User login
VIDEO: Teriflunomide and dimethyl fumarate are comparable in relapsing-remitting MS
SAN DIEGO – New industry-funded research finds that
“The fundamental conclusion is that there’s little to separate them. If you look at efficacy, safety, and MRI scanning, there were no differences between the two medications that matter. ... Both drugs can be used with equal confidence,” said Stanley Cohan, MD, PhD, medical director of Providence MS Center in Portland, Ore.
Dr. Cohan spoke in a video interview at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
The researchers analyzed retrospective data collected in the phase 4 Teri-RADAR study, which tracked two groups of 50 patients each at MS centers in the United States as they took 14 mg of teriflunomide (Aubagio) daily or 240 mg of delayed-release dimethyl fumarate (Tecfidera) twice daily.
In both groups, participants had an average age of about 49 years, about three-quarters were women, and about 93% were white. Twenty-two percent of those in the teriflunomide group had relapses in the 18 months before the index date, compared with 10% of the dimethyl fumarate group.
The study – funded by Sanofi, maker of teriflunomide – followed up with patients at a mean of 15-16 months.
Mean annualized percentage of brain volume change (PBVC) was lower in teriflunomide-treated patients, compared with those treated with dimethyl fumarate (–0.2% vs. –0.5%; P = .02). Also, fewer teriflunomide-treated patients showed signs of annualized PBVC above a mean annualized brain volume loss rate threshold of 0.4% (25.9% vs. 58.6%; P = .01).
As for adverse effects, rashes affected none of the teriflunomide group but 28% of the dimethyl fumarate group. The teriflunomide group experienced skin reactions (12%) and diarrhea (16%), while these adverse events were not reported in the dimethyl fumarate group.
The study authors reported a variety of disclosures, and several are employees of Sanofi. Dr. Cohan reported a variety of disclosures, including multiple relationships with Sanofi.
SAN DIEGO – New industry-funded research finds that
“The fundamental conclusion is that there’s little to separate them. If you look at efficacy, safety, and MRI scanning, there were no differences between the two medications that matter. ... Both drugs can be used with equal confidence,” said Stanley Cohan, MD, PhD, medical director of Providence MS Center in Portland, Ore.
Dr. Cohan spoke in a video interview at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
The researchers analyzed retrospective data collected in the phase 4 Teri-RADAR study, which tracked two groups of 50 patients each at MS centers in the United States as they took 14 mg of teriflunomide (Aubagio) daily or 240 mg of delayed-release dimethyl fumarate (Tecfidera) twice daily.
In both groups, participants had an average age of about 49 years, about three-quarters were women, and about 93% were white. Twenty-two percent of those in the teriflunomide group had relapses in the 18 months before the index date, compared with 10% of the dimethyl fumarate group.
The study – funded by Sanofi, maker of teriflunomide – followed up with patients at a mean of 15-16 months.
Mean annualized percentage of brain volume change (PBVC) was lower in teriflunomide-treated patients, compared with those treated with dimethyl fumarate (–0.2% vs. –0.5%; P = .02). Also, fewer teriflunomide-treated patients showed signs of annualized PBVC above a mean annualized brain volume loss rate threshold of 0.4% (25.9% vs. 58.6%; P = .01).
As for adverse effects, rashes affected none of the teriflunomide group but 28% of the dimethyl fumarate group. The teriflunomide group experienced skin reactions (12%) and diarrhea (16%), while these adverse events were not reported in the dimethyl fumarate group.
The study authors reported a variety of disclosures, and several are employees of Sanofi. Dr. Cohan reported a variety of disclosures, including multiple relationships with Sanofi.
SAN DIEGO – New industry-funded research finds that
“The fundamental conclusion is that there’s little to separate them. If you look at efficacy, safety, and MRI scanning, there were no differences between the two medications that matter. ... Both drugs can be used with equal confidence,” said Stanley Cohan, MD, PhD, medical director of Providence MS Center in Portland, Ore.
Dr. Cohan spoke in a video interview at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
The researchers analyzed retrospective data collected in the phase 4 Teri-RADAR study, which tracked two groups of 50 patients each at MS centers in the United States as they took 14 mg of teriflunomide (Aubagio) daily or 240 mg of delayed-release dimethyl fumarate (Tecfidera) twice daily.
In both groups, participants had an average age of about 49 years, about three-quarters were women, and about 93% were white. Twenty-two percent of those in the teriflunomide group had relapses in the 18 months before the index date, compared with 10% of the dimethyl fumarate group.
The study – funded by Sanofi, maker of teriflunomide – followed up with patients at a mean of 15-16 months.
Mean annualized percentage of brain volume change (PBVC) was lower in teriflunomide-treated patients, compared with those treated with dimethyl fumarate (–0.2% vs. –0.5%; P = .02). Also, fewer teriflunomide-treated patients showed signs of annualized PBVC above a mean annualized brain volume loss rate threshold of 0.4% (25.9% vs. 58.6%; P = .01).
As for adverse effects, rashes affected none of the teriflunomide group but 28% of the dimethyl fumarate group. The teriflunomide group experienced skin reactions (12%) and diarrhea (16%), while these adverse events were not reported in the dimethyl fumarate group.
The study authors reported a variety of disclosures, and several are employees of Sanofi. Dr. Cohan reported a variety of disclosures, including multiple relationships with Sanofi.
REPORTING FROM ACTRIMS Forum 2018
VIDEO: Advanced practice providers take on many roles in MS care
SAN DIEGO – A new survey suggests that advanced practice providers who care for multiple sclerosis patients are highly satisfied with their jobs, which encompass a wide range of responsibilities.
Researchers received survey responses from 215 nurse practitioners and 395 physician assistants who answered Web questionnaires in 2016 and 2017. Of those who care for multiple sclerosis (MS) patients, 92.5% and 77.8% respectively said they provide at least 9 of 11 services, such as direct care, supportive services, and care coordination.
“Nurse practitioners in particular are providing lots of different services from diagnosis to education to symptom management,” said Michael T. Halpern, MD, PhD, MPH, of Temple University, Philadelphia. “Physician assistants are also providing a diverse range of MS services, but not as frequently as nurse practitioners.”
Dr. Halpern was the presenting author of the study reporting the survey results. He spoke in a video interview at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis, where the study findings were presented.
Advanced practice providers also reported high levels of job satisfaction. About 80% in both groups reported being very or extremely satisfied with their careers and with their colleagues; 90% of nurse practitioners reported being very or extremely satisfied by their relationships with patients, as did 86% of physician assistants.
The providers “appear to really enjoy working with individuals with MS,” Dr. Halpern said. But he cautioned that there’s a need for additional training for these providers; some respondents said their lack of knowledge was a hindrance to care.
The study was funded by the National Multiple Sclerosis Society. Dr. Halpern reported no relevant disclosures.
SAN DIEGO – A new survey suggests that advanced practice providers who care for multiple sclerosis patients are highly satisfied with their jobs, which encompass a wide range of responsibilities.
Researchers received survey responses from 215 nurse practitioners and 395 physician assistants who answered Web questionnaires in 2016 and 2017. Of those who care for multiple sclerosis (MS) patients, 92.5% and 77.8% respectively said they provide at least 9 of 11 services, such as direct care, supportive services, and care coordination.
“Nurse practitioners in particular are providing lots of different services from diagnosis to education to symptom management,” said Michael T. Halpern, MD, PhD, MPH, of Temple University, Philadelphia. “Physician assistants are also providing a diverse range of MS services, but not as frequently as nurse practitioners.”
Dr. Halpern was the presenting author of the study reporting the survey results. He spoke in a video interview at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis, where the study findings were presented.
Advanced practice providers also reported high levels of job satisfaction. About 80% in both groups reported being very or extremely satisfied with their careers and with their colleagues; 90% of nurse practitioners reported being very or extremely satisfied by their relationships with patients, as did 86% of physician assistants.
The providers “appear to really enjoy working with individuals with MS,” Dr. Halpern said. But he cautioned that there’s a need for additional training for these providers; some respondents said their lack of knowledge was a hindrance to care.
The study was funded by the National Multiple Sclerosis Society. Dr. Halpern reported no relevant disclosures.
SAN DIEGO – A new survey suggests that advanced practice providers who care for multiple sclerosis patients are highly satisfied with their jobs, which encompass a wide range of responsibilities.
Researchers received survey responses from 215 nurse practitioners and 395 physician assistants who answered Web questionnaires in 2016 and 2017. Of those who care for multiple sclerosis (MS) patients, 92.5% and 77.8% respectively said they provide at least 9 of 11 services, such as direct care, supportive services, and care coordination.
“Nurse practitioners in particular are providing lots of different services from diagnosis to education to symptom management,” said Michael T. Halpern, MD, PhD, MPH, of Temple University, Philadelphia. “Physician assistants are also providing a diverse range of MS services, but not as frequently as nurse practitioners.”
Dr. Halpern was the presenting author of the study reporting the survey results. He spoke in a video interview at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis, where the study findings were presented.
Advanced practice providers also reported high levels of job satisfaction. About 80% in both groups reported being very or extremely satisfied with their careers and with their colleagues; 90% of nurse practitioners reported being very or extremely satisfied by their relationships with patients, as did 86% of physician assistants.
The providers “appear to really enjoy working with individuals with MS,” Dr. Halpern said. But he cautioned that there’s a need for additional training for these providers; some respondents said their lack of knowledge was a hindrance to care.
The study was funded by the National Multiple Sclerosis Society. Dr. Halpern reported no relevant disclosures.
REPORTING FROM ACTRIMS FORUM 2018
Trial of clozapine, risperidone halted in MS
SAN DIEGO – New Zealand researchers halted a small trial that was testing the use of the antipsychotics clozapine and risperidone to treat progressive multiple sclerosis because significant side effects caused participants to withdraw.
The adverse events appeared even though the doses were much smaller than those routinely given to patients with psychiatric illnesses. “The neurologists realized it was in the participants’ best interest to stop,” said study lead author Anne Camille La Flamme, PhD, of Victoria University of Wellington (New Zealand). “Adverse events included dizziness, muscle weakness, and falls.”
The researchers launched the study – a blinded, randomized, placebo-controlled trial – to learn whether the two antipsychotic drugs, also known by the brand names Clozaril and Risperdal, have potential as treatments for progressive multiple sclerosis.
Previous in-vitro research had linked the drugs to anti-inflammatory effects in the central nervous system, Dr. La Flamme said, and researchers believed that the progressive form of MS might be especially vulnerable to their effects because of high immune system involvement.
The researchers planned to randomly assign three groups of 12 patients per arm to placebo, clozapine, and risperidone.
For clozapine, “the doses were very low, much lower than you’d expect for psychiatric use,” Dr. La Flamme said. A typical dose for psychiatric disorders is about 350 mg/day, she said, and the trial aimed to use 100-150 mg/day with an eye toward preventing dose-dependent side effects.
As for risperidone, a typical dose is about 4 mg/day, and the trial began at 2 mg/day and would increase to 3.5 mg/day, she said.
Three subjects in the clozapine group had to withdraw within 2 weeks when their doses had only reached an average of 35 mg/day. Two of three in the risperidone group withdrew within 4 months.
In light of the adverse effects, “it was deemed not wise to continue,” Dr. La Flamme said.
The placebo group, meanwhile, completed the trial at 178 days and had adverse effects that were more indicative of MS, she said.
What happened? One possibility is that disability from MS made the adverse events more evident, Dr. La Flamme said. Another possible explanation is that the underlying MS physiology changed the targets of the medications, she said.
“We have no conclusive evidence that would suggest one over the other,” she said. “But a lot of the evidence supports the idea that it’s a change in the physiology, that something about those pathways has been altered.”
It’s clear, she said, that the doses of the drugs in the trial were not appropriate. However, a big question remains: “We do not know whether these medicines are effective at reducing neuroinflammation.”
It’s possible, she said, that a “whisper of a dose” could still be effective. “It may get back to how these agents metabolize and become an active form.”
The study was funded by New Zealand’s Ministry of Business, Innovation and Employment. Dr. La Flamme disclosed that the study team has a patent for repurposing of clozapine and risperidone to treat MS.
SOURCE: La Flamme A et al. ACTRIMS Forum 2018, abstract P031.
SAN DIEGO – New Zealand researchers halted a small trial that was testing the use of the antipsychotics clozapine and risperidone to treat progressive multiple sclerosis because significant side effects caused participants to withdraw.
The adverse events appeared even though the doses were much smaller than those routinely given to patients with psychiatric illnesses. “The neurologists realized it was in the participants’ best interest to stop,” said study lead author Anne Camille La Flamme, PhD, of Victoria University of Wellington (New Zealand). “Adverse events included dizziness, muscle weakness, and falls.”
The researchers launched the study – a blinded, randomized, placebo-controlled trial – to learn whether the two antipsychotic drugs, also known by the brand names Clozaril and Risperdal, have potential as treatments for progressive multiple sclerosis.
Previous in-vitro research had linked the drugs to anti-inflammatory effects in the central nervous system, Dr. La Flamme said, and researchers believed that the progressive form of MS might be especially vulnerable to their effects because of high immune system involvement.
The researchers planned to randomly assign three groups of 12 patients per arm to placebo, clozapine, and risperidone.
For clozapine, “the doses were very low, much lower than you’d expect for psychiatric use,” Dr. La Flamme said. A typical dose for psychiatric disorders is about 350 mg/day, she said, and the trial aimed to use 100-150 mg/day with an eye toward preventing dose-dependent side effects.
As for risperidone, a typical dose is about 4 mg/day, and the trial began at 2 mg/day and would increase to 3.5 mg/day, she said.
Three subjects in the clozapine group had to withdraw within 2 weeks when their doses had only reached an average of 35 mg/day. Two of three in the risperidone group withdrew within 4 months.
In light of the adverse effects, “it was deemed not wise to continue,” Dr. La Flamme said.
The placebo group, meanwhile, completed the trial at 178 days and had adverse effects that were more indicative of MS, she said.
What happened? One possibility is that disability from MS made the adverse events more evident, Dr. La Flamme said. Another possible explanation is that the underlying MS physiology changed the targets of the medications, she said.
“We have no conclusive evidence that would suggest one over the other,” she said. “But a lot of the evidence supports the idea that it’s a change in the physiology, that something about those pathways has been altered.”
It’s clear, she said, that the doses of the drugs in the trial were not appropriate. However, a big question remains: “We do not know whether these medicines are effective at reducing neuroinflammation.”
It’s possible, she said, that a “whisper of a dose” could still be effective. “It may get back to how these agents metabolize and become an active form.”
The study was funded by New Zealand’s Ministry of Business, Innovation and Employment. Dr. La Flamme disclosed that the study team has a patent for repurposing of clozapine and risperidone to treat MS.
SOURCE: La Flamme A et al. ACTRIMS Forum 2018, abstract P031.
SAN DIEGO – New Zealand researchers halted a small trial that was testing the use of the antipsychotics clozapine and risperidone to treat progressive multiple sclerosis because significant side effects caused participants to withdraw.
The adverse events appeared even though the doses were much smaller than those routinely given to patients with psychiatric illnesses. “The neurologists realized it was in the participants’ best interest to stop,” said study lead author Anne Camille La Flamme, PhD, of Victoria University of Wellington (New Zealand). “Adverse events included dizziness, muscle weakness, and falls.”
The researchers launched the study – a blinded, randomized, placebo-controlled trial – to learn whether the two antipsychotic drugs, also known by the brand names Clozaril and Risperdal, have potential as treatments for progressive multiple sclerosis.
Previous in-vitro research had linked the drugs to anti-inflammatory effects in the central nervous system, Dr. La Flamme said, and researchers believed that the progressive form of MS might be especially vulnerable to their effects because of high immune system involvement.
The researchers planned to randomly assign three groups of 12 patients per arm to placebo, clozapine, and risperidone.
For clozapine, “the doses were very low, much lower than you’d expect for psychiatric use,” Dr. La Flamme said. A typical dose for psychiatric disorders is about 350 mg/day, she said, and the trial aimed to use 100-150 mg/day with an eye toward preventing dose-dependent side effects.
As for risperidone, a typical dose is about 4 mg/day, and the trial began at 2 mg/day and would increase to 3.5 mg/day, she said.
Three subjects in the clozapine group had to withdraw within 2 weeks when their doses had only reached an average of 35 mg/day. Two of three in the risperidone group withdrew within 4 months.
In light of the adverse effects, “it was deemed not wise to continue,” Dr. La Flamme said.
The placebo group, meanwhile, completed the trial at 178 days and had adverse effects that were more indicative of MS, she said.
What happened? One possibility is that disability from MS made the adverse events more evident, Dr. La Flamme said. Another possible explanation is that the underlying MS physiology changed the targets of the medications, she said.
“We have no conclusive evidence that would suggest one over the other,” she said. “But a lot of the evidence supports the idea that it’s a change in the physiology, that something about those pathways has been altered.”
It’s clear, she said, that the doses of the drugs in the trial were not appropriate. However, a big question remains: “We do not know whether these medicines are effective at reducing neuroinflammation.”
It’s possible, she said, that a “whisper of a dose” could still be effective. “It may get back to how these agents metabolize and become an active form.”
The study was funded by New Zealand’s Ministry of Business, Innovation and Employment. Dr. La Flamme disclosed that the study team has a patent for repurposing of clozapine and risperidone to treat MS.
SOURCE: La Flamme A et al. ACTRIMS Forum 2018, abstract P031.
REPORTING FROM ACTRIMS Forum 2018
VIDEO: Oral ozanimod shows promise for relapsing MS
SAN DIEGO – A pair of phase 3 studies offer promising results regarding the safety and efficacy of ozanimod, an experimental immunomodulator, in the treatment of relapsing multiple sclerosis (RMS).
The medication targets sphingosine 1-phosphate 1 and 5 receptors. Industry-funded researchers tested it in two studies against interferon beta-1a.
One study, called SUNBEAM, tested once-daily oral ozanimod (1 mg or 0.5 mg with 7-day dose escalation) against interferon beta-1a (via 30 mcg weekly intramuscular injection) for at least 12 months in 1,346 patients with RMS. The annualized relapse rate, the primary endpoint, was lower in the ozanimod groups versus interferon. For the 1-mg dose, it was 0.181 (P less than .0001), and for 0.5-mg dose, 0.241 (P = .0013).
The number of serious treatment-emergent adverse events in the three groups was low, ranging from 2.5% to 3.5%.
The other study, called RADIANCE, was a similar trial that lasted 24 months. In it, the rate of serious treatment-emergent adverse events in the three groups were similar, ranging from 6.4% to 7.1%.
Ozanimod offers “an excellent therapeutic benefit for patients and a very clean safety profile,” said Bruce Cree, MD, PhD, clinical research director at the University of California, San Francisco, Multiple Sclerosis Center. He is an author on both studies and spoke in a video interview at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
He said the ozanimod should be especially useful as a first-line treatment for MS. The drug is currently being evaluated by the Food and Drug Administration for an RMS indication, and it is also being developed for Crohn’s disease and ulcerative colitis, he said.
The study was funded by Receptos, a wholly owned subsidiary of Celgene. Dr. Cree reported that he has been a consultant to AbbVie, Biogen, EMD Serono, Genzyme, Novartis, and Shire.
SOURCE: Cree B et al. ACTRIMS Forum 2018, abstract P030, and Comi G et al. ACTRIMS Forum 2018, abstract P023
SAN DIEGO – A pair of phase 3 studies offer promising results regarding the safety and efficacy of ozanimod, an experimental immunomodulator, in the treatment of relapsing multiple sclerosis (RMS).
The medication targets sphingosine 1-phosphate 1 and 5 receptors. Industry-funded researchers tested it in two studies against interferon beta-1a.
One study, called SUNBEAM, tested once-daily oral ozanimod (1 mg or 0.5 mg with 7-day dose escalation) against interferon beta-1a (via 30 mcg weekly intramuscular injection) for at least 12 months in 1,346 patients with RMS. The annualized relapse rate, the primary endpoint, was lower in the ozanimod groups versus interferon. For the 1-mg dose, it was 0.181 (P less than .0001), and for 0.5-mg dose, 0.241 (P = .0013).
The number of serious treatment-emergent adverse events in the three groups was low, ranging from 2.5% to 3.5%.
The other study, called RADIANCE, was a similar trial that lasted 24 months. In it, the rate of serious treatment-emergent adverse events in the three groups were similar, ranging from 6.4% to 7.1%.
Ozanimod offers “an excellent therapeutic benefit for patients and a very clean safety profile,” said Bruce Cree, MD, PhD, clinical research director at the University of California, San Francisco, Multiple Sclerosis Center. He is an author on both studies and spoke in a video interview at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
He said the ozanimod should be especially useful as a first-line treatment for MS. The drug is currently being evaluated by the Food and Drug Administration for an RMS indication, and it is also being developed for Crohn’s disease and ulcerative colitis, he said.
The study was funded by Receptos, a wholly owned subsidiary of Celgene. Dr. Cree reported that he has been a consultant to AbbVie, Biogen, EMD Serono, Genzyme, Novartis, and Shire.
SOURCE: Cree B et al. ACTRIMS Forum 2018, abstract P030, and Comi G et al. ACTRIMS Forum 2018, abstract P023
SAN DIEGO – A pair of phase 3 studies offer promising results regarding the safety and efficacy of ozanimod, an experimental immunomodulator, in the treatment of relapsing multiple sclerosis (RMS).
The medication targets sphingosine 1-phosphate 1 and 5 receptors. Industry-funded researchers tested it in two studies against interferon beta-1a.
One study, called SUNBEAM, tested once-daily oral ozanimod (1 mg or 0.5 mg with 7-day dose escalation) against interferon beta-1a (via 30 mcg weekly intramuscular injection) for at least 12 months in 1,346 patients with RMS. The annualized relapse rate, the primary endpoint, was lower in the ozanimod groups versus interferon. For the 1-mg dose, it was 0.181 (P less than .0001), and for 0.5-mg dose, 0.241 (P = .0013).
The number of serious treatment-emergent adverse events in the three groups was low, ranging from 2.5% to 3.5%.
The other study, called RADIANCE, was a similar trial that lasted 24 months. In it, the rate of serious treatment-emergent adverse events in the three groups were similar, ranging from 6.4% to 7.1%.
Ozanimod offers “an excellent therapeutic benefit for patients and a very clean safety profile,” said Bruce Cree, MD, PhD, clinical research director at the University of California, San Francisco, Multiple Sclerosis Center. He is an author on both studies and spoke in a video interview at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
He said the ozanimod should be especially useful as a first-line treatment for MS. The drug is currently being evaluated by the Food and Drug Administration for an RMS indication, and it is also being developed for Crohn’s disease and ulcerative colitis, he said.
The study was funded by Receptos, a wholly owned subsidiary of Celgene. Dr. Cree reported that he has been a consultant to AbbVie, Biogen, EMD Serono, Genzyme, Novartis, and Shire.
SOURCE: Cree B et al. ACTRIMS Forum 2018, abstract P030, and Comi G et al. ACTRIMS Forum 2018, abstract P023
REPORTING FROM ACTRIMS FORUM 2018
MS may be a transmissible protein misfolding disorder, study suggests
SAN DIEGO – MS may even be caused by prions, potentially putting it into the same category as Creutzfeldt-Jakob disease.
Researchers don’t think MS is contagious between humans. But their findings in mice do suggest that the disease is transmissible from “a seed of protein misfolding,” Shigeki Tsutsui, DVM, PhD, of the University of Calgary (Alta.), said in an interview in advance of his presentation at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
“The next question is: ‘What is actually causing primary damage?’ ” he said. “Our hypothesis is that it’s protein misfolding. If protein misfolding targets the central neuron cells, then those damaged cells release a kind of a trigger to start an immune response.”
This isn’t an unusual concept. Protein misfolding is believed to cause several chronic neurodegenerative diseases, including Parkinson’s, Alzheimer’s, and amyotrophic lateral sclerosis, Dr. Tsutsui said.
The researchers focused on the potential transmissibility of human prion protein “since we thought that the prion protein might be the candidate causing the MS,” he said.
Prion diseases are extremely rare and often deadly. They appear when normal cellular prion proteins are induced to misfold when they come in contact with infectious agents known as prions.
In humans, prion diseases include Creutzfeldt-Jakob disease, fatal familial insomnia, and kuru (a condition spread among cannibals who eat the brains of other humans). Most famously, a variant of Creutzfeldt-Jakob disease has been linked – but not conclusively – to the eating of meat from cows infected with mad cow disease.
Depending on the condition, prion diseases can appear spontaneously, via inheritance or through infection.
In the new study, researchers intracerebrally injected brain homogenate from 10 patients with primary or secondary-progressive MS into 36 transgenic mice. All the mice over-expressed human prion protein. A control group of 23 mice were injected with homogenate from six donor brains.
At 6-12 months, the control mice did not develop pathology. However, the mice injected with brain matter from MS patients developed various levels of significant pathology. The researchers also managed to transmit illness from affected mice into another group of transgenic mice that over-expressed human prion protein via injection of brain homogenate.
While the mice in the study represent “a very extreme case” since their levels of prion protein expression were very high, it’s clear that “MS is actually transmissible,” Dr. Tsutsui said.
The study authors speculate that pathogenic prion protein triggers an autoimmune response by degenerating myelin and causing the release of cellular debris.
Could MS also be contagious? It may not be, even if it’s a prion disease, Dr. Tsutsui said. Some prion disease variants are neither contagious nor infectious.
The researchers plan to study the animal model they’ve created and explore the potential for its use in research, Dr. Tsutsui said.
The authors have no disclosures with the exception of one who disclosed serving as deputy editor of the Multiple Sclerosis Journal. Funding sources include Canada Research Chairs, Alberta Innovates-Health Solutions, and the Alberta Prion Research Institute.
SOURCE: Tsutsui S et al. ACTRIMS Forum 2018 Abstract LB282.
SAN DIEGO – MS may even be caused by prions, potentially putting it into the same category as Creutzfeldt-Jakob disease.
Researchers don’t think MS is contagious between humans. But their findings in mice do suggest that the disease is transmissible from “a seed of protein misfolding,” Shigeki Tsutsui, DVM, PhD, of the University of Calgary (Alta.), said in an interview in advance of his presentation at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
“The next question is: ‘What is actually causing primary damage?’ ” he said. “Our hypothesis is that it’s protein misfolding. If protein misfolding targets the central neuron cells, then those damaged cells release a kind of a trigger to start an immune response.”
This isn’t an unusual concept. Protein misfolding is believed to cause several chronic neurodegenerative diseases, including Parkinson’s, Alzheimer’s, and amyotrophic lateral sclerosis, Dr. Tsutsui said.
The researchers focused on the potential transmissibility of human prion protein “since we thought that the prion protein might be the candidate causing the MS,” he said.
Prion diseases are extremely rare and often deadly. They appear when normal cellular prion proteins are induced to misfold when they come in contact with infectious agents known as prions.
In humans, prion diseases include Creutzfeldt-Jakob disease, fatal familial insomnia, and kuru (a condition spread among cannibals who eat the brains of other humans). Most famously, a variant of Creutzfeldt-Jakob disease has been linked – but not conclusively – to the eating of meat from cows infected with mad cow disease.
Depending on the condition, prion diseases can appear spontaneously, via inheritance or through infection.
In the new study, researchers intracerebrally injected brain homogenate from 10 patients with primary or secondary-progressive MS into 36 transgenic mice. All the mice over-expressed human prion protein. A control group of 23 mice were injected with homogenate from six donor brains.
At 6-12 months, the control mice did not develop pathology. However, the mice injected with brain matter from MS patients developed various levels of significant pathology. The researchers also managed to transmit illness from affected mice into another group of transgenic mice that over-expressed human prion protein via injection of brain homogenate.
While the mice in the study represent “a very extreme case” since their levels of prion protein expression were very high, it’s clear that “MS is actually transmissible,” Dr. Tsutsui said.
The study authors speculate that pathogenic prion protein triggers an autoimmune response by degenerating myelin and causing the release of cellular debris.
Could MS also be contagious? It may not be, even if it’s a prion disease, Dr. Tsutsui said. Some prion disease variants are neither contagious nor infectious.
The researchers plan to study the animal model they’ve created and explore the potential for its use in research, Dr. Tsutsui said.
The authors have no disclosures with the exception of one who disclosed serving as deputy editor of the Multiple Sclerosis Journal. Funding sources include Canada Research Chairs, Alberta Innovates-Health Solutions, and the Alberta Prion Research Institute.
SOURCE: Tsutsui S et al. ACTRIMS Forum 2018 Abstract LB282.
SAN DIEGO – MS may even be caused by prions, potentially putting it into the same category as Creutzfeldt-Jakob disease.
Researchers don’t think MS is contagious between humans. But their findings in mice do suggest that the disease is transmissible from “a seed of protein misfolding,” Shigeki Tsutsui, DVM, PhD, of the University of Calgary (Alta.), said in an interview in advance of his presentation at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
“The next question is: ‘What is actually causing primary damage?’ ” he said. “Our hypothesis is that it’s protein misfolding. If protein misfolding targets the central neuron cells, then those damaged cells release a kind of a trigger to start an immune response.”
This isn’t an unusual concept. Protein misfolding is believed to cause several chronic neurodegenerative diseases, including Parkinson’s, Alzheimer’s, and amyotrophic lateral sclerosis, Dr. Tsutsui said.
The researchers focused on the potential transmissibility of human prion protein “since we thought that the prion protein might be the candidate causing the MS,” he said.
Prion diseases are extremely rare and often deadly. They appear when normal cellular prion proteins are induced to misfold when they come in contact with infectious agents known as prions.
In humans, prion diseases include Creutzfeldt-Jakob disease, fatal familial insomnia, and kuru (a condition spread among cannibals who eat the brains of other humans). Most famously, a variant of Creutzfeldt-Jakob disease has been linked – but not conclusively – to the eating of meat from cows infected with mad cow disease.
Depending on the condition, prion diseases can appear spontaneously, via inheritance or through infection.
In the new study, researchers intracerebrally injected brain homogenate from 10 patients with primary or secondary-progressive MS into 36 transgenic mice. All the mice over-expressed human prion protein. A control group of 23 mice were injected with homogenate from six donor brains.
At 6-12 months, the control mice did not develop pathology. However, the mice injected with brain matter from MS patients developed various levels of significant pathology. The researchers also managed to transmit illness from affected mice into another group of transgenic mice that over-expressed human prion protein via injection of brain homogenate.
While the mice in the study represent “a very extreme case” since their levels of prion protein expression were very high, it’s clear that “MS is actually transmissible,” Dr. Tsutsui said.
The study authors speculate that pathogenic prion protein triggers an autoimmune response by degenerating myelin and causing the release of cellular debris.
Could MS also be contagious? It may not be, even if it’s a prion disease, Dr. Tsutsui said. Some prion disease variants are neither contagious nor infectious.
The researchers plan to study the animal model they’ve created and explore the potential for its use in research, Dr. Tsutsui said.
The authors have no disclosures with the exception of one who disclosed serving as deputy editor of the Multiple Sclerosis Journal. Funding sources include Canada Research Chairs, Alberta Innovates-Health Solutions, and the Alberta Prion Research Institute.
SOURCE: Tsutsui S et al. ACTRIMS Forum 2018 Abstract LB282.
REPORTING FROM ACTRIMS FORUM 2018
Key clinical point: Translational research suggests the primary damage in MS may be caused by transmissible protein misfolding.
Major finding: Transgenic mice over-expressing human prion protein that were injected with brain matter from MS patients developed various levels of significant pathology, and brain homogenate from these mice could transmit illness to naive transgenic mice.
Study details: Transgenic mice over-expressing human prion protein received intracerebral injection of brain homogenate from patients with primary or secondary-progressive MS
Disclosures: The authors have no disclosures with the exception of one who disclosed serving as deputy editor of the Multiple Sclerosis Journal. Funding sources include Canada Research Chairs, Alberta Innovates-Health Solutions, and the Alberta Prion Research Institute.
Source: Tsutsui S et al. ACTRIMS Forum 2018 Abstract LB282.
Rituximab fails to eliminate meningeal inflammation in progressive MS
Researchers are heading back to the drawing board after a tiny phase 1 trial of intrathecal rituximab in certain patients with progressive multiple sclerosis failed to eliminate inflammation in the meninges.
The study investigators had hoped the treatment would significantly help progressive MS patients with leptomeningeal inflammation, which has been linked to worsening disease. However, “this paradigm seems to have not gotten rid of the meningeal inflammation significantly. We need a better approach,” said Pavan Bhargava, MD, of Johns Hopkins University, Baltimore, who spoke in an interview in advance of presenting the study findings at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
The new study aimed to test the value of targeting immune cell follicles with doses of rituximab given intrathecally – straight into the spinal fluid – to greatly boost the penetration of the drug into the meninges. When given intravenously, Dr. Bhargava said, only a minuscule amount of rituximab makes it to the brain.
Dr. Bhargava’s team screened 36 patients with progressive MS with MRI scans and found that 15 showed signs of meningeal inflammation. Of those, 11 agreed to take part in the study, and 8 fit the criteria.
The participants (median age 55.5 years; five were female) received two intrathecal treatments of 25 mg of rituximab 2 weeks apart, were monitored via follow-up clinical evaluations and lab tests at weeks 2, 8, 24, and 48, and received lumbar punctures and MRI scans at weeks 8 and 24.
“The main outcome was safety,” he said. “Most patients tolerated the rituximab, and the only side effect occurred in one patient who had abnormal sensations in the leg for 35-45 minutes. Otherwise, the medication was safe.”
The researchers detected no change in the meningeal inflammatory lesions before and after the treatment at 24 weeks.
But the tiny dose, much smaller than the typical two doses of 1,000 mg of IV rituximab over 2 weeks, completely depleted B cells in the blood of almost all patients, Dr. Bhargava said. “It was likely leaking out into the blood and clearing the B cells.” Peripheral blood B cells decreased from a median of 18.55% at baseline to 0.1% at week 2 and 8 with an increase to 3.6% at 24 weeks.
This effect was not as robust in spinal fluid, he said, where B cells were not fully depleted.
The findings suggest more cycles of treatment may be needed to target types of immune cells other than B cells, he noted. Or perhaps another drug would work better if it has a different mechanism and can penetrate more effectively.
The next step, he said, is to screen alternative drug approaches in animal models with meningeal inflammation that’s similar to that found in humans.
The study was funded by the International Progressive MS Alliance and the Race to Erase MS. Dr. Bhargava reported no relevant disclosures.
SOURCE: Bhargava P et al. ACTRIMS Forum 2018 Abstract P027.
Researchers are heading back to the drawing board after a tiny phase 1 trial of intrathecal rituximab in certain patients with progressive multiple sclerosis failed to eliminate inflammation in the meninges.
The study investigators had hoped the treatment would significantly help progressive MS patients with leptomeningeal inflammation, which has been linked to worsening disease. However, “this paradigm seems to have not gotten rid of the meningeal inflammation significantly. We need a better approach,” said Pavan Bhargava, MD, of Johns Hopkins University, Baltimore, who spoke in an interview in advance of presenting the study findings at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
The new study aimed to test the value of targeting immune cell follicles with doses of rituximab given intrathecally – straight into the spinal fluid – to greatly boost the penetration of the drug into the meninges. When given intravenously, Dr. Bhargava said, only a minuscule amount of rituximab makes it to the brain.
Dr. Bhargava’s team screened 36 patients with progressive MS with MRI scans and found that 15 showed signs of meningeal inflammation. Of those, 11 agreed to take part in the study, and 8 fit the criteria.
The participants (median age 55.5 years; five were female) received two intrathecal treatments of 25 mg of rituximab 2 weeks apart, were monitored via follow-up clinical evaluations and lab tests at weeks 2, 8, 24, and 48, and received lumbar punctures and MRI scans at weeks 8 and 24.
“The main outcome was safety,” he said. “Most patients tolerated the rituximab, and the only side effect occurred in one patient who had abnormal sensations in the leg for 35-45 minutes. Otherwise, the medication was safe.”
The researchers detected no change in the meningeal inflammatory lesions before and after the treatment at 24 weeks.
But the tiny dose, much smaller than the typical two doses of 1,000 mg of IV rituximab over 2 weeks, completely depleted B cells in the blood of almost all patients, Dr. Bhargava said. “It was likely leaking out into the blood and clearing the B cells.” Peripheral blood B cells decreased from a median of 18.55% at baseline to 0.1% at week 2 and 8 with an increase to 3.6% at 24 weeks.
This effect was not as robust in spinal fluid, he said, where B cells were not fully depleted.
The findings suggest more cycles of treatment may be needed to target types of immune cells other than B cells, he noted. Or perhaps another drug would work better if it has a different mechanism and can penetrate more effectively.
The next step, he said, is to screen alternative drug approaches in animal models with meningeal inflammation that’s similar to that found in humans.
The study was funded by the International Progressive MS Alliance and the Race to Erase MS. Dr. Bhargava reported no relevant disclosures.
SOURCE: Bhargava P et al. ACTRIMS Forum 2018 Abstract P027.
Researchers are heading back to the drawing board after a tiny phase 1 trial of intrathecal rituximab in certain patients with progressive multiple sclerosis failed to eliminate inflammation in the meninges.
The study investigators had hoped the treatment would significantly help progressive MS patients with leptomeningeal inflammation, which has been linked to worsening disease. However, “this paradigm seems to have not gotten rid of the meningeal inflammation significantly. We need a better approach,” said Pavan Bhargava, MD, of Johns Hopkins University, Baltimore, who spoke in an interview in advance of presenting the study findings at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
The new study aimed to test the value of targeting immune cell follicles with doses of rituximab given intrathecally – straight into the spinal fluid – to greatly boost the penetration of the drug into the meninges. When given intravenously, Dr. Bhargava said, only a minuscule amount of rituximab makes it to the brain.
Dr. Bhargava’s team screened 36 patients with progressive MS with MRI scans and found that 15 showed signs of meningeal inflammation. Of those, 11 agreed to take part in the study, and 8 fit the criteria.
The participants (median age 55.5 years; five were female) received two intrathecal treatments of 25 mg of rituximab 2 weeks apart, were monitored via follow-up clinical evaluations and lab tests at weeks 2, 8, 24, and 48, and received lumbar punctures and MRI scans at weeks 8 and 24.
“The main outcome was safety,” he said. “Most patients tolerated the rituximab, and the only side effect occurred in one patient who had abnormal sensations in the leg for 35-45 minutes. Otherwise, the medication was safe.”
The researchers detected no change in the meningeal inflammatory lesions before and after the treatment at 24 weeks.
But the tiny dose, much smaller than the typical two doses of 1,000 mg of IV rituximab over 2 weeks, completely depleted B cells in the blood of almost all patients, Dr. Bhargava said. “It was likely leaking out into the blood and clearing the B cells.” Peripheral blood B cells decreased from a median of 18.55% at baseline to 0.1% at week 2 and 8 with an increase to 3.6% at 24 weeks.
This effect was not as robust in spinal fluid, he said, where B cells were not fully depleted.
The findings suggest more cycles of treatment may be needed to target types of immune cells other than B cells, he noted. Or perhaps another drug would work better if it has a different mechanism and can penetrate more effectively.
The next step, he said, is to screen alternative drug approaches in animal models with meningeal inflammation that’s similar to that found in humans.
The study was funded by the International Progressive MS Alliance and the Race to Erase MS. Dr. Bhargava reported no relevant disclosures.
SOURCE: Bhargava P et al. ACTRIMS Forum 2018 Abstract P027.
FROM ACTRIMS FORUM 2018
Key clinical point:
Major finding: No change in leptomeningeal inflammation was seen after two intrathecal doses of 25 mg of rituximab administered over 2 weeks.
Data source: Prospective study of eight patients with progressive MS and signs of meningeal inflammation.
Disclosures: The study was funded by the International Progressive MS Alliance and the Race to Erase MS. Study presenter Pavan Bhargava, MD, reported no relevant disclosures.
Source: Bhargava P et al. ACTRIMS Forum 2018 Abstract P027.
Barancik Prize winner to discuss MS biology advances
Two decades ago, multiple sclerosis researchers like the University of Cambridge’s, Robin Franklin, PhD, understood the crucial role of regeneration of the myelin coating on nerve fibers, and they knew the process went awry in MS patients. But, he recalled, “we knew very little about how remyelination came about and the processes that orchestrated it.”
Now, thanks in part to the work of Dr. Franklin’s team, research into remyelination is making tremendous advances. “We’ve begun to understand the mechanisms that are operating, and that is fundamental to devising new therapies,” he said in an interview. “You don’t develop effective new therapies unless you understand the underlying biology. Now, we’re on the brink of that biology leading to regenerative medicine in MS.”
Dr. Franklin is the winner of the international prize, which recognizes exceptional MS research and is administered by the National MS Society.
In recent years, Dr. Franklin and his colleagues have published multiple studies that reveal new details about the workings of myelin regeneration, especially the biology by which oligodendrocyte progenitor cells become new oligodendrocytes.
“There are simple steps in that process,” Dr. Franklin said. “What we’ve been able to do is decorate that simple model with a whole host of mechanisms that allow remyelination to take place.”
In 2011, Franklin and his colleagues published a study in rodents identifying the RXRg gene as a key promoter of remyelination, making it a potential target for therapy (Nat Neurosci. 2011;14:45-53).
“There’s still a lot more to be learned,” Dr. Franklin said, “but we’ve learned a sufficient amount that we can engage in a meaningful way with the pharmaceutical industry and MS clinicians to make regenerative medicines an effective component of the MS therapeutic armory.”
Dr. Franklin has no relevant disclosures.
Two decades ago, multiple sclerosis researchers like the University of Cambridge’s, Robin Franklin, PhD, understood the crucial role of regeneration of the myelin coating on nerve fibers, and they knew the process went awry in MS patients. But, he recalled, “we knew very little about how remyelination came about and the processes that orchestrated it.”
Now, thanks in part to the work of Dr. Franklin’s team, research into remyelination is making tremendous advances. “We’ve begun to understand the mechanisms that are operating, and that is fundamental to devising new therapies,” he said in an interview. “You don’t develop effective new therapies unless you understand the underlying biology. Now, we’re on the brink of that biology leading to regenerative medicine in MS.”
Dr. Franklin is the winner of the international prize, which recognizes exceptional MS research and is administered by the National MS Society.
In recent years, Dr. Franklin and his colleagues have published multiple studies that reveal new details about the workings of myelin regeneration, especially the biology by which oligodendrocyte progenitor cells become new oligodendrocytes.
“There are simple steps in that process,” Dr. Franklin said. “What we’ve been able to do is decorate that simple model with a whole host of mechanisms that allow remyelination to take place.”
In 2011, Franklin and his colleagues published a study in rodents identifying the RXRg gene as a key promoter of remyelination, making it a potential target for therapy (Nat Neurosci. 2011;14:45-53).
“There’s still a lot more to be learned,” Dr. Franklin said, “but we’ve learned a sufficient amount that we can engage in a meaningful way with the pharmaceutical industry and MS clinicians to make regenerative medicines an effective component of the MS therapeutic armory.”
Dr. Franklin has no relevant disclosures.
Two decades ago, multiple sclerosis researchers like the University of Cambridge’s, Robin Franklin, PhD, understood the crucial role of regeneration of the myelin coating on nerve fibers, and they knew the process went awry in MS patients. But, he recalled, “we knew very little about how remyelination came about and the processes that orchestrated it.”
Now, thanks in part to the work of Dr. Franklin’s team, research into remyelination is making tremendous advances. “We’ve begun to understand the mechanisms that are operating, and that is fundamental to devising new therapies,” he said in an interview. “You don’t develop effective new therapies unless you understand the underlying biology. Now, we’re on the brink of that biology leading to regenerative medicine in MS.”
Dr. Franklin is the winner of the international prize, which recognizes exceptional MS research and is administered by the National MS Society.
In recent years, Dr. Franklin and his colleagues have published multiple studies that reveal new details about the workings of myelin regeneration, especially the biology by which oligodendrocyte progenitor cells become new oligodendrocytes.
“There are simple steps in that process,” Dr. Franklin said. “What we’ve been able to do is decorate that simple model with a whole host of mechanisms that allow remyelination to take place.”
In 2011, Franklin and his colleagues published a study in rodents identifying the RXRg gene as a key promoter of remyelination, making it a potential target for therapy (Nat Neurosci. 2011;14:45-53).
“There’s still a lot more to be learned,” Dr. Franklin said, “but we’ve learned a sufficient amount that we can engage in a meaningful way with the pharmaceutical industry and MS clinicians to make regenerative medicines an effective component of the MS therapeutic armory.”
Dr. Franklin has no relevant disclosures.
FROM ACTRIMS FORUM 2018
Researcher’s talk will tackle aging and gut bacteria in MS
Germs, genes, and aging each play a role in the development of multiple sclerosis (MS), but researchers are still figuring out how they’re connected. Now, a speaker at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis will shed light on recent findings that offer new insight into a specific type of germ – gut bacteria.
Suhayl Dhib-Jalbut, MD, ACTRIMS’s former president, will highlight his team’s investigation into the links between aging and alterations in gut microbiota in MS during the Kenneth P. Johnson Memorial Lecture on Feb. 1 at the meeting in San Diego.
Dr. Dhib-Jalbut’s presentation will focus on findings of a study by his team that was published late last year in Proceedings of the National Academy of Sciences of the U.S.A. (Proc Natl Acad Sci U S A. 2017 Oct 31;114[44]:E9318-27).
As the study explains, Dr. Dhib-Jalbut and colleagues triggered spontaneous experimental autoimmune encephalomyelitis (EAE) in transgenic mice by disrupting gut bacteria during adolescence and early young adulthood. But the process of aging in the mice past early young adulthood suppressed EAE onset by boosting immunologic tolerance.
The findings in this animal model offer insight into why the incidence of MS peaks at ages 20-40 years in humans, he said. “The implication is that one can perhaps manipulate gut bacteria with antibiotics or other treatments to impact the course of MS.”
Germs, genes, and aging each play a role in the development of multiple sclerosis (MS), but researchers are still figuring out how they’re connected. Now, a speaker at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis will shed light on recent findings that offer new insight into a specific type of germ – gut bacteria.
Suhayl Dhib-Jalbut, MD, ACTRIMS’s former president, will highlight his team’s investigation into the links between aging and alterations in gut microbiota in MS during the Kenneth P. Johnson Memorial Lecture on Feb. 1 at the meeting in San Diego.
Dr. Dhib-Jalbut’s presentation will focus on findings of a study by his team that was published late last year in Proceedings of the National Academy of Sciences of the U.S.A. (Proc Natl Acad Sci U S A. 2017 Oct 31;114[44]:E9318-27).
As the study explains, Dr. Dhib-Jalbut and colleagues triggered spontaneous experimental autoimmune encephalomyelitis (EAE) in transgenic mice by disrupting gut bacteria during adolescence and early young adulthood. But the process of aging in the mice past early young adulthood suppressed EAE onset by boosting immunologic tolerance.
The findings in this animal model offer insight into why the incidence of MS peaks at ages 20-40 years in humans, he said. “The implication is that one can perhaps manipulate gut bacteria with antibiotics or other treatments to impact the course of MS.”
Germs, genes, and aging each play a role in the development of multiple sclerosis (MS), but researchers are still figuring out how they’re connected. Now, a speaker at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis will shed light on recent findings that offer new insight into a specific type of germ – gut bacteria.
Suhayl Dhib-Jalbut, MD, ACTRIMS’s former president, will highlight his team’s investigation into the links between aging and alterations in gut microbiota in MS during the Kenneth P. Johnson Memorial Lecture on Feb. 1 at the meeting in San Diego.
Dr. Dhib-Jalbut’s presentation will focus on findings of a study by his team that was published late last year in Proceedings of the National Academy of Sciences of the U.S.A. (Proc Natl Acad Sci U S A. 2017 Oct 31;114[44]:E9318-27).
As the study explains, Dr. Dhib-Jalbut and colleagues triggered spontaneous experimental autoimmune encephalomyelitis (EAE) in transgenic mice by disrupting gut bacteria during adolescence and early young adulthood. But the process of aging in the mice past early young adulthood suppressed EAE onset by boosting immunologic tolerance.
The findings in this animal model offer insight into why the incidence of MS peaks at ages 20-40 years in humans, he said. “The implication is that one can perhaps manipulate gut bacteria with antibiotics or other treatments to impact the course of MS.”
REPORTING FROM ACTRIMS FORUM 2018
ACTRIMS Forum 2018 highlights MS therapeutic targets
Current and future therapeutic targets will be the major focus of a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS), according to the meeting’s scientific program committee chair.
As many as 1,000 attendees are expected at the ACTRIMS Forum in San Diego, Feb. 1-3, Dr. Segal said, and about 300 abstracts will be presented. “The idea is for the meeting to be academically rigorous and have a speaker roster composed of the thought leaders in the field to make it akin to more of a keystone symposium.”
The program begins on Feb. 1 with presentations from young investigators about Emerging Concepts in MS. The other sessions will feature discussions about the blood-brain barrier, lymphocytes, cutting-edge developments in MS research, microglia and macrophages, astrocytes, and oligodendrocytes and their precursors.
“The sessions will begin with an overview of the particular target and its role in MS,” Dr. Segal said. “For example, the blood-brain barrier session starts with an overview of the different cell types that maintain the barrier’s integrity and the different ways they could prevent inflammatory cells from entering the central nervous system and causing lesions.”
One presentation in that session, he said, will discuss lessons from research into the mechanism of action of natalizumab (Tysabri). Another presentation focuses on new adhesion molecules – a crucial component of the inflammatory system – that have been discovered in animal models and may lead to new blocking therapies, he said.
The discussion about lymphocytes – white blood cells – will include presentations about new and emerging therapies that deplete them in different ways, he said. “One talk is about alemtuzumab (Lemtrada), which blocks all lymphocytes and can make you susceptible to other autoimmune disorders. What does that mean about how MS is similar to or different than other autoimmune diseases?”
On the myelin front, he said, “there will be a number of talks about how we identify remyelinating agents for clinical trials and how can we enhance precursors of myelination and reconstitute the damaged myelin.”
Another session will look at astrocytes, the glial support cells that are attracting newfound attention. Researchers have known that they’re stimulated in MS, and now new research is suggesting they can cause damage, he said. “They might be a completely new target. There’s evidence that they can have a neuroprotective effect, but it depends on how they’re stimulated.”
Are we on the cusp of being able to reverse damage from MS? “The answers are highly speculative. Research is so unpredictable and clinical trials do take a while,” he said. Still, “we understand repair pathways better than we ever have, and there’s lots of exciting work being done in animal models. We’re closer than we ever have been, and I do believe those drugs will be tested in clinical trials, hopefully within the next 5-10 years.”
Dr. Segal disclosed receiving an investigator-initiated grant from Genentech and support from Mallinckrodt for a clinical trial.
Current and future therapeutic targets will be the major focus of a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS), according to the meeting’s scientific program committee chair.
As many as 1,000 attendees are expected at the ACTRIMS Forum in San Diego, Feb. 1-3, Dr. Segal said, and about 300 abstracts will be presented. “The idea is for the meeting to be academically rigorous and have a speaker roster composed of the thought leaders in the field to make it akin to more of a keystone symposium.”
The program begins on Feb. 1 with presentations from young investigators about Emerging Concepts in MS. The other sessions will feature discussions about the blood-brain barrier, lymphocytes, cutting-edge developments in MS research, microglia and macrophages, astrocytes, and oligodendrocytes and their precursors.
“The sessions will begin with an overview of the particular target and its role in MS,” Dr. Segal said. “For example, the blood-brain barrier session starts with an overview of the different cell types that maintain the barrier’s integrity and the different ways they could prevent inflammatory cells from entering the central nervous system and causing lesions.”
One presentation in that session, he said, will discuss lessons from research into the mechanism of action of natalizumab (Tysabri). Another presentation focuses on new adhesion molecules – a crucial component of the inflammatory system – that have been discovered in animal models and may lead to new blocking therapies, he said.
The discussion about lymphocytes – white blood cells – will include presentations about new and emerging therapies that deplete them in different ways, he said. “One talk is about alemtuzumab (Lemtrada), which blocks all lymphocytes and can make you susceptible to other autoimmune disorders. What does that mean about how MS is similar to or different than other autoimmune diseases?”
On the myelin front, he said, “there will be a number of talks about how we identify remyelinating agents for clinical trials and how can we enhance precursors of myelination and reconstitute the damaged myelin.”
Another session will look at astrocytes, the glial support cells that are attracting newfound attention. Researchers have known that they’re stimulated in MS, and now new research is suggesting they can cause damage, he said. “They might be a completely new target. There’s evidence that they can have a neuroprotective effect, but it depends on how they’re stimulated.”
Are we on the cusp of being able to reverse damage from MS? “The answers are highly speculative. Research is so unpredictable and clinical trials do take a while,” he said. Still, “we understand repair pathways better than we ever have, and there’s lots of exciting work being done in animal models. We’re closer than we ever have been, and I do believe those drugs will be tested in clinical trials, hopefully within the next 5-10 years.”
Dr. Segal disclosed receiving an investigator-initiated grant from Genentech and support from Mallinckrodt for a clinical trial.
Current and future therapeutic targets will be the major focus of a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS), according to the meeting’s scientific program committee chair.
As many as 1,000 attendees are expected at the ACTRIMS Forum in San Diego, Feb. 1-3, Dr. Segal said, and about 300 abstracts will be presented. “The idea is for the meeting to be academically rigorous and have a speaker roster composed of the thought leaders in the field to make it akin to more of a keystone symposium.”
The program begins on Feb. 1 with presentations from young investigators about Emerging Concepts in MS. The other sessions will feature discussions about the blood-brain barrier, lymphocytes, cutting-edge developments in MS research, microglia and macrophages, astrocytes, and oligodendrocytes and their precursors.
“The sessions will begin with an overview of the particular target and its role in MS,” Dr. Segal said. “For example, the blood-brain barrier session starts with an overview of the different cell types that maintain the barrier’s integrity and the different ways they could prevent inflammatory cells from entering the central nervous system and causing lesions.”
One presentation in that session, he said, will discuss lessons from research into the mechanism of action of natalizumab (Tysabri). Another presentation focuses on new adhesion molecules – a crucial component of the inflammatory system – that have been discovered in animal models and may lead to new blocking therapies, he said.
The discussion about lymphocytes – white blood cells – will include presentations about new and emerging therapies that deplete them in different ways, he said. “One talk is about alemtuzumab (Lemtrada), which blocks all lymphocytes and can make you susceptible to other autoimmune disorders. What does that mean about how MS is similar to or different than other autoimmune diseases?”
On the myelin front, he said, “there will be a number of talks about how we identify remyelinating agents for clinical trials and how can we enhance precursors of myelination and reconstitute the damaged myelin.”
Another session will look at astrocytes, the glial support cells that are attracting newfound attention. Researchers have known that they’re stimulated in MS, and now new research is suggesting they can cause damage, he said. “They might be a completely new target. There’s evidence that they can have a neuroprotective effect, but it depends on how they’re stimulated.”
Are we on the cusp of being able to reverse damage from MS? “The answers are highly speculative. Research is so unpredictable and clinical trials do take a while,” he said. Still, “we understand repair pathways better than we ever have, and there’s lots of exciting work being done in animal models. We’re closer than we ever have been, and I do believe those drugs will be tested in clinical trials, hopefully within the next 5-10 years.”
Dr. Segal disclosed receiving an investigator-initiated grant from Genentech and support from Mallinckrodt for a clinical trial.
FROM ACTRIMS FORUM 2018
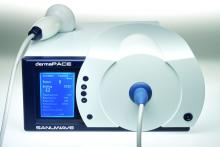
Listen up: Acoustic device useful for diabetic foot ulcers
The Food & Drug Administration has approved the marketing of a device that uses acoustic shock waves to boost wound closure in patients with diabetic foot ulcers (DFUs), an especially stubborn and dangerous condition.
The treatment is experimental, and only limited research into its effectiveness has been published. Still, representatives of its manufacturer say the device, known as dermaPACE, has produced promising results as a secondary treatment in stubborn cases.
A wound care specialist said in an interview that the shock wave technology appears to hold promise.
“A shortcoming in the field of wound care is that providers are typically not trained in a standardized fashion on when and how to a perform meticulous excisional sharp debridement of a wound,” said Bill Tettelbach, MD, systems medical director of Wound Care & Hyperbaric Medicine Services at Intermountain Healthcare in Salt Lake City. “In the majority of cases, the better the debridement, the more rapidly the patient will obtain wound closure.”
This new therapy may provide a benefit as a secondary treatment, especially when the patient cannot tolerate extensive sharp debridement, he said. It also could potentially improve biofilm penetration of antimicrobial topical treatments, he said.
DFUs are believed to affect as many as 1 in 4 people with diabetes over the course of their lifetimes. A 2014 report estimated that care of these wounds costs insurers as much as $13 billion a year in the U.S. alone (Diabetes Care. 2014 Mar;37[3]:651-8).
Treatment options include debridement and, in more extreme cases, hyperbaric oxygen treatment. Amputation can be required if treatment is unsuccessful.
According to Mr. Stegagno, the shock wave device is about the size of a desktop computer from a decade ago. A high-voltage generator box is connected to a handheld therapy head and delivers an acoustic pulse to the patient. The system “is like a spark plug that you see in your automobile,” he said. “It’s pretty much the same technology as lithotripsy, just downsized significantly. The key part is a highly focused, high-energy pulse.”
In a news release, the FDA said it examined the results of two studies of patients with diabetes who received usual DFU care along with either the shock wave therapy or a sham therapy. A total of 336 patients took part in the multicenter, randomized, double-blind studies.
According to the FDA, the studies found a 44% wound closure rate at 24 weeks in patients who had undergone 1-7 shock wave treatments, compared with the 30% wound closure rate in those who received the sham treatment.
Side effects included pain while the device was applied, bruising and numbness, migraines, nausea, fainting, wound infection, fever, and infection beyond the wound such as cellulitis and osteomyelitis.
“There were no meaningful statistical differences in the adverse event rates between the dermaPACE-treated patients and the sham-control group,” Mr. Stegagno said. “There were no issues regarding the tolerability of the treatment, which suggests that a second course of treatment, if needed, is a clinically viable option.”
Mr. Stegagno said the FDA expressed concern about “increased incidences of osteomyelitis at later points in the trials, particularly at the 10-week mark and later.” In response to the agency’s concerns, warning statements were added to labeling, he said.
According to Mr. Stegagno, only one study into the shock wave treatment for DFU has been published, although research has been released through posters and abstracts. The small published study favorably compared shock wave therapy with hyperbaric oxygen therapy. (Diabetes Res Clin Pract. 2011 May;92[2]:187-93)
“Sanuwave will be sponsoring additional studies later this year in the [United States] as follow-on studies to the just-completed DFU trials,” Mr. Stegagno said.
The FDA says the device is intended to be used in adults aged 22 and up with certain types of chronic DFUs. The Sanuwave company says patients should be treated with 4-8 applications over 2-10 weeks.
The shock wave process appears to boost healing through a process that leads to inflammatory responses and oxygenation, Mr. Stegagno said, by first creating an initial compression phase that “squeezes the cell and creates a microtrauma.”
“The cell wakes up and says, ‘Something just punched me,’ ” he said. “This tissue and cellular disruption is believed to initiate the cellular signaling for growth factors and other proteins noted in studies.”
The effects of negative pressure also play a role in stimulation of the wound, he said.
The shock wave therapy will cost an estimated $3,000-$4,000 per protocol of 8 treatments, said Kevin A. Richardson II, the CEO and chairman of the board at Sanuwave, in an interview. The initial plan is for the company to place the devices with doctors while the firm still owns the machines, he said.
The FDA approved the marketing of the device as part of its de novo premarket review pathway, which allows certain new types of devices to be approved when approved similar devices don’t yet exist for the purposes of comparison.
Mr. Stegagno and Mr. Richardson work for Sanuwave. Dr. Tettelbach reported no relevant disclosures.
The Food & Drug Administration has approved the marketing of a device that uses acoustic shock waves to boost wound closure in patients with diabetic foot ulcers (DFUs), an especially stubborn and dangerous condition.
The treatment is experimental, and only limited research into its effectiveness has been published. Still, representatives of its manufacturer say the device, known as dermaPACE, has produced promising results as a secondary treatment in stubborn cases.
A wound care specialist said in an interview that the shock wave technology appears to hold promise.
“A shortcoming in the field of wound care is that providers are typically not trained in a standardized fashion on when and how to a perform meticulous excisional sharp debridement of a wound,” said Bill Tettelbach, MD, systems medical director of Wound Care & Hyperbaric Medicine Services at Intermountain Healthcare in Salt Lake City. “In the majority of cases, the better the debridement, the more rapidly the patient will obtain wound closure.”
This new therapy may provide a benefit as a secondary treatment, especially when the patient cannot tolerate extensive sharp debridement, he said. It also could potentially improve biofilm penetration of antimicrobial topical treatments, he said.
DFUs are believed to affect as many as 1 in 4 people with diabetes over the course of their lifetimes. A 2014 report estimated that care of these wounds costs insurers as much as $13 billion a year in the U.S. alone (Diabetes Care. 2014 Mar;37[3]:651-8).
Treatment options include debridement and, in more extreme cases, hyperbaric oxygen treatment. Amputation can be required if treatment is unsuccessful.
According to Mr. Stegagno, the shock wave device is about the size of a desktop computer from a decade ago. A high-voltage generator box is connected to a handheld therapy head and delivers an acoustic pulse to the patient. The system “is like a spark plug that you see in your automobile,” he said. “It’s pretty much the same technology as lithotripsy, just downsized significantly. The key part is a highly focused, high-energy pulse.”
In a news release, the FDA said it examined the results of two studies of patients with diabetes who received usual DFU care along with either the shock wave therapy or a sham therapy. A total of 336 patients took part in the multicenter, randomized, double-blind studies.
According to the FDA, the studies found a 44% wound closure rate at 24 weeks in patients who had undergone 1-7 shock wave treatments, compared with the 30% wound closure rate in those who received the sham treatment.
Side effects included pain while the device was applied, bruising and numbness, migraines, nausea, fainting, wound infection, fever, and infection beyond the wound such as cellulitis and osteomyelitis.
“There were no meaningful statistical differences in the adverse event rates between the dermaPACE-treated patients and the sham-control group,” Mr. Stegagno said. “There were no issues regarding the tolerability of the treatment, which suggests that a second course of treatment, if needed, is a clinically viable option.”
Mr. Stegagno said the FDA expressed concern about “increased incidences of osteomyelitis at later points in the trials, particularly at the 10-week mark and later.” In response to the agency’s concerns, warning statements were added to labeling, he said.
According to Mr. Stegagno, only one study into the shock wave treatment for DFU has been published, although research has been released through posters and abstracts. The small published study favorably compared shock wave therapy with hyperbaric oxygen therapy. (Diabetes Res Clin Pract. 2011 May;92[2]:187-93)
“Sanuwave will be sponsoring additional studies later this year in the [United States] as follow-on studies to the just-completed DFU trials,” Mr. Stegagno said.
The FDA says the device is intended to be used in adults aged 22 and up with certain types of chronic DFUs. The Sanuwave company says patients should be treated with 4-8 applications over 2-10 weeks.
The shock wave process appears to boost healing through a process that leads to inflammatory responses and oxygenation, Mr. Stegagno said, by first creating an initial compression phase that “squeezes the cell and creates a microtrauma.”
“The cell wakes up and says, ‘Something just punched me,’ ” he said. “This tissue and cellular disruption is believed to initiate the cellular signaling for growth factors and other proteins noted in studies.”
The effects of negative pressure also play a role in stimulation of the wound, he said.
The shock wave therapy will cost an estimated $3,000-$4,000 per protocol of 8 treatments, said Kevin A. Richardson II, the CEO and chairman of the board at Sanuwave, in an interview. The initial plan is for the company to place the devices with doctors while the firm still owns the machines, he said.
The FDA approved the marketing of the device as part of its de novo premarket review pathway, which allows certain new types of devices to be approved when approved similar devices don’t yet exist for the purposes of comparison.
Mr. Stegagno and Mr. Richardson work for Sanuwave. Dr. Tettelbach reported no relevant disclosures.
The Food & Drug Administration has approved the marketing of a device that uses acoustic shock waves to boost wound closure in patients with diabetic foot ulcers (DFUs), an especially stubborn and dangerous condition.
The treatment is experimental, and only limited research into its effectiveness has been published. Still, representatives of its manufacturer say the device, known as dermaPACE, has produced promising results as a secondary treatment in stubborn cases.
A wound care specialist said in an interview that the shock wave technology appears to hold promise.
“A shortcoming in the field of wound care is that providers are typically not trained in a standardized fashion on when and how to a perform meticulous excisional sharp debridement of a wound,” said Bill Tettelbach, MD, systems medical director of Wound Care & Hyperbaric Medicine Services at Intermountain Healthcare in Salt Lake City. “In the majority of cases, the better the debridement, the more rapidly the patient will obtain wound closure.”
This new therapy may provide a benefit as a secondary treatment, especially when the patient cannot tolerate extensive sharp debridement, he said. It also could potentially improve biofilm penetration of antimicrobial topical treatments, he said.
DFUs are believed to affect as many as 1 in 4 people with diabetes over the course of their lifetimes. A 2014 report estimated that care of these wounds costs insurers as much as $13 billion a year in the U.S. alone (Diabetes Care. 2014 Mar;37[3]:651-8).
Treatment options include debridement and, in more extreme cases, hyperbaric oxygen treatment. Amputation can be required if treatment is unsuccessful.
According to Mr. Stegagno, the shock wave device is about the size of a desktop computer from a decade ago. A high-voltage generator box is connected to a handheld therapy head and delivers an acoustic pulse to the patient. The system “is like a spark plug that you see in your automobile,” he said. “It’s pretty much the same technology as lithotripsy, just downsized significantly. The key part is a highly focused, high-energy pulse.”
In a news release, the FDA said it examined the results of two studies of patients with diabetes who received usual DFU care along with either the shock wave therapy or a sham therapy. A total of 336 patients took part in the multicenter, randomized, double-blind studies.
According to the FDA, the studies found a 44% wound closure rate at 24 weeks in patients who had undergone 1-7 shock wave treatments, compared with the 30% wound closure rate in those who received the sham treatment.
Side effects included pain while the device was applied, bruising and numbness, migraines, nausea, fainting, wound infection, fever, and infection beyond the wound such as cellulitis and osteomyelitis.
“There were no meaningful statistical differences in the adverse event rates between the dermaPACE-treated patients and the sham-control group,” Mr. Stegagno said. “There were no issues regarding the tolerability of the treatment, which suggests that a second course of treatment, if needed, is a clinically viable option.”
Mr. Stegagno said the FDA expressed concern about “increased incidences of osteomyelitis at later points in the trials, particularly at the 10-week mark and later.” In response to the agency’s concerns, warning statements were added to labeling, he said.
According to Mr. Stegagno, only one study into the shock wave treatment for DFU has been published, although research has been released through posters and abstracts. The small published study favorably compared shock wave therapy with hyperbaric oxygen therapy. (Diabetes Res Clin Pract. 2011 May;92[2]:187-93)
“Sanuwave will be sponsoring additional studies later this year in the [United States] as follow-on studies to the just-completed DFU trials,” Mr. Stegagno said.
The FDA says the device is intended to be used in adults aged 22 and up with certain types of chronic DFUs. The Sanuwave company says patients should be treated with 4-8 applications over 2-10 weeks.
The shock wave process appears to boost healing through a process that leads to inflammatory responses and oxygenation, Mr. Stegagno said, by first creating an initial compression phase that “squeezes the cell and creates a microtrauma.”
“The cell wakes up and says, ‘Something just punched me,’ ” he said. “This tissue and cellular disruption is believed to initiate the cellular signaling for growth factors and other proteins noted in studies.”
The effects of negative pressure also play a role in stimulation of the wound, he said.
The shock wave therapy will cost an estimated $3,000-$4,000 per protocol of 8 treatments, said Kevin A. Richardson II, the CEO and chairman of the board at Sanuwave, in an interview. The initial plan is for the company to place the devices with doctors while the firm still owns the machines, he said.
The FDA approved the marketing of the device as part of its de novo premarket review pathway, which allows certain new types of devices to be approved when approved similar devices don’t yet exist for the purposes of comparison.
Mr. Stegagno and Mr. Richardson work for Sanuwave. Dr. Tettelbach reported no relevant disclosures.