User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
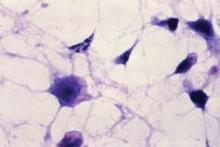
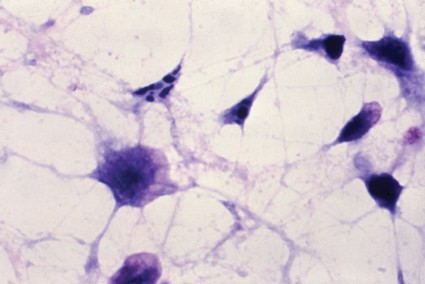
Fecal transplant pill targets C. difficile infection
SAN FRANCISCO – Fecal microbiota transplantation via a pill appears to be as effective as other delivery methods, according to Dr. Thomas Louie.
Typically, fecal microbiota transplantation (FMT) using feces from healthy donors is delivered by enema, colonoscopy, or nasogastric tube to rebalance the bacteria in the gastrointestinal system of patients with recurrent Clostridium difficile infection, but Dr. Louie of the University of Calgary (Alta.) has developed a pill formulation that had 100% efficacy at 3-months’ follow-up in the first 32 patients he treated. Some of those patients have been followed for up to 3 years, and they remain C. difficile free.
The findings were reported at an annual scientific meeting on infectious diseases.
"I’m happy to report that we’ve basically had no recurrences in any of [the 32 patients]," Dr. Louie said during a press conference, noting that one patient who required treatment with antibiotics after FMT developed what Dr. Louie said may be a mild recurrence associated with the antibiotic use.
He has been performing FMT via the enema route since 1996and has treated numerous patients with a very high success rate. He said he came up with the idea for a pill formulation when he encountered a patient who failed to respond to FMT enema on two occasions because of an inability to retain the high-volume treatment for her C. difficile infection, and who was unable to undergo nasogastric delivery because of esophageal varices.
Using freshly passed fecal matter donated, in most cases, by patients’ family members, Dr. Louie said he concentrates the fecal bacteria using a serial centrifugation process that results in pelleted fecal microbes in the sediment of the last centrifugation, and encapsulates the product inside three layers of gelatin capsule to ensure delivery into the small intestine.
The first few patients took 10 pills daily over a period of 4 days with a successful outcome, but the technique has been refined to the ingestion of a "one-shot deal" involving ingestion of 12-34 freshly prepared pills at a single visit.
Patients come in on an empty stomach, take their pills, go home, and wait about an hour and a half, and have some lunch.
"And that’s the end of their C. diff.," he said.
While the dosing isn’t an exact science, it does appear that some patients require fewer pills. For example, a 6-year-old heart transplant patient who had had multiple C. difficile recurrences over a 9-month period was successfully treated with 12 pills, Dr. Louie said.
An analysis of stool following FMT in the treated patients demonstrated that healthy bacteria were significantly increased, while Enterobacteriaceae and Veillonella bacteria were significantly decreased.
Dr. Tom Moore of the University of Kansas in Wichita who moderated the press conference, said in an interview that while none of the methods for delivering FMT have been compared in head-to-head studies, the available evidence suggests they all are highly effective, and the choice depends on patient and physician preference.
Certainly there is a "yuck factor" involved in this treatment for both patients and clinicians. For patients, the pill formulation may seem more palatable; clinicians charged with assembling the capsules may feel otherwise, he said.
But he, along with Dr. Ravi Kamepalli, an infectious disease specialist in group practice in Lima, Ohio, who presented patient satisfaction data for FMT via nasogastric delivery, said clinicians just "need to get over it."
FMT is a life-altering, if not lifesaving procedure for many patients, Dr. Kamepalli said.
In a survey, 28 patients who underwent FMT via nasogastric delivery rated overall satisfaction at 9.6 on a 1-10 scale, and rated ease and likelihood of recommending the procedure to a family member or friend at 9.9 each.
The patients, who had a mean age of 67 years, were surveyed 3 months after the procedure.
To date, Dr. Kamepalli has treated 40 patients with a 98% success rate, he said.
IDWeek is the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The investigators reported having no relevant financial disclosures.
SAN FRANCISCO – Fecal microbiota transplantation via a pill appears to be as effective as other delivery methods, according to Dr. Thomas Louie.
Typically, fecal microbiota transplantation (FMT) using feces from healthy donors is delivered by enema, colonoscopy, or nasogastric tube to rebalance the bacteria in the gastrointestinal system of patients with recurrent Clostridium difficile infection, but Dr. Louie of the University of Calgary (Alta.) has developed a pill formulation that had 100% efficacy at 3-months’ follow-up in the first 32 patients he treated. Some of those patients have been followed for up to 3 years, and they remain C. difficile free.
The findings were reported at an annual scientific meeting on infectious diseases.
"I’m happy to report that we’ve basically had no recurrences in any of [the 32 patients]," Dr. Louie said during a press conference, noting that one patient who required treatment with antibiotics after FMT developed what Dr. Louie said may be a mild recurrence associated with the antibiotic use.
He has been performing FMT via the enema route since 1996and has treated numerous patients with a very high success rate. He said he came up with the idea for a pill formulation when he encountered a patient who failed to respond to FMT enema on two occasions because of an inability to retain the high-volume treatment for her C. difficile infection, and who was unable to undergo nasogastric delivery because of esophageal varices.
Using freshly passed fecal matter donated, in most cases, by patients’ family members, Dr. Louie said he concentrates the fecal bacteria using a serial centrifugation process that results in pelleted fecal microbes in the sediment of the last centrifugation, and encapsulates the product inside three layers of gelatin capsule to ensure delivery into the small intestine.
The first few patients took 10 pills daily over a period of 4 days with a successful outcome, but the technique has been refined to the ingestion of a "one-shot deal" involving ingestion of 12-34 freshly prepared pills at a single visit.
Patients come in on an empty stomach, take their pills, go home, and wait about an hour and a half, and have some lunch.
"And that’s the end of their C. diff.," he said.
While the dosing isn’t an exact science, it does appear that some patients require fewer pills. For example, a 6-year-old heart transplant patient who had had multiple C. difficile recurrences over a 9-month period was successfully treated with 12 pills, Dr. Louie said.
An analysis of stool following FMT in the treated patients demonstrated that healthy bacteria were significantly increased, while Enterobacteriaceae and Veillonella bacteria were significantly decreased.
Dr. Tom Moore of the University of Kansas in Wichita who moderated the press conference, said in an interview that while none of the methods for delivering FMT have been compared in head-to-head studies, the available evidence suggests they all are highly effective, and the choice depends on patient and physician preference.
Certainly there is a "yuck factor" involved in this treatment for both patients and clinicians. For patients, the pill formulation may seem more palatable; clinicians charged with assembling the capsules may feel otherwise, he said.
But he, along with Dr. Ravi Kamepalli, an infectious disease specialist in group practice in Lima, Ohio, who presented patient satisfaction data for FMT via nasogastric delivery, said clinicians just "need to get over it."
FMT is a life-altering, if not lifesaving procedure for many patients, Dr. Kamepalli said.
In a survey, 28 patients who underwent FMT via nasogastric delivery rated overall satisfaction at 9.6 on a 1-10 scale, and rated ease and likelihood of recommending the procedure to a family member or friend at 9.9 each.
The patients, who had a mean age of 67 years, were surveyed 3 months after the procedure.
To date, Dr. Kamepalli has treated 40 patients with a 98% success rate, he said.
IDWeek is the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The investigators reported having no relevant financial disclosures.
SAN FRANCISCO – Fecal microbiota transplantation via a pill appears to be as effective as other delivery methods, according to Dr. Thomas Louie.
Typically, fecal microbiota transplantation (FMT) using feces from healthy donors is delivered by enema, colonoscopy, or nasogastric tube to rebalance the bacteria in the gastrointestinal system of patients with recurrent Clostridium difficile infection, but Dr. Louie of the University of Calgary (Alta.) has developed a pill formulation that had 100% efficacy at 3-months’ follow-up in the first 32 patients he treated. Some of those patients have been followed for up to 3 years, and they remain C. difficile free.
The findings were reported at an annual scientific meeting on infectious diseases.
"I’m happy to report that we’ve basically had no recurrences in any of [the 32 patients]," Dr. Louie said during a press conference, noting that one patient who required treatment with antibiotics after FMT developed what Dr. Louie said may be a mild recurrence associated with the antibiotic use.
He has been performing FMT via the enema route since 1996and has treated numerous patients with a very high success rate. He said he came up with the idea for a pill formulation when he encountered a patient who failed to respond to FMT enema on two occasions because of an inability to retain the high-volume treatment for her C. difficile infection, and who was unable to undergo nasogastric delivery because of esophageal varices.
Using freshly passed fecal matter donated, in most cases, by patients’ family members, Dr. Louie said he concentrates the fecal bacteria using a serial centrifugation process that results in pelleted fecal microbes in the sediment of the last centrifugation, and encapsulates the product inside three layers of gelatin capsule to ensure delivery into the small intestine.
The first few patients took 10 pills daily over a period of 4 days with a successful outcome, but the technique has been refined to the ingestion of a "one-shot deal" involving ingestion of 12-34 freshly prepared pills at a single visit.
Patients come in on an empty stomach, take their pills, go home, and wait about an hour and a half, and have some lunch.
"And that’s the end of their C. diff.," he said.
While the dosing isn’t an exact science, it does appear that some patients require fewer pills. For example, a 6-year-old heart transplant patient who had had multiple C. difficile recurrences over a 9-month period was successfully treated with 12 pills, Dr. Louie said.
An analysis of stool following FMT in the treated patients demonstrated that healthy bacteria were significantly increased, while Enterobacteriaceae and Veillonella bacteria were significantly decreased.
Dr. Tom Moore of the University of Kansas in Wichita who moderated the press conference, said in an interview that while none of the methods for delivering FMT have been compared in head-to-head studies, the available evidence suggests they all are highly effective, and the choice depends on patient and physician preference.
Certainly there is a "yuck factor" involved in this treatment for both patients and clinicians. For patients, the pill formulation may seem more palatable; clinicians charged with assembling the capsules may feel otherwise, he said.
But he, along with Dr. Ravi Kamepalli, an infectious disease specialist in group practice in Lima, Ohio, who presented patient satisfaction data for FMT via nasogastric delivery, said clinicians just "need to get over it."
FMT is a life-altering, if not lifesaving procedure for many patients, Dr. Kamepalli said.
In a survey, 28 patients who underwent FMT via nasogastric delivery rated overall satisfaction at 9.6 on a 1-10 scale, and rated ease and likelihood of recommending the procedure to a family member or friend at 9.9 each.
The patients, who had a mean age of 67 years, were surveyed 3 months after the procedure.
To date, Dr. Kamepalli has treated 40 patients with a 98% success rate, he said.
IDWeek is the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The investigators reported having no relevant financial disclosures.
AT IDWEEK 2013
Major finding: 100% efficacy was seen at 3-months’ follow-up in 32 patients who had FMT via a pill, and patient satisfaction scores were high in 28 patients who had FMT via nasogastric tube.
Data source: A case series and a survey.
Disclosures: The investigators reported having no relevant financial disclosures.
Palmar-plantar psoriasis? Anti-TNF therapy may be culprit
LAS VEGAS – Psoriasiform eruptions are increasingly recognized as a "real event" among patients treated with anti–tumor necrosis factor therapy, according to Dr. Kenneth B. Gordon.
At Perspectives in Rheumatic Diseases 2013, Dr. Gordon of Northwestern University in Chicago presented the case of a 56-year-old woman with Crohn’s disease who developed a painful rash with fissures on both hands about 8 months after starting adalimumab therapy.
The patient first presented with Crohn’s disease at age 28 and was initially treated with mesalamine and systemic corticosteroids for flares. She became prednisone dependent and was started on azathioprine, but could not tolerate the drug, which caused pancreatitis. She was switched to adalimumab at a dose of 80 mg at week 1, 40 mg at week 2, and then 40 mg every other week thereafter.
The patient had an excellent response, including resolution of the gastrointestinal symptoms she experienced on azathioprine, but the rash, which involved well-demarcated plaques suggestive of a psoriasiform structure, was debilitating, Dr. Gordon said.
Another case, presented by Dr. Iain McInnes of Glasgow University, Scotland, involved a patient undergoing anti–tumor necrosis factor (anti-TNF) therapy for rheumatoid arthritis. That patient also developed palmar-plantar pustular psoriasis, which is the most common presentation of psoriasis induced by anti-TNF agents.
Data on the frequency of this condition are lacking, but Dr. Gordon said his "best guess" based on the available literature is that it occurs in about 1%-2% of patients on TNF blockers.
Dr. McInnes described the frequency as "sufficiently common to know it happens and reassure patients about it; sufficiently uncommon enough not to influence decision making" about prescribing anti-TNF therapy.
According to a 2010 systematic literature review by Dr. Angelique N. Collamer and Dr. Daniel F. Battafarano of the rheumatology service at Brooke Army Medical Center in Fort Sam Houston, Tex., pustular psoriasis occurs in 56% of cases, plaque psoriasis occurs in 50%, and guttate psoriasis occurs in 12% of cases. Most (85%) are new-onset cases, and 15% represent an exacerbation of existing psoriasis. Also, patients who are being treated for a variety of diseases – including rheumatoid arthritis, seronegative spondyloarthropathy, inflammatory bowel disease, and psoriasis – may be affected, and all anti-TNF therapies have been implicated (Semin. Arthritis Rheum. 2010;40:233-40).
However, treatment does not necessarily require stopping the TNF blockers, which is important because many patients don’t want to stop – they’re pleased with the effects of treatment on their underlying disease, Dr. Gordon noted.
He said that the first-line treatment is topical therapy, such as a potent topical corticosteroid, but retinoids may also be needed. Other options include phototherapy and switching to another anti-TNF therapy.
"My philosophy is to try to treat the patient while maintaining [anti-TNF] treatment," he said, noting that stopping anti-TNF therapy is a last resort.
Although the patient Dr. Gordon discussed was not a smoker, smoking is a risk factor for worsening of palm psoriasis. Patients with this condition should be advised to quit smoking, and many smokers will get better just by quitting smoking, Dr. Gordon noted.
Dr. McInnes also outlined his approach to patients presenting with possible anti-TNF therapy–induced psoriasis.
He stressed the importance of a good differential diagnosis, noting that infection may be the actual cause.
A biopsy can sometimes help with the diagnosis, he said.
In general, his treatment approach in cases of true anti-TNF–induced psoriasis is to:
• Continue the anti-TNF therapy when less than 5% of body surface area is affected.
• Change to a different anti-TNF drug if the psoriasis is tolerable but appears "a little more severe or is a palmar-pustular variant." The timing of the change will depend on how the patient responds to cutaneous management.
• Stop the drug if the psoriasis is intolerable, and treat the psoriasis aggressively.
In any of these circumstances, a change in the mode of action of treatment may be warranted, he said at the meeting.
In the review by Dr. Collamer and Dr. Battafarano, 24% of patients resolved off anti-TNF therapy, 5% had partial or no resolution off anti-TNF therapy, 25% had partial resolution on anti-TNF therapy, and 6% had no recurrence with change of anti-TNF. Only 1% had no resolution on anti-TNF therapy, and only 2% who resolved after anti-TNF discontinuation then had recurrence after reintroduction of anti-TNF therapy, while 6% resolved off anti-TNF therapy and then had recurrence with introduction of a different anti-TNF. The outcome was unknown in 5% of patients. A Mayo Clinic series showed a similar breakdown (J. Am. Acad. Dermatol. 2012;67:e179-85).
The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Gordon reported receiving research support and/or honoraria from AbbVie, Amgen, and other companies. Dr. McInnes reported serving as a speaker, adviser, and/or researcher for Janssen, Roche, and other companies.
LAS VEGAS – Psoriasiform eruptions are increasingly recognized as a "real event" among patients treated with anti–tumor necrosis factor therapy, according to Dr. Kenneth B. Gordon.
At Perspectives in Rheumatic Diseases 2013, Dr. Gordon of Northwestern University in Chicago presented the case of a 56-year-old woman with Crohn’s disease who developed a painful rash with fissures on both hands about 8 months after starting adalimumab therapy.
The patient first presented with Crohn’s disease at age 28 and was initially treated with mesalamine and systemic corticosteroids for flares. She became prednisone dependent and was started on azathioprine, but could not tolerate the drug, which caused pancreatitis. She was switched to adalimumab at a dose of 80 mg at week 1, 40 mg at week 2, and then 40 mg every other week thereafter.
The patient had an excellent response, including resolution of the gastrointestinal symptoms she experienced on azathioprine, but the rash, which involved well-demarcated plaques suggestive of a psoriasiform structure, was debilitating, Dr. Gordon said.
Another case, presented by Dr. Iain McInnes of Glasgow University, Scotland, involved a patient undergoing anti–tumor necrosis factor (anti-TNF) therapy for rheumatoid arthritis. That patient also developed palmar-plantar pustular psoriasis, which is the most common presentation of psoriasis induced by anti-TNF agents.
Data on the frequency of this condition are lacking, but Dr. Gordon said his "best guess" based on the available literature is that it occurs in about 1%-2% of patients on TNF blockers.
Dr. McInnes described the frequency as "sufficiently common to know it happens and reassure patients about it; sufficiently uncommon enough not to influence decision making" about prescribing anti-TNF therapy.
According to a 2010 systematic literature review by Dr. Angelique N. Collamer and Dr. Daniel F. Battafarano of the rheumatology service at Brooke Army Medical Center in Fort Sam Houston, Tex., pustular psoriasis occurs in 56% of cases, plaque psoriasis occurs in 50%, and guttate psoriasis occurs in 12% of cases. Most (85%) are new-onset cases, and 15% represent an exacerbation of existing psoriasis. Also, patients who are being treated for a variety of diseases – including rheumatoid arthritis, seronegative spondyloarthropathy, inflammatory bowel disease, and psoriasis – may be affected, and all anti-TNF therapies have been implicated (Semin. Arthritis Rheum. 2010;40:233-40).
However, treatment does not necessarily require stopping the TNF blockers, which is important because many patients don’t want to stop – they’re pleased with the effects of treatment on their underlying disease, Dr. Gordon noted.
He said that the first-line treatment is topical therapy, such as a potent topical corticosteroid, but retinoids may also be needed. Other options include phototherapy and switching to another anti-TNF therapy.
"My philosophy is to try to treat the patient while maintaining [anti-TNF] treatment," he said, noting that stopping anti-TNF therapy is a last resort.
Although the patient Dr. Gordon discussed was not a smoker, smoking is a risk factor for worsening of palm psoriasis. Patients with this condition should be advised to quit smoking, and many smokers will get better just by quitting smoking, Dr. Gordon noted.
Dr. McInnes also outlined his approach to patients presenting with possible anti-TNF therapy–induced psoriasis.
He stressed the importance of a good differential diagnosis, noting that infection may be the actual cause.
A biopsy can sometimes help with the diagnosis, he said.
In general, his treatment approach in cases of true anti-TNF–induced psoriasis is to:
• Continue the anti-TNF therapy when less than 5% of body surface area is affected.
• Change to a different anti-TNF drug if the psoriasis is tolerable but appears "a little more severe or is a palmar-pustular variant." The timing of the change will depend on how the patient responds to cutaneous management.
• Stop the drug if the psoriasis is intolerable, and treat the psoriasis aggressively.
In any of these circumstances, a change in the mode of action of treatment may be warranted, he said at the meeting.
In the review by Dr. Collamer and Dr. Battafarano, 24% of patients resolved off anti-TNF therapy, 5% had partial or no resolution off anti-TNF therapy, 25% had partial resolution on anti-TNF therapy, and 6% had no recurrence with change of anti-TNF. Only 1% had no resolution on anti-TNF therapy, and only 2% who resolved after anti-TNF discontinuation then had recurrence after reintroduction of anti-TNF therapy, while 6% resolved off anti-TNF therapy and then had recurrence with introduction of a different anti-TNF. The outcome was unknown in 5% of patients. A Mayo Clinic series showed a similar breakdown (J. Am. Acad. Dermatol. 2012;67:e179-85).
The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Gordon reported receiving research support and/or honoraria from AbbVie, Amgen, and other companies. Dr. McInnes reported serving as a speaker, adviser, and/or researcher for Janssen, Roche, and other companies.
LAS VEGAS – Psoriasiform eruptions are increasingly recognized as a "real event" among patients treated with anti–tumor necrosis factor therapy, according to Dr. Kenneth B. Gordon.
At Perspectives in Rheumatic Diseases 2013, Dr. Gordon of Northwestern University in Chicago presented the case of a 56-year-old woman with Crohn’s disease who developed a painful rash with fissures on both hands about 8 months after starting adalimumab therapy.
The patient first presented with Crohn’s disease at age 28 and was initially treated with mesalamine and systemic corticosteroids for flares. She became prednisone dependent and was started on azathioprine, but could not tolerate the drug, which caused pancreatitis. She was switched to adalimumab at a dose of 80 mg at week 1, 40 mg at week 2, and then 40 mg every other week thereafter.
The patient had an excellent response, including resolution of the gastrointestinal symptoms she experienced on azathioprine, but the rash, which involved well-demarcated plaques suggestive of a psoriasiform structure, was debilitating, Dr. Gordon said.
Another case, presented by Dr. Iain McInnes of Glasgow University, Scotland, involved a patient undergoing anti–tumor necrosis factor (anti-TNF) therapy for rheumatoid arthritis. That patient also developed palmar-plantar pustular psoriasis, which is the most common presentation of psoriasis induced by anti-TNF agents.
Data on the frequency of this condition are lacking, but Dr. Gordon said his "best guess" based on the available literature is that it occurs in about 1%-2% of patients on TNF blockers.
Dr. McInnes described the frequency as "sufficiently common to know it happens and reassure patients about it; sufficiently uncommon enough not to influence decision making" about prescribing anti-TNF therapy.
According to a 2010 systematic literature review by Dr. Angelique N. Collamer and Dr. Daniel F. Battafarano of the rheumatology service at Brooke Army Medical Center in Fort Sam Houston, Tex., pustular psoriasis occurs in 56% of cases, plaque psoriasis occurs in 50%, and guttate psoriasis occurs in 12% of cases. Most (85%) are new-onset cases, and 15% represent an exacerbation of existing psoriasis. Also, patients who are being treated for a variety of diseases – including rheumatoid arthritis, seronegative spondyloarthropathy, inflammatory bowel disease, and psoriasis – may be affected, and all anti-TNF therapies have been implicated (Semin. Arthritis Rheum. 2010;40:233-40).
However, treatment does not necessarily require stopping the TNF blockers, which is important because many patients don’t want to stop – they’re pleased with the effects of treatment on their underlying disease, Dr. Gordon noted.
He said that the first-line treatment is topical therapy, such as a potent topical corticosteroid, but retinoids may also be needed. Other options include phototherapy and switching to another anti-TNF therapy.
"My philosophy is to try to treat the patient while maintaining [anti-TNF] treatment," he said, noting that stopping anti-TNF therapy is a last resort.
Although the patient Dr. Gordon discussed was not a smoker, smoking is a risk factor for worsening of palm psoriasis. Patients with this condition should be advised to quit smoking, and many smokers will get better just by quitting smoking, Dr. Gordon noted.
Dr. McInnes also outlined his approach to patients presenting with possible anti-TNF therapy–induced psoriasis.
He stressed the importance of a good differential diagnosis, noting that infection may be the actual cause.
A biopsy can sometimes help with the diagnosis, he said.
In general, his treatment approach in cases of true anti-TNF–induced psoriasis is to:
• Continue the anti-TNF therapy when less than 5% of body surface area is affected.
• Change to a different anti-TNF drug if the psoriasis is tolerable but appears "a little more severe or is a palmar-pustular variant." The timing of the change will depend on how the patient responds to cutaneous management.
• Stop the drug if the psoriasis is intolerable, and treat the psoriasis aggressively.
In any of these circumstances, a change in the mode of action of treatment may be warranted, he said at the meeting.
In the review by Dr. Collamer and Dr. Battafarano, 24% of patients resolved off anti-TNF therapy, 5% had partial or no resolution off anti-TNF therapy, 25% had partial resolution on anti-TNF therapy, and 6% had no recurrence with change of anti-TNF. Only 1% had no resolution on anti-TNF therapy, and only 2% who resolved after anti-TNF discontinuation then had recurrence after reintroduction of anti-TNF therapy, while 6% resolved off anti-TNF therapy and then had recurrence with introduction of a different anti-TNF. The outcome was unknown in 5% of patients. A Mayo Clinic series showed a similar breakdown (J. Am. Acad. Dermatol. 2012;67:e179-85).
The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Gordon reported receiving research support and/or honoraria from AbbVie, Amgen, and other companies. Dr. McInnes reported serving as a speaker, adviser, and/or researcher for Janssen, Roche, and other companies.
EXPERT ANALYSIS FROM PERSPECTIVES IN RHEUMATIC DISEASES 2013
Study shows no benefit with cesarean section for twins
No significant differences were seen in the risk of adverse neonatal outcomes between women with twin pregnancy who underwent planned cesarean delivery and those who underwent planned vaginal delivery as part of the randomized Twin Birth Study.
The rate of a composite outcome of fetal or neonatal death or serious neonatal morbidity among 1,393 women in 25 countries assigned to undergo planned cesarean delivery was 2.2%, compared with 1.9% among 1,393 women assigned to planned vaginal delivery (odds ratio with planned cesarean delivery, 1.16), Dr. Jon F.R. Barrett of Sunnybrook Health Sciences Centre, Toronto, and his colleagues reported on behalf of the Twin Birth Study Collaborative Group.
Another outcome – a composite of maternal death or serious maternal morbidity – also did not differ between the groups, occurring in 7.3% of the cesarean group and 8.5% of the vaginal delivery group. This finding, however, may be explained in part by the high rate of cesarean section in the planned vaginal delivery group, they said (N. Engl. J. Med. 2013;369:1295-305).
Secondary outcomes, including death or a poor neurodevelopmental outcome among the children at 2 years and problematic incontinence among the mothers at 2 years, will be reported subsequently.
Participants in the Twin Birth Study were women between 32 weeks 0 days gestation and 38 weeks 6 days gestation between Dec. 13, 2003, and April 4, 2011, who were pregnant with twins with an estimated weight of between 1,500 g and 4,000 g each and whose first twin was in the cephalic presentation. The women were randomized to planned cesarean delivery or vaginal delivery with cesarean only if indicated and were followed until 28 days after delivery.
Most (90.7%) in the cesarean delivery group delivered by cesarean as planned, and 43.8% in the planned vaginal delivery group required cesarean delivery, the investigators said
The findings are of note because the twin pregnancy rate has increased over time, largely due to assisted reproductive technologies, and the rate of cesarean section for twins also has increased in the wake of studies suggesting a possible benefit of cesarean delivery with respect to perinatal outcomes.
"There are several possible reasons why our results differ from previous observational data: we avoided selection bias, we ensured the presence of an experienced obstetrician at delivery, and many of the twins in our study were born preterm," the investigators said, adding that no significant interaction between treatment group and baseline variables was seen, which suggests that no significant benefit occurred with planned cesarean delivery for any subgroup tested.
"However, our study was not powered for these subgroup analyses. Further study may be warranted for the gestational-age subgroup of 37 to 38 weeks, particularly given the limited number of infants in this subgroup," they said.
Overall, the results of this study are consistent with no more than a 23% reduction and no more than a 74% increase in the odds of fetal or neonatal death or serious neonatal morbidity with planned cesarean vs. planned vaginal delivery, they said.
"In conclusion, we found no benefits of planned cesarean section, as compared with planned vaginal delivery, for the delivery of twins between 32 and 38 weeks of gestation, if the first twin was in the cephalic presentation," they wrote.
This study was supported by a grant from the Canadian Institutes of Health Research. The authors reported having no relevant conflicts of interest.
Although the Twin Birth Study was a "massive logistic effort," the findings are unlikely to lead to a major change in the use of cesarean delivery for twin pregnancies, according to Dr. Michael F. Greene.
For one thing, the results do not indicate that all sets of twin should be delivered vaginally, he said.
"Obstetricians exercising their best clinical judgment delivered both twins by cesarean section in nearly 40% of the women assigned to planned vaginal delivery, which undoubtedly contributed to the salutary outcomes," he said, noting that the results nonetheless suggest that "a plan to deliver appropriately selected sets of twins vaginally is a reasonably safe choice in skilled hands."
Still, are the results likely to "bend the cesarean delivery curve for twin pregnancies?" he asked.
"Given the trends in patient demographic characteristics and preferences, the virtual disappearance of vaginal delivery in cases of breech presentation, and the dramatic reduction in instrumented vaginal delivery (and the associated gradual disappearance of the skills necessary to perform these procedures among obstetricians), it seems unlikely that we will see a major change in use of cesarean delivery for twins nationwide," he said.
Dr. Greene is an ob.gyn. at Massachusetts General Hospital, Boston, and an associate editor for the New England Journal of Medicine. He reported having no other disclosures. Dr. Greene’s remarks were in an editorial in response to Dr. Barrett and his colleagues’ study (N. Engl. J. Med. 2013;369:1365-6).
Although the Twin Birth Study was a "massive logistic effort," the findings are unlikely to lead to a major change in the use of cesarean delivery for twin pregnancies, according to Dr. Michael F. Greene.
For one thing, the results do not indicate that all sets of twin should be delivered vaginally, he said.
"Obstetricians exercising their best clinical judgment delivered both twins by cesarean section in nearly 40% of the women assigned to planned vaginal delivery, which undoubtedly contributed to the salutary outcomes," he said, noting that the results nonetheless suggest that "a plan to deliver appropriately selected sets of twins vaginally is a reasonably safe choice in skilled hands."
Still, are the results likely to "bend the cesarean delivery curve for twin pregnancies?" he asked.
"Given the trends in patient demographic characteristics and preferences, the virtual disappearance of vaginal delivery in cases of breech presentation, and the dramatic reduction in instrumented vaginal delivery (and the associated gradual disappearance of the skills necessary to perform these procedures among obstetricians), it seems unlikely that we will see a major change in use of cesarean delivery for twins nationwide," he said.
Dr. Greene is an ob.gyn. at Massachusetts General Hospital, Boston, and an associate editor for the New England Journal of Medicine. He reported having no other disclosures. Dr. Greene’s remarks were in an editorial in response to Dr. Barrett and his colleagues’ study (N. Engl. J. Med. 2013;369:1365-6).
Although the Twin Birth Study was a "massive logistic effort," the findings are unlikely to lead to a major change in the use of cesarean delivery for twin pregnancies, according to Dr. Michael F. Greene.
For one thing, the results do not indicate that all sets of twin should be delivered vaginally, he said.
"Obstetricians exercising their best clinical judgment delivered both twins by cesarean section in nearly 40% of the women assigned to planned vaginal delivery, which undoubtedly contributed to the salutary outcomes," he said, noting that the results nonetheless suggest that "a plan to deliver appropriately selected sets of twins vaginally is a reasonably safe choice in skilled hands."
Still, are the results likely to "bend the cesarean delivery curve for twin pregnancies?" he asked.
"Given the trends in patient demographic characteristics and preferences, the virtual disappearance of vaginal delivery in cases of breech presentation, and the dramatic reduction in instrumented vaginal delivery (and the associated gradual disappearance of the skills necessary to perform these procedures among obstetricians), it seems unlikely that we will see a major change in use of cesarean delivery for twins nationwide," he said.
Dr. Greene is an ob.gyn. at Massachusetts General Hospital, Boston, and an associate editor for the New England Journal of Medicine. He reported having no other disclosures. Dr. Greene’s remarks were in an editorial in response to Dr. Barrett and his colleagues’ study (N. Engl. J. Med. 2013;369:1365-6).
No significant differences were seen in the risk of adverse neonatal outcomes between women with twin pregnancy who underwent planned cesarean delivery and those who underwent planned vaginal delivery as part of the randomized Twin Birth Study.
The rate of a composite outcome of fetal or neonatal death or serious neonatal morbidity among 1,393 women in 25 countries assigned to undergo planned cesarean delivery was 2.2%, compared with 1.9% among 1,393 women assigned to planned vaginal delivery (odds ratio with planned cesarean delivery, 1.16), Dr. Jon F.R. Barrett of Sunnybrook Health Sciences Centre, Toronto, and his colleagues reported on behalf of the Twin Birth Study Collaborative Group.
Another outcome – a composite of maternal death or serious maternal morbidity – also did not differ between the groups, occurring in 7.3% of the cesarean group and 8.5% of the vaginal delivery group. This finding, however, may be explained in part by the high rate of cesarean section in the planned vaginal delivery group, they said (N. Engl. J. Med. 2013;369:1295-305).
Secondary outcomes, including death or a poor neurodevelopmental outcome among the children at 2 years and problematic incontinence among the mothers at 2 years, will be reported subsequently.
Participants in the Twin Birth Study were women between 32 weeks 0 days gestation and 38 weeks 6 days gestation between Dec. 13, 2003, and April 4, 2011, who were pregnant with twins with an estimated weight of between 1,500 g and 4,000 g each and whose first twin was in the cephalic presentation. The women were randomized to planned cesarean delivery or vaginal delivery with cesarean only if indicated and were followed until 28 days after delivery.
Most (90.7%) in the cesarean delivery group delivered by cesarean as planned, and 43.8% in the planned vaginal delivery group required cesarean delivery, the investigators said
The findings are of note because the twin pregnancy rate has increased over time, largely due to assisted reproductive technologies, and the rate of cesarean section for twins also has increased in the wake of studies suggesting a possible benefit of cesarean delivery with respect to perinatal outcomes.
"There are several possible reasons why our results differ from previous observational data: we avoided selection bias, we ensured the presence of an experienced obstetrician at delivery, and many of the twins in our study were born preterm," the investigators said, adding that no significant interaction between treatment group and baseline variables was seen, which suggests that no significant benefit occurred with planned cesarean delivery for any subgroup tested.
"However, our study was not powered for these subgroup analyses. Further study may be warranted for the gestational-age subgroup of 37 to 38 weeks, particularly given the limited number of infants in this subgroup," they said.
Overall, the results of this study are consistent with no more than a 23% reduction and no more than a 74% increase in the odds of fetal or neonatal death or serious neonatal morbidity with planned cesarean vs. planned vaginal delivery, they said.
"In conclusion, we found no benefits of planned cesarean section, as compared with planned vaginal delivery, for the delivery of twins between 32 and 38 weeks of gestation, if the first twin was in the cephalic presentation," they wrote.
This study was supported by a grant from the Canadian Institutes of Health Research. The authors reported having no relevant conflicts of interest.
No significant differences were seen in the risk of adverse neonatal outcomes between women with twin pregnancy who underwent planned cesarean delivery and those who underwent planned vaginal delivery as part of the randomized Twin Birth Study.
The rate of a composite outcome of fetal or neonatal death or serious neonatal morbidity among 1,393 women in 25 countries assigned to undergo planned cesarean delivery was 2.2%, compared with 1.9% among 1,393 women assigned to planned vaginal delivery (odds ratio with planned cesarean delivery, 1.16), Dr. Jon F.R. Barrett of Sunnybrook Health Sciences Centre, Toronto, and his colleagues reported on behalf of the Twin Birth Study Collaborative Group.
Another outcome – a composite of maternal death or serious maternal morbidity – also did not differ between the groups, occurring in 7.3% of the cesarean group and 8.5% of the vaginal delivery group. This finding, however, may be explained in part by the high rate of cesarean section in the planned vaginal delivery group, they said (N. Engl. J. Med. 2013;369:1295-305).
Secondary outcomes, including death or a poor neurodevelopmental outcome among the children at 2 years and problematic incontinence among the mothers at 2 years, will be reported subsequently.
Participants in the Twin Birth Study were women between 32 weeks 0 days gestation and 38 weeks 6 days gestation between Dec. 13, 2003, and April 4, 2011, who were pregnant with twins with an estimated weight of between 1,500 g and 4,000 g each and whose first twin was in the cephalic presentation. The women were randomized to planned cesarean delivery or vaginal delivery with cesarean only if indicated and were followed until 28 days after delivery.
Most (90.7%) in the cesarean delivery group delivered by cesarean as planned, and 43.8% in the planned vaginal delivery group required cesarean delivery, the investigators said
The findings are of note because the twin pregnancy rate has increased over time, largely due to assisted reproductive technologies, and the rate of cesarean section for twins also has increased in the wake of studies suggesting a possible benefit of cesarean delivery with respect to perinatal outcomes.
"There are several possible reasons why our results differ from previous observational data: we avoided selection bias, we ensured the presence of an experienced obstetrician at delivery, and many of the twins in our study were born preterm," the investigators said, adding that no significant interaction between treatment group and baseline variables was seen, which suggests that no significant benefit occurred with planned cesarean delivery for any subgroup tested.
"However, our study was not powered for these subgroup analyses. Further study may be warranted for the gestational-age subgroup of 37 to 38 weeks, particularly given the limited number of infants in this subgroup," they said.
Overall, the results of this study are consistent with no more than a 23% reduction and no more than a 74% increase in the odds of fetal or neonatal death or serious neonatal morbidity with planned cesarean vs. planned vaginal delivery, they said.
"In conclusion, we found no benefits of planned cesarean section, as compared with planned vaginal delivery, for the delivery of twins between 32 and 38 weeks of gestation, if the first twin was in the cephalic presentation," they wrote.
This study was supported by a grant from the Canadian Institutes of Health Research. The authors reported having no relevant conflicts of interest.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major finding: The rates of adverse neonatal outcome with cesarean vs. vaginal delivery were similar (2.2% and 1.9%, respectively).
Data source: The randomized Twin Birth Study, involving 2,804 women.
Disclosures: This study was supported by a grant from the Canadian Institutes of Health Research. The authors reported having no relevant conflicts of interest.
Extended delirium raises cognitive risk in critically ill
Critical illness survivors who experienced a long period of delirium during an intensive care unit stay can have long-term global cognition and executive function scores similar to those seen in traumatic brain injury and Alzheimer’s patients, according to a multicenter prospective cohort study.
The finding of an association between longer duration of delirium and worse long-term global cognition and executive function was independent of sedative or analgesic medication use, age, preexisting cognitive impairment, the burden of coexisting conditions, and ongoing organ failure during ICU care, Dr. P.P. Pandharipande of Vanderbilt University, Nashville, Tenn., and his colleagues reported. The findings were published Oct. 2 in The New England Journal of Medicine.
Of 821 patients with respiratory failure, cardiogenic shock, or septic shock who were treated in a medical or surgical ICU, 6% had cognitive impairment at baseline and 74% experienced delirium during their hospital stay. Median global cognition scores at 3 and 12 months as assessed by the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) were 79 and 80, respectively.
"These scores were approximately 1.5 [standard deviations] below the age-adjusted population mean of 100 plus or minus 15 and were similar to scores for patients with mild cognitive impairment. At 3-months, 40% of the patients had global cognition scores that were worse than those typically seen in patients with moderate traumatic brain injury, and 26% had scores 2 SD below the population means, which were similar to scores for patients with mild Alzheimer’s disease," the investigators reported.
The deficits occurred regardless of patient age and persisted to 12 months, with 34% and 24% of patients demonstrating scores similar to those for patients with moderate traumatic brain injury and for patients with mild Alzheimer’s disease, respectively, they said (N. Engl. J. Med. 2013;369:1306-16).
Duration of delirium was significantly associated with worse global cognition and significantly worse executive function at both 3- and 12-month follow-up. For example, patients with a 5-day mean duration of delirium had mean RBANS scores that were 6.3 points lower at 3 months and 5.6 points lower at 12 months than those with no delirium. They had Trails B executive-function scores that were 5.1 points lower at 3 months and 6.0 points lower at 12 months.
"A longer duration of delirium was also a risk factor for worse function in several individual RBANS domains," Dr. Pandharipande and his associates noted.
Although the investigators hypothesized that higher doses of sedative and analgesic use also would be independently associated with more severe cognitive impairment at 12 months, this did not prove to be the case. The use of higher benzodiazepine doses during hospitalization, however, was associated with worse executive function scores at 3 months.
The patients, who had a median age of 61 years and high severity of illness, were enrolled in the Bringing to Light the Risk Factors and Incidence of Neuropsychological Dysfunction in ICU Survivors (BRAIN-ICU) study between March, 2007 and May, 2010. Delirium was assessed by the Confusion Assessment Method for the ICU, and level of consciousness was assessed using the Richmond Agitation-Sedation Scale.
It remains unclear whether any preventive or treatment strategies can reduce the risk of long-term cognitive impairment after critical illness, they said, noting that this is of concern, as "long-term cognitive impairment after critical illness may be a growing public health problem, given the large number of acutely ill patients being treated in intensive care units globally."
Though limited by an inability to test patients’ cognition before their emergent illness (although the investigators took several precautions to address this limitation), and by the fact that some patients were unable to complete all cognitive tests, the findings nevertheless demonstrate that cognitive impairment after critical illness is very common – even more so among those with longer duration of delirium – and can persist for at least 1 year, they said.
It is "possible that patients who are vulnerable to delirium owing to severe critical illness are also vulnerable to long-term cognitive impairment and that delirium does not play a causal role in the development of persistent cognitive impairment," investigators noted.
This study was supported by grants from the National Institutes of Health and the Veteran’s Affairs Clinical Science Research and Development Service, as well as by a Mentored Research Training Grant from the Foundation for Anesthesia Education and Research and the VA Tennessee Valley Geriatric Research Education and Clinical Center. Multiple study authors reported disclosures; details are available with the full text of the article at NEJM.org.
In their study on delirium and cognitive outcomes in critically ill patients, Dr. Pandharipande and colleagues "unequivocally show that neurocognitive dysfunction is an important and prevalent public health concern after critical illness," Dr. Margaret Herridge and Jill I. Cameron, Ph.D., wrote in an editorial accompanying the research article.
The findings underscore that surveillance and intervention for delirium are crucial, and set a new standard for longitudinal cognitive-outcome studies, they said (N. Engl. J. Med. 2013;369:1367-8).
Further delineation of clinical-risk groupings and risk modifiers by means of genetic and basic science work will provide a useful longitudinal approach to critical illness. Crucial next steps include prioritization of basic research and translational collaborations, they said.
"Without this detailed knowledge, we are merely guessing about how to proceed," Dr. Herridge and Dr. Cameron noted.
As more knowledge accumulates about neurocognitive and functional morbidity, better education can be provided to patients, families, physicians, and policymakers, which "should fuel an informed discussion about what it means for our patients to survive an episode of critical illness, how it changes families forever, and when the degree of suffering and futility becomes unacceptable from a patient-centered and societal standpoint," they added.
Dr. Herridge and Dr. Cameron are with the University of Toronto. They reported having no disclosures.
In their study on delirium and cognitive outcomes in critically ill patients, Dr. Pandharipande and colleagues "unequivocally show that neurocognitive dysfunction is an important and prevalent public health concern after critical illness," Dr. Margaret Herridge and Jill I. Cameron, Ph.D., wrote in an editorial accompanying the research article.
The findings underscore that surveillance and intervention for delirium are crucial, and set a new standard for longitudinal cognitive-outcome studies, they said (N. Engl. J. Med. 2013;369:1367-8).
Further delineation of clinical-risk groupings and risk modifiers by means of genetic and basic science work will provide a useful longitudinal approach to critical illness. Crucial next steps include prioritization of basic research and translational collaborations, they said.
"Without this detailed knowledge, we are merely guessing about how to proceed," Dr. Herridge and Dr. Cameron noted.
As more knowledge accumulates about neurocognitive and functional morbidity, better education can be provided to patients, families, physicians, and policymakers, which "should fuel an informed discussion about what it means for our patients to survive an episode of critical illness, how it changes families forever, and when the degree of suffering and futility becomes unacceptable from a patient-centered and societal standpoint," they added.
Dr. Herridge and Dr. Cameron are with the University of Toronto. They reported having no disclosures.
In their study on delirium and cognitive outcomes in critically ill patients, Dr. Pandharipande and colleagues "unequivocally show that neurocognitive dysfunction is an important and prevalent public health concern after critical illness," Dr. Margaret Herridge and Jill I. Cameron, Ph.D., wrote in an editorial accompanying the research article.
The findings underscore that surveillance and intervention for delirium are crucial, and set a new standard for longitudinal cognitive-outcome studies, they said (N. Engl. J. Med. 2013;369:1367-8).
Further delineation of clinical-risk groupings and risk modifiers by means of genetic and basic science work will provide a useful longitudinal approach to critical illness. Crucial next steps include prioritization of basic research and translational collaborations, they said.
"Without this detailed knowledge, we are merely guessing about how to proceed," Dr. Herridge and Dr. Cameron noted.
As more knowledge accumulates about neurocognitive and functional morbidity, better education can be provided to patients, families, physicians, and policymakers, which "should fuel an informed discussion about what it means for our patients to survive an episode of critical illness, how it changes families forever, and when the degree of suffering and futility becomes unacceptable from a patient-centered and societal standpoint," they added.
Dr. Herridge and Dr. Cameron are with the University of Toronto. They reported having no disclosures.
Critical illness survivors who experienced a long period of delirium during an intensive care unit stay can have long-term global cognition and executive function scores similar to those seen in traumatic brain injury and Alzheimer’s patients, according to a multicenter prospective cohort study.
The finding of an association between longer duration of delirium and worse long-term global cognition and executive function was independent of sedative or analgesic medication use, age, preexisting cognitive impairment, the burden of coexisting conditions, and ongoing organ failure during ICU care, Dr. P.P. Pandharipande of Vanderbilt University, Nashville, Tenn., and his colleagues reported. The findings were published Oct. 2 in The New England Journal of Medicine.
Of 821 patients with respiratory failure, cardiogenic shock, or septic shock who were treated in a medical or surgical ICU, 6% had cognitive impairment at baseline and 74% experienced delirium during their hospital stay. Median global cognition scores at 3 and 12 months as assessed by the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) were 79 and 80, respectively.
"These scores were approximately 1.5 [standard deviations] below the age-adjusted population mean of 100 plus or minus 15 and were similar to scores for patients with mild cognitive impairment. At 3-months, 40% of the patients had global cognition scores that were worse than those typically seen in patients with moderate traumatic brain injury, and 26% had scores 2 SD below the population means, which were similar to scores for patients with mild Alzheimer’s disease," the investigators reported.
The deficits occurred regardless of patient age and persisted to 12 months, with 34% and 24% of patients demonstrating scores similar to those for patients with moderate traumatic brain injury and for patients with mild Alzheimer’s disease, respectively, they said (N. Engl. J. Med. 2013;369:1306-16).
Duration of delirium was significantly associated with worse global cognition and significantly worse executive function at both 3- and 12-month follow-up. For example, patients with a 5-day mean duration of delirium had mean RBANS scores that were 6.3 points lower at 3 months and 5.6 points lower at 12 months than those with no delirium. They had Trails B executive-function scores that were 5.1 points lower at 3 months and 6.0 points lower at 12 months.
"A longer duration of delirium was also a risk factor for worse function in several individual RBANS domains," Dr. Pandharipande and his associates noted.
Although the investigators hypothesized that higher doses of sedative and analgesic use also would be independently associated with more severe cognitive impairment at 12 months, this did not prove to be the case. The use of higher benzodiazepine doses during hospitalization, however, was associated with worse executive function scores at 3 months.
The patients, who had a median age of 61 years and high severity of illness, were enrolled in the Bringing to Light the Risk Factors and Incidence of Neuropsychological Dysfunction in ICU Survivors (BRAIN-ICU) study between March, 2007 and May, 2010. Delirium was assessed by the Confusion Assessment Method for the ICU, and level of consciousness was assessed using the Richmond Agitation-Sedation Scale.
It remains unclear whether any preventive or treatment strategies can reduce the risk of long-term cognitive impairment after critical illness, they said, noting that this is of concern, as "long-term cognitive impairment after critical illness may be a growing public health problem, given the large number of acutely ill patients being treated in intensive care units globally."
Though limited by an inability to test patients’ cognition before their emergent illness (although the investigators took several precautions to address this limitation), and by the fact that some patients were unable to complete all cognitive tests, the findings nevertheless demonstrate that cognitive impairment after critical illness is very common – even more so among those with longer duration of delirium – and can persist for at least 1 year, they said.
It is "possible that patients who are vulnerable to delirium owing to severe critical illness are also vulnerable to long-term cognitive impairment and that delirium does not play a causal role in the development of persistent cognitive impairment," investigators noted.
This study was supported by grants from the National Institutes of Health and the Veteran’s Affairs Clinical Science Research and Development Service, as well as by a Mentored Research Training Grant from the Foundation for Anesthesia Education and Research and the VA Tennessee Valley Geriatric Research Education and Clinical Center. Multiple study authors reported disclosures; details are available with the full text of the article at NEJM.org.
Critical illness survivors who experienced a long period of delirium during an intensive care unit stay can have long-term global cognition and executive function scores similar to those seen in traumatic brain injury and Alzheimer’s patients, according to a multicenter prospective cohort study.
The finding of an association between longer duration of delirium and worse long-term global cognition and executive function was independent of sedative or analgesic medication use, age, preexisting cognitive impairment, the burden of coexisting conditions, and ongoing organ failure during ICU care, Dr. P.P. Pandharipande of Vanderbilt University, Nashville, Tenn., and his colleagues reported. The findings were published Oct. 2 in The New England Journal of Medicine.
Of 821 patients with respiratory failure, cardiogenic shock, or septic shock who were treated in a medical or surgical ICU, 6% had cognitive impairment at baseline and 74% experienced delirium during their hospital stay. Median global cognition scores at 3 and 12 months as assessed by the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) were 79 and 80, respectively.
"These scores were approximately 1.5 [standard deviations] below the age-adjusted population mean of 100 plus or minus 15 and were similar to scores for patients with mild cognitive impairment. At 3-months, 40% of the patients had global cognition scores that were worse than those typically seen in patients with moderate traumatic brain injury, and 26% had scores 2 SD below the population means, which were similar to scores for patients with mild Alzheimer’s disease," the investigators reported.
The deficits occurred regardless of patient age and persisted to 12 months, with 34% and 24% of patients demonstrating scores similar to those for patients with moderate traumatic brain injury and for patients with mild Alzheimer’s disease, respectively, they said (N. Engl. J. Med. 2013;369:1306-16).
Duration of delirium was significantly associated with worse global cognition and significantly worse executive function at both 3- and 12-month follow-up. For example, patients with a 5-day mean duration of delirium had mean RBANS scores that were 6.3 points lower at 3 months and 5.6 points lower at 12 months than those with no delirium. They had Trails B executive-function scores that were 5.1 points lower at 3 months and 6.0 points lower at 12 months.
"A longer duration of delirium was also a risk factor for worse function in several individual RBANS domains," Dr. Pandharipande and his associates noted.
Although the investigators hypothesized that higher doses of sedative and analgesic use also would be independently associated with more severe cognitive impairment at 12 months, this did not prove to be the case. The use of higher benzodiazepine doses during hospitalization, however, was associated with worse executive function scores at 3 months.
The patients, who had a median age of 61 years and high severity of illness, were enrolled in the Bringing to Light the Risk Factors and Incidence of Neuropsychological Dysfunction in ICU Survivors (BRAIN-ICU) study between March, 2007 and May, 2010. Delirium was assessed by the Confusion Assessment Method for the ICU, and level of consciousness was assessed using the Richmond Agitation-Sedation Scale.
It remains unclear whether any preventive or treatment strategies can reduce the risk of long-term cognitive impairment after critical illness, they said, noting that this is of concern, as "long-term cognitive impairment after critical illness may be a growing public health problem, given the large number of acutely ill patients being treated in intensive care units globally."
Though limited by an inability to test patients’ cognition before their emergent illness (although the investigators took several precautions to address this limitation), and by the fact that some patients were unable to complete all cognitive tests, the findings nevertheless demonstrate that cognitive impairment after critical illness is very common – even more so among those with longer duration of delirium – and can persist for at least 1 year, they said.
It is "possible that patients who are vulnerable to delirium owing to severe critical illness are also vulnerable to long-term cognitive impairment and that delirium does not play a causal role in the development of persistent cognitive impairment," investigators noted.
This study was supported by grants from the National Institutes of Health and the Veteran’s Affairs Clinical Science Research and Development Service, as well as by a Mentored Research Training Grant from the Foundation for Anesthesia Education and Research and the VA Tennessee Valley Geriatric Research Education and Clinical Center. Multiple study authors reported disclosures; details are available with the full text of the article at NEJM.org.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major finding: Neuropsychological status scores averaged 6.3 points lower at 3 months in patients who experienced 5 days of delirium vs. no delirium while in an ICU.
Data source: A prospective cohort study of 821 ICU patients.
Disclosures: This study was supported by grants from the National Institutes of Health and the Veterans Affairs Clinical Science Research and Development Service, as well as by a Mentored Research Training Grant from the Foundation for Anesthesia Education and Research and the VA Tennessee Valley Geriatric Research Education and Clinical Center. Most authors reported having no conflicts of interest. Dr. Pandharipande and a few others have received grants or other support from industry sources, including Hospira and Orion.
Think Exercise First for Secondary Prevention?
Exercise may be at least as effective as many drug interventions with respect to mortality outcomes for secondary prevention of coronary heart disease.
A "meta-epidemiological" study of 12 drug-related meta-analyses, 4 exercise-related meta-analyses, and 3 recent exercise trials, – including over 339,000 subjects from 305 trials – demonstrated no statistically detectable difference in mortality between exercise and drug interventions for the secondary prevention of coronary heart disease and primary prevention of diabetes and demonstrated a slight increase in the efficacy of physical activity over drug treatment for patients with stroke, judging from findings of a Cochrane review.
In coronary heart disease the risk of dying were reduced with the use of statins (odds ratio, 0.82), beta-blockers (OR, 0.85), angiotensin converting enzyme inhibitors (OR, 0.83), and antiplatelets (OR, 0.83), compared with control, and the credible intervals for these drugs ranged from 0.72 to 0.96. Exercise interventions, however, had similar point estimates but wider credible intervals that extended on both sides of 1.00 (OR, 0.89; 95% confidence interval, 0.76-1.04), Huseyin Naci, a doctoral candidate and research fellow at the London School of Economic and Political Science, and Dr. John P.A. Ioannidis, professor of medicine and director of the prevention research center at Stanford (Calif.) University, reported online Oct. 1 in BMJ.
"When compared head to head in network meta-analyses, all interventions were not different beyond chance: there were no statistically detectable differences among any of the exercise and drug intervention in terms of the effects on mortality outcomes," the investigators said (BMJ 2013 Oct. 1 [doi: 10.1136/bmj.f5577]).
Additionally, with respect to stroke, exercise was significantly more effective than drug interventions were for reducing the risk of dying (odds ratios for exercise vs. anticoagulants, 0.09; and for exercise vs. antiplatelets, 0.10). However, there was considerable uncertainty in these findings, owing to a small number of events in the exercise trials, the investigators noted.
For heart failure, on the other hand, fewer deaths occurred with diuretics and beta-blockers, compared with control (OR, 0.19 and 0.71, respectively), and diuretics were more effective than exercise, angiotensin converting enzyme inhibitors, beta-blockers, and angiotensin receptor blockers (OR, 0.24, 0.21, 0.27, 0.21, respectively). For prediabetes, neither exercise nor drug interventions were clearly effective for reducing mortality.
"In secondary analyses comparing exercise with the drug interventions pooled together, there was no definitive difference between drug and exercise intervention in coronary heart disease, heart failure, and prediabetes, but effect sizes had modestly substantial uncertainty for heart failure and prediabetes," the investigators noted
The study "highlights the near absence of evidence on the comparative effectiveness of exercise and drug interventions on mortality outcomes. Existing evidence on the mortality benefits of physical activity is limited to the secondary prevention of coronary heart disease, rehabilitation after stroke, treatment of heart failure, and prevention of diabetes," they said, adding that the amount of evidence on the mortality benefit of exercise is dwarfed by that of drug interventions, and there is a clear lack of exercise and drug comparisons.
"Albeit this considerable asymmetry in the evidence base, our analysis suggests that exercise potentially had similar effectiveness to drug interventions with two exceptions," they said, noting that exercise appeared more effective for stroke rehabilitation and that diuretics outperformed all comparators for heart failure.
But when the drug interventions were considered together in sensitivity analyses, exercise and drug interventions did not differ in terms of mortality benefit "in all conditions except for stroke rehabilitation, where exercise interventions were associated with a reduction in the odds of mortality more than drug interventions," they said.
The findings highlight a "blind spot" in available scientific evidence resulting from an apparent bias against testing exercise interventions – an observation that highlights "the changing landscape of medical research, which seems to increasingly favor drug interventions over strategies to modify lifestyle," they added, noting that this "lopsided nature of modern medical research" may prohibit detection of the most effective interventions for a given condition if that intervention does not involve a prescription drug.
Nevertheless, the current findings suggest that exercise interventions should be considered as a viable alternative to – or alongside – drug therapy, they said, adding that a more nuanced consideration of the effectiveness of different types of physical activity is warranted.
The findings also point to a need for additional randomized trials on the comparative effectiveness of exercise and drug interventions. Given the scarcity of financial resources to fund such trials, the investigators proposed requiring that pharmaceutical sponsors of new drugs include exercise intervention as an active comparator arm in drug trials.
"In cases where drug options provide only modest benefit, patients deserve to understand the relative impact that physical activity might have on their condition," they concluded.
The investigators reported having no disclosures.
Exercise may be at least as effective as many drug interventions with respect to mortality outcomes for secondary prevention of coronary heart disease.
A "meta-epidemiological" study of 12 drug-related meta-analyses, 4 exercise-related meta-analyses, and 3 recent exercise trials, – including over 339,000 subjects from 305 trials – demonstrated no statistically detectable difference in mortality between exercise and drug interventions for the secondary prevention of coronary heart disease and primary prevention of diabetes and demonstrated a slight increase in the efficacy of physical activity over drug treatment for patients with stroke, judging from findings of a Cochrane review.
In coronary heart disease the risk of dying were reduced with the use of statins (odds ratio, 0.82), beta-blockers (OR, 0.85), angiotensin converting enzyme inhibitors (OR, 0.83), and antiplatelets (OR, 0.83), compared with control, and the credible intervals for these drugs ranged from 0.72 to 0.96. Exercise interventions, however, had similar point estimates but wider credible intervals that extended on both sides of 1.00 (OR, 0.89; 95% confidence interval, 0.76-1.04), Huseyin Naci, a doctoral candidate and research fellow at the London School of Economic and Political Science, and Dr. John P.A. Ioannidis, professor of medicine and director of the prevention research center at Stanford (Calif.) University, reported online Oct. 1 in BMJ.
"When compared head to head in network meta-analyses, all interventions were not different beyond chance: there were no statistically detectable differences among any of the exercise and drug intervention in terms of the effects on mortality outcomes," the investigators said (BMJ 2013 Oct. 1 [doi: 10.1136/bmj.f5577]).
Additionally, with respect to stroke, exercise was significantly more effective than drug interventions were for reducing the risk of dying (odds ratios for exercise vs. anticoagulants, 0.09; and for exercise vs. antiplatelets, 0.10). However, there was considerable uncertainty in these findings, owing to a small number of events in the exercise trials, the investigators noted.
For heart failure, on the other hand, fewer deaths occurred with diuretics and beta-blockers, compared with control (OR, 0.19 and 0.71, respectively), and diuretics were more effective than exercise, angiotensin converting enzyme inhibitors, beta-blockers, and angiotensin receptor blockers (OR, 0.24, 0.21, 0.27, 0.21, respectively). For prediabetes, neither exercise nor drug interventions were clearly effective for reducing mortality.
"In secondary analyses comparing exercise with the drug interventions pooled together, there was no definitive difference between drug and exercise intervention in coronary heart disease, heart failure, and prediabetes, but effect sizes had modestly substantial uncertainty for heart failure and prediabetes," the investigators noted
The study "highlights the near absence of evidence on the comparative effectiveness of exercise and drug interventions on mortality outcomes. Existing evidence on the mortality benefits of physical activity is limited to the secondary prevention of coronary heart disease, rehabilitation after stroke, treatment of heart failure, and prevention of diabetes," they said, adding that the amount of evidence on the mortality benefit of exercise is dwarfed by that of drug interventions, and there is a clear lack of exercise and drug comparisons.
"Albeit this considerable asymmetry in the evidence base, our analysis suggests that exercise potentially had similar effectiveness to drug interventions with two exceptions," they said, noting that exercise appeared more effective for stroke rehabilitation and that diuretics outperformed all comparators for heart failure.
But when the drug interventions were considered together in sensitivity analyses, exercise and drug interventions did not differ in terms of mortality benefit "in all conditions except for stroke rehabilitation, where exercise interventions were associated with a reduction in the odds of mortality more than drug interventions," they said.
The findings highlight a "blind spot" in available scientific evidence resulting from an apparent bias against testing exercise interventions – an observation that highlights "the changing landscape of medical research, which seems to increasingly favor drug interventions over strategies to modify lifestyle," they added, noting that this "lopsided nature of modern medical research" may prohibit detection of the most effective interventions for a given condition if that intervention does not involve a prescription drug.
Nevertheless, the current findings suggest that exercise interventions should be considered as a viable alternative to – or alongside – drug therapy, they said, adding that a more nuanced consideration of the effectiveness of different types of physical activity is warranted.
The findings also point to a need for additional randomized trials on the comparative effectiveness of exercise and drug interventions. Given the scarcity of financial resources to fund such trials, the investigators proposed requiring that pharmaceutical sponsors of new drugs include exercise intervention as an active comparator arm in drug trials.
"In cases where drug options provide only modest benefit, patients deserve to understand the relative impact that physical activity might have on their condition," they concluded.
The investigators reported having no disclosures.
Exercise may be at least as effective as many drug interventions with respect to mortality outcomes for secondary prevention of coronary heart disease.
A "meta-epidemiological" study of 12 drug-related meta-analyses, 4 exercise-related meta-analyses, and 3 recent exercise trials, – including over 339,000 subjects from 305 trials – demonstrated no statistically detectable difference in mortality between exercise and drug interventions for the secondary prevention of coronary heart disease and primary prevention of diabetes and demonstrated a slight increase in the efficacy of physical activity over drug treatment for patients with stroke, judging from findings of a Cochrane review.
In coronary heart disease the risk of dying were reduced with the use of statins (odds ratio, 0.82), beta-blockers (OR, 0.85), angiotensin converting enzyme inhibitors (OR, 0.83), and antiplatelets (OR, 0.83), compared with control, and the credible intervals for these drugs ranged from 0.72 to 0.96. Exercise interventions, however, had similar point estimates but wider credible intervals that extended on both sides of 1.00 (OR, 0.89; 95% confidence interval, 0.76-1.04), Huseyin Naci, a doctoral candidate and research fellow at the London School of Economic and Political Science, and Dr. John P.A. Ioannidis, professor of medicine and director of the prevention research center at Stanford (Calif.) University, reported online Oct. 1 in BMJ.
"When compared head to head in network meta-analyses, all interventions were not different beyond chance: there were no statistically detectable differences among any of the exercise and drug intervention in terms of the effects on mortality outcomes," the investigators said (BMJ 2013 Oct. 1 [doi: 10.1136/bmj.f5577]).
Additionally, with respect to stroke, exercise was significantly more effective than drug interventions were for reducing the risk of dying (odds ratios for exercise vs. anticoagulants, 0.09; and for exercise vs. antiplatelets, 0.10). However, there was considerable uncertainty in these findings, owing to a small number of events in the exercise trials, the investigators noted.
For heart failure, on the other hand, fewer deaths occurred with diuretics and beta-blockers, compared with control (OR, 0.19 and 0.71, respectively), and diuretics were more effective than exercise, angiotensin converting enzyme inhibitors, beta-blockers, and angiotensin receptor blockers (OR, 0.24, 0.21, 0.27, 0.21, respectively). For prediabetes, neither exercise nor drug interventions were clearly effective for reducing mortality.
"In secondary analyses comparing exercise with the drug interventions pooled together, there was no definitive difference between drug and exercise intervention in coronary heart disease, heart failure, and prediabetes, but effect sizes had modestly substantial uncertainty for heart failure and prediabetes," the investigators noted
The study "highlights the near absence of evidence on the comparative effectiveness of exercise and drug interventions on mortality outcomes. Existing evidence on the mortality benefits of physical activity is limited to the secondary prevention of coronary heart disease, rehabilitation after stroke, treatment of heart failure, and prevention of diabetes," they said, adding that the amount of evidence on the mortality benefit of exercise is dwarfed by that of drug interventions, and there is a clear lack of exercise and drug comparisons.
"Albeit this considerable asymmetry in the evidence base, our analysis suggests that exercise potentially had similar effectiveness to drug interventions with two exceptions," they said, noting that exercise appeared more effective for stroke rehabilitation and that diuretics outperformed all comparators for heart failure.
But when the drug interventions were considered together in sensitivity analyses, exercise and drug interventions did not differ in terms of mortality benefit "in all conditions except for stroke rehabilitation, where exercise interventions were associated with a reduction in the odds of mortality more than drug interventions," they said.
The findings highlight a "blind spot" in available scientific evidence resulting from an apparent bias against testing exercise interventions – an observation that highlights "the changing landscape of medical research, which seems to increasingly favor drug interventions over strategies to modify lifestyle," they added, noting that this "lopsided nature of modern medical research" may prohibit detection of the most effective interventions for a given condition if that intervention does not involve a prescription drug.
Nevertheless, the current findings suggest that exercise interventions should be considered as a viable alternative to – or alongside – drug therapy, they said, adding that a more nuanced consideration of the effectiveness of different types of physical activity is warranted.
The findings also point to a need for additional randomized trials on the comparative effectiveness of exercise and drug interventions. Given the scarcity of financial resources to fund such trials, the investigators proposed requiring that pharmaceutical sponsors of new drugs include exercise intervention as an active comparator arm in drug trials.
"In cases where drug options provide only modest benefit, patients deserve to understand the relative impact that physical activity might have on their condition," they concluded.
The investigators reported having no disclosures.
FROM BMJ
Think exercise first for secondary prevention?
Exercise may be at least as effective as many drug interventions with respect to mortality outcomes for secondary prevention of coronary heart disease.
A "meta-epidemiological" study of 12 drug-related meta-analyses, 4 exercise-related meta-analyses, and 3 recent exercise trials, – including over 339,000 subjects from 305 trials – demonstrated no statistically detectable difference in mortality between exercise and drug interventions for the secondary prevention of coronary heart disease and primary prevention of diabetes and demonstrated a slight increase in the efficacy of physical activity over drug treatment for patients with stroke, judging from findings of a Cochrane review.
In coronary heart disease the risk of dying were reduced with the use of statins (odds ratio, 0.82), beta-blockers (OR, 0.85), angiotensin converting enzyme inhibitors (OR, 0.83), and antiplatelets (OR, 0.83), compared with control, and the credible intervals for these drugs ranged from 0.72 to 0.96. Exercise interventions, however, had similar point estimates but wider credible intervals that extended on both sides of 1.00 (OR, 0.89; 95% confidence interval, 0.76-1.04), Huseyin Naci, a doctoral candidate and research fellow at the London School of Economic and Political Science, and Dr. John P.A. Ioannidis, professor of medicine and director of the prevention research center at Stanford (Calif.) University, reported online Oct. 1 in BMJ.
"When compared head to head in network meta-analyses, all interventions were not different beyond chance: there were no statistically detectable differences among any of the exercise and drug intervention in terms of the effects on mortality outcomes," the investigators said (BMJ 2013 Oct. 1 [doi: 10.1136/bmj.f5577]).
Additionally, with respect to stroke, exercise was significantly more effective than drug interventions were for reducing the risk of dying (odds ratios for exercise vs. anticoagulants, 0.09; and for exercise vs. antiplatelets, 0.10). However, there was considerable uncertainty in these findings, owing to a small number of events in the exercise trials, the investigators noted.
For heart failure, on the other hand, fewer deaths occurred with diuretics and beta-blockers, compared with control (OR, 0.19 and 0.71, respectively), and diuretics were more effective than exercise, angiotensin converting enzyme inhibitors, beta-blockers, and angiotensin receptor blockers (OR, 0.24, 0.21, 0.27, 0.21, respectively). For prediabetes, neither exercise nor drug interventions were clearly effective for reducing mortality.
"In secondary analyses comparing exercise with the drug interventions pooled together, there was no definitive difference between drug and exercise intervention in coronary heart disease, heart failure, and prediabetes, but effect sizes had modestly substantial uncertainty for heart failure and prediabetes," the investigators noted
The study "highlights the near absence of evidence on the comparative effectiveness of exercise and drug interventions on mortality outcomes. Existing evidence on the mortality benefits of physical activity is limited to the secondary prevention of coronary heart disease, rehabilitation after stroke, treatment of heart failure, and prevention of diabetes," they said, adding that the amount of evidence on the mortality benefit of exercise is dwarfed by that of drug interventions, and there is a clear lack of exercise and drug comparisons.
"Albeit this considerable asymmetry in the evidence base, our analysis suggests that exercise potentially had similar effectiveness to drug interventions with two exceptions," they said, noting that exercise appeared more effective for stroke rehabilitation and that diuretics outperformed all comparators for heart failure.
But when the drug interventions were considered together in sensitivity analyses, exercise and drug interventions did not differ in terms of mortality benefit "in all conditions except for stroke rehabilitation, where exercise interventions were associated with a reduction in the odds of mortality more than drug interventions," they said.
The findings highlight a "blind spot" in available scientific evidence resulting from an apparent bias against testing exercise interventions – an observation that highlights "the changing landscape of medical research, which seems to increasingly favor drug interventions over strategies to modify lifestyle," they added, noting that this "lopsided nature of modern medical research" may prohibit detection of the most effective interventions for a given condition if that intervention does not involve a prescription drug.
Nevertheless, the current findings suggest that exercise interventions should be considered as a viable alternative to – or alongside – drug therapy, they said, adding that a more nuanced consideration of the effectiveness of different types of physical activity is warranted.
The findings also point to a need for additional randomized trials on the comparative effectiveness of exercise and drug interventions. Given the scarcity of financial resources to fund such trials, the investigators proposed requiring that pharmaceutical sponsors of new drugs include exercise intervention as an active comparator arm in drug trials.
"In cases where drug options provide only modest benefit, patients deserve to understand the relative impact that physical activity might have on their condition," they concluded.
The investigators reported having no disclosures.
Exercise may be at least as effective as many drug interventions with respect to mortality outcomes for secondary prevention of coronary heart disease.
A "meta-epidemiological" study of 12 drug-related meta-analyses, 4 exercise-related meta-analyses, and 3 recent exercise trials, – including over 339,000 subjects from 305 trials – demonstrated no statistically detectable difference in mortality between exercise and drug interventions for the secondary prevention of coronary heart disease and primary prevention of diabetes and demonstrated a slight increase in the efficacy of physical activity over drug treatment for patients with stroke, judging from findings of a Cochrane review.
In coronary heart disease the risk of dying were reduced with the use of statins (odds ratio, 0.82), beta-blockers (OR, 0.85), angiotensin converting enzyme inhibitors (OR, 0.83), and antiplatelets (OR, 0.83), compared with control, and the credible intervals for these drugs ranged from 0.72 to 0.96. Exercise interventions, however, had similar point estimates but wider credible intervals that extended on both sides of 1.00 (OR, 0.89; 95% confidence interval, 0.76-1.04), Huseyin Naci, a doctoral candidate and research fellow at the London School of Economic and Political Science, and Dr. John P.A. Ioannidis, professor of medicine and director of the prevention research center at Stanford (Calif.) University, reported online Oct. 1 in BMJ.
"When compared head to head in network meta-analyses, all interventions were not different beyond chance: there were no statistically detectable differences among any of the exercise and drug intervention in terms of the effects on mortality outcomes," the investigators said (BMJ 2013 Oct. 1 [doi: 10.1136/bmj.f5577]).
Additionally, with respect to stroke, exercise was significantly more effective than drug interventions were for reducing the risk of dying (odds ratios for exercise vs. anticoagulants, 0.09; and for exercise vs. antiplatelets, 0.10). However, there was considerable uncertainty in these findings, owing to a small number of events in the exercise trials, the investigators noted.
For heart failure, on the other hand, fewer deaths occurred with diuretics and beta-blockers, compared with control (OR, 0.19 and 0.71, respectively), and diuretics were more effective than exercise, angiotensin converting enzyme inhibitors, beta-blockers, and angiotensin receptor blockers (OR, 0.24, 0.21, 0.27, 0.21, respectively). For prediabetes, neither exercise nor drug interventions were clearly effective for reducing mortality.
"In secondary analyses comparing exercise with the drug interventions pooled together, there was no definitive difference between drug and exercise intervention in coronary heart disease, heart failure, and prediabetes, but effect sizes had modestly substantial uncertainty for heart failure and prediabetes," the investigators noted
The study "highlights the near absence of evidence on the comparative effectiveness of exercise and drug interventions on mortality outcomes. Existing evidence on the mortality benefits of physical activity is limited to the secondary prevention of coronary heart disease, rehabilitation after stroke, treatment of heart failure, and prevention of diabetes," they said, adding that the amount of evidence on the mortality benefit of exercise is dwarfed by that of drug interventions, and there is a clear lack of exercise and drug comparisons.
"Albeit this considerable asymmetry in the evidence base, our analysis suggests that exercise potentially had similar effectiveness to drug interventions with two exceptions," they said, noting that exercise appeared more effective for stroke rehabilitation and that diuretics outperformed all comparators for heart failure.
But when the drug interventions were considered together in sensitivity analyses, exercise and drug interventions did not differ in terms of mortality benefit "in all conditions except for stroke rehabilitation, where exercise interventions were associated with a reduction in the odds of mortality more than drug interventions," they said.
The findings highlight a "blind spot" in available scientific evidence resulting from an apparent bias against testing exercise interventions – an observation that highlights "the changing landscape of medical research, which seems to increasingly favor drug interventions over strategies to modify lifestyle," they added, noting that this "lopsided nature of modern medical research" may prohibit detection of the most effective interventions for a given condition if that intervention does not involve a prescription drug.
Nevertheless, the current findings suggest that exercise interventions should be considered as a viable alternative to – or alongside – drug therapy, they said, adding that a more nuanced consideration of the effectiveness of different types of physical activity is warranted.
The findings also point to a need for additional randomized trials on the comparative effectiveness of exercise and drug interventions. Given the scarcity of financial resources to fund such trials, the investigators proposed requiring that pharmaceutical sponsors of new drugs include exercise intervention as an active comparator arm in drug trials.
"In cases where drug options provide only modest benefit, patients deserve to understand the relative impact that physical activity might have on their condition," they concluded.
The investigators reported having no disclosures.
Exercise may be at least as effective as many drug interventions with respect to mortality outcomes for secondary prevention of coronary heart disease.
A "meta-epidemiological" study of 12 drug-related meta-analyses, 4 exercise-related meta-analyses, and 3 recent exercise trials, – including over 339,000 subjects from 305 trials – demonstrated no statistically detectable difference in mortality between exercise and drug interventions for the secondary prevention of coronary heart disease and primary prevention of diabetes and demonstrated a slight increase in the efficacy of physical activity over drug treatment for patients with stroke, judging from findings of a Cochrane review.
In coronary heart disease the risk of dying were reduced with the use of statins (odds ratio, 0.82), beta-blockers (OR, 0.85), angiotensin converting enzyme inhibitors (OR, 0.83), and antiplatelets (OR, 0.83), compared with control, and the credible intervals for these drugs ranged from 0.72 to 0.96. Exercise interventions, however, had similar point estimates but wider credible intervals that extended on both sides of 1.00 (OR, 0.89; 95% confidence interval, 0.76-1.04), Huseyin Naci, a doctoral candidate and research fellow at the London School of Economic and Political Science, and Dr. John P.A. Ioannidis, professor of medicine and director of the prevention research center at Stanford (Calif.) University, reported online Oct. 1 in BMJ.
"When compared head to head in network meta-analyses, all interventions were not different beyond chance: there were no statistically detectable differences among any of the exercise and drug intervention in terms of the effects on mortality outcomes," the investigators said (BMJ 2013 Oct. 1 [doi: 10.1136/bmj.f5577]).
Additionally, with respect to stroke, exercise was significantly more effective than drug interventions were for reducing the risk of dying (odds ratios for exercise vs. anticoagulants, 0.09; and for exercise vs. antiplatelets, 0.10). However, there was considerable uncertainty in these findings, owing to a small number of events in the exercise trials, the investigators noted.
For heart failure, on the other hand, fewer deaths occurred with diuretics and beta-blockers, compared with control (OR, 0.19 and 0.71, respectively), and diuretics were more effective than exercise, angiotensin converting enzyme inhibitors, beta-blockers, and angiotensin receptor blockers (OR, 0.24, 0.21, 0.27, 0.21, respectively). For prediabetes, neither exercise nor drug interventions were clearly effective for reducing mortality.
"In secondary analyses comparing exercise with the drug interventions pooled together, there was no definitive difference between drug and exercise intervention in coronary heart disease, heart failure, and prediabetes, but effect sizes had modestly substantial uncertainty for heart failure and prediabetes," the investigators noted
The study "highlights the near absence of evidence on the comparative effectiveness of exercise and drug interventions on mortality outcomes. Existing evidence on the mortality benefits of physical activity is limited to the secondary prevention of coronary heart disease, rehabilitation after stroke, treatment of heart failure, and prevention of diabetes," they said, adding that the amount of evidence on the mortality benefit of exercise is dwarfed by that of drug interventions, and there is a clear lack of exercise and drug comparisons.
"Albeit this considerable asymmetry in the evidence base, our analysis suggests that exercise potentially had similar effectiveness to drug interventions with two exceptions," they said, noting that exercise appeared more effective for stroke rehabilitation and that diuretics outperformed all comparators for heart failure.
But when the drug interventions were considered together in sensitivity analyses, exercise and drug interventions did not differ in terms of mortality benefit "in all conditions except for stroke rehabilitation, where exercise interventions were associated with a reduction in the odds of mortality more than drug interventions," they said.
The findings highlight a "blind spot" in available scientific evidence resulting from an apparent bias against testing exercise interventions – an observation that highlights "the changing landscape of medical research, which seems to increasingly favor drug interventions over strategies to modify lifestyle," they added, noting that this "lopsided nature of modern medical research" may prohibit detection of the most effective interventions for a given condition if that intervention does not involve a prescription drug.
Nevertheless, the current findings suggest that exercise interventions should be considered as a viable alternative to – or alongside – drug therapy, they said, adding that a more nuanced consideration of the effectiveness of different types of physical activity is warranted.
The findings also point to a need for additional randomized trials on the comparative effectiveness of exercise and drug interventions. Given the scarcity of financial resources to fund such trials, the investigators proposed requiring that pharmaceutical sponsors of new drugs include exercise intervention as an active comparator arm in drug trials.
"In cases where drug options provide only modest benefit, patients deserve to understand the relative impact that physical activity might have on their condition," they concluded.
The investigators reported having no disclosures.
FROM BMJ
Major finding: No statistically detectable difference was seen in mortality outcomes between exercise and drug interventions for the secondary prevention of coronary heart disease and pre-diabetes; a slight increase was seen in the efficacy of physical activity over drug treatment for patients with stroke.
Data source: A meta-epidemiological study of meta-analyses and studies involving nearly 340,000 subjects.
Disclosures: The investigators reported having no disclosures.
Most physicians don’t bring up infant bed sharing
While black and Hispanic race were associated with an increase in infant bed sharing between 1993 and 2010, those whose physicians did not agree with the practice were less likely to engage in bed sharing, according to findings from the National Infant Sleep Position study.
Of 18,986 participants who took part in the annual telephone survey, 11.2% reported infant bed sharing as a usual practice, with the percentage increasing from 6.5% in 1993 to 13.5% in 2010, Dr. Eve R. Colson of Yale University, New Haven, Conn., and her colleagues reported Sept. 30 in JAMA Pediatrics.
Based on findings from 2006-2010, when questions about physician communication were added to the survey, less than half (46%) of participants reported talking with a physician about bed sharing; and most (72.6%) said their physician was against bed sharing, while 21.3% reported that their physician was neutral and 6.1% reported their physician was supportive of the practice.
Survey respondents with a physician who had a negative attitude about bed sharing were less likely to engage in the practice (odds ratio, 0.66), while a neutral attitude on the part of the physician was associated with increased bed sharing (OR, 1.38), the investigators noted. No significant association was seen between positive physician attitude toward the practice and bed sharing.
This finding could have particular implications for changing behavior, as the results suggest there needs to be more consistent physician advice in line with AAP recommendations – infants should share a room with parents without sharing a bed when sleeping – might reduce bed sharing, the investigators noted.
Among white respondents, the percentage reporting bed sharing increased significantly from 1993 to 2000, but not from 2001 to 2010. For black and Hispanic respondents, the percentage increased progressively across the study period.
After the researchers adjusted for changes in bed sharing over time, black and Hispanic maternal race was significantly associated with bed sharing (OR, 3.47 and 1.33, respectively), as was "other" maternal race (OR, 2.46), the investigators reported (JAMA Pediatr. 2013 Sept. 30 [doi:10.1001/jamapediatrics.2013.2560]).
Significant associations were seen between bed sharing and maternal educational level less than high school, compared with college or greater (OR, 1.42); household income of less than $20,000 and $20,000-$50,000, compared with income greater than $50,000 (OR, 1.69 and 1.29, respectively); living in the West, South, or mid-Atlantic, compared with the Midwest (OR, 1.61, 1.47, and 0.77, respectively); infant age younger than 8 weeks and infant age 8-15 weeks, compared with age 16 weeks or older (OR, 1.45 and 1.31, respectively); and preterm birth, compared with full-term birth (OR, 1.41).
The National Infant Sleep Position study is a cross-sectional study that tracks infant care practices through annual telephone surveys. About 1,000 nighttime caregivers of infants born within 7 months of the survey administration participated; the respondent was the infant’s mother in 84% of cases.
The findings are important because of the strong association between infant bed sharing, sudden infant death syndrome, and unintentional sleep-related infant death. While quilt and comforter use – another risk factor for sleep-related infant death declined over time, it is also strongly associated with bed sharing.
Though limited by a number of factors – such as survey responses may not reflect actual practice; there was a declining response rate in the later years of the study; and the sample was not nationally representative when compared with nationally collected vital statistics – the findings provide valuable information about trends associated with bed sharing, the investigators noted. "The data may be useful in evaluating the impact of any broad intervention to change behavior."
This study was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development. The authors reported having no disclosures.
The report by Dr. Colson and her colleagues, which calls for increased efforts by pediatricians to discourage bed sharing, is "disquieting" because it ignores both the potential benefits of the practice and the lack of evidence linking bed sharing per se to an increased risk of infant death, according to Dr. Abraham B. Bergman.
The studies used to support the AAP’s position against bed sharing share a common flaw – nonuniform and unverifiable information on the causes of death, Dr. Bergman wrote in an editorial.
"When condemning a widespread cultural practice, the supporting data should be clear," he wrote.
In fact, questions have been raised about whether the decline in the rate of sudden infant death between 1994 and 2004 reflects a true decline or a change in certifier practices (with medical examiners moving away from classifying deaths as SIDS and moving towards classifying the deaths as accidental suffocation or unknown causes). This question is particularly pertinent beacuse the overall postneonatal mortality rate did not decline during that period.
Given the lack of convincing data supporting its position, the AAP has overreached.
"Equal time in counseling should be given to the benefits to bed sharing, such as more sleep for the parent, easier breast-feeding when the infant is nearby, ease of pacifier reinsertion, and the intangible satisfaction of skin-to-skin contact," he wrote.
Dr. Bergman is a physician in the department of pediatrics at Harborview Medical Center in Seattle. He reported having no disclosures.
The report by Dr. Colson and her colleagues, which calls for increased efforts by pediatricians to discourage bed sharing, is "disquieting" because it ignores both the potential benefits of the practice and the lack of evidence linking bed sharing per se to an increased risk of infant death, according to Dr. Abraham B. Bergman.
The studies used to support the AAP’s position against bed sharing share a common flaw – nonuniform and unverifiable information on the causes of death, Dr. Bergman wrote in an editorial.
"When condemning a widespread cultural practice, the supporting data should be clear," he wrote.
In fact, questions have been raised about whether the decline in the rate of sudden infant death between 1994 and 2004 reflects a true decline or a change in certifier practices (with medical examiners moving away from classifying deaths as SIDS and moving towards classifying the deaths as accidental suffocation or unknown causes). This question is particularly pertinent beacuse the overall postneonatal mortality rate did not decline during that period.
Given the lack of convincing data supporting its position, the AAP has overreached.
"Equal time in counseling should be given to the benefits to bed sharing, such as more sleep for the parent, easier breast-feeding when the infant is nearby, ease of pacifier reinsertion, and the intangible satisfaction of skin-to-skin contact," he wrote.
Dr. Bergman is a physician in the department of pediatrics at Harborview Medical Center in Seattle. He reported having no disclosures.
The report by Dr. Colson and her colleagues, which calls for increased efforts by pediatricians to discourage bed sharing, is "disquieting" because it ignores both the potential benefits of the practice and the lack of evidence linking bed sharing per se to an increased risk of infant death, according to Dr. Abraham B. Bergman.
The studies used to support the AAP’s position against bed sharing share a common flaw – nonuniform and unverifiable information on the causes of death, Dr. Bergman wrote in an editorial.
"When condemning a widespread cultural practice, the supporting data should be clear," he wrote.
In fact, questions have been raised about whether the decline in the rate of sudden infant death between 1994 and 2004 reflects a true decline or a change in certifier practices (with medical examiners moving away from classifying deaths as SIDS and moving towards classifying the deaths as accidental suffocation or unknown causes). This question is particularly pertinent beacuse the overall postneonatal mortality rate did not decline during that period.
Given the lack of convincing data supporting its position, the AAP has overreached.
"Equal time in counseling should be given to the benefits to bed sharing, such as more sleep for the parent, easier breast-feeding when the infant is nearby, ease of pacifier reinsertion, and the intangible satisfaction of skin-to-skin contact," he wrote.
Dr. Bergman is a physician in the department of pediatrics at Harborview Medical Center in Seattle. He reported having no disclosures.
While black and Hispanic race were associated with an increase in infant bed sharing between 1993 and 2010, those whose physicians did not agree with the practice were less likely to engage in bed sharing, according to findings from the National Infant Sleep Position study.
Of 18,986 participants who took part in the annual telephone survey, 11.2% reported infant bed sharing as a usual practice, with the percentage increasing from 6.5% in 1993 to 13.5% in 2010, Dr. Eve R. Colson of Yale University, New Haven, Conn., and her colleagues reported Sept. 30 in JAMA Pediatrics.
Based on findings from 2006-2010, when questions about physician communication were added to the survey, less than half (46%) of participants reported talking with a physician about bed sharing; and most (72.6%) said their physician was against bed sharing, while 21.3% reported that their physician was neutral and 6.1% reported their physician was supportive of the practice.
Survey respondents with a physician who had a negative attitude about bed sharing were less likely to engage in the practice (odds ratio, 0.66), while a neutral attitude on the part of the physician was associated with increased bed sharing (OR, 1.38), the investigators noted. No significant association was seen between positive physician attitude toward the practice and bed sharing.
This finding could have particular implications for changing behavior, as the results suggest there needs to be more consistent physician advice in line with AAP recommendations – infants should share a room with parents without sharing a bed when sleeping – might reduce bed sharing, the investigators noted.
Among white respondents, the percentage reporting bed sharing increased significantly from 1993 to 2000, but not from 2001 to 2010. For black and Hispanic respondents, the percentage increased progressively across the study period.
After the researchers adjusted for changes in bed sharing over time, black and Hispanic maternal race was significantly associated with bed sharing (OR, 3.47 and 1.33, respectively), as was "other" maternal race (OR, 2.46), the investigators reported (JAMA Pediatr. 2013 Sept. 30 [doi:10.1001/jamapediatrics.2013.2560]).
Significant associations were seen between bed sharing and maternal educational level less than high school, compared with college or greater (OR, 1.42); household income of less than $20,000 and $20,000-$50,000, compared with income greater than $50,000 (OR, 1.69 and 1.29, respectively); living in the West, South, or mid-Atlantic, compared with the Midwest (OR, 1.61, 1.47, and 0.77, respectively); infant age younger than 8 weeks and infant age 8-15 weeks, compared with age 16 weeks or older (OR, 1.45 and 1.31, respectively); and preterm birth, compared with full-term birth (OR, 1.41).
The National Infant Sleep Position study is a cross-sectional study that tracks infant care practices through annual telephone surveys. About 1,000 nighttime caregivers of infants born within 7 months of the survey administration participated; the respondent was the infant’s mother in 84% of cases.
The findings are important because of the strong association between infant bed sharing, sudden infant death syndrome, and unintentional sleep-related infant death. While quilt and comforter use – another risk factor for sleep-related infant death declined over time, it is also strongly associated with bed sharing.
Though limited by a number of factors – such as survey responses may not reflect actual practice; there was a declining response rate in the later years of the study; and the sample was not nationally representative when compared with nationally collected vital statistics – the findings provide valuable information about trends associated with bed sharing, the investigators noted. "The data may be useful in evaluating the impact of any broad intervention to change behavior."
This study was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development. The authors reported having no disclosures.
While black and Hispanic race were associated with an increase in infant bed sharing between 1993 and 2010, those whose physicians did not agree with the practice were less likely to engage in bed sharing, according to findings from the National Infant Sleep Position study.
Of 18,986 participants who took part in the annual telephone survey, 11.2% reported infant bed sharing as a usual practice, with the percentage increasing from 6.5% in 1993 to 13.5% in 2010, Dr. Eve R. Colson of Yale University, New Haven, Conn., and her colleagues reported Sept. 30 in JAMA Pediatrics.
Based on findings from 2006-2010, when questions about physician communication were added to the survey, less than half (46%) of participants reported talking with a physician about bed sharing; and most (72.6%) said their physician was against bed sharing, while 21.3% reported that their physician was neutral and 6.1% reported their physician was supportive of the practice.
Survey respondents with a physician who had a negative attitude about bed sharing were less likely to engage in the practice (odds ratio, 0.66), while a neutral attitude on the part of the physician was associated with increased bed sharing (OR, 1.38), the investigators noted. No significant association was seen between positive physician attitude toward the practice and bed sharing.
This finding could have particular implications for changing behavior, as the results suggest there needs to be more consistent physician advice in line with AAP recommendations – infants should share a room with parents without sharing a bed when sleeping – might reduce bed sharing, the investigators noted.
Among white respondents, the percentage reporting bed sharing increased significantly from 1993 to 2000, but not from 2001 to 2010. For black and Hispanic respondents, the percentage increased progressively across the study period.
After the researchers adjusted for changes in bed sharing over time, black and Hispanic maternal race was significantly associated with bed sharing (OR, 3.47 and 1.33, respectively), as was "other" maternal race (OR, 2.46), the investigators reported (JAMA Pediatr. 2013 Sept. 30 [doi:10.1001/jamapediatrics.2013.2560]).
Significant associations were seen between bed sharing and maternal educational level less than high school, compared with college or greater (OR, 1.42); household income of less than $20,000 and $20,000-$50,000, compared with income greater than $50,000 (OR, 1.69 and 1.29, respectively); living in the West, South, or mid-Atlantic, compared with the Midwest (OR, 1.61, 1.47, and 0.77, respectively); infant age younger than 8 weeks and infant age 8-15 weeks, compared with age 16 weeks or older (OR, 1.45 and 1.31, respectively); and preterm birth, compared with full-term birth (OR, 1.41).
The National Infant Sleep Position study is a cross-sectional study that tracks infant care practices through annual telephone surveys. About 1,000 nighttime caregivers of infants born within 7 months of the survey administration participated; the respondent was the infant’s mother in 84% of cases.
The findings are important because of the strong association between infant bed sharing, sudden infant death syndrome, and unintentional sleep-related infant death. While quilt and comforter use – another risk factor for sleep-related infant death declined over time, it is also strongly associated with bed sharing.
Though limited by a number of factors – such as survey responses may not reflect actual practice; there was a declining response rate in the later years of the study; and the sample was not nationally representative when compared with nationally collected vital statistics – the findings provide valuable information about trends associated with bed sharing, the investigators noted. "The data may be useful in evaluating the impact of any broad intervention to change behavior."
This study was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development. The authors reported having no disclosures.
FROM JAMA PEDIATRICS
Major finding: The percentage of survey respondents reporting infant bed sharing as a usual practice increased from 6.5% in 1993 to 13.5% in 2010.
Data source: The National Infant Sleep Position study, a telephone survey involving 18,986 participants.
Disclosures: This study was supported by a grant from the Eunice Kennedy Shriver National Institute of Child Health and Human Development. The authors reported having no disclosures.
Lupus treatment: Get back to basics
LAS VEGAS – The future looks bright for lupus treatment given the extensive list of drugs and targets currently undergoing evaluation, but for now treatment remains largely an art, according to Dr. Richard Furie.
"We try to integrate science, but it really is an art – these patients are very difficult," he said at the meeting.
That’s because of the heterogeneity of the disease, and the need to balance the benefits of treatment against the potential harms, which are extensive with currently available drugs, he explained.
Success is best achieved by "getting back to basics," he added.
First, as simple as it sounds, "do good" by controlling disease activity, preventing damage from disease, and preventing flares – as even one flare can cause damage – and "do no harm," which requires preventing damage from treatment and avoiding the treatment of damage with agents intended for active disease, he said.
"This is difficult, but the last thing we want to do is bombard a patient with potentially toxic medication when they are not going to reverse damage. ... We have a lot of potentially harmful medications. ... The name of the game is to minimize damage over time," said Dr. Furie, chief of the division of rheumatology at North Shore–LIJ Health System and professor of medicine at Hofstra North Shore–LIJ School of Medicine in Hempstead, N.Y.
Dr. Furie outlined his principles for treatment design:
• Identify disease manifestations.
• Distinguish activity from chronicity. For example, it is important to determine if a rash represents active disease or a scar, and whether proteinuria represents active nephritis or previous damage.
• Prioritize active disease manifestations.
• View disease activity as a continuum.
• Treat with the least toxic medicine and the lowest dose needed to address the most concerning disease manifestation. This will hopefully bring other manifestations under control as well, but treating too aggressively can lead to unnecessary damage, he said.
As for currently available treatments, "we’ve come a long way and patients are doing better," he said.
But the number of medications remains limited, and a great deal more work must be done, particularly with respect to lupus nephritis.
"At best, we do a good job with treatment in about 30%-40% of [patients with lupus nephritis], so in more than half we’re not doing well," he said.
More efficacious therapies are also needed for severe extrarenal lupus, flare prevention, and remission induction, he said, noting that treatments that are safer than steroids and cyclophosphamide also are needed.
Among currently used treatments, rituximab, while promising in open-label studies and off-label experience, has had disappointing results in randomized, controlled clinical trials. It nonetheless remains "in the toolbox," Dr. Furie said, noting that interest remains high in studying the agent for nephritis.
Mycophenolate mofetil also has been disappointing, in that it failed to show superiority to cyclophosphamide in the Aspreva Lupus Management Study (ALMS). However, Dr. Furie classified this drug as a "successful failure," because ALMS demonstrated what many had already believed – that mycophenolate mofetil is a good drug for lupus nephritis (J. Am. Soc. Nephrol. 2009;20:1103-12) .
Another agent, belimumab, on the other hand, represents a success story – and a history-making success story at that, he said.
Belimumab was the first drug approved for SLE via a randomized controlled trial, he explained, noting that it has favorable effects on steroid tapering to under 7.5 mg/day, 36-Item Short Form Health Survey (SF-36) fatigue scores, flare rates, and DNA antibody and complement levels – all with a favorable safety profile.
Unfortunately, the failures outweigh the successes with respect to the "humbling process" of drug development, but many drugs and targets are currently being investigated, he said.
In fact, there are more studies than there are patients, he said, adding, "but it still strikes me how many companies want to go forward in lupus, and I think it’s great for the field."
"There are a lot of interesting drugs on the horizon. ... We will someday have biomarkers, personalized therapy, and better outcomes," he said at the meeting, held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Furie reported receiving research support and/or serving as a consultant or investigator for several companies, including AstraZeneca, Biogen Idec, Bristol-Myers Squibb, Boehringer Ingelheim, Eli Lilly, GlaxoSmithKline, Merck, and Novartis. He also has received research support from the National Institute of Allergy and Infectious Diseases.
LAS VEGAS – The future looks bright for lupus treatment given the extensive list of drugs and targets currently undergoing evaluation, but for now treatment remains largely an art, according to Dr. Richard Furie.
"We try to integrate science, but it really is an art – these patients are very difficult," he said at the meeting.
That’s because of the heterogeneity of the disease, and the need to balance the benefits of treatment against the potential harms, which are extensive with currently available drugs, he explained.
Success is best achieved by "getting back to basics," he added.
First, as simple as it sounds, "do good" by controlling disease activity, preventing damage from disease, and preventing flares – as even one flare can cause damage – and "do no harm," which requires preventing damage from treatment and avoiding the treatment of damage with agents intended for active disease, he said.
"This is difficult, but the last thing we want to do is bombard a patient with potentially toxic medication when they are not going to reverse damage. ... We have a lot of potentially harmful medications. ... The name of the game is to minimize damage over time," said Dr. Furie, chief of the division of rheumatology at North Shore–LIJ Health System and professor of medicine at Hofstra North Shore–LIJ School of Medicine in Hempstead, N.Y.
Dr. Furie outlined his principles for treatment design:
• Identify disease manifestations.
• Distinguish activity from chronicity. For example, it is important to determine if a rash represents active disease or a scar, and whether proteinuria represents active nephritis or previous damage.
• Prioritize active disease manifestations.
• View disease activity as a continuum.
• Treat with the least toxic medicine and the lowest dose needed to address the most concerning disease manifestation. This will hopefully bring other manifestations under control as well, but treating too aggressively can lead to unnecessary damage, he said.
As for currently available treatments, "we’ve come a long way and patients are doing better," he said.
But the number of medications remains limited, and a great deal more work must be done, particularly with respect to lupus nephritis.
"At best, we do a good job with treatment in about 30%-40% of [patients with lupus nephritis], so in more than half we’re not doing well," he said.
More efficacious therapies are also needed for severe extrarenal lupus, flare prevention, and remission induction, he said, noting that treatments that are safer than steroids and cyclophosphamide also are needed.
Among currently used treatments, rituximab, while promising in open-label studies and off-label experience, has had disappointing results in randomized, controlled clinical trials. It nonetheless remains "in the toolbox," Dr. Furie said, noting that interest remains high in studying the agent for nephritis.
Mycophenolate mofetil also has been disappointing, in that it failed to show superiority to cyclophosphamide in the Aspreva Lupus Management Study (ALMS). However, Dr. Furie classified this drug as a "successful failure," because ALMS demonstrated what many had already believed – that mycophenolate mofetil is a good drug for lupus nephritis (J. Am. Soc. Nephrol. 2009;20:1103-12) .
Another agent, belimumab, on the other hand, represents a success story – and a history-making success story at that, he said.
Belimumab was the first drug approved for SLE via a randomized controlled trial, he explained, noting that it has favorable effects on steroid tapering to under 7.5 mg/day, 36-Item Short Form Health Survey (SF-36) fatigue scores, flare rates, and DNA antibody and complement levels – all with a favorable safety profile.
Unfortunately, the failures outweigh the successes with respect to the "humbling process" of drug development, but many drugs and targets are currently being investigated, he said.
In fact, there are more studies than there are patients, he said, adding, "but it still strikes me how many companies want to go forward in lupus, and I think it’s great for the field."
"There are a lot of interesting drugs on the horizon. ... We will someday have biomarkers, personalized therapy, and better outcomes," he said at the meeting, held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Furie reported receiving research support and/or serving as a consultant or investigator for several companies, including AstraZeneca, Biogen Idec, Bristol-Myers Squibb, Boehringer Ingelheim, Eli Lilly, GlaxoSmithKline, Merck, and Novartis. He also has received research support from the National Institute of Allergy and Infectious Diseases.
LAS VEGAS – The future looks bright for lupus treatment given the extensive list of drugs and targets currently undergoing evaluation, but for now treatment remains largely an art, according to Dr. Richard Furie.
"We try to integrate science, but it really is an art – these patients are very difficult," he said at the meeting.
That’s because of the heterogeneity of the disease, and the need to balance the benefits of treatment against the potential harms, which are extensive with currently available drugs, he explained.
Success is best achieved by "getting back to basics," he added.
First, as simple as it sounds, "do good" by controlling disease activity, preventing damage from disease, and preventing flares – as even one flare can cause damage – and "do no harm," which requires preventing damage from treatment and avoiding the treatment of damage with agents intended for active disease, he said.
"This is difficult, but the last thing we want to do is bombard a patient with potentially toxic medication when they are not going to reverse damage. ... We have a lot of potentially harmful medications. ... The name of the game is to minimize damage over time," said Dr. Furie, chief of the division of rheumatology at North Shore–LIJ Health System and professor of medicine at Hofstra North Shore–LIJ School of Medicine in Hempstead, N.Y.
Dr. Furie outlined his principles for treatment design:
• Identify disease manifestations.
• Distinguish activity from chronicity. For example, it is important to determine if a rash represents active disease or a scar, and whether proteinuria represents active nephritis or previous damage.
• Prioritize active disease manifestations.
• View disease activity as a continuum.
• Treat with the least toxic medicine and the lowest dose needed to address the most concerning disease manifestation. This will hopefully bring other manifestations under control as well, but treating too aggressively can lead to unnecessary damage, he said.
As for currently available treatments, "we’ve come a long way and patients are doing better," he said.
But the number of medications remains limited, and a great deal more work must be done, particularly with respect to lupus nephritis.
"At best, we do a good job with treatment in about 30%-40% of [patients with lupus nephritis], so in more than half we’re not doing well," he said.
More efficacious therapies are also needed for severe extrarenal lupus, flare prevention, and remission induction, he said, noting that treatments that are safer than steroids and cyclophosphamide also are needed.
Among currently used treatments, rituximab, while promising in open-label studies and off-label experience, has had disappointing results in randomized, controlled clinical trials. It nonetheless remains "in the toolbox," Dr. Furie said, noting that interest remains high in studying the agent for nephritis.
Mycophenolate mofetil also has been disappointing, in that it failed to show superiority to cyclophosphamide in the Aspreva Lupus Management Study (ALMS). However, Dr. Furie classified this drug as a "successful failure," because ALMS demonstrated what many had already believed – that mycophenolate mofetil is a good drug for lupus nephritis (J. Am. Soc. Nephrol. 2009;20:1103-12) .
Another agent, belimumab, on the other hand, represents a success story – and a history-making success story at that, he said.
Belimumab was the first drug approved for SLE via a randomized controlled trial, he explained, noting that it has favorable effects on steroid tapering to under 7.5 mg/day, 36-Item Short Form Health Survey (SF-36) fatigue scores, flare rates, and DNA antibody and complement levels – all with a favorable safety profile.
Unfortunately, the failures outweigh the successes with respect to the "humbling process" of drug development, but many drugs and targets are currently being investigated, he said.
In fact, there are more studies than there are patients, he said, adding, "but it still strikes me how many companies want to go forward in lupus, and I think it’s great for the field."
"There are a lot of interesting drugs on the horizon. ... We will someday have biomarkers, personalized therapy, and better outcomes," he said at the meeting, held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
Dr. Furie reported receiving research support and/or serving as a consultant or investigator for several companies, including AstraZeneca, Biogen Idec, Bristol-Myers Squibb, Boehringer Ingelheim, Eli Lilly, GlaxoSmithKline, Merck, and Novartis. He also has received research support from the National Institute of Allergy and Infectious Diseases.
EXPERT ANALYSIS FROM PERSPECTIVES IN RHEUMATIC DISEASES 2013
Legislative update: Initiatives with implications for rheumatologists
LAS VEGAS – A coalition working to address dual-energy X-ray absorptiometry access issues is developing draft legislation that would establish a geographically adjusted national minimum payment for DXA, according to Dr. Michael Schweitz.
Under the draft legislation – a response to a series of reimbursement cuts over the past several years – DXA CPT codes 77080 and 77082 would be paid at $98 and $35, respectively, and a bundled code or bundled payment for bone density and vertebral fracture assessment studies would result in a national minimum payment of $133, Dr. Schweitz, president of the Coalition of State Rheumatology Organizations, said at Perspectives in Rheumatic Diseases 2013 during an update on federal legislative and regulatory issues facing rheumatologists.
Restoring reimbursement is an issue on the "front burner," he said, noting that studies have demonstrated an increase in hip fracture rates in the wake of declining reimbursement as some practices have abandoned DXA.
The DXA draft legislation is just one of a number of current legislative initiatives and issues that should be on rheumatologists’ radar, Dr. Schweitz said.
Sequestration
Under sequestration – a "heavy hammer" proposed as a way to push for agreement on a deficit-reduction plan and implemented when that agreement didn’t materialize – Medicare provider reductions are limited to 2% per year. Claims are reduced by 2% after beneficiary coinsurance or deductible. H.R. 1416, the Cancer Patient Protection Act of 2013, would terminate sequester cuts for all part B drugs, not just cancer drugs.
Medicare physician payment
Based on the formula for the Medicare sustainable growth rate, or SGR, physician payments will be cut by 24.4% if Congress does not intervene by Jan. 1, 2014. The latest cost estimate by the Congressional Budget Office for repeal of the SGR is $176 billion – far less than previous estimates and far less than the combined cost of prior short-term patches. President Barack Obama’s 2014 budget, as well as Congressional budgets and recent federal rulemakings, all assume that Congress will fix the SGR this year. Draft legislation released in July based on a replacement proposal from the bipartisan House Energy & Commerce Committee calls for a two-phase approach. Phase I would repeal the SGR entirely and replace it with a 5-year period of stable payments, including a 0.5% increase across-the-board, and phase II would involve implementation of an enhanced "PQRS-like Update Incentive Program." This would maintain fee-for-service as an option, allow providers to move into other models at any time, and would involve physician-driven quality measurement.
"This is something all of us should be aware of ... you can be sure that this whole system of reimbursement for what we do is going to change in the future," said Dr. Schweitz, who has a private practice in West Palm Beach, Fla.
How the changes would be paid for has not been defined, but there is a proposal from the House Ways & Means Committee that involves entitlement reform with increased part B/D deductibles and an increased age for eligibility.
Part B drug payments
A new bill (H.R. 800) that would affect physicians who provide in-office drug infusion excludes prompt pay discounts from manufacturers to wholesalers from the average sales price (ASP) calculation for part B drugs. This would improve payment for physicians who do not receive the discounts. However, the president’s 2014 budget assumes part B drugs are overpaid and proposes reducing Medicare payment from ASP plus 6% to ASP plus 3%.
This would require legislation to enact. The House and Senate budgets do not include any assumption regarding ASP.
"There’s very little chance, I think, that either side is going to be successful with this," Dr. Schweitz said.
The Independent Payment Advisory Board
The IPAB, a 15-member board created as part of the Affordable Care Act to make recommendations to cut spending when costs exceed growth targets, has been a contentious issue.
"There is a huge outcry in Congress against this IPAB," Dr. Schweitz said, explaining that there is a sense that the IPAB represents a usurping of the authority of Congress in controlling this part of Medicare activity.
A bipartisan repeal effort is underway in the form of H.R. 351/S. 351, the Protecting Seniors’ Access to Medicare Act. This has a chance of moving forward, but opposition in the Senate could hold it back, Dr. Schweitz said.
Of note, the president’s 2014 budget would lower the target rate that triggers IPAB recommendations from gross domestic product plus 1% to GDP plus 0.5%. Also, House Speaker John Boehner (R-Ohio) and Senate Minority Leader Mitch McConnell (R-Ky.) are on record saying that they will not nominate members to the IPAB, he said.
Private contracting
Private contracting is permitted under current law, but physicians who engage in it must opt out of Medicare for 2 years; patients who sign a private contract must pay out of pocket, even for services covered by Medicare. Under S. 236/H.R. 1310 – the Medicare Patient Empowerment Act – the 2-year opt-out requirement would be eliminated and beneficiaries could use their benefits to offset a portion of the fee set by their physician. This would protect low-income patients, "dual-eligibles," and patients with certain emergent or urgent health conditions. It is opposed by the Democratic Party and AARP, who see it as a cost-shifting mechanism; others feel it is a way to help reduce Medicare spending, he noted.
Medical liability reform
There’s been "a lot of noise but not much action" when it comes to medical liability reform, Dr. Schweitz said.
A House bill that would have included a $250,000 cap on noneconomic damages was paired with the IPAB repeal bill in the 112th Congress but did not make it to the Senate.
"There’s no such bill active in this [113th] Congress, but there are other kinds of liability reform acts that are more focused," he said.
These include H.R. 36/S. 961, the Health Care Safety Net Enhancement Act of 2013, which would extend Federal Tort Claims Act liability protections to on-call specialty physicians; H.R. 1733, the Good Samaritan Health Professionals Act of 2013, which would limit liability of health care professionals who volunteer to provide health care in response to a declared natural disaster; and H.R. 1473, the Standard of Care Protection Act, which would protect physicians from the use of quality measures for reimbursement issues in other federal health care laws to be used as standards of care in liability lawsuits.
Biosimilars
The president’s 2014 budget proposes to reduce the exclusivity for brand names from 12 years to 7 years beginning in 2014 and to prohibit "evergreening" – the securing of additional periods of exclusivity for brand biologics because of minor changes in product formulations.
Additionally, the Coalition of State Rheumatology Organizations is lobbying state legislatures to ensure that biosimilars legislation includes appropriate patient protections, such as physician notification of any substitution, appropriate record keeping, and "dispense as written" requirements. To date, five such bills have passed, with North Dakota, Utah, Virginia, and Oregon including all of these measures and Florida including all but notification.
Graduate medical education
Workforce shortages aren’t just a primary care issue. They also affect multiple specialties, including rheumatology. Though not unique in this regard, rheumatology is representative of an aging specialty, with more physicians reaching the end of their careers than young physicians entering the field. Coupled with the aging population, this raises concerns. The coalition and the Alliance of Specialty Medicine Policy Roundtable on Workforce sought cosponsors for H.R. 1180 and S. 577, the Resident Physician Shortage Reduction Act of 2013, and H.R. 1201, the Training Tomorrow’s Doctor Today Act. These legislative initiatives seek to address the problem of residency-slot shortages.
Dr. Schweitz reported having no disclosures.
LAS VEGAS – A coalition working to address dual-energy X-ray absorptiometry access issues is developing draft legislation that would establish a geographically adjusted national minimum payment for DXA, according to Dr. Michael Schweitz.
Under the draft legislation – a response to a series of reimbursement cuts over the past several years – DXA CPT codes 77080 and 77082 would be paid at $98 and $35, respectively, and a bundled code or bundled payment for bone density and vertebral fracture assessment studies would result in a national minimum payment of $133, Dr. Schweitz, president of the Coalition of State Rheumatology Organizations, said at Perspectives in Rheumatic Diseases 2013 during an update on federal legislative and regulatory issues facing rheumatologists.
Restoring reimbursement is an issue on the "front burner," he said, noting that studies have demonstrated an increase in hip fracture rates in the wake of declining reimbursement as some practices have abandoned DXA.
The DXA draft legislation is just one of a number of current legislative initiatives and issues that should be on rheumatologists’ radar, Dr. Schweitz said.
Sequestration
Under sequestration – a "heavy hammer" proposed as a way to push for agreement on a deficit-reduction plan and implemented when that agreement didn’t materialize – Medicare provider reductions are limited to 2% per year. Claims are reduced by 2% after beneficiary coinsurance or deductible. H.R. 1416, the Cancer Patient Protection Act of 2013, would terminate sequester cuts for all part B drugs, not just cancer drugs.
Medicare physician payment
Based on the formula for the Medicare sustainable growth rate, or SGR, physician payments will be cut by 24.4% if Congress does not intervene by Jan. 1, 2014. The latest cost estimate by the Congressional Budget Office for repeal of the SGR is $176 billion – far less than previous estimates and far less than the combined cost of prior short-term patches. President Barack Obama’s 2014 budget, as well as Congressional budgets and recent federal rulemakings, all assume that Congress will fix the SGR this year. Draft legislation released in July based on a replacement proposal from the bipartisan House Energy & Commerce Committee calls for a two-phase approach. Phase I would repeal the SGR entirely and replace it with a 5-year period of stable payments, including a 0.5% increase across-the-board, and phase II would involve implementation of an enhanced "PQRS-like Update Incentive Program." This would maintain fee-for-service as an option, allow providers to move into other models at any time, and would involve physician-driven quality measurement.
"This is something all of us should be aware of ... you can be sure that this whole system of reimbursement for what we do is going to change in the future," said Dr. Schweitz, who has a private practice in West Palm Beach, Fla.
How the changes would be paid for has not been defined, but there is a proposal from the House Ways & Means Committee that involves entitlement reform with increased part B/D deductibles and an increased age for eligibility.
Part B drug payments
A new bill (H.R. 800) that would affect physicians who provide in-office drug infusion excludes prompt pay discounts from manufacturers to wholesalers from the average sales price (ASP) calculation for part B drugs. This would improve payment for physicians who do not receive the discounts. However, the president’s 2014 budget assumes part B drugs are overpaid and proposes reducing Medicare payment from ASP plus 6% to ASP plus 3%.
This would require legislation to enact. The House and Senate budgets do not include any assumption regarding ASP.
"There’s very little chance, I think, that either side is going to be successful with this," Dr. Schweitz said.
The Independent Payment Advisory Board
The IPAB, a 15-member board created as part of the Affordable Care Act to make recommendations to cut spending when costs exceed growth targets, has been a contentious issue.
"There is a huge outcry in Congress against this IPAB," Dr. Schweitz said, explaining that there is a sense that the IPAB represents a usurping of the authority of Congress in controlling this part of Medicare activity.
A bipartisan repeal effort is underway in the form of H.R. 351/S. 351, the Protecting Seniors’ Access to Medicare Act. This has a chance of moving forward, but opposition in the Senate could hold it back, Dr. Schweitz said.
Of note, the president’s 2014 budget would lower the target rate that triggers IPAB recommendations from gross domestic product plus 1% to GDP plus 0.5%. Also, House Speaker John Boehner (R-Ohio) and Senate Minority Leader Mitch McConnell (R-Ky.) are on record saying that they will not nominate members to the IPAB, he said.
Private contracting
Private contracting is permitted under current law, but physicians who engage in it must opt out of Medicare for 2 years; patients who sign a private contract must pay out of pocket, even for services covered by Medicare. Under S. 236/H.R. 1310 – the Medicare Patient Empowerment Act – the 2-year opt-out requirement would be eliminated and beneficiaries could use their benefits to offset a portion of the fee set by their physician. This would protect low-income patients, "dual-eligibles," and patients with certain emergent or urgent health conditions. It is opposed by the Democratic Party and AARP, who see it as a cost-shifting mechanism; others feel it is a way to help reduce Medicare spending, he noted.
Medical liability reform
There’s been "a lot of noise but not much action" when it comes to medical liability reform, Dr. Schweitz said.
A House bill that would have included a $250,000 cap on noneconomic damages was paired with the IPAB repeal bill in the 112th Congress but did not make it to the Senate.
"There’s no such bill active in this [113th] Congress, but there are other kinds of liability reform acts that are more focused," he said.
These include H.R. 36/S. 961, the Health Care Safety Net Enhancement Act of 2013, which would extend Federal Tort Claims Act liability protections to on-call specialty physicians; H.R. 1733, the Good Samaritan Health Professionals Act of 2013, which would limit liability of health care professionals who volunteer to provide health care in response to a declared natural disaster; and H.R. 1473, the Standard of Care Protection Act, which would protect physicians from the use of quality measures for reimbursement issues in other federal health care laws to be used as standards of care in liability lawsuits.
Biosimilars
The president’s 2014 budget proposes to reduce the exclusivity for brand names from 12 years to 7 years beginning in 2014 and to prohibit "evergreening" – the securing of additional periods of exclusivity for brand biologics because of minor changes in product formulations.
Additionally, the Coalition of State Rheumatology Organizations is lobbying state legislatures to ensure that biosimilars legislation includes appropriate patient protections, such as physician notification of any substitution, appropriate record keeping, and "dispense as written" requirements. To date, five such bills have passed, with North Dakota, Utah, Virginia, and Oregon including all of these measures and Florida including all but notification.
Graduate medical education
Workforce shortages aren’t just a primary care issue. They also affect multiple specialties, including rheumatology. Though not unique in this regard, rheumatology is representative of an aging specialty, with more physicians reaching the end of their careers than young physicians entering the field. Coupled with the aging population, this raises concerns. The coalition and the Alliance of Specialty Medicine Policy Roundtable on Workforce sought cosponsors for H.R. 1180 and S. 577, the Resident Physician Shortage Reduction Act of 2013, and H.R. 1201, the Training Tomorrow’s Doctor Today Act. These legislative initiatives seek to address the problem of residency-slot shortages.
Dr. Schweitz reported having no disclosures.
LAS VEGAS – A coalition working to address dual-energy X-ray absorptiometry access issues is developing draft legislation that would establish a geographically adjusted national minimum payment for DXA, according to Dr. Michael Schweitz.
Under the draft legislation – a response to a series of reimbursement cuts over the past several years – DXA CPT codes 77080 and 77082 would be paid at $98 and $35, respectively, and a bundled code or bundled payment for bone density and vertebral fracture assessment studies would result in a national minimum payment of $133, Dr. Schweitz, president of the Coalition of State Rheumatology Organizations, said at Perspectives in Rheumatic Diseases 2013 during an update on federal legislative and regulatory issues facing rheumatologists.
Restoring reimbursement is an issue on the "front burner," he said, noting that studies have demonstrated an increase in hip fracture rates in the wake of declining reimbursement as some practices have abandoned DXA.
The DXA draft legislation is just one of a number of current legislative initiatives and issues that should be on rheumatologists’ radar, Dr. Schweitz said.
Sequestration
Under sequestration – a "heavy hammer" proposed as a way to push for agreement on a deficit-reduction plan and implemented when that agreement didn’t materialize – Medicare provider reductions are limited to 2% per year. Claims are reduced by 2% after beneficiary coinsurance or deductible. H.R. 1416, the Cancer Patient Protection Act of 2013, would terminate sequester cuts for all part B drugs, not just cancer drugs.
Medicare physician payment
Based on the formula for the Medicare sustainable growth rate, or SGR, physician payments will be cut by 24.4% if Congress does not intervene by Jan. 1, 2014. The latest cost estimate by the Congressional Budget Office for repeal of the SGR is $176 billion – far less than previous estimates and far less than the combined cost of prior short-term patches. President Barack Obama’s 2014 budget, as well as Congressional budgets and recent federal rulemakings, all assume that Congress will fix the SGR this year. Draft legislation released in July based on a replacement proposal from the bipartisan House Energy & Commerce Committee calls for a two-phase approach. Phase I would repeal the SGR entirely and replace it with a 5-year period of stable payments, including a 0.5% increase across-the-board, and phase II would involve implementation of an enhanced "PQRS-like Update Incentive Program." This would maintain fee-for-service as an option, allow providers to move into other models at any time, and would involve physician-driven quality measurement.
"This is something all of us should be aware of ... you can be sure that this whole system of reimbursement for what we do is going to change in the future," said Dr. Schweitz, who has a private practice in West Palm Beach, Fla.
How the changes would be paid for has not been defined, but there is a proposal from the House Ways & Means Committee that involves entitlement reform with increased part B/D deductibles and an increased age for eligibility.
Part B drug payments
A new bill (H.R. 800) that would affect physicians who provide in-office drug infusion excludes prompt pay discounts from manufacturers to wholesalers from the average sales price (ASP) calculation for part B drugs. This would improve payment for physicians who do not receive the discounts. However, the president’s 2014 budget assumes part B drugs are overpaid and proposes reducing Medicare payment from ASP plus 6% to ASP plus 3%.
This would require legislation to enact. The House and Senate budgets do not include any assumption regarding ASP.
"There’s very little chance, I think, that either side is going to be successful with this," Dr. Schweitz said.
The Independent Payment Advisory Board
The IPAB, a 15-member board created as part of the Affordable Care Act to make recommendations to cut spending when costs exceed growth targets, has been a contentious issue.
"There is a huge outcry in Congress against this IPAB," Dr. Schweitz said, explaining that there is a sense that the IPAB represents a usurping of the authority of Congress in controlling this part of Medicare activity.
A bipartisan repeal effort is underway in the form of H.R. 351/S. 351, the Protecting Seniors’ Access to Medicare Act. This has a chance of moving forward, but opposition in the Senate could hold it back, Dr. Schweitz said.
Of note, the president’s 2014 budget would lower the target rate that triggers IPAB recommendations from gross domestic product plus 1% to GDP plus 0.5%. Also, House Speaker John Boehner (R-Ohio) and Senate Minority Leader Mitch McConnell (R-Ky.) are on record saying that they will not nominate members to the IPAB, he said.
Private contracting
Private contracting is permitted under current law, but physicians who engage in it must opt out of Medicare for 2 years; patients who sign a private contract must pay out of pocket, even for services covered by Medicare. Under S. 236/H.R. 1310 – the Medicare Patient Empowerment Act – the 2-year opt-out requirement would be eliminated and beneficiaries could use their benefits to offset a portion of the fee set by their physician. This would protect low-income patients, "dual-eligibles," and patients with certain emergent or urgent health conditions. It is opposed by the Democratic Party and AARP, who see it as a cost-shifting mechanism; others feel it is a way to help reduce Medicare spending, he noted.
Medical liability reform
There’s been "a lot of noise but not much action" when it comes to medical liability reform, Dr. Schweitz said.
A House bill that would have included a $250,000 cap on noneconomic damages was paired with the IPAB repeal bill in the 112th Congress but did not make it to the Senate.
"There’s no such bill active in this [113th] Congress, but there are other kinds of liability reform acts that are more focused," he said.
These include H.R. 36/S. 961, the Health Care Safety Net Enhancement Act of 2013, which would extend Federal Tort Claims Act liability protections to on-call specialty physicians; H.R. 1733, the Good Samaritan Health Professionals Act of 2013, which would limit liability of health care professionals who volunteer to provide health care in response to a declared natural disaster; and H.R. 1473, the Standard of Care Protection Act, which would protect physicians from the use of quality measures for reimbursement issues in other federal health care laws to be used as standards of care in liability lawsuits.
Biosimilars
The president’s 2014 budget proposes to reduce the exclusivity for brand names from 12 years to 7 years beginning in 2014 and to prohibit "evergreening" – the securing of additional periods of exclusivity for brand biologics because of minor changes in product formulations.
Additionally, the Coalition of State Rheumatology Organizations is lobbying state legislatures to ensure that biosimilars legislation includes appropriate patient protections, such as physician notification of any substitution, appropriate record keeping, and "dispense as written" requirements. To date, five such bills have passed, with North Dakota, Utah, Virginia, and Oregon including all of these measures and Florida including all but notification.
Graduate medical education
Workforce shortages aren’t just a primary care issue. They also affect multiple specialties, including rheumatology. Though not unique in this regard, rheumatology is representative of an aging specialty, with more physicians reaching the end of their careers than young physicians entering the field. Coupled with the aging population, this raises concerns. The coalition and the Alliance of Specialty Medicine Policy Roundtable on Workforce sought cosponsors for H.R. 1180 and S. 577, the Resident Physician Shortage Reduction Act of 2013, and H.R. 1201, the Training Tomorrow’s Doctor Today Act. These legislative initiatives seek to address the problem of residency-slot shortages.
Dr. Schweitz reported having no disclosures.
AT PERSPECTIVES IN RHEUMATIC DISEASES 2013
Autoantibodies play role in myositis classification, treatment
LAS VEGAS – Autoantibodies and autoantibody subsets can be particularly helpful for classifying and treating patients with myositis, including those with myositis-associated interstitial lung disease, according to Dr. Chester V. Oddis.
Many autoantibody subsets are phenotypically and clinically well defined, and have clinical relevance, said Dr. Oddis, professor of medicine and associate director of the rheumatology fellowship training program at the University of Pittsburgh.
Serological classification isn’t always accurate or routinely available for all clinicians, but this may change in the near future as improved techniques for autoantibody detection become available, he said at Perspectives in Rheumatic Diseases 2013.
Anti-MDA-5 and interstitial lung disease
One autoantibody that has gained attention in recent years is anti-MDA-5, also known as anti-CADM-140, which is often seen in patients with amyopathic dermatomyositis (ADM).
Patients with ADM represent a subset of dermatomyositis patients who have cutaneous manifestations of dermatomyositis for 6 months or longer and have no clinical evidence of proximal muscle weakness but may have mild serum muscle enzyme abnormalities. More extensive muscle testing in these patients generally demonstrates no or minimal abnormalities. However, these patients should not be considered to have simply a benign cutaneous form of disease; in fact, they have a frequency of malignancy similar to that of patients with classic dermatomyositis (14% in one series of nearly 300 patients, compared with 15% in classic dermatomyositis).
In addition, ADM patients also have a relatively high frequency of lung disease, Dr. Oddis said. In a published review of the literature of nearly 200 patients with ADM, 10% had interstitial lung disease (ILD) – an important point given that the rash of dermatomyositis may be subtle and missed, he noted.
The Asian population seems to be particularly at risk for this complication. Two studies in recent years have demonstrated that Japanese ADM patients with anti-MDA-5 present with rapidly progressive ILD. A 2011 study showed an increased incidence of acute or subacute interstitial pneumonitis in Chinese patients. Other studies have shown similar findings in Korean and other Asian populations, Dr. Oddis noted.
The presence of anti-MDA-5 represents a novel cutaneous phenotype involving palmar papules and cutaneous ulcerations, severe vasculopathy, and rapidly progressive ILD. The target autoantigen in these cases is MDA-5, which is involved in innate immune defense against viruses, he explained, noting that this supports the possibility that a viral trigger plays a role in the disease.
"I think this complication is filtering into the U.S. population, as we have seen it in our myositis cohort," Dr. Oddis said of the anti-MDA-5 association with ADM and severe ILD. He noted that he recently cared for a 70-year-old white male with "double pneumonia" (a finding that "should always raise suspicion of autoimmune ILD") in June of 2012, a rash of dermatomyositis in September of 2012, and vasculitic skin changes in January of 2013. He presented without muscle weakness.
Anti-synthetase syndrome and ILD
Another autoantibody myositis subset involves the anti-synthetases, including PL-7, PL-12, EJ, and Jo-1.
Patients with anti-synthetase syndrome are generally a clinically homogeneous patient population characterized by fever, myositis, arthritis, Raynaud’s phenomenon, mechanic’s hands, and ILD. About 30%-40% of myositis patients have ILD, which is a significant contributor to morbidity and mortality.
Anti-Jo-1 is found in 50%-75% of these patients, and the coexistence of Ro52 may portend worse prognosis, Dr. Oddis said.
There are certain clinical features of ILD in polymyositis and dermatomyositis, including progressive dyspnea with or without nonproductive cough, lack of digital clubbing (unlike in idiopathic pulmonary fibrosis), and lack of pleuritis and pleural effusion in most cases (unlike in systemic lupus erythematosus). However, presentation can be variable, with about one-third of patients developing ILD before muscle or skin manifestations are apparent. Some patients present with acute disease (acute respiratory distress syndrome), and others present with subacute disease that is chronic and slowly progressing or asymptomatic.
It is important to understand when making a diagnosis of autoimmune ILD that not all patients will present with the classic anti-synthetase syndrome, Dr. Oddis said.
In some cases, patients will have an "incomplete" clinical syndrome with ILD alone or ILD with only subtle connective tissue disease findings, myositis-specific autoantibodies in the absence of myositis, and/or a negative antinuclear antibody (ANA) test, he explained.
The initial symptoms in patients may vary depending on the anti-synthetase autoantibody that is present. In a University of Pittsburgh study, for example, Jo-1 was found in 60% of cases; non-Jo-1 synthetase positive cases more often experienced Raynaud’s as their initial symptom, less often experienced muscle and joint problems as their initial symptom, and had a longer delay in diagnosis, compared with Jo-1 patients. Survival was also decreased, compared with Jo-1 patients.
As for ANA, about half of patients tested positive, whereas 72% demonstrated positive anti-cytoplasmic staining on indirect immunofluorescence.
The diagnosis of autoimmune ILD can be missed when there is a failure to recognize "incomplete" clinical syndromes, when there is a failure to order or detect myositis-specific autoantibodies – even in patients without myositis – and when a negative ANA is considered to be reassuring, Dr. Oddis said.
Dr. Oddis has served on an advisory board for Questcor.
The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
LAS VEGAS – Autoantibodies and autoantibody subsets can be particularly helpful for classifying and treating patients with myositis, including those with myositis-associated interstitial lung disease, according to Dr. Chester V. Oddis.
Many autoantibody subsets are phenotypically and clinically well defined, and have clinical relevance, said Dr. Oddis, professor of medicine and associate director of the rheumatology fellowship training program at the University of Pittsburgh.
Serological classification isn’t always accurate or routinely available for all clinicians, but this may change in the near future as improved techniques for autoantibody detection become available, he said at Perspectives in Rheumatic Diseases 2013.
Anti-MDA-5 and interstitial lung disease
One autoantibody that has gained attention in recent years is anti-MDA-5, also known as anti-CADM-140, which is often seen in patients with amyopathic dermatomyositis (ADM).
Patients with ADM represent a subset of dermatomyositis patients who have cutaneous manifestations of dermatomyositis for 6 months or longer and have no clinical evidence of proximal muscle weakness but may have mild serum muscle enzyme abnormalities. More extensive muscle testing in these patients generally demonstrates no or minimal abnormalities. However, these patients should not be considered to have simply a benign cutaneous form of disease; in fact, they have a frequency of malignancy similar to that of patients with classic dermatomyositis (14% in one series of nearly 300 patients, compared with 15% in classic dermatomyositis).
In addition, ADM patients also have a relatively high frequency of lung disease, Dr. Oddis said. In a published review of the literature of nearly 200 patients with ADM, 10% had interstitial lung disease (ILD) – an important point given that the rash of dermatomyositis may be subtle and missed, he noted.
The Asian population seems to be particularly at risk for this complication. Two studies in recent years have demonstrated that Japanese ADM patients with anti-MDA-5 present with rapidly progressive ILD. A 2011 study showed an increased incidence of acute or subacute interstitial pneumonitis in Chinese patients. Other studies have shown similar findings in Korean and other Asian populations, Dr. Oddis noted.
The presence of anti-MDA-5 represents a novel cutaneous phenotype involving palmar papules and cutaneous ulcerations, severe vasculopathy, and rapidly progressive ILD. The target autoantigen in these cases is MDA-5, which is involved in innate immune defense against viruses, he explained, noting that this supports the possibility that a viral trigger plays a role in the disease.
"I think this complication is filtering into the U.S. population, as we have seen it in our myositis cohort," Dr. Oddis said of the anti-MDA-5 association with ADM and severe ILD. He noted that he recently cared for a 70-year-old white male with "double pneumonia" (a finding that "should always raise suspicion of autoimmune ILD") in June of 2012, a rash of dermatomyositis in September of 2012, and vasculitic skin changes in January of 2013. He presented without muscle weakness.
Anti-synthetase syndrome and ILD
Another autoantibody myositis subset involves the anti-synthetases, including PL-7, PL-12, EJ, and Jo-1.
Patients with anti-synthetase syndrome are generally a clinically homogeneous patient population characterized by fever, myositis, arthritis, Raynaud’s phenomenon, mechanic’s hands, and ILD. About 30%-40% of myositis patients have ILD, which is a significant contributor to morbidity and mortality.
Anti-Jo-1 is found in 50%-75% of these patients, and the coexistence of Ro52 may portend worse prognosis, Dr. Oddis said.
There are certain clinical features of ILD in polymyositis and dermatomyositis, including progressive dyspnea with or without nonproductive cough, lack of digital clubbing (unlike in idiopathic pulmonary fibrosis), and lack of pleuritis and pleural effusion in most cases (unlike in systemic lupus erythematosus). However, presentation can be variable, with about one-third of patients developing ILD before muscle or skin manifestations are apparent. Some patients present with acute disease (acute respiratory distress syndrome), and others present with subacute disease that is chronic and slowly progressing or asymptomatic.
It is important to understand when making a diagnosis of autoimmune ILD that not all patients will present with the classic anti-synthetase syndrome, Dr. Oddis said.
In some cases, patients will have an "incomplete" clinical syndrome with ILD alone or ILD with only subtle connective tissue disease findings, myositis-specific autoantibodies in the absence of myositis, and/or a negative antinuclear antibody (ANA) test, he explained.
The initial symptoms in patients may vary depending on the anti-synthetase autoantibody that is present. In a University of Pittsburgh study, for example, Jo-1 was found in 60% of cases; non-Jo-1 synthetase positive cases more often experienced Raynaud’s as their initial symptom, less often experienced muscle and joint problems as their initial symptom, and had a longer delay in diagnosis, compared with Jo-1 patients. Survival was also decreased, compared with Jo-1 patients.
As for ANA, about half of patients tested positive, whereas 72% demonstrated positive anti-cytoplasmic staining on indirect immunofluorescence.
The diagnosis of autoimmune ILD can be missed when there is a failure to recognize "incomplete" clinical syndromes, when there is a failure to order or detect myositis-specific autoantibodies – even in patients without myositis – and when a negative ANA is considered to be reassuring, Dr. Oddis said.
Dr. Oddis has served on an advisory board for Questcor.
The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
LAS VEGAS – Autoantibodies and autoantibody subsets can be particularly helpful for classifying and treating patients with myositis, including those with myositis-associated interstitial lung disease, according to Dr. Chester V. Oddis.
Many autoantibody subsets are phenotypically and clinically well defined, and have clinical relevance, said Dr. Oddis, professor of medicine and associate director of the rheumatology fellowship training program at the University of Pittsburgh.
Serological classification isn’t always accurate or routinely available for all clinicians, but this may change in the near future as improved techniques for autoantibody detection become available, he said at Perspectives in Rheumatic Diseases 2013.
Anti-MDA-5 and interstitial lung disease
One autoantibody that has gained attention in recent years is anti-MDA-5, also known as anti-CADM-140, which is often seen in patients with amyopathic dermatomyositis (ADM).
Patients with ADM represent a subset of dermatomyositis patients who have cutaneous manifestations of dermatomyositis for 6 months or longer and have no clinical evidence of proximal muscle weakness but may have mild serum muscle enzyme abnormalities. More extensive muscle testing in these patients generally demonstrates no or minimal abnormalities. However, these patients should not be considered to have simply a benign cutaneous form of disease; in fact, they have a frequency of malignancy similar to that of patients with classic dermatomyositis (14% in one series of nearly 300 patients, compared with 15% in classic dermatomyositis).
In addition, ADM patients also have a relatively high frequency of lung disease, Dr. Oddis said. In a published review of the literature of nearly 200 patients with ADM, 10% had interstitial lung disease (ILD) – an important point given that the rash of dermatomyositis may be subtle and missed, he noted.
The Asian population seems to be particularly at risk for this complication. Two studies in recent years have demonstrated that Japanese ADM patients with anti-MDA-5 present with rapidly progressive ILD. A 2011 study showed an increased incidence of acute or subacute interstitial pneumonitis in Chinese patients. Other studies have shown similar findings in Korean and other Asian populations, Dr. Oddis noted.
The presence of anti-MDA-5 represents a novel cutaneous phenotype involving palmar papules and cutaneous ulcerations, severe vasculopathy, and rapidly progressive ILD. The target autoantigen in these cases is MDA-5, which is involved in innate immune defense against viruses, he explained, noting that this supports the possibility that a viral trigger plays a role in the disease.
"I think this complication is filtering into the U.S. population, as we have seen it in our myositis cohort," Dr. Oddis said of the anti-MDA-5 association with ADM and severe ILD. He noted that he recently cared for a 70-year-old white male with "double pneumonia" (a finding that "should always raise suspicion of autoimmune ILD") in June of 2012, a rash of dermatomyositis in September of 2012, and vasculitic skin changes in January of 2013. He presented without muscle weakness.
Anti-synthetase syndrome and ILD
Another autoantibody myositis subset involves the anti-synthetases, including PL-7, PL-12, EJ, and Jo-1.
Patients with anti-synthetase syndrome are generally a clinically homogeneous patient population characterized by fever, myositis, arthritis, Raynaud’s phenomenon, mechanic’s hands, and ILD. About 30%-40% of myositis patients have ILD, which is a significant contributor to morbidity and mortality.
Anti-Jo-1 is found in 50%-75% of these patients, and the coexistence of Ro52 may portend worse prognosis, Dr. Oddis said.
There are certain clinical features of ILD in polymyositis and dermatomyositis, including progressive dyspnea with or without nonproductive cough, lack of digital clubbing (unlike in idiopathic pulmonary fibrosis), and lack of pleuritis and pleural effusion in most cases (unlike in systemic lupus erythematosus). However, presentation can be variable, with about one-third of patients developing ILD before muscle or skin manifestations are apparent. Some patients present with acute disease (acute respiratory distress syndrome), and others present with subacute disease that is chronic and slowly progressing or asymptomatic.
It is important to understand when making a diagnosis of autoimmune ILD that not all patients will present with the classic anti-synthetase syndrome, Dr. Oddis said.
In some cases, patients will have an "incomplete" clinical syndrome with ILD alone or ILD with only subtle connective tissue disease findings, myositis-specific autoantibodies in the absence of myositis, and/or a negative antinuclear antibody (ANA) test, he explained.
The initial symptoms in patients may vary depending on the anti-synthetase autoantibody that is present. In a University of Pittsburgh study, for example, Jo-1 was found in 60% of cases; non-Jo-1 synthetase positive cases more often experienced Raynaud’s as their initial symptom, less often experienced muscle and joint problems as their initial symptom, and had a longer delay in diagnosis, compared with Jo-1 patients. Survival was also decreased, compared with Jo-1 patients.
As for ANA, about half of patients tested positive, whereas 72% demonstrated positive anti-cytoplasmic staining on indirect immunofluorescence.
The diagnosis of autoimmune ILD can be missed when there is a failure to recognize "incomplete" clinical syndromes, when there is a failure to order or detect myositis-specific autoantibodies – even in patients without myositis – and when a negative ANA is considered to be reassuring, Dr. Oddis said.
Dr. Oddis has served on an advisory board for Questcor.
The meeting was held by Global Academy for Medical Education. GAME and this news organization are owned by Frontline Medical Communications.
AT PERSPECTIVES IN RHEUMATIC DISEASES 2013