User login
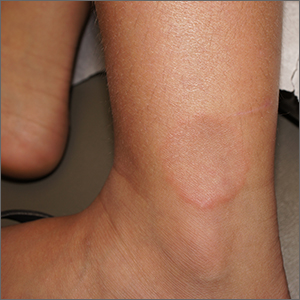
Smooth plaque on ankle
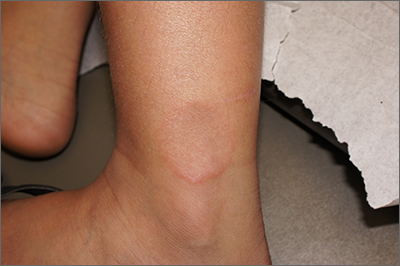
A 4-mm punch biopsy of the annular border confirmed a diagnosis of localized granuloma annulare (GA).
There is a long list of differential diagnoses for annular patches and plaques; it includes tinea corporis and important systemic diseases such as sarcoidosis and Lyme disease. Clinical features of GA include annular, minimally scaly patches to plaques with central clearing on extensor surfaces in children and adults. Sometimes GA is much more widespread. Often, the diagnosis can be made clinically, but a punch biopsy of the deep dermis will confirm the diagnosis by showing palisading or interstitial granulomatous inflammation, necrobiotic collagen, and often mucin.
GA is a common inflammatory disorder with an uncertain etiology. Localized GA affects children and adults and is often self limiting. It may, however, last for months or years before resolving. Disseminated disease is much more recalcitrant with few good treatment options if topical steroids or phototherapy fails. Treatment for localized disease is much more successful with topical or intralesional steroids.
Trauma can cause a localized plaque to resolve; a lesion may resolve soon after a biopsy is performed. Possible related conditions include diabetes, thyroid disease, hepatitis C, and hyperlipidemia; but there is no consensus on focused screening. Similarly, associations or nonassociations with malignancy in adults have been cited, but evidence is lacking.1
In this case, the patient and his family were reassured that the diagnosis wasn’t serious. In a single visit, he received a series of 6 to 7 injections of 10 mg/mL triamcinolone which led to resolution of the lesion in 4 weeks.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Piette EW, Rosenbach M. Granuloma annulare: pathogenesis, disease associations and triggers, and therapeutic options. J Am Acad Dermatol. 2016;75:467-479. doi: 10.1016/j.jaad.2015.03.055

A 4-mm punch biopsy of the annular border confirmed a diagnosis of localized granuloma annulare (GA).
There is a long list of differential diagnoses for annular patches and plaques; it includes tinea corporis and important systemic diseases such as sarcoidosis and Lyme disease. Clinical features of GA include annular, minimally scaly patches to plaques with central clearing on extensor surfaces in children and adults. Sometimes GA is much more widespread. Often, the diagnosis can be made clinically, but a punch biopsy of the deep dermis will confirm the diagnosis by showing palisading or interstitial granulomatous inflammation, necrobiotic collagen, and often mucin.
GA is a common inflammatory disorder with an uncertain etiology. Localized GA affects children and adults and is often self limiting. It may, however, last for months or years before resolving. Disseminated disease is much more recalcitrant with few good treatment options if topical steroids or phototherapy fails. Treatment for localized disease is much more successful with topical or intralesional steroids.
Trauma can cause a localized plaque to resolve; a lesion may resolve soon after a biopsy is performed. Possible related conditions include diabetes, thyroid disease, hepatitis C, and hyperlipidemia; but there is no consensus on focused screening. Similarly, associations or nonassociations with malignancy in adults have been cited, but evidence is lacking.1
In this case, the patient and his family were reassured that the diagnosis wasn’t serious. In a single visit, he received a series of 6 to 7 injections of 10 mg/mL triamcinolone which led to resolution of the lesion in 4 weeks.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

A 4-mm punch biopsy of the annular border confirmed a diagnosis of localized granuloma annulare (GA).
There is a long list of differential diagnoses for annular patches and plaques; it includes tinea corporis and important systemic diseases such as sarcoidosis and Lyme disease. Clinical features of GA include annular, minimally scaly patches to plaques with central clearing on extensor surfaces in children and adults. Sometimes GA is much more widespread. Often, the diagnosis can be made clinically, but a punch biopsy of the deep dermis will confirm the diagnosis by showing palisading or interstitial granulomatous inflammation, necrobiotic collagen, and often mucin.
GA is a common inflammatory disorder with an uncertain etiology. Localized GA affects children and adults and is often self limiting. It may, however, last for months or years before resolving. Disseminated disease is much more recalcitrant with few good treatment options if topical steroids or phototherapy fails. Treatment for localized disease is much more successful with topical or intralesional steroids.
Trauma can cause a localized plaque to resolve; a lesion may resolve soon after a biopsy is performed. Possible related conditions include diabetes, thyroid disease, hepatitis C, and hyperlipidemia; but there is no consensus on focused screening. Similarly, associations or nonassociations with malignancy in adults have been cited, but evidence is lacking.1
In this case, the patient and his family were reassured that the diagnosis wasn’t serious. In a single visit, he received a series of 6 to 7 injections of 10 mg/mL triamcinolone which led to resolution of the lesion in 4 weeks.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Piette EW, Rosenbach M. Granuloma annulare: pathogenesis, disease associations and triggers, and therapeutic options. J Am Acad Dermatol. 2016;75:467-479. doi: 10.1016/j.jaad.2015.03.055
1. Piette EW, Rosenbach M. Granuloma annulare: pathogenesis, disease associations and triggers, and therapeutic options. J Am Acad Dermatol. 2016;75:467-479. doi: 10.1016/j.jaad.2015.03.055
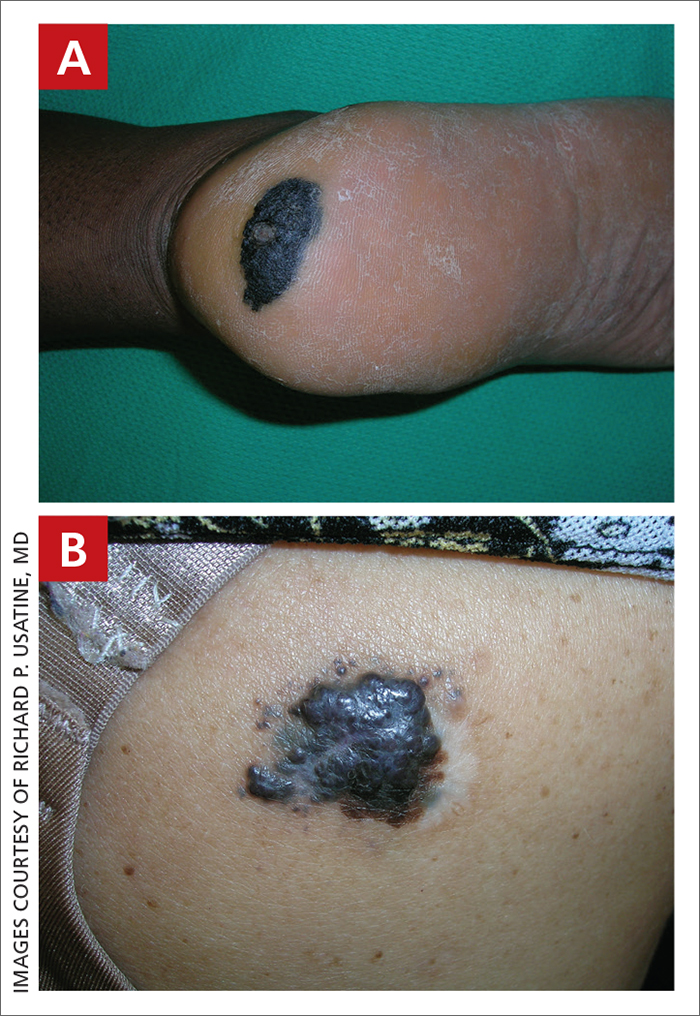
Melanoma
THE COMPARISON
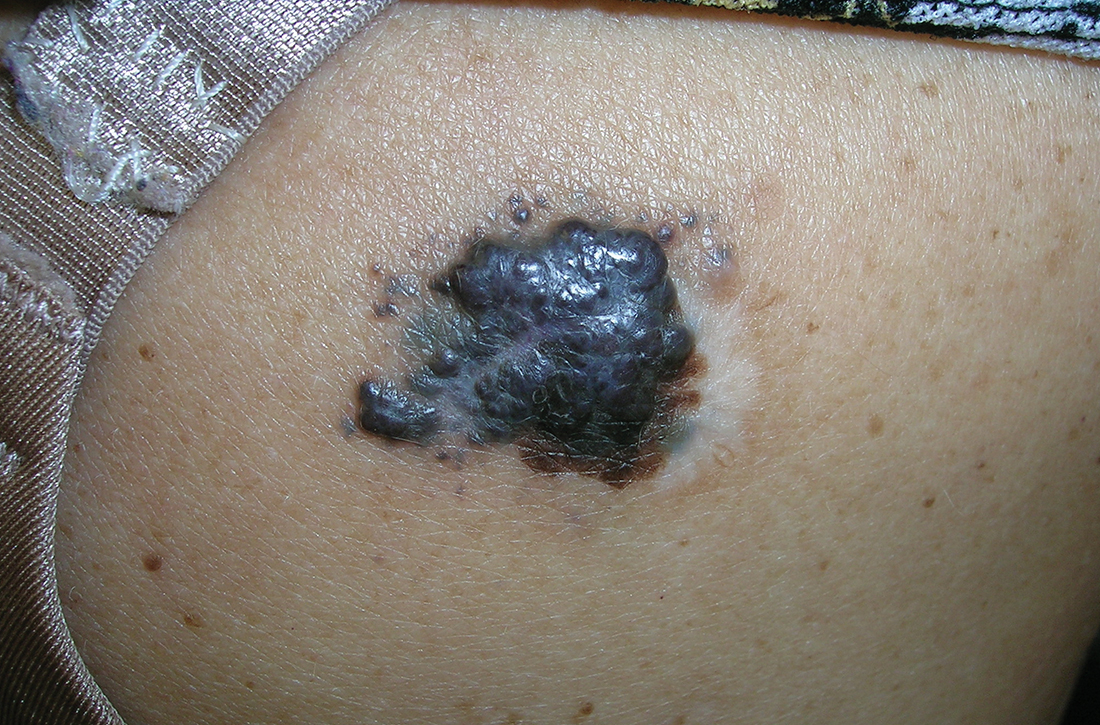
A Acral lentiginous melanoma on the sole of the foot of a 30-year-old Black woman. The depth of the lesion was 2 mm with a positive sentinel lymph node biopsy.
B Nodular melanoma on the shoulder of a 63-year-old Hispanic woman. The depth of the lesion was 5.5 mm with a positive sentinel lymph node biopsy.
Melanoma occurs less frequently in individuals with darker skin types than in those with lighter skin types but is associated with higher rates of morbidity and mortality in this patient population.1-7 In the cases shown here (A and B), both patients had advanced melanomas with large primary lesions and lymph node metastases.
Epidemiology
A systematic review by Higgins et al6 reported the following on the epidemiology of melanomas in patients with skin of color:
- African Americans have deeper tumors at the time of diagnosis, in addition to increased rates of regionally advanced and distant disease. Lesions generally are located on the lower extremities and have an increased propensity for ulceration. Acral lentiginous melanoma is the most common melanoma subtype found in African American patients.6
- In Hispanic individuals, superficial spreading melanoma is the most common melanoma subtype. Lower extremity lesions are more common relative to White individuals. Hispanic individuals have the highest rate of oral cavity melanomas across all ethnic groups.6
- In Asian individuals, acral and subungual sites are most common. Specifically, Pacific Islanders have the highest proportion of mucosal melanomas across all ethnic groups.6
Key clinical features in people with darker skin tones
Melanomas are found more often on the palms, soles, nail units, oral cavity, and mucosae.6 The melanomas have the same clinical and dermoscopic features found in individuals with lighter skin tones.
Worth noting
Factors that may contribute to the diagnosis of more advanced melanomas in racial/ethnic minorities in the United States include:
- decreased access to health care based on lack of health insurance and low socioeconomic status,
- less awareness of the risk of melanoma among patients and health care providers because melanoma is less common in persons of color, and
- lesions found in areas less likely to be seen in screening examinations, such as the soles of the feet and the oral and genital mucosae.
Health disparity highlight
- In a large US study of 96,953 patients with a diagnosis of cutaneous melanoma from 1992 to 2009, the proportion of later-stage melanoma—stages II to IV—was greater in Black patients compared to White patients.7
- Based on this same data set, White patients had the longest survival time (P <. 05), followed by Hispanic (P < .05), Asian American/Native American/Pacific Islander (P < .05), and Black (P < .05) patients, respectively.7
- In Miami-Dade County, one study of 1690 melanoma cases found that 48% of Black patients had regional or distant disease at presentation compared to 22% of White patients (P = .015).5 Analysis of multiple factors found that only race was a significant predictor for late-stage melanoma (P < .001). Black patients in this study were 3 times more likely than others to be diagnosed with melanoma at a late stage (P = .07).5
- Black patients in the United States are more likely to have a delayed time from diagnosis to definitive surgery even when researchers controlled for type of health insurance and stage of diagnosis.8
Final thoughts
Efforts are needed to overcome these disparities by:
- educating patients with skin of color and their health care providers about the risks of advanced melanoma with the goal of prevention and earlier diagnosis;
- breaking down barriers to care caused by poverty, lack of health insurance, and systemic racism; and
- eliminating factors that lead to delays from diagnosis to definitive surgery.
1. Wu XC, Eide MJ, King J, et al. Racial and ethnic variations in incidence and survival of cutaneous melanoma in the United States, 1999-2006. J Am Acad Dermatol. 2011;65(5 suppl 1):S26-S37. doi: 10.1016/j.jaad.2001.05.034
2. Cormier JN, Xing Y, Ding M, et al. Ethnic differences among patients with cutaneous melanoma. Arch Intern Med. 2006;166:1907-1914. doi: 10.1001/archinte.166.17.1907
3. Cress RD, Holly EA. Incidence of cutaneous melanoma among non-Hispanic whites, Hispanics, Asians, and blacks: an analysis of California cancer registry data, 1988-93. Cancer Causes Control. 1997;8:246-252. doi: 10.1023/a:1018432632528
4. Hu S, Parker DF, Thomas AG, et al. Advanced presentation of melanoma in African Americans: the Miami-Dade County experience. J Am Acad Dermatol. 2004;51:1031-1032. doi: 10.1016/ j.jaad.2004.05.005
5. Hu S, Soza-Vento RM, Parker DF, et al. Comparison of stage at diagnosis of melanoma among Hispanic, black, and white patients in Miami-Dade County, Florida. Arch Dermatol. 2006;142: 704-708. doi: 10.1001/archderm.142.6.704
6. Higgins S, Nazemi A, Feinstein S, et al. Clinical presentations of melanoma in African Americans, Hispanics, and Asians. Dermatol Surg. 2019;45:791-801. doi: 10.1097/DSS.0000000000001759
7. Dawes SM, Tsai S, Gittleman H, et al. Racial disparities in melanoma survival [published online July 28, 2016]. J Am Acad Dermatol. 2016;75:983-991. doi: 10.1016/j.jaad.2016.06.006
8. Qian Y, Johannet P, Sawyers A, et al. The ongoing racial disparities in melanoma: an analysis of the Surveillance, Epidemiology, and End Results database (1975-2016) [published online August 27, 2020]. J Am Acad Dermatol. 2021;84:1585-1593. doi: 10.1016/ j.jaad.2020.08.097
THE COMPARISON
A Acral lentiginous melanoma on the sole of the foot of a 30-year-old Black woman. The depth of the lesion was 2 mm with a positive sentinel lymph node biopsy.
B Nodular melanoma on the shoulder of a 63-year-old Hispanic woman. The depth of the lesion was 5.5 mm with a positive sentinel lymph node biopsy.
Melanoma occurs less frequently in individuals with darker skin types than in those with lighter skin types but is associated with higher rates of morbidity and mortality in this patient population.1-7 In the cases shown here (A and B), both patients had advanced melanomas with large primary lesions and lymph node metastases.

Epidemiology
A systematic review by Higgins et al6 reported the following on the epidemiology of melanomas in patients with skin of color:
- African Americans have deeper tumors at the time of diagnosis, in addition to increased rates of regionally advanced and distant disease. Lesions generally are located on the lower extremities and have an increased propensity for ulceration. Acral lentiginous melanoma is the most common melanoma subtype found in African American patients.6
- In Hispanic individuals, superficial spreading melanoma is the most common melanoma subtype. Lower extremity lesions are more common relative to White individuals. Hispanic individuals have the highest rate of oral cavity melanomas across all ethnic groups.6
- In Asian individuals, acral and subungual sites are most common. Specifically, Pacific Islanders have the highest proportion of mucosal melanomas across all ethnic groups.6
Key clinical features in people with darker skin tones
Melanomas are found more often on the palms, soles, nail units, oral cavity, and mucosae.6 The melanomas have the same clinical and dermoscopic features found in individuals with lighter skin tones.
Worth noting
Factors that may contribute to the diagnosis of more advanced melanomas in racial/ethnic minorities in the United States include:
- decreased access to health care based on lack of health insurance and low socioeconomic status,
- less awareness of the risk of melanoma among patients and health care providers because melanoma is less common in persons of color, and
- lesions found in areas less likely to be seen in screening examinations, such as the soles of the feet and the oral and genital mucosae.
Health disparity highlight
- In a large US study of 96,953 patients with a diagnosis of cutaneous melanoma from 1992 to 2009, the proportion of later-stage melanoma—stages II to IV—was greater in Black patients compared to White patients.7
- Based on this same data set, White patients had the longest survival time (P <. 05), followed by Hispanic (P < .05), Asian American/Native American/Pacific Islander (P < .05), and Black (P < .05) patients, respectively.7
- In Miami-Dade County, one study of 1690 melanoma cases found that 48% of Black patients had regional or distant disease at presentation compared to 22% of White patients (P = .015).5 Analysis of multiple factors found that only race was a significant predictor for late-stage melanoma (P < .001). Black patients in this study were 3 times more likely than others to be diagnosed with melanoma at a late stage (P = .07).5
- Black patients in the United States are more likely to have a delayed time from diagnosis to definitive surgery even when researchers controlled for type of health insurance and stage of diagnosis.8
Final thoughts
Efforts are needed to overcome these disparities by:
- educating patients with skin of color and their health care providers about the risks of advanced melanoma with the goal of prevention and earlier diagnosis;
- breaking down barriers to care caused by poverty, lack of health insurance, and systemic racism; and
- eliminating factors that lead to delays from diagnosis to definitive surgery.
THE COMPARISON
A Acral lentiginous melanoma on the sole of the foot of a 30-year-old Black woman. The depth of the lesion was 2 mm with a positive sentinel lymph node biopsy.
B Nodular melanoma on the shoulder of a 63-year-old Hispanic woman. The depth of the lesion was 5.5 mm with a positive sentinel lymph node biopsy.
Melanoma occurs less frequently in individuals with darker skin types than in those with lighter skin types but is associated with higher rates of morbidity and mortality in this patient population.1-7 In the cases shown here (A and B), both patients had advanced melanomas with large primary lesions and lymph node metastases.

Epidemiology
A systematic review by Higgins et al6 reported the following on the epidemiology of melanomas in patients with skin of color:
- African Americans have deeper tumors at the time of diagnosis, in addition to increased rates of regionally advanced and distant disease. Lesions generally are located on the lower extremities and have an increased propensity for ulceration. Acral lentiginous melanoma is the most common melanoma subtype found in African American patients.6
- In Hispanic individuals, superficial spreading melanoma is the most common melanoma subtype. Lower extremity lesions are more common relative to White individuals. Hispanic individuals have the highest rate of oral cavity melanomas across all ethnic groups.6
- In Asian individuals, acral and subungual sites are most common. Specifically, Pacific Islanders have the highest proportion of mucosal melanomas across all ethnic groups.6
Key clinical features in people with darker skin tones
Melanomas are found more often on the palms, soles, nail units, oral cavity, and mucosae.6 The melanomas have the same clinical and dermoscopic features found in individuals with lighter skin tones.
Worth noting
Factors that may contribute to the diagnosis of more advanced melanomas in racial/ethnic minorities in the United States include:
- decreased access to health care based on lack of health insurance and low socioeconomic status,
- less awareness of the risk of melanoma among patients and health care providers because melanoma is less common in persons of color, and
- lesions found in areas less likely to be seen in screening examinations, such as the soles of the feet and the oral and genital mucosae.
Health disparity highlight
- In a large US study of 96,953 patients with a diagnosis of cutaneous melanoma from 1992 to 2009, the proportion of later-stage melanoma—stages II to IV—was greater in Black patients compared to White patients.7
- Based on this same data set, White patients had the longest survival time (P <. 05), followed by Hispanic (P < .05), Asian American/Native American/Pacific Islander (P < .05), and Black (P < .05) patients, respectively.7
- In Miami-Dade County, one study of 1690 melanoma cases found that 48% of Black patients had regional or distant disease at presentation compared to 22% of White patients (P = .015).5 Analysis of multiple factors found that only race was a significant predictor for late-stage melanoma (P < .001). Black patients in this study were 3 times more likely than others to be diagnosed with melanoma at a late stage (P = .07).5
- Black patients in the United States are more likely to have a delayed time from diagnosis to definitive surgery even when researchers controlled for type of health insurance and stage of diagnosis.8
Final thoughts
Efforts are needed to overcome these disparities by:
- educating patients with skin of color and their health care providers about the risks of advanced melanoma with the goal of prevention and earlier diagnosis;
- breaking down barriers to care caused by poverty, lack of health insurance, and systemic racism; and
- eliminating factors that lead to delays from diagnosis to definitive surgery.
1. Wu XC, Eide MJ, King J, et al. Racial and ethnic variations in incidence and survival of cutaneous melanoma in the United States, 1999-2006. J Am Acad Dermatol. 2011;65(5 suppl 1):S26-S37. doi: 10.1016/j.jaad.2001.05.034
2. Cormier JN, Xing Y, Ding M, et al. Ethnic differences among patients with cutaneous melanoma. Arch Intern Med. 2006;166:1907-1914. doi: 10.1001/archinte.166.17.1907
3. Cress RD, Holly EA. Incidence of cutaneous melanoma among non-Hispanic whites, Hispanics, Asians, and blacks: an analysis of California cancer registry data, 1988-93. Cancer Causes Control. 1997;8:246-252. doi: 10.1023/a:1018432632528
4. Hu S, Parker DF, Thomas AG, et al. Advanced presentation of melanoma in African Americans: the Miami-Dade County experience. J Am Acad Dermatol. 2004;51:1031-1032. doi: 10.1016/ j.jaad.2004.05.005
5. Hu S, Soza-Vento RM, Parker DF, et al. Comparison of stage at diagnosis of melanoma among Hispanic, black, and white patients in Miami-Dade County, Florida. Arch Dermatol. 2006;142: 704-708. doi: 10.1001/archderm.142.6.704
6. Higgins S, Nazemi A, Feinstein S, et al. Clinical presentations of melanoma in African Americans, Hispanics, and Asians. Dermatol Surg. 2019;45:791-801. doi: 10.1097/DSS.0000000000001759
7. Dawes SM, Tsai S, Gittleman H, et al. Racial disparities in melanoma survival [published online July 28, 2016]. J Am Acad Dermatol. 2016;75:983-991. doi: 10.1016/j.jaad.2016.06.006
8. Qian Y, Johannet P, Sawyers A, et al. The ongoing racial disparities in melanoma: an analysis of the Surveillance, Epidemiology, and End Results database (1975-2016) [published online August 27, 2020]. J Am Acad Dermatol. 2021;84:1585-1593. doi: 10.1016/ j.jaad.2020.08.097
1. Wu XC, Eide MJ, King J, et al. Racial and ethnic variations in incidence and survival of cutaneous melanoma in the United States, 1999-2006. J Am Acad Dermatol. 2011;65(5 suppl 1):S26-S37. doi: 10.1016/j.jaad.2001.05.034
2. Cormier JN, Xing Y, Ding M, et al. Ethnic differences among patients with cutaneous melanoma. Arch Intern Med. 2006;166:1907-1914. doi: 10.1001/archinte.166.17.1907
3. Cress RD, Holly EA. Incidence of cutaneous melanoma among non-Hispanic whites, Hispanics, Asians, and blacks: an analysis of California cancer registry data, 1988-93. Cancer Causes Control. 1997;8:246-252. doi: 10.1023/a:1018432632528
4. Hu S, Parker DF, Thomas AG, et al. Advanced presentation of melanoma in African Americans: the Miami-Dade County experience. J Am Acad Dermatol. 2004;51:1031-1032. doi: 10.1016/ j.jaad.2004.05.005
5. Hu S, Soza-Vento RM, Parker DF, et al. Comparison of stage at diagnosis of melanoma among Hispanic, black, and white patients in Miami-Dade County, Florida. Arch Dermatol. 2006;142: 704-708. doi: 10.1001/archderm.142.6.704
6. Higgins S, Nazemi A, Feinstein S, et al. Clinical presentations of melanoma in African Americans, Hispanics, and Asians. Dermatol Surg. 2019;45:791-801. doi: 10.1097/DSS.0000000000001759
7. Dawes SM, Tsai S, Gittleman H, et al. Racial disparities in melanoma survival [published online July 28, 2016]. J Am Acad Dermatol. 2016;75:983-991. doi: 10.1016/j.jaad.2016.06.006
8. Qian Y, Johannet P, Sawyers A, et al. The ongoing racial disparities in melanoma: an analysis of the Surveillance, Epidemiology, and End Results database (1975-2016) [published online August 27, 2020]. J Am Acad Dermatol. 2021;84:1585-1593. doi: 10.1016/ j.jaad.2020.08.097
Steroid phobia drives weaker prescribing, nonadherence for AD
, Nanette B. Silverberg, MD, said at the Revolutionizing Atopic Dermatitis meeting.
Up to 40% of parents of children with chronic AD cite anxiety surrounding corticosteroids, according to Dr. Silverberg, chief of pediatric dermatology at the Mount Sinai Health System, New York.
When the potential for adverse events are explained to parents who are anxious about a drug, “they take it in a different way than other individuals,” noted Dr. Silverberg, clinical professor of pediatrics and dermatology at Icahn School of Medicine at Mount Sinai.
In a systematic review of 16 studies examining topical corticosteroid phobia in AD, published between 1946 and 2016, the prevalence of corticosteroid phobia among patients with AD or their caregivers ranged from 21% to 83.7%, with definitions of phobia that ranged from “concern” to “irrational fear.” In two studies where adherence was evaluated, patients with corticosteroid phobia had a higher rate of partial adherence (49.4%) or nonadherence (14.1%) when compared with patients who didn’t have a phobia of corticosteroids (29.3 % and 9.8%, respectively)..
The source of these fears can be information from friends, relatives, media, the Internet, as well as doctors, Dr. Silverberg noted. “We have to be responsible for providing proper data to these individuals,” she said.
Primary care providers also treat young children with AD differently from older children, when compared with other specialties, according to the results of one study that involved a survey and a retrospective chart review, published in 2020. In the survey, 88% of primary care providers in Chicago said they managed AD differently in children under aged 2 years than in older children, with 65% reporting they were more likely to refer a child under 2 years to a specialist, and 64% said they were less likely to prescribe high-potency topical corticosteroids to children in this age group. The retrospective review found that at PCP visits, significantly more children with AD between aged 2 and 5 years were more likely to be prescribed medium-potency topical corticosteroids (0.66% vs. 0.37%, P < .01) and high-potency topical corticosteroids (0.15% vs. 0.05%; P < .01) than children under 2 years old, respectively.
Of the children who had seen a specialist, more dermatologists (57%) prescribed medium-potency and high-potency topical corticosteroids for children under aged 2 years than did allergists (30%) and pediatricians (15%) (P < .01), according to the study.
“These are our colleagues who are often very strong prescribers using systemic agents, and only 15% of pediatricians will do this,” Dr. Silverberg said. “We’re really looking at a big divide between us and other subspecialties and primary care, and [topical corticosteroids] are frequently underutilized because of these fears.”
In another study looking at the use of topical corticosteroids for AD in the pediatric emergency department (mean age of patients, 6.3 years), from 2012 to 2017, patients at 46 of 167 visits were prescribed over-the-counter topical hydrocortisone, while at 63 of 167 visits, patients were not prescribed or recommended any corticosteroid.
The mean class of the topical corticosteroid prescribed was 5.5, and the most commonly recommended corticosteroid was class 7 (the least potent available) in 61 of 104 patients (P < .001). A dermatologist was consulted in 14 of 167 visits (8.6%), and in those cases, topical corticosteroids were often prescribed (P = .018), as was a higher class of corticosteroids (a mean of 3.1 vs. 5.9; P < .001).
Topical corticosteroids also tend to be prescribed less by internal medicine physicians than by family medicine physicians or dermatologists. A 2020 study of ambulatory care data in the United States from 2006 to 2016 found that internists were 22 times less likely to prescribe topical corticosteroids for AD compared with dermatologists (5.1% vs. 52.2%; P = .001). But there was no significant difference in prescribing between family medicine physicians and dermatologists (39.1% vs. 52.2%, P = .27).
“We know they [corticosteroids] work, but so many people are fearful of them ... even with a low, low side effect profile,” Dr. Silverberg said.
For children with AD, corticosteroid use is “suboptimal” across the United States, with evidence that Medicaid-insured pediatric patients with AD are less likely to see a specialist and less likely to be prescribed high-potency topical corticosteroids compared with commercially-insured patients.
Discussing efficacy and safety
Dr. Silverberg said providers who care for children with AD should talk about the fear surrounding these medications and educate parents with anxiety surrounding corticosteroids. “Side effects are usually short term and limited, so we really can assure parents that there is a long safety profile,” she said.
Asked to comment on this topic, Adelaide Hebert, MD, professor of dermatology and director of pediatric dermatology at the University of Texas, Houston, said that she often sees concerns surrounding the use of topical corticosteroids, both in her practice with parents and when teaching residents in other disciplines, such as pediatrics, family medicine, and emergency medicine.
“We don’t do a good job in medical school educating the students about the safety, applicability, and proper use of topical steroids, and I think that leads to some of the confusion when it comes to properly using this class of medications in treating atopic dermatitis,” she said in an interview.
The use of a high-potency topical steroid is important, she noted, as lower doses may not adequately control AD. “If the patient has very mild disease, this may be just fine,” she noted. Those patients often do not see a pediatric dermatologist, “but the ones with moderate or severe atopic dermatitis often do, and I would say [the problem of] undertreatment is all too common.”
Like Dr. Silverberg, Dr. Hebert said that in her clinical experience, side effects from topical corticosteroids have been rare. “I could count on one hand the number of patients in a 38-year pediatric dermatology practice where they had an adverse effect from a topical steroid,” she said.
Dr. Silverberg reports receiving consulting fees from Amryt Pharma, Galderma, Incyte, and Vyne; non-CME related fees from Pfizer and Regeneron; and contracted research fees from Incyte and the Vitiligo Research Foundation. Dr. Hebert reports receiving research funds from GSK, Leo, Ortho Dermatologics, Galderma, Dermavant, Pfizer, and Arcutis Biotherapeutics paid to her institution; honoraria from Pfizer, Arcutis, Incyte; and having served on the data safety monitoring board for Regeneron-Sanofi, GSK, and Ortho Dermatologics.
, Nanette B. Silverberg, MD, said at the Revolutionizing Atopic Dermatitis meeting.
Up to 40% of parents of children with chronic AD cite anxiety surrounding corticosteroids, according to Dr. Silverberg, chief of pediatric dermatology at the Mount Sinai Health System, New York.
When the potential for adverse events are explained to parents who are anxious about a drug, “they take it in a different way than other individuals,” noted Dr. Silverberg, clinical professor of pediatrics and dermatology at Icahn School of Medicine at Mount Sinai.
In a systematic review of 16 studies examining topical corticosteroid phobia in AD, published between 1946 and 2016, the prevalence of corticosteroid phobia among patients with AD or their caregivers ranged from 21% to 83.7%, with definitions of phobia that ranged from “concern” to “irrational fear.” In two studies where adherence was evaluated, patients with corticosteroid phobia had a higher rate of partial adherence (49.4%) or nonadherence (14.1%) when compared with patients who didn’t have a phobia of corticosteroids (29.3 % and 9.8%, respectively)..
The source of these fears can be information from friends, relatives, media, the Internet, as well as doctors, Dr. Silverberg noted. “We have to be responsible for providing proper data to these individuals,” she said.
Primary care providers also treat young children with AD differently from older children, when compared with other specialties, according to the results of one study that involved a survey and a retrospective chart review, published in 2020. In the survey, 88% of primary care providers in Chicago said they managed AD differently in children under aged 2 years than in older children, with 65% reporting they were more likely to refer a child under 2 years to a specialist, and 64% said they were less likely to prescribe high-potency topical corticosteroids to children in this age group. The retrospective review found that at PCP visits, significantly more children with AD between aged 2 and 5 years were more likely to be prescribed medium-potency topical corticosteroids (0.66% vs. 0.37%, P < .01) and high-potency topical corticosteroids (0.15% vs. 0.05%; P < .01) than children under 2 years old, respectively.
Of the children who had seen a specialist, more dermatologists (57%) prescribed medium-potency and high-potency topical corticosteroids for children under aged 2 years than did allergists (30%) and pediatricians (15%) (P < .01), according to the study.
“These are our colleagues who are often very strong prescribers using systemic agents, and only 15% of pediatricians will do this,” Dr. Silverberg said. “We’re really looking at a big divide between us and other subspecialties and primary care, and [topical corticosteroids] are frequently underutilized because of these fears.”
In another study looking at the use of topical corticosteroids for AD in the pediatric emergency department (mean age of patients, 6.3 years), from 2012 to 2017, patients at 46 of 167 visits were prescribed over-the-counter topical hydrocortisone, while at 63 of 167 visits, patients were not prescribed or recommended any corticosteroid.
The mean class of the topical corticosteroid prescribed was 5.5, and the most commonly recommended corticosteroid was class 7 (the least potent available) in 61 of 104 patients (P < .001). A dermatologist was consulted in 14 of 167 visits (8.6%), and in those cases, topical corticosteroids were often prescribed (P = .018), as was a higher class of corticosteroids (a mean of 3.1 vs. 5.9; P < .001).
Topical corticosteroids also tend to be prescribed less by internal medicine physicians than by family medicine physicians or dermatologists. A 2020 study of ambulatory care data in the United States from 2006 to 2016 found that internists were 22 times less likely to prescribe topical corticosteroids for AD compared with dermatologists (5.1% vs. 52.2%; P = .001). But there was no significant difference in prescribing between family medicine physicians and dermatologists (39.1% vs. 52.2%, P = .27).
“We know they [corticosteroids] work, but so many people are fearful of them ... even with a low, low side effect profile,” Dr. Silverberg said.
For children with AD, corticosteroid use is “suboptimal” across the United States, with evidence that Medicaid-insured pediatric patients with AD are less likely to see a specialist and less likely to be prescribed high-potency topical corticosteroids compared with commercially-insured patients.
Discussing efficacy and safety
Dr. Silverberg said providers who care for children with AD should talk about the fear surrounding these medications and educate parents with anxiety surrounding corticosteroids. “Side effects are usually short term and limited, so we really can assure parents that there is a long safety profile,” she said.
Asked to comment on this topic, Adelaide Hebert, MD, professor of dermatology and director of pediatric dermatology at the University of Texas, Houston, said that she often sees concerns surrounding the use of topical corticosteroids, both in her practice with parents and when teaching residents in other disciplines, such as pediatrics, family medicine, and emergency medicine.
“We don’t do a good job in medical school educating the students about the safety, applicability, and proper use of topical steroids, and I think that leads to some of the confusion when it comes to properly using this class of medications in treating atopic dermatitis,” she said in an interview.
The use of a high-potency topical steroid is important, she noted, as lower doses may not adequately control AD. “If the patient has very mild disease, this may be just fine,” she noted. Those patients often do not see a pediatric dermatologist, “but the ones with moderate or severe atopic dermatitis often do, and I would say [the problem of] undertreatment is all too common.”
Like Dr. Silverberg, Dr. Hebert said that in her clinical experience, side effects from topical corticosteroids have been rare. “I could count on one hand the number of patients in a 38-year pediatric dermatology practice where they had an adverse effect from a topical steroid,” she said.
Dr. Silverberg reports receiving consulting fees from Amryt Pharma, Galderma, Incyte, and Vyne; non-CME related fees from Pfizer and Regeneron; and contracted research fees from Incyte and the Vitiligo Research Foundation. Dr. Hebert reports receiving research funds from GSK, Leo, Ortho Dermatologics, Galderma, Dermavant, Pfizer, and Arcutis Biotherapeutics paid to her institution; honoraria from Pfizer, Arcutis, Incyte; and having served on the data safety monitoring board for Regeneron-Sanofi, GSK, and Ortho Dermatologics.
, Nanette B. Silverberg, MD, said at the Revolutionizing Atopic Dermatitis meeting.
Up to 40% of parents of children with chronic AD cite anxiety surrounding corticosteroids, according to Dr. Silverberg, chief of pediatric dermatology at the Mount Sinai Health System, New York.
When the potential for adverse events are explained to parents who are anxious about a drug, “they take it in a different way than other individuals,” noted Dr. Silverberg, clinical professor of pediatrics and dermatology at Icahn School of Medicine at Mount Sinai.
In a systematic review of 16 studies examining topical corticosteroid phobia in AD, published between 1946 and 2016, the prevalence of corticosteroid phobia among patients with AD or their caregivers ranged from 21% to 83.7%, with definitions of phobia that ranged from “concern” to “irrational fear.” In two studies where adherence was evaluated, patients with corticosteroid phobia had a higher rate of partial adherence (49.4%) or nonadherence (14.1%) when compared with patients who didn’t have a phobia of corticosteroids (29.3 % and 9.8%, respectively)..
The source of these fears can be information from friends, relatives, media, the Internet, as well as doctors, Dr. Silverberg noted. “We have to be responsible for providing proper data to these individuals,” she said.
Primary care providers also treat young children with AD differently from older children, when compared with other specialties, according to the results of one study that involved a survey and a retrospective chart review, published in 2020. In the survey, 88% of primary care providers in Chicago said they managed AD differently in children under aged 2 years than in older children, with 65% reporting they were more likely to refer a child under 2 years to a specialist, and 64% said they were less likely to prescribe high-potency topical corticosteroids to children in this age group. The retrospective review found that at PCP visits, significantly more children with AD between aged 2 and 5 years were more likely to be prescribed medium-potency topical corticosteroids (0.66% vs. 0.37%, P < .01) and high-potency topical corticosteroids (0.15% vs. 0.05%; P < .01) than children under 2 years old, respectively.
Of the children who had seen a specialist, more dermatologists (57%) prescribed medium-potency and high-potency topical corticosteroids for children under aged 2 years than did allergists (30%) and pediatricians (15%) (P < .01), according to the study.
“These are our colleagues who are often very strong prescribers using systemic agents, and only 15% of pediatricians will do this,” Dr. Silverberg said. “We’re really looking at a big divide between us and other subspecialties and primary care, and [topical corticosteroids] are frequently underutilized because of these fears.”
In another study looking at the use of topical corticosteroids for AD in the pediatric emergency department (mean age of patients, 6.3 years), from 2012 to 2017, patients at 46 of 167 visits were prescribed over-the-counter topical hydrocortisone, while at 63 of 167 visits, patients were not prescribed or recommended any corticosteroid.
The mean class of the topical corticosteroid prescribed was 5.5, and the most commonly recommended corticosteroid was class 7 (the least potent available) in 61 of 104 patients (P < .001). A dermatologist was consulted in 14 of 167 visits (8.6%), and in those cases, topical corticosteroids were often prescribed (P = .018), as was a higher class of corticosteroids (a mean of 3.1 vs. 5.9; P < .001).
Topical corticosteroids also tend to be prescribed less by internal medicine physicians than by family medicine physicians or dermatologists. A 2020 study of ambulatory care data in the United States from 2006 to 2016 found that internists were 22 times less likely to prescribe topical corticosteroids for AD compared with dermatologists (5.1% vs. 52.2%; P = .001). But there was no significant difference in prescribing between family medicine physicians and dermatologists (39.1% vs. 52.2%, P = .27).
“We know they [corticosteroids] work, but so many people are fearful of them ... even with a low, low side effect profile,” Dr. Silverberg said.
For children with AD, corticosteroid use is “suboptimal” across the United States, with evidence that Medicaid-insured pediatric patients with AD are less likely to see a specialist and less likely to be prescribed high-potency topical corticosteroids compared with commercially-insured patients.
Discussing efficacy and safety
Dr. Silverberg said providers who care for children with AD should talk about the fear surrounding these medications and educate parents with anxiety surrounding corticosteroids. “Side effects are usually short term and limited, so we really can assure parents that there is a long safety profile,” she said.
Asked to comment on this topic, Adelaide Hebert, MD, professor of dermatology and director of pediatric dermatology at the University of Texas, Houston, said that she often sees concerns surrounding the use of topical corticosteroids, both in her practice with parents and when teaching residents in other disciplines, such as pediatrics, family medicine, and emergency medicine.
“We don’t do a good job in medical school educating the students about the safety, applicability, and proper use of topical steroids, and I think that leads to some of the confusion when it comes to properly using this class of medications in treating atopic dermatitis,” she said in an interview.
The use of a high-potency topical steroid is important, she noted, as lower doses may not adequately control AD. “If the patient has very mild disease, this may be just fine,” she noted. Those patients often do not see a pediatric dermatologist, “but the ones with moderate or severe atopic dermatitis often do, and I would say [the problem of] undertreatment is all too common.”
Like Dr. Silverberg, Dr. Hebert said that in her clinical experience, side effects from topical corticosteroids have been rare. “I could count on one hand the number of patients in a 38-year pediatric dermatology practice where they had an adverse effect from a topical steroid,” she said.
Dr. Silverberg reports receiving consulting fees from Amryt Pharma, Galderma, Incyte, and Vyne; non-CME related fees from Pfizer and Regeneron; and contracted research fees from Incyte and the Vitiligo Research Foundation. Dr. Hebert reports receiving research funds from GSK, Leo, Ortho Dermatologics, Galderma, Dermavant, Pfizer, and Arcutis Biotherapeutics paid to her institution; honoraria from Pfizer, Arcutis, Incyte; and having served on the data safety monitoring board for Regeneron-Sanofi, GSK, and Ortho Dermatologics.
FROM RAD 2022
Pick your sunscreen carefully: 75% don’t pass muster
Just in time for Memorial Day outings, a new report on sunscreens is out.
The news isn’t all sunny. , a nonprofit research and advocacy group that just issued its 16th annual Guide to Sunscreens.
In response, dermatologists, including the president of the American Academy of Dermatology, say that although some concerns have been raised about the safety of some sunscreen ingredients, sunscreens themselves remain an important tool in the fight against skin cancer. According to the Skin Cancer Foundation, 1 in 5 Americans will get skin cancer by age 70. Melanoma, the most deadly, has a 5-year survival rate of 99% if caught early.
2022 report
Overall, the Environmental Working Group found that about 1 in 4 sunscreens, or about 500 products, met their standards for providing adequate sun protection and avoiding ingredients linked to known health harms. Products meant for babies and children did slightly better, with about 1 in 3 meeting the standards. The group evaluated mineral sunscreens, also called physical sunscreens, and non-mineral sunscreens, also called chemical sunscreens. Mineral sunscreens contain zinc oxide or titanium dioxide and sit on the skin to deflect the sun’s rays. Chemical sunscreens, with ingredients such as oxybenzone or avobenzone, are partially absorbed into the skin.
Among the group’s concerns:
- The use of oxybenzone in the non-mineral sunscreens. About 30% of the non-mineral sunscreens have it, says Carla Burns, senior director for cosmetic science for the Environmental Working Group. Oxybenzone is a potential hormone disrupter and a skin sensitizer that may harm children and adults, she says. Some progress has been made, as the group found oxybenzone in 66% of the non-mineral sunscreens it reviewed in 2019. (The FDA is seeking more information on oxybenzone and many other sunscreen ingredients.)
- Contamination of sunscreens with benzene, which has been linked to leukemia and other blood disorders, according to the National Cancer Institute. But industry experts stress that that chemical is found in trace amounts in personal care products and does not pose a safety concern. “Benzene is a chemical that is ubiquitous in the environment and not an intentionally added ingredient in personal care products. People worldwide are exposed daily to benzene from indoor and outdoor sources, including air, drinking water, and food and beverages,” the Personal Care Products Council, an industry group, said in a statement.
- Protection from ultraviolet A (UVA) rays is often inadequate, according to research published last year by the Environmental Working Group.
Products on the ‘best’ list
The Environmental Working Group found that 282 recreational sunscreens met its criteria. Among them:
- Coral Safe Sunscreen Lotion, SPF 30
- Neutrogena Sheer Zinc Mineral Sunscreen Lotion, SPF 30
- Mad Hippie Facial Sunscreen Lotion, SPF 30+
The group chose 86 non-mineral sunscreens as better options, including:
- Alba Botanica Hawaiian Sunscreen Lotion, Aloe Vera, SPF 30
- Banana Boat Sport Ultra Sunscreen Stick, SPF 50+
- Black Girl Sunscreen Melanin Boosting Moisturizing Sunscreen Lotion, SPF 30
And 70 sunscreens made the kids’ best list, including:
- True Baby Everyday Play Sunscreen Lotion, SPF 30+
- Sun Biologic Kids’ Sunscreen Stick, SPF 30+
- Kiss My Face Organic Kids’ Defense Sunscreen Lotion, SPF 30
Industry response, FDA actions
In a statement, Alexandra Kowcz, chief scientist at the Personal Care Products Council, pointed out that “as part of a daily safe-sun regimen, sunscreen products help prevent sunburn and reduce skin cancer risk. It is unfortunate that as Americans spend more time outdoors, the Environmental Working Group’s (EWG) 2022 Guide to Sunscreens resorts to fear-mongering with misleading information that could keep consumers from using sunscreens altogether.”
The FDA has asked for more information about certain ingredients to further evaluate products, she says, and industry is working with the agency. The FDA says it is attempting to improve the quality, safety and effectiveness of over-the-counter sunscreen products. In September, 2021, the FDA issued a proposal for regulating OTC sunscreen products, as required under the CARES (Coronavirus Aid, Relief and Economic Security) Act. The effective date for the final order can’t be earlier than September 2022, the CARES Act says.
Dermatologists weigh in
“Every time something like this gets published, my patients come in hysterical,” says Michele Green, MD, a New York City dermatologist who reviewed the report for WebMD. She acknowledges that more research is needed on some sunscreen ingredients. “We really do not know the long-term consequence of oxybenzone,” she says.
Her advice: If her patients have melasma (a skin condition with brown patches on the face), she advises them to use both a chemical and a mineral sunscreen. “I don’t tell my patients in general not to use the chemical [sunscreens].”
For children, she says, the mineral sunscreens may be preferred. On her own children, who are teens, she uses the mineral sunscreens, due to possible concern about hormone disruption.
In a statement, Mark D. Kaufmann, MD, president of the American Academy of Dermatology, says that “sunscreen is an important part of a comprehensive sun protection strategy.”
Besides a broad-spectrum, water-resistant sunscreen with an SPF of 30 or higher for exposed skin, the academy recommends seeking shade and wearing sun-protective clothing to reduce skin cancer risk.
A version of this article first appeared on WebMD.com.
Just in time for Memorial Day outings, a new report on sunscreens is out.
The news isn’t all sunny. , a nonprofit research and advocacy group that just issued its 16th annual Guide to Sunscreens.
In response, dermatologists, including the president of the American Academy of Dermatology, say that although some concerns have been raised about the safety of some sunscreen ingredients, sunscreens themselves remain an important tool in the fight against skin cancer. According to the Skin Cancer Foundation, 1 in 5 Americans will get skin cancer by age 70. Melanoma, the most deadly, has a 5-year survival rate of 99% if caught early.
2022 report
Overall, the Environmental Working Group found that about 1 in 4 sunscreens, or about 500 products, met their standards for providing adequate sun protection and avoiding ingredients linked to known health harms. Products meant for babies and children did slightly better, with about 1 in 3 meeting the standards. The group evaluated mineral sunscreens, also called physical sunscreens, and non-mineral sunscreens, also called chemical sunscreens. Mineral sunscreens contain zinc oxide or titanium dioxide and sit on the skin to deflect the sun’s rays. Chemical sunscreens, with ingredients such as oxybenzone or avobenzone, are partially absorbed into the skin.
Among the group’s concerns:
- The use of oxybenzone in the non-mineral sunscreens. About 30% of the non-mineral sunscreens have it, says Carla Burns, senior director for cosmetic science for the Environmental Working Group. Oxybenzone is a potential hormone disrupter and a skin sensitizer that may harm children and adults, she says. Some progress has been made, as the group found oxybenzone in 66% of the non-mineral sunscreens it reviewed in 2019. (The FDA is seeking more information on oxybenzone and many other sunscreen ingredients.)
- Contamination of sunscreens with benzene, which has been linked to leukemia and other blood disorders, according to the National Cancer Institute. But industry experts stress that that chemical is found in trace amounts in personal care products and does not pose a safety concern. “Benzene is a chemical that is ubiquitous in the environment and not an intentionally added ingredient in personal care products. People worldwide are exposed daily to benzene from indoor and outdoor sources, including air, drinking water, and food and beverages,” the Personal Care Products Council, an industry group, said in a statement.
- Protection from ultraviolet A (UVA) rays is often inadequate, according to research published last year by the Environmental Working Group.
Products on the ‘best’ list
The Environmental Working Group found that 282 recreational sunscreens met its criteria. Among them:
- Coral Safe Sunscreen Lotion, SPF 30
- Neutrogena Sheer Zinc Mineral Sunscreen Lotion, SPF 30
- Mad Hippie Facial Sunscreen Lotion, SPF 30+
The group chose 86 non-mineral sunscreens as better options, including:
- Alba Botanica Hawaiian Sunscreen Lotion, Aloe Vera, SPF 30
- Banana Boat Sport Ultra Sunscreen Stick, SPF 50+
- Black Girl Sunscreen Melanin Boosting Moisturizing Sunscreen Lotion, SPF 30
And 70 sunscreens made the kids’ best list, including:
- True Baby Everyday Play Sunscreen Lotion, SPF 30+
- Sun Biologic Kids’ Sunscreen Stick, SPF 30+
- Kiss My Face Organic Kids’ Defense Sunscreen Lotion, SPF 30
Industry response, FDA actions
In a statement, Alexandra Kowcz, chief scientist at the Personal Care Products Council, pointed out that “as part of a daily safe-sun regimen, sunscreen products help prevent sunburn and reduce skin cancer risk. It is unfortunate that as Americans spend more time outdoors, the Environmental Working Group’s (EWG) 2022 Guide to Sunscreens resorts to fear-mongering with misleading information that could keep consumers from using sunscreens altogether.”
The FDA has asked for more information about certain ingredients to further evaluate products, she says, and industry is working with the agency. The FDA says it is attempting to improve the quality, safety and effectiveness of over-the-counter sunscreen products. In September, 2021, the FDA issued a proposal for regulating OTC sunscreen products, as required under the CARES (Coronavirus Aid, Relief and Economic Security) Act. The effective date for the final order can’t be earlier than September 2022, the CARES Act says.
Dermatologists weigh in
“Every time something like this gets published, my patients come in hysterical,” says Michele Green, MD, a New York City dermatologist who reviewed the report for WebMD. She acknowledges that more research is needed on some sunscreen ingredients. “We really do not know the long-term consequence of oxybenzone,” she says.
Her advice: If her patients have melasma (a skin condition with brown patches on the face), she advises them to use both a chemical and a mineral sunscreen. “I don’t tell my patients in general not to use the chemical [sunscreens].”
For children, she says, the mineral sunscreens may be preferred. On her own children, who are teens, she uses the mineral sunscreens, due to possible concern about hormone disruption.
In a statement, Mark D. Kaufmann, MD, president of the American Academy of Dermatology, says that “sunscreen is an important part of a comprehensive sun protection strategy.”
Besides a broad-spectrum, water-resistant sunscreen with an SPF of 30 or higher for exposed skin, the academy recommends seeking shade and wearing sun-protective clothing to reduce skin cancer risk.
A version of this article first appeared on WebMD.com.
Just in time for Memorial Day outings, a new report on sunscreens is out.
The news isn’t all sunny. , a nonprofit research and advocacy group that just issued its 16th annual Guide to Sunscreens.
In response, dermatologists, including the president of the American Academy of Dermatology, say that although some concerns have been raised about the safety of some sunscreen ingredients, sunscreens themselves remain an important tool in the fight against skin cancer. According to the Skin Cancer Foundation, 1 in 5 Americans will get skin cancer by age 70. Melanoma, the most deadly, has a 5-year survival rate of 99% if caught early.
2022 report
Overall, the Environmental Working Group found that about 1 in 4 sunscreens, or about 500 products, met their standards for providing adequate sun protection and avoiding ingredients linked to known health harms. Products meant for babies and children did slightly better, with about 1 in 3 meeting the standards. The group evaluated mineral sunscreens, also called physical sunscreens, and non-mineral sunscreens, also called chemical sunscreens. Mineral sunscreens contain zinc oxide or titanium dioxide and sit on the skin to deflect the sun’s rays. Chemical sunscreens, with ingredients such as oxybenzone or avobenzone, are partially absorbed into the skin.
Among the group’s concerns:
- The use of oxybenzone in the non-mineral sunscreens. About 30% of the non-mineral sunscreens have it, says Carla Burns, senior director for cosmetic science for the Environmental Working Group. Oxybenzone is a potential hormone disrupter and a skin sensitizer that may harm children and adults, she says. Some progress has been made, as the group found oxybenzone in 66% of the non-mineral sunscreens it reviewed in 2019. (The FDA is seeking more information on oxybenzone and many other sunscreen ingredients.)
- Contamination of sunscreens with benzene, which has been linked to leukemia and other blood disorders, according to the National Cancer Institute. But industry experts stress that that chemical is found in trace amounts in personal care products and does not pose a safety concern. “Benzene is a chemical that is ubiquitous in the environment and not an intentionally added ingredient in personal care products. People worldwide are exposed daily to benzene from indoor and outdoor sources, including air, drinking water, and food and beverages,” the Personal Care Products Council, an industry group, said in a statement.
- Protection from ultraviolet A (UVA) rays is often inadequate, according to research published last year by the Environmental Working Group.
Products on the ‘best’ list
The Environmental Working Group found that 282 recreational sunscreens met its criteria. Among them:
- Coral Safe Sunscreen Lotion, SPF 30
- Neutrogena Sheer Zinc Mineral Sunscreen Lotion, SPF 30
- Mad Hippie Facial Sunscreen Lotion, SPF 30+
The group chose 86 non-mineral sunscreens as better options, including:
- Alba Botanica Hawaiian Sunscreen Lotion, Aloe Vera, SPF 30
- Banana Boat Sport Ultra Sunscreen Stick, SPF 50+
- Black Girl Sunscreen Melanin Boosting Moisturizing Sunscreen Lotion, SPF 30
And 70 sunscreens made the kids’ best list, including:
- True Baby Everyday Play Sunscreen Lotion, SPF 30+
- Sun Biologic Kids’ Sunscreen Stick, SPF 30+
- Kiss My Face Organic Kids’ Defense Sunscreen Lotion, SPF 30
Industry response, FDA actions
In a statement, Alexandra Kowcz, chief scientist at the Personal Care Products Council, pointed out that “as part of a daily safe-sun regimen, sunscreen products help prevent sunburn and reduce skin cancer risk. It is unfortunate that as Americans spend more time outdoors, the Environmental Working Group’s (EWG) 2022 Guide to Sunscreens resorts to fear-mongering with misleading information that could keep consumers from using sunscreens altogether.”
The FDA has asked for more information about certain ingredients to further evaluate products, she says, and industry is working with the agency. The FDA says it is attempting to improve the quality, safety and effectiveness of over-the-counter sunscreen products. In September, 2021, the FDA issued a proposal for regulating OTC sunscreen products, as required under the CARES (Coronavirus Aid, Relief and Economic Security) Act. The effective date for the final order can’t be earlier than September 2022, the CARES Act says.
Dermatologists weigh in
“Every time something like this gets published, my patients come in hysterical,” says Michele Green, MD, a New York City dermatologist who reviewed the report for WebMD. She acknowledges that more research is needed on some sunscreen ingredients. “We really do not know the long-term consequence of oxybenzone,” she says.
Her advice: If her patients have melasma (a skin condition with brown patches on the face), she advises them to use both a chemical and a mineral sunscreen. “I don’t tell my patients in general not to use the chemical [sunscreens].”
For children, she says, the mineral sunscreens may be preferred. On her own children, who are teens, she uses the mineral sunscreens, due to possible concern about hormone disruption.
In a statement, Mark D. Kaufmann, MD, president of the American Academy of Dermatology, says that “sunscreen is an important part of a comprehensive sun protection strategy.”
Besides a broad-spectrum, water-resistant sunscreen with an SPF of 30 or higher for exposed skin, the academy recommends seeking shade and wearing sun-protective clothing to reduce skin cancer risk.
A version of this article first appeared on WebMD.com.
Restoring dignity to sex trafficking survivors, one tattoo removal at a time
SAN DIEGO – , according to the results of an online survey evaluating the need for and impact of tattoo removal in this population.
Sex trafficking involves the use of force, fraud, or coercion to compel another person to engage in commercial sex acts, and traffickers often brand their victims with tattoos that convey ownership, including tattoos of names, symbols, and barcodes. According to data from Polaris, a nonprofit organization that works to combat and prevent sex and labor trafficking in the United States, 16,658 sex trafficking victims were identified in the country in 2020, but tens of thousands go unreported.
“Given the inherently covert nature of this crime, it is difficult to determine exact statistics,” Emily L. Guo, MD, a cosmetic dermatologic surgery fellow at the Dermatology and Laser Surgery Center in Houston, said during a clinical abstract session at the annual meeting of the American Society for Laser Medicine and Surgery. “We have been working with sex trafficking survivors local to our practice in Houston providing pro bono tattoo removal, and we’ve observed how impactful that is in their recovery. We wanted to see if there was a national need for support of these survivors, allowing them to reclaim their lives.”
In collaboration with Elizabeth Kream, MD, a dermatology resident at the University of Illinois at Chicago, and Paul M. Friedman, MD, director of the Dermatology and Laser Surgery Center and the current ASLMS president, Dr. Guo conducted an online needs and impact survey regarding laser removal of branding tattoos. With assistance from the National Trafficking Sheltered Alliance, the researchers distributed the survey to U.S. organizations that support sex trafficking survivors. Representatives from 40 organizations responded to the survey. Most were based in the South (45%), followed by the West (20%) and Midwest (20%), and the Northeast (15%).
“On average, these programs support 81 survivors per year, which translates into 3,240 victims per year,” Dr. Guo said. Survey respondents estimated that 47% of sex trafficking survivors had branding tattoos. Of those, 67% were in a stable situation that would make it possible to undergo tattoo removal.
On a scale of 1 to 10 with 10 being the highest, “pro bono removal of branding tattoos received a survivor impact recovery score of 9.2 by these respondents,” Dr. Guo said. “Breaking down these numbers, there are at least 1,200 survivors per year who would benefit from tattoo removal during recovery. Qualitative responses to our survey echoed the same messages: There is a great need and a large impact for pro bono tattoo removal.”
For example, one survey respondent wrote, “Thank you for being willing to remove tattoos, allowing them to feel as though they are no longer owned by their trafficker.” Another wrote, “Erasing or revising the mark of her trafficker is a critical part of every survivor’s recovery journey.”
Sometimes branding tattoos are placed in highly visible locations. One sex trafficking survivor presented to Dr. Guo with a large dark blue tattoo above an eyebrow. “She shared with me that because the tattoo was so highly visible, nobody would offer her a job,” Dr. Guo said. Another survivor had her trafficker’s initial tattooed on her left ring finger. Yet another had a large tattoo on her forearm branded with her trafficker’s name as well as the word cash, “indicating that she is source of money for him,” she said, noting that on average, one sex trafficking victim generates about $100,000 per year for their trafficker.
Although there has been work published on recognition of branding tattoos in the medical community, including the difficulty in differentiating branding tattoos from voluntary tattoos, Dr. Friedman said that there have not been any studies evaluating the need and impact of laser branding tattoo removal in the recovery of sex trafficking survivors. Findings from the current survey “illuminate that the removal of branding tattoos is highly impactful on recovery and may be preferred over tattoo cover-ups,” Dr. Friedman told this news organization.
“Furthermore, survivors frequently move during their recovery process, so a national partnership is essential to allowing survivors to continue the removal process wherever they may be.”
The findings support a proposed ASLMS campaign that intends to connect sex trafficking survivors with board-certified physicians for pro bono removal of branding tattoos. “This will not only aid in survivors’ recovery, but this work will also be beneficial to allow for an avenue to create a repository of sex trafficking tattoo images to improve branding tattoo identification competency among health care providers,” Dr. Friedman said.
He acknowledged certain limitations of the survey, including the fact that “thorough and exact data collection regarding human trafficking is challenging given the inherently covert and underground nature of this crime.” In addition, the study involved surveying organizations supporting sex trafficking survivors rather than the survivors themselves. However, he noted, “we felt for this initial study we wanted to be sensitive to the survivors.”
In an interview at the meeting, one of the session moderators, Oge Onwudiwe, MD, a dermatologist who practices at AllPhases Dermatology in Alexandria, Va., said that pro bono laser removal of branding tattoos “is something that a lot of us can work on and do, and have an impact on. There’s no reason why we shouldn’t help. I can only imagine the psychological impact of having a daily reminder of that [in the form of a branding tattoo]. That’s like PTSD every day almost. You have a trigger there.”
Another session moderator, Eliot Battle, MD, CEO of Cultura Dermatology and Laser Center in Washington, is a board member of Innocents at Risk, a nonprofit that works to fight child exploitation and human trafficking. With pro bono laser removal of a branded tattoo, “this is not just a cosmetic correction you’re making,” Dr. Battle said. “It’s so much deeper than that. It changes people’s lives.”
The researchers and Dr. Onwudiwe reported having no financial disclosures. Dr. Battle disclosed that he conducts research for Cynosure, and has received discounts from Cynosure, Cutera, Solta Medical, Lumenis, Be Inc., and Sciton.
SAN DIEGO – , according to the results of an online survey evaluating the need for and impact of tattoo removal in this population.
Sex trafficking involves the use of force, fraud, or coercion to compel another person to engage in commercial sex acts, and traffickers often brand their victims with tattoos that convey ownership, including tattoos of names, symbols, and barcodes. According to data from Polaris, a nonprofit organization that works to combat and prevent sex and labor trafficking in the United States, 16,658 sex trafficking victims were identified in the country in 2020, but tens of thousands go unreported.
“Given the inherently covert nature of this crime, it is difficult to determine exact statistics,” Emily L. Guo, MD, a cosmetic dermatologic surgery fellow at the Dermatology and Laser Surgery Center in Houston, said during a clinical abstract session at the annual meeting of the American Society for Laser Medicine and Surgery. “We have been working with sex trafficking survivors local to our practice in Houston providing pro bono tattoo removal, and we’ve observed how impactful that is in their recovery. We wanted to see if there was a national need for support of these survivors, allowing them to reclaim their lives.”
In collaboration with Elizabeth Kream, MD, a dermatology resident at the University of Illinois at Chicago, and Paul M. Friedman, MD, director of the Dermatology and Laser Surgery Center and the current ASLMS president, Dr. Guo conducted an online needs and impact survey regarding laser removal of branding tattoos. With assistance from the National Trafficking Sheltered Alliance, the researchers distributed the survey to U.S. organizations that support sex trafficking survivors. Representatives from 40 organizations responded to the survey. Most were based in the South (45%), followed by the West (20%) and Midwest (20%), and the Northeast (15%).
“On average, these programs support 81 survivors per year, which translates into 3,240 victims per year,” Dr. Guo said. Survey respondents estimated that 47% of sex trafficking survivors had branding tattoos. Of those, 67% were in a stable situation that would make it possible to undergo tattoo removal.
On a scale of 1 to 10 with 10 being the highest, “pro bono removal of branding tattoos received a survivor impact recovery score of 9.2 by these respondents,” Dr. Guo said. “Breaking down these numbers, there are at least 1,200 survivors per year who would benefit from tattoo removal during recovery. Qualitative responses to our survey echoed the same messages: There is a great need and a large impact for pro bono tattoo removal.”
For example, one survey respondent wrote, “Thank you for being willing to remove tattoos, allowing them to feel as though they are no longer owned by their trafficker.” Another wrote, “Erasing or revising the mark of her trafficker is a critical part of every survivor’s recovery journey.”
Sometimes branding tattoos are placed in highly visible locations. One sex trafficking survivor presented to Dr. Guo with a large dark blue tattoo above an eyebrow. “She shared with me that because the tattoo was so highly visible, nobody would offer her a job,” Dr. Guo said. Another survivor had her trafficker’s initial tattooed on her left ring finger. Yet another had a large tattoo on her forearm branded with her trafficker’s name as well as the word cash, “indicating that she is source of money for him,” she said, noting that on average, one sex trafficking victim generates about $100,000 per year for their trafficker.
Although there has been work published on recognition of branding tattoos in the medical community, including the difficulty in differentiating branding tattoos from voluntary tattoos, Dr. Friedman said that there have not been any studies evaluating the need and impact of laser branding tattoo removal in the recovery of sex trafficking survivors. Findings from the current survey “illuminate that the removal of branding tattoos is highly impactful on recovery and may be preferred over tattoo cover-ups,” Dr. Friedman told this news organization.
“Furthermore, survivors frequently move during their recovery process, so a national partnership is essential to allowing survivors to continue the removal process wherever they may be.”
The findings support a proposed ASLMS campaign that intends to connect sex trafficking survivors with board-certified physicians for pro bono removal of branding tattoos. “This will not only aid in survivors’ recovery, but this work will also be beneficial to allow for an avenue to create a repository of sex trafficking tattoo images to improve branding tattoo identification competency among health care providers,” Dr. Friedman said.
He acknowledged certain limitations of the survey, including the fact that “thorough and exact data collection regarding human trafficking is challenging given the inherently covert and underground nature of this crime.” In addition, the study involved surveying organizations supporting sex trafficking survivors rather than the survivors themselves. However, he noted, “we felt for this initial study we wanted to be sensitive to the survivors.”
In an interview at the meeting, one of the session moderators, Oge Onwudiwe, MD, a dermatologist who practices at AllPhases Dermatology in Alexandria, Va., said that pro bono laser removal of branding tattoos “is something that a lot of us can work on and do, and have an impact on. There’s no reason why we shouldn’t help. I can only imagine the psychological impact of having a daily reminder of that [in the form of a branding tattoo]. That’s like PTSD every day almost. You have a trigger there.”
Another session moderator, Eliot Battle, MD, CEO of Cultura Dermatology and Laser Center in Washington, is a board member of Innocents at Risk, a nonprofit that works to fight child exploitation and human trafficking. With pro bono laser removal of a branded tattoo, “this is not just a cosmetic correction you’re making,” Dr. Battle said. “It’s so much deeper than that. It changes people’s lives.”
The researchers and Dr. Onwudiwe reported having no financial disclosures. Dr. Battle disclosed that he conducts research for Cynosure, and has received discounts from Cynosure, Cutera, Solta Medical, Lumenis, Be Inc., and Sciton.
SAN DIEGO – , according to the results of an online survey evaluating the need for and impact of tattoo removal in this population.
Sex trafficking involves the use of force, fraud, or coercion to compel another person to engage in commercial sex acts, and traffickers often brand their victims with tattoos that convey ownership, including tattoos of names, symbols, and barcodes. According to data from Polaris, a nonprofit organization that works to combat and prevent sex and labor trafficking in the United States, 16,658 sex trafficking victims were identified in the country in 2020, but tens of thousands go unreported.
“Given the inherently covert nature of this crime, it is difficult to determine exact statistics,” Emily L. Guo, MD, a cosmetic dermatologic surgery fellow at the Dermatology and Laser Surgery Center in Houston, said during a clinical abstract session at the annual meeting of the American Society for Laser Medicine and Surgery. “We have been working with sex trafficking survivors local to our practice in Houston providing pro bono tattoo removal, and we’ve observed how impactful that is in their recovery. We wanted to see if there was a national need for support of these survivors, allowing them to reclaim their lives.”
In collaboration with Elizabeth Kream, MD, a dermatology resident at the University of Illinois at Chicago, and Paul M. Friedman, MD, director of the Dermatology and Laser Surgery Center and the current ASLMS president, Dr. Guo conducted an online needs and impact survey regarding laser removal of branding tattoos. With assistance from the National Trafficking Sheltered Alliance, the researchers distributed the survey to U.S. organizations that support sex trafficking survivors. Representatives from 40 organizations responded to the survey. Most were based in the South (45%), followed by the West (20%) and Midwest (20%), and the Northeast (15%).
“On average, these programs support 81 survivors per year, which translates into 3,240 victims per year,” Dr. Guo said. Survey respondents estimated that 47% of sex trafficking survivors had branding tattoos. Of those, 67% were in a stable situation that would make it possible to undergo tattoo removal.
On a scale of 1 to 10 with 10 being the highest, “pro bono removal of branding tattoos received a survivor impact recovery score of 9.2 by these respondents,” Dr. Guo said. “Breaking down these numbers, there are at least 1,200 survivors per year who would benefit from tattoo removal during recovery. Qualitative responses to our survey echoed the same messages: There is a great need and a large impact for pro bono tattoo removal.”
For example, one survey respondent wrote, “Thank you for being willing to remove tattoos, allowing them to feel as though they are no longer owned by their trafficker.” Another wrote, “Erasing or revising the mark of her trafficker is a critical part of every survivor’s recovery journey.”
Sometimes branding tattoos are placed in highly visible locations. One sex trafficking survivor presented to Dr. Guo with a large dark blue tattoo above an eyebrow. “She shared with me that because the tattoo was so highly visible, nobody would offer her a job,” Dr. Guo said. Another survivor had her trafficker’s initial tattooed on her left ring finger. Yet another had a large tattoo on her forearm branded with her trafficker’s name as well as the word cash, “indicating that she is source of money for him,” she said, noting that on average, one sex trafficking victim generates about $100,000 per year for their trafficker.
Although there has been work published on recognition of branding tattoos in the medical community, including the difficulty in differentiating branding tattoos from voluntary tattoos, Dr. Friedman said that there have not been any studies evaluating the need and impact of laser branding tattoo removal in the recovery of sex trafficking survivors. Findings from the current survey “illuminate that the removal of branding tattoos is highly impactful on recovery and may be preferred over tattoo cover-ups,” Dr. Friedman told this news organization.
“Furthermore, survivors frequently move during their recovery process, so a national partnership is essential to allowing survivors to continue the removal process wherever they may be.”
The findings support a proposed ASLMS campaign that intends to connect sex trafficking survivors with board-certified physicians for pro bono removal of branding tattoos. “This will not only aid in survivors’ recovery, but this work will also be beneficial to allow for an avenue to create a repository of sex trafficking tattoo images to improve branding tattoo identification competency among health care providers,” Dr. Friedman said.
He acknowledged certain limitations of the survey, including the fact that “thorough and exact data collection regarding human trafficking is challenging given the inherently covert and underground nature of this crime.” In addition, the study involved surveying organizations supporting sex trafficking survivors rather than the survivors themselves. However, he noted, “we felt for this initial study we wanted to be sensitive to the survivors.”
In an interview at the meeting, one of the session moderators, Oge Onwudiwe, MD, a dermatologist who practices at AllPhases Dermatology in Alexandria, Va., said that pro bono laser removal of branding tattoos “is something that a lot of us can work on and do, and have an impact on. There’s no reason why we shouldn’t help. I can only imagine the psychological impact of having a daily reminder of that [in the form of a branding tattoo]. That’s like PTSD every day almost. You have a trigger there.”
Another session moderator, Eliot Battle, MD, CEO of Cultura Dermatology and Laser Center in Washington, is a board member of Innocents at Risk, a nonprofit that works to fight child exploitation and human trafficking. With pro bono laser removal of a branded tattoo, “this is not just a cosmetic correction you’re making,” Dr. Battle said. “It’s so much deeper than that. It changes people’s lives.”
The researchers and Dr. Onwudiwe reported having no financial disclosures. Dr. Battle disclosed that he conducts research for Cynosure, and has received discounts from Cynosure, Cutera, Solta Medical, Lumenis, Be Inc., and Sciton.
AT ASLMS 2022
Q&A with Hubert (Hugh) Greenway, MD
who was also recently selected as program director for cutaneous oncology at Scripps MD Anderson Cancer Center in San Diego. He is also a former president of the American College of Mohs Surgery.
After earning his medical degree from the Medical College of Georgia, Augusta, in 1974, Dr. Greenway was fellowship trained in Mohs skin cancer surgery by Frederic E. Mohs, MD, at the University of Wisconsin–Madison. He completed his dermatology residency at the Naval Medical Center San Diego and joined Scripps Clinic in 1983, where he launched the institution’s first Mohs surgery program, as well as a popular annual intensive course in superficial anatomy and cutaneous surgery that bears his name. He was also the first physician in the world to use interferon as a nonsurgical treatment of basal cell carcinoma.

To date, Dr. Greenway has performed more than 41,000 Mohs surgery cases and has trained 61 fellows who practice in academic and clinical settings. In 2017, he received the Frederic E. Mohs Award from the ACMS at the college’s annual meeting. He is also a past CEO of Scripps Clinic. In this Q&A, Dr. Greenway opens up about what it was like to train with Dr. Mohs, what makes a good Mohs surgeon, and why he’s excited about the future of dermatology.
I understand that you first became interested in a medical career after meeting Dr. Carl Jones, a friend of your father who was your Scoutmaster in the Boy Scouts in Georgia. What about Dr. Jones inspired you to pursue a career in medicine?
Dr. Jones was an internist/allergist in Atlanta, where I grew up. His three sons and I were friends. My dad had dealt with several medical problems being injured in World War II and subsequently undergoing a couple of kidney transplantations, so I developed an interest in medicine personally. Even though Dr. Jones was a specialist, he started out as a family doctor like I did, so he was interested in the whole person and all of his or her medical problems as opposed to those related to his specialty only. I traveled with the Boy Scouts to camp at places like Valley Forge in Pennsylvania, and Dr. Jones was involved with the medical set-ups of those large events. That also contributed to my interest in medicine.
As part of your 9-year service in the U.S. Navy, you spent 2 years as the flight surgeon at NAS Atlanta/Dobbins Air Force Base. What was your most memorable experience from that assignment?
Dobbins is a large facility with two Lockheed plants, and the Air Force had built the medical clinic, which was staffed by the Navy. Getting to know some of the active-duty members of the Air Force, the Navy, and the National Guard, and their commitment to our country, was memorable. Jimmy Carter was the president in those days. When he would fly in Dobbins, one of my jobs as the flight surgeon was to be on base when Air Force One landed or departed. One night, we had a DC-9 commercial aircraft coming from Huntsville, Ala., to Atlanta that got caught in a thunderstorm a little above 30,000 feet. Both engines went out and the aircraft essentially became a glider. The pilots tried to land on our runway but unfortunately, they ended up 4 miles short. We were heavily involved in responding to the crash, which was a tragic event. I also learned to fly (second seat) different types of aircraft during my assignment at NAS Atlanta/Dobbins Air Force Base, everything from the large C-5s to Navy fighter jets and helicopters. Coincidentally, Dr. Jones was involved with a couple of free health clinics in Atlanta when I was stationed there. Every Tuesday night, my wife (who is a nurse) and I would volunteer at a clinic in Cabbagetown, which was one of the poorer areas of Atlanta. It was a chance to give back to a group of people who didn’t have a whole lot.
In the middle your dermatology residency at Naval Medical Center San Diego, you were selected by Dr. Mohs for fellowship training in Mohs skin cancer surgery at the University of Wisconsin–Madison. What do you remember most about your training with Dr. Mohs?
Dr. Mohs was a kind, humble man who had this great idea about skin cancer. He was not a dermatologist; he was a general surgeon. The technique he developed was originally called chemosurgery because he put a chemical onto the skin. This was known as the fixed-tissue technique. Then we had a fresh-tissue technique, where we did not use the chemical, but we were able to use local anesthesia right away. That developed into the Mohs surgery we know today. Dr. Mohs did not name it that; he was very humble, but he was very proud of his technique. He was also a very hard worker. On the first day of my fellowship, I started at 7 in the morning and ended at 7 at night. It was the same for the last day of my fellowship. He also had an excellent office staff, many of whom had worked with him for many years. Patients with difficult skin cancers traveled to Madison from all over the world because there weren’t that many Mohs surgery clinics in those days. During the latter part of my fellowship, Michael McCall, MD, and I had the opportunity to remove a skin cancer from the nose of Dr. Mohs. We presented the case at a national conference, and I titled the talk “Mohs Surgery for Mohs’ Nose.”
Early in your career Dr. Mohs asked you to take over his practice, but you accepted an offer to establish the first Mohs surgery office at Scripps in San Diego instead. What convinced you to head West?
After my fellowship, I returned to San Diego to complete my residency with the Navy, where we opened a Mohs surgery clinic. Dr. Mohs came out for the ribbon cutting. During that time, I was taking care of several patients that he had treated in Wisconsin. Through that my wife and I ended up going to dinner with Cecil and Ida Green, philanthropists who made several financial gifts to Scripps Clinic – and for whom Scripps Green Hospital is named. Cecil cofounded Texas Instruments and was knighted by Queen Elizabeth. During dinner, he suggested that I stay in San Diego for a year and work at Scripps after my residency assignment with the Navy. I agreed and have been here ever since.
What do you find most interesting about Mohs surgery?
In Mohs surgery, you’re able to provide not only surgical care to eliminate the tumor, but also the pathology and the reconstruction. That was interesting to me. Dr. Mohs was not that interested in reconstruction. He was more focused on the tumor, in part because with the original fixed-tissue technique you could not do the reconstruction. You had to wait for an extra layer of tissue to separate. But with the fresh-tissue technique, you were able to provide the reconstruction that day. Mohs surgery deals with a subset of tumors that are challenging to treat. That also spiked my academic and clinical interest.
In your opinion, what’s been the most important advance in Mohs surgery to date?
In recent years, immunology has come into play, so now we have teams of clinicians in dermatology, medical oncology, surgery, and other subspecialties providing patients the best of care. In the arena of Mohs surgery itself, in the 1980s, the American College of Mohs Surgery developed a 1-year fellowship program, which enabled us to train many men and women to practice Mohs surgery. Most of them are dermatologists.
Please complete the sentence: “You can tell a good Mohs surgeon by the way he/she ...”
Treats patients, is willing to spend time with them, and shows an interest in them. One of the things we should strive for is to let patients know that they as a person are important; it’s not just the melanoma on their nose. We’re not only dealing with a skin cancer; we’re dealing with a patient who has skin cancer.
For the past 39 years, you have led Hugh Greenway’s Superficial Anatomy and Cutaneous Surgery course, which takes place every January in San Diego. What’s been key to sustaining this training course for nearly 4 decades?
There have been many people involved in its success, so it’s not just me. When I first started my practice, there really was not a focus on anatomy in the general dermatologic community. Dermatologic surgery textbooks contained very little content on surgical anatomy so I developed an interest a putting together a course that would cover some of this material. I met with Terence Davidson, MD, an otolaryngologist who was dean of continuing medical education at the University of California, San Diego. The course includes lectures from experts in many subspecialties and hands-on laboratories using cadavers to work on anatomy and surgical techniques. After about 16 years of doing the course Dr. Davidson told me: “When we started this course, as a group, the head and neck surgeons were the best to do the reconstructions on the face with skin flaps and grafts and layered closures. But now, as a group, the dermatologists are best at doing that.” That’s what we want to hear in medical education.
During the peak of the COVID-19 pandemic, what were your most significant challenges from both a clinical and a personal standpoint?
I’m fortunate to practice at a place like Scripps, where there are many resources to look at what was happening with COVID-19. Clinically, we had to put a lot of things on hold, but we tried our best to keep our cancer patients in particular in the forefront of care. It has been a challenge, but fortunately we have been able to take care of patients after a brief timeout. Many of us remember the polio vaccine back in the 1950s. Having worked overseas and at missionary hospital where we had children die of measles because they were not vaccinated gave me a larger appreciation for the importance of vaccines. I recommend all young physicians who work with me to read, “The Great Influenza: The Story of the Deadliest Pandemic in History,” by John M. Barry, which recounts the 1918 flu epidemic.
Who inspires you most in your work today?
I don’t view what I do as work. Dr. Jones and Dr. Mohs continue to inspire me with what they accomplished during their careers. You have to love people and love patients. Every patient who comes to see me has a story, so I try to understand their story. One of the things I really enjoy is training the young fellows. We train three Mohs fellows per year at Scripps, and it’s a great challenge every day.
What development in dermatology are you most excited about in the next 5 years?
Dermatology will continue to evolve just like all other medical specialties. We’re going to see a large growth in telemedicine, and immunotherapy is playing a key role in dermatologic oncology. What excites me the most in medicine is the young people who enter the field willing to contribute their lives to helping others.
who was also recently selected as program director for cutaneous oncology at Scripps MD Anderson Cancer Center in San Diego. He is also a former president of the American College of Mohs Surgery.
After earning his medical degree from the Medical College of Georgia, Augusta, in 1974, Dr. Greenway was fellowship trained in Mohs skin cancer surgery by Frederic E. Mohs, MD, at the University of Wisconsin–Madison. He completed his dermatology residency at the Naval Medical Center San Diego and joined Scripps Clinic in 1983, where he launched the institution’s first Mohs surgery program, as well as a popular annual intensive course in superficial anatomy and cutaneous surgery that bears his name. He was also the first physician in the world to use interferon as a nonsurgical treatment of basal cell carcinoma.

To date, Dr. Greenway has performed more than 41,000 Mohs surgery cases and has trained 61 fellows who practice in academic and clinical settings. In 2017, he received the Frederic E. Mohs Award from the ACMS at the college’s annual meeting. He is also a past CEO of Scripps Clinic. In this Q&A, Dr. Greenway opens up about what it was like to train with Dr. Mohs, what makes a good Mohs surgeon, and why he’s excited about the future of dermatology.
I understand that you first became interested in a medical career after meeting Dr. Carl Jones, a friend of your father who was your Scoutmaster in the Boy Scouts in Georgia. What about Dr. Jones inspired you to pursue a career in medicine?
Dr. Jones was an internist/allergist in Atlanta, where I grew up. His three sons and I were friends. My dad had dealt with several medical problems being injured in World War II and subsequently undergoing a couple of kidney transplantations, so I developed an interest in medicine personally. Even though Dr. Jones was a specialist, he started out as a family doctor like I did, so he was interested in the whole person and all of his or her medical problems as opposed to those related to his specialty only. I traveled with the Boy Scouts to camp at places like Valley Forge in Pennsylvania, and Dr. Jones was involved with the medical set-ups of those large events. That also contributed to my interest in medicine.
As part of your 9-year service in the U.S. Navy, you spent 2 years as the flight surgeon at NAS Atlanta/Dobbins Air Force Base. What was your most memorable experience from that assignment?
Dobbins is a large facility with two Lockheed plants, and the Air Force had built the medical clinic, which was staffed by the Navy. Getting to know some of the active-duty members of the Air Force, the Navy, and the National Guard, and their commitment to our country, was memorable. Jimmy Carter was the president in those days. When he would fly in Dobbins, one of my jobs as the flight surgeon was to be on base when Air Force One landed or departed. One night, we had a DC-9 commercial aircraft coming from Huntsville, Ala., to Atlanta that got caught in a thunderstorm a little above 30,000 feet. Both engines went out and the aircraft essentially became a glider. The pilots tried to land on our runway but unfortunately, they ended up 4 miles short. We were heavily involved in responding to the crash, which was a tragic event. I also learned to fly (second seat) different types of aircraft during my assignment at NAS Atlanta/Dobbins Air Force Base, everything from the large C-5s to Navy fighter jets and helicopters. Coincidentally, Dr. Jones was involved with a couple of free health clinics in Atlanta when I was stationed there. Every Tuesday night, my wife (who is a nurse) and I would volunteer at a clinic in Cabbagetown, which was one of the poorer areas of Atlanta. It was a chance to give back to a group of people who didn’t have a whole lot.
In the middle your dermatology residency at Naval Medical Center San Diego, you were selected by Dr. Mohs for fellowship training in Mohs skin cancer surgery at the University of Wisconsin–Madison. What do you remember most about your training with Dr. Mohs?
Dr. Mohs was a kind, humble man who had this great idea about skin cancer. He was not a dermatologist; he was a general surgeon. The technique he developed was originally called chemosurgery because he put a chemical onto the skin. This was known as the fixed-tissue technique. Then we had a fresh-tissue technique, where we did not use the chemical, but we were able to use local anesthesia right away. That developed into the Mohs surgery we know today. Dr. Mohs did not name it that; he was very humble, but he was very proud of his technique. He was also a very hard worker. On the first day of my fellowship, I started at 7 in the morning and ended at 7 at night. It was the same for the last day of my fellowship. He also had an excellent office staff, many of whom had worked with him for many years. Patients with difficult skin cancers traveled to Madison from all over the world because there weren’t that many Mohs surgery clinics in those days. During the latter part of my fellowship, Michael McCall, MD, and I had the opportunity to remove a skin cancer from the nose of Dr. Mohs. We presented the case at a national conference, and I titled the talk “Mohs Surgery for Mohs’ Nose.”
Early in your career Dr. Mohs asked you to take over his practice, but you accepted an offer to establish the first Mohs surgery office at Scripps in San Diego instead. What convinced you to head West?
After my fellowship, I returned to San Diego to complete my residency with the Navy, where we opened a Mohs surgery clinic. Dr. Mohs came out for the ribbon cutting. During that time, I was taking care of several patients that he had treated in Wisconsin. Through that my wife and I ended up going to dinner with Cecil and Ida Green, philanthropists who made several financial gifts to Scripps Clinic – and for whom Scripps Green Hospital is named. Cecil cofounded Texas Instruments and was knighted by Queen Elizabeth. During dinner, he suggested that I stay in San Diego for a year and work at Scripps after my residency assignment with the Navy. I agreed and have been here ever since.
What do you find most interesting about Mohs surgery?
In Mohs surgery, you’re able to provide not only surgical care to eliminate the tumor, but also the pathology and the reconstruction. That was interesting to me. Dr. Mohs was not that interested in reconstruction. He was more focused on the tumor, in part because with the original fixed-tissue technique you could not do the reconstruction. You had to wait for an extra layer of tissue to separate. But with the fresh-tissue technique, you were able to provide the reconstruction that day. Mohs surgery deals with a subset of tumors that are challenging to treat. That also spiked my academic and clinical interest.
In your opinion, what’s been the most important advance in Mohs surgery to date?
In recent years, immunology has come into play, so now we have teams of clinicians in dermatology, medical oncology, surgery, and other subspecialties providing patients the best of care. In the arena of Mohs surgery itself, in the 1980s, the American College of Mohs Surgery developed a 1-year fellowship program, which enabled us to train many men and women to practice Mohs surgery. Most of them are dermatologists.
Please complete the sentence: “You can tell a good Mohs surgeon by the way he/she ...”
Treats patients, is willing to spend time with them, and shows an interest in them. One of the things we should strive for is to let patients know that they as a person are important; it’s not just the melanoma on their nose. We’re not only dealing with a skin cancer; we’re dealing with a patient who has skin cancer.
For the past 39 years, you have led Hugh Greenway’s Superficial Anatomy and Cutaneous Surgery course, which takes place every January in San Diego. What’s been key to sustaining this training course for nearly 4 decades?
There have been many people involved in its success, so it’s not just me. When I first started my practice, there really was not a focus on anatomy in the general dermatologic community. Dermatologic surgery textbooks contained very little content on surgical anatomy so I developed an interest a putting together a course that would cover some of this material. I met with Terence Davidson, MD, an otolaryngologist who was dean of continuing medical education at the University of California, San Diego. The course includes lectures from experts in many subspecialties and hands-on laboratories using cadavers to work on anatomy and surgical techniques. After about 16 years of doing the course Dr. Davidson told me: “When we started this course, as a group, the head and neck surgeons were the best to do the reconstructions on the face with skin flaps and grafts and layered closures. But now, as a group, the dermatologists are best at doing that.” That’s what we want to hear in medical education.
During the peak of the COVID-19 pandemic, what were your most significant challenges from both a clinical and a personal standpoint?
I’m fortunate to practice at a place like Scripps, where there are many resources to look at what was happening with COVID-19. Clinically, we had to put a lot of things on hold, but we tried our best to keep our cancer patients in particular in the forefront of care. It has been a challenge, but fortunately we have been able to take care of patients after a brief timeout. Many of us remember the polio vaccine back in the 1950s. Having worked overseas and at missionary hospital where we had children die of measles because they were not vaccinated gave me a larger appreciation for the importance of vaccines. I recommend all young physicians who work with me to read, “The Great Influenza: The Story of the Deadliest Pandemic in History,” by John M. Barry, which recounts the 1918 flu epidemic.
Who inspires you most in your work today?
I don’t view what I do as work. Dr. Jones and Dr. Mohs continue to inspire me with what they accomplished during their careers. You have to love people and love patients. Every patient who comes to see me has a story, so I try to understand their story. One of the things I really enjoy is training the young fellows. We train three Mohs fellows per year at Scripps, and it’s a great challenge every day.
What development in dermatology are you most excited about in the next 5 years?
Dermatology will continue to evolve just like all other medical specialties. We’re going to see a large growth in telemedicine, and immunotherapy is playing a key role in dermatologic oncology. What excites me the most in medicine is the young people who enter the field willing to contribute their lives to helping others.
who was also recently selected as program director for cutaneous oncology at Scripps MD Anderson Cancer Center in San Diego. He is also a former president of the American College of Mohs Surgery.
After earning his medical degree from the Medical College of Georgia, Augusta, in 1974, Dr. Greenway was fellowship trained in Mohs skin cancer surgery by Frederic E. Mohs, MD, at the University of Wisconsin–Madison. He completed his dermatology residency at the Naval Medical Center San Diego and joined Scripps Clinic in 1983, where he launched the institution’s first Mohs surgery program, as well as a popular annual intensive course in superficial anatomy and cutaneous surgery that bears his name. He was also the first physician in the world to use interferon as a nonsurgical treatment of basal cell carcinoma.

To date, Dr. Greenway has performed more than 41,000 Mohs surgery cases and has trained 61 fellows who practice in academic and clinical settings. In 2017, he received the Frederic E. Mohs Award from the ACMS at the college’s annual meeting. He is also a past CEO of Scripps Clinic. In this Q&A, Dr. Greenway opens up about what it was like to train with Dr. Mohs, what makes a good Mohs surgeon, and why he’s excited about the future of dermatology.
I understand that you first became interested in a medical career after meeting Dr. Carl Jones, a friend of your father who was your Scoutmaster in the Boy Scouts in Georgia. What about Dr. Jones inspired you to pursue a career in medicine?
Dr. Jones was an internist/allergist in Atlanta, where I grew up. His three sons and I were friends. My dad had dealt with several medical problems being injured in World War II and subsequently undergoing a couple of kidney transplantations, so I developed an interest in medicine personally. Even though Dr. Jones was a specialist, he started out as a family doctor like I did, so he was interested in the whole person and all of his or her medical problems as opposed to those related to his specialty only. I traveled with the Boy Scouts to camp at places like Valley Forge in Pennsylvania, and Dr. Jones was involved with the medical set-ups of those large events. That also contributed to my interest in medicine.
As part of your 9-year service in the U.S. Navy, you spent 2 years as the flight surgeon at NAS Atlanta/Dobbins Air Force Base. What was your most memorable experience from that assignment?
Dobbins is a large facility with two Lockheed plants, and the Air Force had built the medical clinic, which was staffed by the Navy. Getting to know some of the active-duty members of the Air Force, the Navy, and the National Guard, and their commitment to our country, was memorable. Jimmy Carter was the president in those days. When he would fly in Dobbins, one of my jobs as the flight surgeon was to be on base when Air Force One landed or departed. One night, we had a DC-9 commercial aircraft coming from Huntsville, Ala., to Atlanta that got caught in a thunderstorm a little above 30,000 feet. Both engines went out and the aircraft essentially became a glider. The pilots tried to land on our runway but unfortunately, they ended up 4 miles short. We were heavily involved in responding to the crash, which was a tragic event. I also learned to fly (second seat) different types of aircraft during my assignment at NAS Atlanta/Dobbins Air Force Base, everything from the large C-5s to Navy fighter jets and helicopters. Coincidentally, Dr. Jones was involved with a couple of free health clinics in Atlanta when I was stationed there. Every Tuesday night, my wife (who is a nurse) and I would volunteer at a clinic in Cabbagetown, which was one of the poorer areas of Atlanta. It was a chance to give back to a group of people who didn’t have a whole lot.
In the middle your dermatology residency at Naval Medical Center San Diego, you were selected by Dr. Mohs for fellowship training in Mohs skin cancer surgery at the University of Wisconsin–Madison. What do you remember most about your training with Dr. Mohs?
Dr. Mohs was a kind, humble man who had this great idea about skin cancer. He was not a dermatologist; he was a general surgeon. The technique he developed was originally called chemosurgery because he put a chemical onto the skin. This was known as the fixed-tissue technique. Then we had a fresh-tissue technique, where we did not use the chemical, but we were able to use local anesthesia right away. That developed into the Mohs surgery we know today. Dr. Mohs did not name it that; he was very humble, but he was very proud of his technique. He was also a very hard worker. On the first day of my fellowship, I started at 7 in the morning and ended at 7 at night. It was the same for the last day of my fellowship. He also had an excellent office staff, many of whom had worked with him for many years. Patients with difficult skin cancers traveled to Madison from all over the world because there weren’t that many Mohs surgery clinics in those days. During the latter part of my fellowship, Michael McCall, MD, and I had the opportunity to remove a skin cancer from the nose of Dr. Mohs. We presented the case at a national conference, and I titled the talk “Mohs Surgery for Mohs’ Nose.”
Early in your career Dr. Mohs asked you to take over his practice, but you accepted an offer to establish the first Mohs surgery office at Scripps in San Diego instead. What convinced you to head West?
After my fellowship, I returned to San Diego to complete my residency with the Navy, where we opened a Mohs surgery clinic. Dr. Mohs came out for the ribbon cutting. During that time, I was taking care of several patients that he had treated in Wisconsin. Through that my wife and I ended up going to dinner with Cecil and Ida Green, philanthropists who made several financial gifts to Scripps Clinic – and for whom Scripps Green Hospital is named. Cecil cofounded Texas Instruments and was knighted by Queen Elizabeth. During dinner, he suggested that I stay in San Diego for a year and work at Scripps after my residency assignment with the Navy. I agreed and have been here ever since.
What do you find most interesting about Mohs surgery?
In Mohs surgery, you’re able to provide not only surgical care to eliminate the tumor, but also the pathology and the reconstruction. That was interesting to me. Dr. Mohs was not that interested in reconstruction. He was more focused on the tumor, in part because with the original fixed-tissue technique you could not do the reconstruction. You had to wait for an extra layer of tissue to separate. But with the fresh-tissue technique, you were able to provide the reconstruction that day. Mohs surgery deals with a subset of tumors that are challenging to treat. That also spiked my academic and clinical interest.
In your opinion, what’s been the most important advance in Mohs surgery to date?
In recent years, immunology has come into play, so now we have teams of clinicians in dermatology, medical oncology, surgery, and other subspecialties providing patients the best of care. In the arena of Mohs surgery itself, in the 1980s, the American College of Mohs Surgery developed a 1-year fellowship program, which enabled us to train many men and women to practice Mohs surgery. Most of them are dermatologists.
Please complete the sentence: “You can tell a good Mohs surgeon by the way he/she ...”
Treats patients, is willing to spend time with them, and shows an interest in them. One of the things we should strive for is to let patients know that they as a person are important; it’s not just the melanoma on their nose. We’re not only dealing with a skin cancer; we’re dealing with a patient who has skin cancer.
For the past 39 years, you have led Hugh Greenway’s Superficial Anatomy and Cutaneous Surgery course, which takes place every January in San Diego. What’s been key to sustaining this training course for nearly 4 decades?
There have been many people involved in its success, so it’s not just me. When I first started my practice, there really was not a focus on anatomy in the general dermatologic community. Dermatologic surgery textbooks contained very little content on surgical anatomy so I developed an interest a putting together a course that would cover some of this material. I met with Terence Davidson, MD, an otolaryngologist who was dean of continuing medical education at the University of California, San Diego. The course includes lectures from experts in many subspecialties and hands-on laboratories using cadavers to work on anatomy and surgical techniques. After about 16 years of doing the course Dr. Davidson told me: “When we started this course, as a group, the head and neck surgeons were the best to do the reconstructions on the face with skin flaps and grafts and layered closures. But now, as a group, the dermatologists are best at doing that.” That’s what we want to hear in medical education.
During the peak of the COVID-19 pandemic, what were your most significant challenges from both a clinical and a personal standpoint?
I’m fortunate to practice at a place like Scripps, where there are many resources to look at what was happening with COVID-19. Clinically, we had to put a lot of things on hold, but we tried our best to keep our cancer patients in particular in the forefront of care. It has been a challenge, but fortunately we have been able to take care of patients after a brief timeout. Many of us remember the polio vaccine back in the 1950s. Having worked overseas and at missionary hospital where we had children die of measles because they were not vaccinated gave me a larger appreciation for the importance of vaccines. I recommend all young physicians who work with me to read, “The Great Influenza: The Story of the Deadliest Pandemic in History,” by John M. Barry, which recounts the 1918 flu epidemic.
Who inspires you most in your work today?
I don’t view what I do as work. Dr. Jones and Dr. Mohs continue to inspire me with what they accomplished during their careers. You have to love people and love patients. Every patient who comes to see me has a story, so I try to understand their story. One of the things I really enjoy is training the young fellows. We train three Mohs fellows per year at Scripps, and it’s a great challenge every day.
What development in dermatology are you most excited about in the next 5 years?
Dermatology will continue to evolve just like all other medical specialties. We’re going to see a large growth in telemedicine, and immunotherapy is playing a key role in dermatologic oncology. What excites me the most in medicine is the young people who enter the field willing to contribute their lives to helping others.
Study provides new analysis of isotretinoin and risk for adverse neuropsychiatric outcomes
The use of , in a large retrospective cohort study published in the British Journal of Dermatology.
Although severe neuropsychiatric effects associated with isotretinoin therapy in patients with acne have been reported, “the evidence base ... is mixed and inconclusive,” and many studies are small, Seena Fazel, MBChB, MD, of the department of psychiatry, Oxford University, England, and co-authors write in the study.
The study results suggest that isotretinoin is conferring protection against adverse neuropsychiatric outcomes, particularly when compared with using oral antibiotics to treat acne, Dr. Fazel, professor of forensic psychiatry at Oxford University and the study’s senior author, said in an interview.
In the study, the investigators reviewed electronic health records (2013-2019) from a primarily United States–based dataset (TriNetX) of patients with acne aged 12-27 who had been followed for up to 1 year after their prescriptions had been dispensed.
There were four arms: those prescribed isotretinoin (30,866), oral antibiotics (44,748), topical anti-acne treatments (108,367), and those who had not been prescribed any acne treatment (78,666). The primary outcomes were diagnoses of a neuropsychiatric disorder (psychotic, mood, anxiety, personality, behavioral, and sleep disorders; and non-fatal self-harm) within one year of being prescribed treatment.
After using propensity score matching to adjust for confounders at baseline, the investigators determined that the odds ratio for any incident neuropsychiatric outcomes among patients with acne treated with isotretinoin was 0.80 (95% confidence interval, 0.74-0.87), compared with patients on oral antibiotics; 0.94 (95% CI, 0.87-1.02), compared with patients on topical anti-acne medications; and 1.06 (95% CI, 0.97-1.16), compared with those without a prescription for anti-acne medicines.
Side effects of isotretinoin – such as headache, dry mouth, and fatigue – were higher among those on isotretinoin than in the other three groups.
The authors concluded that isotretinoin was not independently linked to excess adverse neuropsychiatric outcomes at a population level. “We observed a consistent association between increasing acne severity as indicated by anti-acne treatment options and incidence of adverse neuropsychiatric outcomes, but the findings showed that isotretinoin exposure did not add to the risk of neuropsychiatric adverse outcomes over and above what was associated with oral antibiotics,” they write.
Isotretinoin treatment “appeared to mitigate the excess neuropsychiatric risk associated with recalcitrant moderate-to-severe acne,” they add.
The dermatology community has been interested in the impact isotretinoin has on mental health, and “I think clinically, they see that people get better on isotretinoin and their mental health improves,” Dr. Fazel told this news organization.
Asked to comment on the study results, John Barbieri, MD, MBA, director of the Advanced Acne Therapeutics Clinic, Brigham and Women’s Hospital, Boston, commended the investigators for the design of the trial.
“One of the strengths of this study is that they use a technique called propensity-score matching, where you try to make the groups of patients similar with respect to their other characteristics to minimize the risks of confounding and bias in the study, which I think is a real strength,” he told this news organization. “The other thing that they do, which I think is a strength, is to think about the impact of acne severity on these outcomes, because we know acne itself is associated with depression and risk for suicide and other neuropsychiatric outcomes.”
Including a cohort of patients who had acne and received oral antibiotics for comparison “is a nice way to address the potential for confounding by severity and confounding by indication,” Dr. Barbieri said. “Those who get antibiotics usually have more severe acne. They may not have it as severely as those who get isotretinoin, but it is a nice approach to account for background levels of depression and neuropsychiatric outcomes in patients with acne. I think that is a real strength of the study. This is one of the best studies to have looked at this question.”
However, although the study found that isotretinoin decreased the excess psychiatric risk associated with refractory moderate-to-severe acne, it does not rule out the possibility that individuals may experience an adverse psychiatric outcome while on isotretinoin, Dr. Barbieri said.
“While I think on a population level, we absolutely can feel reassured by these data, I do think there are individual patients who have idiosyncratic, unpredictable reactions to isotretinoin where they have mood changes, whether it be irritability, depression, or other mood changes,” he cautioned. “Given the association of acne itself with mental health comorbidities, it is important to screen for comorbidities such as depression in all patients with acne.”
The study was funded by the Wellcome Trust, which provided Dr. Fazel and the first author with financial support for the study. One author is an employee of TriNetX; the other authors had no relevant disclosures. Dr. Barbieri reported no financial disclosures. He is cochair of the AAD’s Acne Guidelines Workgroup and associate editor at JAMA Dermatology.
A version of this article first appeared on Medscape.com.
The use of , in a large retrospective cohort study published in the British Journal of Dermatology.
Although severe neuropsychiatric effects associated with isotretinoin therapy in patients with acne have been reported, “the evidence base ... is mixed and inconclusive,” and many studies are small, Seena Fazel, MBChB, MD, of the department of psychiatry, Oxford University, England, and co-authors write in the study.
The study results suggest that isotretinoin is conferring protection against adverse neuropsychiatric outcomes, particularly when compared with using oral antibiotics to treat acne, Dr. Fazel, professor of forensic psychiatry at Oxford University and the study’s senior author, said in an interview.
In the study, the investigators reviewed electronic health records (2013-2019) from a primarily United States–based dataset (TriNetX) of patients with acne aged 12-27 who had been followed for up to 1 year after their prescriptions had been dispensed.
There were four arms: those prescribed isotretinoin (30,866), oral antibiotics (44,748), topical anti-acne treatments (108,367), and those who had not been prescribed any acne treatment (78,666). The primary outcomes were diagnoses of a neuropsychiatric disorder (psychotic, mood, anxiety, personality, behavioral, and sleep disorders; and non-fatal self-harm) within one year of being prescribed treatment.
After using propensity score matching to adjust for confounders at baseline, the investigators determined that the odds ratio for any incident neuropsychiatric outcomes among patients with acne treated with isotretinoin was 0.80 (95% confidence interval, 0.74-0.87), compared with patients on oral antibiotics; 0.94 (95% CI, 0.87-1.02), compared with patients on topical anti-acne medications; and 1.06 (95% CI, 0.97-1.16), compared with those without a prescription for anti-acne medicines.
Side effects of isotretinoin – such as headache, dry mouth, and fatigue – were higher among those on isotretinoin than in the other three groups.
The authors concluded that isotretinoin was not independently linked to excess adverse neuropsychiatric outcomes at a population level. “We observed a consistent association between increasing acne severity as indicated by anti-acne treatment options and incidence of adverse neuropsychiatric outcomes, but the findings showed that isotretinoin exposure did not add to the risk of neuropsychiatric adverse outcomes over and above what was associated with oral antibiotics,” they write.
Isotretinoin treatment “appeared to mitigate the excess neuropsychiatric risk associated with recalcitrant moderate-to-severe acne,” they add.
The dermatology community has been interested in the impact isotretinoin has on mental health, and “I think clinically, they see that people get better on isotretinoin and their mental health improves,” Dr. Fazel told this news organization.
Asked to comment on the study results, John Barbieri, MD, MBA, director of the Advanced Acne Therapeutics Clinic, Brigham and Women’s Hospital, Boston, commended the investigators for the design of the trial.
“One of the strengths of this study is that they use a technique called propensity-score matching, where you try to make the groups of patients similar with respect to their other characteristics to minimize the risks of confounding and bias in the study, which I think is a real strength,” he told this news organization. “The other thing that they do, which I think is a strength, is to think about the impact of acne severity on these outcomes, because we know acne itself is associated with depression and risk for suicide and other neuropsychiatric outcomes.”
Including a cohort of patients who had acne and received oral antibiotics for comparison “is a nice way to address the potential for confounding by severity and confounding by indication,” Dr. Barbieri said. “Those who get antibiotics usually have more severe acne. They may not have it as severely as those who get isotretinoin, but it is a nice approach to account for background levels of depression and neuropsychiatric outcomes in patients with acne. I think that is a real strength of the study. This is one of the best studies to have looked at this question.”
However, although the study found that isotretinoin decreased the excess psychiatric risk associated with refractory moderate-to-severe acne, it does not rule out the possibility that individuals may experience an adverse psychiatric outcome while on isotretinoin, Dr. Barbieri said.
“While I think on a population level, we absolutely can feel reassured by these data, I do think there are individual patients who have idiosyncratic, unpredictable reactions to isotretinoin where they have mood changes, whether it be irritability, depression, or other mood changes,” he cautioned. “Given the association of acne itself with mental health comorbidities, it is important to screen for comorbidities such as depression in all patients with acne.”
The study was funded by the Wellcome Trust, which provided Dr. Fazel and the first author with financial support for the study. One author is an employee of TriNetX; the other authors had no relevant disclosures. Dr. Barbieri reported no financial disclosures. He is cochair of the AAD’s Acne Guidelines Workgroup and associate editor at JAMA Dermatology.
A version of this article first appeared on Medscape.com.
The use of , in a large retrospective cohort study published in the British Journal of Dermatology.
Although severe neuropsychiatric effects associated with isotretinoin therapy in patients with acne have been reported, “the evidence base ... is mixed and inconclusive,” and many studies are small, Seena Fazel, MBChB, MD, of the department of psychiatry, Oxford University, England, and co-authors write in the study.
The study results suggest that isotretinoin is conferring protection against adverse neuropsychiatric outcomes, particularly when compared with using oral antibiotics to treat acne, Dr. Fazel, professor of forensic psychiatry at Oxford University and the study’s senior author, said in an interview.
In the study, the investigators reviewed electronic health records (2013-2019) from a primarily United States–based dataset (TriNetX) of patients with acne aged 12-27 who had been followed for up to 1 year after their prescriptions had been dispensed.
There were four arms: those prescribed isotretinoin (30,866), oral antibiotics (44,748), topical anti-acne treatments (108,367), and those who had not been prescribed any acne treatment (78,666). The primary outcomes were diagnoses of a neuropsychiatric disorder (psychotic, mood, anxiety, personality, behavioral, and sleep disorders; and non-fatal self-harm) within one year of being prescribed treatment.
After using propensity score matching to adjust for confounders at baseline, the investigators determined that the odds ratio for any incident neuropsychiatric outcomes among patients with acne treated with isotretinoin was 0.80 (95% confidence interval, 0.74-0.87), compared with patients on oral antibiotics; 0.94 (95% CI, 0.87-1.02), compared with patients on topical anti-acne medications; and 1.06 (95% CI, 0.97-1.16), compared with those without a prescription for anti-acne medicines.
Side effects of isotretinoin – such as headache, dry mouth, and fatigue – were higher among those on isotretinoin than in the other three groups.
The authors concluded that isotretinoin was not independently linked to excess adverse neuropsychiatric outcomes at a population level. “We observed a consistent association between increasing acne severity as indicated by anti-acne treatment options and incidence of adverse neuropsychiatric outcomes, but the findings showed that isotretinoin exposure did not add to the risk of neuropsychiatric adverse outcomes over and above what was associated with oral antibiotics,” they write.
Isotretinoin treatment “appeared to mitigate the excess neuropsychiatric risk associated with recalcitrant moderate-to-severe acne,” they add.
The dermatology community has been interested in the impact isotretinoin has on mental health, and “I think clinically, they see that people get better on isotretinoin and their mental health improves,” Dr. Fazel told this news organization.
Asked to comment on the study results, John Barbieri, MD, MBA, director of the Advanced Acne Therapeutics Clinic, Brigham and Women’s Hospital, Boston, commended the investigators for the design of the trial.
“One of the strengths of this study is that they use a technique called propensity-score matching, where you try to make the groups of patients similar with respect to their other characteristics to minimize the risks of confounding and bias in the study, which I think is a real strength,” he told this news organization. “The other thing that they do, which I think is a strength, is to think about the impact of acne severity on these outcomes, because we know acne itself is associated with depression and risk for suicide and other neuropsychiatric outcomes.”
Including a cohort of patients who had acne and received oral antibiotics for comparison “is a nice way to address the potential for confounding by severity and confounding by indication,” Dr. Barbieri said. “Those who get antibiotics usually have more severe acne. They may not have it as severely as those who get isotretinoin, but it is a nice approach to account for background levels of depression and neuropsychiatric outcomes in patients with acne. I think that is a real strength of the study. This is one of the best studies to have looked at this question.”
However, although the study found that isotretinoin decreased the excess psychiatric risk associated with refractory moderate-to-severe acne, it does not rule out the possibility that individuals may experience an adverse psychiatric outcome while on isotretinoin, Dr. Barbieri said.
“While I think on a population level, we absolutely can feel reassured by these data, I do think there are individual patients who have idiosyncratic, unpredictable reactions to isotretinoin where they have mood changes, whether it be irritability, depression, or other mood changes,” he cautioned. “Given the association of acne itself with mental health comorbidities, it is important to screen for comorbidities such as depression in all patients with acne.”
The study was funded by the Wellcome Trust, which provided Dr. Fazel and the first author with financial support for the study. One author is an employee of TriNetX; the other authors had no relevant disclosures. Dr. Barbieri reported no financial disclosures. He is cochair of the AAD’s Acne Guidelines Workgroup and associate editor at JAMA Dermatology.
A version of this article first appeared on Medscape.com.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Focal hair loss
The findings of smooth, round alopecia occurring rapidly without associated scarring, pain, or itching, is consistent with the diagnosis of alopecia areata.
Alopecia areata is a common autoimmune disease caused by T lymphocytes targeting hair follicles and resulting in rapid and nonscarring hair loss. It is usually self-resolving and about 2% of all individuals are affected at some point during their lifetime, with an average age of onset of 33 years.1 Some patients may progress to loss of all scalp hair (alopecia totalis) or all hair on the scalp and body (alopecia universalis).1
It is important to inspect a patient’s scalp, face, and body for more subtle areas of loss that could signal other disorders, such as lichen planopilaris, discoid lupus, or telogen effluvium. It is worth noting that alopecia areata is not associated with scalp lesions, crusting, or scars without follicles. Such findings should be further investigated with a 4-mm punch biopsy of affected and adjacent follicular units. Carefully labeling biopsy specimens as scalp specimens for hair loss will aid in a correct histopathologic diagnosis.
Systematic data comparing treatments for alopecia areata are lacking. For localized disease, topical or intradermal triamcinolone injections at a concentration of 5 to 10 mg/mL, with about 0.1 mL to 0.05 mL injected every square centimeter of affected area (up to 40 mg per visit), can provide rapid regrowth.1 Within 4 months of the monthly injections, 63% of patients experience complete regrowth.1 Despite this favorable outcome, there is also a high rate of recurrence.
For more widespread disease, contact immunotherapy with squaric acid dibutyl ester or diphencyprone can provoke a low-grade contact allergy and induce antigenic completion. This therapy is painless but can be itchy; medications must be compounded and titrated to activity.
The patient in this case opted to receive monthly triamcinolone injections in an undiluted concentration of 10 mg/mL for 3 months, at which point she experienced excellent hair regrowth. A small patch of recurrence was noted a year later and treated twice with monthly triamcinolone injections.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Darwin E, Hirt PA, Fertig R, et al. Alopecia areata: review of epidemiology, clinical features, pathogenesis, and new treatment options. Int J Trichology. 2018;10:51-60. doi: 10.4103/ijt.ijt_99_17

The findings of smooth, round alopecia occurring rapidly without associated scarring, pain, or itching, is consistent with the diagnosis of alopecia areata.
Alopecia areata is a common autoimmune disease caused by T lymphocytes targeting hair follicles and resulting in rapid and nonscarring hair loss. It is usually self-resolving and about 2% of all individuals are affected at some point during their lifetime, with an average age of onset of 33 years.1 Some patients may progress to loss of all scalp hair (alopecia totalis) or all hair on the scalp and body (alopecia universalis).1
It is important to inspect a patient’s scalp, face, and body for more subtle areas of loss that could signal other disorders, such as lichen planopilaris, discoid lupus, or telogen effluvium. It is worth noting that alopecia areata is not associated with scalp lesions, crusting, or scars without follicles. Such findings should be further investigated with a 4-mm punch biopsy of affected and adjacent follicular units. Carefully labeling biopsy specimens as scalp specimens for hair loss will aid in a correct histopathologic diagnosis.
Systematic data comparing treatments for alopecia areata are lacking. For localized disease, topical or intradermal triamcinolone injections at a concentration of 5 to 10 mg/mL, with about 0.1 mL to 0.05 mL injected every square centimeter of affected area (up to 40 mg per visit), can provide rapid regrowth.1 Within 4 months of the monthly injections, 63% of patients experience complete regrowth.1 Despite this favorable outcome, there is also a high rate of recurrence.
For more widespread disease, contact immunotherapy with squaric acid dibutyl ester or diphencyprone can provoke a low-grade contact allergy and induce antigenic completion. This therapy is painless but can be itchy; medications must be compounded and titrated to activity.
The patient in this case opted to receive monthly triamcinolone injections in an undiluted concentration of 10 mg/mL for 3 months, at which point she experienced excellent hair regrowth. A small patch of recurrence was noted a year later and treated twice with monthly triamcinolone injections.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

The findings of smooth, round alopecia occurring rapidly without associated scarring, pain, or itching, is consistent with the diagnosis of alopecia areata.
Alopecia areata is a common autoimmune disease caused by T lymphocytes targeting hair follicles and resulting in rapid and nonscarring hair loss. It is usually self-resolving and about 2% of all individuals are affected at some point during their lifetime, with an average age of onset of 33 years.1 Some patients may progress to loss of all scalp hair (alopecia totalis) or all hair on the scalp and body (alopecia universalis).1
It is important to inspect a patient’s scalp, face, and body for more subtle areas of loss that could signal other disorders, such as lichen planopilaris, discoid lupus, or telogen effluvium. It is worth noting that alopecia areata is not associated with scalp lesions, crusting, or scars without follicles. Such findings should be further investigated with a 4-mm punch biopsy of affected and adjacent follicular units. Carefully labeling biopsy specimens as scalp specimens for hair loss will aid in a correct histopathologic diagnosis.
Systematic data comparing treatments for alopecia areata are lacking. For localized disease, topical or intradermal triamcinolone injections at a concentration of 5 to 10 mg/mL, with about 0.1 mL to 0.05 mL injected every square centimeter of affected area (up to 40 mg per visit), can provide rapid regrowth.1 Within 4 months of the monthly injections, 63% of patients experience complete regrowth.1 Despite this favorable outcome, there is also a high rate of recurrence.
For more widespread disease, contact immunotherapy with squaric acid dibutyl ester or diphencyprone can provoke a low-grade contact allergy and induce antigenic completion. This therapy is painless but can be itchy; medications must be compounded and titrated to activity.
The patient in this case opted to receive monthly triamcinolone injections in an undiluted concentration of 10 mg/mL for 3 months, at which point she experienced excellent hair regrowth. A small patch of recurrence was noted a year later and treated twice with monthly triamcinolone injections.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Darwin E, Hirt PA, Fertig R, et al. Alopecia areata: review of epidemiology, clinical features, pathogenesis, and new treatment options. Int J Trichology. 2018;10:51-60. doi: 10.4103/ijt.ijt_99_17
1. Darwin E, Hirt PA, Fertig R, et al. Alopecia areata: review of epidemiology, clinical features, pathogenesis, and new treatment options. Int J Trichology. 2018;10:51-60. doi: 10.4103/ijt.ijt_99_17
PIH in patients with dark skin responds to laser treatment: Small case series
SAN DIEGO – , results from a small retrospective case series suggest.
“Postinflammatory hyperpigmentation is a leading chief of complaint of many skin of color persons seeking a dermatologist,” Elizabeth J. Kream, MD, told this news organization in advance of the annual conference of American Society for Laser Medicine and Surgery. “I describe PIH to patients as the ‘ashes after a fire is extinguished.’ It’s the stubborn brown to gray/black spots that persist after conditions like acne and folliculitis, but it can be caused by any insult to the skin including external injury. In fact, there’s a risk of inciting PIH with lasers and energy-based devices and this risk is greater in skin of color given the greater melanin content. Unfortunately, we see patients present after visiting a med spa who were treated with the wrong devices and/or the wrong settings and they have disfiguring scarring and/or dyspigmentation.”
During an abstract session at the meeting, Dr. Kream, a dermatology resident at the University of Illinois at Chicago, discussed three patients with recalcitrant PIH and Fitzpatrick skin phototype V and VI who were treated in San Diego with a combination of topical and laser therapies. She presented the case series on behalf of coauthors Monica Boen, MD and Douglas C. Wu, MD, dermatologists who practice in San Diego.
The first patient was a 37-year-old Black female who presented for evaluation of longstanding hyperpigmentation on the face and neck determined to be PIH secondary to folliculitis on the chin and neck. She was started on 8% hydroquinone with kojic acid daily and received four treatments spaced 4-8 weeks apart with the 1,927-nm fractional nonablative diode laser. Laser settings were 5 mJ pulse energy and 5% coverage after eight passes. Triamcinolone 0.1% ointment was applied immediately after treatment and for 3 days following treatment, and the “patient experienced near complete resolution of PIH with no unexpected adverse events,” Dr. Kream said.
The second patient was a 20-year-old Black male who presented with a 3-month history of facial hyperpigmentation after suffering a laser-induced injury. He was started on a non-hydroquinone topical lightening agent and received five treatments spaced 2 weeks apart with a 1,927-nm fractional nonablative diode laser. The laser settings were 5 mJ pulse energy and 5% coverage after eight passes. The patient experienced 80%-90% resolution of his PIH with no unexpected adverse reactions.
The third patient in the series was a 39-year-old Black male who presented with a 6-month history of hyperpigmentation on his right shin and calf, secondary to minor occupational-related trauma. Treatment was initiated with a fractional 1,064-nm picosecond laser. The laser settings were 2.1 mJ per microbeam microwave pulse energy and a 450 picosecond pulse duration delivered at 2 Hz through a holographic beam splitter with a 6 x 6–mm spot size containing 101 microbeams, for an estimated coverage of 4% per pulse. Four passes were performed for each area. The endpoint was a mild erythema to several treated areas a few minutes following laser treatment. Postoperative care consisted of applying a non-hydroquinone topical lightening agent twice daily to the affected area for 1 month. Near-complete resolution of the PIH was achieved, with no unexpected adverse reactions.
“In our clinical experience, PIH can be treated with the combination of topical skin lighteners and low density, low fluence laser therapy in almost all skin types,” Dr. Kream said. “The rationale behind this combination is to treat and remove existing pigment with the laser therapy while minimizing and preventing any pigmentary recurrence with diligent topical therapy and photoprotection.”
It is important to identify the cause of PIH “because some cases are trickier than others,” such as a lichenoid process that deposits pigment “a little bit deeper into the dermis,” she said. “When selecting an appropriate laser modality for the treatment of PIH in skin types V and VI, it’s especially important to consider the mechanism of action, depth of penetration, degree of tissue damage, and the extent of disruption to the dermal-epidermal junction.”
Following the presentation, one of the session moderators, Albert Wolkerstorfer, MD, PhD, a dermatologist at Amsterdam University Medical Center, the Netherlands, emphasized the importance of proper patient selection for laser treatment of PIH. “Not every patient with PIH is adapted to treatment with the laser,” Dr. Wolkerstorfer said. “I think it’s also important to choose stable PIH, meaning you often see patients with an underlying disorder who want to get rid of the pigment. They often believe that the laser is the solution, but it often isn’t.”
During a question-and-answer session, a meeting attendee pointed out that the study lacked a control area to compare the treatment results to. “This was a retrospective case series,” Dr. Kream replied. “I’d like to see more elegant studies in the future, with a control [area],” she said.
Dr. Kream reported having no financial disclosures, Dr. Boen has no disclosures, and Dr. Wu has conducted research for many pharmaceutical and device companies. Dr. Wolkerstorfer disclosed that he has received grant or research funding from Lumenis, Novartis, and Avita Medical, and is an advisory board member for Incyte.
SAN DIEGO – , results from a small retrospective case series suggest.
“Postinflammatory hyperpigmentation is a leading chief of complaint of many skin of color persons seeking a dermatologist,” Elizabeth J. Kream, MD, told this news organization in advance of the annual conference of American Society for Laser Medicine and Surgery. “I describe PIH to patients as the ‘ashes after a fire is extinguished.’ It’s the stubborn brown to gray/black spots that persist after conditions like acne and folliculitis, but it can be caused by any insult to the skin including external injury. In fact, there’s a risk of inciting PIH with lasers and energy-based devices and this risk is greater in skin of color given the greater melanin content. Unfortunately, we see patients present after visiting a med spa who were treated with the wrong devices and/or the wrong settings and they have disfiguring scarring and/or dyspigmentation.”
During an abstract session at the meeting, Dr. Kream, a dermatology resident at the University of Illinois at Chicago, discussed three patients with recalcitrant PIH and Fitzpatrick skin phototype V and VI who were treated in San Diego with a combination of topical and laser therapies. She presented the case series on behalf of coauthors Monica Boen, MD and Douglas C. Wu, MD, dermatologists who practice in San Diego.
The first patient was a 37-year-old Black female who presented for evaluation of longstanding hyperpigmentation on the face and neck determined to be PIH secondary to folliculitis on the chin and neck. She was started on 8% hydroquinone with kojic acid daily and received four treatments spaced 4-8 weeks apart with the 1,927-nm fractional nonablative diode laser. Laser settings were 5 mJ pulse energy and 5% coverage after eight passes. Triamcinolone 0.1% ointment was applied immediately after treatment and for 3 days following treatment, and the “patient experienced near complete resolution of PIH with no unexpected adverse events,” Dr. Kream said.
The second patient was a 20-year-old Black male who presented with a 3-month history of facial hyperpigmentation after suffering a laser-induced injury. He was started on a non-hydroquinone topical lightening agent and received five treatments spaced 2 weeks apart with a 1,927-nm fractional nonablative diode laser. The laser settings were 5 mJ pulse energy and 5% coverage after eight passes. The patient experienced 80%-90% resolution of his PIH with no unexpected adverse reactions.
The third patient in the series was a 39-year-old Black male who presented with a 6-month history of hyperpigmentation on his right shin and calf, secondary to minor occupational-related trauma. Treatment was initiated with a fractional 1,064-nm picosecond laser. The laser settings were 2.1 mJ per microbeam microwave pulse energy and a 450 picosecond pulse duration delivered at 2 Hz through a holographic beam splitter with a 6 x 6–mm spot size containing 101 microbeams, for an estimated coverage of 4% per pulse. Four passes were performed for each area. The endpoint was a mild erythema to several treated areas a few minutes following laser treatment. Postoperative care consisted of applying a non-hydroquinone topical lightening agent twice daily to the affected area for 1 month. Near-complete resolution of the PIH was achieved, with no unexpected adverse reactions.
“In our clinical experience, PIH can be treated with the combination of topical skin lighteners and low density, low fluence laser therapy in almost all skin types,” Dr. Kream said. “The rationale behind this combination is to treat and remove existing pigment with the laser therapy while minimizing and preventing any pigmentary recurrence with diligent topical therapy and photoprotection.”
It is important to identify the cause of PIH “because some cases are trickier than others,” such as a lichenoid process that deposits pigment “a little bit deeper into the dermis,” she said. “When selecting an appropriate laser modality for the treatment of PIH in skin types V and VI, it’s especially important to consider the mechanism of action, depth of penetration, degree of tissue damage, and the extent of disruption to the dermal-epidermal junction.”
Following the presentation, one of the session moderators, Albert Wolkerstorfer, MD, PhD, a dermatologist at Amsterdam University Medical Center, the Netherlands, emphasized the importance of proper patient selection for laser treatment of PIH. “Not every patient with PIH is adapted to treatment with the laser,” Dr. Wolkerstorfer said. “I think it’s also important to choose stable PIH, meaning you often see patients with an underlying disorder who want to get rid of the pigment. They often believe that the laser is the solution, but it often isn’t.”
During a question-and-answer session, a meeting attendee pointed out that the study lacked a control area to compare the treatment results to. “This was a retrospective case series,” Dr. Kream replied. “I’d like to see more elegant studies in the future, with a control [area],” she said.
Dr. Kream reported having no financial disclosures, Dr. Boen has no disclosures, and Dr. Wu has conducted research for many pharmaceutical and device companies. Dr. Wolkerstorfer disclosed that he has received grant or research funding from Lumenis, Novartis, and Avita Medical, and is an advisory board member for Incyte.
SAN DIEGO – , results from a small retrospective case series suggest.
“Postinflammatory hyperpigmentation is a leading chief of complaint of many skin of color persons seeking a dermatologist,” Elizabeth J. Kream, MD, told this news organization in advance of the annual conference of American Society for Laser Medicine and Surgery. “I describe PIH to patients as the ‘ashes after a fire is extinguished.’ It’s the stubborn brown to gray/black spots that persist after conditions like acne and folliculitis, but it can be caused by any insult to the skin including external injury. In fact, there’s a risk of inciting PIH with lasers and energy-based devices and this risk is greater in skin of color given the greater melanin content. Unfortunately, we see patients present after visiting a med spa who were treated with the wrong devices and/or the wrong settings and they have disfiguring scarring and/or dyspigmentation.”
During an abstract session at the meeting, Dr. Kream, a dermatology resident at the University of Illinois at Chicago, discussed three patients with recalcitrant PIH and Fitzpatrick skin phototype V and VI who were treated in San Diego with a combination of topical and laser therapies. She presented the case series on behalf of coauthors Monica Boen, MD and Douglas C. Wu, MD, dermatologists who practice in San Diego.
The first patient was a 37-year-old Black female who presented for evaluation of longstanding hyperpigmentation on the face and neck determined to be PIH secondary to folliculitis on the chin and neck. She was started on 8% hydroquinone with kojic acid daily and received four treatments spaced 4-8 weeks apart with the 1,927-nm fractional nonablative diode laser. Laser settings were 5 mJ pulse energy and 5% coverage after eight passes. Triamcinolone 0.1% ointment was applied immediately after treatment and for 3 days following treatment, and the “patient experienced near complete resolution of PIH with no unexpected adverse events,” Dr. Kream said.
The second patient was a 20-year-old Black male who presented with a 3-month history of facial hyperpigmentation after suffering a laser-induced injury. He was started on a non-hydroquinone topical lightening agent and received five treatments spaced 2 weeks apart with a 1,927-nm fractional nonablative diode laser. The laser settings were 5 mJ pulse energy and 5% coverage after eight passes. The patient experienced 80%-90% resolution of his PIH with no unexpected adverse reactions.
The third patient in the series was a 39-year-old Black male who presented with a 6-month history of hyperpigmentation on his right shin and calf, secondary to minor occupational-related trauma. Treatment was initiated with a fractional 1,064-nm picosecond laser. The laser settings were 2.1 mJ per microbeam microwave pulse energy and a 450 picosecond pulse duration delivered at 2 Hz through a holographic beam splitter with a 6 x 6–mm spot size containing 101 microbeams, for an estimated coverage of 4% per pulse. Four passes were performed for each area. The endpoint was a mild erythema to several treated areas a few minutes following laser treatment. Postoperative care consisted of applying a non-hydroquinone topical lightening agent twice daily to the affected area for 1 month. Near-complete resolution of the PIH was achieved, with no unexpected adverse reactions.
“In our clinical experience, PIH can be treated with the combination of topical skin lighteners and low density, low fluence laser therapy in almost all skin types,” Dr. Kream said. “The rationale behind this combination is to treat and remove existing pigment with the laser therapy while minimizing and preventing any pigmentary recurrence with diligent topical therapy and photoprotection.”
It is important to identify the cause of PIH “because some cases are trickier than others,” such as a lichenoid process that deposits pigment “a little bit deeper into the dermis,” she said. “When selecting an appropriate laser modality for the treatment of PIH in skin types V and VI, it’s especially important to consider the mechanism of action, depth of penetration, degree of tissue damage, and the extent of disruption to the dermal-epidermal junction.”
Following the presentation, one of the session moderators, Albert Wolkerstorfer, MD, PhD, a dermatologist at Amsterdam University Medical Center, the Netherlands, emphasized the importance of proper patient selection for laser treatment of PIH. “Not every patient with PIH is adapted to treatment with the laser,” Dr. Wolkerstorfer said. “I think it’s also important to choose stable PIH, meaning you often see patients with an underlying disorder who want to get rid of the pigment. They often believe that the laser is the solution, but it often isn’t.”
During a question-and-answer session, a meeting attendee pointed out that the study lacked a control area to compare the treatment results to. “This was a retrospective case series,” Dr. Kream replied. “I’d like to see more elegant studies in the future, with a control [area],” she said.
Dr. Kream reported having no financial disclosures, Dr. Boen has no disclosures, and Dr. Wu has conducted research for many pharmaceutical and device companies. Dr. Wolkerstorfer disclosed that he has received grant or research funding from Lumenis, Novartis, and Avita Medical, and is an advisory board member for Incyte.
AT ASLMS 2022
Upadacitinib earns FDA approval for ankylosing spondylitis
The Food and Drug Administration has approved upadacitinib (Rinvoq) as an oral treatment for active ankylosing spondylitis in adults, its manufacturer AbbVie announced April 29.
Upadacitinib, a selective and reversible Janus kinase inhibitor, is the second drug in its class to be FDA approved for ankylosing spondylitis, after tofacitinib (Xeljanz) in December.
Upadacitinib is now indicated for patients with active ankylosing spondylitis (AS) who have had an insufficient response or intolerance with one or more tumor necrosis factor (TNF) blockers. Upadacitinib is already approved by the FDA for adults with active psoriatic arthritis, moderately to severely active rheumatoid arthritis, and moderately to severely active ulcerative colitis who have had an insufficient response or intolerance with one or more TNF inhibitors. It also has been approved for adults and pediatric patients 12 years of age and older with refractory, moderate to severe atopic dermatitis.
The European Medicines Agency gave marketing approval for upadacitinib in adults with active AS in January 2021.
Two main clinical studies form the basis for the FDA’s approval decision. The phase 3 SELECT-AXIS 2 clinical trial involved patients with an inadequate response or intolerance to one or two biologic disease-modifying antirheumatic drugs (bDMARDs). A total of 44.5% patients with AS who were randomly assigned to upadacitinib 15 mg once daily met the primary endpoint of at least 40% improvement in Assessment in Spondyloarthritis International Society response criteria (ASAS 40) at 14 weeks, compared against 18.2% with placebo.
The second study, the phase 2/3 SELECT-AXIS 1 clinical trial, tested upadacitinib in patients who had never taken bDMARDs and had an inadequate response or intolerance to at least two NSAIDs. In this study, significantly more patients randomly assigned to 15 mg upadacitinib achieved ASAS 40 at 14 weeks, compared with placebo (51% vs. 26%).
Patients randomly assigned to upadacitinib also showed significant improvements in signs and symptoms of AS, as well as improvements in physical function and disease activity, compared with placebo, after 14 weeks. The safety profile for patients with AS treated with upadacitinib was similar to that seen in studies of patients with rheumatoid arthritis or psoriatic arthritis. Potential severe side effects include increased risk for death in patients aged 50 years and older with at least one cardiovascular risk factor; increased risk of serious infections, such as tuberculosis; and increased risk of certain cancers, according to the company statement.
Read the complete prescribing information here.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved upadacitinib (Rinvoq) as an oral treatment for active ankylosing spondylitis in adults, its manufacturer AbbVie announced April 29.
Upadacitinib, a selective and reversible Janus kinase inhibitor, is the second drug in its class to be FDA approved for ankylosing spondylitis, after tofacitinib (Xeljanz) in December.
Upadacitinib is now indicated for patients with active ankylosing spondylitis (AS) who have had an insufficient response or intolerance with one or more tumor necrosis factor (TNF) blockers. Upadacitinib is already approved by the FDA for adults with active psoriatic arthritis, moderately to severely active rheumatoid arthritis, and moderately to severely active ulcerative colitis who have had an insufficient response or intolerance with one or more TNF inhibitors. It also has been approved for adults and pediatric patients 12 years of age and older with refractory, moderate to severe atopic dermatitis.
The European Medicines Agency gave marketing approval for upadacitinib in adults with active AS in January 2021.
Two main clinical studies form the basis for the FDA’s approval decision. The phase 3 SELECT-AXIS 2 clinical trial involved patients with an inadequate response or intolerance to one or two biologic disease-modifying antirheumatic drugs (bDMARDs). A total of 44.5% patients with AS who were randomly assigned to upadacitinib 15 mg once daily met the primary endpoint of at least 40% improvement in Assessment in Spondyloarthritis International Society response criteria (ASAS 40) at 14 weeks, compared against 18.2% with placebo.
The second study, the phase 2/3 SELECT-AXIS 1 clinical trial, tested upadacitinib in patients who had never taken bDMARDs and had an inadequate response or intolerance to at least two NSAIDs. In this study, significantly more patients randomly assigned to 15 mg upadacitinib achieved ASAS 40 at 14 weeks, compared with placebo (51% vs. 26%).
Patients randomly assigned to upadacitinib also showed significant improvements in signs and symptoms of AS, as well as improvements in physical function and disease activity, compared with placebo, after 14 weeks. The safety profile for patients with AS treated with upadacitinib was similar to that seen in studies of patients with rheumatoid arthritis or psoriatic arthritis. Potential severe side effects include increased risk for death in patients aged 50 years and older with at least one cardiovascular risk factor; increased risk of serious infections, such as tuberculosis; and increased risk of certain cancers, according to the company statement.
Read the complete prescribing information here.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved upadacitinib (Rinvoq) as an oral treatment for active ankylosing spondylitis in adults, its manufacturer AbbVie announced April 29.
Upadacitinib, a selective and reversible Janus kinase inhibitor, is the second drug in its class to be FDA approved for ankylosing spondylitis, after tofacitinib (Xeljanz) in December.
Upadacitinib is now indicated for patients with active ankylosing spondylitis (AS) who have had an insufficient response or intolerance with one or more tumor necrosis factor (TNF) blockers. Upadacitinib is already approved by the FDA for adults with active psoriatic arthritis, moderately to severely active rheumatoid arthritis, and moderately to severely active ulcerative colitis who have had an insufficient response or intolerance with one or more TNF inhibitors. It also has been approved for adults and pediatric patients 12 years of age and older with refractory, moderate to severe atopic dermatitis.
The European Medicines Agency gave marketing approval for upadacitinib in adults with active AS in January 2021.
Two main clinical studies form the basis for the FDA’s approval decision. The phase 3 SELECT-AXIS 2 clinical trial involved patients with an inadequate response or intolerance to one or two biologic disease-modifying antirheumatic drugs (bDMARDs). A total of 44.5% patients with AS who were randomly assigned to upadacitinib 15 mg once daily met the primary endpoint of at least 40% improvement in Assessment in Spondyloarthritis International Society response criteria (ASAS 40) at 14 weeks, compared against 18.2% with placebo.
The second study, the phase 2/3 SELECT-AXIS 1 clinical trial, tested upadacitinib in patients who had never taken bDMARDs and had an inadequate response or intolerance to at least two NSAIDs. In this study, significantly more patients randomly assigned to 15 mg upadacitinib achieved ASAS 40 at 14 weeks, compared with placebo (51% vs. 26%).
Patients randomly assigned to upadacitinib also showed significant improvements in signs and symptoms of AS, as well as improvements in physical function and disease activity, compared with placebo, after 14 weeks. The safety profile for patients with AS treated with upadacitinib was similar to that seen in studies of patients with rheumatoid arthritis or psoriatic arthritis. Potential severe side effects include increased risk for death in patients aged 50 years and older with at least one cardiovascular risk factor; increased risk of serious infections, such as tuberculosis; and increased risk of certain cancers, according to the company statement.
Read the complete prescribing information here.
A version of this article first appeared on Medscape.com.