User login
Berdazimer gel under review at FDA for treating molluscum contagiosum
, the manufacturer announced.
If the submission is accepted by the FDA, the topical product could be approved in the first quarter of 2024, according to a press release from Novan, the manufacturer. If approved, it would be the first-in-class topical treatment for MC, the common, contagious viral skin infection that affects approximately six million individuals in the United States each year, most of them children aged 1-14 years, the statement noted. No FDA-approved therapies currently exist for the condition, which causes unsightly lesions on the face, trunk, limbs, and axillae that may persist untreated for a period of years.
The active ingredient in berdazimer gel 10.3% is berdazimer sodium, a nitric oxide–releasing agent. A 3.4% formulation is in development for the topical treatment of acne, according to the company.
The submission for FDA approval is based on data from the B-SIMPLE4 study, a phase 3 randomized trial of nearly 900 individuals with MC aged 6 months and older (mean age, 6.6 years), with 3-70 raised lesions. Participants were randomized to treatment with berdazimer gel 10.3% or a vehicle gel applied in a thin layer to all lesions once daily for 12 weeks. The results were published in JAMA Dermatology.
The primary outcome was complete clearance of all lesions. At 12 weeks, 32.4% of patients in the berdazimer group achieved this outcome vs. 19.7% of those in the vehicle group (P < .001). Overall adverse event rates were low in both groups; 4.1% of patients on berdazimer and 0.7% of those on the vehicle experienced adverse events that led to discontinuation of treatment. The most common adverse events across both groups were application-site pain and erythema, and most of these were mild or moderate.
, the manufacturer announced.
If the submission is accepted by the FDA, the topical product could be approved in the first quarter of 2024, according to a press release from Novan, the manufacturer. If approved, it would be the first-in-class topical treatment for MC, the common, contagious viral skin infection that affects approximately six million individuals in the United States each year, most of them children aged 1-14 years, the statement noted. No FDA-approved therapies currently exist for the condition, which causes unsightly lesions on the face, trunk, limbs, and axillae that may persist untreated for a period of years.
The active ingredient in berdazimer gel 10.3% is berdazimer sodium, a nitric oxide–releasing agent. A 3.4% formulation is in development for the topical treatment of acne, according to the company.
The submission for FDA approval is based on data from the B-SIMPLE4 study, a phase 3 randomized trial of nearly 900 individuals with MC aged 6 months and older (mean age, 6.6 years), with 3-70 raised lesions. Participants were randomized to treatment with berdazimer gel 10.3% or a vehicle gel applied in a thin layer to all lesions once daily for 12 weeks. The results were published in JAMA Dermatology.
The primary outcome was complete clearance of all lesions. At 12 weeks, 32.4% of patients in the berdazimer group achieved this outcome vs. 19.7% of those in the vehicle group (P < .001). Overall adverse event rates were low in both groups; 4.1% of patients on berdazimer and 0.7% of those on the vehicle experienced adverse events that led to discontinuation of treatment. The most common adverse events across both groups were application-site pain and erythema, and most of these were mild or moderate.
, the manufacturer announced.
If the submission is accepted by the FDA, the topical product could be approved in the first quarter of 2024, according to a press release from Novan, the manufacturer. If approved, it would be the first-in-class topical treatment for MC, the common, contagious viral skin infection that affects approximately six million individuals in the United States each year, most of them children aged 1-14 years, the statement noted. No FDA-approved therapies currently exist for the condition, which causes unsightly lesions on the face, trunk, limbs, and axillae that may persist untreated for a period of years.
The active ingredient in berdazimer gel 10.3% is berdazimer sodium, a nitric oxide–releasing agent. A 3.4% formulation is in development for the topical treatment of acne, according to the company.
The submission for FDA approval is based on data from the B-SIMPLE4 study, a phase 3 randomized trial of nearly 900 individuals with MC aged 6 months and older (mean age, 6.6 years), with 3-70 raised lesions. Participants were randomized to treatment with berdazimer gel 10.3% or a vehicle gel applied in a thin layer to all lesions once daily for 12 weeks. The results were published in JAMA Dermatology.
The primary outcome was complete clearance of all lesions. At 12 weeks, 32.4% of patients in the berdazimer group achieved this outcome vs. 19.7% of those in the vehicle group (P < .001). Overall adverse event rates were low in both groups; 4.1% of patients on berdazimer and 0.7% of those on the vehicle experienced adverse events that led to discontinuation of treatment. The most common adverse events across both groups were application-site pain and erythema, and most of these were mild or moderate.
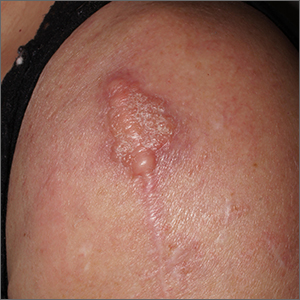
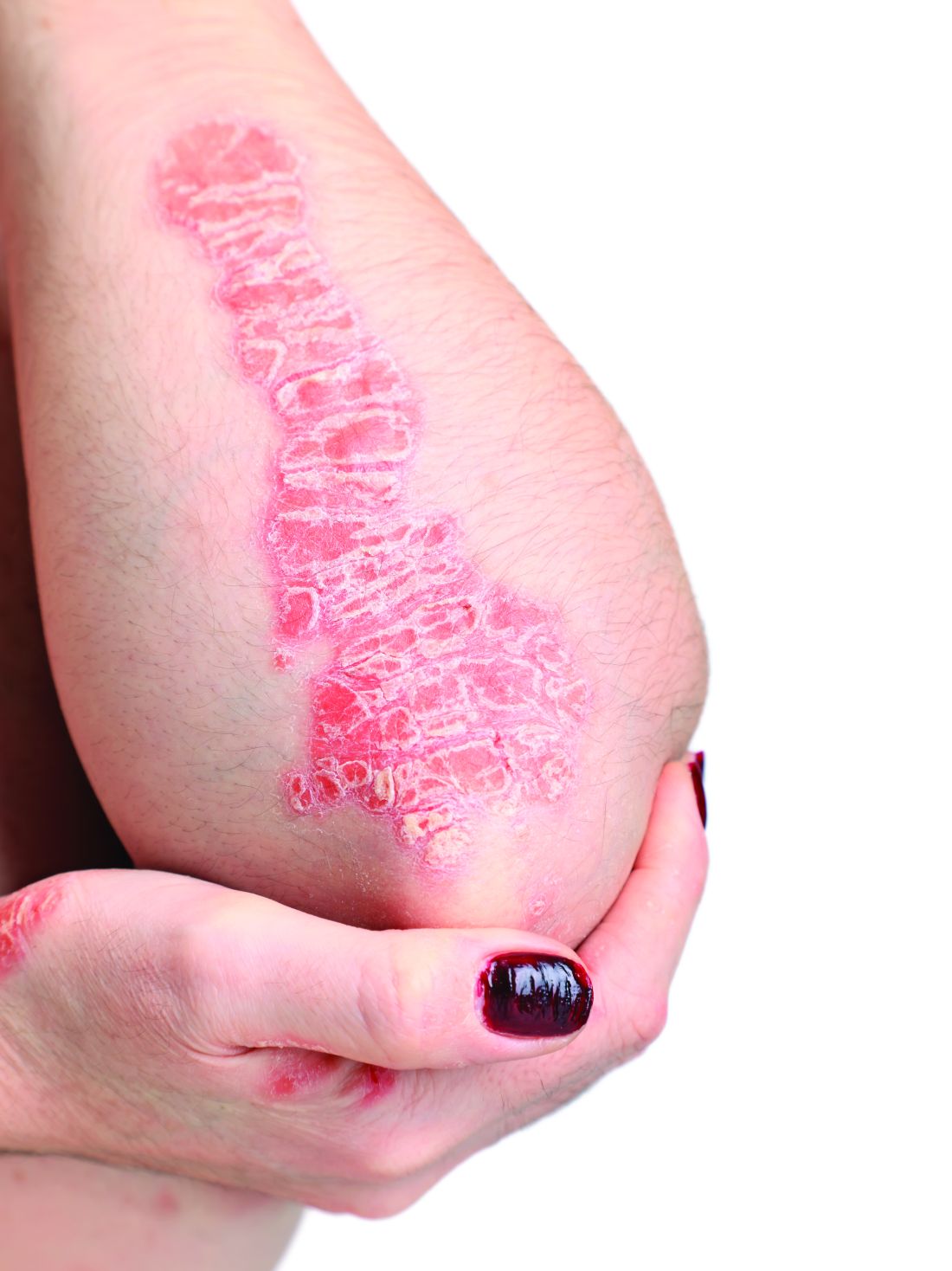
Scar overgrowth
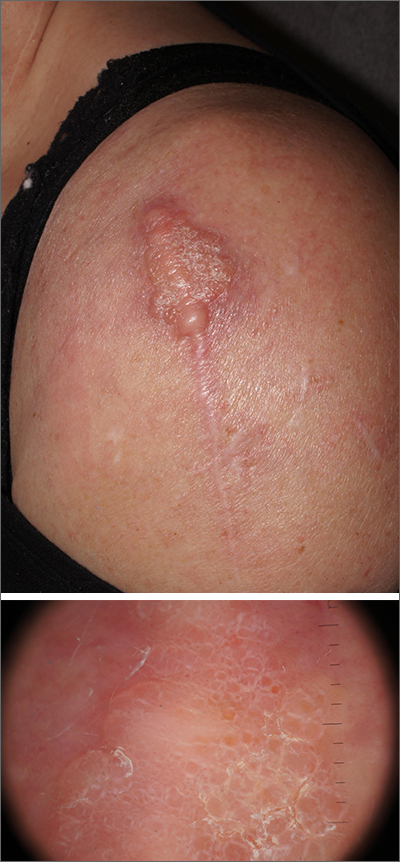
Dermatopathology was consistent with a diagnosis of cutaneous myxoma (CM). There are very few dermoscopic descriptions of CM in the literature, so diagnostic features are not established. However, the absence of more diagnostic features of basal cell carcinoma or squamous cell carcinoma (SCC) increases the likelihood of a rare diagnosis, such as CM.
CMs are rare benign neoplasms that manifest most commonly in young adults as small (< 1 cm) flesh-colored to blue papules on the head, neck, and trunk. The size of this particular CM was an outlier. CMs may be associated with Carney Complex (CNC), a rare inherited syndrome that has been linked to multiple endocrine neoplasias—namely, pituitary adenomas, testicular Sertoli cell tumors, thyroid tumors, and cardiac atrial myxomas.1 Additionally, in CNC, lentigines and multiple blue nevi develop on the skin and mucosal surfaces.
The differential diagnosis for a large, pink to flesh-colored nodule of this size includes benign histiocytoma, SCC, CM, and dermatofibrosarcoma protuberans. Benign histiocytomas and SCCs are much more common than CM. Clinical features only hint at the correct diagnosis, which must be made histologically.
Patients with CMs benefit from ongoing dermatology surveillance to monitor for the development of atypical nevi or new CMs. In this case, a wide excision with generous margins was planned with plastic surgery. (CMs have been reported to recur after surgery, which is why wide margins are essential.)
Additionally, 2 factors prompted an echocardiogram: the association between CMs and possible cardiac tumors and the patient’s need to undergo future orthopedic surgery under general anesthesia. No cardiac tumors were visible on echocardiogram. Thyroid imaging and genetic evaluation were planned but not completed.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Zou Y, Billings SD. Myxoid cutaneous tumors: a review. J Cutan Pathol. 2016;43:903-18. doi: 10.1111/cup.12749.

Dermatopathology was consistent with a diagnosis of cutaneous myxoma (CM). There are very few dermoscopic descriptions of CM in the literature, so diagnostic features are not established. However, the absence of more diagnostic features of basal cell carcinoma or squamous cell carcinoma (SCC) increases the likelihood of a rare diagnosis, such as CM.
CMs are rare benign neoplasms that manifest most commonly in young adults as small (< 1 cm) flesh-colored to blue papules on the head, neck, and trunk. The size of this particular CM was an outlier. CMs may be associated with Carney Complex (CNC), a rare inherited syndrome that has been linked to multiple endocrine neoplasias—namely, pituitary adenomas, testicular Sertoli cell tumors, thyroid tumors, and cardiac atrial myxomas.1 Additionally, in CNC, lentigines and multiple blue nevi develop on the skin and mucosal surfaces.
The differential diagnosis for a large, pink to flesh-colored nodule of this size includes benign histiocytoma, SCC, CM, and dermatofibrosarcoma protuberans. Benign histiocytomas and SCCs are much more common than CM. Clinical features only hint at the correct diagnosis, which must be made histologically.
Patients with CMs benefit from ongoing dermatology surveillance to monitor for the development of atypical nevi or new CMs. In this case, a wide excision with generous margins was planned with plastic surgery. (CMs have been reported to recur after surgery, which is why wide margins are essential.)
Additionally, 2 factors prompted an echocardiogram: the association between CMs and possible cardiac tumors and the patient’s need to undergo future orthopedic surgery under general anesthesia. No cardiac tumors were visible on echocardiogram. Thyroid imaging and genetic evaluation were planned but not completed.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.

Dermatopathology was consistent with a diagnosis of cutaneous myxoma (CM). There are very few dermoscopic descriptions of CM in the literature, so diagnostic features are not established. However, the absence of more diagnostic features of basal cell carcinoma or squamous cell carcinoma (SCC) increases the likelihood of a rare diagnosis, such as CM.
CMs are rare benign neoplasms that manifest most commonly in young adults as small (< 1 cm) flesh-colored to blue papules on the head, neck, and trunk. The size of this particular CM was an outlier. CMs may be associated with Carney Complex (CNC), a rare inherited syndrome that has been linked to multiple endocrine neoplasias—namely, pituitary adenomas, testicular Sertoli cell tumors, thyroid tumors, and cardiac atrial myxomas.1 Additionally, in CNC, lentigines and multiple blue nevi develop on the skin and mucosal surfaces.
The differential diagnosis for a large, pink to flesh-colored nodule of this size includes benign histiocytoma, SCC, CM, and dermatofibrosarcoma protuberans. Benign histiocytomas and SCCs are much more common than CM. Clinical features only hint at the correct diagnosis, which must be made histologically.
Patients with CMs benefit from ongoing dermatology surveillance to monitor for the development of atypical nevi or new CMs. In this case, a wide excision with generous margins was planned with plastic surgery. (CMs have been reported to recur after surgery, which is why wide margins are essential.)
Additionally, 2 factors prompted an echocardiogram: the association between CMs and possible cardiac tumors and the patient’s need to undergo future orthopedic surgery under general anesthesia. No cardiac tumors were visible on echocardiogram. Thyroid imaging and genetic evaluation were planned but not completed.
Photos and text for Photo Rounds Friday courtesy of Jonathan Karnes, MD (copyright retained). Dr. Karnes is the medical director of MDFMR Dermatology Services, Augusta, ME.
1. Zou Y, Billings SD. Myxoid cutaneous tumors: a review. J Cutan Pathol. 2016;43:903-18. doi: 10.1111/cup.12749.
1. Zou Y, Billings SD. Myxoid cutaneous tumors: a review. J Cutan Pathol. 2016;43:903-18. doi: 10.1111/cup.12749.
Surgeon’s license suspension spotlights hazards, ethics of live-streaming surgeries
potentially endangering patients. The surgeon has a large social media following.
In November, the State Medical Board of Ohio temporarily suspended the license of Katherine Roxanne Grawe, MD, who practices in the wealthy Columbus suburb of Powell.
Among other accusations of misconduct, the board stated that “during some videos/live-streams you engage in dialogue to respond to viewers’ online questions while the surgical procedure remains actively ongoing.”
One patient needed emergency treatment following liposuction and was diagnosed with a perforated bowel and serious bacterial infection.
“Despite liposuction being a blind surgery that requires awareness of the tip of the cannula to avoid injury, your attention to the camera meant at those moments you were not looking at the patient or palpating the location of the tip of the cannula,” the medical board said.
Neither Dr. Grawe nor her attorney responded to requests for comment.
Dr. Grawe, known as “Dr. Roxy,” has a popular TikTok account – now set to private – with 841,600 followers and 14.6 million likes. She has another 123,000 followers on her Instagram account, also now private.
The Columbus Dispatch reported that Dr. Grawe had previously been warned to protect patient privacy on social media. The board has yet to make a final decision regarding her license.
According to Columbus TV station WSYX, she said in a TikTok video, “We show our surgeries every single day on Snapchat. Patients get to decide if they want to be part of it. And if you do, you can watch your own surgery.”
The TV station quoted former patients who described surgical complications. One said: “I went to her because, I thought, from all of her social media that she uplifted women. That she helped women empower themselves. But she didn’t.”
Dallas plastic surgeon Rod J. Rohrich, MD, who has written about social-media best practices and has 430,000 followers on Instagram, said in an interview that many surgeons have been reprimanded by state medical boards for being distracted by social media during procedures.
“It is best not to do live-streaming unless it is an educational event to demonstrate techniques and technology with full informed consent of the patient. It should be a very well-rehearsed event for education,” he said.
Nurses also have been disciplined for inappropriate posts on social media. In December 2022, an Atlanta hospital announced that four nurses were no longer on the job after they appeared in a TikTok video in scrubs and revealed their “icks” regarding obstetric care.
“My ick is when you ask me how much the baby weighs,” one worker said in the video, “and it’s still ... in your hands.”
Plastic surgeon Christian J. Vercler, MD, of the University of Michigan, Ann Arbor, who’s studied social-media guidelines for surgeons, said in an interview that plastic surgery content on TikTok has “blown up” in recent years.
“Five years or so ago, it was Snapchat where I saw a lot of inappropriate things posted by surgeons,” Dr. Vercler said in an interview. “That may still be happening on Snapchat, but I actually don’t ever use that platform anymore, and neither do my trainees.”
Dr. Vercler cautioned colleagues to consider their motivations for live-streaming surgery and to think about whether they can fully focus on the patient.
“There are many potential distractions in the OR. We get pages, phone calls, nurses asking us questions, anesthesiologists trying to talk to us. Social media is just one more thing competing for the surgeon’s attention,” he said. “Every surgeon should strive to eliminate unnecessary or unavoidable distractions, so the question becomes, ‘who is best being served by me focusing my attention on recording this operation on someone’s phone so we can post it on social media? Is it the patient?’ ”
Dr. Vercler added, “There are many, many plastic surgeons using social media as the powerful platform that it is to build their brands, to connect with potential patients, and to educate the public about what they do. I believe that most are doing this in a way that is respectful to patients and doesn’t exploit patients for the surgeon’s benefit.
“Unfortunately,” he concluded, “there are some who do see patients as merely instruments by which they can achieve fame, notoriety, and wealth.”
Dr. Rohrich and Dr. Vercler disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
potentially endangering patients. The surgeon has a large social media following.
In November, the State Medical Board of Ohio temporarily suspended the license of Katherine Roxanne Grawe, MD, who practices in the wealthy Columbus suburb of Powell.
Among other accusations of misconduct, the board stated that “during some videos/live-streams you engage in dialogue to respond to viewers’ online questions while the surgical procedure remains actively ongoing.”
One patient needed emergency treatment following liposuction and was diagnosed with a perforated bowel and serious bacterial infection.
“Despite liposuction being a blind surgery that requires awareness of the tip of the cannula to avoid injury, your attention to the camera meant at those moments you were not looking at the patient or palpating the location of the tip of the cannula,” the medical board said.
Neither Dr. Grawe nor her attorney responded to requests for comment.
Dr. Grawe, known as “Dr. Roxy,” has a popular TikTok account – now set to private – with 841,600 followers and 14.6 million likes. She has another 123,000 followers on her Instagram account, also now private.
The Columbus Dispatch reported that Dr. Grawe had previously been warned to protect patient privacy on social media. The board has yet to make a final decision regarding her license.
According to Columbus TV station WSYX, she said in a TikTok video, “We show our surgeries every single day on Snapchat. Patients get to decide if they want to be part of it. And if you do, you can watch your own surgery.”
The TV station quoted former patients who described surgical complications. One said: “I went to her because, I thought, from all of her social media that she uplifted women. That she helped women empower themselves. But she didn’t.”
Dallas plastic surgeon Rod J. Rohrich, MD, who has written about social-media best practices and has 430,000 followers on Instagram, said in an interview that many surgeons have been reprimanded by state medical boards for being distracted by social media during procedures.
“It is best not to do live-streaming unless it is an educational event to demonstrate techniques and technology with full informed consent of the patient. It should be a very well-rehearsed event for education,” he said.
Nurses also have been disciplined for inappropriate posts on social media. In December 2022, an Atlanta hospital announced that four nurses were no longer on the job after they appeared in a TikTok video in scrubs and revealed their “icks” regarding obstetric care.
“My ick is when you ask me how much the baby weighs,” one worker said in the video, “and it’s still ... in your hands.”
Plastic surgeon Christian J. Vercler, MD, of the University of Michigan, Ann Arbor, who’s studied social-media guidelines for surgeons, said in an interview that plastic surgery content on TikTok has “blown up” in recent years.
“Five years or so ago, it was Snapchat where I saw a lot of inappropriate things posted by surgeons,” Dr. Vercler said in an interview. “That may still be happening on Snapchat, but I actually don’t ever use that platform anymore, and neither do my trainees.”
Dr. Vercler cautioned colleagues to consider their motivations for live-streaming surgery and to think about whether they can fully focus on the patient.
“There are many potential distractions in the OR. We get pages, phone calls, nurses asking us questions, anesthesiologists trying to talk to us. Social media is just one more thing competing for the surgeon’s attention,” he said. “Every surgeon should strive to eliminate unnecessary or unavoidable distractions, so the question becomes, ‘who is best being served by me focusing my attention on recording this operation on someone’s phone so we can post it on social media? Is it the patient?’ ”
Dr. Vercler added, “There are many, many plastic surgeons using social media as the powerful platform that it is to build their brands, to connect with potential patients, and to educate the public about what they do. I believe that most are doing this in a way that is respectful to patients and doesn’t exploit patients for the surgeon’s benefit.
“Unfortunately,” he concluded, “there are some who do see patients as merely instruments by which they can achieve fame, notoriety, and wealth.”
Dr. Rohrich and Dr. Vercler disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
potentially endangering patients. The surgeon has a large social media following.
In November, the State Medical Board of Ohio temporarily suspended the license of Katherine Roxanne Grawe, MD, who practices in the wealthy Columbus suburb of Powell.
Among other accusations of misconduct, the board stated that “during some videos/live-streams you engage in dialogue to respond to viewers’ online questions while the surgical procedure remains actively ongoing.”
One patient needed emergency treatment following liposuction and was diagnosed with a perforated bowel and serious bacterial infection.
“Despite liposuction being a blind surgery that requires awareness of the tip of the cannula to avoid injury, your attention to the camera meant at those moments you were not looking at the patient or palpating the location of the tip of the cannula,” the medical board said.
Neither Dr. Grawe nor her attorney responded to requests for comment.
Dr. Grawe, known as “Dr. Roxy,” has a popular TikTok account – now set to private – with 841,600 followers and 14.6 million likes. She has another 123,000 followers on her Instagram account, also now private.
The Columbus Dispatch reported that Dr. Grawe had previously been warned to protect patient privacy on social media. The board has yet to make a final decision regarding her license.
According to Columbus TV station WSYX, she said in a TikTok video, “We show our surgeries every single day on Snapchat. Patients get to decide if they want to be part of it. And if you do, you can watch your own surgery.”
The TV station quoted former patients who described surgical complications. One said: “I went to her because, I thought, from all of her social media that she uplifted women. That she helped women empower themselves. But she didn’t.”
Dallas plastic surgeon Rod J. Rohrich, MD, who has written about social-media best practices and has 430,000 followers on Instagram, said in an interview that many surgeons have been reprimanded by state medical boards for being distracted by social media during procedures.
“It is best not to do live-streaming unless it is an educational event to demonstrate techniques and technology with full informed consent of the patient. It should be a very well-rehearsed event for education,” he said.
Nurses also have been disciplined for inappropriate posts on social media. In December 2022, an Atlanta hospital announced that four nurses were no longer on the job after they appeared in a TikTok video in scrubs and revealed their “icks” regarding obstetric care.
“My ick is when you ask me how much the baby weighs,” one worker said in the video, “and it’s still ... in your hands.”
Plastic surgeon Christian J. Vercler, MD, of the University of Michigan, Ann Arbor, who’s studied social-media guidelines for surgeons, said in an interview that plastic surgery content on TikTok has “blown up” in recent years.
“Five years or so ago, it was Snapchat where I saw a lot of inappropriate things posted by surgeons,” Dr. Vercler said in an interview. “That may still be happening on Snapchat, but I actually don’t ever use that platform anymore, and neither do my trainees.”
Dr. Vercler cautioned colleagues to consider their motivations for live-streaming surgery and to think about whether they can fully focus on the patient.
“There are many potential distractions in the OR. We get pages, phone calls, nurses asking us questions, anesthesiologists trying to talk to us. Social media is just one more thing competing for the surgeon’s attention,” he said. “Every surgeon should strive to eliminate unnecessary or unavoidable distractions, so the question becomes, ‘who is best being served by me focusing my attention on recording this operation on someone’s phone so we can post it on social media? Is it the patient?’ ”
Dr. Vercler added, “There are many, many plastic surgeons using social media as the powerful platform that it is to build their brands, to connect with potential patients, and to educate the public about what they do. I believe that most are doing this in a way that is respectful to patients and doesn’t exploit patients for the surgeon’s benefit.
“Unfortunately,” he concluded, “there are some who do see patients as merely instruments by which they can achieve fame, notoriety, and wealth.”
Dr. Rohrich and Dr. Vercler disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
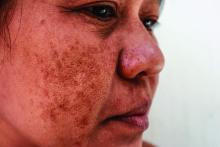
Long-term maintenance required in melasma patients
SAN DIEGO –
“They need to understand that melasma is going to require long-term maintenance,” Dr. Ortiz, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium.
Hydroquinone is a mainstay of melasma therapy, but instead of the commonly used 4% formulation, she prefers to use 12% hydroquinone with 6% kojic acid in VersaBase cream. “It’s a high concentration but the VersaBase makes it more tolerable,” she said. “I have patients take a pea-sized amount and mix it in a regular moisturizer. It’s too strong to spot treat, so it goes on the whole face.”
Mindful that chronic hydroquinone use can cause ochronosis (permanent darkening), she has patients alternate with a nonhydroquinone bleaching agent such as lignin peroxidase, oligopeptide, Lytera, Melaplex, 4-n-butylresorcinol, Cysteamine cream, tranexamic acid, or oral antioxidants. In a study sponsored by SkinMedica, investigators conducted a randomized, double-blind, half-face study in females with moderate to severe facial hyperpigmentation to assess the efficacy and tolerability of three new skin brightener formulations containing SMA-432, a prostaglandin E2 inhibitor, compared with 4% hydroquinone. They found that the nonhydroquinone skin formulations were better tolerated and were just as effective as 4% hydroquinone.
Chemical peels and laser treatments
Chemical peels are another treatment option for melasma, but Dr. Ortiz prefers glycolic peels over salicylic and other peels, “because there is no downtime,” she said.
As for laser-based approaches, melasma patients respond best to low energy devices such as the 1,927-nm fractional diode laser at a 3.75% density. “This also can increase the skin permeability of topicals, so when you’re combining it with hydroquinone it can be more effective,” she said.
In an observational study of 27 women with refractory melasma, with phototypes II-V, New York City–based dermatologist Arielle Kauvar, MD, combined microdermabrasion with the Q-switched Nd:YAG laser. “The settings she used were very low fluence, so there was no clinical endpoint or no whitening,” said Dr. Ortiz, vice president of the American Society for Laser Medicine and Surgery (ASLMS). Specifically, Dr. Kauvar used the laser at 1.6-2 J/cm2 with a 5- or 6-mm spot size immediately following microdermabrasion every 4 weeks; Patients received an average of 2.6 treatments, and were assessed 3-12 months after the last treatment. Study participants were on a standard skin care regimen of a broad spectrum sunscreen, hydroquinone, and tretinoin or vitamin C.
Most of the patients showed at least 50% clearance of melasma 1 month after the first treatment, and 81% showed more than 75% clearance of melasma; remission lasted at least 6 months.
“I personally prefer to use picosecond over Q-switched lasers, because they deliver the energy faster, and you can use a 1,064-nm picosecond laser that is safe in all skin types,” Dr. Ortiz said. “There is minimal downtime, and it doesn’t require anesthesia. You have to consider these things when you’re treating melasma, because this usually requires monthly treatments. If you do something that requires a week of downtime every month, it’s not practical for patients.”
In a study published in 2021, Dr. Ortiz and Tanya Greywal, MD, used three passes of the 1,064-nm Nd:YAG laser to treat melasma in 10 patients with skin types II-V. The device had a 650-microsecond pulse duration, a 6-mm spot size, and an energy mode of 11-14 J/cm2. The researchers observed a mean melasma improvement of 26%-50% as early as 3 weeks. “There was no downtime, and no anesthesia was required,” Dr. Ortiz said.
Researchers have discovered a vascular component to melasma, which may have treatment implications. Houston-based dermatologist Paul M. Friedman, MD, and his colleagues used spectrocolorimetry to detect an underlying prominent vascular component in a retrospective review of 11 patients with melasma, with skin types II-IV. They determined that melasma lesions exhibiting subtle or subclinical telangiectatic erythema may be improved by combining vascular-targeted laser therapy with fractional low-powered diode laser therapy.
“So, combining a vascular laser with a 1,927-nm fractional diode laser showed more improvement than with just the diode laser alone,” said Dr. Ortiz, who was not involved with the analysis.
To optimize results following the laser treatment of melasma, she uses one application of clobetasol immediately after the procedure. “This can help reduce swelling and inflammation to decrease the risk of postinflammatory hyperpigmentation,” she said. “You can also use a skin cooling system like Cryomodulation for controlled cooling.”
Tranexamic acid and PLE
Another strategy for melasma patients involves oral treatment with extract of Polypodium leucotomos (PLE), a fern from the Polypodiaceae family with antioxidant properties that has been shown to be photoprotective against UVA and UVB radiation. “I explain to my patients that it’s like an internal sunscreen,” Dr. Ortiz said. “It does not replace your external sunscreen, but it adds extra protection.”
In a pilot placebo-controlled study of patients with melasma on their normal regimen of hydroquinone and sunscreen, 40 Asian patients with melasma were randomized to receive either oral PLE supplementation or placebo for 12 weeks. The authors found that PLE significantly improved and accelerated the outcome reached with hydroquinone and sunscreen from about the first month of treatment, compared with placebo.
Dr. Ortiz discussed the role of oral tranexamic acid, an antifibrinolytic, procoagulant agent that is approved by the Food and Drug Administration for the treatment of menorrhagia and to prevent hemorrhage in patients with hemophilia undergoing tooth extractions. “This is a game changer for melasma treatment,” she said, but its use has been limited by the risk for thromboembolism.
In a study of 561 patients with melasma, 90% improved after a median treatment duration of 4 months, and only 7% had side effects, most commonly abdominal bloating and pain. Treatment was discontinued in one patient who developed a deep vein thrombosis, and was diagnosed with familial protein S deficiency.
The daily dosing of tranexamic acid for menorrhagia is 3,900 mg daily, while the dose for treating melasma has ranged from 500 mg to 1,500 mg per day, Dr. Ortiz said. It’s available as a 650-mg tablet in the United States. “I prescribe 325 mg twice a day, but studies have shown that 650 mg once a day is just as effective,” she said.
Prior to prescribing tranexamic acid, Dr. Ortiz does not order labs, but she performs an extensive history of current illness and does not prescribe it in patients with an increased risk of clotting, including people who smoke and those who take oral contraceptives or are on hormone supplementation. Use is also contraindicated in people with a current malignancy, those with a history of stroke or DVT, and those who have any clotting disorder.
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies. She is cochair of the Masters of Aesthetics Symposium.
SAN DIEGO –
“They need to understand that melasma is going to require long-term maintenance,” Dr. Ortiz, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium.
Hydroquinone is a mainstay of melasma therapy, but instead of the commonly used 4% formulation, she prefers to use 12% hydroquinone with 6% kojic acid in VersaBase cream. “It’s a high concentration but the VersaBase makes it more tolerable,” she said. “I have patients take a pea-sized amount and mix it in a regular moisturizer. It’s too strong to spot treat, so it goes on the whole face.”
Mindful that chronic hydroquinone use can cause ochronosis (permanent darkening), she has patients alternate with a nonhydroquinone bleaching agent such as lignin peroxidase, oligopeptide, Lytera, Melaplex, 4-n-butylresorcinol, Cysteamine cream, tranexamic acid, or oral antioxidants. In a study sponsored by SkinMedica, investigators conducted a randomized, double-blind, half-face study in females with moderate to severe facial hyperpigmentation to assess the efficacy and tolerability of three new skin brightener formulations containing SMA-432, a prostaglandin E2 inhibitor, compared with 4% hydroquinone. They found that the nonhydroquinone skin formulations were better tolerated and were just as effective as 4% hydroquinone.
Chemical peels and laser treatments
Chemical peels are another treatment option for melasma, but Dr. Ortiz prefers glycolic peels over salicylic and other peels, “because there is no downtime,” she said.
As for laser-based approaches, melasma patients respond best to low energy devices such as the 1,927-nm fractional diode laser at a 3.75% density. “This also can increase the skin permeability of topicals, so when you’re combining it with hydroquinone it can be more effective,” she said.
In an observational study of 27 women with refractory melasma, with phototypes II-V, New York City–based dermatologist Arielle Kauvar, MD, combined microdermabrasion with the Q-switched Nd:YAG laser. “The settings she used were very low fluence, so there was no clinical endpoint or no whitening,” said Dr. Ortiz, vice president of the American Society for Laser Medicine and Surgery (ASLMS). Specifically, Dr. Kauvar used the laser at 1.6-2 J/cm2 with a 5- or 6-mm spot size immediately following microdermabrasion every 4 weeks; Patients received an average of 2.6 treatments, and were assessed 3-12 months after the last treatment. Study participants were on a standard skin care regimen of a broad spectrum sunscreen, hydroquinone, and tretinoin or vitamin C.
Most of the patients showed at least 50% clearance of melasma 1 month after the first treatment, and 81% showed more than 75% clearance of melasma; remission lasted at least 6 months.
“I personally prefer to use picosecond over Q-switched lasers, because they deliver the energy faster, and you can use a 1,064-nm picosecond laser that is safe in all skin types,” Dr. Ortiz said. “There is minimal downtime, and it doesn’t require anesthesia. You have to consider these things when you’re treating melasma, because this usually requires monthly treatments. If you do something that requires a week of downtime every month, it’s not practical for patients.”
In a study published in 2021, Dr. Ortiz and Tanya Greywal, MD, used three passes of the 1,064-nm Nd:YAG laser to treat melasma in 10 patients with skin types II-V. The device had a 650-microsecond pulse duration, a 6-mm spot size, and an energy mode of 11-14 J/cm2. The researchers observed a mean melasma improvement of 26%-50% as early as 3 weeks. “There was no downtime, and no anesthesia was required,” Dr. Ortiz said.
Researchers have discovered a vascular component to melasma, which may have treatment implications. Houston-based dermatologist Paul M. Friedman, MD, and his colleagues used spectrocolorimetry to detect an underlying prominent vascular component in a retrospective review of 11 patients with melasma, with skin types II-IV. They determined that melasma lesions exhibiting subtle or subclinical telangiectatic erythema may be improved by combining vascular-targeted laser therapy with fractional low-powered diode laser therapy.
“So, combining a vascular laser with a 1,927-nm fractional diode laser showed more improvement than with just the diode laser alone,” said Dr. Ortiz, who was not involved with the analysis.
To optimize results following the laser treatment of melasma, she uses one application of clobetasol immediately after the procedure. “This can help reduce swelling and inflammation to decrease the risk of postinflammatory hyperpigmentation,” she said. “You can also use a skin cooling system like Cryomodulation for controlled cooling.”
Tranexamic acid and PLE
Another strategy for melasma patients involves oral treatment with extract of Polypodium leucotomos (PLE), a fern from the Polypodiaceae family with antioxidant properties that has been shown to be photoprotective against UVA and UVB radiation. “I explain to my patients that it’s like an internal sunscreen,” Dr. Ortiz said. “It does not replace your external sunscreen, but it adds extra protection.”
In a pilot placebo-controlled study of patients with melasma on their normal regimen of hydroquinone and sunscreen, 40 Asian patients with melasma were randomized to receive either oral PLE supplementation or placebo for 12 weeks. The authors found that PLE significantly improved and accelerated the outcome reached with hydroquinone and sunscreen from about the first month of treatment, compared with placebo.
Dr. Ortiz discussed the role of oral tranexamic acid, an antifibrinolytic, procoagulant agent that is approved by the Food and Drug Administration for the treatment of menorrhagia and to prevent hemorrhage in patients with hemophilia undergoing tooth extractions. “This is a game changer for melasma treatment,” she said, but its use has been limited by the risk for thromboembolism.
In a study of 561 patients with melasma, 90% improved after a median treatment duration of 4 months, and only 7% had side effects, most commonly abdominal bloating and pain. Treatment was discontinued in one patient who developed a deep vein thrombosis, and was diagnosed with familial protein S deficiency.
The daily dosing of tranexamic acid for menorrhagia is 3,900 mg daily, while the dose for treating melasma has ranged from 500 mg to 1,500 mg per day, Dr. Ortiz said. It’s available as a 650-mg tablet in the United States. “I prescribe 325 mg twice a day, but studies have shown that 650 mg once a day is just as effective,” she said.
Prior to prescribing tranexamic acid, Dr. Ortiz does not order labs, but she performs an extensive history of current illness and does not prescribe it in patients with an increased risk of clotting, including people who smoke and those who take oral contraceptives or are on hormone supplementation. Use is also contraindicated in people with a current malignancy, those with a history of stroke or DVT, and those who have any clotting disorder.
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies. She is cochair of the Masters of Aesthetics Symposium.
SAN DIEGO –
“They need to understand that melasma is going to require long-term maintenance,” Dr. Ortiz, director of laser and cosmetic dermatology at the University of California, San Diego, said at the annual Masters of Aesthetics Symposium.
Hydroquinone is a mainstay of melasma therapy, but instead of the commonly used 4% formulation, she prefers to use 12% hydroquinone with 6% kojic acid in VersaBase cream. “It’s a high concentration but the VersaBase makes it more tolerable,” she said. “I have patients take a pea-sized amount and mix it in a regular moisturizer. It’s too strong to spot treat, so it goes on the whole face.”
Mindful that chronic hydroquinone use can cause ochronosis (permanent darkening), she has patients alternate with a nonhydroquinone bleaching agent such as lignin peroxidase, oligopeptide, Lytera, Melaplex, 4-n-butylresorcinol, Cysteamine cream, tranexamic acid, or oral antioxidants. In a study sponsored by SkinMedica, investigators conducted a randomized, double-blind, half-face study in females with moderate to severe facial hyperpigmentation to assess the efficacy and tolerability of three new skin brightener formulations containing SMA-432, a prostaglandin E2 inhibitor, compared with 4% hydroquinone. They found that the nonhydroquinone skin formulations were better tolerated and were just as effective as 4% hydroquinone.
Chemical peels and laser treatments
Chemical peels are another treatment option for melasma, but Dr. Ortiz prefers glycolic peels over salicylic and other peels, “because there is no downtime,” she said.
As for laser-based approaches, melasma patients respond best to low energy devices such as the 1,927-nm fractional diode laser at a 3.75% density. “This also can increase the skin permeability of topicals, so when you’re combining it with hydroquinone it can be more effective,” she said.
In an observational study of 27 women with refractory melasma, with phototypes II-V, New York City–based dermatologist Arielle Kauvar, MD, combined microdermabrasion with the Q-switched Nd:YAG laser. “The settings she used were very low fluence, so there was no clinical endpoint or no whitening,” said Dr. Ortiz, vice president of the American Society for Laser Medicine and Surgery (ASLMS). Specifically, Dr. Kauvar used the laser at 1.6-2 J/cm2 with a 5- or 6-mm spot size immediately following microdermabrasion every 4 weeks; Patients received an average of 2.6 treatments, and were assessed 3-12 months after the last treatment. Study participants were on a standard skin care regimen of a broad spectrum sunscreen, hydroquinone, and tretinoin or vitamin C.
Most of the patients showed at least 50% clearance of melasma 1 month after the first treatment, and 81% showed more than 75% clearance of melasma; remission lasted at least 6 months.
“I personally prefer to use picosecond over Q-switched lasers, because they deliver the energy faster, and you can use a 1,064-nm picosecond laser that is safe in all skin types,” Dr. Ortiz said. “There is minimal downtime, and it doesn’t require anesthesia. You have to consider these things when you’re treating melasma, because this usually requires monthly treatments. If you do something that requires a week of downtime every month, it’s not practical for patients.”
In a study published in 2021, Dr. Ortiz and Tanya Greywal, MD, used three passes of the 1,064-nm Nd:YAG laser to treat melasma in 10 patients with skin types II-V. The device had a 650-microsecond pulse duration, a 6-mm spot size, and an energy mode of 11-14 J/cm2. The researchers observed a mean melasma improvement of 26%-50% as early as 3 weeks. “There was no downtime, and no anesthesia was required,” Dr. Ortiz said.
Researchers have discovered a vascular component to melasma, which may have treatment implications. Houston-based dermatologist Paul M. Friedman, MD, and his colleagues used spectrocolorimetry to detect an underlying prominent vascular component in a retrospective review of 11 patients with melasma, with skin types II-IV. They determined that melasma lesions exhibiting subtle or subclinical telangiectatic erythema may be improved by combining vascular-targeted laser therapy with fractional low-powered diode laser therapy.
“So, combining a vascular laser with a 1,927-nm fractional diode laser showed more improvement than with just the diode laser alone,” said Dr. Ortiz, who was not involved with the analysis.
To optimize results following the laser treatment of melasma, she uses one application of clobetasol immediately after the procedure. “This can help reduce swelling and inflammation to decrease the risk of postinflammatory hyperpigmentation,” she said. “You can also use a skin cooling system like Cryomodulation for controlled cooling.”
Tranexamic acid and PLE
Another strategy for melasma patients involves oral treatment with extract of Polypodium leucotomos (PLE), a fern from the Polypodiaceae family with antioxidant properties that has been shown to be photoprotective against UVA and UVB radiation. “I explain to my patients that it’s like an internal sunscreen,” Dr. Ortiz said. “It does not replace your external sunscreen, but it adds extra protection.”
In a pilot placebo-controlled study of patients with melasma on their normal regimen of hydroquinone and sunscreen, 40 Asian patients with melasma were randomized to receive either oral PLE supplementation or placebo for 12 weeks. The authors found that PLE significantly improved and accelerated the outcome reached with hydroquinone and sunscreen from about the first month of treatment, compared with placebo.
Dr. Ortiz discussed the role of oral tranexamic acid, an antifibrinolytic, procoagulant agent that is approved by the Food and Drug Administration for the treatment of menorrhagia and to prevent hemorrhage in patients with hemophilia undergoing tooth extractions. “This is a game changer for melasma treatment,” she said, but its use has been limited by the risk for thromboembolism.
In a study of 561 patients with melasma, 90% improved after a median treatment duration of 4 months, and only 7% had side effects, most commonly abdominal bloating and pain. Treatment was discontinued in one patient who developed a deep vein thrombosis, and was diagnosed with familial protein S deficiency.
The daily dosing of tranexamic acid for menorrhagia is 3,900 mg daily, while the dose for treating melasma has ranged from 500 mg to 1,500 mg per day, Dr. Ortiz said. It’s available as a 650-mg tablet in the United States. “I prescribe 325 mg twice a day, but studies have shown that 650 mg once a day is just as effective,” she said.
Prior to prescribing tranexamic acid, Dr. Ortiz does not order labs, but she performs an extensive history of current illness and does not prescribe it in patients with an increased risk of clotting, including people who smoke and those who take oral contraceptives or are on hormone supplementation. Use is also contraindicated in people with a current malignancy, those with a history of stroke or DVT, and those who have any clotting disorder.
Dr. Ortiz disclosed having financial relationships with several pharmaceutical and device companies. She is cochair of the Masters of Aesthetics Symposium.
AT MOAS 2022
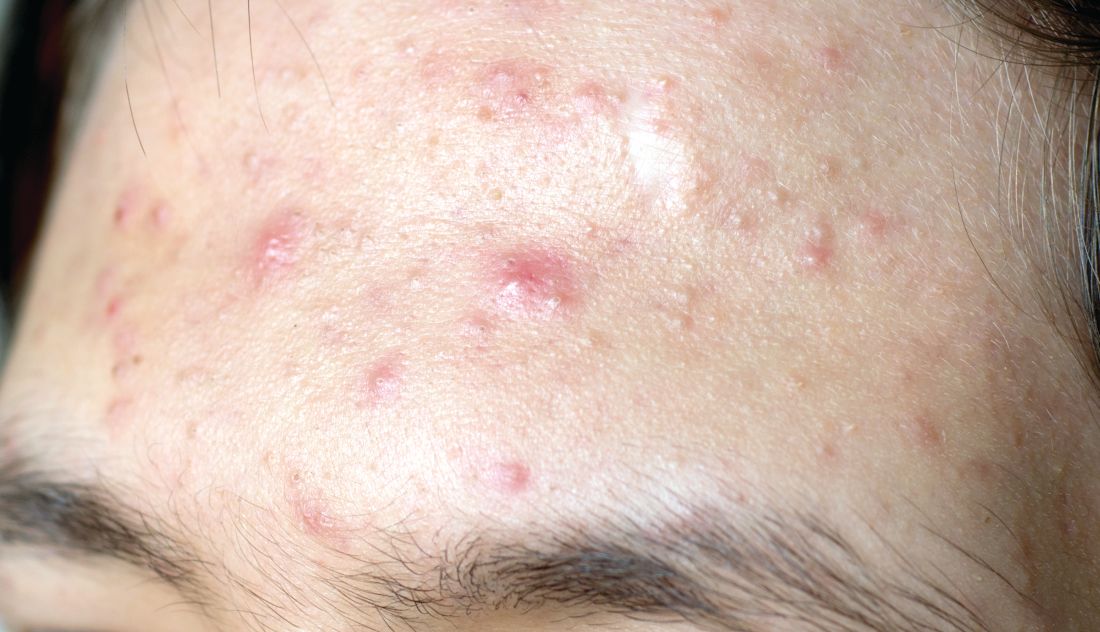
Topical treatment options for acne continue to expand
SAN DIEGO – , according to Lawrence F. Eichenfield, MD.
The product, known as IDP-126 and being developed by Ortho Dermatologics, is a fixed dose triple combination of clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% being evaluated in patients nine years of age and older. According to a 2021 press release from the company, results from a second 12-week pivotal phase 3 trial showed a treatment success of 50.5% and 20.5% for IDP-126 and its vehicle, respectively, along with significant changes from baseline in inflammatory lesion count and non-inflammatory lesion count.
More recently, researchers led by Linda Stein Gold, MD, conducted a 12-week multicenter, randomized, double-blind study of IDP-126 in 741 children, adolescents, and adults with moderate to severe acne. They reported 52.5% of patients treated with IDP-126 gel achieved treatment success by week 12, with over 70% reduction in inflammatory and noninflammatory lesions.
“This will be interesting to follow as it moves along,” Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, said at the annual Masters of Aesthetics Symposium in a presentation on the newest topical acne treatments.
“If approved, we probably will be able to decrease our need for systemic therapies in some individuals,” he said. “It’s something that may become important in practices that mix and match between medical and procedural or surgical approaches to acne.”
Dr. Eichenfield highlighted other products for the topical treatment of acne:
- Trifarotene cream 0.005% (Aklief). In 2019, Food and Drug Administration approval made trifarotene cream the first new retinoid indicated for acne in several decades. It is indicated for the topical treatment of acne vulgaris in patients age 9 and older and has been studied in acne of the face, chest, and back.
- Tazarotene lotion 0.045% (Arazlo). The 0.1% formulation of tazarotene is commonly used for acne, but it can cause skin irritation, dryness, and erythema. The new 0.045% formulation was developed in a three-dimensional mesh matrix, with ingredients from an oil-in-water emulsion. “Many of the new acne products come with a background of vehicle delivery systems that minimize the concentration gradient, so it decreases irritation,” said Dr. Eichenfield, one of the authors of a 2021 review article on the management of acne vulgaris in JAMA. “This has very good efficacy without the traditional irritation of other tazarotene products,” Dr. Eichenfield said.
- Minocycline 4% topical foam (Amzeeq). The 2019 U.S. approval marked the first and so far only topical minocycline prescription treatment for acne. “Its hydrophobic composition allows for stable and efficient delivery of inherently unstable pharmaceutical ingredients,” he said. “It’s generally well tolerated.”
- Clascoterone cream 1% (Winlevi). This first-in-class topical androgen receptor inhibitor is approved for the treatment of acne in patients 12 years and older. It competes with dihydrotestosterone and selectively targets androgen receptors in sebocytes and hair papilla cells. “It is safe for use in men, has been studied on the face and trunk, and has been shown to inhibit sebum production, reduce secretion of inflammatory cytokines, and inhibit inflammatory pathways,” Dr. Eichenfield said.
- Micro-encapsulated benzoyl peroxide 3% and tretinoin 0.1% cream (Twyneo). This is a once-daily fixed-dose combination of tretinoin and benzoyl peroxide indicated for the treatment of acne vulgaris in patients age 9 and older. According to a press release from Sol-Gel, the manufacturer, silica (silicon dioxide) core shell structures separate micro-encapsulate tretinoin crystals and benzoyl peroxide crystals, enabling inclusion of the two active ingredients in the cream.
Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Galderma, and Ortho Dermatologics.
SAN DIEGO – , according to Lawrence F. Eichenfield, MD.
The product, known as IDP-126 and being developed by Ortho Dermatologics, is a fixed dose triple combination of clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% being evaluated in patients nine years of age and older. According to a 2021 press release from the company, results from a second 12-week pivotal phase 3 trial showed a treatment success of 50.5% and 20.5% for IDP-126 and its vehicle, respectively, along with significant changes from baseline in inflammatory lesion count and non-inflammatory lesion count.
More recently, researchers led by Linda Stein Gold, MD, conducted a 12-week multicenter, randomized, double-blind study of IDP-126 in 741 children, adolescents, and adults with moderate to severe acne. They reported 52.5% of patients treated with IDP-126 gel achieved treatment success by week 12, with over 70% reduction in inflammatory and noninflammatory lesions.
“This will be interesting to follow as it moves along,” Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, said at the annual Masters of Aesthetics Symposium in a presentation on the newest topical acne treatments.
“If approved, we probably will be able to decrease our need for systemic therapies in some individuals,” he said. “It’s something that may become important in practices that mix and match between medical and procedural or surgical approaches to acne.”
Dr. Eichenfield highlighted other products for the topical treatment of acne:
- Trifarotene cream 0.005% (Aklief). In 2019, Food and Drug Administration approval made trifarotene cream the first new retinoid indicated for acne in several decades. It is indicated for the topical treatment of acne vulgaris in patients age 9 and older and has been studied in acne of the face, chest, and back.
- Tazarotene lotion 0.045% (Arazlo). The 0.1% formulation of tazarotene is commonly used for acne, but it can cause skin irritation, dryness, and erythema. The new 0.045% formulation was developed in a three-dimensional mesh matrix, with ingredients from an oil-in-water emulsion. “Many of the new acne products come with a background of vehicle delivery systems that minimize the concentration gradient, so it decreases irritation,” said Dr. Eichenfield, one of the authors of a 2021 review article on the management of acne vulgaris in JAMA. “This has very good efficacy without the traditional irritation of other tazarotene products,” Dr. Eichenfield said.
- Minocycline 4% topical foam (Amzeeq). The 2019 U.S. approval marked the first and so far only topical minocycline prescription treatment for acne. “Its hydrophobic composition allows for stable and efficient delivery of inherently unstable pharmaceutical ingredients,” he said. “It’s generally well tolerated.”
- Clascoterone cream 1% (Winlevi). This first-in-class topical androgen receptor inhibitor is approved for the treatment of acne in patients 12 years and older. It competes with dihydrotestosterone and selectively targets androgen receptors in sebocytes and hair papilla cells. “It is safe for use in men, has been studied on the face and trunk, and has been shown to inhibit sebum production, reduce secretion of inflammatory cytokines, and inhibit inflammatory pathways,” Dr. Eichenfield said.
- Micro-encapsulated benzoyl peroxide 3% and tretinoin 0.1% cream (Twyneo). This is a once-daily fixed-dose combination of tretinoin and benzoyl peroxide indicated for the treatment of acne vulgaris in patients age 9 and older. According to a press release from Sol-Gel, the manufacturer, silica (silicon dioxide) core shell structures separate micro-encapsulate tretinoin crystals and benzoyl peroxide crystals, enabling inclusion of the two active ingredients in the cream.
Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Galderma, and Ortho Dermatologics.
SAN DIEGO – , according to Lawrence F. Eichenfield, MD.
The product, known as IDP-126 and being developed by Ortho Dermatologics, is a fixed dose triple combination of clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% being evaluated in patients nine years of age and older. According to a 2021 press release from the company, results from a second 12-week pivotal phase 3 trial showed a treatment success of 50.5% and 20.5% for IDP-126 and its vehicle, respectively, along with significant changes from baseline in inflammatory lesion count and non-inflammatory lesion count.
More recently, researchers led by Linda Stein Gold, MD, conducted a 12-week multicenter, randomized, double-blind study of IDP-126 in 741 children, adolescents, and adults with moderate to severe acne. They reported 52.5% of patients treated with IDP-126 gel achieved treatment success by week 12, with over 70% reduction in inflammatory and noninflammatory lesions.
“This will be interesting to follow as it moves along,” Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, said at the annual Masters of Aesthetics Symposium in a presentation on the newest topical acne treatments.
“If approved, we probably will be able to decrease our need for systemic therapies in some individuals,” he said. “It’s something that may become important in practices that mix and match between medical and procedural or surgical approaches to acne.”
Dr. Eichenfield highlighted other products for the topical treatment of acne:
- Trifarotene cream 0.005% (Aklief). In 2019, Food and Drug Administration approval made trifarotene cream the first new retinoid indicated for acne in several decades. It is indicated for the topical treatment of acne vulgaris in patients age 9 and older and has been studied in acne of the face, chest, and back.
- Tazarotene lotion 0.045% (Arazlo). The 0.1% formulation of tazarotene is commonly used for acne, but it can cause skin irritation, dryness, and erythema. The new 0.045% formulation was developed in a three-dimensional mesh matrix, with ingredients from an oil-in-water emulsion. “Many of the new acne products come with a background of vehicle delivery systems that minimize the concentration gradient, so it decreases irritation,” said Dr. Eichenfield, one of the authors of a 2021 review article on the management of acne vulgaris in JAMA. “This has very good efficacy without the traditional irritation of other tazarotene products,” Dr. Eichenfield said.
- Minocycline 4% topical foam (Amzeeq). The 2019 U.S. approval marked the first and so far only topical minocycline prescription treatment for acne. “Its hydrophobic composition allows for stable and efficient delivery of inherently unstable pharmaceutical ingredients,” he said. “It’s generally well tolerated.”
- Clascoterone cream 1% (Winlevi). This first-in-class topical androgen receptor inhibitor is approved for the treatment of acne in patients 12 years and older. It competes with dihydrotestosterone and selectively targets androgen receptors in sebocytes and hair papilla cells. “It is safe for use in men, has been studied on the face and trunk, and has been shown to inhibit sebum production, reduce secretion of inflammatory cytokines, and inhibit inflammatory pathways,” Dr. Eichenfield said.
- Micro-encapsulated benzoyl peroxide 3% and tretinoin 0.1% cream (Twyneo). This is a once-daily fixed-dose combination of tretinoin and benzoyl peroxide indicated for the treatment of acne vulgaris in patients age 9 and older. According to a press release from Sol-Gel, the manufacturer, silica (silicon dioxide) core shell structures separate micro-encapsulate tretinoin crystals and benzoyl peroxide crystals, enabling inclusion of the two active ingredients in the cream.
Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Galderma, and Ortho Dermatologics.
AT MOAS 2022
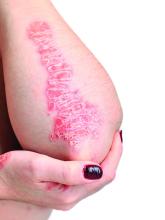
New treatments aim to tame vitiligo
LAS VEGAS – in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
Vitiligo, an autoimmune condition that results in patches of skin depigmentation, occurs in 0.5% to 2% of the population. The average age of onset is 20 years, with 25% of cases occurring before age 10, and 70%-80% of cases by age 30 years, which means a long-term effect on quality of life, especially for younger patients, said Dr. Rosmarin, vice chair of education and research and director of the clinical trials unit at Tufts University, Boston.
Studies have shown that 95% of 15- to 17-year-olds with vitiligo are bothered by it, as are approximately 50% of children aged 6-14 years, he said. Although patients with more extensive lesions on the face, arms, legs, and hands report worse quality of life, they report that uncontrolled progression of vitiligo is more concerning than the presence of lesions in exposed areas, he noted.
The current strategy for getting vitiligo under control is a two-step process, said Dr. Rosmarin. First, improve the skin environment by suppressing the overactive immune system, then encourage repigmentation and “nudge the melanocytes to return,” he said.
Topical ruxolitinib, a Janus kinase (JAK) inhibitor, is the latest tool for dermatologists to help give the melanocytes that nudge. In July 2022, the Food and Drug Administration approved ruxolitinib cream for treating nonsegmental vitiligo in patients 12 years of age and older – the first treatment approved to repigment patients with vitiligo.
Vitiligo is driven in part by interferon (IFN)-gamma signaling through JAK 1 and 2, and ruxolitinib acts as an inhibitor, Dr. Rosmarin said.
In the TRuE-V1 and TRuE-V2 studies presented at the 2022 European Academy of Dermatology and Venereology meeting in Milan, adolescents and adults with vitiligo who were randomized to 1.5% ruxolitinib cream twice daily showed significant improvement over those randomized to the vehicle by 24 weeks, at which time all patients could continue with ruxolitinib through 52 weeks, he said.
Dr. Rosmarin presented 52-week data from the TRuE-V1 and TRuE-V2 studies at the 2022 American Academy of Dermatology meeting in Boston. He was the lead author of the studies that were subsequently published in the New England Journal of Medicine.
In the two studies, 52.6% and 48% of the patients in the ruxolitinib groups achieved the primary outcome of at least 75% improvement on the Facial Vitiligo Area Scoring Index (F-VASI75) by 52 weeks, compared with 26.8% and 29.6% of patients on the vehicle, respectively.
In addition, at 52 weeks, 53.2% and 49.2% of patients treated with ruxolitinib in the two studies achieved 50% improvement on the Total Vitiligo Area Scoring Index (T-VASI50), a clinician assessment of affected body surface area and level of depigmentation, compared with 31.7% and 22.2% of those on vehicle, respectively.
Patient satisfaction was high with ruxolitinib, Dr. Rosmarin said. In the TRuE-V1 and TRuE-V2 studies, 39.9% and 32.8% of patients, respectively, achieved a successful treatment response based on the patient-reported Vitiligo Noticeability Scale (VNS) by week 52, versus 19.5% and 13.6% of those on vehicle.
Ruxolitinib cream was well tolerated, with “no clinically significant application site reactions or serious treatment-related adverse events,” he noted. The most common treatment-related adverse events across the TRuE-V1 and TRuE-V2 studies were acne at the application site (affecting about 6% of patients) and pruritus at the application site about (affecting 5%), said Dr. Rosmarin.
JAK inhibitors, including ruxolitinib, baricitinib, and tofacitinib, have shown effectiveness for vitiligo, which supports the potential role of the IFN-gamma-chemokine signaling axis in the pathogenesis of the disease, said Dr. Rosmarin. However, more studies are required to determine the ideal dosage of JAK inhibitors for the treatment of vitiligo, and to identify other inflammatory pathways that may be implicated in the pathogenesis of this condition.
Ruxolitinib’s success has been consistent across subgroups of age, gender, race, geographic region, and Fitzpatrick skin phototype. Notably, ruxolitinib was effective among the adolescent population, with approximately 60% achieving T-VASI50 and success based on VNS in TRuE-V1 and TRuE-V2.
An oral version of ruxolitinib is in clinical trials, which “makes a lot of sense,” Dr. Rosmarin said. “Patients don’t always have localized disease,” and such patients may benefit from an oral therapy. Topicals may have the advantage in terms of safety, but questions of maintenance remain, he said. Oral treatments may be useful for patients with large body surface areas affected, and those with unstable or progressive disease, he added.
Areas for additional research include combination therapy with ruxolitinib and phototherapy, and an anti-IL 15 therapy in the pipeline has the potential to drive vitiligo into remission, Dr. Rosmarin said. In a study known as REVEAL that is still recruiting patients, researchers will test the efficacy of an IL-15 inhibitor known as AMG 714 to induce facial repigmentation in adults with vitiligo.
Dr. Rosmarin disclosed ties with AbbVie, Abcuro, AltruBio, Amgen, Arena Pharmaceuticals, Boehringer Ingelheim, Bristol Myers Squibb Company, Celgene, Concert Pharmaceuticals, CSL Behring, Dermavant, Dermira, Eli Lilly, Galderma, Incyte, Janssen, Kyowa Kirin, Merck, Novartis, Pfizer, Regeneron, Revolo, Sanofi, Sun, UCB, and Viela Bio.
MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
Vitiligo, an autoimmune condition that results in patches of skin depigmentation, occurs in 0.5% to 2% of the population. The average age of onset is 20 years, with 25% of cases occurring before age 10, and 70%-80% of cases by age 30 years, which means a long-term effect on quality of life, especially for younger patients, said Dr. Rosmarin, vice chair of education and research and director of the clinical trials unit at Tufts University, Boston.
Studies have shown that 95% of 15- to 17-year-olds with vitiligo are bothered by it, as are approximately 50% of children aged 6-14 years, he said. Although patients with more extensive lesions on the face, arms, legs, and hands report worse quality of life, they report that uncontrolled progression of vitiligo is more concerning than the presence of lesions in exposed areas, he noted.
The current strategy for getting vitiligo under control is a two-step process, said Dr. Rosmarin. First, improve the skin environment by suppressing the overactive immune system, then encourage repigmentation and “nudge the melanocytes to return,” he said.
Topical ruxolitinib, a Janus kinase (JAK) inhibitor, is the latest tool for dermatologists to help give the melanocytes that nudge. In July 2022, the Food and Drug Administration approved ruxolitinib cream for treating nonsegmental vitiligo in patients 12 years of age and older – the first treatment approved to repigment patients with vitiligo.
Vitiligo is driven in part by interferon (IFN)-gamma signaling through JAK 1 and 2, and ruxolitinib acts as an inhibitor, Dr. Rosmarin said.
In the TRuE-V1 and TRuE-V2 studies presented at the 2022 European Academy of Dermatology and Venereology meeting in Milan, adolescents and adults with vitiligo who were randomized to 1.5% ruxolitinib cream twice daily showed significant improvement over those randomized to the vehicle by 24 weeks, at which time all patients could continue with ruxolitinib through 52 weeks, he said.
Dr. Rosmarin presented 52-week data from the TRuE-V1 and TRuE-V2 studies at the 2022 American Academy of Dermatology meeting in Boston. He was the lead author of the studies that were subsequently published in the New England Journal of Medicine.
In the two studies, 52.6% and 48% of the patients in the ruxolitinib groups achieved the primary outcome of at least 75% improvement on the Facial Vitiligo Area Scoring Index (F-VASI75) by 52 weeks, compared with 26.8% and 29.6% of patients on the vehicle, respectively.
In addition, at 52 weeks, 53.2% and 49.2% of patients treated with ruxolitinib in the two studies achieved 50% improvement on the Total Vitiligo Area Scoring Index (T-VASI50), a clinician assessment of affected body surface area and level of depigmentation, compared with 31.7% and 22.2% of those on vehicle, respectively.
Patient satisfaction was high with ruxolitinib, Dr. Rosmarin said. In the TRuE-V1 and TRuE-V2 studies, 39.9% and 32.8% of patients, respectively, achieved a successful treatment response based on the patient-reported Vitiligo Noticeability Scale (VNS) by week 52, versus 19.5% and 13.6% of those on vehicle.
Ruxolitinib cream was well tolerated, with “no clinically significant application site reactions or serious treatment-related adverse events,” he noted. The most common treatment-related adverse events across the TRuE-V1 and TRuE-V2 studies were acne at the application site (affecting about 6% of patients) and pruritus at the application site about (affecting 5%), said Dr. Rosmarin.
JAK inhibitors, including ruxolitinib, baricitinib, and tofacitinib, have shown effectiveness for vitiligo, which supports the potential role of the IFN-gamma-chemokine signaling axis in the pathogenesis of the disease, said Dr. Rosmarin. However, more studies are required to determine the ideal dosage of JAK inhibitors for the treatment of vitiligo, and to identify other inflammatory pathways that may be implicated in the pathogenesis of this condition.
Ruxolitinib’s success has been consistent across subgroups of age, gender, race, geographic region, and Fitzpatrick skin phototype. Notably, ruxolitinib was effective among the adolescent population, with approximately 60% achieving T-VASI50 and success based on VNS in TRuE-V1 and TRuE-V2.
An oral version of ruxolitinib is in clinical trials, which “makes a lot of sense,” Dr. Rosmarin said. “Patients don’t always have localized disease,” and such patients may benefit from an oral therapy. Topicals may have the advantage in terms of safety, but questions of maintenance remain, he said. Oral treatments may be useful for patients with large body surface areas affected, and those with unstable or progressive disease, he added.
Areas for additional research include combination therapy with ruxolitinib and phototherapy, and an anti-IL 15 therapy in the pipeline has the potential to drive vitiligo into remission, Dr. Rosmarin said. In a study known as REVEAL that is still recruiting patients, researchers will test the efficacy of an IL-15 inhibitor known as AMG 714 to induce facial repigmentation in adults with vitiligo.
Dr. Rosmarin disclosed ties with AbbVie, Abcuro, AltruBio, Amgen, Arena Pharmaceuticals, Boehringer Ingelheim, Bristol Myers Squibb Company, Celgene, Concert Pharmaceuticals, CSL Behring, Dermavant, Dermira, Eli Lilly, Galderma, Incyte, Janssen, Kyowa Kirin, Merck, Novartis, Pfizer, Regeneron, Revolo, Sanofi, Sun, UCB, and Viela Bio.
MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
Vitiligo, an autoimmune condition that results in patches of skin depigmentation, occurs in 0.5% to 2% of the population. The average age of onset is 20 years, with 25% of cases occurring before age 10, and 70%-80% of cases by age 30 years, which means a long-term effect on quality of life, especially for younger patients, said Dr. Rosmarin, vice chair of education and research and director of the clinical trials unit at Tufts University, Boston.
Studies have shown that 95% of 15- to 17-year-olds with vitiligo are bothered by it, as are approximately 50% of children aged 6-14 years, he said. Although patients with more extensive lesions on the face, arms, legs, and hands report worse quality of life, they report that uncontrolled progression of vitiligo is more concerning than the presence of lesions in exposed areas, he noted.
The current strategy for getting vitiligo under control is a two-step process, said Dr. Rosmarin. First, improve the skin environment by suppressing the overactive immune system, then encourage repigmentation and “nudge the melanocytes to return,” he said.
Topical ruxolitinib, a Janus kinase (JAK) inhibitor, is the latest tool for dermatologists to help give the melanocytes that nudge. In July 2022, the Food and Drug Administration approved ruxolitinib cream for treating nonsegmental vitiligo in patients 12 years of age and older – the first treatment approved to repigment patients with vitiligo.
Vitiligo is driven in part by interferon (IFN)-gamma signaling through JAK 1 and 2, and ruxolitinib acts as an inhibitor, Dr. Rosmarin said.
In the TRuE-V1 and TRuE-V2 studies presented at the 2022 European Academy of Dermatology and Venereology meeting in Milan, adolescents and adults with vitiligo who were randomized to 1.5% ruxolitinib cream twice daily showed significant improvement over those randomized to the vehicle by 24 weeks, at which time all patients could continue with ruxolitinib through 52 weeks, he said.
Dr. Rosmarin presented 52-week data from the TRuE-V1 and TRuE-V2 studies at the 2022 American Academy of Dermatology meeting in Boston. He was the lead author of the studies that were subsequently published in the New England Journal of Medicine.
In the two studies, 52.6% and 48% of the patients in the ruxolitinib groups achieved the primary outcome of at least 75% improvement on the Facial Vitiligo Area Scoring Index (F-VASI75) by 52 weeks, compared with 26.8% and 29.6% of patients on the vehicle, respectively.
In addition, at 52 weeks, 53.2% and 49.2% of patients treated with ruxolitinib in the two studies achieved 50% improvement on the Total Vitiligo Area Scoring Index (T-VASI50), a clinician assessment of affected body surface area and level of depigmentation, compared with 31.7% and 22.2% of those on vehicle, respectively.
Patient satisfaction was high with ruxolitinib, Dr. Rosmarin said. In the TRuE-V1 and TRuE-V2 studies, 39.9% and 32.8% of patients, respectively, achieved a successful treatment response based on the patient-reported Vitiligo Noticeability Scale (VNS) by week 52, versus 19.5% and 13.6% of those on vehicle.
Ruxolitinib cream was well tolerated, with “no clinically significant application site reactions or serious treatment-related adverse events,” he noted. The most common treatment-related adverse events across the TRuE-V1 and TRuE-V2 studies were acne at the application site (affecting about 6% of patients) and pruritus at the application site about (affecting 5%), said Dr. Rosmarin.
JAK inhibitors, including ruxolitinib, baricitinib, and tofacitinib, have shown effectiveness for vitiligo, which supports the potential role of the IFN-gamma-chemokine signaling axis in the pathogenesis of the disease, said Dr. Rosmarin. However, more studies are required to determine the ideal dosage of JAK inhibitors for the treatment of vitiligo, and to identify other inflammatory pathways that may be implicated in the pathogenesis of this condition.
Ruxolitinib’s success has been consistent across subgroups of age, gender, race, geographic region, and Fitzpatrick skin phototype. Notably, ruxolitinib was effective among the adolescent population, with approximately 60% achieving T-VASI50 and success based on VNS in TRuE-V1 and TRuE-V2.
An oral version of ruxolitinib is in clinical trials, which “makes a lot of sense,” Dr. Rosmarin said. “Patients don’t always have localized disease,” and such patients may benefit from an oral therapy. Topicals may have the advantage in terms of safety, but questions of maintenance remain, he said. Oral treatments may be useful for patients with large body surface areas affected, and those with unstable or progressive disease, he added.
Areas for additional research include combination therapy with ruxolitinib and phototherapy, and an anti-IL 15 therapy in the pipeline has the potential to drive vitiligo into remission, Dr. Rosmarin said. In a study known as REVEAL that is still recruiting patients, researchers will test the efficacy of an IL-15 inhibitor known as AMG 714 to induce facial repigmentation in adults with vitiligo.
Dr. Rosmarin disclosed ties with AbbVie, Abcuro, AltruBio, Amgen, Arena Pharmaceuticals, Boehringer Ingelheim, Bristol Myers Squibb Company, Celgene, Concert Pharmaceuticals, CSL Behring, Dermavant, Dermira, Eli Lilly, Galderma, Incyte, Janssen, Kyowa Kirin, Merck, Novartis, Pfizer, Regeneron, Revolo, Sanofi, Sun, UCB, and Viela Bio.
MedscapeLive and this news organization are owned by the same parent company.
AT INNOVATIONS IN DERMATOLOGY
Oral minoxidil improves anticancer treatment–induced alopecia in women with breast cancer
Topical minoxidil is widely used to treat hair loss, but new findings suggest that
In a retrospective cohort study of women with breast cancer and anticancer therapy–induced alopecia, researchers found that combining low-dose oral minoxidil (LDOM) and topical minoxidil achieved better results than topical minoxidil alone and that the treatment was well tolerated. A total of 5 of the 37 patients (13.5%) in the combination therapy group achieved a complete response, defined as an improvement of alopecia severity from grade 2 to grade 1, compared with none of the 19 patients in the topical therapy–only group.
In contrast, none of the patients in the combination group experienced worsening of alopecia, compared with two (10.5%) in the topical monotherapy group.
The study was published online in the Journal of the American Academy of Dermatology. Topical minoxidil is approved by the Food and Drug Administration to treat androgenetic alopecia. Oral minoxidil is not approved for treating hair loss but has been receiving increased attention as an adjunctive therapy for hair loss, particularly for women. Oral minoxidil is approved for treating hypertension but at much higher doses.
An increasing number of studies have been conducted on the use of oral minoxidil for the treatment of female pattern hair loss, dating back to a pilot study in 2017, with promising results. The findings suggest that LDOM might be more effective than topical therapy, well tolerated, and more convenient for individuals to take.
Hypothesis generating
In a comment, Kai Johnson, MD, a medical oncologist who specializes in treating patients with breast cancer at the Ohio State University Comprehensive Cancer Center – Arthur G. James Cancer Hospital and Richard J. Solove Research Institute, Columbus, noted that the study, like most small-scale retrospective studies, is hypothesis generating. However, “I’d be hesitant to broadly recommend this practice of dual therapy – oral and topical minoxidil together – until we see a placebo-controlled prospective study performed demonstrating clinically meaningful benefits for patients.”
Another factor is the study endpoints. “While there was a statistically significant benefit documented with dual therapy in this study, it’s important to have study endpoints that are more patient oriented,” Dr. Johnson said. The most important endpoint for patients would be improvements “in the actual alopecia grade, which did occur in 5 of the 37 of dual-therapy patients, versus 0 topical minoxidil patients.”
George Cotsarelis, MD, chair of the department of dermatology and professor of dermatology at the University of Pennsylvania, Philadelphia, also weighed in. He questioned whether adding the topical therapy to oral minoxidil actually improved the results. “What was missing was a study arm that used the oral alone,” he said in an interview. “So we don’t know how effective the oral therapy would be by itself and if combining it with the topical is really adding anything.”
Oral minoxidil as a treatment for hair loss is gaining traction, and it’s clear that it is effective. However, the risk of side effects is higher, he said. “The risk isn’t that high with the low dose, but it can grow hair on places other than the scalp, and that can be disconcerting.” In this study, two women who took the oral drug reported edema, and one reported headache and dizziness. Hypertrichosis was reported by five patients who received the combination.
Study details
In the study, Jeewoo Kang, MD, and colleagues from the Seoul National University evaluated the efficacy of LDOM in 100 patients with breast cancer who had been diagnosed with persistent chemotherapy-induced alopecia (pCIA) and endocrine therapy–induced alopecia (EIA) at a dermatology clinic.
They conducted an analysis of medical records, standardized clinical photographs, and trichoscopic images to evaluate the alopecia pattern, severity, treatment response, and posttreatment changes in vertex hair density and thickness.
Compared with those with EIA alone, patients with pCIA were significantly more likely to have diffuse alopecia (P < .001), and they were more likely to have more severe alopecia, although this difference was not significant (P = .058). Outcomes were evaluated for 56 patients who were treated with minoxidil (19 with topical minoxidil alone and 37 with both LDOM and topical minoxidil) and for whom clinical and trichoscopic photos were available at baseline and at the last follow-up (all patients were scheduled for follow-up at 3-month intervals).
The results showed that those treated with 1.25-5.0 mg/d of oral minoxidil and 5% topical minoxidil solution once a day had better responses (P = .002) and a higher percentage increase in hair density from baseline (P = .003), compared with those who received topical minoxidil monotherapy.
However, changes in hair thickness after treatment were not significantly different between the two groups (P = .540).
In addition to the five (13.5%) cases of hypertrichosis, two cases of edema (5.4%), and one case of headache/dizziness (2.7%) among those who received the combination, there was also one report of palpitations (2.7%). Palpitations were reported in one patient (5%) who received topical monotherapy, the only adverse event reported in this group.
Dr. Johnson noted that, at his institution, a dermatologist is conducting a clinical trial with oncology patients post chemotherapy and endocrine therapy. “She is looking at a similar question, although she is comparing oral minoxidil to topical minoxidil directly rather than in combination.” There is also an active clinical trial at Northwestern University, Chicago, of LDOM alone for patients with chemotherapy-induced alopecia.
“So there is a lot of momentum surrounding this concept, and I feel we will continue to see it come up as a possible treatment option, but more data are needed at this time before it can become standard of care,” Dr. Johnson added.
No funding for the study was reported. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Topical minoxidil is widely used to treat hair loss, but new findings suggest that
In a retrospective cohort study of women with breast cancer and anticancer therapy–induced alopecia, researchers found that combining low-dose oral minoxidil (LDOM) and topical minoxidil achieved better results than topical minoxidil alone and that the treatment was well tolerated. A total of 5 of the 37 patients (13.5%) in the combination therapy group achieved a complete response, defined as an improvement of alopecia severity from grade 2 to grade 1, compared with none of the 19 patients in the topical therapy–only group.
In contrast, none of the patients in the combination group experienced worsening of alopecia, compared with two (10.5%) in the topical monotherapy group.
The study was published online in the Journal of the American Academy of Dermatology. Topical minoxidil is approved by the Food and Drug Administration to treat androgenetic alopecia. Oral minoxidil is not approved for treating hair loss but has been receiving increased attention as an adjunctive therapy for hair loss, particularly for women. Oral minoxidil is approved for treating hypertension but at much higher doses.
An increasing number of studies have been conducted on the use of oral minoxidil for the treatment of female pattern hair loss, dating back to a pilot study in 2017, with promising results. The findings suggest that LDOM might be more effective than topical therapy, well tolerated, and more convenient for individuals to take.
Hypothesis generating
In a comment, Kai Johnson, MD, a medical oncologist who specializes in treating patients with breast cancer at the Ohio State University Comprehensive Cancer Center – Arthur G. James Cancer Hospital and Richard J. Solove Research Institute, Columbus, noted that the study, like most small-scale retrospective studies, is hypothesis generating. However, “I’d be hesitant to broadly recommend this practice of dual therapy – oral and topical minoxidil together – until we see a placebo-controlled prospective study performed demonstrating clinically meaningful benefits for patients.”
Another factor is the study endpoints. “While there was a statistically significant benefit documented with dual therapy in this study, it’s important to have study endpoints that are more patient oriented,” Dr. Johnson said. The most important endpoint for patients would be improvements “in the actual alopecia grade, which did occur in 5 of the 37 of dual-therapy patients, versus 0 topical minoxidil patients.”
George Cotsarelis, MD, chair of the department of dermatology and professor of dermatology at the University of Pennsylvania, Philadelphia, also weighed in. He questioned whether adding the topical therapy to oral minoxidil actually improved the results. “What was missing was a study arm that used the oral alone,” he said in an interview. “So we don’t know how effective the oral therapy would be by itself and if combining it with the topical is really adding anything.”
Oral minoxidil as a treatment for hair loss is gaining traction, and it’s clear that it is effective. However, the risk of side effects is higher, he said. “The risk isn’t that high with the low dose, but it can grow hair on places other than the scalp, and that can be disconcerting.” In this study, two women who took the oral drug reported edema, and one reported headache and dizziness. Hypertrichosis was reported by five patients who received the combination.
Study details
In the study, Jeewoo Kang, MD, and colleagues from the Seoul National University evaluated the efficacy of LDOM in 100 patients with breast cancer who had been diagnosed with persistent chemotherapy-induced alopecia (pCIA) and endocrine therapy–induced alopecia (EIA) at a dermatology clinic.
They conducted an analysis of medical records, standardized clinical photographs, and trichoscopic images to evaluate the alopecia pattern, severity, treatment response, and posttreatment changes in vertex hair density and thickness.
Compared with those with EIA alone, patients with pCIA were significantly more likely to have diffuse alopecia (P < .001), and they were more likely to have more severe alopecia, although this difference was not significant (P = .058). Outcomes were evaluated for 56 patients who were treated with minoxidil (19 with topical minoxidil alone and 37 with both LDOM and topical minoxidil) and for whom clinical and trichoscopic photos were available at baseline and at the last follow-up (all patients were scheduled for follow-up at 3-month intervals).
The results showed that those treated with 1.25-5.0 mg/d of oral minoxidil and 5% topical minoxidil solution once a day had better responses (P = .002) and a higher percentage increase in hair density from baseline (P = .003), compared with those who received topical minoxidil monotherapy.
However, changes in hair thickness after treatment were not significantly different between the two groups (P = .540).
In addition to the five (13.5%) cases of hypertrichosis, two cases of edema (5.4%), and one case of headache/dizziness (2.7%) among those who received the combination, there was also one report of palpitations (2.7%). Palpitations were reported in one patient (5%) who received topical monotherapy, the only adverse event reported in this group.
Dr. Johnson noted that, at his institution, a dermatologist is conducting a clinical trial with oncology patients post chemotherapy and endocrine therapy. “She is looking at a similar question, although she is comparing oral minoxidil to topical minoxidil directly rather than in combination.” There is also an active clinical trial at Northwestern University, Chicago, of LDOM alone for patients with chemotherapy-induced alopecia.
“So there is a lot of momentum surrounding this concept, and I feel we will continue to see it come up as a possible treatment option, but more data are needed at this time before it can become standard of care,” Dr. Johnson added.
No funding for the study was reported. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Topical minoxidil is widely used to treat hair loss, but new findings suggest that
In a retrospective cohort study of women with breast cancer and anticancer therapy–induced alopecia, researchers found that combining low-dose oral minoxidil (LDOM) and topical minoxidil achieved better results than topical minoxidil alone and that the treatment was well tolerated. A total of 5 of the 37 patients (13.5%) in the combination therapy group achieved a complete response, defined as an improvement of alopecia severity from grade 2 to grade 1, compared with none of the 19 patients in the topical therapy–only group.
In contrast, none of the patients in the combination group experienced worsening of alopecia, compared with two (10.5%) in the topical monotherapy group.
The study was published online in the Journal of the American Academy of Dermatology. Topical minoxidil is approved by the Food and Drug Administration to treat androgenetic alopecia. Oral minoxidil is not approved for treating hair loss but has been receiving increased attention as an adjunctive therapy for hair loss, particularly for women. Oral minoxidil is approved for treating hypertension but at much higher doses.
An increasing number of studies have been conducted on the use of oral minoxidil for the treatment of female pattern hair loss, dating back to a pilot study in 2017, with promising results. The findings suggest that LDOM might be more effective than topical therapy, well tolerated, and more convenient for individuals to take.
Hypothesis generating
In a comment, Kai Johnson, MD, a medical oncologist who specializes in treating patients with breast cancer at the Ohio State University Comprehensive Cancer Center – Arthur G. James Cancer Hospital and Richard J. Solove Research Institute, Columbus, noted that the study, like most small-scale retrospective studies, is hypothesis generating. However, “I’d be hesitant to broadly recommend this practice of dual therapy – oral and topical minoxidil together – until we see a placebo-controlled prospective study performed demonstrating clinically meaningful benefits for patients.”
Another factor is the study endpoints. “While there was a statistically significant benefit documented with dual therapy in this study, it’s important to have study endpoints that are more patient oriented,” Dr. Johnson said. The most important endpoint for patients would be improvements “in the actual alopecia grade, which did occur in 5 of the 37 of dual-therapy patients, versus 0 topical minoxidil patients.”
George Cotsarelis, MD, chair of the department of dermatology and professor of dermatology at the University of Pennsylvania, Philadelphia, also weighed in. He questioned whether adding the topical therapy to oral minoxidil actually improved the results. “What was missing was a study arm that used the oral alone,” he said in an interview. “So we don’t know how effective the oral therapy would be by itself and if combining it with the topical is really adding anything.”
Oral minoxidil as a treatment for hair loss is gaining traction, and it’s clear that it is effective. However, the risk of side effects is higher, he said. “The risk isn’t that high with the low dose, but it can grow hair on places other than the scalp, and that can be disconcerting.” In this study, two women who took the oral drug reported edema, and one reported headache and dizziness. Hypertrichosis was reported by five patients who received the combination.
Study details
In the study, Jeewoo Kang, MD, and colleagues from the Seoul National University evaluated the efficacy of LDOM in 100 patients with breast cancer who had been diagnosed with persistent chemotherapy-induced alopecia (pCIA) and endocrine therapy–induced alopecia (EIA) at a dermatology clinic.
They conducted an analysis of medical records, standardized clinical photographs, and trichoscopic images to evaluate the alopecia pattern, severity, treatment response, and posttreatment changes in vertex hair density and thickness.
Compared with those with EIA alone, patients with pCIA were significantly more likely to have diffuse alopecia (P < .001), and they were more likely to have more severe alopecia, although this difference was not significant (P = .058). Outcomes were evaluated for 56 patients who were treated with minoxidil (19 with topical minoxidil alone and 37 with both LDOM and topical minoxidil) and for whom clinical and trichoscopic photos were available at baseline and at the last follow-up (all patients were scheduled for follow-up at 3-month intervals).
The results showed that those treated with 1.25-5.0 mg/d of oral minoxidil and 5% topical minoxidil solution once a day had better responses (P = .002) and a higher percentage increase in hair density from baseline (P = .003), compared with those who received topical minoxidil monotherapy.
However, changes in hair thickness after treatment were not significantly different between the two groups (P = .540).
In addition to the five (13.5%) cases of hypertrichosis, two cases of edema (5.4%), and one case of headache/dizziness (2.7%) among those who received the combination, there was also one report of palpitations (2.7%). Palpitations were reported in one patient (5%) who received topical monotherapy, the only adverse event reported in this group.
Dr. Johnson noted that, at his institution, a dermatologist is conducting a clinical trial with oncology patients post chemotherapy and endocrine therapy. “She is looking at a similar question, although she is comparing oral minoxidil to topical minoxidil directly rather than in combination.” There is also an active clinical trial at Northwestern University, Chicago, of LDOM alone for patients with chemotherapy-induced alopecia.
“So there is a lot of momentum surrounding this concept, and I feel we will continue to see it come up as a possible treatment option, but more data are needed at this time before it can become standard of care,” Dr. Johnson added.
No funding for the study was reported. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Adverse events linked to better survival with ICIs in melanoma
Among Survival is further improved if the immunotherapy is continued after the adverse event develops, a new study confirms.
“In the largest clinical cohort to date, our data support a positive association with overall survival for patients who develop clinically significant immune-related adverse events while receiving combination immune checkpoint blockade, in keeping with other reported series,” the authors wrote.
The study was published online in JAMA Network Open.
Immune-related adverse events are common with these drugs. Severe events of grade 3 or higher occur in 59% of trial patients who receive combination ICI therapy.
The adverse events have increasingly been positively associated with survival. However, the effects for patients with metastatic melanoma, in particular, are less clear. There is little research on the effects in relation to combination therapy with ipilimumab and nivolumab, which is the standard of care for many patients with metastatic melanoma.
To investigate, Alexander S. Watson, MD, and colleagues evaluated data on 492 patients with metastatic melanoma who had been treated with one or more doses of an anti–programmed death 1 agent as single or combination immune checkpoint blockade in the multicenter Alberta Immunotherapy Database from August 2013 to May 2020.
Of these 492 patients, 198 patients (40%) developed immune-related adverse events. The mean age of the patients who developed adverse events was 61.8 years; of those who did not develop adverse events, the mean age was 65.5 years. Men made up 69.2% and 62.2%, respectively.
A total of 288 patients received pembrolizumab as their first ICI therapy, 80 received nivolumab, and 124 received combination blockade with ipilimumab-nivolumab.
Overall, with a median follow-up of 36.6 months, among patients who experienced clinically significant immune-related adverse events, defined as requiring systemic corticosteroids and/or a treatment delay, median overall survival was significantly improved, at 56.3 months, compared with 18.5 months among those who did not experience immune-related adverse events (P < .001).
In addition, among those who received combination ICI treatment, the median overall survival was 56.2 months for those who experienced adverse events versus 19.0 months for those who did not (P < .001).
There were no significant differences in overall survival between those who were and those who were not hospitalized for their immune-related adverse events (P = .53).
For patients who resumed their ICI therapy following the adverse events, overall survival was longer, compared with those who did not resume the therapy (median, 56.3 months vs. 31.5 months; P = .009).
The improvements in overall survival seen with immune-related adverse events remained consistent after adjustment in a multivariable analysis (hazard ratio for death, 0.382; P < .001).
There were no significant differences in the median number of cycles of ICIs between those with and those without the adverse events.
The risk of recurrence of immune-related adverse events following the reintroduction of therapy after initial events was a concern, so the improved overall survival among those patients is encouraging, although further investigation is needed, commented lead author Dr. Watson, from the department of oncology, University of Calgary (Alta.).
“It may be, for certain patients with immune-related adverse events, that continued immune-priming is safe and optimizes anticancer response,” he told this news organization. “However, in a retrospective analysis such as ours, selection bias can have an impact.”
“Confirming this finding and better identifying patients who may benefit from resumption will be an area for future investigation,” he said.
Patients who developed immune-related adverse events were more likely to be younger than 50 years (21.8% vs. 13.9%), have normal albumin levels (86.4% vs. 74.8%), and have a more robust Eastern Cooperative Oncology Group status, which is consistent with other studies that have shown survival benefits among those who experience adverse events.
“We, and others, speculate this could be due to such groups having immune systems more ready to respond strongly to immunotherapy,” Dr. Watson explained.
After controlling for age and performance status in the multivariable analysis, however, “immune-related adverse events remained strongly associated with survival, potentially [indicating] that robust responses to immunotherapy lead to both cancer control and immune-related adverse events,” he said.
Overall, “we feel these findings will help clinicians in discussions with patients and in clinical decision-making after adverse events develop,” Dr. Watson said.
Dr. Watson has received personal fees from Apobiologix Canada.
A version of this article first appeared on Medscape.com.
Among Survival is further improved if the immunotherapy is continued after the adverse event develops, a new study confirms.
“In the largest clinical cohort to date, our data support a positive association with overall survival for patients who develop clinically significant immune-related adverse events while receiving combination immune checkpoint blockade, in keeping with other reported series,” the authors wrote.
The study was published online in JAMA Network Open.
Immune-related adverse events are common with these drugs. Severe events of grade 3 or higher occur in 59% of trial patients who receive combination ICI therapy.
The adverse events have increasingly been positively associated with survival. However, the effects for patients with metastatic melanoma, in particular, are less clear. There is little research on the effects in relation to combination therapy with ipilimumab and nivolumab, which is the standard of care for many patients with metastatic melanoma.
To investigate, Alexander S. Watson, MD, and colleagues evaluated data on 492 patients with metastatic melanoma who had been treated with one or more doses of an anti–programmed death 1 agent as single or combination immune checkpoint blockade in the multicenter Alberta Immunotherapy Database from August 2013 to May 2020.
Of these 492 patients, 198 patients (40%) developed immune-related adverse events. The mean age of the patients who developed adverse events was 61.8 years; of those who did not develop adverse events, the mean age was 65.5 years. Men made up 69.2% and 62.2%, respectively.
A total of 288 patients received pembrolizumab as their first ICI therapy, 80 received nivolumab, and 124 received combination blockade with ipilimumab-nivolumab.
Overall, with a median follow-up of 36.6 months, among patients who experienced clinically significant immune-related adverse events, defined as requiring systemic corticosteroids and/or a treatment delay, median overall survival was significantly improved, at 56.3 months, compared with 18.5 months among those who did not experience immune-related adverse events (P < .001).
In addition, among those who received combination ICI treatment, the median overall survival was 56.2 months for those who experienced adverse events versus 19.0 months for those who did not (P < .001).
There were no significant differences in overall survival between those who were and those who were not hospitalized for their immune-related adverse events (P = .53).
For patients who resumed their ICI therapy following the adverse events, overall survival was longer, compared with those who did not resume the therapy (median, 56.3 months vs. 31.5 months; P = .009).
The improvements in overall survival seen with immune-related adverse events remained consistent after adjustment in a multivariable analysis (hazard ratio for death, 0.382; P < .001).
There were no significant differences in the median number of cycles of ICIs between those with and those without the adverse events.
The risk of recurrence of immune-related adverse events following the reintroduction of therapy after initial events was a concern, so the improved overall survival among those patients is encouraging, although further investigation is needed, commented lead author Dr. Watson, from the department of oncology, University of Calgary (Alta.).
“It may be, for certain patients with immune-related adverse events, that continued immune-priming is safe and optimizes anticancer response,” he told this news organization. “However, in a retrospective analysis such as ours, selection bias can have an impact.”
“Confirming this finding and better identifying patients who may benefit from resumption will be an area for future investigation,” he said.
Patients who developed immune-related adverse events were more likely to be younger than 50 years (21.8% vs. 13.9%), have normal albumin levels (86.4% vs. 74.8%), and have a more robust Eastern Cooperative Oncology Group status, which is consistent with other studies that have shown survival benefits among those who experience adverse events.
“We, and others, speculate this could be due to such groups having immune systems more ready to respond strongly to immunotherapy,” Dr. Watson explained.
After controlling for age and performance status in the multivariable analysis, however, “immune-related adverse events remained strongly associated with survival, potentially [indicating] that robust responses to immunotherapy lead to both cancer control and immune-related adverse events,” he said.
Overall, “we feel these findings will help clinicians in discussions with patients and in clinical decision-making after adverse events develop,” Dr. Watson said.
Dr. Watson has received personal fees from Apobiologix Canada.
A version of this article first appeared on Medscape.com.
Among Survival is further improved if the immunotherapy is continued after the adverse event develops, a new study confirms.
“In the largest clinical cohort to date, our data support a positive association with overall survival for patients who develop clinically significant immune-related adverse events while receiving combination immune checkpoint blockade, in keeping with other reported series,” the authors wrote.
The study was published online in JAMA Network Open.
Immune-related adverse events are common with these drugs. Severe events of grade 3 or higher occur in 59% of trial patients who receive combination ICI therapy.
The adverse events have increasingly been positively associated with survival. However, the effects for patients with metastatic melanoma, in particular, are less clear. There is little research on the effects in relation to combination therapy with ipilimumab and nivolumab, which is the standard of care for many patients with metastatic melanoma.
To investigate, Alexander S. Watson, MD, and colleagues evaluated data on 492 patients with metastatic melanoma who had been treated with one or more doses of an anti–programmed death 1 agent as single or combination immune checkpoint blockade in the multicenter Alberta Immunotherapy Database from August 2013 to May 2020.
Of these 492 patients, 198 patients (40%) developed immune-related adverse events. The mean age of the patients who developed adverse events was 61.8 years; of those who did not develop adverse events, the mean age was 65.5 years. Men made up 69.2% and 62.2%, respectively.
A total of 288 patients received pembrolizumab as their first ICI therapy, 80 received nivolumab, and 124 received combination blockade with ipilimumab-nivolumab.
Overall, with a median follow-up of 36.6 months, among patients who experienced clinically significant immune-related adverse events, defined as requiring systemic corticosteroids and/or a treatment delay, median overall survival was significantly improved, at 56.3 months, compared with 18.5 months among those who did not experience immune-related adverse events (P < .001).
In addition, among those who received combination ICI treatment, the median overall survival was 56.2 months for those who experienced adverse events versus 19.0 months for those who did not (P < .001).
There were no significant differences in overall survival between those who were and those who were not hospitalized for their immune-related adverse events (P = .53).
For patients who resumed their ICI therapy following the adverse events, overall survival was longer, compared with those who did not resume the therapy (median, 56.3 months vs. 31.5 months; P = .009).
The improvements in overall survival seen with immune-related adverse events remained consistent after adjustment in a multivariable analysis (hazard ratio for death, 0.382; P < .001).
There were no significant differences in the median number of cycles of ICIs between those with and those without the adverse events.
The risk of recurrence of immune-related adverse events following the reintroduction of therapy after initial events was a concern, so the improved overall survival among those patients is encouraging, although further investigation is needed, commented lead author Dr. Watson, from the department of oncology, University of Calgary (Alta.).
“It may be, for certain patients with immune-related adverse events, that continued immune-priming is safe and optimizes anticancer response,” he told this news organization. “However, in a retrospective analysis such as ours, selection bias can have an impact.”
“Confirming this finding and better identifying patients who may benefit from resumption will be an area for future investigation,” he said.
Patients who developed immune-related adverse events were more likely to be younger than 50 years (21.8% vs. 13.9%), have normal albumin levels (86.4% vs. 74.8%), and have a more robust Eastern Cooperative Oncology Group status, which is consistent with other studies that have shown survival benefits among those who experience adverse events.
“We, and others, speculate this could be due to such groups having immune systems more ready to respond strongly to immunotherapy,” Dr. Watson explained.
After controlling for age and performance status in the multivariable analysis, however, “immune-related adverse events remained strongly associated with survival, potentially [indicating] that robust responses to immunotherapy lead to both cancer control and immune-related adverse events,” he said.
Overall, “we feel these findings will help clinicians in discussions with patients and in clinical decision-making after adverse events develop,” Dr. Watson said.
Dr. Watson has received personal fees from Apobiologix Canada.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Topical psoriasis treatments
LAS VEGAS – ,” said Linda Stein Gold, MD, in a presentation at Medscape Live’s annual Las Vegas Dermatology Seminar.
However, when using topical treatments, combination therapy is generally more effective than monotherapy for psoriasis, especially for plaque psoriasis, said Dr. Stein Gold, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit.
Two combination products, calcipotriene/betamethasone (CAL/BDP) and tazarotene/halobetasol lotion, each offer a complimentary mechanism of action that minimizes side effects, with decreased irritation and less atrophy, she said. Calcipotriene/betamethasone (CAL/BDP) is available as a cream or foam, Dr. Stein Gold noted. The cream is engineered for rapid onset, as well as enhanced penetration, she said. CAL/BDP foam also is designed for enhanced penetration, and has been shown to have long-term maintenance efficacy, she said.
The currently available CAL/BDP cream is made using a patented technology known as “PAD,” in which the internal oil of the cream vehicle is stabilized by encapsulation in “a robust aqueous film,” Dr. Stein Gold said, noting that the greater solubility enhances skin penetration. The creation of “a robust oil droplet” addresses the problems associated with the surfactants present in many cream vehicles, namely irritation and impedance of skin penetration of the cream, she said.
In an 8-week study published in 2021, researchers compared CAL/BDP cream with PAD technology to CAL/BDP topical suspension in adults with mild to moderate psoriasis.
Patients randomized to treatment with CAL/BDP cream were significantly more likely to achieve the primary endpoint of Physician Global Assessment (PGA) treatment success than those randomized to the topical solution or vehicle (37.4%, 22.8%, and 3.7%, respectively).
Get proactive to maintain results
With topical psoriasis treatment, a proactive strategy helps maintain results over time, Dr. Stein Gold said. As an example, she cited a study published in 2021. In that study, known as PSO-LONG, which evaluated topical CAL/BDP foam, proactive management with the CAL/BDP foam formulation, “reduced the risk of experiencing relapse by 43%,” compared with reactive management (treatment with the vehicle foam), she said. Patients in the proactive-management group experienced an average of 41 more days in remission, compared with those in the reactive management group over a 1-year period.
Dr. Stein Gold also highlighted the value of tazarotene/halobetasol lotion for psoriasis, which she described as having synergistic efficacy,
She shared data presented at the 2021 Maui Dermatology meeting showing treatment success by 8 weeks with halobetasol/tazarotene with significantly reduced mean scores on measures of itching, dryness, and burning/stinging, compared with those on vehicle.
What’s new and approved
Joining the current topical treatment options for psoriasis is tapinarof, a small molecule that works by down-regulating Th17 cytokines, said Dr. Stein Gold. Tapinarof is Food and Drug Administration approved for treating psoriasis and is being studied in clinical trials for atopic dermatitis, she noted.
Dr. Stein Gold reviewed data from the PSOARING program published in the New England Journal of Medicine that served as a foundation for the FDA approval of tapinarof 1% cream. In the PSOARING 1 and 2 studies, patients with PSORIASIS showed significant improvement compared with vehicle over 12 weeks for the primary endpoint of Physicians’ Global Assessment scores of 0 or 1 (clear or almost clear). In the two studies, 60.7% and 56.9% of patients randomized to tapinarof met the patient-reported outcome of a minimum 4-point improvement in peak pruritus on the numerical rating scale (NRS) from baseline vs. 43.2% and 29.7% of placebo patients in the two studies, respectively.
In PSOARING 1 and 2, folliculitis (mostly mild or moderate), contact dermatitis, headache, pruritus, and dermatitis were the most common treatment-emergent adverse events, occurring in 1% or more of patients. Adverse event profiles for tapinarof are similar to those seen in previous studies, and a long-term extension showed a consistent safety profile, Dr. Stein Gold said.
Another recently approved topical treatment for psoriasis, a cream formulation of roflumilast, a phosphodiesterase (PDE)-4 inhibitor, has shown efficacy for treating plaque psoriasis, she said.
Patients with psoriasis in the DERMIS 1 and DERMIS 2 phase 3 studies randomized to 0.3% roflumilast cream showed significant improvement compared with those randomized to vehicle in terms of Investigator Global Assessment scores of clear or almost clear with an improvement of at least 2 grades from baseline.
Roflumilast foam also has shown success in improving scalp and body psoriasis, but this vehicle and indication has not yet been approved, Dr. Stein Gold said.
Dr. Stein Gold disclosed serving as a consultant or adviser for companies including AbbVie, Amgen, Arcutis, Bristol Myers Squibb, Dermavant, EPI Health, Galderma, Janssen, Incyte, Ortho Dermatologics, Pfizer, Regeneron, Sanofi; UCB, and serving as a speaker or member of speakers’ bureau for Amgen, AbbVie, Incyte, Pfizer, Regeneron, Sanofi, and Sun Research. She also disclosed receiving funding from AbbVie Amgen, Arcutis, Dermata, Dermavant, Galderma, Incyte, Ortho Dermatologics, Pfizer, and UCB.
MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – ,” said Linda Stein Gold, MD, in a presentation at Medscape Live’s annual Las Vegas Dermatology Seminar.
However, when using topical treatments, combination therapy is generally more effective than monotherapy for psoriasis, especially for plaque psoriasis, said Dr. Stein Gold, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit.
Two combination products, calcipotriene/betamethasone (CAL/BDP) and tazarotene/halobetasol lotion, each offer a complimentary mechanism of action that minimizes side effects, with decreased irritation and less atrophy, she said. Calcipotriene/betamethasone (CAL/BDP) is available as a cream or foam, Dr. Stein Gold noted. The cream is engineered for rapid onset, as well as enhanced penetration, she said. CAL/BDP foam also is designed for enhanced penetration, and has been shown to have long-term maintenance efficacy, she said.
The currently available CAL/BDP cream is made using a patented technology known as “PAD,” in which the internal oil of the cream vehicle is stabilized by encapsulation in “a robust aqueous film,” Dr. Stein Gold said, noting that the greater solubility enhances skin penetration. The creation of “a robust oil droplet” addresses the problems associated with the surfactants present in many cream vehicles, namely irritation and impedance of skin penetration of the cream, she said.
In an 8-week study published in 2021, researchers compared CAL/BDP cream with PAD technology to CAL/BDP topical suspension in adults with mild to moderate psoriasis.
Patients randomized to treatment with CAL/BDP cream were significantly more likely to achieve the primary endpoint of Physician Global Assessment (PGA) treatment success than those randomized to the topical solution or vehicle (37.4%, 22.8%, and 3.7%, respectively).
Get proactive to maintain results
With topical psoriasis treatment, a proactive strategy helps maintain results over time, Dr. Stein Gold said. As an example, she cited a study published in 2021. In that study, known as PSO-LONG, which evaluated topical CAL/BDP foam, proactive management with the CAL/BDP foam formulation, “reduced the risk of experiencing relapse by 43%,” compared with reactive management (treatment with the vehicle foam), she said. Patients in the proactive-management group experienced an average of 41 more days in remission, compared with those in the reactive management group over a 1-year period.
Dr. Stein Gold also highlighted the value of tazarotene/halobetasol lotion for psoriasis, which she described as having synergistic efficacy,
She shared data presented at the 2021 Maui Dermatology meeting showing treatment success by 8 weeks with halobetasol/tazarotene with significantly reduced mean scores on measures of itching, dryness, and burning/stinging, compared with those on vehicle.
What’s new and approved
Joining the current topical treatment options for psoriasis is tapinarof, a small molecule that works by down-regulating Th17 cytokines, said Dr. Stein Gold. Tapinarof is Food and Drug Administration approved for treating psoriasis and is being studied in clinical trials for atopic dermatitis, she noted.
Dr. Stein Gold reviewed data from the PSOARING program published in the New England Journal of Medicine that served as a foundation for the FDA approval of tapinarof 1% cream. In the PSOARING 1 and 2 studies, patients with PSORIASIS showed significant improvement compared with vehicle over 12 weeks for the primary endpoint of Physicians’ Global Assessment scores of 0 or 1 (clear or almost clear). In the two studies, 60.7% and 56.9% of patients randomized to tapinarof met the patient-reported outcome of a minimum 4-point improvement in peak pruritus on the numerical rating scale (NRS) from baseline vs. 43.2% and 29.7% of placebo patients in the two studies, respectively.
In PSOARING 1 and 2, folliculitis (mostly mild or moderate), contact dermatitis, headache, pruritus, and dermatitis were the most common treatment-emergent adverse events, occurring in 1% or more of patients. Adverse event profiles for tapinarof are similar to those seen in previous studies, and a long-term extension showed a consistent safety profile, Dr. Stein Gold said.
Another recently approved topical treatment for psoriasis, a cream formulation of roflumilast, a phosphodiesterase (PDE)-4 inhibitor, has shown efficacy for treating plaque psoriasis, she said.
Patients with psoriasis in the DERMIS 1 and DERMIS 2 phase 3 studies randomized to 0.3% roflumilast cream showed significant improvement compared with those randomized to vehicle in terms of Investigator Global Assessment scores of clear or almost clear with an improvement of at least 2 grades from baseline.
Roflumilast foam also has shown success in improving scalp and body psoriasis, but this vehicle and indication has not yet been approved, Dr. Stein Gold said.
Dr. Stein Gold disclosed serving as a consultant or adviser for companies including AbbVie, Amgen, Arcutis, Bristol Myers Squibb, Dermavant, EPI Health, Galderma, Janssen, Incyte, Ortho Dermatologics, Pfizer, Regeneron, Sanofi; UCB, and serving as a speaker or member of speakers’ bureau for Amgen, AbbVie, Incyte, Pfizer, Regeneron, Sanofi, and Sun Research. She also disclosed receiving funding from AbbVie Amgen, Arcutis, Dermata, Dermavant, Galderma, Incyte, Ortho Dermatologics, Pfizer, and UCB.
MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – ,” said Linda Stein Gold, MD, in a presentation at Medscape Live’s annual Las Vegas Dermatology Seminar.
However, when using topical treatments, combination therapy is generally more effective than monotherapy for psoriasis, especially for plaque psoriasis, said Dr. Stein Gold, director of clinical research and division head of dermatology at the Henry Ford Health System, Detroit.
Two combination products, calcipotriene/betamethasone (CAL/BDP) and tazarotene/halobetasol lotion, each offer a complimentary mechanism of action that minimizes side effects, with decreased irritation and less atrophy, she said. Calcipotriene/betamethasone (CAL/BDP) is available as a cream or foam, Dr. Stein Gold noted. The cream is engineered for rapid onset, as well as enhanced penetration, she said. CAL/BDP foam also is designed for enhanced penetration, and has been shown to have long-term maintenance efficacy, she said.
The currently available CAL/BDP cream is made using a patented technology known as “PAD,” in which the internal oil of the cream vehicle is stabilized by encapsulation in “a robust aqueous film,” Dr. Stein Gold said, noting that the greater solubility enhances skin penetration. The creation of “a robust oil droplet” addresses the problems associated with the surfactants present in many cream vehicles, namely irritation and impedance of skin penetration of the cream, she said.
In an 8-week study published in 2021, researchers compared CAL/BDP cream with PAD technology to CAL/BDP topical suspension in adults with mild to moderate psoriasis.
Patients randomized to treatment with CAL/BDP cream were significantly more likely to achieve the primary endpoint of Physician Global Assessment (PGA) treatment success than those randomized to the topical solution or vehicle (37.4%, 22.8%, and 3.7%, respectively).
Get proactive to maintain results
With topical psoriasis treatment, a proactive strategy helps maintain results over time, Dr. Stein Gold said. As an example, she cited a study published in 2021. In that study, known as PSO-LONG, which evaluated topical CAL/BDP foam, proactive management with the CAL/BDP foam formulation, “reduced the risk of experiencing relapse by 43%,” compared with reactive management (treatment with the vehicle foam), she said. Patients in the proactive-management group experienced an average of 41 more days in remission, compared with those in the reactive management group over a 1-year period.
Dr. Stein Gold also highlighted the value of tazarotene/halobetasol lotion for psoriasis, which she described as having synergistic efficacy,
She shared data presented at the 2021 Maui Dermatology meeting showing treatment success by 8 weeks with halobetasol/tazarotene with significantly reduced mean scores on measures of itching, dryness, and burning/stinging, compared with those on vehicle.
What’s new and approved
Joining the current topical treatment options for psoriasis is tapinarof, a small molecule that works by down-regulating Th17 cytokines, said Dr. Stein Gold. Tapinarof is Food and Drug Administration approved for treating psoriasis and is being studied in clinical trials for atopic dermatitis, she noted.
Dr. Stein Gold reviewed data from the PSOARING program published in the New England Journal of Medicine that served as a foundation for the FDA approval of tapinarof 1% cream. In the PSOARING 1 and 2 studies, patients with PSORIASIS showed significant improvement compared with vehicle over 12 weeks for the primary endpoint of Physicians’ Global Assessment scores of 0 or 1 (clear or almost clear). In the two studies, 60.7% and 56.9% of patients randomized to tapinarof met the patient-reported outcome of a minimum 4-point improvement in peak pruritus on the numerical rating scale (NRS) from baseline vs. 43.2% and 29.7% of placebo patients in the two studies, respectively.
In PSOARING 1 and 2, folliculitis (mostly mild or moderate), contact dermatitis, headache, pruritus, and dermatitis were the most common treatment-emergent adverse events, occurring in 1% or more of patients. Adverse event profiles for tapinarof are similar to those seen in previous studies, and a long-term extension showed a consistent safety profile, Dr. Stein Gold said.
Another recently approved topical treatment for psoriasis, a cream formulation of roflumilast, a phosphodiesterase (PDE)-4 inhibitor, has shown efficacy for treating plaque psoriasis, she said.
Patients with psoriasis in the DERMIS 1 and DERMIS 2 phase 3 studies randomized to 0.3% roflumilast cream showed significant improvement compared with those randomized to vehicle in terms of Investigator Global Assessment scores of clear or almost clear with an improvement of at least 2 grades from baseline.
Roflumilast foam also has shown success in improving scalp and body psoriasis, but this vehicle and indication has not yet been approved, Dr. Stein Gold said.
Dr. Stein Gold disclosed serving as a consultant or adviser for companies including AbbVie, Amgen, Arcutis, Bristol Myers Squibb, Dermavant, EPI Health, Galderma, Janssen, Incyte, Ortho Dermatologics, Pfizer, Regeneron, Sanofi; UCB, and serving as a speaker or member of speakers’ bureau for Amgen, AbbVie, Incyte, Pfizer, Regeneron, Sanofi, and Sun Research. She also disclosed receiving funding from AbbVie Amgen, Arcutis, Dermata, Dermavant, Galderma, Incyte, Ortho Dermatologics, Pfizer, and UCB.
MedscapeLive and this news organization are owned by the same parent company.
AT INNOVATIONS IN DERMATOLOGY
Study evaluates features of alopecia areata in Hispanic/Latinx patients
.
Those are among key findings from a retrospective analysis of Hispanic/Latinx patients at the University of California, Irvine (UCI) by Natasha Mesinkovska, MD, PhD, of UCI’s department of dermatology, and her coauthors. The findings were published online in the Journal of the American Academy of Dermatology.
A recent study examined the epidemiology of alopecia areata (AA) in Black patients, wrote Dr. Mesinkovska and coauthors Celine Phong, a UCI medical student, and Amy J. McMichael, MD, professor of dermatology at Wake Forest University, Winston-Salem, N.C. “A similar unmet need exists to describe the characteristics of AA in Hispanic/Latinx (H/L) patients, the prevalent majority in California,” they added.
Drawing from chart reviews, ICD codes, and documented physical exams, they retrospectively identified 197 Hispanic/Latinx patients diagnosed with AA at UCI between 2015 and 2022, including alopecia totalis and alopecia universalis.
Nearly two-thirds of patients with alopecia were female (63%), and their mean age at diagnosis was 33 years. Most patients (79%) presented with patchy pattern AA, 13% had diffuse pattern AA, and only 12% had eyebrow, eyelash, or beard involvement. The most common comorbidity in patients overall was atopy (24%), including allergic rhinitis (12%), asthma (10%), and/or atopic dermatitis (7%).
The authors found that 18% of patients had one or more coexisting autoimmune conditions, most commonly rheumatoid arthritis (9%) and thyroid disease (6%). No patients had celiac disease, myasthenia gravis, or inflammatory bowel disease, but 43% had another dermatologic condition.
In other findings, 22% of patients had vitamin D deficiency, 20% had hyperlipidemia, 18% had obesity, 16% had gastroesophageal reflux disease, and 12% had anemia. At the same time, depression, anxiety, or sleep disorders were identified in 14% of patients.
“Interestingly, the most common autoimmune comorbidity in H/L was rheumatoid arthritis, compared to thyroid disease in Black patients and overall AA patients,” the authors wrote. “This finding may be a reflection of a larger trend, as rheumatoid arthritis in the H/L population has been on the rise.”
The authors acknowledged certain limitations of the study including its small sample size and lack of a control group, and reported having no financial disclosures.
.
Those are among key findings from a retrospective analysis of Hispanic/Latinx patients at the University of California, Irvine (UCI) by Natasha Mesinkovska, MD, PhD, of UCI’s department of dermatology, and her coauthors. The findings were published online in the Journal of the American Academy of Dermatology.
A recent study examined the epidemiology of alopecia areata (AA) in Black patients, wrote Dr. Mesinkovska and coauthors Celine Phong, a UCI medical student, and Amy J. McMichael, MD, professor of dermatology at Wake Forest University, Winston-Salem, N.C. “A similar unmet need exists to describe the characteristics of AA in Hispanic/Latinx (H/L) patients, the prevalent majority in California,” they added.
Drawing from chart reviews, ICD codes, and documented physical exams, they retrospectively identified 197 Hispanic/Latinx patients diagnosed with AA at UCI between 2015 and 2022, including alopecia totalis and alopecia universalis.
Nearly two-thirds of patients with alopecia were female (63%), and their mean age at diagnosis was 33 years. Most patients (79%) presented with patchy pattern AA, 13% had diffuse pattern AA, and only 12% had eyebrow, eyelash, or beard involvement. The most common comorbidity in patients overall was atopy (24%), including allergic rhinitis (12%), asthma (10%), and/or atopic dermatitis (7%).
The authors found that 18% of patients had one or more coexisting autoimmune conditions, most commonly rheumatoid arthritis (9%) and thyroid disease (6%). No patients had celiac disease, myasthenia gravis, or inflammatory bowel disease, but 43% had another dermatologic condition.
In other findings, 22% of patients had vitamin D deficiency, 20% had hyperlipidemia, 18% had obesity, 16% had gastroesophageal reflux disease, and 12% had anemia. At the same time, depression, anxiety, or sleep disorders were identified in 14% of patients.
“Interestingly, the most common autoimmune comorbidity in H/L was rheumatoid arthritis, compared to thyroid disease in Black patients and overall AA patients,” the authors wrote. “This finding may be a reflection of a larger trend, as rheumatoid arthritis in the H/L population has been on the rise.”
The authors acknowledged certain limitations of the study including its small sample size and lack of a control group, and reported having no financial disclosures.
.
Those are among key findings from a retrospective analysis of Hispanic/Latinx patients at the University of California, Irvine (UCI) by Natasha Mesinkovska, MD, PhD, of UCI’s department of dermatology, and her coauthors. The findings were published online in the Journal of the American Academy of Dermatology.
A recent study examined the epidemiology of alopecia areata (AA) in Black patients, wrote Dr. Mesinkovska and coauthors Celine Phong, a UCI medical student, and Amy J. McMichael, MD, professor of dermatology at Wake Forest University, Winston-Salem, N.C. “A similar unmet need exists to describe the characteristics of AA in Hispanic/Latinx (H/L) patients, the prevalent majority in California,” they added.
Drawing from chart reviews, ICD codes, and documented physical exams, they retrospectively identified 197 Hispanic/Latinx patients diagnosed with AA at UCI between 2015 and 2022, including alopecia totalis and alopecia universalis.
Nearly two-thirds of patients with alopecia were female (63%), and their mean age at diagnosis was 33 years. Most patients (79%) presented with patchy pattern AA, 13% had diffuse pattern AA, and only 12% had eyebrow, eyelash, or beard involvement. The most common comorbidity in patients overall was atopy (24%), including allergic rhinitis (12%), asthma (10%), and/or atopic dermatitis (7%).
The authors found that 18% of patients had one or more coexisting autoimmune conditions, most commonly rheumatoid arthritis (9%) and thyroid disease (6%). No patients had celiac disease, myasthenia gravis, or inflammatory bowel disease, but 43% had another dermatologic condition.
In other findings, 22% of patients had vitamin D deficiency, 20% had hyperlipidemia, 18% had obesity, 16% had gastroesophageal reflux disease, and 12% had anemia. At the same time, depression, anxiety, or sleep disorders were identified in 14% of patients.
“Interestingly, the most common autoimmune comorbidity in H/L was rheumatoid arthritis, compared to thyroid disease in Black patients and overall AA patients,” the authors wrote. “This finding may be a reflection of a larger trend, as rheumatoid arthritis in the H/L population has been on the rise.”
The authors acknowledged certain limitations of the study including its small sample size and lack of a control group, and reported having no financial disclosures.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY