User login
MS: Past, Present, and Future
Stuart D. Cook, MD, and Abdul Rahman Alchaki
Dr. Cook is the Ruth Dunietz Kushner and Michael Jay Serwitz Professor of Neurology/Neurosciences at Rutgers, the State University of New Jersey, Newark. Dr. Alchaki is a resident in the Deptartment of Neurology/Neurosciences at Rutgers, the State University of New Jersey, Newark.
Disclosure: Stuart Cook has received honoraria for lectures from Bayer HealthCare and Merck Serono. He has served as a consultant for Merck Serono, Bayer HealthCare, Teva, Novartis, Sanofi-Aventis, Biogen Idec, and Actinobac Biomed. He has served on steering committees for the BEYOND and CLARITY Studies and as a member of Advisory Boards for Merck Serono, Bayer HealthCare, Teva, Biogen Idec, Sanofi Aventis, and Actinobac Biomed.
The Initial Years (1838 to 1930s)
The earliest recognition of MS clinical features and pathology was attributed to Jean-Martin Charcot, Robert Carswell, and Jean Cruveilhier in Europe from 1838 to 1868. Beyond those early descriptions, relatively few MS breakthroughs occurred until the 1930s, when Thomas Rivers discovered experimental autoimmune encephalomyelitis (EAE), a demyelinating disease, in animals. His insightful concepts were widely cited and ultimately contributed to undestanding of the immune mechanisms of MS and acute disseminated encephalomyelitis (ADEM).
Advances in Diagnosis (1965 to 1992)
In 1965, Schumacher et al provided the essential clinical criteria for MS diagnosis. Poser et al refined these criteria in 1983. In 2001, McDonald et al added neuroimaging, CSF analysis, and evoked potentials to further complement MS clinical diagnosis. For the first time, the disease could generally be recognized.
Early Treatments
Various treatments for MS were tried over the years, without great success. However, in 1953, a small descriptive trial by Miller and Gibbons reported clinical benefits in patients using intramuscular (IM) adrenocorticotropic hormone (ACTH) for MS and disseminated encephalomyelitis. This was followed in 1970 by a Cooperative Study of IM ACTH versus placebo by Rose et al, which resulted in ACTH, and subsequently oral corticosteroids, being widely used to treat MS, particularly for acute exacerbations of the disease. However, robust evidence of long-term steroids remain limited, even to the present.
High-Dose Steroids
By 1980, the initial descriptive treatment of high-dose intravenous (IV) steroids for demyelinating diseases, including MS and transverse myelitis, by Dowling et al resulted in rapid clinical improvement in some patients. This result was ultimately confirmed by others. High-dose IV steroids became the gold standard for acute attacks, particularly those aggressive in nature. In the mid 1980s, work by Troiano et al, as well as others, showed that the rapid use of high-dose IV as well as oral steroids showed similar effects, with reduction or elimination of CT contrast-enhancing lesions within as few as eight hours, while lower doses or alternative-day treatments were less effective. In addition, descriptive studies of immune modulatory and immunosuppressive drugs, as well as small randomized studies, were published. These agents did not receive FDA approval.
The Golden Age of Therapy (1993 to 2018)
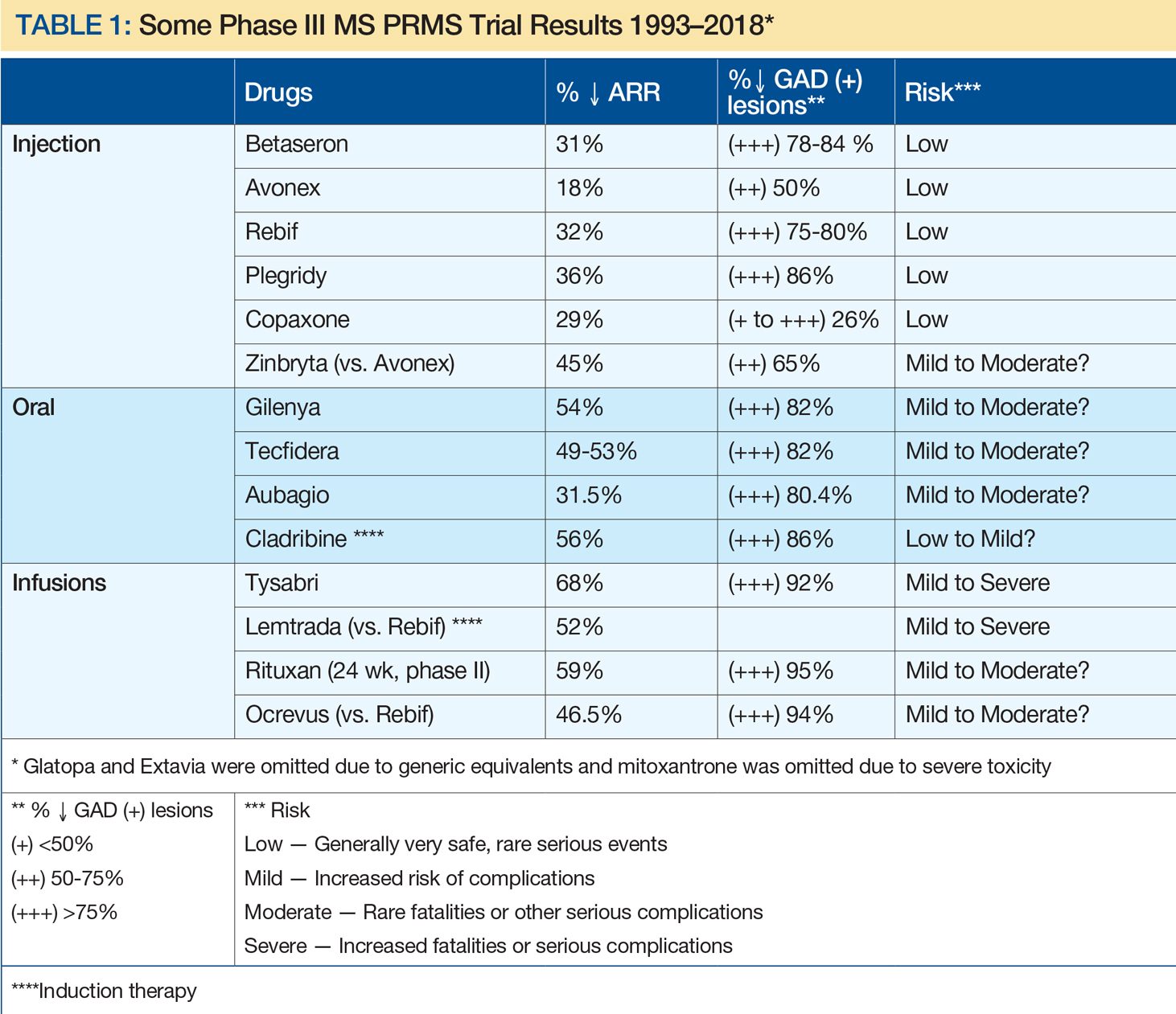
A remarkable era in MS prognosis and treatment began with immunomodulation injections of Betaseron (INFβ-1b), Avonex (INFβ-1a), and Copaxone (glatiramer acetate). This can be attributed, at least in part, to advances in molecular biology, genetics, and neuroimaging, and support by corporate, private, and public funding. Since the initial FDA approval of INFβ-1b, 15 MS therapies have become clinically available, including eight injectables, three orals, and four infusion treatments (see Table 1). In addition, two other drugs have been FDA approved for uses other than MS: rituximab (approved for lymphoma) and cladribine (for hairy cell leukemia), with the latter now approved by the European Medicines Agency for MS. Table 1 depicts characteristics of these therapies approved by US or European agencies (or for other disorders increasingly used off label for MS) in an attempt to compare annual relapse rates (ARR) and decreases in the percent of gadolinium-enhancing MS lesions versus placebo. This information was chosen because ARR has been uniformly selected and defined for such trials, while percent decrease of gadolinium-enhancing lesions on MRI has been the most sensitive barometer available for assessing acute clinical activity. As a result, risk-benefit considerations have been critical in evaluating these drug treatments, with efficacy improving greatly over time, whereas risks have been more variable.
Disease Categories
In 1996, Lublin and Reingold provided a new classification, not specifically for the diagnosis of MS, but rather for the clinical course of the disease. Initially, there were four categories—relapsing-remitting MS, secondary progressive MS, primary progressive MS, and progressive-relapsing MS—that were universally identified. These were thought to be relatively distinct clinical categories, but over time it became clear that the classification did not fully distinguish MS disease activity within these categories. For that reason, it was subsequently recommended, by Lincoln et al in 2009 and Cook et al in 2012, to include MRI, a vastly more sensitive modality, as well as clinical data in assessing disease activity.
On another note, MS and neuromyelitis optica (NMO), although having similar features, were clearly identified as different diseases by Lennon et al in 2004. Differences in pathology, clinical characteristics, immunology, and therapy separate the two disorders.
MRI in MS
Work by Young et al in 1981 established the central role of MRI brain imaging in MS diagnosis and therapeutic considerations. Since then it has become ubiquitous.
An example of a sensitive and highly productive MRI protocol is the BECOME study of MS and clinically isolated syndrome by Cadavid et al from 2009 to 2017. In this study, IFNβ-1b was compared with glatiramer acetate treatment. Cadavid et al used a 3T scanner with triple-dose gadolinium, performed monthly for as long as 24 consecutive months. This unique study brought about a virtual gold mine of valuable research and clinical information. This included proof that gadolinium-enhancing lesions persisted for six months or more, evidence of a 30:1 ratio of new MRI brain lesions to clinical activity, and documentation that 96% of T2 lesions and black holes derive from prior gadolinium-enhancing lesions. It was further noted that 80% to 90% of acute black holes disappeared with treatment and 75% to 80% of patients taking IFNβ-1b or glatiramer acetate had new MRI lesions despite continuing treatment. Perhaps most interestingly, monthly MRIs could predict relapse and disability in a relatively small number of patients, depending upon the frequency and activity of MRI lesions. In 2017, Brown et al documented that magnetization transfer ratio recovery in MS brain lesions occurred more significantly with glatiramer acetate than with IFNβ-1b, whereas more chronic black hole lesions were found with glatiramer acetate. Also in 2017, Maranzano et al found evidence of acute inflammatory leukocortical lesions, which were not as well recognized previously.
In summary, it has become increasingly clear that MRI is the most sen
The Future of MS
While it is not yet a curable disease, there is growing evidence that MS prognosis has improved and will continue to improve. This is based on incremental decreases in acute MS exacerbations, progressive disability, and MRI lesion activity, as well as a combination of the three—no evidence of disease activity (NEDA).
Not only are drug therapies becoming more effective, but patients and physicians now have many more treatment options to carefully consider with regard to efficacy, side effect profiles, treatment frequency, route of administration, cost, and quality of life. Newer drugs with different mechanisms of action such as cladribine, now approved in Europe, fulfill most of these beneficial criteria (see Giovannoni et al, 2010). More promising MS treatments, including long-acting induction therapies, are still being evaluated. As with other complex diseases, multiple therapies are likely to be used as well.
In summary, compared with the time before 1993, MS will be much less likely to be a progressive disease, and quality of life will be much improved. In my opinion, patients will be less fearful about their prognosis than ever before, and with appropriate evaluations and treatments, we may realize that disabling MS will be far less common.
Suggested Reading
Brown JW, Pardini M, Brownlee WJ, et al. An abnormal periventricular magnetization transfer ratio gradient occurs early in multiple sclerosis. Brain. 2017;140(2):387-398.
Cadavid D, Wolansky LJ, Skurnick J, et al. Efficacy of treatment of MS with IFNbeta-1b or glatiramer acetate by monthly brain MRI in the BECOME study. Neurology. 2009;72(23):1976-1983.
Cook SD, Dhib-Jalbut S, Dowling P, et al. Use of magnetic resonance imaging as well as clinical disease activity in the clinical classification of multiple sclerosis and assessment of its course: a report from an international CMSC consensus conference, March 5-7, 2010. Int J MS Care. 2012;14(3):105-114.
Dowling PC, Bosch VV, Cook SD. Possible beneficial effect of high-dose intravenous steroid therapy in acute demyelinating disease and transverse myelitis. Neurology. 1980;30(7 Pt 2):33-36.
Giovannoni G, Comi G, Cook S, et al. A placebo-controlled trial of oral cladribine for relapsing multiple sclerosis. N Engl J Med. 2010;362(5):416-426.
Lennon VA, Wingerchuk DM, Kryzer TJ, et al. A serum autoantibody marker of neuromyelitis optica: distinction from multiple sclerosis. Lancet. 2004;364(9451):2106-2112.
Lincoln JA, Cadavid D, Pollard J, et al. We should use magnetic resonance imaging to classify and monitor the course of multiple sclerosis. Arch Neurol. 2009;66(3):412-414.
Lublin FD, Reingold SC. Defining the clinical course of multiple sclerosis: results of an international survey. National Multiple Sclerosis Society (USA) Advisory Committee on Clinical Trials of New Agents in Multiple Sclerosis. Neurology. 1996;46(4):907-911.
Maranzano J, Rudko DA, Nakamura K, et al. MRI evidence of acute inflammation in leukocortical lesions of patients with early multiple sclerosis. Neurology. 2017;89(7):714-721.
McDonald WI, Compston A, Edan G, et al. Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol. 2001;50(1):121-127.
Miller HG, Gibbons JL. Acute disseminated encephalomyelitis and acute disseminated sclerosis; results of treatment with A.C.T.H. Br Med J. 1953;2(4850):1345-1348.
Poser CM, Paty DW, Scheinberg L, et al. New diagnostic criteria for multiple sclerosis: guidelines for research protocols. Ann Neurol. 1983;13(3):227-231.
Rose AS, Kuzma JW, Kurtzke JF, et al. Cooperative study in the evaluation of therapy in multiple sclerosis. ACTH vs. placebo--final report. Neurology. 1970;20(5):1-59.
Troiano R, Hafstein M, Ruderman M, et al. Effect of high-dose intravenous steroid administration on contrast-enhancing computed tomographic scan lesions in multiple sclerosis. Ann Neurol. 1984;15(3):257-263.
Troiano RA, Hafstein MP, Zito G, et al. The effect of oral corticosteroid dosage on CT enhancing multiple sclerosis plaques. J Neurol Sci. 1985;70(1):67-72.
Young IR, Hall AS, Pallis A, et al. Nuclear magnetic resonance imaging of the brain in multiple sclerosis. Lancet. 1981;2(8255):1063-1066.
Stuart D. Cook, MD, and Abdul Rahman Alchaki
Dr. Cook is the Ruth Dunietz Kushner and Michael Jay Serwitz Professor of Neurology/Neurosciences at Rutgers, the State University of New Jersey, Newark. Dr. Alchaki is a resident in the Deptartment of Neurology/Neurosciences at Rutgers, the State University of New Jersey, Newark.
Disclosure: Stuart Cook has received honoraria for lectures from Bayer HealthCare and Merck Serono. He has served as a consultant for Merck Serono, Bayer HealthCare, Teva, Novartis, Sanofi-Aventis, Biogen Idec, and Actinobac Biomed. He has served on steering committees for the BEYOND and CLARITY Studies and as a member of Advisory Boards for Merck Serono, Bayer HealthCare, Teva, Biogen Idec, Sanofi Aventis, and Actinobac Biomed.
The Initial Years (1838 to 1930s)
The earliest recognition of MS clinical features and pathology was attributed to Jean-Martin Charcot, Robert Carswell, and Jean Cruveilhier in Europe from 1838 to 1868. Beyond those early descriptions, relatively few MS breakthroughs occurred until the 1930s, when Thomas Rivers discovered experimental autoimmune encephalomyelitis (EAE), a demyelinating disease, in animals. His insightful concepts were widely cited and ultimately contributed to undestanding of the immune mechanisms of MS and acute disseminated encephalomyelitis (ADEM).
Advances in Diagnosis (1965 to 1992)
In 1965, Schumacher et al provided the essential clinical criteria for MS diagnosis. Poser et al refined these criteria in 1983. In 2001, McDonald et al added neuroimaging, CSF analysis, and evoked potentials to further complement MS clinical diagnosis. For the first time, the disease could generally be recognized.
Early Treatments
Various treatments for MS were tried over the years, without great success. However, in 1953, a small descriptive trial by Miller and Gibbons reported clinical benefits in patients using intramuscular (IM) adrenocorticotropic hormone (ACTH) for MS and disseminated encephalomyelitis. This was followed in 1970 by a Cooperative Study of IM ACTH versus placebo by Rose et al, which resulted in ACTH, and subsequently oral corticosteroids, being widely used to treat MS, particularly for acute exacerbations of the disease. However, robust evidence of long-term steroids remain limited, even to the present.
High-Dose Steroids
By 1980, the initial descriptive treatment of high-dose intravenous (IV) steroids for demyelinating diseases, including MS and transverse myelitis, by Dowling et al resulted in rapid clinical improvement in some patients. This result was ultimately confirmed by others. High-dose IV steroids became the gold standard for acute attacks, particularly those aggressive in nature. In the mid 1980s, work by Troiano et al, as well as others, showed that the rapid use of high-dose IV as well as oral steroids showed similar effects, with reduction or elimination of CT contrast-enhancing lesions within as few as eight hours, while lower doses or alternative-day treatments were less effective. In addition, descriptive studies of immune modulatory and immunosuppressive drugs, as well as small randomized studies, were published. These agents did not receive FDA approval.
The Golden Age of Therapy (1993 to 2018)
A remarkable era in MS prognosis and treatment began with immunomodulation injections of Betaseron (INFβ-1b), Avonex (INFβ-1a), and Copaxone (glatiramer acetate). This can be attributed, at least in part, to advances in molecular biology, genetics, and neuroimaging, and support by corporate, private, and public funding. Since the initial FDA approval of INFβ-1b, 15 MS therapies have become clinically available, including eight injectables, three orals, and four infusion treatments (see Table 1). In addition, two other drugs have been FDA approved for uses other than MS: rituximab (approved for lymphoma) and cladribine (for hairy cell leukemia), with the latter now approved by the European Medicines Agency for MS. Table 1 depicts characteristics of these therapies approved by US or European agencies (or for other disorders increasingly used off label for MS) in an attempt to compare annual relapse rates (ARR) and decreases in the percent of gadolinium-enhancing MS lesions versus placebo. This information was chosen because ARR has been uniformly selected and defined for such trials, while percent decrease of gadolinium-enhancing lesions on MRI has been the most sensitive barometer available for assessing acute clinical activity. As a result, risk-benefit considerations have been critical in evaluating these drug treatments, with efficacy improving greatly over time, whereas risks have been more variable.
Disease Categories
In 1996, Lublin and Reingold provided a new classification, not specifically for the diagnosis of MS, but rather for the clinical course of the disease. Initially, there were four categories—relapsing-remitting MS, secondary progressive MS, primary progressive MS, and progressive-relapsing MS—that were universally identified. These were thought to be relatively distinct clinical categories, but over time it became clear that the classification did not fully distinguish MS disease activity within these categories. For that reason, it was subsequently recommended, by Lincoln et al in 2009 and Cook et al in 2012, to include MRI, a vastly more sensitive modality, as well as clinical data in assessing disease activity.
On another note, MS and neuromyelitis optica (NMO), although having similar features, were clearly identified as different diseases by Lennon et al in 2004. Differences in pathology, clinical characteristics, immunology, and therapy separate the two disorders.
MRI in MS
Work by Young et al in 1981 established the central role of MRI brain imaging in MS diagnosis and therapeutic considerations. Since then it has become ubiquitous.
An example of a sensitive and highly productive MRI protocol is the BECOME study of MS and clinically isolated syndrome by Cadavid et al from 2009 to 2017. In this study, IFNβ-1b was compared with glatiramer acetate treatment. Cadavid et al used a 3T scanner with triple-dose gadolinium, performed monthly for as long as 24 consecutive months. This unique study brought about a virtual gold mine of valuable research and clinical information. This included proof that gadolinium-enhancing lesions persisted for six months or more, evidence of a 30:1 ratio of new MRI brain lesions to clinical activity, and documentation that 96% of T2 lesions and black holes derive from prior gadolinium-enhancing lesions. It was further noted that 80% to 90% of acute black holes disappeared with treatment and 75% to 80% of patients taking IFNβ-1b or glatiramer acetate had new MRI lesions despite continuing treatment. Perhaps most interestingly, monthly MRIs could predict relapse and disability in a relatively small number of patients, depending upon the frequency and activity of MRI lesions. In 2017, Brown et al documented that magnetization transfer ratio recovery in MS brain lesions occurred more significantly with glatiramer acetate than with IFNβ-1b, whereas more chronic black hole lesions were found with glatiramer acetate. Also in 2017, Maranzano et al found evidence of acute inflammatory leukocortical lesions, which were not as well recognized previously.
In summary, it has become increasingly clear that MRI is the most sen
The Future of MS
While it is not yet a curable disease, there is growing evidence that MS prognosis has improved and will continue to improve. This is based on incremental decreases in acute MS exacerbations, progressive disability, and MRI lesion activity, as well as a combination of the three—no evidence of disease activity (NEDA).
Not only are drug therapies becoming more effective, but patients and physicians now have many more treatment options to carefully consider with regard to efficacy, side effect profiles, treatment frequency, route of administration, cost, and quality of life. Newer drugs with different mechanisms of action such as cladribine, now approved in Europe, fulfill most of these beneficial criteria (see Giovannoni et al, 2010). More promising MS treatments, including long-acting induction therapies, are still being evaluated. As with other complex diseases, multiple therapies are likely to be used as well.
In summary, compared with the time before 1993, MS will be much less likely to be a progressive disease, and quality of life will be much improved. In my opinion, patients will be less fearful about their prognosis than ever before, and with appropriate evaluations and treatments, we may realize that disabling MS will be far less common.

Suggested Reading
Brown JW, Pardini M, Brownlee WJ, et al. An abnormal periventricular magnetization transfer ratio gradient occurs early in multiple sclerosis. Brain. 2017;140(2):387-398.
Cadavid D, Wolansky LJ, Skurnick J, et al. Efficacy of treatment of MS with IFNbeta-1b or glatiramer acetate by monthly brain MRI in the BECOME study. Neurology. 2009;72(23):1976-1983.
Cook SD, Dhib-Jalbut S, Dowling P, et al. Use of magnetic resonance imaging as well as clinical disease activity in the clinical classification of multiple sclerosis and assessment of its course: a report from an international CMSC consensus conference, March 5-7, 2010. Int J MS Care. 2012;14(3):105-114.
Dowling PC, Bosch VV, Cook SD. Possible beneficial effect of high-dose intravenous steroid therapy in acute demyelinating disease and transverse myelitis. Neurology. 1980;30(7 Pt 2):33-36.
Giovannoni G, Comi G, Cook S, et al. A placebo-controlled trial of oral cladribine for relapsing multiple sclerosis. N Engl J Med. 2010;362(5):416-426.
Lennon VA, Wingerchuk DM, Kryzer TJ, et al. A serum autoantibody marker of neuromyelitis optica: distinction from multiple sclerosis. Lancet. 2004;364(9451):2106-2112.
Lincoln JA, Cadavid D, Pollard J, et al. We should use magnetic resonance imaging to classify and monitor the course of multiple sclerosis. Arch Neurol. 2009;66(3):412-414.
Lublin FD, Reingold SC. Defining the clinical course of multiple sclerosis: results of an international survey. National Multiple Sclerosis Society (USA) Advisory Committee on Clinical Trials of New Agents in Multiple Sclerosis. Neurology. 1996;46(4):907-911.
Maranzano J, Rudko DA, Nakamura K, et al. MRI evidence of acute inflammation in leukocortical lesions of patients with early multiple sclerosis. Neurology. 2017;89(7):714-721.
McDonald WI, Compston A, Edan G, et al. Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol. 2001;50(1):121-127.
Miller HG, Gibbons JL. Acute disseminated encephalomyelitis and acute disseminated sclerosis; results of treatment with A.C.T.H. Br Med J. 1953;2(4850):1345-1348.
Poser CM, Paty DW, Scheinberg L, et al. New diagnostic criteria for multiple sclerosis: guidelines for research protocols. Ann Neurol. 1983;13(3):227-231.
Rose AS, Kuzma JW, Kurtzke JF, et al. Cooperative study in the evaluation of therapy in multiple sclerosis. ACTH vs. placebo--final report. Neurology. 1970;20(5):1-59.
Troiano R, Hafstein M, Ruderman M, et al. Effect of high-dose intravenous steroid administration on contrast-enhancing computed tomographic scan lesions in multiple sclerosis. Ann Neurol. 1984;15(3):257-263.
Troiano RA, Hafstein MP, Zito G, et al. The effect of oral corticosteroid dosage on CT enhancing multiple sclerosis plaques. J Neurol Sci. 1985;70(1):67-72.
Young IR, Hall AS, Pallis A, et al. Nuclear magnetic resonance imaging of the brain in multiple sclerosis. Lancet. 1981;2(8255):1063-1066.
Stuart D. Cook, MD, and Abdul Rahman Alchaki
Dr. Cook is the Ruth Dunietz Kushner and Michael Jay Serwitz Professor of Neurology/Neurosciences at Rutgers, the State University of New Jersey, Newark. Dr. Alchaki is a resident in the Deptartment of Neurology/Neurosciences at Rutgers, the State University of New Jersey, Newark.
Disclosure: Stuart Cook has received honoraria for lectures from Bayer HealthCare and Merck Serono. He has served as a consultant for Merck Serono, Bayer HealthCare, Teva, Novartis, Sanofi-Aventis, Biogen Idec, and Actinobac Biomed. He has served on steering committees for the BEYOND and CLARITY Studies and as a member of Advisory Boards for Merck Serono, Bayer HealthCare, Teva, Biogen Idec, Sanofi Aventis, and Actinobac Biomed.
The Initial Years (1838 to 1930s)
The earliest recognition of MS clinical features and pathology was attributed to Jean-Martin Charcot, Robert Carswell, and Jean Cruveilhier in Europe from 1838 to 1868. Beyond those early descriptions, relatively few MS breakthroughs occurred until the 1930s, when Thomas Rivers discovered experimental autoimmune encephalomyelitis (EAE), a demyelinating disease, in animals. His insightful concepts were widely cited and ultimately contributed to undestanding of the immune mechanisms of MS and acute disseminated encephalomyelitis (ADEM).
Advances in Diagnosis (1965 to 1992)
In 1965, Schumacher et al provided the essential clinical criteria for MS diagnosis. Poser et al refined these criteria in 1983. In 2001, McDonald et al added neuroimaging, CSF analysis, and evoked potentials to further complement MS clinical diagnosis. For the first time, the disease could generally be recognized.
Early Treatments
Various treatments for MS were tried over the years, without great success. However, in 1953, a small descriptive trial by Miller and Gibbons reported clinical benefits in patients using intramuscular (IM) adrenocorticotropic hormone (ACTH) for MS and disseminated encephalomyelitis. This was followed in 1970 by a Cooperative Study of IM ACTH versus placebo by Rose et al, which resulted in ACTH, and subsequently oral corticosteroids, being widely used to treat MS, particularly for acute exacerbations of the disease. However, robust evidence of long-term steroids remain limited, even to the present.
High-Dose Steroids
By 1980, the initial descriptive treatment of high-dose intravenous (IV) steroids for demyelinating diseases, including MS and transverse myelitis, by Dowling et al resulted in rapid clinical improvement in some patients. This result was ultimately confirmed by others. High-dose IV steroids became the gold standard for acute attacks, particularly those aggressive in nature. In the mid 1980s, work by Troiano et al, as well as others, showed that the rapid use of high-dose IV as well as oral steroids showed similar effects, with reduction or elimination of CT contrast-enhancing lesions within as few as eight hours, while lower doses or alternative-day treatments were less effective. In addition, descriptive studies of immune modulatory and immunosuppressive drugs, as well as small randomized studies, were published. These agents did not receive FDA approval.
The Golden Age of Therapy (1993 to 2018)
A remarkable era in MS prognosis and treatment began with immunomodulation injections of Betaseron (INFβ-1b), Avonex (INFβ-1a), and Copaxone (glatiramer acetate). This can be attributed, at least in part, to advances in molecular biology, genetics, and neuroimaging, and support by corporate, private, and public funding. Since the initial FDA approval of INFβ-1b, 15 MS therapies have become clinically available, including eight injectables, three orals, and four infusion treatments (see Table 1). In addition, two other drugs have been FDA approved for uses other than MS: rituximab (approved for lymphoma) and cladribine (for hairy cell leukemia), with the latter now approved by the European Medicines Agency for MS. Table 1 depicts characteristics of these therapies approved by US or European agencies (or for other disorders increasingly used off label for MS) in an attempt to compare annual relapse rates (ARR) and decreases in the percent of gadolinium-enhancing MS lesions versus placebo. This information was chosen because ARR has been uniformly selected and defined for such trials, while percent decrease of gadolinium-enhancing lesions on MRI has been the most sensitive barometer available for assessing acute clinical activity. As a result, risk-benefit considerations have been critical in evaluating these drug treatments, with efficacy improving greatly over time, whereas risks have been more variable.
Disease Categories
In 1996, Lublin and Reingold provided a new classification, not specifically for the diagnosis of MS, but rather for the clinical course of the disease. Initially, there were four categories—relapsing-remitting MS, secondary progressive MS, primary progressive MS, and progressive-relapsing MS—that were universally identified. These were thought to be relatively distinct clinical categories, but over time it became clear that the classification did not fully distinguish MS disease activity within these categories. For that reason, it was subsequently recommended, by Lincoln et al in 2009 and Cook et al in 2012, to include MRI, a vastly more sensitive modality, as well as clinical data in assessing disease activity.
On another note, MS and neuromyelitis optica (NMO), although having similar features, were clearly identified as different diseases by Lennon et al in 2004. Differences in pathology, clinical characteristics, immunology, and therapy separate the two disorders.
MRI in MS
Work by Young et al in 1981 established the central role of MRI brain imaging in MS diagnosis and therapeutic considerations. Since then it has become ubiquitous.
An example of a sensitive and highly productive MRI protocol is the BECOME study of MS and clinically isolated syndrome by Cadavid et al from 2009 to 2017. In this study, IFNβ-1b was compared with glatiramer acetate treatment. Cadavid et al used a 3T scanner with triple-dose gadolinium, performed monthly for as long as 24 consecutive months. This unique study brought about a virtual gold mine of valuable research and clinical information. This included proof that gadolinium-enhancing lesions persisted for six months or more, evidence of a 30:1 ratio of new MRI brain lesions to clinical activity, and documentation that 96% of T2 lesions and black holes derive from prior gadolinium-enhancing lesions. It was further noted that 80% to 90% of acute black holes disappeared with treatment and 75% to 80% of patients taking IFNβ-1b or glatiramer acetate had new MRI lesions despite continuing treatment. Perhaps most interestingly, monthly MRIs could predict relapse and disability in a relatively small number of patients, depending upon the frequency and activity of MRI lesions. In 2017, Brown et al documented that magnetization transfer ratio recovery in MS brain lesions occurred more significantly with glatiramer acetate than with IFNβ-1b, whereas more chronic black hole lesions were found with glatiramer acetate. Also in 2017, Maranzano et al found evidence of acute inflammatory leukocortical lesions, which were not as well recognized previously.
In summary, it has become increasingly clear that MRI is the most sen
The Future of MS
While it is not yet a curable disease, there is growing evidence that MS prognosis has improved and will continue to improve. This is based on incremental decreases in acute MS exacerbations, progressive disability, and MRI lesion activity, as well as a combination of the three—no evidence of disease activity (NEDA).
Not only are drug therapies becoming more effective, but patients and physicians now have many more treatment options to carefully consider with regard to efficacy, side effect profiles, treatment frequency, route of administration, cost, and quality of life. Newer drugs with different mechanisms of action such as cladribine, now approved in Europe, fulfill most of these beneficial criteria (see Giovannoni et al, 2010). More promising MS treatments, including long-acting induction therapies, are still being evaluated. As with other complex diseases, multiple therapies are likely to be used as well.
In summary, compared with the time before 1993, MS will be much less likely to be a progressive disease, and quality of life will be much improved. In my opinion, patients will be less fearful about their prognosis than ever before, and with appropriate evaluations and treatments, we may realize that disabling MS will be far less common.

Suggested Reading
Brown JW, Pardini M, Brownlee WJ, et al. An abnormal periventricular magnetization transfer ratio gradient occurs early in multiple sclerosis. Brain. 2017;140(2):387-398.
Cadavid D, Wolansky LJ, Skurnick J, et al. Efficacy of treatment of MS with IFNbeta-1b or glatiramer acetate by monthly brain MRI in the BECOME study. Neurology. 2009;72(23):1976-1983.
Cook SD, Dhib-Jalbut S, Dowling P, et al. Use of magnetic resonance imaging as well as clinical disease activity in the clinical classification of multiple sclerosis and assessment of its course: a report from an international CMSC consensus conference, March 5-7, 2010. Int J MS Care. 2012;14(3):105-114.
Dowling PC, Bosch VV, Cook SD. Possible beneficial effect of high-dose intravenous steroid therapy in acute demyelinating disease and transverse myelitis. Neurology. 1980;30(7 Pt 2):33-36.
Giovannoni G, Comi G, Cook S, et al. A placebo-controlled trial of oral cladribine for relapsing multiple sclerosis. N Engl J Med. 2010;362(5):416-426.
Lennon VA, Wingerchuk DM, Kryzer TJ, et al. A serum autoantibody marker of neuromyelitis optica: distinction from multiple sclerosis. Lancet. 2004;364(9451):2106-2112.
Lincoln JA, Cadavid D, Pollard J, et al. We should use magnetic resonance imaging to classify and monitor the course of multiple sclerosis. Arch Neurol. 2009;66(3):412-414.
Lublin FD, Reingold SC. Defining the clinical course of multiple sclerosis: results of an international survey. National Multiple Sclerosis Society (USA) Advisory Committee on Clinical Trials of New Agents in Multiple Sclerosis. Neurology. 1996;46(4):907-911.
Maranzano J, Rudko DA, Nakamura K, et al. MRI evidence of acute inflammation in leukocortical lesions of patients with early multiple sclerosis. Neurology. 2017;89(7):714-721.
McDonald WI, Compston A, Edan G, et al. Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol. 2001;50(1):121-127.
Miller HG, Gibbons JL. Acute disseminated encephalomyelitis and acute disseminated sclerosis; results of treatment with A.C.T.H. Br Med J. 1953;2(4850):1345-1348.
Poser CM, Paty DW, Scheinberg L, et al. New diagnostic criteria for multiple sclerosis: guidelines for research protocols. Ann Neurol. 1983;13(3):227-231.
Rose AS, Kuzma JW, Kurtzke JF, et al. Cooperative study in the evaluation of therapy in multiple sclerosis. ACTH vs. placebo--final report. Neurology. 1970;20(5):1-59.
Troiano R, Hafstein M, Ruderman M, et al. Effect of high-dose intravenous steroid administration on contrast-enhancing computed tomographic scan lesions in multiple sclerosis. Ann Neurol. 1984;15(3):257-263.
Troiano RA, Hafstein MP, Zito G, et al. The effect of oral corticosteroid dosage on CT enhancing multiple sclerosis plaques. J Neurol Sci. 1985;70(1):67-72.
Young IR, Hall AS, Pallis A, et al. Nuclear magnetic resonance imaging of the brain in multiple sclerosis. Lancet. 1981;2(8255):1063-1066.
VIDEO: Managing the alemtuzumab paradox
SAN DIEGO – A paradox of treating people with the monoclonal antibody alemtuzumab is that the agent can be an effective therapy for many people living with multiple sclerosis, but in some patients is associated with the development of other autoimmune diseases.
“I counsel patients with multiple sclerosis that this is a high risk, high gain drug,” Alasdair Coles, MD, of the University of Cambridge (England), said at the meeting, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
Patients can make it safer through strict compliance with the drug’s risk monitoring program. But without patient compliance, alemtuzumab becomes a dangerous drug, he said in a video interview.
Knowing in advance which patients are at an elevated risk for subsequent autoimmune diseases has been difficult to predict. But researchers are getting closer, Dr. Coles said. In the future, measuring a serum factor, potentially interleukin 21, could produce a pretreatment risk assessment for each individual.
SAN DIEGO – A paradox of treating people with the monoclonal antibody alemtuzumab is that the agent can be an effective therapy for many people living with multiple sclerosis, but in some patients is associated with the development of other autoimmune diseases.
“I counsel patients with multiple sclerosis that this is a high risk, high gain drug,” Alasdair Coles, MD, of the University of Cambridge (England), said at the meeting, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
Patients can make it safer through strict compliance with the drug’s risk monitoring program. But without patient compliance, alemtuzumab becomes a dangerous drug, he said in a video interview.
Knowing in advance which patients are at an elevated risk for subsequent autoimmune diseases has been difficult to predict. But researchers are getting closer, Dr. Coles said. In the future, measuring a serum factor, potentially interleukin 21, could produce a pretreatment risk assessment for each individual.
SAN DIEGO – A paradox of treating people with the monoclonal antibody alemtuzumab is that the agent can be an effective therapy for many people living with multiple sclerosis, but in some patients is associated with the development of other autoimmune diseases.
“I counsel patients with multiple sclerosis that this is a high risk, high gain drug,” Alasdair Coles, MD, of the University of Cambridge (England), said at the meeting, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
Patients can make it safer through strict compliance with the drug’s risk monitoring program. But without patient compliance, alemtuzumab becomes a dangerous drug, he said in a video interview.
Knowing in advance which patients are at an elevated risk for subsequent autoimmune diseases has been difficult to predict. But researchers are getting closer, Dr. Coles said. In the future, measuring a serum factor, potentially interleukin 21, could produce a pretreatment risk assessment for each individual.
REPORTING FROM ACTRIMS FORUM 2018
Third course of alemtuzumab can improve MS outcomes
SAN DIEGO – Approximately 30% of people with active multiple sclerosis who initially responded well to two courses of alemtuzumab in the CARE-MS II trial experience relapse or MRI activity over time. But investigators set out to determine whether retreatment with a subsequent course of alemtuzumab is worthwhile.
“What we found is, after the third course, they continued to do well again – at this point for an average of another 3-4 years,” Ann D. Bass, MD, said in an interview at the meeting, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
“The take-home message is if they do need to be retreated after those two courses, it doesn’t [necessarily] mean they are a treatment failure. Give them another chance and see if they will do well with a third course, which is what they did,” said Dr. Bass, of the Neurology Center of San Antonio.
Through year 6 of an extension study with 393 of the original CARE-MS II trial participants, 45% received one or more additional courses of alemtuzumab. Participants were required to wait 12 months or more after completion of the two initial courses of therapy. This figure includes 30% who received a third course of alemtuzumab, 12% who received a fourth course, and 2% and 1% who received a fifth or sixth course, respectively.
“The average time until they needed a third course was 2.5 years after the second course; so they didn’t need it right away,” Dr. Bass said. “The majority, even at 6 years, did not need to have a third course.”
When patients did require a subsequent course, “about half needed it because of clinical relapse; one-quarter needed it because of MRI relapse; and about one-quarter needed it because of both,” Dr. Bass said during a poster presentation.
In terms of effectiveness, the annual relapse rate significantly decreased following a third course of alemtuzumab, from 0.85 in the 12 months prior to the third course to 0.20 in the 12 months after (P less than .0001). In these patients, the annual relapse rate remained low, at 0.17, up to 3 years later.
Investigators also tracked disability using the Expanded Disability Status Scale. They found that more than two-thirds, 68%, maintained stable or had improved scores after administration of a third alemtuzumab course. In addition, the percentage of patients with confirmed disability improvement increased from 4.4% in the 12 months prior to a third course to 14.4% in the year following pretreatment.
Retreatment was at the patient’s discretion. “The patients have the right to say ‘No, I’m doing great. I don’t want to be retreated’ or ‘I want to explore other options’ for whatever reason,” Dr. Bass said. “That’s rare though; most patients actually say yes.”
To qualify for retreatment based on MRI findings, patients had to have at least two lesions – one enlarging and one enhancing, two enlarging, or two new.
Only patients who opted for a subsequent course of alemtuzumab were included in the current analysis; those who chose a different disease-modifying therapy were excluded.
“Many achieve clinical and MRI remission. I never say cure – you don’t want to say that word,” Dr. Bass said.
Sanofi and Bayer HealthCare Pharmaceuticals supported the study. Dr. Bass reported that she is a principal investigator, speaker, and member of the advisory board for Sanofi Genzyme.
SOURCE: Bass A et al. ACTRIMS Forum 2018 Poster P035.
SAN DIEGO – Approximately 30% of people with active multiple sclerosis who initially responded well to two courses of alemtuzumab in the CARE-MS II trial experience relapse or MRI activity over time. But investigators set out to determine whether retreatment with a subsequent course of alemtuzumab is worthwhile.
“What we found is, after the third course, they continued to do well again – at this point for an average of another 3-4 years,” Ann D. Bass, MD, said in an interview at the meeting, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
“The take-home message is if they do need to be retreated after those two courses, it doesn’t [necessarily] mean they are a treatment failure. Give them another chance and see if they will do well with a third course, which is what they did,” said Dr. Bass, of the Neurology Center of San Antonio.
Through year 6 of an extension study with 393 of the original CARE-MS II trial participants, 45% received one or more additional courses of alemtuzumab. Participants were required to wait 12 months or more after completion of the two initial courses of therapy. This figure includes 30% who received a third course of alemtuzumab, 12% who received a fourth course, and 2% and 1% who received a fifth or sixth course, respectively.
“The average time until they needed a third course was 2.5 years after the second course; so they didn’t need it right away,” Dr. Bass said. “The majority, even at 6 years, did not need to have a third course.”
When patients did require a subsequent course, “about half needed it because of clinical relapse; one-quarter needed it because of MRI relapse; and about one-quarter needed it because of both,” Dr. Bass said during a poster presentation.
In terms of effectiveness, the annual relapse rate significantly decreased following a third course of alemtuzumab, from 0.85 in the 12 months prior to the third course to 0.20 in the 12 months after (P less than .0001). In these patients, the annual relapse rate remained low, at 0.17, up to 3 years later.
Investigators also tracked disability using the Expanded Disability Status Scale. They found that more than two-thirds, 68%, maintained stable or had improved scores after administration of a third alemtuzumab course. In addition, the percentage of patients with confirmed disability improvement increased from 4.4% in the 12 months prior to a third course to 14.4% in the year following pretreatment.
Retreatment was at the patient’s discretion. “The patients have the right to say ‘No, I’m doing great. I don’t want to be retreated’ or ‘I want to explore other options’ for whatever reason,” Dr. Bass said. “That’s rare though; most patients actually say yes.”
To qualify for retreatment based on MRI findings, patients had to have at least two lesions – one enlarging and one enhancing, two enlarging, or two new.
Only patients who opted for a subsequent course of alemtuzumab were included in the current analysis; those who chose a different disease-modifying therapy were excluded.
“Many achieve clinical and MRI remission. I never say cure – you don’t want to say that word,” Dr. Bass said.
Sanofi and Bayer HealthCare Pharmaceuticals supported the study. Dr. Bass reported that she is a principal investigator, speaker, and member of the advisory board for Sanofi Genzyme.
SOURCE: Bass A et al. ACTRIMS Forum 2018 Poster P035.
SAN DIEGO – Approximately 30% of people with active multiple sclerosis who initially responded well to two courses of alemtuzumab in the CARE-MS II trial experience relapse or MRI activity over time. But investigators set out to determine whether retreatment with a subsequent course of alemtuzumab is worthwhile.
“What we found is, after the third course, they continued to do well again – at this point for an average of another 3-4 years,” Ann D. Bass, MD, said in an interview at the meeting, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
“The take-home message is if they do need to be retreated after those two courses, it doesn’t [necessarily] mean they are a treatment failure. Give them another chance and see if they will do well with a third course, which is what they did,” said Dr. Bass, of the Neurology Center of San Antonio.
Through year 6 of an extension study with 393 of the original CARE-MS II trial participants, 45% received one or more additional courses of alemtuzumab. Participants were required to wait 12 months or more after completion of the two initial courses of therapy. This figure includes 30% who received a third course of alemtuzumab, 12% who received a fourth course, and 2% and 1% who received a fifth or sixth course, respectively.
“The average time until they needed a third course was 2.5 years after the second course; so they didn’t need it right away,” Dr. Bass said. “The majority, even at 6 years, did not need to have a third course.”
When patients did require a subsequent course, “about half needed it because of clinical relapse; one-quarter needed it because of MRI relapse; and about one-quarter needed it because of both,” Dr. Bass said during a poster presentation.
In terms of effectiveness, the annual relapse rate significantly decreased following a third course of alemtuzumab, from 0.85 in the 12 months prior to the third course to 0.20 in the 12 months after (P less than .0001). In these patients, the annual relapse rate remained low, at 0.17, up to 3 years later.
Investigators also tracked disability using the Expanded Disability Status Scale. They found that more than two-thirds, 68%, maintained stable or had improved scores after administration of a third alemtuzumab course. In addition, the percentage of patients with confirmed disability improvement increased from 4.4% in the 12 months prior to a third course to 14.4% in the year following pretreatment.
Retreatment was at the patient’s discretion. “The patients have the right to say ‘No, I’m doing great. I don’t want to be retreated’ or ‘I want to explore other options’ for whatever reason,” Dr. Bass said. “That’s rare though; most patients actually say yes.”
To qualify for retreatment based on MRI findings, patients had to have at least two lesions – one enlarging and one enhancing, two enlarging, or two new.
Only patients who opted for a subsequent course of alemtuzumab were included in the current analysis; those who chose a different disease-modifying therapy were excluded.
“Many achieve clinical and MRI remission. I never say cure – you don’t want to say that word,” Dr. Bass said.
Sanofi and Bayer HealthCare Pharmaceuticals supported the study. Dr. Bass reported that she is a principal investigator, speaker, and member of the advisory board for Sanofi Genzyme.
SOURCE: Bass A et al. ACTRIMS Forum 2018 Poster P035.
REPORTING FROM ACTRIMS FORUM 2018
Key clinical point:
Major finding: The annual relapse rate significantly decreased following a third course of alemtuzumab, from 0.85 in the 12 months prior to the third course to 0.20 in the 12 months after (P less than .0001).
Study details: An extension study of the CARE-MS II trial involving 393 of the original study participants.
Disclosures: Sanofi and Bayer HealthCare Pharmaceuticals supported the study. Dr. Bass reported that she is a principal investigator, speaker, and member of the advisory board for Sanofi Genzyme.
Source: Bass A et al. ACTRIMS Forum 2018 Poster P035.
Switching RRMS patients to daclizumab beta appears safe
SAN DIEGO – Switching relapsing-remitting multiple sclerosis patients from glatiramer acetate to daclizumab beta resulted in no increase in the adverse event profile and was associated with superior efficacy, a post hoc analysis of data from the DECIDE study showed.
“There is always a challenge in transitioning patients from one therapeutic agent to another, with concerns for adequate efficacy to justify the switch, and heightened risks of toxicity or adverse events,” lead study author Stanley L. Cohan, MD, PhD, said in an interview prior to the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “Daclizumab is not a first-line or platform therapeutic agent, but has clearly superior efficacy to first-line medication, and, based upon the current data presented, safety and efficacy are not adversely influenced by prior treatment history with a first-line agent.”
In the phase 3 DECIDE trial, daclizumab beta 150 mg demonstrated greater efficacy versus intramuscular (IM) interferon (IFN) beta-1a 30 mcg on several clinical, radiographic, and patient-centered outcomes in patients with relapsing-remitting multiple sclerosis (N Engl J Med 2015; 373:1418-28). The purpose of the current study was to examine the efficacy and safety of daclizumab beta vs. IM IFN beta-1a in the subgroup of RRMS patients treated with glatiramer acetate (GA) as their only previous disease-modifying therapy (DMT) before entering DECIDE.
Dr. Cohan, a neurologist who directs the Providence MS Center at the Providence Brain and Spine Institute in Portland, Ore., and his associates reported data from 42 of 922 (5%) IM IFN beta-1a and 50 of 919 (5%) daclizumab beta patients who had received treatment with GA only prior to DECIDE. Baseline characteristics were balanced between treatment groups, including duration of prior treatment with GA, reasons for discontinuing GA, and time between GA discontinuation and start of treatment in DECIDE. The annualized relapse rate was 42% lower in patients treated with daclizumab beta vs. IM IFN beta-1a (rate ratio of 0.58). Daclizumab beta also reduced risk of relapse by 53% (hazard ratio of 0.47; P = .048) and the mean number of new or newly-enlarging T2-hyperintense lesions at week 96 by 58% (lesion mean ratio, 0.42; P = .021) vs. IM IFN beta-1a.
In patients treated with GA only before DECIDE, 98% of IM IFN beta-1a patients and 94% of daclizumab beta patients reported any adverse event (AE). In all, 2% of IM IFN beta-1a patients and 16% of daclizumab beta patients had a serious AE (excluding MS relapse), and 10% of IM IFN beta-1a and 12% of daclizumab beta patients discontinued treatment because of an AE (excluding MS relapse).
The incidence of elevations of alanine aminotransferase or aspartate aminotransferase three times the upper limit of normal or greater was 10% in the IM IFN beta-1a group and 8% in the daclizumab beta, while the ALT and AST elevations were greater than five times the ULN in 0% and 4% of patients, respectively.
Serious adverse events were reported in eight participants in the daclizumab beta group (including abortion induced, ankle fracture, anal fistula, anxiety, appendicitis perforated, convulsion, pelvic abscess, inguinal hernia, abnormal cervix smear), and one participant in the IM IFN beta-1a group (ligament rupture).
“This post hoc analysis demonstrates that in switching from glatiramer to daclizumab there was no unanticipated or increase in the AE profile, and that a switch from glatiramer to daclizumab was associated with superior efficacy, again in line with overall efficacy observed for daclizumab in this study,” Dr. Cohan said.
He acknowledged certain limitations of the study, including the post hoc nature of the analysis. “The small glatiramer cohort size, and the large percentage of former glatiramer patients who entered DECIDE because of lack of glatiramer efficacy may have introduced a selection bias which would magnify the seeming efficacy of daclizumab, and interferon-beta,” he added.
Dr. Cohan reported that he receives research support from Biogen, Novartis, Roche, Sanofi, and Mallinckrodt, and speaking honoraria from Acorda, Biogen, Roche, and Sanofi. He has served on advisory boards for Biogen, Sanofi, and Novartis.
SOURCE: Cohan et al. ACTRIMS Forum 2018, Poster 42.
SAN DIEGO – Switching relapsing-remitting multiple sclerosis patients from glatiramer acetate to daclizumab beta resulted in no increase in the adverse event profile and was associated with superior efficacy, a post hoc analysis of data from the DECIDE study showed.
“There is always a challenge in transitioning patients from one therapeutic agent to another, with concerns for adequate efficacy to justify the switch, and heightened risks of toxicity or adverse events,” lead study author Stanley L. Cohan, MD, PhD, said in an interview prior to the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “Daclizumab is not a first-line or platform therapeutic agent, but has clearly superior efficacy to first-line medication, and, based upon the current data presented, safety and efficacy are not adversely influenced by prior treatment history with a first-line agent.”
In the phase 3 DECIDE trial, daclizumab beta 150 mg demonstrated greater efficacy versus intramuscular (IM) interferon (IFN) beta-1a 30 mcg on several clinical, radiographic, and patient-centered outcomes in patients with relapsing-remitting multiple sclerosis (N Engl J Med 2015; 373:1418-28). The purpose of the current study was to examine the efficacy and safety of daclizumab beta vs. IM IFN beta-1a in the subgroup of RRMS patients treated with glatiramer acetate (GA) as their only previous disease-modifying therapy (DMT) before entering DECIDE.
Dr. Cohan, a neurologist who directs the Providence MS Center at the Providence Brain and Spine Institute in Portland, Ore., and his associates reported data from 42 of 922 (5%) IM IFN beta-1a and 50 of 919 (5%) daclizumab beta patients who had received treatment with GA only prior to DECIDE. Baseline characteristics were balanced between treatment groups, including duration of prior treatment with GA, reasons for discontinuing GA, and time between GA discontinuation and start of treatment in DECIDE. The annualized relapse rate was 42% lower in patients treated with daclizumab beta vs. IM IFN beta-1a (rate ratio of 0.58). Daclizumab beta also reduced risk of relapse by 53% (hazard ratio of 0.47; P = .048) and the mean number of new or newly-enlarging T2-hyperintense lesions at week 96 by 58% (lesion mean ratio, 0.42; P = .021) vs. IM IFN beta-1a.
In patients treated with GA only before DECIDE, 98% of IM IFN beta-1a patients and 94% of daclizumab beta patients reported any adverse event (AE). In all, 2% of IM IFN beta-1a patients and 16% of daclizumab beta patients had a serious AE (excluding MS relapse), and 10% of IM IFN beta-1a and 12% of daclizumab beta patients discontinued treatment because of an AE (excluding MS relapse).
The incidence of elevations of alanine aminotransferase or aspartate aminotransferase three times the upper limit of normal or greater was 10% in the IM IFN beta-1a group and 8% in the daclizumab beta, while the ALT and AST elevations were greater than five times the ULN in 0% and 4% of patients, respectively.
Serious adverse events were reported in eight participants in the daclizumab beta group (including abortion induced, ankle fracture, anal fistula, anxiety, appendicitis perforated, convulsion, pelvic abscess, inguinal hernia, abnormal cervix smear), and one participant in the IM IFN beta-1a group (ligament rupture).
“This post hoc analysis demonstrates that in switching from glatiramer to daclizumab there was no unanticipated or increase in the AE profile, and that a switch from glatiramer to daclizumab was associated with superior efficacy, again in line with overall efficacy observed for daclizumab in this study,” Dr. Cohan said.
He acknowledged certain limitations of the study, including the post hoc nature of the analysis. “The small glatiramer cohort size, and the large percentage of former glatiramer patients who entered DECIDE because of lack of glatiramer efficacy may have introduced a selection bias which would magnify the seeming efficacy of daclizumab, and interferon-beta,” he added.
Dr. Cohan reported that he receives research support from Biogen, Novartis, Roche, Sanofi, and Mallinckrodt, and speaking honoraria from Acorda, Biogen, Roche, and Sanofi. He has served on advisory boards for Biogen, Sanofi, and Novartis.
SOURCE: Cohan et al. ACTRIMS Forum 2018, Poster 42.
SAN DIEGO – Switching relapsing-remitting multiple sclerosis patients from glatiramer acetate to daclizumab beta resulted in no increase in the adverse event profile and was associated with superior efficacy, a post hoc analysis of data from the DECIDE study showed.
“There is always a challenge in transitioning patients from one therapeutic agent to another, with concerns for adequate efficacy to justify the switch, and heightened risks of toxicity or adverse events,” lead study author Stanley L. Cohan, MD, PhD, said in an interview prior to the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “Daclizumab is not a first-line or platform therapeutic agent, but has clearly superior efficacy to first-line medication, and, based upon the current data presented, safety and efficacy are not adversely influenced by prior treatment history with a first-line agent.”
In the phase 3 DECIDE trial, daclizumab beta 150 mg demonstrated greater efficacy versus intramuscular (IM) interferon (IFN) beta-1a 30 mcg on several clinical, radiographic, and patient-centered outcomes in patients with relapsing-remitting multiple sclerosis (N Engl J Med 2015; 373:1418-28). The purpose of the current study was to examine the efficacy and safety of daclizumab beta vs. IM IFN beta-1a in the subgroup of RRMS patients treated with glatiramer acetate (GA) as their only previous disease-modifying therapy (DMT) before entering DECIDE.
Dr. Cohan, a neurologist who directs the Providence MS Center at the Providence Brain and Spine Institute in Portland, Ore., and his associates reported data from 42 of 922 (5%) IM IFN beta-1a and 50 of 919 (5%) daclizumab beta patients who had received treatment with GA only prior to DECIDE. Baseline characteristics were balanced between treatment groups, including duration of prior treatment with GA, reasons for discontinuing GA, and time between GA discontinuation and start of treatment in DECIDE. The annualized relapse rate was 42% lower in patients treated with daclizumab beta vs. IM IFN beta-1a (rate ratio of 0.58). Daclizumab beta also reduced risk of relapse by 53% (hazard ratio of 0.47; P = .048) and the mean number of new or newly-enlarging T2-hyperintense lesions at week 96 by 58% (lesion mean ratio, 0.42; P = .021) vs. IM IFN beta-1a.
In patients treated with GA only before DECIDE, 98% of IM IFN beta-1a patients and 94% of daclizumab beta patients reported any adverse event (AE). In all, 2% of IM IFN beta-1a patients and 16% of daclizumab beta patients had a serious AE (excluding MS relapse), and 10% of IM IFN beta-1a and 12% of daclizumab beta patients discontinued treatment because of an AE (excluding MS relapse).
The incidence of elevations of alanine aminotransferase or aspartate aminotransferase three times the upper limit of normal or greater was 10% in the IM IFN beta-1a group and 8% in the daclizumab beta, while the ALT and AST elevations were greater than five times the ULN in 0% and 4% of patients, respectively.
Serious adverse events were reported in eight participants in the daclizumab beta group (including abortion induced, ankle fracture, anal fistula, anxiety, appendicitis perforated, convulsion, pelvic abscess, inguinal hernia, abnormal cervix smear), and one participant in the IM IFN beta-1a group (ligament rupture).
“This post hoc analysis demonstrates that in switching from glatiramer to daclizumab there was no unanticipated or increase in the AE profile, and that a switch from glatiramer to daclizumab was associated with superior efficacy, again in line with overall efficacy observed for daclizumab in this study,” Dr. Cohan said.
He acknowledged certain limitations of the study, including the post hoc nature of the analysis. “The small glatiramer cohort size, and the large percentage of former glatiramer patients who entered DECIDE because of lack of glatiramer efficacy may have introduced a selection bias which would magnify the seeming efficacy of daclizumab, and interferon-beta,” he added.
Dr. Cohan reported that he receives research support from Biogen, Novartis, Roche, Sanofi, and Mallinckrodt, and speaking honoraria from Acorda, Biogen, Roche, and Sanofi. He has served on advisory boards for Biogen, Sanofi, and Novartis.
SOURCE: Cohan et al. ACTRIMS Forum 2018, Poster 42.
REPORTING FROM ACTRIMS FORUM 2018
Key clinical point:
Major finding: The annualized relapse rate was 42% lower in patients treated with daclizumab beta vs. IM IFN beta-1a (rate ratio of 0.58).
Study details: A post hoc analysis of 42 of 922 (5%) IM IFN beta-1a and 50 of 919 (5%) daclizumab beta patients who had received treatment with glatiramer acetate only prior to the DECIDE trial.
Disclosures: Dr. Cohan reported that he receives research support from Biogen, Novartis, Roche, Sanofi, and Mallinckrodt, and speaking honoraria from Acorda, Biogen, Roche, and Sanofi. He has served on advisory boards for Biogen, Sanofi, and Novartis.
Source: Cohan S et al. ACTRIMS Forum 2018 Poster 42.
VIDEO: Oral ozanimod shows promise for relapsing MS
SAN DIEGO – A pair of phase 3 studies offer promising results regarding the safety and efficacy of ozanimod, an experimental immunomodulator, in the treatment of relapsing multiple sclerosis (RMS).
The medication targets sphingosine 1-phosphate 1 and 5 receptors. Industry-funded researchers tested it in two studies against interferon beta-1a.
One study, called SUNBEAM, tested once-daily oral ozanimod (1 mg or 0.5 mg with 7-day dose escalation) against interferon beta-1a (via 30 mcg weekly intramuscular injection) for at least 12 months in 1,346 patients with RMS. The annualized relapse rate, the primary endpoint, was lower in the ozanimod groups versus interferon. For the 1-mg dose, it was 0.181 (P less than .0001), and for 0.5-mg dose, 0.241 (P = .0013).
The number of serious treatment-emergent adverse events in the three groups was low, ranging from 2.5% to 3.5%.
The other study, called RADIANCE, was a similar trial that lasted 24 months. In it, the rate of serious treatment-emergent adverse events in the three groups were similar, ranging from 6.4% to 7.1%.
Ozanimod offers “an excellent therapeutic benefit for patients and a very clean safety profile,” said Bruce Cree, MD, PhD, clinical research director at the University of California, San Francisco, Multiple Sclerosis Center. He is an author on both studies and spoke in a video interview at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
He said the ozanimod should be especially useful as a first-line treatment for MS. The drug is currently being evaluated by the Food and Drug Administration for an RMS indication, and it is also being developed for Crohn’s disease and ulcerative colitis, he said.
The study was funded by Receptos, a wholly owned subsidiary of Celgene. Dr. Cree reported that he has been a consultant to AbbVie, Biogen, EMD Serono, Genzyme, Novartis, and Shire.
SOURCE: Cree B et al. ACTRIMS Forum 2018, abstract P030, and Comi G et al. ACTRIMS Forum 2018, abstract P023
SAN DIEGO – A pair of phase 3 studies offer promising results regarding the safety and efficacy of ozanimod, an experimental immunomodulator, in the treatment of relapsing multiple sclerosis (RMS).
The medication targets sphingosine 1-phosphate 1 and 5 receptors. Industry-funded researchers tested it in two studies against interferon beta-1a.
One study, called SUNBEAM, tested once-daily oral ozanimod (1 mg or 0.5 mg with 7-day dose escalation) against interferon beta-1a (via 30 mcg weekly intramuscular injection) for at least 12 months in 1,346 patients with RMS. The annualized relapse rate, the primary endpoint, was lower in the ozanimod groups versus interferon. For the 1-mg dose, it was 0.181 (P less than .0001), and for 0.5-mg dose, 0.241 (P = .0013).
The number of serious treatment-emergent adverse events in the three groups was low, ranging from 2.5% to 3.5%.
The other study, called RADIANCE, was a similar trial that lasted 24 months. In it, the rate of serious treatment-emergent adverse events in the three groups were similar, ranging from 6.4% to 7.1%.
Ozanimod offers “an excellent therapeutic benefit for patients and a very clean safety profile,” said Bruce Cree, MD, PhD, clinical research director at the University of California, San Francisco, Multiple Sclerosis Center. He is an author on both studies and spoke in a video interview at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
He said the ozanimod should be especially useful as a first-line treatment for MS. The drug is currently being evaluated by the Food and Drug Administration for an RMS indication, and it is also being developed for Crohn’s disease and ulcerative colitis, he said.
The study was funded by Receptos, a wholly owned subsidiary of Celgene. Dr. Cree reported that he has been a consultant to AbbVie, Biogen, EMD Serono, Genzyme, Novartis, and Shire.
SOURCE: Cree B et al. ACTRIMS Forum 2018, abstract P030, and Comi G et al. ACTRIMS Forum 2018, abstract P023
SAN DIEGO – A pair of phase 3 studies offer promising results regarding the safety and efficacy of ozanimod, an experimental immunomodulator, in the treatment of relapsing multiple sclerosis (RMS).
The medication targets sphingosine 1-phosphate 1 and 5 receptors. Industry-funded researchers tested it in two studies against interferon beta-1a.
One study, called SUNBEAM, tested once-daily oral ozanimod (1 mg or 0.5 mg with 7-day dose escalation) against interferon beta-1a (via 30 mcg weekly intramuscular injection) for at least 12 months in 1,346 patients with RMS. The annualized relapse rate, the primary endpoint, was lower in the ozanimod groups versus interferon. For the 1-mg dose, it was 0.181 (P less than .0001), and for 0.5-mg dose, 0.241 (P = .0013).
The number of serious treatment-emergent adverse events in the three groups was low, ranging from 2.5% to 3.5%.
The other study, called RADIANCE, was a similar trial that lasted 24 months. In it, the rate of serious treatment-emergent adverse events in the three groups were similar, ranging from 6.4% to 7.1%.
Ozanimod offers “an excellent therapeutic benefit for patients and a very clean safety profile,” said Bruce Cree, MD, PhD, clinical research director at the University of California, San Francisco, Multiple Sclerosis Center. He is an author on both studies and spoke in a video interview at ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
He said the ozanimod should be especially useful as a first-line treatment for MS. The drug is currently being evaluated by the Food and Drug Administration for an RMS indication, and it is also being developed for Crohn’s disease and ulcerative colitis, he said.
The study was funded by Receptos, a wholly owned subsidiary of Celgene. Dr. Cree reported that he has been a consultant to AbbVie, Biogen, EMD Serono, Genzyme, Novartis, and Shire.
SOURCE: Cree B et al. ACTRIMS Forum 2018, abstract P030, and Comi G et al. ACTRIMS Forum 2018, abstract P023
REPORTING FROM ACTRIMS FORUM 2018
VIDEO: Efficacy of DMTs decreases with age
San Diego – , and high-efficacy drugs do a better job of inhibiting MS disability compared with low-efficacy drugs only in patients younger than 40.5 years.
Those are the key conclusions from a meta-analysis of the age-dependent efficacy of MS treatments that was published in the November 2017 issue of Frontiers in Neurology. In a video interview, Ann Marie Weideman, lead study author, discussed highlights from the meta-analysis at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. The meta-analysis drew from more than 28,000 individuals with MS participating in 38 trials of 13 categories of immunomodulatory drugs.
Ms. Weideman is an IRTA Fellow at the National Institute of Neurological Disorders and Stroke, Bethesda, Md. She reported that study coauthor Bibiana Bielekova, MD, is coinventor of several patents related to daclizumab.
San Diego – , and high-efficacy drugs do a better job of inhibiting MS disability compared with low-efficacy drugs only in patients younger than 40.5 years.
Those are the key conclusions from a meta-analysis of the age-dependent efficacy of MS treatments that was published in the November 2017 issue of Frontiers in Neurology. In a video interview, Ann Marie Weideman, lead study author, discussed highlights from the meta-analysis at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. The meta-analysis drew from more than 28,000 individuals with MS participating in 38 trials of 13 categories of immunomodulatory drugs.
Ms. Weideman is an IRTA Fellow at the National Institute of Neurological Disorders and Stroke, Bethesda, Md. She reported that study coauthor Bibiana Bielekova, MD, is coinventor of several patents related to daclizumab.
San Diego – , and high-efficacy drugs do a better job of inhibiting MS disability compared with low-efficacy drugs only in patients younger than 40.5 years.
Those are the key conclusions from a meta-analysis of the age-dependent efficacy of MS treatments that was published in the November 2017 issue of Frontiers in Neurology. In a video interview, Ann Marie Weideman, lead study author, discussed highlights from the meta-analysis at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. The meta-analysis drew from more than 28,000 individuals with MS participating in 38 trials of 13 categories of immunomodulatory drugs.
Ms. Weideman is an IRTA Fellow at the National Institute of Neurological Disorders and Stroke, Bethesda, Md. She reported that study coauthor Bibiana Bielekova, MD, is coinventor of several patents related to daclizumab.
REPORTING FROM ACTRIMS FORUM 2018
VIDEO: New MS ambulatory measure could fill clinical gap
REPORTING FROM ACTRIMS FORUM 2018
SAN DIEGO – Although clinical tools to assess ambulatory function among people with multiple sclerosis exist, some measure it as part of a comprehensive assessment while others require the patient to answer many questions and then clinicians to calculate a score.
To devise a more targeted, simpler instrument, Emily Evans, MD, and her colleagues developed the PDAS or Patient Derived Ambulation Scale. They evaluated the correlation of this single-item scale to assess ambulation – an important measure of patient function – and evaluated how the results correlated with existing tools such as the Patient Determined Disease Steps and 12-item MS Walking Scale. Dr. Evans presented preliminary findings at the ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
“We feel this is a quick test that can be readily implemented into clinical practice,” Dr. Evans, a neurologist at the John L. Trotter MS Center at Washington University in St. Louis, said in a video interview.
REPORTING FROM ACTRIMS FORUM 2018
SAN DIEGO – Although clinical tools to assess ambulatory function among people with multiple sclerosis exist, some measure it as part of a comprehensive assessment while others require the patient to answer many questions and then clinicians to calculate a score.
To devise a more targeted, simpler instrument, Emily Evans, MD, and her colleagues developed the PDAS or Patient Derived Ambulation Scale. They evaluated the correlation of this single-item scale to assess ambulation – an important measure of patient function – and evaluated how the results correlated with existing tools such as the Patient Determined Disease Steps and 12-item MS Walking Scale. Dr. Evans presented preliminary findings at the ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
“We feel this is a quick test that can be readily implemented into clinical practice,” Dr. Evans, a neurologist at the John L. Trotter MS Center at Washington University in St. Louis, said in a video interview.
REPORTING FROM ACTRIMS FORUM 2018
SAN DIEGO – Although clinical tools to assess ambulatory function among people with multiple sclerosis exist, some measure it as part of a comprehensive assessment while others require the patient to answer many questions and then clinicians to calculate a score.
To devise a more targeted, simpler instrument, Emily Evans, MD, and her colleagues developed the PDAS or Patient Derived Ambulation Scale. They evaluated the correlation of this single-item scale to assess ambulation – an important measure of patient function – and evaluated how the results correlated with existing tools such as the Patient Determined Disease Steps and 12-item MS Walking Scale. Dr. Evans presented preliminary findings at the ACTRIMS Forum 2018, held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
“We feel this is a quick test that can be readily implemented into clinical practice,” Dr. Evans, a neurologist at the John L. Trotter MS Center at Washington University in St. Louis, said in a video interview.
VIDEO: Alemtuzumab associated with long-term MS control in TOPAZ study
SAN DIEGO – A majority of patients with active relapsing-remitting multiple sclerosis and inadequate response to previous therapy achieved a durable response after treatment with alemtuzumab in the TOPAZ trial, a 5-year extension to the CARE-MS II study.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Almost half of the 317 participants in TOPAZ received no further therapy beyond their initial two courses of alemtuzumab infusion therapy that they received as part of the CARE-MS II study.
“If you follow patients over time ... you’re seeing a significant group of patients who have improvement. It’s very unexpected, especially when you look at the patients who entered the clinical trial who had a fair amount of active disease,” said Barry A. Singer, MD, director of The MS Center for Innovations in Care at Missouri Baptist Medical Center in St. Louis.
At the 7-year evaluation of patients in TOPAZ, the annualized relapse rate was 0.14. In addition, 87% of patients remained relapse-free in year 7. Dr. Singer and his colleagues also reported that 73% of TOPAZ participants were stable or improved based on their Expanded Disability Status Scale (EDSS) scores.
“As we follow the data out and follow these patients out, we’re seeing how the clinical course for these patients is dramatically improving for the majority of patients,” Dr. Singer said in a video interview at ACTRIMS Forum 2018, the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
The TOPAZ study also revealed that 69% of patients were free of clinical disease worsening and 44% experienced clinical disease improvement in the 6 months before year 7. The majority also had no evidence of disease activity, Dr. Singer reported.
“One of the attributes that makes alemtuzumab so attractive as a clinician and for patients is you can go through a couple of series of medication [treatments] ... and really alter your disease course – that is the exciting thing,” he said.
The Food and Drug Administration approved alemtuzumab (Lemtrada) in November 2014 for the treatment of patients with relapsing forms of multiple sclerosis. Use of alemtuzumab is generally reserved for patients who have had an inadequate response to two or more previous drugs indicated for the treatment of multiple sclerosis.
In CARE-MS II, participants received two annual courses of alemtuzumab: intravenous infusion of 12 mg/day for 5 days at baseline and again for 3 days at 12 months. Additional treatment in TOPAZ for relapse or MRI evidence of disease was at the discretion of the investigator and could include alemtuzumab retreatment 12 mg/day on 3 consecutive days 12 months or more after a previous course, or another disease-modifying therapy at any time. Annual follow-up exams included an MRI scan.
A durable treatment effect was achieved by a majority of patients, even though 47% received no further treatment with alemtuzumab or another disease-modifying therapy after the initial two alemtuzumab courses.
The incidence of most adverse events, including infusion-associated reactions and infections, decreased over the course of the TOPAZ study and were lower than the incidence reported in the 2-year CARE-MS II trial. Of note, the incidence of thyroid-related adverse events peaked in the third year of the follow-up and continued to decline out to 7 years, Dr. Singer said. “We’re not seeing any new safety issues.”
Dr. Singer and his coinvestigators plan to continue the research, monitoring and scoring patients over time.
The TOPAZ trial was funded by Sanofi Genzyme, which markets alemtuzumab. Dr. Singer disclosed that he receives clinical research support and is a speaker for Sanofi Genzyme.
SOURCE: Singer B et al. ACTRIMS Forum 2018, abstract P026.
SAN DIEGO – A majority of patients with active relapsing-remitting multiple sclerosis and inadequate response to previous therapy achieved a durable response after treatment with alemtuzumab in the TOPAZ trial, a 5-year extension to the CARE-MS II study.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Almost half of the 317 participants in TOPAZ received no further therapy beyond their initial two courses of alemtuzumab infusion therapy that they received as part of the CARE-MS II study.
“If you follow patients over time ... you’re seeing a significant group of patients who have improvement. It’s very unexpected, especially when you look at the patients who entered the clinical trial who had a fair amount of active disease,” said Barry A. Singer, MD, director of The MS Center for Innovations in Care at Missouri Baptist Medical Center in St. Louis.
At the 7-year evaluation of patients in TOPAZ, the annualized relapse rate was 0.14. In addition, 87% of patients remained relapse-free in year 7. Dr. Singer and his colleagues also reported that 73% of TOPAZ participants were stable or improved based on their Expanded Disability Status Scale (EDSS) scores.
“As we follow the data out and follow these patients out, we’re seeing how the clinical course for these patients is dramatically improving for the majority of patients,” Dr. Singer said in a video interview at ACTRIMS Forum 2018, the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
The TOPAZ study also revealed that 69% of patients were free of clinical disease worsening and 44% experienced clinical disease improvement in the 6 months before year 7. The majority also had no evidence of disease activity, Dr. Singer reported.
“One of the attributes that makes alemtuzumab so attractive as a clinician and for patients is you can go through a couple of series of medication [treatments] ... and really alter your disease course – that is the exciting thing,” he said.
The Food and Drug Administration approved alemtuzumab (Lemtrada) in November 2014 for the treatment of patients with relapsing forms of multiple sclerosis. Use of alemtuzumab is generally reserved for patients who have had an inadequate response to two or more previous drugs indicated for the treatment of multiple sclerosis.
In CARE-MS II, participants received two annual courses of alemtuzumab: intravenous infusion of 12 mg/day for 5 days at baseline and again for 3 days at 12 months. Additional treatment in TOPAZ for relapse or MRI evidence of disease was at the discretion of the investigator and could include alemtuzumab retreatment 12 mg/day on 3 consecutive days 12 months or more after a previous course, or another disease-modifying therapy at any time. Annual follow-up exams included an MRI scan.
A durable treatment effect was achieved by a majority of patients, even though 47% received no further treatment with alemtuzumab or another disease-modifying therapy after the initial two alemtuzumab courses.
The incidence of most adverse events, including infusion-associated reactions and infections, decreased over the course of the TOPAZ study and were lower than the incidence reported in the 2-year CARE-MS II trial. Of note, the incidence of thyroid-related adverse events peaked in the third year of the follow-up and continued to decline out to 7 years, Dr. Singer said. “We’re not seeing any new safety issues.”
Dr. Singer and his coinvestigators plan to continue the research, monitoring and scoring patients over time.
The TOPAZ trial was funded by Sanofi Genzyme, which markets alemtuzumab. Dr. Singer disclosed that he receives clinical research support and is a speaker for Sanofi Genzyme.
SOURCE: Singer B et al. ACTRIMS Forum 2018, abstract P026.
SAN DIEGO – A majority of patients with active relapsing-remitting multiple sclerosis and inadequate response to previous therapy achieved a durable response after treatment with alemtuzumab in the TOPAZ trial, a 5-year extension to the CARE-MS II study.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Almost half of the 317 participants in TOPAZ received no further therapy beyond their initial two courses of alemtuzumab infusion therapy that they received as part of the CARE-MS II study.
“If you follow patients over time ... you’re seeing a significant group of patients who have improvement. It’s very unexpected, especially when you look at the patients who entered the clinical trial who had a fair amount of active disease,” said Barry A. Singer, MD, director of The MS Center for Innovations in Care at Missouri Baptist Medical Center in St. Louis.
At the 7-year evaluation of patients in TOPAZ, the annualized relapse rate was 0.14. In addition, 87% of patients remained relapse-free in year 7. Dr. Singer and his colleagues also reported that 73% of TOPAZ participants were stable or improved based on their Expanded Disability Status Scale (EDSS) scores.
“As we follow the data out and follow these patients out, we’re seeing how the clinical course for these patients is dramatically improving for the majority of patients,” Dr. Singer said in a video interview at ACTRIMS Forum 2018, the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
The TOPAZ study also revealed that 69% of patients were free of clinical disease worsening and 44% experienced clinical disease improvement in the 6 months before year 7. The majority also had no evidence of disease activity, Dr. Singer reported.
“One of the attributes that makes alemtuzumab so attractive as a clinician and for patients is you can go through a couple of series of medication [treatments] ... and really alter your disease course – that is the exciting thing,” he said.
The Food and Drug Administration approved alemtuzumab (Lemtrada) in November 2014 for the treatment of patients with relapsing forms of multiple sclerosis. Use of alemtuzumab is generally reserved for patients who have had an inadequate response to two or more previous drugs indicated for the treatment of multiple sclerosis.
In CARE-MS II, participants received two annual courses of alemtuzumab: intravenous infusion of 12 mg/day for 5 days at baseline and again for 3 days at 12 months. Additional treatment in TOPAZ for relapse or MRI evidence of disease was at the discretion of the investigator and could include alemtuzumab retreatment 12 mg/day on 3 consecutive days 12 months or more after a previous course, or another disease-modifying therapy at any time. Annual follow-up exams included an MRI scan.
A durable treatment effect was achieved by a majority of patients, even though 47% received no further treatment with alemtuzumab or another disease-modifying therapy after the initial two alemtuzumab courses.
The incidence of most adverse events, including infusion-associated reactions and infections, decreased over the course of the TOPAZ study and were lower than the incidence reported in the 2-year CARE-MS II trial. Of note, the incidence of thyroid-related adverse events peaked in the third year of the follow-up and continued to decline out to 7 years, Dr. Singer said. “We’re not seeing any new safety issues.”
Dr. Singer and his coinvestigators plan to continue the research, monitoring and scoring patients over time.
The TOPAZ trial was funded by Sanofi Genzyme, which markets alemtuzumab. Dr. Singer disclosed that he receives clinical research support and is a speaker for Sanofi Genzyme.
SOURCE: Singer B et al. ACTRIMS Forum 2018, abstract P026.
REPORTING FROM ACTRIMS FORUM 2018
Key clinical point:
Major finding: The annualized relapse rate was 0.14 at year 7 among the 87% of participants who remained in the TOPAZ study.
Study details: A 5-year extension study of 317 participants from the initial CARE-MS II trial.
Disclosures: The TOPAZ trial was funded by Sanofi Genzyme, which markets alemtuzumab. Dr. Singer disclosed that he receives clinical research support and is a speaker for Sanofi Genzyme.
Source: Singer B et al. ACTRIMS Forum 2018, abstract P026.
Ibudilast shows promise in progressive MS
San Diego – In a phase 2 trial, .
“That was a striking result,” one of the study authors, Robert T. Naismith, MD, said in an interview prior to presenting the study at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “We’ll need to see how that translates into a possible clinical benefit.”
In a trial known as SPRINT-MS/NN 102, researchers led by principal investigator Robert Fox, MD, a neurologist at the Cleveland Clinic in Ohio, along with colleagues in the NeuroNEXT Network, randomized 255 subjects with primary or secondary progressive MS to receive either ibudilast up to 100 mg/day (50 mg twice daily) or matching placebo and followed them for 96 weeks. They assessed clinical and imaging outcomes every 24 weeks, and the primary outcome was change in brain atrophy as measured by brain parenchymal fraction (BPF) over 96 weeks. Secondary outcomes included magnetization transfer ratio (MTR), diffusion tensor imaging, and optical coherence tomography. Analysis was based on a modified intention-to-treat approach, and linear mixed effects modeling was used to estimate the rate of change within imaging measures for each treatment group.
The 255 subjects were enrolled at 28 U.S. sites and their last follow-up visit was completed on May 11, 2017. The retention rate through week 96 was 86%, or 219 patients. The researchers found that treatment with ibudilast was associated with a 48% slowing in the rate of decline in brain atrophy as measured by BPF. A per-protocol analysis yielded similar results (P = .045). In addition, no increased rate of serious adverse events and no opportunistic infections or cancer signals were observed. As for tolerability, 25% of subjects on placebo and 30% of those on ibudilast discontinued treatment (P = .30). The main treatment-related adverse events were gastrointestinal in nature (20% in the placebo group vs. 67% in the ibudilast group; P = .002), primarily nausea.
In the analysis of key secondary outcomes, the researchers found that the change in MTR in normal-appearing brain tissue was reduced –0.00558 for ibudilast and –0.03064 for placebo, which was a relative reduction of 82% in MTR decline. At the same time, the change in MTR in normal-appearing gray matter was reduced –0.00753 for ibudilast and –0.03210 for placebo, which was a relative reduction of 77% in MTR decline. No significant difference in white matter changes were observed in transverse diffusivity (P = .15) or longitudinal diffusivity (P = .73), compared with placebo.
For the secondary endpoint of disability, a blinded evaluator assessed Expanded Disability Status Scale (EDSS) score at baseline and every 24 weeks. The researchers used a time-to-event Kaplan-Meier analysis and a Cox proportional hazards model to determine time to confirmed EDSS progression not due to relapse. They found that the hazard ratio for progression in the ibudilast group, compared with the placebo group, was 0.74, with a 90% confidence interval of 0.47-1.17 (P = .29).
“Ibudilast is a novel therapy for progressive MS, with a positive phase 2 study,” Dr. Naismith concluded. “Safety and tolerability were favorable. Clinical endpoints from this trial will be forthcoming.”
The NeuroNEXT Network is supported by the National Institute of Neurological Disorders and Stroke. Dr. Naismith reported having no financial disclosures.
SOURCE: Naismith R et al. ACTRIMS Forum 2018 Abstract P029.
San Diego – In a phase 2 trial, .
“That was a striking result,” one of the study authors, Robert T. Naismith, MD, said in an interview prior to presenting the study at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “We’ll need to see how that translates into a possible clinical benefit.”
In a trial known as SPRINT-MS/NN 102, researchers led by principal investigator Robert Fox, MD, a neurologist at the Cleveland Clinic in Ohio, along with colleagues in the NeuroNEXT Network, randomized 255 subjects with primary or secondary progressive MS to receive either ibudilast up to 100 mg/day (50 mg twice daily) or matching placebo and followed them for 96 weeks. They assessed clinical and imaging outcomes every 24 weeks, and the primary outcome was change in brain atrophy as measured by brain parenchymal fraction (BPF) over 96 weeks. Secondary outcomes included magnetization transfer ratio (MTR), diffusion tensor imaging, and optical coherence tomography. Analysis was based on a modified intention-to-treat approach, and linear mixed effects modeling was used to estimate the rate of change within imaging measures for each treatment group.
The 255 subjects were enrolled at 28 U.S. sites and their last follow-up visit was completed on May 11, 2017. The retention rate through week 96 was 86%, or 219 patients. The researchers found that treatment with ibudilast was associated with a 48% slowing in the rate of decline in brain atrophy as measured by BPF. A per-protocol analysis yielded similar results (P = .045). In addition, no increased rate of serious adverse events and no opportunistic infections or cancer signals were observed. As for tolerability, 25% of subjects on placebo and 30% of those on ibudilast discontinued treatment (P = .30). The main treatment-related adverse events were gastrointestinal in nature (20% in the placebo group vs. 67% in the ibudilast group; P = .002), primarily nausea.
In the analysis of key secondary outcomes, the researchers found that the change in MTR in normal-appearing brain tissue was reduced –0.00558 for ibudilast and –0.03064 for placebo, which was a relative reduction of 82% in MTR decline. At the same time, the change in MTR in normal-appearing gray matter was reduced –0.00753 for ibudilast and –0.03210 for placebo, which was a relative reduction of 77% in MTR decline. No significant difference in white matter changes were observed in transverse diffusivity (P = .15) or longitudinal diffusivity (P = .73), compared with placebo.
For the secondary endpoint of disability, a blinded evaluator assessed Expanded Disability Status Scale (EDSS) score at baseline and every 24 weeks. The researchers used a time-to-event Kaplan-Meier analysis and a Cox proportional hazards model to determine time to confirmed EDSS progression not due to relapse. They found that the hazard ratio for progression in the ibudilast group, compared with the placebo group, was 0.74, with a 90% confidence interval of 0.47-1.17 (P = .29).
“Ibudilast is a novel therapy for progressive MS, with a positive phase 2 study,” Dr. Naismith concluded. “Safety and tolerability were favorable. Clinical endpoints from this trial will be forthcoming.”
The NeuroNEXT Network is supported by the National Institute of Neurological Disorders and Stroke. Dr. Naismith reported having no financial disclosures.
SOURCE: Naismith R et al. ACTRIMS Forum 2018 Abstract P029.
San Diego – In a phase 2 trial, .
“That was a striking result,” one of the study authors, Robert T. Naismith, MD, said in an interview prior to presenting the study at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “We’ll need to see how that translates into a possible clinical benefit.”
In a trial known as SPRINT-MS/NN 102, researchers led by principal investigator Robert Fox, MD, a neurologist at the Cleveland Clinic in Ohio, along with colleagues in the NeuroNEXT Network, randomized 255 subjects with primary or secondary progressive MS to receive either ibudilast up to 100 mg/day (50 mg twice daily) or matching placebo and followed them for 96 weeks. They assessed clinical and imaging outcomes every 24 weeks, and the primary outcome was change in brain atrophy as measured by brain parenchymal fraction (BPF) over 96 weeks. Secondary outcomes included magnetization transfer ratio (MTR), diffusion tensor imaging, and optical coherence tomography. Analysis was based on a modified intention-to-treat approach, and linear mixed effects modeling was used to estimate the rate of change within imaging measures for each treatment group.
The 255 subjects were enrolled at 28 U.S. sites and their last follow-up visit was completed on May 11, 2017. The retention rate through week 96 was 86%, or 219 patients. The researchers found that treatment with ibudilast was associated with a 48% slowing in the rate of decline in brain atrophy as measured by BPF. A per-protocol analysis yielded similar results (P = .045). In addition, no increased rate of serious adverse events and no opportunistic infections or cancer signals were observed. As for tolerability, 25% of subjects on placebo and 30% of those on ibudilast discontinued treatment (P = .30). The main treatment-related adverse events were gastrointestinal in nature (20% in the placebo group vs. 67% in the ibudilast group; P = .002), primarily nausea.
In the analysis of key secondary outcomes, the researchers found that the change in MTR in normal-appearing brain tissue was reduced –0.00558 for ibudilast and –0.03064 for placebo, which was a relative reduction of 82% in MTR decline. At the same time, the change in MTR in normal-appearing gray matter was reduced –0.00753 for ibudilast and –0.03210 for placebo, which was a relative reduction of 77% in MTR decline. No significant difference in white matter changes were observed in transverse diffusivity (P = .15) or longitudinal diffusivity (P = .73), compared with placebo.
For the secondary endpoint of disability, a blinded evaluator assessed Expanded Disability Status Scale (EDSS) score at baseline and every 24 weeks. The researchers used a time-to-event Kaplan-Meier analysis and a Cox proportional hazards model to determine time to confirmed EDSS progression not due to relapse. They found that the hazard ratio for progression in the ibudilast group, compared with the placebo group, was 0.74, with a 90% confidence interval of 0.47-1.17 (P = .29).
“Ibudilast is a novel therapy for progressive MS, with a positive phase 2 study,” Dr. Naismith concluded. “Safety and tolerability were favorable. Clinical endpoints from this trial will be forthcoming.”
The NeuroNEXT Network is supported by the National Institute of Neurological Disorders and Stroke. Dr. Naismith reported having no financial disclosures.
SOURCE: Naismith R et al. ACTRIMS Forum 2018 Abstract P029.
REPORTING FROM ACTRIMS FORUM 2018
Key clinical point: Ibudilast’s positive phase 2 trial results in progressive multiple sclerosis patients bode well for future development.
Major finding: Ibudilast treatment was associated with a 48% slowing in the rate of decline in brain atrophy as measured by brain parenchymal fraction.
Study details: A randomized trial of 255 subjects with progressive MS enrolled at 28 U.S. sites.
Disclosures: The NeuroNEXT Network is supported by the National Institute of Neurological Disorders and Stroke. Dr. Naismith reported having no financial disclosures.
Source: Naismith R et al. ACTRIMS Forum 2018 Abstract P029.
MS may be a transmissible protein misfolding disorder, study suggests
SAN DIEGO – MS may even be caused by prions, potentially putting it into the same category as Creutzfeldt-Jakob disease.
Researchers don’t think MS is contagious between humans. But their findings in mice do suggest that the disease is transmissible from “a seed of protein misfolding,” Shigeki Tsutsui, DVM, PhD, of the University of Calgary (Alta.), said in an interview in advance of his presentation at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
“The next question is: ‘What is actually causing primary damage?’ ” he said. “Our hypothesis is that it’s protein misfolding. If protein misfolding targets the central neuron cells, then those damaged cells release a kind of a trigger to start an immune response.”
This isn’t an unusual concept. Protein misfolding is believed to cause several chronic neurodegenerative diseases, including Parkinson’s, Alzheimer’s, and amyotrophic lateral sclerosis, Dr. Tsutsui said.
The researchers focused on the potential transmissibility of human prion protein “since we thought that the prion protein might be the candidate causing the MS,” he said.
Prion diseases are extremely rare and often deadly. They appear when normal cellular prion proteins are induced to misfold when they come in contact with infectious agents known as prions.
In humans, prion diseases include Creutzfeldt-Jakob disease, fatal familial insomnia, and kuru (a condition spread among cannibals who eat the brains of other humans). Most famously, a variant of Creutzfeldt-Jakob disease has been linked – but not conclusively – to the eating of meat from cows infected with mad cow disease.
Depending on the condition, prion diseases can appear spontaneously, via inheritance or through infection.
In the new study, researchers intracerebrally injected brain homogenate from 10 patients with primary or secondary-progressive MS into 36 transgenic mice. All the mice over-expressed human prion protein. A control group of 23 mice were injected with homogenate from six donor brains.
At 6-12 months, the control mice did not develop pathology. However, the mice injected with brain matter from MS patients developed various levels of significant pathology. The researchers also managed to transmit illness from affected mice into another group of transgenic mice that over-expressed human prion protein via injection of brain homogenate.
While the mice in the study represent “a very extreme case” since their levels of prion protein expression were very high, it’s clear that “MS is actually transmissible,” Dr. Tsutsui said.
The study authors speculate that pathogenic prion protein triggers an autoimmune response by degenerating myelin and causing the release of cellular debris.
Could MS also be contagious? It may not be, even if it’s a prion disease, Dr. Tsutsui said. Some prion disease variants are neither contagious nor infectious.
The researchers plan to study the animal model they’ve created and explore the potential for its use in research, Dr. Tsutsui said.
The authors have no disclosures with the exception of one who disclosed serving as deputy editor of the Multiple Sclerosis Journal. Funding sources include Canada Research Chairs, Alberta Innovates-Health Solutions, and the Alberta Prion Research Institute.
SOURCE: Tsutsui S et al. ACTRIMS Forum 2018 Abstract LB282.
SAN DIEGO – MS may even be caused by prions, potentially putting it into the same category as Creutzfeldt-Jakob disease.
Researchers don’t think MS is contagious between humans. But their findings in mice do suggest that the disease is transmissible from “a seed of protein misfolding,” Shigeki Tsutsui, DVM, PhD, of the University of Calgary (Alta.), said in an interview in advance of his presentation at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
“The next question is: ‘What is actually causing primary damage?’ ” he said. “Our hypothesis is that it’s protein misfolding. If protein misfolding targets the central neuron cells, then those damaged cells release a kind of a trigger to start an immune response.”
This isn’t an unusual concept. Protein misfolding is believed to cause several chronic neurodegenerative diseases, including Parkinson’s, Alzheimer’s, and amyotrophic lateral sclerosis, Dr. Tsutsui said.
The researchers focused on the potential transmissibility of human prion protein “since we thought that the prion protein might be the candidate causing the MS,” he said.
Prion diseases are extremely rare and often deadly. They appear when normal cellular prion proteins are induced to misfold when they come in contact with infectious agents known as prions.
In humans, prion diseases include Creutzfeldt-Jakob disease, fatal familial insomnia, and kuru (a condition spread among cannibals who eat the brains of other humans). Most famously, a variant of Creutzfeldt-Jakob disease has been linked – but not conclusively – to the eating of meat from cows infected with mad cow disease.
Depending on the condition, prion diseases can appear spontaneously, via inheritance or through infection.
In the new study, researchers intracerebrally injected brain homogenate from 10 patients with primary or secondary-progressive MS into 36 transgenic mice. All the mice over-expressed human prion protein. A control group of 23 mice were injected with homogenate from six donor brains.
At 6-12 months, the control mice did not develop pathology. However, the mice injected with brain matter from MS patients developed various levels of significant pathology. The researchers also managed to transmit illness from affected mice into another group of transgenic mice that over-expressed human prion protein via injection of brain homogenate.
While the mice in the study represent “a very extreme case” since their levels of prion protein expression were very high, it’s clear that “MS is actually transmissible,” Dr. Tsutsui said.
The study authors speculate that pathogenic prion protein triggers an autoimmune response by degenerating myelin and causing the release of cellular debris.
Could MS also be contagious? It may not be, even if it’s a prion disease, Dr. Tsutsui said. Some prion disease variants are neither contagious nor infectious.
The researchers plan to study the animal model they’ve created and explore the potential for its use in research, Dr. Tsutsui said.
The authors have no disclosures with the exception of one who disclosed serving as deputy editor of the Multiple Sclerosis Journal. Funding sources include Canada Research Chairs, Alberta Innovates-Health Solutions, and the Alberta Prion Research Institute.
SOURCE: Tsutsui S et al. ACTRIMS Forum 2018 Abstract LB282.
SAN DIEGO – MS may even be caused by prions, potentially putting it into the same category as Creutzfeldt-Jakob disease.
Researchers don’t think MS is contagious between humans. But their findings in mice do suggest that the disease is transmissible from “a seed of protein misfolding,” Shigeki Tsutsui, DVM, PhD, of the University of Calgary (Alta.), said in an interview in advance of his presentation at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
“The next question is: ‘What is actually causing primary damage?’ ” he said. “Our hypothesis is that it’s protein misfolding. If protein misfolding targets the central neuron cells, then those damaged cells release a kind of a trigger to start an immune response.”
This isn’t an unusual concept. Protein misfolding is believed to cause several chronic neurodegenerative diseases, including Parkinson’s, Alzheimer’s, and amyotrophic lateral sclerosis, Dr. Tsutsui said.
The researchers focused on the potential transmissibility of human prion protein “since we thought that the prion protein might be the candidate causing the MS,” he said.
Prion diseases are extremely rare and often deadly. They appear when normal cellular prion proteins are induced to misfold when they come in contact with infectious agents known as prions.
In humans, prion diseases include Creutzfeldt-Jakob disease, fatal familial insomnia, and kuru (a condition spread among cannibals who eat the brains of other humans). Most famously, a variant of Creutzfeldt-Jakob disease has been linked – but not conclusively – to the eating of meat from cows infected with mad cow disease.
Depending on the condition, prion diseases can appear spontaneously, via inheritance or through infection.
In the new study, researchers intracerebrally injected brain homogenate from 10 patients with primary or secondary-progressive MS into 36 transgenic mice. All the mice over-expressed human prion protein. A control group of 23 mice were injected with homogenate from six donor brains.
At 6-12 months, the control mice did not develop pathology. However, the mice injected with brain matter from MS patients developed various levels of significant pathology. The researchers also managed to transmit illness from affected mice into another group of transgenic mice that over-expressed human prion protein via injection of brain homogenate.
While the mice in the study represent “a very extreme case” since their levels of prion protein expression were very high, it’s clear that “MS is actually transmissible,” Dr. Tsutsui said.
The study authors speculate that pathogenic prion protein triggers an autoimmune response by degenerating myelin and causing the release of cellular debris.
Could MS also be contagious? It may not be, even if it’s a prion disease, Dr. Tsutsui said. Some prion disease variants are neither contagious nor infectious.
The researchers plan to study the animal model they’ve created and explore the potential for its use in research, Dr. Tsutsui said.
The authors have no disclosures with the exception of one who disclosed serving as deputy editor of the Multiple Sclerosis Journal. Funding sources include Canada Research Chairs, Alberta Innovates-Health Solutions, and the Alberta Prion Research Institute.
SOURCE: Tsutsui S et al. ACTRIMS Forum 2018 Abstract LB282.
REPORTING FROM ACTRIMS FORUM 2018
Key clinical point: Translational research suggests the primary damage in MS may be caused by transmissible protein misfolding.
Major finding: Transgenic mice over-expressing human prion protein that were injected with brain matter from MS patients developed various levels of significant pathology, and brain homogenate from these mice could transmit illness to naive transgenic mice.
Study details: Transgenic mice over-expressing human prion protein received intracerebral injection of brain homogenate from patients with primary or secondary-progressive MS
Disclosures: The authors have no disclosures with the exception of one who disclosed serving as deputy editor of the Multiple Sclerosis Journal. Funding sources include Canada Research Chairs, Alberta Innovates-Health Solutions, and the Alberta Prion Research Institute.
Source: Tsutsui S et al. ACTRIMS Forum 2018 Abstract LB282.














