User login
Finerenone benefits T2D across spectrum of renal function
Treatment with finerenone produced roughly similar reductions in heart failure–related outcomes in people with type 2 diabetes and chronic kidney disease (CKD) across the spectrum of kidney function, compared with placebo, including those who had albuminuria but a preserved estimated glomerular filtration rate (eGFR), in a post hoc analysis of pooled data from more than 13,000 people.
The findings, from the two pivotal trials for the agent, “reinforce the importance of routine eGFR and UACR [urinary albumin-to-creatinine ratio] screening” in people with type 2 diabetes to identify new candidates for treatment with finerenone (Kerendia), Gerasimos Filippatos, MD, and coauthors said in a report published online in JACC: Heart Failure.
Among the 13,026 patients in the two combined trials, 40% had a preserved eGFR of greater than 60 mL/min per 1.73 m2 despite also having albuminuria with a UACR of at least 30 mg/g, showing how often this combination occurs. But many clinicians “do not follow the guidelines” and fail to measure the UACR in these patients in routine practice, noted Dr. Filippatos at the annual congress of the European Society of Cardiology in August.
“We now have something to do for these patients,” treat them with finerenone, said Dr. Filippatos, professor and director of heart failure at the Attikon University Hospital, Athens.
The availability of finerenone following its U.S. approval in 2021 means clinicians “must get used to measuring UACR” in people with type 2 diabetes even when their eGFR is normal, especially people with type 2 diabetes plus high cardiovascular disease risk, he said.
The Food and Drug Administration approved finerenone, a nonsteroidal mineralocorticoid receptor antagonist, for treating people with type 2 diabetes and CKD in July 2021, but its uptake has been slow, experts say. In a talk in September 2022 during the annual meeting of the European Association for the Study of Diabetes, Jennifer B. Green, MD, estimated that U.S. uptake of finerenone for appropriate people with type 2 diabetes had not advanced beyond 10%.
A recent review also noted that uptake of screening for elevated UACR in U.S. patients with type 2 diabetes was in the range of 10%-40% during 2017-2019, a “shockingly low rate,” said Dr. Green, a professor and diabetes specialist at Duke University, Durham, N.C.
A new reason to screen for albuminuria
“It’s an extremely important message,” Johann Bauersachs, MD, commented in an interview. Results from “many studies have shown that albuminuria is an excellent additional marker for cardiovascular disease risk. But measurement of albuminuria is not widely done, despite guidelines that recommend annual albuminuria testing in people with type 2 diabetes,” said Dr. Bauersachs, professor and head of the department of cardiology at Hannover (Germany) Medical School.
“Even before there was finerenone, there were reasons to measure UACR, but I hope adding finerenone will help, and more clinicians will incorporate UACR into their routine practice,” said Dr. Bauersachs, who was not involved with the finerenone studies.
The analyses reported by Dr. Filippatos and coauthors used data from two related trials of finerenone, FIDELIO-DKD and FIGARO-DKD, combined by prespecified design into a single dataset, FIDELITY, with a total of 13,026 participants eligible for analysis and followed for a median of 3 years. All had type 2 diabetes and CKD based on having a UACR of at least 30 mg/g. Their eGFR levels could run as high as 74 mL/min per 1.73 m2 in FIDELIO-DKD, and as high as 90 mL/min/1.73m2 in FIGARO-DKD. The two trials excluded people with heart failure with reduced ejection fraction, and those with a serum potassium greater than 4.8 mmol/L.
In the FIDELITY dataset treatment with finerenone led to a significant 17% reduction in the combined incidence of cardiovascular death or first hospitalization for heart failure relative to those who received placebo. This relative risk reduction was not affected by either eGFR or UACR values at baseline, the new analysis showed.
The analysis also demonstrated a nonsignificant trend toward greater reductions in heart failure–related outcomes among study participants who began with an eGFR in the normal range of at least 60 mL/min per 1.73 m2. The researchers also found a nonsignificant trend to a greater reduction in heart failure–related events among those with a UACR of less than 300 mg/g.
Finerenone favors patients with less advanced CKD
In short “the magnitude of the treatment benefit tended to favor patients with less advanced CKD,” concluded the researchers, suggesting that “earlier intervention [with finerenone] in the CKD course is likely to provide the greatest long-term benefit on heart failure–related outcomes.” This led them to further infer “the importance of not only routine assessing eGFR, but also perhaps more importantly, routinely screening for UACR to facilitate early diagnosis and early intervention in patients with type 2 diabetes.”
Findings from FIDELIO-DKD and FIGARO-DKD led to recent guideline additions for finerenone by several medical groups. In August 2022, the American Association of Clinical Endocrinologists released an update to its guideline for managing people with diabetes that recommended treating people with type 2 diabetes with finerenone when they have a UACR of at least 30 mg/g if they are already treated with a maximum-tolerated dose of a renin-angiotensin system inhibitor, have a normal serum potassium level, and have an eGFR of at least 25 mL/min per 1.73 m2. The identical recommendation also appeared in a Consensus Report from the American Diabetes Association and KDIGO, an international organization promoting evidence-based management of patients with CKD.
“Finerenone provides a very important contribution because it improves prognosis even in very well managed patients” with type 2 diabetes, commented Lars Rydén, MD, professor of cardiology at the Karolinska Institute in Stockholm, as designated discussant for the report by Dr. Filippatos at the ESC congress.
The findings from the FIDELITY analysis are “trustworthy, and clinically important,” Dr. Rydén said. When left untreated, diabetic kidney disease “reduces life expectancy by an average of 16 years.”
The finerenone trials were sponsored by Bayer, which markets finerenone (Kerendia). Dr. Filippatos has received lecture fees from Bayer as well as from Amgen, Medtronic, Novartis, Servier, and Vifor. Dr. Green has financial ties to Bayer as well as to Anji, AstraZeneca, Boehringer Ingelheim/Lilly, Hawthorne Effect/Omada, Merck, Novo Nordisk, Pfizer, Roche, Sanofi/Lexicon, and Valo. Dr. Bauersachs has been a consultant to Bayer as well as to Amgen, AstraZeneca, Boehringer Ingelheim, Cardior, Cervia, CVRx, Novartis, Pfizer, and Vifor, and he has received research funding from Abiomed. Dr. Rydén has financial ties to Bayer, Boehringer Ingelheim, Eli Lilly, and Novo Nordisk.
Treatment with finerenone produced roughly similar reductions in heart failure–related outcomes in people with type 2 diabetes and chronic kidney disease (CKD) across the spectrum of kidney function, compared with placebo, including those who had albuminuria but a preserved estimated glomerular filtration rate (eGFR), in a post hoc analysis of pooled data from more than 13,000 people.
The findings, from the two pivotal trials for the agent, “reinforce the importance of routine eGFR and UACR [urinary albumin-to-creatinine ratio] screening” in people with type 2 diabetes to identify new candidates for treatment with finerenone (Kerendia), Gerasimos Filippatos, MD, and coauthors said in a report published online in JACC: Heart Failure.
Among the 13,026 patients in the two combined trials, 40% had a preserved eGFR of greater than 60 mL/min per 1.73 m2 despite also having albuminuria with a UACR of at least 30 mg/g, showing how often this combination occurs. But many clinicians “do not follow the guidelines” and fail to measure the UACR in these patients in routine practice, noted Dr. Filippatos at the annual congress of the European Society of Cardiology in August.
“We now have something to do for these patients,” treat them with finerenone, said Dr. Filippatos, professor and director of heart failure at the Attikon University Hospital, Athens.
The availability of finerenone following its U.S. approval in 2021 means clinicians “must get used to measuring UACR” in people with type 2 diabetes even when their eGFR is normal, especially people with type 2 diabetes plus high cardiovascular disease risk, he said.
The Food and Drug Administration approved finerenone, a nonsteroidal mineralocorticoid receptor antagonist, for treating people with type 2 diabetes and CKD in July 2021, but its uptake has been slow, experts say. In a talk in September 2022 during the annual meeting of the European Association for the Study of Diabetes, Jennifer B. Green, MD, estimated that U.S. uptake of finerenone for appropriate people with type 2 diabetes had not advanced beyond 10%.
A recent review also noted that uptake of screening for elevated UACR in U.S. patients with type 2 diabetes was in the range of 10%-40% during 2017-2019, a “shockingly low rate,” said Dr. Green, a professor and diabetes specialist at Duke University, Durham, N.C.
A new reason to screen for albuminuria
“It’s an extremely important message,” Johann Bauersachs, MD, commented in an interview. Results from “many studies have shown that albuminuria is an excellent additional marker for cardiovascular disease risk. But measurement of albuminuria is not widely done, despite guidelines that recommend annual albuminuria testing in people with type 2 diabetes,” said Dr. Bauersachs, professor and head of the department of cardiology at Hannover (Germany) Medical School.
“Even before there was finerenone, there were reasons to measure UACR, but I hope adding finerenone will help, and more clinicians will incorporate UACR into their routine practice,” said Dr. Bauersachs, who was not involved with the finerenone studies.
The analyses reported by Dr. Filippatos and coauthors used data from two related trials of finerenone, FIDELIO-DKD and FIGARO-DKD, combined by prespecified design into a single dataset, FIDELITY, with a total of 13,026 participants eligible for analysis and followed for a median of 3 years. All had type 2 diabetes and CKD based on having a UACR of at least 30 mg/g. Their eGFR levels could run as high as 74 mL/min per 1.73 m2 in FIDELIO-DKD, and as high as 90 mL/min/1.73m2 in FIGARO-DKD. The two trials excluded people with heart failure with reduced ejection fraction, and those with a serum potassium greater than 4.8 mmol/L.
In the FIDELITY dataset treatment with finerenone led to a significant 17% reduction in the combined incidence of cardiovascular death or first hospitalization for heart failure relative to those who received placebo. This relative risk reduction was not affected by either eGFR or UACR values at baseline, the new analysis showed.
The analysis also demonstrated a nonsignificant trend toward greater reductions in heart failure–related outcomes among study participants who began with an eGFR in the normal range of at least 60 mL/min per 1.73 m2. The researchers also found a nonsignificant trend to a greater reduction in heart failure–related events among those with a UACR of less than 300 mg/g.
Finerenone favors patients with less advanced CKD
In short “the magnitude of the treatment benefit tended to favor patients with less advanced CKD,” concluded the researchers, suggesting that “earlier intervention [with finerenone] in the CKD course is likely to provide the greatest long-term benefit on heart failure–related outcomes.” This led them to further infer “the importance of not only routine assessing eGFR, but also perhaps more importantly, routinely screening for UACR to facilitate early diagnosis and early intervention in patients with type 2 diabetes.”
Findings from FIDELIO-DKD and FIGARO-DKD led to recent guideline additions for finerenone by several medical groups. In August 2022, the American Association of Clinical Endocrinologists released an update to its guideline for managing people with diabetes that recommended treating people with type 2 diabetes with finerenone when they have a UACR of at least 30 mg/g if they are already treated with a maximum-tolerated dose of a renin-angiotensin system inhibitor, have a normal serum potassium level, and have an eGFR of at least 25 mL/min per 1.73 m2. The identical recommendation also appeared in a Consensus Report from the American Diabetes Association and KDIGO, an international organization promoting evidence-based management of patients with CKD.
“Finerenone provides a very important contribution because it improves prognosis even in very well managed patients” with type 2 diabetes, commented Lars Rydén, MD, professor of cardiology at the Karolinska Institute in Stockholm, as designated discussant for the report by Dr. Filippatos at the ESC congress.
The findings from the FIDELITY analysis are “trustworthy, and clinically important,” Dr. Rydén said. When left untreated, diabetic kidney disease “reduces life expectancy by an average of 16 years.”
The finerenone trials were sponsored by Bayer, which markets finerenone (Kerendia). Dr. Filippatos has received lecture fees from Bayer as well as from Amgen, Medtronic, Novartis, Servier, and Vifor. Dr. Green has financial ties to Bayer as well as to Anji, AstraZeneca, Boehringer Ingelheim/Lilly, Hawthorne Effect/Omada, Merck, Novo Nordisk, Pfizer, Roche, Sanofi/Lexicon, and Valo. Dr. Bauersachs has been a consultant to Bayer as well as to Amgen, AstraZeneca, Boehringer Ingelheim, Cardior, Cervia, CVRx, Novartis, Pfizer, and Vifor, and he has received research funding from Abiomed. Dr. Rydén has financial ties to Bayer, Boehringer Ingelheim, Eli Lilly, and Novo Nordisk.
Treatment with finerenone produced roughly similar reductions in heart failure–related outcomes in people with type 2 diabetes and chronic kidney disease (CKD) across the spectrum of kidney function, compared with placebo, including those who had albuminuria but a preserved estimated glomerular filtration rate (eGFR), in a post hoc analysis of pooled data from more than 13,000 people.
The findings, from the two pivotal trials for the agent, “reinforce the importance of routine eGFR and UACR [urinary albumin-to-creatinine ratio] screening” in people with type 2 diabetes to identify new candidates for treatment with finerenone (Kerendia), Gerasimos Filippatos, MD, and coauthors said in a report published online in JACC: Heart Failure.
Among the 13,026 patients in the two combined trials, 40% had a preserved eGFR of greater than 60 mL/min per 1.73 m2 despite also having albuminuria with a UACR of at least 30 mg/g, showing how often this combination occurs. But many clinicians “do not follow the guidelines” and fail to measure the UACR in these patients in routine practice, noted Dr. Filippatos at the annual congress of the European Society of Cardiology in August.
“We now have something to do for these patients,” treat them with finerenone, said Dr. Filippatos, professor and director of heart failure at the Attikon University Hospital, Athens.
The availability of finerenone following its U.S. approval in 2021 means clinicians “must get used to measuring UACR” in people with type 2 diabetes even when their eGFR is normal, especially people with type 2 diabetes plus high cardiovascular disease risk, he said.
The Food and Drug Administration approved finerenone, a nonsteroidal mineralocorticoid receptor antagonist, for treating people with type 2 diabetes and CKD in July 2021, but its uptake has been slow, experts say. In a talk in September 2022 during the annual meeting of the European Association for the Study of Diabetes, Jennifer B. Green, MD, estimated that U.S. uptake of finerenone for appropriate people with type 2 diabetes had not advanced beyond 10%.
A recent review also noted that uptake of screening for elevated UACR in U.S. patients with type 2 diabetes was in the range of 10%-40% during 2017-2019, a “shockingly low rate,” said Dr. Green, a professor and diabetes specialist at Duke University, Durham, N.C.
A new reason to screen for albuminuria
“It’s an extremely important message,” Johann Bauersachs, MD, commented in an interview. Results from “many studies have shown that albuminuria is an excellent additional marker for cardiovascular disease risk. But measurement of albuminuria is not widely done, despite guidelines that recommend annual albuminuria testing in people with type 2 diabetes,” said Dr. Bauersachs, professor and head of the department of cardiology at Hannover (Germany) Medical School.
“Even before there was finerenone, there were reasons to measure UACR, but I hope adding finerenone will help, and more clinicians will incorporate UACR into their routine practice,” said Dr. Bauersachs, who was not involved with the finerenone studies.
The analyses reported by Dr. Filippatos and coauthors used data from two related trials of finerenone, FIDELIO-DKD and FIGARO-DKD, combined by prespecified design into a single dataset, FIDELITY, with a total of 13,026 participants eligible for analysis and followed for a median of 3 years. All had type 2 diabetes and CKD based on having a UACR of at least 30 mg/g. Their eGFR levels could run as high as 74 mL/min per 1.73 m2 in FIDELIO-DKD, and as high as 90 mL/min/1.73m2 in FIGARO-DKD. The two trials excluded people with heart failure with reduced ejection fraction, and those with a serum potassium greater than 4.8 mmol/L.
In the FIDELITY dataset treatment with finerenone led to a significant 17% reduction in the combined incidence of cardiovascular death or first hospitalization for heart failure relative to those who received placebo. This relative risk reduction was not affected by either eGFR or UACR values at baseline, the new analysis showed.
The analysis also demonstrated a nonsignificant trend toward greater reductions in heart failure–related outcomes among study participants who began with an eGFR in the normal range of at least 60 mL/min per 1.73 m2. The researchers also found a nonsignificant trend to a greater reduction in heart failure–related events among those with a UACR of less than 300 mg/g.
Finerenone favors patients with less advanced CKD
In short “the magnitude of the treatment benefit tended to favor patients with less advanced CKD,” concluded the researchers, suggesting that “earlier intervention [with finerenone] in the CKD course is likely to provide the greatest long-term benefit on heart failure–related outcomes.” This led them to further infer “the importance of not only routine assessing eGFR, but also perhaps more importantly, routinely screening for UACR to facilitate early diagnosis and early intervention in patients with type 2 diabetes.”
Findings from FIDELIO-DKD and FIGARO-DKD led to recent guideline additions for finerenone by several medical groups. In August 2022, the American Association of Clinical Endocrinologists released an update to its guideline for managing people with diabetes that recommended treating people with type 2 diabetes with finerenone when they have a UACR of at least 30 mg/g if they are already treated with a maximum-tolerated dose of a renin-angiotensin system inhibitor, have a normal serum potassium level, and have an eGFR of at least 25 mL/min per 1.73 m2. The identical recommendation also appeared in a Consensus Report from the American Diabetes Association and KDIGO, an international organization promoting evidence-based management of patients with CKD.
“Finerenone provides a very important contribution because it improves prognosis even in very well managed patients” with type 2 diabetes, commented Lars Rydén, MD, professor of cardiology at the Karolinska Institute in Stockholm, as designated discussant for the report by Dr. Filippatos at the ESC congress.
The findings from the FIDELITY analysis are “trustworthy, and clinically important,” Dr. Rydén said. When left untreated, diabetic kidney disease “reduces life expectancy by an average of 16 years.”
The finerenone trials were sponsored by Bayer, which markets finerenone (Kerendia). Dr. Filippatos has received lecture fees from Bayer as well as from Amgen, Medtronic, Novartis, Servier, and Vifor. Dr. Green has financial ties to Bayer as well as to Anji, AstraZeneca, Boehringer Ingelheim/Lilly, Hawthorne Effect/Omada, Merck, Novo Nordisk, Pfizer, Roche, Sanofi/Lexicon, and Valo. Dr. Bauersachs has been a consultant to Bayer as well as to Amgen, AstraZeneca, Boehringer Ingelheim, Cardior, Cervia, CVRx, Novartis, Pfizer, and Vifor, and he has received research funding from Abiomed. Dr. Rydén has financial ties to Bayer, Boehringer Ingelheim, Eli Lilly, and Novo Nordisk.
FROM JACC: HEART FAILURE
Chronic Kidney Disease in People with Type 2 Diabetes
We know from the literature and in practice that type 2 diabetes (T2D) is one of the most common risk factors for developing chronic kidney disease (CKD). How prevalent is this overlap, and are certain patients more at risk than others?
Dr. McGill: That’s correct, in fact, 20% to 40% of people with T2D have identifiable CKD, and the rest are at risk for developing CKD in the future. All patients with T2D should recognize that risk and undergo annual screening for CKD. If an individual has prediabetes, then step up the screening to perhaps twice a year to see if the person has progressed. At that point, we can think about intervening with a medication to prevent the onset of diabetes, particularly if the patient is unable to adopt significant lifestyle changes.
In your day-to-day practice, what therapeutic approach do you take in managing patients with T2D and CKD?
Dr. McGill: The earliest and arguably the most important treatment for the care of CKD in T2D is glucose control. Establishing and maintaining blood glucose levels near the normal range is our strongest weapon for preventing CKD. Another treatment avenue is controlling blood pressure. The American Diabetes Association and other groups recommend that blood pressure be less than 130/80 mm Hg. It is critical that we treat hypertension effectively to achieve those numbers.
We also have therapies, such as the SGLT2 inhibitors, that offer protection from progression of CKD and from hospitalization for heart failure. Deployment of these newer agents is important for people who have more advanced diabetes or other serious health conditions.
What is the rate of disease progression, related complications, or even mortality for these patients?
Dr. McGill: People with CKD and T2D are at risk for many things. One risk is progression of kidney disease all the way to end-stage kidney disease, which requires dialysis or transplantation. Another huge risk is cardiovascular events such as myocardial infarction (MI) and stroke.
Persons with kidney disease, for reasons we don't understand, are at higher risk of MI and stroke than people who do not have kidney disease. Therefore, the risks of early mortality and events that adversely affect quality of life are greatly increased.
Can you please discuss the economic burdens associated with T2D and CKD, and whether any interventions are in place to help offset those costs?
Dr. McGill: Diabetes itself is wickedly expensive. We have excellent treatments for diabetes today, but they are very costly. The best approach for the prevention of diabetes is to be screened. When a patient presents with prediabetes, it’s important that they take important measures, such as weight loss, exercising 150 minutes per week, or reducing 500 calories a day from their diet, all of which have been shown to forestall the onset of diabetes.
Once diabetes develops, achieving near-normal glucose control can either be very inexpensive with one or more generic drugs, or it can be terribly expensive with the newer branded drugs. Both options can help with the achievement of near-normal glucose levels, but the newer drugs are better for weight loss and provide protection from heart and kidney disease.
It is important to consider where the patient is along the disease spectrum, and to educate them on the benefits of taking a proactive approach to their health. Don’t wait for diabetes to develop before doing something about it. We have to take action earlier, and more definitively.
We do everything we can to help patients with the high cost of diabetes medications. Pharma companies offer various coupons and patient assistance programs, but it's really important that we get people on the right therapy. In order for that to happen, they have to come to office visits and get lab tests done.
Is there anything else you would like to share on this topic?
Dr. McGill: Once a person has been diagnosed with diabetes, then excellent glucose control from the onset has been shown to prevent later complications, and early treatment is inexpensive. As people progress through their journey with diabetes and blood sugars go up, we have excellent therapies that help manage high glucose and help with weight loss.
We have to be realistic and rethink our approach in some ways, but as long as people develop good health care habits and visit the doctor once or twice a year specifically to address diabetes and blood pressure, we might be able to avoid long-term complications.
We know from the literature and in practice that type 2 diabetes (T2D) is one of the most common risk factors for developing chronic kidney disease (CKD). How prevalent is this overlap, and are certain patients more at risk than others?
Dr. McGill: That’s correct, in fact, 20% to 40% of people with T2D have identifiable CKD, and the rest are at risk for developing CKD in the future. All patients with T2D should recognize that risk and undergo annual screening for CKD. If an individual has prediabetes, then step up the screening to perhaps twice a year to see if the person has progressed. At that point, we can think about intervening with a medication to prevent the onset of diabetes, particularly if the patient is unable to adopt significant lifestyle changes.
In your day-to-day practice, what therapeutic approach do you take in managing patients with T2D and CKD?
Dr. McGill: The earliest and arguably the most important treatment for the care of CKD in T2D is glucose control. Establishing and maintaining blood glucose levels near the normal range is our strongest weapon for preventing CKD. Another treatment avenue is controlling blood pressure. The American Diabetes Association and other groups recommend that blood pressure be less than 130/80 mm Hg. It is critical that we treat hypertension effectively to achieve those numbers.
We also have therapies, such as the SGLT2 inhibitors, that offer protection from progression of CKD and from hospitalization for heart failure. Deployment of these newer agents is important for people who have more advanced diabetes or other serious health conditions.
What is the rate of disease progression, related complications, or even mortality for these patients?
Dr. McGill: People with CKD and T2D are at risk for many things. One risk is progression of kidney disease all the way to end-stage kidney disease, which requires dialysis or transplantation. Another huge risk is cardiovascular events such as myocardial infarction (MI) and stroke.
Persons with kidney disease, for reasons we don't understand, are at higher risk of MI and stroke than people who do not have kidney disease. Therefore, the risks of early mortality and events that adversely affect quality of life are greatly increased.
Can you please discuss the economic burdens associated with T2D and CKD, and whether any interventions are in place to help offset those costs?
Dr. McGill: Diabetes itself is wickedly expensive. We have excellent treatments for diabetes today, but they are very costly. The best approach for the prevention of diabetes is to be screened. When a patient presents with prediabetes, it’s important that they take important measures, such as weight loss, exercising 150 minutes per week, or reducing 500 calories a day from their diet, all of which have been shown to forestall the onset of diabetes.
Once diabetes develops, achieving near-normal glucose control can either be very inexpensive with one or more generic drugs, or it can be terribly expensive with the newer branded drugs. Both options can help with the achievement of near-normal glucose levels, but the newer drugs are better for weight loss and provide protection from heart and kidney disease.
It is important to consider where the patient is along the disease spectrum, and to educate them on the benefits of taking a proactive approach to their health. Don’t wait for diabetes to develop before doing something about it. We have to take action earlier, and more definitively.
We do everything we can to help patients with the high cost of diabetes medications. Pharma companies offer various coupons and patient assistance programs, but it's really important that we get people on the right therapy. In order for that to happen, they have to come to office visits and get lab tests done.
Is there anything else you would like to share on this topic?
Dr. McGill: Once a person has been diagnosed with diabetes, then excellent glucose control from the onset has been shown to prevent later complications, and early treatment is inexpensive. As people progress through their journey with diabetes and blood sugars go up, we have excellent therapies that help manage high glucose and help with weight loss.
We have to be realistic and rethink our approach in some ways, but as long as people develop good health care habits and visit the doctor once or twice a year specifically to address diabetes and blood pressure, we might be able to avoid long-term complications.
We know from the literature and in practice that type 2 diabetes (T2D) is one of the most common risk factors for developing chronic kidney disease (CKD). How prevalent is this overlap, and are certain patients more at risk than others?
Dr. McGill: That’s correct, in fact, 20% to 40% of people with T2D have identifiable CKD, and the rest are at risk for developing CKD in the future. All patients with T2D should recognize that risk and undergo annual screening for CKD. If an individual has prediabetes, then step up the screening to perhaps twice a year to see if the person has progressed. At that point, we can think about intervening with a medication to prevent the onset of diabetes, particularly if the patient is unable to adopt significant lifestyle changes.
In your day-to-day practice, what therapeutic approach do you take in managing patients with T2D and CKD?
Dr. McGill: The earliest and arguably the most important treatment for the care of CKD in T2D is glucose control. Establishing and maintaining blood glucose levels near the normal range is our strongest weapon for preventing CKD. Another treatment avenue is controlling blood pressure. The American Diabetes Association and other groups recommend that blood pressure be less than 130/80 mm Hg. It is critical that we treat hypertension effectively to achieve those numbers.
We also have therapies, such as the SGLT2 inhibitors, that offer protection from progression of CKD and from hospitalization for heart failure. Deployment of these newer agents is important for people who have more advanced diabetes or other serious health conditions.
What is the rate of disease progression, related complications, or even mortality for these patients?
Dr. McGill: People with CKD and T2D are at risk for many things. One risk is progression of kidney disease all the way to end-stage kidney disease, which requires dialysis or transplantation. Another huge risk is cardiovascular events such as myocardial infarction (MI) and stroke.
Persons with kidney disease, for reasons we don't understand, are at higher risk of MI and stroke than people who do not have kidney disease. Therefore, the risks of early mortality and events that adversely affect quality of life are greatly increased.
Can you please discuss the economic burdens associated with T2D and CKD, and whether any interventions are in place to help offset those costs?
Dr. McGill: Diabetes itself is wickedly expensive. We have excellent treatments for diabetes today, but they are very costly. The best approach for the prevention of diabetes is to be screened. When a patient presents with prediabetes, it’s important that they take important measures, such as weight loss, exercising 150 minutes per week, or reducing 500 calories a day from their diet, all of which have been shown to forestall the onset of diabetes.
Once diabetes develops, achieving near-normal glucose control can either be very inexpensive with one or more generic drugs, or it can be terribly expensive with the newer branded drugs. Both options can help with the achievement of near-normal glucose levels, but the newer drugs are better for weight loss and provide protection from heart and kidney disease.
It is important to consider where the patient is along the disease spectrum, and to educate them on the benefits of taking a proactive approach to their health. Don’t wait for diabetes to develop before doing something about it. We have to take action earlier, and more definitively.
We do everything we can to help patients with the high cost of diabetes medications. Pharma companies offer various coupons and patient assistance programs, but it's really important that we get people on the right therapy. In order for that to happen, they have to come to office visits and get lab tests done.
Is there anything else you would like to share on this topic?
Dr. McGill: Once a person has been diagnosed with diabetes, then excellent glucose control from the onset has been shown to prevent later complications, and early treatment is inexpensive. As people progress through their journey with diabetes and blood sugars go up, we have excellent therapies that help manage high glucose and help with weight loss.
We have to be realistic and rethink our approach in some ways, but as long as people develop good health care habits and visit the doctor once or twice a year specifically to address diabetes and blood pressure, we might be able to avoid long-term complications.
Tirzepatide’s benefits expand: Lean mass up, serum lipids down
STOCKHOLM – New insights into the benefits of treatment with the “twincretin” tirzepatide for people with overweight or obesity – with or without diabetes – come from new findings reported at the annual meeting of the European Association for the Study of Diabetes.
Additional results from the SURMOUNT-1 trial, which matched tirzepatide against placebo in people with overweight or obesity, provide further details on the favorable changes produced by 72 weeks of tirzepatide treatment on outcomes that included fat and lean mass, insulin sensitivity, and patient-reported outcomes related to functional health and well being, reported Ania M. Jastreboff, MD, PhD.
And results from a meta-analysis of six trials that compared tirzepatide (Mounjaro) against several different comparators in patients with type 2 diabetes further confirm the drug’s ability to reliably produce positive changes in blood lipids, especially by significantly lowering levels of triglycerides, LDL cholesterol, and very LDL (VLDL) cholesterol, said Thomas Karagiannis, MD, PhD, in a separate report at the meeting.
Tirzepatide works as an agonist on receptors for both the glucagonlike peptide–1 (GLP-1), and for the glucose-dependent insulinotropic polypeptide, and received Food and Drug Administration approval for treating people with type 2 diabetes in May 2022. On the basis of results from SURMOUNT-1, the FDA on Oct. 6 granted tirzepatide fast-track designation for a proposed labeling of the agent for treating people with overweight or obesity. This FDA decision will likely remain pending at least until results from a second trial in people with overweight or obesity but without diabetes, SURMOUNT-2, become available in 2023.
SURMOUNT-1 randomized 2,539 people with obesity or overweight and at least one weight-related complication to a weekly injection of tirzepatide or placebo for 72 weeks. The study’s primary efficacy endpoints were the average reduction in weight from baseline, and the percentage of people in each treatment arm achieving weight loss of at least 5% from baseline.
For both endpoints, the outcomes with tirzepatide significantly surpassed placebo effects. Average weight loss ranged from 15%-21% from baseline, depending on dose, compared with 3% on placebo. The rate of participants with at least a 5% weight loss ranged from 85% to 91%, compared with 35% with placebo, as reported in July 2022 in the New England Journal of Medicine.
Cutting fat mass, boosting lean mass
New results from the trial reported by Dr. Jastreboff included a cut in fat mass from 46.2% of total body mass at baseline to 38.5% after 72 weeks, compared with a change from 46.8% at baseline to 44.7% after 72 weeks in the placebo group. Concurrently, lean mass increased with tirzepatide treatment from 51.0% at baseline to 58.1% after 72 weeks.
Participants who received tirzepatide, compared with those who received placebo, had “proportionately greater decrease in fat mass and proportionately greater increase in lean mass” compared with those who received placebo, said Dr. Jastreboff, an endocrinologist and obesity medicine specialist with Yale Medicine in New Haven, Conn. “I was impressed by the amount of visceral fat lost.”
These effects translated into a significant reduction in fat mass-to-lean mass ratio among the people treated with tirzepatide, with the greatest reduction in those who lost at least 15% of their starting weight. In that subgroup the fat-to-lean mass ratio dropped from 0.94 at baseline to 0.64 after 72 weeks of treatment, she said.
Focus on diet quality
People treated with tirzepatide “eat so little food that we need to improve the quality of what they eat to protect their muscle,” commented Carel le Roux, MBChB, PhD, a professor in the Diabetes Complications Research Centre of University College Dublin. “You no longer need a dietitian to help people lose weight, because the drug does that. You need dietitians to look after the nutritional health of patients while they lose weight,” Dr. le Roux said in a separate session at the meeting.
Additional tests showed that blood glucose and insulin levels were all significantly lower among trial participants on all three doses of tirzepatide compared with those on placebo, and the tirzepatide-treated subjects also had significant, roughly twofold elevations in their insulin sensitivity measured by the Matsuda Index.
The impact of tirzepatide on glucose and insulin levels and on insulin sensitivity was similar regardless of whether study participants had normoglycemia or prediabetes at entry. By design, no study participants had diabetes.
The trial assessed patient-reported quality-of-life outcomes using the 36-Item Short Form Survey (SF-36). Participants had significant increases in all eight domains within the SF-36 at all three tirzepatide doses, compared with placebo, at 72 weeks, Dr. Jastreboff reported. Improvements in the physical function domain increased most notably among study participants on tirzepatide who had functional limitations at baseline. Heart rate rose among participants who received either of the two highest tirzepatide doses by 2.3-2.5 beats/min, comparable with the effect of other injected incretin-based treatments.
Lipids improve in those with type 2 diabetes
Tirzepatide treatment also results in a “secondary effect” of improving levels of several lipids in people with type 2 diabetes, according to a meta-analysis of findings from six randomized trials. The meta-analysis collectively involved 4,502 participants treated for numerous weeks with one of three doses of tirzepatide and 2,144 people in comparator groups, reported Dr. Karagiannis, a diabetes researcher at Aristotle University of Thessaloniki (Greece).
Among the significant lipid changes linked with tirzepatide treatment, compared with placebo, were an average 13 mg/dL decrease in LDL cholesterol, an average 6 mg/dL decrease in VLDL cholesterol, and an average 50 mg/dL decrease in triglycerides. In comparison to a GLP-1 receptor agonist, an average 25 mg/dL decrease in triglycerides and an average 4 mg/dL reduction in VLDL cholesterol were seen. And trials comparing tirzepatide with basal insulin saw average reductions of 7% in LDL cholesterol, 15% in VLDL cholesterol, 15% in triglycerides, and an 8% increase in HDL cholesterol.
Dr. Karagiannis highlighted that the clinical impact of these effects is unclear, although he noted that the average reduction in LDL cholesterol relative to placebo is of a magnitude that could have a modest effect on long-term outcomes.
These lipid effects of tirzepatide “should be considered alongside” tirzepatide’s “key metabolic effects” on weight and hemoglobin A1c as well as the drug’s safety, concluded Dr. Karagiannis.
The tirzepatide trials were all funded by Eli Lilly, which markets tirzepatide (Mounjaro). Dr. Jastreboff has been an adviser and consultant to Eli Lilly, as well as to Intellihealth, Novo Nordisk, Pfizer, Rhythm Scholars, Roche, and Weight Watchers, and she has received research funding from Eli Lilly and Novo Nordisk. Dr. Karagiannis had no disclosures. Dr. le Roux has had financial relationships with Eli Lilly, as well as with Boehringer Ingelheim, Consilient Health, Covidion, Fractyl, GL Dynamics, Herbalife, Johnson & Johnson, Keyron, and Novo Nordisk.
STOCKHOLM – New insights into the benefits of treatment with the “twincretin” tirzepatide for people with overweight or obesity – with or without diabetes – come from new findings reported at the annual meeting of the European Association for the Study of Diabetes.
Additional results from the SURMOUNT-1 trial, which matched tirzepatide against placebo in people with overweight or obesity, provide further details on the favorable changes produced by 72 weeks of tirzepatide treatment on outcomes that included fat and lean mass, insulin sensitivity, and patient-reported outcomes related to functional health and well being, reported Ania M. Jastreboff, MD, PhD.
And results from a meta-analysis of six trials that compared tirzepatide (Mounjaro) against several different comparators in patients with type 2 diabetes further confirm the drug’s ability to reliably produce positive changes in blood lipids, especially by significantly lowering levels of triglycerides, LDL cholesterol, and very LDL (VLDL) cholesterol, said Thomas Karagiannis, MD, PhD, in a separate report at the meeting.
Tirzepatide works as an agonist on receptors for both the glucagonlike peptide–1 (GLP-1), and for the glucose-dependent insulinotropic polypeptide, and received Food and Drug Administration approval for treating people with type 2 diabetes in May 2022. On the basis of results from SURMOUNT-1, the FDA on Oct. 6 granted tirzepatide fast-track designation for a proposed labeling of the agent for treating people with overweight or obesity. This FDA decision will likely remain pending at least until results from a second trial in people with overweight or obesity but without diabetes, SURMOUNT-2, become available in 2023.
SURMOUNT-1 randomized 2,539 people with obesity or overweight and at least one weight-related complication to a weekly injection of tirzepatide or placebo for 72 weeks. The study’s primary efficacy endpoints were the average reduction in weight from baseline, and the percentage of people in each treatment arm achieving weight loss of at least 5% from baseline.
For both endpoints, the outcomes with tirzepatide significantly surpassed placebo effects. Average weight loss ranged from 15%-21% from baseline, depending on dose, compared with 3% on placebo. The rate of participants with at least a 5% weight loss ranged from 85% to 91%, compared with 35% with placebo, as reported in July 2022 in the New England Journal of Medicine.
Cutting fat mass, boosting lean mass
New results from the trial reported by Dr. Jastreboff included a cut in fat mass from 46.2% of total body mass at baseline to 38.5% after 72 weeks, compared with a change from 46.8% at baseline to 44.7% after 72 weeks in the placebo group. Concurrently, lean mass increased with tirzepatide treatment from 51.0% at baseline to 58.1% after 72 weeks.
Participants who received tirzepatide, compared with those who received placebo, had “proportionately greater decrease in fat mass and proportionately greater increase in lean mass” compared with those who received placebo, said Dr. Jastreboff, an endocrinologist and obesity medicine specialist with Yale Medicine in New Haven, Conn. “I was impressed by the amount of visceral fat lost.”
These effects translated into a significant reduction in fat mass-to-lean mass ratio among the people treated with tirzepatide, with the greatest reduction in those who lost at least 15% of their starting weight. In that subgroup the fat-to-lean mass ratio dropped from 0.94 at baseline to 0.64 after 72 weeks of treatment, she said.
Focus on diet quality
People treated with tirzepatide “eat so little food that we need to improve the quality of what they eat to protect their muscle,” commented Carel le Roux, MBChB, PhD, a professor in the Diabetes Complications Research Centre of University College Dublin. “You no longer need a dietitian to help people lose weight, because the drug does that. You need dietitians to look after the nutritional health of patients while they lose weight,” Dr. le Roux said in a separate session at the meeting.
Additional tests showed that blood glucose and insulin levels were all significantly lower among trial participants on all three doses of tirzepatide compared with those on placebo, and the tirzepatide-treated subjects also had significant, roughly twofold elevations in their insulin sensitivity measured by the Matsuda Index.
The impact of tirzepatide on glucose and insulin levels and on insulin sensitivity was similar regardless of whether study participants had normoglycemia or prediabetes at entry. By design, no study participants had diabetes.
The trial assessed patient-reported quality-of-life outcomes using the 36-Item Short Form Survey (SF-36). Participants had significant increases in all eight domains within the SF-36 at all three tirzepatide doses, compared with placebo, at 72 weeks, Dr. Jastreboff reported. Improvements in the physical function domain increased most notably among study participants on tirzepatide who had functional limitations at baseline. Heart rate rose among participants who received either of the two highest tirzepatide doses by 2.3-2.5 beats/min, comparable with the effect of other injected incretin-based treatments.
Lipids improve in those with type 2 diabetes
Tirzepatide treatment also results in a “secondary effect” of improving levels of several lipids in people with type 2 diabetes, according to a meta-analysis of findings from six randomized trials. The meta-analysis collectively involved 4,502 participants treated for numerous weeks with one of three doses of tirzepatide and 2,144 people in comparator groups, reported Dr. Karagiannis, a diabetes researcher at Aristotle University of Thessaloniki (Greece).
Among the significant lipid changes linked with tirzepatide treatment, compared with placebo, were an average 13 mg/dL decrease in LDL cholesterol, an average 6 mg/dL decrease in VLDL cholesterol, and an average 50 mg/dL decrease in triglycerides. In comparison to a GLP-1 receptor agonist, an average 25 mg/dL decrease in triglycerides and an average 4 mg/dL reduction in VLDL cholesterol were seen. And trials comparing tirzepatide with basal insulin saw average reductions of 7% in LDL cholesterol, 15% in VLDL cholesterol, 15% in triglycerides, and an 8% increase in HDL cholesterol.
Dr. Karagiannis highlighted that the clinical impact of these effects is unclear, although he noted that the average reduction in LDL cholesterol relative to placebo is of a magnitude that could have a modest effect on long-term outcomes.
These lipid effects of tirzepatide “should be considered alongside” tirzepatide’s “key metabolic effects” on weight and hemoglobin A1c as well as the drug’s safety, concluded Dr. Karagiannis.
The tirzepatide trials were all funded by Eli Lilly, which markets tirzepatide (Mounjaro). Dr. Jastreboff has been an adviser and consultant to Eli Lilly, as well as to Intellihealth, Novo Nordisk, Pfizer, Rhythm Scholars, Roche, and Weight Watchers, and she has received research funding from Eli Lilly and Novo Nordisk. Dr. Karagiannis had no disclosures. Dr. le Roux has had financial relationships with Eli Lilly, as well as with Boehringer Ingelheim, Consilient Health, Covidion, Fractyl, GL Dynamics, Herbalife, Johnson & Johnson, Keyron, and Novo Nordisk.
STOCKHOLM – New insights into the benefits of treatment with the “twincretin” tirzepatide for people with overweight or obesity – with or without diabetes – come from new findings reported at the annual meeting of the European Association for the Study of Diabetes.
Additional results from the SURMOUNT-1 trial, which matched tirzepatide against placebo in people with overweight or obesity, provide further details on the favorable changes produced by 72 weeks of tirzepatide treatment on outcomes that included fat and lean mass, insulin sensitivity, and patient-reported outcomes related to functional health and well being, reported Ania M. Jastreboff, MD, PhD.
And results from a meta-analysis of six trials that compared tirzepatide (Mounjaro) against several different comparators in patients with type 2 diabetes further confirm the drug’s ability to reliably produce positive changes in blood lipids, especially by significantly lowering levels of triglycerides, LDL cholesterol, and very LDL (VLDL) cholesterol, said Thomas Karagiannis, MD, PhD, in a separate report at the meeting.
Tirzepatide works as an agonist on receptors for both the glucagonlike peptide–1 (GLP-1), and for the glucose-dependent insulinotropic polypeptide, and received Food and Drug Administration approval for treating people with type 2 diabetes in May 2022. On the basis of results from SURMOUNT-1, the FDA on Oct. 6 granted tirzepatide fast-track designation for a proposed labeling of the agent for treating people with overweight or obesity. This FDA decision will likely remain pending at least until results from a second trial in people with overweight or obesity but without diabetes, SURMOUNT-2, become available in 2023.
SURMOUNT-1 randomized 2,539 people with obesity or overweight and at least one weight-related complication to a weekly injection of tirzepatide or placebo for 72 weeks. The study’s primary efficacy endpoints were the average reduction in weight from baseline, and the percentage of people in each treatment arm achieving weight loss of at least 5% from baseline.
For both endpoints, the outcomes with tirzepatide significantly surpassed placebo effects. Average weight loss ranged from 15%-21% from baseline, depending on dose, compared with 3% on placebo. The rate of participants with at least a 5% weight loss ranged from 85% to 91%, compared with 35% with placebo, as reported in July 2022 in the New England Journal of Medicine.
Cutting fat mass, boosting lean mass
New results from the trial reported by Dr. Jastreboff included a cut in fat mass from 46.2% of total body mass at baseline to 38.5% after 72 weeks, compared with a change from 46.8% at baseline to 44.7% after 72 weeks in the placebo group. Concurrently, lean mass increased with tirzepatide treatment from 51.0% at baseline to 58.1% after 72 weeks.
Participants who received tirzepatide, compared with those who received placebo, had “proportionately greater decrease in fat mass and proportionately greater increase in lean mass” compared with those who received placebo, said Dr. Jastreboff, an endocrinologist and obesity medicine specialist with Yale Medicine in New Haven, Conn. “I was impressed by the amount of visceral fat lost.”
These effects translated into a significant reduction in fat mass-to-lean mass ratio among the people treated with tirzepatide, with the greatest reduction in those who lost at least 15% of their starting weight. In that subgroup the fat-to-lean mass ratio dropped from 0.94 at baseline to 0.64 after 72 weeks of treatment, she said.
Focus on diet quality
People treated with tirzepatide “eat so little food that we need to improve the quality of what they eat to protect their muscle,” commented Carel le Roux, MBChB, PhD, a professor in the Diabetes Complications Research Centre of University College Dublin. “You no longer need a dietitian to help people lose weight, because the drug does that. You need dietitians to look after the nutritional health of patients while they lose weight,” Dr. le Roux said in a separate session at the meeting.
Additional tests showed that blood glucose and insulin levels were all significantly lower among trial participants on all three doses of tirzepatide compared with those on placebo, and the tirzepatide-treated subjects also had significant, roughly twofold elevations in their insulin sensitivity measured by the Matsuda Index.
The impact of tirzepatide on glucose and insulin levels and on insulin sensitivity was similar regardless of whether study participants had normoglycemia or prediabetes at entry. By design, no study participants had diabetes.
The trial assessed patient-reported quality-of-life outcomes using the 36-Item Short Form Survey (SF-36). Participants had significant increases in all eight domains within the SF-36 at all three tirzepatide doses, compared with placebo, at 72 weeks, Dr. Jastreboff reported. Improvements in the physical function domain increased most notably among study participants on tirzepatide who had functional limitations at baseline. Heart rate rose among participants who received either of the two highest tirzepatide doses by 2.3-2.5 beats/min, comparable with the effect of other injected incretin-based treatments.
Lipids improve in those with type 2 diabetes
Tirzepatide treatment also results in a “secondary effect” of improving levels of several lipids in people with type 2 diabetes, according to a meta-analysis of findings from six randomized trials. The meta-analysis collectively involved 4,502 participants treated for numerous weeks with one of three doses of tirzepatide and 2,144 people in comparator groups, reported Dr. Karagiannis, a diabetes researcher at Aristotle University of Thessaloniki (Greece).
Among the significant lipid changes linked with tirzepatide treatment, compared with placebo, were an average 13 mg/dL decrease in LDL cholesterol, an average 6 mg/dL decrease in VLDL cholesterol, and an average 50 mg/dL decrease in triglycerides. In comparison to a GLP-1 receptor agonist, an average 25 mg/dL decrease in triglycerides and an average 4 mg/dL reduction in VLDL cholesterol were seen. And trials comparing tirzepatide with basal insulin saw average reductions of 7% in LDL cholesterol, 15% in VLDL cholesterol, 15% in triglycerides, and an 8% increase in HDL cholesterol.
Dr. Karagiannis highlighted that the clinical impact of these effects is unclear, although he noted that the average reduction in LDL cholesterol relative to placebo is of a magnitude that could have a modest effect on long-term outcomes.
These lipid effects of tirzepatide “should be considered alongside” tirzepatide’s “key metabolic effects” on weight and hemoglobin A1c as well as the drug’s safety, concluded Dr. Karagiannis.
The tirzepatide trials were all funded by Eli Lilly, which markets tirzepatide (Mounjaro). Dr. Jastreboff has been an adviser and consultant to Eli Lilly, as well as to Intellihealth, Novo Nordisk, Pfizer, Rhythm Scholars, Roche, and Weight Watchers, and she has received research funding from Eli Lilly and Novo Nordisk. Dr. Karagiannis had no disclosures. Dr. le Roux has had financial relationships with Eli Lilly, as well as with Boehringer Ingelheim, Consilient Health, Covidion, Fractyl, GL Dynamics, Herbalife, Johnson & Johnson, Keyron, and Novo Nordisk.
AT EASD 2022
Older diabetes drugs linked to dementia risk -- one lower, one higher
a new observational study in patients with type 2 diabetes suggests.
The data, obtained from nationwide electronic medical records from the Department of Veterans Affairs, yielded a 22% lower risk of dementia with TZD monotherapy and a 12% elevated risk with sulfonylurea monotherapy, compared with metformin monotherapy. The apparent protective effects of TZDs were greater among individuals with overweight or obesity.
“Our findings provide additional information to aid clinicians’ selection of [glucose-lowering medications] for patients with mild or moderate type 2 diabetes and [who] are at high risk of dementia,” Xin Tang and colleagues wrote in their article, published online in BMJ Open Diabetes Research & Care.
The results “add substantially to the literature concerning the effects of [glucose-lowering medications] on dementia where previous findings have been inconsistent. Studies with a follow-up time of less than 3 years have mainly reported null associations, while studies with longer a follow-up time typically yielded protective findings. With a mean follow-up time of 6.8 years, we had a sufficient duration to detect treatment differences,” the investigators wrote.
“Supplementing [a] sulfonylurea with either metformin or [a] TZD may partially offset its prodementia effects. These findings may help inform medication selection for elderly patients with T2D at high risk of dementia,” they added.
Randomized trials needed to determine cause and effect
Ivan Koychev, PhD, a senior clinical researcher in the department of psychiatry at the University of Oxford (England), told the UK Science Media Centre: “This is a large, well-conducted real-world data study that highlights the importance of checking whether already prescribed medications may be useful for preventing dementia.”
The findings regarding TZDs, also known as glitazones, are in line with existing literature suggesting dementia protection with other drugs prescribed for type 2 diabetes that weren’t examined in the current study, such as newer agents like glucagonlike peptide–1 (GLP-1) agonists and sodium-glucose cotransporter 2 (SGLT2) inhibitors, Dr. Koychev said.
“The main limitations of this study is that following the initial 2-year period the authors were interested in, the participants may have been prescribed one of the other type 2 diabetes drugs [GLP-1 agonists or SGLT2 inhibitors] that have been found to reduce dementia risk, thus potentially making the direct glitazone [TZD] effect more difficult to discern,” Dr. Koychev noted.
And, he pointed out that the study design limits attribution of causality. “It is also important to note that people with type 2 diabetes do run a higher risk of both dementia and cognitive deficits and that these medications are only prescribed in these patients, so all this data is from this patient group rather than the general population.”
James Connell, PhD, head of translational science at Alzheimer’s Research UK, agreed. “While this observational study found that those with type 2 diabetes taking thiazolidinedione had a lower dementia risk than those on the most common medication for type 2 diabetes, it only shows an association between taking the drug and dementia risk and not a causal relationship.
“Double-blind and placebo-controlled clinical trials are needed to see whether the drug [TDZ] could help lower dementia risk in people with and without diabetes. Anyone with any questions about what treatments they are receiving should speak to their doctor,” he told the UK Science Media Centre.
Opposite effects of sulfonylureas, TZDs versus metformin
The study authors analyzed 559,106 VA patients with type 2 diabetes who initiated glucose-lowering medication during 2001-2017 and took it for at least a year. They were aged 60 years or older and did not have dementia at baseline. Most were White (76.8%) and male (96.9%), two-thirds (63.1%) had obesity, and mean hemoglobin A1c was 6.8%.
Overall, 31,125 developed all-cause dementia. The incidence rate was 8.2 cases per 1,000 person-years, ranging from 6.2 cases per 1,000 person-years among those taking metformin monotherapy to 13.4 cases per 1,000 person-years in those taking both sulfonylurea and a TZD.
Compared with metformin monotherapy, the hazard ratio for all-cause dementia for sulfonylurea monotherapy was a significant 1.12. The increased risk was also seen for vascular dementia, with an HR of 1.14.
In contrast, TZD monotherapy was associated with a significantly lower risk for all-cause dementia (HR, 0.78), as well as for Alzheimer’s disease (HR, 0.89) and vascular dementia (HR, 0.43), compared with metformin monotherapy.
The combination of metformin and TZD also lowered the risk of all-cause dementia, while regimens including sulfonylureas raised the risks for all-cause and vascular dementia.
Most of the results didn’t change significantly when the drug exposure window was extended to 2 years.
Effects more pronounced in those with obesity
The protective 1-year effects of TZD monotherapy and of metformin plus TZD, compared with metformin alone, were more significant among participants aged 75 or younger and with a body mass index above 25 kg/m2, compared with those who were older than 75 years and with normal BMIs, respectively.
On the other hand, the greater risk for dementia incurred with sulfonylureas was further increased among those with higher BMI.
This research was partially funded by grants from the National Human Genome Research Institute, the National Science Foundation, the National Institute of Diabetes and Digestive and Kidney Disease, and the National Heart, Lung, and Blood Institute. Dr. Koychev is chief investigator for a trial, sponsored by Oxford University and funded by Novo Nordisk, testing whether the GLP-1 agonist semaglutide reduces the risk for dementia in aging adults.
A version of this article first appeared on Medscape.com.
a new observational study in patients with type 2 diabetes suggests.
The data, obtained from nationwide electronic medical records from the Department of Veterans Affairs, yielded a 22% lower risk of dementia with TZD monotherapy and a 12% elevated risk with sulfonylurea monotherapy, compared with metformin monotherapy. The apparent protective effects of TZDs were greater among individuals with overweight or obesity.
“Our findings provide additional information to aid clinicians’ selection of [glucose-lowering medications] for patients with mild or moderate type 2 diabetes and [who] are at high risk of dementia,” Xin Tang and colleagues wrote in their article, published online in BMJ Open Diabetes Research & Care.
The results “add substantially to the literature concerning the effects of [glucose-lowering medications] on dementia where previous findings have been inconsistent. Studies with a follow-up time of less than 3 years have mainly reported null associations, while studies with longer a follow-up time typically yielded protective findings. With a mean follow-up time of 6.8 years, we had a sufficient duration to detect treatment differences,” the investigators wrote.
“Supplementing [a] sulfonylurea with either metformin or [a] TZD may partially offset its prodementia effects. These findings may help inform medication selection for elderly patients with T2D at high risk of dementia,” they added.
Randomized trials needed to determine cause and effect
Ivan Koychev, PhD, a senior clinical researcher in the department of psychiatry at the University of Oxford (England), told the UK Science Media Centre: “This is a large, well-conducted real-world data study that highlights the importance of checking whether already prescribed medications may be useful for preventing dementia.”
The findings regarding TZDs, also known as glitazones, are in line with existing literature suggesting dementia protection with other drugs prescribed for type 2 diabetes that weren’t examined in the current study, such as newer agents like glucagonlike peptide–1 (GLP-1) agonists and sodium-glucose cotransporter 2 (SGLT2) inhibitors, Dr. Koychev said.
“The main limitations of this study is that following the initial 2-year period the authors were interested in, the participants may have been prescribed one of the other type 2 diabetes drugs [GLP-1 agonists or SGLT2 inhibitors] that have been found to reduce dementia risk, thus potentially making the direct glitazone [TZD] effect more difficult to discern,” Dr. Koychev noted.
And, he pointed out that the study design limits attribution of causality. “It is also important to note that people with type 2 diabetes do run a higher risk of both dementia and cognitive deficits and that these medications are only prescribed in these patients, so all this data is from this patient group rather than the general population.”
James Connell, PhD, head of translational science at Alzheimer’s Research UK, agreed. “While this observational study found that those with type 2 diabetes taking thiazolidinedione had a lower dementia risk than those on the most common medication for type 2 diabetes, it only shows an association between taking the drug and dementia risk and not a causal relationship.
“Double-blind and placebo-controlled clinical trials are needed to see whether the drug [TDZ] could help lower dementia risk in people with and without diabetes. Anyone with any questions about what treatments they are receiving should speak to their doctor,” he told the UK Science Media Centre.
Opposite effects of sulfonylureas, TZDs versus metformin
The study authors analyzed 559,106 VA patients with type 2 diabetes who initiated glucose-lowering medication during 2001-2017 and took it for at least a year. They were aged 60 years or older and did not have dementia at baseline. Most were White (76.8%) and male (96.9%), two-thirds (63.1%) had obesity, and mean hemoglobin A1c was 6.8%.
Overall, 31,125 developed all-cause dementia. The incidence rate was 8.2 cases per 1,000 person-years, ranging from 6.2 cases per 1,000 person-years among those taking metformin monotherapy to 13.4 cases per 1,000 person-years in those taking both sulfonylurea and a TZD.
Compared with metformin monotherapy, the hazard ratio for all-cause dementia for sulfonylurea monotherapy was a significant 1.12. The increased risk was also seen for vascular dementia, with an HR of 1.14.
In contrast, TZD monotherapy was associated with a significantly lower risk for all-cause dementia (HR, 0.78), as well as for Alzheimer’s disease (HR, 0.89) and vascular dementia (HR, 0.43), compared with metformin monotherapy.
The combination of metformin and TZD also lowered the risk of all-cause dementia, while regimens including sulfonylureas raised the risks for all-cause and vascular dementia.
Most of the results didn’t change significantly when the drug exposure window was extended to 2 years.
Effects more pronounced in those with obesity
The protective 1-year effects of TZD monotherapy and of metformin plus TZD, compared with metformin alone, were more significant among participants aged 75 or younger and with a body mass index above 25 kg/m2, compared with those who were older than 75 years and with normal BMIs, respectively.
On the other hand, the greater risk for dementia incurred with sulfonylureas was further increased among those with higher BMI.
This research was partially funded by grants from the National Human Genome Research Institute, the National Science Foundation, the National Institute of Diabetes and Digestive and Kidney Disease, and the National Heart, Lung, and Blood Institute. Dr. Koychev is chief investigator for a trial, sponsored by Oxford University and funded by Novo Nordisk, testing whether the GLP-1 agonist semaglutide reduces the risk for dementia in aging adults.
A version of this article first appeared on Medscape.com.
a new observational study in patients with type 2 diabetes suggests.
The data, obtained from nationwide electronic medical records from the Department of Veterans Affairs, yielded a 22% lower risk of dementia with TZD monotherapy and a 12% elevated risk with sulfonylurea monotherapy, compared with metformin monotherapy. The apparent protective effects of TZDs were greater among individuals with overweight or obesity.
“Our findings provide additional information to aid clinicians’ selection of [glucose-lowering medications] for patients with mild or moderate type 2 diabetes and [who] are at high risk of dementia,” Xin Tang and colleagues wrote in their article, published online in BMJ Open Diabetes Research & Care.
The results “add substantially to the literature concerning the effects of [glucose-lowering medications] on dementia where previous findings have been inconsistent. Studies with a follow-up time of less than 3 years have mainly reported null associations, while studies with longer a follow-up time typically yielded protective findings. With a mean follow-up time of 6.8 years, we had a sufficient duration to detect treatment differences,” the investigators wrote.
“Supplementing [a] sulfonylurea with either metformin or [a] TZD may partially offset its prodementia effects. These findings may help inform medication selection for elderly patients with T2D at high risk of dementia,” they added.
Randomized trials needed to determine cause and effect
Ivan Koychev, PhD, a senior clinical researcher in the department of psychiatry at the University of Oxford (England), told the UK Science Media Centre: “This is a large, well-conducted real-world data study that highlights the importance of checking whether already prescribed medications may be useful for preventing dementia.”
The findings regarding TZDs, also known as glitazones, are in line with existing literature suggesting dementia protection with other drugs prescribed for type 2 diabetes that weren’t examined in the current study, such as newer agents like glucagonlike peptide–1 (GLP-1) agonists and sodium-glucose cotransporter 2 (SGLT2) inhibitors, Dr. Koychev said.
“The main limitations of this study is that following the initial 2-year period the authors were interested in, the participants may have been prescribed one of the other type 2 diabetes drugs [GLP-1 agonists or SGLT2 inhibitors] that have been found to reduce dementia risk, thus potentially making the direct glitazone [TZD] effect more difficult to discern,” Dr. Koychev noted.
And, he pointed out that the study design limits attribution of causality. “It is also important to note that people with type 2 diabetes do run a higher risk of both dementia and cognitive deficits and that these medications are only prescribed in these patients, so all this data is from this patient group rather than the general population.”
James Connell, PhD, head of translational science at Alzheimer’s Research UK, agreed. “While this observational study found that those with type 2 diabetes taking thiazolidinedione had a lower dementia risk than those on the most common medication for type 2 diabetes, it only shows an association between taking the drug and dementia risk and not a causal relationship.
“Double-blind and placebo-controlled clinical trials are needed to see whether the drug [TDZ] could help lower dementia risk in people with and without diabetes. Anyone with any questions about what treatments they are receiving should speak to their doctor,” he told the UK Science Media Centre.
Opposite effects of sulfonylureas, TZDs versus metformin
The study authors analyzed 559,106 VA patients with type 2 diabetes who initiated glucose-lowering medication during 2001-2017 and took it for at least a year. They were aged 60 years or older and did not have dementia at baseline. Most were White (76.8%) and male (96.9%), two-thirds (63.1%) had obesity, and mean hemoglobin A1c was 6.8%.
Overall, 31,125 developed all-cause dementia. The incidence rate was 8.2 cases per 1,000 person-years, ranging from 6.2 cases per 1,000 person-years among those taking metformin monotherapy to 13.4 cases per 1,000 person-years in those taking both sulfonylurea and a TZD.
Compared with metformin monotherapy, the hazard ratio for all-cause dementia for sulfonylurea monotherapy was a significant 1.12. The increased risk was also seen for vascular dementia, with an HR of 1.14.
In contrast, TZD monotherapy was associated with a significantly lower risk for all-cause dementia (HR, 0.78), as well as for Alzheimer’s disease (HR, 0.89) and vascular dementia (HR, 0.43), compared with metformin monotherapy.
The combination of metformin and TZD also lowered the risk of all-cause dementia, while regimens including sulfonylureas raised the risks for all-cause and vascular dementia.
Most of the results didn’t change significantly when the drug exposure window was extended to 2 years.
Effects more pronounced in those with obesity
The protective 1-year effects of TZD monotherapy and of metformin plus TZD, compared with metformin alone, were more significant among participants aged 75 or younger and with a body mass index above 25 kg/m2, compared with those who were older than 75 years and with normal BMIs, respectively.
On the other hand, the greater risk for dementia incurred with sulfonylureas was further increased among those with higher BMI.
This research was partially funded by grants from the National Human Genome Research Institute, the National Science Foundation, the National Institute of Diabetes and Digestive and Kidney Disease, and the National Heart, Lung, and Blood Institute. Dr. Koychev is chief investigator for a trial, sponsored by Oxford University and funded by Novo Nordisk, testing whether the GLP-1 agonist semaglutide reduces the risk for dementia in aging adults.
A version of this article first appeared on Medscape.com.
FROM BMJ OPEN DIABETES RESEARCH & CARE
New advice on artificial pancreas insulin delivery systems
A new consensus statement summarizes the benefits, limitations, and challenges of using automated insulin delivery (AID) systems and provides recommendations for use by people with diabetes.
“Automated insulin delivery systems” is becoming the standard terminology – including by the U.S. Food and Drug Administration – to refer to systems that integrate data from a continuous glucose monitoring (CGM) system via a control algorithm into an insulin pump in order to automate subcutaneous insulin delivery. “Hybrid AID” or “hybrid closed-loop” refers to the current status of these systems, which still require some degree of user input to control glucose levels.
The term “artificial pancreas” was used interchangeably with AID in the past, but it doesn’t take into account exocrine pancreatic function. The term “bionic pancreas” refers to a specific system in development that would ultimately include glucagon along with insulin.
The new consensus report, titled “Automated insulin delivery: Benefits, challenges, and recommendations,” was published online in Diabetes Care and Diabetologia.
The document is geared toward not only diabetologists and other specialists, but also diabetes nurses and specialist dietitians. Colleagues working at regulatory agencies, health care organizations, and related media might also benefit from reading it.
It is endorsed by two professional societies – the European Association for the Study of Diabetes and the American Diabetes Association – and contrasts with other statements about AID systems that are sponsored by their manufacturers, noted document co-author Mark Evans, PhD, professor of diabetic medicine, University of Cambridge, England, in a statement.
“Many clinically relevant aspects, including safety, are addressed in this report. The aim ... is to encourage ongoing improvement of this technology, its safe and effective use, and its accessibility to all who can benefit from it,” Dr. Evans said.
Lead author Jennifer Sherr, MD, PhD, pediatric endocrinology, Yale University, New Haven, Conn., commented that the report “addresses the clinical usage of AID systems from a practical point of view rather than as ... a meta-analysis or a review of all relevant clinical studies. ... As such, the benefits and limitations of systems are discussed while also considering safety, regulatory pathways, and access to this technology.”
AID systems do not mean diabetes is “cured”
Separate recommendations provided at the end of the document are aimed at specific stakeholders, including health care providers, patients and their caregivers, manufacturers, regulatory agencies, and the research community.
The authors make clear in the introduction that, while representing “a significant movement toward optimizing glucose management for individuals with diabetes,” the use of AID systems doesn’t mean that diabetes is “cured.” Rather, “expectations need to be set adequately so that individuals with diabetes and providers understand what such systems can and cannot do.”
In particular, current commercially available AID systems require user input for mealtime insulin dosing and sometimes for correction doses of high blood glucose levels, although the systems at least partially automate that.
“When integrated into care, AID systems hold promise to relieve some of the daily burdens of diabetes care,” the authors write.
The statement also details problems that may arise with the physical devices, including skin irritation from adhesives, occlusion of insulin infusion sets, early CGM sensor failure, and inadequate dosing algorithms.
“Individuals with diabetes who are considering this type of advanced diabetes therapy should not only have appropriate technical understanding of the system but also be able to revert to standard diabetes treatment (that is, nonautomated subcutaneous insulin delivery by pump or injections) in case the AID system fails. They should be able to independently troubleshoot and have access to their health care provider if needed.”
To monitor the impact of the technology, the authors emphasize the importance of the time-in-range metric derived from CGM, with the goal of achieving 70% or greater time in target blood glucose range.
Separate sections of the document address the benefits and limitations of AID systems, education and expectations for both patients and providers, and patient and provider perspectives, including how to handle urgent questions.
Other sections cover special populations such as pregnant women and people with type 2 diabetes, considerations for patient selection for current AID systems, safety, improving access to the technology, liability, and do-it-yourself systems.
Recommendations for health care professionals
A table near the end of the document provides specific recommendations for health care professionals, including the following:
- Be knowledgeable about AID systems and nuances of different systems, including their distinguishing features as well as strengths and weaknesses.
- Inform patients with diabetes about AID systems, including review of currently available systems, and create realistic expectations for device use.
- Involve patients with diabetes in shared decision-making when considering use of AID systems.
- Share information with patients with diabetes, as well as their peers, about general standards set by national and international guidelines on AID systems.
- Provide an on-call number or method by which a person with diabetes can always access support from a health care provider at the practice, including weekends and nights.
- Implement, potentially, protocols on times when AID systems should not be used.
- Use an individual’s health data to improve quality of care and health outcomes.
Most members of the ADA/EASD Diabetes Technology Working Group work with industry, but industry had no input on the project. Dr. Sherr has reported conducting clinical trials for Eli Lilly, Insulet, and Medtronic, and has received in-kind support for research studies from Dexcom and Medtronic. She has also reported consulting for Eli Lilly, Lexicon, Medtronic, and Sanofi, and being an advisory board member for Bigfoot Biomedical, Cecelia Health, Eli Lilly, Insulet, T1D Fund, and Vertex Pharmaceuticals. Dr. Evans has reported conducting clinical trials or research collaborations for, serving on advisory boards for, or receiving speakers fees or travel support from Medtronic, Roche, Abbott Diabetes Care, Dexcom, Novo Nordisk, Eli Lilly, Sanofi, Zucara Therapeutics, Pila Pharma, and AstraZeneca. The University of Cambridge has received salary support for Dr. Evans from the National Health Service.
A version of this article first appeared on Medscape.com.
A new consensus statement summarizes the benefits, limitations, and challenges of using automated insulin delivery (AID) systems and provides recommendations for use by people with diabetes.
“Automated insulin delivery systems” is becoming the standard terminology – including by the U.S. Food and Drug Administration – to refer to systems that integrate data from a continuous glucose monitoring (CGM) system via a control algorithm into an insulin pump in order to automate subcutaneous insulin delivery. “Hybrid AID” or “hybrid closed-loop” refers to the current status of these systems, which still require some degree of user input to control glucose levels.
The term “artificial pancreas” was used interchangeably with AID in the past, but it doesn’t take into account exocrine pancreatic function. The term “bionic pancreas” refers to a specific system in development that would ultimately include glucagon along with insulin.
The new consensus report, titled “Automated insulin delivery: Benefits, challenges, and recommendations,” was published online in Diabetes Care and Diabetologia.
The document is geared toward not only diabetologists and other specialists, but also diabetes nurses and specialist dietitians. Colleagues working at regulatory agencies, health care organizations, and related media might also benefit from reading it.
It is endorsed by two professional societies – the European Association for the Study of Diabetes and the American Diabetes Association – and contrasts with other statements about AID systems that are sponsored by their manufacturers, noted document co-author Mark Evans, PhD, professor of diabetic medicine, University of Cambridge, England, in a statement.
“Many clinically relevant aspects, including safety, are addressed in this report. The aim ... is to encourage ongoing improvement of this technology, its safe and effective use, and its accessibility to all who can benefit from it,” Dr. Evans said.
Lead author Jennifer Sherr, MD, PhD, pediatric endocrinology, Yale University, New Haven, Conn., commented that the report “addresses the clinical usage of AID systems from a practical point of view rather than as ... a meta-analysis or a review of all relevant clinical studies. ... As such, the benefits and limitations of systems are discussed while also considering safety, regulatory pathways, and access to this technology.”
AID systems do not mean diabetes is “cured”
Separate recommendations provided at the end of the document are aimed at specific stakeholders, including health care providers, patients and their caregivers, manufacturers, regulatory agencies, and the research community.
The authors make clear in the introduction that, while representing “a significant movement toward optimizing glucose management for individuals with diabetes,” the use of AID systems doesn’t mean that diabetes is “cured.” Rather, “expectations need to be set adequately so that individuals with diabetes and providers understand what such systems can and cannot do.”
In particular, current commercially available AID systems require user input for mealtime insulin dosing and sometimes for correction doses of high blood glucose levels, although the systems at least partially automate that.
“When integrated into care, AID systems hold promise to relieve some of the daily burdens of diabetes care,” the authors write.
The statement also details problems that may arise with the physical devices, including skin irritation from adhesives, occlusion of insulin infusion sets, early CGM sensor failure, and inadequate dosing algorithms.
“Individuals with diabetes who are considering this type of advanced diabetes therapy should not only have appropriate technical understanding of the system but also be able to revert to standard diabetes treatment (that is, nonautomated subcutaneous insulin delivery by pump or injections) in case the AID system fails. They should be able to independently troubleshoot and have access to their health care provider if needed.”
To monitor the impact of the technology, the authors emphasize the importance of the time-in-range metric derived from CGM, with the goal of achieving 70% or greater time in target blood glucose range.
Separate sections of the document address the benefits and limitations of AID systems, education and expectations for both patients and providers, and patient and provider perspectives, including how to handle urgent questions.
Other sections cover special populations such as pregnant women and people with type 2 diabetes, considerations for patient selection for current AID systems, safety, improving access to the technology, liability, and do-it-yourself systems.
Recommendations for health care professionals
A table near the end of the document provides specific recommendations for health care professionals, including the following:
- Be knowledgeable about AID systems and nuances of different systems, including their distinguishing features as well as strengths and weaknesses.
- Inform patients with diabetes about AID systems, including review of currently available systems, and create realistic expectations for device use.
- Involve patients with diabetes in shared decision-making when considering use of AID systems.
- Share information with patients with diabetes, as well as their peers, about general standards set by national and international guidelines on AID systems.
- Provide an on-call number or method by which a person with diabetes can always access support from a health care provider at the practice, including weekends and nights.
- Implement, potentially, protocols on times when AID systems should not be used.
- Use an individual’s health data to improve quality of care and health outcomes.
Most members of the ADA/EASD Diabetes Technology Working Group work with industry, but industry had no input on the project. Dr. Sherr has reported conducting clinical trials for Eli Lilly, Insulet, and Medtronic, and has received in-kind support for research studies from Dexcom and Medtronic. She has also reported consulting for Eli Lilly, Lexicon, Medtronic, and Sanofi, and being an advisory board member for Bigfoot Biomedical, Cecelia Health, Eli Lilly, Insulet, T1D Fund, and Vertex Pharmaceuticals. Dr. Evans has reported conducting clinical trials or research collaborations for, serving on advisory boards for, or receiving speakers fees or travel support from Medtronic, Roche, Abbott Diabetes Care, Dexcom, Novo Nordisk, Eli Lilly, Sanofi, Zucara Therapeutics, Pila Pharma, and AstraZeneca. The University of Cambridge has received salary support for Dr. Evans from the National Health Service.
A version of this article first appeared on Medscape.com.
A new consensus statement summarizes the benefits, limitations, and challenges of using automated insulin delivery (AID) systems and provides recommendations for use by people with diabetes.
“Automated insulin delivery systems” is becoming the standard terminology – including by the U.S. Food and Drug Administration – to refer to systems that integrate data from a continuous glucose monitoring (CGM) system via a control algorithm into an insulin pump in order to automate subcutaneous insulin delivery. “Hybrid AID” or “hybrid closed-loop” refers to the current status of these systems, which still require some degree of user input to control glucose levels.
The term “artificial pancreas” was used interchangeably with AID in the past, but it doesn’t take into account exocrine pancreatic function. The term “bionic pancreas” refers to a specific system in development that would ultimately include glucagon along with insulin.
The new consensus report, titled “Automated insulin delivery: Benefits, challenges, and recommendations,” was published online in Diabetes Care and Diabetologia.
The document is geared toward not only diabetologists and other specialists, but also diabetes nurses and specialist dietitians. Colleagues working at regulatory agencies, health care organizations, and related media might also benefit from reading it.
It is endorsed by two professional societies – the European Association for the Study of Diabetes and the American Diabetes Association – and contrasts with other statements about AID systems that are sponsored by their manufacturers, noted document co-author Mark Evans, PhD, professor of diabetic medicine, University of Cambridge, England, in a statement.
“Many clinically relevant aspects, including safety, are addressed in this report. The aim ... is to encourage ongoing improvement of this technology, its safe and effective use, and its accessibility to all who can benefit from it,” Dr. Evans said.
Lead author Jennifer Sherr, MD, PhD, pediatric endocrinology, Yale University, New Haven, Conn., commented that the report “addresses the clinical usage of AID systems from a practical point of view rather than as ... a meta-analysis or a review of all relevant clinical studies. ... As such, the benefits and limitations of systems are discussed while also considering safety, regulatory pathways, and access to this technology.”
AID systems do not mean diabetes is “cured”
Separate recommendations provided at the end of the document are aimed at specific stakeholders, including health care providers, patients and their caregivers, manufacturers, regulatory agencies, and the research community.
The authors make clear in the introduction that, while representing “a significant movement toward optimizing glucose management for individuals with diabetes,” the use of AID systems doesn’t mean that diabetes is “cured.” Rather, “expectations need to be set adequately so that individuals with diabetes and providers understand what such systems can and cannot do.”
In particular, current commercially available AID systems require user input for mealtime insulin dosing and sometimes for correction doses of high blood glucose levels, although the systems at least partially automate that.
“When integrated into care, AID systems hold promise to relieve some of the daily burdens of diabetes care,” the authors write.
The statement also details problems that may arise with the physical devices, including skin irritation from adhesives, occlusion of insulin infusion sets, early CGM sensor failure, and inadequate dosing algorithms.
“Individuals with diabetes who are considering this type of advanced diabetes therapy should not only have appropriate technical understanding of the system but also be able to revert to standard diabetes treatment (that is, nonautomated subcutaneous insulin delivery by pump or injections) in case the AID system fails. They should be able to independently troubleshoot and have access to their health care provider if needed.”
To monitor the impact of the technology, the authors emphasize the importance of the time-in-range metric derived from CGM, with the goal of achieving 70% or greater time in target blood glucose range.
Separate sections of the document address the benefits and limitations of AID systems, education and expectations for both patients and providers, and patient and provider perspectives, including how to handle urgent questions.
Other sections cover special populations such as pregnant women and people with type 2 diabetes, considerations for patient selection for current AID systems, safety, improving access to the technology, liability, and do-it-yourself systems.
Recommendations for health care professionals
A table near the end of the document provides specific recommendations for health care professionals, including the following:
- Be knowledgeable about AID systems and nuances of different systems, including their distinguishing features as well as strengths and weaknesses.
- Inform patients with diabetes about AID systems, including review of currently available systems, and create realistic expectations for device use.
- Involve patients with diabetes in shared decision-making when considering use of AID systems.
- Share information with patients with diabetes, as well as their peers, about general standards set by national and international guidelines on AID systems.
- Provide an on-call number or method by which a person with diabetes can always access support from a health care provider at the practice, including weekends and nights.
- Implement, potentially, protocols on times when AID systems should not be used.
- Use an individual’s health data to improve quality of care and health outcomes.
Most members of the ADA/EASD Diabetes Technology Working Group work with industry, but industry had no input on the project. Dr. Sherr has reported conducting clinical trials for Eli Lilly, Insulet, and Medtronic, and has received in-kind support for research studies from Dexcom and Medtronic. She has also reported consulting for Eli Lilly, Lexicon, Medtronic, and Sanofi, and being an advisory board member for Bigfoot Biomedical, Cecelia Health, Eli Lilly, Insulet, T1D Fund, and Vertex Pharmaceuticals. Dr. Evans has reported conducting clinical trials or research collaborations for, serving on advisory boards for, or receiving speakers fees or travel support from Medtronic, Roche, Abbott Diabetes Care, Dexcom, Novo Nordisk, Eli Lilly, Sanofi, Zucara Therapeutics, Pila Pharma, and AstraZeneca. The University of Cambridge has received salary support for Dr. Evans from the National Health Service.
A version of this article first appeared on Medscape.com.
FROM DIABETES CARE AND DIABETOLOGIA
Going the distance with our patients
Many years ago, I had a patient I’ll call “Hannah,” who was well into her 80s and always came into the office with her daughter. She was a heavy smoker and had hypertension and type 2 diabetes.
At each visit, I would ask her if she still smoked and if she was interested in talking about quitting. At every visit, she would say that she was still smoking and didn’t want to quit. My response was always something along the lines of: “When you’re ready, we can talk more. But I think it is the most important thing you can do to improve your health.” From there, we would discuss any concerns she or her daughter had.
A few years shy of her 100th birthday, Hannah told me she had quit smoking. I was amazed and asked her why, after all these years, she’d done it.
“I quit,” she said, “because I was tired of you nagging me, sonny!” And we both had a good laugh about that.
Hannah’s story reminds me that, as family physicians, we often have an impact on our patients in ways we don’t see in the short term. It is our longitudinal relationships with patients that allow us to plant seeds and reap the benefits over time.
It is these relationships that we can draw upon when counseling our patients with type 2 diabetes to address lifestyle issues such as exercise and a healthy diet. In this issue, McMullan et al1 provide us with a rather hopeful review of the evidence in support of lifestyle changes. For our patients with type 2 diabetes, lifestyle changes can decrease A1C levels by 0.5% (with environmental changes related to diet)2 and 0.7% (with moderate aerobic exercise).3 This is comparable to what is reported for the starting doses of most medications.4 In fact, a meta-analysis showed that a low-carbohydrate diet induced remission at 6 months in 32% of patients.5 (Caveat: The result was not controlled for weight loss as a possible confounding factor and an A1C cutoff of 6.5% was used.)
And yet, we often focus more on the various medications we can prescribe, with professional guidelines pointing the way.
Continue to: The National Institute for Health and Care Excellence
The National Institute for Health and Care Excellence,6 American Diabetes Association,7 American College of Physicians,8 and American Academy of Family Physicians8 have followed the accumulating evidence that various medications improve outcomes—especially in patients at high risk or with established atherosclerotic cardiovascular disease. They have endorsed a stepwise pharmacologic approach beginning with metformin and recommend assessing each patient’s comorbidities to guide whether to add a sodium glucose co-transporter 2 (SGLT2) inhibitor or another agent. Where the groups diverge is what that second agent should be (glucagon-like peptide 1 receptor agonist, SGLT2 inhibitor, or dipeptidyl peptidase-4 inhibitor).
But what about lifestyle? Each organization’s guidelines address lifestyle changes as a foundation for managing patients with type 2 diabetes. But is that call loud enough? Do we heed it well enough?
Implementing lifestyle changes in office practice can be time consuming. Many clinicians lack adequate training or experience to gain any traction with it. Also, there is skepticism about success and sustainability.
I believe change starts when we recognize that while we have a priority list for each patient encounter, so do our patients. But they may not share that list with us unless we open the door by asking questions, such as:
- Of all the things you have heard about caring for your diabetes, what would you like to work on?
- What are you currently doing and what prevents you from meeting your goals?
- How would you like me to help you?
From there, we can start small and build on successes over time. We can go the distance with our patients. In the case of Hannah, I had the honor of caring for her until she died at age 104.
1. McMullan S, Smith DK, Kimsey J. Maximizing lifestyle changes to manage type 2 diabetes. J Fam Pract. 2022;71;342-348. doi: 10.12788/jfp.0482
2. Cradock KA, ÓLaighin G, Finucane FM, et al. Diet behavior change techniques in type 2 diabetes: a systematic review and meta-analysis. Diabetes Care. 2017;40:1800-1810. doi: 10.2337/dc17-0462
3. Grace A, Chan E, Giallauria F, et al. Clinical outcomes and glycaemic responses to different aerobic exercise training intensities in type II diabetes: a systematic review and meta-analysis. Cardiovasc Diabetol. 2017;16:37. doi: 10.1186/s12933-017-0518-6
Many years ago, I had a patient I’ll call “Hannah,” who was well into her 80s and always came into the office with her daughter. She was a heavy smoker and had hypertension and type 2 diabetes.
At each visit, I would ask her if she still smoked and if she was interested in talking about quitting. At every visit, she would say that she was still smoking and didn’t want to quit. My response was always something along the lines of: “When you’re ready, we can talk more. But I think it is the most important thing you can do to improve your health.” From there, we would discuss any concerns she or her daughter had.
A few years shy of her 100th birthday, Hannah told me she had quit smoking. I was amazed and asked her why, after all these years, she’d done it.
“I quit,” she said, “because I was tired of you nagging me, sonny!” And we both had a good laugh about that.
Hannah’s story reminds me that, as family physicians, we often have an impact on our patients in ways we don’t see in the short term. It is our longitudinal relationships with patients that allow us to plant seeds and reap the benefits over time.
It is these relationships that we can draw upon when counseling our patients with type 2 diabetes to address lifestyle issues such as exercise and a healthy diet. In this issue, McMullan et al1 provide us with a rather hopeful review of the evidence in support of lifestyle changes. For our patients with type 2 diabetes, lifestyle changes can decrease A1C levels by 0.5% (with environmental changes related to diet)2 and 0.7% (with moderate aerobic exercise).3 This is comparable to what is reported for the starting doses of most medications.4 In fact, a meta-analysis showed that a low-carbohydrate diet induced remission at 6 months in 32% of patients.5 (Caveat: The result was not controlled for weight loss as a possible confounding factor and an A1C cutoff of 6.5% was used.)
And yet, we often focus more on the various medications we can prescribe, with professional guidelines pointing the way.
Continue to: The National Institute for Health and Care Excellence
The National Institute for Health and Care Excellence,6 American Diabetes Association,7 American College of Physicians,8 and American Academy of Family Physicians8 have followed the accumulating evidence that various medications improve outcomes—especially in patients at high risk or with established atherosclerotic cardiovascular disease. They have endorsed a stepwise pharmacologic approach beginning with metformin and recommend assessing each patient’s comorbidities to guide whether to add a sodium glucose co-transporter 2 (SGLT2) inhibitor or another agent. Where the groups diverge is what that second agent should be (glucagon-like peptide 1 receptor agonist, SGLT2 inhibitor, or dipeptidyl peptidase-4 inhibitor).
But what about lifestyle? Each organization’s guidelines address lifestyle changes as a foundation for managing patients with type 2 diabetes. But is that call loud enough? Do we heed it well enough?
Implementing lifestyle changes in office practice can be time consuming. Many clinicians lack adequate training or experience to gain any traction with it. Also, there is skepticism about success and sustainability.
I believe change starts when we recognize that while we have a priority list for each patient encounter, so do our patients. But they may not share that list with us unless we open the door by asking questions, such as:
- Of all the things you have heard about caring for your diabetes, what would you like to work on?
- What are you currently doing and what prevents you from meeting your goals?
- How would you like me to help you?
From there, we can start small and build on successes over time. We can go the distance with our patients. In the case of Hannah, I had the honor of caring for her until she died at age 104.
Many years ago, I had a patient I’ll call “Hannah,” who was well into her 80s and always came into the office with her daughter. She was a heavy smoker and had hypertension and type 2 diabetes.
At each visit, I would ask her if she still smoked and if she was interested in talking about quitting. At every visit, she would say that she was still smoking and didn’t want to quit. My response was always something along the lines of: “When you’re ready, we can talk more. But I think it is the most important thing you can do to improve your health.” From there, we would discuss any concerns she or her daughter had.
A few years shy of her 100th birthday, Hannah told me she had quit smoking. I was amazed and asked her why, after all these years, she’d done it.
“I quit,” she said, “because I was tired of you nagging me, sonny!” And we both had a good laugh about that.
Hannah’s story reminds me that, as family physicians, we often have an impact on our patients in ways we don’t see in the short term. It is our longitudinal relationships with patients that allow us to plant seeds and reap the benefits over time.
It is these relationships that we can draw upon when counseling our patients with type 2 diabetes to address lifestyle issues such as exercise and a healthy diet. In this issue, McMullan et al1 provide us with a rather hopeful review of the evidence in support of lifestyle changes. For our patients with type 2 diabetes, lifestyle changes can decrease A1C levels by 0.5% (with environmental changes related to diet)2 and 0.7% (with moderate aerobic exercise).3 This is comparable to what is reported for the starting doses of most medications.4 In fact, a meta-analysis showed that a low-carbohydrate diet induced remission at 6 months in 32% of patients.5 (Caveat: The result was not controlled for weight loss as a possible confounding factor and an A1C cutoff of 6.5% was used.)
And yet, we often focus more on the various medications we can prescribe, with professional guidelines pointing the way.
Continue to: The National Institute for Health and Care Excellence
The National Institute for Health and Care Excellence,6 American Diabetes Association,7 American College of Physicians,8 and American Academy of Family Physicians8 have followed the accumulating evidence that various medications improve outcomes—especially in patients at high risk or with established atherosclerotic cardiovascular disease. They have endorsed a stepwise pharmacologic approach beginning with metformin and recommend assessing each patient’s comorbidities to guide whether to add a sodium glucose co-transporter 2 (SGLT2) inhibitor or another agent. Where the groups diverge is what that second agent should be (glucagon-like peptide 1 receptor agonist, SGLT2 inhibitor, or dipeptidyl peptidase-4 inhibitor).
But what about lifestyle? Each organization’s guidelines address lifestyle changes as a foundation for managing patients with type 2 diabetes. But is that call loud enough? Do we heed it well enough?
Implementing lifestyle changes in office practice can be time consuming. Many clinicians lack adequate training or experience to gain any traction with it. Also, there is skepticism about success and sustainability.
I believe change starts when we recognize that while we have a priority list for each patient encounter, so do our patients. But they may not share that list with us unless we open the door by asking questions, such as:
- Of all the things you have heard about caring for your diabetes, what would you like to work on?
- What are you currently doing and what prevents you from meeting your goals?
- How would you like me to help you?
From there, we can start small and build on successes over time. We can go the distance with our patients. In the case of Hannah, I had the honor of caring for her until she died at age 104.
1. McMullan S, Smith DK, Kimsey J. Maximizing lifestyle changes to manage type 2 diabetes. J Fam Pract. 2022;71;342-348. doi: 10.12788/jfp.0482
2. Cradock KA, ÓLaighin G, Finucane FM, et al. Diet behavior change techniques in type 2 diabetes: a systematic review and meta-analysis. Diabetes Care. 2017;40:1800-1810. doi: 10.2337/dc17-0462
3. Grace A, Chan E, Giallauria F, et al. Clinical outcomes and glycaemic responses to different aerobic exercise training intensities in type II diabetes: a systematic review and meta-analysis. Cardiovasc Diabetol. 2017;16:37. doi: 10.1186/s12933-017-0518-6
1. McMullan S, Smith DK, Kimsey J. Maximizing lifestyle changes to manage type 2 diabetes. J Fam Pract. 2022;71;342-348. doi: 10.12788/jfp.0482
2. Cradock KA, ÓLaighin G, Finucane FM, et al. Diet behavior change techniques in type 2 diabetes: a systematic review and meta-analysis. Diabetes Care. 2017;40:1800-1810. doi: 10.2337/dc17-0462
3. Grace A, Chan E, Giallauria F, et al. Clinical outcomes and glycaemic responses to different aerobic exercise training intensities in type II diabetes: a systematic review and meta-analysis. Cardiovasc Diabetol. 2017;16:37. doi: 10.1186/s12933-017-0518-6
Maximizing lifestyle changes to manage type 2 diabetes
Type 2 diabetes has been increasing in incidence and prevalence over the past 20 years, with worldwide prevalence estimated at 6.28%.1 The estimated cost of diagnosed diabetes in the United States was $327 billion in 2017; this included direct medical costs and reduced productivity.2 Type 2 diabetes can be prevented in most patients, given that it is a metabolic derangement caused by a complicated interaction between a patient’s genetic predisposition and lifestyle. A consensus statement by the American Academy of Clinical Endocrinologists (AACE) and American College of Endocrinology indicates that the recommended lifestyle modifications for diabetes include medical nutrition therapy with healthy eating patterns, regular physical activity, adequate sleep, behavioral support/counseling, and smoking cessation.3 Evidence shows that adherence to these lifestyle changes alone yields a relative reduction in type 2 diabetes mortality of 57%.4

In the discussion that follows, we review the current guideline recommendations for dietary modifications and physical activity and summarize their effectiveness in the treatment of type 2 diabetes. We also describe practical clinical strategies to promote change in patient behavior, and examine current literature supporting intensive lifestyle changes that, if achieved, may induce disease remission.5
Dietary strategies
Low, or very low, carbohydrate diet
Carbohydrates can affect blood glucose levels in varying degrees depending on their intrinsic properties such as fiber content, sugars, and starches . 6 According to the American Diabetes Association’s (ADA) 2019 consensus report, 6 the carbohydrate quality that generally should be recommended is high in fiber, vitamins, and minerals, and low in added sugars, fats, and sodium (processed carbohydrates) ( TABLE 1 7-10 ). A low-carbohydrate diet (LCD) typically has a carbohydrate content < 130 g/d or < 26% of a 2000 kcal/d diet. 11 A very low–carbohydrate diet (VLCD) is 20-50 g/d or < 10% of the 2000 kcal/day diet. 11
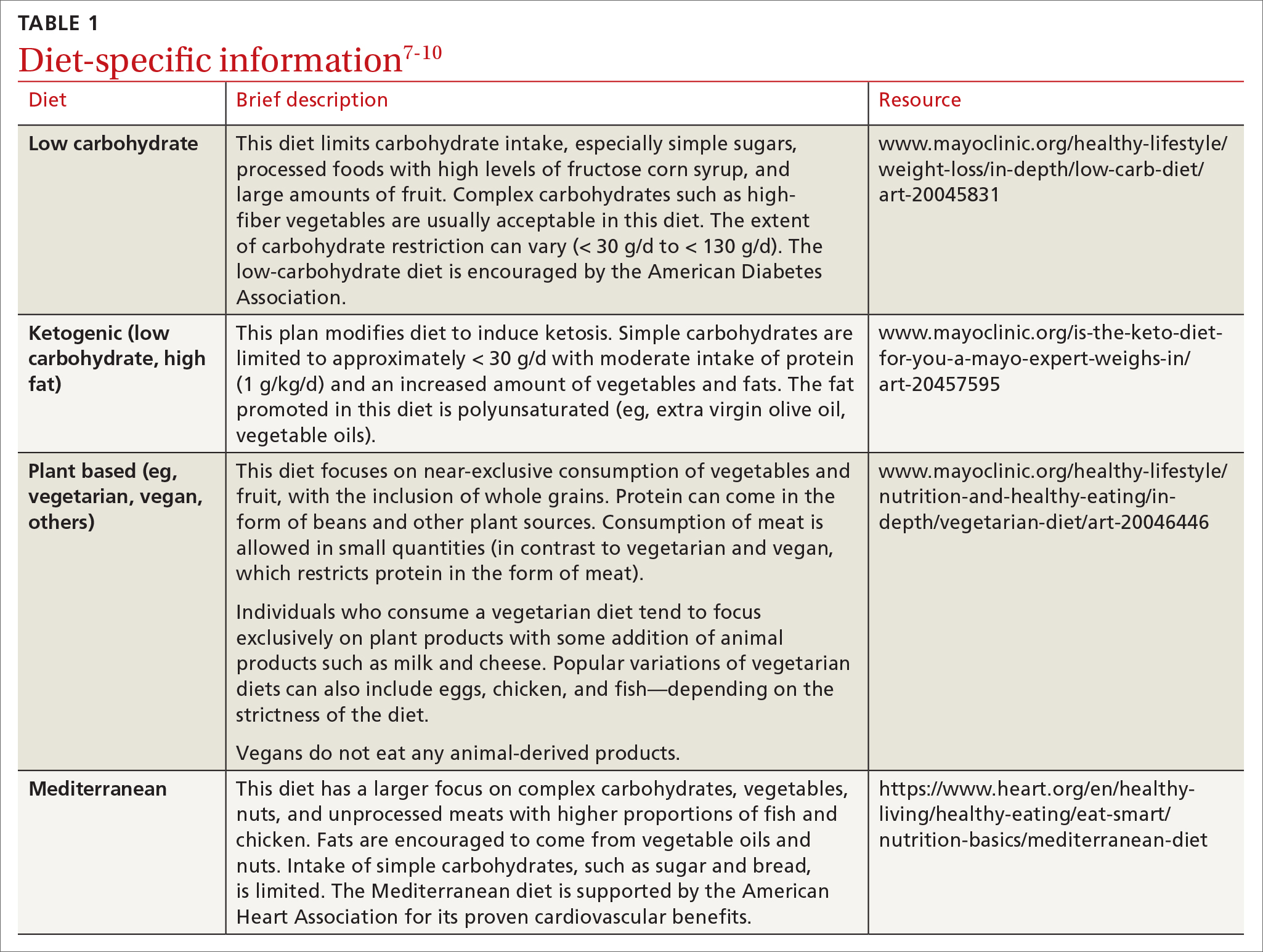
In a meta-analysis by Goldenberg et al11, the LCD was shown to reduce A1C by 0.47% at 6 months (95% CI, –0.6 to –0.34) and by 0.23% at 12 months when compared with control diets. A review of multiple meta-analyses also showed a significant reduction in A1C especially with VLCD patterns; however, the results waned at the 12-month follow-up.5 In addition, confounding factors were seen when comparing adherence between LCD and VLCD, with patients in the latter group having larger problems with adherence, which decreased the benefit seen in the overall group comparison.11
Very low–carbohydrate/high-fat (ketogenic) diet
Ketogenic diets generally follow a VLCD with the carbohydrate portion set at 5% to 10% of total caloric intake (generally < 30 g/d) and the rest of the calories taken up by protein (typically 1 g/kg/d) and fat (TABLE 17-10).12 The fat content recommended is primarily polyunsaturated fat such as olive oil, while saturated fats such as butter and lard (animal fat) should be limited.
A recent meta-analysis by Choi et al12 showed that in overweight or obese patients with type 2 diabetes, the average A1C reduction was 0.62% (95% CI, –0.89 to –0.35) in the ketogenic intervention group. Another meta-analysis showed an even more significant A1C reduction at 1.07% (95% CI, –1.37 to –0.78).13 Concerns have been raised about the ketogenic diet, particularly as it relates to lipid metabolism and cholesterol levels; however, in the 2 referenced meta-analyses, the total cholesterol and triglyceride levels actually declined in the ketogenic intervention groups with minimal effect on LDL-C.12,13 This may alleviate some of the concerns of lipid management with this diet.
Plant-based diet
Popularized by Dr. T. Colin Campbell, a plant-based diet refers to a low-fat, high-fiber, whole-foods diet (whole fruits, vegetables, and naturally occurring carbohydrates, as opposed to processed foods). Examples of this type of diet include the popular vegan diet, which restricts all animal-derived products, and the vegetarian diet, which is generally limited to foods in the plant category with some addition of animal products, such as milk and cheese. Other variations of these diets exist and include other sources of protein (eg, chicken, eggs, or fish) (TABLE 17-10).
Continue to: A review by...
A review by Salas-Salvadó et al14 showed that a vegan diet yields an average A1C reduction of 0.41% (95% CI, –0.58 to –0.23).Several meta-analyses report similar effects on A1C with vegetarian and vegan eating patterns.6,15,16 The ADA review notes that weight loss was more significant in the vegan group and concluded that this diet should be studied further while controlling for weight loss.6
Mediterranean diet
The Mediterranean diet emphasizes vegetables, whole grains, fruits, lean meats, nuts, and olive oil. The benefits of the Mediterranean diet are well known and, as a result, the diet is recommended by organizations including the American Heart Association as part of a strategy to reduce cardiovascular risk (TABLE 17-10).
Mediterranean diet interventions have generally shown mixed effects on A1C reduction, weight management, and lipid control in type 2 diabetes. 6 The PREDIMED trial is the largest and longest randomized controlled trial to date comparing the Mediterranean diet to a low-fat diet. 17 This trial has reliably shown a reduced risk for type 2 diabetes and a trend to reduced A1C. 17 A reduction in the need for glucose-lowering medications was demonstrated in a subgroup analysis of the intervention group (adjusted hazard ratio = 0.78; 95% CI, 0.62-0.98). 18 Also, the Mediterranean diet has shown a significant reduction in the incidence of cardiovascular disease in patients with type 2 diabetes. 6
Physical activity and exercise
What do current guidelines recommend?
For most adults with type 2 diabetes, current guidelines by the ADA and by the National Institute of Diabetes and Digestive and Kidney Diseases recommend at least 150 minutes of moderate-to-vigorous intensity exercise every week spread out over at least 3 days, with no more than 2 consecutive days without exercise; and resistance training at least 2 other days per week which should balance all major muscle groups (TABLE 219-21). The benefits of exercise for type 2 diabetes have been well reviewed: positive effects on glucose control, insulin sensitivity, cardiovascular disease, lipid profiles, skeletal muscle metabolism, and solid-organ functioning.19,22,23
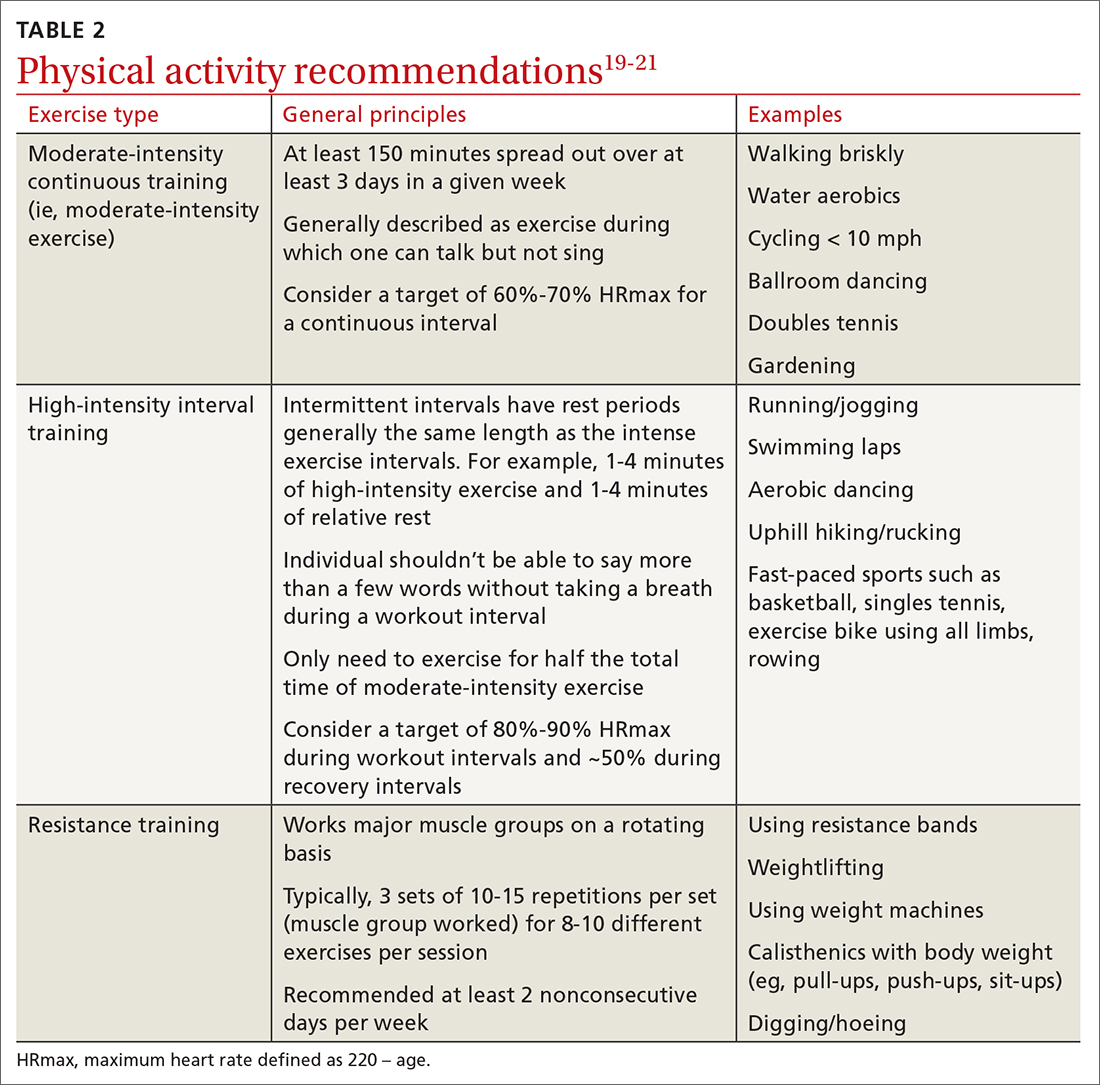
Grace et al24 showed in a meta-analysis that moderate aerobic exercise reduced A1C by 0.69% (95% CI, –1.09 to –0.3) at 13 weeks, and a Cochrane review showed an average A1C reduction of 0.6% with moderate-intensity exercise.25 Borror et al26 demonstrated in a systematic review that postprandial moderate-intensity aerobic exercise starting 1 hour after meals results in a reduced 24-hour prevalence of hyperglycemia (33.5% reduction vs control). A meta-analysis in China showed an average A1C reduction of 0.68% for patients performing a Tai Chi physical activity intervention.27
Continue to: Consider high-intensity interval training
Consider high-intensity interval training
Multiple randomized controlled trials highlight the benefits of high-intensity interval training (HIIT) (TABLE 219-21) compared with moderate-intensity continuous training (MICT) on improving A1C. A meta-analysis showed a weighted mean difference in A1C of 0.23% (95% CI, –0.43 to –0.02%).28 Also, a patient could spend less time performing HIIT as opposed to MICT to achieve the same benefits. For example, a patient typically performing 30 minutes of MICT may only need to perform 15 minutes of HIIT,a time-saving option for patients.20,22
Interrupt sedentary behavior
Risk for incident type 2 diabetes increases when someone is sedentary for more than 6 to 8 hours daily or watches TV for 3 to 4 hours (relative risk [RR] = 1.12).29 Recommendations for interrupting a sedentary lifestyle include standing from a seated position at least every 30 minutes and engaging in a light activity during the break interval for at least 3 minutes.19 Most studies have reliably shown that interrupting sedentary behavior reduces postprandial and 24-hour average blood glucose levels.19 Interrupted sitting/sedentary behavior has also been shown to reduce resting blood pressure in patients with type 2 diabetes.30
O ther important lifestyle factors
Encourage 7 to 8 hours of sleep
There is a U-shaped association between glycemic control and sleep quantity based on a meta-analysis by Lee et al 31 that showed a 0.23% increase in A1C in patients with insufficient sleep (< 4.5-6 hours/night) and a 0.13% increase in patients with ≥ 8 hours of sleep per night. Patients should be encouraged to obtain 7 to 8 hours of sleep per night to help maximize their diabetes control.
Address stress reduction
Although evidence for stress reduction interventions on glycemic control is mixed, there does seem to be a benefit in diminishing emotional distress in patients with diabetes. A systematic review by Noordali et al32 demonstrated that patients who received mindfulness-based interventions had improvements in stress, anxiety, and depression symptoms which resulted in improved quality of life. These psychological benefits may subsequently lead to positive behavioral changes.
Assist patients with smoking cessation
A large meta-analysis showed that active smoking increases the risk of cardiovascular events in patients with type 2 diabetes (RR = 1.44; 95% CI, 1.34-1.54).33 Former smokers still have an increased risk (RR = 1.09; 95% CI, 1.05-1.13), but it is lower than that of current smokers, so patients should be encouraged to quit smoking.3,33
Continue to: How can I get my patient to change?
H ow can I get my patient to change?
The AACE recommends using motivational interviewing, behavioral therapy consultation, and wearable feedback devices (eg, accelerometers/pedometers) to stimulate behavioral change in patients.3 Motivational interviewing is the principal counseling strategy and is supported by multiple studies showing the benefits of using this technique in a clinical encounter to induce behavioral changes.34 In general, offer receptive patients intensive behavioral interventions and provide them with resources to accomplish their goals.35 For example, a 7-step yearly intensive behavioral counseling intervention over 3 years showed significant improvements in activity of any intensity, reduced sedentary time, and led to favorable metabolic outcomes.36 Wearable devices result in up to a 1 hour increase in physical activity per week for the wearers vs control, although there was no appreciable effect on A1C.37
One systematic review showed a 0.5% reduction in A1C (95% CI, –0.65 to –0.34) by focusing on environmental changes related to the diet, with the most effective intervention being full meal replacement for calorie control (ie, each meal was pre-made and provided to the patients based on macronutrient and caloric goals).38 Additionally, diabetes self-management education includes coping strategies, problem solving, self-advocacy, and health care system navigation, which have been shown to reduce A1C by an average of 0.6%.21 Patient resources are available for further assistance with lifestyle modifications (TABLE 3).
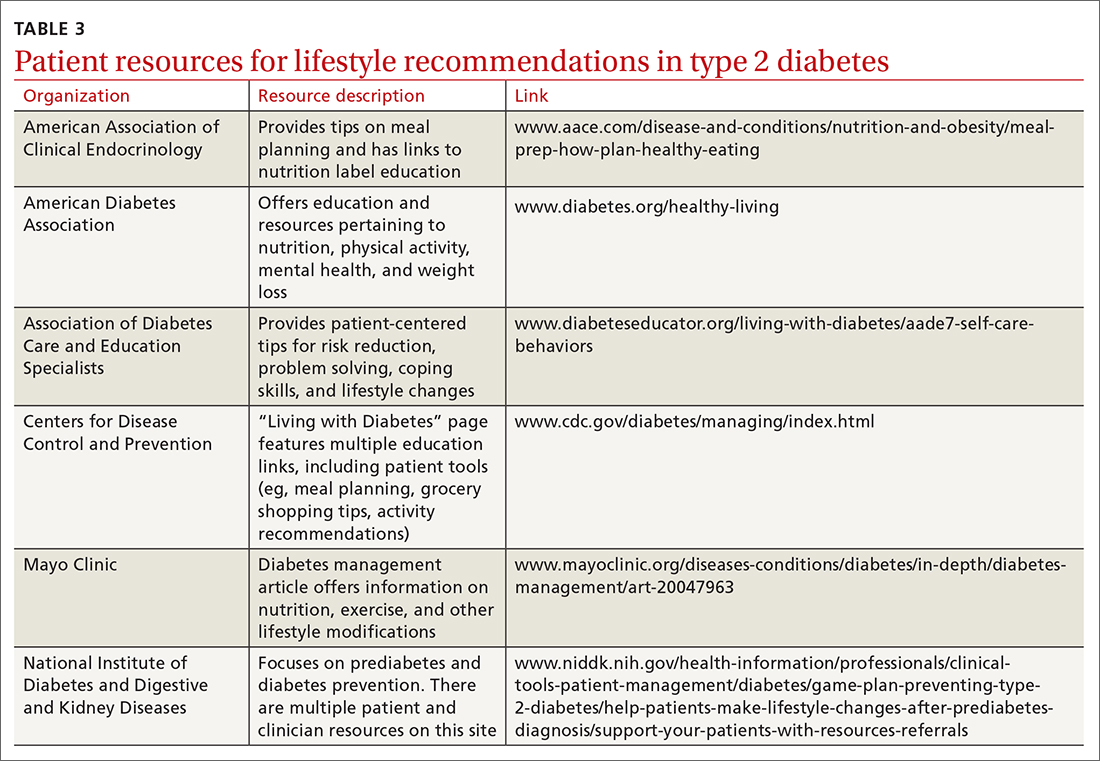
C an your patient achieve remission?
Emerging evidence suggests that patients may achieve remission from type 2 diabetes with intensive lifestyle interventions.39 This is supported by the American College of Lifestyle Medicine.5 Although there is no consensus definition for remission, in general it is reasonable to presume remission if a patient achieves normo-glycemia (A1C < 5.7%) for at least 1 year without any medication therapy.5 These intensive lifestyle interventions would include a mostly plant-based diet with moderate calorie restriction, appropriate and sustained physical activity, adequate sleep, and stress-reduction techniques.5 One study found that 46% of patients in a weight-management program across multiple primary care clinics achieved remission at 12 months.40 A meta-analysis showed that a low-carbohydrate diet induced remission at 6 months in 32% of patients (although the result was not controlled for weight loss as a possible confounding factor and an A1C cutoff of 6.5% was used).11 Thus far, most studies have focused on short-term follow-up intervals, but evidence is emerging that with intensive lifestyle interventions the effects are sustained at the 2-year mark.41
This evidence could reframe our understanding of type 2 diabetes therapy and could change the conversations we have with patients regarding their treatment. Instead of focusing on an A1C goal that is adequate for control of type 2 diabetes, we would instead focus on achieving remission.
CORRESPONDENCE
Stephen McMullan, MD, Mayo Clinic College of Medicine and Science, 4500 San Pablo Road, Jacksonville, FL 32224; mcmullan.stephen@mayo.edu
1. Kahn MAB, Hashim MJ, King JK, et al. Epidemiology of type 2 diabetes – global burden of disease and forecasted trends. J Epidemiol Glob Health. 2020;10:107-111. doi: 10.2991/jegh.k.191028.001
2. American Diabetes Association. Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41:917-928. doi:10.2337/dci18-0007
3. Garber AJ, Handelsman Y, Grunberger G, et al. Consensus Statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the Comprehensive Type 2 Diabetes Management Algorithm – 2020 Executive Summary. Endocr Pract. 2020;26:107-139. doi:10.4158/CS-2019-0472
4. Schlesinger S, Neuenschwander M, Ballon A, et al. Adherence to healthy lifestyles and incidence of diabetes and mortality among individuals with diabetes: a systematic review and meta-analysis of prospective studies. J Epidemiol Community Health. 2020;74:481-487. doi: 10.1136/jech-2019-213415
5. Kelly J, Karlsen M, Steinke G. Type 2 Diabetes Remission and Lifestyle Medicine: A Position Statement from the American College of Lifestyle Medicine. Am J Lifestyle Med. 2020;14:406-419. doi: 10.1177/1559827620930962
6. Evert AB, Dennison M, Gardner CD, et al. Nutrition Therapy for Adults with Diabetes or Prediabetes: A Consensus Report. Diabetes Care. 2019;42:731-754. doi: 10.2337/dci19-0014
7. Mayo Clinic. Low-carb diet: Can it help you lose weight? Accessed August 22, 2022. www.mayoclinic.org/healthylifestyle/weight-loss/in-depth/low-carb-diet/art-20045831
8. Mayo Clinic. Is the keto diet for You? A Mayo expert weighs in. Accessed September 16, 2022. www.mayoclinic.org/is-the-keto-diet-for-you-a-mayo-expert-weighs-in/art-20457595
9. Mayo Clinic. Vegetarian diet: How to get the best nutrition. Accessed August 22, 2022. www.mayoclinic.org/healthy-lifestyle/nutrition-and-healthy-eating/in-depth/vegetarian-diet/art-20046446
10. AHA. What is the Mediterranean diet? Accessed September 16, 2022. www.heart.org/en/healthy-living/healthy-eating/eat-smart/nutrition-basics/mediterranean-diet
11. Goldenberg JZ, Day A, Brinkworth GD, et al. Efficacy and safety of low and very low carbohydrate diets for type 2 diabetes remission: systematic review and meta-analysis of published and unpublished randomized trial data. BMJ. 2021;372:m4743. doi: 10.1136/bmj.m4743
12. Choi YJ, Jeon SM, Shin S. Impact of a ketogenic diet on metabolic parameters in patients with obesity or overweight and with or without type 2 diabetes: a meta-analysis of randomized controlled trials. Nutrients. 2020;12:2005. doi: 10.3390/nu12072005
13. Yuan X, Wang J, Yang S, et al. Effect of the ketogenic diet on glycemic control, insulin resistance, and lipid metabolism in patients with T2DM: a systematic review and meta-analysis. Nutr Diabetes. 2020;10:38. doi: 10.1038/s41387-020-00142-z
14. Salas-Salvadó J, Becerra-Tomás N, Papandreou C, et al. Dietary patterns emphasizing the consumption of plant foods in the management of type 2 diabetes: a narrative review. Adv Nutr. 2019;10(suppl_4):S320-S331. doi: 10.1093/advances/nmy102
15. Viguiliouk E, Kendall CW, Kahleová H, et al. Effect of vegetarian dietary patterns on cardiometabolic risk factors in diabetes: a systematic review and meta-analysis of randomized controlled trials. Clin Nutr. 2018;38:1133-1145. doi: 10.1016/j.clnu.2018.05.032
16. Yokoyama Y, Barnard ND, Levin SM, et al. Vegetarian diets and glycemic control in diabetes: a systematic review and meta-analysis. Cardiovasc Diagn Ther. 2014;4:373-382. doi: 10.3978/j.issn.2223-3652.2014.10.04
17. Estruch R, Ros E, Salas-Salvadó J, et al. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. N Engl J Med. 2018;378:e34. doi: 10.1056/NEJMoa1800389
18. Basterra-Gortari FJ, Ruiz-Canela M, Martínez-González MA, et al. Effects of a Mediterranean eating plan on the need for glucose-lowering medications in participants with type 2 diabetes: a subgroup analysis of the PREDIMED trial. Diabetes Care. 2019;42:1390-1397. doi: 10.2337/dc18-2475
19. Colberg SR, Sigal RJ, Yardley JE, et al. Physical Activity/Exercise and Diabetes: A position Statement of the American Diabetes Association. Diabetes Care. 2016;39:2065-2079. doi:10.2337/dc16-1728
20. Hwang CL, Lim J, Yoo JK, et al. Effect of all-extremity high-intensity interval training vs. moderate-intensity continuous training on aerobic fitness in middle-aged and older adults with type 2 diabetes: a randomized controlled trial. Exp Gerontol. 2019;116:46-53. doi:10.1016/j.exger.2018.12.013
21. Zangeneh F, Boltri J, Dallas A, et al. National Institute of Diabetes and Digestive and Kidney Diseases. Guiding principles for the care of people with or at risk for diabetes. Accessed September 16, 2022. www.niddk.nih.gov/health-information/professionals/clinical-tools-patient-management/diabetes/guiding-principles-care-people-risk-diabetes
22. Kirwan JP, Sacks J, Nieuwoudt S. The essential role of exercise in the management of type 2 diabetes. Cleve Clin J Med. 2017;84(7 suppl 1):S15-S21. doi: 10.3949/ccjm.84.s1.03
23. Zanuso S, Sacchetti M, Sundberg CJ, et al. Exercise in type 2 diabetes: genetic, metabolic and neuromuscular adaptations. a review of the evidence. Br J Sports Med. 2017;51:1533-1538. doi: 10.1136/bjsports-2016-096724
24. Grace A, Chan E, Giallauria F, et al. Clinical outcomes and glycaemic responses to different aerobic exercise training intensities in type II diabetes: a systematic review and meta-analysis. Cardiovasc Diabetol. 2017;16:37. Published 2017 Mar 14. doi: 10.1186/s12933-017-0518-6
25. Thomas DE, Elliott EJ, Naughton GA. Exercise for type 2 diabetes mellitus. Cochrane Database Syst Rev. 2006;(3):CD002968. doi: 10.1002/14651858.CD002968.pub2
26. Borror A, Zieff G, Battaglini C, et al. The effects of postprandial exercise on glucose control in individuals with type 2 diabetes: a systematic review. Sports Med. 2018;48:1479-1491. doi: 10.1007/s40279-018-0864-x
27. Xia TW, Yang Y, Li WH, et al. Different training durations and styles of tai chi for glucose control in patients with type 2 diabetes: a systematic review and meta-analysis of controlled trials. BMC Complement Altern Med. 2019;19:63. doi: 10.1186/s12906-019-2475-y
28. Liubaoerjijin Y, Terada T, Fletcher K, et al. Effect of aerobic exercise intensity on glycemic control in type 2 diabetes: a meta-analysis of head-to-head randomized trials. Acta Diabetol. 2016;53:769-781. doi: 10.1007/s00592-016-0870-0
29. Patterson R, McNamara E, Tainio M, et al. Sedentary behaviour and risk of all-cause, cardiovascular and cancer mortality, and incident type 2 diabetes: a systematic review and dose response meta-analysis. Eur J Epidemiol. 2018;33:811-829. doi: 10.1007/s10654-018-0380-1
30. Dempsey PC, Sacre JW, Larsen RN, et al. Interrupting prolonged sitting with brief bouts of light walking or simple resistance activities reduces resting blood pressure and plasma noradrenaline in type 2 diabetes. J Hypertens. 2016;34:2376-2382. doi: 10.1097/HJH.0000000000001101
31. Lee SWH, Ng KY, Chin WK. The impact of sleep amount and sleep quality on glycemic control in type 2 diabetes: a systematic review and meta-analysis. Sleep Med Rev. 2017;31:91-101. doi: 10.1016/j.smrv.2016.02.001.
32. Noordali F, Cumming J, Thompson JL. Effectiveness of mindfulness-based intervention on physiological and psychological complications in adults with diabetes: a systematic review. J Health Psychol. 2017;22:965-983. doi: 10.1177/1359105315620293
33. Pan A, Wang Y, Talaei M, et al. Relation of smoking with total mortality and cardiovascular events among patients with diabetes mellitus: a meta-analysis and systematic review. Circulation. 2015;132:1795-1804. doi:10.116/circulationaha.115.017926
34. VanBuskirk KA, Wetherell JL. Motivational interviewing with primary care populations: a systematic review and meta-analysis. J Behav Med. 2014;37:768-780. doi:10.1007/s10865-013-9527-4
35. Koenigsberg MR, Corliss J. Diabetes self-management: facilitating lifestyle change. Am Fam Physician. 2017;96:362-370.
36. Balducci S, D’Errico V, Haxhi J, et al. Effect of a behavioral intervention strategy for adoption and maintenance of a physically active lifestyle: the Italian Diabetes and Exercise Study 2 (IDES_2): a randomized controlled trial. Diabetes Care. 2017;40:1444-1452. doi: 10.2337/dc17-0594
37. Baskerville R, Ricci-Cabello I, Roberts N, et al. Impact of accelerometer and pedometer use on physical activity and glycaemic control in people with type 2 diabetes: a systematic review and meta-analysis. Diabet Med. 2017;34:612-620. doi:10.1111/dme.13331
38. Cradock KA, ÓLaighin G, Finucane FM, et al. Diet behavior change techniques in type 2 diabetes: a systematic review and meta-analysis. Diabetes Care. 2017;40:1800-1810. doi: 10.2337/dc17-0462
39. Hallberg SJ, Gershuni VM, Hazbun TL, et al. Reversing type 2 diabetes: a narrative review of the evidence. Nutrients. 2019;11:766. doi: 10.3390/nu11040766
40. Lean MEJ, Leslie WS, Barnes AC, et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): an open-label, cluster-randomised trial. Lancet. 2018;391:541-551. doi: 10.1016/S0140-6736(17)33102-1
41. Sbroma Tomaro E, Pippi R, Reginato E, et al. Intensive lifestyle intervention is particularly advantageous in poorly controlled type 2 diabetes. Nutr Metab Cardiovasc Dis. 2017;27:688-694. doi:10.1016/j.numecd.2017.06.009
Type 2 diabetes has been increasing in incidence and prevalence over the past 20 years, with worldwide prevalence estimated at 6.28%.1 The estimated cost of diagnosed diabetes in the United States was $327 billion in 2017; this included direct medical costs and reduced productivity.2 Type 2 diabetes can be prevented in most patients, given that it is a metabolic derangement caused by a complicated interaction between a patient’s genetic predisposition and lifestyle. A consensus statement by the American Academy of Clinical Endocrinologists (AACE) and American College of Endocrinology indicates that the recommended lifestyle modifications for diabetes include medical nutrition therapy with healthy eating patterns, regular physical activity, adequate sleep, behavioral support/counseling, and smoking cessation.3 Evidence shows that adherence to these lifestyle changes alone yields a relative reduction in type 2 diabetes mortality of 57%.4

In the discussion that follows, we review the current guideline recommendations for dietary modifications and physical activity and summarize their effectiveness in the treatment of type 2 diabetes. We also describe practical clinical strategies to promote change in patient behavior, and examine current literature supporting intensive lifestyle changes that, if achieved, may induce disease remission.5
Dietary strategies
Low, or very low, carbohydrate diet
Carbohydrates can affect blood glucose levels in varying degrees depending on their intrinsic properties such as fiber content, sugars, and starches . 6 According to the American Diabetes Association’s (ADA) 2019 consensus report, 6 the carbohydrate quality that generally should be recommended is high in fiber, vitamins, and minerals, and low in added sugars, fats, and sodium (processed carbohydrates) ( TABLE 1 7-10 ). A low-carbohydrate diet (LCD) typically has a carbohydrate content < 130 g/d or < 26% of a 2000 kcal/d diet. 11 A very low–carbohydrate diet (VLCD) is 20-50 g/d or < 10% of the 2000 kcal/day diet. 11

In a meta-analysis by Goldenberg et al11, the LCD was shown to reduce A1C by 0.47% at 6 months (95% CI, –0.6 to –0.34) and by 0.23% at 12 months when compared with control diets. A review of multiple meta-analyses also showed a significant reduction in A1C especially with VLCD patterns; however, the results waned at the 12-month follow-up.5 In addition, confounding factors were seen when comparing adherence between LCD and VLCD, with patients in the latter group having larger problems with adherence, which decreased the benefit seen in the overall group comparison.11
Very low–carbohydrate/high-fat (ketogenic) diet
Ketogenic diets generally follow a VLCD with the carbohydrate portion set at 5% to 10% of total caloric intake (generally < 30 g/d) and the rest of the calories taken up by protein (typically 1 g/kg/d) and fat (TABLE 17-10).12 The fat content recommended is primarily polyunsaturated fat such as olive oil, while saturated fats such as butter and lard (animal fat) should be limited.
A recent meta-analysis by Choi et al12 showed that in overweight or obese patients with type 2 diabetes, the average A1C reduction was 0.62% (95% CI, –0.89 to –0.35) in the ketogenic intervention group. Another meta-analysis showed an even more significant A1C reduction at 1.07% (95% CI, –1.37 to –0.78).13 Concerns have been raised about the ketogenic diet, particularly as it relates to lipid metabolism and cholesterol levels; however, in the 2 referenced meta-analyses, the total cholesterol and triglyceride levels actually declined in the ketogenic intervention groups with minimal effect on LDL-C.12,13 This may alleviate some of the concerns of lipid management with this diet.
Plant-based diet
Popularized by Dr. T. Colin Campbell, a plant-based diet refers to a low-fat, high-fiber, whole-foods diet (whole fruits, vegetables, and naturally occurring carbohydrates, as opposed to processed foods). Examples of this type of diet include the popular vegan diet, which restricts all animal-derived products, and the vegetarian diet, which is generally limited to foods in the plant category with some addition of animal products, such as milk and cheese. Other variations of these diets exist and include other sources of protein (eg, chicken, eggs, or fish) (TABLE 17-10).
Continue to: A review by...
A review by Salas-Salvadó et al14 showed that a vegan diet yields an average A1C reduction of 0.41% (95% CI, –0.58 to –0.23).Several meta-analyses report similar effects on A1C with vegetarian and vegan eating patterns.6,15,16 The ADA review notes that weight loss was more significant in the vegan group and concluded that this diet should be studied further while controlling for weight loss.6
Mediterranean diet
The Mediterranean diet emphasizes vegetables, whole grains, fruits, lean meats, nuts, and olive oil. The benefits of the Mediterranean diet are well known and, as a result, the diet is recommended by organizations including the American Heart Association as part of a strategy to reduce cardiovascular risk (TABLE 17-10).
Mediterranean diet interventions have generally shown mixed effects on A1C reduction, weight management, and lipid control in type 2 diabetes. 6 The PREDIMED trial is the largest and longest randomized controlled trial to date comparing the Mediterranean diet to a low-fat diet. 17 This trial has reliably shown a reduced risk for type 2 diabetes and a trend to reduced A1C. 17 A reduction in the need for glucose-lowering medications was demonstrated in a subgroup analysis of the intervention group (adjusted hazard ratio = 0.78; 95% CI, 0.62-0.98). 18 Also, the Mediterranean diet has shown a significant reduction in the incidence of cardiovascular disease in patients with type 2 diabetes. 6
Physical activity and exercise
What do current guidelines recommend?
For most adults with type 2 diabetes, current guidelines by the ADA and by the National Institute of Diabetes and Digestive and Kidney Diseases recommend at least 150 minutes of moderate-to-vigorous intensity exercise every week spread out over at least 3 days, with no more than 2 consecutive days without exercise; and resistance training at least 2 other days per week which should balance all major muscle groups (TABLE 219-21). The benefits of exercise for type 2 diabetes have been well reviewed: positive effects on glucose control, insulin sensitivity, cardiovascular disease, lipid profiles, skeletal muscle metabolism, and solid-organ functioning.19,22,23

Grace et al24 showed in a meta-analysis that moderate aerobic exercise reduced A1C by 0.69% (95% CI, –1.09 to –0.3) at 13 weeks, and a Cochrane review showed an average A1C reduction of 0.6% with moderate-intensity exercise.25 Borror et al26 demonstrated in a systematic review that postprandial moderate-intensity aerobic exercise starting 1 hour after meals results in a reduced 24-hour prevalence of hyperglycemia (33.5% reduction vs control). A meta-analysis in China showed an average A1C reduction of 0.68% for patients performing a Tai Chi physical activity intervention.27
Continue to: Consider high-intensity interval training
Consider high-intensity interval training
Multiple randomized controlled trials highlight the benefits of high-intensity interval training (HIIT) (TABLE 219-21) compared with moderate-intensity continuous training (MICT) on improving A1C. A meta-analysis showed a weighted mean difference in A1C of 0.23% (95% CI, –0.43 to –0.02%).28 Also, a patient could spend less time performing HIIT as opposed to MICT to achieve the same benefits. For example, a patient typically performing 30 minutes of MICT may only need to perform 15 minutes of HIIT,a time-saving option for patients.20,22
Interrupt sedentary behavior
Risk for incident type 2 diabetes increases when someone is sedentary for more than 6 to 8 hours daily or watches TV for 3 to 4 hours (relative risk [RR] = 1.12).29 Recommendations for interrupting a sedentary lifestyle include standing from a seated position at least every 30 minutes and engaging in a light activity during the break interval for at least 3 minutes.19 Most studies have reliably shown that interrupting sedentary behavior reduces postprandial and 24-hour average blood glucose levels.19 Interrupted sitting/sedentary behavior has also been shown to reduce resting blood pressure in patients with type 2 diabetes.30
O ther important lifestyle factors
Encourage 7 to 8 hours of sleep
There is a U-shaped association between glycemic control and sleep quantity based on a meta-analysis by Lee et al 31 that showed a 0.23% increase in A1C in patients with insufficient sleep (< 4.5-6 hours/night) and a 0.13% increase in patients with ≥ 8 hours of sleep per night. Patients should be encouraged to obtain 7 to 8 hours of sleep per night to help maximize their diabetes control.
Address stress reduction
Although evidence for stress reduction interventions on glycemic control is mixed, there does seem to be a benefit in diminishing emotional distress in patients with diabetes. A systematic review by Noordali et al32 demonstrated that patients who received mindfulness-based interventions had improvements in stress, anxiety, and depression symptoms which resulted in improved quality of life. These psychological benefits may subsequently lead to positive behavioral changes.
Assist patients with smoking cessation
A large meta-analysis showed that active smoking increases the risk of cardiovascular events in patients with type 2 diabetes (RR = 1.44; 95% CI, 1.34-1.54).33 Former smokers still have an increased risk (RR = 1.09; 95% CI, 1.05-1.13), but it is lower than that of current smokers, so patients should be encouraged to quit smoking.3,33
Continue to: How can I get my patient to change?
H ow can I get my patient to change?
The AACE recommends using motivational interviewing, behavioral therapy consultation, and wearable feedback devices (eg, accelerometers/pedometers) to stimulate behavioral change in patients.3 Motivational interviewing is the principal counseling strategy and is supported by multiple studies showing the benefits of using this technique in a clinical encounter to induce behavioral changes.34 In general, offer receptive patients intensive behavioral interventions and provide them with resources to accomplish their goals.35 For example, a 7-step yearly intensive behavioral counseling intervention over 3 years showed significant improvements in activity of any intensity, reduced sedentary time, and led to favorable metabolic outcomes.36 Wearable devices result in up to a 1 hour increase in physical activity per week for the wearers vs control, although there was no appreciable effect on A1C.37
One systematic review showed a 0.5% reduction in A1C (95% CI, –0.65 to –0.34) by focusing on environmental changes related to the diet, with the most effective intervention being full meal replacement for calorie control (ie, each meal was pre-made and provided to the patients based on macronutrient and caloric goals).38 Additionally, diabetes self-management education includes coping strategies, problem solving, self-advocacy, and health care system navigation, which have been shown to reduce A1C by an average of 0.6%.21 Patient resources are available for further assistance with lifestyle modifications (TABLE 3).

C an your patient achieve remission?
Emerging evidence suggests that patients may achieve remission from type 2 diabetes with intensive lifestyle interventions.39 This is supported by the American College of Lifestyle Medicine.5 Although there is no consensus definition for remission, in general it is reasonable to presume remission if a patient achieves normo-glycemia (A1C < 5.7%) for at least 1 year without any medication therapy.5 These intensive lifestyle interventions would include a mostly plant-based diet with moderate calorie restriction, appropriate and sustained physical activity, adequate sleep, and stress-reduction techniques.5 One study found that 46% of patients in a weight-management program across multiple primary care clinics achieved remission at 12 months.40 A meta-analysis showed that a low-carbohydrate diet induced remission at 6 months in 32% of patients (although the result was not controlled for weight loss as a possible confounding factor and an A1C cutoff of 6.5% was used).11 Thus far, most studies have focused on short-term follow-up intervals, but evidence is emerging that with intensive lifestyle interventions the effects are sustained at the 2-year mark.41
This evidence could reframe our understanding of type 2 diabetes therapy and could change the conversations we have with patients regarding their treatment. Instead of focusing on an A1C goal that is adequate for control of type 2 diabetes, we would instead focus on achieving remission.
CORRESPONDENCE
Stephen McMullan, MD, Mayo Clinic College of Medicine and Science, 4500 San Pablo Road, Jacksonville, FL 32224; mcmullan.stephen@mayo.edu
Type 2 diabetes has been increasing in incidence and prevalence over the past 20 years, with worldwide prevalence estimated at 6.28%.1 The estimated cost of diagnosed diabetes in the United States was $327 billion in 2017; this included direct medical costs and reduced productivity.2 Type 2 diabetes can be prevented in most patients, given that it is a metabolic derangement caused by a complicated interaction between a patient’s genetic predisposition and lifestyle. A consensus statement by the American Academy of Clinical Endocrinologists (AACE) and American College of Endocrinology indicates that the recommended lifestyle modifications for diabetes include medical nutrition therapy with healthy eating patterns, regular physical activity, adequate sleep, behavioral support/counseling, and smoking cessation.3 Evidence shows that adherence to these lifestyle changes alone yields a relative reduction in type 2 diabetes mortality of 57%.4

In the discussion that follows, we review the current guideline recommendations for dietary modifications and physical activity and summarize their effectiveness in the treatment of type 2 diabetes. We also describe practical clinical strategies to promote change in patient behavior, and examine current literature supporting intensive lifestyle changes that, if achieved, may induce disease remission.5
Dietary strategies
Low, or very low, carbohydrate diet
Carbohydrates can affect blood glucose levels in varying degrees depending on their intrinsic properties such as fiber content, sugars, and starches . 6 According to the American Diabetes Association’s (ADA) 2019 consensus report, 6 the carbohydrate quality that generally should be recommended is high in fiber, vitamins, and minerals, and low in added sugars, fats, and sodium (processed carbohydrates) ( TABLE 1 7-10 ). A low-carbohydrate diet (LCD) typically has a carbohydrate content < 130 g/d or < 26% of a 2000 kcal/d diet. 11 A very low–carbohydrate diet (VLCD) is 20-50 g/d or < 10% of the 2000 kcal/day diet. 11

In a meta-analysis by Goldenberg et al11, the LCD was shown to reduce A1C by 0.47% at 6 months (95% CI, –0.6 to –0.34) and by 0.23% at 12 months when compared with control diets. A review of multiple meta-analyses also showed a significant reduction in A1C especially with VLCD patterns; however, the results waned at the 12-month follow-up.5 In addition, confounding factors were seen when comparing adherence between LCD and VLCD, with patients in the latter group having larger problems with adherence, which decreased the benefit seen in the overall group comparison.11
Very low–carbohydrate/high-fat (ketogenic) diet
Ketogenic diets generally follow a VLCD with the carbohydrate portion set at 5% to 10% of total caloric intake (generally < 30 g/d) and the rest of the calories taken up by protein (typically 1 g/kg/d) and fat (TABLE 17-10).12 The fat content recommended is primarily polyunsaturated fat such as olive oil, while saturated fats such as butter and lard (animal fat) should be limited.
A recent meta-analysis by Choi et al12 showed that in overweight or obese patients with type 2 diabetes, the average A1C reduction was 0.62% (95% CI, –0.89 to –0.35) in the ketogenic intervention group. Another meta-analysis showed an even more significant A1C reduction at 1.07% (95% CI, –1.37 to –0.78).13 Concerns have been raised about the ketogenic diet, particularly as it relates to lipid metabolism and cholesterol levels; however, in the 2 referenced meta-analyses, the total cholesterol and triglyceride levels actually declined in the ketogenic intervention groups with minimal effect on LDL-C.12,13 This may alleviate some of the concerns of lipid management with this diet.
Plant-based diet
Popularized by Dr. T. Colin Campbell, a plant-based diet refers to a low-fat, high-fiber, whole-foods diet (whole fruits, vegetables, and naturally occurring carbohydrates, as opposed to processed foods). Examples of this type of diet include the popular vegan diet, which restricts all animal-derived products, and the vegetarian diet, which is generally limited to foods in the plant category with some addition of animal products, such as milk and cheese. Other variations of these diets exist and include other sources of protein (eg, chicken, eggs, or fish) (TABLE 17-10).
Continue to: A review by...
A review by Salas-Salvadó et al14 showed that a vegan diet yields an average A1C reduction of 0.41% (95% CI, –0.58 to –0.23).Several meta-analyses report similar effects on A1C with vegetarian and vegan eating patterns.6,15,16 The ADA review notes that weight loss was more significant in the vegan group and concluded that this diet should be studied further while controlling for weight loss.6
Mediterranean diet
The Mediterranean diet emphasizes vegetables, whole grains, fruits, lean meats, nuts, and olive oil. The benefits of the Mediterranean diet are well known and, as a result, the diet is recommended by organizations including the American Heart Association as part of a strategy to reduce cardiovascular risk (TABLE 17-10).
Mediterranean diet interventions have generally shown mixed effects on A1C reduction, weight management, and lipid control in type 2 diabetes. 6 The PREDIMED trial is the largest and longest randomized controlled trial to date comparing the Mediterranean diet to a low-fat diet. 17 This trial has reliably shown a reduced risk for type 2 diabetes and a trend to reduced A1C. 17 A reduction in the need for glucose-lowering medications was demonstrated in a subgroup analysis of the intervention group (adjusted hazard ratio = 0.78; 95% CI, 0.62-0.98). 18 Also, the Mediterranean diet has shown a significant reduction in the incidence of cardiovascular disease in patients with type 2 diabetes. 6
Physical activity and exercise
What do current guidelines recommend?
For most adults with type 2 diabetes, current guidelines by the ADA and by the National Institute of Diabetes and Digestive and Kidney Diseases recommend at least 150 minutes of moderate-to-vigorous intensity exercise every week spread out over at least 3 days, with no more than 2 consecutive days without exercise; and resistance training at least 2 other days per week which should balance all major muscle groups (TABLE 219-21). The benefits of exercise for type 2 diabetes have been well reviewed: positive effects on glucose control, insulin sensitivity, cardiovascular disease, lipid profiles, skeletal muscle metabolism, and solid-organ functioning.19,22,23

Grace et al24 showed in a meta-analysis that moderate aerobic exercise reduced A1C by 0.69% (95% CI, –1.09 to –0.3) at 13 weeks, and a Cochrane review showed an average A1C reduction of 0.6% with moderate-intensity exercise.25 Borror et al26 demonstrated in a systematic review that postprandial moderate-intensity aerobic exercise starting 1 hour after meals results in a reduced 24-hour prevalence of hyperglycemia (33.5% reduction vs control). A meta-analysis in China showed an average A1C reduction of 0.68% for patients performing a Tai Chi physical activity intervention.27
Continue to: Consider high-intensity interval training
Consider high-intensity interval training
Multiple randomized controlled trials highlight the benefits of high-intensity interval training (HIIT) (TABLE 219-21) compared with moderate-intensity continuous training (MICT) on improving A1C. A meta-analysis showed a weighted mean difference in A1C of 0.23% (95% CI, –0.43 to –0.02%).28 Also, a patient could spend less time performing HIIT as opposed to MICT to achieve the same benefits. For example, a patient typically performing 30 minutes of MICT may only need to perform 15 minutes of HIIT,a time-saving option for patients.20,22
Interrupt sedentary behavior
Risk for incident type 2 diabetes increases when someone is sedentary for more than 6 to 8 hours daily or watches TV for 3 to 4 hours (relative risk [RR] = 1.12).29 Recommendations for interrupting a sedentary lifestyle include standing from a seated position at least every 30 minutes and engaging in a light activity during the break interval for at least 3 minutes.19 Most studies have reliably shown that interrupting sedentary behavior reduces postprandial and 24-hour average blood glucose levels.19 Interrupted sitting/sedentary behavior has also been shown to reduce resting blood pressure in patients with type 2 diabetes.30
O ther important lifestyle factors
Encourage 7 to 8 hours of sleep
There is a U-shaped association between glycemic control and sleep quantity based on a meta-analysis by Lee et al 31 that showed a 0.23% increase in A1C in patients with insufficient sleep (< 4.5-6 hours/night) and a 0.13% increase in patients with ≥ 8 hours of sleep per night. Patients should be encouraged to obtain 7 to 8 hours of sleep per night to help maximize their diabetes control.
Address stress reduction
Although evidence for stress reduction interventions on glycemic control is mixed, there does seem to be a benefit in diminishing emotional distress in patients with diabetes. A systematic review by Noordali et al32 demonstrated that patients who received mindfulness-based interventions had improvements in stress, anxiety, and depression symptoms which resulted in improved quality of life. These psychological benefits may subsequently lead to positive behavioral changes.
Assist patients with smoking cessation
A large meta-analysis showed that active smoking increases the risk of cardiovascular events in patients with type 2 diabetes (RR = 1.44; 95% CI, 1.34-1.54).33 Former smokers still have an increased risk (RR = 1.09; 95% CI, 1.05-1.13), but it is lower than that of current smokers, so patients should be encouraged to quit smoking.3,33
Continue to: How can I get my patient to change?
H ow can I get my patient to change?
The AACE recommends using motivational interviewing, behavioral therapy consultation, and wearable feedback devices (eg, accelerometers/pedometers) to stimulate behavioral change in patients.3 Motivational interviewing is the principal counseling strategy and is supported by multiple studies showing the benefits of using this technique in a clinical encounter to induce behavioral changes.34 In general, offer receptive patients intensive behavioral interventions and provide them with resources to accomplish their goals.35 For example, a 7-step yearly intensive behavioral counseling intervention over 3 years showed significant improvements in activity of any intensity, reduced sedentary time, and led to favorable metabolic outcomes.36 Wearable devices result in up to a 1 hour increase in physical activity per week for the wearers vs control, although there was no appreciable effect on A1C.37
One systematic review showed a 0.5% reduction in A1C (95% CI, –0.65 to –0.34) by focusing on environmental changes related to the diet, with the most effective intervention being full meal replacement for calorie control (ie, each meal was pre-made and provided to the patients based on macronutrient and caloric goals).38 Additionally, diabetes self-management education includes coping strategies, problem solving, self-advocacy, and health care system navigation, which have been shown to reduce A1C by an average of 0.6%.21 Patient resources are available for further assistance with lifestyle modifications (TABLE 3).

C an your patient achieve remission?
Emerging evidence suggests that patients may achieve remission from type 2 diabetes with intensive lifestyle interventions.39 This is supported by the American College of Lifestyle Medicine.5 Although there is no consensus definition for remission, in general it is reasonable to presume remission if a patient achieves normo-glycemia (A1C < 5.7%) for at least 1 year without any medication therapy.5 These intensive lifestyle interventions would include a mostly plant-based diet with moderate calorie restriction, appropriate and sustained physical activity, adequate sleep, and stress-reduction techniques.5 One study found that 46% of patients in a weight-management program across multiple primary care clinics achieved remission at 12 months.40 A meta-analysis showed that a low-carbohydrate diet induced remission at 6 months in 32% of patients (although the result was not controlled for weight loss as a possible confounding factor and an A1C cutoff of 6.5% was used).11 Thus far, most studies have focused on short-term follow-up intervals, but evidence is emerging that with intensive lifestyle interventions the effects are sustained at the 2-year mark.41
This evidence could reframe our understanding of type 2 diabetes therapy and could change the conversations we have with patients regarding their treatment. Instead of focusing on an A1C goal that is adequate for control of type 2 diabetes, we would instead focus on achieving remission.
CORRESPONDENCE
Stephen McMullan, MD, Mayo Clinic College of Medicine and Science, 4500 San Pablo Road, Jacksonville, FL 32224; mcmullan.stephen@mayo.edu
1. Kahn MAB, Hashim MJ, King JK, et al. Epidemiology of type 2 diabetes – global burden of disease and forecasted trends. J Epidemiol Glob Health. 2020;10:107-111. doi: 10.2991/jegh.k.191028.001
2. American Diabetes Association. Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41:917-928. doi:10.2337/dci18-0007
3. Garber AJ, Handelsman Y, Grunberger G, et al. Consensus Statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the Comprehensive Type 2 Diabetes Management Algorithm – 2020 Executive Summary. Endocr Pract. 2020;26:107-139. doi:10.4158/CS-2019-0472
4. Schlesinger S, Neuenschwander M, Ballon A, et al. Adherence to healthy lifestyles and incidence of diabetes and mortality among individuals with diabetes: a systematic review and meta-analysis of prospective studies. J Epidemiol Community Health. 2020;74:481-487. doi: 10.1136/jech-2019-213415
5. Kelly J, Karlsen M, Steinke G. Type 2 Diabetes Remission and Lifestyle Medicine: A Position Statement from the American College of Lifestyle Medicine. Am J Lifestyle Med. 2020;14:406-419. doi: 10.1177/1559827620930962
6. Evert AB, Dennison M, Gardner CD, et al. Nutrition Therapy for Adults with Diabetes or Prediabetes: A Consensus Report. Diabetes Care. 2019;42:731-754. doi: 10.2337/dci19-0014
7. Mayo Clinic. Low-carb diet: Can it help you lose weight? Accessed August 22, 2022. www.mayoclinic.org/healthylifestyle/weight-loss/in-depth/low-carb-diet/art-20045831
8. Mayo Clinic. Is the keto diet for You? A Mayo expert weighs in. Accessed September 16, 2022. www.mayoclinic.org/is-the-keto-diet-for-you-a-mayo-expert-weighs-in/art-20457595
9. Mayo Clinic. Vegetarian diet: How to get the best nutrition. Accessed August 22, 2022. www.mayoclinic.org/healthy-lifestyle/nutrition-and-healthy-eating/in-depth/vegetarian-diet/art-20046446
10. AHA. What is the Mediterranean diet? Accessed September 16, 2022. www.heart.org/en/healthy-living/healthy-eating/eat-smart/nutrition-basics/mediterranean-diet
11. Goldenberg JZ, Day A, Brinkworth GD, et al. Efficacy and safety of low and very low carbohydrate diets for type 2 diabetes remission: systematic review and meta-analysis of published and unpublished randomized trial data. BMJ. 2021;372:m4743. doi: 10.1136/bmj.m4743
12. Choi YJ, Jeon SM, Shin S. Impact of a ketogenic diet on metabolic parameters in patients with obesity or overweight and with or without type 2 diabetes: a meta-analysis of randomized controlled trials. Nutrients. 2020;12:2005. doi: 10.3390/nu12072005
13. Yuan X, Wang J, Yang S, et al. Effect of the ketogenic diet on glycemic control, insulin resistance, and lipid metabolism in patients with T2DM: a systematic review and meta-analysis. Nutr Diabetes. 2020;10:38. doi: 10.1038/s41387-020-00142-z
14. Salas-Salvadó J, Becerra-Tomás N, Papandreou C, et al. Dietary patterns emphasizing the consumption of plant foods in the management of type 2 diabetes: a narrative review. Adv Nutr. 2019;10(suppl_4):S320-S331. doi: 10.1093/advances/nmy102
15. Viguiliouk E, Kendall CW, Kahleová H, et al. Effect of vegetarian dietary patterns on cardiometabolic risk factors in diabetes: a systematic review and meta-analysis of randomized controlled trials. Clin Nutr. 2018;38:1133-1145. doi: 10.1016/j.clnu.2018.05.032
16. Yokoyama Y, Barnard ND, Levin SM, et al. Vegetarian diets and glycemic control in diabetes: a systematic review and meta-analysis. Cardiovasc Diagn Ther. 2014;4:373-382. doi: 10.3978/j.issn.2223-3652.2014.10.04
17. Estruch R, Ros E, Salas-Salvadó J, et al. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. N Engl J Med. 2018;378:e34. doi: 10.1056/NEJMoa1800389
18. Basterra-Gortari FJ, Ruiz-Canela M, Martínez-González MA, et al. Effects of a Mediterranean eating plan on the need for glucose-lowering medications in participants with type 2 diabetes: a subgroup analysis of the PREDIMED trial. Diabetes Care. 2019;42:1390-1397. doi: 10.2337/dc18-2475
19. Colberg SR, Sigal RJ, Yardley JE, et al. Physical Activity/Exercise and Diabetes: A position Statement of the American Diabetes Association. Diabetes Care. 2016;39:2065-2079. doi:10.2337/dc16-1728
20. Hwang CL, Lim J, Yoo JK, et al. Effect of all-extremity high-intensity interval training vs. moderate-intensity continuous training on aerobic fitness in middle-aged and older adults with type 2 diabetes: a randomized controlled trial. Exp Gerontol. 2019;116:46-53. doi:10.1016/j.exger.2018.12.013
21. Zangeneh F, Boltri J, Dallas A, et al. National Institute of Diabetes and Digestive and Kidney Diseases. Guiding principles for the care of people with or at risk for diabetes. Accessed September 16, 2022. www.niddk.nih.gov/health-information/professionals/clinical-tools-patient-management/diabetes/guiding-principles-care-people-risk-diabetes
22. Kirwan JP, Sacks J, Nieuwoudt S. The essential role of exercise in the management of type 2 diabetes. Cleve Clin J Med. 2017;84(7 suppl 1):S15-S21. doi: 10.3949/ccjm.84.s1.03
23. Zanuso S, Sacchetti M, Sundberg CJ, et al. Exercise in type 2 diabetes: genetic, metabolic and neuromuscular adaptations. a review of the evidence. Br J Sports Med. 2017;51:1533-1538. doi: 10.1136/bjsports-2016-096724
24. Grace A, Chan E, Giallauria F, et al. Clinical outcomes and glycaemic responses to different aerobic exercise training intensities in type II diabetes: a systematic review and meta-analysis. Cardiovasc Diabetol. 2017;16:37. Published 2017 Mar 14. doi: 10.1186/s12933-017-0518-6
25. Thomas DE, Elliott EJ, Naughton GA. Exercise for type 2 diabetes mellitus. Cochrane Database Syst Rev. 2006;(3):CD002968. doi: 10.1002/14651858.CD002968.pub2
26. Borror A, Zieff G, Battaglini C, et al. The effects of postprandial exercise on glucose control in individuals with type 2 diabetes: a systematic review. Sports Med. 2018;48:1479-1491. doi: 10.1007/s40279-018-0864-x
27. Xia TW, Yang Y, Li WH, et al. Different training durations and styles of tai chi for glucose control in patients with type 2 diabetes: a systematic review and meta-analysis of controlled trials. BMC Complement Altern Med. 2019;19:63. doi: 10.1186/s12906-019-2475-y
28. Liubaoerjijin Y, Terada T, Fletcher K, et al. Effect of aerobic exercise intensity on glycemic control in type 2 diabetes: a meta-analysis of head-to-head randomized trials. Acta Diabetol. 2016;53:769-781. doi: 10.1007/s00592-016-0870-0
29. Patterson R, McNamara E, Tainio M, et al. Sedentary behaviour and risk of all-cause, cardiovascular and cancer mortality, and incident type 2 diabetes: a systematic review and dose response meta-analysis. Eur J Epidemiol. 2018;33:811-829. doi: 10.1007/s10654-018-0380-1
30. Dempsey PC, Sacre JW, Larsen RN, et al. Interrupting prolonged sitting with brief bouts of light walking or simple resistance activities reduces resting blood pressure and plasma noradrenaline in type 2 diabetes. J Hypertens. 2016;34:2376-2382. doi: 10.1097/HJH.0000000000001101
31. Lee SWH, Ng KY, Chin WK. The impact of sleep amount and sleep quality on glycemic control in type 2 diabetes: a systematic review and meta-analysis. Sleep Med Rev. 2017;31:91-101. doi: 10.1016/j.smrv.2016.02.001.
32. Noordali F, Cumming J, Thompson JL. Effectiveness of mindfulness-based intervention on physiological and psychological complications in adults with diabetes: a systematic review. J Health Psychol. 2017;22:965-983. doi: 10.1177/1359105315620293
33. Pan A, Wang Y, Talaei M, et al. Relation of smoking with total mortality and cardiovascular events among patients with diabetes mellitus: a meta-analysis and systematic review. Circulation. 2015;132:1795-1804. doi:10.116/circulationaha.115.017926
34. VanBuskirk KA, Wetherell JL. Motivational interviewing with primary care populations: a systematic review and meta-analysis. J Behav Med. 2014;37:768-780. doi:10.1007/s10865-013-9527-4
35. Koenigsberg MR, Corliss J. Diabetes self-management: facilitating lifestyle change. Am Fam Physician. 2017;96:362-370.
36. Balducci S, D’Errico V, Haxhi J, et al. Effect of a behavioral intervention strategy for adoption and maintenance of a physically active lifestyle: the Italian Diabetes and Exercise Study 2 (IDES_2): a randomized controlled trial. Diabetes Care. 2017;40:1444-1452. doi: 10.2337/dc17-0594
37. Baskerville R, Ricci-Cabello I, Roberts N, et al. Impact of accelerometer and pedometer use on physical activity and glycaemic control in people with type 2 diabetes: a systematic review and meta-analysis. Diabet Med. 2017;34:612-620. doi:10.1111/dme.13331
38. Cradock KA, ÓLaighin G, Finucane FM, et al. Diet behavior change techniques in type 2 diabetes: a systematic review and meta-analysis. Diabetes Care. 2017;40:1800-1810. doi: 10.2337/dc17-0462
39. Hallberg SJ, Gershuni VM, Hazbun TL, et al. Reversing type 2 diabetes: a narrative review of the evidence. Nutrients. 2019;11:766. doi: 10.3390/nu11040766
40. Lean MEJ, Leslie WS, Barnes AC, et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): an open-label, cluster-randomised trial. Lancet. 2018;391:541-551. doi: 10.1016/S0140-6736(17)33102-1
41. Sbroma Tomaro E, Pippi R, Reginato E, et al. Intensive lifestyle intervention is particularly advantageous in poorly controlled type 2 diabetes. Nutr Metab Cardiovasc Dis. 2017;27:688-694. doi:10.1016/j.numecd.2017.06.009
1. Kahn MAB, Hashim MJ, King JK, et al. Epidemiology of type 2 diabetes – global burden of disease and forecasted trends. J Epidemiol Glob Health. 2020;10:107-111. doi: 10.2991/jegh.k.191028.001
2. American Diabetes Association. Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41:917-928. doi:10.2337/dci18-0007
3. Garber AJ, Handelsman Y, Grunberger G, et al. Consensus Statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the Comprehensive Type 2 Diabetes Management Algorithm – 2020 Executive Summary. Endocr Pract. 2020;26:107-139. doi:10.4158/CS-2019-0472
4. Schlesinger S, Neuenschwander M, Ballon A, et al. Adherence to healthy lifestyles and incidence of diabetes and mortality among individuals with diabetes: a systematic review and meta-analysis of prospective studies. J Epidemiol Community Health. 2020;74:481-487. doi: 10.1136/jech-2019-213415
5. Kelly J, Karlsen M, Steinke G. Type 2 Diabetes Remission and Lifestyle Medicine: A Position Statement from the American College of Lifestyle Medicine. Am J Lifestyle Med. 2020;14:406-419. doi: 10.1177/1559827620930962
6. Evert AB, Dennison M, Gardner CD, et al. Nutrition Therapy for Adults with Diabetes or Prediabetes: A Consensus Report. Diabetes Care. 2019;42:731-754. doi: 10.2337/dci19-0014
7. Mayo Clinic. Low-carb diet: Can it help you lose weight? Accessed August 22, 2022. www.mayoclinic.org/healthylifestyle/weight-loss/in-depth/low-carb-diet/art-20045831
8. Mayo Clinic. Is the keto diet for You? A Mayo expert weighs in. Accessed September 16, 2022. www.mayoclinic.org/is-the-keto-diet-for-you-a-mayo-expert-weighs-in/art-20457595
9. Mayo Clinic. Vegetarian diet: How to get the best nutrition. Accessed August 22, 2022. www.mayoclinic.org/healthy-lifestyle/nutrition-and-healthy-eating/in-depth/vegetarian-diet/art-20046446
10. AHA. What is the Mediterranean diet? Accessed September 16, 2022. www.heart.org/en/healthy-living/healthy-eating/eat-smart/nutrition-basics/mediterranean-diet
11. Goldenberg JZ, Day A, Brinkworth GD, et al. Efficacy and safety of low and very low carbohydrate diets for type 2 diabetes remission: systematic review and meta-analysis of published and unpublished randomized trial data. BMJ. 2021;372:m4743. doi: 10.1136/bmj.m4743
12. Choi YJ, Jeon SM, Shin S. Impact of a ketogenic diet on metabolic parameters in patients with obesity or overweight and with or without type 2 diabetes: a meta-analysis of randomized controlled trials. Nutrients. 2020;12:2005. doi: 10.3390/nu12072005
13. Yuan X, Wang J, Yang S, et al. Effect of the ketogenic diet on glycemic control, insulin resistance, and lipid metabolism in patients with T2DM: a systematic review and meta-analysis. Nutr Diabetes. 2020;10:38. doi: 10.1038/s41387-020-00142-z
14. Salas-Salvadó J, Becerra-Tomás N, Papandreou C, et al. Dietary patterns emphasizing the consumption of plant foods in the management of type 2 diabetes: a narrative review. Adv Nutr. 2019;10(suppl_4):S320-S331. doi: 10.1093/advances/nmy102
15. Viguiliouk E, Kendall CW, Kahleová H, et al. Effect of vegetarian dietary patterns on cardiometabolic risk factors in diabetes: a systematic review and meta-analysis of randomized controlled trials. Clin Nutr. 2018;38:1133-1145. doi: 10.1016/j.clnu.2018.05.032
16. Yokoyama Y, Barnard ND, Levin SM, et al. Vegetarian diets and glycemic control in diabetes: a systematic review and meta-analysis. Cardiovasc Diagn Ther. 2014;4:373-382. doi: 10.3978/j.issn.2223-3652.2014.10.04
17. Estruch R, Ros E, Salas-Salvadó J, et al. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. N Engl J Med. 2018;378:e34. doi: 10.1056/NEJMoa1800389
18. Basterra-Gortari FJ, Ruiz-Canela M, Martínez-González MA, et al. Effects of a Mediterranean eating plan on the need for glucose-lowering medications in participants with type 2 diabetes: a subgroup analysis of the PREDIMED trial. Diabetes Care. 2019;42:1390-1397. doi: 10.2337/dc18-2475
19. Colberg SR, Sigal RJ, Yardley JE, et al. Physical Activity/Exercise and Diabetes: A position Statement of the American Diabetes Association. Diabetes Care. 2016;39:2065-2079. doi:10.2337/dc16-1728
20. Hwang CL, Lim J, Yoo JK, et al. Effect of all-extremity high-intensity interval training vs. moderate-intensity continuous training on aerobic fitness in middle-aged and older adults with type 2 diabetes: a randomized controlled trial. Exp Gerontol. 2019;116:46-53. doi:10.1016/j.exger.2018.12.013
21. Zangeneh F, Boltri J, Dallas A, et al. National Institute of Diabetes and Digestive and Kidney Diseases. Guiding principles for the care of people with or at risk for diabetes. Accessed September 16, 2022. www.niddk.nih.gov/health-information/professionals/clinical-tools-patient-management/diabetes/guiding-principles-care-people-risk-diabetes
22. Kirwan JP, Sacks J, Nieuwoudt S. The essential role of exercise in the management of type 2 diabetes. Cleve Clin J Med. 2017;84(7 suppl 1):S15-S21. doi: 10.3949/ccjm.84.s1.03
23. Zanuso S, Sacchetti M, Sundberg CJ, et al. Exercise in type 2 diabetes: genetic, metabolic and neuromuscular adaptations. a review of the evidence. Br J Sports Med. 2017;51:1533-1538. doi: 10.1136/bjsports-2016-096724
24. Grace A, Chan E, Giallauria F, et al. Clinical outcomes and glycaemic responses to different aerobic exercise training intensities in type II diabetes: a systematic review and meta-analysis. Cardiovasc Diabetol. 2017;16:37. Published 2017 Mar 14. doi: 10.1186/s12933-017-0518-6
25. Thomas DE, Elliott EJ, Naughton GA. Exercise for type 2 diabetes mellitus. Cochrane Database Syst Rev. 2006;(3):CD002968. doi: 10.1002/14651858.CD002968.pub2
26. Borror A, Zieff G, Battaglini C, et al. The effects of postprandial exercise on glucose control in individuals with type 2 diabetes: a systematic review. Sports Med. 2018;48:1479-1491. doi: 10.1007/s40279-018-0864-x
27. Xia TW, Yang Y, Li WH, et al. Different training durations and styles of tai chi for glucose control in patients with type 2 diabetes: a systematic review and meta-analysis of controlled trials. BMC Complement Altern Med. 2019;19:63. doi: 10.1186/s12906-019-2475-y
28. Liubaoerjijin Y, Terada T, Fletcher K, et al. Effect of aerobic exercise intensity on glycemic control in type 2 diabetes: a meta-analysis of head-to-head randomized trials. Acta Diabetol. 2016;53:769-781. doi: 10.1007/s00592-016-0870-0
29. Patterson R, McNamara E, Tainio M, et al. Sedentary behaviour and risk of all-cause, cardiovascular and cancer mortality, and incident type 2 diabetes: a systematic review and dose response meta-analysis. Eur J Epidemiol. 2018;33:811-829. doi: 10.1007/s10654-018-0380-1
30. Dempsey PC, Sacre JW, Larsen RN, et al. Interrupting prolonged sitting with brief bouts of light walking or simple resistance activities reduces resting blood pressure and plasma noradrenaline in type 2 diabetes. J Hypertens. 2016;34:2376-2382. doi: 10.1097/HJH.0000000000001101
31. Lee SWH, Ng KY, Chin WK. The impact of sleep amount and sleep quality on glycemic control in type 2 diabetes: a systematic review and meta-analysis. Sleep Med Rev. 2017;31:91-101. doi: 10.1016/j.smrv.2016.02.001.
32. Noordali F, Cumming J, Thompson JL. Effectiveness of mindfulness-based intervention on physiological and psychological complications in adults with diabetes: a systematic review. J Health Psychol. 2017;22:965-983. doi: 10.1177/1359105315620293
33. Pan A, Wang Y, Talaei M, et al. Relation of smoking with total mortality and cardiovascular events among patients with diabetes mellitus: a meta-analysis and systematic review. Circulation. 2015;132:1795-1804. doi:10.116/circulationaha.115.017926
34. VanBuskirk KA, Wetherell JL. Motivational interviewing with primary care populations: a systematic review and meta-analysis. J Behav Med. 2014;37:768-780. doi:10.1007/s10865-013-9527-4
35. Koenigsberg MR, Corliss J. Diabetes self-management: facilitating lifestyle change. Am Fam Physician. 2017;96:362-370.
36. Balducci S, D’Errico V, Haxhi J, et al. Effect of a behavioral intervention strategy for adoption and maintenance of a physically active lifestyle: the Italian Diabetes and Exercise Study 2 (IDES_2): a randomized controlled trial. Diabetes Care. 2017;40:1444-1452. doi: 10.2337/dc17-0594
37. Baskerville R, Ricci-Cabello I, Roberts N, et al. Impact of accelerometer and pedometer use on physical activity and glycaemic control in people with type 2 diabetes: a systematic review and meta-analysis. Diabet Med. 2017;34:612-620. doi:10.1111/dme.13331
38. Cradock KA, ÓLaighin G, Finucane FM, et al. Diet behavior change techniques in type 2 diabetes: a systematic review and meta-analysis. Diabetes Care. 2017;40:1800-1810. doi: 10.2337/dc17-0462
39. Hallberg SJ, Gershuni VM, Hazbun TL, et al. Reversing type 2 diabetes: a narrative review of the evidence. Nutrients. 2019;11:766. doi: 10.3390/nu11040766
40. Lean MEJ, Leslie WS, Barnes AC, et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): an open-label, cluster-randomised trial. Lancet. 2018;391:541-551. doi: 10.1016/S0140-6736(17)33102-1
41. Sbroma Tomaro E, Pippi R, Reginato E, et al. Intensive lifestyle intervention is particularly advantageous in poorly controlled type 2 diabetes. Nutr Metab Cardiovasc Dis. 2017;27:688-694. doi:10.1016/j.numecd.2017.06.009
PRACTICE RECOMMENDATIONS
› Recommend a reduced-calorie diet that is generally plant based and low in carbohydrates as part of the treatment plan for type 2 diabetes. B
› Counsel all patients with type 2 diabetes to engage in physical activity for at least 150 minutes per week at moderate intensity and to add resistance training on at least 2 days to improve glycemic control. B
› Teach patients techniques to reduce stress and improve sleep quality. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Dapagliflozin DELIVERs regardless of systolic pressure in HFpEF
Whatever the mechanism of benefit from dapagliflozin (Farxiga) in patients with heart failure (HF) – and potentially also other sodium-glucose cotransporter 2 (SGLT2) inhibitors – its blood pressure lowering effects aren’t likely to contribute much.
Indeed, at least in patients with HF and non-reduced ejection fractions, dapagliflozin has only a modest BP-lowering effect and cuts cardiovascular (CV) risk regardless of baseline pressure or change in systolic BP, suggests a secondary analysis from the large placebo-controlled DELIVER trial.
Systolic BP fell over 1 month by just under 2 mmHg, on average, in trial patients with either mildly reduced or preserved ejection fraction (HFmrEF or HFpEF, respectively) assigned to take dapagliflozin versus placebo.
The effect was achieved without increasing the risk for adverse events from dapagliflozin, even among patients with the lowest baseline systolic pressures. Adverse outcomes overall, however, were more common at the lowest systolic BP level than at higher pressures, researchers reported.
They say the findings should help alleviate long-standing concerns that initiating SGLT2 inhibitors, with their recognized diuretic effects, might present a hazard in patients with HF and low systolic BP.
“It is a consistent theme in heart failure trials that the blood pressure–lowering effect of SGLT2 inhibitors is more modest than it is in non–heart-failure populations,” Senthil Selvaraj, MD, Duke University, Durham, N.C., told this news organization.
Changes to antihypertensive drug therapy throughout the trial, which presumably enhanced BP responses and “might occur more frequently in the placebo group,” Dr. Selvaraj said, “might explain why the blood pressure effect is a little bit more modest in this population.”
Dr. Selvaraj presented the analysis at the Annual Scientific Meeting of the Heart Failure Society of America, held in National Harbor, Md., and is lead author on its same-day publication in JACC: Heart Failure.
The findings “reinforce the clinical benefits of SGLT2 inhibitors in patients with heart failure across the full spectrum of ejection fractions and large range of systolic blood pressures,” said Gregg C. Fonarow, MD, University of California, Los Angeles Medical Center, who was not part of the DELIVER analysis.
The study’s greater adjusted risks for CV and all-cause mortality risks at the lowest baseline systolic pressures “parallels a series of observational analyses from registries, including OPTIMIZE-HF,” Dr. Fonarow observed.
In those prior studies of patients with established HFpEF, “systolic BP less than 120 mmHg or even 130 mmHg was associated with worse outcomes than those with higher systolic BP.”
The current findings, therefore, “highlight how optimal blood pressure targets in patients with established heart failure have not been well established,” Dr. Fonarow said.
The analysis included all 6,263 participants in DELIVER, outpatients or patients hospitalized for worsening HF who were in NYHA class 2-4 with a left ventricular ejection fraction (LVEF) greater than 40%. They averaged 72 in age, and 44% were women. Their mean baseline systolic BP was 128 mmHg.
After 1 month, mean systolic BP had fallen by 1.8 mmHg (P < .001) in patients who had been randomly assigned to dapagliflozin versus placebo. The effect was consistent (interaction P = .16) across all systolic BP categories (less than 120 mmHg, 120-129 mmHg, 130-139 mmHg, and 140 mmHg or higher).
The effect was similarly independent of estimated glomerular filtration rate (eGFR) and LVEF (interaction P = .30 and P = .33, respectively), Dr. Selvaraj reported.
In an analysis adjusted for both baseline and 1-month change in systolic BP, the effect of dapagliflozin on the primary endpoint was “minimally attenuated,” compared with the primary analysis, he said. That suggests the clinical benefits “did not significantly relate to the blood pressure–lowering effect” of the SGLT2 inhibitor.
In that analysis, the hazard ratio for CV death or worsening HF for dapagliflozin versus placebo was 0.85 (95% confidence interval, 0.75-0.96; P = .010). The HR had been 0.82 (95% CI, 0.73-0.92; P < .001) overall in the DELIVER primary analysis.
The current study doesn’t shed further light on the main SGLT2 inhibitor mechanism of clinical benefit in nondiabetics with HF, which remains a mystery.
“There is a diuretic effect, but it’s not incredibly robust,” Dr. Selvaraj observed. It may contribute to the drugs’ benefits, “but it’s definitely more than that – a lot more than that.”
DELIVER was funded by AstraZeneca. Dr. Selvaraj reported no relevant conflicts. Disclosures for the other authors are in the report. Dr. Fonarow has reported receiving personal fees from Abbott, Amgen, AstraZeneca, Bayer, Cytokinetics, Edwards, Janssen, Medtronic, Merck, and Novartis.
A version of this article first appeared on Medscape.com.
Whatever the mechanism of benefit from dapagliflozin (Farxiga) in patients with heart failure (HF) – and potentially also other sodium-glucose cotransporter 2 (SGLT2) inhibitors – its blood pressure lowering effects aren’t likely to contribute much.
Indeed, at least in patients with HF and non-reduced ejection fractions, dapagliflozin has only a modest BP-lowering effect and cuts cardiovascular (CV) risk regardless of baseline pressure or change in systolic BP, suggests a secondary analysis from the large placebo-controlled DELIVER trial.
Systolic BP fell over 1 month by just under 2 mmHg, on average, in trial patients with either mildly reduced or preserved ejection fraction (HFmrEF or HFpEF, respectively) assigned to take dapagliflozin versus placebo.
The effect was achieved without increasing the risk for adverse events from dapagliflozin, even among patients with the lowest baseline systolic pressures. Adverse outcomes overall, however, were more common at the lowest systolic BP level than at higher pressures, researchers reported.
They say the findings should help alleviate long-standing concerns that initiating SGLT2 inhibitors, with their recognized diuretic effects, might present a hazard in patients with HF and low systolic BP.
“It is a consistent theme in heart failure trials that the blood pressure–lowering effect of SGLT2 inhibitors is more modest than it is in non–heart-failure populations,” Senthil Selvaraj, MD, Duke University, Durham, N.C., told this news organization.
Changes to antihypertensive drug therapy throughout the trial, which presumably enhanced BP responses and “might occur more frequently in the placebo group,” Dr. Selvaraj said, “might explain why the blood pressure effect is a little bit more modest in this population.”
Dr. Selvaraj presented the analysis at the Annual Scientific Meeting of the Heart Failure Society of America, held in National Harbor, Md., and is lead author on its same-day publication in JACC: Heart Failure.
The findings “reinforce the clinical benefits of SGLT2 inhibitors in patients with heart failure across the full spectrum of ejection fractions and large range of systolic blood pressures,” said Gregg C. Fonarow, MD, University of California, Los Angeles Medical Center, who was not part of the DELIVER analysis.
The study’s greater adjusted risks for CV and all-cause mortality risks at the lowest baseline systolic pressures “parallels a series of observational analyses from registries, including OPTIMIZE-HF,” Dr. Fonarow observed.
In those prior studies of patients with established HFpEF, “systolic BP less than 120 mmHg or even 130 mmHg was associated with worse outcomes than those with higher systolic BP.”
The current findings, therefore, “highlight how optimal blood pressure targets in patients with established heart failure have not been well established,” Dr. Fonarow said.
The analysis included all 6,263 participants in DELIVER, outpatients or patients hospitalized for worsening HF who were in NYHA class 2-4 with a left ventricular ejection fraction (LVEF) greater than 40%. They averaged 72 in age, and 44% were women. Their mean baseline systolic BP was 128 mmHg.
After 1 month, mean systolic BP had fallen by 1.8 mmHg (P < .001) in patients who had been randomly assigned to dapagliflozin versus placebo. The effect was consistent (interaction P = .16) across all systolic BP categories (less than 120 mmHg, 120-129 mmHg, 130-139 mmHg, and 140 mmHg or higher).
The effect was similarly independent of estimated glomerular filtration rate (eGFR) and LVEF (interaction P = .30 and P = .33, respectively), Dr. Selvaraj reported.
In an analysis adjusted for both baseline and 1-month change in systolic BP, the effect of dapagliflozin on the primary endpoint was “minimally attenuated,” compared with the primary analysis, he said. That suggests the clinical benefits “did not significantly relate to the blood pressure–lowering effect” of the SGLT2 inhibitor.
In that analysis, the hazard ratio for CV death or worsening HF for dapagliflozin versus placebo was 0.85 (95% confidence interval, 0.75-0.96; P = .010). The HR had been 0.82 (95% CI, 0.73-0.92; P < .001) overall in the DELIVER primary analysis.
The current study doesn’t shed further light on the main SGLT2 inhibitor mechanism of clinical benefit in nondiabetics with HF, which remains a mystery.
“There is a diuretic effect, but it’s not incredibly robust,” Dr. Selvaraj observed. It may contribute to the drugs’ benefits, “but it’s definitely more than that – a lot more than that.”
DELIVER was funded by AstraZeneca. Dr. Selvaraj reported no relevant conflicts. Disclosures for the other authors are in the report. Dr. Fonarow has reported receiving personal fees from Abbott, Amgen, AstraZeneca, Bayer, Cytokinetics, Edwards, Janssen, Medtronic, Merck, and Novartis.
A version of this article first appeared on Medscape.com.
Whatever the mechanism of benefit from dapagliflozin (Farxiga) in patients with heart failure (HF) – and potentially also other sodium-glucose cotransporter 2 (SGLT2) inhibitors – its blood pressure lowering effects aren’t likely to contribute much.
Indeed, at least in patients with HF and non-reduced ejection fractions, dapagliflozin has only a modest BP-lowering effect and cuts cardiovascular (CV) risk regardless of baseline pressure or change in systolic BP, suggests a secondary analysis from the large placebo-controlled DELIVER trial.
Systolic BP fell over 1 month by just under 2 mmHg, on average, in trial patients with either mildly reduced or preserved ejection fraction (HFmrEF or HFpEF, respectively) assigned to take dapagliflozin versus placebo.
The effect was achieved without increasing the risk for adverse events from dapagliflozin, even among patients with the lowest baseline systolic pressures. Adverse outcomes overall, however, were more common at the lowest systolic BP level than at higher pressures, researchers reported.
They say the findings should help alleviate long-standing concerns that initiating SGLT2 inhibitors, with their recognized diuretic effects, might present a hazard in patients with HF and low systolic BP.
“It is a consistent theme in heart failure trials that the blood pressure–lowering effect of SGLT2 inhibitors is more modest than it is in non–heart-failure populations,” Senthil Selvaraj, MD, Duke University, Durham, N.C., told this news organization.
Changes to antihypertensive drug therapy throughout the trial, which presumably enhanced BP responses and “might occur more frequently in the placebo group,” Dr. Selvaraj said, “might explain why the blood pressure effect is a little bit more modest in this population.”
Dr. Selvaraj presented the analysis at the Annual Scientific Meeting of the Heart Failure Society of America, held in National Harbor, Md., and is lead author on its same-day publication in JACC: Heart Failure.
The findings “reinforce the clinical benefits of SGLT2 inhibitors in patients with heart failure across the full spectrum of ejection fractions and large range of systolic blood pressures,” said Gregg C. Fonarow, MD, University of California, Los Angeles Medical Center, who was not part of the DELIVER analysis.
The study’s greater adjusted risks for CV and all-cause mortality risks at the lowest baseline systolic pressures “parallels a series of observational analyses from registries, including OPTIMIZE-HF,” Dr. Fonarow observed.
In those prior studies of patients with established HFpEF, “systolic BP less than 120 mmHg or even 130 mmHg was associated with worse outcomes than those with higher systolic BP.”
The current findings, therefore, “highlight how optimal blood pressure targets in patients with established heart failure have not been well established,” Dr. Fonarow said.
The analysis included all 6,263 participants in DELIVER, outpatients or patients hospitalized for worsening HF who were in NYHA class 2-4 with a left ventricular ejection fraction (LVEF) greater than 40%. They averaged 72 in age, and 44% were women. Their mean baseline systolic BP was 128 mmHg.
After 1 month, mean systolic BP had fallen by 1.8 mmHg (P < .001) in patients who had been randomly assigned to dapagliflozin versus placebo. The effect was consistent (interaction P = .16) across all systolic BP categories (less than 120 mmHg, 120-129 mmHg, 130-139 mmHg, and 140 mmHg or higher).
The effect was similarly independent of estimated glomerular filtration rate (eGFR) and LVEF (interaction P = .30 and P = .33, respectively), Dr. Selvaraj reported.
In an analysis adjusted for both baseline and 1-month change in systolic BP, the effect of dapagliflozin on the primary endpoint was “minimally attenuated,” compared with the primary analysis, he said. That suggests the clinical benefits “did not significantly relate to the blood pressure–lowering effect” of the SGLT2 inhibitor.
In that analysis, the hazard ratio for CV death or worsening HF for dapagliflozin versus placebo was 0.85 (95% confidence interval, 0.75-0.96; P = .010). The HR had been 0.82 (95% CI, 0.73-0.92; P < .001) overall in the DELIVER primary analysis.
The current study doesn’t shed further light on the main SGLT2 inhibitor mechanism of clinical benefit in nondiabetics with HF, which remains a mystery.
“There is a diuretic effect, but it’s not incredibly robust,” Dr. Selvaraj observed. It may contribute to the drugs’ benefits, “but it’s definitely more than that – a lot more than that.”
DELIVER was funded by AstraZeneca. Dr. Selvaraj reported no relevant conflicts. Disclosures for the other authors are in the report. Dr. Fonarow has reported receiving personal fees from Abbott, Amgen, AstraZeneca, Bayer, Cytokinetics, Edwards, Janssen, Medtronic, Merck, and Novartis.
A version of this article first appeared on Medscape.com.
Bariatric surgery prompts visceral fat reduction, cardiac changes
Weight loss after bariatric surgery was linked with visceral fat reduction as well as reduced blood pressure, fasting glucose, and left ventricular remodeling, based an imaging study in 213 patients.
“We found that ventricular function measured by strain imaging improved in both the left and right sides of the heart, but function measured in the traditional method using endocardial motion [in other words, ejection fraction] actually worsened,” senior investigator Barry A. Borlaug, MD, said in an interview.
Although previous studies have shown positive effects of weight loss on the heart after bariatric surgery, most have been short term and have not specifically examined the effects of visceral fat reduction, wrote the investigators.
“We are in the middle of an increasing epidemic of obesity worldwide, but particularly in the United States, where it is currently projected that one in two adults will be obese by 2030,” added Dr. Borlaug of Mayo Clinic, Rochester, Minn. “Heart failure with preserved ejection fraction (HFpEF) is growing in tandem, and numerous recent studies have shown that obesity is one of the strongest risk factors for developing HFpEF, and that the severity of HFpEF is intimately linked to excess body fat. This suggests that therapies to reduce body fat could improve the cardiac abnormalities that cause HFpEF, which was our focus in this study,” he explained.
In the study, published in the Journal of the American College of Cardiology, the researchers reviewed echocardiography data from 213 obese patients before and more than 180 days after bariatric surgery. They also measured abdominal visceral adipose tissue (VAT) of 52 patients via computed tomography. The average age of the patients was 54 years, the average body mass index was 45 kg/m2, and 67% were women. Comorbidities included hypertension, diabetes, dyslipidemia, and obstructive sleep apnea.
The primary outcome was changes in cardiac structure and function.
After a median follow-up of 5.3 years, patients overall averaged a 23% reduction in body weight and a 22% reduction in BMI. In the 52 patients with abdominal scans, the VAT area decreased by 30% overall. Changes in left ventricular mass were significantly correlated to changes in the VAT.
Epicardial adipose thickness decreased by 14% overall. Left and right ventricular longitudinal strains improved at follow-up, but left atrial strain deteriorated, the researchers noted.
Although the mechanism of action remains unclear, the results suggest that left ventricular remodeling was associated with visceral adiposity rather than subcutaneous fat, the researchers wrote.
They also found that right ventricular strain was negatively correlated with VAT, but not with body weight or BMI.
“These findings suggest that weight loss, particularly reduction in visceral adiposity, benefits [right ventricular] structure and function in a manner akin to that observed in the [left ventricle],” the researchers noted.
Some surprises and limitations
Dr. Borlaug said he found some, but not all, of the results surprising. “Earlier studies had shown evidence for benefit from weight loss on cardiac structure and function, but had been limited by smaller sample sizes, shorter durations of evaluation, and variable methods used,” he said in an interview.
The findings that strain imaging showed both left and right ventricular function improved while EF declined “shows some of the problems with using EF, as it is affected by chamber size and geometry. We have previously shown that patients with HFpEF display an increase in fat around the heart, and this affects cardiac function and interaction between the left and right sides of the heart, so we expected to see that this fat depot would be reduced, and this was indeed the case,” Dr. Borlaug added.
In the current study, “visceral fat was most strongly tied to the heart remodeling in obesity, and changes in visceral fat were most strongly tied to improvements in cardiac structure following weight loss,” Dr. Borlaug told this news organization. “This further supports this concept that excess visceral fat plays a key role in HFpEF, especially in the abdomen and around the heart,” he said.
However, “The biggest surprise was the discordant effects in the left atrium,” Dr. Borlaug said. “Left atrial remodeling and dysfunction play a crucial role in HFpEF as well, and we expected that this would improve following weight loss, but in fact we observed that left atrial function deteriorated, and other indicators of atrial myopathy worsened, including higher estimates of left atrial pressures and increased prevalence of atrial fibrillation,” he said.
This difference emphasizes that weight loss may not address all abnormalities that lead to HFpEF, although a key limitation of the current study was the lack of a control group of patients with the same degree of obesity and no weight-loss intervention, and the deterioration in left atrial function might have been even greater in the absence of weight loss, Dr. Borlaug added.
Larger numbers support effects
Previous research shows that structural heart changes associated with obesity can be reversed through weight loss, but the current study fills a gap by providing long-term data in a larger sample than previously studied, wrote Paul Heidenreich, MD, of Stanford (Calif.) University in an accompanying editorial).
“There has been uncertainty regarding the prolonged effect of weight loss on cardiac function; this study was larger than many prior studies and provided a longer follow-up,” Dr. Heidenreich said in an interview.
“One unusual finding was that, while weight loss led to left ventricle reverse remodeling (reduction in wall thickness), the same effect was not seen for the left atrium; the left atrial size continued to increase,” he said. “I would have expected the left atrial changes to mirror the changes in the left ventricle,” he noted.
The findings support the greater cardiac risk of visceral vs. subcutaneous adipose tissue, and although body mass index will retain prognostic value, measures of central obesity are more likely predictors of cardiac structural changes and events and should be reported in clinical studies, Dr. Heidenreich wrote.
However, “We need a better understanding of the factors that influence left atrial remodeling and reverse remodeling,” Dr. Heidenreich told this news organization. “While left ventricular compliance and pressure play a role, there are other factors that need to be elucidated,” he said.
Studies in progress may inform practice
The current data call for further study to test novel treatments to facilitate weight loss in patients with HFpEF and those at risk for HFpEF, and some of these studies with medicines are underway, Dr. Borlaug said in the interview.
“Until such studies are completed, we will not truly understand the effects of weight loss on the heart, but the present data certainly provide strong support that patients who have obesity and HFpEF or are at risk for HFpEF should try to lose weight through lifestyle interventions,” he said.
Whether the cardiac changes seen in the current study would be different with nonsurgical weight loss remains a key question because many obese patients are reluctant to undergo bariatric surgery, Dr. Borlaug said. “We cannot assess whether the effects would differ with nonsurgical weight loss, and this requires further study,” he added.
As for additional research, “Randomized, controlled trials of weight-loss interventions, with appropriate controls and comprehensive assessments of cardiac structure, function, and hemodynamics will be most informative,” said Dr. Borlaug. “Larger trials powered to evaluate cardiovascular outcomes such as heart failure hospitalization or cardiovascular death also are critically important to better understand the role of weight loss to treat and prevent HFpEF, the ultimate form of obesity-related heart disease,” he emphasized.
The study was supported in part by grants to lead author Dr. Hidemi Sorimachi of the Mayo Clinic from the Uehara Memorial Foundation, Japan, and to corresponding author Dr. Borlaug from the National Institutes of Health. Dr. Borlaug also disclosed previous grants from National Institutes of Health/National Heart, Lung, and Blood Institute, AstraZeneca, Corvia, Medtronic, GlaxoSmithKline, Mesoblast, Novartis, and Tenax Therapeutics; and consulting fees from Actelion, Amgen, Aria, Axon Therapies, Boehringer Ingelheim, Edwards Lifesciences, Eli Lilly, Imbria, Janssen, Merck, Novo Nordisk, and VADovations. Dr. Heidenreich had no financial disclosures.
Weight loss after bariatric surgery was linked with visceral fat reduction as well as reduced blood pressure, fasting glucose, and left ventricular remodeling, based an imaging study in 213 patients.
“We found that ventricular function measured by strain imaging improved in both the left and right sides of the heart, but function measured in the traditional method using endocardial motion [in other words, ejection fraction] actually worsened,” senior investigator Barry A. Borlaug, MD, said in an interview.
Although previous studies have shown positive effects of weight loss on the heart after bariatric surgery, most have been short term and have not specifically examined the effects of visceral fat reduction, wrote the investigators.
“We are in the middle of an increasing epidemic of obesity worldwide, but particularly in the United States, where it is currently projected that one in two adults will be obese by 2030,” added Dr. Borlaug of Mayo Clinic, Rochester, Minn. “Heart failure with preserved ejection fraction (HFpEF) is growing in tandem, and numerous recent studies have shown that obesity is one of the strongest risk factors for developing HFpEF, and that the severity of HFpEF is intimately linked to excess body fat. This suggests that therapies to reduce body fat could improve the cardiac abnormalities that cause HFpEF, which was our focus in this study,” he explained.
In the study, published in the Journal of the American College of Cardiology, the researchers reviewed echocardiography data from 213 obese patients before and more than 180 days after bariatric surgery. They also measured abdominal visceral adipose tissue (VAT) of 52 patients via computed tomography. The average age of the patients was 54 years, the average body mass index was 45 kg/m2, and 67% were women. Comorbidities included hypertension, diabetes, dyslipidemia, and obstructive sleep apnea.
The primary outcome was changes in cardiac structure and function.
After a median follow-up of 5.3 years, patients overall averaged a 23% reduction in body weight and a 22% reduction in BMI. In the 52 patients with abdominal scans, the VAT area decreased by 30% overall. Changes in left ventricular mass were significantly correlated to changes in the VAT.
Epicardial adipose thickness decreased by 14% overall. Left and right ventricular longitudinal strains improved at follow-up, but left atrial strain deteriorated, the researchers noted.
Although the mechanism of action remains unclear, the results suggest that left ventricular remodeling was associated with visceral adiposity rather than subcutaneous fat, the researchers wrote.
They also found that right ventricular strain was negatively correlated with VAT, but not with body weight or BMI.
“These findings suggest that weight loss, particularly reduction in visceral adiposity, benefits [right ventricular] structure and function in a manner akin to that observed in the [left ventricle],” the researchers noted.
Some surprises and limitations
Dr. Borlaug said he found some, but not all, of the results surprising. “Earlier studies had shown evidence for benefit from weight loss on cardiac structure and function, but had been limited by smaller sample sizes, shorter durations of evaluation, and variable methods used,” he said in an interview.
The findings that strain imaging showed both left and right ventricular function improved while EF declined “shows some of the problems with using EF, as it is affected by chamber size and geometry. We have previously shown that patients with HFpEF display an increase in fat around the heart, and this affects cardiac function and interaction between the left and right sides of the heart, so we expected to see that this fat depot would be reduced, and this was indeed the case,” Dr. Borlaug added.
In the current study, “visceral fat was most strongly tied to the heart remodeling in obesity, and changes in visceral fat were most strongly tied to improvements in cardiac structure following weight loss,” Dr. Borlaug told this news organization. “This further supports this concept that excess visceral fat plays a key role in HFpEF, especially in the abdomen and around the heart,” he said.
However, “The biggest surprise was the discordant effects in the left atrium,” Dr. Borlaug said. “Left atrial remodeling and dysfunction play a crucial role in HFpEF as well, and we expected that this would improve following weight loss, but in fact we observed that left atrial function deteriorated, and other indicators of atrial myopathy worsened, including higher estimates of left atrial pressures and increased prevalence of atrial fibrillation,” he said.
This difference emphasizes that weight loss may not address all abnormalities that lead to HFpEF, although a key limitation of the current study was the lack of a control group of patients with the same degree of obesity and no weight-loss intervention, and the deterioration in left atrial function might have been even greater in the absence of weight loss, Dr. Borlaug added.
Larger numbers support effects
Previous research shows that structural heart changes associated with obesity can be reversed through weight loss, but the current study fills a gap by providing long-term data in a larger sample than previously studied, wrote Paul Heidenreich, MD, of Stanford (Calif.) University in an accompanying editorial).
“There has been uncertainty regarding the prolonged effect of weight loss on cardiac function; this study was larger than many prior studies and provided a longer follow-up,” Dr. Heidenreich said in an interview.
“One unusual finding was that, while weight loss led to left ventricle reverse remodeling (reduction in wall thickness), the same effect was not seen for the left atrium; the left atrial size continued to increase,” he said. “I would have expected the left atrial changes to mirror the changes in the left ventricle,” he noted.
The findings support the greater cardiac risk of visceral vs. subcutaneous adipose tissue, and although body mass index will retain prognostic value, measures of central obesity are more likely predictors of cardiac structural changes and events and should be reported in clinical studies, Dr. Heidenreich wrote.
However, “We need a better understanding of the factors that influence left atrial remodeling and reverse remodeling,” Dr. Heidenreich told this news organization. “While left ventricular compliance and pressure play a role, there are other factors that need to be elucidated,” he said.
Studies in progress may inform practice
The current data call for further study to test novel treatments to facilitate weight loss in patients with HFpEF and those at risk for HFpEF, and some of these studies with medicines are underway, Dr. Borlaug said in the interview.
“Until such studies are completed, we will not truly understand the effects of weight loss on the heart, but the present data certainly provide strong support that patients who have obesity and HFpEF or are at risk for HFpEF should try to lose weight through lifestyle interventions,” he said.
Whether the cardiac changes seen in the current study would be different with nonsurgical weight loss remains a key question because many obese patients are reluctant to undergo bariatric surgery, Dr. Borlaug said. “We cannot assess whether the effects would differ with nonsurgical weight loss, and this requires further study,” he added.
As for additional research, “Randomized, controlled trials of weight-loss interventions, with appropriate controls and comprehensive assessments of cardiac structure, function, and hemodynamics will be most informative,” said Dr. Borlaug. “Larger trials powered to evaluate cardiovascular outcomes such as heart failure hospitalization or cardiovascular death also are critically important to better understand the role of weight loss to treat and prevent HFpEF, the ultimate form of obesity-related heart disease,” he emphasized.
The study was supported in part by grants to lead author Dr. Hidemi Sorimachi of the Mayo Clinic from the Uehara Memorial Foundation, Japan, and to corresponding author Dr. Borlaug from the National Institutes of Health. Dr. Borlaug also disclosed previous grants from National Institutes of Health/National Heart, Lung, and Blood Institute, AstraZeneca, Corvia, Medtronic, GlaxoSmithKline, Mesoblast, Novartis, and Tenax Therapeutics; and consulting fees from Actelion, Amgen, Aria, Axon Therapies, Boehringer Ingelheim, Edwards Lifesciences, Eli Lilly, Imbria, Janssen, Merck, Novo Nordisk, and VADovations. Dr. Heidenreich had no financial disclosures.
Weight loss after bariatric surgery was linked with visceral fat reduction as well as reduced blood pressure, fasting glucose, and left ventricular remodeling, based an imaging study in 213 patients.
“We found that ventricular function measured by strain imaging improved in both the left and right sides of the heart, but function measured in the traditional method using endocardial motion [in other words, ejection fraction] actually worsened,” senior investigator Barry A. Borlaug, MD, said in an interview.
Although previous studies have shown positive effects of weight loss on the heart after bariatric surgery, most have been short term and have not specifically examined the effects of visceral fat reduction, wrote the investigators.
“We are in the middle of an increasing epidemic of obesity worldwide, but particularly in the United States, where it is currently projected that one in two adults will be obese by 2030,” added Dr. Borlaug of Mayo Clinic, Rochester, Minn. “Heart failure with preserved ejection fraction (HFpEF) is growing in tandem, and numerous recent studies have shown that obesity is one of the strongest risk factors for developing HFpEF, and that the severity of HFpEF is intimately linked to excess body fat. This suggests that therapies to reduce body fat could improve the cardiac abnormalities that cause HFpEF, which was our focus in this study,” he explained.
In the study, published in the Journal of the American College of Cardiology, the researchers reviewed echocardiography data from 213 obese patients before and more than 180 days after bariatric surgery. They also measured abdominal visceral adipose tissue (VAT) of 52 patients via computed tomography. The average age of the patients was 54 years, the average body mass index was 45 kg/m2, and 67% were women. Comorbidities included hypertension, diabetes, dyslipidemia, and obstructive sleep apnea.
The primary outcome was changes in cardiac structure and function.
After a median follow-up of 5.3 years, patients overall averaged a 23% reduction in body weight and a 22% reduction in BMI. In the 52 patients with abdominal scans, the VAT area decreased by 30% overall. Changes in left ventricular mass were significantly correlated to changes in the VAT.
Epicardial adipose thickness decreased by 14% overall. Left and right ventricular longitudinal strains improved at follow-up, but left atrial strain deteriorated, the researchers noted.
Although the mechanism of action remains unclear, the results suggest that left ventricular remodeling was associated with visceral adiposity rather than subcutaneous fat, the researchers wrote.
They also found that right ventricular strain was negatively correlated with VAT, but not with body weight or BMI.
“These findings suggest that weight loss, particularly reduction in visceral adiposity, benefits [right ventricular] structure and function in a manner akin to that observed in the [left ventricle],” the researchers noted.
Some surprises and limitations
Dr. Borlaug said he found some, but not all, of the results surprising. “Earlier studies had shown evidence for benefit from weight loss on cardiac structure and function, but had been limited by smaller sample sizes, shorter durations of evaluation, and variable methods used,” he said in an interview.
The findings that strain imaging showed both left and right ventricular function improved while EF declined “shows some of the problems with using EF, as it is affected by chamber size and geometry. We have previously shown that patients with HFpEF display an increase in fat around the heart, and this affects cardiac function and interaction between the left and right sides of the heart, so we expected to see that this fat depot would be reduced, and this was indeed the case,” Dr. Borlaug added.
In the current study, “visceral fat was most strongly tied to the heart remodeling in obesity, and changes in visceral fat were most strongly tied to improvements in cardiac structure following weight loss,” Dr. Borlaug told this news organization. “This further supports this concept that excess visceral fat plays a key role in HFpEF, especially in the abdomen and around the heart,” he said.
However, “The biggest surprise was the discordant effects in the left atrium,” Dr. Borlaug said. “Left atrial remodeling and dysfunction play a crucial role in HFpEF as well, and we expected that this would improve following weight loss, but in fact we observed that left atrial function deteriorated, and other indicators of atrial myopathy worsened, including higher estimates of left atrial pressures and increased prevalence of atrial fibrillation,” he said.
This difference emphasizes that weight loss may not address all abnormalities that lead to HFpEF, although a key limitation of the current study was the lack of a control group of patients with the same degree of obesity and no weight-loss intervention, and the deterioration in left atrial function might have been even greater in the absence of weight loss, Dr. Borlaug added.
Larger numbers support effects
Previous research shows that structural heart changes associated with obesity can be reversed through weight loss, but the current study fills a gap by providing long-term data in a larger sample than previously studied, wrote Paul Heidenreich, MD, of Stanford (Calif.) University in an accompanying editorial).
“There has been uncertainty regarding the prolonged effect of weight loss on cardiac function; this study was larger than many prior studies and provided a longer follow-up,” Dr. Heidenreich said in an interview.
“One unusual finding was that, while weight loss led to left ventricle reverse remodeling (reduction in wall thickness), the same effect was not seen for the left atrium; the left atrial size continued to increase,” he said. “I would have expected the left atrial changes to mirror the changes in the left ventricle,” he noted.
The findings support the greater cardiac risk of visceral vs. subcutaneous adipose tissue, and although body mass index will retain prognostic value, measures of central obesity are more likely predictors of cardiac structural changes and events and should be reported in clinical studies, Dr. Heidenreich wrote.
However, “We need a better understanding of the factors that influence left atrial remodeling and reverse remodeling,” Dr. Heidenreich told this news organization. “While left ventricular compliance and pressure play a role, there are other factors that need to be elucidated,” he said.
Studies in progress may inform practice
The current data call for further study to test novel treatments to facilitate weight loss in patients with HFpEF and those at risk for HFpEF, and some of these studies with medicines are underway, Dr. Borlaug said in the interview.
“Until such studies are completed, we will not truly understand the effects of weight loss on the heart, but the present data certainly provide strong support that patients who have obesity and HFpEF or are at risk for HFpEF should try to lose weight through lifestyle interventions,” he said.
Whether the cardiac changes seen in the current study would be different with nonsurgical weight loss remains a key question because many obese patients are reluctant to undergo bariatric surgery, Dr. Borlaug said. “We cannot assess whether the effects would differ with nonsurgical weight loss, and this requires further study,” he added.
As for additional research, “Randomized, controlled trials of weight-loss interventions, with appropriate controls and comprehensive assessments of cardiac structure, function, and hemodynamics will be most informative,” said Dr. Borlaug. “Larger trials powered to evaluate cardiovascular outcomes such as heart failure hospitalization or cardiovascular death also are critically important to better understand the role of weight loss to treat and prevent HFpEF, the ultimate form of obesity-related heart disease,” he emphasized.
The study was supported in part by grants to lead author Dr. Hidemi Sorimachi of the Mayo Clinic from the Uehara Memorial Foundation, Japan, and to corresponding author Dr. Borlaug from the National Institutes of Health. Dr. Borlaug also disclosed previous grants from National Institutes of Health/National Heart, Lung, and Blood Institute, AstraZeneca, Corvia, Medtronic, GlaxoSmithKline, Mesoblast, Novartis, and Tenax Therapeutics; and consulting fees from Actelion, Amgen, Aria, Axon Therapies, Boehringer Ingelheim, Edwards Lifesciences, Eli Lilly, Imbria, Janssen, Merck, Novo Nordisk, and VADovations. Dr. Heidenreich had no financial disclosures.
FROM JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Type 2 Diabetes in Adolescents: We Must Do Better
Type 2 diabetes (T2D) has become a noncommunicable pandemic. Approximately 14.7% of the US adult population has diabetes.1 Additionally, nearly 25% of the geriatric population has diabetes and nearly 50% has prediabetes.2 Needless to say, most practices, regardless of specialty, see many patients with diabetes. We have made major advances in diabetes treatments, yet diabetes mellitus is still the leading cause of legal blindness, nontraumatic amputation, and end-stage renal disease requiring dialysis.3
While the prevalence of diabetes in adults is concerning, what is even more startling is the significant increase of T2D within the pediatric population. It was not too long ago that we considered T2D an “adult-only” disease. Now, 24% of children with diabetes have T2D, and 18% of adolescents have prediabetes.4,5 This is not the end of the story. Recent studies have identified that the earlier you are diagnosed with T2D, the less responsive you are to diabetes treatments—and the disease will progress more rapidly to complications.
We know that pediatric patients are not little adults. There are important physiologic and metabolic differences in our younger patients. The RISE study found that adolescents have lower insulin sensitivity than adults.4,6 The pancreatic beta cells are more responsive at first and there is less clearance by the liver, which may indeed make insulin resistance worse. Finally, pancreatic beta cell function declines more rapidly in adolescents than in adults.4,6 These physiologic changes can be even worse during puberty. The hormonal changes seen in puberty accelerate and amplify insulin secretion and worsen insulin resistance, which can result in hyperglycemia in those at risk.7,8
The other complicating factor is the rapid rise in obesity in Americans. While childhood obesity is not quite at adult levels, it is a major risk factor for adult obesity. The prevalence of obesity in childhood was recently estimated to be 19.7% and is still on the rise.9 Obesity can be diabetogenic as we see an increase in visceral obesity. This triggers an inflammatory response that leads to worsening systemic insulin resistance and lipotoxicity from elevated circulating free fatty acids.8
Lifestyle and behavioral factors are also important in adolescents with T2D. While they are more independent than younger children, they are still largely dependent on the foods that are available in their home. Family food choices have a major impact on our youth. Further, the foods that our adolescents eat outside the home are more likely to be fast food or ultra-processed foods, which have been shown to contribute to obesity and T2D.
Family history is a strong predictor of risk for T2D. In the Treatment Options for Type 2 Diabetes in Adolescents and Youth (TODAY) cohort, 89.4% of pediatric participants had a first-degree relative or grandparent with T2D.10 This highlights the importance of both genetic risk and living environment as risks for T2D.
The American Diabetes Association recommends that all children with specific risk factors be screened for diabetes starting at the age 10 years or at puberty, whichever comes first.11 The screening tests recommended for diabetes are currently the same as for adults, yet there are few data supporting this regimen. To diagnose diabetes, you can use any of the following screening tests: fasting glucose, glucose tolerance, or glycated hemoglobin (HbA1c).1
Risk Factors That Should Prompt Diabetes Screening11
Screening is recommended in children who are overweight (≥85%) or obese (≥95%) and who also have ≥1 of the following risk factors:
- Family history of T2D in a first- or second-degree relative
- Maternal history of gestational diabetes
- Low birth weight for gestational age
- Physical signs of insulin resistance or related conditions (eg, hypertension, dyslipidemia, polycystic ovary syndrome)
- High-risk race/ethnicity (Native American, African American, Pacific Islander, Latino)
Diagnostic Criteria for Diabetes Mellitus11
Testing method | Normal range | Pre-diabetes | Diabetes mellitus |
Fasting glucose | <100 mg/dL | 100-125 mg/dL | ≥126 mg/dL |
2-hour glucose tolerance test | <140 mg/dL | 140-199 mg/dL | ≥200 mg/dL |
HbA1c | <5.7% | 5.7%-6.4% | ≥6.5% |
A childhood or adolescent T2D diagnosis should be taken seriously and communicated to the patient and family in a timely manner. Treatment should start immediately. There are several factors that make managing T2D in adolescents more challenging. Children do not control key aspects of their life, including nutrition and, often, free time activity. There are a lot of social pressures to be “normal,” and having a chronic disease will definitely make the child feel “different” and potentially feel socially isolated. There are high rates of mood disorders in children with diabetes, which can make self-management even harder.12
As mentioned above, treatment should begin immediately upon diagnosis. This is because T2D in younger people tends to be more progressive and less responsive to treatment options, and patients are much more likely to develop.1,13,14 These same complications can be seen in adult patients, but in younger patients they develop earlier in the disease; specifically, renal and neurologic complications occur at even higher rates.14
The initial treatment should include both family-based therapeutic lifestyle changes (ie, nutrition, physical activity intervention) and medication.11 There are fewer US Food and Drug Administration–approved medication options for children and adolescents, and those treatments that have been approved are less durable in this population.
Metformin and insulin are the most-used medications, but their initiation is often delayed, as therapeutic lifestyle change is tried first. This has not been shown to be an effective strategy and may even undermine the value of therapeutic lifestyle change if the family is told later that medication may still need to be added.
Recent studies have shown the benefit of select glucagon-like peptide-1 receptor agonists (GLP-1 RAs) as important therapeutic tools to treat T2D in adolescence. Dulaglutide, exenatide, and liraglutide have been shown to be safe and effective in trials for adolescents with T2D.15-17 These agents reduce glucose and body weight and may be important tools to help reduce extra glycemic risks (eg, cardiovascular disease, kidney disease), but they have not been studied for this purpose yet.
Further, there is good support for the use of bariatric surgery for adolescents. While this is a relatively new treatment option, early and mid-term results are favorable compared with medication-based strategies.18 Further studies are needed to determine the long-term benefits.
Take home points:
- T2D is becoming increasingly common in our youth.
- T2D, when diagnosed earlier in life, is more progressive, less responsive to treatment options, and associated with earlier complications.
- New studies support the use of novel therapies such as GLP-1 RAs and metabolic surgery in this age group.
Dr. Shubrook and Dr. Antonia M. Molinari have written a comprehensive review on treatment options and current guidelines for the management of T2D in the pediatric population, which can supply further information.19
National and state diabetes trends 2021. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/library/reports/reportcard/national-state-diabetes-trends.html
National diabetes statistics report. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/data/statistics-report/index.html
National diabetes statistics report. Estimates of diabetes and its burden in the United States. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf
RISE Consortium. Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: I. Observations using the hyperglycemic clamp. Diabetes Care. 2018;41(8):1696-1706. doi:10.2337/dc18-0244
Andes LJ, Cheng YJ, Rolka DB, Gregg EW, Imperatore G. Prevalence of prediabetes among adolescents and young adults in the United States, 2005-2016. JAMA Pediatr. 2020;174:e194498. doi:10.1001/jamapediatrics.2019.4498
RISE Consortium. Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: II. Observations using the oral glucose tolerance test. Diabetes Care. 2018;41(8):1707-1716. doi:10.2337/dc18-0243.
Cree-Green M, Triolo TM, Nadeau KJ. Etiology of insulin resistance in youth with type 2 diabetes. Curr Diab Rep. 2013;13(1):81-88. doi:10.1007/s11892-012-0341-0
Goran MI, Gower BA. Longitudinal study on pubertal insulin resistance. Diabetes. 2001;50(11):2444-2450. doi:10.2337/diabetes.50.11.2444
Childhood obesity facts. Centers for Disease Control and Prevention. Accessed October 3, 2022. https://www.cdc.gov/obesity/data/childhood.html
Copeland KC, Zeitler P, Geffner M, et al. Characteristics of adolescents and youth with recent-onset type 2 diabetes: the TODAY cohort at baseline. J Clin Endocrinol Metab. 2011;96(1):159-167. doi:10.1210/jc.2010-1642
Arslanian S, Bacha F, Grey M, Marcus MD, White NH, Zeitler P. Evaluation and management of youth-onset type 2 diabetes: a position statement by the American Diabetes Association. Diabetes Care. 2018;41(12):2648-2668. doi:10.2337/dci18-0052
Anderson BJ, Edelstein S, Abramson NW, et al. Depressive symptoms and quality of life in adolescents with type 2 diabetes: baseline data from the TODAY study. Diabetes Care. 2011;34(10):2205-2207. doi:10.2337/dc11-0431
TODAY Study Group. Treatment options for type 2 diabetes in adolescents and youth: a study of the comparative efficacy of metformin alone or in combination with rosiglitazone or lifestyle intervention in adolescents with type 2 diabetes. Pediatr Diabetes. 2007;8(2):74-87. doi:10.1111/j.1399-5448.2007.00237.x
Dart AB, Martens PJ, Rigatto C, Brownell MD, Dean HJ, Sellers EA. Earlier onset of complications in youth with type 2 diabetes. Diabetes Care. 2014;37(2):436-443. doi:10.2337/dc13-0954
Arslanian SA, Hannon T, Zeitler P et al. Once-weekly dulaglutide for the treatment of youths with type 2 diabetes. N Engl J Med. 2022;387(5):433-443. doi:10.1056/NEJMoa2204601
Tamborlane WV, Barrientos-Pérez M, Fainberg U, et al. Liraglutide in children and adolescents with type 2 diabetes. N Engl J Med. 2019;381(7):637-646. doi:10.1056/NEJMoa1903822
Tamborlane WV, Bishai R, Geller D, et al. Once-weekly exenatide in youth with type 2 diabetes. Diabetes Care. 2022;45(8):1833-1840. doi:10.2337/dc21-2275
Inge TH, Laffel LM, Jenkins TM, et al. Comparison of surgical and medical therapy for type 2 diabetes in severely obese adolescents. JAMA Pediatr. 2018;172(5):452-460. doi:10.1001/jamapediatrics.2017.5763
Molinari AM, Shubrook JH. Treatment options and current guidelines of care for pediatric type 2 diabetes patients: a narrative review. J Osteopath Med. 2021;121(4):431-440. doi:10.1515/jom-2020-0172
Type 2 diabetes (T2D) has become a noncommunicable pandemic. Approximately 14.7% of the US adult population has diabetes.1 Additionally, nearly 25% of the geriatric population has diabetes and nearly 50% has prediabetes.2 Needless to say, most practices, regardless of specialty, see many patients with diabetes. We have made major advances in diabetes treatments, yet diabetes mellitus is still the leading cause of legal blindness, nontraumatic amputation, and end-stage renal disease requiring dialysis.3
While the prevalence of diabetes in adults is concerning, what is even more startling is the significant increase of T2D within the pediatric population. It was not too long ago that we considered T2D an “adult-only” disease. Now, 24% of children with diabetes have T2D, and 18% of adolescents have prediabetes.4,5 This is not the end of the story. Recent studies have identified that the earlier you are diagnosed with T2D, the less responsive you are to diabetes treatments—and the disease will progress more rapidly to complications.
We know that pediatric patients are not little adults. There are important physiologic and metabolic differences in our younger patients. The RISE study found that adolescents have lower insulin sensitivity than adults.4,6 The pancreatic beta cells are more responsive at first and there is less clearance by the liver, which may indeed make insulin resistance worse. Finally, pancreatic beta cell function declines more rapidly in adolescents than in adults.4,6 These physiologic changes can be even worse during puberty. The hormonal changes seen in puberty accelerate and amplify insulin secretion and worsen insulin resistance, which can result in hyperglycemia in those at risk.7,8
The other complicating factor is the rapid rise in obesity in Americans. While childhood obesity is not quite at adult levels, it is a major risk factor for adult obesity. The prevalence of obesity in childhood was recently estimated to be 19.7% and is still on the rise.9 Obesity can be diabetogenic as we see an increase in visceral obesity. This triggers an inflammatory response that leads to worsening systemic insulin resistance and lipotoxicity from elevated circulating free fatty acids.8
Lifestyle and behavioral factors are also important in adolescents with T2D. While they are more independent than younger children, they are still largely dependent on the foods that are available in their home. Family food choices have a major impact on our youth. Further, the foods that our adolescents eat outside the home are more likely to be fast food or ultra-processed foods, which have been shown to contribute to obesity and T2D.
Family history is a strong predictor of risk for T2D. In the Treatment Options for Type 2 Diabetes in Adolescents and Youth (TODAY) cohort, 89.4% of pediatric participants had a first-degree relative or grandparent with T2D.10 This highlights the importance of both genetic risk and living environment as risks for T2D.
The American Diabetes Association recommends that all children with specific risk factors be screened for diabetes starting at the age 10 years or at puberty, whichever comes first.11 The screening tests recommended for diabetes are currently the same as for adults, yet there are few data supporting this regimen. To diagnose diabetes, you can use any of the following screening tests: fasting glucose, glucose tolerance, or glycated hemoglobin (HbA1c).1
Risk Factors That Should Prompt Diabetes Screening11
Screening is recommended in children who are overweight (≥85%) or obese (≥95%) and who also have ≥1 of the following risk factors:
- Family history of T2D in a first- or second-degree relative
- Maternal history of gestational diabetes
- Low birth weight for gestational age
- Physical signs of insulin resistance or related conditions (eg, hypertension, dyslipidemia, polycystic ovary syndrome)
- High-risk race/ethnicity (Native American, African American, Pacific Islander, Latino)
Diagnostic Criteria for Diabetes Mellitus11
Testing method | Normal range | Pre-diabetes | Diabetes mellitus |
Fasting glucose | <100 mg/dL | 100-125 mg/dL | ≥126 mg/dL |
2-hour glucose tolerance test | <140 mg/dL | 140-199 mg/dL | ≥200 mg/dL |
HbA1c | <5.7% | 5.7%-6.4% | ≥6.5% |
A childhood or adolescent T2D diagnosis should be taken seriously and communicated to the patient and family in a timely manner. Treatment should start immediately. There are several factors that make managing T2D in adolescents more challenging. Children do not control key aspects of their life, including nutrition and, often, free time activity. There are a lot of social pressures to be “normal,” and having a chronic disease will definitely make the child feel “different” and potentially feel socially isolated. There are high rates of mood disorders in children with diabetes, which can make self-management even harder.12
As mentioned above, treatment should begin immediately upon diagnosis. This is because T2D in younger people tends to be more progressive and less responsive to treatment options, and patients are much more likely to develop.1,13,14 These same complications can be seen in adult patients, but in younger patients they develop earlier in the disease; specifically, renal and neurologic complications occur at even higher rates.14
The initial treatment should include both family-based therapeutic lifestyle changes (ie, nutrition, physical activity intervention) and medication.11 There are fewer US Food and Drug Administration–approved medication options for children and adolescents, and those treatments that have been approved are less durable in this population.
Metformin and insulin are the most-used medications, but their initiation is often delayed, as therapeutic lifestyle change is tried first. This has not been shown to be an effective strategy and may even undermine the value of therapeutic lifestyle change if the family is told later that medication may still need to be added.
Recent studies have shown the benefit of select glucagon-like peptide-1 receptor agonists (GLP-1 RAs) as important therapeutic tools to treat T2D in adolescence. Dulaglutide, exenatide, and liraglutide have been shown to be safe and effective in trials for adolescents with T2D.15-17 These agents reduce glucose and body weight and may be important tools to help reduce extra glycemic risks (eg, cardiovascular disease, kidney disease), but they have not been studied for this purpose yet.
Further, there is good support for the use of bariatric surgery for adolescents. While this is a relatively new treatment option, early and mid-term results are favorable compared with medication-based strategies.18 Further studies are needed to determine the long-term benefits.
Take home points:
- T2D is becoming increasingly common in our youth.
- T2D, when diagnosed earlier in life, is more progressive, less responsive to treatment options, and associated with earlier complications.
- New studies support the use of novel therapies such as GLP-1 RAs and metabolic surgery in this age group.
Dr. Shubrook and Dr. Antonia M. Molinari have written a comprehensive review on treatment options and current guidelines for the management of T2D in the pediatric population, which can supply further information.19
Type 2 diabetes (T2D) has become a noncommunicable pandemic. Approximately 14.7% of the US adult population has diabetes.1 Additionally, nearly 25% of the geriatric population has diabetes and nearly 50% has prediabetes.2 Needless to say, most practices, regardless of specialty, see many patients with diabetes. We have made major advances in diabetes treatments, yet diabetes mellitus is still the leading cause of legal blindness, nontraumatic amputation, and end-stage renal disease requiring dialysis.3
While the prevalence of diabetes in adults is concerning, what is even more startling is the significant increase of T2D within the pediatric population. It was not too long ago that we considered T2D an “adult-only” disease. Now, 24% of children with diabetes have T2D, and 18% of adolescents have prediabetes.4,5 This is not the end of the story. Recent studies have identified that the earlier you are diagnosed with T2D, the less responsive you are to diabetes treatments—and the disease will progress more rapidly to complications.
We know that pediatric patients are not little adults. There are important physiologic and metabolic differences in our younger patients. The RISE study found that adolescents have lower insulin sensitivity than adults.4,6 The pancreatic beta cells are more responsive at first and there is less clearance by the liver, which may indeed make insulin resistance worse. Finally, pancreatic beta cell function declines more rapidly in adolescents than in adults.4,6 These physiologic changes can be even worse during puberty. The hormonal changes seen in puberty accelerate and amplify insulin secretion and worsen insulin resistance, which can result in hyperglycemia in those at risk.7,8
The other complicating factor is the rapid rise in obesity in Americans. While childhood obesity is not quite at adult levels, it is a major risk factor for adult obesity. The prevalence of obesity in childhood was recently estimated to be 19.7% and is still on the rise.9 Obesity can be diabetogenic as we see an increase in visceral obesity. This triggers an inflammatory response that leads to worsening systemic insulin resistance and lipotoxicity from elevated circulating free fatty acids.8
Lifestyle and behavioral factors are also important in adolescents with T2D. While they are more independent than younger children, they are still largely dependent on the foods that are available in their home. Family food choices have a major impact on our youth. Further, the foods that our adolescents eat outside the home are more likely to be fast food or ultra-processed foods, which have been shown to contribute to obesity and T2D.
Family history is a strong predictor of risk for T2D. In the Treatment Options for Type 2 Diabetes in Adolescents and Youth (TODAY) cohort, 89.4% of pediatric participants had a first-degree relative or grandparent with T2D.10 This highlights the importance of both genetic risk and living environment as risks for T2D.
The American Diabetes Association recommends that all children with specific risk factors be screened for diabetes starting at the age 10 years or at puberty, whichever comes first.11 The screening tests recommended for diabetes are currently the same as for adults, yet there are few data supporting this regimen. To diagnose diabetes, you can use any of the following screening tests: fasting glucose, glucose tolerance, or glycated hemoglobin (HbA1c).1
Risk Factors That Should Prompt Diabetes Screening11
Screening is recommended in children who are overweight (≥85%) or obese (≥95%) and who also have ≥1 of the following risk factors:
- Family history of T2D in a first- or second-degree relative
- Maternal history of gestational diabetes
- Low birth weight for gestational age
- Physical signs of insulin resistance or related conditions (eg, hypertension, dyslipidemia, polycystic ovary syndrome)
- High-risk race/ethnicity (Native American, African American, Pacific Islander, Latino)
Diagnostic Criteria for Diabetes Mellitus11
Testing method | Normal range | Pre-diabetes | Diabetes mellitus |
Fasting glucose | <100 mg/dL | 100-125 mg/dL | ≥126 mg/dL |
2-hour glucose tolerance test | <140 mg/dL | 140-199 mg/dL | ≥200 mg/dL |
HbA1c | <5.7% | 5.7%-6.4% | ≥6.5% |
A childhood or adolescent T2D diagnosis should be taken seriously and communicated to the patient and family in a timely manner. Treatment should start immediately. There are several factors that make managing T2D in adolescents more challenging. Children do not control key aspects of their life, including nutrition and, often, free time activity. There are a lot of social pressures to be “normal,” and having a chronic disease will definitely make the child feel “different” and potentially feel socially isolated. There are high rates of mood disorders in children with diabetes, which can make self-management even harder.12
As mentioned above, treatment should begin immediately upon diagnosis. This is because T2D in younger people tends to be more progressive and less responsive to treatment options, and patients are much more likely to develop.1,13,14 These same complications can be seen in adult patients, but in younger patients they develop earlier in the disease; specifically, renal and neurologic complications occur at even higher rates.14
The initial treatment should include both family-based therapeutic lifestyle changes (ie, nutrition, physical activity intervention) and medication.11 There are fewer US Food and Drug Administration–approved medication options for children and adolescents, and those treatments that have been approved are less durable in this population.
Metformin and insulin are the most-used medications, but their initiation is often delayed, as therapeutic lifestyle change is tried first. This has not been shown to be an effective strategy and may even undermine the value of therapeutic lifestyle change if the family is told later that medication may still need to be added.
Recent studies have shown the benefit of select glucagon-like peptide-1 receptor agonists (GLP-1 RAs) as important therapeutic tools to treat T2D in adolescence. Dulaglutide, exenatide, and liraglutide have been shown to be safe and effective in trials for adolescents with T2D.15-17 These agents reduce glucose and body weight and may be important tools to help reduce extra glycemic risks (eg, cardiovascular disease, kidney disease), but they have not been studied for this purpose yet.
Further, there is good support for the use of bariatric surgery for adolescents. While this is a relatively new treatment option, early and mid-term results are favorable compared with medication-based strategies.18 Further studies are needed to determine the long-term benefits.
Take home points:
- T2D is becoming increasingly common in our youth.
- T2D, when diagnosed earlier in life, is more progressive, less responsive to treatment options, and associated with earlier complications.
- New studies support the use of novel therapies such as GLP-1 RAs and metabolic surgery in this age group.
Dr. Shubrook and Dr. Antonia M. Molinari have written a comprehensive review on treatment options and current guidelines for the management of T2D in the pediatric population, which can supply further information.19
National and state diabetes trends 2021. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/library/reports/reportcard/national-state-diabetes-trends.html
National diabetes statistics report. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/data/statistics-report/index.html
National diabetes statistics report. Estimates of diabetes and its burden in the United States. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf
RISE Consortium. Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: I. Observations using the hyperglycemic clamp. Diabetes Care. 2018;41(8):1696-1706. doi:10.2337/dc18-0244
Andes LJ, Cheng YJ, Rolka DB, Gregg EW, Imperatore G. Prevalence of prediabetes among adolescents and young adults in the United States, 2005-2016. JAMA Pediatr. 2020;174:e194498. doi:10.1001/jamapediatrics.2019.4498
RISE Consortium. Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: II. Observations using the oral glucose tolerance test. Diabetes Care. 2018;41(8):1707-1716. doi:10.2337/dc18-0243.
Cree-Green M, Triolo TM, Nadeau KJ. Etiology of insulin resistance in youth with type 2 diabetes. Curr Diab Rep. 2013;13(1):81-88. doi:10.1007/s11892-012-0341-0
Goran MI, Gower BA. Longitudinal study on pubertal insulin resistance. Diabetes. 2001;50(11):2444-2450. doi:10.2337/diabetes.50.11.2444
Childhood obesity facts. Centers for Disease Control and Prevention. Accessed October 3, 2022. https://www.cdc.gov/obesity/data/childhood.html
Copeland KC, Zeitler P, Geffner M, et al. Characteristics of adolescents and youth with recent-onset type 2 diabetes: the TODAY cohort at baseline. J Clin Endocrinol Metab. 2011;96(1):159-167. doi:10.1210/jc.2010-1642
Arslanian S, Bacha F, Grey M, Marcus MD, White NH, Zeitler P. Evaluation and management of youth-onset type 2 diabetes: a position statement by the American Diabetes Association. Diabetes Care. 2018;41(12):2648-2668. doi:10.2337/dci18-0052
Anderson BJ, Edelstein S, Abramson NW, et al. Depressive symptoms and quality of life in adolescents with type 2 diabetes: baseline data from the TODAY study. Diabetes Care. 2011;34(10):2205-2207. doi:10.2337/dc11-0431
TODAY Study Group. Treatment options for type 2 diabetes in adolescents and youth: a study of the comparative efficacy of metformin alone or in combination with rosiglitazone or lifestyle intervention in adolescents with type 2 diabetes. Pediatr Diabetes. 2007;8(2):74-87. doi:10.1111/j.1399-5448.2007.00237.x
Dart AB, Martens PJ, Rigatto C, Brownell MD, Dean HJ, Sellers EA. Earlier onset of complications in youth with type 2 diabetes. Diabetes Care. 2014;37(2):436-443. doi:10.2337/dc13-0954
Arslanian SA, Hannon T, Zeitler P et al. Once-weekly dulaglutide for the treatment of youths with type 2 diabetes. N Engl J Med. 2022;387(5):433-443. doi:10.1056/NEJMoa2204601
Tamborlane WV, Barrientos-Pérez M, Fainberg U, et al. Liraglutide in children and adolescents with type 2 diabetes. N Engl J Med. 2019;381(7):637-646. doi:10.1056/NEJMoa1903822
Tamborlane WV, Bishai R, Geller D, et al. Once-weekly exenatide in youth with type 2 diabetes. Diabetes Care. 2022;45(8):1833-1840. doi:10.2337/dc21-2275
Inge TH, Laffel LM, Jenkins TM, et al. Comparison of surgical and medical therapy for type 2 diabetes in severely obese adolescents. JAMA Pediatr. 2018;172(5):452-460. doi:10.1001/jamapediatrics.2017.5763
Molinari AM, Shubrook JH. Treatment options and current guidelines of care for pediatric type 2 diabetes patients: a narrative review. J Osteopath Med. 2021;121(4):431-440. doi:10.1515/jom-2020-0172
National and state diabetes trends 2021. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/library/reports/reportcard/national-state-diabetes-trends.html
National diabetes statistics report. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/data/statistics-report/index.html
National diabetes statistics report. Estimates of diabetes and its burden in the United States. Centers for Disease Control and Prevention. Accessed September 26, 2022. https://www.cdc.gov/diabetes/pdfs/data/statistics/national-diabetes-statistics-report.pdf
RISE Consortium. Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: I. Observations using the hyperglycemic clamp. Diabetes Care. 2018;41(8):1696-1706. doi:10.2337/dc18-0244
Andes LJ, Cheng YJ, Rolka DB, Gregg EW, Imperatore G. Prevalence of prediabetes among adolescents and young adults in the United States, 2005-2016. JAMA Pediatr. 2020;174:e194498. doi:10.1001/jamapediatrics.2019.4498
RISE Consortium. Metabolic contrasts between youth and adults with impaired glucose tolerance or recently diagnosed type 2 diabetes: II. Observations using the oral glucose tolerance test. Diabetes Care. 2018;41(8):1707-1716. doi:10.2337/dc18-0243.
Cree-Green M, Triolo TM, Nadeau KJ. Etiology of insulin resistance in youth with type 2 diabetes. Curr Diab Rep. 2013;13(1):81-88. doi:10.1007/s11892-012-0341-0
Goran MI, Gower BA. Longitudinal study on pubertal insulin resistance. Diabetes. 2001;50(11):2444-2450. doi:10.2337/diabetes.50.11.2444
Childhood obesity facts. Centers for Disease Control and Prevention. Accessed October 3, 2022. https://www.cdc.gov/obesity/data/childhood.html
Copeland KC, Zeitler P, Geffner M, et al. Characteristics of adolescents and youth with recent-onset type 2 diabetes: the TODAY cohort at baseline. J Clin Endocrinol Metab. 2011;96(1):159-167. doi:10.1210/jc.2010-1642
Arslanian S, Bacha F, Grey M, Marcus MD, White NH, Zeitler P. Evaluation and management of youth-onset type 2 diabetes: a position statement by the American Diabetes Association. Diabetes Care. 2018;41(12):2648-2668. doi:10.2337/dci18-0052
Anderson BJ, Edelstein S, Abramson NW, et al. Depressive symptoms and quality of life in adolescents with type 2 diabetes: baseline data from the TODAY study. Diabetes Care. 2011;34(10):2205-2207. doi:10.2337/dc11-0431
TODAY Study Group. Treatment options for type 2 diabetes in adolescents and youth: a study of the comparative efficacy of metformin alone or in combination with rosiglitazone or lifestyle intervention in adolescents with type 2 diabetes. Pediatr Diabetes. 2007;8(2):74-87. doi:10.1111/j.1399-5448.2007.00237.x
Dart AB, Martens PJ, Rigatto C, Brownell MD, Dean HJ, Sellers EA. Earlier onset of complications in youth with type 2 diabetes. Diabetes Care. 2014;37(2):436-443. doi:10.2337/dc13-0954
Arslanian SA, Hannon T, Zeitler P et al. Once-weekly dulaglutide for the treatment of youths with type 2 diabetes. N Engl J Med. 2022;387(5):433-443. doi:10.1056/NEJMoa2204601
Tamborlane WV, Barrientos-Pérez M, Fainberg U, et al. Liraglutide in children and adolescents with type 2 diabetes. N Engl J Med. 2019;381(7):637-646. doi:10.1056/NEJMoa1903822
Tamborlane WV, Bishai R, Geller D, et al. Once-weekly exenatide in youth with type 2 diabetes. Diabetes Care. 2022;45(8):1833-1840. doi:10.2337/dc21-2275
Inge TH, Laffel LM, Jenkins TM, et al. Comparison of surgical and medical therapy for type 2 diabetes in severely obese adolescents. JAMA Pediatr. 2018;172(5):452-460. doi:10.1001/jamapediatrics.2017.5763
Molinari AM, Shubrook JH. Treatment options and current guidelines of care for pediatric type 2 diabetes patients: a narrative review. J Osteopath Med. 2021;121(4):431-440. doi:10.1515/jom-2020-0172












