User login
Time to end direct-to-consumer ads, says physician
One has to be living off the grid to not be bombarded with direct-to-consumer (DTC) pharmaceutical advertising. Since 1997, when the Food and Drug Administration eased restrictions on this prohibition and allowed pharmaceutical companies to promote prescription-only medications to the public, there has been a deluge of ads in magazines, on the Internet, and, most annoying, on commercial television.
These television ads are quite formulaic:
We are initially introduced to a number of highly functioning patients (typically actors) who are engaged in rewarding pursuits. A voiceover narration then presents the pharmaceutical to be promoted, suggesting (not so subtly) to consumers that taking the advertised drug will improve one’s disease outlook or quality of life such that they too, just like the actors in the minidrama, can lead such highly productive lives.
The potential best-case scenarios of these new treatments may be stated. There then follows a litany of side effects – some of them life threatening – warnings, and contraindications. We’re again treated to another 5 or 10 seconds of patients leading “the good life,” and almost all of the ads end with the narrator concluding: “Ask your doctor (sometimes ‘provider’) if _____ is right for you.”
Americans spend more money on their prescriptions than do citizens of any other highly developed nation. I have personally heard from patients who get their prescriptions from other countries, where they are more affordable. These patients will also cut their pills in half or take a medication every other day instead of every day, to economize on drug costs.
Another “trick” they use to save money – and I have heard pharmacists and pharmaceutical reps themselves recommend this – is to ask for a higher dose of a medication, usually double, and then use a pill cutter to divide a tablet in half, thus making their prescription last twice as long. Why do Americans have to resort to such “workarounds”?
Many of the medications advertised are for relatively rare conditions, such as thyroid eye disease or myasthenia gravis (which affects up to about 60,000 patients in the United States). Why not spend these advertising dollars on programs to make drugs taken by the millions of Americans with common conditions (for example, hypertension, diabetes, heart failure) more affordable?
Very often the television ads contain medical jargon, such as: “If you have the EGFR mutation, or if your cancer is HER2 negative ...”
Do most patients truly understand what these terms mean? And what happens when a patient’s physician doesn’t prescribe a medication that a patient has seen on TV and asks for, or when the physician believes that a generic (nonadvertised) medication might work just as well? This creates conflict and potential discord, adversely affecting the doctor-patient relationship.
An oncologist colleague related to me that he often has to spend time correcting patients’ misperceptions of potential miracle cures offered by these ads, and that several patients have left his practice because he would not prescribe a drug they saw advertised.
Further, while these ads urge patients to try expensive “newest and latest” treatments, pharmacy benefit plans are working with health care insurance conglomerates to reduce costs of pharmaceuticals.
How does this juxtaposition of opposing forces make any sense?
It is time for us to put an end to DTC advertising, at least on television. It will require legislative action by our federal government to end this practice (legal, by the way, only in the United States and New Zealand), and hence the willingness of our politicians to get behind legislation to do so.
Just as a law was passed to prohibit tobacco advertising on television, so should a law be passed to regulate DTC pharmaceutical advertising.
The time to end DTC advertising has come!
Lloyd Alterman, MD, is a retired physician and chairman of the New Jersey Universal Healthcare Coalition. He disclosed having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
One has to be living off the grid to not be bombarded with direct-to-consumer (DTC) pharmaceutical advertising. Since 1997, when the Food and Drug Administration eased restrictions on this prohibition and allowed pharmaceutical companies to promote prescription-only medications to the public, there has been a deluge of ads in magazines, on the Internet, and, most annoying, on commercial television.
These television ads are quite formulaic:
We are initially introduced to a number of highly functioning patients (typically actors) who are engaged in rewarding pursuits. A voiceover narration then presents the pharmaceutical to be promoted, suggesting (not so subtly) to consumers that taking the advertised drug will improve one’s disease outlook or quality of life such that they too, just like the actors in the minidrama, can lead such highly productive lives.
The potential best-case scenarios of these new treatments may be stated. There then follows a litany of side effects – some of them life threatening – warnings, and contraindications. We’re again treated to another 5 or 10 seconds of patients leading “the good life,” and almost all of the ads end with the narrator concluding: “Ask your doctor (sometimes ‘provider’) if _____ is right for you.”
Americans spend more money on their prescriptions than do citizens of any other highly developed nation. I have personally heard from patients who get their prescriptions from other countries, where they are more affordable. These patients will also cut their pills in half or take a medication every other day instead of every day, to economize on drug costs.
Another “trick” they use to save money – and I have heard pharmacists and pharmaceutical reps themselves recommend this – is to ask for a higher dose of a medication, usually double, and then use a pill cutter to divide a tablet in half, thus making their prescription last twice as long. Why do Americans have to resort to such “workarounds”?
Many of the medications advertised are for relatively rare conditions, such as thyroid eye disease or myasthenia gravis (which affects up to about 60,000 patients in the United States). Why not spend these advertising dollars on programs to make drugs taken by the millions of Americans with common conditions (for example, hypertension, diabetes, heart failure) more affordable?
Very often the television ads contain medical jargon, such as: “If you have the EGFR mutation, or if your cancer is HER2 negative ...”
Do most patients truly understand what these terms mean? And what happens when a patient’s physician doesn’t prescribe a medication that a patient has seen on TV and asks for, or when the physician believes that a generic (nonadvertised) medication might work just as well? This creates conflict and potential discord, adversely affecting the doctor-patient relationship.
An oncologist colleague related to me that he often has to spend time correcting patients’ misperceptions of potential miracle cures offered by these ads, and that several patients have left his practice because he would not prescribe a drug they saw advertised.
Further, while these ads urge patients to try expensive “newest and latest” treatments, pharmacy benefit plans are working with health care insurance conglomerates to reduce costs of pharmaceuticals.
How does this juxtaposition of opposing forces make any sense?
It is time for us to put an end to DTC advertising, at least on television. It will require legislative action by our federal government to end this practice (legal, by the way, only in the United States and New Zealand), and hence the willingness of our politicians to get behind legislation to do so.
Just as a law was passed to prohibit tobacco advertising on television, so should a law be passed to regulate DTC pharmaceutical advertising.
The time to end DTC advertising has come!
Lloyd Alterman, MD, is a retired physician and chairman of the New Jersey Universal Healthcare Coalition. He disclosed having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
One has to be living off the grid to not be bombarded with direct-to-consumer (DTC) pharmaceutical advertising. Since 1997, when the Food and Drug Administration eased restrictions on this prohibition and allowed pharmaceutical companies to promote prescription-only medications to the public, there has been a deluge of ads in magazines, on the Internet, and, most annoying, on commercial television.
These television ads are quite formulaic:
We are initially introduced to a number of highly functioning patients (typically actors) who are engaged in rewarding pursuits. A voiceover narration then presents the pharmaceutical to be promoted, suggesting (not so subtly) to consumers that taking the advertised drug will improve one’s disease outlook or quality of life such that they too, just like the actors in the minidrama, can lead such highly productive lives.
The potential best-case scenarios of these new treatments may be stated. There then follows a litany of side effects – some of them life threatening – warnings, and contraindications. We’re again treated to another 5 or 10 seconds of patients leading “the good life,” and almost all of the ads end with the narrator concluding: “Ask your doctor (sometimes ‘provider’) if _____ is right for you.”
Americans spend more money on their prescriptions than do citizens of any other highly developed nation. I have personally heard from patients who get their prescriptions from other countries, where they are more affordable. These patients will also cut their pills in half or take a medication every other day instead of every day, to economize on drug costs.
Another “trick” they use to save money – and I have heard pharmacists and pharmaceutical reps themselves recommend this – is to ask for a higher dose of a medication, usually double, and then use a pill cutter to divide a tablet in half, thus making their prescription last twice as long. Why do Americans have to resort to such “workarounds”?
Many of the medications advertised are for relatively rare conditions, such as thyroid eye disease or myasthenia gravis (which affects up to about 60,000 patients in the United States). Why not spend these advertising dollars on programs to make drugs taken by the millions of Americans with common conditions (for example, hypertension, diabetes, heart failure) more affordable?
Very often the television ads contain medical jargon, such as: “If you have the EGFR mutation, or if your cancer is HER2 negative ...”
Do most patients truly understand what these terms mean? And what happens when a patient’s physician doesn’t prescribe a medication that a patient has seen on TV and asks for, or when the physician believes that a generic (nonadvertised) medication might work just as well? This creates conflict and potential discord, adversely affecting the doctor-patient relationship.
An oncologist colleague related to me that he often has to spend time correcting patients’ misperceptions of potential miracle cures offered by these ads, and that several patients have left his practice because he would not prescribe a drug they saw advertised.
Further, while these ads urge patients to try expensive “newest and latest” treatments, pharmacy benefit plans are working with health care insurance conglomerates to reduce costs of pharmaceuticals.
How does this juxtaposition of opposing forces make any sense?
It is time for us to put an end to DTC advertising, at least on television. It will require legislative action by our federal government to end this practice (legal, by the way, only in the United States and New Zealand), and hence the willingness of our politicians to get behind legislation to do so.
Just as a law was passed to prohibit tobacco advertising on television, so should a law be passed to regulate DTC pharmaceutical advertising.
The time to end DTC advertising has come!
Lloyd Alterman, MD, is a retired physician and chairman of the New Jersey Universal Healthcare Coalition. He disclosed having no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Chaperones for physical exams? Doctors and patients weigh in
News of physicians accused or convicted of sexually inappropriate behavior toward patients during medical exams has been frequent recently. And patient advocates have brought up the fact that many patients are uncomfortable during sensitive exams.
As a result, more
For some institutions, the movement has been toward mandating chaperones. Chaperones can be present for any examination of what are often referred to as sensitive areas, such as breasts, genitalia, and the perianal area. For example, Yale University Health requires patients to use a medical staff member as a chaperone for all sensitive exams except in emergency situations. UCLA Health, on the other hand, allows any adult or child aged 12 years or older to decline a chaperone if they wish to do so. Michigan Medicine provides a chaperone on request, although a patient can opt out.
Many physicians fully support the concept. “If a patient requests a chaperone I think we all, as practitioners, should honor that request respectfully,” said Shieva Ghofrany, MD, an ob.gyn. practicing in Stamford, Conn., and cofounder of tribecalledv, a women’s health knowledge platform. “It’s a clear sign a patient wants to ensure that they feel safe and comfortable.”
However, using a chaperone can be challenging for many primary care physicians and specialists in terms of logistics. Should a chaperone’s job be purely observational? Or should the chaperone also be a medical professional who assists during the procedure? How, specifically, should a chaperone perform their duty during an exam? Where should the chaperone stand? What about legal and ethical ramifications?
Who should be a chaperone?
The role of a chaperone is sometimes vaguely defined. It’s logical that a chaperone should have sufficient medical knowledge. Could it be a medical student? Should it be a nurse or another doctor? Would a patient representative suffice even if they aren’t a direct-care clinician?
There are no set standards about who can assume the role. However, the American Medical Association recommends that a patient chaperone be an authorized member of a health care team. For many patients, this is vital.
“As a survivor of sexual assault by an ob.gyn. while I was pregnant, I can assure you that the heinous crime committed against me and hundreds of other women would have been prevented had there been another medical professional in the room,” said Laurie Kanyok, founder of a dance company in New York City, whose former doctor was recently convicted of federal sex abuse charges and is scheduled to be sentenced in July 2023. “The chaperone should be a physician assistant, nurse or medical professional. Qualified professionals better understand the nature and protocols of an exam.”
For children and adolescents, allowing a parent to be a chaperone might be a logical and comforting choice. However, a new British study found that it’s not appropriate.
Study authors Rebecca Jane Moon and Justin Huw Davies looked at pubertal staging–related exams and contend that a chaperone should always be impartial, and a parent’s presence could conceivably open up a physician to false charges of misconduct.
The solution: A parent should have the option be present during any physical exam of their child, with a medical professional additionally acting as chaperone. This can also work for any patient who wishes to bring a trusted friend or family member to their medical appointments.
How does a chaperoned exam work?
A research team from the Medical College of Wisconsin and Louisiana State University Health conducted a recent systematic review of patient and clinician perspectives on carrying out a chaperoned urology exam. The review found that the protocol could use improvement. For example, it was reported that over one-quarter of patients didn’t feel comfortable asking for a chaperone if they were not offered one. “Patients should not have to request this,” said Diana Londoño, MD, a board-certified urologist and assistant clinical professor at City of Hope National Medical Center in Duarte, Calif.
The researchers also found that 93% of female patients preferred a chaperone of the same gender, whereas male patients were split on this issue. Key duties of a chaperone should be to ensure privacy, help interpret instructions from doctor to patient, and continually clarify that consent is being requested as a sensitive exam proceeds, the researchers report. Although clinical practice standards for chaperones aren’t uniform, keeping these important points in mind can easily be adopted by any health care provider.
Many doctors do follow their own set guidelines. “Often, we have our medical assistants be our chaperones,” explained Dr. Ghofrany. “Ideally they ‘room’ the patient – take vitals and ask the patient what specific concerns they would like to address. This helps with exam set-up.
“For example, if a patient has a breast concern versus a concern about their genital area, this would change what drapes are used. The medical assistant would then stand near me if they are helping with a Pap smear or cultures, or they may stand by the patient’s head at the bedside and offer support if needed. Some patients want to hold someone’s hand during an uncomfortable exam.”
The issue of positioning is important. A patient may feel very uneasy if it appears that the chaperone is looking at their body from the doctor’s angle, negating the point of reassurance. The key is to explain before an exam exactly how a chaperone is needed during the exam itself and whether the chaperone is a medical assistant or nurse.
“Chaperones usually stand next to me on either side or on the side of the patient facing me, depending on if they are immediately assisting me or helping the patient,” said Dr. Londoño. If a chaperone will be moving back and forth during the exam, that should be conveyed as well. For virtual visits, a chaperone can act as a third party and be present on a split screen, a process Michigan Medicine uses.
It’s also important to inform patients that a chaperone can step in and stop an exam at any point, both at the patient’s request or because of any observation of inappropriate physician behavior. Understanding this can help reluctant patients feel less worry.
When Christian Miller was diagnosed with a sleep disorder, his doctor suggested having a chaperone present during his physical exams. At first, he was apprehensive but then found the experience to be very positive. “Having someone there with me gave me an extra measure of assurance that nothing untoward would happen,” he said. “I found that having a chaperone was reassuring. The chaperone was not intrusive but stood close enough to ensure everything went smoothly and respectfully.”
Do chaperones help protect physicians legally?
Some states mandate that chaperones be present by patient request during sensitive exams, such as Texas and Oregon. For the most part, though, physicians have no legal obligation to offer chaperones, although it could be in their best interest to do so.
Ob.gyn. is one of the most litigated specialties, for example, so these physicians may find chaperones can play a key role in averting lawsuits. According to data from Physicians’ Insurance, having a reliable witness in an exam room can mitigate claims, and doctors should follow a clear practice policy regarding chaperones for all patients to further reduce liability.
Another advantage to having a chaperone present: protection for a doctor against a problematic patient. The risk for assault or aggression during an exam can of course go both ways. It’s important to be able to prove any patient misconduct through a witness, and a chaperone can lessen the prospect of violence in any form. “Having a chaperone in an exam room is about protecting the doctors as well as the patient,” said Ms. Kanyok.
A chaperone can also defuse ethical dilemmas. Consider a patient who refuses to allow a chaperone in an exam, but a chaperone is required by the physician’s health care organization. Asking a patient to state their reasons for wanting privacy with the chaperone present before the actual exam can help document the patient’s wish respectfully as well as protect a physician and organization from any potential liability if the patient refuses the exam altogether or if an exception for the patient is made.
Making a chaperone policy work best
- Have your staff inform patients of a medical chaperone policy at the time an appointment is made. Have fact sheets available for the patient before appointments fully outlining your policy.
- Inform the chaperone fully about the details of the exam and the patient’s case prior to the exam, in accordance with HIPAA.
- The chaperone can introduce themselves to the patient in the waiting room or exam room before you enter the room. The chaperone can go over the policy again verbally and answer any questions the patient may have initially. You can then clarify whether the patient understands your chaperone policy when you come in.
- Document, document, document. Write down who the chaperone was for each exam in patient notes and note the details of any interactions that are significant, such as patient questions or conflicts.
- Practice respect, above all. “A patient’s safety and level of comfort should be prioritized,” said Ms. Kanyok.
A version of this article first appeared on Medscape.com.
News of physicians accused or convicted of sexually inappropriate behavior toward patients during medical exams has been frequent recently. And patient advocates have brought up the fact that many patients are uncomfortable during sensitive exams.
As a result, more
For some institutions, the movement has been toward mandating chaperones. Chaperones can be present for any examination of what are often referred to as sensitive areas, such as breasts, genitalia, and the perianal area. For example, Yale University Health requires patients to use a medical staff member as a chaperone for all sensitive exams except in emergency situations. UCLA Health, on the other hand, allows any adult or child aged 12 years or older to decline a chaperone if they wish to do so. Michigan Medicine provides a chaperone on request, although a patient can opt out.
Many physicians fully support the concept. “If a patient requests a chaperone I think we all, as practitioners, should honor that request respectfully,” said Shieva Ghofrany, MD, an ob.gyn. practicing in Stamford, Conn., and cofounder of tribecalledv, a women’s health knowledge platform. “It’s a clear sign a patient wants to ensure that they feel safe and comfortable.”
However, using a chaperone can be challenging for many primary care physicians and specialists in terms of logistics. Should a chaperone’s job be purely observational? Or should the chaperone also be a medical professional who assists during the procedure? How, specifically, should a chaperone perform their duty during an exam? Where should the chaperone stand? What about legal and ethical ramifications?
Who should be a chaperone?
The role of a chaperone is sometimes vaguely defined. It’s logical that a chaperone should have sufficient medical knowledge. Could it be a medical student? Should it be a nurse or another doctor? Would a patient representative suffice even if they aren’t a direct-care clinician?
There are no set standards about who can assume the role. However, the American Medical Association recommends that a patient chaperone be an authorized member of a health care team. For many patients, this is vital.
“As a survivor of sexual assault by an ob.gyn. while I was pregnant, I can assure you that the heinous crime committed against me and hundreds of other women would have been prevented had there been another medical professional in the room,” said Laurie Kanyok, founder of a dance company in New York City, whose former doctor was recently convicted of federal sex abuse charges and is scheduled to be sentenced in July 2023. “The chaperone should be a physician assistant, nurse or medical professional. Qualified professionals better understand the nature and protocols of an exam.”
For children and adolescents, allowing a parent to be a chaperone might be a logical and comforting choice. However, a new British study found that it’s not appropriate.
Study authors Rebecca Jane Moon and Justin Huw Davies looked at pubertal staging–related exams and contend that a chaperone should always be impartial, and a parent’s presence could conceivably open up a physician to false charges of misconduct.
The solution: A parent should have the option be present during any physical exam of their child, with a medical professional additionally acting as chaperone. This can also work for any patient who wishes to bring a trusted friend or family member to their medical appointments.
How does a chaperoned exam work?
A research team from the Medical College of Wisconsin and Louisiana State University Health conducted a recent systematic review of patient and clinician perspectives on carrying out a chaperoned urology exam. The review found that the protocol could use improvement. For example, it was reported that over one-quarter of patients didn’t feel comfortable asking for a chaperone if they were not offered one. “Patients should not have to request this,” said Diana Londoño, MD, a board-certified urologist and assistant clinical professor at City of Hope National Medical Center in Duarte, Calif.
The researchers also found that 93% of female patients preferred a chaperone of the same gender, whereas male patients were split on this issue. Key duties of a chaperone should be to ensure privacy, help interpret instructions from doctor to patient, and continually clarify that consent is being requested as a sensitive exam proceeds, the researchers report. Although clinical practice standards for chaperones aren’t uniform, keeping these important points in mind can easily be adopted by any health care provider.
Many doctors do follow their own set guidelines. “Often, we have our medical assistants be our chaperones,” explained Dr. Ghofrany. “Ideally they ‘room’ the patient – take vitals and ask the patient what specific concerns they would like to address. This helps with exam set-up.
“For example, if a patient has a breast concern versus a concern about their genital area, this would change what drapes are used. The medical assistant would then stand near me if they are helping with a Pap smear or cultures, or they may stand by the patient’s head at the bedside and offer support if needed. Some patients want to hold someone’s hand during an uncomfortable exam.”
The issue of positioning is important. A patient may feel very uneasy if it appears that the chaperone is looking at their body from the doctor’s angle, negating the point of reassurance. The key is to explain before an exam exactly how a chaperone is needed during the exam itself and whether the chaperone is a medical assistant or nurse.
“Chaperones usually stand next to me on either side or on the side of the patient facing me, depending on if they are immediately assisting me or helping the patient,” said Dr. Londoño. If a chaperone will be moving back and forth during the exam, that should be conveyed as well. For virtual visits, a chaperone can act as a third party and be present on a split screen, a process Michigan Medicine uses.
It’s also important to inform patients that a chaperone can step in and stop an exam at any point, both at the patient’s request or because of any observation of inappropriate physician behavior. Understanding this can help reluctant patients feel less worry.
When Christian Miller was diagnosed with a sleep disorder, his doctor suggested having a chaperone present during his physical exams. At first, he was apprehensive but then found the experience to be very positive. “Having someone there with me gave me an extra measure of assurance that nothing untoward would happen,” he said. “I found that having a chaperone was reassuring. The chaperone was not intrusive but stood close enough to ensure everything went smoothly and respectfully.”
Do chaperones help protect physicians legally?
Some states mandate that chaperones be present by patient request during sensitive exams, such as Texas and Oregon. For the most part, though, physicians have no legal obligation to offer chaperones, although it could be in their best interest to do so.
Ob.gyn. is one of the most litigated specialties, for example, so these physicians may find chaperones can play a key role in averting lawsuits. According to data from Physicians’ Insurance, having a reliable witness in an exam room can mitigate claims, and doctors should follow a clear practice policy regarding chaperones for all patients to further reduce liability.
Another advantage to having a chaperone present: protection for a doctor against a problematic patient. The risk for assault or aggression during an exam can of course go both ways. It’s important to be able to prove any patient misconduct through a witness, and a chaperone can lessen the prospect of violence in any form. “Having a chaperone in an exam room is about protecting the doctors as well as the patient,” said Ms. Kanyok.
A chaperone can also defuse ethical dilemmas. Consider a patient who refuses to allow a chaperone in an exam, but a chaperone is required by the physician’s health care organization. Asking a patient to state their reasons for wanting privacy with the chaperone present before the actual exam can help document the patient’s wish respectfully as well as protect a physician and organization from any potential liability if the patient refuses the exam altogether or if an exception for the patient is made.
Making a chaperone policy work best
- Have your staff inform patients of a medical chaperone policy at the time an appointment is made. Have fact sheets available for the patient before appointments fully outlining your policy.
- Inform the chaperone fully about the details of the exam and the patient’s case prior to the exam, in accordance with HIPAA.
- The chaperone can introduce themselves to the patient in the waiting room or exam room before you enter the room. The chaperone can go over the policy again verbally and answer any questions the patient may have initially. You can then clarify whether the patient understands your chaperone policy when you come in.
- Document, document, document. Write down who the chaperone was for each exam in patient notes and note the details of any interactions that are significant, such as patient questions or conflicts.
- Practice respect, above all. “A patient’s safety and level of comfort should be prioritized,” said Ms. Kanyok.
A version of this article first appeared on Medscape.com.
News of physicians accused or convicted of sexually inappropriate behavior toward patients during medical exams has been frequent recently. And patient advocates have brought up the fact that many patients are uncomfortable during sensitive exams.
As a result, more
For some institutions, the movement has been toward mandating chaperones. Chaperones can be present for any examination of what are often referred to as sensitive areas, such as breasts, genitalia, and the perianal area. For example, Yale University Health requires patients to use a medical staff member as a chaperone for all sensitive exams except in emergency situations. UCLA Health, on the other hand, allows any adult or child aged 12 years or older to decline a chaperone if they wish to do so. Michigan Medicine provides a chaperone on request, although a patient can opt out.
Many physicians fully support the concept. “If a patient requests a chaperone I think we all, as practitioners, should honor that request respectfully,” said Shieva Ghofrany, MD, an ob.gyn. practicing in Stamford, Conn., and cofounder of tribecalledv, a women’s health knowledge platform. “It’s a clear sign a patient wants to ensure that they feel safe and comfortable.”
However, using a chaperone can be challenging for many primary care physicians and specialists in terms of logistics. Should a chaperone’s job be purely observational? Or should the chaperone also be a medical professional who assists during the procedure? How, specifically, should a chaperone perform their duty during an exam? Where should the chaperone stand? What about legal and ethical ramifications?
Who should be a chaperone?
The role of a chaperone is sometimes vaguely defined. It’s logical that a chaperone should have sufficient medical knowledge. Could it be a medical student? Should it be a nurse or another doctor? Would a patient representative suffice even if they aren’t a direct-care clinician?
There are no set standards about who can assume the role. However, the American Medical Association recommends that a patient chaperone be an authorized member of a health care team. For many patients, this is vital.
“As a survivor of sexual assault by an ob.gyn. while I was pregnant, I can assure you that the heinous crime committed against me and hundreds of other women would have been prevented had there been another medical professional in the room,” said Laurie Kanyok, founder of a dance company in New York City, whose former doctor was recently convicted of federal sex abuse charges and is scheduled to be sentenced in July 2023. “The chaperone should be a physician assistant, nurse or medical professional. Qualified professionals better understand the nature and protocols of an exam.”
For children and adolescents, allowing a parent to be a chaperone might be a logical and comforting choice. However, a new British study found that it’s not appropriate.
Study authors Rebecca Jane Moon and Justin Huw Davies looked at pubertal staging–related exams and contend that a chaperone should always be impartial, and a parent’s presence could conceivably open up a physician to false charges of misconduct.
The solution: A parent should have the option be present during any physical exam of their child, with a medical professional additionally acting as chaperone. This can also work for any patient who wishes to bring a trusted friend or family member to their medical appointments.
How does a chaperoned exam work?
A research team from the Medical College of Wisconsin and Louisiana State University Health conducted a recent systematic review of patient and clinician perspectives on carrying out a chaperoned urology exam. The review found that the protocol could use improvement. For example, it was reported that over one-quarter of patients didn’t feel comfortable asking for a chaperone if they were not offered one. “Patients should not have to request this,” said Diana Londoño, MD, a board-certified urologist and assistant clinical professor at City of Hope National Medical Center in Duarte, Calif.
The researchers also found that 93% of female patients preferred a chaperone of the same gender, whereas male patients were split on this issue. Key duties of a chaperone should be to ensure privacy, help interpret instructions from doctor to patient, and continually clarify that consent is being requested as a sensitive exam proceeds, the researchers report. Although clinical practice standards for chaperones aren’t uniform, keeping these important points in mind can easily be adopted by any health care provider.
Many doctors do follow their own set guidelines. “Often, we have our medical assistants be our chaperones,” explained Dr. Ghofrany. “Ideally they ‘room’ the patient – take vitals and ask the patient what specific concerns they would like to address. This helps with exam set-up.
“For example, if a patient has a breast concern versus a concern about their genital area, this would change what drapes are used. The medical assistant would then stand near me if they are helping with a Pap smear or cultures, or they may stand by the patient’s head at the bedside and offer support if needed. Some patients want to hold someone’s hand during an uncomfortable exam.”
The issue of positioning is important. A patient may feel very uneasy if it appears that the chaperone is looking at their body from the doctor’s angle, negating the point of reassurance. The key is to explain before an exam exactly how a chaperone is needed during the exam itself and whether the chaperone is a medical assistant or nurse.
“Chaperones usually stand next to me on either side or on the side of the patient facing me, depending on if they are immediately assisting me or helping the patient,” said Dr. Londoño. If a chaperone will be moving back and forth during the exam, that should be conveyed as well. For virtual visits, a chaperone can act as a third party and be present on a split screen, a process Michigan Medicine uses.
It’s also important to inform patients that a chaperone can step in and stop an exam at any point, both at the patient’s request or because of any observation of inappropriate physician behavior. Understanding this can help reluctant patients feel less worry.
When Christian Miller was diagnosed with a sleep disorder, his doctor suggested having a chaperone present during his physical exams. At first, he was apprehensive but then found the experience to be very positive. “Having someone there with me gave me an extra measure of assurance that nothing untoward would happen,” he said. “I found that having a chaperone was reassuring. The chaperone was not intrusive but stood close enough to ensure everything went smoothly and respectfully.”
Do chaperones help protect physicians legally?
Some states mandate that chaperones be present by patient request during sensitive exams, such as Texas and Oregon. For the most part, though, physicians have no legal obligation to offer chaperones, although it could be in their best interest to do so.
Ob.gyn. is one of the most litigated specialties, for example, so these physicians may find chaperones can play a key role in averting lawsuits. According to data from Physicians’ Insurance, having a reliable witness in an exam room can mitigate claims, and doctors should follow a clear practice policy regarding chaperones for all patients to further reduce liability.
Another advantage to having a chaperone present: protection for a doctor against a problematic patient. The risk for assault or aggression during an exam can of course go both ways. It’s important to be able to prove any patient misconduct through a witness, and a chaperone can lessen the prospect of violence in any form. “Having a chaperone in an exam room is about protecting the doctors as well as the patient,” said Ms. Kanyok.
A chaperone can also defuse ethical dilemmas. Consider a patient who refuses to allow a chaperone in an exam, but a chaperone is required by the physician’s health care organization. Asking a patient to state their reasons for wanting privacy with the chaperone present before the actual exam can help document the patient’s wish respectfully as well as protect a physician and organization from any potential liability if the patient refuses the exam altogether or if an exception for the patient is made.
Making a chaperone policy work best
- Have your staff inform patients of a medical chaperone policy at the time an appointment is made. Have fact sheets available for the patient before appointments fully outlining your policy.
- Inform the chaperone fully about the details of the exam and the patient’s case prior to the exam, in accordance with HIPAA.
- The chaperone can introduce themselves to the patient in the waiting room or exam room before you enter the room. The chaperone can go over the policy again verbally and answer any questions the patient may have initially. You can then clarify whether the patient understands your chaperone policy when you come in.
- Document, document, document. Write down who the chaperone was for each exam in patient notes and note the details of any interactions that are significant, such as patient questions or conflicts.
- Practice respect, above all. “A patient’s safety and level of comfort should be prioritized,” said Ms. Kanyok.
A version of this article first appeared on Medscape.com.
Intermittent fasting vs. calorie counting for weight loss
BOSTON –
For the study, 57 overweight and obese participants with type 2 diabetes were randomly assigned to three different groups: The first group ate between noon and 8 p.m., the second was asked to reduce caloric intake by 25% of maintenance calories, and the third, a control group, continued eating normally.
The calorie-restriction group tracked intake on MyFitnessPal, an app that logs the calorie content of different foods. Both the intermittent-fasting and calorie-restriction groups were assigned a dietitian to help with adherence.
After 6 months, participants in the intermittent-fasting group lost about 4.3% of body weight – the equivalent of 10 pounds of weight loss for a person weighing 230 pounds – whereas participants in the calorie-restriction group lost about 2.5% of body weight.
The difference between the two groups was not significant, so one approach isn’t necessarily better than the other for weight loss.
“Let’s not think of this as an approach that’s better than calorie restriction,” William Yancy, MD, MHS, an internist and weight management specialist at Duke Lifestyle and Weight Management Center, Durham, N.C., said in an interview. “It’s an alternative approach to calorie restriction.”
Participants’ willingness to adhere to the diet likely accounted for the percentage difference between the groups, study author Vasiliki Pavlou, RDN, told this news organization. Ms. Pavlou presented the findings at the Nutrition 2023 conference.
“People that have type 2 diabetes, they’ve already been to the doctor, they’ve already been told to count calories,” said Ms. Pavlou, a doctoral student at the University of Illinois at Chicago. “There were many weeks where they came to us with nothing on MyFitnessPal and we’d have to encourage them to start tracking again.”
The intermittent-fasting group adhered to the eating time window 6 out of 7 days of the week, with a 1-hour grace period for the noon-to-8-p.m. window. In comparison, one-third of the calorie-restriction group didn’t stay within 200 calories of the goal, according to Ms. Pavlou.
That meant the fasting group cut about 100 calories more per day than the calorie-restriction group, which was reflected in their weight loss, Ms. Pavlou said.
A1c levels dropped by about 1% in both the intermittent-fasting and calorie-restriction groups – a meaningful decrease, said Dr. Yancy. “I think a 0.5% difference would have some clinical significance in terms of complications from diabetes,” he said. “So 1% would be even more clinically meaningful.”
However, fewer participants taking insulin in the calorie-restriction group could explain the difference, according to Ms. Pavlou. “Usually, when someone goes on insulin, their pancreas is already not functioning as well,” she said. “And it’s way harder to see improvements in their A1c and glycemic control.”
Up to 90% of people with type 2 diabetes are overweight or obese. Weight loss is one of the major components of type 2 diabetes care, according to the American Diabetes Association, and studies have shown that even a 5% reduction in body weight can reduce blood glucose concentration and A1c. Some studies suggest diabetes remission can occur after a 10% loss in body weight, but Dr. Yancy said it depends on the person.
“It depends on the individual, their metabolic situation, how long they’ve had diabetes, what kind of approach they’re following, maybe what medicines they’re taking,” Dr. Yancy said. “There’s a lot of different factors involved in remission.”
The study cohort generally had advanced diabetes and was taking a mix of medications, so the results might not be applicable to people with a more recent diabetes diagnosis, according to Ms. Pavlou.
Dr. Yancy said intermittent fasting could work well for the right person. The success of the approach could depend on a person’s eating habits and whether their meals usually fall outside the time-restricted window, or it could depend on how well a person follows rules, according to Dr. Yancy.
“Some people might not eat much after 8 o’clock, and some people might skip breakfast,” Dr. Yancy said. “And if that’s the case, then it’s not going to make a big impact on their weight probably.”
Medication is also an important consideration. Not eating can be dangerous for patients taking short-acting insulin or sulfonylureas, according to Dr. Yancy.
Ms. Pavlou said these findings show intermittent fasting is another option for patients with type 2 diabetes trying to lose weight. “If you’ve tried calorie counting, that’s not working for you or if you’re kind of burnt out, this is something else that you could try,” she said.
“We have a lot of patients that need to lose weight, and we have patients who respond differently to different approaches,” said Dr. Yancy. “So having various approaches is really valuable.”
The manuscript is currently under review at JAMA Internal Medicine, said Ms. Pavlou.
A version of this article appeared on Medscape.com.
BOSTON –
For the study, 57 overweight and obese participants with type 2 diabetes were randomly assigned to three different groups: The first group ate between noon and 8 p.m., the second was asked to reduce caloric intake by 25% of maintenance calories, and the third, a control group, continued eating normally.
The calorie-restriction group tracked intake on MyFitnessPal, an app that logs the calorie content of different foods. Both the intermittent-fasting and calorie-restriction groups were assigned a dietitian to help with adherence.
After 6 months, participants in the intermittent-fasting group lost about 4.3% of body weight – the equivalent of 10 pounds of weight loss for a person weighing 230 pounds – whereas participants in the calorie-restriction group lost about 2.5% of body weight.
The difference between the two groups was not significant, so one approach isn’t necessarily better than the other for weight loss.
“Let’s not think of this as an approach that’s better than calorie restriction,” William Yancy, MD, MHS, an internist and weight management specialist at Duke Lifestyle and Weight Management Center, Durham, N.C., said in an interview. “It’s an alternative approach to calorie restriction.”
Participants’ willingness to adhere to the diet likely accounted for the percentage difference between the groups, study author Vasiliki Pavlou, RDN, told this news organization. Ms. Pavlou presented the findings at the Nutrition 2023 conference.
“People that have type 2 diabetes, they’ve already been to the doctor, they’ve already been told to count calories,” said Ms. Pavlou, a doctoral student at the University of Illinois at Chicago. “There were many weeks where they came to us with nothing on MyFitnessPal and we’d have to encourage them to start tracking again.”
The intermittent-fasting group adhered to the eating time window 6 out of 7 days of the week, with a 1-hour grace period for the noon-to-8-p.m. window. In comparison, one-third of the calorie-restriction group didn’t stay within 200 calories of the goal, according to Ms. Pavlou.
That meant the fasting group cut about 100 calories more per day than the calorie-restriction group, which was reflected in their weight loss, Ms. Pavlou said.
A1c levels dropped by about 1% in both the intermittent-fasting and calorie-restriction groups – a meaningful decrease, said Dr. Yancy. “I think a 0.5% difference would have some clinical significance in terms of complications from diabetes,” he said. “So 1% would be even more clinically meaningful.”
However, fewer participants taking insulin in the calorie-restriction group could explain the difference, according to Ms. Pavlou. “Usually, when someone goes on insulin, their pancreas is already not functioning as well,” she said. “And it’s way harder to see improvements in their A1c and glycemic control.”
Up to 90% of people with type 2 diabetes are overweight or obese. Weight loss is one of the major components of type 2 diabetes care, according to the American Diabetes Association, and studies have shown that even a 5% reduction in body weight can reduce blood glucose concentration and A1c. Some studies suggest diabetes remission can occur after a 10% loss in body weight, but Dr. Yancy said it depends on the person.
“It depends on the individual, their metabolic situation, how long they’ve had diabetes, what kind of approach they’re following, maybe what medicines they’re taking,” Dr. Yancy said. “There’s a lot of different factors involved in remission.”
The study cohort generally had advanced diabetes and was taking a mix of medications, so the results might not be applicable to people with a more recent diabetes diagnosis, according to Ms. Pavlou.
Dr. Yancy said intermittent fasting could work well for the right person. The success of the approach could depend on a person’s eating habits and whether their meals usually fall outside the time-restricted window, or it could depend on how well a person follows rules, according to Dr. Yancy.
“Some people might not eat much after 8 o’clock, and some people might skip breakfast,” Dr. Yancy said. “And if that’s the case, then it’s not going to make a big impact on their weight probably.”
Medication is also an important consideration. Not eating can be dangerous for patients taking short-acting insulin or sulfonylureas, according to Dr. Yancy.
Ms. Pavlou said these findings show intermittent fasting is another option for patients with type 2 diabetes trying to lose weight. “If you’ve tried calorie counting, that’s not working for you or if you’re kind of burnt out, this is something else that you could try,” she said.
“We have a lot of patients that need to lose weight, and we have patients who respond differently to different approaches,” said Dr. Yancy. “So having various approaches is really valuable.”
The manuscript is currently under review at JAMA Internal Medicine, said Ms. Pavlou.
A version of this article appeared on Medscape.com.
BOSTON –
For the study, 57 overweight and obese participants with type 2 diabetes were randomly assigned to three different groups: The first group ate between noon and 8 p.m., the second was asked to reduce caloric intake by 25% of maintenance calories, and the third, a control group, continued eating normally.
The calorie-restriction group tracked intake on MyFitnessPal, an app that logs the calorie content of different foods. Both the intermittent-fasting and calorie-restriction groups were assigned a dietitian to help with adherence.
After 6 months, participants in the intermittent-fasting group lost about 4.3% of body weight – the equivalent of 10 pounds of weight loss for a person weighing 230 pounds – whereas participants in the calorie-restriction group lost about 2.5% of body weight.
The difference between the two groups was not significant, so one approach isn’t necessarily better than the other for weight loss.
“Let’s not think of this as an approach that’s better than calorie restriction,” William Yancy, MD, MHS, an internist and weight management specialist at Duke Lifestyle and Weight Management Center, Durham, N.C., said in an interview. “It’s an alternative approach to calorie restriction.”
Participants’ willingness to adhere to the diet likely accounted for the percentage difference between the groups, study author Vasiliki Pavlou, RDN, told this news organization. Ms. Pavlou presented the findings at the Nutrition 2023 conference.
“People that have type 2 diabetes, they’ve already been to the doctor, they’ve already been told to count calories,” said Ms. Pavlou, a doctoral student at the University of Illinois at Chicago. “There were many weeks where they came to us with nothing on MyFitnessPal and we’d have to encourage them to start tracking again.”
The intermittent-fasting group adhered to the eating time window 6 out of 7 days of the week, with a 1-hour grace period for the noon-to-8-p.m. window. In comparison, one-third of the calorie-restriction group didn’t stay within 200 calories of the goal, according to Ms. Pavlou.
That meant the fasting group cut about 100 calories more per day than the calorie-restriction group, which was reflected in their weight loss, Ms. Pavlou said.
A1c levels dropped by about 1% in both the intermittent-fasting and calorie-restriction groups – a meaningful decrease, said Dr. Yancy. “I think a 0.5% difference would have some clinical significance in terms of complications from diabetes,” he said. “So 1% would be even more clinically meaningful.”
However, fewer participants taking insulin in the calorie-restriction group could explain the difference, according to Ms. Pavlou. “Usually, when someone goes on insulin, their pancreas is already not functioning as well,” she said. “And it’s way harder to see improvements in their A1c and glycemic control.”
Up to 90% of people with type 2 diabetes are overweight or obese. Weight loss is one of the major components of type 2 diabetes care, according to the American Diabetes Association, and studies have shown that even a 5% reduction in body weight can reduce blood glucose concentration and A1c. Some studies suggest diabetes remission can occur after a 10% loss in body weight, but Dr. Yancy said it depends on the person.
“It depends on the individual, their metabolic situation, how long they’ve had diabetes, what kind of approach they’re following, maybe what medicines they’re taking,” Dr. Yancy said. “There’s a lot of different factors involved in remission.”
The study cohort generally had advanced diabetes and was taking a mix of medications, so the results might not be applicable to people with a more recent diabetes diagnosis, according to Ms. Pavlou.
Dr. Yancy said intermittent fasting could work well for the right person. The success of the approach could depend on a person’s eating habits and whether their meals usually fall outside the time-restricted window, or it could depend on how well a person follows rules, according to Dr. Yancy.
“Some people might not eat much after 8 o’clock, and some people might skip breakfast,” Dr. Yancy said. “And if that’s the case, then it’s not going to make a big impact on their weight probably.”
Medication is also an important consideration. Not eating can be dangerous for patients taking short-acting insulin or sulfonylureas, according to Dr. Yancy.
Ms. Pavlou said these findings show intermittent fasting is another option for patients with type 2 diabetes trying to lose weight. “If you’ve tried calorie counting, that’s not working for you or if you’re kind of burnt out, this is something else that you could try,” she said.
“We have a lot of patients that need to lose weight, and we have patients who respond differently to different approaches,” said Dr. Yancy. “So having various approaches is really valuable.”
The manuscript is currently under review at JAMA Internal Medicine, said Ms. Pavlou.
A version of this article appeared on Medscape.com.
AT NUTRITION 2023
CDC offers guidance on RSV vaccines for adults
Two newly approved respiratory syncytial virus (RSV) vaccines for adults aged 60 years and older may be able to prevent illness in those at risk for severe RSV disease.
Most adult RSV illness occurs among the older age group and results in an estimated 60,000-160,000 hospitalizations and 6,000-10,000 deaths per year among people aged at least 65 years.
Older adults deciding whether to get the vaccines should weigh risks and their own preferences and make the decision in consultation with their clinicians, said authors of a Centers for Disease Control and Prevention report.
Michael Melgar, MD, with the Coronavirus and Other Respiratory Viruses Division at the CDC, was lead author on the report, published in the Morbidity and Mortality Weekly Report.
Two new vaccines
In May, the Food and Drug Administration approved the first of two vaccines for preventing RSV lower respiratory tract disease for adults aged at least 60 years.
On June 21, the Advisory Committee on Immunization Practices (ACIP) recommended that people in that age group receive a single dose of RSV vaccine using shared decision-making.
The recommendation for shared decision-making makes the ACIP decision different from routine and risk-based vaccine recommendations. Rather than targeting all in a particular age group or risk group, the decision calls for consideration of a patients’ risk for disease and their characteristics, preferences, and values; the health care professional’s clinical discretion; and performance of the vaccine.
Dr. Melgar and colleagues reported that vaccination with one dose of the GSK or Pfizer RSV vaccines has proved moderately to highly effective in preventing symptomatic RSV-associated lower respiratory tract disease over two consecutive RSV seasons among people aged 60 and older.
The trials that led to approval weren’t powered to gauge efficacy against RSV-associated hospitalization and death. However, the authors wrote, the prevention of lower respiratory tract disease, including medically attended illness, suggests that the shots might prevent considerable morbidity from RSV disease among those aged 60 and older.
Both vaccines were generally well tolerated with a good safety profile. However, six cases of inflammatory neurologic events (including Guillain-Barré Syndrome, acute disseminated encephalomyelitis, and others) were reported in clinical trials after RSV vaccination.
“Whether these events occurred due to chance, or whether RSV vaccination increases the risk for inflammatory neurologic events, is currently unknown,” the authors wrote.
Postmarketing surveillance may help clarify the existence of any potential risk, but until those results are clearer, the CDC researchers said, RSV vaccinations should be targeted to older adults at highest risk for severe RSV and those most likely to benefit from the shots.
At higher risk
Some adults with certain medical conditions have a higher risk for RSV-associated hospitalization, according to the report.
Those conditions include chronic obstructive pulmonary disease, asthma, heart failure, coronary artery disease, cerebrovascular disease, diabetes mellitus, and chronic kidney disease.
People who are frail and of advanced age also are at higher risk for RSV hospitalization. That risk increases with age and the highest risk is for people aged at least 75 years.
The researchers added that RSV can cause severe disease in those with compromised immunity, including people who have received hematopoietic stem cell transplants and patients taking immunosuppressive drugs such as those used with solid organ transplants, cancer treatment, or other conditions.
As for when physicians should offer the vaccinations, shots are optimally given before the start of the RSV season.
However, the COVID-19 pandemic interrupted the seasonality and the timing has not yet returned to prepandemic patterns.
For the 2023-24 season, this report states, clinicians should offer RSV vaccination to adults aged at least 60 years using shared clinical decision-making as early as vaccine supply is available and should continue to offer vaccination to eligible adults who remain unvaccinated.
RSV vaccines can be administered with other adult vaccines during the same visit, the authors confirmed.
A version of this article first appeared on Medscape.com.
Two newly approved respiratory syncytial virus (RSV) vaccines for adults aged 60 years and older may be able to prevent illness in those at risk for severe RSV disease.
Most adult RSV illness occurs among the older age group and results in an estimated 60,000-160,000 hospitalizations and 6,000-10,000 deaths per year among people aged at least 65 years.
Older adults deciding whether to get the vaccines should weigh risks and their own preferences and make the decision in consultation with their clinicians, said authors of a Centers for Disease Control and Prevention report.
Michael Melgar, MD, with the Coronavirus and Other Respiratory Viruses Division at the CDC, was lead author on the report, published in the Morbidity and Mortality Weekly Report.
Two new vaccines
In May, the Food and Drug Administration approved the first of two vaccines for preventing RSV lower respiratory tract disease for adults aged at least 60 years.
On June 21, the Advisory Committee on Immunization Practices (ACIP) recommended that people in that age group receive a single dose of RSV vaccine using shared decision-making.
The recommendation for shared decision-making makes the ACIP decision different from routine and risk-based vaccine recommendations. Rather than targeting all in a particular age group or risk group, the decision calls for consideration of a patients’ risk for disease and their characteristics, preferences, and values; the health care professional’s clinical discretion; and performance of the vaccine.
Dr. Melgar and colleagues reported that vaccination with one dose of the GSK or Pfizer RSV vaccines has proved moderately to highly effective in preventing symptomatic RSV-associated lower respiratory tract disease over two consecutive RSV seasons among people aged 60 and older.
The trials that led to approval weren’t powered to gauge efficacy against RSV-associated hospitalization and death. However, the authors wrote, the prevention of lower respiratory tract disease, including medically attended illness, suggests that the shots might prevent considerable morbidity from RSV disease among those aged 60 and older.
Both vaccines were generally well tolerated with a good safety profile. However, six cases of inflammatory neurologic events (including Guillain-Barré Syndrome, acute disseminated encephalomyelitis, and others) were reported in clinical trials after RSV vaccination.
“Whether these events occurred due to chance, or whether RSV vaccination increases the risk for inflammatory neurologic events, is currently unknown,” the authors wrote.
Postmarketing surveillance may help clarify the existence of any potential risk, but until those results are clearer, the CDC researchers said, RSV vaccinations should be targeted to older adults at highest risk for severe RSV and those most likely to benefit from the shots.
At higher risk
Some adults with certain medical conditions have a higher risk for RSV-associated hospitalization, according to the report.
Those conditions include chronic obstructive pulmonary disease, asthma, heart failure, coronary artery disease, cerebrovascular disease, diabetes mellitus, and chronic kidney disease.
People who are frail and of advanced age also are at higher risk for RSV hospitalization. That risk increases with age and the highest risk is for people aged at least 75 years.
The researchers added that RSV can cause severe disease in those with compromised immunity, including people who have received hematopoietic stem cell transplants and patients taking immunosuppressive drugs such as those used with solid organ transplants, cancer treatment, or other conditions.
As for when physicians should offer the vaccinations, shots are optimally given before the start of the RSV season.
However, the COVID-19 pandemic interrupted the seasonality and the timing has not yet returned to prepandemic patterns.
For the 2023-24 season, this report states, clinicians should offer RSV vaccination to adults aged at least 60 years using shared clinical decision-making as early as vaccine supply is available and should continue to offer vaccination to eligible adults who remain unvaccinated.
RSV vaccines can be administered with other adult vaccines during the same visit, the authors confirmed.
A version of this article first appeared on Medscape.com.
Two newly approved respiratory syncytial virus (RSV) vaccines for adults aged 60 years and older may be able to prevent illness in those at risk for severe RSV disease.
Most adult RSV illness occurs among the older age group and results in an estimated 60,000-160,000 hospitalizations and 6,000-10,000 deaths per year among people aged at least 65 years.
Older adults deciding whether to get the vaccines should weigh risks and their own preferences and make the decision in consultation with their clinicians, said authors of a Centers for Disease Control and Prevention report.
Michael Melgar, MD, with the Coronavirus and Other Respiratory Viruses Division at the CDC, was lead author on the report, published in the Morbidity and Mortality Weekly Report.
Two new vaccines
In May, the Food and Drug Administration approved the first of two vaccines for preventing RSV lower respiratory tract disease for adults aged at least 60 years.
On June 21, the Advisory Committee on Immunization Practices (ACIP) recommended that people in that age group receive a single dose of RSV vaccine using shared decision-making.
The recommendation for shared decision-making makes the ACIP decision different from routine and risk-based vaccine recommendations. Rather than targeting all in a particular age group or risk group, the decision calls for consideration of a patients’ risk for disease and their characteristics, preferences, and values; the health care professional’s clinical discretion; and performance of the vaccine.
Dr. Melgar and colleagues reported that vaccination with one dose of the GSK or Pfizer RSV vaccines has proved moderately to highly effective in preventing symptomatic RSV-associated lower respiratory tract disease over two consecutive RSV seasons among people aged 60 and older.
The trials that led to approval weren’t powered to gauge efficacy against RSV-associated hospitalization and death. However, the authors wrote, the prevention of lower respiratory tract disease, including medically attended illness, suggests that the shots might prevent considerable morbidity from RSV disease among those aged 60 and older.
Both vaccines were generally well tolerated with a good safety profile. However, six cases of inflammatory neurologic events (including Guillain-Barré Syndrome, acute disseminated encephalomyelitis, and others) were reported in clinical trials after RSV vaccination.
“Whether these events occurred due to chance, or whether RSV vaccination increases the risk for inflammatory neurologic events, is currently unknown,” the authors wrote.
Postmarketing surveillance may help clarify the existence of any potential risk, but until those results are clearer, the CDC researchers said, RSV vaccinations should be targeted to older adults at highest risk for severe RSV and those most likely to benefit from the shots.
At higher risk
Some adults with certain medical conditions have a higher risk for RSV-associated hospitalization, according to the report.
Those conditions include chronic obstructive pulmonary disease, asthma, heart failure, coronary artery disease, cerebrovascular disease, diabetes mellitus, and chronic kidney disease.
People who are frail and of advanced age also are at higher risk for RSV hospitalization. That risk increases with age and the highest risk is for people aged at least 75 years.
The researchers added that RSV can cause severe disease in those with compromised immunity, including people who have received hematopoietic stem cell transplants and patients taking immunosuppressive drugs such as those used with solid organ transplants, cancer treatment, or other conditions.
As for when physicians should offer the vaccinations, shots are optimally given before the start of the RSV season.
However, the COVID-19 pandemic interrupted the seasonality and the timing has not yet returned to prepandemic patterns.
For the 2023-24 season, this report states, clinicians should offer RSV vaccination to adults aged at least 60 years using shared clinical decision-making as early as vaccine supply is available and should continue to offer vaccination to eligible adults who remain unvaccinated.
RSV vaccines can be administered with other adult vaccines during the same visit, the authors confirmed.
A version of this article first appeared on Medscape.com.
FROM THE MMWR
Cancer Patients: Who’s at Risk for Venous Thromboembolism?
Patients with cancer are at a high risk of venous thromboembolism (VTE)—in fact, it’s one of the leading causes of death in patients who receive systemic therapy for cancer. But as cancer treatment has evolved, have the incidence and risk of VTE changed too?
Researchers from Veterans Affairs Boston Healthcare System in Massachusetts conducted a study with 434,203 veterans to evaluate the pattern of VTE incidence over 16 years, focusing on the types of cancer, treatment, race and ethnicity, and other factors related to cancer-associated thrombosis (CAT).
In contrast with other large population studies, this study found the overall incidence of CAT remained largely stable over time. At 12 months, the incidence was 4.5%, with yearly trends ranging between 4.2% and 4.7%. “As expected,” the researchers say, the subset of patients receiving systemic therapy had a higher incidence of VTE at 12 months (7.7%) than did the overall cohort. The pattern was “particularly pronounced” in gynecologic, testicular, and kidney cancers, where the incidence of VTE was 2 to 3 times higher in the treated cohort compared with the overall cohort.
Cancer type and diagnosis were the most statistically and clinically significant associations with CAT, with up to a 6-fold difference between cancer subtypes. The patients at the highest risk of VTE were those with pancreatic cancer and acute lymphoblastic leukemia.
Most studies have focused only on patients with solid tumors, but these researchers observed novel patterns among patients with hematologic neoplasms. Specifically, a higher incidence of VTE among patients with aggressive vs indolent leukemias and lymphomas. This trend, the researchers say, may be associated in part with catheter-related events.
Furthermore, the type of system treatment was associated with the risk of VTE, the researchers say, although to a lesser extent. Chemotherapy- and immunotherapy-based regimens had the highest risk of VTE, relative to no treatment. Targeted and endocrine therapy also carried a higher risk compared with no treatment but to a lesser degree.
The researchers found significant heterogeneity by race and ethnicity across cancer types. Non-Hispanic Black patients had about 20% higher risk of VTE compared with non-Hispanic White patients. Asian and Pacific Islander patients had about 20% lower risk compared with non-Hispanic White patients.
Male sex was also associated with VTE. However, “interestingly,” the researchers note, neighborhood-level socioeconomic factors and patients’ comorbidities were not associated with CAT but were associated with mortality.
Their results suggest that patient- and treatment-specific factors play a critical role in assessing the risk of CAT, and “ongoing efforts to identify these patterns are of utmost importance for risk stratification and prognostic assessment.”
Patients with cancer are at a high risk of venous thromboembolism (VTE)—in fact, it’s one of the leading causes of death in patients who receive systemic therapy for cancer. But as cancer treatment has evolved, have the incidence and risk of VTE changed too?
Researchers from Veterans Affairs Boston Healthcare System in Massachusetts conducted a study with 434,203 veterans to evaluate the pattern of VTE incidence over 16 years, focusing on the types of cancer, treatment, race and ethnicity, and other factors related to cancer-associated thrombosis (CAT).
In contrast with other large population studies, this study found the overall incidence of CAT remained largely stable over time. At 12 months, the incidence was 4.5%, with yearly trends ranging between 4.2% and 4.7%. “As expected,” the researchers say, the subset of patients receiving systemic therapy had a higher incidence of VTE at 12 months (7.7%) than did the overall cohort. The pattern was “particularly pronounced” in gynecologic, testicular, and kidney cancers, where the incidence of VTE was 2 to 3 times higher in the treated cohort compared with the overall cohort.
Cancer type and diagnosis were the most statistically and clinically significant associations with CAT, with up to a 6-fold difference between cancer subtypes. The patients at the highest risk of VTE were those with pancreatic cancer and acute lymphoblastic leukemia.
Most studies have focused only on patients with solid tumors, but these researchers observed novel patterns among patients with hematologic neoplasms. Specifically, a higher incidence of VTE among patients with aggressive vs indolent leukemias and lymphomas. This trend, the researchers say, may be associated in part with catheter-related events.
Furthermore, the type of system treatment was associated with the risk of VTE, the researchers say, although to a lesser extent. Chemotherapy- and immunotherapy-based regimens had the highest risk of VTE, relative to no treatment. Targeted and endocrine therapy also carried a higher risk compared with no treatment but to a lesser degree.
The researchers found significant heterogeneity by race and ethnicity across cancer types. Non-Hispanic Black patients had about 20% higher risk of VTE compared with non-Hispanic White patients. Asian and Pacific Islander patients had about 20% lower risk compared with non-Hispanic White patients.
Male sex was also associated with VTE. However, “interestingly,” the researchers note, neighborhood-level socioeconomic factors and patients’ comorbidities were not associated with CAT but were associated with mortality.
Their results suggest that patient- and treatment-specific factors play a critical role in assessing the risk of CAT, and “ongoing efforts to identify these patterns are of utmost importance for risk stratification and prognostic assessment.”
Patients with cancer are at a high risk of venous thromboembolism (VTE)—in fact, it’s one of the leading causes of death in patients who receive systemic therapy for cancer. But as cancer treatment has evolved, have the incidence and risk of VTE changed too?
Researchers from Veterans Affairs Boston Healthcare System in Massachusetts conducted a study with 434,203 veterans to evaluate the pattern of VTE incidence over 16 years, focusing on the types of cancer, treatment, race and ethnicity, and other factors related to cancer-associated thrombosis (CAT).
In contrast with other large population studies, this study found the overall incidence of CAT remained largely stable over time. At 12 months, the incidence was 4.5%, with yearly trends ranging between 4.2% and 4.7%. “As expected,” the researchers say, the subset of patients receiving systemic therapy had a higher incidence of VTE at 12 months (7.7%) than did the overall cohort. The pattern was “particularly pronounced” in gynecologic, testicular, and kidney cancers, where the incidence of VTE was 2 to 3 times higher in the treated cohort compared with the overall cohort.
Cancer type and diagnosis were the most statistically and clinically significant associations with CAT, with up to a 6-fold difference between cancer subtypes. The patients at the highest risk of VTE were those with pancreatic cancer and acute lymphoblastic leukemia.
Most studies have focused only on patients with solid tumors, but these researchers observed novel patterns among patients with hematologic neoplasms. Specifically, a higher incidence of VTE among patients with aggressive vs indolent leukemias and lymphomas. This trend, the researchers say, may be associated in part with catheter-related events.
Furthermore, the type of system treatment was associated with the risk of VTE, the researchers say, although to a lesser extent. Chemotherapy- and immunotherapy-based regimens had the highest risk of VTE, relative to no treatment. Targeted and endocrine therapy also carried a higher risk compared with no treatment but to a lesser degree.
The researchers found significant heterogeneity by race and ethnicity across cancer types. Non-Hispanic Black patients had about 20% higher risk of VTE compared with non-Hispanic White patients. Asian and Pacific Islander patients had about 20% lower risk compared with non-Hispanic White patients.
Male sex was also associated with VTE. However, “interestingly,” the researchers note, neighborhood-level socioeconomic factors and patients’ comorbidities were not associated with CAT but were associated with mortality.
Their results suggest that patient- and treatment-specific factors play a critical role in assessing the risk of CAT, and “ongoing efforts to identify these patterns are of utmost importance for risk stratification and prognostic assessment.”
Regional Meeting Focuses on Women’s Cancer Survivorship
As the number of female veterans continues to grow, the US Department of Veterans Affairs (VA) is adjusting by focusing more on breast/gynecological cancer and referring fewer cases to outside clinicians.
The VA’s effort reflects the reality that female veterans from the wars in Afghanistan and Iraq are approaching the ages—50s, 60s, and 70s—when cancer diagnoses become more common, said Sarah Colonna, MD, national medical director of breast oncology for VA's Breast and Gynecologic Oncology System of Excellence and an oncologist at the Huntsman Cancer Institute and Wahlen VA Medical Center in Salt Lake City, Utah. “This is preparation for the change that we know is coming.”
In response, the Association of VA Hematology/Oncology (AVAHO) is devoting a regional meeting in Tampa, Florida (July 29, 2023) to improving survivorship for patients with women’s cancers. “This meeting is designed to educate both cancer experts and primary care providers on the care of women who have already gone through breast and gynecological cancer treatment,” Colonna explained.
Adherence Challenges
Colonna will speak in a session about the importance of adherence to endocrine therapy. “When we prescribe endocrine therapy for breast cancer, we usually ask women to stay on it for 5 to 10 years, and sometimes that’s hard for them,” she said. “I’ll talk about tips and tricks to help women stay on endocrine therapy for the long haul because we know that is linked to better survival.”
Between two-thirds and three-quarters of women with breast cancer are advised to stay on endocrine drugs, she said, but the medications can be difficult to tolerate due to adverse effects such as hot flashes and sleep disturbances.
In addition, patients are often anxious about the medications. “Women are very leery of anything that changes or makes their hormones different,” Colonna noted. “They feel like it’s messing with something that is natural for them.”
Colonna urges colleagues to focus on their “soft skills,” the ability to absorb and validate the worries of patients. Instead of dismissing them, she said, focus on messages that acknowledge concerns but are also firm: “That’s real, that sucks. But we’ve got to do it.”
It’s also helpful to guide patients away from thinking that taking a pill every day means they’re sick. “I try to flip that paradigm: ‘You’re taking this pill every day because you have power over this thing that happened to you.’”
Education is also key, she said, so that patients “understand very clearly why this medication is important for them: It increases the chance of surviving breast cancer or it increases the chances that the cancer will never come back in your arm or in your breast. Then, whether they make a decision to take it or not, at least they’re making the choice with knowledge.”
As for adverse effects, Colonna said medications such as antidepressants and painkillers can relieve hot flashes, which can disturb sleep.
Identifying the best strategy to address adverse effects “requires keeping in frequent contact with the patient during the first 6 months of endocrine therapy, which are really critical,” she said. “Once they’ve been on it for a year, they can see the light at the end of the tunnel and hang in there even if they have adverse effects.”
Some guidelines suggest that no doctor visits are needed until the 6-month mark, but Colonna prefers to check in at the 4- to 6-week mark, even if it’s just via a phone call. Otherwise, “often they’ll stop taking the pill, and then you won’t know about it until you see them at 6 six months.” At that point, she said, a critical period for treatment has passed.
The Role of Nurse Navigators
In another session at the Tampa regional meeting, AVAHO president-elect Cindy Bowman, MSN, RN, OCN, will moderate a session about the role of nurse navigators in VA cancer care. She is the coordinator of the Cancer Care Navigation Program at the C. W. Bill Young VA Medical Center in Bay Pines, Florida.
“Veterans become survivors the day they’re diagnosed with cancer,” she said. Within the VA, cancer-care navigator teams developed over the past decade aim to help patients find their way forward through survivorship, she said, and nurses are crucial to the effort.
As Sharp and Scheid reported in a 2018 Journal Oncology Navigation Survivorship article, “research demonstrates that navigation can improve access to the cancer care system by addressing barriers, as well as facilitating quality care. The benefits of patient navigation for improving cancer patient outcomes is considerable.” McKenney and colleagues found that “patient navigation has been demonstrated to increase access to screening, shorten time to diagnostic resolution, and improve cancer outcomes, particularly in health disparity populations, such as women of color, rural populations, and poor women.”
According to Bowman, “it has become standard practice to have nurse navigators be there each step of the way from a high suspicion of cancer to diagnosis and through the clinical workup into active treatment and survivorship.” Within the VA, she said, “the focus right now is to look at standardizing care that all VAs will be able to offer holistic, comprehensive cancer-care navigation teams.”
At the regional meeting, Bowman’s session will include updates from nurse navigators about helping patients through breast/gynecological cancer, abnormal mammograms, and survivorship.
Nurse navigators are typically the second medical professionals who talk to cancer patients after their physicians, Bowman said. The unique knowledge of oncology nurse navigators gives them invaluable insight into treatment plans and cancer drug regimens, she said.
“They’re able to sit down and discuss the actual cancer drug regimen with patients—what each of those drugs do, how they’re administered, the short-term and long-term side effects,” she said. “They have the knowledge about all aspects of cancer care that can really only come from somebody who’s specialty trained.”
Other sessions at the AVAHO regional meeting will highlight breast cancer and lymphedema, breast cancer and bone health; diet, exercise and cancer; sexual health for breast/gynecological cancer survivors; and imaging surveillance after diagnosis.
As the number of female veterans continues to grow, the US Department of Veterans Affairs (VA) is adjusting by focusing more on breast/gynecological cancer and referring fewer cases to outside clinicians.
The VA’s effort reflects the reality that female veterans from the wars in Afghanistan and Iraq are approaching the ages—50s, 60s, and 70s—when cancer diagnoses become more common, said Sarah Colonna, MD, national medical director of breast oncology for VA's Breast and Gynecologic Oncology System of Excellence and an oncologist at the Huntsman Cancer Institute and Wahlen VA Medical Center in Salt Lake City, Utah. “This is preparation for the change that we know is coming.”
In response, the Association of VA Hematology/Oncology (AVAHO) is devoting a regional meeting in Tampa, Florida (July 29, 2023) to improving survivorship for patients with women’s cancers. “This meeting is designed to educate both cancer experts and primary care providers on the care of women who have already gone through breast and gynecological cancer treatment,” Colonna explained.
Adherence Challenges
Colonna will speak in a session about the importance of adherence to endocrine therapy. “When we prescribe endocrine therapy for breast cancer, we usually ask women to stay on it for 5 to 10 years, and sometimes that’s hard for them,” she said. “I’ll talk about tips and tricks to help women stay on endocrine therapy for the long haul because we know that is linked to better survival.”
Between two-thirds and three-quarters of women with breast cancer are advised to stay on endocrine drugs, she said, but the medications can be difficult to tolerate due to adverse effects such as hot flashes and sleep disturbances.
In addition, patients are often anxious about the medications. “Women are very leery of anything that changes or makes their hormones different,” Colonna noted. “They feel like it’s messing with something that is natural for them.”
Colonna urges colleagues to focus on their “soft skills,” the ability to absorb and validate the worries of patients. Instead of dismissing them, she said, focus on messages that acknowledge concerns but are also firm: “That’s real, that sucks. But we’ve got to do it.”
It’s also helpful to guide patients away from thinking that taking a pill every day means they’re sick. “I try to flip that paradigm: ‘You’re taking this pill every day because you have power over this thing that happened to you.’”
Education is also key, she said, so that patients “understand very clearly why this medication is important for them: It increases the chance of surviving breast cancer or it increases the chances that the cancer will never come back in your arm or in your breast. Then, whether they make a decision to take it or not, at least they’re making the choice with knowledge.”
As for adverse effects, Colonna said medications such as antidepressants and painkillers can relieve hot flashes, which can disturb sleep.
Identifying the best strategy to address adverse effects “requires keeping in frequent contact with the patient during the first 6 months of endocrine therapy, which are really critical,” she said. “Once they’ve been on it for a year, they can see the light at the end of the tunnel and hang in there even if they have adverse effects.”
Some guidelines suggest that no doctor visits are needed until the 6-month mark, but Colonna prefers to check in at the 4- to 6-week mark, even if it’s just via a phone call. Otherwise, “often they’ll stop taking the pill, and then you won’t know about it until you see them at 6 six months.” At that point, she said, a critical period for treatment has passed.
The Role of Nurse Navigators
In another session at the Tampa regional meeting, AVAHO president-elect Cindy Bowman, MSN, RN, OCN, will moderate a session about the role of nurse navigators in VA cancer care. She is the coordinator of the Cancer Care Navigation Program at the C. W. Bill Young VA Medical Center in Bay Pines, Florida.
“Veterans become survivors the day they’re diagnosed with cancer,” she said. Within the VA, cancer-care navigator teams developed over the past decade aim to help patients find their way forward through survivorship, she said, and nurses are crucial to the effort.
As Sharp and Scheid reported in a 2018 Journal Oncology Navigation Survivorship article, “research demonstrates that navigation can improve access to the cancer care system by addressing barriers, as well as facilitating quality care. The benefits of patient navigation for improving cancer patient outcomes is considerable.” McKenney and colleagues found that “patient navigation has been demonstrated to increase access to screening, shorten time to diagnostic resolution, and improve cancer outcomes, particularly in health disparity populations, such as women of color, rural populations, and poor women.”
According to Bowman, “it has become standard practice to have nurse navigators be there each step of the way from a high suspicion of cancer to diagnosis and through the clinical workup into active treatment and survivorship.” Within the VA, she said, “the focus right now is to look at standardizing care that all VAs will be able to offer holistic, comprehensive cancer-care navigation teams.”
At the regional meeting, Bowman’s session will include updates from nurse navigators about helping patients through breast/gynecological cancer, abnormal mammograms, and survivorship.
Nurse navigators are typically the second medical professionals who talk to cancer patients after their physicians, Bowman said. The unique knowledge of oncology nurse navigators gives them invaluable insight into treatment plans and cancer drug regimens, she said.
“They’re able to sit down and discuss the actual cancer drug regimen with patients—what each of those drugs do, how they’re administered, the short-term and long-term side effects,” she said. “They have the knowledge about all aspects of cancer care that can really only come from somebody who’s specialty trained.”
Other sessions at the AVAHO regional meeting will highlight breast cancer and lymphedema, breast cancer and bone health; diet, exercise and cancer; sexual health for breast/gynecological cancer survivors; and imaging surveillance after diagnosis.
As the number of female veterans continues to grow, the US Department of Veterans Affairs (VA) is adjusting by focusing more on breast/gynecological cancer and referring fewer cases to outside clinicians.
The VA’s effort reflects the reality that female veterans from the wars in Afghanistan and Iraq are approaching the ages—50s, 60s, and 70s—when cancer diagnoses become more common, said Sarah Colonna, MD, national medical director of breast oncology for VA's Breast and Gynecologic Oncology System of Excellence and an oncologist at the Huntsman Cancer Institute and Wahlen VA Medical Center in Salt Lake City, Utah. “This is preparation for the change that we know is coming.”
In response, the Association of VA Hematology/Oncology (AVAHO) is devoting a regional meeting in Tampa, Florida (July 29, 2023) to improving survivorship for patients with women’s cancers. “This meeting is designed to educate both cancer experts and primary care providers on the care of women who have already gone through breast and gynecological cancer treatment,” Colonna explained.
Adherence Challenges
Colonna will speak in a session about the importance of adherence to endocrine therapy. “When we prescribe endocrine therapy for breast cancer, we usually ask women to stay on it for 5 to 10 years, and sometimes that’s hard for them,” she said. “I’ll talk about tips and tricks to help women stay on endocrine therapy for the long haul because we know that is linked to better survival.”
Between two-thirds and three-quarters of women with breast cancer are advised to stay on endocrine drugs, she said, but the medications can be difficult to tolerate due to adverse effects such as hot flashes and sleep disturbances.
In addition, patients are often anxious about the medications. “Women are very leery of anything that changes or makes their hormones different,” Colonna noted. “They feel like it’s messing with something that is natural for them.”
Colonna urges colleagues to focus on their “soft skills,” the ability to absorb and validate the worries of patients. Instead of dismissing them, she said, focus on messages that acknowledge concerns but are also firm: “That’s real, that sucks. But we’ve got to do it.”
It’s also helpful to guide patients away from thinking that taking a pill every day means they’re sick. “I try to flip that paradigm: ‘You’re taking this pill every day because you have power over this thing that happened to you.’”
Education is also key, she said, so that patients “understand very clearly why this medication is important for them: It increases the chance of surviving breast cancer or it increases the chances that the cancer will never come back in your arm or in your breast. Then, whether they make a decision to take it or not, at least they’re making the choice with knowledge.”
As for adverse effects, Colonna said medications such as antidepressants and painkillers can relieve hot flashes, which can disturb sleep.
Identifying the best strategy to address adverse effects “requires keeping in frequent contact with the patient during the first 6 months of endocrine therapy, which are really critical,” she said. “Once they’ve been on it for a year, they can see the light at the end of the tunnel and hang in there even if they have adverse effects.”
Some guidelines suggest that no doctor visits are needed until the 6-month mark, but Colonna prefers to check in at the 4- to 6-week mark, even if it’s just via a phone call. Otherwise, “often they’ll stop taking the pill, and then you won’t know about it until you see them at 6 six months.” At that point, she said, a critical period for treatment has passed.
The Role of Nurse Navigators
In another session at the Tampa regional meeting, AVAHO president-elect Cindy Bowman, MSN, RN, OCN, will moderate a session about the role of nurse navigators in VA cancer care. She is the coordinator of the Cancer Care Navigation Program at the C. W. Bill Young VA Medical Center in Bay Pines, Florida.
“Veterans become survivors the day they’re diagnosed with cancer,” she said. Within the VA, cancer-care navigator teams developed over the past decade aim to help patients find their way forward through survivorship, she said, and nurses are crucial to the effort.
As Sharp and Scheid reported in a 2018 Journal Oncology Navigation Survivorship article, “research demonstrates that navigation can improve access to the cancer care system by addressing barriers, as well as facilitating quality care. The benefits of patient navigation for improving cancer patient outcomes is considerable.” McKenney and colleagues found that “patient navigation has been demonstrated to increase access to screening, shorten time to diagnostic resolution, and improve cancer outcomes, particularly in health disparity populations, such as women of color, rural populations, and poor women.”
According to Bowman, “it has become standard practice to have nurse navigators be there each step of the way from a high suspicion of cancer to diagnosis and through the clinical workup into active treatment and survivorship.” Within the VA, she said, “the focus right now is to look at standardizing care that all VAs will be able to offer holistic, comprehensive cancer-care navigation teams.”
At the regional meeting, Bowman’s session will include updates from nurse navigators about helping patients through breast/gynecological cancer, abnormal mammograms, and survivorship.
Nurse navigators are typically the second medical professionals who talk to cancer patients after their physicians, Bowman said. The unique knowledge of oncology nurse navigators gives them invaluable insight into treatment plans and cancer drug regimens, she said.
“They’re able to sit down and discuss the actual cancer drug regimen with patients—what each of those drugs do, how they’re administered, the short-term and long-term side effects,” she said. “They have the knowledge about all aspects of cancer care that can really only come from somebody who’s specialty trained.”
Other sessions at the AVAHO regional meeting will highlight breast cancer and lymphedema, breast cancer and bone health; diet, exercise and cancer; sexual health for breast/gynecological cancer survivors; and imaging surveillance after diagnosis.
New cancer survival calculator focuses on oral cancer
This represents the first cancer survival calculator that provides “personalized estimates of the likelihood of surviving or dying from oral cancer or other causes,” according to the experts who developed the tool.
An analysis evaluating the new calculator revealed that people with oral cancer are more likely to die from other causes, compared with their peers without oral cancer, and that noncancer survival worsens with cancer stage.
With its unique design, the calculator “represents perhaps one of the most sophisticated and comprehensive tools to date by integrating multiple population-level data sources to account for general health status [and] disease exposures,” such as alcohol and tobacco, socioeconomic status, and coexisting conditions, the authors of an accompanying commentary wrote.
This calculator may just be the beginning. The broader aim of developing the tool, the study authors explained, is for this new calculator approach to be “applicable for developing future prognostic models of cancer and noncancer aspects of a person’s health in other cancers.”
The analysis was published in JAMA Otolaryngology–Head and Neck Surgery.
When assessing survival, factors such as cancer stage and tumor size are key, but comorbidities also play a crucial role. For oral cancer in particular, where alcohol and tobacco use are notorious risk factors, comorbidities occur frequently and are often serious.
To create a model that provides more “holistic and personalized” estimates and includes a host of factors that can affect the risk of death, the authors tapped into data from the Surveillance, Epidemiology, and End Results database to develop the SEER Oral Cancer Survival Calculator.
Alongside data from the SEER database, the calculator used data from the National Health Interview Survey’s Longitudinal Mortality Files to obtain estimates of general health status, life expectancy without cancer, and the probability of dying from the cancer or from other causes within 1-10 years among people with newly diagnosed oral cancer.
Overall, the data included 22,392 patients, aged 20-94, with oral squamous cell carcinoma, 60.5% of whom were male and 78% White, as well as 402,626 interviewees from the survey. The calculator did not include patients with tonsil- or tongue-based cancers, which were not considered anatomically part of the oral cavity.
The most common conditions coexisting with oral cancer were diabetes and chronic obstructive pulmonary disease among older patients. Among those with oral cancer, more than half (52.8%) had none of the major coexisting conditions, which also included peripheral and cerebrovascular disease, compared with 80% of the Medicare population.
The researchers described and validated four models – one that estimated the probability of death due to oral cancer, and then three others that estimated the probability of death from other causes, with variations based on the specific data and covariates included.
Overall, the models in the calculator estimated that patients with oral cancer have a higher risk of death from other causes, compared with the general population, and survival estimates for noncancer causes got worse with more advanced cancer stage.
For instance, for a patient diagnosed with stage 3 oral cancer after age 50, the chances of being alive at age 70 were 60% for females and 44% for males in the absence of cancer, whereas the corresponding survival estimates in the general U.S. population were 86% for females and 79% for males – an absolute difference of 26 and 35 percentage points.
One key reason for this trend is that patients with later-stage cancers likely also have more coexisting health conditions, first author Louise Davies, MD, from the Geisel School of Medicine at Dartmouth, Lebanon, N.H., explained.
Another reason: For cancers with low enough mortality rates, people might be more likely to die from causes other than their cancer. This can also occur in ductal carcinoma in situ breast cancer or papillary thyroid cancer, noted Dr. Davies, also from the Department of Veterans Affairs Medical Center, White River Junction, Vt.
Commenting on the study, Eric Moore, MD, a head and neck surgeon with the Mayo Clinic in Rochester, Minn., said that while such prediction tools are important, they also come with caveats.
“I think these calculators are helpful and certainly having them widely available to people gives them another piece of knowledge that can be powerful,” he told this news organization. “But you want to make sure you don’t interpret them as the end-all, be-all message, because there are an infinite number of variables that could influence survival that aren’t available in some of these datasets.”
Neil D. Gross, MD, a professor of head and neck surgery at the University of Texas MD Anderson Cancer Center, Houston, agreed. Although this new calculator uses a large dataset, such tools “can be imperfect” and some factors simply can’t be calculated, such as a person’s priorities, Dr. Gross said.
That’s why there’s no substitute for having a “very personal discussion between a patient and a physician to decide what’s best.” And this calculator is just one tool to help with that process, Dr. Gross said.
The commentary authors echoed these sentiments. “This calculator can potentially bridge the gaps between the survival estimates in the literature, life tables, clinical gestalt, and physician attempts to contextualize the inherent limitations of applying survival curves and averages to the one patient with the diagnosis,” wrote Leila J. Mady, MD, PhD, MPH, Wayne M. Koch, MD, and Carole Fakhry, MD, MPH, all from Johns Hopkins School of Medicine, Baltimore.
But a caveat in providing such predictions is the possible psychological effect the news can have.
“Potential risks of revealing personalized prognostic survival estimates to patients include increased anxiety and distress surrounding competing causes of death [and] misinterpretation of data,” the commentary authors cautioned, adding that “we must present such information with grace and sensitivity.”
Dr. Davies recommends that clinicians ask patients what they want to know because that will vary by patient and potentially over time for the same patient.
“People are more than their cancer diagnosis,” said Dr. Davies. “Giving them the opportunity to consider their life as a whole is the aim.”
The oral cancer calculator can be publicly accessed through the National Cancer Institute. The study was supported by the Department of Veterans Affairs and the National Cancer Institute as part of an interagency agreement. The authors report no relevant financial relationships.
A version of this article appeared on Medscape.com.
This represents the first cancer survival calculator that provides “personalized estimates of the likelihood of surviving or dying from oral cancer or other causes,” according to the experts who developed the tool.
An analysis evaluating the new calculator revealed that people with oral cancer are more likely to die from other causes, compared with their peers without oral cancer, and that noncancer survival worsens with cancer stage.
With its unique design, the calculator “represents perhaps one of the most sophisticated and comprehensive tools to date by integrating multiple population-level data sources to account for general health status [and] disease exposures,” such as alcohol and tobacco, socioeconomic status, and coexisting conditions, the authors of an accompanying commentary wrote.
This calculator may just be the beginning. The broader aim of developing the tool, the study authors explained, is for this new calculator approach to be “applicable for developing future prognostic models of cancer and noncancer aspects of a person’s health in other cancers.”
The analysis was published in JAMA Otolaryngology–Head and Neck Surgery.
When assessing survival, factors such as cancer stage and tumor size are key, but comorbidities also play a crucial role. For oral cancer in particular, where alcohol and tobacco use are notorious risk factors, comorbidities occur frequently and are often serious.
To create a model that provides more “holistic and personalized” estimates and includes a host of factors that can affect the risk of death, the authors tapped into data from the Surveillance, Epidemiology, and End Results database to develop the SEER Oral Cancer Survival Calculator.
Alongside data from the SEER database, the calculator used data from the National Health Interview Survey’s Longitudinal Mortality Files to obtain estimates of general health status, life expectancy without cancer, and the probability of dying from the cancer or from other causes within 1-10 years among people with newly diagnosed oral cancer.
Overall, the data included 22,392 patients, aged 20-94, with oral squamous cell carcinoma, 60.5% of whom were male and 78% White, as well as 402,626 interviewees from the survey. The calculator did not include patients with tonsil- or tongue-based cancers, which were not considered anatomically part of the oral cavity.
The most common conditions coexisting with oral cancer were diabetes and chronic obstructive pulmonary disease among older patients. Among those with oral cancer, more than half (52.8%) had none of the major coexisting conditions, which also included peripheral and cerebrovascular disease, compared with 80% of the Medicare population.
The researchers described and validated four models – one that estimated the probability of death due to oral cancer, and then three others that estimated the probability of death from other causes, with variations based on the specific data and covariates included.
Overall, the models in the calculator estimated that patients with oral cancer have a higher risk of death from other causes, compared with the general population, and survival estimates for noncancer causes got worse with more advanced cancer stage.
For instance, for a patient diagnosed with stage 3 oral cancer after age 50, the chances of being alive at age 70 were 60% for females and 44% for males in the absence of cancer, whereas the corresponding survival estimates in the general U.S. population were 86% for females and 79% for males – an absolute difference of 26 and 35 percentage points.
One key reason for this trend is that patients with later-stage cancers likely also have more coexisting health conditions, first author Louise Davies, MD, from the Geisel School of Medicine at Dartmouth, Lebanon, N.H., explained.
Another reason: For cancers with low enough mortality rates, people might be more likely to die from causes other than their cancer. This can also occur in ductal carcinoma in situ breast cancer or papillary thyroid cancer, noted Dr. Davies, also from the Department of Veterans Affairs Medical Center, White River Junction, Vt.
Commenting on the study, Eric Moore, MD, a head and neck surgeon with the Mayo Clinic in Rochester, Minn., said that while such prediction tools are important, they also come with caveats.
“I think these calculators are helpful and certainly having them widely available to people gives them another piece of knowledge that can be powerful,” he told this news organization. “But you want to make sure you don’t interpret them as the end-all, be-all message, because there are an infinite number of variables that could influence survival that aren’t available in some of these datasets.”
Neil D. Gross, MD, a professor of head and neck surgery at the University of Texas MD Anderson Cancer Center, Houston, agreed. Although this new calculator uses a large dataset, such tools “can be imperfect” and some factors simply can’t be calculated, such as a person’s priorities, Dr. Gross said.
That’s why there’s no substitute for having a “very personal discussion between a patient and a physician to decide what’s best.” And this calculator is just one tool to help with that process, Dr. Gross said.
The commentary authors echoed these sentiments. “This calculator can potentially bridge the gaps between the survival estimates in the literature, life tables, clinical gestalt, and physician attempts to contextualize the inherent limitations of applying survival curves and averages to the one patient with the diagnosis,” wrote Leila J. Mady, MD, PhD, MPH, Wayne M. Koch, MD, and Carole Fakhry, MD, MPH, all from Johns Hopkins School of Medicine, Baltimore.
But a caveat in providing such predictions is the possible psychological effect the news can have.
“Potential risks of revealing personalized prognostic survival estimates to patients include increased anxiety and distress surrounding competing causes of death [and] misinterpretation of data,” the commentary authors cautioned, adding that “we must present such information with grace and sensitivity.”
Dr. Davies recommends that clinicians ask patients what they want to know because that will vary by patient and potentially over time for the same patient.
“People are more than their cancer diagnosis,” said Dr. Davies. “Giving them the opportunity to consider their life as a whole is the aim.”
The oral cancer calculator can be publicly accessed through the National Cancer Institute. The study was supported by the Department of Veterans Affairs and the National Cancer Institute as part of an interagency agreement. The authors report no relevant financial relationships.
A version of this article appeared on Medscape.com.
This represents the first cancer survival calculator that provides “personalized estimates of the likelihood of surviving or dying from oral cancer or other causes,” according to the experts who developed the tool.
An analysis evaluating the new calculator revealed that people with oral cancer are more likely to die from other causes, compared with their peers without oral cancer, and that noncancer survival worsens with cancer stage.
With its unique design, the calculator “represents perhaps one of the most sophisticated and comprehensive tools to date by integrating multiple population-level data sources to account for general health status [and] disease exposures,” such as alcohol and tobacco, socioeconomic status, and coexisting conditions, the authors of an accompanying commentary wrote.
This calculator may just be the beginning. The broader aim of developing the tool, the study authors explained, is for this new calculator approach to be “applicable for developing future prognostic models of cancer and noncancer aspects of a person’s health in other cancers.”
The analysis was published in JAMA Otolaryngology–Head and Neck Surgery.
When assessing survival, factors such as cancer stage and tumor size are key, but comorbidities also play a crucial role. For oral cancer in particular, where alcohol and tobacco use are notorious risk factors, comorbidities occur frequently and are often serious.
To create a model that provides more “holistic and personalized” estimates and includes a host of factors that can affect the risk of death, the authors tapped into data from the Surveillance, Epidemiology, and End Results database to develop the SEER Oral Cancer Survival Calculator.
Alongside data from the SEER database, the calculator used data from the National Health Interview Survey’s Longitudinal Mortality Files to obtain estimates of general health status, life expectancy without cancer, and the probability of dying from the cancer or from other causes within 1-10 years among people with newly diagnosed oral cancer.
Overall, the data included 22,392 patients, aged 20-94, with oral squamous cell carcinoma, 60.5% of whom were male and 78% White, as well as 402,626 interviewees from the survey. The calculator did not include patients with tonsil- or tongue-based cancers, which were not considered anatomically part of the oral cavity.
The most common conditions coexisting with oral cancer were diabetes and chronic obstructive pulmonary disease among older patients. Among those with oral cancer, more than half (52.8%) had none of the major coexisting conditions, which also included peripheral and cerebrovascular disease, compared with 80% of the Medicare population.
The researchers described and validated four models – one that estimated the probability of death due to oral cancer, and then three others that estimated the probability of death from other causes, with variations based on the specific data and covariates included.
Overall, the models in the calculator estimated that patients with oral cancer have a higher risk of death from other causes, compared with the general population, and survival estimates for noncancer causes got worse with more advanced cancer stage.
For instance, for a patient diagnosed with stage 3 oral cancer after age 50, the chances of being alive at age 70 were 60% for females and 44% for males in the absence of cancer, whereas the corresponding survival estimates in the general U.S. population were 86% for females and 79% for males – an absolute difference of 26 and 35 percentage points.
One key reason for this trend is that patients with later-stage cancers likely also have more coexisting health conditions, first author Louise Davies, MD, from the Geisel School of Medicine at Dartmouth, Lebanon, N.H., explained.
Another reason: For cancers with low enough mortality rates, people might be more likely to die from causes other than their cancer. This can also occur in ductal carcinoma in situ breast cancer or papillary thyroid cancer, noted Dr. Davies, also from the Department of Veterans Affairs Medical Center, White River Junction, Vt.
Commenting on the study, Eric Moore, MD, a head and neck surgeon with the Mayo Clinic in Rochester, Minn., said that while such prediction tools are important, they also come with caveats.
“I think these calculators are helpful and certainly having them widely available to people gives them another piece of knowledge that can be powerful,” he told this news organization. “But you want to make sure you don’t interpret them as the end-all, be-all message, because there are an infinite number of variables that could influence survival that aren’t available in some of these datasets.”
Neil D. Gross, MD, a professor of head and neck surgery at the University of Texas MD Anderson Cancer Center, Houston, agreed. Although this new calculator uses a large dataset, such tools “can be imperfect” and some factors simply can’t be calculated, such as a person’s priorities, Dr. Gross said.
That’s why there’s no substitute for having a “very personal discussion between a patient and a physician to decide what’s best.” And this calculator is just one tool to help with that process, Dr. Gross said.
The commentary authors echoed these sentiments. “This calculator can potentially bridge the gaps between the survival estimates in the literature, life tables, clinical gestalt, and physician attempts to contextualize the inherent limitations of applying survival curves and averages to the one patient with the diagnosis,” wrote Leila J. Mady, MD, PhD, MPH, Wayne M. Koch, MD, and Carole Fakhry, MD, MPH, all from Johns Hopkins School of Medicine, Baltimore.
But a caveat in providing such predictions is the possible psychological effect the news can have.
“Potential risks of revealing personalized prognostic survival estimates to patients include increased anxiety and distress surrounding competing causes of death [and] misinterpretation of data,” the commentary authors cautioned, adding that “we must present such information with grace and sensitivity.”
Dr. Davies recommends that clinicians ask patients what they want to know because that will vary by patient and potentially over time for the same patient.
“People are more than their cancer diagnosis,” said Dr. Davies. “Giving them the opportunity to consider their life as a whole is the aim.”
The oral cancer calculator can be publicly accessed through the National Cancer Institute. The study was supported by the Department of Veterans Affairs and the National Cancer Institute as part of an interagency agreement. The authors report no relevant financial relationships.
A version of this article appeared on Medscape.com.
FROM JAMA OTOLARYNGOLOGY–HEAD AND NECK SURGERY
Screening for hepatitis B: Where the CDC and USPSTF diverge
The Centers for Disease Control and Prevention (CDC) recently published new recommendations on screening for hepatitis B infection.1 They recommend screening all adults (ages 18 years and older) at least once.
These recommendations differ in a few ways from those of the US Preventive Services Task Force (USPSTF).2 This Practice Alert will highlight these differences but also point out areas of agreement between the 2 sets of recommendations—and discuss why 2 separate agencies in the US Department of Health and Human Services reached different conclusions on some issues.
First, some background on hepatitis B
An estimated 580,000 to 2.4 million people in the United States have chronic hepatitis B (CHB) infection—and as many as two-thirds are unaware of it.3 In 2020, the Department of Health and Human Services published the Viral Hepatitis National Strategic Plan for the United States with a stated goal of increasing awareness of infection status among those with hepatitis B virus (HBV) from 32% to 90% by 2030.4 People living in the United States but born outside the country are at highest risk for CHB; they account for 69% of those with the infection.5
The incidence of acute HBV infection has declined markedly since the HBV vaccine was recommended for high-risk adults in 1982 and universally for infants in 1991.6,7 Overall rates of HBV infection declined fairly steadily starting around 1987—but in 2014, rates began to increase, especially in those ages 40 to 59 years.8,9 In 2019, 3192 cases were reported; but when one factors in underreporting, the CDC estimates that the number is likely closer to 20,700.10 This uptick is one reason the Advisory Committee on Immunization Practices changed its HBV vaccination recommendation for adults from a risk-based to a universal recommendation for all unvaccinated adults through age 60 years.10
Chronic hepatitis B infection has serious consequences
The proportion of those infected with HBV who develop CHB differs by age at infection: 80% to 90% if infected during infancy, 30% if infected before age 6 years, and 1% to 12% if infected as an older child or adult.8
CHB infection can lead to chronic liver disease, including cirrhosis of the liver, liver cancer, and liver failure. About 25% of those who develop CHB infection during childhood and 15% of those who develop chronic infection after childhood will die prematurely from cirrhosis or liver cancer.8
The American Association for the Study of Liver Diseases (AASLD) classifies CHB into 4 phases that reflect the rate of viral replication and the patient’s immune response.11 These phases are:
- immune-tolerant (minimal inflammation and fibrosis)
- hepatitis B e-antigen (HBeAg)-positive immune-active (moderate-to-severe inflammation or fibrosis)
- inactive CHB (minimal necroinflammation but variable fibrosis), and
- HBeAg-negative immune reactivation (moderate-to-severe inflammation or fibrosis).11
Continue to: The progression from one phase...
The progression from one phase to the next varies by patient, and not all patients will progress through each phase. The AASLD recommends periodically monitoring the HBV DNA and alanine aminotransferase (ALT) levels in those with CHB to track the progression from one phase to the next and to guide treatment decisions.
Treatment can be beneficial for those who meet criteria
The evidence report prepared for USPSTF found that antiviral treatment of those with CHB infection resulted in improved intermediate outcomes (histologic improvement, loss of hepatitis B surface antigen [HBsAg], loss of HBeAg, HBeAg seroconversion, virologic suppression, and normalization of ALT levels). The magnitude of benefit varied by location and study design.12
In addition, the evidence review found that antiviral therapy was associated with a decreased risk for overall mortality (relative risk [RR] = 0.15; 95% CI, 0.03-0.69), cirrhosis (RR = 0.72; 95% CI, 0.29-1.77), and hepatocellular carcinoma (RR = 0.60; 95% CI, 0.16-2.33). However, these results came from studies that were “limited due to small numbers of trials, few events, and insufficient duration of follow-up.”12
The USPSTF and the CDC both judged that the intermediate outcome results, as well as findings that improved intermediate outcomes lead to decreases in chronic liver disease, are strong enough evidence for their recommendations.
However, not all patients with CHB infection require treatment; estimates of patients with HBV infection meeting AASLD criteria for treatment range from 24% to 48%.1 The AASLD guideline on the treatment of CHB infection is an excellent resource that makes recommendations on the initial evaluation, ongoing monitoring, and treatment decisions for those with CHB.11
Continue to: How CDC and USPSTF guidance on HBV screeinng differs
How CDC and USPSTF guidance on HBV screening differs
The CDC and USPSTF recommendations for HBV screening differ in 3 aspects: whom to screen, whom to classify as at high risk for HBV infection, and what tests to use for screening.
Who should be screened?
The USPSTF recommends screening adults and adolescents who are at high risk for HBV. The CDC recommends screening all adults at least once. Both entities agree that those who are at increased risk should be screened periodically, although the optimal frequency has not been established. The USPSTF does not recommend against screening for the general population, so universal screening (as advocated by the CDC) is not in direct conflict with the USPSTF’s recommendations.
Who is at increased risk for HBV infection?
The CDC and the USPSTF differ slightly on the factors they consider to constitute increased risk for HBV infection. These are listed in TABLE 1.1,2

The CDC lists 6 categories that the USPSTF does not mention. However, 4 of these categories are mentioned indirectly in the USPSTF evidence report that accompanies the recommendations, via statements that certain settings have high proportions of people at risk for HBV infection: sexually transmitted infection clinics; HIV testing and treatment centers; health care settings that target services toward people who inject drugs and men who have sex with men; correctional facilities; hemodialysis facilities; and institutions and nonresidential daycare centers for developmentally disabled persons. People who are served at most of these facilities are also at risk for hepatitis C virus infection.
Three categories are listed by the CDC and not by the USPSTF, in either the recommendation or evidence report. These include a history of multiple sex partners; elevated ALT or aspartate aminotransferase levels of unknown origin; and patient request for testing (because they may not want to reveal risk factors).
Continue to: What test(s) should be ordered?
What test(s) should be ordered?
The USPSTF recommends screening using HBsAg. The CDC recommends using triple-panel screening: HBsAg, anti-hepatitis B surface antigen (anti-HBs), and total antibody to hepatitis B core antigen (anti-HBc).
HBsAg indicates HBV infection, either acute or chronic, or a recent dose of HBV vaccine. Anti-HBs indicate recovery from HBV infection, response to HBV vaccine, or recent receipt of hepatitis B immune globulin. Total anti-HBc develops in all HBV infections, resolved or current, and usually persists for life. Vaccine-induced immunity does not cause anti-HBc to develop.
The USPSTF’s rationale is that testing for HBsAg is more than 98% sensitive and specific for detecting HBV infections.2 The CDC recommends triple testing because it can detect those with asymptomatic active HBV infections (this would be a rare occurrence); those who have resolved infection and might be susceptible to reactivation (eg, those who are immunosuppressed); and those who are susceptible and need vaccination.
Interpretation of HBV test results and suggested actions are described in TABLE 2.1,8,13
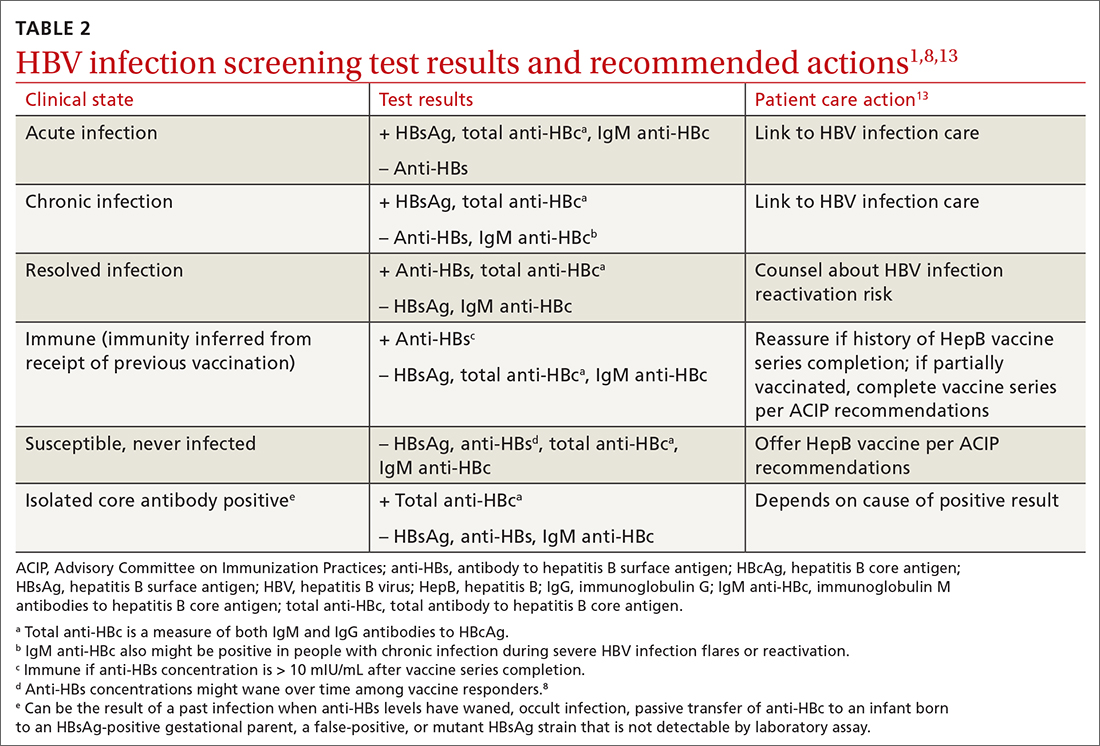
Why do the CDC and USPSTF differ?
While it would be optimal if the CDC and the USPSTF coordinated and harmonized recommendations, this is difficult to achieve given their different missions. The USPSTF is charged to make evidence-based recommendations about preventive services such as screenings, behavioral counseling, and preventive medications, which are provided by clinicians to individual patients. The Task Force uses a very strict evidence-based process and will not make recommendations unless there is adequate evidence of efficacy and safety. Members of the Task Force are primary care professionals, and their collaborating professional organizations are primary care focused.
The CDC takes a community-wide, public health perspective. The professionals that work there are not always clinicians. They strive to prevent as much illness as possible, using public health measures and making recommendations to clinicians. They collaborate with professional organizations; on topics such as hepatitis and other infectious diseases, they collaborate with specialty-oriented societies. Given the imperative to act with the best evidence available, their evidence assessment process is not as strict.
The result, at times, is slight differences in recommendations. However, the HBV screening recommendations from the CDC and the USPSTF agree more than they do not. Based on practice-specific characteristics, family physicians should decide if they want to screen all adults or only those at increased risk, and whether to use single- or triple-test screening.
1. Conners EE, Panagiotakopoulos L, Hofmeister MG, et al. Screening and testing for hepatitis B virus infection: CDC recommendations—United States, 2023. MMWR Recomm Rep. 2023;72:1-25. doi: 10.15585/mmwr.rr7201a1
2. USPSTF. Hepatitis B virus infection in adolescents and adults: screening. Final recommendation statement. Published December 15, 2020. Access June 21, 2023. www.uspreventiveser vicestaskforce.org/uspstf/recommendation/hepatitis-b-virus-infection-screening
3. Roberts H, Ly KN, Yin S, et al. Prevalence of HBV infection, vaccine-induced immunity, and susceptibility among at-risk populations: US households, 2013-2018. Hepatology. 2021;74:2353-2365. doi: 10.1002/hep.31991
4. US Department of Health and Human Services. Viral hepatitis national strategic plan for the United States: a roadmap to elimination (2021-2025). Published January 7, 2021. Accessed June 21, 2023. www.hhs.gov/sites/default/files/Viral-Hepatitis-National-Strategic-Plan-2021-2025.pdf
5. Wong RJ, Brosgart CL, Welch S, et al. An updated assessment of chronic hepatitis B prevalence among foreign-born persons living in the United States. Hepatology. 2021;74:607-626. doi: 10.1002/hep.31782
6. CDC. Recommendation of the Immunization Practices Advisory Committee (ACIP): inactivated hepatitis B virus vaccine. MMWR Morb Mortal Wkly Rep. 1982;31:317-318, 327-288.
7. CDC. Hepatitis B virus: a comprehensive strategy for eliminating transmission in the United States through universal childhood vaccination: recommendations of the Immunization Practices Advisory Committee. MMWR Morb Mortal Wkly Rep. 1991;40:1-25.
8. Schillie S, Vellozzi C, Reingold A, et al. Prevention of hepatitis B virus infection in the United States: recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm Rep. 2018;67:1-31. doi: 10.15585/mmwr.rr6701a1
9. CDC. Viral hepatitis surveillance 2019. Published July 2021. Accessed June 29, 2023. www.cdc.gov/hepatitis/statistics/2019surveillance/
10. Weng MK, Doshani M, Khan MA, et al. Universal hepatitis B vaccination in adults aged 19-59 years: updated recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morb Mortal Wkly Rep. 2022;71:477-483. doi: 10.15585/mmwr.mm7113a1
11. Terrault NA, Bzowej NH, Chang KM, et al; American Association for the Study of Liver Diseases. AASLD guidelines for treatment of chronic hepatitis B. Hepatology. 2016;63:261-283. doi: 10.1002/hep.28156
12. Chou R, Blazina I, Bougatsos C, et al. Screening for hepatitis B virus infection in nonpregnant adolescents and adults: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2020;324:2423-2436. doi: 10.1001/jama.2020.19750
13. Abara WE, Qaseem A, Schillie S, et al. Hepatitis B vaccination, screening, and linkage to care: best practice advice from the American College of Physicians and the Centers for Disease Control and Prevention. Ann Intern Med. 2017;167:794-804. doi: 10.7326/M17-110
The Centers for Disease Control and Prevention (CDC) recently published new recommendations on screening for hepatitis B infection.1 They recommend screening all adults (ages 18 years and older) at least once.
These recommendations differ in a few ways from those of the US Preventive Services Task Force (USPSTF).2 This Practice Alert will highlight these differences but also point out areas of agreement between the 2 sets of recommendations—and discuss why 2 separate agencies in the US Department of Health and Human Services reached different conclusions on some issues.
First, some background on hepatitis B
An estimated 580,000 to 2.4 million people in the United States have chronic hepatitis B (CHB) infection—and as many as two-thirds are unaware of it.3 In 2020, the Department of Health and Human Services published the Viral Hepatitis National Strategic Plan for the United States with a stated goal of increasing awareness of infection status among those with hepatitis B virus (HBV) from 32% to 90% by 2030.4 People living in the United States but born outside the country are at highest risk for CHB; they account for 69% of those with the infection.5
The incidence of acute HBV infection has declined markedly since the HBV vaccine was recommended for high-risk adults in 1982 and universally for infants in 1991.6,7 Overall rates of HBV infection declined fairly steadily starting around 1987—but in 2014, rates began to increase, especially in those ages 40 to 59 years.8,9 In 2019, 3192 cases were reported; but when one factors in underreporting, the CDC estimates that the number is likely closer to 20,700.10 This uptick is one reason the Advisory Committee on Immunization Practices changed its HBV vaccination recommendation for adults from a risk-based to a universal recommendation for all unvaccinated adults through age 60 years.10
Chronic hepatitis B infection has serious consequences
The proportion of those infected with HBV who develop CHB differs by age at infection: 80% to 90% if infected during infancy, 30% if infected before age 6 years, and 1% to 12% if infected as an older child or adult.8
CHB infection can lead to chronic liver disease, including cirrhosis of the liver, liver cancer, and liver failure. About 25% of those who develop CHB infection during childhood and 15% of those who develop chronic infection after childhood will die prematurely from cirrhosis or liver cancer.8
The American Association for the Study of Liver Diseases (AASLD) classifies CHB into 4 phases that reflect the rate of viral replication and the patient’s immune response.11 These phases are:
- immune-tolerant (minimal inflammation and fibrosis)
- hepatitis B e-antigen (HBeAg)-positive immune-active (moderate-to-severe inflammation or fibrosis)
- inactive CHB (minimal necroinflammation but variable fibrosis), and
- HBeAg-negative immune reactivation (moderate-to-severe inflammation or fibrosis).11
Continue to: The progression from one phase...
The progression from one phase to the next varies by patient, and not all patients will progress through each phase. The AASLD recommends periodically monitoring the HBV DNA and alanine aminotransferase (ALT) levels in those with CHB to track the progression from one phase to the next and to guide treatment decisions.
Treatment can be beneficial for those who meet criteria
The evidence report prepared for USPSTF found that antiviral treatment of those with CHB infection resulted in improved intermediate outcomes (histologic improvement, loss of hepatitis B surface antigen [HBsAg], loss of HBeAg, HBeAg seroconversion, virologic suppression, and normalization of ALT levels). The magnitude of benefit varied by location and study design.12
In addition, the evidence review found that antiviral therapy was associated with a decreased risk for overall mortality (relative risk [RR] = 0.15; 95% CI, 0.03-0.69), cirrhosis (RR = 0.72; 95% CI, 0.29-1.77), and hepatocellular carcinoma (RR = 0.60; 95% CI, 0.16-2.33). However, these results came from studies that were “limited due to small numbers of trials, few events, and insufficient duration of follow-up.”12
The USPSTF and the CDC both judged that the intermediate outcome results, as well as findings that improved intermediate outcomes lead to decreases in chronic liver disease, are strong enough evidence for their recommendations.
However, not all patients with CHB infection require treatment; estimates of patients with HBV infection meeting AASLD criteria for treatment range from 24% to 48%.1 The AASLD guideline on the treatment of CHB infection is an excellent resource that makes recommendations on the initial evaluation, ongoing monitoring, and treatment decisions for those with CHB.11
Continue to: How CDC and USPSTF guidance on HBV screeinng differs
How CDC and USPSTF guidance on HBV screening differs
The CDC and USPSTF recommendations for HBV screening differ in 3 aspects: whom to screen, whom to classify as at high risk for HBV infection, and what tests to use for screening.
Who should be screened?
The USPSTF recommends screening adults and adolescents who are at high risk for HBV. The CDC recommends screening all adults at least once. Both entities agree that those who are at increased risk should be screened periodically, although the optimal frequency has not been established. The USPSTF does not recommend against screening for the general population, so universal screening (as advocated by the CDC) is not in direct conflict with the USPSTF’s recommendations.
Who is at increased risk for HBV infection?
The CDC and the USPSTF differ slightly on the factors they consider to constitute increased risk for HBV infection. These are listed in TABLE 1.1,2

The CDC lists 6 categories that the USPSTF does not mention. However, 4 of these categories are mentioned indirectly in the USPSTF evidence report that accompanies the recommendations, via statements that certain settings have high proportions of people at risk for HBV infection: sexually transmitted infection clinics; HIV testing and treatment centers; health care settings that target services toward people who inject drugs and men who have sex with men; correctional facilities; hemodialysis facilities; and institutions and nonresidential daycare centers for developmentally disabled persons. People who are served at most of these facilities are also at risk for hepatitis C virus infection.
Three categories are listed by the CDC and not by the USPSTF, in either the recommendation or evidence report. These include a history of multiple sex partners; elevated ALT or aspartate aminotransferase levels of unknown origin; and patient request for testing (because they may not want to reveal risk factors).
Continue to: What test(s) should be ordered?
What test(s) should be ordered?
The USPSTF recommends screening using HBsAg. The CDC recommends using triple-panel screening: HBsAg, anti-hepatitis B surface antigen (anti-HBs), and total antibody to hepatitis B core antigen (anti-HBc).
HBsAg indicates HBV infection, either acute or chronic, or a recent dose of HBV vaccine. Anti-HBs indicate recovery from HBV infection, response to HBV vaccine, or recent receipt of hepatitis B immune globulin. Total anti-HBc develops in all HBV infections, resolved or current, and usually persists for life. Vaccine-induced immunity does not cause anti-HBc to develop.
The USPSTF’s rationale is that testing for HBsAg is more than 98% sensitive and specific for detecting HBV infections.2 The CDC recommends triple testing because it can detect those with asymptomatic active HBV infections (this would be a rare occurrence); those who have resolved infection and might be susceptible to reactivation (eg, those who are immunosuppressed); and those who are susceptible and need vaccination.
Interpretation of HBV test results and suggested actions are described in TABLE 2.1,8,13

Why do the CDC and USPSTF differ?
While it would be optimal if the CDC and the USPSTF coordinated and harmonized recommendations, this is difficult to achieve given their different missions. The USPSTF is charged to make evidence-based recommendations about preventive services such as screenings, behavioral counseling, and preventive medications, which are provided by clinicians to individual patients. The Task Force uses a very strict evidence-based process and will not make recommendations unless there is adequate evidence of efficacy and safety. Members of the Task Force are primary care professionals, and their collaborating professional organizations are primary care focused.
The CDC takes a community-wide, public health perspective. The professionals that work there are not always clinicians. They strive to prevent as much illness as possible, using public health measures and making recommendations to clinicians. They collaborate with professional organizations; on topics such as hepatitis and other infectious diseases, they collaborate with specialty-oriented societies. Given the imperative to act with the best evidence available, their evidence assessment process is not as strict.
The result, at times, is slight differences in recommendations. However, the HBV screening recommendations from the CDC and the USPSTF agree more than they do not. Based on practice-specific characteristics, family physicians should decide if they want to screen all adults or only those at increased risk, and whether to use single- or triple-test screening.
The Centers for Disease Control and Prevention (CDC) recently published new recommendations on screening for hepatitis B infection.1 They recommend screening all adults (ages 18 years and older) at least once.
These recommendations differ in a few ways from those of the US Preventive Services Task Force (USPSTF).2 This Practice Alert will highlight these differences but also point out areas of agreement between the 2 sets of recommendations—and discuss why 2 separate agencies in the US Department of Health and Human Services reached different conclusions on some issues.
First, some background on hepatitis B
An estimated 580,000 to 2.4 million people in the United States have chronic hepatitis B (CHB) infection—and as many as two-thirds are unaware of it.3 In 2020, the Department of Health and Human Services published the Viral Hepatitis National Strategic Plan for the United States with a stated goal of increasing awareness of infection status among those with hepatitis B virus (HBV) from 32% to 90% by 2030.4 People living in the United States but born outside the country are at highest risk for CHB; they account for 69% of those with the infection.5
The incidence of acute HBV infection has declined markedly since the HBV vaccine was recommended for high-risk adults in 1982 and universally for infants in 1991.6,7 Overall rates of HBV infection declined fairly steadily starting around 1987—but in 2014, rates began to increase, especially in those ages 40 to 59 years.8,9 In 2019, 3192 cases were reported; but when one factors in underreporting, the CDC estimates that the number is likely closer to 20,700.10 This uptick is one reason the Advisory Committee on Immunization Practices changed its HBV vaccination recommendation for adults from a risk-based to a universal recommendation for all unvaccinated adults through age 60 years.10
Chronic hepatitis B infection has serious consequences
The proportion of those infected with HBV who develop CHB differs by age at infection: 80% to 90% if infected during infancy, 30% if infected before age 6 years, and 1% to 12% if infected as an older child or adult.8
CHB infection can lead to chronic liver disease, including cirrhosis of the liver, liver cancer, and liver failure. About 25% of those who develop CHB infection during childhood and 15% of those who develop chronic infection after childhood will die prematurely from cirrhosis or liver cancer.8
The American Association for the Study of Liver Diseases (AASLD) classifies CHB into 4 phases that reflect the rate of viral replication and the patient’s immune response.11 These phases are:
- immune-tolerant (minimal inflammation and fibrosis)
- hepatitis B e-antigen (HBeAg)-positive immune-active (moderate-to-severe inflammation or fibrosis)
- inactive CHB (minimal necroinflammation but variable fibrosis), and
- HBeAg-negative immune reactivation (moderate-to-severe inflammation or fibrosis).11
Continue to: The progression from one phase...
The progression from one phase to the next varies by patient, and not all patients will progress through each phase. The AASLD recommends periodically monitoring the HBV DNA and alanine aminotransferase (ALT) levels in those with CHB to track the progression from one phase to the next and to guide treatment decisions.
Treatment can be beneficial for those who meet criteria
The evidence report prepared for USPSTF found that antiviral treatment of those with CHB infection resulted in improved intermediate outcomes (histologic improvement, loss of hepatitis B surface antigen [HBsAg], loss of HBeAg, HBeAg seroconversion, virologic suppression, and normalization of ALT levels). The magnitude of benefit varied by location and study design.12
In addition, the evidence review found that antiviral therapy was associated with a decreased risk for overall mortality (relative risk [RR] = 0.15; 95% CI, 0.03-0.69), cirrhosis (RR = 0.72; 95% CI, 0.29-1.77), and hepatocellular carcinoma (RR = 0.60; 95% CI, 0.16-2.33). However, these results came from studies that were “limited due to small numbers of trials, few events, and insufficient duration of follow-up.”12
The USPSTF and the CDC both judged that the intermediate outcome results, as well as findings that improved intermediate outcomes lead to decreases in chronic liver disease, are strong enough evidence for their recommendations.
However, not all patients with CHB infection require treatment; estimates of patients with HBV infection meeting AASLD criteria for treatment range from 24% to 48%.1 The AASLD guideline on the treatment of CHB infection is an excellent resource that makes recommendations on the initial evaluation, ongoing monitoring, and treatment decisions for those with CHB.11
Continue to: How CDC and USPSTF guidance on HBV screeinng differs
How CDC and USPSTF guidance on HBV screening differs
The CDC and USPSTF recommendations for HBV screening differ in 3 aspects: whom to screen, whom to classify as at high risk for HBV infection, and what tests to use for screening.
Who should be screened?
The USPSTF recommends screening adults and adolescents who are at high risk for HBV. The CDC recommends screening all adults at least once. Both entities agree that those who are at increased risk should be screened periodically, although the optimal frequency has not been established. The USPSTF does not recommend against screening for the general population, so universal screening (as advocated by the CDC) is not in direct conflict with the USPSTF’s recommendations.
Who is at increased risk for HBV infection?
The CDC and the USPSTF differ slightly on the factors they consider to constitute increased risk for HBV infection. These are listed in TABLE 1.1,2

The CDC lists 6 categories that the USPSTF does not mention. However, 4 of these categories are mentioned indirectly in the USPSTF evidence report that accompanies the recommendations, via statements that certain settings have high proportions of people at risk for HBV infection: sexually transmitted infection clinics; HIV testing and treatment centers; health care settings that target services toward people who inject drugs and men who have sex with men; correctional facilities; hemodialysis facilities; and institutions and nonresidential daycare centers for developmentally disabled persons. People who are served at most of these facilities are also at risk for hepatitis C virus infection.
Three categories are listed by the CDC and not by the USPSTF, in either the recommendation or evidence report. These include a history of multiple sex partners; elevated ALT or aspartate aminotransferase levels of unknown origin; and patient request for testing (because they may not want to reveal risk factors).
Continue to: What test(s) should be ordered?
What test(s) should be ordered?
The USPSTF recommends screening using HBsAg. The CDC recommends using triple-panel screening: HBsAg, anti-hepatitis B surface antigen (anti-HBs), and total antibody to hepatitis B core antigen (anti-HBc).
HBsAg indicates HBV infection, either acute or chronic, or a recent dose of HBV vaccine. Anti-HBs indicate recovery from HBV infection, response to HBV vaccine, or recent receipt of hepatitis B immune globulin. Total anti-HBc develops in all HBV infections, resolved or current, and usually persists for life. Vaccine-induced immunity does not cause anti-HBc to develop.
The USPSTF’s rationale is that testing for HBsAg is more than 98% sensitive and specific for detecting HBV infections.2 The CDC recommends triple testing because it can detect those with asymptomatic active HBV infections (this would be a rare occurrence); those who have resolved infection and might be susceptible to reactivation (eg, those who are immunosuppressed); and those who are susceptible and need vaccination.
Interpretation of HBV test results and suggested actions are described in TABLE 2.1,8,13

Why do the CDC and USPSTF differ?
While it would be optimal if the CDC and the USPSTF coordinated and harmonized recommendations, this is difficult to achieve given their different missions. The USPSTF is charged to make evidence-based recommendations about preventive services such as screenings, behavioral counseling, and preventive medications, which are provided by clinicians to individual patients. The Task Force uses a very strict evidence-based process and will not make recommendations unless there is adequate evidence of efficacy and safety. Members of the Task Force are primary care professionals, and their collaborating professional organizations are primary care focused.
The CDC takes a community-wide, public health perspective. The professionals that work there are not always clinicians. They strive to prevent as much illness as possible, using public health measures and making recommendations to clinicians. They collaborate with professional organizations; on topics such as hepatitis and other infectious diseases, they collaborate with specialty-oriented societies. Given the imperative to act with the best evidence available, their evidence assessment process is not as strict.
The result, at times, is slight differences in recommendations. However, the HBV screening recommendations from the CDC and the USPSTF agree more than they do not. Based on practice-specific characteristics, family physicians should decide if they want to screen all adults or only those at increased risk, and whether to use single- or triple-test screening.
1. Conners EE, Panagiotakopoulos L, Hofmeister MG, et al. Screening and testing for hepatitis B virus infection: CDC recommendations—United States, 2023. MMWR Recomm Rep. 2023;72:1-25. doi: 10.15585/mmwr.rr7201a1
2. USPSTF. Hepatitis B virus infection in adolescents and adults: screening. Final recommendation statement. Published December 15, 2020. Access June 21, 2023. www.uspreventiveser vicestaskforce.org/uspstf/recommendation/hepatitis-b-virus-infection-screening
3. Roberts H, Ly KN, Yin S, et al. Prevalence of HBV infection, vaccine-induced immunity, and susceptibility among at-risk populations: US households, 2013-2018. Hepatology. 2021;74:2353-2365. doi: 10.1002/hep.31991
4. US Department of Health and Human Services. Viral hepatitis national strategic plan for the United States: a roadmap to elimination (2021-2025). Published January 7, 2021. Accessed June 21, 2023. www.hhs.gov/sites/default/files/Viral-Hepatitis-National-Strategic-Plan-2021-2025.pdf
5. Wong RJ, Brosgart CL, Welch S, et al. An updated assessment of chronic hepatitis B prevalence among foreign-born persons living in the United States. Hepatology. 2021;74:607-626. doi: 10.1002/hep.31782
6. CDC. Recommendation of the Immunization Practices Advisory Committee (ACIP): inactivated hepatitis B virus vaccine. MMWR Morb Mortal Wkly Rep. 1982;31:317-318, 327-288.
7. CDC. Hepatitis B virus: a comprehensive strategy for eliminating transmission in the United States through universal childhood vaccination: recommendations of the Immunization Practices Advisory Committee. MMWR Morb Mortal Wkly Rep. 1991;40:1-25.
8. Schillie S, Vellozzi C, Reingold A, et al. Prevention of hepatitis B virus infection in the United States: recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm Rep. 2018;67:1-31. doi: 10.15585/mmwr.rr6701a1
9. CDC. Viral hepatitis surveillance 2019. Published July 2021. Accessed June 29, 2023. www.cdc.gov/hepatitis/statistics/2019surveillance/
10. Weng MK, Doshani M, Khan MA, et al. Universal hepatitis B vaccination in adults aged 19-59 years: updated recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morb Mortal Wkly Rep. 2022;71:477-483. doi: 10.15585/mmwr.mm7113a1
11. Terrault NA, Bzowej NH, Chang KM, et al; American Association for the Study of Liver Diseases. AASLD guidelines for treatment of chronic hepatitis B. Hepatology. 2016;63:261-283. doi: 10.1002/hep.28156
12. Chou R, Blazina I, Bougatsos C, et al. Screening for hepatitis B virus infection in nonpregnant adolescents and adults: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2020;324:2423-2436. doi: 10.1001/jama.2020.19750
13. Abara WE, Qaseem A, Schillie S, et al. Hepatitis B vaccination, screening, and linkage to care: best practice advice from the American College of Physicians and the Centers for Disease Control and Prevention. Ann Intern Med. 2017;167:794-804. doi: 10.7326/M17-110
1. Conners EE, Panagiotakopoulos L, Hofmeister MG, et al. Screening and testing for hepatitis B virus infection: CDC recommendations—United States, 2023. MMWR Recomm Rep. 2023;72:1-25. doi: 10.15585/mmwr.rr7201a1
2. USPSTF. Hepatitis B virus infection in adolescents and adults: screening. Final recommendation statement. Published December 15, 2020. Access June 21, 2023. www.uspreventiveser vicestaskforce.org/uspstf/recommendation/hepatitis-b-virus-infection-screening
3. Roberts H, Ly KN, Yin S, et al. Prevalence of HBV infection, vaccine-induced immunity, and susceptibility among at-risk populations: US households, 2013-2018. Hepatology. 2021;74:2353-2365. doi: 10.1002/hep.31991
4. US Department of Health and Human Services. Viral hepatitis national strategic plan for the United States: a roadmap to elimination (2021-2025). Published January 7, 2021. Accessed June 21, 2023. www.hhs.gov/sites/default/files/Viral-Hepatitis-National-Strategic-Plan-2021-2025.pdf
5. Wong RJ, Brosgart CL, Welch S, et al. An updated assessment of chronic hepatitis B prevalence among foreign-born persons living in the United States. Hepatology. 2021;74:607-626. doi: 10.1002/hep.31782
6. CDC. Recommendation of the Immunization Practices Advisory Committee (ACIP): inactivated hepatitis B virus vaccine. MMWR Morb Mortal Wkly Rep. 1982;31:317-318, 327-288.
7. CDC. Hepatitis B virus: a comprehensive strategy for eliminating transmission in the United States through universal childhood vaccination: recommendations of the Immunization Practices Advisory Committee. MMWR Morb Mortal Wkly Rep. 1991;40:1-25.
8. Schillie S, Vellozzi C, Reingold A, et al. Prevention of hepatitis B virus infection in the United States: recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm Rep. 2018;67:1-31. doi: 10.15585/mmwr.rr6701a1
9. CDC. Viral hepatitis surveillance 2019. Published July 2021. Accessed June 29, 2023. www.cdc.gov/hepatitis/statistics/2019surveillance/
10. Weng MK, Doshani M, Khan MA, et al. Universal hepatitis B vaccination in adults aged 19-59 years: updated recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR Morb Mortal Wkly Rep. 2022;71:477-483. doi: 10.15585/mmwr.mm7113a1
11. Terrault NA, Bzowej NH, Chang KM, et al; American Association for the Study of Liver Diseases. AASLD guidelines for treatment of chronic hepatitis B. Hepatology. 2016;63:261-283. doi: 10.1002/hep.28156
12. Chou R, Blazina I, Bougatsos C, et al. Screening for hepatitis B virus infection in nonpregnant adolescents and adults: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2020;324:2423-2436. doi: 10.1001/jama.2020.19750
13. Abara WE, Qaseem A, Schillie S, et al. Hepatitis B vaccination, screening, and linkage to care: best practice advice from the American College of Physicians and the Centers for Disease Control and Prevention. Ann Intern Med. 2017;167:794-804. doi: 10.7326/M17-110
Even exercise by ‘weekend warriors’ can cut CV risk
Moderate to vigorous physical activity (MVPA) is a familiar and established approach to reducing cardiovascular (CV) risk, but it’s often believed that the exercise should be spread out across the week rather than concentrated within a couple of days.
A challenge to that view comes from an observational study of accelerometer-confirmed exercise in almost 90,000 people in their 60s. It suggests,
Researchers compared three patterns of MVPA in their subjects who wore accelerometers on their wrists for 1 week. Active WW subjects obtained at least 2.5 hours of exercise weekly, with at least half the amount completed over 1-2 days; “active regular” subjects achieved that exercise level but not mostly during 1 or 2 days; and those who were “inactive” fell short of 2.5 hours of exercise during the week. The group used a median exercise threshold of 3 hours, 50 minutes in a separate analysis.
The “active” groups, compared with inactive subjects, achieved similar and significant reductions in risk for incident atrial fibrillation (AF), myocardial infarction (MI), stroke, and heart failure (HF) over a median follow-up of 6.3 years at both weekly exercise thresholds, the group reported.
“The take-home [message] is that efforts to optimize activity, even if concentrated within just a day or 2 each week, should be expected to result in improved cardiovascular risk profiles,” lead author Shaan Khurshid, MD, MPH, Massachusetts General Hospital, Boston, said in an interview.
The study was published online in JAMA.
The research “provides novel data on patterns of physical activity accumulation and the risk of developing cardiovascular diseases,” observed Peter Katzmarzyk, PhD, Pennington Biomedical Research Center, Baton Rouge, La., in an interview. He was not involved with the research. Its “marked strengths,” he noted, include a large sample population and “use of accelerometers to measure physical activity levels and patterns.”
Moreover, Dr. Katzmarzyk said, its findings are “important” for showing that physical activity “can be accumulated throughout the week in different ways, which opens up more options for busy people to get their physical activity in.”
Current guidelines from the World Health Organization and the American Heart Association recommend at least 150 minutes of MVPA weekly to lower risk for cardiovascular disease and death, but do not specify an optimal exercise time frame. The U.K. National Health Service recommends MVPA daily or spread evenly over perhaps 4-5 days.
“The weekend warrior pattern has been studied previously, but typically relying on self-reported data, which may be biased, or [in studies] too small to look at specific cardiovascular outcomes,” Dr. Khurshid explained.
In the UK Biobank database, he said, “We saw the opportunity to leverage the largest sample of measured activity to date” to address the question of whether exercise time pattern “affects specific major cardiovascular diseases differently,” Dr. Khurshid said
The primary analysis assessed exercise amount in a week based on the guideline-recommended threshold of at least 2.5 hours; a 3-hour, 50-minutes threshold was used in a secondary analysis. The group assessed multiple thresholds because optimal MVPS levels derived from wrist-based accelerometers are “unclear,” he said.
The sample consisted of 89,573 participants with a mean age 62; slightly more than half (56%) were women. Based on the weekly MVPA threshold of 2.5 hours , the WW, active regular, and inactive groups made up 42.2%, 24%, and 33.7% of the population, respectively.
Compared with the inactive group, the two active groups both showed significant risk reductions for the four clinical outcomes, to similar degrees, in multivariate analysis. The results were similar at the 230-minute weekly exercise threshold for incident AF, MI, and HF but not for stroke.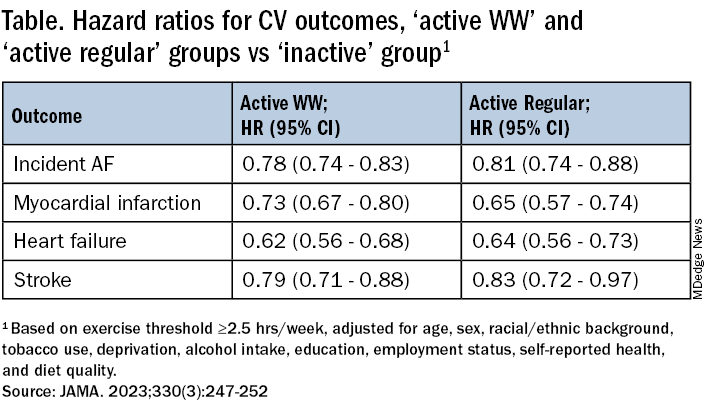
The findings were similarly consistent at the 3-hour, 50-minutes median threshold, although stroke differences were no longer significant.
Patients should be encouraged to exercise at recommended levels, “and should not be discouraged if, for whatever reasons, they are able to focus exercise within only 1 or a few days of the week,” said Dr. Khurshid. “Our findings suggest that it is the volume of activity, rather than the pattern, that matters most.”
The report notes several limitations of the study, including the exercise observation period limited to 1 week and that participants could have modified their behavior during the observation period. Also, the participants were almost all White, so the results may not be generalizable to other populations.
Clinicians should familiarize themselves with the “full range of recommendations” presented in the “Physical Activity Guidelines for Americans, 2nd Edition” “and personalize prescriptions by setting achievable physical activity goals” based on age, physical abilities, and activity levels, states an accompanying editorial from Dr. Katzmarzyk and John M. Jakicic, PhD, University of Kansas Medical Center, Kansas City.
Although MVPA at the recommended level of at least 2.5 hours per week will certainly be beneficial, they write, “the public health message should also clearly convey that every minute counts, especially among the three-quarters of U.S. adults who do not achieve that goal.”
Dr. Khurshid reported no relevant financial relationships; disclosures for the other authors are in the original article. Dr. Katzmarzyk reports no relevant financial relationships. Dr. Jakicic discloses receiving personal fees from Wondr Health, WW International (formerly Weight Watchers), and Educational Initiatives and grants from Epitomee Medical.
A version of this article appeared on Medscape.com.
Moderate to vigorous physical activity (MVPA) is a familiar and established approach to reducing cardiovascular (CV) risk, but it’s often believed that the exercise should be spread out across the week rather than concentrated within a couple of days.
A challenge to that view comes from an observational study of accelerometer-confirmed exercise in almost 90,000 people in their 60s. It suggests,
Researchers compared three patterns of MVPA in their subjects who wore accelerometers on their wrists for 1 week. Active WW subjects obtained at least 2.5 hours of exercise weekly, with at least half the amount completed over 1-2 days; “active regular” subjects achieved that exercise level but not mostly during 1 or 2 days; and those who were “inactive” fell short of 2.5 hours of exercise during the week. The group used a median exercise threshold of 3 hours, 50 minutes in a separate analysis.
The “active” groups, compared with inactive subjects, achieved similar and significant reductions in risk for incident atrial fibrillation (AF), myocardial infarction (MI), stroke, and heart failure (HF) over a median follow-up of 6.3 years at both weekly exercise thresholds, the group reported.
“The take-home [message] is that efforts to optimize activity, even if concentrated within just a day or 2 each week, should be expected to result in improved cardiovascular risk profiles,” lead author Shaan Khurshid, MD, MPH, Massachusetts General Hospital, Boston, said in an interview.
The study was published online in JAMA.
The research “provides novel data on patterns of physical activity accumulation and the risk of developing cardiovascular diseases,” observed Peter Katzmarzyk, PhD, Pennington Biomedical Research Center, Baton Rouge, La., in an interview. He was not involved with the research. Its “marked strengths,” he noted, include a large sample population and “use of accelerometers to measure physical activity levels and patterns.”
Moreover, Dr. Katzmarzyk said, its findings are “important” for showing that physical activity “can be accumulated throughout the week in different ways, which opens up more options for busy people to get their physical activity in.”
Current guidelines from the World Health Organization and the American Heart Association recommend at least 150 minutes of MVPA weekly to lower risk for cardiovascular disease and death, but do not specify an optimal exercise time frame. The U.K. National Health Service recommends MVPA daily or spread evenly over perhaps 4-5 days.
“The weekend warrior pattern has been studied previously, but typically relying on self-reported data, which may be biased, or [in studies] too small to look at specific cardiovascular outcomes,” Dr. Khurshid explained.
In the UK Biobank database, he said, “We saw the opportunity to leverage the largest sample of measured activity to date” to address the question of whether exercise time pattern “affects specific major cardiovascular diseases differently,” Dr. Khurshid said
The primary analysis assessed exercise amount in a week based on the guideline-recommended threshold of at least 2.5 hours; a 3-hour, 50-minutes threshold was used in a secondary analysis. The group assessed multiple thresholds because optimal MVPS levels derived from wrist-based accelerometers are “unclear,” he said.
The sample consisted of 89,573 participants with a mean age 62; slightly more than half (56%) were women. Based on the weekly MVPA threshold of 2.5 hours , the WW, active regular, and inactive groups made up 42.2%, 24%, and 33.7% of the population, respectively.
Compared with the inactive group, the two active groups both showed significant risk reductions for the four clinical outcomes, to similar degrees, in multivariate analysis. The results were similar at the 230-minute weekly exercise threshold for incident AF, MI, and HF but not for stroke.
The findings were similarly consistent at the 3-hour, 50-minutes median threshold, although stroke differences were no longer significant.
Patients should be encouraged to exercise at recommended levels, “and should not be discouraged if, for whatever reasons, they are able to focus exercise within only 1 or a few days of the week,” said Dr. Khurshid. “Our findings suggest that it is the volume of activity, rather than the pattern, that matters most.”
The report notes several limitations of the study, including the exercise observation period limited to 1 week and that participants could have modified their behavior during the observation period. Also, the participants were almost all White, so the results may not be generalizable to other populations.
Clinicians should familiarize themselves with the “full range of recommendations” presented in the “Physical Activity Guidelines for Americans, 2nd Edition” “and personalize prescriptions by setting achievable physical activity goals” based on age, physical abilities, and activity levels, states an accompanying editorial from Dr. Katzmarzyk and John M. Jakicic, PhD, University of Kansas Medical Center, Kansas City.
Although MVPA at the recommended level of at least 2.5 hours per week will certainly be beneficial, they write, “the public health message should also clearly convey that every minute counts, especially among the three-quarters of U.S. adults who do not achieve that goal.”
Dr. Khurshid reported no relevant financial relationships; disclosures for the other authors are in the original article. Dr. Katzmarzyk reports no relevant financial relationships. Dr. Jakicic discloses receiving personal fees from Wondr Health, WW International (formerly Weight Watchers), and Educational Initiatives and grants from Epitomee Medical.
A version of this article appeared on Medscape.com.
Moderate to vigorous physical activity (MVPA) is a familiar and established approach to reducing cardiovascular (CV) risk, but it’s often believed that the exercise should be spread out across the week rather than concentrated within a couple of days.
A challenge to that view comes from an observational study of accelerometer-confirmed exercise in almost 90,000 people in their 60s. It suggests,
Researchers compared three patterns of MVPA in their subjects who wore accelerometers on their wrists for 1 week. Active WW subjects obtained at least 2.5 hours of exercise weekly, with at least half the amount completed over 1-2 days; “active regular” subjects achieved that exercise level but not mostly during 1 or 2 days; and those who were “inactive” fell short of 2.5 hours of exercise during the week. The group used a median exercise threshold of 3 hours, 50 minutes in a separate analysis.
The “active” groups, compared with inactive subjects, achieved similar and significant reductions in risk for incident atrial fibrillation (AF), myocardial infarction (MI), stroke, and heart failure (HF) over a median follow-up of 6.3 years at both weekly exercise thresholds, the group reported.
“The take-home [message] is that efforts to optimize activity, even if concentrated within just a day or 2 each week, should be expected to result in improved cardiovascular risk profiles,” lead author Shaan Khurshid, MD, MPH, Massachusetts General Hospital, Boston, said in an interview.
The study was published online in JAMA.
The research “provides novel data on patterns of physical activity accumulation and the risk of developing cardiovascular diseases,” observed Peter Katzmarzyk, PhD, Pennington Biomedical Research Center, Baton Rouge, La., in an interview. He was not involved with the research. Its “marked strengths,” he noted, include a large sample population and “use of accelerometers to measure physical activity levels and patterns.”
Moreover, Dr. Katzmarzyk said, its findings are “important” for showing that physical activity “can be accumulated throughout the week in different ways, which opens up more options for busy people to get their physical activity in.”
Current guidelines from the World Health Organization and the American Heart Association recommend at least 150 minutes of MVPA weekly to lower risk for cardiovascular disease and death, but do not specify an optimal exercise time frame. The U.K. National Health Service recommends MVPA daily or spread evenly over perhaps 4-5 days.
“The weekend warrior pattern has been studied previously, but typically relying on self-reported data, which may be biased, or [in studies] too small to look at specific cardiovascular outcomes,” Dr. Khurshid explained.
In the UK Biobank database, he said, “We saw the opportunity to leverage the largest sample of measured activity to date” to address the question of whether exercise time pattern “affects specific major cardiovascular diseases differently,” Dr. Khurshid said
The primary analysis assessed exercise amount in a week based on the guideline-recommended threshold of at least 2.5 hours; a 3-hour, 50-minutes threshold was used in a secondary analysis. The group assessed multiple thresholds because optimal MVPS levels derived from wrist-based accelerometers are “unclear,” he said.
The sample consisted of 89,573 participants with a mean age 62; slightly more than half (56%) were women. Based on the weekly MVPA threshold of 2.5 hours , the WW, active regular, and inactive groups made up 42.2%, 24%, and 33.7% of the population, respectively.
Compared with the inactive group, the two active groups both showed significant risk reductions for the four clinical outcomes, to similar degrees, in multivariate analysis. The results were similar at the 230-minute weekly exercise threshold for incident AF, MI, and HF but not for stroke.
The findings were similarly consistent at the 3-hour, 50-minutes median threshold, although stroke differences were no longer significant.
Patients should be encouraged to exercise at recommended levels, “and should not be discouraged if, for whatever reasons, they are able to focus exercise within only 1 or a few days of the week,” said Dr. Khurshid. “Our findings suggest that it is the volume of activity, rather than the pattern, that matters most.”
The report notes several limitations of the study, including the exercise observation period limited to 1 week and that participants could have modified their behavior during the observation period. Also, the participants were almost all White, so the results may not be generalizable to other populations.
Clinicians should familiarize themselves with the “full range of recommendations” presented in the “Physical Activity Guidelines for Americans, 2nd Edition” “and personalize prescriptions by setting achievable physical activity goals” based on age, physical abilities, and activity levels, states an accompanying editorial from Dr. Katzmarzyk and John M. Jakicic, PhD, University of Kansas Medical Center, Kansas City.
Although MVPA at the recommended level of at least 2.5 hours per week will certainly be beneficial, they write, “the public health message should also clearly convey that every minute counts, especially among the three-quarters of U.S. adults who do not achieve that goal.”
Dr. Khurshid reported no relevant financial relationships; disclosures for the other authors are in the original article. Dr. Katzmarzyk reports no relevant financial relationships. Dr. Jakicic discloses receiving personal fees from Wondr Health, WW International (formerly Weight Watchers), and Educational Initiatives and grants from Epitomee Medical.
A version of this article appeared on Medscape.com.
FROM JAMA
FDA approves quizartinib for newly diagnosed AML
On July 20 the FDA also approved the LeukoStrat CDx FLT3 Mutation Assay to determine whether patients have this mutation.
The agency granted quizartinib a first-line indication for use in combination with standard chemotherapy – cytarabine and anthracycline induction followed by cytarabine consolidation – and as maintenance monotherapy afterwards, in adults whose tumors express FLT3-ITD.
The FLT3 protein is a tyrosine kinase receptor found on hematopoietic stem cells. Wild-type FLT3 promotes cell survival, growth, and differentiation, but ITD (internal tandem duplication)-mutated FLT3, which quizartinib targets, is associated with a higher relapse risk and shorter survival. About a quarter of AML patients carry the mutation.
Approval was based on the phase 3 QuANTUM-First trial in over 500 patients with the mutation. Median overall survival among patients on standard chemotherapy randomly assigned to quizartinib was 31.9 months versus 15.1 months in patients randomly assigned to placebo, a 22.4% reduction in the risk of death (P = .0324).
Quizartinib is not indicated as maintenance monotherapy after allogeneic hematopoietic stem cell transplantation.
In a company press release, the drug’s manufacturer Daiichi Sankyo said quizartinib will be available in the United States soon.
Company executive Ken Takeshita, MD, called the approval “an important milestone, as patients with the FLT3-ITD subtype of AML can now be treated with the first-ever FLT3 inhibitor approved across the three phases of treatment these patients typically receive.”
The FDA’s original decision date was April 24, but the agency pushed it back 3 months to review updates Daiichi Sankyo made to quizartinib’s Risk Evaluation and Mitigation Strategies (REMS) program in response to an agency request.
Quizartinib carries a boxed warning of QT prolongation, torsades de pointes, and cardiac arrest. Because of these risks, it’s only available through a new program, dubbed “Vanflyta REMS.”
In the trial, the most common adverse with quizartinib included lymphopenia (60%), hypokalemia (59%), hypoalbuminemia (53%), hypophosphatemia (52%), alkaline phosphatase increased (51%), hypomagnesemia (44%), febrile neutropenia (44%), diarrhea (42%), mucositis (38%), nausea (34%), and hypocalcemia (33%), among others.
The most common grade 3/4 adverse events were febrile neutropenia (43% with quizartinib vs. 41% with placebo), neutropenia (18% vs. 9%), hypokalemia (19% vs. 16%), and pneumonia (11% both). Adverse events were fatal in 11.3% of patients receiving quizartinib versus 9.7% of patients on placebo, mostly caused by infections.
In 2019, the FDA rejected quizartinib for FLT3-ITD mutated relapsed/refractory AML monotherapy in adults, after most of its oncology advisers thought the risk of treatment outweighed the benefits in an earlier trial.
A version of this article first appeared on Medscape.com.
On July 20 the FDA also approved the LeukoStrat CDx FLT3 Mutation Assay to determine whether patients have this mutation.
The agency granted quizartinib a first-line indication for use in combination with standard chemotherapy – cytarabine and anthracycline induction followed by cytarabine consolidation – and as maintenance monotherapy afterwards, in adults whose tumors express FLT3-ITD.
The FLT3 protein is a tyrosine kinase receptor found on hematopoietic stem cells. Wild-type FLT3 promotes cell survival, growth, and differentiation, but ITD (internal tandem duplication)-mutated FLT3, which quizartinib targets, is associated with a higher relapse risk and shorter survival. About a quarter of AML patients carry the mutation.
Approval was based on the phase 3 QuANTUM-First trial in over 500 patients with the mutation. Median overall survival among patients on standard chemotherapy randomly assigned to quizartinib was 31.9 months versus 15.1 months in patients randomly assigned to placebo, a 22.4% reduction in the risk of death (P = .0324).
Quizartinib is not indicated as maintenance monotherapy after allogeneic hematopoietic stem cell transplantation.
In a company press release, the drug’s manufacturer Daiichi Sankyo said quizartinib will be available in the United States soon.
Company executive Ken Takeshita, MD, called the approval “an important milestone, as patients with the FLT3-ITD subtype of AML can now be treated with the first-ever FLT3 inhibitor approved across the three phases of treatment these patients typically receive.”
The FDA’s original decision date was April 24, but the agency pushed it back 3 months to review updates Daiichi Sankyo made to quizartinib’s Risk Evaluation and Mitigation Strategies (REMS) program in response to an agency request.
Quizartinib carries a boxed warning of QT prolongation, torsades de pointes, and cardiac arrest. Because of these risks, it’s only available through a new program, dubbed “Vanflyta REMS.”
In the trial, the most common adverse with quizartinib included lymphopenia (60%), hypokalemia (59%), hypoalbuminemia (53%), hypophosphatemia (52%), alkaline phosphatase increased (51%), hypomagnesemia (44%), febrile neutropenia (44%), diarrhea (42%), mucositis (38%), nausea (34%), and hypocalcemia (33%), among others.
The most common grade 3/4 adverse events were febrile neutropenia (43% with quizartinib vs. 41% with placebo), neutropenia (18% vs. 9%), hypokalemia (19% vs. 16%), and pneumonia (11% both). Adverse events were fatal in 11.3% of patients receiving quizartinib versus 9.7% of patients on placebo, mostly caused by infections.
In 2019, the FDA rejected quizartinib for FLT3-ITD mutated relapsed/refractory AML monotherapy in adults, after most of its oncology advisers thought the risk of treatment outweighed the benefits in an earlier trial.
A version of this article first appeared on Medscape.com.
On July 20 the FDA also approved the LeukoStrat CDx FLT3 Mutation Assay to determine whether patients have this mutation.
The agency granted quizartinib a first-line indication for use in combination with standard chemotherapy – cytarabine and anthracycline induction followed by cytarabine consolidation – and as maintenance monotherapy afterwards, in adults whose tumors express FLT3-ITD.
The FLT3 protein is a tyrosine kinase receptor found on hematopoietic stem cells. Wild-type FLT3 promotes cell survival, growth, and differentiation, but ITD (internal tandem duplication)-mutated FLT3, which quizartinib targets, is associated with a higher relapse risk and shorter survival. About a quarter of AML patients carry the mutation.
Approval was based on the phase 3 QuANTUM-First trial in over 500 patients with the mutation. Median overall survival among patients on standard chemotherapy randomly assigned to quizartinib was 31.9 months versus 15.1 months in patients randomly assigned to placebo, a 22.4% reduction in the risk of death (P = .0324).
Quizartinib is not indicated as maintenance monotherapy after allogeneic hematopoietic stem cell transplantation.
In a company press release, the drug’s manufacturer Daiichi Sankyo said quizartinib will be available in the United States soon.
Company executive Ken Takeshita, MD, called the approval “an important milestone, as patients with the FLT3-ITD subtype of AML can now be treated with the first-ever FLT3 inhibitor approved across the three phases of treatment these patients typically receive.”
The FDA’s original decision date was April 24, but the agency pushed it back 3 months to review updates Daiichi Sankyo made to quizartinib’s Risk Evaluation and Mitigation Strategies (REMS) program in response to an agency request.
Quizartinib carries a boxed warning of QT prolongation, torsades de pointes, and cardiac arrest. Because of these risks, it’s only available through a new program, dubbed “Vanflyta REMS.”
In the trial, the most common adverse with quizartinib included lymphopenia (60%), hypokalemia (59%), hypoalbuminemia (53%), hypophosphatemia (52%), alkaline phosphatase increased (51%), hypomagnesemia (44%), febrile neutropenia (44%), diarrhea (42%), mucositis (38%), nausea (34%), and hypocalcemia (33%), among others.
The most common grade 3/4 adverse events were febrile neutropenia (43% with quizartinib vs. 41% with placebo), neutropenia (18% vs. 9%), hypokalemia (19% vs. 16%), and pneumonia (11% both). Adverse events were fatal in 11.3% of patients receiving quizartinib versus 9.7% of patients on placebo, mostly caused by infections.
In 2019, the FDA rejected quizartinib for FLT3-ITD mutated relapsed/refractory AML monotherapy in adults, after most of its oncology advisers thought the risk of treatment outweighed the benefits in an earlier trial.
A version of this article first appeared on Medscape.com.
