User login
High dose of novel compound for relapsing-remitting MS shows promise
SAN DIEGO – Early results of the novel human endogenous retrovirus-W antagonist GNbAC1 in a phase 2 trial of patients with relapsing-remitting multiple sclerosis demonstrated evidence of remyelination at week 24 among high-dose users, but it did not meet its primary endpoint of active lesions seen on MRI.
In an interview at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis, study author Robert Glanzman, MD, said that GNbAC1 is a monoclonal antibody that targets and blocks the envelope protein pHER-W ENV, a potent agonist of Toll-like receptor 4. It thereby inhibits TLR4-mediated pathogenicity, which includes activation of macrophages and microglia into proinflammatory phenotypes and direct inhibition of remyelination via TLR4.
In a study known as CHANGE-MS, 270 patients with relapsing-remitting MS were randomized to one of three doses of the GNbAC1 (6, 12, or 18 mg/kg), or placebo via monthly IV infusion over 6 months. The study was conducted at 70 centers in 13 European countries over the past 3 years. It had a 24-week, double-blind, placebo-controlled period, followed by a 24-week, dose-blind, active-only treatment period, with placebo patients randomized to the three different doses of GNbA1C. Brain MRI scans were performed at weeks 12, 16, 20, 24, and 48, to look for evidence of remyelination.
The mean age of patients was 38 years and 65% were female. The researchers observed no safety concerns and no significant effect on inflammatory measures over weeks 12-24, even though the absolute number of lesions was reduced by about 50%. Although the primary endpoint of the cumulative number of gadolinium-enhancing lesions seen on brain MRI scans every 4 weeks during weeks 12-24 was not met, post hoc analyses suggest a decrease in neuroinflammation in the 18 mg/kg GNbA1C group at week 24, compared with placebo (P = .008). “A consistent increase in MT [magnetization transfer] ratio signal was observed in normal-appearing white matter and cerebral cortex at the highest dose, suggesting remyelination,” Dr. Glanzman added. “We gained about a quarter or half of percent in normal-appearing white matter at the cerebral cortex at the high dose. Normally, MS patients lose white matter over time, both in the cortex and in gray matter. We’re actually showing evidence of remyelination, which is really exciting. If these data are replicated and confirmed at week 48, we think we really have an exciting compound.”
GeNeuro sponsored the study.
Full 48-week analyses from CHANGE-MS are expected to be unveiled at the 2018 annual meeting American Academy of Neurology.
SOURCE: Glanzman R et al. Abstract P034.
SAN DIEGO – Early results of the novel human endogenous retrovirus-W antagonist GNbAC1 in a phase 2 trial of patients with relapsing-remitting multiple sclerosis demonstrated evidence of remyelination at week 24 among high-dose users, but it did not meet its primary endpoint of active lesions seen on MRI.
In an interview at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis, study author Robert Glanzman, MD, said that GNbAC1 is a monoclonal antibody that targets and blocks the envelope protein pHER-W ENV, a potent agonist of Toll-like receptor 4. It thereby inhibits TLR4-mediated pathogenicity, which includes activation of macrophages and microglia into proinflammatory phenotypes and direct inhibition of remyelination via TLR4.
In a study known as CHANGE-MS, 270 patients with relapsing-remitting MS were randomized to one of three doses of the GNbAC1 (6, 12, or 18 mg/kg), or placebo via monthly IV infusion over 6 months. The study was conducted at 70 centers in 13 European countries over the past 3 years. It had a 24-week, double-blind, placebo-controlled period, followed by a 24-week, dose-blind, active-only treatment period, with placebo patients randomized to the three different doses of GNbA1C. Brain MRI scans were performed at weeks 12, 16, 20, 24, and 48, to look for evidence of remyelination.
The mean age of patients was 38 years and 65% were female. The researchers observed no safety concerns and no significant effect on inflammatory measures over weeks 12-24, even though the absolute number of lesions was reduced by about 50%. Although the primary endpoint of the cumulative number of gadolinium-enhancing lesions seen on brain MRI scans every 4 weeks during weeks 12-24 was not met, post hoc analyses suggest a decrease in neuroinflammation in the 18 mg/kg GNbA1C group at week 24, compared with placebo (P = .008). “A consistent increase in MT [magnetization transfer] ratio signal was observed in normal-appearing white matter and cerebral cortex at the highest dose, suggesting remyelination,” Dr. Glanzman added. “We gained about a quarter or half of percent in normal-appearing white matter at the cerebral cortex at the high dose. Normally, MS patients lose white matter over time, both in the cortex and in gray matter. We’re actually showing evidence of remyelination, which is really exciting. If these data are replicated and confirmed at week 48, we think we really have an exciting compound.”
GeNeuro sponsored the study.
Full 48-week analyses from CHANGE-MS are expected to be unveiled at the 2018 annual meeting American Academy of Neurology.
SOURCE: Glanzman R et al. Abstract P034.
SAN DIEGO – Early results of the novel human endogenous retrovirus-W antagonist GNbAC1 in a phase 2 trial of patients with relapsing-remitting multiple sclerosis demonstrated evidence of remyelination at week 24 among high-dose users, but it did not meet its primary endpoint of active lesions seen on MRI.
In an interview at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis, study author Robert Glanzman, MD, said that GNbAC1 is a monoclonal antibody that targets and blocks the envelope protein pHER-W ENV, a potent agonist of Toll-like receptor 4. It thereby inhibits TLR4-mediated pathogenicity, which includes activation of macrophages and microglia into proinflammatory phenotypes and direct inhibition of remyelination via TLR4.
In a study known as CHANGE-MS, 270 patients with relapsing-remitting MS were randomized to one of three doses of the GNbAC1 (6, 12, or 18 mg/kg), or placebo via monthly IV infusion over 6 months. The study was conducted at 70 centers in 13 European countries over the past 3 years. It had a 24-week, double-blind, placebo-controlled period, followed by a 24-week, dose-blind, active-only treatment period, with placebo patients randomized to the three different doses of GNbA1C. Brain MRI scans were performed at weeks 12, 16, 20, 24, and 48, to look for evidence of remyelination.
The mean age of patients was 38 years and 65% were female. The researchers observed no safety concerns and no significant effect on inflammatory measures over weeks 12-24, even though the absolute number of lesions was reduced by about 50%. Although the primary endpoint of the cumulative number of gadolinium-enhancing lesions seen on brain MRI scans every 4 weeks during weeks 12-24 was not met, post hoc analyses suggest a decrease in neuroinflammation in the 18 mg/kg GNbA1C group at week 24, compared with placebo (P = .008). “A consistent increase in MT [magnetization transfer] ratio signal was observed in normal-appearing white matter and cerebral cortex at the highest dose, suggesting remyelination,” Dr. Glanzman added. “We gained about a quarter or half of percent in normal-appearing white matter at the cerebral cortex at the high dose. Normally, MS patients lose white matter over time, both in the cortex and in gray matter. We’re actually showing evidence of remyelination, which is really exciting. If these data are replicated and confirmed at week 48, we think we really have an exciting compound.”
GeNeuro sponsored the study.
Full 48-week analyses from CHANGE-MS are expected to be unveiled at the 2018 annual meeting American Academy of Neurology.
SOURCE: Glanzman R et al. Abstract P034.
REPORTING FROM ACTRIMS FORUM 2018
Key clinical point:
Major finding: Although the primary endpoint was not met, post hoc analyses suggest a decrease in neuroinflammation in the 18 mg/kg GNbA1C group at week 24, compared with placebo (P = .008).
Study details: A phase 2 study of 270 patients with relapsing-remitting MS who were randomized to one of three doses of GNbAC1.
Disclosures: Dr. Glanzman is chief medical officer for GeNeuro, which sponsored the study.
Source: Glanzman R et al. Abstract P034.
How Are Newer DMTs Used in Pediatric MS?
SAN DIEGO—Newer oral and IV disease-modifying therapies (DMTs) for multiple sclerosis (MS) increasingly are used in children, according to data presented at the ACTRIMS 2018 Forum. These DMTs often are second-line therapies, but some patients receive them as first-line treatment.
“Newer DMTs … are being started more often over time at clinics participating in the US Network of Pediatric MS Centers,” said Kristen M. Krysko, MD, Clinical Fellow at the University of California, San Francisco School of Medicine, and colleagues.
The short-term side effect profile of newer DMTs in children is similar to that in adults. The study may have underestimated side effects, however, and long-term safety data are lacking, the researchers noted.
Treatment Challenges
About 5% of patients with MS develop symptoms before age 18. “Treatment of pediatric MS is challenging, given high relapse rates and the lack of safety and efficacy data for DMTs in children,” Dr. Krysko and colleagues said. “Conventionally, first-line treatments for pediatric MS include interferon beta and glatiramer acetate, but these agents may be poorly tolerated, since they are given by injection, and may not adequately control the disease. Those with breakthrough events require escalation to more potent agents, which are increasingly used off-label in pediatric MS despite limited data about safety or efficacy in children.”
To characterize the use and safety of newer DMTs in children with MS and clinically isolated syndrome (CIS), Dr. Krysko and colleagues conducted a retrospective cohort study of prospectively collected data at 12 clinics participating in the US Network of Pediatric MS Centers as of August 2017.
The researchers examined patterns of newer DMT use and newer DMT side effects. They classified DMTs as injectable (ie, glatiramer acetate and beta-interferons), oral (ie, dimethyl fumarate, fingolimod, and teriflunomide), or IV (ie, natalizumab, rituximab, alemtuzumab, and ocrelizumab). The researchers considered dimethyl fumarate, fingolimod, teriflunomide, natalizumab, rituximab, ocrelizumab, alemtuzumab, and daclizumab to be newer therapies.
The cohort included more than 1,000 patients with MS or CIS. In all, 618 patients had received a DMT before age 18 (587 patients with a diagnosis of MS at last follow-up and 31 patients with a diagnosis of CIS at last follow-up). About 66% were female, and patients’ mean age at MS onset was approximately 13.
The Use of Oral and IV DMTs
From 2008 to 2017, use of newer oral and IV DMTs increased overall and as first-line therapy in patients younger than 12 and in patients 12 or older. Of the 618 patients who received a DMT before age 18, 259 (42%) received a newer DMT, and 104 (17%) received a newer DMT as a first-line therapy.
Newer DMTs used in patients before age 18 included natalizumab in 101 patients, dimethyl fumarate in 100 patients, rituximab in 57 patients, fingolimod in 37 patients, daclizumab in five patients, and teriflunomide in three patients. Newer DMTs used as first-line therapy in patients before age 18 included dimethyl fumarate in 36 patients, natalizumab in 30 patients, rituximab in 22 patients, fingolimod in 14 patients, and teriflunomide in two patients.
Patients were exposed to dimethyl fumarate for 149 person-years, natalizumab for 140 person-years, rituximab for 75 person-years, fingolimod for 55 person-years, daclizumab for 10 person-years, and teriflunomide for one person-year.
Randomized controlled trials of the efficacy of DMTs in children with MS are ongoing. Future studies may explore predictors of newer DMT use in children with MS, the researchers said.
—Jake Remaly
SAN DIEGO—Newer oral and IV disease-modifying therapies (DMTs) for multiple sclerosis (MS) increasingly are used in children, according to data presented at the ACTRIMS 2018 Forum. These DMTs often are second-line therapies, but some patients receive them as first-line treatment.
“Newer DMTs … are being started more often over time at clinics participating in the US Network of Pediatric MS Centers,” said Kristen M. Krysko, MD, Clinical Fellow at the University of California, San Francisco School of Medicine, and colleagues.
The short-term side effect profile of newer DMTs in children is similar to that in adults. The study may have underestimated side effects, however, and long-term safety data are lacking, the researchers noted.
Treatment Challenges
About 5% of patients with MS develop symptoms before age 18. “Treatment of pediatric MS is challenging, given high relapse rates and the lack of safety and efficacy data for DMTs in children,” Dr. Krysko and colleagues said. “Conventionally, first-line treatments for pediatric MS include interferon beta and glatiramer acetate, but these agents may be poorly tolerated, since they are given by injection, and may not adequately control the disease. Those with breakthrough events require escalation to more potent agents, which are increasingly used off-label in pediatric MS despite limited data about safety or efficacy in children.”
To characterize the use and safety of newer DMTs in children with MS and clinically isolated syndrome (CIS), Dr. Krysko and colleagues conducted a retrospective cohort study of prospectively collected data at 12 clinics participating in the US Network of Pediatric MS Centers as of August 2017.
The researchers examined patterns of newer DMT use and newer DMT side effects. They classified DMTs as injectable (ie, glatiramer acetate and beta-interferons), oral (ie, dimethyl fumarate, fingolimod, and teriflunomide), or IV (ie, natalizumab, rituximab, alemtuzumab, and ocrelizumab). The researchers considered dimethyl fumarate, fingolimod, teriflunomide, natalizumab, rituximab, ocrelizumab, alemtuzumab, and daclizumab to be newer therapies.
The cohort included more than 1,000 patients with MS or CIS. In all, 618 patients had received a DMT before age 18 (587 patients with a diagnosis of MS at last follow-up and 31 patients with a diagnosis of CIS at last follow-up). About 66% were female, and patients’ mean age at MS onset was approximately 13.
The Use of Oral and IV DMTs
From 2008 to 2017, use of newer oral and IV DMTs increased overall and as first-line therapy in patients younger than 12 and in patients 12 or older. Of the 618 patients who received a DMT before age 18, 259 (42%) received a newer DMT, and 104 (17%) received a newer DMT as a first-line therapy.
Newer DMTs used in patients before age 18 included natalizumab in 101 patients, dimethyl fumarate in 100 patients, rituximab in 57 patients, fingolimod in 37 patients, daclizumab in five patients, and teriflunomide in three patients. Newer DMTs used as first-line therapy in patients before age 18 included dimethyl fumarate in 36 patients, natalizumab in 30 patients, rituximab in 22 patients, fingolimod in 14 patients, and teriflunomide in two patients.
Patients were exposed to dimethyl fumarate for 149 person-years, natalizumab for 140 person-years, rituximab for 75 person-years, fingolimod for 55 person-years, daclizumab for 10 person-years, and teriflunomide for one person-year.
Randomized controlled trials of the efficacy of DMTs in children with MS are ongoing. Future studies may explore predictors of newer DMT use in children with MS, the researchers said.
—Jake Remaly
SAN DIEGO—Newer oral and IV disease-modifying therapies (DMTs) for multiple sclerosis (MS) increasingly are used in children, according to data presented at the ACTRIMS 2018 Forum. These DMTs often are second-line therapies, but some patients receive them as first-line treatment.
“Newer DMTs … are being started more often over time at clinics participating in the US Network of Pediatric MS Centers,” said Kristen M. Krysko, MD, Clinical Fellow at the University of California, San Francisco School of Medicine, and colleagues.
The short-term side effect profile of newer DMTs in children is similar to that in adults. The study may have underestimated side effects, however, and long-term safety data are lacking, the researchers noted.
Treatment Challenges
About 5% of patients with MS develop symptoms before age 18. “Treatment of pediatric MS is challenging, given high relapse rates and the lack of safety and efficacy data for DMTs in children,” Dr. Krysko and colleagues said. “Conventionally, first-line treatments for pediatric MS include interferon beta and glatiramer acetate, but these agents may be poorly tolerated, since they are given by injection, and may not adequately control the disease. Those with breakthrough events require escalation to more potent agents, which are increasingly used off-label in pediatric MS despite limited data about safety or efficacy in children.”
To characterize the use and safety of newer DMTs in children with MS and clinically isolated syndrome (CIS), Dr. Krysko and colleagues conducted a retrospective cohort study of prospectively collected data at 12 clinics participating in the US Network of Pediatric MS Centers as of August 2017.
The researchers examined patterns of newer DMT use and newer DMT side effects. They classified DMTs as injectable (ie, glatiramer acetate and beta-interferons), oral (ie, dimethyl fumarate, fingolimod, and teriflunomide), or IV (ie, natalizumab, rituximab, alemtuzumab, and ocrelizumab). The researchers considered dimethyl fumarate, fingolimod, teriflunomide, natalizumab, rituximab, ocrelizumab, alemtuzumab, and daclizumab to be newer therapies.
The cohort included more than 1,000 patients with MS or CIS. In all, 618 patients had received a DMT before age 18 (587 patients with a diagnosis of MS at last follow-up and 31 patients with a diagnosis of CIS at last follow-up). About 66% were female, and patients’ mean age at MS onset was approximately 13.
The Use of Oral and IV DMTs
From 2008 to 2017, use of newer oral and IV DMTs increased overall and as first-line therapy in patients younger than 12 and in patients 12 or older. Of the 618 patients who received a DMT before age 18, 259 (42%) received a newer DMT, and 104 (17%) received a newer DMT as a first-line therapy.
Newer DMTs used in patients before age 18 included natalizumab in 101 patients, dimethyl fumarate in 100 patients, rituximab in 57 patients, fingolimod in 37 patients, daclizumab in five patients, and teriflunomide in three patients. Newer DMTs used as first-line therapy in patients before age 18 included dimethyl fumarate in 36 patients, natalizumab in 30 patients, rituximab in 22 patients, fingolimod in 14 patients, and teriflunomide in two patients.
Patients were exposed to dimethyl fumarate for 149 person-years, natalizumab for 140 person-years, rituximab for 75 person-years, fingolimod for 55 person-years, daclizumab for 10 person-years, and teriflunomide for one person-year.
Randomized controlled trials of the efficacy of DMTs in children with MS are ongoing. Future studies may explore predictors of newer DMT use in children with MS, the researchers said.
—Jake Remaly
Can Online Patient Journals Improve MS Care?
SAN DIEGO—Continuous collection of patient-reported outcomes via a web-based platform may help neurologists identify patients with multiple sclerosis (MS) with active disease who would benefit from additional care coordination, according to a study described at the ACTRIMS 2018 Forum.
MS has a “variable and unpredictable disease course,” said Alexis Ahmad, Director of Clinical Trials for the Georgetown University Department of Neurology in Washington, DC, and colleagues. “Given the chronic and complex nature of MS, patients living with this disease require continuous and specialized care.”
The MS and Neuroimmunology Center at Georgetown University aims to become the first MS center recognized as a Patient-Centered Specialty Practice by the National Committee for Quality Assurance.
To assess whether a web-based platform to capture patient-reported outcomes can help MS practices meet Patient-Centered Specialty Practice standards and achieve MS care goals, the researchers analyzed patient responses in their center’s MS-Advance Study. The MS-Advance Study includes patients from the practice who use a web-based platform to relay patient-reported outcomes in between and at clinic visits.
Patients completed scales and questionnaires at baseline (eg, Modified Fatigue Impact Scale, Patient Health Questionnaire-9, Work Productivity and Activity Improvement Questionnaire, and Treatment Satisfaction Questionnaire for Medication) and journal entries about relapses, mood, energy, nutrition, sleep, and health service or resource utilization. The investigators analyzed responses from patients who completed at least one patient-reported outcome or journal entry between January and August 2017.
Five hundred twenty patients agreed to report outcomes using the online platform; 310 patients completed one or more patient-reported outcome or journal entry. In all, 259 patients reported daily MS sympto
“It is challenging for providers and clinicians to incorporate routine review of patient-reported outcomes into their clinic schedules,” the researchers noted. Strategies for reviewing patient-reported outcomes in clinic and eliciting more complete and timely patient reports are warranted, they said.
“Use of an online platform provides an opportunity to learn about patients’ daily experiences. Understanding these experiences can inform care strategies to address total functionality and other clinical outcomes.”
—Jake Remaly
SAN DIEGO—Continuous collection of patient-reported outcomes via a web-based platform may help neurologists identify patients with multiple sclerosis (MS) with active disease who would benefit from additional care coordination, according to a study described at the ACTRIMS 2018 Forum.
MS has a “variable and unpredictable disease course,” said Alexis Ahmad, Director of Clinical Trials for the Georgetown University Department of Neurology in Washington, DC, and colleagues. “Given the chronic and complex nature of MS, patients living with this disease require continuous and specialized care.”
The MS and Neuroimmunology Center at Georgetown University aims to become the first MS center recognized as a Patient-Centered Specialty Practice by the National Committee for Quality Assurance.
To assess whether a web-based platform to capture patient-reported outcomes can help MS practices meet Patient-Centered Specialty Practice standards and achieve MS care goals, the researchers analyzed patient responses in their center’s MS-Advance Study. The MS-Advance Study includes patients from the practice who use a web-based platform to relay patient-reported outcomes in between and at clinic visits.
Patients completed scales and questionnaires at baseline (eg, Modified Fatigue Impact Scale, Patient Health Questionnaire-9, Work Productivity and Activity Improvement Questionnaire, and Treatment Satisfaction Questionnaire for Medication) and journal entries about relapses, mood, energy, nutrition, sleep, and health service or resource utilization. The investigators analyzed responses from patients who completed at least one patient-reported outcome or journal entry between January and August 2017.
Five hundred twenty patients agreed to report outcomes using the online platform; 310 patients completed one or more patient-reported outcome or journal entry. In all, 259 patients reported daily MS sympto
“It is challenging for providers and clinicians to incorporate routine review of patient-reported outcomes into their clinic schedules,” the researchers noted. Strategies for reviewing patient-reported outcomes in clinic and eliciting more complete and timely patient reports are warranted, they said.
“Use of an online platform provides an opportunity to learn about patients’ daily experiences. Understanding these experiences can inform care strategies to address total functionality and other clinical outcomes.”
—Jake Remaly
SAN DIEGO—Continuous collection of patient-reported outcomes via a web-based platform may help neurologists identify patients with multiple sclerosis (MS) with active disease who would benefit from additional care coordination, according to a study described at the ACTRIMS 2018 Forum.
MS has a “variable and unpredictable disease course,” said Alexis Ahmad, Director of Clinical Trials for the Georgetown University Department of Neurology in Washington, DC, and colleagues. “Given the chronic and complex nature of MS, patients living with this disease require continuous and specialized care.”
The MS and Neuroimmunology Center at Georgetown University aims to become the first MS center recognized as a Patient-Centered Specialty Practice by the National Committee for Quality Assurance.
To assess whether a web-based platform to capture patient-reported outcomes can help MS practices meet Patient-Centered Specialty Practice standards and achieve MS care goals, the researchers analyzed patient responses in their center’s MS-Advance Study. The MS-Advance Study includes patients from the practice who use a web-based platform to relay patient-reported outcomes in between and at clinic visits.
Patients completed scales and questionnaires at baseline (eg, Modified Fatigue Impact Scale, Patient Health Questionnaire-9, Work Productivity and Activity Improvement Questionnaire, and Treatment Satisfaction Questionnaire for Medication) and journal entries about relapses, mood, energy, nutrition, sleep, and health service or resource utilization. The investigators analyzed responses from patients who completed at least one patient-reported outcome or journal entry between January and August 2017.
Five hundred twenty patients agreed to report outcomes using the online platform; 310 patients completed one or more patient-reported outcome or journal entry. In all, 259 patients reported daily MS sympto
“It is challenging for providers and clinicians to incorporate routine review of patient-reported outcomes into their clinic schedules,” the researchers noted. Strategies for reviewing patient-reported outcomes in clinic and eliciting more complete and timely patient reports are warranted, they said.
“Use of an online platform provides an opportunity to learn about patients’ daily experiences. Understanding these experiences can inform care strategies to address total functionality and other clinical outcomes.”
—Jake Remaly
Health Care Use May Elucidate MS Prodrome
SAN DIEGO—In the five years before multiple sclerosis (MS) symptom onset, patients are more likely to see a physician or be admitted to the hospital for problems related to the nervous system, sensory organs, musculoskeletal system, and genitourinary system, compared with controls, according to research presented at the ACTRIMS 2018 Forum.
In addition, patients with MS are more likely to see psychiatrists and urologists and to fill prescriptions related to the musculoskeletal system or the genitourinary system in the five years before MS onset.
“It is possible to measure and phenotype the MS prodrome five years before the clinical recognition of MS,” said Elaine Kingwell, PhD, a research associate in the Division of Neurology at the University of British Columbia in Vancouver, and colleagues. “Our findings inform the etiologically relevant time window and suggest that an earlier recognition and diagnosis of MS is feasible.”
A prior study by the researchers had identified increased health care use before a first demyelinating event among patients
Phenotypic Characteristics
To investigate the phenotypic characteristics of the MS prodrome, Dr. Kingwell and colleagues examined the medical diagnoses and therapeutic drug classes related to inpatient and outpatient health care encounters five years before the clinical recognition of MS. The researchers performed a population-based matched cohort study using linked health administrative and clinical data in four Canadian provinces between 1989 and 2014.
The investigators compared the reasons for physician and hospital encounters, using ICD-10 chapters or physician specialty, and prescriptions filled, using Anatomical Therapeutic Chemical System, level 1 drug classes. The researchers matched people with MS with as many as five people without any demyelinating disease by sex, year of birth, and postal code.
The study included two cohorts of people with MS. One cohort encompassed patients with a first demyelinating disease–related claim (ie, the administrative cohort), and the other cohort comprised patients with a neurologist-confirmed diagnosis of MS (ie, the clinical cohort). The researchers compared inpatient and outpatient encounters between cases and controls in the five years before the cases’ first demyelinating claim or clinically reported symptom onset.
Fewer Pregnancy-Related Encounters
The administrative cohort included 13,951 cases and 66,940 controls (about 73% women; average age, 43). The clinical cohort included 3,202 cases and 16,006 controls (about 74% women; average age, 36.5). Compared with controls, in the five years before the first demyelinating claim or symptom onset, cases had more physician and hospital encounters for the nervous system (rate ratio [RR] = 1.70–4.75), sensory organs (RR = 1.40–2.28), musculoskeletal system (RR = 1.19–1.70), and genitourinary system (RR = 1.17–1.59). Cases also had more encounters with psychiatrists (RR = 1.48–1.66) and urologists (RR = 1.49–1.80), and a higher proportion of filled prescriptions for hormonal preparations and for drugs related to the musculoskeletal or genitourinary systems (1.1–1.5 times higher). In contrast, cases had fewer pregnancy-related encounters, compared with controls (RR = 0.78–0.88).
“Research into factors that might cause or prevent MS should take the MS prodrome into account,” the researchers said.
—Jake Remaly
Suggested Reading
Wijnands JMA, Kingwell E, Zhu F, et al. Health-care use before a first demyelinating event suggestive of a multiple sclerosis prodrome: a matched cohort study. Lancet Neurol. 2017;16(6):445-451.
SAN DIEGO—In the five years before multiple sclerosis (MS) symptom onset, patients are more likely to see a physician or be admitted to the hospital for problems related to the nervous system, sensory organs, musculoskeletal system, and genitourinary system, compared with controls, according to research presented at the ACTRIMS 2018 Forum.
In addition, patients with MS are more likely to see psychiatrists and urologists and to fill prescriptions related to the musculoskeletal system or the genitourinary system in the five years before MS onset.
“It is possible to measure and phenotype the MS prodrome five years before the clinical recognition of MS,” said Elaine Kingwell, PhD, a research associate in the Division of Neurology at the University of British Columbia in Vancouver, and colleagues. “Our findings inform the etiologically relevant time window and suggest that an earlier recognition and diagnosis of MS is feasible.”
A prior study by the researchers had identified increased health care use before a first demyelinating event among patients
Phenotypic Characteristics
To investigate the phenotypic characteristics of the MS prodrome, Dr. Kingwell and colleagues examined the medical diagnoses and therapeutic drug classes related to inpatient and outpatient health care encounters five years before the clinical recognition of MS. The researchers performed a population-based matched cohort study using linked health administrative and clinical data in four Canadian provinces between 1989 and 2014.
The investigators compared the reasons for physician and hospital encounters, using ICD-10 chapters or physician specialty, and prescriptions filled, using Anatomical Therapeutic Chemical System, level 1 drug classes. The researchers matched people with MS with as many as five people without any demyelinating disease by sex, year of birth, and postal code.
The study included two cohorts of people with MS. One cohort encompassed patients with a first demyelinating disease–related claim (ie, the administrative cohort), and the other cohort comprised patients with a neurologist-confirmed diagnosis of MS (ie, the clinical cohort). The researchers compared inpatient and outpatient encounters between cases and controls in the five years before the cases’ first demyelinating claim or clinically reported symptom onset.
Fewer Pregnancy-Related Encounters
The administrative cohort included 13,951 cases and 66,940 controls (about 73% women; average age, 43). The clinical cohort included 3,202 cases and 16,006 controls (about 74% women; average age, 36.5). Compared with controls, in the five years before the first demyelinating claim or symptom onset, cases had more physician and hospital encounters for the nervous system (rate ratio [RR] = 1.70–4.75), sensory organs (RR = 1.40–2.28), musculoskeletal system (RR = 1.19–1.70), and genitourinary system (RR = 1.17–1.59). Cases also had more encounters with psychiatrists (RR = 1.48–1.66) and urologists (RR = 1.49–1.80), and a higher proportion of filled prescriptions for hormonal preparations and for drugs related to the musculoskeletal or genitourinary systems (1.1–1.5 times higher). In contrast, cases had fewer pregnancy-related encounters, compared with controls (RR = 0.78–0.88).
“Research into factors that might cause or prevent MS should take the MS prodrome into account,” the researchers said.
—Jake Remaly
Suggested Reading
Wijnands JMA, Kingwell E, Zhu F, et al. Health-care use before a first demyelinating event suggestive of a multiple sclerosis prodrome: a matched cohort study. Lancet Neurol. 2017;16(6):445-451.
SAN DIEGO—In the five years before multiple sclerosis (MS) symptom onset, patients are more likely to see a physician or be admitted to the hospital for problems related to the nervous system, sensory organs, musculoskeletal system, and genitourinary system, compared with controls, according to research presented at the ACTRIMS 2018 Forum.
In addition, patients with MS are more likely to see psychiatrists and urologists and to fill prescriptions related to the musculoskeletal system or the genitourinary system in the five years before MS onset.
“It is possible to measure and phenotype the MS prodrome five years before the clinical recognition of MS,” said Elaine Kingwell, PhD, a research associate in the Division of Neurology at the University of British Columbia in Vancouver, and colleagues. “Our findings inform the etiologically relevant time window and suggest that an earlier recognition and diagnosis of MS is feasible.”
A prior study by the researchers had identified increased health care use before a first demyelinating event among patients
Phenotypic Characteristics
To investigate the phenotypic characteristics of the MS prodrome, Dr. Kingwell and colleagues examined the medical diagnoses and therapeutic drug classes related to inpatient and outpatient health care encounters five years before the clinical recognition of MS. The researchers performed a population-based matched cohort study using linked health administrative and clinical data in four Canadian provinces between 1989 and 2014.
The investigators compared the reasons for physician and hospital encounters, using ICD-10 chapters or physician specialty, and prescriptions filled, using Anatomical Therapeutic Chemical System, level 1 drug classes. The researchers matched people with MS with as many as five people without any demyelinating disease by sex, year of birth, and postal code.
The study included two cohorts of people with MS. One cohort encompassed patients with a first demyelinating disease–related claim (ie, the administrative cohort), and the other cohort comprised patients with a neurologist-confirmed diagnosis of MS (ie, the clinical cohort). The researchers compared inpatient and outpatient encounters between cases and controls in the five years before the cases’ first demyelinating claim or clinically reported symptom onset.
Fewer Pregnancy-Related Encounters
The administrative cohort included 13,951 cases and 66,940 controls (about 73% women; average age, 43). The clinical cohort included 3,202 cases and 16,006 controls (about 74% women; average age, 36.5). Compared with controls, in the five years before the first demyelinating claim or symptom onset, cases had more physician and hospital encounters for the nervous system (rate ratio [RR] = 1.70–4.75), sensory organs (RR = 1.40–2.28), musculoskeletal system (RR = 1.19–1.70), and genitourinary system (RR = 1.17–1.59). Cases also had more encounters with psychiatrists (RR = 1.48–1.66) and urologists (RR = 1.49–1.80), and a higher proportion of filled prescriptions for hormonal preparations and for drugs related to the musculoskeletal or genitourinary systems (1.1–1.5 times higher). In contrast, cases had fewer pregnancy-related encounters, compared with controls (RR = 0.78–0.88).
“Research into factors that might cause or prevent MS should take the MS prodrome into account,” the researchers said.
—Jake Remaly
Suggested Reading
Wijnands JMA, Kingwell E, Zhu F, et al. Health-care use before a first demyelinating event suggestive of a multiple sclerosis prodrome: a matched cohort study. Lancet Neurol. 2017;16(6):445-451.
Researchers find predictors of worse MS outcomes in post hoc study of three trials
SAN DIEGO – Baseline Expanded Disability Status Scale scores and number of relapses during the first year were the most consistent predictors of disability worsening or relapses over the subsequent 3 years in long-term analysis of three phase 3 fingolimod trials.
Those patients identified at higher risk for worse long-term clinical outcomes could benefit from an early review of multiple sclerosis (MS) treatment regimens to help prevent worsening disability, Pavle Repovic, MD, PhD, said in an interview.
“The idea for some time now has been to figure out what will tell whether a patient is responding to a therapy early on or not,” Dr. Repovic said at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
To explore long-term clinical predictors of disability progression and relapse risk, Dr. Repovic and his colleagues analyzed three phase 3 trials assessing fingolimod (Gilenya). They evaluated parameters at baseline and during the first year of the FREEDOMS, FREEDOMS II, and TRANSFORMS studies.
Investigators in the FREEDOMS studies enrolled 1,118 people with relapsing MS and randomized them to fingolimod or placebo for 2 years. The patients who were initially treated with placebo were then re-randomized to one of two fingolimod doses for an additional 18 months. Finally, all participants underwent treatment with the same fingolimod dose during an open-label phase. Similarly, in TRANSFORMS, researchers enrolled 1,292 people with relapsing MS and randomized them to either fingolimod or interferon beta-1a for 1 year. They then re-randomized the interferon beta-1a group to one of two fingolimod doses for a 1-year extension, then assigned all participants to one dose of fingolimod for an open-label phase.
At baseline in the FREEDOMS and FREEDOMS II studies, a longer duration of MS was associated with a higher risk for clinically definite progression or relapses at 6 months (odds ratio, 1.040), as was baseline Expanded Disability Status Scale (EDSS) score (OR, 1.229).
“In the FREEDOMS trials, what we learned is that the duration of multiple sclerosis since diagnosis and the baseline disability predict worsening between 1 and 4 years,” said Dr. Repovic, a neurologist with the Multiple Sclerosis Center at the Swedish Neuroscience Institute in Seattle.
Dr. Repovic added that “there is nothing you can do about them [the baseline predictors]. Patients are as disabled as they are, and they’ve had multiple sclerosis as long as they have.”
The researchers also looked at predictors of 6-month confirmed disability progression or relapses during the first year. “The idea is that year 1 might tell you what happens next,” Dr. Repovic said. The number of confirmed relapses (OR, 2.788) and MRI lesion activity (OR, 1.638), were predictive.
“So breakthrough disease in year 1 predicts more disease down the road.”
In the TRANSFORMS trial, previous treatment for MS (OR, 1.613) and EDSS score at baseline (OR, 1.228) predicted who would do worse. The number of confirmed relapses in the first year of the study was the only predictor (OR, 1.774). There was a trend for MRI lesion activity that approached statistical significance.
The confirmed number of relapses in the first year was the common predictor among all the phase 3 studies. “So with people who relapse, we really need to watch out for them,” Dr. Repovic said.
“The challenge with this particular study is we took everybody,” Dr. Repovic added. Next he would like to look at only those participants who received fingolimod throughout the trials to see if any specific predictors emerge.
Dr. Repovic disclosed he is on the speakers bureau for Novartis, the company that sponsored the study.
SOURCE: Repovic P et al. Abstract P210.
SAN DIEGO – Baseline Expanded Disability Status Scale scores and number of relapses during the first year were the most consistent predictors of disability worsening or relapses over the subsequent 3 years in long-term analysis of three phase 3 fingolimod trials.
Those patients identified at higher risk for worse long-term clinical outcomes could benefit from an early review of multiple sclerosis (MS) treatment regimens to help prevent worsening disability, Pavle Repovic, MD, PhD, said in an interview.
“The idea for some time now has been to figure out what will tell whether a patient is responding to a therapy early on or not,” Dr. Repovic said at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
To explore long-term clinical predictors of disability progression and relapse risk, Dr. Repovic and his colleagues analyzed three phase 3 trials assessing fingolimod (Gilenya). They evaluated parameters at baseline and during the first year of the FREEDOMS, FREEDOMS II, and TRANSFORMS studies.
Investigators in the FREEDOMS studies enrolled 1,118 people with relapsing MS and randomized them to fingolimod or placebo for 2 years. The patients who were initially treated with placebo were then re-randomized to one of two fingolimod doses for an additional 18 months. Finally, all participants underwent treatment with the same fingolimod dose during an open-label phase. Similarly, in TRANSFORMS, researchers enrolled 1,292 people with relapsing MS and randomized them to either fingolimod or interferon beta-1a for 1 year. They then re-randomized the interferon beta-1a group to one of two fingolimod doses for a 1-year extension, then assigned all participants to one dose of fingolimod for an open-label phase.
At baseline in the FREEDOMS and FREEDOMS II studies, a longer duration of MS was associated with a higher risk for clinically definite progression or relapses at 6 months (odds ratio, 1.040), as was baseline Expanded Disability Status Scale (EDSS) score (OR, 1.229).
“In the FREEDOMS trials, what we learned is that the duration of multiple sclerosis since diagnosis and the baseline disability predict worsening between 1 and 4 years,” said Dr. Repovic, a neurologist with the Multiple Sclerosis Center at the Swedish Neuroscience Institute in Seattle.
Dr. Repovic added that “there is nothing you can do about them [the baseline predictors]. Patients are as disabled as they are, and they’ve had multiple sclerosis as long as they have.”
The researchers also looked at predictors of 6-month confirmed disability progression or relapses during the first year. “The idea is that year 1 might tell you what happens next,” Dr. Repovic said. The number of confirmed relapses (OR, 2.788) and MRI lesion activity (OR, 1.638), were predictive.
“So breakthrough disease in year 1 predicts more disease down the road.”
In the TRANSFORMS trial, previous treatment for MS (OR, 1.613) and EDSS score at baseline (OR, 1.228) predicted who would do worse. The number of confirmed relapses in the first year of the study was the only predictor (OR, 1.774). There was a trend for MRI lesion activity that approached statistical significance.
The confirmed number of relapses in the first year was the common predictor among all the phase 3 studies. “So with people who relapse, we really need to watch out for them,” Dr. Repovic said.
“The challenge with this particular study is we took everybody,” Dr. Repovic added. Next he would like to look at only those participants who received fingolimod throughout the trials to see if any specific predictors emerge.
Dr. Repovic disclosed he is on the speakers bureau for Novartis, the company that sponsored the study.
SOURCE: Repovic P et al. Abstract P210.
SAN DIEGO – Baseline Expanded Disability Status Scale scores and number of relapses during the first year were the most consistent predictors of disability worsening or relapses over the subsequent 3 years in long-term analysis of three phase 3 fingolimod trials.
Those patients identified at higher risk for worse long-term clinical outcomes could benefit from an early review of multiple sclerosis (MS) treatment regimens to help prevent worsening disability, Pavle Repovic, MD, PhD, said in an interview.
“The idea for some time now has been to figure out what will tell whether a patient is responding to a therapy early on or not,” Dr. Repovic said at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
To explore long-term clinical predictors of disability progression and relapse risk, Dr. Repovic and his colleagues analyzed three phase 3 trials assessing fingolimod (Gilenya). They evaluated parameters at baseline and during the first year of the FREEDOMS, FREEDOMS II, and TRANSFORMS studies.
Investigators in the FREEDOMS studies enrolled 1,118 people with relapsing MS and randomized them to fingolimod or placebo for 2 years. The patients who were initially treated with placebo were then re-randomized to one of two fingolimod doses for an additional 18 months. Finally, all participants underwent treatment with the same fingolimod dose during an open-label phase. Similarly, in TRANSFORMS, researchers enrolled 1,292 people with relapsing MS and randomized them to either fingolimod or interferon beta-1a for 1 year. They then re-randomized the interferon beta-1a group to one of two fingolimod doses for a 1-year extension, then assigned all participants to one dose of fingolimod for an open-label phase.
At baseline in the FREEDOMS and FREEDOMS II studies, a longer duration of MS was associated with a higher risk for clinically definite progression or relapses at 6 months (odds ratio, 1.040), as was baseline Expanded Disability Status Scale (EDSS) score (OR, 1.229).
“In the FREEDOMS trials, what we learned is that the duration of multiple sclerosis since diagnosis and the baseline disability predict worsening between 1 and 4 years,” said Dr. Repovic, a neurologist with the Multiple Sclerosis Center at the Swedish Neuroscience Institute in Seattle.
Dr. Repovic added that “there is nothing you can do about them [the baseline predictors]. Patients are as disabled as they are, and they’ve had multiple sclerosis as long as they have.”
The researchers also looked at predictors of 6-month confirmed disability progression or relapses during the first year. “The idea is that year 1 might tell you what happens next,” Dr. Repovic said. The number of confirmed relapses (OR, 2.788) and MRI lesion activity (OR, 1.638), were predictive.
“So breakthrough disease in year 1 predicts more disease down the road.”
In the TRANSFORMS trial, previous treatment for MS (OR, 1.613) and EDSS score at baseline (OR, 1.228) predicted who would do worse. The number of confirmed relapses in the first year of the study was the only predictor (OR, 1.774). There was a trend for MRI lesion activity that approached statistical significance.
The confirmed number of relapses in the first year was the common predictor among all the phase 3 studies. “So with people who relapse, we really need to watch out for them,” Dr. Repovic said.
“The challenge with this particular study is we took everybody,” Dr. Repovic added. Next he would like to look at only those participants who received fingolimod throughout the trials to see if any specific predictors emerge.
Dr. Repovic disclosed he is on the speakers bureau for Novartis, the company that sponsored the study.
SOURCE: Repovic P et al. Abstract P210.
REPORTING FROM ACTRIMS FORUM 2018
Key clinical point: Certain factors at baseline and over first year of three pooled studies were associated with worse long-term multiple sclerosis outcomes.
Major finding: The number of confirmed relapses in the first year of the FREEDOMS, FREEDOMS II, and TRANSFORMS studies predicted worse subsequent outcomes.
Study details: Pooled data from three phase 3 trials of fingolimod with 2,355 patients.
Disclosures: Dr. Repovic disclosed he is on the speakers bureau for Novartis, the company that sponsored the study.
Source: Repovic P et al. Abstract P210.
ECTRIMS and EAN Publish Recommendations for Treating MS
The European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) and the European Academy of Neurology (EAN) have published a guideline to offer up-to-date, evidence-based recommendations for the treatment of adult patients with MS. The guideline is intended to fill a perceived need for a comprehensive document that includes information about recently approved MS therapies and helps clinicians and patients resolve difficulties in everyday clinical practice. It was published in the February issue of Multiple Sclerosis.
Authors Addressed 10 Questions
Xavier Montalban, MD, PhD, Chair and Director of the Department of Neurology and Neuroimmunology at Vall d’Hebron University Hospital in Barcelona, and colleagues agreed to investigate 10 questions related to treatment efficacy, response criteria, strategies to address suboptimal response and safety concerns, and treatment of MS in pregnancy. They developed the guideline following the recommendations of the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) Working Group, along with EAN recommendations for writing a neurologic management guideline. Literature searches relied upon databases such as the Cochrane Central Register of Controlled Trials, Excerpta Medica, and Medical Literature Analysis and Retrieval System Online.
The authors evaluated data for all disease-modifying treatments (DMTs) approved by the European Medicines Agency at the time of publication. They did not consider combination therapies, complementary or alternative medicine, or symptomatic treatment. Although focused on Europe, the guideline does not account for regulatory or organizational issues specific to any European country.
Strong Recommendations
Dr. Montalban and colleagues agreed upon 21 recommendations and consensus statements. The recommendations were categorized as strong or weak, according to the quality of evidence and the risk–benefit balance. The authors formulated consensus statements on questions for which evidence was insufficient to support a formal recommendation.
The first of the guideline’s three strong recommendations is that neurologists offer interferon or glatiramer acetate to patients with clinically isolated syndrome (CIS) and an abnormal MRI with lesions suggestive of MS who do not fulfill criteria for MS. The second is to offer early treatment with DMTs to patients with active relapsing-remitting MS, as defined by clinical relapses or MRI activity. The authors defined active lesions as contrast-enhancing lesions or new or unequivocally enlarging T2 lesions assessed at least annually. This recommendation also applies to patients with CIS who fulfill current diagnostic criteria for MS. The third strong recommendation is to offer a more efficacious drug to patients treated with interferon or glatiramer acetate who show evidence of disease activity.
Weak Recommendations
Nine of the guideline’s recommendations are categorized as weak. For example, neurologists should consider treating patients with active secondary progressive MS with interferon beta-1a or -1b, taking into account the treatments’ “dubious efficacy,” as well as their safety and tolerability, according to the authors. Neurologists should consider mitoxantrone, ocrelizumab, and cladribine for this population. The authors recommend considering ocrelizumab as a treatment for patients with primary progressive MS.
As a way of monitoring treatment response, the authors recommend that neurologists consider combining MRI with clinical measures when evaluating disease evolution in treated patients. When making treatment decisions in the event of safety concerns, neurologists should consider the possibility that disease activity may resume or rebound if treatment is stopped, particularly with natalizumab, said Dr. Montalban and colleagues. Continuation of DMT treatment should be considered for patients who are clinically stable, who have stable MRI, and who have no problems with safety or tolerability, according to the guideline.
“For women planning a pregnancy, if there is a high risk of disease reactivation, consider using interferon or glatiramer acetate until pregnancy is confirmed,” said the authors. “In some very specific (active) cases, continuing this treatment during pregnancy could also be considered.” Delaying pregnancy is advisable for women with persistent high disease activity. If such a woman decides to become pregnant or has an unplanned pregnancy, neurologists may consider treatment with natalizumab throughout pregnancy after a full discussion with the patient of the potential implications. “Treatment with alemtuzumab could be an alternative therapeutic option for planned pregnancy in very active cases, provided that a four-month interval is strictly observed from the latest infusion until conception,” said Dr. Montalban and colleagues.
Consensus Statements
Several of the guideline’s consensus statements relate to the monitoring of treatment response. For patients treated with DMTs, the authors recommend performing a standardized reference brain MRI within six months of treatment onset, and comparing it with a subsequent brain MRI performed 12 months after starting treatment. The measurement of new or unequivocally enlarging T2 lesions is the preferred MRI method of gauging treatment response, supplemented by gadolinium-enhancing lesions. To monitor treatment safety, the authors recommend performing a standardized reference brain MRI every year in patients at low risk for progressive multifocal leukoencephalopathy (PML), and every three to six months in patients at high risk for PML.
If the neurologist and patient have decided to switch therapies, they should consider the patient’s characteristics and comorbidities, drug safety profiles, and disease severity when choosing a new DMT. If treatment of a highly efficacious drug is stopped because of safety or efficacy problems, the neurologist should consider starting another highly efficacious drug, according to the guideline. Disease activity, the drug’s half-life and biologic activity, and the potential for resumed disease activity or rebound should be considered when choosing a new therapy, said Dr. Montalban and colleagues.
—Erik Greb
Suggested Reading
Montalban X, Gold R, Thompson AJ, et al. ECTRIMS/EAN Guideline on the pharmacological treatment of people with multiple sclerosis. Mult Scler. 2018;24(2):96-120.
The European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) and the European Academy of Neurology (EAN) have published a guideline to offer up-to-date, evidence-based recommendations for the treatment of adult patients with MS. The guideline is intended to fill a perceived need for a comprehensive document that includes information about recently approved MS therapies and helps clinicians and patients resolve difficulties in everyday clinical practice. It was published in the February issue of Multiple Sclerosis.
Authors Addressed 10 Questions
Xavier Montalban, MD, PhD, Chair and Director of the Department of Neurology and Neuroimmunology at Vall d’Hebron University Hospital in Barcelona, and colleagues agreed to investigate 10 questions related to treatment efficacy, response criteria, strategies to address suboptimal response and safety concerns, and treatment of MS in pregnancy. They developed the guideline following the recommendations of the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) Working Group, along with EAN recommendations for writing a neurologic management guideline. Literature searches relied upon databases such as the Cochrane Central Register of Controlled Trials, Excerpta Medica, and Medical Literature Analysis and Retrieval System Online.
The authors evaluated data for all disease-modifying treatments (DMTs) approved by the European Medicines Agency at the time of publication. They did not consider combination therapies, complementary or alternative medicine, or symptomatic treatment. Although focused on Europe, the guideline does not account for regulatory or organizational issues specific to any European country.
Strong Recommendations
Dr. Montalban and colleagues agreed upon 21 recommendations and consensus statements. The recommendations were categorized as strong or weak, according to the quality of evidence and the risk–benefit balance. The authors formulated consensus statements on questions for which evidence was insufficient to support a formal recommendation.
The first of the guideline’s three strong recommendations is that neurologists offer interferon or glatiramer acetate to patients with clinically isolated syndrome (CIS) and an abnormal MRI with lesions suggestive of MS who do not fulfill criteria for MS. The second is to offer early treatment with DMTs to patients with active relapsing-remitting MS, as defined by clinical relapses or MRI activity. The authors defined active lesions as contrast-enhancing lesions or new or unequivocally enlarging T2 lesions assessed at least annually. This recommendation also applies to patients with CIS who fulfill current diagnostic criteria for MS. The third strong recommendation is to offer a more efficacious drug to patients treated with interferon or glatiramer acetate who show evidence of disease activity.
Weak Recommendations
Nine of the guideline’s recommendations are categorized as weak. For example, neurologists should consider treating patients with active secondary progressive MS with interferon beta-1a or -1b, taking into account the treatments’ “dubious efficacy,” as well as their safety and tolerability, according to the authors. Neurologists should consider mitoxantrone, ocrelizumab, and cladribine for this population. The authors recommend considering ocrelizumab as a treatment for patients with primary progressive MS.
As a way of monitoring treatment response, the authors recommend that neurologists consider combining MRI with clinical measures when evaluating disease evolution in treated patients. When making treatment decisions in the event of safety concerns, neurologists should consider the possibility that disease activity may resume or rebound if treatment is stopped, particularly with natalizumab, said Dr. Montalban and colleagues. Continuation of DMT treatment should be considered for patients who are clinically stable, who have stable MRI, and who have no problems with safety or tolerability, according to the guideline.
“For women planning a pregnancy, if there is a high risk of disease reactivation, consider using interferon or glatiramer acetate until pregnancy is confirmed,” said the authors. “In some very specific (active) cases, continuing this treatment during pregnancy could also be considered.” Delaying pregnancy is advisable for women with persistent high disease activity. If such a woman decides to become pregnant or has an unplanned pregnancy, neurologists may consider treatment with natalizumab throughout pregnancy after a full discussion with the patient of the potential implications. “Treatment with alemtuzumab could be an alternative therapeutic option for planned pregnancy in very active cases, provided that a four-month interval is strictly observed from the latest infusion until conception,” said Dr. Montalban and colleagues.
Consensus Statements
Several of the guideline’s consensus statements relate to the monitoring of treatment response. For patients treated with DMTs, the authors recommend performing a standardized reference brain MRI within six months of treatment onset, and comparing it with a subsequent brain MRI performed 12 months after starting treatment. The measurement of new or unequivocally enlarging T2 lesions is the preferred MRI method of gauging treatment response, supplemented by gadolinium-enhancing lesions. To monitor treatment safety, the authors recommend performing a standardized reference brain MRI every year in patients at low risk for progressive multifocal leukoencephalopathy (PML), and every three to six months in patients at high risk for PML.
If the neurologist and patient have decided to switch therapies, they should consider the patient’s characteristics and comorbidities, drug safety profiles, and disease severity when choosing a new DMT. If treatment of a highly efficacious drug is stopped because of safety or efficacy problems, the neurologist should consider starting another highly efficacious drug, according to the guideline. Disease activity, the drug’s half-life and biologic activity, and the potential for resumed disease activity or rebound should be considered when choosing a new therapy, said Dr. Montalban and colleagues.
—Erik Greb
Suggested Reading
Montalban X, Gold R, Thompson AJ, et al. ECTRIMS/EAN Guideline on the pharmacological treatment of people with multiple sclerosis. Mult Scler. 2018;24(2):96-120.
The European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) and the European Academy of Neurology (EAN) have published a guideline to offer up-to-date, evidence-based recommendations for the treatment of adult patients with MS. The guideline is intended to fill a perceived need for a comprehensive document that includes information about recently approved MS therapies and helps clinicians and patients resolve difficulties in everyday clinical practice. It was published in the February issue of Multiple Sclerosis.
Authors Addressed 10 Questions
Xavier Montalban, MD, PhD, Chair and Director of the Department of Neurology and Neuroimmunology at Vall d’Hebron University Hospital in Barcelona, and colleagues agreed to investigate 10 questions related to treatment efficacy, response criteria, strategies to address suboptimal response and safety concerns, and treatment of MS in pregnancy. They developed the guideline following the recommendations of the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) Working Group, along with EAN recommendations for writing a neurologic management guideline. Literature searches relied upon databases such as the Cochrane Central Register of Controlled Trials, Excerpta Medica, and Medical Literature Analysis and Retrieval System Online.
The authors evaluated data for all disease-modifying treatments (DMTs) approved by the European Medicines Agency at the time of publication. They did not consider combination therapies, complementary or alternative medicine, or symptomatic treatment. Although focused on Europe, the guideline does not account for regulatory or organizational issues specific to any European country.
Strong Recommendations
Dr. Montalban and colleagues agreed upon 21 recommendations and consensus statements. The recommendations were categorized as strong or weak, according to the quality of evidence and the risk–benefit balance. The authors formulated consensus statements on questions for which evidence was insufficient to support a formal recommendation.
The first of the guideline’s three strong recommendations is that neurologists offer interferon or glatiramer acetate to patients with clinically isolated syndrome (CIS) and an abnormal MRI with lesions suggestive of MS who do not fulfill criteria for MS. The second is to offer early treatment with DMTs to patients with active relapsing-remitting MS, as defined by clinical relapses or MRI activity. The authors defined active lesions as contrast-enhancing lesions or new or unequivocally enlarging T2 lesions assessed at least annually. This recommendation also applies to patients with CIS who fulfill current diagnostic criteria for MS. The third strong recommendation is to offer a more efficacious drug to patients treated with interferon or glatiramer acetate who show evidence of disease activity.
Weak Recommendations
Nine of the guideline’s recommendations are categorized as weak. For example, neurologists should consider treating patients with active secondary progressive MS with interferon beta-1a or -1b, taking into account the treatments’ “dubious efficacy,” as well as their safety and tolerability, according to the authors. Neurologists should consider mitoxantrone, ocrelizumab, and cladribine for this population. The authors recommend considering ocrelizumab as a treatment for patients with primary progressive MS.
As a way of monitoring treatment response, the authors recommend that neurologists consider combining MRI with clinical measures when evaluating disease evolution in treated patients. When making treatment decisions in the event of safety concerns, neurologists should consider the possibility that disease activity may resume or rebound if treatment is stopped, particularly with natalizumab, said Dr. Montalban and colleagues. Continuation of DMT treatment should be considered for patients who are clinically stable, who have stable MRI, and who have no problems with safety or tolerability, according to the guideline.
“For women planning a pregnancy, if there is a high risk of disease reactivation, consider using interferon or glatiramer acetate until pregnancy is confirmed,” said the authors. “In some very specific (active) cases, continuing this treatment during pregnancy could also be considered.” Delaying pregnancy is advisable for women with persistent high disease activity. If such a woman decides to become pregnant or has an unplanned pregnancy, neurologists may consider treatment with natalizumab throughout pregnancy after a full discussion with the patient of the potential implications. “Treatment with alemtuzumab could be an alternative therapeutic option for planned pregnancy in very active cases, provided that a four-month interval is strictly observed from the latest infusion until conception,” said Dr. Montalban and colleagues.
Consensus Statements
Several of the guideline’s consensus statements relate to the monitoring of treatment response. For patients treated with DMTs, the authors recommend performing a standardized reference brain MRI within six months of treatment onset, and comparing it with a subsequent brain MRI performed 12 months after starting treatment. The measurement of new or unequivocally enlarging T2 lesions is the preferred MRI method of gauging treatment response, supplemented by gadolinium-enhancing lesions. To monitor treatment safety, the authors recommend performing a standardized reference brain MRI every year in patients at low risk for progressive multifocal leukoencephalopathy (PML), and every three to six months in patients at high risk for PML.
If the neurologist and patient have decided to switch therapies, they should consider the patient’s characteristics and comorbidities, drug safety profiles, and disease severity when choosing a new DMT. If treatment of a highly efficacious drug is stopped because of safety or efficacy problems, the neurologist should consider starting another highly efficacious drug, according to the guideline. Disease activity, the drug’s half-life and biologic activity, and the potential for resumed disease activity or rebound should be considered when choosing a new therapy, said Dr. Montalban and colleagues.
—Erik Greb
Suggested Reading
Montalban X, Gold R, Thompson AJ, et al. ECTRIMS/EAN Guideline on the pharmacological treatment of people with multiple sclerosis. Mult Scler. 2018;24(2):96-120.
Fingolimod switch from an injectable linked to improved outcomes in relapsing MS
SAN DIEGO – People with relapsing multiple sclerosis who switched from an injectable disease-modifying therapy to fingolimod after the randomized phase of the PREFERMS trial experienced improved annualized relapse rates, exposure-adjusted percentage brain volume loss, and satisfaction, compared with study participants who did not switch.
Importantly, there were no differences between switchers who were treatment-naive or previously treated with one injectable therapy class at baseline in the phase 4, active-controlled, open-label, and multicenter PREFERMS trial, according to the researchers.
Of the 875 patients with relapsing multiple sclerosis, 254 or 58% of those initially randomized to an injectable disease-modifying therapy (iDMT) switched to fingolimod (Gilenya) during the 12-month study. Participants in the trial were permitted to switch therapy once after at least 3 months, or sooner if warranted for safety or efficacy reasons.
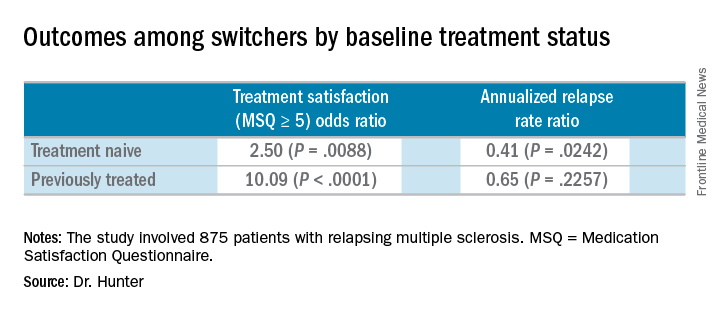
Compared with the end the randomization phase, patients who switched from an iDMT to fingolimod were more satisfied, defined as a Medication Satisfaction Questionnaire score of 5 or greater (odds ratio, 5.65; P less than .001) at 12 months. At the same time, their annualized relapse rate decreased (ARR ratio, 0.53; P = .0158) and their exposure-adjusted brain volume loss decreased from 0.874% to 0.463%.
The benefit of these switches was observed regardless of previous treatment status, Dr. Hunter said during a poster presentation at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
At the start of the PREFERMS trial, patients were randomized 1:1 to fingolimod 0.5 mg/day or a preselected iDMT. Just more than half, 54%, were treatment-naive and 46% had previous interferon or glatiramer acetate treatment.
“Generally, because most of these people were switched, and we’re looking at people who were treatment-naive or previously treated, when you look at brain volume loss, it’s very low on fingolimod versus high while they’re on injectable therapies,” Dr. Hunter said.
Dr. Hunter and his colleagues reported similar relapse rates and brain volume loss outcomes among the 254 patients who switched to fingolimod mid-study according to baseline treatment history. The investigators noted, however, that these post hoc analyses and P values are intended for hypothesis generation only.
In the 99 treatment-naive switchers, brain volume loss was 0.54% at the end of the study, compared with 0.84% at the end of the randomization period. Among the 155 previously treated switchers, brain volume loss also decreased, from 0.90% at the end of randomization to 0.42% at study end.
Dr. Hunter disclosed that he is an investigator for Novartis, the sponsor of the study.
SOURCE: Hunter S et al. ACTRIMS Forum 2018 Abstract P019.
SAN DIEGO – People with relapsing multiple sclerosis who switched from an injectable disease-modifying therapy to fingolimod after the randomized phase of the PREFERMS trial experienced improved annualized relapse rates, exposure-adjusted percentage brain volume loss, and satisfaction, compared with study participants who did not switch.
Importantly, there were no differences between switchers who were treatment-naive or previously treated with one injectable therapy class at baseline in the phase 4, active-controlled, open-label, and multicenter PREFERMS trial, according to the researchers.
Of the 875 patients with relapsing multiple sclerosis, 254 or 58% of those initially randomized to an injectable disease-modifying therapy (iDMT) switched to fingolimod (Gilenya) during the 12-month study. Participants in the trial were permitted to switch therapy once after at least 3 months, or sooner if warranted for safety or efficacy reasons.
Compared with the end the randomization phase, patients who switched from an iDMT to fingolimod were more satisfied, defined as a Medication Satisfaction Questionnaire score of 5 or greater (odds ratio, 5.65; P less than .001) at 12 months. At the same time, their annualized relapse rate decreased (ARR ratio, 0.53; P = .0158) and their exposure-adjusted brain volume loss decreased from 0.874% to 0.463%.
The benefit of these switches was observed regardless of previous treatment status, Dr. Hunter said during a poster presentation at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.

At the start of the PREFERMS trial, patients were randomized 1:1 to fingolimod 0.5 mg/day or a preselected iDMT. Just more than half, 54%, were treatment-naive and 46% had previous interferon or glatiramer acetate treatment.
“Generally, because most of these people were switched, and we’re looking at people who were treatment-naive or previously treated, when you look at brain volume loss, it’s very low on fingolimod versus high while they’re on injectable therapies,” Dr. Hunter said.
Dr. Hunter and his colleagues reported similar relapse rates and brain volume loss outcomes among the 254 patients who switched to fingolimod mid-study according to baseline treatment history. The investigators noted, however, that these post hoc analyses and P values are intended for hypothesis generation only.
In the 99 treatment-naive switchers, brain volume loss was 0.54% at the end of the study, compared with 0.84% at the end of the randomization period. Among the 155 previously treated switchers, brain volume loss also decreased, from 0.90% at the end of randomization to 0.42% at study end.
Dr. Hunter disclosed that he is an investigator for Novartis, the sponsor of the study.
SOURCE: Hunter S et al. ACTRIMS Forum 2018 Abstract P019.
SAN DIEGO – People with relapsing multiple sclerosis who switched from an injectable disease-modifying therapy to fingolimod after the randomized phase of the PREFERMS trial experienced improved annualized relapse rates, exposure-adjusted percentage brain volume loss, and satisfaction, compared with study participants who did not switch.
Importantly, there were no differences between switchers who were treatment-naive or previously treated with one injectable therapy class at baseline in the phase 4, active-controlled, open-label, and multicenter PREFERMS trial, according to the researchers.
Of the 875 patients with relapsing multiple sclerosis, 254 or 58% of those initially randomized to an injectable disease-modifying therapy (iDMT) switched to fingolimod (Gilenya) during the 12-month study. Participants in the trial were permitted to switch therapy once after at least 3 months, or sooner if warranted for safety or efficacy reasons.
Compared with the end the randomization phase, patients who switched from an iDMT to fingolimod were more satisfied, defined as a Medication Satisfaction Questionnaire score of 5 or greater (odds ratio, 5.65; P less than .001) at 12 months. At the same time, their annualized relapse rate decreased (ARR ratio, 0.53; P = .0158) and their exposure-adjusted brain volume loss decreased from 0.874% to 0.463%.
The benefit of these switches was observed regardless of previous treatment status, Dr. Hunter said during a poster presentation at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.

At the start of the PREFERMS trial, patients were randomized 1:1 to fingolimod 0.5 mg/day or a preselected iDMT. Just more than half, 54%, were treatment-naive and 46% had previous interferon or glatiramer acetate treatment.
“Generally, because most of these people were switched, and we’re looking at people who were treatment-naive or previously treated, when you look at brain volume loss, it’s very low on fingolimod versus high while they’re on injectable therapies,” Dr. Hunter said.
Dr. Hunter and his colleagues reported similar relapse rates and brain volume loss outcomes among the 254 patients who switched to fingolimod mid-study according to baseline treatment history. The investigators noted, however, that these post hoc analyses and P values are intended for hypothesis generation only.
In the 99 treatment-naive switchers, brain volume loss was 0.54% at the end of the study, compared with 0.84% at the end of the randomization period. Among the 155 previously treated switchers, brain volume loss also decreased, from 0.90% at the end of randomization to 0.42% at study end.
Dr. Hunter disclosed that he is an investigator for Novartis, the sponsor of the study.
SOURCE: Hunter S et al. ACTRIMS Forum 2018 Abstract P019.
REPORTING FROM ACTRIMS FORUM 2018
Key clinical point:
Major finding: Patients who switched from an injectable disease-modifying therapy to fingolimod were more satisfied, defined as a Medication Satisfaction Questionnaire score of 5 or greater (odds ratio, 5.65; P less than .001).
Study details: Post hoc analyses of the extension study of the PREFERMS trial.
Disclosures: Dr. Hunter disclosed that he is an investigator for Novartis, the sponsor of the study.
Source: Hunter S et al. ACTRIMS Forum 2018 Abstract P019.
Extended-interval dosing of natalizumab linked to lower risk of PML
SAN DIEGO – A large industry-funded study of the multiple sclerosis drug natalizumab suggests that physicians can dramatically lower the likelihood of progressive multifocal leukoencephalopathy in patients at higher risk of the condition by increasing the interval between doses.
Previous research has suggested that natalizumab (Tysabri) doesn’t lose efficacy when given less frequently.
Dr. Zhovtis Ryerson presented the study findings at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
As a 2014 report explains, natalizumab is a “highly effective disease-modifying therapy for the treatment of relapsing forms of multiple sclerosis,” but the risk of progressive multifocal leukoencephalopathy (PML) “has largely contributed to it being relegated to a second-line position” (Ther Adv Chronic Dis. 2014 Mar;5[2]:62-8).
Patients previously exposed to John Cunningham (JC) virus are at higher risk of PML, a rare viral brain disease that can be severely disabling or deadly. A 2017 report estimated the combined cumulative PML probability at 2.7% (with previous immunosuppressant use) and 1.7% (no previous immunosuppressant use) over 6 years in patients with signs of exposure to the JC virus (Lancet Neurol. 2017 Nov;16[11]:925-33). According to Dr. Zhovtis Ryerson, natalizumab manufacturer Biogen reported in December 2017 that it has confirmed 756 cases of PML in natalizumab-treated patients, all except for 3 in MS patients.
Doctors are wary in the at-risk patient population. “In general,” she said, “clinicians treating patients who are JC virus positive, and therefore have a risk for PML, may not utilize the drug or may take the patient off it after 2 years, which is when the risk goes up.”
Some physicians have experimented with longer intervals between treatments of 300 mg given intravenously, administering it every 5-8 weeks instead of every 4 weeks, with an eye on not extending the interval for too long “because MS disease activity returns after 12 weeks of withholding therapy,” Dr. Zhovtis Ryerson said.
In 2016, Dr. Zhovtis Ryerson and colleagues reported in a retrospective analysis that natalizumab dosing intervals of up to 8 weeks, 5 days didn’t affect the drug’s efficacy (J Neurol Neurosurg Psychiatry. 2016 Aug;87[8]:885-9).
For the primary analyses in the new study – whether dosing history in the last 18 months of natalizumab treatment affects PML – researchers tracked 1,988 patients who took the drug via an extended interval schedule and 13,132 who took it via a standard interval. The patients were tracked in the mandated TOUCH Prescribing Program; all participants showed signs of exposure to the JC virus.
The two groups were similar at about 68% female, an average age at first infusion of about 43, and a median number of about 48 natalizumab infusions.
For patients without previous immunosuppressant use who had received 49-60 doses, the researchers estimated the incidence of PML per 1,000 patients as 1.23 in the extended-interval group and 3.96 in the standard-interval group. The numbers were slightly higher for those who received 61-72 doses. At 48 or fewer doses, there were no PML cases in patients on extended-interval dosing.
“The data showed highly significant risk reductions of up to 94%,” Dr. Zhovtis Ryerson said.
Moving forward, “in collaboration with Biogen, we have more sensitivity analysis to be done to assure that our conclusions on this are correct,” she said. “Furthermore, more evidence of efficacy of extended-interval dosing natalizumab is needed, and we hope to move forward with a prospective, randomized, controlled trial to give clinicians the highest level of evidence we can.”
The study was funded by Biogen. Dr. Zhovtis Ryerson reported personal compensation from Teva, speaker/advisory board activities for Biogen, and research support from Biogen. Seven other authors reported being Biogen employees. Other authors reported no disclosures or various disclosures, including connections to Biogen.
SOURCE: Zhovtis R et al. ACTRIMS Forum 2018 Abstract LB250.
SAN DIEGO – A large industry-funded study of the multiple sclerosis drug natalizumab suggests that physicians can dramatically lower the likelihood of progressive multifocal leukoencephalopathy in patients at higher risk of the condition by increasing the interval between doses.
Previous research has suggested that natalizumab (Tysabri) doesn’t lose efficacy when given less frequently.
Dr. Zhovtis Ryerson presented the study findings at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
As a 2014 report explains, natalizumab is a “highly effective disease-modifying therapy for the treatment of relapsing forms of multiple sclerosis,” but the risk of progressive multifocal leukoencephalopathy (PML) “has largely contributed to it being relegated to a second-line position” (Ther Adv Chronic Dis. 2014 Mar;5[2]:62-8).
Patients previously exposed to John Cunningham (JC) virus are at higher risk of PML, a rare viral brain disease that can be severely disabling or deadly. A 2017 report estimated the combined cumulative PML probability at 2.7% (with previous immunosuppressant use) and 1.7% (no previous immunosuppressant use) over 6 years in patients with signs of exposure to the JC virus (Lancet Neurol. 2017 Nov;16[11]:925-33). According to Dr. Zhovtis Ryerson, natalizumab manufacturer Biogen reported in December 2017 that it has confirmed 756 cases of PML in natalizumab-treated patients, all except for 3 in MS patients.
Doctors are wary in the at-risk patient population. “In general,” she said, “clinicians treating patients who are JC virus positive, and therefore have a risk for PML, may not utilize the drug or may take the patient off it after 2 years, which is when the risk goes up.”
Some physicians have experimented with longer intervals between treatments of 300 mg given intravenously, administering it every 5-8 weeks instead of every 4 weeks, with an eye on not extending the interval for too long “because MS disease activity returns after 12 weeks of withholding therapy,” Dr. Zhovtis Ryerson said.
In 2016, Dr. Zhovtis Ryerson and colleagues reported in a retrospective analysis that natalizumab dosing intervals of up to 8 weeks, 5 days didn’t affect the drug’s efficacy (J Neurol Neurosurg Psychiatry. 2016 Aug;87[8]:885-9).
For the primary analyses in the new study – whether dosing history in the last 18 months of natalizumab treatment affects PML – researchers tracked 1,988 patients who took the drug via an extended interval schedule and 13,132 who took it via a standard interval. The patients were tracked in the mandated TOUCH Prescribing Program; all participants showed signs of exposure to the JC virus.
The two groups were similar at about 68% female, an average age at first infusion of about 43, and a median number of about 48 natalizumab infusions.
For patients without previous immunosuppressant use who had received 49-60 doses, the researchers estimated the incidence of PML per 1,000 patients as 1.23 in the extended-interval group and 3.96 in the standard-interval group. The numbers were slightly higher for those who received 61-72 doses. At 48 or fewer doses, there were no PML cases in patients on extended-interval dosing.
“The data showed highly significant risk reductions of up to 94%,” Dr. Zhovtis Ryerson said.
Moving forward, “in collaboration with Biogen, we have more sensitivity analysis to be done to assure that our conclusions on this are correct,” she said. “Furthermore, more evidence of efficacy of extended-interval dosing natalizumab is needed, and we hope to move forward with a prospective, randomized, controlled trial to give clinicians the highest level of evidence we can.”
The study was funded by Biogen. Dr. Zhovtis Ryerson reported personal compensation from Teva, speaker/advisory board activities for Biogen, and research support from Biogen. Seven other authors reported being Biogen employees. Other authors reported no disclosures or various disclosures, including connections to Biogen.
SOURCE: Zhovtis R et al. ACTRIMS Forum 2018 Abstract LB250.
SAN DIEGO – A large industry-funded study of the multiple sclerosis drug natalizumab suggests that physicians can dramatically lower the likelihood of progressive multifocal leukoencephalopathy in patients at higher risk of the condition by increasing the interval between doses.
Previous research has suggested that natalizumab (Tysabri) doesn’t lose efficacy when given less frequently.
Dr. Zhovtis Ryerson presented the study findings at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
As a 2014 report explains, natalizumab is a “highly effective disease-modifying therapy for the treatment of relapsing forms of multiple sclerosis,” but the risk of progressive multifocal leukoencephalopathy (PML) “has largely contributed to it being relegated to a second-line position” (Ther Adv Chronic Dis. 2014 Mar;5[2]:62-8).
Patients previously exposed to John Cunningham (JC) virus are at higher risk of PML, a rare viral brain disease that can be severely disabling or deadly. A 2017 report estimated the combined cumulative PML probability at 2.7% (with previous immunosuppressant use) and 1.7% (no previous immunosuppressant use) over 6 years in patients with signs of exposure to the JC virus (Lancet Neurol. 2017 Nov;16[11]:925-33). According to Dr. Zhovtis Ryerson, natalizumab manufacturer Biogen reported in December 2017 that it has confirmed 756 cases of PML in natalizumab-treated patients, all except for 3 in MS patients.
Doctors are wary in the at-risk patient population. “In general,” she said, “clinicians treating patients who are JC virus positive, and therefore have a risk for PML, may not utilize the drug or may take the patient off it after 2 years, which is when the risk goes up.”
Some physicians have experimented with longer intervals between treatments of 300 mg given intravenously, administering it every 5-8 weeks instead of every 4 weeks, with an eye on not extending the interval for too long “because MS disease activity returns after 12 weeks of withholding therapy,” Dr. Zhovtis Ryerson said.
In 2016, Dr. Zhovtis Ryerson and colleagues reported in a retrospective analysis that natalizumab dosing intervals of up to 8 weeks, 5 days didn’t affect the drug’s efficacy (J Neurol Neurosurg Psychiatry. 2016 Aug;87[8]:885-9).
For the primary analyses in the new study – whether dosing history in the last 18 months of natalizumab treatment affects PML – researchers tracked 1,988 patients who took the drug via an extended interval schedule and 13,132 who took it via a standard interval. The patients were tracked in the mandated TOUCH Prescribing Program; all participants showed signs of exposure to the JC virus.
The two groups were similar at about 68% female, an average age at first infusion of about 43, and a median number of about 48 natalizumab infusions.
For patients without previous immunosuppressant use who had received 49-60 doses, the researchers estimated the incidence of PML per 1,000 patients as 1.23 in the extended-interval group and 3.96 in the standard-interval group. The numbers were slightly higher for those who received 61-72 doses. At 48 or fewer doses, there were no PML cases in patients on extended-interval dosing.
“The data showed highly significant risk reductions of up to 94%,” Dr. Zhovtis Ryerson said.
Moving forward, “in collaboration with Biogen, we have more sensitivity analysis to be done to assure that our conclusions on this are correct,” she said. “Furthermore, more evidence of efficacy of extended-interval dosing natalizumab is needed, and we hope to move forward with a prospective, randomized, controlled trial to give clinicians the highest level of evidence we can.”
The study was funded by Biogen. Dr. Zhovtis Ryerson reported personal compensation from Teva, speaker/advisory board activities for Biogen, and research support from Biogen. Seven other authors reported being Biogen employees. Other authors reported no disclosures or various disclosures, including connections to Biogen.
SOURCE: Zhovtis R et al. ACTRIMS Forum 2018 Abstract LB250.
REPORTING FROM ACTRIMS FORUM 2018
Key clinical point: , compared with standard-interval dosing in patients who showed signs of exposure to the JC virus.
Major finding: Estimated incidence of PML per 1,000 patients was 1.23 in an extended-interval group and 3.96 in a standard-interval group among those who had received 49-60 natalizumab doses.
Study details: 18-month analysis of multiple sclerosis patients on natalizumab who showed signs of exposure to the JC virus: 1,988 on extended-interval dosing and 13,132 on standard-interval dosing.
Disclosures: The study was funded by Biogen, and multiple study authors report being employees of Biogen or having other relationships to the company.
Source: Zhovtis R et al. ACTRIMS Forum 2018, Abstract LB250.
Is MS caused by one-two punch of pinworm and Epstein-Barr virus?
SAN DIEGO – What causes multiple sclerosis (MS)? A team of Scottish researchers offers a new theory that it’s triggered in part by a one-two punch of infection with pinworm – a common condition in the United States, especially among children – and the Epstein-Barr virus (EBV).
The theory identifies pinworm as the prime suspect to be the “missing link” that explains why EBV and MS are so tightly connected, said Patrick Kearns, MBChB, a graduate student at Harvard T.H. Chan School of Public Health in Boston.
Dr. Kearns is the lead author of two reports about the possible role of pinworm that were presented at ACTRIMS Forum 2018, which is held by the Americas Committee for Treatment and Research in Multiple Sclerosis. He spoke in an interview.
Dr. Kearns and his colleagues focused on a well-known cluster of MS cases that began to appear in the Faroe Islands – a Danish possession in the North Atlantic – during World War II. The cases began to appear after British troops occupied the islands.
“Many of the occupation soldiers were from the Scottish Highlands, where the MS prevalence is quite high: 90 cases per 100,000, comparable to the northern U.S.,” according to a National MS Society summary about MS clusters.
In a theory that spawned controversy, the late neurologist John Kurtzke, MD, speculated that the British soldiers brought a transmissible agent to the islands, which triggered MS cases.
Could the agent be EBV alone? The authors of the new studies don’t think so, although they note that EBV is “robustly linked” to MS. Indeed, a 2012 meta-analysis reported that the virus “appears to be present in 100% of MS patients,” based on studies considered to be the strongest (Mult Scler. 2013 Feb;19[2]:162-6).
The authors of the new reports note that, while EBV infection “appears necessary,” it is “clearly not sufficient” to cause the disease on its own.
“Certainly almost everyone gets EBV eventually,” Dr. Kearns said. “So mere presence of the virus is certainly not sufficient for causing the disease. But it seems still to be necessary, and timing of infection might be everything.”
So what’s the missing piece of the puzzle?
Dr. Kearns began to think it might be the lowly pinworm after helping a colleague by analyzing data from appendicitis samples in children. He noted that uninflamed samples often had pinworms in them, but the inflamed samples often didn’t, which suggested that “the rate of pinworms in normal appendices must be very high in the healthy pediatric population at any given time.”
More data confirmed this to be true, and medical literature told Dr. Kearns that pinworms were common in high latitudes – places where people often are especially prone to MS.
“Most remarkably, they are known to have very little migration and stay spatially stable in populations over long periods of time,” Dr. Kearns said, “and typically everyone in an affected population will encounter them because their eggs are transmitted in household dust.”
And, he said, “they are known to be common in soldiers who live in military accommodation.”
According to the Centers for Disease Control and Prevention, pinworm prevalence can be as high as 50% in at-risk groups – children, caregivers of infected children, and people who live in institutions. Pinworms, which are spread through ingestion, are often asymptomatic but may cause anal itching and trigger bacterial infections.
The researchers suggest that pinworm infection comes first, followed by EBV infection. This makes sense because “late EBV infection in the form of infectious mononucleosis is known to be a risk factor for MS,” Dr. Kearns said.
The one-two punch of pinworm and then EBV is a plausible theory “because EBV lives in memory B cells, which are known to be important in MS and could be specific for the previous exposure to pinworm,” Dr. Kearns said. “However, this is very speculative and some researchers will argue this is very unlikely to be the case. But I think there is a chance it could explain some of the epidemiology, so I’m keen to try and test the theory if I can.”
What’s next? Dr. Kearns wants to explore data from Scotland in search of areas of high and low MS incidence that could offer insight into environmental triggers.
He added that the development of a serological blood test to prove a history of pinworm infection would be “the most effective way to prove or disprove this theory.”
“I have approached an investigator who has a track record of doing this for other infections and have been encouraged that he thinks that it would be achievable,” he said. “But this will definitely take time and funding.”
No specific funding was reported. The study authors reported no relevant disclosures.
SOURCE: Kearns P et al. ACTRIMS Forum 2018, Abstracts LB257 and LB264.
SAN DIEGO – What causes multiple sclerosis (MS)? A team of Scottish researchers offers a new theory that it’s triggered in part by a one-two punch of infection with pinworm – a common condition in the United States, especially among children – and the Epstein-Barr virus (EBV).
The theory identifies pinworm as the prime suspect to be the “missing link” that explains why EBV and MS are so tightly connected, said Patrick Kearns, MBChB, a graduate student at Harvard T.H. Chan School of Public Health in Boston.
Dr. Kearns is the lead author of two reports about the possible role of pinworm that were presented at ACTRIMS Forum 2018, which is held by the Americas Committee for Treatment and Research in Multiple Sclerosis. He spoke in an interview.
Dr. Kearns and his colleagues focused on a well-known cluster of MS cases that began to appear in the Faroe Islands – a Danish possession in the North Atlantic – during World War II. The cases began to appear after British troops occupied the islands.
“Many of the occupation soldiers were from the Scottish Highlands, where the MS prevalence is quite high: 90 cases per 100,000, comparable to the northern U.S.,” according to a National MS Society summary about MS clusters.
In a theory that spawned controversy, the late neurologist John Kurtzke, MD, speculated that the British soldiers brought a transmissible agent to the islands, which triggered MS cases.
Could the agent be EBV alone? The authors of the new studies don’t think so, although they note that EBV is “robustly linked” to MS. Indeed, a 2012 meta-analysis reported that the virus “appears to be present in 100% of MS patients,” based on studies considered to be the strongest (Mult Scler. 2013 Feb;19[2]:162-6).
The authors of the new reports note that, while EBV infection “appears necessary,” it is “clearly not sufficient” to cause the disease on its own.
“Certainly almost everyone gets EBV eventually,” Dr. Kearns said. “So mere presence of the virus is certainly not sufficient for causing the disease. But it seems still to be necessary, and timing of infection might be everything.”
So what’s the missing piece of the puzzle?
Dr. Kearns began to think it might be the lowly pinworm after helping a colleague by analyzing data from appendicitis samples in children. He noted that uninflamed samples often had pinworms in them, but the inflamed samples often didn’t, which suggested that “the rate of pinworms in normal appendices must be very high in the healthy pediatric population at any given time.”
More data confirmed this to be true, and medical literature told Dr. Kearns that pinworms were common in high latitudes – places where people often are especially prone to MS.
“Most remarkably, they are known to have very little migration and stay spatially stable in populations over long periods of time,” Dr. Kearns said, “and typically everyone in an affected population will encounter them because their eggs are transmitted in household dust.”
And, he said, “they are known to be common in soldiers who live in military accommodation.”
According to the Centers for Disease Control and Prevention, pinworm prevalence can be as high as 50% in at-risk groups – children, caregivers of infected children, and people who live in institutions. Pinworms, which are spread through ingestion, are often asymptomatic but may cause anal itching and trigger bacterial infections.
The researchers suggest that pinworm infection comes first, followed by EBV infection. This makes sense because “late EBV infection in the form of infectious mononucleosis is known to be a risk factor for MS,” Dr. Kearns said.
The one-two punch of pinworm and then EBV is a plausible theory “because EBV lives in memory B cells, which are known to be important in MS and could be specific for the previous exposure to pinworm,” Dr. Kearns said. “However, this is very speculative and some researchers will argue this is very unlikely to be the case. But I think there is a chance it could explain some of the epidemiology, so I’m keen to try and test the theory if I can.”
What’s next? Dr. Kearns wants to explore data from Scotland in search of areas of high and low MS incidence that could offer insight into environmental triggers.
He added that the development of a serological blood test to prove a history of pinworm infection would be “the most effective way to prove or disprove this theory.”
“I have approached an investigator who has a track record of doing this for other infections and have been encouraged that he thinks that it would be achievable,” he said. “But this will definitely take time and funding.”
No specific funding was reported. The study authors reported no relevant disclosures.
SOURCE: Kearns P et al. ACTRIMS Forum 2018, Abstracts LB257 and LB264.
SAN DIEGO – What causes multiple sclerosis (MS)? A team of Scottish researchers offers a new theory that it’s triggered in part by a one-two punch of infection with pinworm – a common condition in the United States, especially among children – and the Epstein-Barr virus (EBV).
The theory identifies pinworm as the prime suspect to be the “missing link” that explains why EBV and MS are so tightly connected, said Patrick Kearns, MBChB, a graduate student at Harvard T.H. Chan School of Public Health in Boston.
Dr. Kearns is the lead author of two reports about the possible role of pinworm that were presented at ACTRIMS Forum 2018, which is held by the Americas Committee for Treatment and Research in Multiple Sclerosis. He spoke in an interview.
Dr. Kearns and his colleagues focused on a well-known cluster of MS cases that began to appear in the Faroe Islands – a Danish possession in the North Atlantic – during World War II. The cases began to appear after British troops occupied the islands.
“Many of the occupation soldiers were from the Scottish Highlands, where the MS prevalence is quite high: 90 cases per 100,000, comparable to the northern U.S.,” according to a National MS Society summary about MS clusters.
In a theory that spawned controversy, the late neurologist John Kurtzke, MD, speculated that the British soldiers brought a transmissible agent to the islands, which triggered MS cases.
Could the agent be EBV alone? The authors of the new studies don’t think so, although they note that EBV is “robustly linked” to MS. Indeed, a 2012 meta-analysis reported that the virus “appears to be present in 100% of MS patients,” based on studies considered to be the strongest (Mult Scler. 2013 Feb;19[2]:162-6).
The authors of the new reports note that, while EBV infection “appears necessary,” it is “clearly not sufficient” to cause the disease on its own.
“Certainly almost everyone gets EBV eventually,” Dr. Kearns said. “So mere presence of the virus is certainly not sufficient for causing the disease. But it seems still to be necessary, and timing of infection might be everything.”
So what’s the missing piece of the puzzle?
Dr. Kearns began to think it might be the lowly pinworm after helping a colleague by analyzing data from appendicitis samples in children. He noted that uninflamed samples often had pinworms in them, but the inflamed samples often didn’t, which suggested that “the rate of pinworms in normal appendices must be very high in the healthy pediatric population at any given time.”
More data confirmed this to be true, and medical literature told Dr. Kearns that pinworms were common in high latitudes – places where people often are especially prone to MS.
“Most remarkably, they are known to have very little migration and stay spatially stable in populations over long periods of time,” Dr. Kearns said, “and typically everyone in an affected population will encounter them because their eggs are transmitted in household dust.”
And, he said, “they are known to be common in soldiers who live in military accommodation.”
According to the Centers for Disease Control and Prevention, pinworm prevalence can be as high as 50% in at-risk groups – children, caregivers of infected children, and people who live in institutions. Pinworms, which are spread through ingestion, are often asymptomatic but may cause anal itching and trigger bacterial infections.
The researchers suggest that pinworm infection comes first, followed by EBV infection. This makes sense because “late EBV infection in the form of infectious mononucleosis is known to be a risk factor for MS,” Dr. Kearns said.
The one-two punch of pinworm and then EBV is a plausible theory “because EBV lives in memory B cells, which are known to be important in MS and could be specific for the previous exposure to pinworm,” Dr. Kearns said. “However, this is very speculative and some researchers will argue this is very unlikely to be the case. But I think there is a chance it could explain some of the epidemiology, so I’m keen to try and test the theory if I can.”
What’s next? Dr. Kearns wants to explore data from Scotland in search of areas of high and low MS incidence that could offer insight into environmental triggers.
He added that the development of a serological blood test to prove a history of pinworm infection would be “the most effective way to prove or disprove this theory.”
“I have approached an investigator who has a track record of doing this for other infections and have been encouraged that he thinks that it would be achievable,” he said. “But this will definitely take time and funding.”
No specific funding was reported. The study authors reported no relevant disclosures.
SOURCE: Kearns P et al. ACTRIMS Forum 2018, Abstracts LB257 and LB264.
REPORTING FROM ACTRIMS FORUM 2018
Do Neurologists Adequately Screen Patients With MS for Cognitive Impairment and Depression?
SAN DIEGO—Current practice related to screening patients with multiple sclerosis (MS) for cognitive impairment and depression may need improvement, according to a study presented at the ACTRIMS 2018 Forum. Not all patients undergo screening for these comorbidities, and patients who undergo screening are not necessarily those most likely to have cognitive impairment or depression, said the researchers. In addition, the vast majority of clinicians who perform this screening do not use validated tools.
“Education on validated screening tools, development of more accessible tools, and the sharing of best practices by MS experts may all help address these barriers,” said Guy J. Buckle, MD, MPH, Director of Neuroimaging Research at Shepherd Center in Atlanta, and colleagues. “Assisting MS clinicians in cognitive and depression screening and early referral and treatment may all positively impact outcomes for patients with MS.”
Study Included Chart Review and Survey
The National MS Society reported that as much as half of patients with MS may have cognitive impairment. A meta-analysis by Foley et al suggested that 35% of patients with MS met the criteria for clinically significant depression. Yet not all patients with MS undergo screening for these comorbidities.
Dr. Buckle and colleagues examined neurologists’ current screening practices, perceptions about screening, and barriers to screening. The investigators reviewed charts for 300 patients with MS who were seen at two large specialty clinics (150 charts per clinic). Eligible patients were 18 or older and had two or more visits during the study period. The investigators also conducted informal email interviews with 13 MS specialists recognized as leaders in research and treatment.
Dr. Buckle’s group sought to determine the extent to which screening for cognitive impairment and depression are documented in practice, as well as the extent to which clinicians use validated tools for this screening. In addition, the researchers solicited neurologists’ perceptions of the clinical value of formal screening, along with barriers to using screening tools.
Rate of Screening May Have Been Suboptimal
Participants’ median age was 52, and 76% of participants were female. Average time since diagnosis was 13 years. Fifteen patients had had one or more relapse in the previous 24 months.
Screening for cognitive impairment was documented for 52% of patients in the previous 12 months, and cognitive impairment was documented in 37% of those screened. About 2% of clinicians used a validated tool for screening, and 28% of patients were referred for more specialized care.
Screening for depression was documented for 63% of patients in the previous 12 months, and depression was documented in 42% of those screened. About 4% of clinicians used a validated tool for screening, and 25% of patients were referred for specialized care.
About 48% of patients younger than 65 were screened for cognitive impairment, compared with 73% of patients age 65 or older. Cognitive impairment was diagnosed in 69% of patients younger than 65 and in 78% of patients 65 or older. Patients younger than 65 were less likely to be screened for depression than those 65 or older (60% vs 73%), but more likely to be diagnosed with depression (71% vs 54%).
White patients were more likely to be screened for cognitive impairment than black patients (57% vs 52%), but less likely to be diagnosed with cognitive impairment (68% vs 82%). Similarly, white patients were more likely to be screened for depression (71% vs 53%), but less likely to be diagnosed with depression (65% vs 75%).
Patients who were employed were more likely to undergo cognitive screening than unemployed, retired, or disabled patients, but the latter were more likely to be diagnosed with cognitive impairment. “The analyses support previous findings suggesting links between cognitive dysfunction, patient age, and physical disability,” said Dr. Buckle and colleagues.
Specialists Cited Time as a Barrier
About 54% of the MS specialists surveyed reported that they used validated tools to assess cognitive impairment. Among the tools they named were the Symbol Digit Modalities Test, the Mini-Mental State Examination, the California Verbal Learning Test II, and the Brief International Cognitive Assessment for MS. Among the reasons the specialists gave for not using validated tools were time constraints, lack of qualified staff for administration, lack of integration of the tests into electronic medical records, and lack of reimbursement.
Approximately 46% of the MS specialists reported that they used validated tools to assess depression. The tools that they reported using included the Patient Health Questionnaire-9, the Beck Depression Inventory-II, CNS Vital Signs, and routine questions about appetite, weight loss, sleep, sexual activity, and suicidal ideation. The specialists cited time constraints, lack of compensation, cost, and inability to document results in the electronic medical record as reasons for not using validated tools.
“Integration of formal tools in clinical practice may support clinicians in appropriate and consistent identification of patient populations with these conditions,” said the investigators.
Study May Have Limited Generalizability
The study results may not accurately reflect current practice throughout the country. “The small sample size and relatively low proportions of racial minorities may limit the ability to generalize these results and necessitate large-scale studies,” said Dr. Buckle and colleagues. “Future research should examine healthcare disparities in MS and underlying contributors (eg, bias and perceptions) that hinder the use of formal screening tools in MS patients.”
—Erik Greb
Suggested Reading
Benedict RH, Cox D, Thompson LL, et al. Reliable screening for neuropsychological impairment in multiple sclerosis. Mult
Parmenter BA, Weinstock-Guttman B, Garg N, et al. Screening for cognitive impairment in multiple sclerosis using the Symbol digit Modalities Test. Mult Scler. 2007;13(1):52-57.
SAN DIEGO—Current practice related to screening patients with multiple sclerosis (MS) for cognitive impairment and depression may need improvement, according to a study presented at the ACTRIMS 2018 Forum. Not all patients undergo screening for these comorbidities, and patients who undergo screening are not necessarily those most likely to have cognitive impairment or depression, said the researchers. In addition, the vast majority of clinicians who perform this screening do not use validated tools.
“Education on validated screening tools, development of more accessible tools, and the sharing of best practices by MS experts may all help address these barriers,” said Guy J. Buckle, MD, MPH, Director of Neuroimaging Research at Shepherd Center in Atlanta, and colleagues. “Assisting MS clinicians in cognitive and depression screening and early referral and treatment may all positively impact outcomes for patients with MS.”
Study Included Chart Review and Survey
The National MS Society reported that as much as half of patients with MS may have cognitive impairment. A meta-analysis by Foley et al suggested that 35% of patients with MS met the criteria for clinically significant depression. Yet not all patients with MS undergo screening for these comorbidities.
Dr. Buckle and colleagues examined neurologists’ current screening practices, perceptions about screening, and barriers to screening. The investigators reviewed charts for 300 patients with MS who were seen at two large specialty clinics (150 charts per clinic). Eligible patients were 18 or older and had two or more visits during the study period. The investigators also conducted informal email interviews with 13 MS specialists recognized as leaders in research and treatment.
Dr. Buckle’s group sought to determine the extent to which screening for cognitive impairment and depression are documented in practice, as well as the extent to which clinicians use validated tools for this screening. In addition, the researchers solicited neurologists’ perceptions of the clinical value of formal screening, along with barriers to using screening tools.
Rate of Screening May Have Been Suboptimal
Participants’ median age was 52, and 76% of participants were female. Average time since diagnosis was 13 years. Fifteen patients had had one or more relapse in the previous 24 months.
Screening for cognitive impairment was documented for 52% of patients in the previous 12 months, and cognitive impairment was documented in 37% of those screened. About 2% of clinicians used a validated tool for screening, and 28% of patients were referred for more specialized care.
Screening for depression was documented for 63% of patients in the previous 12 months, and depression was documented in 42% of those screened. About 4% of clinicians used a validated tool for screening, and 25% of patients were referred for specialized care.
About 48% of patients younger than 65 were screened for cognitive impairment, compared with 73% of patients age 65 or older. Cognitive impairment was diagnosed in 69% of patients younger than 65 and in 78% of patients 65 or older. Patients younger than 65 were less likely to be screened for depression than those 65 or older (60% vs 73%), but more likely to be diagnosed with depression (71% vs 54%).
White patients were more likely to be screened for cognitive impairment than black patients (57% vs 52%), but less likely to be diagnosed with cognitive impairment (68% vs 82%). Similarly, white patients were more likely to be screened for depression (71% vs 53%), but less likely to be diagnosed with depression (65% vs 75%).
Patients who were employed were more likely to undergo cognitive screening than unemployed, retired, or disabled patients, but the latter were more likely to be diagnosed with cognitive impairment. “The analyses support previous findings suggesting links between cognitive dysfunction, patient age, and physical disability,” said Dr. Buckle and colleagues.
Specialists Cited Time as a Barrier
About 54% of the MS specialists surveyed reported that they used validated tools to assess cognitive impairment. Among the tools they named were the Symbol Digit Modalities Test, the Mini-Mental State Examination, the California Verbal Learning Test II, and the Brief International Cognitive Assessment for MS. Among the reasons the specialists gave for not using validated tools were time constraints, lack of qualified staff for administration, lack of integration of the tests into electronic medical records, and lack of reimbursement.
Approximately 46% of the MS specialists reported that they used validated tools to assess depression. The tools that they reported using included the Patient Health Questionnaire-9, the Beck Depression Inventory-II, CNS Vital Signs, and routine questions about appetite, weight loss, sleep, sexual activity, and suicidal ideation. The specialists cited time constraints, lack of compensation, cost, and inability to document results in the electronic medical record as reasons for not using validated tools.
“Integration of formal tools in clinical practice may support clinicians in appropriate and consistent identification of patient populations with these conditions,” said the investigators.
Study May Have Limited Generalizability
The study results may not accurately reflect current practice throughout the country. “The small sample size and relatively low proportions of racial minorities may limit the ability to generalize these results and necessitate large-scale studies,” said Dr. Buckle and colleagues. “Future research should examine healthcare disparities in MS and underlying contributors (eg, bias and perceptions) that hinder the use of formal screening tools in MS patients.”
—Erik Greb
Suggested Reading
Benedict RH, Cox D, Thompson LL, et al. Reliable screening for neuropsychological impairment in multiple sclerosis. Mult
Parmenter BA, Weinstock-Guttman B, Garg N, et al. Screening for cognitive impairment in multiple sclerosis using the Symbol digit Modalities Test. Mult Scler. 2007;13(1):52-57.
SAN DIEGO—Current practice related to screening patients with multiple sclerosis (MS) for cognitive impairment and depression may need improvement, according to a study presented at the ACTRIMS 2018 Forum. Not all patients undergo screening for these comorbidities, and patients who undergo screening are not necessarily those most likely to have cognitive impairment or depression, said the researchers. In addition, the vast majority of clinicians who perform this screening do not use validated tools.
“Education on validated screening tools, development of more accessible tools, and the sharing of best practices by MS experts may all help address these barriers,” said Guy J. Buckle, MD, MPH, Director of Neuroimaging Research at Shepherd Center in Atlanta, and colleagues. “Assisting MS clinicians in cognitive and depression screening and early referral and treatment may all positively impact outcomes for patients with MS.”
Study Included Chart Review and Survey
The National MS Society reported that as much as half of patients with MS may have cognitive impairment. A meta-analysis by Foley et al suggested that 35% of patients with MS met the criteria for clinically significant depression. Yet not all patients with MS undergo screening for these comorbidities.
Dr. Buckle and colleagues examined neurologists’ current screening practices, perceptions about screening, and barriers to screening. The investigators reviewed charts for 300 patients with MS who were seen at two large specialty clinics (150 charts per clinic). Eligible patients were 18 or older and had two or more visits during the study period. The investigators also conducted informal email interviews with 13 MS specialists recognized as leaders in research and treatment.
Dr. Buckle’s group sought to determine the extent to which screening for cognitive impairment and depression are documented in practice, as well as the extent to which clinicians use validated tools for this screening. In addition, the researchers solicited neurologists’ perceptions of the clinical value of formal screening, along with barriers to using screening tools.
Rate of Screening May Have Been Suboptimal
Participants’ median age was 52, and 76% of participants were female. Average time since diagnosis was 13 years. Fifteen patients had had one or more relapse in the previous 24 months.
Screening for cognitive impairment was documented for 52% of patients in the previous 12 months, and cognitive impairment was documented in 37% of those screened. About 2% of clinicians used a validated tool for screening, and 28% of patients were referred for more specialized care.
Screening for depression was documented for 63% of patients in the previous 12 months, and depression was documented in 42% of those screened. About 4% of clinicians used a validated tool for screening, and 25% of patients were referred for specialized care.
About 48% of patients younger than 65 were screened for cognitive impairment, compared with 73% of patients age 65 or older. Cognitive impairment was diagnosed in 69% of patients younger than 65 and in 78% of patients 65 or older. Patients younger than 65 were less likely to be screened for depression than those 65 or older (60% vs 73%), but more likely to be diagnosed with depression (71% vs 54%).
White patients were more likely to be screened for cognitive impairment than black patients (57% vs 52%), but less likely to be diagnosed with cognitive impairment (68% vs 82%). Similarly, white patients were more likely to be screened for depression (71% vs 53%), but less likely to be diagnosed with depression (65% vs 75%).
Patients who were employed were more likely to undergo cognitive screening than unemployed, retired, or disabled patients, but the latter were more likely to be diagnosed with cognitive impairment. “The analyses support previous findings suggesting links between cognitive dysfunction, patient age, and physical disability,” said Dr. Buckle and colleagues.
Specialists Cited Time as a Barrier
About 54% of the MS specialists surveyed reported that they used validated tools to assess cognitive impairment. Among the tools they named were the Symbol Digit Modalities Test, the Mini-Mental State Examination, the California Verbal Learning Test II, and the Brief International Cognitive Assessment for MS. Among the reasons the specialists gave for not using validated tools were time constraints, lack of qualified staff for administration, lack of integration of the tests into electronic medical records, and lack of reimbursement.
Approximately 46% of the MS specialists reported that they used validated tools to assess depression. The tools that they reported using included the Patient Health Questionnaire-9, the Beck Depression Inventory-II, CNS Vital Signs, and routine questions about appetite, weight loss, sleep, sexual activity, and suicidal ideation. The specialists cited time constraints, lack of compensation, cost, and inability to document results in the electronic medical record as reasons for not using validated tools.
“Integration of formal tools in clinical practice may support clinicians in appropriate and consistent identification of patient populations with these conditions,” said the investigators.
Study May Have Limited Generalizability
The study results may not accurately reflect current practice throughout the country. “The small sample size and relatively low proportions of racial minorities may limit the ability to generalize these results and necessitate large-scale studies,” said Dr. Buckle and colleagues. “Future research should examine healthcare disparities in MS and underlying contributors (eg, bias and perceptions) that hinder the use of formal screening tools in MS patients.”
—Erik Greb
Suggested Reading
Benedict RH, Cox D, Thompson LL, et al. Reliable screening for neuropsychological impairment in multiple sclerosis. Mult
Parmenter BA, Weinstock-Guttman B, Garg N, et al. Screening for cognitive impairment in multiple sclerosis using the Symbol digit Modalities Test. Mult Scler. 2007;13(1):52-57.