User login
Ustekinumab Re-treatment Delivers Excellent Results
LISBON – Roughly 85% of psoriasis patients re-treated with ustekinumab after withdrawing from an earlier successful course of therapy will experience an excellent response in round two.
This benefit was consistently seen in secondary analyses of two separate phase III clinical trials. This finding provides information that is valuable in clinical practice, Dr. Christopher E. Griffiths said at the annual congress of the European Academy of Dermatology and Venereology.
Psoriasis patients often have to halt effective biologic therapy for a variety of reasons – pregnancy, elective major surgery, vaccinations, or just to enjoy a welcome break from potent therapy while their disease has cleared – but not all biologics retain their efficacy upon re-treatment.
Ustekinumab (Stelara) now joins etanercept (Enbrel) and alefacept (Amevive) on the list of biologic therapies that are able to recapture their clinical response upon re-treatment, said Dr. Griffiths, professor of dermatology at the University of Manchester (England).
He reported on 260 ustekinumab-treated patients in the phase-III PHOENIX I trial and 375 in the ACCEPT trial, all of whom attained at least a 75% improvement, compared with baseline, in Psoriasis Area and Severity Index (PASI) scores, and a Physician Global Assessment (PGA) score of 0-2, meaning clear, minimal, or mild disease. The study patients were withdrawn from the biologic at week 40 in PHOENIX I and at week 12 in ACCEPT.
The PHOENIX I participants began re-treatment after losing at least 50% of the improvement obtained during the initial course of therapy. At week 12 of re-treatment, 84% of patients from PHOENIX I who were on the ustekinumab 45 mg dose and 85% on ustekinumab 90 mg achieved a PASI 75 response. The median improvement in PASI scores, compared with when they commenced re-treatment, was 89%-90%.
ACCEPT participants entered re-treatment after their PGA score, which was 0-2 upon treatment withdrawal, regressed to greater than 2.
Among ACCEPT participants undergoing re-treatment with ustekinumab 45 mg after earlier ustekinumab withdrawal, 85% were rated as having a 0-2 score on PGA at week 12. So were 89% on ustekinumab 90 mg. The median improvement in PASI scores from baseline was 85% in the ustekinumab 45 mg group and 90% in the ustekinumab 90 mg arm.
Rates of overall adverse events, serious adverse events, and infections were similar in the initial round of treatment and in re-treatment. No new safety signals arose during 12 weeks of re-treatment. Five patients being re-treated with ustekinumab 45 mg and two on ustekinumab 90 mg developed anti-ustekinumab antibodies; their response to re-treatment was significantly less robust than in patients without antibodies, Dr. Griffiths noted.
The study was funded by Centocor, the manufacturer of ustekinumab. Dr. Griffiths is an advisor to the company.
LISBON – Roughly 85% of psoriasis patients re-treated with ustekinumab after withdrawing from an earlier successful course of therapy will experience an excellent response in round two.
This benefit was consistently seen in secondary analyses of two separate phase III clinical trials. This finding provides information that is valuable in clinical practice, Dr. Christopher E. Griffiths said at the annual congress of the European Academy of Dermatology and Venereology.
Psoriasis patients often have to halt effective biologic therapy for a variety of reasons – pregnancy, elective major surgery, vaccinations, or just to enjoy a welcome break from potent therapy while their disease has cleared – but not all biologics retain their efficacy upon re-treatment.
Ustekinumab (Stelara) now joins etanercept (Enbrel) and alefacept (Amevive) on the list of biologic therapies that are able to recapture their clinical response upon re-treatment, said Dr. Griffiths, professor of dermatology at the University of Manchester (England).
He reported on 260 ustekinumab-treated patients in the phase-III PHOENIX I trial and 375 in the ACCEPT trial, all of whom attained at least a 75% improvement, compared with baseline, in Psoriasis Area and Severity Index (PASI) scores, and a Physician Global Assessment (PGA) score of 0-2, meaning clear, minimal, or mild disease. The study patients were withdrawn from the biologic at week 40 in PHOENIX I and at week 12 in ACCEPT.
The PHOENIX I participants began re-treatment after losing at least 50% of the improvement obtained during the initial course of therapy. At week 12 of re-treatment, 84% of patients from PHOENIX I who were on the ustekinumab 45 mg dose and 85% on ustekinumab 90 mg achieved a PASI 75 response. The median improvement in PASI scores, compared with when they commenced re-treatment, was 89%-90%.
ACCEPT participants entered re-treatment after their PGA score, which was 0-2 upon treatment withdrawal, regressed to greater than 2.
Among ACCEPT participants undergoing re-treatment with ustekinumab 45 mg after earlier ustekinumab withdrawal, 85% were rated as having a 0-2 score on PGA at week 12. So were 89% on ustekinumab 90 mg. The median improvement in PASI scores from baseline was 85% in the ustekinumab 45 mg group and 90% in the ustekinumab 90 mg arm.
Rates of overall adverse events, serious adverse events, and infections were similar in the initial round of treatment and in re-treatment. No new safety signals arose during 12 weeks of re-treatment. Five patients being re-treated with ustekinumab 45 mg and two on ustekinumab 90 mg developed anti-ustekinumab antibodies; their response to re-treatment was significantly less robust than in patients without antibodies, Dr. Griffiths noted.
The study was funded by Centocor, the manufacturer of ustekinumab. Dr. Griffiths is an advisor to the company.
LISBON – Roughly 85% of psoriasis patients re-treated with ustekinumab after withdrawing from an earlier successful course of therapy will experience an excellent response in round two.
This benefit was consistently seen in secondary analyses of two separate phase III clinical trials. This finding provides information that is valuable in clinical practice, Dr. Christopher E. Griffiths said at the annual congress of the European Academy of Dermatology and Venereology.
Psoriasis patients often have to halt effective biologic therapy for a variety of reasons – pregnancy, elective major surgery, vaccinations, or just to enjoy a welcome break from potent therapy while their disease has cleared – but not all biologics retain their efficacy upon re-treatment.
Ustekinumab (Stelara) now joins etanercept (Enbrel) and alefacept (Amevive) on the list of biologic therapies that are able to recapture their clinical response upon re-treatment, said Dr. Griffiths, professor of dermatology at the University of Manchester (England).
He reported on 260 ustekinumab-treated patients in the phase-III PHOENIX I trial and 375 in the ACCEPT trial, all of whom attained at least a 75% improvement, compared with baseline, in Psoriasis Area and Severity Index (PASI) scores, and a Physician Global Assessment (PGA) score of 0-2, meaning clear, minimal, or mild disease. The study patients were withdrawn from the biologic at week 40 in PHOENIX I and at week 12 in ACCEPT.
The PHOENIX I participants began re-treatment after losing at least 50% of the improvement obtained during the initial course of therapy. At week 12 of re-treatment, 84% of patients from PHOENIX I who were on the ustekinumab 45 mg dose and 85% on ustekinumab 90 mg achieved a PASI 75 response. The median improvement in PASI scores, compared with when they commenced re-treatment, was 89%-90%.
ACCEPT participants entered re-treatment after their PGA score, which was 0-2 upon treatment withdrawal, regressed to greater than 2.
Among ACCEPT participants undergoing re-treatment with ustekinumab 45 mg after earlier ustekinumab withdrawal, 85% were rated as having a 0-2 score on PGA at week 12. So were 89% on ustekinumab 90 mg. The median improvement in PASI scores from baseline was 85% in the ustekinumab 45 mg group and 90% in the ustekinumab 90 mg arm.
Rates of overall adverse events, serious adverse events, and infections were similar in the initial round of treatment and in re-treatment. No new safety signals arose during 12 weeks of re-treatment. Five patients being re-treated with ustekinumab 45 mg and two on ustekinumab 90 mg developed anti-ustekinumab antibodies; their response to re-treatment was significantly less robust than in patients without antibodies, Dr. Griffiths noted.
The study was funded by Centocor, the manufacturer of ustekinumab. Dr. Griffiths is an advisor to the company.
FROM THE ANNUAL CONGRESS OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Major Finding: At week 12 of re-treatment, 84% of patients from PHOENIX I who were on the ustekinumab 45 mg dose and 85% on ustekinumab 90 mg achieved a PASI 75 response.
Data Source: Secondary analysis of the phase III PHOENIX 1 and ACCEPT randomized trials.
Disclosures: The study was funded by Centocor, manufacturer of ustekinumab. Dr. Griffiths is an advisor to the company.
Mycophenolate Better Than Azathioprine for Lupus Nephritis Maintenance Therapy
Mycophenolate mofetil was more effective than azathioprine in maintaining renal response and preventing relapse in patients with active lupus nephritis, judging from the findings of a phase III clinical trial reported in the Nov. 17 issue of the New England Journal of Medicine.
Compared with azathioprine maintenance therapy, the use of mycophenolate mofetil prolonged the time to treatment failure, which was the study’s primary end point. It also extended the interval until individual components of this end point were reached, including a renal flare, the need for rescue therapy, sustained doubling time of the serum creatinine level, and the development of end-stage renal disease (ESRD), said Dr. Mary Anne Dooley of the University of North Carolina at Chapel Hill and her associates in the manufacturer-sponsored Aspreva Lupus Management Study (ALMS).
The investigators previously reported the results of the induction phase of ALMS, in which the efficacy and safety of mycophenolate mofetil were compared with that of intravenous cyclophosphamide as induction therapy for lupus nephritis. This latest report covers the results of the 3-year maintenance phase of ALMS, a prospective, double-blind trial comparing oral mycophenolate mofetil against oral azathioprine for maintenance of remission among the study subjects who responded to either induction therapy.
The 227 study subjects were aged 12-75 years at baseline and had class III, IV, or V lupus nephritis. They were enrolled at medical centers in Asia, Latin America, North America, Europe, South Africa, and Australia and were randomly assigned to receive mycophenolate mofetil (116 patients) or azathioprine (111 patients).
A total of 127 subjects (55.9% of the mycophenolate group and 48.6% of the azathioprine group) completed 36 months of treatment.
"Mycophenolate mofetil was significantly superior to azathioprine with respect to the primary end point, the time to treatment failure," and overall rates of treatment failure were 16.4% with mycophenolate, compared with 32.4% with azathioprine, regardless of which type of induction therapy had been used or where patients resided, Dr. Dooley and her colleagues said (N. Engl. J. Med. 2011;365:1886-95).
"Mycophenolate mofetil was significantly superior to azathioprine."
Among subjects who received mycophenolate mofetil maintenance, renal flares developed in 12.9%, rescue therapy was required in 7.8%, and a doubling of the serum creatinine level was reached in 0.9%. Among subjects who received azathioprine maintenance, renal flares developed in nearly twice as many (23.4%), rescue therapy was required in more than twice as many (17.1%), and a doubling of the serum creatinine level was reached in five times as many (4.5%).
Three patients on azathioprine developed ESRD, compared with none of those taking mycophenolate mofetil.
The overall incidence of adverse events was similar between the two groups, and infections were the most common adverse events in both. The rates of serious infection were low in both groups, at 9.6% with mycophenolate mofetil and 11.7% with azathioprine.
The percentage of patients who withdrew from treatment because of adverse effects was higher with azathioprine (39.6%) than with mycophenolate mofetil (25.2%).
"Although our trial included more patients and was substantially longer than many of the controlled trials involving patients with lupus nephritis, potential outcomes that might appear more frequently after 5-20 years [such as cardiovascular complications and ESRD] cannot be determined, since no further follow-up study is planned," they noted.
"The length of time that mycophenolate mofetil needs to be continued is unknown; hence, improved biomarkers of response are needed to distinguish disease remission from remission that occurs while the patient is receiving treatment," they added.
It is important to note that this maintenance study included only patients who responded well to induction therapy. Thus, the results may not apply to those whose disease is more difficult to treat, Dr. Dooley and her associates said.
The ALMS was supported by Vifor Pharma (formerly Aspreva Pharmaceuticals) as part of the Roche-Aspreva rare diseases collaboration, and by Caudex Medical.
Mycophenolate mofetil was more effective than azathioprine in maintaining renal response and preventing relapse in patients with active lupus nephritis, judging from the findings of a phase III clinical trial reported in the Nov. 17 issue of the New England Journal of Medicine.
Compared with azathioprine maintenance therapy, the use of mycophenolate mofetil prolonged the time to treatment failure, which was the study’s primary end point. It also extended the interval until individual components of this end point were reached, including a renal flare, the need for rescue therapy, sustained doubling time of the serum creatinine level, and the development of end-stage renal disease (ESRD), said Dr. Mary Anne Dooley of the University of North Carolina at Chapel Hill and her associates in the manufacturer-sponsored Aspreva Lupus Management Study (ALMS).
The investigators previously reported the results of the induction phase of ALMS, in which the efficacy and safety of mycophenolate mofetil were compared with that of intravenous cyclophosphamide as induction therapy for lupus nephritis. This latest report covers the results of the 3-year maintenance phase of ALMS, a prospective, double-blind trial comparing oral mycophenolate mofetil against oral azathioprine for maintenance of remission among the study subjects who responded to either induction therapy.
The 227 study subjects were aged 12-75 years at baseline and had class III, IV, or V lupus nephritis. They were enrolled at medical centers in Asia, Latin America, North America, Europe, South Africa, and Australia and were randomly assigned to receive mycophenolate mofetil (116 patients) or azathioprine (111 patients).
A total of 127 subjects (55.9% of the mycophenolate group and 48.6% of the azathioprine group) completed 36 months of treatment.
"Mycophenolate mofetil was significantly superior to azathioprine with respect to the primary end point, the time to treatment failure," and overall rates of treatment failure were 16.4% with mycophenolate, compared with 32.4% with azathioprine, regardless of which type of induction therapy had been used or where patients resided, Dr. Dooley and her colleagues said (N. Engl. J. Med. 2011;365:1886-95).
"Mycophenolate mofetil was significantly superior to azathioprine."
Among subjects who received mycophenolate mofetil maintenance, renal flares developed in 12.9%, rescue therapy was required in 7.8%, and a doubling of the serum creatinine level was reached in 0.9%. Among subjects who received azathioprine maintenance, renal flares developed in nearly twice as many (23.4%), rescue therapy was required in more than twice as many (17.1%), and a doubling of the serum creatinine level was reached in five times as many (4.5%).
Three patients on azathioprine developed ESRD, compared with none of those taking mycophenolate mofetil.
The overall incidence of adverse events was similar between the two groups, and infections were the most common adverse events in both. The rates of serious infection were low in both groups, at 9.6% with mycophenolate mofetil and 11.7% with azathioprine.
The percentage of patients who withdrew from treatment because of adverse effects was higher with azathioprine (39.6%) than with mycophenolate mofetil (25.2%).
"Although our trial included more patients and was substantially longer than many of the controlled trials involving patients with lupus nephritis, potential outcomes that might appear more frequently after 5-20 years [such as cardiovascular complications and ESRD] cannot be determined, since no further follow-up study is planned," they noted.
"The length of time that mycophenolate mofetil needs to be continued is unknown; hence, improved biomarkers of response are needed to distinguish disease remission from remission that occurs while the patient is receiving treatment," they added.
It is important to note that this maintenance study included only patients who responded well to induction therapy. Thus, the results may not apply to those whose disease is more difficult to treat, Dr. Dooley and her associates said.
The ALMS was supported by Vifor Pharma (formerly Aspreva Pharmaceuticals) as part of the Roche-Aspreva rare diseases collaboration, and by Caudex Medical.
Mycophenolate mofetil was more effective than azathioprine in maintaining renal response and preventing relapse in patients with active lupus nephritis, judging from the findings of a phase III clinical trial reported in the Nov. 17 issue of the New England Journal of Medicine.
Compared with azathioprine maintenance therapy, the use of mycophenolate mofetil prolonged the time to treatment failure, which was the study’s primary end point. It also extended the interval until individual components of this end point were reached, including a renal flare, the need for rescue therapy, sustained doubling time of the serum creatinine level, and the development of end-stage renal disease (ESRD), said Dr. Mary Anne Dooley of the University of North Carolina at Chapel Hill and her associates in the manufacturer-sponsored Aspreva Lupus Management Study (ALMS).
The investigators previously reported the results of the induction phase of ALMS, in which the efficacy and safety of mycophenolate mofetil were compared with that of intravenous cyclophosphamide as induction therapy for lupus nephritis. This latest report covers the results of the 3-year maintenance phase of ALMS, a prospective, double-blind trial comparing oral mycophenolate mofetil against oral azathioprine for maintenance of remission among the study subjects who responded to either induction therapy.
The 227 study subjects were aged 12-75 years at baseline and had class III, IV, or V lupus nephritis. They were enrolled at medical centers in Asia, Latin America, North America, Europe, South Africa, and Australia and were randomly assigned to receive mycophenolate mofetil (116 patients) or azathioprine (111 patients).
A total of 127 subjects (55.9% of the mycophenolate group and 48.6% of the azathioprine group) completed 36 months of treatment.
"Mycophenolate mofetil was significantly superior to azathioprine with respect to the primary end point, the time to treatment failure," and overall rates of treatment failure were 16.4% with mycophenolate, compared with 32.4% with azathioprine, regardless of which type of induction therapy had been used or where patients resided, Dr. Dooley and her colleagues said (N. Engl. J. Med. 2011;365:1886-95).
"Mycophenolate mofetil was significantly superior to azathioprine."
Among subjects who received mycophenolate mofetil maintenance, renal flares developed in 12.9%, rescue therapy was required in 7.8%, and a doubling of the serum creatinine level was reached in 0.9%. Among subjects who received azathioprine maintenance, renal flares developed in nearly twice as many (23.4%), rescue therapy was required in more than twice as many (17.1%), and a doubling of the serum creatinine level was reached in five times as many (4.5%).
Three patients on azathioprine developed ESRD, compared with none of those taking mycophenolate mofetil.
The overall incidence of adverse events was similar between the two groups, and infections were the most common adverse events in both. The rates of serious infection were low in both groups, at 9.6% with mycophenolate mofetil and 11.7% with azathioprine.
The percentage of patients who withdrew from treatment because of adverse effects was higher with azathioprine (39.6%) than with mycophenolate mofetil (25.2%).
"Although our trial included more patients and was substantially longer than many of the controlled trials involving patients with lupus nephritis, potential outcomes that might appear more frequently after 5-20 years [such as cardiovascular complications and ESRD] cannot be determined, since no further follow-up study is planned," they noted.
"The length of time that mycophenolate mofetil needs to be continued is unknown; hence, improved biomarkers of response are needed to distinguish disease remission from remission that occurs while the patient is receiving treatment," they added.
It is important to note that this maintenance study included only patients who responded well to induction therapy. Thus, the results may not apply to those whose disease is more difficult to treat, Dr. Dooley and her associates said.
The ALMS was supported by Vifor Pharma (formerly Aspreva Pharmaceuticals) as part of the Roche-Aspreva rare diseases collaboration, and by Caudex Medical.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: Mycophenolate mofetil was superior to azathioprine in rates of treatment failure (16.4% vs. 32.4%), renal flares (12.9% vs. 23.4%), need for rescue therapy (7.8% vs. 17.1%), doubling of serum creatinine level (0.9% vs. 4.5%), and development of ESRD (0% vs. 3%).
Data Source: The maintenance phase of an international, randomized, double-blind, phase III clinical trial involving 227 patients aged 12-75 years who had lupus nephritis and were followed for 3 years.
Disclosures: The ALMS was supported by Vifor Pharma (formerly Aspreva Pharmaceuticals) as part of the Roche-Aspreva rare diseases collaboration, and by Caudex Medical.
When Vitiligo and Psoriasis Coexist, Keep Looking
LISBON – Comorbid vitiligo and psoriasis is a red flag signaling the need to dig deeper looking for potentially associated diseases, including cardiovascular, autoimmune, or psychiatric diagnoses.
A study of 154 Italian vitiligo patients showed that a family history of cardiovascular disease was present in none of 54 participants with vitiligo alone compared to one-third of the 19 patients with both vitiligo and psoriasis, Dr. Silvia Moretti reported at the annual congress of the European Academy of Dermatology and Venereology.
Subjects with strong genetic loading for either or both of the skin diseases also tended to have an increased prevalence of a positive family history of cardiovascular disease, although this trend did not reach statistical significance. For example, a family history of cardiovascular disease was present in 10% of 52 patients with vitiligo and a family history of vitiligo, 13% of 15 patients with vitiligo as well as a family history of both vitiligo and psoriasis, and 18% of the 16 patients with vitiligo and a family history of psoriasis, according to Dr. Moretti of the University of Florence.
Similarly, a strong family history of hypertension was present in 4% of vitiligo patients, 8% of those with both vitiligo and a family history of vitiligo, 13% with vitiligo and a family history of both psoriasis and vitiligo, 25% of vitiligo patients with a family history of psoriasis, and 26% of patients carrying diagnoses of both vitiligo and psoriasis but no family history of either dermatologic disease. In only the group with both vitiligo and psoriasis was a family history of hypertension significantly more common than in patients with vitiligo alone.
The investigators also checked for a grab bag of comorbid, nondermatologic diseases they suspect might be genetically linked to vitiligo and psoriasis, including lupus, celiac disease, megaloblastic anemia, and allergic rhinitis. Vitiligo patients with a family history of both vitiligo and psoriasis had a 47% prevalence of one or more of these conditions, significantly greater than in the other subgroups, where the prevalence ranged from 11% to 37%. In patients with comorbid vitiligo and psoriasis but no family history of either skin disease, the rate was 21%.
The prevalence of a diagnosed psychiatric disorder was zero in patients with vitiligo with or without a family history of vitiligo, 7% in those with vitiligo and a family history of both vitiligo and psoriasis, 6% in those with vitiligo and a family history of psoriasis, and 5% in patients with comorbid vitiligo and psoriasis.
Seventy-two percent of patients with both vitiligo and psoriasis were Fitzpatrick phototype 2 and the rest were phototype 3. Seventy-nine percent of those with vitiligo and a family history of both psoriasis and vitiligo were phototype 2, as were 56% of vitiligo patients with a family history of psoriasis.
In contrast, 60% of patients with only vitiligo were phototype 3 and 40% were phototype 2. The same 60/40 split held for patients with vitiligo and a family history of vitiligo.
Dr. Moretti declared having no financial conflicts of interest.
LISBON – Comorbid vitiligo and psoriasis is a red flag signaling the need to dig deeper looking for potentially associated diseases, including cardiovascular, autoimmune, or psychiatric diagnoses.
A study of 154 Italian vitiligo patients showed that a family history of cardiovascular disease was present in none of 54 participants with vitiligo alone compared to one-third of the 19 patients with both vitiligo and psoriasis, Dr. Silvia Moretti reported at the annual congress of the European Academy of Dermatology and Venereology.
Subjects with strong genetic loading for either or both of the skin diseases also tended to have an increased prevalence of a positive family history of cardiovascular disease, although this trend did not reach statistical significance. For example, a family history of cardiovascular disease was present in 10% of 52 patients with vitiligo and a family history of vitiligo, 13% of 15 patients with vitiligo as well as a family history of both vitiligo and psoriasis, and 18% of the 16 patients with vitiligo and a family history of psoriasis, according to Dr. Moretti of the University of Florence.
Similarly, a strong family history of hypertension was present in 4% of vitiligo patients, 8% of those with both vitiligo and a family history of vitiligo, 13% with vitiligo and a family history of both psoriasis and vitiligo, 25% of vitiligo patients with a family history of psoriasis, and 26% of patients carrying diagnoses of both vitiligo and psoriasis but no family history of either dermatologic disease. In only the group with both vitiligo and psoriasis was a family history of hypertension significantly more common than in patients with vitiligo alone.
The investigators also checked for a grab bag of comorbid, nondermatologic diseases they suspect might be genetically linked to vitiligo and psoriasis, including lupus, celiac disease, megaloblastic anemia, and allergic rhinitis. Vitiligo patients with a family history of both vitiligo and psoriasis had a 47% prevalence of one or more of these conditions, significantly greater than in the other subgroups, where the prevalence ranged from 11% to 37%. In patients with comorbid vitiligo and psoriasis but no family history of either skin disease, the rate was 21%.
The prevalence of a diagnosed psychiatric disorder was zero in patients with vitiligo with or without a family history of vitiligo, 7% in those with vitiligo and a family history of both vitiligo and psoriasis, 6% in those with vitiligo and a family history of psoriasis, and 5% in patients with comorbid vitiligo and psoriasis.
Seventy-two percent of patients with both vitiligo and psoriasis were Fitzpatrick phototype 2 and the rest were phototype 3. Seventy-nine percent of those with vitiligo and a family history of both psoriasis and vitiligo were phototype 2, as were 56% of vitiligo patients with a family history of psoriasis.
In contrast, 60% of patients with only vitiligo were phototype 3 and 40% were phototype 2. The same 60/40 split held for patients with vitiligo and a family history of vitiligo.
Dr. Moretti declared having no financial conflicts of interest.
LISBON – Comorbid vitiligo and psoriasis is a red flag signaling the need to dig deeper looking for potentially associated diseases, including cardiovascular, autoimmune, or psychiatric diagnoses.
A study of 154 Italian vitiligo patients showed that a family history of cardiovascular disease was present in none of 54 participants with vitiligo alone compared to one-third of the 19 patients with both vitiligo and psoriasis, Dr. Silvia Moretti reported at the annual congress of the European Academy of Dermatology and Venereology.
Subjects with strong genetic loading for either or both of the skin diseases also tended to have an increased prevalence of a positive family history of cardiovascular disease, although this trend did not reach statistical significance. For example, a family history of cardiovascular disease was present in 10% of 52 patients with vitiligo and a family history of vitiligo, 13% of 15 patients with vitiligo as well as a family history of both vitiligo and psoriasis, and 18% of the 16 patients with vitiligo and a family history of psoriasis, according to Dr. Moretti of the University of Florence.
Similarly, a strong family history of hypertension was present in 4% of vitiligo patients, 8% of those with both vitiligo and a family history of vitiligo, 13% with vitiligo and a family history of both psoriasis and vitiligo, 25% of vitiligo patients with a family history of psoriasis, and 26% of patients carrying diagnoses of both vitiligo and psoriasis but no family history of either dermatologic disease. In only the group with both vitiligo and psoriasis was a family history of hypertension significantly more common than in patients with vitiligo alone.
The investigators also checked for a grab bag of comorbid, nondermatologic diseases they suspect might be genetically linked to vitiligo and psoriasis, including lupus, celiac disease, megaloblastic anemia, and allergic rhinitis. Vitiligo patients with a family history of both vitiligo and psoriasis had a 47% prevalence of one or more of these conditions, significantly greater than in the other subgroups, where the prevalence ranged from 11% to 37%. In patients with comorbid vitiligo and psoriasis but no family history of either skin disease, the rate was 21%.
The prevalence of a diagnosed psychiatric disorder was zero in patients with vitiligo with or without a family history of vitiligo, 7% in those with vitiligo and a family history of both vitiligo and psoriasis, 6% in those with vitiligo and a family history of psoriasis, and 5% in patients with comorbid vitiligo and psoriasis.
Seventy-two percent of patients with both vitiligo and psoriasis were Fitzpatrick phototype 2 and the rest were phototype 3. Seventy-nine percent of those with vitiligo and a family history of both psoriasis and vitiligo were phototype 2, as were 56% of vitiligo patients with a family history of psoriasis.
In contrast, 60% of patients with only vitiligo were phototype 3 and 40% were phototype 2. The same 60/40 split held for patients with vitiligo and a family history of vitiligo.
Dr. Moretti declared having no financial conflicts of interest.
FROM THE ANNUAL CONGRESS OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Major Finding: A family history of cardiovascular disease was present in none of 54 participants with vitiligo alone compared with one-third of the 19 patients with both vitiligo and psoriasis.
Data Source: A study of 154 vitiligo patients and their families.
Disclosures: No financial conflicts were reported.
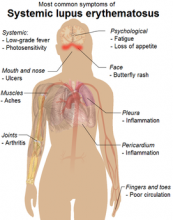
Blog: How Vitamin D Supplements May Help Lupus Patients
It seems like every week brings a new study about the benefits of vitamin D: It builds bones, tames psychotic symptoms in bipolar teens, and strengthens the immune system. It is the immune system benefit that attracted the interest of Dr. Benjamin Terrier and colleagues at the Pitié-Salpêtrière Hospital in Paris, France.
The researchers studied 24 people with lupus to determine the possible benefit of vitamin D supplementation on their immune systems. They presented their findings at this year’s annual meeting of the American College of Rheumatology.
Most importantly, the number of regulatory T cells increased and the number of T helper lymphocytes decreased after 2 months and also after 6 months of vitamin D supplementation. In addition, antibody-producing memory B cells decreased after 2 months, and activated CD8+ T cells (thought to be associated with lupus in particular) decreased after 6 months.
An added bonus: None of the patients reported adverse events associated with vitamin D, including hypercalcemia or lithiasis.
The findings are preliminary, Dr. Terrier said, and large, randomized controlled trials are needed to confirm the results.
Currently, no one is advocating that lupus patients increase their vitamin D with heavy supplementation, said Dr. Sam Lim of Emory University in Atlanta, Georgia. Dr. Lim served as moderator at the press conference in which the findings were presented.
However, “the study is very important because it is a link to take [the research] to the next step,” Dr. Lim said.
--Heidi Splete (On Twitter @hsplete)
Image courtesy of Mikael Häggström, via Wikimedia Commons
It seems like every week brings a new study about the benefits of vitamin D: It builds bones, tames psychotic symptoms in bipolar teens, and strengthens the immune system. It is the immune system benefit that attracted the interest of Dr. Benjamin Terrier and colleagues at the Pitié-Salpêtrière Hospital in Paris, France.
The researchers studied 24 people with lupus to determine the possible benefit of vitamin D supplementation on their immune systems. They presented their findings at this year’s annual meeting of the American College of Rheumatology.
Most importantly, the number of regulatory T cells increased and the number of T helper lymphocytes decreased after 2 months and also after 6 months of vitamin D supplementation. In addition, antibody-producing memory B cells decreased after 2 months, and activated CD8+ T cells (thought to be associated with lupus in particular) decreased after 6 months.
An added bonus: None of the patients reported adverse events associated with vitamin D, including hypercalcemia or lithiasis.
The findings are preliminary, Dr. Terrier said, and large, randomized controlled trials are needed to confirm the results.
Currently, no one is advocating that lupus patients increase their vitamin D with heavy supplementation, said Dr. Sam Lim of Emory University in Atlanta, Georgia. Dr. Lim served as moderator at the press conference in which the findings were presented.
However, “the study is very important because it is a link to take [the research] to the next step,” Dr. Lim said.
--Heidi Splete (On Twitter @hsplete)
Image courtesy of Mikael Häggström, via Wikimedia Commons
It seems like every week brings a new study about the benefits of vitamin D: It builds bones, tames psychotic symptoms in bipolar teens, and strengthens the immune system. It is the immune system benefit that attracted the interest of Dr. Benjamin Terrier and colleagues at the Pitié-Salpêtrière Hospital in Paris, France.
The researchers studied 24 people with lupus to determine the possible benefit of vitamin D supplementation on their immune systems. They presented their findings at this year’s annual meeting of the American College of Rheumatology.
Most importantly, the number of regulatory T cells increased and the number of T helper lymphocytes decreased after 2 months and also after 6 months of vitamin D supplementation. In addition, antibody-producing memory B cells decreased after 2 months, and activated CD8+ T cells (thought to be associated with lupus in particular) decreased after 6 months.
An added bonus: None of the patients reported adverse events associated with vitamin D, including hypercalcemia or lithiasis.
The findings are preliminary, Dr. Terrier said, and large, randomized controlled trials are needed to confirm the results.
Currently, no one is advocating that lupus patients increase their vitamin D with heavy supplementation, said Dr. Sam Lim of Emory University in Atlanta, Georgia. Dr. Lim served as moderator at the press conference in which the findings were presented.
However, “the study is very important because it is a link to take [the research] to the next step,” Dr. Lim said.
--Heidi Splete (On Twitter @hsplete)
Image courtesy of Mikael Häggström, via Wikimedia Commons
Safety Assurance Awaits Further Data for New Psoriasis Drugs
LAS VEGAS – New agents for treating psoriasis have shown great promise in terms of efficacy, but assurances of safety await further data from larger studies.
Thus far, phase II data show impressive efficacy and no worrisome safety signals for the anti-interleukin-17 agents AMG 827 and secukinumab and the small molecules apremilast and tofacitinib. However, the studies have been too small and of insufficient duration to definitively rule out cardiovascular, infectious, and cancer risks, said Dr. Kenneth B. Gordon at the Las Vegas Dermatology Seminar, sponsored by Skin Disease Education Foundation (SDEF).
"It is my hope that these drugs are going to be fantastic. We just have to maintain a level of concern and vigilance for both biologics and small molecules ... We just desperately need large trials," said Dr. Gordon, head of the division of dermatology at NorthShore University HealthSystem, Chicago.
The discovery of IL-17 as a key player in psoriatic plaque formation has led to a new understanding of psoriasis pathophysiology and has become a new target for drug development. However, experience with one agent that blocks IL-12/23 – which induces activated IL-17 – gives pause.
Briakinumab, an extremely effective anti-psoriatic agent that blocks IL 12/23, was withdrawn from development after phase III studies showed a signal – albeit statistically insignificant – for serious infections including cellulitis and pneumonia, MACE events (cardiac arrest, myocardial infarction, and stroke), and malignancies (nonmelanoma skin cancer and squamous cell carcinoma of the lung and nasopharynx).
The absolute MACE event numbers were small, but were counter to what would be expected from a systemic anti-inflammatory agent, which should reduce cardiovascular disease, Dr. Craig L. Leonardi, a clinical professor of dermatology at St. Louis University, said in a separate presentation.
According to Dr. Gordon, "It’s really an exciting time for new psoriatic therapies based on a better understanding of psoriasis pathophysiology. However, we are less excited about phase II data because of what happened to briakinumab in phase III."
It will be essential to determine the etiology of the adverse effects, he said. "If the effects are not related to how well the agent controls psoriasis but to mechanisms like IL-12 blockade that may not have relevance to IL-17, they may not show up [with the new agents in development]. That’s what the phase II studies suggest, but it’s too early to really make a final statement," Dr. Gordon said in an interview.
Phase II data on the anti-IL17 and small molecules were presented earlier this year at the European Academy of Dermatology and Venereology meeting in Lisbon and at the World Congress of Dermatology in Seoul. Among the findings:
• AMG 827: This fully-human monoclonal antibody binds to and blocks the IL-17 receptor. In a phase II, double-blind, randomized, placebo-controlled trial of 198 patients who were randomized to subcutaneous AMG 827 at 280 mg monthly; to 70, 140, or 210 mg every 2 weeks; or to placebo. The primary end point, PASI 75 response rate at week 12, was highest (83%) in patients who received 210 mg every 2 weeks. Moreover, at that dose, the proportion achieving a PASI 100 score, indicating no psoriasis activity, was 63%.
"This is an extraordinarily high-responding drug, similar only to briakinumab," Dr. Gordon commented.
Changes in neutrophil counts were seen, as to be expected from IL-17 and IL-12 blockade. "Some of the changes were significant. We need to keep an eye on this," he said.
• Secukinumab: This novel, fully human antibody to IL-17A was investigated in three separate phase II trials of patients with moderate to severe plaque psoriasis. An intravenous induction dose-ranging study of 100 patients yielded PASI 75 response rates at 12 weeks of 40%-83%. A subcutaneous dose-ranging study of 125 patients produced PASI 75 response rates of 19%-81%, and a subcutaneous regimen-finding trial involving 404 patients determined that a regimen of 150 mg at weeks 0, 1, 2, and 4 produced the best PASI 75 response at week 12, of 55%.
While the safety analysis showed no significant differences from placebo, there were two cases of cardiac disorders – one angina pectoris and one coronary artery disease – in the intravenous dose-ranging study and two cases listed as "cardiac disorders" in the regimen-finding study. "We don’t know if these are significant. We have to look at phase III trials," Dr. Gordon said.
• Apremilast: This small molecule, taken orally, works by inhibiting type 4 phosphodiesterase. In a randomized, placebo-controlled phase II trial of 352 patients with moderate to severe plaque psoriasis, the PASI 75 response rate at 16 weeks was 41% with a 30-mg twice-daily dose.
Adverse effects were dose-dependent. Adverse events that occurred in 5% or more of patients included headache, nausea, diarrhea, and upper respiratory tract infections. These typically occurred early in the course of treatment.
Serious adverse events included one myocardial infarction and one case of prostate cancer in the 30-mg BID group. But there was also a prostate cancer and a sudden death among the placebo recipients. "We really don’t have enough patients to look at adverse effects," Dr. Gordon said.
• Tofacitinib: This oral Janus kinase inhibitor demonstrated dose-dependent efficacy, with 67% of patients randomized at 15 mg twice a day achieving PASI 75 and "clear" or "almost clear." The phase II, 12-week, double-blind, placebo-controlled trial enrolled 197 patients with moderate to severe plaque psoriasis.
Concomitant overall decreases in hemoglobin and neutrophil counts from baseline were also dose-dependent. The finding did not specify the proportion of patients who had the decreases or the individual degree of change. "If everyone had a small change, it’s not going to bother me too much. But, if 10% of patients had a big change, it’s a really important finding. Those are the questions we need to ask," Dr. Gordon said.
Dr. Gordon said that so far the phase II safety data for the agents are encouraging, but enthusiasm should still be tempered. "When multiple drugs with the same mechanism have [similar] results, you start to feel more confident. But still, we need to see larger studies."
"It’s really an exciting time for new psoriatic therapies based on a better understanding of psoriasis pathophysiology."
Dr. Leonardi’s presentation focused on anti-IL 12/23 inhibitor treatment; he noted that the recently-approved monoclonal antibody ustekinumab binds to the same shared p40 subunit of IL-12 and IL-23 as briakinumab, the agent that was withdrawn from development in phase III. "There are more similarities than differences" between the two agents, he commented.
He and his colleagues recently conducted a meta-analysis of 22 randomized controlled clinical trials of biologic therapies comprising 10,183 patients with chronic plaque psoriasis, in which 10 of 3,179 patients receiving either ustekinumab or briakinumab experienced a MACE, compared with 0 events in 1,474 patients receiving placebo, for a MACE rate of 1.33 per 100 patient-years.
In contrast, no difference was seen among patients in the anti-TNF-alpha trials, with only 1 of 3,858 patients receiving anti-TNF-alpha agents experiencing a MACE, compared with 1 of 1,812 patients receiving placebo (JAMA 2011;306:864-71).
Although the difference for the anti-IL 12/23 agents was not statistically significant, the data set was not large enough to detect rare events. "This is a class effect in my mind," Dr. Leonardi said, adding that he uses ustekinumab as a second-line agent, after the TNF antagonists.
"It’s important to remember that all new drugs are ‘new’ ... We will learn more and more about these drugs as time goes on," he said.
Dr. Gordon disclosed that he has received research support or honoraria as a consultant from Abbott, Amgen, Centocor, Eli Lilly, Merck, Novartis, and Pfizer. Dr. Leonardi disclosed that he has had financial relationships with 23 companies that are involved in psoriasis treatment development, including Abbott and Centocor.
SDEF and this news organization are owned by Elsevier.
LAS VEGAS – New agents for treating psoriasis have shown great promise in terms of efficacy, but assurances of safety await further data from larger studies.
Thus far, phase II data show impressive efficacy and no worrisome safety signals for the anti-interleukin-17 agents AMG 827 and secukinumab and the small molecules apremilast and tofacitinib. However, the studies have been too small and of insufficient duration to definitively rule out cardiovascular, infectious, and cancer risks, said Dr. Kenneth B. Gordon at the Las Vegas Dermatology Seminar, sponsored by Skin Disease Education Foundation (SDEF).
"It is my hope that these drugs are going to be fantastic. We just have to maintain a level of concern and vigilance for both biologics and small molecules ... We just desperately need large trials," said Dr. Gordon, head of the division of dermatology at NorthShore University HealthSystem, Chicago.
The discovery of IL-17 as a key player in psoriatic plaque formation has led to a new understanding of psoriasis pathophysiology and has become a new target for drug development. However, experience with one agent that blocks IL-12/23 – which induces activated IL-17 – gives pause.
Briakinumab, an extremely effective anti-psoriatic agent that blocks IL 12/23, was withdrawn from development after phase III studies showed a signal – albeit statistically insignificant – for serious infections including cellulitis and pneumonia, MACE events (cardiac arrest, myocardial infarction, and stroke), and malignancies (nonmelanoma skin cancer and squamous cell carcinoma of the lung and nasopharynx).
The absolute MACE event numbers were small, but were counter to what would be expected from a systemic anti-inflammatory agent, which should reduce cardiovascular disease, Dr. Craig L. Leonardi, a clinical professor of dermatology at St. Louis University, said in a separate presentation.
According to Dr. Gordon, "It’s really an exciting time for new psoriatic therapies based on a better understanding of psoriasis pathophysiology. However, we are less excited about phase II data because of what happened to briakinumab in phase III."
It will be essential to determine the etiology of the adverse effects, he said. "If the effects are not related to how well the agent controls psoriasis but to mechanisms like IL-12 blockade that may not have relevance to IL-17, they may not show up [with the new agents in development]. That’s what the phase II studies suggest, but it’s too early to really make a final statement," Dr. Gordon said in an interview.
Phase II data on the anti-IL17 and small molecules were presented earlier this year at the European Academy of Dermatology and Venereology meeting in Lisbon and at the World Congress of Dermatology in Seoul. Among the findings:
• AMG 827: This fully-human monoclonal antibody binds to and blocks the IL-17 receptor. In a phase II, double-blind, randomized, placebo-controlled trial of 198 patients who were randomized to subcutaneous AMG 827 at 280 mg monthly; to 70, 140, or 210 mg every 2 weeks; or to placebo. The primary end point, PASI 75 response rate at week 12, was highest (83%) in patients who received 210 mg every 2 weeks. Moreover, at that dose, the proportion achieving a PASI 100 score, indicating no psoriasis activity, was 63%.
"This is an extraordinarily high-responding drug, similar only to briakinumab," Dr. Gordon commented.
Changes in neutrophil counts were seen, as to be expected from IL-17 and IL-12 blockade. "Some of the changes were significant. We need to keep an eye on this," he said.
• Secukinumab: This novel, fully human antibody to IL-17A was investigated in three separate phase II trials of patients with moderate to severe plaque psoriasis. An intravenous induction dose-ranging study of 100 patients yielded PASI 75 response rates at 12 weeks of 40%-83%. A subcutaneous dose-ranging study of 125 patients produced PASI 75 response rates of 19%-81%, and a subcutaneous regimen-finding trial involving 404 patients determined that a regimen of 150 mg at weeks 0, 1, 2, and 4 produced the best PASI 75 response at week 12, of 55%.
While the safety analysis showed no significant differences from placebo, there were two cases of cardiac disorders – one angina pectoris and one coronary artery disease – in the intravenous dose-ranging study and two cases listed as "cardiac disorders" in the regimen-finding study. "We don’t know if these are significant. We have to look at phase III trials," Dr. Gordon said.
• Apremilast: This small molecule, taken orally, works by inhibiting type 4 phosphodiesterase. In a randomized, placebo-controlled phase II trial of 352 patients with moderate to severe plaque psoriasis, the PASI 75 response rate at 16 weeks was 41% with a 30-mg twice-daily dose.
Adverse effects were dose-dependent. Adverse events that occurred in 5% or more of patients included headache, nausea, diarrhea, and upper respiratory tract infections. These typically occurred early in the course of treatment.
Serious adverse events included one myocardial infarction and one case of prostate cancer in the 30-mg BID group. But there was also a prostate cancer and a sudden death among the placebo recipients. "We really don’t have enough patients to look at adverse effects," Dr. Gordon said.
• Tofacitinib: This oral Janus kinase inhibitor demonstrated dose-dependent efficacy, with 67% of patients randomized at 15 mg twice a day achieving PASI 75 and "clear" or "almost clear." The phase II, 12-week, double-blind, placebo-controlled trial enrolled 197 patients with moderate to severe plaque psoriasis.
Concomitant overall decreases in hemoglobin and neutrophil counts from baseline were also dose-dependent. The finding did not specify the proportion of patients who had the decreases or the individual degree of change. "If everyone had a small change, it’s not going to bother me too much. But, if 10% of patients had a big change, it’s a really important finding. Those are the questions we need to ask," Dr. Gordon said.
Dr. Gordon said that so far the phase II safety data for the agents are encouraging, but enthusiasm should still be tempered. "When multiple drugs with the same mechanism have [similar] results, you start to feel more confident. But still, we need to see larger studies."
"It’s really an exciting time for new psoriatic therapies based on a better understanding of psoriasis pathophysiology."
Dr. Leonardi’s presentation focused on anti-IL 12/23 inhibitor treatment; he noted that the recently-approved monoclonal antibody ustekinumab binds to the same shared p40 subunit of IL-12 and IL-23 as briakinumab, the agent that was withdrawn from development in phase III. "There are more similarities than differences" between the two agents, he commented.
He and his colleagues recently conducted a meta-analysis of 22 randomized controlled clinical trials of biologic therapies comprising 10,183 patients with chronic plaque psoriasis, in which 10 of 3,179 patients receiving either ustekinumab or briakinumab experienced a MACE, compared with 0 events in 1,474 patients receiving placebo, for a MACE rate of 1.33 per 100 patient-years.
In contrast, no difference was seen among patients in the anti-TNF-alpha trials, with only 1 of 3,858 patients receiving anti-TNF-alpha agents experiencing a MACE, compared with 1 of 1,812 patients receiving placebo (JAMA 2011;306:864-71).
Although the difference for the anti-IL 12/23 agents was not statistically significant, the data set was not large enough to detect rare events. "This is a class effect in my mind," Dr. Leonardi said, adding that he uses ustekinumab as a second-line agent, after the TNF antagonists.
"It’s important to remember that all new drugs are ‘new’ ... We will learn more and more about these drugs as time goes on," he said.
Dr. Gordon disclosed that he has received research support or honoraria as a consultant from Abbott, Amgen, Centocor, Eli Lilly, Merck, Novartis, and Pfizer. Dr. Leonardi disclosed that he has had financial relationships with 23 companies that are involved in psoriasis treatment development, including Abbott and Centocor.
SDEF and this news organization are owned by Elsevier.
LAS VEGAS – New agents for treating psoriasis have shown great promise in terms of efficacy, but assurances of safety await further data from larger studies.
Thus far, phase II data show impressive efficacy and no worrisome safety signals for the anti-interleukin-17 agents AMG 827 and secukinumab and the small molecules apremilast and tofacitinib. However, the studies have been too small and of insufficient duration to definitively rule out cardiovascular, infectious, and cancer risks, said Dr. Kenneth B. Gordon at the Las Vegas Dermatology Seminar, sponsored by Skin Disease Education Foundation (SDEF).
"It is my hope that these drugs are going to be fantastic. We just have to maintain a level of concern and vigilance for both biologics and small molecules ... We just desperately need large trials," said Dr. Gordon, head of the division of dermatology at NorthShore University HealthSystem, Chicago.
The discovery of IL-17 as a key player in psoriatic plaque formation has led to a new understanding of psoriasis pathophysiology and has become a new target for drug development. However, experience with one agent that blocks IL-12/23 – which induces activated IL-17 – gives pause.
Briakinumab, an extremely effective anti-psoriatic agent that blocks IL 12/23, was withdrawn from development after phase III studies showed a signal – albeit statistically insignificant – for serious infections including cellulitis and pneumonia, MACE events (cardiac arrest, myocardial infarction, and stroke), and malignancies (nonmelanoma skin cancer and squamous cell carcinoma of the lung and nasopharynx).
The absolute MACE event numbers were small, but were counter to what would be expected from a systemic anti-inflammatory agent, which should reduce cardiovascular disease, Dr. Craig L. Leonardi, a clinical professor of dermatology at St. Louis University, said in a separate presentation.
According to Dr. Gordon, "It’s really an exciting time for new psoriatic therapies based on a better understanding of psoriasis pathophysiology. However, we are less excited about phase II data because of what happened to briakinumab in phase III."
It will be essential to determine the etiology of the adverse effects, he said. "If the effects are not related to how well the agent controls psoriasis but to mechanisms like IL-12 blockade that may not have relevance to IL-17, they may not show up [with the new agents in development]. That’s what the phase II studies suggest, but it’s too early to really make a final statement," Dr. Gordon said in an interview.
Phase II data on the anti-IL17 and small molecules were presented earlier this year at the European Academy of Dermatology and Venereology meeting in Lisbon and at the World Congress of Dermatology in Seoul. Among the findings:
• AMG 827: This fully-human monoclonal antibody binds to and blocks the IL-17 receptor. In a phase II, double-blind, randomized, placebo-controlled trial of 198 patients who were randomized to subcutaneous AMG 827 at 280 mg monthly; to 70, 140, or 210 mg every 2 weeks; or to placebo. The primary end point, PASI 75 response rate at week 12, was highest (83%) in patients who received 210 mg every 2 weeks. Moreover, at that dose, the proportion achieving a PASI 100 score, indicating no psoriasis activity, was 63%.
"This is an extraordinarily high-responding drug, similar only to briakinumab," Dr. Gordon commented.
Changes in neutrophil counts were seen, as to be expected from IL-17 and IL-12 blockade. "Some of the changes were significant. We need to keep an eye on this," he said.
• Secukinumab: This novel, fully human antibody to IL-17A was investigated in three separate phase II trials of patients with moderate to severe plaque psoriasis. An intravenous induction dose-ranging study of 100 patients yielded PASI 75 response rates at 12 weeks of 40%-83%. A subcutaneous dose-ranging study of 125 patients produced PASI 75 response rates of 19%-81%, and a subcutaneous regimen-finding trial involving 404 patients determined that a regimen of 150 mg at weeks 0, 1, 2, and 4 produced the best PASI 75 response at week 12, of 55%.
While the safety analysis showed no significant differences from placebo, there were two cases of cardiac disorders – one angina pectoris and one coronary artery disease – in the intravenous dose-ranging study and two cases listed as "cardiac disorders" in the regimen-finding study. "We don’t know if these are significant. We have to look at phase III trials," Dr. Gordon said.
• Apremilast: This small molecule, taken orally, works by inhibiting type 4 phosphodiesterase. In a randomized, placebo-controlled phase II trial of 352 patients with moderate to severe plaque psoriasis, the PASI 75 response rate at 16 weeks was 41% with a 30-mg twice-daily dose.
Adverse effects were dose-dependent. Adverse events that occurred in 5% or more of patients included headache, nausea, diarrhea, and upper respiratory tract infections. These typically occurred early in the course of treatment.
Serious adverse events included one myocardial infarction and one case of prostate cancer in the 30-mg BID group. But there was also a prostate cancer and a sudden death among the placebo recipients. "We really don’t have enough patients to look at adverse effects," Dr. Gordon said.
• Tofacitinib: This oral Janus kinase inhibitor demonstrated dose-dependent efficacy, with 67% of patients randomized at 15 mg twice a day achieving PASI 75 and "clear" or "almost clear." The phase II, 12-week, double-blind, placebo-controlled trial enrolled 197 patients with moderate to severe plaque psoriasis.
Concomitant overall decreases in hemoglobin and neutrophil counts from baseline were also dose-dependent. The finding did not specify the proportion of patients who had the decreases or the individual degree of change. "If everyone had a small change, it’s not going to bother me too much. But, if 10% of patients had a big change, it’s a really important finding. Those are the questions we need to ask," Dr. Gordon said.
Dr. Gordon said that so far the phase II safety data for the agents are encouraging, but enthusiasm should still be tempered. "When multiple drugs with the same mechanism have [similar] results, you start to feel more confident. But still, we need to see larger studies."
"It’s really an exciting time for new psoriatic therapies based on a better understanding of psoriasis pathophysiology."
Dr. Leonardi’s presentation focused on anti-IL 12/23 inhibitor treatment; he noted that the recently-approved monoclonal antibody ustekinumab binds to the same shared p40 subunit of IL-12 and IL-23 as briakinumab, the agent that was withdrawn from development in phase III. "There are more similarities than differences" between the two agents, he commented.
He and his colleagues recently conducted a meta-analysis of 22 randomized controlled clinical trials of biologic therapies comprising 10,183 patients with chronic plaque psoriasis, in which 10 of 3,179 patients receiving either ustekinumab or briakinumab experienced a MACE, compared with 0 events in 1,474 patients receiving placebo, for a MACE rate of 1.33 per 100 patient-years.
In contrast, no difference was seen among patients in the anti-TNF-alpha trials, with only 1 of 3,858 patients receiving anti-TNF-alpha agents experiencing a MACE, compared with 1 of 1,812 patients receiving placebo (JAMA 2011;306:864-71).
Although the difference for the anti-IL 12/23 agents was not statistically significant, the data set was not large enough to detect rare events. "This is a class effect in my mind," Dr. Leonardi said, adding that he uses ustekinumab as a second-line agent, after the TNF antagonists.
"It’s important to remember that all new drugs are ‘new’ ... We will learn more and more about these drugs as time goes on," he said.
Dr. Gordon disclosed that he has received research support or honoraria as a consultant from Abbott, Amgen, Centocor, Eli Lilly, Merck, Novartis, and Pfizer. Dr. Leonardi disclosed that he has had financial relationships with 23 companies that are involved in psoriasis treatment development, including Abbott and Centocor.
SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Psoriasis Patients Face Increased Risk of CVD
LAS VEGAS – The list of disease states and behaviors that potentially increase morbidity and mortality and lower quality of life for psoriasis patients is well known and includes psoriatic arthritis, Crohn’s disease, depression, alcoholism, and smoking.
Perhaps less well appreciated is the fact that psoriasis is also associated with multiple comorbidities that increase the risk of cardiovascular disease, including hypertension, diabetes, dyslipidemia, and obesity, Dr. Bruce E. Strober said at the SDEF Las Vegas Dermatology Seminar.
"The metabolic syndrome, a chronic inflammatory state that is associated with increased cardiovascular mortality, is also common in patients with psoriasis," noted Dr. Strober of the department of dermatology at the University of Connecticut, Farmington. The syndrome is associated with at least three of the following five factors: increased waist circumference or abdominal obesity, hypertension, hypertriglyceridemia, reduced high-density lipoprotein levels, and insulin resistance.
In 2006 German researchers published results from a trial that compared the prevalence of metabolic syndrome in 581 adults hospitalized with plaque-type psoriasis to the prevalence in 1,044 controls who were treated surgically for localized melanoma during the same time period (Arch. Dermatol. Res. 2006;298:321-8). Charts were screened for the presence of concomitant forms of chronic internal diseases.
The researchers discovered that compared with controls, psoriasis patients had a significantly higher prevalence of three components of metabolic syndrome, including diabetes mellitus (odds ratio, 2.48); hyperlipidemia (OR, 2.09); and arterial hypertension (OR, 3.27).
A separate, larger trial conducted in the United Kingdom set out to determine whether the prevalence of major cardiovascular risk factors was higher in mild or severe psoriasis patients, or in patients without psoriasis (J. Am. Acad. Dermatol. 2006;55:829-35). Patients were defined as having severe psoriasis if they received a code for psoriasis as well as systemic therapy used for the treatment of psoriasis, while patients were defined as having mild psoriasis if they ever received a psoriasis code but no systemic therapy.
The population-based study included 127,706 patients with mild psoriasis and 3,854 with severe disease. The researchers found that patients with mild psoriasis had a higher adjusted odds of diabetes (odds ratio, 1.13); hypertension (OR, 1.03); hyperlipidemia (OR, 1.16); obesity (OR, 1.27); and smoking (OR, 1.31), compared with controls, while patients with severe psoriasis had a higher adjusted odds of diabetes (OR, 1.62); obesity (OR, 1.79);and smoking (OR, 1.31) than controls, Dr. Strober reported at the seminar sponsored by Skin Disease Education Foundation (SDEF).
In addition, diabetes (OR, 1.39) and obesity (OR 1.47) were more prevalent in those with severe psoriasis than with mild psoriasis.
According to Dr. Strober, one of the largest studies to evaluate the association between obesity and the risk of psoriasis comes from 78,626 women who participated in the ongoing Nurses’ Health Study (Arch. Intern. Med. 2007;167:1670-5). After adjustment for age, alcohol consumption, and smoking status, the researchers found that the relative risk of developing psoriasis increased with advancing body mass index category: 1.40 for a BMI of 25-29.9 kg/m2, 1.48 for a BMI of 30-34.9 kg/m2, and 2.69 for a BMI of 35 kg/m2 or greater.
A higher waist circumference, hip circumference, and waist-hip ratio were also associated with a higher risk of incident psoriasis.
Myocardial infarction is also associated with psoriasis, according to a population-based study in the United Kingdom that evaluated outcomes in 556,995 controls, 127,139 patients with mild psoriasis, and 3,837 patients with severe psoriasis (JAMA 2006;296:1735-41). The researchers found that the adjusted relative risk of myocardial infarction was 3.58 among controls, 4.04 among those with mild psoriasis, and 5.13 among those with severe psoriasis.
"Possible causes for increased cardiovascular risks in psoriasis include the use of dyslipidemic therapies, such as corticosteroids, acitretin, and cyclosporine; the increased prevalence of obesity and other associated risk factors; and uncontrolled inflammation leading to endothelial dysfunction and dyslipidemia," Dr. Strober said.
He emphasized that many questions remain about the association between psoriasis and cardiovascular risk factors. "Do the associations have any clinical relevance?" he asked. "Do specific comorbidities influence the response to therapy for psoriasis? Does psoriasis influence the response to therapy for any specific comorbidity? Will specific psoriasis therapies reduce the negative health consequences of a comorbidity of psoriasis?"
For now, he said, dermatologists might consider screening patients for the prevalence of cardiovascular risk factors through laboratory evaluations such as a comprehensive metabolic panel and fasting lipids, assessing blood pressure and weight status, and asking patients about their history of smoking, alcohol use, depression, and arthritis.
Dr. Strober disclosed that he is a member of the scientific advisory board for Abbott, Amgen, Janssen, Novartis, and Pfizer. He also disclosed that he is a consultant for Abbott, Amgen, Celgene, Centocor, Galderma, Leo, Maruho, Novartis, and Pfizer, and that he has received honoraria from Abbott.
SDEF and this news organization are owned by Elsevier.
LAS VEGAS – The list of disease states and behaviors that potentially increase morbidity and mortality and lower quality of life for psoriasis patients is well known and includes psoriatic arthritis, Crohn’s disease, depression, alcoholism, and smoking.
Perhaps less well appreciated is the fact that psoriasis is also associated with multiple comorbidities that increase the risk of cardiovascular disease, including hypertension, diabetes, dyslipidemia, and obesity, Dr. Bruce E. Strober said at the SDEF Las Vegas Dermatology Seminar.
"The metabolic syndrome, a chronic inflammatory state that is associated with increased cardiovascular mortality, is also common in patients with psoriasis," noted Dr. Strober of the department of dermatology at the University of Connecticut, Farmington. The syndrome is associated with at least three of the following five factors: increased waist circumference or abdominal obesity, hypertension, hypertriglyceridemia, reduced high-density lipoprotein levels, and insulin resistance.
In 2006 German researchers published results from a trial that compared the prevalence of metabolic syndrome in 581 adults hospitalized with plaque-type psoriasis to the prevalence in 1,044 controls who were treated surgically for localized melanoma during the same time period (Arch. Dermatol. Res. 2006;298:321-8). Charts were screened for the presence of concomitant forms of chronic internal diseases.
The researchers discovered that compared with controls, psoriasis patients had a significantly higher prevalence of three components of metabolic syndrome, including diabetes mellitus (odds ratio, 2.48); hyperlipidemia (OR, 2.09); and arterial hypertension (OR, 3.27).
A separate, larger trial conducted in the United Kingdom set out to determine whether the prevalence of major cardiovascular risk factors was higher in mild or severe psoriasis patients, or in patients without psoriasis (J. Am. Acad. Dermatol. 2006;55:829-35). Patients were defined as having severe psoriasis if they received a code for psoriasis as well as systemic therapy used for the treatment of psoriasis, while patients were defined as having mild psoriasis if they ever received a psoriasis code but no systemic therapy.
The population-based study included 127,706 patients with mild psoriasis and 3,854 with severe disease. The researchers found that patients with mild psoriasis had a higher adjusted odds of diabetes (odds ratio, 1.13); hypertension (OR, 1.03); hyperlipidemia (OR, 1.16); obesity (OR, 1.27); and smoking (OR, 1.31), compared with controls, while patients with severe psoriasis had a higher adjusted odds of diabetes (OR, 1.62); obesity (OR, 1.79);and smoking (OR, 1.31) than controls, Dr. Strober reported at the seminar sponsored by Skin Disease Education Foundation (SDEF).
In addition, diabetes (OR, 1.39) and obesity (OR 1.47) were more prevalent in those with severe psoriasis than with mild psoriasis.
According to Dr. Strober, one of the largest studies to evaluate the association between obesity and the risk of psoriasis comes from 78,626 women who participated in the ongoing Nurses’ Health Study (Arch. Intern. Med. 2007;167:1670-5). After adjustment for age, alcohol consumption, and smoking status, the researchers found that the relative risk of developing psoriasis increased with advancing body mass index category: 1.40 for a BMI of 25-29.9 kg/m2, 1.48 for a BMI of 30-34.9 kg/m2, and 2.69 for a BMI of 35 kg/m2 or greater.
A higher waist circumference, hip circumference, and waist-hip ratio were also associated with a higher risk of incident psoriasis.
Myocardial infarction is also associated with psoriasis, according to a population-based study in the United Kingdom that evaluated outcomes in 556,995 controls, 127,139 patients with mild psoriasis, and 3,837 patients with severe psoriasis (JAMA 2006;296:1735-41). The researchers found that the adjusted relative risk of myocardial infarction was 3.58 among controls, 4.04 among those with mild psoriasis, and 5.13 among those with severe psoriasis.
"Possible causes for increased cardiovascular risks in psoriasis include the use of dyslipidemic therapies, such as corticosteroids, acitretin, and cyclosporine; the increased prevalence of obesity and other associated risk factors; and uncontrolled inflammation leading to endothelial dysfunction and dyslipidemia," Dr. Strober said.
He emphasized that many questions remain about the association between psoriasis and cardiovascular risk factors. "Do the associations have any clinical relevance?" he asked. "Do specific comorbidities influence the response to therapy for psoriasis? Does psoriasis influence the response to therapy for any specific comorbidity? Will specific psoriasis therapies reduce the negative health consequences of a comorbidity of psoriasis?"
For now, he said, dermatologists might consider screening patients for the prevalence of cardiovascular risk factors through laboratory evaluations such as a comprehensive metabolic panel and fasting lipids, assessing blood pressure and weight status, and asking patients about their history of smoking, alcohol use, depression, and arthritis.
Dr. Strober disclosed that he is a member of the scientific advisory board for Abbott, Amgen, Janssen, Novartis, and Pfizer. He also disclosed that he is a consultant for Abbott, Amgen, Celgene, Centocor, Galderma, Leo, Maruho, Novartis, and Pfizer, and that he has received honoraria from Abbott.
SDEF and this news organization are owned by Elsevier.
LAS VEGAS – The list of disease states and behaviors that potentially increase morbidity and mortality and lower quality of life for psoriasis patients is well known and includes psoriatic arthritis, Crohn’s disease, depression, alcoholism, and smoking.
Perhaps less well appreciated is the fact that psoriasis is also associated with multiple comorbidities that increase the risk of cardiovascular disease, including hypertension, diabetes, dyslipidemia, and obesity, Dr. Bruce E. Strober said at the SDEF Las Vegas Dermatology Seminar.
"The metabolic syndrome, a chronic inflammatory state that is associated with increased cardiovascular mortality, is also common in patients with psoriasis," noted Dr. Strober of the department of dermatology at the University of Connecticut, Farmington. The syndrome is associated with at least three of the following five factors: increased waist circumference or abdominal obesity, hypertension, hypertriglyceridemia, reduced high-density lipoprotein levels, and insulin resistance.
In 2006 German researchers published results from a trial that compared the prevalence of metabolic syndrome in 581 adults hospitalized with plaque-type psoriasis to the prevalence in 1,044 controls who were treated surgically for localized melanoma during the same time period (Arch. Dermatol. Res. 2006;298:321-8). Charts were screened for the presence of concomitant forms of chronic internal diseases.
The researchers discovered that compared with controls, psoriasis patients had a significantly higher prevalence of three components of metabolic syndrome, including diabetes mellitus (odds ratio, 2.48); hyperlipidemia (OR, 2.09); and arterial hypertension (OR, 3.27).
A separate, larger trial conducted in the United Kingdom set out to determine whether the prevalence of major cardiovascular risk factors was higher in mild or severe psoriasis patients, or in patients without psoriasis (J. Am. Acad. Dermatol. 2006;55:829-35). Patients were defined as having severe psoriasis if they received a code for psoriasis as well as systemic therapy used for the treatment of psoriasis, while patients were defined as having mild psoriasis if they ever received a psoriasis code but no systemic therapy.
The population-based study included 127,706 patients with mild psoriasis and 3,854 with severe disease. The researchers found that patients with mild psoriasis had a higher adjusted odds of diabetes (odds ratio, 1.13); hypertension (OR, 1.03); hyperlipidemia (OR, 1.16); obesity (OR, 1.27); and smoking (OR, 1.31), compared with controls, while patients with severe psoriasis had a higher adjusted odds of diabetes (OR, 1.62); obesity (OR, 1.79);and smoking (OR, 1.31) than controls, Dr. Strober reported at the seminar sponsored by Skin Disease Education Foundation (SDEF).
In addition, diabetes (OR, 1.39) and obesity (OR 1.47) were more prevalent in those with severe psoriasis than with mild psoriasis.
According to Dr. Strober, one of the largest studies to evaluate the association between obesity and the risk of psoriasis comes from 78,626 women who participated in the ongoing Nurses’ Health Study (Arch. Intern. Med. 2007;167:1670-5). After adjustment for age, alcohol consumption, and smoking status, the researchers found that the relative risk of developing psoriasis increased with advancing body mass index category: 1.40 for a BMI of 25-29.9 kg/m2, 1.48 for a BMI of 30-34.9 kg/m2, and 2.69 for a BMI of 35 kg/m2 or greater.
A higher waist circumference, hip circumference, and waist-hip ratio were also associated with a higher risk of incident psoriasis.
Myocardial infarction is also associated with psoriasis, according to a population-based study in the United Kingdom that evaluated outcomes in 556,995 controls, 127,139 patients with mild psoriasis, and 3,837 patients with severe psoriasis (JAMA 2006;296:1735-41). The researchers found that the adjusted relative risk of myocardial infarction was 3.58 among controls, 4.04 among those with mild psoriasis, and 5.13 among those with severe psoriasis.
"Possible causes for increased cardiovascular risks in psoriasis include the use of dyslipidemic therapies, such as corticosteroids, acitretin, and cyclosporine; the increased prevalence of obesity and other associated risk factors; and uncontrolled inflammation leading to endothelial dysfunction and dyslipidemia," Dr. Strober said.
He emphasized that many questions remain about the association between psoriasis and cardiovascular risk factors. "Do the associations have any clinical relevance?" he asked. "Do specific comorbidities influence the response to therapy for psoriasis? Does psoriasis influence the response to therapy for any specific comorbidity? Will specific psoriasis therapies reduce the negative health consequences of a comorbidity of psoriasis?"
For now, he said, dermatologists might consider screening patients for the prevalence of cardiovascular risk factors through laboratory evaluations such as a comprehensive metabolic panel and fasting lipids, assessing blood pressure and weight status, and asking patients about their history of smoking, alcohol use, depression, and arthritis.
Dr. Strober disclosed that he is a member of the scientific advisory board for Abbott, Amgen, Janssen, Novartis, and Pfizer. He also disclosed that he is a consultant for Abbott, Amgen, Celgene, Centocor, Galderma, Leo, Maruho, Novartis, and Pfizer, and that he has received honoraria from Abbott.
SDEF and this news organization are owned by Elsevier.
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
Formula Calculates Need for Heart Catheterization in SSc
A new formula accurately calculates the risk of pulmonary hypertension by predicting the mean pulmonary artery pressure using data obtained from pulmonary function testing and pulse oximetry, according to a report by Dr. Benjamin E. Schreiber, of the Royal Free Hampstead National Health Service Trust, London, and his associates.
They developed the formula based on findings in 257 patients with systemic sclerosis; the investigators validated it with data from another 129 similar patients.
Patients with a predicted mean pulmonary artery pressure (mPAP) below 25 mm Hg had a low pulmonary hypertension (PH) prevalence (4.4%). Those with a predicted mPAP between 25-35 mm Hg had an average PH prevalence (11.3%). Finally, those with a predicted mPAP above 35 mm Hg had a high PH prevalence (62.9%), which justifies using right-sided heart catheterization (RHC) (Arthritis Rheum. 2011;63:3531-9).
The study started with 838 patients who have systemic sclerosis (SSc) who had undergone an RHC. Of those, 386 patients had undergone pulmonary function testing (PFT) within 6 months of the RHC. Data on oxygen saturation as measured by pulse oximetry (SpO2) at the time of the RHC were included in the study. Of the total number of patients, 452 were excluded because they had incomplete PFT data.
The investigators randomly split the remaining 386 into two groups: the derivation and validation cohorts. The derivation cohort had 257 patients and its data were used to create the formula. The validation cohort had 129 subjects and its data were used to validate the formula. An additional 155 patients with connective tissue diseases other than SSc were obtained from the Royal Free Pulmonary Hypertension Service in London, United Kingdom.
The formula was derived using multivariable linear regression in the derivation cohort. Dr. Schreiber and his team examined the relationship between the SpO2, PFTs, clinical subtype, autoimmune serologic features, and the mPAP on RHC and developed the following formula: predicted mPAP = 136 – SpO2 –0.25 x diffusing capacity for carbon monoxide (DLCO)% predicted.
Applying the formula to the validation cohort gave an area under the curve (AUC) of 0.75. The formula performed best in patients with anticentromere antibodies, with an AUC of 0.87. In patients with connective tissue diseases other than SSc, the formula gave an AUC of 0.64.
The researchers suggested the PFT-derived formula can complement data obtained through echocardiography, and that both tests are predictive. Echocardiography is a good tool for diagnosing PH, but it cannot evaluate lung disease and is not always feasible to conduct. Therefore, the formula can calculate which patients are most likely to have PH.
Some of the limitations of this retrospective study included missing data, especially from echocardiograms and PFTs, and lack of rigorous selection of patients for RHC. There were no echocardiographic data available for most patients, and there may have been selection bias in the patients who had a second echocardiography performed.
Dr. Schreiber and his team suggested that further research should combine PFT data with other noninvasive information, such as symptoms, 6-minute walking distance, and brain natriuretic peptide levels. They also said that they plan to continue to validate their formula in patients recruited into the DETECT (Detection of Pulmonary Artery Hypertension in Systemic Sclerosis) study.
No relevant financial disclosures were reported.
A new formula accurately calculates the risk of pulmonary hypertension by predicting the mean pulmonary artery pressure using data obtained from pulmonary function testing and pulse oximetry, according to a report by Dr. Benjamin E. Schreiber, of the Royal Free Hampstead National Health Service Trust, London, and his associates.
They developed the formula based on findings in 257 patients with systemic sclerosis; the investigators validated it with data from another 129 similar patients.
Patients with a predicted mean pulmonary artery pressure (mPAP) below 25 mm Hg had a low pulmonary hypertension (PH) prevalence (4.4%). Those with a predicted mPAP between 25-35 mm Hg had an average PH prevalence (11.3%). Finally, those with a predicted mPAP above 35 mm Hg had a high PH prevalence (62.9%), which justifies using right-sided heart catheterization (RHC) (Arthritis Rheum. 2011;63:3531-9).
The study started with 838 patients who have systemic sclerosis (SSc) who had undergone an RHC. Of those, 386 patients had undergone pulmonary function testing (PFT) within 6 months of the RHC. Data on oxygen saturation as measured by pulse oximetry (SpO2) at the time of the RHC were included in the study. Of the total number of patients, 452 were excluded because they had incomplete PFT data.
The investigators randomly split the remaining 386 into two groups: the derivation and validation cohorts. The derivation cohort had 257 patients and its data were used to create the formula. The validation cohort had 129 subjects and its data were used to validate the formula. An additional 155 patients with connective tissue diseases other than SSc were obtained from the Royal Free Pulmonary Hypertension Service in London, United Kingdom.
The formula was derived using multivariable linear regression in the derivation cohort. Dr. Schreiber and his team examined the relationship between the SpO2, PFTs, clinical subtype, autoimmune serologic features, and the mPAP on RHC and developed the following formula: predicted mPAP = 136 – SpO2 –0.25 x diffusing capacity for carbon monoxide (DLCO)% predicted.
Applying the formula to the validation cohort gave an area under the curve (AUC) of 0.75. The formula performed best in patients with anticentromere antibodies, with an AUC of 0.87. In patients with connective tissue diseases other than SSc, the formula gave an AUC of 0.64.
The researchers suggested the PFT-derived formula can complement data obtained through echocardiography, and that both tests are predictive. Echocardiography is a good tool for diagnosing PH, but it cannot evaluate lung disease and is not always feasible to conduct. Therefore, the formula can calculate which patients are most likely to have PH.
Some of the limitations of this retrospective study included missing data, especially from echocardiograms and PFTs, and lack of rigorous selection of patients for RHC. There were no echocardiographic data available for most patients, and there may have been selection bias in the patients who had a second echocardiography performed.
Dr. Schreiber and his team suggested that further research should combine PFT data with other noninvasive information, such as symptoms, 6-minute walking distance, and brain natriuretic peptide levels. They also said that they plan to continue to validate their formula in patients recruited into the DETECT (Detection of Pulmonary Artery Hypertension in Systemic Sclerosis) study.
No relevant financial disclosures were reported.
A new formula accurately calculates the risk of pulmonary hypertension by predicting the mean pulmonary artery pressure using data obtained from pulmonary function testing and pulse oximetry, according to a report by Dr. Benjamin E. Schreiber, of the Royal Free Hampstead National Health Service Trust, London, and his associates.
They developed the formula based on findings in 257 patients with systemic sclerosis; the investigators validated it with data from another 129 similar patients.
Patients with a predicted mean pulmonary artery pressure (mPAP) below 25 mm Hg had a low pulmonary hypertension (PH) prevalence (4.4%). Those with a predicted mPAP between 25-35 mm Hg had an average PH prevalence (11.3%). Finally, those with a predicted mPAP above 35 mm Hg had a high PH prevalence (62.9%), which justifies using right-sided heart catheterization (RHC) (Arthritis Rheum. 2011;63:3531-9).
The study started with 838 patients who have systemic sclerosis (SSc) who had undergone an RHC. Of those, 386 patients had undergone pulmonary function testing (PFT) within 6 months of the RHC. Data on oxygen saturation as measured by pulse oximetry (SpO2) at the time of the RHC were included in the study. Of the total number of patients, 452 were excluded because they had incomplete PFT data.
The investigators randomly split the remaining 386 into two groups: the derivation and validation cohorts. The derivation cohort had 257 patients and its data were used to create the formula. The validation cohort had 129 subjects and its data were used to validate the formula. An additional 155 patients with connective tissue diseases other than SSc were obtained from the Royal Free Pulmonary Hypertension Service in London, United Kingdom.
The formula was derived using multivariable linear regression in the derivation cohort. Dr. Schreiber and his team examined the relationship between the SpO2, PFTs, clinical subtype, autoimmune serologic features, and the mPAP on RHC and developed the following formula: predicted mPAP = 136 – SpO2 –0.25 x diffusing capacity for carbon monoxide (DLCO)% predicted.
Applying the formula to the validation cohort gave an area under the curve (AUC) of 0.75. The formula performed best in patients with anticentromere antibodies, with an AUC of 0.87. In patients with connective tissue diseases other than SSc, the formula gave an AUC of 0.64.
The researchers suggested the PFT-derived formula can complement data obtained through echocardiography, and that both tests are predictive. Echocardiography is a good tool for diagnosing PH, but it cannot evaluate lung disease and is not always feasible to conduct. Therefore, the formula can calculate which patients are most likely to have PH.
Some of the limitations of this retrospective study included missing data, especially from echocardiograms and PFTs, and lack of rigorous selection of patients for RHC. There were no echocardiographic data available for most patients, and there may have been selection bias in the patients who had a second echocardiography performed.
Dr. Schreiber and his team suggested that further research should combine PFT data with other noninvasive information, such as symptoms, 6-minute walking distance, and brain natriuretic peptide levels. They also said that they plan to continue to validate their formula in patients recruited into the DETECT (Detection of Pulmonary Artery Hypertension in Systemic Sclerosis) study.
No relevant financial disclosures were reported.
FROM ARTHRITIS & RHEUMATISM
Major Finding: Patients with a predicted mean pulmonary artery pressure (mPAP) below 25 mm Hg showed a pulmonary hypertension (PH) prevalence of 4.4%, while those with a predicted mPAP of 25-35 mm Hg showed a PH prevalence of 11.3%, and those with a predicted mPAP above 35 mm Hg showed a PH prevalence of 62.9%, thus justifying use of right-sided heart catheterization.
Data Source: Results of pulmonary function tests of 386 patients with systemic sclerosis were quantified against data obtained from right-sided heart catheterization.
Disclosures: No relevant financial disclosures were reported.
New Lupus Nephritis Guidelines Will Address Therapy
CHICAGO – Newly updated American College of Rheumatology guidelines for the screening and management of lupus nephritis in adults, which are expected to be published in early 2012, focus largely on induction therapy with mycophenolate mofetil or cyclophosphamide and on maintenance therapy with mycophenolate mofetil and azathioprine.
An algorithm for diffuse proliferative nephritis, for example, indicates that in patients with active class III and IV disease without cellular crescents, induction therapy should include mycophenolate mofetil (MMF) at 2-3 g/day for 6 months. Cyclophosphamide is an alternative option in these patients, according to the guidelines, which note that the literature suggests MMF is preferred for initial therapy in African Americans and Hispanics in North America and Latin America.
These recommendations are based on level A evidence from multiple randomized controlled trials or a meta-analysis, Dr. Bevra H. Hahn noted at the annual meeting of the American College of Rheumatology, where she presented the updated guidelines.
In patients who are given cyclophosphamide, the dose should be 500 mg intravenously every 2 weeks for 6 weeks, or 500-1,000 mg/m2 of body surface area. This recommendation is based on level B evidence from a single randomized controlled trial or nonrandomized studies. The latter choice has been studied only in Caucasians of European descent, noted Dr. Hahn, a professor of medicine and chair of the rheumatology department at the University of California, Los Angeles, as well as a member of the core working group for the updated guidelines.
Also recommended for class III and IV patients without cellular crescents is a concomitant pulse of steroids for 3 days, then prednisone at a dose of 0.5-1.0 mg/kg to induce improvement, with tapering to the lowest effective dose after a few weeks. Data supporting a specific dose in this range are lacking, and this recommendation, which is based on level C evidence indicating the recommendation is derived from consensus, expert opinion, or a case series, also states that the prednisone should continue for 6 months.
The algorithm for patients with diffuse proliferative disease also addresses patients with cellular crescents. Such patients should receive the higher 1.0-mg/kg per day prednisone dose. Patients who do not respond to induction should be switched from their initial induction regimen, be it MMF or cyclophosphamide, to the other. Maintenance therapy for responders to either MMF or cyclophosphamide should include MMF at 1-2 g/day or azathioprine at 2 mg/kg per day plus low-dose daily glucocorticoids.
The updated guidelines also address renal biopsy, adjunctive treatments, patients with focal disease, patients with class V disease, lupus nephritis in pregnancy, and recommendations for monitoring.
Notably, the guidelines do not advocate use of belimumab, as it has not been studied in active lupus nephritis, Dr. Hahn said, noting that despite the expanded literature on lupus nephritis that led to development of these updated guidelines, there are a number of unmet needs.
Between 20% and 35% of patients do not respond at all to induction therapies, and in many the response is slow, taking up to 52 weeks to determine response. Furthermore, response rates remain low.
"So it takes a long time, and you certainly don’t get 100% response rates, she said.
The guidelines, developed by a core working group that conducted the literature reviews and wrote scenarios, and by a task force panel of international experts in rheumatology, nephrology, and pathology who voted on the scenarios, will be published in Arthritis Care & Research, most likely in the spring, Dr. Hahn said. Currently they are pending approval by the ACR Guidelines Subcommittee, Quality of Care Committee, and Board of Directors.
Dr. Hahn, however, is already looking forward to new advances in the treatment of lupus nephritis and to the next guidelines update.
"I can’t wait to have something that’s better, so in another decade when we write guidelines, we can talk to you about 80% to 100% response rates," she said.
The lupus nephritis guideline project was sponsored by the ACR via a competitive grant mechanism. Of the 39 working group and task force members, 16 declared potential conflicts of interest, including relationships with Aspreva, Bristol-Myers Squibb, Genentech, and Human Genome Sciences.
CHICAGO – Newly updated American College of Rheumatology guidelines for the screening and management of lupus nephritis in adults, which are expected to be published in early 2012, focus largely on induction therapy with mycophenolate mofetil or cyclophosphamide and on maintenance therapy with mycophenolate mofetil and azathioprine.
An algorithm for diffuse proliferative nephritis, for example, indicates that in patients with active class III and IV disease without cellular crescents, induction therapy should include mycophenolate mofetil (MMF) at 2-3 g/day for 6 months. Cyclophosphamide is an alternative option in these patients, according to the guidelines, which note that the literature suggests MMF is preferred for initial therapy in African Americans and Hispanics in North America and Latin America.
These recommendations are based on level A evidence from multiple randomized controlled trials or a meta-analysis, Dr. Bevra H. Hahn noted at the annual meeting of the American College of Rheumatology, where she presented the updated guidelines.
In patients who are given cyclophosphamide, the dose should be 500 mg intravenously every 2 weeks for 6 weeks, or 500-1,000 mg/m2 of body surface area. This recommendation is based on level B evidence from a single randomized controlled trial or nonrandomized studies. The latter choice has been studied only in Caucasians of European descent, noted Dr. Hahn, a professor of medicine and chair of the rheumatology department at the University of California, Los Angeles, as well as a member of the core working group for the updated guidelines.
Also recommended for class III and IV patients without cellular crescents is a concomitant pulse of steroids for 3 days, then prednisone at a dose of 0.5-1.0 mg/kg to induce improvement, with tapering to the lowest effective dose after a few weeks. Data supporting a specific dose in this range are lacking, and this recommendation, which is based on level C evidence indicating the recommendation is derived from consensus, expert opinion, or a case series, also states that the prednisone should continue for 6 months.
The algorithm for patients with diffuse proliferative disease also addresses patients with cellular crescents. Such patients should receive the higher 1.0-mg/kg per day prednisone dose. Patients who do not respond to induction should be switched from their initial induction regimen, be it MMF or cyclophosphamide, to the other. Maintenance therapy for responders to either MMF or cyclophosphamide should include MMF at 1-2 g/day or azathioprine at 2 mg/kg per day plus low-dose daily glucocorticoids.
The updated guidelines also address renal biopsy, adjunctive treatments, patients with focal disease, patients with class V disease, lupus nephritis in pregnancy, and recommendations for monitoring.
Notably, the guidelines do not advocate use of belimumab, as it has not been studied in active lupus nephritis, Dr. Hahn said, noting that despite the expanded literature on lupus nephritis that led to development of these updated guidelines, there are a number of unmet needs.
Between 20% and 35% of patients do not respond at all to induction therapies, and in many the response is slow, taking up to 52 weeks to determine response. Furthermore, response rates remain low.
"So it takes a long time, and you certainly don’t get 100% response rates, she said.
The guidelines, developed by a core working group that conducted the literature reviews and wrote scenarios, and by a task force panel of international experts in rheumatology, nephrology, and pathology who voted on the scenarios, will be published in Arthritis Care & Research, most likely in the spring, Dr. Hahn said. Currently they are pending approval by the ACR Guidelines Subcommittee, Quality of Care Committee, and Board of Directors.
Dr. Hahn, however, is already looking forward to new advances in the treatment of lupus nephritis and to the next guidelines update.
"I can’t wait to have something that’s better, so in another decade when we write guidelines, we can talk to you about 80% to 100% response rates," she said.
The lupus nephritis guideline project was sponsored by the ACR via a competitive grant mechanism. Of the 39 working group and task force members, 16 declared potential conflicts of interest, including relationships with Aspreva, Bristol-Myers Squibb, Genentech, and Human Genome Sciences.
CHICAGO – Newly updated American College of Rheumatology guidelines for the screening and management of lupus nephritis in adults, which are expected to be published in early 2012, focus largely on induction therapy with mycophenolate mofetil or cyclophosphamide and on maintenance therapy with mycophenolate mofetil and azathioprine.
An algorithm for diffuse proliferative nephritis, for example, indicates that in patients with active class III and IV disease without cellular crescents, induction therapy should include mycophenolate mofetil (MMF) at 2-3 g/day for 6 months. Cyclophosphamide is an alternative option in these patients, according to the guidelines, which note that the literature suggests MMF is preferred for initial therapy in African Americans and Hispanics in North America and Latin America.
These recommendations are based on level A evidence from multiple randomized controlled trials or a meta-analysis, Dr. Bevra H. Hahn noted at the annual meeting of the American College of Rheumatology, where she presented the updated guidelines.
In patients who are given cyclophosphamide, the dose should be 500 mg intravenously every 2 weeks for 6 weeks, or 500-1,000 mg/m2 of body surface area. This recommendation is based on level B evidence from a single randomized controlled trial or nonrandomized studies. The latter choice has been studied only in Caucasians of European descent, noted Dr. Hahn, a professor of medicine and chair of the rheumatology department at the University of California, Los Angeles, as well as a member of the core working group for the updated guidelines.
Also recommended for class III and IV patients without cellular crescents is a concomitant pulse of steroids for 3 days, then prednisone at a dose of 0.5-1.0 mg/kg to induce improvement, with tapering to the lowest effective dose after a few weeks. Data supporting a specific dose in this range are lacking, and this recommendation, which is based on level C evidence indicating the recommendation is derived from consensus, expert opinion, or a case series, also states that the prednisone should continue for 6 months.
The algorithm for patients with diffuse proliferative disease also addresses patients with cellular crescents. Such patients should receive the higher 1.0-mg/kg per day prednisone dose. Patients who do not respond to induction should be switched from their initial induction regimen, be it MMF or cyclophosphamide, to the other. Maintenance therapy for responders to either MMF or cyclophosphamide should include MMF at 1-2 g/day or azathioprine at 2 mg/kg per day plus low-dose daily glucocorticoids.
The updated guidelines also address renal biopsy, adjunctive treatments, patients with focal disease, patients with class V disease, lupus nephritis in pregnancy, and recommendations for monitoring.
Notably, the guidelines do not advocate use of belimumab, as it has not been studied in active lupus nephritis, Dr. Hahn said, noting that despite the expanded literature on lupus nephritis that led to development of these updated guidelines, there are a number of unmet needs.
Between 20% and 35% of patients do not respond at all to induction therapies, and in many the response is slow, taking up to 52 weeks to determine response. Furthermore, response rates remain low.
"So it takes a long time, and you certainly don’t get 100% response rates, she said.
The guidelines, developed by a core working group that conducted the literature reviews and wrote scenarios, and by a task force panel of international experts in rheumatology, nephrology, and pathology who voted on the scenarios, will be published in Arthritis Care & Research, most likely in the spring, Dr. Hahn said. Currently they are pending approval by the ACR Guidelines Subcommittee, Quality of Care Committee, and Board of Directors.
Dr. Hahn, however, is already looking forward to new advances in the treatment of lupus nephritis and to the next guidelines update.
"I can’t wait to have something that’s better, so in another decade when we write guidelines, we can talk to you about 80% to 100% response rates," she said.
The lupus nephritis guideline project was sponsored by the ACR via a competitive grant mechanism. Of the 39 working group and task force members, 16 declared potential conflicts of interest, including relationships with Aspreva, Bristol-Myers Squibb, Genentech, and Human Genome Sciences.
FROM THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF RHEUMATOLOGY
FDA Steps Up Cancer Surveillance of TNF Blockers
The Food and Drug Administration has asked manufacturers of tumor necrosis factor blockers to enhance their follow-up of pediatric and young adult patients who develop malignancies while on these treatments.
The agency also is advising health care professionals to “remain vigilant” for malignancies in these patients, according to a statement issued by the FDA Nov. 4.
Manufacturers of TNF blockers have been asked by the FDA to report cases of malignancies in children, adolescents, and adults aged 30 years and younger treated with TNF blockers and to conduct in-depth follow-up of them. They also are asked to provide the agency with annual summaries and assessments of malignancies and data on the use of TNF blockers in these age groups.
“This type of safety surveillance is important for our improved understanding of malignancies in pediatric and young adult patients treated with TNF blockers, because it will allow FDA to more completely capture and analyze all reported malignancies based on more complete and consistent reports,” the agency said in its statement.
The FDA statement updates the agency’s ongoing safety review of TNF blockers and malignancies in children, adolescents, and young adults, and does not include any new cases or previously unrecognized risks associated with TNF blockers in this patient population.
The agency has previously issued reports about the increased risk of lymphomas and other cancers associated with the use of TNF blockers and malignancies in children and young adults in June 2008, August 2009, and most recently, April 2011.
TNF blockers are used to treat Crohn’s disease, ulcerative colitis, rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis, plaque psoriasis, and/or juvenile idiopathic arthritis. TNF blockers used in the United States are infliximab (Remicade), etanercept (Enbrel), adalimumab (Humira), certolizumab pegol (Cimzia), and golimumab (Simponi).
Health care professionals also should report these cases to the FDA’s Medwatch program.
The Food and Drug Administration has asked manufacturers of tumor necrosis factor blockers to enhance their follow-up of pediatric and young adult patients who develop malignancies while on these treatments.
The agency also is advising health care professionals to “remain vigilant” for malignancies in these patients, according to a statement issued by the FDA Nov. 4.
Manufacturers of TNF blockers have been asked by the FDA to report cases of malignancies in children, adolescents, and adults aged 30 years and younger treated with TNF blockers and to conduct in-depth follow-up of them. They also are asked to provide the agency with annual summaries and assessments of malignancies and data on the use of TNF blockers in these age groups.
“This type of safety surveillance is important for our improved understanding of malignancies in pediatric and young adult patients treated with TNF blockers, because it will allow FDA to more completely capture and analyze all reported malignancies based on more complete and consistent reports,” the agency said in its statement.
The FDA statement updates the agency’s ongoing safety review of TNF blockers and malignancies in children, adolescents, and young adults, and does not include any new cases or previously unrecognized risks associated with TNF blockers in this patient population.
The agency has previously issued reports about the increased risk of lymphomas and other cancers associated with the use of TNF blockers and malignancies in children and young adults in June 2008, August 2009, and most recently, April 2011.
TNF blockers are used to treat Crohn’s disease, ulcerative colitis, rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis, plaque psoriasis, and/or juvenile idiopathic arthritis. TNF blockers used in the United States are infliximab (Remicade), etanercept (Enbrel), adalimumab (Humira), certolizumab pegol (Cimzia), and golimumab (Simponi).
Health care professionals also should report these cases to the FDA’s Medwatch program.
The Food and Drug Administration has asked manufacturers of tumor necrosis factor blockers to enhance their follow-up of pediatric and young adult patients who develop malignancies while on these treatments.
The agency also is advising health care professionals to “remain vigilant” for malignancies in these patients, according to a statement issued by the FDA Nov. 4.
Manufacturers of TNF blockers have been asked by the FDA to report cases of malignancies in children, adolescents, and adults aged 30 years and younger treated with TNF blockers and to conduct in-depth follow-up of them. They also are asked to provide the agency with annual summaries and assessments of malignancies and data on the use of TNF blockers in these age groups.
“This type of safety surveillance is important for our improved understanding of malignancies in pediatric and young adult patients treated with TNF blockers, because it will allow FDA to more completely capture and analyze all reported malignancies based on more complete and consistent reports,” the agency said in its statement.
The FDA statement updates the agency’s ongoing safety review of TNF blockers and malignancies in children, adolescents, and young adults, and does not include any new cases or previously unrecognized risks associated with TNF blockers in this patient population.
The agency has previously issued reports about the increased risk of lymphomas and other cancers associated with the use of TNF blockers and malignancies in children and young adults in June 2008, August 2009, and most recently, April 2011.
TNF blockers are used to treat Crohn’s disease, ulcerative colitis, rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis, plaque psoriasis, and/or juvenile idiopathic arthritis. TNF blockers used in the United States are infliximab (Remicade), etanercept (Enbrel), adalimumab (Humira), certolizumab pegol (Cimzia), and golimumab (Simponi).
Health care professionals also should report these cases to the FDA’s Medwatch program.
Adding Methotrexate to Etanercept Boosts Psoriasis Efficacy
LISBON – Adding methotrexate to etanercept in patients with moderate to severe plaque psoriasis brings a bump in efficacy with no increase in serious adverse events, according to a large, double-blind, randomized clinical trial.
The multicenter trial included 478 patients on etanercept (Enbrel) at 50 mg twice weekly for 12 weeks, followed by once-weekly treatment thereafter. Participants were randomized to methotrexate or placebo from the outset. Methotrexate was dosed at 7.5 mg/week for the first 2 weeks, 10 mg/week for the next 2 weeks, and 15 mg/week after that.
The primary study end point was the proportion of patients attaining a PASI 75 response – that is, at least a 75% improvement over baseline scores on the Psoriasis Area and Severity Index – at week 24. The PASI 75 rate was 77.3% with dual therapy, significantly better than the 60.3% rate with etanercept alone, Dr. Alice B. Gottlieb reported at the annual congress of the European Academy of Dermatology and Venereology.
These results with combination therapy are in line with the enhanced efficacy seen with ustekinumab (Stelara) and other biologics targeting interleukin-23/Th17. But the combination therapy relies upon familiar medications that have been around a long time and have well-understood safety profiles, she noted.
The 24-week PASI 90 rate was also markedly better with dual therapy: 53.8%, compared with 34.2%. Seventy-two percent of patients on methotrexate plus etanercept were rated clear or almost clear on Physician’s Global Assessment, as were 53% on etanercept alone, according to Dr. Gottlieb, professor and chair of dermatology at Tufts Medical Center, Boston.
Adding methotrexate accelerated the time course of improvement. By week 12, 70% of patients on methotrexate plus etanercept had achieved a PASI 75 response, compared with 54% of those on etanercept alone.
Three serious adverse events occurred in each study arm. In the dual-therapy arm, there was one case each of bacterial pneumonia, lumbar spinal stenosis, and synovial cyst. In the etanercept-only arm, there was a case of acute MI, cholecystitis, and asthma.
Dr. Gottlieb disclosed that she serves as a consultant to Amgen, which sponsored the trial. In addition, she is a consultant and/or advisory board member for numerous other pharmaceutical companies.
LISBON – Adding methotrexate to etanercept in patients with moderate to severe plaque psoriasis brings a bump in efficacy with no increase in serious adverse events, according to a large, double-blind, randomized clinical trial.
The multicenter trial included 478 patients on etanercept (Enbrel) at 50 mg twice weekly for 12 weeks, followed by once-weekly treatment thereafter. Participants were randomized to methotrexate or placebo from the outset. Methotrexate was dosed at 7.5 mg/week for the first 2 weeks, 10 mg/week for the next 2 weeks, and 15 mg/week after that.
The primary study end point was the proportion of patients attaining a PASI 75 response – that is, at least a 75% improvement over baseline scores on the Psoriasis Area and Severity Index – at week 24. The PASI 75 rate was 77.3% with dual therapy, significantly better than the 60.3% rate with etanercept alone, Dr. Alice B. Gottlieb reported at the annual congress of the European Academy of Dermatology and Venereology.
These results with combination therapy are in line with the enhanced efficacy seen with ustekinumab (Stelara) and other biologics targeting interleukin-23/Th17. But the combination therapy relies upon familiar medications that have been around a long time and have well-understood safety profiles, she noted.
The 24-week PASI 90 rate was also markedly better with dual therapy: 53.8%, compared with 34.2%. Seventy-two percent of patients on methotrexate plus etanercept were rated clear or almost clear on Physician’s Global Assessment, as were 53% on etanercept alone, according to Dr. Gottlieb, professor and chair of dermatology at Tufts Medical Center, Boston.
Adding methotrexate accelerated the time course of improvement. By week 12, 70% of patients on methotrexate plus etanercept had achieved a PASI 75 response, compared with 54% of those on etanercept alone.
Three serious adverse events occurred in each study arm. In the dual-therapy arm, there was one case each of bacterial pneumonia, lumbar spinal stenosis, and synovial cyst. In the etanercept-only arm, there was a case of acute MI, cholecystitis, and asthma.
Dr. Gottlieb disclosed that she serves as a consultant to Amgen, which sponsored the trial. In addition, she is a consultant and/or advisory board member for numerous other pharmaceutical companies.
LISBON – Adding methotrexate to etanercept in patients with moderate to severe plaque psoriasis brings a bump in efficacy with no increase in serious adverse events, according to a large, double-blind, randomized clinical trial.
The multicenter trial included 478 patients on etanercept (Enbrel) at 50 mg twice weekly for 12 weeks, followed by once-weekly treatment thereafter. Participants were randomized to methotrexate or placebo from the outset. Methotrexate was dosed at 7.5 mg/week for the first 2 weeks, 10 mg/week for the next 2 weeks, and 15 mg/week after that.
The primary study end point was the proportion of patients attaining a PASI 75 response – that is, at least a 75% improvement over baseline scores on the Psoriasis Area and Severity Index – at week 24. The PASI 75 rate was 77.3% with dual therapy, significantly better than the 60.3% rate with etanercept alone, Dr. Alice B. Gottlieb reported at the annual congress of the European Academy of Dermatology and Venereology.
These results with combination therapy are in line with the enhanced efficacy seen with ustekinumab (Stelara) and other biologics targeting interleukin-23/Th17. But the combination therapy relies upon familiar medications that have been around a long time and have well-understood safety profiles, she noted.
The 24-week PASI 90 rate was also markedly better with dual therapy: 53.8%, compared with 34.2%. Seventy-two percent of patients on methotrexate plus etanercept were rated clear or almost clear on Physician’s Global Assessment, as were 53% on etanercept alone, according to Dr. Gottlieb, professor and chair of dermatology at Tufts Medical Center, Boston.
Adding methotrexate accelerated the time course of improvement. By week 12, 70% of patients on methotrexate plus etanercept had achieved a PASI 75 response, compared with 54% of those on etanercept alone.
Three serious adverse events occurred in each study arm. In the dual-therapy arm, there was one case each of bacterial pneumonia, lumbar spinal stenosis, and synovial cyst. In the etanercept-only arm, there was a case of acute MI, cholecystitis, and asthma.
Dr. Gottlieb disclosed that she serves as a consultant to Amgen, which sponsored the trial. In addition, she is a consultant and/or advisory board member for numerous other pharmaceutical companies.
FROM THE ANNUAL CONGRESS OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY
Major Finding: Adding methotrexate to etanercept resulted in a 77% PASI 75 response rate at 24 weeks, compared with 60% with etanercept alone.
Data Source: A double-blind, randomized, placebo-controlled clinical trial involving 478 patients with moderate to severe plaque psoriasis.
Disclosures: Dr. Gottlieb disclosed that she serves as a consultant to Amgen, which sponsored the trial. In addition, she is a consultant and/or advisory board member for numerous other pharmaceutical companies.