User login
Survey: Most military family physicians wary of transgender treatments
The U.S. military now allows transgender people to serve openly, and all medical personnel are supposed to be trained in their treatment – including care related to gender transition – by June 2017.
But a new survey of 204 family physicians who work for the military shows that most aren’t willing to prescribe cross-hormone treatment to eligible adult transgender patients.
In addition, “we found that most military clinicians did not receive any formal training on transgender care during their medical education, most had not treated a patient with gender dysphoria, and most had not received sufficient training to prescribe cross-hormone therapy,” said Natasha A. Schvey, PhD, an assistant professor with the medical and clinical psychology department at the Uniformed Services University of the Health Sciences, Bethesda, Md., and lead author of a new study that summarizes the survey findings.
An estimated 12,800 transgender people serve in the U.S. military, and researchers believe that they make up a larger proportion of the military population than in the general population.
In October 2016, the Department of Defense ended its ban on service by transgender troops. A policy statement says: “Service members with a diagnosis from a military medical provider indicating that gender transition is medically necessary will be provided medical care and treatment for the diagnosed medical condition, in the same manner as other medical care and treatment.”
For the new study, Dr. Schvey and her associates surveyed 204 of the approximately 1,700 family physicians who serve the military (JAMA Intern Med. 2017 Mar 13. doi: 10.1001/jamainternmed.2017.0136). The survey respondents had all attended an annual meeting in March 2016.
Most respondents were male (62.8%) and white (85.5%); 21.7% were residents. The highest number of respondents (42.6%) worked for the U.S. Air Force.
Almost three-quarters of the respondents said that they had received no training or medical education in gender dysphoria, and 62.7% said they had never treated a transgender patient as a physician.
Just under half (47.1%) of respondents – 82 – said they would prescribe cross-hormone therapy to eligible adult patients, but only one would do so independently. The others would only do so with additional education (7.5% of all responding physicians), the help of an experienced clinician (17.2%), or both (21.8%).
The other 52.9% said they wouldn’t prescribe the hormone therapy. Eight percent of all responding physicians blamed ethical concerns, 19.5% pointed to lack of comfort, and 25.3% mentioned both, the investigators reported.
Why are so many physicians so uncomfortable?
Dr. Schvey said the survey design doesn’t allow for speculation about what the ethical concerns might be, but “the data did indicate that hours of training on transgender care were directly associated with the likelihood of prescribing cross-hormone therapy. Therefore, some of the lack of comfort may be due to lack of experience and adequate training.”
Although family physicians are generally used to treating patients with hormones for contraception and to treat perimenopausal symptoms and hypogonadism, many don’t know about dosing regimens and other information regarding their use in transgender patients, study coauthor David A. Klein, MD, assistant professor of family medicine and pediatrics at the Uniformed Services University of the Health Sciences, said in an interview. And, he noted, “some are unaware that provision of cross-hormone therapy is within their scope of care.”
Going forward, “given that greater education in transgender care is associated with increased competency,” Dr. Schvey said in an interview, “it will be important to assess the training of military physicians to ensure skill and sensitivity in treating patients who identify as transgender.”
The study had no specific funding, and the authors reported no relevant conflicts of interest.
I first learned the term transsexual from a 1972 publication by the American Medical Association titled “Human Sexuality.” I found the textbook on my father’s bookshelf as a 10-year-old and for the first time found a word to describe my inner world.
Up until 2016, transgender individuals in the military were not given appropriate medical or psychological assistance. They were forced to seek help in secret, outside the military health care system, or to await discharge proceedings.
Although the situation has improved markedly, there is still a long way to go. We need better protocols for individuals who wish to transition while active duty, for retirees, and their family members. We need to aid them in preserving their fertility, and we need to foster an environment of openness where no soldier feels like he or she is isolated from fellow service members owing to gender identity.
Although I completed my medical training as a male, today I serve as a female physician in every respect within the Department of Defense. Last month, I graduated the Army Medical Department’s Advanced Course with honors, and now I look forward to the second half of my military career being treated like any other capable military physician. My hope is that over time all clinicians gain comfort and skill in treating transgender persons.
Jamie L. Henry, MD, is with the Walter Reed National Military Medical Center in Bethesda, Md. These comments were taken from an editorial (JAMA Intern Med. 2017 Mar 13. doi: 10.1001/jamainternmed.2017.0140) accompanying the report by Dr. Schvey and her associates.
I first learned the term transsexual from a 1972 publication by the American Medical Association titled “Human Sexuality.” I found the textbook on my father’s bookshelf as a 10-year-old and for the first time found a word to describe my inner world.
Up until 2016, transgender individuals in the military were not given appropriate medical or psychological assistance. They were forced to seek help in secret, outside the military health care system, or to await discharge proceedings.
Although the situation has improved markedly, there is still a long way to go. We need better protocols for individuals who wish to transition while active duty, for retirees, and their family members. We need to aid them in preserving their fertility, and we need to foster an environment of openness where no soldier feels like he or she is isolated from fellow service members owing to gender identity.
Although I completed my medical training as a male, today I serve as a female physician in every respect within the Department of Defense. Last month, I graduated the Army Medical Department’s Advanced Course with honors, and now I look forward to the second half of my military career being treated like any other capable military physician. My hope is that over time all clinicians gain comfort and skill in treating transgender persons.
Jamie L. Henry, MD, is with the Walter Reed National Military Medical Center in Bethesda, Md. These comments were taken from an editorial (JAMA Intern Med. 2017 Mar 13. doi: 10.1001/jamainternmed.2017.0140) accompanying the report by Dr. Schvey and her associates.
I first learned the term transsexual from a 1972 publication by the American Medical Association titled “Human Sexuality.” I found the textbook on my father’s bookshelf as a 10-year-old and for the first time found a word to describe my inner world.
Up until 2016, transgender individuals in the military were not given appropriate medical or psychological assistance. They were forced to seek help in secret, outside the military health care system, or to await discharge proceedings.
Although the situation has improved markedly, there is still a long way to go. We need better protocols for individuals who wish to transition while active duty, for retirees, and their family members. We need to aid them in preserving their fertility, and we need to foster an environment of openness where no soldier feels like he or she is isolated from fellow service members owing to gender identity.
Although I completed my medical training as a male, today I serve as a female physician in every respect within the Department of Defense. Last month, I graduated the Army Medical Department’s Advanced Course with honors, and now I look forward to the second half of my military career being treated like any other capable military physician. My hope is that over time all clinicians gain comfort and skill in treating transgender persons.
Jamie L. Henry, MD, is with the Walter Reed National Military Medical Center in Bethesda, Md. These comments were taken from an editorial (JAMA Intern Med. 2017 Mar 13. doi: 10.1001/jamainternmed.2017.0140) accompanying the report by Dr. Schvey and her associates.
The U.S. military now allows transgender people to serve openly, and all medical personnel are supposed to be trained in their treatment – including care related to gender transition – by June 2017.
But a new survey of 204 family physicians who work for the military shows that most aren’t willing to prescribe cross-hormone treatment to eligible adult transgender patients.
In addition, “we found that most military clinicians did not receive any formal training on transgender care during their medical education, most had not treated a patient with gender dysphoria, and most had not received sufficient training to prescribe cross-hormone therapy,” said Natasha A. Schvey, PhD, an assistant professor with the medical and clinical psychology department at the Uniformed Services University of the Health Sciences, Bethesda, Md., and lead author of a new study that summarizes the survey findings.
An estimated 12,800 transgender people serve in the U.S. military, and researchers believe that they make up a larger proportion of the military population than in the general population.
In October 2016, the Department of Defense ended its ban on service by transgender troops. A policy statement says: “Service members with a diagnosis from a military medical provider indicating that gender transition is medically necessary will be provided medical care and treatment for the diagnosed medical condition, in the same manner as other medical care and treatment.”
For the new study, Dr. Schvey and her associates surveyed 204 of the approximately 1,700 family physicians who serve the military (JAMA Intern Med. 2017 Mar 13. doi: 10.1001/jamainternmed.2017.0136). The survey respondents had all attended an annual meeting in March 2016.
Most respondents were male (62.8%) and white (85.5%); 21.7% were residents. The highest number of respondents (42.6%) worked for the U.S. Air Force.
Almost three-quarters of the respondents said that they had received no training or medical education in gender dysphoria, and 62.7% said they had never treated a transgender patient as a physician.
Just under half (47.1%) of respondents – 82 – said they would prescribe cross-hormone therapy to eligible adult patients, but only one would do so independently. The others would only do so with additional education (7.5% of all responding physicians), the help of an experienced clinician (17.2%), or both (21.8%).
The other 52.9% said they wouldn’t prescribe the hormone therapy. Eight percent of all responding physicians blamed ethical concerns, 19.5% pointed to lack of comfort, and 25.3% mentioned both, the investigators reported.
Why are so many physicians so uncomfortable?
Dr. Schvey said the survey design doesn’t allow for speculation about what the ethical concerns might be, but “the data did indicate that hours of training on transgender care were directly associated with the likelihood of prescribing cross-hormone therapy. Therefore, some of the lack of comfort may be due to lack of experience and adequate training.”
Although family physicians are generally used to treating patients with hormones for contraception and to treat perimenopausal symptoms and hypogonadism, many don’t know about dosing regimens and other information regarding their use in transgender patients, study coauthor David A. Klein, MD, assistant professor of family medicine and pediatrics at the Uniformed Services University of the Health Sciences, said in an interview. And, he noted, “some are unaware that provision of cross-hormone therapy is within their scope of care.”
Going forward, “given that greater education in transgender care is associated with increased competency,” Dr. Schvey said in an interview, “it will be important to assess the training of military physicians to ensure skill and sensitivity in treating patients who identify as transgender.”
The study had no specific funding, and the authors reported no relevant conflicts of interest.
The U.S. military now allows transgender people to serve openly, and all medical personnel are supposed to be trained in their treatment – including care related to gender transition – by June 2017.
But a new survey of 204 family physicians who work for the military shows that most aren’t willing to prescribe cross-hormone treatment to eligible adult transgender patients.
In addition, “we found that most military clinicians did not receive any formal training on transgender care during their medical education, most had not treated a patient with gender dysphoria, and most had not received sufficient training to prescribe cross-hormone therapy,” said Natasha A. Schvey, PhD, an assistant professor with the medical and clinical psychology department at the Uniformed Services University of the Health Sciences, Bethesda, Md., and lead author of a new study that summarizes the survey findings.
An estimated 12,800 transgender people serve in the U.S. military, and researchers believe that they make up a larger proportion of the military population than in the general population.
In October 2016, the Department of Defense ended its ban on service by transgender troops. A policy statement says: “Service members with a diagnosis from a military medical provider indicating that gender transition is medically necessary will be provided medical care and treatment for the diagnosed medical condition, in the same manner as other medical care and treatment.”
For the new study, Dr. Schvey and her associates surveyed 204 of the approximately 1,700 family physicians who serve the military (JAMA Intern Med. 2017 Mar 13. doi: 10.1001/jamainternmed.2017.0136). The survey respondents had all attended an annual meeting in March 2016.
Most respondents were male (62.8%) and white (85.5%); 21.7% were residents. The highest number of respondents (42.6%) worked for the U.S. Air Force.
Almost three-quarters of the respondents said that they had received no training or medical education in gender dysphoria, and 62.7% said they had never treated a transgender patient as a physician.
Just under half (47.1%) of respondents – 82 – said they would prescribe cross-hormone therapy to eligible adult patients, but only one would do so independently. The others would only do so with additional education (7.5% of all responding physicians), the help of an experienced clinician (17.2%), or both (21.8%).
The other 52.9% said they wouldn’t prescribe the hormone therapy. Eight percent of all responding physicians blamed ethical concerns, 19.5% pointed to lack of comfort, and 25.3% mentioned both, the investigators reported.
Why are so many physicians so uncomfortable?
Dr. Schvey said the survey design doesn’t allow for speculation about what the ethical concerns might be, but “the data did indicate that hours of training on transgender care were directly associated with the likelihood of prescribing cross-hormone therapy. Therefore, some of the lack of comfort may be due to lack of experience and adequate training.”
Although family physicians are generally used to treating patients with hormones for contraception and to treat perimenopausal symptoms and hypogonadism, many don’t know about dosing regimens and other information regarding their use in transgender patients, study coauthor David A. Klein, MD, assistant professor of family medicine and pediatrics at the Uniformed Services University of the Health Sciences, said in an interview. And, he noted, “some are unaware that provision of cross-hormone therapy is within their scope of care.”
Going forward, “given that greater education in transgender care is associated with increased competency,” Dr. Schvey said in an interview, “it will be important to assess the training of military physicians to ensure skill and sensitivity in treating patients who identify as transgender.”
The study had no specific funding, and the authors reported no relevant conflicts of interest.
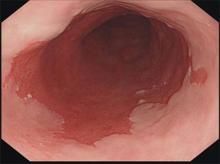
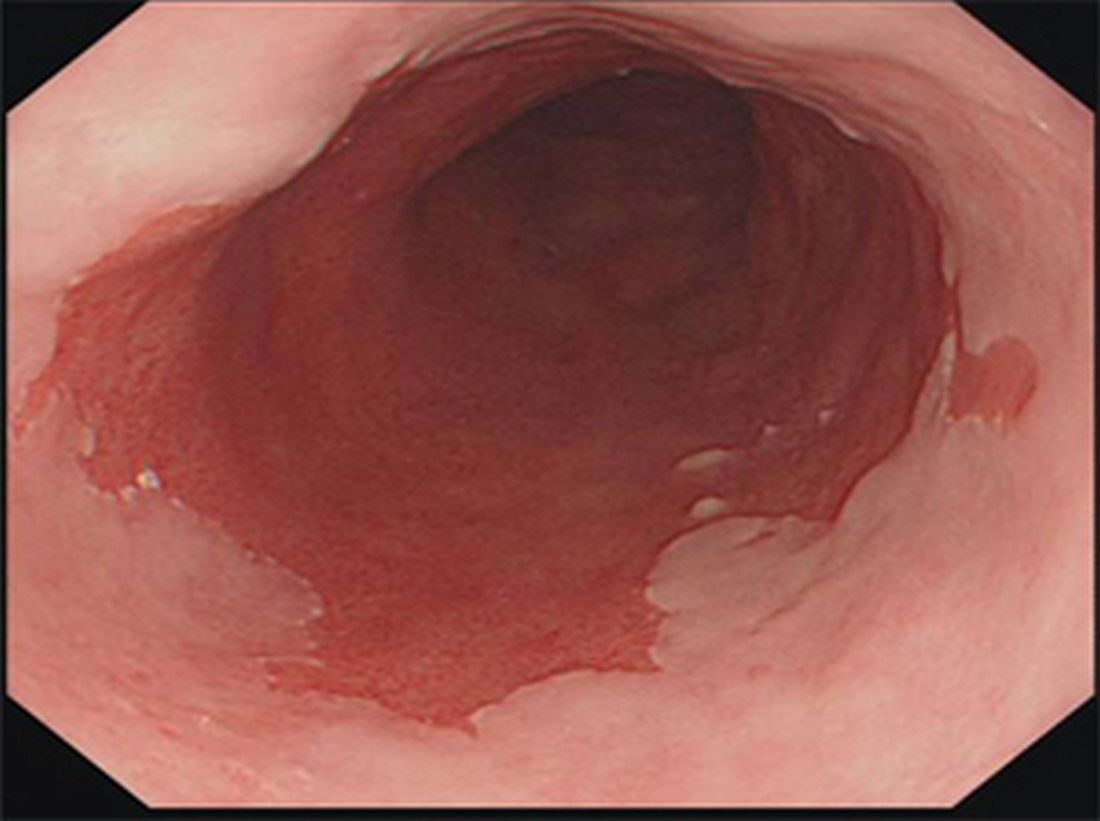
Barrett’s esophagus: Cancer risk is highest in first year after diagnosis
The highest risk of diagnosis with esophageal adenocarcinoma occurs in the first month after diagnosis with Barrett’s esophagus (BE).
Further, the risk of this type of esophageal cancer is lower than previously believed after that first year, according to a study of 7,932 Swedish residents who were diagnosed with BE from 2006 to 2013.
Esophageal adenocarcinoma (EA) is the most common type of esophageal cancer in the United States, and more than half of patients present with late-stage disease. “The connection between BE and EA is well known, and risk factors for both conditions remain vastly the same, including male sex, age over 50 years, central obesity, and symptomatic GERD [gastroesophageal reflux disease],” Dr. Holmberg said in an interview.
“In order to reduce mortality from esophageal cancer through surveillance and early treatment, we need to improve on BE screening programs,” he said. He also noted that, of all EA cases in Sweden, where screening for BE itself is not recommended in national guidelines, an “astonishing” 97% were diagnosed in patients with previously unknown BE.
Screening programs urge regular endoscopic surveillance in nondysplastic BE patients, but Dr. Holmberg said that their cost-effectiveness is questionable. Complicating matters, there has been conflicting research about the risk of EA that these patients actually face.
As the new study notes, patients with BE were previously thought to face a 30-125 times greater relative risk of EA than the general population did and a mean annual risk of 0.5%. But, recent studies in Europe and the U.S. have suggested that the risk is lower, with an estimated annual risk range of 0.12%-0.18%.
Dr. Holmberg said he and his colleagues launched the new study to better understand the risk in the first year after BE diagnosis.
The researchers used the Swedish National Patient Register to track 7,932 Swedish residents who were diagnosed with BE from 2006 to 2013. About two-thirds were male, and the median age at BE diagnosis was 66 years.
The findings appear in the European Journal of Cancer (2017;75:41-6). Of the patients with BE, 89 new cases of EA were diagnosed 62% of the time within a year of BE diagnosis.
Over a median follow-up of 2.13 person-years, the overall incidence of EA in the patients with BE was 1.47 (95% CI, 0.91-2.02) per 1,000 person-years. That is the equivalent of an annual risk of 0.15%.
The highest incidence rate by far was in the period of 7 to 30 days after diagnosis at 15.53 (95% CI, 4.77-26.29). The incident rates varied from 1.44 to 4.10 (95% CI, 0.00-7.38) for other periods (31-100 days, 101 days to 6 months, over 6 months to 1 year, over 1 year to 3 years, over 3 years to end of follow-up).
How should physicians interpret the study findings and look forward? “Nondysplastic BE represents almost 80% of all BE,” Dr. Holmberg said, but “nondiscriminatory long-term surveillance in nondysplastic BE is not cost-efficient.”
However, while this is “the group with the least risk of EA development, it still generates a large chunk of all EA cases. It is likely that future management of nondysplastic BE will include more risk factors than just degree of dysplasia.”
He added that “individuals with multiple risk factors, such as Caucasian males, age over 50 years, obesity, symptomatic reflux, and with long-segment nondysplastic BE would likely benefit more from more frequent surveillance or even prophylactic treatment with radio-frequency ablation compared to individuals without those risk factors.”
The study was funded by the Swedish Cancer Society and the Swedish Research Council. The authors report no relevant disclosures.
The highest risk of diagnosis with esophageal adenocarcinoma occurs in the first month after diagnosis with Barrett’s esophagus (BE).
Further, the risk of this type of esophageal cancer is lower than previously believed after that first year, according to a study of 7,932 Swedish residents who were diagnosed with BE from 2006 to 2013.
Esophageal adenocarcinoma (EA) is the most common type of esophageal cancer in the United States, and more than half of patients present with late-stage disease. “The connection between BE and EA is well known, and risk factors for both conditions remain vastly the same, including male sex, age over 50 years, central obesity, and symptomatic GERD [gastroesophageal reflux disease],” Dr. Holmberg said in an interview.
“In order to reduce mortality from esophageal cancer through surveillance and early treatment, we need to improve on BE screening programs,” he said. He also noted that, of all EA cases in Sweden, where screening for BE itself is not recommended in national guidelines, an “astonishing” 97% were diagnosed in patients with previously unknown BE.
Screening programs urge regular endoscopic surveillance in nondysplastic BE patients, but Dr. Holmberg said that their cost-effectiveness is questionable. Complicating matters, there has been conflicting research about the risk of EA that these patients actually face.
As the new study notes, patients with BE were previously thought to face a 30-125 times greater relative risk of EA than the general population did and a mean annual risk of 0.5%. But, recent studies in Europe and the U.S. have suggested that the risk is lower, with an estimated annual risk range of 0.12%-0.18%.
Dr. Holmberg said he and his colleagues launched the new study to better understand the risk in the first year after BE diagnosis.
The researchers used the Swedish National Patient Register to track 7,932 Swedish residents who were diagnosed with BE from 2006 to 2013. About two-thirds were male, and the median age at BE diagnosis was 66 years.
The findings appear in the European Journal of Cancer (2017;75:41-6). Of the patients with BE, 89 new cases of EA were diagnosed 62% of the time within a year of BE diagnosis.
Over a median follow-up of 2.13 person-years, the overall incidence of EA in the patients with BE was 1.47 (95% CI, 0.91-2.02) per 1,000 person-years. That is the equivalent of an annual risk of 0.15%.
The highest incidence rate by far was in the period of 7 to 30 days after diagnosis at 15.53 (95% CI, 4.77-26.29). The incident rates varied from 1.44 to 4.10 (95% CI, 0.00-7.38) for other periods (31-100 days, 101 days to 6 months, over 6 months to 1 year, over 1 year to 3 years, over 3 years to end of follow-up).
How should physicians interpret the study findings and look forward? “Nondysplastic BE represents almost 80% of all BE,” Dr. Holmberg said, but “nondiscriminatory long-term surveillance in nondysplastic BE is not cost-efficient.”
However, while this is “the group with the least risk of EA development, it still generates a large chunk of all EA cases. It is likely that future management of nondysplastic BE will include more risk factors than just degree of dysplasia.”
He added that “individuals with multiple risk factors, such as Caucasian males, age over 50 years, obesity, symptomatic reflux, and with long-segment nondysplastic BE would likely benefit more from more frequent surveillance or even prophylactic treatment with radio-frequency ablation compared to individuals without those risk factors.”
The study was funded by the Swedish Cancer Society and the Swedish Research Council. The authors report no relevant disclosures.
The highest risk of diagnosis with esophageal adenocarcinoma occurs in the first month after diagnosis with Barrett’s esophagus (BE).
Further, the risk of this type of esophageal cancer is lower than previously believed after that first year, according to a study of 7,932 Swedish residents who were diagnosed with BE from 2006 to 2013.
Esophageal adenocarcinoma (EA) is the most common type of esophageal cancer in the United States, and more than half of patients present with late-stage disease. “The connection between BE and EA is well known, and risk factors for both conditions remain vastly the same, including male sex, age over 50 years, central obesity, and symptomatic GERD [gastroesophageal reflux disease],” Dr. Holmberg said in an interview.
“In order to reduce mortality from esophageal cancer through surveillance and early treatment, we need to improve on BE screening programs,” he said. He also noted that, of all EA cases in Sweden, where screening for BE itself is not recommended in national guidelines, an “astonishing” 97% were diagnosed in patients with previously unknown BE.
Screening programs urge regular endoscopic surveillance in nondysplastic BE patients, but Dr. Holmberg said that their cost-effectiveness is questionable. Complicating matters, there has been conflicting research about the risk of EA that these patients actually face.
As the new study notes, patients with BE were previously thought to face a 30-125 times greater relative risk of EA than the general population did and a mean annual risk of 0.5%. But, recent studies in Europe and the U.S. have suggested that the risk is lower, with an estimated annual risk range of 0.12%-0.18%.
Dr. Holmberg said he and his colleagues launched the new study to better understand the risk in the first year after BE diagnosis.
The researchers used the Swedish National Patient Register to track 7,932 Swedish residents who were diagnosed with BE from 2006 to 2013. About two-thirds were male, and the median age at BE diagnosis was 66 years.
The findings appear in the European Journal of Cancer (2017;75:41-6). Of the patients with BE, 89 new cases of EA were diagnosed 62% of the time within a year of BE diagnosis.
Over a median follow-up of 2.13 person-years, the overall incidence of EA in the patients with BE was 1.47 (95% CI, 0.91-2.02) per 1,000 person-years. That is the equivalent of an annual risk of 0.15%.
The highest incidence rate by far was in the period of 7 to 30 days after diagnosis at 15.53 (95% CI, 4.77-26.29). The incident rates varied from 1.44 to 4.10 (95% CI, 0.00-7.38) for other periods (31-100 days, 101 days to 6 months, over 6 months to 1 year, over 1 year to 3 years, over 3 years to end of follow-up).
How should physicians interpret the study findings and look forward? “Nondysplastic BE represents almost 80% of all BE,” Dr. Holmberg said, but “nondiscriminatory long-term surveillance in nondysplastic BE is not cost-efficient.”
However, while this is “the group with the least risk of EA development, it still generates a large chunk of all EA cases. It is likely that future management of nondysplastic BE will include more risk factors than just degree of dysplasia.”
He added that “individuals with multiple risk factors, such as Caucasian males, age over 50 years, obesity, symptomatic reflux, and with long-segment nondysplastic BE would likely benefit more from more frequent surveillance or even prophylactic treatment with radio-frequency ablation compared to individuals without those risk factors.”
The study was funded by the Swedish Cancer Society and the Swedish Research Council. The authors report no relevant disclosures.
FROM THE EUROPEAN JOURNAL OF CANCER
Key clinical point: While their overall risk over the short term is low, patients with Barrett’s esophagus are especially likely to be diagnosed with esophageal adenocarcinoma in the first weeks after Barrett’s diagnosis.
Major finding: Over a median of 2.13 person-years, the annual risk of esophageal adenocarcinoma diagnosis was 0.15% in patients with Barrett’s esophagus, but the incidence rate was especially high (15.53; 95% confidence interval, 4.77-26.29) from 7 to 30 days after Barrett’s diagnosis, compared with rates of 1.44-4.10 (95% CI, 0.00-7.38) for later periods.
Data source: A population-centered analysis of 7,932 Swedish residents diagnosed with Barrett’s esophagus from 2006 to 2013.
Disclosures: The study was funded by the Swedish Cancer Society and the Swedish Research Council. The authors report no relevant disclosures.
Psoriasis and depression may pack a PsA punch
Patients with both psoriasis and major depressive disorder face an adjusted 37% greater risk of developing psoriatic arthritis (PsA) over a median follow-up of 5.1 years, a new study finds.
The findings don’t definitively prove that depression plays a role in PsA. Still, “,” study coauthor Cheryl Barnabe, MD, MSc, said in an interview.
For the new study published by the Journal of Investigative Dermatology, researchers tracked 73,447 people in the United Kingdom with psoriasis through a primary care medical records database for up to 25 years. The study statistics come from the years 1987-2012, reported Ryan T. Lewinson of the Cumming School of Medicine, in Calgary, Alta., and his associates (J Invest Dermatol. 2017. doi: 10.1016/j.jid.2016.11.032).
The median age at psoriasis diagnosis was 49.5 years (range, 20-90 years) and the median follow-up time was 5.1 years; 2% of the patients developed PsA and 7% developed major depression.
Via an unadjusted model, those with signs of depression were 1.56 times more likely to develop PsA (hazard ratio; 95% CI, 1.28-1.90; P less than .0001). In a model adjusted for factors such as age, gender, and obesity status, the extra risk of PsA was 1.37 (HR; 95% CI, 1.05-1.80; P = .021).
“The study draws into question the biological mechanisms by which depression increases the risk for developing psoriatic arthritis,” said Dr. Barnabe, an associate professor with the departments of medicine and community health sciences at the University of Calgary and a rheumatologist with Alberta Health Services.
The study notes that depression is linked to poor diet and lack of exercise, factors that could contribute to PsA. The authors also point out that researchers have linked depression to inflammation, a crucial component of both psoriasis and PsA, although they note that the study doesn’t examine systemic inflammation.
What’s next? “Mental health in chronic inflammatory diseases is not well addressed at the present time, in our system anyway, and should be a prime area of focus,” Dr. Barnabe noted. “Depression occurs at elevated rates in both psoriasis and psoriatic arthritis, and there is certainly a role for treatment to assist with disease management.”
The study authors reported no specific study funding and no relevant financial disclosures.
Patients with both psoriasis and major depressive disorder face an adjusted 37% greater risk of developing psoriatic arthritis (PsA) over a median follow-up of 5.1 years, a new study finds.
The findings don’t definitively prove that depression plays a role in PsA. Still, “,” study coauthor Cheryl Barnabe, MD, MSc, said in an interview.
For the new study published by the Journal of Investigative Dermatology, researchers tracked 73,447 people in the United Kingdom with psoriasis through a primary care medical records database for up to 25 years. The study statistics come from the years 1987-2012, reported Ryan T. Lewinson of the Cumming School of Medicine, in Calgary, Alta., and his associates (J Invest Dermatol. 2017. doi: 10.1016/j.jid.2016.11.032).
The median age at psoriasis diagnosis was 49.5 years (range, 20-90 years) and the median follow-up time was 5.1 years; 2% of the patients developed PsA and 7% developed major depression.
Via an unadjusted model, those with signs of depression were 1.56 times more likely to develop PsA (hazard ratio; 95% CI, 1.28-1.90; P less than .0001). In a model adjusted for factors such as age, gender, and obesity status, the extra risk of PsA was 1.37 (HR; 95% CI, 1.05-1.80; P = .021).
“The study draws into question the biological mechanisms by which depression increases the risk for developing psoriatic arthritis,” said Dr. Barnabe, an associate professor with the departments of medicine and community health sciences at the University of Calgary and a rheumatologist with Alberta Health Services.
The study notes that depression is linked to poor diet and lack of exercise, factors that could contribute to PsA. The authors also point out that researchers have linked depression to inflammation, a crucial component of both psoriasis and PsA, although they note that the study doesn’t examine systemic inflammation.
What’s next? “Mental health in chronic inflammatory diseases is not well addressed at the present time, in our system anyway, and should be a prime area of focus,” Dr. Barnabe noted. “Depression occurs at elevated rates in both psoriasis and psoriatic arthritis, and there is certainly a role for treatment to assist with disease management.”
The study authors reported no specific study funding and no relevant financial disclosures.
Patients with both psoriasis and major depressive disorder face an adjusted 37% greater risk of developing psoriatic arthritis (PsA) over a median follow-up of 5.1 years, a new study finds.
The findings don’t definitively prove that depression plays a role in PsA. Still, “,” study coauthor Cheryl Barnabe, MD, MSc, said in an interview.
For the new study published by the Journal of Investigative Dermatology, researchers tracked 73,447 people in the United Kingdom with psoriasis through a primary care medical records database for up to 25 years. The study statistics come from the years 1987-2012, reported Ryan T. Lewinson of the Cumming School of Medicine, in Calgary, Alta., and his associates (J Invest Dermatol. 2017. doi: 10.1016/j.jid.2016.11.032).
The median age at psoriasis diagnosis was 49.5 years (range, 20-90 years) and the median follow-up time was 5.1 years; 2% of the patients developed PsA and 7% developed major depression.
Via an unadjusted model, those with signs of depression were 1.56 times more likely to develop PsA (hazard ratio; 95% CI, 1.28-1.90; P less than .0001). In a model adjusted for factors such as age, gender, and obesity status, the extra risk of PsA was 1.37 (HR; 95% CI, 1.05-1.80; P = .021).
“The study draws into question the biological mechanisms by which depression increases the risk for developing psoriatic arthritis,” said Dr. Barnabe, an associate professor with the departments of medicine and community health sciences at the University of Calgary and a rheumatologist with Alberta Health Services.
The study notes that depression is linked to poor diet and lack of exercise, factors that could contribute to PsA. The authors also point out that researchers have linked depression to inflammation, a crucial component of both psoriasis and PsA, although they note that the study doesn’t examine systemic inflammation.
What’s next? “Mental health in chronic inflammatory diseases is not well addressed at the present time, in our system anyway, and should be a prime area of focus,” Dr. Barnabe noted. “Depression occurs at elevated rates in both psoriasis and psoriatic arthritis, and there is certainly a role for treatment to assist with disease management.”
The study authors reported no specific study funding and no relevant financial disclosures.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
Key clinical point: Depression is especially common in patients with psoriasis, and those with both conditions appear to face a higher risk of psoriatic arthritis.
Major finding: In an adjusted model, patients with psoriasis and signs of major depression were 37% more likely to develop PsA.
Data source: Retrospective study of 73,447 patients with psoriasis from the U.K. tracked for up to 25 years (median follow-up, 5.1 years).
Disclosures: The authors reported no specific study funding and no relevant financial disclosures.
Guidelines needed for outpatient opioid use after vaginal delivery
A new study of pregnant Medicaid patients in Pennsylvania found that 12% filled prescriptions for opioids within 5 days of vaginal delivery, even though fewer than one-third of the women had pain-inducing conditions.
“Our study raises the question: Why is outpatient opioid use not rare after vaginal delivery?” lead author Marian Jarlenski, PhD, MPH, said in an interview. “Outpatient opioid prescriptions after a hospitalization may be one potential pathway to opioid use disorder. I hope the study will prompt some thought about why opioids are being prescribed for women after vaginal delivery and how to best manage postdelivery pain. This is especially important in areas of the country that have extraordinarily high rates of opioid use disorder.”
A total of 12% of the women (18,131) filled an outpatient prescription for an opioid with 5 days of giving birth. Of those, just 28.2% (5,110) had one or more conditions that cause an increased level of pain after delivery, such as bilateral tubal ligation, certain kinds of lacerations, and episiotomy.
During the first 5 days after birth, the most commonly prescribed opioid was oxycodone-acetaminophen (53.3%), followed by acetaminophen-codeine (20.5%), and hydrocodone-acetaminophen (19.6%).
Of the 18,131 women with an early postdelivery opioid prescription, 14% (2,592) filled at least one other opioid prescription within 6-60 days – that’s 1.6% of all the women in the study.
“On a positive note, we saw the supply of opioid prescriptions was generally short at 3-7 days,” Dr. Jarlenski said.
The researchers linked tobacco use and a mental health condition (both adjusted odds ratio 1.3; 95% confidence interval, 1.2-1.4) to a higher risk of filling a prescription without being diagnosed with a pain-causing disorder. They also found an association between substance use disorder (not related to opioids) and a higher risk of these types of prescriptions, but only for filling a second opioid prescription in the 6-60 day period (aOR 1.4; 95% CI, 1.2-1.6).
The researchers called for national guidelines regarding the use of opioids after birth.
“Several organizations have developed opioid prescribing guidelines for chronic and acute pain,” Dr. Jarlenski said. “These guidelines are necessary because the risk of opioid use disorder and subsequent overdose events is well established. Although delivery is the most common reason for hospitalization in the United States, there are no national guidelines for outpatient opioid use after vaginal delivery.”
The study was partially supported by the University of Pittsburgh, the Pennsylvania Department of Human Services, the Building Interdisciplinary Research Careers in Women’s Health Program, and the National Institute on Drug Abuse. The researchers reported having no relevant financial disclosures.
A new study of pregnant Medicaid patients in Pennsylvania found that 12% filled prescriptions for opioids within 5 days of vaginal delivery, even though fewer than one-third of the women had pain-inducing conditions.
“Our study raises the question: Why is outpatient opioid use not rare after vaginal delivery?” lead author Marian Jarlenski, PhD, MPH, said in an interview. “Outpatient opioid prescriptions after a hospitalization may be one potential pathway to opioid use disorder. I hope the study will prompt some thought about why opioids are being prescribed for women after vaginal delivery and how to best manage postdelivery pain. This is especially important in areas of the country that have extraordinarily high rates of opioid use disorder.”
A total of 12% of the women (18,131) filled an outpatient prescription for an opioid with 5 days of giving birth. Of those, just 28.2% (5,110) had one or more conditions that cause an increased level of pain after delivery, such as bilateral tubal ligation, certain kinds of lacerations, and episiotomy.
During the first 5 days after birth, the most commonly prescribed opioid was oxycodone-acetaminophen (53.3%), followed by acetaminophen-codeine (20.5%), and hydrocodone-acetaminophen (19.6%).
Of the 18,131 women with an early postdelivery opioid prescription, 14% (2,592) filled at least one other opioid prescription within 6-60 days – that’s 1.6% of all the women in the study.
“On a positive note, we saw the supply of opioid prescriptions was generally short at 3-7 days,” Dr. Jarlenski said.
The researchers linked tobacco use and a mental health condition (both adjusted odds ratio 1.3; 95% confidence interval, 1.2-1.4) to a higher risk of filling a prescription without being diagnosed with a pain-causing disorder. They also found an association between substance use disorder (not related to opioids) and a higher risk of these types of prescriptions, but only for filling a second opioid prescription in the 6-60 day period (aOR 1.4; 95% CI, 1.2-1.6).
The researchers called for national guidelines regarding the use of opioids after birth.
“Several organizations have developed opioid prescribing guidelines for chronic and acute pain,” Dr. Jarlenski said. “These guidelines are necessary because the risk of opioid use disorder and subsequent overdose events is well established. Although delivery is the most common reason for hospitalization in the United States, there are no national guidelines for outpatient opioid use after vaginal delivery.”
The study was partially supported by the University of Pittsburgh, the Pennsylvania Department of Human Services, the Building Interdisciplinary Research Careers in Women’s Health Program, and the National Institute on Drug Abuse. The researchers reported having no relevant financial disclosures.
A new study of pregnant Medicaid patients in Pennsylvania found that 12% filled prescriptions for opioids within 5 days of vaginal delivery, even though fewer than one-third of the women had pain-inducing conditions.
“Our study raises the question: Why is outpatient opioid use not rare after vaginal delivery?” lead author Marian Jarlenski, PhD, MPH, said in an interview. “Outpatient opioid prescriptions after a hospitalization may be one potential pathway to opioid use disorder. I hope the study will prompt some thought about why opioids are being prescribed for women after vaginal delivery and how to best manage postdelivery pain. This is especially important in areas of the country that have extraordinarily high rates of opioid use disorder.”
A total of 12% of the women (18,131) filled an outpatient prescription for an opioid with 5 days of giving birth. Of those, just 28.2% (5,110) had one or more conditions that cause an increased level of pain after delivery, such as bilateral tubal ligation, certain kinds of lacerations, and episiotomy.
During the first 5 days after birth, the most commonly prescribed opioid was oxycodone-acetaminophen (53.3%), followed by acetaminophen-codeine (20.5%), and hydrocodone-acetaminophen (19.6%).
Of the 18,131 women with an early postdelivery opioid prescription, 14% (2,592) filled at least one other opioid prescription within 6-60 days – that’s 1.6% of all the women in the study.
“On a positive note, we saw the supply of opioid prescriptions was generally short at 3-7 days,” Dr. Jarlenski said.
The researchers linked tobacco use and a mental health condition (both adjusted odds ratio 1.3; 95% confidence interval, 1.2-1.4) to a higher risk of filling a prescription without being diagnosed with a pain-causing disorder. They also found an association between substance use disorder (not related to opioids) and a higher risk of these types of prescriptions, but only for filling a second opioid prescription in the 6-60 day period (aOR 1.4; 95% CI, 1.2-1.6).
The researchers called for national guidelines regarding the use of opioids after birth.
“Several organizations have developed opioid prescribing guidelines for chronic and acute pain,” Dr. Jarlenski said. “These guidelines are necessary because the risk of opioid use disorder and subsequent overdose events is well established. Although delivery is the most common reason for hospitalization in the United States, there are no national guidelines for outpatient opioid use after vaginal delivery.”
The study was partially supported by the University of Pittsburgh, the Pennsylvania Department of Human Services, the Building Interdisciplinary Research Careers in Women’s Health Program, and the National Institute on Drug Abuse. The researchers reported having no relevant financial disclosures.
FROM OBSTETRICS AND GYNECOLOGY
Key clinical point:
Major finding: A total of 12% of women on Medicaid filled opioid prescriptions within 5 days of delivery; just 28.2% of those patients had a pain-inducing condition.
Data source: A retrospective cohort study of 164,720 women enrolled in Medicaid in Pennsylvania who delivered live-born babies vaginally from 2008 to 2013.
Disclosures: The study is partially supported by the University of Pittsburgh, the Pennsylvania Department of Human Services, the Building Interdisciplinary Research Careers in Women’s Health Program, and the National Institute on Drug Abuse. The researchers reported having no relevant financial disclosures.
Updated guidelines offer insight into pediatric obesity
Among other things, the guidelines offer new insight into the role of genetics in childhood obesity, provide more extensive guidance regarding bariatric surgery in adolescents, and suggest that measurements of insulin concentrations aren’t useful barometers.
The guidelines also point to research gaps in several areas and call for more studies.
Why issue new guidelines now? “Eight years have passed since the last publication. We did a very thorough job, but there’s been an incredible amount of interest in child obesity, and more information is available,” lead author Dennis M. Styne, MD, professor of pediatrics and the Yocha Dehe endowed chair in pediatric endocrinology at the University of California at Davis, said in an interview. Indeed, recent years have produced hundreds of studies into pediatric obesity, he noted.
The 49-page guidelines are cosponsored by the European Society of Endocrinology and the Pediatric Endocrine Society (J Clin Endocrinol Metab. 2017 March;102[3]:1-49).
Pediatric obesity is of special interest to endocrinologists, Dr. Styne said. “While there’s interest from many specialists now, we are at the forefront of evaluation and treatment of complications like polycystic ovary syndrome, metabolic syndrome, dyslipidemia, and type 2 diabetes.”
The guidelines provide recommendations about a wide variety of obesity-related topics including screening, diagnosis, and treatment. The Endocrine Society commissioned two systematic reviews to support the guidelines: One examined longer randomized controlled trials into medication, surgery, lifestyle, and community-based intervention treatments. The other examined how changing body mass index levels corresponded to cardiometabolic changes.
Several updated areas in the guidelines should be of special interest to endocrinologists: guidance regarding the genetic causes of pediatric obesity, the use of weight-loss medication and surgery, and the roles of insulin tests and breast-feeding, according to Dr. Styne.
In regard to genetics, the guidelines note that research suggests 7% of patients with extreme pediatric obesity “may have rare chromosomal abnormalities and/or highly penetrant genetic mutations that drive their obesity. This percentage is likely to increase with newer methods for genetic testing.”
Dr. Styne calls this finding “remarkable.” As he put it, “we didn’t appreciate that so much in the past.”
The guidelines suggest genetic testing for patients who become obese before the age of 5 years, have a family history of extreme obesity, or show clinical signs of genetic obesity syndromes, especially extreme hyperphagia.
As for the most extreme treatments for the most obese children, the guidelines recommend against weight-loss medication outside of clinical trials and note that “increasing evidence” supports bariatric surgery in teens who haven’t been able to lose enough pounds through lifestyle modification. However, “the use of surgery requires experienced teams with resources for long-term follow-up.”
The guidelines also recommend against measuring serum insulin concentrations as part of pediatric screening for obesity. “A lot of people like to get insulin levels and think it tells them about the future of the child,” Dr. Styne said. “But it doesn’t work very well.”
In another area that reflects new information, the guidelines note that breast-feeding hasn’t been definitively shown to be effective in reducing childhood obesity. “The literature is contradictory,” Dr. Styne said, although he noted that breast-feeding still has many other benefits.
The guidelines point to research gaps in several areas, including the prevention and treatment of pediatric obesity. There’s also “uncharted territory” regarding how to “effectively transition to adult care, with continued necessary monitoring, support, and intervention.”
In regard to the best treatment programs, “we didn’t come up with an answer regarding overall effectiveness,” Dr. Styne said. “School- and community-based programs have promise, but I can’t give you the percentage of success.”
As for the overall picture of pediatric obesity in the United States, “we’re in a better situation than we were 8 years ago,” he said. “Everyone is talking about the problem, and when you talk to families, they’re more aware of it.”
Still, he said, the prevalence of obesity in kids is high, estimated at 17% of those aged 2-19 years in 2014. “That’s not a good place to be,” he said. “We still have to work harder.”
The Endocrine Society funded the guidelines. Dr. Styne reports ownership interests in Teva, Bristol Myers and Organovo. Other authors report various disclosures.
Among other things, the guidelines offer new insight into the role of genetics in childhood obesity, provide more extensive guidance regarding bariatric surgery in adolescents, and suggest that measurements of insulin concentrations aren’t useful barometers.
The guidelines also point to research gaps in several areas and call for more studies.
Why issue new guidelines now? “Eight years have passed since the last publication. We did a very thorough job, but there’s been an incredible amount of interest in child obesity, and more information is available,” lead author Dennis M. Styne, MD, professor of pediatrics and the Yocha Dehe endowed chair in pediatric endocrinology at the University of California at Davis, said in an interview. Indeed, recent years have produced hundreds of studies into pediatric obesity, he noted.
The 49-page guidelines are cosponsored by the European Society of Endocrinology and the Pediatric Endocrine Society (J Clin Endocrinol Metab. 2017 March;102[3]:1-49).
Pediatric obesity is of special interest to endocrinologists, Dr. Styne said. “While there’s interest from many specialists now, we are at the forefront of evaluation and treatment of complications like polycystic ovary syndrome, metabolic syndrome, dyslipidemia, and type 2 diabetes.”
The guidelines provide recommendations about a wide variety of obesity-related topics including screening, diagnosis, and treatment. The Endocrine Society commissioned two systematic reviews to support the guidelines: One examined longer randomized controlled trials into medication, surgery, lifestyle, and community-based intervention treatments. The other examined how changing body mass index levels corresponded to cardiometabolic changes.
Several updated areas in the guidelines should be of special interest to endocrinologists: guidance regarding the genetic causes of pediatric obesity, the use of weight-loss medication and surgery, and the roles of insulin tests and breast-feeding, according to Dr. Styne.
In regard to genetics, the guidelines note that research suggests 7% of patients with extreme pediatric obesity “may have rare chromosomal abnormalities and/or highly penetrant genetic mutations that drive their obesity. This percentage is likely to increase with newer methods for genetic testing.”
Dr. Styne calls this finding “remarkable.” As he put it, “we didn’t appreciate that so much in the past.”
The guidelines suggest genetic testing for patients who become obese before the age of 5 years, have a family history of extreme obesity, or show clinical signs of genetic obesity syndromes, especially extreme hyperphagia.
As for the most extreme treatments for the most obese children, the guidelines recommend against weight-loss medication outside of clinical trials and note that “increasing evidence” supports bariatric surgery in teens who haven’t been able to lose enough pounds through lifestyle modification. However, “the use of surgery requires experienced teams with resources for long-term follow-up.”
The guidelines also recommend against measuring serum insulin concentrations as part of pediatric screening for obesity. “A lot of people like to get insulin levels and think it tells them about the future of the child,” Dr. Styne said. “But it doesn’t work very well.”
In another area that reflects new information, the guidelines note that breast-feeding hasn’t been definitively shown to be effective in reducing childhood obesity. “The literature is contradictory,” Dr. Styne said, although he noted that breast-feeding still has many other benefits.
The guidelines point to research gaps in several areas, including the prevention and treatment of pediatric obesity. There’s also “uncharted territory” regarding how to “effectively transition to adult care, with continued necessary monitoring, support, and intervention.”
In regard to the best treatment programs, “we didn’t come up with an answer regarding overall effectiveness,” Dr. Styne said. “School- and community-based programs have promise, but I can’t give you the percentage of success.”
As for the overall picture of pediatric obesity in the United States, “we’re in a better situation than we were 8 years ago,” he said. “Everyone is talking about the problem, and when you talk to families, they’re more aware of it.”
Still, he said, the prevalence of obesity in kids is high, estimated at 17% of those aged 2-19 years in 2014. “That’s not a good place to be,” he said. “We still have to work harder.”
The Endocrine Society funded the guidelines. Dr. Styne reports ownership interests in Teva, Bristol Myers and Organovo. Other authors report various disclosures.
Among other things, the guidelines offer new insight into the role of genetics in childhood obesity, provide more extensive guidance regarding bariatric surgery in adolescents, and suggest that measurements of insulin concentrations aren’t useful barometers.
The guidelines also point to research gaps in several areas and call for more studies.
Why issue new guidelines now? “Eight years have passed since the last publication. We did a very thorough job, but there’s been an incredible amount of interest in child obesity, and more information is available,” lead author Dennis M. Styne, MD, professor of pediatrics and the Yocha Dehe endowed chair in pediatric endocrinology at the University of California at Davis, said in an interview. Indeed, recent years have produced hundreds of studies into pediatric obesity, he noted.
The 49-page guidelines are cosponsored by the European Society of Endocrinology and the Pediatric Endocrine Society (J Clin Endocrinol Metab. 2017 March;102[3]:1-49).
Pediatric obesity is of special interest to endocrinologists, Dr. Styne said. “While there’s interest from many specialists now, we are at the forefront of evaluation and treatment of complications like polycystic ovary syndrome, metabolic syndrome, dyslipidemia, and type 2 diabetes.”
The guidelines provide recommendations about a wide variety of obesity-related topics including screening, diagnosis, and treatment. The Endocrine Society commissioned two systematic reviews to support the guidelines: One examined longer randomized controlled trials into medication, surgery, lifestyle, and community-based intervention treatments. The other examined how changing body mass index levels corresponded to cardiometabolic changes.
Several updated areas in the guidelines should be of special interest to endocrinologists: guidance regarding the genetic causes of pediatric obesity, the use of weight-loss medication and surgery, and the roles of insulin tests and breast-feeding, according to Dr. Styne.
In regard to genetics, the guidelines note that research suggests 7% of patients with extreme pediatric obesity “may have rare chromosomal abnormalities and/or highly penetrant genetic mutations that drive their obesity. This percentage is likely to increase with newer methods for genetic testing.”
Dr. Styne calls this finding “remarkable.” As he put it, “we didn’t appreciate that so much in the past.”
The guidelines suggest genetic testing for patients who become obese before the age of 5 years, have a family history of extreme obesity, or show clinical signs of genetic obesity syndromes, especially extreme hyperphagia.
As for the most extreme treatments for the most obese children, the guidelines recommend against weight-loss medication outside of clinical trials and note that “increasing evidence” supports bariatric surgery in teens who haven’t been able to lose enough pounds through lifestyle modification. However, “the use of surgery requires experienced teams with resources for long-term follow-up.”
The guidelines also recommend against measuring serum insulin concentrations as part of pediatric screening for obesity. “A lot of people like to get insulin levels and think it tells them about the future of the child,” Dr. Styne said. “But it doesn’t work very well.”
In another area that reflects new information, the guidelines note that breast-feeding hasn’t been definitively shown to be effective in reducing childhood obesity. “The literature is contradictory,” Dr. Styne said, although he noted that breast-feeding still has many other benefits.
The guidelines point to research gaps in several areas, including the prevention and treatment of pediatric obesity. There’s also “uncharted territory” regarding how to “effectively transition to adult care, with continued necessary monitoring, support, and intervention.”
In regard to the best treatment programs, “we didn’t come up with an answer regarding overall effectiveness,” Dr. Styne said. “School- and community-based programs have promise, but I can’t give you the percentage of success.”
As for the overall picture of pediatric obesity in the United States, “we’re in a better situation than we were 8 years ago,” he said. “Everyone is talking about the problem, and when you talk to families, they’re more aware of it.”
Still, he said, the prevalence of obesity in kids is high, estimated at 17% of those aged 2-19 years in 2014. “That’s not a good place to be,” he said. “We still have to work harder.”
The Endocrine Society funded the guidelines. Dr. Styne reports ownership interests in Teva, Bristol Myers and Organovo. Other authors report various disclosures.
Abatacept efficacy differs in trials of giant cell and Takayasu’s arteritis
A pair of new studies offer mixed results regarding the use of the rheumatoid arthritis drug abatacept to treat two forms of large-vessel vasculitis: It appears to help patients with giant cell arteritis but not those with the rarer Takayasu’s arteritis.
“The results from the GCA [giant cell arteritis] study found that treatment with abatacept [Orencia] combined with prednisone resulted in a lower rate of relapse than treatment with prednisone alone,” said Carol A. Langford, MD, lead author of both studies, which were conducted by the Vasculitis Clinical Research Consortium. She is chair in rheumatic and immunologic diseases and director of the Center for Vasculitis Care and Research at the Cleveland Clinic.
Both studies appear online Jan. 30 in Arthritis & Rheumatology.
“GCA is the most common form of vasculitis with an estimated incidence of 19.8 per 100,000,” she said. “It occurs in people over the age of 50 with the average age of onset in the 70s.” Women are most affected by a 2:1 ratio.
She said the disease affects the cranium (causing headaches, scalp tenderness, and a risk of blindness) and causes signs of systemic inflammation.
“Almost one-third of patients with GCA can have large vessel involvement that specifically include thoracic aortic aneurysms and stenosis of the cervical and subclavian arteries,” she said. Fatal thoracic aneurysms are possible, she said, but “studies have shown that in GCA overall, while short-term mortality may be increased, long-term survival is similar to the age-matched general population.”
TAK is much rarer, she said, affecting 3-9 people per 1,000,000. “TAK has an average age of onset in the 20s with an even stronger female predisposition of up to 9:1.”
The condition affects the aorta, its main branches, and pulmonary arteries, she said, “Some of the more frequent vascular symptoms/signs can include extremity claudication, hypertension, chest pain, and features associated with cerebral hypoperfusion. TAK is associated with substantial morbidity which is influenced by a low rate of sustained remission in 28%-50% of patients. Up to 47% of patients experience permanent disability, which has a significant impact on this young population.”
The mortality from TAK is unclear, she said.
Glucocorticoids are the main treatment for both GCA and TAK, Dr. Langford said, but “while glucocorticoids effectively control disease, they do not prevent relapse and they are associated with significant toxicity.”
For GCA, methotrexate has shown a mild benefit at best, she said, while two studies show promise for tocilizumab (Actemra). As for TAK, she said doctors often turn to the use of immunosuppressants and tumor necrosis factor inhibitors, although their use is based on retrospective studies and small, open-label trials.
Dr. Langford and her colleagues launched the two randomized, double-blind, placebo-controlled, multicenter studies in parallel with the same protocols.
In the GCA trial (doi: 10.1002/art.40044), researchers enrolled 49 patients with newly diagnosed GCA or disease that had relapsed within the 2 prior months to prednisone 40-60 mg/day followed by a standardized tapering schedule plus abatacept 10 mg/kg IV on days 1, 15, 29, and week 8. At 12 weeks, 8 patients had withdrawn, relapsed, or were not in remission, and so 41 were randomized to receive placebo or monthly abatacept until they met criteria for early termination or 12 months had passed after the last patient was enrolled. At the time of the randomization at 12 weeks, all patients were taking prednisone 20 mg/day, which was tapered until discontinuation at week 28.
At 12 months, 48% of those who took abatacept survived without relapse, compared with 31% of those who took placebo (P = .049), and the median remission period was longer for abatacept (9.9 months) than placebo (3.9 months; P = .023).
“This difference between groups is clinically meaningful to patients with GCA, corresponding to a prolonged duration of remission during which time they are not exposed to glucocorticoids and their potential toxicities that can impact quality of life,” Dr. Langford said.
No patients died during the trial, and 23 serious adverse events were reported in 15 patients. The frequency and severity of adverse events and infections didn’t differ between the treatment and placebo groups.
The drug may work in GCA patients by blocking T-cell activation, Dr. Langford said. Physicians could consider the drug in clinical practice, she said, although more research is needed to understand the long-term effects of using the medication.
In the other study (doi: 10.1002/art.40037), researchers used the same protocol to treat 34 patients with TAK; 26 reached the 12-week midpoint and were randomized to placebo or continuing abatacept on a monthly basis.
At 12 months, the relapse-free rate was 22% for the abatacept group and 40% for the placebo group (P = .853). The median duration of remission was similar for the groups at 5.5 months for abatacept and 5.7 months for placebo (P = .125).
The researchers reported no difference in frequency or severity of adverse events such as infection.
Based on the study results, Dr. Langford said abatacept is not appropriate in clinical practice to treat TAK.
The National Institute of Arthritis and Musculoskeletal and Skin Diseases funded the studies, and Bristol-Myers Squibb, which markets abatacept, provided the drug. Dr. Langford disclosed receiving research grants from Bristol-Myers Squibb, Genentech, and GlaxoSmithKline.
A pair of new studies offer mixed results regarding the use of the rheumatoid arthritis drug abatacept to treat two forms of large-vessel vasculitis: It appears to help patients with giant cell arteritis but not those with the rarer Takayasu’s arteritis.
“The results from the GCA [giant cell arteritis] study found that treatment with abatacept [Orencia] combined with prednisone resulted in a lower rate of relapse than treatment with prednisone alone,” said Carol A. Langford, MD, lead author of both studies, which were conducted by the Vasculitis Clinical Research Consortium. She is chair in rheumatic and immunologic diseases and director of the Center for Vasculitis Care and Research at the Cleveland Clinic.
Both studies appear online Jan. 30 in Arthritis & Rheumatology.
“GCA is the most common form of vasculitis with an estimated incidence of 19.8 per 100,000,” she said. “It occurs in people over the age of 50 with the average age of onset in the 70s.” Women are most affected by a 2:1 ratio.
She said the disease affects the cranium (causing headaches, scalp tenderness, and a risk of blindness) and causes signs of systemic inflammation.
“Almost one-third of patients with GCA can have large vessel involvement that specifically include thoracic aortic aneurysms and stenosis of the cervical and subclavian arteries,” she said. Fatal thoracic aneurysms are possible, she said, but “studies have shown that in GCA overall, while short-term mortality may be increased, long-term survival is similar to the age-matched general population.”
TAK is much rarer, she said, affecting 3-9 people per 1,000,000. “TAK has an average age of onset in the 20s with an even stronger female predisposition of up to 9:1.”
The condition affects the aorta, its main branches, and pulmonary arteries, she said, “Some of the more frequent vascular symptoms/signs can include extremity claudication, hypertension, chest pain, and features associated with cerebral hypoperfusion. TAK is associated with substantial morbidity which is influenced by a low rate of sustained remission in 28%-50% of patients. Up to 47% of patients experience permanent disability, which has a significant impact on this young population.”
The mortality from TAK is unclear, she said.
Glucocorticoids are the main treatment for both GCA and TAK, Dr. Langford said, but “while glucocorticoids effectively control disease, they do not prevent relapse and they are associated with significant toxicity.”
For GCA, methotrexate has shown a mild benefit at best, she said, while two studies show promise for tocilizumab (Actemra). As for TAK, she said doctors often turn to the use of immunosuppressants and tumor necrosis factor inhibitors, although their use is based on retrospective studies and small, open-label trials.
Dr. Langford and her colleagues launched the two randomized, double-blind, placebo-controlled, multicenter studies in parallel with the same protocols.
In the GCA trial (doi: 10.1002/art.40044), researchers enrolled 49 patients with newly diagnosed GCA or disease that had relapsed within the 2 prior months to prednisone 40-60 mg/day followed by a standardized tapering schedule plus abatacept 10 mg/kg IV on days 1, 15, 29, and week 8. At 12 weeks, 8 patients had withdrawn, relapsed, or were not in remission, and so 41 were randomized to receive placebo or monthly abatacept until they met criteria for early termination or 12 months had passed after the last patient was enrolled. At the time of the randomization at 12 weeks, all patients were taking prednisone 20 mg/day, which was tapered until discontinuation at week 28.
At 12 months, 48% of those who took abatacept survived without relapse, compared with 31% of those who took placebo (P = .049), and the median remission period was longer for abatacept (9.9 months) than placebo (3.9 months; P = .023).
“This difference between groups is clinically meaningful to patients with GCA, corresponding to a prolonged duration of remission during which time they are not exposed to glucocorticoids and their potential toxicities that can impact quality of life,” Dr. Langford said.
No patients died during the trial, and 23 serious adverse events were reported in 15 patients. The frequency and severity of adverse events and infections didn’t differ between the treatment and placebo groups.
The drug may work in GCA patients by blocking T-cell activation, Dr. Langford said. Physicians could consider the drug in clinical practice, she said, although more research is needed to understand the long-term effects of using the medication.
In the other study (doi: 10.1002/art.40037), researchers used the same protocol to treat 34 patients with TAK; 26 reached the 12-week midpoint and were randomized to placebo or continuing abatacept on a monthly basis.
At 12 months, the relapse-free rate was 22% for the abatacept group and 40% for the placebo group (P = .853). The median duration of remission was similar for the groups at 5.5 months for abatacept and 5.7 months for placebo (P = .125).
The researchers reported no difference in frequency or severity of adverse events such as infection.
Based on the study results, Dr. Langford said abatacept is not appropriate in clinical practice to treat TAK.
The National Institute of Arthritis and Musculoskeletal and Skin Diseases funded the studies, and Bristol-Myers Squibb, which markets abatacept, provided the drug. Dr. Langford disclosed receiving research grants from Bristol-Myers Squibb, Genentech, and GlaxoSmithKline.
A pair of new studies offer mixed results regarding the use of the rheumatoid arthritis drug abatacept to treat two forms of large-vessel vasculitis: It appears to help patients with giant cell arteritis but not those with the rarer Takayasu’s arteritis.
“The results from the GCA [giant cell arteritis] study found that treatment with abatacept [Orencia] combined with prednisone resulted in a lower rate of relapse than treatment with prednisone alone,” said Carol A. Langford, MD, lead author of both studies, which were conducted by the Vasculitis Clinical Research Consortium. She is chair in rheumatic and immunologic diseases and director of the Center for Vasculitis Care and Research at the Cleveland Clinic.
Both studies appear online Jan. 30 in Arthritis & Rheumatology.
“GCA is the most common form of vasculitis with an estimated incidence of 19.8 per 100,000,” she said. “It occurs in people over the age of 50 with the average age of onset in the 70s.” Women are most affected by a 2:1 ratio.
She said the disease affects the cranium (causing headaches, scalp tenderness, and a risk of blindness) and causes signs of systemic inflammation.
“Almost one-third of patients with GCA can have large vessel involvement that specifically include thoracic aortic aneurysms and stenosis of the cervical and subclavian arteries,” she said. Fatal thoracic aneurysms are possible, she said, but “studies have shown that in GCA overall, while short-term mortality may be increased, long-term survival is similar to the age-matched general population.”
TAK is much rarer, she said, affecting 3-9 people per 1,000,000. “TAK has an average age of onset in the 20s with an even stronger female predisposition of up to 9:1.”
The condition affects the aorta, its main branches, and pulmonary arteries, she said, “Some of the more frequent vascular symptoms/signs can include extremity claudication, hypertension, chest pain, and features associated with cerebral hypoperfusion. TAK is associated with substantial morbidity which is influenced by a low rate of sustained remission in 28%-50% of patients. Up to 47% of patients experience permanent disability, which has a significant impact on this young population.”
The mortality from TAK is unclear, she said.
Glucocorticoids are the main treatment for both GCA and TAK, Dr. Langford said, but “while glucocorticoids effectively control disease, they do not prevent relapse and they are associated with significant toxicity.”
For GCA, methotrexate has shown a mild benefit at best, she said, while two studies show promise for tocilizumab (Actemra). As for TAK, she said doctors often turn to the use of immunosuppressants and tumor necrosis factor inhibitors, although their use is based on retrospective studies and small, open-label trials.
Dr. Langford and her colleagues launched the two randomized, double-blind, placebo-controlled, multicenter studies in parallel with the same protocols.
In the GCA trial (doi: 10.1002/art.40044), researchers enrolled 49 patients with newly diagnosed GCA or disease that had relapsed within the 2 prior months to prednisone 40-60 mg/day followed by a standardized tapering schedule plus abatacept 10 mg/kg IV on days 1, 15, 29, and week 8. At 12 weeks, 8 patients had withdrawn, relapsed, or were not in remission, and so 41 were randomized to receive placebo or monthly abatacept until they met criteria for early termination or 12 months had passed after the last patient was enrolled. At the time of the randomization at 12 weeks, all patients were taking prednisone 20 mg/day, which was tapered until discontinuation at week 28.
At 12 months, 48% of those who took abatacept survived without relapse, compared with 31% of those who took placebo (P = .049), and the median remission period was longer for abatacept (9.9 months) than placebo (3.9 months; P = .023).
“This difference between groups is clinically meaningful to patients with GCA, corresponding to a prolonged duration of remission during which time they are not exposed to glucocorticoids and their potential toxicities that can impact quality of life,” Dr. Langford said.
No patients died during the trial, and 23 serious adverse events were reported in 15 patients. The frequency and severity of adverse events and infections didn’t differ between the treatment and placebo groups.
The drug may work in GCA patients by blocking T-cell activation, Dr. Langford said. Physicians could consider the drug in clinical practice, she said, although more research is needed to understand the long-term effects of using the medication.
In the other study (doi: 10.1002/art.40037), researchers used the same protocol to treat 34 patients with TAK; 26 reached the 12-week midpoint and were randomized to placebo or continuing abatacept on a monthly basis.
At 12 months, the relapse-free rate was 22% for the abatacept group and 40% for the placebo group (P = .853). The median duration of remission was similar for the groups at 5.5 months for abatacept and 5.7 months for placebo (P = .125).
The researchers reported no difference in frequency or severity of adverse events such as infection.
Based on the study results, Dr. Langford said abatacept is not appropriate in clinical practice to treat TAK.
The National Institute of Arthritis and Musculoskeletal and Skin Diseases funded the studies, and Bristol-Myers Squibb, which markets abatacept, provided the drug. Dr. Langford disclosed receiving research grants from Bristol-Myers Squibb, Genentech, and GlaxoSmithKline.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point:
Major finding: At 12 months, 48% of GCA patients who took abatacept survived without relapse, compared with 31% of those who took placebo (P = .049), and the median remission period was longer for abatacept (9.9 months) than placebo (3.9 months; P = .023) For TAK patients, the relapse-free rates were 22% for the abatacept group and 40% for the placebo group (P = .853). The median duration of remission was similar for the groups at 5.5 months for abatacept and 5.7 months for placebo (P = .125).
Data source: Two randomized, double-blinded, placebo-controlled, multicenter trials with identical protocols of prednisone plus abatacept 10 mg/kg IV on days 1, 15, 29, and week 8, then randomization to placebo or monthly abatacept until patients met criteria for early termination or 12 months passed after the last patient was enrolled.
Disclosures: The National Institute of Arthritis and Musculoskeletal and Skin Diseases funded the studies, and Bristol-Myers Squibb, which markets abatacept, provided the drug. Dr. Langford disclosed receiving research grants from Bristol-Myers Squibb, Genentech, and GlaxoSmithKline.
Immune-suppressing drugs in IBD linked to higher skin cancer rates
In another sign that immune-suppressing drugs may cause skin cancer, a new Irish study links immunomodulator use in younger patients with inflammatory bowel disease (IBD) to higher rates of nonmelanoma skin cancer (NMSC).
The 19-year study lacks information about medication doses or duration, and it doesn’t confirm a cause-and-effect link. Still, researchers recommend that all patients with IBD be urged to comply with skin cancer prevention guidelines.
As the study notes, previous research has linked immunosuppression – such as that in transplant patients and those with AIDS and lymphoma – to higher rates of NMSC.
Studies have also linked IBD to higher rates of NMSC even before the age of 50, possibly as the result of immune system dysfunction and exposure to immunomodulators, especially thiopurines. The risk of tumor necrosis factor–alpha (TNF-alpha) inhibitors, the study says, is less clear.
To better understand the risk of immunomodulators, researchers led by Julianne Clowry, MBBCh, of St Vincent’s University Hospital in Dublin tracked 2,053 IBD patients at a tertiary adult hospital from 1994 to 2013.
The median age at IBD diagnosis was 31 with a median of 19.6 years of illness, and the patients had both Crohn’s disease (41%) and ulcerative colitis (59%). Fifty-seven percent of patients had taken immunomodulating medication, although the database used didn’t disclose details about dose or duration, and 43% had not.
The study findings appeared Jan. 3 in the Journal of the European Academy of Dermatology and Venereology (doi: 10.1111/jdv.14105).
NMSC was diagnosed in 1.7% of the entire cohort, 1.4% of patients who’d taken immunosuppressants, and 1.9% of those who had not.
Older ages may explain the higher rate in those who didn’t take the medications. The researchers found that the standardized incidence ratio for the patients who took immunomodulators overall was 1.76 [confidence interval, 1.0-2.7], compared with a matched general population cohort, while the ratio was not considered significant among the nonimmunosuppressed [1.07; CI, 0.6-1.6].
The study links use of thiopurines alone and use of both thiopurines and TNF-alpha inhibitors to higher rates of NMSC [odds ratio, 5.26; 95% CI, 2.15-12.93; P less than .001, and OR: 6.45; 95% CI, 2.69-15.95; P less than .001, respectively].
The researchers note that 82% of those who had taken a TNF-alpha inhibitor also took a thiopurine at some point.
The study says the “relatively high” standardized incident ratios are worrisome amid more use of dual immunomodulators and higher IBD rates in kids and younger adults. But the medications are “vital,” the study says, and the researchers suggest “targeted dermatology referrals for IBD patients, particularly those exposed to dual immunomodulatory therapy from an early age.”
The study authors disclose no source of funding and report no relevant disclosures.
In another sign that immune-suppressing drugs may cause skin cancer, a new Irish study links immunomodulator use in younger patients with inflammatory bowel disease (IBD) to higher rates of nonmelanoma skin cancer (NMSC).
The 19-year study lacks information about medication doses or duration, and it doesn’t confirm a cause-and-effect link. Still, researchers recommend that all patients with IBD be urged to comply with skin cancer prevention guidelines.
As the study notes, previous research has linked immunosuppression – such as that in transplant patients and those with AIDS and lymphoma – to higher rates of NMSC.
Studies have also linked IBD to higher rates of NMSC even before the age of 50, possibly as the result of immune system dysfunction and exposure to immunomodulators, especially thiopurines. The risk of tumor necrosis factor–alpha (TNF-alpha) inhibitors, the study says, is less clear.
To better understand the risk of immunomodulators, researchers led by Julianne Clowry, MBBCh, of St Vincent’s University Hospital in Dublin tracked 2,053 IBD patients at a tertiary adult hospital from 1994 to 2013.
The median age at IBD diagnosis was 31 with a median of 19.6 years of illness, and the patients had both Crohn’s disease (41%) and ulcerative colitis (59%). Fifty-seven percent of patients had taken immunomodulating medication, although the database used didn’t disclose details about dose or duration, and 43% had not.
The study findings appeared Jan. 3 in the Journal of the European Academy of Dermatology and Venereology (doi: 10.1111/jdv.14105).
NMSC was diagnosed in 1.7% of the entire cohort, 1.4% of patients who’d taken immunosuppressants, and 1.9% of those who had not.
Older ages may explain the higher rate in those who didn’t take the medications. The researchers found that the standardized incidence ratio for the patients who took immunomodulators overall was 1.76 [confidence interval, 1.0-2.7], compared with a matched general population cohort, while the ratio was not considered significant among the nonimmunosuppressed [1.07; CI, 0.6-1.6].
The study links use of thiopurines alone and use of both thiopurines and TNF-alpha inhibitors to higher rates of NMSC [odds ratio, 5.26; 95% CI, 2.15-12.93; P less than .001, and OR: 6.45; 95% CI, 2.69-15.95; P less than .001, respectively].
The researchers note that 82% of those who had taken a TNF-alpha inhibitor also took a thiopurine at some point.
The study says the “relatively high” standardized incident ratios are worrisome amid more use of dual immunomodulators and higher IBD rates in kids and younger adults. But the medications are “vital,” the study says, and the researchers suggest “targeted dermatology referrals for IBD patients, particularly those exposed to dual immunomodulatory therapy from an early age.”
The study authors disclose no source of funding and report no relevant disclosures.
In another sign that immune-suppressing drugs may cause skin cancer, a new Irish study links immunomodulator use in younger patients with inflammatory bowel disease (IBD) to higher rates of nonmelanoma skin cancer (NMSC).
The 19-year study lacks information about medication doses or duration, and it doesn’t confirm a cause-and-effect link. Still, researchers recommend that all patients with IBD be urged to comply with skin cancer prevention guidelines.
As the study notes, previous research has linked immunosuppression – such as that in transplant patients and those with AIDS and lymphoma – to higher rates of NMSC.
Studies have also linked IBD to higher rates of NMSC even before the age of 50, possibly as the result of immune system dysfunction and exposure to immunomodulators, especially thiopurines. The risk of tumor necrosis factor–alpha (TNF-alpha) inhibitors, the study says, is less clear.
To better understand the risk of immunomodulators, researchers led by Julianne Clowry, MBBCh, of St Vincent’s University Hospital in Dublin tracked 2,053 IBD patients at a tertiary adult hospital from 1994 to 2013.
The median age at IBD diagnosis was 31 with a median of 19.6 years of illness, and the patients had both Crohn’s disease (41%) and ulcerative colitis (59%). Fifty-seven percent of patients had taken immunomodulating medication, although the database used didn’t disclose details about dose or duration, and 43% had not.
The study findings appeared Jan. 3 in the Journal of the European Academy of Dermatology and Venereology (doi: 10.1111/jdv.14105).
NMSC was diagnosed in 1.7% of the entire cohort, 1.4% of patients who’d taken immunosuppressants, and 1.9% of those who had not.
Older ages may explain the higher rate in those who didn’t take the medications. The researchers found that the standardized incidence ratio for the patients who took immunomodulators overall was 1.76 [confidence interval, 1.0-2.7], compared with a matched general population cohort, while the ratio was not considered significant among the nonimmunosuppressed [1.07; CI, 0.6-1.6].
The study links use of thiopurines alone and use of both thiopurines and TNF-alpha inhibitors to higher rates of NMSC [odds ratio, 5.26; 95% CI, 2.15-12.93; P less than .001, and OR: 6.45; 95% CI, 2.69-15.95; P less than .001, respectively].
The researchers note that 82% of those who had taken a TNF-alpha inhibitor also took a thiopurine at some point.
The study says the “relatively high” standardized incident ratios are worrisome amid more use of dual immunomodulators and higher IBD rates in kids and younger adults. But the medications are “vital,” the study says, and the researchers suggest “targeted dermatology referrals for IBD patients, particularly those exposed to dual immunomodulatory therapy from an early age.”
The study authors disclose no source of funding and report no relevant disclosures.
Key clinical point: Younger inflammatory bowel disease (IBD) patients who’ve taken immunomodulating drugs have higher rates of nonmelanoma skin cancer (NMSC).
Major finding: IBD patients who took thiopurines alone and both thiopurines and TNF-alpha inhibitors had higher rates of NMSC [OR, 5.26; 95% CI, 2.15-12.93; P less than .001, and OR, 6.45; 95% CI, 2.69-15.95; P less than .001, respectively], compared with an age-matched general population cohort.
Data source: Retrospective single-center cohort study over 19 years of 2,053 IBD patients with Crohn’s disease (41%) and ulcerative colitis (59%); 57% had taken immunomodulating medications.
Disclosures: The study authors disclose no source of funding and report no relevant disclosures.
Unique microbiota mix found in guts of T1D patients
potentially offering early insight into possible links between the disease and gut germs.
The findings by an Italian team don’t confirm any connection between bacteria in the digestive system and diabetes. Still, “this study is probably the best example to date in the literature of inflammatory events happening in the gut that are correlated with type 1 diabetes,” said Aleksandar Kostic, PhD, of the department of microbiology and immunobiology at Harvard Medical School, Boston, who conducts similar research.
At issue: What role, if any, does the gut play in the development of type 1 diabetes (T1D)? Scientists already believe that the gut microbiome directly affects metabolism and the development of type 2 diabetes, according to Dr. Kostic. But T1D is an autoimmune disease, not a metabolic one, he said, “and the mechanisms are very different. For type 1, we don’t know a whole lot. We’re in the very early days.”
Still, “there’s a theory that inflammatory stimulus in the gut that is somehow partially responsible for causing T1D. The idea is that the microbiome is less diverse, which means that it loses its integrity in some way and loses the ability to crowd out inflammatory organisms,” he said in an interview.
For the new study, researchers led by scientists at Milan’s IRCCS San Raffaele Scientific Institute measured inflammation and the microbiome in the duodenal mucosa of 19 patients with T1D, 19 with celiac disease, and 16 healthy controls. They reported their findings online Jan. 19 (J Clin Endocrinol Metab. 2017. doi: 10.1210/jc.2016-3222).
The researchers found a unique inflammation profile through an analysis of gene expression in the patients with T1D. They called it a “peculiar signature” that’s notable for increased numbers of infiltration from the monocyte/macrophage lineage.
“In T1D patients, we didn’t observe any correlation between gene expression and [hemoglobin A1c] level, duration of diabetes, presence of secondary complications or the reason that led to endoscopy, indicating that gene expression was not influenced by these variables,” the researchers write.
They also found a “specific microbiota composition” featuring a reduction in the role of Proteobacteria and an increase in Firmicutes; this was unique to the T1D patients. Bacteroidetes “showed a trend to reduction” in both T1D and celiac patients compared to the controls.
“The expression of genes specific for T1D inflammation was associated with the abundance of specific bacteria in duodenum,” the researchers added.
Elena Barengolts, MD, of the division of endocrinology, diabetes, and metabolism at the University of Illinois, Chicago, who’s familiar with the study, said it appears to be valid. However, the methods used have limited powers to define specific types of bacteria, making it difficult to know if the germs in question are “bad” or “good,” she said in an interview.
For his part, Dr. Kostic said the findings are “really neat” and consistent with previous findings regarding the role of the gut microbiome and T1D. He pointed to a study he led that found less-diverse microbiomes in the guts of Finnish infants with T1D (Cell Host Microbe. 2015 Feb 11;17[2]:260-73).
As a result, the gut microbiome is “functionally capable of doing fewer things, and the community gets overrun by certain pathogens,” he said. “We saw that a lot of organisms were capable of promoting inflammation in the gut.”
Dr. Kostic, Dr. Barengolts, and the study authors report no relevant disclosures.
potentially offering early insight into possible links between the disease and gut germs.
The findings by an Italian team don’t confirm any connection between bacteria in the digestive system and diabetes. Still, “this study is probably the best example to date in the literature of inflammatory events happening in the gut that are correlated with type 1 diabetes,” said Aleksandar Kostic, PhD, of the department of microbiology and immunobiology at Harvard Medical School, Boston, who conducts similar research.
At issue: What role, if any, does the gut play in the development of type 1 diabetes (T1D)? Scientists already believe that the gut microbiome directly affects metabolism and the development of type 2 diabetes, according to Dr. Kostic. But T1D is an autoimmune disease, not a metabolic one, he said, “and the mechanisms are very different. For type 1, we don’t know a whole lot. We’re in the very early days.”
Still, “there’s a theory that inflammatory stimulus in the gut that is somehow partially responsible for causing T1D. The idea is that the microbiome is less diverse, which means that it loses its integrity in some way and loses the ability to crowd out inflammatory organisms,” he said in an interview.
For the new study, researchers led by scientists at Milan’s IRCCS San Raffaele Scientific Institute measured inflammation and the microbiome in the duodenal mucosa of 19 patients with T1D, 19 with celiac disease, and 16 healthy controls. They reported their findings online Jan. 19 (J Clin Endocrinol Metab. 2017. doi: 10.1210/jc.2016-3222).
The researchers found a unique inflammation profile through an analysis of gene expression in the patients with T1D. They called it a “peculiar signature” that’s notable for increased numbers of infiltration from the monocyte/macrophage lineage.
“In T1D patients, we didn’t observe any correlation between gene expression and [hemoglobin A1c] level, duration of diabetes, presence of secondary complications or the reason that led to endoscopy, indicating that gene expression was not influenced by these variables,” the researchers write.
They also found a “specific microbiota composition” featuring a reduction in the role of Proteobacteria and an increase in Firmicutes; this was unique to the T1D patients. Bacteroidetes “showed a trend to reduction” in both T1D and celiac patients compared to the controls.
“The expression of genes specific for T1D inflammation was associated with the abundance of specific bacteria in duodenum,” the researchers added.
Elena Barengolts, MD, of the division of endocrinology, diabetes, and metabolism at the University of Illinois, Chicago, who’s familiar with the study, said it appears to be valid. However, the methods used have limited powers to define specific types of bacteria, making it difficult to know if the germs in question are “bad” or “good,” she said in an interview.
For his part, Dr. Kostic said the findings are “really neat” and consistent with previous findings regarding the role of the gut microbiome and T1D. He pointed to a study he led that found less-diverse microbiomes in the guts of Finnish infants with T1D (Cell Host Microbe. 2015 Feb 11;17[2]:260-73).
As a result, the gut microbiome is “functionally capable of doing fewer things, and the community gets overrun by certain pathogens,” he said. “We saw that a lot of organisms were capable of promoting inflammation in the gut.”
Dr. Kostic, Dr. Barengolts, and the study authors report no relevant disclosures.
potentially offering early insight into possible links between the disease and gut germs.
The findings by an Italian team don’t confirm any connection between bacteria in the digestive system and diabetes. Still, “this study is probably the best example to date in the literature of inflammatory events happening in the gut that are correlated with type 1 diabetes,” said Aleksandar Kostic, PhD, of the department of microbiology and immunobiology at Harvard Medical School, Boston, who conducts similar research.
At issue: What role, if any, does the gut play in the development of type 1 diabetes (T1D)? Scientists already believe that the gut microbiome directly affects metabolism and the development of type 2 diabetes, according to Dr. Kostic. But T1D is an autoimmune disease, not a metabolic one, he said, “and the mechanisms are very different. For type 1, we don’t know a whole lot. We’re in the very early days.”
Still, “there’s a theory that inflammatory stimulus in the gut that is somehow partially responsible for causing T1D. The idea is that the microbiome is less diverse, which means that it loses its integrity in some way and loses the ability to crowd out inflammatory organisms,” he said in an interview.
For the new study, researchers led by scientists at Milan’s IRCCS San Raffaele Scientific Institute measured inflammation and the microbiome in the duodenal mucosa of 19 patients with T1D, 19 with celiac disease, and 16 healthy controls. They reported their findings online Jan. 19 (J Clin Endocrinol Metab. 2017. doi: 10.1210/jc.2016-3222).
The researchers found a unique inflammation profile through an analysis of gene expression in the patients with T1D. They called it a “peculiar signature” that’s notable for increased numbers of infiltration from the monocyte/macrophage lineage.
“In T1D patients, we didn’t observe any correlation between gene expression and [hemoglobin A1c] level, duration of diabetes, presence of secondary complications or the reason that led to endoscopy, indicating that gene expression was not influenced by these variables,” the researchers write.
They also found a “specific microbiota composition” featuring a reduction in the role of Proteobacteria and an increase in Firmicutes; this was unique to the T1D patients. Bacteroidetes “showed a trend to reduction” in both T1D and celiac patients compared to the controls.
“The expression of genes specific for T1D inflammation was associated with the abundance of specific bacteria in duodenum,” the researchers added.
Elena Barengolts, MD, of the division of endocrinology, diabetes, and metabolism at the University of Illinois, Chicago, who’s familiar with the study, said it appears to be valid. However, the methods used have limited powers to define specific types of bacteria, making it difficult to know if the germs in question are “bad” or “good,” she said in an interview.
For his part, Dr. Kostic said the findings are “really neat” and consistent with previous findings regarding the role of the gut microbiome and T1D. He pointed to a study he led that found less-diverse microbiomes in the guts of Finnish infants with T1D (Cell Host Microbe. 2015 Feb 11;17[2]:260-73).
As a result, the gut microbiome is “functionally capable of doing fewer things, and the community gets overrun by certain pathogens,” he said. “We saw that a lot of organisms were capable of promoting inflammation in the gut.”
Dr. Kostic, Dr. Barengolts, and the study authors report no relevant disclosures.
FROM THE JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM
Key clinical point: Patients with type 1 diabetes (T1D) show signs of unique inflammation and microbiota in the duodenal mucosa, compared with controls and celiac patients.
Major finding: T1D patients had a “peculiar” inflammation signature and a unique microbiota composition, and there’s a sign of a link between inflammation and bacteria levels.
Data source: An analysis of 19 patients with T1D, 19 with celiac disease, and 16 healthy controls.
Disclosures: The study was supported by institutional funds, and the authors report no relevant disclosures.
Symptoms don’t worsen during pregnancy in most PsA patients
Psoriatic arthritis disease activity tended to improve or stabilize during pregnancy and the first year after giving birth, even outpacing controls, in a small retrospective cohort study.
“The outcome of pregnancy in PsA [psoriatic arthritis] patients is excellent,” investigators from the University of Toronto Psoriatic Arthritis Program reported. They found that “arthritis activity has a favorable course during pregnancy in almost 60% of the pregnancies, while the skin disease shows a favorable course in close to 90% of the pregnancies.”
According to the researchers, led by Ari Polachek, MD, of the Centre for Prognosis Studies in the Rheumatic Diseases at Toronto Western Hospital, previous research has found that rheumatoid arthritis improves in most pregnant women with the condition but then worsens in the year after birth, while most patients with ankylosing spondylitis don’t get better or worse. Limited research suggests psoriasis tends to improve during pregnancy and then flares afterward.
For the new study, researchers tracked 29 pregnant PsA patients with 42 total pregnancies who had a mean age of about 34 years at the beginning of pregnancy and matched them with 67 control patients with PsA who were not pregnant and had an average age of about 35 years. They had all visited the University of Toronto Psoriatic Arthritis Clinic during 1990-2015 (Semin Arthritis Rheum. 2017 Jan 16. doi: 10.1016/j.semarthrit.2017.01.002).
Among the 41 pregnancies in women who began follow-up prior to pregnancy, 13 (32%) had an unfavorable course of disease activity marked by worsening during 8 (20%) of the pregnancies or stable high disease activity during 5 (12%). The course of disease activity was more favorable for 24 (59%) pregnancies, in which it improved in 11 (27%) and stayed stable at a low disease level in 13 (32%). Four pregnancies (10%) had a mixed pattern of improvement followed by worsening.
The 1-year postpartum period during 40 pregnancies showed that stable or worsening symptoms were most common: 8 (20%) had improvement and 13 (32.5%) had stable low disease activity, while another 16 (4%) worsened and 3 (8%) had a mixed course of improvement followed by worsening.
After they controlled the results to account for various factors, the researchers found that pregnancy appeared to be especially beneficial for skin-related PsA symptoms. The likelihood of improved skin activity during pregnancy rose significantly (odds ratio, 7.7; 95% confidence interval, 1.8-23.5; P = .004) when compared with a matched period among controls, but this was no longer the case during the year after birth (OR, 0.4; 95% CI, 0.2-1.4; P = .60).
The likelihood of improved joint symptoms during pregnancy also rose, but not to a significant extent (OR, 2.1; 95% CI, 0.8-5.1; P = .10), and the effect on joints was not significant in the year after pregnancy (OR, 1.4; 95% CI, 0.5-3.4; P = .50).
There was a declining use of medications among the patients during pregnancy, particularly in the second and third trimesters, but two-thirds of the patients were treated with medications for PsA during this time, including NSAIDs (41%), disease-modifying antirheumatic drugs (35%), and biologic agents (26%).
The researchers speculated that hormonal changes during pregnancy may explain the improvement in skin activity. However, they noted that women with PsA who have multiple pregnancies don’t tend to have the same experiences each time: “Most of the women with more than one pregnancy had different joint disease course during their own different pregnancies and postpartum periods. Accordingly, this suggests that each woman needs specific evaluation and treatment adjustment during each pregnancy.”
The University of Toronto Psoriatic Arthritis Program is supported by a grant from the Krembil Foundation. Dr. Polachek is supported by an educational grant from Janssen Canada.
Psoriatic arthritis disease activity tended to improve or stabilize during pregnancy and the first year after giving birth, even outpacing controls, in a small retrospective cohort study.
“The outcome of pregnancy in PsA [psoriatic arthritis] patients is excellent,” investigators from the University of Toronto Psoriatic Arthritis Program reported. They found that “arthritis activity has a favorable course during pregnancy in almost 60% of the pregnancies, while the skin disease shows a favorable course in close to 90% of the pregnancies.”
According to the researchers, led by Ari Polachek, MD, of the Centre for Prognosis Studies in the Rheumatic Diseases at Toronto Western Hospital, previous research has found that rheumatoid arthritis improves in most pregnant women with the condition but then worsens in the year after birth, while most patients with ankylosing spondylitis don’t get better or worse. Limited research suggests psoriasis tends to improve during pregnancy and then flares afterward.
For the new study, researchers tracked 29 pregnant PsA patients with 42 total pregnancies who had a mean age of about 34 years at the beginning of pregnancy and matched them with 67 control patients with PsA who were not pregnant and had an average age of about 35 years. They had all visited the University of Toronto Psoriatic Arthritis Clinic during 1990-2015 (Semin Arthritis Rheum. 2017 Jan 16. doi: 10.1016/j.semarthrit.2017.01.002).
Among the 41 pregnancies in women who began follow-up prior to pregnancy, 13 (32%) had an unfavorable course of disease activity marked by worsening during 8 (20%) of the pregnancies or stable high disease activity during 5 (12%). The course of disease activity was more favorable for 24 (59%) pregnancies, in which it improved in 11 (27%) and stayed stable at a low disease level in 13 (32%). Four pregnancies (10%) had a mixed pattern of improvement followed by worsening.
The 1-year postpartum period during 40 pregnancies showed that stable or worsening symptoms were most common: 8 (20%) had improvement and 13 (32.5%) had stable low disease activity, while another 16 (4%) worsened and 3 (8%) had a mixed course of improvement followed by worsening.
After they controlled the results to account for various factors, the researchers found that pregnancy appeared to be especially beneficial for skin-related PsA symptoms. The likelihood of improved skin activity during pregnancy rose significantly (odds ratio, 7.7; 95% confidence interval, 1.8-23.5; P = .004) when compared with a matched period among controls, but this was no longer the case during the year after birth (OR, 0.4; 95% CI, 0.2-1.4; P = .60).
The likelihood of improved joint symptoms during pregnancy also rose, but not to a significant extent (OR, 2.1; 95% CI, 0.8-5.1; P = .10), and the effect on joints was not significant in the year after pregnancy (OR, 1.4; 95% CI, 0.5-3.4; P = .50).
There was a declining use of medications among the patients during pregnancy, particularly in the second and third trimesters, but two-thirds of the patients were treated with medications for PsA during this time, including NSAIDs (41%), disease-modifying antirheumatic drugs (35%), and biologic agents (26%).
The researchers speculated that hormonal changes during pregnancy may explain the improvement in skin activity. However, they noted that women with PsA who have multiple pregnancies don’t tend to have the same experiences each time: “Most of the women with more than one pregnancy had different joint disease course during their own different pregnancies and postpartum periods. Accordingly, this suggests that each woman needs specific evaluation and treatment adjustment during each pregnancy.”
The University of Toronto Psoriatic Arthritis Program is supported by a grant from the Krembil Foundation. Dr. Polachek is supported by an educational grant from Janssen Canada.
Psoriatic arthritis disease activity tended to improve or stabilize during pregnancy and the first year after giving birth, even outpacing controls, in a small retrospective cohort study.
“The outcome of pregnancy in PsA [psoriatic arthritis] patients is excellent,” investigators from the University of Toronto Psoriatic Arthritis Program reported. They found that “arthritis activity has a favorable course during pregnancy in almost 60% of the pregnancies, while the skin disease shows a favorable course in close to 90% of the pregnancies.”
According to the researchers, led by Ari Polachek, MD, of the Centre for Prognosis Studies in the Rheumatic Diseases at Toronto Western Hospital, previous research has found that rheumatoid arthritis improves in most pregnant women with the condition but then worsens in the year after birth, while most patients with ankylosing spondylitis don’t get better or worse. Limited research suggests psoriasis tends to improve during pregnancy and then flares afterward.
For the new study, researchers tracked 29 pregnant PsA patients with 42 total pregnancies who had a mean age of about 34 years at the beginning of pregnancy and matched them with 67 control patients with PsA who were not pregnant and had an average age of about 35 years. They had all visited the University of Toronto Psoriatic Arthritis Clinic during 1990-2015 (Semin Arthritis Rheum. 2017 Jan 16. doi: 10.1016/j.semarthrit.2017.01.002).
Among the 41 pregnancies in women who began follow-up prior to pregnancy, 13 (32%) had an unfavorable course of disease activity marked by worsening during 8 (20%) of the pregnancies or stable high disease activity during 5 (12%). The course of disease activity was more favorable for 24 (59%) pregnancies, in which it improved in 11 (27%) and stayed stable at a low disease level in 13 (32%). Four pregnancies (10%) had a mixed pattern of improvement followed by worsening.
The 1-year postpartum period during 40 pregnancies showed that stable or worsening symptoms were most common: 8 (20%) had improvement and 13 (32.5%) had stable low disease activity, while another 16 (4%) worsened and 3 (8%) had a mixed course of improvement followed by worsening.
After they controlled the results to account for various factors, the researchers found that pregnancy appeared to be especially beneficial for skin-related PsA symptoms. The likelihood of improved skin activity during pregnancy rose significantly (odds ratio, 7.7; 95% confidence interval, 1.8-23.5; P = .004) when compared with a matched period among controls, but this was no longer the case during the year after birth (OR, 0.4; 95% CI, 0.2-1.4; P = .60).
The likelihood of improved joint symptoms during pregnancy also rose, but not to a significant extent (OR, 2.1; 95% CI, 0.8-5.1; P = .10), and the effect on joints was not significant in the year after pregnancy (OR, 1.4; 95% CI, 0.5-3.4; P = .50).
There was a declining use of medications among the patients during pregnancy, particularly in the second and third trimesters, but two-thirds of the patients were treated with medications for PsA during this time, including NSAIDs (41%), disease-modifying antirheumatic drugs (35%), and biologic agents (26%).
The researchers speculated that hormonal changes during pregnancy may explain the improvement in skin activity. However, they noted that women with PsA who have multiple pregnancies don’t tend to have the same experiences each time: “Most of the women with more than one pregnancy had different joint disease course during their own different pregnancies and postpartum periods. Accordingly, this suggests that each woman needs specific evaluation and treatment adjustment during each pregnancy.”
The University of Toronto Psoriatic Arthritis Program is supported by a grant from the Krembil Foundation. Dr. Polachek is supported by an educational grant from Janssen Canada.
Key clinical point:
Major finding: PsA symptoms improved in 27% of pregnancies, stabilized at a low level in 32%, and worsened in 20%.
Data source: 29 pregnant PsA patients with 42 total pregnancies and 67 non-pregnant PsA patients.
Disclosures: The University of Toronto Psoriatic Arthritis Program is supported by a grant from the Krembil Foundation. Dr. Polachek is supported by an educational grant from Janssen Canada.
Childhood PCV program produces overall protection
Childhood pneumococcal conjugate vaccines continue to indirectly produce widespread societal protection against invasive pneumococcal disease, a review and meta-analysis showed.
In fact, the reviewed studies suggest that the use of these vaccines in children can lead to an overall 90% drop in invasive pneumococcal disease within fewer than 10 years.
Herd immunity appears to be at work, the review authors said. The effect is so powerful that the findings raise questions about “the merit of offering [the 13-valent pneumococcal vaccine] in older groups” in places that have a children’s pneumococcal conjugate vaccine (PCV) program, the investigators said.
U.S. guidelines recommend vaccinations for older people, although the recommendations are up for review in 2018.
According to the review, childhood PCVs have had a tremendous impact since a seven-valent version (PCV7) was first released in 2000. “In vaccinated young children, disease due to serotypes included in the vaccines has been reduced to negligible levels,” the authors wrote.
But unvaccinated people, especially the elderly, remain susceptible.
The review authors, led by Tinevimbo Shiri, PhD, of the University of Warwick, Coventry, England, updated a 2013 systemic review (Vaccine. 2013 Dec 17;32[1]:133-45). They focused on studies from 1994 to 2016 that examined the effects of introducing PCV in children.
A total of 242 studies were included in the meta-analysis, published in the January issue of Lancet Global Health (2017 Jan;5[1]:e51-e9), including 70 from the previous review. Of these, only 9 (4%) were performed in poor or middle-income countries, with most of the rest having been done in North America (42%) and Europe (38%).
The researchers found that “[herd] immunity effects continued to accumulate over time and reduced disease due to PCV7 serotypes, for which follow-up data have generally been available for the longest period, with a 90% average reduction after about 9 years.”
Specifically, the review estimated it would take 8.9 years for a 90% reduction in invasive pneumococcal disease for grouped serotypes in the PCV7 and 9.5 years for the extra six grouped serotypes in the 13-valent PCV. The latter vaccine was introduced in 2010.
The researchers found evidence of a similar annual reduction in disease linked to grouped serotypes in the 23-valent pneumococcal polysaccharide vaccine in adults aged 19 and up. They noted that the 11 serotypes contained in PPV23 but not in PCV13 “did not change invasive pneumococcal disease at any age.”
The investigators added: “In countries with mature pediatric PCV programmes such as Canada, Germany, the Netherlands, the U.K., and the U.S.A., invasive pneumococcal disease due to PCV7 serotypes has been nearly eliminated through indirect protection - i.e., the average incidence of PCV7-invasive pneumococcal disease after nearly a decade of PCV7 use is less than 10 per 100,000 people. In these countries, consistent decreases in vaccine-type adult community-acquired pneumonia (CAP) or meningitis, and nonbacteraemic CAP, have been observed, indicating substantial indirect protection effects against noninvasive disease from childhood vaccination.”
The review authors noted that a major “evidence gap” in the effectiveness of childhood PCV programs in low-income countries exists. “Because these countries are increasingly undertaking childhood vaccination programs, research to assess the indirect effects in these settings is particularly relevant,” they wrote.
The review’s limitations include the possibility that the results could be thrown off by variations across nations in areas like diagnostic protocols, surveillance, and outcome measures.
The authors of the review, funded by the Policy Research Program of England’s Department of Health, reported no relevant financial disclosures.
The impact of pneumococcal conjugate vaccines has been hampered by serotypes that they don’t address, said David Goldblatt, PhD, MBChB, of the Great Ormond Street Institute of Child Health and University College London, in a commentary accompanying this review and meta-analysis (Lancet Glob Health. 2017 Jan;5[1]:e6-e7).
“Data from this meta-analysis have shown an overall reduction of invasive pneumococcal disease in all unvaccinated age groups of just 1%. … New extended-valency vaccines will be required to halt this erosion of PCV impact,” he wrote.
Dr. Goldblatt reported receiving grants and personal fees from GlaxoSmithKline, Merck, Sharpe, and Dohme and a publication with Pfizer.
The impact of pneumococcal conjugate vaccines has been hampered by serotypes that they don’t address, said David Goldblatt, PhD, MBChB, of the Great Ormond Street Institute of Child Health and University College London, in a commentary accompanying this review and meta-analysis (Lancet Glob Health. 2017 Jan;5[1]:e6-e7).
“Data from this meta-analysis have shown an overall reduction of invasive pneumococcal disease in all unvaccinated age groups of just 1%. … New extended-valency vaccines will be required to halt this erosion of PCV impact,” he wrote.
Dr. Goldblatt reported receiving grants and personal fees from GlaxoSmithKline, Merck, Sharpe, and Dohme and a publication with Pfizer.
The impact of pneumococcal conjugate vaccines has been hampered by serotypes that they don’t address, said David Goldblatt, PhD, MBChB, of the Great Ormond Street Institute of Child Health and University College London, in a commentary accompanying this review and meta-analysis (Lancet Glob Health. 2017 Jan;5[1]:e6-e7).
“Data from this meta-analysis have shown an overall reduction of invasive pneumococcal disease in all unvaccinated age groups of just 1%. … New extended-valency vaccines will be required to halt this erosion of PCV impact,” he wrote.
Dr. Goldblatt reported receiving grants and personal fees from GlaxoSmithKline, Merck, Sharpe, and Dohme and a publication with Pfizer.
Childhood pneumococcal conjugate vaccines continue to indirectly produce widespread societal protection against invasive pneumococcal disease, a review and meta-analysis showed.
In fact, the reviewed studies suggest that the use of these vaccines in children can lead to an overall 90% drop in invasive pneumococcal disease within fewer than 10 years.
Herd immunity appears to be at work, the review authors said. The effect is so powerful that the findings raise questions about “the merit of offering [the 13-valent pneumococcal vaccine] in older groups” in places that have a children’s pneumococcal conjugate vaccine (PCV) program, the investigators said.
U.S. guidelines recommend vaccinations for older people, although the recommendations are up for review in 2018.
According to the review, childhood PCVs have had a tremendous impact since a seven-valent version (PCV7) was first released in 2000. “In vaccinated young children, disease due to serotypes included in the vaccines has been reduced to negligible levels,” the authors wrote.
But unvaccinated people, especially the elderly, remain susceptible.
The review authors, led by Tinevimbo Shiri, PhD, of the University of Warwick, Coventry, England, updated a 2013 systemic review (Vaccine. 2013 Dec 17;32[1]:133-45). They focused on studies from 1994 to 2016 that examined the effects of introducing PCV in children.
A total of 242 studies were included in the meta-analysis, published in the January issue of Lancet Global Health (2017 Jan;5[1]:e51-e9), including 70 from the previous review. Of these, only 9 (4%) were performed in poor or middle-income countries, with most of the rest having been done in North America (42%) and Europe (38%).
The researchers found that “[herd] immunity effects continued to accumulate over time and reduced disease due to PCV7 serotypes, for which follow-up data have generally been available for the longest period, with a 90% average reduction after about 9 years.”
Specifically, the review estimated it would take 8.9 years for a 90% reduction in invasive pneumococcal disease for grouped serotypes in the PCV7 and 9.5 years for the extra six grouped serotypes in the 13-valent PCV. The latter vaccine was introduced in 2010.
The researchers found evidence of a similar annual reduction in disease linked to grouped serotypes in the 23-valent pneumococcal polysaccharide vaccine in adults aged 19 and up. They noted that the 11 serotypes contained in PPV23 but not in PCV13 “did not change invasive pneumococcal disease at any age.”
The investigators added: “In countries with mature pediatric PCV programmes such as Canada, Germany, the Netherlands, the U.K., and the U.S.A., invasive pneumococcal disease due to PCV7 serotypes has been nearly eliminated through indirect protection - i.e., the average incidence of PCV7-invasive pneumococcal disease after nearly a decade of PCV7 use is less than 10 per 100,000 people. In these countries, consistent decreases in vaccine-type adult community-acquired pneumonia (CAP) or meningitis, and nonbacteraemic CAP, have been observed, indicating substantial indirect protection effects against noninvasive disease from childhood vaccination.”
The review authors noted that a major “evidence gap” in the effectiveness of childhood PCV programs in low-income countries exists. “Because these countries are increasingly undertaking childhood vaccination programs, research to assess the indirect effects in these settings is particularly relevant,” they wrote.
The review’s limitations include the possibility that the results could be thrown off by variations across nations in areas like diagnostic protocols, surveillance, and outcome measures.
The authors of the review, funded by the Policy Research Program of England’s Department of Health, reported no relevant financial disclosures.
Childhood pneumococcal conjugate vaccines continue to indirectly produce widespread societal protection against invasive pneumococcal disease, a review and meta-analysis showed.
In fact, the reviewed studies suggest that the use of these vaccines in children can lead to an overall 90% drop in invasive pneumococcal disease within fewer than 10 years.
Herd immunity appears to be at work, the review authors said. The effect is so powerful that the findings raise questions about “the merit of offering [the 13-valent pneumococcal vaccine] in older groups” in places that have a children’s pneumococcal conjugate vaccine (PCV) program, the investigators said.
U.S. guidelines recommend vaccinations for older people, although the recommendations are up for review in 2018.
According to the review, childhood PCVs have had a tremendous impact since a seven-valent version (PCV7) was first released in 2000. “In vaccinated young children, disease due to serotypes included in the vaccines has been reduced to negligible levels,” the authors wrote.
But unvaccinated people, especially the elderly, remain susceptible.
The review authors, led by Tinevimbo Shiri, PhD, of the University of Warwick, Coventry, England, updated a 2013 systemic review (Vaccine. 2013 Dec 17;32[1]:133-45). They focused on studies from 1994 to 2016 that examined the effects of introducing PCV in children.
A total of 242 studies were included in the meta-analysis, published in the January issue of Lancet Global Health (2017 Jan;5[1]:e51-e9), including 70 from the previous review. Of these, only 9 (4%) were performed in poor or middle-income countries, with most of the rest having been done in North America (42%) and Europe (38%).
The researchers found that “[herd] immunity effects continued to accumulate over time and reduced disease due to PCV7 serotypes, for which follow-up data have generally been available for the longest period, with a 90% average reduction after about 9 years.”
Specifically, the review estimated it would take 8.9 years for a 90% reduction in invasive pneumococcal disease for grouped serotypes in the PCV7 and 9.5 years for the extra six grouped serotypes in the 13-valent PCV. The latter vaccine was introduced in 2010.
The researchers found evidence of a similar annual reduction in disease linked to grouped serotypes in the 23-valent pneumococcal polysaccharide vaccine in adults aged 19 and up. They noted that the 11 serotypes contained in PPV23 but not in PCV13 “did not change invasive pneumococcal disease at any age.”
The investigators added: “In countries with mature pediatric PCV programmes such as Canada, Germany, the Netherlands, the U.K., and the U.S.A., invasive pneumococcal disease due to PCV7 serotypes has been nearly eliminated through indirect protection - i.e., the average incidence of PCV7-invasive pneumococcal disease after nearly a decade of PCV7 use is less than 10 per 100,000 people. In these countries, consistent decreases in vaccine-type adult community-acquired pneumonia (CAP) or meningitis, and nonbacteraemic CAP, have been observed, indicating substantial indirect protection effects against noninvasive disease from childhood vaccination.”
The review authors noted that a major “evidence gap” in the effectiveness of childhood PCV programs in low-income countries exists. “Because these countries are increasingly undertaking childhood vaccination programs, research to assess the indirect effects in these settings is particularly relevant,” they wrote.
The review’s limitations include the possibility that the results could be thrown off by variations across nations in areas like diagnostic protocols, surveillance, and outcome measures.
The authors of the review, funded by the Policy Research Program of England’s Department of Health, reported no relevant financial disclosures.
FROM THE LANCET GLOBAL HEALTH
Key clinical point: Through apparent herd immunity, childhood pneumococcal conjugate vaccines produce societywide reductions in invasive pneumococcal disease in less than a decade.
Major finding: When a seven-valent PCV is given to children, it would take an estimated 8.9 years for a 90% reduction in invasive pneumococcal disease to be seen among the grouped serotypes. It would take an estimated 9.5 years for an identical reduction in the extra six grouped serotypes in the 13-valent PCV.
Data source: A review and meta-analysis of 242 studies from 1994 to 2016 that examined the effects of introducing PCVs in children.
Disclosures: The review authors reported no relevant financial disclosures.