User login
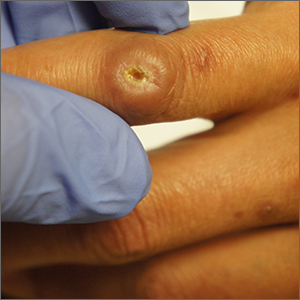
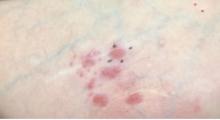
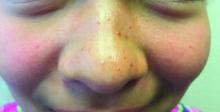
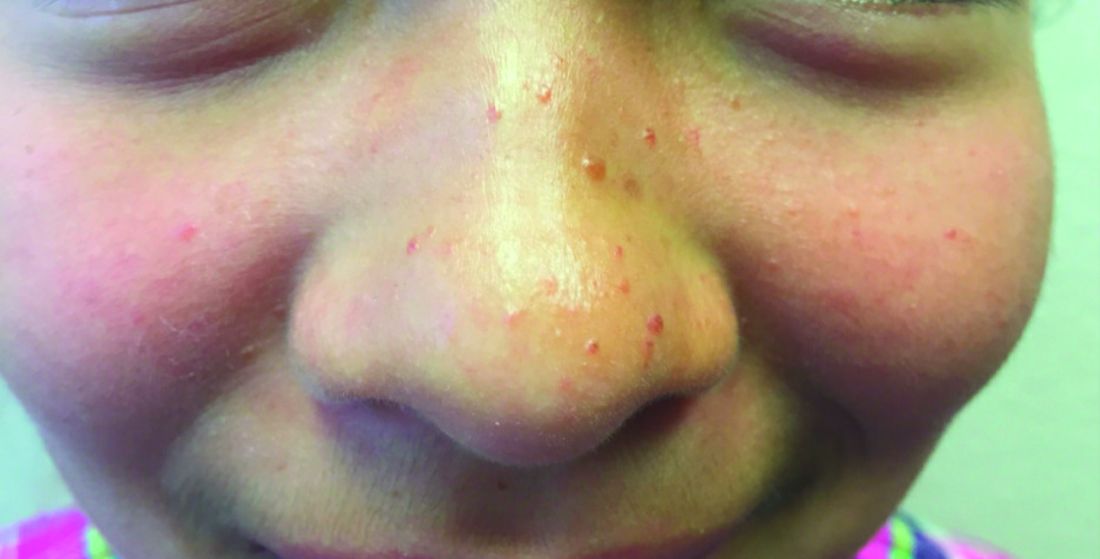
Ulcer on knuckle
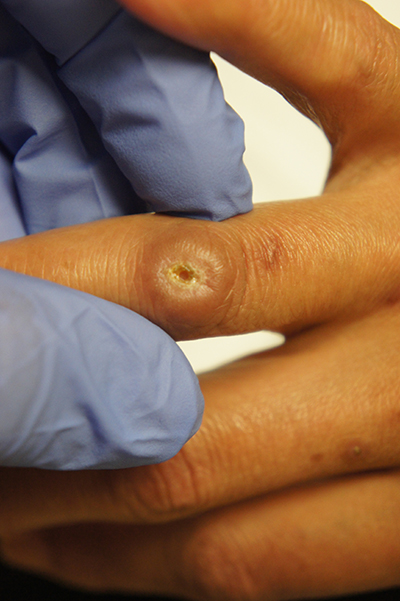
Since the papules were worrisome for vasculitis, 2 punch biopsies were performed on smaller, younger lesions on the hand and 1 was submitted for direct immunofluorescence. Findings revealed a leukocytoclastic vasculitis (LCV) with prominent immunoglobulin A (IgA) deposits around the vessel wall. The clinical and pathologic findings were consistent with a rare fibrosing LCV called erythema elevatum diutinum (EED). The practice of sampling nonblanching purpura or papules for both standard pathology (hematoxylin and eosin) and direct immunofluorescence can facilitate the diagnosis of unusual conditions that may occur only a few times in one’s career.
EED is rare, chronic, and may be associated with IgA gammopathy, IgA antineutrophil cytoplasmic antibodies, recent streptococcal or HIV infections, or myelodysplastic syndrome. A robust work-up to identify whether any of these factors are at work is critical.1 Distinct from other vasculitides, EED forms granulation tissue that becomes reinjured.
This patient was found to have an IgA monoclonal gammopathy that was monitored by Hematology. After checking her glucose-6-phosphate dehydrogenase activity, she was treated with oral dapsone 25 mg/d. Dapsone was titrated up to 100 mg/d, which improved her symptoms considerably and the ulcerated papule was surgically revised and closed. EED can last for years and ultimately clear or can persist indefinitely.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Sandhu JK, Albrecht J, Agnihotri G, et al. Erythema elevatum et diutinum as a systemic disease. Clin Dermatol. 2019;37:679-683. doi: 10.1016/j.clindermatol.2019.07.028

Since the papules were worrisome for vasculitis, 2 punch biopsies were performed on smaller, younger lesions on the hand and 1 was submitted for direct immunofluorescence. Findings revealed a leukocytoclastic vasculitis (LCV) with prominent immunoglobulin A (IgA) deposits around the vessel wall. The clinical and pathologic findings were consistent with a rare fibrosing LCV called erythema elevatum diutinum (EED). The practice of sampling nonblanching purpura or papules for both standard pathology (hematoxylin and eosin) and direct immunofluorescence can facilitate the diagnosis of unusual conditions that may occur only a few times in one’s career.
EED is rare, chronic, and may be associated with IgA gammopathy, IgA antineutrophil cytoplasmic antibodies, recent streptococcal or HIV infections, or myelodysplastic syndrome. A robust work-up to identify whether any of these factors are at work is critical.1 Distinct from other vasculitides, EED forms granulation tissue that becomes reinjured.
This patient was found to have an IgA monoclonal gammopathy that was monitored by Hematology. After checking her glucose-6-phosphate dehydrogenase activity, she was treated with oral dapsone 25 mg/d. Dapsone was titrated up to 100 mg/d, which improved her symptoms considerably and the ulcerated papule was surgically revised and closed. EED can last for years and ultimately clear or can persist indefinitely.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

Since the papules were worrisome for vasculitis, 2 punch biopsies were performed on smaller, younger lesions on the hand and 1 was submitted for direct immunofluorescence. Findings revealed a leukocytoclastic vasculitis (LCV) with prominent immunoglobulin A (IgA) deposits around the vessel wall. The clinical and pathologic findings were consistent with a rare fibrosing LCV called erythema elevatum diutinum (EED). The practice of sampling nonblanching purpura or papules for both standard pathology (hematoxylin and eosin) and direct immunofluorescence can facilitate the diagnosis of unusual conditions that may occur only a few times in one’s career.
EED is rare, chronic, and may be associated with IgA gammopathy, IgA antineutrophil cytoplasmic antibodies, recent streptococcal or HIV infections, or myelodysplastic syndrome. A robust work-up to identify whether any of these factors are at work is critical.1 Distinct from other vasculitides, EED forms granulation tissue that becomes reinjured.
This patient was found to have an IgA monoclonal gammopathy that was monitored by Hematology. After checking her glucose-6-phosphate dehydrogenase activity, she was treated with oral dapsone 25 mg/d. Dapsone was titrated up to 100 mg/d, which improved her symptoms considerably and the ulcerated papule was surgically revised and closed. EED can last for years and ultimately clear or can persist indefinitely.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Sandhu JK, Albrecht J, Agnihotri G, et al. Erythema elevatum et diutinum as a systemic disease. Clin Dermatol. 2019;37:679-683. doi: 10.1016/j.clindermatol.2019.07.028
1. Sandhu JK, Albrecht J, Agnihotri G, et al. Erythema elevatum et diutinum as a systemic disease. Clin Dermatol. 2019;37:679-683. doi: 10.1016/j.clindermatol.2019.07.028
Manufacturer announces FDA approval for molluscum treatment delayed
Pharmaceuticals, which is developing the product.
VP-102 is a proprietary drug-device combination of cantharidin 0.7% administered through a single-use precision applicator, which has been evaluated in phase 3 studies of patients with molluscum aged 2 years and older. It features a visualization agent so the person applying the drug can see which lesions have been treated. It also contains a bittering agent to mitigate oral ingestion by children.
According to a press release from Verrica, the only deficiency listed in the FDA’s complete response letter stemmed from a general reinspection of Sterling Pharmaceuticals Services, which manufactures Verrica’s bulk solution drug product. Although none of the issues identified by the FDA during the reinspection were specific to the manufacturing of VP-102, FDA policy prevents approval of a new drug application when a contract manufacturing organization has an unresolved classification status or is placed on “official action indicated” status.
According to the press release, Verrica will “continue to work collaboratively” with the FDA to bring VP-102 to the market as soon as possible. The company has completed phase 2 studies of VP-102 for the treatment of common warts and for the treatment of external genital warts, the release said.
A version of this article first appeared on Medscape.com.
Pharmaceuticals, which is developing the product.
VP-102 is a proprietary drug-device combination of cantharidin 0.7% administered through a single-use precision applicator, which has been evaluated in phase 3 studies of patients with molluscum aged 2 years and older. It features a visualization agent so the person applying the drug can see which lesions have been treated. It also contains a bittering agent to mitigate oral ingestion by children.
According to a press release from Verrica, the only deficiency listed in the FDA’s complete response letter stemmed from a general reinspection of Sterling Pharmaceuticals Services, which manufactures Verrica’s bulk solution drug product. Although none of the issues identified by the FDA during the reinspection were specific to the manufacturing of VP-102, FDA policy prevents approval of a new drug application when a contract manufacturing organization has an unresolved classification status or is placed on “official action indicated” status.
According to the press release, Verrica will “continue to work collaboratively” with the FDA to bring VP-102 to the market as soon as possible. The company has completed phase 2 studies of VP-102 for the treatment of common warts and for the treatment of external genital warts, the release said.
A version of this article first appeared on Medscape.com.
Pharmaceuticals, which is developing the product.
VP-102 is a proprietary drug-device combination of cantharidin 0.7% administered through a single-use precision applicator, which has been evaluated in phase 3 studies of patients with molluscum aged 2 years and older. It features a visualization agent so the person applying the drug can see which lesions have been treated. It also contains a bittering agent to mitigate oral ingestion by children.
According to a press release from Verrica, the only deficiency listed in the FDA’s complete response letter stemmed from a general reinspection of Sterling Pharmaceuticals Services, which manufactures Verrica’s bulk solution drug product. Although none of the issues identified by the FDA during the reinspection were specific to the manufacturing of VP-102, FDA policy prevents approval of a new drug application when a contract manufacturing organization has an unresolved classification status or is placed on “official action indicated” status.
According to the press release, Verrica will “continue to work collaboratively” with the FDA to bring VP-102 to the market as soon as possible. The company has completed phase 2 studies of VP-102 for the treatment of common warts and for the treatment of external genital warts, the release said.
A version of this article first appeared on Medscape.com.
FDA approves topical tapinarof for plaque psoriasis
The the manufacturer announced.
Tapinarof is an aryl hydrocarbon receptor agonist and is the first FDA-approved steroid-free topical medication in this class, according to a press release from the manufacturer, Dermavant.
Approval was based on results of three studies in a phase 3 clinical trial program (PSOARING 1, PSOARING 2), and an open-label extension study, (PSOARING 3), the company release said. In PSOARING 1 and 2, approximately 1,000 adults aged 18-75 years (median age, 51 years) with plaque psoriasis were randomized to once-daily topical tapinarof or placebo for up to 12 weeks; 85% were White and 57% were men. The study findings were published in the New England Journal of Medicine in December 2021.
The primary endpoint for both trials was the proportion of patients who achieved Physician Global Assessment (PGA) scores score of “clear” (0) or “almost clear” (1) and improvement of at least two grades from baseline.
After 12 weeks, 36% of the patients in PSOARING 1 and 40% in PSOARING 2 who received tapinarof met the primary outcome, compared with 6% of patients on placebo (P < .001 for both studies). Of these, a total of 73 patients from both studies who achieved PGA scores of 0 were entered in PSOARING 3, a 40-week open-label extension study, in which they stopped tapinarof treatment and retained PGA scores of 0 or 1 for approximately 4 months off treatment. An additional 312 patients who were enrolled in the PSOARING 3 extension study achieved PGA scores of 0 at least once during the study period, with “remittive” effects lasting a mean of 130 days off of treatment.
In addition, patients who received tapinarof in the PSOARING 1 and 2 studies showed significant improvement from baseline, compared with patients on placebo, across a range of secondary endpoints including a 75% or greater improvement in Psoriasis Area and Severity Index score (PASI 75).
In PSOARING 1, and 2, respectively, 36.1% and 47.6% of those on tapinarof achieved a PASI 75 response at week 12, compared with 10.2% and 6.9% of those on the vehicle (P < .001 for both).
Across all three studies, the majority adverse events were mild to moderate, and limited to the application site.
The most common adverse events reported by patients in the tapinarof groups were folliculitis, nasopharyngitis, and contact dermatitis. Headaches were more common among those treated with tapinarof than those on vehicle in the studies (3.8% vs. 2.4% in PSOARING 1, and 3.8% vs. 0.6% in PSOARING 2), leading to only three treatment discontinuations.
At the end of the PSOARING 3 study (at either week 40 or early termination), 599 participants responded to satisfaction questionnaires. Of these, 83.6% said they were satisfied with the results of tapinarof treatment, and 81.7% said it was more effective than previous topical treatments they had used, according to the company’s release.
Tapinarof cream can be used on all areas of the body, including the face, skin folds, neck, genitalia, anal crux, inflammatory areas, and axillae, according to the company release.
Full prescribing information is available here.
The the manufacturer announced.
Tapinarof is an aryl hydrocarbon receptor agonist and is the first FDA-approved steroid-free topical medication in this class, according to a press release from the manufacturer, Dermavant.
Approval was based on results of three studies in a phase 3 clinical trial program (PSOARING 1, PSOARING 2), and an open-label extension study, (PSOARING 3), the company release said. In PSOARING 1 and 2, approximately 1,000 adults aged 18-75 years (median age, 51 years) with plaque psoriasis were randomized to once-daily topical tapinarof or placebo for up to 12 weeks; 85% were White and 57% were men. The study findings were published in the New England Journal of Medicine in December 2021.
The primary endpoint for both trials was the proportion of patients who achieved Physician Global Assessment (PGA) scores score of “clear” (0) or “almost clear” (1) and improvement of at least two grades from baseline.
After 12 weeks, 36% of the patients in PSOARING 1 and 40% in PSOARING 2 who received tapinarof met the primary outcome, compared with 6% of patients on placebo (P < .001 for both studies). Of these, a total of 73 patients from both studies who achieved PGA scores of 0 were entered in PSOARING 3, a 40-week open-label extension study, in which they stopped tapinarof treatment and retained PGA scores of 0 or 1 for approximately 4 months off treatment. An additional 312 patients who were enrolled in the PSOARING 3 extension study achieved PGA scores of 0 at least once during the study period, with “remittive” effects lasting a mean of 130 days off of treatment.
In addition, patients who received tapinarof in the PSOARING 1 and 2 studies showed significant improvement from baseline, compared with patients on placebo, across a range of secondary endpoints including a 75% or greater improvement in Psoriasis Area and Severity Index score (PASI 75).
In PSOARING 1, and 2, respectively, 36.1% and 47.6% of those on tapinarof achieved a PASI 75 response at week 12, compared with 10.2% and 6.9% of those on the vehicle (P < .001 for both).
Across all three studies, the majority adverse events were mild to moderate, and limited to the application site.
The most common adverse events reported by patients in the tapinarof groups were folliculitis, nasopharyngitis, and contact dermatitis. Headaches were more common among those treated with tapinarof than those on vehicle in the studies (3.8% vs. 2.4% in PSOARING 1, and 3.8% vs. 0.6% in PSOARING 2), leading to only three treatment discontinuations.
At the end of the PSOARING 3 study (at either week 40 or early termination), 599 participants responded to satisfaction questionnaires. Of these, 83.6% said they were satisfied with the results of tapinarof treatment, and 81.7% said it was more effective than previous topical treatments they had used, according to the company’s release.
Tapinarof cream can be used on all areas of the body, including the face, skin folds, neck, genitalia, anal crux, inflammatory areas, and axillae, according to the company release.
Full prescribing information is available here.
The the manufacturer announced.
Tapinarof is an aryl hydrocarbon receptor agonist and is the first FDA-approved steroid-free topical medication in this class, according to a press release from the manufacturer, Dermavant.
Approval was based on results of three studies in a phase 3 clinical trial program (PSOARING 1, PSOARING 2), and an open-label extension study, (PSOARING 3), the company release said. In PSOARING 1 and 2, approximately 1,000 adults aged 18-75 years (median age, 51 years) with plaque psoriasis were randomized to once-daily topical tapinarof or placebo for up to 12 weeks; 85% were White and 57% were men. The study findings were published in the New England Journal of Medicine in December 2021.
The primary endpoint for both trials was the proportion of patients who achieved Physician Global Assessment (PGA) scores score of “clear” (0) or “almost clear” (1) and improvement of at least two grades from baseline.
After 12 weeks, 36% of the patients in PSOARING 1 and 40% in PSOARING 2 who received tapinarof met the primary outcome, compared with 6% of patients on placebo (P < .001 for both studies). Of these, a total of 73 patients from both studies who achieved PGA scores of 0 were entered in PSOARING 3, a 40-week open-label extension study, in which they stopped tapinarof treatment and retained PGA scores of 0 or 1 for approximately 4 months off treatment. An additional 312 patients who were enrolled in the PSOARING 3 extension study achieved PGA scores of 0 at least once during the study period, with “remittive” effects lasting a mean of 130 days off of treatment.
In addition, patients who received tapinarof in the PSOARING 1 and 2 studies showed significant improvement from baseline, compared with patients on placebo, across a range of secondary endpoints including a 75% or greater improvement in Psoriasis Area and Severity Index score (PASI 75).
In PSOARING 1, and 2, respectively, 36.1% and 47.6% of those on tapinarof achieved a PASI 75 response at week 12, compared with 10.2% and 6.9% of those on the vehicle (P < .001 for both).
Across all three studies, the majority adverse events were mild to moderate, and limited to the application site.
The most common adverse events reported by patients in the tapinarof groups were folliculitis, nasopharyngitis, and contact dermatitis. Headaches were more common among those treated with tapinarof than those on vehicle in the studies (3.8% vs. 2.4% in PSOARING 1, and 3.8% vs. 0.6% in PSOARING 2), leading to only three treatment discontinuations.
At the end of the PSOARING 3 study (at either week 40 or early termination), 599 participants responded to satisfaction questionnaires. Of these, 83.6% said they were satisfied with the results of tapinarof treatment, and 81.7% said it was more effective than previous topical treatments they had used, according to the company’s release.
Tapinarof cream can be used on all areas of the body, including the face, skin folds, neck, genitalia, anal crux, inflammatory areas, and axillae, according to the company release.
Full prescribing information is available here.
European committee recommends approval of baricitinib for severe alopecia areata
The European Medicines Agency’s ( (AA).
The development, which was announced in a May 20, 2022, press release from the manufacturer, Eli Lilly and Incyte, marks the first step toward European regulatory approval of baricitinib (Olumiant) for patients with severe AA, and it is now referred to the European Commission for final action. A decision is expected within the next 2 months.
The committee based its positive opinion on the results of the phase 3 BRAVE-AA1 and BRAVE-AA2 trials, recently published in the New England Journal of Medicine, which evaluated the efficacy and safety of baricitinib in 1,200 patients with severe AA, according to the press release. The primary endpoint was the proportion of patients achieving a Severity of Alopecia Tool (SALT) score of ≤20 at week 36. In both studies, 1 out of 3 patients treated with baricitinib 4-mg achieved 80% or more scalp hair coverage, compared with 1 out of 20 patients and 1 out of 50 patients taking placebo in BRAVE-AA1 and BRAVE-AA2, respectively (P ≤ .001 for all comparisons to placebo).
According to safety profile information from the phase 3 BRAVE-AA clinical program, few patients discontinued treatment because of adverse events (2.6% or less across both studies), and most treatment-emergent adverse events were mild or moderate in severity.
In February 2022, the Food and Drug Administration granted priority review for baricitinib in adults with severe AA. Lilly expects additional regulatory decisions in the United States and Japan in 2022.
Baricitinib is approved in the United States as a treatment for adults with moderate to severe rheumatoid arthritis. Prescribing information can be viewed here.
The European Medicines Agency’s ( (AA).
The development, which was announced in a May 20, 2022, press release from the manufacturer, Eli Lilly and Incyte, marks the first step toward European regulatory approval of baricitinib (Olumiant) for patients with severe AA, and it is now referred to the European Commission for final action. A decision is expected within the next 2 months.
The committee based its positive opinion on the results of the phase 3 BRAVE-AA1 and BRAVE-AA2 trials, recently published in the New England Journal of Medicine, which evaluated the efficacy and safety of baricitinib in 1,200 patients with severe AA, according to the press release. The primary endpoint was the proportion of patients achieving a Severity of Alopecia Tool (SALT) score of ≤20 at week 36. In both studies, 1 out of 3 patients treated with baricitinib 4-mg achieved 80% or more scalp hair coverage, compared with 1 out of 20 patients and 1 out of 50 patients taking placebo in BRAVE-AA1 and BRAVE-AA2, respectively (P ≤ .001 for all comparisons to placebo).
According to safety profile information from the phase 3 BRAVE-AA clinical program, few patients discontinued treatment because of adverse events (2.6% or less across both studies), and most treatment-emergent adverse events were mild or moderate in severity.
In February 2022, the Food and Drug Administration granted priority review for baricitinib in adults with severe AA. Lilly expects additional regulatory decisions in the United States and Japan in 2022.
Baricitinib is approved in the United States as a treatment for adults with moderate to severe rheumatoid arthritis. Prescribing information can be viewed here.
The European Medicines Agency’s ( (AA).
The development, which was announced in a May 20, 2022, press release from the manufacturer, Eli Lilly and Incyte, marks the first step toward European regulatory approval of baricitinib (Olumiant) for patients with severe AA, and it is now referred to the European Commission for final action. A decision is expected within the next 2 months.
The committee based its positive opinion on the results of the phase 3 BRAVE-AA1 and BRAVE-AA2 trials, recently published in the New England Journal of Medicine, which evaluated the efficacy and safety of baricitinib in 1,200 patients with severe AA, according to the press release. The primary endpoint was the proportion of patients achieving a Severity of Alopecia Tool (SALT) score of ≤20 at week 36. In both studies, 1 out of 3 patients treated with baricitinib 4-mg achieved 80% or more scalp hair coverage, compared with 1 out of 20 patients and 1 out of 50 patients taking placebo in BRAVE-AA1 and BRAVE-AA2, respectively (P ≤ .001 for all comparisons to placebo).
According to safety profile information from the phase 3 BRAVE-AA clinical program, few patients discontinued treatment because of adverse events (2.6% or less across both studies), and most treatment-emergent adverse events were mild or moderate in severity.
In February 2022, the Food and Drug Administration granted priority review for baricitinib in adults with severe AA. Lilly expects additional regulatory decisions in the United States and Japan in 2022.
Baricitinib is approved in the United States as a treatment for adults with moderate to severe rheumatoid arthritis. Prescribing information can be viewed here.
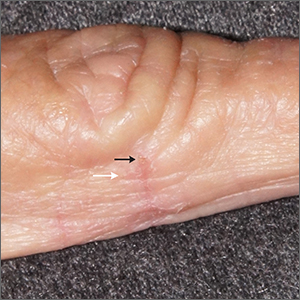
Itching at night
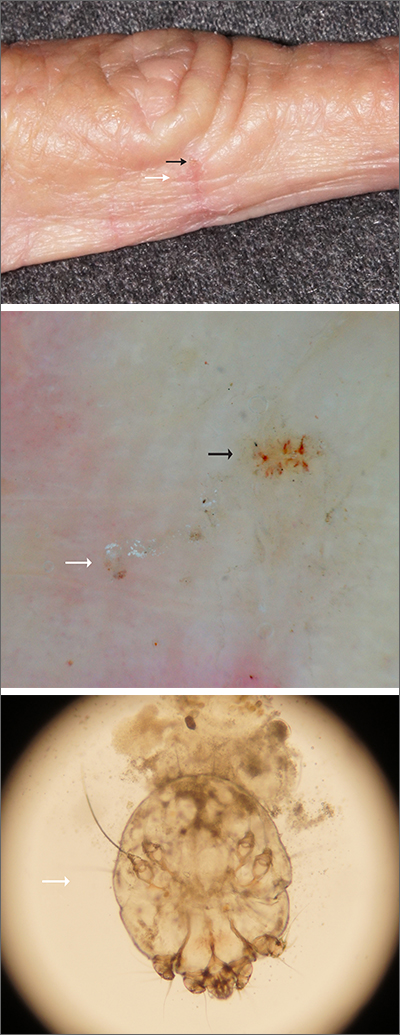
Microscopic evaluation of a dermoscopy-guided skin scraping revealed that this was a case of scabies.
Classically, patients with scabies have many excoriated papules or plaques on their hands, genitals, and trunk with itching so intense that their sleep is interrupted. However, scabies can also be diagnosed in patients who complain of itching but also have very subtle skin findings, such as a small dry patch or fissure (as in this case). Such subtle findings can be easily mistaken for mild hand dermatitis.
In the elderly, itching without a significant rash can arise from many causes. A short list includes dry skin, medications, kidney disease, liver disease, and of course, various dermatologic conditions. Dermoscopy is a sensitive and specific tool for investigating itching and areas of suspected infestation with mites.1 A mite appears as an oval on dermoscopy, but the most recognizable part is the head and front legs, which appear as a single gray triangle. The photo shows that the most erythematous area around a burrow (black arrows) is an excellent place to start when looking through the dermatoscope. In this case, an area of broken skin was connected to a haphazard tunnel that ultimately led to the mite (white arrows).
Scabies may be effectively treated with topical permethrin 5% applied over every inch of the body from the top of the neck to the tips of the toes.
This topical treatment is left on for at least 8 hours and reapplied a week later. Also, remember to take a careful history of close contacts so that others who are affected may receive treatment.
This patient was treated with permethrin, as was her adult son who lived at home with her and had similar itching. Permethrin comes in 60 g tubes, which is enough to treat 1 adult twice. After 6 weeks, all itching symptoms in the patient had cleared.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Dupuy A, Dehen L, Bourrat E, et al. Accuracy of standard dermoscopy for diagnosing scabies. J Am Acad Dermatol. 2007;56:53-62. doi: 10.1016/j.jaad.2006.07.025

Microscopic evaluation of a dermoscopy-guided skin scraping revealed that this was a case of scabies.
Classically, patients with scabies have many excoriated papules or plaques on their hands, genitals, and trunk with itching so intense that their sleep is interrupted. However, scabies can also be diagnosed in patients who complain of itching but also have very subtle skin findings, such as a small dry patch or fissure (as in this case). Such subtle findings can be easily mistaken for mild hand dermatitis.
In the elderly, itching without a significant rash can arise from many causes. A short list includes dry skin, medications, kidney disease, liver disease, and of course, various dermatologic conditions. Dermoscopy is a sensitive and specific tool for investigating itching and areas of suspected infestation with mites.1 A mite appears as an oval on dermoscopy, but the most recognizable part is the head and front legs, which appear as a single gray triangle. The photo shows that the most erythematous area around a burrow (black arrows) is an excellent place to start when looking through the dermatoscope. In this case, an area of broken skin was connected to a haphazard tunnel that ultimately led to the mite (white arrows).
Scabies may be effectively treated with topical permethrin 5% applied over every inch of the body from the top of the neck to the tips of the toes.
This topical treatment is left on for at least 8 hours and reapplied a week later. Also, remember to take a careful history of close contacts so that others who are affected may receive treatment.
This patient was treated with permethrin, as was her adult son who lived at home with her and had similar itching. Permethrin comes in 60 g tubes, which is enough to treat 1 adult twice. After 6 weeks, all itching symptoms in the patient had cleared.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).

Microscopic evaluation of a dermoscopy-guided skin scraping revealed that this was a case of scabies.
Classically, patients with scabies have many excoriated papules or plaques on their hands, genitals, and trunk with itching so intense that their sleep is interrupted. However, scabies can also be diagnosed in patients who complain of itching but also have very subtle skin findings, such as a small dry patch or fissure (as in this case). Such subtle findings can be easily mistaken for mild hand dermatitis.
In the elderly, itching without a significant rash can arise from many causes. A short list includes dry skin, medications, kidney disease, liver disease, and of course, various dermatologic conditions. Dermoscopy is a sensitive and specific tool for investigating itching and areas of suspected infestation with mites.1 A mite appears as an oval on dermoscopy, but the most recognizable part is the head and front legs, which appear as a single gray triangle. The photo shows that the most erythematous area around a burrow (black arrows) is an excellent place to start when looking through the dermatoscope. In this case, an area of broken skin was connected to a haphazard tunnel that ultimately led to the mite (white arrows).
Scabies may be effectively treated with topical permethrin 5% applied over every inch of the body from the top of the neck to the tips of the toes.
This topical treatment is left on for at least 8 hours and reapplied a week later. Also, remember to take a careful history of close contacts so that others who are affected may receive treatment.
This patient was treated with permethrin, as was her adult son who lived at home with her and had similar itching. Permethrin comes in 60 g tubes, which is enough to treat 1 adult twice. After 6 weeks, all itching symptoms in the patient had cleared.
Text courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. Photos courtesy of Jonathan Karnes, MD (copyright retained).
1. Dupuy A, Dehen L, Bourrat E, et al. Accuracy of standard dermoscopy for diagnosing scabies. J Am Acad Dermatol. 2007;56:53-62. doi: 10.1016/j.jaad.2006.07.025
1. Dupuy A, Dehen L, Bourrat E, et al. Accuracy of standard dermoscopy for diagnosing scabies. J Am Acad Dermatol. 2007;56:53-62. doi: 10.1016/j.jaad.2006.07.025
Lenabasum improved skin symptoms in dermatomyositis, but future is uncertain
An – some of it statistically significant – in a phase 2, double-blind, randomized, controlled study.
Patients taking lenabasum experienced greater reductions in the Cutaneous Dermatomyositis Disease Area and Severity Index (CDASI) – a validated outcome designed to assess inflammatory skin involvement in the rare autoimmune disease – and improvements in patient-reported and biomarker outcomes, compared with those on placebo, dermatologist Victoria P. Werth, MD, and coinvestigators reported.
And in a recently completed phase 3 trial, reported by the manufacturer, a subpopulation of patients with active skin disease and no active muscle disease again showed greater reductions in CDASI activity scores – a secondary outcome in the trial.
However, the phase 3 DETERMINE trial produced negative findings overall. It enrolled a more heterogeneous group of patients – including those with both muscle weakness and skin involvement – and its primary outcome measure was a broader composite measure, the Total Improvement Score. The trial failed to meet this primary endpoint, Corbus Pharmaceuticals, the developer of lenabasum, announced in a press release in June 2021.
The phase 3 results are “frustrating” for patients with symptomatic and refractory skin manifestations of dermatomyositis (DM), given the promising findings from the phase 2 trial and from an open-label extension study, said Dr. Werth, professor of dermatology and medicine, University of Pennsylvania, Philadelphia, and principal investigator and coprincipal investigator of the phase 2 and phase 3 studies, respectively.
Dr. Werth is scheduled to present the results from the phase 3 trial at the annual European Alliance of Associations for Rheumatology meeting in June.
“With lenabasum, we have a therapy that doesn’t work for every patient, but does work for quite a number of them,” Dr. Werth said in an interview. “It’s oral, it’s not really that immunosuppressing, and there aren’t many side effects. Right now, patients are often being managed with steroids ... we really need treatments that are not as toxic.”
Robert Spiera, MD, a rheumatologist who led trials of lenabasum for treatment of diffuse cutaneous systemic sclerosis (dcSSc), agreed. “The CB2 agonist strategy is appealing because it’s nonimmunosuppressing and has both anti-inflammatory and antifibrotic properties,” he said in an interview. “I wouldn’t want to give up on it ... especially [for patients] with scleroderma and dermatomyositis who are treated with substantial drugs that are associated with morbidity.”
Lenabasum, he said, has proven to be “incredibly safe, and incredibly safe in the long term.”
While the phase 2 trial of the drug for dcSSc showed clear benefit over placebo, the phase 3 trial did not meet its primary endpoint using the American College of Rheumatology Combined Response Index in Diffuse Cutaneous Systemic Sclerosis.
It allowed background immunosuppressant therapy to reflect real-world clinical practice, and “there was such a high response rate to [that therapy, largely mycophenolate] that there was little room to show benefit beyond that,” said Dr. Spiera, director of the vasculitis and scleroderma program, Hospital for Special Surgery, New York.
The drug led to more improvement in the small subset of participants who were not receiving background immunotherapy during the trial, he noted.
Corbus is currently “seeking a partnership to further explore the drug” for treatment in different subpopulations, according to a company spokesperson. Results of a phase 2 trial of lenabasum for the treatment of systemic lupus erythematosus – with a pain rating as the primary outcome measure – are expected soon.
Phase 2 findings
The single-center phase 2 trial of lenabasum for DM enrolled 22 adults with minimal muscle involvement as evidenced by normal maximal resistance on muscle testing at entry and throughout the study. Most were taking immunosuppressant medication, and all had CDASI scores of at least 20, with mean scores in the severe range (> 26). Symptoms registered on patient-reported outcome measures were moderate to severe.
Patients received a half-dose of lenabasum (20 mg daily) for 1 month and a full dose (20 mg twice daily) for 2 months, or placebo, and were followed for an additional month without dosing.
Starting at day 43 – approximately 2 weeks after the dose was increased – there was “a trend for the change from baseline CDASI to be greater” in the lenabasum group, compared with those on placebo, Dr. Werth and colleagues reported. The differences reached statistical significance on day 113 (P = .038), a month after patients discontinued lenabasum, “suggesting that the modulation of the inflammatory response by lenabasum continued beyond its last dose.”
Five of the 11 patients treated with lenabasum (45%), and none of those on placebo, achieved at least a 40% reduction in the CDASI activity score by the end of the study.
Patients in the lenabasum group also had greater improvement in the Skindex-29 Symptoms scores – an objective measure of itch – and improvements in other secondary efficacy outcomes, including pain, though these did not reach statistical significance.
Skin biopsies before and after treatment showed significant reductions in inflammatory cytokines relevant to DM pathogenesis. Patients treated with the CB2 agonist had a downward trend in the CD4+ T cell population, which correlated with decreased CDASI activity scores, for instance, and a decrease in IL-31 protein expression, which correlated with decreased Skindex-29 Symptoms scores, the investigators reported.
There were no serious adverse events related to the CB2 agonist, and no treatment discontinuations.
The main part of the phase 2 trial, conducted from 2015 to 2017, was followed by a 3-year, open-label extension, in which 20 of the 22 patients took lenabasum 20 mg twice a day. The drug continued to be safe and well tolerated, and the CDASI activity score and other outcomes improved through year 1 and remained stable thereafter, according to a poster presented by Dr. Werth at the 2021 EULAR meeting.
After 1 year in the open-label extension, 60%-70% of patients had achieved mild skin disease, and 75% had achieved at least a 40% reduction in CDASI activity.
“A lot of patients, even if they weren’t completely cleared, were much happier in terms of their itch,” said Dr. Werth, also chief of dermatology, Corporal Michael J. Crescenz Veterans Affairs Medical Center, Philadelphia. “It’s been difficult for a lot of them now that they’re off the long-term extension ... a lot of them are flaring.”
The future
In the lab, with funding from the National Institutes of Health, Dr. Werth is continuing to investigate how lenabasum may be working in DM. A paper just published in the open access journal Arthritis Research & Therapy describes CB2 receptor distribution and up-regulation on key immune cells in the skin and blood, and how, in DM skin, its highest expression is on dendritic cells.
Through both mechanistic and more clinical research, “it’s important to understand the characteristics of the people [lenabasum] worked in or didn’t work in,” she said.
And in clinical trials, it’s important to capture meaningful improvement from the patient perspective. “It may be,” she noted, “that more global, systemic assessments are not the way to go for autoimmune skin disease.”
For dcSSc, Dr. Spiera said, it’s possible that a CB2 agonist may be helpful for patients who have been on immunosuppressants, particularly mycophenolate, for more than 6 months “and are still struggling.”
The phase 2 trial in DM was funded by the National Institutes of Health, the Department of Veterans Affairs, and Corbus Pharmaceuticals. The phase 3 trials in DM and in dcSSc were funded by Corbus. Dr. Werth disclosed grant support from Corbus and several other pharmaceutical companies. Dr. Spiera disclosed that he has received grant support or consulting fees from Roche-Genentech, GlaxoSmithKline, and several other pharmaceutical companies.
A version of this article first appeared on Medscape.com.
An – some of it statistically significant – in a phase 2, double-blind, randomized, controlled study.
Patients taking lenabasum experienced greater reductions in the Cutaneous Dermatomyositis Disease Area and Severity Index (CDASI) – a validated outcome designed to assess inflammatory skin involvement in the rare autoimmune disease – and improvements in patient-reported and biomarker outcomes, compared with those on placebo, dermatologist Victoria P. Werth, MD, and coinvestigators reported.
And in a recently completed phase 3 trial, reported by the manufacturer, a subpopulation of patients with active skin disease and no active muscle disease again showed greater reductions in CDASI activity scores – a secondary outcome in the trial.
However, the phase 3 DETERMINE trial produced negative findings overall. It enrolled a more heterogeneous group of patients – including those with both muscle weakness and skin involvement – and its primary outcome measure was a broader composite measure, the Total Improvement Score. The trial failed to meet this primary endpoint, Corbus Pharmaceuticals, the developer of lenabasum, announced in a press release in June 2021.
The phase 3 results are “frustrating” for patients with symptomatic and refractory skin manifestations of dermatomyositis (DM), given the promising findings from the phase 2 trial and from an open-label extension study, said Dr. Werth, professor of dermatology and medicine, University of Pennsylvania, Philadelphia, and principal investigator and coprincipal investigator of the phase 2 and phase 3 studies, respectively.
Dr. Werth is scheduled to present the results from the phase 3 trial at the annual European Alliance of Associations for Rheumatology meeting in June.
“With lenabasum, we have a therapy that doesn’t work for every patient, but does work for quite a number of them,” Dr. Werth said in an interview. “It’s oral, it’s not really that immunosuppressing, and there aren’t many side effects. Right now, patients are often being managed with steroids ... we really need treatments that are not as toxic.”
Robert Spiera, MD, a rheumatologist who led trials of lenabasum for treatment of diffuse cutaneous systemic sclerosis (dcSSc), agreed. “The CB2 agonist strategy is appealing because it’s nonimmunosuppressing and has both anti-inflammatory and antifibrotic properties,” he said in an interview. “I wouldn’t want to give up on it ... especially [for patients] with scleroderma and dermatomyositis who are treated with substantial drugs that are associated with morbidity.”
Lenabasum, he said, has proven to be “incredibly safe, and incredibly safe in the long term.”
While the phase 2 trial of the drug for dcSSc showed clear benefit over placebo, the phase 3 trial did not meet its primary endpoint using the American College of Rheumatology Combined Response Index in Diffuse Cutaneous Systemic Sclerosis.
It allowed background immunosuppressant therapy to reflect real-world clinical practice, and “there was such a high response rate to [that therapy, largely mycophenolate] that there was little room to show benefit beyond that,” said Dr. Spiera, director of the vasculitis and scleroderma program, Hospital for Special Surgery, New York.
The drug led to more improvement in the small subset of participants who were not receiving background immunotherapy during the trial, he noted.
Corbus is currently “seeking a partnership to further explore the drug” for treatment in different subpopulations, according to a company spokesperson. Results of a phase 2 trial of lenabasum for the treatment of systemic lupus erythematosus – with a pain rating as the primary outcome measure – are expected soon.
Phase 2 findings
The single-center phase 2 trial of lenabasum for DM enrolled 22 adults with minimal muscle involvement as evidenced by normal maximal resistance on muscle testing at entry and throughout the study. Most were taking immunosuppressant medication, and all had CDASI scores of at least 20, with mean scores in the severe range (> 26). Symptoms registered on patient-reported outcome measures were moderate to severe.
Patients received a half-dose of lenabasum (20 mg daily) for 1 month and a full dose (20 mg twice daily) for 2 months, or placebo, and were followed for an additional month without dosing.
Starting at day 43 – approximately 2 weeks after the dose was increased – there was “a trend for the change from baseline CDASI to be greater” in the lenabasum group, compared with those on placebo, Dr. Werth and colleagues reported. The differences reached statistical significance on day 113 (P = .038), a month after patients discontinued lenabasum, “suggesting that the modulation of the inflammatory response by lenabasum continued beyond its last dose.”
Five of the 11 patients treated with lenabasum (45%), and none of those on placebo, achieved at least a 40% reduction in the CDASI activity score by the end of the study.
Patients in the lenabasum group also had greater improvement in the Skindex-29 Symptoms scores – an objective measure of itch – and improvements in other secondary efficacy outcomes, including pain, though these did not reach statistical significance.
Skin biopsies before and after treatment showed significant reductions in inflammatory cytokines relevant to DM pathogenesis. Patients treated with the CB2 agonist had a downward trend in the CD4+ T cell population, which correlated with decreased CDASI activity scores, for instance, and a decrease in IL-31 protein expression, which correlated with decreased Skindex-29 Symptoms scores, the investigators reported.
There were no serious adverse events related to the CB2 agonist, and no treatment discontinuations.
The main part of the phase 2 trial, conducted from 2015 to 2017, was followed by a 3-year, open-label extension, in which 20 of the 22 patients took lenabasum 20 mg twice a day. The drug continued to be safe and well tolerated, and the CDASI activity score and other outcomes improved through year 1 and remained stable thereafter, according to a poster presented by Dr. Werth at the 2021 EULAR meeting.
After 1 year in the open-label extension, 60%-70% of patients had achieved mild skin disease, and 75% had achieved at least a 40% reduction in CDASI activity.
“A lot of patients, even if they weren’t completely cleared, were much happier in terms of their itch,” said Dr. Werth, also chief of dermatology, Corporal Michael J. Crescenz Veterans Affairs Medical Center, Philadelphia. “It’s been difficult for a lot of them now that they’re off the long-term extension ... a lot of them are flaring.”
The future
In the lab, with funding from the National Institutes of Health, Dr. Werth is continuing to investigate how lenabasum may be working in DM. A paper just published in the open access journal Arthritis Research & Therapy describes CB2 receptor distribution and up-regulation on key immune cells in the skin and blood, and how, in DM skin, its highest expression is on dendritic cells.
Through both mechanistic and more clinical research, “it’s important to understand the characteristics of the people [lenabasum] worked in or didn’t work in,” she said.
And in clinical trials, it’s important to capture meaningful improvement from the patient perspective. “It may be,” she noted, “that more global, systemic assessments are not the way to go for autoimmune skin disease.”
For dcSSc, Dr. Spiera said, it’s possible that a CB2 agonist may be helpful for patients who have been on immunosuppressants, particularly mycophenolate, for more than 6 months “and are still struggling.”
The phase 2 trial in DM was funded by the National Institutes of Health, the Department of Veterans Affairs, and Corbus Pharmaceuticals. The phase 3 trials in DM and in dcSSc were funded by Corbus. Dr. Werth disclosed grant support from Corbus and several other pharmaceutical companies. Dr. Spiera disclosed that he has received grant support or consulting fees from Roche-Genentech, GlaxoSmithKline, and several other pharmaceutical companies.
A version of this article first appeared on Medscape.com.
An – some of it statistically significant – in a phase 2, double-blind, randomized, controlled study.
Patients taking lenabasum experienced greater reductions in the Cutaneous Dermatomyositis Disease Area and Severity Index (CDASI) – a validated outcome designed to assess inflammatory skin involvement in the rare autoimmune disease – and improvements in patient-reported and biomarker outcomes, compared with those on placebo, dermatologist Victoria P. Werth, MD, and coinvestigators reported.
And in a recently completed phase 3 trial, reported by the manufacturer, a subpopulation of patients with active skin disease and no active muscle disease again showed greater reductions in CDASI activity scores – a secondary outcome in the trial.
However, the phase 3 DETERMINE trial produced negative findings overall. It enrolled a more heterogeneous group of patients – including those with both muscle weakness and skin involvement – and its primary outcome measure was a broader composite measure, the Total Improvement Score. The trial failed to meet this primary endpoint, Corbus Pharmaceuticals, the developer of lenabasum, announced in a press release in June 2021.
The phase 3 results are “frustrating” for patients with symptomatic and refractory skin manifestations of dermatomyositis (DM), given the promising findings from the phase 2 trial and from an open-label extension study, said Dr. Werth, professor of dermatology and medicine, University of Pennsylvania, Philadelphia, and principal investigator and coprincipal investigator of the phase 2 and phase 3 studies, respectively.
Dr. Werth is scheduled to present the results from the phase 3 trial at the annual European Alliance of Associations for Rheumatology meeting in June.
“With lenabasum, we have a therapy that doesn’t work for every patient, but does work for quite a number of them,” Dr. Werth said in an interview. “It’s oral, it’s not really that immunosuppressing, and there aren’t many side effects. Right now, patients are often being managed with steroids ... we really need treatments that are not as toxic.”
Robert Spiera, MD, a rheumatologist who led trials of lenabasum for treatment of diffuse cutaneous systemic sclerosis (dcSSc), agreed. “The CB2 agonist strategy is appealing because it’s nonimmunosuppressing and has both anti-inflammatory and antifibrotic properties,” he said in an interview. “I wouldn’t want to give up on it ... especially [for patients] with scleroderma and dermatomyositis who are treated with substantial drugs that are associated with morbidity.”
Lenabasum, he said, has proven to be “incredibly safe, and incredibly safe in the long term.”
While the phase 2 trial of the drug for dcSSc showed clear benefit over placebo, the phase 3 trial did not meet its primary endpoint using the American College of Rheumatology Combined Response Index in Diffuse Cutaneous Systemic Sclerosis.
It allowed background immunosuppressant therapy to reflect real-world clinical practice, and “there was such a high response rate to [that therapy, largely mycophenolate] that there was little room to show benefit beyond that,” said Dr. Spiera, director of the vasculitis and scleroderma program, Hospital for Special Surgery, New York.
The drug led to more improvement in the small subset of participants who were not receiving background immunotherapy during the trial, he noted.
Corbus is currently “seeking a partnership to further explore the drug” for treatment in different subpopulations, according to a company spokesperson. Results of a phase 2 trial of lenabasum for the treatment of systemic lupus erythematosus – with a pain rating as the primary outcome measure – are expected soon.
Phase 2 findings
The single-center phase 2 trial of lenabasum for DM enrolled 22 adults with minimal muscle involvement as evidenced by normal maximal resistance on muscle testing at entry and throughout the study. Most were taking immunosuppressant medication, and all had CDASI scores of at least 20, with mean scores in the severe range (> 26). Symptoms registered on patient-reported outcome measures were moderate to severe.
Patients received a half-dose of lenabasum (20 mg daily) for 1 month and a full dose (20 mg twice daily) for 2 months, or placebo, and were followed for an additional month without dosing.
Starting at day 43 – approximately 2 weeks after the dose was increased – there was “a trend for the change from baseline CDASI to be greater” in the lenabasum group, compared with those on placebo, Dr. Werth and colleagues reported. The differences reached statistical significance on day 113 (P = .038), a month after patients discontinued lenabasum, “suggesting that the modulation of the inflammatory response by lenabasum continued beyond its last dose.”
Five of the 11 patients treated with lenabasum (45%), and none of those on placebo, achieved at least a 40% reduction in the CDASI activity score by the end of the study.
Patients in the lenabasum group also had greater improvement in the Skindex-29 Symptoms scores – an objective measure of itch – and improvements in other secondary efficacy outcomes, including pain, though these did not reach statistical significance.
Skin biopsies before and after treatment showed significant reductions in inflammatory cytokines relevant to DM pathogenesis. Patients treated with the CB2 agonist had a downward trend in the CD4+ T cell population, which correlated with decreased CDASI activity scores, for instance, and a decrease in IL-31 protein expression, which correlated with decreased Skindex-29 Symptoms scores, the investigators reported.
There were no serious adverse events related to the CB2 agonist, and no treatment discontinuations.
The main part of the phase 2 trial, conducted from 2015 to 2017, was followed by a 3-year, open-label extension, in which 20 of the 22 patients took lenabasum 20 mg twice a day. The drug continued to be safe and well tolerated, and the CDASI activity score and other outcomes improved through year 1 and remained stable thereafter, according to a poster presented by Dr. Werth at the 2021 EULAR meeting.
After 1 year in the open-label extension, 60%-70% of patients had achieved mild skin disease, and 75% had achieved at least a 40% reduction in CDASI activity.
“A lot of patients, even if they weren’t completely cleared, were much happier in terms of their itch,” said Dr. Werth, also chief of dermatology, Corporal Michael J. Crescenz Veterans Affairs Medical Center, Philadelphia. “It’s been difficult for a lot of them now that they’re off the long-term extension ... a lot of them are flaring.”
The future
In the lab, with funding from the National Institutes of Health, Dr. Werth is continuing to investigate how lenabasum may be working in DM. A paper just published in the open access journal Arthritis Research & Therapy describes CB2 receptor distribution and up-regulation on key immune cells in the skin and blood, and how, in DM skin, its highest expression is on dendritic cells.
Through both mechanistic and more clinical research, “it’s important to understand the characteristics of the people [lenabasum] worked in or didn’t work in,” she said.
And in clinical trials, it’s important to capture meaningful improvement from the patient perspective. “It may be,” she noted, “that more global, systemic assessments are not the way to go for autoimmune skin disease.”
For dcSSc, Dr. Spiera said, it’s possible that a CB2 agonist may be helpful for patients who have been on immunosuppressants, particularly mycophenolate, for more than 6 months “and are still struggling.”
The phase 2 trial in DM was funded by the National Institutes of Health, the Department of Veterans Affairs, and Corbus Pharmaceuticals. The phase 3 trials in DM and in dcSSc were funded by Corbus. Dr. Werth disclosed grant support from Corbus and several other pharmaceutical companies. Dr. Spiera disclosed that he has received grant support or consulting fees from Roche-Genentech, GlaxoSmithKline, and several other pharmaceutical companies.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
Severe infections often accompany severe psoriasis
of nearly 95,000 patients.
Although previous studies have shown a higher risk for comorbid conditions in people with psoriasis, compared with those without psoriasis, data on the occurrence of severe and rare infections in patients with psoriasis are limited, wrote Nikolai Loft, MD, of the department of dermatology and allergy, Copenhagen University Hospital, Gentofte, and colleagues.
Psoriasis patients are often treated with immunosuppressive therapies that may promote or aggravate infections; therefore, a better understanding of psoriasis and risk of infections is needed, they said. In a study published in the British Journal of Dermatology, Dr. Loft and his coinvestigators reviewed data on adults aged 18 years and older from the Danish National Patient Register between Jan. 1, 1997 and Dec. 31, 2018. The study population included 94,450 adults with psoriasis and 566,700 matched controls. Patients with any type of psoriasis and any degree of severity were included.
The primary outcome was the occurrence of severe infections, defined as those requiring assessment at a hospital, and rare infections, defined as HIV, TB, HBV, and HCV. The median age of the participants was 52.3 years, and slightly more than half were women.
Overall, the incidence rate of severe and rare infections among patients with any type of psoriasis was 3,104.9 per 100,000 person-years, compared with 2,381.1 for controls, with a hazard ratio, adjusted for gender, age, ethnicity, socioeconomic status, alcohol-related conditions, and Charlson comorbidity index (aHR) of 1.29.
For any infections resulting in hospitalization, the incidence rate was 2,005.1 vs. 1,531.8 per 100,000 person-years for patients with any type of psoriasis and controls, respectively.
The results were similar when severe infections and rare infections were analyzed separately. The incidence rate of severe infections was 3,080.6 and 2,364.4 per 100,000 person-years for patients with any psoriasis, compared with controls; the incidence rate for rare infections was 42.9 and 31.8 for all psoriasis patients and controls, respectively.
When the data were examined by psoriasis severity, the incidence rate of severe and rare infections among patients with severe psoriasis was 3,847.7 per 100,000 person-years, compared with 2,351.9 per 100,000 person years among controls (aHR, 1.58) and also higher than in patients with mild psoriasis. The incidence rate of severe and rare infections in patients with mild psoriasis (2,979.1 per 100,000 person-years) also was higher than in controls (aHR, 1.26).
Factors that might explain the increased infection risk with severe psoriasis include the altered immune environment in these patients, the researchers wrote in their discussion of the findings. Also, “patients with severe psoriasis are defined by their eligibility for systemics, either conventional or biologic,” and their increased infection risk may stem from these treatments, rather than disease severity itself, they noted.
The study findings were limited by several factors including the lack of data on such confounders as weight, body mass index, and smoking status, they added. Other limitations included potential surveillance bias because of greater TB screening, and the use of prescriptions, rather than the Psoriasis Area Severity Index, to define severity. However, the results were strengthened by the large sample size, and suggest that patients with any type of psoriasis have higher rates of any infection, severe or rare, than the general population, the researchers concluded.
Data show need for clinician vigilance
Based on the 2020 Census data, an estimated 7.55 million adults in the United States have psoriasis, David Robles, MD, said in an interview. “Patients with psoriasis have a high risk for multiple comorbid conditions including metabolic syndrome, which is characterized by obesity, hypertension, and dyslipidemia,” said Dr. Robles, a dermatologist in private practice in Pomona, Calif., who was not involved in the study. “Although these complications were previously attributed to diet and obesity, it has become clear that the proinflammatory cytokines associated with psoriasis may be playing an important role underlying the pathologic basis of these other comorbidities.”
There is an emerging body of literature “indicating that psoriasis is associated with an increased risk of infections,” he added. Research in this area is particularly important because of the increased risk of infections associated with many biologic and immune-modulating treatments for psoriasis, Dr. Robles noted.
The study findings “indicate that, as the severity of psoriasis increases, so does the risk of severe and rare infections,” he said. “This makes it imperative for clinicians to be alert to the possibility of severe or rare infections in patients with psoriasis, especially those with severe psoriasis, so that early intervention can be initiated.”
As for additional research, “as an immunologist and dermatologist, I cannot help but think about the possible role the genetic and cytokine pathways involved in psoriasis may be playing in modulating the immune system and/or microbiome, and whether this contributes to a higher risk of infections,” Dr. Robles said. “Just as it was discovered that patients with atopic dermatitis have decreased levels of antimicrobial peptides in their skin, making them susceptible to recurrent bacterial skin infections, we may find that the genetic and immunological changes associated with psoriasis may independently contribute to infection susceptibility,” he noted. “More basic immunology and virology research may one day shed light on this observation.”
The study was supported by Novartis. Lead author Dr. Loft disclosed serving as a speaker for Eli Lilly and Janssen Cilag, other authors disclosed relationships with multiple companies including Novartis, and two authors are Novartis employees. Dr. Robles had no relevant financial disclosures.
of nearly 95,000 patients.
Although previous studies have shown a higher risk for comorbid conditions in people with psoriasis, compared with those without psoriasis, data on the occurrence of severe and rare infections in patients with psoriasis are limited, wrote Nikolai Loft, MD, of the department of dermatology and allergy, Copenhagen University Hospital, Gentofte, and colleagues.
Psoriasis patients are often treated with immunosuppressive therapies that may promote or aggravate infections; therefore, a better understanding of psoriasis and risk of infections is needed, they said. In a study published in the British Journal of Dermatology, Dr. Loft and his coinvestigators reviewed data on adults aged 18 years and older from the Danish National Patient Register between Jan. 1, 1997 and Dec. 31, 2018. The study population included 94,450 adults with psoriasis and 566,700 matched controls. Patients with any type of psoriasis and any degree of severity were included.
The primary outcome was the occurrence of severe infections, defined as those requiring assessment at a hospital, and rare infections, defined as HIV, TB, HBV, and HCV. The median age of the participants was 52.3 years, and slightly more than half were women.
Overall, the incidence rate of severe and rare infections among patients with any type of psoriasis was 3,104.9 per 100,000 person-years, compared with 2,381.1 for controls, with a hazard ratio, adjusted for gender, age, ethnicity, socioeconomic status, alcohol-related conditions, and Charlson comorbidity index (aHR) of 1.29.
For any infections resulting in hospitalization, the incidence rate was 2,005.1 vs. 1,531.8 per 100,000 person-years for patients with any type of psoriasis and controls, respectively.
The results were similar when severe infections and rare infections were analyzed separately. The incidence rate of severe infections was 3,080.6 and 2,364.4 per 100,000 person-years for patients with any psoriasis, compared with controls; the incidence rate for rare infections was 42.9 and 31.8 for all psoriasis patients and controls, respectively.
When the data were examined by psoriasis severity, the incidence rate of severe and rare infections among patients with severe psoriasis was 3,847.7 per 100,000 person-years, compared with 2,351.9 per 100,000 person years among controls (aHR, 1.58) and also higher than in patients with mild psoriasis. The incidence rate of severe and rare infections in patients with mild psoriasis (2,979.1 per 100,000 person-years) also was higher than in controls (aHR, 1.26).
Factors that might explain the increased infection risk with severe psoriasis include the altered immune environment in these patients, the researchers wrote in their discussion of the findings. Also, “patients with severe psoriasis are defined by their eligibility for systemics, either conventional or biologic,” and their increased infection risk may stem from these treatments, rather than disease severity itself, they noted.
The study findings were limited by several factors including the lack of data on such confounders as weight, body mass index, and smoking status, they added. Other limitations included potential surveillance bias because of greater TB screening, and the use of prescriptions, rather than the Psoriasis Area Severity Index, to define severity. However, the results were strengthened by the large sample size, and suggest that patients with any type of psoriasis have higher rates of any infection, severe or rare, than the general population, the researchers concluded.
Data show need for clinician vigilance
Based on the 2020 Census data, an estimated 7.55 million adults in the United States have psoriasis, David Robles, MD, said in an interview. “Patients with psoriasis have a high risk for multiple comorbid conditions including metabolic syndrome, which is characterized by obesity, hypertension, and dyslipidemia,” said Dr. Robles, a dermatologist in private practice in Pomona, Calif., who was not involved in the study. “Although these complications were previously attributed to diet and obesity, it has become clear that the proinflammatory cytokines associated with psoriasis may be playing an important role underlying the pathologic basis of these other comorbidities.”
There is an emerging body of literature “indicating that psoriasis is associated with an increased risk of infections,” he added. Research in this area is particularly important because of the increased risk of infections associated with many biologic and immune-modulating treatments for psoriasis, Dr. Robles noted.
The study findings “indicate that, as the severity of psoriasis increases, so does the risk of severe and rare infections,” he said. “This makes it imperative for clinicians to be alert to the possibility of severe or rare infections in patients with psoriasis, especially those with severe psoriasis, so that early intervention can be initiated.”
As for additional research, “as an immunologist and dermatologist, I cannot help but think about the possible role the genetic and cytokine pathways involved in psoriasis may be playing in modulating the immune system and/or microbiome, and whether this contributes to a higher risk of infections,” Dr. Robles said. “Just as it was discovered that patients with atopic dermatitis have decreased levels of antimicrobial peptides in their skin, making them susceptible to recurrent bacterial skin infections, we may find that the genetic and immunological changes associated with psoriasis may independently contribute to infection susceptibility,” he noted. “More basic immunology and virology research may one day shed light on this observation.”
The study was supported by Novartis. Lead author Dr. Loft disclosed serving as a speaker for Eli Lilly and Janssen Cilag, other authors disclosed relationships with multiple companies including Novartis, and two authors are Novartis employees. Dr. Robles had no relevant financial disclosures.
of nearly 95,000 patients.
Although previous studies have shown a higher risk for comorbid conditions in people with psoriasis, compared with those without psoriasis, data on the occurrence of severe and rare infections in patients with psoriasis are limited, wrote Nikolai Loft, MD, of the department of dermatology and allergy, Copenhagen University Hospital, Gentofte, and colleagues.
Psoriasis patients are often treated with immunosuppressive therapies that may promote or aggravate infections; therefore, a better understanding of psoriasis and risk of infections is needed, they said. In a study published in the British Journal of Dermatology, Dr. Loft and his coinvestigators reviewed data on adults aged 18 years and older from the Danish National Patient Register between Jan. 1, 1997 and Dec. 31, 2018. The study population included 94,450 adults with psoriasis and 566,700 matched controls. Patients with any type of psoriasis and any degree of severity were included.
The primary outcome was the occurrence of severe infections, defined as those requiring assessment at a hospital, and rare infections, defined as HIV, TB, HBV, and HCV. The median age of the participants was 52.3 years, and slightly more than half were women.
Overall, the incidence rate of severe and rare infections among patients with any type of psoriasis was 3,104.9 per 100,000 person-years, compared with 2,381.1 for controls, with a hazard ratio, adjusted for gender, age, ethnicity, socioeconomic status, alcohol-related conditions, and Charlson comorbidity index (aHR) of 1.29.
For any infections resulting in hospitalization, the incidence rate was 2,005.1 vs. 1,531.8 per 100,000 person-years for patients with any type of psoriasis and controls, respectively.
The results were similar when severe infections and rare infections were analyzed separately. The incidence rate of severe infections was 3,080.6 and 2,364.4 per 100,000 person-years for patients with any psoriasis, compared with controls; the incidence rate for rare infections was 42.9 and 31.8 for all psoriasis patients and controls, respectively.
When the data were examined by psoriasis severity, the incidence rate of severe and rare infections among patients with severe psoriasis was 3,847.7 per 100,000 person-years, compared with 2,351.9 per 100,000 person years among controls (aHR, 1.58) and also higher than in patients with mild psoriasis. The incidence rate of severe and rare infections in patients with mild psoriasis (2,979.1 per 100,000 person-years) also was higher than in controls (aHR, 1.26).
Factors that might explain the increased infection risk with severe psoriasis include the altered immune environment in these patients, the researchers wrote in their discussion of the findings. Also, “patients with severe psoriasis are defined by their eligibility for systemics, either conventional or biologic,” and their increased infection risk may stem from these treatments, rather than disease severity itself, they noted.
The study findings were limited by several factors including the lack of data on such confounders as weight, body mass index, and smoking status, they added. Other limitations included potential surveillance bias because of greater TB screening, and the use of prescriptions, rather than the Psoriasis Area Severity Index, to define severity. However, the results were strengthened by the large sample size, and suggest that patients with any type of psoriasis have higher rates of any infection, severe or rare, than the general population, the researchers concluded.
Data show need for clinician vigilance
Based on the 2020 Census data, an estimated 7.55 million adults in the United States have psoriasis, David Robles, MD, said in an interview. “Patients with psoriasis have a high risk for multiple comorbid conditions including metabolic syndrome, which is characterized by obesity, hypertension, and dyslipidemia,” said Dr. Robles, a dermatologist in private practice in Pomona, Calif., who was not involved in the study. “Although these complications were previously attributed to diet and obesity, it has become clear that the proinflammatory cytokines associated with psoriasis may be playing an important role underlying the pathologic basis of these other comorbidities.”
There is an emerging body of literature “indicating that psoriasis is associated with an increased risk of infections,” he added. Research in this area is particularly important because of the increased risk of infections associated with many biologic and immune-modulating treatments for psoriasis, Dr. Robles noted.
The study findings “indicate that, as the severity of psoriasis increases, so does the risk of severe and rare infections,” he said. “This makes it imperative for clinicians to be alert to the possibility of severe or rare infections in patients with psoriasis, especially those with severe psoriasis, so that early intervention can be initiated.”
As for additional research, “as an immunologist and dermatologist, I cannot help but think about the possible role the genetic and cytokine pathways involved in psoriasis may be playing in modulating the immune system and/or microbiome, and whether this contributes to a higher risk of infections,” Dr. Robles said. “Just as it was discovered that patients with atopic dermatitis have decreased levels of antimicrobial peptides in their skin, making them susceptible to recurrent bacterial skin infections, we may find that the genetic and immunological changes associated with psoriasis may independently contribute to infection susceptibility,” he noted. “More basic immunology and virology research may one day shed light on this observation.”
The study was supported by Novartis. Lead author Dr. Loft disclosed serving as a speaker for Eli Lilly and Janssen Cilag, other authors disclosed relationships with multiple companies including Novartis, and two authors are Novartis employees. Dr. Robles had no relevant financial disclosures.
FROM BRITISH JOURNAL OF DERMATOLOGY
A 64-year-old woman presents with a history of asymptomatic erythematous grouped papules on the right breast
. Recurrences may occur. Rarely, lymph nodes, the gastrointestinal system, lung, bone and bone marrow may be involved as extracutaneous sites.
Primary cutaneous B-cell lymphomas account for approximately 25% of all cutaneous lymphomas. Clinically, patients present with either solitary or multiple papules or plaques, typically on the upper extremities or trunk.
Histopathology is vital for the correct diagnosis. In this patient, the histologic report was written as follows: “The findings are those of a well-differentiated but atypical diffuse mixed small lymphocytic infiltrate representing a mixture of T-cells and B-cells. The minor component of the infiltrate is of T-cell lineage, whereby the cells do not show any phenotypic abnormalities. The background cell population is interpreted as reactive. However, the dominant cell population is in fact of B-cell lineage. It is extensively highlighted by CD20. Only a minor component of the B cell infiltrate appeared to be in the context of representing germinal centers as characterized by small foci of centrocytic and centroblastic infiltration highlighted by BCL6 and CD10. The overwhelming B-cell component is a non–germinal center small B cell that does demonstrate BCL2 positivity and significant immunoreactivity for CD23. This small lymphocytic infiltrate obscures the germinal centers. There are only a few plasma cells; they do not show light chain restriction.”
The pathologist remarked that “this type of morphology of a diffuse small B-cell lymphocytic infiltrate that is without any evidence of light chain restriction amidst plasma cells, whereby the B cell component is dominant over the T-cell component would in fact be consistent with a unique variant of marginal zone lymphoma derived from a naive mantle zone.”
PCMZL has an excellent prognosis. When limited to the skin, local radiation or excision are effective treatments. Intravenous rituximab has been used to treat multifocal PCMZL. This patient was found to have no extracutaneous involvement and was treated with radiation.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Virmani P et al. JAAD Case Rep. 2017 Jun 14;3(4):269-72.
Magro CM and Olson LC. Ann Diagn Pathol. 2018 Jun;34:116-21.
. Recurrences may occur. Rarely, lymph nodes, the gastrointestinal system, lung, bone and bone marrow may be involved as extracutaneous sites.
Primary cutaneous B-cell lymphomas account for approximately 25% of all cutaneous lymphomas. Clinically, patients present with either solitary or multiple papules or plaques, typically on the upper extremities or trunk.
Histopathology is vital for the correct diagnosis. In this patient, the histologic report was written as follows: “The findings are those of a well-differentiated but atypical diffuse mixed small lymphocytic infiltrate representing a mixture of T-cells and B-cells. The minor component of the infiltrate is of T-cell lineage, whereby the cells do not show any phenotypic abnormalities. The background cell population is interpreted as reactive. However, the dominant cell population is in fact of B-cell lineage. It is extensively highlighted by CD20. Only a minor component of the B cell infiltrate appeared to be in the context of representing germinal centers as characterized by small foci of centrocytic and centroblastic infiltration highlighted by BCL6 and CD10. The overwhelming B-cell component is a non–germinal center small B cell that does demonstrate BCL2 positivity and significant immunoreactivity for CD23. This small lymphocytic infiltrate obscures the germinal centers. There are only a few plasma cells; they do not show light chain restriction.”
The pathologist remarked that “this type of morphology of a diffuse small B-cell lymphocytic infiltrate that is without any evidence of light chain restriction amidst plasma cells, whereby the B cell component is dominant over the T-cell component would in fact be consistent with a unique variant of marginal zone lymphoma derived from a naive mantle zone.”
PCMZL has an excellent prognosis. When limited to the skin, local radiation or excision are effective treatments. Intravenous rituximab has been used to treat multifocal PCMZL. This patient was found to have no extracutaneous involvement and was treated with radiation.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Virmani P et al. JAAD Case Rep. 2017 Jun 14;3(4):269-72.
Magro CM and Olson LC. Ann Diagn Pathol. 2018 Jun;34:116-21.
. Recurrences may occur. Rarely, lymph nodes, the gastrointestinal system, lung, bone and bone marrow may be involved as extracutaneous sites.
Primary cutaneous B-cell lymphomas account for approximately 25% of all cutaneous lymphomas. Clinically, patients present with either solitary or multiple papules or plaques, typically on the upper extremities or trunk.
Histopathology is vital for the correct diagnosis. In this patient, the histologic report was written as follows: “The findings are those of a well-differentiated but atypical diffuse mixed small lymphocytic infiltrate representing a mixture of T-cells and B-cells. The minor component of the infiltrate is of T-cell lineage, whereby the cells do not show any phenotypic abnormalities. The background cell population is interpreted as reactive. However, the dominant cell population is in fact of B-cell lineage. It is extensively highlighted by CD20. Only a minor component of the B cell infiltrate appeared to be in the context of representing germinal centers as characterized by small foci of centrocytic and centroblastic infiltration highlighted by BCL6 and CD10. The overwhelming B-cell component is a non–germinal center small B cell that does demonstrate BCL2 positivity and significant immunoreactivity for CD23. This small lymphocytic infiltrate obscures the germinal centers. There are only a few plasma cells; they do not show light chain restriction.”
The pathologist remarked that “this type of morphology of a diffuse small B-cell lymphocytic infiltrate that is without any evidence of light chain restriction amidst plasma cells, whereby the B cell component is dominant over the T-cell component would in fact be consistent with a unique variant of marginal zone lymphoma derived from a naive mantle zone.”
PCMZL has an excellent prognosis. When limited to the skin, local radiation or excision are effective treatments. Intravenous rituximab has been used to treat multifocal PCMZL. This patient was found to have no extracutaneous involvement and was treated with radiation.
This case and photo were submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to dermnews@mdedge.com.
References
Virmani P et al. JAAD Case Rep. 2017 Jun 14;3(4):269-72.
Magro CM and Olson LC. Ann Diagn Pathol. 2018 Jun;34:116-21.
A 7-year-old with red bumps on her nose
The finding of individual, 1- to 4-mm firm, red papules depicted in the image are consistent with facial angiofibromas, which are most commonly seen in pediatric patients as a manifestation of tuberous sclerosis (TSC). Angiofibromas, previously called adenoma sebaceum, a misnomer, are seen in TSC as smooth papules, nodules, and occasionally plaques that typically involve the malar region of the face. These lesions usually develop in childhood and adolescence and can be misdiagnosed as lesions of acne. The number of lesions tend to increase with age, though there is no significant risk of malignant transformation. Ultraviolet-induced DNA damage is thought to play a role in the development of facial angiofibromas, so sun protection is called for.1 Patients may seek treatment to minimize deformity and the stigma of angiofibromas. Recently, the mammalian target of rapamycin inhibitor sirolimus (rapamycin) topical gel received Food and Drug Administration approval for the treatment of facial angiofibromas associated with TSC in patients age at least 6 years.2
The presence of angiofibromas should prompt consideration of TSC and as such, a thorough family history, medical history, and full-body skin examination. TSC is a rare autosomal-dominant genetic disorder, caused by a pathogenic variant in either the TSC1 or TSC2 gene. This neurocutaneous disorder is characterized by the development of multiple benign hamartomas across many organ systems including the brain, eyes, heart, lung, liver, kidney, and skin. The phenotypic expression of TSC is highly variable. Besides angiofibromas, some other characteristic dermatological findings in TSC include periungual fibromas, hypopigmented macules usually elliptical in shape (known as ash-leaf spots), and irregularly shaped elevated flesh-colored fibrous tissue most often found over the lower back (known as shagreen patches).3
What is on the differential?
Agminated spitz nevi refers to multiple spitz nevi in a localized area. Spitz nevi present as a well-circumscribed, dome-shaped, pink-red or brown papules, most commonly located on the face or lower extremities.4 The finding of agminated spitz nevi is very rare and less likely for this patient given the concomitant skin findings of dental pitting, renal cysts, and cortical tubers.
Juvenile xanthogranulomas are benign,proliferations of histiocytic cells that present as reddish or yellowish-to-brown papules, plaques, or nodules that typically develop in young children around the age of 1. With time, juvenile xanthogranulomas may flatten and become more yellow.
Basal cell carcinomas present as dome-shaped lesions with centralized erosions on sun-exposed areas of the skin. They are remarkably uncommon in children but are occasionally seen in basal cell nevus syndrome (also known as nevoid basal cell carcinoma syndrome or Gorlin syndrome). Affected patients may have other findings such as developmental anomalies, bifid ribs, palmar and plantar pitting, odontogenic keratocysts, and/or medulloblastomas.5
Flat warts commonly occur in children and occur by direct skin contact with human papillomavirus. Of the various types of warts, flat warts are smaller and tend to be smooth on top. The diagnosis of cutaneous warts is based on clinical appearance, showing thrombosed capillaries underneath the overlying hyperkeratotic debris. Our patient’s history of having a common wart on her hands raises suspicion for inoculation onto her face, but the morphology, distribution, and lack of response to tretinoin makes this diagnosis much less likely.
Disease workup and course
Our patient’s physical exam revealed dental pits but no evidence of hypopigmented macules, shagreen patches, or periungual lesions. Ultrasound of the kidney displayed renal cortical cysts and brain MRI showed cortical tubers, confirming extracutaneous TSC involvement. Over time, our patient developed angiofibromas on the forehead and was ultimately started on topical sirolimus, which led to marked improvement within months.
Ms. Kleinman is a pediatric dermatology research associate, division of pediatric and adolescent dermatology, University of California, San Diego, and Rady Children’s Hospital, also in San Diego. Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital. They have no relevant financial disclosures.
References
1. Tyburczy ME et al. Hum Molec Genet. 2014;23(8):2023-9.
2. Food & Drug Administration. New drug application (NDA) approval for Hyftor (sirolimus topical gel). https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2022/213478Orig1s000ltr.pdf.
3. Webb DW et al. Br J Dermatol. 1996;135(1):1-5.
4. Ricci F et al. Eur J Dermatol. 2017;27(1):59-62.
5. Evans DG and Farndon PA. Nevoid basal cell carcinoma syndrome, in “GeneReviews®.” Seattle: University of Washington, 2002.
The finding of individual, 1- to 4-mm firm, red papules depicted in the image are consistent with facial angiofibromas, which are most commonly seen in pediatric patients as a manifestation of tuberous sclerosis (TSC). Angiofibromas, previously called adenoma sebaceum, a misnomer, are seen in TSC as smooth papules, nodules, and occasionally plaques that typically involve the malar region of the face. These lesions usually develop in childhood and adolescence and can be misdiagnosed as lesions of acne. The number of lesions tend to increase with age, though there is no significant risk of malignant transformation. Ultraviolet-induced DNA damage is thought to play a role in the development of facial angiofibromas, so sun protection is called for.1 Patients may seek treatment to minimize deformity and the stigma of angiofibromas. Recently, the mammalian target of rapamycin inhibitor sirolimus (rapamycin) topical gel received Food and Drug Administration approval for the treatment of facial angiofibromas associated with TSC in patients age at least 6 years.2
The presence of angiofibromas should prompt consideration of TSC and as such, a thorough family history, medical history, and full-body skin examination. TSC is a rare autosomal-dominant genetic disorder, caused by a pathogenic variant in either the TSC1 or TSC2 gene. This neurocutaneous disorder is characterized by the development of multiple benign hamartomas across many organ systems including the brain, eyes, heart, lung, liver, kidney, and skin. The phenotypic expression of TSC is highly variable. Besides angiofibromas, some other characteristic dermatological findings in TSC include periungual fibromas, hypopigmented macules usually elliptical in shape (known as ash-leaf spots), and irregularly shaped elevated flesh-colored fibrous tissue most often found over the lower back (known as shagreen patches).3
What is on the differential?
Agminated spitz nevi refers to multiple spitz nevi in a localized area. Spitz nevi present as a well-circumscribed, dome-shaped, pink-red or brown papules, most commonly located on the face or lower extremities.4 The finding of agminated spitz nevi is very rare and less likely for this patient given the concomitant skin findings of dental pitting, renal cysts, and cortical tubers.
Juvenile xanthogranulomas are benign,proliferations of histiocytic cells that present as reddish or yellowish-to-brown papules, plaques, or nodules that typically develop in young children around the age of 1. With time, juvenile xanthogranulomas may flatten and become more yellow.
Basal cell carcinomas present as dome-shaped lesions with centralized erosions on sun-exposed areas of the skin. They are remarkably uncommon in children but are occasionally seen in basal cell nevus syndrome (also known as nevoid basal cell carcinoma syndrome or Gorlin syndrome). Affected patients may have other findings such as developmental anomalies, bifid ribs, palmar and plantar pitting, odontogenic keratocysts, and/or medulloblastomas.5
Flat warts commonly occur in children and occur by direct skin contact with human papillomavirus. Of the various types of warts, flat warts are smaller and tend to be smooth on top. The diagnosis of cutaneous warts is based on clinical appearance, showing thrombosed capillaries underneath the overlying hyperkeratotic debris. Our patient’s history of having a common wart on her hands raises suspicion for inoculation onto her face, but the morphology, distribution, and lack of response to tretinoin makes this diagnosis much less likely.
Disease workup and course
Our patient’s physical exam revealed dental pits but no evidence of hypopigmented macules, shagreen patches, or periungual lesions. Ultrasound of the kidney displayed renal cortical cysts and brain MRI showed cortical tubers, confirming extracutaneous TSC involvement. Over time, our patient developed angiofibromas on the forehead and was ultimately started on topical sirolimus, which led to marked improvement within months.
Ms. Kleinman is a pediatric dermatology research associate, division of pediatric and adolescent dermatology, University of California, San Diego, and Rady Children’s Hospital, also in San Diego. Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital. They have no relevant financial disclosures.
References
1. Tyburczy ME et al. Hum Molec Genet. 2014;23(8):2023-9.
2. Food & Drug Administration. New drug application (NDA) approval for Hyftor (sirolimus topical gel). https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2022/213478Orig1s000ltr.pdf.
3. Webb DW et al. Br J Dermatol. 1996;135(1):1-5.
4. Ricci F et al. Eur J Dermatol. 2017;27(1):59-62.
5. Evans DG and Farndon PA. Nevoid basal cell carcinoma syndrome, in “GeneReviews®.” Seattle: University of Washington, 2002.
The finding of individual, 1- to 4-mm firm, red papules depicted in the image are consistent with facial angiofibromas, which are most commonly seen in pediatric patients as a manifestation of tuberous sclerosis (TSC). Angiofibromas, previously called adenoma sebaceum, a misnomer, are seen in TSC as smooth papules, nodules, and occasionally plaques that typically involve the malar region of the face. These lesions usually develop in childhood and adolescence and can be misdiagnosed as lesions of acne. The number of lesions tend to increase with age, though there is no significant risk of malignant transformation. Ultraviolet-induced DNA damage is thought to play a role in the development of facial angiofibromas, so sun protection is called for.1 Patients may seek treatment to minimize deformity and the stigma of angiofibromas. Recently, the mammalian target of rapamycin inhibitor sirolimus (rapamycin) topical gel received Food and Drug Administration approval for the treatment of facial angiofibromas associated with TSC in patients age at least 6 years.2
The presence of angiofibromas should prompt consideration of TSC and as such, a thorough family history, medical history, and full-body skin examination. TSC is a rare autosomal-dominant genetic disorder, caused by a pathogenic variant in either the TSC1 or TSC2 gene. This neurocutaneous disorder is characterized by the development of multiple benign hamartomas across many organ systems including the brain, eyes, heart, lung, liver, kidney, and skin. The phenotypic expression of TSC is highly variable. Besides angiofibromas, some other characteristic dermatological findings in TSC include periungual fibromas, hypopigmented macules usually elliptical in shape (known as ash-leaf spots), and irregularly shaped elevated flesh-colored fibrous tissue most often found over the lower back (known as shagreen patches).3
What is on the differential?
Agminated spitz nevi refers to multiple spitz nevi in a localized area. Spitz nevi present as a well-circumscribed, dome-shaped, pink-red or brown papules, most commonly located on the face or lower extremities.4 The finding of agminated spitz nevi is very rare and less likely for this patient given the concomitant skin findings of dental pitting, renal cysts, and cortical tubers.
Juvenile xanthogranulomas are benign,proliferations of histiocytic cells that present as reddish or yellowish-to-brown papules, plaques, or nodules that typically develop in young children around the age of 1. With time, juvenile xanthogranulomas may flatten and become more yellow.
Basal cell carcinomas present as dome-shaped lesions with centralized erosions on sun-exposed areas of the skin. They are remarkably uncommon in children but are occasionally seen in basal cell nevus syndrome (also known as nevoid basal cell carcinoma syndrome or Gorlin syndrome). Affected patients may have other findings such as developmental anomalies, bifid ribs, palmar and plantar pitting, odontogenic keratocysts, and/or medulloblastomas.5
Flat warts commonly occur in children and occur by direct skin contact with human papillomavirus. Of the various types of warts, flat warts are smaller and tend to be smooth on top. The diagnosis of cutaneous warts is based on clinical appearance, showing thrombosed capillaries underneath the overlying hyperkeratotic debris. Our patient’s history of having a common wart on her hands raises suspicion for inoculation onto her face, but the morphology, distribution, and lack of response to tretinoin makes this diagnosis much less likely.
Disease workup and course
Our patient’s physical exam revealed dental pits but no evidence of hypopigmented macules, shagreen patches, or periungual lesions. Ultrasound of the kidney displayed renal cortical cysts and brain MRI showed cortical tubers, confirming extracutaneous TSC involvement. Over time, our patient developed angiofibromas on the forehead and was ultimately started on topical sirolimus, which led to marked improvement within months.
Ms. Kleinman is a pediatric dermatology research associate, division of pediatric and adolescent dermatology, University of California, San Diego, and Rady Children’s Hospital, also in San Diego. Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital. They have no relevant financial disclosures.
References
1. Tyburczy ME et al. Hum Molec Genet. 2014;23(8):2023-9.
2. Food & Drug Administration. New drug application (NDA) approval for Hyftor (sirolimus topical gel). https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2022/213478Orig1s000ltr.pdf.
3. Webb DW et al. Br J Dermatol. 1996;135(1):1-5.
4. Ricci F et al. Eur J Dermatol. 2017;27(1):59-62.
5. Evans DG and Farndon PA. Nevoid basal cell carcinoma syndrome, in “GeneReviews®.” Seattle: University of Washington, 2002.
A 7-year-old female presented with a bump on the bridge of her nose that was present for 10 months, with subsequent development of multiple papules on the nose and cheeks.
A 7-year-old, previously healthy female presented with a bump on the bridge of her nose that was present for 10 months, with subsequent development of multiple papules on the nose and cheeks. She has no significant medical history aside from a wart on her hand that was recently frozen with liquid nitrogen and resolved. She denied pruritus, bumps, or skin changes elsewhere on the body. The patient was prescribed tretinoin 0.1% cream applied nightly for several months without response.
Myositis guidelines aim to standardize adult and pediatric care
All patients with idiopathic inflammatory myopathies (IIM) should be screened for swallowing difficulties, according to the first evidence-based guideline to be produced.
The guideline, which has been developed by a working group of the British Society for Rheumatology (BSR), also advises that all diagnosed patients should have their myositis antibody levels checked and have their overall well-being assessed. Other recommendations for all patients include the use of glucocorticoids to reduce muscle inflammation and conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) for long-term treatment.
“Finally, now, we’re able to standardize the way we treat adults and children with IIM,” senior guideline author Hector Chinoy, PhD, said at the society’s annual meeting.
It has been a long labor of love, however, taking 4 years to get the guideline published, said Dr. Chinoy, professor of rheumatology and neuromuscular disease at the University of Manchester (England), and a consultant at Salford (England) Royal Hospital.
“We’re not covering diagnosis, classification, or the investigation of suspected IIM,” said Dr. Chinoy. Inclusion body myositis also is not included.
Altogether, there are 13 recommendations that have been developed using a PICO (patient or population, intervention, comparison, outcome) format, graded based on the quality of the available evidence, and then voted on by the working group members to give a score of the strength of agreement. Dr. Chinoy noted that there was a checklist included in the Supplementary Data section of the guideline to help follow the recommendations.
“The target audience for the guideline reflects the variety of clinicians caring for patients with IIM,” Dr. Chinoy said. So that is not just pediatric and adult rheumatologists, but also neurologists, dermatologists, respiratory physicians, oncologists, gastroenterologists, cardiologists, and of course other health care professionals. This includes rheumatology and neurology nurses, psychologists, speech and language therapists, and podiatrists, as well as rheumatology specialist pharmacists, physiotherapists, and occupational therapists.
With reference to the latter, Liza McCann, MBBS, who co-led the development of the guideline, said in a statement released by the BSR that the guideline “highlights the importance of exercise, led and monitored by specialist physiotherapists and occupational therapists.”
Dr. McCann, a consultant pediatric rheumatologist at Alder Hey Hospital, Liverpool, England, and Honorary Clinical Lecturer at the University of Liverpool, added that the guidelines also cover “the need to address psychological wellbeing as an integral part of treatment, in parallel with pharmacological therapies.”
Recommendation highlights
Some of the highlights of the recommendations include the use of high-dose glucocorticoids to manage skeletal muscle inflammation at the time of treatment induction, with specific guidance on the different doses to use in adults and in children. There also is guidance on the use of csDMARDs in both populations and what to use if there is refractory disease – with the strongest evidence supporting the use of intravenous immunoglobulin (IVIG) or cyclophosphamide, and possibly rituximab and abatacept.
“There is insufficient evidence to recommend JAK inhibition,” Dr. Chinoy said. The data search used to develop the guideline had a cutoff of October 2020, but even now there is only anecdotal evidence from case studies, he added.
Importantly, the guidelines recognize that childhood IIM differs from adult disease and call for children to be managed by pediatric specialists.
“Routine assessment of dysphagia should be considered in all patients,” Dr. Chinoy said, “so ask the question.” The recommendation is that a swallowing assessment should involve a speech and language therapist or gastroenterologist, and that IVIG be considered for active disease and dysphagia that is resistant to other treatments.
There also are recommendations to screen adult patients for interstitial lung disease, consider fracture risk, and screen adult patients for cancer if they have specific risk factors that include older age at onset, male gender, dysphagia, and rapid disease onset, among others.
Separate cancer screening guidelines on cards
“Around one in four patients with myositis will develop cancer within the 3 years either before or after myositis onset,” Alexander Oldroyd, MBChB, PhD, said in a separate presentation at the BSR annual meeting.
“It’s a hugely increased risk compared to the general population, and a great worry for patients,” he added. Exactly why there is an increased risk is not known, but “there’s a big link between the biological onset of cancer and myositis.”
Dr. Oldroyd, who is an NIHR Academic Clinical Lecturer at the University of Manchester in England and a coauthor of the BSR myositis guideline, is part of a special interest group set up by the International Myositis Assessment and Clinical Studies Group (IMACS) that is in the process of developing separate guidelines for cancer screening in people newly diagnosed with IIM.
The aim was to produce evidence-based recommendations that were both “pragmatic and practical,” that could help clinicians answer patient’s questions on their risk and how best and how often to screen them, Dr. Oldroyd explained. Importantly, IMACS has endeavored to create recommendations that should be applicable across different countries and health care systems.
“We had to acknowledge that there’s not a lot of evidence base there,” Dr. Oldroyd said, noting that he and colleagues conducted a systematic literature review and meta-analysis and used a Delphi process to draft 20 recommendations. These cover identifying risk factors for cancer in people with myositis and categorizing people into low, medium, and high-risk categories. The recommendations also cover what should constitute basic and enhanced screening, and how often someone should be screened.
Moreover, the authors make recommendations on the use of imaging modalities such as PET and CT scans, as well as upper and lower gastrointestinal endoscopy and naso-endoscopy.
“As rheumatologists, we don’t talk about cancer a lot,” Dr. Oldroyd said. “We pick up a lot of incidental cancers, but we don’t usually talk about cancer screening with patients.” That’s something that needs to change, he said.
“It’s important – just get it out in the open, talk to people about it,” Dr. Oldroyd said.
“Tell them what you’re wanting to do, how you’re wanting to investigate for it, clearly communicate their risk,” he said. “But also acknowledge the limited evidence as well, and clearly communicate the results.”
Dr. Chinoy acknowledged he had received fees for presentations (UCB, Biogen), consultancy (Alexion, Novartis, Eli Lilly, Orphazyme, AstraZeneca), or grant support (Eli Lilly, UCB) that had been paid via his institution for the purpose of furthering myositis research. Dr. Oldroyd had no conflicts of interest to disclose.
All patients with idiopathic inflammatory myopathies (IIM) should be screened for swallowing difficulties, according to the first evidence-based guideline to be produced.
The guideline, which has been developed by a working group of the British Society for Rheumatology (BSR), also advises that all diagnosed patients should have their myositis antibody levels checked and have their overall well-being assessed. Other recommendations for all patients include the use of glucocorticoids to reduce muscle inflammation and conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) for long-term treatment.
“Finally, now, we’re able to standardize the way we treat adults and children with IIM,” senior guideline author Hector Chinoy, PhD, said at the society’s annual meeting.
It has been a long labor of love, however, taking 4 years to get the guideline published, said Dr. Chinoy, professor of rheumatology and neuromuscular disease at the University of Manchester (England), and a consultant at Salford (England) Royal Hospital.
“We’re not covering diagnosis, classification, or the investigation of suspected IIM,” said Dr. Chinoy. Inclusion body myositis also is not included.
Altogether, there are 13 recommendations that have been developed using a PICO (patient or population, intervention, comparison, outcome) format, graded based on the quality of the available evidence, and then voted on by the working group members to give a score of the strength of agreement. Dr. Chinoy noted that there was a checklist included in the Supplementary Data section of the guideline to help follow the recommendations.
“The target audience for the guideline reflects the variety of clinicians caring for patients with IIM,” Dr. Chinoy said. So that is not just pediatric and adult rheumatologists, but also neurologists, dermatologists, respiratory physicians, oncologists, gastroenterologists, cardiologists, and of course other health care professionals. This includes rheumatology and neurology nurses, psychologists, speech and language therapists, and podiatrists, as well as rheumatology specialist pharmacists, physiotherapists, and occupational therapists.
With reference to the latter, Liza McCann, MBBS, who co-led the development of the guideline, said in a statement released by the BSR that the guideline “highlights the importance of exercise, led and monitored by specialist physiotherapists and occupational therapists.”
Dr. McCann, a consultant pediatric rheumatologist at Alder Hey Hospital, Liverpool, England, and Honorary Clinical Lecturer at the University of Liverpool, added that the guidelines also cover “the need to address psychological wellbeing as an integral part of treatment, in parallel with pharmacological therapies.”
Recommendation highlights
Some of the highlights of the recommendations include the use of high-dose glucocorticoids to manage skeletal muscle inflammation at the time of treatment induction, with specific guidance on the different doses to use in adults and in children. There also is guidance on the use of csDMARDs in both populations and what to use if there is refractory disease – with the strongest evidence supporting the use of intravenous immunoglobulin (IVIG) or cyclophosphamide, and possibly rituximab and abatacept.
“There is insufficient evidence to recommend JAK inhibition,” Dr. Chinoy said. The data search used to develop the guideline had a cutoff of October 2020, but even now there is only anecdotal evidence from case studies, he added.
Importantly, the guidelines recognize that childhood IIM differs from adult disease and call for children to be managed by pediatric specialists.
“Routine assessment of dysphagia should be considered in all patients,” Dr. Chinoy said, “so ask the question.” The recommendation is that a swallowing assessment should involve a speech and language therapist or gastroenterologist, and that IVIG be considered for active disease and dysphagia that is resistant to other treatments.
There also are recommendations to screen adult patients for interstitial lung disease, consider fracture risk, and screen adult patients for cancer if they have specific risk factors that include older age at onset, male gender, dysphagia, and rapid disease onset, among others.
Separate cancer screening guidelines on cards
“Around one in four patients with myositis will develop cancer within the 3 years either before or after myositis onset,” Alexander Oldroyd, MBChB, PhD, said in a separate presentation at the BSR annual meeting.
“It’s a hugely increased risk compared to the general population, and a great worry for patients,” he added. Exactly why there is an increased risk is not known, but “there’s a big link between the biological onset of cancer and myositis.”
Dr. Oldroyd, who is an NIHR Academic Clinical Lecturer at the University of Manchester in England and a coauthor of the BSR myositis guideline, is part of a special interest group set up by the International Myositis Assessment and Clinical Studies Group (IMACS) that is in the process of developing separate guidelines for cancer screening in people newly diagnosed with IIM.
The aim was to produce evidence-based recommendations that were both “pragmatic and practical,” that could help clinicians answer patient’s questions on their risk and how best and how often to screen them, Dr. Oldroyd explained. Importantly, IMACS has endeavored to create recommendations that should be applicable across different countries and health care systems.
“We had to acknowledge that there’s not a lot of evidence base there,” Dr. Oldroyd said, noting that he and colleagues conducted a systematic literature review and meta-analysis and used a Delphi process to draft 20 recommendations. These cover identifying risk factors for cancer in people with myositis and categorizing people into low, medium, and high-risk categories. The recommendations also cover what should constitute basic and enhanced screening, and how often someone should be screened.
Moreover, the authors make recommendations on the use of imaging modalities such as PET and CT scans, as well as upper and lower gastrointestinal endoscopy and naso-endoscopy.
“As rheumatologists, we don’t talk about cancer a lot,” Dr. Oldroyd said. “We pick up a lot of incidental cancers, but we don’t usually talk about cancer screening with patients.” That’s something that needs to change, he said.
“It’s important – just get it out in the open, talk to people about it,” Dr. Oldroyd said.
“Tell them what you’re wanting to do, how you’re wanting to investigate for it, clearly communicate their risk,” he said. “But also acknowledge the limited evidence as well, and clearly communicate the results.”
Dr. Chinoy acknowledged he had received fees for presentations (UCB, Biogen), consultancy (Alexion, Novartis, Eli Lilly, Orphazyme, AstraZeneca), or grant support (Eli Lilly, UCB) that had been paid via his institution for the purpose of furthering myositis research. Dr. Oldroyd had no conflicts of interest to disclose.
All patients with idiopathic inflammatory myopathies (IIM) should be screened for swallowing difficulties, according to the first evidence-based guideline to be produced.
The guideline, which has been developed by a working group of the British Society for Rheumatology (BSR), also advises that all diagnosed patients should have their myositis antibody levels checked and have their overall well-being assessed. Other recommendations for all patients include the use of glucocorticoids to reduce muscle inflammation and conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) for long-term treatment.
“Finally, now, we’re able to standardize the way we treat adults and children with IIM,” senior guideline author Hector Chinoy, PhD, said at the society’s annual meeting.
It has been a long labor of love, however, taking 4 years to get the guideline published, said Dr. Chinoy, professor of rheumatology and neuromuscular disease at the University of Manchester (England), and a consultant at Salford (England) Royal Hospital.
“We’re not covering diagnosis, classification, or the investigation of suspected IIM,” said Dr. Chinoy. Inclusion body myositis also is not included.
Altogether, there are 13 recommendations that have been developed using a PICO (patient or population, intervention, comparison, outcome) format, graded based on the quality of the available evidence, and then voted on by the working group members to give a score of the strength of agreement. Dr. Chinoy noted that there was a checklist included in the Supplementary Data section of the guideline to help follow the recommendations.
“The target audience for the guideline reflects the variety of clinicians caring for patients with IIM,” Dr. Chinoy said. So that is not just pediatric and adult rheumatologists, but also neurologists, dermatologists, respiratory physicians, oncologists, gastroenterologists, cardiologists, and of course other health care professionals. This includes rheumatology and neurology nurses, psychologists, speech and language therapists, and podiatrists, as well as rheumatology specialist pharmacists, physiotherapists, and occupational therapists.
With reference to the latter, Liza McCann, MBBS, who co-led the development of the guideline, said in a statement released by the BSR that the guideline “highlights the importance of exercise, led and monitored by specialist physiotherapists and occupational therapists.”
Dr. McCann, a consultant pediatric rheumatologist at Alder Hey Hospital, Liverpool, England, and Honorary Clinical Lecturer at the University of Liverpool, added that the guidelines also cover “the need to address psychological wellbeing as an integral part of treatment, in parallel with pharmacological therapies.”
Recommendation highlights
Some of the highlights of the recommendations include the use of high-dose glucocorticoids to manage skeletal muscle inflammation at the time of treatment induction, with specific guidance on the different doses to use in adults and in children. There also is guidance on the use of csDMARDs in both populations and what to use if there is refractory disease – with the strongest evidence supporting the use of intravenous immunoglobulin (IVIG) or cyclophosphamide, and possibly rituximab and abatacept.
“There is insufficient evidence to recommend JAK inhibition,” Dr. Chinoy said. The data search used to develop the guideline had a cutoff of October 2020, but even now there is only anecdotal evidence from case studies, he added.
Importantly, the guidelines recognize that childhood IIM differs from adult disease and call for children to be managed by pediatric specialists.
“Routine assessment of dysphagia should be considered in all patients,” Dr. Chinoy said, “so ask the question.” The recommendation is that a swallowing assessment should involve a speech and language therapist or gastroenterologist, and that IVIG be considered for active disease and dysphagia that is resistant to other treatments.
There also are recommendations to screen adult patients for interstitial lung disease, consider fracture risk, and screen adult patients for cancer if they have specific risk factors that include older age at onset, male gender, dysphagia, and rapid disease onset, among others.
Separate cancer screening guidelines on cards
“Around one in four patients with myositis will develop cancer within the 3 years either before or after myositis onset,” Alexander Oldroyd, MBChB, PhD, said in a separate presentation at the BSR annual meeting.
“It’s a hugely increased risk compared to the general population, and a great worry for patients,” he added. Exactly why there is an increased risk is not known, but “there’s a big link between the biological onset of cancer and myositis.”
Dr. Oldroyd, who is an NIHR Academic Clinical Lecturer at the University of Manchester in England and a coauthor of the BSR myositis guideline, is part of a special interest group set up by the International Myositis Assessment and Clinical Studies Group (IMACS) that is in the process of developing separate guidelines for cancer screening in people newly diagnosed with IIM.
The aim was to produce evidence-based recommendations that were both “pragmatic and practical,” that could help clinicians answer patient’s questions on their risk and how best and how often to screen them, Dr. Oldroyd explained. Importantly, IMACS has endeavored to create recommendations that should be applicable across different countries and health care systems.
“We had to acknowledge that there’s not a lot of evidence base there,” Dr. Oldroyd said, noting that he and colleagues conducted a systematic literature review and meta-analysis and used a Delphi process to draft 20 recommendations. These cover identifying risk factors for cancer in people with myositis and categorizing people into low, medium, and high-risk categories. The recommendations also cover what should constitute basic and enhanced screening, and how often someone should be screened.
Moreover, the authors make recommendations on the use of imaging modalities such as PET and CT scans, as well as upper and lower gastrointestinal endoscopy and naso-endoscopy.
“As rheumatologists, we don’t talk about cancer a lot,” Dr. Oldroyd said. “We pick up a lot of incidental cancers, but we don’t usually talk about cancer screening with patients.” That’s something that needs to change, he said.
“It’s important – just get it out in the open, talk to people about it,” Dr. Oldroyd said.
“Tell them what you’re wanting to do, how you’re wanting to investigate for it, clearly communicate their risk,” he said. “But also acknowledge the limited evidence as well, and clearly communicate the results.”
Dr. Chinoy acknowledged he had received fees for presentations (UCB, Biogen), consultancy (Alexion, Novartis, Eli Lilly, Orphazyme, AstraZeneca), or grant support (Eli Lilly, UCB) that had been paid via his institution for the purpose of furthering myositis research. Dr. Oldroyd had no conflicts of interest to disclose.
FROM BSR 2022