User login
Monkeypox features include mucocutaneous involvement in almost all cases
MILAN – In the current spread of monkeypox among countries outside of Africa, this zoonotic orthopox DNA virus is sexually transmitted in more than 90% of cases, mostly among men having sex with men (MSM), and can produce severe skin and systemic symptoms but is rarely fatal, according to a breaking news presentation at the annual congress of the European Academy of Dermatology and Venereology.
Synthesizing data from 185 cases in Spain with several sets of recently published data, Alba Català, MD, a dermatologist at Centro Médico Teknon, Barcelona, said at the meeting that there have been only two deaths in Spain in the current epidemic. (As of Sept. 30, after the EADV meeting had concluded, a total of three deaths related to monkeypox in Spain and one death in the United States had been reported, according to the Centers for Disease Control and Prevention).
Hospitalizations have been uncommon, and in Spain, there were only four hospitalizations, according to data collected from the beginning of May through early August, she said. Almost all cases in this Spanish series were from men having high-risk sex with men. Upon screening, 76% had another sexually transmitted disease, including 41% infected with human immunodeficiency virus.
More than 40% of patients with monkeypox have HIV
These data are consistent with several other recently published studies, such as one that evaluated 528 infections in 16 non-African countries, including those in North America, South America, Europe, the Mideast, as well as Australia. In that survey, published in the New England Journal of Medicine, and covering cases between late April and late June, 2022, 41% were HIV positive. Of those who were HIV negative, 57% were taking a pre-exposure prophylaxis regimen of antiretroviral drugs to prevent HIV infection.
However, these data do not preclude a significant risk of nonsexual transmission, according to Dr. Català, who noted that respiratory transmission and transmission through nonsexual skin contact is well documented in endemic areas.
“The virus has no preference for a sexual orientation,” Dr. Català cautioned. Despite the consistency of the data in regard to a largely MSM transmission in the epidemic so far, “these data may change with further spread of infection in the community.”
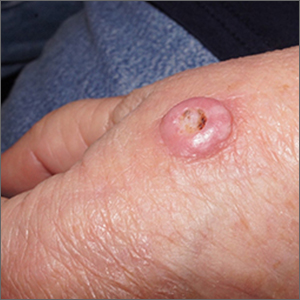
Typically, the incubation period of monkeypox lasts several days before the invasive period, which is commonly accompanied by systemic complaints, particularly fever, headache, and often lymphadenopathy. These systemic features usually but not always precede cutaneous involvement, which is seen in more than 90% of patients, according to Dr. Català. In the Spanish series, mucocutaneous involvement was recorded in 100% of patients.
Monkeypox and smallpox
“The differential diagnosis might include other vesicular eruptions, such as those caused by varicella or smallpox,” reported Dr. Català, who noted that monkeypox and smallpox are related.
Cutaneous lesions often appear first at the site of infection, such as the genitalia, but typically spread in a secondary eruption that is pruritic and may take days to resolve, according to Dr. Català. She reported that single lesions are less common but do occur. While hundreds of lesions have been reported among cases in endemic areas, most patients had 25 lesions or fewer in the Spanish epidemic and other recent series.
Even though there is a common progression in which lesions begin in a papular stage before the vesicular and pustular stages in a given area, new eruptions can occur before a prior eruption develops scabs.
“Frequently, not all the patient’s lesions are in the same stage of development,” said Dr. Català, who explained that disease activity and its complications, such as proctitis, pharyngitis, and penile edema, can take weeks to resolve. Because of the highly invasive nature of monkeypox, it is appropriate to be alert to less common manifestations, such as ocular involvement.
Many of these and other complications, such as secondary bacterial infections, will require targeted treatment, but the mainstay of therapy for the dermatologic manifestations of monkeypox is symptomatic treatment that includes nonsteroidal anti-inflammatory drugs and analgesics.
Re-epithelialization reduces transmission risk
“A clean, moist environment can mitigate transmission potential by covering infectious sores and promoting the re-epithelialization of the damaged exanthem,” Dr. Català advised. Tecovirimat (TPOXX, ST-246), an antiviral drug for smallpox, is approved for treating monkeypox in Europe but not in the United States (but it is approved for smallpox in the United States). Another antiviral drug, brincidofovir (CMX001 or Tembexa), is approved for smallpox in the United States, but not in Europe, according to Dr. Català. (In the United States, no treatment is specifically approved for treating monkeypox, but antivirals developed for smallpox “may prove beneficial against monkeypox,” according to the CDC.)
But she advised weighing the risks and benefits of using either drug in any individual patient.
The data suggest that the risk of viral shedding persists until the late stages of the disease trajectory. “A person is considered infectious from the onset of clinical manifestations until all skin lesions have scabbed over and re-epithelization has occurred,” Dr. Català said.
The prolonged period of contagion might be one reason to expect monkeypox to be transmitted more generally than it is now, according to Boghuma K. Titanji, MD, PhD, assistant professor of infectious diseases, Emory University, Atlanta.
“The longer the outbreak persists, the more likely we will see cases reported in groups other than MSM who have been most affected so far,” said Dr. Titanji, the first author of a recently published review article on monkeypox in Open Forum Infectious Diseases.
However, he acknowledged that a COVID-like spread is not expected. “The spread of monkeypox requires close and prolonged contact and is generally inefficient via fomites and droplet modes of transmission,” Dr. Titanji said in an interview. “Spread in heterosexual networks and congregate settings like crowded jails where close contact is unavoidable remains a concern that we need to educate the public about and maintain a high level of vigilance for.”
Dr. Català and Dr. Titanji report no potential conflicts of interest.
MILAN – In the current spread of monkeypox among countries outside of Africa, this zoonotic orthopox DNA virus is sexually transmitted in more than 90% of cases, mostly among men having sex with men (MSM), and can produce severe skin and systemic symptoms but is rarely fatal, according to a breaking news presentation at the annual congress of the European Academy of Dermatology and Venereology.
Synthesizing data from 185 cases in Spain with several sets of recently published data, Alba Català, MD, a dermatologist at Centro Médico Teknon, Barcelona, said at the meeting that there have been only two deaths in Spain in the current epidemic. (As of Sept. 30, after the EADV meeting had concluded, a total of three deaths related to monkeypox in Spain and one death in the United States had been reported, according to the Centers for Disease Control and Prevention).
Hospitalizations have been uncommon, and in Spain, there were only four hospitalizations, according to data collected from the beginning of May through early August, she said. Almost all cases in this Spanish series were from men having high-risk sex with men. Upon screening, 76% had another sexually transmitted disease, including 41% infected with human immunodeficiency virus.
More than 40% of patients with monkeypox have HIV
These data are consistent with several other recently published studies, such as one that evaluated 528 infections in 16 non-African countries, including those in North America, South America, Europe, the Mideast, as well as Australia. In that survey, published in the New England Journal of Medicine, and covering cases between late April and late June, 2022, 41% were HIV positive. Of those who were HIV negative, 57% were taking a pre-exposure prophylaxis regimen of antiretroviral drugs to prevent HIV infection.
However, these data do not preclude a significant risk of nonsexual transmission, according to Dr. Català, who noted that respiratory transmission and transmission through nonsexual skin contact is well documented in endemic areas.
“The virus has no preference for a sexual orientation,” Dr. Català cautioned. Despite the consistency of the data in regard to a largely MSM transmission in the epidemic so far, “these data may change with further spread of infection in the community.”
Typically, the incubation period of monkeypox lasts several days before the invasive period, which is commonly accompanied by systemic complaints, particularly fever, headache, and often lymphadenopathy. These systemic features usually but not always precede cutaneous involvement, which is seen in more than 90% of patients, according to Dr. Català. In the Spanish series, mucocutaneous involvement was recorded in 100% of patients.
Monkeypox and smallpox
“The differential diagnosis might include other vesicular eruptions, such as those caused by varicella or smallpox,” reported Dr. Català, who noted that monkeypox and smallpox are related.
Cutaneous lesions often appear first at the site of infection, such as the genitalia, but typically spread in a secondary eruption that is pruritic and may take days to resolve, according to Dr. Català. She reported that single lesions are less common but do occur. While hundreds of lesions have been reported among cases in endemic areas, most patients had 25 lesions or fewer in the Spanish epidemic and other recent series.
Even though there is a common progression in which lesions begin in a papular stage before the vesicular and pustular stages in a given area, new eruptions can occur before a prior eruption develops scabs.
“Frequently, not all the patient’s lesions are in the same stage of development,” said Dr. Català, who explained that disease activity and its complications, such as proctitis, pharyngitis, and penile edema, can take weeks to resolve. Because of the highly invasive nature of monkeypox, it is appropriate to be alert to less common manifestations, such as ocular involvement.
Many of these and other complications, such as secondary bacterial infections, will require targeted treatment, but the mainstay of therapy for the dermatologic manifestations of monkeypox is symptomatic treatment that includes nonsteroidal anti-inflammatory drugs and analgesics.
Re-epithelialization reduces transmission risk
“A clean, moist environment can mitigate transmission potential by covering infectious sores and promoting the re-epithelialization of the damaged exanthem,” Dr. Català advised. Tecovirimat (TPOXX, ST-246), an antiviral drug for smallpox, is approved for treating monkeypox in Europe but not in the United States (but it is approved for smallpox in the United States). Another antiviral drug, brincidofovir (CMX001 or Tembexa), is approved for smallpox in the United States, but not in Europe, according to Dr. Català. (In the United States, no treatment is specifically approved for treating monkeypox, but antivirals developed for smallpox “may prove beneficial against monkeypox,” according to the CDC.)
But she advised weighing the risks and benefits of using either drug in any individual patient.
The data suggest that the risk of viral shedding persists until the late stages of the disease trajectory. “A person is considered infectious from the onset of clinical manifestations until all skin lesions have scabbed over and re-epithelization has occurred,” Dr. Català said.
The prolonged period of contagion might be one reason to expect monkeypox to be transmitted more generally than it is now, according to Boghuma K. Titanji, MD, PhD, assistant professor of infectious diseases, Emory University, Atlanta.
“The longer the outbreak persists, the more likely we will see cases reported in groups other than MSM who have been most affected so far,” said Dr. Titanji, the first author of a recently published review article on monkeypox in Open Forum Infectious Diseases.
However, he acknowledged that a COVID-like spread is not expected. “The spread of monkeypox requires close and prolonged contact and is generally inefficient via fomites and droplet modes of transmission,” Dr. Titanji said in an interview. “Spread in heterosexual networks and congregate settings like crowded jails where close contact is unavoidable remains a concern that we need to educate the public about and maintain a high level of vigilance for.”
Dr. Català and Dr. Titanji report no potential conflicts of interest.
MILAN – In the current spread of monkeypox among countries outside of Africa, this zoonotic orthopox DNA virus is sexually transmitted in more than 90% of cases, mostly among men having sex with men (MSM), and can produce severe skin and systemic symptoms but is rarely fatal, according to a breaking news presentation at the annual congress of the European Academy of Dermatology and Venereology.
Synthesizing data from 185 cases in Spain with several sets of recently published data, Alba Català, MD, a dermatologist at Centro Médico Teknon, Barcelona, said at the meeting that there have been only two deaths in Spain in the current epidemic. (As of Sept. 30, after the EADV meeting had concluded, a total of three deaths related to monkeypox in Spain and one death in the United States had been reported, according to the Centers for Disease Control and Prevention).
Hospitalizations have been uncommon, and in Spain, there were only four hospitalizations, according to data collected from the beginning of May through early August, she said. Almost all cases in this Spanish series were from men having high-risk sex with men. Upon screening, 76% had another sexually transmitted disease, including 41% infected with human immunodeficiency virus.
More than 40% of patients with monkeypox have HIV
These data are consistent with several other recently published studies, such as one that evaluated 528 infections in 16 non-African countries, including those in North America, South America, Europe, the Mideast, as well as Australia. In that survey, published in the New England Journal of Medicine, and covering cases between late April and late June, 2022, 41% were HIV positive. Of those who were HIV negative, 57% were taking a pre-exposure prophylaxis regimen of antiretroviral drugs to prevent HIV infection.
However, these data do not preclude a significant risk of nonsexual transmission, according to Dr. Català, who noted that respiratory transmission and transmission through nonsexual skin contact is well documented in endemic areas.
“The virus has no preference for a sexual orientation,” Dr. Català cautioned. Despite the consistency of the data in regard to a largely MSM transmission in the epidemic so far, “these data may change with further spread of infection in the community.”
Typically, the incubation period of monkeypox lasts several days before the invasive period, which is commonly accompanied by systemic complaints, particularly fever, headache, and often lymphadenopathy. These systemic features usually but not always precede cutaneous involvement, which is seen in more than 90% of patients, according to Dr. Català. In the Spanish series, mucocutaneous involvement was recorded in 100% of patients.
Monkeypox and smallpox
“The differential diagnosis might include other vesicular eruptions, such as those caused by varicella or smallpox,” reported Dr. Català, who noted that monkeypox and smallpox are related.
Cutaneous lesions often appear first at the site of infection, such as the genitalia, but typically spread in a secondary eruption that is pruritic and may take days to resolve, according to Dr. Català. She reported that single lesions are less common but do occur. While hundreds of lesions have been reported among cases in endemic areas, most patients had 25 lesions or fewer in the Spanish epidemic and other recent series.
Even though there is a common progression in which lesions begin in a papular stage before the vesicular and pustular stages in a given area, new eruptions can occur before a prior eruption develops scabs.
“Frequently, not all the patient’s lesions are in the same stage of development,” said Dr. Català, who explained that disease activity and its complications, such as proctitis, pharyngitis, and penile edema, can take weeks to resolve. Because of the highly invasive nature of monkeypox, it is appropriate to be alert to less common manifestations, such as ocular involvement.
Many of these and other complications, such as secondary bacterial infections, will require targeted treatment, but the mainstay of therapy for the dermatologic manifestations of monkeypox is symptomatic treatment that includes nonsteroidal anti-inflammatory drugs and analgesics.
Re-epithelialization reduces transmission risk
“A clean, moist environment can mitigate transmission potential by covering infectious sores and promoting the re-epithelialization of the damaged exanthem,” Dr. Català advised. Tecovirimat (TPOXX, ST-246), an antiviral drug for smallpox, is approved for treating monkeypox in Europe but not in the United States (but it is approved for smallpox in the United States). Another antiviral drug, brincidofovir (CMX001 or Tembexa), is approved for smallpox in the United States, but not in Europe, according to Dr. Català. (In the United States, no treatment is specifically approved for treating monkeypox, but antivirals developed for smallpox “may prove beneficial against monkeypox,” according to the CDC.)
But she advised weighing the risks and benefits of using either drug in any individual patient.
The data suggest that the risk of viral shedding persists until the late stages of the disease trajectory. “A person is considered infectious from the onset of clinical manifestations until all skin lesions have scabbed over and re-epithelization has occurred,” Dr. Català said.
The prolonged period of contagion might be one reason to expect monkeypox to be transmitted more generally than it is now, according to Boghuma K. Titanji, MD, PhD, assistant professor of infectious diseases, Emory University, Atlanta.
“The longer the outbreak persists, the more likely we will see cases reported in groups other than MSM who have been most affected so far,” said Dr. Titanji, the first author of a recently published review article on monkeypox in Open Forum Infectious Diseases.
However, he acknowledged that a COVID-like spread is not expected. “The spread of monkeypox requires close and prolonged contact and is generally inefficient via fomites and droplet modes of transmission,” Dr. Titanji said in an interview. “Spread in heterosexual networks and congregate settings like crowded jails where close contact is unavoidable remains a concern that we need to educate the public about and maintain a high level of vigilance for.”
Dr. Català and Dr. Titanji report no potential conflicts of interest.
AT THE EADV CONGRESS
Alopecia areata: Positive results reported for two investigational JAK inhibitors
in separate studies reported at the annual congress of the European Academy of Dermatology and Venereology.
In the THRIVE-AA1 study, the primary endpoint of a Severity of Alopecia Tool (SALT) score of 20 or lower –which indicates that hair regrowth has occurred on at least 80% of the scalp – was achieved among patients taking deuruxolitinib, which was a significantly higher proportion than with placebo (P < .0001). Importantly, the JAK inhibitor’s effects were seen in as early as 4 weeks, and there was significant improvement in both eyelash and eyebrow hair regrowth.
In the unrelated ALLEGRO-LT study, effects from treatment with the JAK inhibitor ritlecitinib appeared to be sustained for 2 years; 69.6% of patients treated with ritlecitinib had a SALT score of 20 or lower by 24 months.
These data are “very exciting for alopecia areata because the patients selected are very severe,” observed Mahtab Samimi, MD, PhD, who cochaired the late-breaking session in which the study findings were discussed.
THRIVE-AA1 included only patients with hair loss of 50% or more. The ALLEGRO-LT study included patients with total scalp or total body hair loss (areata totalis/areata universalis) of 25%-50% at enrollment.
Moreover, “very stringent criteria” were used. SALT scores of 10 or less were evaluated in both studies, observed Dr. Samimi, professor of dermatology at the University of Tours (France).
“We can be ambitious now for our patients with alopecia areata; that’s really good news,” Dr. Samimi added.
Deuruxolitinib and the THRIVE trials
Deuruxolitinib is an oral JAK1/JAK2 inhibitor that has been tested in two similarly designed, multinational, randomized, double-blind, placebo-controlled phase 3 trials in patients with AA, THRIVE-AA1 and THRIVE-AA2.
Two doses of deuruxolitinib, 8 mg and 12 mg given twice daily, were evaluated in the trials, which altogether included just over 1,200 patients.
Results of THRIVE-AA1 have been reported by the manufacturer. Brett King, MD, PhD, associate professor of dermatology, Yale University, New Haven, Conn., presented a more comprehensive review at the EADV meeting.
He reported that at 24 weeks, SALT scores of 20 or lower were achieved by 30% of adults with AA who were treated with deuruxolitinib 8 mg and by 42% of those treated with deuruxolitinib 12 mg. This primary endpoint was seen in only 1% of the placebo-treated patients.
The more stringent endpoint of having a SALT score of 10 or less, which indicates that hair regrowth has occurred over 90% of the scalp, was met by 21% of patients who received deuruxolitinib 8 mg twice a day and by 35% of those who received the 12-mg dose twice a day at 24 weeks. This endpoint was not reached by any of the placebo-treated patients.
“This is truly transformative therapy,” Dr. King said when presenting the findings. “We know that the chances of spontaneous remission when you have severe disease is next to zero,” he added.
There were reasonably high rates of patient satisfaction with the treatment, according to Dr. King. He said that 42% of those who took 8 mg twice a day and 53% of those who took 12 mg twice a day said they were “very satisfied” or “satisfied” with the degree of scalp hair regrowth achieved, compared with 5% for placebo.
Safety was as expected, and there were no signs of any blood clots, said Dr. King. Common treatment-emergent adverse events (TEAEs) that affected 5% or more of patients included acne and headache. Serious TEAEs were reported by 1.1% and 0.5% of those taking the 8-mg and 12-mg twice-daily doses, respectively, compared with 2.9% of those who received placebo.
Overall, the results look promising for deuruxolitinib, he added. He noted that almost all patients included in the trial have opted to continue in the open-label long-term safety study.
Prescribing information of the JAK inhibitors approved by the U.S. Food and Drug Administration includes a boxed warning about risk of serious infections, mortality, malignancy, major adverse cardiovascular events (MACE), and thrombosis. The warning is based on experience with another JAK inhibitor for patients with rheumatoid arthritis.
Ritlecitinib and the ALLEGRO studies
Interim results of the ongoing, open-label, phase 3 ALLEGRO-LT study with ritlecitinib were presented separately by Athanasios Tsianakas, MD, head of the department of dermatology at Fachklinik Bad Bentheim, Germany.
Ritlecitinib, which targets JAK3 and also the TEC family of tyrosine kinases, had met all of its endpoints in the prior ALLEGRO Phase 2b/3 study, Dr. Tsianakas said. Those included the benchmarks of a SALT score of 20 or less and a SALT score of 10 or less.
“Ritlecitinib showed a very good long-term efficacy and good safety profile in our adolescent and adult patients suffering from alopecia areata,” said Dr. Tsianakas.
A total of 447 patients were included in the trial. They were treated with 50 mg of ritlecitinib every day; some had already participated in the ALLEGRO trial, while others had been newly recruited. The latter group entered the trial after a 4-week run-in period, during which a 200-mg daily loading dose was given for 4 weeks.
Most (86%) patients had been exposed to ritlecitinib for at least 12 months; one-fifth had discontinued treatment at the data cutoff, generally because the patients no longer met the eligibility criteria for the trial.
Safety was paramount, Dr. Tsianakas highlighted. There were few adverse events that led to temporary or permanent discontinuation of the study drug. The most common TEAEs that affected 5% or more of patients included headache and acne. There were two cases of MACE (one nonfatal myocardial infarction and one nonfatal stroke).
The proportion of patients with a SALT score of 20 or less was 2.5% at 1 month, 27.9% at 3 months, 50.1% at 6 months, 59.8% at 9 months, and 65.5% at 12 months. Thereafter, there was little shift in the response. A sustained effect, in which a SALT score of 20 or less was seen out to 24 months, occurred in 69.9% of patients.
A similar pattern was seen for SALT scores of 10 or less, ranging from 16.5% at 3 months to 62.5% at 24 months.
Following in baricitinib’s footsteps?
This not the first time that JAK inhibitors have been shown to have beneficial effects for patients with AA. Baricitinib (Olumiant) recently became the first JAK inhibitor to be granted marketing approval for AA in the United States, largely on the basis of two pivotal phase 3 studies, BRAVE-AA1 and BRAVE-AA2.
“This is just such an incredibly exciting time,” said Dr. King. “Our discoveries in the lab are being translated into effective therapies for patients with diseases for which we’ve not previously had therapies,” he commented.
“Our concept of interferon gamma– and interleukin-15–mediated disease is probably not true for everybody,” said, Dr. King, who acknowledged that some patients with AA do not respond to JAK-inhibitor therapy or may need additional or alternative treatment.
“It’s probably not that homogeneous a disease,” he added. “It’s fascinating that the very first drugs for this disease are showing efficacy in as many patients as they are.”
The THRIVE-AAI study was funded by CONCERT Pharmaceuticals. Dr. King has served on advisory boards, has provided consulting services to, or has been a trial investigator for multiple pharmaceutical companies, including CoNCERT Pharmaceuticals. The ALLEGRO-LT study was funded by Pfizer. Dr. Tsianakas has acted as a clinical trial investigator and speaker for Pfizer.
A version of this article first appeared on Medscape.com.
in separate studies reported at the annual congress of the European Academy of Dermatology and Venereology.
In the THRIVE-AA1 study, the primary endpoint of a Severity of Alopecia Tool (SALT) score of 20 or lower –which indicates that hair regrowth has occurred on at least 80% of the scalp – was achieved among patients taking deuruxolitinib, which was a significantly higher proportion than with placebo (P < .0001). Importantly, the JAK inhibitor’s effects were seen in as early as 4 weeks, and there was significant improvement in both eyelash and eyebrow hair regrowth.
In the unrelated ALLEGRO-LT study, effects from treatment with the JAK inhibitor ritlecitinib appeared to be sustained for 2 years; 69.6% of patients treated with ritlecitinib had a SALT score of 20 or lower by 24 months.
These data are “very exciting for alopecia areata because the patients selected are very severe,” observed Mahtab Samimi, MD, PhD, who cochaired the late-breaking session in which the study findings were discussed.
THRIVE-AA1 included only patients with hair loss of 50% or more. The ALLEGRO-LT study included patients with total scalp or total body hair loss (areata totalis/areata universalis) of 25%-50% at enrollment.
Moreover, “very stringent criteria” were used. SALT scores of 10 or less were evaluated in both studies, observed Dr. Samimi, professor of dermatology at the University of Tours (France).
“We can be ambitious now for our patients with alopecia areata; that’s really good news,” Dr. Samimi added.
Deuruxolitinib and the THRIVE trials
Deuruxolitinib is an oral JAK1/JAK2 inhibitor that has been tested in two similarly designed, multinational, randomized, double-blind, placebo-controlled phase 3 trials in patients with AA, THRIVE-AA1 and THRIVE-AA2.
Two doses of deuruxolitinib, 8 mg and 12 mg given twice daily, were evaluated in the trials, which altogether included just over 1,200 patients.
Results of THRIVE-AA1 have been reported by the manufacturer. Brett King, MD, PhD, associate professor of dermatology, Yale University, New Haven, Conn., presented a more comprehensive review at the EADV meeting.
He reported that at 24 weeks, SALT scores of 20 or lower were achieved by 30% of adults with AA who were treated with deuruxolitinib 8 mg and by 42% of those treated with deuruxolitinib 12 mg. This primary endpoint was seen in only 1% of the placebo-treated patients.
The more stringent endpoint of having a SALT score of 10 or less, which indicates that hair regrowth has occurred over 90% of the scalp, was met by 21% of patients who received deuruxolitinib 8 mg twice a day and by 35% of those who received the 12-mg dose twice a day at 24 weeks. This endpoint was not reached by any of the placebo-treated patients.
“This is truly transformative therapy,” Dr. King said when presenting the findings. “We know that the chances of spontaneous remission when you have severe disease is next to zero,” he added.
There were reasonably high rates of patient satisfaction with the treatment, according to Dr. King. He said that 42% of those who took 8 mg twice a day and 53% of those who took 12 mg twice a day said they were “very satisfied” or “satisfied” with the degree of scalp hair regrowth achieved, compared with 5% for placebo.
Safety was as expected, and there were no signs of any blood clots, said Dr. King. Common treatment-emergent adverse events (TEAEs) that affected 5% or more of patients included acne and headache. Serious TEAEs were reported by 1.1% and 0.5% of those taking the 8-mg and 12-mg twice-daily doses, respectively, compared with 2.9% of those who received placebo.
Overall, the results look promising for deuruxolitinib, he added. He noted that almost all patients included in the trial have opted to continue in the open-label long-term safety study.
Prescribing information of the JAK inhibitors approved by the U.S. Food and Drug Administration includes a boxed warning about risk of serious infections, mortality, malignancy, major adverse cardiovascular events (MACE), and thrombosis. The warning is based on experience with another JAK inhibitor for patients with rheumatoid arthritis.
Ritlecitinib and the ALLEGRO studies
Interim results of the ongoing, open-label, phase 3 ALLEGRO-LT study with ritlecitinib were presented separately by Athanasios Tsianakas, MD, head of the department of dermatology at Fachklinik Bad Bentheim, Germany.
Ritlecitinib, which targets JAK3 and also the TEC family of tyrosine kinases, had met all of its endpoints in the prior ALLEGRO Phase 2b/3 study, Dr. Tsianakas said. Those included the benchmarks of a SALT score of 20 or less and a SALT score of 10 or less.
“Ritlecitinib showed a very good long-term efficacy and good safety profile in our adolescent and adult patients suffering from alopecia areata,” said Dr. Tsianakas.
A total of 447 patients were included in the trial. They were treated with 50 mg of ritlecitinib every day; some had already participated in the ALLEGRO trial, while others had been newly recruited. The latter group entered the trial after a 4-week run-in period, during which a 200-mg daily loading dose was given for 4 weeks.
Most (86%) patients had been exposed to ritlecitinib for at least 12 months; one-fifth had discontinued treatment at the data cutoff, generally because the patients no longer met the eligibility criteria for the trial.
Safety was paramount, Dr. Tsianakas highlighted. There were few adverse events that led to temporary or permanent discontinuation of the study drug. The most common TEAEs that affected 5% or more of patients included headache and acne. There were two cases of MACE (one nonfatal myocardial infarction and one nonfatal stroke).
The proportion of patients with a SALT score of 20 or less was 2.5% at 1 month, 27.9% at 3 months, 50.1% at 6 months, 59.8% at 9 months, and 65.5% at 12 months. Thereafter, there was little shift in the response. A sustained effect, in which a SALT score of 20 or less was seen out to 24 months, occurred in 69.9% of patients.
A similar pattern was seen for SALT scores of 10 or less, ranging from 16.5% at 3 months to 62.5% at 24 months.
Following in baricitinib’s footsteps?
This not the first time that JAK inhibitors have been shown to have beneficial effects for patients with AA. Baricitinib (Olumiant) recently became the first JAK inhibitor to be granted marketing approval for AA in the United States, largely on the basis of two pivotal phase 3 studies, BRAVE-AA1 and BRAVE-AA2.
“This is just such an incredibly exciting time,” said Dr. King. “Our discoveries in the lab are being translated into effective therapies for patients with diseases for which we’ve not previously had therapies,” he commented.
“Our concept of interferon gamma– and interleukin-15–mediated disease is probably not true for everybody,” said, Dr. King, who acknowledged that some patients with AA do not respond to JAK-inhibitor therapy or may need additional or alternative treatment.
“It’s probably not that homogeneous a disease,” he added. “It’s fascinating that the very first drugs for this disease are showing efficacy in as many patients as they are.”
The THRIVE-AAI study was funded by CONCERT Pharmaceuticals. Dr. King has served on advisory boards, has provided consulting services to, or has been a trial investigator for multiple pharmaceutical companies, including CoNCERT Pharmaceuticals. The ALLEGRO-LT study was funded by Pfizer. Dr. Tsianakas has acted as a clinical trial investigator and speaker for Pfizer.
A version of this article first appeared on Medscape.com.
in separate studies reported at the annual congress of the European Academy of Dermatology and Venereology.
In the THRIVE-AA1 study, the primary endpoint of a Severity of Alopecia Tool (SALT) score of 20 or lower –which indicates that hair regrowth has occurred on at least 80% of the scalp – was achieved among patients taking deuruxolitinib, which was a significantly higher proportion than with placebo (P < .0001). Importantly, the JAK inhibitor’s effects were seen in as early as 4 weeks, and there was significant improvement in both eyelash and eyebrow hair regrowth.
In the unrelated ALLEGRO-LT study, effects from treatment with the JAK inhibitor ritlecitinib appeared to be sustained for 2 years; 69.6% of patients treated with ritlecitinib had a SALT score of 20 or lower by 24 months.
These data are “very exciting for alopecia areata because the patients selected are very severe,” observed Mahtab Samimi, MD, PhD, who cochaired the late-breaking session in which the study findings were discussed.
THRIVE-AA1 included only patients with hair loss of 50% or more. The ALLEGRO-LT study included patients with total scalp or total body hair loss (areata totalis/areata universalis) of 25%-50% at enrollment.
Moreover, “very stringent criteria” were used. SALT scores of 10 or less were evaluated in both studies, observed Dr. Samimi, professor of dermatology at the University of Tours (France).
“We can be ambitious now for our patients with alopecia areata; that’s really good news,” Dr. Samimi added.
Deuruxolitinib and the THRIVE trials
Deuruxolitinib is an oral JAK1/JAK2 inhibitor that has been tested in two similarly designed, multinational, randomized, double-blind, placebo-controlled phase 3 trials in patients with AA, THRIVE-AA1 and THRIVE-AA2.
Two doses of deuruxolitinib, 8 mg and 12 mg given twice daily, were evaluated in the trials, which altogether included just over 1,200 patients.
Results of THRIVE-AA1 have been reported by the manufacturer. Brett King, MD, PhD, associate professor of dermatology, Yale University, New Haven, Conn., presented a more comprehensive review at the EADV meeting.
He reported that at 24 weeks, SALT scores of 20 or lower were achieved by 30% of adults with AA who were treated with deuruxolitinib 8 mg and by 42% of those treated with deuruxolitinib 12 mg. This primary endpoint was seen in only 1% of the placebo-treated patients.
The more stringent endpoint of having a SALT score of 10 or less, which indicates that hair regrowth has occurred over 90% of the scalp, was met by 21% of patients who received deuruxolitinib 8 mg twice a day and by 35% of those who received the 12-mg dose twice a day at 24 weeks. This endpoint was not reached by any of the placebo-treated patients.
“This is truly transformative therapy,” Dr. King said when presenting the findings. “We know that the chances of spontaneous remission when you have severe disease is next to zero,” he added.
There were reasonably high rates of patient satisfaction with the treatment, according to Dr. King. He said that 42% of those who took 8 mg twice a day and 53% of those who took 12 mg twice a day said they were “very satisfied” or “satisfied” with the degree of scalp hair regrowth achieved, compared with 5% for placebo.
Safety was as expected, and there were no signs of any blood clots, said Dr. King. Common treatment-emergent adverse events (TEAEs) that affected 5% or more of patients included acne and headache. Serious TEAEs were reported by 1.1% and 0.5% of those taking the 8-mg and 12-mg twice-daily doses, respectively, compared with 2.9% of those who received placebo.
Overall, the results look promising for deuruxolitinib, he added. He noted that almost all patients included in the trial have opted to continue in the open-label long-term safety study.
Prescribing information of the JAK inhibitors approved by the U.S. Food and Drug Administration includes a boxed warning about risk of serious infections, mortality, malignancy, major adverse cardiovascular events (MACE), and thrombosis. The warning is based on experience with another JAK inhibitor for patients with rheumatoid arthritis.
Ritlecitinib and the ALLEGRO studies
Interim results of the ongoing, open-label, phase 3 ALLEGRO-LT study with ritlecitinib were presented separately by Athanasios Tsianakas, MD, head of the department of dermatology at Fachklinik Bad Bentheim, Germany.
Ritlecitinib, which targets JAK3 and also the TEC family of tyrosine kinases, had met all of its endpoints in the prior ALLEGRO Phase 2b/3 study, Dr. Tsianakas said. Those included the benchmarks of a SALT score of 20 or less and a SALT score of 10 or less.
“Ritlecitinib showed a very good long-term efficacy and good safety profile in our adolescent and adult patients suffering from alopecia areata,” said Dr. Tsianakas.
A total of 447 patients were included in the trial. They were treated with 50 mg of ritlecitinib every day; some had already participated in the ALLEGRO trial, while others had been newly recruited. The latter group entered the trial after a 4-week run-in period, during which a 200-mg daily loading dose was given for 4 weeks.
Most (86%) patients had been exposed to ritlecitinib for at least 12 months; one-fifth had discontinued treatment at the data cutoff, generally because the patients no longer met the eligibility criteria for the trial.
Safety was paramount, Dr. Tsianakas highlighted. There were few adverse events that led to temporary or permanent discontinuation of the study drug. The most common TEAEs that affected 5% or more of patients included headache and acne. There were two cases of MACE (one nonfatal myocardial infarction and one nonfatal stroke).
The proportion of patients with a SALT score of 20 or less was 2.5% at 1 month, 27.9% at 3 months, 50.1% at 6 months, 59.8% at 9 months, and 65.5% at 12 months. Thereafter, there was little shift in the response. A sustained effect, in which a SALT score of 20 or less was seen out to 24 months, occurred in 69.9% of patients.
A similar pattern was seen for SALT scores of 10 or less, ranging from 16.5% at 3 months to 62.5% at 24 months.
Following in baricitinib’s footsteps?
This not the first time that JAK inhibitors have been shown to have beneficial effects for patients with AA. Baricitinib (Olumiant) recently became the first JAK inhibitor to be granted marketing approval for AA in the United States, largely on the basis of two pivotal phase 3 studies, BRAVE-AA1 and BRAVE-AA2.
“This is just such an incredibly exciting time,” said Dr. King. “Our discoveries in the lab are being translated into effective therapies for patients with diseases for which we’ve not previously had therapies,” he commented.
“Our concept of interferon gamma– and interleukin-15–mediated disease is probably not true for everybody,” said, Dr. King, who acknowledged that some patients with AA do not respond to JAK-inhibitor therapy or may need additional or alternative treatment.
“It’s probably not that homogeneous a disease,” he added. “It’s fascinating that the very first drugs for this disease are showing efficacy in as many patients as they are.”
The THRIVE-AAI study was funded by CONCERT Pharmaceuticals. Dr. King has served on advisory boards, has provided consulting services to, or has been a trial investigator for multiple pharmaceutical companies, including CoNCERT Pharmaceuticals. The ALLEGRO-LT study was funded by Pfizer. Dr. Tsianakas has acted as a clinical trial investigator and speaker for Pfizer.
A version of this article first appeared on Medscape.com.
FROM THE EADV CONGRESS
Training program linked to less hand eczema for hairdressers
The study was conducted in Denmark, where about 40% of hairdressers develop occupational hand eczema (OHE), according to researchers. Hairdressers globally are exposed to wet work and myriad skin irritants and allergens, including dyes, permanent-wave solutions, persulfates, preservatives, and fragrances. The study, which was funded by the Danish hairdressers and beauticians union, was published in Contact Dermatitis.
Lead author Martin Havmose, BSc, of the National Allergy Research Center, department of dermatology and allergy, University of Copenhagen, Hellerup, Denmark, and colleagues wrote that prevention is critical, inasmuch as eczema can cut careers short and have lasting health effects.
Up to 70% of hairdressers experience some sort of work-related skin damage in their careers, as reported by this news organization.
Hand eczema also is common among hairdressers in the United States, Mark Denis Davis, MD, chair of dermatology at the Mayo Clinic in Rochester, Minn., told this news organization. It can be quite debilitating, itchy, and painful, he said.
“Often it is associated with painful fissuring, cracks in the skin, particularly involving the fingers. It may also be unsightly,” he said.
Dr. Davis said he hears anecdotally in his practice that many hairdressers are reluctant to wear gloves because of the touch and dexterity needed in their work.
The researchers evaluated the risk of OHE and compliance with skin protection measures among hairdressers who were trained before Denmark rolled out a nationwide skin protection program in hairdressing vocational schools in 2011.
Questionnaires were sent in May 2009 to all hairdressers (96.4% women; average age, 26) who had graduated from 1985 to 2007; in May 2020, questionnaires were sent to all hairdressers who had graduated from 2008 to 2018.
The average time worked in the trade was 8 years, and 28.8% no longer worked as hairdressers, data show.
The response rate was 66.6% (305/460) for the 2009 survey and 29.9% (363/1215) for the 2020 survey.
Prevalence of OHE dropped after program
The prevalence of OHE during career time dropped from 42.8% to 29% (adjusted odds ratio, 0.55; 95% confidence interval [CI], 0.40-0.77) between the two surveys.
In addition, the incidence rate of OHE decreased from 57.5 (95% CI, 48.4-68.4) to 42.0 (95% CI, 34.6-50.9) per 1,000 person-years (incidence rate ratio, 0.73; 95% CI, 0.560.95) in that period.
There was an increase in the use of gloves between the two surveys. There was more glove use when the hairdressers engaged in wet work and handled dyes, products with bleach, and permanent-wave solutions (P < .05).
The nationwide program educates hairdressing apprentices on contact allergy/urticaria, how to prevent occupational skin disease, and skin biology. Teaching materials focus on 11 recommendations, 7 of which are related to glove use.
“The lack of primary prevention of OHE in hairdressing vocational schools may be a missed opportunity in the prevention of the disease,” the authors concluded.
Dr. Davis said hairdressers with hand eczema should know that in the short term, topical corticosteroids can be used to decrease the inflammation of the skin.
He highlighted the following advice from the authors:
- Gloves should be used when washing, dyeing, bleaching, and creating perms.
- Disposable gloves should never be reused.
- Gloves should be used only as long as necessary.
- Rings should not be worn at work.
- Cotton gloves should be worn underneath protective gloves.
- For clients who are having their hair both cut and dyed, the hair should be cut before it is dyed.
- Nitrile gloves should be used without rubber accelerators.
“In the longer term,” said Dr. Davis, “the most important thing is to avoid exposure to the precipitating factors, such as wet work – working with water, which irritates the skin – and avoiding any allergens that are contributing to the eczema.”
The study was funded by an unrestricted grant from the Danish hairdressers and beauticians union. Two coauthors have links to industry, as listed in the original article. Dr. Davis reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The study was conducted in Denmark, where about 40% of hairdressers develop occupational hand eczema (OHE), according to researchers. Hairdressers globally are exposed to wet work and myriad skin irritants and allergens, including dyes, permanent-wave solutions, persulfates, preservatives, and fragrances. The study, which was funded by the Danish hairdressers and beauticians union, was published in Contact Dermatitis.
Lead author Martin Havmose, BSc, of the National Allergy Research Center, department of dermatology and allergy, University of Copenhagen, Hellerup, Denmark, and colleagues wrote that prevention is critical, inasmuch as eczema can cut careers short and have lasting health effects.
Up to 70% of hairdressers experience some sort of work-related skin damage in their careers, as reported by this news organization.
Hand eczema also is common among hairdressers in the United States, Mark Denis Davis, MD, chair of dermatology at the Mayo Clinic in Rochester, Minn., told this news organization. It can be quite debilitating, itchy, and painful, he said.
“Often it is associated with painful fissuring, cracks in the skin, particularly involving the fingers. It may also be unsightly,” he said.
Dr. Davis said he hears anecdotally in his practice that many hairdressers are reluctant to wear gloves because of the touch and dexterity needed in their work.
The researchers evaluated the risk of OHE and compliance with skin protection measures among hairdressers who were trained before Denmark rolled out a nationwide skin protection program in hairdressing vocational schools in 2011.
Questionnaires were sent in May 2009 to all hairdressers (96.4% women; average age, 26) who had graduated from 1985 to 2007; in May 2020, questionnaires were sent to all hairdressers who had graduated from 2008 to 2018.
The average time worked in the trade was 8 years, and 28.8% no longer worked as hairdressers, data show.
The response rate was 66.6% (305/460) for the 2009 survey and 29.9% (363/1215) for the 2020 survey.
Prevalence of OHE dropped after program
The prevalence of OHE during career time dropped from 42.8% to 29% (adjusted odds ratio, 0.55; 95% confidence interval [CI], 0.40-0.77) between the two surveys.
In addition, the incidence rate of OHE decreased from 57.5 (95% CI, 48.4-68.4) to 42.0 (95% CI, 34.6-50.9) per 1,000 person-years (incidence rate ratio, 0.73; 95% CI, 0.560.95) in that period.
There was an increase in the use of gloves between the two surveys. There was more glove use when the hairdressers engaged in wet work and handled dyes, products with bleach, and permanent-wave solutions (P < .05).
The nationwide program educates hairdressing apprentices on contact allergy/urticaria, how to prevent occupational skin disease, and skin biology. Teaching materials focus on 11 recommendations, 7 of which are related to glove use.
“The lack of primary prevention of OHE in hairdressing vocational schools may be a missed opportunity in the prevention of the disease,” the authors concluded.
Dr. Davis said hairdressers with hand eczema should know that in the short term, topical corticosteroids can be used to decrease the inflammation of the skin.
He highlighted the following advice from the authors:
- Gloves should be used when washing, dyeing, bleaching, and creating perms.
- Disposable gloves should never be reused.
- Gloves should be used only as long as necessary.
- Rings should not be worn at work.
- Cotton gloves should be worn underneath protective gloves.
- For clients who are having their hair both cut and dyed, the hair should be cut before it is dyed.
- Nitrile gloves should be used without rubber accelerators.
“In the longer term,” said Dr. Davis, “the most important thing is to avoid exposure to the precipitating factors, such as wet work – working with water, which irritates the skin – and avoiding any allergens that are contributing to the eczema.”
The study was funded by an unrestricted grant from the Danish hairdressers and beauticians union. Two coauthors have links to industry, as listed in the original article. Dr. Davis reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The study was conducted in Denmark, where about 40% of hairdressers develop occupational hand eczema (OHE), according to researchers. Hairdressers globally are exposed to wet work and myriad skin irritants and allergens, including dyes, permanent-wave solutions, persulfates, preservatives, and fragrances. The study, which was funded by the Danish hairdressers and beauticians union, was published in Contact Dermatitis.
Lead author Martin Havmose, BSc, of the National Allergy Research Center, department of dermatology and allergy, University of Copenhagen, Hellerup, Denmark, and colleagues wrote that prevention is critical, inasmuch as eczema can cut careers short and have lasting health effects.
Up to 70% of hairdressers experience some sort of work-related skin damage in their careers, as reported by this news organization.
Hand eczema also is common among hairdressers in the United States, Mark Denis Davis, MD, chair of dermatology at the Mayo Clinic in Rochester, Minn., told this news organization. It can be quite debilitating, itchy, and painful, he said.
“Often it is associated with painful fissuring, cracks in the skin, particularly involving the fingers. It may also be unsightly,” he said.
Dr. Davis said he hears anecdotally in his practice that many hairdressers are reluctant to wear gloves because of the touch and dexterity needed in their work.
The researchers evaluated the risk of OHE and compliance with skin protection measures among hairdressers who were trained before Denmark rolled out a nationwide skin protection program in hairdressing vocational schools in 2011.
Questionnaires were sent in May 2009 to all hairdressers (96.4% women; average age, 26) who had graduated from 1985 to 2007; in May 2020, questionnaires were sent to all hairdressers who had graduated from 2008 to 2018.
The average time worked in the trade was 8 years, and 28.8% no longer worked as hairdressers, data show.
The response rate was 66.6% (305/460) for the 2009 survey and 29.9% (363/1215) for the 2020 survey.
Prevalence of OHE dropped after program
The prevalence of OHE during career time dropped from 42.8% to 29% (adjusted odds ratio, 0.55; 95% confidence interval [CI], 0.40-0.77) between the two surveys.
In addition, the incidence rate of OHE decreased from 57.5 (95% CI, 48.4-68.4) to 42.0 (95% CI, 34.6-50.9) per 1,000 person-years (incidence rate ratio, 0.73; 95% CI, 0.560.95) in that period.
There was an increase in the use of gloves between the two surveys. There was more glove use when the hairdressers engaged in wet work and handled dyes, products with bleach, and permanent-wave solutions (P < .05).
The nationwide program educates hairdressing apprentices on contact allergy/urticaria, how to prevent occupational skin disease, and skin biology. Teaching materials focus on 11 recommendations, 7 of which are related to glove use.
“The lack of primary prevention of OHE in hairdressing vocational schools may be a missed opportunity in the prevention of the disease,” the authors concluded.
Dr. Davis said hairdressers with hand eczema should know that in the short term, topical corticosteroids can be used to decrease the inflammation of the skin.
He highlighted the following advice from the authors:
- Gloves should be used when washing, dyeing, bleaching, and creating perms.
- Disposable gloves should never be reused.
- Gloves should be used only as long as necessary.
- Rings should not be worn at work.
- Cotton gloves should be worn underneath protective gloves.
- For clients who are having their hair both cut and dyed, the hair should be cut before it is dyed.
- Nitrile gloves should be used without rubber accelerators.
“In the longer term,” said Dr. Davis, “the most important thing is to avoid exposure to the precipitating factors, such as wet work – working with water, which irritates the skin – and avoiding any allergens that are contributing to the eczema.”
The study was funded by an unrestricted grant from the Danish hairdressers and beauticians union. Two coauthors have links to industry, as listed in the original article. Dr. Davis reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CONTACT DERMATITIS
Study identifies skin biomarkers that predict newborn eczema risk
It might be possible to develop a simple test to identify newborn children who are at risk of later developing atopic dermatitis (AD), according to findings from a Danish prospective birth cohort study.
but also for having more severe disease.
“We are able to identify predictive immune biomarkers of atopic dermatitis using a noninvasive method that was not associated with any pain,” one of the study’s investigators, Anne-Sofie Halling, MD, said at a press briefing at the annual congress of the European Academy of Dermatology and Venereology.
“Importantly, we were able to predict atopic dermatitis occurring months after [sample] collection,” said Dr. Halling, who works at Bispebjerg Hospital and is a PhD student at the University of Copenhagen.
These findings could hopefully be used to help identify children “so that preventive strategies can target these children ... and decrease the incidence of this common disease,” she added.
AD is caused “by a complex interplay between skin barrier dysfunction and immune dysregulation,” Dr. Halling said, and it is “the first step in the so-called atopic march, where children also develop food allergy, asthma, and rhinitis.” Almost all cases of AD begin during the first years of life. Approximately 15%-20% of children can be affected, she noted, emphasizing the high burden of the disease and pointing out that strategies are shifting toward trying to prevent the disease in those at risk.
Copenhagen BABY cohort
This is where the BABY study comes in, Dr. Halling said. The study enrolled 450 children at birth and followed them until age 2 years. Gene mutation testing was performed at enrollment. All children underwent skin examination, and skin samples were taken using tape strips. Tape strips were applied to the back of the hand of children born at term and between the shoulder blades on the back of children who were premature.
Skin examinations were repeated, and skin samples were obtained again at age 2 months. They were taken again only if there were any signs of AD. For those diagnosed with AD, disease severity was assessed using the Eczema Area and Severity Index (EASI) by the treating physician. Children were excluded if they had AD at the time the tape strip testing was due to be performed.
Comparing term and preterm children
Dr. Halling noted that analyses were performed separately for the 300 children born at term and for the 150 who were preterm.
The prevalence of AD was higher among children born at term than among the preterm children (34.6% vs. 21.2%), and the median time to onset was shorter (6 months vs. 8 months). There were also differences in the EASI scores among those who developed AD; median scores were higher in the children born at term than in the preterm children (4.1 vs. 1.6).
More children born at term than preterm children had moderate to severe AD (23.3% vs. 8%), Dr. Halling reported.
TARC, IL-8, and IL-18 predictive of AD
Multiple immune biomarkers were tested, including various cytokines and filaggrin degradation products. On examination of skin samples collected at birth, no particular biomarkers were found at higher levels among children who developed AD in comparison with those who did not develop AD.
With regard to biomarkers examined in skin samples at 2 months of age, however, the results were different, Dr. Halling said. One particular cytokine, thymus and activation-regulated chemokine (TARC), was seen to double the risk of AD in the first 2 years of a child’s life.
This doubled risk was seen not only among the children born at term but also among those born preterm, although the data were only significant with regard to the children born at term.
The unadjusted hazard ratios and adjusted HRs (adjusted for parental atopy and filaggrin gene mutations) in term children were 2.11 (95% confidence interval, 1.36-3.26; P = .0008) and 1.85 (95% CI, 1.18-2.89; P = .007), respectively.
For preterm children, the HRs were 2.23 (95% CI, 0.85-5.86; P = .1) and 2.60 (95% CI, 0.98-6.85; P =.05), respectively.
These findings were in line with findings of other studies, Dr. Halling said. “It is well recognized that TARC is currently the best biomarker in patients with established atopic dermatitis.” Moreover, she reported that TARC was associated with a cumulative increase in the risk for AD and that levels were found to be higher in children in whom onset occurred at a later age than among those diagnosed before 6 months of age.
“This is important, as these findings shows that TARC levels predict atopic dermatitis that occurred many months later,” Dr. Halling said.
And, in term-born children at least, TARC upped the chances that the severity of AD would be greater than had it not been present (adjusted HR, 4.65; 95% CI, 1.91-11.31; P = .0007).
Increased levels of interleukin-8 (IL-8) and IL-18 at 2 months of age were also found to be predictive of having moderate to severe AD. The risk was more than double in comparison with those in whom levels were not increased, again only in term-born children.
‘Stimulating and interesting findings’
These data are “very stimulating and interesting,” Dedee Murrell, MD, professor and head of the department of dermatology at St. George Hospital, University of New South Wales, Sydney, observed at the press briefing.
“You found this significant association mainly in the newborn children born at term, and the association in the preterm babies wasn’t as high. Is that anything to do with how they were taken care of in the hospital?” Dr. Murrell asked.
“That’s a really good question,” Dr. Halling said. “Maybe they need to be exposed for a month or two before we are actually able to identify which children will develop atopic dermatitis.”
The study was funded by the Lundbeck Foundation. Dr. Halling has acted as a consultant for Coloplast and as a speaker for Leo Pharma. Dr. Murrell has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
It might be possible to develop a simple test to identify newborn children who are at risk of later developing atopic dermatitis (AD), according to findings from a Danish prospective birth cohort study.
but also for having more severe disease.
“We are able to identify predictive immune biomarkers of atopic dermatitis using a noninvasive method that was not associated with any pain,” one of the study’s investigators, Anne-Sofie Halling, MD, said at a press briefing at the annual congress of the European Academy of Dermatology and Venereology.
“Importantly, we were able to predict atopic dermatitis occurring months after [sample] collection,” said Dr. Halling, who works at Bispebjerg Hospital and is a PhD student at the University of Copenhagen.
These findings could hopefully be used to help identify children “so that preventive strategies can target these children ... and decrease the incidence of this common disease,” she added.
AD is caused “by a complex interplay between skin barrier dysfunction and immune dysregulation,” Dr. Halling said, and it is “the first step in the so-called atopic march, where children also develop food allergy, asthma, and rhinitis.” Almost all cases of AD begin during the first years of life. Approximately 15%-20% of children can be affected, she noted, emphasizing the high burden of the disease and pointing out that strategies are shifting toward trying to prevent the disease in those at risk.
Copenhagen BABY cohort
This is where the BABY study comes in, Dr. Halling said. The study enrolled 450 children at birth and followed them until age 2 years. Gene mutation testing was performed at enrollment. All children underwent skin examination, and skin samples were taken using tape strips. Tape strips were applied to the back of the hand of children born at term and between the shoulder blades on the back of children who were premature.
Skin examinations were repeated, and skin samples were obtained again at age 2 months. They were taken again only if there were any signs of AD. For those diagnosed with AD, disease severity was assessed using the Eczema Area and Severity Index (EASI) by the treating physician. Children were excluded if they had AD at the time the tape strip testing was due to be performed.
Comparing term and preterm children
Dr. Halling noted that analyses were performed separately for the 300 children born at term and for the 150 who were preterm.
The prevalence of AD was higher among children born at term than among the preterm children (34.6% vs. 21.2%), and the median time to onset was shorter (6 months vs. 8 months). There were also differences in the EASI scores among those who developed AD; median scores were higher in the children born at term than in the preterm children (4.1 vs. 1.6).
More children born at term than preterm children had moderate to severe AD (23.3% vs. 8%), Dr. Halling reported.
TARC, IL-8, and IL-18 predictive of AD
Multiple immune biomarkers were tested, including various cytokines and filaggrin degradation products. On examination of skin samples collected at birth, no particular biomarkers were found at higher levels among children who developed AD in comparison with those who did not develop AD.
With regard to biomarkers examined in skin samples at 2 months of age, however, the results were different, Dr. Halling said. One particular cytokine, thymus and activation-regulated chemokine (TARC), was seen to double the risk of AD in the first 2 years of a child’s life.
This doubled risk was seen not only among the children born at term but also among those born preterm, although the data were only significant with regard to the children born at term.
The unadjusted hazard ratios and adjusted HRs (adjusted for parental atopy and filaggrin gene mutations) in term children were 2.11 (95% confidence interval, 1.36-3.26; P = .0008) and 1.85 (95% CI, 1.18-2.89; P = .007), respectively.
For preterm children, the HRs were 2.23 (95% CI, 0.85-5.86; P = .1) and 2.60 (95% CI, 0.98-6.85; P =.05), respectively.
These findings were in line with findings of other studies, Dr. Halling said. “It is well recognized that TARC is currently the best biomarker in patients with established atopic dermatitis.” Moreover, she reported that TARC was associated with a cumulative increase in the risk for AD and that levels were found to be higher in children in whom onset occurred at a later age than among those diagnosed before 6 months of age.
“This is important, as these findings shows that TARC levels predict atopic dermatitis that occurred many months later,” Dr. Halling said.
And, in term-born children at least, TARC upped the chances that the severity of AD would be greater than had it not been present (adjusted HR, 4.65; 95% CI, 1.91-11.31; P = .0007).
Increased levels of interleukin-8 (IL-8) and IL-18 at 2 months of age were also found to be predictive of having moderate to severe AD. The risk was more than double in comparison with those in whom levels were not increased, again only in term-born children.
‘Stimulating and interesting findings’
These data are “very stimulating and interesting,” Dedee Murrell, MD, professor and head of the department of dermatology at St. George Hospital, University of New South Wales, Sydney, observed at the press briefing.
“You found this significant association mainly in the newborn children born at term, and the association in the preterm babies wasn’t as high. Is that anything to do with how they were taken care of in the hospital?” Dr. Murrell asked.
“That’s a really good question,” Dr. Halling said. “Maybe they need to be exposed for a month or two before we are actually able to identify which children will develop atopic dermatitis.”
The study was funded by the Lundbeck Foundation. Dr. Halling has acted as a consultant for Coloplast and as a speaker for Leo Pharma. Dr. Murrell has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
It might be possible to develop a simple test to identify newborn children who are at risk of later developing atopic dermatitis (AD), according to findings from a Danish prospective birth cohort study.
but also for having more severe disease.
“We are able to identify predictive immune biomarkers of atopic dermatitis using a noninvasive method that was not associated with any pain,” one of the study’s investigators, Anne-Sofie Halling, MD, said at a press briefing at the annual congress of the European Academy of Dermatology and Venereology.
“Importantly, we were able to predict atopic dermatitis occurring months after [sample] collection,” said Dr. Halling, who works at Bispebjerg Hospital and is a PhD student at the University of Copenhagen.
These findings could hopefully be used to help identify children “so that preventive strategies can target these children ... and decrease the incidence of this common disease,” she added.
AD is caused “by a complex interplay between skin barrier dysfunction and immune dysregulation,” Dr. Halling said, and it is “the first step in the so-called atopic march, where children also develop food allergy, asthma, and rhinitis.” Almost all cases of AD begin during the first years of life. Approximately 15%-20% of children can be affected, she noted, emphasizing the high burden of the disease and pointing out that strategies are shifting toward trying to prevent the disease in those at risk.
Copenhagen BABY cohort
This is where the BABY study comes in, Dr. Halling said. The study enrolled 450 children at birth and followed them until age 2 years. Gene mutation testing was performed at enrollment. All children underwent skin examination, and skin samples were taken using tape strips. Tape strips were applied to the back of the hand of children born at term and between the shoulder blades on the back of children who were premature.
Skin examinations were repeated, and skin samples were obtained again at age 2 months. They were taken again only if there were any signs of AD. For those diagnosed with AD, disease severity was assessed using the Eczema Area and Severity Index (EASI) by the treating physician. Children were excluded if they had AD at the time the tape strip testing was due to be performed.
Comparing term and preterm children
Dr. Halling noted that analyses were performed separately for the 300 children born at term and for the 150 who were preterm.
The prevalence of AD was higher among children born at term than among the preterm children (34.6% vs. 21.2%), and the median time to onset was shorter (6 months vs. 8 months). There were also differences in the EASI scores among those who developed AD; median scores were higher in the children born at term than in the preterm children (4.1 vs. 1.6).
More children born at term than preterm children had moderate to severe AD (23.3% vs. 8%), Dr. Halling reported.
TARC, IL-8, and IL-18 predictive of AD
Multiple immune biomarkers were tested, including various cytokines and filaggrin degradation products. On examination of skin samples collected at birth, no particular biomarkers were found at higher levels among children who developed AD in comparison with those who did not develop AD.
With regard to biomarkers examined in skin samples at 2 months of age, however, the results were different, Dr. Halling said. One particular cytokine, thymus and activation-regulated chemokine (TARC), was seen to double the risk of AD in the first 2 years of a child’s life.
This doubled risk was seen not only among the children born at term but also among those born preterm, although the data were only significant with regard to the children born at term.
The unadjusted hazard ratios and adjusted HRs (adjusted for parental atopy and filaggrin gene mutations) in term children were 2.11 (95% confidence interval, 1.36-3.26; P = .0008) and 1.85 (95% CI, 1.18-2.89; P = .007), respectively.
For preterm children, the HRs were 2.23 (95% CI, 0.85-5.86; P = .1) and 2.60 (95% CI, 0.98-6.85; P =.05), respectively.
These findings were in line with findings of other studies, Dr. Halling said. “It is well recognized that TARC is currently the best biomarker in patients with established atopic dermatitis.” Moreover, she reported that TARC was associated with a cumulative increase in the risk for AD and that levels were found to be higher in children in whom onset occurred at a later age than among those diagnosed before 6 months of age.
“This is important, as these findings shows that TARC levels predict atopic dermatitis that occurred many months later,” Dr. Halling said.
And, in term-born children at least, TARC upped the chances that the severity of AD would be greater than had it not been present (adjusted HR, 4.65; 95% CI, 1.91-11.31; P = .0007).
Increased levels of interleukin-8 (IL-8) and IL-18 at 2 months of age were also found to be predictive of having moderate to severe AD. The risk was more than double in comparison with those in whom levels were not increased, again only in term-born children.
‘Stimulating and interesting findings’
These data are “very stimulating and interesting,” Dedee Murrell, MD, professor and head of the department of dermatology at St. George Hospital, University of New South Wales, Sydney, observed at the press briefing.
“You found this significant association mainly in the newborn children born at term, and the association in the preterm babies wasn’t as high. Is that anything to do with how they were taken care of in the hospital?” Dr. Murrell asked.
“That’s a really good question,” Dr. Halling said. “Maybe they need to be exposed for a month or two before we are actually able to identify which children will develop atopic dermatitis.”
The study was funded by the Lundbeck Foundation. Dr. Halling has acted as a consultant for Coloplast and as a speaker for Leo Pharma. Dr. Murrell has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE EADV CONGRESS
FDA approves dupilumab for treatment of prurigo nodularis
The according to a press release from the manufacturers.
Recent studies of dupilumab (Dupixent), which inhibits the signaling of the interleukin-4 and IL-13 pathways, show significant improvements in both itchiness and lesion counts, compared with placebo, in adults with prurigo nodularis (PN).
Approval was based on data from two randomized, controlled trials, PRIME and PRIME2, comparing dupilumab with placebo in 311 adults with uncontrolled PN, according to the release issued by Regeneron and Sanofi. Dupilumab is administered via a 300 mg subcutaneous injection every 2 weeks after a loading dose.
The primary endpoint in PRIME and PRIME 2 was a clinically meaningful improvement in itch from baseline as measured by at least a 4-point reduction in the Worst Itch Numeric Rating Scale, a 0-10 scale, at 24 and 12 weeks, respectively. In the studies, 60% and 58% of patients treated with dupilumab met the primary endpoint at 24 weeks, compared with 18% and 20% of those on placebo. At 24 weeks, 48% and 45% of patients on dupilumab achieved clear or almost clear skin, another study endpoint, compared with 18% and 16% among those on placebo.*
In PRIME and PRIME2, 44% and 37% of patients on dupilumab met the primary endpoint at 12 weeks versus16% and 22% among those on placebo.
Safety profiles were similar to those seen in other dupilumab studies, according to the release. The most common adverse events in the two studies combined were nasopharyngitis, reported in 5% of those on dupilumab versus 2% of those on placebo; conjunctivitis in 4% versus 1%; herpes infection in 3% versus 0; dizziness in 3% vs. 1%; muscle pain in 3% versus 1%; and diarrhea in 3% versus 1%.
Phase 3 data on dupilumab for PN were recently presented at the annual congress of the European Academy of Dermatology and Venereology.
A regulatory submission for dupilumab for treating PN is in progress at the European Medicines Agency, and submissions are planned to regulatory agencies in additional countries later in 2022, according to the company press release.
Dupilumab is currently approved in the United States for atopic dermatitis in children aged 6 months and older and adults with moderate to severe atopic dermatitis and in children and adults aged 6 years and older with moderate to severe eosinophilic or oral steroid-dependent asthma, as well as for the treatment of chronic rhinosinusitis with nasal polyposis in adults, and for the treatment of eosinophilic esophagitis in adults and children aged 12 years and older, weighing at least 40 kg. Dupilumab is under clinical development for the treatment of chronic spontaneous urticaria and bullous pemphigoid, according to the manufacturers.
The studies were supported by Regeneron and Sanofi.
A version of this article first appeared on Medscape.com.
*Correction, 9/30/22: An earlier version of this article misstated results of one endpoint.
The according to a press release from the manufacturers.
Recent studies of dupilumab (Dupixent), which inhibits the signaling of the interleukin-4 and IL-13 pathways, show significant improvements in both itchiness and lesion counts, compared with placebo, in adults with prurigo nodularis (PN).
Approval was based on data from two randomized, controlled trials, PRIME and PRIME2, comparing dupilumab with placebo in 311 adults with uncontrolled PN, according to the release issued by Regeneron and Sanofi. Dupilumab is administered via a 300 mg subcutaneous injection every 2 weeks after a loading dose.
The primary endpoint in PRIME and PRIME 2 was a clinically meaningful improvement in itch from baseline as measured by at least a 4-point reduction in the Worst Itch Numeric Rating Scale, a 0-10 scale, at 24 and 12 weeks, respectively. In the studies, 60% and 58% of patients treated with dupilumab met the primary endpoint at 24 weeks, compared with 18% and 20% of those on placebo. At 24 weeks, 48% and 45% of patients on dupilumab achieved clear or almost clear skin, another study endpoint, compared with 18% and 16% among those on placebo.*
In PRIME and PRIME2, 44% and 37% of patients on dupilumab met the primary endpoint at 12 weeks versus16% and 22% among those on placebo.
Safety profiles were similar to those seen in other dupilumab studies, according to the release. The most common adverse events in the two studies combined were nasopharyngitis, reported in 5% of those on dupilumab versus 2% of those on placebo; conjunctivitis in 4% versus 1%; herpes infection in 3% versus 0; dizziness in 3% vs. 1%; muscle pain in 3% versus 1%; and diarrhea in 3% versus 1%.
Phase 3 data on dupilumab for PN were recently presented at the annual congress of the European Academy of Dermatology and Venereology.
A regulatory submission for dupilumab for treating PN is in progress at the European Medicines Agency, and submissions are planned to regulatory agencies in additional countries later in 2022, according to the company press release.
Dupilumab is currently approved in the United States for atopic dermatitis in children aged 6 months and older and adults with moderate to severe atopic dermatitis and in children and adults aged 6 years and older with moderate to severe eosinophilic or oral steroid-dependent asthma, as well as for the treatment of chronic rhinosinusitis with nasal polyposis in adults, and for the treatment of eosinophilic esophagitis in adults and children aged 12 years and older, weighing at least 40 kg. Dupilumab is under clinical development for the treatment of chronic spontaneous urticaria and bullous pemphigoid, according to the manufacturers.
The studies were supported by Regeneron and Sanofi.
A version of this article first appeared on Medscape.com.
*Correction, 9/30/22: An earlier version of this article misstated results of one endpoint.
The according to a press release from the manufacturers.
Recent studies of dupilumab (Dupixent), which inhibits the signaling of the interleukin-4 and IL-13 pathways, show significant improvements in both itchiness and lesion counts, compared with placebo, in adults with prurigo nodularis (PN).
Approval was based on data from two randomized, controlled trials, PRIME and PRIME2, comparing dupilumab with placebo in 311 adults with uncontrolled PN, according to the release issued by Regeneron and Sanofi. Dupilumab is administered via a 300 mg subcutaneous injection every 2 weeks after a loading dose.
The primary endpoint in PRIME and PRIME 2 was a clinically meaningful improvement in itch from baseline as measured by at least a 4-point reduction in the Worst Itch Numeric Rating Scale, a 0-10 scale, at 24 and 12 weeks, respectively. In the studies, 60% and 58% of patients treated with dupilumab met the primary endpoint at 24 weeks, compared with 18% and 20% of those on placebo. At 24 weeks, 48% and 45% of patients on dupilumab achieved clear or almost clear skin, another study endpoint, compared with 18% and 16% among those on placebo.*
In PRIME and PRIME2, 44% and 37% of patients on dupilumab met the primary endpoint at 12 weeks versus16% and 22% among those on placebo.
Safety profiles were similar to those seen in other dupilumab studies, according to the release. The most common adverse events in the two studies combined were nasopharyngitis, reported in 5% of those on dupilumab versus 2% of those on placebo; conjunctivitis in 4% versus 1%; herpes infection in 3% versus 0; dizziness in 3% vs. 1%; muscle pain in 3% versus 1%; and diarrhea in 3% versus 1%.
Phase 3 data on dupilumab for PN were recently presented at the annual congress of the European Academy of Dermatology and Venereology.
A regulatory submission for dupilumab for treating PN is in progress at the European Medicines Agency, and submissions are planned to regulatory agencies in additional countries later in 2022, according to the company press release.
Dupilumab is currently approved in the United States for atopic dermatitis in children aged 6 months and older and adults with moderate to severe atopic dermatitis and in children and adults aged 6 years and older with moderate to severe eosinophilic or oral steroid-dependent asthma, as well as for the treatment of chronic rhinosinusitis with nasal polyposis in adults, and for the treatment of eosinophilic esophagitis in adults and children aged 12 years and older, weighing at least 40 kg. Dupilumab is under clinical development for the treatment of chronic spontaneous urticaria and bullous pemphigoid, according to the manufacturers.
The studies were supported by Regeneron and Sanofi.
A version of this article first appeared on Medscape.com.
*Correction, 9/30/22: An earlier version of this article misstated results of one endpoint.
Fast growing hand lesion
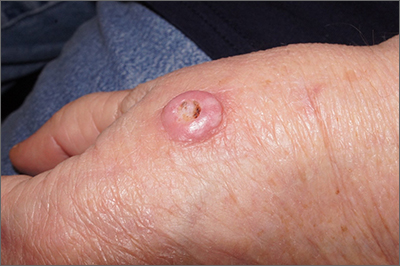
A scoop shave biopsy at the lower edge of the lesion revealed that this was a well-differentiated squamous cell carcinoma.
Squamous cell carcinoma is the second most common cancer in the United States and the most common skin cancer in Black people.1 A patient’s age and their accumulated UV radiation from sun exposure or artificial tanning is a major contributing factor. Lesions may manifest as precancers characterized as rough pink or brown papules with a sandpaper-like texture on sun-exposed skin. These lesions may clear spontaneously or develop into invasive disease, as occurred in this case.
Surgical treatment is often curative. Fusiform excision and Mohs micrographic surgery are 2 common options. More advanced squamous cell carcinomas that are large or found to have poorly differentiated cells or large perineural invasion carry a risk of metastasis.
In elderly patients, optimal treatment isn’t always straightforward.1 Nonsurgical options include radiation and intralesional chemotherapy. These nonsurgical choices may seem less aggressive, but total inconvenience, wound care, and discomfort can be equal to or worse than a single session of curative surgery.
This patient’s lesion was excised with a 5-mm margin. The patient tolerated an in-office procedure lasting about 45 minutes but would have struggled with a longer session with Mohs microsurgery. The postoperative period required limiting full use of his left hand for about 2 weeks.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206; quiz 178. 2. Renzi M Jr, Schimmel J, Decker A, et al. Management of skin cancer in the elderly. Dermatol Clin. 2019;37:279-286. doi: 10.1016/j.det.2019.02.003

A scoop shave biopsy at the lower edge of the lesion revealed that this was a well-differentiated squamous cell carcinoma.
Squamous cell carcinoma is the second most common cancer in the United States and the most common skin cancer in Black people.1 A patient’s age and their accumulated UV radiation from sun exposure or artificial tanning is a major contributing factor. Lesions may manifest as precancers characterized as rough pink or brown papules with a sandpaper-like texture on sun-exposed skin. These lesions may clear spontaneously or develop into invasive disease, as occurred in this case.
Surgical treatment is often curative. Fusiform excision and Mohs micrographic surgery are 2 common options. More advanced squamous cell carcinomas that are large or found to have poorly differentiated cells or large perineural invasion carry a risk of metastasis.
In elderly patients, optimal treatment isn’t always straightforward.1 Nonsurgical options include radiation and intralesional chemotherapy. These nonsurgical choices may seem less aggressive, but total inconvenience, wound care, and discomfort can be equal to or worse than a single session of curative surgery.
This patient’s lesion was excised with a 5-mm margin. The patient tolerated an in-office procedure lasting about 45 minutes but would have struggled with a longer session with Mohs microsurgery. The postoperative period required limiting full use of his left hand for about 2 weeks.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

A scoop shave biopsy at the lower edge of the lesion revealed that this was a well-differentiated squamous cell carcinoma.
Squamous cell carcinoma is the second most common cancer in the United States and the most common skin cancer in Black people.1 A patient’s age and their accumulated UV radiation from sun exposure or artificial tanning is a major contributing factor. Lesions may manifest as precancers characterized as rough pink or brown papules with a sandpaper-like texture on sun-exposed skin. These lesions may clear spontaneously or develop into invasive disease, as occurred in this case.
Surgical treatment is often curative. Fusiform excision and Mohs micrographic surgery are 2 common options. More advanced squamous cell carcinomas that are large or found to have poorly differentiated cells or large perineural invasion carry a risk of metastasis.
In elderly patients, optimal treatment isn’t always straightforward.1 Nonsurgical options include radiation and intralesional chemotherapy. These nonsurgical choices may seem less aggressive, but total inconvenience, wound care, and discomfort can be equal to or worse than a single session of curative surgery.
This patient’s lesion was excised with a 5-mm margin. The patient tolerated an in-office procedure lasting about 45 minutes but would have struggled with a longer session with Mohs microsurgery. The postoperative period required limiting full use of his left hand for about 2 weeks.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206; quiz 178. 2. Renzi M Jr, Schimmel J, Decker A, et al. Management of skin cancer in the elderly. Dermatol Clin. 2019;37:279-286. doi: 10.1016/j.det.2019.02.003
1. Bradford PT. Skin cancer in skin of color. Dermatol Nurs. 2009;21:170-177, 206; quiz 178. 2. Renzi M Jr, Schimmel J, Decker A, et al. Management of skin cancer in the elderly. Dermatol Clin. 2019;37:279-286. doi: 10.1016/j.det.2019.02.003
Consider the mnemonic ‘CLEAR’ when counseling acne patients
to use when treating this group of patients.
During a presentation at Medscape Live’s annual Coastal Dermatology Symposium, Dr. Harper, who practices at Dermatology and Skin Care of Birmingham, Ala., elaborated on the mnemonic, as follows:
C: Communicate expectations. “I look right at the acne patient and say, ‘I know you don’t just want to be better; I know you want to be clear,’ ” she said at the meeting. “ ‘That’s my goal for you, too. That may take us more than one visit and more than one treatment, but I am on your team, and that’s what we’re shooting for.’ If you don’t communicate that, they’re going to think that their acne is not that important to you.”
L: Listen for clues to customize the patient’s treatment. “We’re quick to say, ‘my patients don’t do what I recommend,’ or ‘they didn’t do what the last doctor recommended,’ ” Dr. Harper said. “Sometimes that is true, but there may be a reason why. Maybe the medication was too expensive. Maybe it was bleaching their fabrics. Maybe the regimen was too complex. Listen for opportunities to make adjustments to get their acne closer to clear.”
E: Treat early to improve quality of life and to decrease the risk of scarring. “I have a laser in my practice that is good at treating acne scarring,” she said. “Do I ever look at my patient and say, ‘don’t worry about those scars; I can make them go away?’ No. I look at them and say, ‘we can maybe make this 40% better,’ something like that. We have to prevent acne scars, because we’re not good at treating them.”
A: Treat aggressively with more combination therapies, more hormonal therapies, more isotretinoin, and perhaps more prior authorizations. She characterized the effort to obtain a prior authorization as “our megaphone back to insurance companies that says, ‘we think it is worth taking the time to do this prior authorization because the acne patient will benefit.’ ”
R: Don’t resist isotretinoin. Dr. Harper, who began practicing dermatology more than 20 years ago, said that over time, she has gradually prescribed more isotretinoin for her patients with acne. “It’s not a first-line [treatment], but I’m not afraid of it. If I can’t get somebody clear on other oral or topical treatments, we are going to try isotretinoin.”
The goal of acne treatment, she added, is to affect four key aspects of pathogenesis: follicular epithelial hyperproliferation, inflammation, Cutibacterium acnes (C. acnes), and sebum. “That’s what we’re always shooting for,” she said.
Dr. Harper is a past president of the American Acne & Rosacea Society. She disclosed that she serves as an advisor or consultant for Almirall, BioPharmX, Cassiopeia, Cutanea, Cutera, Dermira, EPI, Galderma, LaRoche-Posay, Ortho, Vyne, Sol Gel, and Sun. She also serves as a speaker or member of a speaker’s bureau for Almirall, EPI, Galderma, Ortho, and Vyne.
Medscape Live and this news organization are owned by the same parent company.
to use when treating this group of patients.
During a presentation at Medscape Live’s annual Coastal Dermatology Symposium, Dr. Harper, who practices at Dermatology and Skin Care of Birmingham, Ala., elaborated on the mnemonic, as follows:
C: Communicate expectations. “I look right at the acne patient and say, ‘I know you don’t just want to be better; I know you want to be clear,’ ” she said at the meeting. “ ‘That’s my goal for you, too. That may take us more than one visit and more than one treatment, but I am on your team, and that’s what we’re shooting for.’ If you don’t communicate that, they’re going to think that their acne is not that important to you.”
L: Listen for clues to customize the patient’s treatment. “We’re quick to say, ‘my patients don’t do what I recommend,’ or ‘they didn’t do what the last doctor recommended,’ ” Dr. Harper said. “Sometimes that is true, but there may be a reason why. Maybe the medication was too expensive. Maybe it was bleaching their fabrics. Maybe the regimen was too complex. Listen for opportunities to make adjustments to get their acne closer to clear.”
E: Treat early to improve quality of life and to decrease the risk of scarring. “I have a laser in my practice that is good at treating acne scarring,” she said. “Do I ever look at my patient and say, ‘don’t worry about those scars; I can make them go away?’ No. I look at them and say, ‘we can maybe make this 40% better,’ something like that. We have to prevent acne scars, because we’re not good at treating them.”
A: Treat aggressively with more combination therapies, more hormonal therapies, more isotretinoin, and perhaps more prior authorizations. She characterized the effort to obtain a prior authorization as “our megaphone back to insurance companies that says, ‘we think it is worth taking the time to do this prior authorization because the acne patient will benefit.’ ”
R: Don’t resist isotretinoin. Dr. Harper, who began practicing dermatology more than 20 years ago, said that over time, she has gradually prescribed more isotretinoin for her patients with acne. “It’s not a first-line [treatment], but I’m not afraid of it. If I can’t get somebody clear on other oral or topical treatments, we are going to try isotretinoin.”
The goal of acne treatment, she added, is to affect four key aspects of pathogenesis: follicular epithelial hyperproliferation, inflammation, Cutibacterium acnes (C. acnes), and sebum. “That’s what we’re always shooting for,” she said.
Dr. Harper is a past president of the American Acne & Rosacea Society. She disclosed that she serves as an advisor or consultant for Almirall, BioPharmX, Cassiopeia, Cutanea, Cutera, Dermira, EPI, Galderma, LaRoche-Posay, Ortho, Vyne, Sol Gel, and Sun. She also serves as a speaker or member of a speaker’s bureau for Almirall, EPI, Galderma, Ortho, and Vyne.
Medscape Live and this news organization are owned by the same parent company.
to use when treating this group of patients.
During a presentation at Medscape Live’s annual Coastal Dermatology Symposium, Dr. Harper, who practices at Dermatology and Skin Care of Birmingham, Ala., elaborated on the mnemonic, as follows:
C: Communicate expectations. “I look right at the acne patient and say, ‘I know you don’t just want to be better; I know you want to be clear,’ ” she said at the meeting. “ ‘That’s my goal for you, too. That may take us more than one visit and more than one treatment, but I am on your team, and that’s what we’re shooting for.’ If you don’t communicate that, they’re going to think that their acne is not that important to you.”
L: Listen for clues to customize the patient’s treatment. “We’re quick to say, ‘my patients don’t do what I recommend,’ or ‘they didn’t do what the last doctor recommended,’ ” Dr. Harper said. “Sometimes that is true, but there may be a reason why. Maybe the medication was too expensive. Maybe it was bleaching their fabrics. Maybe the regimen was too complex. Listen for opportunities to make adjustments to get their acne closer to clear.”
E: Treat early to improve quality of life and to decrease the risk of scarring. “I have a laser in my practice that is good at treating acne scarring,” she said. “Do I ever look at my patient and say, ‘don’t worry about those scars; I can make them go away?’ No. I look at them and say, ‘we can maybe make this 40% better,’ something like that. We have to prevent acne scars, because we’re not good at treating them.”
A: Treat aggressively with more combination therapies, more hormonal therapies, more isotretinoin, and perhaps more prior authorizations. She characterized the effort to obtain a prior authorization as “our megaphone back to insurance companies that says, ‘we think it is worth taking the time to do this prior authorization because the acne patient will benefit.’ ”
R: Don’t resist isotretinoin. Dr. Harper, who began practicing dermatology more than 20 years ago, said that over time, she has gradually prescribed more isotretinoin for her patients with acne. “It’s not a first-line [treatment], but I’m not afraid of it. If I can’t get somebody clear on other oral or topical treatments, we are going to try isotretinoin.”
The goal of acne treatment, she added, is to affect four key aspects of pathogenesis: follicular epithelial hyperproliferation, inflammation, Cutibacterium acnes (C. acnes), and sebum. “That’s what we’re always shooting for,” she said.
Dr. Harper is a past president of the American Acne & Rosacea Society. She disclosed that she serves as an advisor or consultant for Almirall, BioPharmX, Cassiopeia, Cutanea, Cutera, Dermira, EPI, Galderma, LaRoche-Posay, Ortho, Vyne, Sol Gel, and Sun. She also serves as a speaker or member of a speaker’s bureau for Almirall, EPI, Galderma, Ortho, and Vyne.
Medscape Live and this news organization are owned by the same parent company.
FROM MEDSCAPE LIVE COASTAL DERM
Many factors linked with higher, lower risk for hand eczema
“In this population-based study, all atopic diseases, not only atopic dermatitis, were found as individual risk factors for HE. In addition, female gender, obesity and mold exposure increased the risk of HE,” wrote Marjut Koskelo, MD, and her colleagues at the University of Oulu in Finland. Their report was published in Contact Dermatitis.
“Parental allergy was also a risk factor of offspring’s HE. Moderate or high physical activity as well as owning a dog appeared as protective factors of HE. No association was found between other lifestyle factors and HE,” they added.
Hand eczema is one of the most common skin disorders and is the most common occupational skin disease, the authors wrote. Many risk factors, including atopic dermatitis, are known to be linked with HE, but whether various other factors might also be linked has not been well studied.
The research team investigated the link between HE and atopic diseases, parental factors, environmental factors (exposure to mold, keeping animals), and lifestyle factors (physical activity, obesity, tobacco and alcohol use).
They analyzed data of people who took part in the Northern Finland Birth Cohort 1966 Study. The data, collected since 1965, includes details about 12,055 mothers in northern Finland who were expected to deliver babies in 1966, and their 12,058 live-born children. The children have been followed over the years with questionnaires and clinical examinations, and their parents have been followed by national registers and medical reports.
For the 46-year follow-up, 6,830 respondents aged 45-46 years, roughly half of them women, completed a 132-question form covering physical health, lifestyle, environmental factors, socioeconomic status, and history of hand eczema and other atopic diseases.
In the statistical analysis, the researchers adjusted for atopic dermatitis, asthma, allergic rhinoconjunctivitis, education level, body mass index, maternal BMI, parental allergy, physical activity, living on a farm, and mold exposure and symptoms.
Of the 900 respondents who reported having had HE, 592 (65.8%) were women and 308 (34.2%) were men (odds ratio [OR], 1.73; 95% confidence interval [CI], 1.49-2.0).
Various factors linked with hand eczema risk
The authors found the following:
- Atopic diseases and HE were linked: atopic dermatitis (adjusted odds ratio [aOR], 9.66; 95% CI, 8.03-11.66), asthma (aOR, 1.38; 95% CI, 1.12-1.71), and allergic rhinoconjunctivitis (aOR, 1.28; 95% CI, 1.04-1.56). Sex did not affect the link between atopic diseases and HE.
- Respondents who reported visible mold or mold odor in their apartments had higher risk for HE than did those without a history of mold exposure (OR, 1.32; 95% CI, 1.07-1.61).
- Obesity was linked with HE (OR, 3.44; 95% CI, 1.05-22.8), but smoking status, alcohol intake, and education level were not statistically significant risk factors for HE.
- Participants who reported moderate or high physical activity had lower risk for HE (OR, 0.78; 95% CI, 0.64-0.94; and OR, 0.56; 95% CI, 0.33-0.91, respectively) than those who were less active.
- Parental allergy increased risk for HE (OR, 1.98; 95% CI, 1.70-2.30); as maternal age, BMI, and menarche age increased, so did the risk for the child’s HE, but the increases were not statistically significant; and no significant links were found between maternal tobacco smoking, parental asthma, birth weight, parity, gestational age, and HE.
- Dog owners had less risk for HE than did people without a dog (OR, 0.83; 95% CI, 0.71-0.97); links between cat or farm animal owners and HE were not significant.
“There is a strong association between hand eczema and atopic diseases,” Maya Jonas, MD, clinical assistant professor of dermatology at The Ohio State University Wexner Medical Center in Columbus, told this news organization.
“When evaluating patients with hand eczema, it is important to ask if they have a history of atopic dermatitis, asthma, or allergic rhinitis,” said Dr. Jonas, who was not involved in the study.
Elma Baron, MD, professor and director, Skin Study Center, department of dermatology, Case Western Reserve University, Cleveland, was surprised by the inverse link between physical activity and HE.
“What struck me as interesting is the inverse association between hand eczema and physical activity, that greater physical activity will decrease the risk for hand eczema,” she said in an interview. “It’s an interesting finding that’s worth exploring.
“Dermatologists have also speculated about the association with the female gender, because women are more likely to be in situations that involve frequent hand washing or in occupations, such as hairdressing, that involve known irritants and allergens,” added Dr. Baron, who was not involved in the study.
The main weakness, she noted, is the reliance on self-reported diagnosis. “Hand eczema is a common condition, but the etiologies of reported hand eczema may vary.
“Being cognizant of these associations can help us prescribe appropriate medications and advise patients about how they can avoid exposures that will aggravate their condition,” Dr. Baron advised.
The authors recommend further related studies.
The authors, Dr. Jonas, and Dr. Baron report no relevant financial relationships. The study was not funded.
A version of this article first appeared on Medscape.com.
“In this population-based study, all atopic diseases, not only atopic dermatitis, were found as individual risk factors for HE. In addition, female gender, obesity and mold exposure increased the risk of HE,” wrote Marjut Koskelo, MD, and her colleagues at the University of Oulu in Finland. Their report was published in Contact Dermatitis.
“Parental allergy was also a risk factor of offspring’s HE. Moderate or high physical activity as well as owning a dog appeared as protective factors of HE. No association was found between other lifestyle factors and HE,” they added.
Hand eczema is one of the most common skin disorders and is the most common occupational skin disease, the authors wrote. Many risk factors, including atopic dermatitis, are known to be linked with HE, but whether various other factors might also be linked has not been well studied.
The research team investigated the link between HE and atopic diseases, parental factors, environmental factors (exposure to mold, keeping animals), and lifestyle factors (physical activity, obesity, tobacco and alcohol use).
They analyzed data of people who took part in the Northern Finland Birth Cohort 1966 Study. The data, collected since 1965, includes details about 12,055 mothers in northern Finland who were expected to deliver babies in 1966, and their 12,058 live-born children. The children have been followed over the years with questionnaires and clinical examinations, and their parents have been followed by national registers and medical reports.
For the 46-year follow-up, 6,830 respondents aged 45-46 years, roughly half of them women, completed a 132-question form covering physical health, lifestyle, environmental factors, socioeconomic status, and history of hand eczema and other atopic diseases.
In the statistical analysis, the researchers adjusted for atopic dermatitis, asthma, allergic rhinoconjunctivitis, education level, body mass index, maternal BMI, parental allergy, physical activity, living on a farm, and mold exposure and symptoms.
Of the 900 respondents who reported having had HE, 592 (65.8%) were women and 308 (34.2%) were men (odds ratio [OR], 1.73; 95% confidence interval [CI], 1.49-2.0).
Various factors linked with hand eczema risk
The authors found the following:
- Atopic diseases and HE were linked: atopic dermatitis (adjusted odds ratio [aOR], 9.66; 95% CI, 8.03-11.66), asthma (aOR, 1.38; 95% CI, 1.12-1.71), and allergic rhinoconjunctivitis (aOR, 1.28; 95% CI, 1.04-1.56). Sex did not affect the link between atopic diseases and HE.
- Respondents who reported visible mold or mold odor in their apartments had higher risk for HE than did those without a history of mold exposure (OR, 1.32; 95% CI, 1.07-1.61).
- Obesity was linked with HE (OR, 3.44; 95% CI, 1.05-22.8), but smoking status, alcohol intake, and education level were not statistically significant risk factors for HE.
- Participants who reported moderate or high physical activity had lower risk for HE (OR, 0.78; 95% CI, 0.64-0.94; and OR, 0.56; 95% CI, 0.33-0.91, respectively) than those who were less active.
- Parental allergy increased risk for HE (OR, 1.98; 95% CI, 1.70-2.30); as maternal age, BMI, and menarche age increased, so did the risk for the child’s HE, but the increases were not statistically significant; and no significant links were found between maternal tobacco smoking, parental asthma, birth weight, parity, gestational age, and HE.
- Dog owners had less risk for HE than did people without a dog (OR, 0.83; 95% CI, 0.71-0.97); links between cat or farm animal owners and HE were not significant.
“There is a strong association between hand eczema and atopic diseases,” Maya Jonas, MD, clinical assistant professor of dermatology at The Ohio State University Wexner Medical Center in Columbus, told this news organization.
“When evaluating patients with hand eczema, it is important to ask if they have a history of atopic dermatitis, asthma, or allergic rhinitis,” said Dr. Jonas, who was not involved in the study.
Elma Baron, MD, professor and director, Skin Study Center, department of dermatology, Case Western Reserve University, Cleveland, was surprised by the inverse link between physical activity and HE.
“What struck me as interesting is the inverse association between hand eczema and physical activity, that greater physical activity will decrease the risk for hand eczema,” she said in an interview. “It’s an interesting finding that’s worth exploring.
“Dermatologists have also speculated about the association with the female gender, because women are more likely to be in situations that involve frequent hand washing or in occupations, such as hairdressing, that involve known irritants and allergens,” added Dr. Baron, who was not involved in the study.
The main weakness, she noted, is the reliance on self-reported diagnosis. “Hand eczema is a common condition, but the etiologies of reported hand eczema may vary.
“Being cognizant of these associations can help us prescribe appropriate medications and advise patients about how they can avoid exposures that will aggravate their condition,” Dr. Baron advised.
The authors recommend further related studies.
The authors, Dr. Jonas, and Dr. Baron report no relevant financial relationships. The study was not funded.
A version of this article first appeared on Medscape.com.
“In this population-based study, all atopic diseases, not only atopic dermatitis, were found as individual risk factors for HE. In addition, female gender, obesity and mold exposure increased the risk of HE,” wrote Marjut Koskelo, MD, and her colleagues at the University of Oulu in Finland. Their report was published in Contact Dermatitis.
“Parental allergy was also a risk factor of offspring’s HE. Moderate or high physical activity as well as owning a dog appeared as protective factors of HE. No association was found between other lifestyle factors and HE,” they added.
Hand eczema is one of the most common skin disorders and is the most common occupational skin disease, the authors wrote. Many risk factors, including atopic dermatitis, are known to be linked with HE, but whether various other factors might also be linked has not been well studied.
The research team investigated the link between HE and atopic diseases, parental factors, environmental factors (exposure to mold, keeping animals), and lifestyle factors (physical activity, obesity, tobacco and alcohol use).
They analyzed data of people who took part in the Northern Finland Birth Cohort 1966 Study. The data, collected since 1965, includes details about 12,055 mothers in northern Finland who were expected to deliver babies in 1966, and their 12,058 live-born children. The children have been followed over the years with questionnaires and clinical examinations, and their parents have been followed by national registers and medical reports.
For the 46-year follow-up, 6,830 respondents aged 45-46 years, roughly half of them women, completed a 132-question form covering physical health, lifestyle, environmental factors, socioeconomic status, and history of hand eczema and other atopic diseases.
In the statistical analysis, the researchers adjusted for atopic dermatitis, asthma, allergic rhinoconjunctivitis, education level, body mass index, maternal BMI, parental allergy, physical activity, living on a farm, and mold exposure and symptoms.
Of the 900 respondents who reported having had HE, 592 (65.8%) were women and 308 (34.2%) were men (odds ratio [OR], 1.73; 95% confidence interval [CI], 1.49-2.0).
Various factors linked with hand eczema risk
The authors found the following:
- Atopic diseases and HE were linked: atopic dermatitis (adjusted odds ratio [aOR], 9.66; 95% CI, 8.03-11.66), asthma (aOR, 1.38; 95% CI, 1.12-1.71), and allergic rhinoconjunctivitis (aOR, 1.28; 95% CI, 1.04-1.56). Sex did not affect the link between atopic diseases and HE.
- Respondents who reported visible mold or mold odor in their apartments had higher risk for HE than did those without a history of mold exposure (OR, 1.32; 95% CI, 1.07-1.61).
- Obesity was linked with HE (OR, 3.44; 95% CI, 1.05-22.8), but smoking status, alcohol intake, and education level were not statistically significant risk factors for HE.
- Participants who reported moderate or high physical activity had lower risk for HE (OR, 0.78; 95% CI, 0.64-0.94; and OR, 0.56; 95% CI, 0.33-0.91, respectively) than those who were less active.
- Parental allergy increased risk for HE (OR, 1.98; 95% CI, 1.70-2.30); as maternal age, BMI, and menarche age increased, so did the risk for the child’s HE, but the increases were not statistically significant; and no significant links were found between maternal tobacco smoking, parental asthma, birth weight, parity, gestational age, and HE.
- Dog owners had less risk for HE than did people without a dog (OR, 0.83; 95% CI, 0.71-0.97); links between cat or farm animal owners and HE were not significant.
“There is a strong association between hand eczema and atopic diseases,” Maya Jonas, MD, clinical assistant professor of dermatology at The Ohio State University Wexner Medical Center in Columbus, told this news organization.
“When evaluating patients with hand eczema, it is important to ask if they have a history of atopic dermatitis, asthma, or allergic rhinitis,” said Dr. Jonas, who was not involved in the study.
Elma Baron, MD, professor and director, Skin Study Center, department of dermatology, Case Western Reserve University, Cleveland, was surprised by the inverse link between physical activity and HE.
“What struck me as interesting is the inverse association between hand eczema and physical activity, that greater physical activity will decrease the risk for hand eczema,” she said in an interview. “It’s an interesting finding that’s worth exploring.
“Dermatologists have also speculated about the association with the female gender, because women are more likely to be in situations that involve frequent hand washing or in occupations, such as hairdressing, that involve known irritants and allergens,” added Dr. Baron, who was not involved in the study.
The main weakness, she noted, is the reliance on self-reported diagnosis. “Hand eczema is a common condition, but the etiologies of reported hand eczema may vary.
“Being cognizant of these associations can help us prescribe appropriate medications and advise patients about how they can avoid exposures that will aggravate their condition,” Dr. Baron advised.
The authors recommend further related studies.
The authors, Dr. Jonas, and Dr. Baron report no relevant financial relationships. The study was not funded.
A version of this article first appeared on Medscape.com.
FROM CONTACT DERMATITIS
Apremilast may have some cardiometabolic benefits in patients with psoriasis
The trial, led by Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania, Philadelphia, found that apremilast (Otezla) has a neutral effect on aortic vascular inflammation in patients with moderate to severe psoriasis.
It also had variable, but generally favorable, associations with 68 cardiometabolic biomarkers tested and associations with reductions in both visceral and subcutaneous fat. Findings of the study were published online in JAMA Dermatology.
Fat reductions maintained at 1-year mark
The researchers found a 5%-6% reduction in subcutaneous and visceral fat at week 16 of the study that was maintained at the 1-year mark. “The fact that it was rock stable a year later is pretty encouraging,” Dr. Gelfand told this news organization.
As for effects on vascular inflammation, Dr. Gelfand said, “The good news is we didn’t find any adverse effects on aortic vascular inflammation, but we didn’t find any beneficial effects either. That was a little disappointing.
“The most surprising thing was really the effects on visceral adiposity,” he added. “I’m not aware of any other drug having demonstrated that effect.”
Michael S. Garshick, MD, a cardiologist with NYU Langone Health in New York, who was not involved with the trial, told this news organization that despite seemingly good epidemiologic evidence in observational studies that by treating psoriasis surrogates of cardiovascular risk can be reduced, this trial, like others before it, failed to reduce aortic vascular inflammation.
The trial does help answer the question of whether apremilast can induce weight loss, he said, something that earlier trials suggested. “This trial confirms that, which is exciting,” he said. The reduction in both visceral and subcutaneous fat “deserves a lot further study.”
Several questions remain, Dr. Garshick said. Both he and Dr. Gelfand pointed to the need for large, placebo-controlled trials. “We still don’t know which medications may be preferrable in psoriasis to reduce [cardiovascular] risk if any at all,” Dr. Garshick said.
Seventy patients enrolled
In total, 70 patients with moderate to severe psoriasis were enrolled, 60 completed week 16, and 39 completed week 52 of the single-arm, open-label trial conducted between April 2017 and August 2021 at seven dermatology sites in the United States.
Participants took 30 mg of apremilast, an oral phosphodiesterase-4 (PDE-4) inhibitor approved for treating psoriasis and psoriatic arthritis, twice daily. Participants’ average age was 47.5 years; most were male (77.1%) and White (82.9%); almost 6% were Black. Average body mass index was 30 kg/m2. Patients could not have received biologics within 90 days of study baseline (or 180 days for ustekinumab [Stelara]).
There was no change in aortic vascular inflammation at week 16 (target to background ratio, −0.02; 95% confidence interval [CI], −0.08 to 0.05; P = .61) or week 52 (target to background ratio, −0.07; 95% CI, −0.15 to 0.01; P = .09) compared with baseline.
“At week 16, there were reductions in levels of interleukin-1b, fetuin A, valine, leucine, and isoleucine,” the authors wrote, adding that at week 52, compared with baseline, “there were reductions in levels of ferritin, cholesterol efflux capacity, beta-hydroxybutyrate, acetone, and ketone bodies, and an increase in levels of apolipoprotein A-1.”
This study highlights the importance of screening, Dr. Garshick said.
He and Dr. Gelfand said people with psoriatic disease tend to be vastly underscreened for cardiovascular risk factors.
Dr. Gelfand said, “If we did what we knew worked – meaning we screened them for diabetes, we screen their cholesterol, we check their blood pressure, and we adequately treated those traditional cardiovascular risk factors, we probably could narrow the gap quite a bit” in terms of the lower life expectancy people face when they have more significant psoriasis.
Celgene was the initial funding sponsor; sponsorship was then transferred to Amgen. The authors designed, executed, analyzed, and reported the study. Celgene provided nonbinding input into study design, and Amgen provided nonbinding input into the reporting of results. Dr. Gelfand reported numerous disclosures with various pharmaceutical companies and organizations. Dr. Garshick reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The trial, led by Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania, Philadelphia, found that apremilast (Otezla) has a neutral effect on aortic vascular inflammation in patients with moderate to severe psoriasis.
It also had variable, but generally favorable, associations with 68 cardiometabolic biomarkers tested and associations with reductions in both visceral and subcutaneous fat. Findings of the study were published online in JAMA Dermatology.
Fat reductions maintained at 1-year mark
The researchers found a 5%-6% reduction in subcutaneous and visceral fat at week 16 of the study that was maintained at the 1-year mark. “The fact that it was rock stable a year later is pretty encouraging,” Dr. Gelfand told this news organization.
As for effects on vascular inflammation, Dr. Gelfand said, “The good news is we didn’t find any adverse effects on aortic vascular inflammation, but we didn’t find any beneficial effects either. That was a little disappointing.
“The most surprising thing was really the effects on visceral adiposity,” he added. “I’m not aware of any other drug having demonstrated that effect.”
Michael S. Garshick, MD, a cardiologist with NYU Langone Health in New York, who was not involved with the trial, told this news organization that despite seemingly good epidemiologic evidence in observational studies that by treating psoriasis surrogates of cardiovascular risk can be reduced, this trial, like others before it, failed to reduce aortic vascular inflammation.
The trial does help answer the question of whether apremilast can induce weight loss, he said, something that earlier trials suggested. “This trial confirms that, which is exciting,” he said. The reduction in both visceral and subcutaneous fat “deserves a lot further study.”
Several questions remain, Dr. Garshick said. Both he and Dr. Gelfand pointed to the need for large, placebo-controlled trials. “We still don’t know which medications may be preferrable in psoriasis to reduce [cardiovascular] risk if any at all,” Dr. Garshick said.
Seventy patients enrolled
In total, 70 patients with moderate to severe psoriasis were enrolled, 60 completed week 16, and 39 completed week 52 of the single-arm, open-label trial conducted between April 2017 and August 2021 at seven dermatology sites in the United States.
Participants took 30 mg of apremilast, an oral phosphodiesterase-4 (PDE-4) inhibitor approved for treating psoriasis and psoriatic arthritis, twice daily. Participants’ average age was 47.5 years; most were male (77.1%) and White (82.9%); almost 6% were Black. Average body mass index was 30 kg/m2. Patients could not have received biologics within 90 days of study baseline (or 180 days for ustekinumab [Stelara]).
There was no change in aortic vascular inflammation at week 16 (target to background ratio, −0.02; 95% confidence interval [CI], −0.08 to 0.05; P = .61) or week 52 (target to background ratio, −0.07; 95% CI, −0.15 to 0.01; P = .09) compared with baseline.
“At week 16, there were reductions in levels of interleukin-1b, fetuin A, valine, leucine, and isoleucine,” the authors wrote, adding that at week 52, compared with baseline, “there were reductions in levels of ferritin, cholesterol efflux capacity, beta-hydroxybutyrate, acetone, and ketone bodies, and an increase in levels of apolipoprotein A-1.”
This study highlights the importance of screening, Dr. Garshick said.
He and Dr. Gelfand said people with psoriatic disease tend to be vastly underscreened for cardiovascular risk factors.
Dr. Gelfand said, “If we did what we knew worked – meaning we screened them for diabetes, we screen their cholesterol, we check their blood pressure, and we adequately treated those traditional cardiovascular risk factors, we probably could narrow the gap quite a bit” in terms of the lower life expectancy people face when they have more significant psoriasis.
Celgene was the initial funding sponsor; sponsorship was then transferred to Amgen. The authors designed, executed, analyzed, and reported the study. Celgene provided nonbinding input into study design, and Amgen provided nonbinding input into the reporting of results. Dr. Gelfand reported numerous disclosures with various pharmaceutical companies and organizations. Dr. Garshick reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The trial, led by Joel M. Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania, Philadelphia, found that apremilast (Otezla) has a neutral effect on aortic vascular inflammation in patients with moderate to severe psoriasis.
It also had variable, but generally favorable, associations with 68 cardiometabolic biomarkers tested and associations with reductions in both visceral and subcutaneous fat. Findings of the study were published online in JAMA Dermatology.
Fat reductions maintained at 1-year mark
The researchers found a 5%-6% reduction in subcutaneous and visceral fat at week 16 of the study that was maintained at the 1-year mark. “The fact that it was rock stable a year later is pretty encouraging,” Dr. Gelfand told this news organization.
As for effects on vascular inflammation, Dr. Gelfand said, “The good news is we didn’t find any adverse effects on aortic vascular inflammation, but we didn’t find any beneficial effects either. That was a little disappointing.
“The most surprising thing was really the effects on visceral adiposity,” he added. “I’m not aware of any other drug having demonstrated that effect.”
Michael S. Garshick, MD, a cardiologist with NYU Langone Health in New York, who was not involved with the trial, told this news organization that despite seemingly good epidemiologic evidence in observational studies that by treating psoriasis surrogates of cardiovascular risk can be reduced, this trial, like others before it, failed to reduce aortic vascular inflammation.
The trial does help answer the question of whether apremilast can induce weight loss, he said, something that earlier trials suggested. “This trial confirms that, which is exciting,” he said. The reduction in both visceral and subcutaneous fat “deserves a lot further study.”
Several questions remain, Dr. Garshick said. Both he and Dr. Gelfand pointed to the need for large, placebo-controlled trials. “We still don’t know which medications may be preferrable in psoriasis to reduce [cardiovascular] risk if any at all,” Dr. Garshick said.
Seventy patients enrolled
In total, 70 patients with moderate to severe psoriasis were enrolled, 60 completed week 16, and 39 completed week 52 of the single-arm, open-label trial conducted between April 2017 and August 2021 at seven dermatology sites in the United States.
Participants took 30 mg of apremilast, an oral phosphodiesterase-4 (PDE-4) inhibitor approved for treating psoriasis and psoriatic arthritis, twice daily. Participants’ average age was 47.5 years; most were male (77.1%) and White (82.9%); almost 6% were Black. Average body mass index was 30 kg/m2. Patients could not have received biologics within 90 days of study baseline (or 180 days for ustekinumab [Stelara]).
There was no change in aortic vascular inflammation at week 16 (target to background ratio, −0.02; 95% confidence interval [CI], −0.08 to 0.05; P = .61) or week 52 (target to background ratio, −0.07; 95% CI, −0.15 to 0.01; P = .09) compared with baseline.
“At week 16, there were reductions in levels of interleukin-1b, fetuin A, valine, leucine, and isoleucine,” the authors wrote, adding that at week 52, compared with baseline, “there were reductions in levels of ferritin, cholesterol efflux capacity, beta-hydroxybutyrate, acetone, and ketone bodies, and an increase in levels of apolipoprotein A-1.”
This study highlights the importance of screening, Dr. Garshick said.
He and Dr. Gelfand said people with psoriatic disease tend to be vastly underscreened for cardiovascular risk factors.
Dr. Gelfand said, “If we did what we knew worked – meaning we screened them for diabetes, we screen their cholesterol, we check their blood pressure, and we adequately treated those traditional cardiovascular risk factors, we probably could narrow the gap quite a bit” in terms of the lower life expectancy people face when they have more significant psoriasis.
Celgene was the initial funding sponsor; sponsorship was then transferred to Amgen. The authors designed, executed, analyzed, and reported the study. Celgene provided nonbinding input into study design, and Amgen provided nonbinding input into the reporting of results. Dr. Gelfand reported numerous disclosures with various pharmaceutical companies and organizations. Dr. Garshick reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY
Early emollient use reduces dermatitis in at-risk infants
Recent study findings published in Allergy (2022 Aug 23. doi: 10.1111/all.15491) suggest that
The single-center STOP-AD clinical trial recruited term infants within 4 days of birth who were at high risk for AD, as determined on the basis of a parent-reported history of the disease or asthma or allergic rhinitis. Infants were randomly assigned to undergo either a standard skin care routine (control group; n = 160) or twice-daily emollient application for the first 8 weeks of life (intervention group; n = 161).
In the intervention group, infants received an emollient that was specifically formulated for AD-prone skin. The control group received standard skin care advice, which did not include specific advice on bathing frequency or regular emollient use.
The mean age of the infants at randomization was 1.9 days. A total of 41 infants in the intervention group and 20 infants in the control group were withdrawn from the study. Most withdrawals (80%) occurred prior to the 2-week visit.
At 12 months, the cumulative incidence of AD was 32.8% in the intervention group and 46.4% in the control group (P = .036). The investigators note that daily emollient use was associated with a 29% lower risk of cumulative AD at 1 year in comparison with the control intervention.
No significant difference was observed between the groups regarding the incidence of parent-reported skin infections during the treatment period (5.0% vs. 5.7%; P > .05).
Study investigator Jonathan O’Brien Hourihane, MBBS, of the Royal College of Surgeons in Dublin, said in an interview that previously published findings from the BASELINE study supported the rationale for the early use of emollients in infancy to prevent AD.
The investigators of the BASELINE study found that skin barrier function, as measured by transepidermal water loss, increased from birth to 8 weeks but then became stable at 6 months. These observations suggest that the period during early infancy “could be a critical window in which to protect the skin barrier” of infants at risk for AD, Dr. Hourihane added.
Dr. Hourihane, who serves as the head of department of pediatrics at the Royal College of Surgeons, explained that the long-term clinical burden of AD is often more significant if the condition begins earlier in life, underscoring the importance of early prevention and control.
“The casual role [of AD] in other allergic conditions remains suspected but not proven, but its association is clear,” he said. He noted that infants with eczema “also have poorer sleep, and the condition causes increased family disruption,” highlighting the far-reaching burden of AD.
Commenting on the study, Adelaide Hebert, MD, professor of pediatric dermatology at the University of Texas, Houston, said in an interview that the barrier defect observed in AD is one of the prime areas to address as a means of controlling the chronic, relapsing disorder. She noted that the use of emollients can repair this defective barrier.
“Early initiation of emollients has the potential to reduce dryness, itching, transgression of allergens, and infectious agents,” explained Dr. Hebert, who wasn’t involved in the study. “Emollient application also allows the parent to inspect the skin surface and address any challenges in a timely manner.”
In the STOP-AD trial, Dr. Hourihane and colleagues also found that, among patients with loss-of-function (LoF) mutations in the filaggrin gene (FLG), the prevalence of AD at 6 and 12 months seemed to be a higher than among patients with the wild-type gene, but the difference did not reach statistical significance.
Commenting on this finding, Dr. Hebert noted that LoF FLG mutation carriers may benefit especially from emollient use, given that LoF mutations in FLG is associated with reduced production of natural moisturizing factors in the skin.
Regarding future research directions, Dr. Hourihane stated that there is a need for replication and validation of the findings in studies that include infants from different ethnic backgrounds as well as those from various social settings. These studies should also include variable treatment windows to determine both short- and longer-term effects of emollient use in this population, Dr. Hourihane explained.
Dr. Hourihane added that he and the investigators do not yet understand which aspect of the study’s program was key for reducing the incidence of AD in the first year of life. “The timing of emollient initiation, the duration of treatment, the products, or maybe just a combination of these” could be possible explanations.
The study was independently supported. Dr. Hourihand reported receiving grant funding from Aimmune Therapeutics and DBV Technologies. Dr. Hebert reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Recent study findings published in Allergy (2022 Aug 23. doi: 10.1111/all.15491) suggest that
The single-center STOP-AD clinical trial recruited term infants within 4 days of birth who were at high risk for AD, as determined on the basis of a parent-reported history of the disease or asthma or allergic rhinitis. Infants were randomly assigned to undergo either a standard skin care routine (control group; n = 160) or twice-daily emollient application for the first 8 weeks of life (intervention group; n = 161).
In the intervention group, infants received an emollient that was specifically formulated for AD-prone skin. The control group received standard skin care advice, which did not include specific advice on bathing frequency or regular emollient use.
The mean age of the infants at randomization was 1.9 days. A total of 41 infants in the intervention group and 20 infants in the control group were withdrawn from the study. Most withdrawals (80%) occurred prior to the 2-week visit.
At 12 months, the cumulative incidence of AD was 32.8% in the intervention group and 46.4% in the control group (P = .036). The investigators note that daily emollient use was associated with a 29% lower risk of cumulative AD at 1 year in comparison with the control intervention.
No significant difference was observed between the groups regarding the incidence of parent-reported skin infections during the treatment period (5.0% vs. 5.7%; P > .05).
Study investigator Jonathan O’Brien Hourihane, MBBS, of the Royal College of Surgeons in Dublin, said in an interview that previously published findings from the BASELINE study supported the rationale for the early use of emollients in infancy to prevent AD.
The investigators of the BASELINE study found that skin barrier function, as measured by transepidermal water loss, increased from birth to 8 weeks but then became stable at 6 months. These observations suggest that the period during early infancy “could be a critical window in which to protect the skin barrier” of infants at risk for AD, Dr. Hourihane added.
Dr. Hourihane, who serves as the head of department of pediatrics at the Royal College of Surgeons, explained that the long-term clinical burden of AD is often more significant if the condition begins earlier in life, underscoring the importance of early prevention and control.
“The casual role [of AD] in other allergic conditions remains suspected but not proven, but its association is clear,” he said. He noted that infants with eczema “also have poorer sleep, and the condition causes increased family disruption,” highlighting the far-reaching burden of AD.
Commenting on the study, Adelaide Hebert, MD, professor of pediatric dermatology at the University of Texas, Houston, said in an interview that the barrier defect observed in AD is one of the prime areas to address as a means of controlling the chronic, relapsing disorder. She noted that the use of emollients can repair this defective barrier.
“Early initiation of emollients has the potential to reduce dryness, itching, transgression of allergens, and infectious agents,” explained Dr. Hebert, who wasn’t involved in the study. “Emollient application also allows the parent to inspect the skin surface and address any challenges in a timely manner.”
In the STOP-AD trial, Dr. Hourihane and colleagues also found that, among patients with loss-of-function (LoF) mutations in the filaggrin gene (FLG), the prevalence of AD at 6 and 12 months seemed to be a higher than among patients with the wild-type gene, but the difference did not reach statistical significance.
Commenting on this finding, Dr. Hebert noted that LoF FLG mutation carriers may benefit especially from emollient use, given that LoF mutations in FLG is associated with reduced production of natural moisturizing factors in the skin.
Regarding future research directions, Dr. Hourihane stated that there is a need for replication and validation of the findings in studies that include infants from different ethnic backgrounds as well as those from various social settings. These studies should also include variable treatment windows to determine both short- and longer-term effects of emollient use in this population, Dr. Hourihane explained.
Dr. Hourihane added that he and the investigators do not yet understand which aspect of the study’s program was key for reducing the incidence of AD in the first year of life. “The timing of emollient initiation, the duration of treatment, the products, or maybe just a combination of these” could be possible explanations.
The study was independently supported. Dr. Hourihand reported receiving grant funding from Aimmune Therapeutics and DBV Technologies. Dr. Hebert reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Recent study findings published in Allergy (2022 Aug 23. doi: 10.1111/all.15491) suggest that
The single-center STOP-AD clinical trial recruited term infants within 4 days of birth who were at high risk for AD, as determined on the basis of a parent-reported history of the disease or asthma or allergic rhinitis. Infants were randomly assigned to undergo either a standard skin care routine (control group; n = 160) or twice-daily emollient application for the first 8 weeks of life (intervention group; n = 161).
In the intervention group, infants received an emollient that was specifically formulated for AD-prone skin. The control group received standard skin care advice, which did not include specific advice on bathing frequency or regular emollient use.
The mean age of the infants at randomization was 1.9 days. A total of 41 infants in the intervention group and 20 infants in the control group were withdrawn from the study. Most withdrawals (80%) occurred prior to the 2-week visit.
At 12 months, the cumulative incidence of AD was 32.8% in the intervention group and 46.4% in the control group (P = .036). The investigators note that daily emollient use was associated with a 29% lower risk of cumulative AD at 1 year in comparison with the control intervention.
No significant difference was observed between the groups regarding the incidence of parent-reported skin infections during the treatment period (5.0% vs. 5.7%; P > .05).
Study investigator Jonathan O’Brien Hourihane, MBBS, of the Royal College of Surgeons in Dublin, said in an interview that previously published findings from the BASELINE study supported the rationale for the early use of emollients in infancy to prevent AD.
The investigators of the BASELINE study found that skin barrier function, as measured by transepidermal water loss, increased from birth to 8 weeks but then became stable at 6 months. These observations suggest that the period during early infancy “could be a critical window in which to protect the skin barrier” of infants at risk for AD, Dr. Hourihane added.
Dr. Hourihane, who serves as the head of department of pediatrics at the Royal College of Surgeons, explained that the long-term clinical burden of AD is often more significant if the condition begins earlier in life, underscoring the importance of early prevention and control.
“The casual role [of AD] in other allergic conditions remains suspected but not proven, but its association is clear,” he said. He noted that infants with eczema “also have poorer sleep, and the condition causes increased family disruption,” highlighting the far-reaching burden of AD.
Commenting on the study, Adelaide Hebert, MD, professor of pediatric dermatology at the University of Texas, Houston, said in an interview that the barrier defect observed in AD is one of the prime areas to address as a means of controlling the chronic, relapsing disorder. She noted that the use of emollients can repair this defective barrier.
“Early initiation of emollients has the potential to reduce dryness, itching, transgression of allergens, and infectious agents,” explained Dr. Hebert, who wasn’t involved in the study. “Emollient application also allows the parent to inspect the skin surface and address any challenges in a timely manner.”
In the STOP-AD trial, Dr. Hourihane and colleagues also found that, among patients with loss-of-function (LoF) mutations in the filaggrin gene (FLG), the prevalence of AD at 6 and 12 months seemed to be a higher than among patients with the wild-type gene, but the difference did not reach statistical significance.
Commenting on this finding, Dr. Hebert noted that LoF FLG mutation carriers may benefit especially from emollient use, given that LoF mutations in FLG is associated with reduced production of natural moisturizing factors in the skin.
Regarding future research directions, Dr. Hourihane stated that there is a need for replication and validation of the findings in studies that include infants from different ethnic backgrounds as well as those from various social settings. These studies should also include variable treatment windows to determine both short- and longer-term effects of emollient use in this population, Dr. Hourihane explained.
Dr. Hourihane added that he and the investigators do not yet understand which aspect of the study’s program was key for reducing the incidence of AD in the first year of life. “The timing of emollient initiation, the duration of treatment, the products, or maybe just a combination of these” could be possible explanations.
The study was independently supported. Dr. Hourihand reported receiving grant funding from Aimmune Therapeutics and DBV Technologies. Dr. Hebert reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ALLERGY