User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Office-based pediatricians unprepared for emergencies
Emergency preparedness in U.S. pediatric offices is variable and less than ideal, especially in smaller independent practices, a 15-month multicenter study has found.
Researchers led by Kamal Abulebda, MD, associate professor of clinical pediatrics in the division of pediatric critical care medicine at Indiana University and Riley Hospital for Children in Indianapolis, report that adherence to the 2007 policy statement of the American Academy of Pediatrics on emergency preparedness in pediatric primary care offices was suboptimal across 42 offices in 9 states. They suggest that academic and community partnerships use in-situ simulation exercises to address preparedness gaps and implement standard procedures for contacting emergency medical services.
The group’s findings were published online in Pediatrics. “These data can be used to guide the development of interventions to improve emergency preparedness and care delivery in pediatric offices, Dr. Abulebda and coauthors wrote, noting that theirs is the first multicenter study to directly measure preparedness and quality of care in pediatric offices.
According to the authors, the incidence of a child’s requiring emergent stabilization in an individual office ranges from weekly to monthly, with seizures and respiratory distress being the most common events.
The study was conducted from 2018 to 2020 by 48 national teams participating in in-situ simulated sessions in the ambulatory setting. Office teams, recruited from practices by members of regional academic medical centers, included two patients – a child with respiratory distress and a child with a seizure. Almost 40% were from Indiana.
The scenarios and checklists for the mock exercises were created by content experts in pediatric emergency medicine and critical care using evidence-based guidelines and best practices.
Previous research has relied on self-reported surveys rather than direct measurement to assess adherence to the AAP guidelines, the authors say. In-person surveys assessed adherence to AAP recommendations for emergency preparedness. In-person surveys were, however, used to gauge adherence to AAP recommendations for emergency preparedness.
Findings
The overall mean emergency preparedness score was 74.7% (standard deviation [SD] 12.9), with an unweighted percentage of adherence to checklists calculated for each case. By emergency type, the median asthma case performance score was 63.6% (interquartile range [IQR] 43.2-81.2), and the median seizure case score was 69.2% (IQR 46.2-80.8).
On the measure of essential equipment and supplies, the mean subscore (relating to availability of such items as oxygen sources, suction devices, and epinephrine, for example) was 82.2% (SD 15.1).
As for recommended policies and protocols (e.g., regular assessment of the office, maintenance of emergency equipment and medications) the mean subscore fell to 57.1% (SD 25.6).
In multivariable analyses, offices with a standardized procedure for contacting EMS had a higher rate of activating that service during the simulations.
Independent practices and smaller total staff size were associated with lower preparedness compared with larger groups: beta = –11.89, 95% confidence interval [CI], 19.33-4.45).
Higher annual patient volume and larger total staff size were slightly associated with higher scores (beta = .001, 95% CI, .00-001, P = .017; and beta = .51, 95% CI, .19-.83, P = .002, respectively).
Affiliation with an academic medical center and the presence of learners were not associated with higher scores. And in multivariable regression, a higher annual patient volume lost its significant association with greater preparedness.
So why the lag in preparedness despite the long-standing AAP recommendations? “It’s most likely due to the rare occurrence of these emergencies in the office setting, in addition to most offices’ dependence on EMS when they encounter pediatric emergencies in their setting,” Dr. Abulebda said in an interview. “A 2018 study published by Yuknis and associates demonstrated that the average time from EMS notification to arrival on scene was just 6 minutes.”
In other study findings, 82% of offices did not have an infant bag valve mask and would therefore need to wait for EMS to administer lifesaving ventilation. “This highlights the need to have this equipment available and maintain the skills necessary to care for patients in respiratory distress, the most common emergency encountered in the office setting,” Dr. Abulebda and associates wrote.
A cardiac arrest board is another example of a potentially lifesaving piece of equipment that was not available in the majority of offices, likely because of the rarity of this event in the office setting, but lack of this item may result in poor cardiopulmonary resuscitation quality before the arrival of EMS.
In an accompanying editorial, Jesse Hackell, MD, a pediatrician at Boston Children’s Health Physicians and New York Medical College in Pomona, N.Y., noted that data from 2 decades ago suggested that many pediatric offices saw multiple children requiring emergency intervention each week. More recent figures, however, indicate the situation has evolved, with fewer than 1% of current pediatric EMS transports originating from the office setting.
Dr. Hackell agrees that implementation of AAP recommendations has been far from universal and cites the cost of equipment and supplies as well as a lack of access to training and evaluation as significant barriers to implementation. “In addition, the infrequent occurrence of these emergencies makes maintenance of resuscitation skills even more difficult without frequent practice,” he wrote.
Further complicating the issue, preparedness needs vary with practice location, the response time of local EMS, and proximity to an emergency department. “Pediatric offices in more rural areas, which are farther from these services, will require more equipment and more skills to provide optimal emergency care to children living in these underresourced areas,” he wrote.
He called for equitable distribution of preparedness training, equipment, and staffing, with guidance designed to meet patient needs and ensure optimal outcomes. “In discussion of recommendations, one should consider the likely conditions requiring this response, availability of resources beyond the pediatric office, and ongoing training and support needed to maintain provider skills at the level needed for a successful response to any pediatric emergency,” Dr. Hackell wrote.
This study was supported by grants from Indiana University Health Values and the RBaby Foundation. One study coauthor is a board observer of a medical device company. No other authors disclosed financial relationships relevant to this work. Dr. Hackell has disclosed having no competing interests.
Emergency preparedness in U.S. pediatric offices is variable and less than ideal, especially in smaller independent practices, a 15-month multicenter study has found.
Researchers led by Kamal Abulebda, MD, associate professor of clinical pediatrics in the division of pediatric critical care medicine at Indiana University and Riley Hospital for Children in Indianapolis, report that adherence to the 2007 policy statement of the American Academy of Pediatrics on emergency preparedness in pediatric primary care offices was suboptimal across 42 offices in 9 states. They suggest that academic and community partnerships use in-situ simulation exercises to address preparedness gaps and implement standard procedures for contacting emergency medical services.
The group’s findings were published online in Pediatrics. “These data can be used to guide the development of interventions to improve emergency preparedness and care delivery in pediatric offices, Dr. Abulebda and coauthors wrote, noting that theirs is the first multicenter study to directly measure preparedness and quality of care in pediatric offices.
According to the authors, the incidence of a child’s requiring emergent stabilization in an individual office ranges from weekly to monthly, with seizures and respiratory distress being the most common events.
The study was conducted from 2018 to 2020 by 48 national teams participating in in-situ simulated sessions in the ambulatory setting. Office teams, recruited from practices by members of regional academic medical centers, included two patients – a child with respiratory distress and a child with a seizure. Almost 40% were from Indiana.
The scenarios and checklists for the mock exercises were created by content experts in pediatric emergency medicine and critical care using evidence-based guidelines and best practices.
Previous research has relied on self-reported surveys rather than direct measurement to assess adherence to the AAP guidelines, the authors say. In-person surveys assessed adherence to AAP recommendations for emergency preparedness. In-person surveys were, however, used to gauge adherence to AAP recommendations for emergency preparedness.
Findings
The overall mean emergency preparedness score was 74.7% (standard deviation [SD] 12.9), with an unweighted percentage of adherence to checklists calculated for each case. By emergency type, the median asthma case performance score was 63.6% (interquartile range [IQR] 43.2-81.2), and the median seizure case score was 69.2% (IQR 46.2-80.8).
On the measure of essential equipment and supplies, the mean subscore (relating to availability of such items as oxygen sources, suction devices, and epinephrine, for example) was 82.2% (SD 15.1).
As for recommended policies and protocols (e.g., regular assessment of the office, maintenance of emergency equipment and medications) the mean subscore fell to 57.1% (SD 25.6).
In multivariable analyses, offices with a standardized procedure for contacting EMS had a higher rate of activating that service during the simulations.
Independent practices and smaller total staff size were associated with lower preparedness compared with larger groups: beta = –11.89, 95% confidence interval [CI], 19.33-4.45).
Higher annual patient volume and larger total staff size were slightly associated with higher scores (beta = .001, 95% CI, .00-001, P = .017; and beta = .51, 95% CI, .19-.83, P = .002, respectively).
Affiliation with an academic medical center and the presence of learners were not associated with higher scores. And in multivariable regression, a higher annual patient volume lost its significant association with greater preparedness.
So why the lag in preparedness despite the long-standing AAP recommendations? “It’s most likely due to the rare occurrence of these emergencies in the office setting, in addition to most offices’ dependence on EMS when they encounter pediatric emergencies in their setting,” Dr. Abulebda said in an interview. “A 2018 study published by Yuknis and associates demonstrated that the average time from EMS notification to arrival on scene was just 6 minutes.”
In other study findings, 82% of offices did not have an infant bag valve mask and would therefore need to wait for EMS to administer lifesaving ventilation. “This highlights the need to have this equipment available and maintain the skills necessary to care for patients in respiratory distress, the most common emergency encountered in the office setting,” Dr. Abulebda and associates wrote.
A cardiac arrest board is another example of a potentially lifesaving piece of equipment that was not available in the majority of offices, likely because of the rarity of this event in the office setting, but lack of this item may result in poor cardiopulmonary resuscitation quality before the arrival of EMS.
In an accompanying editorial, Jesse Hackell, MD, a pediatrician at Boston Children’s Health Physicians and New York Medical College in Pomona, N.Y., noted that data from 2 decades ago suggested that many pediatric offices saw multiple children requiring emergency intervention each week. More recent figures, however, indicate the situation has evolved, with fewer than 1% of current pediatric EMS transports originating from the office setting.
Dr. Hackell agrees that implementation of AAP recommendations has been far from universal and cites the cost of equipment and supplies as well as a lack of access to training and evaluation as significant barriers to implementation. “In addition, the infrequent occurrence of these emergencies makes maintenance of resuscitation skills even more difficult without frequent practice,” he wrote.
Further complicating the issue, preparedness needs vary with practice location, the response time of local EMS, and proximity to an emergency department. “Pediatric offices in more rural areas, which are farther from these services, will require more equipment and more skills to provide optimal emergency care to children living in these underresourced areas,” he wrote.
He called for equitable distribution of preparedness training, equipment, and staffing, with guidance designed to meet patient needs and ensure optimal outcomes. “In discussion of recommendations, one should consider the likely conditions requiring this response, availability of resources beyond the pediatric office, and ongoing training and support needed to maintain provider skills at the level needed for a successful response to any pediatric emergency,” Dr. Hackell wrote.
This study was supported by grants from Indiana University Health Values and the RBaby Foundation. One study coauthor is a board observer of a medical device company. No other authors disclosed financial relationships relevant to this work. Dr. Hackell has disclosed having no competing interests.
Emergency preparedness in U.S. pediatric offices is variable and less than ideal, especially in smaller independent practices, a 15-month multicenter study has found.
Researchers led by Kamal Abulebda, MD, associate professor of clinical pediatrics in the division of pediatric critical care medicine at Indiana University and Riley Hospital for Children in Indianapolis, report that adherence to the 2007 policy statement of the American Academy of Pediatrics on emergency preparedness in pediatric primary care offices was suboptimal across 42 offices in 9 states. They suggest that academic and community partnerships use in-situ simulation exercises to address preparedness gaps and implement standard procedures for contacting emergency medical services.
The group’s findings were published online in Pediatrics. “These data can be used to guide the development of interventions to improve emergency preparedness and care delivery in pediatric offices, Dr. Abulebda and coauthors wrote, noting that theirs is the first multicenter study to directly measure preparedness and quality of care in pediatric offices.
According to the authors, the incidence of a child’s requiring emergent stabilization in an individual office ranges from weekly to monthly, with seizures and respiratory distress being the most common events.
The study was conducted from 2018 to 2020 by 48 national teams participating in in-situ simulated sessions in the ambulatory setting. Office teams, recruited from practices by members of regional academic medical centers, included two patients – a child with respiratory distress and a child with a seizure. Almost 40% were from Indiana.
The scenarios and checklists for the mock exercises were created by content experts in pediatric emergency medicine and critical care using evidence-based guidelines and best practices.
Previous research has relied on self-reported surveys rather than direct measurement to assess adherence to the AAP guidelines, the authors say. In-person surveys assessed adherence to AAP recommendations for emergency preparedness. In-person surveys were, however, used to gauge adherence to AAP recommendations for emergency preparedness.
Findings
The overall mean emergency preparedness score was 74.7% (standard deviation [SD] 12.9), with an unweighted percentage of adherence to checklists calculated for each case. By emergency type, the median asthma case performance score was 63.6% (interquartile range [IQR] 43.2-81.2), and the median seizure case score was 69.2% (IQR 46.2-80.8).
On the measure of essential equipment and supplies, the mean subscore (relating to availability of such items as oxygen sources, suction devices, and epinephrine, for example) was 82.2% (SD 15.1).
As for recommended policies and protocols (e.g., regular assessment of the office, maintenance of emergency equipment and medications) the mean subscore fell to 57.1% (SD 25.6).
In multivariable analyses, offices with a standardized procedure for contacting EMS had a higher rate of activating that service during the simulations.
Independent practices and smaller total staff size were associated with lower preparedness compared with larger groups: beta = –11.89, 95% confidence interval [CI], 19.33-4.45).
Higher annual patient volume and larger total staff size were slightly associated with higher scores (beta = .001, 95% CI, .00-001, P = .017; and beta = .51, 95% CI, .19-.83, P = .002, respectively).
Affiliation with an academic medical center and the presence of learners were not associated with higher scores. And in multivariable regression, a higher annual patient volume lost its significant association with greater preparedness.
So why the lag in preparedness despite the long-standing AAP recommendations? “It’s most likely due to the rare occurrence of these emergencies in the office setting, in addition to most offices’ dependence on EMS when they encounter pediatric emergencies in their setting,” Dr. Abulebda said in an interview. “A 2018 study published by Yuknis and associates demonstrated that the average time from EMS notification to arrival on scene was just 6 minutes.”
In other study findings, 82% of offices did not have an infant bag valve mask and would therefore need to wait for EMS to administer lifesaving ventilation. “This highlights the need to have this equipment available and maintain the skills necessary to care for patients in respiratory distress, the most common emergency encountered in the office setting,” Dr. Abulebda and associates wrote.
A cardiac arrest board is another example of a potentially lifesaving piece of equipment that was not available in the majority of offices, likely because of the rarity of this event in the office setting, but lack of this item may result in poor cardiopulmonary resuscitation quality before the arrival of EMS.
In an accompanying editorial, Jesse Hackell, MD, a pediatrician at Boston Children’s Health Physicians and New York Medical College in Pomona, N.Y., noted that data from 2 decades ago suggested that many pediatric offices saw multiple children requiring emergency intervention each week. More recent figures, however, indicate the situation has evolved, with fewer than 1% of current pediatric EMS transports originating from the office setting.
Dr. Hackell agrees that implementation of AAP recommendations has been far from universal and cites the cost of equipment and supplies as well as a lack of access to training and evaluation as significant barriers to implementation. “In addition, the infrequent occurrence of these emergencies makes maintenance of resuscitation skills even more difficult without frequent practice,” he wrote.
Further complicating the issue, preparedness needs vary with practice location, the response time of local EMS, and proximity to an emergency department. “Pediatric offices in more rural areas, which are farther from these services, will require more equipment and more skills to provide optimal emergency care to children living in these underresourced areas,” he wrote.
He called for equitable distribution of preparedness training, equipment, and staffing, with guidance designed to meet patient needs and ensure optimal outcomes. “In discussion of recommendations, one should consider the likely conditions requiring this response, availability of resources beyond the pediatric office, and ongoing training and support needed to maintain provider skills at the level needed for a successful response to any pediatric emergency,” Dr. Hackell wrote.
This study was supported by grants from Indiana University Health Values and the RBaby Foundation. One study coauthor is a board observer of a medical device company. No other authors disclosed financial relationships relevant to this work. Dr. Hackell has disclosed having no competing interests.
FROM PEDIATRICS
COVID-clogged ICUs ‘terrify’ those with chronic or emergency illness
Jessica Gosnell, MD, 41, from Portland, Oregon, lives daily with the knowledge that her rare disease — a form of hereditary angioedema — could cause a sudden, severe swelling in her throat that could require quick intubation and land her in an intensive care unit (ICU) for days.
“I’ve been hospitalized for throat swells three times in the last year,” she said in an interview.
Dr. Gosnell no longer practices medicine because of a combination of illnesses, but lives with her husband, Andrew, and two young children, and said they are all “terrified” she will have to go to the hospital amid a COVID-19 surge that had shrunk the number of available ICU beds to 152 from 780 in Oregon as of Aug. 30. Thirty percent of the beds are in use for patients with COVID-19.
She said her life depends on being near hospitals that have ICUs and having access to highly specialized medications, one of which can cost up to $50,000 for the rescue dose.
Her fear has her “literally living bedbound.” In addition to hereditary angioedema, she has Ehlers-Danlos syndrome, which weakens connective tissue. She wears a cervical collar 24/7 to keep from tearing tissues, as any tissue injury can trigger a swell.
Patients worry there won’t be room
As ICU beds in most states are filling with COVID-19 patients as the Delta variant spreads, fears are rising among people like Dr. Gosnell, who have chronic conditions and diseases with unpredictable emergency visits, who worry that if they need emergency care there won’t be room.
As of Aug. 30, in the United States, 79% of ICU beds nationally were in use, 30% of them for COVID-19 patients, according to the U.S. Department of Health and Human Services.
In individual states, the picture is dire. Alabama has fewer than 10% of its ICU beds open across the entire state. In Florida, 93% of ICU beds are filled, 53% of them with COVID patients. In Louisiana, 87% of beds were already in use, 45% of them with COVID patients, just as category 4 hurricane Ida smashed into the coastline on Aug. 29.
News reports have told of people transported and airlifted as hospitals reach capacity.
In Bellville, Tex., U.S. Army veteran Daniel Wilkinson needed advanced care for gallstone pancreatitis that normally would take 30 minutes to treat, his Bellville doctor, Hasan Kakli, MD, told CBS News.
Mr. Wilkinson’s house was three doors from Bellville Hospital, but the hospital was not equipped to treat the condition. Calls to other hospitals found the same answer: no empty ICU beds. After a 7-hour wait on a stretcher, he was airlifted to a Veterans Affairs hospital in Houston, but it was too late. He died on August 22 at age 46.
Dr. Kakli said, “I’ve never lost a patient with this diagnosis. Ever. I’m scared that the next patient I see is someone that I can’t get to where they need to get to. We are playing musical chairs with 100 people and 10 chairs. When the music stops, what happens?”
Also in Texas in August, Joe Valdez, who was shot six times as an unlucky bystander in a domestic dispute, waited for more than a week for surgery at Ben Taub Hospital in Houston, which was over capacity with COVID patients, the Washington Post reported.
Others with chronic diseases fear needing emergency services or even entering a hospital for regular care with the COVID surge.
Nicole Seefeldt, 44, from Easton, Penn., who had a double-lung transplant in 2016, said that she hasn’t been able to see her lung transplant specialists in Philadelphia — an hour-and-a-half drive — for almost 2 years because of fear of contracting COVID. Before the pandemic, she made the trip almost weekly.
“I protect my lungs like they’re children,” she said.
She relies on her local hospital for care, but has put off some needed care, such as a colonoscopy, and has relied on telemedicine because she wants to limit her hospital exposure.
Ms. Seefeldt now faces an eventual kidney transplant, as her kidney function has been reduced to 20%. In the meantime, she worries she will need emergency care for either her lungs or kidneys.
“For those of us who are chronically ill or disabled, what if we have an emergency that is not COVID-related? Are we going to be able to get a bed? Are we going to be able to get treatment? It’s not just COVID patients who come to the [emergency room],” she said.
A pandemic problem
Paul E. Casey, MD, MBA, chief medical officer at Rush University Medical Center in Chicago, said that high vaccination rates in Chicago have helped Rush continue to accommodate both non-COVID and COVID patients in the emergency department.
Though the hospital treated a large volume of COVID patients, “The vast majority of people we see and did see through the pandemic were non-COVID patents,” he said.
Dr. Casey said that in the first wave the hospital noticed a concerning drop in patients coming in for strokes and heart attacks — “things we knew hadn’t gone away.”
And the data backs it up. Over the course of the pandemic, the Centers for Disease Control and Prevention’s National Health Interview Survey found that the percentage of Americans who reported seeing a doctor or health professional fell from 85% at the end of 2019 to about 80% in the first three months of 2021. The survey did not differentiate between in-person visits and telehealth appointments.
Medical practices and patients themselves postponed elective procedures and delayed routine visits during the early months of the crisis.
Patients also reported staying away from hospitals’ emergency departments throughout the pandemic. At the end of 2019, 22% of respondents reported visiting an emergency department in the past year. That dropped to 17% by the end of 2020, and was at 17.7% in the first 3 months of 2021.
Dr. Casey said that, in his hospital’s case, clear messaging became very important to assure patients it was safe to come back. And the message is still critical.
“We want to be loud and clear that patients should continue to seek care for those conditions,” Dr. Casey said. “Deferring healthcare only comes with the long-term sequelae of disease left untreated so we want people to be as proactive in seeking care as they always would be.”
In some cases, fears of entering emergency rooms because of excess patients and risk for infection are keeping some patients from seeking necessary care for minor injuries.
Jim Rickert, MD, an orthopedic surgeon with Indiana University Health in Bloomington, said that some of his patients have expressed fears of coming into the hospital for fractures.
Some patients, particularly elderly patients, he said, are having falls and fractures and wearing slings or braces at home rather than going into the hospital for injuries that need immediate attention.
Bones start healing incorrectly, Dr. Rickert said, and the correction becomes much more difficult.
Plea for vaccinations
Dr. Gosnell made a plea posted on her neighborhood news forum for people to get COVID vaccinations.
“It seems to me it’s easy for other people who are not in bodies like mine to take health for granted,” she said. “But there are a lot of us who live in very fragile bodies and our entire life is at the intersection of us and getting healthcare treatment. Small complications to getting treatment can be life altering.”
Dr. Gosnell, Ms. Seefeldt, Dr. Casey, and Dr. Rickert reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Jessica Gosnell, MD, 41, from Portland, Oregon, lives daily with the knowledge that her rare disease — a form of hereditary angioedema — could cause a sudden, severe swelling in her throat that could require quick intubation and land her in an intensive care unit (ICU) for days.
“I’ve been hospitalized for throat swells three times in the last year,” she said in an interview.
Dr. Gosnell no longer practices medicine because of a combination of illnesses, but lives with her husband, Andrew, and two young children, and said they are all “terrified” she will have to go to the hospital amid a COVID-19 surge that had shrunk the number of available ICU beds to 152 from 780 in Oregon as of Aug. 30. Thirty percent of the beds are in use for patients with COVID-19.
She said her life depends on being near hospitals that have ICUs and having access to highly specialized medications, one of which can cost up to $50,000 for the rescue dose.
Her fear has her “literally living bedbound.” In addition to hereditary angioedema, she has Ehlers-Danlos syndrome, which weakens connective tissue. She wears a cervical collar 24/7 to keep from tearing tissues, as any tissue injury can trigger a swell.
Patients worry there won’t be room
As ICU beds in most states are filling with COVID-19 patients as the Delta variant spreads, fears are rising among people like Dr. Gosnell, who have chronic conditions and diseases with unpredictable emergency visits, who worry that if they need emergency care there won’t be room.
As of Aug. 30, in the United States, 79% of ICU beds nationally were in use, 30% of them for COVID-19 patients, according to the U.S. Department of Health and Human Services.
In individual states, the picture is dire. Alabama has fewer than 10% of its ICU beds open across the entire state. In Florida, 93% of ICU beds are filled, 53% of them with COVID patients. In Louisiana, 87% of beds were already in use, 45% of them with COVID patients, just as category 4 hurricane Ida smashed into the coastline on Aug. 29.
News reports have told of people transported and airlifted as hospitals reach capacity.
In Bellville, Tex., U.S. Army veteran Daniel Wilkinson needed advanced care for gallstone pancreatitis that normally would take 30 minutes to treat, his Bellville doctor, Hasan Kakli, MD, told CBS News.
Mr. Wilkinson’s house was three doors from Bellville Hospital, but the hospital was not equipped to treat the condition. Calls to other hospitals found the same answer: no empty ICU beds. After a 7-hour wait on a stretcher, he was airlifted to a Veterans Affairs hospital in Houston, but it was too late. He died on August 22 at age 46.
Dr. Kakli said, “I’ve never lost a patient with this diagnosis. Ever. I’m scared that the next patient I see is someone that I can’t get to where they need to get to. We are playing musical chairs with 100 people and 10 chairs. When the music stops, what happens?”
Also in Texas in August, Joe Valdez, who was shot six times as an unlucky bystander in a domestic dispute, waited for more than a week for surgery at Ben Taub Hospital in Houston, which was over capacity with COVID patients, the Washington Post reported.
Others with chronic diseases fear needing emergency services or even entering a hospital for regular care with the COVID surge.
Nicole Seefeldt, 44, from Easton, Penn., who had a double-lung transplant in 2016, said that she hasn’t been able to see her lung transplant specialists in Philadelphia — an hour-and-a-half drive — for almost 2 years because of fear of contracting COVID. Before the pandemic, she made the trip almost weekly.
“I protect my lungs like they’re children,” she said.
She relies on her local hospital for care, but has put off some needed care, such as a colonoscopy, and has relied on telemedicine because she wants to limit her hospital exposure.
Ms. Seefeldt now faces an eventual kidney transplant, as her kidney function has been reduced to 20%. In the meantime, she worries she will need emergency care for either her lungs or kidneys.
“For those of us who are chronically ill or disabled, what if we have an emergency that is not COVID-related? Are we going to be able to get a bed? Are we going to be able to get treatment? It’s not just COVID patients who come to the [emergency room],” she said.
A pandemic problem
Paul E. Casey, MD, MBA, chief medical officer at Rush University Medical Center in Chicago, said that high vaccination rates in Chicago have helped Rush continue to accommodate both non-COVID and COVID patients in the emergency department.
Though the hospital treated a large volume of COVID patients, “The vast majority of people we see and did see through the pandemic were non-COVID patents,” he said.
Dr. Casey said that in the first wave the hospital noticed a concerning drop in patients coming in for strokes and heart attacks — “things we knew hadn’t gone away.”
And the data backs it up. Over the course of the pandemic, the Centers for Disease Control and Prevention’s National Health Interview Survey found that the percentage of Americans who reported seeing a doctor or health professional fell from 85% at the end of 2019 to about 80% in the first three months of 2021. The survey did not differentiate between in-person visits and telehealth appointments.
Medical practices and patients themselves postponed elective procedures and delayed routine visits during the early months of the crisis.
Patients also reported staying away from hospitals’ emergency departments throughout the pandemic. At the end of 2019, 22% of respondents reported visiting an emergency department in the past year. That dropped to 17% by the end of 2020, and was at 17.7% in the first 3 months of 2021.
Dr. Casey said that, in his hospital’s case, clear messaging became very important to assure patients it was safe to come back. And the message is still critical.
“We want to be loud and clear that patients should continue to seek care for those conditions,” Dr. Casey said. “Deferring healthcare only comes with the long-term sequelae of disease left untreated so we want people to be as proactive in seeking care as they always would be.”
In some cases, fears of entering emergency rooms because of excess patients and risk for infection are keeping some patients from seeking necessary care for minor injuries.
Jim Rickert, MD, an orthopedic surgeon with Indiana University Health in Bloomington, said that some of his patients have expressed fears of coming into the hospital for fractures.
Some patients, particularly elderly patients, he said, are having falls and fractures and wearing slings or braces at home rather than going into the hospital for injuries that need immediate attention.
Bones start healing incorrectly, Dr. Rickert said, and the correction becomes much more difficult.
Plea for vaccinations
Dr. Gosnell made a plea posted on her neighborhood news forum for people to get COVID vaccinations.
“It seems to me it’s easy for other people who are not in bodies like mine to take health for granted,” she said. “But there are a lot of us who live in very fragile bodies and our entire life is at the intersection of us and getting healthcare treatment. Small complications to getting treatment can be life altering.”
Dr. Gosnell, Ms. Seefeldt, Dr. Casey, and Dr. Rickert reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Jessica Gosnell, MD, 41, from Portland, Oregon, lives daily with the knowledge that her rare disease — a form of hereditary angioedema — could cause a sudden, severe swelling in her throat that could require quick intubation and land her in an intensive care unit (ICU) for days.
“I’ve been hospitalized for throat swells three times in the last year,” she said in an interview.
Dr. Gosnell no longer practices medicine because of a combination of illnesses, but lives with her husband, Andrew, and two young children, and said they are all “terrified” she will have to go to the hospital amid a COVID-19 surge that had shrunk the number of available ICU beds to 152 from 780 in Oregon as of Aug. 30. Thirty percent of the beds are in use for patients with COVID-19.
She said her life depends on being near hospitals that have ICUs and having access to highly specialized medications, one of which can cost up to $50,000 for the rescue dose.
Her fear has her “literally living bedbound.” In addition to hereditary angioedema, she has Ehlers-Danlos syndrome, which weakens connective tissue. She wears a cervical collar 24/7 to keep from tearing tissues, as any tissue injury can trigger a swell.
Patients worry there won’t be room
As ICU beds in most states are filling with COVID-19 patients as the Delta variant spreads, fears are rising among people like Dr. Gosnell, who have chronic conditions and diseases with unpredictable emergency visits, who worry that if they need emergency care there won’t be room.
As of Aug. 30, in the United States, 79% of ICU beds nationally were in use, 30% of them for COVID-19 patients, according to the U.S. Department of Health and Human Services.
In individual states, the picture is dire. Alabama has fewer than 10% of its ICU beds open across the entire state. In Florida, 93% of ICU beds are filled, 53% of them with COVID patients. In Louisiana, 87% of beds were already in use, 45% of them with COVID patients, just as category 4 hurricane Ida smashed into the coastline on Aug. 29.
News reports have told of people transported and airlifted as hospitals reach capacity.
In Bellville, Tex., U.S. Army veteran Daniel Wilkinson needed advanced care for gallstone pancreatitis that normally would take 30 minutes to treat, his Bellville doctor, Hasan Kakli, MD, told CBS News.
Mr. Wilkinson’s house was three doors from Bellville Hospital, but the hospital was not equipped to treat the condition. Calls to other hospitals found the same answer: no empty ICU beds. After a 7-hour wait on a stretcher, he was airlifted to a Veterans Affairs hospital in Houston, but it was too late. He died on August 22 at age 46.
Dr. Kakli said, “I’ve never lost a patient with this diagnosis. Ever. I’m scared that the next patient I see is someone that I can’t get to where they need to get to. We are playing musical chairs with 100 people and 10 chairs. When the music stops, what happens?”
Also in Texas in August, Joe Valdez, who was shot six times as an unlucky bystander in a domestic dispute, waited for more than a week for surgery at Ben Taub Hospital in Houston, which was over capacity with COVID patients, the Washington Post reported.
Others with chronic diseases fear needing emergency services or even entering a hospital for regular care with the COVID surge.
Nicole Seefeldt, 44, from Easton, Penn., who had a double-lung transplant in 2016, said that she hasn’t been able to see her lung transplant specialists in Philadelphia — an hour-and-a-half drive — for almost 2 years because of fear of contracting COVID. Before the pandemic, she made the trip almost weekly.
“I protect my lungs like they’re children,” she said.
She relies on her local hospital for care, but has put off some needed care, such as a colonoscopy, and has relied on telemedicine because she wants to limit her hospital exposure.
Ms. Seefeldt now faces an eventual kidney transplant, as her kidney function has been reduced to 20%. In the meantime, she worries she will need emergency care for either her lungs or kidneys.
“For those of us who are chronically ill or disabled, what if we have an emergency that is not COVID-related? Are we going to be able to get a bed? Are we going to be able to get treatment? It’s not just COVID patients who come to the [emergency room],” she said.
A pandemic problem
Paul E. Casey, MD, MBA, chief medical officer at Rush University Medical Center in Chicago, said that high vaccination rates in Chicago have helped Rush continue to accommodate both non-COVID and COVID patients in the emergency department.
Though the hospital treated a large volume of COVID patients, “The vast majority of people we see and did see through the pandemic were non-COVID patents,” he said.
Dr. Casey said that in the first wave the hospital noticed a concerning drop in patients coming in for strokes and heart attacks — “things we knew hadn’t gone away.”
And the data backs it up. Over the course of the pandemic, the Centers for Disease Control and Prevention’s National Health Interview Survey found that the percentage of Americans who reported seeing a doctor or health professional fell from 85% at the end of 2019 to about 80% in the first three months of 2021. The survey did not differentiate between in-person visits and telehealth appointments.
Medical practices and patients themselves postponed elective procedures and delayed routine visits during the early months of the crisis.
Patients also reported staying away from hospitals’ emergency departments throughout the pandemic. At the end of 2019, 22% of respondents reported visiting an emergency department in the past year. That dropped to 17% by the end of 2020, and was at 17.7% in the first 3 months of 2021.
Dr. Casey said that, in his hospital’s case, clear messaging became very important to assure patients it was safe to come back. And the message is still critical.
“We want to be loud and clear that patients should continue to seek care for those conditions,” Dr. Casey said. “Deferring healthcare only comes with the long-term sequelae of disease left untreated so we want people to be as proactive in seeking care as they always would be.”
In some cases, fears of entering emergency rooms because of excess patients and risk for infection are keeping some patients from seeking necessary care for minor injuries.
Jim Rickert, MD, an orthopedic surgeon with Indiana University Health in Bloomington, said that some of his patients have expressed fears of coming into the hospital for fractures.
Some patients, particularly elderly patients, he said, are having falls and fractures and wearing slings or braces at home rather than going into the hospital for injuries that need immediate attention.
Bones start healing incorrectly, Dr. Rickert said, and the correction becomes much more difficult.
Plea for vaccinations
Dr. Gosnell made a plea posted on her neighborhood news forum for people to get COVID vaccinations.
“It seems to me it’s easy for other people who are not in bodies like mine to take health for granted,” she said. “But there are a lot of us who live in very fragile bodies and our entire life is at the intersection of us and getting healthcare treatment. Small complications to getting treatment can be life altering.”
Dr. Gosnell, Ms. Seefeldt, Dr. Casey, and Dr. Rickert reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Two swings, two misses with colchicine, Vascepa in COVID-19
The anti-inflammatory agents colchicine and icosapent ethyl (Vascepa; Amarin) failed to provide substantial benefits in separate randomized COVID-19 trials.
Both were reported at the European Society of Cardiology (ESC) Congress 2021.
The open-label ECLA PHRI COLCOVID trial randomized 1,277 hospitalized adults (mean age 62 years) to usual care alone or with colchicine at a loading dose of 1.5 mg for 2 hours followed by 0.5 mg on day 1 and then 0.5 mg twice daily for 14 days or until discharge.
The investigators hypothesized that colchicine, which is widely used to treat gout and other inflammatory conditions, might modulate the hyperinflammatory syndrome, or cytokine storm, associated with COVID-19.
Results showed that the need for mechanical ventilation or death occurred in 25.0% of patients receiving colchicine and 28.8% with usual care (P = .08).
The coprimary endpoint of death at 28 days was also not significantly different between groups (20.5% vs. 22.2%), principal investigator Rafael Diaz, MD, said in a late-breaking COVID-19 trials session at the congress.
Among the secondary outcomes at 28 days, colchicine significantly reduced the incidence of new intubation or death from respiratory failure from 27.0% to 22.3% (hazard ratio, 0.79; 95% confidence interval, 0.63-0.99) but not mortality from respiratory failure (19.5% vs. 16.8%).
The only important adverse effect was severe diarrhea, which was reported in 11.3% of the colchicine group vs. 4.5% in the control group, said Dr. Diaz, director of Estudios Clínicos Latinoamérica (ECLA), Rosario, Argentina.
The results are consistent with those from the massive RECOVERY trial, which earlier this year stopped enrollment in the colchicine arm for lack of efficacy in patients hospitalized with COVID-19, and COLCORONA, which missed its primary endpoint using colchicine among nonhospitalized adults with COVID-19.
Session chair and COLCORONA principal investigator Jean-Claude Tardif, MD, pointed out that, as clinicians, it’s fairly uncommon to combine systemic steroids with colchicine, which was the case in 92% of patients in ECLA PHRI COLCOVID.
“I think it is an inherent limitation of testing colchicine on top of steroids,” said Dr. Tardif, of the Montreal Heart Institute.
Icosapent ethyl in PREPARE-IT
Dr. Diaz returned in the ESC session to present the results of the PREPARE-IT trial, which tested whether icosapent ethyl – at a loading dose of 8 grams (4 capsules) for the first 3 days and 4 g/d on days 4-60 – could reduce the risk for SARS-CoV-2 infection in 2,041 health care and other public workers in Argentina at high risk for infection (mean age 40.5 years).
Vascepa was approved by the Food and Drug Administration in 2012 for the reduction of elevated triglyceride levels, with an added indication in 2019 to reduce cardiovascular (CV) events in people with elevated triglycerides and established CV disease or diabetes with other CV risk factors.
The rationale for using the high-dose prescription eicosapentaenoic acid (EPA) preparation includes its anti-inflammatory and antithrombotic effects, and that unsaturated fatty acids, especially EPA, might inactivate the enveloped virus, he explained.
Among 1,712 participants followed for up to 60 days, however, the SARS-CoV-2 infection rate was 7.9% with icosapent ethyl vs. 7.1% with a mineral oil placebo (P = .58).
There were also no significant changes from baseline in the icosapent ethyl and placebo groups for the secondary outcomes of high-sensitivity C-reactive protein (0 vs. 0), triglycerides (median –2 mg/dL vs. 7 mg/dL), or Influenza Patient-Reported Outcome (FLU-PRO) questionnaire scores (median 0.01 vs. 0.03).
The use of a mineral oil placebo has been the subject of controversy in previous fish oil trials, but, Dr. Diaz noted, it did not have a significant proinflammatory effect or cause any excess adverse events.
Overall, adverse events were similar between the active and placebo groups, including atrial fibrillation (none), major bleeding (none), minor bleeding (7 events vs. 10 events), gastrointestinal symptoms (6.8% vs. 7.0%), and diarrhea (8.6% vs. 7.7%).
Although it missed the primary endpoint, Dr. Diaz said, “this is the first large, randomized blinded trial to demonstrate excellent safety and tolerability of an 8-gram-per-day loading dose of icosapent ethyl, opening up the potential for acute use in randomized trials of myocardial infarction, acute coronary syndromes, strokes, and revascularization.”
During a discussion of the results, Dr. Diaz said the Delta variant was not present at the time of the analysis and that the second half of the trial will report on whether icosapent ethyl can reduce the risk for hospitalization or death in participants diagnosed with COVID-19.
ECLA PHRI COLCOVID was supported by the Estudios Clínicos Latinoamérica Population Health Research Institute. PREPARE-IT was supported by Estudios Clínicos Latinoamérica with collaboration from Amarin. Dr. Diaz reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The anti-inflammatory agents colchicine and icosapent ethyl (Vascepa; Amarin) failed to provide substantial benefits in separate randomized COVID-19 trials.
Both were reported at the European Society of Cardiology (ESC) Congress 2021.
The open-label ECLA PHRI COLCOVID trial randomized 1,277 hospitalized adults (mean age 62 years) to usual care alone or with colchicine at a loading dose of 1.5 mg for 2 hours followed by 0.5 mg on day 1 and then 0.5 mg twice daily for 14 days or until discharge.
The investigators hypothesized that colchicine, which is widely used to treat gout and other inflammatory conditions, might modulate the hyperinflammatory syndrome, or cytokine storm, associated with COVID-19.
Results showed that the need for mechanical ventilation or death occurred in 25.0% of patients receiving colchicine and 28.8% with usual care (P = .08).
The coprimary endpoint of death at 28 days was also not significantly different between groups (20.5% vs. 22.2%), principal investigator Rafael Diaz, MD, said in a late-breaking COVID-19 trials session at the congress.
Among the secondary outcomes at 28 days, colchicine significantly reduced the incidence of new intubation or death from respiratory failure from 27.0% to 22.3% (hazard ratio, 0.79; 95% confidence interval, 0.63-0.99) but not mortality from respiratory failure (19.5% vs. 16.8%).
The only important adverse effect was severe diarrhea, which was reported in 11.3% of the colchicine group vs. 4.5% in the control group, said Dr. Diaz, director of Estudios Clínicos Latinoamérica (ECLA), Rosario, Argentina.
The results are consistent with those from the massive RECOVERY trial, which earlier this year stopped enrollment in the colchicine arm for lack of efficacy in patients hospitalized with COVID-19, and COLCORONA, which missed its primary endpoint using colchicine among nonhospitalized adults with COVID-19.
Session chair and COLCORONA principal investigator Jean-Claude Tardif, MD, pointed out that, as clinicians, it’s fairly uncommon to combine systemic steroids with colchicine, which was the case in 92% of patients in ECLA PHRI COLCOVID.
“I think it is an inherent limitation of testing colchicine on top of steroids,” said Dr. Tardif, of the Montreal Heart Institute.
Icosapent ethyl in PREPARE-IT
Dr. Diaz returned in the ESC session to present the results of the PREPARE-IT trial, which tested whether icosapent ethyl – at a loading dose of 8 grams (4 capsules) for the first 3 days and 4 g/d on days 4-60 – could reduce the risk for SARS-CoV-2 infection in 2,041 health care and other public workers in Argentina at high risk for infection (mean age 40.5 years).
Vascepa was approved by the Food and Drug Administration in 2012 for the reduction of elevated triglyceride levels, with an added indication in 2019 to reduce cardiovascular (CV) events in people with elevated triglycerides and established CV disease or diabetes with other CV risk factors.
The rationale for using the high-dose prescription eicosapentaenoic acid (EPA) preparation includes its anti-inflammatory and antithrombotic effects, and that unsaturated fatty acids, especially EPA, might inactivate the enveloped virus, he explained.
Among 1,712 participants followed for up to 60 days, however, the SARS-CoV-2 infection rate was 7.9% with icosapent ethyl vs. 7.1% with a mineral oil placebo (P = .58).
There were also no significant changes from baseline in the icosapent ethyl and placebo groups for the secondary outcomes of high-sensitivity C-reactive protein (0 vs. 0), triglycerides (median –2 mg/dL vs. 7 mg/dL), or Influenza Patient-Reported Outcome (FLU-PRO) questionnaire scores (median 0.01 vs. 0.03).
The use of a mineral oil placebo has been the subject of controversy in previous fish oil trials, but, Dr. Diaz noted, it did not have a significant proinflammatory effect or cause any excess adverse events.
Overall, adverse events were similar between the active and placebo groups, including atrial fibrillation (none), major bleeding (none), minor bleeding (7 events vs. 10 events), gastrointestinal symptoms (6.8% vs. 7.0%), and diarrhea (8.6% vs. 7.7%).
Although it missed the primary endpoint, Dr. Diaz said, “this is the first large, randomized blinded trial to demonstrate excellent safety and tolerability of an 8-gram-per-day loading dose of icosapent ethyl, opening up the potential for acute use in randomized trials of myocardial infarction, acute coronary syndromes, strokes, and revascularization.”
During a discussion of the results, Dr. Diaz said the Delta variant was not present at the time of the analysis and that the second half of the trial will report on whether icosapent ethyl can reduce the risk for hospitalization or death in participants diagnosed with COVID-19.
ECLA PHRI COLCOVID was supported by the Estudios Clínicos Latinoamérica Population Health Research Institute. PREPARE-IT was supported by Estudios Clínicos Latinoamérica with collaboration from Amarin. Dr. Diaz reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The anti-inflammatory agents colchicine and icosapent ethyl (Vascepa; Amarin) failed to provide substantial benefits in separate randomized COVID-19 trials.
Both were reported at the European Society of Cardiology (ESC) Congress 2021.
The open-label ECLA PHRI COLCOVID trial randomized 1,277 hospitalized adults (mean age 62 years) to usual care alone or with colchicine at a loading dose of 1.5 mg for 2 hours followed by 0.5 mg on day 1 and then 0.5 mg twice daily for 14 days or until discharge.
The investigators hypothesized that colchicine, which is widely used to treat gout and other inflammatory conditions, might modulate the hyperinflammatory syndrome, or cytokine storm, associated with COVID-19.
Results showed that the need for mechanical ventilation or death occurred in 25.0% of patients receiving colchicine and 28.8% with usual care (P = .08).
The coprimary endpoint of death at 28 days was also not significantly different between groups (20.5% vs. 22.2%), principal investigator Rafael Diaz, MD, said in a late-breaking COVID-19 trials session at the congress.
Among the secondary outcomes at 28 days, colchicine significantly reduced the incidence of new intubation or death from respiratory failure from 27.0% to 22.3% (hazard ratio, 0.79; 95% confidence interval, 0.63-0.99) but not mortality from respiratory failure (19.5% vs. 16.8%).
The only important adverse effect was severe diarrhea, which was reported in 11.3% of the colchicine group vs. 4.5% in the control group, said Dr. Diaz, director of Estudios Clínicos Latinoamérica (ECLA), Rosario, Argentina.
The results are consistent with those from the massive RECOVERY trial, which earlier this year stopped enrollment in the colchicine arm for lack of efficacy in patients hospitalized with COVID-19, and COLCORONA, which missed its primary endpoint using colchicine among nonhospitalized adults with COVID-19.
Session chair and COLCORONA principal investigator Jean-Claude Tardif, MD, pointed out that, as clinicians, it’s fairly uncommon to combine systemic steroids with colchicine, which was the case in 92% of patients in ECLA PHRI COLCOVID.
“I think it is an inherent limitation of testing colchicine on top of steroids,” said Dr. Tardif, of the Montreal Heart Institute.
Icosapent ethyl in PREPARE-IT
Dr. Diaz returned in the ESC session to present the results of the PREPARE-IT trial, which tested whether icosapent ethyl – at a loading dose of 8 grams (4 capsules) for the first 3 days and 4 g/d on days 4-60 – could reduce the risk for SARS-CoV-2 infection in 2,041 health care and other public workers in Argentina at high risk for infection (mean age 40.5 years).
Vascepa was approved by the Food and Drug Administration in 2012 for the reduction of elevated triglyceride levels, with an added indication in 2019 to reduce cardiovascular (CV) events in people with elevated triglycerides and established CV disease or diabetes with other CV risk factors.
The rationale for using the high-dose prescription eicosapentaenoic acid (EPA) preparation includes its anti-inflammatory and antithrombotic effects, and that unsaturated fatty acids, especially EPA, might inactivate the enveloped virus, he explained.
Among 1,712 participants followed for up to 60 days, however, the SARS-CoV-2 infection rate was 7.9% with icosapent ethyl vs. 7.1% with a mineral oil placebo (P = .58).
There were also no significant changes from baseline in the icosapent ethyl and placebo groups for the secondary outcomes of high-sensitivity C-reactive protein (0 vs. 0), triglycerides (median –2 mg/dL vs. 7 mg/dL), or Influenza Patient-Reported Outcome (FLU-PRO) questionnaire scores (median 0.01 vs. 0.03).
The use of a mineral oil placebo has been the subject of controversy in previous fish oil trials, but, Dr. Diaz noted, it did not have a significant proinflammatory effect or cause any excess adverse events.
Overall, adverse events were similar between the active and placebo groups, including atrial fibrillation (none), major bleeding (none), minor bleeding (7 events vs. 10 events), gastrointestinal symptoms (6.8% vs. 7.0%), and diarrhea (8.6% vs. 7.7%).
Although it missed the primary endpoint, Dr. Diaz said, “this is the first large, randomized blinded trial to demonstrate excellent safety and tolerability of an 8-gram-per-day loading dose of icosapent ethyl, opening up the potential for acute use in randomized trials of myocardial infarction, acute coronary syndromes, strokes, and revascularization.”
During a discussion of the results, Dr. Diaz said the Delta variant was not present at the time of the analysis and that the second half of the trial will report on whether icosapent ethyl can reduce the risk for hospitalization or death in participants diagnosed with COVID-19.
ECLA PHRI COLCOVID was supported by the Estudios Clínicos Latinoamérica Population Health Research Institute. PREPARE-IT was supported by Estudios Clínicos Latinoamérica with collaboration from Amarin. Dr. Diaz reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
‘Deeper dive’ into opioid overdose deaths during COVID pandemic
Opioid overdose deaths were significantly higher during 2020, but occurrences were not homogeneous across nine states. Male deaths were higher than in the 2 previous years in two states, according to a new, granular examination of data collected by researchers at the Massachusetts General Hospital (Mass General), Boston.
The analysis also showed that synthetic opioids such as fentanyl played an outsized role in most of the states that were reviewed. Additional drugs of abuse found in decedents, such as cocaine and psychostimulants, were more prevalent in some states than in others.
The Centers for Disease Control and Prevention used provisional death data in its recent report. It found that opioid-related deaths substantially rose in 2020 and that synthetic opioids were a primary driver.
The current Mass General analysis provides a more timely and detailed dive, senior author Mohammad Jalali, PhD, who is a senior scientist at Mass General’s Institute for Technology Assessment, told this news organization.
The findings, which have not yet been peer reviewed, were published in MedRxiv.
Shifting sands of opioid use disorder
to analyze and project trends and also to be better prepared to address the shifting sands of opioid use disorder in the United States.
They attempted to collect data on confirmed opioid overdose deaths from all 50 states and Washington, D.C. to assess what might have changed during the COVID-19 pandemic. Only nine states provided enough data for the analysis, which has been submitted to a peer reviewed publication.
These states were Alaska, Connecticut, Indiana, Massachusetts, North Carolina, Rhode Island, Colorado, Utah, and Wyoming.
“Drug overdose data are collected and reported more slowly than COVID-19 data,” Dr. Jalali said in a press release. The data reflected a lag time of about 4 to 8 months in Massachusetts and North Carolina to more than a year in Maryland and Ohio, he noted.
The reporting lag “has clouded the understanding of the effects of the COVID-19 pandemic on opioid-related overdose deaths,” said Dr. Jalali.
Commenting on the findings, Brandon Marshall, PhD, associate professor of epidemiology at Brown University, Providence, R.I, said that “the overall pattern of what’s being reported here is not surprising,” given the national trends seen in the CDC data.
“This paper adds a deeper dive into some of the sociodemographic trends that we’re starting to observe in specific states,” Dr. Marshall said.
Also commenting for this news organization, Brian Fuehrlein, MD, PhD, director of the psychiatric emergency department at the VA Connecticut Healthcare System in West Haven, Connecticut, noted that the current study “highlights things that we are currently seeing at VA Connecticut.”
Decrease in heroin, rise in fentanyl
The investigators found a significant reduction in overdose deaths that involved heroin in Alaska, Connecticut, Indiana, Massachusetts, North Carolina, and Rhode Island. That was a new trend for Alaska, Indiana, and Rhode Island, although with only 3 years of data, it’s hard to say whether it will continue, Dr. Jalali noted.
The decrease in heroin involvement seemed to continue a trend previously observed in Colorado, Connecticut, Massachusetts, and North Carolina.
In Connecticut, heroin was involved in 36% of deaths in 2018, 30% in 2019, and 16% in 2020, according to the study.
“We have begun seeing more and more heroin-negative, fentanyl-positive drug screens,” said Dr. Fuehrlein, who is also associate professor of psychiatry at Yale University, New Haven, Conn.
“There is a shift from fentanyl being an adulterant to fentanyl being what is sold and used exclusively,” he added.
In 2020, 92% (n = 887) of deaths in Connecticut involved synthetic opioids, continuing a trend. In Alaska, however, synthetic opioids were involved in 60% (44) of deaths, which is a big jump from 23% (9) in 2018.
Synthetic opioids were involved in the largest percentage of overdoses in all of the states studied. The fewest deaths, 17 (49%), occurred in Wyoming.
Cocaine is also increasingly found in addition to other substances in decedents. In Alaska, about 14% of individuals who overdosed in 2020 also had cocaine in their system, which was a jump from 2% in the prior year.
In Colorado, 19% (94) of those who died also had taken cocaine, up from 13% in 2019. Cocaine was also frequently found in those who died in the northeast: 39% (467) of those who died in Massachusetts, 29% (280) in Connecticut, and 47% (109) in Rhode Island.
There was also an increase in psychostimulants found in those who had died in Massachusetts in 2020.
More male overdoses in 2020
Results also showed that, compared to 2019, significantly more men died from overdoses in 2020 in Colorado (61% vs. 70%, P = .017) and Indiana (62% vs. 70%, P = .026).
This finding was unexpected, said Dr. Marshall, who has observed the same phenomenon in Rhode Island. He is the scientific director of PreventOverdoseRI, Rhode Island’s drug overdose surveillance and information dashboard.
Dr. Marshall and his colleagues conducted a study that also found disproportionate increases in overdoses among men. The findings of that study will be published in September.
“We’re still trying to wrap our head around why that is,” he said. He added that a deeper dive into the Rhode Island data showed that the deaths were increased especially among middle-aged men who had been diagnosed with depression and anxiety.
The same patterns were not seen among women in either Dr. Jalali’s study or his own analysis of the Rhode Island data, said Dr. Marshall.
“That suggests the COVID-19 pandemic impacted men who are at risk for overdose in some particularly severe way,” he noted.
Dr. Fuehrlein said he believes a variety of factors have led to an increase in overdose deaths during the pandemic, including the fact that many patients who would normally seek help avoided care or dropped out of treatment because of COVID fears. In addition, other support systems, such as group therapy and Narcotics Anonymous, were unavailable.
The pandemic increased stress, which can lead to worsening substance use, said Dr. Fuehrlein. He also noted that regular opioid suppliers were often not available, which led some to buy from different dealers, “which can lead to overdose if the fentanyl content is different.”
Identifying at-risk individuals
Dr. Jalali and colleagues note that clinicians and policymakers could use the new study to help identify and treat at-risk individuals.
“Practitioners and policy makers can use our findings to help them anticipate which groups of people might be most affected by opioid overdose and which types of policy interventions might be most effective given each state’s unique situation,” said lead study author Gian-Gabriel P. Garcia, PhD, in a press release. At the time of the study, Dr. Garcia was a postdoctoral fellow at Mass General and Harvard Medical School. He is currently an assistant professor at Georgia Tech, Atlanta.
Dr. Marshall pointed out that Dr. Jalali’s study is also relevant for emergency departments.
ED clinicians “are and will be seeing patients coming in who have no idea they were exposed to an opioid, nevermind fentanyl,” he said. ED clinicians can discuss with patients various harm reduction techniques, including the use of naloxone as well as test strips that can detect fentanyl in the drug supply, he added.
“Given the increasing use of fentanyl, which is very dangerous in overdose, clinicians need to be well versed in a harm reduction/overdose prevention approach to patient care,” Dr. Fuehrlein agreed.
A version of this article first appeared on Medscape.com.
Opioid overdose deaths were significantly higher during 2020, but occurrences were not homogeneous across nine states. Male deaths were higher than in the 2 previous years in two states, according to a new, granular examination of data collected by researchers at the Massachusetts General Hospital (Mass General), Boston.
The analysis also showed that synthetic opioids such as fentanyl played an outsized role in most of the states that were reviewed. Additional drugs of abuse found in decedents, such as cocaine and psychostimulants, were more prevalent in some states than in others.
The Centers for Disease Control and Prevention used provisional death data in its recent report. It found that opioid-related deaths substantially rose in 2020 and that synthetic opioids were a primary driver.
The current Mass General analysis provides a more timely and detailed dive, senior author Mohammad Jalali, PhD, who is a senior scientist at Mass General’s Institute for Technology Assessment, told this news organization.
The findings, which have not yet been peer reviewed, were published in MedRxiv.
Shifting sands of opioid use disorder
to analyze and project trends and also to be better prepared to address the shifting sands of opioid use disorder in the United States.
They attempted to collect data on confirmed opioid overdose deaths from all 50 states and Washington, D.C. to assess what might have changed during the COVID-19 pandemic. Only nine states provided enough data for the analysis, which has been submitted to a peer reviewed publication.
These states were Alaska, Connecticut, Indiana, Massachusetts, North Carolina, Rhode Island, Colorado, Utah, and Wyoming.
“Drug overdose data are collected and reported more slowly than COVID-19 data,” Dr. Jalali said in a press release. The data reflected a lag time of about 4 to 8 months in Massachusetts and North Carolina to more than a year in Maryland and Ohio, he noted.
The reporting lag “has clouded the understanding of the effects of the COVID-19 pandemic on opioid-related overdose deaths,” said Dr. Jalali.
Commenting on the findings, Brandon Marshall, PhD, associate professor of epidemiology at Brown University, Providence, R.I, said that “the overall pattern of what’s being reported here is not surprising,” given the national trends seen in the CDC data.
“This paper adds a deeper dive into some of the sociodemographic trends that we’re starting to observe in specific states,” Dr. Marshall said.
Also commenting for this news organization, Brian Fuehrlein, MD, PhD, director of the psychiatric emergency department at the VA Connecticut Healthcare System in West Haven, Connecticut, noted that the current study “highlights things that we are currently seeing at VA Connecticut.”
Decrease in heroin, rise in fentanyl
The investigators found a significant reduction in overdose deaths that involved heroin in Alaska, Connecticut, Indiana, Massachusetts, North Carolina, and Rhode Island. That was a new trend for Alaska, Indiana, and Rhode Island, although with only 3 years of data, it’s hard to say whether it will continue, Dr. Jalali noted.
The decrease in heroin involvement seemed to continue a trend previously observed in Colorado, Connecticut, Massachusetts, and North Carolina.
In Connecticut, heroin was involved in 36% of deaths in 2018, 30% in 2019, and 16% in 2020, according to the study.
“We have begun seeing more and more heroin-negative, fentanyl-positive drug screens,” said Dr. Fuehrlein, who is also associate professor of psychiatry at Yale University, New Haven, Conn.
“There is a shift from fentanyl being an adulterant to fentanyl being what is sold and used exclusively,” he added.
In 2020, 92% (n = 887) of deaths in Connecticut involved synthetic opioids, continuing a trend. In Alaska, however, synthetic opioids were involved in 60% (44) of deaths, which is a big jump from 23% (9) in 2018.
Synthetic opioids were involved in the largest percentage of overdoses in all of the states studied. The fewest deaths, 17 (49%), occurred in Wyoming.
Cocaine is also increasingly found in addition to other substances in decedents. In Alaska, about 14% of individuals who overdosed in 2020 also had cocaine in their system, which was a jump from 2% in the prior year.
In Colorado, 19% (94) of those who died also had taken cocaine, up from 13% in 2019. Cocaine was also frequently found in those who died in the northeast: 39% (467) of those who died in Massachusetts, 29% (280) in Connecticut, and 47% (109) in Rhode Island.
There was also an increase in psychostimulants found in those who had died in Massachusetts in 2020.
More male overdoses in 2020
Results also showed that, compared to 2019, significantly more men died from overdoses in 2020 in Colorado (61% vs. 70%, P = .017) and Indiana (62% vs. 70%, P = .026).
This finding was unexpected, said Dr. Marshall, who has observed the same phenomenon in Rhode Island. He is the scientific director of PreventOverdoseRI, Rhode Island’s drug overdose surveillance and information dashboard.
Dr. Marshall and his colleagues conducted a study that also found disproportionate increases in overdoses among men. The findings of that study will be published in September.
“We’re still trying to wrap our head around why that is,” he said. He added that a deeper dive into the Rhode Island data showed that the deaths were increased especially among middle-aged men who had been diagnosed with depression and anxiety.
The same patterns were not seen among women in either Dr. Jalali’s study or his own analysis of the Rhode Island data, said Dr. Marshall.
“That suggests the COVID-19 pandemic impacted men who are at risk for overdose in some particularly severe way,” he noted.
Dr. Fuehrlein said he believes a variety of factors have led to an increase in overdose deaths during the pandemic, including the fact that many patients who would normally seek help avoided care or dropped out of treatment because of COVID fears. In addition, other support systems, such as group therapy and Narcotics Anonymous, were unavailable.
The pandemic increased stress, which can lead to worsening substance use, said Dr. Fuehrlein. He also noted that regular opioid suppliers were often not available, which led some to buy from different dealers, “which can lead to overdose if the fentanyl content is different.”
Identifying at-risk individuals
Dr. Jalali and colleagues note that clinicians and policymakers could use the new study to help identify and treat at-risk individuals.
“Practitioners and policy makers can use our findings to help them anticipate which groups of people might be most affected by opioid overdose and which types of policy interventions might be most effective given each state’s unique situation,” said lead study author Gian-Gabriel P. Garcia, PhD, in a press release. At the time of the study, Dr. Garcia was a postdoctoral fellow at Mass General and Harvard Medical School. He is currently an assistant professor at Georgia Tech, Atlanta.
Dr. Marshall pointed out that Dr. Jalali’s study is also relevant for emergency departments.
ED clinicians “are and will be seeing patients coming in who have no idea they were exposed to an opioid, nevermind fentanyl,” he said. ED clinicians can discuss with patients various harm reduction techniques, including the use of naloxone as well as test strips that can detect fentanyl in the drug supply, he added.
“Given the increasing use of fentanyl, which is very dangerous in overdose, clinicians need to be well versed in a harm reduction/overdose prevention approach to patient care,” Dr. Fuehrlein agreed.
A version of this article first appeared on Medscape.com.
Opioid overdose deaths were significantly higher during 2020, but occurrences were not homogeneous across nine states. Male deaths were higher than in the 2 previous years in two states, according to a new, granular examination of data collected by researchers at the Massachusetts General Hospital (Mass General), Boston.
The analysis also showed that synthetic opioids such as fentanyl played an outsized role in most of the states that were reviewed. Additional drugs of abuse found in decedents, such as cocaine and psychostimulants, were more prevalent in some states than in others.
The Centers for Disease Control and Prevention used provisional death data in its recent report. It found that opioid-related deaths substantially rose in 2020 and that synthetic opioids were a primary driver.
The current Mass General analysis provides a more timely and detailed dive, senior author Mohammad Jalali, PhD, who is a senior scientist at Mass General’s Institute for Technology Assessment, told this news organization.
The findings, which have not yet been peer reviewed, were published in MedRxiv.
Shifting sands of opioid use disorder
to analyze and project trends and also to be better prepared to address the shifting sands of opioid use disorder in the United States.
They attempted to collect data on confirmed opioid overdose deaths from all 50 states and Washington, D.C. to assess what might have changed during the COVID-19 pandemic. Only nine states provided enough data for the analysis, which has been submitted to a peer reviewed publication.
These states were Alaska, Connecticut, Indiana, Massachusetts, North Carolina, Rhode Island, Colorado, Utah, and Wyoming.
“Drug overdose data are collected and reported more slowly than COVID-19 data,” Dr. Jalali said in a press release. The data reflected a lag time of about 4 to 8 months in Massachusetts and North Carolina to more than a year in Maryland and Ohio, he noted.
The reporting lag “has clouded the understanding of the effects of the COVID-19 pandemic on opioid-related overdose deaths,” said Dr. Jalali.
Commenting on the findings, Brandon Marshall, PhD, associate professor of epidemiology at Brown University, Providence, R.I, said that “the overall pattern of what’s being reported here is not surprising,” given the national trends seen in the CDC data.
“This paper adds a deeper dive into some of the sociodemographic trends that we’re starting to observe in specific states,” Dr. Marshall said.
Also commenting for this news organization, Brian Fuehrlein, MD, PhD, director of the psychiatric emergency department at the VA Connecticut Healthcare System in West Haven, Connecticut, noted that the current study “highlights things that we are currently seeing at VA Connecticut.”
Decrease in heroin, rise in fentanyl
The investigators found a significant reduction in overdose deaths that involved heroin in Alaska, Connecticut, Indiana, Massachusetts, North Carolina, and Rhode Island. That was a new trend for Alaska, Indiana, and Rhode Island, although with only 3 years of data, it’s hard to say whether it will continue, Dr. Jalali noted.
The decrease in heroin involvement seemed to continue a trend previously observed in Colorado, Connecticut, Massachusetts, and North Carolina.
In Connecticut, heroin was involved in 36% of deaths in 2018, 30% in 2019, and 16% in 2020, according to the study.
“We have begun seeing more and more heroin-negative, fentanyl-positive drug screens,” said Dr. Fuehrlein, who is also associate professor of psychiatry at Yale University, New Haven, Conn.
“There is a shift from fentanyl being an adulterant to fentanyl being what is sold and used exclusively,” he added.
In 2020, 92% (n = 887) of deaths in Connecticut involved synthetic opioids, continuing a trend. In Alaska, however, synthetic opioids were involved in 60% (44) of deaths, which is a big jump from 23% (9) in 2018.
Synthetic opioids were involved in the largest percentage of overdoses in all of the states studied. The fewest deaths, 17 (49%), occurred in Wyoming.
Cocaine is also increasingly found in addition to other substances in decedents. In Alaska, about 14% of individuals who overdosed in 2020 also had cocaine in their system, which was a jump from 2% in the prior year.
In Colorado, 19% (94) of those who died also had taken cocaine, up from 13% in 2019. Cocaine was also frequently found in those who died in the northeast: 39% (467) of those who died in Massachusetts, 29% (280) in Connecticut, and 47% (109) in Rhode Island.
There was also an increase in psychostimulants found in those who had died in Massachusetts in 2020.
More male overdoses in 2020
Results also showed that, compared to 2019, significantly more men died from overdoses in 2020 in Colorado (61% vs. 70%, P = .017) and Indiana (62% vs. 70%, P = .026).
This finding was unexpected, said Dr. Marshall, who has observed the same phenomenon in Rhode Island. He is the scientific director of PreventOverdoseRI, Rhode Island’s drug overdose surveillance and information dashboard.
Dr. Marshall and his colleagues conducted a study that also found disproportionate increases in overdoses among men. The findings of that study will be published in September.
“We’re still trying to wrap our head around why that is,” he said. He added that a deeper dive into the Rhode Island data showed that the deaths were increased especially among middle-aged men who had been diagnosed with depression and anxiety.
The same patterns were not seen among women in either Dr. Jalali’s study or his own analysis of the Rhode Island data, said Dr. Marshall.
“That suggests the COVID-19 pandemic impacted men who are at risk for overdose in some particularly severe way,” he noted.
Dr. Fuehrlein said he believes a variety of factors have led to an increase in overdose deaths during the pandemic, including the fact that many patients who would normally seek help avoided care or dropped out of treatment because of COVID fears. In addition, other support systems, such as group therapy and Narcotics Anonymous, were unavailable.
The pandemic increased stress, which can lead to worsening substance use, said Dr. Fuehrlein. He also noted that regular opioid suppliers were often not available, which led some to buy from different dealers, “which can lead to overdose if the fentanyl content is different.”
Identifying at-risk individuals
Dr. Jalali and colleagues note that clinicians and policymakers could use the new study to help identify and treat at-risk individuals.
“Practitioners and policy makers can use our findings to help them anticipate which groups of people might be most affected by opioid overdose and which types of policy interventions might be most effective given each state’s unique situation,” said lead study author Gian-Gabriel P. Garcia, PhD, in a press release. At the time of the study, Dr. Garcia was a postdoctoral fellow at Mass General and Harvard Medical School. He is currently an assistant professor at Georgia Tech, Atlanta.
Dr. Marshall pointed out that Dr. Jalali’s study is also relevant for emergency departments.
ED clinicians “are and will be seeing patients coming in who have no idea they were exposed to an opioid, nevermind fentanyl,” he said. ED clinicians can discuss with patients various harm reduction techniques, including the use of naloxone as well as test strips that can detect fentanyl in the drug supply, he added.
“Given the increasing use of fentanyl, which is very dangerous in overdose, clinicians need to be well versed in a harm reduction/overdose prevention approach to patient care,” Dr. Fuehrlein agreed.
A version of this article first appeared on Medscape.com.
Children and COVID: Weekly cases top 200,000, vaccinations down
Weekly pediatric cases of COVID-19 exceeded 200,000 for just the second time during the pandemic, while new vaccinations in children continued to decline.
The weekly count has now increased for 9 consecutive weeks, during which time it has risen by over 2,300%, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report. Total cases in children number almost 4.8 million since the pandemic started.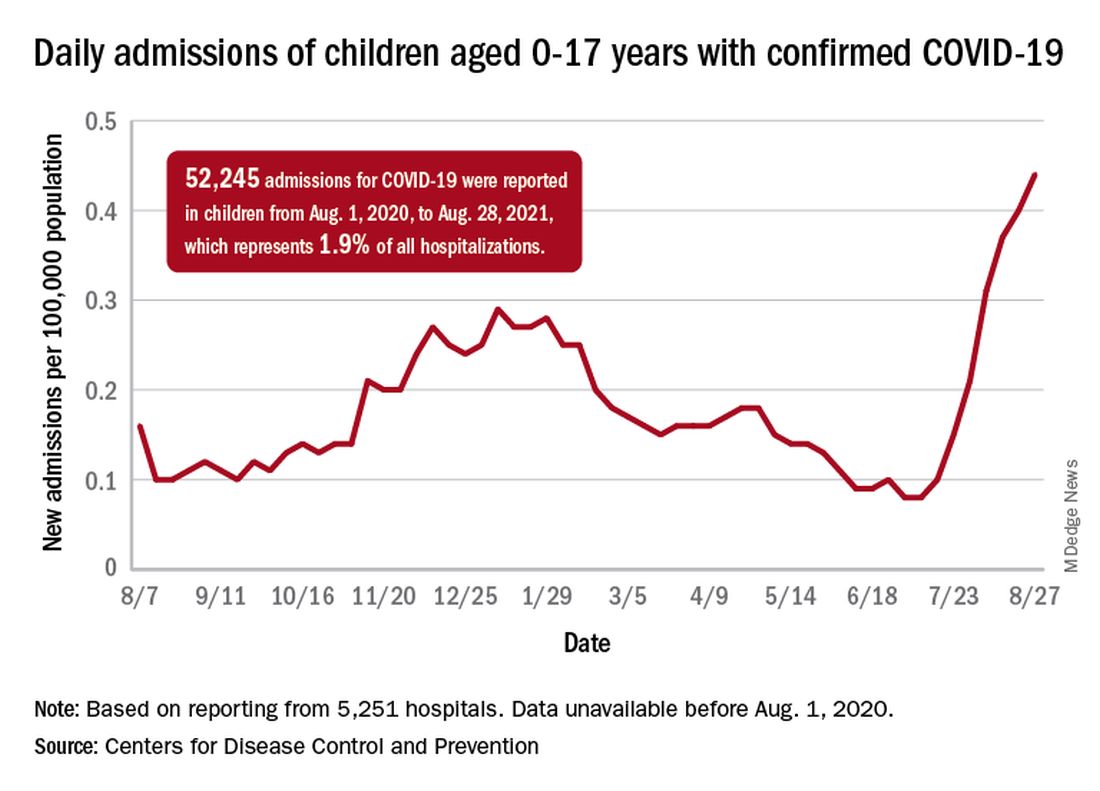
Vaccinations in children are following a different trend. Vaccine initiation has dropped 3 weeks in a row for both of the eligible age groups: First doses administered were down by 29% among 12- to 15-year-olds over that span and by 32% in 16- to 17-year-olds, according to data from the Centers for Disease Control and Prevention.
Since vaccination for children aged 12-15 years started in May, 49% had received at least one dose, and just over 36% were fully vaccinated as of Aug. 30. Among children aged 16-17 years, who have been eligible since December, 57.5% had gotten at least one dose of the vaccine and 46% have completed the two-dose regimen. The total number of children with at least one dose, including those under age 12 who are involved in clinical trials, was about 12 million, the CDC said on its COVID Data Tracker.
Hospitalizations are higher than ever
The recent rise in new child cases has been accompanied by an unprecedented increase in hospitalizations. The daily rate in children aged 0-17 years, which did not surpass 0.30 new admissions per 100,000 population during the worst of the winter surge, had risen to 0.45 per 100,000 by Aug. 26. Since July 4, when the new-admission rate was at its low point of 0.07 per 100,000, hospitalizations in children have jumped by 543%, based on data reported to the CDC by 5,251 hospitals.
A total of 52,245 children were admitted with confirmed COVID-19 from Aug. 1, 2020, when the CDC dataset begins, to Aug. 28, 2021. Those children represent 1.9% of all COVID admissions (2.7 million) in the United States over that period, the CDC said.
Total COVID-related deaths in children are up to 425 in the 48 jurisdictions (45 states, New York City, Puerto Rico, and Guam) that provide mortality data by age, the AAP and the CHA said.
Record-high numbers for the previous 2 reporting weeks – 23 deaths during Aug. 20-26 and 24 deaths during Aug. 13-19, when the previous weekly high was 16 – at least partially reflect the recent addition of South Carolina and New Mexico to the AAP/CHA database, as the two states just started reporting age-related data.
Weekly pediatric cases of COVID-19 exceeded 200,000 for just the second time during the pandemic, while new vaccinations in children continued to decline.
The weekly count has now increased for 9 consecutive weeks, during which time it has risen by over 2,300%, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report. Total cases in children number almost 4.8 million since the pandemic started.
Vaccinations in children are following a different trend. Vaccine initiation has dropped 3 weeks in a row for both of the eligible age groups: First doses administered were down by 29% among 12- to 15-year-olds over that span and by 32% in 16- to 17-year-olds, according to data from the Centers for Disease Control and Prevention.
Since vaccination for children aged 12-15 years started in May, 49% had received at least one dose, and just over 36% were fully vaccinated as of Aug. 30. Among children aged 16-17 years, who have been eligible since December, 57.5% had gotten at least one dose of the vaccine and 46% have completed the two-dose regimen. The total number of children with at least one dose, including those under age 12 who are involved in clinical trials, was about 12 million, the CDC said on its COVID Data Tracker.
Hospitalizations are higher than ever
The recent rise in new child cases has been accompanied by an unprecedented increase in hospitalizations. The daily rate in children aged 0-17 years, which did not surpass 0.30 new admissions per 100,000 population during the worst of the winter surge, had risen to 0.45 per 100,000 by Aug. 26. Since July 4, when the new-admission rate was at its low point of 0.07 per 100,000, hospitalizations in children have jumped by 543%, based on data reported to the CDC by 5,251 hospitals.
A total of 52,245 children were admitted with confirmed COVID-19 from Aug. 1, 2020, when the CDC dataset begins, to Aug. 28, 2021. Those children represent 1.9% of all COVID admissions (2.7 million) in the United States over that period, the CDC said.
Total COVID-related deaths in children are up to 425 in the 48 jurisdictions (45 states, New York City, Puerto Rico, and Guam) that provide mortality data by age, the AAP and the CHA said.
Record-high numbers for the previous 2 reporting weeks – 23 deaths during Aug. 20-26 and 24 deaths during Aug. 13-19, when the previous weekly high was 16 – at least partially reflect the recent addition of South Carolina and New Mexico to the AAP/CHA database, as the two states just started reporting age-related data.
Weekly pediatric cases of COVID-19 exceeded 200,000 for just the second time during the pandemic, while new vaccinations in children continued to decline.
The weekly count has now increased for 9 consecutive weeks, during which time it has risen by over 2,300%, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report. Total cases in children number almost 4.8 million since the pandemic started.
Vaccinations in children are following a different trend. Vaccine initiation has dropped 3 weeks in a row for both of the eligible age groups: First doses administered were down by 29% among 12- to 15-year-olds over that span and by 32% in 16- to 17-year-olds, according to data from the Centers for Disease Control and Prevention.
Since vaccination for children aged 12-15 years started in May, 49% had received at least one dose, and just over 36% were fully vaccinated as of Aug. 30. Among children aged 16-17 years, who have been eligible since December, 57.5% had gotten at least one dose of the vaccine and 46% have completed the two-dose regimen. The total number of children with at least one dose, including those under age 12 who are involved in clinical trials, was about 12 million, the CDC said on its COVID Data Tracker.
Hospitalizations are higher than ever
The recent rise in new child cases has been accompanied by an unprecedented increase in hospitalizations. The daily rate in children aged 0-17 years, which did not surpass 0.30 new admissions per 100,000 population during the worst of the winter surge, had risen to 0.45 per 100,000 by Aug. 26. Since July 4, when the new-admission rate was at its low point of 0.07 per 100,000, hospitalizations in children have jumped by 543%, based on data reported to the CDC by 5,251 hospitals.
A total of 52,245 children were admitted with confirmed COVID-19 from Aug. 1, 2020, when the CDC dataset begins, to Aug. 28, 2021. Those children represent 1.9% of all COVID admissions (2.7 million) in the United States over that period, the CDC said.
Total COVID-related deaths in children are up to 425 in the 48 jurisdictions (45 states, New York City, Puerto Rico, and Guam) that provide mortality data by age, the AAP and the CHA said.
Record-high numbers for the previous 2 reporting weeks – 23 deaths during Aug. 20-26 and 24 deaths during Aug. 13-19, when the previous weekly high was 16 – at least partially reflect the recent addition of South Carolina and New Mexico to the AAP/CHA database, as the two states just started reporting age-related data.
EDs saw more benzodiazepine overdoses, but fewer patients overall, in 2020
In a year when emergency department visits dropped by almost 18%, visits for benzodiazepine overdoses did the opposite, according to a report from the Centers for Disease Control and Prevention.
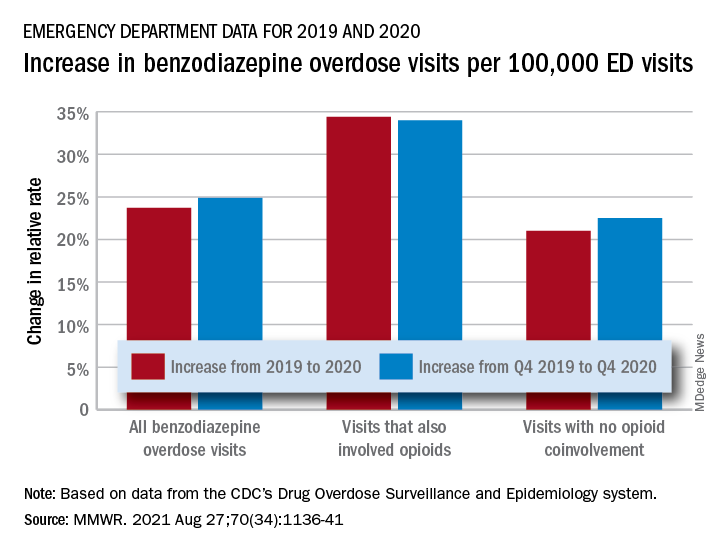
The actual increase in the number of overdose visits for benzodiazepine overdoses was quite small – from 15,547 in 2019 to 15,830 in 2020 (1.8%) – but the 11 million fewer ED visits magnified its effect, Stephen Liu, PhD, and associates said in the Morbidity and Mortality Weekly Report.
The rate of benzodiazepine overdose visits to all visits increased by 23.7% from 2019 (24.22 per 100,000 ED visits) to 2020 (29.97 per 100,000), with the larger share going to those involving opioids, which were up by 34.4%, compared with overdose visits not involving opioids (21.0%), the investigators said, based on data reported by 32 states and the District of Columbia to the CDC’s Drug Overdose Surveillance and Epidemiology system. All of the rate changes are statistically significant.
The number of overdose visits without opioid coinvolvement actually dropped, from 2019 (12,276) to 2020 (12,218), but not by enough to offset the decline in total visits, noted Dr. Liu, of the CDC’s National Center for Injury Prevention and Control and associates.
The number of deaths from benzodiazepine overdose, on the other hand, did not drop in 2020. Those data, coming from 23 states participating in the CDC’s State Unintentional Drug Overdose Reporting System, were available only for the first half of the year.
In those 6 months, The first quarter of 2020 also showed an increase, but exact numbers were not provided in the report. Overdose deaths rose by 22% for prescription forms of benzodiazepine and 520% for illicit forms in Q2 of 2020, compared with 2019, the researchers said.
Almost all of the benzodiazepine deaths (93%) in the first half of 2020 also involved opioids, mostly in the form of illicitly manufactured fentanyls (67% of all deaths). Between Q2 of 2019 and Q2 of 2020, involvement of illicit fentanyls in benzodiazepine overdose deaths increased from almost 57% to 71%, Dr. Liu and associates reported.
“Despite progress in reducing coprescribing [of opioids and benzodiazepines] before 2019, this study suggests a reversal in the decline in benzodiazepine deaths from 2017 to 2019, driven in part by increasing involvement of [illicitly manufactured fentanyls] in benzodiazepine deaths and influxes of illicit benzodiazepines,” they wrote.
In a year when emergency department visits dropped by almost 18%, visits for benzodiazepine overdoses did the opposite, according to a report from the Centers for Disease Control and Prevention.

The actual increase in the number of overdose visits for benzodiazepine overdoses was quite small – from 15,547 in 2019 to 15,830 in 2020 (1.8%) – but the 11 million fewer ED visits magnified its effect, Stephen Liu, PhD, and associates said in the Morbidity and Mortality Weekly Report.
The rate of benzodiazepine overdose visits to all visits increased by 23.7% from 2019 (24.22 per 100,000 ED visits) to 2020 (29.97 per 100,000), with the larger share going to those involving opioids, which were up by 34.4%, compared with overdose visits not involving opioids (21.0%), the investigators said, based on data reported by 32 states and the District of Columbia to the CDC’s Drug Overdose Surveillance and Epidemiology system. All of the rate changes are statistically significant.
The number of overdose visits without opioid coinvolvement actually dropped, from 2019 (12,276) to 2020 (12,218), but not by enough to offset the decline in total visits, noted Dr. Liu, of the CDC’s National Center for Injury Prevention and Control and associates.
The number of deaths from benzodiazepine overdose, on the other hand, did not drop in 2020. Those data, coming from 23 states participating in the CDC’s State Unintentional Drug Overdose Reporting System, were available only for the first half of the year.
In those 6 months, The first quarter of 2020 also showed an increase, but exact numbers were not provided in the report. Overdose deaths rose by 22% for prescription forms of benzodiazepine and 520% for illicit forms in Q2 of 2020, compared with 2019, the researchers said.
Almost all of the benzodiazepine deaths (93%) in the first half of 2020 also involved opioids, mostly in the form of illicitly manufactured fentanyls (67% of all deaths). Between Q2 of 2019 and Q2 of 2020, involvement of illicit fentanyls in benzodiazepine overdose deaths increased from almost 57% to 71%, Dr. Liu and associates reported.
“Despite progress in reducing coprescribing [of opioids and benzodiazepines] before 2019, this study suggests a reversal in the decline in benzodiazepine deaths from 2017 to 2019, driven in part by increasing involvement of [illicitly manufactured fentanyls] in benzodiazepine deaths and influxes of illicit benzodiazepines,” they wrote.
In a year when emergency department visits dropped by almost 18%, visits for benzodiazepine overdoses did the opposite, according to a report from the Centers for Disease Control and Prevention.

The actual increase in the number of overdose visits for benzodiazepine overdoses was quite small – from 15,547 in 2019 to 15,830 in 2020 (1.8%) – but the 11 million fewer ED visits magnified its effect, Stephen Liu, PhD, and associates said in the Morbidity and Mortality Weekly Report.
The rate of benzodiazepine overdose visits to all visits increased by 23.7% from 2019 (24.22 per 100,000 ED visits) to 2020 (29.97 per 100,000), with the larger share going to those involving opioids, which were up by 34.4%, compared with overdose visits not involving opioids (21.0%), the investigators said, based on data reported by 32 states and the District of Columbia to the CDC’s Drug Overdose Surveillance and Epidemiology system. All of the rate changes are statistically significant.
The number of overdose visits without opioid coinvolvement actually dropped, from 2019 (12,276) to 2020 (12,218), but not by enough to offset the decline in total visits, noted Dr. Liu, of the CDC’s National Center for Injury Prevention and Control and associates.
The number of deaths from benzodiazepine overdose, on the other hand, did not drop in 2020. Those data, coming from 23 states participating in the CDC’s State Unintentional Drug Overdose Reporting System, were available only for the first half of the year.
In those 6 months, The first quarter of 2020 also showed an increase, but exact numbers were not provided in the report. Overdose deaths rose by 22% for prescription forms of benzodiazepine and 520% for illicit forms in Q2 of 2020, compared with 2019, the researchers said.
Almost all of the benzodiazepine deaths (93%) in the first half of 2020 also involved opioids, mostly in the form of illicitly manufactured fentanyls (67% of all deaths). Between Q2 of 2019 and Q2 of 2020, involvement of illicit fentanyls in benzodiazepine overdose deaths increased from almost 57% to 71%, Dr. Liu and associates reported.
“Despite progress in reducing coprescribing [of opioids and benzodiazepines] before 2019, this study suggests a reversal in the decline in benzodiazepine deaths from 2017 to 2019, driven in part by increasing involvement of [illicitly manufactured fentanyls] in benzodiazepine deaths and influxes of illicit benzodiazepines,” they wrote.
FROM MMWR
Long COVID symptoms can persist for more than 1 year, study shows
Nearly half of people who are hospitalized with COVID-19 suffer at least one lingering symptom 1 year after discharge, according to the largest study yet to assess the dynamic recovery of a group of COVID-19 survivors 12 months after the illness.
The most common lingering symptoms are fatigue and muscle weakness. One-third continue to have shortness of breath.
Overall, at 12 months, COVID-19 survivors had more problems with mobility, pain or discomfort, and anxiety or depression, and had lower self-assessment scores of quality of life than matched COVID-free peers, the investigators report.
The study was published online Aug. 28 in The Lancet.
“While most had made a good recovery, health problems persisted in some patients, especially those who had been critically ill during their hospital stay,” Bin Cao, MD, from the National Center for Respiratory Medicine at the China-Japan Friendship Hospital and Capital Medical University, both in Beijing, said in a Lancet news release.
“Our findings suggest that recovery for some patients will take longer than 1 year, and this should be taken into account when planning delivery of health care services post pandemic,” Dr. Cao said.
“As the COVID-19 pandemic continues, the need to understand and respond to long COVID is increasingly pressing,” says a Lancet editorial.
“Symptoms such as persistent fatigue, breathlessness, brain fog, and depression could debilitate many millions of people globally. Long COVID is a modern medical challenge of the first order,” it reads.
Study details
Dr. Cao and colleagues studied 1,276 COVID-19 patients (median age 59; 53% men) discharged from a hospital in Wuhan, China, between Jan. 7 and May 29, 2020. The patients were assessed at 6 and 12 months from the date they first experienced COVID-19 symptoms.
Many symptoms resolved over time, regardless of the severity of illness. Yet 49% of patients still had at least one symptom 12 months after their acute illness, down from 68% at the 6-month mark, the authors report.
Fatigue and muscle weakness were the most commonly reported symptoms seen in 52% of patients at 6 months and 20% at 12 months. Compared with men, women were 1.4 times more likely to report fatigue or muscle weakness.
Patients treated with corticosteroids during the acute phase of COVID-19 were 1.5 times as likely to experience fatigue or muscle weakness after 12 months, compared with those who had not received corticosteroids.
Thirty percent of patients reported dyspnea at 12 months, slightly more than at 6 months (26%). Dyspnea was more common in the most severely ill patients needing a ventilator during their hospital stay (39%), compared with those who did not need oxygen treatment (25%).
At the 6-month check, 349 study participants underwent pulmonary function tests and 244 of those patients completed the same test at 12 months.
Spirometric and lung volume parameters of most of these patients were within normal limits at 12 months. But lung diffusion impairment was observed in about 20%-30% of patients who had been moderately ill with COVID-19 and as high as 54% in critically ill patients.
Compared with men, women were almost three times as likely to have lung diffusion impairment after 12 months.
Of 186 patients with abnormal lung CT scan at 6 months, 118 patients had a repeat CT scan at 12 months. The lung imaging abnormality gradually recovered during follow-up, yet 76% of the most critically ill patients still had ground glass opacity at 12 months.
Mental health hit
Among those patients who had been employed full- or part-time before catching COVID, the majority had returned to their original job (88%) and most had returned to their pre-COVID-19 level of work (76%) within 12 months.
Among those who did not return to their original work, 32% cited decreased physical function, 25% were unwilling to do their previous job, and 18% were unemployed.
As shown in multiple other studies, COVID-19 can take a toll on mental health. In this cohort, slightly more patients reported anxiety or depression at 12 months than at 6 months (23% vs. 26%), and the proportion was much greater than in matched community-dwelling adults without COVID-19 (5%).
Compared with men, women were twice as likely to report anxiety or depression.
“We do not yet fully understand why psychiatric symptoms are slightly more common at 1 year than at 6 months in COVID-19 survivors,” study author Xiaoying Gu, PhD, from the Institute of Clinical Medical Sciences, China-Japan Friendship Hospital, said in the news release.
“These could be caused by a biological process linked to the virus infection itself, or the body’s immune response to it. Or they could be linked to reduced social contact, loneliness, incomplete recovery of physical health, or loss of employment associated with illness. Large, long-term studies of COVID-19 survivors are needed so that we can better understand the long-term physical and mental health consequences of COVID-19,” Dr. Gu said.
The authors caution that the findings represent a group of patients from a single hospital in China and the cohort included only a small number of patients who had been admitted to intensive care (94 of 1,276; 7.4%).
The Lancet editorial urges the scientific and medical community to “collaborate to explore the mechanism and pathogenesis of long COVID, estimate the global and regional disease burdens, better delineate who is most at risk, understand how vaccines might affect the condition, and find effective treatments via randomized controlled trials.”
“At the same time, health care providers must acknowledge and validate the toll of the persistent symptoms of long COVID on patients, and health systems need to be prepared to meet individualized, patient-oriented goals, with an appropriately trained workforce involving physical, cognitive, social, and occupational elements,” the editorial states.
“Answering these research questions while providing compassionate and multidisciplinary care will require the full breadth of scientific and medical ingenuity. It is a challenge to which the whole health community must rise,” the editorialists conclude.
The study was funded by the Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences, the National Natural Science Foundation of China, the National Key Research and Development Program of China, Major Projects of National Science and Technology on New Drug Creation and Development of Pulmonary Tuberculosis, the China Evergrande Group, the Jack Ma Foundation, Sino Biopharmaceutical, the Ping An Insurance (Group), and the New Sunshine Charity Foundation. The full list of author disclosures is available with the original article.
A version of this article first appeared on Medscape.com.
Nearly half of people who are hospitalized with COVID-19 suffer at least one lingering symptom 1 year after discharge, according to the largest study yet to assess the dynamic recovery of a group of COVID-19 survivors 12 months after the illness.
The most common lingering symptoms are fatigue and muscle weakness. One-third continue to have shortness of breath.
Overall, at 12 months, COVID-19 survivors had more problems with mobility, pain or discomfort, and anxiety or depression, and had lower self-assessment scores of quality of life than matched COVID-free peers, the investigators report.
The study was published online Aug. 28 in The Lancet.
“While most had made a good recovery, health problems persisted in some patients, especially those who had been critically ill during their hospital stay,” Bin Cao, MD, from the National Center for Respiratory Medicine at the China-Japan Friendship Hospital and Capital Medical University, both in Beijing, said in a Lancet news release.
“Our findings suggest that recovery for some patients will take longer than 1 year, and this should be taken into account when planning delivery of health care services post pandemic,” Dr. Cao said.
“As the COVID-19 pandemic continues, the need to understand and respond to long COVID is increasingly pressing,” says a Lancet editorial.
“Symptoms such as persistent fatigue, breathlessness, brain fog, and depression could debilitate many millions of people globally. Long COVID is a modern medical challenge of the first order,” it reads.
Study details
Dr. Cao and colleagues studied 1,276 COVID-19 patients (median age 59; 53% men) discharged from a hospital in Wuhan, China, between Jan. 7 and May 29, 2020. The patients were assessed at 6 and 12 months from the date they first experienced COVID-19 symptoms.
Many symptoms resolved over time, regardless of the severity of illness. Yet 49% of patients still had at least one symptom 12 months after their acute illness, down from 68% at the 6-month mark, the authors report.
Fatigue and muscle weakness were the most commonly reported symptoms seen in 52% of patients at 6 months and 20% at 12 months. Compared with men, women were 1.4 times more likely to report fatigue or muscle weakness.
Patients treated with corticosteroids during the acute phase of COVID-19 were 1.5 times as likely to experience fatigue or muscle weakness after 12 months, compared with those who had not received corticosteroids.
Thirty percent of patients reported dyspnea at 12 months, slightly more than at 6 months (26%). Dyspnea was more common in the most severely ill patients needing a ventilator during their hospital stay (39%), compared with those who did not need oxygen treatment (25%).
At the 6-month check, 349 study participants underwent pulmonary function tests and 244 of those patients completed the same test at 12 months.
Spirometric and lung volume parameters of most of these patients were within normal limits at 12 months. But lung diffusion impairment was observed in about 20%-30% of patients who had been moderately ill with COVID-19 and as high as 54% in critically ill patients.
Compared with men, women were almost three times as likely to have lung diffusion impairment after 12 months.
Of 186 patients with abnormal lung CT scan at 6 months, 118 patients had a repeat CT scan at 12 months. The lung imaging abnormality gradually recovered during follow-up, yet 76% of the most critically ill patients still had ground glass opacity at 12 months.
Mental health hit
Among those patients who had been employed full- or part-time before catching COVID, the majority had returned to their original job (88%) and most had returned to their pre-COVID-19 level of work (76%) within 12 months.
Among those who did not return to their original work, 32% cited decreased physical function, 25% were unwilling to do their previous job, and 18% were unemployed.
As shown in multiple other studies, COVID-19 can take a toll on mental health. In this cohort, slightly more patients reported anxiety or depression at 12 months than at 6 months (23% vs. 26%), and the proportion was much greater than in matched community-dwelling adults without COVID-19 (5%).
Compared with men, women were twice as likely to report anxiety or depression.
“We do not yet fully understand why psychiatric symptoms are slightly more common at 1 year than at 6 months in COVID-19 survivors,” study author Xiaoying Gu, PhD, from the Institute of Clinical Medical Sciences, China-Japan Friendship Hospital, said in the news release.
“These could be caused by a biological process linked to the virus infection itself, or the body’s immune response to it. Or they could be linked to reduced social contact, loneliness, incomplete recovery of physical health, or loss of employment associated with illness. Large, long-term studies of COVID-19 survivors are needed so that we can better understand the long-term physical and mental health consequences of COVID-19,” Dr. Gu said.
The authors caution that the findings represent a group of patients from a single hospital in China and the cohort included only a small number of patients who had been admitted to intensive care (94 of 1,276; 7.4%).
The Lancet editorial urges the scientific and medical community to “collaborate to explore the mechanism and pathogenesis of long COVID, estimate the global and regional disease burdens, better delineate who is most at risk, understand how vaccines might affect the condition, and find effective treatments via randomized controlled trials.”
“At the same time, health care providers must acknowledge and validate the toll of the persistent symptoms of long COVID on patients, and health systems need to be prepared to meet individualized, patient-oriented goals, with an appropriately trained workforce involving physical, cognitive, social, and occupational elements,” the editorial states.
“Answering these research questions while providing compassionate and multidisciplinary care will require the full breadth of scientific and medical ingenuity. It is a challenge to which the whole health community must rise,” the editorialists conclude.
The study was funded by the Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences, the National Natural Science Foundation of China, the National Key Research and Development Program of China, Major Projects of National Science and Technology on New Drug Creation and Development of Pulmonary Tuberculosis, the China Evergrande Group, the Jack Ma Foundation, Sino Biopharmaceutical, the Ping An Insurance (Group), and the New Sunshine Charity Foundation. The full list of author disclosures is available with the original article.
A version of this article first appeared on Medscape.com.
Nearly half of people who are hospitalized with COVID-19 suffer at least one lingering symptom 1 year after discharge, according to the largest study yet to assess the dynamic recovery of a group of COVID-19 survivors 12 months after the illness.
The most common lingering symptoms are fatigue and muscle weakness. One-third continue to have shortness of breath.
Overall, at 12 months, COVID-19 survivors had more problems with mobility, pain or discomfort, and anxiety or depression, and had lower self-assessment scores of quality of life than matched COVID-free peers, the investigators report.
The study was published online Aug. 28 in The Lancet.
“While most had made a good recovery, health problems persisted in some patients, especially those who had been critically ill during their hospital stay,” Bin Cao, MD, from the National Center for Respiratory Medicine at the China-Japan Friendship Hospital and Capital Medical University, both in Beijing, said in a Lancet news release.
“Our findings suggest that recovery for some patients will take longer than 1 year, and this should be taken into account when planning delivery of health care services post pandemic,” Dr. Cao said.
“As the COVID-19 pandemic continues, the need to understand and respond to long COVID is increasingly pressing,” says a Lancet editorial.
“Symptoms such as persistent fatigue, breathlessness, brain fog, and depression could debilitate many millions of people globally. Long COVID is a modern medical challenge of the first order,” it reads.
Study details
Dr. Cao and colleagues studied 1,276 COVID-19 patients (median age 59; 53% men) discharged from a hospital in Wuhan, China, between Jan. 7 and May 29, 2020. The patients were assessed at 6 and 12 months from the date they first experienced COVID-19 symptoms.
Many symptoms resolved over time, regardless of the severity of illness. Yet 49% of patients still had at least one symptom 12 months after their acute illness, down from 68% at the 6-month mark, the authors report.
Fatigue and muscle weakness were the most commonly reported symptoms seen in 52% of patients at 6 months and 20% at 12 months. Compared with men, women were 1.4 times more likely to report fatigue or muscle weakness.
Patients treated with corticosteroids during the acute phase of COVID-19 were 1.5 times as likely to experience fatigue or muscle weakness after 12 months, compared with those who had not received corticosteroids.
Thirty percent of patients reported dyspnea at 12 months, slightly more than at 6 months (26%). Dyspnea was more common in the most severely ill patients needing a ventilator during their hospital stay (39%), compared with those who did not need oxygen treatment (25%).
At the 6-month check, 349 study participants underwent pulmonary function tests and 244 of those patients completed the same test at 12 months.
Spirometric and lung volume parameters of most of these patients were within normal limits at 12 months. But lung diffusion impairment was observed in about 20%-30% of patients who had been moderately ill with COVID-19 and as high as 54% in critically ill patients.
Compared with men, women were almost three times as likely to have lung diffusion impairment after 12 months.
Of 186 patients with abnormal lung CT scan at 6 months, 118 patients had a repeat CT scan at 12 months. The lung imaging abnormality gradually recovered during follow-up, yet 76% of the most critically ill patients still had ground glass opacity at 12 months.
Mental health hit
Among those patients who had been employed full- or part-time before catching COVID, the majority had returned to their original job (88%) and most had returned to their pre-COVID-19 level of work (76%) within 12 months.
Among those who did not return to their original work, 32% cited decreased physical function, 25% were unwilling to do their previous job, and 18% were unemployed.
As shown in multiple other studies, COVID-19 can take a toll on mental health. In this cohort, slightly more patients reported anxiety or depression at 12 months than at 6 months (23% vs. 26%), and the proportion was much greater than in matched community-dwelling adults without COVID-19 (5%).
Compared with men, women were twice as likely to report anxiety or depression.
“We do not yet fully understand why psychiatric symptoms are slightly more common at 1 year than at 6 months in COVID-19 survivors,” study author Xiaoying Gu, PhD, from the Institute of Clinical Medical Sciences, China-Japan Friendship Hospital, said in the news release.
“These could be caused by a biological process linked to the virus infection itself, or the body’s immune response to it. Or they could be linked to reduced social contact, loneliness, incomplete recovery of physical health, or loss of employment associated with illness. Large, long-term studies of COVID-19 survivors are needed so that we can better understand the long-term physical and mental health consequences of COVID-19,” Dr. Gu said.
The authors caution that the findings represent a group of patients from a single hospital in China and the cohort included only a small number of patients who had been admitted to intensive care (94 of 1,276; 7.4%).
The Lancet editorial urges the scientific and medical community to “collaborate to explore the mechanism and pathogenesis of long COVID, estimate the global and regional disease burdens, better delineate who is most at risk, understand how vaccines might affect the condition, and find effective treatments via randomized controlled trials.”
“At the same time, health care providers must acknowledge and validate the toll of the persistent symptoms of long COVID on patients, and health systems need to be prepared to meet individualized, patient-oriented goals, with an appropriately trained workforce involving physical, cognitive, social, and occupational elements,” the editorial states.
“Answering these research questions while providing compassionate and multidisciplinary care will require the full breadth of scientific and medical ingenuity. It is a challenge to which the whole health community must rise,” the editorialists conclude.
The study was funded by the Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences, the National Natural Science Foundation of China, the National Key Research and Development Program of China, Major Projects of National Science and Technology on New Drug Creation and Development of Pulmonary Tuberculosis, the China Evergrande Group, the Jack Ma Foundation, Sino Biopharmaceutical, the Ping An Insurance (Group), and the New Sunshine Charity Foundation. The full list of author disclosures is available with the original article.
A version of this article first appeared on Medscape.com.
EMPEROR-Preserved spouts torrent of reports on empagliflozin treatment of HFpEF
The featured report from the 6,000-patient EMPEROR-Preserved trial at the virtual annual congress of the European Society of Cardiology drew lots of attention for its headline finding: the first unequivocal demonstration that a medication, empagliflozin, can significantly reduce the rate of cardiovascular death and hospitalization for heart failure in patients with heart failure with preserved ejection fraction (HFpEF, a left ventricular ejection fraction of more than 40%), with the details simultaneously published online.
But at the same time, the EMPEROR-Preserved investigators released four additional reports with a lot more outcome analyses that also deserve some attention.
The puzzling neutral effect on renal events
Perhaps the most surprising and complicated set of findings among the main EMPEROR-Preserved outcomes involved renal outcomes.
The trial’s primary outcome was the combined rate of cardiovascular death or hospitalization for heart failure (HHF), and the results showed that treatment with empagliflozin (Jardiance) for a median of 26 months on top of standard treatment for patients with HFpEF led to a significant 21% relative risk reduction, compared with placebo-treated patients.
The trial had two prespecified secondary outcomes. One was the total number of HHF, which dropped by a significant 27%, compared with placebo. The second was the mean change in slope of estimated glomerular filtration rate (eGFR) on an annualized basis, and the empagliflozin regimen reduced the cumulative annual deficit, compared with placebo by an average of 1.36 mL/min per 1.73 m2, a significant difference.
This preservation of renal function was consistent with results from many prior studies of empagliflozin and all of the other U.S.-approved agents from the sodium-glucose cotransporter 2 inhibitor class. Preservation of renal function and a reduction in renal events has become a hallmark property of all agents in the SGLT2 inhibitor class both in patients with type 2 diabetes, as well as in those without diabetes but with heart failure with reduced ejection fraction (HFrEF) or with chronic kidney disease.
EMPEROR-Preserved threw a wrench into what had been an unbroken history of renal protection by SGLT2 inhibitors. That happened when a prespecified endpoint of the study – a composite renal outcome defined as time to first occurrence of chronic dialysis, renal transplantation, a sustained reduction of at least 40% in eGFR, or a sustained drop in eGFR of more than 10 or 15 mL/min per 1.73 m2 from baseline – yielded an unexpected neutral finding.
For this composite renal outcome, EMPEROR-Preserved showed a nonsignificant 5% reduction, compared with placebo, a result that both differed from what had been seen in essentially all the other SGLT2 inhibitor trials that had looked at this, but which also seemed at odds with the observed significant preservation of renal function that seemed substantial enough to produce a clinically meaningful benefit.
Renal effects blunted in HFpEF
The immediate upshot was a letter published by several EMPEROR-Preserved investigators that spelled out this discrepancy and came to the jolting conclusion that “eGFR slope analysis has limitations as a surrogate for predicting the effect of drugs on renal outcomes in patients with heart failure.”
The same authors, along with some additional associates, also published a second letter that noted a further unexpected twist with the renal outcome: “In prior large-scale clinical trials, the effect of SGLT2 inhibitors on heart failure and renal outcomes had consistently tracked together,” they noted, but in this case it didn’t, a discordance they said was “extraordinarily puzzling”.
This led the study’s leaders to reanalyze the renal outcomes using a different definition, one that Milton Packer, MD, who helped design the trial and oversaw several of its analyses, called “a more conventional definition of renal events,” during his presentation of these findings at the congress. The researchers swapped out a 40% drop from baseline eGFR as an event and replaced it with a 50% decline, a change designed to screen out less severe, and often transient, reductions in kidney function that have less lasting impact on health. They also added an additional component to the composite endpoint, renal death. A revised analysis using this new renal composite outcome appeared in the European Journal of Heart Failure letter.
This change cut the total number of renal events tallied in the trial nearly in half, down to 112, and showed a more robust decline in renal events with empagliflozin treatment compared with the initial analysis, although the drop remained nonsignificant. The revised analysis also showed that the overall, nonsignificant 22% relative reduction in renal events in patients on empagliflozin, compared with placebo, dwindled down to completely nonexistent in the tertile of patients with a left ventricular ejection fraction of 60% or greater. In this tertile the hazard ratio actually showed a nonsignificant point estimate of a 24% increased rate of renal events on empagliflozin, with the caveat that this subgroup now included a total of just 40 total events between the two treatment arms. (Each of the two other tertiles also had roughly the same number of total events.)
The biggest effect on renal-event reduction was in the tertile of patients with an ejection fraction of 41%-49%, in which empagliflozin treatment was linked with a significant 59% cut in renal events, compared with placebo. The analysis also showed significant heterogeneity in thus outcome between this subgroup and the other two tertiles that had higher ejection fractions and showed reduced rates of protection by empagliflozin against renal events.
This apparent blunting of a renal effect despite preservation of renal function seemed to mimic the blunting of the primary cardiovascular outcome effect that also appeared in patients with ejection fractions in the 60%-65% range or above.
“If we knew what blunted the effect of empagliflozin on heart failure outcomes at higher ejection fraction levels, we think the same explanation may also apply to the blunting of effect on renal outcomes, but right now we do not know the answer to either question,” Dr. Packer said in an interview. He’s suggested that one possibility is that many of the enrolled patients identified as having HFpEF, but with these high ejection fractions may have not actually had HFpEF, and their signs and symptoms may have instead resulted from atrial fibrillation.
“Many patients with an ejection fraction of 60%-65% and above had atrial fibrillation,” he noted, with a prevalence at enrollment in this subgroup of about 50%. Atrial fibrillation can cause dyspnea, a hallmark symptom leading to diagnosis of heart failure, and it also increases levels of N-terminal of the prohormone brain natriuretic peptide, a metric that served as a gatekeeper for entry into the trial. “Essentially, we are saying that many of the criteria that we specified to ensure that patients had heart failure probably did not work very well in patients with an ejection fraction of 65% or greater,” said Dr. Packer, a cardiologist at Baylor University Medical Center in Dallas. “We need to figure out who these patients are.”
Some experts not involved with the study voiced skepticism that the renal findings reflected a real issue.
“I’m quite optimistic that in the long-term the effect on eGFR will translate into renal protection,” said Rudolf A. de Boer, MD, PhD, a professor of translational cardiology at University Medical Center Groningen (the Netherlands), and designated discussant at the congress for the presentation by Dr. Packer.
John J.V. McMurray, MD, a professor of cardiology and a heart failure specialist at Glasgow University, speculated that the unexpected renal outcomes data may relate to the initial decline in renal function produced by treatment with SGLT2 inhibitors despite their longer-term enhancement of renal protection.
“If you use a treatment that protects the kidneys in the long-term but causes an initial dip in eGFR, more patients receiving that treatment will have an early ‘event,’ ” he noted in an interview. He also cautioned about the dangers of subgroup analyses that dice the study population into small cohorts.
“Trials are powered to look at the effect of treatment in the overall population. Everything else is exploratory, underpowered, and subject to the play of chance,” Dr. McMurray stressed.
Counting additional cardiovascular disease events allows more analyses
A third auxiliary report from the EMPEROR-Preserved investigators performed several prespecified analyses that depended on adding additional cardiovascular disease endpoints to the core tallies of cardiovascular death or HHF – such as emergent, urgent, and outpatient events that reflected worsening heart failure – and also included information on diuretic and vasopressor use because of worsening heart failure. The increased event numbers allowed the researchers to perform 30 additional analyses included in this report, according to the count kept by Dr. Packer who was the lead author.
He highlighted several of the additional results in this paper that documented benefits from empagliflozin treatment, compared with placebo:
- A significant 29% reduction in the need for admission to a cardiac care unit or intensive care unit during an HHF.
- A nonsignificant 33% reduction in the need for intravenous vasopressors or positive inotropic drugs during HHF.
- A significantly increased rate of patients achieving a higher New York Heart Association functional class. For example, after the first year of treatment patients who received empagliflozin had a 37% higher rate of functional class improvement, compared with patients who received placebo.
Dr. McMurray had his own list of key takeaways from this paper, including:
- Among patients who needed hospitalization, “those treated with empagliflozin were less sick than those in the placebo group.”
- In addition to reducing HHF empagliflozin treatment also reduced episodes of outpatient worsening as reflected by their receipt of intensified diuretic treatment, which occurred a significant 27% less often, compared with patients on placebo.
- Treatment with empagliflozin also linked with a significant 39% relative reduction in emergency or urgent-care visits that required intravenous therapy.
Empagliflozin’s performance relative to sacubitril/valsartan
The fourth additional report focused on a post hoc, cross-trial comparison of the results from EMPEROR-Preserved and from another recent trial that, like EMPEROR-Preserved, assessed in patients with HFpEF a drug previously proven to work quite well in patients with HFrEF. The comparator drug was sacubitril/valsartan (Entresto), which underwent testing in patients with HFpEF in the PARAGON-HF trial.
The primary outcome of PARAGON-HF, which randomized 4,822 patients, was reduction in cardiovascular death and in total HHF. This dropped by a relative 13%, compared with placebo, during a median of 35 months, a between-group difference that came close to but did not achieve significance (P = .06). Despite this limitation, the Food and Drug Administration in February 2021 loosened the indication for using sacubitril/valsartan in patients with heart failure and a “below normal” ejection fraction, a category that can include many patients considered to have HFpEF.
Although the researchers who ran this analysis, including Dr. Packer, who was the first author, admitted that “comparison of effect sizes across trials is fraught with difficulties,” they nonetheless concluded from their analysis that “for all outcomes that included HHF the effect size was larger for empagliflozin than for sacubitril/valsartan.”
Dr. McMurray, a lead instigator for PARAGON-HF, said there was little to take away from this analysis.
“The patient populations were different, and sacubitril/valsartan was compared against an active therapy, valsartan,” while in EMPEROR-Preserved empagliflozin compared against placebo. “Most of us believe that sacubitril/valsartan and SGLT2 inhibitors work in different but complementary ways, and their benefits are additive. You would want patients with HFpEF or HFrEF to take both,” he said in an interview.
Dr. Packer agreed with that approach and added that he would probably also prescribe a third agent, spironolactone, to many patients with HFpEF.
EMPEROR-Preserved was sponsored by Boehringer Ingelheim and Eli Lilly, which jointly market empagliflozin (Jardiance). PARAGON-HF was sponsored by Novartis, which markets sacubitril/valsartan (Entresto). Dr. Packer has received consulting fees from Boehringer Ingelheim and from numerous other companies. Dr. de Boer has research contracts with Boehringer Ingelheim as well as from Abbott, AstraZeneca, Cardior, Ionis, Novo Nordisk, and Roche, and he has been a consultant to Novartis as well as to Abbott, AstraZeneca, Gayer, and Roche. Dr. McMurray led trials of sacubitril/valsartan sponsored by Novartis, and his institution has received compensation for his participation in studies sponsored by Abbvie, AstraZeneca, Cardurion, DalCor, GlaxoSmithKline, Pfizer, and Theracos.
The featured report from the 6,000-patient EMPEROR-Preserved trial at the virtual annual congress of the European Society of Cardiology drew lots of attention for its headline finding: the first unequivocal demonstration that a medication, empagliflozin, can significantly reduce the rate of cardiovascular death and hospitalization for heart failure in patients with heart failure with preserved ejection fraction (HFpEF, a left ventricular ejection fraction of more than 40%), with the details simultaneously published online.
But at the same time, the EMPEROR-Preserved investigators released four additional reports with a lot more outcome analyses that also deserve some attention.
The puzzling neutral effect on renal events
Perhaps the most surprising and complicated set of findings among the main EMPEROR-Preserved outcomes involved renal outcomes.
The trial’s primary outcome was the combined rate of cardiovascular death or hospitalization for heart failure (HHF), and the results showed that treatment with empagliflozin (Jardiance) for a median of 26 months on top of standard treatment for patients with HFpEF led to a significant 21% relative risk reduction, compared with placebo-treated patients.
The trial had two prespecified secondary outcomes. One was the total number of HHF, which dropped by a significant 27%, compared with placebo. The second was the mean change in slope of estimated glomerular filtration rate (eGFR) on an annualized basis, and the empagliflozin regimen reduced the cumulative annual deficit, compared with placebo by an average of 1.36 mL/min per 1.73 m2, a significant difference.
This preservation of renal function was consistent with results from many prior studies of empagliflozin and all of the other U.S.-approved agents from the sodium-glucose cotransporter 2 inhibitor class. Preservation of renal function and a reduction in renal events has become a hallmark property of all agents in the SGLT2 inhibitor class both in patients with type 2 diabetes, as well as in those without diabetes but with heart failure with reduced ejection fraction (HFrEF) or with chronic kidney disease.
EMPEROR-Preserved threw a wrench into what had been an unbroken history of renal protection by SGLT2 inhibitors. That happened when a prespecified endpoint of the study – a composite renal outcome defined as time to first occurrence of chronic dialysis, renal transplantation, a sustained reduction of at least 40% in eGFR, or a sustained drop in eGFR of more than 10 or 15 mL/min per 1.73 m2 from baseline – yielded an unexpected neutral finding.
For this composite renal outcome, EMPEROR-Preserved showed a nonsignificant 5% reduction, compared with placebo, a result that both differed from what had been seen in essentially all the other SGLT2 inhibitor trials that had looked at this, but which also seemed at odds with the observed significant preservation of renal function that seemed substantial enough to produce a clinically meaningful benefit.
Renal effects blunted in HFpEF
The immediate upshot was a letter published by several EMPEROR-Preserved investigators that spelled out this discrepancy and came to the jolting conclusion that “eGFR slope analysis has limitations as a surrogate for predicting the effect of drugs on renal outcomes in patients with heart failure.”
The same authors, along with some additional associates, also published a second letter that noted a further unexpected twist with the renal outcome: “In prior large-scale clinical trials, the effect of SGLT2 inhibitors on heart failure and renal outcomes had consistently tracked together,” they noted, but in this case it didn’t, a discordance they said was “extraordinarily puzzling”.
This led the study’s leaders to reanalyze the renal outcomes using a different definition, one that Milton Packer, MD, who helped design the trial and oversaw several of its analyses, called “a more conventional definition of renal events,” during his presentation of these findings at the congress. The researchers swapped out a 40% drop from baseline eGFR as an event and replaced it with a 50% decline, a change designed to screen out less severe, and often transient, reductions in kidney function that have less lasting impact on health. They also added an additional component to the composite endpoint, renal death. A revised analysis using this new renal composite outcome appeared in the European Journal of Heart Failure letter.
This change cut the total number of renal events tallied in the trial nearly in half, down to 112, and showed a more robust decline in renal events with empagliflozin treatment compared with the initial analysis, although the drop remained nonsignificant. The revised analysis also showed that the overall, nonsignificant 22% relative reduction in renal events in patients on empagliflozin, compared with placebo, dwindled down to completely nonexistent in the tertile of patients with a left ventricular ejection fraction of 60% or greater. In this tertile the hazard ratio actually showed a nonsignificant point estimate of a 24% increased rate of renal events on empagliflozin, with the caveat that this subgroup now included a total of just 40 total events between the two treatment arms. (Each of the two other tertiles also had roughly the same number of total events.)
The biggest effect on renal-event reduction was in the tertile of patients with an ejection fraction of 41%-49%, in which empagliflozin treatment was linked with a significant 59% cut in renal events, compared with placebo. The analysis also showed significant heterogeneity in thus outcome between this subgroup and the other two tertiles that had higher ejection fractions and showed reduced rates of protection by empagliflozin against renal events.
This apparent blunting of a renal effect despite preservation of renal function seemed to mimic the blunting of the primary cardiovascular outcome effect that also appeared in patients with ejection fractions in the 60%-65% range or above.
“If we knew what blunted the effect of empagliflozin on heart failure outcomes at higher ejection fraction levels, we think the same explanation may also apply to the blunting of effect on renal outcomes, but right now we do not know the answer to either question,” Dr. Packer said in an interview. He’s suggested that one possibility is that many of the enrolled patients identified as having HFpEF, but with these high ejection fractions may have not actually had HFpEF, and their signs and symptoms may have instead resulted from atrial fibrillation.
“Many patients with an ejection fraction of 60%-65% and above had atrial fibrillation,” he noted, with a prevalence at enrollment in this subgroup of about 50%. Atrial fibrillation can cause dyspnea, a hallmark symptom leading to diagnosis of heart failure, and it also increases levels of N-terminal of the prohormone brain natriuretic peptide, a metric that served as a gatekeeper for entry into the trial. “Essentially, we are saying that many of the criteria that we specified to ensure that patients had heart failure probably did not work very well in patients with an ejection fraction of 65% or greater,” said Dr. Packer, a cardiologist at Baylor University Medical Center in Dallas. “We need to figure out who these patients are.”
Some experts not involved with the study voiced skepticism that the renal findings reflected a real issue.
“I’m quite optimistic that in the long-term the effect on eGFR will translate into renal protection,” said Rudolf A. de Boer, MD, PhD, a professor of translational cardiology at University Medical Center Groningen (the Netherlands), and designated discussant at the congress for the presentation by Dr. Packer.
John J.V. McMurray, MD, a professor of cardiology and a heart failure specialist at Glasgow University, speculated that the unexpected renal outcomes data may relate to the initial decline in renal function produced by treatment with SGLT2 inhibitors despite their longer-term enhancement of renal protection.
“If you use a treatment that protects the kidneys in the long-term but causes an initial dip in eGFR, more patients receiving that treatment will have an early ‘event,’ ” he noted in an interview. He also cautioned about the dangers of subgroup analyses that dice the study population into small cohorts.
“Trials are powered to look at the effect of treatment in the overall population. Everything else is exploratory, underpowered, and subject to the play of chance,” Dr. McMurray stressed.
Counting additional cardiovascular disease events allows more analyses
A third auxiliary report from the EMPEROR-Preserved investigators performed several prespecified analyses that depended on adding additional cardiovascular disease endpoints to the core tallies of cardiovascular death or HHF – such as emergent, urgent, and outpatient events that reflected worsening heart failure – and also included information on diuretic and vasopressor use because of worsening heart failure. The increased event numbers allowed the researchers to perform 30 additional analyses included in this report, according to the count kept by Dr. Packer who was the lead author.
He highlighted several of the additional results in this paper that documented benefits from empagliflozin treatment, compared with placebo:
- A significant 29% reduction in the need for admission to a cardiac care unit or intensive care unit during an HHF.
- A nonsignificant 33% reduction in the need for intravenous vasopressors or positive inotropic drugs during HHF.
- A significantly increased rate of patients achieving a higher New York Heart Association functional class. For example, after the first year of treatment patients who received empagliflozin had a 37% higher rate of functional class improvement, compared with patients who received placebo.
Dr. McMurray had his own list of key takeaways from this paper, including:
- Among patients who needed hospitalization, “those treated with empagliflozin were less sick than those in the placebo group.”
- In addition to reducing HHF empagliflozin treatment also reduced episodes of outpatient worsening as reflected by their receipt of intensified diuretic treatment, which occurred a significant 27% less often, compared with patients on placebo.
- Treatment with empagliflozin also linked with a significant 39% relative reduction in emergency or urgent-care visits that required intravenous therapy.
Empagliflozin’s performance relative to sacubitril/valsartan
The fourth additional report focused on a post hoc, cross-trial comparison of the results from EMPEROR-Preserved and from another recent trial that, like EMPEROR-Preserved, assessed in patients with HFpEF a drug previously proven to work quite well in patients with HFrEF. The comparator drug was sacubitril/valsartan (Entresto), which underwent testing in patients with HFpEF in the PARAGON-HF trial.
The primary outcome of PARAGON-HF, which randomized 4,822 patients, was reduction in cardiovascular death and in total HHF. This dropped by a relative 13%, compared with placebo, during a median of 35 months, a between-group difference that came close to but did not achieve significance (P = .06). Despite this limitation, the Food and Drug Administration in February 2021 loosened the indication for using sacubitril/valsartan in patients with heart failure and a “below normal” ejection fraction, a category that can include many patients considered to have HFpEF.
Although the researchers who ran this analysis, including Dr. Packer, who was the first author, admitted that “comparison of effect sizes across trials is fraught with difficulties,” they nonetheless concluded from their analysis that “for all outcomes that included HHF the effect size was larger for empagliflozin than for sacubitril/valsartan.”
Dr. McMurray, a lead instigator for PARAGON-HF, said there was little to take away from this analysis.
“The patient populations were different, and sacubitril/valsartan was compared against an active therapy, valsartan,” while in EMPEROR-Preserved empagliflozin compared against placebo. “Most of us believe that sacubitril/valsartan and SGLT2 inhibitors work in different but complementary ways, and their benefits are additive. You would want patients with HFpEF or HFrEF to take both,” he said in an interview.
Dr. Packer agreed with that approach and added that he would probably also prescribe a third agent, spironolactone, to many patients with HFpEF.
EMPEROR-Preserved was sponsored by Boehringer Ingelheim and Eli Lilly, which jointly market empagliflozin (Jardiance). PARAGON-HF was sponsored by Novartis, which markets sacubitril/valsartan (Entresto). Dr. Packer has received consulting fees from Boehringer Ingelheim and from numerous other companies. Dr. de Boer has research contracts with Boehringer Ingelheim as well as from Abbott, AstraZeneca, Cardior, Ionis, Novo Nordisk, and Roche, and he has been a consultant to Novartis as well as to Abbott, AstraZeneca, Gayer, and Roche. Dr. McMurray led trials of sacubitril/valsartan sponsored by Novartis, and his institution has received compensation for his participation in studies sponsored by Abbvie, AstraZeneca, Cardurion, DalCor, GlaxoSmithKline, Pfizer, and Theracos.
The featured report from the 6,000-patient EMPEROR-Preserved trial at the virtual annual congress of the European Society of Cardiology drew lots of attention for its headline finding: the first unequivocal demonstration that a medication, empagliflozin, can significantly reduce the rate of cardiovascular death and hospitalization for heart failure in patients with heart failure with preserved ejection fraction (HFpEF, a left ventricular ejection fraction of more than 40%), with the details simultaneously published online.
But at the same time, the EMPEROR-Preserved investigators released four additional reports with a lot more outcome analyses that also deserve some attention.
The puzzling neutral effect on renal events
Perhaps the most surprising and complicated set of findings among the main EMPEROR-Preserved outcomes involved renal outcomes.
The trial’s primary outcome was the combined rate of cardiovascular death or hospitalization for heart failure (HHF), and the results showed that treatment with empagliflozin (Jardiance) for a median of 26 months on top of standard treatment for patients with HFpEF led to a significant 21% relative risk reduction, compared with placebo-treated patients.
The trial had two prespecified secondary outcomes. One was the total number of HHF, which dropped by a significant 27%, compared with placebo. The second was the mean change in slope of estimated glomerular filtration rate (eGFR) on an annualized basis, and the empagliflozin regimen reduced the cumulative annual deficit, compared with placebo by an average of 1.36 mL/min per 1.73 m2, a significant difference.
This preservation of renal function was consistent with results from many prior studies of empagliflozin and all of the other U.S.-approved agents from the sodium-glucose cotransporter 2 inhibitor class. Preservation of renal function and a reduction in renal events has become a hallmark property of all agents in the SGLT2 inhibitor class both in patients with type 2 diabetes, as well as in those without diabetes but with heart failure with reduced ejection fraction (HFrEF) or with chronic kidney disease.
EMPEROR-Preserved threw a wrench into what had been an unbroken history of renal protection by SGLT2 inhibitors. That happened when a prespecified endpoint of the study – a composite renal outcome defined as time to first occurrence of chronic dialysis, renal transplantation, a sustained reduction of at least 40% in eGFR, or a sustained drop in eGFR of more than 10 or 15 mL/min per 1.73 m2 from baseline – yielded an unexpected neutral finding.
For this composite renal outcome, EMPEROR-Preserved showed a nonsignificant 5% reduction, compared with placebo, a result that both differed from what had been seen in essentially all the other SGLT2 inhibitor trials that had looked at this, but which also seemed at odds with the observed significant preservation of renal function that seemed substantial enough to produce a clinically meaningful benefit.
Renal effects blunted in HFpEF
The immediate upshot was a letter published by several EMPEROR-Preserved investigators that spelled out this discrepancy and came to the jolting conclusion that “eGFR slope analysis has limitations as a surrogate for predicting the effect of drugs on renal outcomes in patients with heart failure.”
The same authors, along with some additional associates, also published a second letter that noted a further unexpected twist with the renal outcome: “In prior large-scale clinical trials, the effect of SGLT2 inhibitors on heart failure and renal outcomes had consistently tracked together,” they noted, but in this case it didn’t, a discordance they said was “extraordinarily puzzling”.
This led the study’s leaders to reanalyze the renal outcomes using a different definition, one that Milton Packer, MD, who helped design the trial and oversaw several of its analyses, called “a more conventional definition of renal events,” during his presentation of these findings at the congress. The researchers swapped out a 40% drop from baseline eGFR as an event and replaced it with a 50% decline, a change designed to screen out less severe, and often transient, reductions in kidney function that have less lasting impact on health. They also added an additional component to the composite endpoint, renal death. A revised analysis using this new renal composite outcome appeared in the European Journal of Heart Failure letter.
This change cut the total number of renal events tallied in the trial nearly in half, down to 112, and showed a more robust decline in renal events with empagliflozin treatment compared with the initial analysis, although the drop remained nonsignificant. The revised analysis also showed that the overall, nonsignificant 22% relative reduction in renal events in patients on empagliflozin, compared with placebo, dwindled down to completely nonexistent in the tertile of patients with a left ventricular ejection fraction of 60% or greater. In this tertile the hazard ratio actually showed a nonsignificant point estimate of a 24% increased rate of renal events on empagliflozin, with the caveat that this subgroup now included a total of just 40 total events between the two treatment arms. (Each of the two other tertiles also had roughly the same number of total events.)
The biggest effect on renal-event reduction was in the tertile of patients with an ejection fraction of 41%-49%, in which empagliflozin treatment was linked with a significant 59% cut in renal events, compared with placebo. The analysis also showed significant heterogeneity in thus outcome between this subgroup and the other two tertiles that had higher ejection fractions and showed reduced rates of protection by empagliflozin against renal events.
This apparent blunting of a renal effect despite preservation of renal function seemed to mimic the blunting of the primary cardiovascular outcome effect that also appeared in patients with ejection fractions in the 60%-65% range or above.
“If we knew what blunted the effect of empagliflozin on heart failure outcomes at higher ejection fraction levels, we think the same explanation may also apply to the blunting of effect on renal outcomes, but right now we do not know the answer to either question,” Dr. Packer said in an interview. He’s suggested that one possibility is that many of the enrolled patients identified as having HFpEF, but with these high ejection fractions may have not actually had HFpEF, and their signs and symptoms may have instead resulted from atrial fibrillation.
“Many patients with an ejection fraction of 60%-65% and above had atrial fibrillation,” he noted, with a prevalence at enrollment in this subgroup of about 50%. Atrial fibrillation can cause dyspnea, a hallmark symptom leading to diagnosis of heart failure, and it also increases levels of N-terminal of the prohormone brain natriuretic peptide, a metric that served as a gatekeeper for entry into the trial. “Essentially, we are saying that many of the criteria that we specified to ensure that patients had heart failure probably did not work very well in patients with an ejection fraction of 65% or greater,” said Dr. Packer, a cardiologist at Baylor University Medical Center in Dallas. “We need to figure out who these patients are.”
Some experts not involved with the study voiced skepticism that the renal findings reflected a real issue.
“I’m quite optimistic that in the long-term the effect on eGFR will translate into renal protection,” said Rudolf A. de Boer, MD, PhD, a professor of translational cardiology at University Medical Center Groningen (the Netherlands), and designated discussant at the congress for the presentation by Dr. Packer.
John J.V. McMurray, MD, a professor of cardiology and a heart failure specialist at Glasgow University, speculated that the unexpected renal outcomes data may relate to the initial decline in renal function produced by treatment with SGLT2 inhibitors despite their longer-term enhancement of renal protection.
“If you use a treatment that protects the kidneys in the long-term but causes an initial dip in eGFR, more patients receiving that treatment will have an early ‘event,’ ” he noted in an interview. He also cautioned about the dangers of subgroup analyses that dice the study population into small cohorts.
“Trials are powered to look at the effect of treatment in the overall population. Everything else is exploratory, underpowered, and subject to the play of chance,” Dr. McMurray stressed.
Counting additional cardiovascular disease events allows more analyses
A third auxiliary report from the EMPEROR-Preserved investigators performed several prespecified analyses that depended on adding additional cardiovascular disease endpoints to the core tallies of cardiovascular death or HHF – such as emergent, urgent, and outpatient events that reflected worsening heart failure – and also included information on diuretic and vasopressor use because of worsening heart failure. The increased event numbers allowed the researchers to perform 30 additional analyses included in this report, according to the count kept by Dr. Packer who was the lead author.
He highlighted several of the additional results in this paper that documented benefits from empagliflozin treatment, compared with placebo:
- A significant 29% reduction in the need for admission to a cardiac care unit or intensive care unit during an HHF.
- A nonsignificant 33% reduction in the need for intravenous vasopressors or positive inotropic drugs during HHF.
- A significantly increased rate of patients achieving a higher New York Heart Association functional class. For example, after the first year of treatment patients who received empagliflozin had a 37% higher rate of functional class improvement, compared with patients who received placebo.
Dr. McMurray had his own list of key takeaways from this paper, including:
- Among patients who needed hospitalization, “those treated with empagliflozin were less sick than those in the placebo group.”
- In addition to reducing HHF empagliflozin treatment also reduced episodes of outpatient worsening as reflected by their receipt of intensified diuretic treatment, which occurred a significant 27% less often, compared with patients on placebo.
- Treatment with empagliflozin also linked with a significant 39% relative reduction in emergency or urgent-care visits that required intravenous therapy.
Empagliflozin’s performance relative to sacubitril/valsartan
The fourth additional report focused on a post hoc, cross-trial comparison of the results from EMPEROR-Preserved and from another recent trial that, like EMPEROR-Preserved, assessed in patients with HFpEF a drug previously proven to work quite well in patients with HFrEF. The comparator drug was sacubitril/valsartan (Entresto), which underwent testing in patients with HFpEF in the PARAGON-HF trial.
The primary outcome of PARAGON-HF, which randomized 4,822 patients, was reduction in cardiovascular death and in total HHF. This dropped by a relative 13%, compared with placebo, during a median of 35 months, a between-group difference that came close to but did not achieve significance (P = .06). Despite this limitation, the Food and Drug Administration in February 2021 loosened the indication for using sacubitril/valsartan in patients with heart failure and a “below normal” ejection fraction, a category that can include many patients considered to have HFpEF.
Although the researchers who ran this analysis, including Dr. Packer, who was the first author, admitted that “comparison of effect sizes across trials is fraught with difficulties,” they nonetheless concluded from their analysis that “for all outcomes that included HHF the effect size was larger for empagliflozin than for sacubitril/valsartan.”
Dr. McMurray, a lead instigator for PARAGON-HF, said there was little to take away from this analysis.
“The patient populations were different, and sacubitril/valsartan was compared against an active therapy, valsartan,” while in EMPEROR-Preserved empagliflozin compared against placebo. “Most of us believe that sacubitril/valsartan and SGLT2 inhibitors work in different but complementary ways, and their benefits are additive. You would want patients with HFpEF or HFrEF to take both,” he said in an interview.
Dr. Packer agreed with that approach and added that he would probably also prescribe a third agent, spironolactone, to many patients with HFpEF.
EMPEROR-Preserved was sponsored by Boehringer Ingelheim and Eli Lilly, which jointly market empagliflozin (Jardiance). PARAGON-HF was sponsored by Novartis, which markets sacubitril/valsartan (Entresto). Dr. Packer has received consulting fees from Boehringer Ingelheim and from numerous other companies. Dr. de Boer has research contracts with Boehringer Ingelheim as well as from Abbott, AstraZeneca, Cardior, Ionis, Novo Nordisk, and Roche, and he has been a consultant to Novartis as well as to Abbott, AstraZeneca, Gayer, and Roche. Dr. McMurray led trials of sacubitril/valsartan sponsored by Novartis, and his institution has received compensation for his participation in studies sponsored by Abbvie, AstraZeneca, Cardurion, DalCor, GlaxoSmithKline, Pfizer, and Theracos.
FROM ESC 2021
Report urges complete residency overhaul
from the Graduate Medical Education Review Committee (UGRC) of the Coalition for Physician Accountability.
The 275-page report presents preliminary findings that were released in April 2021 and a long list of stakeholder comments. According to the report, the coalition will meet soon to discuss the final recommendations and consider next steps toward implementation.
The UGRC includes representatives of national medical organizations, medical schools, and residency programs. Among the organizations that participated in the report’s creation are the American Medical Association, the National Board of Medical Examiners, the American Osteopathic Association, the National Board of Osteopathic Medical Examiners, the Educational Commission for Foreign Medical Graduates, and the Association of American Medical Colleges.
The report identifies a list of challenges that affect the transition of medical students into residency programs and beyond. They include:
- Too much focus on finding and filling residency positions instead of “assuring learner competence and readiness for residency training”
- Inattention to assuring congruence between applicant goals and program missions
- Overreliance on licensure exam scores rather than “valid, trustworthy measures of students’ competence and clinical abilities”
- Increasing financial costs to students
- Individual and systemic biases in the UME-GME transition, as well as inequities related to international medical graduates
Seeking a common framework for competence
Overall, the report calls for increased standardization of how students are evaluated in medical school and how residency programs evaluate students. Less reliance should be placed on the numerical scores of the U.S. Medical Licensing Examination (USMLE), the report says, and more attention should be paid to the direct observation of student performance in clinical situations. In addition, the various organizations involved in the UME-GME transition process are asked to work better together.
To develop better methods of evaluating medical students and residents, UME and GME educators should jointly define and implement a common framework and set of competencies to apply to learners across the UME-GME transition, the report suggests.
While emphasizing the need for a broader student assessment framework, the report says, USMLE scores should also continue to be used in judging residency applicants. “Assessment information should be shared in residency applications and a postmatch learner handover. Licensing examinations should be used for their intended purpose to ensure requisite competence.”
Among the committee’s three dozen recommendations are the following:
- The Centers for Medicare & Medicaid Services should change the GME funding structure so that the initial residency period is calculated starting with the second year of postgraduate training. This change would allow residents to reconsider their career choices. Currently, if a resident decides to switch to another program or specialty after beginning training, the hospital may not receive full GME funding, so may be less likely to approve the change.
- Residency programs should improve recruitment practices to increase specialty-specific diversity of residents. Medical educators should also receive additional training regarding antiracism, avoiding bias, and ensuring equity.
- The self-reported demographic information of applicants to residency programs should be measured and shared with stakeholders, including the programs and medical schools, to promote equity. “A residency program that finds bias in its selection process could go back in real time to find qualified applicants who may have been missed, potentially improving outcomes,” the report notes.
- An interactive database of GME program and specialty track information should be created and made available to all applicants, medical schools, and residency programs at no cost to applicants. “Applicants and their advisors should be able to sort the information according to demographic and educational features that may significantly impact the likelihood of matching at a program.”
Less than half of applicants get in-depth reviews
The 2020 National Resident Matching Program Program Director Survey found that only 49% of applications received in-depth review. In light of this, the report suggests that the application system be updated to use modern information technology, including discrete fields for key data to expedite application reviews.
Many applications have been discarded because of various filters used to block consideration of certain applications. The report suggests that new filters be designed to ensure that each detects meaningful differences among applicants and promotes review based on mission alignment and likelihood of success in a program. Filters should be improved to decrease the likelihood of random exclusions of qualified applicants.
Specialty-specific, just-in-time training for all incoming first-year residents is also suggested to support the transition from the role of student to a physician ready to assume increased responsibility for patient care. In addition, the report urges adequate time be allowed between medical school graduation and residency to enable new residents to relocate and find homes.
The report also calls for a standardized process in the United States for initial licensing of doctors at entrance to residency in order to streamline the process of credentialing for both residency training and continuing practice.
Osteopathic students’ dilemma
To promote equitable treatment of applicants regardless of licensure examination requirements, comparable exams with different scales (COMLEX-USA and USMLE) should be reported within the electronic application system in a single field, the report said.
Osteopathic students, who make up 25% of U.S. medical students, must take the COMLEX-USA exam, but residency programs may filter them out if they don’t also take the USMLE exam. Thus, many osteopathic students take both exams, incurring extra time, cost, and stress.
The UGRC recommends creating a combined field in the electronic residency application service that normalizes the scores between the two exams. Residency programs could then filter applications based only on the single normalized score.
This approach makes sense from the viewpoint that it would reduce the pressure on osteopathic students to take the USMLE, Bryan Carmody, MD, an outspoken critic of various current training policies, said in an interview. But it could also have serious disadvantages.
For one thing, only osteopathic students can take the COMLEX-USA exam, he noted. If they don’t like their score, they can then take the USMLE test to get a higher score – an option that allopathic students don’t have. It’s not clear that they’d be prevented from doing this under the UGRC recommendation.
Second, he said, osteopathic students, on average, don’t do as well as allopathic students on the UMSLE exam. If they only take the COMLEX-USA test, they’re competing against other students who don’t do as well on tests as allopathic students do. If their scores were normalized with those of the USMLE test takers, they’d gain an unfair advantage against students who can only take the USMLE, including international medical graduates.
Although Dr. Carmody admitted that osteopathic students face a harder challenge than allopathic students in matching to residency programs, he said that the UGRC approach to the licensing exams might actually penalize them further. As a result of the scores of the two exams being averaged, residency program directors might discount the scores of all osteopathic students.
A version of this article first appeared on Medscape.com.
from the Graduate Medical Education Review Committee (UGRC) of the Coalition for Physician Accountability.
The 275-page report presents preliminary findings that were released in April 2021 and a long list of stakeholder comments. According to the report, the coalition will meet soon to discuss the final recommendations and consider next steps toward implementation.
The UGRC includes representatives of national medical organizations, medical schools, and residency programs. Among the organizations that participated in the report’s creation are the American Medical Association, the National Board of Medical Examiners, the American Osteopathic Association, the National Board of Osteopathic Medical Examiners, the Educational Commission for Foreign Medical Graduates, and the Association of American Medical Colleges.
The report identifies a list of challenges that affect the transition of medical students into residency programs and beyond. They include:
- Too much focus on finding and filling residency positions instead of “assuring learner competence and readiness for residency training”
- Inattention to assuring congruence between applicant goals and program missions
- Overreliance on licensure exam scores rather than “valid, trustworthy measures of students’ competence and clinical abilities”
- Increasing financial costs to students
- Individual and systemic biases in the UME-GME transition, as well as inequities related to international medical graduates
Seeking a common framework for competence
Overall, the report calls for increased standardization of how students are evaluated in medical school and how residency programs evaluate students. Less reliance should be placed on the numerical scores of the U.S. Medical Licensing Examination (USMLE), the report says, and more attention should be paid to the direct observation of student performance in clinical situations. In addition, the various organizations involved in the UME-GME transition process are asked to work better together.
To develop better methods of evaluating medical students and residents, UME and GME educators should jointly define and implement a common framework and set of competencies to apply to learners across the UME-GME transition, the report suggests.
While emphasizing the need for a broader student assessment framework, the report says, USMLE scores should also continue to be used in judging residency applicants. “Assessment information should be shared in residency applications and a postmatch learner handover. Licensing examinations should be used for their intended purpose to ensure requisite competence.”
Among the committee’s three dozen recommendations are the following:
- The Centers for Medicare & Medicaid Services should change the GME funding structure so that the initial residency period is calculated starting with the second year of postgraduate training. This change would allow residents to reconsider their career choices. Currently, if a resident decides to switch to another program or specialty after beginning training, the hospital may not receive full GME funding, so may be less likely to approve the change.
- Residency programs should improve recruitment practices to increase specialty-specific diversity of residents. Medical educators should also receive additional training regarding antiracism, avoiding bias, and ensuring equity.
- The self-reported demographic information of applicants to residency programs should be measured and shared with stakeholders, including the programs and medical schools, to promote equity. “A residency program that finds bias in its selection process could go back in real time to find qualified applicants who may have been missed, potentially improving outcomes,” the report notes.
- An interactive database of GME program and specialty track information should be created and made available to all applicants, medical schools, and residency programs at no cost to applicants. “Applicants and their advisors should be able to sort the information according to demographic and educational features that may significantly impact the likelihood of matching at a program.”
Less than half of applicants get in-depth reviews
The 2020 National Resident Matching Program Program Director Survey found that only 49% of applications received in-depth review. In light of this, the report suggests that the application system be updated to use modern information technology, including discrete fields for key data to expedite application reviews.
Many applications have been discarded because of various filters used to block consideration of certain applications. The report suggests that new filters be designed to ensure that each detects meaningful differences among applicants and promotes review based on mission alignment and likelihood of success in a program. Filters should be improved to decrease the likelihood of random exclusions of qualified applicants.
Specialty-specific, just-in-time training for all incoming first-year residents is also suggested to support the transition from the role of student to a physician ready to assume increased responsibility for patient care. In addition, the report urges adequate time be allowed between medical school graduation and residency to enable new residents to relocate and find homes.
The report also calls for a standardized process in the United States for initial licensing of doctors at entrance to residency in order to streamline the process of credentialing for both residency training and continuing practice.
Osteopathic students’ dilemma
To promote equitable treatment of applicants regardless of licensure examination requirements, comparable exams with different scales (COMLEX-USA and USMLE) should be reported within the electronic application system in a single field, the report said.
Osteopathic students, who make up 25% of U.S. medical students, must take the COMLEX-USA exam, but residency programs may filter them out if they don’t also take the USMLE exam. Thus, many osteopathic students take both exams, incurring extra time, cost, and stress.
The UGRC recommends creating a combined field in the electronic residency application service that normalizes the scores between the two exams. Residency programs could then filter applications based only on the single normalized score.
This approach makes sense from the viewpoint that it would reduce the pressure on osteopathic students to take the USMLE, Bryan Carmody, MD, an outspoken critic of various current training policies, said in an interview. But it could also have serious disadvantages.
For one thing, only osteopathic students can take the COMLEX-USA exam, he noted. If they don’t like their score, they can then take the USMLE test to get a higher score – an option that allopathic students don’t have. It’s not clear that they’d be prevented from doing this under the UGRC recommendation.
Second, he said, osteopathic students, on average, don’t do as well as allopathic students on the UMSLE exam. If they only take the COMLEX-USA test, they’re competing against other students who don’t do as well on tests as allopathic students do. If their scores were normalized with those of the USMLE test takers, they’d gain an unfair advantage against students who can only take the USMLE, including international medical graduates.
Although Dr. Carmody admitted that osteopathic students face a harder challenge than allopathic students in matching to residency programs, he said that the UGRC approach to the licensing exams might actually penalize them further. As a result of the scores of the two exams being averaged, residency program directors might discount the scores of all osteopathic students.
A version of this article first appeared on Medscape.com.
from the Graduate Medical Education Review Committee (UGRC) of the Coalition for Physician Accountability.
The 275-page report presents preliminary findings that were released in April 2021 and a long list of stakeholder comments. According to the report, the coalition will meet soon to discuss the final recommendations and consider next steps toward implementation.
The UGRC includes representatives of national medical organizations, medical schools, and residency programs. Among the organizations that participated in the report’s creation are the American Medical Association, the National Board of Medical Examiners, the American Osteopathic Association, the National Board of Osteopathic Medical Examiners, the Educational Commission for Foreign Medical Graduates, and the Association of American Medical Colleges.
The report identifies a list of challenges that affect the transition of medical students into residency programs and beyond. They include:
- Too much focus on finding and filling residency positions instead of “assuring learner competence and readiness for residency training”
- Inattention to assuring congruence between applicant goals and program missions
- Overreliance on licensure exam scores rather than “valid, trustworthy measures of students’ competence and clinical abilities”
- Increasing financial costs to students
- Individual and systemic biases in the UME-GME transition, as well as inequities related to international medical graduates
Seeking a common framework for competence
Overall, the report calls for increased standardization of how students are evaluated in medical school and how residency programs evaluate students. Less reliance should be placed on the numerical scores of the U.S. Medical Licensing Examination (USMLE), the report says, and more attention should be paid to the direct observation of student performance in clinical situations. In addition, the various organizations involved in the UME-GME transition process are asked to work better together.
To develop better methods of evaluating medical students and residents, UME and GME educators should jointly define and implement a common framework and set of competencies to apply to learners across the UME-GME transition, the report suggests.
While emphasizing the need for a broader student assessment framework, the report says, USMLE scores should also continue to be used in judging residency applicants. “Assessment information should be shared in residency applications and a postmatch learner handover. Licensing examinations should be used for their intended purpose to ensure requisite competence.”
Among the committee’s three dozen recommendations are the following:
- The Centers for Medicare & Medicaid Services should change the GME funding structure so that the initial residency period is calculated starting with the second year of postgraduate training. This change would allow residents to reconsider their career choices. Currently, if a resident decides to switch to another program or specialty after beginning training, the hospital may not receive full GME funding, so may be less likely to approve the change.
- Residency programs should improve recruitment practices to increase specialty-specific diversity of residents. Medical educators should also receive additional training regarding antiracism, avoiding bias, and ensuring equity.
- The self-reported demographic information of applicants to residency programs should be measured and shared with stakeholders, including the programs and medical schools, to promote equity. “A residency program that finds bias in its selection process could go back in real time to find qualified applicants who may have been missed, potentially improving outcomes,” the report notes.
- An interactive database of GME program and specialty track information should be created and made available to all applicants, medical schools, and residency programs at no cost to applicants. “Applicants and their advisors should be able to sort the information according to demographic and educational features that may significantly impact the likelihood of matching at a program.”
Less than half of applicants get in-depth reviews
The 2020 National Resident Matching Program Program Director Survey found that only 49% of applications received in-depth review. In light of this, the report suggests that the application system be updated to use modern information technology, including discrete fields for key data to expedite application reviews.
Many applications have been discarded because of various filters used to block consideration of certain applications. The report suggests that new filters be designed to ensure that each detects meaningful differences among applicants and promotes review based on mission alignment and likelihood of success in a program. Filters should be improved to decrease the likelihood of random exclusions of qualified applicants.
Specialty-specific, just-in-time training for all incoming first-year residents is also suggested to support the transition from the role of student to a physician ready to assume increased responsibility for patient care. In addition, the report urges adequate time be allowed between medical school graduation and residency to enable new residents to relocate and find homes.
The report also calls for a standardized process in the United States for initial licensing of doctors at entrance to residency in order to streamline the process of credentialing for both residency training and continuing practice.
Osteopathic students’ dilemma
To promote equitable treatment of applicants regardless of licensure examination requirements, comparable exams with different scales (COMLEX-USA and USMLE) should be reported within the electronic application system in a single field, the report said.
Osteopathic students, who make up 25% of U.S. medical students, must take the COMLEX-USA exam, but residency programs may filter them out if they don’t also take the USMLE exam. Thus, many osteopathic students take both exams, incurring extra time, cost, and stress.
The UGRC recommends creating a combined field in the electronic residency application service that normalizes the scores between the two exams. Residency programs could then filter applications based only on the single normalized score.
This approach makes sense from the viewpoint that it would reduce the pressure on osteopathic students to take the USMLE, Bryan Carmody, MD, an outspoken critic of various current training policies, said in an interview. But it could also have serious disadvantages.
For one thing, only osteopathic students can take the COMLEX-USA exam, he noted. If they don’t like their score, they can then take the USMLE test to get a higher score – an option that allopathic students don’t have. It’s not clear that they’d be prevented from doing this under the UGRC recommendation.
Second, he said, osteopathic students, on average, don’t do as well as allopathic students on the UMSLE exam. If they only take the COMLEX-USA test, they’re competing against other students who don’t do as well on tests as allopathic students do. If their scores were normalized with those of the USMLE test takers, they’d gain an unfair advantage against students who can only take the USMLE, including international medical graduates.
Although Dr. Carmody admitted that osteopathic students face a harder challenge than allopathic students in matching to residency programs, he said that the UGRC approach to the licensing exams might actually penalize them further. As a result of the scores of the two exams being averaged, residency program directors might discount the scores of all osteopathic students.
A version of this article first appeared on Medscape.com.
FDA OKs IV Briviact for seizures in kids as young as 1 month
All three brivaracetam formulations (tablets, oral solution, and IV) may now be used. The approval marks the first time that the IV formulation will be available for children, the company said in a news release.
The medication is already approved in the United States as monotherapy and adjunctive therapy in adults with epilepsy.
In an open-label follow-up pediatric study, an estimated 71.4% of patients aged 1 month to 17 years with partial-onset seizures remained on brivaracetam therapy at 1 year, and 64.3% did so at 2 years, the company reported.
“We often see children with seizures hospitalized, so it’s important to have a therapy like Briviact IV that can offer rapid administration in an effective dose when needed and does not require titration,” Raman Sankar, MD, PhD, distinguished professor and chief of pediatric neurology, University of California, Los Angeles, said in the release.
“The availability of the oral dose forms also allows continuity of treatment when these young patients are transitioning from hospital to home,” he added.
Safety profile
Dr. Sankar noted that with approval now of both the IV and oral formulations for partial-onset seizures in such young children, “we have a new option that helps meet a critical need in pediatric epilepsy.”
The most common adverse reactions with brivaracetam include somnolence and sedation, dizziness, fatigue, nausea, and vomiting. In the pediatric clinical trials, the safety profile for pediatric patients was similar to adults.
In the adult trials, psychiatric adverse reactions, including nonpsychotic and psychotic symptoms, were reported in approximately 13% of adults taking at least 50 mg/day of brivaracetam compared with 8% taking placebo.
Psychiatric adverse reactions were also observed in open-label pediatric trials and were generally similar to those observed in adults.
Patients should be advised to report these symptoms immediately to a health care professional, the company noted.
A version of this article first appeared on Medscape.com.
All three brivaracetam formulations (tablets, oral solution, and IV) may now be used. The approval marks the first time that the IV formulation will be available for children, the company said in a news release.
The medication is already approved in the United States as monotherapy and adjunctive therapy in adults with epilepsy.
In an open-label follow-up pediatric study, an estimated 71.4% of patients aged 1 month to 17 years with partial-onset seizures remained on brivaracetam therapy at 1 year, and 64.3% did so at 2 years, the company reported.
“We often see children with seizures hospitalized, so it’s important to have a therapy like Briviact IV that can offer rapid administration in an effective dose when needed and does not require titration,” Raman Sankar, MD, PhD, distinguished professor and chief of pediatric neurology, University of California, Los Angeles, said in the release.
“The availability of the oral dose forms also allows continuity of treatment when these young patients are transitioning from hospital to home,” he added.
Safety profile
Dr. Sankar noted that with approval now of both the IV and oral formulations for partial-onset seizures in such young children, “we have a new option that helps meet a critical need in pediatric epilepsy.”
The most common adverse reactions with brivaracetam include somnolence and sedation, dizziness, fatigue, nausea, and vomiting. In the pediatric clinical trials, the safety profile for pediatric patients was similar to adults.
In the adult trials, psychiatric adverse reactions, including nonpsychotic and psychotic symptoms, were reported in approximately 13% of adults taking at least 50 mg/day of brivaracetam compared with 8% taking placebo.
Psychiatric adverse reactions were also observed in open-label pediatric trials and were generally similar to those observed in adults.
Patients should be advised to report these symptoms immediately to a health care professional, the company noted.
A version of this article first appeared on Medscape.com.
All three brivaracetam formulations (tablets, oral solution, and IV) may now be used. The approval marks the first time that the IV formulation will be available for children, the company said in a news release.
The medication is already approved in the United States as monotherapy and adjunctive therapy in adults with epilepsy.
In an open-label follow-up pediatric study, an estimated 71.4% of patients aged 1 month to 17 years with partial-onset seizures remained on brivaracetam therapy at 1 year, and 64.3% did so at 2 years, the company reported.
“We often see children with seizures hospitalized, so it’s important to have a therapy like Briviact IV that can offer rapid administration in an effective dose when needed and does not require titration,” Raman Sankar, MD, PhD, distinguished professor and chief of pediatric neurology, University of California, Los Angeles, said in the release.
“The availability of the oral dose forms also allows continuity of treatment when these young patients are transitioning from hospital to home,” he added.
Safety profile
Dr. Sankar noted that with approval now of both the IV and oral formulations for partial-onset seizures in such young children, “we have a new option that helps meet a critical need in pediatric epilepsy.”
The most common adverse reactions with brivaracetam include somnolence and sedation, dizziness, fatigue, nausea, and vomiting. In the pediatric clinical trials, the safety profile for pediatric patients was similar to adults.
In the adult trials, psychiatric adverse reactions, including nonpsychotic and psychotic symptoms, were reported in approximately 13% of adults taking at least 50 mg/day of brivaracetam compared with 8% taking placebo.
Psychiatric adverse reactions were also observed in open-label pediatric trials and were generally similar to those observed in adults.
Patients should be advised to report these symptoms immediately to a health care professional, the company noted.
A version of this article first appeared on Medscape.com.











