User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
Earlier epilepsy surgery may have reproductive benefits for women
WASHINGTON – Younger age at the time of surgery, and receipt of fewer medications prior to surgery were associated with higher rates of pregnancy and birth after surgery in a study of women with epilepsy.
The findings, from a retrospective review of the charts of 113 women who underwent surgery involving cortical resection between 1997 and 2008 for intractable focal epilepsy, suggest that earlier may be better when it comes to surgical intervention for young women with epilepsy who desire pregnancy, Dr. Rachel R. Fabris reported at the annual meeting of the American Epilepsy Society.
For the women included in this analysis, the mean age was 13.3 years at epilepsy onset and the mean age was 30.5 years at time of surgery at the Mayo Clinic, Rochester, Minn. They had an average of 5.57 medication trials, 42% had at least monthly seizures, and 21% had daily seizures. They were followed for a mean of 5.7 years after surgery, and 75% had Engel Class I disease after surgery, said Dr. Fabris of the Mayo Clinic.
Prior to surgery, the women had an average of 0.93 pregnancies each, and an average of 0.73 births; after surgery, a total of 17 women had a total of 35 pregnancies and 25 births, for an average of 1.27 pregnancies and 0.96 births each.
One patient reported infertility after surgery.
Younger patients experienced the greatest increases in the number of pregnancies and births (P = .0036 and .0060, respectively), and those taking fewer medications also experienced a significant change in the number of births after surgery (P = .0362). The former finding is likely not meaningful, because younger women have higher reproductive rates than do older women, in general; the latter finding, however, may suggest a role for earlier surgical intervention in women with epilepsy, Dr. Fabris said.
These preliminary findings provide additional evidence of the importance of early surgery for intractable focal epilepsy, but their significance with respect to reproduction is uncertain given the small number of patients, she said. Dr. Fabris said she believes this to be the first study to evaluate the effect of surgery on reproductive outcomes.
"We already know from former studies that women with epilepsy have lower birth rates than women in the general population, and multiple studies have shown this to be a multifactorial process that involves an interplay of endocrine as well as societal factors," she said during a press briefing at the conference.
Since antiepileptic medications also are known to have adverse effects on fertility, it may be that this effect is contributing to the current findings, she noted, concluding that additional study with more patients is needed to provide more meaningful results. There were no relevant financial disclosures.
WASHINGTON – Younger age at the time of surgery, and receipt of fewer medications prior to surgery were associated with higher rates of pregnancy and birth after surgery in a study of women with epilepsy.
The findings, from a retrospective review of the charts of 113 women who underwent surgery involving cortical resection between 1997 and 2008 for intractable focal epilepsy, suggest that earlier may be better when it comes to surgical intervention for young women with epilepsy who desire pregnancy, Dr. Rachel R. Fabris reported at the annual meeting of the American Epilepsy Society.
For the women included in this analysis, the mean age was 13.3 years at epilepsy onset and the mean age was 30.5 years at time of surgery at the Mayo Clinic, Rochester, Minn. They had an average of 5.57 medication trials, 42% had at least monthly seizures, and 21% had daily seizures. They were followed for a mean of 5.7 years after surgery, and 75% had Engel Class I disease after surgery, said Dr. Fabris of the Mayo Clinic.
Prior to surgery, the women had an average of 0.93 pregnancies each, and an average of 0.73 births; after surgery, a total of 17 women had a total of 35 pregnancies and 25 births, for an average of 1.27 pregnancies and 0.96 births each.
One patient reported infertility after surgery.
Younger patients experienced the greatest increases in the number of pregnancies and births (P = .0036 and .0060, respectively), and those taking fewer medications also experienced a significant change in the number of births after surgery (P = .0362). The former finding is likely not meaningful, because younger women have higher reproductive rates than do older women, in general; the latter finding, however, may suggest a role for earlier surgical intervention in women with epilepsy, Dr. Fabris said.
These preliminary findings provide additional evidence of the importance of early surgery for intractable focal epilepsy, but their significance with respect to reproduction is uncertain given the small number of patients, she said. Dr. Fabris said she believes this to be the first study to evaluate the effect of surgery on reproductive outcomes.
"We already know from former studies that women with epilepsy have lower birth rates than women in the general population, and multiple studies have shown this to be a multifactorial process that involves an interplay of endocrine as well as societal factors," she said during a press briefing at the conference.
Since antiepileptic medications also are known to have adverse effects on fertility, it may be that this effect is contributing to the current findings, she noted, concluding that additional study with more patients is needed to provide more meaningful results. There were no relevant financial disclosures.
WASHINGTON – Younger age at the time of surgery, and receipt of fewer medications prior to surgery were associated with higher rates of pregnancy and birth after surgery in a study of women with epilepsy.
The findings, from a retrospective review of the charts of 113 women who underwent surgery involving cortical resection between 1997 and 2008 for intractable focal epilepsy, suggest that earlier may be better when it comes to surgical intervention for young women with epilepsy who desire pregnancy, Dr. Rachel R. Fabris reported at the annual meeting of the American Epilepsy Society.
For the women included in this analysis, the mean age was 13.3 years at epilepsy onset and the mean age was 30.5 years at time of surgery at the Mayo Clinic, Rochester, Minn. They had an average of 5.57 medication trials, 42% had at least monthly seizures, and 21% had daily seizures. They were followed for a mean of 5.7 years after surgery, and 75% had Engel Class I disease after surgery, said Dr. Fabris of the Mayo Clinic.
Prior to surgery, the women had an average of 0.93 pregnancies each, and an average of 0.73 births; after surgery, a total of 17 women had a total of 35 pregnancies and 25 births, for an average of 1.27 pregnancies and 0.96 births each.
One patient reported infertility after surgery.
Younger patients experienced the greatest increases in the number of pregnancies and births (P = .0036 and .0060, respectively), and those taking fewer medications also experienced a significant change in the number of births after surgery (P = .0362). The former finding is likely not meaningful, because younger women have higher reproductive rates than do older women, in general; the latter finding, however, may suggest a role for earlier surgical intervention in women with epilepsy, Dr. Fabris said.
These preliminary findings provide additional evidence of the importance of early surgery for intractable focal epilepsy, but their significance with respect to reproduction is uncertain given the small number of patients, she said. Dr. Fabris said she believes this to be the first study to evaluate the effect of surgery on reproductive outcomes.
"We already know from former studies that women with epilepsy have lower birth rates than women in the general population, and multiple studies have shown this to be a multifactorial process that involves an interplay of endocrine as well as societal factors," she said during a press briefing at the conference.
Since antiepileptic medications also are known to have adverse effects on fertility, it may be that this effect is contributing to the current findings, she noted, concluding that additional study with more patients is needed to provide more meaningful results. There were no relevant financial disclosures.
AT AES 2013
Major finding: Prior to surgery, the women had an average of 0.93 pregnancies each, and an average of 0.73 births; after surgery, a total of 17 women had a total of 35 pregnancies and 25 births, for an average of 1.27 pregnancies and 0.96 births each.
Data source: A chart review involving 113 patients.
Disclosures: There were no relevant financial disclosures.
Multiple antiepileptic drugs may pose risk of aseptic meningitis
WASHINGTON – Three commonly prescribed antiepileptic drugs – gabapentin, levetiracetam, and topiramate – were associated with greater risk of aseptic meningitis than was lamotrigine in a large cohort of new users of antiepileptic drugs.
Although a black box warning regarding the risk of aseptic meningitis was added to lamotrigine in 2010, no such warning is included for these three antiepileptic drugs (AEDs); the findings of this retrospective study have important implications for weighing treatment options in patients with seizures, Alexis Parente reported at the annual meeting of the American Epilepsy Society.
Among 719,749 AED users, including 60,011 children, the hazard ratios for aseptic meningitis were 1.80, 10.22, and 2.65, respectively, for gabapentin, levetiracetam, and topiramate, compared with lamotrigine, said Ms. Parente of Inovalon, Washington.
The median time to development of aseptic meningitis during a follow-up time of up to 12 months was shorter for children, compared with adults (44 days vs. 77 days), and for patients treated with levetiracetam, compared with those treated with lamotrigine (29.5 days vs. 83 days, respectively). There was no difference in time to aseptic meningitis between gabapentin and topiramate, compared with lamotrigine.
The study cohort included patients aged 2 years and older from a large, nationally representative administrative claims database who began taking an AED between 2006 and 2011. The patients were Medicaid-, Medicare-, and privately insured patients who were continuously enrolled with both medical and pharmacy benefits for at least a year prior to the index AED monotherapy prescription was filled. They did not use any other AED in the 90 days prior to the index prescription.
The findings are important, because long-term adherence to AEDs is a critical factor for the successful treatment of epilepsy, psychotic disorders, and pain, and ideal AEDs should have both high potency and tolerability over the long-term, Ms. Parente said. While some small studies have suggested that some second-generation AEDs may be safer than lamotrigine, she noted, their comparative risks have remained largely unclear.
Adverse effects are the leading cause of treatment failures and are associated with up to 25% of treatment discontinuations in these patients, leading to decreased quality of life and increased health care costs, she added.
WASHINGTON – Three commonly prescribed antiepileptic drugs – gabapentin, levetiracetam, and topiramate – were associated with greater risk of aseptic meningitis than was lamotrigine in a large cohort of new users of antiepileptic drugs.
Although a black box warning regarding the risk of aseptic meningitis was added to lamotrigine in 2010, no such warning is included for these three antiepileptic drugs (AEDs); the findings of this retrospective study have important implications for weighing treatment options in patients with seizures, Alexis Parente reported at the annual meeting of the American Epilepsy Society.
Among 719,749 AED users, including 60,011 children, the hazard ratios for aseptic meningitis were 1.80, 10.22, and 2.65, respectively, for gabapentin, levetiracetam, and topiramate, compared with lamotrigine, said Ms. Parente of Inovalon, Washington.
The median time to development of aseptic meningitis during a follow-up time of up to 12 months was shorter for children, compared with adults (44 days vs. 77 days), and for patients treated with levetiracetam, compared with those treated with lamotrigine (29.5 days vs. 83 days, respectively). There was no difference in time to aseptic meningitis between gabapentin and topiramate, compared with lamotrigine.
The study cohort included patients aged 2 years and older from a large, nationally representative administrative claims database who began taking an AED between 2006 and 2011. The patients were Medicaid-, Medicare-, and privately insured patients who were continuously enrolled with both medical and pharmacy benefits for at least a year prior to the index AED monotherapy prescription was filled. They did not use any other AED in the 90 days prior to the index prescription.
The findings are important, because long-term adherence to AEDs is a critical factor for the successful treatment of epilepsy, psychotic disorders, and pain, and ideal AEDs should have both high potency and tolerability over the long-term, Ms. Parente said. While some small studies have suggested that some second-generation AEDs may be safer than lamotrigine, she noted, their comparative risks have remained largely unclear.
Adverse effects are the leading cause of treatment failures and are associated with up to 25% of treatment discontinuations in these patients, leading to decreased quality of life and increased health care costs, she added.
WASHINGTON – Three commonly prescribed antiepileptic drugs – gabapentin, levetiracetam, and topiramate – were associated with greater risk of aseptic meningitis than was lamotrigine in a large cohort of new users of antiepileptic drugs.
Although a black box warning regarding the risk of aseptic meningitis was added to lamotrigine in 2010, no such warning is included for these three antiepileptic drugs (AEDs); the findings of this retrospective study have important implications for weighing treatment options in patients with seizures, Alexis Parente reported at the annual meeting of the American Epilepsy Society.
Among 719,749 AED users, including 60,011 children, the hazard ratios for aseptic meningitis were 1.80, 10.22, and 2.65, respectively, for gabapentin, levetiracetam, and topiramate, compared with lamotrigine, said Ms. Parente of Inovalon, Washington.
The median time to development of aseptic meningitis during a follow-up time of up to 12 months was shorter for children, compared with adults (44 days vs. 77 days), and for patients treated with levetiracetam, compared with those treated with lamotrigine (29.5 days vs. 83 days, respectively). There was no difference in time to aseptic meningitis between gabapentin and topiramate, compared with lamotrigine.
The study cohort included patients aged 2 years and older from a large, nationally representative administrative claims database who began taking an AED between 2006 and 2011. The patients were Medicaid-, Medicare-, and privately insured patients who were continuously enrolled with both medical and pharmacy benefits for at least a year prior to the index AED monotherapy prescription was filled. They did not use any other AED in the 90 days prior to the index prescription.
The findings are important, because long-term adherence to AEDs is a critical factor for the successful treatment of epilepsy, psychotic disorders, and pain, and ideal AEDs should have both high potency and tolerability over the long-term, Ms. Parente said. While some small studies have suggested that some second-generation AEDs may be safer than lamotrigine, she noted, their comparative risks have remained largely unclear.
Adverse effects are the leading cause of treatment failures and are associated with up to 25% of treatment discontinuations in these patients, leading to decreased quality of life and increased health care costs, she added.
AT AES 2013
Major finding: Hazard ratios for aseptic meningitis were 1.80, 10.22, and 2.65, respectively, for gabapentin, levetiracetam, and topiramate, compared with lamotrigine.
Data source: A retrospective cohort study involving nearly 720,000 AED users.
Disclosures: No disclosures were provided.
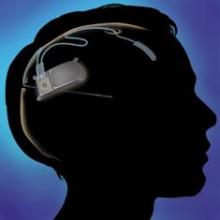
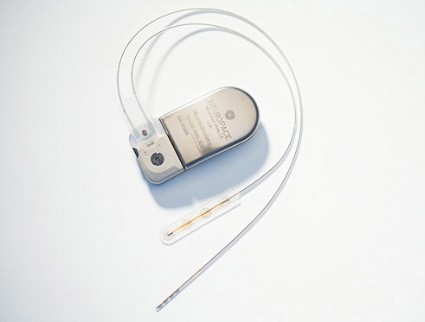
Cortical stimulation device provides safe, sustained seizure reduction
WASHINGTON – A recently approved device that provides responsive cortical stimulation was safe and provided sustained seizure reduction for a substantial number of patients with medically intractable partial onset seizures in a long-term efficacy and safety study.
The device – the RNS system (NeuroPace Inc.), which received Food and Drug Administration approval in November for use in adults – was associated with a median reduction in seizure frequency of 38.9% at 1 year and 51.1% at 2 years in 256 adult patients; 20% of patients were free of seizures for at least 6 months. No safety issues arose over a median of 4.5 years of follow-up (1,200 patient implant-years) as of Nov. 1, 2012, Dr. Gregory K. Bergey reported at the annual meeting of the American Epilepsy Society.
Study subjects had a mean age of 34 years and a mean epilepsy duration of 19.6 years. They were taking a mean of 2.9 antiepileptic drugs. Their seizures were localized to one or two seizure foci, and their median seizure frequency was 10.2 per 28 days, said Dr. Bergey, director of the Johns Hopkins Epilepsy Center in Baltimore.
The study population was extremely refractory, experiencing at least three debilitating seizures monthly and having no months with fewer than two such seizures, he noted.
The first 65 patients were implanted during an earlier 2-year feasibility study, and 191 additional patients were implanted during a 2-year, multicenter, double-blind, randomized, sham-controlled, pivotal study in which the RNS system was also found to be safe and effective as an adjunct treatment for adults with partial-onset seizures.
In the current long-term efficacy and safety study, no unanticipated device-related serious adverse events occurred, and the overall rate of device-related adverse events declined over time. The serious adverse event rate was 0.010 per implant-year for hemorrhage-related events, and 0.023 per implant-year for infection-related events. Most of these events occurred in the first year after implant.
Of 11 deaths that occurred during the study period, 2 involved suicide, 1 occurred because of complications associated with status epilepticus, 1 was because of lymphoma, and 7 were attributed to sudden unexpected death in epilepsy (including 2 among patients not being treated with responsive stimulation) for a rate of 2.5/1,000 patient stimulation-years.
"This new treatment is an option for patients with intractable partial seizures," Dr. Bergey concluded, adding that the current findings, which show progressive improvement over time, support the long-term safety and efficacy of the RNS system.
A 7-year, open-label phase of the study is ongoing to accumulate 9 years of prospective data for those implanted during the feasibility and pivotal studies.
Dr. Bergey reported having no disclosures.
WASHINGTON – A recently approved device that provides responsive cortical stimulation was safe and provided sustained seizure reduction for a substantial number of patients with medically intractable partial onset seizures in a long-term efficacy and safety study.
The device – the RNS system (NeuroPace Inc.), which received Food and Drug Administration approval in November for use in adults – was associated with a median reduction in seizure frequency of 38.9% at 1 year and 51.1% at 2 years in 256 adult patients; 20% of patients were free of seizures for at least 6 months. No safety issues arose over a median of 4.5 years of follow-up (1,200 patient implant-years) as of Nov. 1, 2012, Dr. Gregory K. Bergey reported at the annual meeting of the American Epilepsy Society.
Study subjects had a mean age of 34 years and a mean epilepsy duration of 19.6 years. They were taking a mean of 2.9 antiepileptic drugs. Their seizures were localized to one or two seizure foci, and their median seizure frequency was 10.2 per 28 days, said Dr. Bergey, director of the Johns Hopkins Epilepsy Center in Baltimore.
The study population was extremely refractory, experiencing at least three debilitating seizures monthly and having no months with fewer than two such seizures, he noted.
The first 65 patients were implanted during an earlier 2-year feasibility study, and 191 additional patients were implanted during a 2-year, multicenter, double-blind, randomized, sham-controlled, pivotal study in which the RNS system was also found to be safe and effective as an adjunct treatment for adults with partial-onset seizures.
In the current long-term efficacy and safety study, no unanticipated device-related serious adverse events occurred, and the overall rate of device-related adverse events declined over time. The serious adverse event rate was 0.010 per implant-year for hemorrhage-related events, and 0.023 per implant-year for infection-related events. Most of these events occurred in the first year after implant.
Of 11 deaths that occurred during the study period, 2 involved suicide, 1 occurred because of complications associated with status epilepticus, 1 was because of lymphoma, and 7 were attributed to sudden unexpected death in epilepsy (including 2 among patients not being treated with responsive stimulation) for a rate of 2.5/1,000 patient stimulation-years.
"This new treatment is an option for patients with intractable partial seizures," Dr. Bergey concluded, adding that the current findings, which show progressive improvement over time, support the long-term safety and efficacy of the RNS system.
A 7-year, open-label phase of the study is ongoing to accumulate 9 years of prospective data for those implanted during the feasibility and pivotal studies.
Dr. Bergey reported having no disclosures.
WASHINGTON – A recently approved device that provides responsive cortical stimulation was safe and provided sustained seizure reduction for a substantial number of patients with medically intractable partial onset seizures in a long-term efficacy and safety study.
The device – the RNS system (NeuroPace Inc.), which received Food and Drug Administration approval in November for use in adults – was associated with a median reduction in seizure frequency of 38.9% at 1 year and 51.1% at 2 years in 256 adult patients; 20% of patients were free of seizures for at least 6 months. No safety issues arose over a median of 4.5 years of follow-up (1,200 patient implant-years) as of Nov. 1, 2012, Dr. Gregory K. Bergey reported at the annual meeting of the American Epilepsy Society.
Study subjects had a mean age of 34 years and a mean epilepsy duration of 19.6 years. They were taking a mean of 2.9 antiepileptic drugs. Their seizures were localized to one or two seizure foci, and their median seizure frequency was 10.2 per 28 days, said Dr. Bergey, director of the Johns Hopkins Epilepsy Center in Baltimore.
The study population was extremely refractory, experiencing at least three debilitating seizures monthly and having no months with fewer than two such seizures, he noted.
The first 65 patients were implanted during an earlier 2-year feasibility study, and 191 additional patients were implanted during a 2-year, multicenter, double-blind, randomized, sham-controlled, pivotal study in which the RNS system was also found to be safe and effective as an adjunct treatment for adults with partial-onset seizures.
In the current long-term efficacy and safety study, no unanticipated device-related serious adverse events occurred, and the overall rate of device-related adverse events declined over time. The serious adverse event rate was 0.010 per implant-year for hemorrhage-related events, and 0.023 per implant-year for infection-related events. Most of these events occurred in the first year after implant.
Of 11 deaths that occurred during the study period, 2 involved suicide, 1 occurred because of complications associated with status epilepticus, 1 was because of lymphoma, and 7 were attributed to sudden unexpected death in epilepsy (including 2 among patients not being treated with responsive stimulation) for a rate of 2.5/1,000 patient stimulation-years.
"This new treatment is an option for patients with intractable partial seizures," Dr. Bergey concluded, adding that the current findings, which show progressive improvement over time, support the long-term safety and efficacy of the RNS system.
A 7-year, open-label phase of the study is ongoing to accumulate 9 years of prospective data for those implanted during the feasibility and pivotal studies.
Dr. Bergey reported having no disclosures.
AT AES 2013
Major finding: The median reduction in seizure frequency was 38.9% at 1 year and 51.1% at 2 years; 20% of patients were free of seizures for at least 6 months.
Data source: A study involving 256 patients with medically intractable partial onset seizures who participated in an early feasibility study and a double-blind, randomized, sham-controlled study.
Disclosures: Dr. Bergey reported having no disclosures.
Equipping pediatricians to take on cancer survivor care
ORLANDO – Pediatricians need to be positioned to offer survivor care to the more than 80% of pediatric cancer patients who now survive their disease, according to Dr. Melissa Hudson.
"We have in excess of 479,000 individuals living in the United States diagnosed with cancer before the age of 21 ... and we know with those numbers increasing, you will be increasingly seeing them in your practices," Dr. Hudson, director of the cancer survivorship division, department of oncology at St. Jude Children’s Research Hospital, Memphis, Tenn., said at the annual meeting of the American Academy of Pediatrics.
Health-related issues that affect these cancer survivors include the risk for recurrence, potential late effects of treatment, and psychosocial effects related to the cancer experience, Importantly, health care needs in these patients change over time and across the spectrum of care. Ideally, pediatricians will provide these patients with risk-based survivor care that includes health education, anticipatory guidance based on cancer history and specific cancer-related health risks, and cancer and other health screening and surveillance as recommended based on the patient’s specific treatment exposures, she said.
In particular, patients need to be educated about the impact of their health behaviors on cancer-related health risk and about effective self-management of chronic health conditions.
The common chronic conditions that follow cancer often can impact growth and development – including linear growth, skeletal maturation, intellectual functioning, emotional and social maturation, and sexual development.
Recently published data from the St. Jude Lifetime Cohort Study showed that 95.5% of nearly 1,700 adult survivors of pediatric cancer had at least 1 chronic health condition by age 45 years, and nearly 81% had a serious disabling or life-threatening condition (J. Clin. Oncol. 2013;31:4242-51). Data from the retrospective Childhood Cancer Survivor Study similarly showed that, compared with population controls, survivors have a higher rate of premature mortality, largely from subsequent malignancies, which confer a 15-fold excess risk of death; cardiac conditions, which confer a sevenfold excess risk of death; and pulmonary conditions, which confer a nearly ninefold excess risk of death.
Survivor care also needs to consider that treatment-related issues, ongoing cancer risk, and the cancer experience in general can combine to have important psychosocial effects
Mental health factors can include body image issues, adverse effects of the cancer experience on educational and vocational goals, and late cognitive effects. Survivors also face insurance discrimination and access to care issues due to financial issues associated with medical bills and under- or unemployment.
Many survivors also struggle with ongoing symptoms such as fatigue, disrupted sleep, memory problems, concentration issues, and chronic pain; some have difficulty living independently because of the severity of these symptoms, and the effects can have a negative impact on social interactions with family and friends.
"We need to help them achieve access to resources to optimize their quality of life," Dr. Hudson said.
When cancer survivors return to primary care, they should present with a treatment summary. If they don’t, one should be requested from the cancer treatment center to help guide care, she said.
Become familiar with practice guidelines for caring for survivors of childhood cancers, and be prepared to work with either the oncology care center to help facilitate the transition to pediatric care, she said.
The Children’s Oncology Group (COG), a consortium of medical research institutions, provides numerous tools for care providers and patients. Dr. Hudson, who co-chaired the task force that established the care guidelines, said that they provide recommendations for screening and management of late effects that can arise in pediatric cancer survivors. These patient education handouts can be printed free of charge and many are available in Spanish and French as well as in English. An updated version (4.0) of the COG guidelines is pending release by early 2014, Dr. Hudson said.
Dr. Hudson reported having no disclosures.
ORLANDO – Pediatricians need to be positioned to offer survivor care to the more than 80% of pediatric cancer patients who now survive their disease, according to Dr. Melissa Hudson.
"We have in excess of 479,000 individuals living in the United States diagnosed with cancer before the age of 21 ... and we know with those numbers increasing, you will be increasingly seeing them in your practices," Dr. Hudson, director of the cancer survivorship division, department of oncology at St. Jude Children’s Research Hospital, Memphis, Tenn., said at the annual meeting of the American Academy of Pediatrics.
Health-related issues that affect these cancer survivors include the risk for recurrence, potential late effects of treatment, and psychosocial effects related to the cancer experience, Importantly, health care needs in these patients change over time and across the spectrum of care. Ideally, pediatricians will provide these patients with risk-based survivor care that includes health education, anticipatory guidance based on cancer history and specific cancer-related health risks, and cancer and other health screening and surveillance as recommended based on the patient’s specific treatment exposures, she said.
In particular, patients need to be educated about the impact of their health behaviors on cancer-related health risk and about effective self-management of chronic health conditions.
The common chronic conditions that follow cancer often can impact growth and development – including linear growth, skeletal maturation, intellectual functioning, emotional and social maturation, and sexual development.
Recently published data from the St. Jude Lifetime Cohort Study showed that 95.5% of nearly 1,700 adult survivors of pediatric cancer had at least 1 chronic health condition by age 45 years, and nearly 81% had a serious disabling or life-threatening condition (J. Clin. Oncol. 2013;31:4242-51). Data from the retrospective Childhood Cancer Survivor Study similarly showed that, compared with population controls, survivors have a higher rate of premature mortality, largely from subsequent malignancies, which confer a 15-fold excess risk of death; cardiac conditions, which confer a sevenfold excess risk of death; and pulmonary conditions, which confer a nearly ninefold excess risk of death.
Survivor care also needs to consider that treatment-related issues, ongoing cancer risk, and the cancer experience in general can combine to have important psychosocial effects
Mental health factors can include body image issues, adverse effects of the cancer experience on educational and vocational goals, and late cognitive effects. Survivors also face insurance discrimination and access to care issues due to financial issues associated with medical bills and under- or unemployment.
Many survivors also struggle with ongoing symptoms such as fatigue, disrupted sleep, memory problems, concentration issues, and chronic pain; some have difficulty living independently because of the severity of these symptoms, and the effects can have a negative impact on social interactions with family and friends.
"We need to help them achieve access to resources to optimize their quality of life," Dr. Hudson said.
When cancer survivors return to primary care, they should present with a treatment summary. If they don’t, one should be requested from the cancer treatment center to help guide care, she said.
Become familiar with practice guidelines for caring for survivors of childhood cancers, and be prepared to work with either the oncology care center to help facilitate the transition to pediatric care, she said.
The Children’s Oncology Group (COG), a consortium of medical research institutions, provides numerous tools for care providers and patients. Dr. Hudson, who co-chaired the task force that established the care guidelines, said that they provide recommendations for screening and management of late effects that can arise in pediatric cancer survivors. These patient education handouts can be printed free of charge and many are available in Spanish and French as well as in English. An updated version (4.0) of the COG guidelines is pending release by early 2014, Dr. Hudson said.
Dr. Hudson reported having no disclosures.
ORLANDO – Pediatricians need to be positioned to offer survivor care to the more than 80% of pediatric cancer patients who now survive their disease, according to Dr. Melissa Hudson.
"We have in excess of 479,000 individuals living in the United States diagnosed with cancer before the age of 21 ... and we know with those numbers increasing, you will be increasingly seeing them in your practices," Dr. Hudson, director of the cancer survivorship division, department of oncology at St. Jude Children’s Research Hospital, Memphis, Tenn., said at the annual meeting of the American Academy of Pediatrics.
Health-related issues that affect these cancer survivors include the risk for recurrence, potential late effects of treatment, and psychosocial effects related to the cancer experience, Importantly, health care needs in these patients change over time and across the spectrum of care. Ideally, pediatricians will provide these patients with risk-based survivor care that includes health education, anticipatory guidance based on cancer history and specific cancer-related health risks, and cancer and other health screening and surveillance as recommended based on the patient’s specific treatment exposures, she said.
In particular, patients need to be educated about the impact of their health behaviors on cancer-related health risk and about effective self-management of chronic health conditions.
The common chronic conditions that follow cancer often can impact growth and development – including linear growth, skeletal maturation, intellectual functioning, emotional and social maturation, and sexual development.
Recently published data from the St. Jude Lifetime Cohort Study showed that 95.5% of nearly 1,700 adult survivors of pediatric cancer had at least 1 chronic health condition by age 45 years, and nearly 81% had a serious disabling or life-threatening condition (J. Clin. Oncol. 2013;31:4242-51). Data from the retrospective Childhood Cancer Survivor Study similarly showed that, compared with population controls, survivors have a higher rate of premature mortality, largely from subsequent malignancies, which confer a 15-fold excess risk of death; cardiac conditions, which confer a sevenfold excess risk of death; and pulmonary conditions, which confer a nearly ninefold excess risk of death.
Survivor care also needs to consider that treatment-related issues, ongoing cancer risk, and the cancer experience in general can combine to have important psychosocial effects
Mental health factors can include body image issues, adverse effects of the cancer experience on educational and vocational goals, and late cognitive effects. Survivors also face insurance discrimination and access to care issues due to financial issues associated with medical bills and under- or unemployment.
Many survivors also struggle with ongoing symptoms such as fatigue, disrupted sleep, memory problems, concentration issues, and chronic pain; some have difficulty living independently because of the severity of these symptoms, and the effects can have a negative impact on social interactions with family and friends.
"We need to help them achieve access to resources to optimize their quality of life," Dr. Hudson said.
When cancer survivors return to primary care, they should present with a treatment summary. If they don’t, one should be requested from the cancer treatment center to help guide care, she said.
Become familiar with practice guidelines for caring for survivors of childhood cancers, and be prepared to work with either the oncology care center to help facilitate the transition to pediatric care, she said.
The Children’s Oncology Group (COG), a consortium of medical research institutions, provides numerous tools for care providers and patients. Dr. Hudson, who co-chaired the task force that established the care guidelines, said that they provide recommendations for screening and management of late effects that can arise in pediatric cancer survivors. These patient education handouts can be printed free of charge and many are available in Spanish and French as well as in English. An updated version (4.0) of the COG guidelines is pending release by early 2014, Dr. Hudson said.
Dr. Hudson reported having no disclosures.
AT THE AAP NATIONAL CONFERENCE
Folic acid supplementation low among women with epilepsy
WASHINGTON – Only about 43% of reproductive-age women with epilepsy who completed a Web-based survey were taking a folic acid supplement, according to interim findings from the Epilepsy Birth Control Registry.
Furthermore, only 46% of those considered to be at risk for becoming pregnant (for example, those who were sexually active and who had no history of infertility) reported taking a folic acid supplement, and among respondents who were taking antiepileptic drugs (AEDs), the lowest rate of folic acid supplement use (25%) was in those taking valproate, a folic acid antagonist, Dr. Andrew G. Herzog reported at the annual meeting of the American Epilepsy Society.
Folic acid deficiency is known to be associated with fetal loss and risk of neural tube defects, and valproate is the AED associated with the highest risk of neural tube malformations, said Dr. Herzog of Harvard Medical School, Boston, and Beth Israel Deaconess Medical Center, Wellesley, Mass.
However, respondents taking valproate in this study were the least likely to have a college degree, and having a college degree was found to be associated with greater likelihood of taking a folic acid supplement. In fact, having an associate college degree or higher was the only significant demographic predictor of folic acid use; 50% of those with a college degree were taking folic acid, compared with 39% of those without a college degree, and those with an advanced degree were twice as likely as those with only a high school education to be taking folic acid, but the numbers were insufficient for determining whether AED type or education level predicted folic acid use, Dr. Herzog noted.
Study subjects were women with epilepsy aged 18 to 47 years. The first 650 to complete the survey at the registry site were included in the analysis.
Prior findings from the registry showed that this is largely a population at high risk for pregnancy; about 60% of pregnancies among respondents were unintended, compared with about 47% in the general population, he said.
Yet, the only category of respondents for which folic acid use was more than 50%, including, for example, those at risk for pregnancy, those not at risk for pregnancy, those using birth control, those not using birth control, and those trying to become pregnant, was the latter – those women actively trying to become pregnant, of whom 70% were taking folic acid, he said.
Factors including age, seizure type, and insurance status were not associated with folic acid use.
Importantly, seeing a health care professional within the year prior to the survey also was not associated with increased folic acid use.
This is concerning, given that it has been known for three decades that folic acid deficiency is associated with serious consequences, Dr. Herzog said, suggesting that education about the importance of supplementation may get pushed aside by busy physicians focused more on seizure activity and epilepsy treatment.
Dr. Herzog reported having no disclosures.
WASHINGTON – Only about 43% of reproductive-age women with epilepsy who completed a Web-based survey were taking a folic acid supplement, according to interim findings from the Epilepsy Birth Control Registry.
Furthermore, only 46% of those considered to be at risk for becoming pregnant (for example, those who were sexually active and who had no history of infertility) reported taking a folic acid supplement, and among respondents who were taking antiepileptic drugs (AEDs), the lowest rate of folic acid supplement use (25%) was in those taking valproate, a folic acid antagonist, Dr. Andrew G. Herzog reported at the annual meeting of the American Epilepsy Society.
Folic acid deficiency is known to be associated with fetal loss and risk of neural tube defects, and valproate is the AED associated with the highest risk of neural tube malformations, said Dr. Herzog of Harvard Medical School, Boston, and Beth Israel Deaconess Medical Center, Wellesley, Mass.
However, respondents taking valproate in this study were the least likely to have a college degree, and having a college degree was found to be associated with greater likelihood of taking a folic acid supplement. In fact, having an associate college degree or higher was the only significant demographic predictor of folic acid use; 50% of those with a college degree were taking folic acid, compared with 39% of those without a college degree, and those with an advanced degree were twice as likely as those with only a high school education to be taking folic acid, but the numbers were insufficient for determining whether AED type or education level predicted folic acid use, Dr. Herzog noted.
Study subjects were women with epilepsy aged 18 to 47 years. The first 650 to complete the survey at the registry site were included in the analysis.
Prior findings from the registry showed that this is largely a population at high risk for pregnancy; about 60% of pregnancies among respondents were unintended, compared with about 47% in the general population, he said.
Yet, the only category of respondents for which folic acid use was more than 50%, including, for example, those at risk for pregnancy, those not at risk for pregnancy, those using birth control, those not using birth control, and those trying to become pregnant, was the latter – those women actively trying to become pregnant, of whom 70% were taking folic acid, he said.
Factors including age, seizure type, and insurance status were not associated with folic acid use.
Importantly, seeing a health care professional within the year prior to the survey also was not associated with increased folic acid use.
This is concerning, given that it has been known for three decades that folic acid deficiency is associated with serious consequences, Dr. Herzog said, suggesting that education about the importance of supplementation may get pushed aside by busy physicians focused more on seizure activity and epilepsy treatment.
Dr. Herzog reported having no disclosures.
WASHINGTON – Only about 43% of reproductive-age women with epilepsy who completed a Web-based survey were taking a folic acid supplement, according to interim findings from the Epilepsy Birth Control Registry.
Furthermore, only 46% of those considered to be at risk for becoming pregnant (for example, those who were sexually active and who had no history of infertility) reported taking a folic acid supplement, and among respondents who were taking antiepileptic drugs (AEDs), the lowest rate of folic acid supplement use (25%) was in those taking valproate, a folic acid antagonist, Dr. Andrew G. Herzog reported at the annual meeting of the American Epilepsy Society.
Folic acid deficiency is known to be associated with fetal loss and risk of neural tube defects, and valproate is the AED associated with the highest risk of neural tube malformations, said Dr. Herzog of Harvard Medical School, Boston, and Beth Israel Deaconess Medical Center, Wellesley, Mass.
However, respondents taking valproate in this study were the least likely to have a college degree, and having a college degree was found to be associated with greater likelihood of taking a folic acid supplement. In fact, having an associate college degree or higher was the only significant demographic predictor of folic acid use; 50% of those with a college degree were taking folic acid, compared with 39% of those without a college degree, and those with an advanced degree were twice as likely as those with only a high school education to be taking folic acid, but the numbers were insufficient for determining whether AED type or education level predicted folic acid use, Dr. Herzog noted.
Study subjects were women with epilepsy aged 18 to 47 years. The first 650 to complete the survey at the registry site were included in the analysis.
Prior findings from the registry showed that this is largely a population at high risk for pregnancy; about 60% of pregnancies among respondents were unintended, compared with about 47% in the general population, he said.
Yet, the only category of respondents for which folic acid use was more than 50%, including, for example, those at risk for pregnancy, those not at risk for pregnancy, those using birth control, those not using birth control, and those trying to become pregnant, was the latter – those women actively trying to become pregnant, of whom 70% were taking folic acid, he said.
Factors including age, seizure type, and insurance status were not associated with folic acid use.
Importantly, seeing a health care professional within the year prior to the survey also was not associated with increased folic acid use.
This is concerning, given that it has been known for three decades that folic acid deficiency is associated with serious consequences, Dr. Herzog said, suggesting that education about the importance of supplementation may get pushed aside by busy physicians focused more on seizure activity and epilepsy treatment.
Dr. Herzog reported having no disclosures.
AT AES 2013
Major finding: Only 46% of reproductive-age women with epilepsy who were considered to be at risk for becoming pregnant were taking a folic acid supplement.
Data source: A Web-based survey involving 650 women.
Disclosures: Dr. Herzog reported having no disclosures.
Nursing while taking antiepileptic drugs appears safe
WASHINGTON – Exposure to antiepileptic drugs via breast milk during infancy was not associated with adverse neurodevelopmental effects at age 6 years in the prospective, observational Neurodevelopmental Effects of Antiepileptic Drugs study.
In fact, overall, the children in the study who were exposed to antiepileptic drugs (AEDs) via breast milk exhibited higher IQ and Verbal Index scores than did those who did not breastfeed (adjusted mean IQ scores of 108 vs. 104, and adjusted mean Verbal Index scores of 105 vs. 102, respectively), Dr. Kimford J. Meador of Stanford (Calif.) University reported at the annual meeting of the American Epilepsy Society.
Of 181 children in the multicenter study, 43% were breastfed for an average duration of 7.2 months. AED exposures included carbamazepine, lamotrigine, valproate, or phenytoin. IQ scores were higher for breastfed vs. nonbreastfed children for each of these drug groups except phenytoin (108 vs. 105; 114 vs. 110; 105 vs. 107; and 105 vs. 94, respectively). Significant independent variables predicting age 6 IQ included maternal IQ, AED group, AED dose, periconceptional folate, and breastfeeding.
Scores on cognitive domains other than the Verbal Index, including Nonverbal Index, Memory Index, and Executive Index, also were slightly higher for breastfed vs. nonbreastfed infants, but the differences did not reach statistical significance, Dr. Meador said.
The Neurodevelopmental Effects of Antiepileptic Drugs (NEAD) study, conducted in the United States and the United Kingdom, included pregnant women with epilepsy who were on AED monotherapy. The women were enrolled between 1999 and 2004, and they continued taking AEDs after delivery. Their children were assessed at age 6 years via the Differential Abilities Scales (DAS) and other measures of cognitive ability. Mean scores were adjusted for maternal IQ, AED group, AED dose, and periconceptional folate.
NEAD follows an earlier study that looked at outcomes in the children at age 3 years (Neurology 2010;75:1954-60). That report also demonstrated that children exposed to AEDs via breast milk experienced no adverse neurodevelopmental effects, but outcomes at age 6 years are considered more predictive of school performance and adult ability, Dr. Meador explained during a press briefing at the meeting.
The findings are among the first to evaluate neurodevelopmental outcomes following AED exposure via breast milk, and although they don’t provide a definitive answer about the safety of AED exposure via breast milk, they do provide reassurance for concerned mothers weighing the well-known benefits of breastfeeding with the potential risks of infant AED exposure, he said.
Dr. Meador said that he – along with the NEAD study group – recommends breastfeeding for women who were already taking AEDs during pregnancy, but noted that the findings do not address those who begin taking AEDs after delivery.
"Our recommendation is based on the known positive effects of breastfeeding, the results of our study, and unsubstantiated theoretical risk, and theoretical reasons why breastfeeding on AEDs would not offer additional risk," he concluded.
NEAD was supported by grants from the National Institutes of Health to Dr. Meador and to his coauthor, Dr. N. Browning, and from the U.K. Epilepsy Research Foundation to coauthor Dr. G. A. Baker.
WASHINGTON – Exposure to antiepileptic drugs via breast milk during infancy was not associated with adverse neurodevelopmental effects at age 6 years in the prospective, observational Neurodevelopmental Effects of Antiepileptic Drugs study.
In fact, overall, the children in the study who were exposed to antiepileptic drugs (AEDs) via breast milk exhibited higher IQ and Verbal Index scores than did those who did not breastfeed (adjusted mean IQ scores of 108 vs. 104, and adjusted mean Verbal Index scores of 105 vs. 102, respectively), Dr. Kimford J. Meador of Stanford (Calif.) University reported at the annual meeting of the American Epilepsy Society.
Of 181 children in the multicenter study, 43% were breastfed for an average duration of 7.2 months. AED exposures included carbamazepine, lamotrigine, valproate, or phenytoin. IQ scores were higher for breastfed vs. nonbreastfed children for each of these drug groups except phenytoin (108 vs. 105; 114 vs. 110; 105 vs. 107; and 105 vs. 94, respectively). Significant independent variables predicting age 6 IQ included maternal IQ, AED group, AED dose, periconceptional folate, and breastfeeding.
Scores on cognitive domains other than the Verbal Index, including Nonverbal Index, Memory Index, and Executive Index, also were slightly higher for breastfed vs. nonbreastfed infants, but the differences did not reach statistical significance, Dr. Meador said.
The Neurodevelopmental Effects of Antiepileptic Drugs (NEAD) study, conducted in the United States and the United Kingdom, included pregnant women with epilepsy who were on AED monotherapy. The women were enrolled between 1999 and 2004, and they continued taking AEDs after delivery. Their children were assessed at age 6 years via the Differential Abilities Scales (DAS) and other measures of cognitive ability. Mean scores were adjusted for maternal IQ, AED group, AED dose, and periconceptional folate.
NEAD follows an earlier study that looked at outcomes in the children at age 3 years (Neurology 2010;75:1954-60). That report also demonstrated that children exposed to AEDs via breast milk experienced no adverse neurodevelopmental effects, but outcomes at age 6 years are considered more predictive of school performance and adult ability, Dr. Meador explained during a press briefing at the meeting.
The findings are among the first to evaluate neurodevelopmental outcomes following AED exposure via breast milk, and although they don’t provide a definitive answer about the safety of AED exposure via breast milk, they do provide reassurance for concerned mothers weighing the well-known benefits of breastfeeding with the potential risks of infant AED exposure, he said.
Dr. Meador said that he – along with the NEAD study group – recommends breastfeeding for women who were already taking AEDs during pregnancy, but noted that the findings do not address those who begin taking AEDs after delivery.
"Our recommendation is based on the known positive effects of breastfeeding, the results of our study, and unsubstantiated theoretical risk, and theoretical reasons why breastfeeding on AEDs would not offer additional risk," he concluded.
NEAD was supported by grants from the National Institutes of Health to Dr. Meador and to his coauthor, Dr. N. Browning, and from the U.K. Epilepsy Research Foundation to coauthor Dr. G. A. Baker.
WASHINGTON – Exposure to antiepileptic drugs via breast milk during infancy was not associated with adverse neurodevelopmental effects at age 6 years in the prospective, observational Neurodevelopmental Effects of Antiepileptic Drugs study.
In fact, overall, the children in the study who were exposed to antiepileptic drugs (AEDs) via breast milk exhibited higher IQ and Verbal Index scores than did those who did not breastfeed (adjusted mean IQ scores of 108 vs. 104, and adjusted mean Verbal Index scores of 105 vs. 102, respectively), Dr. Kimford J. Meador of Stanford (Calif.) University reported at the annual meeting of the American Epilepsy Society.
Of 181 children in the multicenter study, 43% were breastfed for an average duration of 7.2 months. AED exposures included carbamazepine, lamotrigine, valproate, or phenytoin. IQ scores were higher for breastfed vs. nonbreastfed children for each of these drug groups except phenytoin (108 vs. 105; 114 vs. 110; 105 vs. 107; and 105 vs. 94, respectively). Significant independent variables predicting age 6 IQ included maternal IQ, AED group, AED dose, periconceptional folate, and breastfeeding.
Scores on cognitive domains other than the Verbal Index, including Nonverbal Index, Memory Index, and Executive Index, also were slightly higher for breastfed vs. nonbreastfed infants, but the differences did not reach statistical significance, Dr. Meador said.
The Neurodevelopmental Effects of Antiepileptic Drugs (NEAD) study, conducted in the United States and the United Kingdom, included pregnant women with epilepsy who were on AED monotherapy. The women were enrolled between 1999 and 2004, and they continued taking AEDs after delivery. Their children were assessed at age 6 years via the Differential Abilities Scales (DAS) and other measures of cognitive ability. Mean scores were adjusted for maternal IQ, AED group, AED dose, and periconceptional folate.
NEAD follows an earlier study that looked at outcomes in the children at age 3 years (Neurology 2010;75:1954-60). That report also demonstrated that children exposed to AEDs via breast milk experienced no adverse neurodevelopmental effects, but outcomes at age 6 years are considered more predictive of school performance and adult ability, Dr. Meador explained during a press briefing at the meeting.
The findings are among the first to evaluate neurodevelopmental outcomes following AED exposure via breast milk, and although they don’t provide a definitive answer about the safety of AED exposure via breast milk, they do provide reassurance for concerned mothers weighing the well-known benefits of breastfeeding with the potential risks of infant AED exposure, he said.
Dr. Meador said that he – along with the NEAD study group – recommends breastfeeding for women who were already taking AEDs during pregnancy, but noted that the findings do not address those who begin taking AEDs after delivery.
"Our recommendation is based on the known positive effects of breastfeeding, the results of our study, and unsubstantiated theoretical risk, and theoretical reasons why breastfeeding on AEDs would not offer additional risk," he concluded.
NEAD was supported by grants from the National Institutes of Health to Dr. Meador and to his coauthor, Dr. N. Browning, and from the U.K. Epilepsy Research Foundation to coauthor Dr. G. A. Baker.
AT AES 2013
Major finding: IQ and Verbal Index scores were higher among children exposed to AEDs via breast milk vs. those not breastfed (adjusted mean IQ scores of 108 vs. 104, and adjusted mean Verbal Index scores of 105 vs. 102, respectively).
Data source: A prospective, observational study involving 181 children either exposed or unexposed to antiepileptic drugs via breast milk.
Disclosures: NEAD was supported by grants from the National Institutes of Health to Dr. Meador and to his coauthor, Dr. N. Browning, and from the UK Epilepsy Research Foundation to coauthor Dr. G. A. Baker.
Anesthetic drugs for status epilepticus linked with death, infection risk
WASHINGTON – The use of anesthetic drugs for the treatment of refractory status epilepticus was associated with a high risk of infection and death among patients treated over a 6-year period at a tertiary academic medical center.
Although prospective, randomized controlled trials are needed to confirm the findings of this retrospective cohort study, the data should heighten awareness of the potential adverse effects of anesthetic drugs, Dr. Peter W. Kaplan said at the annual meeting of the American Epilepsy Society.
Of 171 adults who presented to University Hospital Basel, Switzerland, with status epilepticus between January 2005 and January 2011, 63 (37%) were treated with intravenous anesthetic drugs (IVADs), including thiopental, midazolam, propofol, and/or high-dose phenobarbital, and 18% of the patients died. After adjustment for status epilepticus duration and severity (based on status epilepticus severity score) and for critical medical conditions, those treated with IVADs had a significantly increased risk of death, compared with those who did not receive IVADs (relative risk = 3.16), said Dr. Kaplan of Johns Hopkins University, Baltimore.
Those who received IVADs also had a significantly higher rate of infectious complications (43% vs. 11%). The infections were diagnosed during the course of treatment, and 25 of 27 infections were respiratory infections. The remaining two cases were urinary tract infections.
No significant difference was seen between the groups with respect to seizure control, but ICU stay, a secondary outcome of the study, was significantly longer in those who received IVADs (13.5 days vs. 4.5 days), he said.
Severe hypotension also occurred more often in the patients who received IVADs.
Patients included in the study were adults with a mean age of 64 years. Those who received continuously administered IVADs were refractory to first- and second-line antiepileptic drugs and were treated according to a standard protocol; 29 received midazolam only, 22 received midazolam followed by propofol, and 12 received barbiturates after midazolam. The use of nonanesthetic antiepileptic drugs was similar in the two groups.
The findings, published online Dec. 6 in Neurology ahead of their presentation at the AES meeting, are important because data regarding the risks and benefits of anesthetic drugs for status epilepticus are lacking (Neurology 2013 Dec. 6 [doi:10.1212/WNL.0000000000000009]).
In the Neurology report, principal investigator Dr. Raoul Sutter of University Hospital Basel and his colleagues – including Dr. Kaplan – noted that "most opinion leaders recommend IVADs ... for refractory status epilepticus to induce total seizure suppression, an EEG burst-suppression pattern, or an isoelectric EEG," but explained that "the Neurocritical Care Society outlines the role of IVADs, but notes the lack of supporting data, while the European Federation of Neurological Societies points to the need for further study.
"In our cohort, the relation between the use of IVADs and death was not modified by different grades of status epilepticus severity, suggesting that the association of IVADs with an increased risk of death did not depend on to whom or when, but on the fact that IVADs were used," they wrote.
However, while the findings provide "class 3 evidence that patients with status epilepticus receiving IVADs have a higher proportion of infections and an increased risk of death, compared with those who did not receive IVADs," the findings should be considered preliminary until the association between IVADs and these outcomes are confirmed in prospective randomized trials, Dr. Kaplan said.
Dr. Kaplan reported having no disclosures. Dr. Sutter is supported by the Research Fund of University Basel, the Scientific Society Basel, and the Gottfried Julia Bangerter-Rhyner Foundation. He has held stock from Novartis and Roche since 2005.
WASHINGTON – The use of anesthetic drugs for the treatment of refractory status epilepticus was associated with a high risk of infection and death among patients treated over a 6-year period at a tertiary academic medical center.
Although prospective, randomized controlled trials are needed to confirm the findings of this retrospective cohort study, the data should heighten awareness of the potential adverse effects of anesthetic drugs, Dr. Peter W. Kaplan said at the annual meeting of the American Epilepsy Society.
Of 171 adults who presented to University Hospital Basel, Switzerland, with status epilepticus between January 2005 and January 2011, 63 (37%) were treated with intravenous anesthetic drugs (IVADs), including thiopental, midazolam, propofol, and/or high-dose phenobarbital, and 18% of the patients died. After adjustment for status epilepticus duration and severity (based on status epilepticus severity score) and for critical medical conditions, those treated with IVADs had a significantly increased risk of death, compared with those who did not receive IVADs (relative risk = 3.16), said Dr. Kaplan of Johns Hopkins University, Baltimore.
Those who received IVADs also had a significantly higher rate of infectious complications (43% vs. 11%). The infections were diagnosed during the course of treatment, and 25 of 27 infections were respiratory infections. The remaining two cases were urinary tract infections.
No significant difference was seen between the groups with respect to seizure control, but ICU stay, a secondary outcome of the study, was significantly longer in those who received IVADs (13.5 days vs. 4.5 days), he said.
Severe hypotension also occurred more often in the patients who received IVADs.
Patients included in the study were adults with a mean age of 64 years. Those who received continuously administered IVADs were refractory to first- and second-line antiepileptic drugs and were treated according to a standard protocol; 29 received midazolam only, 22 received midazolam followed by propofol, and 12 received barbiturates after midazolam. The use of nonanesthetic antiepileptic drugs was similar in the two groups.
The findings, published online Dec. 6 in Neurology ahead of their presentation at the AES meeting, are important because data regarding the risks and benefits of anesthetic drugs for status epilepticus are lacking (Neurology 2013 Dec. 6 [doi:10.1212/WNL.0000000000000009]).
In the Neurology report, principal investigator Dr. Raoul Sutter of University Hospital Basel and his colleagues – including Dr. Kaplan – noted that "most opinion leaders recommend IVADs ... for refractory status epilepticus to induce total seizure suppression, an EEG burst-suppression pattern, or an isoelectric EEG," but explained that "the Neurocritical Care Society outlines the role of IVADs, but notes the lack of supporting data, while the European Federation of Neurological Societies points to the need for further study.
"In our cohort, the relation between the use of IVADs and death was not modified by different grades of status epilepticus severity, suggesting that the association of IVADs with an increased risk of death did not depend on to whom or when, but on the fact that IVADs were used," they wrote.
However, while the findings provide "class 3 evidence that patients with status epilepticus receiving IVADs have a higher proportion of infections and an increased risk of death, compared with those who did not receive IVADs," the findings should be considered preliminary until the association between IVADs and these outcomes are confirmed in prospective randomized trials, Dr. Kaplan said.
Dr. Kaplan reported having no disclosures. Dr. Sutter is supported by the Research Fund of University Basel, the Scientific Society Basel, and the Gottfried Julia Bangerter-Rhyner Foundation. He has held stock from Novartis and Roche since 2005.
WASHINGTON – The use of anesthetic drugs for the treatment of refractory status epilepticus was associated with a high risk of infection and death among patients treated over a 6-year period at a tertiary academic medical center.
Although prospective, randomized controlled trials are needed to confirm the findings of this retrospective cohort study, the data should heighten awareness of the potential adverse effects of anesthetic drugs, Dr. Peter W. Kaplan said at the annual meeting of the American Epilepsy Society.
Of 171 adults who presented to University Hospital Basel, Switzerland, with status epilepticus between January 2005 and January 2011, 63 (37%) were treated with intravenous anesthetic drugs (IVADs), including thiopental, midazolam, propofol, and/or high-dose phenobarbital, and 18% of the patients died. After adjustment for status epilepticus duration and severity (based on status epilepticus severity score) and for critical medical conditions, those treated with IVADs had a significantly increased risk of death, compared with those who did not receive IVADs (relative risk = 3.16), said Dr. Kaplan of Johns Hopkins University, Baltimore.
Those who received IVADs also had a significantly higher rate of infectious complications (43% vs. 11%). The infections were diagnosed during the course of treatment, and 25 of 27 infections were respiratory infections. The remaining two cases were urinary tract infections.
No significant difference was seen between the groups with respect to seizure control, but ICU stay, a secondary outcome of the study, was significantly longer in those who received IVADs (13.5 days vs. 4.5 days), he said.
Severe hypotension also occurred more often in the patients who received IVADs.
Patients included in the study were adults with a mean age of 64 years. Those who received continuously administered IVADs were refractory to first- and second-line antiepileptic drugs and were treated according to a standard protocol; 29 received midazolam only, 22 received midazolam followed by propofol, and 12 received barbiturates after midazolam. The use of nonanesthetic antiepileptic drugs was similar in the two groups.
The findings, published online Dec. 6 in Neurology ahead of their presentation at the AES meeting, are important because data regarding the risks and benefits of anesthetic drugs for status epilepticus are lacking (Neurology 2013 Dec. 6 [doi:10.1212/WNL.0000000000000009]).
In the Neurology report, principal investigator Dr. Raoul Sutter of University Hospital Basel and his colleagues – including Dr. Kaplan – noted that "most opinion leaders recommend IVADs ... for refractory status epilepticus to induce total seizure suppression, an EEG burst-suppression pattern, or an isoelectric EEG," but explained that "the Neurocritical Care Society outlines the role of IVADs, but notes the lack of supporting data, while the European Federation of Neurological Societies points to the need for further study.
"In our cohort, the relation between the use of IVADs and death was not modified by different grades of status epilepticus severity, suggesting that the association of IVADs with an increased risk of death did not depend on to whom or when, but on the fact that IVADs were used," they wrote.
However, while the findings provide "class 3 evidence that patients with status epilepticus receiving IVADs have a higher proportion of infections and an increased risk of death, compared with those who did not receive IVADs," the findings should be considered preliminary until the association between IVADs and these outcomes are confirmed in prospective randomized trials, Dr. Kaplan said.
Dr. Kaplan reported having no disclosures. Dr. Sutter is supported by the Research Fund of University Basel, the Scientific Society Basel, and the Gottfried Julia Bangerter-Rhyner Foundation. He has held stock from Novartis and Roche since 2005.
AT AES 2013
Major finding: Patients treated with IVADs vs. those not treated with IVADs had an increased risk of death (relative risk = 3.16) and higher infection rate (43% vs. 11%).
Data source: A retrospective cohort study involving 171 patients.
Disclosures: Dr. Kaplan reported having no disclosures. Dr. Sutter is supported by the Research Fund of the University Basel, the Scientific Society Basel, and the Gottfried Julia Bangerter-Rhyner Foundation. He has held stock from Novartis and Roche since 2005.
Novel agent appears safe, effective for hyperkalemia
ATLANTA – A novel first-in-class agent that selectively binds cations appears safe and effective for the treatment of hyperkalemia in patients with chronic kidney disease, according to findings from a phase II, dose-ranging proof-of-concept study.
Preliminary data from a phase III study of 753 patients, which was initiated based on the positive findings in the phase II study, similarly demonstrate the safety and efficacy of the selective monovalent cation trap known as ZS-9 (ZS Pharma), Dr. Stephen R. Ash reported at Kidney Week 2013, sponsored by the American Society of Nephrology.
In 90 patients from the phase II study who were randomized to receive either placebo or one of three ZS-9 doses (0.3 g in 12 patients, 3 g in 24 patients, and 10 g in 24 patients), active treatment was associated with significantly greater dose-dependent reductions in serum potassium (K+) than was placebo, said Dr. Ash of Indiana University, Lafayette.
The mean serum K+ level at baseline for both the placebo group and the combined active treatment groups was 5.1 mEq/L. Within 48 hours of treatment initiation, 63% of patients in the 10-g ZS-9 group met the primary end point of at least a 1.0-mEq/L reduction in serum K+, compared with only 17% of patients in the placebo group.
The dose reduction in the 10-g group was "rapid and substantial," with a significant decrease, compared with placebo, occurring within 1 hour of the initial dose. Levels were 0.92-mEq/L lower than baseline after 38 hours (4 hours after the last dose), and 0.68-mEq/L lower than baseline at 48 hours (14 hours after the last dose). The levels remained significantly lower than placebo for an additional 3.5 days after the last dose, Dr. Ash said.
"At every point in the trial for these 10-g–dose patients, the serum potassium was lower in the treatment group than in the control group. ... At 38 hours, almost 90% of the patients who were on the 10-g dose had a serum potassium [level] of less than 4.5," he said.
Participants had mild to moderate chronic kidney disease with a glomerular filtration rate of 30-60 mL/min per 1.73 m2, and mild to moderate hyperkalemia, with serum K+ levels of 5-6 mEq/L.
Treatment with the tasteless, odorless substance, which was delivered orally as a suspension in water three times daily at meal times, was well tolerated. No serious adverse events were reported, and no significant gastrointestinal issues, hypokalemia, hypomagnesemia, or hypocalcemia were observed.
Only one adverse event, a mild case of constipation in the 3-g–dose group, was thought to have a causal relationship to the study drug. No patients withdrew from the study.
The preliminary data from the pivotal randomized, controlled phase III trial also suggest a rapid and dose-dependent reduction in serum K+ in patients treated with ZS-9. In that study, patients were randomized to receive placebo or ZS-9 at doses of 1.25 g, 2.5 g, 5 g, or 10 g three times daily for the initial 48 hours.
Patients whose serum K+ levels normalized during that initial phase of treatment were then randomized to receive placebo or one of the four active drug doses administered daily for 12 days.
Patients in the phase III trial had serum K+ levels of 5-6.5 mEq/L. Those in the 2.5-g, 5-g, and 10-g groups met the primary endpoint, with significantly greater reductions in serum K+, compared with those on placebo, at 48 hours.
The mean serum K+ reduction was 0.73 mEq/L at the 10-g dose at 48 hours. The gastrointestinal adverse event rate was 5.1% in the placebo group patients, and 3.5% in the treatment group.
Final results from the phase III trial are expected in the coming months.
The findings are important because currently only one treatment – sodium polystyrene sulfonate (SPS), an organic polymer resin that nonselectively binds cations – is approved in the United States for the treatment of hyperkalemia – a serious condition that is associated with significant mortality and morbidity in patients with cardiovascular disease or chronic kidney disease.
"Even marginally high potassium levels, such as over 4.5, however, have an increased risk of death and ventricular fibrillation," Dr. Ash explained.
Further, hyperkalemia limits the ability to use cardioprotective and renoprotective agents such as angiotensin-converting enzyme inhibitors and angiotensin receptor blockers, he said.
The efficacy of SPS for lowering serum K+ levels is questionable, and the agent has been associated with several serious adverse effects, including sodium loading, colonic necrosis, and other fatal gastrointestinal effects, Dr. Ash explained.
ZS-9 differs from traditional nonspecific and nonselective cation exchange organic polymer resins in that it is an inorganic cation exchanger – a result of advances in chemistry for the development of highly selective drug therapies.
"ZS-9 was designed and engineered to be highly selective for potassium," he said, noting that ZS-9 has more than nine times the potassium-binding ability of SPS, and is not systemically absorbed; thus, the risk of systemic toxicity is minimized or eliminated.
The findings thus far suggest that ZS-9 is a safe, reliable, fast, effective and well-tolerated therapy for lowering serum K+ levels in patients with hyperkalemia, Dr. Ash concluded.
Dr. Ash disclosed ties with Merit Medical, HemoCleanse (which has a minority share interest in ZS Pharma), Fresenius Medical, Ash Access Technology (in which he has ownership interest), Renal Solutions, and DaVita RMS. He also is editor-in-chief of Seminars in Dialysis, ASKIN section.
ATLANTA – A novel first-in-class agent that selectively binds cations appears safe and effective for the treatment of hyperkalemia in patients with chronic kidney disease, according to findings from a phase II, dose-ranging proof-of-concept study.
Preliminary data from a phase III study of 753 patients, which was initiated based on the positive findings in the phase II study, similarly demonstrate the safety and efficacy of the selective monovalent cation trap known as ZS-9 (ZS Pharma), Dr. Stephen R. Ash reported at Kidney Week 2013, sponsored by the American Society of Nephrology.
In 90 patients from the phase II study who were randomized to receive either placebo or one of three ZS-9 doses (0.3 g in 12 patients, 3 g in 24 patients, and 10 g in 24 patients), active treatment was associated with significantly greater dose-dependent reductions in serum potassium (K+) than was placebo, said Dr. Ash of Indiana University, Lafayette.
The mean serum K+ level at baseline for both the placebo group and the combined active treatment groups was 5.1 mEq/L. Within 48 hours of treatment initiation, 63% of patients in the 10-g ZS-9 group met the primary end point of at least a 1.0-mEq/L reduction in serum K+, compared with only 17% of patients in the placebo group.
The dose reduction in the 10-g group was "rapid and substantial," with a significant decrease, compared with placebo, occurring within 1 hour of the initial dose. Levels were 0.92-mEq/L lower than baseline after 38 hours (4 hours after the last dose), and 0.68-mEq/L lower than baseline at 48 hours (14 hours after the last dose). The levels remained significantly lower than placebo for an additional 3.5 days after the last dose, Dr. Ash said.
"At every point in the trial for these 10-g–dose patients, the serum potassium was lower in the treatment group than in the control group. ... At 38 hours, almost 90% of the patients who were on the 10-g dose had a serum potassium [level] of less than 4.5," he said.
Participants had mild to moderate chronic kidney disease with a glomerular filtration rate of 30-60 mL/min per 1.73 m2, and mild to moderate hyperkalemia, with serum K+ levels of 5-6 mEq/L.
Treatment with the tasteless, odorless substance, which was delivered orally as a suspension in water three times daily at meal times, was well tolerated. No serious adverse events were reported, and no significant gastrointestinal issues, hypokalemia, hypomagnesemia, or hypocalcemia were observed.
Only one adverse event, a mild case of constipation in the 3-g–dose group, was thought to have a causal relationship to the study drug. No patients withdrew from the study.
The preliminary data from the pivotal randomized, controlled phase III trial also suggest a rapid and dose-dependent reduction in serum K+ in patients treated with ZS-9. In that study, patients were randomized to receive placebo or ZS-9 at doses of 1.25 g, 2.5 g, 5 g, or 10 g three times daily for the initial 48 hours.
Patients whose serum K+ levels normalized during that initial phase of treatment were then randomized to receive placebo or one of the four active drug doses administered daily for 12 days.
Patients in the phase III trial had serum K+ levels of 5-6.5 mEq/L. Those in the 2.5-g, 5-g, and 10-g groups met the primary endpoint, with significantly greater reductions in serum K+, compared with those on placebo, at 48 hours.
The mean serum K+ reduction was 0.73 mEq/L at the 10-g dose at 48 hours. The gastrointestinal adverse event rate was 5.1% in the placebo group patients, and 3.5% in the treatment group.
Final results from the phase III trial are expected in the coming months.
The findings are important because currently only one treatment – sodium polystyrene sulfonate (SPS), an organic polymer resin that nonselectively binds cations – is approved in the United States for the treatment of hyperkalemia – a serious condition that is associated with significant mortality and morbidity in patients with cardiovascular disease or chronic kidney disease.
"Even marginally high potassium levels, such as over 4.5, however, have an increased risk of death and ventricular fibrillation," Dr. Ash explained.
Further, hyperkalemia limits the ability to use cardioprotective and renoprotective agents such as angiotensin-converting enzyme inhibitors and angiotensin receptor blockers, he said.
The efficacy of SPS for lowering serum K+ levels is questionable, and the agent has been associated with several serious adverse effects, including sodium loading, colonic necrosis, and other fatal gastrointestinal effects, Dr. Ash explained.
ZS-9 differs from traditional nonspecific and nonselective cation exchange organic polymer resins in that it is an inorganic cation exchanger – a result of advances in chemistry for the development of highly selective drug therapies.
"ZS-9 was designed and engineered to be highly selective for potassium," he said, noting that ZS-9 has more than nine times the potassium-binding ability of SPS, and is not systemically absorbed; thus, the risk of systemic toxicity is minimized or eliminated.
The findings thus far suggest that ZS-9 is a safe, reliable, fast, effective and well-tolerated therapy for lowering serum K+ levels in patients with hyperkalemia, Dr. Ash concluded.
Dr. Ash disclosed ties with Merit Medical, HemoCleanse (which has a minority share interest in ZS Pharma), Fresenius Medical, Ash Access Technology (in which he has ownership interest), Renal Solutions, and DaVita RMS. He also is editor-in-chief of Seminars in Dialysis, ASKIN section.
ATLANTA – A novel first-in-class agent that selectively binds cations appears safe and effective for the treatment of hyperkalemia in patients with chronic kidney disease, according to findings from a phase II, dose-ranging proof-of-concept study.
Preliminary data from a phase III study of 753 patients, which was initiated based on the positive findings in the phase II study, similarly demonstrate the safety and efficacy of the selective monovalent cation trap known as ZS-9 (ZS Pharma), Dr. Stephen R. Ash reported at Kidney Week 2013, sponsored by the American Society of Nephrology.
In 90 patients from the phase II study who were randomized to receive either placebo or one of three ZS-9 doses (0.3 g in 12 patients, 3 g in 24 patients, and 10 g in 24 patients), active treatment was associated with significantly greater dose-dependent reductions in serum potassium (K+) than was placebo, said Dr. Ash of Indiana University, Lafayette.
The mean serum K+ level at baseline for both the placebo group and the combined active treatment groups was 5.1 mEq/L. Within 48 hours of treatment initiation, 63% of patients in the 10-g ZS-9 group met the primary end point of at least a 1.0-mEq/L reduction in serum K+, compared with only 17% of patients in the placebo group.
The dose reduction in the 10-g group was "rapid and substantial," with a significant decrease, compared with placebo, occurring within 1 hour of the initial dose. Levels were 0.92-mEq/L lower than baseline after 38 hours (4 hours after the last dose), and 0.68-mEq/L lower than baseline at 48 hours (14 hours after the last dose). The levels remained significantly lower than placebo for an additional 3.5 days after the last dose, Dr. Ash said.
"At every point in the trial for these 10-g–dose patients, the serum potassium was lower in the treatment group than in the control group. ... At 38 hours, almost 90% of the patients who were on the 10-g dose had a serum potassium [level] of less than 4.5," he said.
Participants had mild to moderate chronic kidney disease with a glomerular filtration rate of 30-60 mL/min per 1.73 m2, and mild to moderate hyperkalemia, with serum K+ levels of 5-6 mEq/L.
Treatment with the tasteless, odorless substance, which was delivered orally as a suspension in water three times daily at meal times, was well tolerated. No serious adverse events were reported, and no significant gastrointestinal issues, hypokalemia, hypomagnesemia, or hypocalcemia were observed.
Only one adverse event, a mild case of constipation in the 3-g–dose group, was thought to have a causal relationship to the study drug. No patients withdrew from the study.
The preliminary data from the pivotal randomized, controlled phase III trial also suggest a rapid and dose-dependent reduction in serum K+ in patients treated with ZS-9. In that study, patients were randomized to receive placebo or ZS-9 at doses of 1.25 g, 2.5 g, 5 g, or 10 g three times daily for the initial 48 hours.
Patients whose serum K+ levels normalized during that initial phase of treatment were then randomized to receive placebo or one of the four active drug doses administered daily for 12 days.
Patients in the phase III trial had serum K+ levels of 5-6.5 mEq/L. Those in the 2.5-g, 5-g, and 10-g groups met the primary endpoint, with significantly greater reductions in serum K+, compared with those on placebo, at 48 hours.
The mean serum K+ reduction was 0.73 mEq/L at the 10-g dose at 48 hours. The gastrointestinal adverse event rate was 5.1% in the placebo group patients, and 3.5% in the treatment group.
Final results from the phase III trial are expected in the coming months.
The findings are important because currently only one treatment – sodium polystyrene sulfonate (SPS), an organic polymer resin that nonselectively binds cations – is approved in the United States for the treatment of hyperkalemia – a serious condition that is associated with significant mortality and morbidity in patients with cardiovascular disease or chronic kidney disease.
"Even marginally high potassium levels, such as over 4.5, however, have an increased risk of death and ventricular fibrillation," Dr. Ash explained.
Further, hyperkalemia limits the ability to use cardioprotective and renoprotective agents such as angiotensin-converting enzyme inhibitors and angiotensin receptor blockers, he said.
The efficacy of SPS for lowering serum K+ levels is questionable, and the agent has been associated with several serious adverse effects, including sodium loading, colonic necrosis, and other fatal gastrointestinal effects, Dr. Ash explained.
ZS-9 differs from traditional nonspecific and nonselective cation exchange organic polymer resins in that it is an inorganic cation exchanger – a result of advances in chemistry for the development of highly selective drug therapies.
"ZS-9 was designed and engineered to be highly selective for potassium," he said, noting that ZS-9 has more than nine times the potassium-binding ability of SPS, and is not systemically absorbed; thus, the risk of systemic toxicity is minimized or eliminated.
The findings thus far suggest that ZS-9 is a safe, reliable, fast, effective and well-tolerated therapy for lowering serum K+ levels in patients with hyperkalemia, Dr. Ash concluded.
Dr. Ash disclosed ties with Merit Medical, HemoCleanse (which has a minority share interest in ZS Pharma), Fresenius Medical, Ash Access Technology (in which he has ownership interest), Renal Solutions, and DaVita RMS. He also is editor-in-chief of Seminars in Dialysis, ASKIN section.
AT KIDNEY WEEK 2013
Major finding: Within 48 hours, 63% of patients in the group who got a 10-g dose of ZS-9 met the primary end point of at least a 1.0-mEq/L reduction in serum K+, compared with only 17% of patients in the placebo group (phase II results).
Data source: A phase II study of 90 patients; a phase III study of 753 patients.
Disclosures: Dr. Ash disclosed ties with Merit Medical, HemoCleanse (which has a minority share interest in ZS Pharma), Fresenius Medical, Ash Access Technology (in which he has ownership interest), Renal Solutions, and DaVita RMS. He also is editor-in-chief of Seminars in Dialysis, ASKIN section.
Persistent electrographic seizures occur in one-third of convulsive status epilepticus patients
WASHINGTON – Electrographic seizures persisted following control of convulsive status epilepticus in about a third of children treated in pediatric intensive care units at 11 hospitals, according to a retrospective study.
Of 98 children aged 1 month to 21 years (median, 5 years) who underwent continuous electroencephalographic monitoring (typically for 12-48 hours) after presenting with convulsive status epilepticus (CSE), 32 (33%) had electrographic seizures following control of CSE, including 11 (34%) who had electroencephalographic-only seizures, and 17 (53%) who had electroclinical seizures. In four patients (13%), data regarding clinical correlates were unavailable, Dr. Iván Sánchez Fernández reported at the annual meeting of the American Epilepsy Society.
Factors significantly associated with persistent electrographic seizures were prior diagnosis of epilepsy (P = .029) and the presence of interictal epileptiform discharges (P less than .0005), but about a third of children with persistent electrographic seizures had no clinical correlates associated with the seizure, said Dr. Sánchez Fernández of the department of neurology at Boston Children’s Hospital and Harvard Medical School, Boston.
Few data looking at the rate of electrographic seizures after CSE are available. The current findings are among the first to look at this and to characterize persistent electrographic seizures after CSE, he said.
Of the 32 patients who had electrographic seizures, 15 (47%) had electrographic status epilepticus, including 6 patients (40%) with continuous status epilepticus and 9 (60%) with intermittent status epilepticus. A comparison of patients with electrographic seizures who had status epilepticus and patients with electrographic seizures who did not have status epilepticus, as well as with those who had no electrographic seizures, showed that the presence of an abnormal initial background category on continuous electroencephalogram and the presence of sporadic interictal epileptiform discharges were significantly associated with electrographic status epilepticus, he said.
Slightly more than half (53) of the children included in this study were boys. The typical duration of electrographic seizures was less than 1 minute in 34% of cases, 1-5 minutes in 31% of cases, 6-30 minutes in 22% of cases, and more than 30 minutes in 13% of cases. Seizure onset localization was focal in 41% of cases, multifocal in 20.5% of cases, generalized in 30.8% of cases, and unknown in 7.7% of cases. Maximal spread localization was focal-unilateral in 38% of cases, bilateral in 50% of cases, and unknown in 12% of cases.
Patients with electrographic seizures also stayed in the ICU longer than did those without such seizures. The median ICU stay was 9.5 days for those with electrographic seizures, compared with only 2 days for those without electrographic seizures, but it is unclear from these data whether the seizures were the cause of the prolonged stay, Dr. Sánchez Fernández said.
No disclosures were available at press time.
WASHINGTON – Electrographic seizures persisted following control of convulsive status epilepticus in about a third of children treated in pediatric intensive care units at 11 hospitals, according to a retrospective study.
Of 98 children aged 1 month to 21 years (median, 5 years) who underwent continuous electroencephalographic monitoring (typically for 12-48 hours) after presenting with convulsive status epilepticus (CSE), 32 (33%) had electrographic seizures following control of CSE, including 11 (34%) who had electroencephalographic-only seizures, and 17 (53%) who had electroclinical seizures. In four patients (13%), data regarding clinical correlates were unavailable, Dr. Iván Sánchez Fernández reported at the annual meeting of the American Epilepsy Society.
Factors significantly associated with persistent electrographic seizures were prior diagnosis of epilepsy (P = .029) and the presence of interictal epileptiform discharges (P less than .0005), but about a third of children with persistent electrographic seizures had no clinical correlates associated with the seizure, said Dr. Sánchez Fernández of the department of neurology at Boston Children’s Hospital and Harvard Medical School, Boston.
Few data looking at the rate of electrographic seizures after CSE are available. The current findings are among the first to look at this and to characterize persistent electrographic seizures after CSE, he said.
Of the 32 patients who had electrographic seizures, 15 (47%) had electrographic status epilepticus, including 6 patients (40%) with continuous status epilepticus and 9 (60%) with intermittent status epilepticus. A comparison of patients with electrographic seizures who had status epilepticus and patients with electrographic seizures who did not have status epilepticus, as well as with those who had no electrographic seizures, showed that the presence of an abnormal initial background category on continuous electroencephalogram and the presence of sporadic interictal epileptiform discharges were significantly associated with electrographic status epilepticus, he said.
Slightly more than half (53) of the children included in this study were boys. The typical duration of electrographic seizures was less than 1 minute in 34% of cases, 1-5 minutes in 31% of cases, 6-30 minutes in 22% of cases, and more than 30 minutes in 13% of cases. Seizure onset localization was focal in 41% of cases, multifocal in 20.5% of cases, generalized in 30.8% of cases, and unknown in 7.7% of cases. Maximal spread localization was focal-unilateral in 38% of cases, bilateral in 50% of cases, and unknown in 12% of cases.
Patients with electrographic seizures also stayed in the ICU longer than did those without such seizures. The median ICU stay was 9.5 days for those with electrographic seizures, compared with only 2 days for those without electrographic seizures, but it is unclear from these data whether the seizures were the cause of the prolonged stay, Dr. Sánchez Fernández said.
No disclosures were available at press time.
WASHINGTON – Electrographic seizures persisted following control of convulsive status epilepticus in about a third of children treated in pediatric intensive care units at 11 hospitals, according to a retrospective study.
Of 98 children aged 1 month to 21 years (median, 5 years) who underwent continuous electroencephalographic monitoring (typically for 12-48 hours) after presenting with convulsive status epilepticus (CSE), 32 (33%) had electrographic seizures following control of CSE, including 11 (34%) who had electroencephalographic-only seizures, and 17 (53%) who had electroclinical seizures. In four patients (13%), data regarding clinical correlates were unavailable, Dr. Iván Sánchez Fernández reported at the annual meeting of the American Epilepsy Society.
Factors significantly associated with persistent electrographic seizures were prior diagnosis of epilepsy (P = .029) and the presence of interictal epileptiform discharges (P less than .0005), but about a third of children with persistent electrographic seizures had no clinical correlates associated with the seizure, said Dr. Sánchez Fernández of the department of neurology at Boston Children’s Hospital and Harvard Medical School, Boston.
Few data looking at the rate of electrographic seizures after CSE are available. The current findings are among the first to look at this and to characterize persistent electrographic seizures after CSE, he said.
Of the 32 patients who had electrographic seizures, 15 (47%) had electrographic status epilepticus, including 6 patients (40%) with continuous status epilepticus and 9 (60%) with intermittent status epilepticus. A comparison of patients with electrographic seizures who had status epilepticus and patients with electrographic seizures who did not have status epilepticus, as well as with those who had no electrographic seizures, showed that the presence of an abnormal initial background category on continuous electroencephalogram and the presence of sporadic interictal epileptiform discharges were significantly associated with electrographic status epilepticus, he said.
Slightly more than half (53) of the children included in this study were boys. The typical duration of electrographic seizures was less than 1 minute in 34% of cases, 1-5 minutes in 31% of cases, 6-30 minutes in 22% of cases, and more than 30 minutes in 13% of cases. Seizure onset localization was focal in 41% of cases, multifocal in 20.5% of cases, generalized in 30.8% of cases, and unknown in 7.7% of cases. Maximal spread localization was focal-unilateral in 38% of cases, bilateral in 50% of cases, and unknown in 12% of cases.
Patients with electrographic seizures also stayed in the ICU longer than did those without such seizures. The median ICU stay was 9.5 days for those with electrographic seizures, compared with only 2 days for those without electrographic seizures, but it is unclear from these data whether the seizures were the cause of the prolonged stay, Dr. Sánchez Fernández said.
No disclosures were available at press time.
AT AES 2013
Major finding: Factors significantly associated with persistent electrographic seizures were prior diagnosis of epilepsy (P = .029) and the presence of interictal epileptiform discharges (P less than .0005).
Data source: A retrospective, multicenter study of 98 children with convulsive status epilepticus.
Disclosures: No disclosures were available at press time.
Drug selection, timing for refractory convulsive status epilepticus need improvement
WASHINGTON – The choice of antiepileptic drugs for the treatment of pediatric convulsive status epilepticus was highly variable, and treatment timing and escalation were less than ideal in a pediatric Status Epilepticus Research Group study.
The findings suggest there is "room for improvement" in the management of this serious, life-threatening condition through more timely administration of antiepileptic drugs (AEDs) and through quicker progression from one AED to the next, Dr. Tobias Loddenkemper reported at the annual meeting of the American Epilepsy Society.
The findings represent a first step in addressing the inadequacies in the care of children with refractory convulsive status epilepticus (RCSE), and they underscore the need for parental education and ensuring that rescue medications are available for children with an epilepsy diagnosis, as well as the need for improved management in the inpatient setting, said Dr. Loddenkemper of Boston Children’s Hospital.
The analysis of RCSE episodes in 81 children aged 1 month to 21 years (mean age, 2.5 years) who were treated in the intensive care unit at any of the 11 U.S. tertiary referral hospitals that comprise the pediatric Status Epilepticus Research Group (pSERG) network showed that most episodes (79%) had onset out of the hospital. The median time to emergency medical services arrival was 15 minutes, and the median time to hospital arrival was 47 minutes. The median times to various steps of treatment far exceeded current guideline recommendations, according to Dr. Loddenkemper:
• 30 minutes for the administration of a first benzodiazepine drug by intravenous bolus dosing; guidelines call for treatment within 5 minutes.
• 69 minutes for the administration of a second drug; guidelines recommend 5-10 minutes.
• 120 minutes for the administration of a second non-benzodiazepine drug; guidelines call for 20-30 minutes.
• 180 minutes for the continuous infusion of anesthetic drugs; guidelines call for continuous infusion to begin within 30-70 minutes.
However, delays also occurred for in-hospital RCSE episodes, he said. The median times to first and second drugs for in-hospital episodes were 10 and 20 minutes, respectively.
Of the 81 patients included in the analysis, 75% were intubated and only 67% returned to baseline functioning after the RCSE episode. Four patients (4.9%) died.
"This [mortality] signifies how important this is," he said of RCSE during a press briefing at the meeting, noting that it is the most severe form of epilepsy, and that prompt and appropriate treatment is important for improving outcomes.
Of particular concern was the finding that most of the children in this study had a preexisting diagnosis of epilepsy, yet only half received a rescue medication in the outpatient setting.
"Technically, they should have had some kind of rescue medications, but they either were not given or were not prescribed," he said.
As for drug selection in this study, the most frequent choice of first benzodiazepine was lorazepam in 56% of cases, followed by diazepam in about a third of patients, and midazolam and clobazam in just a few cases each. The choice for a second benzodiazepine was most often lorazepam in 70% of cases, followed by midazolam and diazepam.
The choice of a first non-benzodiazepine drug was fosphenytoin in 52% of cases, followed by phenytoin and levetiracetam, Dr. Loddenkemper said. Phenobarbital, valproate, rufinamide, and propofol were also among the choices for first non-benzodiazepine drug. The choice of a second non-benzodiazepine drug was phenobarbital in 45% of cases, followed by levetiracetam, fosphenytoin, valproate, phenytoin, and propofol. The choice of anesthetic drug for continuous drug infusion was midazolam in 75% of cases. Other drugs used for continuous infusion included pentobarbital, propofol, and isoflurane. Pentobarbital was the first choice for second drug for infusion in 38% of cases.
The first choice of drug in each of those stages of treatment was generally within guidelines, but the high variability with respect to drug choices is concerning, he said.
All consecutive children presenting with RCSE to the ICUs in the pSERG network were included as long as they either failed at least two AEDs or required continuous infusion of an AED drug to abort their seizures.
Quicker progression from one drug to another in RCSE cases and coadministration of the first and second drugs are among strategies for improving outcomes. "Maybe we just have to treat faster," he suggested.
He also suggested that care could be improved by the use of dedicated treatment algorithms and dedicated teams of physicians who take care of patients with RCSE – much like the successful model for stroke care used at many hospitals in the United States.
Additional analysis from this study could shed more light on methods for improving outcomes. Data from an additional 46 patients are currently being analyzed, Dr. Loddenkemper said.
This study was funded by the American Epilepsy Society Infrastructure Award and the Epilepsy Foundation of America. Dr. Loddenkemper reported receiving research funding from Eisai and Lundbeck.
WASHINGTON – The choice of antiepileptic drugs for the treatment of pediatric convulsive status epilepticus was highly variable, and treatment timing and escalation were less than ideal in a pediatric Status Epilepticus Research Group study.
The findings suggest there is "room for improvement" in the management of this serious, life-threatening condition through more timely administration of antiepileptic drugs (AEDs) and through quicker progression from one AED to the next, Dr. Tobias Loddenkemper reported at the annual meeting of the American Epilepsy Society.
The findings represent a first step in addressing the inadequacies in the care of children with refractory convulsive status epilepticus (RCSE), and they underscore the need for parental education and ensuring that rescue medications are available for children with an epilepsy diagnosis, as well as the need for improved management in the inpatient setting, said Dr. Loddenkemper of Boston Children’s Hospital.
The analysis of RCSE episodes in 81 children aged 1 month to 21 years (mean age, 2.5 years) who were treated in the intensive care unit at any of the 11 U.S. tertiary referral hospitals that comprise the pediatric Status Epilepticus Research Group (pSERG) network showed that most episodes (79%) had onset out of the hospital. The median time to emergency medical services arrival was 15 minutes, and the median time to hospital arrival was 47 minutes. The median times to various steps of treatment far exceeded current guideline recommendations, according to Dr. Loddenkemper:
• 30 minutes for the administration of a first benzodiazepine drug by intravenous bolus dosing; guidelines call for treatment within 5 minutes.
• 69 minutes for the administration of a second drug; guidelines recommend 5-10 minutes.
• 120 minutes for the administration of a second non-benzodiazepine drug; guidelines call for 20-30 minutes.
• 180 minutes for the continuous infusion of anesthetic drugs; guidelines call for continuous infusion to begin within 30-70 minutes.
However, delays also occurred for in-hospital RCSE episodes, he said. The median times to first and second drugs for in-hospital episodes were 10 and 20 minutes, respectively.
Of the 81 patients included in the analysis, 75% were intubated and only 67% returned to baseline functioning after the RCSE episode. Four patients (4.9%) died.
"This [mortality] signifies how important this is," he said of RCSE during a press briefing at the meeting, noting that it is the most severe form of epilepsy, and that prompt and appropriate treatment is important for improving outcomes.
Of particular concern was the finding that most of the children in this study had a preexisting diagnosis of epilepsy, yet only half received a rescue medication in the outpatient setting.
"Technically, they should have had some kind of rescue medications, but they either were not given or were not prescribed," he said.
As for drug selection in this study, the most frequent choice of first benzodiazepine was lorazepam in 56% of cases, followed by diazepam in about a third of patients, and midazolam and clobazam in just a few cases each. The choice for a second benzodiazepine was most often lorazepam in 70% of cases, followed by midazolam and diazepam.
The choice of a first non-benzodiazepine drug was fosphenytoin in 52% of cases, followed by phenytoin and levetiracetam, Dr. Loddenkemper said. Phenobarbital, valproate, rufinamide, and propofol were also among the choices for first non-benzodiazepine drug. The choice of a second non-benzodiazepine drug was phenobarbital in 45% of cases, followed by levetiracetam, fosphenytoin, valproate, phenytoin, and propofol. The choice of anesthetic drug for continuous drug infusion was midazolam in 75% of cases. Other drugs used for continuous infusion included pentobarbital, propofol, and isoflurane. Pentobarbital was the first choice for second drug for infusion in 38% of cases.
The first choice of drug in each of those stages of treatment was generally within guidelines, but the high variability with respect to drug choices is concerning, he said.
All consecutive children presenting with RCSE to the ICUs in the pSERG network were included as long as they either failed at least two AEDs or required continuous infusion of an AED drug to abort their seizures.
Quicker progression from one drug to another in RCSE cases and coadministration of the first and second drugs are among strategies for improving outcomes. "Maybe we just have to treat faster," he suggested.
He also suggested that care could be improved by the use of dedicated treatment algorithms and dedicated teams of physicians who take care of patients with RCSE – much like the successful model for stroke care used at many hospitals in the United States.
Additional analysis from this study could shed more light on methods for improving outcomes. Data from an additional 46 patients are currently being analyzed, Dr. Loddenkemper said.
This study was funded by the American Epilepsy Society Infrastructure Award and the Epilepsy Foundation of America. Dr. Loddenkemper reported receiving research funding from Eisai and Lundbeck.
WASHINGTON – The choice of antiepileptic drugs for the treatment of pediatric convulsive status epilepticus was highly variable, and treatment timing and escalation were less than ideal in a pediatric Status Epilepticus Research Group study.
The findings suggest there is "room for improvement" in the management of this serious, life-threatening condition through more timely administration of antiepileptic drugs (AEDs) and through quicker progression from one AED to the next, Dr. Tobias Loddenkemper reported at the annual meeting of the American Epilepsy Society.
The findings represent a first step in addressing the inadequacies in the care of children with refractory convulsive status epilepticus (RCSE), and they underscore the need for parental education and ensuring that rescue medications are available for children with an epilepsy diagnosis, as well as the need for improved management in the inpatient setting, said Dr. Loddenkemper of Boston Children’s Hospital.
The analysis of RCSE episodes in 81 children aged 1 month to 21 years (mean age, 2.5 years) who were treated in the intensive care unit at any of the 11 U.S. tertiary referral hospitals that comprise the pediatric Status Epilepticus Research Group (pSERG) network showed that most episodes (79%) had onset out of the hospital. The median time to emergency medical services arrival was 15 minutes, and the median time to hospital arrival was 47 minutes. The median times to various steps of treatment far exceeded current guideline recommendations, according to Dr. Loddenkemper:
• 30 minutes for the administration of a first benzodiazepine drug by intravenous bolus dosing; guidelines call for treatment within 5 minutes.
• 69 minutes for the administration of a second drug; guidelines recommend 5-10 minutes.
• 120 minutes for the administration of a second non-benzodiazepine drug; guidelines call for 20-30 minutes.
• 180 minutes for the continuous infusion of anesthetic drugs; guidelines call for continuous infusion to begin within 30-70 minutes.
However, delays also occurred for in-hospital RCSE episodes, he said. The median times to first and second drugs for in-hospital episodes were 10 and 20 minutes, respectively.
Of the 81 patients included in the analysis, 75% were intubated and only 67% returned to baseline functioning after the RCSE episode. Four patients (4.9%) died.
"This [mortality] signifies how important this is," he said of RCSE during a press briefing at the meeting, noting that it is the most severe form of epilepsy, and that prompt and appropriate treatment is important for improving outcomes.
Of particular concern was the finding that most of the children in this study had a preexisting diagnosis of epilepsy, yet only half received a rescue medication in the outpatient setting.
"Technically, they should have had some kind of rescue medications, but they either were not given or were not prescribed," he said.
As for drug selection in this study, the most frequent choice of first benzodiazepine was lorazepam in 56% of cases, followed by diazepam in about a third of patients, and midazolam and clobazam in just a few cases each. The choice for a second benzodiazepine was most often lorazepam in 70% of cases, followed by midazolam and diazepam.
The choice of a first non-benzodiazepine drug was fosphenytoin in 52% of cases, followed by phenytoin and levetiracetam, Dr. Loddenkemper said. Phenobarbital, valproate, rufinamide, and propofol were also among the choices for first non-benzodiazepine drug. The choice of a second non-benzodiazepine drug was phenobarbital in 45% of cases, followed by levetiracetam, fosphenytoin, valproate, phenytoin, and propofol. The choice of anesthetic drug for continuous drug infusion was midazolam in 75% of cases. Other drugs used for continuous infusion included pentobarbital, propofol, and isoflurane. Pentobarbital was the first choice for second drug for infusion in 38% of cases.
The first choice of drug in each of those stages of treatment was generally within guidelines, but the high variability with respect to drug choices is concerning, he said.
All consecutive children presenting with RCSE to the ICUs in the pSERG network were included as long as they either failed at least two AEDs or required continuous infusion of an AED drug to abort their seizures.
Quicker progression from one drug to another in RCSE cases and coadministration of the first and second drugs are among strategies for improving outcomes. "Maybe we just have to treat faster," he suggested.
He also suggested that care could be improved by the use of dedicated treatment algorithms and dedicated teams of physicians who take care of patients with RCSE – much like the successful model for stroke care used at many hospitals in the United States.
Additional analysis from this study could shed more light on methods for improving outcomes. Data from an additional 46 patients are currently being analyzed, Dr. Loddenkemper said.
This study was funded by the American Epilepsy Society Infrastructure Award and the Epilepsy Foundation of America. Dr. Loddenkemper reported receiving research funding from Eisai and Lundbeck.
AT AES 2013
Major finding: Median time to first benzodiazepine drug was 30 minutes, versus a recommended time of 0-5 minutes.
Data source: An observational study of RCSE management at 11 tertiary referral hospitals.
Disclosures: This study was funded by the American Epilepsy Society Infrastructure Award and the Epilepsy Foundation of America. Dr. Loddenkemper reported receiving research funding from Eisai and Lundbeck.