User login
Opioids, other causes linked to shorter lifespans, rising midlife mortality
Two reports in the BMJ regarding new research offer grim tidings about mortality in the United States: One study has suggested the opioid epidemic is driving down life expectancy, and another has found that midlife death rates from multiple causes are on the rise across ethnic groups.
“Death rates are increasing across the U.S. population for dozens of conditions,” according to Steven H. Woolf, MD, MPH, a professor at Virginia Commonwealth University, Richmond, and his associates, who together wrote the second study. “Of particular urgency is recognizing that the unfavorable mortality pattern that began for some groups in the 1990s is now unfolding among Hispanics and non-Hispanic blacks, a development made more consequential by their high baseline mortality rates.”
Dr. Woolf and his coinvestigators tracked midlife mortality (ages 25-64 years) from 1999-2016 across the U.S. population.
They found that all-cause mortality jumped among whites, American Indians, and Alaska Natives over that time. A trend toward decreasing all-cause mortality ended in 2009-2011 among African Americans, Hispanic American, Asian Americans, and Americans of Pacific Islander descent.
“Drug overdoses were the leading cause of increased mortality in midlife in each population, but mortality also increased for alcohol related conditions, suicides, and organ diseases involving multiple body systems,” according to the researchers.
For the life expectancy study, which was published in the BMJ, researchers led by Jessica Y. Ho, PhD, of the University of Southern California, Los Angeles, tracked all-cause and cause-specific mortality in 18 high-income nations. They focused on the years 2014-2016.
Most of the nations saw declines in life expectancy from 2014-2015, mainly caused by older people dying from physical diseases and mental disorders. From 2015-2016, most of these nations saw their life expectancy levels rebound and experienced “robust gains.”
In the United States, however, life expectancy at birth for women fell from 81.47 years in 2014 to 81.35 years in 2015, and rose to 81.40 years in 2016.
For men, life expectancy fell continuously from 76.67 years in 2014 to 76.50 years in 2015 to 76.40 years in 2016.
In 2016, these life expectancies were the lowest of 17 nations examined in the study. (Statistics from the 18th nation in the study, Canada, were not included for 2016).
The United States was an outlier in other ways. For one, deaths among people younger than 65 years were the major contributor to the life expectancy decline.
And causes of death were also different than other countries: “For American women, drug overdose and external causes, and respiratory and cardiovascular diseases, contributed roughly equally to the decline in life expectancy, but for American men, nearly all of the decline was attributable to drug overdose and external causes.”
No funding is reported for the death rates study. The study authors report no relevant disclosures.
The life expectancy study was supported by the Robert Wood Johnson Foundation. Authors variously report support from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Institute on Aging, and the National Institute of Child Health and Human Development.
SOURCE: Ho JY et al. BMJ. 2018;362:k2562; Woolf SH et al. BMJ 2018;362:k3096.
Life expectancy in some high-income nations has declined, at least temporarily, in recent years for apparent causes such as flu epidemics and “deaths of despair” because of drug abuse and suicide. While these newer trends may be short lived, they’re worrisome because life expectancy reflects human progress. Meanwhile, some nations and subpopulations continue to lag behind high-income nations despite the benefits of the modern era. Moving forward, we must back up policies with better scientific evidence. Improvements are needed in areas such as timely national mortality data, comparable statistics, and reliable numbers.
Domantas Jasilionis, PhD, is with the Max Planck Institute for Demographic Research, Rostock, Germany
Life expectancy in some high-income nations has declined, at least temporarily, in recent years for apparent causes such as flu epidemics and “deaths of despair” because of drug abuse and suicide. While these newer trends may be short lived, they’re worrisome because life expectancy reflects human progress. Meanwhile, some nations and subpopulations continue to lag behind high-income nations despite the benefits of the modern era. Moving forward, we must back up policies with better scientific evidence. Improvements are needed in areas such as timely national mortality data, comparable statistics, and reliable numbers.
Domantas Jasilionis, PhD, is with the Max Planck Institute for Demographic Research, Rostock, Germany
Life expectancy in some high-income nations has declined, at least temporarily, in recent years for apparent causes such as flu epidemics and “deaths of despair” because of drug abuse and suicide. While these newer trends may be short lived, they’re worrisome because life expectancy reflects human progress. Meanwhile, some nations and subpopulations continue to lag behind high-income nations despite the benefits of the modern era. Moving forward, we must back up policies with better scientific evidence. Improvements are needed in areas such as timely national mortality data, comparable statistics, and reliable numbers.
Domantas Jasilionis, PhD, is with the Max Planck Institute for Demographic Research, Rostock, Germany
Two reports in the BMJ regarding new research offer grim tidings about mortality in the United States: One study has suggested the opioid epidemic is driving down life expectancy, and another has found that midlife death rates from multiple causes are on the rise across ethnic groups.
“Death rates are increasing across the U.S. population for dozens of conditions,” according to Steven H. Woolf, MD, MPH, a professor at Virginia Commonwealth University, Richmond, and his associates, who together wrote the second study. “Of particular urgency is recognizing that the unfavorable mortality pattern that began for some groups in the 1990s is now unfolding among Hispanics and non-Hispanic blacks, a development made more consequential by their high baseline mortality rates.”
Dr. Woolf and his coinvestigators tracked midlife mortality (ages 25-64 years) from 1999-2016 across the U.S. population.
They found that all-cause mortality jumped among whites, American Indians, and Alaska Natives over that time. A trend toward decreasing all-cause mortality ended in 2009-2011 among African Americans, Hispanic American, Asian Americans, and Americans of Pacific Islander descent.
“Drug overdoses were the leading cause of increased mortality in midlife in each population, but mortality also increased for alcohol related conditions, suicides, and organ diseases involving multiple body systems,” according to the researchers.
For the life expectancy study, which was published in the BMJ, researchers led by Jessica Y. Ho, PhD, of the University of Southern California, Los Angeles, tracked all-cause and cause-specific mortality in 18 high-income nations. They focused on the years 2014-2016.
Most of the nations saw declines in life expectancy from 2014-2015, mainly caused by older people dying from physical diseases and mental disorders. From 2015-2016, most of these nations saw their life expectancy levels rebound and experienced “robust gains.”
In the United States, however, life expectancy at birth for women fell from 81.47 years in 2014 to 81.35 years in 2015, and rose to 81.40 years in 2016.
For men, life expectancy fell continuously from 76.67 years in 2014 to 76.50 years in 2015 to 76.40 years in 2016.
In 2016, these life expectancies were the lowest of 17 nations examined in the study. (Statistics from the 18th nation in the study, Canada, were not included for 2016).
The United States was an outlier in other ways. For one, deaths among people younger than 65 years were the major contributor to the life expectancy decline.
And causes of death were also different than other countries: “For American women, drug overdose and external causes, and respiratory and cardiovascular diseases, contributed roughly equally to the decline in life expectancy, but for American men, nearly all of the decline was attributable to drug overdose and external causes.”
No funding is reported for the death rates study. The study authors report no relevant disclosures.
The life expectancy study was supported by the Robert Wood Johnson Foundation. Authors variously report support from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Institute on Aging, and the National Institute of Child Health and Human Development.
SOURCE: Ho JY et al. BMJ. 2018;362:k2562; Woolf SH et al. BMJ 2018;362:k3096.
Two reports in the BMJ regarding new research offer grim tidings about mortality in the United States: One study has suggested the opioid epidemic is driving down life expectancy, and another has found that midlife death rates from multiple causes are on the rise across ethnic groups.
“Death rates are increasing across the U.S. population for dozens of conditions,” according to Steven H. Woolf, MD, MPH, a professor at Virginia Commonwealth University, Richmond, and his associates, who together wrote the second study. “Of particular urgency is recognizing that the unfavorable mortality pattern that began for some groups in the 1990s is now unfolding among Hispanics and non-Hispanic blacks, a development made more consequential by their high baseline mortality rates.”
Dr. Woolf and his coinvestigators tracked midlife mortality (ages 25-64 years) from 1999-2016 across the U.S. population.
They found that all-cause mortality jumped among whites, American Indians, and Alaska Natives over that time. A trend toward decreasing all-cause mortality ended in 2009-2011 among African Americans, Hispanic American, Asian Americans, and Americans of Pacific Islander descent.
“Drug overdoses were the leading cause of increased mortality in midlife in each population, but mortality also increased for alcohol related conditions, suicides, and organ diseases involving multiple body systems,” according to the researchers.
For the life expectancy study, which was published in the BMJ, researchers led by Jessica Y. Ho, PhD, of the University of Southern California, Los Angeles, tracked all-cause and cause-specific mortality in 18 high-income nations. They focused on the years 2014-2016.
Most of the nations saw declines in life expectancy from 2014-2015, mainly caused by older people dying from physical diseases and mental disorders. From 2015-2016, most of these nations saw their life expectancy levels rebound and experienced “robust gains.”
In the United States, however, life expectancy at birth for women fell from 81.47 years in 2014 to 81.35 years in 2015, and rose to 81.40 years in 2016.
For men, life expectancy fell continuously from 76.67 years in 2014 to 76.50 years in 2015 to 76.40 years in 2016.
In 2016, these life expectancies were the lowest of 17 nations examined in the study. (Statistics from the 18th nation in the study, Canada, were not included for 2016).
The United States was an outlier in other ways. For one, deaths among people younger than 65 years were the major contributor to the life expectancy decline.
And causes of death were also different than other countries: “For American women, drug overdose and external causes, and respiratory and cardiovascular diseases, contributed roughly equally to the decline in life expectancy, but for American men, nearly all of the decline was attributable to drug overdose and external causes.”
No funding is reported for the death rates study. The study authors report no relevant disclosures.
The life expectancy study was supported by the Robert Wood Johnson Foundation. Authors variously report support from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Institute on Aging, and the National Institute of Child Health and Human Development.
SOURCE: Ho JY et al. BMJ. 2018;362:k2562; Woolf SH et al. BMJ 2018;362:k3096.
FROM THE BMJ
Sexual minorities seeking abortion report high levels of male violence
Pregnant lesbian and bisexual women who seek abortions are more likely than are their heterosexual counterparts to be the victims of violence by the men who impregnated them, a new study finds.
Rachel K. Jones, PhD, of the Guttmacher Institute, New York, and her associates also found that these sexual minority women, plus a group of individuals who described their sexual orientation as “something else,” were much more likely to report exposure to sexual and physical violence.
“No patient should be presumed to be heterosexual for any reason, including a pregnancy history. All pregnancies – like all patients – should be treated as unique and operating within the dynamic and interconnected circumstances of peoples’ lives, which may encompass differences in sexual orientation and exposure to violence,” the researchers wrote. Their report is in Obstetrics & Gynecology.
Previous research has suggested that nonheterosexual women are more likely than are straight women to become pregnant unintentionally. There also are signs suggesting that they have more abortions, too, although the findings are iffy, the study authors wrote.
For this study, Dr. Jones and her associates examined questionnaire answers of 8,380 women who responded to the Guttmacher Institute’s 2014 Abortion Patient Survey. All were undergoing abortions at 87 U.S. nonhospital facilities that performed 30 or more abortions each per year.
Of the sample, about 9% declined to describe their sexual orientation. Of the rest, 94% described themselves as heterosexual; of those, 41% were white, 28% were black, and 22% were Hispanic. Most were in their 20s, 47% were never married, and 48% had incomes below the federal poverty level.
Women also described themselves as bisexual (4%), “something else” (1%), and lesbian (0.4%). All these groups were more likely than were heterosexuals to be below the federal poverty level; more than half of the lesbian and bisexual respondents said they had previously given birth.
Fifteen percent of lesbians said their current pregnancy was caused by forced sex, compared with 1% of heterosexuals and 3% of bisexuals. (P less than .001).
Bisexuals (9%) and lesbians (33%) were more likely than were heterosexuals (4%) to say the men who impregnated them had physically abused them. The same was true for sexual abuse, which was reported by 7% of bisexuals, 35% of lesbians, and 2% of heterosexuals. After the researchers controlled for various factors including age and race, lesbians remained much more likely to report physical abuse, sexual abuse, and forced sex at the hands of the men who impregnated them (odds ratios = 15, 25, and 10, respectively, P less than .001).
“Exposure to physical and sexual violence was substantially higher among each of the sexual minority groups compared with their heterosexual counterparts, sometimes by a factor of 15 or more,” the study authors wrote. “We found that lesbian respondents had the highest levels of exposure to violence, perhaps because this population was more likely to have had sex with a man only in the context of forced sex.”
The researchers noted that their study has various limitations, such as low numbers of sexual minority women and the 4-year gap since the data were collected.
Still, Dr. Jones and her associates wrote, the study has strengths. “Health care providers, including those working in abortion settings, need to be aware that a proportion of their patient population identifies as something other than heterosexual,” they wrote.
The study was funded by the Susan Thompson Buffett Foundation with support from the National Institutes of Health via a grant to the Guttmacher Center for Population Research Innovation and Dissemination. The study authors reported no relevant financial disclosures.
SOURCE: Jones R et al. Obstet Gynecol. 2018 Sep;132(3):605-11.
Pregnant lesbian and bisexual women who seek abortions are more likely than are their heterosexual counterparts to be the victims of violence by the men who impregnated them, a new study finds.
Rachel K. Jones, PhD, of the Guttmacher Institute, New York, and her associates also found that these sexual minority women, plus a group of individuals who described their sexual orientation as “something else,” were much more likely to report exposure to sexual and physical violence.
“No patient should be presumed to be heterosexual for any reason, including a pregnancy history. All pregnancies – like all patients – should be treated as unique and operating within the dynamic and interconnected circumstances of peoples’ lives, which may encompass differences in sexual orientation and exposure to violence,” the researchers wrote. Their report is in Obstetrics & Gynecology.
Previous research has suggested that nonheterosexual women are more likely than are straight women to become pregnant unintentionally. There also are signs suggesting that they have more abortions, too, although the findings are iffy, the study authors wrote.
For this study, Dr. Jones and her associates examined questionnaire answers of 8,380 women who responded to the Guttmacher Institute’s 2014 Abortion Patient Survey. All were undergoing abortions at 87 U.S. nonhospital facilities that performed 30 or more abortions each per year.
Of the sample, about 9% declined to describe their sexual orientation. Of the rest, 94% described themselves as heterosexual; of those, 41% were white, 28% were black, and 22% were Hispanic. Most were in their 20s, 47% were never married, and 48% had incomes below the federal poverty level.
Women also described themselves as bisexual (4%), “something else” (1%), and lesbian (0.4%). All these groups were more likely than were heterosexuals to be below the federal poverty level; more than half of the lesbian and bisexual respondents said they had previously given birth.
Fifteen percent of lesbians said their current pregnancy was caused by forced sex, compared with 1% of heterosexuals and 3% of bisexuals. (P less than .001).
Bisexuals (9%) and lesbians (33%) were more likely than were heterosexuals (4%) to say the men who impregnated them had physically abused them. The same was true for sexual abuse, which was reported by 7% of bisexuals, 35% of lesbians, and 2% of heterosexuals. After the researchers controlled for various factors including age and race, lesbians remained much more likely to report physical abuse, sexual abuse, and forced sex at the hands of the men who impregnated them (odds ratios = 15, 25, and 10, respectively, P less than .001).
“Exposure to physical and sexual violence was substantially higher among each of the sexual minority groups compared with their heterosexual counterparts, sometimes by a factor of 15 or more,” the study authors wrote. “We found that lesbian respondents had the highest levels of exposure to violence, perhaps because this population was more likely to have had sex with a man only in the context of forced sex.”
The researchers noted that their study has various limitations, such as low numbers of sexual minority women and the 4-year gap since the data were collected.
Still, Dr. Jones and her associates wrote, the study has strengths. “Health care providers, including those working in abortion settings, need to be aware that a proportion of their patient population identifies as something other than heterosexual,” they wrote.
The study was funded by the Susan Thompson Buffett Foundation with support from the National Institutes of Health via a grant to the Guttmacher Center for Population Research Innovation and Dissemination. The study authors reported no relevant financial disclosures.
SOURCE: Jones R et al. Obstet Gynecol. 2018 Sep;132(3):605-11.
Pregnant lesbian and bisexual women who seek abortions are more likely than are their heterosexual counterparts to be the victims of violence by the men who impregnated them, a new study finds.
Rachel K. Jones, PhD, of the Guttmacher Institute, New York, and her associates also found that these sexual minority women, plus a group of individuals who described their sexual orientation as “something else,” were much more likely to report exposure to sexual and physical violence.
“No patient should be presumed to be heterosexual for any reason, including a pregnancy history. All pregnancies – like all patients – should be treated as unique and operating within the dynamic and interconnected circumstances of peoples’ lives, which may encompass differences in sexual orientation and exposure to violence,” the researchers wrote. Their report is in Obstetrics & Gynecology.
Previous research has suggested that nonheterosexual women are more likely than are straight women to become pregnant unintentionally. There also are signs suggesting that they have more abortions, too, although the findings are iffy, the study authors wrote.
For this study, Dr. Jones and her associates examined questionnaire answers of 8,380 women who responded to the Guttmacher Institute’s 2014 Abortion Patient Survey. All were undergoing abortions at 87 U.S. nonhospital facilities that performed 30 or more abortions each per year.
Of the sample, about 9% declined to describe their sexual orientation. Of the rest, 94% described themselves as heterosexual; of those, 41% were white, 28% were black, and 22% were Hispanic. Most were in their 20s, 47% were never married, and 48% had incomes below the federal poverty level.
Women also described themselves as bisexual (4%), “something else” (1%), and lesbian (0.4%). All these groups were more likely than were heterosexuals to be below the federal poverty level; more than half of the lesbian and bisexual respondents said they had previously given birth.
Fifteen percent of lesbians said their current pregnancy was caused by forced sex, compared with 1% of heterosexuals and 3% of bisexuals. (P less than .001).
Bisexuals (9%) and lesbians (33%) were more likely than were heterosexuals (4%) to say the men who impregnated them had physically abused them. The same was true for sexual abuse, which was reported by 7% of bisexuals, 35% of lesbians, and 2% of heterosexuals. After the researchers controlled for various factors including age and race, lesbians remained much more likely to report physical abuse, sexual abuse, and forced sex at the hands of the men who impregnated them (odds ratios = 15, 25, and 10, respectively, P less than .001).
“Exposure to physical and sexual violence was substantially higher among each of the sexual minority groups compared with their heterosexual counterparts, sometimes by a factor of 15 or more,” the study authors wrote. “We found that lesbian respondents had the highest levels of exposure to violence, perhaps because this population was more likely to have had sex with a man only in the context of forced sex.”
The researchers noted that their study has various limitations, such as low numbers of sexual minority women and the 4-year gap since the data were collected.
Still, Dr. Jones and her associates wrote, the study has strengths. “Health care providers, including those working in abortion settings, need to be aware that a proportion of their patient population identifies as something other than heterosexual,” they wrote.
The study was funded by the Susan Thompson Buffett Foundation with support from the National Institutes of Health via a grant to the Guttmacher Center for Population Research Innovation and Dissemination. The study authors reported no relevant financial disclosures.
SOURCE: Jones R et al. Obstet Gynecol. 2018 Sep;132(3):605-11.
FROM OBSTETRICS & GYNECOLOGY
Key clinical point:
Major finding: Fifteen percent of lesbians said their current pregnancy was caused by forced sex, compared with 1% of heterosexuals (P less than .001), and 3% of bisexuals. Lesbians (33%) were more likely than were heterosexuals (4%) to say the man who impregnated them had physically and/or sexually abused them.
Study details: A 2014 survey of 8,380 women seeking abortions at 87 U.S. nonhospital facilities.
Disclosures: The study was funded by the Susan Thompson Buffett Foundation with support from the National Institutes of Health via a grant to the Guttmacher Center for Population Research Innovation and Dissemination. The study authors reported no relevant financial disclosures.
Source: Jones R et al. Obstet Gynecol. 2018 Sep;132(3):605-11.
Continuation, complication rates similar for implants, IUDs
A new study of Medicaid-covered women who sought long-acting reversible contraception via subdermal etonogestrel implants or IUDs found that levels of continuation at 1 year were high, and complication rates were low.
There was little difference between the groups on both fronts.
“This study demonstrates high rates of continued use of subdermal and intrauterine contraceptives by Medicaid clients, particularly among adolescents, and therefore, interventions that increase adolescent access to these contraceptives are likely to be cost effective from the payer perspective,” the study authors wrote in Contraception.
The study, led by Max J. Romano, MD, MPH, of MedStar Franklin Square Medical Center in Baltimore, retrospectively examined the medical claims of 3,305 women treated via MedStar Family Choice, a Medicaid managed-choice insurer.
The women, who lived in Maryland and the District of Columbia, were aged 15-44 years when they underwent contraceptive treatment during 2012-2015. Of the women, 1,335 had subdermal etonogestrel implants inserted, and 1,970 had IUDs inserted. Researchers followed the women for a mean 344 days.
The implant users were younger than the IUD group (mean age, 25 years vs. 28 years; P = less than .001), and women under age 20 years were more than more than twice as likely to get implants than IUDs (71% vs. 29%).
Women older than 20 years were more likely to get IUDs than implants, with those older than 25 preferring them by a ratio of 70% to 30%.
After researchers controlled for such factors as age group and year of insertion, they found that implant recipients were slightly more likely to still have the contraception tool in place at 1 year: 81% (implants) vs. IUDs (77%; P = .01). It’s not clear why women had their implants or IUDs removed.
Claims for complications were similar between the groups (8% for implants, 7% for IUDs), and researchers didn’t find a statistically significant difference after they controlled for various factors. Dysfunctional uterine bleeding was the most common complication, followed by excessive or frequent menstruation.
Few women reported pregnancy among the implant (0.82%) and IUD (0.86%) groups, and there was no statistically significant difference between the groups.
The researchers reported that their study has various limitations: The insurance claims information may provide misleading information about minor complications, and it doesn’t include details about abortions. The claims also may not report IUD expulsions and self-removals.
The authors also noted that they didn’t control for factors like socioeconomic status, psychological conditions, and multimorbidity.
MedStar Family Choice provided study funding. Dr. Romano and Loral Patchen, PhD, disclosed receiving free CME trainings and insertion certification regarding the long-acting contraceptive devices discussed in the study from Merck, Bayer, and Teva. Dr. Patchen disclosed serving on the Diclegis speakers bureau.
A new study of Medicaid-covered women who sought long-acting reversible contraception via subdermal etonogestrel implants or IUDs found that levels of continuation at 1 year were high, and complication rates were low.
There was little difference between the groups on both fronts.
“This study demonstrates high rates of continued use of subdermal and intrauterine contraceptives by Medicaid clients, particularly among adolescents, and therefore, interventions that increase adolescent access to these contraceptives are likely to be cost effective from the payer perspective,” the study authors wrote in Contraception.
The study, led by Max J. Romano, MD, MPH, of MedStar Franklin Square Medical Center in Baltimore, retrospectively examined the medical claims of 3,305 women treated via MedStar Family Choice, a Medicaid managed-choice insurer.
The women, who lived in Maryland and the District of Columbia, were aged 15-44 years when they underwent contraceptive treatment during 2012-2015. Of the women, 1,335 had subdermal etonogestrel implants inserted, and 1,970 had IUDs inserted. Researchers followed the women for a mean 344 days.
The implant users were younger than the IUD group (mean age, 25 years vs. 28 years; P = less than .001), and women under age 20 years were more than more than twice as likely to get implants than IUDs (71% vs. 29%).
Women older than 20 years were more likely to get IUDs than implants, with those older than 25 preferring them by a ratio of 70% to 30%.
After researchers controlled for such factors as age group and year of insertion, they found that implant recipients were slightly more likely to still have the contraception tool in place at 1 year: 81% (implants) vs. IUDs (77%; P = .01). It’s not clear why women had their implants or IUDs removed.
Claims for complications were similar between the groups (8% for implants, 7% for IUDs), and researchers didn’t find a statistically significant difference after they controlled for various factors. Dysfunctional uterine bleeding was the most common complication, followed by excessive or frequent menstruation.
Few women reported pregnancy among the implant (0.82%) and IUD (0.86%) groups, and there was no statistically significant difference between the groups.
The researchers reported that their study has various limitations: The insurance claims information may provide misleading information about minor complications, and it doesn’t include details about abortions. The claims also may not report IUD expulsions and self-removals.
The authors also noted that they didn’t control for factors like socioeconomic status, psychological conditions, and multimorbidity.
MedStar Family Choice provided study funding. Dr. Romano and Loral Patchen, PhD, disclosed receiving free CME trainings and insertion certification regarding the long-acting contraceptive devices discussed in the study from Merck, Bayer, and Teva. Dr. Patchen disclosed serving on the Diclegis speakers bureau.
A new study of Medicaid-covered women who sought long-acting reversible contraception via subdermal etonogestrel implants or IUDs found that levels of continuation at 1 year were high, and complication rates were low.
There was little difference between the groups on both fronts.
“This study demonstrates high rates of continued use of subdermal and intrauterine contraceptives by Medicaid clients, particularly among adolescents, and therefore, interventions that increase adolescent access to these contraceptives are likely to be cost effective from the payer perspective,” the study authors wrote in Contraception.
The study, led by Max J. Romano, MD, MPH, of MedStar Franklin Square Medical Center in Baltimore, retrospectively examined the medical claims of 3,305 women treated via MedStar Family Choice, a Medicaid managed-choice insurer.
The women, who lived in Maryland and the District of Columbia, were aged 15-44 years when they underwent contraceptive treatment during 2012-2015. Of the women, 1,335 had subdermal etonogestrel implants inserted, and 1,970 had IUDs inserted. Researchers followed the women for a mean 344 days.
The implant users were younger than the IUD group (mean age, 25 years vs. 28 years; P = less than .001), and women under age 20 years were more than more than twice as likely to get implants than IUDs (71% vs. 29%).
Women older than 20 years were more likely to get IUDs than implants, with those older than 25 preferring them by a ratio of 70% to 30%.
After researchers controlled for such factors as age group and year of insertion, they found that implant recipients were slightly more likely to still have the contraception tool in place at 1 year: 81% (implants) vs. IUDs (77%; P = .01). It’s not clear why women had their implants or IUDs removed.
Claims for complications were similar between the groups (8% for implants, 7% for IUDs), and researchers didn’t find a statistically significant difference after they controlled for various factors. Dysfunctional uterine bleeding was the most common complication, followed by excessive or frequent menstruation.
Few women reported pregnancy among the implant (0.82%) and IUD (0.86%) groups, and there was no statistically significant difference between the groups.
The researchers reported that their study has various limitations: The insurance claims information may provide misleading information about minor complications, and it doesn’t include details about abortions. The claims also may not report IUD expulsions and self-removals.
The authors also noted that they didn’t control for factors like socioeconomic status, psychological conditions, and multimorbidity.
MedStar Family Choice provided study funding. Dr. Romano and Loral Patchen, PhD, disclosed receiving free CME trainings and insertion certification regarding the long-acting contraceptive devices discussed in the study from Merck, Bayer, and Teva. Dr. Patchen disclosed serving on the Diclegis speakers bureau.
FROM CONTRACEPTION
Key clinical point:
Major finding: For implants and IUDS, respectively, 1-year continuation rates were similar (adjusted 81% vs. 77%, P = .01), as were complication rates (unadjusted 8% for implants, 7% for IUDs).
Study details: Retrospective analysis of 3,305 Medicaid-covered women treated with the two types of contraceptives during 2012-2015 in the District of Columbia and Maryland.
Disclosures: MedStar Family Choice provided study funding. Dr. Romano and Loral Patchen, PhD, disclosed receiving free CME trainings and insertion certification regarding the long-acting contraceptive devices discussed in the study from Merck, Bayer, and Teva. Dr. Patchen discloses serving on the Diclegis speakers bureau.
Source: Romano MJ et al. Contraception. 2018 Aug;98:125-9.
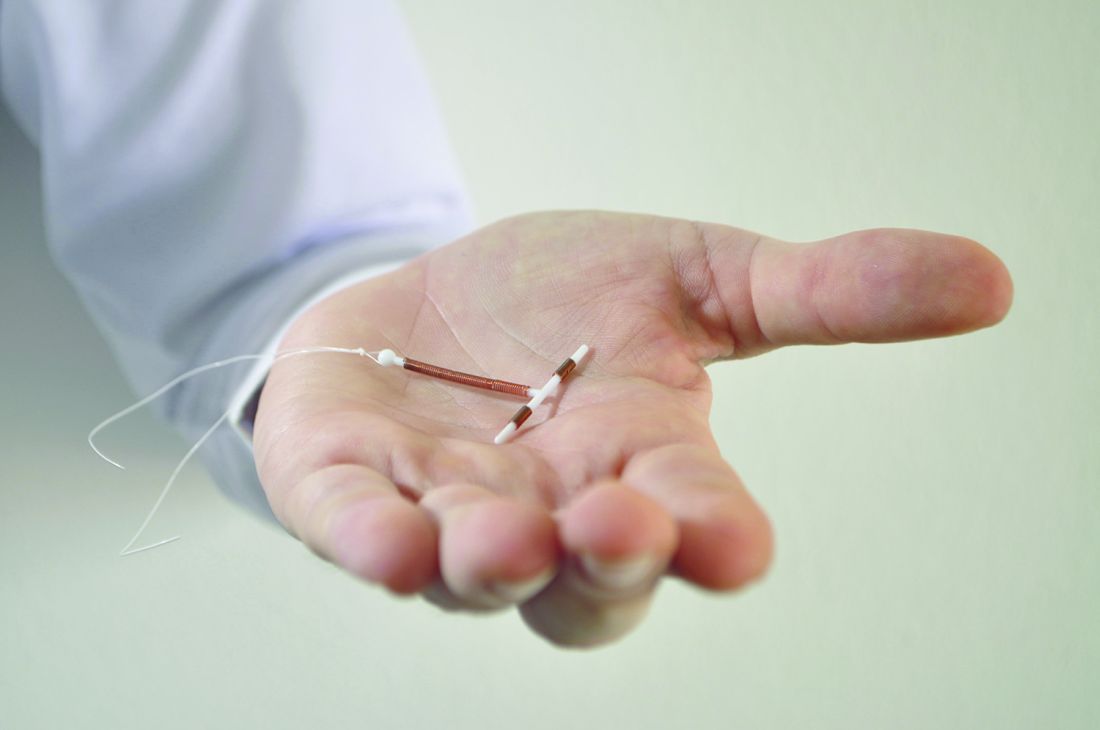
New IUD expelled less often after C-section than older device
that’s inserted the same way, according to results of a Turkish study.
The study authors, led by Ceren Unal, MD, of Zeynep Kamil Women’s and Children’s Disease Training and Research Hospital, Istanbul, wrote that the new device, the frameless copper-releasing Gyn-CS IUD, “could be a major advance, potentially suitable for general use due to the ease and safety of the insertion procedure,” which requires limited training.
According to the study authors, most IUDs are retained in the uterus, and most have a T-shape configuration. Some are inserted immediately following expulsion of the placenta at birth, although techniques “are far from being optimal,” and devices are frequently displaced and sometimes expelled.
The newly developed Gyn-CS IUD, a redesigned version of a previous model, has an alternative “anchor” design.
The investigators tracked 140 pregnant women – 106 who underwent elective cesarean section and 34 who underwent emergency cesarean section – who had the Gyn-CS or the TCu380A IUD inserted shortly after they gave birth. Their median age was around 30 years , all were white, and all were married.
The TCu380A IUD, a decades-old model that’s been recommended by the World Health Organization, and the Gyn-CS IUD were each inserted in 70 women. The researchers then followed them for 3 months. Three women (one in the Gyn-CS IUD group and two in the TCu380A IUD group) were lost to follow-up.
In total, 61 of 70 IUDs (88%) remained in place in the Gyn-CS group, and 54 of 70 (79%) in the TCu380A group (P = .30). One (1%) Gyn-CS IUD was expelled , possibly because it was incorrectly anchored, and eight (11%) TCu380A IUDs were expelled (P = .04). There were equal numbers of medical removals (four) and nonmedical removals (two) in the two groups, and one “other medical removal” in the Gyn-CS IUD group.
“The very low expulsion rate of Gyn-CS, compared with TCu380A, is a very strong argument in favor of the anchored IUD, preventing expulsion and displacement,” Dr. Unal and colleagues reported in Contraception.
They noted that the study has a limited 3-month tracking period, but they also pointed out that most expulsions inserted post partum occurred in the initial 6 weeks.
The low expulsion rate of the Gyn-CS device “will prevent more women becoming pregnant too soon which constitutes an important safety issue during future pregnancy,” the authors wrote. “The device, preferably its high-load 10-year version, could also interest many women as a reversible alternative for tubal sterilization. Additional clinical experience with the Gyn-CS IUD is, therefore, urgently warranted.”
No study funding is reported, although Contrel Research provided the Gyn-CS devices at no charge. The study authors reported no relevant disclosures.
that’s inserted the same way, according to results of a Turkish study.
The study authors, led by Ceren Unal, MD, of Zeynep Kamil Women’s and Children’s Disease Training and Research Hospital, Istanbul, wrote that the new device, the frameless copper-releasing Gyn-CS IUD, “could be a major advance, potentially suitable for general use due to the ease and safety of the insertion procedure,” which requires limited training.
According to the study authors, most IUDs are retained in the uterus, and most have a T-shape configuration. Some are inserted immediately following expulsion of the placenta at birth, although techniques “are far from being optimal,” and devices are frequently displaced and sometimes expelled.
The newly developed Gyn-CS IUD, a redesigned version of a previous model, has an alternative “anchor” design.
The investigators tracked 140 pregnant women – 106 who underwent elective cesarean section and 34 who underwent emergency cesarean section – who had the Gyn-CS or the TCu380A IUD inserted shortly after they gave birth. Their median age was around 30 years , all were white, and all were married.
The TCu380A IUD, a decades-old model that’s been recommended by the World Health Organization, and the Gyn-CS IUD were each inserted in 70 women. The researchers then followed them for 3 months. Three women (one in the Gyn-CS IUD group and two in the TCu380A IUD group) were lost to follow-up.
In total, 61 of 70 IUDs (88%) remained in place in the Gyn-CS group, and 54 of 70 (79%) in the TCu380A group (P = .30). One (1%) Gyn-CS IUD was expelled , possibly because it was incorrectly anchored, and eight (11%) TCu380A IUDs were expelled (P = .04). There were equal numbers of medical removals (four) and nonmedical removals (two) in the two groups, and one “other medical removal” in the Gyn-CS IUD group.
“The very low expulsion rate of Gyn-CS, compared with TCu380A, is a very strong argument in favor of the anchored IUD, preventing expulsion and displacement,” Dr. Unal and colleagues reported in Contraception.
They noted that the study has a limited 3-month tracking period, but they also pointed out that most expulsions inserted post partum occurred in the initial 6 weeks.
The low expulsion rate of the Gyn-CS device “will prevent more women becoming pregnant too soon which constitutes an important safety issue during future pregnancy,” the authors wrote. “The device, preferably its high-load 10-year version, could also interest many women as a reversible alternative for tubal sterilization. Additional clinical experience with the Gyn-CS IUD is, therefore, urgently warranted.”
No study funding is reported, although Contrel Research provided the Gyn-CS devices at no charge. The study authors reported no relevant disclosures.
that’s inserted the same way, according to results of a Turkish study.
The study authors, led by Ceren Unal, MD, of Zeynep Kamil Women’s and Children’s Disease Training and Research Hospital, Istanbul, wrote that the new device, the frameless copper-releasing Gyn-CS IUD, “could be a major advance, potentially suitable for general use due to the ease and safety of the insertion procedure,” which requires limited training.
According to the study authors, most IUDs are retained in the uterus, and most have a T-shape configuration. Some are inserted immediately following expulsion of the placenta at birth, although techniques “are far from being optimal,” and devices are frequently displaced and sometimes expelled.
The newly developed Gyn-CS IUD, a redesigned version of a previous model, has an alternative “anchor” design.
The investigators tracked 140 pregnant women – 106 who underwent elective cesarean section and 34 who underwent emergency cesarean section – who had the Gyn-CS or the TCu380A IUD inserted shortly after they gave birth. Their median age was around 30 years , all were white, and all were married.
The TCu380A IUD, a decades-old model that’s been recommended by the World Health Organization, and the Gyn-CS IUD were each inserted in 70 women. The researchers then followed them for 3 months. Three women (one in the Gyn-CS IUD group and two in the TCu380A IUD group) were lost to follow-up.
In total, 61 of 70 IUDs (88%) remained in place in the Gyn-CS group, and 54 of 70 (79%) in the TCu380A group (P = .30). One (1%) Gyn-CS IUD was expelled , possibly because it was incorrectly anchored, and eight (11%) TCu380A IUDs were expelled (P = .04). There were equal numbers of medical removals (four) and nonmedical removals (two) in the two groups, and one “other medical removal” in the Gyn-CS IUD group.
“The very low expulsion rate of Gyn-CS, compared with TCu380A, is a very strong argument in favor of the anchored IUD, preventing expulsion and displacement,” Dr. Unal and colleagues reported in Contraception.
They noted that the study has a limited 3-month tracking period, but they also pointed out that most expulsions inserted post partum occurred in the initial 6 weeks.
The low expulsion rate of the Gyn-CS device “will prevent more women becoming pregnant too soon which constitutes an important safety issue during future pregnancy,” the authors wrote. “The device, preferably its high-load 10-year version, could also interest many women as a reversible alternative for tubal sterilization. Additional clinical experience with the Gyn-CS IUD is, therefore, urgently warranted.”
No study funding is reported, although Contrel Research provided the Gyn-CS devices at no charge. The study authors reported no relevant disclosures.
FROM CONTRACEPTION
Key clinical point: A newly developed IUD appears to be expelled less frequently than an older device after insertion following cesarean section births.
Major finding: 61 of 70 IUDs (88%) remained in place in women who received the frameless copper-releasing Gyn-CS device, while 54 of 70 (79%) did so in those who received the TCu380A device (P = .30).
Study details: Randomized trial of 140 women who underwent cesarean section followed by insertion of one of the two IUD types (n = 70 for each).
Disclosures: No study funding is reported, although Contrel Research provided the Gyn-CS devices at no charge. The study authors reported no relevant disclosures.
Source: Unal C et al. Contraception. 2018 Aug;98:135-40.
What do you call a koala who is too sweet for its own good? Diabetic
SAN DIEGO – The 14-pound patient with the deep-pile complexion was lethargic, kept drinking a lot of water, and had a glucose level in the range of 600-700 mg/dL. He was nearly comatose by the time medical staff transferred him to a specialized facility.
The diagnosis: Diabetes. The treatment: Insulin. But multiple daily skin pricks were quite a challenge for Quincy the koala. After all, he requires up to 22 hours of shut-eye each day.
What to do? The veterinary staff at the San Diego Zoo turned to the experts – an endocrinologist and a manufacturer of continuous glucose monitors. Now, Quincy has his own CGM, and a medical team that is tracking his glucose levels in real time on their smartphones.
In fact, Athena Philis-Tsimikas, MD, of the Scripps Whittier Diabetes Institute, pulls out her phone and checks on him at least a couple times a day. She also gets alerts if his blood sugar drops too quickly.
“He is definitely another one of my patients,” she said in an interview. But he’s the only one who lives in trees and enjoys a nice eucalyptus smoothie.
Humans are hardly the only mammals who get diabetes
Veterinarians are quite familiar with diabetes. A wide variety of mammals from pigs and apes to horses and dolphins can develop an equivalent of the human condition. Dogs may be prescribed daily insulin shots, and cats even develop peripheral neuropathy and retinopathy like humans with diabetes.
So it’s not entirely surprising that a team at the Los Angeles Zoo diagnosed Quincy, a 3-year-old Queensland koala, with diabetes.
Quincy’s glucose levels should have been around 80-130 mg/dL, similar to the ideal levels in humans, said San Diego Zoo senior veterinarian Cora Singleton, DVM, in an interview. But tests prompted by his symptoms showed his levels were high, she said, and they stayed that way. According to her, that suggested he wasn’t just having a one-time elevation that animals can experience when they’re stressed.
Unfortunately, there are only a few scattered reports of diabetes in koalas, and “there’s not anything documented about treating a koala over a long term,” Dr. Singleton said. “We’re in uncharted territory here.”
So the Los Angeles Zoo sent Quincy down the California coast for more specialized treatment. The San Diego Zoo’s veterinary staff took in Quincy and treated him with glucose tests and insulin shots, Dr. Singleton said. “But we were looking a way for to get more information with less disturbance to Quincy.”
Someone mentioned the idea of a sensor. “We thought, ‘What a great idea,’” Dr. Singleton said. “It would be a way for us to get a lot of information and find out how his highs and lows are related.”
That’s when the team turned to local endocrinologist Dr. Tsimikas for a helping hand.
The key to koala calming: Eucalyptus smoothies
“They did reach out to us and asked what kind of sensors might be available. We connected them to Dexcom,” a CGM company that’s based in San Diego, Dr. Tsimikas said. “We knew the newest one was coming along and suggested they place that on him as a starting point.”
On June 1, a zoo team attached a Dexcom G6 Continuous Glucose Monitoring System to the koala’s side.
“He’s doing very well. He tolerates the CGM superbly,” Dr. Singleton said. And Quincy doesn’t react when sensors are applied, she said, although it helps that he gets to enjoy a eucalyptus smoothie during the procedure. “Put that in a big syringe, and he’ll volunteer for most anything,” she said.
Obesity can trigger diabetes in mammals other than humans. Could eucalyptus overindulgence explain Quincy’s case of diabetes? Nope.
According to Dr. Tsimikas, the ingredients of the eucalyptus smoothie are just pureed eucalyptus leaves that “go down fast and easy.” These naturally have a nice mix of carbohydrates, fat, and protein to better manage the koala’s sugars and other nutritional needs. If he is dropping his blood sugar values fast, there is another dextrose drink they give him in small amounts, which contains 5-10 g carbohydrates. This is enough to help bring his glucose values back up. It is similar to the treatment recommendations provided to humans with diabetes where they are told to take 15 g of carbohydrates such as honey, hard candies, or juice to prevent a severe hypoglycemic episode.
Dr. Singleton noted that Quincy appears to have the koala equivalent is type 1 diabetes mellitus (T1DM).
Dr. Tsimikas noted “We are not finding the typical antibodies that we find in human T1DM. Quincy is showing low insulin levels, which is why it more closely resembles T1DM. We will be doing further analysis and comparisons with nondiabetic koalas in the future to see if it can be better differentiated.
While he appears to have type 1 diabetes, it’s not clear why he developed it, Dr. Singleton said.
While Quincy is only 3 years old, he’s a full-fledged adult in koala terms. Koalas typically live up to their mid-teens, she said.
This speechless patient still manages to communicate
The San Diego Zoo’s veterinary staff is monitoring Quincy and trying to understand how his glucose levels and daily insulin shots affect him. His tiny size has ruled out use of an insulin pump: Although the insulin pumps have been getting smaller and lighter, they are still too large to attach to our tiny friend. Especially since he would need both the CGM device and the pump, there is not a lot of surface area on his body for attachment of all the devices, according to Dr. Tsimikas.
Since Quincy is so tiny, insulin doses must be minuscule to avoid sending him into hypoglycemia, Dr. Tsimikas said. She said the koala’s medical team is planning to try using a NovoPen Echo injector with a half-unit of insulin.
Dr. Singleton noted that for now, “he’s maintaining his body weight, and he has days when he feels spunky. Sometimes, when he knows it’s breakfast time, and he hears his caretakers coming up the doorway with his breakfast, he’ll be very active on his perch.”
But he has sluggish days, too, when he’ll try to sleep in. Dr. Singleton keeps an eye out for grogginess and signs of weakness and hypoglycemia or hyperglycemia like “a little wobble in his step.”
“The biggest thing I’ve learned from Quincy is the value of his particular nonverbal cues,” she said. “I’m starting to understand when he feels like his sugars are a little high or a little low. I imagine that doctors and parents have the same challenges with little patients, along with figuring out how you communicate that this is supposed to help them.”
Dr. Tsimikas agreed, noting that she sees similarities between Quincy and patients who are hospitalized and can’t easily communicate. Now, “we can track the folks who are on the CGM and intervene earlier than before,” said Dr. Tsimikas, who’s part of a clinical trial team testing CGM devices in two hospitals. “It’s almost like having another vital sign.
“It is only when we have all the data on all the other factors that can influence blood sugar, such as eating patterns, insulin dose and timing, and activity level that we can more accurately adjust the medical interventions.” This requires collaboration between all the groups involved in Quincy’s care. In koalas, the collaboration is with the veterinarian, koala zookeepers, dietitian, and the technology monitoring team. Whereas, for humans, we need parents, care providers, diabetes educators, dietitians, and physicians.
It’s not clear if Quincy will need his CGM for the rest of his life. If he’s stable on a specific insulin dose, Dr. Tsimikas said, he may not need it. But it sounds like eucalyptus smoothies will always be a vital part of his regimen.
In the name of thoroughness, take note that Quincy is not the first diabetic zoo animal whose care involved physicians from Scripps. “We have had several other consultations for animals with diabetes. Nearly 25 years ago, a roller-skating chimpanzee with diabetes was brought to the Scripps Whittier Institute labs for evaluation and treatment recommendations. A few years later, one of our medical directors, Alberto Hayek, MD, advised on the care of Lune, a diabetic baboon at the San Diego Zoo, for insulin management. This time we are making house calls to the zoo to treat Quincy in his home environment. Each animal experience offers opportunities to expand our knowledge about diabetes care and exchange approaches that we might not otherwise be aware of. This has been fun and rewarding. I am looking forward to seeing further outcomes from our interactions with Quincy,” according to Dr. Tsimikas.
Dr. Tsimikas reports that her center conducts research with Dexcom and Novo Nordisk. Dr. Singleton reports no relevant disclosures.
SAN DIEGO – The 14-pound patient with the deep-pile complexion was lethargic, kept drinking a lot of water, and had a glucose level in the range of 600-700 mg/dL. He was nearly comatose by the time medical staff transferred him to a specialized facility.
The diagnosis: Diabetes. The treatment: Insulin. But multiple daily skin pricks were quite a challenge for Quincy the koala. After all, he requires up to 22 hours of shut-eye each day.
What to do? The veterinary staff at the San Diego Zoo turned to the experts – an endocrinologist and a manufacturer of continuous glucose monitors. Now, Quincy has his own CGM, and a medical team that is tracking his glucose levels in real time on their smartphones.
In fact, Athena Philis-Tsimikas, MD, of the Scripps Whittier Diabetes Institute, pulls out her phone and checks on him at least a couple times a day. She also gets alerts if his blood sugar drops too quickly.
“He is definitely another one of my patients,” she said in an interview. But he’s the only one who lives in trees and enjoys a nice eucalyptus smoothie.
Humans are hardly the only mammals who get diabetes
Veterinarians are quite familiar with diabetes. A wide variety of mammals from pigs and apes to horses and dolphins can develop an equivalent of the human condition. Dogs may be prescribed daily insulin shots, and cats even develop peripheral neuropathy and retinopathy like humans with diabetes.
So it’s not entirely surprising that a team at the Los Angeles Zoo diagnosed Quincy, a 3-year-old Queensland koala, with diabetes.
Quincy’s glucose levels should have been around 80-130 mg/dL, similar to the ideal levels in humans, said San Diego Zoo senior veterinarian Cora Singleton, DVM, in an interview. But tests prompted by his symptoms showed his levels were high, she said, and they stayed that way. According to her, that suggested he wasn’t just having a one-time elevation that animals can experience when they’re stressed.
Unfortunately, there are only a few scattered reports of diabetes in koalas, and “there’s not anything documented about treating a koala over a long term,” Dr. Singleton said. “We’re in uncharted territory here.”
So the Los Angeles Zoo sent Quincy down the California coast for more specialized treatment. The San Diego Zoo’s veterinary staff took in Quincy and treated him with glucose tests and insulin shots, Dr. Singleton said. “But we were looking a way for to get more information with less disturbance to Quincy.”
Someone mentioned the idea of a sensor. “We thought, ‘What a great idea,’” Dr. Singleton said. “It would be a way for us to get a lot of information and find out how his highs and lows are related.”
That’s when the team turned to local endocrinologist Dr. Tsimikas for a helping hand.
The key to koala calming: Eucalyptus smoothies
“They did reach out to us and asked what kind of sensors might be available. We connected them to Dexcom,” a CGM company that’s based in San Diego, Dr. Tsimikas said. “We knew the newest one was coming along and suggested they place that on him as a starting point.”
On June 1, a zoo team attached a Dexcom G6 Continuous Glucose Monitoring System to the koala’s side.
“He’s doing very well. He tolerates the CGM superbly,” Dr. Singleton said. And Quincy doesn’t react when sensors are applied, she said, although it helps that he gets to enjoy a eucalyptus smoothie during the procedure. “Put that in a big syringe, and he’ll volunteer for most anything,” she said.
Obesity can trigger diabetes in mammals other than humans. Could eucalyptus overindulgence explain Quincy’s case of diabetes? Nope.
According to Dr. Tsimikas, the ingredients of the eucalyptus smoothie are just pureed eucalyptus leaves that “go down fast and easy.” These naturally have a nice mix of carbohydrates, fat, and protein to better manage the koala’s sugars and other nutritional needs. If he is dropping his blood sugar values fast, there is another dextrose drink they give him in small amounts, which contains 5-10 g carbohydrates. This is enough to help bring his glucose values back up. It is similar to the treatment recommendations provided to humans with diabetes where they are told to take 15 g of carbohydrates such as honey, hard candies, or juice to prevent a severe hypoglycemic episode.
Dr. Singleton noted that Quincy appears to have the koala equivalent is type 1 diabetes mellitus (T1DM).
Dr. Tsimikas noted “We are not finding the typical antibodies that we find in human T1DM. Quincy is showing low insulin levels, which is why it more closely resembles T1DM. We will be doing further analysis and comparisons with nondiabetic koalas in the future to see if it can be better differentiated.
While he appears to have type 1 diabetes, it’s not clear why he developed it, Dr. Singleton said.
While Quincy is only 3 years old, he’s a full-fledged adult in koala terms. Koalas typically live up to their mid-teens, she said.
This speechless patient still manages to communicate
The San Diego Zoo’s veterinary staff is monitoring Quincy and trying to understand how his glucose levels and daily insulin shots affect him. His tiny size has ruled out use of an insulin pump: Although the insulin pumps have been getting smaller and lighter, they are still too large to attach to our tiny friend. Especially since he would need both the CGM device and the pump, there is not a lot of surface area on his body for attachment of all the devices, according to Dr. Tsimikas.
Since Quincy is so tiny, insulin doses must be minuscule to avoid sending him into hypoglycemia, Dr. Tsimikas said. She said the koala’s medical team is planning to try using a NovoPen Echo injector with a half-unit of insulin.
Dr. Singleton noted that for now, “he’s maintaining his body weight, and he has days when he feels spunky. Sometimes, when he knows it’s breakfast time, and he hears his caretakers coming up the doorway with his breakfast, he’ll be very active on his perch.”
But he has sluggish days, too, when he’ll try to sleep in. Dr. Singleton keeps an eye out for grogginess and signs of weakness and hypoglycemia or hyperglycemia like “a little wobble in his step.”
“The biggest thing I’ve learned from Quincy is the value of his particular nonverbal cues,” she said. “I’m starting to understand when he feels like his sugars are a little high or a little low. I imagine that doctors and parents have the same challenges with little patients, along with figuring out how you communicate that this is supposed to help them.”
Dr. Tsimikas agreed, noting that she sees similarities between Quincy and patients who are hospitalized and can’t easily communicate. Now, “we can track the folks who are on the CGM and intervene earlier than before,” said Dr. Tsimikas, who’s part of a clinical trial team testing CGM devices in two hospitals. “It’s almost like having another vital sign.
“It is only when we have all the data on all the other factors that can influence blood sugar, such as eating patterns, insulin dose and timing, and activity level that we can more accurately adjust the medical interventions.” This requires collaboration between all the groups involved in Quincy’s care. In koalas, the collaboration is with the veterinarian, koala zookeepers, dietitian, and the technology monitoring team. Whereas, for humans, we need parents, care providers, diabetes educators, dietitians, and physicians.
It’s not clear if Quincy will need his CGM for the rest of his life. If he’s stable on a specific insulin dose, Dr. Tsimikas said, he may not need it. But it sounds like eucalyptus smoothies will always be a vital part of his regimen.
In the name of thoroughness, take note that Quincy is not the first diabetic zoo animal whose care involved physicians from Scripps. “We have had several other consultations for animals with diabetes. Nearly 25 years ago, a roller-skating chimpanzee with diabetes was brought to the Scripps Whittier Institute labs for evaluation and treatment recommendations. A few years later, one of our medical directors, Alberto Hayek, MD, advised on the care of Lune, a diabetic baboon at the San Diego Zoo, for insulin management. This time we are making house calls to the zoo to treat Quincy in his home environment. Each animal experience offers opportunities to expand our knowledge about diabetes care and exchange approaches that we might not otherwise be aware of. This has been fun and rewarding. I am looking forward to seeing further outcomes from our interactions with Quincy,” according to Dr. Tsimikas.
Dr. Tsimikas reports that her center conducts research with Dexcom and Novo Nordisk. Dr. Singleton reports no relevant disclosures.
SAN DIEGO – The 14-pound patient with the deep-pile complexion was lethargic, kept drinking a lot of water, and had a glucose level in the range of 600-700 mg/dL. He was nearly comatose by the time medical staff transferred him to a specialized facility.
The diagnosis: Diabetes. The treatment: Insulin. But multiple daily skin pricks were quite a challenge for Quincy the koala. After all, he requires up to 22 hours of shut-eye each day.
What to do? The veterinary staff at the San Diego Zoo turned to the experts – an endocrinologist and a manufacturer of continuous glucose monitors. Now, Quincy has his own CGM, and a medical team that is tracking his glucose levels in real time on their smartphones.
In fact, Athena Philis-Tsimikas, MD, of the Scripps Whittier Diabetes Institute, pulls out her phone and checks on him at least a couple times a day. She also gets alerts if his blood sugar drops too quickly.
“He is definitely another one of my patients,” she said in an interview. But he’s the only one who lives in trees and enjoys a nice eucalyptus smoothie.
Humans are hardly the only mammals who get diabetes
Veterinarians are quite familiar with diabetes. A wide variety of mammals from pigs and apes to horses and dolphins can develop an equivalent of the human condition. Dogs may be prescribed daily insulin shots, and cats even develop peripheral neuropathy and retinopathy like humans with diabetes.
So it’s not entirely surprising that a team at the Los Angeles Zoo diagnosed Quincy, a 3-year-old Queensland koala, with diabetes.
Quincy’s glucose levels should have been around 80-130 mg/dL, similar to the ideal levels in humans, said San Diego Zoo senior veterinarian Cora Singleton, DVM, in an interview. But tests prompted by his symptoms showed his levels were high, she said, and they stayed that way. According to her, that suggested he wasn’t just having a one-time elevation that animals can experience when they’re stressed.
Unfortunately, there are only a few scattered reports of diabetes in koalas, and “there’s not anything documented about treating a koala over a long term,” Dr. Singleton said. “We’re in uncharted territory here.”
So the Los Angeles Zoo sent Quincy down the California coast for more specialized treatment. The San Diego Zoo’s veterinary staff took in Quincy and treated him with glucose tests and insulin shots, Dr. Singleton said. “But we were looking a way for to get more information with less disturbance to Quincy.”
Someone mentioned the idea of a sensor. “We thought, ‘What a great idea,’” Dr. Singleton said. “It would be a way for us to get a lot of information and find out how his highs and lows are related.”
That’s when the team turned to local endocrinologist Dr. Tsimikas for a helping hand.
The key to koala calming: Eucalyptus smoothies
“They did reach out to us and asked what kind of sensors might be available. We connected them to Dexcom,” a CGM company that’s based in San Diego, Dr. Tsimikas said. “We knew the newest one was coming along and suggested they place that on him as a starting point.”
On June 1, a zoo team attached a Dexcom G6 Continuous Glucose Monitoring System to the koala’s side.
“He’s doing very well. He tolerates the CGM superbly,” Dr. Singleton said. And Quincy doesn’t react when sensors are applied, she said, although it helps that he gets to enjoy a eucalyptus smoothie during the procedure. “Put that in a big syringe, and he’ll volunteer for most anything,” she said.
Obesity can trigger diabetes in mammals other than humans. Could eucalyptus overindulgence explain Quincy’s case of diabetes? Nope.
According to Dr. Tsimikas, the ingredients of the eucalyptus smoothie are just pureed eucalyptus leaves that “go down fast and easy.” These naturally have a nice mix of carbohydrates, fat, and protein to better manage the koala’s sugars and other nutritional needs. If he is dropping his blood sugar values fast, there is another dextrose drink they give him in small amounts, which contains 5-10 g carbohydrates. This is enough to help bring his glucose values back up. It is similar to the treatment recommendations provided to humans with diabetes where they are told to take 15 g of carbohydrates such as honey, hard candies, or juice to prevent a severe hypoglycemic episode.
Dr. Singleton noted that Quincy appears to have the koala equivalent is type 1 diabetes mellitus (T1DM).
Dr. Tsimikas noted “We are not finding the typical antibodies that we find in human T1DM. Quincy is showing low insulin levels, which is why it more closely resembles T1DM. We will be doing further analysis and comparisons with nondiabetic koalas in the future to see if it can be better differentiated.
While he appears to have type 1 diabetes, it’s not clear why he developed it, Dr. Singleton said.
While Quincy is only 3 years old, he’s a full-fledged adult in koala terms. Koalas typically live up to their mid-teens, she said.
This speechless patient still manages to communicate
The San Diego Zoo’s veterinary staff is monitoring Quincy and trying to understand how his glucose levels and daily insulin shots affect him. His tiny size has ruled out use of an insulin pump: Although the insulin pumps have been getting smaller and lighter, they are still too large to attach to our tiny friend. Especially since he would need both the CGM device and the pump, there is not a lot of surface area on his body for attachment of all the devices, according to Dr. Tsimikas.
Since Quincy is so tiny, insulin doses must be minuscule to avoid sending him into hypoglycemia, Dr. Tsimikas said. She said the koala’s medical team is planning to try using a NovoPen Echo injector with a half-unit of insulin.
Dr. Singleton noted that for now, “he’s maintaining his body weight, and he has days when he feels spunky. Sometimes, when he knows it’s breakfast time, and he hears his caretakers coming up the doorway with his breakfast, he’ll be very active on his perch.”
But he has sluggish days, too, when he’ll try to sleep in. Dr. Singleton keeps an eye out for grogginess and signs of weakness and hypoglycemia or hyperglycemia like “a little wobble in his step.”
“The biggest thing I’ve learned from Quincy is the value of his particular nonverbal cues,” she said. “I’m starting to understand when he feels like his sugars are a little high or a little low. I imagine that doctors and parents have the same challenges with little patients, along with figuring out how you communicate that this is supposed to help them.”
Dr. Tsimikas agreed, noting that she sees similarities between Quincy and patients who are hospitalized and can’t easily communicate. Now, “we can track the folks who are on the CGM and intervene earlier than before,” said Dr. Tsimikas, who’s part of a clinical trial team testing CGM devices in two hospitals. “It’s almost like having another vital sign.
“It is only when we have all the data on all the other factors that can influence blood sugar, such as eating patterns, insulin dose and timing, and activity level that we can more accurately adjust the medical interventions.” This requires collaboration between all the groups involved in Quincy’s care. In koalas, the collaboration is with the veterinarian, koala zookeepers, dietitian, and the technology monitoring team. Whereas, for humans, we need parents, care providers, diabetes educators, dietitians, and physicians.
It’s not clear if Quincy will need his CGM for the rest of his life. If he’s stable on a specific insulin dose, Dr. Tsimikas said, he may not need it. But it sounds like eucalyptus smoothies will always be a vital part of his regimen.
In the name of thoroughness, take note that Quincy is not the first diabetic zoo animal whose care involved physicians from Scripps. “We have had several other consultations for animals with diabetes. Nearly 25 years ago, a roller-skating chimpanzee with diabetes was brought to the Scripps Whittier Institute labs for evaluation and treatment recommendations. A few years later, one of our medical directors, Alberto Hayek, MD, advised on the care of Lune, a diabetic baboon at the San Diego Zoo, for insulin management. This time we are making house calls to the zoo to treat Quincy in his home environment. Each animal experience offers opportunities to expand our knowledge about diabetes care and exchange approaches that we might not otherwise be aware of. This has been fun and rewarding. I am looking forward to seeing further outcomes from our interactions with Quincy,” according to Dr. Tsimikas.
Dr. Tsimikas reports that her center conducts research with Dexcom and Novo Nordisk. Dr. Singleton reports no relevant disclosures.
Herceptin linked to doubling of HF risk in women with breast cancer
Adding more evidence to an ongoing debate, a large new study suggests that patients with breast cancer who take trastuzumab (Herceptin) may face double the adjusted risk of developing heart failure, with older women at highest risk.
The study also found that most patients who took trastuzumab didn’t receive recommended cardiac screening.
The researchers said their findings are unique because they tracked both younger and older patients. “By examining the rates of both cardiac monitoring and cardiotoxicity, we could begin to address the controversial issue of whether cardiac monitoring is warranted in young breast cancer patients,” wrote Mariana Chavez-MacGregor, MD, MSC, of the University of Texas MD Anderson Cancer Center, and her associates. The report was published in JACC: Cardiovascular Imaging.
While Trastuzumab has boosted breast cancer survival rates for patients with HER2-positive tumors, it’s also raised concerns about cardiotoxicity that could be an indicator of subsequent congestive heart failure (Cochrane Database Syst Rev. 2014 Jun 12;(6):CD006242).
According to the new study, the risk of trastuzumab risk is linked to damage to cardiac myocytes that can cause reversible cardiotoxicity.
The prescribing information for trastuzumab advises patients to undergo cardiac monitoring before treatment with trastuzumab and every 3 months during treatment. Recommendations by medical organizations have varied.
Now, as a 2016 report put it, it’s “increasingly unclear” whether frequent routine monitoring is appropriate for all patients (J Clin Oncol. 2016 Apr 1;34[10]:1030-3).
For the current study, Dr. Chavez-MacGregor and her associates identified 16,456 adult women in the United States who were diagnosed with nonmetastatic invasive breast cancer from 2009 to 2014. Researchers tracked the group, with a median age of 56, through as late as 2015.
The women were treated with chemotherapy within 6 months of diagnosis, and 4,325 received trastuzumab.
Of all the subjects, 692 patients (4.2%) developed heart failure following chemotherapy. The rate among patients treated with trastuzumab was higher, at 8.3%, compared with 2.7% for those not treated with trastuzumab (P less than .001).
The researchers also looked at anthracycline users and found that they were slightly more likely to develop HF (4.6% vs. 4.0% among nonusers, P = .048).
Increased age boosted the risk of HF in the trastuzumab-treated patients, and the risk was highest in those treated with both anthracyclines and trastuzumab. Other factors linked to more risk were comorbidities, hypertension, and valve disease.
After adjusting for confounders, the researchers estimated that those treated with trastuzumab were 2.01 times more likely to develop HF (HR, 2.01; 95% confidence interval, 1.72-2.36), and those who took anthracycline were 1.53 times more likely (HR, 1.53; 95% CI, 1.30-1.80)
The researchers also examined medical records for evidence that subjects underwent cardiac screening at least once every 4 months, not 3 months, as the prescribing information recommends. The study team chose to focus on 4-month intervals “to compensate for differences in scheduling, resources, or levels of accessibility to medical care.”
Medical records suggest that 73.5% of patients who took trastuzumab underwent cardiac screening at the beginning of therapy, but only 46.2% continued to do so at least every 4 months.
An adjusted model linked more screening to the use of anthracyclines and taxanes, radiation treatment, and living in the Northeast vs. the West.
“HF was more frequently identified among patients undergoing recommended cardiac monitoring (10.4% compared with 6.5%, respectively; P less than.001), suggesting that, as more patients are screened, more patients are likely to be found having HF,” the researchers reported.
However, they added that “our sensitivity analysis using inpatient claims allowed us to determine that the HF identified using cardiac monitoring was not severe enough to require hospitalization and was likely asymptomatic. The clinical implications of the diagnosis of asymptomatic HF are hard to determine and are beyond the scope of this study.”
The researchers also noted that the findings suggest that screening has become more common in recent years.
“The number of cancer survivors is expected to increase over time, and we will continue to see patients develop treatment-related cardiotoxicity,” the researchers wrote. “Thus, more research, evidence-based guidelines, and tools for prediction of cancer treatment–related cardiotoxicity are needed.”
The National Cancer Institute and Cancer Prevention and Research Institute of Texas funded the study. Two study authors reported grant funding from the Susan G. Komen Breast Cancer Foundation, and one reports consulting for Pfizer and Roche. The other authors reported no disclosures.
SOURCE: Chavez-MacGregor M et al. JACC: Cardiovasc Imaging. 2018 Aug;11[8]1084-93.
While trastuzumab clearly benefits patients with HER2-positive breast cancer at various stages of progression, concerns about heart failure persist. Studies have suggested that the drug doesn’t boost the risk of late cardiac events, but it’s not clear if this is due to mandated screening in these trials. The new study provides more evidence that adherence to screening guidelines is limited, and recent trials offer evidence that the general cardiac risk may be overblown. Future studies could be designed to offer insight into the wisdom of adjusting screening regimens based on stratification of risk. A meta-analysis could also be helpful, and the upcoming results of the SAFE-HEART study will provide information about the safety of anti-HER2 antibody therapy in patients with low but asymptomatic left-ventricular ejection fraction.
These comments are excerpted from a commentary by Chau T. Dang, MD, of Memorial Sloan Kettering Cancer Center, New York, and her associates (JACC: Cardiovasc Imaging. 2018 Aug;11[8]:1094-7). Most of the commentary authors report various disclosures.
While trastuzumab clearly benefits patients with HER2-positive breast cancer at various stages of progression, concerns about heart failure persist. Studies have suggested that the drug doesn’t boost the risk of late cardiac events, but it’s not clear if this is due to mandated screening in these trials. The new study provides more evidence that adherence to screening guidelines is limited, and recent trials offer evidence that the general cardiac risk may be overblown. Future studies could be designed to offer insight into the wisdom of adjusting screening regimens based on stratification of risk. A meta-analysis could also be helpful, and the upcoming results of the SAFE-HEART study will provide information about the safety of anti-HER2 antibody therapy in patients with low but asymptomatic left-ventricular ejection fraction.
These comments are excerpted from a commentary by Chau T. Dang, MD, of Memorial Sloan Kettering Cancer Center, New York, and her associates (JACC: Cardiovasc Imaging. 2018 Aug;11[8]:1094-7). Most of the commentary authors report various disclosures.
While trastuzumab clearly benefits patients with HER2-positive breast cancer at various stages of progression, concerns about heart failure persist. Studies have suggested that the drug doesn’t boost the risk of late cardiac events, but it’s not clear if this is due to mandated screening in these trials. The new study provides more evidence that adherence to screening guidelines is limited, and recent trials offer evidence that the general cardiac risk may be overblown. Future studies could be designed to offer insight into the wisdom of adjusting screening regimens based on stratification of risk. A meta-analysis could also be helpful, and the upcoming results of the SAFE-HEART study will provide information about the safety of anti-HER2 antibody therapy in patients with low but asymptomatic left-ventricular ejection fraction.
These comments are excerpted from a commentary by Chau T. Dang, MD, of Memorial Sloan Kettering Cancer Center, New York, and her associates (JACC: Cardiovasc Imaging. 2018 Aug;11[8]:1094-7). Most of the commentary authors report various disclosures.
Adding more evidence to an ongoing debate, a large new study suggests that patients with breast cancer who take trastuzumab (Herceptin) may face double the adjusted risk of developing heart failure, with older women at highest risk.
The study also found that most patients who took trastuzumab didn’t receive recommended cardiac screening.
The researchers said their findings are unique because they tracked both younger and older patients. “By examining the rates of both cardiac monitoring and cardiotoxicity, we could begin to address the controversial issue of whether cardiac monitoring is warranted in young breast cancer patients,” wrote Mariana Chavez-MacGregor, MD, MSC, of the University of Texas MD Anderson Cancer Center, and her associates. The report was published in JACC: Cardiovascular Imaging.
While Trastuzumab has boosted breast cancer survival rates for patients with HER2-positive tumors, it’s also raised concerns about cardiotoxicity that could be an indicator of subsequent congestive heart failure (Cochrane Database Syst Rev. 2014 Jun 12;(6):CD006242).
According to the new study, the risk of trastuzumab risk is linked to damage to cardiac myocytes that can cause reversible cardiotoxicity.
The prescribing information for trastuzumab advises patients to undergo cardiac monitoring before treatment with trastuzumab and every 3 months during treatment. Recommendations by medical organizations have varied.
Now, as a 2016 report put it, it’s “increasingly unclear” whether frequent routine monitoring is appropriate for all patients (J Clin Oncol. 2016 Apr 1;34[10]:1030-3).
For the current study, Dr. Chavez-MacGregor and her associates identified 16,456 adult women in the United States who were diagnosed with nonmetastatic invasive breast cancer from 2009 to 2014. Researchers tracked the group, with a median age of 56, through as late as 2015.
The women were treated with chemotherapy within 6 months of diagnosis, and 4,325 received trastuzumab.
Of all the subjects, 692 patients (4.2%) developed heart failure following chemotherapy. The rate among patients treated with trastuzumab was higher, at 8.3%, compared with 2.7% for those not treated with trastuzumab (P less than .001).
The researchers also looked at anthracycline users and found that they were slightly more likely to develop HF (4.6% vs. 4.0% among nonusers, P = .048).
Increased age boosted the risk of HF in the trastuzumab-treated patients, and the risk was highest in those treated with both anthracyclines and trastuzumab. Other factors linked to more risk were comorbidities, hypertension, and valve disease.
After adjusting for confounders, the researchers estimated that those treated with trastuzumab were 2.01 times more likely to develop HF (HR, 2.01; 95% confidence interval, 1.72-2.36), and those who took anthracycline were 1.53 times more likely (HR, 1.53; 95% CI, 1.30-1.80)
The researchers also examined medical records for evidence that subjects underwent cardiac screening at least once every 4 months, not 3 months, as the prescribing information recommends. The study team chose to focus on 4-month intervals “to compensate for differences in scheduling, resources, or levels of accessibility to medical care.”
Medical records suggest that 73.5% of patients who took trastuzumab underwent cardiac screening at the beginning of therapy, but only 46.2% continued to do so at least every 4 months.
An adjusted model linked more screening to the use of anthracyclines and taxanes, radiation treatment, and living in the Northeast vs. the West.
“HF was more frequently identified among patients undergoing recommended cardiac monitoring (10.4% compared with 6.5%, respectively; P less than.001), suggesting that, as more patients are screened, more patients are likely to be found having HF,” the researchers reported.
However, they added that “our sensitivity analysis using inpatient claims allowed us to determine that the HF identified using cardiac monitoring was not severe enough to require hospitalization and was likely asymptomatic. The clinical implications of the diagnosis of asymptomatic HF are hard to determine and are beyond the scope of this study.”
The researchers also noted that the findings suggest that screening has become more common in recent years.
“The number of cancer survivors is expected to increase over time, and we will continue to see patients develop treatment-related cardiotoxicity,” the researchers wrote. “Thus, more research, evidence-based guidelines, and tools for prediction of cancer treatment–related cardiotoxicity are needed.”
The National Cancer Institute and Cancer Prevention and Research Institute of Texas funded the study. Two study authors reported grant funding from the Susan G. Komen Breast Cancer Foundation, and one reports consulting for Pfizer and Roche. The other authors reported no disclosures.
SOURCE: Chavez-MacGregor M et al. JACC: Cardiovasc Imaging. 2018 Aug;11[8]1084-93.
Adding more evidence to an ongoing debate, a large new study suggests that patients with breast cancer who take trastuzumab (Herceptin) may face double the adjusted risk of developing heart failure, with older women at highest risk.
The study also found that most patients who took trastuzumab didn’t receive recommended cardiac screening.
The researchers said their findings are unique because they tracked both younger and older patients. “By examining the rates of both cardiac monitoring and cardiotoxicity, we could begin to address the controversial issue of whether cardiac monitoring is warranted in young breast cancer patients,” wrote Mariana Chavez-MacGregor, MD, MSC, of the University of Texas MD Anderson Cancer Center, and her associates. The report was published in JACC: Cardiovascular Imaging.
While Trastuzumab has boosted breast cancer survival rates for patients with HER2-positive tumors, it’s also raised concerns about cardiotoxicity that could be an indicator of subsequent congestive heart failure (Cochrane Database Syst Rev. 2014 Jun 12;(6):CD006242).
According to the new study, the risk of trastuzumab risk is linked to damage to cardiac myocytes that can cause reversible cardiotoxicity.
The prescribing information for trastuzumab advises patients to undergo cardiac monitoring before treatment with trastuzumab and every 3 months during treatment. Recommendations by medical organizations have varied.
Now, as a 2016 report put it, it’s “increasingly unclear” whether frequent routine monitoring is appropriate for all patients (J Clin Oncol. 2016 Apr 1;34[10]:1030-3).
For the current study, Dr. Chavez-MacGregor and her associates identified 16,456 adult women in the United States who were diagnosed with nonmetastatic invasive breast cancer from 2009 to 2014. Researchers tracked the group, with a median age of 56, through as late as 2015.
The women were treated with chemotherapy within 6 months of diagnosis, and 4,325 received trastuzumab.
Of all the subjects, 692 patients (4.2%) developed heart failure following chemotherapy. The rate among patients treated with trastuzumab was higher, at 8.3%, compared with 2.7% for those not treated with trastuzumab (P less than .001).
The researchers also looked at anthracycline users and found that they were slightly more likely to develop HF (4.6% vs. 4.0% among nonusers, P = .048).
Increased age boosted the risk of HF in the trastuzumab-treated patients, and the risk was highest in those treated with both anthracyclines and trastuzumab. Other factors linked to more risk were comorbidities, hypertension, and valve disease.
After adjusting for confounders, the researchers estimated that those treated with trastuzumab were 2.01 times more likely to develop HF (HR, 2.01; 95% confidence interval, 1.72-2.36), and those who took anthracycline were 1.53 times more likely (HR, 1.53; 95% CI, 1.30-1.80)
The researchers also examined medical records for evidence that subjects underwent cardiac screening at least once every 4 months, not 3 months, as the prescribing information recommends. The study team chose to focus on 4-month intervals “to compensate for differences in scheduling, resources, or levels of accessibility to medical care.”
Medical records suggest that 73.5% of patients who took trastuzumab underwent cardiac screening at the beginning of therapy, but only 46.2% continued to do so at least every 4 months.
An adjusted model linked more screening to the use of anthracyclines and taxanes, radiation treatment, and living in the Northeast vs. the West.
“HF was more frequently identified among patients undergoing recommended cardiac monitoring (10.4% compared with 6.5%, respectively; P less than.001), suggesting that, as more patients are screened, more patients are likely to be found having HF,” the researchers reported.
However, they added that “our sensitivity analysis using inpatient claims allowed us to determine that the HF identified using cardiac monitoring was not severe enough to require hospitalization and was likely asymptomatic. The clinical implications of the diagnosis of asymptomatic HF are hard to determine and are beyond the scope of this study.”
The researchers also noted that the findings suggest that screening has become more common in recent years.
“The number of cancer survivors is expected to increase over time, and we will continue to see patients develop treatment-related cardiotoxicity,” the researchers wrote. “Thus, more research, evidence-based guidelines, and tools for prediction of cancer treatment–related cardiotoxicity are needed.”
The National Cancer Institute and Cancer Prevention and Research Institute of Texas funded the study. Two study authors reported grant funding from the Susan G. Komen Breast Cancer Foundation, and one reports consulting for Pfizer and Roche. The other authors reported no disclosures.
SOURCE: Chavez-MacGregor M et al. JACC: Cardiovasc Imaging. 2018 Aug;11[8]1084-93.
FROM JACC: CARDIOVASCULAR IMAGING
Key clinical point: Women with breast cancer who take trastuzumab (Herceptin) may face double the adjusted risk of heart failure, but most aren’t screened frequently.
Major finding: Patients who took trastuzumab were 2.01 times more likely to develop HF (HR, 95% CI, 1.72-2.36) than were those who didn’t. Of all patients who took the drug, fewer than half received recommended frequency of screening.
Study details: Analysis of 16,456 U.S. adult women with nonmetastatic breast cancer diagnosed from 2009 to 2014 and tracked through 2015. Of those, 4.2% developed HF.
Disclosures: The National Cancer Institute and Cancer Prevention and Research Institute of Texas funded the study. Two study authors report grant funding from the Susan G. Komen Breast Cancer Foundation, and one reports consulting for Pfizer and Roche. The other authors report no disclosures.
Source: Chavez-MacGregor M et al. JACC: Cardiovasc Imaging 2018 Aug;11[8]1084-93.
Study supports meningococcal B vaccine in children with rare diseases
A new study, the first of its kind, provided support for guidelines suggesting that the capsular meningococcal B vaccine be given to children with three rare conditions that boost infection risk.
For children with terminal chain complement deficiencies or who are undergoing treatment with eculizumab, “it is important that these patients are identified, receive education about sepsis management plans, and are prescribed prophylactic antibiotics according to local guidelines, along with vaccination, to provide every chance for them to be protected against this deadly disease,” the researchers wrote in Pediatrics.
While some countries suggest that the vaccine be given to all healthy infants, U.S. guidelines advise that the vaccine be given to preteenagers, teenagers, and adults who are considered at special risk. These include those with terminal chain complement deficiencies, who are believed to be up to 10,000 times more likely than healthy children to develop invasive meningococcal disease, and those who take eculizumab (Soliris). The risk groups recommended for vaccinations also include those with asplenia and splenic dysfunction, although their excess risk, if any, is unknown.
The new study of the capsular group meningococcal B vaccine, led by Federico Martinón-Torres, PhD, of the Hospital Clinico Universitario de Santiago de Compostela, Spain, adds to previous research that confirmed the effectiveness of vaccinating complement-deficient patients with capsular group A, C, W, and Y meningococcal vaccines.
For the open-label, phase 3b study, researchers in Italy, Spain, Poland, and Russia gave two doses of the vaccine 2 months apart to 239 children aged 2-17 years with an average age of 10 years. Nearly all were white, and 45% were female.
A total of 40 children had complement deficiency, 112 had asplenia or splenic dysfunction, and 87 children in the control group also received the vaccine.
Following vaccination, the percentages of children with exogenous complement serum bactericidal activity titers greater than or equal to 1:5 to the four test strains were similar in the healthy children and those with asplenia/splenic dysfunction. “It is reasonable to expect that this vaccine will be as effective in children with asplenia or splenic deficiency as in children in the control category,” the researchers wrote.
However, these levels were lower in the complement-deficient children, particularly in those with terminal chain complement deficiency and those who took eculizumab.
The proportions of children with exogenous complement serum bactericidal activity titers greater than or equal to 1:5 against the four test strains were 87% (H44/76), 95% (5/99), 68% (NZ98/254), and 73% (M10713) in complement-deficient children, compared with 98%, 99%, 83%, and 99%, respectively, in the healthy controls.
“Ongoing surveillance for vaccine failures is required to determine the significance of the trend to reduced immune response in children with terminal chain complement deficiencies or undergoing treatment with eculizumab,” the researchers wrote.
Eculizumab’s manufacturer has noted the risk of serious meningococcal infections and warned physicians to “immunize patients with meningococcal vaccines at least 2 weeks prior to administering the first dose of Soliris, unless the risks of delaying Soliris therapy outweigh the risk of developing a meningococcal infection,” according to the website.
The study was funded by Novartis Vaccines and Diagnostics (now GlaxoSmithKline Biologicals). Some of the study authors reported various disclosures, including financial relationships with Novartis and GlaxoSmithKline outside the submitted work. Dr. Kaplan reported no relevant financial disclosures.
SOURCE: Martinón-Torres F et al. Pediatrics. 2018 Aug 1. doi: 10.1542/peds.2017-4250.
The Centers for Disease Control and Prevention reported an annual average of 792 cases of meningococcal disease and 98 deaths in the United States from 2006 to 2015 with serotype B isolates causing the highest numbers of cases. In a recent development, two vaccines against this strain have become available in the United States in the past 3 years for people aged 10-25 years. But officials don’t recommend their routine use, instead, guidelines suggest they be given to those at high risk only.
There’s a gap in knowledge because vaccine researchers didn’t include people with complement deficiency (either congenital or related to eculizumab), asplenia, or splenic dysfunction in studies that led to approval. Now, the new study offers reassuring findings regarding the latter two conditions, as bactericidal antibody responses were equal to those in healthy controls.
The findings regarding complement deficiency aren’t surprising, and suggest that vaccine strength in children with the condition only reached the levels in healthy children when an exogenous complement was added.
The study supports guidelines suggesting antibiotic prophylaxis in patients receiving eculizumab even if they already underwent meningococcal vaccination. It’s not clear if this approach also will be effective in those with congenital complement deficiencies (except for complement component 6 deficiency).
It is hoped that surveillance studies will show that use of serogroup B vaccines will prevent invasive meningococcal infections in these high-risk populations for which they are recommended.
Sheldon L. Kaplan, MD, is a pediatrician at Baylor College of Medicine and Texas Children’s Hospital, both in Houston. These comments are summarized from an editorial accompanying the article by Martinón-Torres et al. (Pediatrics. 2018 Aug 1. doi: 10.1542/peds.2018-0554).
The Centers for Disease Control and Prevention reported an annual average of 792 cases of meningococcal disease and 98 deaths in the United States from 2006 to 2015 with serotype B isolates causing the highest numbers of cases. In a recent development, two vaccines against this strain have become available in the United States in the past 3 years for people aged 10-25 years. But officials don’t recommend their routine use, instead, guidelines suggest they be given to those at high risk only.
There’s a gap in knowledge because vaccine researchers didn’t include people with complement deficiency (either congenital or related to eculizumab), asplenia, or splenic dysfunction in studies that led to approval. Now, the new study offers reassuring findings regarding the latter two conditions, as bactericidal antibody responses were equal to those in healthy controls.
The findings regarding complement deficiency aren’t surprising, and suggest that vaccine strength in children with the condition only reached the levels in healthy children when an exogenous complement was added.
The study supports guidelines suggesting antibiotic prophylaxis in patients receiving eculizumab even if they already underwent meningococcal vaccination. It’s not clear if this approach also will be effective in those with congenital complement deficiencies (except for complement component 6 deficiency).
It is hoped that surveillance studies will show that use of serogroup B vaccines will prevent invasive meningococcal infections in these high-risk populations for which they are recommended.
Sheldon L. Kaplan, MD, is a pediatrician at Baylor College of Medicine and Texas Children’s Hospital, both in Houston. These comments are summarized from an editorial accompanying the article by Martinón-Torres et al. (Pediatrics. 2018 Aug 1. doi: 10.1542/peds.2018-0554).
The Centers for Disease Control and Prevention reported an annual average of 792 cases of meningococcal disease and 98 deaths in the United States from 2006 to 2015 with serotype B isolates causing the highest numbers of cases. In a recent development, two vaccines against this strain have become available in the United States in the past 3 years for people aged 10-25 years. But officials don’t recommend their routine use, instead, guidelines suggest they be given to those at high risk only.
There’s a gap in knowledge because vaccine researchers didn’t include people with complement deficiency (either congenital or related to eculizumab), asplenia, or splenic dysfunction in studies that led to approval. Now, the new study offers reassuring findings regarding the latter two conditions, as bactericidal antibody responses were equal to those in healthy controls.
The findings regarding complement deficiency aren’t surprising, and suggest that vaccine strength in children with the condition only reached the levels in healthy children when an exogenous complement was added.
The study supports guidelines suggesting antibiotic prophylaxis in patients receiving eculizumab even if they already underwent meningococcal vaccination. It’s not clear if this approach also will be effective in those with congenital complement deficiencies (except for complement component 6 deficiency).
It is hoped that surveillance studies will show that use of serogroup B vaccines will prevent invasive meningococcal infections in these high-risk populations for which they are recommended.
Sheldon L. Kaplan, MD, is a pediatrician at Baylor College of Medicine and Texas Children’s Hospital, both in Houston. These comments are summarized from an editorial accompanying the article by Martinón-Torres et al. (Pediatrics. 2018 Aug 1. doi: 10.1542/peds.2018-0554).
A new study, the first of its kind, provided support for guidelines suggesting that the capsular meningococcal B vaccine be given to children with three rare conditions that boost infection risk.
For children with terminal chain complement deficiencies or who are undergoing treatment with eculizumab, “it is important that these patients are identified, receive education about sepsis management plans, and are prescribed prophylactic antibiotics according to local guidelines, along with vaccination, to provide every chance for them to be protected against this deadly disease,” the researchers wrote in Pediatrics.
While some countries suggest that the vaccine be given to all healthy infants, U.S. guidelines advise that the vaccine be given to preteenagers, teenagers, and adults who are considered at special risk. These include those with terminal chain complement deficiencies, who are believed to be up to 10,000 times more likely than healthy children to develop invasive meningococcal disease, and those who take eculizumab (Soliris). The risk groups recommended for vaccinations also include those with asplenia and splenic dysfunction, although their excess risk, if any, is unknown.
The new study of the capsular group meningococcal B vaccine, led by Federico Martinón-Torres, PhD, of the Hospital Clinico Universitario de Santiago de Compostela, Spain, adds to previous research that confirmed the effectiveness of vaccinating complement-deficient patients with capsular group A, C, W, and Y meningococcal vaccines.
For the open-label, phase 3b study, researchers in Italy, Spain, Poland, and Russia gave two doses of the vaccine 2 months apart to 239 children aged 2-17 years with an average age of 10 years. Nearly all were white, and 45% were female.
A total of 40 children had complement deficiency, 112 had asplenia or splenic dysfunction, and 87 children in the control group also received the vaccine.
Following vaccination, the percentages of children with exogenous complement serum bactericidal activity titers greater than or equal to 1:5 to the four test strains were similar in the healthy children and those with asplenia/splenic dysfunction. “It is reasonable to expect that this vaccine will be as effective in children with asplenia or splenic deficiency as in children in the control category,” the researchers wrote.
However, these levels were lower in the complement-deficient children, particularly in those with terminal chain complement deficiency and those who took eculizumab.
The proportions of children with exogenous complement serum bactericidal activity titers greater than or equal to 1:5 against the four test strains were 87% (H44/76), 95% (5/99), 68% (NZ98/254), and 73% (M10713) in complement-deficient children, compared with 98%, 99%, 83%, and 99%, respectively, in the healthy controls.
“Ongoing surveillance for vaccine failures is required to determine the significance of the trend to reduced immune response in children with terminal chain complement deficiencies or undergoing treatment with eculizumab,” the researchers wrote.
Eculizumab’s manufacturer has noted the risk of serious meningococcal infections and warned physicians to “immunize patients with meningococcal vaccines at least 2 weeks prior to administering the first dose of Soliris, unless the risks of delaying Soliris therapy outweigh the risk of developing a meningococcal infection,” according to the website.
The study was funded by Novartis Vaccines and Diagnostics (now GlaxoSmithKline Biologicals). Some of the study authors reported various disclosures, including financial relationships with Novartis and GlaxoSmithKline outside the submitted work. Dr. Kaplan reported no relevant financial disclosures.
SOURCE: Martinón-Torres F et al. Pediatrics. 2018 Aug 1. doi: 10.1542/peds.2017-4250.
A new study, the first of its kind, provided support for guidelines suggesting that the capsular meningococcal B vaccine be given to children with three rare conditions that boost infection risk.
For children with terminal chain complement deficiencies or who are undergoing treatment with eculizumab, “it is important that these patients are identified, receive education about sepsis management plans, and are prescribed prophylactic antibiotics according to local guidelines, along with vaccination, to provide every chance for them to be protected against this deadly disease,” the researchers wrote in Pediatrics.
While some countries suggest that the vaccine be given to all healthy infants, U.S. guidelines advise that the vaccine be given to preteenagers, teenagers, and adults who are considered at special risk. These include those with terminal chain complement deficiencies, who are believed to be up to 10,000 times more likely than healthy children to develop invasive meningococcal disease, and those who take eculizumab (Soliris). The risk groups recommended for vaccinations also include those with asplenia and splenic dysfunction, although their excess risk, if any, is unknown.
The new study of the capsular group meningococcal B vaccine, led by Federico Martinón-Torres, PhD, of the Hospital Clinico Universitario de Santiago de Compostela, Spain, adds to previous research that confirmed the effectiveness of vaccinating complement-deficient patients with capsular group A, C, W, and Y meningococcal vaccines.
For the open-label, phase 3b study, researchers in Italy, Spain, Poland, and Russia gave two doses of the vaccine 2 months apart to 239 children aged 2-17 years with an average age of 10 years. Nearly all were white, and 45% were female.
A total of 40 children had complement deficiency, 112 had asplenia or splenic dysfunction, and 87 children in the control group also received the vaccine.
Following vaccination, the percentages of children with exogenous complement serum bactericidal activity titers greater than or equal to 1:5 to the four test strains were similar in the healthy children and those with asplenia/splenic dysfunction. “It is reasonable to expect that this vaccine will be as effective in children with asplenia or splenic deficiency as in children in the control category,” the researchers wrote.
However, these levels were lower in the complement-deficient children, particularly in those with terminal chain complement deficiency and those who took eculizumab.
The proportions of children with exogenous complement serum bactericidal activity titers greater than or equal to 1:5 against the four test strains were 87% (H44/76), 95% (5/99), 68% (NZ98/254), and 73% (M10713) in complement-deficient children, compared with 98%, 99%, 83%, and 99%, respectively, in the healthy controls.
“Ongoing surveillance for vaccine failures is required to determine the significance of the trend to reduced immune response in children with terminal chain complement deficiencies or undergoing treatment with eculizumab,” the researchers wrote.
Eculizumab’s manufacturer has noted the risk of serious meningococcal infections and warned physicians to “immunize patients with meningococcal vaccines at least 2 weeks prior to administering the first dose of Soliris, unless the risks of delaying Soliris therapy outweigh the risk of developing a meningococcal infection,” according to the website.
The study was funded by Novartis Vaccines and Diagnostics (now GlaxoSmithKline Biologicals). Some of the study authors reported various disclosures, including financial relationships with Novartis and GlaxoSmithKline outside the submitted work. Dr. Kaplan reported no relevant financial disclosures.
SOURCE: Martinón-Torres F et al. Pediatrics. 2018 Aug 1. doi: 10.1542/peds.2017-4250.
FROM PEDIATRICS
Key clinical point: The meningococcal B vaccine retained its strength in kids with two rare immunosuppressive diseases, but may be weaker in a third group.
Major finding: The vaccine’s effectiveness was roughly the same in healthy controls and in those with asplenia and splenic dysfunction, but it dipped in those with complement deficiency.
Study details: An open-label, multicenter analysis of children aged 2-17 years who received two doses over 2 months.
Disclosures: The study was funded by Novartis Vaccines and Diagnostics (now GlaxoSmithKline Biologicals). Some of the study authors reported various disclosures, including financial relationships with Novartis and GlaxoSmithKline outside the submitted work. Dr. Kaplan reported no relevant financial disclosures.
Source: Martinón-Torres F et al. Pediatrics. 2018 Aug 1. doi: 10.1542/peds.2017-4250.
Questions abound about availability, legality of new cannabis-derived epilepsy drug
The first-ever federal approval of a marijuana-derived drug, Epidiolex (cannabidiol), comes with a cloud of complications.
Epidiolex is an oral solution of purified cannabidiol (CBD), a component of the marijuana plant that does not make people high.
According to the results of a phase 3 trial in patients with Lennox-Gastaut syndrome published earlier this year, median monthly drop seizures fell by 43.9% in a Epidiolex group compared with 21.8% in a placebo group (Lancet. 2018;391[10125]:1085-96). A 2017 study found that patients with Dravet syndrome who took the drug had fewer median convulsive seizures per month, dropping from 12.4 to 5.9, while there was barely a difference for the placebo group (N Engl J Med. 2017;376:2011-20). In both trials, patients received Epidiolex as an add-on treatment.
“There definitely are side effects such as sleepiness, abnormal liver function values, and other things that people will have to watch out for, such as interactions with other drugs,” Jacqueline A. French, MD, professor of neurology at New York University and chief scientific officer of the Epilepsy Foundation, said in an interview. “But it will definitely be a great benefit to some and a benefit to many.”
It’s not clear how the drug works, she said.
Greenwich Biosciences, the U.S. subsidiary of British drug maker GW Pharmaceuticals, plans to market Epidiolex this fall, spokesman Steve Schultz said in an interview. First, however, the U.S. Drug Enforcement Agency must reschedule the drug so it doesn’t fall under federal antimarijuana laws. It has 90 days after the FDA approval to do so, and its rescheduling is expected.
Then the focus will turn to the states, which have a crazy quilt of laws about marijuana and its medical use. “We’ve worked with the state legislators to enact laws or to modify laws to allow for our medicine to be made available,” Mr. Schultz said. “There may be one or two that we’re still working on.”
At issue: States and the District of Columbia have a wide variety of laws that appear to affect the sale of Epidiolex.
According to the website procon.org, 30 states and the District of Columbia allow medical marijuana, although the laws vary regarding what is allowed and do not address cannabis-derived pharmaceuticals.
Another 17 states have laws that address CBD specifically, often with language that allows its use to treat epilepsy in all or specific cases, according to procon.org.
In Oklahoma, an anticannabis law excludes “from the definition of marijuana ‘any federal Food and Drug Administration–approved cannabidiol drug or substance.’ ”
North Carolina attorney Rod Kight, who represents businesses in the cannabis industry, predicts that “once the federal government [via the DEA] changes the law, the states will fall in line. The only thing that would prevent them would be a lack of understanding of what’s going on.”
Meanwhile, many sellers of CBD-based products continue to promote their use as treatments for epilepsy and many other conditions.
Legal exceptions made by states for FDA-approved CBD-based drugs could affect access to unapproved CBD-based products. It’s also possible that Epidiolex could hurt sellers of cannabis products in other ways. “They’ll now have to compete with a drug made to a specific strength and purity, and that’s probably going to have some impact on their business,” William J. McNichol, adjunct professor at Rutgers Law School, Camden, N.J., said in an interview. “If you have a choice, are you going to choose aspirin I made myself in my artisan aspirin factory that the FDA never saw? Or aspirin made by Bayer?”
What’s next? For one, the FDA’s approval of Epidiolex seems likely to open the door for more cannabis-based medications. “The FDA is quite open to evaluating cannabinoid-based medicines as long as they go through their process,” said Mr. Schultz, the Greenwich Biosciences spokesman.
As for the legal front, Mr. Kight said potential changes in federal law could expand the legality of cannabis-based products by allowing them when they’re derived from “industrial hemp.” This could mean that patients with epilepsy will have more legal ways to buy CBD products.
Regardless of the legal situation, Mr. Kight said, “a physician can explain to patients across the board about how CBD might benefit them. The physician can say there are extract products that are out there and available over the counter.”
As for the maker of Epidiolex, GW Pharmaceuticals is recruiting for a phase 3 trial of a cannabinoid treatment for tuberous sclerosis complex, Mr. Schultz said. The results are due in the first half of 2019.
The company is also conducting cannabinoid research in the areas of autism and Rett syndrome, Mr. Schultz said, adding: “We also have done work in schizophrenia and in oncology and glioblastoma.”
Mr. Kight disclosed that he represents companies in the cannabis industry. Dr. French is president of the Epilepsy Study Consortium and disclosed working with multiple drug makers in the epilepsy field, including GW Pharmaceuticals. Dr. French receives fixed compensation by the consortium and is not paid by any pharmaceutical company. Mr. McNichol reported no relevant disclosures.
The first-ever federal approval of a marijuana-derived drug, Epidiolex (cannabidiol), comes with a cloud of complications.
Epidiolex is an oral solution of purified cannabidiol (CBD), a component of the marijuana plant that does not make people high.
According to the results of a phase 3 trial in patients with Lennox-Gastaut syndrome published earlier this year, median monthly drop seizures fell by 43.9% in a Epidiolex group compared with 21.8% in a placebo group (Lancet. 2018;391[10125]:1085-96). A 2017 study found that patients with Dravet syndrome who took the drug had fewer median convulsive seizures per month, dropping from 12.4 to 5.9, while there was barely a difference for the placebo group (N Engl J Med. 2017;376:2011-20). In both trials, patients received Epidiolex as an add-on treatment.
“There definitely are side effects such as sleepiness, abnormal liver function values, and other things that people will have to watch out for, such as interactions with other drugs,” Jacqueline A. French, MD, professor of neurology at New York University and chief scientific officer of the Epilepsy Foundation, said in an interview. “But it will definitely be a great benefit to some and a benefit to many.”
It’s not clear how the drug works, she said.
Greenwich Biosciences, the U.S. subsidiary of British drug maker GW Pharmaceuticals, plans to market Epidiolex this fall, spokesman Steve Schultz said in an interview. First, however, the U.S. Drug Enforcement Agency must reschedule the drug so it doesn’t fall under federal antimarijuana laws. It has 90 days after the FDA approval to do so, and its rescheduling is expected.
Then the focus will turn to the states, which have a crazy quilt of laws about marijuana and its medical use. “We’ve worked with the state legislators to enact laws or to modify laws to allow for our medicine to be made available,” Mr. Schultz said. “There may be one or two that we’re still working on.”
At issue: States and the District of Columbia have a wide variety of laws that appear to affect the sale of Epidiolex.
According to the website procon.org, 30 states and the District of Columbia allow medical marijuana, although the laws vary regarding what is allowed and do not address cannabis-derived pharmaceuticals.
Another 17 states have laws that address CBD specifically, often with language that allows its use to treat epilepsy in all or specific cases, according to procon.org.
In Oklahoma, an anticannabis law excludes “from the definition of marijuana ‘any federal Food and Drug Administration–approved cannabidiol drug or substance.’ ”
North Carolina attorney Rod Kight, who represents businesses in the cannabis industry, predicts that “once the federal government [via the DEA] changes the law, the states will fall in line. The only thing that would prevent them would be a lack of understanding of what’s going on.”
Meanwhile, many sellers of CBD-based products continue to promote their use as treatments for epilepsy and many other conditions.
Legal exceptions made by states for FDA-approved CBD-based drugs could affect access to unapproved CBD-based products. It’s also possible that Epidiolex could hurt sellers of cannabis products in other ways. “They’ll now have to compete with a drug made to a specific strength and purity, and that’s probably going to have some impact on their business,” William J. McNichol, adjunct professor at Rutgers Law School, Camden, N.J., said in an interview. “If you have a choice, are you going to choose aspirin I made myself in my artisan aspirin factory that the FDA never saw? Or aspirin made by Bayer?”
What’s next? For one, the FDA’s approval of Epidiolex seems likely to open the door for more cannabis-based medications. “The FDA is quite open to evaluating cannabinoid-based medicines as long as they go through their process,” said Mr. Schultz, the Greenwich Biosciences spokesman.
As for the legal front, Mr. Kight said potential changes in federal law could expand the legality of cannabis-based products by allowing them when they’re derived from “industrial hemp.” This could mean that patients with epilepsy will have more legal ways to buy CBD products.
Regardless of the legal situation, Mr. Kight said, “a physician can explain to patients across the board about how CBD might benefit them. The physician can say there are extract products that are out there and available over the counter.”
As for the maker of Epidiolex, GW Pharmaceuticals is recruiting for a phase 3 trial of a cannabinoid treatment for tuberous sclerosis complex, Mr. Schultz said. The results are due in the first half of 2019.
The company is also conducting cannabinoid research in the areas of autism and Rett syndrome, Mr. Schultz said, adding: “We also have done work in schizophrenia and in oncology and glioblastoma.”
Mr. Kight disclosed that he represents companies in the cannabis industry. Dr. French is president of the Epilepsy Study Consortium and disclosed working with multiple drug makers in the epilepsy field, including GW Pharmaceuticals. Dr. French receives fixed compensation by the consortium and is not paid by any pharmaceutical company. Mr. McNichol reported no relevant disclosures.
The first-ever federal approval of a marijuana-derived drug, Epidiolex (cannabidiol), comes with a cloud of complications.
Epidiolex is an oral solution of purified cannabidiol (CBD), a component of the marijuana plant that does not make people high.
According to the results of a phase 3 trial in patients with Lennox-Gastaut syndrome published earlier this year, median monthly drop seizures fell by 43.9% in a Epidiolex group compared with 21.8% in a placebo group (Lancet. 2018;391[10125]:1085-96). A 2017 study found that patients with Dravet syndrome who took the drug had fewer median convulsive seizures per month, dropping from 12.4 to 5.9, while there was barely a difference for the placebo group (N Engl J Med. 2017;376:2011-20). In both trials, patients received Epidiolex as an add-on treatment.
“There definitely are side effects such as sleepiness, abnormal liver function values, and other things that people will have to watch out for, such as interactions with other drugs,” Jacqueline A. French, MD, professor of neurology at New York University and chief scientific officer of the Epilepsy Foundation, said in an interview. “But it will definitely be a great benefit to some and a benefit to many.”
It’s not clear how the drug works, she said.
Greenwich Biosciences, the U.S. subsidiary of British drug maker GW Pharmaceuticals, plans to market Epidiolex this fall, spokesman Steve Schultz said in an interview. First, however, the U.S. Drug Enforcement Agency must reschedule the drug so it doesn’t fall under federal antimarijuana laws. It has 90 days after the FDA approval to do so, and its rescheduling is expected.
Then the focus will turn to the states, which have a crazy quilt of laws about marijuana and its medical use. “We’ve worked with the state legislators to enact laws or to modify laws to allow for our medicine to be made available,” Mr. Schultz said. “There may be one or two that we’re still working on.”
At issue: States and the District of Columbia have a wide variety of laws that appear to affect the sale of Epidiolex.
According to the website procon.org, 30 states and the District of Columbia allow medical marijuana, although the laws vary regarding what is allowed and do not address cannabis-derived pharmaceuticals.
Another 17 states have laws that address CBD specifically, often with language that allows its use to treat epilepsy in all or specific cases, according to procon.org.
In Oklahoma, an anticannabis law excludes “from the definition of marijuana ‘any federal Food and Drug Administration–approved cannabidiol drug or substance.’ ”
North Carolina attorney Rod Kight, who represents businesses in the cannabis industry, predicts that “once the federal government [via the DEA] changes the law, the states will fall in line. The only thing that would prevent them would be a lack of understanding of what’s going on.”
Meanwhile, many sellers of CBD-based products continue to promote their use as treatments for epilepsy and many other conditions.
Legal exceptions made by states for FDA-approved CBD-based drugs could affect access to unapproved CBD-based products. It’s also possible that Epidiolex could hurt sellers of cannabis products in other ways. “They’ll now have to compete with a drug made to a specific strength and purity, and that’s probably going to have some impact on their business,” William J. McNichol, adjunct professor at Rutgers Law School, Camden, N.J., said in an interview. “If you have a choice, are you going to choose aspirin I made myself in my artisan aspirin factory that the FDA never saw? Or aspirin made by Bayer?”
What’s next? For one, the FDA’s approval of Epidiolex seems likely to open the door for more cannabis-based medications. “The FDA is quite open to evaluating cannabinoid-based medicines as long as they go through their process,” said Mr. Schultz, the Greenwich Biosciences spokesman.
As for the legal front, Mr. Kight said potential changes in federal law could expand the legality of cannabis-based products by allowing them when they’re derived from “industrial hemp.” This could mean that patients with epilepsy will have more legal ways to buy CBD products.
Regardless of the legal situation, Mr. Kight said, “a physician can explain to patients across the board about how CBD might benefit them. The physician can say there are extract products that are out there and available over the counter.”
As for the maker of Epidiolex, GW Pharmaceuticals is recruiting for a phase 3 trial of a cannabinoid treatment for tuberous sclerosis complex, Mr. Schultz said. The results are due in the first half of 2019.
The company is also conducting cannabinoid research in the areas of autism and Rett syndrome, Mr. Schultz said, adding: “We also have done work in schizophrenia and in oncology and glioblastoma.”
Mr. Kight disclosed that he represents companies in the cannabis industry. Dr. French is president of the Epilepsy Study Consortium and disclosed working with multiple drug makers in the epilepsy field, including GW Pharmaceuticals. Dr. French receives fixed compensation by the consortium and is not paid by any pharmaceutical company. Mr. McNichol reported no relevant disclosures.
Omega-3 heart benefit: Just another fish tale?
There’s more evidence that the vaunted reputation of fish oil is all wet: An updated Cochrane Library review finds that boosting two components of omega-3 fats – mainly through supplements such as fish oil – has little or no impact on cardiac health or overall mortality.
“Although EPA and DHA reduce triglycerides, supplementary omega-3 fats are probably not useful for preventing or treating heart and circulatory diseases,” the researchers wrote, referring to eicosapentaenoic acid and docosahexaenoic acid, two sources of omega-3 fats in fish.
They did find signs that boosting intake of plant-based alpha-linolenic acid (ALA) “may be slightly protective for some heart and circulatory diseases.” However, they deemed the evidence to be of low quality compared with the high- to moderate-quality evidence supporting the conclusions regarding omega-3 fats from fish.
The review, led by Asmaa S. Abdelhamid, MBBCH, MSc, MD, of the University of East Anglia, was commissioned by the World Health Organization and published on July 18, 2018, in Cochrane Database of Systematic Reviews (doi: 10.1002/14651858.CD003177.pub3). It updates a 2004 Cochrane Library review.
As the review notes, “there is a great deal of public belief in the cardiovascular benefits of omega-3 fats.” But research in recent years has questioned the benefits of fish oil supplementation.
For the new review, the researchers focused on studies that boosted omega-3 fat intake using EPA, DHA and ALA.
The researchers included 79 randomized controlled trials with a total of more than 112,000 adult participants. The studies examined the cardiac and circulatory system effects of greater omega-3 consumption for at least a year compared with controls. Most trials tested supplementation with capsules, but some relied on boosting intake of foods or simply making dietary recommendations.
The studies were conducted in North America, Europe, Australia, and Asia. Researchers determined that 25 trials were designed well enough to be deemed “very trustworthy.”
According to the review’s meta-analysis, increasing intake of long-chain omega-3 fatty acids (EPA and DHA) had little to no effect on all-cause mortality (relative risk, 0.98; 39 trials), cardiovascular mortality (RR, 0.95; 25 trials), cardiovascular events (RR, 0.99; 38 trials), coronary heart disease mortality (RR, 0.93; 21 trials) and arrhythmia (RR, 0.97; 28 trials), while there was a potential boost in stroke risk (RR, 1.06; 28 trials).
The investigators noted that the relative risk of coronary heart disease events fell (RR, 0.93; 28 trials), but this did not hold up after sensitivity analysis. All these trials were deemed of moderate or high quality.
As for increased ALA intake, the researchers found little or no effect on all-cause mortality (RR, 1.01; four trials) and cardiovascular mortality (RR, 0.96; four trials) or coronary heart disease events (RR, 1.00, four trials).
Although the researchers deemed the evidence on ALA to be of low quality, there’s a possible decreased risk of cardiovascular events (RR, 0.95; five trials), coronary heart disease mortality (RR, 0.95; three trials) and arrhythmia (RR, 0.79; one trial). It’s not clear if increased ALA intake affects stroke risk.
“There was no evidence that increasing LCn3 [long-chain omega-3 fatty acids] or ALA altered serious adverse events, adiposity, or lipids, although LCn3 slightly reduced triglycerides and increased HDL. ALA probably reduces HDL (high- or moderate-quality evidence),” the researchers wrote.
Why have some earlier studies suggested a significant effect, enough to help convince millions of Americans to add fish oil pills to their diets? “Previous suggestions of benefits from EPA and DHA supplements appear to spring from trials with higher risk of bias,” the researchers wrote. Robert A. Vogel, MD, a cardiologist at the University of Colorado Denver who is familiar with the review findings, said in an interview that “it’s hard to recommend taking fish oil for heart disease. Even if there’s some effect, it’s got to be a very small, 5%-7% reduction at the best.
“That is not to say that there isn’t an effect on triglycerides,” he added. “[Fish oil] is an accepted way of reducing triglycerides. But you don’t reduce triglycerides to reduce heart disease. There are other risks to high triglycerides, such as pancreatitis.”
Dr. Vogel said he’s never taken fish oil supplements, but he does eat fish 5-6 times a week.
The review was funded by the University of East Anglia, Cochrane Heart Group, and the National Institute for Health Research. The review authors reported no relevant disclosures. Dr. Vogel disclosed consulting for the Pritikin Longevity Center.
SOURCE: Abdelhamid AS et al. Cochrane Database Syst Rev. 2018 Jul 18. doi: 10.1002/14651858.CD003177.pub3.
There’s more evidence that the vaunted reputation of fish oil is all wet: An updated Cochrane Library review finds that boosting two components of omega-3 fats – mainly through supplements such as fish oil – has little or no impact on cardiac health or overall mortality.
“Although EPA and DHA reduce triglycerides, supplementary omega-3 fats are probably not useful for preventing or treating heart and circulatory diseases,” the researchers wrote, referring to eicosapentaenoic acid and docosahexaenoic acid, two sources of omega-3 fats in fish.
They did find signs that boosting intake of plant-based alpha-linolenic acid (ALA) “may be slightly protective for some heart and circulatory diseases.” However, they deemed the evidence to be of low quality compared with the high- to moderate-quality evidence supporting the conclusions regarding omega-3 fats from fish.
The review, led by Asmaa S. Abdelhamid, MBBCH, MSc, MD, of the University of East Anglia, was commissioned by the World Health Organization and published on July 18, 2018, in Cochrane Database of Systematic Reviews (doi: 10.1002/14651858.CD003177.pub3). It updates a 2004 Cochrane Library review.
As the review notes, “there is a great deal of public belief in the cardiovascular benefits of omega-3 fats.” But research in recent years has questioned the benefits of fish oil supplementation.
For the new review, the researchers focused on studies that boosted omega-3 fat intake using EPA, DHA and ALA.
The researchers included 79 randomized controlled trials with a total of more than 112,000 adult participants. The studies examined the cardiac and circulatory system effects of greater omega-3 consumption for at least a year compared with controls. Most trials tested supplementation with capsules, but some relied on boosting intake of foods or simply making dietary recommendations.
The studies were conducted in North America, Europe, Australia, and Asia. Researchers determined that 25 trials were designed well enough to be deemed “very trustworthy.”
According to the review’s meta-analysis, increasing intake of long-chain omega-3 fatty acids (EPA and DHA) had little to no effect on all-cause mortality (relative risk, 0.98; 39 trials), cardiovascular mortality (RR, 0.95; 25 trials), cardiovascular events (RR, 0.99; 38 trials), coronary heart disease mortality (RR, 0.93; 21 trials) and arrhythmia (RR, 0.97; 28 trials), while there was a potential boost in stroke risk (RR, 1.06; 28 trials).
The investigators noted that the relative risk of coronary heart disease events fell (RR, 0.93; 28 trials), but this did not hold up after sensitivity analysis. All these trials were deemed of moderate or high quality.
As for increased ALA intake, the researchers found little or no effect on all-cause mortality (RR, 1.01; four trials) and cardiovascular mortality (RR, 0.96; four trials) or coronary heart disease events (RR, 1.00, four trials).
Although the researchers deemed the evidence on ALA to be of low quality, there’s a possible decreased risk of cardiovascular events (RR, 0.95; five trials), coronary heart disease mortality (RR, 0.95; three trials) and arrhythmia (RR, 0.79; one trial). It’s not clear if increased ALA intake affects stroke risk.
“There was no evidence that increasing LCn3 [long-chain omega-3 fatty acids] or ALA altered serious adverse events, adiposity, or lipids, although LCn3 slightly reduced triglycerides and increased HDL. ALA probably reduces HDL (high- or moderate-quality evidence),” the researchers wrote.
Why have some earlier studies suggested a significant effect, enough to help convince millions of Americans to add fish oil pills to their diets? “Previous suggestions of benefits from EPA and DHA supplements appear to spring from trials with higher risk of bias,” the researchers wrote. Robert A. Vogel, MD, a cardiologist at the University of Colorado Denver who is familiar with the review findings, said in an interview that “it’s hard to recommend taking fish oil for heart disease. Even if there’s some effect, it’s got to be a very small, 5%-7% reduction at the best.
“That is not to say that there isn’t an effect on triglycerides,” he added. “[Fish oil] is an accepted way of reducing triglycerides. But you don’t reduce triglycerides to reduce heart disease. There are other risks to high triglycerides, such as pancreatitis.”
Dr. Vogel said he’s never taken fish oil supplements, but he does eat fish 5-6 times a week.
The review was funded by the University of East Anglia, Cochrane Heart Group, and the National Institute for Health Research. The review authors reported no relevant disclosures. Dr. Vogel disclosed consulting for the Pritikin Longevity Center.
SOURCE: Abdelhamid AS et al. Cochrane Database Syst Rev. 2018 Jul 18. doi: 10.1002/14651858.CD003177.pub3.
There’s more evidence that the vaunted reputation of fish oil is all wet: An updated Cochrane Library review finds that boosting two components of omega-3 fats – mainly through supplements such as fish oil – has little or no impact on cardiac health or overall mortality.
“Although EPA and DHA reduce triglycerides, supplementary omega-3 fats are probably not useful for preventing or treating heart and circulatory diseases,” the researchers wrote, referring to eicosapentaenoic acid and docosahexaenoic acid, two sources of omega-3 fats in fish.
They did find signs that boosting intake of plant-based alpha-linolenic acid (ALA) “may be slightly protective for some heart and circulatory diseases.” However, they deemed the evidence to be of low quality compared with the high- to moderate-quality evidence supporting the conclusions regarding omega-3 fats from fish.
The review, led by Asmaa S. Abdelhamid, MBBCH, MSc, MD, of the University of East Anglia, was commissioned by the World Health Organization and published on July 18, 2018, in Cochrane Database of Systematic Reviews (doi: 10.1002/14651858.CD003177.pub3). It updates a 2004 Cochrane Library review.
As the review notes, “there is a great deal of public belief in the cardiovascular benefits of omega-3 fats.” But research in recent years has questioned the benefits of fish oil supplementation.
For the new review, the researchers focused on studies that boosted omega-3 fat intake using EPA, DHA and ALA.
The researchers included 79 randomized controlled trials with a total of more than 112,000 adult participants. The studies examined the cardiac and circulatory system effects of greater omega-3 consumption for at least a year compared with controls. Most trials tested supplementation with capsules, but some relied on boosting intake of foods or simply making dietary recommendations.
The studies were conducted in North America, Europe, Australia, and Asia. Researchers determined that 25 trials were designed well enough to be deemed “very trustworthy.”
According to the review’s meta-analysis, increasing intake of long-chain omega-3 fatty acids (EPA and DHA) had little to no effect on all-cause mortality (relative risk, 0.98; 39 trials), cardiovascular mortality (RR, 0.95; 25 trials), cardiovascular events (RR, 0.99; 38 trials), coronary heart disease mortality (RR, 0.93; 21 trials) and arrhythmia (RR, 0.97; 28 trials), while there was a potential boost in stroke risk (RR, 1.06; 28 trials).
The investigators noted that the relative risk of coronary heart disease events fell (RR, 0.93; 28 trials), but this did not hold up after sensitivity analysis. All these trials were deemed of moderate or high quality.
As for increased ALA intake, the researchers found little or no effect on all-cause mortality (RR, 1.01; four trials) and cardiovascular mortality (RR, 0.96; four trials) or coronary heart disease events (RR, 1.00, four trials).
Although the researchers deemed the evidence on ALA to be of low quality, there’s a possible decreased risk of cardiovascular events (RR, 0.95; five trials), coronary heart disease mortality (RR, 0.95; three trials) and arrhythmia (RR, 0.79; one trial). It’s not clear if increased ALA intake affects stroke risk.
“There was no evidence that increasing LCn3 [long-chain omega-3 fatty acids] or ALA altered serious adverse events, adiposity, or lipids, although LCn3 slightly reduced triglycerides and increased HDL. ALA probably reduces HDL (high- or moderate-quality evidence),” the researchers wrote.
Why have some earlier studies suggested a significant effect, enough to help convince millions of Americans to add fish oil pills to their diets? “Previous suggestions of benefits from EPA and DHA supplements appear to spring from trials with higher risk of bias,” the researchers wrote. Robert A. Vogel, MD, a cardiologist at the University of Colorado Denver who is familiar with the review findings, said in an interview that “it’s hard to recommend taking fish oil for heart disease. Even if there’s some effect, it’s got to be a very small, 5%-7% reduction at the best.
“That is not to say that there isn’t an effect on triglycerides,” he added. “[Fish oil] is an accepted way of reducing triglycerides. But you don’t reduce triglycerides to reduce heart disease. There are other risks to high triglycerides, such as pancreatitis.”
Dr. Vogel said he’s never taken fish oil supplements, but he does eat fish 5-6 times a week.
The review was funded by the University of East Anglia, Cochrane Heart Group, and the National Institute for Health Research. The review authors reported no relevant disclosures. Dr. Vogel disclosed consulting for the Pritikin Longevity Center.
SOURCE: Abdelhamid AS et al. Cochrane Database Syst Rev. 2018 Jul 18. doi: 10.1002/14651858.CD003177.pub3.
FROM COCHRANE DATABASE OF SYSTEMATIC REVIEWS
Key clinical point: .
Major finding: Increased intake of fish-based omega-3 fats, mainly supplements, seems to barely affect various cardiac/circulatory outcomes. Increased intake of plant-based alpha-linolenic acid may be slightly helpful.
Study details: Review of 79 randomized controlled trials with more than 112,000 adult subjects.
Disclosures: The review was funded by the University of East Anglia, Cochrane Heart Group, and the National Institute for Health Research. The review authors reported no relevant disclosures.
Source: Abdelhamid AS et al. Cochrane Database Syst Rev. 2018 Jul 18. doi: 10.1002/14651858.CD003177.pub3.
Endocrinologists clash over routine CGM during pregnancy
ORLANDO – Diabetes and pregnancy aren’t a good mix, but what about pregnancy and continuous glucose monitors (CGMs)? In a polite but pointed debate, two endocrinologists used each other’s studies as evidence to support their opposing perspectives about routine GCM use by diabetic women during pregnancy.
“This topic shouldn’t really be debated because the evidence is clear” in favor of CGM, said Denice S. Feig, MD, MSc, FRCPC, of the University of Toronto and Mt. Sinai Hospital, also in Toronto, in a presentation at the annual scientific sessions of the American Diabetes Association.
However, Elisabeth R. Mathiesen, MD, DMSc, of Rigshospitalet in Copenhagen, rebutted. She said her own research suggests CGM use may lead to larger babies and more premature births, convincing her to “say no to uncritical use of CGM in pregnancy.”
At issue: What is the best routine treatment for diabetic women before, during, and after pregnancy? As the American Diabetes Association noted in its 2018 Standards of Medical Care in Diabetes report, “specific risks of uncontrolled diabetes in pregnancy include spontaneous abortion, fetal anomalies, preeclampsia, fetal demise, macrosomia, neonatal hypoglycemia, and neonatal hyperbilirubinemia, among others. In addition, diabetes in pregnancy may increase the risk of obesity and type 2 diabetes [mellitus] in offspring later in life.”
In her presentation, Dr. Feig pointed to a 2017 study she led that examined the effectiveness of continuous, real-time CGM on women with type 1 diabetes mellitus who were pregnant or planning to become pregnant (Lancet. 2017 Nov 25;390(10110):2347-2359).
“The study, in effect, was two parallel, randomized trials, one in those who planned pregnancy and one in those who were pregnant,” Dr. Feig said.
Participants, aged 18-40 years, from 31 hospitals in seven European and North American nations, had to have hemoglobin A1c levels greater than or equal to 6.5% during pregnancy or greater than or equal to 7% while planning pregnancy to be included in the study.
“We had a run-in phase to make sure they were able and willing to wear the CGM. Then we had 215 women in the pregnancy arm and 110 in the prepregnancy group randomized to real-time continuous CGM or standard care,” Dr. Feig said. The study ran for 34 weeks in the pregnant patients and for 24 weeks or until conception in the other women.
According to Dr. Feig, 70% of pregnant participants used CGM devices for more than 75% of the time. Compared with the control group, HbA1c levels in those who used CGM fell by 0.19% (P = .0207). The researchers also reported that women in the CGM group spent 100 more minutes a day within the glucose target range.
No differences in outcomes such as gestational age at delivery and rate of preterm delivery was found, although incidence of large-for-gestational-age infants, hypoglycemia requiring dextrose infusion, and neonatal ICU admission were lower in the CGM group to a statistically significant degree. “The numbers needed to treat were very small at six to eight women to reduce one of these events,” said Dr. Feig, who added that the numbers suggest the potential for cost savings.
Whatever the case, she said, “what price would you place on your baby avoiding a prolonged stay in the NICU? I think [it’s] priceless.”
She added that 80% of participants reported having trouble with the devices, which she attributed to the technology being old.
As for the planned pregnancy group, the study noted that “it did not have sufficient power to detect the magnitude of differences that were significant in the pregnancy trial.”
However, Dr. Feig said the study showed a trend toward lower HbA1c levels among CGM users in this population in which “tight glycemic control is absolutely paramount,” and that other studies also provide evidence supporting CGM use through the breastfeeding period.
Dr. Feig also pointed to a similar 2013 study coauthored by Dr. Mathiesen, her debate opponent. Dr. Feig said its findings are weakened because participants used CGM intermittently. She also pointed to the low participation (64%) in CGM by women assigned to a CGM group. (Diabetes Care. 2013 Jul;36[7]:1877-83)
In that study, researchers assigned 123 Danish women with type 1 diabetes mellitus and 31 women with type 2 diabetes mellitus to use real-time CGM for 6 days at various points in pregnancy or to only engage in routine care (including self-monitored plasma glucose seven times daily).
Researchers found no difference in HbA1c levels at 33 weeks between the groups, and they found similar rates of severe hypoglycemia and perinatal outcomes such as large-for-gestational-age infants.
These results, Dr. Mathiesen said, make her skeptical of a blanket recommendation to use CGM in pregnancy. Women aren’t eager to upload their glucose readings, making it difficult for doctors to make adjustments. “My women are Vikings. They come from Denmark,” she said, but “even these women don’t upload their glucose data between visits. ... I rarely have women who upload their data and look at their curves themselves. I think that’s a major disadvantage.”
Dr. Mathiesen also pointed to Dr. Feig’s study and noted that many women used CGM less than 75% of the time. In addition, 80% reported problems with the technology. “I’ve seen lots of skin problems with sensors. One lady used CGM during pregnancy; 4 years later, during another pregnancy, she showed me the mark of her sensor.”
Finally, the cost of CGM use is high considering the ongoing expense of the devices and the nurse time needed to upload data in the clinic. “As a rough estimate, the cost of CGM use in about 20 women during their pregnancies is the cost of the salary for one nurse per year,” she said.
Dr. Feig reported speaking fees from Medtronic, which provided CGM devices at reduced cost to her trial. Dr. Mathiesen reported research funding from the Novo Nordisk Foundation and speaker fees from Novo Nordisk, Lilly, AstraZeneca, and Sanofi-Aventis.
ORLANDO – Diabetes and pregnancy aren’t a good mix, but what about pregnancy and continuous glucose monitors (CGMs)? In a polite but pointed debate, two endocrinologists used each other’s studies as evidence to support their opposing perspectives about routine GCM use by diabetic women during pregnancy.
“This topic shouldn’t really be debated because the evidence is clear” in favor of CGM, said Denice S. Feig, MD, MSc, FRCPC, of the University of Toronto and Mt. Sinai Hospital, also in Toronto, in a presentation at the annual scientific sessions of the American Diabetes Association.
However, Elisabeth R. Mathiesen, MD, DMSc, of Rigshospitalet in Copenhagen, rebutted. She said her own research suggests CGM use may lead to larger babies and more premature births, convincing her to “say no to uncritical use of CGM in pregnancy.”
At issue: What is the best routine treatment for diabetic women before, during, and after pregnancy? As the American Diabetes Association noted in its 2018 Standards of Medical Care in Diabetes report, “specific risks of uncontrolled diabetes in pregnancy include spontaneous abortion, fetal anomalies, preeclampsia, fetal demise, macrosomia, neonatal hypoglycemia, and neonatal hyperbilirubinemia, among others. In addition, diabetes in pregnancy may increase the risk of obesity and type 2 diabetes [mellitus] in offspring later in life.”
In her presentation, Dr. Feig pointed to a 2017 study she led that examined the effectiveness of continuous, real-time CGM on women with type 1 diabetes mellitus who were pregnant or planning to become pregnant (Lancet. 2017 Nov 25;390(10110):2347-2359).
“The study, in effect, was two parallel, randomized trials, one in those who planned pregnancy and one in those who were pregnant,” Dr. Feig said.
Participants, aged 18-40 years, from 31 hospitals in seven European and North American nations, had to have hemoglobin A1c levels greater than or equal to 6.5% during pregnancy or greater than or equal to 7% while planning pregnancy to be included in the study.
“We had a run-in phase to make sure they were able and willing to wear the CGM. Then we had 215 women in the pregnancy arm and 110 in the prepregnancy group randomized to real-time continuous CGM or standard care,” Dr. Feig said. The study ran for 34 weeks in the pregnant patients and for 24 weeks or until conception in the other women.
According to Dr. Feig, 70% of pregnant participants used CGM devices for more than 75% of the time. Compared with the control group, HbA1c levels in those who used CGM fell by 0.19% (P = .0207). The researchers also reported that women in the CGM group spent 100 more minutes a day within the glucose target range.
No differences in outcomes such as gestational age at delivery and rate of preterm delivery was found, although incidence of large-for-gestational-age infants, hypoglycemia requiring dextrose infusion, and neonatal ICU admission were lower in the CGM group to a statistically significant degree. “The numbers needed to treat were very small at six to eight women to reduce one of these events,” said Dr. Feig, who added that the numbers suggest the potential for cost savings.
Whatever the case, she said, “what price would you place on your baby avoiding a prolonged stay in the NICU? I think [it’s] priceless.”
She added that 80% of participants reported having trouble with the devices, which she attributed to the technology being old.
As for the planned pregnancy group, the study noted that “it did not have sufficient power to detect the magnitude of differences that were significant in the pregnancy trial.”
However, Dr. Feig said the study showed a trend toward lower HbA1c levels among CGM users in this population in which “tight glycemic control is absolutely paramount,” and that other studies also provide evidence supporting CGM use through the breastfeeding period.
Dr. Feig also pointed to a similar 2013 study coauthored by Dr. Mathiesen, her debate opponent. Dr. Feig said its findings are weakened because participants used CGM intermittently. She also pointed to the low participation (64%) in CGM by women assigned to a CGM group. (Diabetes Care. 2013 Jul;36[7]:1877-83)
In that study, researchers assigned 123 Danish women with type 1 diabetes mellitus and 31 women with type 2 diabetes mellitus to use real-time CGM for 6 days at various points in pregnancy or to only engage in routine care (including self-monitored plasma glucose seven times daily).
Researchers found no difference in HbA1c levels at 33 weeks between the groups, and they found similar rates of severe hypoglycemia and perinatal outcomes such as large-for-gestational-age infants.
These results, Dr. Mathiesen said, make her skeptical of a blanket recommendation to use CGM in pregnancy. Women aren’t eager to upload their glucose readings, making it difficult for doctors to make adjustments. “My women are Vikings. They come from Denmark,” she said, but “even these women don’t upload their glucose data between visits. ... I rarely have women who upload their data and look at their curves themselves. I think that’s a major disadvantage.”
Dr. Mathiesen also pointed to Dr. Feig’s study and noted that many women used CGM less than 75% of the time. In addition, 80% reported problems with the technology. “I’ve seen lots of skin problems with sensors. One lady used CGM during pregnancy; 4 years later, during another pregnancy, she showed me the mark of her sensor.”
Finally, the cost of CGM use is high considering the ongoing expense of the devices and the nurse time needed to upload data in the clinic. “As a rough estimate, the cost of CGM use in about 20 women during their pregnancies is the cost of the salary for one nurse per year,” she said.
Dr. Feig reported speaking fees from Medtronic, which provided CGM devices at reduced cost to her trial. Dr. Mathiesen reported research funding from the Novo Nordisk Foundation and speaker fees from Novo Nordisk, Lilly, AstraZeneca, and Sanofi-Aventis.
ORLANDO – Diabetes and pregnancy aren’t a good mix, but what about pregnancy and continuous glucose monitors (CGMs)? In a polite but pointed debate, two endocrinologists used each other’s studies as evidence to support their opposing perspectives about routine GCM use by diabetic women during pregnancy.
“This topic shouldn’t really be debated because the evidence is clear” in favor of CGM, said Denice S. Feig, MD, MSc, FRCPC, of the University of Toronto and Mt. Sinai Hospital, also in Toronto, in a presentation at the annual scientific sessions of the American Diabetes Association.
However, Elisabeth R. Mathiesen, MD, DMSc, of Rigshospitalet in Copenhagen, rebutted. She said her own research suggests CGM use may lead to larger babies and more premature births, convincing her to “say no to uncritical use of CGM in pregnancy.”
At issue: What is the best routine treatment for diabetic women before, during, and after pregnancy? As the American Diabetes Association noted in its 2018 Standards of Medical Care in Diabetes report, “specific risks of uncontrolled diabetes in pregnancy include spontaneous abortion, fetal anomalies, preeclampsia, fetal demise, macrosomia, neonatal hypoglycemia, and neonatal hyperbilirubinemia, among others. In addition, diabetes in pregnancy may increase the risk of obesity and type 2 diabetes [mellitus] in offspring later in life.”
In her presentation, Dr. Feig pointed to a 2017 study she led that examined the effectiveness of continuous, real-time CGM on women with type 1 diabetes mellitus who were pregnant or planning to become pregnant (Lancet. 2017 Nov 25;390(10110):2347-2359).
“The study, in effect, was two parallel, randomized trials, one in those who planned pregnancy and one in those who were pregnant,” Dr. Feig said.
Participants, aged 18-40 years, from 31 hospitals in seven European and North American nations, had to have hemoglobin A1c levels greater than or equal to 6.5% during pregnancy or greater than or equal to 7% while planning pregnancy to be included in the study.
“We had a run-in phase to make sure they were able and willing to wear the CGM. Then we had 215 women in the pregnancy arm and 110 in the prepregnancy group randomized to real-time continuous CGM or standard care,” Dr. Feig said. The study ran for 34 weeks in the pregnant patients and for 24 weeks or until conception in the other women.
According to Dr. Feig, 70% of pregnant participants used CGM devices for more than 75% of the time. Compared with the control group, HbA1c levels in those who used CGM fell by 0.19% (P = .0207). The researchers also reported that women in the CGM group spent 100 more minutes a day within the glucose target range.
No differences in outcomes such as gestational age at delivery and rate of preterm delivery was found, although incidence of large-for-gestational-age infants, hypoglycemia requiring dextrose infusion, and neonatal ICU admission were lower in the CGM group to a statistically significant degree. “The numbers needed to treat were very small at six to eight women to reduce one of these events,” said Dr. Feig, who added that the numbers suggest the potential for cost savings.
Whatever the case, she said, “what price would you place on your baby avoiding a prolonged stay in the NICU? I think [it’s] priceless.”
She added that 80% of participants reported having trouble with the devices, which she attributed to the technology being old.
As for the planned pregnancy group, the study noted that “it did not have sufficient power to detect the magnitude of differences that were significant in the pregnancy trial.”
However, Dr. Feig said the study showed a trend toward lower HbA1c levels among CGM users in this population in which “tight glycemic control is absolutely paramount,” and that other studies also provide evidence supporting CGM use through the breastfeeding period.
Dr. Feig also pointed to a similar 2013 study coauthored by Dr. Mathiesen, her debate opponent. Dr. Feig said its findings are weakened because participants used CGM intermittently. She also pointed to the low participation (64%) in CGM by women assigned to a CGM group. (Diabetes Care. 2013 Jul;36[7]:1877-83)
In that study, researchers assigned 123 Danish women with type 1 diabetes mellitus and 31 women with type 2 diabetes mellitus to use real-time CGM for 6 days at various points in pregnancy or to only engage in routine care (including self-monitored plasma glucose seven times daily).
Researchers found no difference in HbA1c levels at 33 weeks between the groups, and they found similar rates of severe hypoglycemia and perinatal outcomes such as large-for-gestational-age infants.
These results, Dr. Mathiesen said, make her skeptical of a blanket recommendation to use CGM in pregnancy. Women aren’t eager to upload their glucose readings, making it difficult for doctors to make adjustments. “My women are Vikings. They come from Denmark,” she said, but “even these women don’t upload their glucose data between visits. ... I rarely have women who upload their data and look at their curves themselves. I think that’s a major disadvantage.”
Dr. Mathiesen also pointed to Dr. Feig’s study and noted that many women used CGM less than 75% of the time. In addition, 80% reported problems with the technology. “I’ve seen lots of skin problems with sensors. One lady used CGM during pregnancy; 4 years later, during another pregnancy, she showed me the mark of her sensor.”
Finally, the cost of CGM use is high considering the ongoing expense of the devices and the nurse time needed to upload data in the clinic. “As a rough estimate, the cost of CGM use in about 20 women during their pregnancies is the cost of the salary for one nurse per year,” she said.
Dr. Feig reported speaking fees from Medtronic, which provided CGM devices at reduced cost to her trial. Dr. Mathiesen reported research funding from the Novo Nordisk Foundation and speaker fees from Novo Nordisk, Lilly, AstraZeneca, and Sanofi-Aventis.
EXPERT ANALYSIS FROM ADA 2018