User login
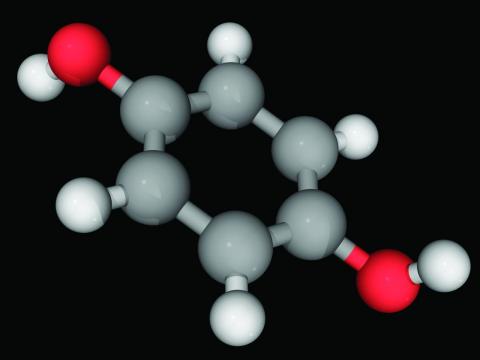
Hydroquinone, found in skin-lightening agents worldwide, linked with increased skin cancer risk
an analysis of records from a large research database suggests.
In the study, hydroquinone use was associated with an approximately threefold increase for skin cancer risk, coauthor Brittany Miles, a fourth-year medical student at the University of Texas Medical Branch at Galveston’s John Sealy School of Medicine, told this news organization. “The magnitude of the risk was surprising. Increased risk should be disclosed to patients considering hydroquinone treatment.”
The results of the study were presented in a poster at the annual meeting of the Society for Investigative Dermatology.
Hydroquinone (multiple brand names), a tyrosinase inhibitor used worldwide for skin lightening because of its inhibition of melanin production, was once considered “generally safe and effective” by the Food and Drug Administration, the authors wrote.
The compound’s use in over-the-counter products in the United States has been restricted based on suspicion of carcinogenicity, but few human studies have been conducted. In April, the FDA issued warning letters to 12 companies that sold hydroquinone in concentrations not generally recognized as safe and effective, because of other concerns including rashes, facial swelling, and ochronosis (skin discoloration).
Ms. Miles and her coauthor, Michael Wilkerson, MD, professor and chair of the department of dermatology at UTMB, analyzed data from TriNetX, the medical research database of anonymized medical record information from 61 million patients in 57 large health care organizations, almost all of them in the United States.
The researchers created two cohorts of patients aged 15 years and older with no prior diagnosis of skin cancer: one group had been treated with hydroquinone (medication code 5509 in the TriNetX system), and the other had not been exposed to the drug. Using ICD-10 codes for melanoma, nonmelanoma skin cancer, and all skin cancers, they investigated which groups of people were likely to develop these cancers.
They found that hydroquinone exposure was linked with a significant increase in melanoma (relative risk, 3.0; 95% confidence interval, 1.704-5.281; P < .0001), nonmelanoma skin cancers (RR, 3.6; 95%; CI, 2.815-4.561; P < .0001), and all reported skin cancers combined (relative risk, 3.4; 95% CI, 2.731-4.268; P < .0001)
While “the source of the data and the number of patients in the study are significant strengths,” Ms. Miles said, “the inability to determine how long and how consistently the patients used hydroquinone is likely the biggest weakness.”
Skin lightening is big business and more research is needed
“The U.S. market for skin-lightening agents was approximately 330 million dollars in 2021, and 330,000 prescriptions containing hydroquinone were dispensed in 2019,” Ms. Miles said.
Valencia D. Thomas, MD, professor in the department of dermatology of the University of Texas MD Anderson Cancer Center, Houston, said in an email that over-the-counter skin-lightening products containing low-concentration hydroquinone are in widespread use and are commonly used in populations of color.
“Hydroquinone preparations in higher concentrations are unfortunately also available in the United States,” added Dr. Thomas, who was not involved in the study and referred to the FDA warning letter issued in April.
Only one hydroquinone-containing medication – Tri-Luma at 4% concentration, used to treat melasma – is currently FDA-approved, she said.
The data in the study do not show an increased risk for skin cancer with hydroquinone exposure, but do show “an increased risk of cancer in the TriNetX medication code 5509 hydroquinone exposure group, which does not prove causation,” Dr. Thomas commented.
“Because ‘hydroquinone exposure’ is not defined, it is unclear how TriNetX identified the hydroquinone exposure cohort,” she noted. “Does ‘exposure’ count prescriptions written and potentially not used, the use of hydroquinone products of high concentration not approved by the FDA, or the use of over-the-counter hydroquinone products?
“The strength of this study is its size,” Dr. Thomas acknowledged. “This study is a wonderful starting point to further investigate the ‘hydroquinone exposure’ cohort to determine if hydroquinone is a driver of cancer, or if hydroquinone is itself a confounder.”
These results highlight the need to examine the social determinants of health that may explain increased risk for cancer, including race, geography, and poverty, she added.
“Given the global consumption of hydroquinone, multinational collaboration investigating hydroquinone and cancer data will likely be needed to provide insight into this continuing question,” Dr. Thomas advised.
Christiane Querfeld, MD, PhD, associate professor of dermatology and dermatopathology at City of Hope in Duarte, Calif., agreed that the occurrence of skin cancer following use of hydroquinone is largely understudied.
“The findings have a huge impact on how we counsel and monitor future patients,” Dr. Querfeld, who also was not involved in the study, said in an email. “There may be a trade-off at the start of treatment: Get rid of melasma but develop a skin cancer or melanoma with potentially severe outcomes.
“It remains to be seen if there is a higher incidence of skin cancer following use of hydroquinone or other voluntary bleaching and depigmentation remedies in ethnic groups such as African American or Hispanic patient populations, who have historically been at low risk of developing skin cancer,” she added. “It also remains to be seen if increased risk is due to direct effects or to indirect effects on already-photodamaged skin.
“These data are critical, and I am sure this will open further investigations to study effects in more detail,” Dr. Querfeld said.
The study authors, Dr. Thomas, and Dr. Querfeld reported no relevant financial relationships. The study did not receive external funding.
A version of this article first appeared on Medscape.com.
an analysis of records from a large research database suggests.
In the study, hydroquinone use was associated with an approximately threefold increase for skin cancer risk, coauthor Brittany Miles, a fourth-year medical student at the University of Texas Medical Branch at Galveston’s John Sealy School of Medicine, told this news organization. “The magnitude of the risk was surprising. Increased risk should be disclosed to patients considering hydroquinone treatment.”
The results of the study were presented in a poster at the annual meeting of the Society for Investigative Dermatology.
Hydroquinone (multiple brand names), a tyrosinase inhibitor used worldwide for skin lightening because of its inhibition of melanin production, was once considered “generally safe and effective” by the Food and Drug Administration, the authors wrote.
The compound’s use in over-the-counter products in the United States has been restricted based on suspicion of carcinogenicity, but few human studies have been conducted. In April, the FDA issued warning letters to 12 companies that sold hydroquinone in concentrations not generally recognized as safe and effective, because of other concerns including rashes, facial swelling, and ochronosis (skin discoloration).
Ms. Miles and her coauthor, Michael Wilkerson, MD, professor and chair of the department of dermatology at UTMB, analyzed data from TriNetX, the medical research database of anonymized medical record information from 61 million patients in 57 large health care organizations, almost all of them in the United States.
The researchers created two cohorts of patients aged 15 years and older with no prior diagnosis of skin cancer: one group had been treated with hydroquinone (medication code 5509 in the TriNetX system), and the other had not been exposed to the drug. Using ICD-10 codes for melanoma, nonmelanoma skin cancer, and all skin cancers, they investigated which groups of people were likely to develop these cancers.
They found that hydroquinone exposure was linked with a significant increase in melanoma (relative risk, 3.0; 95% confidence interval, 1.704-5.281; P < .0001), nonmelanoma skin cancers (RR, 3.6; 95%; CI, 2.815-4.561; P < .0001), and all reported skin cancers combined (relative risk, 3.4; 95% CI, 2.731-4.268; P < .0001)
While “the source of the data and the number of patients in the study are significant strengths,” Ms. Miles said, “the inability to determine how long and how consistently the patients used hydroquinone is likely the biggest weakness.”
Skin lightening is big business and more research is needed
“The U.S. market for skin-lightening agents was approximately 330 million dollars in 2021, and 330,000 prescriptions containing hydroquinone were dispensed in 2019,” Ms. Miles said.
Valencia D. Thomas, MD, professor in the department of dermatology of the University of Texas MD Anderson Cancer Center, Houston, said in an email that over-the-counter skin-lightening products containing low-concentration hydroquinone are in widespread use and are commonly used in populations of color.
“Hydroquinone preparations in higher concentrations are unfortunately also available in the United States,” added Dr. Thomas, who was not involved in the study and referred to the FDA warning letter issued in April.
Only one hydroquinone-containing medication – Tri-Luma at 4% concentration, used to treat melasma – is currently FDA-approved, she said.
The data in the study do not show an increased risk for skin cancer with hydroquinone exposure, but do show “an increased risk of cancer in the TriNetX medication code 5509 hydroquinone exposure group, which does not prove causation,” Dr. Thomas commented.
“Because ‘hydroquinone exposure’ is not defined, it is unclear how TriNetX identified the hydroquinone exposure cohort,” she noted. “Does ‘exposure’ count prescriptions written and potentially not used, the use of hydroquinone products of high concentration not approved by the FDA, or the use of over-the-counter hydroquinone products?
“The strength of this study is its size,” Dr. Thomas acknowledged. “This study is a wonderful starting point to further investigate the ‘hydroquinone exposure’ cohort to determine if hydroquinone is a driver of cancer, or if hydroquinone is itself a confounder.”
These results highlight the need to examine the social determinants of health that may explain increased risk for cancer, including race, geography, and poverty, she added.
“Given the global consumption of hydroquinone, multinational collaboration investigating hydroquinone and cancer data will likely be needed to provide insight into this continuing question,” Dr. Thomas advised.
Christiane Querfeld, MD, PhD, associate professor of dermatology and dermatopathology at City of Hope in Duarte, Calif., agreed that the occurrence of skin cancer following use of hydroquinone is largely understudied.
“The findings have a huge impact on how we counsel and monitor future patients,” Dr. Querfeld, who also was not involved in the study, said in an email. “There may be a trade-off at the start of treatment: Get rid of melasma but develop a skin cancer or melanoma with potentially severe outcomes.
“It remains to be seen if there is a higher incidence of skin cancer following use of hydroquinone or other voluntary bleaching and depigmentation remedies in ethnic groups such as African American or Hispanic patient populations, who have historically been at low risk of developing skin cancer,” she added. “It also remains to be seen if increased risk is due to direct effects or to indirect effects on already-photodamaged skin.
“These data are critical, and I am sure this will open further investigations to study effects in more detail,” Dr. Querfeld said.
The study authors, Dr. Thomas, and Dr. Querfeld reported no relevant financial relationships. The study did not receive external funding.
A version of this article first appeared on Medscape.com.
an analysis of records from a large research database suggests.
In the study, hydroquinone use was associated with an approximately threefold increase for skin cancer risk, coauthor Brittany Miles, a fourth-year medical student at the University of Texas Medical Branch at Galveston’s John Sealy School of Medicine, told this news organization. “The magnitude of the risk was surprising. Increased risk should be disclosed to patients considering hydroquinone treatment.”
The results of the study were presented in a poster at the annual meeting of the Society for Investigative Dermatology.
Hydroquinone (multiple brand names), a tyrosinase inhibitor used worldwide for skin lightening because of its inhibition of melanin production, was once considered “generally safe and effective” by the Food and Drug Administration, the authors wrote.
The compound’s use in over-the-counter products in the United States has been restricted based on suspicion of carcinogenicity, but few human studies have been conducted. In April, the FDA issued warning letters to 12 companies that sold hydroquinone in concentrations not generally recognized as safe and effective, because of other concerns including rashes, facial swelling, and ochronosis (skin discoloration).
Ms. Miles and her coauthor, Michael Wilkerson, MD, professor and chair of the department of dermatology at UTMB, analyzed data from TriNetX, the medical research database of anonymized medical record information from 61 million patients in 57 large health care organizations, almost all of them in the United States.
The researchers created two cohorts of patients aged 15 years and older with no prior diagnosis of skin cancer: one group had been treated with hydroquinone (medication code 5509 in the TriNetX system), and the other had not been exposed to the drug. Using ICD-10 codes for melanoma, nonmelanoma skin cancer, and all skin cancers, they investigated which groups of people were likely to develop these cancers.
They found that hydroquinone exposure was linked with a significant increase in melanoma (relative risk, 3.0; 95% confidence interval, 1.704-5.281; P < .0001), nonmelanoma skin cancers (RR, 3.6; 95%; CI, 2.815-4.561; P < .0001), and all reported skin cancers combined (relative risk, 3.4; 95% CI, 2.731-4.268; P < .0001)
While “the source of the data and the number of patients in the study are significant strengths,” Ms. Miles said, “the inability to determine how long and how consistently the patients used hydroquinone is likely the biggest weakness.”
Skin lightening is big business and more research is needed
“The U.S. market for skin-lightening agents was approximately 330 million dollars in 2021, and 330,000 prescriptions containing hydroquinone were dispensed in 2019,” Ms. Miles said.
Valencia D. Thomas, MD, professor in the department of dermatology of the University of Texas MD Anderson Cancer Center, Houston, said in an email that over-the-counter skin-lightening products containing low-concentration hydroquinone are in widespread use and are commonly used in populations of color.
“Hydroquinone preparations in higher concentrations are unfortunately also available in the United States,” added Dr. Thomas, who was not involved in the study and referred to the FDA warning letter issued in April.
Only one hydroquinone-containing medication – Tri-Luma at 4% concentration, used to treat melasma – is currently FDA-approved, she said.
The data in the study do not show an increased risk for skin cancer with hydroquinone exposure, but do show “an increased risk of cancer in the TriNetX medication code 5509 hydroquinone exposure group, which does not prove causation,” Dr. Thomas commented.
“Because ‘hydroquinone exposure’ is not defined, it is unclear how TriNetX identified the hydroquinone exposure cohort,” she noted. “Does ‘exposure’ count prescriptions written and potentially not used, the use of hydroquinone products of high concentration not approved by the FDA, or the use of over-the-counter hydroquinone products?
“The strength of this study is its size,” Dr. Thomas acknowledged. “This study is a wonderful starting point to further investigate the ‘hydroquinone exposure’ cohort to determine if hydroquinone is a driver of cancer, or if hydroquinone is itself a confounder.”
These results highlight the need to examine the social determinants of health that may explain increased risk for cancer, including race, geography, and poverty, she added.
“Given the global consumption of hydroquinone, multinational collaboration investigating hydroquinone and cancer data will likely be needed to provide insight into this continuing question,” Dr. Thomas advised.
Christiane Querfeld, MD, PhD, associate professor of dermatology and dermatopathology at City of Hope in Duarte, Calif., agreed that the occurrence of skin cancer following use of hydroquinone is largely understudied.
“The findings have a huge impact on how we counsel and monitor future patients,” Dr. Querfeld, who also was not involved in the study, said in an email. “There may be a trade-off at the start of treatment: Get rid of melasma but develop a skin cancer or melanoma with potentially severe outcomes.
“It remains to be seen if there is a higher incidence of skin cancer following use of hydroquinone or other voluntary bleaching and depigmentation remedies in ethnic groups such as African American or Hispanic patient populations, who have historically been at low risk of developing skin cancer,” she added. “It also remains to be seen if increased risk is due to direct effects or to indirect effects on already-photodamaged skin.
“These data are critical, and I am sure this will open further investigations to study effects in more detail,” Dr. Querfeld said.
The study authors, Dr. Thomas, and Dr. Querfeld reported no relevant financial relationships. The study did not receive external funding.
A version of this article first appeared on Medscape.com.
FROM SID 2022
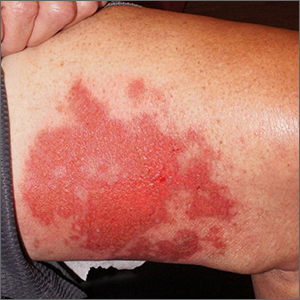
Leg rash
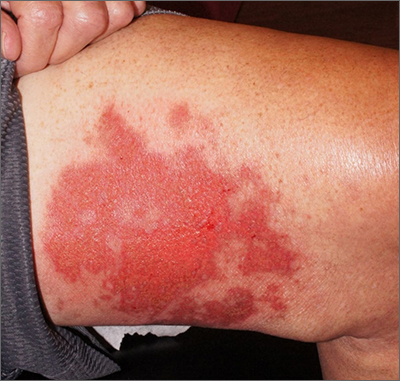
Punch biopsies for standard pathology and direct immunofluorescence were performed and ruled out vesiculobullous disease. Further conversation with the patient revealed that this was a phototoxic drug eruption that resulted from a medication mix-up. The patient had intended to treat an eczema flare with a topical steroid but had inadvertently applied 5-fluorouracil (5-FU), which he had left over from a previous bout of actinic keratosis. While selective to precancerous cells with rapid DNA replication, 5-FU can trigger a significant photodermatitis when applied to heavily sun-exposed skin.
Phototoxic skin reactions can be an adverse result of multiple systemic and topical therapies. Common systemic examples include amiodarone, chlorpromazine, doxycycline, hydrochlorothiazide, isotretinoin, nalidixic acid, naproxen, piroxicam, tetracycline, thioridazine, vemurafenib, and voriconazole.1 Topical examples include retinoids, levulinic acid, and 5-FU. Treatment requires that the patient stop the offending medication and use photoprotection. The patient followed this protocol and his erosions resolved over the course of a few weeks.
This case demonstrates that topical therapies, like systemic medications, can have chemical names that are confusing to patients. Further complicating matters can be the practice of folding metal tubes of cream over their life of use, thus obscuring the label.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Blakely KM, Drucker AM, Rosen CF. Drug-induced photosensitivity-an update: culprit drugs, prevention, and management. Drug Saf. 2019;42:827-847. doi: 10.1007/s40264-019-00806-5

Punch biopsies for standard pathology and direct immunofluorescence were performed and ruled out vesiculobullous disease. Further conversation with the patient revealed that this was a phototoxic drug eruption that resulted from a medication mix-up. The patient had intended to treat an eczema flare with a topical steroid but had inadvertently applied 5-fluorouracil (5-FU), which he had left over from a previous bout of actinic keratosis. While selective to precancerous cells with rapid DNA replication, 5-FU can trigger a significant photodermatitis when applied to heavily sun-exposed skin.
Phototoxic skin reactions can be an adverse result of multiple systemic and topical therapies. Common systemic examples include amiodarone, chlorpromazine, doxycycline, hydrochlorothiazide, isotretinoin, nalidixic acid, naproxen, piroxicam, tetracycline, thioridazine, vemurafenib, and voriconazole.1 Topical examples include retinoids, levulinic acid, and 5-FU. Treatment requires that the patient stop the offending medication and use photoprotection. The patient followed this protocol and his erosions resolved over the course of a few weeks.
This case demonstrates that topical therapies, like systemic medications, can have chemical names that are confusing to patients. Further complicating matters can be the practice of folding metal tubes of cream over their life of use, thus obscuring the label.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

Punch biopsies for standard pathology and direct immunofluorescence were performed and ruled out vesiculobullous disease. Further conversation with the patient revealed that this was a phototoxic drug eruption that resulted from a medication mix-up. The patient had intended to treat an eczema flare with a topical steroid but had inadvertently applied 5-fluorouracil (5-FU), which he had left over from a previous bout of actinic keratosis. While selective to precancerous cells with rapid DNA replication, 5-FU can trigger a significant photodermatitis when applied to heavily sun-exposed skin.
Phototoxic skin reactions can be an adverse result of multiple systemic and topical therapies. Common systemic examples include amiodarone, chlorpromazine, doxycycline, hydrochlorothiazide, isotretinoin, nalidixic acid, naproxen, piroxicam, tetracycline, thioridazine, vemurafenib, and voriconazole.1 Topical examples include retinoids, levulinic acid, and 5-FU. Treatment requires that the patient stop the offending medication and use photoprotection. The patient followed this protocol and his erosions resolved over the course of a few weeks.
This case demonstrates that topical therapies, like systemic medications, can have chemical names that are confusing to patients. Further complicating matters can be the practice of folding metal tubes of cream over their life of use, thus obscuring the label.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Blakely KM, Drucker AM, Rosen CF. Drug-induced photosensitivity-an update: culprit drugs, prevention, and management. Drug Saf. 2019;42:827-847. doi: 10.1007/s40264-019-00806-5
1. Blakely KM, Drucker AM, Rosen CF. Drug-induced photosensitivity-an update: culprit drugs, prevention, and management. Drug Saf. 2019;42:827-847. doi: 10.1007/s40264-019-00806-5
Chlorophyll water can trigger pseudoporphyria, expert warns
PORTLAND, ORE. – If a child presents with pseudoporphyria – a bullous photodermatosis with the clinical and histological features of porphyria cutanea tarda (PCT) but with normal porphyrins – chlorophyll water could be the culprit.
Commercially available, green pigment–infused chlorophyll water is marketed with claims that it supports cancer prevention and digestive health, facilitates weight loss, and improves skin complexion. “It also absorbs light, so lo and behold, if your patient is photosensitive, they might get pseudoporphyria,” Robert Sidbury, MD, MPH, chief of the division of dermatology at Seattle Children’s Hospital, said at the annual meeting of the Pacific Dermatologic Association.
This was one of the clinical pearls he shared during his presentation.
Dr. Sidbury added that the risk of photosensitivity increases in children who are taking other medications such as doxycycline, methotrexate, or even naproxen. At least two cases of pseudoporphyria following self-medication with chlorophyll have been described in the dermatology literature.
Is it SSSS or SJS?
Another clinical pearl that Dr. Sidbury shared at the meeting related to staphylococcal scalded skin syndrome (SSSS), which causes reddening and blistering of the skin that makes it appear scalded or burned. To rule out Stevens-Johnson Syndrome (SJS) in a child who presents with such skin manifestations, he routinely performs the unscientific lollipop test, which he learned from Bernard A. “Buddy” Cohen, MD, professor of dermatology and pediatrics at Johns Hopkins University, Baltimore.
“If they eat it, it’s Staph scalded skin,” said Dr. Sidbury, who is also professor of pediatrics at the University of Washington, Seattle. “If they don’t, it’s likely SJS. It’s not the most specific test, but it’s easy to do, because there’s no mucous membrane involvement in Staph scalded skin.”
In a poster presented during the 2022 annual meeting of the Society for Pediatric Dermatology, Sarah Cipriano, MD, MPH, and colleagues at the University of Utah, Salt Lake City, retrospectively study 85 patients aged younger than 18 years diagnosed with SSSS between Jan. 1, 2010, and Aug. 21, 2021. They found that ancillary blood cultures and CSF cultures did not improve diagnostic precision in SSSS patients.
“They don’t add anything unless there’s an indication beyond the Staph scalded skin,” said Dr. Sidbury, who was not involved in the study. “The researchers also found that clindamycin does not improve outcomes in these patients, so avoid using it.” Instead, a first-generation cephalosporin is indicated, and an alternate diagnosis should be considered if the patient does not improve within 48 hours.
Dr. Sidbury disclosed that he has conducted research for Regeneron, Galderma, and UCB. He is also an adviser for Leo Pharmaceuticals and a speaker for Biersdorf.
PORTLAND, ORE. – If a child presents with pseudoporphyria – a bullous photodermatosis with the clinical and histological features of porphyria cutanea tarda (PCT) but with normal porphyrins – chlorophyll water could be the culprit.
Commercially available, green pigment–infused chlorophyll water is marketed with claims that it supports cancer prevention and digestive health, facilitates weight loss, and improves skin complexion. “It also absorbs light, so lo and behold, if your patient is photosensitive, they might get pseudoporphyria,” Robert Sidbury, MD, MPH, chief of the division of dermatology at Seattle Children’s Hospital, said at the annual meeting of the Pacific Dermatologic Association.
This was one of the clinical pearls he shared during his presentation.
Dr. Sidbury added that the risk of photosensitivity increases in children who are taking other medications such as doxycycline, methotrexate, or even naproxen. At least two cases of pseudoporphyria following self-medication with chlorophyll have been described in the dermatology literature.
Is it SSSS or SJS?
Another clinical pearl that Dr. Sidbury shared at the meeting related to staphylococcal scalded skin syndrome (SSSS), which causes reddening and blistering of the skin that makes it appear scalded or burned. To rule out Stevens-Johnson Syndrome (SJS) in a child who presents with such skin manifestations, he routinely performs the unscientific lollipop test, which he learned from Bernard A. “Buddy” Cohen, MD, professor of dermatology and pediatrics at Johns Hopkins University, Baltimore.
“If they eat it, it’s Staph scalded skin,” said Dr. Sidbury, who is also professor of pediatrics at the University of Washington, Seattle. “If they don’t, it’s likely SJS. It’s not the most specific test, but it’s easy to do, because there’s no mucous membrane involvement in Staph scalded skin.”
In a poster presented during the 2022 annual meeting of the Society for Pediatric Dermatology, Sarah Cipriano, MD, MPH, and colleagues at the University of Utah, Salt Lake City, retrospectively study 85 patients aged younger than 18 years diagnosed with SSSS between Jan. 1, 2010, and Aug. 21, 2021. They found that ancillary blood cultures and CSF cultures did not improve diagnostic precision in SSSS patients.
“They don’t add anything unless there’s an indication beyond the Staph scalded skin,” said Dr. Sidbury, who was not involved in the study. “The researchers also found that clindamycin does not improve outcomes in these patients, so avoid using it.” Instead, a first-generation cephalosporin is indicated, and an alternate diagnosis should be considered if the patient does not improve within 48 hours.
Dr. Sidbury disclosed that he has conducted research for Regeneron, Galderma, and UCB. He is also an adviser for Leo Pharmaceuticals and a speaker for Biersdorf.
PORTLAND, ORE. – If a child presents with pseudoporphyria – a bullous photodermatosis with the clinical and histological features of porphyria cutanea tarda (PCT) but with normal porphyrins – chlorophyll water could be the culprit.
Commercially available, green pigment–infused chlorophyll water is marketed with claims that it supports cancer prevention and digestive health, facilitates weight loss, and improves skin complexion. “It also absorbs light, so lo and behold, if your patient is photosensitive, they might get pseudoporphyria,” Robert Sidbury, MD, MPH, chief of the division of dermatology at Seattle Children’s Hospital, said at the annual meeting of the Pacific Dermatologic Association.
This was one of the clinical pearls he shared during his presentation.
Dr. Sidbury added that the risk of photosensitivity increases in children who are taking other medications such as doxycycline, methotrexate, or even naproxen. At least two cases of pseudoporphyria following self-medication with chlorophyll have been described in the dermatology literature.
Is it SSSS or SJS?
Another clinical pearl that Dr. Sidbury shared at the meeting related to staphylococcal scalded skin syndrome (SSSS), which causes reddening and blistering of the skin that makes it appear scalded or burned. To rule out Stevens-Johnson Syndrome (SJS) in a child who presents with such skin manifestations, he routinely performs the unscientific lollipop test, which he learned from Bernard A. “Buddy” Cohen, MD, professor of dermatology and pediatrics at Johns Hopkins University, Baltimore.
“If they eat it, it’s Staph scalded skin,” said Dr. Sidbury, who is also professor of pediatrics at the University of Washington, Seattle. “If they don’t, it’s likely SJS. It’s not the most specific test, but it’s easy to do, because there’s no mucous membrane involvement in Staph scalded skin.”
In a poster presented during the 2022 annual meeting of the Society for Pediatric Dermatology, Sarah Cipriano, MD, MPH, and colleagues at the University of Utah, Salt Lake City, retrospectively study 85 patients aged younger than 18 years diagnosed with SSSS between Jan. 1, 2010, and Aug. 21, 2021. They found that ancillary blood cultures and CSF cultures did not improve diagnostic precision in SSSS patients.
“They don’t add anything unless there’s an indication beyond the Staph scalded skin,” said Dr. Sidbury, who was not involved in the study. “The researchers also found that clindamycin does not improve outcomes in these patients, so avoid using it.” Instead, a first-generation cephalosporin is indicated, and an alternate diagnosis should be considered if the patient does not improve within 48 hours.
Dr. Sidbury disclosed that he has conducted research for Regeneron, Galderma, and UCB. He is also an adviser for Leo Pharmaceuticals and a speaker for Biersdorf.
AT PDA 2022
VTE risk not elevated in AD patients on JAK inhibitors: Study
, according to a new systemic review and meta-analysis, published online in JAMA Dermatology.
“These findings may provide a reference for clinicians in prescribing JAK inhibitors for patients with AD,” Tai-Li Chen, MD, of Taipei (Taiwan) Veterans General Hospital, Taipei, and colleagues wrote in the study.
The results shed some welcome light on treatment for this dermatologic population, for whom enthusiasm about JAK inhibitors was dampened by the addition of a boxed warning to the labels of JAK inhibitors last year, required by the Food and Drug Administration. The warning, which describes an increased risk of “serious heart-related events such as heart attack or stroke, cancer, blood clots, and death” was triggered by results of the ORAL Surveillance study of patients with rheumatoid arthritis (RA) treated with tofacitinib.
The boxed warning is also included in the labels of topical ruxolitinib, a JAK inhibitor approved by the FDA for mild to moderate AD in 2021, and in the labels of two oral JAK inhibitors, upadacitinib and abrocitinib, approved by the FDA for treating moderate to severe AD in January 2022.
Despite the new findings, some dermatologists are still urging caution.
“All the JAK inhibitor trials are short term. I still think the precautionary principle applies and we need to counsel on the risks of JAKs,” tweeted Aaron Drucker, MD, a dermatologist at Women’s College Hospital, and associate professor at the University of Toronto. “It is great to have these as options for our patients. But we need to be aware of the risks associated with this class of medications, counsel patients about them when we are informing them of the risks and benefits of treatment options, and wait for more data specific to this population to make even more informed decisions,” he told this news organization.
The meta-analysis examined both the risk of incident VTE in untreated patients with AD compared with non-AD patients, as well as the risk of VTE in AD patients treated with JAK inhibitors compared with those on either placebo or dupilumab. Four JAK inhibitors were studied: abrocitinib, baricitinib (under FDA review for AD), upadacitinib, and SHR0302 (in clinical trials).
Two studies (458,206 participants) found the overall incidence rate of VTE for patients with AD was 0.23 events per 100 patient-years. The risk was did not differ from that in non-AD patients (pooled hazard ratio [HR], 0.95; 95% confidence interval [CI], 0.62-1.45).
Another 15 studies included 8,787 participants with AD and found no significant differences in the rates of VTE in AD patients treated with JAK inhibitors (0.05%) versus those treated with placebo or dupilumab (0.03%). However “with the increasing applications of JAK inhibitors in AD, more clinical data are needed to identify patients at high risk for VTE,” noted the authors.
“We need more, long-term data,” agreed Dr. Drucker, adding that a major issue is the short-term nature of AD trials to date (generally up to 16 weeks), which “don’t provide adequate reassurance.” He said although the FDA’s boxed warning was prompted by a trial in RA patients treated with tofacitinib (a less selective JAK inhibitor than those approved by the FDA for AD), and the same risks have not been demonstrated specifically for the JAK inhibitors used for a patients with AD, he still remains cautious.
While agreeing on the need for more long-term data, Andrew Blauvelt, MD, MBA, president of Oregon Medical Research Center, Portland, said that the new findings should “provide reassurance” to dermatologists and are “consonant with recent published meta-analyses reporting no increased VTE risk in patients with psoriasis, RA, or inflammatory bowel disease treated with JAK inhibitors” in Arthritis & Rheumatology, and Mayo Clinic Proceedings.
In an interview, Dr. Blauvelt said that safety profiles emerging for the newer JAK inhibitors, which block JAK 1/2, have been overshadowed by the older RA data for tofacitinib – which is a JAK 1/3 inhibitor, “despite emerging long-term, monotherapy, clinical study data for dermatologic diseases showing no or rare risks of developing severe adverse events outlined in the boxed warnings.”
Both Dr. Blauvelt and Dr. Drucker pointed out that people with RA tend to have more comorbidities than those with AD that would predispose them to adverse events. In fact, “approximately 75% of patients in the ORAL Surveillance study were also on concomitant methotrexate and/or prednisone, which can greatly confound safety results,” said Dr. Blauvelt.
The study authors did not report any disclosures. No funding source for the study was provided. Dr. Drucker has no relevant disclosures. Dr. Blauvelt has been a clinical study investigator in trials for AD treatments, including JAK inhibitors; his disclosures include serving as a speaker, scientific adviser, and/or clinical study investigator for multiple companies including AbbVie, Arcutis, Bristol-Myers Squibb, Pfizer, Incyte, Regeneron, Sanofi Genzyme, and UCB Pharma.
, according to a new systemic review and meta-analysis, published online in JAMA Dermatology.
“These findings may provide a reference for clinicians in prescribing JAK inhibitors for patients with AD,” Tai-Li Chen, MD, of Taipei (Taiwan) Veterans General Hospital, Taipei, and colleagues wrote in the study.
The results shed some welcome light on treatment for this dermatologic population, for whom enthusiasm about JAK inhibitors was dampened by the addition of a boxed warning to the labels of JAK inhibitors last year, required by the Food and Drug Administration. The warning, which describes an increased risk of “serious heart-related events such as heart attack or stroke, cancer, blood clots, and death” was triggered by results of the ORAL Surveillance study of patients with rheumatoid arthritis (RA) treated with tofacitinib.
The boxed warning is also included in the labels of topical ruxolitinib, a JAK inhibitor approved by the FDA for mild to moderate AD in 2021, and in the labels of two oral JAK inhibitors, upadacitinib and abrocitinib, approved by the FDA for treating moderate to severe AD in January 2022.
Despite the new findings, some dermatologists are still urging caution.
“All the JAK inhibitor trials are short term. I still think the precautionary principle applies and we need to counsel on the risks of JAKs,” tweeted Aaron Drucker, MD, a dermatologist at Women’s College Hospital, and associate professor at the University of Toronto. “It is great to have these as options for our patients. But we need to be aware of the risks associated with this class of medications, counsel patients about them when we are informing them of the risks and benefits of treatment options, and wait for more data specific to this population to make even more informed decisions,” he told this news organization.
The meta-analysis examined both the risk of incident VTE in untreated patients with AD compared with non-AD patients, as well as the risk of VTE in AD patients treated with JAK inhibitors compared with those on either placebo or dupilumab. Four JAK inhibitors were studied: abrocitinib, baricitinib (under FDA review for AD), upadacitinib, and SHR0302 (in clinical trials).
Two studies (458,206 participants) found the overall incidence rate of VTE for patients with AD was 0.23 events per 100 patient-years. The risk was did not differ from that in non-AD patients (pooled hazard ratio [HR], 0.95; 95% confidence interval [CI], 0.62-1.45).
Another 15 studies included 8,787 participants with AD and found no significant differences in the rates of VTE in AD patients treated with JAK inhibitors (0.05%) versus those treated with placebo or dupilumab (0.03%). However “with the increasing applications of JAK inhibitors in AD, more clinical data are needed to identify patients at high risk for VTE,” noted the authors.
“We need more, long-term data,” agreed Dr. Drucker, adding that a major issue is the short-term nature of AD trials to date (generally up to 16 weeks), which “don’t provide adequate reassurance.” He said although the FDA’s boxed warning was prompted by a trial in RA patients treated with tofacitinib (a less selective JAK inhibitor than those approved by the FDA for AD), and the same risks have not been demonstrated specifically for the JAK inhibitors used for a patients with AD, he still remains cautious.
While agreeing on the need for more long-term data, Andrew Blauvelt, MD, MBA, president of Oregon Medical Research Center, Portland, said that the new findings should “provide reassurance” to dermatologists and are “consonant with recent published meta-analyses reporting no increased VTE risk in patients with psoriasis, RA, or inflammatory bowel disease treated with JAK inhibitors” in Arthritis & Rheumatology, and Mayo Clinic Proceedings.
In an interview, Dr. Blauvelt said that safety profiles emerging for the newer JAK inhibitors, which block JAK 1/2, have been overshadowed by the older RA data for tofacitinib – which is a JAK 1/3 inhibitor, “despite emerging long-term, monotherapy, clinical study data for dermatologic diseases showing no or rare risks of developing severe adverse events outlined in the boxed warnings.”
Both Dr. Blauvelt and Dr. Drucker pointed out that people with RA tend to have more comorbidities than those with AD that would predispose them to adverse events. In fact, “approximately 75% of patients in the ORAL Surveillance study were also on concomitant methotrexate and/or prednisone, which can greatly confound safety results,” said Dr. Blauvelt.
The study authors did not report any disclosures. No funding source for the study was provided. Dr. Drucker has no relevant disclosures. Dr. Blauvelt has been a clinical study investigator in trials for AD treatments, including JAK inhibitors; his disclosures include serving as a speaker, scientific adviser, and/or clinical study investigator for multiple companies including AbbVie, Arcutis, Bristol-Myers Squibb, Pfizer, Incyte, Regeneron, Sanofi Genzyme, and UCB Pharma.
, according to a new systemic review and meta-analysis, published online in JAMA Dermatology.
“These findings may provide a reference for clinicians in prescribing JAK inhibitors for patients with AD,” Tai-Li Chen, MD, of Taipei (Taiwan) Veterans General Hospital, Taipei, and colleagues wrote in the study.
The results shed some welcome light on treatment for this dermatologic population, for whom enthusiasm about JAK inhibitors was dampened by the addition of a boxed warning to the labels of JAK inhibitors last year, required by the Food and Drug Administration. The warning, which describes an increased risk of “serious heart-related events such as heart attack or stroke, cancer, blood clots, and death” was triggered by results of the ORAL Surveillance study of patients with rheumatoid arthritis (RA) treated with tofacitinib.
The boxed warning is also included in the labels of topical ruxolitinib, a JAK inhibitor approved by the FDA for mild to moderate AD in 2021, and in the labels of two oral JAK inhibitors, upadacitinib and abrocitinib, approved by the FDA for treating moderate to severe AD in January 2022.
Despite the new findings, some dermatologists are still urging caution.
“All the JAK inhibitor trials are short term. I still think the precautionary principle applies and we need to counsel on the risks of JAKs,” tweeted Aaron Drucker, MD, a dermatologist at Women’s College Hospital, and associate professor at the University of Toronto. “It is great to have these as options for our patients. But we need to be aware of the risks associated with this class of medications, counsel patients about them when we are informing them of the risks and benefits of treatment options, and wait for more data specific to this population to make even more informed decisions,” he told this news organization.
The meta-analysis examined both the risk of incident VTE in untreated patients with AD compared with non-AD patients, as well as the risk of VTE in AD patients treated with JAK inhibitors compared with those on either placebo or dupilumab. Four JAK inhibitors were studied: abrocitinib, baricitinib (under FDA review for AD), upadacitinib, and SHR0302 (in clinical trials).
Two studies (458,206 participants) found the overall incidence rate of VTE for patients with AD was 0.23 events per 100 patient-years. The risk was did not differ from that in non-AD patients (pooled hazard ratio [HR], 0.95; 95% confidence interval [CI], 0.62-1.45).
Another 15 studies included 8,787 participants with AD and found no significant differences in the rates of VTE in AD patients treated with JAK inhibitors (0.05%) versus those treated with placebo or dupilumab (0.03%). However “with the increasing applications of JAK inhibitors in AD, more clinical data are needed to identify patients at high risk for VTE,” noted the authors.
“We need more, long-term data,” agreed Dr. Drucker, adding that a major issue is the short-term nature of AD trials to date (generally up to 16 weeks), which “don’t provide adequate reassurance.” He said although the FDA’s boxed warning was prompted by a trial in RA patients treated with tofacitinib (a less selective JAK inhibitor than those approved by the FDA for AD), and the same risks have not been demonstrated specifically for the JAK inhibitors used for a patients with AD, he still remains cautious.
While agreeing on the need for more long-term data, Andrew Blauvelt, MD, MBA, president of Oregon Medical Research Center, Portland, said that the new findings should “provide reassurance” to dermatologists and are “consonant with recent published meta-analyses reporting no increased VTE risk in patients with psoriasis, RA, or inflammatory bowel disease treated with JAK inhibitors” in Arthritis & Rheumatology, and Mayo Clinic Proceedings.
In an interview, Dr. Blauvelt said that safety profiles emerging for the newer JAK inhibitors, which block JAK 1/2, have been overshadowed by the older RA data for tofacitinib – which is a JAK 1/3 inhibitor, “despite emerging long-term, monotherapy, clinical study data for dermatologic diseases showing no or rare risks of developing severe adverse events outlined in the boxed warnings.”
Both Dr. Blauvelt and Dr. Drucker pointed out that people with RA tend to have more comorbidities than those with AD that would predispose them to adverse events. In fact, “approximately 75% of patients in the ORAL Surveillance study were also on concomitant methotrexate and/or prednisone, which can greatly confound safety results,” said Dr. Blauvelt.
The study authors did not report any disclosures. No funding source for the study was provided. Dr. Drucker has no relevant disclosures. Dr. Blauvelt has been a clinical study investigator in trials for AD treatments, including JAK inhibitors; his disclosures include serving as a speaker, scientific adviser, and/or clinical study investigator for multiple companies including AbbVie, Arcutis, Bristol-Myers Squibb, Pfizer, Incyte, Regeneron, Sanofi Genzyme, and UCB Pharma.
FROM JAMA DERMATOLOGY
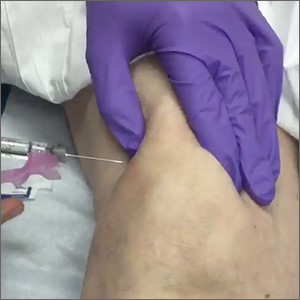
Superolateral knee injection with a patellar tilt for osteoarthritis pain



Autoimmune disease patients’ waxing, waning response to COVID vaccination studied in-depth
A new study in The Lancet Rheumatology examines the strength and duration of SARS-CoV-2 vaccine–induced immunoglobulin-G antibody responses over time for patients with a variety of autoimmune diseases, compared with healthy controls.
The presence of humoral antibodies to SARS-CoV-2 has been shown to correlate with protection against COVID infection. But for patients with immune-mediated inflammatory diseases (IMIDs), host response to COVID infection or to vaccination is affected by the immune dysfunction imposed by the IMID and by the use of immune-modulating drugs to treat it.
This new study finds a weaker – as shown previously – and less sustained immune response to SARS-CoV-2 vaccines in patients with a variety of IMIDs, including rheumatoid arthritis, spondyloarthritis, psoriasis, inflammatory bowel diseases, and other systemic autoimmune diseases such as lupus. It also points toward the possibility of adjusting treatment and vaccination schedules and strategies for these patients based on their antibody levels, among other factors, to preserve best protection against severe COVID.
“It is important to assess immune response in these patients to see if they still have protection against severe COVID infection,” said lead author David Simon, MD, senior clinical scientist in clinical immunology and rheumatology at University Hospital Erlangen (Germany). “We know that antibody response is an immune correlate. Therefore, it is important to see how large and durable the immune response is to the coronavirus vaccine in these IMID patients, and whether specific drugs or therapies have negative effects on their immune response.”
What was studied?
For this large prospective cohort study, researchers registered 5076 coronavirus-vaccinated individuals. They analyzed serum samples obtained between December 15, 2020, and December 1, 2021, from 2,535 patients diagnosed with IMIDs and participating in a prospective coronavirus study program at the Deutsches Zentrum Immuntherapie in Erlangen. The IMID patients had a mean age of 55.0 years, and 58.9% were women.
A healthy control group of 1,198 individuals without IMID who had a mean age of 40.7 years, including 53.8% men, was also recruited for the analysis. All approved coronavirus vaccines were included, following standard vaccination schedules. Antibody response was measured over time by an enzyme-linked immunosorbent assay from 8 weeks after first vaccination to week 40.
Among the findings, the healthy controls had higher postvaccine antibody levels than did those with IMIDs. But the majority of vaccinated patients with IMID were able to build up a humoral immune response to SARS-CoV-2. Patients who were taking B-cell inhibitors like rituximab (Rituxan, Genentech; and biosimilars) and T-cell inhibitors like abatacept (Orencia, Bristol Myers Squibb) for IMIDs had significantly poorer antibody response.
Greater age and the use of combination therapies for IMIDs, compared with monotherapy, further reduced immune response to the vaccine. In terms of vaccination modality, messenger RNA–based vaccines induced higher antibody levels than did vector-based vaccines. The researchers noted that patients with IMID who were given a third vaccine dose could actually catch up well with the antibody responses observed in healthy controls.
“We looked at whether different IMIDs had a different humoral response, and we also assessed if there are effects from different therapeutic strategies,” Dr. Simon explained. “It doesn’t matter so much what kind of IMID patients have; much more important is the specific drug treatment and its impact on their antibody response.” Some participants were advised to briefly stop taking some immunosuppressive treatments before or after vaccination.
One of Dr. Simon’s coauthors, statistician and rheumatologist Koray Tascilar, MD, added, “This research is important because we looked not only at who responded less, which has been previously established, but who are at greater risk of losing their immune response, and how quickly.”
Need to take care
“Most treatments we as rheumatologists give to our patients don’t affect their SARS-CoV-2 humoral response,” Dr. Simon said. “However, there are specific drugs that are associated with lower antibody response. With respect to those drugs, we have to be more careful.”
It is important to be able to tell patients which drugs are safe and won’t have a negative impact on their immune response to vaccinations, Dr. Tascilar said. “But it would be too strong to say we’re ready to choose therapies based on their potential impact on protection against COVID. Yes, there is a risk from catching COVID, but we need to balance that risk with the risk of not giving patients the medications that are necessary to treat their rheumatologic condition.”
These diseases are serious, sometimes life-threatening. “We might think of strategies for how to mitigate the risk of underprotection from COVID that is brought about by these treatments,” he said. For example, offering boosters sooner or more frequently, or prophylactically treating with monoclonal antibodies.
“This study, along other recent studies, has found that antibody levels in patients with immune-mediated diseases wane more rapidly than in healthy controls, and this is especially true of those on medications that interfere with the B and T cells and anticytokine therapies,” Rebecca Haberman, MD, assistant professor, division of rheumatology, New York University Langone Health, noted in an email to this news organization.
“While there is no known antibody level that specifically correlates with clinical protection, and each patient needs to be thought of individually, these findings support the use of supplemental booster dosing in patients with immune-mediated inflammatory diseases,” Dr. Haberman said, adding that her own research in this area has shown similar results.
“As a rheumatologist, I would be more likely to encourage my patients – especially those on immunomodulatory medications – to get boosted.”
Dr. Tascilar said his study does not directly answer the question of whether an earlier booster shot would be an effective strategy for patients with IMID. “In our department, we have an early boosting strategy, based on level of immune response.” But the decision of revaccination or not, and when, is based on a number of factors, not only on the level of antibodies. “It’s just part of the instruments we are using.”
The study was supported by the Deutsche Forschungsgemeinschaft. Dr. Simon and Dr. Tascilar declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study in The Lancet Rheumatology examines the strength and duration of SARS-CoV-2 vaccine–induced immunoglobulin-G antibody responses over time for patients with a variety of autoimmune diseases, compared with healthy controls.
The presence of humoral antibodies to SARS-CoV-2 has been shown to correlate with protection against COVID infection. But for patients with immune-mediated inflammatory diseases (IMIDs), host response to COVID infection or to vaccination is affected by the immune dysfunction imposed by the IMID and by the use of immune-modulating drugs to treat it.
This new study finds a weaker – as shown previously – and less sustained immune response to SARS-CoV-2 vaccines in patients with a variety of IMIDs, including rheumatoid arthritis, spondyloarthritis, psoriasis, inflammatory bowel diseases, and other systemic autoimmune diseases such as lupus. It also points toward the possibility of adjusting treatment and vaccination schedules and strategies for these patients based on their antibody levels, among other factors, to preserve best protection against severe COVID.
“It is important to assess immune response in these patients to see if they still have protection against severe COVID infection,” said lead author David Simon, MD, senior clinical scientist in clinical immunology and rheumatology at University Hospital Erlangen (Germany). “We know that antibody response is an immune correlate. Therefore, it is important to see how large and durable the immune response is to the coronavirus vaccine in these IMID patients, and whether specific drugs or therapies have negative effects on their immune response.”
What was studied?
For this large prospective cohort study, researchers registered 5076 coronavirus-vaccinated individuals. They analyzed serum samples obtained between December 15, 2020, and December 1, 2021, from 2,535 patients diagnosed with IMIDs and participating in a prospective coronavirus study program at the Deutsches Zentrum Immuntherapie in Erlangen. The IMID patients had a mean age of 55.0 years, and 58.9% were women.
A healthy control group of 1,198 individuals without IMID who had a mean age of 40.7 years, including 53.8% men, was also recruited for the analysis. All approved coronavirus vaccines were included, following standard vaccination schedules. Antibody response was measured over time by an enzyme-linked immunosorbent assay from 8 weeks after first vaccination to week 40.
Among the findings, the healthy controls had higher postvaccine antibody levels than did those with IMIDs. But the majority of vaccinated patients with IMID were able to build up a humoral immune response to SARS-CoV-2. Patients who were taking B-cell inhibitors like rituximab (Rituxan, Genentech; and biosimilars) and T-cell inhibitors like abatacept (Orencia, Bristol Myers Squibb) for IMIDs had significantly poorer antibody response.
Greater age and the use of combination therapies for IMIDs, compared with monotherapy, further reduced immune response to the vaccine. In terms of vaccination modality, messenger RNA–based vaccines induced higher antibody levels than did vector-based vaccines. The researchers noted that patients with IMID who were given a third vaccine dose could actually catch up well with the antibody responses observed in healthy controls.
“We looked at whether different IMIDs had a different humoral response, and we also assessed if there are effects from different therapeutic strategies,” Dr. Simon explained. “It doesn’t matter so much what kind of IMID patients have; much more important is the specific drug treatment and its impact on their antibody response.” Some participants were advised to briefly stop taking some immunosuppressive treatments before or after vaccination.
One of Dr. Simon’s coauthors, statistician and rheumatologist Koray Tascilar, MD, added, “This research is important because we looked not only at who responded less, which has been previously established, but who are at greater risk of losing their immune response, and how quickly.”
Need to take care
“Most treatments we as rheumatologists give to our patients don’t affect their SARS-CoV-2 humoral response,” Dr. Simon said. “However, there are specific drugs that are associated with lower antibody response. With respect to those drugs, we have to be more careful.”
It is important to be able to tell patients which drugs are safe and won’t have a negative impact on their immune response to vaccinations, Dr. Tascilar said. “But it would be too strong to say we’re ready to choose therapies based on their potential impact on protection against COVID. Yes, there is a risk from catching COVID, but we need to balance that risk with the risk of not giving patients the medications that are necessary to treat their rheumatologic condition.”
These diseases are serious, sometimes life-threatening. “We might think of strategies for how to mitigate the risk of underprotection from COVID that is brought about by these treatments,” he said. For example, offering boosters sooner or more frequently, or prophylactically treating with monoclonal antibodies.
“This study, along other recent studies, has found that antibody levels in patients with immune-mediated diseases wane more rapidly than in healthy controls, and this is especially true of those on medications that interfere with the B and T cells and anticytokine therapies,” Rebecca Haberman, MD, assistant professor, division of rheumatology, New York University Langone Health, noted in an email to this news organization.
“While there is no known antibody level that specifically correlates with clinical protection, and each patient needs to be thought of individually, these findings support the use of supplemental booster dosing in patients with immune-mediated inflammatory diseases,” Dr. Haberman said, adding that her own research in this area has shown similar results.
“As a rheumatologist, I would be more likely to encourage my patients – especially those on immunomodulatory medications – to get boosted.”
Dr. Tascilar said his study does not directly answer the question of whether an earlier booster shot would be an effective strategy for patients with IMID. “In our department, we have an early boosting strategy, based on level of immune response.” But the decision of revaccination or not, and when, is based on a number of factors, not only on the level of antibodies. “It’s just part of the instruments we are using.”
The study was supported by the Deutsche Forschungsgemeinschaft. Dr. Simon and Dr. Tascilar declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new study in The Lancet Rheumatology examines the strength and duration of SARS-CoV-2 vaccine–induced immunoglobulin-G antibody responses over time for patients with a variety of autoimmune diseases, compared with healthy controls.
The presence of humoral antibodies to SARS-CoV-2 has been shown to correlate with protection against COVID infection. But for patients with immune-mediated inflammatory diseases (IMIDs), host response to COVID infection or to vaccination is affected by the immune dysfunction imposed by the IMID and by the use of immune-modulating drugs to treat it.
This new study finds a weaker – as shown previously – and less sustained immune response to SARS-CoV-2 vaccines in patients with a variety of IMIDs, including rheumatoid arthritis, spondyloarthritis, psoriasis, inflammatory bowel diseases, and other systemic autoimmune diseases such as lupus. It also points toward the possibility of adjusting treatment and vaccination schedules and strategies for these patients based on their antibody levels, among other factors, to preserve best protection against severe COVID.
“It is important to assess immune response in these patients to see if they still have protection against severe COVID infection,” said lead author David Simon, MD, senior clinical scientist in clinical immunology and rheumatology at University Hospital Erlangen (Germany). “We know that antibody response is an immune correlate. Therefore, it is important to see how large and durable the immune response is to the coronavirus vaccine in these IMID patients, and whether specific drugs or therapies have negative effects on their immune response.”
What was studied?
For this large prospective cohort study, researchers registered 5076 coronavirus-vaccinated individuals. They analyzed serum samples obtained between December 15, 2020, and December 1, 2021, from 2,535 patients diagnosed with IMIDs and participating in a prospective coronavirus study program at the Deutsches Zentrum Immuntherapie in Erlangen. The IMID patients had a mean age of 55.0 years, and 58.9% were women.
A healthy control group of 1,198 individuals without IMID who had a mean age of 40.7 years, including 53.8% men, was also recruited for the analysis. All approved coronavirus vaccines were included, following standard vaccination schedules. Antibody response was measured over time by an enzyme-linked immunosorbent assay from 8 weeks after first vaccination to week 40.
Among the findings, the healthy controls had higher postvaccine antibody levels than did those with IMIDs. But the majority of vaccinated patients with IMID were able to build up a humoral immune response to SARS-CoV-2. Patients who were taking B-cell inhibitors like rituximab (Rituxan, Genentech; and biosimilars) and T-cell inhibitors like abatacept (Orencia, Bristol Myers Squibb) for IMIDs had significantly poorer antibody response.
Greater age and the use of combination therapies for IMIDs, compared with monotherapy, further reduced immune response to the vaccine. In terms of vaccination modality, messenger RNA–based vaccines induced higher antibody levels than did vector-based vaccines. The researchers noted that patients with IMID who were given a third vaccine dose could actually catch up well with the antibody responses observed in healthy controls.
“We looked at whether different IMIDs had a different humoral response, and we also assessed if there are effects from different therapeutic strategies,” Dr. Simon explained. “It doesn’t matter so much what kind of IMID patients have; much more important is the specific drug treatment and its impact on their antibody response.” Some participants were advised to briefly stop taking some immunosuppressive treatments before or after vaccination.
One of Dr. Simon’s coauthors, statistician and rheumatologist Koray Tascilar, MD, added, “This research is important because we looked not only at who responded less, which has been previously established, but who are at greater risk of losing their immune response, and how quickly.”
Need to take care
“Most treatments we as rheumatologists give to our patients don’t affect their SARS-CoV-2 humoral response,” Dr. Simon said. “However, there are specific drugs that are associated with lower antibody response. With respect to those drugs, we have to be more careful.”
It is important to be able to tell patients which drugs are safe and won’t have a negative impact on their immune response to vaccinations, Dr. Tascilar said. “But it would be too strong to say we’re ready to choose therapies based on their potential impact on protection against COVID. Yes, there is a risk from catching COVID, but we need to balance that risk with the risk of not giving patients the medications that are necessary to treat their rheumatologic condition.”
These diseases are serious, sometimes life-threatening. “We might think of strategies for how to mitigate the risk of underprotection from COVID that is brought about by these treatments,” he said. For example, offering boosters sooner or more frequently, or prophylactically treating with monoclonal antibodies.
“This study, along other recent studies, has found that antibody levels in patients with immune-mediated diseases wane more rapidly than in healthy controls, and this is especially true of those on medications that interfere with the B and T cells and anticytokine therapies,” Rebecca Haberman, MD, assistant professor, division of rheumatology, New York University Langone Health, noted in an email to this news organization.
“While there is no known antibody level that specifically correlates with clinical protection, and each patient needs to be thought of individually, these findings support the use of supplemental booster dosing in patients with immune-mediated inflammatory diseases,” Dr. Haberman said, adding that her own research in this area has shown similar results.
“As a rheumatologist, I would be more likely to encourage my patients – especially those on immunomodulatory medications – to get boosted.”
Dr. Tascilar said his study does not directly answer the question of whether an earlier booster shot would be an effective strategy for patients with IMID. “In our department, we have an early boosting strategy, based on level of immune response.” But the decision of revaccination or not, and when, is based on a number of factors, not only on the level of antibodies. “It’s just part of the instruments we are using.”
The study was supported by the Deutsche Forschungsgemeinschaft. Dr. Simon and Dr. Tascilar declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE LANCET RHEUMATOLOGY
Body contouring tops list of cosmetic procedures with adverse event reports
of data from the Manufacturer and User Facility Device Experience (MAUDE).
The number of noninvasive body-contouring procedures performed in the United States increased by fivefold from 2011 to 2019, attributed in part to a combination of improved technology and new medical devices, as well as a “cosmetically savvy consumer base heavily influenced by social media,” wrote Young Lim, MD, PhD, of the department of dermatology, Massachusetts General Hospital, Boston, and coauthors.
However, premarket evaluations of many new medical devices fail to capture rare or delayed onset complications, and consumers and providers may not be fully aware of potential adverse events, they said. The MAUDE database was created by the Food and Drug Administration in 1991 to collect information on device-related deaths, serious injuries, or malfunctions based on reports from manufacturers, patients, and health care providers.
The researchers used the MAUDE database to identify and highlight adverse events associated with noninvasive body contouring technology in order to improve patient safety and satisfaction.
In their report, published in Lasers in Surgery and Medicine, they analyzed 723 medical device reports (MDRs) reported between 2015 and 2021: 660 for noninvasive body contouring, 55 for cellulite treatments, and 8 for muscle stimulation.
“Notably, of the 723 total MDRs between 2015 and 2021, 515 (71.2%) were reported in 2021, with the next highest reported being 64 in 2019 (8.8%),” the researchers wrote.
Overall, paradoxical hyperplasia (PAH) accounted for the majority of adverse reactions in the noninvasive body-contouring category (73.2%). In PAH, patients develop additional adipose tissue in areas treated with cryolipolysis. In this study, all reports of PAH as well as all 47 reported cases of abdominal hernias were attributed to the CoolSculpting device.
For cellulite treatments, the most common MDRs – 11 of 55 – were scars and keloids (20%). The Cellfina subcision technique accounted for 47% (26 of 55) of the MDRs in this category, including 9 of the scar and keloid cases.
Only eight of the MDRs analyzed were in the muscle stimulation category; of these, burns were the most common adverse event and accounted for three of the reports. The other reported AEs were two cases of pain and one report each of electrical shock, urticaria, and arrhythmia.
Patients are increasingly opting for noninvasive cosmetic procedures, but adverse events may be underreported despite the existence of databases such as MAUDE, the researchers wrote in their discussion.
“PAH, first reported in 2014 as an adverse sequelae of cryolipolysis, remains without known pathophysiology, though it proportionately affects men more than women,” they noted. The incidence of PAH varies widely, and the current treatment of choice is power-assisted liposuction, they said, although surgical abdominoplasty may be needed in severe cases.
The findings were limited by several factors including the reliance of the quality of submissions, the selection biases of the MAUDE database, and the potential for underreporting, the researchers noted.
However, “by cataloging the AEs of the growing noninvasive cosmetics market, the MAUDE can educate providers and inform patients to maximize safety and efficacy,” they said.
The size of the database and volume of reports provides a picture that likely reflects overall trends occurring in clinical practice, but in order to be effective, such databases require diligence on the part of manufacturers and clinicians to provide accurate, up-to-date information, the researchers concluded.
More procedures mean more complications
“As the market for minimally and noninvasive cosmetic procedures continues to expand, clinicians will likely encounter a greater number of patients with complications from these procedures,” said Jacqueline Watchmaker, MD, a general and cosmetic dermatologist in Scottsdale, Ariz., in an interview.
“Now more than ever, it is important for providers to understand potential side effects of procedures so that they can adequately counsel patients and optimize patient safety,” and therefore the current study is important at this time, she commented.
Dr. Watchmaker, who was not involved in the study, said that, overall, she was not surprised by the findings. “The adverse events analyzed from the Manufacturer and User Facility Device Experience parallel what is seen in clinical practice,” she said. “I did find it slightly surprising that an overwhelming majority of the medical device reports (515 of 723) were from 2021.” As the authors discuss, the reasons for this increase may include such factors as more flexible pandemic work schedules, pandemic weight gain, and the rise in MedSpas in recent years, she added.
“Some patients mistakenly think that ‘noninvasive’ or ‘minimally invasive’ procedures are risk free,” said Dr. Watchmaker. “However, as this review clearly demonstrates, complications can and do occur with these procedures. It is our job as clinicians to educate our patients on potential adverse events prior to treatment,” she emphasized. Also, she added, it is important for clinicians to report all adverse events to the MAUDE database so the true risks of noninvasive procedures can be more accurately assessed.
As for additional research, “It would be interesting to repeat the same study but to look at other minimally and noninvasive cosmetic devices such as radiofrequency and ultrasound devices,” Dr. Watchmaker noted.
The study received no outside funding. Dr. Lim and his coauthors, Adam Wulkan, MD, of the Lahey Clinic, Burlington, Mass., and Mathew Avram, MD, JD, of Massachusetts General Hospital, had no financial conflicts to disclose. Dr. Watchmaker had no financial conflicts to disclose.
Medical device–related adverse events can be reported to the FDA’s MAUDE database here .
of data from the Manufacturer and User Facility Device Experience (MAUDE).
The number of noninvasive body-contouring procedures performed in the United States increased by fivefold from 2011 to 2019, attributed in part to a combination of improved technology and new medical devices, as well as a “cosmetically savvy consumer base heavily influenced by social media,” wrote Young Lim, MD, PhD, of the department of dermatology, Massachusetts General Hospital, Boston, and coauthors.
However, premarket evaluations of many new medical devices fail to capture rare or delayed onset complications, and consumers and providers may not be fully aware of potential adverse events, they said. The MAUDE database was created by the Food and Drug Administration in 1991 to collect information on device-related deaths, serious injuries, or malfunctions based on reports from manufacturers, patients, and health care providers.
The researchers used the MAUDE database to identify and highlight adverse events associated with noninvasive body contouring technology in order to improve patient safety and satisfaction.
In their report, published in Lasers in Surgery and Medicine, they analyzed 723 medical device reports (MDRs) reported between 2015 and 2021: 660 for noninvasive body contouring, 55 for cellulite treatments, and 8 for muscle stimulation.
“Notably, of the 723 total MDRs between 2015 and 2021, 515 (71.2%) were reported in 2021, with the next highest reported being 64 in 2019 (8.8%),” the researchers wrote.
Overall, paradoxical hyperplasia (PAH) accounted for the majority of adverse reactions in the noninvasive body-contouring category (73.2%). In PAH, patients develop additional adipose tissue in areas treated with cryolipolysis. In this study, all reports of PAH as well as all 47 reported cases of abdominal hernias were attributed to the CoolSculpting device.
For cellulite treatments, the most common MDRs – 11 of 55 – were scars and keloids (20%). The Cellfina subcision technique accounted for 47% (26 of 55) of the MDRs in this category, including 9 of the scar and keloid cases.
Only eight of the MDRs analyzed were in the muscle stimulation category; of these, burns were the most common adverse event and accounted for three of the reports. The other reported AEs were two cases of pain and one report each of electrical shock, urticaria, and arrhythmia.
Patients are increasingly opting for noninvasive cosmetic procedures, but adverse events may be underreported despite the existence of databases such as MAUDE, the researchers wrote in their discussion.
“PAH, first reported in 2014 as an adverse sequelae of cryolipolysis, remains without known pathophysiology, though it proportionately affects men more than women,” they noted. The incidence of PAH varies widely, and the current treatment of choice is power-assisted liposuction, they said, although surgical abdominoplasty may be needed in severe cases.
The findings were limited by several factors including the reliance of the quality of submissions, the selection biases of the MAUDE database, and the potential for underreporting, the researchers noted.
However, “by cataloging the AEs of the growing noninvasive cosmetics market, the MAUDE can educate providers and inform patients to maximize safety and efficacy,” they said.
The size of the database and volume of reports provides a picture that likely reflects overall trends occurring in clinical practice, but in order to be effective, such databases require diligence on the part of manufacturers and clinicians to provide accurate, up-to-date information, the researchers concluded.
More procedures mean more complications
“As the market for minimally and noninvasive cosmetic procedures continues to expand, clinicians will likely encounter a greater number of patients with complications from these procedures,” said Jacqueline Watchmaker, MD, a general and cosmetic dermatologist in Scottsdale, Ariz., in an interview.
“Now more than ever, it is important for providers to understand potential side effects of procedures so that they can adequately counsel patients and optimize patient safety,” and therefore the current study is important at this time, she commented.
Dr. Watchmaker, who was not involved in the study, said that, overall, she was not surprised by the findings. “The adverse events analyzed from the Manufacturer and User Facility Device Experience parallel what is seen in clinical practice,” she said. “I did find it slightly surprising that an overwhelming majority of the medical device reports (515 of 723) were from 2021.” As the authors discuss, the reasons for this increase may include such factors as more flexible pandemic work schedules, pandemic weight gain, and the rise in MedSpas in recent years, she added.
“Some patients mistakenly think that ‘noninvasive’ or ‘minimally invasive’ procedures are risk free,” said Dr. Watchmaker. “However, as this review clearly demonstrates, complications can and do occur with these procedures. It is our job as clinicians to educate our patients on potential adverse events prior to treatment,” she emphasized. Also, she added, it is important for clinicians to report all adverse events to the MAUDE database so the true risks of noninvasive procedures can be more accurately assessed.
As for additional research, “It would be interesting to repeat the same study but to look at other minimally and noninvasive cosmetic devices such as radiofrequency and ultrasound devices,” Dr. Watchmaker noted.
The study received no outside funding. Dr. Lim and his coauthors, Adam Wulkan, MD, of the Lahey Clinic, Burlington, Mass., and Mathew Avram, MD, JD, of Massachusetts General Hospital, had no financial conflicts to disclose. Dr. Watchmaker had no financial conflicts to disclose.
Medical device–related adverse events can be reported to the FDA’s MAUDE database here .
of data from the Manufacturer and User Facility Device Experience (MAUDE).
The number of noninvasive body-contouring procedures performed in the United States increased by fivefold from 2011 to 2019, attributed in part to a combination of improved technology and new medical devices, as well as a “cosmetically savvy consumer base heavily influenced by social media,” wrote Young Lim, MD, PhD, of the department of dermatology, Massachusetts General Hospital, Boston, and coauthors.
However, premarket evaluations of many new medical devices fail to capture rare or delayed onset complications, and consumers and providers may not be fully aware of potential adverse events, they said. The MAUDE database was created by the Food and Drug Administration in 1991 to collect information on device-related deaths, serious injuries, or malfunctions based on reports from manufacturers, patients, and health care providers.
The researchers used the MAUDE database to identify and highlight adverse events associated with noninvasive body contouring technology in order to improve patient safety and satisfaction.
In their report, published in Lasers in Surgery and Medicine, they analyzed 723 medical device reports (MDRs) reported between 2015 and 2021: 660 for noninvasive body contouring, 55 for cellulite treatments, and 8 for muscle stimulation.
“Notably, of the 723 total MDRs between 2015 and 2021, 515 (71.2%) were reported in 2021, with the next highest reported being 64 in 2019 (8.8%),” the researchers wrote.
Overall, paradoxical hyperplasia (PAH) accounted for the majority of adverse reactions in the noninvasive body-contouring category (73.2%). In PAH, patients develop additional adipose tissue in areas treated with cryolipolysis. In this study, all reports of PAH as well as all 47 reported cases of abdominal hernias were attributed to the CoolSculpting device.
For cellulite treatments, the most common MDRs – 11 of 55 – were scars and keloids (20%). The Cellfina subcision technique accounted for 47% (26 of 55) of the MDRs in this category, including 9 of the scar and keloid cases.
Only eight of the MDRs analyzed were in the muscle stimulation category; of these, burns were the most common adverse event and accounted for three of the reports. The other reported AEs were two cases of pain and one report each of electrical shock, urticaria, and arrhythmia.
Patients are increasingly opting for noninvasive cosmetic procedures, but adverse events may be underreported despite the existence of databases such as MAUDE, the researchers wrote in their discussion.
“PAH, first reported in 2014 as an adverse sequelae of cryolipolysis, remains without known pathophysiology, though it proportionately affects men more than women,” they noted. The incidence of PAH varies widely, and the current treatment of choice is power-assisted liposuction, they said, although surgical abdominoplasty may be needed in severe cases.
The findings were limited by several factors including the reliance of the quality of submissions, the selection biases of the MAUDE database, and the potential for underreporting, the researchers noted.
However, “by cataloging the AEs of the growing noninvasive cosmetics market, the MAUDE can educate providers and inform patients to maximize safety and efficacy,” they said.
The size of the database and volume of reports provides a picture that likely reflects overall trends occurring in clinical practice, but in order to be effective, such databases require diligence on the part of manufacturers and clinicians to provide accurate, up-to-date information, the researchers concluded.
More procedures mean more complications
“As the market for minimally and noninvasive cosmetic procedures continues to expand, clinicians will likely encounter a greater number of patients with complications from these procedures,” said Jacqueline Watchmaker, MD, a general and cosmetic dermatologist in Scottsdale, Ariz., in an interview.
“Now more than ever, it is important for providers to understand potential side effects of procedures so that they can adequately counsel patients and optimize patient safety,” and therefore the current study is important at this time, she commented.
Dr. Watchmaker, who was not involved in the study, said that, overall, she was not surprised by the findings. “The adverse events analyzed from the Manufacturer and User Facility Device Experience parallel what is seen in clinical practice,” she said. “I did find it slightly surprising that an overwhelming majority of the medical device reports (515 of 723) were from 2021.” As the authors discuss, the reasons for this increase may include such factors as more flexible pandemic work schedules, pandemic weight gain, and the rise in MedSpas in recent years, she added.
“Some patients mistakenly think that ‘noninvasive’ or ‘minimally invasive’ procedures are risk free,” said Dr. Watchmaker. “However, as this review clearly demonstrates, complications can and do occur with these procedures. It is our job as clinicians to educate our patients on potential adverse events prior to treatment,” she emphasized. Also, she added, it is important for clinicians to report all adverse events to the MAUDE database so the true risks of noninvasive procedures can be more accurately assessed.
As for additional research, “It would be interesting to repeat the same study but to look at other minimally and noninvasive cosmetic devices such as radiofrequency and ultrasound devices,” Dr. Watchmaker noted.
The study received no outside funding. Dr. Lim and his coauthors, Adam Wulkan, MD, of the Lahey Clinic, Burlington, Mass., and Mathew Avram, MD, JD, of Massachusetts General Hospital, had no financial conflicts to disclose. Dr. Watchmaker had no financial conflicts to disclose.
Medical device–related adverse events can be reported to the FDA’s MAUDE database here .
FROM LASERS IN SURGERY AND MEDICINE
Asian patients with psoriasis have shortest visits, study shows
(NAMCS) from 2010 to 2016.
Yet the reasons for the difference are unclear and in need of further research, said the investigators and dermatologists who were asked to comment on the research.
The study covered over 4 million visits for psoriasis and found that the mean duration of visits for Asian patients was 9.2 minutes, compared with 15.7 minutes for Hispanic or Latino patients, 20.7 minutes for non-Hispanic Black patients, and 15.4 minutes for non-Hispanic White patients.
The mean duration of visits with Asian patients was 39.9% shorter, compared with visits with White patients (beta coefficient, –5,747; 95% confidence interval, –11.026 to –0.469; P = .03), and 40.6% shorter, compared with visits with non-Asian patients combined (beta coefficient, –5.908; 95% CI, –11.147 to –0.669, P = .03), April W. Armstrong, MD, MPH, professor of dermatology and director of the psoriasis program at the University of Southern California, Los Angeles, and Kevin K. Wu, MD, a dermatology resident at USC, said in a research letter published in JAMA Dermatology.
“The etiology of these differences is unclear,” they wrote. “It is possible that factors such as unconscious bias, cultural differences in communication, or residual confounding may be responsible for the observed findings.”
Their findings came from multivariable linear regression analyses that adjusted for age, sex, type of visit (new or follow-up), visit complexity based on the number of reasons for the visit, insurance status (such as private insurance or Medicaid), psoriasis severity on the basis of systemic psoriasis treatment or phototherapy, and complex topical regimens (three or more topical agents).
Commenting on the results, Deborah A. Scott, MD, codirector of the skin of color dermatology program at Brigham and Women’s Hospital and assistant professor at Harvard Medical School, both in Boston, said in an interview that visit length “is a reasonable parameter to look at among many others” when investigating potential disparities in care.
“They’re equating [shorter visit times] with lack of time spent counseling patients,” said Dr. Scott, who was not involved in the research. But there are “many variables” that can affect visit time, such as language differences, time spent with interpreters, and differences in patient educational levels.
Clarissa Yang, MD, dermatologist-in-chief at Tufts Medical Center, Boston, agreed. “We’re worried about there being a quality of care issue. However, there could also be differences culturally in how [the patients] interact with their physicians – their styles and the questions they ask,” she said in an interview. “The study is a good first step to noting that there may be a disparity,” and there is a need to break down the differences “into more granularity.”
Previous research, the authors wrote, has found that Asian patients were less likely to receive counseling from physicians, compared with White patients. And “paradoxically,” they noted, Asian individuals tend to present with more severe psoriasis than patients of other races and ethnicities.
Dr. Scott said the tendency to present with more severe psoriasis has been documented in patients with skin of color broadly – likely because of delays in recognition and treatment.
Race and ethnicity in the study were self-reported by patients, and missing data were imputed by NAMCS researchers using a sequential regression method. Patients who did not report race and ethnicity may have different characteristics affecting visit duration than those who did report the information, Dr. Armstrong and Dr. Wu said in describing their study’s limitations.
Other differences found
In addition to visit length, they found significant differences in mean age and in the use of complex topical regimens. The mean ages of Asian, Hispanic or Latino, and non-Hispanic Black patients were 37.2, 44.7, and 33.3 years, respectively. Complex topical regimens were prescribed to 11.8% of Asian patients, compared with 1.5% of Black and 1.1% of White patients.
For practicing dermatologists, knowing for now that Asian patients have shorter visits “may bring to light some consciousness to how we practice,” Dr. Yang noted. “We may counsel differently, we may spend differing amounts of time – for reasons still unknown. But being generally aware can help us to shift any unconscious bias that may be there.”
Dermatologists, Dr. Armstrong and Dr. Wu wrote, “need to allow sufficient time to develop strong physician-patient communication regardless of patient background.”
The NAMCS – administered by the Center for Disease Control and Prevention’s National Center for Health Statistics – collects data on a sample of visits provided by non–federally employed office-based physicians.
Dr. Armstrong disclosed receiving personal fees from AbbVie and Regeneron for research funding and serving as a scientific adviser and speaker for additional pharmaceutical and therapeutic companies. Dr. Wu, Dr. Scott, and Dr. Yang did not report any disclosures.
(NAMCS) from 2010 to 2016.
Yet the reasons for the difference are unclear and in need of further research, said the investigators and dermatologists who were asked to comment on the research.
The study covered over 4 million visits for psoriasis and found that the mean duration of visits for Asian patients was 9.2 minutes, compared with 15.7 minutes for Hispanic or Latino patients, 20.7 minutes for non-Hispanic Black patients, and 15.4 minutes for non-Hispanic White patients.
The mean duration of visits with Asian patients was 39.9% shorter, compared with visits with White patients (beta coefficient, –5,747; 95% confidence interval, –11.026 to –0.469; P = .03), and 40.6% shorter, compared with visits with non-Asian patients combined (beta coefficient, –5.908; 95% CI, –11.147 to –0.669, P = .03), April W. Armstrong, MD, MPH, professor of dermatology and director of the psoriasis program at the University of Southern California, Los Angeles, and Kevin K. Wu, MD, a dermatology resident at USC, said in a research letter published in JAMA Dermatology.
“The etiology of these differences is unclear,” they wrote. “It is possible that factors such as unconscious bias, cultural differences in communication, or residual confounding may be responsible for the observed findings.”
Their findings came from multivariable linear regression analyses that adjusted for age, sex, type of visit (new or follow-up), visit complexity based on the number of reasons for the visit, insurance status (such as private insurance or Medicaid), psoriasis severity on the basis of systemic psoriasis treatment or phototherapy, and complex topical regimens (three or more topical agents).
Commenting on the results, Deborah A. Scott, MD, codirector of the skin of color dermatology program at Brigham and Women’s Hospital and assistant professor at Harvard Medical School, both in Boston, said in an interview that visit length “is a reasonable parameter to look at among many others” when investigating potential disparities in care.
“They’re equating [shorter visit times] with lack of time spent counseling patients,” said Dr. Scott, who was not involved in the research. But there are “many variables” that can affect visit time, such as language differences, time spent with interpreters, and differences in patient educational levels.
Clarissa Yang, MD, dermatologist-in-chief at Tufts Medical Center, Boston, agreed. “We’re worried about there being a quality of care issue. However, there could also be differences culturally in how [the patients] interact with their physicians – their styles and the questions they ask,” she said in an interview. “The study is a good first step to noting that there may be a disparity,” and there is a need to break down the differences “into more granularity.”
Previous research, the authors wrote, has found that Asian patients were less likely to receive counseling from physicians, compared with White patients. And “paradoxically,” they noted, Asian individuals tend to present with more severe psoriasis than patients of other races and ethnicities.
Dr. Scott said the tendency to present with more severe psoriasis has been documented in patients with skin of color broadly – likely because of delays in recognition and treatment.
Race and ethnicity in the study were self-reported by patients, and missing data were imputed by NAMCS researchers using a sequential regression method. Patients who did not report race and ethnicity may have different characteristics affecting visit duration than those who did report the information, Dr. Armstrong and Dr. Wu said in describing their study’s limitations.
Other differences found
In addition to visit length, they found significant differences in mean age and in the use of complex topical regimens. The mean ages of Asian, Hispanic or Latino, and non-Hispanic Black patients were 37.2, 44.7, and 33.3 years, respectively. Complex topical regimens were prescribed to 11.8% of Asian patients, compared with 1.5% of Black and 1.1% of White patients.
For practicing dermatologists, knowing for now that Asian patients have shorter visits “may bring to light some consciousness to how we practice,” Dr. Yang noted. “We may counsel differently, we may spend differing amounts of time – for reasons still unknown. But being generally aware can help us to shift any unconscious bias that may be there.”
Dermatologists, Dr. Armstrong and Dr. Wu wrote, “need to allow sufficient time to develop strong physician-patient communication regardless of patient background.”
The NAMCS – administered by the Center for Disease Control and Prevention’s National Center for Health Statistics – collects data on a sample of visits provided by non–federally employed office-based physicians.
Dr. Armstrong disclosed receiving personal fees from AbbVie and Regeneron for research funding and serving as a scientific adviser and speaker for additional pharmaceutical and therapeutic companies. Dr. Wu, Dr. Scott, and Dr. Yang did not report any disclosures.
(NAMCS) from 2010 to 2016.
Yet the reasons for the difference are unclear and in need of further research, said the investigators and dermatologists who were asked to comment on the research.
The study covered over 4 million visits for psoriasis and found that the mean duration of visits for Asian patients was 9.2 minutes, compared with 15.7 minutes for Hispanic or Latino patients, 20.7 minutes for non-Hispanic Black patients, and 15.4 minutes for non-Hispanic White patients.
The mean duration of visits with Asian patients was 39.9% shorter, compared with visits with White patients (beta coefficient, –5,747; 95% confidence interval, –11.026 to –0.469; P = .03), and 40.6% shorter, compared with visits with non-Asian patients combined (beta coefficient, –5.908; 95% CI, –11.147 to –0.669, P = .03), April W. Armstrong, MD, MPH, professor of dermatology and director of the psoriasis program at the University of Southern California, Los Angeles, and Kevin K. Wu, MD, a dermatology resident at USC, said in a research letter published in JAMA Dermatology.
“The etiology of these differences is unclear,” they wrote. “It is possible that factors such as unconscious bias, cultural differences in communication, or residual confounding may be responsible for the observed findings.”
Their findings came from multivariable linear regression analyses that adjusted for age, sex, type of visit (new or follow-up), visit complexity based on the number of reasons for the visit, insurance status (such as private insurance or Medicaid), psoriasis severity on the basis of systemic psoriasis treatment or phototherapy, and complex topical regimens (three or more topical agents).
Commenting on the results, Deborah A. Scott, MD, codirector of the skin of color dermatology program at Brigham and Women’s Hospital and assistant professor at Harvard Medical School, both in Boston, said in an interview that visit length “is a reasonable parameter to look at among many others” when investigating potential disparities in care.
“They’re equating [shorter visit times] with lack of time spent counseling patients,” said Dr. Scott, who was not involved in the research. But there are “many variables” that can affect visit time, such as language differences, time spent with interpreters, and differences in patient educational levels.
Clarissa Yang, MD, dermatologist-in-chief at Tufts Medical Center, Boston, agreed. “We’re worried about there being a quality of care issue. However, there could also be differences culturally in how [the patients] interact with their physicians – their styles and the questions they ask,” she said in an interview. “The study is a good first step to noting that there may be a disparity,” and there is a need to break down the differences “into more granularity.”
Previous research, the authors wrote, has found that Asian patients were less likely to receive counseling from physicians, compared with White patients. And “paradoxically,” they noted, Asian individuals tend to present with more severe psoriasis than patients of other races and ethnicities.
Dr. Scott said the tendency to present with more severe psoriasis has been documented in patients with skin of color broadly – likely because of delays in recognition and treatment.
Race and ethnicity in the study were self-reported by patients, and missing data were imputed by NAMCS researchers using a sequential regression method. Patients who did not report race and ethnicity may have different characteristics affecting visit duration than those who did report the information, Dr. Armstrong and Dr. Wu said in describing their study’s limitations.
Other differences found
In addition to visit length, they found significant differences in mean age and in the use of complex topical regimens. The mean ages of Asian, Hispanic or Latino, and non-Hispanic Black patients were 37.2, 44.7, and 33.3 years, respectively. Complex topical regimens were prescribed to 11.8% of Asian patients, compared with 1.5% of Black and 1.1% of White patients.
For practicing dermatologists, knowing for now that Asian patients have shorter visits “may bring to light some consciousness to how we practice,” Dr. Yang noted. “We may counsel differently, we may spend differing amounts of time – for reasons still unknown. But being generally aware can help us to shift any unconscious bias that may be there.”
Dermatologists, Dr. Armstrong and Dr. Wu wrote, “need to allow sufficient time to develop strong physician-patient communication regardless of patient background.”
The NAMCS – administered by the Center for Disease Control and Prevention’s National Center for Health Statistics – collects data on a sample of visits provided by non–federally employed office-based physicians.
Dr. Armstrong disclosed receiving personal fees from AbbVie and Regeneron for research funding and serving as a scientific adviser and speaker for additional pharmaceutical and therapeutic companies. Dr. Wu, Dr. Scott, and Dr. Yang did not report any disclosures.
FROM JAMA DERMATOLOGY
Second opinions on melanocytic lesions swayed when first opinion is known
, diminishing the value and accuracy of an independent analysis.
In a novel effort to determine whether previous interpretations sway second opinions, 149 dermatopathologists were asked to read melanocytic skin biopsy specimens without access to the initial pathology report. A year or more later they read them again but now with access to the initial reading.
The study showed that the participants, independent of many variables, such as years of experience or frequency with which they offered second options, were more likely to upgrade or downgrade the severity of the specimens in accordance with the initial report even if their original reading was correct.
If the goal of a second dermatopathologist opinion is to obtain an independent diagnostic opinion, the message from this study is that they “should be blinded to first opinions,” according to the authors of this study, led by Joann G. Elmore, MD, professor of medicine, University of California, Los Angeles. The study was published online in JAMA Dermatology.
Two-phase study has 1-year washout
The study was conducted in two phases. In phase 1, a nationally representative sample of volunteer dermatopathologists performed 878 interpretations. In phase 2, conducted after a washout period of 12 months or more, the dermatopathologists read a random subset of the same cases evaluated in phase 1, but this time, unlike the first, they were first exposed to prior pathology reports.
Ultimately, “the dermatologists provided more than 5,000 interpretations of study cases, which was a big contribution of time,” Dr. Elmore said in an interview. Grateful for their critical contribution, she speculated that they were driven by the importance of the question being asked.
When categorized by the Melanocytic Pathology Assessment Tool (MPAT), which rates specimens from benign (class 1) to pT1b invasive melanoma (class 4), the influence of the prior report went in both directions, so that the likelihood of upgrading or downgrading went in accordance with the grading in the original dermatopathology report.
As a result, the risk of a less severe interpretation on the second relative to the first reading was 38% greater if the initial dermatopathology report had a lower grade (relative risk, 1.38; 95% confidence interval [CI], 1.19-1.59). The risk of upgrading the second report if the initial pathology report had a higher grade was increased by more than 50% (RR, 1.52; 95% CI, 1.34-1.73).
The greater likelihood of upgrading than downgrading is “understandable,” Dr. Elmore said. “I think this is consistent with the concern about missing something,” she explained.
According to Dr. Elmore, one of the greatest concerns regarding the bias imposed by the original pathology report is that the switch of opinions often went from one that was accurate to one that was inaccurate.
If the phase 1 diagnosis was accurate but upgraded in the phase 2 diagnosis, the risk of inaccuracy was almost doubled (RR, 1.96; 95% CI, 1.31-2.93). If the phase 1 report was inaccurate, the relative risk of changing the phase 2 diagnosis was still high but lower than if it was accurate (RR, 1.46; 95% CI, 1.27-1.68).
“That is, even when the phase 1 diagnoses agreed with the consensus reference diagnosis, they were swayed away from the correct diagnosis in phase 2 [when the initial pathology report characterized the specimen as higher grade],” Dr. Elmore reported.
Conversely, the risk of downgrading was about the same whether the phase 1 evaluation was accurate (RR, 1.37; 95% CI, 1.14-1.64) or inaccurate (RR 1.32; 95% CI, 1.07-1.64).
Downward and upward shifts in severity from an accurate diagnosis are concerning because of the likelihood they will lead to overtreatment or undertreatment. The problem, according to data from this study, is that dermatologists making a second opinion cannot judge their own susceptibility to being swayed by the original report.
Pathologists might be unaware of bias
At baseline, the participants were asked whether they thought they were influenced by the first interpretation when providing a second opinion. Although 69% acknowledged that they might be “somewhat influenced,” 31% maintained that they do not take initial reports into consideration. When the two groups were compared, the risk of downgrading was nearly identical. The risk of upgrading was lower in those claiming to disregard initial reports (RR, 1.29) relative to those who said they were “somewhat influenced” by a previous diagnosis (RR, 1.64), but the difference was not significant.
The actual risk of bias incurred by prior pathology reports might be greater than that captured in this study for several reasons, according to the investigators. They pointed out that all participants were experienced and board-certified and might therefore be expected to be more confident in their interpretations than an unselected group of dermatopathologists. In addition, participants might have been more careful in their interpretations knowing they were participating in a study.
“There are a lot of data to support the value of second opinions [in dermatopathology and other areas], but we need to consider the process of how they are being obtained,” Dr. Elmore said. “There needs to be a greater emphasis on providing an independent analysis.”
More than 60% of the dermatologists participating in this study reported that they agreed or strongly agreed with the premise that they prefer to have the original dermatopathology report when they offer a second opinion. Dr. Elmore said that the desire of those offering a second opinion to have as much information in front of them as possible is understandable, but the bias imposed by the original report weakens the value of the second opinion.
Blind reading of pathology reports needed
“These data suggest that seeing the original report sways opinions and that includes swaying opinions away from an accurate reading,” Dr. Elmore said. She thinks that for dermatopathologists to render a valuable and independent second opinion, the specimens should be examined “at least initially” without access to the first report.
The results of this study were not surprising to Vishal Anil Patel, MD, director of the Cutaneous Oncology Program, George Washington University Cancer Center, Washington. He made the point that physicians “are human first and foremost and not perfect machines.” As a result, he suggested bias and error are inevitable.
Although strategies to avoid bias are likely to offer some protection against inaccuracy, he said that diagnostic support tools such as artificial intelligence might be the right direction for improving inter- and intra-rater reliability.
Ruifeng Guo, MD, PhD, a consultant in the division of anatomic pathology at the Mayo Clinic, Rochester, Minn., agreed with the basic premise of the study, but he cautioned that restricting access to the initial pathology report might not always be the right approach.
It is true that “dermatopathologists providing a second opinion in diagnosing cutaneous melanoma are mostly unaware of the risk of bias if they read the initial pathology report,” said Dr. Guo, but restricting access comes with risks.
“There are also times critical information may be contained in the initial pathology report that needs to be considered when providing a second opinion consultation,” he noted. Ultimately, the decision to read or not read the initial report should be decided “on an individual basis.”
The study was funded by grants from the National Cancer Institute. Dr. Elmore, Dr. Patel, and Dr. Guo reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, diminishing the value and accuracy of an independent analysis.
In a novel effort to determine whether previous interpretations sway second opinions, 149 dermatopathologists were asked to read melanocytic skin biopsy specimens without access to the initial pathology report. A year or more later they read them again but now with access to the initial reading.
The study showed that the participants, independent of many variables, such as years of experience or frequency with which they offered second options, were more likely to upgrade or downgrade the severity of the specimens in accordance with the initial report even if their original reading was correct.
If the goal of a second dermatopathologist opinion is to obtain an independent diagnostic opinion, the message from this study is that they “should be blinded to first opinions,” according to the authors of this study, led by Joann G. Elmore, MD, professor of medicine, University of California, Los Angeles. The study was published online in JAMA Dermatology.
Two-phase study has 1-year washout
The study was conducted in two phases. In phase 1, a nationally representative sample of volunteer dermatopathologists performed 878 interpretations. In phase 2, conducted after a washout period of 12 months or more, the dermatopathologists read a random subset of the same cases evaluated in phase 1, but this time, unlike the first, they were first exposed to prior pathology reports.
Ultimately, “the dermatologists provided more than 5,000 interpretations of study cases, which was a big contribution of time,” Dr. Elmore said in an interview. Grateful for their critical contribution, she speculated that they were driven by the importance of the question being asked.
When categorized by the Melanocytic Pathology Assessment Tool (MPAT), which rates specimens from benign (class 1) to pT1b invasive melanoma (class 4), the influence of the prior report went in both directions, so that the likelihood of upgrading or downgrading went in accordance with the grading in the original dermatopathology report.
As a result, the risk of a less severe interpretation on the second relative to the first reading was 38% greater if the initial dermatopathology report had a lower grade (relative risk, 1.38; 95% confidence interval [CI], 1.19-1.59). The risk of upgrading the second report if the initial pathology report had a higher grade was increased by more than 50% (RR, 1.52; 95% CI, 1.34-1.73).
The greater likelihood of upgrading than downgrading is “understandable,” Dr. Elmore said. “I think this is consistent with the concern about missing something,” she explained.
According to Dr. Elmore, one of the greatest concerns regarding the bias imposed by the original pathology report is that the switch of opinions often went from one that was accurate to one that was inaccurate.
If the phase 1 diagnosis was accurate but upgraded in the phase 2 diagnosis, the risk of inaccuracy was almost doubled (RR, 1.96; 95% CI, 1.31-2.93). If the phase 1 report was inaccurate, the relative risk of changing the phase 2 diagnosis was still high but lower than if it was accurate (RR, 1.46; 95% CI, 1.27-1.68).
“That is, even when the phase 1 diagnoses agreed with the consensus reference diagnosis, they were swayed away from the correct diagnosis in phase 2 [when the initial pathology report characterized the specimen as higher grade],” Dr. Elmore reported.
Conversely, the risk of downgrading was about the same whether the phase 1 evaluation was accurate (RR, 1.37; 95% CI, 1.14-1.64) or inaccurate (RR 1.32; 95% CI, 1.07-1.64).
Downward and upward shifts in severity from an accurate diagnosis are concerning because of the likelihood they will lead to overtreatment or undertreatment. The problem, according to data from this study, is that dermatologists making a second opinion cannot judge their own susceptibility to being swayed by the original report.
Pathologists might be unaware of bias
At baseline, the participants were asked whether they thought they were influenced by the first interpretation when providing a second opinion. Although 69% acknowledged that they might be “somewhat influenced,” 31% maintained that they do not take initial reports into consideration. When the two groups were compared, the risk of downgrading was nearly identical. The risk of upgrading was lower in those claiming to disregard initial reports (RR, 1.29) relative to those who said they were “somewhat influenced” by a previous diagnosis (RR, 1.64), but the difference was not significant.
The actual risk of bias incurred by prior pathology reports might be greater than that captured in this study for several reasons, according to the investigators. They pointed out that all participants were experienced and board-certified and might therefore be expected to be more confident in their interpretations than an unselected group of dermatopathologists. In addition, participants might have been more careful in their interpretations knowing they were participating in a study.
“There are a lot of data to support the value of second opinions [in dermatopathology and other areas], but we need to consider the process of how they are being obtained,” Dr. Elmore said. “There needs to be a greater emphasis on providing an independent analysis.”
More than 60% of the dermatologists participating in this study reported that they agreed or strongly agreed with the premise that they prefer to have the original dermatopathology report when they offer a second opinion. Dr. Elmore said that the desire of those offering a second opinion to have as much information in front of them as possible is understandable, but the bias imposed by the original report weakens the value of the second opinion.
Blind reading of pathology reports needed
“These data suggest that seeing the original report sways opinions and that includes swaying opinions away from an accurate reading,” Dr. Elmore said. She thinks that for dermatopathologists to render a valuable and independent second opinion, the specimens should be examined “at least initially” without access to the first report.
The results of this study were not surprising to Vishal Anil Patel, MD, director of the Cutaneous Oncology Program, George Washington University Cancer Center, Washington. He made the point that physicians “are human first and foremost and not perfect machines.” As a result, he suggested bias and error are inevitable.
Although strategies to avoid bias are likely to offer some protection against inaccuracy, he said that diagnostic support tools such as artificial intelligence might be the right direction for improving inter- and intra-rater reliability.
Ruifeng Guo, MD, PhD, a consultant in the division of anatomic pathology at the Mayo Clinic, Rochester, Minn., agreed with the basic premise of the study, but he cautioned that restricting access to the initial pathology report might not always be the right approach.
It is true that “dermatopathologists providing a second opinion in diagnosing cutaneous melanoma are mostly unaware of the risk of bias if they read the initial pathology report,” said Dr. Guo, but restricting access comes with risks.
“There are also times critical information may be contained in the initial pathology report that needs to be considered when providing a second opinion consultation,” he noted. Ultimately, the decision to read or not read the initial report should be decided “on an individual basis.”
The study was funded by grants from the National Cancer Institute. Dr. Elmore, Dr. Patel, and Dr. Guo reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, diminishing the value and accuracy of an independent analysis.
In a novel effort to determine whether previous interpretations sway second opinions, 149 dermatopathologists were asked to read melanocytic skin biopsy specimens without access to the initial pathology report. A year or more later they read them again but now with access to the initial reading.
The study showed that the participants, independent of many variables, such as years of experience or frequency with which they offered second options, were more likely to upgrade or downgrade the severity of the specimens in accordance with the initial report even if their original reading was correct.
If the goal of a second dermatopathologist opinion is to obtain an independent diagnostic opinion, the message from this study is that they “should be blinded to first opinions,” according to the authors of this study, led by Joann G. Elmore, MD, professor of medicine, University of California, Los Angeles. The study was published online in JAMA Dermatology.
Two-phase study has 1-year washout
The study was conducted in two phases. In phase 1, a nationally representative sample of volunteer dermatopathologists performed 878 interpretations. In phase 2, conducted after a washout period of 12 months or more, the dermatopathologists read a random subset of the same cases evaluated in phase 1, but this time, unlike the first, they were first exposed to prior pathology reports.
Ultimately, “the dermatologists provided more than 5,000 interpretations of study cases, which was a big contribution of time,” Dr. Elmore said in an interview. Grateful for their critical contribution, she speculated that they were driven by the importance of the question being asked.
When categorized by the Melanocytic Pathology Assessment Tool (MPAT), which rates specimens from benign (class 1) to pT1b invasive melanoma (class 4), the influence of the prior report went in both directions, so that the likelihood of upgrading or downgrading went in accordance with the grading in the original dermatopathology report.
As a result, the risk of a less severe interpretation on the second relative to the first reading was 38% greater if the initial dermatopathology report had a lower grade (relative risk, 1.38; 95% confidence interval [CI], 1.19-1.59). The risk of upgrading the second report if the initial pathology report had a higher grade was increased by more than 50% (RR, 1.52; 95% CI, 1.34-1.73).
The greater likelihood of upgrading than downgrading is “understandable,” Dr. Elmore said. “I think this is consistent with the concern about missing something,” she explained.
According to Dr. Elmore, one of the greatest concerns regarding the bias imposed by the original pathology report is that the switch of opinions often went from one that was accurate to one that was inaccurate.
If the phase 1 diagnosis was accurate but upgraded in the phase 2 diagnosis, the risk of inaccuracy was almost doubled (RR, 1.96; 95% CI, 1.31-2.93). If the phase 1 report was inaccurate, the relative risk of changing the phase 2 diagnosis was still high but lower than if it was accurate (RR, 1.46; 95% CI, 1.27-1.68).
“That is, even when the phase 1 diagnoses agreed with the consensus reference diagnosis, they were swayed away from the correct diagnosis in phase 2 [when the initial pathology report characterized the specimen as higher grade],” Dr. Elmore reported.
Conversely, the risk of downgrading was about the same whether the phase 1 evaluation was accurate (RR, 1.37; 95% CI, 1.14-1.64) or inaccurate (RR 1.32; 95% CI, 1.07-1.64).
Downward and upward shifts in severity from an accurate diagnosis are concerning because of the likelihood they will lead to overtreatment or undertreatment. The problem, according to data from this study, is that dermatologists making a second opinion cannot judge their own susceptibility to being swayed by the original report.
Pathologists might be unaware of bias
At baseline, the participants were asked whether they thought they were influenced by the first interpretation when providing a second opinion. Although 69% acknowledged that they might be “somewhat influenced,” 31% maintained that they do not take initial reports into consideration. When the two groups were compared, the risk of downgrading was nearly identical. The risk of upgrading was lower in those claiming to disregard initial reports (RR, 1.29) relative to those who said they were “somewhat influenced” by a previous diagnosis (RR, 1.64), but the difference was not significant.
The actual risk of bias incurred by prior pathology reports might be greater than that captured in this study for several reasons, according to the investigators. They pointed out that all participants were experienced and board-certified and might therefore be expected to be more confident in their interpretations than an unselected group of dermatopathologists. In addition, participants might have been more careful in their interpretations knowing they were participating in a study.
“There are a lot of data to support the value of second opinions [in dermatopathology and other areas], but we need to consider the process of how they are being obtained,” Dr. Elmore said. “There needs to be a greater emphasis on providing an independent analysis.”
More than 60% of the dermatologists participating in this study reported that they agreed or strongly agreed with the premise that they prefer to have the original dermatopathology report when they offer a second opinion. Dr. Elmore said that the desire of those offering a second opinion to have as much information in front of them as possible is understandable, but the bias imposed by the original report weakens the value of the second opinion.
Blind reading of pathology reports needed
“These data suggest that seeing the original report sways opinions and that includes swaying opinions away from an accurate reading,” Dr. Elmore said. She thinks that for dermatopathologists to render a valuable and independent second opinion, the specimens should be examined “at least initially” without access to the first report.
The results of this study were not surprising to Vishal Anil Patel, MD, director of the Cutaneous Oncology Program, George Washington University Cancer Center, Washington. He made the point that physicians “are human first and foremost and not perfect machines.” As a result, he suggested bias and error are inevitable.
Although strategies to avoid bias are likely to offer some protection against inaccuracy, he said that diagnostic support tools such as artificial intelligence might be the right direction for improving inter- and intra-rater reliability.
Ruifeng Guo, MD, PhD, a consultant in the division of anatomic pathology at the Mayo Clinic, Rochester, Minn., agreed with the basic premise of the study, but he cautioned that restricting access to the initial pathology report might not always be the right approach.
It is true that “dermatopathologists providing a second opinion in diagnosing cutaneous melanoma are mostly unaware of the risk of bias if they read the initial pathology report,” said Dr. Guo, but restricting access comes with risks.
“There are also times critical information may be contained in the initial pathology report that needs to be considered when providing a second opinion consultation,” he noted. Ultimately, the decision to read or not read the initial report should be decided “on an individual basis.”
The study was funded by grants from the National Cancer Institute. Dr. Elmore, Dr. Patel, and Dr. Guo reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Abrocitinib evaluated in patients with and without prior dupilumab treatment
an industry-sponsored study reports.
“In this post hoc analysis, both the efficacy and the safety profiles of abrocitinib were consistent in patients with moderate-to-severe atopic dermatitis, regardless of prior biologic therapy use,” lead author Melinda Gooderham, MD, medical director of the SKiN Centre for Dermatology, Peterborough, Ont., said during an oral presentation at the Society for Investigative Dermatology (SID) 2022 Annual Meeting.
“These results ... support the use of abrocitinib in patients who might have received biologic therapy prior,” she added.
“Prior biologic use did not reveal any new safety signals ... keeping in mind the key limitation of this analysis is that it was done post hoc,” she noted.
Guidelines for moderate-to-severe atopic dermatitis refractory to topical or systemic therapy include systemic immunosuppressants and dupilumab, a monoclonal antibody that inhibits interleukin-4 and interleukin-13 cytokine-induced responses, Dr. Gooderham said.
The Food and Drug Administration recently approved abrocitinib, an oral once-a-day Janus kinase 1 (JAK1) inhibitor, to treat the disease. The approval came with a boxed warning about increased risk for serious infections, mortality, malignancy, and lymphoproliferative disorders, major adverse cardiovascular events, thrombosis, and laboratory abnormalities.
Comparing the bio-experienced with the bio-naive
Dr. Gooderham and colleagues investigated whether patients who’d been treated with a biologic would respond to abrocitinib differently than patients who had not received prior biologic treatment.
Researchers pooled data from two phase 3 placebo-controlled trials of abrocitinib that led to approval and an earlier phase 2b study. They identified 67 patients previously treated with dupilumab and 867 patients who were bio-naive. They repeated their analysis using data from another phase 3 study of abrocitinib on 86 patients previously treated with dupilumab and 1,147 who were bio-naive. On average, the bio-experienced patients were in their mid-30s to early 40s, and the bio-naive group was several years younger.
In the pooled phase 2b and phase 3 JADE MONO-1 and JADE MONO-2 monotherapy trials, patients received once-daily abrocitinib 100 or 200 mg or placebo for 12 weeks. In the phase 3 JADE REGIMEN, which they analyzed separately, eligible patients were enrolled in a 12-week open-label run-in period during which they received an induction treatment of abrocitinib 200 mg once a day.
Researchers compared results of two assessments: the IGA (Investigator Global Assessment) and EASI-75 (Eczema Area and Severity Index, 75% or greater improvement from baseline).
- At week 12, IGA 0/1 dose-dependent response rates were similar in the pooled groups, regardless of whether they had received prior biologic therapy. With abrocitinib 200 mg, 43.5% of those with prior dupilumab therapy responded versus 41.4% of bio-naive patients; with abrocitinib 100 mg, 24.1% versus 26.7% responded. In JADE REGIMEN, corresponding response rates with abrocitinib 200 mg were 53.5% versus 66.9%, respectively.
- At week 12, EASI-75 responses were also comparable. In the pooled groups by dose, with abrocitinib 200 mg, EASI-75 response rates were 65.2% in patients with prior dupilumab therapy versus 62.4% in those without; at abrocitinib 100 mg, 34.5% versus 42.7% responded. Corresponding rates in JADE REGIMEN were 64.0% versus 76.4%, respectively.
- Treatment-emergent adverse event rates among patients with versus without prior biologic therapy were, respectively, 71.7% versus 69.9% (abrocitinib 200 mg + 100 mg groups) in the pooled population. Rates in JADE REGIMEN with abrocitinib 200 mg were, respectively, 66.3% versus 66.5%.
- Abrocitinib efficacy and safety were consistent in patients with moderate-to-severe atopic dermatitis, regardless of prior biologic therapy. Adverse events in the pooled monotherapy trials and in JADE REGIMEN included acne, atopic dermatitis, diarrhea, headache, nasopharyngitis, nausea, upper abdominal pain, and upper respiratory tract infection.
The authors acknowledge that the post hoc study design is a limitation and recommend confirming these findings in a large, long-term prospective study.
JAK inhibitors expand treatment options
The results will help doctors treat their patients, Jami L. Miller, MD, associate professor of dermatology and dermatology clinic medical director at Vanderbilt University Medical Center, Nashville, Tenn., told this news organization.
“Because JAK inhibitors have potentially more side effects than inhibitors of interleukin-4 and interleukin-13, in clinical practice most dermatologists are more likely to treat patients first with dupilumab or similar meds and step up to a JAK inhibitor if they do not respond,” she added in an email.
“With more meds coming out to meet the needs of this population, this is an exciting time for patients with moderate-to-severe atopic dermatitis,” she commented.
Lindsay C. Strowd, MD, associate professor and vice chair of the department of dermatology at Wake Forest University, Winston-Salem, N.C., said JAK inhibitors are increasingly being studied and approved for use in various dermatologic diseases.
An oral JAK inhibitor (upadacitinib) is currently FDA approved for moderate-to-severe atopic dermatitis, and a topical JAK inhibitor (ruxolitinib) is also approved for use in atopic dermatitis, Dr. Strowd noted.
“The study results give providers important practical information,” added Dr. Strowd, who also was not involved with the study. “Those of us who care for patients with severe atopic dermatitis need to know how patients with prior biologic exposure will respond as newer agents come to market and the options for biologic use in atopic dermatitis continue to grow.”
The study was sponsored by Pfizer. All study authors have reported relevant financial relationships with, and several authors are employees of, Pfizer, the developer of abrocitinib. Dr. Strowd and Dr. Miller have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
an industry-sponsored study reports.
“In this post hoc analysis, both the efficacy and the safety profiles of abrocitinib were consistent in patients with moderate-to-severe atopic dermatitis, regardless of prior biologic therapy use,” lead author Melinda Gooderham, MD, medical director of the SKiN Centre for Dermatology, Peterborough, Ont., said during an oral presentation at the Society for Investigative Dermatology (SID) 2022 Annual Meeting.
“These results ... support the use of abrocitinib in patients who might have received biologic therapy prior,” she added.
“Prior biologic use did not reveal any new safety signals ... keeping in mind the key limitation of this analysis is that it was done post hoc,” she noted.
Guidelines for moderate-to-severe atopic dermatitis refractory to topical or systemic therapy include systemic immunosuppressants and dupilumab, a monoclonal antibody that inhibits interleukin-4 and interleukin-13 cytokine-induced responses, Dr. Gooderham said.
The Food and Drug Administration recently approved abrocitinib, an oral once-a-day Janus kinase 1 (JAK1) inhibitor, to treat the disease. The approval came with a boxed warning about increased risk for serious infections, mortality, malignancy, and lymphoproliferative disorders, major adverse cardiovascular events, thrombosis, and laboratory abnormalities.
Comparing the bio-experienced with the bio-naive
Dr. Gooderham and colleagues investigated whether patients who’d been treated with a biologic would respond to abrocitinib differently than patients who had not received prior biologic treatment.
Researchers pooled data from two phase 3 placebo-controlled trials of abrocitinib that led to approval and an earlier phase 2b study. They identified 67 patients previously treated with dupilumab and 867 patients who were bio-naive. They repeated their analysis using data from another phase 3 study of abrocitinib on 86 patients previously treated with dupilumab and 1,147 who were bio-naive. On average, the bio-experienced patients were in their mid-30s to early 40s, and the bio-naive group was several years younger.
In the pooled phase 2b and phase 3 JADE MONO-1 and JADE MONO-2 monotherapy trials, patients received once-daily abrocitinib 100 or 200 mg or placebo for 12 weeks. In the phase 3 JADE REGIMEN, which they analyzed separately, eligible patients were enrolled in a 12-week open-label run-in period during which they received an induction treatment of abrocitinib 200 mg once a day.
Researchers compared results of two assessments: the IGA (Investigator Global Assessment) and EASI-75 (Eczema Area and Severity Index, 75% or greater improvement from baseline).
- At week 12, IGA 0/1 dose-dependent response rates were similar in the pooled groups, regardless of whether they had received prior biologic therapy. With abrocitinib 200 mg, 43.5% of those with prior dupilumab therapy responded versus 41.4% of bio-naive patients; with abrocitinib 100 mg, 24.1% versus 26.7% responded. In JADE REGIMEN, corresponding response rates with abrocitinib 200 mg were 53.5% versus 66.9%, respectively.
- At week 12, EASI-75 responses were also comparable. In the pooled groups by dose, with abrocitinib 200 mg, EASI-75 response rates were 65.2% in patients with prior dupilumab therapy versus 62.4% in those without; at abrocitinib 100 mg, 34.5% versus 42.7% responded. Corresponding rates in JADE REGIMEN were 64.0% versus 76.4%, respectively.
- Treatment-emergent adverse event rates among patients with versus without prior biologic therapy were, respectively, 71.7% versus 69.9% (abrocitinib 200 mg + 100 mg groups) in the pooled population. Rates in JADE REGIMEN with abrocitinib 200 mg were, respectively, 66.3% versus 66.5%.
- Abrocitinib efficacy and safety were consistent in patients with moderate-to-severe atopic dermatitis, regardless of prior biologic therapy. Adverse events in the pooled monotherapy trials and in JADE REGIMEN included acne, atopic dermatitis, diarrhea, headache, nasopharyngitis, nausea, upper abdominal pain, and upper respiratory tract infection.
The authors acknowledge that the post hoc study design is a limitation and recommend confirming these findings in a large, long-term prospective study.
JAK inhibitors expand treatment options
The results will help doctors treat their patients, Jami L. Miller, MD, associate professor of dermatology and dermatology clinic medical director at Vanderbilt University Medical Center, Nashville, Tenn., told this news organization.
“Because JAK inhibitors have potentially more side effects than inhibitors of interleukin-4 and interleukin-13, in clinical practice most dermatologists are more likely to treat patients first with dupilumab or similar meds and step up to a JAK inhibitor if they do not respond,” she added in an email.
“With more meds coming out to meet the needs of this population, this is an exciting time for patients with moderate-to-severe atopic dermatitis,” she commented.
Lindsay C. Strowd, MD, associate professor and vice chair of the department of dermatology at Wake Forest University, Winston-Salem, N.C., said JAK inhibitors are increasingly being studied and approved for use in various dermatologic diseases.
An oral JAK inhibitor (upadacitinib) is currently FDA approved for moderate-to-severe atopic dermatitis, and a topical JAK inhibitor (ruxolitinib) is also approved for use in atopic dermatitis, Dr. Strowd noted.
“The study results give providers important practical information,” added Dr. Strowd, who also was not involved with the study. “Those of us who care for patients with severe atopic dermatitis need to know how patients with prior biologic exposure will respond as newer agents come to market and the options for biologic use in atopic dermatitis continue to grow.”
The study was sponsored by Pfizer. All study authors have reported relevant financial relationships with, and several authors are employees of, Pfizer, the developer of abrocitinib. Dr. Strowd and Dr. Miller have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
an industry-sponsored study reports.
“In this post hoc analysis, both the efficacy and the safety profiles of abrocitinib were consistent in patients with moderate-to-severe atopic dermatitis, regardless of prior biologic therapy use,” lead author Melinda Gooderham, MD, medical director of the SKiN Centre for Dermatology, Peterborough, Ont., said during an oral presentation at the Society for Investigative Dermatology (SID) 2022 Annual Meeting.
“These results ... support the use of abrocitinib in patients who might have received biologic therapy prior,” she added.
“Prior biologic use did not reveal any new safety signals ... keeping in mind the key limitation of this analysis is that it was done post hoc,” she noted.
Guidelines for moderate-to-severe atopic dermatitis refractory to topical or systemic therapy include systemic immunosuppressants and dupilumab, a monoclonal antibody that inhibits interleukin-4 and interleukin-13 cytokine-induced responses, Dr. Gooderham said.
The Food and Drug Administration recently approved abrocitinib, an oral once-a-day Janus kinase 1 (JAK1) inhibitor, to treat the disease. The approval came with a boxed warning about increased risk for serious infections, mortality, malignancy, and lymphoproliferative disorders, major adverse cardiovascular events, thrombosis, and laboratory abnormalities.
Comparing the bio-experienced with the bio-naive
Dr. Gooderham and colleagues investigated whether patients who’d been treated with a biologic would respond to abrocitinib differently than patients who had not received prior biologic treatment.
Researchers pooled data from two phase 3 placebo-controlled trials of abrocitinib that led to approval and an earlier phase 2b study. They identified 67 patients previously treated with dupilumab and 867 patients who were bio-naive. They repeated their analysis using data from another phase 3 study of abrocitinib on 86 patients previously treated with dupilumab and 1,147 who were bio-naive. On average, the bio-experienced patients were in their mid-30s to early 40s, and the bio-naive group was several years younger.
In the pooled phase 2b and phase 3 JADE MONO-1 and JADE MONO-2 monotherapy trials, patients received once-daily abrocitinib 100 or 200 mg or placebo for 12 weeks. In the phase 3 JADE REGIMEN, which they analyzed separately, eligible patients were enrolled in a 12-week open-label run-in period during which they received an induction treatment of abrocitinib 200 mg once a day.
Researchers compared results of two assessments: the IGA (Investigator Global Assessment) and EASI-75 (Eczema Area and Severity Index, 75% or greater improvement from baseline).
- At week 12, IGA 0/1 dose-dependent response rates were similar in the pooled groups, regardless of whether they had received prior biologic therapy. With abrocitinib 200 mg, 43.5% of those with prior dupilumab therapy responded versus 41.4% of bio-naive patients; with abrocitinib 100 mg, 24.1% versus 26.7% responded. In JADE REGIMEN, corresponding response rates with abrocitinib 200 mg were 53.5% versus 66.9%, respectively.
- At week 12, EASI-75 responses were also comparable. In the pooled groups by dose, with abrocitinib 200 mg, EASI-75 response rates were 65.2% in patients with prior dupilumab therapy versus 62.4% in those without; at abrocitinib 100 mg, 34.5% versus 42.7% responded. Corresponding rates in JADE REGIMEN were 64.0% versus 76.4%, respectively.
- Treatment-emergent adverse event rates among patients with versus without prior biologic therapy were, respectively, 71.7% versus 69.9% (abrocitinib 200 mg + 100 mg groups) in the pooled population. Rates in JADE REGIMEN with abrocitinib 200 mg were, respectively, 66.3% versus 66.5%.
- Abrocitinib efficacy and safety were consistent in patients with moderate-to-severe atopic dermatitis, regardless of prior biologic therapy. Adverse events in the pooled monotherapy trials and in JADE REGIMEN included acne, atopic dermatitis, diarrhea, headache, nasopharyngitis, nausea, upper abdominal pain, and upper respiratory tract infection.
The authors acknowledge that the post hoc study design is a limitation and recommend confirming these findings in a large, long-term prospective study.
JAK inhibitors expand treatment options
The results will help doctors treat their patients, Jami L. Miller, MD, associate professor of dermatology and dermatology clinic medical director at Vanderbilt University Medical Center, Nashville, Tenn., told this news organization.
“Because JAK inhibitors have potentially more side effects than inhibitors of interleukin-4 and interleukin-13, in clinical practice most dermatologists are more likely to treat patients first with dupilumab or similar meds and step up to a JAK inhibitor if they do not respond,” she added in an email.
“With more meds coming out to meet the needs of this population, this is an exciting time for patients with moderate-to-severe atopic dermatitis,” she commented.
Lindsay C. Strowd, MD, associate professor and vice chair of the department of dermatology at Wake Forest University, Winston-Salem, N.C., said JAK inhibitors are increasingly being studied and approved for use in various dermatologic diseases.
An oral JAK inhibitor (upadacitinib) is currently FDA approved for moderate-to-severe atopic dermatitis, and a topical JAK inhibitor (ruxolitinib) is also approved for use in atopic dermatitis, Dr. Strowd noted.
“The study results give providers important practical information,” added Dr. Strowd, who also was not involved with the study. “Those of us who care for patients with severe atopic dermatitis need to know how patients with prior biologic exposure will respond as newer agents come to market and the options for biologic use in atopic dermatitis continue to grow.”
The study was sponsored by Pfizer. All study authors have reported relevant financial relationships with, and several authors are employees of, Pfizer, the developer of abrocitinib. Dr. Strowd and Dr. Miller have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.