User login
Optimizing treatment selection in MS often tricky
INDIANAPOLIS – Consider disease activity, patient comorbidities, treatment tolerability, and patient or physician preference when prescribing disease-modifying therapy for multiple sclerosis patients.
“Where I practice, patient preference can sound something like, ‘My sister-in-law’s cousin’s friend at church takes drug X, and that’s the one I’m interested in,’ ” said Dr. Myla D. Goldman, a neurologist who directs the James Q. Miller Multiple Sclerosis Clinic at the University of Virginia, Charlottesville. “It’s sometimes guided by just familiarity or word of mouth, or something that they’ve read or someone that they know. In terms of our own preferences as clinicians, those are much harder to characterize. I think they become shaped over time and our own experiences, as well as being guided by evidence-based medicine.”
In a survey, 102 neurologists were asked about how they made decisions regarding the prescription of disease-modifying therapies (DMTs) for patients with MS (Patient Prefer. Adherence 2014;8:415-22). More than 80% ranked efficacy as the most important attribute of DMT medication selection, followed by safety, tolerability, patient preference, and convenience (including dosing frequency and administration method). In a separate study presented at the 2014 meeting of the American Academy of Neurology, 1,628 MS patients in Germany were asked about the importance of different DMT features (Neurology 2014;82[10 Suppl.]:P3.137). They ranked oral administration as the most important attribute in guiding their DMT preference, while the second most relevant attribute was frequency of administration.
In a more recent study, 50 patients with relapsing-remitting MS were asked about their preferences for attributes of DMTs (Int. J. MS Care 2015;17:74-82). Survey respondents showed the highest preference for medications that would improve their symptoms, which is not a proven DMT outcome. Preventing relapses, which is a key clinical trial outcome, was not associated with significant preferences.
“This is important to know but adds another layer of challenge and complication [because] what we understand most about many of these agents is their effect on relapse rates,” Dr. Goldman said at the annual meeting of the Consortium of Multiple Sclerosis Centers. “It’s another disconnect in terms of what we’re thinking about, the tools that we have, and where our patients are coming from.”
Other factors to consider when selecting a DMT include disease activity/burden, risk/prognostic factors, treatment safety/tolerability, comorbidity, and treatment access/logistics. “As we’ve had more and more [DMT] choices available to us, we’ve become more restricted in how we make choices – which patients can have what choices in what order,” she said. “This is a reality that we all have to face but is something that varies from third-party payers and region, and can be really complicated to navigate.”
In general, Dr. Goldman continued, it’s recognized that more active disease requires more effective therapies with greater tolerance for risk. However, evidence suggesting that early disease activity predicts long-term outcome is poor, “not because it may not be true but because we don’t have as robust data as we would like,” she said. “We also have few [clinical] trials focused on this group. So when we look at high disease activity, it’s typically in a subgroup analysis. There are risks for underpowering and a lack of direct comparisons among therapies. I have found that my idea of disease activity and my individual patient’s idea of disease activity sometimes differ. Perhaps I think we need to make a change, and they feel comfortable with what they’re taking, or vice versa.”
There are various predictors of disease activity and outcomes based on early signs and symptoms, “but these outcomes are variable when we are comparing across studies,” she noted. These predictors and outcomes include:
• Early disease activity predicts relapse disease activity early, especially with posterior fossa lesions and enhancing lesions.
• Early MRI activity predicts long-term disability.
• A baseline T2 lesion burden predicts change in the Kurtzke Expanded Disability Status Scale (EDSS) and progression to secondary-progressive MS.
• A T2 lesion change at 1 year predicts EDSS change.
• Brain atrophy over 2 years predicts EDSS at 8 years.
Favorable prognostic factors include monofocal onset, onset with optic neuritis or isolated sensory symptoms, younger age at onset, being white, being female, having a low relapse rate in the first 2-5 years, and a long interval between the first and second relapse. “That’s where early treatment and impact on relapse rate has a really important role,” she said.
Unfavorable factors in relapsing-remitting MS include multifocal onset, being black, older age at onset, being male, high relapse rate in the first 2-5 years, disability at 5 years, cognitive impairment, high MRI lesion load at baseline, atrophy and T1 holes on MRI, and being a smoker.
Dr. Goldman pointed out that efficacy, safety, and tolerability of most DMTs are unknown in patients with comorbidities such as obesity or cardiovascular disease. “It’s important to remember that for many patients with comorbidity, these are exclusion criteria in the clinical trials program,” she said. “So while we know that having comorbidities in an MS population does affect quality of life and increase disability progression, what we are lacking understanding [about] is how these may affect treatment response.”
Dr. Goldman disclosed that she is a consultant and advisory board member for Questcor Pharmaceuticals.
The session was sponsored by an educational grant from Biogen.
On Twitter @dougbrunk
INDIANAPOLIS – Consider disease activity, patient comorbidities, treatment tolerability, and patient or physician preference when prescribing disease-modifying therapy for multiple sclerosis patients.
“Where I practice, patient preference can sound something like, ‘My sister-in-law’s cousin’s friend at church takes drug X, and that’s the one I’m interested in,’ ” said Dr. Myla D. Goldman, a neurologist who directs the James Q. Miller Multiple Sclerosis Clinic at the University of Virginia, Charlottesville. “It’s sometimes guided by just familiarity or word of mouth, or something that they’ve read or someone that they know. In terms of our own preferences as clinicians, those are much harder to characterize. I think they become shaped over time and our own experiences, as well as being guided by evidence-based medicine.”
In a survey, 102 neurologists were asked about how they made decisions regarding the prescription of disease-modifying therapies (DMTs) for patients with MS (Patient Prefer. Adherence 2014;8:415-22). More than 80% ranked efficacy as the most important attribute of DMT medication selection, followed by safety, tolerability, patient preference, and convenience (including dosing frequency and administration method). In a separate study presented at the 2014 meeting of the American Academy of Neurology, 1,628 MS patients in Germany were asked about the importance of different DMT features (Neurology 2014;82[10 Suppl.]:P3.137). They ranked oral administration as the most important attribute in guiding their DMT preference, while the second most relevant attribute was frequency of administration.
In a more recent study, 50 patients with relapsing-remitting MS were asked about their preferences for attributes of DMTs (Int. J. MS Care 2015;17:74-82). Survey respondents showed the highest preference for medications that would improve their symptoms, which is not a proven DMT outcome. Preventing relapses, which is a key clinical trial outcome, was not associated with significant preferences.
“This is important to know but adds another layer of challenge and complication [because] what we understand most about many of these agents is their effect on relapse rates,” Dr. Goldman said at the annual meeting of the Consortium of Multiple Sclerosis Centers. “It’s another disconnect in terms of what we’re thinking about, the tools that we have, and where our patients are coming from.”
Other factors to consider when selecting a DMT include disease activity/burden, risk/prognostic factors, treatment safety/tolerability, comorbidity, and treatment access/logistics. “As we’ve had more and more [DMT] choices available to us, we’ve become more restricted in how we make choices – which patients can have what choices in what order,” she said. “This is a reality that we all have to face but is something that varies from third-party payers and region, and can be really complicated to navigate.”
In general, Dr. Goldman continued, it’s recognized that more active disease requires more effective therapies with greater tolerance for risk. However, evidence suggesting that early disease activity predicts long-term outcome is poor, “not because it may not be true but because we don’t have as robust data as we would like,” she said. “We also have few [clinical] trials focused on this group. So when we look at high disease activity, it’s typically in a subgroup analysis. There are risks for underpowering and a lack of direct comparisons among therapies. I have found that my idea of disease activity and my individual patient’s idea of disease activity sometimes differ. Perhaps I think we need to make a change, and they feel comfortable with what they’re taking, or vice versa.”
There are various predictors of disease activity and outcomes based on early signs and symptoms, “but these outcomes are variable when we are comparing across studies,” she noted. These predictors and outcomes include:
• Early disease activity predicts relapse disease activity early, especially with posterior fossa lesions and enhancing lesions.
• Early MRI activity predicts long-term disability.
• A baseline T2 lesion burden predicts change in the Kurtzke Expanded Disability Status Scale (EDSS) and progression to secondary-progressive MS.
• A T2 lesion change at 1 year predicts EDSS change.
• Brain atrophy over 2 years predicts EDSS at 8 years.
Favorable prognostic factors include monofocal onset, onset with optic neuritis or isolated sensory symptoms, younger age at onset, being white, being female, having a low relapse rate in the first 2-5 years, and a long interval between the first and second relapse. “That’s where early treatment and impact on relapse rate has a really important role,” she said.
Unfavorable factors in relapsing-remitting MS include multifocal onset, being black, older age at onset, being male, high relapse rate in the first 2-5 years, disability at 5 years, cognitive impairment, high MRI lesion load at baseline, atrophy and T1 holes on MRI, and being a smoker.
Dr. Goldman pointed out that efficacy, safety, and tolerability of most DMTs are unknown in patients with comorbidities such as obesity or cardiovascular disease. “It’s important to remember that for many patients with comorbidity, these are exclusion criteria in the clinical trials program,” she said. “So while we know that having comorbidities in an MS population does affect quality of life and increase disability progression, what we are lacking understanding [about] is how these may affect treatment response.”
Dr. Goldman disclosed that she is a consultant and advisory board member for Questcor Pharmaceuticals.
The session was sponsored by an educational grant from Biogen.
On Twitter @dougbrunk
INDIANAPOLIS – Consider disease activity, patient comorbidities, treatment tolerability, and patient or physician preference when prescribing disease-modifying therapy for multiple sclerosis patients.
“Where I practice, patient preference can sound something like, ‘My sister-in-law’s cousin’s friend at church takes drug X, and that’s the one I’m interested in,’ ” said Dr. Myla D. Goldman, a neurologist who directs the James Q. Miller Multiple Sclerosis Clinic at the University of Virginia, Charlottesville. “It’s sometimes guided by just familiarity or word of mouth, or something that they’ve read or someone that they know. In terms of our own preferences as clinicians, those are much harder to characterize. I think they become shaped over time and our own experiences, as well as being guided by evidence-based medicine.”
In a survey, 102 neurologists were asked about how they made decisions regarding the prescription of disease-modifying therapies (DMTs) for patients with MS (Patient Prefer. Adherence 2014;8:415-22). More than 80% ranked efficacy as the most important attribute of DMT medication selection, followed by safety, tolerability, patient preference, and convenience (including dosing frequency and administration method). In a separate study presented at the 2014 meeting of the American Academy of Neurology, 1,628 MS patients in Germany were asked about the importance of different DMT features (Neurology 2014;82[10 Suppl.]:P3.137). They ranked oral administration as the most important attribute in guiding their DMT preference, while the second most relevant attribute was frequency of administration.
In a more recent study, 50 patients with relapsing-remitting MS were asked about their preferences for attributes of DMTs (Int. J. MS Care 2015;17:74-82). Survey respondents showed the highest preference for medications that would improve their symptoms, which is not a proven DMT outcome. Preventing relapses, which is a key clinical trial outcome, was not associated with significant preferences.
“This is important to know but adds another layer of challenge and complication [because] what we understand most about many of these agents is their effect on relapse rates,” Dr. Goldman said at the annual meeting of the Consortium of Multiple Sclerosis Centers. “It’s another disconnect in terms of what we’re thinking about, the tools that we have, and where our patients are coming from.”
Other factors to consider when selecting a DMT include disease activity/burden, risk/prognostic factors, treatment safety/tolerability, comorbidity, and treatment access/logistics. “As we’ve had more and more [DMT] choices available to us, we’ve become more restricted in how we make choices – which patients can have what choices in what order,” she said. “This is a reality that we all have to face but is something that varies from third-party payers and region, and can be really complicated to navigate.”
In general, Dr. Goldman continued, it’s recognized that more active disease requires more effective therapies with greater tolerance for risk. However, evidence suggesting that early disease activity predicts long-term outcome is poor, “not because it may not be true but because we don’t have as robust data as we would like,” she said. “We also have few [clinical] trials focused on this group. So when we look at high disease activity, it’s typically in a subgroup analysis. There are risks for underpowering and a lack of direct comparisons among therapies. I have found that my idea of disease activity and my individual patient’s idea of disease activity sometimes differ. Perhaps I think we need to make a change, and they feel comfortable with what they’re taking, or vice versa.”
There are various predictors of disease activity and outcomes based on early signs and symptoms, “but these outcomes are variable when we are comparing across studies,” she noted. These predictors and outcomes include:
• Early disease activity predicts relapse disease activity early, especially with posterior fossa lesions and enhancing lesions.
• Early MRI activity predicts long-term disability.
• A baseline T2 lesion burden predicts change in the Kurtzke Expanded Disability Status Scale (EDSS) and progression to secondary-progressive MS.
• A T2 lesion change at 1 year predicts EDSS change.
• Brain atrophy over 2 years predicts EDSS at 8 years.
Favorable prognostic factors include monofocal onset, onset with optic neuritis or isolated sensory symptoms, younger age at onset, being white, being female, having a low relapse rate in the first 2-5 years, and a long interval between the first and second relapse. “That’s where early treatment and impact on relapse rate has a really important role,” she said.
Unfavorable factors in relapsing-remitting MS include multifocal onset, being black, older age at onset, being male, high relapse rate in the first 2-5 years, disability at 5 years, cognitive impairment, high MRI lesion load at baseline, atrophy and T1 holes on MRI, and being a smoker.
Dr. Goldman pointed out that efficacy, safety, and tolerability of most DMTs are unknown in patients with comorbidities such as obesity or cardiovascular disease. “It’s important to remember that for many patients with comorbidity, these are exclusion criteria in the clinical trials program,” she said. “So while we know that having comorbidities in an MS population does affect quality of life and increase disability progression, what we are lacking understanding [about] is how these may affect treatment response.”
Dr. Goldman disclosed that she is a consultant and advisory board member for Questcor Pharmaceuticals.
The session was sponsored by an educational grant from Biogen.
On Twitter @dougbrunk
EXPERT ANALYSIS FROM THE CMSC ANNUAL MEETING
Adherence to MS treatments lower in oral vs. injectable drugs
INDIANAPOLIS – Lack of adherence was significantly higher in multiple sclerosis (MS) patients who received oral versus injectable disease-modifying therapies, a single-center study showed.
Side effect profile may have been a contributing factor to this outcome, study author Caitlin Dionne said in an interview during a poster session at the annual meeting of the Consortium of Multiple Sclerosis Centers. “There are more GI-related side effects with the oral MS drugs, compared with the injectables, so that could be part of it,” said Ms. Dionne, a registered nurse in the neurology department at the Lahey Clinic Outpatient Center, Lexington, Mass. In addition, taking an oral DMT “may not be as serious in the minds [of patients] as ‘I have to take a shot.’”
In an effort to determine if adherence and tolerability of oral disease-modifying therapies (DMTs) is better than with injectables, Ms. Dionne and her associates developed the MS Treatment Adherence Questionnaire (MS-TAQ) and collected data from October and November of 2014. The questionnaire is composed of six questions related to a patient’s current DMT: number of missed doses in 4 weeks, reason dose was missed, perceived side effects, ease of administration, and medication satisfaction. Medication types were divided into three groups: oral DMTs, subcutaneous or intramuscular (SC/IM) DMTs, and monthly IV injections.
Of the 209 patients surveyed, 75% were female and their mean age was 50. A similar number of patients were on oral and SC/IM medications (89 and 90, respectively), while the remaining 30 were on an IV infusion. Ms. Dionne reported that 55% of patients taking oral medications reported missing no doses, compared with 71% of those taking SC/IM agents and 93% of those receiving IV infusions (P = .005). Ease of medication administration was reported by 77% of patients taking oral medications, 60% of those taking SC/IM agents, and 33% of those receiving IV infusions (P < .0001).
Reasons for missing a dose differed with respect to each DMT type (P = .017), with forgetfulness being reported in 38% and 27% of patients taking oral and SC/IM agents, respectively. Side effects also differed with respect to each DMT. Nearly three-quarters (74%) of those receiving IV infusions did not experience side effects, compared with 19% of those taking oral medications and 20% of those taking SC/IM agents.
The researchers had no relevant financial conflicts to disclose.
On Twitter @dougbrunk
INDIANAPOLIS – Lack of adherence was significantly higher in multiple sclerosis (MS) patients who received oral versus injectable disease-modifying therapies, a single-center study showed.
Side effect profile may have been a contributing factor to this outcome, study author Caitlin Dionne said in an interview during a poster session at the annual meeting of the Consortium of Multiple Sclerosis Centers. “There are more GI-related side effects with the oral MS drugs, compared with the injectables, so that could be part of it,” said Ms. Dionne, a registered nurse in the neurology department at the Lahey Clinic Outpatient Center, Lexington, Mass. In addition, taking an oral DMT “may not be as serious in the minds [of patients] as ‘I have to take a shot.’”
In an effort to determine if adherence and tolerability of oral disease-modifying therapies (DMTs) is better than with injectables, Ms. Dionne and her associates developed the MS Treatment Adherence Questionnaire (MS-TAQ) and collected data from October and November of 2014. The questionnaire is composed of six questions related to a patient’s current DMT: number of missed doses in 4 weeks, reason dose was missed, perceived side effects, ease of administration, and medication satisfaction. Medication types were divided into three groups: oral DMTs, subcutaneous or intramuscular (SC/IM) DMTs, and monthly IV injections.
Of the 209 patients surveyed, 75% were female and their mean age was 50. A similar number of patients were on oral and SC/IM medications (89 and 90, respectively), while the remaining 30 were on an IV infusion. Ms. Dionne reported that 55% of patients taking oral medications reported missing no doses, compared with 71% of those taking SC/IM agents and 93% of those receiving IV infusions (P = .005). Ease of medication administration was reported by 77% of patients taking oral medications, 60% of those taking SC/IM agents, and 33% of those receiving IV infusions (P < .0001).
Reasons for missing a dose differed with respect to each DMT type (P = .017), with forgetfulness being reported in 38% and 27% of patients taking oral and SC/IM agents, respectively. Side effects also differed with respect to each DMT. Nearly three-quarters (74%) of those receiving IV infusions did not experience side effects, compared with 19% of those taking oral medications and 20% of those taking SC/IM agents.
The researchers had no relevant financial conflicts to disclose.
On Twitter @dougbrunk
INDIANAPOLIS – Lack of adherence was significantly higher in multiple sclerosis (MS) patients who received oral versus injectable disease-modifying therapies, a single-center study showed.
Side effect profile may have been a contributing factor to this outcome, study author Caitlin Dionne said in an interview during a poster session at the annual meeting of the Consortium of Multiple Sclerosis Centers. “There are more GI-related side effects with the oral MS drugs, compared with the injectables, so that could be part of it,” said Ms. Dionne, a registered nurse in the neurology department at the Lahey Clinic Outpatient Center, Lexington, Mass. In addition, taking an oral DMT “may not be as serious in the minds [of patients] as ‘I have to take a shot.’”
In an effort to determine if adherence and tolerability of oral disease-modifying therapies (DMTs) is better than with injectables, Ms. Dionne and her associates developed the MS Treatment Adherence Questionnaire (MS-TAQ) and collected data from October and November of 2014. The questionnaire is composed of six questions related to a patient’s current DMT: number of missed doses in 4 weeks, reason dose was missed, perceived side effects, ease of administration, and medication satisfaction. Medication types were divided into three groups: oral DMTs, subcutaneous or intramuscular (SC/IM) DMTs, and monthly IV injections.
Of the 209 patients surveyed, 75% were female and their mean age was 50. A similar number of patients were on oral and SC/IM medications (89 and 90, respectively), while the remaining 30 were on an IV infusion. Ms. Dionne reported that 55% of patients taking oral medications reported missing no doses, compared with 71% of those taking SC/IM agents and 93% of those receiving IV infusions (P = .005). Ease of medication administration was reported by 77% of patients taking oral medications, 60% of those taking SC/IM agents, and 33% of those receiving IV infusions (P < .0001).
Reasons for missing a dose differed with respect to each DMT type (P = .017), with forgetfulness being reported in 38% and 27% of patients taking oral and SC/IM agents, respectively. Side effects also differed with respect to each DMT. Nearly three-quarters (74%) of those receiving IV infusions did not experience side effects, compared with 19% of those taking oral medications and 20% of those taking SC/IM agents.
The researchers had no relevant financial conflicts to disclose.
On Twitter @dougbrunk
AT THE CMSC ANNUAL MEETING
Key clinical point: MS patients were less adherent to oral than to injectable disease-modifying therapies.
Major finding: Just over half of MS patients taking oral medications (55%) reported missing no doses, compared with 71% of those taking SC/IM agents and 93% of those receiving IV infusions (P = .005).
Data source: Survey responses from 209 MS patients at the Lahey Outpatient Center, Lexington, Mass.
Disclosures: The researchers had no relevant financial conflicts to disclose.
Only moderate-quality evidence supports medical cannabinoids
At best, only moderate-quality evidence supports the use of medical cannabinoids, and for only two conditions. And low-quality evidence only “suggests” that these agents may improve other medical conditions, but that limited effectiveness applies only to the four conditions that have been studied, according to a report published online June 23 in JAMA.
This doesn’t necessarily mean that cannabinoids have poor efficacy but instead likely reflects the dearth of high-quality research into their medical usefulness.
In what they described as the first comprehensive review to evaluate the efficacy of numerous cannabinoids across a broad range of indications, researchers analyzed data from 79 studies involving 6,462 participants performed from 1975 to early 2015. The studies assessed nabilone, dronabinol, nabiximols, levonantradol, THC, THC/CBD, and ajuvenic acid, delivered via oral capsules, cigarettes, vaporizers, oromucosal sprays, or intramuscular injection, said Penny F. Whiting, Ph.D., of University Hospitals Bristol (England) and her associates.
Most of the studies suggested that cannabinoids improved symptoms for nearly all the 10 medical conditions included in this meta-analysis, but most of the studies were of poor quality so their conclusions were questionable. These agents’ efficacy did not vary according to the type of cannabinoid assessed or the mode of delivery.
Moderate-quality evidence indicated that cannabinoids may be beneficial for chronic neuropathic or cancer pain, and moderate-quality evidence indicated that they may also be beneficial for spasticity due to multiple sclerosis or traumatic paraplegia. Low-quality evidence suggested that cannabinoids may improve nausea and vomiting due to chemotherapy, may improve appetite and induce weight gain in HIV infection, and may improve sleep in primary sleep disorders as well as in conditions that disrupt sleep such as fibromyalgia, multiple sclerosis, and chronic pain. Very low-quality evidence (due to extremely small sample sizes) suggested that cannabinoids may greatly improve tic severity in Tourette’s syndrome, Dr. Whiting and her associates said (JAMA 2015 June 23 [doi:10.1001/jama.2015.6358]).
Otherwise, the evidence did not support cannabinoids’ efficacy in anxiety, depression, psychosis, or glaucoma. Adverse events included asthenia, problems with balance, confusion, dizziness, disorientation, dry mouth, fatigue, and somnolence.
It is unclear why medical marijuana has averted the usual Food and Drug Administration approval process required of other medications.
Adequately powered, double-blind, randomized, placebo- or treatment-controlled clinical trials are critical to establish cannabinoids’ short- and long-term efficacy and safety. Yet they already qualify by state law for use in conditions as varied as hepatitis C, Crohn’s disease, Parkinson’s disease, psoriasis, sickle cell disease, and posttraumatic stress disorder.
In particular, the risks of repeated exposure to cannabinoids needs further study. Addiction, tolerance, and a distinct withdrawal syndrome have been documented, and there is also a small but definite risk of psychotic disorder.
Dr. Deepak Cyril D’Souza and Dr. Mohini Ranganathan are in the department of psychiatry at Yale University, New Haven; in the psychiatry service at Veterans Affairs Connecticut Healthcare System, West Haven; and at Abraham Ribicoff Research Facilities at the Connecticut Mental Health Center, New Haven. Dr. D’Souza reported receiving grants from AbbVie and Pfizer, and serves on the Connecticut Board of Physicians that advises the Commissioner of Consumer Protection about the Act Concerning the Palliative Use of Marijuana. Dr. Ranganathan reported receiving grants from Insys Therapeutics. Dr. D’Souza and Dr. Ranganathan made these remarks in an editorial accompanying Dr. Whiting’s report (JAMA 2015;313:2431-2).
It is unclear why medical marijuana has averted the usual Food and Drug Administration approval process required of other medications.
Adequately powered, double-blind, randomized, placebo- or treatment-controlled clinical trials are critical to establish cannabinoids’ short- and long-term efficacy and safety. Yet they already qualify by state law for use in conditions as varied as hepatitis C, Crohn’s disease, Parkinson’s disease, psoriasis, sickle cell disease, and posttraumatic stress disorder.
In particular, the risks of repeated exposure to cannabinoids needs further study. Addiction, tolerance, and a distinct withdrawal syndrome have been documented, and there is also a small but definite risk of psychotic disorder.
Dr. Deepak Cyril D’Souza and Dr. Mohini Ranganathan are in the department of psychiatry at Yale University, New Haven; in the psychiatry service at Veterans Affairs Connecticut Healthcare System, West Haven; and at Abraham Ribicoff Research Facilities at the Connecticut Mental Health Center, New Haven. Dr. D’Souza reported receiving grants from AbbVie and Pfizer, and serves on the Connecticut Board of Physicians that advises the Commissioner of Consumer Protection about the Act Concerning the Palliative Use of Marijuana. Dr. Ranganathan reported receiving grants from Insys Therapeutics. Dr. D’Souza and Dr. Ranganathan made these remarks in an editorial accompanying Dr. Whiting’s report (JAMA 2015;313:2431-2).
It is unclear why medical marijuana has averted the usual Food and Drug Administration approval process required of other medications.
Adequately powered, double-blind, randomized, placebo- or treatment-controlled clinical trials are critical to establish cannabinoids’ short- and long-term efficacy and safety. Yet they already qualify by state law for use in conditions as varied as hepatitis C, Crohn’s disease, Parkinson’s disease, psoriasis, sickle cell disease, and posttraumatic stress disorder.
In particular, the risks of repeated exposure to cannabinoids needs further study. Addiction, tolerance, and a distinct withdrawal syndrome have been documented, and there is also a small but definite risk of psychotic disorder.
Dr. Deepak Cyril D’Souza and Dr. Mohini Ranganathan are in the department of psychiatry at Yale University, New Haven; in the psychiatry service at Veterans Affairs Connecticut Healthcare System, West Haven; and at Abraham Ribicoff Research Facilities at the Connecticut Mental Health Center, New Haven. Dr. D’Souza reported receiving grants from AbbVie and Pfizer, and serves on the Connecticut Board of Physicians that advises the Commissioner of Consumer Protection about the Act Concerning the Palliative Use of Marijuana. Dr. Ranganathan reported receiving grants from Insys Therapeutics. Dr. D’Souza and Dr. Ranganathan made these remarks in an editorial accompanying Dr. Whiting’s report (JAMA 2015;313:2431-2).
At best, only moderate-quality evidence supports the use of medical cannabinoids, and for only two conditions. And low-quality evidence only “suggests” that these agents may improve other medical conditions, but that limited effectiveness applies only to the four conditions that have been studied, according to a report published online June 23 in JAMA.
This doesn’t necessarily mean that cannabinoids have poor efficacy but instead likely reflects the dearth of high-quality research into their medical usefulness.
In what they described as the first comprehensive review to evaluate the efficacy of numerous cannabinoids across a broad range of indications, researchers analyzed data from 79 studies involving 6,462 participants performed from 1975 to early 2015. The studies assessed nabilone, dronabinol, nabiximols, levonantradol, THC, THC/CBD, and ajuvenic acid, delivered via oral capsules, cigarettes, vaporizers, oromucosal sprays, or intramuscular injection, said Penny F. Whiting, Ph.D., of University Hospitals Bristol (England) and her associates.
Most of the studies suggested that cannabinoids improved symptoms for nearly all the 10 medical conditions included in this meta-analysis, but most of the studies were of poor quality so their conclusions were questionable. These agents’ efficacy did not vary according to the type of cannabinoid assessed or the mode of delivery.
Moderate-quality evidence indicated that cannabinoids may be beneficial for chronic neuropathic or cancer pain, and moderate-quality evidence indicated that they may also be beneficial for spasticity due to multiple sclerosis or traumatic paraplegia. Low-quality evidence suggested that cannabinoids may improve nausea and vomiting due to chemotherapy, may improve appetite and induce weight gain in HIV infection, and may improve sleep in primary sleep disorders as well as in conditions that disrupt sleep such as fibromyalgia, multiple sclerosis, and chronic pain. Very low-quality evidence (due to extremely small sample sizes) suggested that cannabinoids may greatly improve tic severity in Tourette’s syndrome, Dr. Whiting and her associates said (JAMA 2015 June 23 [doi:10.1001/jama.2015.6358]).
Otherwise, the evidence did not support cannabinoids’ efficacy in anxiety, depression, psychosis, or glaucoma. Adverse events included asthenia, problems with balance, confusion, dizziness, disorientation, dry mouth, fatigue, and somnolence.
At best, only moderate-quality evidence supports the use of medical cannabinoids, and for only two conditions. And low-quality evidence only “suggests” that these agents may improve other medical conditions, but that limited effectiveness applies only to the four conditions that have been studied, according to a report published online June 23 in JAMA.
This doesn’t necessarily mean that cannabinoids have poor efficacy but instead likely reflects the dearth of high-quality research into their medical usefulness.
In what they described as the first comprehensive review to evaluate the efficacy of numerous cannabinoids across a broad range of indications, researchers analyzed data from 79 studies involving 6,462 participants performed from 1975 to early 2015. The studies assessed nabilone, dronabinol, nabiximols, levonantradol, THC, THC/CBD, and ajuvenic acid, delivered via oral capsules, cigarettes, vaporizers, oromucosal sprays, or intramuscular injection, said Penny F. Whiting, Ph.D., of University Hospitals Bristol (England) and her associates.
Most of the studies suggested that cannabinoids improved symptoms for nearly all the 10 medical conditions included in this meta-analysis, but most of the studies were of poor quality so their conclusions were questionable. These agents’ efficacy did not vary according to the type of cannabinoid assessed or the mode of delivery.
Moderate-quality evidence indicated that cannabinoids may be beneficial for chronic neuropathic or cancer pain, and moderate-quality evidence indicated that they may also be beneficial for spasticity due to multiple sclerosis or traumatic paraplegia. Low-quality evidence suggested that cannabinoids may improve nausea and vomiting due to chemotherapy, may improve appetite and induce weight gain in HIV infection, and may improve sleep in primary sleep disorders as well as in conditions that disrupt sleep such as fibromyalgia, multiple sclerosis, and chronic pain. Very low-quality evidence (due to extremely small sample sizes) suggested that cannabinoids may greatly improve tic severity in Tourette’s syndrome, Dr. Whiting and her associates said (JAMA 2015 June 23 [doi:10.1001/jama.2015.6358]).
Otherwise, the evidence did not support cannabinoids’ efficacy in anxiety, depression, psychosis, or glaucoma. Adverse events included asthenia, problems with balance, confusion, dizziness, disorientation, dry mouth, fatigue, and somnolence.
FROM JAMA
Key clinical point: Only moderate-quality evidence supports the use of medical cannabinoids, and only for two conditions.
Major finding: Moderate-quality evidence indicated that cannabinoids may be beneficial for chronic neuropathic or cancer pain and for spasticity due to multiple sclerosis or traumatic paraplegia.
Data source: A comprehensive review of the literature since 1975 and a meta-analysis of 79 clinical trials involving 6,462 participants.
Disclosures: This study was funded by the Swiss Federal Office of Public Health. Dr. Whiting and her associates reported having no relevant financial conflicts of interest.
Can MS Progression be Predicted?
In patients with multiple sclerosis (MS), disease progression and disability endpoints can be predicted by time to secondary progression, according to a 50-year follow up of an incidence cohort of 254 MS patients.
The study followed 212 adults with initial relapsing-remitting MS and 42 patients with a monophasic disease course and found:
• Median time to secondary progression was 15 years.
• Median times to Expanded Disability Status Scale 6 (EDSS6) and EDSS7 were 26 and 48 years, respectively.
• The cumulative risk of reaching EDSS6 was 50% at 55 years and 80% at 80 years.
• Age at onset predicted the disease course in men, with a 3 to 6% yearly increase in the risk of reaching disability milestones.
Citation: Tedeholm H, Skoog B, Lisovskaja V, Runmarker B, Nerman O, Andersen O. The outcome spectrum of multiple sclerosis: disability, mortality, and a cluster of predictors from onset. J Neurol. 2015;262(5):1148-1163. doi: 10.1007/s00415-015-7674-y.
Commentary: Long-term cohort information related to disability outcome and natural history of disease can provide long-term milestones to judge therapy efficacy. MS is a chronic disease with expensive therapies and increasing disability over time. A reliance on short-term studies, relapse rates, and physical examination remains a recipe for economic disaster. A cumulative risk of half of all patients reaching EDSS6 at age 55 is sobering and reflects the fact that therapy initiation at diagnosis, long-term treatment, and maintaining effective interventions to prevent accumulation of disability are key. Perhaps the long-term benchmark of disease stability with therapy intervention might justify early expenditure. More objective information is needed about disability milestones and reliable predictors of disease change at critical times to justify therapy efficacy for prevention of long-term disability. A sobering fact that remains is that this disease, despite a greater appearance of benign cases, can still be severe in the long term for many people. — Mark Gudesblatt, MD, Medical Director of the Comprehensive MS Care Center at South Shore Neurologic Associates in Islip, NY
In patients with multiple sclerosis (MS), disease progression and disability endpoints can be predicted by time to secondary progression, according to a 50-year follow up of an incidence cohort of 254 MS patients.
The study followed 212 adults with initial relapsing-remitting MS and 42 patients with a monophasic disease course and found:
• Median time to secondary progression was 15 years.
• Median times to Expanded Disability Status Scale 6 (EDSS6) and EDSS7 were 26 and 48 years, respectively.
• The cumulative risk of reaching EDSS6 was 50% at 55 years and 80% at 80 years.
• Age at onset predicted the disease course in men, with a 3 to 6% yearly increase in the risk of reaching disability milestones.
Citation: Tedeholm H, Skoog B, Lisovskaja V, Runmarker B, Nerman O, Andersen O. The outcome spectrum of multiple sclerosis: disability, mortality, and a cluster of predictors from onset. J Neurol. 2015;262(5):1148-1163. doi: 10.1007/s00415-015-7674-y.
Commentary: Long-term cohort information related to disability outcome and natural history of disease can provide long-term milestones to judge therapy efficacy. MS is a chronic disease with expensive therapies and increasing disability over time. A reliance on short-term studies, relapse rates, and physical examination remains a recipe for economic disaster. A cumulative risk of half of all patients reaching EDSS6 at age 55 is sobering and reflects the fact that therapy initiation at diagnosis, long-term treatment, and maintaining effective interventions to prevent accumulation of disability are key. Perhaps the long-term benchmark of disease stability with therapy intervention might justify early expenditure. More objective information is needed about disability milestones and reliable predictors of disease change at critical times to justify therapy efficacy for prevention of long-term disability. A sobering fact that remains is that this disease, despite a greater appearance of benign cases, can still be severe in the long term for many people. — Mark Gudesblatt, MD, Medical Director of the Comprehensive MS Care Center at South Shore Neurologic Associates in Islip, NY
In patients with multiple sclerosis (MS), disease progression and disability endpoints can be predicted by time to secondary progression, according to a 50-year follow up of an incidence cohort of 254 MS patients.
The study followed 212 adults with initial relapsing-remitting MS and 42 patients with a monophasic disease course and found:
• Median time to secondary progression was 15 years.
• Median times to Expanded Disability Status Scale 6 (EDSS6) and EDSS7 were 26 and 48 years, respectively.
• The cumulative risk of reaching EDSS6 was 50% at 55 years and 80% at 80 years.
• Age at onset predicted the disease course in men, with a 3 to 6% yearly increase in the risk of reaching disability milestones.
Citation: Tedeholm H, Skoog B, Lisovskaja V, Runmarker B, Nerman O, Andersen O. The outcome spectrum of multiple sclerosis: disability, mortality, and a cluster of predictors from onset. J Neurol. 2015;262(5):1148-1163. doi: 10.1007/s00415-015-7674-y.
Commentary: Long-term cohort information related to disability outcome and natural history of disease can provide long-term milestones to judge therapy efficacy. MS is a chronic disease with expensive therapies and increasing disability over time. A reliance on short-term studies, relapse rates, and physical examination remains a recipe for economic disaster. A cumulative risk of half of all patients reaching EDSS6 at age 55 is sobering and reflects the fact that therapy initiation at diagnosis, long-term treatment, and maintaining effective interventions to prevent accumulation of disability are key. Perhaps the long-term benchmark of disease stability with therapy intervention might justify early expenditure. More objective information is needed about disability milestones and reliable predictors of disease change at critical times to justify therapy efficacy for prevention of long-term disability. A sobering fact that remains is that this disease, despite a greater appearance of benign cases, can still be severe in the long term for many people. — Mark Gudesblatt, MD, Medical Director of the Comprehensive MS Care Center at South Shore Neurologic Associates in Islip, NY
Depression, Anxiety, and Multiple Sclerosis
Patients with multiple sclerosis are at increased risk of depression and anxiety diagnoses as well as antidepressant and anxiolytic drug use both before and after diagnosis, according to a matched cohort study of 5,084 patients and 24,771 controls from a nationwide sample.
Researchers used logistic regression to estimate odds ratios for pre- and post-MS diagnosis rates of depression and anxiety, and redemption of prescriptions for tricyclic antidepressants (TCAs) and selective serotonin reuptake inhibitors (SSRIs). They found:
|
| Pre-MS diagnosis | Post-MS diagnosis |
| Depression/anxiety diagnosis | 1.4 | 1.23 |
| TCAs | 1.9 | 6.70 |
| SSRIs | 1.34 | 2.41 |
Citation: Hoang H, Laursen B, Stenager EN, Stenager E. Psychiatric co-morbidity in multiple sclerosis: the risk of depression and anxiety before and after MS diagnosis. Mult Scler. 2015. pii:1352458515588973. [Epub ahead of print]
Patients with multiple sclerosis are at increased risk of depression and anxiety diagnoses as well as antidepressant and anxiolytic drug use both before and after diagnosis, according to a matched cohort study of 5,084 patients and 24,771 controls from a nationwide sample.
Researchers used logistic regression to estimate odds ratios for pre- and post-MS diagnosis rates of depression and anxiety, and redemption of prescriptions for tricyclic antidepressants (TCAs) and selective serotonin reuptake inhibitors (SSRIs). They found:
|
| Pre-MS diagnosis | Post-MS diagnosis |
| Depression/anxiety diagnosis | 1.4 | 1.23 |
| TCAs | 1.9 | 6.70 |
| SSRIs | 1.34 | 2.41 |
Citation: Hoang H, Laursen B, Stenager EN, Stenager E. Psychiatric co-morbidity in multiple sclerosis: the risk of depression and anxiety before and after MS diagnosis. Mult Scler. 2015. pii:1352458515588973. [Epub ahead of print]
Patients with multiple sclerosis are at increased risk of depression and anxiety diagnoses as well as antidepressant and anxiolytic drug use both before and after diagnosis, according to a matched cohort study of 5,084 patients and 24,771 controls from a nationwide sample.
Researchers used logistic regression to estimate odds ratios for pre- and post-MS diagnosis rates of depression and anxiety, and redemption of prescriptions for tricyclic antidepressants (TCAs) and selective serotonin reuptake inhibitors (SSRIs). They found:
|
| Pre-MS diagnosis | Post-MS diagnosis |
| Depression/anxiety diagnosis | 1.4 | 1.23 |
| TCAs | 1.9 | 6.70 |
| SSRIs | 1.34 | 2.41 |
Citation: Hoang H, Laursen B, Stenager EN, Stenager E. Psychiatric co-morbidity in multiple sclerosis: the risk of depression and anxiety before and after MS diagnosis. Mult Scler. 2015. pii:1352458515588973. [Epub ahead of print]
Information Processing Differs by Age of MS Diagnosis
A younger age at diagnosis of pediatric-onset multiple sclerosis (POMS) is associated with a greater impairment in information processing speeds in adulthood, compared with those with adult onset MS (AOMS), according to data from the comprehensive Longitudinal Investigations in MS at Brigham and Women’s Hospital (CLIMB) study.
Investigators compared 51 POMS and 550 AOMS patients’ scores on Multiple Sclerosis Quality of Life-54 (MSQOL54), Modified Fatigue Impact Scale (MFIS), Center for Epidemiologic Studies Depression Scale (CES-D), and Symbol Digit Modalities Test (SDMT), and found:
• SDMT scores were significantly lower in POMS after adjusting for age, but not after adjusting for disease duration.
• Estimated group difference showed lower normative z scores in POMS than AOMS in unadjusted analysis and after adjusting for disease duration.
• No significant differences in health-related quality-of-life, fatigue, depression, or social support between POMS and AOMS measures.
Citation: Baruch NF, O'Donnell EH, Glanz BI, et al. Cognitive and patient-reported outcomes in adults with pediatric-onset multiple sclerosis. Mult Scler. 2015. pii: 1352458515588781. [Epub ahead of print]
A younger age at diagnosis of pediatric-onset multiple sclerosis (POMS) is associated with a greater impairment in information processing speeds in adulthood, compared with those with adult onset MS (AOMS), according to data from the comprehensive Longitudinal Investigations in MS at Brigham and Women’s Hospital (CLIMB) study.
Investigators compared 51 POMS and 550 AOMS patients’ scores on Multiple Sclerosis Quality of Life-54 (MSQOL54), Modified Fatigue Impact Scale (MFIS), Center for Epidemiologic Studies Depression Scale (CES-D), and Symbol Digit Modalities Test (SDMT), and found:
• SDMT scores were significantly lower in POMS after adjusting for age, but not after adjusting for disease duration.
• Estimated group difference showed lower normative z scores in POMS than AOMS in unadjusted analysis and after adjusting for disease duration.
• No significant differences in health-related quality-of-life, fatigue, depression, or social support between POMS and AOMS measures.
Citation: Baruch NF, O'Donnell EH, Glanz BI, et al. Cognitive and patient-reported outcomes in adults with pediatric-onset multiple sclerosis. Mult Scler. 2015. pii: 1352458515588781. [Epub ahead of print]
A younger age at diagnosis of pediatric-onset multiple sclerosis (POMS) is associated with a greater impairment in information processing speeds in adulthood, compared with those with adult onset MS (AOMS), according to data from the comprehensive Longitudinal Investigations in MS at Brigham and Women’s Hospital (CLIMB) study.
Investigators compared 51 POMS and 550 AOMS patients’ scores on Multiple Sclerosis Quality of Life-54 (MSQOL54), Modified Fatigue Impact Scale (MFIS), Center for Epidemiologic Studies Depression Scale (CES-D), and Symbol Digit Modalities Test (SDMT), and found:
• SDMT scores were significantly lower in POMS after adjusting for age, but not after adjusting for disease duration.
• Estimated group difference showed lower normative z scores in POMS than AOMS in unadjusted analysis and after adjusting for disease duration.
• No significant differences in health-related quality-of-life, fatigue, depression, or social support between POMS and AOMS measures.
Citation: Baruch NF, O'Donnell EH, Glanz BI, et al. Cognitive and patient-reported outcomes in adults with pediatric-onset multiple sclerosis. Mult Scler. 2015. pii: 1352458515588781. [Epub ahead of print]
Weight in Adolescence Correlates to Age of MS Diagnosis
A heavier weight in adolescence is associated with a multiple sclerosis diagnosis at a younger age, according to subgroup cohort of 184 women who provided weight and height information from the age of first menstruation and at age 25.
Investigators calculated BMI and used regression analyses to investigate an association, and found having a higher weight at menarche, weight at age 25, and BMI at age 25 were each significantly related to younger mean age at symptom onset by an average of 5 years:
• overweight: 26.9 years old
• not overweight: 32.1 years old
Citation: Kavak KS, Teter BE, Hagemeier J, Zakalik K, Weinstock-Guttman B; New York State Multiple Sclerosis Consortium. Higher weight in adolescence and young adulthood is associated with an earlier age at multiple sclerosis onset. Mult Scler. 2015;21(7):858-865. doi:10.1177/1352458514555787.
A heavier weight in adolescence is associated with a multiple sclerosis diagnosis at a younger age, according to subgroup cohort of 184 women who provided weight and height information from the age of first menstruation and at age 25.
Investigators calculated BMI and used regression analyses to investigate an association, and found having a higher weight at menarche, weight at age 25, and BMI at age 25 were each significantly related to younger mean age at symptom onset by an average of 5 years:
• overweight: 26.9 years old
• not overweight: 32.1 years old
Citation: Kavak KS, Teter BE, Hagemeier J, Zakalik K, Weinstock-Guttman B; New York State Multiple Sclerosis Consortium. Higher weight in adolescence and young adulthood is associated with an earlier age at multiple sclerosis onset. Mult Scler. 2015;21(7):858-865. doi:10.1177/1352458514555787.
A heavier weight in adolescence is associated with a multiple sclerosis diagnosis at a younger age, according to subgroup cohort of 184 women who provided weight and height information from the age of first menstruation and at age 25.
Investigators calculated BMI and used regression analyses to investigate an association, and found having a higher weight at menarche, weight at age 25, and BMI at age 25 were each significantly related to younger mean age at symptom onset by an average of 5 years:
• overweight: 26.9 years old
• not overweight: 32.1 years old
Citation: Kavak KS, Teter BE, Hagemeier J, Zakalik K, Weinstock-Guttman B; New York State Multiple Sclerosis Consortium. Higher weight in adolescence and young adulthood is associated with an earlier age at multiple sclerosis onset. Mult Scler. 2015;21(7):858-865. doi:10.1177/1352458514555787.
Cervical Cord Atrophy in CIS and RRMS
Cervical cord atrophy occurs in clinical isolated syndrome (CIS) as well as more progressive forms of multiple sclerosis (MS), a study of 267 patients with CIS or relapsing-remitting MS (RRMS) and 64 healthy controls reports.
Investigators used structural brain magnetic resonance imaging (MRI) to determine upper cervical cord cross-sectional area (UCCA) at the level of C2/C3 and adjusted for focal MS lesions. They found:
• UCCA was significantly reduced in CIS patients compared to healthy controls.
• Structural variability was higher in patients than in controls, particularly in the case of focal lesions.
• UCCA and the coefficient of variation (CV) were associated with Expanded Disability Status Scale (EDDS) sores and disease duration.
• CV was also associated with hand and arm function.
Citation: Biberacher V, Boucard CC, Schmidt P, et al. Atrophy and structural variability of the upper cervical cord in early multiple sclerosis. Mult Scler. 2015;21(7):875-884. doi:10.1177/1352458514546514.
Cervical cord atrophy occurs in clinical isolated syndrome (CIS) as well as more progressive forms of multiple sclerosis (MS), a study of 267 patients with CIS or relapsing-remitting MS (RRMS) and 64 healthy controls reports.
Investigators used structural brain magnetic resonance imaging (MRI) to determine upper cervical cord cross-sectional area (UCCA) at the level of C2/C3 and adjusted for focal MS lesions. They found:
• UCCA was significantly reduced in CIS patients compared to healthy controls.
• Structural variability was higher in patients than in controls, particularly in the case of focal lesions.
• UCCA and the coefficient of variation (CV) were associated with Expanded Disability Status Scale (EDDS) sores and disease duration.
• CV was also associated with hand and arm function.
Citation: Biberacher V, Boucard CC, Schmidt P, et al. Atrophy and structural variability of the upper cervical cord in early multiple sclerosis. Mult Scler. 2015;21(7):875-884. doi:10.1177/1352458514546514.
Cervical cord atrophy occurs in clinical isolated syndrome (CIS) as well as more progressive forms of multiple sclerosis (MS), a study of 267 patients with CIS or relapsing-remitting MS (RRMS) and 64 healthy controls reports.
Investigators used structural brain magnetic resonance imaging (MRI) to determine upper cervical cord cross-sectional area (UCCA) at the level of C2/C3 and adjusted for focal MS lesions. They found:
• UCCA was significantly reduced in CIS patients compared to healthy controls.
• Structural variability was higher in patients than in controls, particularly in the case of focal lesions.
• UCCA and the coefficient of variation (CV) were associated with Expanded Disability Status Scale (EDDS) sores and disease duration.
• CV was also associated with hand and arm function.
Citation: Biberacher V, Boucard CC, Schmidt P, et al. Atrophy and structural variability of the upper cervical cord in early multiple sclerosis. Mult Scler. 2015;21(7):875-884. doi:10.1177/1352458514546514.
New drugs saw ‘unprecedented’ spending growth in 2014
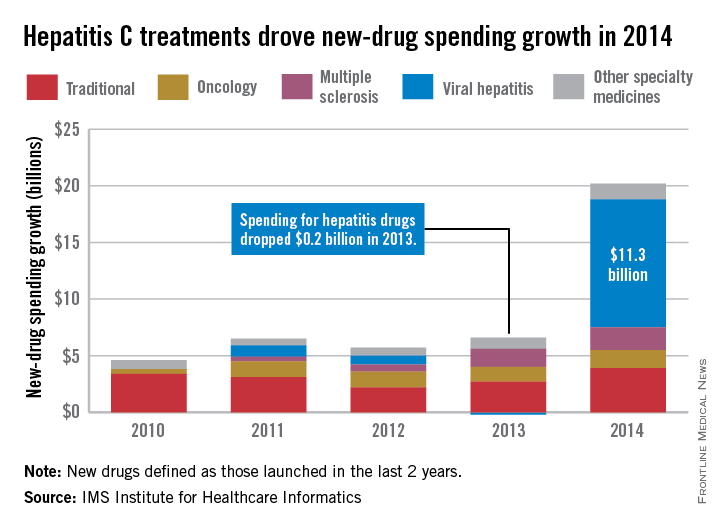
Led by new treatments for hepatitis C, there was an “unprecedented $20.2 billion increase in spending” for new medicines in 2014, which was triple the 2013 level, according to a report from the IMS Institute for Healthcare Informatics.
Compared with 2013, spending for new viral hepatitis drugs was up by $11.3 billion in 2014, accounting for more than half of the total increase. Hepatitis C drugs and other specialty medicines – defined as “products that are often injectable, high-cost, biologic … and include treatment for cancer and other serious chronic conditions” – accounted for 81% of the $20.2 billion, the report noted.
The number of patients who sought treatment for hepatitis C jumped from 17,000 in 2013 to 161,000 in 2014, “owing to new treatments with cure rates over 90% and dramatically fewer side effects,” the IMS Institute said.
Spending on new drugs in the traditional sector, such as those for diabetes, was up by $3.9 billion in 2014. The other leading areas of spending increase in the specialty sector – oncology and multiple sclerosis – were up by $1.6 billion and $2.0 billion, respectively, the report said.
The IMS Institute defines new drugs as those launched in the last 2 years. The report includes data from IMS National Sales Perspectives, which “reports 100% coverage of the retail and nonretail channels for national [non-OTC] pharmaceutical sales at actual transaction prices.”
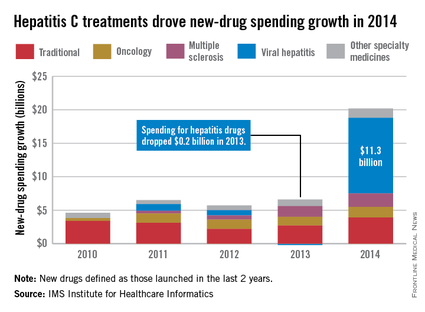
Led by new treatments for hepatitis C, there was an “unprecedented $20.2 billion increase in spending” for new medicines in 2014, which was triple the 2013 level, according to a report from the IMS Institute for Healthcare Informatics.
Compared with 2013, spending for new viral hepatitis drugs was up by $11.3 billion in 2014, accounting for more than half of the total increase. Hepatitis C drugs and other specialty medicines – defined as “products that are often injectable, high-cost, biologic … and include treatment for cancer and other serious chronic conditions” – accounted for 81% of the $20.2 billion, the report noted.
The number of patients who sought treatment for hepatitis C jumped from 17,000 in 2013 to 161,000 in 2014, “owing to new treatments with cure rates over 90% and dramatically fewer side effects,” the IMS Institute said.
Spending on new drugs in the traditional sector, such as those for diabetes, was up by $3.9 billion in 2014. The other leading areas of spending increase in the specialty sector – oncology and multiple sclerosis – were up by $1.6 billion and $2.0 billion, respectively, the report said.
The IMS Institute defines new drugs as those launched in the last 2 years. The report includes data from IMS National Sales Perspectives, which “reports 100% coverage of the retail and nonretail channels for national [non-OTC] pharmaceutical sales at actual transaction prices.”

Led by new treatments for hepatitis C, there was an “unprecedented $20.2 billion increase in spending” for new medicines in 2014, which was triple the 2013 level, according to a report from the IMS Institute for Healthcare Informatics.
Compared with 2013, spending for new viral hepatitis drugs was up by $11.3 billion in 2014, accounting for more than half of the total increase. Hepatitis C drugs and other specialty medicines – defined as “products that are often injectable, high-cost, biologic … and include treatment for cancer and other serious chronic conditions” – accounted for 81% of the $20.2 billion, the report noted.
The number of patients who sought treatment for hepatitis C jumped from 17,000 in 2013 to 161,000 in 2014, “owing to new treatments with cure rates over 90% and dramatically fewer side effects,” the IMS Institute said.
Spending on new drugs in the traditional sector, such as those for diabetes, was up by $3.9 billion in 2014. The other leading areas of spending increase in the specialty sector – oncology and multiple sclerosis – were up by $1.6 billion and $2.0 billion, respectively, the report said.
The IMS Institute defines new drugs as those launched in the last 2 years. The report includes data from IMS National Sales Perspectives, which “reports 100% coverage of the retail and nonretail channels for national [non-OTC] pharmaceutical sales at actual transaction prices.”

Thoracic gray matter atrophy linked to MS disability
Atrophy of gray matter in the thoracic cords of multiple sclerosis patients can be assessed in vivo using magnetic resonance imaging, and this shows a significant association between the area of gray matter in the lower thoracic cord and disease type and disability, according to a paper published online June 8.
A cross-sectional observational study of 142 patients with multiple sclerosis (MS) and 20 healthy controls, using phase-sensitive inversion recovery MRI, showed that, while patients with relapsing multiple sclerosis had smaller thoracic cord gray matter areas than controls, patients with progressive MS showed even more substantial reductions in thoracic cord gray matter.
Researchers also found an inverse correlation between lower thoracic cord gray matter and Expanded Disability Status Scale score, even after accounting for cervical gray matter area, Dr. Regina Schlaeger and her associates reported in JAMA Neurology.
“Magnetic resonance imaging–based estimations of cord GM [gray matter] that have strong correlations with MS disability may better track the disease process in the context of both clinical trials and longitudinal observational studies,” wrote Dr. Schlaeger of the department of neurology at the University of California, San Francisco, and her coauthors (JAMA Neurology 2015 June 8 [doi:10.1001/jamaneurol.2015.0993]).
The study was supported by the National Multiple Sclerosis Society, the Conrad H. Hilton Foundation, the Department of Defense, the National Institutes of Health, the National Defense Science and Engineering Fellowship, and the Nancy Davis Foundation. Some authors reported personal fees, committee roles, consultancies, board positions and support from numerous pharmaceutical companies.
Atrophy of gray matter in the thoracic cords of multiple sclerosis patients can be assessed in vivo using magnetic resonance imaging, and this shows a significant association between the area of gray matter in the lower thoracic cord and disease type and disability, according to a paper published online June 8.
A cross-sectional observational study of 142 patients with multiple sclerosis (MS) and 20 healthy controls, using phase-sensitive inversion recovery MRI, showed that, while patients with relapsing multiple sclerosis had smaller thoracic cord gray matter areas than controls, patients with progressive MS showed even more substantial reductions in thoracic cord gray matter.
Researchers also found an inverse correlation between lower thoracic cord gray matter and Expanded Disability Status Scale score, even after accounting for cervical gray matter area, Dr. Regina Schlaeger and her associates reported in JAMA Neurology.
“Magnetic resonance imaging–based estimations of cord GM [gray matter] that have strong correlations with MS disability may better track the disease process in the context of both clinical trials and longitudinal observational studies,” wrote Dr. Schlaeger of the department of neurology at the University of California, San Francisco, and her coauthors (JAMA Neurology 2015 June 8 [doi:10.1001/jamaneurol.2015.0993]).
The study was supported by the National Multiple Sclerosis Society, the Conrad H. Hilton Foundation, the Department of Defense, the National Institutes of Health, the National Defense Science and Engineering Fellowship, and the Nancy Davis Foundation. Some authors reported personal fees, committee roles, consultancies, board positions and support from numerous pharmaceutical companies.
Atrophy of gray matter in the thoracic cords of multiple sclerosis patients can be assessed in vivo using magnetic resonance imaging, and this shows a significant association between the area of gray matter in the lower thoracic cord and disease type and disability, according to a paper published online June 8.
A cross-sectional observational study of 142 patients with multiple sclerosis (MS) and 20 healthy controls, using phase-sensitive inversion recovery MRI, showed that, while patients with relapsing multiple sclerosis had smaller thoracic cord gray matter areas than controls, patients with progressive MS showed even more substantial reductions in thoracic cord gray matter.
Researchers also found an inverse correlation between lower thoracic cord gray matter and Expanded Disability Status Scale score, even after accounting for cervical gray matter area, Dr. Regina Schlaeger and her associates reported in JAMA Neurology.
“Magnetic resonance imaging–based estimations of cord GM [gray matter] that have strong correlations with MS disability may better track the disease process in the context of both clinical trials and longitudinal observational studies,” wrote Dr. Schlaeger of the department of neurology at the University of California, San Francisco, and her coauthors (JAMA Neurology 2015 June 8 [doi:10.1001/jamaneurol.2015.0993]).
The study was supported by the National Multiple Sclerosis Society, the Conrad H. Hilton Foundation, the Department of Defense, the National Institutes of Health, the National Defense Science and Engineering Fellowship, and the Nancy Davis Foundation. Some authors reported personal fees, committee roles, consultancies, board positions and support from numerous pharmaceutical companies.
FROM JAMA NEUROLOGY
Key clinical point: Atrophy of gray matter in the thoracic cord in multiple sclerosis patients can be assessed in vivo and is associated with disease type and disability.
Major finding: Patients with relapsing multiple sclerosis had smaller thoracic cord gray matter areas than controls, while patients with progressive MS showed even more substantial reductions in thoracic cord gray matter.
Data source: A cross-sectional observational study of 142 patients with MS and 20 healthy controls.
Disclosures: The study was supported by the National Multiple Sclerosis Society, the Conrad H. Hilton Foundation, the Department of Defense, the National Institutes of Health, the National Defense Science and Engineering Fellowship, and the Nancy Davis Foundation. Some authors reported personal fees, committee roles, consultancies, board positions, and support from numerous pharmaceutical companies.