User login
Twin study highlights environmental factors that may aggravate acne
A survey conducted at the world’s largest twin celebration provides more evidence that twins share a genetic propensity toward acne, and provides information about several aggravating factors.
The study “further supports that there may be a genetic phenotypic link, though social and environmental factors may also have an influence in the disease process,” the authors wrote.
Previous twin research has linked genetic factors to 80% of acne variance, with environmental factors, such as stress and low intake of produce, believed to account for the rest of the risk (J Invest Dermat. 2002;119[6]:1317-22). For the new study, researchers surveyed twins at the 2016 Twins Day Festival in Twinsburg, Ohio. Thousand of twins – and triplets and quadruplets – from around the world attend the annual event.
After incomplete surveys were discarded, the survey population included 202 identical twins (101 pairs) and 53 fraternal twins or triplets. (A set of triplets was included in addition to 25 pairs of twins.) The majority of participants were female: 23% of identical twins and 17% of the fraternal twins and triplets were male. The mean age was 29 years among the identical twins and 21 years among fraternal twins.
that suggest “acne is largely attributable to genetics,” the authors observed. Among identical twins, those with acne were more likely to have polycystic ovarian syndrome (P = .045), anxiety (P = .014), and asthma (P = .026).
“Identical twin pairs with acne had a higher BMI [body mass index] and exercised less than those without,” the researchers added. These two associations were statistically significant, both for higher BMI (P = .020) and for less exercise (P = .001). “This suggests that a higher BMI and lack of exercise may contribute [along with genetics of course] to acne development. Thus, regular exercise and lower BMI may keep acne at bay,” they noted.
They also analyzed 56 pairs of identical twins with acne, who reported different severities, and found that the twin with more severe acne was more likely to report that sun exposure (P = .048), cosmetic product use (P = .002), and sugar intake (P = .048) aggravated their acne. Refined carbohydrates, as an aggravating factor, approached statistical significance, they said.
A separate analysis of 45 pairs of female identical twins with different degrees of acne severity produced similar findings. There were no significant difference between acne severity groups in terms of menstruation flare frequency or with oral contraceptive use. The twin with more severe acne, however, “was more likely to report aggravation of acne with sun exposure,” cosmetic use, and sugar intake, all associations which reached statistical significance. They were also more likely to report that refined carbohydrates and intake of fried foods aggravated their acne, associations that approached statistical significance.
“This twin study provides further support for reducing intake of sugar and refined carbohydrates to decrease acne severity in susceptible individuals,” the authors wrote. “For females, reducing intake of fried foods may also help,” they added.
There’s a twist to their results: The finding that those with more severe acne reported worsening symptoms with sun exposure “conflicts with prior research, which has found that acne improves with sun,” the authors wrote, adding that “perhaps the data was confounded by comedogenic sunscreen use.”
No specific study funding was reported. The study authors reported no disclosures.
SOURCE: Suggs A et al. J Drugs Dermatol. 2018 Apr;17(4):380-2.
A survey conducted at the world’s largest twin celebration provides more evidence that twins share a genetic propensity toward acne, and provides information about several aggravating factors.
The study “further supports that there may be a genetic phenotypic link, though social and environmental factors may also have an influence in the disease process,” the authors wrote.
Previous twin research has linked genetic factors to 80% of acne variance, with environmental factors, such as stress and low intake of produce, believed to account for the rest of the risk (J Invest Dermat. 2002;119[6]:1317-22). For the new study, researchers surveyed twins at the 2016 Twins Day Festival in Twinsburg, Ohio. Thousand of twins – and triplets and quadruplets – from around the world attend the annual event.
After incomplete surveys were discarded, the survey population included 202 identical twins (101 pairs) and 53 fraternal twins or triplets. (A set of triplets was included in addition to 25 pairs of twins.) The majority of participants were female: 23% of identical twins and 17% of the fraternal twins and triplets were male. The mean age was 29 years among the identical twins and 21 years among fraternal twins.
that suggest “acne is largely attributable to genetics,” the authors observed. Among identical twins, those with acne were more likely to have polycystic ovarian syndrome (P = .045), anxiety (P = .014), and asthma (P = .026).
“Identical twin pairs with acne had a higher BMI [body mass index] and exercised less than those without,” the researchers added. These two associations were statistically significant, both for higher BMI (P = .020) and for less exercise (P = .001). “This suggests that a higher BMI and lack of exercise may contribute [along with genetics of course] to acne development. Thus, regular exercise and lower BMI may keep acne at bay,” they noted.
They also analyzed 56 pairs of identical twins with acne, who reported different severities, and found that the twin with more severe acne was more likely to report that sun exposure (P = .048), cosmetic product use (P = .002), and sugar intake (P = .048) aggravated their acne. Refined carbohydrates, as an aggravating factor, approached statistical significance, they said.
A separate analysis of 45 pairs of female identical twins with different degrees of acne severity produced similar findings. There were no significant difference between acne severity groups in terms of menstruation flare frequency or with oral contraceptive use. The twin with more severe acne, however, “was more likely to report aggravation of acne with sun exposure,” cosmetic use, and sugar intake, all associations which reached statistical significance. They were also more likely to report that refined carbohydrates and intake of fried foods aggravated their acne, associations that approached statistical significance.
“This twin study provides further support for reducing intake of sugar and refined carbohydrates to decrease acne severity in susceptible individuals,” the authors wrote. “For females, reducing intake of fried foods may also help,” they added.
There’s a twist to their results: The finding that those with more severe acne reported worsening symptoms with sun exposure “conflicts with prior research, which has found that acne improves with sun,” the authors wrote, adding that “perhaps the data was confounded by comedogenic sunscreen use.”
No specific study funding was reported. The study authors reported no disclosures.
SOURCE: Suggs A et al. J Drugs Dermatol. 2018 Apr;17(4):380-2.
A survey conducted at the world’s largest twin celebration provides more evidence that twins share a genetic propensity toward acne, and provides information about several aggravating factors.
The study “further supports that there may be a genetic phenotypic link, though social and environmental factors may also have an influence in the disease process,” the authors wrote.
Previous twin research has linked genetic factors to 80% of acne variance, with environmental factors, such as stress and low intake of produce, believed to account for the rest of the risk (J Invest Dermat. 2002;119[6]:1317-22). For the new study, researchers surveyed twins at the 2016 Twins Day Festival in Twinsburg, Ohio. Thousand of twins – and triplets and quadruplets – from around the world attend the annual event.
After incomplete surveys were discarded, the survey population included 202 identical twins (101 pairs) and 53 fraternal twins or triplets. (A set of triplets was included in addition to 25 pairs of twins.) The majority of participants were female: 23% of identical twins and 17% of the fraternal twins and triplets were male. The mean age was 29 years among the identical twins and 21 years among fraternal twins.
that suggest “acne is largely attributable to genetics,” the authors observed. Among identical twins, those with acne were more likely to have polycystic ovarian syndrome (P = .045), anxiety (P = .014), and asthma (P = .026).
“Identical twin pairs with acne had a higher BMI [body mass index] and exercised less than those without,” the researchers added. These two associations were statistically significant, both for higher BMI (P = .020) and for less exercise (P = .001). “This suggests that a higher BMI and lack of exercise may contribute [along with genetics of course] to acne development. Thus, regular exercise and lower BMI may keep acne at bay,” they noted.
They also analyzed 56 pairs of identical twins with acne, who reported different severities, and found that the twin with more severe acne was more likely to report that sun exposure (P = .048), cosmetic product use (P = .002), and sugar intake (P = .048) aggravated their acne. Refined carbohydrates, as an aggravating factor, approached statistical significance, they said.
A separate analysis of 45 pairs of female identical twins with different degrees of acne severity produced similar findings. There were no significant difference between acne severity groups in terms of menstruation flare frequency or with oral contraceptive use. The twin with more severe acne, however, “was more likely to report aggravation of acne with sun exposure,” cosmetic use, and sugar intake, all associations which reached statistical significance. They were also more likely to report that refined carbohydrates and intake of fried foods aggravated their acne, associations that approached statistical significance.
“This twin study provides further support for reducing intake of sugar and refined carbohydrates to decrease acne severity in susceptible individuals,” the authors wrote. “For females, reducing intake of fried foods may also help,” they added.
There’s a twist to their results: The finding that those with more severe acne reported worsening symptoms with sun exposure “conflicts with prior research, which has found that acne improves with sun,” the authors wrote, adding that “perhaps the data was confounded by comedogenic sunscreen use.”
No specific study funding was reported. The study authors reported no disclosures.
SOURCE: Suggs A et al. J Drugs Dermatol. 2018 Apr;17(4):380-2.
FROM THE JOURNAL OF DRUGS IN DERMATOLOGY
Key clinical point: Moderating environmental factors, such as sugar intake and refined carbohydrates, may help reduce the severity of acne.
Major finding: Sun exposure (P = .048), cosmetic product use (P = .002), and sugar intake (P = .048) were among the factors identified that aggravated acne.
Study details: A survey of 202 identical twins (101 pairs) and 53 fraternal twins or triplets conducted at the annual Twins Day Festival in 2016.
Disclosures: No specific study funding was reported. The study authors reported no disclosures.
Source: Suggs A et al. J Drugs Dermatol. 2018 Apr;17(4):380-2.
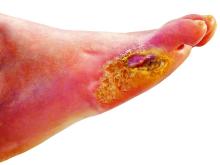
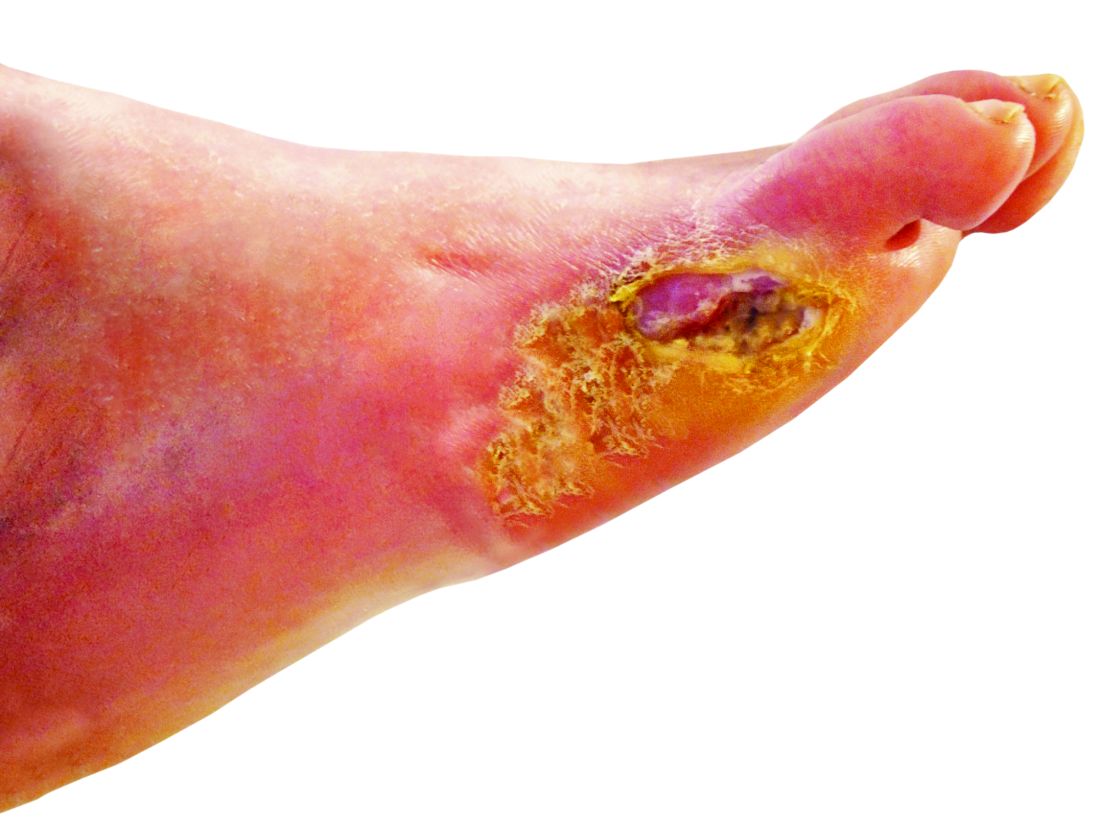
Nitric oxide–generating dressing holds promise for diabetic foot ulcers
Physicians and nurses turn to a wide variety of kinds of dressings to treat patients with diabetic foot ulcers (DFU).
The treatment is still in the research stage, and it’s not clear whether more studies will be conducted. For now, though, “we have a topical agent which specifically treats infection as well as increases perfusion of the ulcer,” study lead author Michael E. Edmonds, MD, a professor of diabetes and endocrinology at King’s College Hospital in London, said in an interview. “The study also showed that the agent not only improved healing but significantly reduced serious adverse events related to the ulcer, which included hospitalizations and amputations.”
The study appeared online April 4 in Wound Repair and Regeneration.
Researchers estimate that DFUs affect as many as 4% of patients with diabetes each year, with about a quarter developing the condition over their lifetimes.
A 2014 U.S. study found that 4%-5% percentage of patients with DFUs underwent lower limb amputations over a 12-month period. The same study also estimated that DFU-related care costs as much as $13 billion a year. (Diabetes Care. 2014 Mar;37[3]:651-8)
“There is no straightforward guideline to choose dressing,” said wound care specialist William H. Tettelbach, MD, the medical director of infection prevention, wound care, and antibiotic stewardship at Landmark Hospital in Salt Lake City, in an interview. Instead, he said, there are just some general tenets: Use an absorbing dressing for a wet ulcer, a moist dressing for a dry ulcer, and an antimicrobial dressing for a bacterial ulcer.
The new multi-center, randomized, controlled phase 2/3 study – funded by the biotech company Edixomed – examined the use of a nitric oxide–generating dressing known as EDX110. The dressing consists of a moist mesh and a second layer that keeps the first layer in place.
“The critical factors that delay the healing of diabetic foot ulcers are ischemia and infection,” Dr. Edmonds said. “Nitric oxide plays a crucial role in maintaining the microvascular supply and infection control in the skin, and its absence in diabetes contributes to poor ulcer healing. EDX110 generates a sustained release of nitric oxide which can treat both infection and ischemia simultaneously.”
The average age of patients in both groups was 59 years, and males made up 82%-87% of the total. Some had more than 1 ulcer.
All patients received standard DFU care for their institution with the exception of members of the treatment group, who were given the EDX110 dressing. Participants were treated for 12 weeks or until their ulcers healed followed by a 12-week follow-up period.
The institutes used a wide variety of dressings including absorbent pad, alginate, antimicrobial, foam, gauze, and other types. About a third were antimicrobial.
In the intent-to-treat population at 12 weeks, the median percentage area reduction of the ulcers was 89% in the treatment group, compared with 47% in the control group (P = .016).
The researchers reported significantly fewer serious adverse events in the treatment group, and none were reported to be linked to the various dressings used.
According to Dr. Edmonds, pricing information for the treatment is unavailable.
Dr. Tettelbach cautioned about the limitations of the study. For one, it doesn’t focus on chronic DFUs that can last well beyond a month and “are more problematic to heal and pose a greater relative risk of infection than acute DFUs.”
He added: “Surrogate end points such as 80% reduction in surface area at 12 weeks are difficult to extrapolate to expected closure. An open chronic ulcer is at risk for complicating infection no matter what size,” he said.
Overall, Dr. Tettelbach said, he doesn’t see the study as a “big deal,” but it’s “a welcomed addition to the wound dressing family that works using a novel mechanism of stimulating angiogenesis and antimicrobial properties.”
The biotech company Edixomed funded the study. The study authors report various disclosures or no disclosures; two disclose links to Edixomed.
SOURCE: Edmonds ME et al. Wound Repair Regen. 2018 April 4. doi: 10.1111/wrr.12630.
Physicians and nurses turn to a wide variety of kinds of dressings to treat patients with diabetic foot ulcers (DFU).
The treatment is still in the research stage, and it’s not clear whether more studies will be conducted. For now, though, “we have a topical agent which specifically treats infection as well as increases perfusion of the ulcer,” study lead author Michael E. Edmonds, MD, a professor of diabetes and endocrinology at King’s College Hospital in London, said in an interview. “The study also showed that the agent not only improved healing but significantly reduced serious adverse events related to the ulcer, which included hospitalizations and amputations.”
The study appeared online April 4 in Wound Repair and Regeneration.
Researchers estimate that DFUs affect as many as 4% of patients with diabetes each year, with about a quarter developing the condition over their lifetimes.
A 2014 U.S. study found that 4%-5% percentage of patients with DFUs underwent lower limb amputations over a 12-month period. The same study also estimated that DFU-related care costs as much as $13 billion a year. (Diabetes Care. 2014 Mar;37[3]:651-8)
“There is no straightforward guideline to choose dressing,” said wound care specialist William H. Tettelbach, MD, the medical director of infection prevention, wound care, and antibiotic stewardship at Landmark Hospital in Salt Lake City, in an interview. Instead, he said, there are just some general tenets: Use an absorbing dressing for a wet ulcer, a moist dressing for a dry ulcer, and an antimicrobial dressing for a bacterial ulcer.
The new multi-center, randomized, controlled phase 2/3 study – funded by the biotech company Edixomed – examined the use of a nitric oxide–generating dressing known as EDX110. The dressing consists of a moist mesh and a second layer that keeps the first layer in place.
“The critical factors that delay the healing of diabetic foot ulcers are ischemia and infection,” Dr. Edmonds said. “Nitric oxide plays a crucial role in maintaining the microvascular supply and infection control in the skin, and its absence in diabetes contributes to poor ulcer healing. EDX110 generates a sustained release of nitric oxide which can treat both infection and ischemia simultaneously.”
The average age of patients in both groups was 59 years, and males made up 82%-87% of the total. Some had more than 1 ulcer.
All patients received standard DFU care for their institution with the exception of members of the treatment group, who were given the EDX110 dressing. Participants were treated for 12 weeks or until their ulcers healed followed by a 12-week follow-up period.
The institutes used a wide variety of dressings including absorbent pad, alginate, antimicrobial, foam, gauze, and other types. About a third were antimicrobial.
In the intent-to-treat population at 12 weeks, the median percentage area reduction of the ulcers was 89% in the treatment group, compared with 47% in the control group (P = .016).
The researchers reported significantly fewer serious adverse events in the treatment group, and none were reported to be linked to the various dressings used.
According to Dr. Edmonds, pricing information for the treatment is unavailable.
Dr. Tettelbach cautioned about the limitations of the study. For one, it doesn’t focus on chronic DFUs that can last well beyond a month and “are more problematic to heal and pose a greater relative risk of infection than acute DFUs.”
He added: “Surrogate end points such as 80% reduction in surface area at 12 weeks are difficult to extrapolate to expected closure. An open chronic ulcer is at risk for complicating infection no matter what size,” he said.
Overall, Dr. Tettelbach said, he doesn’t see the study as a “big deal,” but it’s “a welcomed addition to the wound dressing family that works using a novel mechanism of stimulating angiogenesis and antimicrobial properties.”
The biotech company Edixomed funded the study. The study authors report various disclosures or no disclosures; two disclose links to Edixomed.
SOURCE: Edmonds ME et al. Wound Repair Regen. 2018 April 4. doi: 10.1111/wrr.12630.
Physicians and nurses turn to a wide variety of kinds of dressings to treat patients with diabetic foot ulcers (DFU).
The treatment is still in the research stage, and it’s not clear whether more studies will be conducted. For now, though, “we have a topical agent which specifically treats infection as well as increases perfusion of the ulcer,” study lead author Michael E. Edmonds, MD, a professor of diabetes and endocrinology at King’s College Hospital in London, said in an interview. “The study also showed that the agent not only improved healing but significantly reduced serious adverse events related to the ulcer, which included hospitalizations and amputations.”
The study appeared online April 4 in Wound Repair and Regeneration.
Researchers estimate that DFUs affect as many as 4% of patients with diabetes each year, with about a quarter developing the condition over their lifetimes.
A 2014 U.S. study found that 4%-5% percentage of patients with DFUs underwent lower limb amputations over a 12-month period. The same study also estimated that DFU-related care costs as much as $13 billion a year. (Diabetes Care. 2014 Mar;37[3]:651-8)
“There is no straightforward guideline to choose dressing,” said wound care specialist William H. Tettelbach, MD, the medical director of infection prevention, wound care, and antibiotic stewardship at Landmark Hospital in Salt Lake City, in an interview. Instead, he said, there are just some general tenets: Use an absorbing dressing for a wet ulcer, a moist dressing for a dry ulcer, and an antimicrobial dressing for a bacterial ulcer.
The new multi-center, randomized, controlled phase 2/3 study – funded by the biotech company Edixomed – examined the use of a nitric oxide–generating dressing known as EDX110. The dressing consists of a moist mesh and a second layer that keeps the first layer in place.
“The critical factors that delay the healing of diabetic foot ulcers are ischemia and infection,” Dr. Edmonds said. “Nitric oxide plays a crucial role in maintaining the microvascular supply and infection control in the skin, and its absence in diabetes contributes to poor ulcer healing. EDX110 generates a sustained release of nitric oxide which can treat both infection and ischemia simultaneously.”
The average age of patients in both groups was 59 years, and males made up 82%-87% of the total. Some had more than 1 ulcer.
All patients received standard DFU care for their institution with the exception of members of the treatment group, who were given the EDX110 dressing. Participants were treated for 12 weeks or until their ulcers healed followed by a 12-week follow-up period.
The institutes used a wide variety of dressings including absorbent pad, alginate, antimicrobial, foam, gauze, and other types. About a third were antimicrobial.
In the intent-to-treat population at 12 weeks, the median percentage area reduction of the ulcers was 89% in the treatment group, compared with 47% in the control group (P = .016).
The researchers reported significantly fewer serious adverse events in the treatment group, and none were reported to be linked to the various dressings used.
According to Dr. Edmonds, pricing information for the treatment is unavailable.
Dr. Tettelbach cautioned about the limitations of the study. For one, it doesn’t focus on chronic DFUs that can last well beyond a month and “are more problematic to heal and pose a greater relative risk of infection than acute DFUs.”
He added: “Surrogate end points such as 80% reduction in surface area at 12 weeks are difficult to extrapolate to expected closure. An open chronic ulcer is at risk for complicating infection no matter what size,” he said.
Overall, Dr. Tettelbach said, he doesn’t see the study as a “big deal,” but it’s “a welcomed addition to the wound dressing family that works using a novel mechanism of stimulating angiogenesis and antimicrobial properties.”
The biotech company Edixomed funded the study. The study authors report various disclosures or no disclosures; two disclose links to Edixomed.
SOURCE: Edmonds ME et al. Wound Repair Regen. 2018 April 4. doi: 10.1111/wrr.12630.
FROM WOUND REPAIR AND REGENERATION
Hyaluronic acid filler preferred for infraorbital hollowing
Most patients who responded to surveys reported being satisfied after off-label treatment with Juvéderm Voluma XC hyaluronic acid filler for infraorbital hollowing, a study finds.
Adverse effects were reported in 12% of patients.
The treatment’s “high patient satisfaction profile and a similar safety profile among other soft-tissue fillers make it an excellent adjunct in the plastic surgeon’s armamentarium,” reported Michael B. Hall, MD, and his associates at their private, ambulatory facial plastic and reconstructive surgery practice in Austin, Texas, in JAMA Facial Plastic Surgery.
According to the researchers, the Food and Drug Administration has not approved any soft-tissue fillers for the periorbital complex. At their practice, Dr. Hall and his associates treat infraorbital hollows with Juvéderm Voluma XC, which is approved by the FDA for certain types of cheek augmentation. Other studies have examined Belotero or Restylane as treatments for building volume in the periorbital area, the authors wrote, but research into cosmetic injections of Juvéderm Voluma XC is lacking.
For the new study, the researchers retrospectively analyzed the cases of 101 patients (aged 32-75 years, with average age of 54 years; 89% female; 54% Fitzpatrick skin type II; racial breakdown not reported) who were electively treated with the filler for infraorbital hollowing in 2016 and 2017. The patients received an average 1 mL of the treatment gel.
The patients were photographed and answered surveys, and they were evaluated using the Allergan Infraorbital Hollows Scale. Follow-up time averaged 12 months.
A total of 18 patients (18%) required touch-up within 3 months, and 2 required multiple touch-ups. A total of 12 subjects (12%) had adverse effects (including 3 who had more than one), which included bruising (10%), contour irregularities (2%), edema (3%) and Tyndall effect (1%). Hyaluronidase was required in 3 patients (3%), and 24 patients sought further treatment after 3 months.
The researchers sent two satisfaction surveys to the participants. A total of 41% responded to the FACE-Q Satisfaction With Eyes survey, and 42% responded to the FACE-Q Satisfaction With Decision survey.
Depending on the question, 70%-85% of the respondents to the Satisfaction With Eyes survey said they were “definitely” or “somewhat” satisfied with the treatment outcome.
The highest levels of dissatisfaction came in response to a questions about whether the subjects felt their eyes looked alert (not tired) or youthful. The highest levels of satisfaction were in response to questions about whether the subjects were happy with the shape, attractiveness, and openness of their eyes.
Depending on the question, 73%-85% of the subjects who took the Satisfaction With Decision survey reported that they “definitely” or “somewhat” agree with positive statements about the treatment. While differences were small, they agreed the most with a statement saying the procedure was “worth the time and effort.”
No external funding or remuneration was received. The study authors reported no relevant disclosures.
SOURCE: Hall MB et al. JAMA Facial Plast Surg. 2018 Apr 5. doi:10.1001/jamafacial.2018.0230.
Most patients who responded to surveys reported being satisfied after off-label treatment with Juvéderm Voluma XC hyaluronic acid filler for infraorbital hollowing, a study finds.
Adverse effects were reported in 12% of patients.
The treatment’s “high patient satisfaction profile and a similar safety profile among other soft-tissue fillers make it an excellent adjunct in the plastic surgeon’s armamentarium,” reported Michael B. Hall, MD, and his associates at their private, ambulatory facial plastic and reconstructive surgery practice in Austin, Texas, in JAMA Facial Plastic Surgery.
According to the researchers, the Food and Drug Administration has not approved any soft-tissue fillers for the periorbital complex. At their practice, Dr. Hall and his associates treat infraorbital hollows with Juvéderm Voluma XC, which is approved by the FDA for certain types of cheek augmentation. Other studies have examined Belotero or Restylane as treatments for building volume in the periorbital area, the authors wrote, but research into cosmetic injections of Juvéderm Voluma XC is lacking.
For the new study, the researchers retrospectively analyzed the cases of 101 patients (aged 32-75 years, with average age of 54 years; 89% female; 54% Fitzpatrick skin type II; racial breakdown not reported) who were electively treated with the filler for infraorbital hollowing in 2016 and 2017. The patients received an average 1 mL of the treatment gel.
The patients were photographed and answered surveys, and they were evaluated using the Allergan Infraorbital Hollows Scale. Follow-up time averaged 12 months.
A total of 18 patients (18%) required touch-up within 3 months, and 2 required multiple touch-ups. A total of 12 subjects (12%) had adverse effects (including 3 who had more than one), which included bruising (10%), contour irregularities (2%), edema (3%) and Tyndall effect (1%). Hyaluronidase was required in 3 patients (3%), and 24 patients sought further treatment after 3 months.
The researchers sent two satisfaction surveys to the participants. A total of 41% responded to the FACE-Q Satisfaction With Eyes survey, and 42% responded to the FACE-Q Satisfaction With Decision survey.
Depending on the question, 70%-85% of the respondents to the Satisfaction With Eyes survey said they were “definitely” or “somewhat” satisfied with the treatment outcome.
The highest levels of dissatisfaction came in response to a questions about whether the subjects felt their eyes looked alert (not tired) or youthful. The highest levels of satisfaction were in response to questions about whether the subjects were happy with the shape, attractiveness, and openness of their eyes.
Depending on the question, 73%-85% of the subjects who took the Satisfaction With Decision survey reported that they “definitely” or “somewhat” agree with positive statements about the treatment. While differences were small, they agreed the most with a statement saying the procedure was “worth the time and effort.”
No external funding or remuneration was received. The study authors reported no relevant disclosures.
SOURCE: Hall MB et al. JAMA Facial Plast Surg. 2018 Apr 5. doi:10.1001/jamafacial.2018.0230.
Most patients who responded to surveys reported being satisfied after off-label treatment with Juvéderm Voluma XC hyaluronic acid filler for infraorbital hollowing, a study finds.
Adverse effects were reported in 12% of patients.
The treatment’s “high patient satisfaction profile and a similar safety profile among other soft-tissue fillers make it an excellent adjunct in the plastic surgeon’s armamentarium,” reported Michael B. Hall, MD, and his associates at their private, ambulatory facial plastic and reconstructive surgery practice in Austin, Texas, in JAMA Facial Plastic Surgery.
According to the researchers, the Food and Drug Administration has not approved any soft-tissue fillers for the periorbital complex. At their practice, Dr. Hall and his associates treat infraorbital hollows with Juvéderm Voluma XC, which is approved by the FDA for certain types of cheek augmentation. Other studies have examined Belotero or Restylane as treatments for building volume in the periorbital area, the authors wrote, but research into cosmetic injections of Juvéderm Voluma XC is lacking.
For the new study, the researchers retrospectively analyzed the cases of 101 patients (aged 32-75 years, with average age of 54 years; 89% female; 54% Fitzpatrick skin type II; racial breakdown not reported) who were electively treated with the filler for infraorbital hollowing in 2016 and 2017. The patients received an average 1 mL of the treatment gel.
The patients were photographed and answered surveys, and they were evaluated using the Allergan Infraorbital Hollows Scale. Follow-up time averaged 12 months.
A total of 18 patients (18%) required touch-up within 3 months, and 2 required multiple touch-ups. A total of 12 subjects (12%) had adverse effects (including 3 who had more than one), which included bruising (10%), contour irregularities (2%), edema (3%) and Tyndall effect (1%). Hyaluronidase was required in 3 patients (3%), and 24 patients sought further treatment after 3 months.
The researchers sent two satisfaction surveys to the participants. A total of 41% responded to the FACE-Q Satisfaction With Eyes survey, and 42% responded to the FACE-Q Satisfaction With Decision survey.
Depending on the question, 70%-85% of the respondents to the Satisfaction With Eyes survey said they were “definitely” or “somewhat” satisfied with the treatment outcome.
The highest levels of dissatisfaction came in response to a questions about whether the subjects felt their eyes looked alert (not tired) or youthful. The highest levels of satisfaction were in response to questions about whether the subjects were happy with the shape, attractiveness, and openness of their eyes.
Depending on the question, 73%-85% of the subjects who took the Satisfaction With Decision survey reported that they “definitely” or “somewhat” agree with positive statements about the treatment. While differences were small, they agreed the most with a statement saying the procedure was “worth the time and effort.”
No external funding or remuneration was received. The study authors reported no relevant disclosures.
SOURCE: Hall MB et al. JAMA Facial Plast Surg. 2018 Apr 5. doi:10.1001/jamafacial.2018.0230.
FROM JAMA FACIAL PLASTIC SURGERY
Key clinical point:
Major finding: Adverse effects occurred at a rate of 12%, and most who responded to surveys reported satisfaction postprocedure (70%-85%).
Study details: A retrospective observational study of 101 patients.
Disclosures: No external funding or remuneration was received. The study authors reported no relevant disclosures.
Source: Hall MB et al. JAMA Facial Plast Surg. 2018 Apr 5. doi:10.1001/jamafacial.2018.0230.
Multidisciplinary teams improve diagnoses in ILD
New research provides strong statistical support for the use of dynamic multidisciplinary discussion in the diagnosis of patients who may have interstitial lung diseases (ILD).
and it changed the diagnosis in 41% of the other cases.
The American Thoracic Society, European Respiratory Society, Japanese Respiratory Society, and Latin American Thoracic Association adopted joint guidelines for the treatment of idiopathic pulmonary fibrosis in 2015, and the ATS and ERS updated guidelines for the classification and terminology for idiopathic interstitial pneumonias in 2013. The Lancet Respiratory Medicine published what some consider to be a landmark evaluation of multidisciplinary team agreement on diagnosis of interstitial lung disease following the adoption of these guidelines (Walsh SLF et al. 2016;4[7]:557-65). This study showed that in idiopathic pulmonary fibrosis, multidisciplinary team meetings “have a higher level of agreement on diagnoses, assign diagnoses with higher confidence more frequently, and provide diagnoses that have nonsignificant greater prognostic separation than do clinicians or radiologists in most cases,” the researchers wrote.
In the new study, MDD failed to produce a diagnosis or suggestions about a way forward in only 3.5% of patients, according to the study, which appeared March 30 in CHEST®.
According to Dr. Antin-Ozerkis, accurate diagnosis of ILD is crucial to treatment, but it can be challenging to achieve. The MDD approach has been recommended since 2002 by the ATS and ERS, she said.
The study authors, led by Laurens J. De Sadeleer, MD, of Belgium’s University Hospitals Leuven, define the MDD approach as one “in which expert ILD clinicians, radiologists, and pathologists integrate all available clinical data, laboratory results, high-resolution computed tomography [HRCT] findings, and lung biopsy [when performed].”
For the study, the researchers tracked pre-MDD and MDD diagnoses of 938 consecutive patients with possible ILD who were discussed during 2005-2015. Of these patients, referring physicians made preliminary diagnoses in 49% of cases; in the rest, physicians either failed to develop a diagnosis or offered multiple possible diagnoses.
MDD teams produced a change in diagnosis in 191 – 42% – of patients with a pre-MDD diagnosis. Another condition was diagnosed in 118 of these patients, and the MDD teams declined to classify the other 73 patients pending further investigation.
The MDD teams also were able to produce diagnoses in 80% of cases when referring physicians could not come up with diagnoses.
“Discrepancy between pre-MDD diagnosis before work-up and discussion was remarkable,” the study authors wrote, estimating that MDD added value for 70% of referred patients.
“We believe MDD should be a common practice in the diagnosis of every patient with suspected ILD,” the researchers said.
The study doesn’t examine the challenges of putting MDD into practice, but Dr. Antin-Ozerkis provided some perspective. “It may be difficult for physicians to take the time from a busy practice to meet with a multidisciplinary team. It can require resources to gather the data necessary to comprehensively assess each patient case. Additionally, maintaining staff with experienced pulmonologists, radiologists and pathologists may be costly.”
She added that “there are various ways in which MDD may occur,” and that the pros and cons of different methods have not been well studied. “This practice will likely evolve with the development of new biomarkers and other diagnostic strategies in IPF [idiopathic pulmonary fibrosis].”
Still, she said, “this joint undertaking is clearly vital in helping to guide clinical practice, including therapeutic decisions and discussion of prognosis. For now, any discussion between clinician, radiologist, and pathologist is of benefit.”
Research Foundation-Flanders and University Hospitals Leuven funded the study. Some study authors reported various disclosures. Dr. Antin-Ozerkis disclosed serving as an investigator on several clinical trials for IPF and other ILDs by Boehringer, Promedior, Fibrogen and Roche. She noted that payments go directly to the university with no direct payments to the investigator.
SOURCE: De Sadeleer LJ et al. Chest. 2018 Mar 30. doi: 10.1016/j.chest.2018.03.026.
MDD strategy is crucial for accurate ILD diagnoses
The field of interstitial lung diseases (ILD) is challenging, with more than 200 disorders as possible diagnoses for patients who present to clinicians with similar symptoms and chest x-ray findings. The multidisciplinary discussion (MDD) strategy is very important for attaining an accurate ILD diagnosis.
We have had routine, formal, multidisciplinary discussions at our center since 2008. My guesstimate is that at least a third of patients referred as having idiopathic pulmonary fibrosis or another form of ILD by pulmonologists had been given the wrong diagnosis. Frequently, this was because of incorrect impressions provided by local radiologists and/or pathologists along with the clinician’s own limited knowledge of ILD.
In my experience, some patients described their pulmonologists as becoming irate with them when they asked for a second opinion, and I have had to try to avoid confrontations with referring physicians when trying to explain why the referral diagnosis was inaccurate.
Challenges to instituting the multidisciplinary discussion approach include coverage by health plans for a second-opinion evaluation, the willingness of physicians (for example, pulmonologists) outside of academic referral centers to refer patients to a center capable of adequately conducting an MDD, and patients’ desire to undergo an evaluation at centers of excellence where an MDD can be performed.
One must have also adequate resources to perform a proper MDD. But even in centers that refer patients, pulmonologists should confer with their colleague radiologists – and pathologists when appropriate – to try to make the most accurate diagnosis. And they should continue to question their diagnosis at follow-up appointments, as new symptoms and findings may arise or additional crucial information can become available over time that can point to an alternative diagnosis.
Kenneth C. Meyer, MD, MS, served as medical director of the lung transplant program and head of ILD at the University of Wisconsin–Madison. He reported no relevant disclosures.
Second MDD may be helpful for CTD-related ILD
Accumulating evidence suggests that multidisciplinary committees play a central role in improving the diagnostic accuracy of complex medical conditions. Interstitial lung disease (ILD) encompasses a number of clinical entities and no single diagnostic test alone can discriminate among the various causes of ILD. Instead, these diagnoses are based on a constellation of signs and symptoms, and radiographic, pathologic, and laboratory studies.
However, unanswered questions remain. First, it is unclear whether a single MDD is sufficient. The present study found that 20% of cases were unclassifiable after the MDD. A second MDD may be helpful, especially in patients with ILDs related to connective tissue disease (CTD). The rheumatic diseases most commonly associated with ILD (for example, systemic sclerosis, rheumatoid arthritis, myositis) often evolve at different rates, and not all of the signs and symptoms of these conditions may be present or apparent at the time of the ILD presentation. A second MDD discussion may be particularly helpful in patients presenting with a specific CTD-related autoantibody in the absence of clinical signs and symptoms of a CTD. Another unanswered question is whether MDDs actually improve clinically meaningful outcomes for patients, such as survival and quality of life. At our CTD-ILD Program at the University of California, Los Angeles, we have found that our MDD has augmented patient satisfaction with their care, and it has also improved our ability to identify patients who are eligible for specific clinical studies. Future research is needed to determine to assess the impact of MDD on a variety of patient-centered and practice/research-focused outcomes.
Elizabeth Volkmann, MD, is founder and codirector of the CTD-ILD Program at the University of California, Los Angeles. She disclosed serving as a consultant or as a member of an advisory board for Boehringer Ingelheim and Astellas Pharma. She has received grants from Boehringer Ingelheim, Merck Serono, and the Rheumatology Research Foundation.
MDD strategy is crucial for accurate ILD diagnoses
The field of interstitial lung diseases (ILD) is challenging, with more than 200 disorders as possible diagnoses for patients who present to clinicians with similar symptoms and chest x-ray findings. The multidisciplinary discussion (MDD) strategy is very important for attaining an accurate ILD diagnosis.
We have had routine, formal, multidisciplinary discussions at our center since 2008. My guesstimate is that at least a third of patients referred as having idiopathic pulmonary fibrosis or another form of ILD by pulmonologists had been given the wrong diagnosis. Frequently, this was because of incorrect impressions provided by local radiologists and/or pathologists along with the clinician’s own limited knowledge of ILD.
In my experience, some patients described their pulmonologists as becoming irate with them when they asked for a second opinion, and I have had to try to avoid confrontations with referring physicians when trying to explain why the referral diagnosis was inaccurate.
Challenges to instituting the multidisciplinary discussion approach include coverage by health plans for a second-opinion evaluation, the willingness of physicians (for example, pulmonologists) outside of academic referral centers to refer patients to a center capable of adequately conducting an MDD, and patients’ desire to undergo an evaluation at centers of excellence where an MDD can be performed.
One must have also adequate resources to perform a proper MDD. But even in centers that refer patients, pulmonologists should confer with their colleague radiologists – and pathologists when appropriate – to try to make the most accurate diagnosis. And they should continue to question their diagnosis at follow-up appointments, as new symptoms and findings may arise or additional crucial information can become available over time that can point to an alternative diagnosis.
Kenneth C. Meyer, MD, MS, served as medical director of the lung transplant program and head of ILD at the University of Wisconsin–Madison. He reported no relevant disclosures.
Second MDD may be helpful for CTD-related ILD
Accumulating evidence suggests that multidisciplinary committees play a central role in improving the diagnostic accuracy of complex medical conditions. Interstitial lung disease (ILD) encompasses a number of clinical entities and no single diagnostic test alone can discriminate among the various causes of ILD. Instead, these diagnoses are based on a constellation of signs and symptoms, and radiographic, pathologic, and laboratory studies.
However, unanswered questions remain. First, it is unclear whether a single MDD is sufficient. The present study found that 20% of cases were unclassifiable after the MDD. A second MDD may be helpful, especially in patients with ILDs related to connective tissue disease (CTD). The rheumatic diseases most commonly associated with ILD (for example, systemic sclerosis, rheumatoid arthritis, myositis) often evolve at different rates, and not all of the signs and symptoms of these conditions may be present or apparent at the time of the ILD presentation. A second MDD discussion may be particularly helpful in patients presenting with a specific CTD-related autoantibody in the absence of clinical signs and symptoms of a CTD. Another unanswered question is whether MDDs actually improve clinically meaningful outcomes for patients, such as survival and quality of life. At our CTD-ILD Program at the University of California, Los Angeles, we have found that our MDD has augmented patient satisfaction with their care, and it has also improved our ability to identify patients who are eligible for specific clinical studies. Future research is needed to determine to assess the impact of MDD on a variety of patient-centered and practice/research-focused outcomes.
Elizabeth Volkmann, MD, is founder and codirector of the CTD-ILD Program at the University of California, Los Angeles. She disclosed serving as a consultant or as a member of an advisory board for Boehringer Ingelheim and Astellas Pharma. She has received grants from Boehringer Ingelheim, Merck Serono, and the Rheumatology Research Foundation.
MDD strategy is crucial for accurate ILD diagnoses
The field of interstitial lung diseases (ILD) is challenging, with more than 200 disorders as possible diagnoses for patients who present to clinicians with similar symptoms and chest x-ray findings. The multidisciplinary discussion (MDD) strategy is very important for attaining an accurate ILD diagnosis.
We have had routine, formal, multidisciplinary discussions at our center since 2008. My guesstimate is that at least a third of patients referred as having idiopathic pulmonary fibrosis or another form of ILD by pulmonologists had been given the wrong diagnosis. Frequently, this was because of incorrect impressions provided by local radiologists and/or pathologists along with the clinician’s own limited knowledge of ILD.
In my experience, some patients described their pulmonologists as becoming irate with them when they asked for a second opinion, and I have had to try to avoid confrontations with referring physicians when trying to explain why the referral diagnosis was inaccurate.
Challenges to instituting the multidisciplinary discussion approach include coverage by health plans for a second-opinion evaluation, the willingness of physicians (for example, pulmonologists) outside of academic referral centers to refer patients to a center capable of adequately conducting an MDD, and patients’ desire to undergo an evaluation at centers of excellence where an MDD can be performed.
One must have also adequate resources to perform a proper MDD. But even in centers that refer patients, pulmonologists should confer with their colleague radiologists – and pathologists when appropriate – to try to make the most accurate diagnosis. And they should continue to question their diagnosis at follow-up appointments, as new symptoms and findings may arise or additional crucial information can become available over time that can point to an alternative diagnosis.
Kenneth C. Meyer, MD, MS, served as medical director of the lung transplant program and head of ILD at the University of Wisconsin–Madison. He reported no relevant disclosures.
Second MDD may be helpful for CTD-related ILD
Accumulating evidence suggests that multidisciplinary committees play a central role in improving the diagnostic accuracy of complex medical conditions. Interstitial lung disease (ILD) encompasses a number of clinical entities and no single diagnostic test alone can discriminate among the various causes of ILD. Instead, these diagnoses are based on a constellation of signs and symptoms, and radiographic, pathologic, and laboratory studies.
However, unanswered questions remain. First, it is unclear whether a single MDD is sufficient. The present study found that 20% of cases were unclassifiable after the MDD. A second MDD may be helpful, especially in patients with ILDs related to connective tissue disease (CTD). The rheumatic diseases most commonly associated with ILD (for example, systemic sclerosis, rheumatoid arthritis, myositis) often evolve at different rates, and not all of the signs and symptoms of these conditions may be present or apparent at the time of the ILD presentation. A second MDD discussion may be particularly helpful in patients presenting with a specific CTD-related autoantibody in the absence of clinical signs and symptoms of a CTD. Another unanswered question is whether MDDs actually improve clinically meaningful outcomes for patients, such as survival and quality of life. At our CTD-ILD Program at the University of California, Los Angeles, we have found that our MDD has augmented patient satisfaction with their care, and it has also improved our ability to identify patients who are eligible for specific clinical studies. Future research is needed to determine to assess the impact of MDD on a variety of patient-centered and practice/research-focused outcomes.
Elizabeth Volkmann, MD, is founder and codirector of the CTD-ILD Program at the University of California, Los Angeles. She disclosed serving as a consultant or as a member of an advisory board for Boehringer Ingelheim and Astellas Pharma. She has received grants from Boehringer Ingelheim, Merck Serono, and the Rheumatology Research Foundation.
New research provides strong statistical support for the use of dynamic multidisciplinary discussion in the diagnosis of patients who may have interstitial lung diseases (ILD).
and it changed the diagnosis in 41% of the other cases.
The American Thoracic Society, European Respiratory Society, Japanese Respiratory Society, and Latin American Thoracic Association adopted joint guidelines for the treatment of idiopathic pulmonary fibrosis in 2015, and the ATS and ERS updated guidelines for the classification and terminology for idiopathic interstitial pneumonias in 2013. The Lancet Respiratory Medicine published what some consider to be a landmark evaluation of multidisciplinary team agreement on diagnosis of interstitial lung disease following the adoption of these guidelines (Walsh SLF et al. 2016;4[7]:557-65). This study showed that in idiopathic pulmonary fibrosis, multidisciplinary team meetings “have a higher level of agreement on diagnoses, assign diagnoses with higher confidence more frequently, and provide diagnoses that have nonsignificant greater prognostic separation than do clinicians or radiologists in most cases,” the researchers wrote.
In the new study, MDD failed to produce a diagnosis or suggestions about a way forward in only 3.5% of patients, according to the study, which appeared March 30 in CHEST®.
According to Dr. Antin-Ozerkis, accurate diagnosis of ILD is crucial to treatment, but it can be challenging to achieve. The MDD approach has been recommended since 2002 by the ATS and ERS, she said.
The study authors, led by Laurens J. De Sadeleer, MD, of Belgium’s University Hospitals Leuven, define the MDD approach as one “in which expert ILD clinicians, radiologists, and pathologists integrate all available clinical data, laboratory results, high-resolution computed tomography [HRCT] findings, and lung biopsy [when performed].”
For the study, the researchers tracked pre-MDD and MDD diagnoses of 938 consecutive patients with possible ILD who were discussed during 2005-2015. Of these patients, referring physicians made preliminary diagnoses in 49% of cases; in the rest, physicians either failed to develop a diagnosis or offered multiple possible diagnoses.
MDD teams produced a change in diagnosis in 191 – 42% – of patients with a pre-MDD diagnosis. Another condition was diagnosed in 118 of these patients, and the MDD teams declined to classify the other 73 patients pending further investigation.
The MDD teams also were able to produce diagnoses in 80% of cases when referring physicians could not come up with diagnoses.
“Discrepancy between pre-MDD diagnosis before work-up and discussion was remarkable,” the study authors wrote, estimating that MDD added value for 70% of referred patients.
“We believe MDD should be a common practice in the diagnosis of every patient with suspected ILD,” the researchers said.
The study doesn’t examine the challenges of putting MDD into practice, but Dr. Antin-Ozerkis provided some perspective. “It may be difficult for physicians to take the time from a busy practice to meet with a multidisciplinary team. It can require resources to gather the data necessary to comprehensively assess each patient case. Additionally, maintaining staff with experienced pulmonologists, radiologists and pathologists may be costly.”
She added that “there are various ways in which MDD may occur,” and that the pros and cons of different methods have not been well studied. “This practice will likely evolve with the development of new biomarkers and other diagnostic strategies in IPF [idiopathic pulmonary fibrosis].”
Still, she said, “this joint undertaking is clearly vital in helping to guide clinical practice, including therapeutic decisions and discussion of prognosis. For now, any discussion between clinician, radiologist, and pathologist is of benefit.”
Research Foundation-Flanders and University Hospitals Leuven funded the study. Some study authors reported various disclosures. Dr. Antin-Ozerkis disclosed serving as an investigator on several clinical trials for IPF and other ILDs by Boehringer, Promedior, Fibrogen and Roche. She noted that payments go directly to the university with no direct payments to the investigator.
SOURCE: De Sadeleer LJ et al. Chest. 2018 Mar 30. doi: 10.1016/j.chest.2018.03.026.
New research provides strong statistical support for the use of dynamic multidisciplinary discussion in the diagnosis of patients who may have interstitial lung diseases (ILD).
and it changed the diagnosis in 41% of the other cases.
The American Thoracic Society, European Respiratory Society, Japanese Respiratory Society, and Latin American Thoracic Association adopted joint guidelines for the treatment of idiopathic pulmonary fibrosis in 2015, and the ATS and ERS updated guidelines for the classification and terminology for idiopathic interstitial pneumonias in 2013. The Lancet Respiratory Medicine published what some consider to be a landmark evaluation of multidisciplinary team agreement on diagnosis of interstitial lung disease following the adoption of these guidelines (Walsh SLF et al. 2016;4[7]:557-65). This study showed that in idiopathic pulmonary fibrosis, multidisciplinary team meetings “have a higher level of agreement on diagnoses, assign diagnoses with higher confidence more frequently, and provide diagnoses that have nonsignificant greater prognostic separation than do clinicians or radiologists in most cases,” the researchers wrote.
In the new study, MDD failed to produce a diagnosis or suggestions about a way forward in only 3.5% of patients, according to the study, which appeared March 30 in CHEST®.
According to Dr. Antin-Ozerkis, accurate diagnosis of ILD is crucial to treatment, but it can be challenging to achieve. The MDD approach has been recommended since 2002 by the ATS and ERS, she said.
The study authors, led by Laurens J. De Sadeleer, MD, of Belgium’s University Hospitals Leuven, define the MDD approach as one “in which expert ILD clinicians, radiologists, and pathologists integrate all available clinical data, laboratory results, high-resolution computed tomography [HRCT] findings, and lung biopsy [when performed].”
For the study, the researchers tracked pre-MDD and MDD diagnoses of 938 consecutive patients with possible ILD who were discussed during 2005-2015. Of these patients, referring physicians made preliminary diagnoses in 49% of cases; in the rest, physicians either failed to develop a diagnosis or offered multiple possible diagnoses.
MDD teams produced a change in diagnosis in 191 – 42% – of patients with a pre-MDD diagnosis. Another condition was diagnosed in 118 of these patients, and the MDD teams declined to classify the other 73 patients pending further investigation.
The MDD teams also were able to produce diagnoses in 80% of cases when referring physicians could not come up with diagnoses.
“Discrepancy between pre-MDD diagnosis before work-up and discussion was remarkable,” the study authors wrote, estimating that MDD added value for 70% of referred patients.
“We believe MDD should be a common practice in the diagnosis of every patient with suspected ILD,” the researchers said.
The study doesn’t examine the challenges of putting MDD into practice, but Dr. Antin-Ozerkis provided some perspective. “It may be difficult for physicians to take the time from a busy practice to meet with a multidisciplinary team. It can require resources to gather the data necessary to comprehensively assess each patient case. Additionally, maintaining staff with experienced pulmonologists, radiologists and pathologists may be costly.”
She added that “there are various ways in which MDD may occur,” and that the pros and cons of different methods have not been well studied. “This practice will likely evolve with the development of new biomarkers and other diagnostic strategies in IPF [idiopathic pulmonary fibrosis].”
Still, she said, “this joint undertaking is clearly vital in helping to guide clinical practice, including therapeutic decisions and discussion of prognosis. For now, any discussion between clinician, radiologist, and pathologist is of benefit.”
Research Foundation-Flanders and University Hospitals Leuven funded the study. Some study authors reported various disclosures. Dr. Antin-Ozerkis disclosed serving as an investigator on several clinical trials for IPF and other ILDs by Boehringer, Promedior, Fibrogen and Roche. She noted that payments go directly to the university with no direct payments to the investigator.
SOURCE: De Sadeleer LJ et al. Chest. 2018 Mar 30. doi: 10.1016/j.chest.2018.03.026.
FROM CHEST
Key clinical point: Multidisciplinary discussion (MDD) in cases of suspected interstitial lung disease frequently produces adjustments of previous diagnoses and new diagnoses when none existed previously.
Major finding: MDD teams produced a change in diagnosis in 42% of patients with a pre-MDD diagnosis and in 80% of those without one.
Study details: 938 consecutive patients at University Hospitals Leuven, Belgium, with possible ILD who underwent MDD diagnostics during 2005-2015.
Disclosures: Research Foundation–Flanders and University Hospitals Leuven funded the study. Some study authors reported various disclosures. Dr. Antin-Ozerkis disclosed serving as an investigator on several clinical trials for idiopathic pulmonary fibrosis and other ILDs by Boehringer, Promedior, FibroGen, and Roche. She noted that payments go directly to the university, with no direct payments to the investigator.
Source: De Sadeleer LJ et al. Chest 2018. 2018 Mar 30. doi: 10.1016/j.chest.2018.03.026.
Pregnant women in clinical trials: FDA questions how to include them
Pregnant women are rarely included in clinical drug trials, creating a significant and potentially dangerous gap in knowledge. Now, a new draft guidance from the Food and Drug Administration broadens the discussion about these trials, suggesting issues to consider – including ethics and risks – when testing medications in pregnant women.
“The guidance opens the possibility of ethical conduct of trials in pregnant women but carefully lays out the caveats to be considered,” Christina Chambers, PhD, a perinatal epidemiologist at the University of California, San Diego, said in an interview. “With proper planning and thoughtful consultation with the relevant experts, this change in regulatory limitations will benefit pregnant women and their children.”
Attitudes have evolved toward more acceptance of including pregnant women in drug trials, according to a 2015 committee opinion from the American College of Obstetricians and Gynecologists. Still, “concerns about the potential for pregnancy in research trial participants have led to practices involving overly burdensome contraception requirements,” the opinion states. “Although changes have been made to encourage and recruit more women into research studies, a gap still exists in the available data on health and disease in women, including those who are pregnant” (Obstet Gynecol 2015;126:e100-7).
[polldaddy:9979976]
The draft guidance, released April 6 by the FDA, is “intended to advance scientific research in pregnant women, and discusses issues that should be considered within the framework of human subject protection regulations,” according to posting comments in the Federal Register.
The draft notes that in some cases, the lack of data about drugs may harm pregnant women and their fetuses by leading physicians to be fearful about prescribing medication. Conversely, physicians and pregnant women are often in the dark about the risks and benefits of medications that are prescribed and used, according to the draft.
In terms of research going forward, the guidance says “development of accessible treatment options for the pregnant population is a significant public health issue.”
The guidance, which recommends that clinical trial sponsors consider enlisting ethicists to take part in drug development program, offers these guidelines, among others, to drugmakers:
- It is “ethically justifiable” to include pregnant women in clinical trials under specific circumstances. “Sponsors should consider meeting with the appropriate FDA review division early in the development phase to discuss when and how to include pregnant women in the drug development plan. These discussions should involve FDA experts in bioethics and maternal health.”
- “Pregnant women can be enrolled in clinical trials that involve greater than minimal risk to the fetuses if the trials offer the potential for direct clinical benefit to the enrolled pregnant women and/or their fetuses.”
- A new pregnancy during a randomized, blinded clinical trial should prompt unblinding “so that counseling may be offered based on whether the fetus has been exposed to the investigational drug, placebo, or control.”
- The pregnant woman may continue the trial if potential benefits outweigh the risks.
- In general, pregnant women should not be enrolled in phase 1 and phase 2 clinical trials. Instead, those trials should be completed first “in a nonpregnant population that include females of reproductive potential.”
- Several types of events may call for the cessation of a clinical trial that includes pregnant women, such as serious maternal or fetal adverse events.
The draft guidance should take note of the fact that birth defects often don’t appear for months or even longer, according to Gerald Briggs, BPharm, FCCP, clinical professor of pharmacy at the University of California, San Francisco. “Until first year of life or later, the babies need to be monitored,” he said in an interview.
Mr. Briggs, who led a 2015 report examining the role of pregnant women in phase 4 clinical drug trials, added that the document should take note of recommendations from clinical teratologists regarding the design of animal studies that should be performed prior to human trials (Am J Obstet Gynecol. 2015;213(6):810-5).
Comments on the draft guidance can be made at www.federalregister.gov and are due by June 8, 2018.
Dr. Chambers and Mr. Briggs reported no relevant disclosures.
Pregnant women are rarely included in clinical drug trials, creating a significant and potentially dangerous gap in knowledge. Now, a new draft guidance from the Food and Drug Administration broadens the discussion about these trials, suggesting issues to consider – including ethics and risks – when testing medications in pregnant women.
“The guidance opens the possibility of ethical conduct of trials in pregnant women but carefully lays out the caveats to be considered,” Christina Chambers, PhD, a perinatal epidemiologist at the University of California, San Diego, said in an interview. “With proper planning and thoughtful consultation with the relevant experts, this change in regulatory limitations will benefit pregnant women and their children.”
Attitudes have evolved toward more acceptance of including pregnant women in drug trials, according to a 2015 committee opinion from the American College of Obstetricians and Gynecologists. Still, “concerns about the potential for pregnancy in research trial participants have led to practices involving overly burdensome contraception requirements,” the opinion states. “Although changes have been made to encourage and recruit more women into research studies, a gap still exists in the available data on health and disease in women, including those who are pregnant” (Obstet Gynecol 2015;126:e100-7).
[polldaddy:9979976]
The draft guidance, released April 6 by the FDA, is “intended to advance scientific research in pregnant women, and discusses issues that should be considered within the framework of human subject protection regulations,” according to posting comments in the Federal Register.
The draft notes that in some cases, the lack of data about drugs may harm pregnant women and their fetuses by leading physicians to be fearful about prescribing medication. Conversely, physicians and pregnant women are often in the dark about the risks and benefits of medications that are prescribed and used, according to the draft.
In terms of research going forward, the guidance says “development of accessible treatment options for the pregnant population is a significant public health issue.”
The guidance, which recommends that clinical trial sponsors consider enlisting ethicists to take part in drug development program, offers these guidelines, among others, to drugmakers:
- It is “ethically justifiable” to include pregnant women in clinical trials under specific circumstances. “Sponsors should consider meeting with the appropriate FDA review division early in the development phase to discuss when and how to include pregnant women in the drug development plan. These discussions should involve FDA experts in bioethics and maternal health.”
- “Pregnant women can be enrolled in clinical trials that involve greater than minimal risk to the fetuses if the trials offer the potential for direct clinical benefit to the enrolled pregnant women and/or their fetuses.”
- A new pregnancy during a randomized, blinded clinical trial should prompt unblinding “so that counseling may be offered based on whether the fetus has been exposed to the investigational drug, placebo, or control.”
- The pregnant woman may continue the trial if potential benefits outweigh the risks.
- In general, pregnant women should not be enrolled in phase 1 and phase 2 clinical trials. Instead, those trials should be completed first “in a nonpregnant population that include females of reproductive potential.”
- Several types of events may call for the cessation of a clinical trial that includes pregnant women, such as serious maternal or fetal adverse events.
The draft guidance should take note of the fact that birth defects often don’t appear for months or even longer, according to Gerald Briggs, BPharm, FCCP, clinical professor of pharmacy at the University of California, San Francisco. “Until first year of life or later, the babies need to be monitored,” he said in an interview.
Mr. Briggs, who led a 2015 report examining the role of pregnant women in phase 4 clinical drug trials, added that the document should take note of recommendations from clinical teratologists regarding the design of animal studies that should be performed prior to human trials (Am J Obstet Gynecol. 2015;213(6):810-5).
Comments on the draft guidance can be made at www.federalregister.gov and are due by June 8, 2018.
Dr. Chambers and Mr. Briggs reported no relevant disclosures.
Pregnant women are rarely included in clinical drug trials, creating a significant and potentially dangerous gap in knowledge. Now, a new draft guidance from the Food and Drug Administration broadens the discussion about these trials, suggesting issues to consider – including ethics and risks – when testing medications in pregnant women.
“The guidance opens the possibility of ethical conduct of trials in pregnant women but carefully lays out the caveats to be considered,” Christina Chambers, PhD, a perinatal epidemiologist at the University of California, San Diego, said in an interview. “With proper planning and thoughtful consultation with the relevant experts, this change in regulatory limitations will benefit pregnant women and their children.”
Attitudes have evolved toward more acceptance of including pregnant women in drug trials, according to a 2015 committee opinion from the American College of Obstetricians and Gynecologists. Still, “concerns about the potential for pregnancy in research trial participants have led to practices involving overly burdensome contraception requirements,” the opinion states. “Although changes have been made to encourage and recruit more women into research studies, a gap still exists in the available data on health and disease in women, including those who are pregnant” (Obstet Gynecol 2015;126:e100-7).
[polldaddy:9979976]
The draft guidance, released April 6 by the FDA, is “intended to advance scientific research in pregnant women, and discusses issues that should be considered within the framework of human subject protection regulations,” according to posting comments in the Federal Register.
The draft notes that in some cases, the lack of data about drugs may harm pregnant women and their fetuses by leading physicians to be fearful about prescribing medication. Conversely, physicians and pregnant women are often in the dark about the risks and benefits of medications that are prescribed and used, according to the draft.
In terms of research going forward, the guidance says “development of accessible treatment options for the pregnant population is a significant public health issue.”
The guidance, which recommends that clinical trial sponsors consider enlisting ethicists to take part in drug development program, offers these guidelines, among others, to drugmakers:
- It is “ethically justifiable” to include pregnant women in clinical trials under specific circumstances. “Sponsors should consider meeting with the appropriate FDA review division early in the development phase to discuss when and how to include pregnant women in the drug development plan. These discussions should involve FDA experts in bioethics and maternal health.”
- “Pregnant women can be enrolled in clinical trials that involve greater than minimal risk to the fetuses if the trials offer the potential for direct clinical benefit to the enrolled pregnant women and/or their fetuses.”
- A new pregnancy during a randomized, blinded clinical trial should prompt unblinding “so that counseling may be offered based on whether the fetus has been exposed to the investigational drug, placebo, or control.”
- The pregnant woman may continue the trial if potential benefits outweigh the risks.
- In general, pregnant women should not be enrolled in phase 1 and phase 2 clinical trials. Instead, those trials should be completed first “in a nonpregnant population that include females of reproductive potential.”
- Several types of events may call for the cessation of a clinical trial that includes pregnant women, such as serious maternal or fetal adverse events.
The draft guidance should take note of the fact that birth defects often don’t appear for months or even longer, according to Gerald Briggs, BPharm, FCCP, clinical professor of pharmacy at the University of California, San Francisco. “Until first year of life or later, the babies need to be monitored,” he said in an interview.
Mr. Briggs, who led a 2015 report examining the role of pregnant women in phase 4 clinical drug trials, added that the document should take note of recommendations from clinical teratologists regarding the design of animal studies that should be performed prior to human trials (Am J Obstet Gynecol. 2015;213(6):810-5).
Comments on the draft guidance can be made at www.federalregister.gov and are due by June 8, 2018.
Dr. Chambers and Mr. Briggs reported no relevant disclosures.
Study: Type 2 narcolepsy is significantly different from type 1
Patients with type 1 narcolepsy have more clinical impairments and distinct functional abnormalities than do patients with type 2 narcolepsy, according to investigators.
Patients with type 2 “do not present with such severe handicaps and are clinically closer to hypersomniac patients than the patients with type 1 narcolepsy,” reported Yu-Shu Huang, MD, of Chang Gung Memorial Hospital and Chang Gung University, Taoyuan City, Taiwan, and associates. The study was published in Neurology.
The researchers used brain scans, neuropsychological tests, and other screening tests to analyze three groups of subjects – 104 patients with Na-1, 29 with Na-2, and a control group of 26 subjects. Depending on the group, 62%-66% of the subjects were men, and the mean age ranged from 19 to 20.
The mean age of onset for the narcolepsy groups was 12-13. Those with Na-1 had higher mean body mass indexes – 27 kg/m2 vs. 24 (Na-2) vs. 20 (control), (P = .001).
The patients in both narcolepsy groups showed similar levels of sleepiness, but those with Na-2 had significantly fewer abnormal findings and disturbances.
Patients with Na-2 had significantly fewer sleep-onset REM periods, longer mean sleep latencies, and lower apnea-hypopnea indexes. The human leukocyte antigen DQ-Beta1*0602 was also found less frequently in Na-2 compared to Na-1 (52% vs. 97%, respectively, P less than .001).
PET findings also revealed less impairment in Na-2 compared to Na-1. The researchers noted increased metabolic rate in several brain areas in Na-1, although hypometabolism is more common in some areas in Na-2.
Based on their findings, the authors challenge a previous study of insurance data that suggests patients with both types have similarly poor outcomes over the long term. (PLoS One 2012;7:e33525.)
“In our study,” the authors wrote, “compared to patients with type 2 narcolepsy, patients with type 1 narcolepsy present with much more severe handicaps as early as adolescence. Further studies are needed to clarify the issue.”
The Taiwan Ministry of Science and Technology funded the study. The authors report no relevant disclosures.
SOURCE: Huang Y, et al. March 30, 2018, Neurology. 2018 Mar 30. doi: 10.1212/WNL.0000000000005346.
Patients with type 1 narcolepsy have more clinical impairments and distinct functional abnormalities than do patients with type 2 narcolepsy, according to investigators.
Patients with type 2 “do not present with such severe handicaps and are clinically closer to hypersomniac patients than the patients with type 1 narcolepsy,” reported Yu-Shu Huang, MD, of Chang Gung Memorial Hospital and Chang Gung University, Taoyuan City, Taiwan, and associates. The study was published in Neurology.
The researchers used brain scans, neuropsychological tests, and other screening tests to analyze three groups of subjects – 104 patients with Na-1, 29 with Na-2, and a control group of 26 subjects. Depending on the group, 62%-66% of the subjects were men, and the mean age ranged from 19 to 20.
The mean age of onset for the narcolepsy groups was 12-13. Those with Na-1 had higher mean body mass indexes – 27 kg/m2 vs. 24 (Na-2) vs. 20 (control), (P = .001).
The patients in both narcolepsy groups showed similar levels of sleepiness, but those with Na-2 had significantly fewer abnormal findings and disturbances.
Patients with Na-2 had significantly fewer sleep-onset REM periods, longer mean sleep latencies, and lower apnea-hypopnea indexes. The human leukocyte antigen DQ-Beta1*0602 was also found less frequently in Na-2 compared to Na-1 (52% vs. 97%, respectively, P less than .001).
PET findings also revealed less impairment in Na-2 compared to Na-1. The researchers noted increased metabolic rate in several brain areas in Na-1, although hypometabolism is more common in some areas in Na-2.
Based on their findings, the authors challenge a previous study of insurance data that suggests patients with both types have similarly poor outcomes over the long term. (PLoS One 2012;7:e33525.)
“In our study,” the authors wrote, “compared to patients with type 2 narcolepsy, patients with type 1 narcolepsy present with much more severe handicaps as early as adolescence. Further studies are needed to clarify the issue.”
The Taiwan Ministry of Science and Technology funded the study. The authors report no relevant disclosures.
SOURCE: Huang Y, et al. March 30, 2018, Neurology. 2018 Mar 30. doi: 10.1212/WNL.0000000000005346.
Patients with type 1 narcolepsy have more clinical impairments and distinct functional abnormalities than do patients with type 2 narcolepsy, according to investigators.
Patients with type 2 “do not present with such severe handicaps and are clinically closer to hypersomniac patients than the patients with type 1 narcolepsy,” reported Yu-Shu Huang, MD, of Chang Gung Memorial Hospital and Chang Gung University, Taoyuan City, Taiwan, and associates. The study was published in Neurology.
The researchers used brain scans, neuropsychological tests, and other screening tests to analyze three groups of subjects – 104 patients with Na-1, 29 with Na-2, and a control group of 26 subjects. Depending on the group, 62%-66% of the subjects were men, and the mean age ranged from 19 to 20.
The mean age of onset for the narcolepsy groups was 12-13. Those with Na-1 had higher mean body mass indexes – 27 kg/m2 vs. 24 (Na-2) vs. 20 (control), (P = .001).
The patients in both narcolepsy groups showed similar levels of sleepiness, but those with Na-2 had significantly fewer abnormal findings and disturbances.
Patients with Na-2 had significantly fewer sleep-onset REM periods, longer mean sleep latencies, and lower apnea-hypopnea indexes. The human leukocyte antigen DQ-Beta1*0602 was also found less frequently in Na-2 compared to Na-1 (52% vs. 97%, respectively, P less than .001).
PET findings also revealed less impairment in Na-2 compared to Na-1. The researchers noted increased metabolic rate in several brain areas in Na-1, although hypometabolism is more common in some areas in Na-2.
Based on their findings, the authors challenge a previous study of insurance data that suggests patients with both types have similarly poor outcomes over the long term. (PLoS One 2012;7:e33525.)
“In our study,” the authors wrote, “compared to patients with type 2 narcolepsy, patients with type 1 narcolepsy present with much more severe handicaps as early as adolescence. Further studies are needed to clarify the issue.”
The Taiwan Ministry of Science and Technology funded the study. The authors report no relevant disclosures.
SOURCE: Huang Y, et al. March 30, 2018, Neurology. 2018 Mar 30. doi: 10.1212/WNL.0000000000005346.
FROM NEUROLOGY
Prioritize topical treatments in pregnant women
SAN DIEGO – “Some doctors don’t treat them at all, or they undertreat because they’re afraid of prescribing anything,” said dermatologist Kelly H. Tyler, MD.
But the Food and Drug Administration’s new system of prescription drug labeling for pregnant women offers improved insight for dermatologists, and a variety of alternatives are available to commonly used dermatologic drugs that pose risks, said Dr. Tyler, who has a unique perspective on this issue: She used to practice as an ob.gyn.
In her presentation, she provided the following tips when caring for pregnant patients.
Check the prescription drug labeling for pregnant and lactating women
Prior to 2015, the FDA used a letter ratings to denote the risk of medications during pregnancy. The ratings – A, B, C, D, and X – go from “A” showing no fetal risk in controlled studies to “X” in which a drug is contraindicated because “there is no reason to risk use of drug in pregnancy.”
This rating system is problematic, Dr. Tyler said, since it’s imprecise. She noted that about two-thirds of medications have a risk rating of C, which means “Risk cannot be ruled out; human studies may or may not show risk; potential benefits may justify potential risk.” But, she asked, “How are you supposed to know what to do with that kind of information?”
By June 30, 2015, FDA had retired the letter classifications and replaced this system with the Pregnancy and Lactation Labeling final rule, which requires that labels include a pregnancy section with information on pregnancy exposure registries, in addition to risk summaries, clinical considerations, and available data. There are also sections on lactation, including nursing mothers, and females and males of reproductive potential. (This information appears in the prescription drug labeling sections 8.1-8.3.) Labels for medications approved prior to June 30, 2001 did not need to be updated. Labels for newer medications are required to comply within 3-5 years of the 2015 policy change.
Ask female patients about sexual activity
It’s important to ask all premenopausal female patients whether they’re sexually active and whether they are using birth control, trying to conceive, or are currently pregnant. If they’re pregnant, what trimester are they in?
Keep in mind, she said, that women can be pregnant for quite some time without knowing it. Home pregnancy tests may not show positive results for up to 5 weeks after conception, she said. But the embryonic/organogenesis stage – from 2 to 8 weeks – is the most important period for a pregnant mother avoid drugs that could damage the unborn child and is “when you want to avoid most medications,” she said.
Risks don’t end after a few months, however. Dr. Tyler urged dermatologists to keep in mind that the brain, teeth, and bones remain susceptible to damage from teratogenic medications after the eighth week.
Keep topical medications in mind
When it comes to pregnant women, “for most dermatologic therapies, topical medications will be your No. 1 choice” because of less absorption, Dr. Tyler said.
But there are exceptions, she noted. In acne and rosacea, for example, there’s controversy about the safety of topical retinoids. “I would just urge you to not use those medications in pregnancy because you don’t really need to use them,” Dr. Tyler said. “You have other options that are safer.”
Adapalene and tretinoin are listed as category C (risk cannot be ruled out in pregnancy) and topical tazarotene is category X (contraindicated in pregnancy) because of retinoid-like anomalies in animal studies, she said.
Avoid 3 major contraindicated systemic drugs
Isotretinoin, acitretin, and methotrexate are “absolutely contraindicated” for anyone who is pregnant or could become pregnant, Dr. Tyler said.
Use some systemic drugs with caution
Tetracyclines are category D (positive evidence of risk to human fetus, but benefits may outweigh risks of drug) with the highest risk in the second and third trimesters, Dr. Tyler said. “If for some reason you have a patient who’s on tetracycline, stop before she’s in her second trimester.”
She recommended avoiding the antibiotic erythromycin because of studies hinting at risks when used early in pregnancy. Spironolactone is theoretically risky after the week 8 of pregnancy, she said. “Because of the animal studies, we typically do not use this during pregnancy.”
Psoriasis often improves during pregnancy
Psoriasis improves during pregnancy in about half of women with psoriasis and worsens in about 20%, Dr. Tyler said. During pregnancy, topical treatments are the first-line treatment, said Dr. Tyler, who recommends that treatment should begin with topical steroids, then calcipotriene or tacrolimus if needed (J Am Acad Dermatol. 2013 Apr;68[4]:663-71). Cyclosporine is an option, as is phototherapy, she said. In an interview, she noted that phototherapy (narrowband UVB) would be considered next-line therapy in patients who are past the first 28 days of gestation and have failed topical therapy, given they are taking adequate folic acid supplementation, which is present in prenatal vitamins.
“As for biologics, you have to go with older ones that have more data. When we look at newer medications, we don’t know a lot about them,” she said during the presentation.
It’s okay to prescribe oral steroids and antihistamines
Oral steroids are safe during pregnancy, Dr. Tyler said, but “just be judicious” with moderate doses and short durations.
Antihistamines are also appropriate, she said, but be aware of the potential for neonatal sedation during lactation.
As for antibiotics for bacterial infections, azithromycin, penicillins, and cephalosporins are all category B (no risk to human fetus despite possible animal risk; or no risk in animal studies and human studies not done), as are all topical antibiotics except dapsone, which is category C because of a theoretical risk of neonatal hyperbilirubinemia if used near the time of delivery.
Hydroxychloroquine may be appropriate for connective tissue disease, she said, although steroids may be a better option in some cases.
And topical antifungals are considered safer for fungal diseases than systemic medications. She said she prefers clotrimazole and oxiconazole, both category B.
Finally, Dr. Tyler recommended permethrin (category B) for parasitic infections since it has been used extensively in pregnancy without a sign of risk and is the preferred treatment for scabies. It’s a better option than ivermectin, she said.
Postpone surgery until at least the second trimester
If it’s not possible to delay nonemergent dermatologic surgery until after pregnancy, she recommended performing procedures during the second trimester. Destruction of local lesions, however, is safe without anesthesia.
“In summary,” when treating pregnant patients, Dr. Tyler said, “a conservative approach is always best, topical medications are always first-line for any condition, and certain oral medications are safe.”
Dr. Tyler reported no relevant disclosures.
SAN DIEGO – “Some doctors don’t treat them at all, or they undertreat because they’re afraid of prescribing anything,” said dermatologist Kelly H. Tyler, MD.
But the Food and Drug Administration’s new system of prescription drug labeling for pregnant women offers improved insight for dermatologists, and a variety of alternatives are available to commonly used dermatologic drugs that pose risks, said Dr. Tyler, who has a unique perspective on this issue: She used to practice as an ob.gyn.
In her presentation, she provided the following tips when caring for pregnant patients.
Check the prescription drug labeling for pregnant and lactating women
Prior to 2015, the FDA used a letter ratings to denote the risk of medications during pregnancy. The ratings – A, B, C, D, and X – go from “A” showing no fetal risk in controlled studies to “X” in which a drug is contraindicated because “there is no reason to risk use of drug in pregnancy.”
This rating system is problematic, Dr. Tyler said, since it’s imprecise. She noted that about two-thirds of medications have a risk rating of C, which means “Risk cannot be ruled out; human studies may or may not show risk; potential benefits may justify potential risk.” But, she asked, “How are you supposed to know what to do with that kind of information?”
By June 30, 2015, FDA had retired the letter classifications and replaced this system with the Pregnancy and Lactation Labeling final rule, which requires that labels include a pregnancy section with information on pregnancy exposure registries, in addition to risk summaries, clinical considerations, and available data. There are also sections on lactation, including nursing mothers, and females and males of reproductive potential. (This information appears in the prescription drug labeling sections 8.1-8.3.) Labels for medications approved prior to June 30, 2001 did not need to be updated. Labels for newer medications are required to comply within 3-5 years of the 2015 policy change.
Ask female patients about sexual activity
It’s important to ask all premenopausal female patients whether they’re sexually active and whether they are using birth control, trying to conceive, or are currently pregnant. If they’re pregnant, what trimester are they in?
Keep in mind, she said, that women can be pregnant for quite some time without knowing it. Home pregnancy tests may not show positive results for up to 5 weeks after conception, she said. But the embryonic/organogenesis stage – from 2 to 8 weeks – is the most important period for a pregnant mother avoid drugs that could damage the unborn child and is “when you want to avoid most medications,” she said.
Risks don’t end after a few months, however. Dr. Tyler urged dermatologists to keep in mind that the brain, teeth, and bones remain susceptible to damage from teratogenic medications after the eighth week.
Keep topical medications in mind
When it comes to pregnant women, “for most dermatologic therapies, topical medications will be your No. 1 choice” because of less absorption, Dr. Tyler said.
But there are exceptions, she noted. In acne and rosacea, for example, there’s controversy about the safety of topical retinoids. “I would just urge you to not use those medications in pregnancy because you don’t really need to use them,” Dr. Tyler said. “You have other options that are safer.”
Adapalene and tretinoin are listed as category C (risk cannot be ruled out in pregnancy) and topical tazarotene is category X (contraindicated in pregnancy) because of retinoid-like anomalies in animal studies, she said.
Avoid 3 major contraindicated systemic drugs
Isotretinoin, acitretin, and methotrexate are “absolutely contraindicated” for anyone who is pregnant or could become pregnant, Dr. Tyler said.
Use some systemic drugs with caution
Tetracyclines are category D (positive evidence of risk to human fetus, but benefits may outweigh risks of drug) with the highest risk in the second and third trimesters, Dr. Tyler said. “If for some reason you have a patient who’s on tetracycline, stop before she’s in her second trimester.”
She recommended avoiding the antibiotic erythromycin because of studies hinting at risks when used early in pregnancy. Spironolactone is theoretically risky after the week 8 of pregnancy, she said. “Because of the animal studies, we typically do not use this during pregnancy.”
Psoriasis often improves during pregnancy
Psoriasis improves during pregnancy in about half of women with psoriasis and worsens in about 20%, Dr. Tyler said. During pregnancy, topical treatments are the first-line treatment, said Dr. Tyler, who recommends that treatment should begin with topical steroids, then calcipotriene or tacrolimus if needed (J Am Acad Dermatol. 2013 Apr;68[4]:663-71). Cyclosporine is an option, as is phototherapy, she said. In an interview, she noted that phototherapy (narrowband UVB) would be considered next-line therapy in patients who are past the first 28 days of gestation and have failed topical therapy, given they are taking adequate folic acid supplementation, which is present in prenatal vitamins.
“As for biologics, you have to go with older ones that have more data. When we look at newer medications, we don’t know a lot about them,” she said during the presentation.
It’s okay to prescribe oral steroids and antihistamines
Oral steroids are safe during pregnancy, Dr. Tyler said, but “just be judicious” with moderate doses and short durations.
Antihistamines are also appropriate, she said, but be aware of the potential for neonatal sedation during lactation.
As for antibiotics for bacterial infections, azithromycin, penicillins, and cephalosporins are all category B (no risk to human fetus despite possible animal risk; or no risk in animal studies and human studies not done), as are all topical antibiotics except dapsone, which is category C because of a theoretical risk of neonatal hyperbilirubinemia if used near the time of delivery.
Hydroxychloroquine may be appropriate for connective tissue disease, she said, although steroids may be a better option in some cases.
And topical antifungals are considered safer for fungal diseases than systemic medications. She said she prefers clotrimazole and oxiconazole, both category B.
Finally, Dr. Tyler recommended permethrin (category B) for parasitic infections since it has been used extensively in pregnancy without a sign of risk and is the preferred treatment for scabies. It’s a better option than ivermectin, she said.
Postpone surgery until at least the second trimester
If it’s not possible to delay nonemergent dermatologic surgery until after pregnancy, she recommended performing procedures during the second trimester. Destruction of local lesions, however, is safe without anesthesia.
“In summary,” when treating pregnant patients, Dr. Tyler said, “a conservative approach is always best, topical medications are always first-line for any condition, and certain oral medications are safe.”
Dr. Tyler reported no relevant disclosures.
SAN DIEGO – “Some doctors don’t treat them at all, or they undertreat because they’re afraid of prescribing anything,” said dermatologist Kelly H. Tyler, MD.
But the Food and Drug Administration’s new system of prescription drug labeling for pregnant women offers improved insight for dermatologists, and a variety of alternatives are available to commonly used dermatologic drugs that pose risks, said Dr. Tyler, who has a unique perspective on this issue: She used to practice as an ob.gyn.
In her presentation, she provided the following tips when caring for pregnant patients.
Check the prescription drug labeling for pregnant and lactating women
Prior to 2015, the FDA used a letter ratings to denote the risk of medications during pregnancy. The ratings – A, B, C, D, and X – go from “A” showing no fetal risk in controlled studies to “X” in which a drug is contraindicated because “there is no reason to risk use of drug in pregnancy.”
This rating system is problematic, Dr. Tyler said, since it’s imprecise. She noted that about two-thirds of medications have a risk rating of C, which means “Risk cannot be ruled out; human studies may or may not show risk; potential benefits may justify potential risk.” But, she asked, “How are you supposed to know what to do with that kind of information?”
By June 30, 2015, FDA had retired the letter classifications and replaced this system with the Pregnancy and Lactation Labeling final rule, which requires that labels include a pregnancy section with information on pregnancy exposure registries, in addition to risk summaries, clinical considerations, and available data. There are also sections on lactation, including nursing mothers, and females and males of reproductive potential. (This information appears in the prescription drug labeling sections 8.1-8.3.) Labels for medications approved prior to June 30, 2001 did not need to be updated. Labels for newer medications are required to comply within 3-5 years of the 2015 policy change.
Ask female patients about sexual activity
It’s important to ask all premenopausal female patients whether they’re sexually active and whether they are using birth control, trying to conceive, or are currently pregnant. If they’re pregnant, what trimester are they in?
Keep in mind, she said, that women can be pregnant for quite some time without knowing it. Home pregnancy tests may not show positive results for up to 5 weeks after conception, she said. But the embryonic/organogenesis stage – from 2 to 8 weeks – is the most important period for a pregnant mother avoid drugs that could damage the unborn child and is “when you want to avoid most medications,” she said.
Risks don’t end after a few months, however. Dr. Tyler urged dermatologists to keep in mind that the brain, teeth, and bones remain susceptible to damage from teratogenic medications after the eighth week.
Keep topical medications in mind
When it comes to pregnant women, “for most dermatologic therapies, topical medications will be your No. 1 choice” because of less absorption, Dr. Tyler said.
But there are exceptions, she noted. In acne and rosacea, for example, there’s controversy about the safety of topical retinoids. “I would just urge you to not use those medications in pregnancy because you don’t really need to use them,” Dr. Tyler said. “You have other options that are safer.”
Adapalene and tretinoin are listed as category C (risk cannot be ruled out in pregnancy) and topical tazarotene is category X (contraindicated in pregnancy) because of retinoid-like anomalies in animal studies, she said.
Avoid 3 major contraindicated systemic drugs
Isotretinoin, acitretin, and methotrexate are “absolutely contraindicated” for anyone who is pregnant or could become pregnant, Dr. Tyler said.
Use some systemic drugs with caution
Tetracyclines are category D (positive evidence of risk to human fetus, but benefits may outweigh risks of drug) with the highest risk in the second and third trimesters, Dr. Tyler said. “If for some reason you have a patient who’s on tetracycline, stop before she’s in her second trimester.”
She recommended avoiding the antibiotic erythromycin because of studies hinting at risks when used early in pregnancy. Spironolactone is theoretically risky after the week 8 of pregnancy, she said. “Because of the animal studies, we typically do not use this during pregnancy.”
Psoriasis often improves during pregnancy
Psoriasis improves during pregnancy in about half of women with psoriasis and worsens in about 20%, Dr. Tyler said. During pregnancy, topical treatments are the first-line treatment, said Dr. Tyler, who recommends that treatment should begin with topical steroids, then calcipotriene or tacrolimus if needed (J Am Acad Dermatol. 2013 Apr;68[4]:663-71). Cyclosporine is an option, as is phototherapy, she said. In an interview, she noted that phototherapy (narrowband UVB) would be considered next-line therapy in patients who are past the first 28 days of gestation and have failed topical therapy, given they are taking adequate folic acid supplementation, which is present in prenatal vitamins.
“As for biologics, you have to go with older ones that have more data. When we look at newer medications, we don’t know a lot about them,” she said during the presentation.
It’s okay to prescribe oral steroids and antihistamines
Oral steroids are safe during pregnancy, Dr. Tyler said, but “just be judicious” with moderate doses and short durations.
Antihistamines are also appropriate, she said, but be aware of the potential for neonatal sedation during lactation.
As for antibiotics for bacterial infections, azithromycin, penicillins, and cephalosporins are all category B (no risk to human fetus despite possible animal risk; or no risk in animal studies and human studies not done), as are all topical antibiotics except dapsone, which is category C because of a theoretical risk of neonatal hyperbilirubinemia if used near the time of delivery.
Hydroxychloroquine may be appropriate for connective tissue disease, she said, although steroids may be a better option in some cases.
And topical antifungals are considered safer for fungal diseases than systemic medications. She said she prefers clotrimazole and oxiconazole, both category B.
Finally, Dr. Tyler recommended permethrin (category B) for parasitic infections since it has been used extensively in pregnancy without a sign of risk and is the preferred treatment for scabies. It’s a better option than ivermectin, she said.
Postpone surgery until at least the second trimester
If it’s not possible to delay nonemergent dermatologic surgery until after pregnancy, she recommended performing procedures during the second trimester. Destruction of local lesions, however, is safe without anesthesia.
“In summary,” when treating pregnant patients, Dr. Tyler said, “a conservative approach is always best, topical medications are always first-line for any condition, and certain oral medications are safe.”
Dr. Tyler reported no relevant disclosures.
EXPERT ANALYSIS FROM AAD 18
Dramatic improvements reported after surgery for hidradenitis suppurativa
A retrospective German study found that the majority of , with many saying they no longer suffered from everyday impairment from the disease.
“We were able to show that surgical therapy resulted in convincing improvement of life quality and long-term results for HS that are at least as effective as biologicals,” the researchers wrote. The study was published online in the Journal of the European Academy of Dermatology and Venereology.
Lukas Kofler, MD, and associates from the department of dermatology at Eberhard Karls University’s University Medical Center, Tübingen, Germany, surveyed 910 of the facility’s patients who had undergone wide local excision for HS from 2006 to 2015. Surgery was “designed to reach into clinically disease-free subcutaneous fatty tissue,” followed by second intention healing, they wrote.
A total of 255 patients answered the survey, a response rate of 28%. There were 103 men and 152 women with a median age of 38 years (range, 14-66 years); 75% reported prior “nicotine abuse.” Almost half had been treated previously, most often with systemic antibiotics in 68%. The mean follow-up time was 57 months (range, 19-127 months);
All cases were Hurley grade III. Just over three-quarters of the patients described disease-related limitations in private life prior to surgery as “very strong” or “strong,” and 95% reported that their day-to-day life was impaired. Sixty percent said the disease impaired their work life (another 8% were not employed).
After surgery, 27% experienced postoperative complications, including minor bleeding, infection, and limited mobility; 65% experienced pain but 38% of the patients required analgesics postoperatively.
After surgery, 80% were satisfied or very satisfied with the results, and more than two-thirds were satisfied with the cosmetic results. Just over half said their private life was not impaired by the disease at all, compared with 3% who said so before surgery. After surgery, 20% reported being strongly or very strongly impaired, compared with 77% before surgery.
Nearly 70% reported that HS recurred after surgery, but 62% of those with recurrences said the disease was not as severe as before surgery.
“Surgery represents an important treatment option by itself but should also be part of combined therapeutic strategies, especially in severe disease stages. However, consistent approaches combining systemic and surgical treatments have not been established yet,” the authors noted.
The study had no funding source. The authors had no conflicts to disclose.
SOURCE: Kofler L et al. J Eur Acad Dermatol Venereol. 2018 Mar 23. doi: 10.1111/jdv.14892.
A retrospective German study found that the majority of , with many saying they no longer suffered from everyday impairment from the disease.
“We were able to show that surgical therapy resulted in convincing improvement of life quality and long-term results for HS that are at least as effective as biologicals,” the researchers wrote. The study was published online in the Journal of the European Academy of Dermatology and Venereology.
Lukas Kofler, MD, and associates from the department of dermatology at Eberhard Karls University’s University Medical Center, Tübingen, Germany, surveyed 910 of the facility’s patients who had undergone wide local excision for HS from 2006 to 2015. Surgery was “designed to reach into clinically disease-free subcutaneous fatty tissue,” followed by second intention healing, they wrote.
A total of 255 patients answered the survey, a response rate of 28%. There were 103 men and 152 women with a median age of 38 years (range, 14-66 years); 75% reported prior “nicotine abuse.” Almost half had been treated previously, most often with systemic antibiotics in 68%. The mean follow-up time was 57 months (range, 19-127 months);
All cases were Hurley grade III. Just over three-quarters of the patients described disease-related limitations in private life prior to surgery as “very strong” or “strong,” and 95% reported that their day-to-day life was impaired. Sixty percent said the disease impaired their work life (another 8% were not employed).
After surgery, 27% experienced postoperative complications, including minor bleeding, infection, and limited mobility; 65% experienced pain but 38% of the patients required analgesics postoperatively.
After surgery, 80% were satisfied or very satisfied with the results, and more than two-thirds were satisfied with the cosmetic results. Just over half said their private life was not impaired by the disease at all, compared with 3% who said so before surgery. After surgery, 20% reported being strongly or very strongly impaired, compared with 77% before surgery.
Nearly 70% reported that HS recurred after surgery, but 62% of those with recurrences said the disease was not as severe as before surgery.
“Surgery represents an important treatment option by itself but should also be part of combined therapeutic strategies, especially in severe disease stages. However, consistent approaches combining systemic and surgical treatments have not been established yet,” the authors noted.
The study had no funding source. The authors had no conflicts to disclose.
SOURCE: Kofler L et al. J Eur Acad Dermatol Venereol. 2018 Mar 23. doi: 10.1111/jdv.14892.
A retrospective German study found that the majority of , with many saying they no longer suffered from everyday impairment from the disease.
“We were able to show that surgical therapy resulted in convincing improvement of life quality and long-term results for HS that are at least as effective as biologicals,” the researchers wrote. The study was published online in the Journal of the European Academy of Dermatology and Venereology.
Lukas Kofler, MD, and associates from the department of dermatology at Eberhard Karls University’s University Medical Center, Tübingen, Germany, surveyed 910 of the facility’s patients who had undergone wide local excision for HS from 2006 to 2015. Surgery was “designed to reach into clinically disease-free subcutaneous fatty tissue,” followed by second intention healing, they wrote.
A total of 255 patients answered the survey, a response rate of 28%. There were 103 men and 152 women with a median age of 38 years (range, 14-66 years); 75% reported prior “nicotine abuse.” Almost half had been treated previously, most often with systemic antibiotics in 68%. The mean follow-up time was 57 months (range, 19-127 months);
All cases were Hurley grade III. Just over three-quarters of the patients described disease-related limitations in private life prior to surgery as “very strong” or “strong,” and 95% reported that their day-to-day life was impaired. Sixty percent said the disease impaired their work life (another 8% were not employed).
After surgery, 27% experienced postoperative complications, including minor bleeding, infection, and limited mobility; 65% experienced pain but 38% of the patients required analgesics postoperatively.
After surgery, 80% were satisfied or very satisfied with the results, and more than two-thirds were satisfied with the cosmetic results. Just over half said their private life was not impaired by the disease at all, compared with 3% who said so before surgery. After surgery, 20% reported being strongly or very strongly impaired, compared with 77% before surgery.
Nearly 70% reported that HS recurred after surgery, but 62% of those with recurrences said the disease was not as severe as before surgery.
“Surgery represents an important treatment option by itself but should also be part of combined therapeutic strategies, especially in severe disease stages. However, consistent approaches combining systemic and surgical treatments have not been established yet,” the authors noted.
The study had no funding source. The authors had no conflicts to disclose.
SOURCE: Kofler L et al. J Eur Acad Dermatol Venereol. 2018 Mar 23. doi: 10.1111/jdv.14892.
FROM THE JOURNAL OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY.
Key clinical point: Patients with hidradenitis suppurativa (HS) report dramatic improvement after radical surgery.
Major finding: The percentage reporting strong or very strong impairment of private life fell from 77% before surgery to 20% afterward.
Study details: A retrospective survey of 255 patients who had undergone surgery for HS (Hurley stage III).
Disclosures: The study had no funding source. The authors had no conflicts to disclose.
Source: Kofler L et al. J Eur Acad Dermatol Venereol. 2018 Mar 23. doi: 10.1111/jdv.14892.
From weekend warriors to pros, athletes are plagued by skin disorders
SAN DIEGO – Think beyond the foot: Fungal infections are just the beginning when it comes to skin disorders in athletes, which include ringworm in wrestlers, “jogger’s nipple” in runners, and more serious conditions – like skin cancer.
“from weekend warriors to professional athletes,” said Brian B. Adams, MD, MPH, professor and chair of the department of dermatology at the University of Cincinnati.
In an interview, he discussed specific risks for athletes, the scarcity of data on skin cancer in athletes, and the hazards posed by cotton clothing.
Skin cancer: Risk seems clear, but data are sparse
“Athletes in general appear to be at increased risk in the long term for skin cancer” since they often play and practice during the hours between 10 a.m. and 4 p.m., when the danger of sun exposure is at its highest, he said. In addition, “sweating removes the sunscreen that athletes put on, and there is evidence that sweating actually increases the chance of burning,” he said.
Skiers and snowboarders face unique sun exposure risks, he added. “Snow reflects up to 100% of UV, so even though they may be in shade, they experience UV. And mountain athletes experience greater amounts of UV as the altitude of the mountain increases: At higher altitudes, the atmosphere has less chance of filtering out the damaging rays.”
While it’s obvious that many athletes face extra sun exposure, Dr. Adams pointed out, “very little is definitively known about the actual degree of risk of athletic activities.”
Still, the research that does exist provides plenty of hints about risk. “Large epidemiological studies showed that recreational activities such as sun exposure on the beach or during water sports were associated with an increased risk of basal cell carcinoma, whereas skiing has been shown to be at increased risk for squamous cell carcinoma,” according to a 2008 report on outdoor sports and skin cancer (Clin Dermatol. 2008 Jan-Feb;26[1]:12-5).
“Risk factors of cutaneous melanoma, such as the number of melanocytic nevi and solar lentigines, have been found to be more frequent in subjects practicing endurance outdoor sports. An increased risk for cutaneous melanoma may be assumed for these athletes,” Dr. Adams commented.
Another study, this one published in 2006, found more atypical melanocytic nevi, solar lentigines, and lesions suggestive of nonmelanoma skin cancer in marathon runners, compared with a control group, and the risk was associated with the level of training intensity. The control subjects were more sensitive to the sun and had more common melanocytic nevi (Arch Dermatol. 2006 Nov;142[11]:1471-4).
Counseling about sunscreen may actually work
One strategy to reduce sun exposure is to advise athletes to avoid peak sun hours. However, “the key to caring for the athletes is not only recognizing that their sport may play a role in their disease but also realizing that your therapeutic approach must be tailored to minimize disruption to their practices and competitions,” Dr. Adams said.
However, there’s good news for dermatologists who are willing to push: The study also reported that athletes who were encouraged to use sunscreen were significantly more likely to use sunscreen (P less than .0001).
Watch for other conditions, from jogger’s nipple to ringworm
Dr. Adams offered advice about detection, treatment, and prevention of other skin disorders that affect athletes:
- “Jogger’s nipples” and other kinds of chafing. He has learned to recognize the “red eleven” – two vertical streaks of blood on a runner’s shirt – that represent a case of “jogger’s nipples” caused by chafing. Antibacterial ointment or petroleum jelly are useful treatments, he said, and an application of plenty of petroleum jelly on the nipples prior to a run can be helpful. Cotton shirts should be avoided, he said, in favor of synthetic, moisture-wicking shirts and bras. Chafing can also occur in the underarms and inner thighs, he said, and the same treatments and preventive techniques are useful.
- Callused and bleeding “jogger’s toes.” This can strike runners, especially on the second toe, which is often the longest and most likely to strike the toe box of a shoe. Specialty shoes can help prevent this condition, he said.
- Tinea corporis (ringworm) and herpes gladiatorum. In wrestlers, ringworm is known as tinea corporis gladiatorum because the intensity of skin-to-skin contact in wrestling makes the condition especially common in these athletes. Lesions don’t develop as rings at first; instead, they first appear as relatively nonspecific red round lesions and are most likely to be found in the head, neck, and upper extremities, Dr. Adams noted. Herpes gladiatorum is caused by herpes simplex virus 1; it is also seen in wrestlers and caused by skin-to-skin contact. Topical and oral antifungals clear ringworm, while oral antiviral agents are appropriate for herpes gladiatorum, Dr. Adams said. While herpes gladiatorum clears up and is no longer contagious after 4-5 days, he said, it’s not clear how long wrestlers with ringworm should be disqualified from playing.
Dr. Adams disclosed advising Mission, a company that focuses on sunscreen designed by and for athletes.
SAN DIEGO – Think beyond the foot: Fungal infections are just the beginning when it comes to skin disorders in athletes, which include ringworm in wrestlers, “jogger’s nipple” in runners, and more serious conditions – like skin cancer.
“from weekend warriors to professional athletes,” said Brian B. Adams, MD, MPH, professor and chair of the department of dermatology at the University of Cincinnati.
In an interview, he discussed specific risks for athletes, the scarcity of data on skin cancer in athletes, and the hazards posed by cotton clothing.
Skin cancer: Risk seems clear, but data are sparse
“Athletes in general appear to be at increased risk in the long term for skin cancer” since they often play and practice during the hours between 10 a.m. and 4 p.m., when the danger of sun exposure is at its highest, he said. In addition, “sweating removes the sunscreen that athletes put on, and there is evidence that sweating actually increases the chance of burning,” he said.
Skiers and snowboarders face unique sun exposure risks, he added. “Snow reflects up to 100% of UV, so even though they may be in shade, they experience UV. And mountain athletes experience greater amounts of UV as the altitude of the mountain increases: At higher altitudes, the atmosphere has less chance of filtering out the damaging rays.”
While it’s obvious that many athletes face extra sun exposure, Dr. Adams pointed out, “very little is definitively known about the actual degree of risk of athletic activities.”
Still, the research that does exist provides plenty of hints about risk. “Large epidemiological studies showed that recreational activities such as sun exposure on the beach or during water sports were associated with an increased risk of basal cell carcinoma, whereas skiing has been shown to be at increased risk for squamous cell carcinoma,” according to a 2008 report on outdoor sports and skin cancer (Clin Dermatol. 2008 Jan-Feb;26[1]:12-5).
“Risk factors of cutaneous melanoma, such as the number of melanocytic nevi and solar lentigines, have been found to be more frequent in subjects practicing endurance outdoor sports. An increased risk for cutaneous melanoma may be assumed for these athletes,” Dr. Adams commented.
Another study, this one published in 2006, found more atypical melanocytic nevi, solar lentigines, and lesions suggestive of nonmelanoma skin cancer in marathon runners, compared with a control group, and the risk was associated with the level of training intensity. The control subjects were more sensitive to the sun and had more common melanocytic nevi (Arch Dermatol. 2006 Nov;142[11]:1471-4).
Counseling about sunscreen may actually work
One strategy to reduce sun exposure is to advise athletes to avoid peak sun hours. However, “the key to caring for the athletes is not only recognizing that their sport may play a role in their disease but also realizing that your therapeutic approach must be tailored to minimize disruption to their practices and competitions,” Dr. Adams said.
However, there’s good news for dermatologists who are willing to push: The study also reported that athletes who were encouraged to use sunscreen were significantly more likely to use sunscreen (P less than .0001).
Watch for other conditions, from jogger’s nipple to ringworm
Dr. Adams offered advice about detection, treatment, and prevention of other skin disorders that affect athletes:
- “Jogger’s nipples” and other kinds of chafing. He has learned to recognize the “red eleven” – two vertical streaks of blood on a runner’s shirt – that represent a case of “jogger’s nipples” caused by chafing. Antibacterial ointment or petroleum jelly are useful treatments, he said, and an application of plenty of petroleum jelly on the nipples prior to a run can be helpful. Cotton shirts should be avoided, he said, in favor of synthetic, moisture-wicking shirts and bras. Chafing can also occur in the underarms and inner thighs, he said, and the same treatments and preventive techniques are useful.
- Callused and bleeding “jogger’s toes.” This can strike runners, especially on the second toe, which is often the longest and most likely to strike the toe box of a shoe. Specialty shoes can help prevent this condition, he said.
- Tinea corporis (ringworm) and herpes gladiatorum. In wrestlers, ringworm is known as tinea corporis gladiatorum because the intensity of skin-to-skin contact in wrestling makes the condition especially common in these athletes. Lesions don’t develop as rings at first; instead, they first appear as relatively nonspecific red round lesions and are most likely to be found in the head, neck, and upper extremities, Dr. Adams noted. Herpes gladiatorum is caused by herpes simplex virus 1; it is also seen in wrestlers and caused by skin-to-skin contact. Topical and oral antifungals clear ringworm, while oral antiviral agents are appropriate for herpes gladiatorum, Dr. Adams said. While herpes gladiatorum clears up and is no longer contagious after 4-5 days, he said, it’s not clear how long wrestlers with ringworm should be disqualified from playing.
Dr. Adams disclosed advising Mission, a company that focuses on sunscreen designed by and for athletes.
SAN DIEGO – Think beyond the foot: Fungal infections are just the beginning when it comes to skin disorders in athletes, which include ringworm in wrestlers, “jogger’s nipple” in runners, and more serious conditions – like skin cancer.
“from weekend warriors to professional athletes,” said Brian B. Adams, MD, MPH, professor and chair of the department of dermatology at the University of Cincinnati.
In an interview, he discussed specific risks for athletes, the scarcity of data on skin cancer in athletes, and the hazards posed by cotton clothing.
Skin cancer: Risk seems clear, but data are sparse
“Athletes in general appear to be at increased risk in the long term for skin cancer” since they often play and practice during the hours between 10 a.m. and 4 p.m., when the danger of sun exposure is at its highest, he said. In addition, “sweating removes the sunscreen that athletes put on, and there is evidence that sweating actually increases the chance of burning,” he said.
Skiers and snowboarders face unique sun exposure risks, he added. “Snow reflects up to 100% of UV, so even though they may be in shade, they experience UV. And mountain athletes experience greater amounts of UV as the altitude of the mountain increases: At higher altitudes, the atmosphere has less chance of filtering out the damaging rays.”
While it’s obvious that many athletes face extra sun exposure, Dr. Adams pointed out, “very little is definitively known about the actual degree of risk of athletic activities.”
Still, the research that does exist provides plenty of hints about risk. “Large epidemiological studies showed that recreational activities such as sun exposure on the beach or during water sports were associated with an increased risk of basal cell carcinoma, whereas skiing has been shown to be at increased risk for squamous cell carcinoma,” according to a 2008 report on outdoor sports and skin cancer (Clin Dermatol. 2008 Jan-Feb;26[1]:12-5).
“Risk factors of cutaneous melanoma, such as the number of melanocytic nevi and solar lentigines, have been found to be more frequent in subjects practicing endurance outdoor sports. An increased risk for cutaneous melanoma may be assumed for these athletes,” Dr. Adams commented.
Another study, this one published in 2006, found more atypical melanocytic nevi, solar lentigines, and lesions suggestive of nonmelanoma skin cancer in marathon runners, compared with a control group, and the risk was associated with the level of training intensity. The control subjects were more sensitive to the sun and had more common melanocytic nevi (Arch Dermatol. 2006 Nov;142[11]:1471-4).
Counseling about sunscreen may actually work
One strategy to reduce sun exposure is to advise athletes to avoid peak sun hours. However, “the key to caring for the athletes is not only recognizing that their sport may play a role in their disease but also realizing that your therapeutic approach must be tailored to minimize disruption to their practices and competitions,” Dr. Adams said.
However, there’s good news for dermatologists who are willing to push: The study also reported that athletes who were encouraged to use sunscreen were significantly more likely to use sunscreen (P less than .0001).
Watch for other conditions, from jogger’s nipple to ringworm
Dr. Adams offered advice about detection, treatment, and prevention of other skin disorders that affect athletes:
- “Jogger’s nipples” and other kinds of chafing. He has learned to recognize the “red eleven” – two vertical streaks of blood on a runner’s shirt – that represent a case of “jogger’s nipples” caused by chafing. Antibacterial ointment or petroleum jelly are useful treatments, he said, and an application of plenty of petroleum jelly on the nipples prior to a run can be helpful. Cotton shirts should be avoided, he said, in favor of synthetic, moisture-wicking shirts and bras. Chafing can also occur in the underarms and inner thighs, he said, and the same treatments and preventive techniques are useful.
- Callused and bleeding “jogger’s toes.” This can strike runners, especially on the second toe, which is often the longest and most likely to strike the toe box of a shoe. Specialty shoes can help prevent this condition, he said.
- Tinea corporis (ringworm) and herpes gladiatorum. In wrestlers, ringworm is known as tinea corporis gladiatorum because the intensity of skin-to-skin contact in wrestling makes the condition especially common in these athletes. Lesions don’t develop as rings at first; instead, they first appear as relatively nonspecific red round lesions and are most likely to be found in the head, neck, and upper extremities, Dr. Adams noted. Herpes gladiatorum is caused by herpes simplex virus 1; it is also seen in wrestlers and caused by skin-to-skin contact. Topical and oral antifungals clear ringworm, while oral antiviral agents are appropriate for herpes gladiatorum, Dr. Adams said. While herpes gladiatorum clears up and is no longer contagious after 4-5 days, he said, it’s not clear how long wrestlers with ringworm should be disqualified from playing.
Dr. Adams disclosed advising Mission, a company that focuses on sunscreen designed by and for athletes.
EXPERT ANALYSIS FROM AAD 18
Sensitivity of vibration-based neuropathy detectors varies widely
Three vibration-based detection tools vary widely in their sensitivity for detection of diabetic peripheral neuropathy. Researchers are suggesting that physicians use more than a single vibration-based device when they check the sensitivity of their diabetic patients’ feet.
“This would significantly reduce the proportion of patients with diabetes who would be falsely identified as having no peripheral neuropathy and subsequently denied the benefit of beneficial and effective secondary risk factor control,” said study coauthor Cynthia Formosa, PhD, of the University of Malta, Msida, and Staffordshire University, Stoke-on-Trent, England, in an interview.
Researchers have estimated that 50% of patients with diabetes develop diabetic polyneuropathy over their lives, and that the condition is responsible for more than a quarter of all diabetes spending. “Despite a long history of research in this area, we are only starting to understand the pathophysiology of the disease,” reports a 2016 review of recent findings.
In a previous report, several of the authors of the new study analyzed 10 sets of guidelines regarding diabetic foot and found that some lacked information to back up the recommendations. Guideline limitations “are responsible for the current gaps between guidelines, standard clinical practice, and development of complications,” the researchers wrote (Rev Diabet Stud. 2016, 13[2-3]:158-86).
Most of the guidelines recommended the use of a 10-g monofilament test, in which a monofilament is placed against the skin, plus a vibration-based test, the researchers reported.
For the new study in Primary Care Diabetes, the researchers examined the efficacy of three noninvasive tools that are used to detect neuropathy in the foot: the 128-Hz tuning fork; the VibraTip, a pocket-sized, motorized device; and the neurothesiometer, a 7-pound electromechanical device that comes in a carrying case and includes an attachment that delivers vibrations to the body.
According to Dr. Formosa, the tuning fork is the most commonly used testing device in normal clinical practice.
Physicians such as endocrinologists and podiatrists use the vibrations produced by the devices to detect whether patients have normal levels of sensation in various parts of the foot and ankle. “Vibration testing is extremely important,” the study investigators wrote, “since in the initial stages of neuropathy, the vibratory sensory system is amongst the first component of the nervous system to be affected.”
However, research into the accuracy of the devices has been inconsistent, according to the investigators.
The researchers prospectively recruited 100 patients at primary health centers diagnosed with type 2 diabetes for at least 10 years. The subjects included 57 males and 43 females, with a mean age of 73 years (± 7.8 years). Practitioners tested the neuropathy detection tools on the patients and recorded whether they felt vibrations.
The study authors reported that 29% of patients didn’t perceive vibrations produced by the VibraTip, compared with 21% for the neurothesiometer and 12% for the 128-Hz tuning fork (P less than .001).
The VibraTip device is available for about $90 on Amazon. The cost of a neurothesiometer is listed by one medical supply company as $2,850. Prices for 128-Hz tuning forks vary on Amazon, with prices listed from $6 to $40.
The study researchers did not confirm neuropathy via more sophisticated neurologic testing, so it remains unclear which device is actually the most accurate at detecting cases of neuropathy.
Still, “the findings of this study suggest that use of only one screening tool to assess vibration perception is likely to yield high false negative results,” the researchers wrote. “We recommend that peripheral neuropathy in patients with diabetes should be assessed utilizing two or more modalities.”
Moving forward, Dr. Chockalingam said, the research team plans to launch “a structured clinical trial using a gold-standard tool such as nerve conduction testing to confirm which simple screening method best detects neuropathy.”
No study funding was reported. The authors reported no relevant disclosures.
SOURCE: Azzopardia K et al. Prim Care Diabetes. 2018 Apr;12(2):111-5.
Three vibration-based detection tools vary widely in their sensitivity for detection of diabetic peripheral neuropathy. Researchers are suggesting that physicians use more than a single vibration-based device when they check the sensitivity of their diabetic patients’ feet.
“This would significantly reduce the proportion of patients with diabetes who would be falsely identified as having no peripheral neuropathy and subsequently denied the benefit of beneficial and effective secondary risk factor control,” said study coauthor Cynthia Formosa, PhD, of the University of Malta, Msida, and Staffordshire University, Stoke-on-Trent, England, in an interview.
Researchers have estimated that 50% of patients with diabetes develop diabetic polyneuropathy over their lives, and that the condition is responsible for more than a quarter of all diabetes spending. “Despite a long history of research in this area, we are only starting to understand the pathophysiology of the disease,” reports a 2016 review of recent findings.
In a previous report, several of the authors of the new study analyzed 10 sets of guidelines regarding diabetic foot and found that some lacked information to back up the recommendations. Guideline limitations “are responsible for the current gaps between guidelines, standard clinical practice, and development of complications,” the researchers wrote (Rev Diabet Stud. 2016, 13[2-3]:158-86).
Most of the guidelines recommended the use of a 10-g monofilament test, in which a monofilament is placed against the skin, plus a vibration-based test, the researchers reported.
For the new study in Primary Care Diabetes, the researchers examined the efficacy of three noninvasive tools that are used to detect neuropathy in the foot: the 128-Hz tuning fork; the VibraTip, a pocket-sized, motorized device; and the neurothesiometer, a 7-pound electromechanical device that comes in a carrying case and includes an attachment that delivers vibrations to the body.
According to Dr. Formosa, the tuning fork is the most commonly used testing device in normal clinical practice.
Physicians such as endocrinologists and podiatrists use the vibrations produced by the devices to detect whether patients have normal levels of sensation in various parts of the foot and ankle. “Vibration testing is extremely important,” the study investigators wrote, “since in the initial stages of neuropathy, the vibratory sensory system is amongst the first component of the nervous system to be affected.”
However, research into the accuracy of the devices has been inconsistent, according to the investigators.
The researchers prospectively recruited 100 patients at primary health centers diagnosed with type 2 diabetes for at least 10 years. The subjects included 57 males and 43 females, with a mean age of 73 years (± 7.8 years). Practitioners tested the neuropathy detection tools on the patients and recorded whether they felt vibrations.
The study authors reported that 29% of patients didn’t perceive vibrations produced by the VibraTip, compared with 21% for the neurothesiometer and 12% for the 128-Hz tuning fork (P less than .001).
The VibraTip device is available for about $90 on Amazon. The cost of a neurothesiometer is listed by one medical supply company as $2,850. Prices for 128-Hz tuning forks vary on Amazon, with prices listed from $6 to $40.
The study researchers did not confirm neuropathy via more sophisticated neurologic testing, so it remains unclear which device is actually the most accurate at detecting cases of neuropathy.
Still, “the findings of this study suggest that use of only one screening tool to assess vibration perception is likely to yield high false negative results,” the researchers wrote. “We recommend that peripheral neuropathy in patients with diabetes should be assessed utilizing two or more modalities.”
Moving forward, Dr. Chockalingam said, the research team plans to launch “a structured clinical trial using a gold-standard tool such as nerve conduction testing to confirm which simple screening method best detects neuropathy.”
No study funding was reported. The authors reported no relevant disclosures.
SOURCE: Azzopardia K et al. Prim Care Diabetes. 2018 Apr;12(2):111-5.
Three vibration-based detection tools vary widely in their sensitivity for detection of diabetic peripheral neuropathy. Researchers are suggesting that physicians use more than a single vibration-based device when they check the sensitivity of their diabetic patients’ feet.
“This would significantly reduce the proportion of patients with diabetes who would be falsely identified as having no peripheral neuropathy and subsequently denied the benefit of beneficial and effective secondary risk factor control,” said study coauthor Cynthia Formosa, PhD, of the University of Malta, Msida, and Staffordshire University, Stoke-on-Trent, England, in an interview.
Researchers have estimated that 50% of patients with diabetes develop diabetic polyneuropathy over their lives, and that the condition is responsible for more than a quarter of all diabetes spending. “Despite a long history of research in this area, we are only starting to understand the pathophysiology of the disease,” reports a 2016 review of recent findings.
In a previous report, several of the authors of the new study analyzed 10 sets of guidelines regarding diabetic foot and found that some lacked information to back up the recommendations. Guideline limitations “are responsible for the current gaps between guidelines, standard clinical practice, and development of complications,” the researchers wrote (Rev Diabet Stud. 2016, 13[2-3]:158-86).
Most of the guidelines recommended the use of a 10-g monofilament test, in which a monofilament is placed against the skin, plus a vibration-based test, the researchers reported.
For the new study in Primary Care Diabetes, the researchers examined the efficacy of three noninvasive tools that are used to detect neuropathy in the foot: the 128-Hz tuning fork; the VibraTip, a pocket-sized, motorized device; and the neurothesiometer, a 7-pound electromechanical device that comes in a carrying case and includes an attachment that delivers vibrations to the body.
According to Dr. Formosa, the tuning fork is the most commonly used testing device in normal clinical practice.
Physicians such as endocrinologists and podiatrists use the vibrations produced by the devices to detect whether patients have normal levels of sensation in various parts of the foot and ankle. “Vibration testing is extremely important,” the study investigators wrote, “since in the initial stages of neuropathy, the vibratory sensory system is amongst the first component of the nervous system to be affected.”
However, research into the accuracy of the devices has been inconsistent, according to the investigators.
The researchers prospectively recruited 100 patients at primary health centers diagnosed with type 2 diabetes for at least 10 years. The subjects included 57 males and 43 females, with a mean age of 73 years (± 7.8 years). Practitioners tested the neuropathy detection tools on the patients and recorded whether they felt vibrations.
The study authors reported that 29% of patients didn’t perceive vibrations produced by the VibraTip, compared with 21% for the neurothesiometer and 12% for the 128-Hz tuning fork (P less than .001).
The VibraTip device is available for about $90 on Amazon. The cost of a neurothesiometer is listed by one medical supply company as $2,850. Prices for 128-Hz tuning forks vary on Amazon, with prices listed from $6 to $40.
The study researchers did not confirm neuropathy via more sophisticated neurologic testing, so it remains unclear which device is actually the most accurate at detecting cases of neuropathy.
Still, “the findings of this study suggest that use of only one screening tool to assess vibration perception is likely to yield high false negative results,” the researchers wrote. “We recommend that peripheral neuropathy in patients with diabetes should be assessed utilizing two or more modalities.”
Moving forward, Dr. Chockalingam said, the research team plans to launch “a structured clinical trial using a gold-standard tool such as nerve conduction testing to confirm which simple screening method best detects neuropathy.”
No study funding was reported. The authors reported no relevant disclosures.
SOURCE: Azzopardia K et al. Prim Care Diabetes. 2018 Apr;12(2):111-5.
FROM PRIMARY CARE DIABETES
Key clinical point: Results vary widely among three vibration-based devices used to detect peripheral neuropathy.
Major finding: 29% of patients didn’t perceive vibrations produced by the VibraTip to 21% for the neurothesiometer and 12% for the 128 Hz tuning fork (P less than .001).
Study details: A prospective, multicenter, cross-sectional study of 100 patients diagnosed with type 2 diabetes for at least 10 years.
Disclosures: No study funding was reported. The authors reported no relevant disclosures.
Source: Azzopardia K et al. Prim Care Diabetes. 2018 Apr;12(2):111-5.