User login
New Insights Gained Into Biologics' Infection Risk
SNOWMASS, COLO. – A recent, much-ballyhooed, large, observational U.S. study that revised downward the risk of serious infections posed by tumor necrosis factor–alpha inhibitor therapy may have rendered false comfort to prescribing physicians.
The findings of the U.S. multicenter Safety Assessment of Biologic Therapy study (SABER) were roundly celebrated by many rheumatologists, dermatologists, and gastroenterologists who prescribe biologic agents for autoimmune diseases. But the SABER results are strikingly out of step with those of many large, well-conducted patient registries.
Moreover, at least one SABER organizer is skeptical about the validity of the study’s key conclusion that the serious infection risk in patients who’ve started on anti-TNF therapy isn’t significantly different than the risk in those started on methotrexate or another nonbiologic disease-modifying antirheumatic drug (DMARD). He cautioned against taking that conclusion as a lesson from SABER.
"Actually, my belief is that most of our findings in SABER are explained by the fact that anti-TNF agents are prednisone-sparing drugs," Dr. Kevin L. Winthrop said at the symposium, which was sponsored by the American College of Rheumatology. "My belief is that anti-TNF drugs do increase the risk of serious bacterial infections. The fact that they allow people to decrease their prednisone may result in roughly a canceling out of that risk."
SABER was a retrospective cohort study combining data from four large U.S. automated health databases. Investigators were able to compare outcomes in 16,022 patients with rheumatoid arthritis (RA), psoriasis, psoriatic arthritis, ankylosing spondylitis, or inflammatory bowel disease (IBD) who were placed on an anti-TNF biologic vs. an equal number of matched patients who were started on a conventional DMARD.
The rate of serious infections requiring hospitalization during the first year after starting therapy – while impressively high – didn’t differ significantly between the two groups: roughly 8% per year in the RA patients regardless of whether they were on anti-TNF therapy or a conventional DMARD, 5.4% in those with psoriasis or spondyloarthropathies, and 10% in patients with IBD (JAMA 2011;306:2331-9).
But SABER had a major limitation: Data on prednisone use were collected for the year before the new therapy was introduced, but not after, noted Dr. Winthrop, an infectious disease specialist at Oregon Health and Science University, Portland.
"When you put someone on a biologic, you do a lot of things for that person differently than when you put someone on, say, Plaquenil [hydroxychloroquine]. You screen them for tuberculosis, you may counsel them, you give them vaccines, hopefully you decrease their prednisone, and you may even take off some methotrexate if they have good disease control. All of that probably happens to a much greater extent in the biologic arm than the nonbiologic arm," he explained.
Although infliximab was associated with a 23%-26% higher risk of serious infection compared with adalimumab or etanercept in SABER participants with RA, patients on infliximab were also more likely to concurrently be on methotrexate. "That might have been enough to explain this risk difference," according to Dr. Winthrop.
Unlike the SABER findings, a significantly elevated serious infection risk in association with RA patients receiving anti-TNF agents has been reported from the British Society for Rheumatology registry (Arthritis Rheum. 2006;54:2368-76), from a Japanese prospective registry (J. Rheumatol. 2011;38:1258-64), from the CORONNA (Consortium of Rheumatology Researchers of North American) registry (Ann. Rheum. Dis. 2010;69:380-6), and from the German RABBIT registry (Ann. Rheum. Dis. 2011;70:1914-20).
These studies generally showed a time-dependent decline in infection risk in patients treated with anti-TNF agents. But physicians shouldn’t assume that this apparent drop off in risk over time is a real phenomenon; rather, it is largely due to what epidemiologists call survivor bias. In other words, people who develop serious infections or other problems early on drop out of treatment, so the case mix in the registries changes, he explained.
Dr. Winthrop was particularly impressed with what he termed the "very elegant" 5,044-patient RABBIT study, which was designed to be able to answer the question SABER couldn’t – namely, what happens to prednisone dosing when patients go on a TNF inhibitor? The answer, as was well documented in RABBIT, is that the use of prednisone drastically decreases.
Moreover, the German investigators, using a multivariate analysis, identified a set of independent risk factors for serious infection. These included treatment with a TNF inhibitor, which was associated with a 1.8-fold increased risk, compared with conventional DMARDS; treatment with glucocorticoids at 7.5-14 mg/day during the study period, which had a 2.1-fold risk; and treatment with steroids at 15 mg/day or more, which conferred a 4.7-fold increased risk of infection. Other studies have also demonstrated that glucocorticoids convey a high and dose-dependent risk of serious infection.
A key lesson of both RABBIT and SABER is that much of the increased serious infection risk stems from comorbid conditions. The RABBIT investigators identified three additional risk factors: chronic lung disease, chronic kidney disease, and age older than 60 years. RA patients who had these three additional risk factors and were on at least 15 mg/day of prednisone had a serious infection rate of 45% per year if they were on an anti-TNF agent and 25% per year when on a conventional DMARD. If they had two additional risk factors rather than three, their serious infection risk dropped to less than 20% per year on anti-TNF therapy, and about half that on a conventional DMARD.
In contrast, patients with all three additional risk factors who were on less than 7.5 mg/day of prednisone or none at all had a serious infection rate below 10% per year if they were on a TNF inhibitor, and 5% if on a nonbiologic DMARD, Dr. Winthrop noted.
Similarly, SABER participants with chronic obstructive pulmonary disease at baseline had an absolute two- to threefold greater risk of serious infection on a TNF inhibitor or DMARD, compared with patients without COPD. Baseline diabetes mellitus also magnified the serious infection risk in SABER, although not in RABBIT, he continued.
The RABBIT investigators found that improvement in functional capacity resulting from effective treatment significantly reduced the risk of serious infections. Indeed, functional improvement had a greater impact on infection risk than did improvement in the DAS28 (disease activity score based upon a 28-joint count).
SABER was funded by the Food and Drug Administration, the Agency for Healthcare Research and Quality, and the Department of Health and Human Services.
Dr. Winthrop reported having received consultant fees from Abbott, Amgen, and Pfizer as well as research funding from Pfizer.
SNOWMASS, COLO. – A recent, much-ballyhooed, large, observational U.S. study that revised downward the risk of serious infections posed by tumor necrosis factor–alpha inhibitor therapy may have rendered false comfort to prescribing physicians.
The findings of the U.S. multicenter Safety Assessment of Biologic Therapy study (SABER) were roundly celebrated by many rheumatologists, dermatologists, and gastroenterologists who prescribe biologic agents for autoimmune diseases. But the SABER results are strikingly out of step with those of many large, well-conducted patient registries.
Moreover, at least one SABER organizer is skeptical about the validity of the study’s key conclusion that the serious infection risk in patients who’ve started on anti-TNF therapy isn’t significantly different than the risk in those started on methotrexate or another nonbiologic disease-modifying antirheumatic drug (DMARD). He cautioned against taking that conclusion as a lesson from SABER.
"Actually, my belief is that most of our findings in SABER are explained by the fact that anti-TNF agents are prednisone-sparing drugs," Dr. Kevin L. Winthrop said at the symposium, which was sponsored by the American College of Rheumatology. "My belief is that anti-TNF drugs do increase the risk of serious bacterial infections. The fact that they allow people to decrease their prednisone may result in roughly a canceling out of that risk."
SABER was a retrospective cohort study combining data from four large U.S. automated health databases. Investigators were able to compare outcomes in 16,022 patients with rheumatoid arthritis (RA), psoriasis, psoriatic arthritis, ankylosing spondylitis, or inflammatory bowel disease (IBD) who were placed on an anti-TNF biologic vs. an equal number of matched patients who were started on a conventional DMARD.
The rate of serious infections requiring hospitalization during the first year after starting therapy – while impressively high – didn’t differ significantly between the two groups: roughly 8% per year in the RA patients regardless of whether they were on anti-TNF therapy or a conventional DMARD, 5.4% in those with psoriasis or spondyloarthropathies, and 10% in patients with IBD (JAMA 2011;306:2331-9).
But SABER had a major limitation: Data on prednisone use were collected for the year before the new therapy was introduced, but not after, noted Dr. Winthrop, an infectious disease specialist at Oregon Health and Science University, Portland.
"When you put someone on a biologic, you do a lot of things for that person differently than when you put someone on, say, Plaquenil [hydroxychloroquine]. You screen them for tuberculosis, you may counsel them, you give them vaccines, hopefully you decrease their prednisone, and you may even take off some methotrexate if they have good disease control. All of that probably happens to a much greater extent in the biologic arm than the nonbiologic arm," he explained.
Although infliximab was associated with a 23%-26% higher risk of serious infection compared with adalimumab or etanercept in SABER participants with RA, patients on infliximab were also more likely to concurrently be on methotrexate. "That might have been enough to explain this risk difference," according to Dr. Winthrop.
Unlike the SABER findings, a significantly elevated serious infection risk in association with RA patients receiving anti-TNF agents has been reported from the British Society for Rheumatology registry (Arthritis Rheum. 2006;54:2368-76), from a Japanese prospective registry (J. Rheumatol. 2011;38:1258-64), from the CORONNA (Consortium of Rheumatology Researchers of North American) registry (Ann. Rheum. Dis. 2010;69:380-6), and from the German RABBIT registry (Ann. Rheum. Dis. 2011;70:1914-20).
These studies generally showed a time-dependent decline in infection risk in patients treated with anti-TNF agents. But physicians shouldn’t assume that this apparent drop off in risk over time is a real phenomenon; rather, it is largely due to what epidemiologists call survivor bias. In other words, people who develop serious infections or other problems early on drop out of treatment, so the case mix in the registries changes, he explained.
Dr. Winthrop was particularly impressed with what he termed the "very elegant" 5,044-patient RABBIT study, which was designed to be able to answer the question SABER couldn’t – namely, what happens to prednisone dosing when patients go on a TNF inhibitor? The answer, as was well documented in RABBIT, is that the use of prednisone drastically decreases.
Moreover, the German investigators, using a multivariate analysis, identified a set of independent risk factors for serious infection. These included treatment with a TNF inhibitor, which was associated with a 1.8-fold increased risk, compared with conventional DMARDS; treatment with glucocorticoids at 7.5-14 mg/day during the study period, which had a 2.1-fold risk; and treatment with steroids at 15 mg/day or more, which conferred a 4.7-fold increased risk of infection. Other studies have also demonstrated that glucocorticoids convey a high and dose-dependent risk of serious infection.
A key lesson of both RABBIT and SABER is that much of the increased serious infection risk stems from comorbid conditions. The RABBIT investigators identified three additional risk factors: chronic lung disease, chronic kidney disease, and age older than 60 years. RA patients who had these three additional risk factors and were on at least 15 mg/day of prednisone had a serious infection rate of 45% per year if they were on an anti-TNF agent and 25% per year when on a conventional DMARD. If they had two additional risk factors rather than three, their serious infection risk dropped to less than 20% per year on anti-TNF therapy, and about half that on a conventional DMARD.
In contrast, patients with all three additional risk factors who were on less than 7.5 mg/day of prednisone or none at all had a serious infection rate below 10% per year if they were on a TNF inhibitor, and 5% if on a nonbiologic DMARD, Dr. Winthrop noted.
Similarly, SABER participants with chronic obstructive pulmonary disease at baseline had an absolute two- to threefold greater risk of serious infection on a TNF inhibitor or DMARD, compared with patients without COPD. Baseline diabetes mellitus also magnified the serious infection risk in SABER, although not in RABBIT, he continued.
The RABBIT investigators found that improvement in functional capacity resulting from effective treatment significantly reduced the risk of serious infections. Indeed, functional improvement had a greater impact on infection risk than did improvement in the DAS28 (disease activity score based upon a 28-joint count).
SABER was funded by the Food and Drug Administration, the Agency for Healthcare Research and Quality, and the Department of Health and Human Services.
Dr. Winthrop reported having received consultant fees from Abbott, Amgen, and Pfizer as well as research funding from Pfizer.
SNOWMASS, COLO. – A recent, much-ballyhooed, large, observational U.S. study that revised downward the risk of serious infections posed by tumor necrosis factor–alpha inhibitor therapy may have rendered false comfort to prescribing physicians.
The findings of the U.S. multicenter Safety Assessment of Biologic Therapy study (SABER) were roundly celebrated by many rheumatologists, dermatologists, and gastroenterologists who prescribe biologic agents for autoimmune diseases. But the SABER results are strikingly out of step with those of many large, well-conducted patient registries.
Moreover, at least one SABER organizer is skeptical about the validity of the study’s key conclusion that the serious infection risk in patients who’ve started on anti-TNF therapy isn’t significantly different than the risk in those started on methotrexate or another nonbiologic disease-modifying antirheumatic drug (DMARD). He cautioned against taking that conclusion as a lesson from SABER.
"Actually, my belief is that most of our findings in SABER are explained by the fact that anti-TNF agents are prednisone-sparing drugs," Dr. Kevin L. Winthrop said at the symposium, which was sponsored by the American College of Rheumatology. "My belief is that anti-TNF drugs do increase the risk of serious bacterial infections. The fact that they allow people to decrease their prednisone may result in roughly a canceling out of that risk."
SABER was a retrospective cohort study combining data from four large U.S. automated health databases. Investigators were able to compare outcomes in 16,022 patients with rheumatoid arthritis (RA), psoriasis, psoriatic arthritis, ankylosing spondylitis, or inflammatory bowel disease (IBD) who were placed on an anti-TNF biologic vs. an equal number of matched patients who were started on a conventional DMARD.
The rate of serious infections requiring hospitalization during the first year after starting therapy – while impressively high – didn’t differ significantly between the two groups: roughly 8% per year in the RA patients regardless of whether they were on anti-TNF therapy or a conventional DMARD, 5.4% in those with psoriasis or spondyloarthropathies, and 10% in patients with IBD (JAMA 2011;306:2331-9).
But SABER had a major limitation: Data on prednisone use were collected for the year before the new therapy was introduced, but not after, noted Dr. Winthrop, an infectious disease specialist at Oregon Health and Science University, Portland.
"When you put someone on a biologic, you do a lot of things for that person differently than when you put someone on, say, Plaquenil [hydroxychloroquine]. You screen them for tuberculosis, you may counsel them, you give them vaccines, hopefully you decrease their prednisone, and you may even take off some methotrexate if they have good disease control. All of that probably happens to a much greater extent in the biologic arm than the nonbiologic arm," he explained.
Although infliximab was associated with a 23%-26% higher risk of serious infection compared with adalimumab or etanercept in SABER participants with RA, patients on infliximab were also more likely to concurrently be on methotrexate. "That might have been enough to explain this risk difference," according to Dr. Winthrop.
Unlike the SABER findings, a significantly elevated serious infection risk in association with RA patients receiving anti-TNF agents has been reported from the British Society for Rheumatology registry (Arthritis Rheum. 2006;54:2368-76), from a Japanese prospective registry (J. Rheumatol. 2011;38:1258-64), from the CORONNA (Consortium of Rheumatology Researchers of North American) registry (Ann. Rheum. Dis. 2010;69:380-6), and from the German RABBIT registry (Ann. Rheum. Dis. 2011;70:1914-20).
These studies generally showed a time-dependent decline in infection risk in patients treated with anti-TNF agents. But physicians shouldn’t assume that this apparent drop off in risk over time is a real phenomenon; rather, it is largely due to what epidemiologists call survivor bias. In other words, people who develop serious infections or other problems early on drop out of treatment, so the case mix in the registries changes, he explained.
Dr. Winthrop was particularly impressed with what he termed the "very elegant" 5,044-patient RABBIT study, which was designed to be able to answer the question SABER couldn’t – namely, what happens to prednisone dosing when patients go on a TNF inhibitor? The answer, as was well documented in RABBIT, is that the use of prednisone drastically decreases.
Moreover, the German investigators, using a multivariate analysis, identified a set of independent risk factors for serious infection. These included treatment with a TNF inhibitor, which was associated with a 1.8-fold increased risk, compared with conventional DMARDS; treatment with glucocorticoids at 7.5-14 mg/day during the study period, which had a 2.1-fold risk; and treatment with steroids at 15 mg/day or more, which conferred a 4.7-fold increased risk of infection. Other studies have also demonstrated that glucocorticoids convey a high and dose-dependent risk of serious infection.
A key lesson of both RABBIT and SABER is that much of the increased serious infection risk stems from comorbid conditions. The RABBIT investigators identified three additional risk factors: chronic lung disease, chronic kidney disease, and age older than 60 years. RA patients who had these three additional risk factors and were on at least 15 mg/day of prednisone had a serious infection rate of 45% per year if they were on an anti-TNF agent and 25% per year when on a conventional DMARD. If they had two additional risk factors rather than three, their serious infection risk dropped to less than 20% per year on anti-TNF therapy, and about half that on a conventional DMARD.
In contrast, patients with all three additional risk factors who were on less than 7.5 mg/day of prednisone or none at all had a serious infection rate below 10% per year if they were on a TNF inhibitor, and 5% if on a nonbiologic DMARD, Dr. Winthrop noted.
Similarly, SABER participants with chronic obstructive pulmonary disease at baseline had an absolute two- to threefold greater risk of serious infection on a TNF inhibitor or DMARD, compared with patients without COPD. Baseline diabetes mellitus also magnified the serious infection risk in SABER, although not in RABBIT, he continued.
The RABBIT investigators found that improvement in functional capacity resulting from effective treatment significantly reduced the risk of serious infections. Indeed, functional improvement had a greater impact on infection risk than did improvement in the DAS28 (disease activity score based upon a 28-joint count).
SABER was funded by the Food and Drug Administration, the Agency for Healthcare Research and Quality, and the Department of Health and Human Services.
Dr. Winthrop reported having received consultant fees from Abbott, Amgen, and Pfizer as well as research funding from Pfizer.
EXPERT ANALYSIS FROM A SYMPOSIUM SPONSORED BY THE AMERICAN COLLEGE OF RHEUMATOLOGY
Foundation Takes Bold Approach to Psoriasis Management
The new National Psoriasis Foundation consensus guidelines on the management of plaque psoriasis take a practical approach to helping physicians manage the disease beyond topical treatment.
The guidelines feature a table summarizing the latest consensus on eight systemic medications, and they hone in on specific forms of psoriasis in a way that is more quickly understandable than in the past, according to lead author Dr. Sylvia Hsu, chief of Ben Taub Dermatology Service and a dermatology professor at Baylor College of Medicine in Houston.
"The aim is to simplify and clarify when to use these drugs for patients whose psoriasis is beyond topical treatment," she said. "A lot of the drugs can be used first line, so it’s not like a treatment algorithm."
Dr. Hsu acknowledged that the guidelines are a bit bold because they directly state, for example, that acitretin doesn’t work well for plaque psoriasis, and that evidence is scant to support the use of alefacept to achieve full clearance. The latter position was devised well before Astellas announced in December 2011 that it was pulling alefacept from the market, she said in an interview.
Physicians may also be surprised that the guidelines, published in Archives of Dermatology (2012;148:95-102), recommend cyclosporine as a short-term solution for up to 12 weeks to control a psoriasis flare.
"We know that you can use cyclosporine for up to a year because it’s FDA-approved for up to 12 months, but the typical use is as a quick-fix to transition to other agents that are slower acting," she said. "It’s a practical table."
Established drugs such as etanercept (Enbrel) and adalimumab (Humira) are recommended as first-line therapies for plaque psoriasis, but so is the newer monoclonal antibody ustekinumab (Stelara), so avoid the misconception that dermatologists need to prescribe systemic medications in a particular order based upon time on the market, Dr. Hsu noted.
The National Psoriasis Foundation (NPF) guidelines, however, do not provide a formula for how to treat patients, said coauthor Dr. Steven R. Feldman, professor of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C.
"They don’t say if you have this patient, this must be done first and then if that doesn’t work, do this second, and that third," he said. "There is no guideline of that type for psoriasis because no one treatment is better than all the other treatments in every way and patients weigh the various variables differently."
The new guidelines were adapted from the Canadian Guidelines for the Management of Plaque Psoriasis to reflect United States practice patterns, and are very similar to those published on the subject by the American Academy of Dermatology (AAD), Dr. Feldman said.
"I think these guidelines are clearly consensus guidelines, getting the experts together and deciding what they feel is best, whereas the AAD made a special effort to create evidence-based guidelines based on the available data," he said. "In the end, I think the recommendations are basically the same thing. Neither one tells physicians exactly what to do in a given situation; it’s more about describing the reasonable options that a patient and physician could choose in any situation."
NPF and AAD guidelines coauthor Dr. Mark Lebwohl, professor and chair of the department dermatology at Mount Sinai School of Medicine in New York, said the AAD guidelines are superb, but that "having this second set of guidelines allows us to answer even more questions."
He noted that there is often information in one guideline that is missing from another because new psoriasis developments happen so quickly. For example, a section recommending that physicians consider withholding tumor necrosis factor antagonists in psoriasis patients undergoing elective surgery was borrowed from the Canadian guidelines, but does not appear in the AAD guidelines, he said. The section discusses the controversy over the potential for increased risk of postsurgical infection in patients on biologics, and provides guidance on when specific agents should be withheld.
Conversely, the NPF guidelines discuss the pros and cons of the excimer laser, but this discussion is missing from the Canadian guidelines because the laser is not approved there.
"In a year, the guidelines will all be out of date again; they are a work in progress," said Dr. Lebwohl, who added that the NPF is trying to find a mechanism to update the guidelines in real time as new developments arise.
What the NPF/Canadian guidelines offer physicians is a rigorous evaluation of the literature with a patient-focused approach to psoriasis treatment, said Dr. Kim A. Papp, director of research at Probity Medical Research, Waterloo, Ont., who helped coauthor both sets of guidelines.
"We all appreciate that the art of medicine incorporates habit, consensus, experience, and hopefully sound data," he said. "Consequently, a second and perhaps equally important offering of the guidelines to physicians is an independent affirmation or an independent negation of one’s art in treating psoriasis."
Where the NPF guidelines may prove particularly helpful is in supporting that therapeutic choice with insurers. If a physician thought that a particular biologic agent was the best treatment option for a patient, that physician could point to the guidelines as a way to let insurers know that what is being recommended is within the standard of care, said Dr. Feldman.
The new guidelines could also push physicians to be more aggressive in using some of the available systemic agents. He pointed out that one of the "major enthusiasms" for creating the guidelines was a 2009 National Psoriasis Foundation survey finding that 57% of patients with severe psoriasis were receiving treatment insufficient to achieve adequate control (J. Am. Acad. Dermatol. 2007;57:957-62).
"There needed to be guidelines that would encourage physicians to provide patients treatment that met the severity of their illness," said Dr. Feldman, who suggested that safety concerns, dated training, and a perception that psoriasis isn’t such a bad disease, all played into the lack of aggressive treatment.
The NPF guidelines clearly detail the safety risks associated with each agent, and have a specific section on special populations, including pregnant women, patients with HIV, and those with hepatitis B or C.
For the purpose of the new guidelines, patients are considered to have moderate to severe psoriasis if they cannot achieve or would not be expected to achieve adequate control using topical agents, with adequacy defined by the patient’s own perception of the disease and its burdens. Cutoffs based on clinical metrics, such as the Psoriasis Area and Severity Index, percentage of body surface area affected, and the Dermatological Life Quality Index are necessary in clinical trials, but "have little value in daily practice," the authors noted.
Strict numeric cutoffs can also cause problems with insurers and reimbursement.
"This [document] provides the insurers and other regulators the opportunity to set more meaningful standards for their policies," Dr. Feldman said. "When they see guidelines published in the Archives and supported by the National Psoriasis Foundation, it lends a lot of weight to doing things the way doctors do things in the patient’s best interest."
The physicians interviewed for this story all disclosed consultancies and research arrangements with multiple companies that manufacture psoriasis treatments.
The new National Psoriasis Foundation consensus guidelines on the management of plaque psoriasis take a practical approach to helping physicians manage the disease beyond topical treatment.
The guidelines feature a table summarizing the latest consensus on eight systemic medications, and they hone in on specific forms of psoriasis in a way that is more quickly understandable than in the past, according to lead author Dr. Sylvia Hsu, chief of Ben Taub Dermatology Service and a dermatology professor at Baylor College of Medicine in Houston.
"The aim is to simplify and clarify when to use these drugs for patients whose psoriasis is beyond topical treatment," she said. "A lot of the drugs can be used first line, so it’s not like a treatment algorithm."
Dr. Hsu acknowledged that the guidelines are a bit bold because they directly state, for example, that acitretin doesn’t work well for plaque psoriasis, and that evidence is scant to support the use of alefacept to achieve full clearance. The latter position was devised well before Astellas announced in December 2011 that it was pulling alefacept from the market, she said in an interview.
Physicians may also be surprised that the guidelines, published in Archives of Dermatology (2012;148:95-102), recommend cyclosporine as a short-term solution for up to 12 weeks to control a psoriasis flare.
"We know that you can use cyclosporine for up to a year because it’s FDA-approved for up to 12 months, but the typical use is as a quick-fix to transition to other agents that are slower acting," she said. "It’s a practical table."
Established drugs such as etanercept (Enbrel) and adalimumab (Humira) are recommended as first-line therapies for plaque psoriasis, but so is the newer monoclonal antibody ustekinumab (Stelara), so avoid the misconception that dermatologists need to prescribe systemic medications in a particular order based upon time on the market, Dr. Hsu noted.
The National Psoriasis Foundation (NPF) guidelines, however, do not provide a formula for how to treat patients, said coauthor Dr. Steven R. Feldman, professor of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C.
"They don’t say if you have this patient, this must be done first and then if that doesn’t work, do this second, and that third," he said. "There is no guideline of that type for psoriasis because no one treatment is better than all the other treatments in every way and patients weigh the various variables differently."
The new guidelines were adapted from the Canadian Guidelines for the Management of Plaque Psoriasis to reflect United States practice patterns, and are very similar to those published on the subject by the American Academy of Dermatology (AAD), Dr. Feldman said.
"I think these guidelines are clearly consensus guidelines, getting the experts together and deciding what they feel is best, whereas the AAD made a special effort to create evidence-based guidelines based on the available data," he said. "In the end, I think the recommendations are basically the same thing. Neither one tells physicians exactly what to do in a given situation; it’s more about describing the reasonable options that a patient and physician could choose in any situation."
NPF and AAD guidelines coauthor Dr. Mark Lebwohl, professor and chair of the department dermatology at Mount Sinai School of Medicine in New York, said the AAD guidelines are superb, but that "having this second set of guidelines allows us to answer even more questions."
He noted that there is often information in one guideline that is missing from another because new psoriasis developments happen so quickly. For example, a section recommending that physicians consider withholding tumor necrosis factor antagonists in psoriasis patients undergoing elective surgery was borrowed from the Canadian guidelines, but does not appear in the AAD guidelines, he said. The section discusses the controversy over the potential for increased risk of postsurgical infection in patients on biologics, and provides guidance on when specific agents should be withheld.
Conversely, the NPF guidelines discuss the pros and cons of the excimer laser, but this discussion is missing from the Canadian guidelines because the laser is not approved there.
"In a year, the guidelines will all be out of date again; they are a work in progress," said Dr. Lebwohl, who added that the NPF is trying to find a mechanism to update the guidelines in real time as new developments arise.
What the NPF/Canadian guidelines offer physicians is a rigorous evaluation of the literature with a patient-focused approach to psoriasis treatment, said Dr. Kim A. Papp, director of research at Probity Medical Research, Waterloo, Ont., who helped coauthor both sets of guidelines.
"We all appreciate that the art of medicine incorporates habit, consensus, experience, and hopefully sound data," he said. "Consequently, a second and perhaps equally important offering of the guidelines to physicians is an independent affirmation or an independent negation of one’s art in treating psoriasis."
Where the NPF guidelines may prove particularly helpful is in supporting that therapeutic choice with insurers. If a physician thought that a particular biologic agent was the best treatment option for a patient, that physician could point to the guidelines as a way to let insurers know that what is being recommended is within the standard of care, said Dr. Feldman.
The new guidelines could also push physicians to be more aggressive in using some of the available systemic agents. He pointed out that one of the "major enthusiasms" for creating the guidelines was a 2009 National Psoriasis Foundation survey finding that 57% of patients with severe psoriasis were receiving treatment insufficient to achieve adequate control (J. Am. Acad. Dermatol. 2007;57:957-62).
"There needed to be guidelines that would encourage physicians to provide patients treatment that met the severity of their illness," said Dr. Feldman, who suggested that safety concerns, dated training, and a perception that psoriasis isn’t such a bad disease, all played into the lack of aggressive treatment.
The NPF guidelines clearly detail the safety risks associated with each agent, and have a specific section on special populations, including pregnant women, patients with HIV, and those with hepatitis B or C.
For the purpose of the new guidelines, patients are considered to have moderate to severe psoriasis if they cannot achieve or would not be expected to achieve adequate control using topical agents, with adequacy defined by the patient’s own perception of the disease and its burdens. Cutoffs based on clinical metrics, such as the Psoriasis Area and Severity Index, percentage of body surface area affected, and the Dermatological Life Quality Index are necessary in clinical trials, but "have little value in daily practice," the authors noted.
Strict numeric cutoffs can also cause problems with insurers and reimbursement.
"This [document] provides the insurers and other regulators the opportunity to set more meaningful standards for their policies," Dr. Feldman said. "When they see guidelines published in the Archives and supported by the National Psoriasis Foundation, it lends a lot of weight to doing things the way doctors do things in the patient’s best interest."
The physicians interviewed for this story all disclosed consultancies and research arrangements with multiple companies that manufacture psoriasis treatments.
The new National Psoriasis Foundation consensus guidelines on the management of plaque psoriasis take a practical approach to helping physicians manage the disease beyond topical treatment.
The guidelines feature a table summarizing the latest consensus on eight systemic medications, and they hone in on specific forms of psoriasis in a way that is more quickly understandable than in the past, according to lead author Dr. Sylvia Hsu, chief of Ben Taub Dermatology Service and a dermatology professor at Baylor College of Medicine in Houston.
"The aim is to simplify and clarify when to use these drugs for patients whose psoriasis is beyond topical treatment," she said. "A lot of the drugs can be used first line, so it’s not like a treatment algorithm."
Dr. Hsu acknowledged that the guidelines are a bit bold because they directly state, for example, that acitretin doesn’t work well for plaque psoriasis, and that evidence is scant to support the use of alefacept to achieve full clearance. The latter position was devised well before Astellas announced in December 2011 that it was pulling alefacept from the market, she said in an interview.
Physicians may also be surprised that the guidelines, published in Archives of Dermatology (2012;148:95-102), recommend cyclosporine as a short-term solution for up to 12 weeks to control a psoriasis flare.
"We know that you can use cyclosporine for up to a year because it’s FDA-approved for up to 12 months, but the typical use is as a quick-fix to transition to other agents that are slower acting," she said. "It’s a practical table."
Established drugs such as etanercept (Enbrel) and adalimumab (Humira) are recommended as first-line therapies for plaque psoriasis, but so is the newer monoclonal antibody ustekinumab (Stelara), so avoid the misconception that dermatologists need to prescribe systemic medications in a particular order based upon time on the market, Dr. Hsu noted.
The National Psoriasis Foundation (NPF) guidelines, however, do not provide a formula for how to treat patients, said coauthor Dr. Steven R. Feldman, professor of dermatology at Wake Forest Baptist Medical Center in Winston-Salem, N.C.
"They don’t say if you have this patient, this must be done first and then if that doesn’t work, do this second, and that third," he said. "There is no guideline of that type for psoriasis because no one treatment is better than all the other treatments in every way and patients weigh the various variables differently."
The new guidelines were adapted from the Canadian Guidelines for the Management of Plaque Psoriasis to reflect United States practice patterns, and are very similar to those published on the subject by the American Academy of Dermatology (AAD), Dr. Feldman said.
"I think these guidelines are clearly consensus guidelines, getting the experts together and deciding what they feel is best, whereas the AAD made a special effort to create evidence-based guidelines based on the available data," he said. "In the end, I think the recommendations are basically the same thing. Neither one tells physicians exactly what to do in a given situation; it’s more about describing the reasonable options that a patient and physician could choose in any situation."
NPF and AAD guidelines coauthor Dr. Mark Lebwohl, professor and chair of the department dermatology at Mount Sinai School of Medicine in New York, said the AAD guidelines are superb, but that "having this second set of guidelines allows us to answer even more questions."
He noted that there is often information in one guideline that is missing from another because new psoriasis developments happen so quickly. For example, a section recommending that physicians consider withholding tumor necrosis factor antagonists in psoriasis patients undergoing elective surgery was borrowed from the Canadian guidelines, but does not appear in the AAD guidelines, he said. The section discusses the controversy over the potential for increased risk of postsurgical infection in patients on biologics, and provides guidance on when specific agents should be withheld.
Conversely, the NPF guidelines discuss the pros and cons of the excimer laser, but this discussion is missing from the Canadian guidelines because the laser is not approved there.
"In a year, the guidelines will all be out of date again; they are a work in progress," said Dr. Lebwohl, who added that the NPF is trying to find a mechanism to update the guidelines in real time as new developments arise.
What the NPF/Canadian guidelines offer physicians is a rigorous evaluation of the literature with a patient-focused approach to psoriasis treatment, said Dr. Kim A. Papp, director of research at Probity Medical Research, Waterloo, Ont., who helped coauthor both sets of guidelines.
"We all appreciate that the art of medicine incorporates habit, consensus, experience, and hopefully sound data," he said. "Consequently, a second and perhaps equally important offering of the guidelines to physicians is an independent affirmation or an independent negation of one’s art in treating psoriasis."
Where the NPF guidelines may prove particularly helpful is in supporting that therapeutic choice with insurers. If a physician thought that a particular biologic agent was the best treatment option for a patient, that physician could point to the guidelines as a way to let insurers know that what is being recommended is within the standard of care, said Dr. Feldman.
The new guidelines could also push physicians to be more aggressive in using some of the available systemic agents. He pointed out that one of the "major enthusiasms" for creating the guidelines was a 2009 National Psoriasis Foundation survey finding that 57% of patients with severe psoriasis were receiving treatment insufficient to achieve adequate control (J. Am. Acad. Dermatol. 2007;57:957-62).
"There needed to be guidelines that would encourage physicians to provide patients treatment that met the severity of their illness," said Dr. Feldman, who suggested that safety concerns, dated training, and a perception that psoriasis isn’t such a bad disease, all played into the lack of aggressive treatment.
The NPF guidelines clearly detail the safety risks associated with each agent, and have a specific section on special populations, including pregnant women, patients with HIV, and those with hepatitis B or C.
For the purpose of the new guidelines, patients are considered to have moderate to severe psoriasis if they cannot achieve or would not be expected to achieve adequate control using topical agents, with adequacy defined by the patient’s own perception of the disease and its burdens. Cutoffs based on clinical metrics, such as the Psoriasis Area and Severity Index, percentage of body surface area affected, and the Dermatological Life Quality Index are necessary in clinical trials, but "have little value in daily practice," the authors noted.
Strict numeric cutoffs can also cause problems with insurers and reimbursement.
"This [document] provides the insurers and other regulators the opportunity to set more meaningful standards for their policies," Dr. Feldman said. "When they see guidelines published in the Archives and supported by the National Psoriasis Foundation, it lends a lot of weight to doing things the way doctors do things in the patient’s best interest."
The physicians interviewed for this story all disclosed consultancies and research arrangements with multiple companies that manufacture psoriasis treatments.
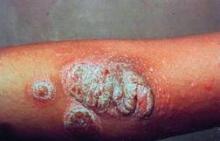
Anticipate Mycobacterial Lung Disease in Anti-TNF Users
SNOWMASS, COLO. – Rheumatoid arthritis patients on tumor necrosis factor inhibitors are at markedly increased risk for both tuberculosis and nontuberculous mycobacterial lung disease, as highlighted in data not yet published from Kaiser Permanente of Northern California that was discussed by investigator Dr. Kevin L. Winthrop at the symposium.
The crude incidence rate of nontuberculous mycobacterial disease in this 3.1-million-member health plan during the study years of 2000-2008 was 4.1 cases/100,000 person-years. The risk rose with age such that among plan members aged 50 years or older the rate was 11.8 cases/100,000 person-years. Plan members with rheumatoid arthritis (RA) who’d never been on a tumor necrosis factor (TNF) inhibitor had a moderately higher rate of 19.2 cases/100,000 person-years, probably because of their use of prednisone. But among 8,418 RA patients on TNF inhibitor therapy, the incidence of nontuberculous mycobacterial pulmonary disease shot up to 112 cases/100,000 person-years.
The tuberculosis incidence followed a similar pattern: 2.8 cases/100,000 person-years among the general Kaiser membership, 5.2 in those aged 50 years or older, 8.7 in RA patients never exposed to a TNF inhibitor, jumping to 56 cases/100,000 person-years among RA patients on an anti-TNF biologic, according to Dr. Winthrop.
In light of these data, physicians need to be on the lookout for nontuberculous mycobacterial pulmonary disease arising in RA patients using a TNF inhibitor.
"You will have patients with this. It’s best to diagnose them early, if possible, and get them off their biologic and also limit or discontinue prednisone," said Dr. Winthrop, an infectious diseases specialist at Oregon Health and Science University, Portland.
It’s his clinical impression, as well as that of other physicians participating in the Infectious Diseases Society of America’s Emerging Infections Network, that people who develop nontuberculous mycobacterial lung disease while on a TNF inhibitor tend to experience more rapid progression of their lung disease, he said at the meeting.
He and his coinvestigators at Kaiser also examined the pulmonary disease rates associated with individual TNF inhibitors. The nontuberculous mycobacterial lung disease rate in patients on etanercept was 35 cases/100,000 person-years of exposure, significantly less than the 116 cases/100,000 person-years with infliximab or 122 with adalimumab.
Similarly, the tuberculosis rate was lowest with etanercept at 17 cases/100,000 person-years as compared to 83 with infliximab and 61 with adalimumab.
The Kaiser experience confirms a 2010 report from the British Society for Rheumatology biologic registry that provided the first solid epidemiologic data showing that TNF inhibitors carry an increased tuberculosis risk. In the U.K. study, etanercept use was associated with a tuberculosis incidence of 39 cases per 100,000 person-years, significantly lower than the 136 cases per 100,000 person-years with infliximab or 144 with adalimumab. In contrast, the tuberculosis rate among more than 3,200 RA patients on a conventional disease-modifying anti-rheumatic drug was zero. The background tuberculosis incidence in the United Kingdom during that time period was about 12 cases/100,000 person-years (Ann. Rheum. Dis. 2010;69:522-8).
"I’m convinced that the monoclonal antibody TNF inhibitors [infliximab and adalimumab] cause more tuberculosis than [does] etanercept. I’m not convinced I know why," Dr. Winthrop admitted.
Numerous potential mechanisms have been floated to explain this differential effect. The two he finds most plausible are that etanercept is less able to penetrate tuberculosis granulomas than are the monoclonal antibody TNF inhibitors, as shown in a mouse model, and the possibility – as yet unproven – that etanercept might also cause less downregulation of CD8 cells producing the antimicrobial peptides perforin and granulysin, which are directed against Mycobacterium tuberculosis.
Dr. Winthrop said the epidemiology of nontuberculous mycobacterial pulmonary disease is changing. Decades ago, it was viewed as a disease of elderly men. As the incidence has climbed during the past 2 decades, however, the disease has come to be recognized as mainly one of postmenopausal women, typically with no history of underlying lung disease or smoking. The phenotype is one of an elderly woman who is tall, slender, and underweight, often with mitral valve prolapse, scoliosis, or pectus defects.
In a large study conducted at four geographically diverse large health plans, the annual prevalence of nontuberculous mycobacterial lung disease among persons age 60 years or older rose from 19.6 cases/100,000 in 1994-1996 to 26.7 cases/100,000 person-years in 2004-2006, a rate two- to threefold greater than the prevalence of tuberculosis at those sites during 2004-2006 (Am. J. Respir. Crit. Care Med. 2010;182:970-6).
Nontuberculous mycobacteria are ubiquitous in tap water and soil. Unlike tuberculosis, which is spread from person to person by coughing, nontuberculous mycobacterial infections are acquired directly from the environment.
Dr. Winthrop said he is reluctant to recommend resuming biologic therapy after a RA patient has been treated for nontuberculous mycobacterial lung disease or coccidioidomycosis.
Dr. Winthrop reported having received consultant fees from Abbott, Amgen, and Pfizer as well as research funding from Pfizer.
SNOWMASS, COLO. – Rheumatoid arthritis patients on tumor necrosis factor inhibitors are at markedly increased risk for both tuberculosis and nontuberculous mycobacterial lung disease, as highlighted in data not yet published from Kaiser Permanente of Northern California that was discussed by investigator Dr. Kevin L. Winthrop at the symposium.
The crude incidence rate of nontuberculous mycobacterial disease in this 3.1-million-member health plan during the study years of 2000-2008 was 4.1 cases/100,000 person-years. The risk rose with age such that among plan members aged 50 years or older the rate was 11.8 cases/100,000 person-years. Plan members with rheumatoid arthritis (RA) who’d never been on a tumor necrosis factor (TNF) inhibitor had a moderately higher rate of 19.2 cases/100,000 person-years, probably because of their use of prednisone. But among 8,418 RA patients on TNF inhibitor therapy, the incidence of nontuberculous mycobacterial pulmonary disease shot up to 112 cases/100,000 person-years.
The tuberculosis incidence followed a similar pattern: 2.8 cases/100,000 person-years among the general Kaiser membership, 5.2 in those aged 50 years or older, 8.7 in RA patients never exposed to a TNF inhibitor, jumping to 56 cases/100,000 person-years among RA patients on an anti-TNF biologic, according to Dr. Winthrop.
In light of these data, physicians need to be on the lookout for nontuberculous mycobacterial pulmonary disease arising in RA patients using a TNF inhibitor.
"You will have patients with this. It’s best to diagnose them early, if possible, and get them off their biologic and also limit or discontinue prednisone," said Dr. Winthrop, an infectious diseases specialist at Oregon Health and Science University, Portland.
It’s his clinical impression, as well as that of other physicians participating in the Infectious Diseases Society of America’s Emerging Infections Network, that people who develop nontuberculous mycobacterial lung disease while on a TNF inhibitor tend to experience more rapid progression of their lung disease, he said at the meeting.
He and his coinvestigators at Kaiser also examined the pulmonary disease rates associated with individual TNF inhibitors. The nontuberculous mycobacterial lung disease rate in patients on etanercept was 35 cases/100,000 person-years of exposure, significantly less than the 116 cases/100,000 person-years with infliximab or 122 with adalimumab.
Similarly, the tuberculosis rate was lowest with etanercept at 17 cases/100,000 person-years as compared to 83 with infliximab and 61 with adalimumab.
The Kaiser experience confirms a 2010 report from the British Society for Rheumatology biologic registry that provided the first solid epidemiologic data showing that TNF inhibitors carry an increased tuberculosis risk. In the U.K. study, etanercept use was associated with a tuberculosis incidence of 39 cases per 100,000 person-years, significantly lower than the 136 cases per 100,000 person-years with infliximab or 144 with adalimumab. In contrast, the tuberculosis rate among more than 3,200 RA patients on a conventional disease-modifying anti-rheumatic drug was zero. The background tuberculosis incidence in the United Kingdom during that time period was about 12 cases/100,000 person-years (Ann. Rheum. Dis. 2010;69:522-8).
"I’m convinced that the monoclonal antibody TNF inhibitors [infliximab and adalimumab] cause more tuberculosis than [does] etanercept. I’m not convinced I know why," Dr. Winthrop admitted.
Numerous potential mechanisms have been floated to explain this differential effect. The two he finds most plausible are that etanercept is less able to penetrate tuberculosis granulomas than are the monoclonal antibody TNF inhibitors, as shown in a mouse model, and the possibility – as yet unproven – that etanercept might also cause less downregulation of CD8 cells producing the antimicrobial peptides perforin and granulysin, which are directed against Mycobacterium tuberculosis.
Dr. Winthrop said the epidemiology of nontuberculous mycobacterial pulmonary disease is changing. Decades ago, it was viewed as a disease of elderly men. As the incidence has climbed during the past 2 decades, however, the disease has come to be recognized as mainly one of postmenopausal women, typically with no history of underlying lung disease or smoking. The phenotype is one of an elderly woman who is tall, slender, and underweight, often with mitral valve prolapse, scoliosis, or pectus defects.
In a large study conducted at four geographically diverse large health plans, the annual prevalence of nontuberculous mycobacterial lung disease among persons age 60 years or older rose from 19.6 cases/100,000 in 1994-1996 to 26.7 cases/100,000 person-years in 2004-2006, a rate two- to threefold greater than the prevalence of tuberculosis at those sites during 2004-2006 (Am. J. Respir. Crit. Care Med. 2010;182:970-6).
Nontuberculous mycobacteria are ubiquitous in tap water and soil. Unlike tuberculosis, which is spread from person to person by coughing, nontuberculous mycobacterial infections are acquired directly from the environment.
Dr. Winthrop said he is reluctant to recommend resuming biologic therapy after a RA patient has been treated for nontuberculous mycobacterial lung disease or coccidioidomycosis.
Dr. Winthrop reported having received consultant fees from Abbott, Amgen, and Pfizer as well as research funding from Pfizer.
SNOWMASS, COLO. – Rheumatoid arthritis patients on tumor necrosis factor inhibitors are at markedly increased risk for both tuberculosis and nontuberculous mycobacterial lung disease, as highlighted in data not yet published from Kaiser Permanente of Northern California that was discussed by investigator Dr. Kevin L. Winthrop at the symposium.
The crude incidence rate of nontuberculous mycobacterial disease in this 3.1-million-member health plan during the study years of 2000-2008 was 4.1 cases/100,000 person-years. The risk rose with age such that among plan members aged 50 years or older the rate was 11.8 cases/100,000 person-years. Plan members with rheumatoid arthritis (RA) who’d never been on a tumor necrosis factor (TNF) inhibitor had a moderately higher rate of 19.2 cases/100,000 person-years, probably because of their use of prednisone. But among 8,418 RA patients on TNF inhibitor therapy, the incidence of nontuberculous mycobacterial pulmonary disease shot up to 112 cases/100,000 person-years.
The tuberculosis incidence followed a similar pattern: 2.8 cases/100,000 person-years among the general Kaiser membership, 5.2 in those aged 50 years or older, 8.7 in RA patients never exposed to a TNF inhibitor, jumping to 56 cases/100,000 person-years among RA patients on an anti-TNF biologic, according to Dr. Winthrop.
In light of these data, physicians need to be on the lookout for nontuberculous mycobacterial pulmonary disease arising in RA patients using a TNF inhibitor.
"You will have patients with this. It’s best to diagnose them early, if possible, and get them off their biologic and also limit or discontinue prednisone," said Dr. Winthrop, an infectious diseases specialist at Oregon Health and Science University, Portland.
It’s his clinical impression, as well as that of other physicians participating in the Infectious Diseases Society of America’s Emerging Infections Network, that people who develop nontuberculous mycobacterial lung disease while on a TNF inhibitor tend to experience more rapid progression of their lung disease, he said at the meeting.
He and his coinvestigators at Kaiser also examined the pulmonary disease rates associated with individual TNF inhibitors. The nontuberculous mycobacterial lung disease rate in patients on etanercept was 35 cases/100,000 person-years of exposure, significantly less than the 116 cases/100,000 person-years with infliximab or 122 with adalimumab.
Similarly, the tuberculosis rate was lowest with etanercept at 17 cases/100,000 person-years as compared to 83 with infliximab and 61 with adalimumab.
The Kaiser experience confirms a 2010 report from the British Society for Rheumatology biologic registry that provided the first solid epidemiologic data showing that TNF inhibitors carry an increased tuberculosis risk. In the U.K. study, etanercept use was associated with a tuberculosis incidence of 39 cases per 100,000 person-years, significantly lower than the 136 cases per 100,000 person-years with infliximab or 144 with adalimumab. In contrast, the tuberculosis rate among more than 3,200 RA patients on a conventional disease-modifying anti-rheumatic drug was zero. The background tuberculosis incidence in the United Kingdom during that time period was about 12 cases/100,000 person-years (Ann. Rheum. Dis. 2010;69:522-8).
"I’m convinced that the monoclonal antibody TNF inhibitors [infliximab and adalimumab] cause more tuberculosis than [does] etanercept. I’m not convinced I know why," Dr. Winthrop admitted.
Numerous potential mechanisms have been floated to explain this differential effect. The two he finds most plausible are that etanercept is less able to penetrate tuberculosis granulomas than are the monoclonal antibody TNF inhibitors, as shown in a mouse model, and the possibility – as yet unproven – that etanercept might also cause less downregulation of CD8 cells producing the antimicrobial peptides perforin and granulysin, which are directed against Mycobacterium tuberculosis.
Dr. Winthrop said the epidemiology of nontuberculous mycobacterial pulmonary disease is changing. Decades ago, it was viewed as a disease of elderly men. As the incidence has climbed during the past 2 decades, however, the disease has come to be recognized as mainly one of postmenopausal women, typically with no history of underlying lung disease or smoking. The phenotype is one of an elderly woman who is tall, slender, and underweight, often with mitral valve prolapse, scoliosis, or pectus defects.
In a large study conducted at four geographically diverse large health plans, the annual prevalence of nontuberculous mycobacterial lung disease among persons age 60 years or older rose from 19.6 cases/100,000 in 1994-1996 to 26.7 cases/100,000 person-years in 2004-2006, a rate two- to threefold greater than the prevalence of tuberculosis at those sites during 2004-2006 (Am. J. Respir. Crit. Care Med. 2010;182:970-6).
Nontuberculous mycobacteria are ubiquitous in tap water and soil. Unlike tuberculosis, which is spread from person to person by coughing, nontuberculous mycobacterial infections are acquired directly from the environment.
Dr. Winthrop said he is reluctant to recommend resuming biologic therapy after a RA patient has been treated for nontuberculous mycobacterial lung disease or coccidioidomycosis.
Dr. Winthrop reported having received consultant fees from Abbott, Amgen, and Pfizer as well as research funding from Pfizer.
EXPERT ANALYSIS FROM A SYMPOSIUM SPONSORED BY THE AMERICAN COLLEGE OF RHEUMATOLOGY
FDA Issues Guidelines on Developing Biosimilar Products
The Food and Drug Administration on Feb. 9 issued some guidelines for industry on how to develop so-called biosimilar products.
The agency has been open to receiving applications for such products since a specific approval pathway was created under the Affordable Care Act in March 2010, but without specific guidance, there have been no such applications so far, according to FDA officials.
"These draft documents are designed to help industry develop biosimilar versions of approved biological products, which can enhance competition and may lead to better patient access and lower cost to consumers," said Dr. Janet Woodcock, director of FDA’s Center for Drug Evaluation and Research, in a statement.
The three "draft guidances" the agency issued "should help biosimilar developers move forward to understand the agency’s expectations," and also provide a "transparent, clear, predictable regulatory pathway," said Dr. Rachel Sherman, director in the Office of Medical Policy at the FDA’s Center for Drug Evaluation and Research.
Biosimilars are biological products that are highly similar to an already approved biological product, according to the FDA.
The first draft guidance, "Scientific Considerations in Demonstrating Biosimilarity to a Reference Product," is considered the key document, said Dr. Sherman. It outlines the basic requirements for how a company can show that its product is similar to the biologic that’s already approved. Developers will have to primarily go through a series of analytical tests to prove similarity.
The FDA would then weigh that information in determining whether the product would require further animal or human testing, said Dr. Sherman.
A biosimilar will not automatically be deemed interchangeable with an existing product. That is a separate determination, she said, noting that it would require further safety and efficacy testing.
So far, the agency has received requests for consultations from 35 companies seeking to develop biosimilars for 11 reference products, said Dr. Sherman. The agency has held 21 meetings. But there have been no formal applications for approval.
The two other guidance documents were, "Quality Considerations in Demonstrating Biosimilarity to a Reference Protein Product, which gives "excruciating detail" about the analytics to be used, said Dr. Sherman, and "Biosimilars: Questions and Answers Regarding Implementation of the Biologics Price Competition and Innovation Act of 2009."
The latter document contains many of the questions companies have asked in previous consultations with the FDA, said Dr. Sherman. She expects that to be updated as the process evolves. The FDA will hold a public meeting to gather comments on the draft guidances, Dr. Sherman said.
Dr. Richard Dolinar, a clinical endocrinologist in Phoenix and chairman of the Alliance for Safe Biologic Medicines said in a statement that the organization welcomed the draft guidance documents as "an important step forward in expanding access to existing biological therapies."
ASBM members include companies concerned about biosimilar encroachment, such as Amgen and Genentech, and the Biotech Industry Organization, and along with several patient advocacy groups.
The Food and Drug Administration on Feb. 9 issued some guidelines for industry on how to develop so-called biosimilar products.
The agency has been open to receiving applications for such products since a specific approval pathway was created under the Affordable Care Act in March 2010, but without specific guidance, there have been no such applications so far, according to FDA officials.
"These draft documents are designed to help industry develop biosimilar versions of approved biological products, which can enhance competition and may lead to better patient access and lower cost to consumers," said Dr. Janet Woodcock, director of FDA’s Center for Drug Evaluation and Research, in a statement.
The three "draft guidances" the agency issued "should help biosimilar developers move forward to understand the agency’s expectations," and also provide a "transparent, clear, predictable regulatory pathway," said Dr. Rachel Sherman, director in the Office of Medical Policy at the FDA’s Center for Drug Evaluation and Research.
Biosimilars are biological products that are highly similar to an already approved biological product, according to the FDA.
The first draft guidance, "Scientific Considerations in Demonstrating Biosimilarity to a Reference Product," is considered the key document, said Dr. Sherman. It outlines the basic requirements for how a company can show that its product is similar to the biologic that’s already approved. Developers will have to primarily go through a series of analytical tests to prove similarity.
The FDA would then weigh that information in determining whether the product would require further animal or human testing, said Dr. Sherman.
A biosimilar will not automatically be deemed interchangeable with an existing product. That is a separate determination, she said, noting that it would require further safety and efficacy testing.
So far, the agency has received requests for consultations from 35 companies seeking to develop biosimilars for 11 reference products, said Dr. Sherman. The agency has held 21 meetings. But there have been no formal applications for approval.
The two other guidance documents were, "Quality Considerations in Demonstrating Biosimilarity to a Reference Protein Product, which gives "excruciating detail" about the analytics to be used, said Dr. Sherman, and "Biosimilars: Questions and Answers Regarding Implementation of the Biologics Price Competition and Innovation Act of 2009."
The latter document contains many of the questions companies have asked in previous consultations with the FDA, said Dr. Sherman. She expects that to be updated as the process evolves. The FDA will hold a public meeting to gather comments on the draft guidances, Dr. Sherman said.
Dr. Richard Dolinar, a clinical endocrinologist in Phoenix and chairman of the Alliance for Safe Biologic Medicines said in a statement that the organization welcomed the draft guidance documents as "an important step forward in expanding access to existing biological therapies."
ASBM members include companies concerned about biosimilar encroachment, such as Amgen and Genentech, and the Biotech Industry Organization, and along with several patient advocacy groups.
The Food and Drug Administration on Feb. 9 issued some guidelines for industry on how to develop so-called biosimilar products.
The agency has been open to receiving applications for such products since a specific approval pathway was created under the Affordable Care Act in March 2010, but without specific guidance, there have been no such applications so far, according to FDA officials.
"These draft documents are designed to help industry develop biosimilar versions of approved biological products, which can enhance competition and may lead to better patient access and lower cost to consumers," said Dr. Janet Woodcock, director of FDA’s Center for Drug Evaluation and Research, in a statement.
The three "draft guidances" the agency issued "should help biosimilar developers move forward to understand the agency’s expectations," and also provide a "transparent, clear, predictable regulatory pathway," said Dr. Rachel Sherman, director in the Office of Medical Policy at the FDA’s Center for Drug Evaluation and Research.
Biosimilars are biological products that are highly similar to an already approved biological product, according to the FDA.
The first draft guidance, "Scientific Considerations in Demonstrating Biosimilarity to a Reference Product," is considered the key document, said Dr. Sherman. It outlines the basic requirements for how a company can show that its product is similar to the biologic that’s already approved. Developers will have to primarily go through a series of analytical tests to prove similarity.
The FDA would then weigh that information in determining whether the product would require further animal or human testing, said Dr. Sherman.
A biosimilar will not automatically be deemed interchangeable with an existing product. That is a separate determination, she said, noting that it would require further safety and efficacy testing.
So far, the agency has received requests for consultations from 35 companies seeking to develop biosimilars for 11 reference products, said Dr. Sherman. The agency has held 21 meetings. But there have been no formal applications for approval.
The two other guidance documents were, "Quality Considerations in Demonstrating Biosimilarity to a Reference Protein Product, which gives "excruciating detail" about the analytics to be used, said Dr. Sherman, and "Biosimilars: Questions and Answers Regarding Implementation of the Biologics Price Competition and Innovation Act of 2009."
The latter document contains many of the questions companies have asked in previous consultations with the FDA, said Dr. Sherman. She expects that to be updated as the process evolves. The FDA will hold a public meeting to gather comments on the draft guidances, Dr. Sherman said.
Dr. Richard Dolinar, a clinical endocrinologist in Phoenix and chairman of the Alliance for Safe Biologic Medicines said in a statement that the organization welcomed the draft guidance documents as "an important step forward in expanding access to existing biological therapies."
ASBM members include companies concerned about biosimilar encroachment, such as Amgen and Genentech, and the Biotech Industry Organization, and along with several patient advocacy groups.
Hair: The Long and Short of It
Lasers and Light Devices for Psoriasis, Part 2: PDL, Nd:YAG Laser, CO2 Laser, and PDT
Earlier Is Better in Psoriasis Treatment
RIO GRANDE, P.R. – Treating psoriasis patients earlier in life could help prevent later physical and psychological problems, according to Dr. Alexa B. Kimball.
"We used to think that we should save our therapies until our patients really needed them, because we were afraid that toxicity might accumulate," Dr. Kimball said at the annual Caribbean Dermatology Symposium. However, that thinking has changed, thanks in part to the availability of safer treatment options.
But of equal importance, "treating patients early in their disease may have an impact that affects the rest of their lives," including jobs, education, socioeconomic status, and curbing the development of health problems like obesity, cardiovascular disease, and psychiatric disorders, she said.
The quality of life issues associated with psoriasis are well known, but recent data confirm that physical and mental comorbidities start in childhood.
According to recent data from the National Psoriasis Foundation, 38% of children with psoriasis reported being bullied because of their condition, noted Dr. Kimball of Massachusetts General Hospital and Harvard Medical School, both in Boston.
Another study found that approximately one-third of children aged 4-17 years with psoriasis had a body mass index greater than the 95th percentile (N. Engl. J. Med. 2008;358:241-51). Conditions such as childhood obesity are not easily managed, and have significant implications for future health, she said.
In a retrospective study of 7,404 psoriasis patients younger than 18 years and 37,020 healthy controls, children with psoriasis were significantly more likely than controls to develop any psychiatric disorder (5% vs. 4%), depression (3% vs. 2%), and anxiety (2% vs. 1%), Dr. Kimball and her colleagues found (J. Am. Acad. Dermatol. 2012 Jan. 16 [doi:10.1016/j.jaad.2011.11.948]).
And the likelihood of comorbidities in psoriasis patients continues as they grow up, she said. "Chronic disease interacts with psychosocial and health events in a complex and ongoing manner throughout a person’s life."
Comorbidities in psoriasis patients appear to accumulate over time. Dr. Kimball cited data from the Nurses’ Health Study II, a cohort including more than 100,000 women who were aged 27-44 years in 1991. In a subset of 1,813 women with psoriasis, the risk of diabetes was approximately 60% higher, and the risk of hypertension was almost 20% higher, compared with women without psoriasis (Arch. Dermatol. 2009;145:379-82).
In a case-control study conducted by Dr. Kimball and her colleagues, cardiovascular disease, diabetes, depression, hyperlipidemia, hypertension, and obesity all increased significantly in psoriasis patients, compared with healthy controls, over a 4-year follow-up period (J. Am. Acad. Dermatol. 2010;62[suppl. 1]:AB3).
These findings suggest that medical comorbidities associated with psoriasis accumulate over time; therefore, aggressive treatment of psoriasis in younger patients could improve their psychological and physical quality of life, Dr. Kimball said. Although there are no recommendations for additional health screening for psoriasis patients beyond the age-recommended preventive health measures, "younger patients especially need to be monitored for psychiatric issues," she said. And these patients should be kept up to date on vaccinations, particularly the annual flu vaccine and the human papillomavirus vaccine.
Dr. Kimball reported receiving grants, honoraria, consulting fees, and other support from Abbott, Amgen, and Janssen Pharmaceuticals.
RIO GRANDE, P.R. – Treating psoriasis patients earlier in life could help prevent later physical and psychological problems, according to Dr. Alexa B. Kimball.
"We used to think that we should save our therapies until our patients really needed them, because we were afraid that toxicity might accumulate," Dr. Kimball said at the annual Caribbean Dermatology Symposium. However, that thinking has changed, thanks in part to the availability of safer treatment options.
But of equal importance, "treating patients early in their disease may have an impact that affects the rest of their lives," including jobs, education, socioeconomic status, and curbing the development of health problems like obesity, cardiovascular disease, and psychiatric disorders, she said.
The quality of life issues associated with psoriasis are well known, but recent data confirm that physical and mental comorbidities start in childhood.
According to recent data from the National Psoriasis Foundation, 38% of children with psoriasis reported being bullied because of their condition, noted Dr. Kimball of Massachusetts General Hospital and Harvard Medical School, both in Boston.
Another study found that approximately one-third of children aged 4-17 years with psoriasis had a body mass index greater than the 95th percentile (N. Engl. J. Med. 2008;358:241-51). Conditions such as childhood obesity are not easily managed, and have significant implications for future health, she said.
In a retrospective study of 7,404 psoriasis patients younger than 18 years and 37,020 healthy controls, children with psoriasis were significantly more likely than controls to develop any psychiatric disorder (5% vs. 4%), depression (3% vs. 2%), and anxiety (2% vs. 1%), Dr. Kimball and her colleagues found (J. Am. Acad. Dermatol. 2012 Jan. 16 [doi:10.1016/j.jaad.2011.11.948]).
And the likelihood of comorbidities in psoriasis patients continues as they grow up, she said. "Chronic disease interacts with psychosocial and health events in a complex and ongoing manner throughout a person’s life."
Comorbidities in psoriasis patients appear to accumulate over time. Dr. Kimball cited data from the Nurses’ Health Study II, a cohort including more than 100,000 women who were aged 27-44 years in 1991. In a subset of 1,813 women with psoriasis, the risk of diabetes was approximately 60% higher, and the risk of hypertension was almost 20% higher, compared with women without psoriasis (Arch. Dermatol. 2009;145:379-82).
In a case-control study conducted by Dr. Kimball and her colleagues, cardiovascular disease, diabetes, depression, hyperlipidemia, hypertension, and obesity all increased significantly in psoriasis patients, compared with healthy controls, over a 4-year follow-up period (J. Am. Acad. Dermatol. 2010;62[suppl. 1]:AB3).
These findings suggest that medical comorbidities associated with psoriasis accumulate over time; therefore, aggressive treatment of psoriasis in younger patients could improve their psychological and physical quality of life, Dr. Kimball said. Although there are no recommendations for additional health screening for psoriasis patients beyond the age-recommended preventive health measures, "younger patients especially need to be monitored for psychiatric issues," she said. And these patients should be kept up to date on vaccinations, particularly the annual flu vaccine and the human papillomavirus vaccine.
Dr. Kimball reported receiving grants, honoraria, consulting fees, and other support from Abbott, Amgen, and Janssen Pharmaceuticals.
RIO GRANDE, P.R. – Treating psoriasis patients earlier in life could help prevent later physical and psychological problems, according to Dr. Alexa B. Kimball.
"We used to think that we should save our therapies until our patients really needed them, because we were afraid that toxicity might accumulate," Dr. Kimball said at the annual Caribbean Dermatology Symposium. However, that thinking has changed, thanks in part to the availability of safer treatment options.
But of equal importance, "treating patients early in their disease may have an impact that affects the rest of their lives," including jobs, education, socioeconomic status, and curbing the development of health problems like obesity, cardiovascular disease, and psychiatric disorders, she said.
The quality of life issues associated with psoriasis are well known, but recent data confirm that physical and mental comorbidities start in childhood.
According to recent data from the National Psoriasis Foundation, 38% of children with psoriasis reported being bullied because of their condition, noted Dr. Kimball of Massachusetts General Hospital and Harvard Medical School, both in Boston.
Another study found that approximately one-third of children aged 4-17 years with psoriasis had a body mass index greater than the 95th percentile (N. Engl. J. Med. 2008;358:241-51). Conditions such as childhood obesity are not easily managed, and have significant implications for future health, she said.
In a retrospective study of 7,404 psoriasis patients younger than 18 years and 37,020 healthy controls, children with psoriasis were significantly more likely than controls to develop any psychiatric disorder (5% vs. 4%), depression (3% vs. 2%), and anxiety (2% vs. 1%), Dr. Kimball and her colleagues found (J. Am. Acad. Dermatol. 2012 Jan. 16 [doi:10.1016/j.jaad.2011.11.948]).
And the likelihood of comorbidities in psoriasis patients continues as they grow up, she said. "Chronic disease interacts with psychosocial and health events in a complex and ongoing manner throughout a person’s life."
Comorbidities in psoriasis patients appear to accumulate over time. Dr. Kimball cited data from the Nurses’ Health Study II, a cohort including more than 100,000 women who were aged 27-44 years in 1991. In a subset of 1,813 women with psoriasis, the risk of diabetes was approximately 60% higher, and the risk of hypertension was almost 20% higher, compared with women without psoriasis (Arch. Dermatol. 2009;145:379-82).
In a case-control study conducted by Dr. Kimball and her colleagues, cardiovascular disease, diabetes, depression, hyperlipidemia, hypertension, and obesity all increased significantly in psoriasis patients, compared with healthy controls, over a 4-year follow-up period (J. Am. Acad. Dermatol. 2010;62[suppl. 1]:AB3).
These findings suggest that medical comorbidities associated with psoriasis accumulate over time; therefore, aggressive treatment of psoriasis in younger patients could improve their psychological and physical quality of life, Dr. Kimball said. Although there are no recommendations for additional health screening for psoriasis patients beyond the age-recommended preventive health measures, "younger patients especially need to be monitored for psychiatric issues," she said. And these patients should be kept up to date on vaccinations, particularly the annual flu vaccine and the human papillomavirus vaccine.
Dr. Kimball reported receiving grants, honoraria, consulting fees, and other support from Abbott, Amgen, and Janssen Pharmaceuticals.
EXPERT ANALYSIS FROM THE ANNUAL CARIBBEAN DERMATOLOGY SYMPOSIUM
Malignancy Rates Remain Stable in Ustekinumab-Treated Patients
RIO GRANDE, P.R. – Malignancy rates in psoriasis patients treated with ustekinumab did not increase significantly over 4 years of follow-up, based on pooled data from 3,117 patients enrolled in ustekinumab clinical trials.
Ustekinumab has shown effectiveness for treating moderate to severe psoriasis, but due to the potential of increased risk for cancer associated with its use, patients from several clinical trials (including PHOENIX I, PHOENIX II, and ACCEPT) are still being followed, said Dr. Kim A. Papp, director of research at Probity Medical Research, Waterloo, Ont., and colleagues.
The cumulative rates for nonmelanoma skin cancer in patients treated with ustekinumab for psoriasis remained low and stable throughout the follow-up period. A total of 0.57 cancer events per 100 person-years were reported in 2009 and 0.62 cancer events per 100 person-years were reported in 2010. The findings were presented at the annual Caribbean Dermatology Symposium.
In the complete analysis that included 6,791 patient-years of follow-up, 41 patients treated with any dose of ustekinumab developed at least one nonmelanoma skin cancer, and 3 patients developed both basal cell carcinoma and squamous cell carcinoma.
Another 42 patients developed at least one other malignancy, including 4 patients with melanoma in situ. However, no cases of invasive melanoma were observed during the study period. The other most common malignancies were prostate cancer (12 patients), colorectal cancer (4 patients) and breast cancer (3 patients).
By comparison, 39 individuals in the general population (based on the National Cancer Institute’s Surveillance, Epidemiology, and End Results database) developed at least one malignancy.
The findings were limited by the inclusion of several studies of varying lengths and by the inclusion criteria that can make comparison with the general population difficult, the researchers noted. The results suggest that rates of nonmelanoma skin cancer and other malignancies in psoriasis patients taking ustekinumab do not increase over time.
However, "additional long term data from clinical trials, observational registries, and postmarketing reporting databases will continue to define the ustekinumab malignancy risk profile," they wrote.
The study was sponsored by Centocor, which manufactures ustekinmab.
RIO GRANDE, P.R. – Malignancy rates in psoriasis patients treated with ustekinumab did not increase significantly over 4 years of follow-up, based on pooled data from 3,117 patients enrolled in ustekinumab clinical trials.
Ustekinumab has shown effectiveness for treating moderate to severe psoriasis, but due to the potential of increased risk for cancer associated with its use, patients from several clinical trials (including PHOENIX I, PHOENIX II, and ACCEPT) are still being followed, said Dr. Kim A. Papp, director of research at Probity Medical Research, Waterloo, Ont., and colleagues.
The cumulative rates for nonmelanoma skin cancer in patients treated with ustekinumab for psoriasis remained low and stable throughout the follow-up period. A total of 0.57 cancer events per 100 person-years were reported in 2009 and 0.62 cancer events per 100 person-years were reported in 2010. The findings were presented at the annual Caribbean Dermatology Symposium.
In the complete analysis that included 6,791 patient-years of follow-up, 41 patients treated with any dose of ustekinumab developed at least one nonmelanoma skin cancer, and 3 patients developed both basal cell carcinoma and squamous cell carcinoma.
Another 42 patients developed at least one other malignancy, including 4 patients with melanoma in situ. However, no cases of invasive melanoma were observed during the study period. The other most common malignancies were prostate cancer (12 patients), colorectal cancer (4 patients) and breast cancer (3 patients).
By comparison, 39 individuals in the general population (based on the National Cancer Institute’s Surveillance, Epidemiology, and End Results database) developed at least one malignancy.
The findings were limited by the inclusion of several studies of varying lengths and by the inclusion criteria that can make comparison with the general population difficult, the researchers noted. The results suggest that rates of nonmelanoma skin cancer and other malignancies in psoriasis patients taking ustekinumab do not increase over time.
However, "additional long term data from clinical trials, observational registries, and postmarketing reporting databases will continue to define the ustekinumab malignancy risk profile," they wrote.
The study was sponsored by Centocor, which manufactures ustekinmab.
RIO GRANDE, P.R. – Malignancy rates in psoriasis patients treated with ustekinumab did not increase significantly over 4 years of follow-up, based on pooled data from 3,117 patients enrolled in ustekinumab clinical trials.
Ustekinumab has shown effectiveness for treating moderate to severe psoriasis, but due to the potential of increased risk for cancer associated with its use, patients from several clinical trials (including PHOENIX I, PHOENIX II, and ACCEPT) are still being followed, said Dr. Kim A. Papp, director of research at Probity Medical Research, Waterloo, Ont., and colleagues.
The cumulative rates for nonmelanoma skin cancer in patients treated with ustekinumab for psoriasis remained low and stable throughout the follow-up period. A total of 0.57 cancer events per 100 person-years were reported in 2009 and 0.62 cancer events per 100 person-years were reported in 2010. The findings were presented at the annual Caribbean Dermatology Symposium.
In the complete analysis that included 6,791 patient-years of follow-up, 41 patients treated with any dose of ustekinumab developed at least one nonmelanoma skin cancer, and 3 patients developed both basal cell carcinoma and squamous cell carcinoma.
Another 42 patients developed at least one other malignancy, including 4 patients with melanoma in situ. However, no cases of invasive melanoma were observed during the study period. The other most common malignancies were prostate cancer (12 patients), colorectal cancer (4 patients) and breast cancer (3 patients).
By comparison, 39 individuals in the general population (based on the National Cancer Institute’s Surveillance, Epidemiology, and End Results database) developed at least one malignancy.
The findings were limited by the inclusion of several studies of varying lengths and by the inclusion criteria that can make comparison with the general population difficult, the researchers noted. The results suggest that rates of nonmelanoma skin cancer and other malignancies in psoriasis patients taking ustekinumab do not increase over time.
However, "additional long term data from clinical trials, observational registries, and postmarketing reporting databases will continue to define the ustekinumab malignancy risk profile," they wrote.
The study was sponsored by Centocor, which manufactures ustekinmab.
FROM THE ANNUAL CARIBBEAN DERMATOLOGY SYMPOSIUM
Major Finding: A total of 0.57 cancer events per 100 person-years were reported in 2009 and 0.62 cancer events per 100 person-years were reported in 2010.
Data Source: A review of 3,117 adults with psoriasis who were enrolled in ustekinumab clinical trials.
Disclosures: The study was sponsored by Centocor, which manufactures ustekinumab.
Infliximab Switch Clears Psoriasis in Etanercept Nonresponders
Switching to infliximab after an inadequate response to etanercept was associated with a Physician Global Assessment score of clear or minimal in nearly two-thirds of patients with plaque psoriasis, based on results of a multicenter, open-label study.
This approach "offers an alternative therapeutic paradigm for patients with psoriasis who do not respond to initial treatment with a TNF [tumor necrosis factor]-alpha antagonist or those who lose response during anti-TNF-alpha treatment," the researchers wrote in a report that appeared online Dec. 8, 2011, in the Journal of the American Academy of Dermatology (doi:10.1016/j.jaad.2011.10.020). "Additional studies are necessary to fully define the risks and benefits of transitions of this type for the treatment of psoriasis."
In an interview, lead study author Dr. Alice B. Gottlieb, professor and chair of dermatology at Tufts Medical Center, Boston, characterized the findings as important, "because many practitioners assume that if a patient does not respond to one TNF-alpha blocker – or if they have lost response to one TNF-alpha blocker – that another TNF-alpha blocker will not work. That’s just not true."
For the open-label study, known as PSUNRISE, Dr. Gottlieb and her associates at 49 centers in North America enrolled 215 adults with moderate to severe plaque psoriasis and an inadequate response to at least 4 months of treatment with etanercept (Enbrel). They were eligible for the trial if they had a Physician Global Assessment (PGA) score of at least 2 (mild) on a 5-point scale with etanercept, with or without concomitant oral systemic methotrexate or cyclosporine at baseline and during the study. Treatment consisted of intravenous infusions of infliximab (Remicade) 5 mg/kg at baseline and weeks 2, 6, 14, and 22.
The study’s primary end point was the proportion of patients who achieved a PGA score of clear (0) or minimal (1) at week 10. Secondary end points included the proportion of patients who achieved a PGA score of 0 or 1 at week 26. Adverse events were documented through week 30.
Of the 215 patients, 36 withdrew consent or terminated their participation, one of whom returned at week 30. This left 179 patients who completed the study and 180 who completed the 30-week follow-up. The mean age of patients was 44 years, 64% were male, and 90% were white.
By week 10, 65% of patients achieved a PGA of 0 or 1, a score that was maintained in 61% of patients at week 26. Overall, 54% of patients had at least two grades of improvement in their PGA score.
In addition, mean Psoriasis Area and Severity Index scores decreased from 11.6 at baseline to 2.8 at week 10 and week 26, while Dermatology Life Quality Index scores of 0 or 1 were achieved by 44% of patients at week 10 and 41% of patients at week 26.
No unexpected side effects or safety concerns were reported. Eight patients (4%) had a serious adverse event; no cases of tuberculosis were observed. "Overall, the type and incidence of adverse events observed after switching from etanercept to infliximab in this study were similar to those reported in large, controlled trials of infliximab in patients with moderate to severe psoriasis," the researchers wrote.
They acknowledged certain limitations of their study, including data bias "and uncontrolled systematic errors, that are not present in a blinded, controlled randomized study. In addition, it would be easier to interpret the study results if a comparator group were included in the study design."
Dr. Gottlieb, who reported the first double-blind, placebo-controlled study of infliximab monotherapy in psoriasis (Lancet 2001;357:1842-7), encouraged more dermatologists to add infliximab to their drug repertoire for severe psoriasis, "because it works in a high percentage of patients, it works quickly, and it can work when other TNF-alpha blockers don’t."
The study was supported by Janssen Biotech, which manufactures Remicade and is a subsidiary of Johnson & Johnson. Dr. Gottlieb disclosed that she has current consulting/advisory board agreements with several pharmaceutical companies, including Amgen and Pfizer, which market Enbrel, but not with Janssen Biotech. Two of the study authors, Dr. Marc Chevrier and Stephen Calabro, are employees of Janssen Biotech.
Switching to infliximab after an inadequate response to etanercept was associated with a Physician Global Assessment score of clear or minimal in nearly two-thirds of patients with plaque psoriasis, based on results of a multicenter, open-label study.
This approach "offers an alternative therapeutic paradigm for patients with psoriasis who do not respond to initial treatment with a TNF [tumor necrosis factor]-alpha antagonist or those who lose response during anti-TNF-alpha treatment," the researchers wrote in a report that appeared online Dec. 8, 2011, in the Journal of the American Academy of Dermatology (doi:10.1016/j.jaad.2011.10.020). "Additional studies are necessary to fully define the risks and benefits of transitions of this type for the treatment of psoriasis."
In an interview, lead study author Dr. Alice B. Gottlieb, professor and chair of dermatology at Tufts Medical Center, Boston, characterized the findings as important, "because many practitioners assume that if a patient does not respond to one TNF-alpha blocker – or if they have lost response to one TNF-alpha blocker – that another TNF-alpha blocker will not work. That’s just not true."
For the open-label study, known as PSUNRISE, Dr. Gottlieb and her associates at 49 centers in North America enrolled 215 adults with moderate to severe plaque psoriasis and an inadequate response to at least 4 months of treatment with etanercept (Enbrel). They were eligible for the trial if they had a Physician Global Assessment (PGA) score of at least 2 (mild) on a 5-point scale with etanercept, with or without concomitant oral systemic methotrexate or cyclosporine at baseline and during the study. Treatment consisted of intravenous infusions of infliximab (Remicade) 5 mg/kg at baseline and weeks 2, 6, 14, and 22.
The study’s primary end point was the proportion of patients who achieved a PGA score of clear (0) or minimal (1) at week 10. Secondary end points included the proportion of patients who achieved a PGA score of 0 or 1 at week 26. Adverse events were documented through week 30.
Of the 215 patients, 36 withdrew consent or terminated their participation, one of whom returned at week 30. This left 179 patients who completed the study and 180 who completed the 30-week follow-up. The mean age of patients was 44 years, 64% were male, and 90% were white.
By week 10, 65% of patients achieved a PGA of 0 or 1, a score that was maintained in 61% of patients at week 26. Overall, 54% of patients had at least two grades of improvement in their PGA score.
In addition, mean Psoriasis Area and Severity Index scores decreased from 11.6 at baseline to 2.8 at week 10 and week 26, while Dermatology Life Quality Index scores of 0 or 1 were achieved by 44% of patients at week 10 and 41% of patients at week 26.
No unexpected side effects or safety concerns were reported. Eight patients (4%) had a serious adverse event; no cases of tuberculosis were observed. "Overall, the type and incidence of adverse events observed after switching from etanercept to infliximab in this study were similar to those reported in large, controlled trials of infliximab in patients with moderate to severe psoriasis," the researchers wrote.
They acknowledged certain limitations of their study, including data bias "and uncontrolled systematic errors, that are not present in a blinded, controlled randomized study. In addition, it would be easier to interpret the study results if a comparator group were included in the study design."
Dr. Gottlieb, who reported the first double-blind, placebo-controlled study of infliximab monotherapy in psoriasis (Lancet 2001;357:1842-7), encouraged more dermatologists to add infliximab to their drug repertoire for severe psoriasis, "because it works in a high percentage of patients, it works quickly, and it can work when other TNF-alpha blockers don’t."
The study was supported by Janssen Biotech, which manufactures Remicade and is a subsidiary of Johnson & Johnson. Dr. Gottlieb disclosed that she has current consulting/advisory board agreements with several pharmaceutical companies, including Amgen and Pfizer, which market Enbrel, but not with Janssen Biotech. Two of the study authors, Dr. Marc Chevrier and Stephen Calabro, are employees of Janssen Biotech.
Switching to infliximab after an inadequate response to etanercept was associated with a Physician Global Assessment score of clear or minimal in nearly two-thirds of patients with plaque psoriasis, based on results of a multicenter, open-label study.
This approach "offers an alternative therapeutic paradigm for patients with psoriasis who do not respond to initial treatment with a TNF [tumor necrosis factor]-alpha antagonist or those who lose response during anti-TNF-alpha treatment," the researchers wrote in a report that appeared online Dec. 8, 2011, in the Journal of the American Academy of Dermatology (doi:10.1016/j.jaad.2011.10.020). "Additional studies are necessary to fully define the risks and benefits of transitions of this type for the treatment of psoriasis."
In an interview, lead study author Dr. Alice B. Gottlieb, professor and chair of dermatology at Tufts Medical Center, Boston, characterized the findings as important, "because many practitioners assume that if a patient does not respond to one TNF-alpha blocker – or if they have lost response to one TNF-alpha blocker – that another TNF-alpha blocker will not work. That’s just not true."
For the open-label study, known as PSUNRISE, Dr. Gottlieb and her associates at 49 centers in North America enrolled 215 adults with moderate to severe plaque psoriasis and an inadequate response to at least 4 months of treatment with etanercept (Enbrel). They were eligible for the trial if they had a Physician Global Assessment (PGA) score of at least 2 (mild) on a 5-point scale with etanercept, with or without concomitant oral systemic methotrexate or cyclosporine at baseline and during the study. Treatment consisted of intravenous infusions of infliximab (Remicade) 5 mg/kg at baseline and weeks 2, 6, 14, and 22.
The study’s primary end point was the proportion of patients who achieved a PGA score of clear (0) or minimal (1) at week 10. Secondary end points included the proportion of patients who achieved a PGA score of 0 or 1 at week 26. Adverse events were documented through week 30.
Of the 215 patients, 36 withdrew consent or terminated their participation, one of whom returned at week 30. This left 179 patients who completed the study and 180 who completed the 30-week follow-up. The mean age of patients was 44 years, 64% were male, and 90% were white.
By week 10, 65% of patients achieved a PGA of 0 or 1, a score that was maintained in 61% of patients at week 26. Overall, 54% of patients had at least two grades of improvement in their PGA score.
In addition, mean Psoriasis Area and Severity Index scores decreased from 11.6 at baseline to 2.8 at week 10 and week 26, while Dermatology Life Quality Index scores of 0 or 1 were achieved by 44% of patients at week 10 and 41% of patients at week 26.
No unexpected side effects or safety concerns were reported. Eight patients (4%) had a serious adverse event; no cases of tuberculosis were observed. "Overall, the type and incidence of adverse events observed after switching from etanercept to infliximab in this study were similar to those reported in large, controlled trials of infliximab in patients with moderate to severe psoriasis," the researchers wrote.
They acknowledged certain limitations of their study, including data bias "and uncontrolled systematic errors, that are not present in a blinded, controlled randomized study. In addition, it would be easier to interpret the study results if a comparator group were included in the study design."
Dr. Gottlieb, who reported the first double-blind, placebo-controlled study of infliximab monotherapy in psoriasis (Lancet 2001;357:1842-7), encouraged more dermatologists to add infliximab to their drug repertoire for severe psoriasis, "because it works in a high percentage of patients, it works quickly, and it can work when other TNF-alpha blockers don’t."
The study was supported by Janssen Biotech, which manufactures Remicade and is a subsidiary of Johnson & Johnson. Dr. Gottlieb disclosed that she has current consulting/advisory board agreements with several pharmaceutical companies, including Amgen and Pfizer, which market Enbrel, but not with Janssen Biotech. Two of the study authors, Dr. Marc Chevrier and Stephen Calabro, are employees of Janssen Biotech.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Major Finding: After switching to infliximab, 65% of patients with plaque psoriasis had a Physician Global Assessment score of clear (0) or minimal (1) at week 10. This response was durable through week 26.
Data Source: An open-label, multicenter study of 215 patients with plaque psoriasis who had an inadequate response to etanercept and who received intravenous infusions of infliximab 5 mg/kg at weeks 0, 2, 6, 14, and 22.
Disclosures: Dr. Gottlieb disclosed that she has current consulting/advisory board agreements with several pharmaceutical companies, including Amgen and Pfizer, but not with Janssen Biotech. Two of the study authors, Dr. Marc Chevrier and Stephen Calabro, are employees of Janssen Biotech.
Ondansetron Quells Methotrexate-Induced Nausea
LISBON – Getting an earful of complaints of nausea from your patients on oral methotrexate therapy?
Consider bracketing the antifolate drug with the antiemetic ondansetron before and afterward, Dr. Richard B. Warren suggested at the congress.
"One successful means of helping patients overcome their nausea that we’ve used a lot in Manchester is ondansetron. For some people it can literally sort out the nausea altogether," said Dr. Warren, senior lecturer and honorary consultant dermatologist at the University of Manchester (England).
The dosing is 8 mg of ondansetron (Zofran) – a serotonin receptor antagonist used to treat nausea caused by chemotherapy, radiation therapy, and/or surgery – administered 2 hours before and once again 12 hours after taking methotrexate.
Nausea is a common nuisance side effect of the drug. It affects roughly one-quarter of patients, typically beginning about 12 hours after they take their weekly dose. The nausea can last for up to 72 hours.
In addition to prophylactic ondansetron, other strategies that may be effective in overcoming methotrexate-induced nausea include the following:
– Divide the dose.
– Have patients take their oral methotrexate with the evening meal.
– Consider switching to subcutaneous methotrexate. "It has been shown to be more efficacious, and with less in the way of adverse events than oral methotrexate," Dr. Warren said.
He declared having no relevant financial interests.
LISBON – Getting an earful of complaints of nausea from your patients on oral methotrexate therapy?
Consider bracketing the antifolate drug with the antiemetic ondansetron before and afterward, Dr. Richard B. Warren suggested at the congress.
"One successful means of helping patients overcome their nausea that we’ve used a lot in Manchester is ondansetron. For some people it can literally sort out the nausea altogether," said Dr. Warren, senior lecturer and honorary consultant dermatologist at the University of Manchester (England).
The dosing is 8 mg of ondansetron (Zofran) – a serotonin receptor antagonist used to treat nausea caused by chemotherapy, radiation therapy, and/or surgery – administered 2 hours before and once again 12 hours after taking methotrexate.
Nausea is a common nuisance side effect of the drug. It affects roughly one-quarter of patients, typically beginning about 12 hours after they take their weekly dose. The nausea can last for up to 72 hours.
In addition to prophylactic ondansetron, other strategies that may be effective in overcoming methotrexate-induced nausea include the following:
– Divide the dose.
– Have patients take their oral methotrexate with the evening meal.
– Consider switching to subcutaneous methotrexate. "It has been shown to be more efficacious, and with less in the way of adverse events than oral methotrexate," Dr. Warren said.
He declared having no relevant financial interests.
LISBON – Getting an earful of complaints of nausea from your patients on oral methotrexate therapy?
Consider bracketing the antifolate drug with the antiemetic ondansetron before and afterward, Dr. Richard B. Warren suggested at the congress.
"One successful means of helping patients overcome their nausea that we’ve used a lot in Manchester is ondansetron. For some people it can literally sort out the nausea altogether," said Dr. Warren, senior lecturer and honorary consultant dermatologist at the University of Manchester (England).
The dosing is 8 mg of ondansetron (Zofran) – a serotonin receptor antagonist used to treat nausea caused by chemotherapy, radiation therapy, and/or surgery – administered 2 hours before and once again 12 hours after taking methotrexate.
Nausea is a common nuisance side effect of the drug. It affects roughly one-quarter of patients, typically beginning about 12 hours after they take their weekly dose. The nausea can last for up to 72 hours.
In addition to prophylactic ondansetron, other strategies that may be effective in overcoming methotrexate-induced nausea include the following:
– Divide the dose.
– Have patients take their oral methotrexate with the evening meal.
– Consider switching to subcutaneous methotrexate. "It has been shown to be more efficacious, and with less in the way of adverse events than oral methotrexate," Dr. Warren said.
He declared having no relevant financial interests.
EXPERT OPINION FROM THE ANNUAL CONGRESS OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY