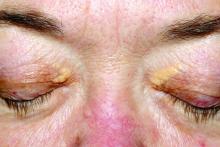
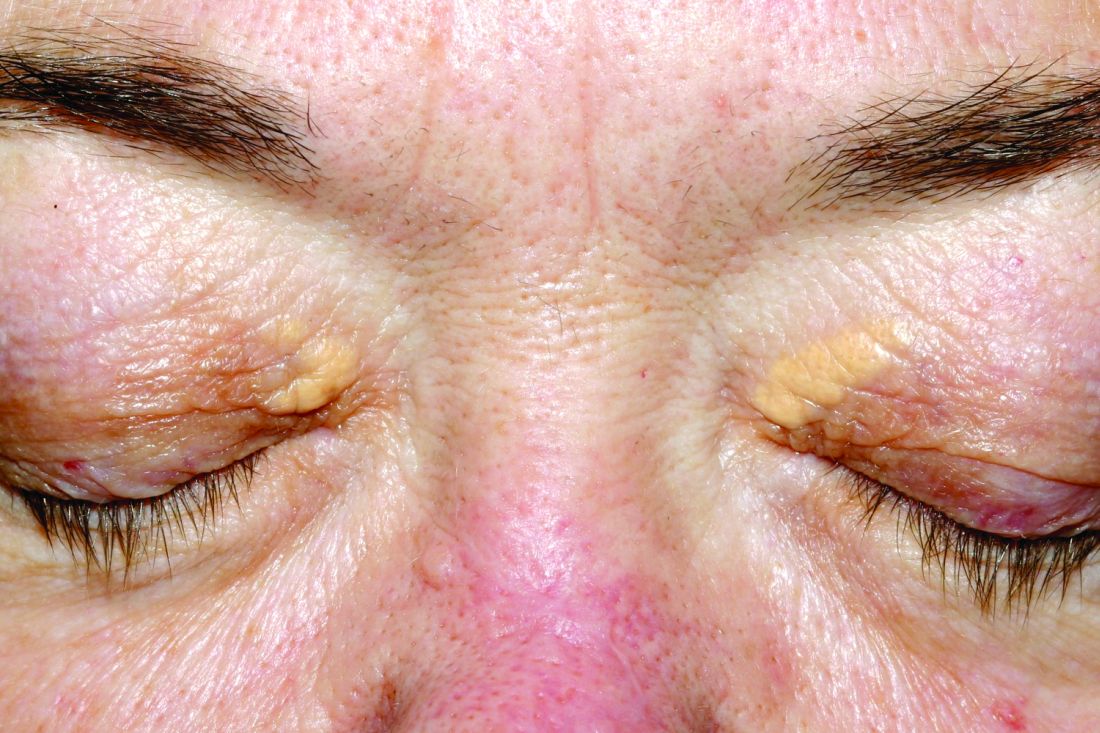
User login
Cutaneous laser surgery: Basic caution isn’t enough to prevent lawsuits
SAN DIEGO – Injuries and lawsuits related to laser cosmetic surgery are increasing and potential legal threats are not always easy to predict, according to two dermatologists who spoke at the annual meeting of the American Society for Laser Medicine and Surgery (ASLMS).
A laser procedure could go smoothly, for example, but the patient might be able to successfully sue if he or she is allowed to drive home after receiving a sedative. Or a physician might get sued because his or her nurse set a laser at the wrong setting and singed a patient.
The risk of a lawsuit is high, H. Ray Jalian, MD, a dermatologist in Los Angeles, said at the meeting. “The reality is that we’re all at some point going to face this.”
The most common procedure litigated was laser hair removal, making up almost 40% of the cases, which is not an indication that this particular procedure is dangerous, Dr. Jalian said. “It’s quite safe, and the complication rate is quite low,” but more of these procedures are being done, he noted. Rejuvenation procedures followed, accounting for 25% of cases.
The alleged injuries sustained from laser surgery included burns (47%), scars (39%), and pigmentation problems (24%). Deaths occurred in just over 2% of the cases. In the study, almost a third of plaintiffs alleged that they were not provided informed consent. Plaintiffs also alleged fraud (9%) and assault/battery (5%), and a family member occasionally sued for loss of consortium (8% of cases). The specialty with the largest percentage of the cases was plastic surgery (26%), followed by dermatology (21%).
Dr. Jalian and his copresenter, Mathew Avram, MD, JD, director of the Dermatology Laser & Cosmetic Center, and director of dermatologic surgery at Massachusetts General Hospital, Boston, offered these lessons about the legal risks associated with laser procedures:
• You may have a duty to protect your patient from bad choices.
Physicians aren’t expected to keep patients from making certain bad decisions such as sunbathing after a traditional resurfacing procedure, said Dr. Avram, of the department of dermatology, Harvard Medical School, Boston, and the ASLMS president. But in some cases, he said, the law may expect the physician to step in to prevent harm. For example, he said, a patient who has undergone a fractional ablative laser procedure and has received a sedative should not be allowed to drive home.
• You may get sued even if your employee is at fault.
The 2013 study found physicians were often sued even when they did not perform the laser procedure in question. Nonphysicians such as physician assistants and nurses often perform laser operations, and many states allow them to do so. “Nonphysicians were less likely to be sued even if they were the operators,” Dr. Jalian said. In the study, almost 38% of the 174 analyzed cases involved nonphysician operators, but they were sued in just 26% of the cases. In 33 of the 174 cases in the study, plaintiffs alleged failure to properly hire, train, or supervise staff.
He recommended looking at state laws, which differ greatly in their regulations – or lack of them – regarding the operation of medical lasers. In some cases, physicians must supervise laser use, he said. “But what are the requirements? Can you be available by phone down the street or in the Caribbean?”
Dr. Jalian, Dr. Avram, and a colleague followed up the 2013 study with another study that tracked 175 legal cases from 1999 to 2012 involving alleged injuries from cutaneous laser surgery. During this time period, 75 (43%) involved a nonphysician operating a laser, increasing from 36% in 2008 to 78% in 2012.
In almost two-thirds of cases, the procedures in question were done by nonphysicians outside a “traditional medical setting” such as a salon or spa (JAMA Dermatol. 2014 Apr;150[4]:407-11).
• Delayed side effects could mean delayed lawsuits.
According to Dr. Avram, statutes of limitations – the length of time in which a patient can file a lawsuit – typically last for 2-3 years in malpractice cases. But he said that the period begins when the physician is alleged to have made a mistake or when the patient becomes aware of – or should reasonably be aware of – an injury. Therefore, physicians could face legal trouble over delayed hypopigmentation that appears 6 months after a laser resurfacing treatment, or granulomas that appear years after a filler treatment, he said.
• A signed form is not a cure-all.
It is wise to make patients sign an extensive informed consent form, but this will not protect a physician against a claim of negligence, Dr. Avram said. And the reverse is also true: If a patient did not sign a proper consent form, he or she could still sue even if the procedure went perfectly, he noted.
• Your instincts are worth trusting.
When it comes to lawsuit prevention, Dr. Avram said, “by far the most important thing you can do happens within a minute of when you see the patient. Assess and trust your own intuition and your staff’s intuition. For elective, cosmetic treatments, don’t be afraid to say no. There’s no legal obligation to perform a cosmetic treatment on a patient.”
If you do choose to treat a patient, he advised, be open about the procedure and “maybe even tell them some of the tougher, worse-case scenarios.” If a procedure goes poorly, he said, consider how to fix it. “Many complications can be significantly improved or cleared with timely and appropriate intervention,” he said.
In some cases, refunding the patient’s money can be considered, with the patient signing a release, he said. “Document that you are refunding the money in order to preserve the doctor-patient relationship, not to avoid negligence.”
Dr. Jalian and Dr. Avram reported no relevant disclosures.
SAN DIEGO – Injuries and lawsuits related to laser cosmetic surgery are increasing and potential legal threats are not always easy to predict, according to two dermatologists who spoke at the annual meeting of the American Society for Laser Medicine and Surgery (ASLMS).
A laser procedure could go smoothly, for example, but the patient might be able to successfully sue if he or she is allowed to drive home after receiving a sedative. Or a physician might get sued because his or her nurse set a laser at the wrong setting and singed a patient.
The risk of a lawsuit is high, H. Ray Jalian, MD, a dermatologist in Los Angeles, said at the meeting. “The reality is that we’re all at some point going to face this.”
The most common procedure litigated was laser hair removal, making up almost 40% of the cases, which is not an indication that this particular procedure is dangerous, Dr. Jalian said. “It’s quite safe, and the complication rate is quite low,” but more of these procedures are being done, he noted. Rejuvenation procedures followed, accounting for 25% of cases.
The alleged injuries sustained from laser surgery included burns (47%), scars (39%), and pigmentation problems (24%). Deaths occurred in just over 2% of the cases. In the study, almost a third of plaintiffs alleged that they were not provided informed consent. Plaintiffs also alleged fraud (9%) and assault/battery (5%), and a family member occasionally sued for loss of consortium (8% of cases). The specialty with the largest percentage of the cases was plastic surgery (26%), followed by dermatology (21%).
Dr. Jalian and his copresenter, Mathew Avram, MD, JD, director of the Dermatology Laser & Cosmetic Center, and director of dermatologic surgery at Massachusetts General Hospital, Boston, offered these lessons about the legal risks associated with laser procedures:
• You may have a duty to protect your patient from bad choices.
Physicians aren’t expected to keep patients from making certain bad decisions such as sunbathing after a traditional resurfacing procedure, said Dr. Avram, of the department of dermatology, Harvard Medical School, Boston, and the ASLMS president. But in some cases, he said, the law may expect the physician to step in to prevent harm. For example, he said, a patient who has undergone a fractional ablative laser procedure and has received a sedative should not be allowed to drive home.
• You may get sued even if your employee is at fault.
The 2013 study found physicians were often sued even when they did not perform the laser procedure in question. Nonphysicians such as physician assistants and nurses often perform laser operations, and many states allow them to do so. “Nonphysicians were less likely to be sued even if they were the operators,” Dr. Jalian said. In the study, almost 38% of the 174 analyzed cases involved nonphysician operators, but they were sued in just 26% of the cases. In 33 of the 174 cases in the study, plaintiffs alleged failure to properly hire, train, or supervise staff.
He recommended looking at state laws, which differ greatly in their regulations – or lack of them – regarding the operation of medical lasers. In some cases, physicians must supervise laser use, he said. “But what are the requirements? Can you be available by phone down the street or in the Caribbean?”
Dr. Jalian, Dr. Avram, and a colleague followed up the 2013 study with another study that tracked 175 legal cases from 1999 to 2012 involving alleged injuries from cutaneous laser surgery. During this time period, 75 (43%) involved a nonphysician operating a laser, increasing from 36% in 2008 to 78% in 2012.
In almost two-thirds of cases, the procedures in question were done by nonphysicians outside a “traditional medical setting” such as a salon or spa (JAMA Dermatol. 2014 Apr;150[4]:407-11).
• Delayed side effects could mean delayed lawsuits.
According to Dr. Avram, statutes of limitations – the length of time in which a patient can file a lawsuit – typically last for 2-3 years in malpractice cases. But he said that the period begins when the physician is alleged to have made a mistake or when the patient becomes aware of – or should reasonably be aware of – an injury. Therefore, physicians could face legal trouble over delayed hypopigmentation that appears 6 months after a laser resurfacing treatment, or granulomas that appear years after a filler treatment, he said.
• A signed form is not a cure-all.
It is wise to make patients sign an extensive informed consent form, but this will not protect a physician against a claim of negligence, Dr. Avram said. And the reverse is also true: If a patient did not sign a proper consent form, he or she could still sue even if the procedure went perfectly, he noted.
• Your instincts are worth trusting.
When it comes to lawsuit prevention, Dr. Avram said, “by far the most important thing you can do happens within a minute of when you see the patient. Assess and trust your own intuition and your staff’s intuition. For elective, cosmetic treatments, don’t be afraid to say no. There’s no legal obligation to perform a cosmetic treatment on a patient.”
If you do choose to treat a patient, he advised, be open about the procedure and “maybe even tell them some of the tougher, worse-case scenarios.” If a procedure goes poorly, he said, consider how to fix it. “Many complications can be significantly improved or cleared with timely and appropriate intervention,” he said.
In some cases, refunding the patient’s money can be considered, with the patient signing a release, he said. “Document that you are refunding the money in order to preserve the doctor-patient relationship, not to avoid negligence.”
Dr. Jalian and Dr. Avram reported no relevant disclosures.
SAN DIEGO – Injuries and lawsuits related to laser cosmetic surgery are increasing and potential legal threats are not always easy to predict, according to two dermatologists who spoke at the annual meeting of the American Society for Laser Medicine and Surgery (ASLMS).
A laser procedure could go smoothly, for example, but the patient might be able to successfully sue if he or she is allowed to drive home after receiving a sedative. Or a physician might get sued because his or her nurse set a laser at the wrong setting and singed a patient.
The risk of a lawsuit is high, H. Ray Jalian, MD, a dermatologist in Los Angeles, said at the meeting. “The reality is that we’re all at some point going to face this.”
The most common procedure litigated was laser hair removal, making up almost 40% of the cases, which is not an indication that this particular procedure is dangerous, Dr. Jalian said. “It’s quite safe, and the complication rate is quite low,” but more of these procedures are being done, he noted. Rejuvenation procedures followed, accounting for 25% of cases.
The alleged injuries sustained from laser surgery included burns (47%), scars (39%), and pigmentation problems (24%). Deaths occurred in just over 2% of the cases. In the study, almost a third of plaintiffs alleged that they were not provided informed consent. Plaintiffs also alleged fraud (9%) and assault/battery (5%), and a family member occasionally sued for loss of consortium (8% of cases). The specialty with the largest percentage of the cases was plastic surgery (26%), followed by dermatology (21%).
Dr. Jalian and his copresenter, Mathew Avram, MD, JD, director of the Dermatology Laser & Cosmetic Center, and director of dermatologic surgery at Massachusetts General Hospital, Boston, offered these lessons about the legal risks associated with laser procedures:
• You may have a duty to protect your patient from bad choices.
Physicians aren’t expected to keep patients from making certain bad decisions such as sunbathing after a traditional resurfacing procedure, said Dr. Avram, of the department of dermatology, Harvard Medical School, Boston, and the ASLMS president. But in some cases, he said, the law may expect the physician to step in to prevent harm. For example, he said, a patient who has undergone a fractional ablative laser procedure and has received a sedative should not be allowed to drive home.
• You may get sued even if your employee is at fault.
The 2013 study found physicians were often sued even when they did not perform the laser procedure in question. Nonphysicians such as physician assistants and nurses often perform laser operations, and many states allow them to do so. “Nonphysicians were less likely to be sued even if they were the operators,” Dr. Jalian said. In the study, almost 38% of the 174 analyzed cases involved nonphysician operators, but they were sued in just 26% of the cases. In 33 of the 174 cases in the study, plaintiffs alleged failure to properly hire, train, or supervise staff.
He recommended looking at state laws, which differ greatly in their regulations – or lack of them – regarding the operation of medical lasers. In some cases, physicians must supervise laser use, he said. “But what are the requirements? Can you be available by phone down the street or in the Caribbean?”
Dr. Jalian, Dr. Avram, and a colleague followed up the 2013 study with another study that tracked 175 legal cases from 1999 to 2012 involving alleged injuries from cutaneous laser surgery. During this time period, 75 (43%) involved a nonphysician operating a laser, increasing from 36% in 2008 to 78% in 2012.
In almost two-thirds of cases, the procedures in question were done by nonphysicians outside a “traditional medical setting” such as a salon or spa (JAMA Dermatol. 2014 Apr;150[4]:407-11).
• Delayed side effects could mean delayed lawsuits.
According to Dr. Avram, statutes of limitations – the length of time in which a patient can file a lawsuit – typically last for 2-3 years in malpractice cases. But he said that the period begins when the physician is alleged to have made a mistake or when the patient becomes aware of – or should reasonably be aware of – an injury. Therefore, physicians could face legal trouble over delayed hypopigmentation that appears 6 months after a laser resurfacing treatment, or granulomas that appear years after a filler treatment, he said.
• A signed form is not a cure-all.
It is wise to make patients sign an extensive informed consent form, but this will not protect a physician against a claim of negligence, Dr. Avram said. And the reverse is also true: If a patient did not sign a proper consent form, he or she could still sue even if the procedure went perfectly, he noted.
• Your instincts are worth trusting.
When it comes to lawsuit prevention, Dr. Avram said, “by far the most important thing you can do happens within a minute of when you see the patient. Assess and trust your own intuition and your staff’s intuition. For elective, cosmetic treatments, don’t be afraid to say no. There’s no legal obligation to perform a cosmetic treatment on a patient.”
If you do choose to treat a patient, he advised, be open about the procedure and “maybe even tell them some of the tougher, worse-case scenarios.” If a procedure goes poorly, he said, consider how to fix it. “Many complications can be significantly improved or cleared with timely and appropriate intervention,” he said.
In some cases, refunding the patient’s money can be considered, with the patient signing a release, he said. “Document that you are refunding the money in order to preserve the doctor-patient relationship, not to avoid negligence.”
Dr. Jalian and Dr. Avram reported no relevant disclosures.
AT LASER 2017
Nusinersen for early spinal muscular atrophy: Final findings beat interim results
BOSTON – Last December, the Food and Drug Administration approved nusinersen (Spinraza) as the only treatment for spinal muscular atrophy (SMA) based in part on startling interim results from a study in infants that ended early so all participants could get access to the drug.
Now the final study results are in, and they’re even more impressive: Patients who took the drug were less than half as likely to die as were those in a sham control group, and motor function improved in more than half of infants who took the medication, compared with none in the sham group.
The findings are “incredibly exciting,” said Charlotte J. Sumner, MD, in a discussant presentation at the annual meeting of the American Academy of Neurology. “The data teaches us that this drug is efficacious.”
However, Dr. Sumner, professor of neurology and neuroscience at Johns Hopkins University, Baltimore, cautioned that the drug is tremendously expensive, challenging to administer, and there are several questions about its use that remain unanswered.
SMA is most often caused by mutations in the SMN1 gene, leading to a loss of the protein called survival motor neuron (SMN), which is essential for the survival of motor neurons, except for a small amount produced from the SMN2 gene. The loss of motor neurons in the spinal cord and brainstem leads to weakness and atrophy of muscles used for crawling, walking, sitting up, controlling head movement, and in severe cases, breathing and swallowing. It affects an estimated 1 in 6,000 newborns and is the most common genetic cause of death in infants. Nusinersen is a antisense oligonucleotide that promotes transcription of the full-length SMN protein from the SMN2 gene.
At the AAN meeting, investigators presented the final results of two nusinersen phase III studies as well as interim data from a separate phase II trial.
The ENDEAR trial, a randomized, double-blind, sham-procedure controlled study, assigned 80 patients to 12-mg scaled equivalent doses of nusinersen delivered intrathecally (four doses over 2 months, then one every 4 months) and 41 to a sham procedure (a needle prick in the lower back). The subjects, aged 30-252 days at first dose, underwent 13 months of treatment and follow-up.
Researchers presented an end-of-study analysis of 110 subjects. They found that 51% of those who received the drug were “motor milestone responders” – meaning they had more modified Hammersmith Infant Neurological Examination Section 2 categories with improvement than worsening – compared with 41% at the interim analysis (P less than .0001). None of the sham group patients improved.
The patients treated with nusinersen lived longer: 39% of those in the sham procedure group died, compared with 16% of those in the nusinersen group (hazard ratio, 0.372; P = .0041). Event-free survival (survival without permanent ventilation) was also better in those who received the drugs: 61% in those who took nusinersen, compared with 32% in those who did not (HR, 0.53; P = .0046)
Also, 71% of those treated with the drug were considered responders at day 183 onward based on an improvement of 4 or more points on the Children’s Hospital of Philadelphia Infant Test of Neuromuscular Disorders scale, whereas the response rate was only 3% in the sham group (P less than .0001).
The study authors report that nusinersen was well tolerated with adverse events that “were consistent with those expected in the general population of infants with SMA.” No adverse events were linked to the nusinersen treatment.
Researchers also presented the end-of-study results of the phase III CHERISH trial of 126 patients aged 2-12 years with later-onset SMA who could sit up but not walk. All participants were believed to have a life expectancy of at least 2 years.
The trial is a multicenter, randomized, double-blind, sham-procedure controlled study. The 84 subjects who received the drug improved an average of 3.9 points by the end of study (15 months) on the Hammersmith Functional Motor Scale Expanded score, a measurement of motor function in children with SMA (P = .0000001). The 42 children who received the sham treatment (a small needle prick on the lower back) declined by an average of 1.0 point.
Researchers report that children taking the drug had fewer adverse events, and most were not related to the drug itself.
All surviving participants who completed nusinersen investigational studies are being enrolled in the SHINE open-label extension trial.
Also at the AAN meeting, researchers released interim phase II results from the NURTURE trial, an open-label study of nusinersen in patients with diagnosed but presymptomatic SMA. The study, which is expected to be completed in 2022, recruited participants up to the age of 6 weeks.
The 20 enrolled infants were all alive at a median study period of 317.5 days, and none required assistance with respiration.
In terms of motor function and growth, most were making gains “generally consistent with normal development such as head control, independent sitting, standing and walking independently, as measured by validated scales,” according to a statement released by Biogen.
Three patients developed adverse events that may have been drug related, but the events were resolved and they remained in the trial.
In Dr. Sumner’s discussant presentation on the ENDEAR study, she cautioned that it’s still not known how much older patients with SMA will benefit from the drug treatment. It’s possible that younger patients may gain motor function while older patients will maintain what they have, she said.
In addition, it seems that “certain body segments may respond more robustly than some others. It’s possible that an infant may improve limb strength quite robustly but not lumbar strength.”
She pointed out other questions: Can the drug, which is difficult to administer, be stopped at some point? “We do think of SMA as a disease of development,” she said. “Maybe after development is finished we can withdraw the drug.”
Then there’s the issue of cost and whether insurers are willing to cover the drug. As Dr. Sumner pointed out, the drug is $125,000 per dose and $750,000 over the first year, then $325,000 a year.
It is clear, however, that “the early and efficient diagnosis of SMA is really important, particularly for infantile SMA,” she said. “Time is motor function.”
The studies were funded by Ionis Pharmaceuticals and Biogen. Dr. Sumner reports serving as a consultant for Biogen, Ionis, Avexis, and Roche. She has served on scientific advisory boards for the Cure SMA, SMA Foundation, and Muscular Dystrophy Association nonprofit foundations.
BOSTON – Last December, the Food and Drug Administration approved nusinersen (Spinraza) as the only treatment for spinal muscular atrophy (SMA) based in part on startling interim results from a study in infants that ended early so all participants could get access to the drug.
Now the final study results are in, and they’re even more impressive: Patients who took the drug were less than half as likely to die as were those in a sham control group, and motor function improved in more than half of infants who took the medication, compared with none in the sham group.
The findings are “incredibly exciting,” said Charlotte J. Sumner, MD, in a discussant presentation at the annual meeting of the American Academy of Neurology. “The data teaches us that this drug is efficacious.”
However, Dr. Sumner, professor of neurology and neuroscience at Johns Hopkins University, Baltimore, cautioned that the drug is tremendously expensive, challenging to administer, and there are several questions about its use that remain unanswered.
SMA is most often caused by mutations in the SMN1 gene, leading to a loss of the protein called survival motor neuron (SMN), which is essential for the survival of motor neurons, except for a small amount produced from the SMN2 gene. The loss of motor neurons in the spinal cord and brainstem leads to weakness and atrophy of muscles used for crawling, walking, sitting up, controlling head movement, and in severe cases, breathing and swallowing. It affects an estimated 1 in 6,000 newborns and is the most common genetic cause of death in infants. Nusinersen is a antisense oligonucleotide that promotes transcription of the full-length SMN protein from the SMN2 gene.
At the AAN meeting, investigators presented the final results of two nusinersen phase III studies as well as interim data from a separate phase II trial.
The ENDEAR trial, a randomized, double-blind, sham-procedure controlled study, assigned 80 patients to 12-mg scaled equivalent doses of nusinersen delivered intrathecally (four doses over 2 months, then one every 4 months) and 41 to a sham procedure (a needle prick in the lower back). The subjects, aged 30-252 days at first dose, underwent 13 months of treatment and follow-up.
Researchers presented an end-of-study analysis of 110 subjects. They found that 51% of those who received the drug were “motor milestone responders” – meaning they had more modified Hammersmith Infant Neurological Examination Section 2 categories with improvement than worsening – compared with 41% at the interim analysis (P less than .0001). None of the sham group patients improved.
The patients treated with nusinersen lived longer: 39% of those in the sham procedure group died, compared with 16% of those in the nusinersen group (hazard ratio, 0.372; P = .0041). Event-free survival (survival without permanent ventilation) was also better in those who received the drugs: 61% in those who took nusinersen, compared with 32% in those who did not (HR, 0.53; P = .0046)
Also, 71% of those treated with the drug were considered responders at day 183 onward based on an improvement of 4 or more points on the Children’s Hospital of Philadelphia Infant Test of Neuromuscular Disorders scale, whereas the response rate was only 3% in the sham group (P less than .0001).
The study authors report that nusinersen was well tolerated with adverse events that “were consistent with those expected in the general population of infants with SMA.” No adverse events were linked to the nusinersen treatment.
Researchers also presented the end-of-study results of the phase III CHERISH trial of 126 patients aged 2-12 years with later-onset SMA who could sit up but not walk. All participants were believed to have a life expectancy of at least 2 years.
The trial is a multicenter, randomized, double-blind, sham-procedure controlled study. The 84 subjects who received the drug improved an average of 3.9 points by the end of study (15 months) on the Hammersmith Functional Motor Scale Expanded score, a measurement of motor function in children with SMA (P = .0000001). The 42 children who received the sham treatment (a small needle prick on the lower back) declined by an average of 1.0 point.
Researchers report that children taking the drug had fewer adverse events, and most were not related to the drug itself.
All surviving participants who completed nusinersen investigational studies are being enrolled in the SHINE open-label extension trial.
Also at the AAN meeting, researchers released interim phase II results from the NURTURE trial, an open-label study of nusinersen in patients with diagnosed but presymptomatic SMA. The study, which is expected to be completed in 2022, recruited participants up to the age of 6 weeks.
The 20 enrolled infants were all alive at a median study period of 317.5 days, and none required assistance with respiration.
In terms of motor function and growth, most were making gains “generally consistent with normal development such as head control, independent sitting, standing and walking independently, as measured by validated scales,” according to a statement released by Biogen.
Three patients developed adverse events that may have been drug related, but the events were resolved and they remained in the trial.
In Dr. Sumner’s discussant presentation on the ENDEAR study, she cautioned that it’s still not known how much older patients with SMA will benefit from the drug treatment. It’s possible that younger patients may gain motor function while older patients will maintain what they have, she said.
In addition, it seems that “certain body segments may respond more robustly than some others. It’s possible that an infant may improve limb strength quite robustly but not lumbar strength.”
She pointed out other questions: Can the drug, which is difficult to administer, be stopped at some point? “We do think of SMA as a disease of development,” she said. “Maybe after development is finished we can withdraw the drug.”
Then there’s the issue of cost and whether insurers are willing to cover the drug. As Dr. Sumner pointed out, the drug is $125,000 per dose and $750,000 over the first year, then $325,000 a year.
It is clear, however, that “the early and efficient diagnosis of SMA is really important, particularly for infantile SMA,” she said. “Time is motor function.”
The studies were funded by Ionis Pharmaceuticals and Biogen. Dr. Sumner reports serving as a consultant for Biogen, Ionis, Avexis, and Roche. She has served on scientific advisory boards for the Cure SMA, SMA Foundation, and Muscular Dystrophy Association nonprofit foundations.
BOSTON – Last December, the Food and Drug Administration approved nusinersen (Spinraza) as the only treatment for spinal muscular atrophy (SMA) based in part on startling interim results from a study in infants that ended early so all participants could get access to the drug.
Now the final study results are in, and they’re even more impressive: Patients who took the drug were less than half as likely to die as were those in a sham control group, and motor function improved in more than half of infants who took the medication, compared with none in the sham group.
The findings are “incredibly exciting,” said Charlotte J. Sumner, MD, in a discussant presentation at the annual meeting of the American Academy of Neurology. “The data teaches us that this drug is efficacious.”
However, Dr. Sumner, professor of neurology and neuroscience at Johns Hopkins University, Baltimore, cautioned that the drug is tremendously expensive, challenging to administer, and there are several questions about its use that remain unanswered.
SMA is most often caused by mutations in the SMN1 gene, leading to a loss of the protein called survival motor neuron (SMN), which is essential for the survival of motor neurons, except for a small amount produced from the SMN2 gene. The loss of motor neurons in the spinal cord and brainstem leads to weakness and atrophy of muscles used for crawling, walking, sitting up, controlling head movement, and in severe cases, breathing and swallowing. It affects an estimated 1 in 6,000 newborns and is the most common genetic cause of death in infants. Nusinersen is a antisense oligonucleotide that promotes transcription of the full-length SMN protein from the SMN2 gene.
At the AAN meeting, investigators presented the final results of two nusinersen phase III studies as well as interim data from a separate phase II trial.
The ENDEAR trial, a randomized, double-blind, sham-procedure controlled study, assigned 80 patients to 12-mg scaled equivalent doses of nusinersen delivered intrathecally (four doses over 2 months, then one every 4 months) and 41 to a sham procedure (a needle prick in the lower back). The subjects, aged 30-252 days at first dose, underwent 13 months of treatment and follow-up.
Researchers presented an end-of-study analysis of 110 subjects. They found that 51% of those who received the drug were “motor milestone responders” – meaning they had more modified Hammersmith Infant Neurological Examination Section 2 categories with improvement than worsening – compared with 41% at the interim analysis (P less than .0001). None of the sham group patients improved.
The patients treated with nusinersen lived longer: 39% of those in the sham procedure group died, compared with 16% of those in the nusinersen group (hazard ratio, 0.372; P = .0041). Event-free survival (survival without permanent ventilation) was also better in those who received the drugs: 61% in those who took nusinersen, compared with 32% in those who did not (HR, 0.53; P = .0046)
Also, 71% of those treated with the drug were considered responders at day 183 onward based on an improvement of 4 or more points on the Children’s Hospital of Philadelphia Infant Test of Neuromuscular Disorders scale, whereas the response rate was only 3% in the sham group (P less than .0001).
The study authors report that nusinersen was well tolerated with adverse events that “were consistent with those expected in the general population of infants with SMA.” No adverse events were linked to the nusinersen treatment.
Researchers also presented the end-of-study results of the phase III CHERISH trial of 126 patients aged 2-12 years with later-onset SMA who could sit up but not walk. All participants were believed to have a life expectancy of at least 2 years.
The trial is a multicenter, randomized, double-blind, sham-procedure controlled study. The 84 subjects who received the drug improved an average of 3.9 points by the end of study (15 months) on the Hammersmith Functional Motor Scale Expanded score, a measurement of motor function in children with SMA (P = .0000001). The 42 children who received the sham treatment (a small needle prick on the lower back) declined by an average of 1.0 point.
Researchers report that children taking the drug had fewer adverse events, and most were not related to the drug itself.
All surviving participants who completed nusinersen investigational studies are being enrolled in the SHINE open-label extension trial.
Also at the AAN meeting, researchers released interim phase II results from the NURTURE trial, an open-label study of nusinersen in patients with diagnosed but presymptomatic SMA. The study, which is expected to be completed in 2022, recruited participants up to the age of 6 weeks.
The 20 enrolled infants were all alive at a median study period of 317.5 days, and none required assistance with respiration.
In terms of motor function and growth, most were making gains “generally consistent with normal development such as head control, independent sitting, standing and walking independently, as measured by validated scales,” according to a statement released by Biogen.
Three patients developed adverse events that may have been drug related, but the events were resolved and they remained in the trial.
In Dr. Sumner’s discussant presentation on the ENDEAR study, she cautioned that it’s still not known how much older patients with SMA will benefit from the drug treatment. It’s possible that younger patients may gain motor function while older patients will maintain what they have, she said.
In addition, it seems that “certain body segments may respond more robustly than some others. It’s possible that an infant may improve limb strength quite robustly but not lumbar strength.”
She pointed out other questions: Can the drug, which is difficult to administer, be stopped at some point? “We do think of SMA as a disease of development,” she said. “Maybe after development is finished we can withdraw the drug.”
Then there’s the issue of cost and whether insurers are willing to cover the drug. As Dr. Sumner pointed out, the drug is $125,000 per dose and $750,000 over the first year, then $325,000 a year.
It is clear, however, that “the early and efficient diagnosis of SMA is really important, particularly for infantile SMA,” she said. “Time is motor function.”
The studies were funded by Ionis Pharmaceuticals and Biogen. Dr. Sumner reports serving as a consultant for Biogen, Ionis, Avexis, and Roche. She has served on scientific advisory boards for the Cure SMA, SMA Foundation, and Muscular Dystrophy Association nonprofit foundations.
AT AAN 2017
Key clinical point:
Major finding: 51% of subjects who took the drug were judged to be “motor milestone responders”; none in the sham group improved (P less than .0001).
Data source: Randomized, double-blind, sham-controlled, 13-month study of 110 subjects with infantile-onset SMA aged 30-252 days at first dose. They received 12-mg scaled equivalent doses of nusinersen delivered intrathecally (four doses over 2 months then one every 4 months) or a sham procedure (needle prick).
Disclosures: The studies were funded by Ionis Pharmaceuticals and Biogen.
Trump presidency prompts Goldwater Rule debate at APA
SAN DIEGO – Over the past year, the American Psychiatric Association has reasserted its commitment to the Goldwater Rule, yet many psychiatrists have questioned – some publicly – whether the rule needs to be revisited based on their concerns about Donald Trump’s mental fitness to be president.
[polldaddy:9767626]Both sides of the issue got a hearing during a pro and con discussion of the Goldwater Rule at the annual meeting of the American Psychiatric Association. A trio of leading psychiatrists called for the Goldwater Rule to be revised and APA past leadership defended the ethical standards of the existing rule. One past-president of the APA remarked that psychiatrists who object to the rule can choose to quit the APA, a comment that one audience member said harkened back to the Vietnam War–era rebuttal to protesters: “Love it or leave it.”
Several news stories in recent months have highlighted the Trump-spawned debate over the Goldwater Rule, which states that “it is unethical for a psychiatrist to offer a professional opinion unless he or she has conducted an examination and has been granted proper authorization for such a statement.”
The APA created the rule in 1973 after a backlash over the hundreds of U.S. psychiatrists who responded to a magazine survey by diagnosing 1964 GOP presidential candidate Barry Goldwater. He later won a libel suit against the magazine.
Prior to last year’s presidential election, the APA stood by its rule. Then, in March 2017, the APA’s Ethics Committee affirmed that “the rule applies to all professional opinions offered by psychiatrists, not just diagnoses. For example, saying an individual does not have a mental disorder would also constitute a professional opinion.”
During the pro and con discussion at the annual meeting, Paul S. Appelbaum, MD, professor of psychiatry, medicine, and law at Columbia University in New York, called the Goldwater incident “a major embarrassment for psychiatry.”
But, he added, “there are more important justifications than whether we’re embarrassed or not. Part of [the rule] is to protect persons who may be harmed by our blithe speculation to the media. We’re also interested in avoiding an impact on people who may be in need of psychiatric care but are discouraged by what they see and hear about psychiatry in the media,” he said.
“A diagnosis based on casual encounters with a person through the media is necessarily inadequate as a basis for a psychiatric diagnosis or formulation of any reliable sort,” he pointed out.
Claire Pouncey, MD, PhD, a psychiatrist who practices in Philadelphia, questioned why speaking publicly about a public figure is inherently harmful.
“How does it threaten my integrity or integrity of the profession for me to choose to speak publicly about a public figure? The APA seems to not trust its members or recognize us as moral agents who are the arbiters of our own integrity. I wish the APA would trust me,” Dr. Pouncey said.
Last year, she outlined her concerns in an article written with Jerome Kroll, MD, and published in the Journal of the American Academy of Psychiatry and the Law (June 2016;44[2]:226-35).
As for concerns about the adequacy of information, she added, “we never have complete information. And I know we don’t always have a full consent of the person being evaluated. Anyone who’s been in an emergency room knows that in their heart and soul.”
Nassir Ghaemi, MD, MPH, who chaired the session and is professor and director of the mood disorders program at Tufts University in Boston, said that strictly abiding by the Goldwater Rule is “sending out the message that the world’s psychiatrists think it’s bad to get diagnosed with a psychiatric illness. … You are not going to harm patients by talking about psychiatric illness. You help them. We know that.”
Further, Dr. Ghaemi, author of the book “A First-Rate Madness: Uncovering the Links Between Leadership and Mental Illness” (Penguin Books, 2012), argued that the “very qualities that mark those with mood disorders also make for the best leaders in times of crisis.”
President Trump’s reported traits, such as lack of sleep and talkativeness, could be portrayed as mania, he said. However, those qualities also could boost his creativity and resilience.
Jerrold M. Post, MD, a pioneer in profiling for the Central Intelligence Agency and professor of psychiatry, political psychology, and international affairs at George Washington University in Washington, D.C., said that psychiatrists have a role to play as experts, especially now. “I’m increasingly uncomfortable in not having commented on a welter of psychiatric diagnoses by people without psychiatric training,” he said. “It seems unethical to not comment at this time.”
Paul Summergrad, MD, a former APA president and chair of the department of psychiatry at Tufts, dismissed the idea that psychiatrists could mold public debate. “The assumption that we’ll somehow change opinion if we all speak out about these matters is giving us much more credit than we deserve.”
“The reality,” he said, “is that we need to wear a certain yoke of limitation and responsibility rather than thinking this is simply an issue of freedom.”
Dr. Post, Dr. Summergrad, and Dr. Pouncey reported no relevant disclosures. Dr. Ghaemi reported support (consultant/advisory board, speakers bureau, honoraria) from Sunovion. Dr. Appelbaum disclosed stock/other financial relationship with COVR.
[polldaddy:9767626]
SAN DIEGO – Over the past year, the American Psychiatric Association has reasserted its commitment to the Goldwater Rule, yet many psychiatrists have questioned – some publicly – whether the rule needs to be revisited based on their concerns about Donald Trump’s mental fitness to be president.
[polldaddy:9767626]Both sides of the issue got a hearing during a pro and con discussion of the Goldwater Rule at the annual meeting of the American Psychiatric Association. A trio of leading psychiatrists called for the Goldwater Rule to be revised and APA past leadership defended the ethical standards of the existing rule. One past-president of the APA remarked that psychiatrists who object to the rule can choose to quit the APA, a comment that one audience member said harkened back to the Vietnam War–era rebuttal to protesters: “Love it or leave it.”
Several news stories in recent months have highlighted the Trump-spawned debate over the Goldwater Rule, which states that “it is unethical for a psychiatrist to offer a professional opinion unless he or she has conducted an examination and has been granted proper authorization for such a statement.”
The APA created the rule in 1973 after a backlash over the hundreds of U.S. psychiatrists who responded to a magazine survey by diagnosing 1964 GOP presidential candidate Barry Goldwater. He later won a libel suit against the magazine.
Prior to last year’s presidential election, the APA stood by its rule. Then, in March 2017, the APA’s Ethics Committee affirmed that “the rule applies to all professional opinions offered by psychiatrists, not just diagnoses. For example, saying an individual does not have a mental disorder would also constitute a professional opinion.”
During the pro and con discussion at the annual meeting, Paul S. Appelbaum, MD, professor of psychiatry, medicine, and law at Columbia University in New York, called the Goldwater incident “a major embarrassment for psychiatry.”
But, he added, “there are more important justifications than whether we’re embarrassed or not. Part of [the rule] is to protect persons who may be harmed by our blithe speculation to the media. We’re also interested in avoiding an impact on people who may be in need of psychiatric care but are discouraged by what they see and hear about psychiatry in the media,” he said.
“A diagnosis based on casual encounters with a person through the media is necessarily inadequate as a basis for a psychiatric diagnosis or formulation of any reliable sort,” he pointed out.
Claire Pouncey, MD, PhD, a psychiatrist who practices in Philadelphia, questioned why speaking publicly about a public figure is inherently harmful.
“How does it threaten my integrity or integrity of the profession for me to choose to speak publicly about a public figure? The APA seems to not trust its members or recognize us as moral agents who are the arbiters of our own integrity. I wish the APA would trust me,” Dr. Pouncey said.
Last year, she outlined her concerns in an article written with Jerome Kroll, MD, and published in the Journal of the American Academy of Psychiatry and the Law (June 2016;44[2]:226-35).
As for concerns about the adequacy of information, she added, “we never have complete information. And I know we don’t always have a full consent of the person being evaluated. Anyone who’s been in an emergency room knows that in their heart and soul.”
Nassir Ghaemi, MD, MPH, who chaired the session and is professor and director of the mood disorders program at Tufts University in Boston, said that strictly abiding by the Goldwater Rule is “sending out the message that the world’s psychiatrists think it’s bad to get diagnosed with a psychiatric illness. … You are not going to harm patients by talking about psychiatric illness. You help them. We know that.”
Further, Dr. Ghaemi, author of the book “A First-Rate Madness: Uncovering the Links Between Leadership and Mental Illness” (Penguin Books, 2012), argued that the “very qualities that mark those with mood disorders also make for the best leaders in times of crisis.”
President Trump’s reported traits, such as lack of sleep and talkativeness, could be portrayed as mania, he said. However, those qualities also could boost his creativity and resilience.
Jerrold M. Post, MD, a pioneer in profiling for the Central Intelligence Agency and professor of psychiatry, political psychology, and international affairs at George Washington University in Washington, D.C., said that psychiatrists have a role to play as experts, especially now. “I’m increasingly uncomfortable in not having commented on a welter of psychiatric diagnoses by people without psychiatric training,” he said. “It seems unethical to not comment at this time.”
Paul Summergrad, MD, a former APA president and chair of the department of psychiatry at Tufts, dismissed the idea that psychiatrists could mold public debate. “The assumption that we’ll somehow change opinion if we all speak out about these matters is giving us much more credit than we deserve.”
“The reality,” he said, “is that we need to wear a certain yoke of limitation and responsibility rather than thinking this is simply an issue of freedom.”
Dr. Post, Dr. Summergrad, and Dr. Pouncey reported no relevant disclosures. Dr. Ghaemi reported support (consultant/advisory board, speakers bureau, honoraria) from Sunovion. Dr. Appelbaum disclosed stock/other financial relationship with COVR.
[polldaddy:9767626]
SAN DIEGO – Over the past year, the American Psychiatric Association has reasserted its commitment to the Goldwater Rule, yet many psychiatrists have questioned – some publicly – whether the rule needs to be revisited based on their concerns about Donald Trump’s mental fitness to be president.
[polldaddy:9767626]Both sides of the issue got a hearing during a pro and con discussion of the Goldwater Rule at the annual meeting of the American Psychiatric Association. A trio of leading psychiatrists called for the Goldwater Rule to be revised and APA past leadership defended the ethical standards of the existing rule. One past-president of the APA remarked that psychiatrists who object to the rule can choose to quit the APA, a comment that one audience member said harkened back to the Vietnam War–era rebuttal to protesters: “Love it or leave it.”
Several news stories in recent months have highlighted the Trump-spawned debate over the Goldwater Rule, which states that “it is unethical for a psychiatrist to offer a professional opinion unless he or she has conducted an examination and has been granted proper authorization for such a statement.”
The APA created the rule in 1973 after a backlash over the hundreds of U.S. psychiatrists who responded to a magazine survey by diagnosing 1964 GOP presidential candidate Barry Goldwater. He later won a libel suit against the magazine.
Prior to last year’s presidential election, the APA stood by its rule. Then, in March 2017, the APA’s Ethics Committee affirmed that “the rule applies to all professional opinions offered by psychiatrists, not just diagnoses. For example, saying an individual does not have a mental disorder would also constitute a professional opinion.”
During the pro and con discussion at the annual meeting, Paul S. Appelbaum, MD, professor of psychiatry, medicine, and law at Columbia University in New York, called the Goldwater incident “a major embarrassment for psychiatry.”
But, he added, “there are more important justifications than whether we’re embarrassed or not. Part of [the rule] is to protect persons who may be harmed by our blithe speculation to the media. We’re also interested in avoiding an impact on people who may be in need of psychiatric care but are discouraged by what they see and hear about psychiatry in the media,” he said.
“A diagnosis based on casual encounters with a person through the media is necessarily inadequate as a basis for a psychiatric diagnosis or formulation of any reliable sort,” he pointed out.
Claire Pouncey, MD, PhD, a psychiatrist who practices in Philadelphia, questioned why speaking publicly about a public figure is inherently harmful.
“How does it threaten my integrity or integrity of the profession for me to choose to speak publicly about a public figure? The APA seems to not trust its members or recognize us as moral agents who are the arbiters of our own integrity. I wish the APA would trust me,” Dr. Pouncey said.
Last year, she outlined her concerns in an article written with Jerome Kroll, MD, and published in the Journal of the American Academy of Psychiatry and the Law (June 2016;44[2]:226-35).
As for concerns about the adequacy of information, she added, “we never have complete information. And I know we don’t always have a full consent of the person being evaluated. Anyone who’s been in an emergency room knows that in their heart and soul.”
Nassir Ghaemi, MD, MPH, who chaired the session and is professor and director of the mood disorders program at Tufts University in Boston, said that strictly abiding by the Goldwater Rule is “sending out the message that the world’s psychiatrists think it’s bad to get diagnosed with a psychiatric illness. … You are not going to harm patients by talking about psychiatric illness. You help them. We know that.”
Further, Dr. Ghaemi, author of the book “A First-Rate Madness: Uncovering the Links Between Leadership and Mental Illness” (Penguin Books, 2012), argued that the “very qualities that mark those with mood disorders also make for the best leaders in times of crisis.”
President Trump’s reported traits, such as lack of sleep and talkativeness, could be portrayed as mania, he said. However, those qualities also could boost his creativity and resilience.
Jerrold M. Post, MD, a pioneer in profiling for the Central Intelligence Agency and professor of psychiatry, political psychology, and international affairs at George Washington University in Washington, D.C., said that psychiatrists have a role to play as experts, especially now. “I’m increasingly uncomfortable in not having commented on a welter of psychiatric diagnoses by people without psychiatric training,” he said. “It seems unethical to not comment at this time.”
Paul Summergrad, MD, a former APA president and chair of the department of psychiatry at Tufts, dismissed the idea that psychiatrists could mold public debate. “The assumption that we’ll somehow change opinion if we all speak out about these matters is giving us much more credit than we deserve.”
“The reality,” he said, “is that we need to wear a certain yoke of limitation and responsibility rather than thinking this is simply an issue of freedom.”
Dr. Post, Dr. Summergrad, and Dr. Pouncey reported no relevant disclosures. Dr. Ghaemi reported support (consultant/advisory board, speakers bureau, honoraria) from Sunovion. Dr. Appelbaum disclosed stock/other financial relationship with COVR.
[polldaddy:9767626]
EXPERT ANALYSIS FROM APA
VIDEO: Care withdrawal becoming more common in ischemic stroke patients
BOSTON – U.S. physicians chose to withdraw life-sustaining care from critically ill ischemic stroke patients at much higher rates in 2011 than in 2006, according to results of a new study, although overall percentages remain low.
The trend was bolstered by big jumps in “withdrawal of care” among patients who underwent thrombolysis or both thrombolysis and endovascular treatment: Over the 5-year period, their likelihood of having care withdrawn increased fivefold.
The study authors don’t know whether the trends have continued over the past 6 years, and it’s not clear why the rates rose so much from 2006 to 2011. The researchers speculate that the increase could be linked to disease severity and the preferences of patients and their families.
Whatever the case, study lead author Malik Muhammad Adil, MD, a neurology resident at the Ochsner Clinic Foundation in New Orleans, cautioned that prematurely withdrawing care can throw off stroke prognostication estimates that are already fuzzy. In an interview, he said this can then lead to “significant consequences, including suboptimal outcomes and higher risk of short-term mortality.”
Dr. Adil defines withdrawal of care as “discontinuation of life-sustaining interventions from a patient who is expected to die without this support.” These interventions include such treatments as intubation, mechanical ventilation, feeding tubes, antibiotics, and brain surgery, he said.
For the new study, researchers examined the Nationwide Inpatient Survey database for the years 2006-2011. They reported their findings at the annual meeting of the American Academy of Neurology.
The study reports the following regarding withdrawal of care among ischemic stroke patients:
- The rate grew in those who received neither thrombolysis nor endovascular treatment from 0.8% in 2006 to 3.0% in 2011 (P less than or equal to .0001).
- In those who received thrombolysis alone, the rate rose from 0.9% to 5.5% (P less than or equal to .0001).
- In those who received endovascular treatment alone, the rate increased from 2.8% to 9.0% (P = .0006).
- In patients who received both thrombolysis and endovascular treatment, the rate grew from 2.0% to 10.3% (P = .0009).
Dr. Adil said several factors can affect rates of withdrawal of care in ischemic stroke patients, such as the level of illness (patients receiving aggressive treatment are sicker), advance directives, and the decisions of family members. Some institutions may be more likely to push for withdrawal of care, too, he said. “At my institution, we are not aggressive with withdrawal of care, and I have seen a few better outcomes than expected,” he said.
Also, the lack of useful data regarding prognosis for these patients may lead to premature decisions regarding withdrawal of care, he said.
“We have few prognostic models/scores that predict mortality, and these models are not very sensitive and specific,” Dr. Adil said. “Often, physicians make these decisions based on their previous experiences. All of this leads to premature withdrawal of care. On the other hand, because of premature withdrawal of care, we do not have the data on long-term outcomes on these patients, leading to errors in prognostication.”
In an interview, Adam G. Kelly, MD, a neurologist at the University of Rochester (N.Y.) and chief of neurology at Highland Hospital in Rochester, said the study is important but lacks crucial information such as the circumstances surrounding the care decisions. He hasn’t noticed trends in withdrawal of care among his patients.
“It’s possible that providers have become better at documenting discussions with patients and families which allowed this information to be better captured,” he said. “As the authors mention, it’s also possible that providers are consciously or unconsciously delivering prognoses that are biased towards negative outcomes, leading patients and families to be more apt to choose a palliative approach.”
Dr. Kelly added that neurologists need to objectively offer prognoses in stroke cases. “When the outcome is in doubt, I recommend time-limited trials of interventions of mechanical ventilation, artificial feeding, and other high-intensity interventions to allow patients and families to make the decision they feel is best,” he said.
What about trends in the years since 2011? Dr. Adil said information regarding the years since 2011 wasn’t available to him, but he hopes to analyze the period from 2012 to 2016.
Dr. Adil discussed his study and its implications in a video interview.
No funding is reported. Dr. Adil reports no relevant disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
cnnews@frontlinemedcom.com
BOSTON – U.S. physicians chose to withdraw life-sustaining care from critically ill ischemic stroke patients at much higher rates in 2011 than in 2006, according to results of a new study, although overall percentages remain low.
The trend was bolstered by big jumps in “withdrawal of care” among patients who underwent thrombolysis or both thrombolysis and endovascular treatment: Over the 5-year period, their likelihood of having care withdrawn increased fivefold.
The study authors don’t know whether the trends have continued over the past 6 years, and it’s not clear why the rates rose so much from 2006 to 2011. The researchers speculate that the increase could be linked to disease severity and the preferences of patients and their families.
Whatever the case, study lead author Malik Muhammad Adil, MD, a neurology resident at the Ochsner Clinic Foundation in New Orleans, cautioned that prematurely withdrawing care can throw off stroke prognostication estimates that are already fuzzy. In an interview, he said this can then lead to “significant consequences, including suboptimal outcomes and higher risk of short-term mortality.”
Dr. Adil defines withdrawal of care as “discontinuation of life-sustaining interventions from a patient who is expected to die without this support.” These interventions include such treatments as intubation, mechanical ventilation, feeding tubes, antibiotics, and brain surgery, he said.
For the new study, researchers examined the Nationwide Inpatient Survey database for the years 2006-2011. They reported their findings at the annual meeting of the American Academy of Neurology.
The study reports the following regarding withdrawal of care among ischemic stroke patients:
- The rate grew in those who received neither thrombolysis nor endovascular treatment from 0.8% in 2006 to 3.0% in 2011 (P less than or equal to .0001).
- In those who received thrombolysis alone, the rate rose from 0.9% to 5.5% (P less than or equal to .0001).
- In those who received endovascular treatment alone, the rate increased from 2.8% to 9.0% (P = .0006).
- In patients who received both thrombolysis and endovascular treatment, the rate grew from 2.0% to 10.3% (P = .0009).
Dr. Adil said several factors can affect rates of withdrawal of care in ischemic stroke patients, such as the level of illness (patients receiving aggressive treatment are sicker), advance directives, and the decisions of family members. Some institutions may be more likely to push for withdrawal of care, too, he said. “At my institution, we are not aggressive with withdrawal of care, and I have seen a few better outcomes than expected,” he said.
Also, the lack of useful data regarding prognosis for these patients may lead to premature decisions regarding withdrawal of care, he said.
“We have few prognostic models/scores that predict mortality, and these models are not very sensitive and specific,” Dr. Adil said. “Often, physicians make these decisions based on their previous experiences. All of this leads to premature withdrawal of care. On the other hand, because of premature withdrawal of care, we do not have the data on long-term outcomes on these patients, leading to errors in prognostication.”
In an interview, Adam G. Kelly, MD, a neurologist at the University of Rochester (N.Y.) and chief of neurology at Highland Hospital in Rochester, said the study is important but lacks crucial information such as the circumstances surrounding the care decisions. He hasn’t noticed trends in withdrawal of care among his patients.
“It’s possible that providers have become better at documenting discussions with patients and families which allowed this information to be better captured,” he said. “As the authors mention, it’s also possible that providers are consciously or unconsciously delivering prognoses that are biased towards negative outcomes, leading patients and families to be more apt to choose a palliative approach.”
Dr. Kelly added that neurologists need to objectively offer prognoses in stroke cases. “When the outcome is in doubt, I recommend time-limited trials of interventions of mechanical ventilation, artificial feeding, and other high-intensity interventions to allow patients and families to make the decision they feel is best,” he said.
What about trends in the years since 2011? Dr. Adil said information regarding the years since 2011 wasn’t available to him, but he hopes to analyze the period from 2012 to 2016.
Dr. Adil discussed his study and its implications in a video interview.
No funding is reported. Dr. Adil reports no relevant disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
cnnews@frontlinemedcom.com
BOSTON – U.S. physicians chose to withdraw life-sustaining care from critically ill ischemic stroke patients at much higher rates in 2011 than in 2006, according to results of a new study, although overall percentages remain low.
The trend was bolstered by big jumps in “withdrawal of care” among patients who underwent thrombolysis or both thrombolysis and endovascular treatment: Over the 5-year period, their likelihood of having care withdrawn increased fivefold.
The study authors don’t know whether the trends have continued over the past 6 years, and it’s not clear why the rates rose so much from 2006 to 2011. The researchers speculate that the increase could be linked to disease severity and the preferences of patients and their families.
Whatever the case, study lead author Malik Muhammad Adil, MD, a neurology resident at the Ochsner Clinic Foundation in New Orleans, cautioned that prematurely withdrawing care can throw off stroke prognostication estimates that are already fuzzy. In an interview, he said this can then lead to “significant consequences, including suboptimal outcomes and higher risk of short-term mortality.”
Dr. Adil defines withdrawal of care as “discontinuation of life-sustaining interventions from a patient who is expected to die without this support.” These interventions include such treatments as intubation, mechanical ventilation, feeding tubes, antibiotics, and brain surgery, he said.
For the new study, researchers examined the Nationwide Inpatient Survey database for the years 2006-2011. They reported their findings at the annual meeting of the American Academy of Neurology.
The study reports the following regarding withdrawal of care among ischemic stroke patients:
- The rate grew in those who received neither thrombolysis nor endovascular treatment from 0.8% in 2006 to 3.0% in 2011 (P less than or equal to .0001).
- In those who received thrombolysis alone, the rate rose from 0.9% to 5.5% (P less than or equal to .0001).
- In those who received endovascular treatment alone, the rate increased from 2.8% to 9.0% (P = .0006).
- In patients who received both thrombolysis and endovascular treatment, the rate grew from 2.0% to 10.3% (P = .0009).
Dr. Adil said several factors can affect rates of withdrawal of care in ischemic stroke patients, such as the level of illness (patients receiving aggressive treatment are sicker), advance directives, and the decisions of family members. Some institutions may be more likely to push for withdrawal of care, too, he said. “At my institution, we are not aggressive with withdrawal of care, and I have seen a few better outcomes than expected,” he said.
Also, the lack of useful data regarding prognosis for these patients may lead to premature decisions regarding withdrawal of care, he said.
“We have few prognostic models/scores that predict mortality, and these models are not very sensitive and specific,” Dr. Adil said. “Often, physicians make these decisions based on their previous experiences. All of this leads to premature withdrawal of care. On the other hand, because of premature withdrawal of care, we do not have the data on long-term outcomes on these patients, leading to errors in prognostication.”
In an interview, Adam G. Kelly, MD, a neurologist at the University of Rochester (N.Y.) and chief of neurology at Highland Hospital in Rochester, said the study is important but lacks crucial information such as the circumstances surrounding the care decisions. He hasn’t noticed trends in withdrawal of care among his patients.
“It’s possible that providers have become better at documenting discussions with patients and families which allowed this information to be better captured,” he said. “As the authors mention, it’s also possible that providers are consciously or unconsciously delivering prognoses that are biased towards negative outcomes, leading patients and families to be more apt to choose a palliative approach.”
Dr. Kelly added that neurologists need to objectively offer prognoses in stroke cases. “When the outcome is in doubt, I recommend time-limited trials of interventions of mechanical ventilation, artificial feeding, and other high-intensity interventions to allow patients and families to make the decision they feel is best,” he said.
What about trends in the years since 2011? Dr. Adil said information regarding the years since 2011 wasn’t available to him, but he hopes to analyze the period from 2012 to 2016.
Dr. Adil discussed his study and its implications in a video interview.
No funding is reported. Dr. Adil reports no relevant disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
cnnews@frontlinemedcom.com
Key clinical point:
Major finding: The percentages of ischemic stroke patients whose care was withdrawn grew from 0.8%-2.8% in 2006 to 3.0%-10.3% in 2011, depending on the kind of treatment they received (thrombolysis, endovascular treatment, both, or neither).
Data source: Nationwide Inpatient Survey database.
Disclosures: No funding is reported. Dr. Adil reports no relevant disclosures.
Schizophrenia researchers seek elusive ‘quantum leap’
Mental health researchers like to say a half-century has passed since the last major advance in schizophrenia treatment came along. Gavin P. Reynolds, PhD, a schizophrenia researcher, says that’s wrong. In fact, he says, “it’s more like 60-plus!”
Yes, there have been advances in drug treatment since the early 1950s, when chlorpromazine (Thorazine) was introduced. Available are new types of antipsychotics, a whole bunch in fact, and they’ve helped many patients. “But these have been incremental, rather than the much-needed quantum leap,” said Dr. Reynolds in an interview. “We still need improved treatments. About one-third of patients do not respond to standard drug treatment. And of those who do respond, the negative symptoms and cognitive problems caused by schizophrenia may still be very limiting.”
That’s quite optimistic. The “revolution” hasn’t yet jumped from medical journals and clinical trials to prescription pads and drugstore shelves, and it’s not likely to do so any time soon. “This isn’t around the corner,” said Joshua T. Kantrowitz, MD, also a schizophrenia researcher. “But I can imagine a day where someone with schizophrenia will undergo a full genetic scan or a specific type of MRI or EEG, and we’ll then be able to recommend the drug they’d be able to use.” And he believes that a glutamate-based medication will be among the available options.
Thorazine: A pioneer with major limits
Like every person, each illness has a history. But schizophrenia’s past is fuzzier than that of many diseases, and this lack of clarity continues into the present as researchers try to understand exactly what it is – a single disorder? a collection of conditions? – and what it isn’t.
“Schizophrenia is a serious mental illness with a remarkably short recorded history. Unlike depression and mania, which are recognizable in ancient texts, schizophrenia-like disorder appeared rather suddenly in the early 19th century,” wrote R. Walter Heinrichs, PhD, a psychologist at Toronto’s York University (J Hist Behav Sci. 2003 Fall;39[4]:349-63). “This could mean that the illness is a recent disease that was largely unknown in earlier times. But perhaps schizophrenia existed, embedded and disguised within more general concepts of madness, and within the arcane languages and cultures of remote times,” he wrote.
As Dr. Heinrichs noted, schizophrenia’s history has spawned at least three theories: It’s a fairly new disease that just popped up in recent centuries. It has been around a long time but just didn’t get identified. It’s not a real disease but a product of modern thought.
Psychiatrists and researchers have rejected the latter possibility and its implications that psychotherapy could be the best treatment. Instead, they have tried to adjust the workings of the schizophrenic brain via medication.
The main breakthrough came in the 1950s through the development of the antipsychotic chlorpromazine. Its success “was instrumental in the reintegration of psychiatry with the other medical disciplines,” wrote Thomas A. Ban, MD, an emeritus professor at Vanderbilt University, Nashville, Tenn. “It turned psychiatrists from caregivers to full-fledged physicians who can help their patients and not only listen to their problems” (Neuropsychiatr Dis Treat. 2007 Aug; 3[4]:495-500).
Since chlorpromazine, however, medical treatment for schizophrenia has barely evolved, said Dr. Reynolds, professor emeritus at Queen’s University Belfast (Northern Ireland) and honorary professor at Sheffield (England) Hallam University. “The two most important developments have been the introduction of clozapine (Clozaril) for patients who don’t respond to other treatments and aripiprazole (Abilify), which has a somewhat different pharmacological action from other antipsychotics and thereby avoids some of the side effects.”
Negative symptoms fail to crumble
But, he said, side effects such as severe weight gain still can hamper the use of antipsychotics. And antipsychotics don’t fare well at treating the negative symptoms of schizophrenia.
As one overview puts it, “negative symptoms, e.g., social withdrawal, reduced initiative, anhedonia, and affective flattening, are notoriously difficult to treat.” Nonmedical treatments have shown some promise, the overview authors write: “Some positive findings have been reported, with the most robust improvements observed for social skills training. Although cognitive-behavior therapy shows significant effects for negative symptoms as a secondary outcome measure, there is a lack of data to allow for definite conclusions of its effectiveness for patients with predominant negative symptoms.”
As for medications, “antipsychotics have been shown to improve negative symptoms, but this seems to be limited to secondary negative symptoms in acute patients. It has also been suggested that antipsychotics may aggravate negative symptoms” (Schizophr Res. 2016 Jun 9. doi: 10.1016/j.schres.2016.05.015).
Looking to dopamine... and beyond
One solution to the vexing schizophrenia treatment gap is to develop a new medication that does a better job of adjusting the brain’s processing of dopamine.
Some researchers have found evidence that dopamine levels rise in the brains of people with schizophrenia, potentially explaining why chlorpromazine – which blocks receptors that process dopamine – is so effective. There also are signs that genes linked to dopamine play a role in schizophrenia.
But chlorpromazine is not effective enough to help many patients. Some gain no benefit, while others only see improvement in positive schizophrenia symptoms, those that are considered to be brought on – added – such as hallucinations and delusions.
And chlorpromazine isn’t believed to improve cognitive symptoms of schizophrenia like disorganized thoughts and concentration difficulties.
Atypical antipsychotics like clozapine (Clozaril) and risperidone (Risperdal), add the blocking of serotonin receptors to their dopamine focus. But they have serious limitations just like chlorpromazine.
The conclusion to take from the failure of dopamine-based drugs to fully combat schizophrenia, Dr. Kantrowitz and a colleague wrote, is that “dopaminergic dysfunction appears to account for only a part of schizophrenia’s symptomatic and neurocognitive profile.” Something else must be going on.
Enter the neurotransmitter known as glutamate.
The angel dust connection
Children who grew up in the 1970s and 1980s were flooded plenty of media messages about the dangers of illegal drugs. But one drug, “angel dust,” also known as phencyclidine or PCP, stood apart from the rest.
Using PCP, television shows and films suggested, led to consequences that could be deadly in ways that were entirely unlike the usual causes of death by drug use – a car accident, say, or a heroin overdose. Taking PCP, it seemed, was tied to crazed misadventure.
Consider a 1982 TV movie, “Desperate Lives,” starring a young actress named Helen Hunt. In a startling scene, she takes the drug and promptly jumps through a second-story window before screeching and cutting herself with jagged glass. The portrayal of PCP users as vividly unhinged – detached from reality and willing to hurt themselves or others – reflected a real phenomenon. As scientists had discovered, angel dust could made people act as if they were psychotic.
In the 1960s, “people started observing that drugs like PCP and ketamine produced schizophrenia-like symptoms,” Dr. Kantrowitz said. “But these observations were mostly ignored until the 1980s and 1990s.”
That’s when scientists started looking into the connections between these drugs and the psychotic states that they could induce. “These drugs can mimic, in animals and humans, the negative and cognitive symptoms as well as the positive symptoms of schizophrenia,” Dr. Reynolds said. “Combined with postmortem evidence of changes in various indicators of glutamate neurotransmission in the brain, these findings provide the basis for the glutamate hypothesis.”
The hypothesis theorizes that disruptions in the brain’s processing of glutamate are crucial to schizophrenia. Specifically, scientists believe the key lies in the N-methyl-D-aspartate (NMDA) receptor in nerve cells.
“Glutamatergic models are based upon the observation that the psychotomimetic agents such as phencyclidine and ketamine induce psychotic symptoms and neurocognitive disturbances similar to those of schizophrenia by blocking neurotransmission at N-methyl-D-aspartate (NMDA)-type glutamate receptors,” writes Daniel C. Javitt, MD, professor of psychiatry and director of schizophrenia research at the Nathan Kline Institute for Psychiatric Research at Columbia University. “Because glutamate/NMDA receptors are located throughout the brain, glutamatergic models predict widespread cortical dysfunction with particular involvement of NMDA receptors throughout the brain.
“Further, NMDA receptors are located on brain circuits that regulate dopamine release, suggesting that dopaminergic deficits in schizophrenia may also be secondary to underlying glutamatergic dysfunction.” (Isr J Psychiatry Relat Sci. 2010;47[1]:4-16).
There’s even more to the story, said Dr. Reynolds, who cautions that the term “glutamate hypothesis” is misleading, because it misses the whole picture.
“The original research identifying the importance of the NMDA receptor highlighted how these receptors were actually on GABA neurons, reflecting the close interaction and interdependence of the two transmitter systems,” he said. (GABA receptors respond to the neurotransmitter gamma-aminobutyric acid, or GABA.)
“Subsequently, it has been established that there is a dysfunction, if not a deficit, of a subgroup of GABA neurons in schizophrenia,” Dr. Reynolds said, “and these neurons, which are important in the control of glutamate neuronal activity, are also a focus of current research.”
Challenges on the drug development front
Earlier findings linking glutamate to schizophrenia “have been strengthened by recent large genetic studies identifying that variability in some genes involved in glutamate’s action is a risk factor for schizophrenia,” Dr. Reynolds said, “while in animal models, some drugs influencing glutamate transmission have shown promise as potential treatments.”
But glutamate-based drugs for schizophrenia remain elusive. “It is disappointing that the new antipsychotic drugs becoming available now are still all variations on the dopamine antagonist or partial agonist theme, even if they have some advantages in terms of side effects and evidence that they may have some limited effects on negative or cognitive symptoms,” Dr. Reynolds said. “Several drugs addressing glutamate systems – indirectly modulating the NMDA receptor or synaptic glutamate – have been developed that had good potential but have failed in clinical trials. Nevertheless, this strategy is still being pursued by the pharma industry.”
Dr. Reynolds points to drug development challenges on several fronts. For one, research subjects don’t necessarily represent patients who’d benefit the most from treatment.
“Drug treatments are likely to be most effective, and most important, in the early stages of the disease,” he said. “However, drug trials are almost inevitably undertaken on subjects who may have had many years of treatment prior to the trial without, perhaps, always responding very well.”
Another challenge: The tight interconnectedness of neural systems linked to schizophrenia. Tinkering with something in one part of the brain may lead to something going wrong somewhere else. “Glutamate and GABA neurons are ubiquitous throughout the brain,” Dr. Reynolds said, “and targeting systems particularly affected in schizophrenia is difficult without influencing other neuronal systems, potentially leading to further side effects.”
Targets offer hope for new medications
The challenges are significant, but the latest schizophrenia research also is opening up opportunities for new medications.
The GABA connection, for example, is a fertile field for researchers. “One interesting and novel approach that seems promising is the development of a drug that modulates ion channels in GABA/parvalbumin neurons, thereby potentially restoring their function,” Dr. Reynolds said. “We are still years away from availability of these new treatments, however.”
But what if drugs targeted specific types of schizophrenia just like antibiotics aim to treat different types of bacterial infections?
Dr. Tamminga is pushing researchers to focus on how disruptions in glutamate processing affect specific parts of the brain and specific brain functions. “Some people want to have a single glutamate defect throughout the whole brain,” she said. But she notes that “the glutamate system is prominent in every brain region,” suggesting that a global problem would entirely incapacitate schizophrenia patients.
In fact, “while people with schizophrenia do have glutamate-mediated troubles with their condition, with their memory, they don’t have glutamate problems with their motor system or sensory system,” she said. “There’s very little evidence for a generalized brain defect. We ought to be taking another tack: We need to figure out pathophysiology by region and brain function.”
Dr. Kantrowitz also is working on developing biomarkers. He said he’s collaborating with colleagues to follow directives by the National Institute on Mental Health to develop schizophrenia biomarkers in early-stage studies.
“It’s a way to assess how well a specific drug is hitting its target,” he said. “Once you’ve developed a good understanding of how well they’re hitting their target, then you can more finely tune doses before you put the drugs in thousands of people.”
The research may shed light on a question that continues to divide schizophrenia researchers: Is the disease one single condition or multiple disorders masquerading as one?
“I think that eventually, once we get better at genetics and looking at schizophrenia, we’ll realize it’s not one thing, that it’s many different things,” Dr. Kantrowitz said. “The brain is a very complex organ, and there are lots of different areas of things that can go wrong to produce similar symptoms that are grouped together as schizophrenia.”
It’s possible, he believes, that one group of schizophrenia patients, maybe 10 percent, will respond to a glutamate-based treatment. An additional 10 percent, perhaps will require something else.
“We’re not good enough to discern these things yet,” he said. “But eventually we’ll get to more targeted treatment.”
And then we’ll finally get to reset the clock marking the time – now almost 70 years – since the last major milestone in schizophrenia treatment.
Dr. Reynolds reported that over the past 3 years, he has received honoraria for lectures, advisory board membership, travel support and/or research support from Janssen, Lundbeck, Otsuka, Sunovion, and Sumitomo. Dr. Kantrowitz reported having received consulting payments within the last 24 months from Vindico Medical Education, Annenberg Center for Health Sciences at Eisenhower, Health Advances, SlingShot, Strategic Edge Communications, Havas Life, and Cowen and Company. He has conducted clinical research supported by the NIMH, the Stanley Foundation, Merck, Roche-Genentech, Forum, Sunovion, Novartis, Lundbeck, Alkermes, NeuroRx, Pfizer, and Lilly. He owns a small number of shares of common stock in GlaxoSmithKline.
Dr. Tamminga reported no relevant disclosures.
Lessons from the latest research
One major thrust of research is to understand exactly how the glutamate system works and what goes wrong in schizophrenia patients. Here are some questions and answers from recent research:
Question: If NMDA receptors are crucial to glutamate, would an NMDA receptor channel blocker help improve symptoms in schizophrenia patients? What about memantine (Namenda), an Alzheimer’s disease medication that treats the cognitive problems caused by dementia?
Answer: A 2017 systematic review of 10 studies (including two case reports) found “unclear results”: “memantine therapy in schizophrenic patients seems to improve mainly negative symptoms while positive symptoms and cognitive symptoms did not improve significantly.”
The researchers add that the “use of memantine should be considered in patients with prevalent negative symptoms and cognitive impairment, even if further trials are required. Memantine could be a new opportunity to treat young patients in order to prevent further cognitive decline that will lead to global impairment” (J Amino Acids. 2017;2017:7021071).
The review did not include a 2017 double-blind, placebo-controlled study of 46 adult male patients (average age around 45 years) comparing the antipsychotic risperidone with risperidone plus memantine. The researchers found no difference in positive symptom improvement between the two groups, but the combination treatment group showed significant improvement in cognitive function (at 6 weeks and study completion at 12 weeks) and negative symptoms (at 12 weeks) (Psychiatry Res. 2017 Jan;247:291-5).
Q: We know that the cognitive symptoms of schizophrenia are difficult to treat. What hope does the glutamate system hold in this area?
A: A 2017 systematic review of 44 studies says “memory, working memory and executive functions appear to be most influenced by the glutamatergic pathway.” But there’s some added complexity: “evidence from the literature suggests that presynaptic components synthesis and uptake of glutamate is involved in memory, while postsynaptic signaling appears to be involved in working memory.”
In the big picture, the review says, “the glutamatergic system appears to contribute to the cognitive deficits in schizophrenia, whereby different parts of the pathway are associated with different cognitive domains” (Neurosci Biobehav Rev. 2017 Apr 13;77:369-87).
Q: Could imaging via proton magnetic resonance spectroscopy prove a link between schizophrenia and certain kinds of glutamate activity in the brain, giving mental health professionals a tool to identify at-risk people who are most likely to develop psychosis?
A: A 2016 meta-analysis examined 59 studies and reported that “schizophrenia is associated with elevations in glutamatergic metabolites across several brain regions. This finding supports the hypothesis that schizophrenia is associated with excess glutamatergic neurotransmission in several limbic areas and further indicates that compounds that reduce glutamatergic transmission may have therapeutic potential” (JAMA Psychiatry. 2016 Jul 1;73[7]:665-74).
A review published in 2016 examined 11 proton magnetic resonance spectroscopy studies in search of signs that the state of the glutamate system could predict future psychosis.
Researchers found that six studies reported “significant alterations in glutamate metabolites across different cerebral areas (frontal lobe, thalamus, and the associative striatum)” in at-risk patients. “A longitudinal analysis in two of these trials confirmed an association between these abnormalities and worsening of symptoms and final transition to psychosis.”
But the other five studies analyzed in the review “found no significant differences across these same areas” (Schizophr Res. 2016 Mar;171[1-3]:166-75).
Mental health researchers like to say a half-century has passed since the last major advance in schizophrenia treatment came along. Gavin P. Reynolds, PhD, a schizophrenia researcher, says that’s wrong. In fact, he says, “it’s more like 60-plus!”
Yes, there have been advances in drug treatment since the early 1950s, when chlorpromazine (Thorazine) was introduced. Available are new types of antipsychotics, a whole bunch in fact, and they’ve helped many patients. “But these have been incremental, rather than the much-needed quantum leap,” said Dr. Reynolds in an interview. “We still need improved treatments. About one-third of patients do not respond to standard drug treatment. And of those who do respond, the negative symptoms and cognitive problems caused by schizophrenia may still be very limiting.”
That’s quite optimistic. The “revolution” hasn’t yet jumped from medical journals and clinical trials to prescription pads and drugstore shelves, and it’s not likely to do so any time soon. “This isn’t around the corner,” said Joshua T. Kantrowitz, MD, also a schizophrenia researcher. “But I can imagine a day where someone with schizophrenia will undergo a full genetic scan or a specific type of MRI or EEG, and we’ll then be able to recommend the drug they’d be able to use.” And he believes that a glutamate-based medication will be among the available options.
Thorazine: A pioneer with major limits
Like every person, each illness has a history. But schizophrenia’s past is fuzzier than that of many diseases, and this lack of clarity continues into the present as researchers try to understand exactly what it is – a single disorder? a collection of conditions? – and what it isn’t.
“Schizophrenia is a serious mental illness with a remarkably short recorded history. Unlike depression and mania, which are recognizable in ancient texts, schizophrenia-like disorder appeared rather suddenly in the early 19th century,” wrote R. Walter Heinrichs, PhD, a psychologist at Toronto’s York University (J Hist Behav Sci. 2003 Fall;39[4]:349-63). “This could mean that the illness is a recent disease that was largely unknown in earlier times. But perhaps schizophrenia existed, embedded and disguised within more general concepts of madness, and within the arcane languages and cultures of remote times,” he wrote.
As Dr. Heinrichs noted, schizophrenia’s history has spawned at least three theories: It’s a fairly new disease that just popped up in recent centuries. It has been around a long time but just didn’t get identified. It’s not a real disease but a product of modern thought.
Psychiatrists and researchers have rejected the latter possibility and its implications that psychotherapy could be the best treatment. Instead, they have tried to adjust the workings of the schizophrenic brain via medication.
The main breakthrough came in the 1950s through the development of the antipsychotic chlorpromazine. Its success “was instrumental in the reintegration of psychiatry with the other medical disciplines,” wrote Thomas A. Ban, MD, an emeritus professor at Vanderbilt University, Nashville, Tenn. “It turned psychiatrists from caregivers to full-fledged physicians who can help their patients and not only listen to their problems” (Neuropsychiatr Dis Treat. 2007 Aug; 3[4]:495-500).
Since chlorpromazine, however, medical treatment for schizophrenia has barely evolved, said Dr. Reynolds, professor emeritus at Queen’s University Belfast (Northern Ireland) and honorary professor at Sheffield (England) Hallam University. “The two most important developments have been the introduction of clozapine (Clozaril) for patients who don’t respond to other treatments and aripiprazole (Abilify), which has a somewhat different pharmacological action from other antipsychotics and thereby avoids some of the side effects.”
Negative symptoms fail to crumble
But, he said, side effects such as severe weight gain still can hamper the use of antipsychotics. And antipsychotics don’t fare well at treating the negative symptoms of schizophrenia.
As one overview puts it, “negative symptoms, e.g., social withdrawal, reduced initiative, anhedonia, and affective flattening, are notoriously difficult to treat.” Nonmedical treatments have shown some promise, the overview authors write: “Some positive findings have been reported, with the most robust improvements observed for social skills training. Although cognitive-behavior therapy shows significant effects for negative symptoms as a secondary outcome measure, there is a lack of data to allow for definite conclusions of its effectiveness for patients with predominant negative symptoms.”
As for medications, “antipsychotics have been shown to improve negative symptoms, but this seems to be limited to secondary negative symptoms in acute patients. It has also been suggested that antipsychotics may aggravate negative symptoms” (Schizophr Res. 2016 Jun 9. doi: 10.1016/j.schres.2016.05.015).
Looking to dopamine... and beyond
One solution to the vexing schizophrenia treatment gap is to develop a new medication that does a better job of adjusting the brain’s processing of dopamine.
Some researchers have found evidence that dopamine levels rise in the brains of people with schizophrenia, potentially explaining why chlorpromazine – which blocks receptors that process dopamine – is so effective. There also are signs that genes linked to dopamine play a role in schizophrenia.
But chlorpromazine is not effective enough to help many patients. Some gain no benefit, while others only see improvement in positive schizophrenia symptoms, those that are considered to be brought on – added – such as hallucinations and delusions.
And chlorpromazine isn’t believed to improve cognitive symptoms of schizophrenia like disorganized thoughts and concentration difficulties.
Atypical antipsychotics like clozapine (Clozaril) and risperidone (Risperdal), add the blocking of serotonin receptors to their dopamine focus. But they have serious limitations just like chlorpromazine.
The conclusion to take from the failure of dopamine-based drugs to fully combat schizophrenia, Dr. Kantrowitz and a colleague wrote, is that “dopaminergic dysfunction appears to account for only a part of schizophrenia’s symptomatic and neurocognitive profile.” Something else must be going on.
Enter the neurotransmitter known as glutamate.
The angel dust connection
Children who grew up in the 1970s and 1980s were flooded plenty of media messages about the dangers of illegal drugs. But one drug, “angel dust,” also known as phencyclidine or PCP, stood apart from the rest.
Using PCP, television shows and films suggested, led to consequences that could be deadly in ways that were entirely unlike the usual causes of death by drug use – a car accident, say, or a heroin overdose. Taking PCP, it seemed, was tied to crazed misadventure.
Consider a 1982 TV movie, “Desperate Lives,” starring a young actress named Helen Hunt. In a startling scene, she takes the drug and promptly jumps through a second-story window before screeching and cutting herself with jagged glass. The portrayal of PCP users as vividly unhinged – detached from reality and willing to hurt themselves or others – reflected a real phenomenon. As scientists had discovered, angel dust could made people act as if they were psychotic.
In the 1960s, “people started observing that drugs like PCP and ketamine produced schizophrenia-like symptoms,” Dr. Kantrowitz said. “But these observations were mostly ignored until the 1980s and 1990s.”
That’s when scientists started looking into the connections between these drugs and the psychotic states that they could induce. “These drugs can mimic, in animals and humans, the negative and cognitive symptoms as well as the positive symptoms of schizophrenia,” Dr. Reynolds said. “Combined with postmortem evidence of changes in various indicators of glutamate neurotransmission in the brain, these findings provide the basis for the glutamate hypothesis.”
The hypothesis theorizes that disruptions in the brain’s processing of glutamate are crucial to schizophrenia. Specifically, scientists believe the key lies in the N-methyl-D-aspartate (NMDA) receptor in nerve cells.
“Glutamatergic models are based upon the observation that the psychotomimetic agents such as phencyclidine and ketamine induce psychotic symptoms and neurocognitive disturbances similar to those of schizophrenia by blocking neurotransmission at N-methyl-D-aspartate (NMDA)-type glutamate receptors,” writes Daniel C. Javitt, MD, professor of psychiatry and director of schizophrenia research at the Nathan Kline Institute for Psychiatric Research at Columbia University. “Because glutamate/NMDA receptors are located throughout the brain, glutamatergic models predict widespread cortical dysfunction with particular involvement of NMDA receptors throughout the brain.
“Further, NMDA receptors are located on brain circuits that regulate dopamine release, suggesting that dopaminergic deficits in schizophrenia may also be secondary to underlying glutamatergic dysfunction.” (Isr J Psychiatry Relat Sci. 2010;47[1]:4-16).
There’s even more to the story, said Dr. Reynolds, who cautions that the term “glutamate hypothesis” is misleading, because it misses the whole picture.
“The original research identifying the importance of the NMDA receptor highlighted how these receptors were actually on GABA neurons, reflecting the close interaction and interdependence of the two transmitter systems,” he said. (GABA receptors respond to the neurotransmitter gamma-aminobutyric acid, or GABA.)
“Subsequently, it has been established that there is a dysfunction, if not a deficit, of a subgroup of GABA neurons in schizophrenia,” Dr. Reynolds said, “and these neurons, which are important in the control of glutamate neuronal activity, are also a focus of current research.”
Challenges on the drug development front
Earlier findings linking glutamate to schizophrenia “have been strengthened by recent large genetic studies identifying that variability in some genes involved in glutamate’s action is a risk factor for schizophrenia,” Dr. Reynolds said, “while in animal models, some drugs influencing glutamate transmission have shown promise as potential treatments.”
But glutamate-based drugs for schizophrenia remain elusive. “It is disappointing that the new antipsychotic drugs becoming available now are still all variations on the dopamine antagonist or partial agonist theme, even if they have some advantages in terms of side effects and evidence that they may have some limited effects on negative or cognitive symptoms,” Dr. Reynolds said. “Several drugs addressing glutamate systems – indirectly modulating the NMDA receptor or synaptic glutamate – have been developed that had good potential but have failed in clinical trials. Nevertheless, this strategy is still being pursued by the pharma industry.”
Dr. Reynolds points to drug development challenges on several fronts. For one, research subjects don’t necessarily represent patients who’d benefit the most from treatment.
“Drug treatments are likely to be most effective, and most important, in the early stages of the disease,” he said. “However, drug trials are almost inevitably undertaken on subjects who may have had many years of treatment prior to the trial without, perhaps, always responding very well.”
Another challenge: The tight interconnectedness of neural systems linked to schizophrenia. Tinkering with something in one part of the brain may lead to something going wrong somewhere else. “Glutamate and GABA neurons are ubiquitous throughout the brain,” Dr. Reynolds said, “and targeting systems particularly affected in schizophrenia is difficult without influencing other neuronal systems, potentially leading to further side effects.”
Targets offer hope for new medications
The challenges are significant, but the latest schizophrenia research also is opening up opportunities for new medications.
The GABA connection, for example, is a fertile field for researchers. “One interesting and novel approach that seems promising is the development of a drug that modulates ion channels in GABA/parvalbumin neurons, thereby potentially restoring their function,” Dr. Reynolds said. “We are still years away from availability of these new treatments, however.”
But what if drugs targeted specific types of schizophrenia just like antibiotics aim to treat different types of bacterial infections?
Dr. Tamminga is pushing researchers to focus on how disruptions in glutamate processing affect specific parts of the brain and specific brain functions. “Some people want to have a single glutamate defect throughout the whole brain,” she said. But she notes that “the glutamate system is prominent in every brain region,” suggesting that a global problem would entirely incapacitate schizophrenia patients.
In fact, “while people with schizophrenia do have glutamate-mediated troubles with their condition, with their memory, they don’t have glutamate problems with their motor system or sensory system,” she said. “There’s very little evidence for a generalized brain defect. We ought to be taking another tack: We need to figure out pathophysiology by region and brain function.”
Dr. Kantrowitz also is working on developing biomarkers. He said he’s collaborating with colleagues to follow directives by the National Institute on Mental Health to develop schizophrenia biomarkers in early-stage studies.
“It’s a way to assess how well a specific drug is hitting its target,” he said. “Once you’ve developed a good understanding of how well they’re hitting their target, then you can more finely tune doses before you put the drugs in thousands of people.”
The research may shed light on a question that continues to divide schizophrenia researchers: Is the disease one single condition or multiple disorders masquerading as one?
“I think that eventually, once we get better at genetics and looking at schizophrenia, we’ll realize it’s not one thing, that it’s many different things,” Dr. Kantrowitz said. “The brain is a very complex organ, and there are lots of different areas of things that can go wrong to produce similar symptoms that are grouped together as schizophrenia.”
It’s possible, he believes, that one group of schizophrenia patients, maybe 10 percent, will respond to a glutamate-based treatment. An additional 10 percent, perhaps will require something else.
“We’re not good enough to discern these things yet,” he said. “But eventually we’ll get to more targeted treatment.”
And then we’ll finally get to reset the clock marking the time – now almost 70 years – since the last major milestone in schizophrenia treatment.
Dr. Reynolds reported that over the past 3 years, he has received honoraria for lectures, advisory board membership, travel support and/or research support from Janssen, Lundbeck, Otsuka, Sunovion, and Sumitomo. Dr. Kantrowitz reported having received consulting payments within the last 24 months from Vindico Medical Education, Annenberg Center for Health Sciences at Eisenhower, Health Advances, SlingShot, Strategic Edge Communications, Havas Life, and Cowen and Company. He has conducted clinical research supported by the NIMH, the Stanley Foundation, Merck, Roche-Genentech, Forum, Sunovion, Novartis, Lundbeck, Alkermes, NeuroRx, Pfizer, and Lilly. He owns a small number of shares of common stock in GlaxoSmithKline.
Dr. Tamminga reported no relevant disclosures.
Lessons from the latest research
One major thrust of research is to understand exactly how the glutamate system works and what goes wrong in schizophrenia patients. Here are some questions and answers from recent research:
Question: If NMDA receptors are crucial to glutamate, would an NMDA receptor channel blocker help improve symptoms in schizophrenia patients? What about memantine (Namenda), an Alzheimer’s disease medication that treats the cognitive problems caused by dementia?
Answer: A 2017 systematic review of 10 studies (including two case reports) found “unclear results”: “memantine therapy in schizophrenic patients seems to improve mainly negative symptoms while positive symptoms and cognitive symptoms did not improve significantly.”
The researchers add that the “use of memantine should be considered in patients with prevalent negative symptoms and cognitive impairment, even if further trials are required. Memantine could be a new opportunity to treat young patients in order to prevent further cognitive decline that will lead to global impairment” (J Amino Acids. 2017;2017:7021071).
The review did not include a 2017 double-blind, placebo-controlled study of 46 adult male patients (average age around 45 years) comparing the antipsychotic risperidone with risperidone plus memantine. The researchers found no difference in positive symptom improvement between the two groups, but the combination treatment group showed significant improvement in cognitive function (at 6 weeks and study completion at 12 weeks) and negative symptoms (at 12 weeks) (Psychiatry Res. 2017 Jan;247:291-5).
Q: We know that the cognitive symptoms of schizophrenia are difficult to treat. What hope does the glutamate system hold in this area?
A: A 2017 systematic review of 44 studies says “memory, working memory and executive functions appear to be most influenced by the glutamatergic pathway.” But there’s some added complexity: “evidence from the literature suggests that presynaptic components synthesis and uptake of glutamate is involved in memory, while postsynaptic signaling appears to be involved in working memory.”
In the big picture, the review says, “the glutamatergic system appears to contribute to the cognitive deficits in schizophrenia, whereby different parts of the pathway are associated with different cognitive domains” (Neurosci Biobehav Rev. 2017 Apr 13;77:369-87).
Q: Could imaging via proton magnetic resonance spectroscopy prove a link between schizophrenia and certain kinds of glutamate activity in the brain, giving mental health professionals a tool to identify at-risk people who are most likely to develop psychosis?
A: A 2016 meta-analysis examined 59 studies and reported that “schizophrenia is associated with elevations in glutamatergic metabolites across several brain regions. This finding supports the hypothesis that schizophrenia is associated with excess glutamatergic neurotransmission in several limbic areas and further indicates that compounds that reduce glutamatergic transmission may have therapeutic potential” (JAMA Psychiatry. 2016 Jul 1;73[7]:665-74).
A review published in 2016 examined 11 proton magnetic resonance spectroscopy studies in search of signs that the state of the glutamate system could predict future psychosis.
Researchers found that six studies reported “significant alterations in glutamate metabolites across different cerebral areas (frontal lobe, thalamus, and the associative striatum)” in at-risk patients. “A longitudinal analysis in two of these trials confirmed an association between these abnormalities and worsening of symptoms and final transition to psychosis.”
But the other five studies analyzed in the review “found no significant differences across these same areas” (Schizophr Res. 2016 Mar;171[1-3]:166-75).
Mental health researchers like to say a half-century has passed since the last major advance in schizophrenia treatment came along. Gavin P. Reynolds, PhD, a schizophrenia researcher, says that’s wrong. In fact, he says, “it’s more like 60-plus!”
Yes, there have been advances in drug treatment since the early 1950s, when chlorpromazine (Thorazine) was introduced. Available are new types of antipsychotics, a whole bunch in fact, and they’ve helped many patients. “But these have been incremental, rather than the much-needed quantum leap,” said Dr. Reynolds in an interview. “We still need improved treatments. About one-third of patients do not respond to standard drug treatment. And of those who do respond, the negative symptoms and cognitive problems caused by schizophrenia may still be very limiting.”
That’s quite optimistic. The “revolution” hasn’t yet jumped from medical journals and clinical trials to prescription pads and drugstore shelves, and it’s not likely to do so any time soon. “This isn’t around the corner,” said Joshua T. Kantrowitz, MD, also a schizophrenia researcher. “But I can imagine a day where someone with schizophrenia will undergo a full genetic scan or a specific type of MRI or EEG, and we’ll then be able to recommend the drug they’d be able to use.” And he believes that a glutamate-based medication will be among the available options.
Thorazine: A pioneer with major limits
Like every person, each illness has a history. But schizophrenia’s past is fuzzier than that of many diseases, and this lack of clarity continues into the present as researchers try to understand exactly what it is – a single disorder? a collection of conditions? – and what it isn’t.
“Schizophrenia is a serious mental illness with a remarkably short recorded history. Unlike depression and mania, which are recognizable in ancient texts, schizophrenia-like disorder appeared rather suddenly in the early 19th century,” wrote R. Walter Heinrichs, PhD, a psychologist at Toronto’s York University (J Hist Behav Sci. 2003 Fall;39[4]:349-63). “This could mean that the illness is a recent disease that was largely unknown in earlier times. But perhaps schizophrenia existed, embedded and disguised within more general concepts of madness, and within the arcane languages and cultures of remote times,” he wrote.
As Dr. Heinrichs noted, schizophrenia’s history has spawned at least three theories: It’s a fairly new disease that just popped up in recent centuries. It has been around a long time but just didn’t get identified. It’s not a real disease but a product of modern thought.
Psychiatrists and researchers have rejected the latter possibility and its implications that psychotherapy could be the best treatment. Instead, they have tried to adjust the workings of the schizophrenic brain via medication.
The main breakthrough came in the 1950s through the development of the antipsychotic chlorpromazine. Its success “was instrumental in the reintegration of psychiatry with the other medical disciplines,” wrote Thomas A. Ban, MD, an emeritus professor at Vanderbilt University, Nashville, Tenn. “It turned psychiatrists from caregivers to full-fledged physicians who can help their patients and not only listen to their problems” (Neuropsychiatr Dis Treat. 2007 Aug; 3[4]:495-500).
Since chlorpromazine, however, medical treatment for schizophrenia has barely evolved, said Dr. Reynolds, professor emeritus at Queen’s University Belfast (Northern Ireland) and honorary professor at Sheffield (England) Hallam University. “The two most important developments have been the introduction of clozapine (Clozaril) for patients who don’t respond to other treatments and aripiprazole (Abilify), which has a somewhat different pharmacological action from other antipsychotics and thereby avoids some of the side effects.”
Negative symptoms fail to crumble
But, he said, side effects such as severe weight gain still can hamper the use of antipsychotics. And antipsychotics don’t fare well at treating the negative symptoms of schizophrenia.
As one overview puts it, “negative symptoms, e.g., social withdrawal, reduced initiative, anhedonia, and affective flattening, are notoriously difficult to treat.” Nonmedical treatments have shown some promise, the overview authors write: “Some positive findings have been reported, with the most robust improvements observed for social skills training. Although cognitive-behavior therapy shows significant effects for negative symptoms as a secondary outcome measure, there is a lack of data to allow for definite conclusions of its effectiveness for patients with predominant negative symptoms.”
As for medications, “antipsychotics have been shown to improve negative symptoms, but this seems to be limited to secondary negative symptoms in acute patients. It has also been suggested that antipsychotics may aggravate negative symptoms” (Schizophr Res. 2016 Jun 9. doi: 10.1016/j.schres.2016.05.015).
Looking to dopamine... and beyond
One solution to the vexing schizophrenia treatment gap is to develop a new medication that does a better job of adjusting the brain’s processing of dopamine.
Some researchers have found evidence that dopamine levels rise in the brains of people with schizophrenia, potentially explaining why chlorpromazine – which blocks receptors that process dopamine – is so effective. There also are signs that genes linked to dopamine play a role in schizophrenia.
But chlorpromazine is not effective enough to help many patients. Some gain no benefit, while others only see improvement in positive schizophrenia symptoms, those that are considered to be brought on – added – such as hallucinations and delusions.
And chlorpromazine isn’t believed to improve cognitive symptoms of schizophrenia like disorganized thoughts and concentration difficulties.
Atypical antipsychotics like clozapine (Clozaril) and risperidone (Risperdal), add the blocking of serotonin receptors to their dopamine focus. But they have serious limitations just like chlorpromazine.
The conclusion to take from the failure of dopamine-based drugs to fully combat schizophrenia, Dr. Kantrowitz and a colleague wrote, is that “dopaminergic dysfunction appears to account for only a part of schizophrenia’s symptomatic and neurocognitive profile.” Something else must be going on.
Enter the neurotransmitter known as glutamate.
The angel dust connection
Children who grew up in the 1970s and 1980s were flooded plenty of media messages about the dangers of illegal drugs. But one drug, “angel dust,” also known as phencyclidine or PCP, stood apart from the rest.
Using PCP, television shows and films suggested, led to consequences that could be deadly in ways that were entirely unlike the usual causes of death by drug use – a car accident, say, or a heroin overdose. Taking PCP, it seemed, was tied to crazed misadventure.
Consider a 1982 TV movie, “Desperate Lives,” starring a young actress named Helen Hunt. In a startling scene, she takes the drug and promptly jumps through a second-story window before screeching and cutting herself with jagged glass. The portrayal of PCP users as vividly unhinged – detached from reality and willing to hurt themselves or others – reflected a real phenomenon. As scientists had discovered, angel dust could made people act as if they were psychotic.
In the 1960s, “people started observing that drugs like PCP and ketamine produced schizophrenia-like symptoms,” Dr. Kantrowitz said. “But these observations were mostly ignored until the 1980s and 1990s.”
That’s when scientists started looking into the connections between these drugs and the psychotic states that they could induce. “These drugs can mimic, in animals and humans, the negative and cognitive symptoms as well as the positive symptoms of schizophrenia,” Dr. Reynolds said. “Combined with postmortem evidence of changes in various indicators of glutamate neurotransmission in the brain, these findings provide the basis for the glutamate hypothesis.”
The hypothesis theorizes that disruptions in the brain’s processing of glutamate are crucial to schizophrenia. Specifically, scientists believe the key lies in the N-methyl-D-aspartate (NMDA) receptor in nerve cells.
“Glutamatergic models are based upon the observation that the psychotomimetic agents such as phencyclidine and ketamine induce psychotic symptoms and neurocognitive disturbances similar to those of schizophrenia by blocking neurotransmission at N-methyl-D-aspartate (NMDA)-type glutamate receptors,” writes Daniel C. Javitt, MD, professor of psychiatry and director of schizophrenia research at the Nathan Kline Institute for Psychiatric Research at Columbia University. “Because glutamate/NMDA receptors are located throughout the brain, glutamatergic models predict widespread cortical dysfunction with particular involvement of NMDA receptors throughout the brain.
“Further, NMDA receptors are located on brain circuits that regulate dopamine release, suggesting that dopaminergic deficits in schizophrenia may also be secondary to underlying glutamatergic dysfunction.” (Isr J Psychiatry Relat Sci. 2010;47[1]:4-16).
There’s even more to the story, said Dr. Reynolds, who cautions that the term “glutamate hypothesis” is misleading, because it misses the whole picture.
“The original research identifying the importance of the NMDA receptor highlighted how these receptors were actually on GABA neurons, reflecting the close interaction and interdependence of the two transmitter systems,” he said. (GABA receptors respond to the neurotransmitter gamma-aminobutyric acid, or GABA.)
“Subsequently, it has been established that there is a dysfunction, if not a deficit, of a subgroup of GABA neurons in schizophrenia,” Dr. Reynolds said, “and these neurons, which are important in the control of glutamate neuronal activity, are also a focus of current research.”
Challenges on the drug development front
Earlier findings linking glutamate to schizophrenia “have been strengthened by recent large genetic studies identifying that variability in some genes involved in glutamate’s action is a risk factor for schizophrenia,” Dr. Reynolds said, “while in animal models, some drugs influencing glutamate transmission have shown promise as potential treatments.”
But glutamate-based drugs for schizophrenia remain elusive. “It is disappointing that the new antipsychotic drugs becoming available now are still all variations on the dopamine antagonist or partial agonist theme, even if they have some advantages in terms of side effects and evidence that they may have some limited effects on negative or cognitive symptoms,” Dr. Reynolds said. “Several drugs addressing glutamate systems – indirectly modulating the NMDA receptor or synaptic glutamate – have been developed that had good potential but have failed in clinical trials. Nevertheless, this strategy is still being pursued by the pharma industry.”
Dr. Reynolds points to drug development challenges on several fronts. For one, research subjects don’t necessarily represent patients who’d benefit the most from treatment.
“Drug treatments are likely to be most effective, and most important, in the early stages of the disease,” he said. “However, drug trials are almost inevitably undertaken on subjects who may have had many years of treatment prior to the trial without, perhaps, always responding very well.”
Another challenge: The tight interconnectedness of neural systems linked to schizophrenia. Tinkering with something in one part of the brain may lead to something going wrong somewhere else. “Glutamate and GABA neurons are ubiquitous throughout the brain,” Dr. Reynolds said, “and targeting systems particularly affected in schizophrenia is difficult without influencing other neuronal systems, potentially leading to further side effects.”
Targets offer hope for new medications
The challenges are significant, but the latest schizophrenia research also is opening up opportunities for new medications.
The GABA connection, for example, is a fertile field for researchers. “One interesting and novel approach that seems promising is the development of a drug that modulates ion channels in GABA/parvalbumin neurons, thereby potentially restoring their function,” Dr. Reynolds said. “We are still years away from availability of these new treatments, however.”
But what if drugs targeted specific types of schizophrenia just like antibiotics aim to treat different types of bacterial infections?
Dr. Tamminga is pushing researchers to focus on how disruptions in glutamate processing affect specific parts of the brain and specific brain functions. “Some people want to have a single glutamate defect throughout the whole brain,” she said. But she notes that “the glutamate system is prominent in every brain region,” suggesting that a global problem would entirely incapacitate schizophrenia patients.
In fact, “while people with schizophrenia do have glutamate-mediated troubles with their condition, with their memory, they don’t have glutamate problems with their motor system or sensory system,” she said. “There’s very little evidence for a generalized brain defect. We ought to be taking another tack: We need to figure out pathophysiology by region and brain function.”
Dr. Kantrowitz also is working on developing biomarkers. He said he’s collaborating with colleagues to follow directives by the National Institute on Mental Health to develop schizophrenia biomarkers in early-stage studies.
“It’s a way to assess how well a specific drug is hitting its target,” he said. “Once you’ve developed a good understanding of how well they’re hitting their target, then you can more finely tune doses before you put the drugs in thousands of people.”
The research may shed light on a question that continues to divide schizophrenia researchers: Is the disease one single condition or multiple disorders masquerading as one?
“I think that eventually, once we get better at genetics and looking at schizophrenia, we’ll realize it’s not one thing, that it’s many different things,” Dr. Kantrowitz said. “The brain is a very complex organ, and there are lots of different areas of things that can go wrong to produce similar symptoms that are grouped together as schizophrenia.”
It’s possible, he believes, that one group of schizophrenia patients, maybe 10 percent, will respond to a glutamate-based treatment. An additional 10 percent, perhaps will require something else.
“We’re not good enough to discern these things yet,” he said. “But eventually we’ll get to more targeted treatment.”
And then we’ll finally get to reset the clock marking the time – now almost 70 years – since the last major milestone in schizophrenia treatment.
Dr. Reynolds reported that over the past 3 years, he has received honoraria for lectures, advisory board membership, travel support and/or research support from Janssen, Lundbeck, Otsuka, Sunovion, and Sumitomo. Dr. Kantrowitz reported having received consulting payments within the last 24 months from Vindico Medical Education, Annenberg Center for Health Sciences at Eisenhower, Health Advances, SlingShot, Strategic Edge Communications, Havas Life, and Cowen and Company. He has conducted clinical research supported by the NIMH, the Stanley Foundation, Merck, Roche-Genentech, Forum, Sunovion, Novartis, Lundbeck, Alkermes, NeuroRx, Pfizer, and Lilly. He owns a small number of shares of common stock in GlaxoSmithKline.
Dr. Tamminga reported no relevant disclosures.
Lessons from the latest research
One major thrust of research is to understand exactly how the glutamate system works and what goes wrong in schizophrenia patients. Here are some questions and answers from recent research:
Question: If NMDA receptors are crucial to glutamate, would an NMDA receptor channel blocker help improve symptoms in schizophrenia patients? What about memantine (Namenda), an Alzheimer’s disease medication that treats the cognitive problems caused by dementia?
Answer: A 2017 systematic review of 10 studies (including two case reports) found “unclear results”: “memantine therapy in schizophrenic patients seems to improve mainly negative symptoms while positive symptoms and cognitive symptoms did not improve significantly.”
The researchers add that the “use of memantine should be considered in patients with prevalent negative symptoms and cognitive impairment, even if further trials are required. Memantine could be a new opportunity to treat young patients in order to prevent further cognitive decline that will lead to global impairment” (J Amino Acids. 2017;2017:7021071).
The review did not include a 2017 double-blind, placebo-controlled study of 46 adult male patients (average age around 45 years) comparing the antipsychotic risperidone with risperidone plus memantine. The researchers found no difference in positive symptom improvement between the two groups, but the combination treatment group showed significant improvement in cognitive function (at 6 weeks and study completion at 12 weeks) and negative symptoms (at 12 weeks) (Psychiatry Res. 2017 Jan;247:291-5).
Q: We know that the cognitive symptoms of schizophrenia are difficult to treat. What hope does the glutamate system hold in this area?
A: A 2017 systematic review of 44 studies says “memory, working memory and executive functions appear to be most influenced by the glutamatergic pathway.” But there’s some added complexity: “evidence from the literature suggests that presynaptic components synthesis and uptake of glutamate is involved in memory, while postsynaptic signaling appears to be involved in working memory.”
In the big picture, the review says, “the glutamatergic system appears to contribute to the cognitive deficits in schizophrenia, whereby different parts of the pathway are associated with different cognitive domains” (Neurosci Biobehav Rev. 2017 Apr 13;77:369-87).
Q: Could imaging via proton magnetic resonance spectroscopy prove a link between schizophrenia and certain kinds of glutamate activity in the brain, giving mental health professionals a tool to identify at-risk people who are most likely to develop psychosis?
A: A 2016 meta-analysis examined 59 studies and reported that “schizophrenia is associated with elevations in glutamatergic metabolites across several brain regions. This finding supports the hypothesis that schizophrenia is associated with excess glutamatergic neurotransmission in several limbic areas and further indicates that compounds that reduce glutamatergic transmission may have therapeutic potential” (JAMA Psychiatry. 2016 Jul 1;73[7]:665-74).
A review published in 2016 examined 11 proton magnetic resonance spectroscopy studies in search of signs that the state of the glutamate system could predict future psychosis.
Researchers found that six studies reported “significant alterations in glutamate metabolites across different cerebral areas (frontal lobe, thalamus, and the associative striatum)” in at-risk patients. “A longitudinal analysis in two of these trials confirmed an association between these abnormalities and worsening of symptoms and final transition to psychosis.”
But the other five studies analyzed in the review “found no significant differences across these same areas” (Schizophr Res. 2016 Mar;171[1-3]:166-75).
RF, IPL score highest for facial rejuvenation in review
SAN DIEGO – A review of existing research found that newer technologies are safer and more effective at skin rejuvenation than older ones, with two types – intense pulsed laser (IPL) and radiofrequency (RF) – at or near the head of the pack on both fronts.
In addition, 10 types of treatments, including multiple laser technologies, scored higher than facial peels on safety, although the peels beat almost all comers in terms of efficacy, the study’s lead author, Caerwyn Ash, PhD, said in an interview after presenting the results at the annual meeting of the American Society for Laser Medicine and Surgery.
The review is unique because it maps the various treatments on two axes – safety and efficacy – in a chart. Viewers gain an instant perspective on how the individual treatments fare when stacked up against each other. “The study gives credence to the new technologies,” said Dr. Ash, associate professor of medical devices at the University of Wales Trinity Saint David, Swansea. “We’re moving toward safer and more efficacious results.”
He and his colleagues analyzed more than 500 studies published since 1985, which evaluated 15 different types of technology used for facial rejuvenation. They reviewed at least 35 clinical studies per platform. They also reported on the safety and efficacy of facial peels.
The technologies examined included RF, light emitting diode (LED), pulsed dyed laser (PDL), frequency doubled 532-nm potassium-titanyl-phosphate neodymium:YAG laser (which had a low number of clinical studies), 980-nm diode laser, Q-switched Nd:YAG laser, 1320-nm Nd:YAG laser, 1540-nm Erbium:glass laser, 1450-nm diode laser, IPL, long pulsed Nd:YAG laser, plasma, and copper bromide laser.
Another technology reviewed was fractional selective photothermolysis, but studies of this modality were difficult to compare, according to the authors. Also included was the carbon dioxide laser, which the authors described as being hampered by long healing times and discomfort that requires anesthesia.
The review notes that comparisons between studies of the different treatments in the review were limited by such factors as varied beam and tip sizes (even within individual studies), various pulse durations, and different treatment times and intervals.
The researchers mapped the treatments in a single chart on two axes, safety and efficacy.
Three treatments scored the best in terms of efficacy: RF, IPL and chemical peels. However, while RF and IPL scored near the top of all treatments on the safety scale, chemical peels pulled up the rear.
The treatment strategies on the safety scale, ranked from most safe to least safe, were LED, IPL, diode laser, RF, argon laser, PDL and carbon dioxide laser, Nd:YAG, ablative Erbium, plasma, and chemical peel.
Radiofrequency topped the efficacy scale, followed closely by chemical peel and IPL. Following them, from most efficacious to least efficacious, were carbon dioxide laser, ablative Erbium, plasma treatments, and PDL, Nd:YAG and argon laser, diode laser, and LED.
The study was funded by the University of Wales Trinity Saint David. The authors had no disclosures.
SAN DIEGO – A review of existing research found that newer technologies are safer and more effective at skin rejuvenation than older ones, with two types – intense pulsed laser (IPL) and radiofrequency (RF) – at or near the head of the pack on both fronts.
In addition, 10 types of treatments, including multiple laser technologies, scored higher than facial peels on safety, although the peels beat almost all comers in terms of efficacy, the study’s lead author, Caerwyn Ash, PhD, said in an interview after presenting the results at the annual meeting of the American Society for Laser Medicine and Surgery.
The review is unique because it maps the various treatments on two axes – safety and efficacy – in a chart. Viewers gain an instant perspective on how the individual treatments fare when stacked up against each other. “The study gives credence to the new technologies,” said Dr. Ash, associate professor of medical devices at the University of Wales Trinity Saint David, Swansea. “We’re moving toward safer and more efficacious results.”
He and his colleagues analyzed more than 500 studies published since 1985, which evaluated 15 different types of technology used for facial rejuvenation. They reviewed at least 35 clinical studies per platform. They also reported on the safety and efficacy of facial peels.
The technologies examined included RF, light emitting diode (LED), pulsed dyed laser (PDL), frequency doubled 532-nm potassium-titanyl-phosphate neodymium:YAG laser (which had a low number of clinical studies), 980-nm diode laser, Q-switched Nd:YAG laser, 1320-nm Nd:YAG laser, 1540-nm Erbium:glass laser, 1450-nm diode laser, IPL, long pulsed Nd:YAG laser, plasma, and copper bromide laser.
Another technology reviewed was fractional selective photothermolysis, but studies of this modality were difficult to compare, according to the authors. Also included was the carbon dioxide laser, which the authors described as being hampered by long healing times and discomfort that requires anesthesia.
The review notes that comparisons between studies of the different treatments in the review were limited by such factors as varied beam and tip sizes (even within individual studies), various pulse durations, and different treatment times and intervals.
The researchers mapped the treatments in a single chart on two axes, safety and efficacy.
Three treatments scored the best in terms of efficacy: RF, IPL and chemical peels. However, while RF and IPL scored near the top of all treatments on the safety scale, chemical peels pulled up the rear.
The treatment strategies on the safety scale, ranked from most safe to least safe, were LED, IPL, diode laser, RF, argon laser, PDL and carbon dioxide laser, Nd:YAG, ablative Erbium, plasma, and chemical peel.
Radiofrequency topped the efficacy scale, followed closely by chemical peel and IPL. Following them, from most efficacious to least efficacious, were carbon dioxide laser, ablative Erbium, plasma treatments, and PDL, Nd:YAG and argon laser, diode laser, and LED.
The study was funded by the University of Wales Trinity Saint David. The authors had no disclosures.
SAN DIEGO – A review of existing research found that newer technologies are safer and more effective at skin rejuvenation than older ones, with two types – intense pulsed laser (IPL) and radiofrequency (RF) – at or near the head of the pack on both fronts.
In addition, 10 types of treatments, including multiple laser technologies, scored higher than facial peels on safety, although the peels beat almost all comers in terms of efficacy, the study’s lead author, Caerwyn Ash, PhD, said in an interview after presenting the results at the annual meeting of the American Society for Laser Medicine and Surgery.
The review is unique because it maps the various treatments on two axes – safety and efficacy – in a chart. Viewers gain an instant perspective on how the individual treatments fare when stacked up against each other. “The study gives credence to the new technologies,” said Dr. Ash, associate professor of medical devices at the University of Wales Trinity Saint David, Swansea. “We’re moving toward safer and more efficacious results.”
He and his colleagues analyzed more than 500 studies published since 1985, which evaluated 15 different types of technology used for facial rejuvenation. They reviewed at least 35 clinical studies per platform. They also reported on the safety and efficacy of facial peels.
The technologies examined included RF, light emitting diode (LED), pulsed dyed laser (PDL), frequency doubled 532-nm potassium-titanyl-phosphate neodymium:YAG laser (which had a low number of clinical studies), 980-nm diode laser, Q-switched Nd:YAG laser, 1320-nm Nd:YAG laser, 1540-nm Erbium:glass laser, 1450-nm diode laser, IPL, long pulsed Nd:YAG laser, plasma, and copper bromide laser.
Another technology reviewed was fractional selective photothermolysis, but studies of this modality were difficult to compare, according to the authors. Also included was the carbon dioxide laser, which the authors described as being hampered by long healing times and discomfort that requires anesthesia.
The review notes that comparisons between studies of the different treatments in the review were limited by such factors as varied beam and tip sizes (even within individual studies), various pulse durations, and different treatment times and intervals.
The researchers mapped the treatments in a single chart on two axes, safety and efficacy.
Three treatments scored the best in terms of efficacy: RF, IPL and chemical peels. However, while RF and IPL scored near the top of all treatments on the safety scale, chemical peels pulled up the rear.
The treatment strategies on the safety scale, ranked from most safe to least safe, were LED, IPL, diode laser, RF, argon laser, PDL and carbon dioxide laser, Nd:YAG, ablative Erbium, plasma, and chemical peel.
Radiofrequency topped the efficacy scale, followed closely by chemical peel and IPL. Following them, from most efficacious to least efficacious, were carbon dioxide laser, ablative Erbium, plasma treatments, and PDL, Nd:YAG and argon laser, diode laser, and LED.
The study was funded by the University of Wales Trinity Saint David. The authors had no disclosures.
AT LASER 2017
Key clinical point:
Major finding: IPL and RF were among the modalities that were safer and more effective at skin rejuvenation than were older ones.
Data source: An analysis of more than 500 studies published since 1985 evaluating of 15 different types of technology used for facial rejuvenation, reviewing at least 35 clinical studies per platform.
Disclosures: The study was funded by the University of Wales Trinity Saint David. The authors had no disclosures.
Top Acthar prescribers reap hefty payments from drug maker
BOSTON – A new analysis finds that some of the biggest neurologist prescribers of repository corticotropin gel (Acthar) – the extraordinarily expensive multiple sclerosis (MS) relapse drug – reaped extensive payments from its manufacturer, with one taking in $130,307 in a single year.
Together, 51 neurologists accounted for 980 Medicare claims worth more than $39 million in Acthar spending in 2014, almost half of the entire estimated $83 million in Medicare spending on neurologist-prescribed Acthar that year.
“There is a small group of neurologists – less than 1% – who are prescribing Acthar at considerable cost to Medicare and may do this in part because of financial relationships with the company that sells Acthar,” said study lead author Dennis Bourdette, MD, chair and research professor of neurology at Oregon Health and Science University, Portland.
Dr. Bourdette acknowledges that the research doesn’t prove a causal relationship between payments and prescriptions. In response to questions, Acthar manufacturer Mallinckrodt Pharmaceuticals, already under fire for the $34,000-per-vial cost of the drug, questioned the study design and denied wrongdoing in a statement: “Mallinckrodt is committed to following the highest standards for integrity and compliance in all of our business practices, including our collaboration with physicians.”
Acthar, also known as H.P. Acthar, is the poster child for stunningly expensive medication. According to the Federal Trade Commission, the cost of the drug rose from $40 per vial in 2001 to more than $34,000 this year. The New York Times reports that Medicare spending on Acthar topped half a billion in 2015.
Earlier this year, Mallinckrodt ARD (formerly Questcor) and its parent company agreed to pay a $100 million fine to settle charges that it created an illegal monopoly over the drug.
Dr. Bourdette and colleagues released their findings at the annual meeting of the American Academy of Neurology.
In an interview, Dr. Bourdette said the study’s roots lie in his concern about the medication, whose transformation from an inexpensive 1950s-era medication for pulmonary sarcoidosis to high-priced MS treatment has drawn national media attention.
“I believe that Acthar is tremendously overpriced and a waste of health care money,” Dr. Bourdette said. “I wanted to find out how extensive an economic problem it was and how much money Medicare was spending on it since this data was easily accessible.”
According to the study, Medicare spent $1.3 billion from 2011 to 2015 on Acthar, with about 25% of the cost due to prescriptions from neurologists. “When I discovered that a relatively small number of neurologists was prescribing it commonly at a cost of $40-$50 million a year to Medicare, I was interested in determining why they were prescribing this expensive therapy and postulated that it might be related to financial support they were receiving from the manufacturer,” Dr. Bourdette said.
The researchers examined the Medicare Part D Public Use File to determine which neurologists prescribed Acthar frequently in 2014. They identified 51 who prescribed Acthar 10 or more times that year and were frequent prescribers of MS disease-modifying therapy, indicating they treated many people with MS.
The 51 neurologists accounted for a mean of 19 Acthar claims each in 2014 (range, 11-50) totaling a mean annual cost of $770,145 (range, $354,479-$3,623,509). Together, the neurologists accounted for total Medicare spending on Acthar of $39,277,380.
The researchers also chose a control group – 51 neurologists who prescribed glatiramer acetate (Copaxone) more than 10 times in 2014 and also prescribed a similar frequency of all MS disease-modifying therapies as the high-frequency Acthar group.
“Acthar is used episodically to treat MS relapses, and glatiramer acetate is taken chronically to prevent relapses and disability,” Dr. Bourdette said. He added that glatiramer acetate is now available in a generic, but it wasn’t in 2014.
The Acthar and Copaxone groups were nearly identical in terms of gender (about two-thirds men) and years since graduation (a mean of 26), but the Acthar prescribers were more likely to work in small practices (1-10 doctors), file more prescription claims, see more Medicare patients, and practice in the South or West. The demographic information came from CMS Physician Compare.
In terms of overall drug maker payments, neurologists in the Acthar group accepted much more (median, $54,270; range, $623-$369,847) than did the Copaxone group (median, $1,747, range, $0-$256,305, P less than .001). Payment information came from the federal Open Payments database.
As for payments directly from the manufacturers of the two drugs, the Acthar prescribers accepted a median of $5,344 (range, $0-$130,307) from its manufacturer, while the Copaxone prescribers accepted a median of $137 (range, $0-$168,373) from Teva (P = .003).
“The payments are primarily for giving lectures or serving on advisory boards,” Dr. Bourdette said. “These types of payments are commonly made by pharmaceutical companies to physicians who participate in these types of activities.”
In a statement, Mallinckrodt Pharmaceuticals contends that the study inappropriately compares Acthar, often a later-line therapy, to Copaxone, which it says is often a first-line therapy.
Dr. Bourdette responded that the researchers chose a comparison group of top prescribers of Copaxone “as a marker of neurologists who treated a significant number of patients with MS.”
Mallinckrodt also notes that “there may be unmeasured confounding factors in the matching process between the comparator physician groups. These could include differences in the patient characteristics managed by these physicians including disease severity affecting prescribing patterns.”
The researchers agree that there may be differences between the groups, Dr. Bourdette said. However, he added, “the fact remains that the two groups differed in the amount of money they received as open-source payments from pharmaceutical companies. We doubt that the severity of their case mixes should lead to one group receiving more pharmaceutical open-source payments than another.”
Finally, Mallinckrodt says there’s no proof of a causal connection between the payments and the prescriptions; the study authors agree. And, the company says, “this pattern of correlation would be expected in any scenario where a small number of prescribers are the experts in the use of a later-line drug to treat a limited subset of patients.”
Dr. Bourdette rejects this contention. Few of the high Acthar prescribers practice at academic centers, he said, and few are recognized for their MS expertise. “So to suggest that they are experts in the use of Acthar for the treatment of MS when they are not as a group recognized as being experts on the treatment of MS in general is incredible,” he said. “Why a small group of neurologists prescribe Acthar remains a mystery, but it is not because the majority are leaders in the field of MS therapeutics.”
In an interview, Eric G. Campbell, PhD, professor of medicine and director of research at the Mongan Institute Health Policy Center at Harvard Medical School, Boston, said the study findings fit in with previous research that has found that “the more money that people get, the more they use the drug.
“Any reasonable person looking at this data would assume or at least consider very strongly that there is a causal relationship here,” he said.
What should be done? “There are lots of ways to stop this,” Dr. Campbell said. “One could simply impose rules that forbid doctors who accept payments for marketing drugs from billing Medicare or private payers for the care they provide. Large provider organizations could pass rules that forbid this kind of behavior. Finally, the government could vigorously pursue stiff penalties against physicians who accept payments that are really nothing more than incentives to encourage or sustain prescribing practices.”
The study was supported by a National Multiple Sclerosis Society grant. Dr. Bourdette said that he provides consulting to Magellan Healthcare, a company that provides recommendations to insurance companies on the approval of high-cost therapies. Dr. Campbell disclosed that he serves as an expert witness on law cases related to conflicts of interest in medicine.
BOSTON – A new analysis finds that some of the biggest neurologist prescribers of repository corticotropin gel (Acthar) – the extraordinarily expensive multiple sclerosis (MS) relapse drug – reaped extensive payments from its manufacturer, with one taking in $130,307 in a single year.
Together, 51 neurologists accounted for 980 Medicare claims worth more than $39 million in Acthar spending in 2014, almost half of the entire estimated $83 million in Medicare spending on neurologist-prescribed Acthar that year.
“There is a small group of neurologists – less than 1% – who are prescribing Acthar at considerable cost to Medicare and may do this in part because of financial relationships with the company that sells Acthar,” said study lead author Dennis Bourdette, MD, chair and research professor of neurology at Oregon Health and Science University, Portland.
Dr. Bourdette acknowledges that the research doesn’t prove a causal relationship between payments and prescriptions. In response to questions, Acthar manufacturer Mallinckrodt Pharmaceuticals, already under fire for the $34,000-per-vial cost of the drug, questioned the study design and denied wrongdoing in a statement: “Mallinckrodt is committed to following the highest standards for integrity and compliance in all of our business practices, including our collaboration with physicians.”
Acthar, also known as H.P. Acthar, is the poster child for stunningly expensive medication. According to the Federal Trade Commission, the cost of the drug rose from $40 per vial in 2001 to more than $34,000 this year. The New York Times reports that Medicare spending on Acthar topped half a billion in 2015.
Earlier this year, Mallinckrodt ARD (formerly Questcor) and its parent company agreed to pay a $100 million fine to settle charges that it created an illegal monopoly over the drug.
Dr. Bourdette and colleagues released their findings at the annual meeting of the American Academy of Neurology.
In an interview, Dr. Bourdette said the study’s roots lie in his concern about the medication, whose transformation from an inexpensive 1950s-era medication for pulmonary sarcoidosis to high-priced MS treatment has drawn national media attention.
“I believe that Acthar is tremendously overpriced and a waste of health care money,” Dr. Bourdette said. “I wanted to find out how extensive an economic problem it was and how much money Medicare was spending on it since this data was easily accessible.”
According to the study, Medicare spent $1.3 billion from 2011 to 2015 on Acthar, with about 25% of the cost due to prescriptions from neurologists. “When I discovered that a relatively small number of neurologists was prescribing it commonly at a cost of $40-$50 million a year to Medicare, I was interested in determining why they were prescribing this expensive therapy and postulated that it might be related to financial support they were receiving from the manufacturer,” Dr. Bourdette said.
The researchers examined the Medicare Part D Public Use File to determine which neurologists prescribed Acthar frequently in 2014. They identified 51 who prescribed Acthar 10 or more times that year and were frequent prescribers of MS disease-modifying therapy, indicating they treated many people with MS.
The 51 neurologists accounted for a mean of 19 Acthar claims each in 2014 (range, 11-50) totaling a mean annual cost of $770,145 (range, $354,479-$3,623,509). Together, the neurologists accounted for total Medicare spending on Acthar of $39,277,380.
The researchers also chose a control group – 51 neurologists who prescribed glatiramer acetate (Copaxone) more than 10 times in 2014 and also prescribed a similar frequency of all MS disease-modifying therapies as the high-frequency Acthar group.
“Acthar is used episodically to treat MS relapses, and glatiramer acetate is taken chronically to prevent relapses and disability,” Dr. Bourdette said. He added that glatiramer acetate is now available in a generic, but it wasn’t in 2014.
The Acthar and Copaxone groups were nearly identical in terms of gender (about two-thirds men) and years since graduation (a mean of 26), but the Acthar prescribers were more likely to work in small practices (1-10 doctors), file more prescription claims, see more Medicare patients, and practice in the South or West. The demographic information came from CMS Physician Compare.
In terms of overall drug maker payments, neurologists in the Acthar group accepted much more (median, $54,270; range, $623-$369,847) than did the Copaxone group (median, $1,747, range, $0-$256,305, P less than .001). Payment information came from the federal Open Payments database.
As for payments directly from the manufacturers of the two drugs, the Acthar prescribers accepted a median of $5,344 (range, $0-$130,307) from its manufacturer, while the Copaxone prescribers accepted a median of $137 (range, $0-$168,373) from Teva (P = .003).
“The payments are primarily for giving lectures or serving on advisory boards,” Dr. Bourdette said. “These types of payments are commonly made by pharmaceutical companies to physicians who participate in these types of activities.”
In a statement, Mallinckrodt Pharmaceuticals contends that the study inappropriately compares Acthar, often a later-line therapy, to Copaxone, which it says is often a first-line therapy.
Dr. Bourdette responded that the researchers chose a comparison group of top prescribers of Copaxone “as a marker of neurologists who treated a significant number of patients with MS.”
Mallinckrodt also notes that “there may be unmeasured confounding factors in the matching process between the comparator physician groups. These could include differences in the patient characteristics managed by these physicians including disease severity affecting prescribing patterns.”
The researchers agree that there may be differences between the groups, Dr. Bourdette said. However, he added, “the fact remains that the two groups differed in the amount of money they received as open-source payments from pharmaceutical companies. We doubt that the severity of their case mixes should lead to one group receiving more pharmaceutical open-source payments than another.”
Finally, Mallinckrodt says there’s no proof of a causal connection between the payments and the prescriptions; the study authors agree. And, the company says, “this pattern of correlation would be expected in any scenario where a small number of prescribers are the experts in the use of a later-line drug to treat a limited subset of patients.”
Dr. Bourdette rejects this contention. Few of the high Acthar prescribers practice at academic centers, he said, and few are recognized for their MS expertise. “So to suggest that they are experts in the use of Acthar for the treatment of MS when they are not as a group recognized as being experts on the treatment of MS in general is incredible,” he said. “Why a small group of neurologists prescribe Acthar remains a mystery, but it is not because the majority are leaders in the field of MS therapeutics.”
In an interview, Eric G. Campbell, PhD, professor of medicine and director of research at the Mongan Institute Health Policy Center at Harvard Medical School, Boston, said the study findings fit in with previous research that has found that “the more money that people get, the more they use the drug.
“Any reasonable person looking at this data would assume or at least consider very strongly that there is a causal relationship here,” he said.
What should be done? “There are lots of ways to stop this,” Dr. Campbell said. “One could simply impose rules that forbid doctors who accept payments for marketing drugs from billing Medicare or private payers for the care they provide. Large provider organizations could pass rules that forbid this kind of behavior. Finally, the government could vigorously pursue stiff penalties against physicians who accept payments that are really nothing more than incentives to encourage or sustain prescribing practices.”
The study was supported by a National Multiple Sclerosis Society grant. Dr. Bourdette said that he provides consulting to Magellan Healthcare, a company that provides recommendations to insurance companies on the approval of high-cost therapies. Dr. Campbell disclosed that he serves as an expert witness on law cases related to conflicts of interest in medicine.
BOSTON – A new analysis finds that some of the biggest neurologist prescribers of repository corticotropin gel (Acthar) – the extraordinarily expensive multiple sclerosis (MS) relapse drug – reaped extensive payments from its manufacturer, with one taking in $130,307 in a single year.
Together, 51 neurologists accounted for 980 Medicare claims worth more than $39 million in Acthar spending in 2014, almost half of the entire estimated $83 million in Medicare spending on neurologist-prescribed Acthar that year.
“There is a small group of neurologists – less than 1% – who are prescribing Acthar at considerable cost to Medicare and may do this in part because of financial relationships with the company that sells Acthar,” said study lead author Dennis Bourdette, MD, chair and research professor of neurology at Oregon Health and Science University, Portland.
Dr. Bourdette acknowledges that the research doesn’t prove a causal relationship between payments and prescriptions. In response to questions, Acthar manufacturer Mallinckrodt Pharmaceuticals, already under fire for the $34,000-per-vial cost of the drug, questioned the study design and denied wrongdoing in a statement: “Mallinckrodt is committed to following the highest standards for integrity and compliance in all of our business practices, including our collaboration with physicians.”
Acthar, also known as H.P. Acthar, is the poster child for stunningly expensive medication. According to the Federal Trade Commission, the cost of the drug rose from $40 per vial in 2001 to more than $34,000 this year. The New York Times reports that Medicare spending on Acthar topped half a billion in 2015.
Earlier this year, Mallinckrodt ARD (formerly Questcor) and its parent company agreed to pay a $100 million fine to settle charges that it created an illegal monopoly over the drug.
Dr. Bourdette and colleagues released their findings at the annual meeting of the American Academy of Neurology.
In an interview, Dr. Bourdette said the study’s roots lie in his concern about the medication, whose transformation from an inexpensive 1950s-era medication for pulmonary sarcoidosis to high-priced MS treatment has drawn national media attention.
“I believe that Acthar is tremendously overpriced and a waste of health care money,” Dr. Bourdette said. “I wanted to find out how extensive an economic problem it was and how much money Medicare was spending on it since this data was easily accessible.”
According to the study, Medicare spent $1.3 billion from 2011 to 2015 on Acthar, with about 25% of the cost due to prescriptions from neurologists. “When I discovered that a relatively small number of neurologists was prescribing it commonly at a cost of $40-$50 million a year to Medicare, I was interested in determining why they were prescribing this expensive therapy and postulated that it might be related to financial support they were receiving from the manufacturer,” Dr. Bourdette said.
The researchers examined the Medicare Part D Public Use File to determine which neurologists prescribed Acthar frequently in 2014. They identified 51 who prescribed Acthar 10 or more times that year and were frequent prescribers of MS disease-modifying therapy, indicating they treated many people with MS.
The 51 neurologists accounted for a mean of 19 Acthar claims each in 2014 (range, 11-50) totaling a mean annual cost of $770,145 (range, $354,479-$3,623,509). Together, the neurologists accounted for total Medicare spending on Acthar of $39,277,380.
The researchers also chose a control group – 51 neurologists who prescribed glatiramer acetate (Copaxone) more than 10 times in 2014 and also prescribed a similar frequency of all MS disease-modifying therapies as the high-frequency Acthar group.
“Acthar is used episodically to treat MS relapses, and glatiramer acetate is taken chronically to prevent relapses and disability,” Dr. Bourdette said. He added that glatiramer acetate is now available in a generic, but it wasn’t in 2014.
The Acthar and Copaxone groups were nearly identical in terms of gender (about two-thirds men) and years since graduation (a mean of 26), but the Acthar prescribers were more likely to work in small practices (1-10 doctors), file more prescription claims, see more Medicare patients, and practice in the South or West. The demographic information came from CMS Physician Compare.
In terms of overall drug maker payments, neurologists in the Acthar group accepted much more (median, $54,270; range, $623-$369,847) than did the Copaxone group (median, $1,747, range, $0-$256,305, P less than .001). Payment information came from the federal Open Payments database.
As for payments directly from the manufacturers of the two drugs, the Acthar prescribers accepted a median of $5,344 (range, $0-$130,307) from its manufacturer, while the Copaxone prescribers accepted a median of $137 (range, $0-$168,373) from Teva (P = .003).
“The payments are primarily for giving lectures or serving on advisory boards,” Dr. Bourdette said. “These types of payments are commonly made by pharmaceutical companies to physicians who participate in these types of activities.”
In a statement, Mallinckrodt Pharmaceuticals contends that the study inappropriately compares Acthar, often a later-line therapy, to Copaxone, which it says is often a first-line therapy.
Dr. Bourdette responded that the researchers chose a comparison group of top prescribers of Copaxone “as a marker of neurologists who treated a significant number of patients with MS.”
Mallinckrodt also notes that “there may be unmeasured confounding factors in the matching process between the comparator physician groups. These could include differences in the patient characteristics managed by these physicians including disease severity affecting prescribing patterns.”
The researchers agree that there may be differences between the groups, Dr. Bourdette said. However, he added, “the fact remains that the two groups differed in the amount of money they received as open-source payments from pharmaceutical companies. We doubt that the severity of their case mixes should lead to one group receiving more pharmaceutical open-source payments than another.”
Finally, Mallinckrodt says there’s no proof of a causal connection between the payments and the prescriptions; the study authors agree. And, the company says, “this pattern of correlation would be expected in any scenario where a small number of prescribers are the experts in the use of a later-line drug to treat a limited subset of patients.”
Dr. Bourdette rejects this contention. Few of the high Acthar prescribers practice at academic centers, he said, and few are recognized for their MS expertise. “So to suggest that they are experts in the use of Acthar for the treatment of MS when they are not as a group recognized as being experts on the treatment of MS in general is incredible,” he said. “Why a small group of neurologists prescribe Acthar remains a mystery, but it is not because the majority are leaders in the field of MS therapeutics.”
In an interview, Eric G. Campbell, PhD, professor of medicine and director of research at the Mongan Institute Health Policy Center at Harvard Medical School, Boston, said the study findings fit in with previous research that has found that “the more money that people get, the more they use the drug.
“Any reasonable person looking at this data would assume or at least consider very strongly that there is a causal relationship here,” he said.
What should be done? “There are lots of ways to stop this,” Dr. Campbell said. “One could simply impose rules that forbid doctors who accept payments for marketing drugs from billing Medicare or private payers for the care they provide. Large provider organizations could pass rules that forbid this kind of behavior. Finally, the government could vigorously pursue stiff penalties against physicians who accept payments that are really nothing more than incentives to encourage or sustain prescribing practices.”
The study was supported by a National Multiple Sclerosis Society grant. Dr. Bourdette said that he provides consulting to Magellan Healthcare, a company that provides recommendations to insurance companies on the approval of high-cost therapies. Dr. Campbell disclosed that he serves as an expert witness on law cases related to conflicts of interest in medicine.
AT AAN 2017
Key clinical point:
Major finding: The top Acthar prescribers reaped much more in payments from drug makers overall in 2014 than did a control group of top prescribers of another MS drug (median of $54,270 vs. $1,747; P less than .001) and from the manufacturer of each drug (median of $5,344 vs. $137; P = .003).
Data source: Medicare Part D Public Use File, CMS Physician Compare, Open Payments database, 2014.
Disclosures: The work was supported by a National Multiple Sclerosis Society grant. Dr. Bourdette provides consulting to Magellan Healthcare, a company that provides recommendations to insurance companies on the approval of high-cost therapies.
Lasers may be effective for treating xanthelasma
SAN DIEGO – Laser treatments may be effective for xanthelasma palpebrarum lesions, based on a systematic review of existing studies, although the research is limited.
“The number of cases we looked at was relatively small, so you can’t come up with any definite conclusions,” said review coauthor Christopher J. Huerter, MD, head of the division of dermatology at Creighton University, Omaha. “But it’s promising since the lasers we examined all work with some efficacy, with the CO2 and Er:YAG [erbium:YAG] lasers probably having the best results.”
Xanthelasma lesions appear as small yellowish plaques on the eyelids. “About half the people who have it have some blood lipid abnormality,” Dr. Huerter said in an interview. “If a person has it, it’s worthwhile to do a cholesterol screen or a lipid profile.”
Treatment with trichloroacetic acid is one option, although it was more common before lasers began to be used. In addition, “surgical incision can be very effective,” he said, although the review notes that it can create undesirable scarring.
Researchers have studied laser treatment for xanthelasma for at least 30 years. Dr. Huerter and his colleagues examined 21 studies published since 1987, with the following lasers: CO2 laser (three studies), argon laser (one study), Er:YAG laser (four studies), ultrapulse CO2 laser (five studies), 1,450-nm diode laser (one study), pulsed dye laser (PDL, two studies), superpulsed or fractional CO2 laser (one study), and Q-switched neodymium:YAG laser (three studies). An additional study examined both argon and Er:YAG lasers.
The number of treated patients in the studies ranged from 1 to 50, and the number of treated lesions ranged from 1 to 76. (Patients often had more than one lesion.) “It would be nice to have bigger studies and bigger numbers,” Dr. Huerter said at the annual meeting of the American Society for Laser Medicine and Surgery.
Although the studies were limited by small cohorts, short follow-up, and lack of comparison groups, the findings did reveal signs of effectiveness: Clearance rates were 100% in CO2, argon, and PDL cases and about 85% with Er:YAG lasers. Clearance rates were lower with Nd:YAG (about 55%) and 1,450-nm diode (about 48%) lasers.
Edema was reported in all PDL cases and erythema in almost 20% of CO2 cases. Dyspigmentation was most common – at about 30% – in Er:YAG and 1,450-nm diode cases. Visible scars were reported in more than 5% of Er:YAG cases.
The review concluded that “sufficient evidence is available to suggest laser therapies to be a cosmetically excellent treatment option for xanthelasma , particularly applicable in patients who are not good candidates for surgical excision,” he said.
As for advice to dermatologists, Dr. Huerter pointed to the positive results for CO2 and Er:YAG lasers. He said PDL lasers could also be used. As for argon lasers, he noted that it’s not as likely for dermatologists to have them on hand, he said.
In regard to choosing which xanthelasma lesions to treat with laser, he said thicker ones may not be as amenable. “But if you do laser treatment and don’t get the results you want, you can always excise.”
No funding for the study was reported. Dr. Huerter reported no disclosures.
SAN DIEGO – Laser treatments may be effective for xanthelasma palpebrarum lesions, based on a systematic review of existing studies, although the research is limited.
“The number of cases we looked at was relatively small, so you can’t come up with any definite conclusions,” said review coauthor Christopher J. Huerter, MD, head of the division of dermatology at Creighton University, Omaha. “But it’s promising since the lasers we examined all work with some efficacy, with the CO2 and Er:YAG [erbium:YAG] lasers probably having the best results.”
Xanthelasma lesions appear as small yellowish plaques on the eyelids. “About half the people who have it have some blood lipid abnormality,” Dr. Huerter said in an interview. “If a person has it, it’s worthwhile to do a cholesterol screen or a lipid profile.”
Treatment with trichloroacetic acid is one option, although it was more common before lasers began to be used. In addition, “surgical incision can be very effective,” he said, although the review notes that it can create undesirable scarring.
Researchers have studied laser treatment for xanthelasma for at least 30 years. Dr. Huerter and his colleagues examined 21 studies published since 1987, with the following lasers: CO2 laser (three studies), argon laser (one study), Er:YAG laser (four studies), ultrapulse CO2 laser (five studies), 1,450-nm diode laser (one study), pulsed dye laser (PDL, two studies), superpulsed or fractional CO2 laser (one study), and Q-switched neodymium:YAG laser (three studies). An additional study examined both argon and Er:YAG lasers.
The number of treated patients in the studies ranged from 1 to 50, and the number of treated lesions ranged from 1 to 76. (Patients often had more than one lesion.) “It would be nice to have bigger studies and bigger numbers,” Dr. Huerter said at the annual meeting of the American Society for Laser Medicine and Surgery.
Although the studies were limited by small cohorts, short follow-up, and lack of comparison groups, the findings did reveal signs of effectiveness: Clearance rates were 100% in CO2, argon, and PDL cases and about 85% with Er:YAG lasers. Clearance rates were lower with Nd:YAG (about 55%) and 1,450-nm diode (about 48%) lasers.
Edema was reported in all PDL cases and erythema in almost 20% of CO2 cases. Dyspigmentation was most common – at about 30% – in Er:YAG and 1,450-nm diode cases. Visible scars were reported in more than 5% of Er:YAG cases.
The review concluded that “sufficient evidence is available to suggest laser therapies to be a cosmetically excellent treatment option for xanthelasma , particularly applicable in patients who are not good candidates for surgical excision,” he said.
As for advice to dermatologists, Dr. Huerter pointed to the positive results for CO2 and Er:YAG lasers. He said PDL lasers could also be used. As for argon lasers, he noted that it’s not as likely for dermatologists to have them on hand, he said.
In regard to choosing which xanthelasma lesions to treat with laser, he said thicker ones may not be as amenable. “But if you do laser treatment and don’t get the results you want, you can always excise.”
No funding for the study was reported. Dr. Huerter reported no disclosures.
SAN DIEGO – Laser treatments may be effective for xanthelasma palpebrarum lesions, based on a systematic review of existing studies, although the research is limited.
“The number of cases we looked at was relatively small, so you can’t come up with any definite conclusions,” said review coauthor Christopher J. Huerter, MD, head of the division of dermatology at Creighton University, Omaha. “But it’s promising since the lasers we examined all work with some efficacy, with the CO2 and Er:YAG [erbium:YAG] lasers probably having the best results.”
Xanthelasma lesions appear as small yellowish plaques on the eyelids. “About half the people who have it have some blood lipid abnormality,” Dr. Huerter said in an interview. “If a person has it, it’s worthwhile to do a cholesterol screen or a lipid profile.”
Treatment with trichloroacetic acid is one option, although it was more common before lasers began to be used. In addition, “surgical incision can be very effective,” he said, although the review notes that it can create undesirable scarring.
Researchers have studied laser treatment for xanthelasma for at least 30 years. Dr. Huerter and his colleagues examined 21 studies published since 1987, with the following lasers: CO2 laser (three studies), argon laser (one study), Er:YAG laser (four studies), ultrapulse CO2 laser (five studies), 1,450-nm diode laser (one study), pulsed dye laser (PDL, two studies), superpulsed or fractional CO2 laser (one study), and Q-switched neodymium:YAG laser (three studies). An additional study examined both argon and Er:YAG lasers.
The number of treated patients in the studies ranged from 1 to 50, and the number of treated lesions ranged from 1 to 76. (Patients often had more than one lesion.) “It would be nice to have bigger studies and bigger numbers,” Dr. Huerter said at the annual meeting of the American Society for Laser Medicine and Surgery.
Although the studies were limited by small cohorts, short follow-up, and lack of comparison groups, the findings did reveal signs of effectiveness: Clearance rates were 100% in CO2, argon, and PDL cases and about 85% with Er:YAG lasers. Clearance rates were lower with Nd:YAG (about 55%) and 1,450-nm diode (about 48%) lasers.
Edema was reported in all PDL cases and erythema in almost 20% of CO2 cases. Dyspigmentation was most common – at about 30% – in Er:YAG and 1,450-nm diode cases. Visible scars were reported in more than 5% of Er:YAG cases.
The review concluded that “sufficient evidence is available to suggest laser therapies to be a cosmetically excellent treatment option for xanthelasma , particularly applicable in patients who are not good candidates for surgical excision,” he said.
As for advice to dermatologists, Dr. Huerter pointed to the positive results for CO2 and Er:YAG lasers. He said PDL lasers could also be used. As for argon lasers, he noted that it’s not as likely for dermatologists to have them on hand, he said.
In regard to choosing which xanthelasma lesions to treat with laser, he said thicker ones may not be as amenable. “But if you do laser treatment and don’t get the results you want, you can always excise.”
No funding for the study was reported. Dr. Huerter reported no disclosures.
Key clinical point:
Major finding: Clearance rates of about 85%-100% were reported for xanthelasma treatment with CO2, argon, pulsed dye, and Er:YAG laser treatments.
Data source: A systematic review of 21 studies evaluating different laser treatments for xanthelasma.
Disclosures: No funding was reported. Dr. Huerter reported no disclosures.
VIDEO: About 1 in 20 ALS patients in Washington state chose assisted suicide
BOSTON – A new study estimates that 3.4%-6.7% of amyotrophic lateral sclerosis (ALS) patients in Washington state sought to commit physician-assisted suicide over a 5-year period.
The rate is many times higher than that among cancer patients in the state, researchers found. They also discovered that ALS patients were significantly more likely than were other terminally ill people to use the deadly medication after getting prescriptions for it.
The findings appear to reflect the unique hopelessness facing ALS patients. “They’re not afforded as much denial of decline and death as are patients with other terminal illnesses,” said Linda Ganzini, MD, MPH, a professor of psychiatry and medicine at Oregon Health & Science University, Portland, who has studied end of life in ALS patients.
“Many cancer patients, even in the final days of life, receive treatments that they hope will extend their lives,” she said in an interview after reviewing the study findings. “In contrast, treatments for ALS are minimally effective.”
Physician-assisted suicide is legal in California, Colorado, the District of Columbia, Montana, Oregon, Vermont, and Washington.
A team led by Leo H. Wang, MD, PhD, of the University of Washington, Seattle, examined the medical records of 39 ALS patients who sought medication to end their lives at three hospitals in Seattle from March 2009 to Dec. 31, 2014.
Washington’s Death with Dignity (DWD) law, which went into effect in 2009, allows physicians to prescribe lethal medication if the patient has a terminal illness and a prognosis of less than 6 months to live as judged by two physicians.
The researchers reported their findings, a follow-up to a previous study (Neurology. 2016 Nov 15;87[20]:2117-22), at the annual meeting of the American Academy of Neurology.
The median age of the ALS patients at symptom onset was 64 (range, 42-83), and a median of 712 days passed (range, 207-2,407) from the date of diagnosis to date of prescription for lethal medication.
The median time from prescription to death was 22 days, with at least one patient dying immediately (range, 0-386 days). All 39 patients had limb involvement, and 82%-92% had bulbar involvement, dysarthria, dysphagia, and/or dyspnea.
The researchers estimate that 3.4%-6.7% of 1,146 ALS patients in Washington who died over the time period of the study sought a physician-assisted death. The 3.4% figure assumes that the 39 patients at the three hospitals make up all the ALS patients who received medication prescriptions. The 6.7% figure assumes that all patients with neurodegenerative disease who sought DWD in the state over that period had ALS.
“Similarly, 5% (92 of 1,795) of Oregon ALS patient who died sought medication under DWD between 1998 and 2014,” Dr. Wang said. “This is slightly increased compared to the percentage during the first decade, following enactment of the Oregon law (1998-2007), when 2.7% (26 of 962) of ALS patients died using DWD medication.”
Using Washington state data, researchers also estimated that 0.6% of 73,319 cancer patients and 0.2% of 298,178 people in the state who died of all causes sought DWD over the study period.
A total of 30 (77%) ALS patients who received the deadly prescriptions chose to take them, compared with 67% of all-cause patients who took advantage of the DWD law and 60% of cancer patients.
All 30 patients died. The nine who chose to not take the prescribed medication died after a median of 76 days. The patients who did not take the medication were more likely to be married (88% vs. 69%), to be college educated (100% vs. 74%), and to use a motorized wheelchair (78% vs. 31%).
Those who chose to not take the prescribed medication were also less motivated by loss of dignity (63% vs. 93% among those who took the medication) and by being a burden on others (25% vs. 66%). They were more likely to identify themselves as religious (80% vs. 35%).
Multiple factors may explain why ALS patients made different choices regarding the deadly drugs, lead study author Dr. Wang said in an interview. “We thought that the loss of communication may have played a role based on our finding, as most patients who followed through had more substantial trouble speaking,” he said. “For the patients who ultimately did not choose to take the medication, we found more of them had stronger religious beliefs than those who did not.”
As for pain, he reported that it was not a major issue. “Only about 10% of ALS patients were worried about pain, as opposed to 30% of the general Death with Dignity patients,” he said.
Dr. Ganzini noted that some patients who seek the prescribed drugs “want reassurance that, if their quality of life becomes unbearable, they have the option of physician-assisted death. But, they continue to cope and find reasons to live. As such, they ultimately die of their disease without taking the medications. Others lose the ability to ingest the medications, often because of sudden worsening of their disease.”
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
No specific funding was reported. Dr. Ganzini and Dr. Wang had no disclosures.
BOSTON – A new study estimates that 3.4%-6.7% of amyotrophic lateral sclerosis (ALS) patients in Washington state sought to commit physician-assisted suicide over a 5-year period.
The rate is many times higher than that among cancer patients in the state, researchers found. They also discovered that ALS patients were significantly more likely than were other terminally ill people to use the deadly medication after getting prescriptions for it.
The findings appear to reflect the unique hopelessness facing ALS patients. “They’re not afforded as much denial of decline and death as are patients with other terminal illnesses,” said Linda Ganzini, MD, MPH, a professor of psychiatry and medicine at Oregon Health & Science University, Portland, who has studied end of life in ALS patients.
“Many cancer patients, even in the final days of life, receive treatments that they hope will extend their lives,” she said in an interview after reviewing the study findings. “In contrast, treatments for ALS are minimally effective.”
Physician-assisted suicide is legal in California, Colorado, the District of Columbia, Montana, Oregon, Vermont, and Washington.
A team led by Leo H. Wang, MD, PhD, of the University of Washington, Seattle, examined the medical records of 39 ALS patients who sought medication to end their lives at three hospitals in Seattle from March 2009 to Dec. 31, 2014.
Washington’s Death with Dignity (DWD) law, which went into effect in 2009, allows physicians to prescribe lethal medication if the patient has a terminal illness and a prognosis of less than 6 months to live as judged by two physicians.
The researchers reported their findings, a follow-up to a previous study (Neurology. 2016 Nov 15;87[20]:2117-22), at the annual meeting of the American Academy of Neurology.
The median age of the ALS patients at symptom onset was 64 (range, 42-83), and a median of 712 days passed (range, 207-2,407) from the date of diagnosis to date of prescription for lethal medication.
The median time from prescription to death was 22 days, with at least one patient dying immediately (range, 0-386 days). All 39 patients had limb involvement, and 82%-92% had bulbar involvement, dysarthria, dysphagia, and/or dyspnea.
The researchers estimate that 3.4%-6.7% of 1,146 ALS patients in Washington who died over the time period of the study sought a physician-assisted death. The 3.4% figure assumes that the 39 patients at the three hospitals make up all the ALS patients who received medication prescriptions. The 6.7% figure assumes that all patients with neurodegenerative disease who sought DWD in the state over that period had ALS.
“Similarly, 5% (92 of 1,795) of Oregon ALS patient who died sought medication under DWD between 1998 and 2014,” Dr. Wang said. “This is slightly increased compared to the percentage during the first decade, following enactment of the Oregon law (1998-2007), when 2.7% (26 of 962) of ALS patients died using DWD medication.”
Using Washington state data, researchers also estimated that 0.6% of 73,319 cancer patients and 0.2% of 298,178 people in the state who died of all causes sought DWD over the study period.
A total of 30 (77%) ALS patients who received the deadly prescriptions chose to take them, compared with 67% of all-cause patients who took advantage of the DWD law and 60% of cancer patients.
All 30 patients died. The nine who chose to not take the prescribed medication died after a median of 76 days. The patients who did not take the medication were more likely to be married (88% vs. 69%), to be college educated (100% vs. 74%), and to use a motorized wheelchair (78% vs. 31%).
Those who chose to not take the prescribed medication were also less motivated by loss of dignity (63% vs. 93% among those who took the medication) and by being a burden on others (25% vs. 66%). They were more likely to identify themselves as religious (80% vs. 35%).
Multiple factors may explain why ALS patients made different choices regarding the deadly drugs, lead study author Dr. Wang said in an interview. “We thought that the loss of communication may have played a role based on our finding, as most patients who followed through had more substantial trouble speaking,” he said. “For the patients who ultimately did not choose to take the medication, we found more of them had stronger religious beliefs than those who did not.”
As for pain, he reported that it was not a major issue. “Only about 10% of ALS patients were worried about pain, as opposed to 30% of the general Death with Dignity patients,” he said.
Dr. Ganzini noted that some patients who seek the prescribed drugs “want reassurance that, if their quality of life becomes unbearable, they have the option of physician-assisted death. But, they continue to cope and find reasons to live. As such, they ultimately die of their disease without taking the medications. Others lose the ability to ingest the medications, often because of sudden worsening of their disease.”
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
No specific funding was reported. Dr. Ganzini and Dr. Wang had no disclosures.
BOSTON – A new study estimates that 3.4%-6.7% of amyotrophic lateral sclerosis (ALS) patients in Washington state sought to commit physician-assisted suicide over a 5-year period.
The rate is many times higher than that among cancer patients in the state, researchers found. They also discovered that ALS patients were significantly more likely than were other terminally ill people to use the deadly medication after getting prescriptions for it.
The findings appear to reflect the unique hopelessness facing ALS patients. “They’re not afforded as much denial of decline and death as are patients with other terminal illnesses,” said Linda Ganzini, MD, MPH, a professor of psychiatry and medicine at Oregon Health & Science University, Portland, who has studied end of life in ALS patients.
“Many cancer patients, even in the final days of life, receive treatments that they hope will extend their lives,” she said in an interview after reviewing the study findings. “In contrast, treatments for ALS are minimally effective.”
Physician-assisted suicide is legal in California, Colorado, the District of Columbia, Montana, Oregon, Vermont, and Washington.
A team led by Leo H. Wang, MD, PhD, of the University of Washington, Seattle, examined the medical records of 39 ALS patients who sought medication to end their lives at three hospitals in Seattle from March 2009 to Dec. 31, 2014.
Washington’s Death with Dignity (DWD) law, which went into effect in 2009, allows physicians to prescribe lethal medication if the patient has a terminal illness and a prognosis of less than 6 months to live as judged by two physicians.
The researchers reported their findings, a follow-up to a previous study (Neurology. 2016 Nov 15;87[20]:2117-22), at the annual meeting of the American Academy of Neurology.
The median age of the ALS patients at symptom onset was 64 (range, 42-83), and a median of 712 days passed (range, 207-2,407) from the date of diagnosis to date of prescription for lethal medication.
The median time from prescription to death was 22 days, with at least one patient dying immediately (range, 0-386 days). All 39 patients had limb involvement, and 82%-92% had bulbar involvement, dysarthria, dysphagia, and/or dyspnea.
The researchers estimate that 3.4%-6.7% of 1,146 ALS patients in Washington who died over the time period of the study sought a physician-assisted death. The 3.4% figure assumes that the 39 patients at the three hospitals make up all the ALS patients who received medication prescriptions. The 6.7% figure assumes that all patients with neurodegenerative disease who sought DWD in the state over that period had ALS.
“Similarly, 5% (92 of 1,795) of Oregon ALS patient who died sought medication under DWD between 1998 and 2014,” Dr. Wang said. “This is slightly increased compared to the percentage during the first decade, following enactment of the Oregon law (1998-2007), when 2.7% (26 of 962) of ALS patients died using DWD medication.”
Using Washington state data, researchers also estimated that 0.6% of 73,319 cancer patients and 0.2% of 298,178 people in the state who died of all causes sought DWD over the study period.
A total of 30 (77%) ALS patients who received the deadly prescriptions chose to take them, compared with 67% of all-cause patients who took advantage of the DWD law and 60% of cancer patients.
All 30 patients died. The nine who chose to not take the prescribed medication died after a median of 76 days. The patients who did not take the medication were more likely to be married (88% vs. 69%), to be college educated (100% vs. 74%), and to use a motorized wheelchair (78% vs. 31%).
Those who chose to not take the prescribed medication were also less motivated by loss of dignity (63% vs. 93% among those who took the medication) and by being a burden on others (25% vs. 66%). They were more likely to identify themselves as religious (80% vs. 35%).
Multiple factors may explain why ALS patients made different choices regarding the deadly drugs, lead study author Dr. Wang said in an interview. “We thought that the loss of communication may have played a role based on our finding, as most patients who followed through had more substantial trouble speaking,” he said. “For the patients who ultimately did not choose to take the medication, we found more of them had stronger religious beliefs than those who did not.”
As for pain, he reported that it was not a major issue. “Only about 10% of ALS patients were worried about pain, as opposed to 30% of the general Death with Dignity patients,” he said.
Dr. Ganzini noted that some patients who seek the prescribed drugs “want reassurance that, if their quality of life becomes unbearable, they have the option of physician-assisted death. But, they continue to cope and find reasons to live. As such, they ultimately die of their disease without taking the medications. Others lose the ability to ingest the medications, often because of sudden worsening of their disease.”
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
No specific funding was reported. Dr. Ganzini and Dr. Wang had no disclosures.
At AAN 2017
Key clinical point:
Major finding: An estimated 3.4%-6.7% of ALS patients in Washington state sought physician-assisted death, and 77% took the prescribed deadly medication, a higher rate than all-cause (67%) and cancer patients (60%).
Data source: Analysis of 39 ALS patients who sought deadly medication from three Seattle hospitals from March 2009 to Dec. 31, 2014.
Disclosures: No specific funding was reported, and Dr. Wang had no disclosures.
Schizophrenia patients’ mortality is 14.5 years shorter
SAN DIEGO – A new systematic review and meta-analysis, the first of its kind, suggests that most people with schizophrenia – especially men – do not survive past their 60s.
According to findings released at the 2017 International Congress on Schizophrenia Research, schizophrenia patients appear to die a weighted average of 14.5 years earlier than the rest of the population (95% confidence interval, 11.2-17.8).
The weighted average life expectancy for schizophrenia patients was 64.7 years (95% CI, 61.1-71.3) – 59.9 years for men (95% CI, 55.5-64.3) and 67.6 years for women (95% CI, 63.1-72.1).
“There were no indications that this is improving over time,” lead author Carsten Hjorthøj, PhD, senior researcher at the University of Copenhagen, said in an interview.
The researchers launched their paper in order to provide a wider perspective on lives lost in schizophrenia.
Previous studies had used measures like the standardized mortality ratio, which compared schizophrenia patients with a matched group from the general population, Dr. Hjorthøj said. “Saying that people with schizophrenia are, for instance, 2.5 times more likely to die at any given age may be a little difficult to understand. Identifying the actual number of years they die earlier than the rest of the population is easily understood and will probably also make the issue more obvious to policymakers.”
The researchers, whose findings were previously published in Lancet Psychiatry (2017 Apr;4[4]:295-301), examined 11 studies, mostly from Europe (n = 5) and North America (n = 3) with single studies from Africa, Asia, and Australia. (The lowest life expectancies were reported in Asia and Africa.)
One study was published in 1991, and the rest from 2001-2016. Together, the studies tracked 302,691 patients with schizophrenia.
What explains the gap between the life expectancies in men and women? “We did not investigate the reasons for this,” Dr. Hjorthøj said, “but men typically have worse adherence to treatment than women, and men typically also have poorer health-seeking behavior than women even in the general population. Given that schizophrenia is associated with a multitude of adverse somatic outcomes, this could then be aggravated by this difference in health-seeking behavior.”
In addition, he said, “It could also be the case that use of alcohol, tobacco, and illicit substances is even more skewed in schizophrenia than in the general population. Finally, it could also be attributed in part to the fact that men are more likely than women to die from suicide.”
Dr. Hjorthøj said that the findings emphasize the importance of treating physical symptoms in people with schizophrenia.
“I am aware of several cases where physical complaints were not taken seriously because they were raised by people who were known to suffer from delusions,” he said. “But, I do believe that most psychiatrists are aware of this, and the problem is perhaps more that the system in general is underfunded and that somatic and psychiatric hospitals are not good enough at communicating with each other.”
The researchers report no funding and no disclosures.
SAN DIEGO – A new systematic review and meta-analysis, the first of its kind, suggests that most people with schizophrenia – especially men – do not survive past their 60s.
According to findings released at the 2017 International Congress on Schizophrenia Research, schizophrenia patients appear to die a weighted average of 14.5 years earlier than the rest of the population (95% confidence interval, 11.2-17.8).
The weighted average life expectancy for schizophrenia patients was 64.7 years (95% CI, 61.1-71.3) – 59.9 years for men (95% CI, 55.5-64.3) and 67.6 years for women (95% CI, 63.1-72.1).
“There were no indications that this is improving over time,” lead author Carsten Hjorthøj, PhD, senior researcher at the University of Copenhagen, said in an interview.
The researchers launched their paper in order to provide a wider perspective on lives lost in schizophrenia.
Previous studies had used measures like the standardized mortality ratio, which compared schizophrenia patients with a matched group from the general population, Dr. Hjorthøj said. “Saying that people with schizophrenia are, for instance, 2.5 times more likely to die at any given age may be a little difficult to understand. Identifying the actual number of years they die earlier than the rest of the population is easily understood and will probably also make the issue more obvious to policymakers.”
The researchers, whose findings were previously published in Lancet Psychiatry (2017 Apr;4[4]:295-301), examined 11 studies, mostly from Europe (n = 5) and North America (n = 3) with single studies from Africa, Asia, and Australia. (The lowest life expectancies were reported in Asia and Africa.)
One study was published in 1991, and the rest from 2001-2016. Together, the studies tracked 302,691 patients with schizophrenia.
What explains the gap between the life expectancies in men and women? “We did not investigate the reasons for this,” Dr. Hjorthøj said, “but men typically have worse adherence to treatment than women, and men typically also have poorer health-seeking behavior than women even in the general population. Given that schizophrenia is associated with a multitude of adverse somatic outcomes, this could then be aggravated by this difference in health-seeking behavior.”
In addition, he said, “It could also be the case that use of alcohol, tobacco, and illicit substances is even more skewed in schizophrenia than in the general population. Finally, it could also be attributed in part to the fact that men are more likely than women to die from suicide.”
Dr. Hjorthøj said that the findings emphasize the importance of treating physical symptoms in people with schizophrenia.
“I am aware of several cases where physical complaints were not taken seriously because they were raised by people who were known to suffer from delusions,” he said. “But, I do believe that most psychiatrists are aware of this, and the problem is perhaps more that the system in general is underfunded and that somatic and psychiatric hospitals are not good enough at communicating with each other.”
The researchers report no funding and no disclosures.
SAN DIEGO – A new systematic review and meta-analysis, the first of its kind, suggests that most people with schizophrenia – especially men – do not survive past their 60s.
According to findings released at the 2017 International Congress on Schizophrenia Research, schizophrenia patients appear to die a weighted average of 14.5 years earlier than the rest of the population (95% confidence interval, 11.2-17.8).
The weighted average life expectancy for schizophrenia patients was 64.7 years (95% CI, 61.1-71.3) – 59.9 years for men (95% CI, 55.5-64.3) and 67.6 years for women (95% CI, 63.1-72.1).
“There were no indications that this is improving over time,” lead author Carsten Hjorthøj, PhD, senior researcher at the University of Copenhagen, said in an interview.
The researchers launched their paper in order to provide a wider perspective on lives lost in schizophrenia.
Previous studies had used measures like the standardized mortality ratio, which compared schizophrenia patients with a matched group from the general population, Dr. Hjorthøj said. “Saying that people with schizophrenia are, for instance, 2.5 times more likely to die at any given age may be a little difficult to understand. Identifying the actual number of years they die earlier than the rest of the population is easily understood and will probably also make the issue more obvious to policymakers.”
The researchers, whose findings were previously published in Lancet Psychiatry (2017 Apr;4[4]:295-301), examined 11 studies, mostly from Europe (n = 5) and North America (n = 3) with single studies from Africa, Asia, and Australia. (The lowest life expectancies were reported in Asia and Africa.)
One study was published in 1991, and the rest from 2001-2016. Together, the studies tracked 302,691 patients with schizophrenia.
What explains the gap between the life expectancies in men and women? “We did not investigate the reasons for this,” Dr. Hjorthøj said, “but men typically have worse adherence to treatment than women, and men typically also have poorer health-seeking behavior than women even in the general population. Given that schizophrenia is associated with a multitude of adverse somatic outcomes, this could then be aggravated by this difference in health-seeking behavior.”
In addition, he said, “It could also be the case that use of alcohol, tobacco, and illicit substances is even more skewed in schizophrenia than in the general population. Finally, it could also be attributed in part to the fact that men are more likely than women to die from suicide.”
Dr. Hjorthøj said that the findings emphasize the importance of treating physical symptoms in people with schizophrenia.
“I am aware of several cases where physical complaints were not taken seriously because they were raised by people who were known to suffer from delusions,” he said. “But, I do believe that most psychiatrists are aware of this, and the problem is perhaps more that the system in general is underfunded and that somatic and psychiatric hospitals are not good enough at communicating with each other.”
The researchers report no funding and no disclosures.
At the ICSR Biennial Meeting
Key clinical point:
Major finding: Schizophrenia patients die a weighted average of 14.5 years earlier than those in the general population (95% CI, 11.2-17.8), and their weighted average life expectancy is 64.7 years (95% CI, 61-71.3) – 59.9 for men (95% CI, 55.5-64.3) and 67.6 for women (95% CI, 63.1-72.1).
Data source: Review and meta-analysis of 11 studies of 302,691 schizophrenia patients.
Disclosures: No specific funding was reported, and the authors reported no disclosures.