User login
Serum dupilumab levels do not predict clinical response
The finding that serum levels, according to a study published in JAMA Dermatology.
The study results mean that researchers should continue exploring potential AD drugs with novel mechanisms to help patients who fail type 2 inflammatory inhibition, experts told this news organization. The search for accurate augurs of clinical performance also must continue.
Addressing inadequate response
Quantifying nonresponse and incomplete response levels with dupilumab is difficult, said Jonathan I. Silverberg, MD, PhD, MPH, offering perspective on the study. “True nonresponse is probably less than 20%, but many other patients are inadequate responders even if they are having partial response.” Dr. Silverberg, professor of dermatology and director of clinical research, at George Washington University, Washington, was not an investigator.
Robert Sidbury, MD, MPH, added, “When a patient doesn’t respond to a medication that you expect they should, we always ask ourselves why.” Dermatologists have long assumed that, as with biologics for psoriasis, low blood levels were to blame for dupilumab nonresponse, said Dr. Sidbury, who is division chief of dermatology at Seattle Children’s Hospital and was not involved with the study. “This study showed that there was no correlation between response and blood levels.”
In the study, Lotte S. Spekhorst, MD, of National Expertise Center for Atopic Dermatitis, department of dermatology and allergology, University Medical Center Utrecht (the Netherlands) and coinvestigators prospectively followed 295 consecutive adult patients with moderate AD who were treated with dupilumab for 1 year. All patients received the same loading (600 mg) and biweekly (300 mg) doses.
The median dupilumab level at 16 weeks was 86.6 mcg/mL, which is higher than serum levels observed with other monoclonal antibodies used for other indications, such as psoriasis and inflammatory bowel disease, the authors noted. More importantly, researchers found no significant relationship between median week 16 dupilumab levels and 1-year clinical responses measured either discretely (Eczema Area and Severity Index [EASI] < 50, 50, 75, or 90; P = .18) or as quartiles (P = .06).
“It may be that response is dependent on target availability of the IL-4R-alpha, with an interpatient variability producing heterogeneity in response,” the authors wrote. But because serum dupilumab levels were relatively high, they said, all patients’ IL-4R-alpha “was likely fully saturated” at 16 weeks.
“This would explain why serum dupilumab levels were not related to effectiveness,” they noted, “although we cannot rule out differential effects in the tissue associated with heterogeneity in serum dupilumab levels.”
The study helps explain why some patients do not fully respond to dupilumab, said Eric L. Simpson, MD, professor of dermatology, Oregon Health & Science University, Portland, who was not involved with the study.
“One hypothesis would be that drug serum levels differ due to metabolism or absorption reasons,” Dr. Simpson said in an interview. Results also suggest that heterogeneity in disease biology, such as other uninhibited cytokine pathways, might explain differences in clinical results. “Thus, more therapeutics that target different inflammatory pathways are needed to capture responses in patients not adequately responding to type 2 inflammatory blockade,” he said.
Assessing AEs
As with response levels, serum dupilumab levels at week 16 did not predict AEs including dupilumab-associated ocular surface disease (DAOSD), which impacted 46.4% of 216 patients who reported AEs. These findings also contradict what happens with biologics in other diseases such as psoriasis and inflammatory bowel disease, said Dr. Sidbury, wherein serum drug levels may predict both clinical response and side-effect risks.
A previous study showed that lowering dupilumab levels led to improvement in DAOSD. Authors of the current study therefore surmised that DAOSD development might be more associated with interpatient variability in IL-4R-alpha expression than with serum drug levels. “More research is necessary to confirm the hypothesis of interpatient variability of the IL-4Ra and the pharmacokinetics of dupilumab,” they concluded.
For now, said Dr. Sidbury, the study helps clinicians look beyond serum drug levels when patients respond inadequately to dupilumab. Moreover, added Dr. Silverberg, study results mean that physicians must find other ways to predict dupilumab response levels. “We need better predictors of clinical response – theranostic markers that we could test the patient to and understand how well they’re going to do,” he said.
Be it dupilumab or any other medication, he said, physicians lack even confirmatory biomarkers to reflect when a drug is working well. “Right now, we go with clinical assessments. But if it’s not drug levels, we have to figure out why some patients do markedly better than others.”
It was not unreasonable, Dr. Silverberg said, for the investigators to seek a biomarker in blood rather than tissue. “But in this disease, we believe that the more important place to look for biomarkers and drug levels would be in the skin itself. So we are still left with the issue” that drug levels in tissue might reflect response when serum levels do not.
The study was supported by grants from AbbVie, Eli Lilly, Leo Pharma, Pfizer, and Sanofi. Study patients participated in the BioDay Registry, which is sponsored by Sanofi, Regeneron, AbbVie, Eli Lilly, LEO Pharma, and Pfizer; the sponsors had no role in the study design and conduct. Dr. Spekhorst discloses receiving speaking fees from Abbvie outside the work; disclosures of other authors included receiving advisory, speaking consulting, and/or investigator fees from Sanofi Genzyme during the study. Several authors had no disclosures.
Dr. Simpson has been an investigator and consultant for Regeneron and Sanofi, makers of dupilumab. Dr. Silverberg has been an investigator, consultant, and speaker for Regeneron and Sanofi. Dr. Sidbury has been a clinical investigator for all dupilumab pediatric trials. (His institution has a contract with Regeneron and Sanofi, but he receives no money from the arrangement.)
The finding that serum levels, according to a study published in JAMA Dermatology.
The study results mean that researchers should continue exploring potential AD drugs with novel mechanisms to help patients who fail type 2 inflammatory inhibition, experts told this news organization. The search for accurate augurs of clinical performance also must continue.
Addressing inadequate response
Quantifying nonresponse and incomplete response levels with dupilumab is difficult, said Jonathan I. Silverberg, MD, PhD, MPH, offering perspective on the study. “True nonresponse is probably less than 20%, but many other patients are inadequate responders even if they are having partial response.” Dr. Silverberg, professor of dermatology and director of clinical research, at George Washington University, Washington, was not an investigator.
Robert Sidbury, MD, MPH, added, “When a patient doesn’t respond to a medication that you expect they should, we always ask ourselves why.” Dermatologists have long assumed that, as with biologics for psoriasis, low blood levels were to blame for dupilumab nonresponse, said Dr. Sidbury, who is division chief of dermatology at Seattle Children’s Hospital and was not involved with the study. “This study showed that there was no correlation between response and blood levels.”
In the study, Lotte S. Spekhorst, MD, of National Expertise Center for Atopic Dermatitis, department of dermatology and allergology, University Medical Center Utrecht (the Netherlands) and coinvestigators prospectively followed 295 consecutive adult patients with moderate AD who were treated with dupilumab for 1 year. All patients received the same loading (600 mg) and biweekly (300 mg) doses.
The median dupilumab level at 16 weeks was 86.6 mcg/mL, which is higher than serum levels observed with other monoclonal antibodies used for other indications, such as psoriasis and inflammatory bowel disease, the authors noted. More importantly, researchers found no significant relationship between median week 16 dupilumab levels and 1-year clinical responses measured either discretely (Eczema Area and Severity Index [EASI] < 50, 50, 75, or 90; P = .18) or as quartiles (P = .06).
“It may be that response is dependent on target availability of the IL-4R-alpha, with an interpatient variability producing heterogeneity in response,” the authors wrote. But because serum dupilumab levels were relatively high, they said, all patients’ IL-4R-alpha “was likely fully saturated” at 16 weeks.
“This would explain why serum dupilumab levels were not related to effectiveness,” they noted, “although we cannot rule out differential effects in the tissue associated with heterogeneity in serum dupilumab levels.”
The study helps explain why some patients do not fully respond to dupilumab, said Eric L. Simpson, MD, professor of dermatology, Oregon Health & Science University, Portland, who was not involved with the study.
“One hypothesis would be that drug serum levels differ due to metabolism or absorption reasons,” Dr. Simpson said in an interview. Results also suggest that heterogeneity in disease biology, such as other uninhibited cytokine pathways, might explain differences in clinical results. “Thus, more therapeutics that target different inflammatory pathways are needed to capture responses in patients not adequately responding to type 2 inflammatory blockade,” he said.
Assessing AEs
As with response levels, serum dupilumab levels at week 16 did not predict AEs including dupilumab-associated ocular surface disease (DAOSD), which impacted 46.4% of 216 patients who reported AEs. These findings also contradict what happens with biologics in other diseases such as psoriasis and inflammatory bowel disease, said Dr. Sidbury, wherein serum drug levels may predict both clinical response and side-effect risks.
A previous study showed that lowering dupilumab levels led to improvement in DAOSD. Authors of the current study therefore surmised that DAOSD development might be more associated with interpatient variability in IL-4R-alpha expression than with serum drug levels. “More research is necessary to confirm the hypothesis of interpatient variability of the IL-4Ra and the pharmacokinetics of dupilumab,” they concluded.
For now, said Dr. Sidbury, the study helps clinicians look beyond serum drug levels when patients respond inadequately to dupilumab. Moreover, added Dr. Silverberg, study results mean that physicians must find other ways to predict dupilumab response levels. “We need better predictors of clinical response – theranostic markers that we could test the patient to and understand how well they’re going to do,” he said.
Be it dupilumab or any other medication, he said, physicians lack even confirmatory biomarkers to reflect when a drug is working well. “Right now, we go with clinical assessments. But if it’s not drug levels, we have to figure out why some patients do markedly better than others.”
It was not unreasonable, Dr. Silverberg said, for the investigators to seek a biomarker in blood rather than tissue. “But in this disease, we believe that the more important place to look for biomarkers and drug levels would be in the skin itself. So we are still left with the issue” that drug levels in tissue might reflect response when serum levels do not.
The study was supported by grants from AbbVie, Eli Lilly, Leo Pharma, Pfizer, and Sanofi. Study patients participated in the BioDay Registry, which is sponsored by Sanofi, Regeneron, AbbVie, Eli Lilly, LEO Pharma, and Pfizer; the sponsors had no role in the study design and conduct. Dr. Spekhorst discloses receiving speaking fees from Abbvie outside the work; disclosures of other authors included receiving advisory, speaking consulting, and/or investigator fees from Sanofi Genzyme during the study. Several authors had no disclosures.
Dr. Simpson has been an investigator and consultant for Regeneron and Sanofi, makers of dupilumab. Dr. Silverberg has been an investigator, consultant, and speaker for Regeneron and Sanofi. Dr. Sidbury has been a clinical investigator for all dupilumab pediatric trials. (His institution has a contract with Regeneron and Sanofi, but he receives no money from the arrangement.)
The finding that serum levels, according to a study published in JAMA Dermatology.
The study results mean that researchers should continue exploring potential AD drugs with novel mechanisms to help patients who fail type 2 inflammatory inhibition, experts told this news organization. The search for accurate augurs of clinical performance also must continue.
Addressing inadequate response
Quantifying nonresponse and incomplete response levels with dupilumab is difficult, said Jonathan I. Silverberg, MD, PhD, MPH, offering perspective on the study. “True nonresponse is probably less than 20%, but many other patients are inadequate responders even if they are having partial response.” Dr. Silverberg, professor of dermatology and director of clinical research, at George Washington University, Washington, was not an investigator.
Robert Sidbury, MD, MPH, added, “When a patient doesn’t respond to a medication that you expect they should, we always ask ourselves why.” Dermatologists have long assumed that, as with biologics for psoriasis, low blood levels were to blame for dupilumab nonresponse, said Dr. Sidbury, who is division chief of dermatology at Seattle Children’s Hospital and was not involved with the study. “This study showed that there was no correlation between response and blood levels.”
In the study, Lotte S. Spekhorst, MD, of National Expertise Center for Atopic Dermatitis, department of dermatology and allergology, University Medical Center Utrecht (the Netherlands) and coinvestigators prospectively followed 295 consecutive adult patients with moderate AD who were treated with dupilumab for 1 year. All patients received the same loading (600 mg) and biweekly (300 mg) doses.
The median dupilumab level at 16 weeks was 86.6 mcg/mL, which is higher than serum levels observed with other monoclonal antibodies used for other indications, such as psoriasis and inflammatory bowel disease, the authors noted. More importantly, researchers found no significant relationship between median week 16 dupilumab levels and 1-year clinical responses measured either discretely (Eczema Area and Severity Index [EASI] < 50, 50, 75, or 90; P = .18) or as quartiles (P = .06).
“It may be that response is dependent on target availability of the IL-4R-alpha, with an interpatient variability producing heterogeneity in response,” the authors wrote. But because serum dupilumab levels were relatively high, they said, all patients’ IL-4R-alpha “was likely fully saturated” at 16 weeks.
“This would explain why serum dupilumab levels were not related to effectiveness,” they noted, “although we cannot rule out differential effects in the tissue associated with heterogeneity in serum dupilumab levels.”
The study helps explain why some patients do not fully respond to dupilumab, said Eric L. Simpson, MD, professor of dermatology, Oregon Health & Science University, Portland, who was not involved with the study.
“One hypothesis would be that drug serum levels differ due to metabolism or absorption reasons,” Dr. Simpson said in an interview. Results also suggest that heterogeneity in disease biology, such as other uninhibited cytokine pathways, might explain differences in clinical results. “Thus, more therapeutics that target different inflammatory pathways are needed to capture responses in patients not adequately responding to type 2 inflammatory blockade,” he said.
Assessing AEs
As with response levels, serum dupilumab levels at week 16 did not predict AEs including dupilumab-associated ocular surface disease (DAOSD), which impacted 46.4% of 216 patients who reported AEs. These findings also contradict what happens with biologics in other diseases such as psoriasis and inflammatory bowel disease, said Dr. Sidbury, wherein serum drug levels may predict both clinical response and side-effect risks.
A previous study showed that lowering dupilumab levels led to improvement in DAOSD. Authors of the current study therefore surmised that DAOSD development might be more associated with interpatient variability in IL-4R-alpha expression than with serum drug levels. “More research is necessary to confirm the hypothesis of interpatient variability of the IL-4Ra and the pharmacokinetics of dupilumab,” they concluded.
For now, said Dr. Sidbury, the study helps clinicians look beyond serum drug levels when patients respond inadequately to dupilumab. Moreover, added Dr. Silverberg, study results mean that physicians must find other ways to predict dupilumab response levels. “We need better predictors of clinical response – theranostic markers that we could test the patient to and understand how well they’re going to do,” he said.
Be it dupilumab or any other medication, he said, physicians lack even confirmatory biomarkers to reflect when a drug is working well. “Right now, we go with clinical assessments. But if it’s not drug levels, we have to figure out why some patients do markedly better than others.”
It was not unreasonable, Dr. Silverberg said, for the investigators to seek a biomarker in blood rather than tissue. “But in this disease, we believe that the more important place to look for biomarkers and drug levels would be in the skin itself. So we are still left with the issue” that drug levels in tissue might reflect response when serum levels do not.
The study was supported by grants from AbbVie, Eli Lilly, Leo Pharma, Pfizer, and Sanofi. Study patients participated in the BioDay Registry, which is sponsored by Sanofi, Regeneron, AbbVie, Eli Lilly, LEO Pharma, and Pfizer; the sponsors had no role in the study design and conduct. Dr. Spekhorst discloses receiving speaking fees from Abbvie outside the work; disclosures of other authors included receiving advisory, speaking consulting, and/or investigator fees from Sanofi Genzyme during the study. Several authors had no disclosures.
Dr. Simpson has been an investigator and consultant for Regeneron and Sanofi, makers of dupilumab. Dr. Silverberg has been an investigator, consultant, and speaker for Regeneron and Sanofi. Dr. Sidbury has been a clinical investigator for all dupilumab pediatric trials. (His institution has a contract with Regeneron and Sanofi, but he receives no money from the arrangement.)
FROM JAMA DERMATOLOGY
Monkeypox in children appears rare and relatively mild
Monkeypox virus infections in children and adolescents in the United States are rare, and young patients with known infections have all recovered, according to a study from the Centers for Disease Control and Prevention.
In addition, evidence suggests that secondary transmission in schools or childcare facilities may be unlikely.
The study was the first comprehensive study on the impact of monkeypox on children during the 2022 outbreak, according to a statement emailed to this news organization from the California Department of Public Health, one of the state health departments that partnered with the CDC to share information.
News of low infection rates and relatively mild disease was welcome to clinicians, who had braced for severe findings on the basis of sparse prior data, according to Peter Chin-Hong, MD, a professor of medicine and an infectious diseases physician at the University of California, San Francisco.
“We were on heightened alert that kids may do poorly,” said Dr. Chin-Hong, who was not involved in the study but who cared for monkeypox patients during the outbreak. “I think this study is reassuring.
“The other silver lining about it is that most of the kids got infected in the household setting from ways that you would expect them to get [infected],” Dr. Chin-Hong said in an interview.
However, Black and Hispanic children were more likely to contract the disease, underscoring troubling inequities.
“Early on, individuals of color were much less likely to be able to successfully access vaccination,” said first author Ian Hennessee, PhD, MPH, an epidemic intelligence service officer with the CDC and a member of the Special Case Investigation Unit of the Multinational Monkeypox Response Team at the CDC. “We think those kinds of structural inequities really trickled down towards the children and adolescents that have been affected by this outbreak.”
The study was published in Morbidity and Mortality Weekly Report.
A nationwide look at the data
The researchers discussed 83 children and adolescents with monkeypox who came to the CDC’s attention between May 17 and Sept. 24, 2022.
The 83 cases represent 0.3% of the 25,038 reported monkeypox cases in the United States over that period. Of the 28 children aged 12 years or younger, 18 (64%) were boys. Sixteen children were younger than 4 years.
Exposure data were available for 20 (71%) of those aged 0-12. In that group, 19 were exposed at home; 17 cases were due to routine skin-to-skin contact with a household caregiver; and one case was suspected to be caused by fomites (such as a shared towel). Exposure information was unavailable for the remaining case.
Most of the children experienced lesions on the trunk. No lesions were anogenital. Two patients in the youngest age group were hospitalized because of widespread rash that involved the eyelids, and a patient in the 5- to 12-year-old group was hospitalized because of periorbital cellulitis and conjunctivitis.
Among those aged 13-17, there were 55 cases. Of these patients, 89% were boys. Exposure data were available for 35 (64%). In 32 of these patients, the infection occurred from presumed sexual contact. Twenty-three of those adolescents reported male-to-male sexual contact. No case was found to be connected with sexual abuse.
Lesions in the adolescents were mostly truncal or anogenital. Six in this group were hospitalized, and all of them recovered. One adolescent was found to be HIV positive.
Black and Hispanic children accounted for 47% and 35% of all cases, respectively.
Eleven percent of all the children and adolescents were hospitalized, and none received intensive care.
Treatments, when given, included the antiviral drug tecovirimat, intravenous vaccinia immune globulin, and topical trifluridine. There were no deaths.
Ten symptomatic patients attended school or daycare. Among these patients, no secondary transmissions were found to have occurred. Some contacts were offered the JYNNEOS monkeypox vaccine as postexposure prophylaxis.
Limitations of the study included potentially overlooked cases. Data were collected through routine surveillance, children frequently experience rashes, and access to testing has been a challenge, Dr. Hennessee explained.
In addition, data on exposure characteristics were missing for some children.
Inequities and the risks of being judged
The outbreak in the United States has eased in recent months. However, though uncommon in children, monkeypox has affected some racial groups disproportionately.
“Especially in the later course of the outbreak, the majority of cases were among Black and Hispanic individuals,” said co-author Rachel E. Harold, MD, an infectious diseases specialist and supervisory medical officer with the District of Columbia Department of Health’s HIV/AIDS, Hepatitis, STDs, and TB Administration.
“Unfortunately, the pediatric cases do reflect the outbreak overall,” she told this news organization.
Dr. Harold noted there have been efforts in D.C. and other jurisdictions, as well as by the White House monkeypox response team, to reach populations at greatest risk and that they were “really trying to make vaccine available to people of color.”
Vaccination clinics often popped up in unexpected locations at short notice, and that made it hard for some people to get to them, Dr. Chin-Hong pointed out.
Another factor was “the public aspect of accessing diagnostics and vaccines and the way that that’s linked to potential judgment or sexual risk,” he added.
“Not everybody’s out,” Dr. Chin-Hong said, referring to members of the LGBTQ community. “In many communities of color, going to get a test or going to get a vaccine essentially means that you’re out.”
For clinicians who suspect monkeypox in a child, Dr. Harold suggests keeping a broad differential diagnosis, looking for an epidemiologic link, and contacting the CDC for assistance. Infected children should be encouraged to avoid touching their own eyes or mucous membranes, she added.
In addition, she said, tecovirimat is a reasonable treatment and is well tolerated by pediatric monkeypox patients with eczema, an underlying condition that could lead to severe disease.
For infected caregivers, Dr. Hennessee said, measures to prevent infecting children at home include isolation, contact precautions, and in some cases, postexposure prophylaxis via vaccination.
For sexually active adolescents, he advised that clinicians offer vaccination, education on sexual health, and testing for HIV and other sexually transmitted infections.
“It’s important to remember that adolescents may be sexually active, and clinicians should do a thorough and nonjudgmental sexual history,” Dr. Harold added. “That is always true, but especially if there is concern for [monkeypox].”
Dr. Hennessee, Dr. Chin-Hong, and Dr. Harold have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Monkeypox virus infections in children and adolescents in the United States are rare, and young patients with known infections have all recovered, according to a study from the Centers for Disease Control and Prevention.
In addition, evidence suggests that secondary transmission in schools or childcare facilities may be unlikely.
The study was the first comprehensive study on the impact of monkeypox on children during the 2022 outbreak, according to a statement emailed to this news organization from the California Department of Public Health, one of the state health departments that partnered with the CDC to share information.
News of low infection rates and relatively mild disease was welcome to clinicians, who had braced for severe findings on the basis of sparse prior data, according to Peter Chin-Hong, MD, a professor of medicine and an infectious diseases physician at the University of California, San Francisco.
“We were on heightened alert that kids may do poorly,” said Dr. Chin-Hong, who was not involved in the study but who cared for monkeypox patients during the outbreak. “I think this study is reassuring.
“The other silver lining about it is that most of the kids got infected in the household setting from ways that you would expect them to get [infected],” Dr. Chin-Hong said in an interview.
However, Black and Hispanic children were more likely to contract the disease, underscoring troubling inequities.
“Early on, individuals of color were much less likely to be able to successfully access vaccination,” said first author Ian Hennessee, PhD, MPH, an epidemic intelligence service officer with the CDC and a member of the Special Case Investigation Unit of the Multinational Monkeypox Response Team at the CDC. “We think those kinds of structural inequities really trickled down towards the children and adolescents that have been affected by this outbreak.”
The study was published in Morbidity and Mortality Weekly Report.
A nationwide look at the data
The researchers discussed 83 children and adolescents with monkeypox who came to the CDC’s attention between May 17 and Sept. 24, 2022.
The 83 cases represent 0.3% of the 25,038 reported monkeypox cases in the United States over that period. Of the 28 children aged 12 years or younger, 18 (64%) were boys. Sixteen children were younger than 4 years.
Exposure data were available for 20 (71%) of those aged 0-12. In that group, 19 were exposed at home; 17 cases were due to routine skin-to-skin contact with a household caregiver; and one case was suspected to be caused by fomites (such as a shared towel). Exposure information was unavailable for the remaining case.
Most of the children experienced lesions on the trunk. No lesions were anogenital. Two patients in the youngest age group were hospitalized because of widespread rash that involved the eyelids, and a patient in the 5- to 12-year-old group was hospitalized because of periorbital cellulitis and conjunctivitis.
Among those aged 13-17, there were 55 cases. Of these patients, 89% were boys. Exposure data were available for 35 (64%). In 32 of these patients, the infection occurred from presumed sexual contact. Twenty-three of those adolescents reported male-to-male sexual contact. No case was found to be connected with sexual abuse.
Lesions in the adolescents were mostly truncal or anogenital. Six in this group were hospitalized, and all of them recovered. One adolescent was found to be HIV positive.
Black and Hispanic children accounted for 47% and 35% of all cases, respectively.
Eleven percent of all the children and adolescents were hospitalized, and none received intensive care.
Treatments, when given, included the antiviral drug tecovirimat, intravenous vaccinia immune globulin, and topical trifluridine. There were no deaths.
Ten symptomatic patients attended school or daycare. Among these patients, no secondary transmissions were found to have occurred. Some contacts were offered the JYNNEOS monkeypox vaccine as postexposure prophylaxis.
Limitations of the study included potentially overlooked cases. Data were collected through routine surveillance, children frequently experience rashes, and access to testing has been a challenge, Dr. Hennessee explained.
In addition, data on exposure characteristics were missing for some children.
Inequities and the risks of being judged
The outbreak in the United States has eased in recent months. However, though uncommon in children, monkeypox has affected some racial groups disproportionately.
“Especially in the later course of the outbreak, the majority of cases were among Black and Hispanic individuals,” said co-author Rachel E. Harold, MD, an infectious diseases specialist and supervisory medical officer with the District of Columbia Department of Health’s HIV/AIDS, Hepatitis, STDs, and TB Administration.
“Unfortunately, the pediatric cases do reflect the outbreak overall,” she told this news organization.
Dr. Harold noted there have been efforts in D.C. and other jurisdictions, as well as by the White House monkeypox response team, to reach populations at greatest risk and that they were “really trying to make vaccine available to people of color.”
Vaccination clinics often popped up in unexpected locations at short notice, and that made it hard for some people to get to them, Dr. Chin-Hong pointed out.
Another factor was “the public aspect of accessing diagnostics and vaccines and the way that that’s linked to potential judgment or sexual risk,” he added.
“Not everybody’s out,” Dr. Chin-Hong said, referring to members of the LGBTQ community. “In many communities of color, going to get a test or going to get a vaccine essentially means that you’re out.”
For clinicians who suspect monkeypox in a child, Dr. Harold suggests keeping a broad differential diagnosis, looking for an epidemiologic link, and contacting the CDC for assistance. Infected children should be encouraged to avoid touching their own eyes or mucous membranes, she added.
In addition, she said, tecovirimat is a reasonable treatment and is well tolerated by pediatric monkeypox patients with eczema, an underlying condition that could lead to severe disease.
For infected caregivers, Dr. Hennessee said, measures to prevent infecting children at home include isolation, contact precautions, and in some cases, postexposure prophylaxis via vaccination.
For sexually active adolescents, he advised that clinicians offer vaccination, education on sexual health, and testing for HIV and other sexually transmitted infections.
“It’s important to remember that adolescents may be sexually active, and clinicians should do a thorough and nonjudgmental sexual history,” Dr. Harold added. “That is always true, but especially if there is concern for [monkeypox].”
Dr. Hennessee, Dr. Chin-Hong, and Dr. Harold have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Monkeypox virus infections in children and adolescents in the United States are rare, and young patients with known infections have all recovered, according to a study from the Centers for Disease Control and Prevention.
In addition, evidence suggests that secondary transmission in schools or childcare facilities may be unlikely.
The study was the first comprehensive study on the impact of monkeypox on children during the 2022 outbreak, according to a statement emailed to this news organization from the California Department of Public Health, one of the state health departments that partnered with the CDC to share information.
News of low infection rates and relatively mild disease was welcome to clinicians, who had braced for severe findings on the basis of sparse prior data, according to Peter Chin-Hong, MD, a professor of medicine and an infectious diseases physician at the University of California, San Francisco.
“We were on heightened alert that kids may do poorly,” said Dr. Chin-Hong, who was not involved in the study but who cared for monkeypox patients during the outbreak. “I think this study is reassuring.
“The other silver lining about it is that most of the kids got infected in the household setting from ways that you would expect them to get [infected],” Dr. Chin-Hong said in an interview.
However, Black and Hispanic children were more likely to contract the disease, underscoring troubling inequities.
“Early on, individuals of color were much less likely to be able to successfully access vaccination,” said first author Ian Hennessee, PhD, MPH, an epidemic intelligence service officer with the CDC and a member of the Special Case Investigation Unit of the Multinational Monkeypox Response Team at the CDC. “We think those kinds of structural inequities really trickled down towards the children and adolescents that have been affected by this outbreak.”
The study was published in Morbidity and Mortality Weekly Report.
A nationwide look at the data
The researchers discussed 83 children and adolescents with monkeypox who came to the CDC’s attention between May 17 and Sept. 24, 2022.
The 83 cases represent 0.3% of the 25,038 reported monkeypox cases in the United States over that period. Of the 28 children aged 12 years or younger, 18 (64%) were boys. Sixteen children were younger than 4 years.
Exposure data were available for 20 (71%) of those aged 0-12. In that group, 19 were exposed at home; 17 cases were due to routine skin-to-skin contact with a household caregiver; and one case was suspected to be caused by fomites (such as a shared towel). Exposure information was unavailable for the remaining case.
Most of the children experienced lesions on the trunk. No lesions were anogenital. Two patients in the youngest age group were hospitalized because of widespread rash that involved the eyelids, and a patient in the 5- to 12-year-old group was hospitalized because of periorbital cellulitis and conjunctivitis.
Among those aged 13-17, there were 55 cases. Of these patients, 89% were boys. Exposure data were available for 35 (64%). In 32 of these patients, the infection occurred from presumed sexual contact. Twenty-three of those adolescents reported male-to-male sexual contact. No case was found to be connected with sexual abuse.
Lesions in the adolescents were mostly truncal or anogenital. Six in this group were hospitalized, and all of them recovered. One adolescent was found to be HIV positive.
Black and Hispanic children accounted for 47% and 35% of all cases, respectively.
Eleven percent of all the children and adolescents were hospitalized, and none received intensive care.
Treatments, when given, included the antiviral drug tecovirimat, intravenous vaccinia immune globulin, and topical trifluridine. There were no deaths.
Ten symptomatic patients attended school or daycare. Among these patients, no secondary transmissions were found to have occurred. Some contacts were offered the JYNNEOS monkeypox vaccine as postexposure prophylaxis.
Limitations of the study included potentially overlooked cases. Data were collected through routine surveillance, children frequently experience rashes, and access to testing has been a challenge, Dr. Hennessee explained.
In addition, data on exposure characteristics were missing for some children.
Inequities and the risks of being judged
The outbreak in the United States has eased in recent months. However, though uncommon in children, monkeypox has affected some racial groups disproportionately.
“Especially in the later course of the outbreak, the majority of cases were among Black and Hispanic individuals,” said co-author Rachel E. Harold, MD, an infectious diseases specialist and supervisory medical officer with the District of Columbia Department of Health’s HIV/AIDS, Hepatitis, STDs, and TB Administration.
“Unfortunately, the pediatric cases do reflect the outbreak overall,” she told this news organization.
Dr. Harold noted there have been efforts in D.C. and other jurisdictions, as well as by the White House monkeypox response team, to reach populations at greatest risk and that they were “really trying to make vaccine available to people of color.”
Vaccination clinics often popped up in unexpected locations at short notice, and that made it hard for some people to get to them, Dr. Chin-Hong pointed out.
Another factor was “the public aspect of accessing diagnostics and vaccines and the way that that’s linked to potential judgment or sexual risk,” he added.
“Not everybody’s out,” Dr. Chin-Hong said, referring to members of the LGBTQ community. “In many communities of color, going to get a test or going to get a vaccine essentially means that you’re out.”
For clinicians who suspect monkeypox in a child, Dr. Harold suggests keeping a broad differential diagnosis, looking for an epidemiologic link, and contacting the CDC for assistance. Infected children should be encouraged to avoid touching their own eyes or mucous membranes, she added.
In addition, she said, tecovirimat is a reasonable treatment and is well tolerated by pediatric monkeypox patients with eczema, an underlying condition that could lead to severe disease.
For infected caregivers, Dr. Hennessee said, measures to prevent infecting children at home include isolation, contact precautions, and in some cases, postexposure prophylaxis via vaccination.
For sexually active adolescents, he advised that clinicians offer vaccination, education on sexual health, and testing for HIV and other sexually transmitted infections.
“It’s important to remember that adolescents may be sexually active, and clinicians should do a thorough and nonjudgmental sexual history,” Dr. Harold added. “That is always true, but especially if there is concern for [monkeypox].”
Dr. Hennessee, Dr. Chin-Hong, and Dr. Harold have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
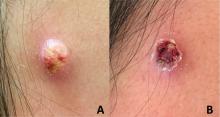
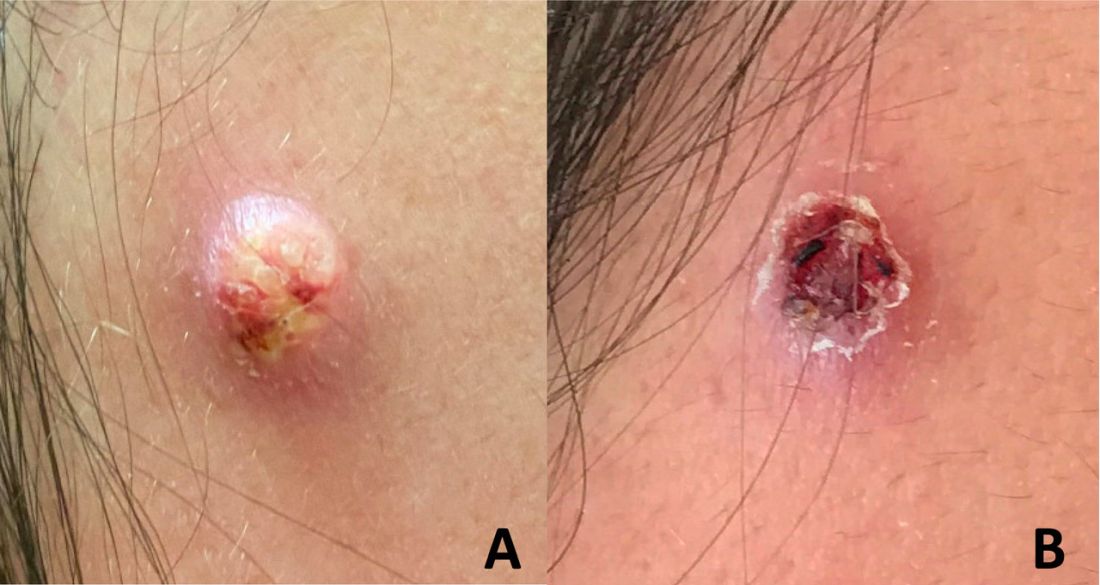
An adolescent male presents with an eroded bump on the temple
The correct answer is (D), molluscum contagiosum. Upon surgical excision, the pathology indicated the lesion was consistent with molluscum contagiosum.
Molluscum contagiosum is a benign skin disorder caused by a pox virus and is frequently seen in children. This disease is transmitted primarily through direct skin contact with an infected individual.1 Contaminated fomites have been suggested as another source of infection.2 The typical lesion appears dome-shaped, round, and pinkish-purple in color.1 The incubation period ranges from 2 weeks to 6 months and is typically self-limited in immunocompetent hosts; however, in immunocompromised persons, molluscum contagiosum lesions may present atypically such that they are larger in size and/or resemble malignancies, such as basal cell carcinoma or keratoacanthoma (for single lesions), or other infectious diseases, such as cryptococcosis and histoplasmosis (for more numerous lesions).3,4 A giant atypical molluscum contagiosum is rarely seen in healthy individuals.
What’s on the differential?
The recent episode of bleeding raises concern for other neoplastic processes of the skin including squamous cell carcinoma or basal cell carcinoma as well as cutaneous metastatic rhabdoid tumor, given the patient’s history.
Eruptive keratoacanthomas are also reported in patients taking nivolumab, an anti-PD-1 immunotherapy, which the patient has received for treatment of his recurrent metastatic rhabdoid tumor.5 More common entities such as a pyogenic granuloma or verruca are also included on the differential. The initial presentation of the lesion, however, is more consistent with the pearly umbilicated papules associated with molluscum contagiosum.
Comments from Dr. Eichenfield
This is a very hard diagnosis to make with the clinical findings and history.
Molluscum contagiosum infections are common, but with this patient’s medical history, biopsy and excision with pathologic examination was an appropriate approach to make a certain diagnosis.
Ms. Moyal is a research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego.
References
1. Brown J et al. Int J Dermatol. 2006 Feb;45(2):93-9.
2. Hanson D and Diven DG. Dermatol Online J. 2003 Mar;9(2).
3. Badri T and Gandhi GR. Molluscum contagiosum. 2022. In: StatPearls [Internet]. Treasure Island, Fla.: StatPearls Publishing.
4. Schwartz JJ and Myskowski PL. J Am Acad Dermatol. 1992 Oct 1;27(4):583-8.
5. Antonov NK et al. JAAD Case Rep. 2019 Apr 5;5(4):342-5.
The correct answer is (D), molluscum contagiosum. Upon surgical excision, the pathology indicated the lesion was consistent with molluscum contagiosum.
Molluscum contagiosum is a benign skin disorder caused by a pox virus and is frequently seen in children. This disease is transmitted primarily through direct skin contact with an infected individual.1 Contaminated fomites have been suggested as another source of infection.2 The typical lesion appears dome-shaped, round, and pinkish-purple in color.1 The incubation period ranges from 2 weeks to 6 months and is typically self-limited in immunocompetent hosts; however, in immunocompromised persons, molluscum contagiosum lesions may present atypically such that they are larger in size and/or resemble malignancies, such as basal cell carcinoma or keratoacanthoma (for single lesions), or other infectious diseases, such as cryptococcosis and histoplasmosis (for more numerous lesions).3,4 A giant atypical molluscum contagiosum is rarely seen in healthy individuals.
What’s on the differential?
The recent episode of bleeding raises concern for other neoplastic processes of the skin including squamous cell carcinoma or basal cell carcinoma as well as cutaneous metastatic rhabdoid tumor, given the patient’s history.
Eruptive keratoacanthomas are also reported in patients taking nivolumab, an anti-PD-1 immunotherapy, which the patient has received for treatment of his recurrent metastatic rhabdoid tumor.5 More common entities such as a pyogenic granuloma or verruca are also included on the differential. The initial presentation of the lesion, however, is more consistent with the pearly umbilicated papules associated with molluscum contagiosum.
Comments from Dr. Eichenfield
This is a very hard diagnosis to make with the clinical findings and history.
Molluscum contagiosum infections are common, but with this patient’s medical history, biopsy and excision with pathologic examination was an appropriate approach to make a certain diagnosis.
Ms. Moyal is a research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego.
References
1. Brown J et al. Int J Dermatol. 2006 Feb;45(2):93-9.
2. Hanson D and Diven DG. Dermatol Online J. 2003 Mar;9(2).
3. Badri T and Gandhi GR. Molluscum contagiosum. 2022. In: StatPearls [Internet]. Treasure Island, Fla.: StatPearls Publishing.
4. Schwartz JJ and Myskowski PL. J Am Acad Dermatol. 1992 Oct 1;27(4):583-8.
5. Antonov NK et al. JAAD Case Rep. 2019 Apr 5;5(4):342-5.
The correct answer is (D), molluscum contagiosum. Upon surgical excision, the pathology indicated the lesion was consistent with molluscum contagiosum.
Molluscum contagiosum is a benign skin disorder caused by a pox virus and is frequently seen in children. This disease is transmitted primarily through direct skin contact with an infected individual.1 Contaminated fomites have been suggested as another source of infection.2 The typical lesion appears dome-shaped, round, and pinkish-purple in color.1 The incubation period ranges from 2 weeks to 6 months and is typically self-limited in immunocompetent hosts; however, in immunocompromised persons, molluscum contagiosum lesions may present atypically such that they are larger in size and/or resemble malignancies, such as basal cell carcinoma or keratoacanthoma (for single lesions), or other infectious diseases, such as cryptococcosis and histoplasmosis (for more numerous lesions).3,4 A giant atypical molluscum contagiosum is rarely seen in healthy individuals.
What’s on the differential?
The recent episode of bleeding raises concern for other neoplastic processes of the skin including squamous cell carcinoma or basal cell carcinoma as well as cutaneous metastatic rhabdoid tumor, given the patient’s history.
Eruptive keratoacanthomas are also reported in patients taking nivolumab, an anti-PD-1 immunotherapy, which the patient has received for treatment of his recurrent metastatic rhabdoid tumor.5 More common entities such as a pyogenic granuloma or verruca are also included on the differential. The initial presentation of the lesion, however, is more consistent with the pearly umbilicated papules associated with molluscum contagiosum.
Comments from Dr. Eichenfield
This is a very hard diagnosis to make with the clinical findings and history.
Molluscum contagiosum infections are common, but with this patient’s medical history, biopsy and excision with pathologic examination was an appropriate approach to make a certain diagnosis.
Ms. Moyal is a research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital, San Diego.
References
1. Brown J et al. Int J Dermatol. 2006 Feb;45(2):93-9.
2. Hanson D and Diven DG. Dermatol Online J. 2003 Mar;9(2).
3. Badri T and Gandhi GR. Molluscum contagiosum. 2022. In: StatPearls [Internet]. Treasure Island, Fla.: StatPearls Publishing.
4. Schwartz JJ and Myskowski PL. J Am Acad Dermatol. 1992 Oct 1;27(4):583-8.
5. Antonov NK et al. JAAD Case Rep. 2019 Apr 5;5(4):342-5.
Breaking the itch-scratch cycle with mindfulness
Apple A. Bodemer, MD, a dermatologist at the University of Wisconsin, Madison, teaches patients how to breathe mindfully. So does Kathy Farah, MD, an integrative family physician who practices in Roberts, Wis.
, they said at the annual Integrative Dermatology Symposium.
“As with any integrative modality, if it’s safe and effective, then let’s use it,” Dr. Farah said in a presentation on the mind-body approach to pain and itch.
“A breathwork session can literally take 1 minute,” said Dr. Bodemer, associate professor of dermatology at the University of Wisconsin and director of an integrative dermatology clinic. Dr. Bodemer, who completed a fellowship in integrative medicine at the Andrew Weil Center for Integrative Medicine at the University of Arizona and sits on the American Board of Integrative Medicine, spoke on a mindfulness panel at the meeting.
Her favorite breathing practice is the “4-7-8” breath taught by Andrew Weil, MD, founder and director of the center. This involves inhaling through the nose for a count of 4, holding for 7, and exhaling through the mouth for a count of 8. “It doesn’t matter how slow or fast, it’s the tempo that matters ... On exhale, squeeze your abs in to engage your core and get air out of your lungs as much as you can,” she said, advising a cycle of three at a time.
A technique known as “square breathing” (breath in 4, hold for 4, breath out for 4, hold for 4) is another helpful technique to “reset the nervous system” said Dr. Farah, who worked for many years in a children’s hospital. With children, she said, “I often do five finger breathing.”
For five finger breathing, the children spread their fingers apart in front of them or on the ground and use the pointer finger of the opposite hand to trace each finger, inhaling while tracing upward, and exhaling while tracing down.
Dr. Farah, associate clinical director of The Center for Mind-Body Medicine in Washington, DC, said her commitment to mindfulness was influenced by a “seminal” study published over 20 years ago showing that patients with moderate to severe psoriasis who used a meditation-based, audiotape-guided stress reduction intervention during phototherapy sessions had more rapid resolution of psoriatic lesions than did patients who didn’t use the mindfulness exercise.
Among more recent findings: A cross-sectional study of 120 adult dermatology patients, published in the British Journal of Dermatology in 2016, assessed skin shame, social anxiety, anxiety, depression, dermatological quality of life, and levels of mindfulness, and found that higher levels of mindfulness were associated with lower levels of psychosocial distress.
Another cross-sectional questionnaire study looked at mindfulness and “itch catastrophizing” in 155 adult patients with atopic dermatitis. Higher levels of a specific facet of mindfulness termed “acting with awareness” were associated with lower levels of itch catastrophizing, the researchers found. “Catastrophizing is a negative way of thinking, this itching will never stop,” Dr. Farah explained. The study shows that “mindfulness can actually help reduce some of the automatic scratching and response to itch. So it’s a great adjunct to pharmaceuticals.”
Affirmations – phrases and statements that are repeated to oneself to help challenge negative thoughts – can also help reverse itch catastrophizing. Statements such as “I can breathe through this feeling of itching,” or “I can move to feel comfortable and relaxed” encourage positive change, she said.
“I teach [mindfulness skills like breathing] a lot, without any expectations. I’ll say ‘give it a try and see what you think.’ If patients feel even a micron better, then they’re invested” and can then find numerous tools online, Dr. Farah said. “Can I do this [in a busy schedule] with every patient? Absolutely not. But can I do it with every 10th patient? Maybe.”
Dr. Bodemer’s experience has shown her that “breathing with your patient builds rapport,” she said. “There’s something very powerful in that in terms of building trust. ... I’ll just do it [during a visit, to show them] and almost always, patients start breathing with me, with an invitation or without.”
For her own health, 4-7-8 breathing has “been a gateway to meditation and deeper practices,” she said. “But even without going very deep, it has a long history of being able to modulate the stress response. It’s the parasympathetic-sympathetic rebalancing I’m interested in.”
Mindful breathing and other mind-body practices also can be helpful for parents of children with eczema, she and Dr. Farah said.
Dr. Bodemer and Dr. Farah reported no financial relationships to disclose.
Apple A. Bodemer, MD, a dermatologist at the University of Wisconsin, Madison, teaches patients how to breathe mindfully. So does Kathy Farah, MD, an integrative family physician who practices in Roberts, Wis.
, they said at the annual Integrative Dermatology Symposium.
“As with any integrative modality, if it’s safe and effective, then let’s use it,” Dr. Farah said in a presentation on the mind-body approach to pain and itch.
“A breathwork session can literally take 1 minute,” said Dr. Bodemer, associate professor of dermatology at the University of Wisconsin and director of an integrative dermatology clinic. Dr. Bodemer, who completed a fellowship in integrative medicine at the Andrew Weil Center for Integrative Medicine at the University of Arizona and sits on the American Board of Integrative Medicine, spoke on a mindfulness panel at the meeting.
Her favorite breathing practice is the “4-7-8” breath taught by Andrew Weil, MD, founder and director of the center. This involves inhaling through the nose for a count of 4, holding for 7, and exhaling through the mouth for a count of 8. “It doesn’t matter how slow or fast, it’s the tempo that matters ... On exhale, squeeze your abs in to engage your core and get air out of your lungs as much as you can,” she said, advising a cycle of three at a time.
A technique known as “square breathing” (breath in 4, hold for 4, breath out for 4, hold for 4) is another helpful technique to “reset the nervous system” said Dr. Farah, who worked for many years in a children’s hospital. With children, she said, “I often do five finger breathing.”
For five finger breathing, the children spread their fingers apart in front of them or on the ground and use the pointer finger of the opposite hand to trace each finger, inhaling while tracing upward, and exhaling while tracing down.
Dr. Farah, associate clinical director of The Center for Mind-Body Medicine in Washington, DC, said her commitment to mindfulness was influenced by a “seminal” study published over 20 years ago showing that patients with moderate to severe psoriasis who used a meditation-based, audiotape-guided stress reduction intervention during phototherapy sessions had more rapid resolution of psoriatic lesions than did patients who didn’t use the mindfulness exercise.
Among more recent findings: A cross-sectional study of 120 adult dermatology patients, published in the British Journal of Dermatology in 2016, assessed skin shame, social anxiety, anxiety, depression, dermatological quality of life, and levels of mindfulness, and found that higher levels of mindfulness were associated with lower levels of psychosocial distress.
Another cross-sectional questionnaire study looked at mindfulness and “itch catastrophizing” in 155 adult patients with atopic dermatitis. Higher levels of a specific facet of mindfulness termed “acting with awareness” were associated with lower levels of itch catastrophizing, the researchers found. “Catastrophizing is a negative way of thinking, this itching will never stop,” Dr. Farah explained. The study shows that “mindfulness can actually help reduce some of the automatic scratching and response to itch. So it’s a great adjunct to pharmaceuticals.”
Affirmations – phrases and statements that are repeated to oneself to help challenge negative thoughts – can also help reverse itch catastrophizing. Statements such as “I can breathe through this feeling of itching,” or “I can move to feel comfortable and relaxed” encourage positive change, she said.
“I teach [mindfulness skills like breathing] a lot, without any expectations. I’ll say ‘give it a try and see what you think.’ If patients feel even a micron better, then they’re invested” and can then find numerous tools online, Dr. Farah said. “Can I do this [in a busy schedule] with every patient? Absolutely not. But can I do it with every 10th patient? Maybe.”
Dr. Bodemer’s experience has shown her that “breathing with your patient builds rapport,” she said. “There’s something very powerful in that in terms of building trust. ... I’ll just do it [during a visit, to show them] and almost always, patients start breathing with me, with an invitation or without.”
For her own health, 4-7-8 breathing has “been a gateway to meditation and deeper practices,” she said. “But even without going very deep, it has a long history of being able to modulate the stress response. It’s the parasympathetic-sympathetic rebalancing I’m interested in.”
Mindful breathing and other mind-body practices also can be helpful for parents of children with eczema, she and Dr. Farah said.
Dr. Bodemer and Dr. Farah reported no financial relationships to disclose.
Apple A. Bodemer, MD, a dermatologist at the University of Wisconsin, Madison, teaches patients how to breathe mindfully. So does Kathy Farah, MD, an integrative family physician who practices in Roberts, Wis.
, they said at the annual Integrative Dermatology Symposium.
“As with any integrative modality, if it’s safe and effective, then let’s use it,” Dr. Farah said in a presentation on the mind-body approach to pain and itch.
“A breathwork session can literally take 1 minute,” said Dr. Bodemer, associate professor of dermatology at the University of Wisconsin and director of an integrative dermatology clinic. Dr. Bodemer, who completed a fellowship in integrative medicine at the Andrew Weil Center for Integrative Medicine at the University of Arizona and sits on the American Board of Integrative Medicine, spoke on a mindfulness panel at the meeting.
Her favorite breathing practice is the “4-7-8” breath taught by Andrew Weil, MD, founder and director of the center. This involves inhaling through the nose for a count of 4, holding for 7, and exhaling through the mouth for a count of 8. “It doesn’t matter how slow or fast, it’s the tempo that matters ... On exhale, squeeze your abs in to engage your core and get air out of your lungs as much as you can,” she said, advising a cycle of three at a time.
A technique known as “square breathing” (breath in 4, hold for 4, breath out for 4, hold for 4) is another helpful technique to “reset the nervous system” said Dr. Farah, who worked for many years in a children’s hospital. With children, she said, “I often do five finger breathing.”
For five finger breathing, the children spread their fingers apart in front of them or on the ground and use the pointer finger of the opposite hand to trace each finger, inhaling while tracing upward, and exhaling while tracing down.
Dr. Farah, associate clinical director of The Center for Mind-Body Medicine in Washington, DC, said her commitment to mindfulness was influenced by a “seminal” study published over 20 years ago showing that patients with moderate to severe psoriasis who used a meditation-based, audiotape-guided stress reduction intervention during phototherapy sessions had more rapid resolution of psoriatic lesions than did patients who didn’t use the mindfulness exercise.
Among more recent findings: A cross-sectional study of 120 adult dermatology patients, published in the British Journal of Dermatology in 2016, assessed skin shame, social anxiety, anxiety, depression, dermatological quality of life, and levels of mindfulness, and found that higher levels of mindfulness were associated with lower levels of psychosocial distress.
Another cross-sectional questionnaire study looked at mindfulness and “itch catastrophizing” in 155 adult patients with atopic dermatitis. Higher levels of a specific facet of mindfulness termed “acting with awareness” were associated with lower levels of itch catastrophizing, the researchers found. “Catastrophizing is a negative way of thinking, this itching will never stop,” Dr. Farah explained. The study shows that “mindfulness can actually help reduce some of the automatic scratching and response to itch. So it’s a great adjunct to pharmaceuticals.”
Affirmations – phrases and statements that are repeated to oneself to help challenge negative thoughts – can also help reverse itch catastrophizing. Statements such as “I can breathe through this feeling of itching,” or “I can move to feel comfortable and relaxed” encourage positive change, she said.
“I teach [mindfulness skills like breathing] a lot, without any expectations. I’ll say ‘give it a try and see what you think.’ If patients feel even a micron better, then they’re invested” and can then find numerous tools online, Dr. Farah said. “Can I do this [in a busy schedule] with every patient? Absolutely not. But can I do it with every 10th patient? Maybe.”
Dr. Bodemer’s experience has shown her that “breathing with your patient builds rapport,” she said. “There’s something very powerful in that in terms of building trust. ... I’ll just do it [during a visit, to show them] and almost always, patients start breathing with me, with an invitation or without.”
For her own health, 4-7-8 breathing has “been a gateway to meditation and deeper practices,” she said. “But even without going very deep, it has a long history of being able to modulate the stress response. It’s the parasympathetic-sympathetic rebalancing I’m interested in.”
Mindful breathing and other mind-body practices also can be helpful for parents of children with eczema, she and Dr. Farah said.
Dr. Bodemer and Dr. Farah reported no financial relationships to disclose.
FROM IDS 2022
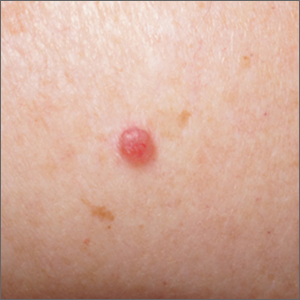
Pink shoulder lesion
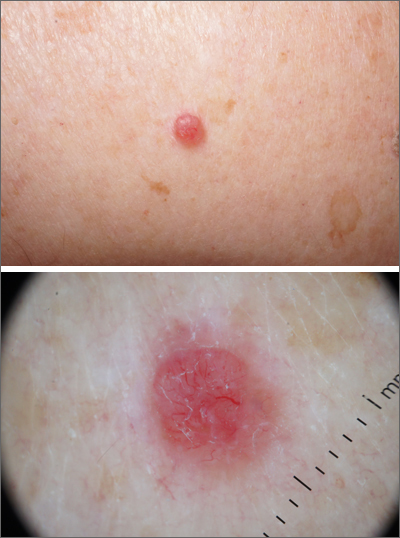
A scoop shave biopsy was performed and histology was consistent with a nodular basal cell carcinoma. BCC is the most common skin cancer in the United States, occurring in approximately 30% of patients with skin types I and II.1 In patients who are Black, squamous cell carcinoma is more common than BCC.2 The overall incidence of BCC is increasing by 4% to 8% every year in the United States.1
BCC most often affects sun-damaged areas—especially on the head and neck—and frequently causes significant tissue damage. It is, however, associated with a low risk of metastasis and mortality.
BCCs may appear as a pink, brown, blue, or white papule or macule. The surface is frequently shiny or pearly in appearance with a rolled border. Dilated, angulated, tree-branch like vessels termed “arborizing vessels” are common. Infiltrative BCC subtypes may look like melted candlewax and extend beyond the area that is clinically apparent.
Partial shave biopsies of a lesion can confirm the diagnosis. A punch biopsy can make it easier to evaluate flat (or even sunken) lesions.
The patient described here was treated with electrodessication and curettage (EDC)—a fast, economical, and effective treatment for the low-risk subtypes of superficial or nodular BCCs on the trunk or extremities. EDC should be avoided with higher risk subtypes of micronodular and infiltrative BCC. With these subtypes, excision (with 4- to 6-mm margins) or Mohs microsurgery is recommended.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME. References
1. Kim DP, Kus KJB, Ruiz E. Basal cell carcinoma review. Hematol Oncol Clin North Am. 2019;33:13-24. doi:10.1016/j.hoc.2018.09.004
2. Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206.

A scoop shave biopsy was performed and histology was consistent with a nodular basal cell carcinoma. BCC is the most common skin cancer in the United States, occurring in approximately 30% of patients with skin types I and II.1 In patients who are Black, squamous cell carcinoma is more common than BCC.2 The overall incidence of BCC is increasing by 4% to 8% every year in the United States.1
BCC most often affects sun-damaged areas—especially on the head and neck—and frequently causes significant tissue damage. It is, however, associated with a low risk of metastasis and mortality.
BCCs may appear as a pink, brown, blue, or white papule or macule. The surface is frequently shiny or pearly in appearance with a rolled border. Dilated, angulated, tree-branch like vessels termed “arborizing vessels” are common. Infiltrative BCC subtypes may look like melted candlewax and extend beyond the area that is clinically apparent.
Partial shave biopsies of a lesion can confirm the diagnosis. A punch biopsy can make it easier to evaluate flat (or even sunken) lesions.
The patient described here was treated with electrodessication and curettage (EDC)—a fast, economical, and effective treatment for the low-risk subtypes of superficial or nodular BCCs on the trunk or extremities. EDC should be avoided with higher risk subtypes of micronodular and infiltrative BCC. With these subtypes, excision (with 4- to 6-mm margins) or Mohs microsurgery is recommended.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME. References

A scoop shave biopsy was performed and histology was consistent with a nodular basal cell carcinoma. BCC is the most common skin cancer in the United States, occurring in approximately 30% of patients with skin types I and II.1 In patients who are Black, squamous cell carcinoma is more common than BCC.2 The overall incidence of BCC is increasing by 4% to 8% every year in the United States.1
BCC most often affects sun-damaged areas—especially on the head and neck—and frequently causes significant tissue damage. It is, however, associated with a low risk of metastasis and mortality.
BCCs may appear as a pink, brown, blue, or white papule or macule. The surface is frequently shiny or pearly in appearance with a rolled border. Dilated, angulated, tree-branch like vessels termed “arborizing vessels” are common. Infiltrative BCC subtypes may look like melted candlewax and extend beyond the area that is clinically apparent.
Partial shave biopsies of a lesion can confirm the diagnosis. A punch biopsy can make it easier to evaluate flat (or even sunken) lesions.
The patient described here was treated with electrodessication and curettage (EDC)—a fast, economical, and effective treatment for the low-risk subtypes of superficial or nodular BCCs on the trunk or extremities. EDC should be avoided with higher risk subtypes of micronodular and infiltrative BCC. With these subtypes, excision (with 4- to 6-mm margins) or Mohs microsurgery is recommended.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME. References
1. Kim DP, Kus KJB, Ruiz E. Basal cell carcinoma review. Hematol Oncol Clin North Am. 2019;33:13-24. doi:10.1016/j.hoc.2018.09.004
2. Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206.
1. Kim DP, Kus KJB, Ruiz E. Basal cell carcinoma review. Hematol Oncol Clin North Am. 2019;33:13-24. doi:10.1016/j.hoc.2018.09.004
2. Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206.
Study finds high rate of psychiatric burden in cosmetic dermatology patients
results from a large retrospective analysis showed.
“As the rate of cosmetic procedures continues to increase, it is crucial that physicians understand that many patients with a psychiatric disorder require clear communication and appropriate consultation visits,” lead study author Patricia Richey, MD, told this news organization.
While studies have displayed links between the desire for a cosmetic procedure and psychiatric stressors and disorders – most commonly mood disorders, personality disorders, body dysmorphic disorder, and addiction-like behavior – the scarce literature on the subject mostly comes from the realm of plastic surgery.
“The relationship between psychiatric disease and the motivation for dermatologic cosmetic procedures has never been fully elucidated,” said Dr. Richey, who practices Mohs surgery and cosmetic dermatology in Washington, D.C., and conducts research for the Wellman Center for Photomedicine and the Dermatology Laser and Cosmetic Center at Massachusetts General Hospital, Boston. “A possible association between psychiatric disorder and the motivation for cosmetic procedures is critical to understand given increasing procedure rates and the need for clear communication and appropriate consultation visits with these patients.”
For the retrospective cohort study, which was published online in the Journal of the American Academy of Dermatology, Dr. Richey; Mathew Avram, MD, JD, director of the Dermatology Laser and Cosmetic Center at MGH; and Ryan W. Chapin, PharmD, of Beth Israel Deaconess Medical Center, Boston, reviewed the medical records of 1,000 patients from a cosmetic dermatology clinic and 1,000 patients from a medical dermatology clinic, both at MGH. Those who crossed over between the two clinics were excluded from the analysis.
Patients in the cosmetic group were significantly younger than those in the medical group (a mean of 48 vs. 56 years, respectively; P < .0001), and there was a higher percentage of women than men in both groups (78.5% vs. 21.5% in the cosmetic group and 61.4% vs. 38.6% in the medical group; P < .00001).
The researchers found that 49% of patients in the cosmetic group had been diagnosed with at least one psychiatric disorder, compared with 33% in the medical group (P < .00001), most commonly anxiety, depression, ADHD, and insomnia. In addition, 39 patients in the cosmetic group had 2 or more psychiatric disorders, compared with 22 of those in the medical group.
Similarly, 44% of patients in the cosmetic group were on a psychiatric medication, compared with 28% in the medical group (P < .00001). The average number of medications among those on more than one psychiatric medication was 1.67 among those in the cosmetic dermatology group versus 1.48 among those in the medical dermatology group (P = .020).
By drug class, a higher percentage of patients in the cosmetic group, compared with those in the medical group, were taking antidepressants (33% vs. 21%, respectively; P < .00001), anxiolytics (26% vs. 13%; P < .00001), mood stabilizers (2.80% vs. 1.10%; P = .006), and stimulants (15.2% vs. 7.20%; P < .00001). The proportion of those taking antipsychotics was essentially even in the two groups (2.50% vs. 2.70%; P = .779).
Dr. Richey and colleagues also observed that patients in the cosmetic group had significantly higher rates of obsessive-compulsive disorder (OCD) and ADHD than those in the medical group. “This finding did not particularly surprise me,” she said, since she and her colleagues recently published a study on the association of stimulant use with psychocutaneous disease.
“Stimulants are used to treat ADHD and are also known to trigger OCD-like symptoms,” she said. “I was surprised that no patients had been diagnosed with body dysmorphic disorder, but we know that with increased patient access to medical records, physicians are often cautious in their documentation.”
She added that the overall results of the new study underscore the importance of consultation visits with cosmetic patients, including obtaining a full medication list and accurate medical history, if possible. “One could also consider well-studied screening tools mostly from the mood disorder realm, such as the Patient Health Questionnaire–2,” Dr. Richey said. “Much can be gained from simply talking to the patient and trying to understand him/her and underlying motivations prior to performing a procedure.”
Evan Rieder, MD, a New York City–based dermatologist and psychiatrist who was asked to comment on the study, characterized the analysis as demonstrating what medical and cosmetic dermatologists have been seeing in their practices for years. “While this study is limited by its single-center retrospective nature in an academic center that may not be representative of the general population, it does demonstrate a high burden of psychopathology and psychopharmacologic treatments in aesthetic patients,” Dr. Rieder said in an interview.
“While psychiatric illness is not a contraindication to cosmetic treatment, a high percentage of patients with ADHD, OCD, and likely [body dysmorphic disorder] in cosmetic dermatology practices should give us pause.” The nature of these diseases may indicate that some people are seeking aesthetic treatments for reasons yet to be elucidated, he added.
“It certainly indicates that dermatologists should be equipped to screen for, identify, and provide such patients with the appropriate resources for psychological treatment, regardless if they are deemed appropriate candidates for cosmetic intervention,” he said.
In an interview, Pooja Sodha, MD, director of the Center for Laser and Cosmetic Dermatology at George Washington University, Washington, noted that previous studies have demonstrated the interplay between mood disorders and dermatologic conditions for years, namely in acne, atopic dermatitis, psoriasis, and immune mediated disorders.
“In these conditions, the psychiatric stressors can worsen the skin condition and impede treatment,” Dr. Sodha said. “This study is an important segue into further elucidating our cosmetic patient population, and we should try to ask the next important question: how do we as physicians build a better rapport with these patients, understand their motivations for care, and effectively guide the patient through the consultation process to realistically address their concerns? It might help us both.”
Neither the researchers nor Dr. Sodha reported having financial disclosures. Dr. Rieder disclosed that he is a consultant for Allergan, Almirall, Bristol-Myers Squibb, Dr. Brandt, L’Oreal, Procter & Gamble, and Unilever.
results from a large retrospective analysis showed.
“As the rate of cosmetic procedures continues to increase, it is crucial that physicians understand that many patients with a psychiatric disorder require clear communication and appropriate consultation visits,” lead study author Patricia Richey, MD, told this news organization.
While studies have displayed links between the desire for a cosmetic procedure and psychiatric stressors and disorders – most commonly mood disorders, personality disorders, body dysmorphic disorder, and addiction-like behavior – the scarce literature on the subject mostly comes from the realm of plastic surgery.
“The relationship between psychiatric disease and the motivation for dermatologic cosmetic procedures has never been fully elucidated,” said Dr. Richey, who practices Mohs surgery and cosmetic dermatology in Washington, D.C., and conducts research for the Wellman Center for Photomedicine and the Dermatology Laser and Cosmetic Center at Massachusetts General Hospital, Boston. “A possible association between psychiatric disorder and the motivation for cosmetic procedures is critical to understand given increasing procedure rates and the need for clear communication and appropriate consultation visits with these patients.”
For the retrospective cohort study, which was published online in the Journal of the American Academy of Dermatology, Dr. Richey; Mathew Avram, MD, JD, director of the Dermatology Laser and Cosmetic Center at MGH; and Ryan W. Chapin, PharmD, of Beth Israel Deaconess Medical Center, Boston, reviewed the medical records of 1,000 patients from a cosmetic dermatology clinic and 1,000 patients from a medical dermatology clinic, both at MGH. Those who crossed over between the two clinics were excluded from the analysis.
Patients in the cosmetic group were significantly younger than those in the medical group (a mean of 48 vs. 56 years, respectively; P < .0001), and there was a higher percentage of women than men in both groups (78.5% vs. 21.5% in the cosmetic group and 61.4% vs. 38.6% in the medical group; P < .00001).
The researchers found that 49% of patients in the cosmetic group had been diagnosed with at least one psychiatric disorder, compared with 33% in the medical group (P < .00001), most commonly anxiety, depression, ADHD, and insomnia. In addition, 39 patients in the cosmetic group had 2 or more psychiatric disorders, compared with 22 of those in the medical group.
Similarly, 44% of patients in the cosmetic group were on a psychiatric medication, compared with 28% in the medical group (P < .00001). The average number of medications among those on more than one psychiatric medication was 1.67 among those in the cosmetic dermatology group versus 1.48 among those in the medical dermatology group (P = .020).
By drug class, a higher percentage of patients in the cosmetic group, compared with those in the medical group, were taking antidepressants (33% vs. 21%, respectively; P < .00001), anxiolytics (26% vs. 13%; P < .00001), mood stabilizers (2.80% vs. 1.10%; P = .006), and stimulants (15.2% vs. 7.20%; P < .00001). The proportion of those taking antipsychotics was essentially even in the two groups (2.50% vs. 2.70%; P = .779).
Dr. Richey and colleagues also observed that patients in the cosmetic group had significantly higher rates of obsessive-compulsive disorder (OCD) and ADHD than those in the medical group. “This finding did not particularly surprise me,” she said, since she and her colleagues recently published a study on the association of stimulant use with psychocutaneous disease.
“Stimulants are used to treat ADHD and are also known to trigger OCD-like symptoms,” she said. “I was surprised that no patients had been diagnosed with body dysmorphic disorder, but we know that with increased patient access to medical records, physicians are often cautious in their documentation.”
She added that the overall results of the new study underscore the importance of consultation visits with cosmetic patients, including obtaining a full medication list and accurate medical history, if possible. “One could also consider well-studied screening tools mostly from the mood disorder realm, such as the Patient Health Questionnaire–2,” Dr. Richey said. “Much can be gained from simply talking to the patient and trying to understand him/her and underlying motivations prior to performing a procedure.”
Evan Rieder, MD, a New York City–based dermatologist and psychiatrist who was asked to comment on the study, characterized the analysis as demonstrating what medical and cosmetic dermatologists have been seeing in their practices for years. “While this study is limited by its single-center retrospective nature in an academic center that may not be representative of the general population, it does demonstrate a high burden of psychopathology and psychopharmacologic treatments in aesthetic patients,” Dr. Rieder said in an interview.
“While psychiatric illness is not a contraindication to cosmetic treatment, a high percentage of patients with ADHD, OCD, and likely [body dysmorphic disorder] in cosmetic dermatology practices should give us pause.” The nature of these diseases may indicate that some people are seeking aesthetic treatments for reasons yet to be elucidated, he added.
“It certainly indicates that dermatologists should be equipped to screen for, identify, and provide such patients with the appropriate resources for psychological treatment, regardless if they are deemed appropriate candidates for cosmetic intervention,” he said.
In an interview, Pooja Sodha, MD, director of the Center for Laser and Cosmetic Dermatology at George Washington University, Washington, noted that previous studies have demonstrated the interplay between mood disorders and dermatologic conditions for years, namely in acne, atopic dermatitis, psoriasis, and immune mediated disorders.
“In these conditions, the psychiatric stressors can worsen the skin condition and impede treatment,” Dr. Sodha said. “This study is an important segue into further elucidating our cosmetic patient population, and we should try to ask the next important question: how do we as physicians build a better rapport with these patients, understand their motivations for care, and effectively guide the patient through the consultation process to realistically address their concerns? It might help us both.”
Neither the researchers nor Dr. Sodha reported having financial disclosures. Dr. Rieder disclosed that he is a consultant for Allergan, Almirall, Bristol-Myers Squibb, Dr. Brandt, L’Oreal, Procter & Gamble, and Unilever.
results from a large retrospective analysis showed.
“As the rate of cosmetic procedures continues to increase, it is crucial that physicians understand that many patients with a psychiatric disorder require clear communication and appropriate consultation visits,” lead study author Patricia Richey, MD, told this news organization.
While studies have displayed links between the desire for a cosmetic procedure and psychiatric stressors and disorders – most commonly mood disorders, personality disorders, body dysmorphic disorder, and addiction-like behavior – the scarce literature on the subject mostly comes from the realm of plastic surgery.
“The relationship between psychiatric disease and the motivation for dermatologic cosmetic procedures has never been fully elucidated,” said Dr. Richey, who practices Mohs surgery and cosmetic dermatology in Washington, D.C., and conducts research for the Wellman Center for Photomedicine and the Dermatology Laser and Cosmetic Center at Massachusetts General Hospital, Boston. “A possible association between psychiatric disorder and the motivation for cosmetic procedures is critical to understand given increasing procedure rates and the need for clear communication and appropriate consultation visits with these patients.”
For the retrospective cohort study, which was published online in the Journal of the American Academy of Dermatology, Dr. Richey; Mathew Avram, MD, JD, director of the Dermatology Laser and Cosmetic Center at MGH; and Ryan W. Chapin, PharmD, of Beth Israel Deaconess Medical Center, Boston, reviewed the medical records of 1,000 patients from a cosmetic dermatology clinic and 1,000 patients from a medical dermatology clinic, both at MGH. Those who crossed over between the two clinics were excluded from the analysis.
Patients in the cosmetic group were significantly younger than those in the medical group (a mean of 48 vs. 56 years, respectively; P < .0001), and there was a higher percentage of women than men in both groups (78.5% vs. 21.5% in the cosmetic group and 61.4% vs. 38.6% in the medical group; P < .00001).
The researchers found that 49% of patients in the cosmetic group had been diagnosed with at least one psychiatric disorder, compared with 33% in the medical group (P < .00001), most commonly anxiety, depression, ADHD, and insomnia. In addition, 39 patients in the cosmetic group had 2 or more psychiatric disorders, compared with 22 of those in the medical group.
Similarly, 44% of patients in the cosmetic group were on a psychiatric medication, compared with 28% in the medical group (P < .00001). The average number of medications among those on more than one psychiatric medication was 1.67 among those in the cosmetic dermatology group versus 1.48 among those in the medical dermatology group (P = .020).
By drug class, a higher percentage of patients in the cosmetic group, compared with those in the medical group, were taking antidepressants (33% vs. 21%, respectively; P < .00001), anxiolytics (26% vs. 13%; P < .00001), mood stabilizers (2.80% vs. 1.10%; P = .006), and stimulants (15.2% vs. 7.20%; P < .00001). The proportion of those taking antipsychotics was essentially even in the two groups (2.50% vs. 2.70%; P = .779).
Dr. Richey and colleagues also observed that patients in the cosmetic group had significantly higher rates of obsessive-compulsive disorder (OCD) and ADHD than those in the medical group. “This finding did not particularly surprise me,” she said, since she and her colleagues recently published a study on the association of stimulant use with psychocutaneous disease.
“Stimulants are used to treat ADHD and are also known to trigger OCD-like symptoms,” she said. “I was surprised that no patients had been diagnosed with body dysmorphic disorder, but we know that with increased patient access to medical records, physicians are often cautious in their documentation.”
She added that the overall results of the new study underscore the importance of consultation visits with cosmetic patients, including obtaining a full medication list and accurate medical history, if possible. “One could also consider well-studied screening tools mostly from the mood disorder realm, such as the Patient Health Questionnaire–2,” Dr. Richey said. “Much can be gained from simply talking to the patient and trying to understand him/her and underlying motivations prior to performing a procedure.”
Evan Rieder, MD, a New York City–based dermatologist and psychiatrist who was asked to comment on the study, characterized the analysis as demonstrating what medical and cosmetic dermatologists have been seeing in their practices for years. “While this study is limited by its single-center retrospective nature in an academic center that may not be representative of the general population, it does demonstrate a high burden of psychopathology and psychopharmacologic treatments in aesthetic patients,” Dr. Rieder said in an interview.
“While psychiatric illness is not a contraindication to cosmetic treatment, a high percentage of patients with ADHD, OCD, and likely [body dysmorphic disorder] in cosmetic dermatology practices should give us pause.” The nature of these diseases may indicate that some people are seeking aesthetic treatments for reasons yet to be elucidated, he added.
“It certainly indicates that dermatologists should be equipped to screen for, identify, and provide such patients with the appropriate resources for psychological treatment, regardless if they are deemed appropriate candidates for cosmetic intervention,” he said.
In an interview, Pooja Sodha, MD, director of the Center for Laser and Cosmetic Dermatology at George Washington University, Washington, noted that previous studies have demonstrated the interplay between mood disorders and dermatologic conditions for years, namely in acne, atopic dermatitis, psoriasis, and immune mediated disorders.
“In these conditions, the psychiatric stressors can worsen the skin condition and impede treatment,” Dr. Sodha said. “This study is an important segue into further elucidating our cosmetic patient population, and we should try to ask the next important question: how do we as physicians build a better rapport with these patients, understand their motivations for care, and effectively guide the patient through the consultation process to realistically address their concerns? It might help us both.”
Neither the researchers nor Dr. Sodha reported having financial disclosures. Dr. Rieder disclosed that he is a consultant for Allergan, Almirall, Bristol-Myers Squibb, Dr. Brandt, L’Oreal, Procter & Gamble, and Unilever.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Medicaid coverage of HPV vaccine in adults: Implications in dermatology
, according to the authors of a review of Medicaid policies across all 50 states.
The human papillomavirus (HPV) vaccine is approved for people aged 9-45 years, for preventing genital, cervical, anal, and oropharyngeal cancers, and genital warts. And the Centers for Disease Control and Prevention Advisory Committee on Immunization Practices recommends routine vaccination with the HPV vaccine for individuals aged 9-26 years, with “shared clinical decision-making” recommended for vaccination of those aged 27-45 years, wrote Nathaniel Goldman of New York Medical College, Valhalla, and coauthors, from the University of Missouri–Kansas City and Harvard Medical School, Boston.
A total of 33 states offered formal statewide Medicaid coverage policies that were accessible online or through the state’s Medicaid office. Another 11 states provided coverage through Medicaid managed care organizations, and 4 states had HPV vaccination as part of their formal Medicaid adult vaccination programs.
Overall, 43 states covered HPV vaccination through age 45 years with no need for prior authorization, and another 4 states (Ohio, Maine, Nebraska, and New York) provided coverage with prior authorization for adults older than 26 years.
The study findings were limited by the use of Medicaid coverage only, the researchers noted. Consequently, patients eligible for HPV vaccination who are uninsured or have other types of insurance may face additional barriers in the form of high costs, given that the current retail price is $250-$350 per shot for the three-shot series, the researchers noted.
However, the results suggest that Medicaid coverage for HPV vaccination may inform dermatologists’ recommendations for patients at increased risk, they said. More research is needed to “better identify dermatology patients at risk for new HPV infection and ways to improve vaccination rates in these vulnerable individuals,” they added.
Vaccine discussions are important in dermatology
“Dermatologists care for patients who may be an increased risk of vaccine-preventable illnesses, either from a skin disease or a dermatology medication,” corresponding author Megan H. Noe, MD, a dermatologist at Brigham and Women’s Hospital, and assistant professor of dermatology, Harvard Medical School, Boston, said in an interview. “Over the last several years, we have seen that all physicians, whether they provide vaccinations or not, can play an important role in discussing vaccines with their patients,” she said.
“Vaccines can be cost-prohibitive for patients without insurance coverage, so we hope that dermatologists will be more likely to recommend the HPV vaccine to patients 27-45 years of age if they know that it is likely covered by insurance,” Dr. Noe noted.
However, “time may be a barrier for many dermatologists who have many important things to discuss with patients during their appointments,” she said. “We are currently working on developing educational information to help facilitate this conversation,” she added.
Looking ahead, she said that “additional research is necessary to create vaccine guidelines specific to dermatology patients and dermatology medications, so we can provide clear recommendations to our patients and ensure appropriate insurance coverage for all necessary vaccines.”
Vaccine discussions
“I think it’s great that many Medicaid plans are covering HPV vaccination,” said Karl Saardi, MD, of the department of dermatology, George Washington University, Washington, who was asked to comment on the study. “I routinely recommend [vaccination] for patients who have viral warts, since it does lead to improvement in some cases,” Dr. Saardi, who was not involved in the current study, said in an interview. “Although we don’t have the HPV vaccines in our clinic for administration, my experience has been that patients are very open to discussing it with their primary care doctors.”
Although the upper age range continues to rise, “I think getting younger people vaccinated will also prove to be important,” said Dr. Saardi, director of the inpatient dermatology service at the George Washington University Hospital.
The point made in the current study about the importance of HPV vaccination in patients with hidradenitis suppurativa is also crucial, he added. “Since chronic skin inflammation in hidradenitis drives squamous cell carcinoma, reducing the impact of HPV on such cancers makes perfect sense.”
The study received no outside funding. Dr. Noe disclosed grants from Boehringer Ingelheim unrelated to the current study. Dr. Saardi had no financial conflicts to disclose.
, according to the authors of a review of Medicaid policies across all 50 states.
The human papillomavirus (HPV) vaccine is approved for people aged 9-45 years, for preventing genital, cervical, anal, and oropharyngeal cancers, and genital warts. And the Centers for Disease Control and Prevention Advisory Committee on Immunization Practices recommends routine vaccination with the HPV vaccine for individuals aged 9-26 years, with “shared clinical decision-making” recommended for vaccination of those aged 27-45 years, wrote Nathaniel Goldman of New York Medical College, Valhalla, and coauthors, from the University of Missouri–Kansas City and Harvard Medical School, Boston.
A total of 33 states offered formal statewide Medicaid coverage policies that were accessible online or through the state’s Medicaid office. Another 11 states provided coverage through Medicaid managed care organizations, and 4 states had HPV vaccination as part of their formal Medicaid adult vaccination programs.
Overall, 43 states covered HPV vaccination through age 45 years with no need for prior authorization, and another 4 states (Ohio, Maine, Nebraska, and New York) provided coverage with prior authorization for adults older than 26 years.
The study findings were limited by the use of Medicaid coverage only, the researchers noted. Consequently, patients eligible for HPV vaccination who are uninsured or have other types of insurance may face additional barriers in the form of high costs, given that the current retail price is $250-$350 per shot for the three-shot series, the researchers noted.
However, the results suggest that Medicaid coverage for HPV vaccination may inform dermatologists’ recommendations for patients at increased risk, they said. More research is needed to “better identify dermatology patients at risk for new HPV infection and ways to improve vaccination rates in these vulnerable individuals,” they added.
Vaccine discussions are important in dermatology
“Dermatologists care for patients who may be an increased risk of vaccine-preventable illnesses, either from a skin disease or a dermatology medication,” corresponding author Megan H. Noe, MD, a dermatologist at Brigham and Women’s Hospital, and assistant professor of dermatology, Harvard Medical School, Boston, said in an interview. “Over the last several years, we have seen that all physicians, whether they provide vaccinations or not, can play an important role in discussing vaccines with their patients,” she said.
“Vaccines can be cost-prohibitive for patients without insurance coverage, so we hope that dermatologists will be more likely to recommend the HPV vaccine to patients 27-45 years of age if they know that it is likely covered by insurance,” Dr. Noe noted.
However, “time may be a barrier for many dermatologists who have many important things to discuss with patients during their appointments,” she said. “We are currently working on developing educational information to help facilitate this conversation,” she added.
Looking ahead, she said that “additional research is necessary to create vaccine guidelines specific to dermatology patients and dermatology medications, so we can provide clear recommendations to our patients and ensure appropriate insurance coverage for all necessary vaccines.”
Vaccine discussions
“I think it’s great that many Medicaid plans are covering HPV vaccination,” said Karl Saardi, MD, of the department of dermatology, George Washington University, Washington, who was asked to comment on the study. “I routinely recommend [vaccination] for patients who have viral warts, since it does lead to improvement in some cases,” Dr. Saardi, who was not involved in the current study, said in an interview. “Although we don’t have the HPV vaccines in our clinic for administration, my experience has been that patients are very open to discussing it with their primary care doctors.”
Although the upper age range continues to rise, “I think getting younger people vaccinated will also prove to be important,” said Dr. Saardi, director of the inpatient dermatology service at the George Washington University Hospital.
The point made in the current study about the importance of HPV vaccination in patients with hidradenitis suppurativa is also crucial, he added. “Since chronic skin inflammation in hidradenitis drives squamous cell carcinoma, reducing the impact of HPV on such cancers makes perfect sense.”
The study received no outside funding. Dr. Noe disclosed grants from Boehringer Ingelheim unrelated to the current study. Dr. Saardi had no financial conflicts to disclose.
, according to the authors of a review of Medicaid policies across all 50 states.
The human papillomavirus (HPV) vaccine is approved for people aged 9-45 years, for preventing genital, cervical, anal, and oropharyngeal cancers, and genital warts. And the Centers for Disease Control and Prevention Advisory Committee on Immunization Practices recommends routine vaccination with the HPV vaccine for individuals aged 9-26 years, with “shared clinical decision-making” recommended for vaccination of those aged 27-45 years, wrote Nathaniel Goldman of New York Medical College, Valhalla, and coauthors, from the University of Missouri–Kansas City and Harvard Medical School, Boston.
A total of 33 states offered formal statewide Medicaid coverage policies that were accessible online or through the state’s Medicaid office. Another 11 states provided coverage through Medicaid managed care organizations, and 4 states had HPV vaccination as part of their formal Medicaid adult vaccination programs.
Overall, 43 states covered HPV vaccination through age 45 years with no need for prior authorization, and another 4 states (Ohio, Maine, Nebraska, and New York) provided coverage with prior authorization for adults older than 26 years.
The study findings were limited by the use of Medicaid coverage only, the researchers noted. Consequently, patients eligible for HPV vaccination who are uninsured or have other types of insurance may face additional barriers in the form of high costs, given that the current retail price is $250-$350 per shot for the three-shot series, the researchers noted.
However, the results suggest that Medicaid coverage for HPV vaccination may inform dermatologists’ recommendations for patients at increased risk, they said. More research is needed to “better identify dermatology patients at risk for new HPV infection and ways to improve vaccination rates in these vulnerable individuals,” they added.
Vaccine discussions are important in dermatology
“Dermatologists care for patients who may be an increased risk of vaccine-preventable illnesses, either from a skin disease or a dermatology medication,” corresponding author Megan H. Noe, MD, a dermatologist at Brigham and Women’s Hospital, and assistant professor of dermatology, Harvard Medical School, Boston, said in an interview. “Over the last several years, we have seen that all physicians, whether they provide vaccinations or not, can play an important role in discussing vaccines with their patients,” she said.
“Vaccines can be cost-prohibitive for patients without insurance coverage, so we hope that dermatologists will be more likely to recommend the HPV vaccine to patients 27-45 years of age if they know that it is likely covered by insurance,” Dr. Noe noted.
However, “time may be a barrier for many dermatologists who have many important things to discuss with patients during their appointments,” she said. “We are currently working on developing educational information to help facilitate this conversation,” she added.
Looking ahead, she said that “additional research is necessary to create vaccine guidelines specific to dermatology patients and dermatology medications, so we can provide clear recommendations to our patients and ensure appropriate insurance coverage for all necessary vaccines.”
Vaccine discussions
“I think it’s great that many Medicaid plans are covering HPV vaccination,” said Karl Saardi, MD, of the department of dermatology, George Washington University, Washington, who was asked to comment on the study. “I routinely recommend [vaccination] for patients who have viral warts, since it does lead to improvement in some cases,” Dr. Saardi, who was not involved in the current study, said in an interview. “Although we don’t have the HPV vaccines in our clinic for administration, my experience has been that patients are very open to discussing it with their primary care doctors.”
Although the upper age range continues to rise, “I think getting younger people vaccinated will also prove to be important,” said Dr. Saardi, director of the inpatient dermatology service at the George Washington University Hospital.
The point made in the current study about the importance of HPV vaccination in patients with hidradenitis suppurativa is also crucial, he added. “Since chronic skin inflammation in hidradenitis drives squamous cell carcinoma, reducing the impact of HPV on such cancers makes perfect sense.”
The study received no outside funding. Dr. Noe disclosed grants from Boehringer Ingelheim unrelated to the current study. Dr. Saardi had no financial conflicts to disclose.
FROM JAMA DERMATOLOGY
Lego introduces first character with vitiligo
The
The character appears with the customizable array of players to assemble for a table football team.
It’s the latest representation of the disease as toymakers diversify their lines.
In May 2022, Mattel released a Ken doll with vitiligo after a Barbie with vitiligo was released in 2020. Rainbow High and other toy makers also have character versions.
The Lego addition follows a big summer medically for vitiligo as the first treatment was approved for repigmentation. In July, a cream formulation of ruxolitinib (Opzelura), a Janus kinase inhibitor, became the first repigmentation treatment approved by the Food and Drug Administration for nonsegmental vitiligo, the most common form of the disease.
Vitiligo is estimated to affect 1.9 million–2.8 million adults in the United States and more than 100 million people worldwide. It cuts across races and genders and can be psychologically painful for many who live with it.
John E. Harris, MD, director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester, wrote about the Lego character in his blog “Speaking of Vitiligo ...” saying: “I could not be more excited. This new minifigure also serves as a way to educate both children and adults who are not familiar with vitiligo about the disease.”
He noted that until recently vitiligo representation in kids’ toys has been limited. “By adding diversity such as representations of vitiligo in toys, it can help remove stigmas associated with vitiligo and give children more options that they can relate to.”
Erika Page of Richmond, Va., who founded and edits the vitiligo blog “Living Dappled,” told this news organization she was thrilled to see the new Lego character.
“Growing up I didn’t know anyone who looked like me, let alone a toy or a character,” she said. The message the representations send is important not just for the kids but for the parents of kids with vitiligo who want to help their kids in any way they can.
Ms. Page was diagnosed with vitiligo at age 7 and struggled emotionally in her high school and college years when she often looked in the mirror, saw “giraffe-like” spots, and cried. Over time she lost 100% of her pigment to the condition and today at age 33, lives with universal vitiligo or overall very pale skin.
She founded the Living Dappled blog 6 years ago to help people with the disease feel less alone. The Lego character will also help with that, she said.
“Growing up with vitiligo was so isolating and you felt so different,” Ms. Page said. “Today we see billboards and models and dolls and now Legos that look like us. I hope this is a first of many to come for Lego.”
Dr. Harris and Ms. Page declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The
The character appears with the customizable array of players to assemble for a table football team.
It’s the latest representation of the disease as toymakers diversify their lines.
In May 2022, Mattel released a Ken doll with vitiligo after a Barbie with vitiligo was released in 2020. Rainbow High and other toy makers also have character versions.
The Lego addition follows a big summer medically for vitiligo as the first treatment was approved for repigmentation. In July, a cream formulation of ruxolitinib (Opzelura), a Janus kinase inhibitor, became the first repigmentation treatment approved by the Food and Drug Administration for nonsegmental vitiligo, the most common form of the disease.
Vitiligo is estimated to affect 1.9 million–2.8 million adults in the United States and more than 100 million people worldwide. It cuts across races and genders and can be psychologically painful for many who live with it.
John E. Harris, MD, director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester, wrote about the Lego character in his blog “Speaking of Vitiligo ...” saying: “I could not be more excited. This new minifigure also serves as a way to educate both children and adults who are not familiar with vitiligo about the disease.”
He noted that until recently vitiligo representation in kids’ toys has been limited. “By adding diversity such as representations of vitiligo in toys, it can help remove stigmas associated with vitiligo and give children more options that they can relate to.”
Erika Page of Richmond, Va., who founded and edits the vitiligo blog “Living Dappled,” told this news organization she was thrilled to see the new Lego character.
“Growing up I didn’t know anyone who looked like me, let alone a toy or a character,” she said. The message the representations send is important not just for the kids but for the parents of kids with vitiligo who want to help their kids in any way they can.
Ms. Page was diagnosed with vitiligo at age 7 and struggled emotionally in her high school and college years when she often looked in the mirror, saw “giraffe-like” spots, and cried. Over time she lost 100% of her pigment to the condition and today at age 33, lives with universal vitiligo or overall very pale skin.
She founded the Living Dappled blog 6 years ago to help people with the disease feel less alone. The Lego character will also help with that, she said.
“Growing up with vitiligo was so isolating and you felt so different,” Ms. Page said. “Today we see billboards and models and dolls and now Legos that look like us. I hope this is a first of many to come for Lego.”
Dr. Harris and Ms. Page declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The
The character appears with the customizable array of players to assemble for a table football team.
It’s the latest representation of the disease as toymakers diversify their lines.
In May 2022, Mattel released a Ken doll with vitiligo after a Barbie with vitiligo was released in 2020. Rainbow High and other toy makers also have character versions.
The Lego addition follows a big summer medically for vitiligo as the first treatment was approved for repigmentation. In July, a cream formulation of ruxolitinib (Opzelura), a Janus kinase inhibitor, became the first repigmentation treatment approved by the Food and Drug Administration for nonsegmental vitiligo, the most common form of the disease.
Vitiligo is estimated to affect 1.9 million–2.8 million adults in the United States and more than 100 million people worldwide. It cuts across races and genders and can be psychologically painful for many who live with it.
John E. Harris, MD, director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester, wrote about the Lego character in his blog “Speaking of Vitiligo ...” saying: “I could not be more excited. This new minifigure also serves as a way to educate both children and adults who are not familiar with vitiligo about the disease.”
He noted that until recently vitiligo representation in kids’ toys has been limited. “By adding diversity such as representations of vitiligo in toys, it can help remove stigmas associated with vitiligo and give children more options that they can relate to.”
Erika Page of Richmond, Va., who founded and edits the vitiligo blog “Living Dappled,” told this news organization she was thrilled to see the new Lego character.
“Growing up I didn’t know anyone who looked like me, let alone a toy or a character,” she said. The message the representations send is important not just for the kids but for the parents of kids with vitiligo who want to help their kids in any way they can.
Ms. Page was diagnosed with vitiligo at age 7 and struggled emotionally in her high school and college years when she often looked in the mirror, saw “giraffe-like” spots, and cried. Over time she lost 100% of her pigment to the condition and today at age 33, lives with universal vitiligo or overall very pale skin.
She founded the Living Dappled blog 6 years ago to help people with the disease feel less alone. The Lego character will also help with that, she said.
“Growing up with vitiligo was so isolating and you felt so different,” Ms. Page said. “Today we see billboards and models and dolls and now Legos that look like us. I hope this is a first of many to come for Lego.”
Dr. Harris and Ms. Page declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Practical pearls guide treatment of psoriasis in tricky areas
LAS VEGAS – With the right regimen, a majority of patients with psoriasis can achieve at least a Psoriasis Area and Severity Index (PASI) 75 score, Jennifer Soung, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
The array of treatment options includes mainstay topicals, new nonsteroidals, traditional oral systemics, new oral systemics, biologics, and light therapy, said Dr. Soung, director of clinical research at Harbor-UCLA Medical Center, Santa Ana, Calif. However, she said.
For these areas, make sure the diagnosis of psoriasis is correct, to avoid wasting time on the wrong course of treatment, Dr. Soung emphasized.
Scalp strategies
The scalp is often the first area of the body affected by psoriasis, and patients with severe scalp psoriasis may have minimal plaques on the body, Dr. Soung said. However, a differential diagnosis should include seborrheic dermatitis, she noted.
For most cases of scalp psoriasis, “start with localized topical treatment,” such as vitamin D and corticosteroid combination therapy, or excimer laser, Dr. Soung advised.
Systemic treatments with demonstrated effectiveness on scalp psoriasis in post hoc analyses of patients with moderate to severe plaque psoriasis include adalimumab, etanercept, ixekizumab, and secukinumab. Studies specifically focused on treatment of scalp psoriasis have shown success with secukinumab and apremilast, she noted.
Roflumilast foam, 0.3%, is in development and is an emerging option for scalp psoriasis. (A cream formulation of roflumilast, a topical phosphodiesterase 4 inhibitor, was approved for treatment of plaque psoriasis in July 2022.) A phase 2b study of roflumilast foam showed that approximately one-third of patients with scalp psoriasis achieved a status of clear based on scalp-investigator global assessment, compared with approximately 3% of those on vehicle, and similar results were seen in a recently completed phase 3 trial for scalp and body psoriasis, she added.
Facial psoriasis
Patients with facial psoriasis tend to be younger, and they may have more severe disease overall, Dr. Soung said. Given the sensitivity of facial skin, “it is nice to have a nonsteroidal option,” she noted. Current novel nonsteroidal therapies include a cream formulation of tapinarof, an aryl hydrocarbon receptor agonist, which was approved earlier this year for plaque psoriasis in adults, and roflumilast cream. Vitamin D and topical calcineurin inhibitors are options as well, she said.
Intertriginous tricks
Intertriginous (inverse) psoriasis is distinct from other areas in that the plaques are usually smooth and well-demarcated, with little or no scaling, Dr. Soung said. Erosions or signs of maceration may be present. The prevalence of inverse psoriasis is approximately 30%, but the prevalence on external genitalia is 80%, she noted. For inverse psoriasis, topical corticosteroids can result in adverse events such as poor wound healing and skin fragility, and some patients resist the idea of a steroid and don’t adhere to the medication, she added. Dr. Soung recommended topical corticosteroids for the short term, and topical calcineurin inhibitors or calcipotriol for the long term.
New topical options for inverse psoriasis include tapinarof and roflumilast, Dr. Soung said. For tapinarof, the phase 3 PSOARING program included assessment of tolerability in sensitive skin areas and found little to no irritation. Similarly, treatment with roflumilast cream was effective and well tolerated by patients with intertriginous plaque psoriasis in the DERMIS-1 and DERMIS-2 studies, she said.
Genital psoriasis
Ask patients with psoriasis about genital psoriasis, because they often are too embarrassed to provide that information on their own, said Dr. Soung. In fact, 63% of patients with psoriasis report ever experiencing genital psoriasis, but it often goes undiagnosed and undertreated, which has a significant impact on patient quality of life and sexual health.
A differential diagnosis of genital psoriasis should include dermatitis, tinea or candidiasis, and even squamous cell carcinoma, she noted. Other considerations include fixed drug eruption, lichen nitidus, lichen sclerosus, and scabies.
Dr. Soung’s first line of treatment for genital psoriasis is low-potency topical corticosteroids for 2-4 weeks. If long-term topical therapy is needed, alternatives include calcineurin inhibitors and vitamin D analogs, she said. The new topicals roflumilast and tapinarof are options as well, she said.
For those patients with severe and extensive genital psoriasis, consider systemic therapy, possibly with ixekizumab or secukinumab, she added. Patients with moderate to severe genital psoriasis treated with apremilast have shown improvement at week 16, in an ongoing clinical trial, she noted.
Palmoplantar involvement
For patients with palmar plantar psoriasis, “don’t underestimate the impact on quality of life,” said Dr. Soung. Approximately 12%-16% of patients with psoriasis report palmoplantar involvement, she noted.
Palmoplantar psoriasis can be stubborn, and many patients will need combination therapy with topicals and systemics, she said. “I am very curious about how well our new topical nonsteroidals will work in these areas,” she added.
Dr. Soung starts patients with palmoplantar psoriasis with a “potent to super-potent” twice daily topical corticosteroid, with or without occlusion. Her first-line systemic therapy is acitretin, 10-50 mg daily. However, keep in mind that acitretin is contraindicated in pregnancy, and also may cause side effects including cheilitis, alopecia, and peeling skin, she cautioned.
During the question and answer session, Dr. Soung was asked whether she routinely biopsies patients with palmoplantar psoriasis. “Not always,” was her answer. Instead, she looks for clues elsewhere on the body to confirm the diagnosis.
Nail know-how
Approximately 23%-27% of patients with psoriasis experience nail involvement, said Dr. Soung. Nail psoriasis can appear on the nail plate as pitting, onycholysis, or subungual hyperkeratosis, or in the nail bed as splinter hemorrhages or oil spots, she said.
For patients with psoriasis of the nails only, Dr. Soung described the use of high-potency topical corticosteroids, with or without calcipotriol. In her experience, she said that intralesional steroids for nail psoriasis are torturous to patients. For patients who have failed topical therapy or have psoriasis in other areas, with or without psoriatic arthritis, she advised the use of either IL-17 antagonists (secukinumab, ixekizumab, brodalumab) or IL-23 antagonists (risankizumab, guselkumab).
Dr. Soung disclosed serving as a consultant or advisor for Arcutis, Bristol Myers Squibb Company, Dermavant, and Novartis. She also disclosed serving as a speaker or member of the speakers’ bureau for AbbVie, Amgen, Arcutis, Bristol Myers Squibb Company, Celgene, Leo Pharma, Eli Lilly, Novartis, Ortho Dermatologics, Pfizer, Regeneron, and Sanofi, as well as research funding from AbbVie, Amgen, Arcutis, Castle Biosciences, Dermavant, KoBio, Kyowa Kirin, Leo Pharma, Eli Lilly, Novartis, Pfizer, and UCB.
MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – With the right regimen, a majority of patients with psoriasis can achieve at least a Psoriasis Area and Severity Index (PASI) 75 score, Jennifer Soung, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
The array of treatment options includes mainstay topicals, new nonsteroidals, traditional oral systemics, new oral systemics, biologics, and light therapy, said Dr. Soung, director of clinical research at Harbor-UCLA Medical Center, Santa Ana, Calif. However, she said.
For these areas, make sure the diagnosis of psoriasis is correct, to avoid wasting time on the wrong course of treatment, Dr. Soung emphasized.
Scalp strategies
The scalp is often the first area of the body affected by psoriasis, and patients with severe scalp psoriasis may have minimal plaques on the body, Dr. Soung said. However, a differential diagnosis should include seborrheic dermatitis, she noted.
For most cases of scalp psoriasis, “start with localized topical treatment,” such as vitamin D and corticosteroid combination therapy, or excimer laser, Dr. Soung advised.
Systemic treatments with demonstrated effectiveness on scalp psoriasis in post hoc analyses of patients with moderate to severe plaque psoriasis include adalimumab, etanercept, ixekizumab, and secukinumab. Studies specifically focused on treatment of scalp psoriasis have shown success with secukinumab and apremilast, she noted.
Roflumilast foam, 0.3%, is in development and is an emerging option for scalp psoriasis. (A cream formulation of roflumilast, a topical phosphodiesterase 4 inhibitor, was approved for treatment of plaque psoriasis in July 2022.) A phase 2b study of roflumilast foam showed that approximately one-third of patients with scalp psoriasis achieved a status of clear based on scalp-investigator global assessment, compared with approximately 3% of those on vehicle, and similar results were seen in a recently completed phase 3 trial for scalp and body psoriasis, she added.
Facial psoriasis
Patients with facial psoriasis tend to be younger, and they may have more severe disease overall, Dr. Soung said. Given the sensitivity of facial skin, “it is nice to have a nonsteroidal option,” she noted. Current novel nonsteroidal therapies include a cream formulation of tapinarof, an aryl hydrocarbon receptor agonist, which was approved earlier this year for plaque psoriasis in adults, and roflumilast cream. Vitamin D and topical calcineurin inhibitors are options as well, she said.
Intertriginous tricks
Intertriginous (inverse) psoriasis is distinct from other areas in that the plaques are usually smooth and well-demarcated, with little or no scaling, Dr. Soung said. Erosions or signs of maceration may be present. The prevalence of inverse psoriasis is approximately 30%, but the prevalence on external genitalia is 80%, she noted. For inverse psoriasis, topical corticosteroids can result in adverse events such as poor wound healing and skin fragility, and some patients resist the idea of a steroid and don’t adhere to the medication, she added. Dr. Soung recommended topical corticosteroids for the short term, and topical calcineurin inhibitors or calcipotriol for the long term.
New topical options for inverse psoriasis include tapinarof and roflumilast, Dr. Soung said. For tapinarof, the phase 3 PSOARING program included assessment of tolerability in sensitive skin areas and found little to no irritation. Similarly, treatment with roflumilast cream was effective and well tolerated by patients with intertriginous plaque psoriasis in the DERMIS-1 and DERMIS-2 studies, she said.
Genital psoriasis
Ask patients with psoriasis about genital psoriasis, because they often are too embarrassed to provide that information on their own, said Dr. Soung. In fact, 63% of patients with psoriasis report ever experiencing genital psoriasis, but it often goes undiagnosed and undertreated, which has a significant impact on patient quality of life and sexual health.
A differential diagnosis of genital psoriasis should include dermatitis, tinea or candidiasis, and even squamous cell carcinoma, she noted. Other considerations include fixed drug eruption, lichen nitidus, lichen sclerosus, and scabies.
Dr. Soung’s first line of treatment for genital psoriasis is low-potency topical corticosteroids for 2-4 weeks. If long-term topical therapy is needed, alternatives include calcineurin inhibitors and vitamin D analogs, she said. The new topicals roflumilast and tapinarof are options as well, she said.
For those patients with severe and extensive genital psoriasis, consider systemic therapy, possibly with ixekizumab or secukinumab, she added. Patients with moderate to severe genital psoriasis treated with apremilast have shown improvement at week 16, in an ongoing clinical trial, she noted.
Palmoplantar involvement
For patients with palmar plantar psoriasis, “don’t underestimate the impact on quality of life,” said Dr. Soung. Approximately 12%-16% of patients with psoriasis report palmoplantar involvement, she noted.
Palmoplantar psoriasis can be stubborn, and many patients will need combination therapy with topicals and systemics, she said. “I am very curious about how well our new topical nonsteroidals will work in these areas,” she added.
Dr. Soung starts patients with palmoplantar psoriasis with a “potent to super-potent” twice daily topical corticosteroid, with or without occlusion. Her first-line systemic therapy is acitretin, 10-50 mg daily. However, keep in mind that acitretin is contraindicated in pregnancy, and also may cause side effects including cheilitis, alopecia, and peeling skin, she cautioned.
During the question and answer session, Dr. Soung was asked whether she routinely biopsies patients with palmoplantar psoriasis. “Not always,” was her answer. Instead, she looks for clues elsewhere on the body to confirm the diagnosis.
Nail know-how
Approximately 23%-27% of patients with psoriasis experience nail involvement, said Dr. Soung. Nail psoriasis can appear on the nail plate as pitting, onycholysis, or subungual hyperkeratosis, or in the nail bed as splinter hemorrhages or oil spots, she said.
For patients with psoriasis of the nails only, Dr. Soung described the use of high-potency topical corticosteroids, with or without calcipotriol. In her experience, she said that intralesional steroids for nail psoriasis are torturous to patients. For patients who have failed topical therapy or have psoriasis in other areas, with or without psoriatic arthritis, she advised the use of either IL-17 antagonists (secukinumab, ixekizumab, brodalumab) or IL-23 antagonists (risankizumab, guselkumab).
Dr. Soung disclosed serving as a consultant or advisor for Arcutis, Bristol Myers Squibb Company, Dermavant, and Novartis. She also disclosed serving as a speaker or member of the speakers’ bureau for AbbVie, Amgen, Arcutis, Bristol Myers Squibb Company, Celgene, Leo Pharma, Eli Lilly, Novartis, Ortho Dermatologics, Pfizer, Regeneron, and Sanofi, as well as research funding from AbbVie, Amgen, Arcutis, Castle Biosciences, Dermavant, KoBio, Kyowa Kirin, Leo Pharma, Eli Lilly, Novartis, Pfizer, and UCB.
MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – With the right regimen, a majority of patients with psoriasis can achieve at least a Psoriasis Area and Severity Index (PASI) 75 score, Jennifer Soung, MD, said in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
The array of treatment options includes mainstay topicals, new nonsteroidals, traditional oral systemics, new oral systemics, biologics, and light therapy, said Dr. Soung, director of clinical research at Harbor-UCLA Medical Center, Santa Ana, Calif. However, she said.
For these areas, make sure the diagnosis of psoriasis is correct, to avoid wasting time on the wrong course of treatment, Dr. Soung emphasized.
Scalp strategies
The scalp is often the first area of the body affected by psoriasis, and patients with severe scalp psoriasis may have minimal plaques on the body, Dr. Soung said. However, a differential diagnosis should include seborrheic dermatitis, she noted.
For most cases of scalp psoriasis, “start with localized topical treatment,” such as vitamin D and corticosteroid combination therapy, or excimer laser, Dr. Soung advised.
Systemic treatments with demonstrated effectiveness on scalp psoriasis in post hoc analyses of patients with moderate to severe plaque psoriasis include adalimumab, etanercept, ixekizumab, and secukinumab. Studies specifically focused on treatment of scalp psoriasis have shown success with secukinumab and apremilast, she noted.
Roflumilast foam, 0.3%, is in development and is an emerging option for scalp psoriasis. (A cream formulation of roflumilast, a topical phosphodiesterase 4 inhibitor, was approved for treatment of plaque psoriasis in July 2022.) A phase 2b study of roflumilast foam showed that approximately one-third of patients with scalp psoriasis achieved a status of clear based on scalp-investigator global assessment, compared with approximately 3% of those on vehicle, and similar results were seen in a recently completed phase 3 trial for scalp and body psoriasis, she added.
Facial psoriasis
Patients with facial psoriasis tend to be younger, and they may have more severe disease overall, Dr. Soung said. Given the sensitivity of facial skin, “it is nice to have a nonsteroidal option,” she noted. Current novel nonsteroidal therapies include a cream formulation of tapinarof, an aryl hydrocarbon receptor agonist, which was approved earlier this year for plaque psoriasis in adults, and roflumilast cream. Vitamin D and topical calcineurin inhibitors are options as well, she said.
Intertriginous tricks
Intertriginous (inverse) psoriasis is distinct from other areas in that the plaques are usually smooth and well-demarcated, with little or no scaling, Dr. Soung said. Erosions or signs of maceration may be present. The prevalence of inverse psoriasis is approximately 30%, but the prevalence on external genitalia is 80%, she noted. For inverse psoriasis, topical corticosteroids can result in adverse events such as poor wound healing and skin fragility, and some patients resist the idea of a steroid and don’t adhere to the medication, she added. Dr. Soung recommended topical corticosteroids for the short term, and topical calcineurin inhibitors or calcipotriol for the long term.
New topical options for inverse psoriasis include tapinarof and roflumilast, Dr. Soung said. For tapinarof, the phase 3 PSOARING program included assessment of tolerability in sensitive skin areas and found little to no irritation. Similarly, treatment with roflumilast cream was effective and well tolerated by patients with intertriginous plaque psoriasis in the DERMIS-1 and DERMIS-2 studies, she said.
Genital psoriasis
Ask patients with psoriasis about genital psoriasis, because they often are too embarrassed to provide that information on their own, said Dr. Soung. In fact, 63% of patients with psoriasis report ever experiencing genital psoriasis, but it often goes undiagnosed and undertreated, which has a significant impact on patient quality of life and sexual health.
A differential diagnosis of genital psoriasis should include dermatitis, tinea or candidiasis, and even squamous cell carcinoma, she noted. Other considerations include fixed drug eruption, lichen nitidus, lichen sclerosus, and scabies.
Dr. Soung’s first line of treatment for genital psoriasis is low-potency topical corticosteroids for 2-4 weeks. If long-term topical therapy is needed, alternatives include calcineurin inhibitors and vitamin D analogs, she said. The new topicals roflumilast and tapinarof are options as well, she said.
For those patients with severe and extensive genital psoriasis, consider systemic therapy, possibly with ixekizumab or secukinumab, she added. Patients with moderate to severe genital psoriasis treated with apremilast have shown improvement at week 16, in an ongoing clinical trial, she noted.
Palmoplantar involvement
For patients with palmar plantar psoriasis, “don’t underestimate the impact on quality of life,” said Dr. Soung. Approximately 12%-16% of patients with psoriasis report palmoplantar involvement, she noted.
Palmoplantar psoriasis can be stubborn, and many patients will need combination therapy with topicals and systemics, she said. “I am very curious about how well our new topical nonsteroidals will work in these areas,” she added.
Dr. Soung starts patients with palmoplantar psoriasis with a “potent to super-potent” twice daily topical corticosteroid, with or without occlusion. Her first-line systemic therapy is acitretin, 10-50 mg daily. However, keep in mind that acitretin is contraindicated in pregnancy, and also may cause side effects including cheilitis, alopecia, and peeling skin, she cautioned.
During the question and answer session, Dr. Soung was asked whether she routinely biopsies patients with palmoplantar psoriasis. “Not always,” was her answer. Instead, she looks for clues elsewhere on the body to confirm the diagnosis.
Nail know-how
Approximately 23%-27% of patients with psoriasis experience nail involvement, said Dr. Soung. Nail psoriasis can appear on the nail plate as pitting, onycholysis, or subungual hyperkeratosis, or in the nail bed as splinter hemorrhages or oil spots, she said.
For patients with psoriasis of the nails only, Dr. Soung described the use of high-potency topical corticosteroids, with or without calcipotriol. In her experience, she said that intralesional steroids for nail psoriasis are torturous to patients. For patients who have failed topical therapy or have psoriasis in other areas, with or without psoriatic arthritis, she advised the use of either IL-17 antagonists (secukinumab, ixekizumab, brodalumab) or IL-23 antagonists (risankizumab, guselkumab).
Dr. Soung disclosed serving as a consultant or advisor for Arcutis, Bristol Myers Squibb Company, Dermavant, and Novartis. She also disclosed serving as a speaker or member of the speakers’ bureau for AbbVie, Amgen, Arcutis, Bristol Myers Squibb Company, Celgene, Leo Pharma, Eli Lilly, Novartis, Ortho Dermatologics, Pfizer, Regeneron, and Sanofi, as well as research funding from AbbVie, Amgen, Arcutis, Castle Biosciences, Dermavant, KoBio, Kyowa Kirin, Leo Pharma, Eli Lilly, Novartis, Pfizer, and UCB.
MedscapeLive and this news organization are owned by the same parent company.
AT INNOVATIONS IN DERMATOLOGY
A cost-effective de-escalation strategy in advanced melanoma
Response-adapted , an economic analysis found.
“The rising costs of cancer therapies are becoming untenable for both patients and payers, and there is both clinical and economic benefit to finding less expensive treatment alternatives,” Wolfgang Kunz, MD, University Hospital, Ludwig Maximilian University of Munich, told this news organization.
This economic analysis “highlights that leveraging modern diagnostic capabilities can do just that: Pairing drug regimens with CT-image analysis to optimize dosages can reduce health care costs and improve clinical outcomes,” Dr. Kunz said.
The study was published online in JAMA Dermatology.
While the use of immunotherapies over the past decade has improved the prognosis for patients with advanced melanoma, these drugs come with a hefty price tag.
One potential way to help reduce costs: de-escalate therapy. The ADAPT-IT trial demonstrated similar progression-free and overall survival among patients who received response-adapted ipilimumab discontinuation and those who received standard of care.
In the current analysis, Dr. Kunz and colleagues wanted to understand whether this response-adapted approach was also cost effective.
The team applied economic modeling to data from the ADAPT-IT trial as well as CheckMate 067, in which patients received standard of care four doses of combination ipilimumab-nivolumab followed by nivolumab monotherapy. In the ADAPT-IT trial, patients also initially received the immunotherapy combination but had CT scans to determine their response after two doses; if they responded, patients discontinued ipilimumab and continued with nivolumab monotherapy.
Overall, ADAPT-IT showed that responders could forgo the additional two doses of ipilimumab plus nivolumab while maintaining similar progression-free survival and overall survival seen at 18 months in the CheckMate 067 trial.
The current economic analysis, based on 41 patients from ADAPT-IT and 314 from CheckMate 067, showed a potential reduction in health care costs of $19,891 per patient with the response-adapted approach.
Response-adapted treatment was the cost-effective option in 94% of simulated scenarios.
When extrapolated to 2019 incidence rates of distant melanoma cases, yearly national savings could reach about $58 million.
“In the relatively small space of immunotherapies in advanced melanoma, we hope this analysis motivates clinicians to consider response-adapted treatment,” Dr. Kunz told this news organization.
“On the larger scale, this analysis serves as a stepping stone to more response-guided treatment protocols,” Dr. Kunz added. “With drug costs rising and imaging capabilities growing, more frequent image-guided adjustments are a perfect fit into the personalized care model.”
When applying the cost savings noted in this analysis across all treated patients, “the economic impact may be profound,” said Joseph Skitzki, MD, surgical oncologist, Roswell Park Comprehensive Cancer Center, Buffalo, N.Y., who wasn’t involved in the study. The “financial toxicity of cancer care is increasingly recognized as a potential barrier to optimal outcomes and any measures to mitigate cost may be impactful.”
However, Dr. Skitzki said several caveats need to be considered.
One is that the data included from ADAPT-IT only included 41 patients, compared with 314 patients from CheckMate 067.
“It is possible that a larger real-world study utilizing the ADAPT-IT protocol may not be as favorable in terms of outcomes and could lessen the economic impact of de-escalation, although any form of de-escalation is likely to have a cost benefit,” Dr. Skitzki said in an interview.
A real-world response–adapted de-escalation clinical trial, with an emphasis on costs and a benchmark of similar progression-free and overall survival, should be conducted before the de-escalated option becomes “practice changing,” Dr. Skitzki said.
Jeffrey Weber, MD, PhD, deputy director, Perlmutter Cancer Center, NYU Langone Health, New York, also urged caution in interpreting the results.
“I would not base treatment decisions on a small sampling of 41 patients in the absence of a randomized comparison,” Dr. Weber told this news organization. “Without a proper comparison, I would not advocate using only two doses of ipilimumab-nivolumab to make decisions on treatment.”
Dr. Skitzki added that, while “studies like this one are desperately needed to lessen the economic impact of new and emerging combination immunotherapies,” there is likely also a “disincentive for pharmaceutical companies to conduct this type of research.”
This research had no specific funding. Dr. Kunz and Dr. Skitzki reported no relevant conflicts of interest. Dr. Weber disclosed relationships with Merck, Genentech, AstraZeneca, Pfizer, Regeneron, and GSK, among others, and holds equity in Cytomx, Biond, NexImmune, and Immunomax.
A version of this article first appeared on Medscape.com.
Response-adapted , an economic analysis found.
“The rising costs of cancer therapies are becoming untenable for both patients and payers, and there is both clinical and economic benefit to finding less expensive treatment alternatives,” Wolfgang Kunz, MD, University Hospital, Ludwig Maximilian University of Munich, told this news organization.
This economic analysis “highlights that leveraging modern diagnostic capabilities can do just that: Pairing drug regimens with CT-image analysis to optimize dosages can reduce health care costs and improve clinical outcomes,” Dr. Kunz said.
The study was published online in JAMA Dermatology.
While the use of immunotherapies over the past decade has improved the prognosis for patients with advanced melanoma, these drugs come with a hefty price tag.
One potential way to help reduce costs: de-escalate therapy. The ADAPT-IT trial demonstrated similar progression-free and overall survival among patients who received response-adapted ipilimumab discontinuation and those who received standard of care.
In the current analysis, Dr. Kunz and colleagues wanted to understand whether this response-adapted approach was also cost effective.
The team applied economic modeling to data from the ADAPT-IT trial as well as CheckMate 067, in which patients received standard of care four doses of combination ipilimumab-nivolumab followed by nivolumab monotherapy. In the ADAPT-IT trial, patients also initially received the immunotherapy combination but had CT scans to determine their response after two doses; if they responded, patients discontinued ipilimumab and continued with nivolumab monotherapy.
Overall, ADAPT-IT showed that responders could forgo the additional two doses of ipilimumab plus nivolumab while maintaining similar progression-free survival and overall survival seen at 18 months in the CheckMate 067 trial.
The current economic analysis, based on 41 patients from ADAPT-IT and 314 from CheckMate 067, showed a potential reduction in health care costs of $19,891 per patient with the response-adapted approach.
Response-adapted treatment was the cost-effective option in 94% of simulated scenarios.
When extrapolated to 2019 incidence rates of distant melanoma cases, yearly national savings could reach about $58 million.
“In the relatively small space of immunotherapies in advanced melanoma, we hope this analysis motivates clinicians to consider response-adapted treatment,” Dr. Kunz told this news organization.
“On the larger scale, this analysis serves as a stepping stone to more response-guided treatment protocols,” Dr. Kunz added. “With drug costs rising and imaging capabilities growing, more frequent image-guided adjustments are a perfect fit into the personalized care model.”
When applying the cost savings noted in this analysis across all treated patients, “the economic impact may be profound,” said Joseph Skitzki, MD, surgical oncologist, Roswell Park Comprehensive Cancer Center, Buffalo, N.Y., who wasn’t involved in the study. The “financial toxicity of cancer care is increasingly recognized as a potential barrier to optimal outcomes and any measures to mitigate cost may be impactful.”
However, Dr. Skitzki said several caveats need to be considered.
One is that the data included from ADAPT-IT only included 41 patients, compared with 314 patients from CheckMate 067.
“It is possible that a larger real-world study utilizing the ADAPT-IT protocol may not be as favorable in terms of outcomes and could lessen the economic impact of de-escalation, although any form of de-escalation is likely to have a cost benefit,” Dr. Skitzki said in an interview.
A real-world response–adapted de-escalation clinical trial, with an emphasis on costs and a benchmark of similar progression-free and overall survival, should be conducted before the de-escalated option becomes “practice changing,” Dr. Skitzki said.
Jeffrey Weber, MD, PhD, deputy director, Perlmutter Cancer Center, NYU Langone Health, New York, also urged caution in interpreting the results.
“I would not base treatment decisions on a small sampling of 41 patients in the absence of a randomized comparison,” Dr. Weber told this news organization. “Without a proper comparison, I would not advocate using only two doses of ipilimumab-nivolumab to make decisions on treatment.”
Dr. Skitzki added that, while “studies like this one are desperately needed to lessen the economic impact of new and emerging combination immunotherapies,” there is likely also a “disincentive for pharmaceutical companies to conduct this type of research.”
This research had no specific funding. Dr. Kunz and Dr. Skitzki reported no relevant conflicts of interest. Dr. Weber disclosed relationships with Merck, Genentech, AstraZeneca, Pfizer, Regeneron, and GSK, among others, and holds equity in Cytomx, Biond, NexImmune, and Immunomax.
A version of this article first appeared on Medscape.com.
Response-adapted , an economic analysis found.
“The rising costs of cancer therapies are becoming untenable for both patients and payers, and there is both clinical and economic benefit to finding less expensive treatment alternatives,” Wolfgang Kunz, MD, University Hospital, Ludwig Maximilian University of Munich, told this news organization.
This economic analysis “highlights that leveraging modern diagnostic capabilities can do just that: Pairing drug regimens with CT-image analysis to optimize dosages can reduce health care costs and improve clinical outcomes,” Dr. Kunz said.
The study was published online in JAMA Dermatology.
While the use of immunotherapies over the past decade has improved the prognosis for patients with advanced melanoma, these drugs come with a hefty price tag.
One potential way to help reduce costs: de-escalate therapy. The ADAPT-IT trial demonstrated similar progression-free and overall survival among patients who received response-adapted ipilimumab discontinuation and those who received standard of care.
In the current analysis, Dr. Kunz and colleagues wanted to understand whether this response-adapted approach was also cost effective.
The team applied economic modeling to data from the ADAPT-IT trial as well as CheckMate 067, in which patients received standard of care four doses of combination ipilimumab-nivolumab followed by nivolumab monotherapy. In the ADAPT-IT trial, patients also initially received the immunotherapy combination but had CT scans to determine their response after two doses; if they responded, patients discontinued ipilimumab and continued with nivolumab monotherapy.
Overall, ADAPT-IT showed that responders could forgo the additional two doses of ipilimumab plus nivolumab while maintaining similar progression-free survival and overall survival seen at 18 months in the CheckMate 067 trial.
The current economic analysis, based on 41 patients from ADAPT-IT and 314 from CheckMate 067, showed a potential reduction in health care costs of $19,891 per patient with the response-adapted approach.
Response-adapted treatment was the cost-effective option in 94% of simulated scenarios.
When extrapolated to 2019 incidence rates of distant melanoma cases, yearly national savings could reach about $58 million.
“In the relatively small space of immunotherapies in advanced melanoma, we hope this analysis motivates clinicians to consider response-adapted treatment,” Dr. Kunz told this news organization.
“On the larger scale, this analysis serves as a stepping stone to more response-guided treatment protocols,” Dr. Kunz added. “With drug costs rising and imaging capabilities growing, more frequent image-guided adjustments are a perfect fit into the personalized care model.”
When applying the cost savings noted in this analysis across all treated patients, “the economic impact may be profound,” said Joseph Skitzki, MD, surgical oncologist, Roswell Park Comprehensive Cancer Center, Buffalo, N.Y., who wasn’t involved in the study. The “financial toxicity of cancer care is increasingly recognized as a potential barrier to optimal outcomes and any measures to mitigate cost may be impactful.”
However, Dr. Skitzki said several caveats need to be considered.
One is that the data included from ADAPT-IT only included 41 patients, compared with 314 patients from CheckMate 067.
“It is possible that a larger real-world study utilizing the ADAPT-IT protocol may not be as favorable in terms of outcomes and could lessen the economic impact of de-escalation, although any form of de-escalation is likely to have a cost benefit,” Dr. Skitzki said in an interview.
A real-world response–adapted de-escalation clinical trial, with an emphasis on costs and a benchmark of similar progression-free and overall survival, should be conducted before the de-escalated option becomes “practice changing,” Dr. Skitzki said.
Jeffrey Weber, MD, PhD, deputy director, Perlmutter Cancer Center, NYU Langone Health, New York, also urged caution in interpreting the results.
“I would not base treatment decisions on a small sampling of 41 patients in the absence of a randomized comparison,” Dr. Weber told this news organization. “Without a proper comparison, I would not advocate using only two doses of ipilimumab-nivolumab to make decisions on treatment.”
Dr. Skitzki added that, while “studies like this one are desperately needed to lessen the economic impact of new and emerging combination immunotherapies,” there is likely also a “disincentive for pharmaceutical companies to conduct this type of research.”
This research had no specific funding. Dr. Kunz and Dr. Skitzki reported no relevant conflicts of interest. Dr. Weber disclosed relationships with Merck, Genentech, AstraZeneca, Pfizer, Regeneron, and GSK, among others, and holds equity in Cytomx, Biond, NexImmune, and Immunomax.
A version of this article first appeared on Medscape.com.