User login
The Integration of Extended Reality in Arthroplasty: Reviewing Technological Progress and Clinical Benefits
The Integration of Extended Reality in Arthroplasty: Reviewing Technological Progress and Clinical Benefits
The introduction of extended reality (XR) to the operating room (OR) has proved promising for enhancing surgical precision and improving patient outcomes. In the field of orthopedic surgery, precise alignment of implants is integral to maintaining functional range of motion and preventing impingement of adjacent neurovascular structures. XR systems have shown promise in arthroplasty including by improving precision and streamlining surgery by allowing surgeons to create 3D preoperative plans that are accessible intraoperatively. This article explores the current applications of XR in arthroplasty, highlights recent advancements and benefits, and describes limitations in comparison to traditional techniques.
Methods
A literature search identified studies involving the use of XR in arthroplasty and current US Food and Drug Administration (FDA)-approved XR systems. Multiple electronic databases were used, including PubMed, Google Scholar, and IEEE Xplore. Search terms included: extended reality, augmented reality, virtual reality, arthroplasty, joint replacement, total knee arthroplasty, total shoulder arthroplasty, and total hip arthroplasty. The study design, intervention details, outcomes, and comparisons with traditional surgical techniques were thematically analyzed, with identification of common ideas associated with XR use in arthroplasty. This narrative report highlights the integration of XR in arthroplasty.
Extended Reality Fundamentals
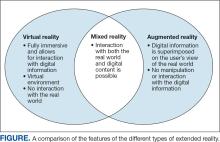
XR encompasses augmented reality (AR), virtual reality (VR), and mixed reality (MR). AR involves superimposing digitally rendered information and images onto the surgeon’s view of the real world, typically through the use of a headset and smart glasses.1 AR allows the surgeon to move and interact freely within the OR, removing the need for additional screens or devices to display patient information or imaging. VR is a fully immersive simulation using a headset that obstructs the view of the real world but allows the user to move freely within this virtual setting, often with audio or other sensory stimuli. MR combines AR and VR to create a digital model that allows for real-world interaction, with the advantage of adapting information and models in real time.2 Whereas in AR the surgeon can view the data projected from the headset, MR provides the ability to interact with and manipulate the digital content (Figure). Both AR and MR have been adapted for use in the OR, while VR has been adapted for use in surgical planning and training.
Extended Reality Use in Orthopedics
The HipNav system was introduced in 1995 to create preoperative plans that assist surgeons in accurately implanting the acetabular cup during total hip arthroplasty (THA).3 Although not commercially successful, this system spurred surgeons to experiment with XR to improve the accuracy and alignment of orthopedic implants. Systems capable of displaying the desired intraoperative implant placement have flourished, with applications in fracture reduction, arthroplasty, solid tumor resection, and hardware placement.4-7 Accurate alignment has been linked to improvements in patient outcomes.8-10 XR has great potential within the field of arthroplasty, with multiple new systems approved by the FDA and currently available in the US (Table).
Hip Arthroplasty
Orientation of the acetabular cup is a technically challenging part of THA. Accuracy in the anteversion and inclination angles of the acetabular cup is required to maintain implant stability, preserve functional range of motion (ROM), and prevent precocious wear.11,12 Despite preoperative planning, surgeons often overestimate the inclination angle and underestimate anteversion.13 Improper implantation of the acetabular cup can lead to joint instability caused by aseptic loosening, increasing the risk of dislocation and the need for revision surgery.14,15 Dislocations typically present to the emergency department, but primary care practitioners may encounter patients with pain or diminished sensation due to impingement or instability.16
The introduction of XR into the OR has provided the opportunity for real-time navigation and adjustment of the acetabular cup to maximize anteversion and inclination angles. Currently, 2 FDA-approved systems are available for THA: the Zimmer and Surgical Planning Associates HipInsight system, and the Insight Augmented Reality Visualization and Information System (ARVIS). The HipInsight system consists of a hologram projection using the Microsoft HoloLens2 device and optimizes preoperative planning, producing accuracy of anteversion and inclination angles within 3°.17 ARVIS employs existing surgical helmets and 2 mounted tracking cameras to provide navigation intraoperatively. ARVIS has also been approved for use in total knee arthroplasty (TKA) and unicompartmental knee arthroplasty.18
HipInsight has shown utility in increasing the accuracy of acetabular cup placement along with the use of biplanar radiographic scans.19 However, there are no studies validating the efficacy of ARVIS and HipInsight and assessing long-term disease-oriented or patient-oriented outcomes.
Knee Arthroplasty
In the setting of TKA, XR is most effective in ensuring accurate resection of the tibial and femoral components. Achieving the planned femoral coronal, axial, and sagittal angles allows the prosthesis to be on the femoral axis of rotation, improving functional outcomes. XR systems for TKA have been shown to increase the accuracy of distal femoral resection with a limited increase in surgery duration.20,21 For TKA in particular, patients are often less satisfied with the result than surgeons expect.22 Accurate alignment can improve patient satisfaction and reduce return-to-clinic rates for postoperative pain management, a factor that primary care practitioners should consider when recommending a patient for TKA.23
Along with ARVIS, 3 additional XR systems are FDA-approved for use in TKA. The Pixee Medical Knee+ system uses smart glasses and trackers to aid in the positioning of instruments for improved accuracy while allowing real-time navigation.24 The Medacta NextAR Knee’s single-use tracking system allows for intraoperative navigation with the use of AR glasses.25 The Polaris STELLAR Knee uses MR and avoids the need for preoperative imaging by capturing real-time anatomic data.26
The Pixee Medical Knee+ system was commercially available in Europe for several years prior to FDA approval, so more research exists on its efficacy. One study found that the Pixee Medical Knee+ system initially demonstrated an inferior clinical outcome, attributed to the learning curve associated with using the system.27 However, more recent studies have shown its utility in improving alignment, regardless of implant specifications.28,29 The Medacta NextAR Knee system has been shown to improve accuracy of tibial rotation and soft tissue balance and even increase OR efficiency.30,31 The Polaris STELLAR Knee system received FDA approval in 2023; no published research exists on its accuracy and outcomes.26
Shoulder Arthroplasty
Minimally invasive techniques are favored in total shoulder arthroplasty (TSA) due to the vitality of maintaining the surrounding soft tissue to maximize preservation of motility and strength.32 However, this complicates the procedure by decreasing the ability to effectively access and visualize key structures of the shoulder. Accordingly, issues with implant positioning and alignment are more common with TSA than other joint arthroplasties, making XR particularly promising.33 Some studies report that up to 67% of patients experience glenohumeral instability, which can clinically present as weakness, decreased range of motion, and persistent shoulder pain.34,35 The use of preoperative computed tomography to improve understanding of glenoid anatomy and glenohumeral subluxation is becoming increasingly common, and it can be combined with XR to improve accuracy.36,37
Two FDA-approved systems are available. The Stryker Blueprint MR system is used for intraoperative guidance and integration for patient imaging used for preoperative planning. The Medacta NextAR Shoulder system is a parallel of the company’s TKA system. The Stryker Blueprint MR system combines the Microsoft HoloLens 2 headset to display preoperative plans with a secondary display for coordination with the rest of the surgical team.38 Similar to the Medacta NextAR Knee, the Medacta NextAR Shoulder system uses the same single-use tracking system and AR glasses for intraoperative guidance.39
Data on the long-term outcomes of using these systems are still limited, but the Stryker Blueprint MR system has not been shown to accurately predict postoperative ROM.40 Cadaveric studies have demonstrated that the Medacta NextAR Shoulder system can provide accurate inclination, retroversion, entry point, depth, and rotation values based on the preoperative planned values.41,42 However, this accuracy has yet to be confirmed in vivo, and the impact of using XR in TSA on long-term outcomes is still unknown.
Challenges and Limitations
Though XR has proven to be promising in arthroplasty, several limitations regarding widespread implementation exist. In particular, there is a steep learning curve associated with the use of XR systems, which can cause increased operative time and even initial inferior outcomes, as demonstrated with the Pixee Medical Knee+ system. The need for extensive practice and training prior to use could delay widespread adoption and may cause discrepancies in surgical outcomes. Unfamiliarity with the system and technological difficulties that may require troubleshooting can also increase operative time, particularly for surgeons new to using the XR system. Though intraoperative navigation is expected to improve accuracy of implant alignment, its added complexity may also result in longer surgeries.
In addition to the steep learning curve and increased operative time, there is a high upfront cost associated with XR systems. Exact costs of XR systems are not typically disclosed, but available estimates suggest an average sales price of about $1000 per case. Given the proprietary nature of these technologies, publicly available cost data are limited, making it challenging to fully assess the financial burden on health care institutions. Though some systems, such as ARVIS, can be integrated with existing surgical helmets, many require the purchase of AR glasses and secondary displays. This can cause further variation in the total expense for each system. In low-resource settings, this represents a significant challenge to widespread implementation. To justify this cost, additional research on long-term patient outcomes is needed to ensure the benefits of XR systems outweigh the cost.
Although early studies on XR systems in arthroplasty have shown improvements in precision and short-term outcomes, long-term data regarding effectiveness remains. Even systems such as ARVIS and HipInsight have limited long-term follow-up, making it difficult to assess whether the improved accuracy with these XR systems translates into improved patient outcomes compared with traditional arthroplasty.
CONCLUSIONS
XR technologies have shown significant potential in enhancing precision and patient outcomes. Through the integration of XR in the OR, surgeons can visualize preoperative plans and even make intraoperative changes, with the benefit of improving implant alignment.
There are some disadvantages to its use, however, including high cost and increased operative time. Despite this, the integration of XR into surgical practice can deliver more precise implant alignment and address other challenges faced with conventional techniques. As these technologies evolve and studies on long-term outcomes validate their utility, XR has the potential to transform the field of arthroplasty.
Azuma RT. A survey of augmented reality. Presence-Teleop Virt. 1997;6:355-385. doi:10.1162/pres.1997.6.4.355
Speicher M, Hall BD, Nebeling M. What is Mixed Reality? In: Proceedings of the 2019 CHI Conference on Human Factors in Computing Systems. Association for Computing Machinery; 2019:1-15. doi:10.1145/3290605.3300767
Digioia AM, Jaramaz B, Nikou C, et al. Surgical navigation for total hip replacement with the use of hipnav. Oper Tech Orthop. 2000;10:3-8. doi:10.1016/S1048-6666(00)80036-1
Ogawa H, Hasegawa S, Tsukada S, et al. A pilot study of augmented reality technology applied to the acetabular cup placement during total hip arthroplasty. J Arthroplasty. 2018;33:1833-1837. doi:10.1016/j.arth.2018.01.067
Shen F, Chen B, Guo Q, et al. Augmented reality patient-specific reconstruction plate design for pelvic and acetabular fracture surgery. Int J CARS. 2013;8:169-179. doi:10.1007/s11548-012-0775-5
Cho HS, Park YK, Gupta S, et al. Augmented reality in bone tumour resection: an experimental study. Bone Joint Res. 2017;6:137-143. doi:10.1302/2046-3758.63.bjr-2016-0289.r1
Wu X, Liu R, Yu J, et al. Mixed reality technology launches in orthopedic surgery for comprehensive preoperative management of complicated cervical fractures. Surg Innov. 2018;25:421-422. doi:10.1177/1553350618761758
Dossett HG, Arthur JR, Makovicka JL, et al. A randomized controlled trial of kinematically and mechanically aligned total knee arthroplasties: long-term follow-up. J Arthroplasty. 2023;38:S209-S214. doi:10.1016/j.arth.2023.03.065
Kazarian GS, Haddad FS, Donaldson MJ, et al. Implant malalignment may be a risk factor for poor patient-reported outcomes measures (PROMs) following total knee arthroplasty (TKA). J Arthroplasty. 2022;37:S129-S133. doi:10.1016/j.arth.2022.02.087
Peng Y, Arauz P, An S, et al. Does component alignment affect patient reported outcomes following bicruciate retaining total knee arthroplasty? An in vivo three-dimensional analysis. J Knee Surg. 2020;33:798-803. doi:10.1055/s-0039-1688500
D’Lima DD, Urquhart AG, Buehler KO, et al. The effect of the orientation of the acetabular and femoral components on the range of motion of the hip at different head-neck ratios. J Bone Joint Surg Am. 2000;82:315-321. doi:10.2106/00004623-200003000-00003
Yamaguchi M, Akisue T, Bauer TW, et al. The spatial location of impingement in total hip arthroplasty. J Arthroplasty. 2000;15:305-313. doi:10.1016/s0883-5403(00)90601-6
Grammatopoulos G, Alvand A, Monk AP, et al. Surgeons’ accuracy in achieving their desired acetabular component orientation. J Bone Joint Surg. 2016;98:e72. doi:10.2106/JBJS.15.01080
Kennedy JG, Rogers WB, Soffe KE, et al. Effect of acetabular component orientation on recurrent dislocation, pelvic osteolysis, polyethylene wear, and component migration. J Arthroplasty. 1998;13:530-534. doi:10.1016/S0883-5403(98)90052-3
Del Schutte H, Lipman AJ, Bannar SM, et al. Effects of acetabular abduction on cup wear rates in total hip arthroplasty. J Arthroplasty. 1998;13:621-626. doi:10.1016/S0883-5403(98)80003-X
Aresti N, Kassam J, Bartlett D, et al. Primary care management of postoperative shoulder, hip, and knee arthroplasty. BMJ. 2017;359:j4431. doi:10.1136/bmj.j4431
HipInsightTM System. Zimmer Biomet. Accessed September 3, 2025. https://www.zimmerbiomet.com/en/products-and-solutions/zb-edge/mixed-reality-portfolio/hipinsight-system.html
ARVIS. Insight Medical Systems. Accessed September 3, 2025. https://www.insightmedsys.com/arvis
Sun DC, Murphy WS, Amundson AJ, et al. Validation of a novel method of measuring cup orientation using biplanar simultaneous radiographic images. J Arthroplasty. 2023;38:S252-S256. doi:10.1016/j.arth.2023.04.011
Tsukada S, Ogawa H, Nishino M, et al. Augmented reality-assisted femoral bone resection in total knee arthroplasty. JBJS Open Access. 2021;6:e21.00001. doi:10.2106/JBJS.OA.21.00001
Castellarin G, Bori E, Barbieux E, et al. Is total knee arthroplasty surgical performance enhanced using augmented reality? A single-center study on 76 consecutive patients. J Arthroplasty. 2024;39:332-335. doi:10.1016/j.arth.2023.08.013
Choi YJ, Ra HJ. Patient satisfaction after total knee arthroplasty. Knee Surg Relat Res. 2016;28:1. doi:10.5792/ksrr.2016.28.1.1
Hazratwala K, Gouk C, Wilkinson MPR, et al. Navigated functional alignment total knee arthroplasty achieves reliable, reproducible and accurate results with high patient satisfaction. Knee Surg Sports Traumatol Arthrosc. 2023;31:3861-3870. doi:10.1007/s00167-023-07327-w
Knee+. Pixee Medical. Accessed September 3, 2025. https://www.pixee-medical.com/en/products/knee-nexsight/
KNEE | NEXTAR. Nextar. Accessed September 3, 2025. https://nextar.medacta.com/knee
POLARIS AR receives clearance from the U.S. Food and Drug Administration for STELLAR Knee. News release. PRNewswire. November 3, 2023. Accessed September 3, 2025. https://www.prnewswire.com/news-releases/polarisar-receives-clearance-from-the-us-food-and-drug-administration-for-stellar-knee-301976747.html
van Overschelde P, Vansintjan P, Byn P, Lapierre C, van Lysebettens W. Does augmented reality improve clinical outcome in TKA? A prospective observational report. In: The 20th Annual Meeting of the International Society for Computer Assisted Orthopaedic Surgery. 2022:170-174.
Sakellariou E, Alevrogiannis P, Alevrogianni F, et al. Single-center experience with Knee+TM augmented reality navigation system in primary total knee arthroplasty. World J Orthop. 2024;15:247-256. doi:10.5312/wjo.v15.i3.247
León-Muñoz VJ, Moya-Angeler J, López-López M, et al. Integration of square fiducial markers in patient-specific instrumentation and their applicability in knee surgery. J Pers Med. 2023;13:727. doi:10.3390/jpm13050727
Fucentese SF, Koch PP. A novel augmented reality-based surgical guidance system for total knee arthroplasty. Arch Orthop Trauma Surg. 2021;141:2227-2233. doi:10.1007/s00402-021-04204-4
Sabatini L, Ascani D, Vezza D, et al. Novel surgical technique for total knee arthroplasty integrating kinematic alignment and real-time elongation of the ligaments using the NextAR system. J Pers Med. 2024;14:794. doi:10.3390/jpm14080794
Daher M, Ghanimeh J, Otayek J, et al. Augmented reality and shoulder replacement: a state-of-the-art review article. JSES Rev Rep Tech. 2023;3:274-278. doi:10.1016/j.xrrt.2023.01.008
Atmani H, Merienne F, Fofi D, et al. Computer aided surgery system for shoulder prosthesis placement. Comput Aided Surg. 2007;12:60-70. doi:10.3109/10929080701210832
Eichinger JK, Galvin JW. Management of complications after total shoulder arthroplasty. Curr Rev Musculoskelet Med. 2015;8:83-91. doi:10.1007/s12178-014-9251-x
Bonnevialle N, Melis B, Neyton L, et al. Aseptic glenoid loosening or failure in total shoulder arthroplasty: revision with glenoid reimplantation. J Shoulder Elbow Surg. 2013;22:745-751. doi:10.1016/j.jse.2012.08.009
Erickson BJ, Chalmers PN, Denard P, et al. Does commercially available shoulder arthroplasty preoperative planning software agree with surgeon measurements of version, inclination, and subluxation? J Shoulder Elbow Surg. 2021;30:413-420. doi:10.1016/j.jse.2020.05.027
Werner BS, Hudek R, Burkhart KJ, et al. The influence of three-dimensional planning on decision-making in total shoulder arthroplasty. J Shoulder Elbow Surg. 2017;26:1477-1483. doi:10.1016/j.jse.2017.01.006
Blueprint. Stryker. Updated August 2025. Accessed September 3, 2025. https://www.stryker.com/us/en/trauma-and-extremities/products/blueprint.html
NextAR Shoulder. Medacta. Accessed September 3, 2025. https://nextar.medacta.com/shoulder
Baumgarten KM. Accuracy of Blueprint software in predicting range of motion 1 year after reverse total shoulder arthroplasty. J Shoulder Elbow Surg. 2023;32:1088-1094. doi:10.1016/j.jse.2022.12.009
Rojas JT, Jost B, Zipeto C, et al. Glenoid component placement in reverse shoulder arthroplasty assisted with augmented reality through a head-mounted display leads to low deviation between planned and postoperative parameters. J Shoulder Elbow Surg. 2023;32:e587-e596. doi:10.1016/j.jse.2023.05.002
Dey Hazra RO, Paksoy A, Imiolczyk JP, et al. Augmented reality–assisted intraoperative navigation increases precision of glenoid inclination in reverse shoulder arthroplasty. J Shoulder Elbow Surg. 2025;34(2):577-583. doi:10.1016/j.jse.2024.05.039
The introduction of extended reality (XR) to the operating room (OR) has proved promising for enhancing surgical precision and improving patient outcomes. In the field of orthopedic surgery, precise alignment of implants is integral to maintaining functional range of motion and preventing impingement of adjacent neurovascular structures. XR systems have shown promise in arthroplasty including by improving precision and streamlining surgery by allowing surgeons to create 3D preoperative plans that are accessible intraoperatively. This article explores the current applications of XR in arthroplasty, highlights recent advancements and benefits, and describes limitations in comparison to traditional techniques.
Methods
A literature search identified studies involving the use of XR in arthroplasty and current US Food and Drug Administration (FDA)-approved XR systems. Multiple electronic databases were used, including PubMed, Google Scholar, and IEEE Xplore. Search terms included: extended reality, augmented reality, virtual reality, arthroplasty, joint replacement, total knee arthroplasty, total shoulder arthroplasty, and total hip arthroplasty. The study design, intervention details, outcomes, and comparisons with traditional surgical techniques were thematically analyzed, with identification of common ideas associated with XR use in arthroplasty. This narrative report highlights the integration of XR in arthroplasty.
Extended Reality Fundamentals
XR encompasses augmented reality (AR), virtual reality (VR), and mixed reality (MR). AR involves superimposing digitally rendered information and images onto the surgeon’s view of the real world, typically through the use of a headset and smart glasses.1 AR allows the surgeon to move and interact freely within the OR, removing the need for additional screens or devices to display patient information or imaging. VR is a fully immersive simulation using a headset that obstructs the view of the real world but allows the user to move freely within this virtual setting, often with audio or other sensory stimuli. MR combines AR and VR to create a digital model that allows for real-world interaction, with the advantage of adapting information and models in real time.2 Whereas in AR the surgeon can view the data projected from the headset, MR provides the ability to interact with and manipulate the digital content (Figure). Both AR and MR have been adapted for use in the OR, while VR has been adapted for use in surgical planning and training.
Extended Reality Use in Orthopedics
The HipNav system was introduced in 1995 to create preoperative plans that assist surgeons in accurately implanting the acetabular cup during total hip arthroplasty (THA).3 Although not commercially successful, this system spurred surgeons to experiment with XR to improve the accuracy and alignment of orthopedic implants. Systems capable of displaying the desired intraoperative implant placement have flourished, with applications in fracture reduction, arthroplasty, solid tumor resection, and hardware placement.4-7 Accurate alignment has been linked to improvements in patient outcomes.8-10 XR has great potential within the field of arthroplasty, with multiple new systems approved by the FDA and currently available in the US (Table).
Hip Arthroplasty
Orientation of the acetabular cup is a technically challenging part of THA. Accuracy in the anteversion and inclination angles of the acetabular cup is required to maintain implant stability, preserve functional range of motion (ROM), and prevent precocious wear.11,12 Despite preoperative planning, surgeons often overestimate the inclination angle and underestimate anteversion.13 Improper implantation of the acetabular cup can lead to joint instability caused by aseptic loosening, increasing the risk of dislocation and the need for revision surgery.14,15 Dislocations typically present to the emergency department, but primary care practitioners may encounter patients with pain or diminished sensation due to impingement or instability.16
The introduction of XR into the OR has provided the opportunity for real-time navigation and adjustment of the acetabular cup to maximize anteversion and inclination angles. Currently, 2 FDA-approved systems are available for THA: the Zimmer and Surgical Planning Associates HipInsight system, and the Insight Augmented Reality Visualization and Information System (ARVIS). The HipInsight system consists of a hologram projection using the Microsoft HoloLens2 device and optimizes preoperative planning, producing accuracy of anteversion and inclination angles within 3°.17 ARVIS employs existing surgical helmets and 2 mounted tracking cameras to provide navigation intraoperatively. ARVIS has also been approved for use in total knee arthroplasty (TKA) and unicompartmental knee arthroplasty.18
HipInsight has shown utility in increasing the accuracy of acetabular cup placement along with the use of biplanar radiographic scans.19 However, there are no studies validating the efficacy of ARVIS and HipInsight and assessing long-term disease-oriented or patient-oriented outcomes.
Knee Arthroplasty
In the setting of TKA, XR is most effective in ensuring accurate resection of the tibial and femoral components. Achieving the planned femoral coronal, axial, and sagittal angles allows the prosthesis to be on the femoral axis of rotation, improving functional outcomes. XR systems for TKA have been shown to increase the accuracy of distal femoral resection with a limited increase in surgery duration.20,21 For TKA in particular, patients are often less satisfied with the result than surgeons expect.22 Accurate alignment can improve patient satisfaction and reduce return-to-clinic rates for postoperative pain management, a factor that primary care practitioners should consider when recommending a patient for TKA.23
Along with ARVIS, 3 additional XR systems are FDA-approved for use in TKA. The Pixee Medical Knee+ system uses smart glasses and trackers to aid in the positioning of instruments for improved accuracy while allowing real-time navigation.24 The Medacta NextAR Knee’s single-use tracking system allows for intraoperative navigation with the use of AR glasses.25 The Polaris STELLAR Knee uses MR and avoids the need for preoperative imaging by capturing real-time anatomic data.26
The Pixee Medical Knee+ system was commercially available in Europe for several years prior to FDA approval, so more research exists on its efficacy. One study found that the Pixee Medical Knee+ system initially demonstrated an inferior clinical outcome, attributed to the learning curve associated with using the system.27 However, more recent studies have shown its utility in improving alignment, regardless of implant specifications.28,29 The Medacta NextAR Knee system has been shown to improve accuracy of tibial rotation and soft tissue balance and even increase OR efficiency.30,31 The Polaris STELLAR Knee system received FDA approval in 2023; no published research exists on its accuracy and outcomes.26
Shoulder Arthroplasty
Minimally invasive techniques are favored in total shoulder arthroplasty (TSA) due to the vitality of maintaining the surrounding soft tissue to maximize preservation of motility and strength.32 However, this complicates the procedure by decreasing the ability to effectively access and visualize key structures of the shoulder. Accordingly, issues with implant positioning and alignment are more common with TSA than other joint arthroplasties, making XR particularly promising.33 Some studies report that up to 67% of patients experience glenohumeral instability, which can clinically present as weakness, decreased range of motion, and persistent shoulder pain.34,35 The use of preoperative computed tomography to improve understanding of glenoid anatomy and glenohumeral subluxation is becoming increasingly common, and it can be combined with XR to improve accuracy.36,37
Two FDA-approved systems are available. The Stryker Blueprint MR system is used for intraoperative guidance and integration for patient imaging used for preoperative planning. The Medacta NextAR Shoulder system is a parallel of the company’s TKA system. The Stryker Blueprint MR system combines the Microsoft HoloLens 2 headset to display preoperative plans with a secondary display for coordination with the rest of the surgical team.38 Similar to the Medacta NextAR Knee, the Medacta NextAR Shoulder system uses the same single-use tracking system and AR glasses for intraoperative guidance.39
Data on the long-term outcomes of using these systems are still limited, but the Stryker Blueprint MR system has not been shown to accurately predict postoperative ROM.40 Cadaveric studies have demonstrated that the Medacta NextAR Shoulder system can provide accurate inclination, retroversion, entry point, depth, and rotation values based on the preoperative planned values.41,42 However, this accuracy has yet to be confirmed in vivo, and the impact of using XR in TSA on long-term outcomes is still unknown.
Challenges and Limitations
Though XR has proven to be promising in arthroplasty, several limitations regarding widespread implementation exist. In particular, there is a steep learning curve associated with the use of XR systems, which can cause increased operative time and even initial inferior outcomes, as demonstrated with the Pixee Medical Knee+ system. The need for extensive practice and training prior to use could delay widespread adoption and may cause discrepancies in surgical outcomes. Unfamiliarity with the system and technological difficulties that may require troubleshooting can also increase operative time, particularly for surgeons new to using the XR system. Though intraoperative navigation is expected to improve accuracy of implant alignment, its added complexity may also result in longer surgeries.
In addition to the steep learning curve and increased operative time, there is a high upfront cost associated with XR systems. Exact costs of XR systems are not typically disclosed, but available estimates suggest an average sales price of about $1000 per case. Given the proprietary nature of these technologies, publicly available cost data are limited, making it challenging to fully assess the financial burden on health care institutions. Though some systems, such as ARVIS, can be integrated with existing surgical helmets, many require the purchase of AR glasses and secondary displays. This can cause further variation in the total expense for each system. In low-resource settings, this represents a significant challenge to widespread implementation. To justify this cost, additional research on long-term patient outcomes is needed to ensure the benefits of XR systems outweigh the cost.
Although early studies on XR systems in arthroplasty have shown improvements in precision and short-term outcomes, long-term data regarding effectiveness remains. Even systems such as ARVIS and HipInsight have limited long-term follow-up, making it difficult to assess whether the improved accuracy with these XR systems translates into improved patient outcomes compared with traditional arthroplasty.
CONCLUSIONS
XR technologies have shown significant potential in enhancing precision and patient outcomes. Through the integration of XR in the OR, surgeons can visualize preoperative plans and even make intraoperative changes, with the benefit of improving implant alignment.
There are some disadvantages to its use, however, including high cost and increased operative time. Despite this, the integration of XR into surgical practice can deliver more precise implant alignment and address other challenges faced with conventional techniques. As these technologies evolve and studies on long-term outcomes validate their utility, XR has the potential to transform the field of arthroplasty.
The introduction of extended reality (XR) to the operating room (OR) has proved promising for enhancing surgical precision and improving patient outcomes. In the field of orthopedic surgery, precise alignment of implants is integral to maintaining functional range of motion and preventing impingement of adjacent neurovascular structures. XR systems have shown promise in arthroplasty including by improving precision and streamlining surgery by allowing surgeons to create 3D preoperative plans that are accessible intraoperatively. This article explores the current applications of XR in arthroplasty, highlights recent advancements and benefits, and describes limitations in comparison to traditional techniques.
Methods
A literature search identified studies involving the use of XR in arthroplasty and current US Food and Drug Administration (FDA)-approved XR systems. Multiple electronic databases were used, including PubMed, Google Scholar, and IEEE Xplore. Search terms included: extended reality, augmented reality, virtual reality, arthroplasty, joint replacement, total knee arthroplasty, total shoulder arthroplasty, and total hip arthroplasty. The study design, intervention details, outcomes, and comparisons with traditional surgical techniques were thematically analyzed, with identification of common ideas associated with XR use in arthroplasty. This narrative report highlights the integration of XR in arthroplasty.
Extended Reality Fundamentals
XR encompasses augmented reality (AR), virtual reality (VR), and mixed reality (MR). AR involves superimposing digitally rendered information and images onto the surgeon’s view of the real world, typically through the use of a headset and smart glasses.1 AR allows the surgeon to move and interact freely within the OR, removing the need for additional screens or devices to display patient information or imaging. VR is a fully immersive simulation using a headset that obstructs the view of the real world but allows the user to move freely within this virtual setting, often with audio or other sensory stimuli. MR combines AR and VR to create a digital model that allows for real-world interaction, with the advantage of adapting information and models in real time.2 Whereas in AR the surgeon can view the data projected from the headset, MR provides the ability to interact with and manipulate the digital content (Figure). Both AR and MR have been adapted for use in the OR, while VR has been adapted for use in surgical planning and training.
Extended Reality Use in Orthopedics
The HipNav system was introduced in 1995 to create preoperative plans that assist surgeons in accurately implanting the acetabular cup during total hip arthroplasty (THA).3 Although not commercially successful, this system spurred surgeons to experiment with XR to improve the accuracy and alignment of orthopedic implants. Systems capable of displaying the desired intraoperative implant placement have flourished, with applications in fracture reduction, arthroplasty, solid tumor resection, and hardware placement.4-7 Accurate alignment has been linked to improvements in patient outcomes.8-10 XR has great potential within the field of arthroplasty, with multiple new systems approved by the FDA and currently available in the US (Table).
Hip Arthroplasty
Orientation of the acetabular cup is a technically challenging part of THA. Accuracy in the anteversion and inclination angles of the acetabular cup is required to maintain implant stability, preserve functional range of motion (ROM), and prevent precocious wear.11,12 Despite preoperative planning, surgeons often overestimate the inclination angle and underestimate anteversion.13 Improper implantation of the acetabular cup can lead to joint instability caused by aseptic loosening, increasing the risk of dislocation and the need for revision surgery.14,15 Dislocations typically present to the emergency department, but primary care practitioners may encounter patients with pain or diminished sensation due to impingement or instability.16
The introduction of XR into the OR has provided the opportunity for real-time navigation and adjustment of the acetabular cup to maximize anteversion and inclination angles. Currently, 2 FDA-approved systems are available for THA: the Zimmer and Surgical Planning Associates HipInsight system, and the Insight Augmented Reality Visualization and Information System (ARVIS). The HipInsight system consists of a hologram projection using the Microsoft HoloLens2 device and optimizes preoperative planning, producing accuracy of anteversion and inclination angles within 3°.17 ARVIS employs existing surgical helmets and 2 mounted tracking cameras to provide navigation intraoperatively. ARVIS has also been approved for use in total knee arthroplasty (TKA) and unicompartmental knee arthroplasty.18
HipInsight has shown utility in increasing the accuracy of acetabular cup placement along with the use of biplanar radiographic scans.19 However, there are no studies validating the efficacy of ARVIS and HipInsight and assessing long-term disease-oriented or patient-oriented outcomes.
Knee Arthroplasty
In the setting of TKA, XR is most effective in ensuring accurate resection of the tibial and femoral components. Achieving the planned femoral coronal, axial, and sagittal angles allows the prosthesis to be on the femoral axis of rotation, improving functional outcomes. XR systems for TKA have been shown to increase the accuracy of distal femoral resection with a limited increase in surgery duration.20,21 For TKA in particular, patients are often less satisfied with the result than surgeons expect.22 Accurate alignment can improve patient satisfaction and reduce return-to-clinic rates for postoperative pain management, a factor that primary care practitioners should consider when recommending a patient for TKA.23
Along with ARVIS, 3 additional XR systems are FDA-approved for use in TKA. The Pixee Medical Knee+ system uses smart glasses and trackers to aid in the positioning of instruments for improved accuracy while allowing real-time navigation.24 The Medacta NextAR Knee’s single-use tracking system allows for intraoperative navigation with the use of AR glasses.25 The Polaris STELLAR Knee uses MR and avoids the need for preoperative imaging by capturing real-time anatomic data.26
The Pixee Medical Knee+ system was commercially available in Europe for several years prior to FDA approval, so more research exists on its efficacy. One study found that the Pixee Medical Knee+ system initially demonstrated an inferior clinical outcome, attributed to the learning curve associated with using the system.27 However, more recent studies have shown its utility in improving alignment, regardless of implant specifications.28,29 The Medacta NextAR Knee system has been shown to improve accuracy of tibial rotation and soft tissue balance and even increase OR efficiency.30,31 The Polaris STELLAR Knee system received FDA approval in 2023; no published research exists on its accuracy and outcomes.26
Shoulder Arthroplasty
Minimally invasive techniques are favored in total shoulder arthroplasty (TSA) due to the vitality of maintaining the surrounding soft tissue to maximize preservation of motility and strength.32 However, this complicates the procedure by decreasing the ability to effectively access and visualize key structures of the shoulder. Accordingly, issues with implant positioning and alignment are more common with TSA than other joint arthroplasties, making XR particularly promising.33 Some studies report that up to 67% of patients experience glenohumeral instability, which can clinically present as weakness, decreased range of motion, and persistent shoulder pain.34,35 The use of preoperative computed tomography to improve understanding of glenoid anatomy and glenohumeral subluxation is becoming increasingly common, and it can be combined with XR to improve accuracy.36,37
Two FDA-approved systems are available. The Stryker Blueprint MR system is used for intraoperative guidance and integration for patient imaging used for preoperative planning. The Medacta NextAR Shoulder system is a parallel of the company’s TKA system. The Stryker Blueprint MR system combines the Microsoft HoloLens 2 headset to display preoperative plans with a secondary display for coordination with the rest of the surgical team.38 Similar to the Medacta NextAR Knee, the Medacta NextAR Shoulder system uses the same single-use tracking system and AR glasses for intraoperative guidance.39
Data on the long-term outcomes of using these systems are still limited, but the Stryker Blueprint MR system has not been shown to accurately predict postoperative ROM.40 Cadaveric studies have demonstrated that the Medacta NextAR Shoulder system can provide accurate inclination, retroversion, entry point, depth, and rotation values based on the preoperative planned values.41,42 However, this accuracy has yet to be confirmed in vivo, and the impact of using XR in TSA on long-term outcomes is still unknown.
Challenges and Limitations
Though XR has proven to be promising in arthroplasty, several limitations regarding widespread implementation exist. In particular, there is a steep learning curve associated with the use of XR systems, which can cause increased operative time and even initial inferior outcomes, as demonstrated with the Pixee Medical Knee+ system. The need for extensive practice and training prior to use could delay widespread adoption and may cause discrepancies in surgical outcomes. Unfamiliarity with the system and technological difficulties that may require troubleshooting can also increase operative time, particularly for surgeons new to using the XR system. Though intraoperative navigation is expected to improve accuracy of implant alignment, its added complexity may also result in longer surgeries.
In addition to the steep learning curve and increased operative time, there is a high upfront cost associated with XR systems. Exact costs of XR systems are not typically disclosed, but available estimates suggest an average sales price of about $1000 per case. Given the proprietary nature of these technologies, publicly available cost data are limited, making it challenging to fully assess the financial burden on health care institutions. Though some systems, such as ARVIS, can be integrated with existing surgical helmets, many require the purchase of AR glasses and secondary displays. This can cause further variation in the total expense for each system. In low-resource settings, this represents a significant challenge to widespread implementation. To justify this cost, additional research on long-term patient outcomes is needed to ensure the benefits of XR systems outweigh the cost.
Although early studies on XR systems in arthroplasty have shown improvements in precision and short-term outcomes, long-term data regarding effectiveness remains. Even systems such as ARVIS and HipInsight have limited long-term follow-up, making it difficult to assess whether the improved accuracy with these XR systems translates into improved patient outcomes compared with traditional arthroplasty.
CONCLUSIONS
XR technologies have shown significant potential in enhancing precision and patient outcomes. Through the integration of XR in the OR, surgeons can visualize preoperative plans and even make intraoperative changes, with the benefit of improving implant alignment.
There are some disadvantages to its use, however, including high cost and increased operative time. Despite this, the integration of XR into surgical practice can deliver more precise implant alignment and address other challenges faced with conventional techniques. As these technologies evolve and studies on long-term outcomes validate their utility, XR has the potential to transform the field of arthroplasty.
Azuma RT. A survey of augmented reality. Presence-Teleop Virt. 1997;6:355-385. doi:10.1162/pres.1997.6.4.355
Speicher M, Hall BD, Nebeling M. What is Mixed Reality? In: Proceedings of the 2019 CHI Conference on Human Factors in Computing Systems. Association for Computing Machinery; 2019:1-15. doi:10.1145/3290605.3300767
Digioia AM, Jaramaz B, Nikou C, et al. Surgical navigation for total hip replacement with the use of hipnav. Oper Tech Orthop. 2000;10:3-8. doi:10.1016/S1048-6666(00)80036-1
Ogawa H, Hasegawa S, Tsukada S, et al. A pilot study of augmented reality technology applied to the acetabular cup placement during total hip arthroplasty. J Arthroplasty. 2018;33:1833-1837. doi:10.1016/j.arth.2018.01.067
Shen F, Chen B, Guo Q, et al. Augmented reality patient-specific reconstruction plate design for pelvic and acetabular fracture surgery. Int J CARS. 2013;8:169-179. doi:10.1007/s11548-012-0775-5
Cho HS, Park YK, Gupta S, et al. Augmented reality in bone tumour resection: an experimental study. Bone Joint Res. 2017;6:137-143. doi:10.1302/2046-3758.63.bjr-2016-0289.r1
Wu X, Liu R, Yu J, et al. Mixed reality technology launches in orthopedic surgery for comprehensive preoperative management of complicated cervical fractures. Surg Innov. 2018;25:421-422. doi:10.1177/1553350618761758
Dossett HG, Arthur JR, Makovicka JL, et al. A randomized controlled trial of kinematically and mechanically aligned total knee arthroplasties: long-term follow-up. J Arthroplasty. 2023;38:S209-S214. doi:10.1016/j.arth.2023.03.065
Kazarian GS, Haddad FS, Donaldson MJ, et al. Implant malalignment may be a risk factor for poor patient-reported outcomes measures (PROMs) following total knee arthroplasty (TKA). J Arthroplasty. 2022;37:S129-S133. doi:10.1016/j.arth.2022.02.087
Peng Y, Arauz P, An S, et al. Does component alignment affect patient reported outcomes following bicruciate retaining total knee arthroplasty? An in vivo three-dimensional analysis. J Knee Surg. 2020;33:798-803. doi:10.1055/s-0039-1688500
D’Lima DD, Urquhart AG, Buehler KO, et al. The effect of the orientation of the acetabular and femoral components on the range of motion of the hip at different head-neck ratios. J Bone Joint Surg Am. 2000;82:315-321. doi:10.2106/00004623-200003000-00003
Yamaguchi M, Akisue T, Bauer TW, et al. The spatial location of impingement in total hip arthroplasty. J Arthroplasty. 2000;15:305-313. doi:10.1016/s0883-5403(00)90601-6
Grammatopoulos G, Alvand A, Monk AP, et al. Surgeons’ accuracy in achieving their desired acetabular component orientation. J Bone Joint Surg. 2016;98:e72. doi:10.2106/JBJS.15.01080
Kennedy JG, Rogers WB, Soffe KE, et al. Effect of acetabular component orientation on recurrent dislocation, pelvic osteolysis, polyethylene wear, and component migration. J Arthroplasty. 1998;13:530-534. doi:10.1016/S0883-5403(98)90052-3
Del Schutte H, Lipman AJ, Bannar SM, et al. Effects of acetabular abduction on cup wear rates in total hip arthroplasty. J Arthroplasty. 1998;13:621-626. doi:10.1016/S0883-5403(98)80003-X
Aresti N, Kassam J, Bartlett D, et al. Primary care management of postoperative shoulder, hip, and knee arthroplasty. BMJ. 2017;359:j4431. doi:10.1136/bmj.j4431
HipInsightTM System. Zimmer Biomet. Accessed September 3, 2025. https://www.zimmerbiomet.com/en/products-and-solutions/zb-edge/mixed-reality-portfolio/hipinsight-system.html
ARVIS. Insight Medical Systems. Accessed September 3, 2025. https://www.insightmedsys.com/arvis
Sun DC, Murphy WS, Amundson AJ, et al. Validation of a novel method of measuring cup orientation using biplanar simultaneous radiographic images. J Arthroplasty. 2023;38:S252-S256. doi:10.1016/j.arth.2023.04.011
Tsukada S, Ogawa H, Nishino M, et al. Augmented reality-assisted femoral bone resection in total knee arthroplasty. JBJS Open Access. 2021;6:e21.00001. doi:10.2106/JBJS.OA.21.00001
Castellarin G, Bori E, Barbieux E, et al. Is total knee arthroplasty surgical performance enhanced using augmented reality? A single-center study on 76 consecutive patients. J Arthroplasty. 2024;39:332-335. doi:10.1016/j.arth.2023.08.013
Choi YJ, Ra HJ. Patient satisfaction after total knee arthroplasty. Knee Surg Relat Res. 2016;28:1. doi:10.5792/ksrr.2016.28.1.1
Hazratwala K, Gouk C, Wilkinson MPR, et al. Navigated functional alignment total knee arthroplasty achieves reliable, reproducible and accurate results with high patient satisfaction. Knee Surg Sports Traumatol Arthrosc. 2023;31:3861-3870. doi:10.1007/s00167-023-07327-w
Knee+. Pixee Medical. Accessed September 3, 2025. https://www.pixee-medical.com/en/products/knee-nexsight/
KNEE | NEXTAR. Nextar. Accessed September 3, 2025. https://nextar.medacta.com/knee
POLARIS AR receives clearance from the U.S. Food and Drug Administration for STELLAR Knee. News release. PRNewswire. November 3, 2023. Accessed September 3, 2025. https://www.prnewswire.com/news-releases/polarisar-receives-clearance-from-the-us-food-and-drug-administration-for-stellar-knee-301976747.html
van Overschelde P, Vansintjan P, Byn P, Lapierre C, van Lysebettens W. Does augmented reality improve clinical outcome in TKA? A prospective observational report. In: The 20th Annual Meeting of the International Society for Computer Assisted Orthopaedic Surgery. 2022:170-174.
Sakellariou E, Alevrogiannis P, Alevrogianni F, et al. Single-center experience with Knee+TM augmented reality navigation system in primary total knee arthroplasty. World J Orthop. 2024;15:247-256. doi:10.5312/wjo.v15.i3.247
León-Muñoz VJ, Moya-Angeler J, López-López M, et al. Integration of square fiducial markers in patient-specific instrumentation and their applicability in knee surgery. J Pers Med. 2023;13:727. doi:10.3390/jpm13050727
Fucentese SF, Koch PP. A novel augmented reality-based surgical guidance system for total knee arthroplasty. Arch Orthop Trauma Surg. 2021;141:2227-2233. doi:10.1007/s00402-021-04204-4
Sabatini L, Ascani D, Vezza D, et al. Novel surgical technique for total knee arthroplasty integrating kinematic alignment and real-time elongation of the ligaments using the NextAR system. J Pers Med. 2024;14:794. doi:10.3390/jpm14080794
Daher M, Ghanimeh J, Otayek J, et al. Augmented reality and shoulder replacement: a state-of-the-art review article. JSES Rev Rep Tech. 2023;3:274-278. doi:10.1016/j.xrrt.2023.01.008
Atmani H, Merienne F, Fofi D, et al. Computer aided surgery system for shoulder prosthesis placement. Comput Aided Surg. 2007;12:60-70. doi:10.3109/10929080701210832
Eichinger JK, Galvin JW. Management of complications after total shoulder arthroplasty. Curr Rev Musculoskelet Med. 2015;8:83-91. doi:10.1007/s12178-014-9251-x
Bonnevialle N, Melis B, Neyton L, et al. Aseptic glenoid loosening or failure in total shoulder arthroplasty: revision with glenoid reimplantation. J Shoulder Elbow Surg. 2013;22:745-751. doi:10.1016/j.jse.2012.08.009
Erickson BJ, Chalmers PN, Denard P, et al. Does commercially available shoulder arthroplasty preoperative planning software agree with surgeon measurements of version, inclination, and subluxation? J Shoulder Elbow Surg. 2021;30:413-420. doi:10.1016/j.jse.2020.05.027
Werner BS, Hudek R, Burkhart KJ, et al. The influence of three-dimensional planning on decision-making in total shoulder arthroplasty. J Shoulder Elbow Surg. 2017;26:1477-1483. doi:10.1016/j.jse.2017.01.006
Blueprint. Stryker. Updated August 2025. Accessed September 3, 2025. https://www.stryker.com/us/en/trauma-and-extremities/products/blueprint.html
NextAR Shoulder. Medacta. Accessed September 3, 2025. https://nextar.medacta.com/shoulder
Baumgarten KM. Accuracy of Blueprint software in predicting range of motion 1 year after reverse total shoulder arthroplasty. J Shoulder Elbow Surg. 2023;32:1088-1094. doi:10.1016/j.jse.2022.12.009
Rojas JT, Jost B, Zipeto C, et al. Glenoid component placement in reverse shoulder arthroplasty assisted with augmented reality through a head-mounted display leads to low deviation between planned and postoperative parameters. J Shoulder Elbow Surg. 2023;32:e587-e596. doi:10.1016/j.jse.2023.05.002
Dey Hazra RO, Paksoy A, Imiolczyk JP, et al. Augmented reality–assisted intraoperative navigation increases precision of glenoid inclination in reverse shoulder arthroplasty. J Shoulder Elbow Surg. 2025;34(2):577-583. doi:10.1016/j.jse.2024.05.039
Azuma RT. A survey of augmented reality. Presence-Teleop Virt. 1997;6:355-385. doi:10.1162/pres.1997.6.4.355
Speicher M, Hall BD, Nebeling M. What is Mixed Reality? In: Proceedings of the 2019 CHI Conference on Human Factors in Computing Systems. Association for Computing Machinery; 2019:1-15. doi:10.1145/3290605.3300767
Digioia AM, Jaramaz B, Nikou C, et al. Surgical navigation for total hip replacement with the use of hipnav. Oper Tech Orthop. 2000;10:3-8. doi:10.1016/S1048-6666(00)80036-1
Ogawa H, Hasegawa S, Tsukada S, et al. A pilot study of augmented reality technology applied to the acetabular cup placement during total hip arthroplasty. J Arthroplasty. 2018;33:1833-1837. doi:10.1016/j.arth.2018.01.067
Shen F, Chen B, Guo Q, et al. Augmented reality patient-specific reconstruction plate design for pelvic and acetabular fracture surgery. Int J CARS. 2013;8:169-179. doi:10.1007/s11548-012-0775-5
Cho HS, Park YK, Gupta S, et al. Augmented reality in bone tumour resection: an experimental study. Bone Joint Res. 2017;6:137-143. doi:10.1302/2046-3758.63.bjr-2016-0289.r1
Wu X, Liu R, Yu J, et al. Mixed reality technology launches in orthopedic surgery for comprehensive preoperative management of complicated cervical fractures. Surg Innov. 2018;25:421-422. doi:10.1177/1553350618761758
Dossett HG, Arthur JR, Makovicka JL, et al. A randomized controlled trial of kinematically and mechanically aligned total knee arthroplasties: long-term follow-up. J Arthroplasty. 2023;38:S209-S214. doi:10.1016/j.arth.2023.03.065
Kazarian GS, Haddad FS, Donaldson MJ, et al. Implant malalignment may be a risk factor for poor patient-reported outcomes measures (PROMs) following total knee arthroplasty (TKA). J Arthroplasty. 2022;37:S129-S133. doi:10.1016/j.arth.2022.02.087
Peng Y, Arauz P, An S, et al. Does component alignment affect patient reported outcomes following bicruciate retaining total knee arthroplasty? An in vivo three-dimensional analysis. J Knee Surg. 2020;33:798-803. doi:10.1055/s-0039-1688500
D’Lima DD, Urquhart AG, Buehler KO, et al. The effect of the orientation of the acetabular and femoral components on the range of motion of the hip at different head-neck ratios. J Bone Joint Surg Am. 2000;82:315-321. doi:10.2106/00004623-200003000-00003
Yamaguchi M, Akisue T, Bauer TW, et al. The spatial location of impingement in total hip arthroplasty. J Arthroplasty. 2000;15:305-313. doi:10.1016/s0883-5403(00)90601-6
Grammatopoulos G, Alvand A, Monk AP, et al. Surgeons’ accuracy in achieving their desired acetabular component orientation. J Bone Joint Surg. 2016;98:e72. doi:10.2106/JBJS.15.01080
Kennedy JG, Rogers WB, Soffe KE, et al. Effect of acetabular component orientation on recurrent dislocation, pelvic osteolysis, polyethylene wear, and component migration. J Arthroplasty. 1998;13:530-534. doi:10.1016/S0883-5403(98)90052-3
Del Schutte H, Lipman AJ, Bannar SM, et al. Effects of acetabular abduction on cup wear rates in total hip arthroplasty. J Arthroplasty. 1998;13:621-626. doi:10.1016/S0883-5403(98)80003-X
Aresti N, Kassam J, Bartlett D, et al. Primary care management of postoperative shoulder, hip, and knee arthroplasty. BMJ. 2017;359:j4431. doi:10.1136/bmj.j4431
HipInsightTM System. Zimmer Biomet. Accessed September 3, 2025. https://www.zimmerbiomet.com/en/products-and-solutions/zb-edge/mixed-reality-portfolio/hipinsight-system.html
ARVIS. Insight Medical Systems. Accessed September 3, 2025. https://www.insightmedsys.com/arvis
Sun DC, Murphy WS, Amundson AJ, et al. Validation of a novel method of measuring cup orientation using biplanar simultaneous radiographic images. J Arthroplasty. 2023;38:S252-S256. doi:10.1016/j.arth.2023.04.011
Tsukada S, Ogawa H, Nishino M, et al. Augmented reality-assisted femoral bone resection in total knee arthroplasty. JBJS Open Access. 2021;6:e21.00001. doi:10.2106/JBJS.OA.21.00001
Castellarin G, Bori E, Barbieux E, et al. Is total knee arthroplasty surgical performance enhanced using augmented reality? A single-center study on 76 consecutive patients. J Arthroplasty. 2024;39:332-335. doi:10.1016/j.arth.2023.08.013
Choi YJ, Ra HJ. Patient satisfaction after total knee arthroplasty. Knee Surg Relat Res. 2016;28:1. doi:10.5792/ksrr.2016.28.1.1
Hazratwala K, Gouk C, Wilkinson MPR, et al. Navigated functional alignment total knee arthroplasty achieves reliable, reproducible and accurate results with high patient satisfaction. Knee Surg Sports Traumatol Arthrosc. 2023;31:3861-3870. doi:10.1007/s00167-023-07327-w
Knee+. Pixee Medical. Accessed September 3, 2025. https://www.pixee-medical.com/en/products/knee-nexsight/
KNEE | NEXTAR. Nextar. Accessed September 3, 2025. https://nextar.medacta.com/knee
POLARIS AR receives clearance from the U.S. Food and Drug Administration for STELLAR Knee. News release. PRNewswire. November 3, 2023. Accessed September 3, 2025. https://www.prnewswire.com/news-releases/polarisar-receives-clearance-from-the-us-food-and-drug-administration-for-stellar-knee-301976747.html
van Overschelde P, Vansintjan P, Byn P, Lapierre C, van Lysebettens W. Does augmented reality improve clinical outcome in TKA? A prospective observational report. In: The 20th Annual Meeting of the International Society for Computer Assisted Orthopaedic Surgery. 2022:170-174.
Sakellariou E, Alevrogiannis P, Alevrogianni F, et al. Single-center experience with Knee+TM augmented reality navigation system in primary total knee arthroplasty. World J Orthop. 2024;15:247-256. doi:10.5312/wjo.v15.i3.247
León-Muñoz VJ, Moya-Angeler J, López-López M, et al. Integration of square fiducial markers in patient-specific instrumentation and their applicability in knee surgery. J Pers Med. 2023;13:727. doi:10.3390/jpm13050727
Fucentese SF, Koch PP. A novel augmented reality-based surgical guidance system for total knee arthroplasty. Arch Orthop Trauma Surg. 2021;141:2227-2233. doi:10.1007/s00402-021-04204-4
Sabatini L, Ascani D, Vezza D, et al. Novel surgical technique for total knee arthroplasty integrating kinematic alignment and real-time elongation of the ligaments using the NextAR system. J Pers Med. 2024;14:794. doi:10.3390/jpm14080794
Daher M, Ghanimeh J, Otayek J, et al. Augmented reality and shoulder replacement: a state-of-the-art review article. JSES Rev Rep Tech. 2023;3:274-278. doi:10.1016/j.xrrt.2023.01.008
Atmani H, Merienne F, Fofi D, et al. Computer aided surgery system for shoulder prosthesis placement. Comput Aided Surg. 2007;12:60-70. doi:10.3109/10929080701210832
Eichinger JK, Galvin JW. Management of complications after total shoulder arthroplasty. Curr Rev Musculoskelet Med. 2015;8:83-91. doi:10.1007/s12178-014-9251-x
Bonnevialle N, Melis B, Neyton L, et al. Aseptic glenoid loosening or failure in total shoulder arthroplasty: revision with glenoid reimplantation. J Shoulder Elbow Surg. 2013;22:745-751. doi:10.1016/j.jse.2012.08.009
Erickson BJ, Chalmers PN, Denard P, et al. Does commercially available shoulder arthroplasty preoperative planning software agree with surgeon measurements of version, inclination, and subluxation? J Shoulder Elbow Surg. 2021;30:413-420. doi:10.1016/j.jse.2020.05.027
Werner BS, Hudek R, Burkhart KJ, et al. The influence of three-dimensional planning on decision-making in total shoulder arthroplasty. J Shoulder Elbow Surg. 2017;26:1477-1483. doi:10.1016/j.jse.2017.01.006
Blueprint. Stryker. Updated August 2025. Accessed September 3, 2025. https://www.stryker.com/us/en/trauma-and-extremities/products/blueprint.html
NextAR Shoulder. Medacta. Accessed September 3, 2025. https://nextar.medacta.com/shoulder
Baumgarten KM. Accuracy of Blueprint software in predicting range of motion 1 year after reverse total shoulder arthroplasty. J Shoulder Elbow Surg. 2023;32:1088-1094. doi:10.1016/j.jse.2022.12.009
Rojas JT, Jost B, Zipeto C, et al. Glenoid component placement in reverse shoulder arthroplasty assisted with augmented reality through a head-mounted display leads to low deviation between planned and postoperative parameters. J Shoulder Elbow Surg. 2023;32:e587-e596. doi:10.1016/j.jse.2023.05.002
Dey Hazra RO, Paksoy A, Imiolczyk JP, et al. Augmented reality–assisted intraoperative navigation increases precision of glenoid inclination in reverse shoulder arthroplasty. J Shoulder Elbow Surg. 2025;34(2):577-583. doi:10.1016/j.jse.2024.05.039
The Integration of Extended Reality in Arthroplasty: Reviewing Technological Progress and Clinical Benefits
The Integration of Extended Reality in Arthroplasty: Reviewing Technological Progress and Clinical Benefits
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
More than 40 million surgeries are performed annually in the United States, of which > 18 million are orthopedic, including > 1 million emergency orthopedic surgeries and > 2 million joint replacements.1-4 Notably, > 50% of patients undergoing orthopedic surgery are aged ≥ 65 years, a demographic shift driven by longer life expectancies and an increasing number of older adults remaining physically active for extended periods.5 Osteoarthritis, the most common joint disease, affects 10% of men and 18% of women aged > 60 years, often necessitating an orthopedic joint replacement.6 Perioperative morbidity and mortality are 2.9- to 6.7-times higher in older adults compared with younger adults.7 These risks include infection, venous thromboembolism (VTE), pressure ulcers, reduced mobility, and increased mortality. Due to the high incidence of these complications in older surgical patients, special perioperative protocols and considerations are needed when preparing an older patient for surgery. This review aims to establish concrete considerations and guidelines for perioperative management.
METHODOLOGY
A literature review of PubMed, Google Scholar, and IEEE Xplore identified research on perioperative challenges in geriatric orthopedic surgery. Keywords included geriatrics and orthopedic surgery, perioperative care in geriatric populations, and orthopedic perioperative care. Inclusion criteria were strictly defined to ensure relevance to the geriatric population, with studies focusing on patients aged ≥ 65 years. Exclusion criteria were applied to remove studies that did not involve geriatric populations or orthopedic surgeries or that lacked a clear perioperative focus. Studies were analyzed for design, interventions, and outcomes. Special attention was given to identifying common challenges and trends related to perioperative considerations. We developed a narrative report providing a comprehensive overview of the current understanding of perioperative care for geriatric orthopedic patients to offer practical recommendations for clinicians to use in their practice.
RESULTS
Consistent with the narrative review methodology described, the literature search yielded a broad range of publications addressing perioperative considerations in geriatric orthopedic patients. Articles were screened for relevance to patients aged ≥ 65 years undergoing orthopedic surgery and for applicability to perioperative optimization and postoperative outcomes. Given the heterogeneity in study design, population characteristics, and outcome reporting, findings are presented descriptively rather than being quantitatively pooled. Studies not focused on geriatric populations, orthopedic procedures, or perioperative management were excluded. Key themes included multimorbidity and comorbidity optimization, age-related physiologic changes, frailty assessment and fracture risk stratification, nutritional and bone health management, mechanism of injury considerations, prevention of postoperative complications, and the role of multidisciplinary perioperative care.
Unique Physiological Challenges
The aging process induces a range of physiological changes that can increase morbidity and mortality following surgery. One of the most essential elements to surgical recovery is wound healing, as impairments in this process can lead to adverse events, including infection, cosmetic deformity, and wound dehiscence. The general paradigm of aging involves cell senescence resulting in slower or disorganized functional capacity of these cells.8 While wound healing in older individuals was once thought to be defective, recent research has demonstrated that this process is not absent, but delayed.9
Wound healing is a tightly regulated and evolutionarily conserved process that proceeds through 3 main phases: inflammation, proliferation, and remodeling. Re-epithelialization begins with the migration of epithelial cells from hair follicles, sweat glands, or wound margins (depending on wound depth) and is influenced by oxygen levels, moisture, and growth factors.9 Several characteristics of aged skin contribute to the delayed healing process. Aged skin has fewer hair follicles and eccrine sweat glands, as well as decreased follicle thickness.10 This results in fewer proliferating cells for wound healing and lower amounts of sebum production for skin moisture.11 Furthermore, aged fibroblasts are fewer in number and less effective in synthesizing extracellular matrices, resulting in slower and less tensile wound healing.12 Additionally, microvascular changes associated with aging result in disorganized vasculature, which impairs oxygen delivery to the wound bed and diminishes the influx of proinflammatory cells necessary for effective healing.13 These senescent traits of aged skin contribute to the delayed wound healing process found in geriatric patients.
Compounding these age-related factors is the prevalence of multimorbidity, or coexisting chronic diagnoses, in 55% to 98% of older patients.14 Common comorbidities include peripheral arterial disease, chronic venous insufficiency, type 1 and type 2 diabetes, neoplasms, atherosclerotic disease, and hypertension. Older patients are more likely to be prescribed corticosteroids and chemotherapeutic agents that impair the function of inflammatory cells necessary for wound healing.15,16 Additionally, decreased mobility is more common in geriatric patients, which can increase the risk of wound formation, particularly pressure ulcers.17
Perioperative Considerations
All surgical patients undergo a formal or informal preoperative evaluation to assess their fitness for surgery, with the goal of minimizing both anesthesia-related risks and postoperative complications. A widely used tool in this assessment is the American Society of Anesthesiologists (ASA) physical status classification, which stratifies patients into 6 categories based on their medical history and overall health status.18 Classes range from healthy patients (Class I) to organ donors who are brain-dead (Class VI).
Cardiac optimization is an essential component of preoperative evaluation for older adults due to their higher risk of underlying cardiovascular disease.19 This process involves an in-depth review of the patient’s cardiac history, including the timing and nature of any prior interventions and the recurrence rate. Functional capacity is assessed through metabolic equivalents, where a threshold of > 4 metabolic equivalents (the ability to walk up a flight of stairs) is considered adequate for surgery. Risk is assessed based on the specific surgical procedure, and nonemergent orthopedic procedures are considered intermediate risk. If a patient is deemed high risk at any stage of this evaluation, further cardiac testing is indicated.
Pulmonary optimization is typically necessary for geriatric patients, who are more likely to have conditions such as chronic obstructive pulmonary disease or interstitial lung disease.14,20 In patients without severe systemic lung disease, pulmonary optimization involves assessing the functional expiratory volume and diffusing capacity for carbon monoxide. In addition, aggressive modification of risk factors, such as smoking cessation, is strongly recommended.
Additional perioperative conditions are disease-specific and involve evaluation of comorbid illnesses and recognition of absolute contraindications to noncardiac surgery. For instance, an ejection fraction of < 35%, a history of myocardial infarction within 6 months, or active diabetic ketoacidosis are all absolute contraindications to elective surgery. For orthopedic procedures, additional contraindications include symptomatic bacteremia, active joint or local tissue infection, severe malnutrition, uncontrolled metabolic syndrome or chronic disease, untreated immunodeficiency, and active deep venous thrombosis (DVT) or pulmonary embolism.21
Bone Health and Nutrition
In the context of orthopedic surgery, the hallmark of clinically defined optimal bone health is a musculoskeletal system that provides the ability for pain-free functional and occupational tasks with an adequate capacity to withstand the mechanical forces imparted by everyday life. Back pain and arthritis are the fourth- and sixth-most common complaints in primary care, underscoring suboptimal bone health management in developed countries.22
Optimizing bone health through proper nutrition is crucial in the perioperative management of geriatric orthopedic patients. The clinical diagnosis of malnutrition has well-studied associations with worse outcomes after orthopedic surgery, which include increased mortality, hospital length of stay, readmission rates, and health institution spending.23-25 Some studies show that up to 60% of geriatric patients may be malnourished.26
Regarding vitamin and mineral supplements, the general consensus before orthopedic surgery is that vitamins A, C, D, and E, and zinc are predictive in determining postoperative health.27 However, Curtis et al state that therapy should be targeted at correcting relative deficiencies; supraphysiologic concentrations of these vitamins do not appear to be helpful.27 This claim may merit serum studies to rule out deficiencies. Dietitians should be involved in the creation of a patient care plan in the spirit of multidisciplinary orthopedic surgery approaches, which have proven to result in superior patient outcomes.28 Additionally, directive counseling should be provided when necessary.
In patients with adequately managed nutrition, 7 to 10 days of diet optimization is typically sufficient, but patients with malnutrition may require sustained nutritional support for up to 6 weeks; a standardized time for adequate nutrition supplementation has not been identified.25-27 Postoperative management is similar in older patients who are malnourished and those receiving adequate nutrition after orthopedic surgery, which typically involves 3 weeks of a high-protein diet.26
Evaluating Mechanisms of Injury
Assessing the mechanism of injury (MOI) is essential to developing an appropriate and successful orthopedic treatment plan. MOI is typically categorized as low energy, which consists of ground-level falls and other minor trauma, or high energy, which can include motor vehicle crashes or falls from a height.29 Unlike younger patients who typically experience trauma from high-energy MOIs, geriatric patients often sustain fractures from low-energy MOIs. The importance of assessing MOI for the geriatric population is magnified as it provides vital clues that not only help determine the nature of the injury, but also highlight underlying frailty, comorbidities, and potential complications. Weakness or deconditioning related to older age is often not discovered before trauma, which is why assessing the MOI can provide valuable information regarding overall patient health.30
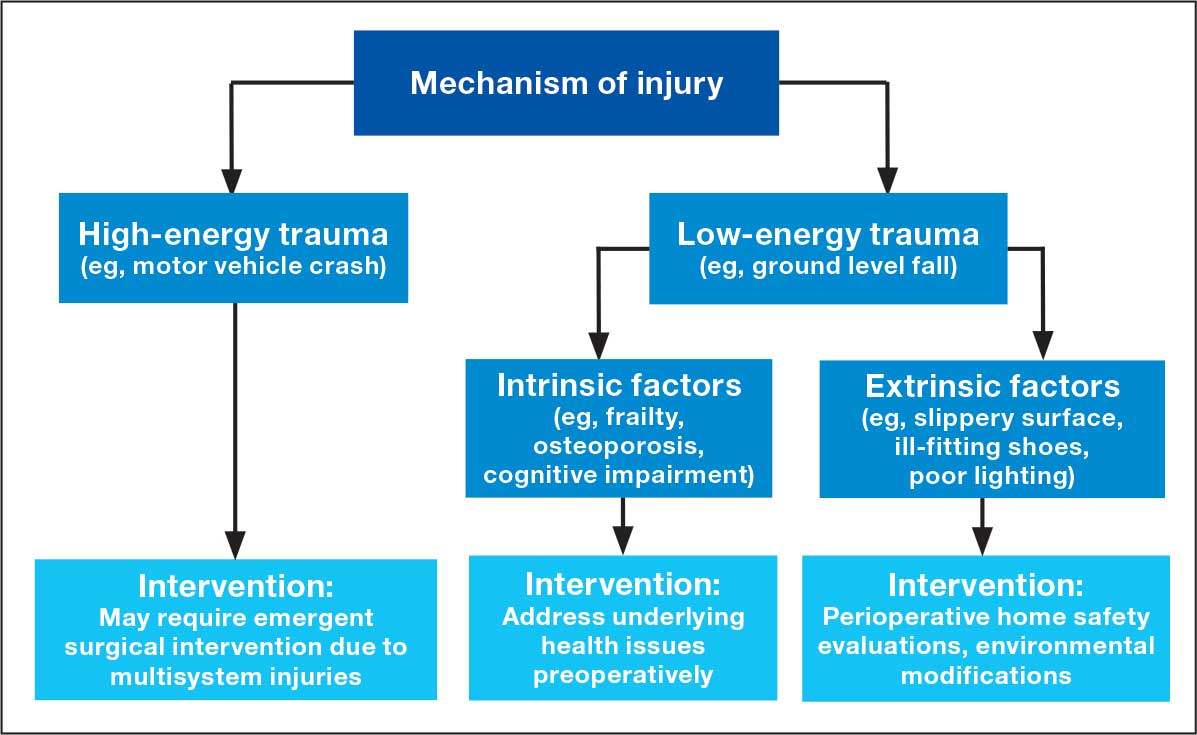
The MOI of trauma also is correlated with factors that influence postoperative recovery and overall prognosis (Figure). Falls comprise more than three-quarters of the MOI in geriatric patients with trauma, and > 90% of these falls are ground-level or other simple falls.30 Falls secondary to an intrinsic disorder, rather than an extrinsic environmental hazard, are more common in geriatric patients.31
These events may be associated with an underlying medical condition, such as osteopenia, osteoporosis, or neuromuscular disorders, such as Parkinson disease.32 They may also be attributed to normal age-related changes, such as decreased visual acuity, reduced reaction time, or mild cognitive impairment.30 An estimated 6% to 35% of geriatric patients who present to the emergency department have some degree of cognitive dysfunction.33 Accordingly, a thorough understanding of the events leading up to injury is vital for the management of older patients. Knowing the specific circumstances of a fall can provide insight into the patient’s gait, balance, and need for further investigations such as cognitive screening or evaluation of home safety. This information can guide decisions regarding preoperative optimization of medications and postoperative rehabilitation interventions.
Frailty and Risk of Fracture
Frailty is a clinical syndrome defined by overall decreased capacity for the body’s adaptive changes to various stressors.34 It is a common condition in geriatric populations due to cumulative degenerative changes and multisystem decline over a lifetime’s worth of disruptions to natural homeostasis.34 In orthopedics, frailty typically refers to musculoskeletal durability and resilience in response to mechanical forces (ie, falls, trauma, and high-acceleration movements). Globally, > 200 million people have osteoporotic frailty, leading to 9 million hip fractures annually.35 More than 30% of people aged ≥ 65 years fall ≥ 1 time per calendar year.36
Assessing frailty in geriatric patients undergoing orthopedic surgery is vital, as it predisposes patients to higher rates of morbidity, mortality, and institutionalization, particularly from falls and resultant fragility fractures.37-39 This is true for a wide range of orthopedic procedures, spanning elective to urgent surgeries and involving the axial and appendicular skeleton.40,41 Given the high rates of fractures, subsequent patient morbidity, and financial burden on the health care system, effective frailty screening is essential.
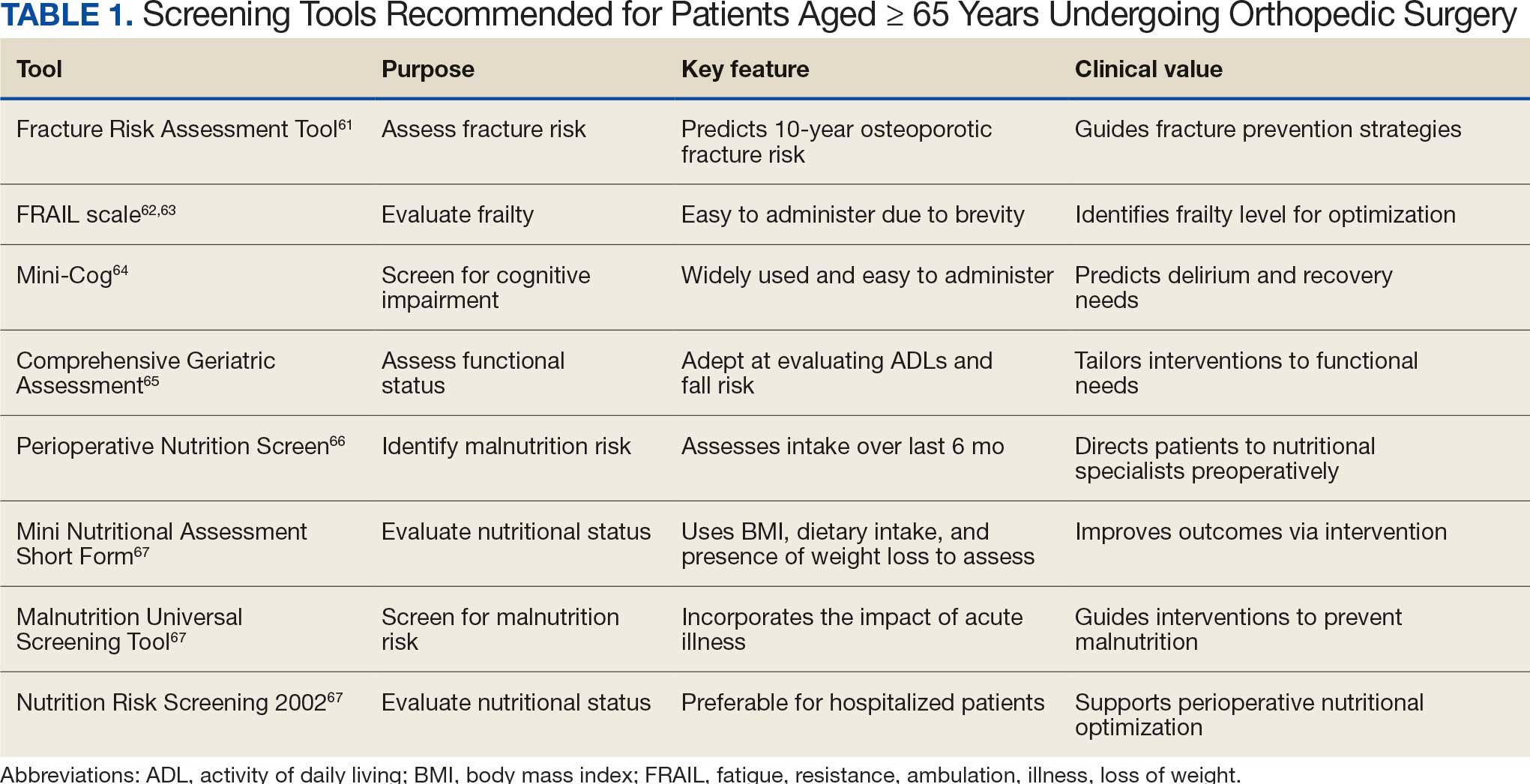
There are many strategies to assess frailty risk and subsequent fracture risk.42 Questionnaires or online medical calculators serve as easy-to-use tools for assessment of frailty or associated predictors of fragility fractures. Validated assessment tools are provided in Table 1.
Dual-emission X-ray absorptiometry is a well-established way to determine bone density and establish fracture risk. The Fried Frailty Phenotype score and Short Performance Physical Battery test are clinically applicable methods of assessing frailty in older outpatient populations. Although these examinations focus on different aspects of the patient, they have moderate agreeability, are sensitive, and can be readily performed in the clinical setting as demonstrated by a > 90% patient participation rate for both methods.42 Finally, several serum studies can be predictive of frailty, the most readily modifiable of which are vitamin D3, ferritin, albumin, and calcium.43 Although they are more invasive for the patient, serum studies can provide additional modifiable targets for perioperative optimization and contribute to risk stratification.
Risk stratification should take place around 6 weeks before surgery, which should provide adequate time for rectification of preoperative barriers to elective surgical intervention—namely nutritional status. In cases of urgent or emergent procedures (ie, femoral neck fracture with concern for avascular necrosis of the femoral head), this may not be possible but should be conducted nonetheless for patient-specific postoperative rehabilitation and risk reduction.
Postoperative Complication Risks
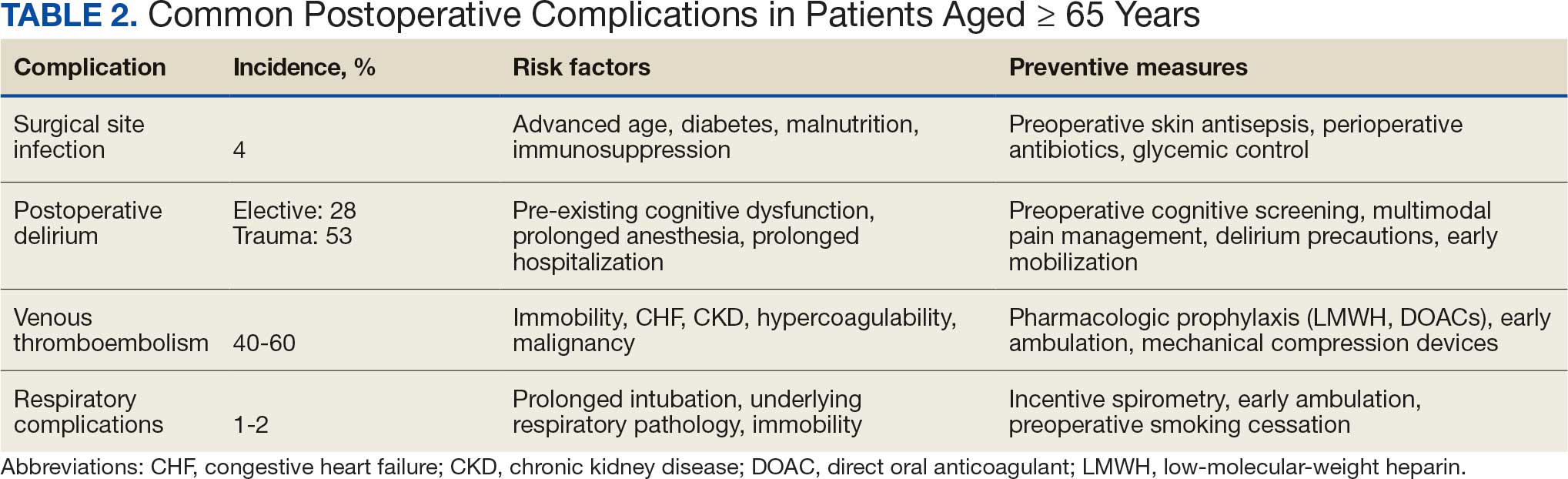
Postoperative complications affect nearly 15% of geriatric orthopedic patients, highlighting the need for comprehensive preoperative evaluations to assess risk factors.44 Age-related physiological changes, frailty, and comorbidities complicate recovery and management (Table 2).
Wound healing is impaired in older individuals due to suboptimal circulation and decreased oxygenation that is secondary to age-related changes, as well as the increased likelihood of comorbid conditions (eg, diabetes).7 Surgical site infections can be particularly malicious in geriatric patients, with a 4% incidence.45,46 Hospitalization can be prolonged by a mean 2 weeks, which increases the risk of hospital-associated delirium and iatrogenic complications.46 Both the mortality rate and costs associated with hospitalization are higher for older patients who develop surgical site infections compared with patients aged < 65 years, underscoring the importance of vigilant monitoring, early detection, and effective preoperative screening to identify and manage modifiable risk factors.47
Postoperative delirium is another common complication of orthopedic surgery in the geriatric population, increasing morbidity and mortality. The incidence is reported to be as high as 53.3% in the trauma setting and 28.3% in the elective setting, indicating a need to assess patient risk preoperatively.48,49 Several factors contribute to the high incidence of delirium, including advanced age, longer surgical durations, intraoperative hypotension and hypercapnia, pre-existing cognitive dysfunction, and postoperative sleep disorders.50
VTE is another common cause of complications following orthopedic surgery. The development of DVT can lead to subsequent pulmonary embolism, which can be fatal. Orthopedic surgery patients are already at higher risk of DVT and VTE than other surgical patients, with an incidence as high as 40% to 60%, though it is frequently asymptomatic.51,52 Geriatric patients may be more likely to have concurrent comorbidities that increase hypercoagulability.53 Congestive heart failure, chronic kidney disease, and cardiovascular disease are all more common in older individuals and can increase the risk of VTE by 2-fold.53 While anticoagulation is the standard of care to prevent VTE after orthopedic surgery, geriatric patients require more careful monitoring due to the higher incidence of bleeding complications. Additionally, early postoperative mobilization is critical to reduce the risk of DVT without significantly increasing pain or causing other adverse events.54
Respiratory complications are common after orthopedic surgery, particularly atelectasis and bronchospasm, which can result from intraoperative mechanical ventilation.55 While these conditions are typically self-limiting, more severe respiratory issues such as pneumonia are a significant concern because they may lead to the need for mechanical ventilation and admission to the intensive care unit (ICU). The more severe complications have an incidence of about 1% to 2% in orthopedic surgery patients.56 Preventive strategies, such as respiratory physiotherapy and guided breathing exercises, are crucial to minimize perioperative pulmonary complications and promote optimal recovery. Addressing these challenges through early intervention is essential to improve outcomes.
Multidisciplinary Perioperative Care
Multidisciplinary care in orthopedic surgery involves collaborative management of patient care by general practitioners, surgeons, anesthesiologists, dietitians, physical and occupational therapists, inpatient health care practitioners (HCPs), and social services. The goal of this form of care is to provide a longitudinal sequence of health-optimization tactics that prepare a patient for surgery and give them the best chance of postoperative recovery.
Given that many aspects of geriatric health play a role in orthopedic postoperative outcomes, there are many preoperative factors to consider. As previously discussed, preoperative evaluation of geriatric patients should include nutritional and fragility screening to determine surgical candidacy and target modifiable risk factors for risk reduction. This screening can be conducted by primary care practitioners and orthopedic surgeons in an outpatient setting. A multidisciplinary approach benefits patients by decreasing time to surgery.35
Several large studies have demonstrated the positive influence of a multidisciplinary approach on patient-oriented outcomes in orthopedic patients. Incorporation of this style of care in contrast to surgeon-led perioperative optimization leads to fewer floor and ICU admissions, shorter lengths of stay, and decreased mortality rates.35,57 These findings are broadly applicable to a wide range of orthopedic surgeries and even surgeries outside of the musculoskeletal system.58,59 In addition, this strategy has demonstrated reduced in-hospital health care costs due to shorter lengths of stay and fewer ICU admissions. Physical and occupational therapy also have irreplaceable roles in outcomes after orthopedic surgeries. They have independently been shown to decrease pain, increase range of motion, and increase functionality in daily life.60 These aspects of recovery are essential for geriatric well-being.
Screening Tools
The World Health Organization FRAX fracture risk assessment tool (www.fraxplus.org/calculation-tool) was developed to identify patients at high risk of fracture and subsequent complications and to guide clinical decision-making regarding pharmacologic interventions.61 FRAX calculates the 10-year probability of fracture based on demographic factors, such as age and body mass index, clinical measures (eg, femoral neck bone mineral density), and risk factors (eg, prior fragility fractures, substance use history, and prolonged glucocorticoid use).61 The online tool is easy to use, making it a valuable resource for assessing fracture risk and determining appropriate treatment strategies.
The fatigue, resistance, ambulation, illnesses, loss of weight (FRAIL) scale assesses frailty in older adults. The scale classifies patients into 3 categories: robust, prefrail, and frail. The frail category is associated with an increased frequency of hip fracture and an elevated ASA class.62 Additionally, the FRAIL scale has demonstrated value in predicting hospital length of stay and the risk of postoperative complications.62 It also has shown utility in quantifying frailty status, which is traditionally challenging to assess systematically.63
The Mini-Cog is commonly used in geriatric populations to screen for cognitive impairment. Preoperative Mini-Cog screening has been shown to predict the development of postoperative complications.64 Geriatric patients who screened positive for cognitive impairment prior to orthopedic surgery were more likely to develop postoperative delirium, require alternative discharge disposition, and have a longer hospital length of stay.64 Mini-Cog serves as an important preoperative tool for identifying patients who may benefit from closer postoperative monitoring and tailored care.
The Comprehensive Geriatric Assessment (CGA) is a multidimensional evaluation that has been validated for use in geriatric patients undergoing orthopedic surgery.65 The CGA assesses functional status and the ability to perform activities of daily living (ADLs), such as eating, dressing, and ambulating. Poor ADLs are associated with increased risk of falls and cardiopulmonary complications. The CGA allows HCPs to identify patients at higher risk of complications and tailor interventions that optimize functional recovery during the perioperative period.
Nutritional screening is another component of preoperative evaluation in older adults undergoing orthopedic surgery. The Perioperative Nutrition Screen is a preoperative phone assessment of unintentional weight loss in the past 6 months.66 Patients who screen positive are asked to come in for a preoperative visit with a registered dietitian who can further evaluate the nutritional status of the patient.
The Mini Nutritional Assessment Short Form (MNA-SF), Malnutrition Universal Screening Tool, and Nutrition Risk Screening 2002 have all been validated for use in older patients undergoing orthopedic surgery. Among these, the MNA-SF has demonstrated superior utility in predicting hospital readmission and mortality.67 Given the established links between malnutrition and poor surgical outcomes, routine nutritional screening is important for identifying geriatric patients who may require preoperative nutritional interventions.
CONCLUSIONS AND RECOMMENDATIONS
Perioperative management of geriatric patients undergoing orthopedic surgery requires an assessment and strategy focused on risk stratification, patient optimization, and mitigation of potential complications and mortality. Due to the complexity and comprehensive nature of an optimal perioperative plan, creating the plan early is essential to ensure adequate time for patient optimization and care coordination.
Nutrition plays a critical role in the success of surgical procedures, and orthopedics is no exception. Extra care should be taken to preoperatively optimize patient bone health before surgical intervention to enhance recovery and reduce the risk of complications. After an appropriate patient history and clinical picture are gathered, screening tools should be used on a case-by-case basis to further characterize comorbid conditions that may contribute to suboptimal outcomes. Additionally, given the proven association between frailty and fracture risk, frailty serves as a readily quantifiable predictor of patient-oriented outcomes. This should be assessed preoperatively with appropriate risk-stratification tools to determine appropriate postoperative measures to prevent morbidity and mortality.
Orthopedic surgery is increasingly common in geriatric patients, who face higher perioperative risks due to age-related physiological changes, multimorbidity, and frailty. Optimizing preoperative assessment and adopting a multidisciplinary approach—integrating surgeons, anesthesiologists, physical therapists, and dietitians—can improve outcomes, reduce complications, and enhance recovery. The successful use of the tools and strategies outlined in this article by primary care should facilitate access to and recovery from orthopedic surgery in the geriatric population.
Dobson GP. Trauma of major surgery: a global problem that is not going away. Int J Surg. 2020;81:47-54. doi:10.1016/j.ijsu.2020.07.017
United States (US) orthopedic procedures count by segments and forecast to 2030. GlobalData. February 17, 2023. Accessed April 29, 2026. https://www.globaldata.com/store/report/usa-orthopedic-procedures-analysis/
Jarman MP, Weaver MJ, Haider AH, Salim A, Harris MB. The national burden of orthopedic injury: cross-sectional estimates for trauma system planning and optimization. J Surg Res. 2020;249:197-204. doi:10.1016/j.jss.2019.12.023
Hegde V, Stambough JB, Levine BR, et al. Highlights of the 2022 American Joint Replacement Registry Annual Report. Arthroplast Today. 2023;21:101137. doi:10.1016/j.artd.2023.101137
Nakamura K, Ogata T. Locomotive syndrome: definition and management. Clin Rev Bone Miner Metab. 2016;14:56-67. doi:10.1007/s12018-016-9208-2
Glyn-Jones S, Palmer AJR, Agricola R, et al. Osteoarthritis. Lancet. 2015;386:376-387. doi:10.1016/S0140-6736(14)60802-3
Hughes S, Leary A, Zweizig S, Cain J. Surgery in elderly people: preoperative, operative and postoperative care to assist healing. Best Pract Res Clin Obstet Gynaecol. 2013;27:753-765. doi:10.1016/j.bpobgyn.2013.02.006
Regulski MJ. Cellular senescence: what, why, and how. Wounds. 2017;29:168-174.
Kremer M, Burkemper N. Aging skin and wound healing. Clin Geriatr Med. 2024;40:1-10. doi:10.1016/j.cger.2023.06.001
Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol. 1986;15:571-585. doi:10.1016/S0190-9622(86)70208-9
Van Neste D, Tobin DJ. Hair cycle and hair pigmentation: dynamic interactions and changes associated with aging. Micron. 2004;35:193-200. doi:10.1016/j.micron.2003.11.006
Salzer MC, Lafzi A, Berenguer-Llergo A, et al. Identity noise and adipogenic traits characterize dermal fibroblast aging. Cell. 2018;175:1575-1590.e22. doi:10.1016/j.cell.2018.10.012
Jin K. A microcirculatory theory of aging. Aging Dis. 2019;10:676-683. doi:10.14336/AD.2019.0315
Marengoni A, Angleman S, Melis R, et al. Aging with multimorbidity: a systematic review of the literature. Ageing Res Rev. 2011;10:430-439. doi:10.1016/j.arr.2011.03.003
Waljee AK, Rogers MAM, Lin P, et al. Short term use of oral corticosteroids and related harms among adults in the United States: population based cohort study. BMJ. 2017;357:j1415. doi:10.1136/bmj.j1415
Given B, Given CW. Older adults and cancer treatment. Cancer. 2008;113:3505-3511. doi:10.1002/cncr.23939
Ferrucci L, Cooper R, Shardell M, Simonsick EM, Schrack JA, Kuh D. Age-related change in mobility: perspectives from life course epidemiology and geroscience. J Gerontol A Biol Sci Med Sci. 2016;71:1184-1194. doi:10.1093/gerona/glw043
Mayhew D, Mendonca V, Murthy BVS. A review of ASA physical status - historical perspectives and modern developments. Anaesthesia. 2019;74:373-379. doi:10.1111/anae.14569
Eagle KA, Berger PB, Calkins H, et al. ACC/AHA guideline update for perioperative cardiovascular evaluation for noncardiac surgery—executive summary a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1996 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation. 2002;105:1257-1267. doi:10.1161/circ.105.10.1257
Bapoje SR, Whitaker JF, Schulz T, Chu ES, Albert RK. Preoperative evaluation of the patient with pulmonary disease. Chest. 2007;132:1637-1645. doi:10.1378/chest.07-0347
Choe H, Indelli PF, Ricciardi B, et al. What are the absolute contraindications for elective total knee or hip arthroplasty? J Arthroplasty. 2025;40(2 suppl 1):S45-S47. doi:10.1016/j.arth.2024.10.041
Finley CR, Chan DS, Garrison S, et al. What are the most common conditions in primary care? Systematic review. Can Fam Physician. 2018;64:832-840.
Vaid S, Bell T, Grim R, Ahuja V. Predicting risk of death in general surgery patients on the basis of preoperative variables using American College of Surgeons National Surgical Quality Improvement Program data. Perm J. 2012;16:10-17. doi:10.7812/TPP/12-019
Correia MTD, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22:235-239. doi:10.1016/S0261-5614(02)00215-7
Friedman J, Lussiez A, Sullivan J, Wang S, Englesbe M. Implications of sarcopenia in major surgery. Nutr Clin Pract. 2015;30:175-179. doi:10.1177/0884533615569888
Hirsch KR, Wolfe RR, Ferrando AA. Pre- and post-surgical nutrition for preservation of muscle mass, strength, and functionality following orthopedic surgery. Nutrients. 2021;13:1675. doi:10.3390/nu13051675
Curtis W, Choi T, Ahmad A, Shultz C. Perioperative nutritional considerations in orthopaedic surgery: a review of the literature. West J Orthop. 2023;12:1. https://digitalrepository.unm.edu/wjo/vol12/iss1/1
Wischmeyer PE, Carli F, Evans DC, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on nutrition screening and therapy within a surgical enhanced recovery pathway. Anesth Analg. 2018;126:1883-1895. doi:10.1213/ANE.0000000000002743
Mun F, Ringenbach K, Baer B, et al. Factors influencing geriatric orthopaedic trauma mortality. Injury. 2022;53:919-924. doi:10.1016/j.injury.2022.01.005
Bonne S, Schuerer DJE. Trauma in the older adult: epidemiology and evolving geriatric trauma principles. Clin Geriatr Med. 2013;29:137-150. doi:10.1016/j.cger.2012.10.008
Montero-Odasso MM. Falls as a geriatric syndrome: mechanisms and risk identification. In: Duque G, Kiel DP, eds. Osteoporosis in Older Persons: Advances in Pathophysiology and Therapeutic Approaches. 2nd ed. Springer International Publishing; 2016:171-186. doi:10.1007/978-3-319-25976-5_10
Lach HW, Reed AT, Arfken CL, et al. Falls in the elderly: reliability of a classification system. J Am Geriatr Soc. 1991;39:197-202. doi:10.1111/j.1532-5415.1991.tb01626.x
Carpenter CR, DesPain B, Keeling TN, Shah M, Rothenberger M. The six-item screener and AD8 for the detection of cognitive impairment in geriatric emergency department patients. Ann Emerg Med. 2011;57:653-661. doi:10.1016/j.annemergmed.2010.06.560
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381:752-762. doi:10.1016/S0140-6736(12)62167-9
Patel JN, Klein DS, Sreekumar S, Liporace FA, Yoon RS. Outcomes in multidisciplinary team-based approach in geriatric hip fracture care: a systematic review. J Am Acad Orthop Surg. 2020;28:128-133. doi:10.5435/JAAOS-D-18-00425
Amador LF, Loera JA. Preventing postoperative falls in the older adult. J Am Coll Surg. 2007;204:447-453. doi:10.1016/j.jamcollsurg.2006.12.010
Tembo MC, Holloway-Kew KL, Mohebbi M, et al. The association between a fracture risk tool and frailty: Geelong Osteoporosis Study. BMC Geriatr. 2020;20:196. doi:10.1186/s12877-020-01595-8
Demiris¸ B, Basat S, Kurt F, Aksakal B, Basat O. Evaluation of the relationship between frailty and fracture risk using Fracture Risk Assessment Tool in patients 65 years and over. South Clin Istanb Eurasia. 2023;34:42-48. doi:10.14744/scie.2022.66564
Partridge JSL, Harari D, Dhesi JK. Frailty in the older surgical patient: a review. Age Ageing. 2012;41:142-147. doi:10.1093/ageing/afr182
Mamtora PH, Fortier MA, Barnett SR, Schmid LN, Kain ZN. Peri-operative management of frailty in the orthopedic patient. J Orthop. 2020;22:304-307. doi:10.1016/j.jor.2020.05.024
Leven DM, Lee NJ, Kim JS, et al. Frailty is predictive of adverse postoperative events in patients undergoing lumbar fusion. Global Spine J. 2017;7:529-535. doi:10.1177/2192568217700099
Pritchard JM, Kennedy CC, Karampatos S, et al. Measuring frailty in clinical practice: a comparison of physical frailty assessment methods in a geriatric out-patient clinic. BMC Geriatr. 2017;17:264. doi:10.1186/s12877-017-0623-0
Kumar A, Dhar M, Agarwal M, Mukherjee A, Saxena V. Predictors of frailty in the elderly population: a cross-sectional study at a tertiary care center. Cureus. 2022;14:e30557. doi:10.7759/cureus.30557
Scarano KA, Philp FH, Westrick ER, Altman GT, Altman DT. Evaluating postoperative complications and outcomes of orthopedic fracture repair in nonagenarian patients. Geriatr Orthop Surg Rehabil. 2018;9:2151459318758106. doi:10.1177/2151459318758106
Liang Z, Rong K, Gu W, et al. Surgical site infection following elective orthopaedic surgeries in geriatric patients: incidence and associated risk factors. Int Wound J. 2019;16:773-780. doi:10.1111/iwj.13096
Ren M, Liang W, Wu Z, Zhao H, Wang J. Risk factors of surgical site infection in geriatric orthopedic surgery: a retrospective multicenter cohort study. Geriatr Gerontol Int. 2019;19:213-217. doi:10.1111/ggi.13590
Kaye KS, Schmader KE, Sawyer R. Surgical site infection in the elderly population. Clin Infect Dis. 2004;39:1835-1841. doi:10.1086/425744
Bruce AJ, Ritchie CW, Blizard R, Lai R, Raven P. The incidence of delirium associated with orthopedic surgery: a meta-analytic review. Int Psychogeriatr. 2007;19:197-214. doi:10.1017/S104161020600425X
Williams-Russo P, Urquhart BL, Sharrock NE, Charlson ME. Post-operative delirium: predictors and prognosis in elderly orthopedic patients. J Am Geriatr Soc. 1992;40:759-767. doi:10.1111/j.1532-5415.1992.tb01846.x
Wang J, Li Z, Yu Y, Li B, Shao G, Wang Q. Risk factors contributing to postoperative delirium in geriatric patients postorthopedic surgery. Asia Pac Psychiatry. 2015;7:375-382. doi:10.1111/appy.12193
Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126:338S-400S. doi:10.1378/chest.126.3_suppl.338S
Kahn SR, Shivakumar S. What’s new in VTE risk and prevention in orthopedic surgery. Res Pract Thromb Haemost. 2020;4:366-376. doi:10.1002/rth2.12323
Uzel K, Azboy I·, Parvizi J. Venous thromboembolism in orthopedic surgery: global guidelines. Acta Orthop Traumatol Turc. 2023;57:192-203. doi:10.5152/j.aott.2023.23074
Peck M, Holthaus A, Kingsbury K, Salsberry MG, Duggirala V. Mobility in acute care for geriatric patients with orthopedic conditions: a review of recent literature. Curr Geri Rep. 2020;9:300-310. doi:10.1007/s13670-020-00347-1
Leme LEG, Sitta MC, Toledo M, Henriques SS. Orthopedic surgery among the elderly: clinical characteristics. Rev Bras Ortop. 2015;46:238-246. doi:10.1016/S2255-4971(15)30189-0
Malcolm TL, Knezevic NN, Zouki CC, Tharian AR. Pulmonary complications after hip and knee arthroplasty in the United States, 2004-2014. Anesth Analg. 2020;130:917-924. doi:10.1213/ANE.0000000000004265
Kamal T, Conway RM, Littlejohn I, Ricketts D. The role of a multidisciplinary pre-assessment clinic in reducing mortality after complex orthopaedic surgery. Ann R Coll Surg Engl. 2011;93:149-151. doi:10.1308/003588411X561026
Davis MJ, Luu BC, Raj S, Abu-Ghname A, Buchanan EP. Multidisciplinary care in surgery: Are team-based interventions cost-effective? Surgeon. 2021;19:49-60. doi:10.1016/j.surge.2020.02.005
Frassanito L, Vergari A, Nestorini R, et al. Enhanced recovery after surgery (ERAS) in hip and knee replacement surgery: description of a multidisciplinary program to improve management of the patients undergoing major orthopedic surgery. Musculoskelet Surg. 2020;104:87-92. doi:10.1007/s12306-019-00603-4
Reddy RS, Alahmari KA, Alshahrani MS, et al. Exploring the impact of physiotherapy on health outcomes in older adults with chronic diseases: a cross-sectional analysis. Front Public Health. 2024;12:1415882. doi:10.3389/fpubh.2024.1415882
Watts NB. The Fracture Risk Assessment Tool (FRAX®): applications in clinical practice. J Womens Health (Larchmt). 2011;20:525-531. doi:10.1089/jwh.2010.2294
Gleason LJ, Benton EA, Alvarez-Nebreda ML, Weaver MJ, Harris MB, Javedan H. FRAIL questionnaire screening tool and short-term outcomes in geriatric fracture patients. J Am Med Dir Assoc. 2017;18:1082-1086. doi:10.1016/j.jamda.2017.07.005
Kojima G. Frailty defined by FRAIL scale as a predictor of mortality: a systematic review and meta-analysis. J Am Med Dir Assoc. 2018;19:480-483. doi:10.1016/j.jamda.2018.04.006
Culley DJ, Flaherty D, Fahey MC, et al. Poor performance on a preoperative cognitive screening test predicts postoperative complications in older orthopedic surgical patients. Anesthesiology. 2017;127:765-774. doi:10.1097/ALN.0000000000001859
Kong C, Zhang Y, Wang C, et al. Comprehensive geriatric assessment for older orthopedic patients and analysis of risk factors for postoperative complications. BMC Geriatr. 2022;22:644. doi:10.1186/s12877-022-03328-5
Williams DGA, Wischmeyer PE. Perioperative nutrition care of orthopedic surgery patient. Tech Orthop. 2020;35:15-18. doi:10.1097/BTO.0000000000000412
Koren-Hakim T, Weiss A, Hershkovitz A, et al. Comparing the adequacy of the MNA-SF, NRS-2002 and MUST nutritional tools in assessing malnutrition in hip fracture operated elderly patients. Clin Nutr. 2016;35:1053-1058. doi:10.1016/j.clnu.2015.07.014
More than 40 million surgeries are performed annually in the United States, of which > 18 million are orthopedic, including > 1 million emergency orthopedic surgeries and > 2 million joint replacements.1-4 Notably, > 50% of patients undergoing orthopedic surgery are aged ≥ 65 years, a demographic shift driven by longer life expectancies and an increasing number of older adults remaining physically active for extended periods.5 Osteoarthritis, the most common joint disease, affects 10% of men and 18% of women aged > 60 years, often necessitating an orthopedic joint replacement.6 Perioperative morbidity and mortality are 2.9- to 6.7-times higher in older adults compared with younger adults.7 These risks include infection, venous thromboembolism (VTE), pressure ulcers, reduced mobility, and increased mortality. Due to the high incidence of these complications in older surgical patients, special perioperative protocols and considerations are needed when preparing an older patient for surgery. This review aims to establish concrete considerations and guidelines for perioperative management.
METHODOLOGY
A literature review of PubMed, Google Scholar, and IEEE Xplore identified research on perioperative challenges in geriatric orthopedic surgery. Keywords included geriatrics and orthopedic surgery, perioperative care in geriatric populations, and orthopedic perioperative care. Inclusion criteria were strictly defined to ensure relevance to the geriatric population, with studies focusing on patients aged ≥ 65 years. Exclusion criteria were applied to remove studies that did not involve geriatric populations or orthopedic surgeries or that lacked a clear perioperative focus. Studies were analyzed for design, interventions, and outcomes. Special attention was given to identifying common challenges and trends related to perioperative considerations. We developed a narrative report providing a comprehensive overview of the current understanding of perioperative care for geriatric orthopedic patients to offer practical recommendations for clinicians to use in their practice.
RESULTS
Consistent with the narrative review methodology described, the literature search yielded a broad range of publications addressing perioperative considerations in geriatric orthopedic patients. Articles were screened for relevance to patients aged ≥ 65 years undergoing orthopedic surgery and for applicability to perioperative optimization and postoperative outcomes. Given the heterogeneity in study design, population characteristics, and outcome reporting, findings are presented descriptively rather than being quantitatively pooled. Studies not focused on geriatric populations, orthopedic procedures, or perioperative management were excluded. Key themes included multimorbidity and comorbidity optimization, age-related physiologic changes, frailty assessment and fracture risk stratification, nutritional and bone health management, mechanism of injury considerations, prevention of postoperative complications, and the role of multidisciplinary perioperative care.
Unique Physiological Challenges
The aging process induces a range of physiological changes that can increase morbidity and mortality following surgery. One of the most essential elements to surgical recovery is wound healing, as impairments in this process can lead to adverse events, including infection, cosmetic deformity, and wound dehiscence. The general paradigm of aging involves cell senescence resulting in slower or disorganized functional capacity of these cells.8 While wound healing in older individuals was once thought to be defective, recent research has demonstrated that this process is not absent, but delayed.9
Wound healing is a tightly regulated and evolutionarily conserved process that proceeds through 3 main phases: inflammation, proliferation, and remodeling. Re-epithelialization begins with the migration of epithelial cells from hair follicles, sweat glands, or wound margins (depending on wound depth) and is influenced by oxygen levels, moisture, and growth factors.9 Several characteristics of aged skin contribute to the delayed healing process. Aged skin has fewer hair follicles and eccrine sweat glands, as well as decreased follicle thickness.10 This results in fewer proliferating cells for wound healing and lower amounts of sebum production for skin moisture.11 Furthermore, aged fibroblasts are fewer in number and less effective in synthesizing extracellular matrices, resulting in slower and less tensile wound healing.12 Additionally, microvascular changes associated with aging result in disorganized vasculature, which impairs oxygen delivery to the wound bed and diminishes the influx of proinflammatory cells necessary for effective healing.13 These senescent traits of aged skin contribute to the delayed wound healing process found in geriatric patients.
Compounding these age-related factors is the prevalence of multimorbidity, or coexisting chronic diagnoses, in 55% to 98% of older patients.14 Common comorbidities include peripheral arterial disease, chronic venous insufficiency, type 1 and type 2 diabetes, neoplasms, atherosclerotic disease, and hypertension. Older patients are more likely to be prescribed corticosteroids and chemotherapeutic agents that impair the function of inflammatory cells necessary for wound healing.15,16 Additionally, decreased mobility is more common in geriatric patients, which can increase the risk of wound formation, particularly pressure ulcers.17
Perioperative Considerations
All surgical patients undergo a formal or informal preoperative evaluation to assess their fitness for surgery, with the goal of minimizing both anesthesia-related risks and postoperative complications. A widely used tool in this assessment is the American Society of Anesthesiologists (ASA) physical status classification, which stratifies patients into 6 categories based on their medical history and overall health status.18 Classes range from healthy patients (Class I) to organ donors who are brain-dead (Class VI).
Cardiac optimization is an essential component of preoperative evaluation for older adults due to their higher risk of underlying cardiovascular disease.19 This process involves an in-depth review of the patient’s cardiac history, including the timing and nature of any prior interventions and the recurrence rate. Functional capacity is assessed through metabolic equivalents, where a threshold of > 4 metabolic equivalents (the ability to walk up a flight of stairs) is considered adequate for surgery. Risk is assessed based on the specific surgical procedure, and nonemergent orthopedic procedures are considered intermediate risk. If a patient is deemed high risk at any stage of this evaluation, further cardiac testing is indicated.
Pulmonary optimization is typically necessary for geriatric patients, who are more likely to have conditions such as chronic obstructive pulmonary disease or interstitial lung disease.14,20 In patients without severe systemic lung disease, pulmonary optimization involves assessing the functional expiratory volume and diffusing capacity for carbon monoxide. In addition, aggressive modification of risk factors, such as smoking cessation, is strongly recommended.
Additional perioperative conditions are disease-specific and involve evaluation of comorbid illnesses and recognition of absolute contraindications to noncardiac surgery. For instance, an ejection fraction of < 35%, a history of myocardial infarction within 6 months, or active diabetic ketoacidosis are all absolute contraindications to elective surgery. For orthopedic procedures, additional contraindications include symptomatic bacteremia, active joint or local tissue infection, severe malnutrition, uncontrolled metabolic syndrome or chronic disease, untreated immunodeficiency, and active deep venous thrombosis (DVT) or pulmonary embolism.21
Bone Health and Nutrition
In the context of orthopedic surgery, the hallmark of clinically defined optimal bone health is a musculoskeletal system that provides the ability for pain-free functional and occupational tasks with an adequate capacity to withstand the mechanical forces imparted by everyday life. Back pain and arthritis are the fourth- and sixth-most common complaints in primary care, underscoring suboptimal bone health management in developed countries.22
Optimizing bone health through proper nutrition is crucial in the perioperative management of geriatric orthopedic patients. The clinical diagnosis of malnutrition has well-studied associations with worse outcomes after orthopedic surgery, which include increased mortality, hospital length of stay, readmission rates, and health institution spending.23-25 Some studies show that up to 60% of geriatric patients may be malnourished.26
Regarding vitamin and mineral supplements, the general consensus before orthopedic surgery is that vitamins A, C, D, and E, and zinc are predictive in determining postoperative health.27 However, Curtis et al state that therapy should be targeted at correcting relative deficiencies; supraphysiologic concentrations of these vitamins do not appear to be helpful.27 This claim may merit serum studies to rule out deficiencies. Dietitians should be involved in the creation of a patient care plan in the spirit of multidisciplinary orthopedic surgery approaches, which have proven to result in superior patient outcomes.28 Additionally, directive counseling should be provided when necessary.
In patients with adequately managed nutrition, 7 to 10 days of diet optimization is typically sufficient, but patients with malnutrition may require sustained nutritional support for up to 6 weeks; a standardized time for adequate nutrition supplementation has not been identified.25-27 Postoperative management is similar in older patients who are malnourished and those receiving adequate nutrition after orthopedic surgery, which typically involves 3 weeks of a high-protein diet.26
Evaluating Mechanisms of Injury
Assessing the mechanism of injury (MOI) is essential to developing an appropriate and successful orthopedic treatment plan. MOI is typically categorized as low energy, which consists of ground-level falls and other minor trauma, or high energy, which can include motor vehicle crashes or falls from a height.29 Unlike younger patients who typically experience trauma from high-energy MOIs, geriatric patients often sustain fractures from low-energy MOIs. The importance of assessing MOI for the geriatric population is magnified as it provides vital clues that not only help determine the nature of the injury, but also highlight underlying frailty, comorbidities, and potential complications. Weakness or deconditioning related to older age is often not discovered before trauma, which is why assessing the MOI can provide valuable information regarding overall patient health.30

The MOI of trauma also is correlated with factors that influence postoperative recovery and overall prognosis (Figure). Falls comprise more than three-quarters of the MOI in geriatric patients with trauma, and > 90% of these falls are ground-level or other simple falls.30 Falls secondary to an intrinsic disorder, rather than an extrinsic environmental hazard, are more common in geriatric patients.31
These events may be associated with an underlying medical condition, such as osteopenia, osteoporosis, or neuromuscular disorders, such as Parkinson disease.32 They may also be attributed to normal age-related changes, such as decreased visual acuity, reduced reaction time, or mild cognitive impairment.30 An estimated 6% to 35% of geriatric patients who present to the emergency department have some degree of cognitive dysfunction.33 Accordingly, a thorough understanding of the events leading up to injury is vital for the management of older patients. Knowing the specific circumstances of a fall can provide insight into the patient’s gait, balance, and need for further investigations such as cognitive screening or evaluation of home safety. This information can guide decisions regarding preoperative optimization of medications and postoperative rehabilitation interventions.
Frailty and Risk of Fracture
Frailty is a clinical syndrome defined by overall decreased capacity for the body’s adaptive changes to various stressors.34 It is a common condition in geriatric populations due to cumulative degenerative changes and multisystem decline over a lifetime’s worth of disruptions to natural homeostasis.34 In orthopedics, frailty typically refers to musculoskeletal durability and resilience in response to mechanical forces (ie, falls, trauma, and high-acceleration movements). Globally, > 200 million people have osteoporotic frailty, leading to 9 million hip fractures annually.35 More than 30% of people aged ≥ 65 years fall ≥ 1 time per calendar year.36
Assessing frailty in geriatric patients undergoing orthopedic surgery is vital, as it predisposes patients to higher rates of morbidity, mortality, and institutionalization, particularly from falls and resultant fragility fractures.37-39 This is true for a wide range of orthopedic procedures, spanning elective to urgent surgeries and involving the axial and appendicular skeleton.40,41 Given the high rates of fractures, subsequent patient morbidity, and financial burden on the health care system, effective frailty screening is essential.
There are many strategies to assess frailty risk and subsequent fracture risk.42 Questionnaires or online medical calculators serve as easy-to-use tools for assessment of frailty or associated predictors of fragility fractures. Validated assessment tools are provided in Table 1.
Dual-emission X-ray absorptiometry is a well-established way to determine bone density and establish fracture risk. The Fried Frailty Phenotype score and Short Performance Physical Battery test are clinically applicable methods of assessing frailty in older outpatient populations. Although these examinations focus on different aspects of the patient, they have moderate agreeability, are sensitive, and can be readily performed in the clinical setting as demonstrated by a > 90% patient participation rate for both methods.42 Finally, several serum studies can be predictive of frailty, the most readily modifiable of which are vitamin D3, ferritin, albumin, and calcium.43 Although they are more invasive for the patient, serum studies can provide additional modifiable targets for perioperative optimization and contribute to risk stratification.
Risk stratification should take place around 6 weeks before surgery, which should provide adequate time for rectification of preoperative barriers to elective surgical intervention—namely nutritional status. In cases of urgent or emergent procedures (ie, femoral neck fracture with concern for avascular necrosis of the femoral head), this may not be possible but should be conducted nonetheless for patient-specific postoperative rehabilitation and risk reduction.


Postoperative Complication Risks
Postoperative complications affect nearly 15% of geriatric orthopedic patients, highlighting the need for comprehensive preoperative evaluations to assess risk factors.44 Age-related physiological changes, frailty, and comorbidities complicate recovery and management (Table 2).
Wound healing is impaired in older individuals due to suboptimal circulation and decreased oxygenation that is secondary to age-related changes, as well as the increased likelihood of comorbid conditions (eg, diabetes).7 Surgical site infections can be particularly malicious in geriatric patients, with a 4% incidence.45,46 Hospitalization can be prolonged by a mean 2 weeks, which increases the risk of hospital-associated delirium and iatrogenic complications.46 Both the mortality rate and costs associated with hospitalization are higher for older patients who develop surgical site infections compared with patients aged < 65 years, underscoring the importance of vigilant monitoring, early detection, and effective preoperative screening to identify and manage modifiable risk factors.47
Postoperative delirium is another common complication of orthopedic surgery in the geriatric population, increasing morbidity and mortality. The incidence is reported to be as high as 53.3% in the trauma setting and 28.3% in the elective setting, indicating a need to assess patient risk preoperatively.48,49 Several factors contribute to the high incidence of delirium, including advanced age, longer surgical durations, intraoperative hypotension and hypercapnia, pre-existing cognitive dysfunction, and postoperative sleep disorders.50
VTE is another common cause of complications following orthopedic surgery. The development of DVT can lead to subsequent pulmonary embolism, which can be fatal. Orthopedic surgery patients are already at higher risk of DVT and VTE than other surgical patients, with an incidence as high as 40% to 60%, though it is frequently asymptomatic.51,52 Geriatric patients may be more likely to have concurrent comorbidities that increase hypercoagulability.53 Congestive heart failure, chronic kidney disease, and cardiovascular disease are all more common in older individuals and can increase the risk of VTE by 2-fold.53 While anticoagulation is the standard of care to prevent VTE after orthopedic surgery, geriatric patients require more careful monitoring due to the higher incidence of bleeding complications. Additionally, early postoperative mobilization is critical to reduce the risk of DVT without significantly increasing pain or causing other adverse events.54
Respiratory complications are common after orthopedic surgery, particularly atelectasis and bronchospasm, which can result from intraoperative mechanical ventilation.55 While these conditions are typically self-limiting, more severe respiratory issues such as pneumonia are a significant concern because they may lead to the need for mechanical ventilation and admission to the intensive care unit (ICU). The more severe complications have an incidence of about 1% to 2% in orthopedic surgery patients.56 Preventive strategies, such as respiratory physiotherapy and guided breathing exercises, are crucial to minimize perioperative pulmonary complications and promote optimal recovery. Addressing these challenges through early intervention is essential to improve outcomes.
Multidisciplinary Perioperative Care
Multidisciplinary care in orthopedic surgery involves collaborative management of patient care by general practitioners, surgeons, anesthesiologists, dietitians, physical and occupational therapists, inpatient health care practitioners (HCPs), and social services. The goal of this form of care is to provide a longitudinal sequence of health-optimization tactics that prepare a patient for surgery and give them the best chance of postoperative recovery.
Given that many aspects of geriatric health play a role in orthopedic postoperative outcomes, there are many preoperative factors to consider. As previously discussed, preoperative evaluation of geriatric patients should include nutritional and fragility screening to determine surgical candidacy and target modifiable risk factors for risk reduction. This screening can be conducted by primary care practitioners and orthopedic surgeons in an outpatient setting. A multidisciplinary approach benefits patients by decreasing time to surgery.35
Several large studies have demonstrated the positive influence of a multidisciplinary approach on patient-oriented outcomes in orthopedic patients. Incorporation of this style of care in contrast to surgeon-led perioperative optimization leads to fewer floor and ICU admissions, shorter lengths of stay, and decreased mortality rates.35,57 These findings are broadly applicable to a wide range of orthopedic surgeries and even surgeries outside of the musculoskeletal system.58,59 In addition, this strategy has demonstrated reduced in-hospital health care costs due to shorter lengths of stay and fewer ICU admissions. Physical and occupational therapy also have irreplaceable roles in outcomes after orthopedic surgeries. They have independently been shown to decrease pain, increase range of motion, and increase functionality in daily life.60 These aspects of recovery are essential for geriatric well-being.
Screening Tools
The World Health Organization FRAX fracture risk assessment tool (www.fraxplus.org/calculation-tool) was developed to identify patients at high risk of fracture and subsequent complications and to guide clinical decision-making regarding pharmacologic interventions.61 FRAX calculates the 10-year probability of fracture based on demographic factors, such as age and body mass index, clinical measures (eg, femoral neck bone mineral density), and risk factors (eg, prior fragility fractures, substance use history, and prolonged glucocorticoid use).61 The online tool is easy to use, making it a valuable resource for assessing fracture risk and determining appropriate treatment strategies.
The fatigue, resistance, ambulation, illnesses, loss of weight (FRAIL) scale assesses frailty in older adults. The scale classifies patients into 3 categories: robust, prefrail, and frail. The frail category is associated with an increased frequency of hip fracture and an elevated ASA class.62 Additionally, the FRAIL scale has demonstrated value in predicting hospital length of stay and the risk of postoperative complications.62 It also has shown utility in quantifying frailty status, which is traditionally challenging to assess systematically.63
The Mini-Cog is commonly used in geriatric populations to screen for cognitive impairment. Preoperative Mini-Cog screening has been shown to predict the development of postoperative complications.64 Geriatric patients who screened positive for cognitive impairment prior to orthopedic surgery were more likely to develop postoperative delirium, require alternative discharge disposition, and have a longer hospital length of stay.64 Mini-Cog serves as an important preoperative tool for identifying patients who may benefit from closer postoperative monitoring and tailored care.
The Comprehensive Geriatric Assessment (CGA) is a multidimensional evaluation that has been validated for use in geriatric patients undergoing orthopedic surgery.65 The CGA assesses functional status and the ability to perform activities of daily living (ADLs), such as eating, dressing, and ambulating. Poor ADLs are associated with increased risk of falls and cardiopulmonary complications. The CGA allows HCPs to identify patients at higher risk of complications and tailor interventions that optimize functional recovery during the perioperative period.
Nutritional screening is another component of preoperative evaluation in older adults undergoing orthopedic surgery. The Perioperative Nutrition Screen is a preoperative phone assessment of unintentional weight loss in the past 6 months.66 Patients who screen positive are asked to come in for a preoperative visit with a registered dietitian who can further evaluate the nutritional status of the patient.
The Mini Nutritional Assessment Short Form (MNA-SF), Malnutrition Universal Screening Tool, and Nutrition Risk Screening 2002 have all been validated for use in older patients undergoing orthopedic surgery. Among these, the MNA-SF has demonstrated superior utility in predicting hospital readmission and mortality.67 Given the established links between malnutrition and poor surgical outcomes, routine nutritional screening is important for identifying geriatric patients who may require preoperative nutritional interventions.
CONCLUSIONS AND RECOMMENDATIONS
Perioperative management of geriatric patients undergoing orthopedic surgery requires an assessment and strategy focused on risk stratification, patient optimization, and mitigation of potential complications and mortality. Due to the complexity and comprehensive nature of an optimal perioperative plan, creating the plan early is essential to ensure adequate time for patient optimization and care coordination.
Nutrition plays a critical role in the success of surgical procedures, and orthopedics is no exception. Extra care should be taken to preoperatively optimize patient bone health before surgical intervention to enhance recovery and reduce the risk of complications. After an appropriate patient history and clinical picture are gathered, screening tools should be used on a case-by-case basis to further characterize comorbid conditions that may contribute to suboptimal outcomes. Additionally, given the proven association between frailty and fracture risk, frailty serves as a readily quantifiable predictor of patient-oriented outcomes. This should be assessed preoperatively with appropriate risk-stratification tools to determine appropriate postoperative measures to prevent morbidity and mortality.
Orthopedic surgery is increasingly common in geriatric patients, who face higher perioperative risks due to age-related physiological changes, multimorbidity, and frailty. Optimizing preoperative assessment and adopting a multidisciplinary approach—integrating surgeons, anesthesiologists, physical therapists, and dietitians—can improve outcomes, reduce complications, and enhance recovery. The successful use of the tools and strategies outlined in this article by primary care should facilitate access to and recovery from orthopedic surgery in the geriatric population.
More than 40 million surgeries are performed annually in the United States, of which > 18 million are orthopedic, including > 1 million emergency orthopedic surgeries and > 2 million joint replacements.1-4 Notably, > 50% of patients undergoing orthopedic surgery are aged ≥ 65 years, a demographic shift driven by longer life expectancies and an increasing number of older adults remaining physically active for extended periods.5 Osteoarthritis, the most common joint disease, affects 10% of men and 18% of women aged > 60 years, often necessitating an orthopedic joint replacement.6 Perioperative morbidity and mortality are 2.9- to 6.7-times higher in older adults compared with younger adults.7 These risks include infection, venous thromboembolism (VTE), pressure ulcers, reduced mobility, and increased mortality. Due to the high incidence of these complications in older surgical patients, special perioperative protocols and considerations are needed when preparing an older patient for surgery. This review aims to establish concrete considerations and guidelines for perioperative management.
METHODOLOGY
A literature review of PubMed, Google Scholar, and IEEE Xplore identified research on perioperative challenges in geriatric orthopedic surgery. Keywords included geriatrics and orthopedic surgery, perioperative care in geriatric populations, and orthopedic perioperative care. Inclusion criteria were strictly defined to ensure relevance to the geriatric population, with studies focusing on patients aged ≥ 65 years. Exclusion criteria were applied to remove studies that did not involve geriatric populations or orthopedic surgeries or that lacked a clear perioperative focus. Studies were analyzed for design, interventions, and outcomes. Special attention was given to identifying common challenges and trends related to perioperative considerations. We developed a narrative report providing a comprehensive overview of the current understanding of perioperative care for geriatric orthopedic patients to offer practical recommendations for clinicians to use in their practice.
RESULTS
Consistent with the narrative review methodology described, the literature search yielded a broad range of publications addressing perioperative considerations in geriatric orthopedic patients. Articles were screened for relevance to patients aged ≥ 65 years undergoing orthopedic surgery and for applicability to perioperative optimization and postoperative outcomes. Given the heterogeneity in study design, population characteristics, and outcome reporting, findings are presented descriptively rather than being quantitatively pooled. Studies not focused on geriatric populations, orthopedic procedures, or perioperative management were excluded. Key themes included multimorbidity and comorbidity optimization, age-related physiologic changes, frailty assessment and fracture risk stratification, nutritional and bone health management, mechanism of injury considerations, prevention of postoperative complications, and the role of multidisciplinary perioperative care.
Unique Physiological Challenges
The aging process induces a range of physiological changes that can increase morbidity and mortality following surgery. One of the most essential elements to surgical recovery is wound healing, as impairments in this process can lead to adverse events, including infection, cosmetic deformity, and wound dehiscence. The general paradigm of aging involves cell senescence resulting in slower or disorganized functional capacity of these cells.8 While wound healing in older individuals was once thought to be defective, recent research has demonstrated that this process is not absent, but delayed.9
Wound healing is a tightly regulated and evolutionarily conserved process that proceeds through 3 main phases: inflammation, proliferation, and remodeling. Re-epithelialization begins with the migration of epithelial cells from hair follicles, sweat glands, or wound margins (depending on wound depth) and is influenced by oxygen levels, moisture, and growth factors.9 Several characteristics of aged skin contribute to the delayed healing process. Aged skin has fewer hair follicles and eccrine sweat glands, as well as decreased follicle thickness.10 This results in fewer proliferating cells for wound healing and lower amounts of sebum production for skin moisture.11 Furthermore, aged fibroblasts are fewer in number and less effective in synthesizing extracellular matrices, resulting in slower and less tensile wound healing.12 Additionally, microvascular changes associated with aging result in disorganized vasculature, which impairs oxygen delivery to the wound bed and diminishes the influx of proinflammatory cells necessary for effective healing.13 These senescent traits of aged skin contribute to the delayed wound healing process found in geriatric patients.
Compounding these age-related factors is the prevalence of multimorbidity, or coexisting chronic diagnoses, in 55% to 98% of older patients.14 Common comorbidities include peripheral arterial disease, chronic venous insufficiency, type 1 and type 2 diabetes, neoplasms, atherosclerotic disease, and hypertension. Older patients are more likely to be prescribed corticosteroids and chemotherapeutic agents that impair the function of inflammatory cells necessary for wound healing.15,16 Additionally, decreased mobility is more common in geriatric patients, which can increase the risk of wound formation, particularly pressure ulcers.17
Perioperative Considerations
All surgical patients undergo a formal or informal preoperative evaluation to assess their fitness for surgery, with the goal of minimizing both anesthesia-related risks and postoperative complications. A widely used tool in this assessment is the American Society of Anesthesiologists (ASA) physical status classification, which stratifies patients into 6 categories based on their medical history and overall health status.18 Classes range from healthy patients (Class I) to organ donors who are brain-dead (Class VI).
Cardiac optimization is an essential component of preoperative evaluation for older adults due to their higher risk of underlying cardiovascular disease.19 This process involves an in-depth review of the patient’s cardiac history, including the timing and nature of any prior interventions and the recurrence rate. Functional capacity is assessed through metabolic equivalents, where a threshold of > 4 metabolic equivalents (the ability to walk up a flight of stairs) is considered adequate for surgery. Risk is assessed based on the specific surgical procedure, and nonemergent orthopedic procedures are considered intermediate risk. If a patient is deemed high risk at any stage of this evaluation, further cardiac testing is indicated.
Pulmonary optimization is typically necessary for geriatric patients, who are more likely to have conditions such as chronic obstructive pulmonary disease or interstitial lung disease.14,20 In patients without severe systemic lung disease, pulmonary optimization involves assessing the functional expiratory volume and diffusing capacity for carbon monoxide. In addition, aggressive modification of risk factors, such as smoking cessation, is strongly recommended.
Additional perioperative conditions are disease-specific and involve evaluation of comorbid illnesses and recognition of absolute contraindications to noncardiac surgery. For instance, an ejection fraction of < 35%, a history of myocardial infarction within 6 months, or active diabetic ketoacidosis are all absolute contraindications to elective surgery. For orthopedic procedures, additional contraindications include symptomatic bacteremia, active joint or local tissue infection, severe malnutrition, uncontrolled metabolic syndrome or chronic disease, untreated immunodeficiency, and active deep venous thrombosis (DVT) or pulmonary embolism.21
Bone Health and Nutrition
In the context of orthopedic surgery, the hallmark of clinically defined optimal bone health is a musculoskeletal system that provides the ability for pain-free functional and occupational tasks with an adequate capacity to withstand the mechanical forces imparted by everyday life. Back pain and arthritis are the fourth- and sixth-most common complaints in primary care, underscoring suboptimal bone health management in developed countries.22
Optimizing bone health through proper nutrition is crucial in the perioperative management of geriatric orthopedic patients. The clinical diagnosis of malnutrition has well-studied associations with worse outcomes after orthopedic surgery, which include increased mortality, hospital length of stay, readmission rates, and health institution spending.23-25 Some studies show that up to 60% of geriatric patients may be malnourished.26
Regarding vitamin and mineral supplements, the general consensus before orthopedic surgery is that vitamins A, C, D, and E, and zinc are predictive in determining postoperative health.27 However, Curtis et al state that therapy should be targeted at correcting relative deficiencies; supraphysiologic concentrations of these vitamins do not appear to be helpful.27 This claim may merit serum studies to rule out deficiencies. Dietitians should be involved in the creation of a patient care plan in the spirit of multidisciplinary orthopedic surgery approaches, which have proven to result in superior patient outcomes.28 Additionally, directive counseling should be provided when necessary.
In patients with adequately managed nutrition, 7 to 10 days of diet optimization is typically sufficient, but patients with malnutrition may require sustained nutritional support for up to 6 weeks; a standardized time for adequate nutrition supplementation has not been identified.25-27 Postoperative management is similar in older patients who are malnourished and those receiving adequate nutrition after orthopedic surgery, which typically involves 3 weeks of a high-protein diet.26
Evaluating Mechanisms of Injury
Assessing the mechanism of injury (MOI) is essential to developing an appropriate and successful orthopedic treatment plan. MOI is typically categorized as low energy, which consists of ground-level falls and other minor trauma, or high energy, which can include motor vehicle crashes or falls from a height.29 Unlike younger patients who typically experience trauma from high-energy MOIs, geriatric patients often sustain fractures from low-energy MOIs. The importance of assessing MOI for the geriatric population is magnified as it provides vital clues that not only help determine the nature of the injury, but also highlight underlying frailty, comorbidities, and potential complications. Weakness or deconditioning related to older age is often not discovered before trauma, which is why assessing the MOI can provide valuable information regarding overall patient health.30

The MOI of trauma also is correlated with factors that influence postoperative recovery and overall prognosis (Figure). Falls comprise more than three-quarters of the MOI in geriatric patients with trauma, and > 90% of these falls are ground-level or other simple falls.30 Falls secondary to an intrinsic disorder, rather than an extrinsic environmental hazard, are more common in geriatric patients.31
These events may be associated with an underlying medical condition, such as osteopenia, osteoporosis, or neuromuscular disorders, such as Parkinson disease.32 They may also be attributed to normal age-related changes, such as decreased visual acuity, reduced reaction time, or mild cognitive impairment.30 An estimated 6% to 35% of geriatric patients who present to the emergency department have some degree of cognitive dysfunction.33 Accordingly, a thorough understanding of the events leading up to injury is vital for the management of older patients. Knowing the specific circumstances of a fall can provide insight into the patient’s gait, balance, and need for further investigations such as cognitive screening or evaluation of home safety. This information can guide decisions regarding preoperative optimization of medications and postoperative rehabilitation interventions.
Frailty and Risk of Fracture
Frailty is a clinical syndrome defined by overall decreased capacity for the body’s adaptive changes to various stressors.34 It is a common condition in geriatric populations due to cumulative degenerative changes and multisystem decline over a lifetime’s worth of disruptions to natural homeostasis.34 In orthopedics, frailty typically refers to musculoskeletal durability and resilience in response to mechanical forces (ie, falls, trauma, and high-acceleration movements). Globally, > 200 million people have osteoporotic frailty, leading to 9 million hip fractures annually.35 More than 30% of people aged ≥ 65 years fall ≥ 1 time per calendar year.36
Assessing frailty in geriatric patients undergoing orthopedic surgery is vital, as it predisposes patients to higher rates of morbidity, mortality, and institutionalization, particularly from falls and resultant fragility fractures.37-39 This is true for a wide range of orthopedic procedures, spanning elective to urgent surgeries and involving the axial and appendicular skeleton.40,41 Given the high rates of fractures, subsequent patient morbidity, and financial burden on the health care system, effective frailty screening is essential.
There are many strategies to assess frailty risk and subsequent fracture risk.42 Questionnaires or online medical calculators serve as easy-to-use tools for assessment of frailty or associated predictors of fragility fractures. Validated assessment tools are provided in Table 1.
Dual-emission X-ray absorptiometry is a well-established way to determine bone density and establish fracture risk. The Fried Frailty Phenotype score and Short Performance Physical Battery test are clinically applicable methods of assessing frailty in older outpatient populations. Although these examinations focus on different aspects of the patient, they have moderate agreeability, are sensitive, and can be readily performed in the clinical setting as demonstrated by a > 90% patient participation rate for both methods.42 Finally, several serum studies can be predictive of frailty, the most readily modifiable of which are vitamin D3, ferritin, albumin, and calcium.43 Although they are more invasive for the patient, serum studies can provide additional modifiable targets for perioperative optimization and contribute to risk stratification.
Risk stratification should take place around 6 weeks before surgery, which should provide adequate time for rectification of preoperative barriers to elective surgical intervention—namely nutritional status. In cases of urgent or emergent procedures (ie, femoral neck fracture with concern for avascular necrosis of the femoral head), this may not be possible but should be conducted nonetheless for patient-specific postoperative rehabilitation and risk reduction.


Postoperative Complication Risks
Postoperative complications affect nearly 15% of geriatric orthopedic patients, highlighting the need for comprehensive preoperative evaluations to assess risk factors.44 Age-related physiological changes, frailty, and comorbidities complicate recovery and management (Table 2).
Wound healing is impaired in older individuals due to suboptimal circulation and decreased oxygenation that is secondary to age-related changes, as well as the increased likelihood of comorbid conditions (eg, diabetes).7 Surgical site infections can be particularly malicious in geriatric patients, with a 4% incidence.45,46 Hospitalization can be prolonged by a mean 2 weeks, which increases the risk of hospital-associated delirium and iatrogenic complications.46 Both the mortality rate and costs associated with hospitalization are higher for older patients who develop surgical site infections compared with patients aged < 65 years, underscoring the importance of vigilant monitoring, early detection, and effective preoperative screening to identify and manage modifiable risk factors.47
Postoperative delirium is another common complication of orthopedic surgery in the geriatric population, increasing morbidity and mortality. The incidence is reported to be as high as 53.3% in the trauma setting and 28.3% in the elective setting, indicating a need to assess patient risk preoperatively.48,49 Several factors contribute to the high incidence of delirium, including advanced age, longer surgical durations, intraoperative hypotension and hypercapnia, pre-existing cognitive dysfunction, and postoperative sleep disorders.50
VTE is another common cause of complications following orthopedic surgery. The development of DVT can lead to subsequent pulmonary embolism, which can be fatal. Orthopedic surgery patients are already at higher risk of DVT and VTE than other surgical patients, with an incidence as high as 40% to 60%, though it is frequently asymptomatic.51,52 Geriatric patients may be more likely to have concurrent comorbidities that increase hypercoagulability.53 Congestive heart failure, chronic kidney disease, and cardiovascular disease are all more common in older individuals and can increase the risk of VTE by 2-fold.53 While anticoagulation is the standard of care to prevent VTE after orthopedic surgery, geriatric patients require more careful monitoring due to the higher incidence of bleeding complications. Additionally, early postoperative mobilization is critical to reduce the risk of DVT without significantly increasing pain or causing other adverse events.54
Respiratory complications are common after orthopedic surgery, particularly atelectasis and bronchospasm, which can result from intraoperative mechanical ventilation.55 While these conditions are typically self-limiting, more severe respiratory issues such as pneumonia are a significant concern because they may lead to the need for mechanical ventilation and admission to the intensive care unit (ICU). The more severe complications have an incidence of about 1% to 2% in orthopedic surgery patients.56 Preventive strategies, such as respiratory physiotherapy and guided breathing exercises, are crucial to minimize perioperative pulmonary complications and promote optimal recovery. Addressing these challenges through early intervention is essential to improve outcomes.
Multidisciplinary Perioperative Care
Multidisciplinary care in orthopedic surgery involves collaborative management of patient care by general practitioners, surgeons, anesthesiologists, dietitians, physical and occupational therapists, inpatient health care practitioners (HCPs), and social services. The goal of this form of care is to provide a longitudinal sequence of health-optimization tactics that prepare a patient for surgery and give them the best chance of postoperative recovery.
Given that many aspects of geriatric health play a role in orthopedic postoperative outcomes, there are many preoperative factors to consider. As previously discussed, preoperative evaluation of geriatric patients should include nutritional and fragility screening to determine surgical candidacy and target modifiable risk factors for risk reduction. This screening can be conducted by primary care practitioners and orthopedic surgeons in an outpatient setting. A multidisciplinary approach benefits patients by decreasing time to surgery.35
Several large studies have demonstrated the positive influence of a multidisciplinary approach on patient-oriented outcomes in orthopedic patients. Incorporation of this style of care in contrast to surgeon-led perioperative optimization leads to fewer floor and ICU admissions, shorter lengths of stay, and decreased mortality rates.35,57 These findings are broadly applicable to a wide range of orthopedic surgeries and even surgeries outside of the musculoskeletal system.58,59 In addition, this strategy has demonstrated reduced in-hospital health care costs due to shorter lengths of stay and fewer ICU admissions. Physical and occupational therapy also have irreplaceable roles in outcomes after orthopedic surgeries. They have independently been shown to decrease pain, increase range of motion, and increase functionality in daily life.60 These aspects of recovery are essential for geriatric well-being.
Screening Tools
The World Health Organization FRAX fracture risk assessment tool (www.fraxplus.org/calculation-tool) was developed to identify patients at high risk of fracture and subsequent complications and to guide clinical decision-making regarding pharmacologic interventions.61 FRAX calculates the 10-year probability of fracture based on demographic factors, such as age and body mass index, clinical measures (eg, femoral neck bone mineral density), and risk factors (eg, prior fragility fractures, substance use history, and prolonged glucocorticoid use).61 The online tool is easy to use, making it a valuable resource for assessing fracture risk and determining appropriate treatment strategies.
The fatigue, resistance, ambulation, illnesses, loss of weight (FRAIL) scale assesses frailty in older adults. The scale classifies patients into 3 categories: robust, prefrail, and frail. The frail category is associated with an increased frequency of hip fracture and an elevated ASA class.62 Additionally, the FRAIL scale has demonstrated value in predicting hospital length of stay and the risk of postoperative complications.62 It also has shown utility in quantifying frailty status, which is traditionally challenging to assess systematically.63
The Mini-Cog is commonly used in geriatric populations to screen for cognitive impairment. Preoperative Mini-Cog screening has been shown to predict the development of postoperative complications.64 Geriatric patients who screened positive for cognitive impairment prior to orthopedic surgery were more likely to develop postoperative delirium, require alternative discharge disposition, and have a longer hospital length of stay.64 Mini-Cog serves as an important preoperative tool for identifying patients who may benefit from closer postoperative monitoring and tailored care.
The Comprehensive Geriatric Assessment (CGA) is a multidimensional evaluation that has been validated for use in geriatric patients undergoing orthopedic surgery.65 The CGA assesses functional status and the ability to perform activities of daily living (ADLs), such as eating, dressing, and ambulating. Poor ADLs are associated with increased risk of falls and cardiopulmonary complications. The CGA allows HCPs to identify patients at higher risk of complications and tailor interventions that optimize functional recovery during the perioperative period.
Nutritional screening is another component of preoperative evaluation in older adults undergoing orthopedic surgery. The Perioperative Nutrition Screen is a preoperative phone assessment of unintentional weight loss in the past 6 months.66 Patients who screen positive are asked to come in for a preoperative visit with a registered dietitian who can further evaluate the nutritional status of the patient.
The Mini Nutritional Assessment Short Form (MNA-SF), Malnutrition Universal Screening Tool, and Nutrition Risk Screening 2002 have all been validated for use in older patients undergoing orthopedic surgery. Among these, the MNA-SF has demonstrated superior utility in predicting hospital readmission and mortality.67 Given the established links between malnutrition and poor surgical outcomes, routine nutritional screening is important for identifying geriatric patients who may require preoperative nutritional interventions.
CONCLUSIONS AND RECOMMENDATIONS
Perioperative management of geriatric patients undergoing orthopedic surgery requires an assessment and strategy focused on risk stratification, patient optimization, and mitigation of potential complications and mortality. Due to the complexity and comprehensive nature of an optimal perioperative plan, creating the plan early is essential to ensure adequate time for patient optimization and care coordination.
Nutrition plays a critical role in the success of surgical procedures, and orthopedics is no exception. Extra care should be taken to preoperatively optimize patient bone health before surgical intervention to enhance recovery and reduce the risk of complications. After an appropriate patient history and clinical picture are gathered, screening tools should be used on a case-by-case basis to further characterize comorbid conditions that may contribute to suboptimal outcomes. Additionally, given the proven association between frailty and fracture risk, frailty serves as a readily quantifiable predictor of patient-oriented outcomes. This should be assessed preoperatively with appropriate risk-stratification tools to determine appropriate postoperative measures to prevent morbidity and mortality.
Orthopedic surgery is increasingly common in geriatric patients, who face higher perioperative risks due to age-related physiological changes, multimorbidity, and frailty. Optimizing preoperative assessment and adopting a multidisciplinary approach—integrating surgeons, anesthesiologists, physical therapists, and dietitians—can improve outcomes, reduce complications, and enhance recovery. The successful use of the tools and strategies outlined in this article by primary care should facilitate access to and recovery from orthopedic surgery in the geriatric population.
Dobson GP. Trauma of major surgery: a global problem that is not going away. Int J Surg. 2020;81:47-54. doi:10.1016/j.ijsu.2020.07.017
United States (US) orthopedic procedures count by segments and forecast to 2030. GlobalData. February 17, 2023. Accessed April 29, 2026. https://www.globaldata.com/store/report/usa-orthopedic-procedures-analysis/
Jarman MP, Weaver MJ, Haider AH, Salim A, Harris MB. The national burden of orthopedic injury: cross-sectional estimates for trauma system planning and optimization. J Surg Res. 2020;249:197-204. doi:10.1016/j.jss.2019.12.023
Hegde V, Stambough JB, Levine BR, et al. Highlights of the 2022 American Joint Replacement Registry Annual Report. Arthroplast Today. 2023;21:101137. doi:10.1016/j.artd.2023.101137
Nakamura K, Ogata T. Locomotive syndrome: definition and management. Clin Rev Bone Miner Metab. 2016;14:56-67. doi:10.1007/s12018-016-9208-2
Glyn-Jones S, Palmer AJR, Agricola R, et al. Osteoarthritis. Lancet. 2015;386:376-387. doi:10.1016/S0140-6736(14)60802-3
Hughes S, Leary A, Zweizig S, Cain J. Surgery in elderly people: preoperative, operative and postoperative care to assist healing. Best Pract Res Clin Obstet Gynaecol. 2013;27:753-765. doi:10.1016/j.bpobgyn.2013.02.006
Regulski MJ. Cellular senescence: what, why, and how. Wounds. 2017;29:168-174.
Kremer M, Burkemper N. Aging skin and wound healing. Clin Geriatr Med. 2024;40:1-10. doi:10.1016/j.cger.2023.06.001
Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol. 1986;15:571-585. doi:10.1016/S0190-9622(86)70208-9
Van Neste D, Tobin DJ. Hair cycle and hair pigmentation: dynamic interactions and changes associated with aging. Micron. 2004;35:193-200. doi:10.1016/j.micron.2003.11.006
Salzer MC, Lafzi A, Berenguer-Llergo A, et al. Identity noise and adipogenic traits characterize dermal fibroblast aging. Cell. 2018;175:1575-1590.e22. doi:10.1016/j.cell.2018.10.012
Jin K. A microcirculatory theory of aging. Aging Dis. 2019;10:676-683. doi:10.14336/AD.2019.0315
Marengoni A, Angleman S, Melis R, et al. Aging with multimorbidity: a systematic review of the literature. Ageing Res Rev. 2011;10:430-439. doi:10.1016/j.arr.2011.03.003
Waljee AK, Rogers MAM, Lin P, et al. Short term use of oral corticosteroids and related harms among adults in the United States: population based cohort study. BMJ. 2017;357:j1415. doi:10.1136/bmj.j1415
Given B, Given CW. Older adults and cancer treatment. Cancer. 2008;113:3505-3511. doi:10.1002/cncr.23939
Ferrucci L, Cooper R, Shardell M, Simonsick EM, Schrack JA, Kuh D. Age-related change in mobility: perspectives from life course epidemiology and geroscience. J Gerontol A Biol Sci Med Sci. 2016;71:1184-1194. doi:10.1093/gerona/glw043
Mayhew D, Mendonca V, Murthy BVS. A review of ASA physical status - historical perspectives and modern developments. Anaesthesia. 2019;74:373-379. doi:10.1111/anae.14569
Eagle KA, Berger PB, Calkins H, et al. ACC/AHA guideline update for perioperative cardiovascular evaluation for noncardiac surgery—executive summary a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1996 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation. 2002;105:1257-1267. doi:10.1161/circ.105.10.1257
Bapoje SR, Whitaker JF, Schulz T, Chu ES, Albert RK. Preoperative evaluation of the patient with pulmonary disease. Chest. 2007;132:1637-1645. doi:10.1378/chest.07-0347
Choe H, Indelli PF, Ricciardi B, et al. What are the absolute contraindications for elective total knee or hip arthroplasty? J Arthroplasty. 2025;40(2 suppl 1):S45-S47. doi:10.1016/j.arth.2024.10.041
Finley CR, Chan DS, Garrison S, et al. What are the most common conditions in primary care? Systematic review. Can Fam Physician. 2018;64:832-840.
Vaid S, Bell T, Grim R, Ahuja V. Predicting risk of death in general surgery patients on the basis of preoperative variables using American College of Surgeons National Surgical Quality Improvement Program data. Perm J. 2012;16:10-17. doi:10.7812/TPP/12-019
Correia MTD, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22:235-239. doi:10.1016/S0261-5614(02)00215-7
Friedman J, Lussiez A, Sullivan J, Wang S, Englesbe M. Implications of sarcopenia in major surgery. Nutr Clin Pract. 2015;30:175-179. doi:10.1177/0884533615569888
Hirsch KR, Wolfe RR, Ferrando AA. Pre- and post-surgical nutrition for preservation of muscle mass, strength, and functionality following orthopedic surgery. Nutrients. 2021;13:1675. doi:10.3390/nu13051675
Curtis W, Choi T, Ahmad A, Shultz C. Perioperative nutritional considerations in orthopaedic surgery: a review of the literature. West J Orthop. 2023;12:1. https://digitalrepository.unm.edu/wjo/vol12/iss1/1
Wischmeyer PE, Carli F, Evans DC, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on nutrition screening and therapy within a surgical enhanced recovery pathway. Anesth Analg. 2018;126:1883-1895. doi:10.1213/ANE.0000000000002743
Mun F, Ringenbach K, Baer B, et al. Factors influencing geriatric orthopaedic trauma mortality. Injury. 2022;53:919-924. doi:10.1016/j.injury.2022.01.005
Bonne S, Schuerer DJE. Trauma in the older adult: epidemiology and evolving geriatric trauma principles. Clin Geriatr Med. 2013;29:137-150. doi:10.1016/j.cger.2012.10.008
Montero-Odasso MM. Falls as a geriatric syndrome: mechanisms and risk identification. In: Duque G, Kiel DP, eds. Osteoporosis in Older Persons: Advances in Pathophysiology and Therapeutic Approaches. 2nd ed. Springer International Publishing; 2016:171-186. doi:10.1007/978-3-319-25976-5_10
Lach HW, Reed AT, Arfken CL, et al. Falls in the elderly: reliability of a classification system. J Am Geriatr Soc. 1991;39:197-202. doi:10.1111/j.1532-5415.1991.tb01626.x
Carpenter CR, DesPain B, Keeling TN, Shah M, Rothenberger M. The six-item screener and AD8 for the detection of cognitive impairment in geriatric emergency department patients. Ann Emerg Med. 2011;57:653-661. doi:10.1016/j.annemergmed.2010.06.560
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381:752-762. doi:10.1016/S0140-6736(12)62167-9
Patel JN, Klein DS, Sreekumar S, Liporace FA, Yoon RS. Outcomes in multidisciplinary team-based approach in geriatric hip fracture care: a systematic review. J Am Acad Orthop Surg. 2020;28:128-133. doi:10.5435/JAAOS-D-18-00425
Amador LF, Loera JA. Preventing postoperative falls in the older adult. J Am Coll Surg. 2007;204:447-453. doi:10.1016/j.jamcollsurg.2006.12.010
Tembo MC, Holloway-Kew KL, Mohebbi M, et al. The association between a fracture risk tool and frailty: Geelong Osteoporosis Study. BMC Geriatr. 2020;20:196. doi:10.1186/s12877-020-01595-8
Demiris¸ B, Basat S, Kurt F, Aksakal B, Basat O. Evaluation of the relationship between frailty and fracture risk using Fracture Risk Assessment Tool in patients 65 years and over. South Clin Istanb Eurasia. 2023;34:42-48. doi:10.14744/scie.2022.66564
Partridge JSL, Harari D, Dhesi JK. Frailty in the older surgical patient: a review. Age Ageing. 2012;41:142-147. doi:10.1093/ageing/afr182
Mamtora PH, Fortier MA, Barnett SR, Schmid LN, Kain ZN. Peri-operative management of frailty in the orthopedic patient. J Orthop. 2020;22:304-307. doi:10.1016/j.jor.2020.05.024
Leven DM, Lee NJ, Kim JS, et al. Frailty is predictive of adverse postoperative events in patients undergoing lumbar fusion. Global Spine J. 2017;7:529-535. doi:10.1177/2192568217700099
Pritchard JM, Kennedy CC, Karampatos S, et al. Measuring frailty in clinical practice: a comparison of physical frailty assessment methods in a geriatric out-patient clinic. BMC Geriatr. 2017;17:264. doi:10.1186/s12877-017-0623-0
Kumar A, Dhar M, Agarwal M, Mukherjee A, Saxena V. Predictors of frailty in the elderly population: a cross-sectional study at a tertiary care center. Cureus. 2022;14:e30557. doi:10.7759/cureus.30557
Scarano KA, Philp FH, Westrick ER, Altman GT, Altman DT. Evaluating postoperative complications and outcomes of orthopedic fracture repair in nonagenarian patients. Geriatr Orthop Surg Rehabil. 2018;9:2151459318758106. doi:10.1177/2151459318758106
Liang Z, Rong K, Gu W, et al. Surgical site infection following elective orthopaedic surgeries in geriatric patients: incidence and associated risk factors. Int Wound J. 2019;16:773-780. doi:10.1111/iwj.13096
Ren M, Liang W, Wu Z, Zhao H, Wang J. Risk factors of surgical site infection in geriatric orthopedic surgery: a retrospective multicenter cohort study. Geriatr Gerontol Int. 2019;19:213-217. doi:10.1111/ggi.13590
Kaye KS, Schmader KE, Sawyer R. Surgical site infection in the elderly population. Clin Infect Dis. 2004;39:1835-1841. doi:10.1086/425744
Bruce AJ, Ritchie CW, Blizard R, Lai R, Raven P. The incidence of delirium associated with orthopedic surgery: a meta-analytic review. Int Psychogeriatr. 2007;19:197-214. doi:10.1017/S104161020600425X
Williams-Russo P, Urquhart BL, Sharrock NE, Charlson ME. Post-operative delirium: predictors and prognosis in elderly orthopedic patients. J Am Geriatr Soc. 1992;40:759-767. doi:10.1111/j.1532-5415.1992.tb01846.x
Wang J, Li Z, Yu Y, Li B, Shao G, Wang Q. Risk factors contributing to postoperative delirium in geriatric patients postorthopedic surgery. Asia Pac Psychiatry. 2015;7:375-382. doi:10.1111/appy.12193
Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126:338S-400S. doi:10.1378/chest.126.3_suppl.338S
Kahn SR, Shivakumar S. What’s new in VTE risk and prevention in orthopedic surgery. Res Pract Thromb Haemost. 2020;4:366-376. doi:10.1002/rth2.12323
Uzel K, Azboy I·, Parvizi J. Venous thromboembolism in orthopedic surgery: global guidelines. Acta Orthop Traumatol Turc. 2023;57:192-203. doi:10.5152/j.aott.2023.23074
Peck M, Holthaus A, Kingsbury K, Salsberry MG, Duggirala V. Mobility in acute care for geriatric patients with orthopedic conditions: a review of recent literature. Curr Geri Rep. 2020;9:300-310. doi:10.1007/s13670-020-00347-1
Leme LEG, Sitta MC, Toledo M, Henriques SS. Orthopedic surgery among the elderly: clinical characteristics. Rev Bras Ortop. 2015;46:238-246. doi:10.1016/S2255-4971(15)30189-0
Malcolm TL, Knezevic NN, Zouki CC, Tharian AR. Pulmonary complications after hip and knee arthroplasty in the United States, 2004-2014. Anesth Analg. 2020;130:917-924. doi:10.1213/ANE.0000000000004265
Kamal T, Conway RM, Littlejohn I, Ricketts D. The role of a multidisciplinary pre-assessment clinic in reducing mortality after complex orthopaedic surgery. Ann R Coll Surg Engl. 2011;93:149-151. doi:10.1308/003588411X561026
Davis MJ, Luu BC, Raj S, Abu-Ghname A, Buchanan EP. Multidisciplinary care in surgery: Are team-based interventions cost-effective? Surgeon. 2021;19:49-60. doi:10.1016/j.surge.2020.02.005
Frassanito L, Vergari A, Nestorini R, et al. Enhanced recovery after surgery (ERAS) in hip and knee replacement surgery: description of a multidisciplinary program to improve management of the patients undergoing major orthopedic surgery. Musculoskelet Surg. 2020;104:87-92. doi:10.1007/s12306-019-00603-4
Reddy RS, Alahmari KA, Alshahrani MS, et al. Exploring the impact of physiotherapy on health outcomes in older adults with chronic diseases: a cross-sectional analysis. Front Public Health. 2024;12:1415882. doi:10.3389/fpubh.2024.1415882
Watts NB. The Fracture Risk Assessment Tool (FRAX®): applications in clinical practice. J Womens Health (Larchmt). 2011;20:525-531. doi:10.1089/jwh.2010.2294
Gleason LJ, Benton EA, Alvarez-Nebreda ML, Weaver MJ, Harris MB, Javedan H. FRAIL questionnaire screening tool and short-term outcomes in geriatric fracture patients. J Am Med Dir Assoc. 2017;18:1082-1086. doi:10.1016/j.jamda.2017.07.005
Kojima G. Frailty defined by FRAIL scale as a predictor of mortality: a systematic review and meta-analysis. J Am Med Dir Assoc. 2018;19:480-483. doi:10.1016/j.jamda.2018.04.006
Culley DJ, Flaherty D, Fahey MC, et al. Poor performance on a preoperative cognitive screening test predicts postoperative complications in older orthopedic surgical patients. Anesthesiology. 2017;127:765-774. doi:10.1097/ALN.0000000000001859
Kong C, Zhang Y, Wang C, et al. Comprehensive geriatric assessment for older orthopedic patients and analysis of risk factors for postoperative complications. BMC Geriatr. 2022;22:644. doi:10.1186/s12877-022-03328-5
Williams DGA, Wischmeyer PE. Perioperative nutrition care of orthopedic surgery patient. Tech Orthop. 2020;35:15-18. doi:10.1097/BTO.0000000000000412
Koren-Hakim T, Weiss A, Hershkovitz A, et al. Comparing the adequacy of the MNA-SF, NRS-2002 and MUST nutritional tools in assessing malnutrition in hip fracture operated elderly patients. Clin Nutr. 2016;35:1053-1058. doi:10.1016/j.clnu.2015.07.014
Dobson GP. Trauma of major surgery: a global problem that is not going away. Int J Surg. 2020;81:47-54. doi:10.1016/j.ijsu.2020.07.017
United States (US) orthopedic procedures count by segments and forecast to 2030. GlobalData. February 17, 2023. Accessed April 29, 2026. https://www.globaldata.com/store/report/usa-orthopedic-procedures-analysis/
Jarman MP, Weaver MJ, Haider AH, Salim A, Harris MB. The national burden of orthopedic injury: cross-sectional estimates for trauma system planning and optimization. J Surg Res. 2020;249:197-204. doi:10.1016/j.jss.2019.12.023
Hegde V, Stambough JB, Levine BR, et al. Highlights of the 2022 American Joint Replacement Registry Annual Report. Arthroplast Today. 2023;21:101137. doi:10.1016/j.artd.2023.101137
Nakamura K, Ogata T. Locomotive syndrome: definition and management. Clin Rev Bone Miner Metab. 2016;14:56-67. doi:10.1007/s12018-016-9208-2
Glyn-Jones S, Palmer AJR, Agricola R, et al. Osteoarthritis. Lancet. 2015;386:376-387. doi:10.1016/S0140-6736(14)60802-3
Hughes S, Leary A, Zweizig S, Cain J. Surgery in elderly people: preoperative, operative and postoperative care to assist healing. Best Pract Res Clin Obstet Gynaecol. 2013;27:753-765. doi:10.1016/j.bpobgyn.2013.02.006
Regulski MJ. Cellular senescence: what, why, and how. Wounds. 2017;29:168-174.
Kremer M, Burkemper N. Aging skin and wound healing. Clin Geriatr Med. 2024;40:1-10. doi:10.1016/j.cger.2023.06.001
Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol. 1986;15:571-585. doi:10.1016/S0190-9622(86)70208-9
Van Neste D, Tobin DJ. Hair cycle and hair pigmentation: dynamic interactions and changes associated with aging. Micron. 2004;35:193-200. doi:10.1016/j.micron.2003.11.006
Salzer MC, Lafzi A, Berenguer-Llergo A, et al. Identity noise and adipogenic traits characterize dermal fibroblast aging. Cell. 2018;175:1575-1590.e22. doi:10.1016/j.cell.2018.10.012
Jin K. A microcirculatory theory of aging. Aging Dis. 2019;10:676-683. doi:10.14336/AD.2019.0315
Marengoni A, Angleman S, Melis R, et al. Aging with multimorbidity: a systematic review of the literature. Ageing Res Rev. 2011;10:430-439. doi:10.1016/j.arr.2011.03.003
Waljee AK, Rogers MAM, Lin P, et al. Short term use of oral corticosteroids and related harms among adults in the United States: population based cohort study. BMJ. 2017;357:j1415. doi:10.1136/bmj.j1415
Given B, Given CW. Older adults and cancer treatment. Cancer. 2008;113:3505-3511. doi:10.1002/cncr.23939
Ferrucci L, Cooper R, Shardell M, Simonsick EM, Schrack JA, Kuh D. Age-related change in mobility: perspectives from life course epidemiology and geroscience. J Gerontol A Biol Sci Med Sci. 2016;71:1184-1194. doi:10.1093/gerona/glw043
Mayhew D, Mendonca V, Murthy BVS. A review of ASA physical status - historical perspectives and modern developments. Anaesthesia. 2019;74:373-379. doi:10.1111/anae.14569
Eagle KA, Berger PB, Calkins H, et al. ACC/AHA guideline update for perioperative cardiovascular evaluation for noncardiac surgery—executive summary a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1996 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation. 2002;105:1257-1267. doi:10.1161/circ.105.10.1257
Bapoje SR, Whitaker JF, Schulz T, Chu ES, Albert RK. Preoperative evaluation of the patient with pulmonary disease. Chest. 2007;132:1637-1645. doi:10.1378/chest.07-0347
Choe H, Indelli PF, Ricciardi B, et al. What are the absolute contraindications for elective total knee or hip arthroplasty? J Arthroplasty. 2025;40(2 suppl 1):S45-S47. doi:10.1016/j.arth.2024.10.041
Finley CR, Chan DS, Garrison S, et al. What are the most common conditions in primary care? Systematic review. Can Fam Physician. 2018;64:832-840.
Vaid S, Bell T, Grim R, Ahuja V. Predicting risk of death in general surgery patients on the basis of preoperative variables using American College of Surgeons National Surgical Quality Improvement Program data. Perm J. 2012;16:10-17. doi:10.7812/TPP/12-019
Correia MTD, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22:235-239. doi:10.1016/S0261-5614(02)00215-7
Friedman J, Lussiez A, Sullivan J, Wang S, Englesbe M. Implications of sarcopenia in major surgery. Nutr Clin Pract. 2015;30:175-179. doi:10.1177/0884533615569888
Hirsch KR, Wolfe RR, Ferrando AA. Pre- and post-surgical nutrition for preservation of muscle mass, strength, and functionality following orthopedic surgery. Nutrients. 2021;13:1675. doi:10.3390/nu13051675
Curtis W, Choi T, Ahmad A, Shultz C. Perioperative nutritional considerations in orthopaedic surgery: a review of the literature. West J Orthop. 2023;12:1. https://digitalrepository.unm.edu/wjo/vol12/iss1/1
Wischmeyer PE, Carli F, Evans DC, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on nutrition screening and therapy within a surgical enhanced recovery pathway. Anesth Analg. 2018;126:1883-1895. doi:10.1213/ANE.0000000000002743
Mun F, Ringenbach K, Baer B, et al. Factors influencing geriatric orthopaedic trauma mortality. Injury. 2022;53:919-924. doi:10.1016/j.injury.2022.01.005
Bonne S, Schuerer DJE. Trauma in the older adult: epidemiology and evolving geriatric trauma principles. Clin Geriatr Med. 2013;29:137-150. doi:10.1016/j.cger.2012.10.008
Montero-Odasso MM. Falls as a geriatric syndrome: mechanisms and risk identification. In: Duque G, Kiel DP, eds. Osteoporosis in Older Persons: Advances in Pathophysiology and Therapeutic Approaches. 2nd ed. Springer International Publishing; 2016:171-186. doi:10.1007/978-3-319-25976-5_10
Lach HW, Reed AT, Arfken CL, et al. Falls in the elderly: reliability of a classification system. J Am Geriatr Soc. 1991;39:197-202. doi:10.1111/j.1532-5415.1991.tb01626.x
Carpenter CR, DesPain B, Keeling TN, Shah M, Rothenberger M. The six-item screener and AD8 for the detection of cognitive impairment in geriatric emergency department patients. Ann Emerg Med. 2011;57:653-661. doi:10.1016/j.annemergmed.2010.06.560
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381:752-762. doi:10.1016/S0140-6736(12)62167-9
Patel JN, Klein DS, Sreekumar S, Liporace FA, Yoon RS. Outcomes in multidisciplinary team-based approach in geriatric hip fracture care: a systematic review. J Am Acad Orthop Surg. 2020;28:128-133. doi:10.5435/JAAOS-D-18-00425
Amador LF, Loera JA. Preventing postoperative falls in the older adult. J Am Coll Surg. 2007;204:447-453. doi:10.1016/j.jamcollsurg.2006.12.010
Tembo MC, Holloway-Kew KL, Mohebbi M, et al. The association between a fracture risk tool and frailty: Geelong Osteoporosis Study. BMC Geriatr. 2020;20:196. doi:10.1186/s12877-020-01595-8
Demiris¸ B, Basat S, Kurt F, Aksakal B, Basat O. Evaluation of the relationship between frailty and fracture risk using Fracture Risk Assessment Tool in patients 65 years and over. South Clin Istanb Eurasia. 2023;34:42-48. doi:10.14744/scie.2022.66564
Partridge JSL, Harari D, Dhesi JK. Frailty in the older surgical patient: a review. Age Ageing. 2012;41:142-147. doi:10.1093/ageing/afr182
Mamtora PH, Fortier MA, Barnett SR, Schmid LN, Kain ZN. Peri-operative management of frailty in the orthopedic patient. J Orthop. 2020;22:304-307. doi:10.1016/j.jor.2020.05.024
Leven DM, Lee NJ, Kim JS, et al. Frailty is predictive of adverse postoperative events in patients undergoing lumbar fusion. Global Spine J. 2017;7:529-535. doi:10.1177/2192568217700099
Pritchard JM, Kennedy CC, Karampatos S, et al. Measuring frailty in clinical practice: a comparison of physical frailty assessment methods in a geriatric out-patient clinic. BMC Geriatr. 2017;17:264. doi:10.1186/s12877-017-0623-0
Kumar A, Dhar M, Agarwal M, Mukherjee A, Saxena V. Predictors of frailty in the elderly population: a cross-sectional study at a tertiary care center. Cureus. 2022;14:e30557. doi:10.7759/cureus.30557
Scarano KA, Philp FH, Westrick ER, Altman GT, Altman DT. Evaluating postoperative complications and outcomes of orthopedic fracture repair in nonagenarian patients. Geriatr Orthop Surg Rehabil. 2018;9:2151459318758106. doi:10.1177/2151459318758106
Liang Z, Rong K, Gu W, et al. Surgical site infection following elective orthopaedic surgeries in geriatric patients: incidence and associated risk factors. Int Wound J. 2019;16:773-780. doi:10.1111/iwj.13096
Ren M, Liang W, Wu Z, Zhao H, Wang J. Risk factors of surgical site infection in geriatric orthopedic surgery: a retrospective multicenter cohort study. Geriatr Gerontol Int. 2019;19:213-217. doi:10.1111/ggi.13590
Kaye KS, Schmader KE, Sawyer R. Surgical site infection in the elderly population. Clin Infect Dis. 2004;39:1835-1841. doi:10.1086/425744
Bruce AJ, Ritchie CW, Blizard R, Lai R, Raven P. The incidence of delirium associated with orthopedic surgery: a meta-analytic review. Int Psychogeriatr. 2007;19:197-214. doi:10.1017/S104161020600425X
Williams-Russo P, Urquhart BL, Sharrock NE, Charlson ME. Post-operative delirium: predictors and prognosis in elderly orthopedic patients. J Am Geriatr Soc. 1992;40:759-767. doi:10.1111/j.1532-5415.1992.tb01846.x
Wang J, Li Z, Yu Y, Li B, Shao G, Wang Q. Risk factors contributing to postoperative delirium in geriatric patients postorthopedic surgery. Asia Pac Psychiatry. 2015;7:375-382. doi:10.1111/appy.12193
Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126:338S-400S. doi:10.1378/chest.126.3_suppl.338S
Kahn SR, Shivakumar S. What’s new in VTE risk and prevention in orthopedic surgery. Res Pract Thromb Haemost. 2020;4:366-376. doi:10.1002/rth2.12323
Uzel K, Azboy I·, Parvizi J. Venous thromboembolism in orthopedic surgery: global guidelines. Acta Orthop Traumatol Turc. 2023;57:192-203. doi:10.5152/j.aott.2023.23074
Peck M, Holthaus A, Kingsbury K, Salsberry MG, Duggirala V. Mobility in acute care for geriatric patients with orthopedic conditions: a review of recent literature. Curr Geri Rep. 2020;9:300-310. doi:10.1007/s13670-020-00347-1
Leme LEG, Sitta MC, Toledo M, Henriques SS. Orthopedic surgery among the elderly: clinical characteristics. Rev Bras Ortop. 2015;46:238-246. doi:10.1016/S2255-4971(15)30189-0
Malcolm TL, Knezevic NN, Zouki CC, Tharian AR. Pulmonary complications after hip and knee arthroplasty in the United States, 2004-2014. Anesth Analg. 2020;130:917-924. doi:10.1213/ANE.0000000000004265
Kamal T, Conway RM, Littlejohn I, Ricketts D. The role of a multidisciplinary pre-assessment clinic in reducing mortality after complex orthopaedic surgery. Ann R Coll Surg Engl. 2011;93:149-151. doi:10.1308/003588411X561026
Davis MJ, Luu BC, Raj S, Abu-Ghname A, Buchanan EP. Multidisciplinary care in surgery: Are team-based interventions cost-effective? Surgeon. 2021;19:49-60. doi:10.1016/j.surge.2020.02.005
Frassanito L, Vergari A, Nestorini R, et al. Enhanced recovery after surgery (ERAS) in hip and knee replacement surgery: description of a multidisciplinary program to improve management of the patients undergoing major orthopedic surgery. Musculoskelet Surg. 2020;104:87-92. doi:10.1007/s12306-019-00603-4
Reddy RS, Alahmari KA, Alshahrani MS, et al. Exploring the impact of physiotherapy on health outcomes in older adults with chronic diseases: a cross-sectional analysis. Front Public Health. 2024;12:1415882. doi:10.3389/fpubh.2024.1415882
Watts NB. The Fracture Risk Assessment Tool (FRAX®): applications in clinical practice. J Womens Health (Larchmt). 2011;20:525-531. doi:10.1089/jwh.2010.2294
Gleason LJ, Benton EA, Alvarez-Nebreda ML, Weaver MJ, Harris MB, Javedan H. FRAIL questionnaire screening tool and short-term outcomes in geriatric fracture patients. J Am Med Dir Assoc. 2017;18:1082-1086. doi:10.1016/j.jamda.2017.07.005
Kojima G. Frailty defined by FRAIL scale as a predictor of mortality: a systematic review and meta-analysis. J Am Med Dir Assoc. 2018;19:480-483. doi:10.1016/j.jamda.2018.04.006
Culley DJ, Flaherty D, Fahey MC, et al. Poor performance on a preoperative cognitive screening test predicts postoperative complications in older orthopedic surgical patients. Anesthesiology. 2017;127:765-774. doi:10.1097/ALN.0000000000001859
Kong C, Zhang Y, Wang C, et al. Comprehensive geriatric assessment for older orthopedic patients and analysis of risk factors for postoperative complications. BMC Geriatr. 2022;22:644. doi:10.1186/s12877-022-03328-5
Williams DGA, Wischmeyer PE. Perioperative nutrition care of orthopedic surgery patient. Tech Orthop. 2020;35:15-18. doi:10.1097/BTO.0000000000000412
Koren-Hakim T, Weiss A, Hershkovitz A, et al. Comparing the adequacy of the MNA-SF, NRS-2002 and MUST nutritional tools in assessing malnutrition in hip fracture operated elderly patients. Clin Nutr. 2016;35:1053-1058. doi:10.1016/j.clnu.2015.07.014
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population