User login
Welcome to Current Psychiatry, a leading source of information, online and in print, for practitioners of psychiatry and its related subspecialties, including addiction psychiatry, child and adolescent psychiatry, and geriatric psychiatry. This Web site contains evidence-based reviews of the prevention, diagnosis, and treatment of mental illness and psychological disorders; case reports; updates on psychopharmacology; news about the specialty of psychiatry; pearls for practice; and other topics of interest and use to this audience.
Dear Drupal User: You're seeing this because you're logged in to Drupal, and not redirected to MDedge.com/psychiatry.
Depression
adolescent depression
adolescent major depressive disorder
adolescent schizophrenia
adolescent with major depressive disorder
animals
autism
baby
brexpiprazole
child
child bipolar
child depression
child schizophrenia
children with bipolar disorder
children with depression
children with major depressive disorder
compulsive behaviors
cure
elderly bipolar
elderly depression
elderly major depressive disorder
elderly schizophrenia
elderly with dementia
first break
first episode
gambling
gaming
geriatric depression
geriatric major depressive disorder
geriatric schizophrenia
infant
kid
major depressive disorder
major depressive disorder in adolescents
major depressive disorder in children
parenting
pediatric
pediatric bipolar
pediatric depression
pediatric major depressive disorder
pediatric schizophrenia
pregnancy
pregnant
rexulti
skin care
teen
wine
section[contains(@class, 'nav-hidden')]
footer[@id='footer']
div[contains(@class, 'pane-pub-article-current-psychiatry')]
div[contains(@class, 'pane-pub-home-current-psychiatry')]
div[contains(@class, 'pane-pub-topic-current-psychiatry')]
div[contains(@class, 'panel-panel-inner')]
div[contains(@class, 'pane-node-field-article-topics')]
section[contains(@class, 'footer-nav-section-wrapper')]
Perinatal psychiatry: 5 key principles
Perinatal mood and anxiety disorders are the most common complication of pregnancy and childbirth.1 Mental health concerns are a leading cause of maternal mortality in the United States, which has rising maternal mortality rates and glaring racial and socioeconomic disparities.2 Inconsistent perinatal psychiatry training likely contributes to perceived discomfort of patients who are pregnant.3 This is why it is critical for all psychiatrists to understand the principles of perinatal psychiatry. Here is a brief description of 5 key principles.
1. Discuss preconception planning
Reproductive life planning should occur with all patients who are capable of becoming pregnant. This planning should include not just a risks/benefits analysis and anticipatory planning regarding medications but also a discussion of prior perinatal symptoms, pregnancy intentions and contraception (especially in light of increasingly limited access to abortion), and the bidirectional nature of pregnancy and mental health conditions.
The acronym PATH provides a framework for these conversations:
- Pregnancy Attitudes: “Do you think you might like to have (more) children at some point?”
- Timing: “If considering future parenthood, when do you think that might be?”
- How important is prevention: “How important is it to you to prevent pregnancy (until then)?”4
2. Focus on perinatal mental health
Discussion often centers on medication risks to the fetus at the expense of considering risks of under- or nontreatment for both members of the dyad. Undertreating perinatal mental health conditions results in dual exposures (medication and illness), and untreated illness is associated with negative effects on obstetric and neonatal outcomes and the well-being of the parent and offspring.1
3. Resist experimentation
It is common for clinicians to reflexively switch patients who are pregnant from an effective medication to one viewed as the “safest” or “best” because it has more data. This exposes the fetus to 2 medications and the dyad to potential symptoms of the illness. Decisions about medication changes should instead be made on an individual basis considering the risks and benefits of all exposures as well as the patient’s current symptoms, previous treatment, and family history.
4. Collaborate and communicate
Despite effective interventions, many perinatal mental health conditions go untreated.1 Normalize perinatal mental health symptoms with patients to reduce stigma and barriers to disclosure, and respect their decisions regarding perinatal medication use. Proper communication with the obstetric team ensures appropriate perinatal mental health screening and fetal monitoring (eg, possible fetal growth ultrasounds for a patient taking prazosin, or assessing for neonatal adaptation syndrome if there is selective serotonin reuptake inhibitor exposure in utero).
5. Recognize your limitations
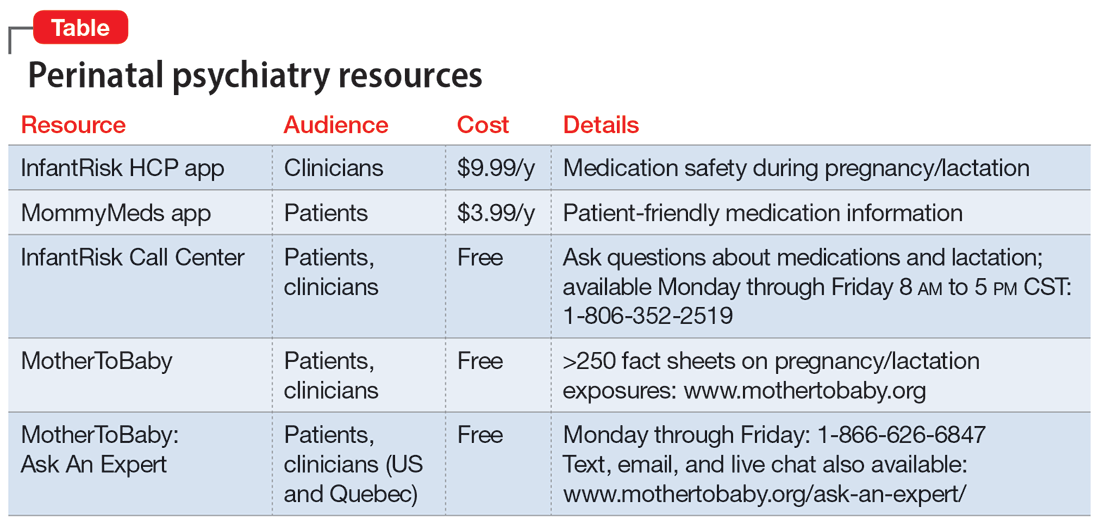
Our understanding of psychotropics’ teratogenicity is constantly evolving, and we must recognize when we don’t know something. In addition to medication databases such as Reprotox (https://reprotox.org/) and LactMed (https://www.ncbi.nlm.nih.gov/books/NBK501922/), several perinatal psychiatry resources are available for both patients and clinicians (Table). Additionally, Postpartum Support International maintains a National Perinatal Consult Line (1-877-499-4773) as well as a list of state perinatal psychiatry access lines (https://www.postpartum.net/professionals/state-perinatal-psychiatry-access-lines/) for clinicians. The Massachusetts General Hospital Center for Women’s Mental Health (https://womensmentalhealth.org) is also a helpful resource for clinicians.
1. Luca DL, Garlow N, Staatz C, et al. Societal costs of untreated perinatal mood and anxiety disorders in the United States. Mathematica Policy Research. April 29, 2019. Accessed July 13, 2023. https://www.mathematica.org/publications/societal-costs-of-untreated-perinatal-mood-and-anxiety-disorders-in-the-united-states
2. Singh GK. Trends and social inequalities in maternal mortality in the United States, 1969-2018. Int J MCH AIDS. 2021;10(1):29-42. doi:10.21106/ijma.444
3. Weinreb L, Byatt N, Moore Simas TA, et al. What happens to mental health treatment during pregnancy? Women’s experience with prescribing providers. Psychiatr Q. 2014;85(3):349-355. doi:10.1007/s11126-014-9293-7
4. Callegari LS, Aiken AR, Dehlendorf C, et al. Addressing potential pitfalls of reproductive life planning with patient-centered counseling. Am J Obstet Gynecol. 2017;216(2):129-134. doi:10.1016/j.ajog.2016.10.004
Perinatal mood and anxiety disorders are the most common complication of pregnancy and childbirth.1 Mental health concerns are a leading cause of maternal mortality in the United States, which has rising maternal mortality rates and glaring racial and socioeconomic disparities.2 Inconsistent perinatal psychiatry training likely contributes to perceived discomfort of patients who are pregnant.3 This is why it is critical for all psychiatrists to understand the principles of perinatal psychiatry. Here is a brief description of 5 key principles.
1. Discuss preconception planning
Reproductive life planning should occur with all patients who are capable of becoming pregnant. This planning should include not just a risks/benefits analysis and anticipatory planning regarding medications but also a discussion of prior perinatal symptoms, pregnancy intentions and contraception (especially in light of increasingly limited access to abortion), and the bidirectional nature of pregnancy and mental health conditions.
The acronym PATH provides a framework for these conversations:
- Pregnancy Attitudes: “Do you think you might like to have (more) children at some point?”
- Timing: “If considering future parenthood, when do you think that might be?”
- How important is prevention: “How important is it to you to prevent pregnancy (until then)?”4
2. Focus on perinatal mental health
Discussion often centers on medication risks to the fetus at the expense of considering risks of under- or nontreatment for both members of the dyad. Undertreating perinatal mental health conditions results in dual exposures (medication and illness), and untreated illness is associated with negative effects on obstetric and neonatal outcomes and the well-being of the parent and offspring.1
3. Resist experimentation
It is common for clinicians to reflexively switch patients who are pregnant from an effective medication to one viewed as the “safest” or “best” because it has more data. This exposes the fetus to 2 medications and the dyad to potential symptoms of the illness. Decisions about medication changes should instead be made on an individual basis considering the risks and benefits of all exposures as well as the patient’s current symptoms, previous treatment, and family history.
4. Collaborate and communicate
Despite effective interventions, many perinatal mental health conditions go untreated.1 Normalize perinatal mental health symptoms with patients to reduce stigma and barriers to disclosure, and respect their decisions regarding perinatal medication use. Proper communication with the obstetric team ensures appropriate perinatal mental health screening and fetal monitoring (eg, possible fetal growth ultrasounds for a patient taking prazosin, or assessing for neonatal adaptation syndrome if there is selective serotonin reuptake inhibitor exposure in utero).
5. Recognize your limitations
Our understanding of psychotropics’ teratogenicity is constantly evolving, and we must recognize when we don’t know something. In addition to medication databases such as Reprotox (https://reprotox.org/) and LactMed (https://www.ncbi.nlm.nih.gov/books/NBK501922/), several perinatal psychiatry resources are available for both patients and clinicians (Table). Additionally, Postpartum Support International maintains a National Perinatal Consult Line (1-877-499-4773) as well as a list of state perinatal psychiatry access lines (https://www.postpartum.net/professionals/state-perinatal-psychiatry-access-lines/) for clinicians. The Massachusetts General Hospital Center for Women’s Mental Health (https://womensmentalhealth.org) is also a helpful resource for clinicians.

Perinatal mood and anxiety disorders are the most common complication of pregnancy and childbirth.1 Mental health concerns are a leading cause of maternal mortality in the United States, which has rising maternal mortality rates and glaring racial and socioeconomic disparities.2 Inconsistent perinatal psychiatry training likely contributes to perceived discomfort of patients who are pregnant.3 This is why it is critical for all psychiatrists to understand the principles of perinatal psychiatry. Here is a brief description of 5 key principles.
1. Discuss preconception planning
Reproductive life planning should occur with all patients who are capable of becoming pregnant. This planning should include not just a risks/benefits analysis and anticipatory planning regarding medications but also a discussion of prior perinatal symptoms, pregnancy intentions and contraception (especially in light of increasingly limited access to abortion), and the bidirectional nature of pregnancy and mental health conditions.
The acronym PATH provides a framework for these conversations:
- Pregnancy Attitudes: “Do you think you might like to have (more) children at some point?”
- Timing: “If considering future parenthood, when do you think that might be?”
- How important is prevention: “How important is it to you to prevent pregnancy (until then)?”4
2. Focus on perinatal mental health
Discussion often centers on medication risks to the fetus at the expense of considering risks of under- or nontreatment for both members of the dyad. Undertreating perinatal mental health conditions results in dual exposures (medication and illness), and untreated illness is associated with negative effects on obstetric and neonatal outcomes and the well-being of the parent and offspring.1
3. Resist experimentation
It is common for clinicians to reflexively switch patients who are pregnant from an effective medication to one viewed as the “safest” or “best” because it has more data. This exposes the fetus to 2 medications and the dyad to potential symptoms of the illness. Decisions about medication changes should instead be made on an individual basis considering the risks and benefits of all exposures as well as the patient’s current symptoms, previous treatment, and family history.
4. Collaborate and communicate
Despite effective interventions, many perinatal mental health conditions go untreated.1 Normalize perinatal mental health symptoms with patients to reduce stigma and barriers to disclosure, and respect their decisions regarding perinatal medication use. Proper communication with the obstetric team ensures appropriate perinatal mental health screening and fetal monitoring (eg, possible fetal growth ultrasounds for a patient taking prazosin, or assessing for neonatal adaptation syndrome if there is selective serotonin reuptake inhibitor exposure in utero).
5. Recognize your limitations
Our understanding of psychotropics’ teratogenicity is constantly evolving, and we must recognize when we don’t know something. In addition to medication databases such as Reprotox (https://reprotox.org/) and LactMed (https://www.ncbi.nlm.nih.gov/books/NBK501922/), several perinatal psychiatry resources are available for both patients and clinicians (Table). Additionally, Postpartum Support International maintains a National Perinatal Consult Line (1-877-499-4773) as well as a list of state perinatal psychiatry access lines (https://www.postpartum.net/professionals/state-perinatal-psychiatry-access-lines/) for clinicians. The Massachusetts General Hospital Center for Women’s Mental Health (https://womensmentalhealth.org) is also a helpful resource for clinicians.

1. Luca DL, Garlow N, Staatz C, et al. Societal costs of untreated perinatal mood and anxiety disorders in the United States. Mathematica Policy Research. April 29, 2019. Accessed July 13, 2023. https://www.mathematica.org/publications/societal-costs-of-untreated-perinatal-mood-and-anxiety-disorders-in-the-united-states
2. Singh GK. Trends and social inequalities in maternal mortality in the United States, 1969-2018. Int J MCH AIDS. 2021;10(1):29-42. doi:10.21106/ijma.444
3. Weinreb L, Byatt N, Moore Simas TA, et al. What happens to mental health treatment during pregnancy? Women’s experience with prescribing providers. Psychiatr Q. 2014;85(3):349-355. doi:10.1007/s11126-014-9293-7
4. Callegari LS, Aiken AR, Dehlendorf C, et al. Addressing potential pitfalls of reproductive life planning with patient-centered counseling. Am J Obstet Gynecol. 2017;216(2):129-134. doi:10.1016/j.ajog.2016.10.004
1. Luca DL, Garlow N, Staatz C, et al. Societal costs of untreated perinatal mood and anxiety disorders in the United States. Mathematica Policy Research. April 29, 2019. Accessed July 13, 2023. https://www.mathematica.org/publications/societal-costs-of-untreated-perinatal-mood-and-anxiety-disorders-in-the-united-states
2. Singh GK. Trends and social inequalities in maternal mortality in the United States, 1969-2018. Int J MCH AIDS. 2021;10(1):29-42. doi:10.21106/ijma.444
3. Weinreb L, Byatt N, Moore Simas TA, et al. What happens to mental health treatment during pregnancy? Women’s experience with prescribing providers. Psychiatr Q. 2014;85(3):349-355. doi:10.1007/s11126-014-9293-7
4. Callegari LS, Aiken AR, Dehlendorf C, et al. Addressing potential pitfalls of reproductive life planning with patient-centered counseling. Am J Obstet Gynecol. 2017;216(2):129-134. doi:10.1016/j.ajog.2016.10.004
Diagnosing borderline personality disorder: Avoid these pitfalls
Borderline personality disorder (BPD) is associated with impaired psychosocial functioning, reduced quality of life, increased use of health care services, and excess mortality.1 Unfortunately, this disorder is often underrecognized and underdiagnosed, and patients with BPD may not receive an accurate diagnosis for years after first seeking treatment.1 Problems in diagnosing BPD include:
Stigma. Some patients may view the term “borderline” as stigmatizing, as if we are calling these patients borderline human beings. One of the symptoms of BPD is a “markedly and persistently unstable self-image.”2 Such patients do not need a stigmatizing label to worsen their self-image.
Terminology. The word borderline may also imply relatively mild psychiatric symptoms. However, “borderline personality disorder” does not refer to a mild personality disorder. DSM-5 describes potential BPD symptoms as “intense,” “marked,” or “severe,” and 1 of the symptoms is suicidal behavior.2
Symptoms. To meet the criteria for a BPD diagnosis, a patient must exhibit ≥5 of 9 severe symptoms2:
- frantic efforts to avoid abandonment
- unstable and intense interpersonal relationships
- unstable self-image
- impulsivity in ≥2 areas that are potentially self-damaging
- suicidal behavior
- affective instability
- chronic feelings of emptiness
- inappropriate anger
- transient paranoid ideation or dissociative symptoms.
Asking about all 9 of these criteria and their severity is not part of a routine psychiatric evaluation. A patient might not volunteer any of this information because they are concerned about potential stigma. Additionally, perhaps most of the general population has had a “BPD-like” symptom at least once during their lives. This symptom might not have been severe enough to qualify as a true BPD symptom. Clinicians might have difficulty discerning BPD-like symptoms from true BPD symptoms.
Comorbidities. Many patients with BPD also have a comorbid mood disorder or substance use disorder.1,3 Clinicians might focus on a comorbid diagnosis and not recognize BPD.
Stress. BPD symptoms may become more severe when the patient faces a stressful situation. The BPD symptoms might seem more severe than the stress would warrant.2 However, clinicians might blame the BPD symptoms solely on stress and not acknowledge the underlying BPD diagnosis.
Awareness of these factors can help clinicians keep BPD in the differential diagnosis when conducting a psychiatric evaluation, thus reducing the chances of overlooking this serious disorder.
1. Zimmerman M. Improving the recognition of borderline personality disorder. Current Psychiatry. 2017;16(10):13-19.
2. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Association; 2013:663-666.
3. Grant BF, Chou SP, Goldstein RB, et al. Prevalence, correlates, disability, and comorbidity of DSM-IV borderline personality disorder: results from the Wave 2 National Epidemiologic Survey on Alcohol and Related Conditions. J Clin Psychiatry. 2008:69(4)533-545.
Borderline personality disorder (BPD) is associated with impaired psychosocial functioning, reduced quality of life, increased use of health care services, and excess mortality.1 Unfortunately, this disorder is often underrecognized and underdiagnosed, and patients with BPD may not receive an accurate diagnosis for years after first seeking treatment.1 Problems in diagnosing BPD include:
Stigma. Some patients may view the term “borderline” as stigmatizing, as if we are calling these patients borderline human beings. One of the symptoms of BPD is a “markedly and persistently unstable self-image.”2 Such patients do not need a stigmatizing label to worsen their self-image.
Terminology. The word borderline may also imply relatively mild psychiatric symptoms. However, “borderline personality disorder” does not refer to a mild personality disorder. DSM-5 describes potential BPD symptoms as “intense,” “marked,” or “severe,” and 1 of the symptoms is suicidal behavior.2
Symptoms. To meet the criteria for a BPD diagnosis, a patient must exhibit ≥5 of 9 severe symptoms2:
- frantic efforts to avoid abandonment
- unstable and intense interpersonal relationships
- unstable self-image
- impulsivity in ≥2 areas that are potentially self-damaging
- suicidal behavior
- affective instability
- chronic feelings of emptiness
- inappropriate anger
- transient paranoid ideation or dissociative symptoms.
Asking about all 9 of these criteria and their severity is not part of a routine psychiatric evaluation. A patient might not volunteer any of this information because they are concerned about potential stigma. Additionally, perhaps most of the general population has had a “BPD-like” symptom at least once during their lives. This symptom might not have been severe enough to qualify as a true BPD symptom. Clinicians might have difficulty discerning BPD-like symptoms from true BPD symptoms.
Comorbidities. Many patients with BPD also have a comorbid mood disorder or substance use disorder.1,3 Clinicians might focus on a comorbid diagnosis and not recognize BPD.
Stress. BPD symptoms may become more severe when the patient faces a stressful situation. The BPD symptoms might seem more severe than the stress would warrant.2 However, clinicians might blame the BPD symptoms solely on stress and not acknowledge the underlying BPD diagnosis.
Awareness of these factors can help clinicians keep BPD in the differential diagnosis when conducting a psychiatric evaluation, thus reducing the chances of overlooking this serious disorder.
Borderline personality disorder (BPD) is associated with impaired psychosocial functioning, reduced quality of life, increased use of health care services, and excess mortality.1 Unfortunately, this disorder is often underrecognized and underdiagnosed, and patients with BPD may not receive an accurate diagnosis for years after first seeking treatment.1 Problems in diagnosing BPD include:
Stigma. Some patients may view the term “borderline” as stigmatizing, as if we are calling these patients borderline human beings. One of the symptoms of BPD is a “markedly and persistently unstable self-image.”2 Such patients do not need a stigmatizing label to worsen their self-image.
Terminology. The word borderline may also imply relatively mild psychiatric symptoms. However, “borderline personality disorder” does not refer to a mild personality disorder. DSM-5 describes potential BPD symptoms as “intense,” “marked,” or “severe,” and 1 of the symptoms is suicidal behavior.2
Symptoms. To meet the criteria for a BPD diagnosis, a patient must exhibit ≥5 of 9 severe symptoms2:
- frantic efforts to avoid abandonment
- unstable and intense interpersonal relationships
- unstable self-image
- impulsivity in ≥2 areas that are potentially self-damaging
- suicidal behavior
- affective instability
- chronic feelings of emptiness
- inappropriate anger
- transient paranoid ideation or dissociative symptoms.
Asking about all 9 of these criteria and their severity is not part of a routine psychiatric evaluation. A patient might not volunteer any of this information because they are concerned about potential stigma. Additionally, perhaps most of the general population has had a “BPD-like” symptom at least once during their lives. This symptom might not have been severe enough to qualify as a true BPD symptom. Clinicians might have difficulty discerning BPD-like symptoms from true BPD symptoms.
Comorbidities. Many patients with BPD also have a comorbid mood disorder or substance use disorder.1,3 Clinicians might focus on a comorbid diagnosis and not recognize BPD.
Stress. BPD symptoms may become more severe when the patient faces a stressful situation. The BPD symptoms might seem more severe than the stress would warrant.2 However, clinicians might blame the BPD symptoms solely on stress and not acknowledge the underlying BPD diagnosis.
Awareness of these factors can help clinicians keep BPD in the differential diagnosis when conducting a psychiatric evaluation, thus reducing the chances of overlooking this serious disorder.
1. Zimmerman M. Improving the recognition of borderline personality disorder. Current Psychiatry. 2017;16(10):13-19.
2. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Association; 2013:663-666.
3. Grant BF, Chou SP, Goldstein RB, et al. Prevalence, correlates, disability, and comorbidity of DSM-IV borderline personality disorder: results from the Wave 2 National Epidemiologic Survey on Alcohol and Related Conditions. J Clin Psychiatry. 2008:69(4)533-545.
1. Zimmerman M. Improving the recognition of borderline personality disorder. Current Psychiatry. 2017;16(10):13-19.
2. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Association; 2013:663-666.
3. Grant BF, Chou SP, Goldstein RB, et al. Prevalence, correlates, disability, and comorbidity of DSM-IV borderline personality disorder: results from the Wave 2 National Epidemiologic Survey on Alcohol and Related Conditions. J Clin Psychiatry. 2008:69(4)533-545.
Extended-release injectable naltrexone for opioid use disorder
We appreciate the important review by Gluck et al (“Managing patients with comorbid opioid and alcohol use disorders,”
XR-NTX should be considered an equal OUD treatment alternative to buprenorphine-naloxone, especially for patients who prefer an opioid-free option.1,2 It has the added advantage of being FDA-approved for both AUD and OUD.
One obstacle to the success of XR-NTX is the induction period. The National Institute on Drug Abuse Clinical Trials Network X:BOT trial found that once the induction hurdle was surmounted, XR-NTX and buprenorphine were equally effective in a population of approximately 80% heroin users and two-thirds injection drug users.2 Patient variables that predict successful induction include young age, baseline preference for XR-NTX, fewer drug complications, and fewer family/social complications.3 If the length of the induction (usually 7 to 10 days) is a deterrent, a study supported the feasibility of a 5-day outpatient XR-NTX induction.4 Further research is needed to improve successful induction for XR-NTX.
Ashmeer Ogbuchi, MD
Karen Drexler, MD
Atlanta, Georgia
References
1. Tanum L, Solli KK, Latif Z, et al. Effectiveness of injectable extended-release naltrexone vs daily buprenorphine-naloxone for opioid dependence. JAMA Psychiatry. 2017;74(12):1197-1205. doi:10.1001/ jamapsychiatry.2017.3206
2. Lee JD, Nunes EV Jr, Novo P, et al. Comparative effectiveness of extended-release naltrexone versus buprenorphine-naloxone for opioid relapse prevention (X:BOT): a multicentre, open-label, randomised controlled trial. Lancet. 2018;391(10118):309-318. doi:10.1016/s0140-6736(17)32812-x
3. Murphy SM, Jeng PJ, McCollister KE, et al. Cost‐effectiveness implications of increasing the efficiency of the extended‐release naltrexone induction process for the treatment of opioid use disorder: a secondary analysis. Addiction. 2021;116(12)3444-3453. doi:10.1111/add.15531
4. Sibai M, Mishlen K, Nunes EV, et al. A week-long outpatient induction onto XR-naltrexone in patients with opioid use disorder. Am J Drug Alcohol Abuse. 2020;46(3):289-296. doi:10.1080/00952990.2019.1700265
Continue to: The authors respond
The authors respond
We appreciate Drs. Ogbuchi and Drexler for their thoughtful attention to our review. They proposed amending our original algorithm, recommending that XR-NTX be considered as another first-line option for patients with OUD. We agree with this suggestion, particularly for inpatients. However, we have some reservations about applying this suggestion to outpatient treatment. Though research evidence from Lee et al1 indicates that once initiation is completed, both medications are equally safe and effective, the initial attrition rate in the XR-NTX group was much higher (28% vs 6%, P < .0001), which suggests lower acceptability/tolerability compared with buprenorphine. Notably, the initiation of both medications in Lee et al1 was done in an inpatient setting. Moreover, although some medications are endorsed as “first-line,” the actual utilization rate is often influenced by many factors, including the ease of treatment initiation. Wakeman et al2 found the most common treatment modality received by patients with OUD was nonintensive behavioral health (59.5%), followed by inpatient withdrawal management and residential treatment (15.2%). Among all patients in the Wakeman study,2 only 12.5% received buprenorphine or methadone, and 2.4% received naltrexone.
Data from our clinic corroborate this trend. Currently, in our clinic approximately 300 patients with OUD are receiving medications, including approximately 250 on buprenorphine (including 5 to 10 on the long-acting injectable formulation), 50 on methadone, and only 1 or 2 on XR-NTX. Though this disparity may reflect bias in our clinicians’ prescribing practices, in the past few years we have had many unsuccessful attempts at initiating XR-NTX. To our disappointment, a theoretically excellent medication has not translated clinically. The recent surge in fentanyl use further complicates XR-NTX initiation for OUD, because the length of induction may be longer.
In conclusion, we agree that XR-NTX is a potential treatment option for patients with OUD, but clinicians should be cognizant of the potential barriers; inform patients of the advantages, expectations, and challenges; and respect patients’ informed decisions.
Rachel Gluck, MD
Karen Hochman, MD
Yi-lang Tang, MD, PhD
Atlanta, Georgia
References
1. Lee JD, Nunes EV Jr, Novo P, et al. Comparative effectiveness of extended-release naltrexone versus buprenorphine-naloxone for opioid relapse prevention (X:BOT): a multicentre, open-label, randomised controlled trial. Lancet. 2018;391(10118):309-318. doi:10.1016/s0140-6736(17)32812-x
2. Wakeman SE, Larochelle MR, Ameli O, et al. Comparative effectiveness of different treatment pathways for opioid use disorder. JAMA Netw Open. 2020;3(2):e1920622. doi:10.1001/jamanetworkopen.2019.20622
We appreciate the important review by Gluck et al (“Managing patients with comorbid opioid and alcohol use disorders,”
XR-NTX should be considered an equal OUD treatment alternative to buprenorphine-naloxone, especially for patients who prefer an opioid-free option.1,2 It has the added advantage of being FDA-approved for both AUD and OUD.
One obstacle to the success of XR-NTX is the induction period. The National Institute on Drug Abuse Clinical Trials Network X:BOT trial found that once the induction hurdle was surmounted, XR-NTX and buprenorphine were equally effective in a population of approximately 80% heroin users and two-thirds injection drug users.2 Patient variables that predict successful induction include young age, baseline preference for XR-NTX, fewer drug complications, and fewer family/social complications.3 If the length of the induction (usually 7 to 10 days) is a deterrent, a study supported the feasibility of a 5-day outpatient XR-NTX induction.4 Further research is needed to improve successful induction for XR-NTX.
Ashmeer Ogbuchi, MD
Karen Drexler, MD
Atlanta, Georgia
References
1. Tanum L, Solli KK, Latif Z, et al. Effectiveness of injectable extended-release naltrexone vs daily buprenorphine-naloxone for opioid dependence. JAMA Psychiatry. 2017;74(12):1197-1205. doi:10.1001/ jamapsychiatry.2017.3206
2. Lee JD, Nunes EV Jr, Novo P, et al. Comparative effectiveness of extended-release naltrexone versus buprenorphine-naloxone for opioid relapse prevention (X:BOT): a multicentre, open-label, randomised controlled trial. Lancet. 2018;391(10118):309-318. doi:10.1016/s0140-6736(17)32812-x
3. Murphy SM, Jeng PJ, McCollister KE, et al. Cost‐effectiveness implications of increasing the efficiency of the extended‐release naltrexone induction process for the treatment of opioid use disorder: a secondary analysis. Addiction. 2021;116(12)3444-3453. doi:10.1111/add.15531
4. Sibai M, Mishlen K, Nunes EV, et al. A week-long outpatient induction onto XR-naltrexone in patients with opioid use disorder. Am J Drug Alcohol Abuse. 2020;46(3):289-296. doi:10.1080/00952990.2019.1700265
Continue to: The authors respond
The authors respond
We appreciate Drs. Ogbuchi and Drexler for their thoughtful attention to our review. They proposed amending our original algorithm, recommending that XR-NTX be considered as another first-line option for patients with OUD. We agree with this suggestion, particularly for inpatients. However, we have some reservations about applying this suggestion to outpatient treatment. Though research evidence from Lee et al1 indicates that once initiation is completed, both medications are equally safe and effective, the initial attrition rate in the XR-NTX group was much higher (28% vs 6%, P < .0001), which suggests lower acceptability/tolerability compared with buprenorphine. Notably, the initiation of both medications in Lee et al1 was done in an inpatient setting. Moreover, although some medications are endorsed as “first-line,” the actual utilization rate is often influenced by many factors, including the ease of treatment initiation. Wakeman et al2 found the most common treatment modality received by patients with OUD was nonintensive behavioral health (59.5%), followed by inpatient withdrawal management and residential treatment (15.2%). Among all patients in the Wakeman study,2 only 12.5% received buprenorphine or methadone, and 2.4% received naltrexone.
Data from our clinic corroborate this trend. Currently, in our clinic approximately 300 patients with OUD are receiving medications, including approximately 250 on buprenorphine (including 5 to 10 on the long-acting injectable formulation), 50 on methadone, and only 1 or 2 on XR-NTX. Though this disparity may reflect bias in our clinicians’ prescribing practices, in the past few years we have had many unsuccessful attempts at initiating XR-NTX. To our disappointment, a theoretically excellent medication has not translated clinically. The recent surge in fentanyl use further complicates XR-NTX initiation for OUD, because the length of induction may be longer.
In conclusion, we agree that XR-NTX is a potential treatment option for patients with OUD, but clinicians should be cognizant of the potential barriers; inform patients of the advantages, expectations, and challenges; and respect patients’ informed decisions.
Rachel Gluck, MD
Karen Hochman, MD
Yi-lang Tang, MD, PhD
Atlanta, Georgia
References
1. Lee JD, Nunes EV Jr, Novo P, et al. Comparative effectiveness of extended-release naltrexone versus buprenorphine-naloxone for opioid relapse prevention (X:BOT): a multicentre, open-label, randomised controlled trial. Lancet. 2018;391(10118):309-318. doi:10.1016/s0140-6736(17)32812-x
2. Wakeman SE, Larochelle MR, Ameli O, et al. Comparative effectiveness of different treatment pathways for opioid use disorder. JAMA Netw Open. 2020;3(2):e1920622. doi:10.1001/jamanetworkopen.2019.20622
We appreciate the important review by Gluck et al (“Managing patients with comorbid opioid and alcohol use disorders,”
XR-NTX should be considered an equal OUD treatment alternative to buprenorphine-naloxone, especially for patients who prefer an opioid-free option.1,2 It has the added advantage of being FDA-approved for both AUD and OUD.
One obstacle to the success of XR-NTX is the induction period. The National Institute on Drug Abuse Clinical Trials Network X:BOT trial found that once the induction hurdle was surmounted, XR-NTX and buprenorphine were equally effective in a population of approximately 80% heroin users and two-thirds injection drug users.2 Patient variables that predict successful induction include young age, baseline preference for XR-NTX, fewer drug complications, and fewer family/social complications.3 If the length of the induction (usually 7 to 10 days) is a deterrent, a study supported the feasibility of a 5-day outpatient XR-NTX induction.4 Further research is needed to improve successful induction for XR-NTX.
Ashmeer Ogbuchi, MD
Karen Drexler, MD
Atlanta, Georgia
References
1. Tanum L, Solli KK, Latif Z, et al. Effectiveness of injectable extended-release naltrexone vs daily buprenorphine-naloxone for opioid dependence. JAMA Psychiatry. 2017;74(12):1197-1205. doi:10.1001/ jamapsychiatry.2017.3206
2. Lee JD, Nunes EV Jr, Novo P, et al. Comparative effectiveness of extended-release naltrexone versus buprenorphine-naloxone for opioid relapse prevention (X:BOT): a multicentre, open-label, randomised controlled trial. Lancet. 2018;391(10118):309-318. doi:10.1016/s0140-6736(17)32812-x
3. Murphy SM, Jeng PJ, McCollister KE, et al. Cost‐effectiveness implications of increasing the efficiency of the extended‐release naltrexone induction process for the treatment of opioid use disorder: a secondary analysis. Addiction. 2021;116(12)3444-3453. doi:10.1111/add.15531
4. Sibai M, Mishlen K, Nunes EV, et al. A week-long outpatient induction onto XR-naltrexone in patients with opioid use disorder. Am J Drug Alcohol Abuse. 2020;46(3):289-296. doi:10.1080/00952990.2019.1700265
Continue to: The authors respond
The authors respond
We appreciate Drs. Ogbuchi and Drexler for their thoughtful attention to our review. They proposed amending our original algorithm, recommending that XR-NTX be considered as another first-line option for patients with OUD. We agree with this suggestion, particularly for inpatients. However, we have some reservations about applying this suggestion to outpatient treatment. Though research evidence from Lee et al1 indicates that once initiation is completed, both medications are equally safe and effective, the initial attrition rate in the XR-NTX group was much higher (28% vs 6%, P < .0001), which suggests lower acceptability/tolerability compared with buprenorphine. Notably, the initiation of both medications in Lee et al1 was done in an inpatient setting. Moreover, although some medications are endorsed as “first-line,” the actual utilization rate is often influenced by many factors, including the ease of treatment initiation. Wakeman et al2 found the most common treatment modality received by patients with OUD was nonintensive behavioral health (59.5%), followed by inpatient withdrawal management and residential treatment (15.2%). Among all patients in the Wakeman study,2 only 12.5% received buprenorphine or methadone, and 2.4% received naltrexone.
Data from our clinic corroborate this trend. Currently, in our clinic approximately 300 patients with OUD are receiving medications, including approximately 250 on buprenorphine (including 5 to 10 on the long-acting injectable formulation), 50 on methadone, and only 1 or 2 on XR-NTX. Though this disparity may reflect bias in our clinicians’ prescribing practices, in the past few years we have had many unsuccessful attempts at initiating XR-NTX. To our disappointment, a theoretically excellent medication has not translated clinically. The recent surge in fentanyl use further complicates XR-NTX initiation for OUD, because the length of induction may be longer.
In conclusion, we agree that XR-NTX is a potential treatment option for patients with OUD, but clinicians should be cognizant of the potential barriers; inform patients of the advantages, expectations, and challenges; and respect patients’ informed decisions.
Rachel Gluck, MD
Karen Hochman, MD
Yi-lang Tang, MD, PhD
Atlanta, Georgia
References
1. Lee JD, Nunes EV Jr, Novo P, et al. Comparative effectiveness of extended-release naltrexone versus buprenorphine-naloxone for opioid relapse prevention (X:BOT): a multicentre, open-label, randomised controlled trial. Lancet. 2018;391(10118):309-318. doi:10.1016/s0140-6736(17)32812-x
2. Wakeman SE, Larochelle MR, Ameli O, et al. Comparative effectiveness of different treatment pathways for opioid use disorder. JAMA Netw Open. 2020;3(2):e1920622. doi:10.1001/jamanetworkopen.2019.20622
Risk Evaluation and Mitigation Strategy programs: How they can be improved
A Risk Evaluation and Mitigation Strategy (REMS) is a drug safety program the FDA can require for certain medications with serious safety concerns to help ensure the benefits of the medication outweigh its risks (Box1). The FDA may require medication guides, patient package inserts, communication plans for health care professionals, and/or certain packaging and safe disposal technologies for medications that pose a serious risk of abuse or overdose. The FDA may also require elements to assure safe use and/or an implementation system be included in the REMS. Pharmaceutical manufacturers then develop a proposed REMS for FDA review.2 If the FDA approves the proposed REMS, the manufacturer is responsible for implementing the REMS requirements.
Box
There are many myths and misconceptions surrounding psychiatry, the branch of medicine that deals with the diagnosis, treatment, and prevention of mental illness. Some of the most common myths include:
The FDA provides this description of a Risk Evaluation and Mitigation Strategy (REMS):
“A [REMS] is a drug safety program that the U.S. Food and Drug Administration (FDA) can require for certain medications with serious safety concerns to help ensure the benefits of the medication outweigh its risks. REMS are designed to reinforce medication use behaviors and actions that support the safe use of that medication. While all medications have labeling that informs health care stakeholders about medication risks, only a few medications require a REMS. REMS are not designed to mitigate all the adverse events of a medication, these are communicated to health care providers in the medication’s prescribing information. Rather, REMS focus on preventing, monitoring and/or managing a specific serious risk by informing, educating and/or reinforcing actions to reduce the frequency and/or severity of the event.”1
The REMS program for clozapine3 has been the subject of much discussion in the psychiatric community. The adverse impact of the 2015 update to the clozapine REMS program was emphasized at meetings of both the American Psychiatric Association and the College of Psychiatric and Neurologic Pharmacists. A white paper published by the National Association of State Mental Health Program Directors shortly after the 2015 update concluded, “clozapine is underused due to a variety of barriers related to the drug and its properties, the health care system, regulatory requirements, and reimbursement issues.”4 After an update to the clozapine REMS program in 2021, the FDA temporarily suspended enforcement of certain requirements due to concerns from health care professionals about patient access to the medication because of problems with implementing the clozapine REMS program.5,6 In November 2022, the FDA issued a second announcement of enforcement discretion related to additional requirements of the REMS program.5 The FDA had previously announced a decision to not take action regarding adherence to REMS requirements for certain laboratory tests in March 2020, during the COVID-19 pandemic.7
REMS programs for other psychiatric medications may also present challenges. The REMS programs for esketamine8 and olanzapine for extended-release (ER) injectable suspension9 include certain risks that require postadministration monitoring. Some facilities have had to dedicate additional space and clinician time to ensure REMS requirements are met.
To further understand health care professionals’ perspectives regarding the value and burden of these REMS programs, a collaborative effort of the University of Maryland (College Park and Baltimore campuses) Center of Excellence in Regulatory Science and Innovation with the FDA was undertaken. The REMS for clozapine, olanzapine for ER injectable suspension, and esketamine were examined to develop recommendations for improving patient access while ensuring safe medication use and limiting the impact on health care professionals.
Assessing the REMS programs
Focus groups were held with health care professionals nominated by professional organizations to gather their perspectives on the REMS requirements. There was 1 focus group for each of the 3 medications. A facilitator’s guide was developed that contained the details of how to conduct the focus group along with the medication-specific questions. The questions were based on the REMS requirements as of May 2021 and assessed the impact of the REMS on patient safety, patient access, and health care professional workload; effects from the COVID-19 pandemic; and suggestions to improve the REMS programs. The University of Maryland Institutional Review Board reviewed the materials and processes and made the determination of exempt.
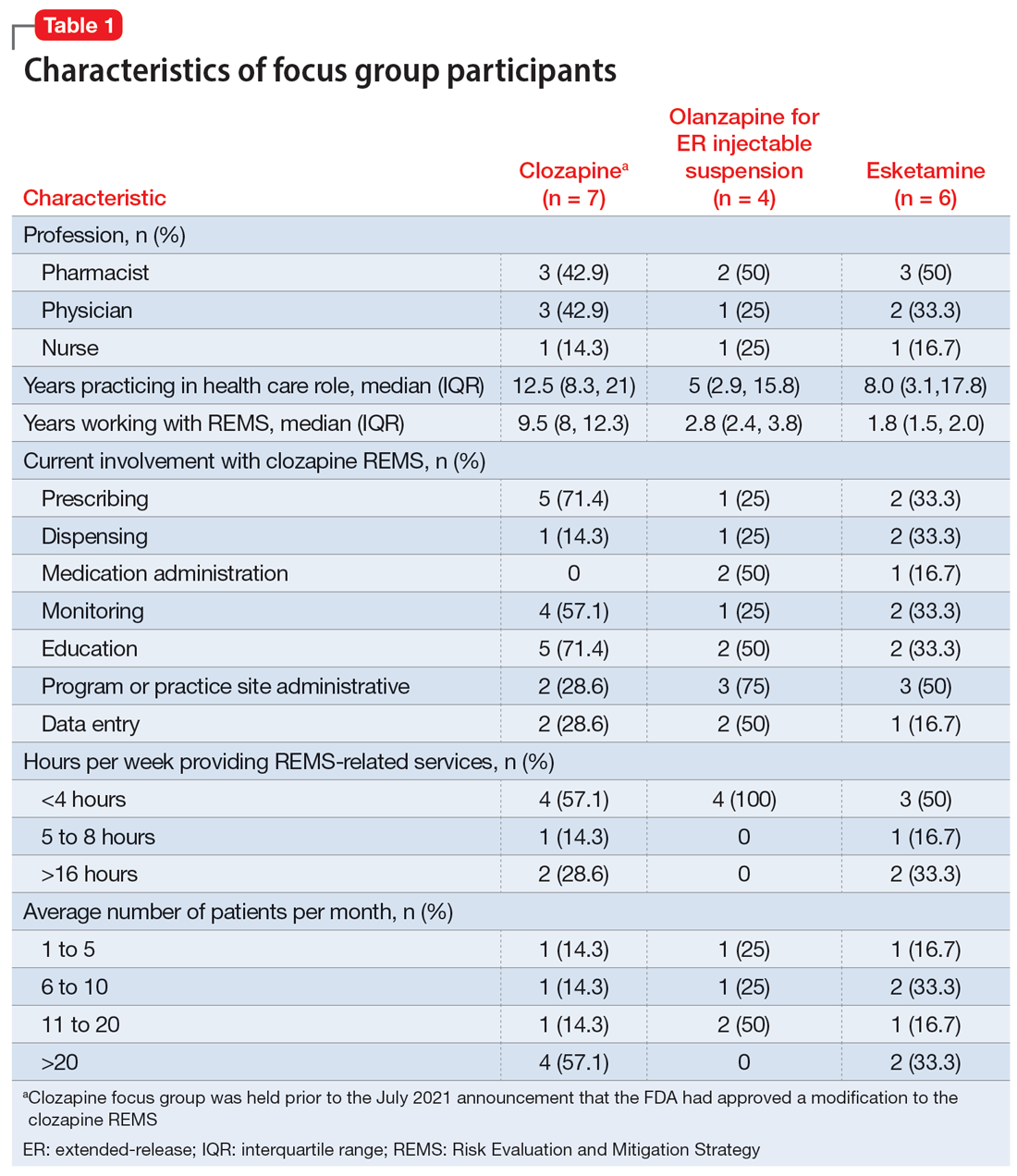
Health care professionals were eligible to participate in a focus group if they had ≥1 year of experience working with patients who use the specific medication and ≥6 months of experience within the past year working with the REMS program for that medication. Participants were excluded if they were employed by a pharmaceutical manufacturer or the FDA. The focus groups were conducted virtually using an online conferencing service during summer 2021 and were scheduled for 90 minutes. Prior to the focus group, participants received information from the “Goals” and “Summary” tabs of the FDA REMS website10 for the specific medication along with patient/caregiver guides, which were available for clozapine and olanzapine for ER injectable suspension. For each focus group, there was a target sample size of 6 to 9 participants. However, there were only 4 participants in the olanzapine for ER injectable suspension focus group, which we believed was due to lower national utilization of this medication. Individuals were only able to participate in 1 focus group, so the unique participant count for all 3 focus groups totaled 17 (Table 1).
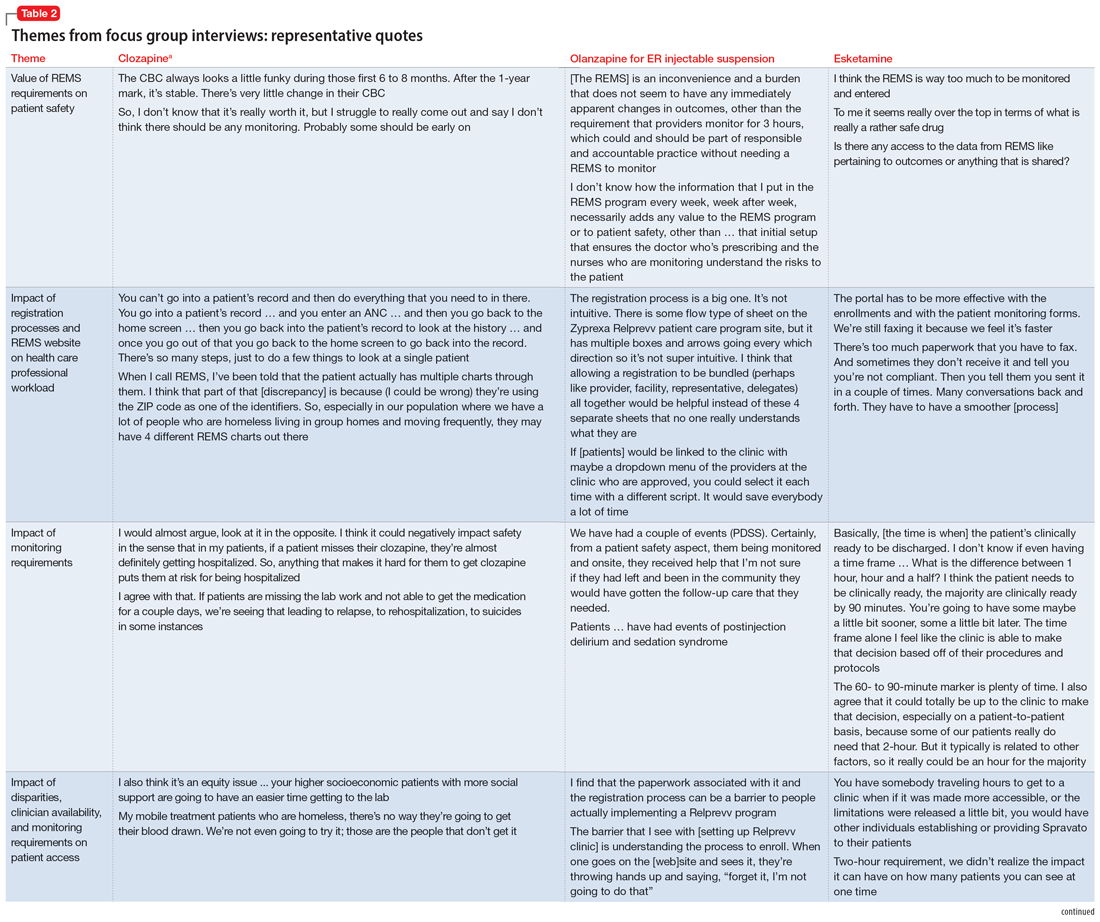
Themes extracted from qualitative analysis of the focus group responses were the value of the REMS programs; registration/enrollment processes and REMS websites; monitoring requirements; care transitions; and COVID considerations (Table 2). While the REMS programs were perceived to increase practitioner and patient awareness of potential harms, discussions centered on the relative cost-to-benefit of the required reporting and other REMS requirements. There were challenges with the registration/enrollment processes and REMS websites that also affected patient care during transitions to different health care settings or clinicians. Patient access was affected by disparities in care related to monitoring requirements and clinician availability.

Continue to: COVID impacted all REMS...
COVID impacted all REMS programs. Physical distancing was an issue for medications that required extensive postadministration monitoring (ie, esketamine and olanzapine for ER injectable suspension). Access to laboratory services was an issue for clozapine.
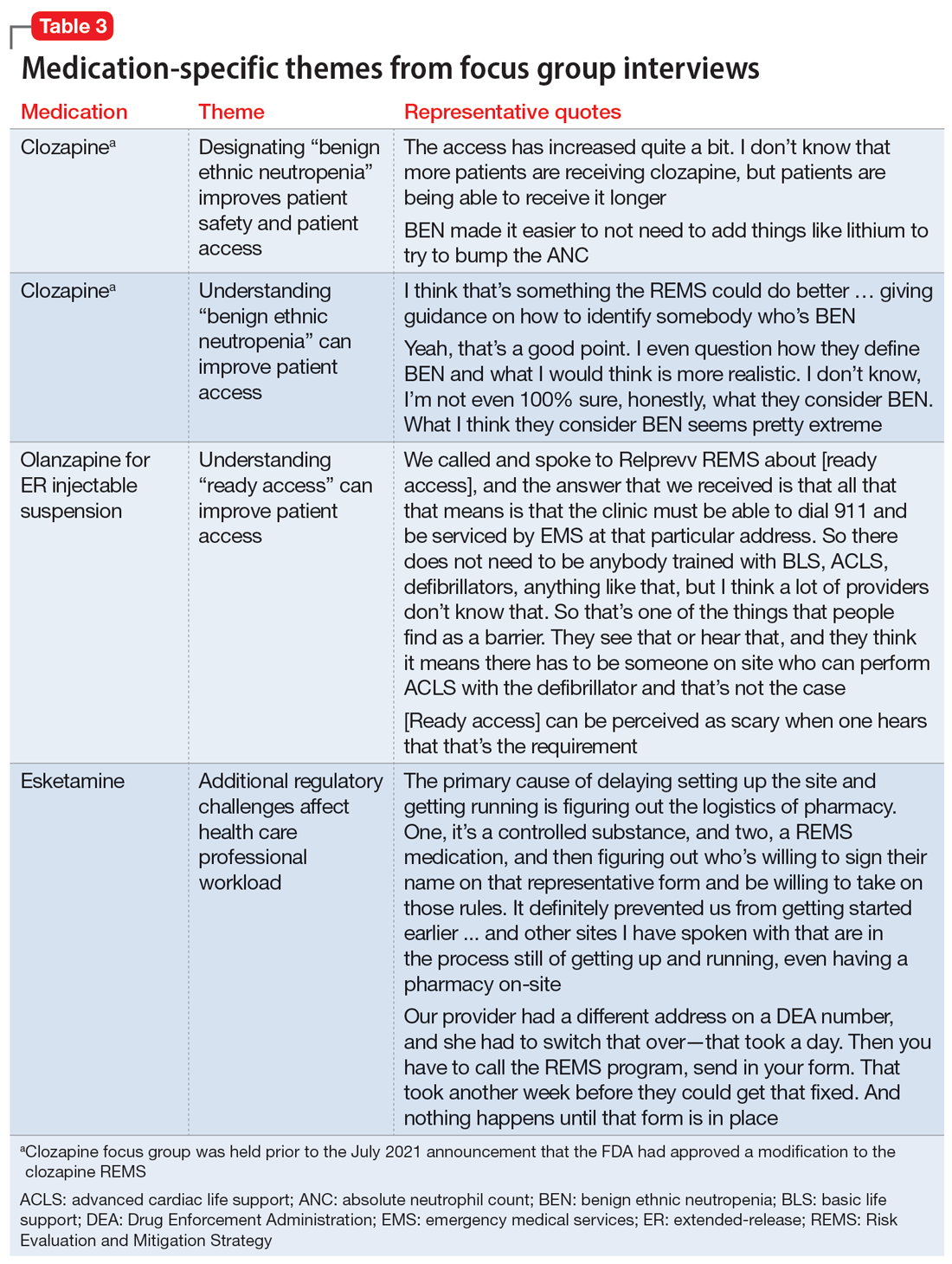
Medication-specific themes are listed in Table 3 and relate to terms and descriptions in the REMS or additional regulatory requirements from the Drug Enforcement Agency (DEA). Suggestions for improvement to the REMS are presented in Table 4.
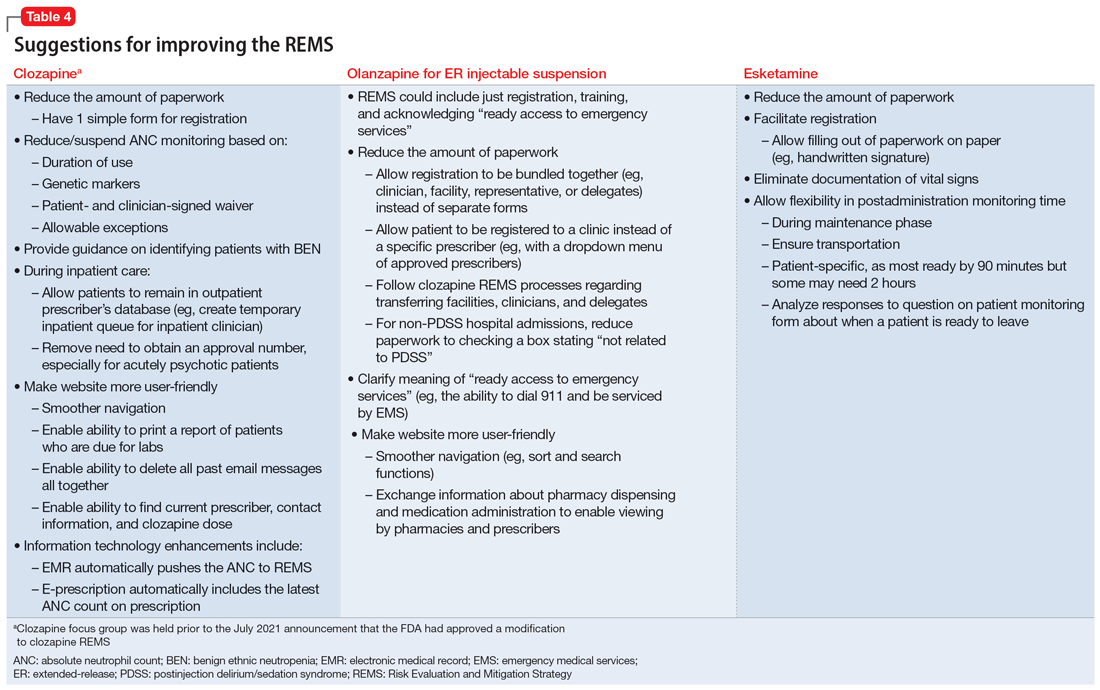
Recommendations for improving REMS
A group consisting of health care professionals, policy experts, and mental health advocates reviewed the information provided by the focus groups and developed the following recommendations.
Overarching recommendations
Each REMS should include a section providing justification for its existence, including a risk analysis of the data regarding the risk the REMS is designed to mitigate. This analysis should be repeated on a regular basis as scientific evidence regarding the risk and its epidemiology evolves. This additional section should also explain how the program requirements of the REMS as implemented (or planned) will achieve the aims of the REMS and weigh the potential benefits of the REMS requirements as implemented (or planned) by the manufacturer vs the potential risks of the REMS requirements as implemented (or planned) by the manufacturer.
Each REMS should have specific quantifiable outcomes. For example, it should specify a reduction in occurrence of the rate of the concerned risk by a specified amount.
Continue to: Ensure adequate...
Ensure adequate stakeholder input during the REMS development and real-world testing in multiple environments before implementing the REMS to identify unanticipated consequences that might impact patient access, patient safety, and health care professional burden. Implementation testing should explore issues such as purchasing and procurement, billing and reimbursement, and relevant factors such as other federal regulations or requirements (eg, the DEA or Medicare).
Ensure harmonization of the REMS forms and processes (eg, initiation and monitoring) for different medications where possible. A prescriber, pharmacist, or system should not face additional barriers to participate in a REMS based on REMS-specific intricacies (ie, prescription systems, data submission systems, or ordering systems). This streamlining will likely decrease clinical inertia to initiate care with the REMS medication, decrease health care professional burden, and improve compliance with REMS requirements.
REMS should anticipate the need for care transitions and employ provisions to ensure seamless care. Considerations should be given to transitions that occur due to:
- Different care settings (eg, inpatient, outpatient, or long-term care)
- Different geographies (eg, patient moves)
- Changes in clinicians, including leaves or absences
- Changes in facilities (eg, pharmacies).
REMS should mirror normal health care professional workflow, including how monitoring data are collected and how and with which frequency pharmacies fill prescriptions.Enhanced information technology to support REMS programs is needed. For example, REMS should be integrated with major electronic patient health record and pharmacy systems to reduce the effort required for clinicians to supply data and automate REMS processes.
For medications that are subject to other agencies and their regulations (eg, the CDC, Centers for Medicare & Medicaid Services, or the DEA), REMS should be required to meet all standards of all agencies with a single system that accommodates normal health care professional workflow.
Continue to: REMS should have a...
REMS should have a standard disclaimer that allows the health care professional to waive certain provisions of the REMS in cases when the specific provisions of the REMS pose a greater risk to the patient than the risk posed by waiving the requirement.
Assure the actions implemented by the industry to meet the requirements for each REMS program are based on peer-reviewed evidence and provide a reasonable expectation to achieve the anticipated benefit.
Ensure that manufacturers make all accumulated REMS data available in a deidentified manner for use by qualified scientific researchers. Additionally, each REMS should have a plan for data access upon initiation and termination of the REMS.
Each REMS should collect data on the performance of the centers and/or personnel who operate the REMS and submit this data for review by qualified outside reviewers. Parameters to assess could include:
- timeliness of response
- timeliness of problem resolution
- data availability and its helpfulness to patient care
- adequacy of resources.
Recommendations for clozapine REMS
These comments relate to the clozapine REMS program prior to the July 2021 announcement that FDA had approved a modification.
Provide a clear definition for “benign ethnic neutropenia.”
Ensure the REMS includes patient-specific adjustments to allow flexibility for monitoring. During COVID, the FDA allowed clinicians to “use their best medical judgment in weighing the benefits and risks of continuing treatment in the absence of laboratory testing.”7 This guidance, which allowed flexibility to absolute neutrophil count (ANC) monitoring, was perceived as positive and safe. Before the changes in the REMS requirements, patients with benign ethnic neutropenia were restricted from accessing their medication or encountered harm from additional pharmacotherapy to mitigate ANC levels.
Continue to: Recommendations for olanzapine for ER injectable suspension REMS
Recommendations for olanzapine for ER injectable suspension REMS
Provide clear explicit instructions on what is required to have “ready access to emergency services.”
Ensure the REMS include patient-specific adjustments to allow flexibility for postadministration monitoring (eg, sedation or blood pressure). Specific patient groups may have differential access to certain types of facilities, transportation, or other resources. For example, consider the administration of olanzapine for ER injectable suspension by a mobile treatment team with an adequate protocol (eg, via videoconferencing or phone calls).
Ensure actions with peer-reviewed evidence demonstrating efficacy/effectiveness are included in the REMS. How was the 3-hour cut-point determined? Has it been reevaluated?
Ensure the REMS requirements allow for seamless care during transitions, particularly when clinicians are on vacation.
Continue to: Recommendations for esketamine REMS
Recommendations for esketamine REMS
Ensure the REMS includes patient-specific adjustments to allow flexibility for postadministration monitoring. Specific patient groups may have differential access to certain types of facilities, transportation, or other resources. For example, consider the administration of esketamine by a mobile treatment team with an adequate protocol (eg, via videoconferencing or phone calls).
Ensure actions with peer-reviewed evidence demonstrating efficacy/effectiveness of requirements are included in the REMS. How was the 2-hour cut-point determined? Has it been reevaluated?
Ensure that the REMS meet all standards of the DEA, with a single system that accommodates normal health care professional workflow.
A summary of the findings
Overall, the REMS programs for these 3 medications were positively perceived for raising awareness of safe medication use for clinicians and patients. Monitoring patients for safety concerns is important and REMS requirements provide accountability.
Continue to: The use of a single shared...
The use of a single shared REMS system for documenting requirements for clozapine (compared to separate systems for each manufacturer) was a positive move forward in implementation. The focus group welcomed the increased awareness of benign ethnic neutropenia as a result of this condition being incorporated in the revised monitoring requirements of the clozapine REMS.
Focus group participants raised the issue of the real-world efficiency of the REMS programs (reduced access and increased clinician workload) vs the benefits (patient safety). They noted that excessive workload could lead to clinicians becoming unwilling to use a medication that requires a REMS. Clinician workload may be further compromised when REMS logistics disrupt the normal workflow and transitions of care between clinicians or settings. This latter aspect is of particular concern for clozapine.
The complexities of the registration and reporting system for olanzapine for ER injectable suspension and the lack of clarity about monitoring were noted to have discouraged the opening of treatment sites. This scarcity of sites may make clinicians hesitant to use this medication, and instead opt for alternative treatments in patients who may be appropriate candidates.
There has also been limited growth of esketamine treatment sites, especially in comparison to ketamine treatment sites.11-14 Esketamine is FDA-approved for treatment-resistant depression in adults and for depressive symptoms in adults with major depressive disorder with acute suicidal ideation or behavior. Ketamine is not FDA-approved for treating depression but is being used off-label to treat this disorder.15 The FDA determined that ketamine does not require a REMS to ensure the benefits outweigh the risks for its approved indications as an anesthetic agent, anesthesia-inducing agent, or supplement to anesthesia. Since ketamine has no REMS requirements, there may be a lower burden for its use. Thus, clinicians are treating patients for depression with this medication without needing to comply with a REMS.16
Technology plays a role in workload burden, and integrating health care processes within current workflow systems, such as using electronic patient health records and pharmacy systems, is recommended. The FDA has been exploring technologies to facilitate the completion of REMS requirements, including mandatory education within the prescribers’ and pharmacists’ workflow.17 This is a complex task that requires multiple stakeholders with differing perspectives and incentives to align.
Continue to: The data collected for the REMS...
The data collected for the REMS program belongs to the medication’s manufacturer. Current regulations do not require manufacturers to make this data available to qualified scientific researchers. A regulatory mandate to establish data sharing methods would improve transparency and enhance efforts to better understand the outcomes of the REMS programs.
A few caveats
Both the overarching and medication-specific recommendations were based on a small number of participants’ discussions related to clozapine, olanzapine for ER injectable suspension, and esketamine. These recommendations do not include other medications with REMS that are used to treat psychiatric disorders, such as loxapine, buprenorphine ER, and buprenorphine transmucosal products. Larger-scale qualitative and quantitative research is needed to better understand health care professionals’ perspectives. Lastly, some of the recommendations outlined in this article are beyond the current purview or authority of the FDA and may require legislative or regulatory action to implement.
Bottom Line
Risk Evaluation and Mitigation Strategy (REMS) programs are designed to help reduce the occurrence and/or severity of serious risks or to inform decision-making. However, REMS requirements may adversely impact patient access to certain REMS medications and clinician burden. Health care professionals can provide informed recommendations for improving the REMS programs for clozapine, olanzapine for extended-release injectable suspension, and esketamine.
Related Resources
- FDA. Frequently asked questions (FAQs) about REMS. www.fda.gov/drugs/risk-evaluation-and-mitigation-strategies-rems/frequently-asked-questions-faqs-about-rems
Drug Brand Names
Buprenorphine extended-release • Sublocade
Buprenorphine transmucosal • Subutex, Suboxone
Clozapine • Clozaril
Esketamine • Spravato
Ketamine • Ketalar
Lithium • Eskalith, Lithobid
Loxapine • Adasuve
Olanzapine extended-release injectable suspension • Zyprexa Relprevv
1. U.S. Food and Drug Administration. Risk Evaluation and Mitigation Strategies. Accessed January 18, 2023. https://www.fda.gov/drugs/drug-safety-and-availability/risk-evaluation-and-mitigation-strategies-rems
2. U.S. Department of Health and Human Services, Food and Drug Administration. Format and Content of a REMS Document. Guidance for Industry. Accessed January 18, 2023. https://www.fda.gov/media/77846/download
3. U.S. Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS), Clozapine. Accessed January 18, 2023. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=RemsDetails.page&REMS=351
4. The National Association of State Mental Health Program Directors. Clozapine underutilization: addressing the barriers. Accessed September 30, 2019. https://nasmhpd.org/sites/default/files/Assessment%201_Clozapine%20Underutilization.pdf
5. U.S. Food and Drug Administration. FDA is temporarily exercising enforcement discretion with respect to certain clozapine REMS program requirements to ensure continuity of care for patients taking clozapine. Updated November 22, 2022. Accessed June 1, 2023. https://www.fda.gov/drugs/drug-safety-and-availability/fda-temporarily-exercising-enforcement-discretion-respect-certain-clozapine-rems-program
6. Tanzi M. REMS issues affect clozapine, isotretinoin. Pharmacy Today. 2022;28(3):49.
7. U.S. Food and Drug Administration. Coronavirus (COVID-19) update: FDA provides update on patient access to certain REMS drugs during COVID-19 public health emergency. Accessed June 1, 2023. https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-provides-update-patient-access-certain-rems-drugs-during-covid-19
8. U.S. Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS), Spravato (esketamine). Accessed January 18, 2023. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=386
9. U.S. Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS), Zyprexa Relprevv (olanzapine). Accessed January 18, 2023. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=74
10. U.S. Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS). Accessed January 18, 2023. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm
11. Parikh SV, Lopez D, Vande Voort JL, et al. Developing an IV ketamine clinic for treatment-resistant depression: a primer. Psychopharmacol Bull. 2021;51(3):109-124.
12. Dodge D. The ketamine cure. The New York Times. November 4, 2021. Updated November 5, 2021. Accessed June 1, 2023. https://www.nytimes.com/2021/11/04/well/ketamine-therapy-depression.html
13. Burton KW. Time for a national ketamine registry, experts say. Medscape. February 15, 2023. Accessed June 1, 2023. https://www.medscape.com/viewarticle/988310
14. Wilkinson ST, Howard DH, Busch SH. Psychiatric practice patterns and barriers to the adoption of esketamine. JAMA. 2019;322(11):1039-1040. doi:10.1001/jama.2019.10728
15. Wilkinson ST, Toprak M, Turner MS, et al. A survey of the clinical, off-label use of ketamine as a treatment for psychiatric disorders. Am J Psychiatry. 2017;174(7):695-696. doi:10.1176/appi.ajp.2017.17020239
16. Pai SM, Gries JM; ACCP Public Policy Committee. Off-label use of ketamine: a challenging drug treatment delivery model with an inherently unfavorable risk-benefit profile. J Clin Pharmacol. 2022;62(1):10-13. doi:10.1002/jcph.1983
17. Risk Evaluation and Mitigation Strategies (REMS) Integration. Accessed June 1, 2023. https://confluence.hl7.org/display/COD/Risk+Evaluation+and+Mitigation+Strategies+%28REMS%29+Integration
A Risk Evaluation and Mitigation Strategy (REMS) is a drug safety program the FDA can require for certain medications with serious safety concerns to help ensure the benefits of the medication outweigh its risks (Box1). The FDA may require medication guides, patient package inserts, communication plans for health care professionals, and/or certain packaging and safe disposal technologies for medications that pose a serious risk of abuse or overdose. The FDA may also require elements to assure safe use and/or an implementation system be included in the REMS. Pharmaceutical manufacturers then develop a proposed REMS for FDA review.2 If the FDA approves the proposed REMS, the manufacturer is responsible for implementing the REMS requirements.
Box
There are many myths and misconceptions surrounding psychiatry, the branch of medicine that deals with the diagnosis, treatment, and prevention of mental illness. Some of the most common myths include:
The FDA provides this description of a Risk Evaluation and Mitigation Strategy (REMS):
“A [REMS] is a drug safety program that the U.S. Food and Drug Administration (FDA) can require for certain medications with serious safety concerns to help ensure the benefits of the medication outweigh its risks. REMS are designed to reinforce medication use behaviors and actions that support the safe use of that medication. While all medications have labeling that informs health care stakeholders about medication risks, only a few medications require a REMS. REMS are not designed to mitigate all the adverse events of a medication, these are communicated to health care providers in the medication’s prescribing information. Rather, REMS focus on preventing, monitoring and/or managing a specific serious risk by informing, educating and/or reinforcing actions to reduce the frequency and/or severity of the event.”1
The REMS program for clozapine3 has been the subject of much discussion in the psychiatric community. The adverse impact of the 2015 update to the clozapine REMS program was emphasized at meetings of both the American Psychiatric Association and the College of Psychiatric and Neurologic Pharmacists. A white paper published by the National Association of State Mental Health Program Directors shortly after the 2015 update concluded, “clozapine is underused due to a variety of barriers related to the drug and its properties, the health care system, regulatory requirements, and reimbursement issues.”4 After an update to the clozapine REMS program in 2021, the FDA temporarily suspended enforcement of certain requirements due to concerns from health care professionals about patient access to the medication because of problems with implementing the clozapine REMS program.5,6 In November 2022, the FDA issued a second announcement of enforcement discretion related to additional requirements of the REMS program.5 The FDA had previously announced a decision to not take action regarding adherence to REMS requirements for certain laboratory tests in March 2020, during the COVID-19 pandemic.7
REMS programs for other psychiatric medications may also present challenges. The REMS programs for esketamine8 and olanzapine for extended-release (ER) injectable suspension9 include certain risks that require postadministration monitoring. Some facilities have had to dedicate additional space and clinician time to ensure REMS requirements are met.
To further understand health care professionals’ perspectives regarding the value and burden of these REMS programs, a collaborative effort of the University of Maryland (College Park and Baltimore campuses) Center of Excellence in Regulatory Science and Innovation with the FDA was undertaken. The REMS for clozapine, olanzapine for ER injectable suspension, and esketamine were examined to develop recommendations for improving patient access while ensuring safe medication use and limiting the impact on health care professionals.
Assessing the REMS programs
Focus groups were held with health care professionals nominated by professional organizations to gather their perspectives on the REMS requirements. There was 1 focus group for each of the 3 medications. A facilitator’s guide was developed that contained the details of how to conduct the focus group along with the medication-specific questions. The questions were based on the REMS requirements as of May 2021 and assessed the impact of the REMS on patient safety, patient access, and health care professional workload; effects from the COVID-19 pandemic; and suggestions to improve the REMS programs. The University of Maryland Institutional Review Board reviewed the materials and processes and made the determination of exempt.
Health care professionals were eligible to participate in a focus group if they had ≥1 year of experience working with patients who use the specific medication and ≥6 months of experience within the past year working with the REMS program for that medication. Participants were excluded if they were employed by a pharmaceutical manufacturer or the FDA. The focus groups were conducted virtually using an online conferencing service during summer 2021 and were scheduled for 90 minutes. Prior to the focus group, participants received information from the “Goals” and “Summary” tabs of the FDA REMS website10 for the specific medication along with patient/caregiver guides, which were available for clozapine and olanzapine for ER injectable suspension. For each focus group, there was a target sample size of 6 to 9 participants. However, there were only 4 participants in the olanzapine for ER injectable suspension focus group, which we believed was due to lower national utilization of this medication. Individuals were only able to participate in 1 focus group, so the unique participant count for all 3 focus groups totaled 17 (Table 1).

Themes extracted from qualitative analysis of the focus group responses were the value of the REMS programs; registration/enrollment processes and REMS websites; monitoring requirements; care transitions; and COVID considerations (Table 2). While the REMS programs were perceived to increase practitioner and patient awareness of potential harms, discussions centered on the relative cost-to-benefit of the required reporting and other REMS requirements. There were challenges with the registration/enrollment processes and REMS websites that also affected patient care during transitions to different health care settings or clinicians. Patient access was affected by disparities in care related to monitoring requirements and clinician availability.


Continue to: COVID impacted all REMS...
COVID impacted all REMS programs. Physical distancing was an issue for medications that required extensive postadministration monitoring (ie, esketamine and olanzapine for ER injectable suspension). Access to laboratory services was an issue for clozapine.

Medication-specific themes are listed in Table 3 and relate to terms and descriptions in the REMS or additional regulatory requirements from the Drug Enforcement Agency (DEA). Suggestions for improvement to the REMS are presented in Table 4.

Recommendations for improving REMS
A group consisting of health care professionals, policy experts, and mental health advocates reviewed the information provided by the focus groups and developed the following recommendations.
Overarching recommendations
Each REMS should include a section providing justification for its existence, including a risk analysis of the data regarding the risk the REMS is designed to mitigate. This analysis should be repeated on a regular basis as scientific evidence regarding the risk and its epidemiology evolves. This additional section should also explain how the program requirements of the REMS as implemented (or planned) will achieve the aims of the REMS and weigh the potential benefits of the REMS requirements as implemented (or planned) by the manufacturer vs the potential risks of the REMS requirements as implemented (or planned) by the manufacturer.
Each REMS should have specific quantifiable outcomes. For example, it should specify a reduction in occurrence of the rate of the concerned risk by a specified amount.
Continue to: Ensure adequate...
Ensure adequate stakeholder input during the REMS development and real-world testing in multiple environments before implementing the REMS to identify unanticipated consequences that might impact patient access, patient safety, and health care professional burden. Implementation testing should explore issues such as purchasing and procurement, billing and reimbursement, and relevant factors such as other federal regulations or requirements (eg, the DEA or Medicare).
Ensure harmonization of the REMS forms and processes (eg, initiation and monitoring) for different medications where possible. A prescriber, pharmacist, or system should not face additional barriers to participate in a REMS based on REMS-specific intricacies (ie, prescription systems, data submission systems, or ordering systems). This streamlining will likely decrease clinical inertia to initiate care with the REMS medication, decrease health care professional burden, and improve compliance with REMS requirements.
REMS should anticipate the need for care transitions and employ provisions to ensure seamless care. Considerations should be given to transitions that occur due to:
- Different care settings (eg, inpatient, outpatient, or long-term care)
- Different geographies (eg, patient moves)
- Changes in clinicians, including leaves or absences
- Changes in facilities (eg, pharmacies).
REMS should mirror normal health care professional workflow, including how monitoring data are collected and how and with which frequency pharmacies fill prescriptions.Enhanced information technology to support REMS programs is needed. For example, REMS should be integrated with major electronic patient health record and pharmacy systems to reduce the effort required for clinicians to supply data and automate REMS processes.
For medications that are subject to other agencies and their regulations (eg, the CDC, Centers for Medicare & Medicaid Services, or the DEA), REMS should be required to meet all standards of all agencies with a single system that accommodates normal health care professional workflow.
Continue to: REMS should have a...
REMS should have a standard disclaimer that allows the health care professional to waive certain provisions of the REMS in cases when the specific provisions of the REMS pose a greater risk to the patient than the risk posed by waiving the requirement.
Assure the actions implemented by the industry to meet the requirements for each REMS program are based on peer-reviewed evidence and provide a reasonable expectation to achieve the anticipated benefit.
Ensure that manufacturers make all accumulated REMS data available in a deidentified manner for use by qualified scientific researchers. Additionally, each REMS should have a plan for data access upon initiation and termination of the REMS.
Each REMS should collect data on the performance of the centers and/or personnel who operate the REMS and submit this data for review by qualified outside reviewers. Parameters to assess could include:
- timeliness of response
- timeliness of problem resolution
- data availability and its helpfulness to patient care
- adequacy of resources.
Recommendations for clozapine REMS
These comments relate to the clozapine REMS program prior to the July 2021 announcement that FDA had approved a modification.
Provide a clear definition for “benign ethnic neutropenia.”
Ensure the REMS includes patient-specific adjustments to allow flexibility for monitoring. During COVID, the FDA allowed clinicians to “use their best medical judgment in weighing the benefits and risks of continuing treatment in the absence of laboratory testing.”7 This guidance, which allowed flexibility to absolute neutrophil count (ANC) monitoring, was perceived as positive and safe. Before the changes in the REMS requirements, patients with benign ethnic neutropenia were restricted from accessing their medication or encountered harm from additional pharmacotherapy to mitigate ANC levels.
Continue to: Recommendations for olanzapine for ER injectable suspension REMS
Recommendations for olanzapine for ER injectable suspension REMS
Provide clear explicit instructions on what is required to have “ready access to emergency services.”
Ensure the REMS include patient-specific adjustments to allow flexibility for postadministration monitoring (eg, sedation or blood pressure). Specific patient groups may have differential access to certain types of facilities, transportation, or other resources. For example, consider the administration of olanzapine for ER injectable suspension by a mobile treatment team with an adequate protocol (eg, via videoconferencing or phone calls).
Ensure actions with peer-reviewed evidence demonstrating efficacy/effectiveness are included in the REMS. How was the 3-hour cut-point determined? Has it been reevaluated?
Ensure the REMS requirements allow for seamless care during transitions, particularly when clinicians are on vacation.
Continue to: Recommendations for esketamine REMS
Recommendations for esketamine REMS
Ensure the REMS includes patient-specific adjustments to allow flexibility for postadministration monitoring. Specific patient groups may have differential access to certain types of facilities, transportation, or other resources. For example, consider the administration of esketamine by a mobile treatment team with an adequate protocol (eg, via videoconferencing or phone calls).
Ensure actions with peer-reviewed evidence demonstrating efficacy/effectiveness of requirements are included in the REMS. How was the 2-hour cut-point determined? Has it been reevaluated?
Ensure that the REMS meet all standards of the DEA, with a single system that accommodates normal health care professional workflow.
A summary of the findings
Overall, the REMS programs for these 3 medications were positively perceived for raising awareness of safe medication use for clinicians and patients. Monitoring patients for safety concerns is important and REMS requirements provide accountability.
Continue to: The use of a single shared...
The use of a single shared REMS system for documenting requirements for clozapine (compared to separate systems for each manufacturer) was a positive move forward in implementation. The focus group welcomed the increased awareness of benign ethnic neutropenia as a result of this condition being incorporated in the revised monitoring requirements of the clozapine REMS.
Focus group participants raised the issue of the real-world efficiency of the REMS programs (reduced access and increased clinician workload) vs the benefits (patient safety). They noted that excessive workload could lead to clinicians becoming unwilling to use a medication that requires a REMS. Clinician workload may be further compromised when REMS logistics disrupt the normal workflow and transitions of care between clinicians or settings. This latter aspect is of particular concern for clozapine.
The complexities of the registration and reporting system for olanzapine for ER injectable suspension and the lack of clarity about monitoring were noted to have discouraged the opening of treatment sites. This scarcity of sites may make clinicians hesitant to use this medication, and instead opt for alternative treatments in patients who may be appropriate candidates.
There has also been limited growth of esketamine treatment sites, especially in comparison to ketamine treatment sites.11-14 Esketamine is FDA-approved for treatment-resistant depression in adults and for depressive symptoms in adults with major depressive disorder with acute suicidal ideation or behavior. Ketamine is not FDA-approved for treating depression but is being used off-label to treat this disorder.15 The FDA determined that ketamine does not require a REMS to ensure the benefits outweigh the risks for its approved indications as an anesthetic agent, anesthesia-inducing agent, or supplement to anesthesia. Since ketamine has no REMS requirements, there may be a lower burden for its use. Thus, clinicians are treating patients for depression with this medication without needing to comply with a REMS.16
Technology plays a role in workload burden, and integrating health care processes within current workflow systems, such as using electronic patient health records and pharmacy systems, is recommended. The FDA has been exploring technologies to facilitate the completion of REMS requirements, including mandatory education within the prescribers’ and pharmacists’ workflow.17 This is a complex task that requires multiple stakeholders with differing perspectives and incentives to align.
Continue to: The data collected for the REMS...
The data collected for the REMS program belongs to the medication’s manufacturer. Current regulations do not require manufacturers to make this data available to qualified scientific researchers. A regulatory mandate to establish data sharing methods would improve transparency and enhance efforts to better understand the outcomes of the REMS programs.
A few caveats
Both the overarching and medication-specific recommendations were based on a small number of participants’ discussions related to clozapine, olanzapine for ER injectable suspension, and esketamine. These recommendations do not include other medications with REMS that are used to treat psychiatric disorders, such as loxapine, buprenorphine ER, and buprenorphine transmucosal products. Larger-scale qualitative and quantitative research is needed to better understand health care professionals’ perspectives. Lastly, some of the recommendations outlined in this article are beyond the current purview or authority of the FDA and may require legislative or regulatory action to implement.
Bottom Line
Risk Evaluation and Mitigation Strategy (REMS) programs are designed to help reduce the occurrence and/or severity of serious risks or to inform decision-making. However, REMS requirements may adversely impact patient access to certain REMS medications and clinician burden. Health care professionals can provide informed recommendations for improving the REMS programs for clozapine, olanzapine for extended-release injectable suspension, and esketamine.
Related Resources
- FDA. Frequently asked questions (FAQs) about REMS. www.fda.gov/drugs/risk-evaluation-and-mitigation-strategies-rems/frequently-asked-questions-faqs-about-rems
Drug Brand Names
Buprenorphine extended-release • Sublocade
Buprenorphine transmucosal • Subutex, Suboxone
Clozapine • Clozaril
Esketamine • Spravato
Ketamine • Ketalar
Lithium • Eskalith, Lithobid
Loxapine • Adasuve
Olanzapine extended-release injectable suspension • Zyprexa Relprevv
A Risk Evaluation and Mitigation Strategy (REMS) is a drug safety program the FDA can require for certain medications with serious safety concerns to help ensure the benefits of the medication outweigh its risks (Box1). The FDA may require medication guides, patient package inserts, communication plans for health care professionals, and/or certain packaging and safe disposal technologies for medications that pose a serious risk of abuse or overdose. The FDA may also require elements to assure safe use and/or an implementation system be included in the REMS. Pharmaceutical manufacturers then develop a proposed REMS for FDA review.2 If the FDA approves the proposed REMS, the manufacturer is responsible for implementing the REMS requirements.
Box
There are many myths and misconceptions surrounding psychiatry, the branch of medicine that deals with the diagnosis, treatment, and prevention of mental illness. Some of the most common myths include:
The FDA provides this description of a Risk Evaluation and Mitigation Strategy (REMS):
“A [REMS] is a drug safety program that the U.S. Food and Drug Administration (FDA) can require for certain medications with serious safety concerns to help ensure the benefits of the medication outweigh its risks. REMS are designed to reinforce medication use behaviors and actions that support the safe use of that medication. While all medications have labeling that informs health care stakeholders about medication risks, only a few medications require a REMS. REMS are not designed to mitigate all the adverse events of a medication, these are communicated to health care providers in the medication’s prescribing information. Rather, REMS focus on preventing, monitoring and/or managing a specific serious risk by informing, educating and/or reinforcing actions to reduce the frequency and/or severity of the event.”1
The REMS program for clozapine3 has been the subject of much discussion in the psychiatric community. The adverse impact of the 2015 update to the clozapine REMS program was emphasized at meetings of both the American Psychiatric Association and the College of Psychiatric and Neurologic Pharmacists. A white paper published by the National Association of State Mental Health Program Directors shortly after the 2015 update concluded, “clozapine is underused due to a variety of barriers related to the drug and its properties, the health care system, regulatory requirements, and reimbursement issues.”4 After an update to the clozapine REMS program in 2021, the FDA temporarily suspended enforcement of certain requirements due to concerns from health care professionals about patient access to the medication because of problems with implementing the clozapine REMS program.5,6 In November 2022, the FDA issued a second announcement of enforcement discretion related to additional requirements of the REMS program.5 The FDA had previously announced a decision to not take action regarding adherence to REMS requirements for certain laboratory tests in March 2020, during the COVID-19 pandemic.7
REMS programs for other psychiatric medications may also present challenges. The REMS programs for esketamine8 and olanzapine for extended-release (ER) injectable suspension9 include certain risks that require postadministration monitoring. Some facilities have had to dedicate additional space and clinician time to ensure REMS requirements are met.
To further understand health care professionals’ perspectives regarding the value and burden of these REMS programs, a collaborative effort of the University of Maryland (College Park and Baltimore campuses) Center of Excellence in Regulatory Science and Innovation with the FDA was undertaken. The REMS for clozapine, olanzapine for ER injectable suspension, and esketamine were examined to develop recommendations for improving patient access while ensuring safe medication use and limiting the impact on health care professionals.
Assessing the REMS programs
Focus groups were held with health care professionals nominated by professional organizations to gather their perspectives on the REMS requirements. There was 1 focus group for each of the 3 medications. A facilitator’s guide was developed that contained the details of how to conduct the focus group along with the medication-specific questions. The questions were based on the REMS requirements as of May 2021 and assessed the impact of the REMS on patient safety, patient access, and health care professional workload; effects from the COVID-19 pandemic; and suggestions to improve the REMS programs. The University of Maryland Institutional Review Board reviewed the materials and processes and made the determination of exempt.
Health care professionals were eligible to participate in a focus group if they had ≥1 year of experience working with patients who use the specific medication and ≥6 months of experience within the past year working with the REMS program for that medication. Participants were excluded if they were employed by a pharmaceutical manufacturer or the FDA. The focus groups were conducted virtually using an online conferencing service during summer 2021 and were scheduled for 90 minutes. Prior to the focus group, participants received information from the “Goals” and “Summary” tabs of the FDA REMS website10 for the specific medication along with patient/caregiver guides, which were available for clozapine and olanzapine for ER injectable suspension. For each focus group, there was a target sample size of 6 to 9 participants. However, there were only 4 participants in the olanzapine for ER injectable suspension focus group, which we believed was due to lower national utilization of this medication. Individuals were only able to participate in 1 focus group, so the unique participant count for all 3 focus groups totaled 17 (Table 1).

Themes extracted from qualitative analysis of the focus group responses were the value of the REMS programs; registration/enrollment processes and REMS websites; monitoring requirements; care transitions; and COVID considerations (Table 2). While the REMS programs were perceived to increase practitioner and patient awareness of potential harms, discussions centered on the relative cost-to-benefit of the required reporting and other REMS requirements. There were challenges with the registration/enrollment processes and REMS websites that also affected patient care during transitions to different health care settings or clinicians. Patient access was affected by disparities in care related to monitoring requirements and clinician availability.


Continue to: COVID impacted all REMS...
COVID impacted all REMS programs. Physical distancing was an issue for medications that required extensive postadministration monitoring (ie, esketamine and olanzapine for ER injectable suspension). Access to laboratory services was an issue for clozapine.

Medication-specific themes are listed in Table 3 and relate to terms and descriptions in the REMS or additional regulatory requirements from the Drug Enforcement Agency (DEA). Suggestions for improvement to the REMS are presented in Table 4.

Recommendations for improving REMS
A group consisting of health care professionals, policy experts, and mental health advocates reviewed the information provided by the focus groups and developed the following recommendations.
Overarching recommendations
Each REMS should include a section providing justification for its existence, including a risk analysis of the data regarding the risk the REMS is designed to mitigate. This analysis should be repeated on a regular basis as scientific evidence regarding the risk and its epidemiology evolves. This additional section should also explain how the program requirements of the REMS as implemented (or planned) will achieve the aims of the REMS and weigh the potential benefits of the REMS requirements as implemented (or planned) by the manufacturer vs the potential risks of the REMS requirements as implemented (or planned) by the manufacturer.
Each REMS should have specific quantifiable outcomes. For example, it should specify a reduction in occurrence of the rate of the concerned risk by a specified amount.
Continue to: Ensure adequate...
Ensure adequate stakeholder input during the REMS development and real-world testing in multiple environments before implementing the REMS to identify unanticipated consequences that might impact patient access, patient safety, and health care professional burden. Implementation testing should explore issues such as purchasing and procurement, billing and reimbursement, and relevant factors such as other federal regulations or requirements (eg, the DEA or Medicare).
Ensure harmonization of the REMS forms and processes (eg, initiation and monitoring) for different medications where possible. A prescriber, pharmacist, or system should not face additional barriers to participate in a REMS based on REMS-specific intricacies (ie, prescription systems, data submission systems, or ordering systems). This streamlining will likely decrease clinical inertia to initiate care with the REMS medication, decrease health care professional burden, and improve compliance with REMS requirements.
REMS should anticipate the need for care transitions and employ provisions to ensure seamless care. Considerations should be given to transitions that occur due to:
- Different care settings (eg, inpatient, outpatient, or long-term care)
- Different geographies (eg, patient moves)
- Changes in clinicians, including leaves or absences
- Changes in facilities (eg, pharmacies).
REMS should mirror normal health care professional workflow, including how monitoring data are collected and how and with which frequency pharmacies fill prescriptions.Enhanced information technology to support REMS programs is needed. For example, REMS should be integrated with major electronic patient health record and pharmacy systems to reduce the effort required for clinicians to supply data and automate REMS processes.
For medications that are subject to other agencies and their regulations (eg, the CDC, Centers for Medicare & Medicaid Services, or the DEA), REMS should be required to meet all standards of all agencies with a single system that accommodates normal health care professional workflow.
Continue to: REMS should have a...
REMS should have a standard disclaimer that allows the health care professional to waive certain provisions of the REMS in cases when the specific provisions of the REMS pose a greater risk to the patient than the risk posed by waiving the requirement.
Assure the actions implemented by the industry to meet the requirements for each REMS program are based on peer-reviewed evidence and provide a reasonable expectation to achieve the anticipated benefit.
Ensure that manufacturers make all accumulated REMS data available in a deidentified manner for use by qualified scientific researchers. Additionally, each REMS should have a plan for data access upon initiation and termination of the REMS.
Each REMS should collect data on the performance of the centers and/or personnel who operate the REMS and submit this data for review by qualified outside reviewers. Parameters to assess could include:
- timeliness of response
- timeliness of problem resolution
- data availability and its helpfulness to patient care
- adequacy of resources.
Recommendations for clozapine REMS
These comments relate to the clozapine REMS program prior to the July 2021 announcement that FDA had approved a modification.
Provide a clear definition for “benign ethnic neutropenia.”
Ensure the REMS includes patient-specific adjustments to allow flexibility for monitoring. During COVID, the FDA allowed clinicians to “use their best medical judgment in weighing the benefits and risks of continuing treatment in the absence of laboratory testing.”7 This guidance, which allowed flexibility to absolute neutrophil count (ANC) monitoring, was perceived as positive and safe. Before the changes in the REMS requirements, patients with benign ethnic neutropenia were restricted from accessing their medication or encountered harm from additional pharmacotherapy to mitigate ANC levels.
Continue to: Recommendations for olanzapine for ER injectable suspension REMS
Recommendations for olanzapine for ER injectable suspension REMS
Provide clear explicit instructions on what is required to have “ready access to emergency services.”
Ensure the REMS include patient-specific adjustments to allow flexibility for postadministration monitoring (eg, sedation or blood pressure). Specific patient groups may have differential access to certain types of facilities, transportation, or other resources. For example, consider the administration of olanzapine for ER injectable suspension by a mobile treatment team with an adequate protocol (eg, via videoconferencing or phone calls).
Ensure actions with peer-reviewed evidence demonstrating efficacy/effectiveness are included in the REMS. How was the 3-hour cut-point determined? Has it been reevaluated?
Ensure the REMS requirements allow for seamless care during transitions, particularly when clinicians are on vacation.
Continue to: Recommendations for esketamine REMS
Recommendations for esketamine REMS
Ensure the REMS includes patient-specific adjustments to allow flexibility for postadministration monitoring. Specific patient groups may have differential access to certain types of facilities, transportation, or other resources. For example, consider the administration of esketamine by a mobile treatment team with an adequate protocol (eg, via videoconferencing or phone calls).
Ensure actions with peer-reviewed evidence demonstrating efficacy/effectiveness of requirements are included in the REMS. How was the 2-hour cut-point determined? Has it been reevaluated?
Ensure that the REMS meet all standards of the DEA, with a single system that accommodates normal health care professional workflow.
A summary of the findings
Overall, the REMS programs for these 3 medications were positively perceived for raising awareness of safe medication use for clinicians and patients. Monitoring patients for safety concerns is important and REMS requirements provide accountability.
Continue to: The use of a single shared...
The use of a single shared REMS system for documenting requirements for clozapine (compared to separate systems for each manufacturer) was a positive move forward in implementation. The focus group welcomed the increased awareness of benign ethnic neutropenia as a result of this condition being incorporated in the revised monitoring requirements of the clozapine REMS.
Focus group participants raised the issue of the real-world efficiency of the REMS programs (reduced access and increased clinician workload) vs the benefits (patient safety). They noted that excessive workload could lead to clinicians becoming unwilling to use a medication that requires a REMS. Clinician workload may be further compromised when REMS logistics disrupt the normal workflow and transitions of care between clinicians or settings. This latter aspect is of particular concern for clozapine.
The complexities of the registration and reporting system for olanzapine for ER injectable suspension and the lack of clarity about monitoring were noted to have discouraged the opening of treatment sites. This scarcity of sites may make clinicians hesitant to use this medication, and instead opt for alternative treatments in patients who may be appropriate candidates.
There has also been limited growth of esketamine treatment sites, especially in comparison to ketamine treatment sites.11-14 Esketamine is FDA-approved for treatment-resistant depression in adults and for depressive symptoms in adults with major depressive disorder with acute suicidal ideation or behavior. Ketamine is not FDA-approved for treating depression but is being used off-label to treat this disorder.15 The FDA determined that ketamine does not require a REMS to ensure the benefits outweigh the risks for its approved indications as an anesthetic agent, anesthesia-inducing agent, or supplement to anesthesia. Since ketamine has no REMS requirements, there may be a lower burden for its use. Thus, clinicians are treating patients for depression with this medication without needing to comply with a REMS.16
Technology plays a role in workload burden, and integrating health care processes within current workflow systems, such as using electronic patient health records and pharmacy systems, is recommended. The FDA has been exploring technologies to facilitate the completion of REMS requirements, including mandatory education within the prescribers’ and pharmacists’ workflow.17 This is a complex task that requires multiple stakeholders with differing perspectives and incentives to align.
Continue to: The data collected for the REMS...
The data collected for the REMS program belongs to the medication’s manufacturer. Current regulations do not require manufacturers to make this data available to qualified scientific researchers. A regulatory mandate to establish data sharing methods would improve transparency and enhance efforts to better understand the outcomes of the REMS programs.
A few caveats
Both the overarching and medication-specific recommendations were based on a small number of participants’ discussions related to clozapine, olanzapine for ER injectable suspension, and esketamine. These recommendations do not include other medications with REMS that are used to treat psychiatric disorders, such as loxapine, buprenorphine ER, and buprenorphine transmucosal products. Larger-scale qualitative and quantitative research is needed to better understand health care professionals’ perspectives. Lastly, some of the recommendations outlined in this article are beyond the current purview or authority of the FDA and may require legislative or regulatory action to implement.
Bottom Line
Risk Evaluation and Mitigation Strategy (REMS) programs are designed to help reduce the occurrence and/or severity of serious risks or to inform decision-making. However, REMS requirements may adversely impact patient access to certain REMS medications and clinician burden. Health care professionals can provide informed recommendations for improving the REMS programs for clozapine, olanzapine for extended-release injectable suspension, and esketamine.
Related Resources
- FDA. Frequently asked questions (FAQs) about REMS. www.fda.gov/drugs/risk-evaluation-and-mitigation-strategies-rems/frequently-asked-questions-faqs-about-rems
Drug Brand Names
Buprenorphine extended-release • Sublocade
Buprenorphine transmucosal • Subutex, Suboxone
Clozapine • Clozaril
Esketamine • Spravato
Ketamine • Ketalar
Lithium • Eskalith, Lithobid
Loxapine • Adasuve
Olanzapine extended-release injectable suspension • Zyprexa Relprevv
1. U.S. Food and Drug Administration. Risk Evaluation and Mitigation Strategies. Accessed January 18, 2023. https://www.fda.gov/drugs/drug-safety-and-availability/risk-evaluation-and-mitigation-strategies-rems
2. U.S. Department of Health and Human Services, Food and Drug Administration. Format and Content of a REMS Document. Guidance for Industry. Accessed January 18, 2023. https://www.fda.gov/media/77846/download
3. U.S. Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS), Clozapine. Accessed January 18, 2023. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=RemsDetails.page&REMS=351
4. The National Association of State Mental Health Program Directors. Clozapine underutilization: addressing the barriers. Accessed September 30, 2019. https://nasmhpd.org/sites/default/files/Assessment%201_Clozapine%20Underutilization.pdf
5. U.S. Food and Drug Administration. FDA is temporarily exercising enforcement discretion with respect to certain clozapine REMS program requirements to ensure continuity of care for patients taking clozapine. Updated November 22, 2022. Accessed June 1, 2023. https://www.fda.gov/drugs/drug-safety-and-availability/fda-temporarily-exercising-enforcement-discretion-respect-certain-clozapine-rems-program
6. Tanzi M. REMS issues affect clozapine, isotretinoin. Pharmacy Today. 2022;28(3):49.
7. U.S. Food and Drug Administration. Coronavirus (COVID-19) update: FDA provides update on patient access to certain REMS drugs during COVID-19 public health emergency. Accessed June 1, 2023. https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-provides-update-patient-access-certain-rems-drugs-during-covid-19
8. U.S. Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS), Spravato (esketamine). Accessed January 18, 2023. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=386
9. U.S. Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS), Zyprexa Relprevv (olanzapine). Accessed January 18, 2023. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=74
10. U.S. Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS). Accessed January 18, 2023. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm
11. Parikh SV, Lopez D, Vande Voort JL, et al. Developing an IV ketamine clinic for treatment-resistant depression: a primer. Psychopharmacol Bull. 2021;51(3):109-124.
12. Dodge D. The ketamine cure. The New York Times. November 4, 2021. Updated November 5, 2021. Accessed June 1, 2023. https://www.nytimes.com/2021/11/04/well/ketamine-therapy-depression.html
13. Burton KW. Time for a national ketamine registry, experts say. Medscape. February 15, 2023. Accessed June 1, 2023. https://www.medscape.com/viewarticle/988310
14. Wilkinson ST, Howard DH, Busch SH. Psychiatric practice patterns and barriers to the adoption of esketamine. JAMA. 2019;322(11):1039-1040. doi:10.1001/jama.2019.10728
15. Wilkinson ST, Toprak M, Turner MS, et al. A survey of the clinical, off-label use of ketamine as a treatment for psychiatric disorders. Am J Psychiatry. 2017;174(7):695-696. doi:10.1176/appi.ajp.2017.17020239
16. Pai SM, Gries JM; ACCP Public Policy Committee. Off-label use of ketamine: a challenging drug treatment delivery model with an inherently unfavorable risk-benefit profile. J Clin Pharmacol. 2022;62(1):10-13. doi:10.1002/jcph.1983
17. Risk Evaluation and Mitigation Strategies (REMS) Integration. Accessed June 1, 2023. https://confluence.hl7.org/display/COD/Risk+Evaluation+and+Mitigation+Strategies+%28REMS%29+Integration
1. U.S. Food and Drug Administration. Risk Evaluation and Mitigation Strategies. Accessed January 18, 2023. https://www.fda.gov/drugs/drug-safety-and-availability/risk-evaluation-and-mitigation-strategies-rems
2. U.S. Department of Health and Human Services, Food and Drug Administration. Format and Content of a REMS Document. Guidance for Industry. Accessed January 18, 2023. https://www.fda.gov/media/77846/download
3. U.S. Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS), Clozapine. Accessed January 18, 2023. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=RemsDetails.page&REMS=351
4. The National Association of State Mental Health Program Directors. Clozapine underutilization: addressing the barriers. Accessed September 30, 2019. https://nasmhpd.org/sites/default/files/Assessment%201_Clozapine%20Underutilization.pdf
5. U.S. Food and Drug Administration. FDA is temporarily exercising enforcement discretion with respect to certain clozapine REMS program requirements to ensure continuity of care for patients taking clozapine. Updated November 22, 2022. Accessed June 1, 2023. https://www.fda.gov/drugs/drug-safety-and-availability/fda-temporarily-exercising-enforcement-discretion-respect-certain-clozapine-rems-program
6. Tanzi M. REMS issues affect clozapine, isotretinoin. Pharmacy Today. 2022;28(3):49.
7. U.S. Food and Drug Administration. Coronavirus (COVID-19) update: FDA provides update on patient access to certain REMS drugs during COVID-19 public health emergency. Accessed June 1, 2023. https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-provides-update-patient-access-certain-rems-drugs-during-covid-19
8. U.S. Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS), Spravato (esketamine). Accessed January 18, 2023. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=386
9. U.S. Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS), Zyprexa Relprevv (olanzapine). Accessed January 18, 2023. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm?event=IndvRemsDetails.page&REMS=74
10. U.S. Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS). Accessed January 18, 2023. https://www.accessdata.fda.gov/scripts/cder/rems/index.cfm
11. Parikh SV, Lopez D, Vande Voort JL, et al. Developing an IV ketamine clinic for treatment-resistant depression: a primer. Psychopharmacol Bull. 2021;51(3):109-124.
12. Dodge D. The ketamine cure. The New York Times. November 4, 2021. Updated November 5, 2021. Accessed June 1, 2023. https://www.nytimes.com/2021/11/04/well/ketamine-therapy-depression.html
13. Burton KW. Time for a national ketamine registry, experts say. Medscape. February 15, 2023. Accessed June 1, 2023. https://www.medscape.com/viewarticle/988310
14. Wilkinson ST, Howard DH, Busch SH. Psychiatric practice patterns and barriers to the adoption of esketamine. JAMA. 2019;322(11):1039-1040. doi:10.1001/jama.2019.10728
15. Wilkinson ST, Toprak M, Turner MS, et al. A survey of the clinical, off-label use of ketamine as a treatment for psychiatric disorders. Am J Psychiatry. 2017;174(7):695-696. doi:10.1176/appi.ajp.2017.17020239
16. Pai SM, Gries JM; ACCP Public Policy Committee. Off-label use of ketamine: a challenging drug treatment delivery model with an inherently unfavorable risk-benefit profile. J Clin Pharmacol. 2022;62(1):10-13. doi:10.1002/jcph.1983
17. Risk Evaluation and Mitigation Strategies (REMS) Integration. Accessed June 1, 2023. https://confluence.hl7.org/display/COD/Risk+Evaluation+and+Mitigation+Strategies+%28REMS%29+Integration
Interventional psychiatry (Part 2)
While most psychiatric treatments have traditionally consisted of pharmacotherapy with oral medications, a better understanding of the pathophysiology underlying many mental illnesses has led to the recent increased use of treatments that require specialized administration and the creation of a subspecialty called interventional psychiatry. In Part 1 of this 2-part article (“Interventional psychiatry [Part 1],"
Neuromodulation treatments
Neuromodulation—the alteration of nerve activity through targeted delivery of a stimulus, such as electrical stimulation, to specific neurologic sites—is an increasingly common approach to treating a variety of psychiatric conditions. The use of some form of neuromodulation as a medical treatment has a long history (Box1-6). Modern electric neuromodulation began in the 1930s with electroconvulsive therapy (ECT). The 1960s saw the introduction of deep brain stimulation (DBS), spinal cord stimulation, and later, vagus nerve stimulation (VNS). Target-specific noninvasive brain stimulation became possible with transcranial magnetic stimulation (TMS). These approaches are used for treating major depressive disorder (MDD), obsessive-compulsive disorder (OCD), anxiety disorders, and insomnia. Nearly all these neuromodulatory approaches require clinicians to undergo special training and patients to participate in an invasive procedure. These factors also increase cost. Nonetheless, the high rates of success of some of these approaches have led to relatively rapid and widespread acceptance.
Box
The depth and breadth of human anatomical knowledge has evolved over millennia. The time frame “thousands of years” may appear to be an overstatement, but evidence exists for successful therapeutic limb amputation as early as 31,000 years ago.1 This suggests that human knowledge of bone, muscle, and blood supply was developed much earlier than initially believed. Early Homo sapiens were altering the body—regulating or adjusting it— to serve a purpose; in this case, the purpose was survival.
In 46 AD, electrical modulation was introduced by Scribonius Largus, a physician in court of the emperor Tiberius, who used “torpedoes” (most likely electric eels) to treat headaches and pain from arthritis. Loosely, these early clinicians were modulating human function.
In the late 1800s, electrotherapeutics was a growing branch of medicine, with its own national organization—the American ElectroTherapeutic Association.2 In that era, electricity was novel, powerful, and seen as “the future.” Because such novel therapeutics were offered by both mainstream and dubious sources,3 “many of these products were marketed with the promise of curing everything from cancer to headaches.”4
Modern electric neuromodulation began in the 1930s with electroconvulsive therapy,5 followed by deep brain stimulation and spinal cord stimulation in the 1960s. Target-specific noninvasive brain stimulation became possible when Anthony Barker’s team developed the first device that permitted transcranial magnetic stimulation in 1985.6
Electroconvulsive therapy
In ECT, electric current is applied to the brain to induce a self-limiting seizure. It is the oldest and best-known interventional psychiatric treatment. ECT can also be considered one of the first treatments specifically developed to address pathophysiologic changes. In 1934, Ladislas J. Meduna, who had observed in neuropathologic studies that microglia were more numerous in patients with epilepsy compared with patients with schizophrenia, injected a patient who had been hospitalized with catatonia for 4 years with camphor, a proconvulsant.7 After 5 seizures, the patient began to recover. The therapeutic use of electricity was subsequently developed and optimized in animal models, and first used on human patients in Italy in 1939 and in the United States in 1940.8 The link between psychiatric illness and microglia, which was initially observed nearly a century ago, is making a comeback, as excessive microglial activation has been demonstrated in animal and human models of depression.9
Administering ECT requires specialized equipment, anesthesia, physician training, and nursing observation. ECT also has a negative public image.10 All of these factors conspire to reduce the availability of ECT. Despite this, approximately 100,000 patients in the United States and >1 million worldwide receive ECT each year.10 Patients generally require 6 to 12 ECT treatments11 to achieve sufficient response and may require additional maintenance treatments.12
Although ECT is used to treat psychiatric illnesses ranging from mood disorders to psychotic disorders and catatonia, it is mainly employed to treat people with severe treatment-resistant depression (TRD).13 ECT is associated with significant improvements in depressive symptoms and improvements in quality of life.14 It is superior to other treatments for TRD, such as ketamine,15 though a recent study did not show IV ketamine inferiority.16 ECT is also used to treat other neuropsychiatric disorders, such as Parkinson disease.17
Clinicians have explored alternate methods of inducing therapeutic seizures. Magnetic seizure therapy (MST) utilizes a modified magnetic stimulation device to deliver a higher energy in such a way to induce a generalized seizure under anesthesia.18 While patients receiving MST generally experience fewer adverse effects than with ECT, the procedure may be equal to19 or less effective than ECT.20
Transcranial magnetic stimulation
In neuroimaging research, certain aberrant brain circuits have been implicated in the pathogenesis of depression.21 Specifically, anatomical and functional imaging suggests connections in the prefrontal cortex are involved in the depression process. In TMS, a series of magnetic pulses are administered via the scalp to stimulate neurons in areas of the brain associated with MDD. Early case reports on using TMS to stimulate the prefrontal cortex found significant improvement of symptoms in patients with depression.22 These promising results spurred great interest in the procedure. Over time, the dose and duration of stimulation has increased, along with FDA-approved indications. TMS was first FDA-approved for TRD.23 Although the primary endpoint of the initial clinical trial did not meet criteria for FDA approval, TMS did result in improvement across multiple other measures of depression.23 After the FDA approved the first TMS device, numerous companies began to produce TMS technology. Most of these companies manufacture devices with the figure-of-eight coil, with 1 company producing the Hesed-coil helmet.24
Continue to: An unintended outcome...
An unintended outcome of the increased interest in TMS has been an increased understanding of brain regions involved in psychiatric illness. TMS was able to bring knowledge of mental health from synapses to circuits.25 Work in this area has further stratified the circuits involved in the manifestation of symptom clusters in depression.26 The exact taxonomy of these brain circuits has not been fully realized, but the default mode, salience, attention, cognitive control, and other circuits have been shown to be involved in specific symptom presentations.26,27 These circuits can be hyperactive, hypoactive, hyperconnected, or hypoconnected, with the aberrancies compared to normal controls resulting in symptoms of psychiatric illness.28
This enhanced understanding of brain function has led to further research and development of protocols and subsequent FDA approval of TMS for OCD, anxious depression, and smoking cessation.29 In addition, it has allowed for a proliferation of off-label uses for TMS, including (but not limited to) tinnitus, pain, migraines, and various substance use disorders.30 TMS treatment for these conditions involves stimulation of specific anatomical brain regions that are thought to play a role in the pathology of the target disorder. For example, subthreshold stimulation of the motor cortex has shown some utility in managing symptoms of pain disorders and movement disorders,31,32 the ventromedial prefrontal cortex has been implicated in disorders in the OCD spectrum,33 stimulation of the frontal poles may help treat substance use disorders,34 and the auditory cortex has been a target for treating tinnitus and auditory hallucinations.35
The location of stimulation for treating depression has evolved. The Talairach-Tournoux coordinate system has been used to determine the location of the dorsolateral prefrontal cortex (DLPFC) in relation to the motor cortex. This was measured to be 5 cm from the motor hotspot and subsequently became “the 5.5 cm rule,” taking skull convexity into account. The treatment paradigm for the Hesed coil also uses a measurement from the motor hotspot. Another commonly used methodology for coil placement involves using the 10 to 20 EEG coordinate system to individualize scalp landmarks. In this method, the F3 location corresponds most accurately to the DLPFC target. More recently, using fMRI-guided navigation for coil placement has been shown to lead to a significant reduction in depressive symptoms.36
For depression, the initial recommended course of treatment is 6 weeks, but most improvement is seen in the first 2 to 3 weeks.14 Therefore, many clinicians administer an initial course of 3 weeks unless the response is inadequate, in which case a 6-week course is administered. Many patients require ongoing maintenance treatment, which can be weekly or monthly based on response.37
Research to determine the optimal TMS dose for treating neuropsychiatric symptoms is ongoing. Location, intensity of stimulation, and pulse are the components of stimulation. The pulse can be subdivided into frequency, pattern (single pulse, standard, burst), train (numbers of pulse groups), interval between trains, and total number of pulses per session. The Clinical TMS Society has published TMS protocols.38 The standard intensity of stimulation is 120% of the motor threshold (MT), which is defined as the amount of stimulation over the motor cortex required to produce movement in the extensor hallucis longus. Although treatment for depression traditionally utilizes rapid TMS (3,000 pulses delivered per session at a frequency of 10 Hz in 4-second trains), in controlled studies, accelerated protocols such as intermittent theta burst stimulation (iTBS; standard stimulation parameters: triplet 50 Hz bursts at 5 Hz, with an interval of 8 seconds for 600 pulses per session) have shown noninferiority.36,39
Recent research has explored fMRI-guided iTBS in an even more accelerated format. The Stanford Neuromodulation Therapy trial involved 1,800 pulses per session for 10 sessions a day for 5 days at 90% MT.36 This treatment paradigm was shown to be more effective than standard protocols and was FDA-approved in 2022. Although this specific iTBS protocol exhibited encouraging results, the need for fMRI for adequate delivery might limit its use.
Continue to: Transcranial direct current stimulation
Transcranial direct current stimulation
Therapeutic noninvasive brain stimulation technology is plausible due to the relative lack of adverse effects and ease of administration. In transcranial direct current stimulation (tDCS), a low-intensity, constant electric current is delivered to stimulate the brain via electrodes attached to the scalp. tDCS modulates spontaneous neuronal network activity40,41 and induces polarization of resting membrane potential at the neuronal level,42 though the exact mechanism is yet to be proven. N-methyl-
tDCS has been suggested as a treatment for various psychiatric and medical conditions. However, the small sample sizes and experimental design of published studies have limited tDCS from being clinically recommended.30 No recommendation of Level A (definite efficacy) for its use was found for any indication. Level B recommendation (probable efficacy) was proposed for fibromyalgia, MDD episode without drug resistance, and addiction/craving. Level C recommendation (possible efficacy) is proposed for chronic lower limb neuropathic pain secondary to spinal cord lesion. tDCS was found to be probably ineffective as a treatment for tinnitus and drug-resistant MDD.30 Some research has suggested that tDCS targeting the DLPFC is associated with cognitive improvements in healthy individuals as well as those with schizophrenia.44 tDCS treatment remains experimental and investigational.
Deep brain stimulation
DBS is a neurosurgical procedure that uses electrical current to directly modulate specific areas of the CNS. In terms of accurate, site-specific anatomical targeting, there can be little doubt of the superiority of DBS. DBS involves the placement of leads into the brain parenchyma. Image guidance techniques are used for accurate placement. DBS is a mainstay for the symptomatic treatment of treatment-resistant movement disorders such as Parkinson disease, essential tremor, and some dystonic disorders. It also has been studied as a potential treatment for chronic pain, cluster headache, Huntington disease, and Tourette syndrome.
For treating depression, researched targets include the subgenual cingulate gyrus (SCG), ventral striatum, nucleus accumbens, inferior thalamic peduncle, medial forebrain bundle, and the red nucleus.45 In systematic reviews, improvement of depression is greatest when DBS targets the subgenual cingulate cortex and the medial forebrain bundle.46
The major limitation of DBS for treating depression is the invasive nature of the procedure. Deep TMS can achieve noninvasive stimulation of the SCG and may be associated with fewer risks, fewer adverse events, and less collateral damage. However, given the evolving concept of abnormal neurologic circuits in depression, as our understanding of circuitry in pathological psychiatric processes increases, DBS may be an attractive option for personalized targeting of symptoms in some patients.
DBS may also be beneficial for severe, treatment-resistant OCD. Electrode implantation in the region of the internal capsule/ventral striatum, including the nucleus accumbens, is used47; there is little difference in placement as a treatment for OCD vs for movement disorders.48
Continue to: A critical review of 23 trials...
A critical review of 23 trials and case reports of DBS as a treatment for OCD demonstrated a 47.7% mean reduction in score on the Yale-Brown Obsessive-Compulsive Scale (Y-BOCS) and a mean response percentage (minimum 35% Y-BOCS reduction) of 58.2%.49 Most patients regained a normal quality of life after DBS.49 A more rigorous review of 15 meta-analyses of DBS found that conclusions about its efficacy or comparative effectiveness cannot be drawn.50 Because of the nature of neurosurgery, DBS has many potential complications, including cognitive changes, headache, infection, seizures, stroke, and hardware failure.
Vagus nerve stimulation
VNS, in which an implanted device stimulates the left vagus nerve with electrical impulses, was FDA-approved for treating chronic TRD in 2005.51 It had been approved for treatment-resistant epilepsy in 1997. In patients with epilepsy, VNS was shown to improve mood independent of seizure control.52 VNS requires a battery-powered pacemaker device to be implanted under the skin over the anterior chest wall, and a wire tunneled to an electrode is wrapped around the left vagus nerve in the neck.53 The pacemaker is then programmed, monitored, and reprogrammed to optimize response.
VNS is believed to stimulate deep brain nuclei that may play a role in depression.54 The onset of improvement is slow (it may take many months) but in carefully selected patients VNS can provide significant control of TRD. In addition to rare surgery-related complications such as a trauma to the vagal nerve and surrounding tissues (vocal cord paralysis, implant site infection, left facial nerve paralysis and Horner syndrome), VNS may cause hoarseness, dyspnea, and cough related to the intensity of the current output.51 Hypomania and mania were also reported; no suicidal behavior has been associated with VNS.51
Noninvasive vagus nerve stimulationIn noninvasive vagus nerve stimulation (nVNS) or transcutaneous VNS, an external handheld device is applied to the neck overlying the course of the vagus nerve to deliver a sinusoidal alternating current.55 nVNS is currently FDA-approved for treating migraine headaches.55,56 It has demonstrated actions on neurophysiology57 and inflammation in patients with MDD.58 Exploratory research has found a small beneficial effect in patients with depression.59,60 A lack of adequate reproducibility prevents this treatment from being more widely recommended, although attempts to standardize the field are evolving.61
Cranial electrical stimulation
Cranial electrical stimulation (CES) is an older form of electric stimulation developed in the 1970s. In CES, mild electrical pulses are delivered to the ear lobes bilaterally in an episodic fashion (usually 20 to 60 minutes once or twice daily). While CES can be considered a form of neuromodulation, it is not strictly interventional. Patients self-administer CES. The procedure has minimal effects on improving sleep, anxiety, and mood.62-66 Potential adverse effects include a tingling sensation in the ear lobes, lightheadedness, and fogginess. A review and meta-analysis of CES for treating addiction by Kirsch67 showed a wide range of symptoms responding positively to CES treatment, although this study was not peer-reviewed. Because of the low quality of nearly all research that evaluated CES, this form of electric stimulation cannot be viewed as an accepted treatment for any of its listed indications.
Continue to: Other neuromodulation techniques
Other
In addition to the forms of neuromodulation we have already described, there are many other techniques. Several are promising but not yet ready for clinical use. Table 1 and Table 2 summarize the neuromodulation techniques described in this article as well as several that are under development.

Acupuncture
Acupuncture is a Chinese form of medical treatment that began >3,000 years ago; there are written descriptions of it from >2,000 years ago.68 It is based on the belief that there are channels within the body through which the Qi (vital energy or life force) flow, and that inserting fine needles into these channels via the skin can rebalance Qi.68 Modern mechanistic hypotheses invoke involvement of inflammatory or pain pathways.69 Acupuncture frequently uses electric stimulation (electro-acupuncture) to increase the potency of the procedure. Alternatively, in a related procedure (acupressure), pressure can replace the needle. Accreditation in acupuncture generally requires a master’s degree in traditional Chinese medicine but does not require any specific medical training. Acupuncture training courses for physicians are widely available.
All forms of acupuncture are experimental for a wide variety of mental and medical conditions. A meta-analysis found that most research of the utility of acupuncture for depression suffered from various forms of potential bias and was considered low quality.70 Nonetheless, active acupuncture was shown to be minimally superior to placebo acupuncture.70 A meta-analysis of acupuncture for preoperative anxiety71,72 and poststroke insomnia73 reported a similar low study quality. A study of 72 patients with primary insomnia revealed that acupuncture was more effective than sham acupuncture for most sleep measures.74
Psychiatry is increasingly integrating medical tools in addition to psychological tools. Pharmacology remains a cornerstone of biological psychiatry and this will not soon change. However, nonpharmacologic psychiatric treatments such as therapeutic neuromodulation are rapidly emerging. These and novel methods of medication administration may present a challenge to psychiatrists who do not have access to medical personnel or may have forgotten general medical skills.
Our 2-part article has highlighted several interventional psychiatry tools—old and new—that may interest clinicians and benefit patients. As a rule, such treatments are reserved for the most treatment-resistant, challenging psychiatric patients, those with hard-to-treat chronic conditions, and patients who are not helped by more commonly used treatments. An additional complication is that such treatments are frequently not appropriately researched, vetted, or FDA-approved, and therefore are higher risk. Appropriate clinical judgment is always necessary, and potential benefits must be thoroughly weighed against possible adverse effects.
Bottom Line
Several forms of neuromodulation, including electroconvulsive therapy, transcranial magnetic stimulation, transcranial direct current stimulation, deep brain stimulation, and vagus nerve stimulation, may be beneficial for patients with certain treatment-resistant psychiatric disorders, including major depressive disorder and obsessive-compulsive disorder.
Related Resources
- Janicak PG. What’s new in transcranial magnetic stimulation. Current Psychiatry. 2019;18(3):10-16.
- Sharma MS, Ang-Rabanes M, Selek S, et al. Neuromodulatory options for treatment-resistant depression. Current Psychiatry. 2018;17(3):26-28,33-37.
1. Maloney TR, Dilkes-Hall IE, Vlok M, et al. Surgical amputation of a limb 31,000 years ago in Borneo. Nature. 2022;609(7927):547-551. doi:10.1038/s41586-022-05160-8
2. The American Electro-Therapeutic Association. JAMA. 1893;21(14):500. doi:10.1001/jama.1893.02420660030004
3. The American Electro-Therapeutic Association. JAMA. 1894;23(15):590-591. doi:10.1001/jama.1894.02421200024006
4. Wexler A. The medical battery in the United States (1870-1920): electrotherapy at home and in the clinic. J Hist Med Allied Sci. 2017;72(2):166-192. doi:10.1093/jhmas/jrx001
5. Gazdag G, Ungvari GS. Electroconvulsive therapy: 80 years old and still going strong. World J Psychiatry. 2019;9(1):1-6. doi:10.5498/wjp.v9.i1.1
6. Barker AT, Jalinous R, Freeston IL. Non-invasive magnetic stimulation of human motor cortex. Lancet. 1985;1(8437):1106-1107. doi:10.1016/s0140-6736(85)92413-4
7. Fink M. Historical article: autobiography of L. J. Meduna. Convuls Ther. 1985;1(1):43-57.
8. Suleman R. A brief history of electroconvulsive therapy. Am J Psychiatry. 2020;16(1):6. doi:10.1176/appi.ajp-rj.2020.160103
9. Ménard C, Hodes GE, Russo SJ. Pathogenesis of depression: insights from human and rodent studies. Neuroscience. 2016;321:138-162. doi:10.1016/j.neuroscience.2015.05.053
10. Payne NA, Prudic J. Electroconvulsive therapy: part II: a biopsychosocial perspective. J Psychiatr Pract. 2009;15(5):369-390. doi:10.1097/01.pra.0000361278.73092.85
11. Tirmizi O, Raza A, Trevino K, et al. Electroconvulsive therapy: how modern techniques improve patient outcomes. Current Psychiatry. 2012;11(10):24-46.
12. Kolar D. Current status of electroconvulsive therapy for mood disorders: a clinical review. Evid Based Ment Health. 2017;20(1):12-14. doi:10.1136/eb-2016-102498
13. Andrade C. Active placebo, the parachute meta-analysis, the Nobel Prize, and the efficacy of electroconvulsive therapy. J Clin Psychiatry. 2021;82(2):21f13992. doi:10.4088/JCP.21f13992
14. Giacobbe P, Rakita U, Penner-Goeke K, et al. Improvements in health-related quality of life with electroconvulsive therapy: a meta-analysis. J ECT. 2018;34(2):87-94. doi:10.1097/YCT.0000000000000486
15. Rhee TG, Shim SR, Forester BP, et al. Efficacy and safety of ketamine vs electroconvulsive therapy among patients with major depressive episode: a systematic review and meta-analysis. JAMA Psychiatry. 2022;79(12):1162-1172. doi:10.1001/jamapsychiatry.2022.3352
16. Anand A, Mathew SJ, Sanacora G, et al. Ketamine versus ECT for nonpsychotic treatment-resistant major depression. N Engl J Med. 2023. doi: 10.1056/NEJMoa2302399
17. Takamiya A, Seki M, Kudo S, et al. Electroconvulsive therapy for Parkinson’s disease: a systematic review and meta-analysis. Mov Disord. 2021;36(1):50-58. doi:10.1002/mds.28335
18. Singh R, Sharma R, Prakash J, et al. Magnetic seizure therapy. Ind Psychiatry J. 2021;30(Suppl 1):S320-S321. doi:10.4103/0972-6748.328841
19. Chen M, Yang X, Liu C, et al. Comparative efficacy and cognitive function of magnetic seizure therapy vs. electroconvulsive therapy for major depressive disorder: a systematic review and meta-analysis. Transl Psychiatry. 2021;11(1):437. doi:10.1038/s41398-021-01560-y
20. Cretaz E, Brunoni AR, Lafer B. Magnetic seizure therapy for unipolar and bipolar depression: a systematic review. Neural Plast. 2015;2015:521398. doi:10.1155/2015/521398
21. George MS, Ketter TA, Post RM. Prefrontal cortex dysfunction in clinical depression. In: Nemeroff CB, Weiss JM, Schatzberg AF, et al, eds. Depression. 2nd ed. Wiley Online Library; 1994:59-72. https://doi.org/10.1002/depr.3050020202
22. George MS, Wassermann EM, Williams WA, et al. Daily repetitive transcranial magnetic stimulation (rTMS) improves mood in depression. Neuroreport. 1995;6(14):1853-1856.
23. O’Reardon JP, Solvason HB, Janicak PG, et al. Efficacy and safety of transcranial magnetic stimulation in the acute treatment of major depression: a multisite randomized controlled trial. Biol Psychiatry. 2007;62(11):1208-1216.
24. Clinical TMS Society. TMS devices. Accessed January 2, 2023. https://www.clinicaltmssociety.org/devices
25. Goldstein-Piekarski AN, Ball TM, Samara Z, et al. Mapping neural circuit biotypes to symptoms and behavioral dimensions of depression and anxiety. Biol Psychiatry. 2022;91(6):561-571. doi:10.1016/j.biopsych.2021.06.024
26. Siddiqi SH, Taylor SF, Cooke D, et al. Distinct symptom-specific treatment targets for circuit-based neuromodulation. Am J Psychiatry. 2020;177(5):435-446. doi:10.1176/appi.ajp.2019.19090915
27. Williams LM. Defining biotypes for depression and anxiety based on large-scale circuit dysfunction: a theoretical review of the evidence and future directions for clinical translation. Depress Anxiety. 2017;34(1):9-24. doi:10.1002/da.22556
28. Drysdale AT, Grosenick L, Downar J, et al. Resting-state connectivity biomarkers define neurophysiological subtypes of depression. Nat Med. 2017;23(1):28-38. doi:10.1038/nm.4246
29. Cohen SL, Bikson M, Badran BW, et al. A visual and narrative timeline of US FDA milestones for transcranial magnetic stimulation (TMS) devices. Brain Stimul. 2022;15(1):73-75. doi:10.1016/j.brs.2021.11.010
30. Lefaucheur JP, Antal A, Ayache SS, et al. Evidence-based guidelines on the therapeutic use of transcranial direct current stimulation (tDCS). Clin Neurophysiol. 2017;128(1):56-92. doi:10.1016/j.clinph.2016.10.087
31. Li R, He Y, Qin W, et al. Effects of repetitive transcranial magnetic stimulation on motor symptoms in Parkinson’s disease: a meta-analysis. Neurorehabil Neural Repair. 2022;36(7):395-404. doi:10.1177/15459683221095034
32. Leung A, Shirvalkar P, Chen R, et al. Transcranial magnetic stimulation for pain, headache, and comorbid depression: INS-NANS expert consensus panel review and recommendation. Neuromodulation. 2020;23(3):267-290. doi:10.1111/ner.13094
33. Carmi L, Tendler A, Bystritsky A, et al. Efficacy and safety of deep transcranial magnetic stimulation for obsessive-compulsive disorder: a prospective multicenter randomized double-blind placebo-controlled trial. Am J Psychiatry. 2019;176(11):931-938. doi:10.1176/appi.ajp.2019.18101180
34. Harel M, Perini I, Kämpe R, et al. Repetitive transcranial magnetic stimulation in alcohol dependence: a randomized, double-blind, sham-controlled proof-of-concept trial targeting the medial prefrontal and anterior cingulate cortices. Biol Psychiatry. 2022;91(12):1061-1069. doi:10.1016/j.biopsych.2021.11.020
35. Folmer RL, Theodoroff SM, Casiana L, et al. Repetitive transcranial magnetic stimulation treatment for chronic tinnitus: a randomized clinical trial. JAMA Otolaryngol Head Neck Surg. 2015;141(8):716-722. doi:10.1001/jamaoto.2015.1219
36. Cole EJ, Phillips AL, Bentzley BS, et al. Stanford Neuromodulation Therapy (SNT): a double-blind randomized controlled trial. Am J Psychiatry. 2022;179(2):132-141. doi:10.1176/appi.ajp.2021.20101429
37. Wilson S, Croarkin PE, Aaronson ST, et al. Systematic review of preservation TMS that includes continuation, maintenance, relapse-prevention, and rescue TMS. J Affect Disord. 2022;296:79-88. doi:10.1016/j.jad.2021.09.040
38. Perera T, George MS, Grammer G, et al. The Clinical TMS Society consensus review and treatment recommendations for TMS therapy for major depressive disorder. Brain Stimul. 2016;9(3):336-346. doi:10.1016/j.brs.2016.03.010
39. Blumberger DM, Vila-Rodriguez F, Thorpe KE, et al. Effectiveness of theta burst versus high-frequency repetitive transcranial magnetic stimulation in patients with depression (THREE-D): a randomized non-inferiority trial. Lancet. 2018;391(10131):1683-1692. doi:10.1016/S0140-6736(18)30295-2
40. Nitsche MA, Cohen LG, Wassermann EM, et al. Transcranial direct current stimulation: state of the art 2008. Brain Stimul. 2008;1(3):206-223. doi:10.1016/j.brs.2008.06.004
41. Priori A, Hallett M, Rothwell JC. Repetitive transcranial magnetic stimulation or transcranial direct current stimulation? Brain Stimul. 2009;2(4):241-245.
42. Priori A, Berardelli A, Rona S, et al. Polarization of the human motor cortex through the scalp. Neuroreport. 1998;9(10):2257-2260. doi:10.1097/00001756-199807130-00020
43. Nitsche MA, Liebetanz D, Antal A, et al. Modulation of cortical excitability by weak direct current stimulation-- technical, safety and functional aspects. Suppl Clin Neurophysiol. 2003;56:255-276. doi:10.1016/s1567-424x(09)70230-2
44. Agarwal SM, Venkataram Shivakumar V, et al. Transcranial direct current stimulation in schizophrenia. Clin Psychopharmacol Neurosci. 2013;11(3):118-125.
45. Drobisz D, Damborská A. Deep brain stimulation targets for treating depression. Behav Brain Res. 2019;359:266-273. doi:10.1016/j.bbr.2018.11.004
46. Kisely S, Li A, Warren N, et al. A systematic review and meta-analysis of deep brain stimulation for depression. Depress Anxiety. 2018;35(5):468-480. doi:10.1002/da.22746
47. Blomstedt P, Sjöberg RL, Hansson M, et al. Deep brain stimulation in the treatment of obsessive-compulsive disorder. World Neurosurg. 2013;80(6):e245-e253. doi:10.1016/j.wneu.2012.10.006
48. Denys D, Mantione M, Figee M, et al. Deep brain stimulation of the nucleus accumbens for treatment-refractory obsessive-compulsive disorder. Arch Gen Psychiatry. 2010;67(10):1061-1068. doi:10.1001/archgenpsychiatry.2010.122
49. van Westen M, Rietveld E, Figee M, et al. Clinical outcome and mechanisms of deep brain stimulation for obsessive-compulsive disorder. Curr Behav Neurosci Rep. 2015;2(2):41-48. doi:10.1007/s40473-015-0036-3
50. Papageorgiou PN, Deschner J, Papageorgiou SN. Effectiveness and adverse effects of deep brain stimulation: umbrella review of meta-analyses. J Neurol Surg A Cent Eur Neurosurg. 2017;78(2):180-190. doi:10.1055/s-0036-1592158
51. O’Reardon JP, Cristancho P, Peshek AD. Vagus nerve stimulation (VNS) and treatment of depression: to the brainstem and beyond. Psychiatry (Edgmont). 2006;3(5):54-63.
52. Harden CL, Pulver MC, Ravdin LD, et al. A pilot study of mood in epilepsy patients treated with vagus nerve stimulation. Epilepsy Behav. 2000;1(2):93-99. doi:10.1006/ebeh.2000.0046
53. Giordano F, Zicca A, Barba C, et al. Vagus nerve stimulation: surgical technique of implantation and revision and related morbidity. Epilepsia. 2017;58(S1):85-90. doi:10.1111/epi.13687
54. George MS, Nahas Z, Bohning DE, et al. Mechanisms of action of vagus nerve stimulation (VNS). Clin Neurosci Res. 2004;4(1-2):71-79.
55. Nesbitt AD, Marin JCA, Tompkins E, et al. Initial use of a novel noninvasive vagus nerve stimulator for cluster headache treatment. Neurology. 2015;84:1249-1253. doi:10.1212/WNL.0000000000001394
56. Goadsby PJ, Grosberg BM, Mauskop A, et al. Effect of noninvasive vagus nerve stimulation on acute migraine: an open-label pilot study. Cephalalgia. 2014;34:986-993. doi:10.1177/0333102414524494
57. Fang J, Rong P, Hong Y, et al. Transcutaneous vagus nerve stimulation modulates default mode network in major depressive disorder. Biol Psychiatry. 2016;79(4):266-273. doi:10.1016/j.biopsych.2015.03.025
58. Liu CH, Yang MH, Zhang GZ, et al. Neural networks and the anti-inflammatory effect of transcutaneous auricular vagus nerve stimulation in depression. J Neuroinflammation. 2020;17(1):54. doi:10.1186/s12974-020-01732-5
59. Hein E, Nowak M, Kiess O, et al. Auricular transcutaneous electrical nerve stimulation in depressed patients: a randomized controlled pilot study. J Neural Transm (Vienna). 2013;120(5):821-827. doi:10.1007/s00702-012-0908-6
60. Rong P, Liu J, Wang L, et al. Effect of transcutaneous auricular vagus nerve stimulation on major depressive disorder: a nonrandomized controlled pilot study. J Affect Disord. 2016;195:172-179. doi:10.1016/j.jad.2016.02.031
61. Farmer AD, Strzelczyk A, Finisguerra A, et al. International consensus based review and recommendations for minimum reporting standards in research on transcutaneous vagus nerve stimulation (Version 2020). Front Hum Neurosci. 2021;14:568051. doi:10.3389/fnhum.2020.568051
62. Amr M, El-Wasify M, Elmaadawi AZ, et al. Cranial electrotherapy stimulation for the treatment of chronically symptomatic bipolar patients. J ECT. 2013;29(2):e31-e32. doi:10.1097/YCT.0b013e31828a344d
63. Kirsch DL, Nichols F. Cranial electrotherapy stimulation for treatment of anxiety, depression, and insomnia. Psychiatr Clin North Am. 2013;36(1):169-176. doi:10.1016/j.psc.2013.01.006
64. Lande RG, Gragnani C. Efficacy of cranial electric stimulation for the treatment of insomnia: a randomized pilot study. Complement Ther Med. 2013;21(1):8-13. doi:10.1016/j.ctim.2012.11.007
65. Ou Y, Li, C. Sertraline combined alpha-stim clinical observations on the treatment of 30 cases of generalized anxiety disorder. Chinese Journal of Ethnomedicine and Ethnopharmacy. 2015;24(17):73-75.
66. Price L, Briley J, Haltiwanger S, et al. A meta-analysis of cranial electrotherapy stimulation in the treatment of depression. J Psychiatr Res. 2021;135:119-134. doi:10.1016/j.jpsychires.2020.12.043
67. Kirsch D, Gilula M. CES in the treatment of addictions: a review and meta-analysis. Pract Pain Manag. 2007;7(9).
68. Hao JJ, Mittelman M. Acupuncture: past, present, and future. Glob Adv Health Med. 2014;3(4):6-8. doi:10.7453/gahmj.2014.042
69. Napadow V, Ahn A, Longhurst J, et al. The status and future of acupuncture mechanism research. J Altern Complement Med. 2008;14(7):861-869. doi:10.1089/acm.2008.SAR-3
70. Smith CA, Armour M, Lee MS, et al. Acupuncture for depression. Cochrane Database Syst Rev. 2018;3(3):CD004046. doi:10.1002/14651858.CD004046.pub4
71. Tong QY, Liu R, Zhang K, et al. Can acupuncture therapy reduce preoperative anxiety? A systematic review and meta-analysis. J Integr Med. 2021;19(1):20-28. doi:10.1016/j.joim.2020.10.007
72. Usichenko TI, Hua K, Cummings M, et al. Auricular stimulation for preoperative anxiety – a systematic review and meta-analysis of randomized controlled clinical trials. J Clin Anesth. 2022;76:110581. doi:10.1016/j.jclinane.2021.110581
73. Zhou L, Hu X, Yu Z, et al. Efficacy and safety of acupuncture in the treatment of poststroke insomnia: a systematic review and meta-analysis of twenty-six randomized controlled trials. Evid Based Complement Alternat Med. 2022;2022:5188311. doi:10.1155/2022/5188311
74. Yin X, Gou M, Xu J, et al. Efficacy and safety of acupuncture treatment on primary insomnia: a randomized controlled trial. Sleep Med. 2017;37:193-200. doi:10.1016/j.sleep.2017.02.012
While most psychiatric treatments have traditionally consisted of pharmacotherapy with oral medications, a better understanding of the pathophysiology underlying many mental illnesses has led to the recent increased use of treatments that require specialized administration and the creation of a subspecialty called interventional psychiatry. In Part 1 of this 2-part article (“Interventional psychiatry [Part 1],"
Neuromodulation treatments
Neuromodulation—the alteration of nerve activity through targeted delivery of a stimulus, such as electrical stimulation, to specific neurologic sites—is an increasingly common approach to treating a variety of psychiatric conditions. The use of some form of neuromodulation as a medical treatment has a long history (Box1-6). Modern electric neuromodulation began in the 1930s with electroconvulsive therapy (ECT). The 1960s saw the introduction of deep brain stimulation (DBS), spinal cord stimulation, and later, vagus nerve stimulation (VNS). Target-specific noninvasive brain stimulation became possible with transcranial magnetic stimulation (TMS). These approaches are used for treating major depressive disorder (MDD), obsessive-compulsive disorder (OCD), anxiety disorders, and insomnia. Nearly all these neuromodulatory approaches require clinicians to undergo special training and patients to participate in an invasive procedure. These factors also increase cost. Nonetheless, the high rates of success of some of these approaches have led to relatively rapid and widespread acceptance.
Box
The depth and breadth of human anatomical knowledge has evolved over millennia. The time frame “thousands of years” may appear to be an overstatement, but evidence exists for successful therapeutic limb amputation as early as 31,000 years ago.1 This suggests that human knowledge of bone, muscle, and blood supply was developed much earlier than initially believed. Early Homo sapiens were altering the body—regulating or adjusting it— to serve a purpose; in this case, the purpose was survival.
In 46 AD, electrical modulation was introduced by Scribonius Largus, a physician in court of the emperor Tiberius, who used “torpedoes” (most likely electric eels) to treat headaches and pain from arthritis. Loosely, these early clinicians were modulating human function.
In the late 1800s, electrotherapeutics was a growing branch of medicine, with its own national organization—the American ElectroTherapeutic Association.2 In that era, electricity was novel, powerful, and seen as “the future.” Because such novel therapeutics were offered by both mainstream and dubious sources,3 “many of these products were marketed with the promise of curing everything from cancer to headaches.”4
Modern electric neuromodulation began in the 1930s with electroconvulsive therapy,5 followed by deep brain stimulation and spinal cord stimulation in the 1960s. Target-specific noninvasive brain stimulation became possible when Anthony Barker’s team developed the first device that permitted transcranial magnetic stimulation in 1985.6
Electroconvulsive therapy
In ECT, electric current is applied to the brain to induce a self-limiting seizure. It is the oldest and best-known interventional psychiatric treatment. ECT can also be considered one of the first treatments specifically developed to address pathophysiologic changes. In 1934, Ladislas J. Meduna, who had observed in neuropathologic studies that microglia were more numerous in patients with epilepsy compared with patients with schizophrenia, injected a patient who had been hospitalized with catatonia for 4 years with camphor, a proconvulsant.7 After 5 seizures, the patient began to recover. The therapeutic use of electricity was subsequently developed and optimized in animal models, and first used on human patients in Italy in 1939 and in the United States in 1940.8 The link between psychiatric illness and microglia, which was initially observed nearly a century ago, is making a comeback, as excessive microglial activation has been demonstrated in animal and human models of depression.9
Administering ECT requires specialized equipment, anesthesia, physician training, and nursing observation. ECT also has a negative public image.10 All of these factors conspire to reduce the availability of ECT. Despite this, approximately 100,000 patients in the United States and >1 million worldwide receive ECT each year.10 Patients generally require 6 to 12 ECT treatments11 to achieve sufficient response and may require additional maintenance treatments.12
Although ECT is used to treat psychiatric illnesses ranging from mood disorders to psychotic disorders and catatonia, it is mainly employed to treat people with severe treatment-resistant depression (TRD).13 ECT is associated with significant improvements in depressive symptoms and improvements in quality of life.14 It is superior to other treatments for TRD, such as ketamine,15 though a recent study did not show IV ketamine inferiority.16 ECT is also used to treat other neuropsychiatric disorders, such as Parkinson disease.17
Clinicians have explored alternate methods of inducing therapeutic seizures. Magnetic seizure therapy (MST) utilizes a modified magnetic stimulation device to deliver a higher energy in such a way to induce a generalized seizure under anesthesia.18 While patients receiving MST generally experience fewer adverse effects than with ECT, the procedure may be equal to19 or less effective than ECT.20
Transcranial magnetic stimulation
In neuroimaging research, certain aberrant brain circuits have been implicated in the pathogenesis of depression.21 Specifically, anatomical and functional imaging suggests connections in the prefrontal cortex are involved in the depression process. In TMS, a series of magnetic pulses are administered via the scalp to stimulate neurons in areas of the brain associated with MDD. Early case reports on using TMS to stimulate the prefrontal cortex found significant improvement of symptoms in patients with depression.22 These promising results spurred great interest in the procedure. Over time, the dose and duration of stimulation has increased, along with FDA-approved indications. TMS was first FDA-approved for TRD.23 Although the primary endpoint of the initial clinical trial did not meet criteria for FDA approval, TMS did result in improvement across multiple other measures of depression.23 After the FDA approved the first TMS device, numerous companies began to produce TMS technology. Most of these companies manufacture devices with the figure-of-eight coil, with 1 company producing the Hesed-coil helmet.24
Continue to: An unintended outcome...
An unintended outcome of the increased interest in TMS has been an increased understanding of brain regions involved in psychiatric illness. TMS was able to bring knowledge of mental health from synapses to circuits.25 Work in this area has further stratified the circuits involved in the manifestation of symptom clusters in depression.26 The exact taxonomy of these brain circuits has not been fully realized, but the default mode, salience, attention, cognitive control, and other circuits have been shown to be involved in specific symptom presentations.26,27 These circuits can be hyperactive, hypoactive, hyperconnected, or hypoconnected, with the aberrancies compared to normal controls resulting in symptoms of psychiatric illness.28
This enhanced understanding of brain function has led to further research and development of protocols and subsequent FDA approval of TMS for OCD, anxious depression, and smoking cessation.29 In addition, it has allowed for a proliferation of off-label uses for TMS, including (but not limited to) tinnitus, pain, migraines, and various substance use disorders.30 TMS treatment for these conditions involves stimulation of specific anatomical brain regions that are thought to play a role in the pathology of the target disorder. For example, subthreshold stimulation of the motor cortex has shown some utility in managing symptoms of pain disorders and movement disorders,31,32 the ventromedial prefrontal cortex has been implicated in disorders in the OCD spectrum,33 stimulation of the frontal poles may help treat substance use disorders,34 and the auditory cortex has been a target for treating tinnitus and auditory hallucinations.35
The location of stimulation for treating depression has evolved. The Talairach-Tournoux coordinate system has been used to determine the location of the dorsolateral prefrontal cortex (DLPFC) in relation to the motor cortex. This was measured to be 5 cm from the motor hotspot and subsequently became “the 5.5 cm rule,” taking skull convexity into account. The treatment paradigm for the Hesed coil also uses a measurement from the motor hotspot. Another commonly used methodology for coil placement involves using the 10 to 20 EEG coordinate system to individualize scalp landmarks. In this method, the F3 location corresponds most accurately to the DLPFC target. More recently, using fMRI-guided navigation for coil placement has been shown to lead to a significant reduction in depressive symptoms.36
For depression, the initial recommended course of treatment is 6 weeks, but most improvement is seen in the first 2 to 3 weeks.14 Therefore, many clinicians administer an initial course of 3 weeks unless the response is inadequate, in which case a 6-week course is administered. Many patients require ongoing maintenance treatment, which can be weekly or monthly based on response.37
Research to determine the optimal TMS dose for treating neuropsychiatric symptoms is ongoing. Location, intensity of stimulation, and pulse are the components of stimulation. The pulse can be subdivided into frequency, pattern (single pulse, standard, burst), train (numbers of pulse groups), interval between trains, and total number of pulses per session. The Clinical TMS Society has published TMS protocols.38 The standard intensity of stimulation is 120% of the motor threshold (MT), which is defined as the amount of stimulation over the motor cortex required to produce movement in the extensor hallucis longus. Although treatment for depression traditionally utilizes rapid TMS (3,000 pulses delivered per session at a frequency of 10 Hz in 4-second trains), in controlled studies, accelerated protocols such as intermittent theta burst stimulation (iTBS; standard stimulation parameters: triplet 50 Hz bursts at 5 Hz, with an interval of 8 seconds for 600 pulses per session) have shown noninferiority.36,39
Recent research has explored fMRI-guided iTBS in an even more accelerated format. The Stanford Neuromodulation Therapy trial involved 1,800 pulses per session for 10 sessions a day for 5 days at 90% MT.36 This treatment paradigm was shown to be more effective than standard protocols and was FDA-approved in 2022. Although this specific iTBS protocol exhibited encouraging results, the need for fMRI for adequate delivery might limit its use.
Continue to: Transcranial direct current stimulation
Transcranial direct current stimulation
Therapeutic noninvasive brain stimulation technology is plausible due to the relative lack of adverse effects and ease of administration. In transcranial direct current stimulation (tDCS), a low-intensity, constant electric current is delivered to stimulate the brain via electrodes attached to the scalp. tDCS modulates spontaneous neuronal network activity40,41 and induces polarization of resting membrane potential at the neuronal level,42 though the exact mechanism is yet to be proven. N-methyl-
tDCS has been suggested as a treatment for various psychiatric and medical conditions. However, the small sample sizes and experimental design of published studies have limited tDCS from being clinically recommended.30 No recommendation of Level A (definite efficacy) for its use was found for any indication. Level B recommendation (probable efficacy) was proposed for fibromyalgia, MDD episode without drug resistance, and addiction/craving. Level C recommendation (possible efficacy) is proposed for chronic lower limb neuropathic pain secondary to spinal cord lesion. tDCS was found to be probably ineffective as a treatment for tinnitus and drug-resistant MDD.30 Some research has suggested that tDCS targeting the DLPFC is associated with cognitive improvements in healthy individuals as well as those with schizophrenia.44 tDCS treatment remains experimental and investigational.
Deep brain stimulation
DBS is a neurosurgical procedure that uses electrical current to directly modulate specific areas of the CNS. In terms of accurate, site-specific anatomical targeting, there can be little doubt of the superiority of DBS. DBS involves the placement of leads into the brain parenchyma. Image guidance techniques are used for accurate placement. DBS is a mainstay for the symptomatic treatment of treatment-resistant movement disorders such as Parkinson disease, essential tremor, and some dystonic disorders. It also has been studied as a potential treatment for chronic pain, cluster headache, Huntington disease, and Tourette syndrome.
For treating depression, researched targets include the subgenual cingulate gyrus (SCG), ventral striatum, nucleus accumbens, inferior thalamic peduncle, medial forebrain bundle, and the red nucleus.45 In systematic reviews, improvement of depression is greatest when DBS targets the subgenual cingulate cortex and the medial forebrain bundle.46
The major limitation of DBS for treating depression is the invasive nature of the procedure. Deep TMS can achieve noninvasive stimulation of the SCG and may be associated with fewer risks, fewer adverse events, and less collateral damage. However, given the evolving concept of abnormal neurologic circuits in depression, as our understanding of circuitry in pathological psychiatric processes increases, DBS may be an attractive option for personalized targeting of symptoms in some patients.
DBS may also be beneficial for severe, treatment-resistant OCD. Electrode implantation in the region of the internal capsule/ventral striatum, including the nucleus accumbens, is used47; there is little difference in placement as a treatment for OCD vs for movement disorders.48
Continue to: A critical review of 23 trials...
A critical review of 23 trials and case reports of DBS as a treatment for OCD demonstrated a 47.7% mean reduction in score on the Yale-Brown Obsessive-Compulsive Scale (Y-BOCS) and a mean response percentage (minimum 35% Y-BOCS reduction) of 58.2%.49 Most patients regained a normal quality of life after DBS.49 A more rigorous review of 15 meta-analyses of DBS found that conclusions about its efficacy or comparative effectiveness cannot be drawn.50 Because of the nature of neurosurgery, DBS has many potential complications, including cognitive changes, headache, infection, seizures, stroke, and hardware failure.
Vagus nerve stimulation
VNS, in which an implanted device stimulates the left vagus nerve with electrical impulses, was FDA-approved for treating chronic TRD in 2005.51 It had been approved for treatment-resistant epilepsy in 1997. In patients with epilepsy, VNS was shown to improve mood independent of seizure control.52 VNS requires a battery-powered pacemaker device to be implanted under the skin over the anterior chest wall, and a wire tunneled to an electrode is wrapped around the left vagus nerve in the neck.53 The pacemaker is then programmed, monitored, and reprogrammed to optimize response.
VNS is believed to stimulate deep brain nuclei that may play a role in depression.54 The onset of improvement is slow (it may take many months) but in carefully selected patients VNS can provide significant control of TRD. In addition to rare surgery-related complications such as a trauma to the vagal nerve and surrounding tissues (vocal cord paralysis, implant site infection, left facial nerve paralysis and Horner syndrome), VNS may cause hoarseness, dyspnea, and cough related to the intensity of the current output.51 Hypomania and mania were also reported; no suicidal behavior has been associated with VNS.51
Noninvasive vagus nerve stimulationIn noninvasive vagus nerve stimulation (nVNS) or transcutaneous VNS, an external handheld device is applied to the neck overlying the course of the vagus nerve to deliver a sinusoidal alternating current.55 nVNS is currently FDA-approved for treating migraine headaches.55,56 It has demonstrated actions on neurophysiology57 and inflammation in patients with MDD.58 Exploratory research has found a small beneficial effect in patients with depression.59,60 A lack of adequate reproducibility prevents this treatment from being more widely recommended, although attempts to standardize the field are evolving.61
Cranial electrical stimulation
Cranial electrical stimulation (CES) is an older form of electric stimulation developed in the 1970s. In CES, mild electrical pulses are delivered to the ear lobes bilaterally in an episodic fashion (usually 20 to 60 minutes once or twice daily). While CES can be considered a form of neuromodulation, it is not strictly interventional. Patients self-administer CES. The procedure has minimal effects on improving sleep, anxiety, and mood.62-66 Potential adverse effects include a tingling sensation in the ear lobes, lightheadedness, and fogginess. A review and meta-analysis of CES for treating addiction by Kirsch67 showed a wide range of symptoms responding positively to CES treatment, although this study was not peer-reviewed. Because of the low quality of nearly all research that evaluated CES, this form of electric stimulation cannot be viewed as an accepted treatment for any of its listed indications.
Continue to: Other neuromodulation techniques
Other
In addition to the forms of neuromodulation we have already described, there are many other techniques. Several are promising but not yet ready for clinical use. Table 1 and Table 2 summarize the neuromodulation techniques described in this article as well as several that are under development.


Acupuncture
Acupuncture is a Chinese form of medical treatment that began >3,000 years ago; there are written descriptions of it from >2,000 years ago.68 It is based on the belief that there are channels within the body through which the Qi (vital energy or life force) flow, and that inserting fine needles into these channels via the skin can rebalance Qi.68 Modern mechanistic hypotheses invoke involvement of inflammatory or pain pathways.69 Acupuncture frequently uses electric stimulation (electro-acupuncture) to increase the potency of the procedure. Alternatively, in a related procedure (acupressure), pressure can replace the needle. Accreditation in acupuncture generally requires a master’s degree in traditional Chinese medicine but does not require any specific medical training. Acupuncture training courses for physicians are widely available.
All forms of acupuncture are experimental for a wide variety of mental and medical conditions. A meta-analysis found that most research of the utility of acupuncture for depression suffered from various forms of potential bias and was considered low quality.70 Nonetheless, active acupuncture was shown to be minimally superior to placebo acupuncture.70 A meta-analysis of acupuncture for preoperative anxiety71,72 and poststroke insomnia73 reported a similar low study quality. A study of 72 patients with primary insomnia revealed that acupuncture was more effective than sham acupuncture for most sleep measures.74
Psychiatry is increasingly integrating medical tools in addition to psychological tools. Pharmacology remains a cornerstone of biological psychiatry and this will not soon change. However, nonpharmacologic psychiatric treatments such as therapeutic neuromodulation are rapidly emerging. These and novel methods of medication administration may present a challenge to psychiatrists who do not have access to medical personnel or may have forgotten general medical skills.
Our 2-part article has highlighted several interventional psychiatry tools—old and new—that may interest clinicians and benefit patients. As a rule, such treatments are reserved for the most treatment-resistant, challenging psychiatric patients, those with hard-to-treat chronic conditions, and patients who are not helped by more commonly used treatments. An additional complication is that such treatments are frequently not appropriately researched, vetted, or FDA-approved, and therefore are higher risk. Appropriate clinical judgment is always necessary, and potential benefits must be thoroughly weighed against possible adverse effects.
Bottom Line
Several forms of neuromodulation, including electroconvulsive therapy, transcranial magnetic stimulation, transcranial direct current stimulation, deep brain stimulation, and vagus nerve stimulation, may be beneficial for patients with certain treatment-resistant psychiatric disorders, including major depressive disorder and obsessive-compulsive disorder.
Related Resources
- Janicak PG. What’s new in transcranial magnetic stimulation. Current Psychiatry. 2019;18(3):10-16.
- Sharma MS, Ang-Rabanes M, Selek S, et al. Neuromodulatory options for treatment-resistant depression. Current Psychiatry. 2018;17(3):26-28,33-37.
While most psychiatric treatments have traditionally consisted of pharmacotherapy with oral medications, a better understanding of the pathophysiology underlying many mental illnesses has led to the recent increased use of treatments that require specialized administration and the creation of a subspecialty called interventional psychiatry. In Part 1 of this 2-part article (“Interventional psychiatry [Part 1],"
Neuromodulation treatments
Neuromodulation—the alteration of nerve activity through targeted delivery of a stimulus, such as electrical stimulation, to specific neurologic sites—is an increasingly common approach to treating a variety of psychiatric conditions. The use of some form of neuromodulation as a medical treatment has a long history (Box1-6). Modern electric neuromodulation began in the 1930s with electroconvulsive therapy (ECT). The 1960s saw the introduction of deep brain stimulation (DBS), spinal cord stimulation, and later, vagus nerve stimulation (VNS). Target-specific noninvasive brain stimulation became possible with transcranial magnetic stimulation (TMS). These approaches are used for treating major depressive disorder (MDD), obsessive-compulsive disorder (OCD), anxiety disorders, and insomnia. Nearly all these neuromodulatory approaches require clinicians to undergo special training and patients to participate in an invasive procedure. These factors also increase cost. Nonetheless, the high rates of success of some of these approaches have led to relatively rapid and widespread acceptance.
Box
The depth and breadth of human anatomical knowledge has evolved over millennia. The time frame “thousands of years” may appear to be an overstatement, but evidence exists for successful therapeutic limb amputation as early as 31,000 years ago.1 This suggests that human knowledge of bone, muscle, and blood supply was developed much earlier than initially believed. Early Homo sapiens were altering the body—regulating or adjusting it— to serve a purpose; in this case, the purpose was survival.
In 46 AD, electrical modulation was introduced by Scribonius Largus, a physician in court of the emperor Tiberius, who used “torpedoes” (most likely electric eels) to treat headaches and pain from arthritis. Loosely, these early clinicians were modulating human function.
In the late 1800s, electrotherapeutics was a growing branch of medicine, with its own national organization—the American ElectroTherapeutic Association.2 In that era, electricity was novel, powerful, and seen as “the future.” Because such novel therapeutics were offered by both mainstream and dubious sources,3 “many of these products were marketed with the promise of curing everything from cancer to headaches.”4
Modern electric neuromodulation began in the 1930s with electroconvulsive therapy,5 followed by deep brain stimulation and spinal cord stimulation in the 1960s. Target-specific noninvasive brain stimulation became possible when Anthony Barker’s team developed the first device that permitted transcranial magnetic stimulation in 1985.6
Electroconvulsive therapy
In ECT, electric current is applied to the brain to induce a self-limiting seizure. It is the oldest and best-known interventional psychiatric treatment. ECT can also be considered one of the first treatments specifically developed to address pathophysiologic changes. In 1934, Ladislas J. Meduna, who had observed in neuropathologic studies that microglia were more numerous in patients with epilepsy compared with patients with schizophrenia, injected a patient who had been hospitalized with catatonia for 4 years with camphor, a proconvulsant.7 After 5 seizures, the patient began to recover. The therapeutic use of electricity was subsequently developed and optimized in animal models, and first used on human patients in Italy in 1939 and in the United States in 1940.8 The link between psychiatric illness and microglia, which was initially observed nearly a century ago, is making a comeback, as excessive microglial activation has been demonstrated in animal and human models of depression.9
Administering ECT requires specialized equipment, anesthesia, physician training, and nursing observation. ECT also has a negative public image.10 All of these factors conspire to reduce the availability of ECT. Despite this, approximately 100,000 patients in the United States and >1 million worldwide receive ECT each year.10 Patients generally require 6 to 12 ECT treatments11 to achieve sufficient response and may require additional maintenance treatments.12
Although ECT is used to treat psychiatric illnesses ranging from mood disorders to psychotic disorders and catatonia, it is mainly employed to treat people with severe treatment-resistant depression (TRD).13 ECT is associated with significant improvements in depressive symptoms and improvements in quality of life.14 It is superior to other treatments for TRD, such as ketamine,15 though a recent study did not show IV ketamine inferiority.16 ECT is also used to treat other neuropsychiatric disorders, such as Parkinson disease.17
Clinicians have explored alternate methods of inducing therapeutic seizures. Magnetic seizure therapy (MST) utilizes a modified magnetic stimulation device to deliver a higher energy in such a way to induce a generalized seizure under anesthesia.18 While patients receiving MST generally experience fewer adverse effects than with ECT, the procedure may be equal to19 or less effective than ECT.20
Transcranial magnetic stimulation
In neuroimaging research, certain aberrant brain circuits have been implicated in the pathogenesis of depression.21 Specifically, anatomical and functional imaging suggests connections in the prefrontal cortex are involved in the depression process. In TMS, a series of magnetic pulses are administered via the scalp to stimulate neurons in areas of the brain associated with MDD. Early case reports on using TMS to stimulate the prefrontal cortex found significant improvement of symptoms in patients with depression.22 These promising results spurred great interest in the procedure. Over time, the dose and duration of stimulation has increased, along with FDA-approved indications. TMS was first FDA-approved for TRD.23 Although the primary endpoint of the initial clinical trial did not meet criteria for FDA approval, TMS did result in improvement across multiple other measures of depression.23 After the FDA approved the first TMS device, numerous companies began to produce TMS technology. Most of these companies manufacture devices with the figure-of-eight coil, with 1 company producing the Hesed-coil helmet.24
Continue to: An unintended outcome...
An unintended outcome of the increased interest in TMS has been an increased understanding of brain regions involved in psychiatric illness. TMS was able to bring knowledge of mental health from synapses to circuits.25 Work in this area has further stratified the circuits involved in the manifestation of symptom clusters in depression.26 The exact taxonomy of these brain circuits has not been fully realized, but the default mode, salience, attention, cognitive control, and other circuits have been shown to be involved in specific symptom presentations.26,27 These circuits can be hyperactive, hypoactive, hyperconnected, or hypoconnected, with the aberrancies compared to normal controls resulting in symptoms of psychiatric illness.28
This enhanced understanding of brain function has led to further research and development of protocols and subsequent FDA approval of TMS for OCD, anxious depression, and smoking cessation.29 In addition, it has allowed for a proliferation of off-label uses for TMS, including (but not limited to) tinnitus, pain, migraines, and various substance use disorders.30 TMS treatment for these conditions involves stimulation of specific anatomical brain regions that are thought to play a role in the pathology of the target disorder. For example, subthreshold stimulation of the motor cortex has shown some utility in managing symptoms of pain disorders and movement disorders,31,32 the ventromedial prefrontal cortex has been implicated in disorders in the OCD spectrum,33 stimulation of the frontal poles may help treat substance use disorders,34 and the auditory cortex has been a target for treating tinnitus and auditory hallucinations.35
The location of stimulation for treating depression has evolved. The Talairach-Tournoux coordinate system has been used to determine the location of the dorsolateral prefrontal cortex (DLPFC) in relation to the motor cortex. This was measured to be 5 cm from the motor hotspot and subsequently became “the 5.5 cm rule,” taking skull convexity into account. The treatment paradigm for the Hesed coil also uses a measurement from the motor hotspot. Another commonly used methodology for coil placement involves using the 10 to 20 EEG coordinate system to individualize scalp landmarks. In this method, the F3 location corresponds most accurately to the DLPFC target. More recently, using fMRI-guided navigation for coil placement has been shown to lead to a significant reduction in depressive symptoms.36
For depression, the initial recommended course of treatment is 6 weeks, but most improvement is seen in the first 2 to 3 weeks.14 Therefore, many clinicians administer an initial course of 3 weeks unless the response is inadequate, in which case a 6-week course is administered. Many patients require ongoing maintenance treatment, which can be weekly or monthly based on response.37
Research to determine the optimal TMS dose for treating neuropsychiatric symptoms is ongoing. Location, intensity of stimulation, and pulse are the components of stimulation. The pulse can be subdivided into frequency, pattern (single pulse, standard, burst), train (numbers of pulse groups), interval between trains, and total number of pulses per session. The Clinical TMS Society has published TMS protocols.38 The standard intensity of stimulation is 120% of the motor threshold (MT), which is defined as the amount of stimulation over the motor cortex required to produce movement in the extensor hallucis longus. Although treatment for depression traditionally utilizes rapid TMS (3,000 pulses delivered per session at a frequency of 10 Hz in 4-second trains), in controlled studies, accelerated protocols such as intermittent theta burst stimulation (iTBS; standard stimulation parameters: triplet 50 Hz bursts at 5 Hz, with an interval of 8 seconds for 600 pulses per session) have shown noninferiority.36,39
Recent research has explored fMRI-guided iTBS in an even more accelerated format. The Stanford Neuromodulation Therapy trial involved 1,800 pulses per session for 10 sessions a day for 5 days at 90% MT.36 This treatment paradigm was shown to be more effective than standard protocols and was FDA-approved in 2022. Although this specific iTBS protocol exhibited encouraging results, the need for fMRI for adequate delivery might limit its use.
Continue to: Transcranial direct current stimulation
Transcranial direct current stimulation
Therapeutic noninvasive brain stimulation technology is plausible due to the relative lack of adverse effects and ease of administration. In transcranial direct current stimulation (tDCS), a low-intensity, constant electric current is delivered to stimulate the brain via electrodes attached to the scalp. tDCS modulates spontaneous neuronal network activity40,41 and induces polarization of resting membrane potential at the neuronal level,42 though the exact mechanism is yet to be proven. N-methyl-
tDCS has been suggested as a treatment for various psychiatric and medical conditions. However, the small sample sizes and experimental design of published studies have limited tDCS from being clinically recommended.30 No recommendation of Level A (definite efficacy) for its use was found for any indication. Level B recommendation (probable efficacy) was proposed for fibromyalgia, MDD episode without drug resistance, and addiction/craving. Level C recommendation (possible efficacy) is proposed for chronic lower limb neuropathic pain secondary to spinal cord lesion. tDCS was found to be probably ineffective as a treatment for tinnitus and drug-resistant MDD.30 Some research has suggested that tDCS targeting the DLPFC is associated with cognitive improvements in healthy individuals as well as those with schizophrenia.44 tDCS treatment remains experimental and investigational.
Deep brain stimulation
DBS is a neurosurgical procedure that uses electrical current to directly modulate specific areas of the CNS. In terms of accurate, site-specific anatomical targeting, there can be little doubt of the superiority of DBS. DBS involves the placement of leads into the brain parenchyma. Image guidance techniques are used for accurate placement. DBS is a mainstay for the symptomatic treatment of treatment-resistant movement disorders such as Parkinson disease, essential tremor, and some dystonic disorders. It also has been studied as a potential treatment for chronic pain, cluster headache, Huntington disease, and Tourette syndrome.
For treating depression, researched targets include the subgenual cingulate gyrus (SCG), ventral striatum, nucleus accumbens, inferior thalamic peduncle, medial forebrain bundle, and the red nucleus.45 In systematic reviews, improvement of depression is greatest when DBS targets the subgenual cingulate cortex and the medial forebrain bundle.46
The major limitation of DBS for treating depression is the invasive nature of the procedure. Deep TMS can achieve noninvasive stimulation of the SCG and may be associated with fewer risks, fewer adverse events, and less collateral damage. However, given the evolving concept of abnormal neurologic circuits in depression, as our understanding of circuitry in pathological psychiatric processes increases, DBS may be an attractive option for personalized targeting of symptoms in some patients.
DBS may also be beneficial for severe, treatment-resistant OCD. Electrode implantation in the region of the internal capsule/ventral striatum, including the nucleus accumbens, is used47; there is little difference in placement as a treatment for OCD vs for movement disorders.48
Continue to: A critical review of 23 trials...
A critical review of 23 trials and case reports of DBS as a treatment for OCD demonstrated a 47.7% mean reduction in score on the Yale-Brown Obsessive-Compulsive Scale (Y-BOCS) and a mean response percentage (minimum 35% Y-BOCS reduction) of 58.2%.49 Most patients regained a normal quality of life after DBS.49 A more rigorous review of 15 meta-analyses of DBS found that conclusions about its efficacy or comparative effectiveness cannot be drawn.50 Because of the nature of neurosurgery, DBS has many potential complications, including cognitive changes, headache, infection, seizures, stroke, and hardware failure.
Vagus nerve stimulation
VNS, in which an implanted device stimulates the left vagus nerve with electrical impulses, was FDA-approved for treating chronic TRD in 2005.51 It had been approved for treatment-resistant epilepsy in 1997. In patients with epilepsy, VNS was shown to improve mood independent of seizure control.52 VNS requires a battery-powered pacemaker device to be implanted under the skin over the anterior chest wall, and a wire tunneled to an electrode is wrapped around the left vagus nerve in the neck.53 The pacemaker is then programmed, monitored, and reprogrammed to optimize response.
VNS is believed to stimulate deep brain nuclei that may play a role in depression.54 The onset of improvement is slow (it may take many months) but in carefully selected patients VNS can provide significant control of TRD. In addition to rare surgery-related complications such as a trauma to the vagal nerve and surrounding tissues (vocal cord paralysis, implant site infection, left facial nerve paralysis and Horner syndrome), VNS may cause hoarseness, dyspnea, and cough related to the intensity of the current output.51 Hypomania and mania were also reported; no suicidal behavior has been associated with VNS.51
Noninvasive vagus nerve stimulationIn noninvasive vagus nerve stimulation (nVNS) or transcutaneous VNS, an external handheld device is applied to the neck overlying the course of the vagus nerve to deliver a sinusoidal alternating current.55 nVNS is currently FDA-approved for treating migraine headaches.55,56 It has demonstrated actions on neurophysiology57 and inflammation in patients with MDD.58 Exploratory research has found a small beneficial effect in patients with depression.59,60 A lack of adequate reproducibility prevents this treatment from being more widely recommended, although attempts to standardize the field are evolving.61
Cranial electrical stimulation
Cranial electrical stimulation (CES) is an older form of electric stimulation developed in the 1970s. In CES, mild electrical pulses are delivered to the ear lobes bilaterally in an episodic fashion (usually 20 to 60 minutes once or twice daily). While CES can be considered a form of neuromodulation, it is not strictly interventional. Patients self-administer CES. The procedure has minimal effects on improving sleep, anxiety, and mood.62-66 Potential adverse effects include a tingling sensation in the ear lobes, lightheadedness, and fogginess. A review and meta-analysis of CES for treating addiction by Kirsch67 showed a wide range of symptoms responding positively to CES treatment, although this study was not peer-reviewed. Because of the low quality of nearly all research that evaluated CES, this form of electric stimulation cannot be viewed as an accepted treatment for any of its listed indications.
Continue to: Other neuromodulation techniques
Other
In addition to the forms of neuromodulation we have already described, there are many other techniques. Several are promising but not yet ready for clinical use. Table 1 and Table 2 summarize the neuromodulation techniques described in this article as well as several that are under development.


Acupuncture
Acupuncture is a Chinese form of medical treatment that began >3,000 years ago; there are written descriptions of it from >2,000 years ago.68 It is based on the belief that there are channels within the body through which the Qi (vital energy or life force) flow, and that inserting fine needles into these channels via the skin can rebalance Qi.68 Modern mechanistic hypotheses invoke involvement of inflammatory or pain pathways.69 Acupuncture frequently uses electric stimulation (electro-acupuncture) to increase the potency of the procedure. Alternatively, in a related procedure (acupressure), pressure can replace the needle. Accreditation in acupuncture generally requires a master’s degree in traditional Chinese medicine but does not require any specific medical training. Acupuncture training courses for physicians are widely available.
All forms of acupuncture are experimental for a wide variety of mental and medical conditions. A meta-analysis found that most research of the utility of acupuncture for depression suffered from various forms of potential bias and was considered low quality.70 Nonetheless, active acupuncture was shown to be minimally superior to placebo acupuncture.70 A meta-analysis of acupuncture for preoperative anxiety71,72 and poststroke insomnia73 reported a similar low study quality. A study of 72 patients with primary insomnia revealed that acupuncture was more effective than sham acupuncture for most sleep measures.74
Psychiatry is increasingly integrating medical tools in addition to psychological tools. Pharmacology remains a cornerstone of biological psychiatry and this will not soon change. However, nonpharmacologic psychiatric treatments such as therapeutic neuromodulation are rapidly emerging. These and novel methods of medication administration may present a challenge to psychiatrists who do not have access to medical personnel or may have forgotten general medical skills.
Our 2-part article has highlighted several interventional psychiatry tools—old and new—that may interest clinicians and benefit patients. As a rule, such treatments are reserved for the most treatment-resistant, challenging psychiatric patients, those with hard-to-treat chronic conditions, and patients who are not helped by more commonly used treatments. An additional complication is that such treatments are frequently not appropriately researched, vetted, or FDA-approved, and therefore are higher risk. Appropriate clinical judgment is always necessary, and potential benefits must be thoroughly weighed against possible adverse effects.
Bottom Line
Several forms of neuromodulation, including electroconvulsive therapy, transcranial magnetic stimulation, transcranial direct current stimulation, deep brain stimulation, and vagus nerve stimulation, may be beneficial for patients with certain treatment-resistant psychiatric disorders, including major depressive disorder and obsessive-compulsive disorder.
Related Resources
- Janicak PG. What’s new in transcranial magnetic stimulation. Current Psychiatry. 2019;18(3):10-16.
- Sharma MS, Ang-Rabanes M, Selek S, et al. Neuromodulatory options for treatment-resistant depression. Current Psychiatry. 2018;17(3):26-28,33-37.
1. Maloney TR, Dilkes-Hall IE, Vlok M, et al. Surgical amputation of a limb 31,000 years ago in Borneo. Nature. 2022;609(7927):547-551. doi:10.1038/s41586-022-05160-8
2. The American Electro-Therapeutic Association. JAMA. 1893;21(14):500. doi:10.1001/jama.1893.02420660030004
3. The American Electro-Therapeutic Association. JAMA. 1894;23(15):590-591. doi:10.1001/jama.1894.02421200024006
4. Wexler A. The medical battery in the United States (1870-1920): electrotherapy at home and in the clinic. J Hist Med Allied Sci. 2017;72(2):166-192. doi:10.1093/jhmas/jrx001
5. Gazdag G, Ungvari GS. Electroconvulsive therapy: 80 years old and still going strong. World J Psychiatry. 2019;9(1):1-6. doi:10.5498/wjp.v9.i1.1
6. Barker AT, Jalinous R, Freeston IL. Non-invasive magnetic stimulation of human motor cortex. Lancet. 1985;1(8437):1106-1107. doi:10.1016/s0140-6736(85)92413-4
7. Fink M. Historical article: autobiography of L. J. Meduna. Convuls Ther. 1985;1(1):43-57.
8. Suleman R. A brief history of electroconvulsive therapy. Am J Psychiatry. 2020;16(1):6. doi:10.1176/appi.ajp-rj.2020.160103
9. Ménard C, Hodes GE, Russo SJ. Pathogenesis of depression: insights from human and rodent studies. Neuroscience. 2016;321:138-162. doi:10.1016/j.neuroscience.2015.05.053
10. Payne NA, Prudic J. Electroconvulsive therapy: part II: a biopsychosocial perspective. J Psychiatr Pract. 2009;15(5):369-390. doi:10.1097/01.pra.0000361278.73092.85
11. Tirmizi O, Raza A, Trevino K, et al. Electroconvulsive therapy: how modern techniques improve patient outcomes. Current Psychiatry. 2012;11(10):24-46.
12. Kolar D. Current status of electroconvulsive therapy for mood disorders: a clinical review. Evid Based Ment Health. 2017;20(1):12-14. doi:10.1136/eb-2016-102498
13. Andrade C. Active placebo, the parachute meta-analysis, the Nobel Prize, and the efficacy of electroconvulsive therapy. J Clin Psychiatry. 2021;82(2):21f13992. doi:10.4088/JCP.21f13992
14. Giacobbe P, Rakita U, Penner-Goeke K, et al. Improvements in health-related quality of life with electroconvulsive therapy: a meta-analysis. J ECT. 2018;34(2):87-94. doi:10.1097/YCT.0000000000000486
15. Rhee TG, Shim SR, Forester BP, et al. Efficacy and safety of ketamine vs electroconvulsive therapy among patients with major depressive episode: a systematic review and meta-analysis. JAMA Psychiatry. 2022;79(12):1162-1172. doi:10.1001/jamapsychiatry.2022.3352
16. Anand A, Mathew SJ, Sanacora G, et al. Ketamine versus ECT for nonpsychotic treatment-resistant major depression. N Engl J Med. 2023. doi: 10.1056/NEJMoa2302399
17. Takamiya A, Seki M, Kudo S, et al. Electroconvulsive therapy for Parkinson’s disease: a systematic review and meta-analysis. Mov Disord. 2021;36(1):50-58. doi:10.1002/mds.28335
18. Singh R, Sharma R, Prakash J, et al. Magnetic seizure therapy. Ind Psychiatry J. 2021;30(Suppl 1):S320-S321. doi:10.4103/0972-6748.328841
19. Chen M, Yang X, Liu C, et al. Comparative efficacy and cognitive function of magnetic seizure therapy vs. electroconvulsive therapy for major depressive disorder: a systematic review and meta-analysis. Transl Psychiatry. 2021;11(1):437. doi:10.1038/s41398-021-01560-y
20. Cretaz E, Brunoni AR, Lafer B. Magnetic seizure therapy for unipolar and bipolar depression: a systematic review. Neural Plast. 2015;2015:521398. doi:10.1155/2015/521398
21. George MS, Ketter TA, Post RM. Prefrontal cortex dysfunction in clinical depression. In: Nemeroff CB, Weiss JM, Schatzberg AF, et al, eds. Depression. 2nd ed. Wiley Online Library; 1994:59-72. https://doi.org/10.1002/depr.3050020202
22. George MS, Wassermann EM, Williams WA, et al. Daily repetitive transcranial magnetic stimulation (rTMS) improves mood in depression. Neuroreport. 1995;6(14):1853-1856.
23. O’Reardon JP, Solvason HB, Janicak PG, et al. Efficacy and safety of transcranial magnetic stimulation in the acute treatment of major depression: a multisite randomized controlled trial. Biol Psychiatry. 2007;62(11):1208-1216.
24. Clinical TMS Society. TMS devices. Accessed January 2, 2023. https://www.clinicaltmssociety.org/devices
25. Goldstein-Piekarski AN, Ball TM, Samara Z, et al. Mapping neural circuit biotypes to symptoms and behavioral dimensions of depression and anxiety. Biol Psychiatry. 2022;91(6):561-571. doi:10.1016/j.biopsych.2021.06.024
26. Siddiqi SH, Taylor SF, Cooke D, et al. Distinct symptom-specific treatment targets for circuit-based neuromodulation. Am J Psychiatry. 2020;177(5):435-446. doi:10.1176/appi.ajp.2019.19090915
27. Williams LM. Defining biotypes for depression and anxiety based on large-scale circuit dysfunction: a theoretical review of the evidence and future directions for clinical translation. Depress Anxiety. 2017;34(1):9-24. doi:10.1002/da.22556
28. Drysdale AT, Grosenick L, Downar J, et al. Resting-state connectivity biomarkers define neurophysiological subtypes of depression. Nat Med. 2017;23(1):28-38. doi:10.1038/nm.4246
29. Cohen SL, Bikson M, Badran BW, et al. A visual and narrative timeline of US FDA milestones for transcranial magnetic stimulation (TMS) devices. Brain Stimul. 2022;15(1):73-75. doi:10.1016/j.brs.2021.11.010
30. Lefaucheur JP, Antal A, Ayache SS, et al. Evidence-based guidelines on the therapeutic use of transcranial direct current stimulation (tDCS). Clin Neurophysiol. 2017;128(1):56-92. doi:10.1016/j.clinph.2016.10.087
31. Li R, He Y, Qin W, et al. Effects of repetitive transcranial magnetic stimulation on motor symptoms in Parkinson’s disease: a meta-analysis. Neurorehabil Neural Repair. 2022;36(7):395-404. doi:10.1177/15459683221095034
32. Leung A, Shirvalkar P, Chen R, et al. Transcranial magnetic stimulation for pain, headache, and comorbid depression: INS-NANS expert consensus panel review and recommendation. Neuromodulation. 2020;23(3):267-290. doi:10.1111/ner.13094
33. Carmi L, Tendler A, Bystritsky A, et al. Efficacy and safety of deep transcranial magnetic stimulation for obsessive-compulsive disorder: a prospective multicenter randomized double-blind placebo-controlled trial. Am J Psychiatry. 2019;176(11):931-938. doi:10.1176/appi.ajp.2019.18101180
34. Harel M, Perini I, Kämpe R, et al. Repetitive transcranial magnetic stimulation in alcohol dependence: a randomized, double-blind, sham-controlled proof-of-concept trial targeting the medial prefrontal and anterior cingulate cortices. Biol Psychiatry. 2022;91(12):1061-1069. doi:10.1016/j.biopsych.2021.11.020
35. Folmer RL, Theodoroff SM, Casiana L, et al. Repetitive transcranial magnetic stimulation treatment for chronic tinnitus: a randomized clinical trial. JAMA Otolaryngol Head Neck Surg. 2015;141(8):716-722. doi:10.1001/jamaoto.2015.1219
36. Cole EJ, Phillips AL, Bentzley BS, et al. Stanford Neuromodulation Therapy (SNT): a double-blind randomized controlled trial. Am J Psychiatry. 2022;179(2):132-141. doi:10.1176/appi.ajp.2021.20101429
37. Wilson S, Croarkin PE, Aaronson ST, et al. Systematic review of preservation TMS that includes continuation, maintenance, relapse-prevention, and rescue TMS. J Affect Disord. 2022;296:79-88. doi:10.1016/j.jad.2021.09.040
38. Perera T, George MS, Grammer G, et al. The Clinical TMS Society consensus review and treatment recommendations for TMS therapy for major depressive disorder. Brain Stimul. 2016;9(3):336-346. doi:10.1016/j.brs.2016.03.010
39. Blumberger DM, Vila-Rodriguez F, Thorpe KE, et al. Effectiveness of theta burst versus high-frequency repetitive transcranial magnetic stimulation in patients with depression (THREE-D): a randomized non-inferiority trial. Lancet. 2018;391(10131):1683-1692. doi:10.1016/S0140-6736(18)30295-2
40. Nitsche MA, Cohen LG, Wassermann EM, et al. Transcranial direct current stimulation: state of the art 2008. Brain Stimul. 2008;1(3):206-223. doi:10.1016/j.brs.2008.06.004
41. Priori A, Hallett M, Rothwell JC. Repetitive transcranial magnetic stimulation or transcranial direct current stimulation? Brain Stimul. 2009;2(4):241-245.
42. Priori A, Berardelli A, Rona S, et al. Polarization of the human motor cortex through the scalp. Neuroreport. 1998;9(10):2257-2260. doi:10.1097/00001756-199807130-00020
43. Nitsche MA, Liebetanz D, Antal A, et al. Modulation of cortical excitability by weak direct current stimulation-- technical, safety and functional aspects. Suppl Clin Neurophysiol. 2003;56:255-276. doi:10.1016/s1567-424x(09)70230-2
44. Agarwal SM, Venkataram Shivakumar V, et al. Transcranial direct current stimulation in schizophrenia. Clin Psychopharmacol Neurosci. 2013;11(3):118-125.
45. Drobisz D, Damborská A. Deep brain stimulation targets for treating depression. Behav Brain Res. 2019;359:266-273. doi:10.1016/j.bbr.2018.11.004
46. Kisely S, Li A, Warren N, et al. A systematic review and meta-analysis of deep brain stimulation for depression. Depress Anxiety. 2018;35(5):468-480. doi:10.1002/da.22746
47. Blomstedt P, Sjöberg RL, Hansson M, et al. Deep brain stimulation in the treatment of obsessive-compulsive disorder. World Neurosurg. 2013;80(6):e245-e253. doi:10.1016/j.wneu.2012.10.006
48. Denys D, Mantione M, Figee M, et al. Deep brain stimulation of the nucleus accumbens for treatment-refractory obsessive-compulsive disorder. Arch Gen Psychiatry. 2010;67(10):1061-1068. doi:10.1001/archgenpsychiatry.2010.122
49. van Westen M, Rietveld E, Figee M, et al. Clinical outcome and mechanisms of deep brain stimulation for obsessive-compulsive disorder. Curr Behav Neurosci Rep. 2015;2(2):41-48. doi:10.1007/s40473-015-0036-3
50. Papageorgiou PN, Deschner J, Papageorgiou SN. Effectiveness and adverse effects of deep brain stimulation: umbrella review of meta-analyses. J Neurol Surg A Cent Eur Neurosurg. 2017;78(2):180-190. doi:10.1055/s-0036-1592158
51. O’Reardon JP, Cristancho P, Peshek AD. Vagus nerve stimulation (VNS) and treatment of depression: to the brainstem and beyond. Psychiatry (Edgmont). 2006;3(5):54-63.
52. Harden CL, Pulver MC, Ravdin LD, et al. A pilot study of mood in epilepsy patients treated with vagus nerve stimulation. Epilepsy Behav. 2000;1(2):93-99. doi:10.1006/ebeh.2000.0046
53. Giordano F, Zicca A, Barba C, et al. Vagus nerve stimulation: surgical technique of implantation and revision and related morbidity. Epilepsia. 2017;58(S1):85-90. doi:10.1111/epi.13687
54. George MS, Nahas Z, Bohning DE, et al. Mechanisms of action of vagus nerve stimulation (VNS). Clin Neurosci Res. 2004;4(1-2):71-79.
55. Nesbitt AD, Marin JCA, Tompkins E, et al. Initial use of a novel noninvasive vagus nerve stimulator for cluster headache treatment. Neurology. 2015;84:1249-1253. doi:10.1212/WNL.0000000000001394
56. Goadsby PJ, Grosberg BM, Mauskop A, et al. Effect of noninvasive vagus nerve stimulation on acute migraine: an open-label pilot study. Cephalalgia. 2014;34:986-993. doi:10.1177/0333102414524494
57. Fang J, Rong P, Hong Y, et al. Transcutaneous vagus nerve stimulation modulates default mode network in major depressive disorder. Biol Psychiatry. 2016;79(4):266-273. doi:10.1016/j.biopsych.2015.03.025
58. Liu CH, Yang MH, Zhang GZ, et al. Neural networks and the anti-inflammatory effect of transcutaneous auricular vagus nerve stimulation in depression. J Neuroinflammation. 2020;17(1):54. doi:10.1186/s12974-020-01732-5
59. Hein E, Nowak M, Kiess O, et al. Auricular transcutaneous electrical nerve stimulation in depressed patients: a randomized controlled pilot study. J Neural Transm (Vienna). 2013;120(5):821-827. doi:10.1007/s00702-012-0908-6
60. Rong P, Liu J, Wang L, et al. Effect of transcutaneous auricular vagus nerve stimulation on major depressive disorder: a nonrandomized controlled pilot study. J Affect Disord. 2016;195:172-179. doi:10.1016/j.jad.2016.02.031
61. Farmer AD, Strzelczyk A, Finisguerra A, et al. International consensus based review and recommendations for minimum reporting standards in research on transcutaneous vagus nerve stimulation (Version 2020). Front Hum Neurosci. 2021;14:568051. doi:10.3389/fnhum.2020.568051
62. Amr M, El-Wasify M, Elmaadawi AZ, et al. Cranial electrotherapy stimulation for the treatment of chronically symptomatic bipolar patients. J ECT. 2013;29(2):e31-e32. doi:10.1097/YCT.0b013e31828a344d
63. Kirsch DL, Nichols F. Cranial electrotherapy stimulation for treatment of anxiety, depression, and insomnia. Psychiatr Clin North Am. 2013;36(1):169-176. doi:10.1016/j.psc.2013.01.006
64. Lande RG, Gragnani C. Efficacy of cranial electric stimulation for the treatment of insomnia: a randomized pilot study. Complement Ther Med. 2013;21(1):8-13. doi:10.1016/j.ctim.2012.11.007
65. Ou Y, Li, C. Sertraline combined alpha-stim clinical observations on the treatment of 30 cases of generalized anxiety disorder. Chinese Journal of Ethnomedicine and Ethnopharmacy. 2015;24(17):73-75.
66. Price L, Briley J, Haltiwanger S, et al. A meta-analysis of cranial electrotherapy stimulation in the treatment of depression. J Psychiatr Res. 2021;135:119-134. doi:10.1016/j.jpsychires.2020.12.043
67. Kirsch D, Gilula M. CES in the treatment of addictions: a review and meta-analysis. Pract Pain Manag. 2007;7(9).
68. Hao JJ, Mittelman M. Acupuncture: past, present, and future. Glob Adv Health Med. 2014;3(4):6-8. doi:10.7453/gahmj.2014.042
69. Napadow V, Ahn A, Longhurst J, et al. The status and future of acupuncture mechanism research. J Altern Complement Med. 2008;14(7):861-869. doi:10.1089/acm.2008.SAR-3
70. Smith CA, Armour M, Lee MS, et al. Acupuncture for depression. Cochrane Database Syst Rev. 2018;3(3):CD004046. doi:10.1002/14651858.CD004046.pub4
71. Tong QY, Liu R, Zhang K, et al. Can acupuncture therapy reduce preoperative anxiety? A systematic review and meta-analysis. J Integr Med. 2021;19(1):20-28. doi:10.1016/j.joim.2020.10.007
72. Usichenko TI, Hua K, Cummings M, et al. Auricular stimulation for preoperative anxiety – a systematic review and meta-analysis of randomized controlled clinical trials. J Clin Anesth. 2022;76:110581. doi:10.1016/j.jclinane.2021.110581
73. Zhou L, Hu X, Yu Z, et al. Efficacy and safety of acupuncture in the treatment of poststroke insomnia: a systematic review and meta-analysis of twenty-six randomized controlled trials. Evid Based Complement Alternat Med. 2022;2022:5188311. doi:10.1155/2022/5188311
74. Yin X, Gou M, Xu J, et al. Efficacy and safety of acupuncture treatment on primary insomnia: a randomized controlled trial. Sleep Med. 2017;37:193-200. doi:10.1016/j.sleep.2017.02.012
1. Maloney TR, Dilkes-Hall IE, Vlok M, et al. Surgical amputation of a limb 31,000 years ago in Borneo. Nature. 2022;609(7927):547-551. doi:10.1038/s41586-022-05160-8
2. The American Electro-Therapeutic Association. JAMA. 1893;21(14):500. doi:10.1001/jama.1893.02420660030004
3. The American Electro-Therapeutic Association. JAMA. 1894;23(15):590-591. doi:10.1001/jama.1894.02421200024006
4. Wexler A. The medical battery in the United States (1870-1920): electrotherapy at home and in the clinic. J Hist Med Allied Sci. 2017;72(2):166-192. doi:10.1093/jhmas/jrx001
5. Gazdag G, Ungvari GS. Electroconvulsive therapy: 80 years old and still going strong. World J Psychiatry. 2019;9(1):1-6. doi:10.5498/wjp.v9.i1.1
6. Barker AT, Jalinous R, Freeston IL. Non-invasive magnetic stimulation of human motor cortex. Lancet. 1985;1(8437):1106-1107. doi:10.1016/s0140-6736(85)92413-4
7. Fink M. Historical article: autobiography of L. J. Meduna. Convuls Ther. 1985;1(1):43-57.
8. Suleman R. A brief history of electroconvulsive therapy. Am J Psychiatry. 2020;16(1):6. doi:10.1176/appi.ajp-rj.2020.160103
9. Ménard C, Hodes GE, Russo SJ. Pathogenesis of depression: insights from human and rodent studies. Neuroscience. 2016;321:138-162. doi:10.1016/j.neuroscience.2015.05.053
10. Payne NA, Prudic J. Electroconvulsive therapy: part II: a biopsychosocial perspective. J Psychiatr Pract. 2009;15(5):369-390. doi:10.1097/01.pra.0000361278.73092.85
11. Tirmizi O, Raza A, Trevino K, et al. Electroconvulsive therapy: how modern techniques improve patient outcomes. Current Psychiatry. 2012;11(10):24-46.
12. Kolar D. Current status of electroconvulsive therapy for mood disorders: a clinical review. Evid Based Ment Health. 2017;20(1):12-14. doi:10.1136/eb-2016-102498
13. Andrade C. Active placebo, the parachute meta-analysis, the Nobel Prize, and the efficacy of electroconvulsive therapy. J Clin Psychiatry. 2021;82(2):21f13992. doi:10.4088/JCP.21f13992
14. Giacobbe P, Rakita U, Penner-Goeke K, et al. Improvements in health-related quality of life with electroconvulsive therapy: a meta-analysis. J ECT. 2018;34(2):87-94. doi:10.1097/YCT.0000000000000486
15. Rhee TG, Shim SR, Forester BP, et al. Efficacy and safety of ketamine vs electroconvulsive therapy among patients with major depressive episode: a systematic review and meta-analysis. JAMA Psychiatry. 2022;79(12):1162-1172. doi:10.1001/jamapsychiatry.2022.3352
16. Anand A, Mathew SJ, Sanacora G, et al. Ketamine versus ECT for nonpsychotic treatment-resistant major depression. N Engl J Med. 2023. doi: 10.1056/NEJMoa2302399
17. Takamiya A, Seki M, Kudo S, et al. Electroconvulsive therapy for Parkinson’s disease: a systematic review and meta-analysis. Mov Disord. 2021;36(1):50-58. doi:10.1002/mds.28335
18. Singh R, Sharma R, Prakash J, et al. Magnetic seizure therapy. Ind Psychiatry J. 2021;30(Suppl 1):S320-S321. doi:10.4103/0972-6748.328841
19. Chen M, Yang X, Liu C, et al. Comparative efficacy and cognitive function of magnetic seizure therapy vs. electroconvulsive therapy for major depressive disorder: a systematic review and meta-analysis. Transl Psychiatry. 2021;11(1):437. doi:10.1038/s41398-021-01560-y
20. Cretaz E, Brunoni AR, Lafer B. Magnetic seizure therapy for unipolar and bipolar depression: a systematic review. Neural Plast. 2015;2015:521398. doi:10.1155/2015/521398
21. George MS, Ketter TA, Post RM. Prefrontal cortex dysfunction in clinical depression. In: Nemeroff CB, Weiss JM, Schatzberg AF, et al, eds. Depression. 2nd ed. Wiley Online Library; 1994:59-72. https://doi.org/10.1002/depr.3050020202
22. George MS, Wassermann EM, Williams WA, et al. Daily repetitive transcranial magnetic stimulation (rTMS) improves mood in depression. Neuroreport. 1995;6(14):1853-1856.
23. O’Reardon JP, Solvason HB, Janicak PG, et al. Efficacy and safety of transcranial magnetic stimulation in the acute treatment of major depression: a multisite randomized controlled trial. Biol Psychiatry. 2007;62(11):1208-1216.
24. Clinical TMS Society. TMS devices. Accessed January 2, 2023. https://www.clinicaltmssociety.org/devices
25. Goldstein-Piekarski AN, Ball TM, Samara Z, et al. Mapping neural circuit biotypes to symptoms and behavioral dimensions of depression and anxiety. Biol Psychiatry. 2022;91(6):561-571. doi:10.1016/j.biopsych.2021.06.024
26. Siddiqi SH, Taylor SF, Cooke D, et al. Distinct symptom-specific treatment targets for circuit-based neuromodulation. Am J Psychiatry. 2020;177(5):435-446. doi:10.1176/appi.ajp.2019.19090915
27. Williams LM. Defining biotypes for depression and anxiety based on large-scale circuit dysfunction: a theoretical review of the evidence and future directions for clinical translation. Depress Anxiety. 2017;34(1):9-24. doi:10.1002/da.22556
28. Drysdale AT, Grosenick L, Downar J, et al. Resting-state connectivity biomarkers define neurophysiological subtypes of depression. Nat Med. 2017;23(1):28-38. doi:10.1038/nm.4246
29. Cohen SL, Bikson M, Badran BW, et al. A visual and narrative timeline of US FDA milestones for transcranial magnetic stimulation (TMS) devices. Brain Stimul. 2022;15(1):73-75. doi:10.1016/j.brs.2021.11.010
30. Lefaucheur JP, Antal A, Ayache SS, et al. Evidence-based guidelines on the therapeutic use of transcranial direct current stimulation (tDCS). Clin Neurophysiol. 2017;128(1):56-92. doi:10.1016/j.clinph.2016.10.087
31. Li R, He Y, Qin W, et al. Effects of repetitive transcranial magnetic stimulation on motor symptoms in Parkinson’s disease: a meta-analysis. Neurorehabil Neural Repair. 2022;36(7):395-404. doi:10.1177/15459683221095034
32. Leung A, Shirvalkar P, Chen R, et al. Transcranial magnetic stimulation for pain, headache, and comorbid depression: INS-NANS expert consensus panel review and recommendation. Neuromodulation. 2020;23(3):267-290. doi:10.1111/ner.13094
33. Carmi L, Tendler A, Bystritsky A, et al. Efficacy and safety of deep transcranial magnetic stimulation for obsessive-compulsive disorder: a prospective multicenter randomized double-blind placebo-controlled trial. Am J Psychiatry. 2019;176(11):931-938. doi:10.1176/appi.ajp.2019.18101180
34. Harel M, Perini I, Kämpe R, et al. Repetitive transcranial magnetic stimulation in alcohol dependence: a randomized, double-blind, sham-controlled proof-of-concept trial targeting the medial prefrontal and anterior cingulate cortices. Biol Psychiatry. 2022;91(12):1061-1069. doi:10.1016/j.biopsych.2021.11.020
35. Folmer RL, Theodoroff SM, Casiana L, et al. Repetitive transcranial magnetic stimulation treatment for chronic tinnitus: a randomized clinical trial. JAMA Otolaryngol Head Neck Surg. 2015;141(8):716-722. doi:10.1001/jamaoto.2015.1219
36. Cole EJ, Phillips AL, Bentzley BS, et al. Stanford Neuromodulation Therapy (SNT): a double-blind randomized controlled trial. Am J Psychiatry. 2022;179(2):132-141. doi:10.1176/appi.ajp.2021.20101429
37. Wilson S, Croarkin PE, Aaronson ST, et al. Systematic review of preservation TMS that includes continuation, maintenance, relapse-prevention, and rescue TMS. J Affect Disord. 2022;296:79-88. doi:10.1016/j.jad.2021.09.040
38. Perera T, George MS, Grammer G, et al. The Clinical TMS Society consensus review and treatment recommendations for TMS therapy for major depressive disorder. Brain Stimul. 2016;9(3):336-346. doi:10.1016/j.brs.2016.03.010
39. Blumberger DM, Vila-Rodriguez F, Thorpe KE, et al. Effectiveness of theta burst versus high-frequency repetitive transcranial magnetic stimulation in patients with depression (THREE-D): a randomized non-inferiority trial. Lancet. 2018;391(10131):1683-1692. doi:10.1016/S0140-6736(18)30295-2
40. Nitsche MA, Cohen LG, Wassermann EM, et al. Transcranial direct current stimulation: state of the art 2008. Brain Stimul. 2008;1(3):206-223. doi:10.1016/j.brs.2008.06.004
41. Priori A, Hallett M, Rothwell JC. Repetitive transcranial magnetic stimulation or transcranial direct current stimulation? Brain Stimul. 2009;2(4):241-245.
42. Priori A, Berardelli A, Rona S, et al. Polarization of the human motor cortex through the scalp. Neuroreport. 1998;9(10):2257-2260. doi:10.1097/00001756-199807130-00020
43. Nitsche MA, Liebetanz D, Antal A, et al. Modulation of cortical excitability by weak direct current stimulation-- technical, safety and functional aspects. Suppl Clin Neurophysiol. 2003;56:255-276. doi:10.1016/s1567-424x(09)70230-2
44. Agarwal SM, Venkataram Shivakumar V, et al. Transcranial direct current stimulation in schizophrenia. Clin Psychopharmacol Neurosci. 2013;11(3):118-125.
45. Drobisz D, Damborská A. Deep brain stimulation targets for treating depression. Behav Brain Res. 2019;359:266-273. doi:10.1016/j.bbr.2018.11.004
46. Kisely S, Li A, Warren N, et al. A systematic review and meta-analysis of deep brain stimulation for depression. Depress Anxiety. 2018;35(5):468-480. doi:10.1002/da.22746
47. Blomstedt P, Sjöberg RL, Hansson M, et al. Deep brain stimulation in the treatment of obsessive-compulsive disorder. World Neurosurg. 2013;80(6):e245-e253. doi:10.1016/j.wneu.2012.10.006
48. Denys D, Mantione M, Figee M, et al. Deep brain stimulation of the nucleus accumbens for treatment-refractory obsessive-compulsive disorder. Arch Gen Psychiatry. 2010;67(10):1061-1068. doi:10.1001/archgenpsychiatry.2010.122
49. van Westen M, Rietveld E, Figee M, et al. Clinical outcome and mechanisms of deep brain stimulation for obsessive-compulsive disorder. Curr Behav Neurosci Rep. 2015;2(2):41-48. doi:10.1007/s40473-015-0036-3
50. Papageorgiou PN, Deschner J, Papageorgiou SN. Effectiveness and adverse effects of deep brain stimulation: umbrella review of meta-analyses. J Neurol Surg A Cent Eur Neurosurg. 2017;78(2):180-190. doi:10.1055/s-0036-1592158
51. O’Reardon JP, Cristancho P, Peshek AD. Vagus nerve stimulation (VNS) and treatment of depression: to the brainstem and beyond. Psychiatry (Edgmont). 2006;3(5):54-63.
52. Harden CL, Pulver MC, Ravdin LD, et al. A pilot study of mood in epilepsy patients treated with vagus nerve stimulation. Epilepsy Behav. 2000;1(2):93-99. doi:10.1006/ebeh.2000.0046
53. Giordano F, Zicca A, Barba C, et al. Vagus nerve stimulation: surgical technique of implantation and revision and related morbidity. Epilepsia. 2017;58(S1):85-90. doi:10.1111/epi.13687
54. George MS, Nahas Z, Bohning DE, et al. Mechanisms of action of vagus nerve stimulation (VNS). Clin Neurosci Res. 2004;4(1-2):71-79.
55. Nesbitt AD, Marin JCA, Tompkins E, et al. Initial use of a novel noninvasive vagus nerve stimulator for cluster headache treatment. Neurology. 2015;84:1249-1253. doi:10.1212/WNL.0000000000001394
56. Goadsby PJ, Grosberg BM, Mauskop A, et al. Effect of noninvasive vagus nerve stimulation on acute migraine: an open-label pilot study. Cephalalgia. 2014;34:986-993. doi:10.1177/0333102414524494
57. Fang J, Rong P, Hong Y, et al. Transcutaneous vagus nerve stimulation modulates default mode network in major depressive disorder. Biol Psychiatry. 2016;79(4):266-273. doi:10.1016/j.biopsych.2015.03.025
58. Liu CH, Yang MH, Zhang GZ, et al. Neural networks and the anti-inflammatory effect of transcutaneous auricular vagus nerve stimulation in depression. J Neuroinflammation. 2020;17(1):54. doi:10.1186/s12974-020-01732-5
59. Hein E, Nowak M, Kiess O, et al. Auricular transcutaneous electrical nerve stimulation in depressed patients: a randomized controlled pilot study. J Neural Transm (Vienna). 2013;120(5):821-827. doi:10.1007/s00702-012-0908-6
60. Rong P, Liu J, Wang L, et al. Effect of transcutaneous auricular vagus nerve stimulation on major depressive disorder: a nonrandomized controlled pilot study. J Affect Disord. 2016;195:172-179. doi:10.1016/j.jad.2016.02.031
61. Farmer AD, Strzelczyk A, Finisguerra A, et al. International consensus based review and recommendations for minimum reporting standards in research on transcutaneous vagus nerve stimulation (Version 2020). Front Hum Neurosci. 2021;14:568051. doi:10.3389/fnhum.2020.568051
62. Amr M, El-Wasify M, Elmaadawi AZ, et al. Cranial electrotherapy stimulation for the treatment of chronically symptomatic bipolar patients. J ECT. 2013;29(2):e31-e32. doi:10.1097/YCT.0b013e31828a344d
63. Kirsch DL, Nichols F. Cranial electrotherapy stimulation for treatment of anxiety, depression, and insomnia. Psychiatr Clin North Am. 2013;36(1):169-176. doi:10.1016/j.psc.2013.01.006
64. Lande RG, Gragnani C. Efficacy of cranial electric stimulation for the treatment of insomnia: a randomized pilot study. Complement Ther Med. 2013;21(1):8-13. doi:10.1016/j.ctim.2012.11.007
65. Ou Y, Li, C. Sertraline combined alpha-stim clinical observations on the treatment of 30 cases of generalized anxiety disorder. Chinese Journal of Ethnomedicine and Ethnopharmacy. 2015;24(17):73-75.
66. Price L, Briley J, Haltiwanger S, et al. A meta-analysis of cranial electrotherapy stimulation in the treatment of depression. J Psychiatr Res. 2021;135:119-134. doi:10.1016/j.jpsychires.2020.12.043
67. Kirsch D, Gilula M. CES in the treatment of addictions: a review and meta-analysis. Pract Pain Manag. 2007;7(9).
68. Hao JJ, Mittelman M. Acupuncture: past, present, and future. Glob Adv Health Med. 2014;3(4):6-8. doi:10.7453/gahmj.2014.042
69. Napadow V, Ahn A, Longhurst J, et al. The status and future of acupuncture mechanism research. J Altern Complement Med. 2008;14(7):861-869. doi:10.1089/acm.2008.SAR-3
70. Smith CA, Armour M, Lee MS, et al. Acupuncture for depression. Cochrane Database Syst Rev. 2018;3(3):CD004046. doi:10.1002/14651858.CD004046.pub4
71. Tong QY, Liu R, Zhang K, et al. Can acupuncture therapy reduce preoperative anxiety? A systematic review and meta-analysis. J Integr Med. 2021;19(1):20-28. doi:10.1016/j.joim.2020.10.007
72. Usichenko TI, Hua K, Cummings M, et al. Auricular stimulation for preoperative anxiety – a systematic review and meta-analysis of randomized controlled clinical trials. J Clin Anesth. 2022;76:110581. doi:10.1016/j.jclinane.2021.110581
73. Zhou L, Hu X, Yu Z, et al. Efficacy and safety of acupuncture in the treatment of poststroke insomnia: a systematic review and meta-analysis of twenty-six randomized controlled trials. Evid Based Complement Alternat Med. 2022;2022:5188311. doi:10.1155/2022/5188311
74. Yin X, Gou M, Xu J, et al. Efficacy and safety of acupuncture treatment on primary insomnia: a randomized controlled trial. Sleep Med. 2017;37:193-200. doi:10.1016/j.sleep.2017.02.012
Interventional psychiatry: What are the next steps?
The explosion of interest in interventional psychiatry is highlighted by 2 recent reviews published in
Psychiatry’s failure to address these changes would be a dire error, as psychiatrists could lose control of our field’s advances and growth. But this creates an even larger question: what are the next steps we need to take? We believe interventional psychiatry must be recognized as its own psychiatric subspeciality, receive greater emphasis in psychiatry residency training, and be subject to standardization by professional organizations.
Psychiatry has incorporated procedures into patient care for almost 100 years, starting with electroconvulsive therapy (ECT) and insulin shock therapy in the 1930s.3,4 However, in the last 10 years, the rapid expansion of FDA approvals of neuromodulation procedures to treat psychiatric conditions (including vagus nerve stimulation in 2005, transcranial magnetic stimulation [TMS] in 2008, and the device exception granted for the use of deep brain stimulation in 2009) has produced the moniker “interventional psychiatry” for this unofficial psychiatric subspeciality.5,6
If we are to establish interventional psychiatry as a recognized subspeciality, it is important to create a universally accepted definition. We propose the term refer to therapeutic techniques or processes that may or may not be invasive but require special training to perform. Additionally, interventional psychiatry should include even minimally invasive procedures, such as ketamine infusions, medication implants, long-acting injectable (LAI) medications, and processes that require a Risk Evaluation and Mitigation Strategy (REMS), such as those utilized with clozapine, esketamine, or olanzapine for extended-release injectable suspension7 (see “Risk Evaluation and Mitigation Strategy programs: How they can be improved”). The proportions of clinicians who prescribe clozapine (7%)8 or LAIs (32.1% to 77.7%, depending on the patient population being treated)9,10 is evidence that the interventional nature of these treatments creates obstacles to their use.
This vacuum of adequate training among psychiatrists has caused interventional psychiatry to grow beyond the confines of the psychiatric field. In most metropolitan areas of the United States, there are clinicians who focus on a specific interventional treatment, such as ketamine infusions or TMS administration. The creation of these specialized clinics has frequently been pioneered by nonpsychiatrists, such as anesthesiologists. This may be attributed to these clinicians’ level of comfort with procedures, or because they possess an infrastructure within their practice that facilitates delivery of the services. In certain states with independent-practice laws, midlevel clinicians are granted permission to open these clinics. However, having nonpsychiatrists provide these treatments to patients with complex psychiatric disorders without psychiatrist involvement makes it less likely that the appropriateness of treatment will be determined, or that the treatment will be incorporated into the patient’s overall biopsychosocial treatment plan.
A gap in training
There is evidence the growth of interventional psychiatry has exceeded the capacity of the current training infrastructure to provide trainees with adequate exposure to these procedures. The Accreditation Council for Graduate Medical Education requires that psychiatry residents be trained in the indications for and use of ECT and neuromodulation therapies but does not provide any specifics about how this training should occur,11 and the Psychiatry Milestones do not indicate how competency in these therapies can be achieved.12 Most trainees have exposure to some interventional treatments, such as ECT or clozapine administration, during residency. However, in 1 survey, only 63% of residents had prescribed clozapine, and 83% indicated they wanted additional experience.13 In a survey of 91 training programs, 75% stated that ECT was required of residents, but 37% estimated that a typical resident would participate in <10 treatments.14 Even more surprising, 27% estimated that the typical resident would care for <5 patients receiving ECT.14
Addressing the changing role of interventional practices in our field must occur on multiple levels, starting with a core curriculum during residency training, expanded learning opportunities for residents with a specific interest in interventional psychiatry, and, most important, a formal interventional psychiatry fellowship leading to certification from the American Board of Medical Specialties.5,6 There are growing numbers of 1-year fellowship programs that offer extensive experiences in neuromodulation and novel pharmacologic treatment and may produce the next generation of leaders in this field. However, training in interventional psychiatry techniques for practicing psychiatrists wishing to expand their treatment offerings is generally quite limited.
Oversight of interventional psychiatry training should be performed by peers. Therefore, creation of an interventional psychiatry society, or a work group within a larger organization, is necessary. While much of this already exists, it is fragmented into associations focused on unique aspects of interventional psychiatry, such as just ECT (eg, International Society for ECT and Neurostimulation), just TMS (eg, Clinical TMS Society), or just ketamine (eg, the American Society of Ketamine Physicians). Despite disparate foci, the goal would be for all to unite into a parent interventional organization that can face these challenges. These organizations have already united a core of individual interventional psychiatrists who can lead psychiatry into the future. They can provide input into guidelines, minimal standards, procedures, protocols, and outcome measures. They also can address any ethical issues that may arise with the use of more invasive treatments.
Change, especially the monumental changes in practice that accompany interventional psychiatry, is both exciting and intimidating. However, certain “growing pains” along the way require urgent consideration. Ultimately, as a field, we either adapt to change or get left behind.
1. Arbuck D, Farooqui A, El-Mallakh RS. Interventional psychiatry (Part 1). Current Psychiatry. 2023;22(5):25-35. doi:10.12788/cp.0356
2. Arbuck D, Farooqui A, El-Mallakh RS. Interventional psychiatry (Part 2). Current Psychiatry. 2023;22(7):27-35. doi:10.12788/cp.0364
3. Jones K. Insulin coma therapy in schizophrenia. J R Soc Med. 2000;93(3):147-149. doi:10.1177/014107680009300313
4. Gazdag G, Ungvari GS. Electroconvulsive therapy: 80 years old and still going strong. World J Psychiatry. 2019;9(1):1-6. doi:10.5498/wjp.v9.i1.1
5. Williams NR, Taylor JJ, Snipes JM, et al. Interventional psychiatry: how should psychiatric educators incorporate neuromodulation into training? Acad Psychiatry. 2014;38(2):168-176. doi:10.1007/s40596-014-0050-x
6. Trapp NT, Williams NR. The future of training and practice in neuromodulation: an interventional psychiatry perspective. Front Psychiatry. 2021;12:734487. doi:10.3389/fpsyt.2021.734487
7. Vincent KM, Ryan M, Palmer E, et al. Interventional psychiatry. Postgrad Med. 2020;132(7):573-574. doi:10.1080/00325481.2020.1727671
8. Tang Y, Horvitz-Lennon M, Gellad WF, et al. Prescribing of clozapine and antipsychotic polypharmacy for schizophrenia in a large Medicaid program. Psychiatr Serv. 2017;68(6):579-586. doi:10.1176/appi.ps.201600041
9. Zhdanava M, Starr HL, Lefebvre P, et al. Understanding the health system conditions affecting the use of long-acting injectable antipsychotics in the treatment of schizophrenia in clinical practice: a US healthcare provider survey. Neuropsychiatr Dis Treat. 2022;18:1479-1493. doi:10.2147/NDT.S369494
10. Bunting SR, Chalmers K, Yohanna D, et al. Prescription of long-acting injectable antipsychotic medications among outpatient mental health care service providers. Psychiatr Serv. 2023:appips20220586. doi:10.1176/appi.ps.20220586
11. Accreditation Council for Graduate Medical Education. Common program requirements. July 2022. Accessed June 6, 2023. https://www.acgme.org/programs-and-institutions/programs/common-program-requirements
12. Kinzie JM, DeJong SM, Edgar L, et al. Psychiatry Milestones 2.0: using the supplemental guide to create a shared model of the development of professional identity and expertise. Acad Psychiatry. 2021;45(4):500-505. doi:10.1007/s40596-021-01455-6
13. Singh B, Hughes AJ, Roerig JL. Comfort level and barriers to the appropriate use of clozapine: a preliminary survey of US psychiatric residents. Acad Psychiatry. 2020;44(1):53-58 doi:10.1007/s40596-019-01134-7
14. Dinwiddie SH, Spitz D. Resident education in electroconvulsive therapy. J ECT. 2010;26(4):310-316. doi:10.1097/YCT.0b013e3181cb5f78
The explosion of interest in interventional psychiatry is highlighted by 2 recent reviews published in
Psychiatry’s failure to address these changes would be a dire error, as psychiatrists could lose control of our field’s advances and growth. But this creates an even larger question: what are the next steps we need to take? We believe interventional psychiatry must be recognized as its own psychiatric subspeciality, receive greater emphasis in psychiatry residency training, and be subject to standardization by professional organizations.
Psychiatry has incorporated procedures into patient care for almost 100 years, starting with electroconvulsive therapy (ECT) and insulin shock therapy in the 1930s.3,4 However, in the last 10 years, the rapid expansion of FDA approvals of neuromodulation procedures to treat psychiatric conditions (including vagus nerve stimulation in 2005, transcranial magnetic stimulation [TMS] in 2008, and the device exception granted for the use of deep brain stimulation in 2009) has produced the moniker “interventional psychiatry” for this unofficial psychiatric subspeciality.5,6
If we are to establish interventional psychiatry as a recognized subspeciality, it is important to create a universally accepted definition. We propose the term refer to therapeutic techniques or processes that may or may not be invasive but require special training to perform. Additionally, interventional psychiatry should include even minimally invasive procedures, such as ketamine infusions, medication implants, long-acting injectable (LAI) medications, and processes that require a Risk Evaluation and Mitigation Strategy (REMS), such as those utilized with clozapine, esketamine, or olanzapine for extended-release injectable suspension7 (see “Risk Evaluation and Mitigation Strategy programs: How they can be improved”). The proportions of clinicians who prescribe clozapine (7%)8 or LAIs (32.1% to 77.7%, depending on the patient population being treated)9,10 is evidence that the interventional nature of these treatments creates obstacles to their use.
This vacuum of adequate training among psychiatrists has caused interventional psychiatry to grow beyond the confines of the psychiatric field. In most metropolitan areas of the United States, there are clinicians who focus on a specific interventional treatment, such as ketamine infusions or TMS administration. The creation of these specialized clinics has frequently been pioneered by nonpsychiatrists, such as anesthesiologists. This may be attributed to these clinicians’ level of comfort with procedures, or because they possess an infrastructure within their practice that facilitates delivery of the services. In certain states with independent-practice laws, midlevel clinicians are granted permission to open these clinics. However, having nonpsychiatrists provide these treatments to patients with complex psychiatric disorders without psychiatrist involvement makes it less likely that the appropriateness of treatment will be determined, or that the treatment will be incorporated into the patient’s overall biopsychosocial treatment plan.
A gap in training
There is evidence the growth of interventional psychiatry has exceeded the capacity of the current training infrastructure to provide trainees with adequate exposure to these procedures. The Accreditation Council for Graduate Medical Education requires that psychiatry residents be trained in the indications for and use of ECT and neuromodulation therapies but does not provide any specifics about how this training should occur,11 and the Psychiatry Milestones do not indicate how competency in these therapies can be achieved.12 Most trainees have exposure to some interventional treatments, such as ECT or clozapine administration, during residency. However, in 1 survey, only 63% of residents had prescribed clozapine, and 83% indicated they wanted additional experience.13 In a survey of 91 training programs, 75% stated that ECT was required of residents, but 37% estimated that a typical resident would participate in <10 treatments.14 Even more surprising, 27% estimated that the typical resident would care for <5 patients receiving ECT.14
Addressing the changing role of interventional practices in our field must occur on multiple levels, starting with a core curriculum during residency training, expanded learning opportunities for residents with a specific interest in interventional psychiatry, and, most important, a formal interventional psychiatry fellowship leading to certification from the American Board of Medical Specialties.5,6 There are growing numbers of 1-year fellowship programs that offer extensive experiences in neuromodulation and novel pharmacologic treatment and may produce the next generation of leaders in this field. However, training in interventional psychiatry techniques for practicing psychiatrists wishing to expand their treatment offerings is generally quite limited.
Oversight of interventional psychiatry training should be performed by peers. Therefore, creation of an interventional psychiatry society, or a work group within a larger organization, is necessary. While much of this already exists, it is fragmented into associations focused on unique aspects of interventional psychiatry, such as just ECT (eg, International Society for ECT and Neurostimulation), just TMS (eg, Clinical TMS Society), or just ketamine (eg, the American Society of Ketamine Physicians). Despite disparate foci, the goal would be for all to unite into a parent interventional organization that can face these challenges. These organizations have already united a core of individual interventional psychiatrists who can lead psychiatry into the future. They can provide input into guidelines, minimal standards, procedures, protocols, and outcome measures. They also can address any ethical issues that may arise with the use of more invasive treatments.
Change, especially the monumental changes in practice that accompany interventional psychiatry, is both exciting and intimidating. However, certain “growing pains” along the way require urgent consideration. Ultimately, as a field, we either adapt to change or get left behind.
The explosion of interest in interventional psychiatry is highlighted by 2 recent reviews published in
Psychiatry’s failure to address these changes would be a dire error, as psychiatrists could lose control of our field’s advances and growth. But this creates an even larger question: what are the next steps we need to take? We believe interventional psychiatry must be recognized as its own psychiatric subspeciality, receive greater emphasis in psychiatry residency training, and be subject to standardization by professional organizations.
Psychiatry has incorporated procedures into patient care for almost 100 years, starting with electroconvulsive therapy (ECT) and insulin shock therapy in the 1930s.3,4 However, in the last 10 years, the rapid expansion of FDA approvals of neuromodulation procedures to treat psychiatric conditions (including vagus nerve stimulation in 2005, transcranial magnetic stimulation [TMS] in 2008, and the device exception granted for the use of deep brain stimulation in 2009) has produced the moniker “interventional psychiatry” for this unofficial psychiatric subspeciality.5,6
If we are to establish interventional psychiatry as a recognized subspeciality, it is important to create a universally accepted definition. We propose the term refer to therapeutic techniques or processes that may or may not be invasive but require special training to perform. Additionally, interventional psychiatry should include even minimally invasive procedures, such as ketamine infusions, medication implants, long-acting injectable (LAI) medications, and processes that require a Risk Evaluation and Mitigation Strategy (REMS), such as those utilized with clozapine, esketamine, or olanzapine for extended-release injectable suspension7 (see “Risk Evaluation and Mitigation Strategy programs: How they can be improved”). The proportions of clinicians who prescribe clozapine (7%)8 or LAIs (32.1% to 77.7%, depending on the patient population being treated)9,10 is evidence that the interventional nature of these treatments creates obstacles to their use.
This vacuum of adequate training among psychiatrists has caused interventional psychiatry to grow beyond the confines of the psychiatric field. In most metropolitan areas of the United States, there are clinicians who focus on a specific interventional treatment, such as ketamine infusions or TMS administration. The creation of these specialized clinics has frequently been pioneered by nonpsychiatrists, such as anesthesiologists. This may be attributed to these clinicians’ level of comfort with procedures, or because they possess an infrastructure within their practice that facilitates delivery of the services. In certain states with independent-practice laws, midlevel clinicians are granted permission to open these clinics. However, having nonpsychiatrists provide these treatments to patients with complex psychiatric disorders without psychiatrist involvement makes it less likely that the appropriateness of treatment will be determined, or that the treatment will be incorporated into the patient’s overall biopsychosocial treatment plan.
A gap in training
There is evidence the growth of interventional psychiatry has exceeded the capacity of the current training infrastructure to provide trainees with adequate exposure to these procedures. The Accreditation Council for Graduate Medical Education requires that psychiatry residents be trained in the indications for and use of ECT and neuromodulation therapies but does not provide any specifics about how this training should occur,11 and the Psychiatry Milestones do not indicate how competency in these therapies can be achieved.12 Most trainees have exposure to some interventional treatments, such as ECT or clozapine administration, during residency. However, in 1 survey, only 63% of residents had prescribed clozapine, and 83% indicated they wanted additional experience.13 In a survey of 91 training programs, 75% stated that ECT was required of residents, but 37% estimated that a typical resident would participate in <10 treatments.14 Even more surprising, 27% estimated that the typical resident would care for <5 patients receiving ECT.14
Addressing the changing role of interventional practices in our field must occur on multiple levels, starting with a core curriculum during residency training, expanded learning opportunities for residents with a specific interest in interventional psychiatry, and, most important, a formal interventional psychiatry fellowship leading to certification from the American Board of Medical Specialties.5,6 There are growing numbers of 1-year fellowship programs that offer extensive experiences in neuromodulation and novel pharmacologic treatment and may produce the next generation of leaders in this field. However, training in interventional psychiatry techniques for practicing psychiatrists wishing to expand their treatment offerings is generally quite limited.
Oversight of interventional psychiatry training should be performed by peers. Therefore, creation of an interventional psychiatry society, or a work group within a larger organization, is necessary. While much of this already exists, it is fragmented into associations focused on unique aspects of interventional psychiatry, such as just ECT (eg, International Society for ECT and Neurostimulation), just TMS (eg, Clinical TMS Society), or just ketamine (eg, the American Society of Ketamine Physicians). Despite disparate foci, the goal would be for all to unite into a parent interventional organization that can face these challenges. These organizations have already united a core of individual interventional psychiatrists who can lead psychiatry into the future. They can provide input into guidelines, minimal standards, procedures, protocols, and outcome measures. They also can address any ethical issues that may arise with the use of more invasive treatments.
Change, especially the monumental changes in practice that accompany interventional psychiatry, is both exciting and intimidating. However, certain “growing pains” along the way require urgent consideration. Ultimately, as a field, we either adapt to change or get left behind.
1. Arbuck D, Farooqui A, El-Mallakh RS. Interventional psychiatry (Part 1). Current Psychiatry. 2023;22(5):25-35. doi:10.12788/cp.0356
2. Arbuck D, Farooqui A, El-Mallakh RS. Interventional psychiatry (Part 2). Current Psychiatry. 2023;22(7):27-35. doi:10.12788/cp.0364
3. Jones K. Insulin coma therapy in schizophrenia. J R Soc Med. 2000;93(3):147-149. doi:10.1177/014107680009300313
4. Gazdag G, Ungvari GS. Electroconvulsive therapy: 80 years old and still going strong. World J Psychiatry. 2019;9(1):1-6. doi:10.5498/wjp.v9.i1.1
5. Williams NR, Taylor JJ, Snipes JM, et al. Interventional psychiatry: how should psychiatric educators incorporate neuromodulation into training? Acad Psychiatry. 2014;38(2):168-176. doi:10.1007/s40596-014-0050-x
6. Trapp NT, Williams NR. The future of training and practice in neuromodulation: an interventional psychiatry perspective. Front Psychiatry. 2021;12:734487. doi:10.3389/fpsyt.2021.734487
7. Vincent KM, Ryan M, Palmer E, et al. Interventional psychiatry. Postgrad Med. 2020;132(7):573-574. doi:10.1080/00325481.2020.1727671
8. Tang Y, Horvitz-Lennon M, Gellad WF, et al. Prescribing of clozapine and antipsychotic polypharmacy for schizophrenia in a large Medicaid program. Psychiatr Serv. 2017;68(6):579-586. doi:10.1176/appi.ps.201600041
9. Zhdanava M, Starr HL, Lefebvre P, et al. Understanding the health system conditions affecting the use of long-acting injectable antipsychotics in the treatment of schizophrenia in clinical practice: a US healthcare provider survey. Neuropsychiatr Dis Treat. 2022;18:1479-1493. doi:10.2147/NDT.S369494
10. Bunting SR, Chalmers K, Yohanna D, et al. Prescription of long-acting injectable antipsychotic medications among outpatient mental health care service providers. Psychiatr Serv. 2023:appips20220586. doi:10.1176/appi.ps.20220586
11. Accreditation Council for Graduate Medical Education. Common program requirements. July 2022. Accessed June 6, 2023. https://www.acgme.org/programs-and-institutions/programs/common-program-requirements
12. Kinzie JM, DeJong SM, Edgar L, et al. Psychiatry Milestones 2.0: using the supplemental guide to create a shared model of the development of professional identity and expertise. Acad Psychiatry. 2021;45(4):500-505. doi:10.1007/s40596-021-01455-6
13. Singh B, Hughes AJ, Roerig JL. Comfort level and barriers to the appropriate use of clozapine: a preliminary survey of US psychiatric residents. Acad Psychiatry. 2020;44(1):53-58 doi:10.1007/s40596-019-01134-7
14. Dinwiddie SH, Spitz D. Resident education in electroconvulsive therapy. J ECT. 2010;26(4):310-316. doi:10.1097/YCT.0b013e3181cb5f78
1. Arbuck D, Farooqui A, El-Mallakh RS. Interventional psychiatry (Part 1). Current Psychiatry. 2023;22(5):25-35. doi:10.12788/cp.0356
2. Arbuck D, Farooqui A, El-Mallakh RS. Interventional psychiatry (Part 2). Current Psychiatry. 2023;22(7):27-35. doi:10.12788/cp.0364
3. Jones K. Insulin coma therapy in schizophrenia. J R Soc Med. 2000;93(3):147-149. doi:10.1177/014107680009300313
4. Gazdag G, Ungvari GS. Electroconvulsive therapy: 80 years old and still going strong. World J Psychiatry. 2019;9(1):1-6. doi:10.5498/wjp.v9.i1.1
5. Williams NR, Taylor JJ, Snipes JM, et al. Interventional psychiatry: how should psychiatric educators incorporate neuromodulation into training? Acad Psychiatry. 2014;38(2):168-176. doi:10.1007/s40596-014-0050-x
6. Trapp NT, Williams NR. The future of training and practice in neuromodulation: an interventional psychiatry perspective. Front Psychiatry. 2021;12:734487. doi:10.3389/fpsyt.2021.734487
7. Vincent KM, Ryan M, Palmer E, et al. Interventional psychiatry. Postgrad Med. 2020;132(7):573-574. doi:10.1080/00325481.2020.1727671
8. Tang Y, Horvitz-Lennon M, Gellad WF, et al. Prescribing of clozapine and antipsychotic polypharmacy for schizophrenia in a large Medicaid program. Psychiatr Serv. 2017;68(6):579-586. doi:10.1176/appi.ps.201600041
9. Zhdanava M, Starr HL, Lefebvre P, et al. Understanding the health system conditions affecting the use of long-acting injectable antipsychotics in the treatment of schizophrenia in clinical practice: a US healthcare provider survey. Neuropsychiatr Dis Treat. 2022;18:1479-1493. doi:10.2147/NDT.S369494
10. Bunting SR, Chalmers K, Yohanna D, et al. Prescription of long-acting injectable antipsychotic medications among outpatient mental health care service providers. Psychiatr Serv. 2023:appips20220586. doi:10.1176/appi.ps.20220586
11. Accreditation Council for Graduate Medical Education. Common program requirements. July 2022. Accessed June 6, 2023. https://www.acgme.org/programs-and-institutions/programs/common-program-requirements
12. Kinzie JM, DeJong SM, Edgar L, et al. Psychiatry Milestones 2.0: using the supplemental guide to create a shared model of the development of professional identity and expertise. Acad Psychiatry. 2021;45(4):500-505. doi:10.1007/s40596-021-01455-6
13. Singh B, Hughes AJ, Roerig JL. Comfort level and barriers to the appropriate use of clozapine: a preliminary survey of US psychiatric residents. Acad Psychiatry. 2020;44(1):53-58 doi:10.1007/s40596-019-01134-7
14. Dinwiddie SH, Spitz D. Resident education in electroconvulsive therapy. J ECT. 2010;26(4):310-316. doi:10.1097/YCT.0b013e3181cb5f78
A pivot in training: My path to reproductive psychiatry
In March 2020, as I was wheeling my patient into the operating room to perform a Caesarean section, covered head-to-toe in COVID personal protective equipment, my phone rang. It was Jody Schindelheim, MD, Director of the Psychiatry Residency Program at Tufts Medical Center in Boston, calling to offer me a PGY-2 spot in their program.
As COVID upended the world, I was struggling with my own major change. My path had been planned since before medical school: I would grind through a 4-year OB/GYN residency, complete a fellowship, and establish myself as a reproductive endocrinology and infertility specialist. My personal statement emphasized my dream that no woman should be made to feel useless based on infertility. OB/GYN, genetics, and ultrasound were my favorite rotations at the Albert Einstein College of Medicine in the Bronx.
However, 6 months into my OB/GYN intern year, I grew curious about the possibility of a future in reproductive psychiatry and women’s mental health. This decision was not easy. As someone who loved the adrenaline rush of delivering babies and performing surgery, I had paid little attention to psychiatry in medical school. However, my experience in gynecologic oncology in January 2020 made me realize my love of stories and trauma-informed care. I recall a woman, cachectic with only days left to live due to ovarian cancer, talking to me about her trauma and the power of her lifelong partner. Another woman, experiencing complications from chemotherapy to treat fallopian tube cancer, shared about her coping skill of chair yoga.
Fulfilling an unmet need
As I spent time with these 2 women and heard their stories, I felt compelled to help them with these psychological challenges. As a gynecologist, I addressed their physical needs, but not their personal needs. I spoke to many psychiatrists, including reproductive psychiatrists, in New York, who shared their stories and taught me about the prevalence of postpartum depression and psychosis. After caring for hundreds of pregnant and postpartum women in the Bronx, I thought about the unmet need for women’s mental health and how this career change could still fulfill my purpose of helping women feel empowered regardless of their fertility status.
In the inpatient and outpatient settings at Tufts, I have loved hearing my patients’ stories and providing continuity of care with medical management and therapy. My mentors in reproductive psychiatry inspired me to create the Reproductive Psychiatry Trainee Interest Group (https://www.repropsychtrainees.com), a national group for the burgeoning field that now has more than 650 members. With monthly lectures, journal clubs, and book clubs, I have surrounded myself with like-minded individuals who love learning about the perinatal, postpartum, and perimenopausal experiences.
As I prepare to begin a full-time faculty position in psychiatry at the University of Pennsylvania, I know I have found my joy and my calling. I once feared the life of a psychiatrist would be too sedentary for someone accustomed to the pace of OB/GYN. Now I know that my patients’ stories are all the motivation I need.
In March 2020, as I was wheeling my patient into the operating room to perform a Caesarean section, covered head-to-toe in COVID personal protective equipment, my phone rang. It was Jody Schindelheim, MD, Director of the Psychiatry Residency Program at Tufts Medical Center in Boston, calling to offer me a PGY-2 spot in their program.
As COVID upended the world, I was struggling with my own major change. My path had been planned since before medical school: I would grind through a 4-year OB/GYN residency, complete a fellowship, and establish myself as a reproductive endocrinology and infertility specialist. My personal statement emphasized my dream that no woman should be made to feel useless based on infertility. OB/GYN, genetics, and ultrasound were my favorite rotations at the Albert Einstein College of Medicine in the Bronx.
However, 6 months into my OB/GYN intern year, I grew curious about the possibility of a future in reproductive psychiatry and women’s mental health. This decision was not easy. As someone who loved the adrenaline rush of delivering babies and performing surgery, I had paid little attention to psychiatry in medical school. However, my experience in gynecologic oncology in January 2020 made me realize my love of stories and trauma-informed care. I recall a woman, cachectic with only days left to live due to ovarian cancer, talking to me about her trauma and the power of her lifelong partner. Another woman, experiencing complications from chemotherapy to treat fallopian tube cancer, shared about her coping skill of chair yoga.
Fulfilling an unmet need
As I spent time with these 2 women and heard their stories, I felt compelled to help them with these psychological challenges. As a gynecologist, I addressed their physical needs, but not their personal needs. I spoke to many psychiatrists, including reproductive psychiatrists, in New York, who shared their stories and taught me about the prevalence of postpartum depression and psychosis. After caring for hundreds of pregnant and postpartum women in the Bronx, I thought about the unmet need for women’s mental health and how this career change could still fulfill my purpose of helping women feel empowered regardless of their fertility status.
In the inpatient and outpatient settings at Tufts, I have loved hearing my patients’ stories and providing continuity of care with medical management and therapy. My mentors in reproductive psychiatry inspired me to create the Reproductive Psychiatry Trainee Interest Group (https://www.repropsychtrainees.com), a national group for the burgeoning field that now has more than 650 members. With monthly lectures, journal clubs, and book clubs, I have surrounded myself with like-minded individuals who love learning about the perinatal, postpartum, and perimenopausal experiences.
As I prepare to begin a full-time faculty position in psychiatry at the University of Pennsylvania, I know I have found my joy and my calling. I once feared the life of a psychiatrist would be too sedentary for someone accustomed to the pace of OB/GYN. Now I know that my patients’ stories are all the motivation I need.
In March 2020, as I was wheeling my patient into the operating room to perform a Caesarean section, covered head-to-toe in COVID personal protective equipment, my phone rang. It was Jody Schindelheim, MD, Director of the Psychiatry Residency Program at Tufts Medical Center in Boston, calling to offer me a PGY-2 spot in their program.
As COVID upended the world, I was struggling with my own major change. My path had been planned since before medical school: I would grind through a 4-year OB/GYN residency, complete a fellowship, and establish myself as a reproductive endocrinology and infertility specialist. My personal statement emphasized my dream that no woman should be made to feel useless based on infertility. OB/GYN, genetics, and ultrasound were my favorite rotations at the Albert Einstein College of Medicine in the Bronx.
However, 6 months into my OB/GYN intern year, I grew curious about the possibility of a future in reproductive psychiatry and women’s mental health. This decision was not easy. As someone who loved the adrenaline rush of delivering babies and performing surgery, I had paid little attention to psychiatry in medical school. However, my experience in gynecologic oncology in January 2020 made me realize my love of stories and trauma-informed care. I recall a woman, cachectic with only days left to live due to ovarian cancer, talking to me about her trauma and the power of her lifelong partner. Another woman, experiencing complications from chemotherapy to treat fallopian tube cancer, shared about her coping skill of chair yoga.
Fulfilling an unmet need
As I spent time with these 2 women and heard their stories, I felt compelled to help them with these psychological challenges. As a gynecologist, I addressed their physical needs, but not their personal needs. I spoke to many psychiatrists, including reproductive psychiatrists, in New York, who shared their stories and taught me about the prevalence of postpartum depression and psychosis. After caring for hundreds of pregnant and postpartum women in the Bronx, I thought about the unmet need for women’s mental health and how this career change could still fulfill my purpose of helping women feel empowered regardless of their fertility status.
In the inpatient and outpatient settings at Tufts, I have loved hearing my patients’ stories and providing continuity of care with medical management and therapy. My mentors in reproductive psychiatry inspired me to create the Reproductive Psychiatry Trainee Interest Group (https://www.repropsychtrainees.com), a national group for the burgeoning field that now has more than 650 members. With monthly lectures, journal clubs, and book clubs, I have surrounded myself with like-minded individuals who love learning about the perinatal, postpartum, and perimenopausal experiences.
As I prepare to begin a full-time faculty position in psychiatry at the University of Pennsylvania, I know I have found my joy and my calling. I once feared the life of a psychiatrist would be too sedentary for someone accustomed to the pace of OB/GYN. Now I know that my patients’ stories are all the motivation I need.
Using apps in clinical practice: 8 studies
COVID-19’s increased demand on the mental health care delivery system led to expanded utilization of technology-based solutions, including digital tools to deliver care.1 Technology-based solutions include both synchronous telehealth (eg, real-time interactive audio/video visits) and asynchronous tools such as smartphone applications (apps). Both real-time telehealth and apps continue to gain popularity. More than 10,000 mental health–related apps are available, and that number continues to rise.2 Numerous web- or mobile-based apps are available to aid in the treatment of various psychiatric conditions, including generalized anxiety disorder (GAD), major depressive disorder, insomnia, and posttraumatic stress disorder (PTSD).
Clinicians may find it challenging to choose the best psychiatry-related apps to recommend to patients. This dilemma calls for an approach to help clinicians select apps that are safe and effective.2 The American Psychiatric Association provides information to help mental health professionals navigate these issues and identify which aspects to consider when selecting an app for clinical use.3 The M-Health Index and Navigation Database also provides a set of objective evaluative criteria and offers guidance on choosing apps.4
In this article, we review 8 randomized controlled trials (RCTs) of mental health–related apps. We took several steps to ensure the RCTs we included were impactful and meaningful. First, we conducted a general search using mainstream search engines to assess which psychiatric apps were most popular for use in clinical practice. Using this list, we conducted a scholarly search engine query of RCTs using the name of the apps as a search parameter along with the following keywords: “mobile,” “web,” “applications,” and “psychiatry.” This search yielded approximately 50 results, which were narrowed down based on content and interest to a list of 8 articles (Table5-12). These articles were then graded using the limitations of each study as the primary substrate for evaluation.

1. Linardon J, Shatte A, Rosato J, et al. Efficacy of a transdiagnostic cognitive-behavioral intervention for eating disorder psychopathology delivered through a smartphone app: a randomized controlled trial. Psychol Med. 2022;52(9):1679-1690. doi:10.1017/S0033291720003426
Many patients with eating disorders are unable to receive effective treatment due to problems with accessing health care. Smartphone apps may help bridge the treatment gap for patients in this position. Linardon et al5 developed an app that uses the principles of cognitive-behavioral therapy (CBT) for treating eating disorders and conducted this study to evaluate its effectiveness.
Study design
- This RCT assigned individuals who reported episodes of binge eating to a group that used a mobile app (n = 197) or to a waiting list (n = 195). At baseline, 42% of participants exhibited diagnostic-level symptoms of bulimia nervosa and 31% had symptoms of binge-eating disorder.
- Assessments took place at baseline, Week 4, and Week 8.
- The primary outcome was global levels of eating disorder psychopathology.
- Secondary outcomes were other eating disorder symptoms, impairment, and distress.
Outcomes
- Compared to the control group, participants who used the mobile app reported greater reductions in global eating disorder psychopathology (d = -0.80).
- Significant effects were also observed for secondary outcomes except compensatory behavior frequency.
- Overall, participants reported they were satisfied with the app.
Continue to: Conclusions/limitations
Conclusions/limitations
- Findings show this app could potentially be a cost-effective and easily accessible option for patients who cannot receive standard treatment for eating disorders.
- Limitations: The overall posttest attrition rate was 35%.
2. Christoforou M, Sáez Fonseca JA, Tsakanikos E. Two novel cognitive behavioral therapy–based mobile apps for agoraphobia: randomized controlled trial. J Med Internet Res. 2017;19(11):e398. doi:10.2196/jmir.7747
CBT is generally the most accepted first-line treatment for agoraphobia. However, numerous barriers to obtaining CBT can prevent successful treatment. Limited research has evaluated the efficacy of apps for treating agoraphobia. Christoforou et al6 conducted an RCT to determine the effectiveness of a self-guided smartphone app for improving agoraphobic symptoms, compared to a mobile app used to treat anxiety.
Study design
- Participants (N = 170) who self-identified as having agoraphobia were randomly assigned to use a smartphone app designed to target agoraphobia (Agoraphobia Free) or a smartphone app designed to help with symptoms of anxiety (Stress Free) for 12 weeks. Both apps were based on established cognitive behavioral principles.
- Assessment occurred at baseline, midpoint, and end point.
- The primary outcome was symptom severity as measured by the Panic and Agoraphobia Scale (PAS).
Outcomes
- Both groups experienced statistically significant improvements in symptom severity over time. The differences in PAS score were -5.97 (95% CI, -8.49 to -3.44, P < .001) for Agoraphobia Free and -6.35 (95% CI, -8.82 to -3.87, P < .001) for Stress Free.
- There were no significant between-group differences in symptom severity.
Continue to: Conclusions/limitations
Conclusions/limitations
- This study is the first RCT to show that patients with agoraphobia could benefit from mobile-based interventions.
- Limitations: There was no waitlist control group. Limited information was collected about participant characteristics; there were no data on comorbid disorders, other psychological or physiological treatments, or other demographic characteristics such as ethnicity or computer literacy.
3. Everitt N, Broadbent J, Richardson B, et al. Exploring the features of an app-based just-in-time intervention for depression. J Affect Disord. 2021;291:279-287. doi:10.1016/j.jad.2021.05.021
The apps MoodTracker, ImproveYourMood, and ImproveYourMood+ deliver content “just in time” (in response to acute negative symptoms) to help patients with depression. In an RCT, Everitt et al7 evaluated delivering acute care for depressive mood states via a smartphone app. They sought to delineate whether symptom improvement was due to microintervention content, mood augmentation, or just-in-time prompts to use content.
Study design
- Participants (N = 235) from the general population who said they wanted to improve their mood were randomly assigned to a waitlist control group (n = 55) or 1 of 3 intervention groups: MoodTracker (monitoring-only; n = 58), ImproveYourMood (monitoring and content; n = 62), or ImproveYourMood+ (monitoring, content, and prompts; n = 60).
- The microintervention content provided by these apps consisted of 4 audio files of brief (2- to 3-minute) mindfulness and relaxation exercises. Participants used the assigned app for 3 weeks.
- Depressive symptoms, anxiety symptoms, and negative automatic thoughts were assessed at baseline, immediately following the intervention, and 1 month after the intervention using the 9-item Patient Health Questionnaire (PHQ-9), 7-item GAD scale (GAD-7), and 8-item Automatic Thoughts Questionnaire, respectively.
Outcomes
- Compared to the waitlist control group, participants in the ImproveYourMood group showed greater declines in depressive symptoms and anxiety symptoms (at follow-up only), and negative automatic thoughts (at both postintervention and follow-up).
- Those in the ImproveYourMood+ group only showed significantly greater improvements for automatic negative thoughts (at postintervention).
- MoodTracker participants did not differ from waitlist controls for any variables at any timepoints.
Continue to: Conclusions/limitations
Conclusions/limitations
- This study suggests that using microinterventions in acute settings can effectively reduce depressive symptoms both as they occur, and 1 to 2 months later.
- Limitations: The study featured a naturalistic design, where participants self-selected whether they wanted to use the program. Participants did not complete eligibility assessments or receive compensation, and the study had high dropout rates, ranging from 20% for the waitlist control group to 67% for the ImproveYourMood+ group.
4. McLean C, Davis CA, Miller M, et al. The effects of an exposure-based mobile app on symptoms of posttraumatic stress disorder in veterans: pilot randomized controlled trial. JMIR Mhealth Uhealth. 2022;10(11):e38951. doi:10.2196/38951
Veterans with PTSD face barriers when receiving trauma-focused treatments such as exposure therapy or CBT. Smartphone apps may help veterans self-treat and self-manage their PTSD symptoms. McLean et al8 studied the efficacy of Renew, a smartphone app that uses exposure therapy and social support to treat PTSD.
Study design
- In this pilot RCT, 93 veterans with clinically significant PTSD symptoms were randomly assigned to use the Renew app with and without support from a research staff member (active use group) or to a waitlist (delayed use group) for 6 weeks.
- The PTSD Checklist for DSM-5 (PCL-5) was used to measure PTSD symptoms at preintervention, postintervention, and 6-week follow-up.
- Most participants (69%) were women, and the mean age was 49.
Outcomes
- Compared to the delayed use group, participants in the active use group experienced a larger decrease in PCL-5 score (-6.14 vs -1.84). However, this difference was not statistically significant (P = .29), and the effect size was small (d = -0.39).
- There was no difference in engagement with the app between participants who received support from a research staff member and those who did not receive such support.
Continue to: Conclusions/limitations
Conclusions/limitations
- Renew may show promise as a tool to reduce PTSD symptoms in veterans.
- Educating family and friends on how to best support a patient using a mobile mental health app may help improve the efficacy of Renew and increase app engagement.
- Limitations: Because the study was conducted in veterans, the results may not be generalizable to other populations. Because most data collection occurred during the first wave of the COVID-19 pandemic in the United States, COVID-19–related stress may have impacted PTSD symptoms, app engagement, or outcomes.
5. Graham AK, Greene CJ, Kwasny MJ, et al. Coached mobile app platform for the treatment of depression and anxiety among primary care patients: a randomized clinical trial. JAMA Psychiatry. 2020;77(9):906-914. doi:10.1001/jamapsychiatry.2020.1011
Many cases of depression and anxiety are initially treated in primary care settings. However, these settings may have limited resources and inadequate training, and mobile interventions might be helpful to augment patient care. Graham et al9 studied the mobile platform IntelliCare to determine its efficacy as a tool to be used in primary care settings to treat depression and anxiety.
Study design
- This RCT randomly assigned adult primary care patients (N = 146) who screened positive for depression on the PHQ-9 (score ≥10) or anxiety on the GAD-7 (score ≥8) to the coach-supported IntelliCare platform, which consisted of 5 clinically focused apps, or to a waitlist control group. Interventions were delivered over 8 weeks.
- Overall, 122 (83.6%) patients were diagnosed with depression and 131 (89.7%) were diagnosed with anxiety.
- The primary outcomes were changes in depression (as measured by change in PHQ-9 score) and anxiety (change in GAD-7 score) during the intervention period.
Outcomes
- Participants who used the IntelliCare platform had a greater reduction in depression and anxiety symptoms compared to waitlist controls, and changes were sustained over 2-month follow-up.
- The least square means (LSM) difference in depression scores at Week 4 was 2.91 (SE = 0.83; d = 0.43) and at Week 8 was 4.37 (SE = 0.83; d = 0.64). The LSM difference in anxiety scores at Week 4 was 2.51 (SE = 0.78; d = 0.41) and at Week 8 was 3.33 (SE = 0.76; d = 0.55).
- A median number of 93 and 98 sessions among participants with depression and anxiety were recorded, respectively, indicating high use of the IntelliCare platform.
Continue to: Conclusions/limitations
Conclusions/limitations
- The IntelliCare platform was shown to be effective in reducing depression and anxiety among primary care patients. Simple apps can be bundled together and used by patients in conjunction to treat their individual needs.
- Limitations: The study had a limited follow-up period and did not record participants’ use of other apps. Slightly more than one-half (56%) of participants were taking an antidepressant.
6. Wilhelm S, Weingarden H, Greenberg JL, et al. Efficacy of app-based cognitive behavioral therapy for body dysmorphic disorder with coach support: initial randomized controlled clinical trial. Psychother Psychosom. 2022;91(4):277-285. doi:10.1159/000524628
Body dysmorphic disorder (BDD) is a severe yet undertreated disorder. Apps can improve access to treatment for patients experiencing BDD. Wilhelm et al10 studied the usability and efficacy of a coach-supported app called Perspectives that was specifically designed for treating BDD. Perspectives provide CBT in 7 modules: psychoeducation, cognitive restructuring, exposure, response prevention, mindfulness, attention retraining, and relapse prevention.
Study design
- Adults (N = 80) with primary BDD were assigned to use the Perspectives app for 12 weeks or to a waitlist control group. Participants were predominately female (84%) and White (71%), with a mean age of 27.
- Coaches promoted engagement and answered questions via in-app messaging and phone calls.
- Blinded independent evaluators used the Yale-Brown Obsessive Compulsive Scale Modified for BDD (BDD-YBOCS) to measure BDD severity at baseline, midtreatment (Week 6), and end of treatment (Week 12).
- Secondary outcomes included BDD-related insight, depression, quality of life, and functioning. Various scales were used to measure these outcomes.
Outcomes
- In intent-to-treat analyses, patients who received CBT via the Perspectives app had significantly lower BDD severity at the end of treatment compared to the waitlist control group, with a mean (SD) BDD-YBOCS score of 16.8 (7.5) vs 26.7 (6.2), with P < .001 and d = 1.44.
- Slightly more than one-half (52%) of those who used Perspectives achieved full or partial remission, compared to 8% in the waitlist control group.
Continue to: Conclusions/limitations
Conclusions/limitations
- CBT delivered via the Perspectives app and a coach proved to be effective treatment for adults with BDD.
- Adoption of the application was relatively high; 86% of Perspectives users were very or mostly satisfied.
- Limitations: Because the participants in this study were predominantly female and White, the findings might not be generalizable to other populations.
7. Kuhn E, Miller KE, Puran D, et al. A pilot randomized controlled trial of the Insomnia Coach mobile app to assess its feasibility, acceptability, and potential efficacy. Behav Ther. 2022;53(3):440-457. doi:10.1016/j.beth.2021.11.003
Insomnia remains a substantial problem among military veterans. First-line treatments for the disorder are sleep hygiene modification and CBT. Access to CBT is limited, especially for veterans. Kuhn et al11 studied the effectiveness of using Insomnia Coach, a CBT for insomnia–based app, to improve insomnia symptoms.
Study design
- Fifty US veterans who were mostly male (58%) with a mean age of 44.5 and moderate insomnia symptoms were randomized to use Insomnia Coach (n = 25) or to a waitlist control group (n = 25) for 6 weeks.
- All participants completed self-report measures and sleep diaries at baseline, posttreatment, and follow-up (12 weeks). Those who used the app (n = 15) completed a qualitative interview at posttreatment.
Outcomes
- At posttreatment, 28% of participants who used Insomnia Coach achieved clinically significant improvement, vs 4% of waitlist control participants. There was also a significant treatment effect on daytime sleep-related impairment (P = .044, d = -0.6).
- Additional treatment effects emerged at follow-up for insomnia severity, sleep onset latency, global sleep quality, and depression symptoms.
- Based on self-reports and qualitative interview responses, participants’ perceptions of Insomnia Coach were favorable. Three-fourths of participants used the app through 6 weeks and engaged with active elements.
Continue to: Conclusions/limitations
Conclusions/limitations
- Insomnia Coach may provide an accessible and convenient public health intervention for patients who aren’t receiving adequate care or CBT.
- Limitations: Because this study evaluated only veterans, the findings might not be generalizable to other populations.
8. Dahne J, Lejuez CW, Diaz VA, et al. Pilot randomized trial of a self-help behavioral activation mobile app for utilization in primary care. Behav Ther. 2019;50(4):817-827. doi:10.1016/j.beth.2018.12.003
Previous mobile technologies have shown the ability to treat depression in primary care settings. Moodivate is a self-help mobile app based on the Brief Behavioral Activation Treatment for Depression, which is an evidence-based treatment. This app is designed to help the user reengage in positive, nondepressed activities by identifying, scheduling, and completing activities. Dahne et al12 investigated the feasibility and efficacy of Moodivate for depressive symptoms in primary care patients.
Study design
- Participants (N = 52) were recruited from primary care practices and randomized 2:2:1 to receive Moodivate, a CBT-based mobile app called MoodKit, or treatment as usual (no app). All participants had an initial PHQ-8 score >10.
- Participants completed assessments of depressive symptoms (PHQ-8) weekly for 8 weeks.
- App analytics data were captured to examine if the use of Moodivate was feasible. (Analytics were not available for MoodKit).
Outcomes
- Participants who used Moodivate had a mean (SD) of 46.76 (30.10) sessions throughout the trial, spent 3.50 (2.76) minutes using the app per session, and spent 120.76 (101.02) minutes using the app in total.
- Nearly 70% of Moodivate participants continued to use the app 1 month after trial enrollment and 50% at the end of the 8-week follow-up period.
- Compared to the treatment as usual group, participants who used Moodivate and those who used MoodKit experienced significant decreases in depressive symptoms over time.
Conclusions/limitations
- The results show that for primary care patients with depression, the use of Moodivate is feasible and may reduce depressive symptoms.
- Limitations: For the first 3 months of enrollment, patients who met diagnostic criteria for a current major depressive episode were excluded. This study did not assess duration of medication use (ie, whether a study participant was stabilized on medication or recently started taking a new medication) and therefore could not ascertain whether treatment gains were a result of the use of the app or of possible new medication use.
1. Torous J, Jän Myrick K, Rauseo-Ricupero N, et al. Digital mental health and COVID-19: using technology today to accelerate the curve on access and quality tomorrow. JMIR Ment Health. 2020;7(3):e18848. doi:10.2196/18848
2. Camacho E, Cohen A, Torous J. Assessment of mental health services available through smartphone apps. JAMA Netw Open. 2022;5(12):e2248784. doi:10.1001/jamanetworkopen.2022.48784
3. American Psychiatric Association. APP Advisor: An American Psychiatric Association Initiative. Accessed April 28, 2023. https://www.psychiatry.org/psychiatrists/practice/mental-health-apps
4. Lagan S, Aquino P, Emerson MR, et al. Actionable health app evaluation: translating expert frameworks into objective metrics. NPJ Digit Med. 2020;3:100. doi:10.1038/s41746-020-00312-4
5. Linardon J, Shatte A, Rosato J, et al. Efficacy of a transdiagnostic cognitive-behavioral intervention for eating disorder psychopathology delivered through a smartphone app: a randomized controlled trial. Psychol Med. 2022;52(9):1679-1690. doi:10.1017/S0033291720003426
6. Christoforou M, Sáez Fonseca JA, Tsakanikos E. Two novel cognitive behavioral therapy–based mobile apps for agoraphobia: randomized controlled trial. J Med Internet Res. 2017;19(11):e398. doi:10.2196/jmir.7747
7. Everitt N, Broadbent J, Richardson B, et al. Exploring the features of an app-based just-in-time intervention for depression. J Affect Disord. 2021;291:279-287. doi:10.1016/j.jad.2021.05.021
8. McLean C, Davis CA, Miller M, et al. The effects of an exposure-based mobile app on symptoms of posttraumatic stress disorder in veterans: pilot randomized controlled trial. JMIR Mhealth Uhealth. 2022;10(11):e38951. doi:10.2196/38951
9. Graham AK, Greene CJ, Kwasny MJ, et al. Coached mobile app platform for the treatment of depression and anxiety among primary care patients: a randomized clinical trial. JAMA Psychiatry. 2020;77(9):906-914. doi:10.1001/jamapsychiatry.2020.1011
10. Wilhelm S, Weingarden H, Greenberg JL, et al. Efficacy of app-based cognitive behavioral therapy for body dysmorphic disorder with coach support: initial randomized controlled clinical trial. Psychother Psychosom. 2022;91(4):277-285. doi:10.1159/000524628
11. Kuhn E, Miller KE, Puran D, et al. A pilot randomized controlled trial of the Insomnia Coach mobile app to assess its feasibility, acceptability, and potential efficacy. Behav Ther. 2022;53(3):440-457. doi:10.1016/j.beth.2021.11.003
12. Dahne J, Lejuez CW, Diaz VA, et al. Pilot randomized trial of a self-help behavioral activation mobile app for utilization in primary care. Behav Ther. 2019;50(4):817-827. doi:10.1016/j.beth.2018.12.003
COVID-19’s increased demand on the mental health care delivery system led to expanded utilization of technology-based solutions, including digital tools to deliver care.1 Technology-based solutions include both synchronous telehealth (eg, real-time interactive audio/video visits) and asynchronous tools such as smartphone applications (apps). Both real-time telehealth and apps continue to gain popularity. More than 10,000 mental health–related apps are available, and that number continues to rise.2 Numerous web- or mobile-based apps are available to aid in the treatment of various psychiatric conditions, including generalized anxiety disorder (GAD), major depressive disorder, insomnia, and posttraumatic stress disorder (PTSD).
Clinicians may find it challenging to choose the best psychiatry-related apps to recommend to patients. This dilemma calls for an approach to help clinicians select apps that are safe and effective.2 The American Psychiatric Association provides information to help mental health professionals navigate these issues and identify which aspects to consider when selecting an app for clinical use.3 The M-Health Index and Navigation Database also provides a set of objective evaluative criteria and offers guidance on choosing apps.4
In this article, we review 8 randomized controlled trials (RCTs) of mental health–related apps. We took several steps to ensure the RCTs we included were impactful and meaningful. First, we conducted a general search using mainstream search engines to assess which psychiatric apps were most popular for use in clinical practice. Using this list, we conducted a scholarly search engine query of RCTs using the name of the apps as a search parameter along with the following keywords: “mobile,” “web,” “applications,” and “psychiatry.” This search yielded approximately 50 results, which were narrowed down based on content and interest to a list of 8 articles (Table5-12). These articles were then graded using the limitations of each study as the primary substrate for evaluation.

1. Linardon J, Shatte A, Rosato J, et al. Efficacy of a transdiagnostic cognitive-behavioral intervention for eating disorder psychopathology delivered through a smartphone app: a randomized controlled trial. Psychol Med. 2022;52(9):1679-1690. doi:10.1017/S0033291720003426
Many patients with eating disorders are unable to receive effective treatment due to problems with accessing health care. Smartphone apps may help bridge the treatment gap for patients in this position. Linardon et al5 developed an app that uses the principles of cognitive-behavioral therapy (CBT) for treating eating disorders and conducted this study to evaluate its effectiveness.
Study design
- This RCT assigned individuals who reported episodes of binge eating to a group that used a mobile app (n = 197) or to a waiting list (n = 195). At baseline, 42% of participants exhibited diagnostic-level symptoms of bulimia nervosa and 31% had symptoms of binge-eating disorder.
- Assessments took place at baseline, Week 4, and Week 8.
- The primary outcome was global levels of eating disorder psychopathology.
- Secondary outcomes were other eating disorder symptoms, impairment, and distress.
Outcomes
- Compared to the control group, participants who used the mobile app reported greater reductions in global eating disorder psychopathology (d = -0.80).
- Significant effects were also observed for secondary outcomes except compensatory behavior frequency.
- Overall, participants reported they were satisfied with the app.
Continue to: Conclusions/limitations
Conclusions/limitations
- Findings show this app could potentially be a cost-effective and easily accessible option for patients who cannot receive standard treatment for eating disorders.
- Limitations: The overall posttest attrition rate was 35%.
2. Christoforou M, Sáez Fonseca JA, Tsakanikos E. Two novel cognitive behavioral therapy–based mobile apps for agoraphobia: randomized controlled trial. J Med Internet Res. 2017;19(11):e398. doi:10.2196/jmir.7747
CBT is generally the most accepted first-line treatment for agoraphobia. However, numerous barriers to obtaining CBT can prevent successful treatment. Limited research has evaluated the efficacy of apps for treating agoraphobia. Christoforou et al6 conducted an RCT to determine the effectiveness of a self-guided smartphone app for improving agoraphobic symptoms, compared to a mobile app used to treat anxiety.
Study design
- Participants (N = 170) who self-identified as having agoraphobia were randomly assigned to use a smartphone app designed to target agoraphobia (Agoraphobia Free) or a smartphone app designed to help with symptoms of anxiety (Stress Free) for 12 weeks. Both apps were based on established cognitive behavioral principles.
- Assessment occurred at baseline, midpoint, and end point.
- The primary outcome was symptom severity as measured by the Panic and Agoraphobia Scale (PAS).
Outcomes
- Both groups experienced statistically significant improvements in symptom severity over time. The differences in PAS score were -5.97 (95% CI, -8.49 to -3.44, P < .001) for Agoraphobia Free and -6.35 (95% CI, -8.82 to -3.87, P < .001) for Stress Free.
- There were no significant between-group differences in symptom severity.
Continue to: Conclusions/limitations
Conclusions/limitations
- This study is the first RCT to show that patients with agoraphobia could benefit from mobile-based interventions.
- Limitations: There was no waitlist control group. Limited information was collected about participant characteristics; there were no data on comorbid disorders, other psychological or physiological treatments, or other demographic characteristics such as ethnicity or computer literacy.
3. Everitt N, Broadbent J, Richardson B, et al. Exploring the features of an app-based just-in-time intervention for depression. J Affect Disord. 2021;291:279-287. doi:10.1016/j.jad.2021.05.021
The apps MoodTracker, ImproveYourMood, and ImproveYourMood+ deliver content “just in time” (in response to acute negative symptoms) to help patients with depression. In an RCT, Everitt et al7 evaluated delivering acute care for depressive mood states via a smartphone app. They sought to delineate whether symptom improvement was due to microintervention content, mood augmentation, or just-in-time prompts to use content.
Study design
- Participants (N = 235) from the general population who said they wanted to improve their mood were randomly assigned to a waitlist control group (n = 55) or 1 of 3 intervention groups: MoodTracker (monitoring-only; n = 58), ImproveYourMood (monitoring and content; n = 62), or ImproveYourMood+ (monitoring, content, and prompts; n = 60).
- The microintervention content provided by these apps consisted of 4 audio files of brief (2- to 3-minute) mindfulness and relaxation exercises. Participants used the assigned app for 3 weeks.
- Depressive symptoms, anxiety symptoms, and negative automatic thoughts were assessed at baseline, immediately following the intervention, and 1 month after the intervention using the 9-item Patient Health Questionnaire (PHQ-9), 7-item GAD scale (GAD-7), and 8-item Automatic Thoughts Questionnaire, respectively.
Outcomes
- Compared to the waitlist control group, participants in the ImproveYourMood group showed greater declines in depressive symptoms and anxiety symptoms (at follow-up only), and negative automatic thoughts (at both postintervention and follow-up).
- Those in the ImproveYourMood+ group only showed significantly greater improvements for automatic negative thoughts (at postintervention).
- MoodTracker participants did not differ from waitlist controls for any variables at any timepoints.
Continue to: Conclusions/limitations
Conclusions/limitations
- This study suggests that using microinterventions in acute settings can effectively reduce depressive symptoms both as they occur, and 1 to 2 months later.
- Limitations: The study featured a naturalistic design, where participants self-selected whether they wanted to use the program. Participants did not complete eligibility assessments or receive compensation, and the study had high dropout rates, ranging from 20% for the waitlist control group to 67% for the ImproveYourMood+ group.
4. McLean C, Davis CA, Miller M, et al. The effects of an exposure-based mobile app on symptoms of posttraumatic stress disorder in veterans: pilot randomized controlled trial. JMIR Mhealth Uhealth. 2022;10(11):e38951. doi:10.2196/38951
Veterans with PTSD face barriers when receiving trauma-focused treatments such as exposure therapy or CBT. Smartphone apps may help veterans self-treat and self-manage their PTSD symptoms. McLean et al8 studied the efficacy of Renew, a smartphone app that uses exposure therapy and social support to treat PTSD.
Study design
- In this pilot RCT, 93 veterans with clinically significant PTSD symptoms were randomly assigned to use the Renew app with and without support from a research staff member (active use group) or to a waitlist (delayed use group) for 6 weeks.
- The PTSD Checklist for DSM-5 (PCL-5) was used to measure PTSD symptoms at preintervention, postintervention, and 6-week follow-up.
- Most participants (69%) were women, and the mean age was 49.
Outcomes
- Compared to the delayed use group, participants in the active use group experienced a larger decrease in PCL-5 score (-6.14 vs -1.84). However, this difference was not statistically significant (P = .29), and the effect size was small (d = -0.39).
- There was no difference in engagement with the app between participants who received support from a research staff member and those who did not receive such support.
Continue to: Conclusions/limitations
Conclusions/limitations
- Renew may show promise as a tool to reduce PTSD symptoms in veterans.
- Educating family and friends on how to best support a patient using a mobile mental health app may help improve the efficacy of Renew and increase app engagement.
- Limitations: Because the study was conducted in veterans, the results may not be generalizable to other populations. Because most data collection occurred during the first wave of the COVID-19 pandemic in the United States, COVID-19–related stress may have impacted PTSD symptoms, app engagement, or outcomes.
5. Graham AK, Greene CJ, Kwasny MJ, et al. Coached mobile app platform for the treatment of depression and anxiety among primary care patients: a randomized clinical trial. JAMA Psychiatry. 2020;77(9):906-914. doi:10.1001/jamapsychiatry.2020.1011
Many cases of depression and anxiety are initially treated in primary care settings. However, these settings may have limited resources and inadequate training, and mobile interventions might be helpful to augment patient care. Graham et al9 studied the mobile platform IntelliCare to determine its efficacy as a tool to be used in primary care settings to treat depression and anxiety.
Study design
- This RCT randomly assigned adult primary care patients (N = 146) who screened positive for depression on the PHQ-9 (score ≥10) or anxiety on the GAD-7 (score ≥8) to the coach-supported IntelliCare platform, which consisted of 5 clinically focused apps, or to a waitlist control group. Interventions were delivered over 8 weeks.
- Overall, 122 (83.6%) patients were diagnosed with depression and 131 (89.7%) were diagnosed with anxiety.
- The primary outcomes were changes in depression (as measured by change in PHQ-9 score) and anxiety (change in GAD-7 score) during the intervention period.
Outcomes
- Participants who used the IntelliCare platform had a greater reduction in depression and anxiety symptoms compared to waitlist controls, and changes were sustained over 2-month follow-up.
- The least square means (LSM) difference in depression scores at Week 4 was 2.91 (SE = 0.83; d = 0.43) and at Week 8 was 4.37 (SE = 0.83; d = 0.64). The LSM difference in anxiety scores at Week 4 was 2.51 (SE = 0.78; d = 0.41) and at Week 8 was 3.33 (SE = 0.76; d = 0.55).
- A median number of 93 and 98 sessions among participants with depression and anxiety were recorded, respectively, indicating high use of the IntelliCare platform.
Continue to: Conclusions/limitations
Conclusions/limitations
- The IntelliCare platform was shown to be effective in reducing depression and anxiety among primary care patients. Simple apps can be bundled together and used by patients in conjunction to treat their individual needs.
- Limitations: The study had a limited follow-up period and did not record participants’ use of other apps. Slightly more than one-half (56%) of participants were taking an antidepressant.
6. Wilhelm S, Weingarden H, Greenberg JL, et al. Efficacy of app-based cognitive behavioral therapy for body dysmorphic disorder with coach support: initial randomized controlled clinical trial. Psychother Psychosom. 2022;91(4):277-285. doi:10.1159/000524628
Body dysmorphic disorder (BDD) is a severe yet undertreated disorder. Apps can improve access to treatment for patients experiencing BDD. Wilhelm et al10 studied the usability and efficacy of a coach-supported app called Perspectives that was specifically designed for treating BDD. Perspectives provide CBT in 7 modules: psychoeducation, cognitive restructuring, exposure, response prevention, mindfulness, attention retraining, and relapse prevention.
Study design
- Adults (N = 80) with primary BDD were assigned to use the Perspectives app for 12 weeks or to a waitlist control group. Participants were predominately female (84%) and White (71%), with a mean age of 27.
- Coaches promoted engagement and answered questions via in-app messaging and phone calls.
- Blinded independent evaluators used the Yale-Brown Obsessive Compulsive Scale Modified for BDD (BDD-YBOCS) to measure BDD severity at baseline, midtreatment (Week 6), and end of treatment (Week 12).
- Secondary outcomes included BDD-related insight, depression, quality of life, and functioning. Various scales were used to measure these outcomes.
Outcomes
- In intent-to-treat analyses, patients who received CBT via the Perspectives app had significantly lower BDD severity at the end of treatment compared to the waitlist control group, with a mean (SD) BDD-YBOCS score of 16.8 (7.5) vs 26.7 (6.2), with P < .001 and d = 1.44.
- Slightly more than one-half (52%) of those who used Perspectives achieved full or partial remission, compared to 8% in the waitlist control group.
Continue to: Conclusions/limitations
Conclusions/limitations
- CBT delivered via the Perspectives app and a coach proved to be effective treatment for adults with BDD.
- Adoption of the application was relatively high; 86% of Perspectives users were very or mostly satisfied.
- Limitations: Because the participants in this study were predominantly female and White, the findings might not be generalizable to other populations.
7. Kuhn E, Miller KE, Puran D, et al. A pilot randomized controlled trial of the Insomnia Coach mobile app to assess its feasibility, acceptability, and potential efficacy. Behav Ther. 2022;53(3):440-457. doi:10.1016/j.beth.2021.11.003
Insomnia remains a substantial problem among military veterans. First-line treatments for the disorder are sleep hygiene modification and CBT. Access to CBT is limited, especially for veterans. Kuhn et al11 studied the effectiveness of using Insomnia Coach, a CBT for insomnia–based app, to improve insomnia symptoms.
Study design
- Fifty US veterans who were mostly male (58%) with a mean age of 44.5 and moderate insomnia symptoms were randomized to use Insomnia Coach (n = 25) or to a waitlist control group (n = 25) for 6 weeks.
- All participants completed self-report measures and sleep diaries at baseline, posttreatment, and follow-up (12 weeks). Those who used the app (n = 15) completed a qualitative interview at posttreatment.
Outcomes
- At posttreatment, 28% of participants who used Insomnia Coach achieved clinically significant improvement, vs 4% of waitlist control participants. There was also a significant treatment effect on daytime sleep-related impairment (P = .044, d = -0.6).
- Additional treatment effects emerged at follow-up for insomnia severity, sleep onset latency, global sleep quality, and depression symptoms.
- Based on self-reports and qualitative interview responses, participants’ perceptions of Insomnia Coach were favorable. Three-fourths of participants used the app through 6 weeks and engaged with active elements.
Continue to: Conclusions/limitations
Conclusions/limitations
- Insomnia Coach may provide an accessible and convenient public health intervention for patients who aren’t receiving adequate care or CBT.
- Limitations: Because this study evaluated only veterans, the findings might not be generalizable to other populations.
8. Dahne J, Lejuez CW, Diaz VA, et al. Pilot randomized trial of a self-help behavioral activation mobile app for utilization in primary care. Behav Ther. 2019;50(4):817-827. doi:10.1016/j.beth.2018.12.003
Previous mobile technologies have shown the ability to treat depression in primary care settings. Moodivate is a self-help mobile app based on the Brief Behavioral Activation Treatment for Depression, which is an evidence-based treatment. This app is designed to help the user reengage in positive, nondepressed activities by identifying, scheduling, and completing activities. Dahne et al12 investigated the feasibility and efficacy of Moodivate for depressive symptoms in primary care patients.
Study design
- Participants (N = 52) were recruited from primary care practices and randomized 2:2:1 to receive Moodivate, a CBT-based mobile app called MoodKit, or treatment as usual (no app). All participants had an initial PHQ-8 score >10.
- Participants completed assessments of depressive symptoms (PHQ-8) weekly for 8 weeks.
- App analytics data were captured to examine if the use of Moodivate was feasible. (Analytics were not available for MoodKit).
Outcomes
- Participants who used Moodivate had a mean (SD) of 46.76 (30.10) sessions throughout the trial, spent 3.50 (2.76) minutes using the app per session, and spent 120.76 (101.02) minutes using the app in total.
- Nearly 70% of Moodivate participants continued to use the app 1 month after trial enrollment and 50% at the end of the 8-week follow-up period.
- Compared to the treatment as usual group, participants who used Moodivate and those who used MoodKit experienced significant decreases in depressive symptoms over time.
Conclusions/limitations
- The results show that for primary care patients with depression, the use of Moodivate is feasible and may reduce depressive symptoms.
- Limitations: For the first 3 months of enrollment, patients who met diagnostic criteria for a current major depressive episode were excluded. This study did not assess duration of medication use (ie, whether a study participant was stabilized on medication or recently started taking a new medication) and therefore could not ascertain whether treatment gains were a result of the use of the app or of possible new medication use.
COVID-19’s increased demand on the mental health care delivery system led to expanded utilization of technology-based solutions, including digital tools to deliver care.1 Technology-based solutions include both synchronous telehealth (eg, real-time interactive audio/video visits) and asynchronous tools such as smartphone applications (apps). Both real-time telehealth and apps continue to gain popularity. More than 10,000 mental health–related apps are available, and that number continues to rise.2 Numerous web- or mobile-based apps are available to aid in the treatment of various psychiatric conditions, including generalized anxiety disorder (GAD), major depressive disorder, insomnia, and posttraumatic stress disorder (PTSD).
Clinicians may find it challenging to choose the best psychiatry-related apps to recommend to patients. This dilemma calls for an approach to help clinicians select apps that are safe and effective.2 The American Psychiatric Association provides information to help mental health professionals navigate these issues and identify which aspects to consider when selecting an app for clinical use.3 The M-Health Index and Navigation Database also provides a set of objective evaluative criteria and offers guidance on choosing apps.4
In this article, we review 8 randomized controlled trials (RCTs) of mental health–related apps. We took several steps to ensure the RCTs we included were impactful and meaningful. First, we conducted a general search using mainstream search engines to assess which psychiatric apps were most popular for use in clinical practice. Using this list, we conducted a scholarly search engine query of RCTs using the name of the apps as a search parameter along with the following keywords: “mobile,” “web,” “applications,” and “psychiatry.” This search yielded approximately 50 results, which were narrowed down based on content and interest to a list of 8 articles (Table5-12). These articles were then graded using the limitations of each study as the primary substrate for evaluation.

1. Linardon J, Shatte A, Rosato J, et al. Efficacy of a transdiagnostic cognitive-behavioral intervention for eating disorder psychopathology delivered through a smartphone app: a randomized controlled trial. Psychol Med. 2022;52(9):1679-1690. doi:10.1017/S0033291720003426
Many patients with eating disorders are unable to receive effective treatment due to problems with accessing health care. Smartphone apps may help bridge the treatment gap for patients in this position. Linardon et al5 developed an app that uses the principles of cognitive-behavioral therapy (CBT) for treating eating disorders and conducted this study to evaluate its effectiveness.
Study design
- This RCT assigned individuals who reported episodes of binge eating to a group that used a mobile app (n = 197) or to a waiting list (n = 195). At baseline, 42% of participants exhibited diagnostic-level symptoms of bulimia nervosa and 31% had symptoms of binge-eating disorder.
- Assessments took place at baseline, Week 4, and Week 8.
- The primary outcome was global levels of eating disorder psychopathology.
- Secondary outcomes were other eating disorder symptoms, impairment, and distress.
Outcomes
- Compared to the control group, participants who used the mobile app reported greater reductions in global eating disorder psychopathology (d = -0.80).
- Significant effects were also observed for secondary outcomes except compensatory behavior frequency.
- Overall, participants reported they were satisfied with the app.
Continue to: Conclusions/limitations
Conclusions/limitations
- Findings show this app could potentially be a cost-effective and easily accessible option for patients who cannot receive standard treatment for eating disorders.
- Limitations: The overall posttest attrition rate was 35%.
2. Christoforou M, Sáez Fonseca JA, Tsakanikos E. Two novel cognitive behavioral therapy–based mobile apps for agoraphobia: randomized controlled trial. J Med Internet Res. 2017;19(11):e398. doi:10.2196/jmir.7747
CBT is generally the most accepted first-line treatment for agoraphobia. However, numerous barriers to obtaining CBT can prevent successful treatment. Limited research has evaluated the efficacy of apps for treating agoraphobia. Christoforou et al6 conducted an RCT to determine the effectiveness of a self-guided smartphone app for improving agoraphobic symptoms, compared to a mobile app used to treat anxiety.
Study design
- Participants (N = 170) who self-identified as having agoraphobia were randomly assigned to use a smartphone app designed to target agoraphobia (Agoraphobia Free) or a smartphone app designed to help with symptoms of anxiety (Stress Free) for 12 weeks. Both apps were based on established cognitive behavioral principles.
- Assessment occurred at baseline, midpoint, and end point.
- The primary outcome was symptom severity as measured by the Panic and Agoraphobia Scale (PAS).
Outcomes
- Both groups experienced statistically significant improvements in symptom severity over time. The differences in PAS score were -5.97 (95% CI, -8.49 to -3.44, P < .001) for Agoraphobia Free and -6.35 (95% CI, -8.82 to -3.87, P < .001) for Stress Free.
- There were no significant between-group differences in symptom severity.
Continue to: Conclusions/limitations
Conclusions/limitations
- This study is the first RCT to show that patients with agoraphobia could benefit from mobile-based interventions.
- Limitations: There was no waitlist control group. Limited information was collected about participant characteristics; there were no data on comorbid disorders, other psychological or physiological treatments, or other demographic characteristics such as ethnicity or computer literacy.
3. Everitt N, Broadbent J, Richardson B, et al. Exploring the features of an app-based just-in-time intervention for depression. J Affect Disord. 2021;291:279-287. doi:10.1016/j.jad.2021.05.021
The apps MoodTracker, ImproveYourMood, and ImproveYourMood+ deliver content “just in time” (in response to acute negative symptoms) to help patients with depression. In an RCT, Everitt et al7 evaluated delivering acute care for depressive mood states via a smartphone app. They sought to delineate whether symptom improvement was due to microintervention content, mood augmentation, or just-in-time prompts to use content.
Study design
- Participants (N = 235) from the general population who said they wanted to improve their mood were randomly assigned to a waitlist control group (n = 55) or 1 of 3 intervention groups: MoodTracker (monitoring-only; n = 58), ImproveYourMood (monitoring and content; n = 62), or ImproveYourMood+ (monitoring, content, and prompts; n = 60).
- The microintervention content provided by these apps consisted of 4 audio files of brief (2- to 3-minute) mindfulness and relaxation exercises. Participants used the assigned app for 3 weeks.
- Depressive symptoms, anxiety symptoms, and negative automatic thoughts were assessed at baseline, immediately following the intervention, and 1 month after the intervention using the 9-item Patient Health Questionnaire (PHQ-9), 7-item GAD scale (GAD-7), and 8-item Automatic Thoughts Questionnaire, respectively.
Outcomes
- Compared to the waitlist control group, participants in the ImproveYourMood group showed greater declines in depressive symptoms and anxiety symptoms (at follow-up only), and negative automatic thoughts (at both postintervention and follow-up).
- Those in the ImproveYourMood+ group only showed significantly greater improvements for automatic negative thoughts (at postintervention).
- MoodTracker participants did not differ from waitlist controls for any variables at any timepoints.
Continue to: Conclusions/limitations
Conclusions/limitations
- This study suggests that using microinterventions in acute settings can effectively reduce depressive symptoms both as they occur, and 1 to 2 months later.
- Limitations: The study featured a naturalistic design, where participants self-selected whether they wanted to use the program. Participants did not complete eligibility assessments or receive compensation, and the study had high dropout rates, ranging from 20% for the waitlist control group to 67% for the ImproveYourMood+ group.
4. McLean C, Davis CA, Miller M, et al. The effects of an exposure-based mobile app on symptoms of posttraumatic stress disorder in veterans: pilot randomized controlled trial. JMIR Mhealth Uhealth. 2022;10(11):e38951. doi:10.2196/38951
Veterans with PTSD face barriers when receiving trauma-focused treatments such as exposure therapy or CBT. Smartphone apps may help veterans self-treat and self-manage their PTSD symptoms. McLean et al8 studied the efficacy of Renew, a smartphone app that uses exposure therapy and social support to treat PTSD.
Study design
- In this pilot RCT, 93 veterans with clinically significant PTSD symptoms were randomly assigned to use the Renew app with and without support from a research staff member (active use group) or to a waitlist (delayed use group) for 6 weeks.
- The PTSD Checklist for DSM-5 (PCL-5) was used to measure PTSD symptoms at preintervention, postintervention, and 6-week follow-up.
- Most participants (69%) were women, and the mean age was 49.
Outcomes
- Compared to the delayed use group, participants in the active use group experienced a larger decrease in PCL-5 score (-6.14 vs -1.84). However, this difference was not statistically significant (P = .29), and the effect size was small (d = -0.39).
- There was no difference in engagement with the app between participants who received support from a research staff member and those who did not receive such support.
Continue to: Conclusions/limitations
Conclusions/limitations
- Renew may show promise as a tool to reduce PTSD symptoms in veterans.
- Educating family and friends on how to best support a patient using a mobile mental health app may help improve the efficacy of Renew and increase app engagement.
- Limitations: Because the study was conducted in veterans, the results may not be generalizable to other populations. Because most data collection occurred during the first wave of the COVID-19 pandemic in the United States, COVID-19–related stress may have impacted PTSD symptoms, app engagement, or outcomes.
5. Graham AK, Greene CJ, Kwasny MJ, et al. Coached mobile app platform for the treatment of depression and anxiety among primary care patients: a randomized clinical trial. JAMA Psychiatry. 2020;77(9):906-914. doi:10.1001/jamapsychiatry.2020.1011
Many cases of depression and anxiety are initially treated in primary care settings. However, these settings may have limited resources and inadequate training, and mobile interventions might be helpful to augment patient care. Graham et al9 studied the mobile platform IntelliCare to determine its efficacy as a tool to be used in primary care settings to treat depression and anxiety.
Study design
- This RCT randomly assigned adult primary care patients (N = 146) who screened positive for depression on the PHQ-9 (score ≥10) or anxiety on the GAD-7 (score ≥8) to the coach-supported IntelliCare platform, which consisted of 5 clinically focused apps, or to a waitlist control group. Interventions were delivered over 8 weeks.
- Overall, 122 (83.6%) patients were diagnosed with depression and 131 (89.7%) were diagnosed with anxiety.
- The primary outcomes were changes in depression (as measured by change in PHQ-9 score) and anxiety (change in GAD-7 score) during the intervention period.
Outcomes
- Participants who used the IntelliCare platform had a greater reduction in depression and anxiety symptoms compared to waitlist controls, and changes were sustained over 2-month follow-up.
- The least square means (LSM) difference in depression scores at Week 4 was 2.91 (SE = 0.83; d = 0.43) and at Week 8 was 4.37 (SE = 0.83; d = 0.64). The LSM difference in anxiety scores at Week 4 was 2.51 (SE = 0.78; d = 0.41) and at Week 8 was 3.33 (SE = 0.76; d = 0.55).
- A median number of 93 and 98 sessions among participants with depression and anxiety were recorded, respectively, indicating high use of the IntelliCare platform.
Continue to: Conclusions/limitations
Conclusions/limitations
- The IntelliCare platform was shown to be effective in reducing depression and anxiety among primary care patients. Simple apps can be bundled together and used by patients in conjunction to treat their individual needs.
- Limitations: The study had a limited follow-up period and did not record participants’ use of other apps. Slightly more than one-half (56%) of participants were taking an antidepressant.
6. Wilhelm S, Weingarden H, Greenberg JL, et al. Efficacy of app-based cognitive behavioral therapy for body dysmorphic disorder with coach support: initial randomized controlled clinical trial. Psychother Psychosom. 2022;91(4):277-285. doi:10.1159/000524628
Body dysmorphic disorder (BDD) is a severe yet undertreated disorder. Apps can improve access to treatment for patients experiencing BDD. Wilhelm et al10 studied the usability and efficacy of a coach-supported app called Perspectives that was specifically designed for treating BDD. Perspectives provide CBT in 7 modules: psychoeducation, cognitive restructuring, exposure, response prevention, mindfulness, attention retraining, and relapse prevention.
Study design
- Adults (N = 80) with primary BDD were assigned to use the Perspectives app for 12 weeks or to a waitlist control group. Participants were predominately female (84%) and White (71%), with a mean age of 27.
- Coaches promoted engagement and answered questions via in-app messaging and phone calls.
- Blinded independent evaluators used the Yale-Brown Obsessive Compulsive Scale Modified for BDD (BDD-YBOCS) to measure BDD severity at baseline, midtreatment (Week 6), and end of treatment (Week 12).
- Secondary outcomes included BDD-related insight, depression, quality of life, and functioning. Various scales were used to measure these outcomes.
Outcomes
- In intent-to-treat analyses, patients who received CBT via the Perspectives app had significantly lower BDD severity at the end of treatment compared to the waitlist control group, with a mean (SD) BDD-YBOCS score of 16.8 (7.5) vs 26.7 (6.2), with P < .001 and d = 1.44.
- Slightly more than one-half (52%) of those who used Perspectives achieved full or partial remission, compared to 8% in the waitlist control group.
Continue to: Conclusions/limitations
Conclusions/limitations
- CBT delivered via the Perspectives app and a coach proved to be effective treatment for adults with BDD.
- Adoption of the application was relatively high; 86% of Perspectives users were very or mostly satisfied.
- Limitations: Because the participants in this study were predominantly female and White, the findings might not be generalizable to other populations.
7. Kuhn E, Miller KE, Puran D, et al. A pilot randomized controlled trial of the Insomnia Coach mobile app to assess its feasibility, acceptability, and potential efficacy. Behav Ther. 2022;53(3):440-457. doi:10.1016/j.beth.2021.11.003
Insomnia remains a substantial problem among military veterans. First-line treatments for the disorder are sleep hygiene modification and CBT. Access to CBT is limited, especially for veterans. Kuhn et al11 studied the effectiveness of using Insomnia Coach, a CBT for insomnia–based app, to improve insomnia symptoms.
Study design
- Fifty US veterans who were mostly male (58%) with a mean age of 44.5 and moderate insomnia symptoms were randomized to use Insomnia Coach (n = 25) or to a waitlist control group (n = 25) for 6 weeks.
- All participants completed self-report measures and sleep diaries at baseline, posttreatment, and follow-up (12 weeks). Those who used the app (n = 15) completed a qualitative interview at posttreatment.
Outcomes
- At posttreatment, 28% of participants who used Insomnia Coach achieved clinically significant improvement, vs 4% of waitlist control participants. There was also a significant treatment effect on daytime sleep-related impairment (P = .044, d = -0.6).
- Additional treatment effects emerged at follow-up for insomnia severity, sleep onset latency, global sleep quality, and depression symptoms.
- Based on self-reports and qualitative interview responses, participants’ perceptions of Insomnia Coach were favorable. Three-fourths of participants used the app through 6 weeks and engaged with active elements.
Continue to: Conclusions/limitations
Conclusions/limitations
- Insomnia Coach may provide an accessible and convenient public health intervention for patients who aren’t receiving adequate care or CBT.
- Limitations: Because this study evaluated only veterans, the findings might not be generalizable to other populations.
8. Dahne J, Lejuez CW, Diaz VA, et al. Pilot randomized trial of a self-help behavioral activation mobile app for utilization in primary care. Behav Ther. 2019;50(4):817-827. doi:10.1016/j.beth.2018.12.003
Previous mobile technologies have shown the ability to treat depression in primary care settings. Moodivate is a self-help mobile app based on the Brief Behavioral Activation Treatment for Depression, which is an evidence-based treatment. This app is designed to help the user reengage in positive, nondepressed activities by identifying, scheduling, and completing activities. Dahne et al12 investigated the feasibility and efficacy of Moodivate for depressive symptoms in primary care patients.
Study design
- Participants (N = 52) were recruited from primary care practices and randomized 2:2:1 to receive Moodivate, a CBT-based mobile app called MoodKit, or treatment as usual (no app). All participants had an initial PHQ-8 score >10.
- Participants completed assessments of depressive symptoms (PHQ-8) weekly for 8 weeks.
- App analytics data were captured to examine if the use of Moodivate was feasible. (Analytics were not available for MoodKit).
Outcomes
- Participants who used Moodivate had a mean (SD) of 46.76 (30.10) sessions throughout the trial, spent 3.50 (2.76) minutes using the app per session, and spent 120.76 (101.02) minutes using the app in total.
- Nearly 70% of Moodivate participants continued to use the app 1 month after trial enrollment and 50% at the end of the 8-week follow-up period.
- Compared to the treatment as usual group, participants who used Moodivate and those who used MoodKit experienced significant decreases in depressive symptoms over time.
Conclusions/limitations
- The results show that for primary care patients with depression, the use of Moodivate is feasible and may reduce depressive symptoms.
- Limitations: For the first 3 months of enrollment, patients who met diagnostic criteria for a current major depressive episode were excluded. This study did not assess duration of medication use (ie, whether a study participant was stabilized on medication or recently started taking a new medication) and therefore could not ascertain whether treatment gains were a result of the use of the app or of possible new medication use.
1. Torous J, Jän Myrick K, Rauseo-Ricupero N, et al. Digital mental health and COVID-19: using technology today to accelerate the curve on access and quality tomorrow. JMIR Ment Health. 2020;7(3):e18848. doi:10.2196/18848
2. Camacho E, Cohen A, Torous J. Assessment of mental health services available through smartphone apps. JAMA Netw Open. 2022;5(12):e2248784. doi:10.1001/jamanetworkopen.2022.48784
3. American Psychiatric Association. APP Advisor: An American Psychiatric Association Initiative. Accessed April 28, 2023. https://www.psychiatry.org/psychiatrists/practice/mental-health-apps
4. Lagan S, Aquino P, Emerson MR, et al. Actionable health app evaluation: translating expert frameworks into objective metrics. NPJ Digit Med. 2020;3:100. doi:10.1038/s41746-020-00312-4
5. Linardon J, Shatte A, Rosato J, et al. Efficacy of a transdiagnostic cognitive-behavioral intervention for eating disorder psychopathology delivered through a smartphone app: a randomized controlled trial. Psychol Med. 2022;52(9):1679-1690. doi:10.1017/S0033291720003426
6. Christoforou M, Sáez Fonseca JA, Tsakanikos E. Two novel cognitive behavioral therapy–based mobile apps for agoraphobia: randomized controlled trial. J Med Internet Res. 2017;19(11):e398. doi:10.2196/jmir.7747
7. Everitt N, Broadbent J, Richardson B, et al. Exploring the features of an app-based just-in-time intervention for depression. J Affect Disord. 2021;291:279-287. doi:10.1016/j.jad.2021.05.021
8. McLean C, Davis CA, Miller M, et al. The effects of an exposure-based mobile app on symptoms of posttraumatic stress disorder in veterans: pilot randomized controlled trial. JMIR Mhealth Uhealth. 2022;10(11):e38951. doi:10.2196/38951
9. Graham AK, Greene CJ, Kwasny MJ, et al. Coached mobile app platform for the treatment of depression and anxiety among primary care patients: a randomized clinical trial. JAMA Psychiatry. 2020;77(9):906-914. doi:10.1001/jamapsychiatry.2020.1011
10. Wilhelm S, Weingarden H, Greenberg JL, et al. Efficacy of app-based cognitive behavioral therapy for body dysmorphic disorder with coach support: initial randomized controlled clinical trial. Psychother Psychosom. 2022;91(4):277-285. doi:10.1159/000524628
11. Kuhn E, Miller KE, Puran D, et al. A pilot randomized controlled trial of the Insomnia Coach mobile app to assess its feasibility, acceptability, and potential efficacy. Behav Ther. 2022;53(3):440-457. doi:10.1016/j.beth.2021.11.003
12. Dahne J, Lejuez CW, Diaz VA, et al. Pilot randomized trial of a self-help behavioral activation mobile app for utilization in primary care. Behav Ther. 2019;50(4):817-827. doi:10.1016/j.beth.2018.12.003
1. Torous J, Jän Myrick K, Rauseo-Ricupero N, et al. Digital mental health and COVID-19: using technology today to accelerate the curve on access and quality tomorrow. JMIR Ment Health. 2020;7(3):e18848. doi:10.2196/18848
2. Camacho E, Cohen A, Torous J. Assessment of mental health services available through smartphone apps. JAMA Netw Open. 2022;5(12):e2248784. doi:10.1001/jamanetworkopen.2022.48784
3. American Psychiatric Association. APP Advisor: An American Psychiatric Association Initiative. Accessed April 28, 2023. https://www.psychiatry.org/psychiatrists/practice/mental-health-apps
4. Lagan S, Aquino P, Emerson MR, et al. Actionable health app evaluation: translating expert frameworks into objective metrics. NPJ Digit Med. 2020;3:100. doi:10.1038/s41746-020-00312-4
5. Linardon J, Shatte A, Rosato J, et al. Efficacy of a transdiagnostic cognitive-behavioral intervention for eating disorder psychopathology delivered through a smartphone app: a randomized controlled trial. Psychol Med. 2022;52(9):1679-1690. doi:10.1017/S0033291720003426
6. Christoforou M, Sáez Fonseca JA, Tsakanikos E. Two novel cognitive behavioral therapy–based mobile apps for agoraphobia: randomized controlled trial. J Med Internet Res. 2017;19(11):e398. doi:10.2196/jmir.7747
7. Everitt N, Broadbent J, Richardson B, et al. Exploring the features of an app-based just-in-time intervention for depression. J Affect Disord. 2021;291:279-287. doi:10.1016/j.jad.2021.05.021
8. McLean C, Davis CA, Miller M, et al. The effects of an exposure-based mobile app on symptoms of posttraumatic stress disorder in veterans: pilot randomized controlled trial. JMIR Mhealth Uhealth. 2022;10(11):e38951. doi:10.2196/38951
9. Graham AK, Greene CJ, Kwasny MJ, et al. Coached mobile app platform for the treatment of depression and anxiety among primary care patients: a randomized clinical trial. JAMA Psychiatry. 2020;77(9):906-914. doi:10.1001/jamapsychiatry.2020.1011
10. Wilhelm S, Weingarden H, Greenberg JL, et al. Efficacy of app-based cognitive behavioral therapy for body dysmorphic disorder with coach support: initial randomized controlled clinical trial. Psychother Psychosom. 2022;91(4):277-285. doi:10.1159/000524628
11. Kuhn E, Miller KE, Puran D, et al. A pilot randomized controlled trial of the Insomnia Coach mobile app to assess its feasibility, acceptability, and potential efficacy. Behav Ther. 2022;53(3):440-457. doi:10.1016/j.beth.2021.11.003
12. Dahne J, Lejuez CW, Diaz VA, et al. Pilot randomized trial of a self-help behavioral activation mobile app for utilization in primary care. Behav Ther. 2019;50(4):817-827. doi:10.1016/j.beth.2018.12.003
Lamotrigine interactions with oral contraceptives
Ms. A, age 20, presents to the clinic after experiencing difficulty sleeping, depressed mood, fatigue, and difficulty concentrating. Her psychiatric history includes bipolar II disorder (BD II), predominantly with depressive episodes. Ms. A’s current medications include a combination of lamotrigine 200 mg/d and bupropion extended-release 450 mg/d, and her symptoms were well maintained until 2 weeks ago. When her psychiatrist performs a medication reconciliation at her medication management appointment, Ms. A indicates she started taking an oral contraceptive, ethinyl estradiol and norgestimate, approximately 1 month ago for management of endometriosis symptoms. She is not currently taking any other medications or supplements.
Lamotrigine is indicated for epilepsy and as maintenance treatment for BD I. It is also used off-label to treat other mood disorders. After oral administration, lamotrigine is rapidly and fully absorbed with a high bioavailability (98%).The principal metabolic pathway is via glucuronic acid conjugation, leading to the major inactive metabolite 2-N-glucuronide. Minor metabolites include 5-N-glucuronide and a 2-N-glucuronide metabolite.1
Combined oral contraceptives contain an estrogen component, typically ethinyl estradiol, and a progestin component, which varies based on the specific formulation. The metabolism of ethinyl estradiol occurs through cytochrome P450 (CYP)3A4, CYP2C9, sulfation, and glucuronidation. For progestin—the second component of combined oral contraceptives and the lone component of progestin-only oral contraceptives—metabolism occurs via CYP3A4 and conjugation reactions.2 This article focuses on lamotrigine interactions specifically with oral contraceptives, but it is important to note that other formulations of combined hormonal contraceptives, such as the combined contraceptive patch (Ortho Evra) and vaginal ring (NuvaRing), would be expected to interact in the same way as oral formulations.3
Bidirectional interaction
While many antiseizure medications are known to interact with and potentially decrease the efficacy of oral contraceptives (Table 13-6), the interactions between lamotrigine and oral contraceptives is uniquely bidirectional. Combined oral contraceptives are thought to interact with lamotrigine primarily via the estrogen component, which causes increased metabolism of lamotrigine through induction of glucuronidation. This drug interaction decreases the plasma concentrations of lamotrigine in the body by up to 2-fold, resulting in an increased risk of seizures or inadequate mood stabilization.1 This effect on metabolism is very rapid, resulting in decreases in lamotrigine concentrations within 1 week.4,7 A recent study suggested that certain progestins may also contribute to decreased plasma levels of lamotrigine, but the mechanism for this is unknown (Table 23-7).8
Clinicians should consider increasing the lamotrigine dose (potentially as much as 2-fold) in a patient who initiates treatment with a combined hormonal contraceptive. Dose increases should not be >50 to 100 mg/d every week.1 Collect lamotrigine blood levels before starting a hormonal contraceptive and during dose titration. While there is not a well-established therapeutic range for lamotrigine in BD, expert consensus recommends a range of 1 to 6 mcg/mL.8

The lamotrigine dose should be decreased if combined hormonal contraceptives are discontinued. Dose decreases should not exceed 25% of the total daily dose per week.1 Desogestrel, a progestin-only medication, may increase exposure to lamotrigine, but this has not been observed in research with other progestins.5,9 When starting a progestin-only pill, monitor patients for signs of lamotrigine toxicity (ataxia, diplopia, dizziness) and consider monitoring their blood levels.
An important consideration to note with combined oral contraceptives is the hormone-free interval, also known as the pill-free week. Due to the rapid effect of estrogens, the lamotrigine concentrations have been shown to rise, even double, during this hormone-free interval, so patients should be closely monitored for adverse effects.3 Some recommend use of an extended cycle regimen (with a limited hormone-free interval), or continuous cycle regimen (with no hormone-free interval) to avoid fluctuations in lamotrigine levels.3,5 Additionally, data suggest that in patients taking lamotrigine and valproate, which inhibits glucuronidation, oral contraceptives do not cause reductions in lamotrigine concentrations.2,5 In these instances, dose increases of lamotrigine are not needed.
Continue to: The metabolism of ethinyl estradiol...
The metabolism of ethinyl estradiol and progestin are susceptible to CYP3A4 induction and increased glucuronidation. Serum concentrations may be reduced by ≥50% when used concomitantly with CYP enzyme–inducing medications, which could possibly result in subtherapeutic levels and unplanned pregnancy.3 CYP3A4 induction occurs for up to 4 weeks after discontinuation of an enzyme-inducing agent, pointing to the need for alternative or backup contraception during this time.3 Lamotrigine is not a CYP enzyme–inducing medication; it is unlikely to affect the efficacy of oral contraceptives in the same manner as other antiseizure medications. However, a study of lamotrigine and the combined hormonal contraceptive ethinyl estradiol and levonorgestrel demonstrated reduced exposure to levonorgestrel, resulting in breakthrough bleeding.5
In a study on the coadministration of lamotrigine and combined oral contraceptives, Sidhu et al4 observed a small mean reduction (20%) in progestin concentrations when lamotrigine was used at a dose of 300 mg/d. Although there is no research suggesting decreased effectiveness in preventing pregnancy when lamotrigine is used with combined oral contraceptives, progestin-only oral contraceptives, or progestin implants, additional or alternative contraceptive methods may be considered based on this pharmacokinetic data, particularly in patients who require lamotrigine doses ≥300 mg/d.5
CASE CONTINUED
Given when Ms. A started the oral contraceptive, the treatment team determines it is likely that an interaction with lamotrigine is causing her resurgence of depressive symptoms. Her care team decides to titrate the lamotrigine gradually to 300 mg/d, then 400 mg/d if needed, while carefully monitoring for signs of a serious rash. This dosage increase may help Ms. A achieve symptom remission. Monitoring plasma levels may be considered, although it is unknown what plasma level was effective for Ms. A before she started the oral contraceptive. Ms. A would need to be counseled regarding the effect of higher doses of lamotrigine on the effectiveness of the oral contraceptive.
Although it does not appear Ms. A is using the oral contraceptive specifically to prevent pregnancy, the team informs her about the possibility of unintended pregnancy with this medication combination. If Ms. A was also using the medication for this indication, alternative contraceptive options would include medroxyprogesterone acetate, levonorgestrel implants, or an intrauterine device (levonorgestrel or copper, though copper would not be effective for endometriosis symptom management). Ms. A should consult with her gynecologist regarding the most appropriate option for her endometriosis. If the decision is made to discontinue her oral contraceptive in the future, the lamotrigine dose should be decreased to her previously effective dose of 200 mg/d.
Related Resources
- Makino KK, Hatters Friedman S, Amin J. Emergency contraception for psychiatric patients. Current Psychiatry. 2022;21(11):34-39,44-45. doi:10.12788/cp.0300
- MGH Center for Women’s Mental Health. You asked: is there an interaction between lamotrigine and oral contraceptives? September 29, 2015. https://womensmentalhealth.org/posts/you-asked-is-there-an-interaction-between-lamotrigine-andoral-contraceptives/
Drug Brand Names
Bupropion extended-release • Wellbutrin XL
Carbamazepine • Equetro, Tegretol
Desogestrel • Cerazette
Divalproex sodium • Depakote
Ethinyl estradiol and etonogestrel • NuvaRing
Ethinyl estradiol and norelgestromin • Ortho Evra
Ethinyl estradiol and norgestimate • Ortho Tri-Cyclen, TriNessa, others
Etonogestrel • Implanon, Nexplanon
Gabapentin • Neurontin
Lamotrigine • Lamictal
Levonorgestrel emergency contraceptive pill • AfterPill, Plan B
Levonorgestrel intrauterine device • Mirena, Skyla
Medroxyprogesterone acetate • Depo-Provera
Oxcarbazepine • Trileptal
Topiramate • Topamax
Valproic acid • Depakene
1. Lamictal [package insert]. Research Triangle Park, NC: GlaxoSmithKline; 2020.
2. Lee CR. Drug interactions and hormonal contraception. Trends in Urology Gynaecology & Sexual Health. 2009;14(3):23-26.
3. Williams D. Antiepileptic drugs and contraception. US Pharm. 2014;39(1):39-42.
4. Sidhu J, Job S, Singh S, et al. The pharmacokinetic and pharmacodynamic consequences of the co-administration of lamotrigine and a combined oral contraceptive in healthy female subjects. Br J Clin Pharmacol. 2006;61(2):191-199. doi:10.1111/j.1365-2125.2005.02539.x
5. Faculty of Sexual & Reproductive Healthcare. Clinical guidance: drug interactions with hormonal contraception. Published May 9, 2022. Accessed September 28, 2022. https://www.fsrh.org/documents/ceu-clinical-guidance-drug-interactions-with-hormonal/
6. Johnston CA, Crawford PM. Anti-epileptic drugs and hormonal treatments. Curr Treat Options Neurol. 2014;16(5):288. doi:10.1007/s11940-014-0288-3
7. Christensen J, Petrenaite V, Atterman J, et al. Oral contraceptives induce lamotrigine metabolism: evidence from a double-blind, placebo-controlled trial. Epilepsia. 2007;48(3):484-489. doi:10.1111/j.1528-1167.2007.00997.x
8. Hiemke C, Bergemann N, Clement HW, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51(1-02):9-62. doi:10.1055/s-0043-116492
9. Rauchenzauner M, Deichmann S, Pittschieler, et al. Bidirectional interaction between oral contraception and lamotrigine in women with epilepsy – role of progestins. Seizure. 2020;74:89-92. doi:10.1016/j.seizure.2019.11.011
Ms. A, age 20, presents to the clinic after experiencing difficulty sleeping, depressed mood, fatigue, and difficulty concentrating. Her psychiatric history includes bipolar II disorder (BD II), predominantly with depressive episodes. Ms. A’s current medications include a combination of lamotrigine 200 mg/d and bupropion extended-release 450 mg/d, and her symptoms were well maintained until 2 weeks ago. When her psychiatrist performs a medication reconciliation at her medication management appointment, Ms. A indicates she started taking an oral contraceptive, ethinyl estradiol and norgestimate, approximately 1 month ago for management of endometriosis symptoms. She is not currently taking any other medications or supplements.
Lamotrigine is indicated for epilepsy and as maintenance treatment for BD I. It is also used off-label to treat other mood disorders. After oral administration, lamotrigine is rapidly and fully absorbed with a high bioavailability (98%).The principal metabolic pathway is via glucuronic acid conjugation, leading to the major inactive metabolite 2-N-glucuronide. Minor metabolites include 5-N-glucuronide and a 2-N-glucuronide metabolite.1
Combined oral contraceptives contain an estrogen component, typically ethinyl estradiol, and a progestin component, which varies based on the specific formulation. The metabolism of ethinyl estradiol occurs through cytochrome P450 (CYP)3A4, CYP2C9, sulfation, and glucuronidation. For progestin—the second component of combined oral contraceptives and the lone component of progestin-only oral contraceptives—metabolism occurs via CYP3A4 and conjugation reactions.2 This article focuses on lamotrigine interactions specifically with oral contraceptives, but it is important to note that other formulations of combined hormonal contraceptives, such as the combined contraceptive patch (Ortho Evra) and vaginal ring (NuvaRing), would be expected to interact in the same way as oral formulations.3
Bidirectional interaction
While many antiseizure medications are known to interact with and potentially decrease the efficacy of oral contraceptives (Table 13-6), the interactions between lamotrigine and oral contraceptives is uniquely bidirectional. Combined oral contraceptives are thought to interact with lamotrigine primarily via the estrogen component, which causes increased metabolism of lamotrigine through induction of glucuronidation. This drug interaction decreases the plasma concentrations of lamotrigine in the body by up to 2-fold, resulting in an increased risk of seizures or inadequate mood stabilization.1 This effect on metabolism is very rapid, resulting in decreases in lamotrigine concentrations within 1 week.4,7 A recent study suggested that certain progestins may also contribute to decreased plasma levels of lamotrigine, but the mechanism for this is unknown (Table 23-7).8

Clinicians should consider increasing the lamotrigine dose (potentially as much as 2-fold) in a patient who initiates treatment with a combined hormonal contraceptive. Dose increases should not be >50 to 100 mg/d every week.1 Collect lamotrigine blood levels before starting a hormonal contraceptive and during dose titration. While there is not a well-established therapeutic range for lamotrigine in BD, expert consensus recommends a range of 1 to 6 mcg/mL.8

The lamotrigine dose should be decreased if combined hormonal contraceptives are discontinued. Dose decreases should not exceed 25% of the total daily dose per week.1 Desogestrel, a progestin-only medication, may increase exposure to lamotrigine, but this has not been observed in research with other progestins.5,9 When starting a progestin-only pill, monitor patients for signs of lamotrigine toxicity (ataxia, diplopia, dizziness) and consider monitoring their blood levels.
An important consideration to note with combined oral contraceptives is the hormone-free interval, also known as the pill-free week. Due to the rapid effect of estrogens, the lamotrigine concentrations have been shown to rise, even double, during this hormone-free interval, so patients should be closely monitored for adverse effects.3 Some recommend use of an extended cycle regimen (with a limited hormone-free interval), or continuous cycle regimen (with no hormone-free interval) to avoid fluctuations in lamotrigine levels.3,5 Additionally, data suggest that in patients taking lamotrigine and valproate, which inhibits glucuronidation, oral contraceptives do not cause reductions in lamotrigine concentrations.2,5 In these instances, dose increases of lamotrigine are not needed.
Continue to: The metabolism of ethinyl estradiol...
The metabolism of ethinyl estradiol and progestin are susceptible to CYP3A4 induction and increased glucuronidation. Serum concentrations may be reduced by ≥50% when used concomitantly with CYP enzyme–inducing medications, which could possibly result in subtherapeutic levels and unplanned pregnancy.3 CYP3A4 induction occurs for up to 4 weeks after discontinuation of an enzyme-inducing agent, pointing to the need for alternative or backup contraception during this time.3 Lamotrigine is not a CYP enzyme–inducing medication; it is unlikely to affect the efficacy of oral contraceptives in the same manner as other antiseizure medications. However, a study of lamotrigine and the combined hormonal contraceptive ethinyl estradiol and levonorgestrel demonstrated reduced exposure to levonorgestrel, resulting in breakthrough bleeding.5
In a study on the coadministration of lamotrigine and combined oral contraceptives, Sidhu et al4 observed a small mean reduction (20%) in progestin concentrations when lamotrigine was used at a dose of 300 mg/d. Although there is no research suggesting decreased effectiveness in preventing pregnancy when lamotrigine is used with combined oral contraceptives, progestin-only oral contraceptives, or progestin implants, additional or alternative contraceptive methods may be considered based on this pharmacokinetic data, particularly in patients who require lamotrigine doses ≥300 mg/d.5
CASE CONTINUED
Given when Ms. A started the oral contraceptive, the treatment team determines it is likely that an interaction with lamotrigine is causing her resurgence of depressive symptoms. Her care team decides to titrate the lamotrigine gradually to 300 mg/d, then 400 mg/d if needed, while carefully monitoring for signs of a serious rash. This dosage increase may help Ms. A achieve symptom remission. Monitoring plasma levels may be considered, although it is unknown what plasma level was effective for Ms. A before she started the oral contraceptive. Ms. A would need to be counseled regarding the effect of higher doses of lamotrigine on the effectiveness of the oral contraceptive.
Although it does not appear Ms. A is using the oral contraceptive specifically to prevent pregnancy, the team informs her about the possibility of unintended pregnancy with this medication combination. If Ms. A was also using the medication for this indication, alternative contraceptive options would include medroxyprogesterone acetate, levonorgestrel implants, or an intrauterine device (levonorgestrel or copper, though copper would not be effective for endometriosis symptom management). Ms. A should consult with her gynecologist regarding the most appropriate option for her endometriosis. If the decision is made to discontinue her oral contraceptive in the future, the lamotrigine dose should be decreased to her previously effective dose of 200 mg/d.
Related Resources
- Makino KK, Hatters Friedman S, Amin J. Emergency contraception for psychiatric patients. Current Psychiatry. 2022;21(11):34-39,44-45. doi:10.12788/cp.0300
- MGH Center for Women’s Mental Health. You asked: is there an interaction between lamotrigine and oral contraceptives? September 29, 2015. https://womensmentalhealth.org/posts/you-asked-is-there-an-interaction-between-lamotrigine-andoral-contraceptives/
Drug Brand Names
Bupropion extended-release • Wellbutrin XL
Carbamazepine • Equetro, Tegretol
Desogestrel • Cerazette
Divalproex sodium • Depakote
Ethinyl estradiol and etonogestrel • NuvaRing
Ethinyl estradiol and norelgestromin • Ortho Evra
Ethinyl estradiol and norgestimate • Ortho Tri-Cyclen, TriNessa, others
Etonogestrel • Implanon, Nexplanon
Gabapentin • Neurontin
Lamotrigine • Lamictal
Levonorgestrel emergency contraceptive pill • AfterPill, Plan B
Levonorgestrel intrauterine device • Mirena, Skyla
Medroxyprogesterone acetate • Depo-Provera
Oxcarbazepine • Trileptal
Topiramate • Topamax
Valproic acid • Depakene
Ms. A, age 20, presents to the clinic after experiencing difficulty sleeping, depressed mood, fatigue, and difficulty concentrating. Her psychiatric history includes bipolar II disorder (BD II), predominantly with depressive episodes. Ms. A’s current medications include a combination of lamotrigine 200 mg/d and bupropion extended-release 450 mg/d, and her symptoms were well maintained until 2 weeks ago. When her psychiatrist performs a medication reconciliation at her medication management appointment, Ms. A indicates she started taking an oral contraceptive, ethinyl estradiol and norgestimate, approximately 1 month ago for management of endometriosis symptoms. She is not currently taking any other medications or supplements.
Lamotrigine is indicated for epilepsy and as maintenance treatment for BD I. It is also used off-label to treat other mood disorders. After oral administration, lamotrigine is rapidly and fully absorbed with a high bioavailability (98%).The principal metabolic pathway is via glucuronic acid conjugation, leading to the major inactive metabolite 2-N-glucuronide. Minor metabolites include 5-N-glucuronide and a 2-N-glucuronide metabolite.1
Combined oral contraceptives contain an estrogen component, typically ethinyl estradiol, and a progestin component, which varies based on the specific formulation. The metabolism of ethinyl estradiol occurs through cytochrome P450 (CYP)3A4, CYP2C9, sulfation, and glucuronidation. For progestin—the second component of combined oral contraceptives and the lone component of progestin-only oral contraceptives—metabolism occurs via CYP3A4 and conjugation reactions.2 This article focuses on lamotrigine interactions specifically with oral contraceptives, but it is important to note that other formulations of combined hormonal contraceptives, such as the combined contraceptive patch (Ortho Evra) and vaginal ring (NuvaRing), would be expected to interact in the same way as oral formulations.3
Bidirectional interaction
While many antiseizure medications are known to interact with and potentially decrease the efficacy of oral contraceptives (Table 13-6), the interactions between lamotrigine and oral contraceptives is uniquely bidirectional. Combined oral contraceptives are thought to interact with lamotrigine primarily via the estrogen component, which causes increased metabolism of lamotrigine through induction of glucuronidation. This drug interaction decreases the plasma concentrations of lamotrigine in the body by up to 2-fold, resulting in an increased risk of seizures or inadequate mood stabilization.1 This effect on metabolism is very rapid, resulting in decreases in lamotrigine concentrations within 1 week.4,7 A recent study suggested that certain progestins may also contribute to decreased plasma levels of lamotrigine, but the mechanism for this is unknown (Table 23-7).8

Clinicians should consider increasing the lamotrigine dose (potentially as much as 2-fold) in a patient who initiates treatment with a combined hormonal contraceptive. Dose increases should not be >50 to 100 mg/d every week.1 Collect lamotrigine blood levels before starting a hormonal contraceptive and during dose titration. While there is not a well-established therapeutic range for lamotrigine in BD, expert consensus recommends a range of 1 to 6 mcg/mL.8

The lamotrigine dose should be decreased if combined hormonal contraceptives are discontinued. Dose decreases should not exceed 25% of the total daily dose per week.1 Desogestrel, a progestin-only medication, may increase exposure to lamotrigine, but this has not been observed in research with other progestins.5,9 When starting a progestin-only pill, monitor patients for signs of lamotrigine toxicity (ataxia, diplopia, dizziness) and consider monitoring their blood levels.
An important consideration to note with combined oral contraceptives is the hormone-free interval, also known as the pill-free week. Due to the rapid effect of estrogens, the lamotrigine concentrations have been shown to rise, even double, during this hormone-free interval, so patients should be closely monitored for adverse effects.3 Some recommend use of an extended cycle regimen (with a limited hormone-free interval), or continuous cycle regimen (with no hormone-free interval) to avoid fluctuations in lamotrigine levels.3,5 Additionally, data suggest that in patients taking lamotrigine and valproate, which inhibits glucuronidation, oral contraceptives do not cause reductions in lamotrigine concentrations.2,5 In these instances, dose increases of lamotrigine are not needed.
Continue to: The metabolism of ethinyl estradiol...
The metabolism of ethinyl estradiol and progestin are susceptible to CYP3A4 induction and increased glucuronidation. Serum concentrations may be reduced by ≥50% when used concomitantly with CYP enzyme–inducing medications, which could possibly result in subtherapeutic levels and unplanned pregnancy.3 CYP3A4 induction occurs for up to 4 weeks after discontinuation of an enzyme-inducing agent, pointing to the need for alternative or backup contraception during this time.3 Lamotrigine is not a CYP enzyme–inducing medication; it is unlikely to affect the efficacy of oral contraceptives in the same manner as other antiseizure medications. However, a study of lamotrigine and the combined hormonal contraceptive ethinyl estradiol and levonorgestrel demonstrated reduced exposure to levonorgestrel, resulting in breakthrough bleeding.5
In a study on the coadministration of lamotrigine and combined oral contraceptives, Sidhu et al4 observed a small mean reduction (20%) in progestin concentrations when lamotrigine was used at a dose of 300 mg/d. Although there is no research suggesting decreased effectiveness in preventing pregnancy when lamotrigine is used with combined oral contraceptives, progestin-only oral contraceptives, or progestin implants, additional or alternative contraceptive methods may be considered based on this pharmacokinetic data, particularly in patients who require lamotrigine doses ≥300 mg/d.5
CASE CONTINUED
Given when Ms. A started the oral contraceptive, the treatment team determines it is likely that an interaction with lamotrigine is causing her resurgence of depressive symptoms. Her care team decides to titrate the lamotrigine gradually to 300 mg/d, then 400 mg/d if needed, while carefully monitoring for signs of a serious rash. This dosage increase may help Ms. A achieve symptom remission. Monitoring plasma levels may be considered, although it is unknown what plasma level was effective for Ms. A before she started the oral contraceptive. Ms. A would need to be counseled regarding the effect of higher doses of lamotrigine on the effectiveness of the oral contraceptive.
Although it does not appear Ms. A is using the oral contraceptive specifically to prevent pregnancy, the team informs her about the possibility of unintended pregnancy with this medication combination. If Ms. A was also using the medication for this indication, alternative contraceptive options would include medroxyprogesterone acetate, levonorgestrel implants, or an intrauterine device (levonorgestrel or copper, though copper would not be effective for endometriosis symptom management). Ms. A should consult with her gynecologist regarding the most appropriate option for her endometriosis. If the decision is made to discontinue her oral contraceptive in the future, the lamotrigine dose should be decreased to her previously effective dose of 200 mg/d.
Related Resources
- Makino KK, Hatters Friedman S, Amin J. Emergency contraception for psychiatric patients. Current Psychiatry. 2022;21(11):34-39,44-45. doi:10.12788/cp.0300
- MGH Center for Women’s Mental Health. You asked: is there an interaction between lamotrigine and oral contraceptives? September 29, 2015. https://womensmentalhealth.org/posts/you-asked-is-there-an-interaction-between-lamotrigine-andoral-contraceptives/
Drug Brand Names
Bupropion extended-release • Wellbutrin XL
Carbamazepine • Equetro, Tegretol
Desogestrel • Cerazette
Divalproex sodium • Depakote
Ethinyl estradiol and etonogestrel • NuvaRing
Ethinyl estradiol and norelgestromin • Ortho Evra
Ethinyl estradiol and norgestimate • Ortho Tri-Cyclen, TriNessa, others
Etonogestrel • Implanon, Nexplanon
Gabapentin • Neurontin
Lamotrigine • Lamictal
Levonorgestrel emergency contraceptive pill • AfterPill, Plan B
Levonorgestrel intrauterine device • Mirena, Skyla
Medroxyprogesterone acetate • Depo-Provera
Oxcarbazepine • Trileptal
Topiramate • Topamax
Valproic acid • Depakene
1. Lamictal [package insert]. Research Triangle Park, NC: GlaxoSmithKline; 2020.
2. Lee CR. Drug interactions and hormonal contraception. Trends in Urology Gynaecology & Sexual Health. 2009;14(3):23-26.
3. Williams D. Antiepileptic drugs and contraception. US Pharm. 2014;39(1):39-42.
4. Sidhu J, Job S, Singh S, et al. The pharmacokinetic and pharmacodynamic consequences of the co-administration of lamotrigine and a combined oral contraceptive in healthy female subjects. Br J Clin Pharmacol. 2006;61(2):191-199. doi:10.1111/j.1365-2125.2005.02539.x
5. Faculty of Sexual & Reproductive Healthcare. Clinical guidance: drug interactions with hormonal contraception. Published May 9, 2022. Accessed September 28, 2022. https://www.fsrh.org/documents/ceu-clinical-guidance-drug-interactions-with-hormonal/
6. Johnston CA, Crawford PM. Anti-epileptic drugs and hormonal treatments. Curr Treat Options Neurol. 2014;16(5):288. doi:10.1007/s11940-014-0288-3
7. Christensen J, Petrenaite V, Atterman J, et al. Oral contraceptives induce lamotrigine metabolism: evidence from a double-blind, placebo-controlled trial. Epilepsia. 2007;48(3):484-489. doi:10.1111/j.1528-1167.2007.00997.x
8. Hiemke C, Bergemann N, Clement HW, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51(1-02):9-62. doi:10.1055/s-0043-116492
9. Rauchenzauner M, Deichmann S, Pittschieler, et al. Bidirectional interaction between oral contraception and lamotrigine in women with epilepsy – role of progestins. Seizure. 2020;74:89-92. doi:10.1016/j.seizure.2019.11.011
1. Lamictal [package insert]. Research Triangle Park, NC: GlaxoSmithKline; 2020.
2. Lee CR. Drug interactions and hormonal contraception. Trends in Urology Gynaecology & Sexual Health. 2009;14(3):23-26.
3. Williams D. Antiepileptic drugs and contraception. US Pharm. 2014;39(1):39-42.
4. Sidhu J, Job S, Singh S, et al. The pharmacokinetic and pharmacodynamic consequences of the co-administration of lamotrigine and a combined oral contraceptive in healthy female subjects. Br J Clin Pharmacol. 2006;61(2):191-199. doi:10.1111/j.1365-2125.2005.02539.x
5. Faculty of Sexual & Reproductive Healthcare. Clinical guidance: drug interactions with hormonal contraception. Published May 9, 2022. Accessed September 28, 2022. https://www.fsrh.org/documents/ceu-clinical-guidance-drug-interactions-with-hormonal/
6. Johnston CA, Crawford PM. Anti-epileptic drugs and hormonal treatments. Curr Treat Options Neurol. 2014;16(5):288. doi:10.1007/s11940-014-0288-3
7. Christensen J, Petrenaite V, Atterman J, et al. Oral contraceptives induce lamotrigine metabolism: evidence from a double-blind, placebo-controlled trial. Epilepsia. 2007;48(3):484-489. doi:10.1111/j.1528-1167.2007.00997.x
8. Hiemke C, Bergemann N, Clement HW, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51(1-02):9-62. doi:10.1055/s-0043-116492
9. Rauchenzauner M, Deichmann S, Pittschieler, et al. Bidirectional interaction between oral contraception and lamotrigine in women with epilepsy – role of progestins. Seizure. 2020;74:89-92. doi:10.1016/j.seizure.2019.11.011









