User login
Hospital factors play key role in readmission risk after surgery
CORONADO, CALIF. – Variation in readmission risk across hospitals following certain surgical procedures is more attributable to hospital factors than to patient characteristics, results from a large analysis demonstrated.
Such is the impact of the care delivery macro environment (CDM), which Sarah A. Brownlee and coauthors defined as a series of complex interactions between patient characteristics and imposed hospital attributes than can impact patient outcomes postoperatively.
The purpose of the current study was to determine the relative contribution of various aspects of the CDM to 1-year readmission risk after surgery. Working with colleagues Anai Kothari, MD, and Paul Kuo MD, in the One:MAP Section of Clinical informatics and Analytics in the department of surgery at Loyola University Medical Center, Ms. Brownlee analyzed the Healthcare Cost and Utilization Project State Inpatient Databases from Florida, New York, and Washington between 2009 and 2013, which were linked to the American Hospital Association Annual Survey from that same time period.
The researchers used smoothed hazard estimates to determine all-cause readmission in the year after surgery, and multilevel survival models with shared frailty to determine the relative impact of hospital versus patient characteristics on the heterogeneity of readmission risk between hospitals. They limited the analysis to patients aged 18 years and older who underwent one the following procedures: abdominal aortic aneurysm repair, pancreatectomy, colectomy, coronary artery bypass graft, and total hip arthroplasty.
Ms. Brownlee reported results from 502,157 patients who underwent surgical procedures at 347 hospitals. The 1-year readmission rate was 23.5%, and ranged from 12% to 36% across procedures. After controlling for procedure, the researchers observed a 7.9% variation in readmission risk between hospitals. Staffing accounted for 9.8% of variance, followed by hospital structural characteristics such as teaching status and clinical programs (7.5%), patient ZIP code (3.8%), hospital perioperative resources such as inpatient rehab (2.9%), hospital volume (2.8%), and patient clinical characteristics (2.1%). The following hospital characteristics were significantly associated with a lower risk of 1-year readmission: high physician/bed ratio (hazard ratio 0.85; P = .00017); transplant status (HR 0.87; P = .022); high-income ZIP code (HR 0.89; P less than .001); high nurse bed/bed ratio (HR 0.90; P = .047), and cancer center designation (HR 0.93; P = .021).
“Compared to patient clinical characteristics, hospital factors such as staffing ratios, perioperative resources, and structural elements account for more variation in postoperative outcomes,” Ms. Brownlee concluded. “However, it’s important to note that in the present study, over 70% of variation in readmission rates is not explained by the covariates that we analyzed. It’s possible that there are other factors we need to consider. That’s where the direction of this research is going. Much of the variation in readmission risk across hospitals cannot be characterized with currently utilized administrative data.”
The National Institutes of Health provided funding for the study. Ms. Brownlee reported having no financial disclosures.
CORONADO, CALIF. – Variation in readmission risk across hospitals following certain surgical procedures is more attributable to hospital factors than to patient characteristics, results from a large analysis demonstrated.
Such is the impact of the care delivery macro environment (CDM), which Sarah A. Brownlee and coauthors defined as a series of complex interactions between patient characteristics and imposed hospital attributes than can impact patient outcomes postoperatively.
The purpose of the current study was to determine the relative contribution of various aspects of the CDM to 1-year readmission risk after surgery. Working with colleagues Anai Kothari, MD, and Paul Kuo MD, in the One:MAP Section of Clinical informatics and Analytics in the department of surgery at Loyola University Medical Center, Ms. Brownlee analyzed the Healthcare Cost and Utilization Project State Inpatient Databases from Florida, New York, and Washington between 2009 and 2013, which were linked to the American Hospital Association Annual Survey from that same time period.
The researchers used smoothed hazard estimates to determine all-cause readmission in the year after surgery, and multilevel survival models with shared frailty to determine the relative impact of hospital versus patient characteristics on the heterogeneity of readmission risk between hospitals. They limited the analysis to patients aged 18 years and older who underwent one the following procedures: abdominal aortic aneurysm repair, pancreatectomy, colectomy, coronary artery bypass graft, and total hip arthroplasty.
Ms. Brownlee reported results from 502,157 patients who underwent surgical procedures at 347 hospitals. The 1-year readmission rate was 23.5%, and ranged from 12% to 36% across procedures. After controlling for procedure, the researchers observed a 7.9% variation in readmission risk between hospitals. Staffing accounted for 9.8% of variance, followed by hospital structural characteristics such as teaching status and clinical programs (7.5%), patient ZIP code (3.8%), hospital perioperative resources such as inpatient rehab (2.9%), hospital volume (2.8%), and patient clinical characteristics (2.1%). The following hospital characteristics were significantly associated with a lower risk of 1-year readmission: high physician/bed ratio (hazard ratio 0.85; P = .00017); transplant status (HR 0.87; P = .022); high-income ZIP code (HR 0.89; P less than .001); high nurse bed/bed ratio (HR 0.90; P = .047), and cancer center designation (HR 0.93; P = .021).
“Compared to patient clinical characteristics, hospital factors such as staffing ratios, perioperative resources, and structural elements account for more variation in postoperative outcomes,” Ms. Brownlee concluded. “However, it’s important to note that in the present study, over 70% of variation in readmission rates is not explained by the covariates that we analyzed. It’s possible that there are other factors we need to consider. That’s where the direction of this research is going. Much of the variation in readmission risk across hospitals cannot be characterized with currently utilized administrative data.”
The National Institutes of Health provided funding for the study. Ms. Brownlee reported having no financial disclosures.
CORONADO, CALIF. – Variation in readmission risk across hospitals following certain surgical procedures is more attributable to hospital factors than to patient characteristics, results from a large analysis demonstrated.
Such is the impact of the care delivery macro environment (CDM), which Sarah A. Brownlee and coauthors defined as a series of complex interactions between patient characteristics and imposed hospital attributes than can impact patient outcomes postoperatively.
The purpose of the current study was to determine the relative contribution of various aspects of the CDM to 1-year readmission risk after surgery. Working with colleagues Anai Kothari, MD, and Paul Kuo MD, in the One:MAP Section of Clinical informatics and Analytics in the department of surgery at Loyola University Medical Center, Ms. Brownlee analyzed the Healthcare Cost and Utilization Project State Inpatient Databases from Florida, New York, and Washington between 2009 and 2013, which were linked to the American Hospital Association Annual Survey from that same time period.
The researchers used smoothed hazard estimates to determine all-cause readmission in the year after surgery, and multilevel survival models with shared frailty to determine the relative impact of hospital versus patient characteristics on the heterogeneity of readmission risk between hospitals. They limited the analysis to patients aged 18 years and older who underwent one the following procedures: abdominal aortic aneurysm repair, pancreatectomy, colectomy, coronary artery bypass graft, and total hip arthroplasty.
Ms. Brownlee reported results from 502,157 patients who underwent surgical procedures at 347 hospitals. The 1-year readmission rate was 23.5%, and ranged from 12% to 36% across procedures. After controlling for procedure, the researchers observed a 7.9% variation in readmission risk between hospitals. Staffing accounted for 9.8% of variance, followed by hospital structural characteristics such as teaching status and clinical programs (7.5%), patient ZIP code (3.8%), hospital perioperative resources such as inpatient rehab (2.9%), hospital volume (2.8%), and patient clinical characteristics (2.1%). The following hospital characteristics were significantly associated with a lower risk of 1-year readmission: high physician/bed ratio (hazard ratio 0.85; P = .00017); transplant status (HR 0.87; P = .022); high-income ZIP code (HR 0.89; P less than .001); high nurse bed/bed ratio (HR 0.90; P = .047), and cancer center designation (HR 0.93; P = .021).
“Compared to patient clinical characteristics, hospital factors such as staffing ratios, perioperative resources, and structural elements account for more variation in postoperative outcomes,” Ms. Brownlee concluded. “However, it’s important to note that in the present study, over 70% of variation in readmission rates is not explained by the covariates that we analyzed. It’s possible that there are other factors we need to consider. That’s where the direction of this research is going. Much of the variation in readmission risk across hospitals cannot be characterized with currently utilized administrative data.”
The National Institutes of Health provided funding for the study. Ms. Brownlee reported having no financial disclosures.
AT WSA 2016
Key clinical point:
Major finding: Staffing accounted for 9.8% of variance in readmission risk between hospitals, followed by hospital structural characteristics such as teaching status and clinical programs (7.5%).
Data source: Results from 502,157 patients who underwent surgical procedures at 347 hospitals in three states.
Disclosures: The National Institutes of Health provided funding for the study. Ms. Brownlee reported having no financial disclosures.
Complete colpectomy & colpocleisis: Model for simulation

Visit the Society of Gynecologic Surgeons online: sgsonline.org
Related articles:
- Natural orifice sacral colpopexy
- Alternative options for visualizing ureteral patency during intraoperative cystoscopy
- Use of suprapubic Carter-Thomason needle to assist in cystoscopic excision of an intravesical foreign object
- Uterine artery ligation: Advanced techniques and considerations for the difficult laparoscopic hysterectomy
- Cervical injection of methylene blue for identification of sentinel lymph nodes in cervical cancer
- Misplaced hysteroscopic sterilization micro-insert in the peritoneal cavity: A corpus alienum
- Laparoscopic cystectomy for large, bilateral ovarian dermoids
- Small bowel surgery for the benign gynecologist

Visit the Society of Gynecologic Surgeons online: sgsonline.org
Related articles:
- Natural orifice sacral colpopexy
- Alternative options for visualizing ureteral patency during intraoperative cystoscopy
- Use of suprapubic Carter-Thomason needle to assist in cystoscopic excision of an intravesical foreign object
- Uterine artery ligation: Advanced techniques and considerations for the difficult laparoscopic hysterectomy
- Cervical injection of methylene blue for identification of sentinel lymph nodes in cervical cancer
- Misplaced hysteroscopic sterilization micro-insert in the peritoneal cavity: A corpus alienum
- Laparoscopic cystectomy for large, bilateral ovarian dermoids
- Small bowel surgery for the benign gynecologist

Visit the Society of Gynecologic Surgeons online: sgsonline.org
Related articles:
- Natural orifice sacral colpopexy
- Alternative options for visualizing ureteral patency during intraoperative cystoscopy
- Use of suprapubic Carter-Thomason needle to assist in cystoscopic excision of an intravesical foreign object
- Uterine artery ligation: Advanced techniques and considerations for the difficult laparoscopic hysterectomy
- Cervical injection of methylene blue for identification of sentinel lymph nodes in cervical cancer
- Misplaced hysteroscopic sterilization micro-insert in the peritoneal cavity: A corpus alienum
- Laparoscopic cystectomy for large, bilateral ovarian dermoids
- Small bowel surgery for the benign gynecologist
This video is brought to you by
View more videos from SGS here
Low rate of occult uterine malignancy with vaginal morcellation
ORLANDO – The Food and Drug Administration’s 2014 warning that laparoscopic power morcellation during hysterectomy or myomectomy could spread unsuspected cancerous tissue had a chilling effect across the specialty, but what about the risks associated with morcellation during vaginal hysterectomy?
“There is only one case of morcellation during vaginal hysterectomy with a leiomyosarcoma recorded in the literature,” Megan N. Wasson, DO, a fellow in minimally invasive gynecologic surgery at the Mayo Clinic in Phoenix said at the meeting sponsored by AAGL. “It is really unclear if vaginal and electromechanical morcellation carry the same inherent risk.”
To find out more, Dr. Wasson and her colleagues identified 2,296 patients who underwent total vaginal hysterectomy at one of three academic medical centers. A total of 611 of these women had uterine removal with uncontained morcellation via cold-knife wedge resection. The investigators assessed this group for incidence of occult malignancy, perioperative outcomes, and long-term survival in a retrospective cohort study.
Of the 611 women who underwent morcellation during the study, five patients had an occult malignancy, for a rate less than one percent, 0.82%. Three patients had a stage IA, grade I endometrial adenocarcinoma, and two patients had a low-grade stromal sarcoma. No patients had a leiomyosarcoma.
This group of five patients had a mean age of 49 years, a mean BMI of 32 kg/m2 and a median parity of two. Abnormal uterine bleeding was the indication for surgery for all five patients with a malignancy. The mean uterine weight was elevated at 231 g. One patient with endometrial adenocarcinoma later underwent pelvic lymphadenectomy and vaginal brachytherapy.
“So far, thankfully, all of these patients show no evidence of disease recurrence,” Dr. Wasson said. All five patients are alive, with a mean disease-free survival of 43 months among those with endometrial adenocarcinoma and 37 months for the low-grade stroma sarcoma patients.
“Overall, the incidence of occult uterine carcinoma at the time of vaginal hysterectomy is less than 1%,” Dr. Wasson said. “Thankfully, it does not appear to have a negative effect on patient outcomes when it occurs.”
More research is needed, however. “The risk is very limited in terms of what we know,” she said. “We investigated cancer in this study, but there is also a risk of dissemination of benign conditions.”
All patients underwent a preoperative evaluation that included sampling of the lining of the uterus and imaging. “Out of the five patients with carcinomas, two of the adenocarcinomas had completely benign preoperative sampling and one had hyperplasia, which unfortunately did develop into occult disease,” Dr. Wasson said. “We wouldn’t recommend morcellating any patient with hyperplasia. In the two patients with low-grade stromal sarcoma, neither had any hyperplasia on preoperative sampling.”
Following the 2014 FDA Safety Communication on power morcellation, the AAGL released its own guidance on morcellation during uterine tissue extraction. The AAGL recommended that clinicians avoid morcellation for any patient who had a premalignant or malignant condition or who was at risk for malignancy, and use caution when considering morcellation. “This was for all types of morcellation, including electromechanical and vaginal morcellation,” Dr. Wasson said.
“This was in response to studies and awareness of increased risk of disease with morcellation – specifically leiomyosarcomas – for dissemination of disease in the abdomen and pelvis, but also for an increased risk of recurrence,” she said. “This means, in turn, that patients can have decreased overall survival and disease-free survival, so this is very important when we are talking to our patients.”
Dr. Wasson reported having no relevant financial disclosures.
ORLANDO – The Food and Drug Administration’s 2014 warning that laparoscopic power morcellation during hysterectomy or myomectomy could spread unsuspected cancerous tissue had a chilling effect across the specialty, but what about the risks associated with morcellation during vaginal hysterectomy?
“There is only one case of morcellation during vaginal hysterectomy with a leiomyosarcoma recorded in the literature,” Megan N. Wasson, DO, a fellow in minimally invasive gynecologic surgery at the Mayo Clinic in Phoenix said at the meeting sponsored by AAGL. “It is really unclear if vaginal and electromechanical morcellation carry the same inherent risk.”
To find out more, Dr. Wasson and her colleagues identified 2,296 patients who underwent total vaginal hysterectomy at one of three academic medical centers. A total of 611 of these women had uterine removal with uncontained morcellation via cold-knife wedge resection. The investigators assessed this group for incidence of occult malignancy, perioperative outcomes, and long-term survival in a retrospective cohort study.
Of the 611 women who underwent morcellation during the study, five patients had an occult malignancy, for a rate less than one percent, 0.82%. Three patients had a stage IA, grade I endometrial adenocarcinoma, and two patients had a low-grade stromal sarcoma. No patients had a leiomyosarcoma.
This group of five patients had a mean age of 49 years, a mean BMI of 32 kg/m2 and a median parity of two. Abnormal uterine bleeding was the indication for surgery for all five patients with a malignancy. The mean uterine weight was elevated at 231 g. One patient with endometrial adenocarcinoma later underwent pelvic lymphadenectomy and vaginal brachytherapy.
“So far, thankfully, all of these patients show no evidence of disease recurrence,” Dr. Wasson said. All five patients are alive, with a mean disease-free survival of 43 months among those with endometrial adenocarcinoma and 37 months for the low-grade stroma sarcoma patients.
“Overall, the incidence of occult uterine carcinoma at the time of vaginal hysterectomy is less than 1%,” Dr. Wasson said. “Thankfully, it does not appear to have a negative effect on patient outcomes when it occurs.”
More research is needed, however. “The risk is very limited in terms of what we know,” she said. “We investigated cancer in this study, but there is also a risk of dissemination of benign conditions.”
All patients underwent a preoperative evaluation that included sampling of the lining of the uterus and imaging. “Out of the five patients with carcinomas, two of the adenocarcinomas had completely benign preoperative sampling and one had hyperplasia, which unfortunately did develop into occult disease,” Dr. Wasson said. “We wouldn’t recommend morcellating any patient with hyperplasia. In the two patients with low-grade stromal sarcoma, neither had any hyperplasia on preoperative sampling.”
Following the 2014 FDA Safety Communication on power morcellation, the AAGL released its own guidance on morcellation during uterine tissue extraction. The AAGL recommended that clinicians avoid morcellation for any patient who had a premalignant or malignant condition or who was at risk for malignancy, and use caution when considering morcellation. “This was for all types of morcellation, including electromechanical and vaginal morcellation,” Dr. Wasson said.
“This was in response to studies and awareness of increased risk of disease with morcellation – specifically leiomyosarcomas – for dissemination of disease in the abdomen and pelvis, but also for an increased risk of recurrence,” she said. “This means, in turn, that patients can have decreased overall survival and disease-free survival, so this is very important when we are talking to our patients.”
Dr. Wasson reported having no relevant financial disclosures.
ORLANDO – The Food and Drug Administration’s 2014 warning that laparoscopic power morcellation during hysterectomy or myomectomy could spread unsuspected cancerous tissue had a chilling effect across the specialty, but what about the risks associated with morcellation during vaginal hysterectomy?
“There is only one case of morcellation during vaginal hysterectomy with a leiomyosarcoma recorded in the literature,” Megan N. Wasson, DO, a fellow in minimally invasive gynecologic surgery at the Mayo Clinic in Phoenix said at the meeting sponsored by AAGL. “It is really unclear if vaginal and electromechanical morcellation carry the same inherent risk.”
To find out more, Dr. Wasson and her colleagues identified 2,296 patients who underwent total vaginal hysterectomy at one of three academic medical centers. A total of 611 of these women had uterine removal with uncontained morcellation via cold-knife wedge resection. The investigators assessed this group for incidence of occult malignancy, perioperative outcomes, and long-term survival in a retrospective cohort study.
Of the 611 women who underwent morcellation during the study, five patients had an occult malignancy, for a rate less than one percent, 0.82%. Three patients had a stage IA, grade I endometrial adenocarcinoma, and two patients had a low-grade stromal sarcoma. No patients had a leiomyosarcoma.
This group of five patients had a mean age of 49 years, a mean BMI of 32 kg/m2 and a median parity of two. Abnormal uterine bleeding was the indication for surgery for all five patients with a malignancy. The mean uterine weight was elevated at 231 g. One patient with endometrial adenocarcinoma later underwent pelvic lymphadenectomy and vaginal brachytherapy.
“So far, thankfully, all of these patients show no evidence of disease recurrence,” Dr. Wasson said. All five patients are alive, with a mean disease-free survival of 43 months among those with endometrial adenocarcinoma and 37 months for the low-grade stroma sarcoma patients.
“Overall, the incidence of occult uterine carcinoma at the time of vaginal hysterectomy is less than 1%,” Dr. Wasson said. “Thankfully, it does not appear to have a negative effect on patient outcomes when it occurs.”
More research is needed, however. “The risk is very limited in terms of what we know,” she said. “We investigated cancer in this study, but there is also a risk of dissemination of benign conditions.”
All patients underwent a preoperative evaluation that included sampling of the lining of the uterus and imaging. “Out of the five patients with carcinomas, two of the adenocarcinomas had completely benign preoperative sampling and one had hyperplasia, which unfortunately did develop into occult disease,” Dr. Wasson said. “We wouldn’t recommend morcellating any patient with hyperplasia. In the two patients with low-grade stromal sarcoma, neither had any hyperplasia on preoperative sampling.”
Following the 2014 FDA Safety Communication on power morcellation, the AAGL released its own guidance on morcellation during uterine tissue extraction. The AAGL recommended that clinicians avoid morcellation for any patient who had a premalignant or malignant condition or who was at risk for malignancy, and use caution when considering morcellation. “This was for all types of morcellation, including electromechanical and vaginal morcellation,” Dr. Wasson said.
“This was in response to studies and awareness of increased risk of disease with morcellation – specifically leiomyosarcomas – for dissemination of disease in the abdomen and pelvis, but also for an increased risk of recurrence,” she said. “This means, in turn, that patients can have decreased overall survival and disease-free survival, so this is very important when we are talking to our patients.”
Dr. Wasson reported having no relevant financial disclosures.
AT THE AAGL GLOBAL CONGRESS
Key clinical point:
Major finding: Of 611 patients who underwent morcellation during total vaginal hysterectomy, five patients (0.82%) had occult uterine carcinoma.
Data source: A retrospective cohort study of 611 women who had uterine removal with uncontained morcellation.
Disclosures: Dr. Wasson reported having no relevant financial disclosures.
Should surgeons change gloves during total laparoscopic hysterectomy?
ORLANDO – Many gynecologic surgeons change gloves, gowns, and even surgical drapes during total laparoscopic hysterectomy to prevent bacterial infections, but little data support the practice.
In a small study of women undergoing total laparoscopic hysterectomy, investigators found that the overall risk of infection from contaminated gowns, gloves, and instruments was very low, with bacterial growth below the infection threshold in 98.9% of samples and no surgical site infections reported during 6 weeks of follow-up after surgery.
“Tradition dictates that even after both fields have been prepped, we refer to the perineum and vagina as ‘dirty,’ and the abdomen as ‘clean,’ ” Dr. Shockley said, “And surgeons habitually change their gown and gloves when inadvertent contact with the perineum or vagina occurs.”
To elucidate the true pathogen picture, Dr. Shockley and her colleagues assessed 31 women undergoing total laparoscopic hysterectomy for a benign indication during 2016. They evaluated the type and quantity of bacteria found intraoperatively on the abdomen, vagina, surgical gloves, instrument tips, and uterus.
All patients received perioperative antibiotic prophylaxis and standard, separate perineovaginal and abdominal prep with chlorhexidine. Investigators swabbed the vaginal fornices and abdomen at six sites, as well as the surgeon’s gloves following placement of the uterine manipulator, tips of instruments used to close the vaginal cuff, uterine fundus after extraction, and surgeon’s gloves following removal of the uterus.
They detected no anaerobic bacterial growth from samples taken from the abdomen, in the vagina, or on the tips of instruments used for cuff closure. Similarly, there was no aerobic growth observed in the vagina of any patient. However, they did detect aerobic bacterial growth in the abdomen, which in all cases was consistent with skin flora.
Three patients demonstrated some growth with the surgeon’s gloves following manipulator placement. Nearly one-third – 32% – of surgeon’s gloves cultured bacteria after removal of the uterus. One sample yielded cumulative growth for a bacterial count considered high enough to potentially cause infection, defined as more than 5,000 colony-forming units (CFU) per milliliter. This was the highest growth sample out of the 180 samples collected.
Additionally, 39% of samples from the uterine fundus were positive, a higher percentage than at any other site, Dr. Shockley reported. “And the one sample with growth exceeding 5,000 CFU/mL – you guessed it – was from the same patient.”
Bacterial growth was scant on the instrument tips used to close the vaginal cuffs.
Overall, bacterial growth in 98.9% of samples was below the infection threshold. “We did not identify any post–surgical site infections during 6 weeks of follow-up,” Dr. Shockley said at the meeting sponsored by AAGL.
“This study does provide a good description and count of the bacteria encountered during total laparoscopic hysterectomy. They are unlikely to cause surgical site infections … but based on concentration and frequency of bacterial growth on the surgeon’s gloves after specimen extraction, we would recommend if you are going to change gloves, do it after this step, before turning your attention back to the abdomen for vaginal cuff closure,” she said.
But changing gloves after placing the Foley and uterine manipulator “seems to be a wasted exercise,” Dr. Shockley said. “There was no growth on the vaginal fornices of any patient.”
The bacteria on the gloves in those three cases developed very low colony counts. “Yes, there was growth after the removal of the specimen, but with the exception of one patient, the colony counts were all below 5,000,” she said. “I think we need more data to reassure ourselves [attire changes are] unnecessary at every step of the [total laparoscopic hysterectomy].”
The study was supported by an educational grant from the Foundation of the AAGL Jerome J. Hoffman Endowment. Dr. Shockley reported having no relevant financial disclosures.
ORLANDO – Many gynecologic surgeons change gloves, gowns, and even surgical drapes during total laparoscopic hysterectomy to prevent bacterial infections, but little data support the practice.
In a small study of women undergoing total laparoscopic hysterectomy, investigators found that the overall risk of infection from contaminated gowns, gloves, and instruments was very low, with bacterial growth below the infection threshold in 98.9% of samples and no surgical site infections reported during 6 weeks of follow-up after surgery.
“Tradition dictates that even after both fields have been prepped, we refer to the perineum and vagina as ‘dirty,’ and the abdomen as ‘clean,’ ” Dr. Shockley said, “And surgeons habitually change their gown and gloves when inadvertent contact with the perineum or vagina occurs.”
To elucidate the true pathogen picture, Dr. Shockley and her colleagues assessed 31 women undergoing total laparoscopic hysterectomy for a benign indication during 2016. They evaluated the type and quantity of bacteria found intraoperatively on the abdomen, vagina, surgical gloves, instrument tips, and uterus.
All patients received perioperative antibiotic prophylaxis and standard, separate perineovaginal and abdominal prep with chlorhexidine. Investigators swabbed the vaginal fornices and abdomen at six sites, as well as the surgeon’s gloves following placement of the uterine manipulator, tips of instruments used to close the vaginal cuff, uterine fundus after extraction, and surgeon’s gloves following removal of the uterus.
They detected no anaerobic bacterial growth from samples taken from the abdomen, in the vagina, or on the tips of instruments used for cuff closure. Similarly, there was no aerobic growth observed in the vagina of any patient. However, they did detect aerobic bacterial growth in the abdomen, which in all cases was consistent with skin flora.
Three patients demonstrated some growth with the surgeon’s gloves following manipulator placement. Nearly one-third – 32% – of surgeon’s gloves cultured bacteria after removal of the uterus. One sample yielded cumulative growth for a bacterial count considered high enough to potentially cause infection, defined as more than 5,000 colony-forming units (CFU) per milliliter. This was the highest growth sample out of the 180 samples collected.
Additionally, 39% of samples from the uterine fundus were positive, a higher percentage than at any other site, Dr. Shockley reported. “And the one sample with growth exceeding 5,000 CFU/mL – you guessed it – was from the same patient.”
Bacterial growth was scant on the instrument tips used to close the vaginal cuffs.
Overall, bacterial growth in 98.9% of samples was below the infection threshold. “We did not identify any post–surgical site infections during 6 weeks of follow-up,” Dr. Shockley said at the meeting sponsored by AAGL.
“This study does provide a good description and count of the bacteria encountered during total laparoscopic hysterectomy. They are unlikely to cause surgical site infections … but based on concentration and frequency of bacterial growth on the surgeon’s gloves after specimen extraction, we would recommend if you are going to change gloves, do it after this step, before turning your attention back to the abdomen for vaginal cuff closure,” she said.
But changing gloves after placing the Foley and uterine manipulator “seems to be a wasted exercise,” Dr. Shockley said. “There was no growth on the vaginal fornices of any patient.”
The bacteria on the gloves in those three cases developed very low colony counts. “Yes, there was growth after the removal of the specimen, but with the exception of one patient, the colony counts were all below 5,000,” she said. “I think we need more data to reassure ourselves [attire changes are] unnecessary at every step of the [total laparoscopic hysterectomy].”
The study was supported by an educational grant from the Foundation of the AAGL Jerome J. Hoffman Endowment. Dr. Shockley reported having no relevant financial disclosures.
ORLANDO – Many gynecologic surgeons change gloves, gowns, and even surgical drapes during total laparoscopic hysterectomy to prevent bacterial infections, but little data support the practice.
In a small study of women undergoing total laparoscopic hysterectomy, investigators found that the overall risk of infection from contaminated gowns, gloves, and instruments was very low, with bacterial growth below the infection threshold in 98.9% of samples and no surgical site infections reported during 6 weeks of follow-up after surgery.
“Tradition dictates that even after both fields have been prepped, we refer to the perineum and vagina as ‘dirty,’ and the abdomen as ‘clean,’ ” Dr. Shockley said, “And surgeons habitually change their gown and gloves when inadvertent contact with the perineum or vagina occurs.”
To elucidate the true pathogen picture, Dr. Shockley and her colleagues assessed 31 women undergoing total laparoscopic hysterectomy for a benign indication during 2016. They evaluated the type and quantity of bacteria found intraoperatively on the abdomen, vagina, surgical gloves, instrument tips, and uterus.
All patients received perioperative antibiotic prophylaxis and standard, separate perineovaginal and abdominal prep with chlorhexidine. Investigators swabbed the vaginal fornices and abdomen at six sites, as well as the surgeon’s gloves following placement of the uterine manipulator, tips of instruments used to close the vaginal cuff, uterine fundus after extraction, and surgeon’s gloves following removal of the uterus.
They detected no anaerobic bacterial growth from samples taken from the abdomen, in the vagina, or on the tips of instruments used for cuff closure. Similarly, there was no aerobic growth observed in the vagina of any patient. However, they did detect aerobic bacterial growth in the abdomen, which in all cases was consistent with skin flora.
Three patients demonstrated some growth with the surgeon’s gloves following manipulator placement. Nearly one-third – 32% – of surgeon’s gloves cultured bacteria after removal of the uterus. One sample yielded cumulative growth for a bacterial count considered high enough to potentially cause infection, defined as more than 5,000 colony-forming units (CFU) per milliliter. This was the highest growth sample out of the 180 samples collected.
Additionally, 39% of samples from the uterine fundus were positive, a higher percentage than at any other site, Dr. Shockley reported. “And the one sample with growth exceeding 5,000 CFU/mL – you guessed it – was from the same patient.”
Bacterial growth was scant on the instrument tips used to close the vaginal cuffs.
Overall, bacterial growth in 98.9% of samples was below the infection threshold. “We did not identify any post–surgical site infections during 6 weeks of follow-up,” Dr. Shockley said at the meeting sponsored by AAGL.
“This study does provide a good description and count of the bacteria encountered during total laparoscopic hysterectomy. They are unlikely to cause surgical site infections … but based on concentration and frequency of bacterial growth on the surgeon’s gloves after specimen extraction, we would recommend if you are going to change gloves, do it after this step, before turning your attention back to the abdomen for vaginal cuff closure,” she said.
But changing gloves after placing the Foley and uterine manipulator “seems to be a wasted exercise,” Dr. Shockley said. “There was no growth on the vaginal fornices of any patient.”
The bacteria on the gloves in those three cases developed very low colony counts. “Yes, there was growth after the removal of the specimen, but with the exception of one patient, the colony counts were all below 5,000,” she said. “I think we need more data to reassure ourselves [attire changes are] unnecessary at every step of the [total laparoscopic hysterectomy].”
The study was supported by an educational grant from the Foundation of the AAGL Jerome J. Hoffman Endowment. Dr. Shockley reported having no relevant financial disclosures.
AT THE AAGL GLOBAL CONGRESS
Key clinical point:
Major finding: Bacterial concentrations did not exceed thresholds required to trigger potential infection in almost 99% of cultures.
Data source: A study of 31 women undergoing total laparoscopic hysterectomy for benign indications in 2016.
Disclosures: The study was supported by an educational grant from the Foundation of the AAGL Jerome J. Hoffman Endowment. Dr. Shockley reported having no relevant financial disclosures.
Risk-reducing salpingectomy at benign hysterectomy: Have surgeons embraced this practice?
According to its January 2015 Committee Opinion, the American College of Obstetricians and Gynecologists supported the following recommendations and conclusions regarding salpingectomy for ovarian cancer prevention1:
- The surgeon and patient should discuss the potential benefits of the removal of the fallopian tubes during a hysterectomy in women at population risk of ovarian cancer who are not having an oophorectomy.
- When counseling women about laparoscopic sterilization methods, clinicians can communicate that bilateral salpingectomy can be considered a method that provides effective contraception.
- Prophylactic salpingectomy may offer clinicians the opportunity to prevent ovarian cancer in their patients.
- Randomized controlled trials are needed to support the validity of this approach to reduce the incidence of ovarian cancer.
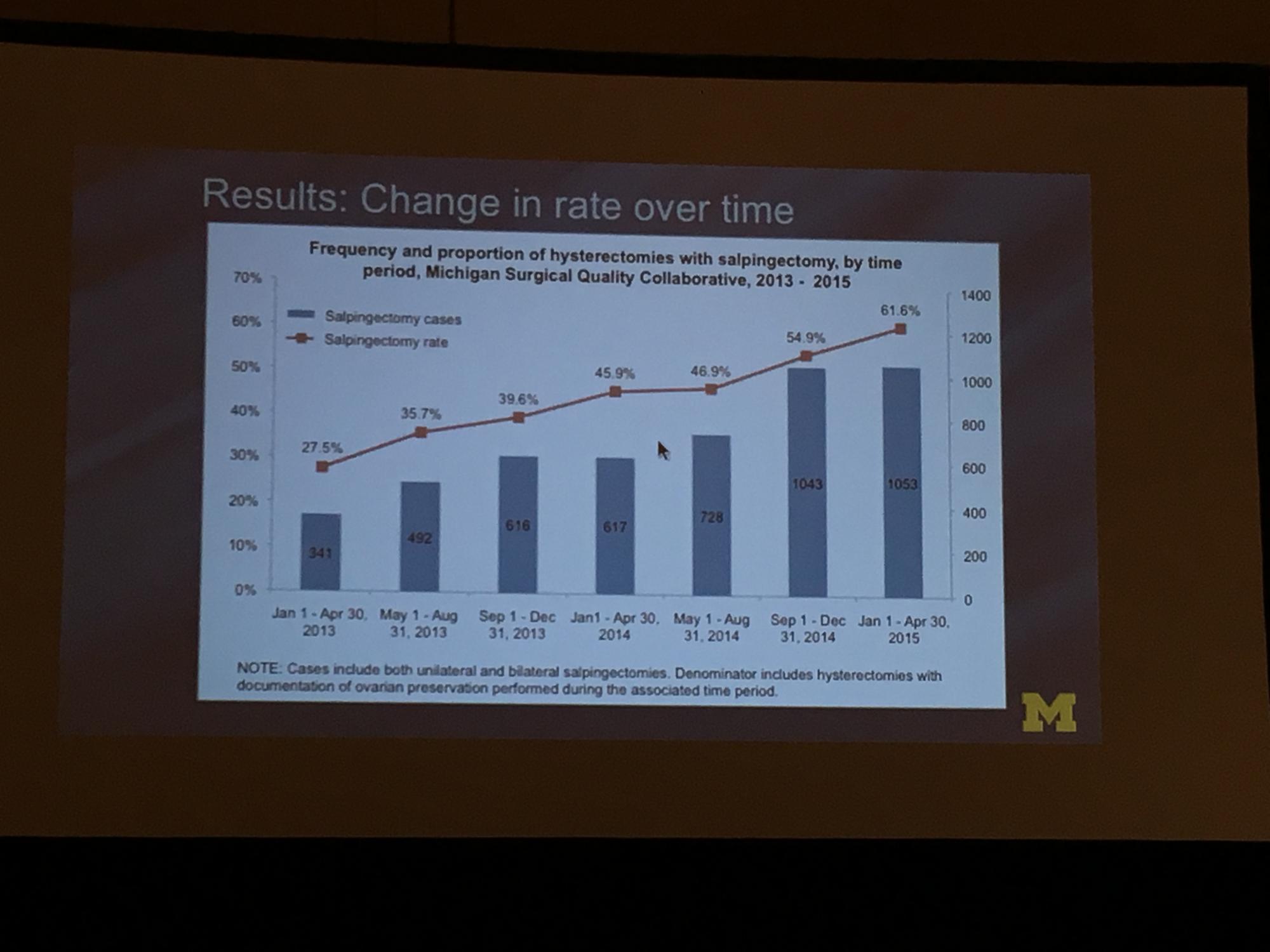
To determine the change in rate of salpingectomy performed at benign hysterectomy at Michigan hospitals, Sara Till, MD, MPH, and colleagues from the University of Michigan Health System performed a retrospective cross-sectioned study of data from the Michigan Surgical Quality Collaborative. They examined hysterectomies performed for all surgical routes between January 2013 and April 2015. Exclusion criteria included malignancy and obstetric indication. The primary objective was to measure salpingectomy at the time of hysterectomy with ovarian preservation. Measures studied included demographics; comorbidities; perioperative and postoperative results; and hospital/surgeon-related data; including surgeon volume, hospital type (ie, teaching), and hospital size.2
During the study period (January 1, 2013, to April 30, 2015), 18,642 hysterectomies were performed for benign indications, of which 55.7% (n = 10,382) were ovarian conserving. Among patients who underwent ovarian conserving hysterectomy, 44.9% (n = 4,668) had salpingectomy, with rates increasing steadily from 26.4% to 61.1% across the study period (P<.001). Salpingectomy was more likely with a laparoscopic approach (odds ratio [OR], 2.93; 95% confidence interval [CI], 2.69–3.20) and among women aged <60 years (OR, 2.60; 95% CI, 1.42–1.98), but did not vary with surgeon volume. After adjustments for age, body mass index, and surgical approach using a mixed model, the researchers found substantial variation in rates of salpingectomy across hospital sites, ranging from 3.7% to 88.3%. Variation in adjusted salpingectomy rates was not associated with academic affiliation or hospital size.2
Dr. Till and colleagues concluded that there was a substantial rise in risk-reducing salpingectomy from January 1, 2013, to April 30, 2015, and that there is substantial variation in the practice of salpingectomy, which is not accounted for by patient, surgeon, or hospital characteristics.2
- American College of Obstetricians and Gynecologists, Committee on Gynecologic Practice. Salpingectomy for ovarian cancer prevention. Committee Opinion No. 620 [published correction appears in: Obstet Gynecol. 2016;127(2):405]. Obstet Gynecol. 2015;125(1):279–281.
- Till SR, Edwards MG, Kobernik EK, Kamdar NS, As-Sanie S, Morgan DM. Implementation rate of risk-reducing salpingectomy at time of benign hysterectomy. Poster presented at: AAGL Global Congress of Minimally Invasive Gynecology; November 16, 2016; Orlando, Florida. J Minim Invasiv Gynecol. 2016;23(7 suppl):S1.
According to its January 2015 Committee Opinion, the American College of Obstetricians and Gynecologists supported the following recommendations and conclusions regarding salpingectomy for ovarian cancer prevention1:
- The surgeon and patient should discuss the potential benefits of the removal of the fallopian tubes during a hysterectomy in women at population risk of ovarian cancer who are not having an oophorectomy.
- When counseling women about laparoscopic sterilization methods, clinicians can communicate that bilateral salpingectomy can be considered a method that provides effective contraception.
- Prophylactic salpingectomy may offer clinicians the opportunity to prevent ovarian cancer in their patients.
- Randomized controlled trials are needed to support the validity of this approach to reduce the incidence of ovarian cancer.
To determine the change in rate of salpingectomy performed at benign hysterectomy at Michigan hospitals, Sara Till, MD, MPH, and colleagues from the University of Michigan Health System performed a retrospective cross-sectioned study of data from the Michigan Surgical Quality Collaborative. They examined hysterectomies performed for all surgical routes between January 2013 and April 2015. Exclusion criteria included malignancy and obstetric indication. The primary objective was to measure salpingectomy at the time of hysterectomy with ovarian preservation. Measures studied included demographics; comorbidities; perioperative and postoperative results; and hospital/surgeon-related data; including surgeon volume, hospital type (ie, teaching), and hospital size.2
During the study period (January 1, 2013, to April 30, 2015), 18,642 hysterectomies were performed for benign indications, of which 55.7% (n = 10,382) were ovarian conserving. Among patients who underwent ovarian conserving hysterectomy, 44.9% (n = 4,668) had salpingectomy, with rates increasing steadily from 26.4% to 61.1% across the study period (P<.001). Salpingectomy was more likely with a laparoscopic approach (odds ratio [OR], 2.93; 95% confidence interval [CI], 2.69–3.20) and among women aged <60 years (OR, 2.60; 95% CI, 1.42–1.98), but did not vary with surgeon volume. After adjustments for age, body mass index, and surgical approach using a mixed model, the researchers found substantial variation in rates of salpingectomy across hospital sites, ranging from 3.7% to 88.3%. Variation in adjusted salpingectomy rates was not associated with academic affiliation or hospital size.2
Dr. Till and colleagues concluded that there was a substantial rise in risk-reducing salpingectomy from January 1, 2013, to April 30, 2015, and that there is substantial variation in the practice of salpingectomy, which is not accounted for by patient, surgeon, or hospital characteristics.2
According to its January 2015 Committee Opinion, the American College of Obstetricians and Gynecologists supported the following recommendations and conclusions regarding salpingectomy for ovarian cancer prevention1:
- The surgeon and patient should discuss the potential benefits of the removal of the fallopian tubes during a hysterectomy in women at population risk of ovarian cancer who are not having an oophorectomy.
- When counseling women about laparoscopic sterilization methods, clinicians can communicate that bilateral salpingectomy can be considered a method that provides effective contraception.
- Prophylactic salpingectomy may offer clinicians the opportunity to prevent ovarian cancer in their patients.
- Randomized controlled trials are needed to support the validity of this approach to reduce the incidence of ovarian cancer.
To determine the change in rate of salpingectomy performed at benign hysterectomy at Michigan hospitals, Sara Till, MD, MPH, and colleagues from the University of Michigan Health System performed a retrospective cross-sectioned study of data from the Michigan Surgical Quality Collaborative. They examined hysterectomies performed for all surgical routes between January 2013 and April 2015. Exclusion criteria included malignancy and obstetric indication. The primary objective was to measure salpingectomy at the time of hysterectomy with ovarian preservation. Measures studied included demographics; comorbidities; perioperative and postoperative results; and hospital/surgeon-related data; including surgeon volume, hospital type (ie, teaching), and hospital size.2
During the study period (January 1, 2013, to April 30, 2015), 18,642 hysterectomies were performed for benign indications, of which 55.7% (n = 10,382) were ovarian conserving. Among patients who underwent ovarian conserving hysterectomy, 44.9% (n = 4,668) had salpingectomy, with rates increasing steadily from 26.4% to 61.1% across the study period (P<.001). Salpingectomy was more likely with a laparoscopic approach (odds ratio [OR], 2.93; 95% confidence interval [CI], 2.69–3.20) and among women aged <60 years (OR, 2.60; 95% CI, 1.42–1.98), but did not vary with surgeon volume. After adjustments for age, body mass index, and surgical approach using a mixed model, the researchers found substantial variation in rates of salpingectomy across hospital sites, ranging from 3.7% to 88.3%. Variation in adjusted salpingectomy rates was not associated with academic affiliation or hospital size.2
Dr. Till and colleagues concluded that there was a substantial rise in risk-reducing salpingectomy from January 1, 2013, to April 30, 2015, and that there is substantial variation in the practice of salpingectomy, which is not accounted for by patient, surgeon, or hospital characteristics.2
- American College of Obstetricians and Gynecologists, Committee on Gynecologic Practice. Salpingectomy for ovarian cancer prevention. Committee Opinion No. 620 [published correction appears in: Obstet Gynecol. 2016;127(2):405]. Obstet Gynecol. 2015;125(1):279–281.
- Till SR, Edwards MG, Kobernik EK, Kamdar NS, As-Sanie S, Morgan DM. Implementation rate of risk-reducing salpingectomy at time of benign hysterectomy. Poster presented at: AAGL Global Congress of Minimally Invasive Gynecology; November 16, 2016; Orlando, Florida. J Minim Invasiv Gynecol. 2016;23(7 suppl):S1.
- American College of Obstetricians and Gynecologists, Committee on Gynecologic Practice. Salpingectomy for ovarian cancer prevention. Committee Opinion No. 620 [published correction appears in: Obstet Gynecol. 2016;127(2):405]. Obstet Gynecol. 2015;125(1):279–281.
- Till SR, Edwards MG, Kobernik EK, Kamdar NS, As-Sanie S, Morgan DM. Implementation rate of risk-reducing salpingectomy at time of benign hysterectomy. Poster presented at: AAGL Global Congress of Minimally Invasive Gynecology; November 16, 2016; Orlando, Florida. J Minim Invasiv Gynecol. 2016;23(7 suppl):S1.
Risk stratification important for aortic valve disease in pregnancy
CHICAGO – Pregnancy-associated hemodynamic and physiologic changes can challenge hearts with diseased aortic valves. Understanding pregnancy’s toll on the body can help physicians take better care of pregnant women with aortic stenosis or aortic regurgitation, said Patrick T. O’Gara, MD. “This is a relatively infrequent condition, and it causes all of us a great deal of apprehension,” he said.
Dr. O’Gara, director of clinical cardiology at Brigham and Women’s Hospital, Boston, walked attendees through current recommendations for caring for pregnant women with aortic valve disease in a presentation at the annual Heart Valve Summit.
When talking to women with aortic valve disease, it’s important to let them know that a pregnancy during which they experience an adverse maternal cardiac event appears to increase their risk for later events as well, said Dr. O’Gara. One study of pregnancy in women with aortic valve disease, said Dr. O’Gara, found that the risk of later events for women who had had a cardiac event in pregnancy was 27% plus or minus 9 percentage points in the 5 years following pregnancy, while the risk was just 15% plus or minus 3 percentage points for women whose pregnancies were not complicated by cardiac adverse events (P = .02) (Heart. 2010 Oct;96(20):1656-61).
The World Health Organization classification of pregnancy risk, said Dr. O’Gara, provides some guidance. Patients who are WHO class III are considered to be high risk; pregnancy in these individuals calls for counseling and a multidisciplinary care team. Patients who fall into this class are those with mechanical heart valves, those with Marfan syndrome and aortic involvement with a valve diameter of 40-45 mm, and those with bicuspid aortic valve disease with a valve diameter of 45-50 mm.
WHO class IV patients, according to the guidelines, are at prohibitive risk. These patients should consider terminating a pregnancy if one occurs. Conditions that place women into WHO class IV include symptomatic severe aortic stenosis (AS), severe left ventricular dysfunction with an ejection fraction less than 40%, and severe coarctation of the aorta. Women with Marfan syndrome with aortic valve diameters greater than 45 mm and women with bicuspid aortic valves and a diameter over 50 mm also fall into this category of prohibitive risk.
If a physician is lucky enough to have a pre-conception relationship with a patient, then a host of considerations can come into play. In addition to cardiac risk stratification, obstetric and intrapartum risk to the mother as well as neonatal risk should be considered. Some factors to take into account are maternal life expectancy and potential long-term complications to the mother from pregnancy and childbirth.
Plasma volume increases rapidly through the first trimester, plateauing about halfway through pregnancy. With this normal dilutional anemia, “Most normal pregnancies are accompanied by a grade II systolic murmur, best heard at the upper left sternal border,” said Dr. O’Gara. Since stroke volume and heart rate both also increase through pregnancy, cardiac output goes up as well.
“Importantly, systemic vascular resistance is reduced throughout pregnancy,” said Dr. O’Gara. Consequently, “regurgitant heart valve problems are generally much better tolerated than stenotic lesions in pregnancy. That’s physiologic principle number one.”
In labor, abrupt hemodynamic changes include increases in cardiac output, heart rate, blood pressure, and venous return. “Labor and delivery are very stressful hemodynamic times for women,” said Dr. O’Gara.
In the immediate postpartum period, uterine contraction results in a return of a significant amount of blood to the maternal circulation. This effectively creates an autotransfusion, with resulting increased preload and cardiac output. The inferior vena cava is also freed from the pressure of a gravid uterus, increasing venous return further. Finally, there’s also a marked increase in systemic vascular resistance.
Aside from the risk of adverse cardiac events, other adverse outcomes for women can include an increased risk of premature rupture of membranes and postpartum hemorrhage. For the neonate, preterm birth and respiratory distress are more likely. Newborns are more likely to be small for gestational age, to suffer intraventricular hemorrhage, and to die. Depending on the parental genetic status, the infant may be at risk of congenital heart disease as well.
Neonatal risk may also be increased, said Dr. O’Gara. Some maternal conditions that can increase risk for the infant include a baseline New York Heart Association (NYHA) class of greater than II or having cyanosis; having a left heart obstruction; being a smoker; having a multiple gestation; having a mechanical heart valve; and taking an oral anticoagulant.
Ideally, a full discussion would include a genetic referral if indicated, as well as coordination with a gynecologist or primary care provider to provide contraception and to assist with planning for and optimizing outcomes of a pregnancy.
The American College of Cardiology and the American Heart Association have issued guidelines that include a class I recommendation regarding valve intervention before pregnancy for symptomatic patients with severe AS (those with aortic velocity of at least 4.0 m/sec, or mean pressure gradient greater than 41 mm Hg; stage D). For patients who meet the same hemodynamic criteria for severe AS but who are asymptomatic (stage C), the guidelines have a class IIa recommendation that pre-pregnancy valve intervention is “reasonable.” These recommendations have level C evidence supporting them.
However, another class IIa recommendation for patients with severe AS who are already pregnant advises valve intervention only in the case of hemodynamic deterioration, or the development of NYHA class III to IV heart failure symptoms. This is backed by grade B evidence, according to the associations.
For aortic regurgitation (AR), a level I recommendation advises valve repair or replacement before pregnancy if patients are symptomatic and have severe (stage D) regurgitation. Pregnant patients with severe regurgitation should only be considered for a valve operation if they have refractory NYHA class IV heart failure symptoms. These recommendations have level C evidence supporting them.
Since AR is better tolerated in pregnancy than AS, the nature of the valve disease is one of many factors to consider when deciding whether to perform intervention before the patient becomes pregnant. Plans for future pregnancy may also affect the choice of prosthesis, as anticoagulation management during pregnancy can be extremely tricky.
The recommendations overall make clear that, based on available evidence, “there should be a high threshold for intervention during pregnancy,” said Dr. O’Gara. And no one should go it alone when taking care of these complicated patients. “Care of pregnant women with heart disease requires coordination with a multidisciplinary team,” he said.
Dr. O’Gara reported no relevant disclosures.
koakes@frontlinemedcom.com
On Twitter @karioakes
CHICAGO – Pregnancy-associated hemodynamic and physiologic changes can challenge hearts with diseased aortic valves. Understanding pregnancy’s toll on the body can help physicians take better care of pregnant women with aortic stenosis or aortic regurgitation, said Patrick T. O’Gara, MD. “This is a relatively infrequent condition, and it causes all of us a great deal of apprehension,” he said.
Dr. O’Gara, director of clinical cardiology at Brigham and Women’s Hospital, Boston, walked attendees through current recommendations for caring for pregnant women with aortic valve disease in a presentation at the annual Heart Valve Summit.
When talking to women with aortic valve disease, it’s important to let them know that a pregnancy during which they experience an adverse maternal cardiac event appears to increase their risk for later events as well, said Dr. O’Gara. One study of pregnancy in women with aortic valve disease, said Dr. O’Gara, found that the risk of later events for women who had had a cardiac event in pregnancy was 27% plus or minus 9 percentage points in the 5 years following pregnancy, while the risk was just 15% plus or minus 3 percentage points for women whose pregnancies were not complicated by cardiac adverse events (P = .02) (Heart. 2010 Oct;96(20):1656-61).
The World Health Organization classification of pregnancy risk, said Dr. O’Gara, provides some guidance. Patients who are WHO class III are considered to be high risk; pregnancy in these individuals calls for counseling and a multidisciplinary care team. Patients who fall into this class are those with mechanical heart valves, those with Marfan syndrome and aortic involvement with a valve diameter of 40-45 mm, and those with bicuspid aortic valve disease with a valve diameter of 45-50 mm.
WHO class IV patients, according to the guidelines, are at prohibitive risk. These patients should consider terminating a pregnancy if one occurs. Conditions that place women into WHO class IV include symptomatic severe aortic stenosis (AS), severe left ventricular dysfunction with an ejection fraction less than 40%, and severe coarctation of the aorta. Women with Marfan syndrome with aortic valve diameters greater than 45 mm and women with bicuspid aortic valves and a diameter over 50 mm also fall into this category of prohibitive risk.
If a physician is lucky enough to have a pre-conception relationship with a patient, then a host of considerations can come into play. In addition to cardiac risk stratification, obstetric and intrapartum risk to the mother as well as neonatal risk should be considered. Some factors to take into account are maternal life expectancy and potential long-term complications to the mother from pregnancy and childbirth.
Plasma volume increases rapidly through the first trimester, plateauing about halfway through pregnancy. With this normal dilutional anemia, “Most normal pregnancies are accompanied by a grade II systolic murmur, best heard at the upper left sternal border,” said Dr. O’Gara. Since stroke volume and heart rate both also increase through pregnancy, cardiac output goes up as well.
“Importantly, systemic vascular resistance is reduced throughout pregnancy,” said Dr. O’Gara. Consequently, “regurgitant heart valve problems are generally much better tolerated than stenotic lesions in pregnancy. That’s physiologic principle number one.”
In labor, abrupt hemodynamic changes include increases in cardiac output, heart rate, blood pressure, and venous return. “Labor and delivery are very stressful hemodynamic times for women,” said Dr. O’Gara.
In the immediate postpartum period, uterine contraction results in a return of a significant amount of blood to the maternal circulation. This effectively creates an autotransfusion, with resulting increased preload and cardiac output. The inferior vena cava is also freed from the pressure of a gravid uterus, increasing venous return further. Finally, there’s also a marked increase in systemic vascular resistance.
Aside from the risk of adverse cardiac events, other adverse outcomes for women can include an increased risk of premature rupture of membranes and postpartum hemorrhage. For the neonate, preterm birth and respiratory distress are more likely. Newborns are more likely to be small for gestational age, to suffer intraventricular hemorrhage, and to die. Depending on the parental genetic status, the infant may be at risk of congenital heart disease as well.
Neonatal risk may also be increased, said Dr. O’Gara. Some maternal conditions that can increase risk for the infant include a baseline New York Heart Association (NYHA) class of greater than II or having cyanosis; having a left heart obstruction; being a smoker; having a multiple gestation; having a mechanical heart valve; and taking an oral anticoagulant.
Ideally, a full discussion would include a genetic referral if indicated, as well as coordination with a gynecologist or primary care provider to provide contraception and to assist with planning for and optimizing outcomes of a pregnancy.
The American College of Cardiology and the American Heart Association have issued guidelines that include a class I recommendation regarding valve intervention before pregnancy for symptomatic patients with severe AS (those with aortic velocity of at least 4.0 m/sec, or mean pressure gradient greater than 41 mm Hg; stage D). For patients who meet the same hemodynamic criteria for severe AS but who are asymptomatic (stage C), the guidelines have a class IIa recommendation that pre-pregnancy valve intervention is “reasonable.” These recommendations have level C evidence supporting them.
However, another class IIa recommendation for patients with severe AS who are already pregnant advises valve intervention only in the case of hemodynamic deterioration, or the development of NYHA class III to IV heart failure symptoms. This is backed by grade B evidence, according to the associations.
For aortic regurgitation (AR), a level I recommendation advises valve repair or replacement before pregnancy if patients are symptomatic and have severe (stage D) regurgitation. Pregnant patients with severe regurgitation should only be considered for a valve operation if they have refractory NYHA class IV heart failure symptoms. These recommendations have level C evidence supporting them.
Since AR is better tolerated in pregnancy than AS, the nature of the valve disease is one of many factors to consider when deciding whether to perform intervention before the patient becomes pregnant. Plans for future pregnancy may also affect the choice of prosthesis, as anticoagulation management during pregnancy can be extremely tricky.
The recommendations overall make clear that, based on available evidence, “there should be a high threshold for intervention during pregnancy,” said Dr. O’Gara. And no one should go it alone when taking care of these complicated patients. “Care of pregnant women with heart disease requires coordination with a multidisciplinary team,” he said.
Dr. O’Gara reported no relevant disclosures.
koakes@frontlinemedcom.com
On Twitter @karioakes
CHICAGO – Pregnancy-associated hemodynamic and physiologic changes can challenge hearts with diseased aortic valves. Understanding pregnancy’s toll on the body can help physicians take better care of pregnant women with aortic stenosis or aortic regurgitation, said Patrick T. O’Gara, MD. “This is a relatively infrequent condition, and it causes all of us a great deal of apprehension,” he said.
Dr. O’Gara, director of clinical cardiology at Brigham and Women’s Hospital, Boston, walked attendees through current recommendations for caring for pregnant women with aortic valve disease in a presentation at the annual Heart Valve Summit.
When talking to women with aortic valve disease, it’s important to let them know that a pregnancy during which they experience an adverse maternal cardiac event appears to increase their risk for later events as well, said Dr. O’Gara. One study of pregnancy in women with aortic valve disease, said Dr. O’Gara, found that the risk of later events for women who had had a cardiac event in pregnancy was 27% plus or minus 9 percentage points in the 5 years following pregnancy, while the risk was just 15% plus or minus 3 percentage points for women whose pregnancies were not complicated by cardiac adverse events (P = .02) (Heart. 2010 Oct;96(20):1656-61).
The World Health Organization classification of pregnancy risk, said Dr. O’Gara, provides some guidance. Patients who are WHO class III are considered to be high risk; pregnancy in these individuals calls for counseling and a multidisciplinary care team. Patients who fall into this class are those with mechanical heart valves, those with Marfan syndrome and aortic involvement with a valve diameter of 40-45 mm, and those with bicuspid aortic valve disease with a valve diameter of 45-50 mm.
WHO class IV patients, according to the guidelines, are at prohibitive risk. These patients should consider terminating a pregnancy if one occurs. Conditions that place women into WHO class IV include symptomatic severe aortic stenosis (AS), severe left ventricular dysfunction with an ejection fraction less than 40%, and severe coarctation of the aorta. Women with Marfan syndrome with aortic valve diameters greater than 45 mm and women with bicuspid aortic valves and a diameter over 50 mm also fall into this category of prohibitive risk.
If a physician is lucky enough to have a pre-conception relationship with a patient, then a host of considerations can come into play. In addition to cardiac risk stratification, obstetric and intrapartum risk to the mother as well as neonatal risk should be considered. Some factors to take into account are maternal life expectancy and potential long-term complications to the mother from pregnancy and childbirth.
Plasma volume increases rapidly through the first trimester, plateauing about halfway through pregnancy. With this normal dilutional anemia, “Most normal pregnancies are accompanied by a grade II systolic murmur, best heard at the upper left sternal border,” said Dr. O’Gara. Since stroke volume and heart rate both also increase through pregnancy, cardiac output goes up as well.
“Importantly, systemic vascular resistance is reduced throughout pregnancy,” said Dr. O’Gara. Consequently, “regurgitant heart valve problems are generally much better tolerated than stenotic lesions in pregnancy. That’s physiologic principle number one.”
In labor, abrupt hemodynamic changes include increases in cardiac output, heart rate, blood pressure, and venous return. “Labor and delivery are very stressful hemodynamic times for women,” said Dr. O’Gara.
In the immediate postpartum period, uterine contraction results in a return of a significant amount of blood to the maternal circulation. This effectively creates an autotransfusion, with resulting increased preload and cardiac output. The inferior vena cava is also freed from the pressure of a gravid uterus, increasing venous return further. Finally, there’s also a marked increase in systemic vascular resistance.
Aside from the risk of adverse cardiac events, other adverse outcomes for women can include an increased risk of premature rupture of membranes and postpartum hemorrhage. For the neonate, preterm birth and respiratory distress are more likely. Newborns are more likely to be small for gestational age, to suffer intraventricular hemorrhage, and to die. Depending on the parental genetic status, the infant may be at risk of congenital heart disease as well.
Neonatal risk may also be increased, said Dr. O’Gara. Some maternal conditions that can increase risk for the infant include a baseline New York Heart Association (NYHA) class of greater than II or having cyanosis; having a left heart obstruction; being a smoker; having a multiple gestation; having a mechanical heart valve; and taking an oral anticoagulant.
Ideally, a full discussion would include a genetic referral if indicated, as well as coordination with a gynecologist or primary care provider to provide contraception and to assist with planning for and optimizing outcomes of a pregnancy.
The American College of Cardiology and the American Heart Association have issued guidelines that include a class I recommendation regarding valve intervention before pregnancy for symptomatic patients with severe AS (those with aortic velocity of at least 4.0 m/sec, or mean pressure gradient greater than 41 mm Hg; stage D). For patients who meet the same hemodynamic criteria for severe AS but who are asymptomatic (stage C), the guidelines have a class IIa recommendation that pre-pregnancy valve intervention is “reasonable.” These recommendations have level C evidence supporting them.
However, another class IIa recommendation for patients with severe AS who are already pregnant advises valve intervention only in the case of hemodynamic deterioration, or the development of NYHA class III to IV heart failure symptoms. This is backed by grade B evidence, according to the associations.
For aortic regurgitation (AR), a level I recommendation advises valve repair or replacement before pregnancy if patients are symptomatic and have severe (stage D) regurgitation. Pregnant patients with severe regurgitation should only be considered for a valve operation if they have refractory NYHA class IV heart failure symptoms. These recommendations have level C evidence supporting them.
Since AR is better tolerated in pregnancy than AS, the nature of the valve disease is one of many factors to consider when deciding whether to perform intervention before the patient becomes pregnant. Plans for future pregnancy may also affect the choice of prosthesis, as anticoagulation management during pregnancy can be extremely tricky.
The recommendations overall make clear that, based on available evidence, “there should be a high threshold for intervention during pregnancy,” said Dr. O’Gara. And no one should go it alone when taking care of these complicated patients. “Care of pregnant women with heart disease requires coordination with a multidisciplinary team,” he said.
Dr. O’Gara reported no relevant disclosures.
koakes@frontlinemedcom.com
On Twitter @karioakes
EXPERT ANALYSIS FROM THE HEART VALVE SUMMIT
Newly available tissue containment system brings back power morcellation to advanced MIG surgeons
In the 11 studies that have examined the incidence of leiomyosarcoma (LMS) since the 2014 communications by the US Food and Drug Administration (FDA),1 Dr. Matthew Siedhoff and colleagues found that the cumulative LMS incidence in 318,006 women was 0.0017%, or approximately 1 in 600. This is according to data Dr. Seidhoff presented November 16, 2016, at the 45th annual AAGL Global Congress on MIGS.2 This reported risk is smaller than the 1 in 350 cited by the FDA in its 2014 notice.1 Dr. Seidhoff concluded that, particularly in women aged younger than 50 years, minimally invasive hysterectomy remains a safe option for the informed patient to consider.
Regardless of the incidence of unsuspected LMS found in the studies published since 2014, what have been the practice changes among gynecologic surgeons as a result of the FDA’s 2014 actions? Kerac N. Falk, MD, explored this question with his colleagues at the Icahn School of Medicine in New York, New York. He presented these findings to AAGL congress attendees on November 16 in Orlando, Florida.3 Notably, of 197 responders to a survey sent to members of the Society of Gynecologic Oncologists, 12.5% reported decreasing their use of power morcellation. A full 38.8% reported discontinuing altogether the use of a power morcellator.
One in 5 (20%) of gyn oncologists previously using power morcellation reported switching to laparotomy. Importantly, the drive for the switch was “driven by media, patient request, and the FDA rather than physician choice,” Falk said.
What if physicians, and patients, have another choice?
Here at the AAGL meeting, Olympus announced the FDA clearance, marketing, and initial training on its contained tissue extraction system—the PneumoLiner containment device and its accompanying laparoscopic PK Morcellator (FIGURE).
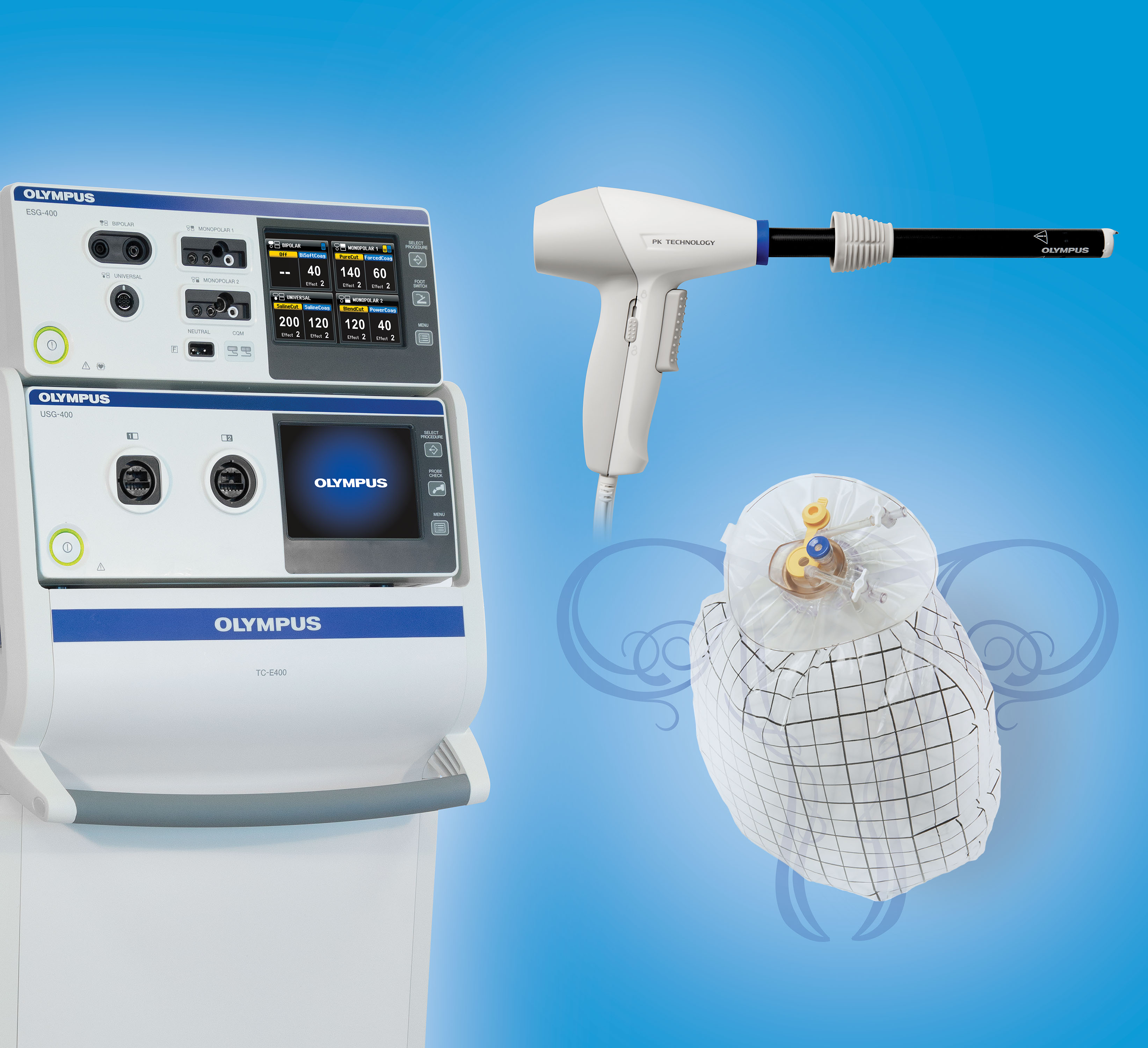
Both tools, FDA cleared in April and October 2016, respectively, provide low-risk, appropriate women with “an improved safety device,” said Jubilee Brown, MD, AAGL representative on power morcellation to the FDA and Associate Director of Gynecologic Oncology at the Levine Cancer Institute of the Carolinas HealthCare System in Charlotte, North Carolina.
“We have found at our institution that we have had to do more opens and minilaps on patients without the option for power morcellation, and this new device offers a way for us not to have to do that. Minimally invasive surgery stays truly minimally invasive surgery,” said Dr. Brown.
“Although I have a handful of patients who have chosen open surgery over minimally invasive surgery when MIS was appropriate, by and large most patients prefer the minimally invasive approach to surgery—especially when they learn that they have reduced risk for complications, blood loss, and pain; will be back to normal life faster; and will have less cosmetic incisions,” said Dr. Brown. “We all have tried workarounds, but for low-risk, appropriate patients under FDA guidelines, this new device by Olympus brings us back to minimally invasive options, and that is a good thing.”
How does the Olympus containment system work?
The PneumoLiner is the first containment device to receive FDA market clearance that is designed for use with certain laparoscopic morcellators to isolate uterine tissue that is not suspected to contain cancer.4
The containment bag and morcellator are not indicated for use in women with tissue that is known or suspected to contain malignancy; and should not be used for removal of uterine tissue containing suspected fibroids in patients who are peri- or post-menopausal, or candidates for en bloc tissue removal vaginally or via mini-laparotomy.4
When insufflated, the PneumoLiner bag allows for space and surgeon visibility throughout the power morcellation procedure while maintaining a barrier to the escape of fluids, cells, and tissue fragments. The bag creates a barrier between the targeted tissue and nontargeted abdominal contents, minimizing the risk of inadvertent damage to adjacent structures, says Olympus.4 See this video, which demonstrates the procedure.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Filtration, immersion, and stress testing were conducted to demonstrate the PneumoLiner as impermeable to human cells, that it maintains its integrity following morcellation, and that its mechanical strength withholds forces in excess of those demonstrated during actual use.5 To support FDA 510 clearance of the PK Morcellator, Olympus performed additional testing with both devices used together, demonstrating compatibility.4
Surgeon training on new device use is rigid, comprehensive, and not to be overlooked
“This is one of the rare times in my career when, right out of the gate, we have a very rigid and comprehensive way of educating and training physicians on the proper utilization of a new technology (avoiding the cart-before-the-horse phenomenon, in which a clinician is given the tools without the proper education),” said AAGL immediate past president Arnold Advincula, MD, “and it is important to underscore that.” Dr. Advincula is the Levine Family Professor, Vice-Chair of Women’s Health, and Chief of Gynecology at Sloane Women’s Hospital, Columbia University Medical Center/New York-Presbyterian Hospital, New York, New York.
Training plans
Instead of its sales force, Olympus is having surgeons train other surgeons. Clinical Education Specialists, a small, select group of individuals within the company, also will be able to train. The required training follows a rigid protocol that was validated and submitted to the FDA as part of the approval process, said Jerilyn Hitchings, Director of Procedure Marketing at Olympus. After training up to 40 surgeons in the past 3 weeks, Olympus expects to train another 100 at AAGL.
“We have strict criteria for training, including that they are advanced laparoscopists,” fulfilling a minimum number of surgeries within a 90-day period, said Hitchings.
The 1-hour training requires surgeons to perform a 4-step protocol and demonstrate unaided proficiency at training conclusion. Olympus will not sell its product to a facility until an advanced surgeon has been trained there, according to Hitchings. And many physicians are approaching their institutions inquiring about training now, she reports.
Applications for training will be submitted to the Olympus Professional Education Team for approval. “We want this to go well. We know that there are many physicians who are hoping that this becomes a good reality, and we want to make sure that it is done properly. The training process adheres to that agenda.”
Having the technology is step 1
OBG Management Board of Editors member Dr. Advincula struck an optimistic tone in his June 2016 Guest Editorial for the journal,6 indicating his belief that the “tissue morcellation pendulum,” which has swung toward non−minimally invasive approaches since 2014, can change direction. At the Olympus containment system official introduction event at AAGL, he expressed the same optimism:
“We now have technology that has the promise of being able to deliver to women a safe way to undergo tissue extraction, to undergo a minimally invasive surgical procedure. …When you combine innovation and education together, you ultimately are going to advance MIS worldwide. With all the things going on around the world today, I think it is important that women be able to have a choice, an autonomous choice with her physician. Having the technology that allows physicians to extract tissue safely, to give women the option of undergoing a minimally invasive surgery, is critically important. I look forward to seeing how we can continue to advance surgery. Partnership with clinicians and with industry, such as Olympus, is going to be key to the success of how we advance women’s health care in general.”
What do AAGL attendees have to say about it?
“It’s a good thing for the minimally invasive market,” said John B. Gebhart, MD, MS, vaginal hysterectomy representative at the star-studded Operating with the Stars event on Thursday, November 17, and Professor of Obstetrics and Gynecology at the Mayo Clinic in Rochester, Minnesota.
May Thomassee, MD, who practices in Lafayette, Louisiana, said that, for the past 3 years, she has been performing extracorporeal morcellation at her institution. “I think the new containment bag and morcellator devices that are attempting to be safer are a very good thing; however, my concern of introducing this new technology, such as bags, is that it may increase the cost within our health care system. We have had great success, and patients feel that they have options when we offer them—after informed consent and appropriate preoperative workup—an abdominal, vaginal, or extracorporeal morcellation approach.”
As a developer and proponent of the Extracorporeal Tissue Extraction (ExCITE) technique, Mireille Truong, MD, who demonstrated the ExCITE surgical approach with Dr. Advincula as part of the Research and Science Plenary here at AAGL, said, “I think the PneumoLiner is a great example of creativity and innovation. It’s good that we now have an FDA-approved device to offer and counsel patients as one of many options.” She added that, overall, the morcellation controversy has been “a positive experience. We have learned better as a community about how to approach new technology.”
- US Food and Drug Administration. Laparoscopic uterine power morcellation in hysterectomy and myomectomy: FDA safety communication. April 17, 2014. http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm393576.htm. Updated November 24, 2014. Accessed November 17, 2016.
- Siedhoff MT, Doll KM, Rutstein SE, et al. Laparoscopic hysterectomy with morcellation versus abdominal hysterectomy for presumed uterine leiomyomata: an updated decision analysis. J Minim Invasiv Gynecol. 2016;23(7):S4-S5.
- Mandelberger AH, Mathews S, Chuang L. Practice changes in power morcellation among gynecologic-oncologists since 2014. J Minim Invasiv Gynecol. 2016;23(7):S3.
Olympus introduces first-of-its-kind contained tissue extraction system, restoring healthcare option for gynecologists and women [press release]. November 16, 2016. Olympus website. http://www.olympusamerica.com/corporate/corp_presscenter_headline.asp?pressNo=2079. Accessed November 16, 2016.
- FDA allows marketing of first-of-kind tissue containment system for use with certain laparoscopic power morcellators in select patients. FDA website. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm494650.htm. Updated April 7, 2016. Accessed November 16, 2016.
- Advincula AP. Tissue extraction: can the pendulum change direction? OBG Manag. 2016;28(6):8, 10, 12.
In the 11 studies that have examined the incidence of leiomyosarcoma (LMS) since the 2014 communications by the US Food and Drug Administration (FDA),1 Dr. Matthew Siedhoff and colleagues found that the cumulative LMS incidence in 318,006 women was 0.0017%, or approximately 1 in 600. This is according to data Dr. Seidhoff presented November 16, 2016, at the 45th annual AAGL Global Congress on MIGS.2 This reported risk is smaller than the 1 in 350 cited by the FDA in its 2014 notice.1 Dr. Seidhoff concluded that, particularly in women aged younger than 50 years, minimally invasive hysterectomy remains a safe option for the informed patient to consider.
Regardless of the incidence of unsuspected LMS found in the studies published since 2014, what have been the practice changes among gynecologic surgeons as a result of the FDA’s 2014 actions? Kerac N. Falk, MD, explored this question with his colleagues at the Icahn School of Medicine in New York, New York. He presented these findings to AAGL congress attendees on November 16 in Orlando, Florida.3 Notably, of 197 responders to a survey sent to members of the Society of Gynecologic Oncologists, 12.5% reported decreasing their use of power morcellation. A full 38.8% reported discontinuing altogether the use of a power morcellator.
One in 5 (20%) of gyn oncologists previously using power morcellation reported switching to laparotomy. Importantly, the drive for the switch was “driven by media, patient request, and the FDA rather than physician choice,” Falk said.
What if physicians, and patients, have another choice?
Here at the AAGL meeting, Olympus announced the FDA clearance, marketing, and initial training on its contained tissue extraction system—the PneumoLiner containment device and its accompanying laparoscopic PK Morcellator (FIGURE).

Both tools, FDA cleared in April and October 2016, respectively, provide low-risk, appropriate women with “an improved safety device,” said Jubilee Brown, MD, AAGL representative on power morcellation to the FDA and Associate Director of Gynecologic Oncology at the Levine Cancer Institute of the Carolinas HealthCare System in Charlotte, North Carolina.
“We have found at our institution that we have had to do more opens and minilaps on patients without the option for power morcellation, and this new device offers a way for us not to have to do that. Minimally invasive surgery stays truly minimally invasive surgery,” said Dr. Brown.
“Although I have a handful of patients who have chosen open surgery over minimally invasive surgery when MIS was appropriate, by and large most patients prefer the minimally invasive approach to surgery—especially when they learn that they have reduced risk for complications, blood loss, and pain; will be back to normal life faster; and will have less cosmetic incisions,” said Dr. Brown. “We all have tried workarounds, but for low-risk, appropriate patients under FDA guidelines, this new device by Olympus brings us back to minimally invasive options, and that is a good thing.”
How does the Olympus containment system work?
The PneumoLiner is the first containment device to receive FDA market clearance that is designed for use with certain laparoscopic morcellators to isolate uterine tissue that is not suspected to contain cancer.4
The containment bag and morcellator are not indicated for use in women with tissue that is known or suspected to contain malignancy; and should not be used for removal of uterine tissue containing suspected fibroids in patients who are peri- or post-menopausal, or candidates for en bloc tissue removal vaginally or via mini-laparotomy.4
When insufflated, the PneumoLiner bag allows for space and surgeon visibility throughout the power morcellation procedure while maintaining a barrier to the escape of fluids, cells, and tissue fragments. The bag creates a barrier between the targeted tissue and nontargeted abdominal contents, minimizing the risk of inadvertent damage to adjacent structures, says Olympus.4 See this video, which demonstrates the procedure.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Filtration, immersion, and stress testing were conducted to demonstrate the PneumoLiner as impermeable to human cells, that it maintains its integrity following morcellation, and that its mechanical strength withholds forces in excess of those demonstrated during actual use.5 To support FDA 510 clearance of the PK Morcellator, Olympus performed additional testing with both devices used together, demonstrating compatibility.4
Surgeon training on new device use is rigid, comprehensive, and not to be overlooked
“This is one of the rare times in my career when, right out of the gate, we have a very rigid and comprehensive way of educating and training physicians on the proper utilization of a new technology (avoiding the cart-before-the-horse phenomenon, in which a clinician is given the tools without the proper education),” said AAGL immediate past president Arnold Advincula, MD, “and it is important to underscore that.” Dr. Advincula is the Levine Family Professor, Vice-Chair of Women’s Health, and Chief of Gynecology at Sloane Women’s Hospital, Columbia University Medical Center/New York-Presbyterian Hospital, New York, New York.
Training plans
Instead of its sales force, Olympus is having surgeons train other surgeons. Clinical Education Specialists, a small, select group of individuals within the company, also will be able to train. The required training follows a rigid protocol that was validated and submitted to the FDA as part of the approval process, said Jerilyn Hitchings, Director of Procedure Marketing at Olympus. After training up to 40 surgeons in the past 3 weeks, Olympus expects to train another 100 at AAGL.
“We have strict criteria for training, including that they are advanced laparoscopists,” fulfilling a minimum number of surgeries within a 90-day period, said Hitchings.
The 1-hour training requires surgeons to perform a 4-step protocol and demonstrate unaided proficiency at training conclusion. Olympus will not sell its product to a facility until an advanced surgeon has been trained there, according to Hitchings. And many physicians are approaching their institutions inquiring about training now, she reports.
Applications for training will be submitted to the Olympus Professional Education Team for approval. “We want this to go well. We know that there are many physicians who are hoping that this becomes a good reality, and we want to make sure that it is done properly. The training process adheres to that agenda.”
Having the technology is step 1
OBG Management Board of Editors member Dr. Advincula struck an optimistic tone in his June 2016 Guest Editorial for the journal,6 indicating his belief that the “tissue morcellation pendulum,” which has swung toward non−minimally invasive approaches since 2014, can change direction. At the Olympus containment system official introduction event at AAGL, he expressed the same optimism:
“We now have technology that has the promise of being able to deliver to women a safe way to undergo tissue extraction, to undergo a minimally invasive surgical procedure. …When you combine innovation and education together, you ultimately are going to advance MIS worldwide. With all the things going on around the world today, I think it is important that women be able to have a choice, an autonomous choice with her physician. Having the technology that allows physicians to extract tissue safely, to give women the option of undergoing a minimally invasive surgery, is critically important. I look forward to seeing how we can continue to advance surgery. Partnership with clinicians and with industry, such as Olympus, is going to be key to the success of how we advance women’s health care in general.”
What do AAGL attendees have to say about it?
“It’s a good thing for the minimally invasive market,” said John B. Gebhart, MD, MS, vaginal hysterectomy representative at the star-studded Operating with the Stars event on Thursday, November 17, and Professor of Obstetrics and Gynecology at the Mayo Clinic in Rochester, Minnesota.
May Thomassee, MD, who practices in Lafayette, Louisiana, said that, for the past 3 years, she has been performing extracorporeal morcellation at her institution. “I think the new containment bag and morcellator devices that are attempting to be safer are a very good thing; however, my concern of introducing this new technology, such as bags, is that it may increase the cost within our health care system. We have had great success, and patients feel that they have options when we offer them—after informed consent and appropriate preoperative workup—an abdominal, vaginal, or extracorporeal morcellation approach.”
As a developer and proponent of the Extracorporeal Tissue Extraction (ExCITE) technique, Mireille Truong, MD, who demonstrated the ExCITE surgical approach with Dr. Advincula as part of the Research and Science Plenary here at AAGL, said, “I think the PneumoLiner is a great example of creativity and innovation. It’s good that we now have an FDA-approved device to offer and counsel patients as one of many options.” She added that, overall, the morcellation controversy has been “a positive experience. We have learned better as a community about how to approach new technology.”
In the 11 studies that have examined the incidence of leiomyosarcoma (LMS) since the 2014 communications by the US Food and Drug Administration (FDA),1 Dr. Matthew Siedhoff and colleagues found that the cumulative LMS incidence in 318,006 women was 0.0017%, or approximately 1 in 600. This is according to data Dr. Seidhoff presented November 16, 2016, at the 45th annual AAGL Global Congress on MIGS.2 This reported risk is smaller than the 1 in 350 cited by the FDA in its 2014 notice.1 Dr. Seidhoff concluded that, particularly in women aged younger than 50 years, minimally invasive hysterectomy remains a safe option for the informed patient to consider.
Regardless of the incidence of unsuspected LMS found in the studies published since 2014, what have been the practice changes among gynecologic surgeons as a result of the FDA’s 2014 actions? Kerac N. Falk, MD, explored this question with his colleagues at the Icahn School of Medicine in New York, New York. He presented these findings to AAGL congress attendees on November 16 in Orlando, Florida.3 Notably, of 197 responders to a survey sent to members of the Society of Gynecologic Oncologists, 12.5% reported decreasing their use of power morcellation. A full 38.8% reported discontinuing altogether the use of a power morcellator.
One in 5 (20%) of gyn oncologists previously using power morcellation reported switching to laparotomy. Importantly, the drive for the switch was “driven by media, patient request, and the FDA rather than physician choice,” Falk said.
What if physicians, and patients, have another choice?
Here at the AAGL meeting, Olympus announced the FDA clearance, marketing, and initial training on its contained tissue extraction system—the PneumoLiner containment device and its accompanying laparoscopic PK Morcellator (FIGURE).

Both tools, FDA cleared in April and October 2016, respectively, provide low-risk, appropriate women with “an improved safety device,” said Jubilee Brown, MD, AAGL representative on power morcellation to the FDA and Associate Director of Gynecologic Oncology at the Levine Cancer Institute of the Carolinas HealthCare System in Charlotte, North Carolina.
“We have found at our institution that we have had to do more opens and minilaps on patients without the option for power morcellation, and this new device offers a way for us not to have to do that. Minimally invasive surgery stays truly minimally invasive surgery,” said Dr. Brown.
“Although I have a handful of patients who have chosen open surgery over minimally invasive surgery when MIS was appropriate, by and large most patients prefer the minimally invasive approach to surgery—especially when they learn that they have reduced risk for complications, blood loss, and pain; will be back to normal life faster; and will have less cosmetic incisions,” said Dr. Brown. “We all have tried workarounds, but for low-risk, appropriate patients under FDA guidelines, this new device by Olympus brings us back to minimally invasive options, and that is a good thing.”
How does the Olympus containment system work?
The PneumoLiner is the first containment device to receive FDA market clearance that is designed for use with certain laparoscopic morcellators to isolate uterine tissue that is not suspected to contain cancer.4
The containment bag and morcellator are not indicated for use in women with tissue that is known or suspected to contain malignancy; and should not be used for removal of uterine tissue containing suspected fibroids in patients who are peri- or post-menopausal, or candidates for en bloc tissue removal vaginally or via mini-laparotomy.4
When insufflated, the PneumoLiner bag allows for space and surgeon visibility throughout the power morcellation procedure while maintaining a barrier to the escape of fluids, cells, and tissue fragments. The bag creates a barrier between the targeted tissue and nontargeted abdominal contents, minimizing the risk of inadvertent damage to adjacent structures, says Olympus.4 See this video, which demonstrates the procedure.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Filtration, immersion, and stress testing were conducted to demonstrate the PneumoLiner as impermeable to human cells, that it maintains its integrity following morcellation, and that its mechanical strength withholds forces in excess of those demonstrated during actual use.5 To support FDA 510 clearance of the PK Morcellator, Olympus performed additional testing with both devices used together, demonstrating compatibility.4
Surgeon training on new device use is rigid, comprehensive, and not to be overlooked
“This is one of the rare times in my career when, right out of the gate, we have a very rigid and comprehensive way of educating and training physicians on the proper utilization of a new technology (avoiding the cart-before-the-horse phenomenon, in which a clinician is given the tools without the proper education),” said AAGL immediate past president Arnold Advincula, MD, “and it is important to underscore that.” Dr. Advincula is the Levine Family Professor, Vice-Chair of Women’s Health, and Chief of Gynecology at Sloane Women’s Hospital, Columbia University Medical Center/New York-Presbyterian Hospital, New York, New York.
Training plans
Instead of its sales force, Olympus is having surgeons train other surgeons. Clinical Education Specialists, a small, select group of individuals within the company, also will be able to train. The required training follows a rigid protocol that was validated and submitted to the FDA as part of the approval process, said Jerilyn Hitchings, Director of Procedure Marketing at Olympus. After training up to 40 surgeons in the past 3 weeks, Olympus expects to train another 100 at AAGL.
“We have strict criteria for training, including that they are advanced laparoscopists,” fulfilling a minimum number of surgeries within a 90-day period, said Hitchings.
The 1-hour training requires surgeons to perform a 4-step protocol and demonstrate unaided proficiency at training conclusion. Olympus will not sell its product to a facility until an advanced surgeon has been trained there, according to Hitchings. And many physicians are approaching their institutions inquiring about training now, she reports.
Applications for training will be submitted to the Olympus Professional Education Team for approval. “We want this to go well. We know that there are many physicians who are hoping that this becomes a good reality, and we want to make sure that it is done properly. The training process adheres to that agenda.”
Having the technology is step 1
OBG Management Board of Editors member Dr. Advincula struck an optimistic tone in his June 2016 Guest Editorial for the journal,6 indicating his belief that the “tissue morcellation pendulum,” which has swung toward non−minimally invasive approaches since 2014, can change direction. At the Olympus containment system official introduction event at AAGL, he expressed the same optimism:
“We now have technology that has the promise of being able to deliver to women a safe way to undergo tissue extraction, to undergo a minimally invasive surgical procedure. …When you combine innovation and education together, you ultimately are going to advance MIS worldwide. With all the things going on around the world today, I think it is important that women be able to have a choice, an autonomous choice with her physician. Having the technology that allows physicians to extract tissue safely, to give women the option of undergoing a minimally invasive surgery, is critically important. I look forward to seeing how we can continue to advance surgery. Partnership with clinicians and with industry, such as Olympus, is going to be key to the success of how we advance women’s health care in general.”
What do AAGL attendees have to say about it?
“It’s a good thing for the minimally invasive market,” said John B. Gebhart, MD, MS, vaginal hysterectomy representative at the star-studded Operating with the Stars event on Thursday, November 17, and Professor of Obstetrics and Gynecology at the Mayo Clinic in Rochester, Minnesota.
May Thomassee, MD, who practices in Lafayette, Louisiana, said that, for the past 3 years, she has been performing extracorporeal morcellation at her institution. “I think the new containment bag and morcellator devices that are attempting to be safer are a very good thing; however, my concern of introducing this new technology, such as bags, is that it may increase the cost within our health care system. We have had great success, and patients feel that they have options when we offer them—after informed consent and appropriate preoperative workup—an abdominal, vaginal, or extracorporeal morcellation approach.”
As a developer and proponent of the Extracorporeal Tissue Extraction (ExCITE) technique, Mireille Truong, MD, who demonstrated the ExCITE surgical approach with Dr. Advincula as part of the Research and Science Plenary here at AAGL, said, “I think the PneumoLiner is a great example of creativity and innovation. It’s good that we now have an FDA-approved device to offer and counsel patients as one of many options.” She added that, overall, the morcellation controversy has been “a positive experience. We have learned better as a community about how to approach new technology.”
- US Food and Drug Administration. Laparoscopic uterine power morcellation in hysterectomy and myomectomy: FDA safety communication. April 17, 2014. http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm393576.htm. Updated November 24, 2014. Accessed November 17, 2016.
- Siedhoff MT, Doll KM, Rutstein SE, et al. Laparoscopic hysterectomy with morcellation versus abdominal hysterectomy for presumed uterine leiomyomata: an updated decision analysis. J Minim Invasiv Gynecol. 2016;23(7):S4-S5.
- Mandelberger AH, Mathews S, Chuang L. Practice changes in power morcellation among gynecologic-oncologists since 2014. J Minim Invasiv Gynecol. 2016;23(7):S3.
Olympus introduces first-of-its-kind contained tissue extraction system, restoring healthcare option for gynecologists and women [press release]. November 16, 2016. Olympus website. http://www.olympusamerica.com/corporate/corp_presscenter_headline.asp?pressNo=2079. Accessed November 16, 2016.
- FDA allows marketing of first-of-kind tissue containment system for use with certain laparoscopic power morcellators in select patients. FDA website. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm494650.htm. Updated April 7, 2016. Accessed November 16, 2016.
- Advincula AP. Tissue extraction: can the pendulum change direction? OBG Manag. 2016;28(6):8, 10, 12.
- US Food and Drug Administration. Laparoscopic uterine power morcellation in hysterectomy and myomectomy: FDA safety communication. April 17, 2014. http://www.fda.gov/MedicalDevices/Safety/AlertsandNotices/ucm393576.htm. Updated November 24, 2014. Accessed November 17, 2016.
- Siedhoff MT, Doll KM, Rutstein SE, et al. Laparoscopic hysterectomy with morcellation versus abdominal hysterectomy for presumed uterine leiomyomata: an updated decision analysis. J Minim Invasiv Gynecol. 2016;23(7):S4-S5.
- Mandelberger AH, Mathews S, Chuang L. Practice changes in power morcellation among gynecologic-oncologists since 2014. J Minim Invasiv Gynecol. 2016;23(7):S3.
Olympus introduces first-of-its-kind contained tissue extraction system, restoring healthcare option for gynecologists and women [press release]. November 16, 2016. Olympus website. http://www.olympusamerica.com/corporate/corp_presscenter_headline.asp?pressNo=2079. Accessed November 16, 2016.
- FDA allows marketing of first-of-kind tissue containment system for use with certain laparoscopic power morcellators in select patients. FDA website. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm494650.htm. Updated April 7, 2016. Accessed November 16, 2016.
- Advincula AP. Tissue extraction: can the pendulum change direction? OBG Manag. 2016;28(6):8, 10, 12.
VIDEO: Biologics: Proposed guideline addresses perioperative management
WASHINGTON – Biologic agents should be stopped prior to elective total knee or hip arthroplasty in patients with rheumatic diseases, according to a draft guideline developed by the American College of Rheumatology and the American Association of Hip and Knee Surgeons.
The guideline, which address the perioperative management of antirheumatic medications in patients with rheumatoid arthritis, spondyloarthritis, psoriatic arthritis, juvenile idiopathic arthritis (JIA), or lupus who are undergoing such surgery, is currently under review, Dr. Susan Goodman, MD, coprincipal investigator, reported at the annual meeting of the American College of Rheumatology.
The draft guideline was created because “guidance was needed for common clinical situations, even where data were sparse. We didn’t want to configure treatment mandates – that’s not what these are,” Dr. Goodman of Cornell University, New York, said.
The recommendations are conditional, she said, meaning that the benefits probably outweigh the harms, that the recommendations apply to most but not all patients, and that future research may lead to changes.
“They’re also preference sensitive,” she said, explaining that patients’ values and preferences should be carefully considered, as they might differ from those of the patient panel consulted during guideline development; the panel expressed greater concern about the risk of infection following surgery than about perioperative flares resulting from medication discontinuation.
Based on agreement by at least 80% of a voting panel which considered available evidence in the context of their clinical experience along with the input from the patient panel, the draft guideline states that:
• Current doses of methotrexate, leflunomide, hydroxychloroquine, and sulfasalazine should be continued in patients with rheumatic diseases undergoing elective hip and knee replacement. This recommendation is based on an extensive literature review that showed the infection rate is decreased in patients who continue these medications, Dr. Goodman said.
• All biologics should be withheld prior to surgery in patients with inflammatory arthritis, and surgery should be planned for the end of the dosing cycle. This matter wasn’t specifically addressed in the literature; however, numerous randomized controlled trials outside of the surgical setting demonstrate an increased risk of infection associated with their use, she noted.
“All of the biologic medications were found to be associated with an increased risk of infection,” she said. “Because of this and the level of importance patients place on minimizing infection risk, we’ve recommended that biologics be withheld prior to surgery.”
• Tofacitinib, which was considered in a separate oral, targeted therapy category, should be withheld for at least 7 days prior to surgery in patients with RA, spondyloarthritis, and JIA. Data from systematic reviews and meta-analyses showed an increased risk of infection with tofacitinib, although more research is needed in order to “firm up” this recommendation, Dr. Goodman said.
• In lupus patients, rituximab and belimumab should be withheld prior to surgery, and surgery should be planned for the end of the dosing period.
“Again, this was not answered in the literature. We depended on observational studies that we reviewed that did show that patients with severe active lupus were at much higher risk for adverse events. But since rituximab isn’t approved by the [Food and Drug Administration] for use in lupus, and belimumab isn’t approved for use in severe lupus – and those seem to be the high-risk patients – we thought withholding them was more prudent,” she said.
• Patients with severe lupus should continue on current doses of methotrexate, mycophenolic acid, azathioprine, mizoribine, cyclosporine, and tacrolimus through surgery. This recommendation is based on indirect data from experience in organ transplant patients.
• All medications should be discontinued in patients whose lupus is not severe.
“Our recommendation is to withhold for 7 days to 2-5 days after surgery in the absence of any wound healing complications or any other complications,” she said, noting that the literature does not directly address this; the recommendation is based on indirect evidence in patients with either active infection or who are at risk for infection.
“We thought that careful monitoring of the patient would permit us to identify flare and intervene quickly. … and that, for mild cases of lupus, the morbidity associated with infection might not be greater than the morbidity associated with the disease flare,” she said.
• Biologics should be restarted once surgical wounds show evidence of healing and there is no clinical evidence of infection. The literature does not directly address this; the recommendation is based on the rationale for use of these medications in patients with either active infection or risk for infection.
• Current daily doses of glucocorticoids, rather than supraphysiologic doses, should be continued in adults with RA, lupus, or inflammatory arthritis. A meta-analysis and systematic review of randomized controlled trial data and observational data showed no hemodynamic difference between daily doses and stress doses.
“In addition, there are abundant observational data demonstrating an increase in infection in patients on chronic steroids greater than 15 mg, and we thought that part of the optimization of the patient would be getting them on the lowest possible steroid dose,” she said, stressing that this refers only to adults receiving glucocorticoids for their rheumatic disease, and not to those with a history of JIA who may have received steroids during development, or to those receiving glucocorticoids for primary, adrenal, or hypothalamic disease.
According to Dr. Goodman, the time is right for the introduction of these recommendations, because the increased use of disease-modifying drugs and biologics means that most patients coming in for these surgeries will be taking these medications.
Further, despite the widespread use of the medications, the rate of total knee and hip arthroplasty surgeries among patients with rheumatic diseases is about the same as it was 20 or 30 years ago – and their risk for devastating complications, including infections, remains high, she said, noting that appropriate medication management provides an opportunity to mitigate risk.
Coprincipal investigator, Bryan Springer, MD, further emphasized the importance of the guideline, noting that the 5-year survival among rheumatic disease patients who develop certain perioperative complications is lower than for many common cancers, and that the literature offers little guidance on managing medications in the perioperative period.
“We now have a document that’s based on the available evidence, and also based on expert opinion, to help us manage these patients much more thoroughly in the perioperative period,” Dr. Springer, an orthopedic surgeon in Charlotte, N.C., said during a press briefing on the guideline.
Dr. Springer highlighted the value of the unique collaboration between the ACR and the AAHKS, calling the effort a win both for patients, and for “collaborative efforts, collaborative research, which we just really don’t do enough of,” he said. “I hope this is a huge step towards that direction.”
This guideline development process was funded by the ACR and AAHKS.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WASHINGTON – Biologic agents should be stopped prior to elective total knee or hip arthroplasty in patients with rheumatic diseases, according to a draft guideline developed by the American College of Rheumatology and the American Association of Hip and Knee Surgeons.
The guideline, which address the perioperative management of antirheumatic medications in patients with rheumatoid arthritis, spondyloarthritis, psoriatic arthritis, juvenile idiopathic arthritis (JIA), or lupus who are undergoing such surgery, is currently under review, Dr. Susan Goodman, MD, coprincipal investigator, reported at the annual meeting of the American College of Rheumatology.
The draft guideline was created because “guidance was needed for common clinical situations, even where data were sparse. We didn’t want to configure treatment mandates – that’s not what these are,” Dr. Goodman of Cornell University, New York, said.
The recommendations are conditional, she said, meaning that the benefits probably outweigh the harms, that the recommendations apply to most but not all patients, and that future research may lead to changes.
“They’re also preference sensitive,” she said, explaining that patients’ values and preferences should be carefully considered, as they might differ from those of the patient panel consulted during guideline development; the panel expressed greater concern about the risk of infection following surgery than about perioperative flares resulting from medication discontinuation.
Based on agreement by at least 80% of a voting panel which considered available evidence in the context of their clinical experience along with the input from the patient panel, the draft guideline states that:
• Current doses of methotrexate, leflunomide, hydroxychloroquine, and sulfasalazine should be continued in patients with rheumatic diseases undergoing elective hip and knee replacement. This recommendation is based on an extensive literature review that showed the infection rate is decreased in patients who continue these medications, Dr. Goodman said.
• All biologics should be withheld prior to surgery in patients with inflammatory arthritis, and surgery should be planned for the end of the dosing cycle. This matter wasn’t specifically addressed in the literature; however, numerous randomized controlled trials outside of the surgical setting demonstrate an increased risk of infection associated with their use, she noted.
“All of the biologic medications were found to be associated with an increased risk of infection,” she said. “Because of this and the level of importance patients place on minimizing infection risk, we’ve recommended that biologics be withheld prior to surgery.”
• Tofacitinib, which was considered in a separate oral, targeted therapy category, should be withheld for at least 7 days prior to surgery in patients with RA, spondyloarthritis, and JIA. Data from systematic reviews and meta-analyses showed an increased risk of infection with tofacitinib, although more research is needed in order to “firm up” this recommendation, Dr. Goodman said.
• In lupus patients, rituximab and belimumab should be withheld prior to surgery, and surgery should be planned for the end of the dosing period.
“Again, this was not answered in the literature. We depended on observational studies that we reviewed that did show that patients with severe active lupus were at much higher risk for adverse events. But since rituximab isn’t approved by the [Food and Drug Administration] for use in lupus, and belimumab isn’t approved for use in severe lupus – and those seem to be the high-risk patients – we thought withholding them was more prudent,” she said.
• Patients with severe lupus should continue on current doses of methotrexate, mycophenolic acid, azathioprine, mizoribine, cyclosporine, and tacrolimus through surgery. This recommendation is based on indirect data from experience in organ transplant patients.
• All medications should be discontinued in patients whose lupus is not severe.
“Our recommendation is to withhold for 7 days to 2-5 days after surgery in the absence of any wound healing complications or any other complications,” she said, noting that the literature does not directly address this; the recommendation is based on indirect evidence in patients with either active infection or who are at risk for infection.
“We thought that careful monitoring of the patient would permit us to identify flare and intervene quickly. … and that, for mild cases of lupus, the morbidity associated with infection might not be greater than the morbidity associated with the disease flare,” she said.
• Biologics should be restarted once surgical wounds show evidence of healing and there is no clinical evidence of infection. The literature does not directly address this; the recommendation is based on the rationale for use of these medications in patients with either active infection or risk for infection.
• Current daily doses of glucocorticoids, rather than supraphysiologic doses, should be continued in adults with RA, lupus, or inflammatory arthritis. A meta-analysis and systematic review of randomized controlled trial data and observational data showed no hemodynamic difference between daily doses and stress doses.
“In addition, there are abundant observational data demonstrating an increase in infection in patients on chronic steroids greater than 15 mg, and we thought that part of the optimization of the patient would be getting them on the lowest possible steroid dose,” she said, stressing that this refers only to adults receiving glucocorticoids for their rheumatic disease, and not to those with a history of JIA who may have received steroids during development, or to those receiving glucocorticoids for primary, adrenal, or hypothalamic disease.
According to Dr. Goodman, the time is right for the introduction of these recommendations, because the increased use of disease-modifying drugs and biologics means that most patients coming in for these surgeries will be taking these medications.
Further, despite the widespread use of the medications, the rate of total knee and hip arthroplasty surgeries among patients with rheumatic diseases is about the same as it was 20 or 30 years ago – and their risk for devastating complications, including infections, remains high, she said, noting that appropriate medication management provides an opportunity to mitigate risk.
Coprincipal investigator, Bryan Springer, MD, further emphasized the importance of the guideline, noting that the 5-year survival among rheumatic disease patients who develop certain perioperative complications is lower than for many common cancers, and that the literature offers little guidance on managing medications in the perioperative period.
“We now have a document that’s based on the available evidence, and also based on expert opinion, to help us manage these patients much more thoroughly in the perioperative period,” Dr. Springer, an orthopedic surgeon in Charlotte, N.C., said during a press briefing on the guideline.
Dr. Springer highlighted the value of the unique collaboration between the ACR and the AAHKS, calling the effort a win both for patients, and for “collaborative efforts, collaborative research, which we just really don’t do enough of,” he said. “I hope this is a huge step towards that direction.”
This guideline development process was funded by the ACR and AAHKS.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WASHINGTON – Biologic agents should be stopped prior to elective total knee or hip arthroplasty in patients with rheumatic diseases, according to a draft guideline developed by the American College of Rheumatology and the American Association of Hip and Knee Surgeons.
The guideline, which address the perioperative management of antirheumatic medications in patients with rheumatoid arthritis, spondyloarthritis, psoriatic arthritis, juvenile idiopathic arthritis (JIA), or lupus who are undergoing such surgery, is currently under review, Dr. Susan Goodman, MD, coprincipal investigator, reported at the annual meeting of the American College of Rheumatology.
The draft guideline was created because “guidance was needed for common clinical situations, even where data were sparse. We didn’t want to configure treatment mandates – that’s not what these are,” Dr. Goodman of Cornell University, New York, said.
The recommendations are conditional, she said, meaning that the benefits probably outweigh the harms, that the recommendations apply to most but not all patients, and that future research may lead to changes.
“They’re also preference sensitive,” she said, explaining that patients’ values and preferences should be carefully considered, as they might differ from those of the patient panel consulted during guideline development; the panel expressed greater concern about the risk of infection following surgery than about perioperative flares resulting from medication discontinuation.
Based on agreement by at least 80% of a voting panel which considered available evidence in the context of their clinical experience along with the input from the patient panel, the draft guideline states that:
• Current doses of methotrexate, leflunomide, hydroxychloroquine, and sulfasalazine should be continued in patients with rheumatic diseases undergoing elective hip and knee replacement. This recommendation is based on an extensive literature review that showed the infection rate is decreased in patients who continue these medications, Dr. Goodman said.
• All biologics should be withheld prior to surgery in patients with inflammatory arthritis, and surgery should be planned for the end of the dosing cycle. This matter wasn’t specifically addressed in the literature; however, numerous randomized controlled trials outside of the surgical setting demonstrate an increased risk of infection associated with their use, she noted.
“All of the biologic medications were found to be associated with an increased risk of infection,” she said. “Because of this and the level of importance patients place on minimizing infection risk, we’ve recommended that biologics be withheld prior to surgery.”
• Tofacitinib, which was considered in a separate oral, targeted therapy category, should be withheld for at least 7 days prior to surgery in patients with RA, spondyloarthritis, and JIA. Data from systematic reviews and meta-analyses showed an increased risk of infection with tofacitinib, although more research is needed in order to “firm up” this recommendation, Dr. Goodman said.
• In lupus patients, rituximab and belimumab should be withheld prior to surgery, and surgery should be planned for the end of the dosing period.
“Again, this was not answered in the literature. We depended on observational studies that we reviewed that did show that patients with severe active lupus were at much higher risk for adverse events. But since rituximab isn’t approved by the [Food and Drug Administration] for use in lupus, and belimumab isn’t approved for use in severe lupus – and those seem to be the high-risk patients – we thought withholding them was more prudent,” she said.
• Patients with severe lupus should continue on current doses of methotrexate, mycophenolic acid, azathioprine, mizoribine, cyclosporine, and tacrolimus through surgery. This recommendation is based on indirect data from experience in organ transplant patients.
• All medications should be discontinued in patients whose lupus is not severe.
“Our recommendation is to withhold for 7 days to 2-5 days after surgery in the absence of any wound healing complications or any other complications,” she said, noting that the literature does not directly address this; the recommendation is based on indirect evidence in patients with either active infection or who are at risk for infection.
“We thought that careful monitoring of the patient would permit us to identify flare and intervene quickly. … and that, for mild cases of lupus, the morbidity associated with infection might not be greater than the morbidity associated with the disease flare,” she said.
• Biologics should be restarted once surgical wounds show evidence of healing and there is no clinical evidence of infection. The literature does not directly address this; the recommendation is based on the rationale for use of these medications in patients with either active infection or risk for infection.
• Current daily doses of glucocorticoids, rather than supraphysiologic doses, should be continued in adults with RA, lupus, or inflammatory arthritis. A meta-analysis and systematic review of randomized controlled trial data and observational data showed no hemodynamic difference between daily doses and stress doses.
“In addition, there are abundant observational data demonstrating an increase in infection in patients on chronic steroids greater than 15 mg, and we thought that part of the optimization of the patient would be getting them on the lowest possible steroid dose,” she said, stressing that this refers only to adults receiving glucocorticoids for their rheumatic disease, and not to those with a history of JIA who may have received steroids during development, or to those receiving glucocorticoids for primary, adrenal, or hypothalamic disease.
According to Dr. Goodman, the time is right for the introduction of these recommendations, because the increased use of disease-modifying drugs and biologics means that most patients coming in for these surgeries will be taking these medications.
Further, despite the widespread use of the medications, the rate of total knee and hip arthroplasty surgeries among patients with rheumatic diseases is about the same as it was 20 or 30 years ago – and their risk for devastating complications, including infections, remains high, she said, noting that appropriate medication management provides an opportunity to mitigate risk.
Coprincipal investigator, Bryan Springer, MD, further emphasized the importance of the guideline, noting that the 5-year survival among rheumatic disease patients who develop certain perioperative complications is lower than for many common cancers, and that the literature offers little guidance on managing medications in the perioperative period.
“We now have a document that’s based on the available evidence, and also based on expert opinion, to help us manage these patients much more thoroughly in the perioperative period,” Dr. Springer, an orthopedic surgeon in Charlotte, N.C., said during a press briefing on the guideline.
Dr. Springer highlighted the value of the unique collaboration between the ACR and the AAHKS, calling the effort a win both for patients, and for “collaborative efforts, collaborative research, which we just really don’t do enough of,” he said. “I hope this is a huge step towards that direction.”
This guideline development process was funded by the ACR and AAHKS.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE ACR ANNUAL MEETING
Restrictive Blood Transfusion Strategies May Increase the Risk of Mortality, Morbidity for Elderly Patients Undergoing Orthopedic Surgery
Clinical Question: Are there particular groups of patients in which lower transfusion thresholds (transfusion only at lower hemoglobin levels) may be harmful?
Background: Previously published meta-analyses have examined transfusion thresholds for critically ill, surgical, and medical patients. By combining these patients, previous meta-analyses are limited in the identification of intervention effects. A more refined understanding of how transfusion thresholds impact outcomes for a variety of patients in different clinical settings is needed.
Study Design: Context-specific systematic review and meta-analysis of randomized clinical trials.
Setting: Adult patients in perioperative, emergency, or intensive-care settings.
Synopsis: Patient information was extracted from 31 randomized clinical trials. The authors found that among 3,465 elderly patients undergoing orthopedic surgery, those given restrictive transfusion strategies had significantly more events reflecting inadequate oxygen supply (relative risk, 1.41; 95% CI, 1.03–1.92). No statistically significant effect from restrictive transfusions was seen in 3,322 patients with cardiovascular disease undergoing cardiac or vascular procedures; 3,590 mixed medical-surgical patients in emergency and intensive-care settings; and 823 patients in a combined group of postpartum women, hematologic malignancy patients, and younger patients with neurologic injury.
The authors argue that even statistically nonsignificant differences in morbidity and mortality should encourage more liberal transfusion; apart from orthopedic surgery patients, this argument is not well-supported by the available data.
Bottom Line: It remains unclear whether restrictive transfusion strategies have a negative impact on certain types of patients, although the authors argue that there may be a trend in that direction. Further study is needed for specific patient populations.
Citation: Hovaguimian F, Myles PS. Restrictive versus liberal transfusion strategy in the perioperative and acute care settings: a context-specific systematic review and meta-analysis of randomized clinical trials. Anesthesiology. 2016;125(1):46-61.
Clinical Question: Are there particular groups of patients in which lower transfusion thresholds (transfusion only at lower hemoglobin levels) may be harmful?
Background: Previously published meta-analyses have examined transfusion thresholds for critically ill, surgical, and medical patients. By combining these patients, previous meta-analyses are limited in the identification of intervention effects. A more refined understanding of how transfusion thresholds impact outcomes for a variety of patients in different clinical settings is needed.
Study Design: Context-specific systematic review and meta-analysis of randomized clinical trials.
Setting: Adult patients in perioperative, emergency, or intensive-care settings.
Synopsis: Patient information was extracted from 31 randomized clinical trials. The authors found that among 3,465 elderly patients undergoing orthopedic surgery, those given restrictive transfusion strategies had significantly more events reflecting inadequate oxygen supply (relative risk, 1.41; 95% CI, 1.03–1.92). No statistically significant effect from restrictive transfusions was seen in 3,322 patients with cardiovascular disease undergoing cardiac or vascular procedures; 3,590 mixed medical-surgical patients in emergency and intensive-care settings; and 823 patients in a combined group of postpartum women, hematologic malignancy patients, and younger patients with neurologic injury.
The authors argue that even statistically nonsignificant differences in morbidity and mortality should encourage more liberal transfusion; apart from orthopedic surgery patients, this argument is not well-supported by the available data.
Bottom Line: It remains unclear whether restrictive transfusion strategies have a negative impact on certain types of patients, although the authors argue that there may be a trend in that direction. Further study is needed for specific patient populations.
Citation: Hovaguimian F, Myles PS. Restrictive versus liberal transfusion strategy in the perioperative and acute care settings: a context-specific systematic review and meta-analysis of randomized clinical trials. Anesthesiology. 2016;125(1):46-61.
Clinical Question: Are there particular groups of patients in which lower transfusion thresholds (transfusion only at lower hemoglobin levels) may be harmful?
Background: Previously published meta-analyses have examined transfusion thresholds for critically ill, surgical, and medical patients. By combining these patients, previous meta-analyses are limited in the identification of intervention effects. A more refined understanding of how transfusion thresholds impact outcomes for a variety of patients in different clinical settings is needed.
Study Design: Context-specific systematic review and meta-analysis of randomized clinical trials.
Setting: Adult patients in perioperative, emergency, or intensive-care settings.
Synopsis: Patient information was extracted from 31 randomized clinical trials. The authors found that among 3,465 elderly patients undergoing orthopedic surgery, those given restrictive transfusion strategies had significantly more events reflecting inadequate oxygen supply (relative risk, 1.41; 95% CI, 1.03–1.92). No statistically significant effect from restrictive transfusions was seen in 3,322 patients with cardiovascular disease undergoing cardiac or vascular procedures; 3,590 mixed medical-surgical patients in emergency and intensive-care settings; and 823 patients in a combined group of postpartum women, hematologic malignancy patients, and younger patients with neurologic injury.
The authors argue that even statistically nonsignificant differences in morbidity and mortality should encourage more liberal transfusion; apart from orthopedic surgery patients, this argument is not well-supported by the available data.
Bottom Line: It remains unclear whether restrictive transfusion strategies have a negative impact on certain types of patients, although the authors argue that there may be a trend in that direction. Further study is needed for specific patient populations.
Citation: Hovaguimian F, Myles PS. Restrictive versus liberal transfusion strategy in the perioperative and acute care settings: a context-specific systematic review and meta-analysis of randomized clinical trials. Anesthesiology. 2016;125(1):46-61.
Robotic surgery instruments ‘virtually impossible’ to clean completely
Instruments used for robotic surgery are “virtually impossible” to clean completely, according to a report published in Infection Control & Hospital Epidemiology.
“A new standard for the cleaning of complex surgical instruments needs to be established, especially for those used in robotic surgery,” wrote Yuhei Saito of the surgical center at the University of Tokyo Hospital and associates.
They assessed the residual contamination of both robotic and regular surgical instruments at their medical center because “hospital staff in central sterile supply departments are troubled by the reprocessing of robotic instruments because they cannot be disassembled for cleaning like other endoscopic instruments. Their complex structure impairs brushing the inner surface of narrow lumens, resulting in failure to [completely] remove contaminants,” the researchers wrote.
In the first phase of the study, the researchers examined 41 instruments immediately after they were used in robotic surgery (7 radical prostatectomies and 2 anterior resections of the rectum) and 27 regular instruments immediately after they were used for open surgery (gastrectomy and colectomy). The robotic instruments were contaminated with 72.3 × 103 mcg of protein each, compared with 5.5 × 103 mcg of protein on the regular instruments, the investigators reported (Infect Control Hosp Epidemiol. 2016 Oct 31. doi: 10.1017/ice.2016.249).
In the second phase of the study, the researchers examined another 24 robotic instruments and 40 regular instruments after they were used in surgery and then cleaned according to the manufacturers’ instructions three successive times. For the robotic instruments, this involved manually brushing the outer surface while moving the instrument “wrists” through their full range of motion, followed by 15 minutes of ultrasonication with enzymatic detergent, flushing the lumen with a water gun through flush ports, and rinsing the entire instrument. For regular instruments, cleaning involved washer-disinfectors and included 5 minutes of ultrasonication, 10 minutes of spraying with an alkaline detergent, and 10 minutes of disinfection via heating.
The level of contamination declined with each successive cleaning but still remained comparatively high for the robotic instruments. The amount of protein contaminants released in the three cleanings was 650, 550, and 530 mcg per robotic instrument, compared with 16, 17, and 17 mcg per ordinary instrument.
The efficacy of cleaning was 97.6% for robotic instruments and 99.1% for regular instruments, the researchers reported.
This study was not designed to assess whether residual contamination is associated with adverse events such as infection in subsequent patients, and there are few data available on this topic.
“We have to recognize that there might be a considerable volume of insufficient cleaning or occult surgical site infections,” the investigators wrote.
New instrument washers equipped with a specific cleaning function for narrow lumens are becoming available, they noted, and “further study should be conducted using these washers with improved cleaning efficacy.”
The study was supported by the Japan Society for the Promotion of Science. The investigators reported having no relevant financial disclosures.
Instruments used for robotic surgery are “virtually impossible” to clean completely, according to a report published in Infection Control & Hospital Epidemiology.
“A new standard for the cleaning of complex surgical instruments needs to be established, especially for those used in robotic surgery,” wrote Yuhei Saito of the surgical center at the University of Tokyo Hospital and associates.
They assessed the residual contamination of both robotic and regular surgical instruments at their medical center because “hospital staff in central sterile supply departments are troubled by the reprocessing of robotic instruments because they cannot be disassembled for cleaning like other endoscopic instruments. Their complex structure impairs brushing the inner surface of narrow lumens, resulting in failure to [completely] remove contaminants,” the researchers wrote.
In the first phase of the study, the researchers examined 41 instruments immediately after they were used in robotic surgery (7 radical prostatectomies and 2 anterior resections of the rectum) and 27 regular instruments immediately after they were used for open surgery (gastrectomy and colectomy). The robotic instruments were contaminated with 72.3 × 103 mcg of protein each, compared with 5.5 × 103 mcg of protein on the regular instruments, the investigators reported (Infect Control Hosp Epidemiol. 2016 Oct 31. doi: 10.1017/ice.2016.249).
In the second phase of the study, the researchers examined another 24 robotic instruments and 40 regular instruments after they were used in surgery and then cleaned according to the manufacturers’ instructions three successive times. For the robotic instruments, this involved manually brushing the outer surface while moving the instrument “wrists” through their full range of motion, followed by 15 minutes of ultrasonication with enzymatic detergent, flushing the lumen with a water gun through flush ports, and rinsing the entire instrument. For regular instruments, cleaning involved washer-disinfectors and included 5 minutes of ultrasonication, 10 minutes of spraying with an alkaline detergent, and 10 minutes of disinfection via heating.
The level of contamination declined with each successive cleaning but still remained comparatively high for the robotic instruments. The amount of protein contaminants released in the three cleanings was 650, 550, and 530 mcg per robotic instrument, compared with 16, 17, and 17 mcg per ordinary instrument.
The efficacy of cleaning was 97.6% for robotic instruments and 99.1% for regular instruments, the researchers reported.
This study was not designed to assess whether residual contamination is associated with adverse events such as infection in subsequent patients, and there are few data available on this topic.
“We have to recognize that there might be a considerable volume of insufficient cleaning or occult surgical site infections,” the investigators wrote.
New instrument washers equipped with a specific cleaning function for narrow lumens are becoming available, they noted, and “further study should be conducted using these washers with improved cleaning efficacy.”
The study was supported by the Japan Society for the Promotion of Science. The investigators reported having no relevant financial disclosures.
Instruments used for robotic surgery are “virtually impossible” to clean completely, according to a report published in Infection Control & Hospital Epidemiology.
“A new standard for the cleaning of complex surgical instruments needs to be established, especially for those used in robotic surgery,” wrote Yuhei Saito of the surgical center at the University of Tokyo Hospital and associates.
They assessed the residual contamination of both robotic and regular surgical instruments at their medical center because “hospital staff in central sterile supply departments are troubled by the reprocessing of robotic instruments because they cannot be disassembled for cleaning like other endoscopic instruments. Their complex structure impairs brushing the inner surface of narrow lumens, resulting in failure to [completely] remove contaminants,” the researchers wrote.
In the first phase of the study, the researchers examined 41 instruments immediately after they were used in robotic surgery (7 radical prostatectomies and 2 anterior resections of the rectum) and 27 regular instruments immediately after they were used for open surgery (gastrectomy and colectomy). The robotic instruments were contaminated with 72.3 × 103 mcg of protein each, compared with 5.5 × 103 mcg of protein on the regular instruments, the investigators reported (Infect Control Hosp Epidemiol. 2016 Oct 31. doi: 10.1017/ice.2016.249).
In the second phase of the study, the researchers examined another 24 robotic instruments and 40 regular instruments after they were used in surgery and then cleaned according to the manufacturers’ instructions three successive times. For the robotic instruments, this involved manually brushing the outer surface while moving the instrument “wrists” through their full range of motion, followed by 15 minutes of ultrasonication with enzymatic detergent, flushing the lumen with a water gun through flush ports, and rinsing the entire instrument. For regular instruments, cleaning involved washer-disinfectors and included 5 minutes of ultrasonication, 10 minutes of spraying with an alkaline detergent, and 10 minutes of disinfection via heating.
The level of contamination declined with each successive cleaning but still remained comparatively high for the robotic instruments. The amount of protein contaminants released in the three cleanings was 650, 550, and 530 mcg per robotic instrument, compared with 16, 17, and 17 mcg per ordinary instrument.
The efficacy of cleaning was 97.6% for robotic instruments and 99.1% for regular instruments, the researchers reported.
This study was not designed to assess whether residual contamination is associated with adverse events such as infection in subsequent patients, and there are few data available on this topic.
“We have to recognize that there might be a considerable volume of insufficient cleaning or occult surgical site infections,” the investigators wrote.
New instrument washers equipped with a specific cleaning function for narrow lumens are becoming available, they noted, and “further study should be conducted using these washers with improved cleaning efficacy.”
The study was supported by the Japan Society for the Promotion of Science. The investigators reported having no relevant financial disclosures.
FROM INFECTION CONTROL AND HOSPITAL EPIDEMIOLOGY
Key clinical point:
Major finding: Immediately after surgery, robotic instruments were contaminated with 72.3 × 103 mcg of protein each, compared with 5.5 × 103 mcg of protein on regular instruments.
Data source: A single-center analysis of protein contamination before and after three successive cleanings on 65 instruments used for robotic surgery and 67 regular instruments used for open surgery.
Disclosures: This study was supported by the Japan Society for the Promotion of Science. The investigators reported having no relevant financial disclosures.