User login
FDA approves generic drugs for APL
The U.S. Food and Drug Administration (FDA) has now approved three generic arsenic trioxide products for use in patients with acute promyelocytic leukemia (APL).
Two of the products—from Zydus Cadila and Amring Pharmaceuticals—were approved on November 13.
The third—from Fresenius Kabi—was approved in August and launched in the United States last month.
All three injectable arsenic trioxide products (1 mg/mL) are generic versions of Teva’s Trisenox.
Since 2000, Trisenox has been FDA-approved to induce remission and as consolidation therapy for patients with APL who are refractory to, or have relapsed after, retinoid and anthracycline chemotherapy, and whose APL is characterized by presence of the t(15;17) translocation or PML/RAR-alpha gene expression.
In January, the FDA approved Trisenox for use in combination with tretinoin to treat adults with newly diagnosed, low-risk APL with the t(15;17) translocation or PML/RAR-alpha gene expression.
The U.S. Food and Drug Administration (FDA) has now approved three generic arsenic trioxide products for use in patients with acute promyelocytic leukemia (APL).
Two of the products—from Zydus Cadila and Amring Pharmaceuticals—were approved on November 13.
The third—from Fresenius Kabi—was approved in August and launched in the United States last month.
All three injectable arsenic trioxide products (1 mg/mL) are generic versions of Teva’s Trisenox.
Since 2000, Trisenox has been FDA-approved to induce remission and as consolidation therapy for patients with APL who are refractory to, or have relapsed after, retinoid and anthracycline chemotherapy, and whose APL is characterized by presence of the t(15;17) translocation or PML/RAR-alpha gene expression.
In January, the FDA approved Trisenox for use in combination with tretinoin to treat adults with newly diagnosed, low-risk APL with the t(15;17) translocation or PML/RAR-alpha gene expression.
The U.S. Food and Drug Administration (FDA) has now approved three generic arsenic trioxide products for use in patients with acute promyelocytic leukemia (APL).
Two of the products—from Zydus Cadila and Amring Pharmaceuticals—were approved on November 13.
The third—from Fresenius Kabi—was approved in August and launched in the United States last month.
All three injectable arsenic trioxide products (1 mg/mL) are generic versions of Teva’s Trisenox.
Since 2000, Trisenox has been FDA-approved to induce remission and as consolidation therapy for patients with APL who are refractory to, or have relapsed after, retinoid and anthracycline chemotherapy, and whose APL is characterized by presence of the t(15;17) translocation or PML/RAR-alpha gene expression.
In January, the FDA approved Trisenox for use in combination with tretinoin to treat adults with newly diagnosed, low-risk APL with the t(15;17) translocation or PML/RAR-alpha gene expression.
FDA approves generic decitabine for MDS
The U.S. Food and Drug Administration has approved Lupin’s decitabine product, a generic version of Otsuka Pharmaceutical Co. Ltd.’s Dacogen, to treat patients with myelodysplastic syndromes (MDS).
Lupin’s decitabine for injection (50 mg, single-dose vial) is approved to treat patients with intermediate-1, intermediate-2, and high-risk MDS.
This includes previously treated, untreated, de novo, and secondary MDS of all French-American-British subtypes—refractory anemia, refractory anemia with ringed sideroblasts, refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, and chronic myelomonocytic leukemia.
The U.S. Food and Drug Administration has approved Lupin’s decitabine product, a generic version of Otsuka Pharmaceutical Co. Ltd.’s Dacogen, to treat patients with myelodysplastic syndromes (MDS).
Lupin’s decitabine for injection (50 mg, single-dose vial) is approved to treat patients with intermediate-1, intermediate-2, and high-risk MDS.
This includes previously treated, untreated, de novo, and secondary MDS of all French-American-British subtypes—refractory anemia, refractory anemia with ringed sideroblasts, refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, and chronic myelomonocytic leukemia.
The U.S. Food and Drug Administration has approved Lupin’s decitabine product, a generic version of Otsuka Pharmaceutical Co. Ltd.’s Dacogen, to treat patients with myelodysplastic syndromes (MDS).
Lupin’s decitabine for injection (50 mg, single-dose vial) is approved to treat patients with intermediate-1, intermediate-2, and high-risk MDS.
This includes previously treated, untreated, de novo, and secondary MDS of all French-American-British subtypes—refractory anemia, refractory anemia with ringed sideroblasts, refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, and chronic myelomonocytic leukemia.
Score can predict thrombosis in ITP
New research suggests a scoring system can predict the risk of thrombosis in patients with immune thrombocytopenia (ITP) who are taking anticoagulants.
Researchers tested their Thrombosis and Thrombocytopenia (TH2) risk assessment score in a small group of ITP patients on anticoagulants, and the score identified all seven patients who developed thrombosis.
The researchers also found that patients’ TH2 scores changed quickly—within a matter of days—which suggests they should be re-evaluated for thrombosis risk frequently.
Amaris K. Balitsky, MD, of McMaster University in Hamilton, Ontario, Canada, and her colleagues detailed this research in Blood.
About the score
To develop the TH2 score, the researchers conducted a review of the literature and existing tools used to assess the risk of thrombosis and bleeding. The resulting score consists of two thrombosis items and two bleeding items.
The thrombosis items are:
- High thrombotic risk, which includes patients with atrial fibrillation and a CHA2DS2-VASc score greater than five; unprovoked, recurrent, or cancer-associated thrombosis; or antiphospholipid antibody syndrome
- Receipt of ITP therapies known to increase the risk of thrombosis in the previous 14 days or splenectomy in the previous 30 days.
The score’s bleeding items are:
- Platelet count less than 20 x 109/L
- Major bleeding (grade 2 bleeding that does not involve the skin) observed at a clinical encounter.
Each thrombosis item is assigned a score of +1, and each bleeding item is assigned a score of -1. A positive score or score of 0 suggests a net increased risk of thrombosis, and a negative score suggests a net increased risk of bleeding.
Patient population
The researchers tested the TH2 score in patients enrolled in the McMaster ITP Registry from 2010 to 2017.
There were 314 patients enrolled, but only 13 were receiving anticoagulation and had a platelet count less than 50 x 109/L. Six of these patients were receiving anticoagulation for atrial fibrillation and seven for venous thrombosis. Four patients were taking antiplatelet agents as well.
The median follow up was 9 months (interquartile range, 4.5 to 24 months). During that time, there were 41 clinical encounters. Data on treatment decisions and clinical outcomes were available for 32 of these encounters.
Ten of the 13 patients had anticoagulation withheld at some point during their 22 clinical encounters. Major bleeding was present at five of the encounters. At 17 encounters, patients received additional ITP treatments.
Six of the 10 patients who stopped anticoagulation had new thrombotic events, and two of these events were fatal. Three of the patients had thrombotic events even though they resumed anticoagulation.
The remaining three patients (of the 13) did not stop anticoagulation. These patients received additional ITP treatments at six of their 10 clinical encounters.
Major bleeding was present at two of the 10 encounters, and one new thrombotic event occurred in a patient with metastatic squamous cell cancer (despite continued treatment with warfarin).
Testing the score
The TH2 score accurately predicted all seven thrombotic events.
There were four patients who initially had a negative TH2 score, which suggested an increased risk of bleeding.
However, these patients had a positive or 0 score—suggesting an increased risk of thrombosis—when they were assessed again, after their platelet counts increased above 50 x 109/L.
The remaining three patients had initial scores of 0 and subsequent positive scores, both suggesting an increased risk of thrombosis.
The researchers said these findings suggest patients should be re-evaluated for thrombosis risk frequently, as ITP treatments are given and platelet counts increase.
“The results of our study suggest that the risk of thrombosis is high in patients with ITP who have a separate indication for anticoagulation, especially after ITP therapies are administered and the severe thrombocytopenia improves,” the researchers wrote. “Early resumption of anticoagulation should be considered in this population.”
The researchers also noted that this study was limited by its retrospective, single-center design and the small number of patients evaluated. Therefore, the TH2 score should be validated in additional, larger studies.
One researcher reported relationships with Amgen, Novartis, Rigel Pharmaceuticals, UCB, and Principia Biopharma. The other researchers said they had no competing financial interests.
New research suggests a scoring system can predict the risk of thrombosis in patients with immune thrombocytopenia (ITP) who are taking anticoagulants.
Researchers tested their Thrombosis and Thrombocytopenia (TH2) risk assessment score in a small group of ITP patients on anticoagulants, and the score identified all seven patients who developed thrombosis.
The researchers also found that patients’ TH2 scores changed quickly—within a matter of days—which suggests they should be re-evaluated for thrombosis risk frequently.
Amaris K. Balitsky, MD, of McMaster University in Hamilton, Ontario, Canada, and her colleagues detailed this research in Blood.
About the score
To develop the TH2 score, the researchers conducted a review of the literature and existing tools used to assess the risk of thrombosis and bleeding. The resulting score consists of two thrombosis items and two bleeding items.
The thrombosis items are:
- High thrombotic risk, which includes patients with atrial fibrillation and a CHA2DS2-VASc score greater than five; unprovoked, recurrent, or cancer-associated thrombosis; or antiphospholipid antibody syndrome
- Receipt of ITP therapies known to increase the risk of thrombosis in the previous 14 days or splenectomy in the previous 30 days.
The score’s bleeding items are:
- Platelet count less than 20 x 109/L
- Major bleeding (grade 2 bleeding that does not involve the skin) observed at a clinical encounter.
Each thrombosis item is assigned a score of +1, and each bleeding item is assigned a score of -1. A positive score or score of 0 suggests a net increased risk of thrombosis, and a negative score suggests a net increased risk of bleeding.
Patient population
The researchers tested the TH2 score in patients enrolled in the McMaster ITP Registry from 2010 to 2017.
There were 314 patients enrolled, but only 13 were receiving anticoagulation and had a platelet count less than 50 x 109/L. Six of these patients were receiving anticoagulation for atrial fibrillation and seven for venous thrombosis. Four patients were taking antiplatelet agents as well.
The median follow up was 9 months (interquartile range, 4.5 to 24 months). During that time, there were 41 clinical encounters. Data on treatment decisions and clinical outcomes were available for 32 of these encounters.
Ten of the 13 patients had anticoagulation withheld at some point during their 22 clinical encounters. Major bleeding was present at five of the encounters. At 17 encounters, patients received additional ITP treatments.
Six of the 10 patients who stopped anticoagulation had new thrombotic events, and two of these events were fatal. Three of the patients had thrombotic events even though they resumed anticoagulation.
The remaining three patients (of the 13) did not stop anticoagulation. These patients received additional ITP treatments at six of their 10 clinical encounters.
Major bleeding was present at two of the 10 encounters, and one new thrombotic event occurred in a patient with metastatic squamous cell cancer (despite continued treatment with warfarin).
Testing the score
The TH2 score accurately predicted all seven thrombotic events.
There were four patients who initially had a negative TH2 score, which suggested an increased risk of bleeding.
However, these patients had a positive or 0 score—suggesting an increased risk of thrombosis—when they were assessed again, after their platelet counts increased above 50 x 109/L.
The remaining three patients had initial scores of 0 and subsequent positive scores, both suggesting an increased risk of thrombosis.
The researchers said these findings suggest patients should be re-evaluated for thrombosis risk frequently, as ITP treatments are given and platelet counts increase.
“The results of our study suggest that the risk of thrombosis is high in patients with ITP who have a separate indication for anticoagulation, especially after ITP therapies are administered and the severe thrombocytopenia improves,” the researchers wrote. “Early resumption of anticoagulation should be considered in this population.”
The researchers also noted that this study was limited by its retrospective, single-center design and the small number of patients evaluated. Therefore, the TH2 score should be validated in additional, larger studies.
One researcher reported relationships with Amgen, Novartis, Rigel Pharmaceuticals, UCB, and Principia Biopharma. The other researchers said they had no competing financial interests.
New research suggests a scoring system can predict the risk of thrombosis in patients with immune thrombocytopenia (ITP) who are taking anticoagulants.
Researchers tested their Thrombosis and Thrombocytopenia (TH2) risk assessment score in a small group of ITP patients on anticoagulants, and the score identified all seven patients who developed thrombosis.
The researchers also found that patients’ TH2 scores changed quickly—within a matter of days—which suggests they should be re-evaluated for thrombosis risk frequently.
Amaris K. Balitsky, MD, of McMaster University in Hamilton, Ontario, Canada, and her colleagues detailed this research in Blood.
About the score
To develop the TH2 score, the researchers conducted a review of the literature and existing tools used to assess the risk of thrombosis and bleeding. The resulting score consists of two thrombosis items and two bleeding items.
The thrombosis items are:
- High thrombotic risk, which includes patients with atrial fibrillation and a CHA2DS2-VASc score greater than five; unprovoked, recurrent, or cancer-associated thrombosis; or antiphospholipid antibody syndrome
- Receipt of ITP therapies known to increase the risk of thrombosis in the previous 14 days or splenectomy in the previous 30 days.
The score’s bleeding items are:
- Platelet count less than 20 x 109/L
- Major bleeding (grade 2 bleeding that does not involve the skin) observed at a clinical encounter.
Each thrombosis item is assigned a score of +1, and each bleeding item is assigned a score of -1. A positive score or score of 0 suggests a net increased risk of thrombosis, and a negative score suggests a net increased risk of bleeding.
Patient population
The researchers tested the TH2 score in patients enrolled in the McMaster ITP Registry from 2010 to 2017.
There were 314 patients enrolled, but only 13 were receiving anticoagulation and had a platelet count less than 50 x 109/L. Six of these patients were receiving anticoagulation for atrial fibrillation and seven for venous thrombosis. Four patients were taking antiplatelet agents as well.
The median follow up was 9 months (interquartile range, 4.5 to 24 months). During that time, there were 41 clinical encounters. Data on treatment decisions and clinical outcomes were available for 32 of these encounters.
Ten of the 13 patients had anticoagulation withheld at some point during their 22 clinical encounters. Major bleeding was present at five of the encounters. At 17 encounters, patients received additional ITP treatments.
Six of the 10 patients who stopped anticoagulation had new thrombotic events, and two of these events were fatal. Three of the patients had thrombotic events even though they resumed anticoagulation.
The remaining three patients (of the 13) did not stop anticoagulation. These patients received additional ITP treatments at six of their 10 clinical encounters.
Major bleeding was present at two of the 10 encounters, and one new thrombotic event occurred in a patient with metastatic squamous cell cancer (despite continued treatment with warfarin).
Testing the score
The TH2 score accurately predicted all seven thrombotic events.
There were four patients who initially had a negative TH2 score, which suggested an increased risk of bleeding.
However, these patients had a positive or 0 score—suggesting an increased risk of thrombosis—when they were assessed again, after their platelet counts increased above 50 x 109/L.
The remaining three patients had initial scores of 0 and subsequent positive scores, both suggesting an increased risk of thrombosis.
The researchers said these findings suggest patients should be re-evaluated for thrombosis risk frequently, as ITP treatments are given and platelet counts increase.
“The results of our study suggest that the risk of thrombosis is high in patients with ITP who have a separate indication for anticoagulation, especially after ITP therapies are administered and the severe thrombocytopenia improves,” the researchers wrote. “Early resumption of anticoagulation should be considered in this population.”
The researchers also noted that this study was limited by its retrospective, single-center design and the small number of patients evaluated. Therefore, the TH2 score should be validated in additional, larger studies.
One researcher reported relationships with Amgen, Novartis, Rigel Pharmaceuticals, UCB, and Principia Biopharma. The other researchers said they had no competing financial interests.
Health Canada approves two tests for CML patients
Health Canada has approved the use of two tests designed to detect BCR-ABL transcripts in patients with chronic myeloid leukemia (CML)—the MRDx® BCR-ABL Test and the QuantideX qPCR BCR-ABL IS Kit.
MolecularMD’s MRDx® BCR-ABL Test is approved as an aid to monitor tyrosine kinase inhibitor (TKI) therapy in Philadelphia chromosome-positive CML patients.
This test is also approved to identify CML patients in the chronic phase being treated with nilotinib who, after sustaining a deep molecular response of MR4.5, may be eligible to stop treatment and be monitored for treatment-free remission.
Asuragen, Inc.’s QuantideX qPCR BCR-ABL IS Kit is approved for use in CML patients, regardless of whether they are taking a TKI or how their disease is being managed.
The kit is designed to detect and quantify major breakpoint (e13a2, e14a2) BCR-ABL1 fusion transcripts for use in monitoring molecular response.
Both the MRDx® BCR-ABL Test and the QuantideX qPCR BCR-ABL IS Kit are approved in the United States as well.
The QuantideX qPCR BCR-ABL IS Kit is also CE marked for clinical use in the European Union.
Health Canada has approved the use of two tests designed to detect BCR-ABL transcripts in patients with chronic myeloid leukemia (CML)—the MRDx® BCR-ABL Test and the QuantideX qPCR BCR-ABL IS Kit.
MolecularMD’s MRDx® BCR-ABL Test is approved as an aid to monitor tyrosine kinase inhibitor (TKI) therapy in Philadelphia chromosome-positive CML patients.
This test is also approved to identify CML patients in the chronic phase being treated with nilotinib who, after sustaining a deep molecular response of MR4.5, may be eligible to stop treatment and be monitored for treatment-free remission.
Asuragen, Inc.’s QuantideX qPCR BCR-ABL IS Kit is approved for use in CML patients, regardless of whether they are taking a TKI or how their disease is being managed.
The kit is designed to detect and quantify major breakpoint (e13a2, e14a2) BCR-ABL1 fusion transcripts for use in monitoring molecular response.
Both the MRDx® BCR-ABL Test and the QuantideX qPCR BCR-ABL IS Kit are approved in the United States as well.
The QuantideX qPCR BCR-ABL IS Kit is also CE marked for clinical use in the European Union.
Health Canada has approved the use of two tests designed to detect BCR-ABL transcripts in patients with chronic myeloid leukemia (CML)—the MRDx® BCR-ABL Test and the QuantideX qPCR BCR-ABL IS Kit.
MolecularMD’s MRDx® BCR-ABL Test is approved as an aid to monitor tyrosine kinase inhibitor (TKI) therapy in Philadelphia chromosome-positive CML patients.
This test is also approved to identify CML patients in the chronic phase being treated with nilotinib who, after sustaining a deep molecular response of MR4.5, may be eligible to stop treatment and be monitored for treatment-free remission.
Asuragen, Inc.’s QuantideX qPCR BCR-ABL IS Kit is approved for use in CML patients, regardless of whether they are taking a TKI or how their disease is being managed.
The kit is designed to detect and quantify major breakpoint (e13a2, e14a2) BCR-ABL1 fusion transcripts for use in monitoring molecular response.
Both the MRDx® BCR-ABL Test and the QuantideX qPCR BCR-ABL IS Kit are approved in the United States as well.
The QuantideX qPCR BCR-ABL IS Kit is also CE marked for clinical use in the European Union.
Preoperative anemia management saves blood, money
BOSTON—Results of a pilot program suggest preoperative management of anemia can reduce transfusion rates and cut costs, but the effect on patient outcomes isn’t clear.
For this program, anemic patients received dietary guidance and supplementation prior to surgery.
This increased day-of-surgery hemoglobin levels, reduced intraoperative and postoperative transfusions, and resulted in a cost savings of more than $100,000 over the life of the program.
Christine M. Cahill, BSN, MS, RN, of the University of Rochester and Strong Memorial Hospital in Rochester, New York, presented these results at AABB 2018 (abstract PBM4-ST4-22*).
“Anemia has been thought of as a relatively benign thing our patients live with, traditionally, but what we have been finding lately is that anemia is actually more serious than we once thought and is an independent risk factor for hospitalization, readmission, increased patient length of stay, loss of function, and diminished quality of life,” Cahill said.
She added that anemia also increases the likelihood that a patient will require transfusions.
The pilot program was implemented with this in mind. The program, which ran from February 2016 through September 2017, was designed to test the feasibility of diagnosing anemia during a cardiology consult visit and implementing an anemia management plan.
During the study period, 240 patients presenting for elective cardiac surgery were screened for anemia, and 58 were diagnosed as anemic (hemoglobin <12 g/dL). These patients were referred for anemia workups, which showed that 33 patients had iron-deficiency anemia, and 25 had anemia from other causes.
Preoperative anemia management for the iron-deficient patients included oral iron for seven patients, intravenous (IV) iron with or without folate for 20 patients, and oral folate with or without vitamin B12 for five patients. One iron-deficient patient could not have surgery delayed for anemia management.
Of the iron-replete patients, one received oral iron, 17 received folate with or without B12, and seven patients were not treated for anemia.
One iron-deficient patient had a reaction to the infusion and did not receive a scheduled second dose due to the need for immediate surgery. A second patient scheduled for IV iron and folate broke an arm and therefore missed an IV infusion appointment. No other complications or reactions occurred.
Results
The researchers compared the 58 patients from the pilot program to control subjects—patients who underwent cardiac surgery from March through July 2015, matched by age, sex, and procedures.
The anemia management group received 10 red blood cell (RBC) units intraoperatively, compared to 68 intraoperative RBC units for controls. The total number of postoperative RBC units was 13 and 22, respectively.
The rate of RBC transfusions was 24% in the anemia management group and 60% in controls (P<0.0001). The average RBC units per patient was 0.4 and 2.07, respectively (P<0.0001).
Patients in the anemia management program also had significantly higher day-of-surgery hemoglobin than controls—11.01 and 10.16 g/dL, respectively (P<0.001).
The program provided an average per-patient savings in acquisition costs of $367.40, an average transfusion cost savings of $1,837, and a total cost savings of $106,546 over the life of the program.
The key to success of a similar program is “to make sure you do your homework,” Cahill said.
Specifically, she recommended feasibility studies, evaluation of the potential impact of infusions on the service, work flow analyses, and cost analyses. It’s also important to get high-level administrative support as well as buy-in from surgeons and patients, she added.
Future studies should include assessment of patient outcomes, safety, and length of intensive care unit and hospital stay, Cahill emphasized.
This study was internally funded. Cahill reported no conflicts of interest.
*The data in the presentation differ from the abstract.
BOSTON—Results of a pilot program suggest preoperative management of anemia can reduce transfusion rates and cut costs, but the effect on patient outcomes isn’t clear.
For this program, anemic patients received dietary guidance and supplementation prior to surgery.
This increased day-of-surgery hemoglobin levels, reduced intraoperative and postoperative transfusions, and resulted in a cost savings of more than $100,000 over the life of the program.
Christine M. Cahill, BSN, MS, RN, of the University of Rochester and Strong Memorial Hospital in Rochester, New York, presented these results at AABB 2018 (abstract PBM4-ST4-22*).
“Anemia has been thought of as a relatively benign thing our patients live with, traditionally, but what we have been finding lately is that anemia is actually more serious than we once thought and is an independent risk factor for hospitalization, readmission, increased patient length of stay, loss of function, and diminished quality of life,” Cahill said.
She added that anemia also increases the likelihood that a patient will require transfusions.
The pilot program was implemented with this in mind. The program, which ran from February 2016 through September 2017, was designed to test the feasibility of diagnosing anemia during a cardiology consult visit and implementing an anemia management plan.
During the study period, 240 patients presenting for elective cardiac surgery were screened for anemia, and 58 were diagnosed as anemic (hemoglobin <12 g/dL). These patients were referred for anemia workups, which showed that 33 patients had iron-deficiency anemia, and 25 had anemia from other causes.
Preoperative anemia management for the iron-deficient patients included oral iron for seven patients, intravenous (IV) iron with or without folate for 20 patients, and oral folate with or without vitamin B12 for five patients. One iron-deficient patient could not have surgery delayed for anemia management.
Of the iron-replete patients, one received oral iron, 17 received folate with or without B12, and seven patients were not treated for anemia.
One iron-deficient patient had a reaction to the infusion and did not receive a scheduled second dose due to the need for immediate surgery. A second patient scheduled for IV iron and folate broke an arm and therefore missed an IV infusion appointment. No other complications or reactions occurred.
Results
The researchers compared the 58 patients from the pilot program to control subjects—patients who underwent cardiac surgery from March through July 2015, matched by age, sex, and procedures.
The anemia management group received 10 red blood cell (RBC) units intraoperatively, compared to 68 intraoperative RBC units for controls. The total number of postoperative RBC units was 13 and 22, respectively.
The rate of RBC transfusions was 24% in the anemia management group and 60% in controls (P<0.0001). The average RBC units per patient was 0.4 and 2.07, respectively (P<0.0001).
Patients in the anemia management program also had significantly higher day-of-surgery hemoglobin than controls—11.01 and 10.16 g/dL, respectively (P<0.001).
The program provided an average per-patient savings in acquisition costs of $367.40, an average transfusion cost savings of $1,837, and a total cost savings of $106,546 over the life of the program.
The key to success of a similar program is “to make sure you do your homework,” Cahill said.
Specifically, she recommended feasibility studies, evaluation of the potential impact of infusions on the service, work flow analyses, and cost analyses. It’s also important to get high-level administrative support as well as buy-in from surgeons and patients, she added.
Future studies should include assessment of patient outcomes, safety, and length of intensive care unit and hospital stay, Cahill emphasized.
This study was internally funded. Cahill reported no conflicts of interest.
*The data in the presentation differ from the abstract.
BOSTON—Results of a pilot program suggest preoperative management of anemia can reduce transfusion rates and cut costs, but the effect on patient outcomes isn’t clear.
For this program, anemic patients received dietary guidance and supplementation prior to surgery.
This increased day-of-surgery hemoglobin levels, reduced intraoperative and postoperative transfusions, and resulted in a cost savings of more than $100,000 over the life of the program.
Christine M. Cahill, BSN, MS, RN, of the University of Rochester and Strong Memorial Hospital in Rochester, New York, presented these results at AABB 2018 (abstract PBM4-ST4-22*).
“Anemia has been thought of as a relatively benign thing our patients live with, traditionally, but what we have been finding lately is that anemia is actually more serious than we once thought and is an independent risk factor for hospitalization, readmission, increased patient length of stay, loss of function, and diminished quality of life,” Cahill said.
She added that anemia also increases the likelihood that a patient will require transfusions.
The pilot program was implemented with this in mind. The program, which ran from February 2016 through September 2017, was designed to test the feasibility of diagnosing anemia during a cardiology consult visit and implementing an anemia management plan.
During the study period, 240 patients presenting for elective cardiac surgery were screened for anemia, and 58 were diagnosed as anemic (hemoglobin <12 g/dL). These patients were referred for anemia workups, which showed that 33 patients had iron-deficiency anemia, and 25 had anemia from other causes.
Preoperative anemia management for the iron-deficient patients included oral iron for seven patients, intravenous (IV) iron with or without folate for 20 patients, and oral folate with or without vitamin B12 for five patients. One iron-deficient patient could not have surgery delayed for anemia management.
Of the iron-replete patients, one received oral iron, 17 received folate with or without B12, and seven patients were not treated for anemia.
One iron-deficient patient had a reaction to the infusion and did not receive a scheduled second dose due to the need for immediate surgery. A second patient scheduled for IV iron and folate broke an arm and therefore missed an IV infusion appointment. No other complications or reactions occurred.
Results
The researchers compared the 58 patients from the pilot program to control subjects—patients who underwent cardiac surgery from March through July 2015, matched by age, sex, and procedures.
The anemia management group received 10 red blood cell (RBC) units intraoperatively, compared to 68 intraoperative RBC units for controls. The total number of postoperative RBC units was 13 and 22, respectively.
The rate of RBC transfusions was 24% in the anemia management group and 60% in controls (P<0.0001). The average RBC units per patient was 0.4 and 2.07, respectively (P<0.0001).
Patients in the anemia management program also had significantly higher day-of-surgery hemoglobin than controls—11.01 and 10.16 g/dL, respectively (P<0.001).
The program provided an average per-patient savings in acquisition costs of $367.40, an average transfusion cost savings of $1,837, and a total cost savings of $106,546 over the life of the program.
The key to success of a similar program is “to make sure you do your homework,” Cahill said.
Specifically, she recommended feasibility studies, evaluation of the potential impact of infusions on the service, work flow analyses, and cost analyses. It’s also important to get high-level administrative support as well as buy-in from surgeons and patients, she added.
Future studies should include assessment of patient outcomes, safety, and length of intensive care unit and hospital stay, Cahill emphasized.
This study was internally funded. Cahill reported no conflicts of interest.
*The data in the presentation differ from the abstract.
Markers associated with efficacy of malaria vaccine
New research has revealed markers associated with efficacy of the RTS,S/AS01E malaria vaccine (Mosquirix™).
The study suggests malaria protection depends on the amount and subclass of antibodies generated upon vaccination and on previous exposure levels to the malaria parasite.
Researchers believe these findings, published in BMC Medicine, could aid the development of more effective vaccines and guide efforts to improve the effectiveness of RTS,S.
The RTS,S vaccine demonstrated partial effectiveness in a phase 3 study—31% in infants ages 6 weeks to 12 weeks and 56% in children ages 5 months to 17 months.
Carlota Dobaño Lázaro, PhD, of ISGlobal in Barcelona, Spain, and her colleagues have been working to understand the reasons for this variability and identify vaccine protection correlates.
The team used a quantitative assay to investigate the levels and types of antibodies induced by RTS,S. In particular, they measured total IgM, IgG, and IgG1–4 subclass antibodies to hepatitis B surface antigen (HBsAg) and three constructs of the Plasmodium falciparum circumsporozoite protein (CSP).
The researchers analyzed serum and plasma from 195 infants and children from Kintampo, Ghana (an area with high malaria transmission) and Manhiça, Mozambique (low malaria transmission), who were vaccinated during the phase 3 trial for RTS,S.
The results confirmed that RTS,S induces significant levels of IgG antibodies against both CSP and HBsAg, which are higher in children than in infants.
The researchers found that higher HBsAg antibody levels post-vaccination were associated with protection from malaria.
However, the same could not be said for all subclasses of CSP antibodies. Higher levels of IgG1 and IgG3 antibodies were associated with protection, while higher levels of IgG2 and IgG4 were associated with a greater risk of developing malaria.
“The balance between the different subclasses seems to be more important than the total IgG levels,” said study author Itzi Ubillos Escriche, of ISGlobal.
“This could be because IgG1 and IgG3 antibodies have the capacity to stick to the parasite and give an ‘eat-me’ signal to cells of the immune system.”
The results also showed that subjects with higher pre-vaccine levels of anti-P falciparum and anti-CSP antibodies were less protected against malaria post-vaccination.
“This means that the vaccine will exert a larger benefit to infants who have not been exposed to the parasite in utero or during the first weeks of life,” Dr. Dobaño Lázaro said.
“This study . . . identifies new correlates of vaccine success and failure in African children and provides a basis for designing more efficacious vaccines.”
This research was funded by the National Institutes of Health-National Institute of Allergy and Infectious Diseases, PATH Malaria Vaccine Initiative, Ministerio de Economía y Competitividad, and EVIMalaR and AGAUR-Catalonia. ISGlobal is a member of the CERCA Program, Generalitat de Catalunya.
The authors said they have no competing interests.
The phase 3 trial of RTS,S was supported by GlaxoSmithKline Biologicals SA and the PATH Malaria Vaccine Initiative.
New research has revealed markers associated with efficacy of the RTS,S/AS01E malaria vaccine (Mosquirix™).
The study suggests malaria protection depends on the amount and subclass of antibodies generated upon vaccination and on previous exposure levels to the malaria parasite.
Researchers believe these findings, published in BMC Medicine, could aid the development of more effective vaccines and guide efforts to improve the effectiveness of RTS,S.
The RTS,S vaccine demonstrated partial effectiveness in a phase 3 study—31% in infants ages 6 weeks to 12 weeks and 56% in children ages 5 months to 17 months.
Carlota Dobaño Lázaro, PhD, of ISGlobal in Barcelona, Spain, and her colleagues have been working to understand the reasons for this variability and identify vaccine protection correlates.
The team used a quantitative assay to investigate the levels and types of antibodies induced by RTS,S. In particular, they measured total IgM, IgG, and IgG1–4 subclass antibodies to hepatitis B surface antigen (HBsAg) and three constructs of the Plasmodium falciparum circumsporozoite protein (CSP).
The researchers analyzed serum and plasma from 195 infants and children from Kintampo, Ghana (an area with high malaria transmission) and Manhiça, Mozambique (low malaria transmission), who were vaccinated during the phase 3 trial for RTS,S.
The results confirmed that RTS,S induces significant levels of IgG antibodies against both CSP and HBsAg, which are higher in children than in infants.
The researchers found that higher HBsAg antibody levels post-vaccination were associated with protection from malaria.
However, the same could not be said for all subclasses of CSP antibodies. Higher levels of IgG1 and IgG3 antibodies were associated with protection, while higher levels of IgG2 and IgG4 were associated with a greater risk of developing malaria.
“The balance between the different subclasses seems to be more important than the total IgG levels,” said study author Itzi Ubillos Escriche, of ISGlobal.
“This could be because IgG1 and IgG3 antibodies have the capacity to stick to the parasite and give an ‘eat-me’ signal to cells of the immune system.”
The results also showed that subjects with higher pre-vaccine levels of anti-P falciparum and anti-CSP antibodies were less protected against malaria post-vaccination.
“This means that the vaccine will exert a larger benefit to infants who have not been exposed to the parasite in utero or during the first weeks of life,” Dr. Dobaño Lázaro said.
“This study . . . identifies new correlates of vaccine success and failure in African children and provides a basis for designing more efficacious vaccines.”
This research was funded by the National Institutes of Health-National Institute of Allergy and Infectious Diseases, PATH Malaria Vaccine Initiative, Ministerio de Economía y Competitividad, and EVIMalaR and AGAUR-Catalonia. ISGlobal is a member of the CERCA Program, Generalitat de Catalunya.
The authors said they have no competing interests.
The phase 3 trial of RTS,S was supported by GlaxoSmithKline Biologicals SA and the PATH Malaria Vaccine Initiative.
New research has revealed markers associated with efficacy of the RTS,S/AS01E malaria vaccine (Mosquirix™).
The study suggests malaria protection depends on the amount and subclass of antibodies generated upon vaccination and on previous exposure levels to the malaria parasite.
Researchers believe these findings, published in BMC Medicine, could aid the development of more effective vaccines and guide efforts to improve the effectiveness of RTS,S.
The RTS,S vaccine demonstrated partial effectiveness in a phase 3 study—31% in infants ages 6 weeks to 12 weeks and 56% in children ages 5 months to 17 months.
Carlota Dobaño Lázaro, PhD, of ISGlobal in Barcelona, Spain, and her colleagues have been working to understand the reasons for this variability and identify vaccine protection correlates.
The team used a quantitative assay to investigate the levels and types of antibodies induced by RTS,S. In particular, they measured total IgM, IgG, and IgG1–4 subclass antibodies to hepatitis B surface antigen (HBsAg) and three constructs of the Plasmodium falciparum circumsporozoite protein (CSP).
The researchers analyzed serum and plasma from 195 infants and children from Kintampo, Ghana (an area with high malaria transmission) and Manhiça, Mozambique (low malaria transmission), who were vaccinated during the phase 3 trial for RTS,S.
The results confirmed that RTS,S induces significant levels of IgG antibodies against both CSP and HBsAg, which are higher in children than in infants.
The researchers found that higher HBsAg antibody levels post-vaccination were associated with protection from malaria.
However, the same could not be said for all subclasses of CSP antibodies. Higher levels of IgG1 and IgG3 antibodies were associated with protection, while higher levels of IgG2 and IgG4 were associated with a greater risk of developing malaria.
“The balance between the different subclasses seems to be more important than the total IgG levels,” said study author Itzi Ubillos Escriche, of ISGlobal.
“This could be because IgG1 and IgG3 antibodies have the capacity to stick to the parasite and give an ‘eat-me’ signal to cells of the immune system.”
The results also showed that subjects with higher pre-vaccine levels of anti-P falciparum and anti-CSP antibodies were less protected against malaria post-vaccination.
“This means that the vaccine will exert a larger benefit to infants who have not been exposed to the parasite in utero or during the first weeks of life,” Dr. Dobaño Lázaro said.
“This study . . . identifies new correlates of vaccine success and failure in African children and provides a basis for designing more efficacious vaccines.”
This research was funded by the National Institutes of Health-National Institute of Allergy and Infectious Diseases, PATH Malaria Vaccine Initiative, Ministerio de Economía y Competitividad, and EVIMalaR and AGAUR-Catalonia. ISGlobal is a member of the CERCA Program, Generalitat de Catalunya.
The authors said they have no competing interests.
The phase 3 trial of RTS,S was supported by GlaxoSmithKline Biologicals SA and the PATH Malaria Vaccine Initiative.
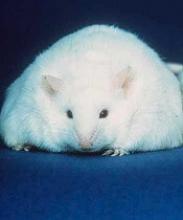
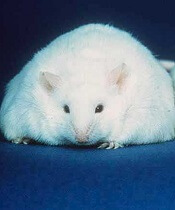
Diet change can improve survival in obese mice with ALL
Switching to a low-fat diet can improve survival in obese mice with acute lymphoblastic leukemia (ALL), according to new research.
Diet-induced obese (DIO) mice with ALL had a survival rate of 17% if they remained on a high-fat diet while treated with vincristine, but survival rose to 92% for mice that were switched to a low-fat diet before treatment.
However, the dietary switch did not impact the survival of DIO mice treated with dexamethasone or L-asparaginase monotherapy.
Researchers reported these findings in Cancer & Metabolism.
“The most exciting thing, to me, about this study is the fact that this shows that a dietary intervention could potentially help us kill leukemia cells in children with acute lymphoblastic leukemia,” said study author Steven Mittelman, MD, PhD, of the University of Southern California, Los Angeles.
“The current treatments for leukemia are very toxic, so finding a way to use a healthy diet, without increasing the toxicity of therapy to treat people with cancer, would be incredible.”
Building on previous research that showed obesity reduced the effectiveness of chemotherapeutic drugs in children with leukemia, the researchers tested whether a dietary intervention could improve ALL outcomes in obese mice.
Methods
The team used NOD/SCID IL2-receptor gamma chain knockout mice raised on either a 60% or 10% fat-calorie diet. Only male mice were studied because female mice do not become as significantly obese on the high-fat diet.
The researchers implanted GFP+ pre-B-cell ALL transgenic mouse cells into DIO and control mice at about 20 weeks old.
Six to seven days after ALL implantation, the researchers randomized the DIO mice to continue their high-fat diet or switch to the control diet (10% calories from fat).
In some experiments, mice received monotherapy with vincristine, and other experiments used L-asparaginase or dexamethasone.
In additional experiments, the researchers implanted DIO and control mice with patient-derived ALL cells with normal karyotype.
After 17 days of engraftment, the researchers switched half the DIO mice to the control diet. The next day, they treated these mice with vincristine, L-asparaginase, or dexamethasone for 4 weeks.
The team monitored the mice daily for food intake, body weight, and onset of progressive leukemia.
Results
Diet had no impact on DIO or control mice that did not receive chemotherapy. Time to progression was the same in these mice.
When vincristine was started on day 7 after ALL implantation, DIO mice switched to the low-fat diet had the best survival, even better than the control mice.
Overall survival was 92% for the DIO mice that switched diets, 42% for the control group, and 17% for the DIO group maintained on the high-fat diet.
Survival experiments performed using L-asparaginase or dexamethasone monotherapy showed no impact of switching diets. The researchers reported there was “no detectable effect on survival in these experiments.”
The team believes this is the first study to test a diet intervention on treatment outcome in hematologic malignancy.
The researchers plan to study dietary intervention further in both obese and non-obese patients.
This study was supported by the National Cancer Institute and funds from the Saban Research Institute, Children’s Hospital of Los Angeles. The researchers had no competing interests to declare.
Switching to a low-fat diet can improve survival in obese mice with acute lymphoblastic leukemia (ALL), according to new research.
Diet-induced obese (DIO) mice with ALL had a survival rate of 17% if they remained on a high-fat diet while treated with vincristine, but survival rose to 92% for mice that were switched to a low-fat diet before treatment.
However, the dietary switch did not impact the survival of DIO mice treated with dexamethasone or L-asparaginase monotherapy.
Researchers reported these findings in Cancer & Metabolism.
“The most exciting thing, to me, about this study is the fact that this shows that a dietary intervention could potentially help us kill leukemia cells in children with acute lymphoblastic leukemia,” said study author Steven Mittelman, MD, PhD, of the University of Southern California, Los Angeles.
“The current treatments for leukemia are very toxic, so finding a way to use a healthy diet, without increasing the toxicity of therapy to treat people with cancer, would be incredible.”
Building on previous research that showed obesity reduced the effectiveness of chemotherapeutic drugs in children with leukemia, the researchers tested whether a dietary intervention could improve ALL outcomes in obese mice.
Methods
The team used NOD/SCID IL2-receptor gamma chain knockout mice raised on either a 60% or 10% fat-calorie diet. Only male mice were studied because female mice do not become as significantly obese on the high-fat diet.
The researchers implanted GFP+ pre-B-cell ALL transgenic mouse cells into DIO and control mice at about 20 weeks old.
Six to seven days after ALL implantation, the researchers randomized the DIO mice to continue their high-fat diet or switch to the control diet (10% calories from fat).
In some experiments, mice received monotherapy with vincristine, and other experiments used L-asparaginase or dexamethasone.
In additional experiments, the researchers implanted DIO and control mice with patient-derived ALL cells with normal karyotype.
After 17 days of engraftment, the researchers switched half the DIO mice to the control diet. The next day, they treated these mice with vincristine, L-asparaginase, or dexamethasone for 4 weeks.
The team monitored the mice daily for food intake, body weight, and onset of progressive leukemia.
Results
Diet had no impact on DIO or control mice that did not receive chemotherapy. Time to progression was the same in these mice.
When vincristine was started on day 7 after ALL implantation, DIO mice switched to the low-fat diet had the best survival, even better than the control mice.
Overall survival was 92% for the DIO mice that switched diets, 42% for the control group, and 17% for the DIO group maintained on the high-fat diet.
Survival experiments performed using L-asparaginase or dexamethasone monotherapy showed no impact of switching diets. The researchers reported there was “no detectable effect on survival in these experiments.”
The team believes this is the first study to test a diet intervention on treatment outcome in hematologic malignancy.
The researchers plan to study dietary intervention further in both obese and non-obese patients.
This study was supported by the National Cancer Institute and funds from the Saban Research Institute, Children’s Hospital of Los Angeles. The researchers had no competing interests to declare.
Switching to a low-fat diet can improve survival in obese mice with acute lymphoblastic leukemia (ALL), according to new research.
Diet-induced obese (DIO) mice with ALL had a survival rate of 17% if they remained on a high-fat diet while treated with vincristine, but survival rose to 92% for mice that were switched to a low-fat diet before treatment.
However, the dietary switch did not impact the survival of DIO mice treated with dexamethasone or L-asparaginase monotherapy.
Researchers reported these findings in Cancer & Metabolism.
“The most exciting thing, to me, about this study is the fact that this shows that a dietary intervention could potentially help us kill leukemia cells in children with acute lymphoblastic leukemia,” said study author Steven Mittelman, MD, PhD, of the University of Southern California, Los Angeles.
“The current treatments for leukemia are very toxic, so finding a way to use a healthy diet, without increasing the toxicity of therapy to treat people with cancer, would be incredible.”
Building on previous research that showed obesity reduced the effectiveness of chemotherapeutic drugs in children with leukemia, the researchers tested whether a dietary intervention could improve ALL outcomes in obese mice.
Methods
The team used NOD/SCID IL2-receptor gamma chain knockout mice raised on either a 60% or 10% fat-calorie diet. Only male mice were studied because female mice do not become as significantly obese on the high-fat diet.
The researchers implanted GFP+ pre-B-cell ALL transgenic mouse cells into DIO and control mice at about 20 weeks old.
Six to seven days after ALL implantation, the researchers randomized the DIO mice to continue their high-fat diet or switch to the control diet (10% calories from fat).
In some experiments, mice received monotherapy with vincristine, and other experiments used L-asparaginase or dexamethasone.
In additional experiments, the researchers implanted DIO and control mice with patient-derived ALL cells with normal karyotype.
After 17 days of engraftment, the researchers switched half the DIO mice to the control diet. The next day, they treated these mice with vincristine, L-asparaginase, or dexamethasone for 4 weeks.
The team monitored the mice daily for food intake, body weight, and onset of progressive leukemia.
Results
Diet had no impact on DIO or control mice that did not receive chemotherapy. Time to progression was the same in these mice.
When vincristine was started on day 7 after ALL implantation, DIO mice switched to the low-fat diet had the best survival, even better than the control mice.
Overall survival was 92% for the DIO mice that switched diets, 42% for the control group, and 17% for the DIO group maintained on the high-fat diet.
Survival experiments performed using L-asparaginase or dexamethasone monotherapy showed no impact of switching diets. The researchers reported there was “no detectable effect on survival in these experiments.”
The team believes this is the first study to test a diet intervention on treatment outcome in hematologic malignancy.
The researchers plan to study dietary intervention further in both obese and non-obese patients.
This study was supported by the National Cancer Institute and funds from the Saban Research Institute, Children’s Hospital of Los Angeles. The researchers had no competing interests to declare.
ADAR1 linked to MM pathogenesis, outcomes
Overly zealous editing of messenger RNA in multiple myeloma (MM) cells contributes to MM pathogenesis and is associated with poor outcomes after certain treatments, investigators contend.
The team found evidence to suggest that overexpression of the RNA editing enzyme ADAR1 leads to hyperediting of the MM transcriptome that appears related to a drug-resistant disease phenotype and worse prognosis.
Phaik Ju Teoh, PhD, of the Cancer Science Institute of Singapore, and colleagues reported these findings in Blood.
The investigators implicate aberrant editing of adenosine (A) to inosine (I) in malignant plasma cells and its effects on NEIL1, a gene that encodes proteins involved in base-excision repair of DNA, as important mechanisms in MM pathogenesis.
A to I editing is the most prevalent form of RNA editing in humans, and aberrant editing mediated by ADAR1 has recently been linked to the development of several cancer types, the investigators noted.
To see whether this process may also be involved in MM, the investigators examined whole blood or bone marrow samples from healthy volunteers and MM patients.
The team found that ADAR1 was overexpressed in MM cells compared to nonmalignant plasma cells.
Furthermore, ADAR1 was expressed at higher levels in patients with newly diagnosed or relapsed MM compared to patients who had smoldering myeloma or monoclonal gammopathy of undetermined significance.
Response to treatment
The investigators also assessed ADAR1 expression in relation to MM patients’ responsiveness to treatment using data from the CoMMpass study.
The team found that patients with poor responses (stable or progressive disease) to bortezomib-based and immunomodulatory-based therapies had high ADAR1 mRNA.
There was no correlation between ADAR1 and responsiveness to carfilzomib-based treatments, but the investigators said this may be because of the relatively lower number of patients who received carfilzomib in this study.
The investigators also found that bortezomib was more effective in inhibiting the growth of MM cells with low ADAR1, and bortezomib-treated cells showed downregulation of ADAR1 expression in a dose- and time-dependent manner.
ADAR1-mediated editing
The investigators determined that ADAR1 directly regulates hyperediting of the MM transcriptome. This was evidenced by a significant increase in A to guanosine (G) editing in newly diagnosed and relapsed MM samples, compared with normal plasma cells.
The team confirmed this finding by observing the effects of ADAR1 levels on editing events across the transcriptome.
The investigators followed this observation with experiments to see whether ADAR1-mediated editing contributes to oncogenesis in MM cells. The MM growth rate slowed when the team silenced ADAR1, and introducing wild-type ADAR1 into cells promoted growth and proliferation.
The investigators also identified NEIL1 as an important ADAR1 editing target in MM. The editing compromised NEIL1’s ability to accurately repair DNA damage.
This study was supported by the National Research Foundation Singapore, the Singapore Ministry of Education, and the National University of Singapore. The investigators reported no competing financial interests.
Overly zealous editing of messenger RNA in multiple myeloma (MM) cells contributes to MM pathogenesis and is associated with poor outcomes after certain treatments, investigators contend.
The team found evidence to suggest that overexpression of the RNA editing enzyme ADAR1 leads to hyperediting of the MM transcriptome that appears related to a drug-resistant disease phenotype and worse prognosis.
Phaik Ju Teoh, PhD, of the Cancer Science Institute of Singapore, and colleagues reported these findings in Blood.
The investigators implicate aberrant editing of adenosine (A) to inosine (I) in malignant plasma cells and its effects on NEIL1, a gene that encodes proteins involved in base-excision repair of DNA, as important mechanisms in MM pathogenesis.
A to I editing is the most prevalent form of RNA editing in humans, and aberrant editing mediated by ADAR1 has recently been linked to the development of several cancer types, the investigators noted.
To see whether this process may also be involved in MM, the investigators examined whole blood or bone marrow samples from healthy volunteers and MM patients.
The team found that ADAR1 was overexpressed in MM cells compared to nonmalignant plasma cells.
Furthermore, ADAR1 was expressed at higher levels in patients with newly diagnosed or relapsed MM compared to patients who had smoldering myeloma or monoclonal gammopathy of undetermined significance.
Response to treatment
The investigators also assessed ADAR1 expression in relation to MM patients’ responsiveness to treatment using data from the CoMMpass study.
The team found that patients with poor responses (stable or progressive disease) to bortezomib-based and immunomodulatory-based therapies had high ADAR1 mRNA.
There was no correlation between ADAR1 and responsiveness to carfilzomib-based treatments, but the investigators said this may be because of the relatively lower number of patients who received carfilzomib in this study.
The investigators also found that bortezomib was more effective in inhibiting the growth of MM cells with low ADAR1, and bortezomib-treated cells showed downregulation of ADAR1 expression in a dose- and time-dependent manner.
ADAR1-mediated editing
The investigators determined that ADAR1 directly regulates hyperediting of the MM transcriptome. This was evidenced by a significant increase in A to guanosine (G) editing in newly diagnosed and relapsed MM samples, compared with normal plasma cells.
The team confirmed this finding by observing the effects of ADAR1 levels on editing events across the transcriptome.
The investigators followed this observation with experiments to see whether ADAR1-mediated editing contributes to oncogenesis in MM cells. The MM growth rate slowed when the team silenced ADAR1, and introducing wild-type ADAR1 into cells promoted growth and proliferation.
The investigators also identified NEIL1 as an important ADAR1 editing target in MM. The editing compromised NEIL1’s ability to accurately repair DNA damage.
This study was supported by the National Research Foundation Singapore, the Singapore Ministry of Education, and the National University of Singapore. The investigators reported no competing financial interests.
Overly zealous editing of messenger RNA in multiple myeloma (MM) cells contributes to MM pathogenesis and is associated with poor outcomes after certain treatments, investigators contend.
The team found evidence to suggest that overexpression of the RNA editing enzyme ADAR1 leads to hyperediting of the MM transcriptome that appears related to a drug-resistant disease phenotype and worse prognosis.
Phaik Ju Teoh, PhD, of the Cancer Science Institute of Singapore, and colleagues reported these findings in Blood.
The investigators implicate aberrant editing of adenosine (A) to inosine (I) in malignant plasma cells and its effects on NEIL1, a gene that encodes proteins involved in base-excision repair of DNA, as important mechanisms in MM pathogenesis.
A to I editing is the most prevalent form of RNA editing in humans, and aberrant editing mediated by ADAR1 has recently been linked to the development of several cancer types, the investigators noted.
To see whether this process may also be involved in MM, the investigators examined whole blood or bone marrow samples from healthy volunteers and MM patients.
The team found that ADAR1 was overexpressed in MM cells compared to nonmalignant plasma cells.
Furthermore, ADAR1 was expressed at higher levels in patients with newly diagnosed or relapsed MM compared to patients who had smoldering myeloma or monoclonal gammopathy of undetermined significance.
Response to treatment
The investigators also assessed ADAR1 expression in relation to MM patients’ responsiveness to treatment using data from the CoMMpass study.
The team found that patients with poor responses (stable or progressive disease) to bortezomib-based and immunomodulatory-based therapies had high ADAR1 mRNA.
There was no correlation between ADAR1 and responsiveness to carfilzomib-based treatments, but the investigators said this may be because of the relatively lower number of patients who received carfilzomib in this study.
The investigators also found that bortezomib was more effective in inhibiting the growth of MM cells with low ADAR1, and bortezomib-treated cells showed downregulation of ADAR1 expression in a dose- and time-dependent manner.
ADAR1-mediated editing
The investigators determined that ADAR1 directly regulates hyperediting of the MM transcriptome. This was evidenced by a significant increase in A to guanosine (G) editing in newly diagnosed and relapsed MM samples, compared with normal plasma cells.
The team confirmed this finding by observing the effects of ADAR1 levels on editing events across the transcriptome.
The investigators followed this observation with experiments to see whether ADAR1-mediated editing contributes to oncogenesis in MM cells. The MM growth rate slowed when the team silenced ADAR1, and introducing wild-type ADAR1 into cells promoted growth and proliferation.
The investigators also identified NEIL1 as an important ADAR1 editing target in MM. The editing compromised NEIL1’s ability to accurately repair DNA damage.
This study was supported by the National Research Foundation Singapore, the Singapore Ministry of Education, and the National University of Singapore. The investigators reported no competing financial interests.
Study supports PBM program for HSCT recipients
BOSTON—A blood management program for patients undergoing hematopoietic stem cell transplant (HSCT) can reduce inappropriate transfusions and costs without compromising patient outcomes, a new study suggests.
Researchers retrospectively compared outcomes before and after implementation of a patient blood management (PBM) program at a single institution.
After the program was implemented, the number of transfusions and the units transfused declined without affecting patient mortality, intensive care unit (ICU) admission rates, or other transfusion-related complications.
In addition, the program saved the hospital more than $600,000 over a year.
Nilesh Jambhekar, MD, of the Mayo Clinic in Rochester, Minnesota, reported these results in a presentation at AABB 2018 (abstract PBM3-ST4-22*).
Study design
Dr. Jambhekar and his colleagues looked at blood product use both before and after the Mayo Clinic started a PBM program that included emphasis on AABB best practice guidelines and electronic clinical-decision support for transfusion orders.
The researchers evaluated the frequency and proportion of red blood cell (RBC) and platelet transfusions, total transfusion quantities, transfusions that occurred outside of the clinical guidelines, and the activity-based costs of transfusions.
Dr. Jambhekar acknowledged that the study relied on rigid hemoglobin and platelet thresholds when considering transfusions conducted outside of the guidelines, defined as RBCs administered for hemoglobin values greater than 7 g/dL and platelet transfusions for platelet counts greater than 10 x 109/L.
He noted, however, that the researchers conducted sensitivity analyses to account for exceptions such as patients with coronary disease or neutropenic fever.
The patient-centered outcomes the researchers evaluated included mortality, hospital and ICU admission rates, transfusion reactions, cerebrovascular and coronary ischemic events, and infections.
The study included data on 360 adults older than 18 who underwent HSCT in 2013, before the PBM program was implemented, and 368 transplanted in 2015, after implementation.
In each cohort, patients were followed out to 90 days after transplant.
Results
The total number of platelet units transfused dropped from 1,660 pre-PBM program to 1,417 post-PBM implementation. The total number of RBC units dropped from 1,158 to 826.
The researchers also saw changes in the proportions of inappropriate (outside guidelines) transfusions between the two time periods.
In 2013, 94.2% of RBC transfusions occurred outside the guidelines, compared with 35.4% in 2015 (P<0.0001). Similarly, the proportion of inappropriate platelet transfusions declined from 73.4% to 48.7% over the same time period (P<0.0001).
In addition, all-cause mortality at 3 months was significantly lower after the PBM program was introduced. The 3-month mortality rate was 30.7% for the 2013 cohort and 20.2% for the 2015 cohort (P=0.001).
Dr. Jambhekar noted that, in a multivariable analysis accounting for baseline differences between the groups, mortality for patients treated before the PBM program remained significantly higher, with an odds ratio of 1.85 (P=0.0008).
Neither hospital nor ICU admission within 30 days differed significantly between the groups, and there were no significant between-group differences in hospital or ICU lengths of stay.
Likewise, there were no significant between-group differences in myocardial infarctions, cerebrovascular events, sepsis, and febrile or allergic transfusion reactions.
Dr. Jambhekar noted that this study was retrospective in design and therefore could not fully account for potential confounders. It’s also unclear whether the results could be generalized for adoption by other institutions.
“In general, PBM implementation is probably helpful in reducing both platelet and PRBC [packed red blood cell] utilization, but it’s not an easy thing to do,” Dr. Jambhekar said.
“It requires institutional buy-in and key players to make it happen. Ongoing PBM-related activities like surveillance, education, and clinical decision feedback are critical to maintaining success that we’ve had.”
This study was internally funded. Dr. Jambhekar reported having nothing to disclose.
*Data presented differ from the abstract.
BOSTON—A blood management program for patients undergoing hematopoietic stem cell transplant (HSCT) can reduce inappropriate transfusions and costs without compromising patient outcomes, a new study suggests.
Researchers retrospectively compared outcomes before and after implementation of a patient blood management (PBM) program at a single institution.
After the program was implemented, the number of transfusions and the units transfused declined without affecting patient mortality, intensive care unit (ICU) admission rates, or other transfusion-related complications.
In addition, the program saved the hospital more than $600,000 over a year.
Nilesh Jambhekar, MD, of the Mayo Clinic in Rochester, Minnesota, reported these results in a presentation at AABB 2018 (abstract PBM3-ST4-22*).
Study design
Dr. Jambhekar and his colleagues looked at blood product use both before and after the Mayo Clinic started a PBM program that included emphasis on AABB best practice guidelines and electronic clinical-decision support for transfusion orders.
The researchers evaluated the frequency and proportion of red blood cell (RBC) and platelet transfusions, total transfusion quantities, transfusions that occurred outside of the clinical guidelines, and the activity-based costs of transfusions.
Dr. Jambhekar acknowledged that the study relied on rigid hemoglobin and platelet thresholds when considering transfusions conducted outside of the guidelines, defined as RBCs administered for hemoglobin values greater than 7 g/dL and platelet transfusions for platelet counts greater than 10 x 109/L.
He noted, however, that the researchers conducted sensitivity analyses to account for exceptions such as patients with coronary disease or neutropenic fever.
The patient-centered outcomes the researchers evaluated included mortality, hospital and ICU admission rates, transfusion reactions, cerebrovascular and coronary ischemic events, and infections.
The study included data on 360 adults older than 18 who underwent HSCT in 2013, before the PBM program was implemented, and 368 transplanted in 2015, after implementation.
In each cohort, patients were followed out to 90 days after transplant.
Results
The total number of platelet units transfused dropped from 1,660 pre-PBM program to 1,417 post-PBM implementation. The total number of RBC units dropped from 1,158 to 826.
The researchers also saw changes in the proportions of inappropriate (outside guidelines) transfusions between the two time periods.
In 2013, 94.2% of RBC transfusions occurred outside the guidelines, compared with 35.4% in 2015 (P<0.0001). Similarly, the proportion of inappropriate platelet transfusions declined from 73.4% to 48.7% over the same time period (P<0.0001).
In addition, all-cause mortality at 3 months was significantly lower after the PBM program was introduced. The 3-month mortality rate was 30.7% for the 2013 cohort and 20.2% for the 2015 cohort (P=0.001).
Dr. Jambhekar noted that, in a multivariable analysis accounting for baseline differences between the groups, mortality for patients treated before the PBM program remained significantly higher, with an odds ratio of 1.85 (P=0.0008).
Neither hospital nor ICU admission within 30 days differed significantly between the groups, and there were no significant between-group differences in hospital or ICU lengths of stay.
Likewise, there were no significant between-group differences in myocardial infarctions, cerebrovascular events, sepsis, and febrile or allergic transfusion reactions.
Dr. Jambhekar noted that this study was retrospective in design and therefore could not fully account for potential confounders. It’s also unclear whether the results could be generalized for adoption by other institutions.
“In general, PBM implementation is probably helpful in reducing both platelet and PRBC [packed red blood cell] utilization, but it’s not an easy thing to do,” Dr. Jambhekar said.
“It requires institutional buy-in and key players to make it happen. Ongoing PBM-related activities like surveillance, education, and clinical decision feedback are critical to maintaining success that we’ve had.”
This study was internally funded. Dr. Jambhekar reported having nothing to disclose.
*Data presented differ from the abstract.
BOSTON—A blood management program for patients undergoing hematopoietic stem cell transplant (HSCT) can reduce inappropriate transfusions and costs without compromising patient outcomes, a new study suggests.
Researchers retrospectively compared outcomes before and after implementation of a patient blood management (PBM) program at a single institution.
After the program was implemented, the number of transfusions and the units transfused declined without affecting patient mortality, intensive care unit (ICU) admission rates, or other transfusion-related complications.
In addition, the program saved the hospital more than $600,000 over a year.
Nilesh Jambhekar, MD, of the Mayo Clinic in Rochester, Minnesota, reported these results in a presentation at AABB 2018 (abstract PBM3-ST4-22*).
Study design
Dr. Jambhekar and his colleagues looked at blood product use both before and after the Mayo Clinic started a PBM program that included emphasis on AABB best practice guidelines and electronic clinical-decision support for transfusion orders.
The researchers evaluated the frequency and proportion of red blood cell (RBC) and platelet transfusions, total transfusion quantities, transfusions that occurred outside of the clinical guidelines, and the activity-based costs of transfusions.
Dr. Jambhekar acknowledged that the study relied on rigid hemoglobin and platelet thresholds when considering transfusions conducted outside of the guidelines, defined as RBCs administered for hemoglobin values greater than 7 g/dL and platelet transfusions for platelet counts greater than 10 x 109/L.
He noted, however, that the researchers conducted sensitivity analyses to account for exceptions such as patients with coronary disease or neutropenic fever.
The patient-centered outcomes the researchers evaluated included mortality, hospital and ICU admission rates, transfusion reactions, cerebrovascular and coronary ischemic events, and infections.
The study included data on 360 adults older than 18 who underwent HSCT in 2013, before the PBM program was implemented, and 368 transplanted in 2015, after implementation.
In each cohort, patients were followed out to 90 days after transplant.
Results
The total number of platelet units transfused dropped from 1,660 pre-PBM program to 1,417 post-PBM implementation. The total number of RBC units dropped from 1,158 to 826.
The researchers also saw changes in the proportions of inappropriate (outside guidelines) transfusions between the two time periods.
In 2013, 94.2% of RBC transfusions occurred outside the guidelines, compared with 35.4% in 2015 (P<0.0001). Similarly, the proportion of inappropriate platelet transfusions declined from 73.4% to 48.7% over the same time period (P<0.0001).
In addition, all-cause mortality at 3 months was significantly lower after the PBM program was introduced. The 3-month mortality rate was 30.7% for the 2013 cohort and 20.2% for the 2015 cohort (P=0.001).
Dr. Jambhekar noted that, in a multivariable analysis accounting for baseline differences between the groups, mortality for patients treated before the PBM program remained significantly higher, with an odds ratio of 1.85 (P=0.0008).
Neither hospital nor ICU admission within 30 days differed significantly between the groups, and there were no significant between-group differences in hospital or ICU lengths of stay.
Likewise, there were no significant between-group differences in myocardial infarctions, cerebrovascular events, sepsis, and febrile or allergic transfusion reactions.
Dr. Jambhekar noted that this study was retrospective in design and therefore could not fully account for potential confounders. It’s also unclear whether the results could be generalized for adoption by other institutions.
“In general, PBM implementation is probably helpful in reducing both platelet and PRBC [packed red blood cell] utilization, but it’s not an easy thing to do,” Dr. Jambhekar said.
“It requires institutional buy-in and key players to make it happen. Ongoing PBM-related activities like surveillance, education, and clinical decision feedback are critical to maintaining success that we’ve had.”
This study was internally funded. Dr. Jambhekar reported having nothing to disclose.
*Data presented differ from the abstract.
AP-1 plays key role in various AML subtypes, team says
The AP-1 transcription factor family is of “major importance” in acute myeloid leukemia (AML), according to researchers.
The team said they identified transcription factor networks specific to AML subtypes, which showed that leukemic growth is dependent upon certain transcription factors, and “the global activation of signaling pathways parallels a growth dependence on AP-1 activity in multiple types of AML.”
Constanze Bonifer, PhD, of the University of Birmingham in the U.K., and her colleagues conducted this research and detailed their findings in Nature Genetics.
The researchers noted that previous work revealed the existence of gene regulatory networks in different types of AML classified by gene expression and DNA methylation patterns.
“Our work now defines these networks in detail and shows that leukemic drivers determine the regulatory phenotype by establishing and maintaining specific gene regulatory and signaling networks that are distinct from those in normal cells,” Dr. Bonifer and her colleagues wrote.
The researchers combined data obtained via several analytic techniques to construct transcription factor networks in normal CD34+ cells and cells from AML patients with defined mutations, including RUNX1 mutations, t(8;21) translocations, mutations of both alleles of the CEBPA gene, and FLT3-ITD with or without NPM1 mutation.
The AP-1 family network was of “high regulatory relevance” for all AML subtypes evaluated, the team reported.
Follow-up in vitro and in vivo studies confirmed the importance of AP-1 for different AML subtypes.
In the in vitro study, the researchers transduced AML cells with a doxycycline-inducible version of a dominant-negative (dn) FOS protein.
“AP-1 is a heterodimer formed by members of the FOS, JUN, ATF, CREB, and JDP families of transcription factors,” the researchers wrote. “[T]hus, it is challenging to target by defined RNA interference approaches.”
Results of the in vitro study showed that induction of dnFOS, mediated by doxycycline, inhibited proliferation of t(8;21)+ Kasumi-1 cells and FLT3-ITD-expressing MV4-11 cells.
Induction of dnFOS also inhibited the colony-forming ability of primary CD34+ FLT3-ITD cells but not CD34+ hematopoietic stem and progenitor cells.
To evaluate the relevance of AP-1 for leukemia propagation in vivo, the researchers transplanted either of two cell lines—Kasumi-1 or MV4-11—expressing inducible dnFOS in immunodeficient mice.
With Kasumi-1, granulosarcomas developed in six of seven untreated control mice and two mice treated with doxycycline, neither of which expressed the inducible protein.
With MV4-11, doxycycline inhibited leukemia development, and untreated mice rapidly developed tumors.
The researchers declared no competing interests related to this work, which was funded by Bloodwise, Cancer Research UK, a Kay Kendall Clinical Training Fellowship, and an MRC/Leuka Clinical Training Fellowship.
The AP-1 transcription factor family is of “major importance” in acute myeloid leukemia (AML), according to researchers.
The team said they identified transcription factor networks specific to AML subtypes, which showed that leukemic growth is dependent upon certain transcription factors, and “the global activation of signaling pathways parallels a growth dependence on AP-1 activity in multiple types of AML.”
Constanze Bonifer, PhD, of the University of Birmingham in the U.K., and her colleagues conducted this research and detailed their findings in Nature Genetics.
The researchers noted that previous work revealed the existence of gene regulatory networks in different types of AML classified by gene expression and DNA methylation patterns.
“Our work now defines these networks in detail and shows that leukemic drivers determine the regulatory phenotype by establishing and maintaining specific gene regulatory and signaling networks that are distinct from those in normal cells,” Dr. Bonifer and her colleagues wrote.
The researchers combined data obtained via several analytic techniques to construct transcription factor networks in normal CD34+ cells and cells from AML patients with defined mutations, including RUNX1 mutations, t(8;21) translocations, mutations of both alleles of the CEBPA gene, and FLT3-ITD with or without NPM1 mutation.
The AP-1 family network was of “high regulatory relevance” for all AML subtypes evaluated, the team reported.
Follow-up in vitro and in vivo studies confirmed the importance of AP-1 for different AML subtypes.
In the in vitro study, the researchers transduced AML cells with a doxycycline-inducible version of a dominant-negative (dn) FOS protein.
“AP-1 is a heterodimer formed by members of the FOS, JUN, ATF, CREB, and JDP families of transcription factors,” the researchers wrote. “[T]hus, it is challenging to target by defined RNA interference approaches.”
Results of the in vitro study showed that induction of dnFOS, mediated by doxycycline, inhibited proliferation of t(8;21)+ Kasumi-1 cells and FLT3-ITD-expressing MV4-11 cells.
Induction of dnFOS also inhibited the colony-forming ability of primary CD34+ FLT3-ITD cells but not CD34+ hematopoietic stem and progenitor cells.
To evaluate the relevance of AP-1 for leukemia propagation in vivo, the researchers transplanted either of two cell lines—Kasumi-1 or MV4-11—expressing inducible dnFOS in immunodeficient mice.
With Kasumi-1, granulosarcomas developed in six of seven untreated control mice and two mice treated with doxycycline, neither of which expressed the inducible protein.
With MV4-11, doxycycline inhibited leukemia development, and untreated mice rapidly developed tumors.
The researchers declared no competing interests related to this work, which was funded by Bloodwise, Cancer Research UK, a Kay Kendall Clinical Training Fellowship, and an MRC/Leuka Clinical Training Fellowship.
The AP-1 transcription factor family is of “major importance” in acute myeloid leukemia (AML), according to researchers.
The team said they identified transcription factor networks specific to AML subtypes, which showed that leukemic growth is dependent upon certain transcription factors, and “the global activation of signaling pathways parallels a growth dependence on AP-1 activity in multiple types of AML.”
Constanze Bonifer, PhD, of the University of Birmingham in the U.K., and her colleagues conducted this research and detailed their findings in Nature Genetics.
The researchers noted that previous work revealed the existence of gene regulatory networks in different types of AML classified by gene expression and DNA methylation patterns.
“Our work now defines these networks in detail and shows that leukemic drivers determine the regulatory phenotype by establishing and maintaining specific gene regulatory and signaling networks that are distinct from those in normal cells,” Dr. Bonifer and her colleagues wrote.
The researchers combined data obtained via several analytic techniques to construct transcription factor networks in normal CD34+ cells and cells from AML patients with defined mutations, including RUNX1 mutations, t(8;21) translocations, mutations of both alleles of the CEBPA gene, and FLT3-ITD with or without NPM1 mutation.
The AP-1 family network was of “high regulatory relevance” for all AML subtypes evaluated, the team reported.
Follow-up in vitro and in vivo studies confirmed the importance of AP-1 for different AML subtypes.
In the in vitro study, the researchers transduced AML cells with a doxycycline-inducible version of a dominant-negative (dn) FOS protein.
“AP-1 is a heterodimer formed by members of the FOS, JUN, ATF, CREB, and JDP families of transcription factors,” the researchers wrote. “[T]hus, it is challenging to target by defined RNA interference approaches.”
Results of the in vitro study showed that induction of dnFOS, mediated by doxycycline, inhibited proliferation of t(8;21)+ Kasumi-1 cells and FLT3-ITD-expressing MV4-11 cells.
Induction of dnFOS also inhibited the colony-forming ability of primary CD34+ FLT3-ITD cells but not CD34+ hematopoietic stem and progenitor cells.
To evaluate the relevance of AP-1 for leukemia propagation in vivo, the researchers transplanted either of two cell lines—Kasumi-1 or MV4-11—expressing inducible dnFOS in immunodeficient mice.
With Kasumi-1, granulosarcomas developed in six of seven untreated control mice and two mice treated with doxycycline, neither of which expressed the inducible protein.
With MV4-11, doxycycline inhibited leukemia development, and untreated mice rapidly developed tumors.
The researchers declared no competing interests related to this work, which was funded by Bloodwise, Cancer Research UK, a Kay Kendall Clinical Training Fellowship, and an MRC/Leuka Clinical Training Fellowship.