User login
AAN calls oral cannabinoids effective for MS pain, spasticity
An expert panel organized by the American Academy of Neurology called oral cannabis extract the only complementary and alternative medicine unequivocally effective for helping patients with multiple sclerosis, specifically easing their pain and symptoms of spasticity, possibly for as long as 1 year of treatment.
The academy’s Guideline Development Subcommittee also found existing evidence "insufficient to support or refute the effectiveness" of 25 other complementary and alternative medicine (CAM) treatments, including acupuncture, chelation therapy, mindfulness training, and muscle-relaxation therapy. The panel noted that two of these inadequately assessed treatments – dental amalgam removal and transdermal histamine – have received substantial media attention despite having "little or no evidence to support recommendations."
Aside from various forms and delivery methods for cannabinoids, the nine-member panel found six other treatments with adequate evidence to develop practice recommendations that either endorsed their efficacy or lack of effect. Ginkgo biloba, reflexology, and magnetic therapy all had some proven level of efficacy, while bee venom, low-fat diet with omega-3 supplementation, and lofepramine plus L-phenylalanine with B12 were all found ineffective, the subcommittee said in guidelines released on March 24 (Neurology 2014;82:1083-92).
The efficacy of CAM therapies in patients with multiple sclerosis (MS) is an important clinical issue. Ten reports cited by the subcommittee and published during 1999-2009 documented that anywhere from a third to 80% of MS patients – particularly women, patients with higher education levels, and patients who report poorer health – used one or more CAM therapies, according to the panel, which was led by Dr. Vijayshree Yadav of the department of neurology at Oregon Health and Science University, Portland, and the Portland VA Medical Center.
The group also determined that oral cannabis extract and another orally delivered cannabinoid, synthetic tetrahydrocannabinol (THC), were possibly effective for reducing symptoms and objective measures of spasticity during treatment beyond 1 year, and that THC is probably effective for reducing symptoms of spasticity and pain during the first year of treatment. The panel decided that, based on existing evidence, both of these oral agents are "probably ineffective" for reducing both objective spasticity measures and MS-related tremor symptoms.
The subcommittee reviewed two other delivery forms of cannabinoids. The members concluded that Sativex oromucosal cannabinoid spray is probably effective for improving subjective spasticity symptoms for periods of 5-10 weeks and possibly ineffective when used for longer periods or for reducing MS-related tremor. When it came to smoked cannabis, the panel decided that the data were inadequate to draw any conclusions on safety or efficacy.
It also deemed the evidence inadequate to draw conclusions about oral cannabis extract or THC for bladder-urge incontinence or for treating overall symptoms; synthetic THC for central neuropathic pain; and Sativex spray for overall bladder symptoms, anxiety, sleep problems, cognitive symptoms, quality of life, or fatigue.
In addition, cannabinoid studies have been of short duration (6-15 weeks), and central side effects may have caused unblinding in studies. The panel cautioned clinicians to counsel patients about potential psychopathologic effects, cognitive effects, or both with cannabinoid use, and cautioned against extrapolating from findings with standardized oral cannabis extract to other, nonstandardized cannabis extracts.
For other treatments with an adequate evidence base, the panel concluded that magnetic therapy is probably effective for reducing fatigue and probably ineffective for reducing depression, with inadequate data to support or refute other effects in MS patients.
The subcommittee said that study findings established Ginkgo biloba as ineffective for improving cognitive function in patients with MS but possibly effective during 4 weeks of treatment to reduce fatigue. The members also warned that Ginkgo biloba and other supplements not regulated by the Food and Drug Administration may vary considerably in efficacy and adverse effects and may interact with other medications, especially disease-modifying therapies for MS.
The panel called low-fat diet with omega-3 fatty acid supplementation probably ineffective for reducing MS relapses, disability, or MRI lesions, or for improving fatigue or quality of life. It found lofepramine plus L-phenylalanine and vitamin B12 possibly ineffective for treating disability, symptoms, depression, or fatigue, and bee-sting therapy possibly ineffective for reducing relapses, disability, fatigue, total MRI-lesion burden, and gadolinium-enhancing lesion volume, or for improving health-related quality of life.
The subcommittee said that reflexology is possibly effective for reducing MS-associated paresthesia during 11 weeks of treatment, but that data were inadequate to support or refute its use for pain, spasticity, fatigue, anxiety, or several other MS manifestations.
The guidelines were funded by the American Academy of Neurology. Most of the panel members reported some potential conflicts of interest in relationships with pharmaceutical companies that market drugs for MS as well as ties to MS medical societies.
On Twitter @mitchelzoler
An expert panel organized by the American Academy of Neurology called oral cannabis extract the only complementary and alternative medicine unequivocally effective for helping patients with multiple sclerosis, specifically easing their pain and symptoms of spasticity, possibly for as long as 1 year of treatment.
The academy’s Guideline Development Subcommittee also found existing evidence "insufficient to support or refute the effectiveness" of 25 other complementary and alternative medicine (CAM) treatments, including acupuncture, chelation therapy, mindfulness training, and muscle-relaxation therapy. The panel noted that two of these inadequately assessed treatments – dental amalgam removal and transdermal histamine – have received substantial media attention despite having "little or no evidence to support recommendations."
Aside from various forms and delivery methods for cannabinoids, the nine-member panel found six other treatments with adequate evidence to develop practice recommendations that either endorsed their efficacy or lack of effect. Ginkgo biloba, reflexology, and magnetic therapy all had some proven level of efficacy, while bee venom, low-fat diet with omega-3 supplementation, and lofepramine plus L-phenylalanine with B12 were all found ineffective, the subcommittee said in guidelines released on March 24 (Neurology 2014;82:1083-92).
The efficacy of CAM therapies in patients with multiple sclerosis (MS) is an important clinical issue. Ten reports cited by the subcommittee and published during 1999-2009 documented that anywhere from a third to 80% of MS patients – particularly women, patients with higher education levels, and patients who report poorer health – used one or more CAM therapies, according to the panel, which was led by Dr. Vijayshree Yadav of the department of neurology at Oregon Health and Science University, Portland, and the Portland VA Medical Center.
The group also determined that oral cannabis extract and another orally delivered cannabinoid, synthetic tetrahydrocannabinol (THC), were possibly effective for reducing symptoms and objective measures of spasticity during treatment beyond 1 year, and that THC is probably effective for reducing symptoms of spasticity and pain during the first year of treatment. The panel decided that, based on existing evidence, both of these oral agents are "probably ineffective" for reducing both objective spasticity measures and MS-related tremor symptoms.
The subcommittee reviewed two other delivery forms of cannabinoids. The members concluded that Sativex oromucosal cannabinoid spray is probably effective for improving subjective spasticity symptoms for periods of 5-10 weeks and possibly ineffective when used for longer periods or for reducing MS-related tremor. When it came to smoked cannabis, the panel decided that the data were inadequate to draw any conclusions on safety or efficacy.
It also deemed the evidence inadequate to draw conclusions about oral cannabis extract or THC for bladder-urge incontinence or for treating overall symptoms; synthetic THC for central neuropathic pain; and Sativex spray for overall bladder symptoms, anxiety, sleep problems, cognitive symptoms, quality of life, or fatigue.
In addition, cannabinoid studies have been of short duration (6-15 weeks), and central side effects may have caused unblinding in studies. The panel cautioned clinicians to counsel patients about potential psychopathologic effects, cognitive effects, or both with cannabinoid use, and cautioned against extrapolating from findings with standardized oral cannabis extract to other, nonstandardized cannabis extracts.
For other treatments with an adequate evidence base, the panel concluded that magnetic therapy is probably effective for reducing fatigue and probably ineffective for reducing depression, with inadequate data to support or refute other effects in MS patients.
The subcommittee said that study findings established Ginkgo biloba as ineffective for improving cognitive function in patients with MS but possibly effective during 4 weeks of treatment to reduce fatigue. The members also warned that Ginkgo biloba and other supplements not regulated by the Food and Drug Administration may vary considerably in efficacy and adverse effects and may interact with other medications, especially disease-modifying therapies for MS.
The panel called low-fat diet with omega-3 fatty acid supplementation probably ineffective for reducing MS relapses, disability, or MRI lesions, or for improving fatigue or quality of life. It found lofepramine plus L-phenylalanine and vitamin B12 possibly ineffective for treating disability, symptoms, depression, or fatigue, and bee-sting therapy possibly ineffective for reducing relapses, disability, fatigue, total MRI-lesion burden, and gadolinium-enhancing lesion volume, or for improving health-related quality of life.
The subcommittee said that reflexology is possibly effective for reducing MS-associated paresthesia during 11 weeks of treatment, but that data were inadequate to support or refute its use for pain, spasticity, fatigue, anxiety, or several other MS manifestations.
The guidelines were funded by the American Academy of Neurology. Most of the panel members reported some potential conflicts of interest in relationships with pharmaceutical companies that market drugs for MS as well as ties to MS medical societies.
On Twitter @mitchelzoler
An expert panel organized by the American Academy of Neurology called oral cannabis extract the only complementary and alternative medicine unequivocally effective for helping patients with multiple sclerosis, specifically easing their pain and symptoms of spasticity, possibly for as long as 1 year of treatment.
The academy’s Guideline Development Subcommittee also found existing evidence "insufficient to support or refute the effectiveness" of 25 other complementary and alternative medicine (CAM) treatments, including acupuncture, chelation therapy, mindfulness training, and muscle-relaxation therapy. The panel noted that two of these inadequately assessed treatments – dental amalgam removal and transdermal histamine – have received substantial media attention despite having "little or no evidence to support recommendations."
Aside from various forms and delivery methods for cannabinoids, the nine-member panel found six other treatments with adequate evidence to develop practice recommendations that either endorsed their efficacy or lack of effect. Ginkgo biloba, reflexology, and magnetic therapy all had some proven level of efficacy, while bee venom, low-fat diet with omega-3 supplementation, and lofepramine plus L-phenylalanine with B12 were all found ineffective, the subcommittee said in guidelines released on March 24 (Neurology 2014;82:1083-92).
The efficacy of CAM therapies in patients with multiple sclerosis (MS) is an important clinical issue. Ten reports cited by the subcommittee and published during 1999-2009 documented that anywhere from a third to 80% of MS patients – particularly women, patients with higher education levels, and patients who report poorer health – used one or more CAM therapies, according to the panel, which was led by Dr. Vijayshree Yadav of the department of neurology at Oregon Health and Science University, Portland, and the Portland VA Medical Center.
The group also determined that oral cannabis extract and another orally delivered cannabinoid, synthetic tetrahydrocannabinol (THC), were possibly effective for reducing symptoms and objective measures of spasticity during treatment beyond 1 year, and that THC is probably effective for reducing symptoms of spasticity and pain during the first year of treatment. The panel decided that, based on existing evidence, both of these oral agents are "probably ineffective" for reducing both objective spasticity measures and MS-related tremor symptoms.
The subcommittee reviewed two other delivery forms of cannabinoids. The members concluded that Sativex oromucosal cannabinoid spray is probably effective for improving subjective spasticity symptoms for periods of 5-10 weeks and possibly ineffective when used for longer periods or for reducing MS-related tremor. When it came to smoked cannabis, the panel decided that the data were inadequate to draw any conclusions on safety or efficacy.
It also deemed the evidence inadequate to draw conclusions about oral cannabis extract or THC for bladder-urge incontinence or for treating overall symptoms; synthetic THC for central neuropathic pain; and Sativex spray for overall bladder symptoms, anxiety, sleep problems, cognitive symptoms, quality of life, or fatigue.
In addition, cannabinoid studies have been of short duration (6-15 weeks), and central side effects may have caused unblinding in studies. The panel cautioned clinicians to counsel patients about potential psychopathologic effects, cognitive effects, or both with cannabinoid use, and cautioned against extrapolating from findings with standardized oral cannabis extract to other, nonstandardized cannabis extracts.
For other treatments with an adequate evidence base, the panel concluded that magnetic therapy is probably effective for reducing fatigue and probably ineffective for reducing depression, with inadequate data to support or refute other effects in MS patients.
The subcommittee said that study findings established Ginkgo biloba as ineffective for improving cognitive function in patients with MS but possibly effective during 4 weeks of treatment to reduce fatigue. The members also warned that Ginkgo biloba and other supplements not regulated by the Food and Drug Administration may vary considerably in efficacy and adverse effects and may interact with other medications, especially disease-modifying therapies for MS.
The panel called low-fat diet with omega-3 fatty acid supplementation probably ineffective for reducing MS relapses, disability, or MRI lesions, or for improving fatigue or quality of life. It found lofepramine plus L-phenylalanine and vitamin B12 possibly ineffective for treating disability, symptoms, depression, or fatigue, and bee-sting therapy possibly ineffective for reducing relapses, disability, fatigue, total MRI-lesion burden, and gadolinium-enhancing lesion volume, or for improving health-related quality of life.
The subcommittee said that reflexology is possibly effective for reducing MS-associated paresthesia during 11 weeks of treatment, but that data were inadequate to support or refute its use for pain, spasticity, fatigue, anxiety, or several other MS manifestations.
The guidelines were funded by the American Academy of Neurology. Most of the panel members reported some potential conflicts of interest in relationships with pharmaceutical companies that market drugs for MS as well as ties to MS medical societies.
On Twitter @mitchelzoler
FROM NEUROLOGY
Simvastatin slows brain atrophy in secondary progressive multiple sclerosis
A high dose of simvastatin decreased whole-brain atrophy by 43% per year over 24 months, compared with placebo, in a phase II, randomized, controlled trial in patients with secondary progressive multiple sclerosis.
The drug had no apparent effect on relapses or new lesions. But patients who took it showed significantly better scores on the Multiple Sclerosis Impact Scale–29 (MSIS-29) and the Expanded Disability Status Scale (EDSS), suggesting that slowing brain atrophy may have a beneficial effect on function, reported Jeremy Chataway, Ph.D., of the University College London (Lancet 2014 March 19 [doi:10.1016/S0140-6736(13)62242-4]).
However, those results should be interpreted cautiously because functional status wasn’t the primary endpoint of the trial, wrote Dr. Chataway and his coauthors.
The study randomized 140 patients with secondary progressive multiple sclerosis to placebo or 80 mg/day simvastatin for 24 months. Brain volume and functional and psychological status were assessed at baseline and 1, 6, 12, and 24 months. The mean age of the patients was 52 years, and average disease duration was 21 years. The mean whole-brain volume was 1,100 mL. The mean EDSS score was 5.8, and the mean MSIS-29 score was 70.
Nine patients did not complete the full trial. Treatment compliance was 90% or greater for 77%-78% of the remaining patients.
At 24 months, whole-brain volume had declined in both groups. But the annual decline was significantly less in those taking simvastatin (–0.288% vs. –0.584%). The adjusted difference in annual atrophy was 0.254% per year, resulting in 43% less volume loss per year in the simvastatin group than in the placebo group. This effect was seen in MRI scans at both 12 and 24 months, and occurred in three-fourths of the treated population.
The rate of new or enlarging lesions was not significantly different, although there was a trend toward benefit in the treated group (1.50 vs. 2.19 per person per year). The relapse rates were similar (0.20 vs. 0.16 events per person/year).
At 24 months, the mean EDSS score in the simvastatin group was significantly lower than in the placebo group (a mean difference of –0.254 after adjustment for baseline measurements). The total mean MSIS-29 also showed a significant between-group difference in favor of simvastatin (–4.78 after adjustment). The difference was most pronounced in the physical subscale of the MSIS-29 (–3.73), but the psychological subscale was not significantly different. Overall, changes in these secondary measures by 24 months reflected worsening in both groups, but more so in the placebo group.
Cholesterol also decreased significantly in the simvastatin group (from 5.5 mmol/L to 4.1 mmol/L); it did not change in the placebo group. However, there were no differences in inflammatory markers.
Drug-related adverse events occurred in 23% of patients on simvastatin, compared with 19% of those on placebo. Simvastatin was generally well tolerated, with no safety issues.
It’s not entirely clear how a statin would slow disease progression, the researchers said. "gAccumulating evidence shows that statins have cell protective properties. For example, inhibition of inducible nitric oxide synthase, thus reducing release of free radicals from activated microglia and astrocytes or exerting a neuroprotective effect by prevention of glutamate-mediated excitotoxic effects."h
Simvastatin might also improve cerebral vasomotor reactivity, which could protect against hypoxic damage, they added. The general benefit of statins on vascular health could also be a contributor.
The study was partially supported by a grant from the U.K. National Institute for Health Research, the Moulton Foundation, the Berkeley Foundation, the Multiple Sclerosis Trials Collaboration, the Rosetrees Trust, and a personal contribution.
None of the authors had any financial disclosures.
On Twitter @alz_gal
Cautious optimism is the catchphrase for this new study of simvastatin in multiple sclerosis, Dr. Jacqueline Palace and Dr. Neil Robertson wrote in an accompanying editorial.
The study "is a promising and novel development," for a disease that has no long-term progression-modifying therapies. "This form of multiple sclerosis has been largely neglected by a pharmaceutical industry that has so far focused on the early inflammatory processes. A substantial advantage of this study is the fairly low cost, availability, and documented safety profile of the drug."
Simvastatin’s effect on neurodegeneration rather than inflammation also suggests a novel mechanism that might be utilized in combination with anti-inflammatory treatments.
"Caution in interpretation of these preliminary data is, however, needed. ... The expected effects of simvastatin on inflammation were not shown. Furthermore, the disability outcome was a surrogate measure and not the usual outcome used in phase III trials. This finding is relevant because single disability measures show great variability in the short term, and the trial was clearly underpowered to identify an effect on arguably the least sensitive of disability measures (EDSS). The meaning of an effect on atrophy, without a reduction in lesion activity, in predicting the future effect on sustained disability outcomes is unknown."
Nonetheless, the authors wrote, the study uncovers a potential that needs exploration. (Lancet 2014 March 19 [doi: 10.1016/S0140-6736(13)62641-0]).
"Further phase III studies to measure the effect of simvastatin on sustained disability, particularly in patients with nonrelapsing secondary progressive and primary progressive multiple sclerosis, are clearly needed, but this trial represents a promising point from which to develop trials of progressive disease."
Dr. Palace leads the neuromyelitis optica service at John Radcliffe Hospital, Oxford, England. Dr. Robertston is professor of neurology in the Institute of Psychological Medicine and Clinical Neurosciences at Cardiff (Wales) University. Both reported financial relationships, including personal remuneration, from several pharmaceutical companies marketing drugs for multiple sclerosis.
Cautious optimism is the catchphrase for this new study of simvastatin in multiple sclerosis, Dr. Jacqueline Palace and Dr. Neil Robertson wrote in an accompanying editorial.
The study "is a promising and novel development," for a disease that has no long-term progression-modifying therapies. "This form of multiple sclerosis has been largely neglected by a pharmaceutical industry that has so far focused on the early inflammatory processes. A substantial advantage of this study is the fairly low cost, availability, and documented safety profile of the drug."
Simvastatin’s effect on neurodegeneration rather than inflammation also suggests a novel mechanism that might be utilized in combination with anti-inflammatory treatments.
"Caution in interpretation of these preliminary data is, however, needed. ... The expected effects of simvastatin on inflammation were not shown. Furthermore, the disability outcome was a surrogate measure and not the usual outcome used in phase III trials. This finding is relevant because single disability measures show great variability in the short term, and the trial was clearly underpowered to identify an effect on arguably the least sensitive of disability measures (EDSS). The meaning of an effect on atrophy, without a reduction in lesion activity, in predicting the future effect on sustained disability outcomes is unknown."
Nonetheless, the authors wrote, the study uncovers a potential that needs exploration. (Lancet 2014 March 19 [doi: 10.1016/S0140-6736(13)62641-0]).
"Further phase III studies to measure the effect of simvastatin on sustained disability, particularly in patients with nonrelapsing secondary progressive and primary progressive multiple sclerosis, are clearly needed, but this trial represents a promising point from which to develop trials of progressive disease."
Dr. Palace leads the neuromyelitis optica service at John Radcliffe Hospital, Oxford, England. Dr. Robertston is professor of neurology in the Institute of Psychological Medicine and Clinical Neurosciences at Cardiff (Wales) University. Both reported financial relationships, including personal remuneration, from several pharmaceutical companies marketing drugs for multiple sclerosis.
Cautious optimism is the catchphrase for this new study of simvastatin in multiple sclerosis, Dr. Jacqueline Palace and Dr. Neil Robertson wrote in an accompanying editorial.
The study "is a promising and novel development," for a disease that has no long-term progression-modifying therapies. "This form of multiple sclerosis has been largely neglected by a pharmaceutical industry that has so far focused on the early inflammatory processes. A substantial advantage of this study is the fairly low cost, availability, and documented safety profile of the drug."
Simvastatin’s effect on neurodegeneration rather than inflammation also suggests a novel mechanism that might be utilized in combination with anti-inflammatory treatments.
"Caution in interpretation of these preliminary data is, however, needed. ... The expected effects of simvastatin on inflammation were not shown. Furthermore, the disability outcome was a surrogate measure and not the usual outcome used in phase III trials. This finding is relevant because single disability measures show great variability in the short term, and the trial was clearly underpowered to identify an effect on arguably the least sensitive of disability measures (EDSS). The meaning of an effect on atrophy, without a reduction in lesion activity, in predicting the future effect on sustained disability outcomes is unknown."
Nonetheless, the authors wrote, the study uncovers a potential that needs exploration. (Lancet 2014 March 19 [doi: 10.1016/S0140-6736(13)62641-0]).
"Further phase III studies to measure the effect of simvastatin on sustained disability, particularly in patients with nonrelapsing secondary progressive and primary progressive multiple sclerosis, are clearly needed, but this trial represents a promising point from which to develop trials of progressive disease."
Dr. Palace leads the neuromyelitis optica service at John Radcliffe Hospital, Oxford, England. Dr. Robertston is professor of neurology in the Institute of Psychological Medicine and Clinical Neurosciences at Cardiff (Wales) University. Both reported financial relationships, including personal remuneration, from several pharmaceutical companies marketing drugs for multiple sclerosis.
A high dose of simvastatin decreased whole-brain atrophy by 43% per year over 24 months, compared with placebo, in a phase II, randomized, controlled trial in patients with secondary progressive multiple sclerosis.
The drug had no apparent effect on relapses or new lesions. But patients who took it showed significantly better scores on the Multiple Sclerosis Impact Scale–29 (MSIS-29) and the Expanded Disability Status Scale (EDSS), suggesting that slowing brain atrophy may have a beneficial effect on function, reported Jeremy Chataway, Ph.D., of the University College London (Lancet 2014 March 19 [doi:10.1016/S0140-6736(13)62242-4]).
However, those results should be interpreted cautiously because functional status wasn’t the primary endpoint of the trial, wrote Dr. Chataway and his coauthors.
The study randomized 140 patients with secondary progressive multiple sclerosis to placebo or 80 mg/day simvastatin for 24 months. Brain volume and functional and psychological status were assessed at baseline and 1, 6, 12, and 24 months. The mean age of the patients was 52 years, and average disease duration was 21 years. The mean whole-brain volume was 1,100 mL. The mean EDSS score was 5.8, and the mean MSIS-29 score was 70.
Nine patients did not complete the full trial. Treatment compliance was 90% or greater for 77%-78% of the remaining patients.
At 24 months, whole-brain volume had declined in both groups. But the annual decline was significantly less in those taking simvastatin (–0.288% vs. –0.584%). The adjusted difference in annual atrophy was 0.254% per year, resulting in 43% less volume loss per year in the simvastatin group than in the placebo group. This effect was seen in MRI scans at both 12 and 24 months, and occurred in three-fourths of the treated population.
The rate of new or enlarging lesions was not significantly different, although there was a trend toward benefit in the treated group (1.50 vs. 2.19 per person per year). The relapse rates were similar (0.20 vs. 0.16 events per person/year).
At 24 months, the mean EDSS score in the simvastatin group was significantly lower than in the placebo group (a mean difference of –0.254 after adjustment for baseline measurements). The total mean MSIS-29 also showed a significant between-group difference in favor of simvastatin (–4.78 after adjustment). The difference was most pronounced in the physical subscale of the MSIS-29 (–3.73), but the psychological subscale was not significantly different. Overall, changes in these secondary measures by 24 months reflected worsening in both groups, but more so in the placebo group.
Cholesterol also decreased significantly in the simvastatin group (from 5.5 mmol/L to 4.1 mmol/L); it did not change in the placebo group. However, there were no differences in inflammatory markers.
Drug-related adverse events occurred in 23% of patients on simvastatin, compared with 19% of those on placebo. Simvastatin was generally well tolerated, with no safety issues.
It’s not entirely clear how a statin would slow disease progression, the researchers said. "gAccumulating evidence shows that statins have cell protective properties. For example, inhibition of inducible nitric oxide synthase, thus reducing release of free radicals from activated microglia and astrocytes or exerting a neuroprotective effect by prevention of glutamate-mediated excitotoxic effects."h
Simvastatin might also improve cerebral vasomotor reactivity, which could protect against hypoxic damage, they added. The general benefit of statins on vascular health could also be a contributor.
The study was partially supported by a grant from the U.K. National Institute for Health Research, the Moulton Foundation, the Berkeley Foundation, the Multiple Sclerosis Trials Collaboration, the Rosetrees Trust, and a personal contribution.
None of the authors had any financial disclosures.
On Twitter @alz_gal
A high dose of simvastatin decreased whole-brain atrophy by 43% per year over 24 months, compared with placebo, in a phase II, randomized, controlled trial in patients with secondary progressive multiple sclerosis.
The drug had no apparent effect on relapses or new lesions. But patients who took it showed significantly better scores on the Multiple Sclerosis Impact Scale–29 (MSIS-29) and the Expanded Disability Status Scale (EDSS), suggesting that slowing brain atrophy may have a beneficial effect on function, reported Jeremy Chataway, Ph.D., of the University College London (Lancet 2014 March 19 [doi:10.1016/S0140-6736(13)62242-4]).
However, those results should be interpreted cautiously because functional status wasn’t the primary endpoint of the trial, wrote Dr. Chataway and his coauthors.
The study randomized 140 patients with secondary progressive multiple sclerosis to placebo or 80 mg/day simvastatin for 24 months. Brain volume and functional and psychological status were assessed at baseline and 1, 6, 12, and 24 months. The mean age of the patients was 52 years, and average disease duration was 21 years. The mean whole-brain volume was 1,100 mL. The mean EDSS score was 5.8, and the mean MSIS-29 score was 70.
Nine patients did not complete the full trial. Treatment compliance was 90% or greater for 77%-78% of the remaining patients.
At 24 months, whole-brain volume had declined in both groups. But the annual decline was significantly less in those taking simvastatin (–0.288% vs. –0.584%). The adjusted difference in annual atrophy was 0.254% per year, resulting in 43% less volume loss per year in the simvastatin group than in the placebo group. This effect was seen in MRI scans at both 12 and 24 months, and occurred in three-fourths of the treated population.
The rate of new or enlarging lesions was not significantly different, although there was a trend toward benefit in the treated group (1.50 vs. 2.19 per person per year). The relapse rates were similar (0.20 vs. 0.16 events per person/year).
At 24 months, the mean EDSS score in the simvastatin group was significantly lower than in the placebo group (a mean difference of –0.254 after adjustment for baseline measurements). The total mean MSIS-29 also showed a significant between-group difference in favor of simvastatin (–4.78 after adjustment). The difference was most pronounced in the physical subscale of the MSIS-29 (–3.73), but the psychological subscale was not significantly different. Overall, changes in these secondary measures by 24 months reflected worsening in both groups, but more so in the placebo group.
Cholesterol also decreased significantly in the simvastatin group (from 5.5 mmol/L to 4.1 mmol/L); it did not change in the placebo group. However, there were no differences in inflammatory markers.
Drug-related adverse events occurred in 23% of patients on simvastatin, compared with 19% of those on placebo. Simvastatin was generally well tolerated, with no safety issues.
It’s not entirely clear how a statin would slow disease progression, the researchers said. "gAccumulating evidence shows that statins have cell protective properties. For example, inhibition of inducible nitric oxide synthase, thus reducing release of free radicals from activated microglia and astrocytes or exerting a neuroprotective effect by prevention of glutamate-mediated excitotoxic effects."h
Simvastatin might also improve cerebral vasomotor reactivity, which could protect against hypoxic damage, they added. The general benefit of statins on vascular health could also be a contributor.
The study was partially supported by a grant from the U.K. National Institute for Health Research, the Moulton Foundation, the Berkeley Foundation, the Multiple Sclerosis Trials Collaboration, the Rosetrees Trust, and a personal contribution.
None of the authors had any financial disclosures.
On Twitter @alz_gal
FROM THE LANCET
Major finding: The annual decline in whole-brain volume was significantly less in those taking simvastatin (–0.288% vs. –0.584%). The adjusted difference in annual atrophy was 0.254% per year.
Data source: A phase II, randomized, placebo-controlled trial of 140 patients.
Disclosures: The study was partially supported by a grant from the U.K. National Institute for Health Research, the Moulton Foundation, the Berkeley Foundation, the Multiple Sclerosis Trials Collaboration, the Rosetrees Trust, and a personal contribution.
Autoimmune disease coalition seeks to increase physician knowledge
WASHINGTON – Some 64% of family physicians are "uncomfortable" or "stressed" when diagnosing autoimmune disease, and almost three-quarters said they have not been given adequate training in diagnosing and treating the conditions, according to a small survey.
The survey of 130 family physicians was conducted by the American Autoimmune Related Diseases Association (AARDA) last fall. The association has queried physicians each year since the mid-1990s on a variety of issues relating to the care and treatment of patients with any one of the 100 or so diseases that fall into the autoimmune category.
The AARDA, along with the National Coalition of Autoimmune Patient Groups, is pushing for more comprehensive autoimmune disorder centers where patients can receive focused and coordinated care from specialists who are more intimately involved with the diseases.
The autoimmune facilities would be modeled on comprehensive cancer centers.
Now, patients struggle to find specialists who can accurately diagnose and treat their conditions. "There’s no such thing as the autoimmunologist," said Stanley Finger, Ph.D., the AARDA’s vice chairman of the board, at a briefing.
Patients responding to AARDA surveys report that it takes 4-5 years to get an accurate diagnosis, and that they see an average of five physicians before they get that diagnosis. At least half of patients are labeled chronic complainers and told that their symptoms are figments of their imagination, Dr. Finger said.
But 75% say they would seek care at a specialized center if it existed.
Improving diagnosis also requires increasing physician awareness and education. In the AARDA’s most recent survey, almost 60% of family physicians said that they had only one or two lectures on autoimmune disease in medical school, said Dr. Finger, who is also president of Environmental Consulting and Investigations in Bluffton, S.C.
"It doesn’t give a lot of time for these physicians to become experts," Dr. Finger said. "Because of that, they don’t feel very good about the training they have received."
The AARDA plans to develop a syllabus for medical schools and a continuing education program to help fill physicians’ knowledge gaps.
The group also surveys about 1,000 members of the general public every 5-7 years to gauge awareness of how patients are interacting with physicians. In 1992, only 5% could name an autoimmune disease. That has increased, but only to 15%. In the first survey, 93% of the public thought AIDS was an autoimmune disease. Now, just 21% have that belief.
Borrowing another page from the cancer model, the AARDA and the coalition are seeking to establish an autoimmune disease registry that would be similar to the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program. The SEER program compiles data about cancer incidence, mortality, and cost. It is widely used by researchers, patients, physicians, and public health agencies.
The autoimmune registry is still a work in progress. There is a huge absence of data in the autoimmune field – for example, no one knows with certainty just how many Americans have any of the various conditions, said Aaron H. Abend, the AARDA’s informatics director.
The registry would aggregate data already being compiled by various individual autoimmune associations. But the effort is in its infancy. The groups still need to agree on governance, data protocols, and other issues, Mr. Abend said. However, the registry will use software that is the standard for registries operated by the National Institutes of Health.
aault@frontlinemedcom.com
On Twitter @aliciaault
WASHINGTON – Some 64% of family physicians are "uncomfortable" or "stressed" when diagnosing autoimmune disease, and almost three-quarters said they have not been given adequate training in diagnosing and treating the conditions, according to a small survey.
The survey of 130 family physicians was conducted by the American Autoimmune Related Diseases Association (AARDA) last fall. The association has queried physicians each year since the mid-1990s on a variety of issues relating to the care and treatment of patients with any one of the 100 or so diseases that fall into the autoimmune category.
The AARDA, along with the National Coalition of Autoimmune Patient Groups, is pushing for more comprehensive autoimmune disorder centers where patients can receive focused and coordinated care from specialists who are more intimately involved with the diseases.
The autoimmune facilities would be modeled on comprehensive cancer centers.
Now, patients struggle to find specialists who can accurately diagnose and treat their conditions. "There’s no such thing as the autoimmunologist," said Stanley Finger, Ph.D., the AARDA’s vice chairman of the board, at a briefing.
Patients responding to AARDA surveys report that it takes 4-5 years to get an accurate diagnosis, and that they see an average of five physicians before they get that diagnosis. At least half of patients are labeled chronic complainers and told that their symptoms are figments of their imagination, Dr. Finger said.
But 75% say they would seek care at a specialized center if it existed.
Improving diagnosis also requires increasing physician awareness and education. In the AARDA’s most recent survey, almost 60% of family physicians said that they had only one or two lectures on autoimmune disease in medical school, said Dr. Finger, who is also president of Environmental Consulting and Investigations in Bluffton, S.C.
"It doesn’t give a lot of time for these physicians to become experts," Dr. Finger said. "Because of that, they don’t feel very good about the training they have received."
The AARDA plans to develop a syllabus for medical schools and a continuing education program to help fill physicians’ knowledge gaps.
The group also surveys about 1,000 members of the general public every 5-7 years to gauge awareness of how patients are interacting with physicians. In 1992, only 5% could name an autoimmune disease. That has increased, but only to 15%. In the first survey, 93% of the public thought AIDS was an autoimmune disease. Now, just 21% have that belief.
Borrowing another page from the cancer model, the AARDA and the coalition are seeking to establish an autoimmune disease registry that would be similar to the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program. The SEER program compiles data about cancer incidence, mortality, and cost. It is widely used by researchers, patients, physicians, and public health agencies.
The autoimmune registry is still a work in progress. There is a huge absence of data in the autoimmune field – for example, no one knows with certainty just how many Americans have any of the various conditions, said Aaron H. Abend, the AARDA’s informatics director.
The registry would aggregate data already being compiled by various individual autoimmune associations. But the effort is in its infancy. The groups still need to agree on governance, data protocols, and other issues, Mr. Abend said. However, the registry will use software that is the standard for registries operated by the National Institutes of Health.
aault@frontlinemedcom.com
On Twitter @aliciaault
WASHINGTON – Some 64% of family physicians are "uncomfortable" or "stressed" when diagnosing autoimmune disease, and almost three-quarters said they have not been given adequate training in diagnosing and treating the conditions, according to a small survey.
The survey of 130 family physicians was conducted by the American Autoimmune Related Diseases Association (AARDA) last fall. The association has queried physicians each year since the mid-1990s on a variety of issues relating to the care and treatment of patients with any one of the 100 or so diseases that fall into the autoimmune category.
The AARDA, along with the National Coalition of Autoimmune Patient Groups, is pushing for more comprehensive autoimmune disorder centers where patients can receive focused and coordinated care from specialists who are more intimately involved with the diseases.
The autoimmune facilities would be modeled on comprehensive cancer centers.
Now, patients struggle to find specialists who can accurately diagnose and treat their conditions. "There’s no such thing as the autoimmunologist," said Stanley Finger, Ph.D., the AARDA’s vice chairman of the board, at a briefing.
Patients responding to AARDA surveys report that it takes 4-5 years to get an accurate diagnosis, and that they see an average of five physicians before they get that diagnosis. At least half of patients are labeled chronic complainers and told that their symptoms are figments of their imagination, Dr. Finger said.
But 75% say they would seek care at a specialized center if it existed.
Improving diagnosis also requires increasing physician awareness and education. In the AARDA’s most recent survey, almost 60% of family physicians said that they had only one or two lectures on autoimmune disease in medical school, said Dr. Finger, who is also president of Environmental Consulting and Investigations in Bluffton, S.C.
"It doesn’t give a lot of time for these physicians to become experts," Dr. Finger said. "Because of that, they don’t feel very good about the training they have received."
The AARDA plans to develop a syllabus for medical schools and a continuing education program to help fill physicians’ knowledge gaps.
The group also surveys about 1,000 members of the general public every 5-7 years to gauge awareness of how patients are interacting with physicians. In 1992, only 5% could name an autoimmune disease. That has increased, but only to 15%. In the first survey, 93% of the public thought AIDS was an autoimmune disease. Now, just 21% have that belief.
Borrowing another page from the cancer model, the AARDA and the coalition are seeking to establish an autoimmune disease registry that would be similar to the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) program. The SEER program compiles data about cancer incidence, mortality, and cost. It is widely used by researchers, patients, physicians, and public health agencies.
The autoimmune registry is still a work in progress. There is a huge absence of data in the autoimmune field – for example, no one knows with certainty just how many Americans have any of the various conditions, said Aaron H. Abend, the AARDA’s informatics director.
The registry would aggregate data already being compiled by various individual autoimmune associations. But the effort is in its infancy. The groups still need to agree on governance, data protocols, and other issues, Mr. Abend said. However, the registry will use software that is the standard for registries operated by the National Institutes of Health.
aault@frontlinemedcom.com
On Twitter @aliciaault
FROM A MEDIA BRIEFING BY THE AMERICAN AUTOIMMUNE RELATED DISEASES ASSOCIATION
Pseudobulbar affect: More common than you’d think
ORLANDO – The prevalence of pseudobulbar affect symptoms – that is, uncontrollable, disruptive outbursts of crying and/or laughing – is considerably greater across a range of neurologic disorders than previously appreciated, according to the largest-ever study to screen for this condition.
Pseudobulbar affect (PBA) symptoms were found in the study to be more common among neurology patients under age 65; however, the adverse impact of PBA symptoms upon quality of life was greater in the elderly, Dr. David W. Crumpacker reported at the annual meeting of the American Association for Geriatric Psychiatry.
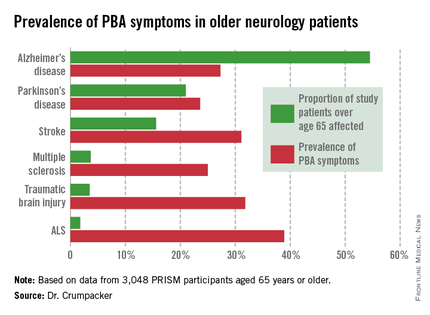
He presented the results of the PRISM (PBA Registry Series) study, which enrolled 5,290 patients on the basis of having any of six neurologic disorders: Alzheimer’s disease, amyotrophic lateral sclerosis (ALS), multiple sclerosis, Parkinson’s disease, stroke, or traumatic brain injury. They were screened for the presence of PBA symptoms using the validated Center for Neurologic Study–Lability Scale (CNS-LS). A score of 13 or more was deemed positive, based upon its demonstrated good predictive value for physician diagnosis of PBA in patients with ALS.
The CNS-LS is a simple test that can be completed quickly by either the patient or caregiver. The test is well-suited for routine use in clinical practice, noted Dr. W. Crumpacker, a psychiatrist at Baylor University Medical Center, Dallas.
The overall prevalence of PBA symptoms among the 3,048 PRISM participants aged 65 years or older was 27.4%, with the highest rate seen in patients having ALS (see chart). In contrast, the prevalence of PBA symptoms among patients under age 65 years was 49.5%, with the highest rate – 56.9% – being seen in traumatic brain injury patients.
Patients or caregivers were asked to rate on a 0-10 scale the impact their primary neurologic disease has had on their quality of life. Patients 65 years and older with PBA symptoms reported a significantly greater negative impact than did those without PBA symptoms, with mean scores on the quality of life impact scale of 6.3 vs. 4.6. The quality of life difference between those with PBA symptoms and those without was significant for patients with each of the neurologic diseases except for ALS.
As another measure of the adverse impact of having PBA symptoms, 56% of affected older patients were on at least one antipsychotic or antidepressant, compared with 35% of older patients without PBA symptoms.
PBA is thought to result from injury to neurologic pathways that regulate emotional expression as a secondary consequence of a variety of neurologic disorders.
In an interview, Dr. Crumpacker said PBA is greatly underdiagnosed and often gets misdiagnosed as depression.
"The symptoms are extremely disturbing to others, and patients are acutely aware of that. I tell my friends in neurology, it’s the psychiatric pathology that causes people problems in their lives. No one gets divorced over neurologic pathology, they get divorced over psychiatric pathology. It’s not, ‘I got a divorce because he had a stroke.’ " "It’s "We got divorced because he had a stroke and it changed his personality; he was a different person and I couldn’t be around him anymore,’ " the psychiatrist said.
PBA became a diagnosable disorder with its own ICD-9 code, albeit a diagnosis that can’t be made in the absence of neurologic pathology, at the behest of the Food and Drug Administration, Dr. Crumpacker explained. The impetus was the discovery of an effective treatment, dextromethorphan HBr and quinidine sulfate (Nuedexta), which received FDA approval for PBA 3 years ago.
Nuedexta’s development as the sole medication indicated for PBA was serendipitous, according to Dr. Crumpacker.
"The drug was being tested in Alzheimer’s disease. The jury is still out on whether it helps. But families of study participants came back saying, ‘You know that stuff dad used to do – the crying, the inappropriate laughter, the anger? He doesn’t do those kinds of things anymore,’ " Dr. Crumpacker recalled.
He reported serving on a scientific advisory board for Avanir Pharmaceuticals, which markets Nuedexta.
ORLANDO – The prevalence of pseudobulbar affect symptoms – that is, uncontrollable, disruptive outbursts of crying and/or laughing – is considerably greater across a range of neurologic disorders than previously appreciated, according to the largest-ever study to screen for this condition.
Pseudobulbar affect (PBA) symptoms were found in the study to be more common among neurology patients under age 65; however, the adverse impact of PBA symptoms upon quality of life was greater in the elderly, Dr. David W. Crumpacker reported at the annual meeting of the American Association for Geriatric Psychiatry.

He presented the results of the PRISM (PBA Registry Series) study, which enrolled 5,290 patients on the basis of having any of six neurologic disorders: Alzheimer’s disease, amyotrophic lateral sclerosis (ALS), multiple sclerosis, Parkinson’s disease, stroke, or traumatic brain injury. They were screened for the presence of PBA symptoms using the validated Center for Neurologic Study–Lability Scale (CNS-LS). A score of 13 or more was deemed positive, based upon its demonstrated good predictive value for physician diagnosis of PBA in patients with ALS.
The CNS-LS is a simple test that can be completed quickly by either the patient or caregiver. The test is well-suited for routine use in clinical practice, noted Dr. W. Crumpacker, a psychiatrist at Baylor University Medical Center, Dallas.
The overall prevalence of PBA symptoms among the 3,048 PRISM participants aged 65 years or older was 27.4%, with the highest rate seen in patients having ALS (see chart). In contrast, the prevalence of PBA symptoms among patients under age 65 years was 49.5%, with the highest rate – 56.9% – being seen in traumatic brain injury patients.
Patients or caregivers were asked to rate on a 0-10 scale the impact their primary neurologic disease has had on their quality of life. Patients 65 years and older with PBA symptoms reported a significantly greater negative impact than did those without PBA symptoms, with mean scores on the quality of life impact scale of 6.3 vs. 4.6. The quality of life difference between those with PBA symptoms and those without was significant for patients with each of the neurologic diseases except for ALS.
As another measure of the adverse impact of having PBA symptoms, 56% of affected older patients were on at least one antipsychotic or antidepressant, compared with 35% of older patients without PBA symptoms.
PBA is thought to result from injury to neurologic pathways that regulate emotional expression as a secondary consequence of a variety of neurologic disorders.
In an interview, Dr. Crumpacker said PBA is greatly underdiagnosed and often gets misdiagnosed as depression.
"The symptoms are extremely disturbing to others, and patients are acutely aware of that. I tell my friends in neurology, it’s the psychiatric pathology that causes people problems in their lives. No one gets divorced over neurologic pathology, they get divorced over psychiatric pathology. It’s not, ‘I got a divorce because he had a stroke.’ " "It’s "We got divorced because he had a stroke and it changed his personality; he was a different person and I couldn’t be around him anymore,’ " the psychiatrist said.
PBA became a diagnosable disorder with its own ICD-9 code, albeit a diagnosis that can’t be made in the absence of neurologic pathology, at the behest of the Food and Drug Administration, Dr. Crumpacker explained. The impetus was the discovery of an effective treatment, dextromethorphan HBr and quinidine sulfate (Nuedexta), which received FDA approval for PBA 3 years ago.
Nuedexta’s development as the sole medication indicated for PBA was serendipitous, according to Dr. Crumpacker.
"The drug was being tested in Alzheimer’s disease. The jury is still out on whether it helps. But families of study participants came back saying, ‘You know that stuff dad used to do – the crying, the inappropriate laughter, the anger? He doesn’t do those kinds of things anymore,’ " Dr. Crumpacker recalled.
He reported serving on a scientific advisory board for Avanir Pharmaceuticals, which markets Nuedexta.
ORLANDO – The prevalence of pseudobulbar affect symptoms – that is, uncontrollable, disruptive outbursts of crying and/or laughing – is considerably greater across a range of neurologic disorders than previously appreciated, according to the largest-ever study to screen for this condition.
Pseudobulbar affect (PBA) symptoms were found in the study to be more common among neurology patients under age 65; however, the adverse impact of PBA symptoms upon quality of life was greater in the elderly, Dr. David W. Crumpacker reported at the annual meeting of the American Association for Geriatric Psychiatry.

He presented the results of the PRISM (PBA Registry Series) study, which enrolled 5,290 patients on the basis of having any of six neurologic disorders: Alzheimer’s disease, amyotrophic lateral sclerosis (ALS), multiple sclerosis, Parkinson’s disease, stroke, or traumatic brain injury. They were screened for the presence of PBA symptoms using the validated Center for Neurologic Study–Lability Scale (CNS-LS). A score of 13 or more was deemed positive, based upon its demonstrated good predictive value for physician diagnosis of PBA in patients with ALS.
The CNS-LS is a simple test that can be completed quickly by either the patient or caregiver. The test is well-suited for routine use in clinical practice, noted Dr. W. Crumpacker, a psychiatrist at Baylor University Medical Center, Dallas.
The overall prevalence of PBA symptoms among the 3,048 PRISM participants aged 65 years or older was 27.4%, with the highest rate seen in patients having ALS (see chart). In contrast, the prevalence of PBA symptoms among patients under age 65 years was 49.5%, with the highest rate – 56.9% – being seen in traumatic brain injury patients.
Patients or caregivers were asked to rate on a 0-10 scale the impact their primary neurologic disease has had on their quality of life. Patients 65 years and older with PBA symptoms reported a significantly greater negative impact than did those without PBA symptoms, with mean scores on the quality of life impact scale of 6.3 vs. 4.6. The quality of life difference between those with PBA symptoms and those without was significant for patients with each of the neurologic diseases except for ALS.
As another measure of the adverse impact of having PBA symptoms, 56% of affected older patients were on at least one antipsychotic or antidepressant, compared with 35% of older patients without PBA symptoms.
PBA is thought to result from injury to neurologic pathways that regulate emotional expression as a secondary consequence of a variety of neurologic disorders.
In an interview, Dr. Crumpacker said PBA is greatly underdiagnosed and often gets misdiagnosed as depression.
"The symptoms are extremely disturbing to others, and patients are acutely aware of that. I tell my friends in neurology, it’s the psychiatric pathology that causes people problems in their lives. No one gets divorced over neurologic pathology, they get divorced over psychiatric pathology. It’s not, ‘I got a divorce because he had a stroke.’ " "It’s "We got divorced because he had a stroke and it changed his personality; he was a different person and I couldn’t be around him anymore,’ " the psychiatrist said.
PBA became a diagnosable disorder with its own ICD-9 code, albeit a diagnosis that can’t be made in the absence of neurologic pathology, at the behest of the Food and Drug Administration, Dr. Crumpacker explained. The impetus was the discovery of an effective treatment, dextromethorphan HBr and quinidine sulfate (Nuedexta), which received FDA approval for PBA 3 years ago.
Nuedexta’s development as the sole medication indicated for PBA was serendipitous, according to Dr. Crumpacker.
"The drug was being tested in Alzheimer’s disease. The jury is still out on whether it helps. But families of study participants came back saying, ‘You know that stuff dad used to do – the crying, the inappropriate laughter, the anger? He doesn’t do those kinds of things anymore,’ " Dr. Crumpacker recalled.
He reported serving on a scientific advisory board for Avanir Pharmaceuticals, which markets Nuedexta.
AT THE AAGP ANNUAL MEETING
Major finding: The prevalence of PBA symptoms among patients over age 65 years with any of six underlying neurologic disorders was 27.4%. That was significantly less than in younger patients with the same disorders, but the adverse effect of having PBA symptoms upon quality of life was markedly greater in the older group.
Data source: The PRISM study included 5,290 patients with Alzheimer’s disease or any of five other less common neurologic disorders, all of whom were screened for the presence of PBA symptoms using a brief validated measure.
Disclosures: The presenter serves on a scientific advisory board for Avanir Pharmaceuticals, which funded the PRISM study.
FDA extends review of pegylated interferon for MS, Biogen says
The Food and Drug Administration review of a pegylated interferon for multiple sclerosis has been extended by 3 months, according to the manufacturer.
A statement issued by Biogen Idec on March 18 said that the agency had extended the Prescription Drug User Fee Act (PDUFA) date for the product’s review by 3 months, "to allow additional time for review of the application." The agency is not requesting additional studies, the statement said.
The product is a subcutaneously administered formulation of pegylated interferon beta-1a for treating relapsing forms of MS. If the agent is approved, Biogen plans to market it as Plegridy.
In the ADVANCE trial of about 1,500 patients with relapsing-remitting MS, treatment with this pegylated interferon administered every 2 weeks was associated with statistically significant advantages for all clinical and radiologic outcomes, compared with placebo, over 1 year.
The product is also being reviewed in Europe for the same indication.
The Food and Drug Administration review of a pegylated interferon for multiple sclerosis has been extended by 3 months, according to the manufacturer.
A statement issued by Biogen Idec on March 18 said that the agency had extended the Prescription Drug User Fee Act (PDUFA) date for the product’s review by 3 months, "to allow additional time for review of the application." The agency is not requesting additional studies, the statement said.
The product is a subcutaneously administered formulation of pegylated interferon beta-1a for treating relapsing forms of MS. If the agent is approved, Biogen plans to market it as Plegridy.
In the ADVANCE trial of about 1,500 patients with relapsing-remitting MS, treatment with this pegylated interferon administered every 2 weeks was associated with statistically significant advantages for all clinical and radiologic outcomes, compared with placebo, over 1 year.
The product is also being reviewed in Europe for the same indication.
The Food and Drug Administration review of a pegylated interferon for multiple sclerosis has been extended by 3 months, according to the manufacturer.
A statement issued by Biogen Idec on March 18 said that the agency had extended the Prescription Drug User Fee Act (PDUFA) date for the product’s review by 3 months, "to allow additional time for review of the application." The agency is not requesting additional studies, the statement said.
The product is a subcutaneously administered formulation of pegylated interferon beta-1a for treating relapsing forms of MS. If the agent is approved, Biogen plans to market it as Plegridy.
In the ADVANCE trial of about 1,500 patients with relapsing-remitting MS, treatment with this pegylated interferon administered every 2 weeks was associated with statistically significant advantages for all clinical and radiologic outcomes, compared with placebo, over 1 year.
The product is also being reviewed in Europe for the same indication.
Results finally published for failed MS trial with B-cell targeting drug atacicept
Atacicept, a drug designed to interfere with B-cell differentiation and maturation, was associated with up to twice as many relapses as placebo among patients with multiple sclerosis in the 36-week, phase II ATAMS (Atacicept in Multiple Sclerosis Extension Study).
Interim findings were enough to halt the drug’s placebo-controlled dose-ranging study, Dr. Ludwig Kappos of the University Hospital Basel, Switzerland, and his colleagues reported (Lancet Neurol. 2014 [doi: 10.1016/S1474-4422(14)70028-6]). The trial results were originally reported at the 2011 joint triennial congress of the European and Americas Committees for Treatment and Research in Multiple Sclerosis.
The increase in disease activity was surprising, wrote Dr. Kappos and his coauthors, especially because atacicept did not cause such problems in other studies.
"Studies of atacicept in other autoimmune diseases, including rheumatoid arthritis and systemic lupus erythematosus, had not suggested that such B-cell-targeted interventions might enhance, rather than suppress, inflammatory activity," they said.
"Our finding of an early and robust increase in relapse activity with atacicept treatment in a typical population with relapsing-remitting multiple sclerosis was therefore unexpected. This increase in clinical disease activity occurred in parallel with reductions in serum immunoglobulin concentrations and mature B-cell counts of the same dynamics and degree as reported in previous atacicept studies."
Fortunately, the authors noted, the relapses didn’t translate into any increase in disease severity, and measurements of all the affected immune factors returned to normal shortly after treatment ceased.
In an editorial accompanying the published paper, Dr. Fred Lühder and Dr. Ralf Gold wrote that there’s little doubt that B cells are a valid therapeutic target in multiple sclerosis. But they seem to exist in a fine-tune balance that, if disrupted, can harm, rather than help (Lancet Neurol. 2014 March 6 [doi: 10.1016/S1474-4422(14)70050-X]).
Atacicept binds to B-lymphocyte stimulator (BLyS), a cytokine that helps control B-cell differentiation, maturation, and survival. But unlike other B-cell modulators, it doesn’t kill all of them immediately. In ATAMS, the maximum reduction was 60%-70%.
"A probably more important issue is the types of B cell that are preferentially depleted," Dr. Lühder and Dr. Gold wrote. "Experimental evidence in animal models suggests that B cells potentially have a dual role in the pathogenesis of neuroinflammation. ... Spontaneous demyelinating disease occurs when both receptors are brought together, suggesting that antigen-specific B-cells could provide the essential stimulus for antigen-specific T-cells."
The second type of B cells, which produce interleukin-10, exert a regulatory effect, controlling the action of activated T cells, and thus reduce disease severity.
"Since BLyS is believed to be involved in the differentiation of these regulatory B-cells, the targeting of BLyS might disturb the fine-tuned balance of conventional and regulatory B-cells in favor of the conventional cells, eventually resulting in increased disease activity, as seen in ATAMS," they wrote.
Researchers will learn as much from this failed trial as they would from any successful trial, Dr. Lühder and Dr. Gold said. "Many other candidates have gone the same way. B-cells should still be regarded as a valid target, but there are good and bad B-cells in multiple sclerosis, and the net effect of a particular treatment on this complex scenario can be unpredictable."
Dr. Lühder is with the neuroimmunology department at the Institute for Multiple Sclerosis Research and the Max Planck Institute for Experimental Medicine at the University of Göttingen, Germany. Dr. Gold is with the department of neurology, St. Josef-Hospital, Ruhr-University Bochum, Germany.
The study was sponsored by Merck Serono. Dr. Kappos’s institution has received Merck grant money. Most of the coauthors reported numerous financial disclosures, including financial relationships with Merck Serono.
Atacicept, a drug designed to interfere with B-cell differentiation and maturation, was associated with up to twice as many relapses as placebo among patients with multiple sclerosis in the 36-week, phase II ATAMS (Atacicept in Multiple Sclerosis Extension Study).
Interim findings were enough to halt the drug’s placebo-controlled dose-ranging study, Dr. Ludwig Kappos of the University Hospital Basel, Switzerland, and his colleagues reported (Lancet Neurol. 2014 [doi: 10.1016/S1474-4422(14)70028-6]). The trial results were originally reported at the 2011 joint triennial congress of the European and Americas Committees for Treatment and Research in Multiple Sclerosis.
The increase in disease activity was surprising, wrote Dr. Kappos and his coauthors, especially because atacicept did not cause such problems in other studies.
"Studies of atacicept in other autoimmune diseases, including rheumatoid arthritis and systemic lupus erythematosus, had not suggested that such B-cell-targeted interventions might enhance, rather than suppress, inflammatory activity," they said.
"Our finding of an early and robust increase in relapse activity with atacicept treatment in a typical population with relapsing-remitting multiple sclerosis was therefore unexpected. This increase in clinical disease activity occurred in parallel with reductions in serum immunoglobulin concentrations and mature B-cell counts of the same dynamics and degree as reported in previous atacicept studies."
Fortunately, the authors noted, the relapses didn’t translate into any increase in disease severity, and measurements of all the affected immune factors returned to normal shortly after treatment ceased.
In an editorial accompanying the published paper, Dr. Fred Lühder and Dr. Ralf Gold wrote that there’s little doubt that B cells are a valid therapeutic target in multiple sclerosis. But they seem to exist in a fine-tune balance that, if disrupted, can harm, rather than help (Lancet Neurol. 2014 March 6 [doi: 10.1016/S1474-4422(14)70050-X]).
Atacicept binds to B-lymphocyte stimulator (BLyS), a cytokine that helps control B-cell differentiation, maturation, and survival. But unlike other B-cell modulators, it doesn’t kill all of them immediately. In ATAMS, the maximum reduction was 60%-70%.
"A probably more important issue is the types of B cell that are preferentially depleted," Dr. Lühder and Dr. Gold wrote. "Experimental evidence in animal models suggests that B cells potentially have a dual role in the pathogenesis of neuroinflammation. ... Spontaneous demyelinating disease occurs when both receptors are brought together, suggesting that antigen-specific B-cells could provide the essential stimulus for antigen-specific T-cells."
The second type of B cells, which produce interleukin-10, exert a regulatory effect, controlling the action of activated T cells, and thus reduce disease severity.
"Since BLyS is believed to be involved in the differentiation of these regulatory B-cells, the targeting of BLyS might disturb the fine-tuned balance of conventional and regulatory B-cells in favor of the conventional cells, eventually resulting in increased disease activity, as seen in ATAMS," they wrote.
Researchers will learn as much from this failed trial as they would from any successful trial, Dr. Lühder and Dr. Gold said. "Many other candidates have gone the same way. B-cells should still be regarded as a valid target, but there are good and bad B-cells in multiple sclerosis, and the net effect of a particular treatment on this complex scenario can be unpredictable."
Dr. Lühder is with the neuroimmunology department at the Institute for Multiple Sclerosis Research and the Max Planck Institute for Experimental Medicine at the University of Göttingen, Germany. Dr. Gold is with the department of neurology, St. Josef-Hospital, Ruhr-University Bochum, Germany.
The study was sponsored by Merck Serono. Dr. Kappos’s institution has received Merck grant money. Most of the coauthors reported numerous financial disclosures, including financial relationships with Merck Serono.
Atacicept, a drug designed to interfere with B-cell differentiation and maturation, was associated with up to twice as many relapses as placebo among patients with multiple sclerosis in the 36-week, phase II ATAMS (Atacicept in Multiple Sclerosis Extension Study).
Interim findings were enough to halt the drug’s placebo-controlled dose-ranging study, Dr. Ludwig Kappos of the University Hospital Basel, Switzerland, and his colleagues reported (Lancet Neurol. 2014 [doi: 10.1016/S1474-4422(14)70028-6]). The trial results were originally reported at the 2011 joint triennial congress of the European and Americas Committees for Treatment and Research in Multiple Sclerosis.
The increase in disease activity was surprising, wrote Dr. Kappos and his coauthors, especially because atacicept did not cause such problems in other studies.
"Studies of atacicept in other autoimmune diseases, including rheumatoid arthritis and systemic lupus erythematosus, had not suggested that such B-cell-targeted interventions might enhance, rather than suppress, inflammatory activity," they said.
"Our finding of an early and robust increase in relapse activity with atacicept treatment in a typical population with relapsing-remitting multiple sclerosis was therefore unexpected. This increase in clinical disease activity occurred in parallel with reductions in serum immunoglobulin concentrations and mature B-cell counts of the same dynamics and degree as reported in previous atacicept studies."
Fortunately, the authors noted, the relapses didn’t translate into any increase in disease severity, and measurements of all the affected immune factors returned to normal shortly after treatment ceased.
In an editorial accompanying the published paper, Dr. Fred Lühder and Dr. Ralf Gold wrote that there’s little doubt that B cells are a valid therapeutic target in multiple sclerosis. But they seem to exist in a fine-tune balance that, if disrupted, can harm, rather than help (Lancet Neurol. 2014 March 6 [doi: 10.1016/S1474-4422(14)70050-X]).
Atacicept binds to B-lymphocyte stimulator (BLyS), a cytokine that helps control B-cell differentiation, maturation, and survival. But unlike other B-cell modulators, it doesn’t kill all of them immediately. In ATAMS, the maximum reduction was 60%-70%.
"A probably more important issue is the types of B cell that are preferentially depleted," Dr. Lühder and Dr. Gold wrote. "Experimental evidence in animal models suggests that B cells potentially have a dual role in the pathogenesis of neuroinflammation. ... Spontaneous demyelinating disease occurs when both receptors are brought together, suggesting that antigen-specific B-cells could provide the essential stimulus for antigen-specific T-cells."
The second type of B cells, which produce interleukin-10, exert a regulatory effect, controlling the action of activated T cells, and thus reduce disease severity.
"Since BLyS is believed to be involved in the differentiation of these regulatory B-cells, the targeting of BLyS might disturb the fine-tuned balance of conventional and regulatory B-cells in favor of the conventional cells, eventually resulting in increased disease activity, as seen in ATAMS," they wrote.
Researchers will learn as much from this failed trial as they would from any successful trial, Dr. Lühder and Dr. Gold said. "Many other candidates have gone the same way. B-cells should still be regarded as a valid target, but there are good and bad B-cells in multiple sclerosis, and the net effect of a particular treatment on this complex scenario can be unpredictable."
Dr. Lühder is with the neuroimmunology department at the Institute for Multiple Sclerosis Research and the Max Planck Institute for Experimental Medicine at the University of Göttingen, Germany. Dr. Gold is with the department of neurology, St. Josef-Hospital, Ruhr-University Bochum, Germany.
The study was sponsored by Merck Serono. Dr. Kappos’s institution has received Merck grant money. Most of the coauthors reported numerous financial disclosures, including financial relationships with Merck Serono.
FROM LANCET NEUROLOGY
MS relapse rates higher with 2-month washout before switch to fingolimod
Switching from natalizumab to fingolimod kept 85% of multiple sclerosis patients relapse-free for at least 6 months in an observational cohort study.
Although relapse rates remained low after patients started fingolimod, patients who had a gap of 2-4 months between cessation of prior treatment and commencement of fingolimod had significantly higher relapse rates than did those with no treatment gap.
"The main risk factor for time to relapse on fingolimod was recent prior relapse," said Dr. Vilija Jokubaitis of the University of Melbourne and her associates. "Our data support choosing a short switch period (2 months or less) between prior treatment and fingolimod to decrease the hazard of relapse," they added.
Because of concerns about progressive multifocal leukoencephalopathy (PML), patients with multiple sclerosis (MS) often are switched to fingolimod (Gilenya) if they have taken natalizumab (Tysabri) for more than 24 months and test positive for anti-JC virus antibodies. To evaluate the effects of switching on MS relapse, the researchers studied 536 patients from the MSBase Registry who took fingolimod as initial therapy (n = 97), switched to fingolimod after failing natalizumab (n = 89), or switched to fingolimod from interferon-beta/glatiramer acetate (n = 350). The median washout period for patients switching from natalizumab to fingolimod was 79 days (interquartile range, 57-96 days), and the median follow-up for all patients was 10.3 months, the investigators reported (Neurology 2014 March 7 [doi: 10.1212/WNL.0000000000000283]).
Relapse rates were "relatively stable" among the patients who switched from natalizumab to fingolimod during the first 9 months on fingolimod (quarterly relapse rate range, 0.079-0.13), compared with the 15-month period before starting fingolimod (quarterly relapse rate range, 0.05-0.11). Patients who switched from natalizumab to fingolimod had a small increase in annualized relapse rate (0.38 vs. 0.26 for natalizumab; P = .002), which the investigators attributed to possible differences in efficacy between the two medications. In survival analyses, predictors of time to first relapse on fingolimod included number of relapses in the past 6 months (hazard ratio, 1.60 per relapse; P = .002) and a treatment gap of 2-4 months vs. no gap (HR, 2.10; P = .041).
"In this study, the largest of its kind to date, we found no evidence to support the occurrence of clinical rebound in patients switching from natalizumab to fingolimod," wrote Dr. Jokubaitis and her associates. Relapse activity was well-controlled in this patient group, and it was similar to relapse activity in patients switching to fingolimod from interferon-beta/glatiramer acetate or those starting fingolimod as their first disease-modifying therapy for MS.
The study was funded by a grant from the Australian National Health and Medical Research Council and by the MSBase Foundation. Dr. Jokubaitis and 13 of her associates reported having received honoraria or other support from Novartis, which makes fingolimod. Ten associates reported receiving honoraria or other support from Biogen Idec, which makes natalizumab.
The study "adds to an emerging literature suggesting that a prolonged washout does more harm than good" in MS patients who are switching therapies, said Dr. Gavin Giovannoni and Dr. Robert T. Naismith.
These and other recent results show that washout should be minimized and should not exceed 2 months in patients who switch to fingolimod, they said, adding that some studies supported no washout at all.
"Currently, the evidence-based harm of a break-through relapse causing sustained disability outweighs the theoretical concern that overlapping therapies will result in more PML. These studies indicate that fingolimod, when start-up is properly timed, can be effective post-natalizumab," they said.
The results also underscored the need for real-world patient registry studies and industry-sponsored switching studies to better elucidate how to tailor emerging MS therapies for individual patients, they said.
Dr. Giovannoni is a neurologist with Queen Mary University of London, Blizard Institute, Barts and The London School of Medicine and Dentistry. He reported receiving research funding from Novartis, the maker of fingolimod. Dr. Naismith is a neurologist at Washington University, St. Louis. He did not report relevant financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Jokubaitis’s report (Neurology 2014 March 7 [doi: 10.1212/WNL.0000000000000296]).
The study "adds to an emerging literature suggesting that a prolonged washout does more harm than good" in MS patients who are switching therapies, said Dr. Gavin Giovannoni and Dr. Robert T. Naismith.
These and other recent results show that washout should be minimized and should not exceed 2 months in patients who switch to fingolimod, they said, adding that some studies supported no washout at all.
"Currently, the evidence-based harm of a break-through relapse causing sustained disability outweighs the theoretical concern that overlapping therapies will result in more PML. These studies indicate that fingolimod, when start-up is properly timed, can be effective post-natalizumab," they said.
The results also underscored the need for real-world patient registry studies and industry-sponsored switching studies to better elucidate how to tailor emerging MS therapies for individual patients, they said.
Dr. Giovannoni is a neurologist with Queen Mary University of London, Blizard Institute, Barts and The London School of Medicine and Dentistry. He reported receiving research funding from Novartis, the maker of fingolimod. Dr. Naismith is a neurologist at Washington University, St. Louis. He did not report relevant financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Jokubaitis’s report (Neurology 2014 March 7 [doi: 10.1212/WNL.0000000000000296]).
The study "adds to an emerging literature suggesting that a prolonged washout does more harm than good" in MS patients who are switching therapies, said Dr. Gavin Giovannoni and Dr. Robert T. Naismith.
These and other recent results show that washout should be minimized and should not exceed 2 months in patients who switch to fingolimod, they said, adding that some studies supported no washout at all.
"Currently, the evidence-based harm of a break-through relapse causing sustained disability outweighs the theoretical concern that overlapping therapies will result in more PML. These studies indicate that fingolimod, when start-up is properly timed, can be effective post-natalizumab," they said.
The results also underscored the need for real-world patient registry studies and industry-sponsored switching studies to better elucidate how to tailor emerging MS therapies for individual patients, they said.
Dr. Giovannoni is a neurologist with Queen Mary University of London, Blizard Institute, Barts and The London School of Medicine and Dentistry. He reported receiving research funding from Novartis, the maker of fingolimod. Dr. Naismith is a neurologist at Washington University, St. Louis. He did not report relevant financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Jokubaitis’s report (Neurology 2014 March 7 [doi: 10.1212/WNL.0000000000000296]).
Switching from natalizumab to fingolimod kept 85% of multiple sclerosis patients relapse-free for at least 6 months in an observational cohort study.
Although relapse rates remained low after patients started fingolimod, patients who had a gap of 2-4 months between cessation of prior treatment and commencement of fingolimod had significantly higher relapse rates than did those with no treatment gap.
"The main risk factor for time to relapse on fingolimod was recent prior relapse," said Dr. Vilija Jokubaitis of the University of Melbourne and her associates. "Our data support choosing a short switch period (2 months or less) between prior treatment and fingolimod to decrease the hazard of relapse," they added.
Because of concerns about progressive multifocal leukoencephalopathy (PML), patients with multiple sclerosis (MS) often are switched to fingolimod (Gilenya) if they have taken natalizumab (Tysabri) for more than 24 months and test positive for anti-JC virus antibodies. To evaluate the effects of switching on MS relapse, the researchers studied 536 patients from the MSBase Registry who took fingolimod as initial therapy (n = 97), switched to fingolimod after failing natalizumab (n = 89), or switched to fingolimod from interferon-beta/glatiramer acetate (n = 350). The median washout period for patients switching from natalizumab to fingolimod was 79 days (interquartile range, 57-96 days), and the median follow-up for all patients was 10.3 months, the investigators reported (Neurology 2014 March 7 [doi: 10.1212/WNL.0000000000000283]).
Relapse rates were "relatively stable" among the patients who switched from natalizumab to fingolimod during the first 9 months on fingolimod (quarterly relapse rate range, 0.079-0.13), compared with the 15-month period before starting fingolimod (quarterly relapse rate range, 0.05-0.11). Patients who switched from natalizumab to fingolimod had a small increase in annualized relapse rate (0.38 vs. 0.26 for natalizumab; P = .002), which the investigators attributed to possible differences in efficacy between the two medications. In survival analyses, predictors of time to first relapse on fingolimod included number of relapses in the past 6 months (hazard ratio, 1.60 per relapse; P = .002) and a treatment gap of 2-4 months vs. no gap (HR, 2.10; P = .041).
"In this study, the largest of its kind to date, we found no evidence to support the occurrence of clinical rebound in patients switching from natalizumab to fingolimod," wrote Dr. Jokubaitis and her associates. Relapse activity was well-controlled in this patient group, and it was similar to relapse activity in patients switching to fingolimod from interferon-beta/glatiramer acetate or those starting fingolimod as their first disease-modifying therapy for MS.
The study was funded by a grant from the Australian National Health and Medical Research Council and by the MSBase Foundation. Dr. Jokubaitis and 13 of her associates reported having received honoraria or other support from Novartis, which makes fingolimod. Ten associates reported receiving honoraria or other support from Biogen Idec, which makes natalizumab.
Switching from natalizumab to fingolimod kept 85% of multiple sclerosis patients relapse-free for at least 6 months in an observational cohort study.
Although relapse rates remained low after patients started fingolimod, patients who had a gap of 2-4 months between cessation of prior treatment and commencement of fingolimod had significantly higher relapse rates than did those with no treatment gap.
"The main risk factor for time to relapse on fingolimod was recent prior relapse," said Dr. Vilija Jokubaitis of the University of Melbourne and her associates. "Our data support choosing a short switch period (2 months or less) between prior treatment and fingolimod to decrease the hazard of relapse," they added.
Because of concerns about progressive multifocal leukoencephalopathy (PML), patients with multiple sclerosis (MS) often are switched to fingolimod (Gilenya) if they have taken natalizumab (Tysabri) for more than 24 months and test positive for anti-JC virus antibodies. To evaluate the effects of switching on MS relapse, the researchers studied 536 patients from the MSBase Registry who took fingolimod as initial therapy (n = 97), switched to fingolimod after failing natalizumab (n = 89), or switched to fingolimod from interferon-beta/glatiramer acetate (n = 350). The median washout period for patients switching from natalizumab to fingolimod was 79 days (interquartile range, 57-96 days), and the median follow-up for all patients was 10.3 months, the investigators reported (Neurology 2014 March 7 [doi: 10.1212/WNL.0000000000000283]).
Relapse rates were "relatively stable" among the patients who switched from natalizumab to fingolimod during the first 9 months on fingolimod (quarterly relapse rate range, 0.079-0.13), compared with the 15-month period before starting fingolimod (quarterly relapse rate range, 0.05-0.11). Patients who switched from natalizumab to fingolimod had a small increase in annualized relapse rate (0.38 vs. 0.26 for natalizumab; P = .002), which the investigators attributed to possible differences in efficacy between the two medications. In survival analyses, predictors of time to first relapse on fingolimod included number of relapses in the past 6 months (hazard ratio, 1.60 per relapse; P = .002) and a treatment gap of 2-4 months vs. no gap (HR, 2.10; P = .041).
"In this study, the largest of its kind to date, we found no evidence to support the occurrence of clinical rebound in patients switching from natalizumab to fingolimod," wrote Dr. Jokubaitis and her associates. Relapse activity was well-controlled in this patient group, and it was similar to relapse activity in patients switching to fingolimod from interferon-beta/glatiramer acetate or those starting fingolimod as their first disease-modifying therapy for MS.
The study was funded by a grant from the Australian National Health and Medical Research Council and by the MSBase Foundation. Dr. Jokubaitis and 13 of her associates reported having received honoraria or other support from Novartis, which makes fingolimod. Ten associates reported receiving honoraria or other support from Biogen Idec, which makes natalizumab.
FROM NEUROLOGY
Major Finding: Relapse rates after patients switched to fingolimod were low (quarterly relapse rate range, 0.079-0.13). Predictors of time to first relapse included number of relapses in the prior 6 months (hazard ratio, 1.59 per relapse; P = .002) and a 2- to 4-month treatment gap vs. no gap (HR, 2.10; P = .041).
Data Source: Cohort study of 536 patients with multiple sclerosis from the MSBase Registry.
Disclosures: Dr. Jokubaitis and 13 of her associates reported having received honoraria or other support from Novartis, which makes fingolimod. Ten associates reported receiving honoraria or other support from Biogen Idec, which makes natalizumab.
Maintenance percutaneous tibial nerve stimulation improved bladder symptoms in MS
Maintenance percutaneous tibial nerve stimulation in patients with multiple sclerosis led to sustained improvements in medically refractory lower urinary tract symptoms in a prospective, multicenter open-label trial.
All initial responders continued to respond after an average of 2 years of maintenance therapy, reported Dr. Claudio Gobbi of the Neurocentre of Southern Switzerland, Lugano, and his associates.
The prospective, multicenter open-label study recruited patients with multiple sclerosis who had lower urinary tract symptoms that had not responded to medical therapy. Symptom criteria included overactive bladder, incomplete voiding, hesitancy, poor or slow flow, prolonged and interrupted flow, and straining to void (J. Urol. 2014;191:697-702).
A total of 83 patients (mean age, 49 years; 62 women) completed 12 initial weekly sessions of percutaneous tibial nerve stimulation (PTNS). In all, 74 (89%) reported at least 50% improvement in lower urinary tract symptoms on the Patient Perception of Bladder Condition questionnaire.
These responders then underwent 30-minute PTNS maintenance sessions every 2, 3, or 4 weeks as needed. Most patients (60%) required treatment every 2 weeks, the researchers said.
After the 12 weekly PTNS treatments, most responders reported their bladder symptoms as moderately improved (based on a global response assessment), and mean treatment satisfaction was 70% (based on a visual analog scale). At 24 months’ follow-up, most patients described their symptoms as markedly improved, and mean treatment satisfaction was 82% (P less than .05).
The investigators also reported significant improvements over a 24-month period in measures of voiding and bladder diary parameters (including frequency of daytime urination, nocturia, and voided volume). Patients reported no relevant adverse effects of treatment.
Dr. Gobbi and his associates noted several caveats to their findings. The study probably lacked sufficient power to detect differences between subgroups, they said. The investigators also did not collect data on drinking habits or lifestyle modifications, each of which could have independently affected treatment outcomes. And the study included only patients who responded to and completed the initial treatment, which created a selection bias for patients willing to adhere to maintenance PTNS.
"Although the efficacy of PTNS was sustained for 2 years, the progressive neurologic course of MS with deteriorating bladder function may affect longer positive treatment outcomes," they added.
One author disclosed having financial interests or other relationships with Medtronic, Allergan, and Astellas. Dr. Gobbi and his other coauthors reported no conflicts of interest.
The most encouraging findings of this study were the clear improvements in voiding and voiding efficiency as well as overactive bladder symptoms, according to Dr. Gary E. Lemack.
He called the minimally invasive approach based on a nonimplantable technique "a welcome tool in our armamentarium" because it could benefit patients with multiple medical problems who need a repeat MRI.
Neuromodulation approaches have naturally evolved during the past 2 decades, said Dr. Lemack, citing recent reports that pudendal nerve stimulation, perianal stimulation, and sacral neuromodulation all directly affect overactive bladder contractions.
As new peripheral transcutaneous treatments for lower urinary tract symptoms emerge, we can expect an expansion of less-invasive neuromodulatory treatments that can be applied or maintained at home, he said.
These techniques will also be used in a wider range of patients, he added, including patients with neurogenic bladder symptoms who often suffer most from symptoms of overactive bladder.
Dr. Lemack is a urologist with the University of Texas Southwestern Medical Center at Dallas. He reported that he has financial interests or other relationships with Afferent Pharmaceuticals, Ferring, Merck, Pfizer, Astellas, and Allergan. These remarks were taken from his editorial accompanying Dr. Gobbi’s report (J. Urol. 2014;191:582-3).
The most encouraging findings of this study were the clear improvements in voiding and voiding efficiency as well as overactive bladder symptoms, according to Dr. Gary E. Lemack.
He called the minimally invasive approach based on a nonimplantable technique "a welcome tool in our armamentarium" because it could benefit patients with multiple medical problems who need a repeat MRI.
Neuromodulation approaches have naturally evolved during the past 2 decades, said Dr. Lemack, citing recent reports that pudendal nerve stimulation, perianal stimulation, and sacral neuromodulation all directly affect overactive bladder contractions.
As new peripheral transcutaneous treatments for lower urinary tract symptoms emerge, we can expect an expansion of less-invasive neuromodulatory treatments that can be applied or maintained at home, he said.
These techniques will also be used in a wider range of patients, he added, including patients with neurogenic bladder symptoms who often suffer most from symptoms of overactive bladder.
Dr. Lemack is a urologist with the University of Texas Southwestern Medical Center at Dallas. He reported that he has financial interests or other relationships with Afferent Pharmaceuticals, Ferring, Merck, Pfizer, Astellas, and Allergan. These remarks were taken from his editorial accompanying Dr. Gobbi’s report (J. Urol. 2014;191:582-3).
The most encouraging findings of this study were the clear improvements in voiding and voiding efficiency as well as overactive bladder symptoms, according to Dr. Gary E. Lemack.
He called the minimally invasive approach based on a nonimplantable technique "a welcome tool in our armamentarium" because it could benefit patients with multiple medical problems who need a repeat MRI.
Neuromodulation approaches have naturally evolved during the past 2 decades, said Dr. Lemack, citing recent reports that pudendal nerve stimulation, perianal stimulation, and sacral neuromodulation all directly affect overactive bladder contractions.
As new peripheral transcutaneous treatments for lower urinary tract symptoms emerge, we can expect an expansion of less-invasive neuromodulatory treatments that can be applied or maintained at home, he said.
These techniques will also be used in a wider range of patients, he added, including patients with neurogenic bladder symptoms who often suffer most from symptoms of overactive bladder.
Dr. Lemack is a urologist with the University of Texas Southwestern Medical Center at Dallas. He reported that he has financial interests or other relationships with Afferent Pharmaceuticals, Ferring, Merck, Pfizer, Astellas, and Allergan. These remarks were taken from his editorial accompanying Dr. Gobbi’s report (J. Urol. 2014;191:582-3).
Maintenance percutaneous tibial nerve stimulation in patients with multiple sclerosis led to sustained improvements in medically refractory lower urinary tract symptoms in a prospective, multicenter open-label trial.
All initial responders continued to respond after an average of 2 years of maintenance therapy, reported Dr. Claudio Gobbi of the Neurocentre of Southern Switzerland, Lugano, and his associates.
The prospective, multicenter open-label study recruited patients with multiple sclerosis who had lower urinary tract symptoms that had not responded to medical therapy. Symptom criteria included overactive bladder, incomplete voiding, hesitancy, poor or slow flow, prolonged and interrupted flow, and straining to void (J. Urol. 2014;191:697-702).
A total of 83 patients (mean age, 49 years; 62 women) completed 12 initial weekly sessions of percutaneous tibial nerve stimulation (PTNS). In all, 74 (89%) reported at least 50% improvement in lower urinary tract symptoms on the Patient Perception of Bladder Condition questionnaire.
These responders then underwent 30-minute PTNS maintenance sessions every 2, 3, or 4 weeks as needed. Most patients (60%) required treatment every 2 weeks, the researchers said.
After the 12 weekly PTNS treatments, most responders reported their bladder symptoms as moderately improved (based on a global response assessment), and mean treatment satisfaction was 70% (based on a visual analog scale). At 24 months’ follow-up, most patients described their symptoms as markedly improved, and mean treatment satisfaction was 82% (P less than .05).
The investigators also reported significant improvements over a 24-month period in measures of voiding and bladder diary parameters (including frequency of daytime urination, nocturia, and voided volume). Patients reported no relevant adverse effects of treatment.
Dr. Gobbi and his associates noted several caveats to their findings. The study probably lacked sufficient power to detect differences between subgroups, they said. The investigators also did not collect data on drinking habits or lifestyle modifications, each of which could have independently affected treatment outcomes. And the study included only patients who responded to and completed the initial treatment, which created a selection bias for patients willing to adhere to maintenance PTNS.
"Although the efficacy of PTNS was sustained for 2 years, the progressive neurologic course of MS with deteriorating bladder function may affect longer positive treatment outcomes," they added.
One author disclosed having financial interests or other relationships with Medtronic, Allergan, and Astellas. Dr. Gobbi and his other coauthors reported no conflicts of interest.
Maintenance percutaneous tibial nerve stimulation in patients with multiple sclerosis led to sustained improvements in medically refractory lower urinary tract symptoms in a prospective, multicenter open-label trial.
All initial responders continued to respond after an average of 2 years of maintenance therapy, reported Dr. Claudio Gobbi of the Neurocentre of Southern Switzerland, Lugano, and his associates.
The prospective, multicenter open-label study recruited patients with multiple sclerosis who had lower urinary tract symptoms that had not responded to medical therapy. Symptom criteria included overactive bladder, incomplete voiding, hesitancy, poor or slow flow, prolonged and interrupted flow, and straining to void (J. Urol. 2014;191:697-702).
A total of 83 patients (mean age, 49 years; 62 women) completed 12 initial weekly sessions of percutaneous tibial nerve stimulation (PTNS). In all, 74 (89%) reported at least 50% improvement in lower urinary tract symptoms on the Patient Perception of Bladder Condition questionnaire.
These responders then underwent 30-minute PTNS maintenance sessions every 2, 3, or 4 weeks as needed. Most patients (60%) required treatment every 2 weeks, the researchers said.
After the 12 weekly PTNS treatments, most responders reported their bladder symptoms as moderately improved (based on a global response assessment), and mean treatment satisfaction was 70% (based on a visual analog scale). At 24 months’ follow-up, most patients described their symptoms as markedly improved, and mean treatment satisfaction was 82% (P less than .05).
The investigators also reported significant improvements over a 24-month period in measures of voiding and bladder diary parameters (including frequency of daytime urination, nocturia, and voided volume). Patients reported no relevant adverse effects of treatment.
Dr. Gobbi and his associates noted several caveats to their findings. The study probably lacked sufficient power to detect differences between subgroups, they said. The investigators also did not collect data on drinking habits or lifestyle modifications, each of which could have independently affected treatment outcomes. And the study included only patients who responded to and completed the initial treatment, which created a selection bias for patients willing to adhere to maintenance PTNS.
"Although the efficacy of PTNS was sustained for 2 years, the progressive neurologic course of MS with deteriorating bladder function may affect longer positive treatment outcomes," they added.
One author disclosed having financial interests or other relationships with Medtronic, Allergan, and Astellas. Dr. Gobbi and his other coauthors reported no conflicts of interest.
FROM THE JOURNAL OF UROLOGY
Major finding: 74 of 83 patients (89%) reported at least 50% improvement after 12 weekly sessions of percutaneous tibial nerve stimulation. All initial responders had sustained benefits from maintenance therapy at 24 months’ follow-up.
Data source: A prospective, open-label multicenter trial of 83 patients with multiple sclerosis and medically refractory lower urinary tract symptoms.
Disclosures: One author disclosed having financial interests or other relationships with Medtronic, Allergan, and Astellas. Dr. Gobbi and his other coauthors reported no conflicts of interest.
Teriflunomide Reduces Annual MS Relapse Rate
Teriflunomide significantly reduced the annualized relapse rate in patients with relapsing-remitting multiple sclerosis (MS) by more than one-third, compared with placebo, in the second of two large, randomized, phase III trials that have been conducted with the drug.
The latest study—Teriflunomide Oral in People With Relapsing Multiple Sclerosis (TOWER)—demonstrated that the 14-mg dose of teriflunomide produced a 36% relative risk reduction in the annualized relapse rate, compared with placebo. A 7-mg dose of the oral MS agent yielded a 22% relative risk reduction. Teriflunomide 14 mg, but not 7 mg, was also associated with a significantly lower sustained disability accumulation than placebo.
“These data support the use of teriflunomide as an initial therapy for patients with relapsing-remitting MS and as an option for patients who are unable to tolerate other disease-modifying therapies,” Christian Confavreux, MD, of the Université Claude Bernard Lyon 1 in France, and associates wrote online ahead of print January 22 in Lancet Neurology.
The TOWER trial involved 1,169 patients with relapsing-remitting MS who were randomized to treatment—408 were assigned to teriflunomide 14 mg, 372 to teriflunomide 7 mg, and 389 to placebo. The mean age of patients at study entry was approximately 38—slightly more than 80% were white, 15% were Asian, and 2% were black.
The annualized relapse rates were 0.50 for patients treated with placebo, 0.32 for patients treated with teriflunomide 14 mg, and 0.39 for those treated with teriflunomide 7 mg. Absolute risk reductions for teriflunomide 14 mg and 7 mg versus placebo were –0.11 and –0.18, respectively.
A key secondary end point was sustained accumulation of disability. This outcome was defined as an increase of at least 1 point on the Expanded Disability Status Scale from baseline that persisted for at least 12 weeks. The hazard ratios for the time to sustained accumulation of disability were 0.68 for the 14-mg dose of teriflunomide and 0.95 for the 7-mg dose versus placebo.
“The safety and tolerability profile of teriflunomide ... was similar for both the 7-mg and 14-mg doses, including long-term treatment observations,” the investigators wrote. Common side effects in patients treated with teriflunomide were hair thinning and headache.
—Sara Freeman
Suggested Reading
Confavreux C, O’Connor P, Comi G, et al. Oral teriflunomide for patients with relapsing multiple sclerosis (TOWER): a randomized, double-blind, placebo-controlled, phase 3 trial. Lancet Neurol. 2014 Jan 22 [Epub ahead of print].
Teriflunomide significantly reduced the annualized relapse rate in patients with relapsing-remitting multiple sclerosis (MS) by more than one-third, compared with placebo, in the second of two large, randomized, phase III trials that have been conducted with the drug.
The latest study—Teriflunomide Oral in People With Relapsing Multiple Sclerosis (TOWER)—demonstrated that the 14-mg dose of teriflunomide produced a 36% relative risk reduction in the annualized relapse rate, compared with placebo. A 7-mg dose of the oral MS agent yielded a 22% relative risk reduction. Teriflunomide 14 mg, but not 7 mg, was also associated with a significantly lower sustained disability accumulation than placebo.
“These data support the use of teriflunomide as an initial therapy for patients with relapsing-remitting MS and as an option for patients who are unable to tolerate other disease-modifying therapies,” Christian Confavreux, MD, of the Université Claude Bernard Lyon 1 in France, and associates wrote online ahead of print January 22 in Lancet Neurology.
The TOWER trial involved 1,169 patients with relapsing-remitting MS who were randomized to treatment—408 were assigned to teriflunomide 14 mg, 372 to teriflunomide 7 mg, and 389 to placebo. The mean age of patients at study entry was approximately 38—slightly more than 80% were white, 15% were Asian, and 2% were black.
The annualized relapse rates were 0.50 for patients treated with placebo, 0.32 for patients treated with teriflunomide 14 mg, and 0.39 for those treated with teriflunomide 7 mg. Absolute risk reductions for teriflunomide 14 mg and 7 mg versus placebo were –0.11 and –0.18, respectively.
A key secondary end point was sustained accumulation of disability. This outcome was defined as an increase of at least 1 point on the Expanded Disability Status Scale from baseline that persisted for at least 12 weeks. The hazard ratios for the time to sustained accumulation of disability were 0.68 for the 14-mg dose of teriflunomide and 0.95 for the 7-mg dose versus placebo.
“The safety and tolerability profile of teriflunomide ... was similar for both the 7-mg and 14-mg doses, including long-term treatment observations,” the investigators wrote. Common side effects in patients treated with teriflunomide were hair thinning and headache.
—Sara Freeman
Teriflunomide significantly reduced the annualized relapse rate in patients with relapsing-remitting multiple sclerosis (MS) by more than one-third, compared with placebo, in the second of two large, randomized, phase III trials that have been conducted with the drug.
The latest study—Teriflunomide Oral in People With Relapsing Multiple Sclerosis (TOWER)—demonstrated that the 14-mg dose of teriflunomide produced a 36% relative risk reduction in the annualized relapse rate, compared with placebo. A 7-mg dose of the oral MS agent yielded a 22% relative risk reduction. Teriflunomide 14 mg, but not 7 mg, was also associated with a significantly lower sustained disability accumulation than placebo.
“These data support the use of teriflunomide as an initial therapy for patients with relapsing-remitting MS and as an option for patients who are unable to tolerate other disease-modifying therapies,” Christian Confavreux, MD, of the Université Claude Bernard Lyon 1 in France, and associates wrote online ahead of print January 22 in Lancet Neurology.
The TOWER trial involved 1,169 patients with relapsing-remitting MS who were randomized to treatment—408 were assigned to teriflunomide 14 mg, 372 to teriflunomide 7 mg, and 389 to placebo. The mean age of patients at study entry was approximately 38—slightly more than 80% were white, 15% were Asian, and 2% were black.
The annualized relapse rates were 0.50 for patients treated with placebo, 0.32 for patients treated with teriflunomide 14 mg, and 0.39 for those treated with teriflunomide 7 mg. Absolute risk reductions for teriflunomide 14 mg and 7 mg versus placebo were –0.11 and –0.18, respectively.
A key secondary end point was sustained accumulation of disability. This outcome was defined as an increase of at least 1 point on the Expanded Disability Status Scale from baseline that persisted for at least 12 weeks. The hazard ratios for the time to sustained accumulation of disability were 0.68 for the 14-mg dose of teriflunomide and 0.95 for the 7-mg dose versus placebo.
“The safety and tolerability profile of teriflunomide ... was similar for both the 7-mg and 14-mg doses, including long-term treatment observations,” the investigators wrote. Common side effects in patients treated with teriflunomide were hair thinning and headache.
—Sara Freeman
Suggested Reading
Confavreux C, O’Connor P, Comi G, et al. Oral teriflunomide for patients with relapsing multiple sclerosis (TOWER): a randomized, double-blind, placebo-controlled, phase 3 trial. Lancet Neurol. 2014 Jan 22 [Epub ahead of print].
Suggested Reading
Confavreux C, O’Connor P, Comi G, et al. Oral teriflunomide for patients with relapsing multiple sclerosis (TOWER): a randomized, double-blind, placebo-controlled, phase 3 trial. Lancet Neurol. 2014 Jan 22 [Epub ahead of print].
Obesity at Age 20 Is Associated With an Increased Risk for MS
Multiple sclerosis (MS) has a “striking” association with obesity at age 20 that strongly interacts with genetic susceptibility, according to an analysis of data from two case–control studies that examined environmental and genetic risk factors for MS and was published online ahead of print February 5 in Neurology.
This relationship between adolescent obesity and MS is of the same magnitude as the separate associations between MS and carriage of the high-risk HLA-DRB1*15 allele, absence of the protective HLA-A*02 allele, and smoking, according to Anna Karin Hedström, MD, of the Institute of Environmental Medicine, Karolinska Institutet, Stockholm, and colleagues.
“The biologic explanations for these interactions are far from clear, but the data open [the way] for mechanistically oriented studies,” the study authors stated.
Three previous studies have suggested that obesity in early life may be linked to an increased risk of developing MS later. Dr. Hedström and her colleagues examined this association using data from a Swedish population-based, case–control study and from a separate American case–control study.
In the Swedish study, 1,510 adults with incident MS who were treated at 40 clinics across the country during a seven-year period and 2,017 control subjects completed detailed questionnaires concerning environmental exposures and other factors. The controls were matched for age, sex, and area of residence, and all the participants gave blood samples for HLA typing.
The American study involved 937 white adults with prevalent MS who were members of a single large health maintenance organization covering northern California and 609 white control subjects matched for age, sex, and area of residence. All the participants completed computer-assisted telephone interviews regarding environmental exposures and lifestyle factors.
All the subjects in both studies reported what their height and weight had been at age 20, from which the investigators calculated BMI.
In both studies, participants whose BMI at age 20 was 27 kg/m2 or greater showed an increased risk of developing MS later in life, compared with those whose BMI was 18.5 to 21 kg/m2. The odds ratios (ORs) were 2.2 for subjects in the Swedish study and 1.8 for those in the American study, Dr. Hedström and her associates said.
Similarly, participants with a slightly lower but still above-normal BMI of 25 to 27 kg/m2 had a modestly increased risk of developing MS later in life. The ORs were 1.4 in the Swedish study and 1.3 in the American study.
These ORs were unchanged when a sensitivity analysis was performed, including only the study subjects who had been genotyped.
Participants who carried the high-risk HLA-DRB1*15 gene, did not carry the protective HLA-A*02 gene, and had a BMI of 27 kg/m2 or greater at age 20 had an OR of 16.2 for developing later MS, compared with those who had none of those risk factors. In contrast, subjects who had the same HLA profile but had not been obese at age 20 had an OR of 5.1.
The investigators proposed that the low-grade chronic inflammation associated with obesity, together with obesity’s adverse effects on autoimmunity, may increase the risk of HLA-related activation of T cells that attack the CNS.
Both studies were limited in that they were retrospective and relied on participants’ self-reports. In addition, Dr. Hedström and her associates modified the usual definition of obesity for the purposes of their study. The typical standard for obesity is a BMI of greater than 30 kg/m2, not greater than 27 kg/m2. However, the number of subjects at this level of BMI was too small in the Swedish cohort to allow accurate analysis, so the researchers combined the top two categories of BMI into one designation of obese.
—Mary Ann Moon
Suggested Reading
Hedström AK, Lima Bomfim I, Barcellos L, et al. Interaction between adolescent obesity and HLA risk genes in the etiology of multiple sclerosis. Neurology. 2014 Feb 5 [Epub ahead of print].
Multiple sclerosis (MS) has a “striking” association with obesity at age 20 that strongly interacts with genetic susceptibility, according to an analysis of data from two case–control studies that examined environmental and genetic risk factors for MS and was published online ahead of print February 5 in Neurology.
This relationship between adolescent obesity and MS is of the same magnitude as the separate associations between MS and carriage of the high-risk HLA-DRB1*15 allele, absence of the protective HLA-A*02 allele, and smoking, according to Anna Karin Hedström, MD, of the Institute of Environmental Medicine, Karolinska Institutet, Stockholm, and colleagues.
“The biologic explanations for these interactions are far from clear, but the data open [the way] for mechanistically oriented studies,” the study authors stated.
Three previous studies have suggested that obesity in early life may be linked to an increased risk of developing MS later. Dr. Hedström and her colleagues examined this association using data from a Swedish population-based, case–control study and from a separate American case–control study.
In the Swedish study, 1,510 adults with incident MS who were treated at 40 clinics across the country during a seven-year period and 2,017 control subjects completed detailed questionnaires concerning environmental exposures and other factors. The controls were matched for age, sex, and area of residence, and all the participants gave blood samples for HLA typing.
The American study involved 937 white adults with prevalent MS who were members of a single large health maintenance organization covering northern California and 609 white control subjects matched for age, sex, and area of residence. All the participants completed computer-assisted telephone interviews regarding environmental exposures and lifestyle factors.
All the subjects in both studies reported what their height and weight had been at age 20, from which the investigators calculated BMI.
In both studies, participants whose BMI at age 20 was 27 kg/m2 or greater showed an increased risk of developing MS later in life, compared with those whose BMI was 18.5 to 21 kg/m2. The odds ratios (ORs) were 2.2 for subjects in the Swedish study and 1.8 for those in the American study, Dr. Hedström and her associates said.
Similarly, participants with a slightly lower but still above-normal BMI of 25 to 27 kg/m2 had a modestly increased risk of developing MS later in life. The ORs were 1.4 in the Swedish study and 1.3 in the American study.
These ORs were unchanged when a sensitivity analysis was performed, including only the study subjects who had been genotyped.
Participants who carried the high-risk HLA-DRB1*15 gene, did not carry the protective HLA-A*02 gene, and had a BMI of 27 kg/m2 or greater at age 20 had an OR of 16.2 for developing later MS, compared with those who had none of those risk factors. In contrast, subjects who had the same HLA profile but had not been obese at age 20 had an OR of 5.1.
The investigators proposed that the low-grade chronic inflammation associated with obesity, together with obesity’s adverse effects on autoimmunity, may increase the risk of HLA-related activation of T cells that attack the CNS.
Both studies were limited in that they were retrospective and relied on participants’ self-reports. In addition, Dr. Hedström and her associates modified the usual definition of obesity for the purposes of their study. The typical standard for obesity is a BMI of greater than 30 kg/m2, not greater than 27 kg/m2. However, the number of subjects at this level of BMI was too small in the Swedish cohort to allow accurate analysis, so the researchers combined the top two categories of BMI into one designation of obese.
—Mary Ann Moon
Multiple sclerosis (MS) has a “striking” association with obesity at age 20 that strongly interacts with genetic susceptibility, according to an analysis of data from two case–control studies that examined environmental and genetic risk factors for MS and was published online ahead of print February 5 in Neurology.
This relationship between adolescent obesity and MS is of the same magnitude as the separate associations between MS and carriage of the high-risk HLA-DRB1*15 allele, absence of the protective HLA-A*02 allele, and smoking, according to Anna Karin Hedström, MD, of the Institute of Environmental Medicine, Karolinska Institutet, Stockholm, and colleagues.
“The biologic explanations for these interactions are far from clear, but the data open [the way] for mechanistically oriented studies,” the study authors stated.
Three previous studies have suggested that obesity in early life may be linked to an increased risk of developing MS later. Dr. Hedström and her colleagues examined this association using data from a Swedish population-based, case–control study and from a separate American case–control study.
In the Swedish study, 1,510 adults with incident MS who were treated at 40 clinics across the country during a seven-year period and 2,017 control subjects completed detailed questionnaires concerning environmental exposures and other factors. The controls were matched for age, sex, and area of residence, and all the participants gave blood samples for HLA typing.
The American study involved 937 white adults with prevalent MS who were members of a single large health maintenance organization covering northern California and 609 white control subjects matched for age, sex, and area of residence. All the participants completed computer-assisted telephone interviews regarding environmental exposures and lifestyle factors.
All the subjects in both studies reported what their height and weight had been at age 20, from which the investigators calculated BMI.
In both studies, participants whose BMI at age 20 was 27 kg/m2 or greater showed an increased risk of developing MS later in life, compared with those whose BMI was 18.5 to 21 kg/m2. The odds ratios (ORs) were 2.2 for subjects in the Swedish study and 1.8 for those in the American study, Dr. Hedström and her associates said.
Similarly, participants with a slightly lower but still above-normal BMI of 25 to 27 kg/m2 had a modestly increased risk of developing MS later in life. The ORs were 1.4 in the Swedish study and 1.3 in the American study.
These ORs were unchanged when a sensitivity analysis was performed, including only the study subjects who had been genotyped.
Participants who carried the high-risk HLA-DRB1*15 gene, did not carry the protective HLA-A*02 gene, and had a BMI of 27 kg/m2 or greater at age 20 had an OR of 16.2 for developing later MS, compared with those who had none of those risk factors. In contrast, subjects who had the same HLA profile but had not been obese at age 20 had an OR of 5.1.
The investigators proposed that the low-grade chronic inflammation associated with obesity, together with obesity’s adverse effects on autoimmunity, may increase the risk of HLA-related activation of T cells that attack the CNS.
Both studies were limited in that they were retrospective and relied on participants’ self-reports. In addition, Dr. Hedström and her associates modified the usual definition of obesity for the purposes of their study. The typical standard for obesity is a BMI of greater than 30 kg/m2, not greater than 27 kg/m2. However, the number of subjects at this level of BMI was too small in the Swedish cohort to allow accurate analysis, so the researchers combined the top two categories of BMI into one designation of obese.
—Mary Ann Moon
Suggested Reading
Hedström AK, Lima Bomfim I, Barcellos L, et al. Interaction between adolescent obesity and HLA risk genes in the etiology of multiple sclerosis. Neurology. 2014 Feb 5 [Epub ahead of print].
Suggested Reading
Hedström AK, Lima Bomfim I, Barcellos L, et al. Interaction between adolescent obesity and HLA risk genes in the etiology of multiple sclerosis. Neurology. 2014 Feb 5 [Epub ahead of print].