User login
Legacy ICDs exposed to MRI still shock, pace as needed
Functions like sensing and pacing in implantable cardioverter defibrillators (ICDs) tend to resist interference from the energy fields generated by MRI, as long as device programming is properly adjusted before the scan.
That applies even to patients with older “legacy” devices implanted before the 2015 advent of MRI-conditional ICDs despite, in practice, prevalent but misguided resistance to obtaining MRI scans in such cases.
Less is known whether such non–MRI-conditional devices, once exposed to MRI, will then reliably deliver antiarrhythmic shocks or antitachycardia pacing (ATP) when needed.
A new cohort study has tried to fill in some of that knowledge gap. It showed no evidence of an excess risk for death or ICD failure to deliver therapy within about 2 years of clinically indicated MRI scans in 629 patients with non–MRI-conditional devices.
The findings, published online in the Annals of Internal Medicine, come with caveats. For example, they’re based on the experience of one, albeit major, center and on MRIs that were for varied indications using 1.5-tesla equipment only.
Despite such safety evidence for appropriately adjusted non–MRI-conditional ICDs, many patients with the devices don›t receive clinically indicated MRI scans due to “perceived risk” that the ICDs won’t then reliably deliver appropriate therapy, observe the authors, led by Joshua Ra, MD, University of California, San Francisco.
Any such risks are “largely theoretical,” but may still explain “why some institutions are shying away from offering MRI exams” to patients with non–MRI-conditional ICDs, Dr. Ra told this news organization.
Many such hospitals refer such patients to more experienced centers, creating “significant logistical barriers in terms of patient access to these MRIs,” he said. “That seems to still be prevalent, unfortunately.”
The current findings “provide another layer of reassurance” that MRI scans in patients with non–MRI-conditional ICDs don’t impair a device’s ability to deliver shocks or ATP, Dr. Ra said.
The cohort consisted of 629 patients with non–MRI-conditional ICDs who underwent 813 clinically indicated MRI exams from 2003 to early 2015 at Johns Hopkins University, Baltimore.
Scans performed within 4 weeks of device implantation were excluded because, the report notes, that’s when spontaneous lead dislodgements or changes to device parameters are most likely to occur. Also excluded were patients with permanent epicardial leads, abandoned leads, or subcutaneous ICD lead systems, the report states.
Still, Dr. Ra said, the cohort is fairly representative of “the modern patient population” of non–MRI-conditional ICD recipients.
A total of 4,177 arrhythmia episodes were documented during a median 2.2 years between scans and last device interrogation prior to pulse-generator change-out or lead exchange.
Of note, Dr. Ra observed, the arrhythmias were confirmed in only 85% of the cohort. Most of the remainder were referral patients who were lost to follow-up whose devices were unavailable for interrogation.
Device therapy terminated “nearly all” documented spontaneous arrhythmias in that 85% of patients, the report states. They included 757 episodes of ventricular tachycardia (VT) or ventricular fibrillation (VF), including 130 that were shocked and the remainder that were managed with ATP. There were also 105 supraventricular tachycardias, all successfully terminated with shocks.
There were no cases of VT or VF detection delay from undersensing or instances of syncope because of “abnormalities” in device detection of arrhythmias, the report states.
Of the 210 known deaths, which occurred a median 1.7 years after the scan, about half were noncardiac and more than a third were cardiac but nonarrhythmic.
Ten patients died from arrhythmia-related cardiac causes, representing 5% of deaths; but 7% of deaths were of undetermined cause.
“No direct relationship of deaths attributable to prior MRI exposure was found or reported,” the report states.
The researchers informally compared outcomes between older and more recently implanted non–MRI-conditional ICDs, the latter presumably with more modern design features. Their data, based on device interrogations, Dr. Ra said, “seem to suggest there were no differences.”
The study was supported by Johns Hopkins University and the National Institutes of Health. Author disclosures are available at apconline.org.
A version of this article first appeared on Medscape.com.
Functions like sensing and pacing in implantable cardioverter defibrillators (ICDs) tend to resist interference from the energy fields generated by MRI, as long as device programming is properly adjusted before the scan.
That applies even to patients with older “legacy” devices implanted before the 2015 advent of MRI-conditional ICDs despite, in practice, prevalent but misguided resistance to obtaining MRI scans in such cases.
Less is known whether such non–MRI-conditional devices, once exposed to MRI, will then reliably deliver antiarrhythmic shocks or antitachycardia pacing (ATP) when needed.
A new cohort study has tried to fill in some of that knowledge gap. It showed no evidence of an excess risk for death or ICD failure to deliver therapy within about 2 years of clinically indicated MRI scans in 629 patients with non–MRI-conditional devices.
The findings, published online in the Annals of Internal Medicine, come with caveats. For example, they’re based on the experience of one, albeit major, center and on MRIs that were for varied indications using 1.5-tesla equipment only.
Despite such safety evidence for appropriately adjusted non–MRI-conditional ICDs, many patients with the devices don›t receive clinically indicated MRI scans due to “perceived risk” that the ICDs won’t then reliably deliver appropriate therapy, observe the authors, led by Joshua Ra, MD, University of California, San Francisco.
Any such risks are “largely theoretical,” but may still explain “why some institutions are shying away from offering MRI exams” to patients with non–MRI-conditional ICDs, Dr. Ra told this news organization.
Many such hospitals refer such patients to more experienced centers, creating “significant logistical barriers in terms of patient access to these MRIs,” he said. “That seems to still be prevalent, unfortunately.”
The current findings “provide another layer of reassurance” that MRI scans in patients with non–MRI-conditional ICDs don’t impair a device’s ability to deliver shocks or ATP, Dr. Ra said.
The cohort consisted of 629 patients with non–MRI-conditional ICDs who underwent 813 clinically indicated MRI exams from 2003 to early 2015 at Johns Hopkins University, Baltimore.
Scans performed within 4 weeks of device implantation were excluded because, the report notes, that’s when spontaneous lead dislodgements or changes to device parameters are most likely to occur. Also excluded were patients with permanent epicardial leads, abandoned leads, or subcutaneous ICD lead systems, the report states.
Still, Dr. Ra said, the cohort is fairly representative of “the modern patient population” of non–MRI-conditional ICD recipients.
A total of 4,177 arrhythmia episodes were documented during a median 2.2 years between scans and last device interrogation prior to pulse-generator change-out or lead exchange.
Of note, Dr. Ra observed, the arrhythmias were confirmed in only 85% of the cohort. Most of the remainder were referral patients who were lost to follow-up whose devices were unavailable for interrogation.
Device therapy terminated “nearly all” documented spontaneous arrhythmias in that 85% of patients, the report states. They included 757 episodes of ventricular tachycardia (VT) or ventricular fibrillation (VF), including 130 that were shocked and the remainder that were managed with ATP. There were also 105 supraventricular tachycardias, all successfully terminated with shocks.
There were no cases of VT or VF detection delay from undersensing or instances of syncope because of “abnormalities” in device detection of arrhythmias, the report states.
Of the 210 known deaths, which occurred a median 1.7 years after the scan, about half were noncardiac and more than a third were cardiac but nonarrhythmic.
Ten patients died from arrhythmia-related cardiac causes, representing 5% of deaths; but 7% of deaths were of undetermined cause.
“No direct relationship of deaths attributable to prior MRI exposure was found or reported,” the report states.
The researchers informally compared outcomes between older and more recently implanted non–MRI-conditional ICDs, the latter presumably with more modern design features. Their data, based on device interrogations, Dr. Ra said, “seem to suggest there were no differences.”
The study was supported by Johns Hopkins University and the National Institutes of Health. Author disclosures are available at apconline.org.
A version of this article first appeared on Medscape.com.
Functions like sensing and pacing in implantable cardioverter defibrillators (ICDs) tend to resist interference from the energy fields generated by MRI, as long as device programming is properly adjusted before the scan.
That applies even to patients with older “legacy” devices implanted before the 2015 advent of MRI-conditional ICDs despite, in practice, prevalent but misguided resistance to obtaining MRI scans in such cases.
Less is known whether such non–MRI-conditional devices, once exposed to MRI, will then reliably deliver antiarrhythmic shocks or antitachycardia pacing (ATP) when needed.
A new cohort study has tried to fill in some of that knowledge gap. It showed no evidence of an excess risk for death or ICD failure to deliver therapy within about 2 years of clinically indicated MRI scans in 629 patients with non–MRI-conditional devices.
The findings, published online in the Annals of Internal Medicine, come with caveats. For example, they’re based on the experience of one, albeit major, center and on MRIs that were for varied indications using 1.5-tesla equipment only.
Despite such safety evidence for appropriately adjusted non–MRI-conditional ICDs, many patients with the devices don›t receive clinically indicated MRI scans due to “perceived risk” that the ICDs won’t then reliably deliver appropriate therapy, observe the authors, led by Joshua Ra, MD, University of California, San Francisco.
Any such risks are “largely theoretical,” but may still explain “why some institutions are shying away from offering MRI exams” to patients with non–MRI-conditional ICDs, Dr. Ra told this news organization.
Many such hospitals refer such patients to more experienced centers, creating “significant logistical barriers in terms of patient access to these MRIs,” he said. “That seems to still be prevalent, unfortunately.”
The current findings “provide another layer of reassurance” that MRI scans in patients with non–MRI-conditional ICDs don’t impair a device’s ability to deliver shocks or ATP, Dr. Ra said.
The cohort consisted of 629 patients with non–MRI-conditional ICDs who underwent 813 clinically indicated MRI exams from 2003 to early 2015 at Johns Hopkins University, Baltimore.
Scans performed within 4 weeks of device implantation were excluded because, the report notes, that’s when spontaneous lead dislodgements or changes to device parameters are most likely to occur. Also excluded were patients with permanent epicardial leads, abandoned leads, or subcutaneous ICD lead systems, the report states.
Still, Dr. Ra said, the cohort is fairly representative of “the modern patient population” of non–MRI-conditional ICD recipients.
A total of 4,177 arrhythmia episodes were documented during a median 2.2 years between scans and last device interrogation prior to pulse-generator change-out or lead exchange.
Of note, Dr. Ra observed, the arrhythmias were confirmed in only 85% of the cohort. Most of the remainder were referral patients who were lost to follow-up whose devices were unavailable for interrogation.
Device therapy terminated “nearly all” documented spontaneous arrhythmias in that 85% of patients, the report states. They included 757 episodes of ventricular tachycardia (VT) or ventricular fibrillation (VF), including 130 that were shocked and the remainder that were managed with ATP. There were also 105 supraventricular tachycardias, all successfully terminated with shocks.
There were no cases of VT or VF detection delay from undersensing or instances of syncope because of “abnormalities” in device detection of arrhythmias, the report states.
Of the 210 known deaths, which occurred a median 1.7 years after the scan, about half were noncardiac and more than a third were cardiac but nonarrhythmic.
Ten patients died from arrhythmia-related cardiac causes, representing 5% of deaths; but 7% of deaths were of undetermined cause.
“No direct relationship of deaths attributable to prior MRI exposure was found or reported,” the report states.
The researchers informally compared outcomes between older and more recently implanted non–MRI-conditional ICDs, the latter presumably with more modern design features. Their data, based on device interrogations, Dr. Ra said, “seem to suggest there were no differences.”
The study was supported by Johns Hopkins University and the National Institutes of Health. Author disclosures are available at apconline.org.
A version of this article first appeared on Medscape.com.
The new blood pressure target in primary care
This transcript has been edited for clarity.
I’m Dr. Neil Skolnik. There are very few things that we treat more often than hypertension, so you’d think the guidelines would have been clear a long time ago. Less than 10 years ago, in 2014, JNC 8 (Eighth Joint National Committee) recommended target blood pressure for individuals under 60 to be less than 140/90, and for those older than 60, less than 150/90.
Then, based primarily on the SPRINT trial (which included only people with or at significantly elevated risk for atherosclerotic cardiovascular disease), in 2017 the American Heart Association’s hypertension guidelines lowered the target BP to less than 130/80 for most individuals. It’s a little more nuanced than that, but most of us don’t remember the nuance. I’ve written about my reservations with that statement in the AHA’s journal, Circulation.
Now the American Academy of Family Physicians has updated its recommendations, and they recommend a BP less than 140/90. This is not a small change, as it often takes additional medication to achieve lower BP targets, and additional medicines lead to additional adverse effects. I’m going share with you some details from the new guideline, and then I’m going share my opinion about it.
The AAFP guideline applies to adults with hypertension, with or without cardiovascular disease. In the comprehensive literature review, the trials ran for an average of 3.7 years, and about 75% of the patients in the trials did not have preexisting cardiovascular disease.
The key to their recommendations is that target BPs lower than 140/90 did not show a statistically significant decrease in total mortality. In regard to serious adverse events, though, lower targets led to a nominal increase that didn’t reach statistical significance. Serious adverse events were defined as death or events that required hospitalization or resulted in significant disability. In regard to all other adverse events, including syncope and hypotension, there was a significant increase, with a relative risk of 1.44 (a 44% increase in adverse events). This reflected an absolute risk increase of 3%, compared with the standard target group (specifically 9.8% vs. 6.8%), with a number needed to harm of 33 over 3.7 years.
Another potential harm of low BP targets was the need for an average of one additional medicine to reach lower BP targets. One systematic review cited an eightfold higher withdrawal rate because of adverse events in the lower-target BP groups.
The AAFP guidelines said that, in the comprehensive review of the literature, while there was no difference in mortality or stroke with lower BP targets, a small additional benefit was observed in myocardial infarction – a 16% lower incidence, with a number needed to treat of 137 over 3.7 years.
So that’s the background. Let me now go over the specifics of the AAFP recommendations.
AAFP gives a strong recommendation for a standard BP target of less than 140/90. They go on to say – and grade this next statement as a weak recommendation – that, while treating to a lower BP target does not provide additional mortality benefit, a target BP of less than 135/85 can be considered to lower the risk for MI, noting that lower BP may increase harms. They state that the lower BP target could be considered based on patient preferences and values.
The AAFP guideline is incredibly helpful. The difference in the recommendations of two large societies – American Heart Association and AAFP — stems from two things. I believe that AHA focused on the composite endpoints in trials such as SPRINT, which included only high-risk patients, and the AAFP uses mortality as the driving endpoint in a broader group of patients that included both high- and lower-risk patients.
In addition, it appears that the two organizations weigh adverse events differently in coming to their conclusions. Clearly, we see more adverse events when aiming for a lower BP level, and in my experience, patients care a lot about adverse events.
Interestingly, the International Society of Hypertension recommends an “essential” BP target of less than 140/90 for most individuals, and for those under 65, they provide the option of an “optimal” BP of less than 130/80. Remember that for certain comorbidities there are also other guidelines out there. The American Diabetes Association this year revised its target BP to less than 130/80 for people with diabetes; for prevention of recurrent stroke, guidelines from the AHA/American Stroke Association in 2021 recommend BP less than 130/80, and the International Society for Hypertension as well as the AHA recommends a BP of less than 130/80 for those with established atherosclerotic cardiovascular disease.
To repeat, though, the main topic for today is that as a general target, the AAFP guidelines recommend a BP less than 140/90.
Dr. Skolnik is professor, department of family medicine, Sidney Kimmel Medical College, Philadelphia, and associate director, department of family medicine, Abington (Pa.) Jefferson Health. He disclosed conflicts of interest with AstraZeneca, Teva, Eli Lilly, Boehringer Ingelheim, Sanofi, Sanofi Pasteur, GlaxoSmithKline, Merck, and Bayer.
A version of this article first appeared on Medscape.com.
*This article was updated on 2/7/2023.
This transcript has been edited for clarity.
I’m Dr. Neil Skolnik. There are very few things that we treat more often than hypertension, so you’d think the guidelines would have been clear a long time ago. Less than 10 years ago, in 2014, JNC 8 (Eighth Joint National Committee) recommended target blood pressure for individuals under 60 to be less than 140/90, and for those older than 60, less than 150/90.
Then, based primarily on the SPRINT trial (which included only people with or at significantly elevated risk for atherosclerotic cardiovascular disease), in 2017 the American Heart Association’s hypertension guidelines lowered the target BP to less than 130/80 for most individuals. It’s a little more nuanced than that, but most of us don’t remember the nuance. I’ve written about my reservations with that statement in the AHA’s journal, Circulation.
Now the American Academy of Family Physicians has updated its recommendations, and they recommend a BP less than 140/90. This is not a small change, as it often takes additional medication to achieve lower BP targets, and additional medicines lead to additional adverse effects. I’m going share with you some details from the new guideline, and then I’m going share my opinion about it.
The AAFP guideline applies to adults with hypertension, with or without cardiovascular disease. In the comprehensive literature review, the trials ran for an average of 3.7 years, and about 75% of the patients in the trials did not have preexisting cardiovascular disease.
The key to their recommendations is that target BPs lower than 140/90 did not show a statistically significant decrease in total mortality. In regard to serious adverse events, though, lower targets led to a nominal increase that didn’t reach statistical significance. Serious adverse events were defined as death or events that required hospitalization or resulted in significant disability. In regard to all other adverse events, including syncope and hypotension, there was a significant increase, with a relative risk of 1.44 (a 44% increase in adverse events). This reflected an absolute risk increase of 3%, compared with the standard target group (specifically 9.8% vs. 6.8%), with a number needed to harm of 33 over 3.7 years.
Another potential harm of low BP targets was the need for an average of one additional medicine to reach lower BP targets. One systematic review cited an eightfold higher withdrawal rate because of adverse events in the lower-target BP groups.
The AAFP guidelines said that, in the comprehensive review of the literature, while there was no difference in mortality or stroke with lower BP targets, a small additional benefit was observed in myocardial infarction – a 16% lower incidence, with a number needed to treat of 137 over 3.7 years.
So that’s the background. Let me now go over the specifics of the AAFP recommendations.
AAFP gives a strong recommendation for a standard BP target of less than 140/90. They go on to say – and grade this next statement as a weak recommendation – that, while treating to a lower BP target does not provide additional mortality benefit, a target BP of less than 135/85 can be considered to lower the risk for MI, noting that lower BP may increase harms. They state that the lower BP target could be considered based on patient preferences and values.
The AAFP guideline is incredibly helpful. The difference in the recommendations of two large societies – American Heart Association and AAFP — stems from two things. I believe that AHA focused on the composite endpoints in trials such as SPRINT, which included only high-risk patients, and the AAFP uses mortality as the driving endpoint in a broader group of patients that included both high- and lower-risk patients.
In addition, it appears that the two organizations weigh adverse events differently in coming to their conclusions. Clearly, we see more adverse events when aiming for a lower BP level, and in my experience, patients care a lot about adverse events.
Interestingly, the International Society of Hypertension recommends an “essential” BP target of less than 140/90 for most individuals, and for those under 65, they provide the option of an “optimal” BP of less than 130/80. Remember that for certain comorbidities there are also other guidelines out there. The American Diabetes Association this year revised its target BP to less than 130/80 for people with diabetes; for prevention of recurrent stroke, guidelines from the AHA/American Stroke Association in 2021 recommend BP less than 130/80, and the International Society for Hypertension as well as the AHA recommends a BP of less than 130/80 for those with established atherosclerotic cardiovascular disease.
To repeat, though, the main topic for today is that as a general target, the AAFP guidelines recommend a BP less than 140/90.
Dr. Skolnik is professor, department of family medicine, Sidney Kimmel Medical College, Philadelphia, and associate director, department of family medicine, Abington (Pa.) Jefferson Health. He disclosed conflicts of interest with AstraZeneca, Teva, Eli Lilly, Boehringer Ingelheim, Sanofi, Sanofi Pasteur, GlaxoSmithKline, Merck, and Bayer.
A version of this article first appeared on Medscape.com.
*This article was updated on 2/7/2023.
This transcript has been edited for clarity.
I’m Dr. Neil Skolnik. There are very few things that we treat more often than hypertension, so you’d think the guidelines would have been clear a long time ago. Less than 10 years ago, in 2014, JNC 8 (Eighth Joint National Committee) recommended target blood pressure for individuals under 60 to be less than 140/90, and for those older than 60, less than 150/90.
Then, based primarily on the SPRINT trial (which included only people with or at significantly elevated risk for atherosclerotic cardiovascular disease), in 2017 the American Heart Association’s hypertension guidelines lowered the target BP to less than 130/80 for most individuals. It’s a little more nuanced than that, but most of us don’t remember the nuance. I’ve written about my reservations with that statement in the AHA’s journal, Circulation.
Now the American Academy of Family Physicians has updated its recommendations, and they recommend a BP less than 140/90. This is not a small change, as it often takes additional medication to achieve lower BP targets, and additional medicines lead to additional adverse effects. I’m going share with you some details from the new guideline, and then I’m going share my opinion about it.
The AAFP guideline applies to adults with hypertension, with or without cardiovascular disease. In the comprehensive literature review, the trials ran for an average of 3.7 years, and about 75% of the patients in the trials did not have preexisting cardiovascular disease.
The key to their recommendations is that target BPs lower than 140/90 did not show a statistically significant decrease in total mortality. In regard to serious adverse events, though, lower targets led to a nominal increase that didn’t reach statistical significance. Serious adverse events were defined as death or events that required hospitalization or resulted in significant disability. In regard to all other adverse events, including syncope and hypotension, there was a significant increase, with a relative risk of 1.44 (a 44% increase in adverse events). This reflected an absolute risk increase of 3%, compared with the standard target group (specifically 9.8% vs. 6.8%), with a number needed to harm of 33 over 3.7 years.
Another potential harm of low BP targets was the need for an average of one additional medicine to reach lower BP targets. One systematic review cited an eightfold higher withdrawal rate because of adverse events in the lower-target BP groups.
The AAFP guidelines said that, in the comprehensive review of the literature, while there was no difference in mortality or stroke with lower BP targets, a small additional benefit was observed in myocardial infarction – a 16% lower incidence, with a number needed to treat of 137 over 3.7 years.
So that’s the background. Let me now go over the specifics of the AAFP recommendations.
AAFP gives a strong recommendation for a standard BP target of less than 140/90. They go on to say – and grade this next statement as a weak recommendation – that, while treating to a lower BP target does not provide additional mortality benefit, a target BP of less than 135/85 can be considered to lower the risk for MI, noting that lower BP may increase harms. They state that the lower BP target could be considered based on patient preferences and values.
The AAFP guideline is incredibly helpful. The difference in the recommendations of two large societies – American Heart Association and AAFP — stems from two things. I believe that AHA focused on the composite endpoints in trials such as SPRINT, which included only high-risk patients, and the AAFP uses mortality as the driving endpoint in a broader group of patients that included both high- and lower-risk patients.
In addition, it appears that the two organizations weigh adverse events differently in coming to their conclusions. Clearly, we see more adverse events when aiming for a lower BP level, and in my experience, patients care a lot about adverse events.
Interestingly, the International Society of Hypertension recommends an “essential” BP target of less than 140/90 for most individuals, and for those under 65, they provide the option of an “optimal” BP of less than 130/80. Remember that for certain comorbidities there are also other guidelines out there. The American Diabetes Association this year revised its target BP to less than 130/80 for people with diabetes; for prevention of recurrent stroke, guidelines from the AHA/American Stroke Association in 2021 recommend BP less than 130/80, and the International Society for Hypertension as well as the AHA recommends a BP of less than 130/80 for those with established atherosclerotic cardiovascular disease.
To repeat, though, the main topic for today is that as a general target, the AAFP guidelines recommend a BP less than 140/90.
Dr. Skolnik is professor, department of family medicine, Sidney Kimmel Medical College, Philadelphia, and associate director, department of family medicine, Abington (Pa.) Jefferson Health. He disclosed conflicts of interest with AstraZeneca, Teva, Eli Lilly, Boehringer Ingelheim, Sanofi, Sanofi Pasteur, GlaxoSmithKline, Merck, and Bayer.
A version of this article first appeared on Medscape.com.
*This article was updated on 2/7/2023.
Universal testing for Lp(a): What are we waiting for?
It soon became clear that Lp(a) was associated with atherosclerotic cardiovascular disease (ASCVD), but whether an elevated blood level was a biomarker or a causal factor proved difficult to determine. Studies of inheritance patterns confirmed that blood levels were primarily genetically determined and largely resistant to lifestyle and pharmacologic intervention. It seemed senseless to test for something that was deemed “unmodifiable,” so untreatable. That label stuck for decades.
Fortunately, a resurgent interest in molecular pathophysiology this past decade has clarified Lp(a)’s unique contribution to atherothrombotic disease and calcific aortic stenosis. While there remains much to be learned about this complex, highly atherogenic molecule and its role in cardiac disease, it seems shortsighted not to take the simple step of identifying who carries this risk. Why are we not testing everyone for an extremely common and potent risk factor for the most lethal disease on the planet?
Epidemiologic studies project a stunning number of people in the United States to be at increased risk for Lp(a)-mediated coronary and cerebrovascular events. Because the LPA gene which codes for the apo(a) component of the Lp(a) molecule is fully expressed at age 2, this is a truly lifelong risk factor for a projected 64 million individuals with blood levels (> 60 mg/dL) high enough to double their risk for ASCVD. Because risk increases linearly, this includes 16 million, like me, with levels > 116 mg/dL, who are at four times the risk for ASCVD as those with normal levels (< 30 mg/dL).
Because Lp(a) level remains relatively constant throughout life, a single blood test would help stratify the risk it confers on millions of people who, under current U.S. guidelines, would never be tested. Until Lp(a) is integrated into its algorithms, the commonly used ASCVD Risk Calculator will substantially underestimate risk in 20% of the population.
A potential barrier to universal testing is that the ideal method to measure Lp(a) has yet to be determined. Lp(a) comprises an apoB particle bonded to an apo(a) particle. Apo(a) is complex and has a number of isoforms that can result in large heterogeneity in apo(a) size between, as well as within, individuals. This contributes to controversy about the ideal assay and whether Lp(a) levels should be expressed as mass (mg/dL) or number of particles (nmols/L). This should not, however, deter universal testing.
One-time cost, lifetime benefit?
Absent universal testing, it’s impossible to estimate the economic toll that Lp(a) exacts, but it’s surely an extraordinary number, particularly because the highest-risk individuals are prone to recurrent, nonfatal vascular events. The substantial price tag for my personal decade of Lp(a)-induced vascular havoc included four percutaneous coronary interventions with rapid stent restenosis, an eventual bypass surgery, and an aborted left hemispheric stroke, requiring an urgent carotid endarterectomy.
As a frame of reference, U.S. expenditures related to ASCVD are estimated to be $351 billion annually. If everyone in the United States over the age of 18 were tested for Lp(a) at a cost of $100 per person, this would be a $21 billion expenditure. This nonrecurring expense would identify the 20% – or almost 42 million individuals – at high risk for ASCVD, a number of whom would have already had vascular events. This one-time cost would be a foundational step in securing year-after-year savings from enhanced ASCVD prevention and reduction in recurrent vascular events.
Such savings would be significantly enhanced if and when targeted, effective Lp(a) treatments become available, but it seems shortsighted to make this the linchpin for universal testing. It’s noteworthy that Canadian and European guidelines already endorse one-time testing for all.
The confirmation of Lp(a)’s causal role in ASCVD remains underappreciated by medical providers across all specialties. Much of the elegant Lp(a)-related science of the past decade has yet to translate to the clinical world. What better way to rectify this than by identifying those with high Lp(a)? Since the advent of the statin era, “good” and “bad” cholesterol values are common conversational fare, in part because virtually every adult has had not one, but many lipid panels. Universal Lp(a) testing would spotlight this pervasive and important risk factor that was referred to as the “horrible” cholesterol in a recent review.
U.S. guidelines need updating
To foster this, U.S. guidelines, which influence every aspect of care, including testing, prevention, treatment, reimbursement, and medical legal issues, need to be simplified. The discussion of Lp(a) testing in the 2018 U.S. guidelines on cholesterol management is already obsolete. The contingencies on when testing is “reasonable” or “may be reasonable” are dated and cumbersome. In contrast, a recommendation to test everyone once, perhaps in adolescence, would be a useful, forward-looking strategy.
To date, trials of an antisense oligonucleotide and a small interfering RNA molecule targeting hepatic LPA messenger RNA have confirmed that plasma Lp(a) levels can be significantly and safely lowered. If the ongoing Lp(a) HORIZON and OCEAN(a) phase 3 trials have positive outcomes in patients with known ASCVD, this would spawn a host of clinical trials to explore the possibilities of these therapies in primary prevention as well. These will require tens of thousands of enrollees, and universal testing would expand the pool of potential participants.
The majority of at-risk individuals identified through universal testing would be candidates for primary prevention. This large, currently unidentified cohort should have all coexisting risk factors assessed and managed; lowering elevated LDL cholesterol early and aggressively is paramount. Recent data from the United Kingdom suggest that attainment of specific LDL cholesterol levels may offset the risk for vascular events in those with high Lp(a) levels.
Of note, this was the advice given to the small fraction of high-risk individuals like me, who had their Lp(a) level tested long before its ominous implications were understood. This recommendation was informed mostly by common sense. For any number of reasons, the same might be said for universal testing.
Dr. Leahy, a retired cardiologist in San Diego, has an abiding professional and personal interest in Lp(a), which has been responsible for a number of cardiovascular events in his own life over the past 2 decades. He was a participant in the phase 2 clinical trial of the Lp(a)-lowering antisense oligonucleotide being studied in the Lp(a) HORIZON trial, funded by Novartis, and is currently undergoing apheresis treatment. A version of this article originally appeared on Medscape.com.
It soon became clear that Lp(a) was associated with atherosclerotic cardiovascular disease (ASCVD), but whether an elevated blood level was a biomarker or a causal factor proved difficult to determine. Studies of inheritance patterns confirmed that blood levels were primarily genetically determined and largely resistant to lifestyle and pharmacologic intervention. It seemed senseless to test for something that was deemed “unmodifiable,” so untreatable. That label stuck for decades.
Fortunately, a resurgent interest in molecular pathophysiology this past decade has clarified Lp(a)’s unique contribution to atherothrombotic disease and calcific aortic stenosis. While there remains much to be learned about this complex, highly atherogenic molecule and its role in cardiac disease, it seems shortsighted not to take the simple step of identifying who carries this risk. Why are we not testing everyone for an extremely common and potent risk factor for the most lethal disease on the planet?
Epidemiologic studies project a stunning number of people in the United States to be at increased risk for Lp(a)-mediated coronary and cerebrovascular events. Because the LPA gene which codes for the apo(a) component of the Lp(a) molecule is fully expressed at age 2, this is a truly lifelong risk factor for a projected 64 million individuals with blood levels (> 60 mg/dL) high enough to double their risk for ASCVD. Because risk increases linearly, this includes 16 million, like me, with levels > 116 mg/dL, who are at four times the risk for ASCVD as those with normal levels (< 30 mg/dL).
Because Lp(a) level remains relatively constant throughout life, a single blood test would help stratify the risk it confers on millions of people who, under current U.S. guidelines, would never be tested. Until Lp(a) is integrated into its algorithms, the commonly used ASCVD Risk Calculator will substantially underestimate risk in 20% of the population.
A potential barrier to universal testing is that the ideal method to measure Lp(a) has yet to be determined. Lp(a) comprises an apoB particle bonded to an apo(a) particle. Apo(a) is complex and has a number of isoforms that can result in large heterogeneity in apo(a) size between, as well as within, individuals. This contributes to controversy about the ideal assay and whether Lp(a) levels should be expressed as mass (mg/dL) or number of particles (nmols/L). This should not, however, deter universal testing.
One-time cost, lifetime benefit?
Absent universal testing, it’s impossible to estimate the economic toll that Lp(a) exacts, but it’s surely an extraordinary number, particularly because the highest-risk individuals are prone to recurrent, nonfatal vascular events. The substantial price tag for my personal decade of Lp(a)-induced vascular havoc included four percutaneous coronary interventions with rapid stent restenosis, an eventual bypass surgery, and an aborted left hemispheric stroke, requiring an urgent carotid endarterectomy.
As a frame of reference, U.S. expenditures related to ASCVD are estimated to be $351 billion annually. If everyone in the United States over the age of 18 were tested for Lp(a) at a cost of $100 per person, this would be a $21 billion expenditure. This nonrecurring expense would identify the 20% – or almost 42 million individuals – at high risk for ASCVD, a number of whom would have already had vascular events. This one-time cost would be a foundational step in securing year-after-year savings from enhanced ASCVD prevention and reduction in recurrent vascular events.
Such savings would be significantly enhanced if and when targeted, effective Lp(a) treatments become available, but it seems shortsighted to make this the linchpin for universal testing. It’s noteworthy that Canadian and European guidelines already endorse one-time testing for all.
The confirmation of Lp(a)’s causal role in ASCVD remains underappreciated by medical providers across all specialties. Much of the elegant Lp(a)-related science of the past decade has yet to translate to the clinical world. What better way to rectify this than by identifying those with high Lp(a)? Since the advent of the statin era, “good” and “bad” cholesterol values are common conversational fare, in part because virtually every adult has had not one, but many lipid panels. Universal Lp(a) testing would spotlight this pervasive and important risk factor that was referred to as the “horrible” cholesterol in a recent review.
U.S. guidelines need updating
To foster this, U.S. guidelines, which influence every aspect of care, including testing, prevention, treatment, reimbursement, and medical legal issues, need to be simplified. The discussion of Lp(a) testing in the 2018 U.S. guidelines on cholesterol management is already obsolete. The contingencies on when testing is “reasonable” or “may be reasonable” are dated and cumbersome. In contrast, a recommendation to test everyone once, perhaps in adolescence, would be a useful, forward-looking strategy.
To date, trials of an antisense oligonucleotide and a small interfering RNA molecule targeting hepatic LPA messenger RNA have confirmed that plasma Lp(a) levels can be significantly and safely lowered. If the ongoing Lp(a) HORIZON and OCEAN(a) phase 3 trials have positive outcomes in patients with known ASCVD, this would spawn a host of clinical trials to explore the possibilities of these therapies in primary prevention as well. These will require tens of thousands of enrollees, and universal testing would expand the pool of potential participants.
The majority of at-risk individuals identified through universal testing would be candidates for primary prevention. This large, currently unidentified cohort should have all coexisting risk factors assessed and managed; lowering elevated LDL cholesterol early and aggressively is paramount. Recent data from the United Kingdom suggest that attainment of specific LDL cholesterol levels may offset the risk for vascular events in those with high Lp(a) levels.
Of note, this was the advice given to the small fraction of high-risk individuals like me, who had their Lp(a) level tested long before its ominous implications were understood. This recommendation was informed mostly by common sense. For any number of reasons, the same might be said for universal testing.
Dr. Leahy, a retired cardiologist in San Diego, has an abiding professional and personal interest in Lp(a), which has been responsible for a number of cardiovascular events in his own life over the past 2 decades. He was a participant in the phase 2 clinical trial of the Lp(a)-lowering antisense oligonucleotide being studied in the Lp(a) HORIZON trial, funded by Novartis, and is currently undergoing apheresis treatment. A version of this article originally appeared on Medscape.com.
It soon became clear that Lp(a) was associated with atherosclerotic cardiovascular disease (ASCVD), but whether an elevated blood level was a biomarker or a causal factor proved difficult to determine. Studies of inheritance patterns confirmed that blood levels were primarily genetically determined and largely resistant to lifestyle and pharmacologic intervention. It seemed senseless to test for something that was deemed “unmodifiable,” so untreatable. That label stuck for decades.
Fortunately, a resurgent interest in molecular pathophysiology this past decade has clarified Lp(a)’s unique contribution to atherothrombotic disease and calcific aortic stenosis. While there remains much to be learned about this complex, highly atherogenic molecule and its role in cardiac disease, it seems shortsighted not to take the simple step of identifying who carries this risk. Why are we not testing everyone for an extremely common and potent risk factor for the most lethal disease on the planet?
Epidemiologic studies project a stunning number of people in the United States to be at increased risk for Lp(a)-mediated coronary and cerebrovascular events. Because the LPA gene which codes for the apo(a) component of the Lp(a) molecule is fully expressed at age 2, this is a truly lifelong risk factor for a projected 64 million individuals with blood levels (> 60 mg/dL) high enough to double their risk for ASCVD. Because risk increases linearly, this includes 16 million, like me, with levels > 116 mg/dL, who are at four times the risk for ASCVD as those with normal levels (< 30 mg/dL).
Because Lp(a) level remains relatively constant throughout life, a single blood test would help stratify the risk it confers on millions of people who, under current U.S. guidelines, would never be tested. Until Lp(a) is integrated into its algorithms, the commonly used ASCVD Risk Calculator will substantially underestimate risk in 20% of the population.
A potential barrier to universal testing is that the ideal method to measure Lp(a) has yet to be determined. Lp(a) comprises an apoB particle bonded to an apo(a) particle. Apo(a) is complex and has a number of isoforms that can result in large heterogeneity in apo(a) size between, as well as within, individuals. This contributes to controversy about the ideal assay and whether Lp(a) levels should be expressed as mass (mg/dL) or number of particles (nmols/L). This should not, however, deter universal testing.
One-time cost, lifetime benefit?
Absent universal testing, it’s impossible to estimate the economic toll that Lp(a) exacts, but it’s surely an extraordinary number, particularly because the highest-risk individuals are prone to recurrent, nonfatal vascular events. The substantial price tag for my personal decade of Lp(a)-induced vascular havoc included four percutaneous coronary interventions with rapid stent restenosis, an eventual bypass surgery, and an aborted left hemispheric stroke, requiring an urgent carotid endarterectomy.
As a frame of reference, U.S. expenditures related to ASCVD are estimated to be $351 billion annually. If everyone in the United States over the age of 18 were tested for Lp(a) at a cost of $100 per person, this would be a $21 billion expenditure. This nonrecurring expense would identify the 20% – or almost 42 million individuals – at high risk for ASCVD, a number of whom would have already had vascular events. This one-time cost would be a foundational step in securing year-after-year savings from enhanced ASCVD prevention and reduction in recurrent vascular events.
Such savings would be significantly enhanced if and when targeted, effective Lp(a) treatments become available, but it seems shortsighted to make this the linchpin for universal testing. It’s noteworthy that Canadian and European guidelines already endorse one-time testing for all.
The confirmation of Lp(a)’s causal role in ASCVD remains underappreciated by medical providers across all specialties. Much of the elegant Lp(a)-related science of the past decade has yet to translate to the clinical world. What better way to rectify this than by identifying those with high Lp(a)? Since the advent of the statin era, “good” and “bad” cholesterol values are common conversational fare, in part because virtually every adult has had not one, but many lipid panels. Universal Lp(a) testing would spotlight this pervasive and important risk factor that was referred to as the “horrible” cholesterol in a recent review.
U.S. guidelines need updating
To foster this, U.S. guidelines, which influence every aspect of care, including testing, prevention, treatment, reimbursement, and medical legal issues, need to be simplified. The discussion of Lp(a) testing in the 2018 U.S. guidelines on cholesterol management is already obsolete. The contingencies on when testing is “reasonable” or “may be reasonable” are dated and cumbersome. In contrast, a recommendation to test everyone once, perhaps in adolescence, would be a useful, forward-looking strategy.
To date, trials of an antisense oligonucleotide and a small interfering RNA molecule targeting hepatic LPA messenger RNA have confirmed that plasma Lp(a) levels can be significantly and safely lowered. If the ongoing Lp(a) HORIZON and OCEAN(a) phase 3 trials have positive outcomes in patients with known ASCVD, this would spawn a host of clinical trials to explore the possibilities of these therapies in primary prevention as well. These will require tens of thousands of enrollees, and universal testing would expand the pool of potential participants.
The majority of at-risk individuals identified through universal testing would be candidates for primary prevention. This large, currently unidentified cohort should have all coexisting risk factors assessed and managed; lowering elevated LDL cholesterol early and aggressively is paramount. Recent data from the United Kingdom suggest that attainment of specific LDL cholesterol levels may offset the risk for vascular events in those with high Lp(a) levels.
Of note, this was the advice given to the small fraction of high-risk individuals like me, who had their Lp(a) level tested long before its ominous implications were understood. This recommendation was informed mostly by common sense. For any number of reasons, the same might be said for universal testing.
Dr. Leahy, a retired cardiologist in San Diego, has an abiding professional and personal interest in Lp(a), which has been responsible for a number of cardiovascular events in his own life over the past 2 decades. He was a participant in the phase 2 clinical trial of the Lp(a)-lowering antisense oligonucleotide being studied in the Lp(a) HORIZON trial, funded by Novartis, and is currently undergoing apheresis treatment. A version of this article originally appeared on Medscape.com.
CV deaths jumped in 2020, reflecting pandemic toll
Cardiovascular-related deaths increased dramatically in 2020, marking the largest single-year increase since 2015 and surpassing the previous record from 2003, according to the American Heart Association’s 2023 Statistical Update.
During the first year of the COVID-19 pandemic, the largest increases in cardiovascular disease (CVD) deaths were seen among Asian, Black, and Hispanic people.
“We thought we had been improving as a country with respect to CVD deaths over the past few decades,” Connie Tsao, MD, chair of the AHA Statistical Update writing committee, told this news organization.
Since 2020, however, those trends have changed. Dr. Tsao, a staff cardiologist at Beth Israel Deaconess Medical Center and assistant professor of medicine at Harvard Medical School, both in Boston, noted the firsthand experience that many clinicians had in seeing the shift.
“We observed this sharp rise in age-adjusted CVD deaths, which corresponds to the COVID-19 pandemic,” she said. “Those of us health care providers knew from the overfull hospitals and ICUs that clearly COVID took a toll, particularly in those with cardiovascular risk factors.”
The AHA Statistical Update was published online in the journal Circulation.
Data on deaths
Each year, the American Heart Association and National Institutes of Health report the latest statistics related to heart disease, stroke, and cardiovascular risk factors. The 2023 update includes additional information about pandemic-related data.
Overall, the number of people who died from cardiovascular disease increased during the first year of the pandemic, rising from 876,613 in 2019 to 928,741 in 2020. This topped the previous high of 910,000 in 2003.
In addition, the age-adjusted mortality rate increased for the first time in several years, Dr. Tsao said, by a “fairly substantial” 4.6%. The age-adjusted mortality rate incorporates the variability in the aging population from year to year, accounting for higher death rates among older people.
“Even though our total number of deaths has been slowly increasing over the past decade, we have seen a decline each year in our age-adjusted rates – until 2020,” she said. “I think that is very indicative of what has been going on within our country – and the world – in light of people of all ages being impacted by the COVID-19 pandemic, especially before vaccines were available to slow the spread.”
The largest increases in CVD-related deaths occurred among Asian, Black, and Hispanic people, who were most heavily affected during the first year of the pandemic.
“People from communities of color were among those most highly impacted, especially early on, often due to a disproportionate burden of cardiovascular risk factors, such as hypertension and obesity,” Michelle Albert, MD, MPH, president of AHA and a professor of medicine at the University of California, San Francisco, said in a statement.
Dr. Albert, who is also the director of UCSF’s Center for the Study of Adversity and Cardiovascular Disease, does research on health equity and noted the disparities seen in the 2020 numbers. “Additionally, there are socioeconomic considerations, as well as the ongoing impact of structural racism on multiple factors, including limiting the ability to access quality health care,” she said.
Additional considerations
In a special commentary, the Statistical Update writing committee pointed to the need to track data for other underrepresented communities, including LGBTQ people and those living in rural or urban areas. The authors outlined several ways to better understand the effects of identity and social determinants of health, as well as strategies to reduce cardiovascular-related disparities.
“This year’s writing group made a concerted effort to gather information on specific social factors related to health risk and outcomes, including sexual orientation, gender identity, urbanization, and socioeconomic position,” Dr. Tsao said. “However, the data are lacking because these communities are grossly underrepresented in clinical and epidemiological research.”
For the next several years, the AHA Statistical Update will likely include more insights about the effects of the COVID-19 pandemic, as well as ongoing disparities.
“For sure, we will be continuing to see the effects of the pandemic for years to come,” Dr. Tsao said. “Recognition of the disparities in outcomes among vulnerable groups should be a call to action among health care providers and researchers, administration, and policy leaders to investigate the reasons and make changes to reverse these trends.”
The statistical update was prepared by a volunteer writing group on behalf of the American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee.
A version of this article first appeared on Medscape.com.
Cardiovascular-related deaths increased dramatically in 2020, marking the largest single-year increase since 2015 and surpassing the previous record from 2003, according to the American Heart Association’s 2023 Statistical Update.
During the first year of the COVID-19 pandemic, the largest increases in cardiovascular disease (CVD) deaths were seen among Asian, Black, and Hispanic people.
“We thought we had been improving as a country with respect to CVD deaths over the past few decades,” Connie Tsao, MD, chair of the AHA Statistical Update writing committee, told this news organization.
Since 2020, however, those trends have changed. Dr. Tsao, a staff cardiologist at Beth Israel Deaconess Medical Center and assistant professor of medicine at Harvard Medical School, both in Boston, noted the firsthand experience that many clinicians had in seeing the shift.
“We observed this sharp rise in age-adjusted CVD deaths, which corresponds to the COVID-19 pandemic,” she said. “Those of us health care providers knew from the overfull hospitals and ICUs that clearly COVID took a toll, particularly in those with cardiovascular risk factors.”
The AHA Statistical Update was published online in the journal Circulation.
Data on deaths
Each year, the American Heart Association and National Institutes of Health report the latest statistics related to heart disease, stroke, and cardiovascular risk factors. The 2023 update includes additional information about pandemic-related data.
Overall, the number of people who died from cardiovascular disease increased during the first year of the pandemic, rising from 876,613 in 2019 to 928,741 in 2020. This topped the previous high of 910,000 in 2003.
In addition, the age-adjusted mortality rate increased for the first time in several years, Dr. Tsao said, by a “fairly substantial” 4.6%. The age-adjusted mortality rate incorporates the variability in the aging population from year to year, accounting for higher death rates among older people.
“Even though our total number of deaths has been slowly increasing over the past decade, we have seen a decline each year in our age-adjusted rates – until 2020,” she said. “I think that is very indicative of what has been going on within our country – and the world – in light of people of all ages being impacted by the COVID-19 pandemic, especially before vaccines were available to slow the spread.”
The largest increases in CVD-related deaths occurred among Asian, Black, and Hispanic people, who were most heavily affected during the first year of the pandemic.
“People from communities of color were among those most highly impacted, especially early on, often due to a disproportionate burden of cardiovascular risk factors, such as hypertension and obesity,” Michelle Albert, MD, MPH, president of AHA and a professor of medicine at the University of California, San Francisco, said in a statement.
Dr. Albert, who is also the director of UCSF’s Center for the Study of Adversity and Cardiovascular Disease, does research on health equity and noted the disparities seen in the 2020 numbers. “Additionally, there are socioeconomic considerations, as well as the ongoing impact of structural racism on multiple factors, including limiting the ability to access quality health care,” she said.
Additional considerations
In a special commentary, the Statistical Update writing committee pointed to the need to track data for other underrepresented communities, including LGBTQ people and those living in rural or urban areas. The authors outlined several ways to better understand the effects of identity and social determinants of health, as well as strategies to reduce cardiovascular-related disparities.
“This year’s writing group made a concerted effort to gather information on specific social factors related to health risk and outcomes, including sexual orientation, gender identity, urbanization, and socioeconomic position,” Dr. Tsao said. “However, the data are lacking because these communities are grossly underrepresented in clinical and epidemiological research.”
For the next several years, the AHA Statistical Update will likely include more insights about the effects of the COVID-19 pandemic, as well as ongoing disparities.
“For sure, we will be continuing to see the effects of the pandemic for years to come,” Dr. Tsao said. “Recognition of the disparities in outcomes among vulnerable groups should be a call to action among health care providers and researchers, administration, and policy leaders to investigate the reasons and make changes to reverse these trends.”
The statistical update was prepared by a volunteer writing group on behalf of the American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee.
A version of this article first appeared on Medscape.com.
Cardiovascular-related deaths increased dramatically in 2020, marking the largest single-year increase since 2015 and surpassing the previous record from 2003, according to the American Heart Association’s 2023 Statistical Update.
During the first year of the COVID-19 pandemic, the largest increases in cardiovascular disease (CVD) deaths were seen among Asian, Black, and Hispanic people.
“We thought we had been improving as a country with respect to CVD deaths over the past few decades,” Connie Tsao, MD, chair of the AHA Statistical Update writing committee, told this news organization.
Since 2020, however, those trends have changed. Dr. Tsao, a staff cardiologist at Beth Israel Deaconess Medical Center and assistant professor of medicine at Harvard Medical School, both in Boston, noted the firsthand experience that many clinicians had in seeing the shift.
“We observed this sharp rise in age-adjusted CVD deaths, which corresponds to the COVID-19 pandemic,” she said. “Those of us health care providers knew from the overfull hospitals and ICUs that clearly COVID took a toll, particularly in those with cardiovascular risk factors.”
The AHA Statistical Update was published online in the journal Circulation.
Data on deaths
Each year, the American Heart Association and National Institutes of Health report the latest statistics related to heart disease, stroke, and cardiovascular risk factors. The 2023 update includes additional information about pandemic-related data.
Overall, the number of people who died from cardiovascular disease increased during the first year of the pandemic, rising from 876,613 in 2019 to 928,741 in 2020. This topped the previous high of 910,000 in 2003.
In addition, the age-adjusted mortality rate increased for the first time in several years, Dr. Tsao said, by a “fairly substantial” 4.6%. The age-adjusted mortality rate incorporates the variability in the aging population from year to year, accounting for higher death rates among older people.
“Even though our total number of deaths has been slowly increasing over the past decade, we have seen a decline each year in our age-adjusted rates – until 2020,” she said. “I think that is very indicative of what has been going on within our country – and the world – in light of people of all ages being impacted by the COVID-19 pandemic, especially before vaccines were available to slow the spread.”
The largest increases in CVD-related deaths occurred among Asian, Black, and Hispanic people, who were most heavily affected during the first year of the pandemic.
“People from communities of color were among those most highly impacted, especially early on, often due to a disproportionate burden of cardiovascular risk factors, such as hypertension and obesity,” Michelle Albert, MD, MPH, president of AHA and a professor of medicine at the University of California, San Francisco, said in a statement.
Dr. Albert, who is also the director of UCSF’s Center for the Study of Adversity and Cardiovascular Disease, does research on health equity and noted the disparities seen in the 2020 numbers. “Additionally, there are socioeconomic considerations, as well as the ongoing impact of structural racism on multiple factors, including limiting the ability to access quality health care,” she said.
Additional considerations
In a special commentary, the Statistical Update writing committee pointed to the need to track data for other underrepresented communities, including LGBTQ people and those living in rural or urban areas. The authors outlined several ways to better understand the effects of identity and social determinants of health, as well as strategies to reduce cardiovascular-related disparities.
“This year’s writing group made a concerted effort to gather information on specific social factors related to health risk and outcomes, including sexual orientation, gender identity, urbanization, and socioeconomic position,” Dr. Tsao said. “However, the data are lacking because these communities are grossly underrepresented in clinical and epidemiological research.”
For the next several years, the AHA Statistical Update will likely include more insights about the effects of the COVID-19 pandemic, as well as ongoing disparities.
“For sure, we will be continuing to see the effects of the pandemic for years to come,” Dr. Tsao said. “Recognition of the disparities in outcomes among vulnerable groups should be a call to action among health care providers and researchers, administration, and policy leaders to investigate the reasons and make changes to reverse these trends.”
The statistical update was prepared by a volunteer writing group on behalf of the American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee.
A version of this article first appeared on Medscape.com.
FROM CIRCULATION
The long-range thrombolysis forecast calls for tiny ultrasonic tornadoes
Sticks and stones may break my bones, but clots will never hurt me
You’ve probably seen “Ghostbusters” or at least heard the theme song. Maybe you even know about the Discovery Channel’s “Mythbusters.” But now there’s a new buster in town, and it eats platitudes for breakfast: Meet Cliche-busters, LOTME’s new recurring feature.
This week, Cliche-busters takes on “Two wrongs don’t make a right.” Yum.
We start with blood clots, which are bad. Doctors go to a lot of trouble to get rid of the things because they are dangerous. A blood clot, then, is a bodily function gone wrong.
Tornadoes are also bad. Out there in the world, these violently rotating columns of air can destroy buildings, toss large objects long distances, and inspire mediocre action movies. They are examples of nature gone wrong.
Seemingly, these two wrongs – blood clots and tornadoes – are not about to make a right. Has Cliche-busters bitten off more than it can chew?
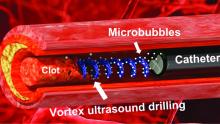
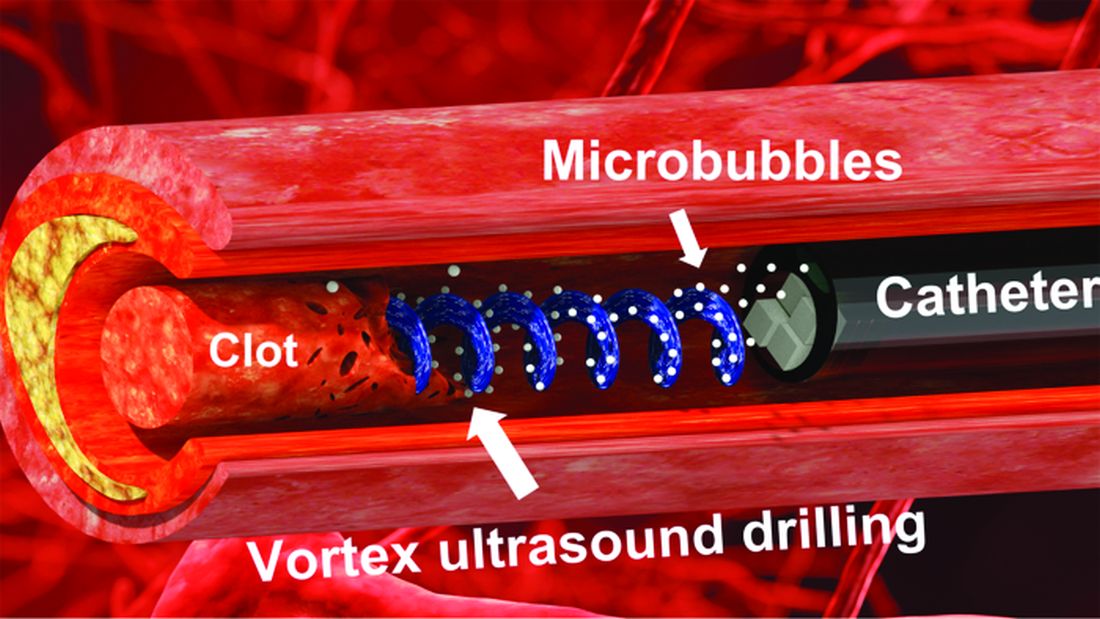
Not according to Xiaoning Jiang of North Carolina State University, Raleigh, and his team of researchers. They’ve figured out a way to use a tiny ultrasonic tornado to break down clots in the brain. “Our new work uses vortex ultrasound, where the ultrasound waves have a helical wavefront. In other words, the ultrasound is swirling as it moves forward,” he said in a statement from the university.
Their new tool’s single transducer is small enough to fit in a catheter, and its “vortex ultrasound-induced shear force has the potential to break down clots safely and improve the efficacy of thrombolysis,” they explained in the open-access journal Research.
The investigators used cow blood in a 3D-printed model of the cerebral venous sinus for the proof-of-concept study and were able to dissolve an acute blood clot in less than 30 minutes, compared with the 15-30 hours needed with a pharmaceutical intervention, according to the written statement.
Can you hear the sound of two wrongs making a right? We can, and that closes the curtain on this cliche.
With age does not come wisdom
We’ve all met this person before. The sort of person who takes a 10-minute IQ test on a shifty-looking website and then proceeds to brag about a 180 IQ until the heat death of the universe. The one who worships at the altar of Mensa. Yeah, that guy. They’re never as smart as they think they are, but they’ll never, ever admit it.
It’s not exactly a secret that IQ as a measurement of intelligence is highly overrated. A lot of scientists doubt we should bother measuring it at all. That said, a higher IQ is associated with greater success in academic and financial endeavors, so it’s not absolutely worthless. And if we’re stuck with it, we may as well study it.
That brings us neatly to new research published in Brain and Behavior. Most studies into IQ and self-estimated intelligence have focused on younger adults, and the author of this study was curious if the stereotype of young men inflating their IQ, a stereotype backed up by research, persisted into older adulthood. So she conducted a survey of 159 younger adults and 152 older adults to find out.
The results in younger adults were not surprising: Younger men overestimated their actual IQ by 5-15 points, which tracks with previous research. We’re in for a bit of a surprise with the older adults, though, because the older men were more humble about their intelligence, with their estimation falling in line with their actual IQ. Older women, however, not so much. In fact, they overestimated their intelligence just as much as the younger men.
In addition, older women who perceived themselves as more attractive reported the highest self-estimated intelligence of all. That isn’t how intelligence works, but honestly, if Grandma’s out and about thinking she looks good and has the brains to go and win “Jeopardy!” do you really have the heart to tell her otherwise?
Fight temptation with empathy … and shoes
Relationships are tough. They all go through their respective ups and downs, but what happens when one person is feeling so down in the partnership that cheating comes to mind? Is there any way to stop it from happening?
Well, a recent study suggests that there is, and it’s as simple as putting yourself in the other person’s shoes. By observing 408 heterosexual, monogamous participants in a series of experiments, psychologists in Israel and New York found that practicing empathy and “perspective taking” doesn’t necessarily stop people from cheating but it does reduces the desire.
People cheat on their significant others for many different reasons – men for a lack of sexual needs being met and women for shortfalls regarding emotional needs – but prioritizing the other person’s perspective gives the idea of being unfaithful a different view and could make one act differently, the investigators said.
Perspective taking also promotes other positive attributes to the relationship, such as the promotion of compassion and the feeling of being understood, lead author Gurit Birnbaum of Reichman University in Herzliya, Israel, said in a written statement. These things ultimately help couples navigate the rough patches and strengthen bonds, making them even less likely to cheat.
The researchers noted that even people in satisfying relationships do cheat, but this approach does encourage people to stop and think before they act. It could ultimately prevent what might be a huge mistake.
Think before they act. Hmm, that’s kind of like look before they leap, right? Sounds like a job for the Cliche-busters.
Sticks and stones may break my bones, but clots will never hurt me
You’ve probably seen “Ghostbusters” or at least heard the theme song. Maybe you even know about the Discovery Channel’s “Mythbusters.” But now there’s a new buster in town, and it eats platitudes for breakfast: Meet Cliche-busters, LOTME’s new recurring feature.
This week, Cliche-busters takes on “Two wrongs don’t make a right.” Yum.
We start with blood clots, which are bad. Doctors go to a lot of trouble to get rid of the things because they are dangerous. A blood clot, then, is a bodily function gone wrong.
Tornadoes are also bad. Out there in the world, these violently rotating columns of air can destroy buildings, toss large objects long distances, and inspire mediocre action movies. They are examples of nature gone wrong.
Seemingly, these two wrongs – blood clots and tornadoes – are not about to make a right. Has Cliche-busters bitten off more than it can chew?
Not according to Xiaoning Jiang of North Carolina State University, Raleigh, and his team of researchers. They’ve figured out a way to use a tiny ultrasonic tornado to break down clots in the brain. “Our new work uses vortex ultrasound, where the ultrasound waves have a helical wavefront. In other words, the ultrasound is swirling as it moves forward,” he said in a statement from the university.
Their new tool’s single transducer is small enough to fit in a catheter, and its “vortex ultrasound-induced shear force has the potential to break down clots safely and improve the efficacy of thrombolysis,” they explained in the open-access journal Research.
The investigators used cow blood in a 3D-printed model of the cerebral venous sinus for the proof-of-concept study and were able to dissolve an acute blood clot in less than 30 minutes, compared with the 15-30 hours needed with a pharmaceutical intervention, according to the written statement.
Can you hear the sound of two wrongs making a right? We can, and that closes the curtain on this cliche.
With age does not come wisdom
We’ve all met this person before. The sort of person who takes a 10-minute IQ test on a shifty-looking website and then proceeds to brag about a 180 IQ until the heat death of the universe. The one who worships at the altar of Mensa. Yeah, that guy. They’re never as smart as they think they are, but they’ll never, ever admit it.
It’s not exactly a secret that IQ as a measurement of intelligence is highly overrated. A lot of scientists doubt we should bother measuring it at all. That said, a higher IQ is associated with greater success in academic and financial endeavors, so it’s not absolutely worthless. And if we’re stuck with it, we may as well study it.
That brings us neatly to new research published in Brain and Behavior. Most studies into IQ and self-estimated intelligence have focused on younger adults, and the author of this study was curious if the stereotype of young men inflating their IQ, a stereotype backed up by research, persisted into older adulthood. So she conducted a survey of 159 younger adults and 152 older adults to find out.
The results in younger adults were not surprising: Younger men overestimated their actual IQ by 5-15 points, which tracks with previous research. We’re in for a bit of a surprise with the older adults, though, because the older men were more humble about their intelligence, with their estimation falling in line with their actual IQ. Older women, however, not so much. In fact, they overestimated their intelligence just as much as the younger men.
In addition, older women who perceived themselves as more attractive reported the highest self-estimated intelligence of all. That isn’t how intelligence works, but honestly, if Grandma’s out and about thinking she looks good and has the brains to go and win “Jeopardy!” do you really have the heart to tell her otherwise?
Fight temptation with empathy … and shoes
Relationships are tough. They all go through their respective ups and downs, but what happens when one person is feeling so down in the partnership that cheating comes to mind? Is there any way to stop it from happening?
Well, a recent study suggests that there is, and it’s as simple as putting yourself in the other person’s shoes. By observing 408 heterosexual, monogamous participants in a series of experiments, psychologists in Israel and New York found that practicing empathy and “perspective taking” doesn’t necessarily stop people from cheating but it does reduces the desire.
People cheat on their significant others for many different reasons – men for a lack of sexual needs being met and women for shortfalls regarding emotional needs – but prioritizing the other person’s perspective gives the idea of being unfaithful a different view and could make one act differently, the investigators said.
Perspective taking also promotes other positive attributes to the relationship, such as the promotion of compassion and the feeling of being understood, lead author Gurit Birnbaum of Reichman University in Herzliya, Israel, said in a written statement. These things ultimately help couples navigate the rough patches and strengthen bonds, making them even less likely to cheat.
The researchers noted that even people in satisfying relationships do cheat, but this approach does encourage people to stop and think before they act. It could ultimately prevent what might be a huge mistake.
Think before they act. Hmm, that’s kind of like look before they leap, right? Sounds like a job for the Cliche-busters.
Sticks and stones may break my bones, but clots will never hurt me
You’ve probably seen “Ghostbusters” or at least heard the theme song. Maybe you even know about the Discovery Channel’s “Mythbusters.” But now there’s a new buster in town, and it eats platitudes for breakfast: Meet Cliche-busters, LOTME’s new recurring feature.
This week, Cliche-busters takes on “Two wrongs don’t make a right.” Yum.
We start with blood clots, which are bad. Doctors go to a lot of trouble to get rid of the things because they are dangerous. A blood clot, then, is a bodily function gone wrong.
Tornadoes are also bad. Out there in the world, these violently rotating columns of air can destroy buildings, toss large objects long distances, and inspire mediocre action movies. They are examples of nature gone wrong.
Seemingly, these two wrongs – blood clots and tornadoes – are not about to make a right. Has Cliche-busters bitten off more than it can chew?
Not according to Xiaoning Jiang of North Carolina State University, Raleigh, and his team of researchers. They’ve figured out a way to use a tiny ultrasonic tornado to break down clots in the brain. “Our new work uses vortex ultrasound, where the ultrasound waves have a helical wavefront. In other words, the ultrasound is swirling as it moves forward,” he said in a statement from the university.
Their new tool’s single transducer is small enough to fit in a catheter, and its “vortex ultrasound-induced shear force has the potential to break down clots safely and improve the efficacy of thrombolysis,” they explained in the open-access journal Research.
The investigators used cow blood in a 3D-printed model of the cerebral venous sinus for the proof-of-concept study and were able to dissolve an acute blood clot in less than 30 minutes, compared with the 15-30 hours needed with a pharmaceutical intervention, according to the written statement.
Can you hear the sound of two wrongs making a right? We can, and that closes the curtain on this cliche.
With age does not come wisdom
We’ve all met this person before. The sort of person who takes a 10-minute IQ test on a shifty-looking website and then proceeds to brag about a 180 IQ until the heat death of the universe. The one who worships at the altar of Mensa. Yeah, that guy. They’re never as smart as they think they are, but they’ll never, ever admit it.
It’s not exactly a secret that IQ as a measurement of intelligence is highly overrated. A lot of scientists doubt we should bother measuring it at all. That said, a higher IQ is associated with greater success in academic and financial endeavors, so it’s not absolutely worthless. And if we’re stuck with it, we may as well study it.
That brings us neatly to new research published in Brain and Behavior. Most studies into IQ and self-estimated intelligence have focused on younger adults, and the author of this study was curious if the stereotype of young men inflating their IQ, a stereotype backed up by research, persisted into older adulthood. So she conducted a survey of 159 younger adults and 152 older adults to find out.
The results in younger adults were not surprising: Younger men overestimated their actual IQ by 5-15 points, which tracks with previous research. We’re in for a bit of a surprise with the older adults, though, because the older men were more humble about their intelligence, with their estimation falling in line with their actual IQ. Older women, however, not so much. In fact, they overestimated their intelligence just as much as the younger men.
In addition, older women who perceived themselves as more attractive reported the highest self-estimated intelligence of all. That isn’t how intelligence works, but honestly, if Grandma’s out and about thinking she looks good and has the brains to go and win “Jeopardy!” do you really have the heart to tell her otherwise?
Fight temptation with empathy … and shoes
Relationships are tough. They all go through their respective ups and downs, but what happens when one person is feeling so down in the partnership that cheating comes to mind? Is there any way to stop it from happening?
Well, a recent study suggests that there is, and it’s as simple as putting yourself in the other person’s shoes. By observing 408 heterosexual, monogamous participants in a series of experiments, psychologists in Israel and New York found that practicing empathy and “perspective taking” doesn’t necessarily stop people from cheating but it does reduces the desire.
People cheat on their significant others for many different reasons – men for a lack of sexual needs being met and women for shortfalls regarding emotional needs – but prioritizing the other person’s perspective gives the idea of being unfaithful a different view and could make one act differently, the investigators said.
Perspective taking also promotes other positive attributes to the relationship, such as the promotion of compassion and the feeling of being understood, lead author Gurit Birnbaum of Reichman University in Herzliya, Israel, said in a written statement. These things ultimately help couples navigate the rough patches and strengthen bonds, making them even less likely to cheat.
The researchers noted that even people in satisfying relationships do cheat, but this approach does encourage people to stop and think before they act. It could ultimately prevent what might be a huge mistake.
Think before they act. Hmm, that’s kind of like look before they leap, right? Sounds like a job for the Cliche-busters.
Autism linked to problems with cardiovascular health
People with autism are more likely to face diabetes, high cholesterol, and heart disease than those without the neurologic condition, according to a study published in JAMA Pediatrics. Researchers also found that children with autism are especially likely to develop diabetes compared with their peers, and are at greater risk of hypertension, too.
While the link between autism and risk for obesity and gastrointestinal ailments is well-established, the new findings suggest that clinicians who care for these patients – particularly children – should focus on cardiometabolic health more broadly.
“Clinicians who are treating kids with autism need to pay more attention to this,” said Chanaka N. Kahathuduwa, MD, PhD, MPhil, of the department of neurology at Texas Tech University Health Sciences Center, in Lubbock, and a coauthor of the new study.
A pediatrician may prescribe an atypical antipsychotic medication such as risperidone to regulate the behavior of an autistic child, Dr. Kahathuduwa said, which may increase their cholesterol levels. Although this or similar drugs may be necessary in some cases, Dr. Kahathuduwa advised that clinicians explore other treatment options first.
Mining data from previously published studies
For the new analysis, Dr. Kahathuduwa and his colleagues pooled the results of 34 previously published studies, which included medical records of more than 276,000 people with autism and close to 8 million people without the condition.
Study participants were an average age of 31 years, and 47% were female. Some studies reported age ranges that enabled the researchers to differentiate between children and adults.
People with autism were 64% more likely to develop type 1 diabetes, 146% more likely to experience type 2 diabetes, and 46% more likely to have heart disease, overall, the study found. Children with autism were almost twice as likely as their peers to develop diabetes (184%) and high blood pressure (154%).
The study found associations, not causation, and does not include detailed data about medication prescribing patterns. While it would be ideal to understand why autism is linked to cardiometabolic risk, to address the link most effectively, Dr. Kahathuduwa said the causes likely are multifactorial. Medication history and genetics each play a role in a way that is hard to untangle. Even so, Dr. Kahathuduwa said he hoped the findings prompt clinicians to reevaluate how they treat their patients with autism.
“This may be an eye opener,” he said.
An editorial accompanying the study noted that people with autism may die up to 30 years earlier than people without autism, in part because of the physical health problems surfaced in the new research. They also are more likely than others to attempt suicide.
Elizabeth M. Weir, PhD, of the Autism Research Centre at the University of Cambridge (England) and author of the editorial, argued that current health delivery models often fail autistic people by not taking their needs into account.
Dr. Weir told this news organization that making adjustments such as dimming the lights for a light-sensitive patient or allowing people with autism to bring an advocate to appointments could build rapport.
“I diagnose autism pretty much every day and I know families get so overwhelmed with all the recommendations that we give,” said Sonia Monteiro, MD, a developmental and behavioral pediatrician at Texas Children’s Hospital in Houston. Still, Dr. Monteiro said clinicians should help parents of children with autism address the potential long-term cardiovascular risks – but to do so by layering in the information rather than merely adding more bullet points to an already long presentation.
“We know this information now, but finding a way to share that with families without overwhelming them even more, I think is challenging,” Dr. Monteiro said. “But it’s not something we can ignore.”
Dr. Kahathuduwa, Dr. Weir, and Dr. Monteiro report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People with autism are more likely to face diabetes, high cholesterol, and heart disease than those without the neurologic condition, according to a study published in JAMA Pediatrics. Researchers also found that children with autism are especially likely to develop diabetes compared with their peers, and are at greater risk of hypertension, too.
While the link between autism and risk for obesity and gastrointestinal ailments is well-established, the new findings suggest that clinicians who care for these patients – particularly children – should focus on cardiometabolic health more broadly.
“Clinicians who are treating kids with autism need to pay more attention to this,” said Chanaka N. Kahathuduwa, MD, PhD, MPhil, of the department of neurology at Texas Tech University Health Sciences Center, in Lubbock, and a coauthor of the new study.
A pediatrician may prescribe an atypical antipsychotic medication such as risperidone to regulate the behavior of an autistic child, Dr. Kahathuduwa said, which may increase their cholesterol levels. Although this or similar drugs may be necessary in some cases, Dr. Kahathuduwa advised that clinicians explore other treatment options first.
Mining data from previously published studies
For the new analysis, Dr. Kahathuduwa and his colleagues pooled the results of 34 previously published studies, which included medical records of more than 276,000 people with autism and close to 8 million people without the condition.
Study participants were an average age of 31 years, and 47% were female. Some studies reported age ranges that enabled the researchers to differentiate between children and adults.
People with autism were 64% more likely to develop type 1 diabetes, 146% more likely to experience type 2 diabetes, and 46% more likely to have heart disease, overall, the study found. Children with autism were almost twice as likely as their peers to develop diabetes (184%) and high blood pressure (154%).
The study found associations, not causation, and does not include detailed data about medication prescribing patterns. While it would be ideal to understand why autism is linked to cardiometabolic risk, to address the link most effectively, Dr. Kahathuduwa said the causes likely are multifactorial. Medication history and genetics each play a role in a way that is hard to untangle. Even so, Dr. Kahathuduwa said he hoped the findings prompt clinicians to reevaluate how they treat their patients with autism.
“This may be an eye opener,” he said.
An editorial accompanying the study noted that people with autism may die up to 30 years earlier than people without autism, in part because of the physical health problems surfaced in the new research. They also are more likely than others to attempt suicide.
Elizabeth M. Weir, PhD, of the Autism Research Centre at the University of Cambridge (England) and author of the editorial, argued that current health delivery models often fail autistic people by not taking their needs into account.
Dr. Weir told this news organization that making adjustments such as dimming the lights for a light-sensitive patient or allowing people with autism to bring an advocate to appointments could build rapport.
“I diagnose autism pretty much every day and I know families get so overwhelmed with all the recommendations that we give,” said Sonia Monteiro, MD, a developmental and behavioral pediatrician at Texas Children’s Hospital in Houston. Still, Dr. Monteiro said clinicians should help parents of children with autism address the potential long-term cardiovascular risks – but to do so by layering in the information rather than merely adding more bullet points to an already long presentation.
“We know this information now, but finding a way to share that with families without overwhelming them even more, I think is challenging,” Dr. Monteiro said. “But it’s not something we can ignore.”
Dr. Kahathuduwa, Dr. Weir, and Dr. Monteiro report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People with autism are more likely to face diabetes, high cholesterol, and heart disease than those without the neurologic condition, according to a study published in JAMA Pediatrics. Researchers also found that children with autism are especially likely to develop diabetes compared with their peers, and are at greater risk of hypertension, too.
While the link between autism and risk for obesity and gastrointestinal ailments is well-established, the new findings suggest that clinicians who care for these patients – particularly children – should focus on cardiometabolic health more broadly.
“Clinicians who are treating kids with autism need to pay more attention to this,” said Chanaka N. Kahathuduwa, MD, PhD, MPhil, of the department of neurology at Texas Tech University Health Sciences Center, in Lubbock, and a coauthor of the new study.
A pediatrician may prescribe an atypical antipsychotic medication such as risperidone to regulate the behavior of an autistic child, Dr. Kahathuduwa said, which may increase their cholesterol levels. Although this or similar drugs may be necessary in some cases, Dr. Kahathuduwa advised that clinicians explore other treatment options first.
Mining data from previously published studies
For the new analysis, Dr. Kahathuduwa and his colleagues pooled the results of 34 previously published studies, which included medical records of more than 276,000 people with autism and close to 8 million people without the condition.
Study participants were an average age of 31 years, and 47% were female. Some studies reported age ranges that enabled the researchers to differentiate between children and adults.
People with autism were 64% more likely to develop type 1 diabetes, 146% more likely to experience type 2 diabetes, and 46% more likely to have heart disease, overall, the study found. Children with autism were almost twice as likely as their peers to develop diabetes (184%) and high blood pressure (154%).
The study found associations, not causation, and does not include detailed data about medication prescribing patterns. While it would be ideal to understand why autism is linked to cardiometabolic risk, to address the link most effectively, Dr. Kahathuduwa said the causes likely are multifactorial. Medication history and genetics each play a role in a way that is hard to untangle. Even so, Dr. Kahathuduwa said he hoped the findings prompt clinicians to reevaluate how they treat their patients with autism.
“This may be an eye opener,” he said.
An editorial accompanying the study noted that people with autism may die up to 30 years earlier than people without autism, in part because of the physical health problems surfaced in the new research. They also are more likely than others to attempt suicide.
Elizabeth M. Weir, PhD, of the Autism Research Centre at the University of Cambridge (England) and author of the editorial, argued that current health delivery models often fail autistic people by not taking their needs into account.
Dr. Weir told this news organization that making adjustments such as dimming the lights for a light-sensitive patient or allowing people with autism to bring an advocate to appointments could build rapport.
“I diagnose autism pretty much every day and I know families get so overwhelmed with all the recommendations that we give,” said Sonia Monteiro, MD, a developmental and behavioral pediatrician at Texas Children’s Hospital in Houston. Still, Dr. Monteiro said clinicians should help parents of children with autism address the potential long-term cardiovascular risks – but to do so by layering in the information rather than merely adding more bullet points to an already long presentation.
“We know this information now, but finding a way to share that with families without overwhelming them even more, I think is challenging,” Dr. Monteiro said. “But it’s not something we can ignore.”
Dr. Kahathuduwa, Dr. Weir, and Dr. Monteiro report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Long QT syndrome overdiagnosis persists
Five factors underlie the ongoing overdiagnosis and misdiagnosis of long QT syndrome (LQTS), including temporary QT prolongation following vasovagal syncope, a “pseudo”-positive genetic test result, family history of sudden cardiac death, transient QT prolongation, and misinterpretation of the QTc interval, a new study suggests.
Awareness of these characteristics, which led to a diagnostic reversal in 290 of 1,841 (16%) patients, could reduce the burden of overdiagnosis on the health care system and on patients and families, senior author Michael J. Ackerman, MD, PhD, of Mayo Clinic, Rochester, Minn., and colleagues conclude.
“The findings are a disturbing and disappointing sequel to the paper we published about LQTS overdiagnosis back in 2007, which showed that 2 out of every 5 patients who came to Mayo Clinic for a second opinion left without the diagnosis,” Dr. Ackerman told this news organization.
To date, Dr. Ackerman has reversed the diagnosis for 350 patients, he said.
The consequences of an LQTS diagnosis are “profound,” he noted, including years of unnecessary drug therapy, implantation of a cardioverter defibrillator, disqualification from competitive sports, and emotional stress to the individual and family.
By pointing out the five biggest mistakes his team has seen, he said, “we hope to equip the diagnostician with the means to challenge and assess the veracity of a LQTS diagnosis.”
The study was published online in the Journal of the American College of Cardiology.
Time to do better
Dr. Ackerman and colleagues analyzed electronic medical records on 290 of 1,841 (16%) patients who presented with an outside diagnosis of LQTS but subsequently were dismissed as having normal findings. The mean age of these patients at their first Mayo Clinic evaluation was 22, 60% were female, and the mean QTc interval was 427 ±25 milliseconds.
Overall, 38% of misdiagnoses were the result of misinterpretation of clinical factors; 29%, to diagnostic test misinterpretations; 17%, to an apparently positive genetic test in the context of a weak or absent phenotype; and 16%, to a family history of false LQTS or of sudden cardiac or sudden unexplained death.
More specifically, the most common cause of an LQTS misdiagnosis was QT prolongation following vasovagal syncope, which was misinterpreted as LQTS-attributed syncope.
The second most common cause was an apparently positive genetic test for an LQTS gene that turned out to be a benign or likely benign variant.
The third most common cause was an LQTS diagnosis based solely on a family history of sudden unexplained death (26 patients), QT prolongation (11 patients), or sudden cardiac arrest (9 patients).
The fourth most common cause was an isolated event of QT prolongation (44 patients). The transient QT prolongation was observed under myriad conditions unrelated to LQTS. Yet, 31 patients received a diagnosis based solely on the event.
The fifth most common cause was inclusion of the U-wave in the calculation of the QTc interval (40 patients), leading to an inaccurate interpretation of the electrocardiogram.
Dr. Ackerman noted that these LQTS diagnoses were given by heart-rhythm specialists, and most patients self-referred for a second opinion because a family member questioned the diagnosis after doing their own research.
“It’s time that we step up to the plate and do better,” Dr. Ackerman said. The team’s evaluation of the impact of the misdiagnosis on the patients’ lifestyle and quality of life showed that 45% had been restricted from competitive sports (and subsequently resumed sports activity with no adverse events); 80% had been started on beta-blockers (the drugs were discontinued in 84% as a result of the Mayo Clinic evaluation, whereas 16% opted to continue); and 10 of 22 patients (45%) who received an implanted cardioverter device underwent an extraction of the device without complications.
The authors conclude: “Although missing a patient who truly has LQTS can lead to a tragic outcome, the implications of overdiagnosed LQTS are not trivial and are potentially tragic as well.”
‘Tricky diagnosis’
LQTS specialist Peter Aziz, MD, director of pediatric electrophysiology at the Cleveland Clinic, agreed with these findings.
“Most of us ‘channelopathists’ who see LQTS for a living have a good grasp of the disease, but it can be elusive for others,” he said in an interview. “This is a tricky diagnosis. There are ends of the spectrum where people for sure don’t have it and people for sure do. Most clinicians are able to identify that.”
However, he added, “A lot of patients fall into that gray area where it may not be clear at first, even to an expert. But the expert knows how to do a comprehensive evaluation, examining episodes and symptoms and understanding whether they are relevant to LQTS or completely red herrings, and feeling confident about how they calculate the acute interval on an electrocardiogram.”
“All of these may seem mundane, but without the experience, clinicians are vulnerable to miscalculations,” he said. “That’s why our bias, as channelopathists, is that every patient who has a suspected diagnosis or is being treated for LQTS really should see an expert.”
Similarly, Arthur A.M. Wilde, MD, PhD, of the University of Amsterdam, and Peter J. Schwartz, MD, of IRCCS Istituto Auxologico Italiano, Milan, write in a related editorial that it “has to be kept in mind that both diagnostic scores and risk scores are dynamic and can be modified by time and by appropriate therapy.
“Therefore, to make hasty diagnosis of a disease that requires life-long treatment is inappropriate, especially when this is done without the support of adequate, specific experience.”
No commercial funding or relevant financial relationships were reported.
A version of this article first appeared on Medscape.com.
Five factors underlie the ongoing overdiagnosis and misdiagnosis of long QT syndrome (LQTS), including temporary QT prolongation following vasovagal syncope, a “pseudo”-positive genetic test result, family history of sudden cardiac death, transient QT prolongation, and misinterpretation of the QTc interval, a new study suggests.
Awareness of these characteristics, which led to a diagnostic reversal in 290 of 1,841 (16%) patients, could reduce the burden of overdiagnosis on the health care system and on patients and families, senior author Michael J. Ackerman, MD, PhD, of Mayo Clinic, Rochester, Minn., and colleagues conclude.
“The findings are a disturbing and disappointing sequel to the paper we published about LQTS overdiagnosis back in 2007, which showed that 2 out of every 5 patients who came to Mayo Clinic for a second opinion left without the diagnosis,” Dr. Ackerman told this news organization.
To date, Dr. Ackerman has reversed the diagnosis for 350 patients, he said.
The consequences of an LQTS diagnosis are “profound,” he noted, including years of unnecessary drug therapy, implantation of a cardioverter defibrillator, disqualification from competitive sports, and emotional stress to the individual and family.
By pointing out the five biggest mistakes his team has seen, he said, “we hope to equip the diagnostician with the means to challenge and assess the veracity of a LQTS diagnosis.”
The study was published online in the Journal of the American College of Cardiology.
Time to do better
Dr. Ackerman and colleagues analyzed electronic medical records on 290 of 1,841 (16%) patients who presented with an outside diagnosis of LQTS but subsequently were dismissed as having normal findings. The mean age of these patients at their first Mayo Clinic evaluation was 22, 60% were female, and the mean QTc interval was 427 ±25 milliseconds.
Overall, 38% of misdiagnoses were the result of misinterpretation of clinical factors; 29%, to diagnostic test misinterpretations; 17%, to an apparently positive genetic test in the context of a weak or absent phenotype; and 16%, to a family history of false LQTS or of sudden cardiac or sudden unexplained death.
More specifically, the most common cause of an LQTS misdiagnosis was QT prolongation following vasovagal syncope, which was misinterpreted as LQTS-attributed syncope.
The second most common cause was an apparently positive genetic test for an LQTS gene that turned out to be a benign or likely benign variant.
The third most common cause was an LQTS diagnosis based solely on a family history of sudden unexplained death (26 patients), QT prolongation (11 patients), or sudden cardiac arrest (9 patients).
The fourth most common cause was an isolated event of QT prolongation (44 patients). The transient QT prolongation was observed under myriad conditions unrelated to LQTS. Yet, 31 patients received a diagnosis based solely on the event.
The fifth most common cause was inclusion of the U-wave in the calculation of the QTc interval (40 patients), leading to an inaccurate interpretation of the electrocardiogram.
Dr. Ackerman noted that these LQTS diagnoses were given by heart-rhythm specialists, and most patients self-referred for a second opinion because a family member questioned the diagnosis after doing their own research.
“It’s time that we step up to the plate and do better,” Dr. Ackerman said. The team’s evaluation of the impact of the misdiagnosis on the patients’ lifestyle and quality of life showed that 45% had been restricted from competitive sports (and subsequently resumed sports activity with no adverse events); 80% had been started on beta-blockers (the drugs were discontinued in 84% as a result of the Mayo Clinic evaluation, whereas 16% opted to continue); and 10 of 22 patients (45%) who received an implanted cardioverter device underwent an extraction of the device without complications.
The authors conclude: “Although missing a patient who truly has LQTS can lead to a tragic outcome, the implications of overdiagnosed LQTS are not trivial and are potentially tragic as well.”
‘Tricky diagnosis’
LQTS specialist Peter Aziz, MD, director of pediatric electrophysiology at the Cleveland Clinic, agreed with these findings.
“Most of us ‘channelopathists’ who see LQTS for a living have a good grasp of the disease, but it can be elusive for others,” he said in an interview. “This is a tricky diagnosis. There are ends of the spectrum where people for sure don’t have it and people for sure do. Most clinicians are able to identify that.”
However, he added, “A lot of patients fall into that gray area where it may not be clear at first, even to an expert. But the expert knows how to do a comprehensive evaluation, examining episodes and symptoms and understanding whether they are relevant to LQTS or completely red herrings, and feeling confident about how they calculate the acute interval on an electrocardiogram.”
“All of these may seem mundane, but without the experience, clinicians are vulnerable to miscalculations,” he said. “That’s why our bias, as channelopathists, is that every patient who has a suspected diagnosis or is being treated for LQTS really should see an expert.”
Similarly, Arthur A.M. Wilde, MD, PhD, of the University of Amsterdam, and Peter J. Schwartz, MD, of IRCCS Istituto Auxologico Italiano, Milan, write in a related editorial that it “has to be kept in mind that both diagnostic scores and risk scores are dynamic and can be modified by time and by appropriate therapy.
“Therefore, to make hasty diagnosis of a disease that requires life-long treatment is inappropriate, especially when this is done without the support of adequate, specific experience.”
No commercial funding or relevant financial relationships were reported.
A version of this article first appeared on Medscape.com.
Five factors underlie the ongoing overdiagnosis and misdiagnosis of long QT syndrome (LQTS), including temporary QT prolongation following vasovagal syncope, a “pseudo”-positive genetic test result, family history of sudden cardiac death, transient QT prolongation, and misinterpretation of the QTc interval, a new study suggests.
Awareness of these characteristics, which led to a diagnostic reversal in 290 of 1,841 (16%) patients, could reduce the burden of overdiagnosis on the health care system and on patients and families, senior author Michael J. Ackerman, MD, PhD, of Mayo Clinic, Rochester, Minn., and colleagues conclude.
“The findings are a disturbing and disappointing sequel to the paper we published about LQTS overdiagnosis back in 2007, which showed that 2 out of every 5 patients who came to Mayo Clinic for a second opinion left without the diagnosis,” Dr. Ackerman told this news organization.
To date, Dr. Ackerman has reversed the diagnosis for 350 patients, he said.
The consequences of an LQTS diagnosis are “profound,” he noted, including years of unnecessary drug therapy, implantation of a cardioverter defibrillator, disqualification from competitive sports, and emotional stress to the individual and family.
By pointing out the five biggest mistakes his team has seen, he said, “we hope to equip the diagnostician with the means to challenge and assess the veracity of a LQTS diagnosis.”
The study was published online in the Journal of the American College of Cardiology.
Time to do better
Dr. Ackerman and colleagues analyzed electronic medical records on 290 of 1,841 (16%) patients who presented with an outside diagnosis of LQTS but subsequently were dismissed as having normal findings. The mean age of these patients at their first Mayo Clinic evaluation was 22, 60% were female, and the mean QTc interval was 427 ±25 milliseconds.
Overall, 38% of misdiagnoses were the result of misinterpretation of clinical factors; 29%, to diagnostic test misinterpretations; 17%, to an apparently positive genetic test in the context of a weak or absent phenotype; and 16%, to a family history of false LQTS or of sudden cardiac or sudden unexplained death.
More specifically, the most common cause of an LQTS misdiagnosis was QT prolongation following vasovagal syncope, which was misinterpreted as LQTS-attributed syncope.
The second most common cause was an apparently positive genetic test for an LQTS gene that turned out to be a benign or likely benign variant.
The third most common cause was an LQTS diagnosis based solely on a family history of sudden unexplained death (26 patients), QT prolongation (11 patients), or sudden cardiac arrest (9 patients).
The fourth most common cause was an isolated event of QT prolongation (44 patients). The transient QT prolongation was observed under myriad conditions unrelated to LQTS. Yet, 31 patients received a diagnosis based solely on the event.
The fifth most common cause was inclusion of the U-wave in the calculation of the QTc interval (40 patients), leading to an inaccurate interpretation of the electrocardiogram.
Dr. Ackerman noted that these LQTS diagnoses were given by heart-rhythm specialists, and most patients self-referred for a second opinion because a family member questioned the diagnosis after doing their own research.
“It’s time that we step up to the plate and do better,” Dr. Ackerman said. The team’s evaluation of the impact of the misdiagnosis on the patients’ lifestyle and quality of life showed that 45% had been restricted from competitive sports (and subsequently resumed sports activity with no adverse events); 80% had been started on beta-blockers (the drugs were discontinued in 84% as a result of the Mayo Clinic evaluation, whereas 16% opted to continue); and 10 of 22 patients (45%) who received an implanted cardioverter device underwent an extraction of the device without complications.
The authors conclude: “Although missing a patient who truly has LQTS can lead to a tragic outcome, the implications of overdiagnosed LQTS are not trivial and are potentially tragic as well.”
‘Tricky diagnosis’
LQTS specialist Peter Aziz, MD, director of pediatric electrophysiology at the Cleveland Clinic, agreed with these findings.
“Most of us ‘channelopathists’ who see LQTS for a living have a good grasp of the disease, but it can be elusive for others,” he said in an interview. “This is a tricky diagnosis. There are ends of the spectrum where people for sure don’t have it and people for sure do. Most clinicians are able to identify that.”
However, he added, “A lot of patients fall into that gray area where it may not be clear at first, even to an expert. But the expert knows how to do a comprehensive evaluation, examining episodes and symptoms and understanding whether they are relevant to LQTS or completely red herrings, and feeling confident about how they calculate the acute interval on an electrocardiogram.”
“All of these may seem mundane, but without the experience, clinicians are vulnerable to miscalculations,” he said. “That’s why our bias, as channelopathists, is that every patient who has a suspected diagnosis or is being treated for LQTS really should see an expert.”
Similarly, Arthur A.M. Wilde, MD, PhD, of the University of Amsterdam, and Peter J. Schwartz, MD, of IRCCS Istituto Auxologico Italiano, Milan, write in a related editorial that it “has to be kept in mind that both diagnostic scores and risk scores are dynamic and can be modified by time and by appropriate therapy.
“Therefore, to make hasty diagnosis of a disease that requires life-long treatment is inappropriate, especially when this is done without the support of adequate, specific experience.”
No commercial funding or relevant financial relationships were reported.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Elevated PCSK9 levels associated with psoriasis suggest new treatment target
A Mendelian randomization study employing data from nearly 300,000 individuals has linked elevated levels of the PCSK9 enzyme with an increased risk of psoriasis, suggesting it might be targetable as an intervention.
. Conversely, psoriasis risk did not appear to be affected when LDL-C was reduced by other pathways of lipid control.
This study “suggests that PCSK9 inhibition is causally associated with reduced risk of psoriasis,” reported a team of investigators led by Sizheng Steven Zhao, MD, PhD, of the division of musculoskeletal and dermatological sciences, University of Manchester (England). “Existing PCSK9 inhibitors hold potential as therapeutic targets for prevention, and possibly treatment, of psoriasis, although further clinical studies are needed,” they concluded.
In an interview, Dr. Zhao also noted that it will be interesting to look at psoriasis susceptibility in post hoc analyses of large randomized controlled trials of PCSK9 inhibitors for cardiovascular disease.
“Genetically proxied” inhibition of HMG-CoA reductase, which is targeted by statins, and NPC1L1 which is targeted by ezetimibe, “were not associated with psoriasis risk,” the investigators reported in the study, published in JAMA Dermatology.
Abnormal lipid metabolism is sufficiently common among people with psoriasis that screening in patients with moderate to severe disease is recommended in 2019 psoriasis guidelines from the American Academy of Dermatology and the National Psoriasis Foundation. However, the link between these diseases is unclear. This study was launched to explore genetically proxied relationships between psoriasis and LDL-C reductions as well as specific treatments for elevated LDL-C.
Mendelian randomizations were applied to deidentified data from two sources, a UK biobank and FinnGen, a Finnish-based project for identifying genotype-to-phenotype correlations. Genetic proxies for these variables were established on the basis of genomewide association studies on large population samples.
Ultimately, 34 genetic variants were selected to proxy for lipid lowering by PCSK9, 19 were selected to proxy for HMG-CoA reductase, and 9 for NPC1L1. In the Mendelian analyses performed on the two sources, genetically proxied PCSK9 inhibition was associated with about a 30% reduction in the odds ratio of psoriasis (OR, 0.69; P = .003). There were no robust associations with proxies for reductions in either HMG-CoA reductase or NPC1L1.
In sensitivity analyses, there was no evidence of bias from pleiotropy or genetic confounding, according to Dr. Zhao and his coauthors, who noted that the relationship between reductions in PCSK9 and reduced risk of psoriasis appeared to be independent of change in circulating LDL-C.
Given the prior evidence implicating the PCSK9 enzyme in psoriasis risk, “this is an exciting study that really highlights the importance of studying and targeting lipid metabolism in psoriasis for a few reasons,” according to Michael S. Garshick, MD, a researcher, cardiologist, and director of the cardio-rheumatology program, New York University Langone Health.
An investigator who has participated in several studies evaluating the relationship between cardiovascular risk and psoriasis, Dr. Garshick said there is increasing interest in PCSK9 as a biomarker or even a mediator of inflammation independent of blood lipid levels.
“In psoriasis regarding PCSK9, we and others have shown PCSK9 is elevated in psoriatic lesion skin, and studies are starting to investigate the unique lipidomic profile in psoriasis,” Dr. Garshick said in an interview. The study he led that showed elevated PCSK9 levels in psoriatic skin was published in 2021 in the Journal of Investigative Dermatology.
While the Mendelian randomization provides only “an inference” that PCSK9 plays a role in mediating risk of psoriasis, Dr. Zhao and coauthors cited numerous studies linking elevated PCSK9 to psoriasis pathophysiology. This not only includes the elevated PCSK9 expression in psoriatic plaques as shown by Dr. Garshick and others but several sets of experimental evidence linking PCSK9 to inflammatory pathways, including upregulation of interleukin-17 and stimulation of macrophage activation.
While Dr. Zhao and coauthors suggested that clinical trials are now needed to test the potential of PCSK9 inhibitors to modify the risk of psoriasis, Dr. Garshick indicated that there are numerous variables to unravel in the relationship between elevated lipids, PCSK9, and psoriasis.
“In our own studies, we did see a statistical correlation between circulating PCSK9 and psoriasis severity,” Dr. Garshick said. But he added, “I think we are just beginning to understand the functions of circulating (extrahepatic) PCSK9 independent of lipid metabolism.”
While he is intrigued by the evidence that PCSK9 is linked to systemic inflammation, he pointed out that several medications used to treat dyslipidemias, such as statins, are associated with an anti-inflammatory effect.
This study “further emphasizes the need to conduct clinical trials treating dyslipidemia in psoriasis, including the targeting of PCSK9, whether it is with statins with lipid lowering and potential pleiotropic anti-inflammatory properties or PCSK9 inhibition,” he said. If positive, “both would be exciting.“
From a cardiologist’s point of view, there is an upside for including patients with psoriasis in lipid-lowering trials even if the effect on psoriasis is modest. Either way, “you still get the lipid-lowering benefit, which is important for reducing atherosclerotic cardiovascular disease,” Dr. Garshick said.
Dr. Zhao reported financial relationships with UCB, although UCB did not provide funding for this study. One author reported grants from Versus Arthritis and the National Institute for Health Research Manchester Biomedical Research Centre during the study, grants from Bristol Myers Squibb, Galapagos, and Pfizer, and personal fees from Chugai Roche outside the submitted work. No other disclosures were reported. The study was supported by grants from Versus Arthritis and the NIHR Manchester Biomedical Research Centre. Dr. Garshick reported financial relationships with AbbVie and Horizon Therapeutics.
A Mendelian randomization study employing data from nearly 300,000 individuals has linked elevated levels of the PCSK9 enzyme with an increased risk of psoriasis, suggesting it might be targetable as an intervention.
. Conversely, psoriasis risk did not appear to be affected when LDL-C was reduced by other pathways of lipid control.
This study “suggests that PCSK9 inhibition is causally associated with reduced risk of psoriasis,” reported a team of investigators led by Sizheng Steven Zhao, MD, PhD, of the division of musculoskeletal and dermatological sciences, University of Manchester (England). “Existing PCSK9 inhibitors hold potential as therapeutic targets for prevention, and possibly treatment, of psoriasis, although further clinical studies are needed,” they concluded.
In an interview, Dr. Zhao also noted that it will be interesting to look at psoriasis susceptibility in post hoc analyses of large randomized controlled trials of PCSK9 inhibitors for cardiovascular disease.
“Genetically proxied” inhibition of HMG-CoA reductase, which is targeted by statins, and NPC1L1 which is targeted by ezetimibe, “were not associated with psoriasis risk,” the investigators reported in the study, published in JAMA Dermatology.
Abnormal lipid metabolism is sufficiently common among people with psoriasis that screening in patients with moderate to severe disease is recommended in 2019 psoriasis guidelines from the American Academy of Dermatology and the National Psoriasis Foundation. However, the link between these diseases is unclear. This study was launched to explore genetically proxied relationships between psoriasis and LDL-C reductions as well as specific treatments for elevated LDL-C.
Mendelian randomizations were applied to deidentified data from two sources, a UK biobank and FinnGen, a Finnish-based project for identifying genotype-to-phenotype correlations. Genetic proxies for these variables were established on the basis of genomewide association studies on large population samples.
Ultimately, 34 genetic variants were selected to proxy for lipid lowering by PCSK9, 19 were selected to proxy for HMG-CoA reductase, and 9 for NPC1L1. In the Mendelian analyses performed on the two sources, genetically proxied PCSK9 inhibition was associated with about a 30% reduction in the odds ratio of psoriasis (OR, 0.69; P = .003). There were no robust associations with proxies for reductions in either HMG-CoA reductase or NPC1L1.
In sensitivity analyses, there was no evidence of bias from pleiotropy or genetic confounding, according to Dr. Zhao and his coauthors, who noted that the relationship between reductions in PCSK9 and reduced risk of psoriasis appeared to be independent of change in circulating LDL-C.
Given the prior evidence implicating the PCSK9 enzyme in psoriasis risk, “this is an exciting study that really highlights the importance of studying and targeting lipid metabolism in psoriasis for a few reasons,” according to Michael S. Garshick, MD, a researcher, cardiologist, and director of the cardio-rheumatology program, New York University Langone Health.
An investigator who has participated in several studies evaluating the relationship between cardiovascular risk and psoriasis, Dr. Garshick said there is increasing interest in PCSK9 as a biomarker or even a mediator of inflammation independent of blood lipid levels.
“In psoriasis regarding PCSK9, we and others have shown PCSK9 is elevated in psoriatic lesion skin, and studies are starting to investigate the unique lipidomic profile in psoriasis,” Dr. Garshick said in an interview. The study he led that showed elevated PCSK9 levels in psoriatic skin was published in 2021 in the Journal of Investigative Dermatology.
While the Mendelian randomization provides only “an inference” that PCSK9 plays a role in mediating risk of psoriasis, Dr. Zhao and coauthors cited numerous studies linking elevated PCSK9 to psoriasis pathophysiology. This not only includes the elevated PCSK9 expression in psoriatic plaques as shown by Dr. Garshick and others but several sets of experimental evidence linking PCSK9 to inflammatory pathways, including upregulation of interleukin-17 and stimulation of macrophage activation.
While Dr. Zhao and coauthors suggested that clinical trials are now needed to test the potential of PCSK9 inhibitors to modify the risk of psoriasis, Dr. Garshick indicated that there are numerous variables to unravel in the relationship between elevated lipids, PCSK9, and psoriasis.
“In our own studies, we did see a statistical correlation between circulating PCSK9 and psoriasis severity,” Dr. Garshick said. But he added, “I think we are just beginning to understand the functions of circulating (extrahepatic) PCSK9 independent of lipid metabolism.”
While he is intrigued by the evidence that PCSK9 is linked to systemic inflammation, he pointed out that several medications used to treat dyslipidemias, such as statins, are associated with an anti-inflammatory effect.
This study “further emphasizes the need to conduct clinical trials treating dyslipidemia in psoriasis, including the targeting of PCSK9, whether it is with statins with lipid lowering and potential pleiotropic anti-inflammatory properties or PCSK9 inhibition,” he said. If positive, “both would be exciting.“
From a cardiologist’s point of view, there is an upside for including patients with psoriasis in lipid-lowering trials even if the effect on psoriasis is modest. Either way, “you still get the lipid-lowering benefit, which is important for reducing atherosclerotic cardiovascular disease,” Dr. Garshick said.
Dr. Zhao reported financial relationships with UCB, although UCB did not provide funding for this study. One author reported grants from Versus Arthritis and the National Institute for Health Research Manchester Biomedical Research Centre during the study, grants from Bristol Myers Squibb, Galapagos, and Pfizer, and personal fees from Chugai Roche outside the submitted work. No other disclosures were reported. The study was supported by grants from Versus Arthritis and the NIHR Manchester Biomedical Research Centre. Dr. Garshick reported financial relationships with AbbVie and Horizon Therapeutics.
A Mendelian randomization study employing data from nearly 300,000 individuals has linked elevated levels of the PCSK9 enzyme with an increased risk of psoriasis, suggesting it might be targetable as an intervention.
. Conversely, psoriasis risk did not appear to be affected when LDL-C was reduced by other pathways of lipid control.
This study “suggests that PCSK9 inhibition is causally associated with reduced risk of psoriasis,” reported a team of investigators led by Sizheng Steven Zhao, MD, PhD, of the division of musculoskeletal and dermatological sciences, University of Manchester (England). “Existing PCSK9 inhibitors hold potential as therapeutic targets for prevention, and possibly treatment, of psoriasis, although further clinical studies are needed,” they concluded.
In an interview, Dr. Zhao also noted that it will be interesting to look at psoriasis susceptibility in post hoc analyses of large randomized controlled trials of PCSK9 inhibitors for cardiovascular disease.
“Genetically proxied” inhibition of HMG-CoA reductase, which is targeted by statins, and NPC1L1 which is targeted by ezetimibe, “were not associated with psoriasis risk,” the investigators reported in the study, published in JAMA Dermatology.
Abnormal lipid metabolism is sufficiently common among people with psoriasis that screening in patients with moderate to severe disease is recommended in 2019 psoriasis guidelines from the American Academy of Dermatology and the National Psoriasis Foundation. However, the link between these diseases is unclear. This study was launched to explore genetically proxied relationships between psoriasis and LDL-C reductions as well as specific treatments for elevated LDL-C.
Mendelian randomizations were applied to deidentified data from two sources, a UK biobank and FinnGen, a Finnish-based project for identifying genotype-to-phenotype correlations. Genetic proxies for these variables were established on the basis of genomewide association studies on large population samples.
Ultimately, 34 genetic variants were selected to proxy for lipid lowering by PCSK9, 19 were selected to proxy for HMG-CoA reductase, and 9 for NPC1L1. In the Mendelian analyses performed on the two sources, genetically proxied PCSK9 inhibition was associated with about a 30% reduction in the odds ratio of psoriasis (OR, 0.69; P = .003). There were no robust associations with proxies for reductions in either HMG-CoA reductase or NPC1L1.
In sensitivity analyses, there was no evidence of bias from pleiotropy or genetic confounding, according to Dr. Zhao and his coauthors, who noted that the relationship between reductions in PCSK9 and reduced risk of psoriasis appeared to be independent of change in circulating LDL-C.
Given the prior evidence implicating the PCSK9 enzyme in psoriasis risk, “this is an exciting study that really highlights the importance of studying and targeting lipid metabolism in psoriasis for a few reasons,” according to Michael S. Garshick, MD, a researcher, cardiologist, and director of the cardio-rheumatology program, New York University Langone Health.
An investigator who has participated in several studies evaluating the relationship between cardiovascular risk and psoriasis, Dr. Garshick said there is increasing interest in PCSK9 as a biomarker or even a mediator of inflammation independent of blood lipid levels.
“In psoriasis regarding PCSK9, we and others have shown PCSK9 is elevated in psoriatic lesion skin, and studies are starting to investigate the unique lipidomic profile in psoriasis,” Dr. Garshick said in an interview. The study he led that showed elevated PCSK9 levels in psoriatic skin was published in 2021 in the Journal of Investigative Dermatology.
While the Mendelian randomization provides only “an inference” that PCSK9 plays a role in mediating risk of psoriasis, Dr. Zhao and coauthors cited numerous studies linking elevated PCSK9 to psoriasis pathophysiology. This not only includes the elevated PCSK9 expression in psoriatic plaques as shown by Dr. Garshick and others but several sets of experimental evidence linking PCSK9 to inflammatory pathways, including upregulation of interleukin-17 and stimulation of macrophage activation.
While Dr. Zhao and coauthors suggested that clinical trials are now needed to test the potential of PCSK9 inhibitors to modify the risk of psoriasis, Dr. Garshick indicated that there are numerous variables to unravel in the relationship between elevated lipids, PCSK9, and psoriasis.
“In our own studies, we did see a statistical correlation between circulating PCSK9 and psoriasis severity,” Dr. Garshick said. But he added, “I think we are just beginning to understand the functions of circulating (extrahepatic) PCSK9 independent of lipid metabolism.”
While he is intrigued by the evidence that PCSK9 is linked to systemic inflammation, he pointed out that several medications used to treat dyslipidemias, such as statins, are associated with an anti-inflammatory effect.
This study “further emphasizes the need to conduct clinical trials treating dyslipidemia in psoriasis, including the targeting of PCSK9, whether it is with statins with lipid lowering and potential pleiotropic anti-inflammatory properties or PCSK9 inhibition,” he said. If positive, “both would be exciting.“
From a cardiologist’s point of view, there is an upside for including patients with psoriasis in lipid-lowering trials even if the effect on psoriasis is modest. Either way, “you still get the lipid-lowering benefit, which is important for reducing atherosclerotic cardiovascular disease,” Dr. Garshick said.
Dr. Zhao reported financial relationships with UCB, although UCB did not provide funding for this study. One author reported grants from Versus Arthritis and the National Institute for Health Research Manchester Biomedical Research Centre during the study, grants from Bristol Myers Squibb, Galapagos, and Pfizer, and personal fees from Chugai Roche outside the submitted work. No other disclosures were reported. The study was supported by grants from Versus Arthritis and the NIHR Manchester Biomedical Research Centre. Dr. Garshick reported financial relationships with AbbVie and Horizon Therapeutics.
FROM JAMA DERMATOLOGY
Angioedema risk jumps when switching HF meds
New renin-angiotensin-system (RAS) inhibitor therapy using sacubitril-valsartan (Entresto) is no more likely to cause angioedema than starting out with an ACE inhibitor or angiotensin receptor blocker (ARB).
But the risk climbs when such patients start on an ACE inhibitor or ARB and then switch to sacubitril-valsartan, compared with those prescribed the newer drug, the only available angiotensin receptor-neprilysin inhibitor (ARNI), in the first place.
Those findings and others from a large database analysis, by researchers at the Food and Drug Administration and Harvard Medical School, may clarify and help alleviate a residual safety concern about the ARNI – that it might promote angioedema – that persists after the drug’s major HF trials.
The angioedema risk increased the most right after the switch to the ARNI from one of the older RAS inhibitors. For example, the overall risk doubled for patients who started with an ARB then switched to sacubitril-valsartan, compared with those who started on the newer drug. But it went up about 2.5 times during the first 14 days after the switch.
A similar pattern emerged for ACE inhibitors, but the increased angioedema risk reached significance only within 2 weeks of the switch from an ACE inhibitor to sacubitril-valsartan compared to starting on the latter.
The analysis, based on data from the FDA’s Sentinel adverse event reporting system, was published in the Journal of the American College of Cardiology.
A rare complication, but ...
Angioedema was rare overall in the study, with an unadjusted rate of about 6.75 per 1,000 person-years for users of ACE inhibitors, less than half that rate for ARB users, and only one-fifth that rate for sacubitril-valsartan recipients.
But even a rare complication can be a worry for drugs as widely used as RAS inhibitors. And it’s not unusual for patients cautiously started on an ACE inhibitor or ARB to be switched to sacubitril-valsartan, which is only recently a core guideline–recommended therapy for HF with reduced ejection fraction.
Such patients transitioning to the ARNI, the current study suggests, should probably be watched closely for signs of angioedema for 2 weeks but especially during the first few days. Indeed, the study’s event curves show most of the extra risk “popping up” right after the switch to sacubitril-valsartan, lead author Efe Eworuke, PhD, told this news organization.
The ARNI’s labeling, which states the drug should follow ACE inhibitors only after 36-hour washout period, “has done justice to this issue,” she said. But “whether clinicians are adhering to that, we can’t tell.”
Potentially, patients who miss the 36-hour washout between ACE inhibitors or ARBs and sacubitril-valsartan may account for the excess angioedema risk seen in the analysis, said Dr. Eworuke, with the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md.
But the analysis doesn’t nail down the window of excess risk to only 36 hours. It suggests that patients switching to the ARNI – even those pausing for 36 hours in between drugs – should probably be monitored “2 weeks or longer,” she said. “They could still have angioedema after the washout period.”
Indeed, the “timing of the switch may be critical,” according to an editorial accompanying the report. “Perhaps a longer initial exposure period of ACE inhibitor or ARB,” beyond 2 weeks, “should be considered before switching to an ARNI,” contended Robert L. Page II, PharmD, MSPH, University of Colorado Skaggs School of Pharmacy and Pharmaceutical Sciences, Aurora.
Moreover, he wrote, the study suggests that “initiation of an ARNI de novo may be safer compared with trialing an ACE inhibitor or ARB then switching to an ARNI,” and “should be a consideration when beginning guideline-directed medical therapy for patients with HF.”
New RAS inhibition with ARNI ‘protective’
Compared with ARNI “new users” who had not received any RAS inhibitor in the prior 6 months, patients in the study who switched from an ACE inhibitor to ARNI (41,548 matched pairs) showed a hazard ratio (HR) for angioedema of 1.62 (95% confidence interval [CI], 0.91-2.89), that is, only a “trend,” the report states.
But that trend became significant when the analysis considered only angioedema cases in the first 14 days after the drug switch: HR, 1.98 (95% CI, 1.11-3.53).
Those switching from an ARB to ARNI, compared with ARNI new users (37,893 matched pairs), showed a significant HR for angioedema of 2.03 (95% CI, 1.16-3.54). The effect was more pronounced when considering only angioedema arising in the first 2 weeks: HR, 2.45 (95% CI, 1.36-4.43).
Compared with new use of ACE inhibitors, new ARNI use (41,998 matched pairs) was “protective,” the report states, with an HR for angioedema of 0.18 (95% CI, 0.11-0.29). So was a switch from ACE inhibitors to the ARNI (69,639 matched pairs), with an HR of 0.31 (95% CI, 0.23-0.43).
But compared with starting with an ARB, ARNI new use (43,755 matched pairs) had a null effect on angioedema risk, HR, 0.59 (95% CI, 0.35-1.01); as did switching from an ARB to ARNI (49,137 matched pairs), HR, 0.85 (95% CI, 0.58-1.26).
The analysis has limitations, Dr. Eworuke acknowledged. The comparator groups probably differed in unknown ways given the limits of propensity matching, for example, and because the FDA’s Sentinel system data can reflect only cases that are reported, the study probably underestimates the true prevalence of angioedema.
For example, a patient may see a clinician for a milder case that resolves without a significant intervention, she noted. But “those types of angioedema would not have been captured by our study.”
Dr. Eworuke disclosed that her comments reflect her views and are not those of the Food and Drug Administration; she and the other authors, as well as editorialist Dr. Page, report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New renin-angiotensin-system (RAS) inhibitor therapy using sacubitril-valsartan (Entresto) is no more likely to cause angioedema than starting out with an ACE inhibitor or angiotensin receptor blocker (ARB).
But the risk climbs when such patients start on an ACE inhibitor or ARB and then switch to sacubitril-valsartan, compared with those prescribed the newer drug, the only available angiotensin receptor-neprilysin inhibitor (ARNI), in the first place.
Those findings and others from a large database analysis, by researchers at the Food and Drug Administration and Harvard Medical School, may clarify and help alleviate a residual safety concern about the ARNI – that it might promote angioedema – that persists after the drug’s major HF trials.
The angioedema risk increased the most right after the switch to the ARNI from one of the older RAS inhibitors. For example, the overall risk doubled for patients who started with an ARB then switched to sacubitril-valsartan, compared with those who started on the newer drug. But it went up about 2.5 times during the first 14 days after the switch.
A similar pattern emerged for ACE inhibitors, but the increased angioedema risk reached significance only within 2 weeks of the switch from an ACE inhibitor to sacubitril-valsartan compared to starting on the latter.
The analysis, based on data from the FDA’s Sentinel adverse event reporting system, was published in the Journal of the American College of Cardiology.
A rare complication, but ...
Angioedema was rare overall in the study, with an unadjusted rate of about 6.75 per 1,000 person-years for users of ACE inhibitors, less than half that rate for ARB users, and only one-fifth that rate for sacubitril-valsartan recipients.
But even a rare complication can be a worry for drugs as widely used as RAS inhibitors. And it’s not unusual for patients cautiously started on an ACE inhibitor or ARB to be switched to sacubitril-valsartan, which is only recently a core guideline–recommended therapy for HF with reduced ejection fraction.
Such patients transitioning to the ARNI, the current study suggests, should probably be watched closely for signs of angioedema for 2 weeks but especially during the first few days. Indeed, the study’s event curves show most of the extra risk “popping up” right after the switch to sacubitril-valsartan, lead author Efe Eworuke, PhD, told this news organization.
The ARNI’s labeling, which states the drug should follow ACE inhibitors only after 36-hour washout period, “has done justice to this issue,” she said. But “whether clinicians are adhering to that, we can’t tell.”
Potentially, patients who miss the 36-hour washout between ACE inhibitors or ARBs and sacubitril-valsartan may account for the excess angioedema risk seen in the analysis, said Dr. Eworuke, with the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md.
But the analysis doesn’t nail down the window of excess risk to only 36 hours. It suggests that patients switching to the ARNI – even those pausing for 36 hours in between drugs – should probably be monitored “2 weeks or longer,” she said. “They could still have angioedema after the washout period.”
Indeed, the “timing of the switch may be critical,” according to an editorial accompanying the report. “Perhaps a longer initial exposure period of ACE inhibitor or ARB,” beyond 2 weeks, “should be considered before switching to an ARNI,” contended Robert L. Page II, PharmD, MSPH, University of Colorado Skaggs School of Pharmacy and Pharmaceutical Sciences, Aurora.
Moreover, he wrote, the study suggests that “initiation of an ARNI de novo may be safer compared with trialing an ACE inhibitor or ARB then switching to an ARNI,” and “should be a consideration when beginning guideline-directed medical therapy for patients with HF.”
New RAS inhibition with ARNI ‘protective’
Compared with ARNI “new users” who had not received any RAS inhibitor in the prior 6 months, patients in the study who switched from an ACE inhibitor to ARNI (41,548 matched pairs) showed a hazard ratio (HR) for angioedema of 1.62 (95% confidence interval [CI], 0.91-2.89), that is, only a “trend,” the report states.
But that trend became significant when the analysis considered only angioedema cases in the first 14 days after the drug switch: HR, 1.98 (95% CI, 1.11-3.53).
Those switching from an ARB to ARNI, compared with ARNI new users (37,893 matched pairs), showed a significant HR for angioedema of 2.03 (95% CI, 1.16-3.54). The effect was more pronounced when considering only angioedema arising in the first 2 weeks: HR, 2.45 (95% CI, 1.36-4.43).
Compared with new use of ACE inhibitors, new ARNI use (41,998 matched pairs) was “protective,” the report states, with an HR for angioedema of 0.18 (95% CI, 0.11-0.29). So was a switch from ACE inhibitors to the ARNI (69,639 matched pairs), with an HR of 0.31 (95% CI, 0.23-0.43).
But compared with starting with an ARB, ARNI new use (43,755 matched pairs) had a null effect on angioedema risk, HR, 0.59 (95% CI, 0.35-1.01); as did switching from an ARB to ARNI (49,137 matched pairs), HR, 0.85 (95% CI, 0.58-1.26).
The analysis has limitations, Dr. Eworuke acknowledged. The comparator groups probably differed in unknown ways given the limits of propensity matching, for example, and because the FDA’s Sentinel system data can reflect only cases that are reported, the study probably underestimates the true prevalence of angioedema.
For example, a patient may see a clinician for a milder case that resolves without a significant intervention, she noted. But “those types of angioedema would not have been captured by our study.”
Dr. Eworuke disclosed that her comments reflect her views and are not those of the Food and Drug Administration; she and the other authors, as well as editorialist Dr. Page, report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New renin-angiotensin-system (RAS) inhibitor therapy using sacubitril-valsartan (Entresto) is no more likely to cause angioedema than starting out with an ACE inhibitor or angiotensin receptor blocker (ARB).
But the risk climbs when such patients start on an ACE inhibitor or ARB and then switch to sacubitril-valsartan, compared with those prescribed the newer drug, the only available angiotensin receptor-neprilysin inhibitor (ARNI), in the first place.
Those findings and others from a large database analysis, by researchers at the Food and Drug Administration and Harvard Medical School, may clarify and help alleviate a residual safety concern about the ARNI – that it might promote angioedema – that persists after the drug’s major HF trials.
The angioedema risk increased the most right after the switch to the ARNI from one of the older RAS inhibitors. For example, the overall risk doubled for patients who started with an ARB then switched to sacubitril-valsartan, compared with those who started on the newer drug. But it went up about 2.5 times during the first 14 days after the switch.
A similar pattern emerged for ACE inhibitors, but the increased angioedema risk reached significance only within 2 weeks of the switch from an ACE inhibitor to sacubitril-valsartan compared to starting on the latter.
The analysis, based on data from the FDA’s Sentinel adverse event reporting system, was published in the Journal of the American College of Cardiology.
A rare complication, but ...
Angioedema was rare overall in the study, with an unadjusted rate of about 6.75 per 1,000 person-years for users of ACE inhibitors, less than half that rate for ARB users, and only one-fifth that rate for sacubitril-valsartan recipients.
But even a rare complication can be a worry for drugs as widely used as RAS inhibitors. And it’s not unusual for patients cautiously started on an ACE inhibitor or ARB to be switched to sacubitril-valsartan, which is only recently a core guideline–recommended therapy for HF with reduced ejection fraction.
Such patients transitioning to the ARNI, the current study suggests, should probably be watched closely for signs of angioedema for 2 weeks but especially during the first few days. Indeed, the study’s event curves show most of the extra risk “popping up” right after the switch to sacubitril-valsartan, lead author Efe Eworuke, PhD, told this news organization.
The ARNI’s labeling, which states the drug should follow ACE inhibitors only after 36-hour washout period, “has done justice to this issue,” she said. But “whether clinicians are adhering to that, we can’t tell.”
Potentially, patients who miss the 36-hour washout between ACE inhibitors or ARBs and sacubitril-valsartan may account for the excess angioedema risk seen in the analysis, said Dr. Eworuke, with the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md.
But the analysis doesn’t nail down the window of excess risk to only 36 hours. It suggests that patients switching to the ARNI – even those pausing for 36 hours in between drugs – should probably be monitored “2 weeks or longer,” she said. “They could still have angioedema after the washout period.”
Indeed, the “timing of the switch may be critical,” according to an editorial accompanying the report. “Perhaps a longer initial exposure period of ACE inhibitor or ARB,” beyond 2 weeks, “should be considered before switching to an ARNI,” contended Robert L. Page II, PharmD, MSPH, University of Colorado Skaggs School of Pharmacy and Pharmaceutical Sciences, Aurora.
Moreover, he wrote, the study suggests that “initiation of an ARNI de novo may be safer compared with trialing an ACE inhibitor or ARB then switching to an ARNI,” and “should be a consideration when beginning guideline-directed medical therapy for patients with HF.”
New RAS inhibition with ARNI ‘protective’
Compared with ARNI “new users” who had not received any RAS inhibitor in the prior 6 months, patients in the study who switched from an ACE inhibitor to ARNI (41,548 matched pairs) showed a hazard ratio (HR) for angioedema of 1.62 (95% confidence interval [CI], 0.91-2.89), that is, only a “trend,” the report states.
But that trend became significant when the analysis considered only angioedema cases in the first 14 days after the drug switch: HR, 1.98 (95% CI, 1.11-3.53).
Those switching from an ARB to ARNI, compared with ARNI new users (37,893 matched pairs), showed a significant HR for angioedema of 2.03 (95% CI, 1.16-3.54). The effect was more pronounced when considering only angioedema arising in the first 2 weeks: HR, 2.45 (95% CI, 1.36-4.43).
Compared with new use of ACE inhibitors, new ARNI use (41,998 matched pairs) was “protective,” the report states, with an HR for angioedema of 0.18 (95% CI, 0.11-0.29). So was a switch from ACE inhibitors to the ARNI (69,639 matched pairs), with an HR of 0.31 (95% CI, 0.23-0.43).
But compared with starting with an ARB, ARNI new use (43,755 matched pairs) had a null effect on angioedema risk, HR, 0.59 (95% CI, 0.35-1.01); as did switching from an ARB to ARNI (49,137 matched pairs), HR, 0.85 (95% CI, 0.58-1.26).
The analysis has limitations, Dr. Eworuke acknowledged. The comparator groups probably differed in unknown ways given the limits of propensity matching, for example, and because the FDA’s Sentinel system data can reflect only cases that are reported, the study probably underestimates the true prevalence of angioedema.
For example, a patient may see a clinician for a milder case that resolves without a significant intervention, she noted. But “those types of angioedema would not have been captured by our study.”
Dr. Eworuke disclosed that her comments reflect her views and are not those of the Food and Drug Administration; she and the other authors, as well as editorialist Dr. Page, report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Noninvasive liver test may help select asymptomatic candidates for heart failure tests
A noninvasive test for liver disease may be a useful, low-cost screening tool to select asymptomatic candidates for a detailed examination of heart failure with preserved ejection fraction (HFpEF), say authors of a report published in Gastro Hep Advances.
The fibrosis-4 (FIB-4) index was a significant predictor of high HFpEF risk, wrote Chisato Okamoto, MD, of the department of medical biochemistry at Osaka University Graduate School of Medicine and the National Cerebral and Cardiovascular Center in Japan, and colleagues.
“Recognition of heart failure with preserved ejection fraction at an early stage in mass screening is desirable, but difficult to achieve,” the authors wrote. “The FIB-4 index is calculated using only four parameters that are routinely evaluated in general health check-up programs.”
HFpEF is an emerging disease in recent years with a poor prognosis, they wrote. Early diagnosis can be challenging for several reasons, particularly because HFpEF patients are often asymptomatic until late in the disease process and have normal left ventricular filling pressures at rest. By using a tool to select probable cases from subclinical participants in a health check-up program, clinicians can refer patients for a diastolic stress test, which is considered the gold standard for diagnosing HFpEF.
Previous studies have found that the FIB-4 index, a noninvasive tool to estimate liver stiffness and fibrosis, is associated with a higher risk of major adverse cardiovascular events (MACE) in patients with HFpEF. In addition, patients with nonalcoholic fatty liver disease (NAFLD) have a twofold higher prevalence of HFpEF than the general population.
Dr. Okamoto and colleagues examined the association between the FIB-4 index and HFpEF risk based on the Heart Failure Association’s diagnostic algorithm for HFpEF in patients with breathlessness (HFA-PEFF). The researchers looked at the prognostic impact of the FIB-4 index in 710 patients who participated in a health check-up program in the rural community of Arita-cho, Japan, between 2006 and 2007. They excluded participants with a history of cardiovascular disease or reduced left ventricular systolic function (LVEF < 50%). Researchers calculated the FIB-4 index and HFA-PEFF score for all participants.
First, using the HFA-PEFF scores, the researchers sorted participants into five groups by HFpEF risk: 215 (30%) with zero points, 100 (14%) with 1 point, 171 (24%) with 2 points, 163 (23%) with 3 points, and 61 (9%) with 4-6 points. Participants in the high-risk group (scores 4-6) were older, mostly men, and had higher blood pressure, alcohol intake, hypertension, dyslipidemia, and liver disease. The higher the HFpEF risk group, the higher the rates of all-cause mortality, hospitalization for heart failure, and MACE.
Overall, the FIB-4 index was correlated with the HFpEF risk groups and showed a stepwise increase across the groups, with .94 for the low-risk group, 1.45 for the intermediate-risk group, and 1.99 for the high-risk group, the authors wrote. The FIB-4 index also correlated with markers associated with components of the HFA-PEFF scoring system.
Using multivariate logistic regression analysis, the FIB-4 index was associated with a high HFpEF risk, and an increase in FIB-4 was associated with increased odds of high HFpEF risk. The association remained significant across four separate models that accounted for risk factors associated with lifestyle-related diseases, blood parameters associated with liver disease, and chronic conditions such as hypertension, dyslipidemia, diabetes mellitus, and liver disease.
In additional area under the curve (AUC) analyses, the FIB-4 index was a significant predictor of high HFpEF risk. At cutoff values typically used for advanced liver fibrosis in NAFLD, a FIB-4 cutoff of 1.3 or less had a sensitivity of 85.2%, while a FIB-4 cutoff of 2.67 or higher had a specificity of 94.8%. At alternate cutoff values typically used for patients with HIV/hepatitis C virus infection, a FIB-4 cutoff of less than 1.45 had a sensitivity of 75.4%, while a FIB-4 cutoff of greater than 3.25 had a specificity of 98%.
Using cutoffs of 1.3 and 2.67, a higher FIB-4 was associated with higher rates of clinical events and MACE, as well as a higher HFpEF risk. Using the alternate cutoffs of 1.45 and 3.25, prognostic stratification of clinical events and MACE was also possible.
When all variables were included in the multivariate model, the FIB-4 index remained a significant prognostic predictor. The FIB-4 index stratified clinical prognosis was also an independent predictor of all-cause mortality and hospitalization for heart failure.
Although additional studies are needed to reveal the interaction between liver and heart function, the study authors wrote, the findings provide valuable insights that can help discover the cardiohepatic interaction to reduce the development of HFpEF.
“Since it can be easily, quickly, and inexpensively measured, routine or repeated measurements of the FIB-4 index could help in selecting preferred candidates for detailed examination of HFpEF risk, which may improve clinical outcomes by diagnosing HFpEF at an early stage,” they wrote.
The study was supported by grants from the Osaka Medical Research Foundation for Intractable Disease, the Japan Arteriosclerosis Prevention Fund, the Japan Society for the Promotion of Science, and the Japan Heart Foundation. The authors disclosed no conflicts.
The 2021 NAFLD clinical care pathway is a shining example of how a simple score like the fibrosis-4 (FIB-4) index – paired sequentially with a second noninvasive test like vibration-controlled elastography – can provide an accurate, cost-effective screening tool and risk stratification and further limit invasive testing such as liver biopsy.
Broader use of FIB-4 by cardiovascular and hepatology providers may increase earlier identification of NAFLD or HFpEF or both.
Anand S. Shah, MD, is director of hepatology at Atlanta VA Healthcare and assistant professor of medicine, division of digestive disease, department of medicine, Emory University, Atlanta. He has no financial conflicts.
The 2021 NAFLD clinical care pathway is a shining example of how a simple score like the fibrosis-4 (FIB-4) index – paired sequentially with a second noninvasive test like vibration-controlled elastography – can provide an accurate, cost-effective screening tool and risk stratification and further limit invasive testing such as liver biopsy.
Broader use of FIB-4 by cardiovascular and hepatology providers may increase earlier identification of NAFLD or HFpEF or both.
Anand S. Shah, MD, is director of hepatology at Atlanta VA Healthcare and assistant professor of medicine, division of digestive disease, department of medicine, Emory University, Atlanta. He has no financial conflicts.
The 2021 NAFLD clinical care pathway is a shining example of how a simple score like the fibrosis-4 (FIB-4) index – paired sequentially with a second noninvasive test like vibration-controlled elastography – can provide an accurate, cost-effective screening tool and risk stratification and further limit invasive testing such as liver biopsy.
Broader use of FIB-4 by cardiovascular and hepatology providers may increase earlier identification of NAFLD or HFpEF or both.
Anand S. Shah, MD, is director of hepatology at Atlanta VA Healthcare and assistant professor of medicine, division of digestive disease, department of medicine, Emory University, Atlanta. He has no financial conflicts.
A noninvasive test for liver disease may be a useful, low-cost screening tool to select asymptomatic candidates for a detailed examination of heart failure with preserved ejection fraction (HFpEF), say authors of a report published in Gastro Hep Advances.
The fibrosis-4 (FIB-4) index was a significant predictor of high HFpEF risk, wrote Chisato Okamoto, MD, of the department of medical biochemistry at Osaka University Graduate School of Medicine and the National Cerebral and Cardiovascular Center in Japan, and colleagues.
“Recognition of heart failure with preserved ejection fraction at an early stage in mass screening is desirable, but difficult to achieve,” the authors wrote. “The FIB-4 index is calculated using only four parameters that are routinely evaluated in general health check-up programs.”
HFpEF is an emerging disease in recent years with a poor prognosis, they wrote. Early diagnosis can be challenging for several reasons, particularly because HFpEF patients are often asymptomatic until late in the disease process and have normal left ventricular filling pressures at rest. By using a tool to select probable cases from subclinical participants in a health check-up program, clinicians can refer patients for a diastolic stress test, which is considered the gold standard for diagnosing HFpEF.
Previous studies have found that the FIB-4 index, a noninvasive tool to estimate liver stiffness and fibrosis, is associated with a higher risk of major adverse cardiovascular events (MACE) in patients with HFpEF. In addition, patients with nonalcoholic fatty liver disease (NAFLD) have a twofold higher prevalence of HFpEF than the general population.
Dr. Okamoto and colleagues examined the association between the FIB-4 index and HFpEF risk based on the Heart Failure Association’s diagnostic algorithm for HFpEF in patients with breathlessness (HFA-PEFF). The researchers looked at the prognostic impact of the FIB-4 index in 710 patients who participated in a health check-up program in the rural community of Arita-cho, Japan, between 2006 and 2007. They excluded participants with a history of cardiovascular disease or reduced left ventricular systolic function (LVEF < 50%). Researchers calculated the FIB-4 index and HFA-PEFF score for all participants.
First, using the HFA-PEFF scores, the researchers sorted participants into five groups by HFpEF risk: 215 (30%) with zero points, 100 (14%) with 1 point, 171 (24%) with 2 points, 163 (23%) with 3 points, and 61 (9%) with 4-6 points. Participants in the high-risk group (scores 4-6) were older, mostly men, and had higher blood pressure, alcohol intake, hypertension, dyslipidemia, and liver disease. The higher the HFpEF risk group, the higher the rates of all-cause mortality, hospitalization for heart failure, and MACE.
Overall, the FIB-4 index was correlated with the HFpEF risk groups and showed a stepwise increase across the groups, with .94 for the low-risk group, 1.45 for the intermediate-risk group, and 1.99 for the high-risk group, the authors wrote. The FIB-4 index also correlated with markers associated with components of the HFA-PEFF scoring system.
Using multivariate logistic regression analysis, the FIB-4 index was associated with a high HFpEF risk, and an increase in FIB-4 was associated with increased odds of high HFpEF risk. The association remained significant across four separate models that accounted for risk factors associated with lifestyle-related diseases, blood parameters associated with liver disease, and chronic conditions such as hypertension, dyslipidemia, diabetes mellitus, and liver disease.
In additional area under the curve (AUC) analyses, the FIB-4 index was a significant predictor of high HFpEF risk. At cutoff values typically used for advanced liver fibrosis in NAFLD, a FIB-4 cutoff of 1.3 or less had a sensitivity of 85.2%, while a FIB-4 cutoff of 2.67 or higher had a specificity of 94.8%. At alternate cutoff values typically used for patients with HIV/hepatitis C virus infection, a FIB-4 cutoff of less than 1.45 had a sensitivity of 75.4%, while a FIB-4 cutoff of greater than 3.25 had a specificity of 98%.
Using cutoffs of 1.3 and 2.67, a higher FIB-4 was associated with higher rates of clinical events and MACE, as well as a higher HFpEF risk. Using the alternate cutoffs of 1.45 and 3.25, prognostic stratification of clinical events and MACE was also possible.
When all variables were included in the multivariate model, the FIB-4 index remained a significant prognostic predictor. The FIB-4 index stratified clinical prognosis was also an independent predictor of all-cause mortality and hospitalization for heart failure.
Although additional studies are needed to reveal the interaction between liver and heart function, the study authors wrote, the findings provide valuable insights that can help discover the cardiohepatic interaction to reduce the development of HFpEF.
“Since it can be easily, quickly, and inexpensively measured, routine or repeated measurements of the FIB-4 index could help in selecting preferred candidates for detailed examination of HFpEF risk, which may improve clinical outcomes by diagnosing HFpEF at an early stage,” they wrote.
The study was supported by grants from the Osaka Medical Research Foundation for Intractable Disease, the Japan Arteriosclerosis Prevention Fund, the Japan Society for the Promotion of Science, and the Japan Heart Foundation. The authors disclosed no conflicts.
A noninvasive test for liver disease may be a useful, low-cost screening tool to select asymptomatic candidates for a detailed examination of heart failure with preserved ejection fraction (HFpEF), say authors of a report published in Gastro Hep Advances.
The fibrosis-4 (FIB-4) index was a significant predictor of high HFpEF risk, wrote Chisato Okamoto, MD, of the department of medical biochemistry at Osaka University Graduate School of Medicine and the National Cerebral and Cardiovascular Center in Japan, and colleagues.
“Recognition of heart failure with preserved ejection fraction at an early stage in mass screening is desirable, but difficult to achieve,” the authors wrote. “The FIB-4 index is calculated using only four parameters that are routinely evaluated in general health check-up programs.”
HFpEF is an emerging disease in recent years with a poor prognosis, they wrote. Early diagnosis can be challenging for several reasons, particularly because HFpEF patients are often asymptomatic until late in the disease process and have normal left ventricular filling pressures at rest. By using a tool to select probable cases from subclinical participants in a health check-up program, clinicians can refer patients for a diastolic stress test, which is considered the gold standard for diagnosing HFpEF.
Previous studies have found that the FIB-4 index, a noninvasive tool to estimate liver stiffness and fibrosis, is associated with a higher risk of major adverse cardiovascular events (MACE) in patients with HFpEF. In addition, patients with nonalcoholic fatty liver disease (NAFLD) have a twofold higher prevalence of HFpEF than the general population.
Dr. Okamoto and colleagues examined the association between the FIB-4 index and HFpEF risk based on the Heart Failure Association’s diagnostic algorithm for HFpEF in patients with breathlessness (HFA-PEFF). The researchers looked at the prognostic impact of the FIB-4 index in 710 patients who participated in a health check-up program in the rural community of Arita-cho, Japan, between 2006 and 2007. They excluded participants with a history of cardiovascular disease or reduced left ventricular systolic function (LVEF < 50%). Researchers calculated the FIB-4 index and HFA-PEFF score for all participants.
First, using the HFA-PEFF scores, the researchers sorted participants into five groups by HFpEF risk: 215 (30%) with zero points, 100 (14%) with 1 point, 171 (24%) with 2 points, 163 (23%) with 3 points, and 61 (9%) with 4-6 points. Participants in the high-risk group (scores 4-6) were older, mostly men, and had higher blood pressure, alcohol intake, hypertension, dyslipidemia, and liver disease. The higher the HFpEF risk group, the higher the rates of all-cause mortality, hospitalization for heart failure, and MACE.
Overall, the FIB-4 index was correlated with the HFpEF risk groups and showed a stepwise increase across the groups, with .94 for the low-risk group, 1.45 for the intermediate-risk group, and 1.99 for the high-risk group, the authors wrote. The FIB-4 index also correlated with markers associated with components of the HFA-PEFF scoring system.
Using multivariate logistic regression analysis, the FIB-4 index was associated with a high HFpEF risk, and an increase in FIB-4 was associated with increased odds of high HFpEF risk. The association remained significant across four separate models that accounted for risk factors associated with lifestyle-related diseases, blood parameters associated with liver disease, and chronic conditions such as hypertension, dyslipidemia, diabetes mellitus, and liver disease.
In additional area under the curve (AUC) analyses, the FIB-4 index was a significant predictor of high HFpEF risk. At cutoff values typically used for advanced liver fibrosis in NAFLD, a FIB-4 cutoff of 1.3 or less had a sensitivity of 85.2%, while a FIB-4 cutoff of 2.67 or higher had a specificity of 94.8%. At alternate cutoff values typically used for patients with HIV/hepatitis C virus infection, a FIB-4 cutoff of less than 1.45 had a sensitivity of 75.4%, while a FIB-4 cutoff of greater than 3.25 had a specificity of 98%.
Using cutoffs of 1.3 and 2.67, a higher FIB-4 was associated with higher rates of clinical events and MACE, as well as a higher HFpEF risk. Using the alternate cutoffs of 1.45 and 3.25, prognostic stratification of clinical events and MACE was also possible.
When all variables were included in the multivariate model, the FIB-4 index remained a significant prognostic predictor. The FIB-4 index stratified clinical prognosis was also an independent predictor of all-cause mortality and hospitalization for heart failure.
Although additional studies are needed to reveal the interaction between liver and heart function, the study authors wrote, the findings provide valuable insights that can help discover the cardiohepatic interaction to reduce the development of HFpEF.
“Since it can be easily, quickly, and inexpensively measured, routine or repeated measurements of the FIB-4 index could help in selecting preferred candidates for detailed examination of HFpEF risk, which may improve clinical outcomes by diagnosing HFpEF at an early stage,” they wrote.
The study was supported by grants from the Osaka Medical Research Foundation for Intractable Disease, the Japan Arteriosclerosis Prevention Fund, the Japan Society for the Promotion of Science, and the Japan Heart Foundation. The authors disclosed no conflicts.
FROM GASTRO HEP ADVANCES