User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
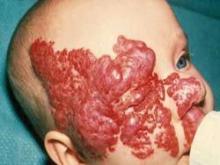
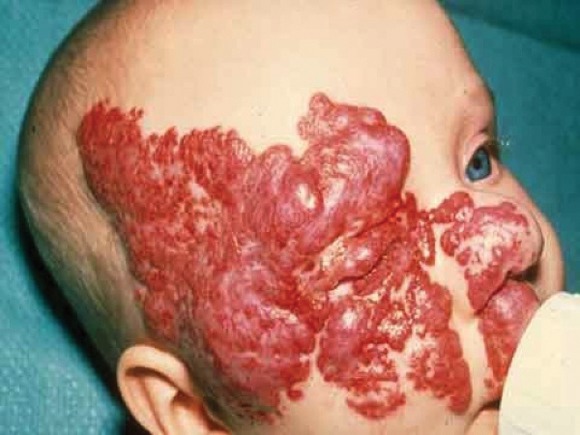
Propranolol safely clears most infantile hemangiomas
MIAMI BEACH – Approximately 60% of infants with proliferating hemangiomas showed significant improvement after treatment with oral propranolol, compared with a placebo, in a randomized, double-blind adaptive phase 2/3 study.
After 24 weeks, 61 of 101 patients (60.4%) treated daily with 3 mg/kg of propranolol had complete or nearly complete resolution of the hemangiomas, compared with only 2 of 55 patients (3.6%) who received a placebo, Dr. Christine Léauté-Labrèze of University Hospital of Bordeaux (France) reported at the annual meeting of the American Academy of Dermatology.
The complete or nearly complete resolution of hemangiomas was assessed by blinded independent investigators, based on a comparison of baseline photographs of the target lesions and photographs taken at week 24.
Patients in the international, multicenter study included infants aged 1-5 months with proliferating infantile hemangiomas requiring systemic therapy. Initially, 456 infants from 60 sites in 15 countries were randomized to receive one of four treatment regimens with oral propranolol: 1 or 3 mg/kg per day for either 3 or 6 months. After an interim efficacy and safety analysis based on the first 188 patients, only the treatment group receiving 3 mg/kg per day for 6 months was retained for trial completion.
The outcomes in both 3-month treatment arms were similar to those in the placebo group. However, the outcomes in the patients who received 3 mg/kg per day for 6 months were better – with no evidence of an increased risk of adverse events – than those in the patients who received 1 mg/kg per day for 6 months, reported Dr. Léauté-Labrèze, principal investigator in the study.
Of note, about half of the placebo patients dropped out of the study after 1 month because of nonefficacy; at the end of the study only about a third of the babies remained in the placebo arm. Similarly, in both of the 3-month treatment arms, dropout rates were high when patients switched from active treatment to placebo. However, approximately 80% of the patients in the group receiving 3 mg/kg per day for 6 months completed the study.
Treatment was well tolerated, and no unexpected safety signals emerged during the course of the study.
The findings of "a highly significant efficacy level" in this randomized, controlled study are important, because 5% of all babies born in the United States are affected by infantile hemangiomas, Dr. Léauté-Labrèze said. Approximately 12% of those babies will likely have complications such as ulceration, impairment of vision, or risk of disfigurement; therefore, effective treatment is needed, she said.
Although several smaller studies and open-label case series involving propranolol have demonstrated efficacy, no treatment has yet received Food and Drug Administration approval for the treatment of infantile hemangiomas, she added.
The study was sponsored by Pierre Fabre Dermatologie.
MIAMI BEACH – Approximately 60% of infants with proliferating hemangiomas showed significant improvement after treatment with oral propranolol, compared with a placebo, in a randomized, double-blind adaptive phase 2/3 study.
After 24 weeks, 61 of 101 patients (60.4%) treated daily with 3 mg/kg of propranolol had complete or nearly complete resolution of the hemangiomas, compared with only 2 of 55 patients (3.6%) who received a placebo, Dr. Christine Léauté-Labrèze of University Hospital of Bordeaux (France) reported at the annual meeting of the American Academy of Dermatology.
The complete or nearly complete resolution of hemangiomas was assessed by blinded independent investigators, based on a comparison of baseline photographs of the target lesions and photographs taken at week 24.
Patients in the international, multicenter study included infants aged 1-5 months with proliferating infantile hemangiomas requiring systemic therapy. Initially, 456 infants from 60 sites in 15 countries were randomized to receive one of four treatment regimens with oral propranolol: 1 or 3 mg/kg per day for either 3 or 6 months. After an interim efficacy and safety analysis based on the first 188 patients, only the treatment group receiving 3 mg/kg per day for 6 months was retained for trial completion.
The outcomes in both 3-month treatment arms were similar to those in the placebo group. However, the outcomes in the patients who received 3 mg/kg per day for 6 months were better – with no evidence of an increased risk of adverse events – than those in the patients who received 1 mg/kg per day for 6 months, reported Dr. Léauté-Labrèze, principal investigator in the study.
Of note, about half of the placebo patients dropped out of the study after 1 month because of nonefficacy; at the end of the study only about a third of the babies remained in the placebo arm. Similarly, in both of the 3-month treatment arms, dropout rates were high when patients switched from active treatment to placebo. However, approximately 80% of the patients in the group receiving 3 mg/kg per day for 6 months completed the study.
Treatment was well tolerated, and no unexpected safety signals emerged during the course of the study.
The findings of "a highly significant efficacy level" in this randomized, controlled study are important, because 5% of all babies born in the United States are affected by infantile hemangiomas, Dr. Léauté-Labrèze said. Approximately 12% of those babies will likely have complications such as ulceration, impairment of vision, or risk of disfigurement; therefore, effective treatment is needed, she said.
Although several smaller studies and open-label case series involving propranolol have demonstrated efficacy, no treatment has yet received Food and Drug Administration approval for the treatment of infantile hemangiomas, she added.
The study was sponsored by Pierre Fabre Dermatologie.
MIAMI BEACH – Approximately 60% of infants with proliferating hemangiomas showed significant improvement after treatment with oral propranolol, compared with a placebo, in a randomized, double-blind adaptive phase 2/3 study.
After 24 weeks, 61 of 101 patients (60.4%) treated daily with 3 mg/kg of propranolol had complete or nearly complete resolution of the hemangiomas, compared with only 2 of 55 patients (3.6%) who received a placebo, Dr. Christine Léauté-Labrèze of University Hospital of Bordeaux (France) reported at the annual meeting of the American Academy of Dermatology.
The complete or nearly complete resolution of hemangiomas was assessed by blinded independent investigators, based on a comparison of baseline photographs of the target lesions and photographs taken at week 24.
Patients in the international, multicenter study included infants aged 1-5 months with proliferating infantile hemangiomas requiring systemic therapy. Initially, 456 infants from 60 sites in 15 countries were randomized to receive one of four treatment regimens with oral propranolol: 1 or 3 mg/kg per day for either 3 or 6 months. After an interim efficacy and safety analysis based on the first 188 patients, only the treatment group receiving 3 mg/kg per day for 6 months was retained for trial completion.
The outcomes in both 3-month treatment arms were similar to those in the placebo group. However, the outcomes in the patients who received 3 mg/kg per day for 6 months were better – with no evidence of an increased risk of adverse events – than those in the patients who received 1 mg/kg per day for 6 months, reported Dr. Léauté-Labrèze, principal investigator in the study.
Of note, about half of the placebo patients dropped out of the study after 1 month because of nonefficacy; at the end of the study only about a third of the babies remained in the placebo arm. Similarly, in both of the 3-month treatment arms, dropout rates were high when patients switched from active treatment to placebo. However, approximately 80% of the patients in the group receiving 3 mg/kg per day for 6 months completed the study.
Treatment was well tolerated, and no unexpected safety signals emerged during the course of the study.
The findings of "a highly significant efficacy level" in this randomized, controlled study are important, because 5% of all babies born in the United States are affected by infantile hemangiomas, Dr. Léauté-Labrèze said. Approximately 12% of those babies will likely have complications such as ulceration, impairment of vision, or risk of disfigurement; therefore, effective treatment is needed, she said.
Although several smaller studies and open-label case series involving propranolol have demonstrated efficacy, no treatment has yet received Food and Drug Administration approval for the treatment of infantile hemangiomas, she added.
The study was sponsored by Pierre Fabre Dermatologie.
AT THE AAD ANNUAL MEETING
Major finding: Among children treated with propranolol, 60% had complete or nearly complete resolution of hemangiomas after 24 weeks, compared with 4% of children treated with placebo.
Data source: A randomized, double-blind, placebo-controlled adaptive phase 2/3 study.
Disclosures: This study was sponsored by Pierre Fabre Dermatologie.
Cognition predicts functional ability in older adults with schizophrenia
LOS ANGELES – Certain cognitive measures predict functional ability in older adults with schizophrenia, according to findings from a study involving 110 patients over age 50.
Cognitive deficits are a common and core feature of schizophrenia and are known to be a strong predictor of function in younger adults with the disease. These findings, which indicate that the same is true in older adults, highlight deficits that might be targets for intervention, Angel Y. Liu reported in a poster at the annual meeting of the American Association for Geriatric Psychiatry.
Three of seven tests that are part of the MATRICS Consensus Cognitive Battery – the Mayer-Salovey-Caruso Emotional Intelligence Test (MSCEIT), the Hopkins Verbal Learning Test Revised (HVLT-R), and the Trail Making Test (TMT) – were found on regression analysis to be more predictive of functional ability in 76 patients than in 34 healthy controls, said Ms. Liu of the University of Toronto and the Centre for Addiction and Mental Health, Toronto.
Specifically, the MSCEIT, a measure of social cognition, predicted scores for communication on the University of San Diego Performance-Based Skills Assessment–Brief (UPSA), which was used in the study to assess functional competence; the HVLT-R, a measure of verbal memory, predicted scores for finances on the UPSA and on the Medication Management Ability Assessment (MMAA), which also was used to assess functional competence; and the TMT predicted total MMAA score.
Patients in the study were clinically stable community-dwelling adults with a current diagnosis of schizophrenia, and controls were community-dwelling adults who did not meet the criteria for any mental disorder. The patients were more impaired than were controls on all clinical measures, including the Positive and Negative Syndrome Scale (mean scores of 55.3 v.32.8), the Abnormal Involuntary Movement Scale (mean scores of 2.7 vs. 0.5), the Cumulative Illness Rating Scale for Geriatrics (mean scores of 5.9 vs. 2.8), the Simpson Angus Scale (mean scores of 3.6 vs. 0.1), and the Subjective Well-Being on Neuroleptic Medications scale (mean scores of 88.1 vs. 106.5).
"The schizophrenia group was also impaired, compared with the control group, on cognitive and functional measures. While about half of the cognitive measures predicted functional ability in the schizophrenia group, none except one predicted functional ability in the control group," Ms. Liu noted.
Mean scores in the patients vs. controls on MSCEIT, HVLT-R, and the TMT, for example, were 86.8 vs. 98.3, 17.1 vs. 23.0, and 68.3 vs. 45.6, respectively, and mean scores on the UPSA Communication, UPSA Finances, and MMAA total were 127 vs. 16.1, 15.5 vs. 18.4, and 19.6 vs. 24.0, respectively.
Though limited by a ceiling effect in healthy controls, the findings suggest that the MSCEIT, the HVLT-R, and the TMT reveal deficits specific to schizophrenia, she said, noting that targeting these deficits with appropriate interventions could improve function in older patients with schizophrenia.
Identifying methods for improving the care of older patients with schizophrenia is particularly important given the aging of the population; an estimated 20% of patients with schizophrenia will be aged 65 years or older by 2025, she said.
Ms. Liu reported having no disclosures.
LOS ANGELES – Certain cognitive measures predict functional ability in older adults with schizophrenia, according to findings from a study involving 110 patients over age 50.
Cognitive deficits are a common and core feature of schizophrenia and are known to be a strong predictor of function in younger adults with the disease. These findings, which indicate that the same is true in older adults, highlight deficits that might be targets for intervention, Angel Y. Liu reported in a poster at the annual meeting of the American Association for Geriatric Psychiatry.
Three of seven tests that are part of the MATRICS Consensus Cognitive Battery – the Mayer-Salovey-Caruso Emotional Intelligence Test (MSCEIT), the Hopkins Verbal Learning Test Revised (HVLT-R), and the Trail Making Test (TMT) – were found on regression analysis to be more predictive of functional ability in 76 patients than in 34 healthy controls, said Ms. Liu of the University of Toronto and the Centre for Addiction and Mental Health, Toronto.
Specifically, the MSCEIT, a measure of social cognition, predicted scores for communication on the University of San Diego Performance-Based Skills Assessment–Brief (UPSA), which was used in the study to assess functional competence; the HVLT-R, a measure of verbal memory, predicted scores for finances on the UPSA and on the Medication Management Ability Assessment (MMAA), which also was used to assess functional competence; and the TMT predicted total MMAA score.
Patients in the study were clinically stable community-dwelling adults with a current diagnosis of schizophrenia, and controls were community-dwelling adults who did not meet the criteria for any mental disorder. The patients were more impaired than were controls on all clinical measures, including the Positive and Negative Syndrome Scale (mean scores of 55.3 v.32.8), the Abnormal Involuntary Movement Scale (mean scores of 2.7 vs. 0.5), the Cumulative Illness Rating Scale for Geriatrics (mean scores of 5.9 vs. 2.8), the Simpson Angus Scale (mean scores of 3.6 vs. 0.1), and the Subjective Well-Being on Neuroleptic Medications scale (mean scores of 88.1 vs. 106.5).
"The schizophrenia group was also impaired, compared with the control group, on cognitive and functional measures. While about half of the cognitive measures predicted functional ability in the schizophrenia group, none except one predicted functional ability in the control group," Ms. Liu noted.
Mean scores in the patients vs. controls on MSCEIT, HVLT-R, and the TMT, for example, were 86.8 vs. 98.3, 17.1 vs. 23.0, and 68.3 vs. 45.6, respectively, and mean scores on the UPSA Communication, UPSA Finances, and MMAA total were 127 vs. 16.1, 15.5 vs. 18.4, and 19.6 vs. 24.0, respectively.
Though limited by a ceiling effect in healthy controls, the findings suggest that the MSCEIT, the HVLT-R, and the TMT reveal deficits specific to schizophrenia, she said, noting that targeting these deficits with appropriate interventions could improve function in older patients with schizophrenia.
Identifying methods for improving the care of older patients with schizophrenia is particularly important given the aging of the population; an estimated 20% of patients with schizophrenia will be aged 65 years or older by 2025, she said.
Ms. Liu reported having no disclosures.
LOS ANGELES – Certain cognitive measures predict functional ability in older adults with schizophrenia, according to findings from a study involving 110 patients over age 50.
Cognitive deficits are a common and core feature of schizophrenia and are known to be a strong predictor of function in younger adults with the disease. These findings, which indicate that the same is true in older adults, highlight deficits that might be targets for intervention, Angel Y. Liu reported in a poster at the annual meeting of the American Association for Geriatric Psychiatry.
Three of seven tests that are part of the MATRICS Consensus Cognitive Battery – the Mayer-Salovey-Caruso Emotional Intelligence Test (MSCEIT), the Hopkins Verbal Learning Test Revised (HVLT-R), and the Trail Making Test (TMT) – were found on regression analysis to be more predictive of functional ability in 76 patients than in 34 healthy controls, said Ms. Liu of the University of Toronto and the Centre for Addiction and Mental Health, Toronto.
Specifically, the MSCEIT, a measure of social cognition, predicted scores for communication on the University of San Diego Performance-Based Skills Assessment–Brief (UPSA), which was used in the study to assess functional competence; the HVLT-R, a measure of verbal memory, predicted scores for finances on the UPSA and on the Medication Management Ability Assessment (MMAA), which also was used to assess functional competence; and the TMT predicted total MMAA score.
Patients in the study were clinically stable community-dwelling adults with a current diagnosis of schizophrenia, and controls were community-dwelling adults who did not meet the criteria for any mental disorder. The patients were more impaired than were controls on all clinical measures, including the Positive and Negative Syndrome Scale (mean scores of 55.3 v.32.8), the Abnormal Involuntary Movement Scale (mean scores of 2.7 vs. 0.5), the Cumulative Illness Rating Scale for Geriatrics (mean scores of 5.9 vs. 2.8), the Simpson Angus Scale (mean scores of 3.6 vs. 0.1), and the Subjective Well-Being on Neuroleptic Medications scale (mean scores of 88.1 vs. 106.5).
"The schizophrenia group was also impaired, compared with the control group, on cognitive and functional measures. While about half of the cognitive measures predicted functional ability in the schizophrenia group, none except one predicted functional ability in the control group," Ms. Liu noted.
Mean scores in the patients vs. controls on MSCEIT, HVLT-R, and the TMT, for example, were 86.8 vs. 98.3, 17.1 vs. 23.0, and 68.3 vs. 45.6, respectively, and mean scores on the UPSA Communication, UPSA Finances, and MMAA total were 127 vs. 16.1, 15.5 vs. 18.4, and 19.6 vs. 24.0, respectively.
Though limited by a ceiling effect in healthy controls, the findings suggest that the MSCEIT, the HVLT-R, and the TMT reveal deficits specific to schizophrenia, she said, noting that targeting these deficits with appropriate interventions could improve function in older patients with schizophrenia.
Identifying methods for improving the care of older patients with schizophrenia is particularly important given the aging of the population; an estimated 20% of patients with schizophrenia will be aged 65 years or older by 2025, she said.
Ms. Liu reported having no disclosures.
AT THE AAGP ANNUAL MEETING
Major finding: Three cognitive function tests were found to predict function in older adults. Mean scores in the patients vs. controls on MSCEIT, HVLT-R, and the TMT were 86.8 vs. 98.3, 17.1 vs. 23.0, and 68.3 vs. 45.6, respectively. Mean scores on the UPSA Communication, UPSA Finances, and MMAA total were 127 vs. 16.1, 15.5 vs. 18.4, and 19.6 vs. 24.0, respectively.
Data source: An evaluation of the relationship between cognition and function in 110 older adults with schizophrenia.
Disclosures: Ms. Liu reported having no disclosures.
Vismodegib shrinks BCCs, reduces Mohs defect size
MIAMI BEACH – Vismodegib treatment for 3 months prior to Mohs surgery for operable basal cell carcinomas shrunk tumors by 46% and reduced the Mohs defect size by 38%, based on data from an open-label intervention trial. The findings were reported at the annual meeting of the American Academy of Dermatology.
The estimated Mohs surgical defect area (based on tumor size) in the first five patients treated in the single-arm study decreased by a mean of 1.4 cm2 from baseline, and actual Mohs surgical defect size decreased by 1.1 cm2 after a mean of 3.4 months of treatment with vismodegib. The changes were statistically significant.
The Mohs defect size was considered a secondary endpoint, because actual defect size can be influenced by skin tension and lesion location, said Dr. Mina Ally, a research fellow at Stanford (Calif.) University.
Patients included in the study were five adults with a total of seven basal cell carcinomas (BCCs) of varying histologic subtypes. One occurred on the shoulder, and the rest occurred on the face; one was a recurrence. All patients were treated for at least 3 months at a vismodegib dosage of 150 mg daily, and all required only a single Mohs stage of excision.
Vismodegib, an oral hedgehog pathway inhibitor approved for the indefinite treatment of advanced and metastatic basal cell carcinomas (that result from aberrant hedgehog pathway signaling), was generally safe, said Dr. Ally.
"All patients experienced mild grade 1 side effects, including muscle cramps, hair loss, and taste loss, and we only needed to discontinue the medication early – after 2 months – in one patient due to a grade 2 elevation in liver enzymes," she said, noting that all of the adverse events resolved after treatment discontinuation.
Further sectioning of Mohs specimens revealed no evidence of residual BCC in three cases and residual BCC in one case. The diagnosis was equivocal in the remaining cases.
"This was because we were seeing an increased number of aberrant follicular structures after (vismodegib) treatment, which were sometimes difficult to differentiate from residual BCC," Dr. Ally explained.
Further staining using a panel that included pleckstrin homology-like domain, family A, member 1, a hair follicle stem cell marker, helped differentiate the follicular structures from BCC, she noted.
None of the patients experienced tumor recurrence during a median 5 months of follow-up.
The findings are of interest because about 1 million people in the United States are affected by BCCs each year, said Dr. Ally. While vismodegib appears to represent a useful adjuvant therapy when given for 3 months prior to Mohs surgery in some cases, certain challenges must be overcome, she said.
"Suppression of the Hedgehog pathway does appear to alter normal follicular development, and this was causing us difficulty in analyzing our histology specimens," Dr. Ally noted. "Future challenges really lie in interpreting these histology specimens and being able to differentiate these follicular structures from residual BCC, which can confound tumor margin clearance."
Dr. Ally reported having no disclosures.
MIAMI BEACH – Vismodegib treatment for 3 months prior to Mohs surgery for operable basal cell carcinomas shrunk tumors by 46% and reduced the Mohs defect size by 38%, based on data from an open-label intervention trial. The findings were reported at the annual meeting of the American Academy of Dermatology.
The estimated Mohs surgical defect area (based on tumor size) in the first five patients treated in the single-arm study decreased by a mean of 1.4 cm2 from baseline, and actual Mohs surgical defect size decreased by 1.1 cm2 after a mean of 3.4 months of treatment with vismodegib. The changes were statistically significant.
The Mohs defect size was considered a secondary endpoint, because actual defect size can be influenced by skin tension and lesion location, said Dr. Mina Ally, a research fellow at Stanford (Calif.) University.
Patients included in the study were five adults with a total of seven basal cell carcinomas (BCCs) of varying histologic subtypes. One occurred on the shoulder, and the rest occurred on the face; one was a recurrence. All patients were treated for at least 3 months at a vismodegib dosage of 150 mg daily, and all required only a single Mohs stage of excision.
Vismodegib, an oral hedgehog pathway inhibitor approved for the indefinite treatment of advanced and metastatic basal cell carcinomas (that result from aberrant hedgehog pathway signaling), was generally safe, said Dr. Ally.
"All patients experienced mild grade 1 side effects, including muscle cramps, hair loss, and taste loss, and we only needed to discontinue the medication early – after 2 months – in one patient due to a grade 2 elevation in liver enzymes," she said, noting that all of the adverse events resolved after treatment discontinuation.
Further sectioning of Mohs specimens revealed no evidence of residual BCC in three cases and residual BCC in one case. The diagnosis was equivocal in the remaining cases.
"This was because we were seeing an increased number of aberrant follicular structures after (vismodegib) treatment, which were sometimes difficult to differentiate from residual BCC," Dr. Ally explained.
Further staining using a panel that included pleckstrin homology-like domain, family A, member 1, a hair follicle stem cell marker, helped differentiate the follicular structures from BCC, she noted.
None of the patients experienced tumor recurrence during a median 5 months of follow-up.
The findings are of interest because about 1 million people in the United States are affected by BCCs each year, said Dr. Ally. While vismodegib appears to represent a useful adjuvant therapy when given for 3 months prior to Mohs surgery in some cases, certain challenges must be overcome, she said.
"Suppression of the Hedgehog pathway does appear to alter normal follicular development, and this was causing us difficulty in analyzing our histology specimens," Dr. Ally noted. "Future challenges really lie in interpreting these histology specimens and being able to differentiate these follicular structures from residual BCC, which can confound tumor margin clearance."
Dr. Ally reported having no disclosures.
MIAMI BEACH – Vismodegib treatment for 3 months prior to Mohs surgery for operable basal cell carcinomas shrunk tumors by 46% and reduced the Mohs defect size by 38%, based on data from an open-label intervention trial. The findings were reported at the annual meeting of the American Academy of Dermatology.
The estimated Mohs surgical defect area (based on tumor size) in the first five patients treated in the single-arm study decreased by a mean of 1.4 cm2 from baseline, and actual Mohs surgical defect size decreased by 1.1 cm2 after a mean of 3.4 months of treatment with vismodegib. The changes were statistically significant.
The Mohs defect size was considered a secondary endpoint, because actual defect size can be influenced by skin tension and lesion location, said Dr. Mina Ally, a research fellow at Stanford (Calif.) University.
Patients included in the study were five adults with a total of seven basal cell carcinomas (BCCs) of varying histologic subtypes. One occurred on the shoulder, and the rest occurred on the face; one was a recurrence. All patients were treated for at least 3 months at a vismodegib dosage of 150 mg daily, and all required only a single Mohs stage of excision.
Vismodegib, an oral hedgehog pathway inhibitor approved for the indefinite treatment of advanced and metastatic basal cell carcinomas (that result from aberrant hedgehog pathway signaling), was generally safe, said Dr. Ally.
"All patients experienced mild grade 1 side effects, including muscle cramps, hair loss, and taste loss, and we only needed to discontinue the medication early – after 2 months – in one patient due to a grade 2 elevation in liver enzymes," she said, noting that all of the adverse events resolved after treatment discontinuation.
Further sectioning of Mohs specimens revealed no evidence of residual BCC in three cases and residual BCC in one case. The diagnosis was equivocal in the remaining cases.
"This was because we were seeing an increased number of aberrant follicular structures after (vismodegib) treatment, which were sometimes difficult to differentiate from residual BCC," Dr. Ally explained.
Further staining using a panel that included pleckstrin homology-like domain, family A, member 1, a hair follicle stem cell marker, helped differentiate the follicular structures from BCC, she noted.
None of the patients experienced tumor recurrence during a median 5 months of follow-up.
The findings are of interest because about 1 million people in the United States are affected by BCCs each year, said Dr. Ally. While vismodegib appears to represent a useful adjuvant therapy when given for 3 months prior to Mohs surgery in some cases, certain challenges must be overcome, she said.
"Suppression of the Hedgehog pathway does appear to alter normal follicular development, and this was causing us difficulty in analyzing our histology specimens," Dr. Ally noted. "Future challenges really lie in interpreting these histology specimens and being able to differentiate these follicular structures from residual BCC, which can confound tumor margin clearance."
Dr. Ally reported having no disclosures.
AT THE AAD ANNUAL MEETING
Major finding: Vismodegib reduced tumor size by an average of 46% after 3 months.
Data source: Interim results from an open-label single-arm intervention study.
Disclosures: Dr. Ally reported having no disclosures.
For overweight children, inject epinephrine in lower thigh
SAN ANTONIO – Overweight and obese children in need of epinephrine for anaphylaxis should be injected in the calf or in the lower thigh, rather than upper half of the thigh, to ensure intramuscular administration, according to findings from an ultrasound study of 93 children.
Ultrasound measurement demonstrated that the distance from skin surface to muscle depth was greater than auto-injector needle length at one quarter of the distance down the thigh in 82% of obese children vs. 25% of nonobese children. At three-quarters of the way down the thigh, this was the case in only 17% of obese children and 2% of nonobese children, Dr. Peter Arkwright reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
At a point midway down the calf, the skin surface to muscle depth was less than the length of the auto-injector needle in all of the children, said Dr. Arkwright of the University of Manchester (England).
Intramuscular injection, rather than subcutaneous injection, is imperative for effective delivery of epinephrine, he said, noting that this study was undertaken because of growing concerns that increasing obesity among children could make existing auto-injectors inadequate for providing intramuscular delivery in a significant proportion of patients.
Children included in the study were patients from regional pediatric allergy clinics. All were measured for height, weight, and body mass index, and all underwent ultrasound measurement at set distances down the thigh and leg. Higher weight, BMI, and waist circumference – but not age or gender – were associated with skin surface to muscle depth greater than auto-injector needle length, he noted.
"Based on our study, injecting epinephrine into the lower rather than upper thigh would be advised in overweight or obese children," he said, adding that caregivers of children at risk of anaphylaxis should be advised about the importance of administering epinephrine into the muscle in the most effective way.
For overweight and obese children, this involves injecting into the lower half of the thigh, and for very obese children it involves injecting at the middle of the calf, he said.
Dr. Arkwright reported having no disclosures.
SAN ANTONIO – Overweight and obese children in need of epinephrine for anaphylaxis should be injected in the calf or in the lower thigh, rather than upper half of the thigh, to ensure intramuscular administration, according to findings from an ultrasound study of 93 children.
Ultrasound measurement demonstrated that the distance from skin surface to muscle depth was greater than auto-injector needle length at one quarter of the distance down the thigh in 82% of obese children vs. 25% of nonobese children. At three-quarters of the way down the thigh, this was the case in only 17% of obese children and 2% of nonobese children, Dr. Peter Arkwright reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
At a point midway down the calf, the skin surface to muscle depth was less than the length of the auto-injector needle in all of the children, said Dr. Arkwright of the University of Manchester (England).
Intramuscular injection, rather than subcutaneous injection, is imperative for effective delivery of epinephrine, he said, noting that this study was undertaken because of growing concerns that increasing obesity among children could make existing auto-injectors inadequate for providing intramuscular delivery in a significant proportion of patients.
Children included in the study were patients from regional pediatric allergy clinics. All were measured for height, weight, and body mass index, and all underwent ultrasound measurement at set distances down the thigh and leg. Higher weight, BMI, and waist circumference – but not age or gender – were associated with skin surface to muscle depth greater than auto-injector needle length, he noted.
"Based on our study, injecting epinephrine into the lower rather than upper thigh would be advised in overweight or obese children," he said, adding that caregivers of children at risk of anaphylaxis should be advised about the importance of administering epinephrine into the muscle in the most effective way.
For overweight and obese children, this involves injecting into the lower half of the thigh, and for very obese children it involves injecting at the middle of the calf, he said.
Dr. Arkwright reported having no disclosures.
SAN ANTONIO – Overweight and obese children in need of epinephrine for anaphylaxis should be injected in the calf or in the lower thigh, rather than upper half of the thigh, to ensure intramuscular administration, according to findings from an ultrasound study of 93 children.
Ultrasound measurement demonstrated that the distance from skin surface to muscle depth was greater than auto-injector needle length at one quarter of the distance down the thigh in 82% of obese children vs. 25% of nonobese children. At three-quarters of the way down the thigh, this was the case in only 17% of obese children and 2% of nonobese children, Dr. Peter Arkwright reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
At a point midway down the calf, the skin surface to muscle depth was less than the length of the auto-injector needle in all of the children, said Dr. Arkwright of the University of Manchester (England).
Intramuscular injection, rather than subcutaneous injection, is imperative for effective delivery of epinephrine, he said, noting that this study was undertaken because of growing concerns that increasing obesity among children could make existing auto-injectors inadequate for providing intramuscular delivery in a significant proportion of patients.
Children included in the study were patients from regional pediatric allergy clinics. All were measured for height, weight, and body mass index, and all underwent ultrasound measurement at set distances down the thigh and leg. Higher weight, BMI, and waist circumference – but not age or gender – were associated with skin surface to muscle depth greater than auto-injector needle length, he noted.
"Based on our study, injecting epinephrine into the lower rather than upper thigh would be advised in overweight or obese children," he said, adding that caregivers of children at risk of anaphylaxis should be advised about the importance of administering epinephrine into the muscle in the most effective way.
For overweight and obese children, this involves injecting into the lower half of the thigh, and for very obese children it involves injecting at the middle of the calf, he said.
Dr. Arkwright reported having no disclosures.
AT THE AAAAI ANNUAL MEETING
For overweight children, inject epinephrine in lower thigh
SAN ANTONIO – Overweight and obese children in need of epinephrine for anaphylaxis should be injected in the calf or in the lower thigh, rather than upper half of the thigh, to ensure intramuscular administration, according to findings from an ultrasound study of 93 children.
Ultrasound measurement demonstrated that the distance from skin surface to muscle depth was greater than auto-injector needle length at one quarter of the distance down the thigh in 82% of obese children vs. 25% of nonobese children. At three-quarters of the way down the thigh, this was the case in only 17% of obese children and 2% of nonobese children, Dr. Peter Arkwright reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
At a point midway down the calf, the skin surface to muscle depth was less than the length of the auto-injector needle in all of the children, said Dr. Arkwright of the University of Manchester (England).
Intramuscular injection, rather than subcutaneous injection, is imperative for effective delivery of epinephrine, he said, noting that this study was undertaken because of growing concerns that increasing obesity among children could make existing auto-injectors inadequate for providing intramuscular delivery in a significant proportion of patients.
Children included in the study were patients from regional pediatric allergy clinics. All were measured for height, weight, and body mass index, and all underwent ultrasound measurement at set distances down the thigh and leg. Higher weight, BMI, and waist circumference – but not age or gender – were associated with skin surface to muscle depth greater than auto-injector needle length, he noted.
"Based on our study, injecting epinephrine into the lower rather than upper thigh would be advised in overweight or obese children," he said, adding that caregivers of children at risk of anaphylaxis should be advised about the importance of administering epinephrine into the muscle in the most effective way.
For overweight and obese children, this involves injecting into the lower half of the thigh, and for very obese children it involves injecting at the middle of the calf, he said.
Dr. Arkwright reported having no disclosures.
SAN ANTONIO – Overweight and obese children in need of epinephrine for anaphylaxis should be injected in the calf or in the lower thigh, rather than upper half of the thigh, to ensure intramuscular administration, according to findings from an ultrasound study of 93 children.
Ultrasound measurement demonstrated that the distance from skin surface to muscle depth was greater than auto-injector needle length at one quarter of the distance down the thigh in 82% of obese children vs. 25% of nonobese children. At three-quarters of the way down the thigh, this was the case in only 17% of obese children and 2% of nonobese children, Dr. Peter Arkwright reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
At a point midway down the calf, the skin surface to muscle depth was less than the length of the auto-injector needle in all of the children, said Dr. Arkwright of the University of Manchester (England).
Intramuscular injection, rather than subcutaneous injection, is imperative for effective delivery of epinephrine, he said, noting that this study was undertaken because of growing concerns that increasing obesity among children could make existing auto-injectors inadequate for providing intramuscular delivery in a significant proportion of patients.
Children included in the study were patients from regional pediatric allergy clinics. All were measured for height, weight, and body mass index, and all underwent ultrasound measurement at set distances down the thigh and leg. Higher weight, BMI, and waist circumference – but not age or gender – were associated with skin surface to muscle depth greater than auto-injector needle length, he noted.
"Based on our study, injecting epinephrine into the lower rather than upper thigh would be advised in overweight or obese children," he said, adding that caregivers of children at risk of anaphylaxis should be advised about the importance of administering epinephrine into the muscle in the most effective way.
For overweight and obese children, this involves injecting into the lower half of the thigh, and for very obese children it involves injecting at the middle of the calf, he said.
Dr. Arkwright reported having no disclosures.
SAN ANTONIO – Overweight and obese children in need of epinephrine for anaphylaxis should be injected in the calf or in the lower thigh, rather than upper half of the thigh, to ensure intramuscular administration, according to findings from an ultrasound study of 93 children.
Ultrasound measurement demonstrated that the distance from skin surface to muscle depth was greater than auto-injector needle length at one quarter of the distance down the thigh in 82% of obese children vs. 25% of nonobese children. At three-quarters of the way down the thigh, this was the case in only 17% of obese children and 2% of nonobese children, Dr. Peter Arkwright reported at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
At a point midway down the calf, the skin surface to muscle depth was less than the length of the auto-injector needle in all of the children, said Dr. Arkwright of the University of Manchester (England).
Intramuscular injection, rather than subcutaneous injection, is imperative for effective delivery of epinephrine, he said, noting that this study was undertaken because of growing concerns that increasing obesity among children could make existing auto-injectors inadequate for providing intramuscular delivery in a significant proportion of patients.
Children included in the study were patients from regional pediatric allergy clinics. All were measured for height, weight, and body mass index, and all underwent ultrasound measurement at set distances down the thigh and leg. Higher weight, BMI, and waist circumference – but not age or gender – were associated with skin surface to muscle depth greater than auto-injector needle length, he noted.
"Based on our study, injecting epinephrine into the lower rather than upper thigh would be advised in overweight or obese children," he said, adding that caregivers of children at risk of anaphylaxis should be advised about the importance of administering epinephrine into the muscle in the most effective way.
For overweight and obese children, this involves injecting into the lower half of the thigh, and for very obese children it involves injecting at the middle of the calf, he said.
Dr. Arkwright reported having no disclosures.
AT THE AAAAI ANNUAL MEETING
Major finding: 82% of obese children and 25% of nonobese children had inadequate upper thigh skin surface to muscle depth.
Data source: A prospective study of 93 children.
Disclosures: Dr. Arkwright reported having no disclosures.
Collaborative care improves response in late-life depression
LOS ANGELES – A collaborative-care model for depression management resulted in significantly greater reductions in depression scores and treatment response, compared with usual care, among 186 older adults included in a records review.
After 6 months of treatment, the mean score on the nine-item depression scale of the Patient Health Questionnaire (PHQ-9) was significantly lower for 93 patients enrolled in the collaborative-care model than for 93 patients who received usual care (6.42 vs. 13.19). The mean improvement in PHQ-9 score from baseline also was significantly greater in the collaborative-care group, Dr. Ramona DeJesus reported in a poster at the annual meeting of the American Association for Geriatric Psychiatry.
Antidepressant medication use was similar in the two groups at 6 months, said Dr. DeJesus, an internist and assistant professor of medicine at the Mayo Clinic in Rochester, Minn.
Collaborative-care models that bring primary care providers and psychiatrists together to care for patients have been shown in numerous studies to be more effective than usual care for the management of depression. One such model – Depression Improvement Across Minnesota, Offering a New Direction (DIAMOND) – was implemented in March 2008 at primary care sites at the Mayo Clinic.
The DIAMOND program, which was developed by the Institute for Clinical Systems Improvement, provides for consistent patient assessment, a registry that tracks treatment response over time, and a focus on relapse prevention. The model involves a primary care manager and a liaison or consultative relationship with a psychiatrist, Dr. DeJesus explained in an interview.
In a prior study, she found that patients treated using the collaborative-care model had significantly greater use of antidepressant medications after 1 year, compared with those receiving usual care, but a subgroup analysis of those over age 65 years in that study did not yield similar results.
For the current study, medical records of patients aged 65 years and older who had received a diagnosis of depression between March 2008, when the DIAMOND program was implemented, and December 2010 were reviewed. All had a screening score of 10 or greater on the PHQ-9. The collaborative-care and usual-care patient groups were similar with respect to demographic characteristics, including age, gender, race, ethnicity, marital status, and baseline PHQ-9 score.
As in the earlier trial, these findings show that antidepressant use is not increased among older adults treated using a collaborative-care model. This might be a result of increased caution with respect to prescribing in older adults, Dr. DeJesus said.
"The collaborative-care model consistently demonstrates efficacy across both young and elderly populations," she said, noting that the current findings provide further support for use of this model in the elderly population.
Dr. DeJesus reported having no disclosures.
Antidepressant medication, psychiatrists, Depression Improvement Across Minnesota, Offering a New Direction, DIAMOND, Institute for Clinical Systems Improvement,
LOS ANGELES – A collaborative-care model for depression management resulted in significantly greater reductions in depression scores and treatment response, compared with usual care, among 186 older adults included in a records review.
After 6 months of treatment, the mean score on the nine-item depression scale of the Patient Health Questionnaire (PHQ-9) was significantly lower for 93 patients enrolled in the collaborative-care model than for 93 patients who received usual care (6.42 vs. 13.19). The mean improvement in PHQ-9 score from baseline also was significantly greater in the collaborative-care group, Dr. Ramona DeJesus reported in a poster at the annual meeting of the American Association for Geriatric Psychiatry.
Antidepressant medication use was similar in the two groups at 6 months, said Dr. DeJesus, an internist and assistant professor of medicine at the Mayo Clinic in Rochester, Minn.
Collaborative-care models that bring primary care providers and psychiatrists together to care for patients have been shown in numerous studies to be more effective than usual care for the management of depression. One such model – Depression Improvement Across Minnesota, Offering a New Direction (DIAMOND) – was implemented in March 2008 at primary care sites at the Mayo Clinic.
The DIAMOND program, which was developed by the Institute for Clinical Systems Improvement, provides for consistent patient assessment, a registry that tracks treatment response over time, and a focus on relapse prevention. The model involves a primary care manager and a liaison or consultative relationship with a psychiatrist, Dr. DeJesus explained in an interview.
In a prior study, she found that patients treated using the collaborative-care model had significantly greater use of antidepressant medications after 1 year, compared with those receiving usual care, but a subgroup analysis of those over age 65 years in that study did not yield similar results.
For the current study, medical records of patients aged 65 years and older who had received a diagnosis of depression between March 2008, when the DIAMOND program was implemented, and December 2010 were reviewed. All had a screening score of 10 or greater on the PHQ-9. The collaborative-care and usual-care patient groups were similar with respect to demographic characteristics, including age, gender, race, ethnicity, marital status, and baseline PHQ-9 score.
As in the earlier trial, these findings show that antidepressant use is not increased among older adults treated using a collaborative-care model. This might be a result of increased caution with respect to prescribing in older adults, Dr. DeJesus said.
"The collaborative-care model consistently demonstrates efficacy across both young and elderly populations," she said, noting that the current findings provide further support for use of this model in the elderly population.
Dr. DeJesus reported having no disclosures.
LOS ANGELES – A collaborative-care model for depression management resulted in significantly greater reductions in depression scores and treatment response, compared with usual care, among 186 older adults included in a records review.
After 6 months of treatment, the mean score on the nine-item depression scale of the Patient Health Questionnaire (PHQ-9) was significantly lower for 93 patients enrolled in the collaborative-care model than for 93 patients who received usual care (6.42 vs. 13.19). The mean improvement in PHQ-9 score from baseline also was significantly greater in the collaborative-care group, Dr. Ramona DeJesus reported in a poster at the annual meeting of the American Association for Geriatric Psychiatry.
Antidepressant medication use was similar in the two groups at 6 months, said Dr. DeJesus, an internist and assistant professor of medicine at the Mayo Clinic in Rochester, Minn.
Collaborative-care models that bring primary care providers and psychiatrists together to care for patients have been shown in numerous studies to be more effective than usual care for the management of depression. One such model – Depression Improvement Across Minnesota, Offering a New Direction (DIAMOND) – was implemented in March 2008 at primary care sites at the Mayo Clinic.
The DIAMOND program, which was developed by the Institute for Clinical Systems Improvement, provides for consistent patient assessment, a registry that tracks treatment response over time, and a focus on relapse prevention. The model involves a primary care manager and a liaison or consultative relationship with a psychiatrist, Dr. DeJesus explained in an interview.
In a prior study, she found that patients treated using the collaborative-care model had significantly greater use of antidepressant medications after 1 year, compared with those receiving usual care, but a subgroup analysis of those over age 65 years in that study did not yield similar results.
For the current study, medical records of patients aged 65 years and older who had received a diagnosis of depression between March 2008, when the DIAMOND program was implemented, and December 2010 were reviewed. All had a screening score of 10 or greater on the PHQ-9. The collaborative-care and usual-care patient groups were similar with respect to demographic characteristics, including age, gender, race, ethnicity, marital status, and baseline PHQ-9 score.
As in the earlier trial, these findings show that antidepressant use is not increased among older adults treated using a collaborative-care model. This might be a result of increased caution with respect to prescribing in older adults, Dr. DeJesus said.
"The collaborative-care model consistently demonstrates efficacy across both young and elderly populations," she said, noting that the current findings provide further support for use of this model in the elderly population.
Dr. DeJesus reported having no disclosures.
Antidepressant medication, psychiatrists, Depression Improvement Across Minnesota, Offering a New Direction, DIAMOND, Institute for Clinical Systems Improvement,
Antidepressant medication, psychiatrists, Depression Improvement Across Minnesota, Offering a New Direction, DIAMOND, Institute for Clinical Systems Improvement,
AT THE AAGP ANNUAL MEETING
Major finding: Six-month PHQ-9 scores were significantly lower with collaborative care vs. usual care (6.42 vs. 13.19).
Data source: Review of medical records of 186 patients.
Disclosures: Dr. DeJesus reported having no disclosures.
Escitalopram during cancer treatment reduces depression risk
LOS ANGELES – Prophylactic escitalopram reduces the risk for depression in patients undergoing treatment for head and neck cancer, according to findings from a randomized placebo-controlled trial involving 148 patients.
Of those randomized to receive the selective serotonin reuptake inhibitor, 10% developed moderate to severe depression during the course of their cancer treatment, compared with 24.6% of those who received placebo, Dr. William J. Burke reported in a poster at the annual meeting of the American Association for Geriatric Psychiatry.
After adjustment for age, baseline smoking status, and stratification variables, including gender, cancer site, cancer stage, and primary treatment modality, those receiving active treatment remained significantly less likely to develop depression (hazard ratio, 0.37), said Dr. Burke of the University of Nebraska Medical Center, Omaha.
Of note, patients undergoing radiotherapy as the primary treatment modality were significantly more likely than those undergoing surgery as the primary treatment modality to develop depression (hazard ratio, 3.6).
Patients in the escitalopram group who did not become depressed during the course of the study reported a significantly better quality of life for 3 consecutive months after escitalopram cessation than those in either the treatment or placebo group who did develop depression, Dr. Burke noted.
Patients included in this double-blind study had head and neck cancers and were about to enter cancer treatment. Depression was assessed using the QIDS-SR(Quick Inventory of Depressive Symptomatology-Self Rated).
The findings have important implications for the management of patients entering treatment for head and neck cancer, which can be a particularly devastating disease. For example, larynx and tongue cancers comprise only 2% of all cancers, but patients with these cancers commit 19% of all cancer-related suicides, Dr. Burke noted.
Up to half of all patients with head and neck cancer develop major depressive disorder, which can adversely affect adherence to treatment, reduce quality of life, and result in significant morbidity and reduced survival.
"Preventing depression during the course of cancer treatment may, thus, be of great benefit," he said.
The project was supported by a grant from the National Institute of Mental Health. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIMH or the National Institutes of Health. Additional support was provided by a research support fund grant from the Nebraska Medical Center and the University of Nebraska Medical Center. Forest Pharmaceuticals provided the matching placebo and drugs.
LOS ANGELES – Prophylactic escitalopram reduces the risk for depression in patients undergoing treatment for head and neck cancer, according to findings from a randomized placebo-controlled trial involving 148 patients.
Of those randomized to receive the selective serotonin reuptake inhibitor, 10% developed moderate to severe depression during the course of their cancer treatment, compared with 24.6% of those who received placebo, Dr. William J. Burke reported in a poster at the annual meeting of the American Association for Geriatric Psychiatry.
After adjustment for age, baseline smoking status, and stratification variables, including gender, cancer site, cancer stage, and primary treatment modality, those receiving active treatment remained significantly less likely to develop depression (hazard ratio, 0.37), said Dr. Burke of the University of Nebraska Medical Center, Omaha.
Of note, patients undergoing radiotherapy as the primary treatment modality were significantly more likely than those undergoing surgery as the primary treatment modality to develop depression (hazard ratio, 3.6).
Patients in the escitalopram group who did not become depressed during the course of the study reported a significantly better quality of life for 3 consecutive months after escitalopram cessation than those in either the treatment or placebo group who did develop depression, Dr. Burke noted.
Patients included in this double-blind study had head and neck cancers and were about to enter cancer treatment. Depression was assessed using the QIDS-SR(Quick Inventory of Depressive Symptomatology-Self Rated).
The findings have important implications for the management of patients entering treatment for head and neck cancer, which can be a particularly devastating disease. For example, larynx and tongue cancers comprise only 2% of all cancers, but patients with these cancers commit 19% of all cancer-related suicides, Dr. Burke noted.
Up to half of all patients with head and neck cancer develop major depressive disorder, which can adversely affect adherence to treatment, reduce quality of life, and result in significant morbidity and reduced survival.
"Preventing depression during the course of cancer treatment may, thus, be of great benefit," he said.
The project was supported by a grant from the National Institute of Mental Health. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIMH or the National Institutes of Health. Additional support was provided by a research support fund grant from the Nebraska Medical Center and the University of Nebraska Medical Center. Forest Pharmaceuticals provided the matching placebo and drugs.
LOS ANGELES – Prophylactic escitalopram reduces the risk for depression in patients undergoing treatment for head and neck cancer, according to findings from a randomized placebo-controlled trial involving 148 patients.
Of those randomized to receive the selective serotonin reuptake inhibitor, 10% developed moderate to severe depression during the course of their cancer treatment, compared with 24.6% of those who received placebo, Dr. William J. Burke reported in a poster at the annual meeting of the American Association for Geriatric Psychiatry.
After adjustment for age, baseline smoking status, and stratification variables, including gender, cancer site, cancer stage, and primary treatment modality, those receiving active treatment remained significantly less likely to develop depression (hazard ratio, 0.37), said Dr. Burke of the University of Nebraska Medical Center, Omaha.
Of note, patients undergoing radiotherapy as the primary treatment modality were significantly more likely than those undergoing surgery as the primary treatment modality to develop depression (hazard ratio, 3.6).
Patients in the escitalopram group who did not become depressed during the course of the study reported a significantly better quality of life for 3 consecutive months after escitalopram cessation than those in either the treatment or placebo group who did develop depression, Dr. Burke noted.
Patients included in this double-blind study had head and neck cancers and were about to enter cancer treatment. Depression was assessed using the QIDS-SR(Quick Inventory of Depressive Symptomatology-Self Rated).
The findings have important implications for the management of patients entering treatment for head and neck cancer, which can be a particularly devastating disease. For example, larynx and tongue cancers comprise only 2% of all cancers, but patients with these cancers commit 19% of all cancer-related suicides, Dr. Burke noted.
Up to half of all patients with head and neck cancer develop major depressive disorder, which can adversely affect adherence to treatment, reduce quality of life, and result in significant morbidity and reduced survival.
"Preventing depression during the course of cancer treatment may, thus, be of great benefit," he said.
The project was supported by a grant from the National Institute of Mental Health. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIMH or the National Institutes of Health. Additional support was provided by a research support fund grant from the Nebraska Medical Center and the University of Nebraska Medical Center. Forest Pharmaceuticals provided the matching placebo and drugs.
AT THE AAGP ANNUAL MEETING
Major finding: During cancer treatment 10% of the treatment group developed depression, versus 24.6% of the placebo group.
Data source: A randomized, double-blind, placebo-controlled trial involving 148 patients.
Disclosures: The project was supported by a grant from the National Institute of Mental Health. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIMH or the National Institutes of Health. Additional support was provided by a research support fund grant from the Nebraska Medical Center and the University of Nebraska Medical Center. Forest Pharmaceuticals provided the matching placebo and drugs.
Executive function moderates emotion regulation in MDD
LOS ANGELES – Poor executive function ability in older patients with major depressive disorder was associated with reduced ability to implement emotion-regulation strategies in a prospective study involving 70 subjects.
The findings suggest that older patients with poor executive functioning might have a reduced capacity for benefiting from standard interventions for depression, such as cognitive-behavioral therapy, which rely on the implementation of emotion-regulation skills, Moria J. Smoski, Ph.D., reported in a poster at the annual meeting of the American Association for Geriatric Psychiatry.
A total of 70 adults aged 60-87 years – 30 with major depressive disorder (MDD) and 40 without MDD – participated in the study. All subjects completed an emotion-regulation task involving stress induction achieved by ruminating for 2 minutes about a personally relevant stressor, followed by guided emotion regulation by either reappraisal or distraction techniques. A control group received no instruction on emotion regulation, said Dr. Smoski of Duke University Medical Center, Durham, N.C.
Mood was assessed using a three-item scale administered before and after stress induction, and at 1-minute intervals throughout the emotion-regulation phase. This protocol was repeated three times.
A subset of participants underwent neuropsychological executive-function testing, including a verbal fluency test (FAS) and assessment of task-switching and cognitive flexibility using the Trail Making Test-B (Trails B), Dr. Smoski said in an interview.
Subjects with MDD who had mid-level Trails B scores (scores at the 34th to 66th percentile) or high-level scores (scores at the 67th to 100th percentile) – indicating adequate or high levels of executive functioning – experienced significantly different degrees of negative affect reduction based on the emotion-regulation strategy used. The greatest decrease in negative affect scores was seen in those instructed to use distraction, followed by those instructed to use reappraisal and those who received no instruction on emotion regulation.
Subjects with low-level Trails B scores (scores at the 0 to 33rd percentile) – indicating poor executive function ability – experienced no benefit with use of any of the regulation strategies, Dr. Smoski said.
No Trails B–dependent effects were seen among those without MDD.
"Participants with low executive function ability may have less ability to capitalize on conscious emotion regulation–strategy use to manage negative affect," she noted.
The findings indicate that executive function moderates emotion regulation in late-life depression and might have important implications for treatment in this population, Dr. Smoski said.
Since patients with late-life depression and poor executive function may have reduced response to standard interventions, referral for behavioral-activation therapy – a "promising and interesting treatment approach" – merits consideration, she said.
This study was supported by grants from the National Institutes of Health. Dr. Smoski reported having no other disclosures.
LOS ANGELES – Poor executive function ability in older patients with major depressive disorder was associated with reduced ability to implement emotion-regulation strategies in a prospective study involving 70 subjects.
The findings suggest that older patients with poor executive functioning might have a reduced capacity for benefiting from standard interventions for depression, such as cognitive-behavioral therapy, which rely on the implementation of emotion-regulation skills, Moria J. Smoski, Ph.D., reported in a poster at the annual meeting of the American Association for Geriatric Psychiatry.
A total of 70 adults aged 60-87 years – 30 with major depressive disorder (MDD) and 40 without MDD – participated in the study. All subjects completed an emotion-regulation task involving stress induction achieved by ruminating for 2 minutes about a personally relevant stressor, followed by guided emotion regulation by either reappraisal or distraction techniques. A control group received no instruction on emotion regulation, said Dr. Smoski of Duke University Medical Center, Durham, N.C.
Mood was assessed using a three-item scale administered before and after stress induction, and at 1-minute intervals throughout the emotion-regulation phase. This protocol was repeated three times.
A subset of participants underwent neuropsychological executive-function testing, including a verbal fluency test (FAS) and assessment of task-switching and cognitive flexibility using the Trail Making Test-B (Trails B), Dr. Smoski said in an interview.
Subjects with MDD who had mid-level Trails B scores (scores at the 34th to 66th percentile) or high-level scores (scores at the 67th to 100th percentile) – indicating adequate or high levels of executive functioning – experienced significantly different degrees of negative affect reduction based on the emotion-regulation strategy used. The greatest decrease in negative affect scores was seen in those instructed to use distraction, followed by those instructed to use reappraisal and those who received no instruction on emotion regulation.
Subjects with low-level Trails B scores (scores at the 0 to 33rd percentile) – indicating poor executive function ability – experienced no benefit with use of any of the regulation strategies, Dr. Smoski said.
No Trails B–dependent effects were seen among those without MDD.
"Participants with low executive function ability may have less ability to capitalize on conscious emotion regulation–strategy use to manage negative affect," she noted.
The findings indicate that executive function moderates emotion regulation in late-life depression and might have important implications for treatment in this population, Dr. Smoski said.
Since patients with late-life depression and poor executive function may have reduced response to standard interventions, referral for behavioral-activation therapy – a "promising and interesting treatment approach" – merits consideration, she said.
This study was supported by grants from the National Institutes of Health. Dr. Smoski reported having no other disclosures.
LOS ANGELES – Poor executive function ability in older patients with major depressive disorder was associated with reduced ability to implement emotion-regulation strategies in a prospective study involving 70 subjects.
The findings suggest that older patients with poor executive functioning might have a reduced capacity for benefiting from standard interventions for depression, such as cognitive-behavioral therapy, which rely on the implementation of emotion-regulation skills, Moria J. Smoski, Ph.D., reported in a poster at the annual meeting of the American Association for Geriatric Psychiatry.
A total of 70 adults aged 60-87 years – 30 with major depressive disorder (MDD) and 40 without MDD – participated in the study. All subjects completed an emotion-regulation task involving stress induction achieved by ruminating for 2 minutes about a personally relevant stressor, followed by guided emotion regulation by either reappraisal or distraction techniques. A control group received no instruction on emotion regulation, said Dr. Smoski of Duke University Medical Center, Durham, N.C.
Mood was assessed using a three-item scale administered before and after stress induction, and at 1-minute intervals throughout the emotion-regulation phase. This protocol was repeated three times.
A subset of participants underwent neuropsychological executive-function testing, including a verbal fluency test (FAS) and assessment of task-switching and cognitive flexibility using the Trail Making Test-B (Trails B), Dr. Smoski said in an interview.
Subjects with MDD who had mid-level Trails B scores (scores at the 34th to 66th percentile) or high-level scores (scores at the 67th to 100th percentile) – indicating adequate or high levels of executive functioning – experienced significantly different degrees of negative affect reduction based on the emotion-regulation strategy used. The greatest decrease in negative affect scores was seen in those instructed to use distraction, followed by those instructed to use reappraisal and those who received no instruction on emotion regulation.
Subjects with low-level Trails B scores (scores at the 0 to 33rd percentile) – indicating poor executive function ability – experienced no benefit with use of any of the regulation strategies, Dr. Smoski said.
No Trails B–dependent effects were seen among those without MDD.
"Participants with low executive function ability may have less ability to capitalize on conscious emotion regulation–strategy use to manage negative affect," she noted.
The findings indicate that executive function moderates emotion regulation in late-life depression and might have important implications for treatment in this population, Dr. Smoski said.
Since patients with late-life depression and poor executive function may have reduced response to standard interventions, referral for behavioral-activation therapy – a "promising and interesting treatment approach" – merits consideration, she said.
This study was supported by grants from the National Institutes of Health. Dr. Smoski reported having no other disclosures.
AT THE AAGP ANNUAL MEETING
Major finding: Lower Trails B scores were associated with lack of benefit from use of emotion-regulation strategies in depressed older adults.
Data source: A prospective study involving 70 adults aged 60-87 years.
Disclosures: This study was supported by grants from the National Institutes of Health. Dr. Smoski reported having no other disclosures.
NHANES follow-up characterizes asthma/allergy patient mortality
SAN ANTONIO – A diagnosis of asthma, allergic disease, or obstructive or restrictive lung disease among participants in the first National Health and Nutrition Examination Survey conferred a significantly increased long-term risk of all-cause mortality for adults who were aged 40-75 years at baseline but not for those who were aged 25-39 years at baseline, according to findings from the study.
A diagnosis of asthma in the younger group conferred an increased long-term risk of death due to respiratory causes – as did a diagnosis of asthma in the older group, Dr. Jessica R. Savage reported in a poster at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
"This association was not likely explained by underlying bronchitis or an increased risk of death due to respiratory infection but was likely due to asthma itself," said Dr. Savage of Brigham and Women’s Hospital, Boston.
"I think the main conclusions are reassuring – no increase in mortality if you are young and have allergies. Some studies show an association between allergy and stroke/heart disease. We were worried that with the rising increase in allergy, there would be also an increase in these other diseases. But we did not see that, fortunately.
"We saw an association with asthma and respiratory death even in the young. Of course, one always needs to remember to be vigilant with asthmatics, but overall for young people the news is good," she said during an interview.
Data were obtained from the National Health and Nutrition Examination Survey (NHANES I), which was conducted from 1971 to 1975 and included 31,937 adults. Of these participants, 14,407 were included in the NHANES I Epidemiologic Follow-Up Study (NHEFS) and were assessed for doctor-diagnosed asthma, allergic rhinitis, food allergy, and urticaria. A subcohort of 6,913 subjects received a more detailed health interview and examination, including prebronchodilator spirometry and percent predicted forced expiratory volume and forced vital capacity. Vital status and cause of death were obtained in 2006.
After adjustment for age, gender, income, education, race, and smoking history, a sensitivity analysis for the association between asthma and mortality demonstrated a significantly increased long-term risk of death in those who were aged 40-75 years at baseline (hazard ratio, 1.22), but not for those aged 25-29 years at baseline (HR, 1.20). The hazard ratios for all-cause mortality in these groups, after exclusion of subjects with bronchitis were not statistically significant (1.16 and 1.52, respectively).
Hazard ratios for the association between asthma and respiratory mortality were significant at 2.03 and 5.87 for the older and younger groups, respectively. The hazard ratios for these groups remained statistically significant at 8.56 and 1.82, respectively, after exclusion of subjects with bronchitis.
This study also demonstrated that older subjects with obstructive lung disease were at significantly increased risk of both all-cause and respiratory mortality and that older subjects with restrictive lung disease were at significantly increased risk of both all-cause and cardiovascular mortality.
Conversely, older adults with urticaria had a reduced risk of cardiovascular mortality.
Cancer-related mortality was slightly, but not significantly, increased in the younger subjects diagnosed with urticaria, and in the older subjects diagnosed with asthma or moderate to severe lung obstruction.
"Asthma and allergic diseases, which typically manifest in childhood, have increased in the United States over the last 3 decades. Asthma and allergy may increase mortality by directly reducing lung function or may be markers of immune dysregulation that could lead to systemic inflammation," Dr. Savage noted, adding that although prior studies have demonstrated associations between allergic sensitization and stroke, hives and cancer, asthma and mortality, and obstructive lung disease and cardiovascular events, the effects of asthma and allergic disease on long-term mortality have been unclear.
"The findings (of this follow-up study) provide some insight regarding the effects of asthma and allergic disease on long-term mortality, Dr. Savage said.
The NHEFS is a joint project of the National Center for Health Statistics and the National Institute on Aging in collaboration with other agencies of the U.S. Public Health Service. Dr. Savage reported having no disclosures.
SAN ANTONIO – A diagnosis of asthma, allergic disease, or obstructive or restrictive lung disease among participants in the first National Health and Nutrition Examination Survey conferred a significantly increased long-term risk of all-cause mortality for adults who were aged 40-75 years at baseline but not for those who were aged 25-39 years at baseline, according to findings from the study.
A diagnosis of asthma in the younger group conferred an increased long-term risk of death due to respiratory causes – as did a diagnosis of asthma in the older group, Dr. Jessica R. Savage reported in a poster at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
"This association was not likely explained by underlying bronchitis or an increased risk of death due to respiratory infection but was likely due to asthma itself," said Dr. Savage of Brigham and Women’s Hospital, Boston.
"I think the main conclusions are reassuring – no increase in mortality if you are young and have allergies. Some studies show an association between allergy and stroke/heart disease. We were worried that with the rising increase in allergy, there would be also an increase in these other diseases. But we did not see that, fortunately.
"We saw an association with asthma and respiratory death even in the young. Of course, one always needs to remember to be vigilant with asthmatics, but overall for young people the news is good," she said during an interview.
Data were obtained from the National Health and Nutrition Examination Survey (NHANES I), which was conducted from 1971 to 1975 and included 31,937 adults. Of these participants, 14,407 were included in the NHANES I Epidemiologic Follow-Up Study (NHEFS) and were assessed for doctor-diagnosed asthma, allergic rhinitis, food allergy, and urticaria. A subcohort of 6,913 subjects received a more detailed health interview and examination, including prebronchodilator spirometry and percent predicted forced expiratory volume and forced vital capacity. Vital status and cause of death were obtained in 2006.
After adjustment for age, gender, income, education, race, and smoking history, a sensitivity analysis for the association between asthma and mortality demonstrated a significantly increased long-term risk of death in those who were aged 40-75 years at baseline (hazard ratio, 1.22), but not for those aged 25-29 years at baseline (HR, 1.20). The hazard ratios for all-cause mortality in these groups, after exclusion of subjects with bronchitis were not statistically significant (1.16 and 1.52, respectively).
Hazard ratios for the association between asthma and respiratory mortality were significant at 2.03 and 5.87 for the older and younger groups, respectively. The hazard ratios for these groups remained statistically significant at 8.56 and 1.82, respectively, after exclusion of subjects with bronchitis.
This study also demonstrated that older subjects with obstructive lung disease were at significantly increased risk of both all-cause and respiratory mortality and that older subjects with restrictive lung disease were at significantly increased risk of both all-cause and cardiovascular mortality.
Conversely, older adults with urticaria had a reduced risk of cardiovascular mortality.
Cancer-related mortality was slightly, but not significantly, increased in the younger subjects diagnosed with urticaria, and in the older subjects diagnosed with asthma or moderate to severe lung obstruction.
"Asthma and allergic diseases, which typically manifest in childhood, have increased in the United States over the last 3 decades. Asthma and allergy may increase mortality by directly reducing lung function or may be markers of immune dysregulation that could lead to systemic inflammation," Dr. Savage noted, adding that although prior studies have demonstrated associations between allergic sensitization and stroke, hives and cancer, asthma and mortality, and obstructive lung disease and cardiovascular events, the effects of asthma and allergic disease on long-term mortality have been unclear.
"The findings (of this follow-up study) provide some insight regarding the effects of asthma and allergic disease on long-term mortality, Dr. Savage said.
The NHEFS is a joint project of the National Center for Health Statistics and the National Institute on Aging in collaboration with other agencies of the U.S. Public Health Service. Dr. Savage reported having no disclosures.
SAN ANTONIO – A diagnosis of asthma, allergic disease, or obstructive or restrictive lung disease among participants in the first National Health and Nutrition Examination Survey conferred a significantly increased long-term risk of all-cause mortality for adults who were aged 40-75 years at baseline but not for those who were aged 25-39 years at baseline, according to findings from the study.
A diagnosis of asthma in the younger group conferred an increased long-term risk of death due to respiratory causes – as did a diagnosis of asthma in the older group, Dr. Jessica R. Savage reported in a poster at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
"This association was not likely explained by underlying bronchitis or an increased risk of death due to respiratory infection but was likely due to asthma itself," said Dr. Savage of Brigham and Women’s Hospital, Boston.
"I think the main conclusions are reassuring – no increase in mortality if you are young and have allergies. Some studies show an association between allergy and stroke/heart disease. We were worried that with the rising increase in allergy, there would be also an increase in these other diseases. But we did not see that, fortunately.
"We saw an association with asthma and respiratory death even in the young. Of course, one always needs to remember to be vigilant with asthmatics, but overall for young people the news is good," she said during an interview.
Data were obtained from the National Health and Nutrition Examination Survey (NHANES I), which was conducted from 1971 to 1975 and included 31,937 adults. Of these participants, 14,407 were included in the NHANES I Epidemiologic Follow-Up Study (NHEFS) and were assessed for doctor-diagnosed asthma, allergic rhinitis, food allergy, and urticaria. A subcohort of 6,913 subjects received a more detailed health interview and examination, including prebronchodilator spirometry and percent predicted forced expiratory volume and forced vital capacity. Vital status and cause of death were obtained in 2006.
After adjustment for age, gender, income, education, race, and smoking history, a sensitivity analysis for the association between asthma and mortality demonstrated a significantly increased long-term risk of death in those who were aged 40-75 years at baseline (hazard ratio, 1.22), but not for those aged 25-29 years at baseline (HR, 1.20). The hazard ratios for all-cause mortality in these groups, after exclusion of subjects with bronchitis were not statistically significant (1.16 and 1.52, respectively).
Hazard ratios for the association between asthma and respiratory mortality were significant at 2.03 and 5.87 for the older and younger groups, respectively. The hazard ratios for these groups remained statistically significant at 8.56 and 1.82, respectively, after exclusion of subjects with bronchitis.
This study also demonstrated that older subjects with obstructive lung disease were at significantly increased risk of both all-cause and respiratory mortality and that older subjects with restrictive lung disease were at significantly increased risk of both all-cause and cardiovascular mortality.
Conversely, older adults with urticaria had a reduced risk of cardiovascular mortality.
Cancer-related mortality was slightly, but not significantly, increased in the younger subjects diagnosed with urticaria, and in the older subjects diagnosed with asthma or moderate to severe lung obstruction.
"Asthma and allergic diseases, which typically manifest in childhood, have increased in the United States over the last 3 decades. Asthma and allergy may increase mortality by directly reducing lung function or may be markers of immune dysregulation that could lead to systemic inflammation," Dr. Savage noted, adding that although prior studies have demonstrated associations between allergic sensitization and stroke, hives and cancer, asthma and mortality, and obstructive lung disease and cardiovascular events, the effects of asthma and allergic disease on long-term mortality have been unclear.
"The findings (of this follow-up study) provide some insight regarding the effects of asthma and allergic disease on long-term mortality, Dr. Savage said.
The NHEFS is a joint project of the National Center for Health Statistics and the National Institute on Aging in collaboration with other agencies of the U.S. Public Health Service. Dr. Savage reported having no disclosures.
AT THE AAAAI ANNUAL MEETING
Major finding: The long-term risk of death was significantly increased among NHANES 1 subjects with asthma who were aged 40-75 at baseline (hazard ratio, 1.22), but not among those aged 25-29 years at baseline (HR, 1.20).
Data source: Epidemiologic follow-up study (NHEFS) of the longitudinal NHANES 1 study.
Disclosures: The NHEFS is a joint project of the National Center for Health Statistics and the National Institute on Aging in collaboration with other agencies of the Public Health Service. Dr. Savage reported having no disclosures.
EBSOS implementation improves asthma guideline compliance
SAN ANTONIO – More of the children who present to the pediatric emergency department with asthma exacerbation received recommended care when the staff had instituted a nurse-initiated, evidence-based, standardized order set, according to Dr. Moira E. Breslin.
Specifically, the percentage of patients receiving at least one dose of ipratropium bromide improved from 55.4% before implementation of the order set to 90.9% after implementation. Compliance with the recommendation of the National Asthma Guidelines that patients receive three consecutive nebulized treatments of ipratropium bromide increased from 13.5% to 40.9%, Dr. Breslin of Duke University Medical Center, Durham, N.C., reported in a poster at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The median time to delivery of rescue medication also improved from 21 minutes to 14 minutes for first inhaled bronchodilator administration, and from 41 minutes to 19 minutes for delivery of systemic corticosteroids.
All differences were statistically significant.
The findings are based on a review of charts for 193 patients treated in the pediatric emergency department for status asthmaticus before implementation of the evidence-based standardized order set, or EBSOS, (between Feb. 23, 2009, and Feb. 22, 2012), and for 22 patients treated after implementation (between Feb. 23, 2012, and July 31, 2012).
The EBSOS for the treatment of pediatric asthma used in this study was developed and incorporated into the emergency department electronic ordering system because personnel were not consistently following national asthma treatment guidelines, according to a separate 2010 emergency department records review.
That review showed that 24% of patients admitted for status asthmaticus had not received the recommended ipratropium bromide treatment, and that only 14% of those who did receive ipratropium bromide received the recommended three consecutive doses.
Implementation of the EBSOS involved the use of an algorithm based on a validated Modified Pulmonary Index Score that allowed for triage nurse initiation of the EBSOS. The EBSOS called for continuous pulse oximetry, supplemental oxygen as needed, evaluation by a respiratory therapist, nebulized albuterol administration at 5 mg every 20 minutes for three treatments, administration of nebulized ipratropium bromide at 0.5 mg every 20 minutes for three treatments, and administration of one dose of oral prednisolone at 2 mg/kg up to a maximum of 60 mg.
"Implementation of an EBSOS improved compliance to national asthma guidelines, as evidenced by a higher proportion of pediatric emergency department patients in status asthmaticus receiving ipratropium bromide, as well as shortened time to delivery of inhaled bronchodilators and systemic steroids," Dr. Breslin concluded, noting that future analysis of this review will focus on patient-centered outcomes.
Dr. Breslin reported having no relevant financial disclosures
SAN ANTONIO – More of the children who present to the pediatric emergency department with asthma exacerbation received recommended care when the staff had instituted a nurse-initiated, evidence-based, standardized order set, according to Dr. Moira E. Breslin.
Specifically, the percentage of patients receiving at least one dose of ipratropium bromide improved from 55.4% before implementation of the order set to 90.9% after implementation. Compliance with the recommendation of the National Asthma Guidelines that patients receive three consecutive nebulized treatments of ipratropium bromide increased from 13.5% to 40.9%, Dr. Breslin of Duke University Medical Center, Durham, N.C., reported in a poster at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The median time to delivery of rescue medication also improved from 21 minutes to 14 minutes for first inhaled bronchodilator administration, and from 41 minutes to 19 minutes for delivery of systemic corticosteroids.
All differences were statistically significant.
The findings are based on a review of charts for 193 patients treated in the pediatric emergency department for status asthmaticus before implementation of the evidence-based standardized order set, or EBSOS, (between Feb. 23, 2009, and Feb. 22, 2012), and for 22 patients treated after implementation (between Feb. 23, 2012, and July 31, 2012).
The EBSOS for the treatment of pediatric asthma used in this study was developed and incorporated into the emergency department electronic ordering system because personnel were not consistently following national asthma treatment guidelines, according to a separate 2010 emergency department records review.
That review showed that 24% of patients admitted for status asthmaticus had not received the recommended ipratropium bromide treatment, and that only 14% of those who did receive ipratropium bromide received the recommended three consecutive doses.
Implementation of the EBSOS involved the use of an algorithm based on a validated Modified Pulmonary Index Score that allowed for triage nurse initiation of the EBSOS. The EBSOS called for continuous pulse oximetry, supplemental oxygen as needed, evaluation by a respiratory therapist, nebulized albuterol administration at 5 mg every 20 minutes for three treatments, administration of nebulized ipratropium bromide at 0.5 mg every 20 minutes for three treatments, and administration of one dose of oral prednisolone at 2 mg/kg up to a maximum of 60 mg.
"Implementation of an EBSOS improved compliance to national asthma guidelines, as evidenced by a higher proportion of pediatric emergency department patients in status asthmaticus receiving ipratropium bromide, as well as shortened time to delivery of inhaled bronchodilators and systemic steroids," Dr. Breslin concluded, noting that future analysis of this review will focus on patient-centered outcomes.
Dr. Breslin reported having no relevant financial disclosures
SAN ANTONIO – More of the children who present to the pediatric emergency department with asthma exacerbation received recommended care when the staff had instituted a nurse-initiated, evidence-based, standardized order set, according to Dr. Moira E. Breslin.
Specifically, the percentage of patients receiving at least one dose of ipratropium bromide improved from 55.4% before implementation of the order set to 90.9% after implementation. Compliance with the recommendation of the National Asthma Guidelines that patients receive three consecutive nebulized treatments of ipratropium bromide increased from 13.5% to 40.9%, Dr. Breslin of Duke University Medical Center, Durham, N.C., reported in a poster at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
The median time to delivery of rescue medication also improved from 21 minutes to 14 minutes for first inhaled bronchodilator administration, and from 41 minutes to 19 minutes for delivery of systemic corticosteroids.
All differences were statistically significant.
The findings are based on a review of charts for 193 patients treated in the pediatric emergency department for status asthmaticus before implementation of the evidence-based standardized order set, or EBSOS, (between Feb. 23, 2009, and Feb. 22, 2012), and for 22 patients treated after implementation (between Feb. 23, 2012, and July 31, 2012).
The EBSOS for the treatment of pediatric asthma used in this study was developed and incorporated into the emergency department electronic ordering system because personnel were not consistently following national asthma treatment guidelines, according to a separate 2010 emergency department records review.
That review showed that 24% of patients admitted for status asthmaticus had not received the recommended ipratropium bromide treatment, and that only 14% of those who did receive ipratropium bromide received the recommended three consecutive doses.
Implementation of the EBSOS involved the use of an algorithm based on a validated Modified Pulmonary Index Score that allowed for triage nurse initiation of the EBSOS. The EBSOS called for continuous pulse oximetry, supplemental oxygen as needed, evaluation by a respiratory therapist, nebulized albuterol administration at 5 mg every 20 minutes for three treatments, administration of nebulized ipratropium bromide at 0.5 mg every 20 minutes for three treatments, and administration of one dose of oral prednisolone at 2 mg/kg up to a maximum of 60 mg.
"Implementation of an EBSOS improved compliance to national asthma guidelines, as evidenced by a higher proportion of pediatric emergency department patients in status asthmaticus receiving ipratropium bromide, as well as shortened time to delivery of inhaled bronchodilators and systemic steroids," Dr. Breslin concluded, noting that future analysis of this review will focus on patient-centered outcomes.
Dr. Breslin reported having no relevant financial disclosures
AT THE AAAAI ANNUAL MEETING