User login
Best Practices When Using POEM to Treat Achalasia: AGA Clinical Update
The American Gastroenterological Association (AGA) has released
“Any patient suspected to have achalasia, or difficulty swallowing for that matter, should undergo a comprehensive diagnostic workup, and that should include clinical history, review of medication, as well as tests. The diagnosis should not be based on isolated tests but on the clinical picture as a whole,” first author Dennis Yang, MD, AGAF, with the Center for Interventional Endoscopy, AdventHealth, Orlando, Florida, noted in an AGA podcast about the update.
The clinical practice update, published in Gastroenterology, includes 12 “best practice advice” statements.
Since its introduction to clinical practice more than a decade ago, POEM has matured and gained widespread acceptance because of its efficacy and safety profile.
POEM has at least similar outcomes to laparoscopic Heller myotomy and pneumatic dilation for type I and type II achalasia with better results for those with type III achalasia, Yang noted.
“However, besides disease phenotype, we need to remember that choosing the right treatment for the patient is going to be based on multiple factors including patient characteristics as well as local expertise,” Yang added.
In terms of technical considerations, the update states that both anterior and posterior tunnel approaches demonstrate comparable success and postprocedure reflux rates. Tunnel orientation should be tailored to the patient’s surgical history and endoscopist’s preference.
It further states that optimal length of the myotomy in the esophagus and cardia, as it pertains to treatment efficacy and risk for postprocedure reflux, remains to be determined.
Adjunct techniques, including real-time intraprocedure functional luminal impedance planimetry, may be considered to tailor or confirm the adequacy of the myotomy.
Same-day discharge after POEM can be considered in select patients who meet discharge criteria. Patients with advanced age, significant comorbidities, poor social support, and/or access to specialized care should be considered for hospital admission, irrespective of symptoms.
The update notes that specific guidelines on the role and extent of antibiotic prophylaxis before and after POEM are lacking. A single dose of antibiotics at the time of POEM “may be sufficient” for antibiotic prophylaxis.
In terms of immediate post-POEM care, the update notes that the clinical impact of routine esophagram or endoscopy immediately post-POEM remains unclear. Testing can be considered based on local practice preferences and in cases in which intraprocedural events or postprocedural findings warrant further evaluation.
Proton pump inhibitors are recommended immediately following POEM, as gastroesophageal reflux disease (GERD) is common following POEM, occurring in up to 65% of cases.
Routine endoscopic surveillance is advised to monitor GERD, disease progression, and esophageal cancer risk, which is significantly higher in achalasia patients.
“Just like diabetes and hypertension, we need to remember that achalasia is a chronic disease and long-term postprocedural surveillance is strongly encouraged to monitor disease progression as well as potential complications of reflux,” Yang said.
He noted that surveillance should be considered irrespective of patient symptoms because many of these patients may remain asymptomatic.
“Primary gastroenterologists should have a very low threshold in referring the patient back to the POEM endoscopist or any specialized esophageal center because the ideology of symptoms in these patients can be quite difficult to tease out and often require comprehensive diagnostic workup,” Yang said.
Evidence for POEM in esophagogastric outflow obstruction and other nonachalasia spastic motility disorders is limited and should only be considered on a case-by-case basis after other less invasive approaches have been exhausted, the update states.
For perspective on the POEM clinical practice update, this news organization spoke with Mouen Khashab, MD, director of therapeutic endoscopy, Johns Hopkins University, Baltimore, Maryland.
“The document is very well written and comprehensive,” Khashab said.
However, Khashab said he would have liked to see greater emphasis on the value or role of a short myotomy in the esophagus and cardia.
“There is level I evidence that the short esophageal myotomy is equivalent to a long esophageal myotomy for type I and II achalasia. When you do a short myotomy, you save procedure time and there is potentially a lower incidence of blown-out myotomy or BOM,” Khashab said.
Khashab also noted that a long myotomy on the gastric side “likely increases the risk of reflux disease, and therefore a limited myotomy on the gastric side likely also is advantageous.”
This research had no commercial funding. Yang serves as a consultant for Boston Scientific, Olympus, FujiFilm, Microtech, Medtronic, 3D-Matrix, and Neptune Medical, and has received research support from Microtech and 3D-Matrix. Khashab had no relevant disclosures.
A version of this article appeared on Medscape.com .
The American Gastroenterological Association (AGA) has released
“Any patient suspected to have achalasia, or difficulty swallowing for that matter, should undergo a comprehensive diagnostic workup, and that should include clinical history, review of medication, as well as tests. The diagnosis should not be based on isolated tests but on the clinical picture as a whole,” first author Dennis Yang, MD, AGAF, with the Center for Interventional Endoscopy, AdventHealth, Orlando, Florida, noted in an AGA podcast about the update.
The clinical practice update, published in Gastroenterology, includes 12 “best practice advice” statements.
Since its introduction to clinical practice more than a decade ago, POEM has matured and gained widespread acceptance because of its efficacy and safety profile.
POEM has at least similar outcomes to laparoscopic Heller myotomy and pneumatic dilation for type I and type II achalasia with better results for those with type III achalasia, Yang noted.
“However, besides disease phenotype, we need to remember that choosing the right treatment for the patient is going to be based on multiple factors including patient characteristics as well as local expertise,” Yang added.
In terms of technical considerations, the update states that both anterior and posterior tunnel approaches demonstrate comparable success and postprocedure reflux rates. Tunnel orientation should be tailored to the patient’s surgical history and endoscopist’s preference.
It further states that optimal length of the myotomy in the esophagus and cardia, as it pertains to treatment efficacy and risk for postprocedure reflux, remains to be determined.
Adjunct techniques, including real-time intraprocedure functional luminal impedance planimetry, may be considered to tailor or confirm the adequacy of the myotomy.
Same-day discharge after POEM can be considered in select patients who meet discharge criteria. Patients with advanced age, significant comorbidities, poor social support, and/or access to specialized care should be considered for hospital admission, irrespective of symptoms.
The update notes that specific guidelines on the role and extent of antibiotic prophylaxis before and after POEM are lacking. A single dose of antibiotics at the time of POEM “may be sufficient” for antibiotic prophylaxis.
In terms of immediate post-POEM care, the update notes that the clinical impact of routine esophagram or endoscopy immediately post-POEM remains unclear. Testing can be considered based on local practice preferences and in cases in which intraprocedural events or postprocedural findings warrant further evaluation.
Proton pump inhibitors are recommended immediately following POEM, as gastroesophageal reflux disease (GERD) is common following POEM, occurring in up to 65% of cases.
Routine endoscopic surveillance is advised to monitor GERD, disease progression, and esophageal cancer risk, which is significantly higher in achalasia patients.
“Just like diabetes and hypertension, we need to remember that achalasia is a chronic disease and long-term postprocedural surveillance is strongly encouraged to monitor disease progression as well as potential complications of reflux,” Yang said.
He noted that surveillance should be considered irrespective of patient symptoms because many of these patients may remain asymptomatic.
“Primary gastroenterologists should have a very low threshold in referring the patient back to the POEM endoscopist or any specialized esophageal center because the ideology of symptoms in these patients can be quite difficult to tease out and often require comprehensive diagnostic workup,” Yang said.
Evidence for POEM in esophagogastric outflow obstruction and other nonachalasia spastic motility disorders is limited and should only be considered on a case-by-case basis after other less invasive approaches have been exhausted, the update states.
For perspective on the POEM clinical practice update, this news organization spoke with Mouen Khashab, MD, director of therapeutic endoscopy, Johns Hopkins University, Baltimore, Maryland.
“The document is very well written and comprehensive,” Khashab said.
However, Khashab said he would have liked to see greater emphasis on the value or role of a short myotomy in the esophagus and cardia.
“There is level I evidence that the short esophageal myotomy is equivalent to a long esophageal myotomy for type I and II achalasia. When you do a short myotomy, you save procedure time and there is potentially a lower incidence of blown-out myotomy or BOM,” Khashab said.
Khashab also noted that a long myotomy on the gastric side “likely increases the risk of reflux disease, and therefore a limited myotomy on the gastric side likely also is advantageous.”
This research had no commercial funding. Yang serves as a consultant for Boston Scientific, Olympus, FujiFilm, Microtech, Medtronic, 3D-Matrix, and Neptune Medical, and has received research support from Microtech and 3D-Matrix. Khashab had no relevant disclosures.
A version of this article appeared on Medscape.com .
The American Gastroenterological Association (AGA) has released
“Any patient suspected to have achalasia, or difficulty swallowing for that matter, should undergo a comprehensive diagnostic workup, and that should include clinical history, review of medication, as well as tests. The diagnosis should not be based on isolated tests but on the clinical picture as a whole,” first author Dennis Yang, MD, AGAF, with the Center for Interventional Endoscopy, AdventHealth, Orlando, Florida, noted in an AGA podcast about the update.
The clinical practice update, published in Gastroenterology, includes 12 “best practice advice” statements.
Since its introduction to clinical practice more than a decade ago, POEM has matured and gained widespread acceptance because of its efficacy and safety profile.
POEM has at least similar outcomes to laparoscopic Heller myotomy and pneumatic dilation for type I and type II achalasia with better results for those with type III achalasia, Yang noted.
“However, besides disease phenotype, we need to remember that choosing the right treatment for the patient is going to be based on multiple factors including patient characteristics as well as local expertise,” Yang added.
In terms of technical considerations, the update states that both anterior and posterior tunnel approaches demonstrate comparable success and postprocedure reflux rates. Tunnel orientation should be tailored to the patient’s surgical history and endoscopist’s preference.
It further states that optimal length of the myotomy in the esophagus and cardia, as it pertains to treatment efficacy and risk for postprocedure reflux, remains to be determined.
Adjunct techniques, including real-time intraprocedure functional luminal impedance planimetry, may be considered to tailor or confirm the adequacy of the myotomy.
Same-day discharge after POEM can be considered in select patients who meet discharge criteria. Patients with advanced age, significant comorbidities, poor social support, and/or access to specialized care should be considered for hospital admission, irrespective of symptoms.
The update notes that specific guidelines on the role and extent of antibiotic prophylaxis before and after POEM are lacking. A single dose of antibiotics at the time of POEM “may be sufficient” for antibiotic prophylaxis.
In terms of immediate post-POEM care, the update notes that the clinical impact of routine esophagram or endoscopy immediately post-POEM remains unclear. Testing can be considered based on local practice preferences and in cases in which intraprocedural events or postprocedural findings warrant further evaluation.
Proton pump inhibitors are recommended immediately following POEM, as gastroesophageal reflux disease (GERD) is common following POEM, occurring in up to 65% of cases.
Routine endoscopic surveillance is advised to monitor GERD, disease progression, and esophageal cancer risk, which is significantly higher in achalasia patients.
“Just like diabetes and hypertension, we need to remember that achalasia is a chronic disease and long-term postprocedural surveillance is strongly encouraged to monitor disease progression as well as potential complications of reflux,” Yang said.
He noted that surveillance should be considered irrespective of patient symptoms because many of these patients may remain asymptomatic.
“Primary gastroenterologists should have a very low threshold in referring the patient back to the POEM endoscopist or any specialized esophageal center because the ideology of symptoms in these patients can be quite difficult to tease out and often require comprehensive diagnostic workup,” Yang said.
Evidence for POEM in esophagogastric outflow obstruction and other nonachalasia spastic motility disorders is limited and should only be considered on a case-by-case basis after other less invasive approaches have been exhausted, the update states.
For perspective on the POEM clinical practice update, this news organization spoke with Mouen Khashab, MD, director of therapeutic endoscopy, Johns Hopkins University, Baltimore, Maryland.
“The document is very well written and comprehensive,” Khashab said.
However, Khashab said he would have liked to see greater emphasis on the value or role of a short myotomy in the esophagus and cardia.
“There is level I evidence that the short esophageal myotomy is equivalent to a long esophageal myotomy for type I and II achalasia. When you do a short myotomy, you save procedure time and there is potentially a lower incidence of blown-out myotomy or BOM,” Khashab said.
Khashab also noted that a long myotomy on the gastric side “likely increases the risk of reflux disease, and therefore a limited myotomy on the gastric side likely also is advantageous.”
This research had no commercial funding. Yang serves as a consultant for Boston Scientific, Olympus, FujiFilm, Microtech, Medtronic, 3D-Matrix, and Neptune Medical, and has received research support from Microtech and 3D-Matrix. Khashab had no relevant disclosures.
A version of this article appeared on Medscape.com .
FROM GASTROENTEROLOGY
Journal Highlights: October-December 2024
Esophagus
Reed CC et al. Daily or Twice Daily Treatment with Topical Steroids Results in Similar Responses in Eosinophilic Esophagitis. Clin Gastroenterol Hepatol. 2024 Nov. doi: 10.1016/j.cgh.2024.10.016.
Patel RV et al. Functional Lumen Imaging Probe Provides an Accurate Assessment of Esophageal Diameter in Patients With Eosinophilic Esophagitis. Clin Gastroenterol Hepatol. 2024 Dec. doi: 10.1016/j.cgh.2024.10.032.
Stomach
Shah SC et al. AGA Clinical Practice Update on Screening and Surveillance in Individuals at Increased Risk for Gastric Cancer in the United States: Expert Review. Gastroenterology. 2024 Dec. doi: 10.1053/j.gastro.2024.11.001.
IBD
Griffiths BJ et al. Hypercoagulation after Hospital Discharge in Acute Severe Ulcerative Colitis: A Prospective Study. Clin Gastroenterol Hepatol. 2024 Dec. doi: 10.1016/j.cgh.2024.10.031.
Liver
Lassailly G et al. Resolution of MASH with no worsening of fibrosis after bariatric surgery improves 15-year survival: a prospective cohort study. Clin Gastroenterol Hepatol. 2024 Dec. doi: 10.1016/j.cgh.2024.10.025.
Norman JS et al. Model for Urgency for Liver Transplantation in Hepatocellular Carcinoma: A Practical Model to Prioritize Patients With Hepatocellular Carcinoma on the Liver Transplant Waiting List. Gastroenterology. 2024 Nov. doi: 10.1053/j.gastro.2024.11.015.
Davis JPE et al. AGA Clinical Practice Update on Management of Portal Vein Thrombosis in Patients With Cirrhosis: Expert Review. Gastroenterology. 2024 Dec. doi: 10.1053/j.gastro.2024.10.038.
Pancreas
Drewes AM et al. Pain in Chronic Pancreatitis: Navigating the Maze of Blocked Tubes and Tangled Wires. Gastroenterology. 2024 Dec. doi: 10.1053/j.gastro.2024.11.026.
Endoscopy
Kindel TL et al; American Gastroenterological Association; American Society for Metabolic and Bariatric Surgery; American Society of Anesthesiologists; International Society of Perioperative Care of Patients with Obesity; Society of American Gastrointestinal and Endoscopic Surgeons. Multisociety Clinical Practice Guidance for the Safe Use of Glucagon-like Peptide-1 Receptor Agonists in the Perioperative Period. Clin Gastroenterol Hepatol. 2024 Oct. doi: 10.1016/j.cgh.2024.10.003.
Schmidt KA et al. Understanding Patients’ Current Acceptability of Artificial Intelligence During Colonoscopy for Polyp Detection: A Single-Center Study. Techniques and Innovations in Gastrointestinal Endoscopy. 2024 Dec. doi: 10.1016/j.tige.2024.250905.
Chandramouli S et al. Endoscopic Surveillance Patterns and Management of Helicobacter pylori in Newly Diagnosed Gastric Intestinal Metaplasia. Techniques and Innovations in Gastrointestinal Endoscopy. 2024 Dec. doi: 10.1016/j.tige.2024.250904.
Practice Management
Tsai C et al. Trauma-Informed Care in Gastroenterology: A Survey of Provider Attitudes, Knowledge, and Skills. Clin Gastroenterol Hepatol. 2024 Oct. doi: 10.1016/j.cgh.2024.09.015.
Mintz KM et al. Incorporating a GI Dietitian into Your GI Practice. Gastroenterology. 2024 Nov. doi: 10.1053/j.gastro.2024.10.022.
Dr. Trieu is assistant professor of medicine, interventional endoscopy, in the Division of Gastroenterology at Washington University in St. Louis School of Medicine, Missouri.
Esophagus
Reed CC et al. Daily or Twice Daily Treatment with Topical Steroids Results in Similar Responses in Eosinophilic Esophagitis. Clin Gastroenterol Hepatol. 2024 Nov. doi: 10.1016/j.cgh.2024.10.016.
Patel RV et al. Functional Lumen Imaging Probe Provides an Accurate Assessment of Esophageal Diameter in Patients With Eosinophilic Esophagitis. Clin Gastroenterol Hepatol. 2024 Dec. doi: 10.1016/j.cgh.2024.10.032.
Stomach
Shah SC et al. AGA Clinical Practice Update on Screening and Surveillance in Individuals at Increased Risk for Gastric Cancer in the United States: Expert Review. Gastroenterology. 2024 Dec. doi: 10.1053/j.gastro.2024.11.001.
IBD
Griffiths BJ et al. Hypercoagulation after Hospital Discharge in Acute Severe Ulcerative Colitis: A Prospective Study. Clin Gastroenterol Hepatol. 2024 Dec. doi: 10.1016/j.cgh.2024.10.031.
Liver
Lassailly G et al. Resolution of MASH with no worsening of fibrosis after bariatric surgery improves 15-year survival: a prospective cohort study. Clin Gastroenterol Hepatol. 2024 Dec. doi: 10.1016/j.cgh.2024.10.025.
Norman JS et al. Model for Urgency for Liver Transplantation in Hepatocellular Carcinoma: A Practical Model to Prioritize Patients With Hepatocellular Carcinoma on the Liver Transplant Waiting List. Gastroenterology. 2024 Nov. doi: 10.1053/j.gastro.2024.11.015.
Davis JPE et al. AGA Clinical Practice Update on Management of Portal Vein Thrombosis in Patients With Cirrhosis: Expert Review. Gastroenterology. 2024 Dec. doi: 10.1053/j.gastro.2024.10.038.
Pancreas
Drewes AM et al. Pain in Chronic Pancreatitis: Navigating the Maze of Blocked Tubes and Tangled Wires. Gastroenterology. 2024 Dec. doi: 10.1053/j.gastro.2024.11.026.
Endoscopy
Kindel TL et al; American Gastroenterological Association; American Society for Metabolic and Bariatric Surgery; American Society of Anesthesiologists; International Society of Perioperative Care of Patients with Obesity; Society of American Gastrointestinal and Endoscopic Surgeons. Multisociety Clinical Practice Guidance for the Safe Use of Glucagon-like Peptide-1 Receptor Agonists in the Perioperative Period. Clin Gastroenterol Hepatol. 2024 Oct. doi: 10.1016/j.cgh.2024.10.003.
Schmidt KA et al. Understanding Patients’ Current Acceptability of Artificial Intelligence During Colonoscopy for Polyp Detection: A Single-Center Study. Techniques and Innovations in Gastrointestinal Endoscopy. 2024 Dec. doi: 10.1016/j.tige.2024.250905.
Chandramouli S et al. Endoscopic Surveillance Patterns and Management of Helicobacter pylori in Newly Diagnosed Gastric Intestinal Metaplasia. Techniques and Innovations in Gastrointestinal Endoscopy. 2024 Dec. doi: 10.1016/j.tige.2024.250904.
Practice Management
Tsai C et al. Trauma-Informed Care in Gastroenterology: A Survey of Provider Attitudes, Knowledge, and Skills. Clin Gastroenterol Hepatol. 2024 Oct. doi: 10.1016/j.cgh.2024.09.015.
Mintz KM et al. Incorporating a GI Dietitian into Your GI Practice. Gastroenterology. 2024 Nov. doi: 10.1053/j.gastro.2024.10.022.
Dr. Trieu is assistant professor of medicine, interventional endoscopy, in the Division of Gastroenterology at Washington University in St. Louis School of Medicine, Missouri.
Esophagus
Reed CC et al. Daily or Twice Daily Treatment with Topical Steroids Results in Similar Responses in Eosinophilic Esophagitis. Clin Gastroenterol Hepatol. 2024 Nov. doi: 10.1016/j.cgh.2024.10.016.
Patel RV et al. Functional Lumen Imaging Probe Provides an Accurate Assessment of Esophageal Diameter in Patients With Eosinophilic Esophagitis. Clin Gastroenterol Hepatol. 2024 Dec. doi: 10.1016/j.cgh.2024.10.032.
Stomach
Shah SC et al. AGA Clinical Practice Update on Screening and Surveillance in Individuals at Increased Risk for Gastric Cancer in the United States: Expert Review. Gastroenterology. 2024 Dec. doi: 10.1053/j.gastro.2024.11.001.
IBD
Griffiths BJ et al. Hypercoagulation after Hospital Discharge in Acute Severe Ulcerative Colitis: A Prospective Study. Clin Gastroenterol Hepatol. 2024 Dec. doi: 10.1016/j.cgh.2024.10.031.
Liver
Lassailly G et al. Resolution of MASH with no worsening of fibrosis after bariatric surgery improves 15-year survival: a prospective cohort study. Clin Gastroenterol Hepatol. 2024 Dec. doi: 10.1016/j.cgh.2024.10.025.
Norman JS et al. Model for Urgency for Liver Transplantation in Hepatocellular Carcinoma: A Practical Model to Prioritize Patients With Hepatocellular Carcinoma on the Liver Transplant Waiting List. Gastroenterology. 2024 Nov. doi: 10.1053/j.gastro.2024.11.015.
Davis JPE et al. AGA Clinical Practice Update on Management of Portal Vein Thrombosis in Patients With Cirrhosis: Expert Review. Gastroenterology. 2024 Dec. doi: 10.1053/j.gastro.2024.10.038.
Pancreas
Drewes AM et al. Pain in Chronic Pancreatitis: Navigating the Maze of Blocked Tubes and Tangled Wires. Gastroenterology. 2024 Dec. doi: 10.1053/j.gastro.2024.11.026.
Endoscopy
Kindel TL et al; American Gastroenterological Association; American Society for Metabolic and Bariatric Surgery; American Society of Anesthesiologists; International Society of Perioperative Care of Patients with Obesity; Society of American Gastrointestinal and Endoscopic Surgeons. Multisociety Clinical Practice Guidance for the Safe Use of Glucagon-like Peptide-1 Receptor Agonists in the Perioperative Period. Clin Gastroenterol Hepatol. 2024 Oct. doi: 10.1016/j.cgh.2024.10.003.
Schmidt KA et al. Understanding Patients’ Current Acceptability of Artificial Intelligence During Colonoscopy for Polyp Detection: A Single-Center Study. Techniques and Innovations in Gastrointestinal Endoscopy. 2024 Dec. doi: 10.1016/j.tige.2024.250905.
Chandramouli S et al. Endoscopic Surveillance Patterns and Management of Helicobacter pylori in Newly Diagnosed Gastric Intestinal Metaplasia. Techniques and Innovations in Gastrointestinal Endoscopy. 2024 Dec. doi: 10.1016/j.tige.2024.250904.
Practice Management
Tsai C et al. Trauma-Informed Care in Gastroenterology: A Survey of Provider Attitudes, Knowledge, and Skills. Clin Gastroenterol Hepatol. 2024 Oct. doi: 10.1016/j.cgh.2024.09.015.
Mintz KM et al. Incorporating a GI Dietitian into Your GI Practice. Gastroenterology. 2024 Nov. doi: 10.1053/j.gastro.2024.10.022.
Dr. Trieu is assistant professor of medicine, interventional endoscopy, in the Division of Gastroenterology at Washington University in St. Louis School of Medicine, Missouri.
Improving Care for Sexual and Gender Minority Patients with Disorders of Gut-Brain Interaction
Brief Introduction to the SGM Communities
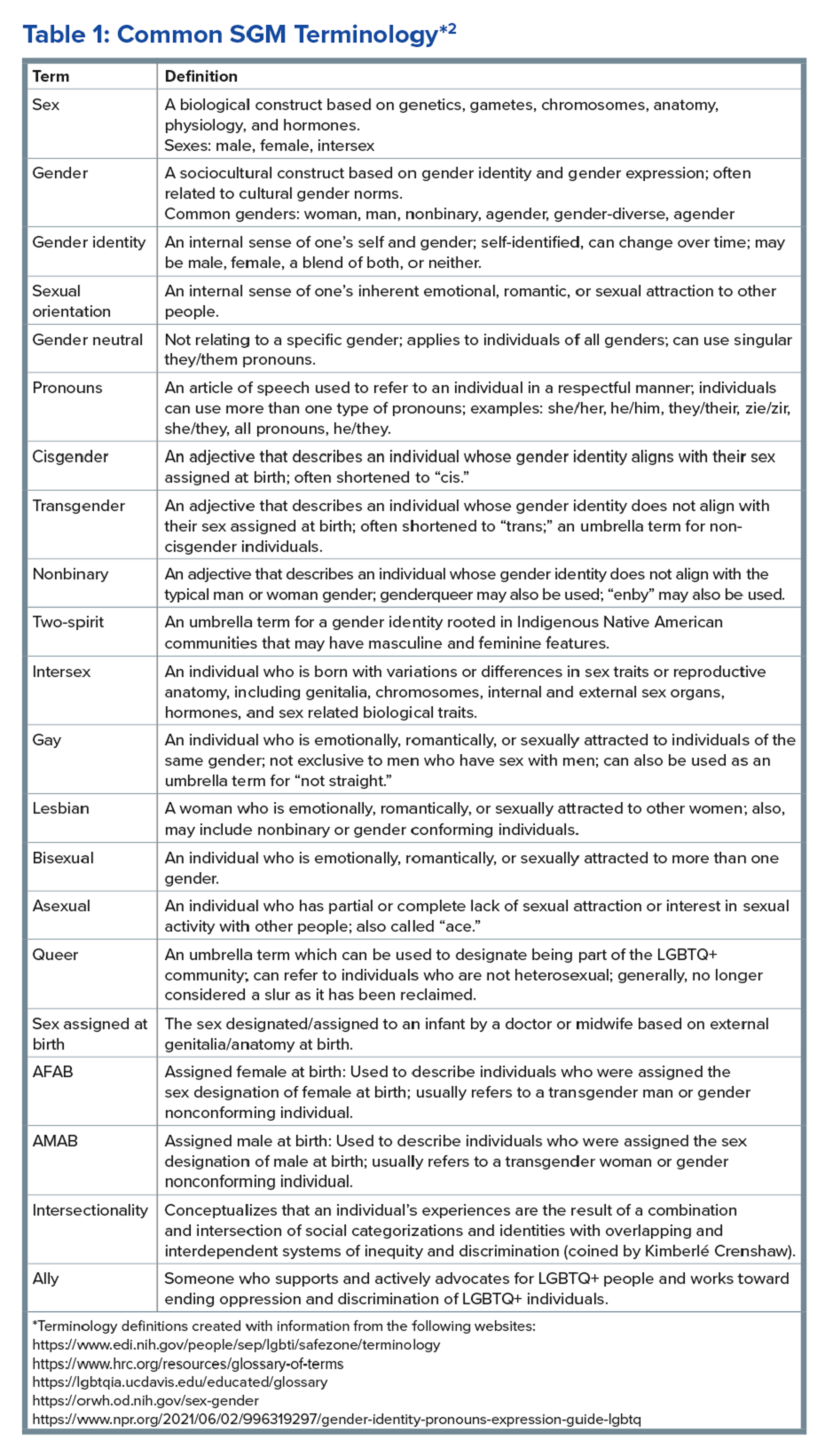
The sexual and gender minority (SGM) communities (see Table 1), also termed “LGBTQIA+ community” (lesbian, gay, bisexual, transgender, queer, intersex, asexual, plus — including two spirit) are historically minoritized with unique risks for inequities in gastrointestinal health outcomes.1 These potential disparities remain largely uninvestigated because of continued systemic discrimination and inadequate collection of sexual orientation and gender identity (SOGI) data,2 with the National Institutes of Health Sexual & Gender Minority Research Office (SGMRO) having been instructed to address these failures. There is increased SGM self-identification (7.1% of all people in the United States and 20.8% of generation Z).3 Given the high worldwide prevalence of disorders of gut-brain interaction (DGBIs)and the influence of biopsychosocial determinants of health in DGBI incidence,4 it becomes increasingly likely that research in DGBI-related factors in SGM people will be fruitful.
Disorders of Gut-Brain Interaction and the Potential Minority Stress Link in SGM People
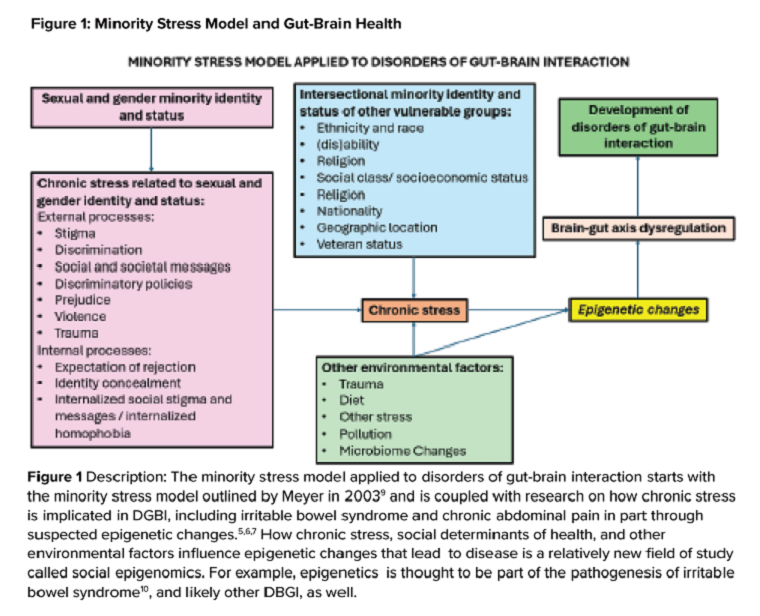
DGBIs are gastrointestinal conditions that occur because of brain-gut axis dysregulation. There is evidence that chronic stress and trauma negatively influence brain-gut interaction, which likely results in minority communities who face increased levels of trauma, stress, discrimination, and social injustice being at higher risk of DGBI development.5-7 Given increased rates of trauma in the SGM community, practicing trauma-informed care is essential to increase patient comfort and decrease the chance of retraumatization in medical settings.8 Trauma-informed care focuses on how trauma influences a patient’s life and response to medical care. To practice trauma-informed care, screening for trauma when appropriate, actively creating a supportive environment with active listening and communication, with informing the patient of planned actions prior to doing them, like physical exams, is key.
Trauma-Informed Care: Examples of Verbiage
Asking about Identity
- Begin by introducing yourself with your pronouns to create a safe environment for patient disclosure. Example: “Hello, I am Dr. Kara Jencks, and my pronouns are she/her. I am one of the gastroenterologists here at XYZ Clinic. How would you prefer to be addressed?”
- You can also wear a pronoun lapel pin or a pronoun button on your ID badge to indicate you are someone who your patient can be themselves around.
- The easiest way to obtain sexual orientation and gender identity is through intake forms. Below are examples of how to ask these questions on intake forms. It is important to offer the option to select more than one option when applicable and to opt out of answering if the patient is not comfortable answering these questions.
Sample Questions for Intake Forms
1. What is your sex assigned at birth? (Select one)
- Female
- Male
- Intersex
- Do not know
- Prefer not to disclose
2. What is your gender identity? (Select all that apply)
- Nonbinary
- Gender queer
- Woman
- Man
- Transwoman
- Transman
- Gender fluid
- Two-spirit
- Agender
- Intersex
- Other: type in response
- Prefer not to disclose
3. What are your pronouns? (Select all that apply)
- They/them/theirs
- She/her/hers
- He/him/his
- Zie/zir/zirs
- Other: type in response
- Prefer not to disclose
4. What is your sexual orientation? (Select all that apply)
- Bisexual
- Pansexual
- Queer
- Lesbian
- Gay
- Asexual
- Demisexual
- Heterosexual or straight
- Other: type in response
- Prefer not to disclose
Screening for Trauma
While there are questionnaires that exist to ask about trauma history, if time allows, it can be helpful to screen verbally with the patient. See reference number 8, for additional prompts and actions to practice trauma-informed care.
- Example: “Many patients with gastrointestinal symptoms and disorders have experienced trauma in the past. We do our best to ensure we are keeping you as comfortable as possible while caring for you. Are you comfortable sharing this information? [if yes->] Do you have a history of trauma, including physical, emotional, or sexual abuse? ... Have these experiences impacted the way in which you navigate your healthcare? ... Is there anything we can do to make you more comfortable today?”
General Physical Examination
Provide details for what you are going to do before you do it. Ask for permission for the examination. Here are two examples:
- “I would like to perform a physical exam to help better understand your symptoms. Is that okay with you?”
- “I would like to examine your abdomen with my stethoscope and my hands. Here is a sheet that we can use to help with your privacy. Please let me know if and when you feel any tenderness or pain.”
Rectal Physical Examination
Let the patient know why it would be helpful to perform a rectal exam, what the rectal exam will entail, and the benefits and risks to doing a rectal exam. An example follows:
- “Based on the symptoms you are describing, I think it would be helpful to perform a rectal exam to make sure you don’t have any fissures or hemorrhoids on the outside around the anus, any blockages or major issues inside the rectum, and to assess the strength and ability of your nerves and muscles or the pelvic floor to coordinate bowel movements. There are no risks aside from discomfort. If it is painful, and you would like me to stop, you tell me to stop, and I will stop right away. What questions do you have? Are we okay to proceed with the rectal exam?”
- “Please pull down your undergarments and your pants to either midthigh, your ankles, or all the way off, whatever your preference is, lie down on the left side on the exam table, and cover yourself with this sheet. In the meantime, I will be getting a chaperone to keep us safe and serve as a patient advocate during the procedure.”
- Upon returning to the exam room: “Here is Sara, who will be chaperoning today. Let myself or Sara know if you are uncomfortable or having pain during this exam. I will be lifting up the sheet to get a good look around the anus. [lifts up sheet] You will feel my hand helping to spread apart the buttocks. I am looking around the anus, and I do not see any fissures, hemorrhoids, or anything else concerning. Please squeeze in like you are trying to hold in gas. Please bear down like you are trying to have a bowel movement or let out gas. Okay, now you may feel some cold gel around the anus, and you will feel my finger go inside. Take a deep breath in. Do you feel any pain as I palpate? Please squeeze in like you are trying to hold in gas. Please bear down like you are trying to have a bowel movement or let out gas. I will be stopping the exam now.”
- You would then wash your hands and allow the patient to get dressed, and then disclose the exam findings and the rest of your visit.
Ilan H. Meyer coined the minority stress model when discussing mental health disorders in SGM patients in the early 2000s.9 With it being well known that DGBIs can overlap with (but are not necessarily caused by) mental health disorders, this model can easily apply to unify multiple individual and societal factors that can combine to result in disorders of brain-gut interaction (see Figure 1) in SGM communities. Let us keep this framework in mind when evaluating the following cases.
Case Presentations
Case 1
A 56-year-old man (pronouns: he/him) assigned male sex at birth, who identifies as gay, presents to your gastroenterology clinic for treatment-refractory constipation-predominant irritable bowel syndrome. It has impacted his sexual function. Outside hospital records report a normal colonoscopy 1 year ago and an unremarkable abdominal computerized tomography 4 months ago, aside from increased stool burden in the entire colon. He has tried to use enemas prior to sex, though these do not always help. Fiber-rich diet and fermentable food avoidance has not been successful. He is currently taking two capfuls of polyethylene glycol 3350 twice per day, as well as senna at night and continues to have a bowel movement every 2-3 days that is Bristol stool form scale type 1-2 unless he uses enemas. How do you counsel this patient about his IBS-C and rectal discomfort?
After assessing for sexual violence or other potential trauma-related factors, your digital rectal examination suggests that an anorectal defecatory disorder is less likely with normal relaxation and perineal movement. You recommend linaclotide. He notices improvement within 1 week, with improved comfort during anoreceptive sex.
Case 2
A 30-year-old woman (pronouns: she/her) assigned male sex at birth who has sex with men underwent vaginoplasty 2 years ago and is referred to the gastroenterology clinic for fecal incontinence and diarrhea. On review of her anatomic inventory, her vaginoplasty was a penile inversion vaginoplasty (no intestinal tissue was used for creation), and her prostate was left intact. The vaginal vault was created in between the urethra and rectum, similar to the pelvic floor anatomy of a woman assigned female sex at birth. Blood, imaging, and endoscopic workup has been negative. She is also not taking any medications associated with diarrhea, only taking estrogen and spironolactone. The diarrhea is not daily, but when present, about once per week, can be up to 10 episodes per day, and she has a sense of incomplete evacuation regularly. She notes having a rectal exam in the past but is not sure if her pelvic floor muscles have ever been assessed. How do you manage this patient?
To complete her evaluation in the office, you perform a trauma-informed rectal exam which reveals a decreased resting anal sphincter tone and paradoxical defecatory maneuvers without tenderness to the puborectalis muscle. Augmentation of the squeeze is also weak. Given her pelvic floor related surgical history, her symptoms, and her rectal exam, you recommend anorectal manometry which is abnormal and send her for anorectal biofeedback pelvic floor physical therapy, which improves her symptoms significantly.
Case 3
A 36-year-old woman (pronouns: she/her) assigned female sex at birth, who identifies as a lesbian, has a history of posttraumatic stress disorder and chronic nausea and vomiting that has begun to affect her quality of life. She notes the nausea and vomiting used to be managed well with evening cannabis gummies, though in the past 3 months, the nausea and vomiting has worsened, and she has lost 20 pounds as a result. As symptom predated cannabis usage, cannabis hyperemesis syndrome (CHS) was less likely (an important point as she has been stigmatized during prior encounters for her cannabis usage). Her primary care physician recommended a gastroscopy which was normal, aside from some residual solid food material in the stomach. Her bowel movements are normal, and she doesn’t have other gastrointestinal symptoms. She and her wife are considering having a third child, so she is worried about medications that may affect pregnancy or breast-feeding. How do you manage her nausea and vomiting?
After validating her concerns and performing a trauma-informed physical exam and encounter, you recommend a 4-hour gastric emptying test with a standard radiolabeled egg meal. Her gastric emptying does reveal significantly delayed gastric emptying at 2 and 4 hours. You discuss the risks and benefits of lifestyle modification (smaller frequent meals), initiating medications (erythromycin and metoclopramide) or cessation of cannabis (despite low likelihood of CHS). Desiring to avoid starting medications around initiation of pregnancy, she opts for the dietary approach and cessation of cannabis. You see her at a follow-up visit in 6 months, and her nausea is now only once a month, and she is excited to begin planning for a pregnancy using assisted reproductive technology.
Case 4
A 20-year-old nonbinary intersex individual (pronouns: he/they) (incorrectly assigned female at birth — is intersex with congenital adrenal hyperplasia) presents to the gastroenterology clinic with 8 years of heartburn, acid reflux, postprandial bloating, alternating diarrhea and constipation, nausea, and vomiting, complicated by avoidant restrictive food intake disorder. They have a history of bipolar II disorder with prior suicidal ideation. He has not yet had diagnostic workup as he previously had a bad encounter with a gastroenterologist where the gastroenterologist blamed his symptoms on his gender-affirming therapy, misgendered the patient, and told the patient their symptoms were “all in her [sic] head.”
You recognize that affirming their gender and using proper pronouns is the best first way to start rapport and help break the cycle of medicalized trauma. You then recommend a holistic work up with interdisciplinary management because of the complexity of his symptoms. For testing, you recommend a colonoscopy, upper endoscopy, a gastric emptying test with a 48-hour transit scintigraphy test, anorectal manometry, a dietitian referral, and a gastrointestinal psychology referral. Their anorectal manometry is consistent with an evacuation disorder. The rest of the work up is unremarkable. You diagnose them with anorectal pelvic floor dysfunction and functional dyspepsia, recommending biofeedback pelvic floor physical therapy, a proton-pump inhibitor, and neuromodulation in coordination with psychiatry and psychology to start with a plan for follow-up. The patient appreciates you for helping them and listening to their symptoms.
Discussion
When approaching DGBIs in the SGM community, it is vital to validate their concerns and be inclusive with diagnostic and treatment modalities. The diagnostic tools and treatments for DGBI are not different for patients in the SGM community. Like with other patients, trauma-informed care should be utilized, particularly given higher rates of trauma and discrimination in this community. Importantly, their DGBI is not a result of their sexual orientation or gender identity, and hormone therapy is not the cause of their DGBI. Recommending cessation of gender-affirming care or recommending lifestyle measures against their identity is generally not appropriate or necessary. among members of the SGM communities.
Dr. Jencks (@karajencks) is based in the division of gastroenterology and hepatology, Mayo Clinic, Rochester, Minnesota. Dr. Vélez (@Chris_Velez_MD) is based in the division of gastroenterology, Massachusetts General Hospital and Harvard Medical School, both in Boston. Both authors do not have any conflicts of interest for this article.
References
1. Duong N et al. 2023 Apr. doi: 10.1016/S2468-1253(23)00005-5.
2. Vélez C et al. Am J Gastroenterol. 2022 Jun. doi: 10.14309/ajg.0000000000001804.
3. Jones JM. Gallup. LGBTQ+ identification in U.S. now at 7.6%. 2024 Mar 13. https://news.gallup.com/poll/611864/lgbtq-identification.aspx
4. Sperber AD et al. Gastroenterology. 2021 Jan. doi: 10.1053/j.gastro.2020.04.014.
5. Wiley JW et al. Neurogastroenterol Motil. 2016 Jan. doi: 10.1111/nmo.12706.
6. Labanski A et al. Psychoneuroendocrinology. 2020 Jan. doi: 10.1016/j.psyneuen.2019.104501.
7. Khlevner J et al. Gastroenterol Clin North Am. 2018 Dec. doi: 10.1016/j.gtc.2018.07.002.
8. Jagielski CH and Harer KN. Gastroenterol Clin North Am. 2022 Dec. doi: 10.1016/j.gtc.2022.07.012.
9. Meyer IH. Psychol Bull. 2003 Sep. doi: 10.1037/0033-2909.129.5.674.
10. Mahurkar-Joshi S and Chang L. Front Psychiatry. 2020 Aug. doi: 10.3389/fpsyt.2020.00805.
Brief Introduction to the SGM Communities
The sexual and gender minority (SGM) communities (see Table 1), also termed “LGBTQIA+ community” (lesbian, gay, bisexual, transgender, queer, intersex, asexual, plus — including two spirit) are historically minoritized with unique risks for inequities in gastrointestinal health outcomes.1 These potential disparities remain largely uninvestigated because of continued systemic discrimination and inadequate collection of sexual orientation and gender identity (SOGI) data,2 with the National Institutes of Health Sexual & Gender Minority Research Office (SGMRO) having been instructed to address these failures. There is increased SGM self-identification (7.1% of all people in the United States and 20.8% of generation Z).3 Given the high worldwide prevalence of disorders of gut-brain interaction (DGBIs)and the influence of biopsychosocial determinants of health in DGBI incidence,4 it becomes increasingly likely that research in DGBI-related factors in SGM people will be fruitful.

Disorders of Gut-Brain Interaction and the Potential Minority Stress Link in SGM People
DGBIs are gastrointestinal conditions that occur because of brain-gut axis dysregulation. There is evidence that chronic stress and trauma negatively influence brain-gut interaction, which likely results in minority communities who face increased levels of trauma, stress, discrimination, and social injustice being at higher risk of DGBI development.5-7 Given increased rates of trauma in the SGM community, practicing trauma-informed care is essential to increase patient comfort and decrease the chance of retraumatization in medical settings.8 Trauma-informed care focuses on how trauma influences a patient’s life and response to medical care. To practice trauma-informed care, screening for trauma when appropriate, actively creating a supportive environment with active listening and communication, with informing the patient of planned actions prior to doing them, like physical exams, is key.
Trauma-Informed Care: Examples of Verbiage
Asking about Identity
- Begin by introducing yourself with your pronouns to create a safe environment for patient disclosure. Example: “Hello, I am Dr. Kara Jencks, and my pronouns are she/her. I am one of the gastroenterologists here at XYZ Clinic. How would you prefer to be addressed?”
- You can also wear a pronoun lapel pin or a pronoun button on your ID badge to indicate you are someone who your patient can be themselves around.
- The easiest way to obtain sexual orientation and gender identity is through intake forms. Below are examples of how to ask these questions on intake forms. It is important to offer the option to select more than one option when applicable and to opt out of answering if the patient is not comfortable answering these questions.
Sample Questions for Intake Forms
1. What is your sex assigned at birth? (Select one)
- Female
- Male
- Intersex
- Do not know
- Prefer not to disclose
2. What is your gender identity? (Select all that apply)
- Nonbinary
- Gender queer
- Woman
- Man
- Transwoman
- Transman
- Gender fluid
- Two-spirit
- Agender
- Intersex
- Other: type in response
- Prefer not to disclose
3. What are your pronouns? (Select all that apply)
- They/them/theirs
- She/her/hers
- He/him/his
- Zie/zir/zirs
- Other: type in response
- Prefer not to disclose
4. What is your sexual orientation? (Select all that apply)
- Bisexual
- Pansexual
- Queer
- Lesbian
- Gay
- Asexual
- Demisexual
- Heterosexual or straight
- Other: type in response
- Prefer not to disclose
Screening for Trauma
While there are questionnaires that exist to ask about trauma history, if time allows, it can be helpful to screen verbally with the patient. See reference number 8, for additional prompts and actions to practice trauma-informed care.
- Example: “Many patients with gastrointestinal symptoms and disorders have experienced trauma in the past. We do our best to ensure we are keeping you as comfortable as possible while caring for you. Are you comfortable sharing this information? [if yes->] Do you have a history of trauma, including physical, emotional, or sexual abuse? ... Have these experiences impacted the way in which you navigate your healthcare? ... Is there anything we can do to make you more comfortable today?”
General Physical Examination
Provide details for what you are going to do before you do it. Ask for permission for the examination. Here are two examples:
- “I would like to perform a physical exam to help better understand your symptoms. Is that okay with you?”
- “I would like to examine your abdomen with my stethoscope and my hands. Here is a sheet that we can use to help with your privacy. Please let me know if and when you feel any tenderness or pain.”
Rectal Physical Examination
Let the patient know why it would be helpful to perform a rectal exam, what the rectal exam will entail, and the benefits and risks to doing a rectal exam. An example follows:
- “Based on the symptoms you are describing, I think it would be helpful to perform a rectal exam to make sure you don’t have any fissures or hemorrhoids on the outside around the anus, any blockages or major issues inside the rectum, and to assess the strength and ability of your nerves and muscles or the pelvic floor to coordinate bowel movements. There are no risks aside from discomfort. If it is painful, and you would like me to stop, you tell me to stop, and I will stop right away. What questions do you have? Are we okay to proceed with the rectal exam?”
- “Please pull down your undergarments and your pants to either midthigh, your ankles, or all the way off, whatever your preference is, lie down on the left side on the exam table, and cover yourself with this sheet. In the meantime, I will be getting a chaperone to keep us safe and serve as a patient advocate during the procedure.”
- Upon returning to the exam room: “Here is Sara, who will be chaperoning today. Let myself or Sara know if you are uncomfortable or having pain during this exam. I will be lifting up the sheet to get a good look around the anus. [lifts up sheet] You will feel my hand helping to spread apart the buttocks. I am looking around the anus, and I do not see any fissures, hemorrhoids, or anything else concerning. Please squeeze in like you are trying to hold in gas. Please bear down like you are trying to have a bowel movement or let out gas. Okay, now you may feel some cold gel around the anus, and you will feel my finger go inside. Take a deep breath in. Do you feel any pain as I palpate? Please squeeze in like you are trying to hold in gas. Please bear down like you are trying to have a bowel movement or let out gas. I will be stopping the exam now.”
- You would then wash your hands and allow the patient to get dressed, and then disclose the exam findings and the rest of your visit.
Ilan H. Meyer coined the minority stress model when discussing mental health disorders in SGM patients in the early 2000s.9 With it being well known that DGBIs can overlap with (but are not necessarily caused by) mental health disorders, this model can easily apply to unify multiple individual and societal factors that can combine to result in disorders of brain-gut interaction (see Figure 1) in SGM communities. Let us keep this framework in mind when evaluating the following cases.

Case Presentations
Case 1
A 56-year-old man (pronouns: he/him) assigned male sex at birth, who identifies as gay, presents to your gastroenterology clinic for treatment-refractory constipation-predominant irritable bowel syndrome. It has impacted his sexual function. Outside hospital records report a normal colonoscopy 1 year ago and an unremarkable abdominal computerized tomography 4 months ago, aside from increased stool burden in the entire colon. He has tried to use enemas prior to sex, though these do not always help. Fiber-rich diet and fermentable food avoidance has not been successful. He is currently taking two capfuls of polyethylene glycol 3350 twice per day, as well as senna at night and continues to have a bowel movement every 2-3 days that is Bristol stool form scale type 1-2 unless he uses enemas. How do you counsel this patient about his IBS-C and rectal discomfort?
After assessing for sexual violence or other potential trauma-related factors, your digital rectal examination suggests that an anorectal defecatory disorder is less likely with normal relaxation and perineal movement. You recommend linaclotide. He notices improvement within 1 week, with improved comfort during anoreceptive sex.
Case 2
A 30-year-old woman (pronouns: she/her) assigned male sex at birth who has sex with men underwent vaginoplasty 2 years ago and is referred to the gastroenterology clinic for fecal incontinence and diarrhea. On review of her anatomic inventory, her vaginoplasty was a penile inversion vaginoplasty (no intestinal tissue was used for creation), and her prostate was left intact. The vaginal vault was created in between the urethra and rectum, similar to the pelvic floor anatomy of a woman assigned female sex at birth. Blood, imaging, and endoscopic workup has been negative. She is also not taking any medications associated with diarrhea, only taking estrogen and spironolactone. The diarrhea is not daily, but when present, about once per week, can be up to 10 episodes per day, and she has a sense of incomplete evacuation regularly. She notes having a rectal exam in the past but is not sure if her pelvic floor muscles have ever been assessed. How do you manage this patient?
To complete her evaluation in the office, you perform a trauma-informed rectal exam which reveals a decreased resting anal sphincter tone and paradoxical defecatory maneuvers without tenderness to the puborectalis muscle. Augmentation of the squeeze is also weak. Given her pelvic floor related surgical history, her symptoms, and her rectal exam, you recommend anorectal manometry which is abnormal and send her for anorectal biofeedback pelvic floor physical therapy, which improves her symptoms significantly.
Case 3
A 36-year-old woman (pronouns: she/her) assigned female sex at birth, who identifies as a lesbian, has a history of posttraumatic stress disorder and chronic nausea and vomiting that has begun to affect her quality of life. She notes the nausea and vomiting used to be managed well with evening cannabis gummies, though in the past 3 months, the nausea and vomiting has worsened, and she has lost 20 pounds as a result. As symptom predated cannabis usage, cannabis hyperemesis syndrome (CHS) was less likely (an important point as she has been stigmatized during prior encounters for her cannabis usage). Her primary care physician recommended a gastroscopy which was normal, aside from some residual solid food material in the stomach. Her bowel movements are normal, and she doesn’t have other gastrointestinal symptoms. She and her wife are considering having a third child, so she is worried about medications that may affect pregnancy or breast-feeding. How do you manage her nausea and vomiting?
After validating her concerns and performing a trauma-informed physical exam and encounter, you recommend a 4-hour gastric emptying test with a standard radiolabeled egg meal. Her gastric emptying does reveal significantly delayed gastric emptying at 2 and 4 hours. You discuss the risks and benefits of lifestyle modification (smaller frequent meals), initiating medications (erythromycin and metoclopramide) or cessation of cannabis (despite low likelihood of CHS). Desiring to avoid starting medications around initiation of pregnancy, she opts for the dietary approach and cessation of cannabis. You see her at a follow-up visit in 6 months, and her nausea is now only once a month, and she is excited to begin planning for a pregnancy using assisted reproductive technology.
Case 4
A 20-year-old nonbinary intersex individual (pronouns: he/they) (incorrectly assigned female at birth — is intersex with congenital adrenal hyperplasia) presents to the gastroenterology clinic with 8 years of heartburn, acid reflux, postprandial bloating, alternating diarrhea and constipation, nausea, and vomiting, complicated by avoidant restrictive food intake disorder. They have a history of bipolar II disorder with prior suicidal ideation. He has not yet had diagnostic workup as he previously had a bad encounter with a gastroenterologist where the gastroenterologist blamed his symptoms on his gender-affirming therapy, misgendered the patient, and told the patient their symptoms were “all in her [sic] head.”
You recognize that affirming their gender and using proper pronouns is the best first way to start rapport and help break the cycle of medicalized trauma. You then recommend a holistic work up with interdisciplinary management because of the complexity of his symptoms. For testing, you recommend a colonoscopy, upper endoscopy, a gastric emptying test with a 48-hour transit scintigraphy test, anorectal manometry, a dietitian referral, and a gastrointestinal psychology referral. Their anorectal manometry is consistent with an evacuation disorder. The rest of the work up is unremarkable. You diagnose them with anorectal pelvic floor dysfunction and functional dyspepsia, recommending biofeedback pelvic floor physical therapy, a proton-pump inhibitor, and neuromodulation in coordination with psychiatry and psychology to start with a plan for follow-up. The patient appreciates you for helping them and listening to their symptoms.
Discussion
When approaching DGBIs in the SGM community, it is vital to validate their concerns and be inclusive with diagnostic and treatment modalities. The diagnostic tools and treatments for DGBI are not different for patients in the SGM community. Like with other patients, trauma-informed care should be utilized, particularly given higher rates of trauma and discrimination in this community. Importantly, their DGBI is not a result of their sexual orientation or gender identity, and hormone therapy is not the cause of their DGBI. Recommending cessation of gender-affirming care or recommending lifestyle measures against their identity is generally not appropriate or necessary. among members of the SGM communities.
Dr. Jencks (@karajencks) is based in the division of gastroenterology and hepatology, Mayo Clinic, Rochester, Minnesota. Dr. Vélez (@Chris_Velez_MD) is based in the division of gastroenterology, Massachusetts General Hospital and Harvard Medical School, both in Boston. Both authors do not have any conflicts of interest for this article.
References
1. Duong N et al. 2023 Apr. doi: 10.1016/S2468-1253(23)00005-5.
2. Vélez C et al. Am J Gastroenterol. 2022 Jun. doi: 10.14309/ajg.0000000000001804.
3. Jones JM. Gallup. LGBTQ+ identification in U.S. now at 7.6%. 2024 Mar 13. https://news.gallup.com/poll/611864/lgbtq-identification.aspx
4. Sperber AD et al. Gastroenterology. 2021 Jan. doi: 10.1053/j.gastro.2020.04.014.
5. Wiley JW et al. Neurogastroenterol Motil. 2016 Jan. doi: 10.1111/nmo.12706.
6. Labanski A et al. Psychoneuroendocrinology. 2020 Jan. doi: 10.1016/j.psyneuen.2019.104501.
7. Khlevner J et al. Gastroenterol Clin North Am. 2018 Dec. doi: 10.1016/j.gtc.2018.07.002.
8. Jagielski CH and Harer KN. Gastroenterol Clin North Am. 2022 Dec. doi: 10.1016/j.gtc.2022.07.012.
9. Meyer IH. Psychol Bull. 2003 Sep. doi: 10.1037/0033-2909.129.5.674.
10. Mahurkar-Joshi S and Chang L. Front Psychiatry. 2020 Aug. doi: 10.3389/fpsyt.2020.00805.
Brief Introduction to the SGM Communities
The sexual and gender minority (SGM) communities (see Table 1), also termed “LGBTQIA+ community” (lesbian, gay, bisexual, transgender, queer, intersex, asexual, plus — including two spirit) are historically minoritized with unique risks for inequities in gastrointestinal health outcomes.1 These potential disparities remain largely uninvestigated because of continued systemic discrimination and inadequate collection of sexual orientation and gender identity (SOGI) data,2 with the National Institutes of Health Sexual & Gender Minority Research Office (SGMRO) having been instructed to address these failures. There is increased SGM self-identification (7.1% of all people in the United States and 20.8% of generation Z).3 Given the high worldwide prevalence of disorders of gut-brain interaction (DGBIs)and the influence of biopsychosocial determinants of health in DGBI incidence,4 it becomes increasingly likely that research in DGBI-related factors in SGM people will be fruitful.

Disorders of Gut-Brain Interaction and the Potential Minority Stress Link in SGM People
DGBIs are gastrointestinal conditions that occur because of brain-gut axis dysregulation. There is evidence that chronic stress and trauma negatively influence brain-gut interaction, which likely results in minority communities who face increased levels of trauma, stress, discrimination, and social injustice being at higher risk of DGBI development.5-7 Given increased rates of trauma in the SGM community, practicing trauma-informed care is essential to increase patient comfort and decrease the chance of retraumatization in medical settings.8 Trauma-informed care focuses on how trauma influences a patient’s life and response to medical care. To practice trauma-informed care, screening for trauma when appropriate, actively creating a supportive environment with active listening and communication, with informing the patient of planned actions prior to doing them, like physical exams, is key.
Trauma-Informed Care: Examples of Verbiage
Asking about Identity
- Begin by introducing yourself with your pronouns to create a safe environment for patient disclosure. Example: “Hello, I am Dr. Kara Jencks, and my pronouns are she/her. I am one of the gastroenterologists here at XYZ Clinic. How would you prefer to be addressed?”
- You can also wear a pronoun lapel pin or a pronoun button on your ID badge to indicate you are someone who your patient can be themselves around.
- The easiest way to obtain sexual orientation and gender identity is through intake forms. Below are examples of how to ask these questions on intake forms. It is important to offer the option to select more than one option when applicable and to opt out of answering if the patient is not comfortable answering these questions.
Sample Questions for Intake Forms
1. What is your sex assigned at birth? (Select one)
- Female
- Male
- Intersex
- Do not know
- Prefer not to disclose
2. What is your gender identity? (Select all that apply)
- Nonbinary
- Gender queer
- Woman
- Man
- Transwoman
- Transman
- Gender fluid
- Two-spirit
- Agender
- Intersex
- Other: type in response
- Prefer not to disclose
3. What are your pronouns? (Select all that apply)
- They/them/theirs
- She/her/hers
- He/him/his
- Zie/zir/zirs
- Other: type in response
- Prefer not to disclose
4. What is your sexual orientation? (Select all that apply)
- Bisexual
- Pansexual
- Queer
- Lesbian
- Gay
- Asexual
- Demisexual
- Heterosexual or straight
- Other: type in response
- Prefer not to disclose
Screening for Trauma
While there are questionnaires that exist to ask about trauma history, if time allows, it can be helpful to screen verbally with the patient. See reference number 8, for additional prompts and actions to practice trauma-informed care.
- Example: “Many patients with gastrointestinal symptoms and disorders have experienced trauma in the past. We do our best to ensure we are keeping you as comfortable as possible while caring for you. Are you comfortable sharing this information? [if yes->] Do you have a history of trauma, including physical, emotional, or sexual abuse? ... Have these experiences impacted the way in which you navigate your healthcare? ... Is there anything we can do to make you more comfortable today?”
General Physical Examination
Provide details for what you are going to do before you do it. Ask for permission for the examination. Here are two examples:
- “I would like to perform a physical exam to help better understand your symptoms. Is that okay with you?”
- “I would like to examine your abdomen with my stethoscope and my hands. Here is a sheet that we can use to help with your privacy. Please let me know if and when you feel any tenderness or pain.”
Rectal Physical Examination
Let the patient know why it would be helpful to perform a rectal exam, what the rectal exam will entail, and the benefits and risks to doing a rectal exam. An example follows:
- “Based on the symptoms you are describing, I think it would be helpful to perform a rectal exam to make sure you don’t have any fissures or hemorrhoids on the outside around the anus, any blockages or major issues inside the rectum, and to assess the strength and ability of your nerves and muscles or the pelvic floor to coordinate bowel movements. There are no risks aside from discomfort. If it is painful, and you would like me to stop, you tell me to stop, and I will stop right away. What questions do you have? Are we okay to proceed with the rectal exam?”
- “Please pull down your undergarments and your pants to either midthigh, your ankles, or all the way off, whatever your preference is, lie down on the left side on the exam table, and cover yourself with this sheet. In the meantime, I will be getting a chaperone to keep us safe and serve as a patient advocate during the procedure.”
- Upon returning to the exam room: “Here is Sara, who will be chaperoning today. Let myself or Sara know if you are uncomfortable or having pain during this exam. I will be lifting up the sheet to get a good look around the anus. [lifts up sheet] You will feel my hand helping to spread apart the buttocks. I am looking around the anus, and I do not see any fissures, hemorrhoids, or anything else concerning. Please squeeze in like you are trying to hold in gas. Please bear down like you are trying to have a bowel movement or let out gas. Okay, now you may feel some cold gel around the anus, and you will feel my finger go inside. Take a deep breath in. Do you feel any pain as I palpate? Please squeeze in like you are trying to hold in gas. Please bear down like you are trying to have a bowel movement or let out gas. I will be stopping the exam now.”
- You would then wash your hands and allow the patient to get dressed, and then disclose the exam findings and the rest of your visit.
Ilan H. Meyer coined the minority stress model when discussing mental health disorders in SGM patients in the early 2000s.9 With it being well known that DGBIs can overlap with (but are not necessarily caused by) mental health disorders, this model can easily apply to unify multiple individual and societal factors that can combine to result in disorders of brain-gut interaction (see Figure 1) in SGM communities. Let us keep this framework in mind when evaluating the following cases.

Case Presentations
Case 1
A 56-year-old man (pronouns: he/him) assigned male sex at birth, who identifies as gay, presents to your gastroenterology clinic for treatment-refractory constipation-predominant irritable bowel syndrome. It has impacted his sexual function. Outside hospital records report a normal colonoscopy 1 year ago and an unremarkable abdominal computerized tomography 4 months ago, aside from increased stool burden in the entire colon. He has tried to use enemas prior to sex, though these do not always help. Fiber-rich diet and fermentable food avoidance has not been successful. He is currently taking two capfuls of polyethylene glycol 3350 twice per day, as well as senna at night and continues to have a bowel movement every 2-3 days that is Bristol stool form scale type 1-2 unless he uses enemas. How do you counsel this patient about his IBS-C and rectal discomfort?
After assessing for sexual violence or other potential trauma-related factors, your digital rectal examination suggests that an anorectal defecatory disorder is less likely with normal relaxation and perineal movement. You recommend linaclotide. He notices improvement within 1 week, with improved comfort during anoreceptive sex.
Case 2
A 30-year-old woman (pronouns: she/her) assigned male sex at birth who has sex with men underwent vaginoplasty 2 years ago and is referred to the gastroenterology clinic for fecal incontinence and diarrhea. On review of her anatomic inventory, her vaginoplasty was a penile inversion vaginoplasty (no intestinal tissue was used for creation), and her prostate was left intact. The vaginal vault was created in between the urethra and rectum, similar to the pelvic floor anatomy of a woman assigned female sex at birth. Blood, imaging, and endoscopic workup has been negative. She is also not taking any medications associated with diarrhea, only taking estrogen and spironolactone. The diarrhea is not daily, but when present, about once per week, can be up to 10 episodes per day, and she has a sense of incomplete evacuation regularly. She notes having a rectal exam in the past but is not sure if her pelvic floor muscles have ever been assessed. How do you manage this patient?
To complete her evaluation in the office, you perform a trauma-informed rectal exam which reveals a decreased resting anal sphincter tone and paradoxical defecatory maneuvers without tenderness to the puborectalis muscle. Augmentation of the squeeze is also weak. Given her pelvic floor related surgical history, her symptoms, and her rectal exam, you recommend anorectal manometry which is abnormal and send her for anorectal biofeedback pelvic floor physical therapy, which improves her symptoms significantly.
Case 3
A 36-year-old woman (pronouns: she/her) assigned female sex at birth, who identifies as a lesbian, has a history of posttraumatic stress disorder and chronic nausea and vomiting that has begun to affect her quality of life. She notes the nausea and vomiting used to be managed well with evening cannabis gummies, though in the past 3 months, the nausea and vomiting has worsened, and she has lost 20 pounds as a result. As symptom predated cannabis usage, cannabis hyperemesis syndrome (CHS) was less likely (an important point as she has been stigmatized during prior encounters for her cannabis usage). Her primary care physician recommended a gastroscopy which was normal, aside from some residual solid food material in the stomach. Her bowel movements are normal, and she doesn’t have other gastrointestinal symptoms. She and her wife are considering having a third child, so she is worried about medications that may affect pregnancy or breast-feeding. How do you manage her nausea and vomiting?
After validating her concerns and performing a trauma-informed physical exam and encounter, you recommend a 4-hour gastric emptying test with a standard radiolabeled egg meal. Her gastric emptying does reveal significantly delayed gastric emptying at 2 and 4 hours. You discuss the risks and benefits of lifestyle modification (smaller frequent meals), initiating medications (erythromycin and metoclopramide) or cessation of cannabis (despite low likelihood of CHS). Desiring to avoid starting medications around initiation of pregnancy, she opts for the dietary approach and cessation of cannabis. You see her at a follow-up visit in 6 months, and her nausea is now only once a month, and she is excited to begin planning for a pregnancy using assisted reproductive technology.
Case 4
A 20-year-old nonbinary intersex individual (pronouns: he/they) (incorrectly assigned female at birth — is intersex with congenital adrenal hyperplasia) presents to the gastroenterology clinic with 8 years of heartburn, acid reflux, postprandial bloating, alternating diarrhea and constipation, nausea, and vomiting, complicated by avoidant restrictive food intake disorder. They have a history of bipolar II disorder with prior suicidal ideation. He has not yet had diagnostic workup as he previously had a bad encounter with a gastroenterologist where the gastroenterologist blamed his symptoms on his gender-affirming therapy, misgendered the patient, and told the patient their symptoms were “all in her [sic] head.”
You recognize that affirming their gender and using proper pronouns is the best first way to start rapport and help break the cycle of medicalized trauma. You then recommend a holistic work up with interdisciplinary management because of the complexity of his symptoms. For testing, you recommend a colonoscopy, upper endoscopy, a gastric emptying test with a 48-hour transit scintigraphy test, anorectal manometry, a dietitian referral, and a gastrointestinal psychology referral. Their anorectal manometry is consistent with an evacuation disorder. The rest of the work up is unremarkable. You diagnose them with anorectal pelvic floor dysfunction and functional dyspepsia, recommending biofeedback pelvic floor physical therapy, a proton-pump inhibitor, and neuromodulation in coordination with psychiatry and psychology to start with a plan for follow-up. The patient appreciates you for helping them and listening to their symptoms.
Discussion
When approaching DGBIs in the SGM community, it is vital to validate their concerns and be inclusive with diagnostic and treatment modalities. The diagnostic tools and treatments for DGBI are not different for patients in the SGM community. Like with other patients, trauma-informed care should be utilized, particularly given higher rates of trauma and discrimination in this community. Importantly, their DGBI is not a result of their sexual orientation or gender identity, and hormone therapy is not the cause of their DGBI. Recommending cessation of gender-affirming care or recommending lifestyle measures against their identity is generally not appropriate or necessary. among members of the SGM communities.
Dr. Jencks (@karajencks) is based in the division of gastroenterology and hepatology, Mayo Clinic, Rochester, Minnesota. Dr. Vélez (@Chris_Velez_MD) is based in the division of gastroenterology, Massachusetts General Hospital and Harvard Medical School, both in Boston. Both authors do not have any conflicts of interest for this article.
References
1. Duong N et al. 2023 Apr. doi: 10.1016/S2468-1253(23)00005-5.
2. Vélez C et al. Am J Gastroenterol. 2022 Jun. doi: 10.14309/ajg.0000000000001804.
3. Jones JM. Gallup. LGBTQ+ identification in U.S. now at 7.6%. 2024 Mar 13. https://news.gallup.com/poll/611864/lgbtq-identification.aspx
4. Sperber AD et al. Gastroenterology. 2021 Jan. doi: 10.1053/j.gastro.2020.04.014.
5. Wiley JW et al. Neurogastroenterol Motil. 2016 Jan. doi: 10.1111/nmo.12706.
6. Labanski A et al. Psychoneuroendocrinology. 2020 Jan. doi: 10.1016/j.psyneuen.2019.104501.
7. Khlevner J et al. Gastroenterol Clin North Am. 2018 Dec. doi: 10.1016/j.gtc.2018.07.002.
8. Jagielski CH and Harer KN. Gastroenterol Clin North Am. 2022 Dec. doi: 10.1016/j.gtc.2022.07.012.
9. Meyer IH. Psychol Bull. 2003 Sep. doi: 10.1037/0033-2909.129.5.674.
10. Mahurkar-Joshi S and Chang L. Front Psychiatry. 2020 Aug. doi: 10.3389/fpsyt.2020.00805.
Immunotherapy Reduces Skin Cancer Precursors
TOPLINE:
Immune checkpoint inhibitors (ICIs) show promise for field cancerization, based on their ability to reduce actinic keratoses (AKs) in a new study.
METHODOLOGY:
- This prospective cohort study included 23 immunocompetent participants (26.1% women; mean age, 69.7 years) from Australia who received ICIs for any cancer between April 2022 and November 2023.
- The most frequently prescribed ICI regimen was a combination of nivolumab and ipilimumab (34.8%), followed by nivolumab monotherapy (26.1%) and cemiplimab (21.7%) or pembrolizumab (17.4%) monotherapy.
- More than half of the patients received ICI therapy for skin cancer (melanoma, 30.4%; cutaneous squamous cell carcinoma, 26.1%); 34.8% had lung cancer; two had other carcinomas.
- The primary outcome was the number of AKs at 12 months after starting ICI therapy; the secondary outcome was the number of keratinocyte carcinomas (KCs) excised 12 months before and after ICI therapy.
TAKEAWAY:
- At 12 months, one patient had complete resolution from AK, and the mean number of AKs significantly decreased from 47.2 at baseline to 14.3 (P < .001).
- Younger patients (66.7% vs 33.3%; P = .007) and those with a history of blistering sunburn (100% vs 0; P = .005) were more likely to experience ≥ 65% reduction in AK count.
- KC incidence in the year before ICI therapy vs the year after initiation dropped from 42 to 17 cases, respectively, and the number of cutaneous squamous cell carcinomas decreased from 16 to 5.
- Adverse events occurred in 11 participants (47.8%), with maculopapular rash or pruritus the most common.
IN PRACTICE:
“This pilot cohort study highlights the potential association of ICI therapy, originally used in cancer treatment, with significant reduction of clinical AKs,” the authors wrote. These findings, they said, “underscore ICIs’ potential as a novel approach to mitigating field cancerization in high-risk populations.”
SOURCE:
Charlotte Cox, MD, MPhil, MPHTM, BMSt, University of Queensland, Brisbane, Australia, led the study, which was published online in JAMA Dermatology.
LIMITATIONS:
Limitations included interrater reliability issues in AK counting. Not all patients completed the follow-up period, and observations about changes after stopping ICI therapy were limited. Surveillance bias could be present in KC reporting.
DISCLOSURES:
This work was supported by grants from the Metro South Health SERTA project and by the French Society of Dermatology, La Ligue Contre le Cancer, the Collège des Enseignants en Dermatologie de France, and the European Association of Dermatology and Venereology. Cox received personal fees from the University of Queensland scholarship funds during this work. Some authors reported receiving personal fees and support from pharmaceutical and cosmetic companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Immune checkpoint inhibitors (ICIs) show promise for field cancerization, based on their ability to reduce actinic keratoses (AKs) in a new study.
METHODOLOGY:
- This prospective cohort study included 23 immunocompetent participants (26.1% women; mean age, 69.7 years) from Australia who received ICIs for any cancer between April 2022 and November 2023.
- The most frequently prescribed ICI regimen was a combination of nivolumab and ipilimumab (34.8%), followed by nivolumab monotherapy (26.1%) and cemiplimab (21.7%) or pembrolizumab (17.4%) monotherapy.
- More than half of the patients received ICI therapy for skin cancer (melanoma, 30.4%; cutaneous squamous cell carcinoma, 26.1%); 34.8% had lung cancer; two had other carcinomas.
- The primary outcome was the number of AKs at 12 months after starting ICI therapy; the secondary outcome was the number of keratinocyte carcinomas (KCs) excised 12 months before and after ICI therapy.
TAKEAWAY:
- At 12 months, one patient had complete resolution from AK, and the mean number of AKs significantly decreased from 47.2 at baseline to 14.3 (P < .001).
- Younger patients (66.7% vs 33.3%; P = .007) and those with a history of blistering sunburn (100% vs 0; P = .005) were more likely to experience ≥ 65% reduction in AK count.
- KC incidence in the year before ICI therapy vs the year after initiation dropped from 42 to 17 cases, respectively, and the number of cutaneous squamous cell carcinomas decreased from 16 to 5.
- Adverse events occurred in 11 participants (47.8%), with maculopapular rash or pruritus the most common.
IN PRACTICE:
“This pilot cohort study highlights the potential association of ICI therapy, originally used in cancer treatment, with significant reduction of clinical AKs,” the authors wrote. These findings, they said, “underscore ICIs’ potential as a novel approach to mitigating field cancerization in high-risk populations.”
SOURCE:
Charlotte Cox, MD, MPhil, MPHTM, BMSt, University of Queensland, Brisbane, Australia, led the study, which was published online in JAMA Dermatology.
LIMITATIONS:
Limitations included interrater reliability issues in AK counting. Not all patients completed the follow-up period, and observations about changes after stopping ICI therapy were limited. Surveillance bias could be present in KC reporting.
DISCLOSURES:
This work was supported by grants from the Metro South Health SERTA project and by the French Society of Dermatology, La Ligue Contre le Cancer, the Collège des Enseignants en Dermatologie de France, and the European Association of Dermatology and Venereology. Cox received personal fees from the University of Queensland scholarship funds during this work. Some authors reported receiving personal fees and support from pharmaceutical and cosmetic companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
Immune checkpoint inhibitors (ICIs) show promise for field cancerization, based on their ability to reduce actinic keratoses (AKs) in a new study.
METHODOLOGY:
- This prospective cohort study included 23 immunocompetent participants (26.1% women; mean age, 69.7 years) from Australia who received ICIs for any cancer between April 2022 and November 2023.
- The most frequently prescribed ICI regimen was a combination of nivolumab and ipilimumab (34.8%), followed by nivolumab monotherapy (26.1%) and cemiplimab (21.7%) or pembrolizumab (17.4%) monotherapy.
- More than half of the patients received ICI therapy for skin cancer (melanoma, 30.4%; cutaneous squamous cell carcinoma, 26.1%); 34.8% had lung cancer; two had other carcinomas.
- The primary outcome was the number of AKs at 12 months after starting ICI therapy; the secondary outcome was the number of keratinocyte carcinomas (KCs) excised 12 months before and after ICI therapy.
TAKEAWAY:
- At 12 months, one patient had complete resolution from AK, and the mean number of AKs significantly decreased from 47.2 at baseline to 14.3 (P < .001).
- Younger patients (66.7% vs 33.3%; P = .007) and those with a history of blistering sunburn (100% vs 0; P = .005) were more likely to experience ≥ 65% reduction in AK count.
- KC incidence in the year before ICI therapy vs the year after initiation dropped from 42 to 17 cases, respectively, and the number of cutaneous squamous cell carcinomas decreased from 16 to 5.
- Adverse events occurred in 11 participants (47.8%), with maculopapular rash or pruritus the most common.
IN PRACTICE:
“This pilot cohort study highlights the potential association of ICI therapy, originally used in cancer treatment, with significant reduction of clinical AKs,” the authors wrote. These findings, they said, “underscore ICIs’ potential as a novel approach to mitigating field cancerization in high-risk populations.”
SOURCE:
Charlotte Cox, MD, MPhil, MPHTM, BMSt, University of Queensland, Brisbane, Australia, led the study, which was published online in JAMA Dermatology.
LIMITATIONS:
Limitations included interrater reliability issues in AK counting. Not all patients completed the follow-up period, and observations about changes after stopping ICI therapy were limited. Surveillance bias could be present in KC reporting.
DISCLOSURES:
This work was supported by grants from the Metro South Health SERTA project and by the French Society of Dermatology, La Ligue Contre le Cancer, the Collège des Enseignants en Dermatologie de France, and the European Association of Dermatology and Venereology. Cox received personal fees from the University of Queensland scholarship funds during this work. Some authors reported receiving personal fees and support from pharmaceutical and cosmetic companies.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Promise for CAR T-Cell Therapies in Solid Tumors?
Chimeric antigen receptor (CAR) T-cell therapy has shown efficacy in blood cancers — with six CAR T-cell products now approved by the Food and Drug Administration (FDA) to treat six hematologic malignancies.
For solid tumors, however, the efficacy of CAR T-cell treatments has been limited and progress “more incremental,” Christian Hinrichs, MD, with Rutgers Cancer Institute in New Brunswick, New Jersey, told this news organization. Currently, there are no CAR T-cell therapies approved in the United States to treat solid tumors.
Why have CAR T-cell therapies been less effective against solid tumors?
Perhaps the biggest hurdle is the ability to identify and selectively target specific molecular structures in cancer cells without causing severe toxicity by injuring healthy cells, Hinrichs and coauthors wrote in a recent JAMA review.
CAR T-cells are made up of “T cells genetically engineered to express a synthetic receptor that recognizes a tumor cell-surface protein,” Hinrichs and colleagues explained. But identifying cell surface antigens that are exclusive to solid tumor cells has been a challenge, which means CAR T-cell therapies end up affecting both tumor and healthy tissues.
“This makes it difficult to target and kill all the tumor cells without causing severe toxicity from injury to healthy cells,” Hinrichs explained.
Other common obstacles include challenges penetrating the dense extracellular matrix of solid tumors and the need to overcome inhibitory cells and molecules in the tumor microenvironment.
Despite the challenges and slow progress, some “promising results” have begun to emerge in the solid tumor, CAR T-cell space, Hinrichs said.
A recent phase 1-2 study, for instance, found that 63% (17 of 27) of pediatric patients with heavily pretreated neuroblastoma achieved an overall response with an investigational CAR T-cell therapy, GD2-CART01.
In a recent phase 1 trial, 38 of 98 patients with gastrointestinal cancers (39%) achieved partial or complete responses after receiving an investigational CAR T-cell treatment directed at Claudin18.2. However, the responses were short overall and could have been related to the chemotherapy given before the CAR T-cell infusion.
Another phase 1 trial found that a GPC3-targeted CAR T-cell therapy led to an objective response rate in half (12 of 24) of heavily treated patients with advanced hepatocellular carcinoma, with a disease control rate of almost 91%.
Outside of CAR-T cell therapies, other cell-based treatments have shown promise against solid tumors, including two T-cell therapies recently approved by the FDA.
Last February, the FDA approved the tumor-infiltrating lymphocyte (TIL) therapy lifileucel (Amtagvi) for advanced melanoma. In August, the agency approved the T-cell receptor (TCR) therapy afamitresgene autoleucel for advanced synovial sarcoma.
“Response rates for these cellular therapies are in the 30% range, but already there is clear data that there’s durability for some patients, which is very exciting because previously treated patients really have very few treatment options,” Jennifer Brudno, MD, with the National Cancer Institute and coauthor of the JAMA review, said in a journal podcast.
Several cell-based agents are in early trials to treat a range of solid tumors.
Hinrichs and colleagues previously reported findings from a phase 2 clinical trial of TIL therapy for human papillomavirus (HPV) — associated cancers including cervical, oropharyngeal, and anal cancers. Responses occurred in 5 of 18 patients with cervical cancer and 2 of 11 patients with noncervical cancers. “Two of the patients with cervical cancer had complete responses that are ongoing years after a single infusion of cells,” Hinrichs told this news organization.
Hinrichs was also involved in a phase 1 trial of gene-engineered TCR T-cells targeting HPV E7 for HPV-associated cancers reported tumor responses in 6 of 12 patients, including 4 of 8 with tumors refractory to checkpoint blockade immunotherapy. A phase 2 trial is now open at Rutgers Cancer Institute, as is an early trial testing a new TCR T-cell therapy targeting Kita-Kyushu Lung Cancer Antigen-1 to treat metastatic gastric, lung, breast, and cervical cancers.
Despite the encouraging findings, for CAR T-cell and other cell-based therapies to be successful against solid tumors, “we need to develop more treatments directed against antigens that are expressed by most or all the cells in a tumor but not by critical healthy tissues,” Hinrichs said.
“It may also be important to increase the potency of therapeutic cells and develop more sophisticated methods of antigen targeting that can better distinguish between tumors and healthy tissues,” he noted.
Brudno reported being an unpaid scientific advisory board member for and receiving travel expenses from Kyverna Therapeutics. Hinrichs reported receiving personal fees from Neogene Therapeutics, Capstan Therapeutics, GlaxoSmithKline, Vir Biotechnology, and PACT Pharma; equity from Scarlet TCR (company officer); and sponsored research agreements from T-Cure Biosciences and Neogene Therapeutics outside the submitted work. He also holds several patents related to cellular therapies.
A version of this article first appeared on Medscape.com.
Chimeric antigen receptor (CAR) T-cell therapy has shown efficacy in blood cancers — with six CAR T-cell products now approved by the Food and Drug Administration (FDA) to treat six hematologic malignancies.
For solid tumors, however, the efficacy of CAR T-cell treatments has been limited and progress “more incremental,” Christian Hinrichs, MD, with Rutgers Cancer Institute in New Brunswick, New Jersey, told this news organization. Currently, there are no CAR T-cell therapies approved in the United States to treat solid tumors.
Why have CAR T-cell therapies been less effective against solid tumors?
Perhaps the biggest hurdle is the ability to identify and selectively target specific molecular structures in cancer cells without causing severe toxicity by injuring healthy cells, Hinrichs and coauthors wrote in a recent JAMA review.
CAR T-cells are made up of “T cells genetically engineered to express a synthetic receptor that recognizes a tumor cell-surface protein,” Hinrichs and colleagues explained. But identifying cell surface antigens that are exclusive to solid tumor cells has been a challenge, which means CAR T-cell therapies end up affecting both tumor and healthy tissues.
“This makes it difficult to target and kill all the tumor cells without causing severe toxicity from injury to healthy cells,” Hinrichs explained.
Other common obstacles include challenges penetrating the dense extracellular matrix of solid tumors and the need to overcome inhibitory cells and molecules in the tumor microenvironment.
Despite the challenges and slow progress, some “promising results” have begun to emerge in the solid tumor, CAR T-cell space, Hinrichs said.
A recent phase 1-2 study, for instance, found that 63% (17 of 27) of pediatric patients with heavily pretreated neuroblastoma achieved an overall response with an investigational CAR T-cell therapy, GD2-CART01.
In a recent phase 1 trial, 38 of 98 patients with gastrointestinal cancers (39%) achieved partial or complete responses after receiving an investigational CAR T-cell treatment directed at Claudin18.2. However, the responses were short overall and could have been related to the chemotherapy given before the CAR T-cell infusion.
Another phase 1 trial found that a GPC3-targeted CAR T-cell therapy led to an objective response rate in half (12 of 24) of heavily treated patients with advanced hepatocellular carcinoma, with a disease control rate of almost 91%.
Outside of CAR-T cell therapies, other cell-based treatments have shown promise against solid tumors, including two T-cell therapies recently approved by the FDA.
Last February, the FDA approved the tumor-infiltrating lymphocyte (TIL) therapy lifileucel (Amtagvi) for advanced melanoma. In August, the agency approved the T-cell receptor (TCR) therapy afamitresgene autoleucel for advanced synovial sarcoma.
“Response rates for these cellular therapies are in the 30% range, but already there is clear data that there’s durability for some patients, which is very exciting because previously treated patients really have very few treatment options,” Jennifer Brudno, MD, with the National Cancer Institute and coauthor of the JAMA review, said in a journal podcast.
Several cell-based agents are in early trials to treat a range of solid tumors.
Hinrichs and colleagues previously reported findings from a phase 2 clinical trial of TIL therapy for human papillomavirus (HPV) — associated cancers including cervical, oropharyngeal, and anal cancers. Responses occurred in 5 of 18 patients with cervical cancer and 2 of 11 patients with noncervical cancers. “Two of the patients with cervical cancer had complete responses that are ongoing years after a single infusion of cells,” Hinrichs told this news organization.
Hinrichs was also involved in a phase 1 trial of gene-engineered TCR T-cells targeting HPV E7 for HPV-associated cancers reported tumor responses in 6 of 12 patients, including 4 of 8 with tumors refractory to checkpoint blockade immunotherapy. A phase 2 trial is now open at Rutgers Cancer Institute, as is an early trial testing a new TCR T-cell therapy targeting Kita-Kyushu Lung Cancer Antigen-1 to treat metastatic gastric, lung, breast, and cervical cancers.
Despite the encouraging findings, for CAR T-cell and other cell-based therapies to be successful against solid tumors, “we need to develop more treatments directed against antigens that are expressed by most or all the cells in a tumor but not by critical healthy tissues,” Hinrichs said.
“It may also be important to increase the potency of therapeutic cells and develop more sophisticated methods of antigen targeting that can better distinguish between tumors and healthy tissues,” he noted.
Brudno reported being an unpaid scientific advisory board member for and receiving travel expenses from Kyverna Therapeutics. Hinrichs reported receiving personal fees from Neogene Therapeutics, Capstan Therapeutics, GlaxoSmithKline, Vir Biotechnology, and PACT Pharma; equity from Scarlet TCR (company officer); and sponsored research agreements from T-Cure Biosciences and Neogene Therapeutics outside the submitted work. He also holds several patents related to cellular therapies.
A version of this article first appeared on Medscape.com.
Chimeric antigen receptor (CAR) T-cell therapy has shown efficacy in blood cancers — with six CAR T-cell products now approved by the Food and Drug Administration (FDA) to treat six hematologic malignancies.
For solid tumors, however, the efficacy of CAR T-cell treatments has been limited and progress “more incremental,” Christian Hinrichs, MD, with Rutgers Cancer Institute in New Brunswick, New Jersey, told this news organization. Currently, there are no CAR T-cell therapies approved in the United States to treat solid tumors.
Why have CAR T-cell therapies been less effective against solid tumors?
Perhaps the biggest hurdle is the ability to identify and selectively target specific molecular structures in cancer cells without causing severe toxicity by injuring healthy cells, Hinrichs and coauthors wrote in a recent JAMA review.
CAR T-cells are made up of “T cells genetically engineered to express a synthetic receptor that recognizes a tumor cell-surface protein,” Hinrichs and colleagues explained. But identifying cell surface antigens that are exclusive to solid tumor cells has been a challenge, which means CAR T-cell therapies end up affecting both tumor and healthy tissues.
“This makes it difficult to target and kill all the tumor cells without causing severe toxicity from injury to healthy cells,” Hinrichs explained.
Other common obstacles include challenges penetrating the dense extracellular matrix of solid tumors and the need to overcome inhibitory cells and molecules in the tumor microenvironment.
Despite the challenges and slow progress, some “promising results” have begun to emerge in the solid tumor, CAR T-cell space, Hinrichs said.
A recent phase 1-2 study, for instance, found that 63% (17 of 27) of pediatric patients with heavily pretreated neuroblastoma achieved an overall response with an investigational CAR T-cell therapy, GD2-CART01.
In a recent phase 1 trial, 38 of 98 patients with gastrointestinal cancers (39%) achieved partial or complete responses after receiving an investigational CAR T-cell treatment directed at Claudin18.2. However, the responses were short overall and could have been related to the chemotherapy given before the CAR T-cell infusion.
Another phase 1 trial found that a GPC3-targeted CAR T-cell therapy led to an objective response rate in half (12 of 24) of heavily treated patients with advanced hepatocellular carcinoma, with a disease control rate of almost 91%.
Outside of CAR-T cell therapies, other cell-based treatments have shown promise against solid tumors, including two T-cell therapies recently approved by the FDA.
Last February, the FDA approved the tumor-infiltrating lymphocyte (TIL) therapy lifileucel (Amtagvi) for advanced melanoma. In August, the agency approved the T-cell receptor (TCR) therapy afamitresgene autoleucel for advanced synovial sarcoma.
“Response rates for these cellular therapies are in the 30% range, but already there is clear data that there’s durability for some patients, which is very exciting because previously treated patients really have very few treatment options,” Jennifer Brudno, MD, with the National Cancer Institute and coauthor of the JAMA review, said in a journal podcast.
Several cell-based agents are in early trials to treat a range of solid tumors.
Hinrichs and colleagues previously reported findings from a phase 2 clinical trial of TIL therapy for human papillomavirus (HPV) — associated cancers including cervical, oropharyngeal, and anal cancers. Responses occurred in 5 of 18 patients with cervical cancer and 2 of 11 patients with noncervical cancers. “Two of the patients with cervical cancer had complete responses that are ongoing years after a single infusion of cells,” Hinrichs told this news organization.
Hinrichs was also involved in a phase 1 trial of gene-engineered TCR T-cells targeting HPV E7 for HPV-associated cancers reported tumor responses in 6 of 12 patients, including 4 of 8 with tumors refractory to checkpoint blockade immunotherapy. A phase 2 trial is now open at Rutgers Cancer Institute, as is an early trial testing a new TCR T-cell therapy targeting Kita-Kyushu Lung Cancer Antigen-1 to treat metastatic gastric, lung, breast, and cervical cancers.
Despite the encouraging findings, for CAR T-cell and other cell-based therapies to be successful against solid tumors, “we need to develop more treatments directed against antigens that are expressed by most or all the cells in a tumor but not by critical healthy tissues,” Hinrichs said.
“It may also be important to increase the potency of therapeutic cells and develop more sophisticated methods of antigen targeting that can better distinguish between tumors and healthy tissues,” he noted.
Brudno reported being an unpaid scientific advisory board member for and receiving travel expenses from Kyverna Therapeutics. Hinrichs reported receiving personal fees from Neogene Therapeutics, Capstan Therapeutics, GlaxoSmithKline, Vir Biotechnology, and PACT Pharma; equity from Scarlet TCR (company officer); and sponsored research agreements from T-Cure Biosciences and Neogene Therapeutics outside the submitted work. He also holds several patents related to cellular therapies.
A version of this article first appeared on Medscape.com.
Landmark VA Study Uncovers Gene Variant Linked to Prostate Cancer
Only about 5% of hereditary prostate cancer (HPC) cases can be explained by known genetic variants, but a groundbreaking US Department of Veterans Affairs (VA) study could revolutionize the diagnosis, prevention, and treatment of HPC in a similar fashion that the discovery of the BRAC2 gene did in breast cancer.
The study, conducted at the VA Tennessee Valley Healthcare System in accordance with Vanderbilt University Medical Center and the VA Million Veteran Program (MVP), linked variants of the WNT9B gene with a greater risk of prostate cancer.
About 15,000 veterans are diagnosed with prostate cancer and treated at the VA annually, and > 200,000 veterans are prostate cancer survivors. According to Bruce Montgomery, MD, an oncologist with VA Puget Sound Health Care System, “Veterans are unique in that those men exposed to Agent Orange during the Vietnam War are at elevated risk for prostate cancer.” Montgomery added that germline pathogenic variants in genes such as BRCA2 and HOXB13 are other risk factors.
This genome-wide study searched for recurrently observed variants that carried the most risk. The study gathered data from a familial case-control population in the Nashville Familial Prostate Cancer Study (NFPCS) and International Consortium for Prostate Cancer Genetics (ICPCG). For evidence of replication, the study turned to 4 biobanks: the MVP, All of Us, the UK Biobank, and FinnGen.
The NFPCS is a case-control study based on family history. Patients included those undergoing treatment for prostate cancer and controls undergoing routine screening at Vanderbilt University Medical Center and the Nashville VA Medical Center between 2003 and 2009. Patients were included in the analysis if they had also had a first- or second-degree relative with prostate cancer.
The ICPCG dataset encompasses unrelated HPC cases aggregated from 12 study sites across Finland, France, Germany, the UK, and the US. The MVP is the nation’s largest biorepository of veteran data and has one of the world’s most diverse cohorts of any genetic research program. More than 1 million veterans are enrolled, and 800-plus researchers are working on > 100 projects.
Pathogenic variants of only 2 genes met the replication requirement with genome-wide significance: HOXB13 and WNT9B. HOXB13 has been reported on in the literature, but this is the first study to investigate WNT9B.
Researchers identified 2 variants of the WNT9B gene: WNT9B E152K carried 2.5-fold risk and reached genome-wide significance under meta-analysis, collectively encompassing one-half million patients. The association of WNT9B E152K with prostate cancer was supported by the familial study populations and each biobank, with genome-wide significance. Variant WNT9B Q47R reached genome-wide significance in the Finnish study. The Q47R founder haplotype was also carried by familial prostate cancer cases in the US and UK.
Autosomal dominant WNT9B pathogenic variants are already known to cause embryonic developmental sequence defects, leading later to prostatic cysts, enlarged prostate, and seminal vesicle cysts. Seminal vesicle adenocarcinoma (or squamous cell carcinoma) and clear cell carcinoma of the prostate have also been reported.
The study found that HOXB13 and WNT9B “share an unexpected commonality.” Both genes function in embryonic genitourinary development. WNT9B pathogenic variants cause the autosomal dominant Mayer-Rokitansky-Küster-Hauser syndrome, featuring genitourinary developmental defects. The study concluded: “Collectively, our observations implicate inherited variation in pathways guiding embryonic genitourinary development in the development of prostate cancer.”
“Significant investments” in VA-specific clinical trials recently have been pursued through a joint agreement between the VA and the Prostate Cancer Foundation, Montgomery said: “The Prostate Cancer Foundation is supporting tumor and germline sequencing of prostate cancer for veterans with advanced disease and providing resources to set up research infrastructure at 10 centers nationwide.”
The VA has also published a prostate cancer clinical pathway and is in the process of creating a national prostate cancer registry. Such a database, as well as the MVP are both unique to the VA and key to research such as the Predicting Metastatic Progression of High Risk Localized Prostate Cancer study, which began in 2023. Five VA medical centers are collaborating on an artificial intelligence algorithm that will detect patterns indicative of aggressive prostate cancer.
“A digital repository for data will allow for development, testing, and validation of prognostic classifiers that could positively impact clinical management of veterans with high-risk prostate cancer,” said Matthew Rettig, MD, chief of oncology and hematology at the Greater Los Angeles VA Medical Center who was coprincipal investigator for the study. “The infrastructure developed by this research will serve as a valuable hub for future discovery.”
About 12% of men with metastatic prostate cancer carry a pathogenic germline alteration that could warrant the use of PARP (poly [ADP-ribose] polymerase) inhibitors or platinum chemotherapy, neither of which is part of standard care. National Comprehensive Cancer Network guidelines recommend germline testing in men with metastatic prostate cancer. In addition, “the family members of veterans who carry these alterations could benefit from undergoing testing and taking advantage of potentially life-saving interventions and surveillance strategies if they are also carriers,” Montgomery wrote.
The VA is committed to improving access to germline testing for men with metastatic prostate cancer in several ways. Montgomery pointed to the system-wide VA genetic counseling and testing resource, the Genomic Medicine Service, and said somatic testing is available across the VA through the National Precision Oncology Program. Both programs can be extremely important to veterans because they provide access to precision oncology studies, along with off-label use of effective treatments.
Precision oncology is the most rapidly moving area in prostate cancer, according to Montgomery. “In the VA, this has been embraced as a very specific need to find these therapeutic options for all veterans as quickly as possible. I am most excited by how the enthusiasm for these approaches is supported at all levels, both nationally and locally, because it makes implementing very significant changes to research and treatment possible.”
Only about 5% of hereditary prostate cancer (HPC) cases can be explained by known genetic variants, but a groundbreaking US Department of Veterans Affairs (VA) study could revolutionize the diagnosis, prevention, and treatment of HPC in a similar fashion that the discovery of the BRAC2 gene did in breast cancer.
The study, conducted at the VA Tennessee Valley Healthcare System in accordance with Vanderbilt University Medical Center and the VA Million Veteran Program (MVP), linked variants of the WNT9B gene with a greater risk of prostate cancer.
About 15,000 veterans are diagnosed with prostate cancer and treated at the VA annually, and > 200,000 veterans are prostate cancer survivors. According to Bruce Montgomery, MD, an oncologist with VA Puget Sound Health Care System, “Veterans are unique in that those men exposed to Agent Orange during the Vietnam War are at elevated risk for prostate cancer.” Montgomery added that germline pathogenic variants in genes such as BRCA2 and HOXB13 are other risk factors.
This genome-wide study searched for recurrently observed variants that carried the most risk. The study gathered data from a familial case-control population in the Nashville Familial Prostate Cancer Study (NFPCS) and International Consortium for Prostate Cancer Genetics (ICPCG). For evidence of replication, the study turned to 4 biobanks: the MVP, All of Us, the UK Biobank, and FinnGen.
The NFPCS is a case-control study based on family history. Patients included those undergoing treatment for prostate cancer and controls undergoing routine screening at Vanderbilt University Medical Center and the Nashville VA Medical Center between 2003 and 2009. Patients were included in the analysis if they had also had a first- or second-degree relative with prostate cancer.
The ICPCG dataset encompasses unrelated HPC cases aggregated from 12 study sites across Finland, France, Germany, the UK, and the US. The MVP is the nation’s largest biorepository of veteran data and has one of the world’s most diverse cohorts of any genetic research program. More than 1 million veterans are enrolled, and 800-plus researchers are working on > 100 projects.
Pathogenic variants of only 2 genes met the replication requirement with genome-wide significance: HOXB13 and WNT9B. HOXB13 has been reported on in the literature, but this is the first study to investigate WNT9B.
Researchers identified 2 variants of the WNT9B gene: WNT9B E152K carried 2.5-fold risk and reached genome-wide significance under meta-analysis, collectively encompassing one-half million patients. The association of WNT9B E152K with prostate cancer was supported by the familial study populations and each biobank, with genome-wide significance. Variant WNT9B Q47R reached genome-wide significance in the Finnish study. The Q47R founder haplotype was also carried by familial prostate cancer cases in the US and UK.
Autosomal dominant WNT9B pathogenic variants are already known to cause embryonic developmental sequence defects, leading later to prostatic cysts, enlarged prostate, and seminal vesicle cysts. Seminal vesicle adenocarcinoma (or squamous cell carcinoma) and clear cell carcinoma of the prostate have also been reported.
The study found that HOXB13 and WNT9B “share an unexpected commonality.” Both genes function in embryonic genitourinary development. WNT9B pathogenic variants cause the autosomal dominant Mayer-Rokitansky-Küster-Hauser syndrome, featuring genitourinary developmental defects. The study concluded: “Collectively, our observations implicate inherited variation in pathways guiding embryonic genitourinary development in the development of prostate cancer.”
“Significant investments” in VA-specific clinical trials recently have been pursued through a joint agreement between the VA and the Prostate Cancer Foundation, Montgomery said: “The Prostate Cancer Foundation is supporting tumor and germline sequencing of prostate cancer for veterans with advanced disease and providing resources to set up research infrastructure at 10 centers nationwide.”
The VA has also published a prostate cancer clinical pathway and is in the process of creating a national prostate cancer registry. Such a database, as well as the MVP are both unique to the VA and key to research such as the Predicting Metastatic Progression of High Risk Localized Prostate Cancer study, which began in 2023. Five VA medical centers are collaborating on an artificial intelligence algorithm that will detect patterns indicative of aggressive prostate cancer.
“A digital repository for data will allow for development, testing, and validation of prognostic classifiers that could positively impact clinical management of veterans with high-risk prostate cancer,” said Matthew Rettig, MD, chief of oncology and hematology at the Greater Los Angeles VA Medical Center who was coprincipal investigator for the study. “The infrastructure developed by this research will serve as a valuable hub for future discovery.”
About 12% of men with metastatic prostate cancer carry a pathogenic germline alteration that could warrant the use of PARP (poly [ADP-ribose] polymerase) inhibitors or platinum chemotherapy, neither of which is part of standard care. National Comprehensive Cancer Network guidelines recommend germline testing in men with metastatic prostate cancer. In addition, “the family members of veterans who carry these alterations could benefit from undergoing testing and taking advantage of potentially life-saving interventions and surveillance strategies if they are also carriers,” Montgomery wrote.
The VA is committed to improving access to germline testing for men with metastatic prostate cancer in several ways. Montgomery pointed to the system-wide VA genetic counseling and testing resource, the Genomic Medicine Service, and said somatic testing is available across the VA through the National Precision Oncology Program. Both programs can be extremely important to veterans because they provide access to precision oncology studies, along with off-label use of effective treatments.
Precision oncology is the most rapidly moving area in prostate cancer, according to Montgomery. “In the VA, this has been embraced as a very specific need to find these therapeutic options for all veterans as quickly as possible. I am most excited by how the enthusiasm for these approaches is supported at all levels, both nationally and locally, because it makes implementing very significant changes to research and treatment possible.”
Only about 5% of hereditary prostate cancer (HPC) cases can be explained by known genetic variants, but a groundbreaking US Department of Veterans Affairs (VA) study could revolutionize the diagnosis, prevention, and treatment of HPC in a similar fashion that the discovery of the BRAC2 gene did in breast cancer.
The study, conducted at the VA Tennessee Valley Healthcare System in accordance with Vanderbilt University Medical Center and the VA Million Veteran Program (MVP), linked variants of the WNT9B gene with a greater risk of prostate cancer.
About 15,000 veterans are diagnosed with prostate cancer and treated at the VA annually, and > 200,000 veterans are prostate cancer survivors. According to Bruce Montgomery, MD, an oncologist with VA Puget Sound Health Care System, “Veterans are unique in that those men exposed to Agent Orange during the Vietnam War are at elevated risk for prostate cancer.” Montgomery added that germline pathogenic variants in genes such as BRCA2 and HOXB13 are other risk factors.
This genome-wide study searched for recurrently observed variants that carried the most risk. The study gathered data from a familial case-control population in the Nashville Familial Prostate Cancer Study (NFPCS) and International Consortium for Prostate Cancer Genetics (ICPCG). For evidence of replication, the study turned to 4 biobanks: the MVP, All of Us, the UK Biobank, and FinnGen.
The NFPCS is a case-control study based on family history. Patients included those undergoing treatment for prostate cancer and controls undergoing routine screening at Vanderbilt University Medical Center and the Nashville VA Medical Center between 2003 and 2009. Patients were included in the analysis if they had also had a first- or second-degree relative with prostate cancer.
The ICPCG dataset encompasses unrelated HPC cases aggregated from 12 study sites across Finland, France, Germany, the UK, and the US. The MVP is the nation’s largest biorepository of veteran data and has one of the world’s most diverse cohorts of any genetic research program. More than 1 million veterans are enrolled, and 800-plus researchers are working on > 100 projects.
Pathogenic variants of only 2 genes met the replication requirement with genome-wide significance: HOXB13 and WNT9B. HOXB13 has been reported on in the literature, but this is the first study to investigate WNT9B.
Researchers identified 2 variants of the WNT9B gene: WNT9B E152K carried 2.5-fold risk and reached genome-wide significance under meta-analysis, collectively encompassing one-half million patients. The association of WNT9B E152K with prostate cancer was supported by the familial study populations and each biobank, with genome-wide significance. Variant WNT9B Q47R reached genome-wide significance in the Finnish study. The Q47R founder haplotype was also carried by familial prostate cancer cases in the US and UK.
Autosomal dominant WNT9B pathogenic variants are already known to cause embryonic developmental sequence defects, leading later to prostatic cysts, enlarged prostate, and seminal vesicle cysts. Seminal vesicle adenocarcinoma (or squamous cell carcinoma) and clear cell carcinoma of the prostate have also been reported.
The study found that HOXB13 and WNT9B “share an unexpected commonality.” Both genes function in embryonic genitourinary development. WNT9B pathogenic variants cause the autosomal dominant Mayer-Rokitansky-Küster-Hauser syndrome, featuring genitourinary developmental defects. The study concluded: “Collectively, our observations implicate inherited variation in pathways guiding embryonic genitourinary development in the development of prostate cancer.”
“Significant investments” in VA-specific clinical trials recently have been pursued through a joint agreement between the VA and the Prostate Cancer Foundation, Montgomery said: “The Prostate Cancer Foundation is supporting tumor and germline sequencing of prostate cancer for veterans with advanced disease and providing resources to set up research infrastructure at 10 centers nationwide.”
The VA has also published a prostate cancer clinical pathway and is in the process of creating a national prostate cancer registry. Such a database, as well as the MVP are both unique to the VA and key to research such as the Predicting Metastatic Progression of High Risk Localized Prostate Cancer study, which began in 2023. Five VA medical centers are collaborating on an artificial intelligence algorithm that will detect patterns indicative of aggressive prostate cancer.
“A digital repository for data will allow for development, testing, and validation of prognostic classifiers that could positively impact clinical management of veterans with high-risk prostate cancer,” said Matthew Rettig, MD, chief of oncology and hematology at the Greater Los Angeles VA Medical Center who was coprincipal investigator for the study. “The infrastructure developed by this research will serve as a valuable hub for future discovery.”
About 12% of men with metastatic prostate cancer carry a pathogenic germline alteration that could warrant the use of PARP (poly [ADP-ribose] polymerase) inhibitors or platinum chemotherapy, neither of which is part of standard care. National Comprehensive Cancer Network guidelines recommend germline testing in men with metastatic prostate cancer. In addition, “the family members of veterans who carry these alterations could benefit from undergoing testing and taking advantage of potentially life-saving interventions and surveillance strategies if they are also carriers,” Montgomery wrote.
The VA is committed to improving access to germline testing for men with metastatic prostate cancer in several ways. Montgomery pointed to the system-wide VA genetic counseling and testing resource, the Genomic Medicine Service, and said somatic testing is available across the VA through the National Precision Oncology Program. Both programs can be extremely important to veterans because they provide access to precision oncology studies, along with off-label use of effective treatments.
Precision oncology is the most rapidly moving area in prostate cancer, according to Montgomery. “In the VA, this has been embraced as a very specific need to find these therapeutic options for all veterans as quickly as possible. I am most excited by how the enthusiasm for these approaches is supported at all levels, both nationally and locally, because it makes implementing very significant changes to research and treatment possible.”
COPD Guidelines Face Implementation Hurdles in Primary Care
TOPLINE: Chronic obstructive pulmonary disease (COPD) guidelines are significantly underutilized in clinical practice, with studies attempting to improve implementation yielding inconsistent results. A team of US Department of Veterans Affairs (VA) researchers developed a pilot program and surveyed both patients and primary care practitioners (PCPs) to better understand the barriers to guideline-based COPD care primary care settings.
METHODOLOGY:
- Researchers conducted a pilot study using an implementation design at the Central Arkansas Veterans Healthcare System (CAVHS) to explore implementation gaps in a primary care setting
- Analysis included semi-structured interviews with 17 respondents, comprising both patients and PCPs, to explore barriers and facilitators to 4 COPD clinical practice guidelines
- The Consolidated Framework of Implementation Science was used to design interview guides focusing on inhaler education, spirometry, pulmonary rehabilitation, and COPD-specific patient education
- Primary care teams followed a collaborative model including physicians, advanced practice nurses, nurses, social workers, pharmacists, and administrative staff working together with patients
TAKEAWAY:
- A total of 17 respondents, including patients and PCPs participated in the study, with the patient sample reflecting the general COPD population at CAVHS
- Both PCPs and patients consistently rated all assessed COPD clinical practice guidelines as highly important, despite significant practice gaps in implementation
- PCPs reported very low rates of providing education on inhaler use, citing time constraints, lack of educational resources, and low familiarity as primary barriers
- The main PCP-related barriers to pulmonary rehabilitation included limited knowledge about the program, unfamiliarity with CAVHS resources, and challenges with the referral process
IN PRACTICE: "Reasons behind this insufficient uptake of COPD guidelines include providers' low familiarity with guidelines, perception of minimal value of guidelines, and time constraints. Studies attempting to improve COPD-CPG uptake have shown mixed results and the best practice to bridge this implementation gap remains unknown," wrote the authors of the study.[Note To Staff: This quote was picked by Plume]
SOURCE: The study was led by Deepa Raghavan, Karen L Drummond, Sonya Sanders, and JoAnn Kirchner at Central Arkansas Veterans Healthcare System. It was published online in Chronic Respiratory Disease.
TOPLINE: Chronic obstructive pulmonary disease (COPD) guidelines are significantly underutilized in clinical practice, with studies attempting to improve implementation yielding inconsistent results. A team of US Department of Veterans Affairs (VA) researchers developed a pilot program and surveyed both patients and primary care practitioners (PCPs) to better understand the barriers to guideline-based COPD care primary care settings.
METHODOLOGY:
- Researchers conducted a pilot study using an implementation design at the Central Arkansas Veterans Healthcare System (CAVHS) to explore implementation gaps in a primary care setting
- Analysis included semi-structured interviews with 17 respondents, comprising both patients and PCPs, to explore barriers and facilitators to 4 COPD clinical practice guidelines
- The Consolidated Framework of Implementation Science was used to design interview guides focusing on inhaler education, spirometry, pulmonary rehabilitation, and COPD-specific patient education
- Primary care teams followed a collaborative model including physicians, advanced practice nurses, nurses, social workers, pharmacists, and administrative staff working together with patients
TAKEAWAY:
- A total of 17 respondents, including patients and PCPs participated in the study, with the patient sample reflecting the general COPD population at CAVHS
- Both PCPs and patients consistently rated all assessed COPD clinical practice guidelines as highly important, despite significant practice gaps in implementation
- PCPs reported very low rates of providing education on inhaler use, citing time constraints, lack of educational resources, and low familiarity as primary barriers
- The main PCP-related barriers to pulmonary rehabilitation included limited knowledge about the program, unfamiliarity with CAVHS resources, and challenges with the referral process
IN PRACTICE: "Reasons behind this insufficient uptake of COPD guidelines include providers' low familiarity with guidelines, perception of minimal value of guidelines, and time constraints. Studies attempting to improve COPD-CPG uptake have shown mixed results and the best practice to bridge this implementation gap remains unknown," wrote the authors of the study.[Note To Staff: This quote was picked by Plume]
SOURCE: The study was led by Deepa Raghavan, Karen L Drummond, Sonya Sanders, and JoAnn Kirchner at Central Arkansas Veterans Healthcare System. It was published online in Chronic Respiratory Disease.
TOPLINE: Chronic obstructive pulmonary disease (COPD) guidelines are significantly underutilized in clinical practice, with studies attempting to improve implementation yielding inconsistent results. A team of US Department of Veterans Affairs (VA) researchers developed a pilot program and surveyed both patients and primary care practitioners (PCPs) to better understand the barriers to guideline-based COPD care primary care settings.
METHODOLOGY:
- Researchers conducted a pilot study using an implementation design at the Central Arkansas Veterans Healthcare System (CAVHS) to explore implementation gaps in a primary care setting
- Analysis included semi-structured interviews with 17 respondents, comprising both patients and PCPs, to explore barriers and facilitators to 4 COPD clinical practice guidelines
- The Consolidated Framework of Implementation Science was used to design interview guides focusing on inhaler education, spirometry, pulmonary rehabilitation, and COPD-specific patient education
- Primary care teams followed a collaborative model including physicians, advanced practice nurses, nurses, social workers, pharmacists, and administrative staff working together with patients
TAKEAWAY:
- A total of 17 respondents, including patients and PCPs participated in the study, with the patient sample reflecting the general COPD population at CAVHS
- Both PCPs and patients consistently rated all assessed COPD clinical practice guidelines as highly important, despite significant practice gaps in implementation
- PCPs reported very low rates of providing education on inhaler use, citing time constraints, lack of educational resources, and low familiarity as primary barriers
- The main PCP-related barriers to pulmonary rehabilitation included limited knowledge about the program, unfamiliarity with CAVHS resources, and challenges with the referral process
IN PRACTICE: "Reasons behind this insufficient uptake of COPD guidelines include providers' low familiarity with guidelines, perception of minimal value of guidelines, and time constraints. Studies attempting to improve COPD-CPG uptake have shown mixed results and the best practice to bridge this implementation gap remains unknown," wrote the authors of the study.[Note To Staff: This quote was picked by Plume]
SOURCE: The study was led by Deepa Raghavan, Karen L Drummond, Sonya Sanders, and JoAnn Kirchner at Central Arkansas Veterans Healthcare System. It was published online in Chronic Respiratory Disease.
Post-COVID Cough Linked to Neurological Dysfunction
Chronic cough remains a common reason for consultation in pulmonology post–COVID-19. But what do we really know about this condition, now 5 years after the pandemic’s onset? This topic was discussed at the recent French-Speaking Pneumology Congress held in Marseille, France, from January 24-26, 2025.
Before discussing post-COVID cough, it is crucial to differentiate between an acute cough, often viral in origin (including those associated with SARS-CoV-2), a subacute cough (lasting 3-8 weeks), and a chronic cough (persisting over 8 weeks).
“This distinction allows us to tailor treatment and prescribe the appropriate investigations, according to the duration and the probability of symptom resolution,” explained Laurent Guilleminault, MD, PhD, pulmonologist at Toulouse University Hospital Centre, Toulouse, France.
In the case of an acute cough, for instance, after a viral infection, the probability of spontaneous resolution is very high. It is often unnecessary to carry out additional examinations or initiate specific treatments because none has proven its effectiveness in shortening this type of cough. On the other hand, when a cough persists beyond 8 weeks, the chance of spontaneous resolution decreases considerably. “This is when an assessment is necessary to identify a possible underlying cause,” Guilleminault noted.
“The absence of coughing during the consultation should not lead to ruling out a diagnosis,” he added.
Neurological Link
A large-scale French study of 70,000 patients examined the demographic profiles of patients with COVID-19. It revealed a lower frequency of coughing among children and older individuals, with a notable prevalence among adults aged 30-60 years.
Furthermore, during the acute phase of COVID, coughing did not appear to indicate severity. A comparison between survivors and nonsurvivors revealed no significant differences in the frequency and severity of coughing. Another study concluded that, contrary to expectations, COVID-related pneumonia, although potentially severe, does not necessarily involve severe cough.
These findings highlight the absence of a direct link between coughing and pulmonary involvement in patients with COVID-19.
“Coughing appears to be more closely linked to neurological dysfunction than to classic respiratory involvement. A distinction that is essential for better understanding the pathophysiology of the disease and guiding therapeutic strategies,” Guilleminault noted.
Cough Mechanism
“The analysis of cough in the context of phylogenetic evolution is fascinating,” explained Guilleminault. “It illustrates how this reflex has provided an advantage to the virus for its propagation.” Studies on the transmission of SARS-CoV-2 have confirmed that coughing plays a key role in the spread of viral particles. However, this mechanism does not involve severe pulmonary damage. The primary goal of the virus is to induce neurological dysfunction in the host by triggering a cough reflex. This neurological activation enables the virus to trigger a cough reflex for dissemination even without significant pulmonary damage. This mechanism provides an evolutionary advantage by enhancing the ability of the virus to spread and colonize new hosts.
The cough mechanism remains partially understood and involves cough hypersensitivity, characterized by increased neural responsivity to a range of stimuli that affect the airways, lungs, and other tissues innervated by common nerve supplies. The cough reflex begins with the activation of sensitive peripheral receptors located mainly in the respiratory tract that detect irritants or abnormalities.
These receptors, such as P2X2, P2X3, and others, transmit information to the brainstem, which coordinates the reflex response. This process is modulated by cortical controls that normally inhibit spontaneous coughing, explaining why we do not cough constantly even in the presence of moderate stimuli.
However, when there is an imbalance in this inhibition mechanism, coughing can be triggered either excessively or uncontrollably. SARS-CoV-2 appears to interact directly with these peripheral receptors, stimulating the cough reflex. The widespread presence and density of these receptors make this mechanism highly effective for the virus’s transmission.
Additionally, the vagus nerve likely plays a central role in triggering cough, particularly in viral infections. Studies of influenza have shown the involvement of sensory cells associated with the vagus nerve.
The virus stimulates the vagus nerve, which activates the cough reflex. Research suggests that neurotropism, neuroinflammation, and neuroimmunomodulation via the vagal sensory nerves, which are involved in SARS-CoV-2 infection, lead to cough hypersensitivity.
One question remains: Could vagus nerve involvement prolong coughing beyond the active phase of viral infection? The data indicate that viral infection significantly increases the sensitivity of the cough reflex, regardless of the level of irritation. The brain areas involved in inhibiting this reflex appear less effective during viral infection, resulting in reduced inhibitory control and easier triggering of cough. This phenomenon reflects temporary dysfunction of the neurological modulation system, which gradually recovers after recovery.
Long-Term Effects
The epidemiology of post-COVID cough and its integration into the framework of the long COVID framework remain subjects of ongoing debate. Early studies have revealed that cough could be either an isolated symptom or associated with other manifestations of long COVID. These studies were often conducted over relatively short periods (14-110 days) and estimated that approximately 19% of patients with long COVID experienced persistent cough. Another study found that 14% of patients reported cough between 3 weeks and 3 months after hospital discharge for COVID-19.
Longer follow-up periods showed a significant decrease in the prevalence of cough over time. For instance, a 1-year study reported that only 2.5% of patients had episodes of chronic cough.
However, a 2023 study published in JAMA found that the prevalence of post-COVID chronic cough exceeded 30% in some groups of patients.
“It is not relevant to wait so long before acting,” Guilleminault said. A reasonable threshold for evaluation and treatment is 8-12 weeks postinfection to begin investigations and consider appropriate treatment. What should be done when a patient presents with “Doctor, I had COVID, I have a cough, and it hasn’t stopped?” These situations are common in clinical practice. In terms of severity, quality of life, and overall impact, patients with chronic post-COVID cough are not significantly different from those with other chronic coughs. Moreover, both conditions involve a real neurological dysfunction.
Same Diagnostic Steps
Management should follow existing guidelines, including the recent French recommendations for chronic cough.
A visual analog scale can be used, and possible complications should be assessed. A chest x-ray is recommended to identify any warning signs, such as cough, although linked to COVID — may coincide with other conditions, such as bronchial cancer. In smokers, chest CT should be considered to rule out neoplastic pathology. The presence of interstitial lesions, particularly fibrosing lesions, suggests that fibrosing interstitial pneumonia requires specialized management.
Smoking, which is an aggravating factor, should be discontinued. Discontinuing angiotensin-converting enzyme inhibitors for 4 weeks can help determine if they contribute to cough.
The three most common causes of chronic cough — rhinosinusitis, asthma, and gastroesophageal reflux disease — should be ruled out. Diagnosis is based on history, physical examination, and specific tests: Nasofibroscopy for rhinosinusitis, spirometry, fractional exhaled nitric oxide for asthma and clinical history of gastroesophageal reflux disease. Studies have indicated that asthma may develop after a COVID infection.
Laryngeal abnormalities are also common in chronic post-COVID cough. One study found that a quarter of patients had increased laryngeal sensitivity or voice changes. “The larynx, a highly cough-producing organ, causes more coughing than the lungs,” Guilleminault explained.
Laryngeal abnormalities are frequently observed. A study found that 63% of patients experienced dysphonia, 56% had a sensation of a foreign body in the larynx, and 10% experienced laryngospasms.
These issues are common in patients with post-COVID cough and are often associated with neurological dysfunction. Innervation of the larynx is complex and can be affected by viruses, leading to hypersensitivity, paresthesia, and other sensory disturbances, which may explain the laryngeal symptoms observed in these patients.
Next Steps
If common causes such as asthma, abnormal imaging findings, or laryngeal pathology are ruled out, the condition may be classified as a chronic refractory or unexplained cough. In these cases, the neurological origin is likely due to nervous system dysfunction. Neuromodulatory treatments including amitriptyline, pregabalin, and gabapentin may be considered in some cases. Corticosteroids are generally ineffective against chronic coughs.
This story was translated from Medscape’s French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Chronic cough remains a common reason for consultation in pulmonology post–COVID-19. But what do we really know about this condition, now 5 years after the pandemic’s onset? This topic was discussed at the recent French-Speaking Pneumology Congress held in Marseille, France, from January 24-26, 2025.
Before discussing post-COVID cough, it is crucial to differentiate between an acute cough, often viral in origin (including those associated with SARS-CoV-2), a subacute cough (lasting 3-8 weeks), and a chronic cough (persisting over 8 weeks).
“This distinction allows us to tailor treatment and prescribe the appropriate investigations, according to the duration and the probability of symptom resolution,” explained Laurent Guilleminault, MD, PhD, pulmonologist at Toulouse University Hospital Centre, Toulouse, France.
In the case of an acute cough, for instance, after a viral infection, the probability of spontaneous resolution is very high. It is often unnecessary to carry out additional examinations or initiate specific treatments because none has proven its effectiveness in shortening this type of cough. On the other hand, when a cough persists beyond 8 weeks, the chance of spontaneous resolution decreases considerably. “This is when an assessment is necessary to identify a possible underlying cause,” Guilleminault noted.
“The absence of coughing during the consultation should not lead to ruling out a diagnosis,” he added.
Neurological Link
A large-scale French study of 70,000 patients examined the demographic profiles of patients with COVID-19. It revealed a lower frequency of coughing among children and older individuals, with a notable prevalence among adults aged 30-60 years.
Furthermore, during the acute phase of COVID, coughing did not appear to indicate severity. A comparison between survivors and nonsurvivors revealed no significant differences in the frequency and severity of coughing. Another study concluded that, contrary to expectations, COVID-related pneumonia, although potentially severe, does not necessarily involve severe cough.
These findings highlight the absence of a direct link between coughing and pulmonary involvement in patients with COVID-19.
“Coughing appears to be more closely linked to neurological dysfunction than to classic respiratory involvement. A distinction that is essential for better understanding the pathophysiology of the disease and guiding therapeutic strategies,” Guilleminault noted.
Cough Mechanism
“The analysis of cough in the context of phylogenetic evolution is fascinating,” explained Guilleminault. “It illustrates how this reflex has provided an advantage to the virus for its propagation.” Studies on the transmission of SARS-CoV-2 have confirmed that coughing plays a key role in the spread of viral particles. However, this mechanism does not involve severe pulmonary damage. The primary goal of the virus is to induce neurological dysfunction in the host by triggering a cough reflex. This neurological activation enables the virus to trigger a cough reflex for dissemination even without significant pulmonary damage. This mechanism provides an evolutionary advantage by enhancing the ability of the virus to spread and colonize new hosts.
The cough mechanism remains partially understood and involves cough hypersensitivity, characterized by increased neural responsivity to a range of stimuli that affect the airways, lungs, and other tissues innervated by common nerve supplies. The cough reflex begins with the activation of sensitive peripheral receptors located mainly in the respiratory tract that detect irritants or abnormalities.
These receptors, such as P2X2, P2X3, and others, transmit information to the brainstem, which coordinates the reflex response. This process is modulated by cortical controls that normally inhibit spontaneous coughing, explaining why we do not cough constantly even in the presence of moderate stimuli.
However, when there is an imbalance in this inhibition mechanism, coughing can be triggered either excessively or uncontrollably. SARS-CoV-2 appears to interact directly with these peripheral receptors, stimulating the cough reflex. The widespread presence and density of these receptors make this mechanism highly effective for the virus’s transmission.
Additionally, the vagus nerve likely plays a central role in triggering cough, particularly in viral infections. Studies of influenza have shown the involvement of sensory cells associated with the vagus nerve.
The virus stimulates the vagus nerve, which activates the cough reflex. Research suggests that neurotropism, neuroinflammation, and neuroimmunomodulation via the vagal sensory nerves, which are involved in SARS-CoV-2 infection, lead to cough hypersensitivity.
One question remains: Could vagus nerve involvement prolong coughing beyond the active phase of viral infection? The data indicate that viral infection significantly increases the sensitivity of the cough reflex, regardless of the level of irritation. The brain areas involved in inhibiting this reflex appear less effective during viral infection, resulting in reduced inhibitory control and easier triggering of cough. This phenomenon reflects temporary dysfunction of the neurological modulation system, which gradually recovers after recovery.
Long-Term Effects
The epidemiology of post-COVID cough and its integration into the framework of the long COVID framework remain subjects of ongoing debate. Early studies have revealed that cough could be either an isolated symptom or associated with other manifestations of long COVID. These studies were often conducted over relatively short periods (14-110 days) and estimated that approximately 19% of patients with long COVID experienced persistent cough. Another study found that 14% of patients reported cough between 3 weeks and 3 months after hospital discharge for COVID-19.
Longer follow-up periods showed a significant decrease in the prevalence of cough over time. For instance, a 1-year study reported that only 2.5% of patients had episodes of chronic cough.
However, a 2023 study published in JAMA found that the prevalence of post-COVID chronic cough exceeded 30% in some groups of patients.
“It is not relevant to wait so long before acting,” Guilleminault said. A reasonable threshold for evaluation and treatment is 8-12 weeks postinfection to begin investigations and consider appropriate treatment. What should be done when a patient presents with “Doctor, I had COVID, I have a cough, and it hasn’t stopped?” These situations are common in clinical practice. In terms of severity, quality of life, and overall impact, patients with chronic post-COVID cough are not significantly different from those with other chronic coughs. Moreover, both conditions involve a real neurological dysfunction.
Same Diagnostic Steps
Management should follow existing guidelines, including the recent French recommendations for chronic cough.
A visual analog scale can be used, and possible complications should be assessed. A chest x-ray is recommended to identify any warning signs, such as cough, although linked to COVID — may coincide with other conditions, such as bronchial cancer. In smokers, chest CT should be considered to rule out neoplastic pathology. The presence of interstitial lesions, particularly fibrosing lesions, suggests that fibrosing interstitial pneumonia requires specialized management.
Smoking, which is an aggravating factor, should be discontinued. Discontinuing angiotensin-converting enzyme inhibitors for 4 weeks can help determine if they contribute to cough.
The three most common causes of chronic cough — rhinosinusitis, asthma, and gastroesophageal reflux disease — should be ruled out. Diagnosis is based on history, physical examination, and specific tests: Nasofibroscopy for rhinosinusitis, spirometry, fractional exhaled nitric oxide for asthma and clinical history of gastroesophageal reflux disease. Studies have indicated that asthma may develop after a COVID infection.
Laryngeal abnormalities are also common in chronic post-COVID cough. One study found that a quarter of patients had increased laryngeal sensitivity or voice changes. “The larynx, a highly cough-producing organ, causes more coughing than the lungs,” Guilleminault explained.
Laryngeal abnormalities are frequently observed. A study found that 63% of patients experienced dysphonia, 56% had a sensation of a foreign body in the larynx, and 10% experienced laryngospasms.
These issues are common in patients with post-COVID cough and are often associated with neurological dysfunction. Innervation of the larynx is complex and can be affected by viruses, leading to hypersensitivity, paresthesia, and other sensory disturbances, which may explain the laryngeal symptoms observed in these patients.
Next Steps
If common causes such as asthma, abnormal imaging findings, or laryngeal pathology are ruled out, the condition may be classified as a chronic refractory or unexplained cough. In these cases, the neurological origin is likely due to nervous system dysfunction. Neuromodulatory treatments including amitriptyline, pregabalin, and gabapentin may be considered in some cases. Corticosteroids are generally ineffective against chronic coughs.
This story was translated from Medscape’s French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Chronic cough remains a common reason for consultation in pulmonology post–COVID-19. But what do we really know about this condition, now 5 years after the pandemic’s onset? This topic was discussed at the recent French-Speaking Pneumology Congress held in Marseille, France, from January 24-26, 2025.
Before discussing post-COVID cough, it is crucial to differentiate between an acute cough, often viral in origin (including those associated with SARS-CoV-2), a subacute cough (lasting 3-8 weeks), and a chronic cough (persisting over 8 weeks).
“This distinction allows us to tailor treatment and prescribe the appropriate investigations, according to the duration and the probability of symptom resolution,” explained Laurent Guilleminault, MD, PhD, pulmonologist at Toulouse University Hospital Centre, Toulouse, France.
In the case of an acute cough, for instance, after a viral infection, the probability of spontaneous resolution is very high. It is often unnecessary to carry out additional examinations or initiate specific treatments because none has proven its effectiveness in shortening this type of cough. On the other hand, when a cough persists beyond 8 weeks, the chance of spontaneous resolution decreases considerably. “This is when an assessment is necessary to identify a possible underlying cause,” Guilleminault noted.
“The absence of coughing during the consultation should not lead to ruling out a diagnosis,” he added.
Neurological Link
A large-scale French study of 70,000 patients examined the demographic profiles of patients with COVID-19. It revealed a lower frequency of coughing among children and older individuals, with a notable prevalence among adults aged 30-60 years.
Furthermore, during the acute phase of COVID, coughing did not appear to indicate severity. A comparison between survivors and nonsurvivors revealed no significant differences in the frequency and severity of coughing. Another study concluded that, contrary to expectations, COVID-related pneumonia, although potentially severe, does not necessarily involve severe cough.
These findings highlight the absence of a direct link between coughing and pulmonary involvement in patients with COVID-19.
“Coughing appears to be more closely linked to neurological dysfunction than to classic respiratory involvement. A distinction that is essential for better understanding the pathophysiology of the disease and guiding therapeutic strategies,” Guilleminault noted.
Cough Mechanism
“The analysis of cough in the context of phylogenetic evolution is fascinating,” explained Guilleminault. “It illustrates how this reflex has provided an advantage to the virus for its propagation.” Studies on the transmission of SARS-CoV-2 have confirmed that coughing plays a key role in the spread of viral particles. However, this mechanism does not involve severe pulmonary damage. The primary goal of the virus is to induce neurological dysfunction in the host by triggering a cough reflex. This neurological activation enables the virus to trigger a cough reflex for dissemination even without significant pulmonary damage. This mechanism provides an evolutionary advantage by enhancing the ability of the virus to spread and colonize new hosts.
The cough mechanism remains partially understood and involves cough hypersensitivity, characterized by increased neural responsivity to a range of stimuli that affect the airways, lungs, and other tissues innervated by common nerve supplies. The cough reflex begins with the activation of sensitive peripheral receptors located mainly in the respiratory tract that detect irritants or abnormalities.
These receptors, such as P2X2, P2X3, and others, transmit information to the brainstem, which coordinates the reflex response. This process is modulated by cortical controls that normally inhibit spontaneous coughing, explaining why we do not cough constantly even in the presence of moderate stimuli.
However, when there is an imbalance in this inhibition mechanism, coughing can be triggered either excessively or uncontrollably. SARS-CoV-2 appears to interact directly with these peripheral receptors, stimulating the cough reflex. The widespread presence and density of these receptors make this mechanism highly effective for the virus’s transmission.
Additionally, the vagus nerve likely plays a central role in triggering cough, particularly in viral infections. Studies of influenza have shown the involvement of sensory cells associated with the vagus nerve.
The virus stimulates the vagus nerve, which activates the cough reflex. Research suggests that neurotropism, neuroinflammation, and neuroimmunomodulation via the vagal sensory nerves, which are involved in SARS-CoV-2 infection, lead to cough hypersensitivity.
One question remains: Could vagus nerve involvement prolong coughing beyond the active phase of viral infection? The data indicate that viral infection significantly increases the sensitivity of the cough reflex, regardless of the level of irritation. The brain areas involved in inhibiting this reflex appear less effective during viral infection, resulting in reduced inhibitory control and easier triggering of cough. This phenomenon reflects temporary dysfunction of the neurological modulation system, which gradually recovers after recovery.
Long-Term Effects
The epidemiology of post-COVID cough and its integration into the framework of the long COVID framework remain subjects of ongoing debate. Early studies have revealed that cough could be either an isolated symptom or associated with other manifestations of long COVID. These studies were often conducted over relatively short periods (14-110 days) and estimated that approximately 19% of patients with long COVID experienced persistent cough. Another study found that 14% of patients reported cough between 3 weeks and 3 months after hospital discharge for COVID-19.
Longer follow-up periods showed a significant decrease in the prevalence of cough over time. For instance, a 1-year study reported that only 2.5% of patients had episodes of chronic cough.
However, a 2023 study published in JAMA found that the prevalence of post-COVID chronic cough exceeded 30% in some groups of patients.
“It is not relevant to wait so long before acting,” Guilleminault said. A reasonable threshold for evaluation and treatment is 8-12 weeks postinfection to begin investigations and consider appropriate treatment. What should be done when a patient presents with “Doctor, I had COVID, I have a cough, and it hasn’t stopped?” These situations are common in clinical practice. In terms of severity, quality of life, and overall impact, patients with chronic post-COVID cough are not significantly different from those with other chronic coughs. Moreover, both conditions involve a real neurological dysfunction.
Same Diagnostic Steps
Management should follow existing guidelines, including the recent French recommendations for chronic cough.
A visual analog scale can be used, and possible complications should be assessed. A chest x-ray is recommended to identify any warning signs, such as cough, although linked to COVID — may coincide with other conditions, such as bronchial cancer. In smokers, chest CT should be considered to rule out neoplastic pathology. The presence of interstitial lesions, particularly fibrosing lesions, suggests that fibrosing interstitial pneumonia requires specialized management.
Smoking, which is an aggravating factor, should be discontinued. Discontinuing angiotensin-converting enzyme inhibitors for 4 weeks can help determine if they contribute to cough.
The three most common causes of chronic cough — rhinosinusitis, asthma, and gastroesophageal reflux disease — should be ruled out. Diagnosis is based on history, physical examination, and specific tests: Nasofibroscopy for rhinosinusitis, spirometry, fractional exhaled nitric oxide for asthma and clinical history of gastroesophageal reflux disease. Studies have indicated that asthma may develop after a COVID infection.
Laryngeal abnormalities are also common in chronic post-COVID cough. One study found that a quarter of patients had increased laryngeal sensitivity or voice changes. “The larynx, a highly cough-producing organ, causes more coughing than the lungs,” Guilleminault explained.
Laryngeal abnormalities are frequently observed. A study found that 63% of patients experienced dysphonia, 56% had a sensation of a foreign body in the larynx, and 10% experienced laryngospasms.
These issues are common in patients with post-COVID cough and are often associated with neurological dysfunction. Innervation of the larynx is complex and can be affected by viruses, leading to hypersensitivity, paresthesia, and other sensory disturbances, which may explain the laryngeal symptoms observed in these patients.
Next Steps
If common causes such as asthma, abnormal imaging findings, or laryngeal pathology are ruled out, the condition may be classified as a chronic refractory or unexplained cough. In these cases, the neurological origin is likely due to nervous system dysfunction. Neuromodulatory treatments including amitriptyline, pregabalin, and gabapentin may be considered in some cases. Corticosteroids are generally ineffective against chronic coughs.
This story was translated from Medscape’s French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Higher Forced Vital Capacity Associated With Lower Risk for Cardiometabolic Disease Onset
TOPLINE:
Better lung function, expressed as higher forced vital capacity (FVC), is associated with a reduced risk for the onset of heart disease, diabetes, and stroke over a follow-up period of approximately 10 years, according to a cross-sectional analysis of a population-based study.
METHODOLOGY:
- Researchers conducted a cross-sectional analysis of a population-based study (the BOLD study) between 2003 and 2016 to investigate the association between lung function and the onset of cardiometabolic diseases by using data from participants across 15 countries.
- Overall, 5916 participants (mean age, 54 years; 55% women) were included, and the mean follow-up duration was 9.5 years.
- Lung function was evaluated as forced expiratory volume in 1 second (FEV1), FVC, and FEV1/FVC ratio, measured using spirometry at baseline, and postbronchodilator values of these measures were expressed as the percent of the predicted values at baseline.
- The onset of cardiometabolic diseases was identified through participant-reported doctor diagnoses of hypertension, heart disease, diabetes, and stroke at follow-up but not at baseline.
TAKEAWAY:
- Each 10% predicted FVC was associated with a 9% reduced risk for the onset of diabetes (adjusted odds ratio [aOR], 0.91; 95% CI, 0.84-0.99), a 14% reduced risk for the onset of heart disease (aOR, 0.86; 95% CI, 0.80-0.92), and a 19% reduced risk for the onset of stroke (aOR, 0.81; 95% CI, 0.73-0.89).
- Each 10% predicted FEV1 was associated with a reduced risk for the onset of heart disease (aOR, 0.88; 95% CI, 0.83-0.94) and stroke (aOR, 0.83; 95% CI, 0.76-0.90).
- A high FEV1/FVC ratio was associated with an increased risk for the onset of diabetes (aOR per 10%, 1.21; 95% CI, 1.08-1.35) but not associated with other cardiometabolic diseases.
- Moderate heterogeneity was observed across study sites regarding the association between high lung function and the risk for the onset of diabetes and stroke.
IN PRACTICE:
“FVC is not included in any risk score for predicting the risk of cardiometabolic events, although data also suggests that FVC predicted mortality more strongly than systolic blood pressure or BMI [body mass index]. Our results and several previous studies suggest that including FVC will improve the precision of risk scores used to predict the onset of diabetes and cardiovascular diseases,” the authors wrote.
SOURCE:
This study was led by Christer Janson, Department of Medical Sciences Respiratory Medicine, Uppsala Universitet, Uppsala, Sweden. It was published online on January 19, 2025, in BMJ Open Respiratory Research.
LIMITATIONS:
The primary limitation of this study was the reliance on the self-reported onset of cardiometabolic diseases, which is particularly challenging in low- and middle-income countries with underdeveloped healthcare systems. The observed outcomes could be the result of an undiagnosed condition. The data did not allow differentiation between various types of heart diseases or strokes.
DISCLOSURES:
The BOLD study received support through grants from the Wellcome Trust and Medical Research Council, and the follow-up study at some centers was supported by an unrestricted grant from AstraZeneca. Four authors reported receiving support from various sources related or unrelated to this work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE:
Better lung function, expressed as higher forced vital capacity (FVC), is associated with a reduced risk for the onset of heart disease, diabetes, and stroke over a follow-up period of approximately 10 years, according to a cross-sectional analysis of a population-based study.
METHODOLOGY:
- Researchers conducted a cross-sectional analysis of a population-based study (the BOLD study) between 2003 and 2016 to investigate the association between lung function and the onset of cardiometabolic diseases by using data from participants across 15 countries.
- Overall, 5916 participants (mean age, 54 years; 55% women) were included, and the mean follow-up duration was 9.5 years.
- Lung function was evaluated as forced expiratory volume in 1 second (FEV1), FVC, and FEV1/FVC ratio, measured using spirometry at baseline, and postbronchodilator values of these measures were expressed as the percent of the predicted values at baseline.
- The onset of cardiometabolic diseases was identified through participant-reported doctor diagnoses of hypertension, heart disease, diabetes, and stroke at follow-up but not at baseline.
TAKEAWAY:
- Each 10% predicted FVC was associated with a 9% reduced risk for the onset of diabetes (adjusted odds ratio [aOR], 0.91; 95% CI, 0.84-0.99), a 14% reduced risk for the onset of heart disease (aOR, 0.86; 95% CI, 0.80-0.92), and a 19% reduced risk for the onset of stroke (aOR, 0.81; 95% CI, 0.73-0.89).
- Each 10% predicted FEV1 was associated with a reduced risk for the onset of heart disease (aOR, 0.88; 95% CI, 0.83-0.94) and stroke (aOR, 0.83; 95% CI, 0.76-0.90).
- A high FEV1/FVC ratio was associated with an increased risk for the onset of diabetes (aOR per 10%, 1.21; 95% CI, 1.08-1.35) but not associated with other cardiometabolic diseases.
- Moderate heterogeneity was observed across study sites regarding the association between high lung function and the risk for the onset of diabetes and stroke.
IN PRACTICE:
“FVC is not included in any risk score for predicting the risk of cardiometabolic events, although data also suggests that FVC predicted mortality more strongly than systolic blood pressure or BMI [body mass index]. Our results and several previous studies suggest that including FVC will improve the precision of risk scores used to predict the onset of diabetes and cardiovascular diseases,” the authors wrote.
SOURCE:
This study was led by Christer Janson, Department of Medical Sciences Respiratory Medicine, Uppsala Universitet, Uppsala, Sweden. It was published online on January 19, 2025, in BMJ Open Respiratory Research.
LIMITATIONS:
The primary limitation of this study was the reliance on the self-reported onset of cardiometabolic diseases, which is particularly challenging in low- and middle-income countries with underdeveloped healthcare systems. The observed outcomes could be the result of an undiagnosed condition. The data did not allow differentiation between various types of heart diseases or strokes.
DISCLOSURES:
The BOLD study received support through grants from the Wellcome Trust and Medical Research Council, and the follow-up study at some centers was supported by an unrestricted grant from AstraZeneca. Four authors reported receiving support from various sources related or unrelated to this work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
TOPLINE:
Better lung function, expressed as higher forced vital capacity (FVC), is associated with a reduced risk for the onset of heart disease, diabetes, and stroke over a follow-up period of approximately 10 years, according to a cross-sectional analysis of a population-based study.
METHODOLOGY:
- Researchers conducted a cross-sectional analysis of a population-based study (the BOLD study) between 2003 and 2016 to investigate the association between lung function and the onset of cardiometabolic diseases by using data from participants across 15 countries.
- Overall, 5916 participants (mean age, 54 years; 55% women) were included, and the mean follow-up duration was 9.5 years.
- Lung function was evaluated as forced expiratory volume in 1 second (FEV1), FVC, and FEV1/FVC ratio, measured using spirometry at baseline, and postbronchodilator values of these measures were expressed as the percent of the predicted values at baseline.
- The onset of cardiometabolic diseases was identified through participant-reported doctor diagnoses of hypertension, heart disease, diabetes, and stroke at follow-up but not at baseline.
TAKEAWAY:
- Each 10% predicted FVC was associated with a 9% reduced risk for the onset of diabetes (adjusted odds ratio [aOR], 0.91; 95% CI, 0.84-0.99), a 14% reduced risk for the onset of heart disease (aOR, 0.86; 95% CI, 0.80-0.92), and a 19% reduced risk for the onset of stroke (aOR, 0.81; 95% CI, 0.73-0.89).
- Each 10% predicted FEV1 was associated with a reduced risk for the onset of heart disease (aOR, 0.88; 95% CI, 0.83-0.94) and stroke (aOR, 0.83; 95% CI, 0.76-0.90).
- A high FEV1/FVC ratio was associated with an increased risk for the onset of diabetes (aOR per 10%, 1.21; 95% CI, 1.08-1.35) but not associated with other cardiometabolic diseases.
- Moderate heterogeneity was observed across study sites regarding the association between high lung function and the risk for the onset of diabetes and stroke.
IN PRACTICE:
“FVC is not included in any risk score for predicting the risk of cardiometabolic events, although data also suggests that FVC predicted mortality more strongly than systolic blood pressure or BMI [body mass index]. Our results and several previous studies suggest that including FVC will improve the precision of risk scores used to predict the onset of diabetes and cardiovascular diseases,” the authors wrote.
SOURCE:
This study was led by Christer Janson, Department of Medical Sciences Respiratory Medicine, Uppsala Universitet, Uppsala, Sweden. It was published online on January 19, 2025, in BMJ Open Respiratory Research.
LIMITATIONS:
The primary limitation of this study was the reliance on the self-reported onset of cardiometabolic diseases, which is particularly challenging in low- and middle-income countries with underdeveloped healthcare systems. The observed outcomes could be the result of an undiagnosed condition. The data did not allow differentiation between various types of heart diseases or strokes.
DISCLOSURES:
The BOLD study received support through grants from the Wellcome Trust and Medical Research Council, and the follow-up study at some centers was supported by an unrestricted grant from AstraZeneca. Four authors reported receiving support from various sources related or unrelated to this work.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
Surgical vs Endoscopic Excision of Large Colon Polyps
Dear colleagues,
We now have the ability to remove almost any large colon polyp endoscopically using a variety of techniques — from the widely used endoscopic mucosal resection to the increasingly prevalent endoscopic submucosal dissection. Yet, in this new era,
In this issue of Perspectives, Dr. Jeffrey Mosko and Dr. Moamen Gabr discuss the importance of careful polyp selection and argue that almost all polyps can be safely removed endoscopically, with low recurrence rates. In contrast, Dr. Ira Leeds from colorectal surgery offers a counterpoint, urging caution when managing polyps in the cecum and rectum while highlighting the role of minimally invasive surgical approaches. We hope these discussions provide valuable insights to support your approach to managing large colorectal polyps, especially in an era of increasing colon cancer screening.
We also welcome your thoughts on this topic — join the conversation on X at @AGA_GIHN.
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, and chief of endoscopy at West Haven VA Medical Center, both in Connecticut. He is an associate editor for GI & Hepatology News.
Advantages of Endoscopic Resection for Large Colon Polyps
BY MOAMEN GABR, MD, MSC, AND JEFFREY D. MOSKO MD, MSC
General Advantages
Endoscopy has revolutionized the management of large colorectal polyps, offering a minimally invasive alternative to surgical resection. The dawn of endoscopic resection in the late 20th century, particularly the evolution of endoscopic mucosal resection (EMR) in Japan, marked a paradigm shift in the treatment of colonic lesions by enabling the removal of lesions that would otherwise necessitate surgery.
Endoscopic resection of colorectal polyps is generally performed in an outpatient setting, allowing patients to recover at home the same day. This not only minimizes disruption to daily life but also significantly enhances patient satisfaction.
Most procedures are performed under moderate or deep sedation eliminating the need for general anesthesia. This represents a critical benefit, particularly for older or medically frail patients who are at higher risk of anesthesia-related complications.
From an economic perspective, endoscopic resection reduces healthcare costs by eliminating prolonged hospital stays and complex perioperative care. Additionally, preserving the colon’s structure and function avoids long-term consequences such as altered bowel habits or ostomy dependence, common with surgical interventions.
The advantages of endoscopic intervention are clear: safety, cost-effectiveness, organ preservation, and convenience for patients.
Lesion Selection
The superiority of endoscopic resection relies on selecting lesions appropriately, specifically those with a low risk of lymph node metastases. This meticulous process should include assessing a lesion’s size, location, morphology, granularity, microvascular and surface pit pattern using a combination of high-definition white light endoscopy, virtual chromoendoscopy and image magnification (when available).
Gross morphologic assessment utilizes the Paris and LST classifications. Combining the Paris classification, lesion granularity and location is both straightforward and revealing. Ulcerated/excavated lesions (0-III) are concerning for deep invasion. Depressed (0-IIc) morphologies are strongly associated with T1 CRC. Nodular lesions (0-Is or IIa + Is) have a higher risk of T1 colorectal cancer (CRC), compared with flat lesions (0-IIa or 0-IIb). Non-granular lesions (0-Is and 0-IIa + Is) have a higher risk of covert cancer. Finally, the rectosigmoid location is associated with an increased risk of T1 CRC (vs. proximal locations).
Endoscopic surface pattern assessment increases one’s diagnostic accuracy. There are three primary endoscopic surface pattern classifications: NBI International Colorectal Endoscopic (NICE), Japanese NBI expert team (JNET), and Kudo pit pattern classifications. Colonic lesions that have a NICE Type 3, JNET 3, or Kudo type Vn pattern should be referred promptly for surgical resection. Lesions with a JNET 2B or Kudo type VI carry a higher risk of superficial T1 CRC but can still be removed endoscopically (see below) in expert centers. All other lesions should undergo endoscopic resection.
Endoscopic Resection Techniques
Endoscopic resection of large colorectal polyps encompasses two primary techniques: EMR and endoscopic submucosal dissection (ESD), each tailored to specific lesion characteristics and operator expertise.
EMR, the technique of choice for the vast majority of lesions, relies on injecting a submucosal cushion to lift the lesion before excision. Recent advances, including enhanced snare designs and underwater EMR, have improved en-bloc resection rates, significantly reducing recurrence and enhancing the efficacy of this technique.
ESD offers unparalleled precision for en-bloc resection of complex lesions, particularly those with fibrosis or high-risk features. Cutting-edge innovations, such as traction devices, have streamlined the procedure, addressing the traditional challenges of ESD. Despite being more time intensive, ESD minimizes recurrence and provides complete histopathological evaluation, critical for the management of malignant or pre-malignant lesions.
For non-lifting polyps, newer techniques such as endoscopic full-thickness resection (eFTR), using tools like the Full-Thickness Resection Device (FTRD), enable resection of up to 2-3 cm of the colonic or rectal wall. This ensures complete removal of any lesion and its underlying tissue, effectively preventing recurrence.
These advancements demonstrate how endoscopy can tackle even the most challenging colorectal polyps, reinforcing its position as the preferred treatment modality.
Perceived Limitations
With ongoing refinement over the last 2 decades, many of the perceived limitations (below) of endoscopic resection have now been overcome.
- Difficult locations/access: Historically lesions at the anorectal junction, ileocecal valve, appendiceal orifice and anastomoses were preferentially sent for surgery. In spite of unique technical challenges at each of these locations, there is now compelling data supporting EMR for these scenarios. We now also have techniques aimed at enabling the resection of lesions with poor access including patient repositioning, distal attachments, variable endoscope diameter/flexibility, traction and overtube devices.
- Recurrence: In the past, recurrence after endoscopic resection of lesions > 20 mm has been reported to be as high as 20%. With our current systematic approach to complete resection, meticulous examination of the post-resection defect for residual polyp tissue, adjunctive techniques to address submucosal fibrosis (hot avulsion, CAST, submucosal release) and thermal ablation to the resection margin (EMR-T), the risk of recurrence for piecemeal resections can be decreased to < 5%. In fact, some groups argue for the en-bloc resection of all large colorectal lesions based on the extremely low (< 1%) recurrence rates and potential for decreased follow-up.
- Post-resection bleeding: Post-resection bleeding is no longer a major limitation of any endoscopic approach because of the combination of improved intra-procedural hemostatic and resection techniques, optimized electrosurgical technology, and enhanced defect closure capabilities and devices (with prophylactic defect closure now supported by randomized control trial level data).
- Perforation: Deep mural injury, once an endoscopists’ worst fear during resection, is no longer a surgical emergency. It can now be predicted, identified (Sydney classification) and successfully managed. In spite of more widespread aggressive resection strategies, the risk of emergency surgery in patients undergoing EMR and even ESD (where the risk of DMI is significantly higher) is extremely low.
Endoscopic resection for large colorectal polyps is effective, available, minimally invasive and organ sparing making it the standard of care for the management of colonic polyps. With ongoing iteration in techniques, more invasive surgical approaches can be avoided in almost all patients with benign and low-risk T1 colorectal cancers.
Dr. Gabr is associate GI division director at the University of Cincinnati, Ohio. Dr. Mosko is based in the division of gastroenterology at St. Michael’s Hospital, Toronto, Ontario, Canada. The authors declare no conflicts of interest.
Blurred Lines: Polyp Needing Surgical versus Endoscopic Excision
BY IRA LEEDS, MD
I am grateful for the invitation to join in discussion with Dr. Gabr and Dr. Mosko on the ever-increasing role of endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD). However, as a surgeon, I do carry at least mild trepidation entering one of the literary “safe spaces” of my gastroenterology colleagues.
With the increasing evidentiary support of EMR approaches and the increasing experience of those performing ESD, these two techniques are quickly becoming the options of choice. As these practices become ubiquitous, it is important to recognize both their advantages and limitations, compared with available surgical options. The decision to proceed with EMR and ESD is essentially a turning point away from early surgical referral for a complex lesion. In this discussion, I intend to highlight when EMR and ESD have a clear advantage to early surgical referral, why I believe that early surgical referral is still superior to advanced endoscopic techniques in the rectum, and why the approach for right-sided lesions should hinge on careful shared decision-making.
Endoscopic approaches nearly always beat surgical approaches when considering short-term risks. Even in the best surgical series, colorectal surgery typically leads to complications in 10%-15% of patients, 1%-5% being serious. Moreover, transabdominal surgical interventions (ie, colectomy) require considerable recovery involving at least a few days in the inpatient setting and over a month of activity restrictions. Finally, there is a minority of chronically unwell patients who cannot tolerate surgical intervention but may be fortunate enough to have a lesion that with enhanced attention can be endoscopically resectioned. While EMR and ESD also contribute a disproportionate burden of complications to endoscopy practice, overall complication rates are still favorable when compared with surgical resection.
Moreover, the most feared short-term complication of EMR and ESD, perforation, has the added benefit of a “controlled failure” to colectomy. Advanced endoscopic approaches already require a prepared colon, and patients are given strict return instructions. Hence, the yearly handful of postprocedural perforations that I get called upon to assist with typically tolerate a routine surgical exploration, repair or resection, and recover at rates equal to or better than elective colon resections. For these reasons, lesions that can be endoscopically removed within appropriate risk tolerances, can and should be considered for EMR or ESD at time of diagnosis.
There are two clinical scenarios where this consideration for up-front EMR or ESD requires further caution. First, any rectal lesion considered for advanced endoscopic techniques really needs to be done in multidisciplinary conference with a colorectal surgeon. In the modern era of colorectal surgery, surgeons now have numerous approaches to reach the rectum that bridge the gap between traditional endoscopy and transabdominal resection. For many rectal lesions, transanal laparoscopic and robotic approaches offer the opportunity for local excision. The most commonly practiced approach, transanal minimally invasive microsurgery (TAMIS), provides many of the benefits of endoscopy (eg, same-day discharge, no activity restrictions, limited periprocedural physiologic stress, low complication rates) while providing the surgical precision, repair strategies, and specimen orientation of conventional surgery. Anecdotally, the time it takes to do a high-quality TAMIS excision in the rectum can be substantially less than that required for a comparable ESD.
For rectal lesions in particular, specimen quality is paramount for oncologic prognosis. Regardless of any intrinsic favorable histopathology or deft hand of the endoscopist, a TAMIS approach will typically provide for a deeper partial thickness or even full thickness excision. More times each year than I would like, I find myself at a multidisciplinary tumor board discussing an endoscopically removed rectal lesion done in a piecemeal fashion or insufficient deep ESD where appropriate risk stratification is impossible and we end up offering patients a likely overly aggressive proctectomy or a potentially oncologically unsound re-excision. Consideration of EMR/ESD vs TAMIS up front would allow better sorting of which technique is most suited to which lesion and avoid these diagnostic dilemmas that only seem to be more common as EMR and ESD practices proliferate.
For a different set of reasons, an advanced cecal adenoma may also be more suited to upfront surgical considerations. Right colon lesions can be more challenging for surveillance for a host of reasons. Procedurally, right colon lesions are undeniably more difficult. The thin-walled cecum can be unforgiving for repeated polypectomies. Despite it being an uncomfortable subject for colonoscopists, the evidence suggests that getting to the cecum is not consistent or 100% expected. Finally, patients can be unwilling to undergo serial bowel preparation and endoscopic examination. In contrast, a laparoscopic right colectomy avoids these issues while also attributing little additional risk. Laparoscopic right-colon operations have overall complication rates of less than 10% and major complications of less than 1%. Hospital stays for laparoscopic right colectomy are typically 3 days or less. Finally, surgery reduces both the frequency of surveillance, and a shortened colon makes surveillance easier.
Advanced polypectomy techniques broaden our ability to address even difficult lesions under the ideally aligned degree of invasive procedure. However, like any procedure, these techniques have their own advantages and limitations. There will always be a minority of premalignant colon lesions that are best suited to surgery-first approaches to treatment. In my practice, maintaining open lines of communication and regular interaction with my endoscopy colleagues naturally leads to polyps being addressed in their most suitable fashion.
Dr. Leeds is assistant professor of surgery at the Yale School of Medicine and a staff surgeon at the VA Connecticut Healthcare System. He declares no conflicts of interest.
Dear colleagues,
We now have the ability to remove almost any large colon polyp endoscopically using a variety of techniques — from the widely used endoscopic mucosal resection to the increasingly prevalent endoscopic submucosal dissection. Yet, in this new era,
In this issue of Perspectives, Dr. Jeffrey Mosko and Dr. Moamen Gabr discuss the importance of careful polyp selection and argue that almost all polyps can be safely removed endoscopically, with low recurrence rates. In contrast, Dr. Ira Leeds from colorectal surgery offers a counterpoint, urging caution when managing polyps in the cecum and rectum while highlighting the role of minimally invasive surgical approaches. We hope these discussions provide valuable insights to support your approach to managing large colorectal polyps, especially in an era of increasing colon cancer screening.
We also welcome your thoughts on this topic — join the conversation on X at @AGA_GIHN.
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, and chief of endoscopy at West Haven VA Medical Center, both in Connecticut. He is an associate editor for GI & Hepatology News.
Advantages of Endoscopic Resection for Large Colon Polyps
BY MOAMEN GABR, MD, MSC, AND JEFFREY D. MOSKO MD, MSC
General Advantages
Endoscopy has revolutionized the management of large colorectal polyps, offering a minimally invasive alternative to surgical resection. The dawn of endoscopic resection in the late 20th century, particularly the evolution of endoscopic mucosal resection (EMR) in Japan, marked a paradigm shift in the treatment of colonic lesions by enabling the removal of lesions that would otherwise necessitate surgery.
Endoscopic resection of colorectal polyps is generally performed in an outpatient setting, allowing patients to recover at home the same day. This not only minimizes disruption to daily life but also significantly enhances patient satisfaction.
Most procedures are performed under moderate or deep sedation eliminating the need for general anesthesia. This represents a critical benefit, particularly for older or medically frail patients who are at higher risk of anesthesia-related complications.
From an economic perspective, endoscopic resection reduces healthcare costs by eliminating prolonged hospital stays and complex perioperative care. Additionally, preserving the colon’s structure and function avoids long-term consequences such as altered bowel habits or ostomy dependence, common with surgical interventions.
The advantages of endoscopic intervention are clear: safety, cost-effectiveness, organ preservation, and convenience for patients.
Lesion Selection
The superiority of endoscopic resection relies on selecting lesions appropriately, specifically those with a low risk of lymph node metastases. This meticulous process should include assessing a lesion’s size, location, morphology, granularity, microvascular and surface pit pattern using a combination of high-definition white light endoscopy, virtual chromoendoscopy and image magnification (when available).
Gross morphologic assessment utilizes the Paris and LST classifications. Combining the Paris classification, lesion granularity and location is both straightforward and revealing. Ulcerated/excavated lesions (0-III) are concerning for deep invasion. Depressed (0-IIc) morphologies are strongly associated with T1 CRC. Nodular lesions (0-Is or IIa + Is) have a higher risk of T1 colorectal cancer (CRC), compared with flat lesions (0-IIa or 0-IIb). Non-granular lesions (0-Is and 0-IIa + Is) have a higher risk of covert cancer. Finally, the rectosigmoid location is associated with an increased risk of T1 CRC (vs. proximal locations).
Endoscopic surface pattern assessment increases one’s diagnostic accuracy. There are three primary endoscopic surface pattern classifications: NBI International Colorectal Endoscopic (NICE), Japanese NBI expert team (JNET), and Kudo pit pattern classifications. Colonic lesions that have a NICE Type 3, JNET 3, or Kudo type Vn pattern should be referred promptly for surgical resection. Lesions with a JNET 2B or Kudo type VI carry a higher risk of superficial T1 CRC but can still be removed endoscopically (see below) in expert centers. All other lesions should undergo endoscopic resection.
Endoscopic Resection Techniques
Endoscopic resection of large colorectal polyps encompasses two primary techniques: EMR and endoscopic submucosal dissection (ESD), each tailored to specific lesion characteristics and operator expertise.
EMR, the technique of choice for the vast majority of lesions, relies on injecting a submucosal cushion to lift the lesion before excision. Recent advances, including enhanced snare designs and underwater EMR, have improved en-bloc resection rates, significantly reducing recurrence and enhancing the efficacy of this technique.
ESD offers unparalleled precision for en-bloc resection of complex lesions, particularly those with fibrosis or high-risk features. Cutting-edge innovations, such as traction devices, have streamlined the procedure, addressing the traditional challenges of ESD. Despite being more time intensive, ESD minimizes recurrence and provides complete histopathological evaluation, critical for the management of malignant or pre-malignant lesions.
For non-lifting polyps, newer techniques such as endoscopic full-thickness resection (eFTR), using tools like the Full-Thickness Resection Device (FTRD), enable resection of up to 2-3 cm of the colonic or rectal wall. This ensures complete removal of any lesion and its underlying tissue, effectively preventing recurrence.
These advancements demonstrate how endoscopy can tackle even the most challenging colorectal polyps, reinforcing its position as the preferred treatment modality.
Perceived Limitations
With ongoing refinement over the last 2 decades, many of the perceived limitations (below) of endoscopic resection have now been overcome.
- Difficult locations/access: Historically lesions at the anorectal junction, ileocecal valve, appendiceal orifice and anastomoses were preferentially sent for surgery. In spite of unique technical challenges at each of these locations, there is now compelling data supporting EMR for these scenarios. We now also have techniques aimed at enabling the resection of lesions with poor access including patient repositioning, distal attachments, variable endoscope diameter/flexibility, traction and overtube devices.
- Recurrence: In the past, recurrence after endoscopic resection of lesions > 20 mm has been reported to be as high as 20%. With our current systematic approach to complete resection, meticulous examination of the post-resection defect for residual polyp tissue, adjunctive techniques to address submucosal fibrosis (hot avulsion, CAST, submucosal release) and thermal ablation to the resection margin (EMR-T), the risk of recurrence for piecemeal resections can be decreased to < 5%. In fact, some groups argue for the en-bloc resection of all large colorectal lesions based on the extremely low (< 1%) recurrence rates and potential for decreased follow-up.
- Post-resection bleeding: Post-resection bleeding is no longer a major limitation of any endoscopic approach because of the combination of improved intra-procedural hemostatic and resection techniques, optimized electrosurgical technology, and enhanced defect closure capabilities and devices (with prophylactic defect closure now supported by randomized control trial level data).
- Perforation: Deep mural injury, once an endoscopists’ worst fear during resection, is no longer a surgical emergency. It can now be predicted, identified (Sydney classification) and successfully managed. In spite of more widespread aggressive resection strategies, the risk of emergency surgery in patients undergoing EMR and even ESD (where the risk of DMI is significantly higher) is extremely low.
Endoscopic resection for large colorectal polyps is effective, available, minimally invasive and organ sparing making it the standard of care for the management of colonic polyps. With ongoing iteration in techniques, more invasive surgical approaches can be avoided in almost all patients with benign and low-risk T1 colorectal cancers.
Dr. Gabr is associate GI division director at the University of Cincinnati, Ohio. Dr. Mosko is based in the division of gastroenterology at St. Michael’s Hospital, Toronto, Ontario, Canada. The authors declare no conflicts of interest.
Blurred Lines: Polyp Needing Surgical versus Endoscopic Excision
BY IRA LEEDS, MD
I am grateful for the invitation to join in discussion with Dr. Gabr and Dr. Mosko on the ever-increasing role of endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD). However, as a surgeon, I do carry at least mild trepidation entering one of the literary “safe spaces” of my gastroenterology colleagues.
With the increasing evidentiary support of EMR approaches and the increasing experience of those performing ESD, these two techniques are quickly becoming the options of choice. As these practices become ubiquitous, it is important to recognize both their advantages and limitations, compared with available surgical options. The decision to proceed with EMR and ESD is essentially a turning point away from early surgical referral for a complex lesion. In this discussion, I intend to highlight when EMR and ESD have a clear advantage to early surgical referral, why I believe that early surgical referral is still superior to advanced endoscopic techniques in the rectum, and why the approach for right-sided lesions should hinge on careful shared decision-making.
Endoscopic approaches nearly always beat surgical approaches when considering short-term risks. Even in the best surgical series, colorectal surgery typically leads to complications in 10%-15% of patients, 1%-5% being serious. Moreover, transabdominal surgical interventions (ie, colectomy) require considerable recovery involving at least a few days in the inpatient setting and over a month of activity restrictions. Finally, there is a minority of chronically unwell patients who cannot tolerate surgical intervention but may be fortunate enough to have a lesion that with enhanced attention can be endoscopically resectioned. While EMR and ESD also contribute a disproportionate burden of complications to endoscopy practice, overall complication rates are still favorable when compared with surgical resection.
Moreover, the most feared short-term complication of EMR and ESD, perforation, has the added benefit of a “controlled failure” to colectomy. Advanced endoscopic approaches already require a prepared colon, and patients are given strict return instructions. Hence, the yearly handful of postprocedural perforations that I get called upon to assist with typically tolerate a routine surgical exploration, repair or resection, and recover at rates equal to or better than elective colon resections. For these reasons, lesions that can be endoscopically removed within appropriate risk tolerances, can and should be considered for EMR or ESD at time of diagnosis.
There are two clinical scenarios where this consideration for up-front EMR or ESD requires further caution. First, any rectal lesion considered for advanced endoscopic techniques really needs to be done in multidisciplinary conference with a colorectal surgeon. In the modern era of colorectal surgery, surgeons now have numerous approaches to reach the rectum that bridge the gap between traditional endoscopy and transabdominal resection. For many rectal lesions, transanal laparoscopic and robotic approaches offer the opportunity for local excision. The most commonly practiced approach, transanal minimally invasive microsurgery (TAMIS), provides many of the benefits of endoscopy (eg, same-day discharge, no activity restrictions, limited periprocedural physiologic stress, low complication rates) while providing the surgical precision, repair strategies, and specimen orientation of conventional surgery. Anecdotally, the time it takes to do a high-quality TAMIS excision in the rectum can be substantially less than that required for a comparable ESD.
For rectal lesions in particular, specimen quality is paramount for oncologic prognosis. Regardless of any intrinsic favorable histopathology or deft hand of the endoscopist, a TAMIS approach will typically provide for a deeper partial thickness or even full thickness excision. More times each year than I would like, I find myself at a multidisciplinary tumor board discussing an endoscopically removed rectal lesion done in a piecemeal fashion or insufficient deep ESD where appropriate risk stratification is impossible and we end up offering patients a likely overly aggressive proctectomy or a potentially oncologically unsound re-excision. Consideration of EMR/ESD vs TAMIS up front would allow better sorting of which technique is most suited to which lesion and avoid these diagnostic dilemmas that only seem to be more common as EMR and ESD practices proliferate.
For a different set of reasons, an advanced cecal adenoma may also be more suited to upfront surgical considerations. Right colon lesions can be more challenging for surveillance for a host of reasons. Procedurally, right colon lesions are undeniably more difficult. The thin-walled cecum can be unforgiving for repeated polypectomies. Despite it being an uncomfortable subject for colonoscopists, the evidence suggests that getting to the cecum is not consistent or 100% expected. Finally, patients can be unwilling to undergo serial bowel preparation and endoscopic examination. In contrast, a laparoscopic right colectomy avoids these issues while also attributing little additional risk. Laparoscopic right-colon operations have overall complication rates of less than 10% and major complications of less than 1%. Hospital stays for laparoscopic right colectomy are typically 3 days or less. Finally, surgery reduces both the frequency of surveillance, and a shortened colon makes surveillance easier.
Advanced polypectomy techniques broaden our ability to address even difficult lesions under the ideally aligned degree of invasive procedure. However, like any procedure, these techniques have their own advantages and limitations. There will always be a minority of premalignant colon lesions that are best suited to surgery-first approaches to treatment. In my practice, maintaining open lines of communication and regular interaction with my endoscopy colleagues naturally leads to polyps being addressed in their most suitable fashion.
Dr. Leeds is assistant professor of surgery at the Yale School of Medicine and a staff surgeon at the VA Connecticut Healthcare System. He declares no conflicts of interest.
Dear colleagues,
We now have the ability to remove almost any large colon polyp endoscopically using a variety of techniques — from the widely used endoscopic mucosal resection to the increasingly prevalent endoscopic submucosal dissection. Yet, in this new era,
In this issue of Perspectives, Dr. Jeffrey Mosko and Dr. Moamen Gabr discuss the importance of careful polyp selection and argue that almost all polyps can be safely removed endoscopically, with low recurrence rates. In contrast, Dr. Ira Leeds from colorectal surgery offers a counterpoint, urging caution when managing polyps in the cecum and rectum while highlighting the role of minimally invasive surgical approaches. We hope these discussions provide valuable insights to support your approach to managing large colorectal polyps, especially in an era of increasing colon cancer screening.
We also welcome your thoughts on this topic — join the conversation on X at @AGA_GIHN.
Gyanprakash A. Ketwaroo, MD, MSc, is associate professor of medicine, Yale University, New Haven, and chief of endoscopy at West Haven VA Medical Center, both in Connecticut. He is an associate editor for GI & Hepatology News.
Advantages of Endoscopic Resection for Large Colon Polyps
BY MOAMEN GABR, MD, MSC, AND JEFFREY D. MOSKO MD, MSC
General Advantages
Endoscopy has revolutionized the management of large colorectal polyps, offering a minimally invasive alternative to surgical resection. The dawn of endoscopic resection in the late 20th century, particularly the evolution of endoscopic mucosal resection (EMR) in Japan, marked a paradigm shift in the treatment of colonic lesions by enabling the removal of lesions that would otherwise necessitate surgery.
Endoscopic resection of colorectal polyps is generally performed in an outpatient setting, allowing patients to recover at home the same day. This not only minimizes disruption to daily life but also significantly enhances patient satisfaction.
Most procedures are performed under moderate or deep sedation eliminating the need for general anesthesia. This represents a critical benefit, particularly for older or medically frail patients who are at higher risk of anesthesia-related complications.
From an economic perspective, endoscopic resection reduces healthcare costs by eliminating prolonged hospital stays and complex perioperative care. Additionally, preserving the colon’s structure and function avoids long-term consequences such as altered bowel habits or ostomy dependence, common with surgical interventions.
The advantages of endoscopic intervention are clear: safety, cost-effectiveness, organ preservation, and convenience for patients.
Lesion Selection
The superiority of endoscopic resection relies on selecting lesions appropriately, specifically those with a low risk of lymph node metastases. This meticulous process should include assessing a lesion’s size, location, morphology, granularity, microvascular and surface pit pattern using a combination of high-definition white light endoscopy, virtual chromoendoscopy and image magnification (when available).
Gross morphologic assessment utilizes the Paris and LST classifications. Combining the Paris classification, lesion granularity and location is both straightforward and revealing. Ulcerated/excavated lesions (0-III) are concerning for deep invasion. Depressed (0-IIc) morphologies are strongly associated with T1 CRC. Nodular lesions (0-Is or IIa + Is) have a higher risk of T1 colorectal cancer (CRC), compared with flat lesions (0-IIa or 0-IIb). Non-granular lesions (0-Is and 0-IIa + Is) have a higher risk of covert cancer. Finally, the rectosigmoid location is associated with an increased risk of T1 CRC (vs. proximal locations).
Endoscopic surface pattern assessment increases one’s diagnostic accuracy. There are three primary endoscopic surface pattern classifications: NBI International Colorectal Endoscopic (NICE), Japanese NBI expert team (JNET), and Kudo pit pattern classifications. Colonic lesions that have a NICE Type 3, JNET 3, or Kudo type Vn pattern should be referred promptly for surgical resection. Lesions with a JNET 2B or Kudo type VI carry a higher risk of superficial T1 CRC but can still be removed endoscopically (see below) in expert centers. All other lesions should undergo endoscopic resection.
Endoscopic Resection Techniques
Endoscopic resection of large colorectal polyps encompasses two primary techniques: EMR and endoscopic submucosal dissection (ESD), each tailored to specific lesion characteristics and operator expertise.
EMR, the technique of choice for the vast majority of lesions, relies on injecting a submucosal cushion to lift the lesion before excision. Recent advances, including enhanced snare designs and underwater EMR, have improved en-bloc resection rates, significantly reducing recurrence and enhancing the efficacy of this technique.
ESD offers unparalleled precision for en-bloc resection of complex lesions, particularly those with fibrosis or high-risk features. Cutting-edge innovations, such as traction devices, have streamlined the procedure, addressing the traditional challenges of ESD. Despite being more time intensive, ESD minimizes recurrence and provides complete histopathological evaluation, critical for the management of malignant or pre-malignant lesions.
For non-lifting polyps, newer techniques such as endoscopic full-thickness resection (eFTR), using tools like the Full-Thickness Resection Device (FTRD), enable resection of up to 2-3 cm of the colonic or rectal wall. This ensures complete removal of any lesion and its underlying tissue, effectively preventing recurrence.
These advancements demonstrate how endoscopy can tackle even the most challenging colorectal polyps, reinforcing its position as the preferred treatment modality.
Perceived Limitations
With ongoing refinement over the last 2 decades, many of the perceived limitations (below) of endoscopic resection have now been overcome.
- Difficult locations/access: Historically lesions at the anorectal junction, ileocecal valve, appendiceal orifice and anastomoses were preferentially sent for surgery. In spite of unique technical challenges at each of these locations, there is now compelling data supporting EMR for these scenarios. We now also have techniques aimed at enabling the resection of lesions with poor access including patient repositioning, distal attachments, variable endoscope diameter/flexibility, traction and overtube devices.
- Recurrence: In the past, recurrence after endoscopic resection of lesions > 20 mm has been reported to be as high as 20%. With our current systematic approach to complete resection, meticulous examination of the post-resection defect for residual polyp tissue, adjunctive techniques to address submucosal fibrosis (hot avulsion, CAST, submucosal release) and thermal ablation to the resection margin (EMR-T), the risk of recurrence for piecemeal resections can be decreased to < 5%. In fact, some groups argue for the en-bloc resection of all large colorectal lesions based on the extremely low (< 1%) recurrence rates and potential for decreased follow-up.
- Post-resection bleeding: Post-resection bleeding is no longer a major limitation of any endoscopic approach because of the combination of improved intra-procedural hemostatic and resection techniques, optimized electrosurgical technology, and enhanced defect closure capabilities and devices (with prophylactic defect closure now supported by randomized control trial level data).
- Perforation: Deep mural injury, once an endoscopists’ worst fear during resection, is no longer a surgical emergency. It can now be predicted, identified (Sydney classification) and successfully managed. In spite of more widespread aggressive resection strategies, the risk of emergency surgery in patients undergoing EMR and even ESD (where the risk of DMI is significantly higher) is extremely low.
Endoscopic resection for large colorectal polyps is effective, available, minimally invasive and organ sparing making it the standard of care for the management of colonic polyps. With ongoing iteration in techniques, more invasive surgical approaches can be avoided in almost all patients with benign and low-risk T1 colorectal cancers.
Dr. Gabr is associate GI division director at the University of Cincinnati, Ohio. Dr. Mosko is based in the division of gastroenterology at St. Michael’s Hospital, Toronto, Ontario, Canada. The authors declare no conflicts of interest.
Blurred Lines: Polyp Needing Surgical versus Endoscopic Excision
BY IRA LEEDS, MD
I am grateful for the invitation to join in discussion with Dr. Gabr and Dr. Mosko on the ever-increasing role of endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD). However, as a surgeon, I do carry at least mild trepidation entering one of the literary “safe spaces” of my gastroenterology colleagues.
With the increasing evidentiary support of EMR approaches and the increasing experience of those performing ESD, these two techniques are quickly becoming the options of choice. As these practices become ubiquitous, it is important to recognize both their advantages and limitations, compared with available surgical options. The decision to proceed with EMR and ESD is essentially a turning point away from early surgical referral for a complex lesion. In this discussion, I intend to highlight when EMR and ESD have a clear advantage to early surgical referral, why I believe that early surgical referral is still superior to advanced endoscopic techniques in the rectum, and why the approach for right-sided lesions should hinge on careful shared decision-making.
Endoscopic approaches nearly always beat surgical approaches when considering short-term risks. Even in the best surgical series, colorectal surgery typically leads to complications in 10%-15% of patients, 1%-5% being serious. Moreover, transabdominal surgical interventions (ie, colectomy) require considerable recovery involving at least a few days in the inpatient setting and over a month of activity restrictions. Finally, there is a minority of chronically unwell patients who cannot tolerate surgical intervention but may be fortunate enough to have a lesion that with enhanced attention can be endoscopically resectioned. While EMR and ESD also contribute a disproportionate burden of complications to endoscopy practice, overall complication rates are still favorable when compared with surgical resection.
Moreover, the most feared short-term complication of EMR and ESD, perforation, has the added benefit of a “controlled failure” to colectomy. Advanced endoscopic approaches already require a prepared colon, and patients are given strict return instructions. Hence, the yearly handful of postprocedural perforations that I get called upon to assist with typically tolerate a routine surgical exploration, repair or resection, and recover at rates equal to or better than elective colon resections. For these reasons, lesions that can be endoscopically removed within appropriate risk tolerances, can and should be considered for EMR or ESD at time of diagnosis.
There are two clinical scenarios where this consideration for up-front EMR or ESD requires further caution. First, any rectal lesion considered for advanced endoscopic techniques really needs to be done in multidisciplinary conference with a colorectal surgeon. In the modern era of colorectal surgery, surgeons now have numerous approaches to reach the rectum that bridge the gap between traditional endoscopy and transabdominal resection. For many rectal lesions, transanal laparoscopic and robotic approaches offer the opportunity for local excision. The most commonly practiced approach, transanal minimally invasive microsurgery (TAMIS), provides many of the benefits of endoscopy (eg, same-day discharge, no activity restrictions, limited periprocedural physiologic stress, low complication rates) while providing the surgical precision, repair strategies, and specimen orientation of conventional surgery. Anecdotally, the time it takes to do a high-quality TAMIS excision in the rectum can be substantially less than that required for a comparable ESD.
For rectal lesions in particular, specimen quality is paramount for oncologic prognosis. Regardless of any intrinsic favorable histopathology or deft hand of the endoscopist, a TAMIS approach will typically provide for a deeper partial thickness or even full thickness excision. More times each year than I would like, I find myself at a multidisciplinary tumor board discussing an endoscopically removed rectal lesion done in a piecemeal fashion or insufficient deep ESD where appropriate risk stratification is impossible and we end up offering patients a likely overly aggressive proctectomy or a potentially oncologically unsound re-excision. Consideration of EMR/ESD vs TAMIS up front would allow better sorting of which technique is most suited to which lesion and avoid these diagnostic dilemmas that only seem to be more common as EMR and ESD practices proliferate.
For a different set of reasons, an advanced cecal adenoma may also be more suited to upfront surgical considerations. Right colon lesions can be more challenging for surveillance for a host of reasons. Procedurally, right colon lesions are undeniably more difficult. The thin-walled cecum can be unforgiving for repeated polypectomies. Despite it being an uncomfortable subject for colonoscopists, the evidence suggests that getting to the cecum is not consistent or 100% expected. Finally, patients can be unwilling to undergo serial bowel preparation and endoscopic examination. In contrast, a laparoscopic right colectomy avoids these issues while also attributing little additional risk. Laparoscopic right-colon operations have overall complication rates of less than 10% and major complications of less than 1%. Hospital stays for laparoscopic right colectomy are typically 3 days or less. Finally, surgery reduces both the frequency of surveillance, and a shortened colon makes surveillance easier.
Advanced polypectomy techniques broaden our ability to address even difficult lesions under the ideally aligned degree of invasive procedure. However, like any procedure, these techniques have their own advantages and limitations. There will always be a minority of premalignant colon lesions that are best suited to surgery-first approaches to treatment. In my practice, maintaining open lines of communication and regular interaction with my endoscopy colleagues naturally leads to polyps being addressed in their most suitable fashion.
Dr. Leeds is assistant professor of surgery at the Yale School of Medicine and a staff surgeon at the VA Connecticut Healthcare System. He declares no conflicts of interest.









