User login
Cochrane Review bolsters case that emollients don’t prevent AD
associated with early use of emollients.
The document, published in November 2022, updates a February 2021 version, said Robert Boyle, MD, PhD, senior author of the Cochrane Review and a pediatric allergist at Imperial College London. “The differences were slight,” he told this news organization. “Mainly, we had a little more data about food allergy outcomes, which slightly strengthened the concern about a possible increase in food allergy with emollients; and we had some new genetic information, which allowed us to add some further interaction analyses and confirm that chromosome 11 intergenic variant rs2212434 doesn’t seem to impact the effect – or lack of effect – of emollient on eczema development.”
The updated Cochrane Review concludes that, “based on low‐ to moderate-certainty evidence, skin care interventions such as emollients during the first year of life in healthy infants are probably not effective for preventing eczema; may increase risk of food allergy; and probably increase risk of skin infection.”
The latest publication should strengthen clinicians’ confidence in not recommending emollient use for preventing AD in at-risk infants – however, that message is being diluted by a stream of contradictory conclusions from poor-quality systematic reviews, say Dr. Boyle and two coauthors. “It’s a systematic problem of people churning out endless systematic reviews without much rigor,” explained the lead author Maeve Kelleher, MD, from Children’s Health Ireland, Crumlin. There have been “misleading systematic reviews published, often in high-ranking journals,” agreed Dr. Boyle.
“I have been an advocate of systematic reviews for the last 20 years, but they have gone completely out of control,” added Hywel Williams, MD, PhD, another of the Cochrane Review coauthors, who is professor of dermato-epidemiology and codirector of the Centre of Evidence Based Dermatology, at Nottingham (England) University Hospitals NHS Trust. In an editorial, published last year, Dr. Williams even posed the question: “Are Dermatology Systematic Reviews Spinning Out of Control?” in which he blamed “the misrepresentation of study results” – which he calls “the sin of spin” – for degrading the quality of science in dermatology.
“The field has become a ‘sausage machine’ industry that undermines the value of systematic reviews in providing a summary of the best evidence to inform patient care,” he wrote. “Fewer systematic reviews are needed in dermatology,” but “better ones” are needed, he continued, calling for all systematic reviews to be registered prospectively, and reported according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines.
Earlier this year, in a letter to the editor, Dr. Kelleher, Dr. Boyle, Dr. Williams, and several others outlined their concerns after a systemic review and meta-analysis was published, “which came to very different conclusions” than their Cochrane Review.
“It is quite common to see non-Cochrane reviews published in leading specialty journals, which interpret data in a more positive light than Cochrane reviews, which have assessed a similar dataset/topic,” Dr. Boyle said in the interview.
Such concerns also apply to the publication of another systematic review that was recently published. “Overall, early application of emollients is an effective strategy for preventing AD development in high-risk infants,” reported senior author Xiaojing Kang, MD, PhD, from People’s Hospital of Xinjiang Uygur Autonomous Region, Urumchi, China, and coauthors, who could not be reached for comment. In their discussion, the authors cite several criticisms of the Cochrane Review: that it included two meeting abstracts and two “ineligible” studies; did not do subgroup analysis of high-risk infants; did not look at different types of emollients; and did not examine the risk of food sensitization.
“A Cochrane Review can be quite a large and complex document to negotiate for those who are not very familiar with Cochrane’s methodology,” said Dr. Boyle. He dismissed the criticism, saying “we did do subgroup analysis of high risk infants, we did look at different types of emollient, and we did look at food sensitization and food allergy risk. We only included eligible studies. … Certainly we would include abstracts of trials, which are not reported in any other form, in order to capture as complete a picture.”
Ultimately, Dr. Boyle said, the discrepancy in conclusions between such systematic reviews and the Cochrane Review relates to quality of methodology. “Our Cochrane review was an individual participant data (IPD) meta-analysis, meaning that authors of the main trials in this area shared their original datasets with us,” he said in the interview. “This is the ‘gold standard’ in systematic reviews, and allowed us to check data/ query inconsistencies and to apply a single-analysis methodology across all studies. It also allowed us to undertake some analyses, which are just not possible in aggregate data analysis based on published work without IPD.”
The most recently published systematic review had no registered protocol, “so, there is no transparency about the methods used,” he noted. “It is free and simple to register a protocol – multiple websites such as PROSPERO, open science framework, and zenodo allow this,” he said “In the journal I edit, we use availability of a registered protocol as a marker of quality. We find that systematic reviews with no registered protocol are almost universally poor quality.”
Dr. Williams is a founding member and coordinating editor of the Cochrane Skin Group 1998 to 2017. Dr. Boyle was paid by Cochrane for senior editor work, until recently, and had no other relevant disclosures. Dr. Kelleher had no relevant disclosures.
associated with early use of emollients.
The document, published in November 2022, updates a February 2021 version, said Robert Boyle, MD, PhD, senior author of the Cochrane Review and a pediatric allergist at Imperial College London. “The differences were slight,” he told this news organization. “Mainly, we had a little more data about food allergy outcomes, which slightly strengthened the concern about a possible increase in food allergy with emollients; and we had some new genetic information, which allowed us to add some further interaction analyses and confirm that chromosome 11 intergenic variant rs2212434 doesn’t seem to impact the effect – or lack of effect – of emollient on eczema development.”
The updated Cochrane Review concludes that, “based on low‐ to moderate-certainty evidence, skin care interventions such as emollients during the first year of life in healthy infants are probably not effective for preventing eczema; may increase risk of food allergy; and probably increase risk of skin infection.”
The latest publication should strengthen clinicians’ confidence in not recommending emollient use for preventing AD in at-risk infants – however, that message is being diluted by a stream of contradictory conclusions from poor-quality systematic reviews, say Dr. Boyle and two coauthors. “It’s a systematic problem of people churning out endless systematic reviews without much rigor,” explained the lead author Maeve Kelleher, MD, from Children’s Health Ireland, Crumlin. There have been “misleading systematic reviews published, often in high-ranking journals,” agreed Dr. Boyle.
“I have been an advocate of systematic reviews for the last 20 years, but they have gone completely out of control,” added Hywel Williams, MD, PhD, another of the Cochrane Review coauthors, who is professor of dermato-epidemiology and codirector of the Centre of Evidence Based Dermatology, at Nottingham (England) University Hospitals NHS Trust. In an editorial, published last year, Dr. Williams even posed the question: “Are Dermatology Systematic Reviews Spinning Out of Control?” in which he blamed “the misrepresentation of study results” – which he calls “the sin of spin” – for degrading the quality of science in dermatology.
“The field has become a ‘sausage machine’ industry that undermines the value of systematic reviews in providing a summary of the best evidence to inform patient care,” he wrote. “Fewer systematic reviews are needed in dermatology,” but “better ones” are needed, he continued, calling for all systematic reviews to be registered prospectively, and reported according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines.
Earlier this year, in a letter to the editor, Dr. Kelleher, Dr. Boyle, Dr. Williams, and several others outlined their concerns after a systemic review and meta-analysis was published, “which came to very different conclusions” than their Cochrane Review.
“It is quite common to see non-Cochrane reviews published in leading specialty journals, which interpret data in a more positive light than Cochrane reviews, which have assessed a similar dataset/topic,” Dr. Boyle said in the interview.
Such concerns also apply to the publication of another systematic review that was recently published. “Overall, early application of emollients is an effective strategy for preventing AD development in high-risk infants,” reported senior author Xiaojing Kang, MD, PhD, from People’s Hospital of Xinjiang Uygur Autonomous Region, Urumchi, China, and coauthors, who could not be reached for comment. In their discussion, the authors cite several criticisms of the Cochrane Review: that it included two meeting abstracts and two “ineligible” studies; did not do subgroup analysis of high-risk infants; did not look at different types of emollients; and did not examine the risk of food sensitization.
“A Cochrane Review can be quite a large and complex document to negotiate for those who are not very familiar with Cochrane’s methodology,” said Dr. Boyle. He dismissed the criticism, saying “we did do subgroup analysis of high risk infants, we did look at different types of emollient, and we did look at food sensitization and food allergy risk. We only included eligible studies. … Certainly we would include abstracts of trials, which are not reported in any other form, in order to capture as complete a picture.”
Ultimately, Dr. Boyle said, the discrepancy in conclusions between such systematic reviews and the Cochrane Review relates to quality of methodology. “Our Cochrane review was an individual participant data (IPD) meta-analysis, meaning that authors of the main trials in this area shared their original datasets with us,” he said in the interview. “This is the ‘gold standard’ in systematic reviews, and allowed us to check data/ query inconsistencies and to apply a single-analysis methodology across all studies. It also allowed us to undertake some analyses, which are just not possible in aggregate data analysis based on published work without IPD.”
The most recently published systematic review had no registered protocol, “so, there is no transparency about the methods used,” he noted. “It is free and simple to register a protocol – multiple websites such as PROSPERO, open science framework, and zenodo allow this,” he said “In the journal I edit, we use availability of a registered protocol as a marker of quality. We find that systematic reviews with no registered protocol are almost universally poor quality.”
Dr. Williams is a founding member and coordinating editor of the Cochrane Skin Group 1998 to 2017. Dr. Boyle was paid by Cochrane for senior editor work, until recently, and had no other relevant disclosures. Dr. Kelleher had no relevant disclosures.
associated with early use of emollients.
The document, published in November 2022, updates a February 2021 version, said Robert Boyle, MD, PhD, senior author of the Cochrane Review and a pediatric allergist at Imperial College London. “The differences were slight,” he told this news organization. “Mainly, we had a little more data about food allergy outcomes, which slightly strengthened the concern about a possible increase in food allergy with emollients; and we had some new genetic information, which allowed us to add some further interaction analyses and confirm that chromosome 11 intergenic variant rs2212434 doesn’t seem to impact the effect – or lack of effect – of emollient on eczema development.”
The updated Cochrane Review concludes that, “based on low‐ to moderate-certainty evidence, skin care interventions such as emollients during the first year of life in healthy infants are probably not effective for preventing eczema; may increase risk of food allergy; and probably increase risk of skin infection.”
The latest publication should strengthen clinicians’ confidence in not recommending emollient use for preventing AD in at-risk infants – however, that message is being diluted by a stream of contradictory conclusions from poor-quality systematic reviews, say Dr. Boyle and two coauthors. “It’s a systematic problem of people churning out endless systematic reviews without much rigor,” explained the lead author Maeve Kelleher, MD, from Children’s Health Ireland, Crumlin. There have been “misleading systematic reviews published, often in high-ranking journals,” agreed Dr. Boyle.
“I have been an advocate of systematic reviews for the last 20 years, but they have gone completely out of control,” added Hywel Williams, MD, PhD, another of the Cochrane Review coauthors, who is professor of dermato-epidemiology and codirector of the Centre of Evidence Based Dermatology, at Nottingham (England) University Hospitals NHS Trust. In an editorial, published last year, Dr. Williams even posed the question: “Are Dermatology Systematic Reviews Spinning Out of Control?” in which he blamed “the misrepresentation of study results” – which he calls “the sin of spin” – for degrading the quality of science in dermatology.
“The field has become a ‘sausage machine’ industry that undermines the value of systematic reviews in providing a summary of the best evidence to inform patient care,” he wrote. “Fewer systematic reviews are needed in dermatology,” but “better ones” are needed, he continued, calling for all systematic reviews to be registered prospectively, and reported according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines.
Earlier this year, in a letter to the editor, Dr. Kelleher, Dr. Boyle, Dr. Williams, and several others outlined their concerns after a systemic review and meta-analysis was published, “which came to very different conclusions” than their Cochrane Review.
“It is quite common to see non-Cochrane reviews published in leading specialty journals, which interpret data in a more positive light than Cochrane reviews, which have assessed a similar dataset/topic,” Dr. Boyle said in the interview.
Such concerns also apply to the publication of another systematic review that was recently published. “Overall, early application of emollients is an effective strategy for preventing AD development in high-risk infants,” reported senior author Xiaojing Kang, MD, PhD, from People’s Hospital of Xinjiang Uygur Autonomous Region, Urumchi, China, and coauthors, who could not be reached for comment. In their discussion, the authors cite several criticisms of the Cochrane Review: that it included two meeting abstracts and two “ineligible” studies; did not do subgroup analysis of high-risk infants; did not look at different types of emollients; and did not examine the risk of food sensitization.
“A Cochrane Review can be quite a large and complex document to negotiate for those who are not very familiar with Cochrane’s methodology,” said Dr. Boyle. He dismissed the criticism, saying “we did do subgroup analysis of high risk infants, we did look at different types of emollient, and we did look at food sensitization and food allergy risk. We only included eligible studies. … Certainly we would include abstracts of trials, which are not reported in any other form, in order to capture as complete a picture.”
Ultimately, Dr. Boyle said, the discrepancy in conclusions between such systematic reviews and the Cochrane Review relates to quality of methodology. “Our Cochrane review was an individual participant data (IPD) meta-analysis, meaning that authors of the main trials in this area shared their original datasets with us,” he said in the interview. “This is the ‘gold standard’ in systematic reviews, and allowed us to check data/ query inconsistencies and to apply a single-analysis methodology across all studies. It also allowed us to undertake some analyses, which are just not possible in aggregate data analysis based on published work without IPD.”
The most recently published systematic review had no registered protocol, “so, there is no transparency about the methods used,” he noted. “It is free and simple to register a protocol – multiple websites such as PROSPERO, open science framework, and zenodo allow this,” he said “In the journal I edit, we use availability of a registered protocol as a marker of quality. We find that systematic reviews with no registered protocol are almost universally poor quality.”
Dr. Williams is a founding member and coordinating editor of the Cochrane Skin Group 1998 to 2017. Dr. Boyle was paid by Cochrane for senior editor work, until recently, and had no other relevant disclosures. Dr. Kelleher had no relevant disclosures.
FROM THE COCHRANE REVIEW
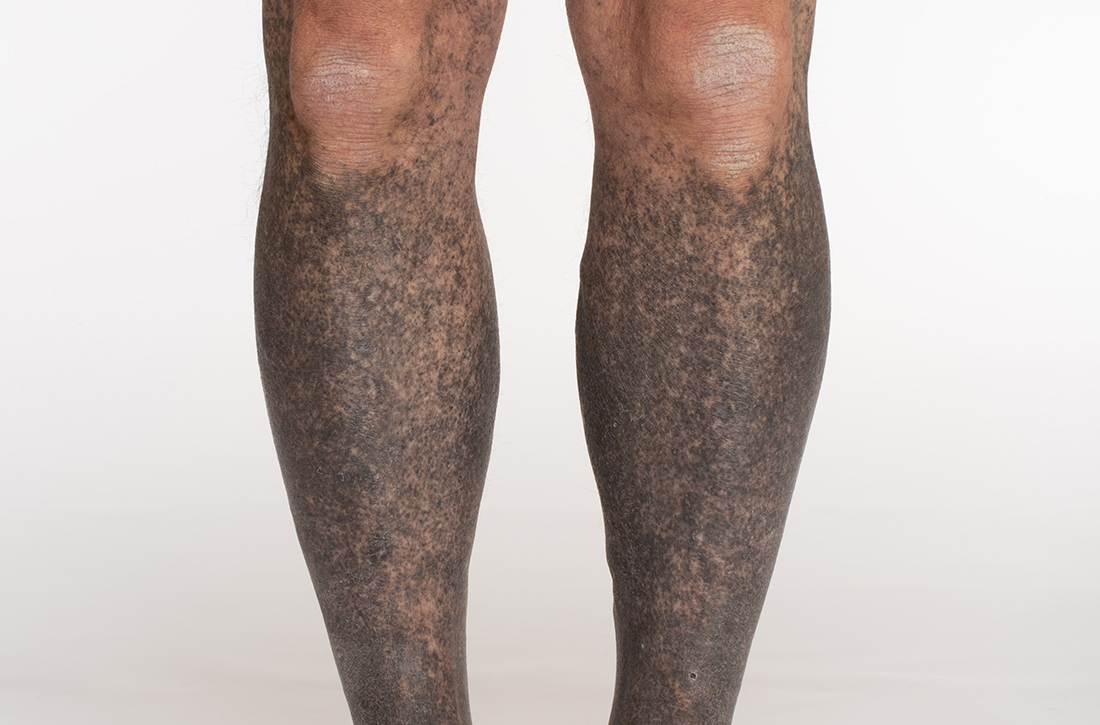
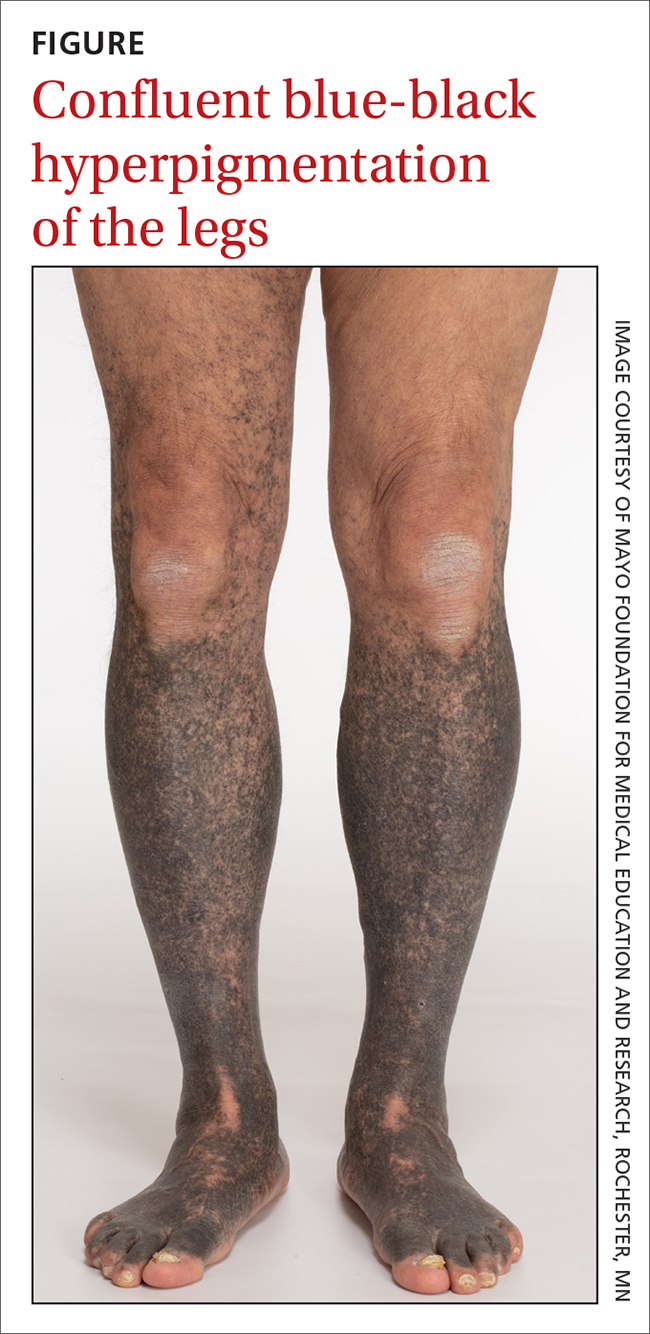
Blue-black hyperpigmentation on the extremities
A 68-year-old man with type 2 diabetes presented with progressive hyperpigmentation of the lower extremities and face over the past 3 years. Clinical examination revealed confluent, blue-black hyperpigmentation of the lower extremities (Figure), upper extremities, neck, and face. Laboratory tests and arterial studies were within normal ranges. The patient’s medication list included lisinopril 10 mg/d, metformin 1000 mg twice daily, minocycline 100 mg twice daily, and omeprazole 20 mg/d.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Minocycline-induced hyperpigmentation
Hyperpigmentation is a rare but not uncommon adverse effect of long-term minocycline use. In this case, our patient had been taking minocycline for more than 5 years. When seen in our clinic, he said he could not remember why he was taking minocycline and incorrectly assumed it was for his diabetes. Chart review of outside records revealed that it had been prescribed, and refilled annually, by his primary physician for rosacea.
Minocycline hyperpigmentation is subdivided into 3 types:
- Type I manifests with blue-black discoloration in previously inflamed areas of skin.
- Type II manifests with blue-gray pigmentation in previously normal skin areas.
- Type III manifests diffusely with muddy-brown hyperpigmentation on photoexposed skin.
Furthermore, noncutaneous manifestations may occur on the sclera, nails, ear cartilage, bone, oral mucosa, teeth, and thyroid gland.1
Diagnosis focuses on identifying the source
Minocycline is one of many drugs that can induce hyperpigmentation of the skin. In addition to history, examination, and review of the patient’s medication list, there are some clues on exam that may suggest a certain type of medication at play.
Continue to: Antimalarials
Antimalarials. Chloroquine, hydroxychloroquine, and quinacrine can cause blue-black skin hyperpigmentation in as many as 25% of patients. Common locations include the shins, face, oral mucosa, and subungual skin. This hyperpigmentation rarely fully resolves.2
Amiodarone. Hyperpigmentation secondary to amiodarone use typically is slate-gray in color and involves photoexposed skin. Patients should be counseled that pigmentation may—but does not always—fade with time after discontinuation of the drug.2
Heavy metals. Argyria results from exposure to silver, either ingested orally or applied externally. A common cause of argyria is ingestion of excessive amounts of silver-containing supplements.3 Affected patients present with diffuse slate-gray discoloration of the skin.
Other metals implicated in skin hyperpigmentation include arsenic, gold, mercury, and iron. Review of all supplements and herbal remedies in patients presenting with skin hyperpigmentation is crucial.
Bleomycin is a chemotherapeutic agent with a rare but unique adverse effect of inducing flagellate hyperpigmentation that favors the chest, abdomen, or back. This may be induced by trauma or scratching and is often transient. Hyperpigmentation can occur secondary to either intravenous or intralesional injection of the medication.2
Continue to: In addition to medication...
In addition to medication- or supplement-induced hyperpigmentation, there is a physiologic source that should be considered when a patient presents with lower-extremity hyperpigmentation:
Stasis hyperpigmentation. Patients with chronic venous insufficiency may present with hyperpigmentation of the lower extremities. Commonly due to dysfunctional venous valves or obstruction, stasis hyperpigmentation manifests with red-brown discoloration from dermal hemosiderin deposition.4
Unlike our patient, those with stasis hyperpigmentation may present symptomatically, with associated dry skin, pruritus, induration, and inflammation. Treatment involves management of the underlying venous insufficiency.4
When there’s no obvious cause, be prepared to dig deeper
At the time of initial assessment, a thorough review of systems and detailed medication history, including over-the-counter supplements, should be obtained. Physical examination revealing diffuse, generalized hyperpigmentation with no reliable culprit medication in the patient’s history warrants further laboratory evaluation. This includes ordering renal and liver studies and tests for thyroid-stimulating hormone and ferritin and cortisol levels to rule out metabolic or endocrine hyperpigmentation disorders.
Stopping the offending medication is the first step
Discontinuation of the offending medication may result in mild improvement in skin hyperpigmentation over time. Some patients may not experience any improvement. If improvement occurs, it is important to educate patients that it can take several months to years. Dermatology guidelines favor discontinuation of antibiotics for acne or rosacea after 3 to 6 months to avoid bacterial resistance.5 Worsening hyperpigmentation despite medication discontinuation warrants further work-up.
Patients who are distressed by persistent hyperpigmentation can be treated using picosecond or Q-switched lasers.6
Our patient was advised to discontinue the minocycline. Three test spots on his face were treated with pulsed-dye laser, carbon dioxide laser, and dermabrasion. The patient noted that the spots responded better to the carbon dioxide laser and dermabrasion compared to the pulsed-dye laser. He did not follow up for further treatment.
1. Wetter DA. Minocycline hyperpigmentation. Mayo Clin Proc. 2012;87:e33. doi: 10.1016/j.mayocp.2012.02.013
2. Chang MW. Chapter 67: Disorders of hyperpigmentation. In: Bolognia J, Schaffer J, Cerroni L, et al (eds). Dermatology. 4th ed. Elsevier; 2018:1122-1124.
3. Bowden LP, Royer MC, Hallman JR, et al. Rapid onset of argyria induced by a silver-containing dietary supplement. J Cutan Pathol. 2011;38:832-835. doi: 10.1111/j.1600-0560.2011.01755.x
4. Patterson J. Stasis dermatitis. In: Weedon’s Skin Pathology. 3rd ed. Churchill Livingstone Elsevier;2010: 121-153.
5. Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945-73.e33. doi: 10.1016/j.jaad.2015.12.037
6. Barrett T, de Zwaan S. Picosecond alexandrite laser is superior to Q-switched Nd:YAG laser in treatment of minocycline-induced hyperpigmentation: a case study and review of the literature. J Cosmet Laser Ther. 2018;20:387-390. doi: 10.1080/14764172.2017.1418514
A 68-year-old man with type 2 diabetes presented with progressive hyperpigmentation of the lower extremities and face over the past 3 years. Clinical examination revealed confluent, blue-black hyperpigmentation of the lower extremities (Figure), upper extremities, neck, and face. Laboratory tests and arterial studies were within normal ranges. The patient’s medication list included lisinopril 10 mg/d, metformin 1000 mg twice daily, minocycline 100 mg twice daily, and omeprazole 20 mg/d.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Minocycline-induced hyperpigmentation
Hyperpigmentation is a rare but not uncommon adverse effect of long-term minocycline use. In this case, our patient had been taking minocycline for more than 5 years. When seen in our clinic, he said he could not remember why he was taking minocycline and incorrectly assumed it was for his diabetes. Chart review of outside records revealed that it had been prescribed, and refilled annually, by his primary physician for rosacea.
Minocycline hyperpigmentation is subdivided into 3 types:
- Type I manifests with blue-black discoloration in previously inflamed areas of skin.
- Type II manifests with blue-gray pigmentation in previously normal skin areas.
- Type III manifests diffusely with muddy-brown hyperpigmentation on photoexposed skin.
Furthermore, noncutaneous manifestations may occur on the sclera, nails, ear cartilage, bone, oral mucosa, teeth, and thyroid gland.1
Diagnosis focuses on identifying the source
Minocycline is one of many drugs that can induce hyperpigmentation of the skin. In addition to history, examination, and review of the patient’s medication list, there are some clues on exam that may suggest a certain type of medication at play.
Continue to: Antimalarials
Antimalarials. Chloroquine, hydroxychloroquine, and quinacrine can cause blue-black skin hyperpigmentation in as many as 25% of patients. Common locations include the shins, face, oral mucosa, and subungual skin. This hyperpigmentation rarely fully resolves.2
Amiodarone. Hyperpigmentation secondary to amiodarone use typically is slate-gray in color and involves photoexposed skin. Patients should be counseled that pigmentation may—but does not always—fade with time after discontinuation of the drug.2
Heavy metals. Argyria results from exposure to silver, either ingested orally or applied externally. A common cause of argyria is ingestion of excessive amounts of silver-containing supplements.3 Affected patients present with diffuse slate-gray discoloration of the skin.
Other metals implicated in skin hyperpigmentation include arsenic, gold, mercury, and iron. Review of all supplements and herbal remedies in patients presenting with skin hyperpigmentation is crucial.
Bleomycin is a chemotherapeutic agent with a rare but unique adverse effect of inducing flagellate hyperpigmentation that favors the chest, abdomen, or back. This may be induced by trauma or scratching and is often transient. Hyperpigmentation can occur secondary to either intravenous or intralesional injection of the medication.2
Continue to: In addition to medication...
In addition to medication- or supplement-induced hyperpigmentation, there is a physiologic source that should be considered when a patient presents with lower-extremity hyperpigmentation:
Stasis hyperpigmentation. Patients with chronic venous insufficiency may present with hyperpigmentation of the lower extremities. Commonly due to dysfunctional venous valves or obstruction, stasis hyperpigmentation manifests with red-brown discoloration from dermal hemosiderin deposition.4
Unlike our patient, those with stasis hyperpigmentation may present symptomatically, with associated dry skin, pruritus, induration, and inflammation. Treatment involves management of the underlying venous insufficiency.4
When there’s no obvious cause, be prepared to dig deeper
At the time of initial assessment, a thorough review of systems and detailed medication history, including over-the-counter supplements, should be obtained. Physical examination revealing diffuse, generalized hyperpigmentation with no reliable culprit medication in the patient’s history warrants further laboratory evaluation. This includes ordering renal and liver studies and tests for thyroid-stimulating hormone and ferritin and cortisol levels to rule out metabolic or endocrine hyperpigmentation disorders.
Stopping the offending medication is the first step
Discontinuation of the offending medication may result in mild improvement in skin hyperpigmentation over time. Some patients may not experience any improvement. If improvement occurs, it is important to educate patients that it can take several months to years. Dermatology guidelines favor discontinuation of antibiotics for acne or rosacea after 3 to 6 months to avoid bacterial resistance.5 Worsening hyperpigmentation despite medication discontinuation warrants further work-up.
Patients who are distressed by persistent hyperpigmentation can be treated using picosecond or Q-switched lasers.6
Our patient was advised to discontinue the minocycline. Three test spots on his face were treated with pulsed-dye laser, carbon dioxide laser, and dermabrasion. The patient noted that the spots responded better to the carbon dioxide laser and dermabrasion compared to the pulsed-dye laser. He did not follow up for further treatment.
A 68-year-old man with type 2 diabetes presented with progressive hyperpigmentation of the lower extremities and face over the past 3 years. Clinical examination revealed confluent, blue-black hyperpigmentation of the lower extremities (Figure), upper extremities, neck, and face. Laboratory tests and arterial studies were within normal ranges. The patient’s medication list included lisinopril 10 mg/d, metformin 1000 mg twice daily, minocycline 100 mg twice daily, and omeprazole 20 mg/d.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Minocycline-induced hyperpigmentation
Hyperpigmentation is a rare but not uncommon adverse effect of long-term minocycline use. In this case, our patient had been taking minocycline for more than 5 years. When seen in our clinic, he said he could not remember why he was taking minocycline and incorrectly assumed it was for his diabetes. Chart review of outside records revealed that it had been prescribed, and refilled annually, by his primary physician for rosacea.
Minocycline hyperpigmentation is subdivided into 3 types:
- Type I manifests with blue-black discoloration in previously inflamed areas of skin.
- Type II manifests with blue-gray pigmentation in previously normal skin areas.
- Type III manifests diffusely with muddy-brown hyperpigmentation on photoexposed skin.
Furthermore, noncutaneous manifestations may occur on the sclera, nails, ear cartilage, bone, oral mucosa, teeth, and thyroid gland.1
Diagnosis focuses on identifying the source
Minocycline is one of many drugs that can induce hyperpigmentation of the skin. In addition to history, examination, and review of the patient’s medication list, there are some clues on exam that may suggest a certain type of medication at play.
Continue to: Antimalarials
Antimalarials. Chloroquine, hydroxychloroquine, and quinacrine can cause blue-black skin hyperpigmentation in as many as 25% of patients. Common locations include the shins, face, oral mucosa, and subungual skin. This hyperpigmentation rarely fully resolves.2
Amiodarone. Hyperpigmentation secondary to amiodarone use typically is slate-gray in color and involves photoexposed skin. Patients should be counseled that pigmentation may—but does not always—fade with time after discontinuation of the drug.2
Heavy metals. Argyria results from exposure to silver, either ingested orally or applied externally. A common cause of argyria is ingestion of excessive amounts of silver-containing supplements.3 Affected patients present with diffuse slate-gray discoloration of the skin.
Other metals implicated in skin hyperpigmentation include arsenic, gold, mercury, and iron. Review of all supplements and herbal remedies in patients presenting with skin hyperpigmentation is crucial.
Bleomycin is a chemotherapeutic agent with a rare but unique adverse effect of inducing flagellate hyperpigmentation that favors the chest, abdomen, or back. This may be induced by trauma or scratching and is often transient. Hyperpigmentation can occur secondary to either intravenous or intralesional injection of the medication.2
Continue to: In addition to medication...
In addition to medication- or supplement-induced hyperpigmentation, there is a physiologic source that should be considered when a patient presents with lower-extremity hyperpigmentation:
Stasis hyperpigmentation. Patients with chronic venous insufficiency may present with hyperpigmentation of the lower extremities. Commonly due to dysfunctional venous valves or obstruction, stasis hyperpigmentation manifests with red-brown discoloration from dermal hemosiderin deposition.4
Unlike our patient, those with stasis hyperpigmentation may present symptomatically, with associated dry skin, pruritus, induration, and inflammation. Treatment involves management of the underlying venous insufficiency.4
When there’s no obvious cause, be prepared to dig deeper
At the time of initial assessment, a thorough review of systems and detailed medication history, including over-the-counter supplements, should be obtained. Physical examination revealing diffuse, generalized hyperpigmentation with no reliable culprit medication in the patient’s history warrants further laboratory evaluation. This includes ordering renal and liver studies and tests for thyroid-stimulating hormone and ferritin and cortisol levels to rule out metabolic or endocrine hyperpigmentation disorders.
Stopping the offending medication is the first step
Discontinuation of the offending medication may result in mild improvement in skin hyperpigmentation over time. Some patients may not experience any improvement. If improvement occurs, it is important to educate patients that it can take several months to years. Dermatology guidelines favor discontinuation of antibiotics for acne or rosacea after 3 to 6 months to avoid bacterial resistance.5 Worsening hyperpigmentation despite medication discontinuation warrants further work-up.
Patients who are distressed by persistent hyperpigmentation can be treated using picosecond or Q-switched lasers.6
Our patient was advised to discontinue the minocycline. Three test spots on his face were treated with pulsed-dye laser, carbon dioxide laser, and dermabrasion. The patient noted that the spots responded better to the carbon dioxide laser and dermabrasion compared to the pulsed-dye laser. He did not follow up for further treatment.
1. Wetter DA. Minocycline hyperpigmentation. Mayo Clin Proc. 2012;87:e33. doi: 10.1016/j.mayocp.2012.02.013
2. Chang MW. Chapter 67: Disorders of hyperpigmentation. In: Bolognia J, Schaffer J, Cerroni L, et al (eds). Dermatology. 4th ed. Elsevier; 2018:1122-1124.
3. Bowden LP, Royer MC, Hallman JR, et al. Rapid onset of argyria induced by a silver-containing dietary supplement. J Cutan Pathol. 2011;38:832-835. doi: 10.1111/j.1600-0560.2011.01755.x
4. Patterson J. Stasis dermatitis. In: Weedon’s Skin Pathology. 3rd ed. Churchill Livingstone Elsevier;2010: 121-153.
5. Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945-73.e33. doi: 10.1016/j.jaad.2015.12.037
6. Barrett T, de Zwaan S. Picosecond alexandrite laser is superior to Q-switched Nd:YAG laser in treatment of minocycline-induced hyperpigmentation: a case study and review of the literature. J Cosmet Laser Ther. 2018;20:387-390. doi: 10.1080/14764172.2017.1418514
1. Wetter DA. Minocycline hyperpigmentation. Mayo Clin Proc. 2012;87:e33. doi: 10.1016/j.mayocp.2012.02.013
2. Chang MW. Chapter 67: Disorders of hyperpigmentation. In: Bolognia J, Schaffer J, Cerroni L, et al (eds). Dermatology. 4th ed. Elsevier; 2018:1122-1124.
3. Bowden LP, Royer MC, Hallman JR, et al. Rapid onset of argyria induced by a silver-containing dietary supplement. J Cutan Pathol. 2011;38:832-835. doi: 10.1111/j.1600-0560.2011.01755.x
4. Patterson J. Stasis dermatitis. In: Weedon’s Skin Pathology. 3rd ed. Churchill Livingstone Elsevier;2010: 121-153.
5. Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74:945-73.e33. doi: 10.1016/j.jaad.2015.12.037
6. Barrett T, de Zwaan S. Picosecond alexandrite laser is superior to Q-switched Nd:YAG laser in treatment of minocycline-induced hyperpigmentation: a case study and review of the literature. J Cosmet Laser Ther. 2018;20:387-390. doi: 10.1080/14764172.2017.1418514
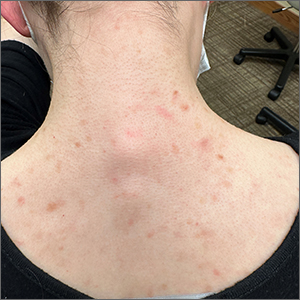
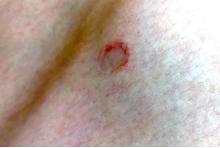
Incidental skin finding
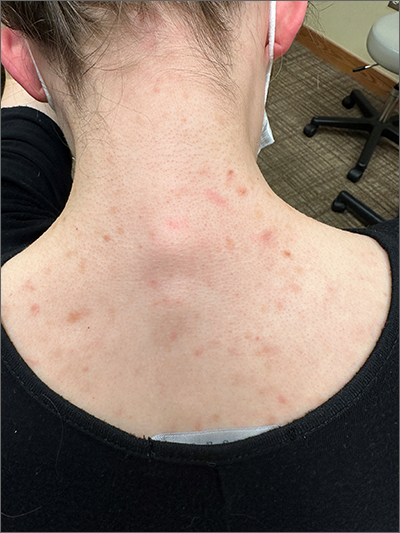
This patient was given a diagnosis of cutaneous mastocytosis. The condition was previously known as urticaria pigmentosa, but in 2016 the World Health Organization reclassified the disease to better suit its pathophysiology as a myeloid cell disorder.1
As the name suggests, this condition—which involves lesions in a sporadic, truncal distribution—involves overactivation of mastocytes at the tissue level from various stimuli, resulting in histocyte degranulation and hyperpigmentation. The exact cause is unknown. What is known is that it is often associated with other allergic or immunologic conditions and is thought to be related to mutations in the gene for CD-117’s receptor for tyrosine kinase.1 Incidence is similar to that of asthma, in that it occurs more often in younger patients; a majority of affected individuals will grow out of the disease by adolescence.1
Although most patients do not experience severe symptomatology, it is still important to differentiate cutaneous vs systemic mastocytosis. If a patient presents with inexplicable systemic symptoms of malaise, vague abdominal pain, heartburn, or flushing, the physician should consider systemic mastocytosis, idiopathic anaphylaxis, or hereditary alpha-tryptasemia.2
The test of choice is a serum tryptase test; levels will be elevated with systemic mastocytosis. Consider obtaining a skin biopsy if lesions are ambiguous or nondistinct.
There is no definitive cure for systemic or cutaneous mastocytosis, so treatment is directed at symptoms. Start by advising patients to avoid triggers and to refrain from scratching the affected areas. Topical antihistamines and oral nonsedating antihistamines can be helpful. If symptoms are more severe, refer the patient to an allergist/immunologist or to a hematologist for further medical management.2
The patient in this case had no systemic symptoms, so she was advised to continue taking oral loratadine 10 mg/d, which had been helpful, and to avoid rubbing her skin.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Murtaza Rizvi, MD, and Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Arber DA, Orazi A, Hasserjian R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127:2391-2405. doi: 10.1182/blood-2016-03-643544.
2. Hartmann K, Escribano L, Grattan C, et al. Cutaneous manifestations in patients with mastocytosis: Consensus report of the European Competence Network on Mastocytosis; the American Academy of Allergy, Asthma & Immunology; and the European Academy of Allergology and Clinical Immunology. J Allergy Clin Immunol 2016; 137:35-45. doi: 10.1016/j.jaci.2015.08.034

This patient was given a diagnosis of cutaneous mastocytosis. The condition was previously known as urticaria pigmentosa, but in 2016 the World Health Organization reclassified the disease to better suit its pathophysiology as a myeloid cell disorder.1
As the name suggests, this condition—which involves lesions in a sporadic, truncal distribution—involves overactivation of mastocytes at the tissue level from various stimuli, resulting in histocyte degranulation and hyperpigmentation. The exact cause is unknown. What is known is that it is often associated with other allergic or immunologic conditions and is thought to be related to mutations in the gene for CD-117’s receptor for tyrosine kinase.1 Incidence is similar to that of asthma, in that it occurs more often in younger patients; a majority of affected individuals will grow out of the disease by adolescence.1
Although most patients do not experience severe symptomatology, it is still important to differentiate cutaneous vs systemic mastocytosis. If a patient presents with inexplicable systemic symptoms of malaise, vague abdominal pain, heartburn, or flushing, the physician should consider systemic mastocytosis, idiopathic anaphylaxis, or hereditary alpha-tryptasemia.2
The test of choice is a serum tryptase test; levels will be elevated with systemic mastocytosis. Consider obtaining a skin biopsy if lesions are ambiguous or nondistinct.
There is no definitive cure for systemic or cutaneous mastocytosis, so treatment is directed at symptoms. Start by advising patients to avoid triggers and to refrain from scratching the affected areas. Topical antihistamines and oral nonsedating antihistamines can be helpful. If symptoms are more severe, refer the patient to an allergist/immunologist or to a hematologist for further medical management.2
The patient in this case had no systemic symptoms, so she was advised to continue taking oral loratadine 10 mg/d, which had been helpful, and to avoid rubbing her skin.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Murtaza Rizvi, MD, and Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

This patient was given a diagnosis of cutaneous mastocytosis. The condition was previously known as urticaria pigmentosa, but in 2016 the World Health Organization reclassified the disease to better suit its pathophysiology as a myeloid cell disorder.1
As the name suggests, this condition—which involves lesions in a sporadic, truncal distribution—involves overactivation of mastocytes at the tissue level from various stimuli, resulting in histocyte degranulation and hyperpigmentation. The exact cause is unknown. What is known is that it is often associated with other allergic or immunologic conditions and is thought to be related to mutations in the gene for CD-117’s receptor for tyrosine kinase.1 Incidence is similar to that of asthma, in that it occurs more often in younger patients; a majority of affected individuals will grow out of the disease by adolescence.1
Although most patients do not experience severe symptomatology, it is still important to differentiate cutaneous vs systemic mastocytosis. If a patient presents with inexplicable systemic symptoms of malaise, vague abdominal pain, heartburn, or flushing, the physician should consider systemic mastocytosis, idiopathic anaphylaxis, or hereditary alpha-tryptasemia.2
The test of choice is a serum tryptase test; levels will be elevated with systemic mastocytosis. Consider obtaining a skin biopsy if lesions are ambiguous or nondistinct.
There is no definitive cure for systemic or cutaneous mastocytosis, so treatment is directed at symptoms. Start by advising patients to avoid triggers and to refrain from scratching the affected areas. Topical antihistamines and oral nonsedating antihistamines can be helpful. If symptoms are more severe, refer the patient to an allergist/immunologist or to a hematologist for further medical management.2
The patient in this case had no systemic symptoms, so she was advised to continue taking oral loratadine 10 mg/d, which had been helpful, and to avoid rubbing her skin.
Image courtesy of Daniel Stulberg, MD. Text courtesy of Murtaza Rizvi, MD, and Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Arber DA, Orazi A, Hasserjian R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127:2391-2405. doi: 10.1182/blood-2016-03-643544.
2. Hartmann K, Escribano L, Grattan C, et al. Cutaneous manifestations in patients with mastocytosis: Consensus report of the European Competence Network on Mastocytosis; the American Academy of Allergy, Asthma & Immunology; and the European Academy of Allergology and Clinical Immunology. J Allergy Clin Immunol 2016; 137:35-45. doi: 10.1016/j.jaci.2015.08.034
1. Arber DA, Orazi A, Hasserjian R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127:2391-2405. doi: 10.1182/blood-2016-03-643544.
2. Hartmann K, Escribano L, Grattan C, et al. Cutaneous manifestations in patients with mastocytosis: Consensus report of the European Competence Network on Mastocytosis; the American Academy of Allergy, Asthma & Immunology; and the European Academy of Allergology and Clinical Immunology. J Allergy Clin Immunol 2016; 137:35-45. doi: 10.1016/j.jaci.2015.08.034
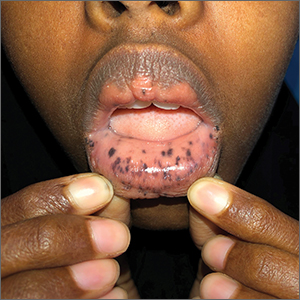
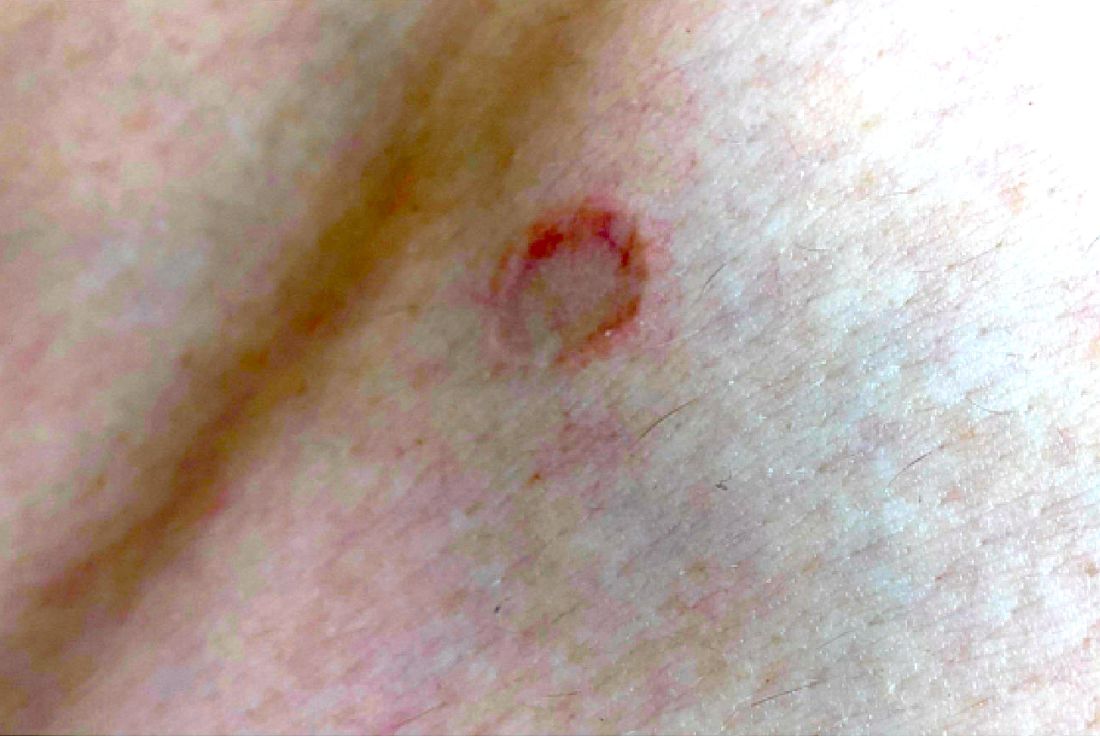
Macules and abdominal pain
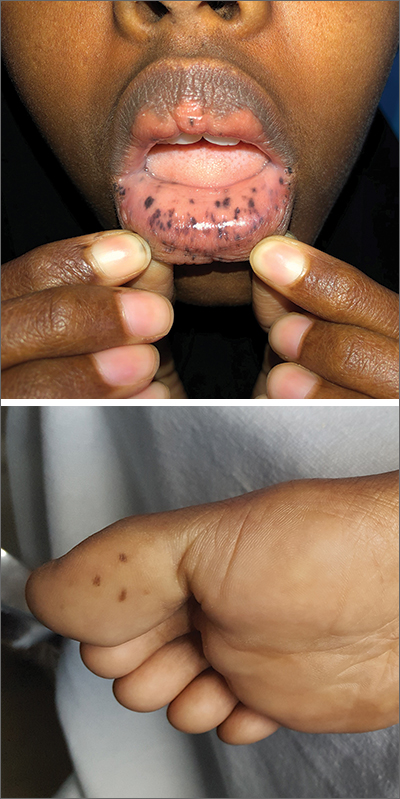
This patient was given a diagnosis of Peutz-Jeghers syndrome (PJS) based on the characteristic pigmented mucocutaneous macules and numerous polyps in her stomach and small bowel. PJS is an autosomal dominant syndrome characterized by mucocutaneous pigmentation, polyposis of the GI tract, and increased cancer risk. The prevalence is approximately 1 in 100,000.1 Genetic testing for the STK11 gene mutation, which is found in 70% of familial cases and 30% to 67% of sporadic cases, is not required for diagnosis.1
The bluish brown to black spots of PJS often are apparent at birth or in early infancy. They are most common on the lips, buccal mucosa, perioral region, palms, and soles.
The polyps may cause bleeding, anemia, and abdominal pain due to intussusception, obstruction, or infarction.2 Polyps usually are benign, but patients are at increased risk of GI and non-GI malignancies such as breast, pancreas, lung, and reproductive tract cancers.1
PJS can be differentiated from other causes of hyperpigmentation by clinical presentation and/or genetic testing. The diagnosis of PJS is made using the following criteria: (1) two or more histologically confirmed PJS polyps, (2) any number of PJS polyps and a family history of PJS, (3) characteristic mucocutaneous pigmentation and a family history of PJS, or (4) any number of PJS polyps and characteristic mucocutaneous pigmentation.2
It’s recommended that polyps be removed when technically feasible.3 Pigmented macules do not require treatment. Macules on the lips may disappear with time, while those on the buccal mucosa persist. The lip lesions can be lightened with chemical peels or laser.
This patient underwent laparotomy, which revealed a grossly dilated and gangrenous small bowel segment. Intussusception was not present and was thought to have spontaneously reduced. Resection and anastomosis of the affected small bowel was performed. The patient’s postoperative course was uneventful, and her diarrhea and abdominal pain resolved. A colonoscopy was normal, and the health care team explained that she would require follow-up and surveillance of her condition due to the high risk of future cancers.
This case was adapted from: Warsame MO, McMichael JR. Chronic abdominal pain and diarrhea. J Fam Pract. 2020;69:365,366,368.
Photos courtesy of Mohamed Omar Warsame, MBBS, and Josette R. McMichael, MD
1. Kopacova M, Tacheci I, Rejchrt S, et al. Peutz-Jeghers syndrome: diagnostic and therapeutic approach. World J Gastroenterol. 2009;15:5397-5408.
2. Beggs AD, Latchford AR, Vasen HF, et al. Peutz-Jeghers syndrome: a systematic review and recommendations for management. Gut. 2010;59:975-986.
3. van Lier MG, Mathus-Vliegen EM, Wagner A, et al. High cumulative risk of intussusception in patients with Peutz-Jeghers syndrome: time to update surveillance guidelines? Am J Gastroenterol. 2011;106:940-945.

This patient was given a diagnosis of Peutz-Jeghers syndrome (PJS) based on the characteristic pigmented mucocutaneous macules and numerous polyps in her stomach and small bowel. PJS is an autosomal dominant syndrome characterized by mucocutaneous pigmentation, polyposis of the GI tract, and increased cancer risk. The prevalence is approximately 1 in 100,000.1 Genetic testing for the STK11 gene mutation, which is found in 70% of familial cases and 30% to 67% of sporadic cases, is not required for diagnosis.1
The bluish brown to black spots of PJS often are apparent at birth or in early infancy. They are most common on the lips, buccal mucosa, perioral region, palms, and soles.
The polyps may cause bleeding, anemia, and abdominal pain due to intussusception, obstruction, or infarction.2 Polyps usually are benign, but patients are at increased risk of GI and non-GI malignancies such as breast, pancreas, lung, and reproductive tract cancers.1
PJS can be differentiated from other causes of hyperpigmentation by clinical presentation and/or genetic testing. The diagnosis of PJS is made using the following criteria: (1) two or more histologically confirmed PJS polyps, (2) any number of PJS polyps and a family history of PJS, (3) characteristic mucocutaneous pigmentation and a family history of PJS, or (4) any number of PJS polyps and characteristic mucocutaneous pigmentation.2
It’s recommended that polyps be removed when technically feasible.3 Pigmented macules do not require treatment. Macules on the lips may disappear with time, while those on the buccal mucosa persist. The lip lesions can be lightened with chemical peels or laser.
This patient underwent laparotomy, which revealed a grossly dilated and gangrenous small bowel segment. Intussusception was not present and was thought to have spontaneously reduced. Resection and anastomosis of the affected small bowel was performed. The patient’s postoperative course was uneventful, and her diarrhea and abdominal pain resolved. A colonoscopy was normal, and the health care team explained that she would require follow-up and surveillance of her condition due to the high risk of future cancers.
This case was adapted from: Warsame MO, McMichael JR. Chronic abdominal pain and diarrhea. J Fam Pract. 2020;69:365,366,368.
Photos courtesy of Mohamed Omar Warsame, MBBS, and Josette R. McMichael, MD

This patient was given a diagnosis of Peutz-Jeghers syndrome (PJS) based on the characteristic pigmented mucocutaneous macules and numerous polyps in her stomach and small bowel. PJS is an autosomal dominant syndrome characterized by mucocutaneous pigmentation, polyposis of the GI tract, and increased cancer risk. The prevalence is approximately 1 in 100,000.1 Genetic testing for the STK11 gene mutation, which is found in 70% of familial cases and 30% to 67% of sporadic cases, is not required for diagnosis.1
The bluish brown to black spots of PJS often are apparent at birth or in early infancy. They are most common on the lips, buccal mucosa, perioral region, palms, and soles.
The polyps may cause bleeding, anemia, and abdominal pain due to intussusception, obstruction, or infarction.2 Polyps usually are benign, but patients are at increased risk of GI and non-GI malignancies such as breast, pancreas, lung, and reproductive tract cancers.1
PJS can be differentiated from other causes of hyperpigmentation by clinical presentation and/or genetic testing. The diagnosis of PJS is made using the following criteria: (1) two or more histologically confirmed PJS polyps, (2) any number of PJS polyps and a family history of PJS, (3) characteristic mucocutaneous pigmentation and a family history of PJS, or (4) any number of PJS polyps and characteristic mucocutaneous pigmentation.2
It’s recommended that polyps be removed when technically feasible.3 Pigmented macules do not require treatment. Macules on the lips may disappear with time, while those on the buccal mucosa persist. The lip lesions can be lightened with chemical peels or laser.
This patient underwent laparotomy, which revealed a grossly dilated and gangrenous small bowel segment. Intussusception was not present and was thought to have spontaneously reduced. Resection and anastomosis of the affected small bowel was performed. The patient’s postoperative course was uneventful, and her diarrhea and abdominal pain resolved. A colonoscopy was normal, and the health care team explained that she would require follow-up and surveillance of her condition due to the high risk of future cancers.
This case was adapted from: Warsame MO, McMichael JR. Chronic abdominal pain and diarrhea. J Fam Pract. 2020;69:365,366,368.
Photos courtesy of Mohamed Omar Warsame, MBBS, and Josette R. McMichael, MD
1. Kopacova M, Tacheci I, Rejchrt S, et al. Peutz-Jeghers syndrome: diagnostic and therapeutic approach. World J Gastroenterol. 2009;15:5397-5408.
2. Beggs AD, Latchford AR, Vasen HF, et al. Peutz-Jeghers syndrome: a systematic review and recommendations for management. Gut. 2010;59:975-986.
3. van Lier MG, Mathus-Vliegen EM, Wagner A, et al. High cumulative risk of intussusception in patients with Peutz-Jeghers syndrome: time to update surveillance guidelines? Am J Gastroenterol. 2011;106:940-945.
1. Kopacova M, Tacheci I, Rejchrt S, et al. Peutz-Jeghers syndrome: diagnostic and therapeutic approach. World J Gastroenterol. 2009;15:5397-5408.
2. Beggs AD, Latchford AR, Vasen HF, et al. Peutz-Jeghers syndrome: a systematic review and recommendations for management. Gut. 2010;59:975-986.
3. van Lier MG, Mathus-Vliegen EM, Wagner A, et al. High cumulative risk of intussusception in patients with Peutz-Jeghers syndrome: time to update surveillance guidelines? Am J Gastroenterol. 2011;106:940-945.
FDA will review pediatric indication for roflumilast cream
, according to a press release from the manufacturer.
The company, Arcutis Biotherapeutics, announced the submission of a supplemental new drug application for approval of roflumilast cream (Zoryve), a topical phosphodiesterase-4 (PDE-4) inhibitor, to treat psoriasis in children aged 2-11 years. If approved, this would be the first such product for young children with plaque psoriasis, according to the press release. In July 2022, the FDA approved roflumilast cream 0.3% for the treatment of plaque psoriasis in people 12 years of age and older, including in intertriginous areas, based on data from the phase 3 DERMIS-1 and DERMIS-2 trials.
The new submission is supported by data from two 4-week Maximal Usage Systemic Exposure (MUSE) studies in children ages 2-11 years with plaque psoriasis. In these phase 2, open-label studies, one study of children aged 2-5 years and another study of children aged 6-11 years, participants were treated with roflumilast cream 0.3% once daily for 4 weeks. The MUSE studies are also intended to fulfill postmarketing requirements for roflumilast, according to the company. The MUSE results were consistent with those from DERMIS-1 and DERMIS-2, according to the company press release. In DERMIS-1 and DERMIS-2, significantly more patients randomized to roflumilast met criteria for Investigators Global Success (IGA) scores after 8 weeks of daily treatment compared with placebo patients, and significantly more achieved a 75% reduction in Psoriasis Area and Severity Index (PASI) scores compared with those on placebo.
Common adverse events associated with roflumilast include diarrhea, headache, insomnia, nausea, application site pain, upper respiratory tract infection, and urinary tract infection. None of these have been reported in more than 3% of patients, the press release noted.
, according to a press release from the manufacturer.
The company, Arcutis Biotherapeutics, announced the submission of a supplemental new drug application for approval of roflumilast cream (Zoryve), a topical phosphodiesterase-4 (PDE-4) inhibitor, to treat psoriasis in children aged 2-11 years. If approved, this would be the first such product for young children with plaque psoriasis, according to the press release. In July 2022, the FDA approved roflumilast cream 0.3% for the treatment of plaque psoriasis in people 12 years of age and older, including in intertriginous areas, based on data from the phase 3 DERMIS-1 and DERMIS-2 trials.
The new submission is supported by data from two 4-week Maximal Usage Systemic Exposure (MUSE) studies in children ages 2-11 years with plaque psoriasis. In these phase 2, open-label studies, one study of children aged 2-5 years and another study of children aged 6-11 years, participants were treated with roflumilast cream 0.3% once daily for 4 weeks. The MUSE studies are also intended to fulfill postmarketing requirements for roflumilast, according to the company. The MUSE results were consistent with those from DERMIS-1 and DERMIS-2, according to the company press release. In DERMIS-1 and DERMIS-2, significantly more patients randomized to roflumilast met criteria for Investigators Global Success (IGA) scores after 8 weeks of daily treatment compared with placebo patients, and significantly more achieved a 75% reduction in Psoriasis Area and Severity Index (PASI) scores compared with those on placebo.
Common adverse events associated with roflumilast include diarrhea, headache, insomnia, nausea, application site pain, upper respiratory tract infection, and urinary tract infection. None of these have been reported in more than 3% of patients, the press release noted.
, according to a press release from the manufacturer.
The company, Arcutis Biotherapeutics, announced the submission of a supplemental new drug application for approval of roflumilast cream (Zoryve), a topical phosphodiesterase-4 (PDE-4) inhibitor, to treat psoriasis in children aged 2-11 years. If approved, this would be the first such product for young children with plaque psoriasis, according to the press release. In July 2022, the FDA approved roflumilast cream 0.3% for the treatment of plaque psoriasis in people 12 years of age and older, including in intertriginous areas, based on data from the phase 3 DERMIS-1 and DERMIS-2 trials.
The new submission is supported by data from two 4-week Maximal Usage Systemic Exposure (MUSE) studies in children ages 2-11 years with plaque psoriasis. In these phase 2, open-label studies, one study of children aged 2-5 years and another study of children aged 6-11 years, participants were treated with roflumilast cream 0.3% once daily for 4 weeks. The MUSE studies are also intended to fulfill postmarketing requirements for roflumilast, according to the company. The MUSE results were consistent with those from DERMIS-1 and DERMIS-2, according to the company press release. In DERMIS-1 and DERMIS-2, significantly more patients randomized to roflumilast met criteria for Investigators Global Success (IGA) scores after 8 weeks of daily treatment compared with placebo patients, and significantly more achieved a 75% reduction in Psoriasis Area and Severity Index (PASI) scores compared with those on placebo.
Common adverse events associated with roflumilast include diarrhea, headache, insomnia, nausea, application site pain, upper respiratory tract infection, and urinary tract infection. None of these have been reported in more than 3% of patients, the press release noted.
FDA approves Idacio as eighth adalimumab biosimilar in U.S.
A biosimilar drug to the tumor necrosis factor inhibitor adalimumab, marketed as Idacio (adalimumab-aacf), has been approved by the Food and Drug Administration for use in the United States, according to a press release from manufacturer Fresenius Kabi.
Idacio is a citrate-free, low-concentration formulation of adalimumab and is now approved for use for all but three of the indications that currently apply to the reference adalimumab product (Humira): rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis in adults, ankylosing spondylitis, Crohn’s disease in adults and children aged 6 years or older, ulcerative colitis in adults, and plaque psoriasis in adults. It does not apply to Humira’s indications for hidradenitis suppurativa, uveitis, or ulcerative colitis in pediatric patients aged 5 years and older.
Idacio is the eighth adalimumab biosimilar to be approved in the United States. Its approval was based on evidence of a similar profile of pharmacokinetics, safety, efficacy, and immunogenicity to Humira.
Idacio was first launched in 2019 and has been marketed in more than 37 countries worldwide, according to Fresenius Kabi. The U.S. launch is scheduled for July, and Idacio will be available as a self-administered prefilled syringe or prefilled pen.
A version of this article first appeared on Medscape.com.
A biosimilar drug to the tumor necrosis factor inhibitor adalimumab, marketed as Idacio (adalimumab-aacf), has been approved by the Food and Drug Administration for use in the United States, according to a press release from manufacturer Fresenius Kabi.
Idacio is a citrate-free, low-concentration formulation of adalimumab and is now approved for use for all but three of the indications that currently apply to the reference adalimumab product (Humira): rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis in adults, ankylosing spondylitis, Crohn’s disease in adults and children aged 6 years or older, ulcerative colitis in adults, and plaque psoriasis in adults. It does not apply to Humira’s indications for hidradenitis suppurativa, uveitis, or ulcerative colitis in pediatric patients aged 5 years and older.
Idacio is the eighth adalimumab biosimilar to be approved in the United States. Its approval was based on evidence of a similar profile of pharmacokinetics, safety, efficacy, and immunogenicity to Humira.
Idacio was first launched in 2019 and has been marketed in more than 37 countries worldwide, according to Fresenius Kabi. The U.S. launch is scheduled for July, and Idacio will be available as a self-administered prefilled syringe or prefilled pen.
A version of this article first appeared on Medscape.com.
A biosimilar drug to the tumor necrosis factor inhibitor adalimumab, marketed as Idacio (adalimumab-aacf), has been approved by the Food and Drug Administration for use in the United States, according to a press release from manufacturer Fresenius Kabi.
Idacio is a citrate-free, low-concentration formulation of adalimumab and is now approved for use for all but three of the indications that currently apply to the reference adalimumab product (Humira): rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis in adults, ankylosing spondylitis, Crohn’s disease in adults and children aged 6 years or older, ulcerative colitis in adults, and plaque psoriasis in adults. It does not apply to Humira’s indications for hidradenitis suppurativa, uveitis, or ulcerative colitis in pediatric patients aged 5 years and older.
Idacio is the eighth adalimumab biosimilar to be approved in the United States. Its approval was based on evidence of a similar profile of pharmacokinetics, safety, efficacy, and immunogenicity to Humira.
Idacio was first launched in 2019 and has been marketed in more than 37 countries worldwide, according to Fresenius Kabi. The U.S. launch is scheduled for July, and Idacio will be available as a self-administered prefilled syringe or prefilled pen.
A version of this article first appeared on Medscape.com.
Dupilumab significantly improves markers of AD severity in pediatric patients
A registry-based study provides further evidence that treatment with dupilumab significantly reduces severity and symptoms of atopic dermatitis (AD) in clinical practice.
Dupilumab also decreased severity-associated biomarkers in pediatric patients with moderate to severe AD, researchers in the Netherlands reported.
Obtaining serum biomarkers is not the current standard in everyday practice, but studying them may improve understanding of who might respond best to dupilumab, said Jessica Hui, MD, a pediatric allergist and immunologist at National Jewish Health in Denver, in an email comment to this news organization.
“AD is heterogeneous, as each patient may have different presentations and underlying biology,” said Dr. Hui, who wasn’t involved in the research. “Studying biomarkers can eventually assist us in providing targeted therapy to each individual patient.”
Dr. Hui added, “As blood biomarkers can inform us of severity and treatment response, we can be hopeful that this will assist us in the management of AD patients in the future.”
Examining effect on disease severity
Dupilumab, a monoclonal antibody that inhibits interleukin (IL)-4 and IL-13 signaling, is approved in Europe and the United States to treat moderate to severe AD in patients 6 months of age or older, and to treat certain other inflammatory conditions.
Phase 3 studies show that dupilumab is effective for improving AD symptoms and quality of life in pediatric patients, but few clinical practice studies have researched the effect of the therapy on severity- and disease-related biomarkers in this population, the study authors write.
The study was published online in Pediatric Allergy Immunology.
In a new study, a team led by Esmé Kamphuis, MD, of the University of Groningen, the Netherlands, and colleagues evaluated the efficacy and safety of a 28-week dupilumab treatment course in 61 pediatric patients with moderate to severe AD. Additionally, the investigators examined the effect of this treatment regimen on serum biomarkers associated with disease severity.
Patients in the study were registered in the multicenter BioDay registry, which includes patients with moderate to severe AD receiving biologics or small-molecule agents. The AD cohort included children between 6 and 12 years of age (n = 16) and adolescents between 12 and less than 18 years of age (n = 45), all of whom received dupilumab on a dosing regimen indicated by age and body weight.
Over one-third (36.1%) of dupilumab-treated patients achieved an Investigator Global Assessment score of “almost clear” by 28 weeks of treatment. Approximately 75.4% of patients reached an Eczema Area and Severity Index (EASI) of 50, 49.2% reached EASI-75, and 24.6% reached EASI-90 at the 7-month follow-up.
Among patient-reported outcomes, 84.7% experienced improvements of 4 or more points on the Patient-Oriented Eczema Measure after the 28-week dupilumab treatment. In addition, improvements of 4 or more points on the Numeric Rating Scale for pruritus and pain were achieved by 45.3% and 77.4% of patients, respectively.
The most frequently reported side effects included conjunctivitis (n = 10) and headache (n = 4).
Of the 19 severity-associated serum biomarkers measured at baseline, week 4, and week 16, markers related to AD severity and treatment response significantly decreased during treatment (thymus- and activation-regulated chemokine, pulmonary and activation-regulated chemokine, periostin, soluble IL-2 receptor alpha).
A predicted EASI, calculated from selected biomarkers, demonstrated a significant association with disease severity in the cohort.
Implications for practice
When asked to comment on the study findings, Raegan Hunt, MD, the division chief of pediatric dermatology at Texas Children’s Hospital in Houston, said it is important to validate the changes in AD serum biomarkers in pediatric patients on dupilumab therapy, given that this treatment has historically been better studied in adults.
“This study adds to daily practice outcomes data, which in many cases is more relevant to the everyday care of patients than structured clinical trial data,” said Dr. Hunt, an associate professor at the Baylor College of Medicine, Houston.
Dr. Hunt, who didn’t participate in the study, noted that more research is needed on the adverse effects of dupilumab in the pediatric AD population.
Dr. Hui added that there is a lack of clear understanding of the exact underlying mechanisms for certain side effects, such as conjunctivitis, warranting further study.
The study’s BioDay registry is funded by Sanofi/Regeneron, AbbVie, Leo Pharma, Pfizer, and Eli Lilly. Several study coauthors report relationships with several pharmaceutical companies. Dr. Hunt and Dr. Hui report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A registry-based study provides further evidence that treatment with dupilumab significantly reduces severity and symptoms of atopic dermatitis (AD) in clinical practice.
Dupilumab also decreased severity-associated biomarkers in pediatric patients with moderate to severe AD, researchers in the Netherlands reported.
Obtaining serum biomarkers is not the current standard in everyday practice, but studying them may improve understanding of who might respond best to dupilumab, said Jessica Hui, MD, a pediatric allergist and immunologist at National Jewish Health in Denver, in an email comment to this news organization.
“AD is heterogeneous, as each patient may have different presentations and underlying biology,” said Dr. Hui, who wasn’t involved in the research. “Studying biomarkers can eventually assist us in providing targeted therapy to each individual patient.”
Dr. Hui added, “As blood biomarkers can inform us of severity and treatment response, we can be hopeful that this will assist us in the management of AD patients in the future.”
Examining effect on disease severity
Dupilumab, a monoclonal antibody that inhibits interleukin (IL)-4 and IL-13 signaling, is approved in Europe and the United States to treat moderate to severe AD in patients 6 months of age or older, and to treat certain other inflammatory conditions.
Phase 3 studies show that dupilumab is effective for improving AD symptoms and quality of life in pediatric patients, but few clinical practice studies have researched the effect of the therapy on severity- and disease-related biomarkers in this population, the study authors write.
The study was published online in Pediatric Allergy Immunology.
In a new study, a team led by Esmé Kamphuis, MD, of the University of Groningen, the Netherlands, and colleagues evaluated the efficacy and safety of a 28-week dupilumab treatment course in 61 pediatric patients with moderate to severe AD. Additionally, the investigators examined the effect of this treatment regimen on serum biomarkers associated with disease severity.
Patients in the study were registered in the multicenter BioDay registry, which includes patients with moderate to severe AD receiving biologics or small-molecule agents. The AD cohort included children between 6 and 12 years of age (n = 16) and adolescents between 12 and less than 18 years of age (n = 45), all of whom received dupilumab on a dosing regimen indicated by age and body weight.
Over one-third (36.1%) of dupilumab-treated patients achieved an Investigator Global Assessment score of “almost clear” by 28 weeks of treatment. Approximately 75.4% of patients reached an Eczema Area and Severity Index (EASI) of 50, 49.2% reached EASI-75, and 24.6% reached EASI-90 at the 7-month follow-up.
Among patient-reported outcomes, 84.7% experienced improvements of 4 or more points on the Patient-Oriented Eczema Measure after the 28-week dupilumab treatment. In addition, improvements of 4 or more points on the Numeric Rating Scale for pruritus and pain were achieved by 45.3% and 77.4% of patients, respectively.
The most frequently reported side effects included conjunctivitis (n = 10) and headache (n = 4).
Of the 19 severity-associated serum biomarkers measured at baseline, week 4, and week 16, markers related to AD severity and treatment response significantly decreased during treatment (thymus- and activation-regulated chemokine, pulmonary and activation-regulated chemokine, periostin, soluble IL-2 receptor alpha).
A predicted EASI, calculated from selected biomarkers, demonstrated a significant association with disease severity in the cohort.
Implications for practice
When asked to comment on the study findings, Raegan Hunt, MD, the division chief of pediatric dermatology at Texas Children’s Hospital in Houston, said it is important to validate the changes in AD serum biomarkers in pediatric patients on dupilumab therapy, given that this treatment has historically been better studied in adults.
“This study adds to daily practice outcomes data, which in many cases is more relevant to the everyday care of patients than structured clinical trial data,” said Dr. Hunt, an associate professor at the Baylor College of Medicine, Houston.
Dr. Hunt, who didn’t participate in the study, noted that more research is needed on the adverse effects of dupilumab in the pediatric AD population.
Dr. Hui added that there is a lack of clear understanding of the exact underlying mechanisms for certain side effects, such as conjunctivitis, warranting further study.
The study’s BioDay registry is funded by Sanofi/Regeneron, AbbVie, Leo Pharma, Pfizer, and Eli Lilly. Several study coauthors report relationships with several pharmaceutical companies. Dr. Hunt and Dr. Hui report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A registry-based study provides further evidence that treatment with dupilumab significantly reduces severity and symptoms of atopic dermatitis (AD) in clinical practice.
Dupilumab also decreased severity-associated biomarkers in pediatric patients with moderate to severe AD, researchers in the Netherlands reported.
Obtaining serum biomarkers is not the current standard in everyday practice, but studying them may improve understanding of who might respond best to dupilumab, said Jessica Hui, MD, a pediatric allergist and immunologist at National Jewish Health in Denver, in an email comment to this news organization.
“AD is heterogeneous, as each patient may have different presentations and underlying biology,” said Dr. Hui, who wasn’t involved in the research. “Studying biomarkers can eventually assist us in providing targeted therapy to each individual patient.”
Dr. Hui added, “As blood biomarkers can inform us of severity and treatment response, we can be hopeful that this will assist us in the management of AD patients in the future.”
Examining effect on disease severity
Dupilumab, a monoclonal antibody that inhibits interleukin (IL)-4 and IL-13 signaling, is approved in Europe and the United States to treat moderate to severe AD in patients 6 months of age or older, and to treat certain other inflammatory conditions.
Phase 3 studies show that dupilumab is effective for improving AD symptoms and quality of life in pediatric patients, but few clinical practice studies have researched the effect of the therapy on severity- and disease-related biomarkers in this population, the study authors write.
The study was published online in Pediatric Allergy Immunology.
In a new study, a team led by Esmé Kamphuis, MD, of the University of Groningen, the Netherlands, and colleagues evaluated the efficacy and safety of a 28-week dupilumab treatment course in 61 pediatric patients with moderate to severe AD. Additionally, the investigators examined the effect of this treatment regimen on serum biomarkers associated with disease severity.
Patients in the study were registered in the multicenter BioDay registry, which includes patients with moderate to severe AD receiving biologics or small-molecule agents. The AD cohort included children between 6 and 12 years of age (n = 16) and adolescents between 12 and less than 18 years of age (n = 45), all of whom received dupilumab on a dosing regimen indicated by age and body weight.
Over one-third (36.1%) of dupilumab-treated patients achieved an Investigator Global Assessment score of “almost clear” by 28 weeks of treatment. Approximately 75.4% of patients reached an Eczema Area and Severity Index (EASI) of 50, 49.2% reached EASI-75, and 24.6% reached EASI-90 at the 7-month follow-up.
Among patient-reported outcomes, 84.7% experienced improvements of 4 or more points on the Patient-Oriented Eczema Measure after the 28-week dupilumab treatment. In addition, improvements of 4 or more points on the Numeric Rating Scale for pruritus and pain were achieved by 45.3% and 77.4% of patients, respectively.
The most frequently reported side effects included conjunctivitis (n = 10) and headache (n = 4).
Of the 19 severity-associated serum biomarkers measured at baseline, week 4, and week 16, markers related to AD severity and treatment response significantly decreased during treatment (thymus- and activation-regulated chemokine, pulmonary and activation-regulated chemokine, periostin, soluble IL-2 receptor alpha).
A predicted EASI, calculated from selected biomarkers, demonstrated a significant association with disease severity in the cohort.
Implications for practice
When asked to comment on the study findings, Raegan Hunt, MD, the division chief of pediatric dermatology at Texas Children’s Hospital in Houston, said it is important to validate the changes in AD serum biomarkers in pediatric patients on dupilumab therapy, given that this treatment has historically been better studied in adults.
“This study adds to daily practice outcomes data, which in many cases is more relevant to the everyday care of patients than structured clinical trial data,” said Dr. Hunt, an associate professor at the Baylor College of Medicine, Houston.
Dr. Hunt, who didn’t participate in the study, noted that more research is needed on the adverse effects of dupilumab in the pediatric AD population.
Dr. Hui added that there is a lack of clear understanding of the exact underlying mechanisms for certain side effects, such as conjunctivitis, warranting further study.
The study’s BioDay registry is funded by Sanofi/Regeneron, AbbVie, Leo Pharma, Pfizer, and Eli Lilly. Several study coauthors report relationships with several pharmaceutical companies. Dr. Hunt and Dr. Hui report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM PEDIATRIC ALLERGY IMMUNOLOGY
Ustekinumab matches TNF inhibitors for psoriatic arthritis in 3-year, real-world study
The interleukin-12/23 inhibitor ustekinumab (Stelara) is nearly as effective as a tumor necrosis factor (TNF) inhibitor for psoriatic arthritis, and patients are slightly more likely to persist with it and have a lower rate of adverse events, a 3-year, real-world study has found.
In a paper published online in Annals of the Rheumatic Diseases, researchers presented the outcomes of the prospective, observational PsABio study of 895 adults with psoriatic arthritis, who were starting treatment for the first time with either ustekinumab or a TNF inhibitor as first-, second-, or third-line treatment.
At 3 years after starting therapy, 49.9% of the 439 patients prescribed ustekinumab were still on that treatment, compared with 47.8% of the 456 patients prescribed a TNF inhibitor. However, there were differences in persistence based on clinical presentation. Patients who had severe skin involvement who were treated with ustekinumab stayed on the drug for longer than did those with severe skin involvement treated with a TNF inhibitor, and they were more likely to persist with their treatment for the 3 years of the study. However, there were numerically more patients with mild or moderate skin involvement taking a TNF inhibitor who stayed persistent with the treatment, compared with those taking ustekinumab, although the differences were not statistically significant.
“In the ustekinumab group, skin response was an important reason for prolonged persistence, with more patients in the ustekinumab group stopping/switching due to lack of effectiveness,” wrote Laure Gossec, MD, of Pitié-Salpêtrière Hospital and Sorbonne University, Paris, and coauthors. “This is expected, as psoriasis can significantly affect morbidity, and successfully treating skin symptoms improves patients’ health-related quality of life.”
The authors also noted that patients on ustekinumab monotherapy had the highest rate of persistence and stayed on treatment longer than did those on TNF inhibitor monotherapy, or on dual therapy with either drug combined with methotrexate. They suggested this could be because patients on TNF inhibitor monotherapy may be more likely to develop antidrug antibodies than those on ustekinumab monotherapy. It could also be because adding methotrexate may increase the risk of adverse events, but without necessarily increasing the effectiveness of ustekinumab on skin involvement.
In terms of efficacy, researchers saw that 69.8% of patients in the TNF inhibitor group had achieved low disease activity and 45% had achieved remission, compared with 58.6% of patients in the ustekinumab group who achieved low disease activity and 31.4% who achieved remission.
A similar pattern was seen for minimal disease activity and very low disease activity, which were achieved by 54.2% and 26.9% respectively of those in the TNF inhibitor group, and 41.4% and 19.2% respectively of those in the ustekinumab group.
Because the study was observational and real-world, the choice of therapy was made by the treating rheumatologist rather than patients being randomized. There were some baseline differences between the ustekinumab and TNF inhibitor groups; for example, patients in ustekinumab group were generally older and with more comorbidities, and were more likely to have previous been treated with biologics. However, they were also less likely to be concurrently treated with methotrexate and NSAIDs, and more likely to have severe skin involvement.
The study saw a higher rate of adverse events in the TNF inhibitor group, compared with the ustekinumab, with 39.7% of patients treated with TNF inhibitor and 34.6% of those treated with ustekinumab reporting at least one adverse event. The rates of serious adverse events and malignancies were similar for the two groups, but overall the ustekinumab group had a lower rate of clinically-relevant adverse events including infections.
The study was sponsored by Janssen, which markets ustekinumab. Ten authors declared personal fees, grants, and nonfinancial support from the pharmaceutical sector, including Janssen. One author was an employee of Janssen, one an employee of Johnson & Johnson, and two are editorial board members of Annals of the Rheumatic Diseases.
The interleukin-12/23 inhibitor ustekinumab (Stelara) is nearly as effective as a tumor necrosis factor (TNF) inhibitor for psoriatic arthritis, and patients are slightly more likely to persist with it and have a lower rate of adverse events, a 3-year, real-world study has found.
In a paper published online in Annals of the Rheumatic Diseases, researchers presented the outcomes of the prospective, observational PsABio study of 895 adults with psoriatic arthritis, who were starting treatment for the first time with either ustekinumab or a TNF inhibitor as first-, second-, or third-line treatment.
At 3 years after starting therapy, 49.9% of the 439 patients prescribed ustekinumab were still on that treatment, compared with 47.8% of the 456 patients prescribed a TNF inhibitor. However, there were differences in persistence based on clinical presentation. Patients who had severe skin involvement who were treated with ustekinumab stayed on the drug for longer than did those with severe skin involvement treated with a TNF inhibitor, and they were more likely to persist with their treatment for the 3 years of the study. However, there were numerically more patients with mild or moderate skin involvement taking a TNF inhibitor who stayed persistent with the treatment, compared with those taking ustekinumab, although the differences were not statistically significant.
“In the ustekinumab group, skin response was an important reason for prolonged persistence, with more patients in the ustekinumab group stopping/switching due to lack of effectiveness,” wrote Laure Gossec, MD, of Pitié-Salpêtrière Hospital and Sorbonne University, Paris, and coauthors. “This is expected, as psoriasis can significantly affect morbidity, and successfully treating skin symptoms improves patients’ health-related quality of life.”
The authors also noted that patients on ustekinumab monotherapy had the highest rate of persistence and stayed on treatment longer than did those on TNF inhibitor monotherapy, or on dual therapy with either drug combined with methotrexate. They suggested this could be because patients on TNF inhibitor monotherapy may be more likely to develop antidrug antibodies than those on ustekinumab monotherapy. It could also be because adding methotrexate may increase the risk of adverse events, but without necessarily increasing the effectiveness of ustekinumab on skin involvement.
In terms of efficacy, researchers saw that 69.8% of patients in the TNF inhibitor group had achieved low disease activity and 45% had achieved remission, compared with 58.6% of patients in the ustekinumab group who achieved low disease activity and 31.4% who achieved remission.
A similar pattern was seen for minimal disease activity and very low disease activity, which were achieved by 54.2% and 26.9% respectively of those in the TNF inhibitor group, and 41.4% and 19.2% respectively of those in the ustekinumab group.
Because the study was observational and real-world, the choice of therapy was made by the treating rheumatologist rather than patients being randomized. There were some baseline differences between the ustekinumab and TNF inhibitor groups; for example, patients in ustekinumab group were generally older and with more comorbidities, and were more likely to have previous been treated with biologics. However, they were also less likely to be concurrently treated with methotrexate and NSAIDs, and more likely to have severe skin involvement.
The study saw a higher rate of adverse events in the TNF inhibitor group, compared with the ustekinumab, with 39.7% of patients treated with TNF inhibitor and 34.6% of those treated with ustekinumab reporting at least one adverse event. The rates of serious adverse events and malignancies were similar for the two groups, but overall the ustekinumab group had a lower rate of clinically-relevant adverse events including infections.
The study was sponsored by Janssen, which markets ustekinumab. Ten authors declared personal fees, grants, and nonfinancial support from the pharmaceutical sector, including Janssen. One author was an employee of Janssen, one an employee of Johnson & Johnson, and two are editorial board members of Annals of the Rheumatic Diseases.
The interleukin-12/23 inhibitor ustekinumab (Stelara) is nearly as effective as a tumor necrosis factor (TNF) inhibitor for psoriatic arthritis, and patients are slightly more likely to persist with it and have a lower rate of adverse events, a 3-year, real-world study has found.
In a paper published online in Annals of the Rheumatic Diseases, researchers presented the outcomes of the prospective, observational PsABio study of 895 adults with psoriatic arthritis, who were starting treatment for the first time with either ustekinumab or a TNF inhibitor as first-, second-, or third-line treatment.
At 3 years after starting therapy, 49.9% of the 439 patients prescribed ustekinumab were still on that treatment, compared with 47.8% of the 456 patients prescribed a TNF inhibitor. However, there were differences in persistence based on clinical presentation. Patients who had severe skin involvement who were treated with ustekinumab stayed on the drug for longer than did those with severe skin involvement treated with a TNF inhibitor, and they were more likely to persist with their treatment for the 3 years of the study. However, there were numerically more patients with mild or moderate skin involvement taking a TNF inhibitor who stayed persistent with the treatment, compared with those taking ustekinumab, although the differences were not statistically significant.
“In the ustekinumab group, skin response was an important reason for prolonged persistence, with more patients in the ustekinumab group stopping/switching due to lack of effectiveness,” wrote Laure Gossec, MD, of Pitié-Salpêtrière Hospital and Sorbonne University, Paris, and coauthors. “This is expected, as psoriasis can significantly affect morbidity, and successfully treating skin symptoms improves patients’ health-related quality of life.”
The authors also noted that patients on ustekinumab monotherapy had the highest rate of persistence and stayed on treatment longer than did those on TNF inhibitor monotherapy, or on dual therapy with either drug combined with methotrexate. They suggested this could be because patients on TNF inhibitor monotherapy may be more likely to develop antidrug antibodies than those on ustekinumab monotherapy. It could also be because adding methotrexate may increase the risk of adverse events, but without necessarily increasing the effectiveness of ustekinumab on skin involvement.
In terms of efficacy, researchers saw that 69.8% of patients in the TNF inhibitor group had achieved low disease activity and 45% had achieved remission, compared with 58.6% of patients in the ustekinumab group who achieved low disease activity and 31.4% who achieved remission.
A similar pattern was seen for minimal disease activity and very low disease activity, which were achieved by 54.2% and 26.9% respectively of those in the TNF inhibitor group, and 41.4% and 19.2% respectively of those in the ustekinumab group.
Because the study was observational and real-world, the choice of therapy was made by the treating rheumatologist rather than patients being randomized. There were some baseline differences between the ustekinumab and TNF inhibitor groups; for example, patients in ustekinumab group were generally older and with more comorbidities, and were more likely to have previous been treated with biologics. However, they were also less likely to be concurrently treated with methotrexate and NSAIDs, and more likely to have severe skin involvement.
The study saw a higher rate of adverse events in the TNF inhibitor group, compared with the ustekinumab, with 39.7% of patients treated with TNF inhibitor and 34.6% of those treated with ustekinumab reporting at least one adverse event. The rates of serious adverse events and malignancies were similar for the two groups, but overall the ustekinumab group had a lower rate of clinically-relevant adverse events including infections.
The study was sponsored by Janssen, which markets ustekinumab. Ten authors declared personal fees, grants, and nonfinancial support from the pharmaceutical sector, including Janssen. One author was an employee of Janssen, one an employee of Johnson & Johnson, and two are editorial board members of Annals of the Rheumatic Diseases.
FROM ANNALS OF THE RHEUMATIC DISEASES
A 17-year-old male was referred by his pediatrician for evaluation of a year-long rash
A biopsy of the edge of one of lesions on the torso was performed. Histopathology demonstrated hyperkeratosis of the stratum corneum with focal thickening of the granular cell layer, basal layer degeneration of the epidermis, and a band-like subepidermal lymphocytic infiltrate with Civatte bodies consistent with lichen planus. There was some reduction in the elastic fibers on the papillary dermis.
Given the morphology of the lesions and the histopathologic presentation, he was diagnosed with annular atrophic lichen planus (AALP). Lichen planus is a chronic inflammatory condition that can affect the skin, nails, hair, and mucosa. Lichen planus is seen in less than 1% of the population, occurring mainly in middle-aged adults and rarely seen in children. Though, there appears to be no clear racial predilection, a small study in the United States showed a higher incidence of lichen planus in Black children. Lesions with classic characteristics are pruritic, polygonal, violaceous, flat-topped papules and plaques.
There are different subtypes of lichen planus, which include papular or classic form, hypertrophic, vesiculobullous, actinic, annular, atrophic, annular atrophic, linear, follicular, lichen planus pigmentosus, lichen pigmentosa pigmentosus-inversus, lichen planus–lupus erythematosus overlap syndrome, and lichen planus pemphigoides. The annular atrophic form is the least common of all, and there are few reports in the pediatric population. AALP presents as annular papules and plaques with atrophic centers that resolve within a few months leaving postinflammatory hypo- or hyperpigmentation and, in some patients, permanent atrophic scarring.
In histopathology, the lesions show the classic characteristics of lichen planus including vacuolar interface changes and necrotic keratinocytes, hypergranulosis, band-like infiltrate in the dermis, melanin incontinence, and Civatte bodies. In AALP, the center of the lesion shows an atrophic epidermis, and there is also a characteristic partial reduction to complete destruction of elastic fibers in the papillary dermis in the center of the lesion and sometimes in the periphery as well, which helps differentiate AALP from other forms of lichen planus.
The differential diagnosis for AALP includes tinea corporis, which can present with annular lesions, but they are usually scaly and rarely resolve on their own. Pityriasis rosea lesions can also look very similar to AALP lesions, but the difference is the presence of an inner collaret of scale and a lack of atrophy in pityriasis rosea. Pityriasis rosea is a rash that can be triggered by viral infections, medications, and vaccines and self-resolves within 10-12 weeks. Secondary syphilis can also be annular and resemble lesions of AALP. Syphilis patients are usually sexually active and may have lesions present on the palms and soles, which were not seen in our patient.
Granuloma annulare should also be included in the differential diagnosis of AALP. Granuloma annulare lesions present as annular papules or plaques with raised borders and a slightly hyperpigmented center that may appear more depressed compared to the edges of the lesion, though not atrophic as seen in AALP. Pityriasis lichenoides chronica is an inflammatory condition of the skin in which patients present with erythematous to brown papules in different stages which may have a mica-like scale, usually not seen on AALP. Sometimes a skin biopsy will be needed to differentiate between these conditions.
It is very important to make a timely diagnosis of AALP and treat the lesions early as it may leave long-lasting dyspigmentation and scarring. Though AAPL lesions can be resistant to treatment with topical medications, there are reports of improvement with superpotent topical corticosteroids and calcineurin inhibitors. In recalcitrant cases, systemic therapy with isotretinoin, acitretin, methotrexate, systemic corticosteroids, dapsone, and hydroxychloroquine can be considered. Our patient was treated with clobetasol propionate ointment 0.05% with good response.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Bowers S and Warshaw EM. J Am Acad Dermatol. 2006 Oct;55(4):557-72; quiz 573-6.
Gorouhi F et al. Scientific World Journal. 2014 Jan 30;2014:742826.
Santhosh P and George M. Int J Dermatol. 2022.61:1213-7.
Sears S et al. Pediatr Dermatol. 2021;38:1283-7.
Weston G and Payette M. Int J Womens Dermatol. 2015 Sep 16;1(3):140-9.
A biopsy of the edge of one of lesions on the torso was performed. Histopathology demonstrated hyperkeratosis of the stratum corneum with focal thickening of the granular cell layer, basal layer degeneration of the epidermis, and a band-like subepidermal lymphocytic infiltrate with Civatte bodies consistent with lichen planus. There was some reduction in the elastic fibers on the papillary dermis.
Given the morphology of the lesions and the histopathologic presentation, he was diagnosed with annular atrophic lichen planus (AALP). Lichen planus is a chronic inflammatory condition that can affect the skin, nails, hair, and mucosa. Lichen planus is seen in less than 1% of the population, occurring mainly in middle-aged adults and rarely seen in children. Though, there appears to be no clear racial predilection, a small study in the United States showed a higher incidence of lichen planus in Black children. Lesions with classic characteristics are pruritic, polygonal, violaceous, flat-topped papules and plaques.
There are different subtypes of lichen planus, which include papular or classic form, hypertrophic, vesiculobullous, actinic, annular, atrophic, annular atrophic, linear, follicular, lichen planus pigmentosus, lichen pigmentosa pigmentosus-inversus, lichen planus–lupus erythematosus overlap syndrome, and lichen planus pemphigoides. The annular atrophic form is the least common of all, and there are few reports in the pediatric population. AALP presents as annular papules and plaques with atrophic centers that resolve within a few months leaving postinflammatory hypo- or hyperpigmentation and, in some patients, permanent atrophic scarring.
In histopathology, the lesions show the classic characteristics of lichen planus including vacuolar interface changes and necrotic keratinocytes, hypergranulosis, band-like infiltrate in the dermis, melanin incontinence, and Civatte bodies. In AALP, the center of the lesion shows an atrophic epidermis, and there is also a characteristic partial reduction to complete destruction of elastic fibers in the papillary dermis in the center of the lesion and sometimes in the periphery as well, which helps differentiate AALP from other forms of lichen planus.
The differential diagnosis for AALP includes tinea corporis, which can present with annular lesions, but they are usually scaly and rarely resolve on their own. Pityriasis rosea lesions can also look very similar to AALP lesions, but the difference is the presence of an inner collaret of scale and a lack of atrophy in pityriasis rosea. Pityriasis rosea is a rash that can be triggered by viral infections, medications, and vaccines and self-resolves within 10-12 weeks. Secondary syphilis can also be annular and resemble lesions of AALP. Syphilis patients are usually sexually active and may have lesions present on the palms and soles, which were not seen in our patient.
Granuloma annulare should also be included in the differential diagnosis of AALP. Granuloma annulare lesions present as annular papules or plaques with raised borders and a slightly hyperpigmented center that may appear more depressed compared to the edges of the lesion, though not atrophic as seen in AALP. Pityriasis lichenoides chronica is an inflammatory condition of the skin in which patients present with erythematous to brown papules in different stages which may have a mica-like scale, usually not seen on AALP. Sometimes a skin biopsy will be needed to differentiate between these conditions.
It is very important to make a timely diagnosis of AALP and treat the lesions early as it may leave long-lasting dyspigmentation and scarring. Though AAPL lesions can be resistant to treatment with topical medications, there are reports of improvement with superpotent topical corticosteroids and calcineurin inhibitors. In recalcitrant cases, systemic therapy with isotretinoin, acitretin, methotrexate, systemic corticosteroids, dapsone, and hydroxychloroquine can be considered. Our patient was treated with clobetasol propionate ointment 0.05% with good response.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Bowers S and Warshaw EM. J Am Acad Dermatol. 2006 Oct;55(4):557-72; quiz 573-6.
Gorouhi F et al. Scientific World Journal. 2014 Jan 30;2014:742826.
Santhosh P and George M. Int J Dermatol. 2022.61:1213-7.
Sears S et al. Pediatr Dermatol. 2021;38:1283-7.
Weston G and Payette M. Int J Womens Dermatol. 2015 Sep 16;1(3):140-9.
A biopsy of the edge of one of lesions on the torso was performed. Histopathology demonstrated hyperkeratosis of the stratum corneum with focal thickening of the granular cell layer, basal layer degeneration of the epidermis, and a band-like subepidermal lymphocytic infiltrate with Civatte bodies consistent with lichen planus. There was some reduction in the elastic fibers on the papillary dermis.
Given the morphology of the lesions and the histopathologic presentation, he was diagnosed with annular atrophic lichen planus (AALP). Lichen planus is a chronic inflammatory condition that can affect the skin, nails, hair, and mucosa. Lichen planus is seen in less than 1% of the population, occurring mainly in middle-aged adults and rarely seen in children. Though, there appears to be no clear racial predilection, a small study in the United States showed a higher incidence of lichen planus in Black children. Lesions with classic characteristics are pruritic, polygonal, violaceous, flat-topped papules and plaques.
There are different subtypes of lichen planus, which include papular or classic form, hypertrophic, vesiculobullous, actinic, annular, atrophic, annular atrophic, linear, follicular, lichen planus pigmentosus, lichen pigmentosa pigmentosus-inversus, lichen planus–lupus erythematosus overlap syndrome, and lichen planus pemphigoides. The annular atrophic form is the least common of all, and there are few reports in the pediatric population. AALP presents as annular papules and plaques with atrophic centers that resolve within a few months leaving postinflammatory hypo- or hyperpigmentation and, in some patients, permanent atrophic scarring.
In histopathology, the lesions show the classic characteristics of lichen planus including vacuolar interface changes and necrotic keratinocytes, hypergranulosis, band-like infiltrate in the dermis, melanin incontinence, and Civatte bodies. In AALP, the center of the lesion shows an atrophic epidermis, and there is also a characteristic partial reduction to complete destruction of elastic fibers in the papillary dermis in the center of the lesion and sometimes in the periphery as well, which helps differentiate AALP from other forms of lichen planus.
The differential diagnosis for AALP includes tinea corporis, which can present with annular lesions, but they are usually scaly and rarely resolve on their own. Pityriasis rosea lesions can also look very similar to AALP lesions, but the difference is the presence of an inner collaret of scale and a lack of atrophy in pityriasis rosea. Pityriasis rosea is a rash that can be triggered by viral infections, medications, and vaccines and self-resolves within 10-12 weeks. Secondary syphilis can also be annular and resemble lesions of AALP. Syphilis patients are usually sexually active and may have lesions present on the palms and soles, which were not seen in our patient.
Granuloma annulare should also be included in the differential diagnosis of AALP. Granuloma annulare lesions present as annular papules or plaques with raised borders and a slightly hyperpigmented center that may appear more depressed compared to the edges of the lesion, though not atrophic as seen in AALP. Pityriasis lichenoides chronica is an inflammatory condition of the skin in which patients present with erythematous to brown papules in different stages which may have a mica-like scale, usually not seen on AALP. Sometimes a skin biopsy will be needed to differentiate between these conditions.
It is very important to make a timely diagnosis of AALP and treat the lesions early as it may leave long-lasting dyspigmentation and scarring. Though AAPL lesions can be resistant to treatment with topical medications, there are reports of improvement with superpotent topical corticosteroids and calcineurin inhibitors. In recalcitrant cases, systemic therapy with isotretinoin, acitretin, methotrexate, systemic corticosteroids, dapsone, and hydroxychloroquine can be considered. Our patient was treated with clobetasol propionate ointment 0.05% with good response.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
Bowers S and Warshaw EM. J Am Acad Dermatol. 2006 Oct;55(4):557-72; quiz 573-6.
Gorouhi F et al. Scientific World Journal. 2014 Jan 30;2014:742826.
Santhosh P and George M. Int J Dermatol. 2022.61:1213-7.
Sears S et al. Pediatr Dermatol. 2021;38:1283-7.
Weston G and Payette M. Int J Womens Dermatol. 2015 Sep 16;1(3):140-9.
A 17-year-old healthy male was referred by his pediatrician for evaluation of a rash on the skin which has been present on and off for a year. During the initial presentation, the lesions were clustered on the back, were slightly itchy, and resolved after 3 months. Several new lesions have developed on the neck, torso, and extremities, leaving hypopigmented marks on the skin. He has previously been treated with topical antifungal creams, oral fluconazole, and triamcinolone ointment without resolution of the lesions.
He is not involved in any contact sports, he has not traveled outside the country, and is not taking any other medications. He is not sexually active. He also has a diagnosis of mild acne that he is currently treating with over-the-counter medications.
On physical exam he had several annular plaques with central atrophic centers and no scale. He also had some hypo- and hyperpigmented macules at the sites of prior skin lesions
Teen with hyperpigmented skin lesions
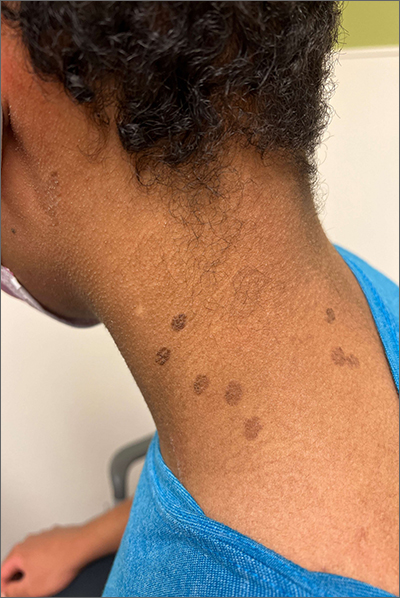
These hyperpigmented lesions with fine scale on the outer edges are characteristic of pityriasis versicolor, also known as tinea versicolor. These macules (or patches) are commonly found on the trunk, proximal extremities, or neck. The lesions can be hypopigmented (pityriasis versicolor alba), hyperpigmented (pityriasis versicolor nigra), or erythematous (pityriasis versicolor rubra).1 It is common to see hyperpigmentation in people with darker skin tones, as was the case with this patient. Pityriasis versicolor is often asymptomatic, but patients may describe mild to moderate pruritis.
Pityriasis versicolor is a common fungal infection of the superficial layers of the dermis caused by Malassezia furfur.2 Impaired immunity and excessive sweating are risk factors for pityriasis versicolor; other risk factors include having a family member with tinea versicolor and living in a hot, humid region.1 Adolescents and young adults are most often affected.1
An evoked scale sign is a helpful tool to confirm the diagnosis clinically.3 Scale will appear when you stretch the affected skin with your thumb and index finger (or you scrape the area with a scalpel blade).3 If there is doubt about the diagnosis, use a potassium hydroxide (KOH) preparation; a positive test will reveal the classic “spaghetti and meatballs” pattern.1
Selenium sulfide 2.5%, zinc pyrithione 1%, and ketoconazole 2% shampoo are effective topical treatments. The shampoo is applied full strength to the affected skin daily for 5 to 10 minutes before it’s washed off. This can be done for 1 to 4 weeks, with longer treatment courses resulting in higher cure rates. Systemic therapy is reserved for patients with widespread infection or those who do not respond to topical treatment.4 It’s important to advise patients that the restoration of normal skin pigmentation can takes months.
This patient was treated with ketoconazole 2% shampoo for 3 weeks. A complete return of skin color was achieved 6 months after completion of therapy.
Image courtesy of Judy Jasser, MD. Text courtesy of Judy Jasser, MD, Department of Pediatrics, and Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Crespo-Erchiga, V, Florencio, VD. Malassezia yeasts and pityriasis versicolor. Curr Opin Infect Dis. 2006;19:139-147. doi: 10.1097/01.qco.0000216624.21069.61
2. Gupta, AK, Bluhm R, Summerbell R. Pityriasis versicolor. J Eur Acad Dermatol Venereol. 2002;16:19-33. doi: 10.1046/j.1468-3083.2002.00378.x
3. Han, A, Calcara DA, Stoecker WV, et al. Evoked scale sign of tinea versicolor. Arch Dermatol. 2009;145:1078. doi: 10.1001/archdermatol.2009.203
4. Renati S, Cukras A, Bigby M. Pityriasis versicolor. BMJ. 2015;350:h1394. doi: 10.1136/bmj.h1394

These hyperpigmented lesions with fine scale on the outer edges are characteristic of pityriasis versicolor, also known as tinea versicolor. These macules (or patches) are commonly found on the trunk, proximal extremities, or neck. The lesions can be hypopigmented (pityriasis versicolor alba), hyperpigmented (pityriasis versicolor nigra), or erythematous (pityriasis versicolor rubra).1 It is common to see hyperpigmentation in people with darker skin tones, as was the case with this patient. Pityriasis versicolor is often asymptomatic, but patients may describe mild to moderate pruritis.
Pityriasis versicolor is a common fungal infection of the superficial layers of the dermis caused by Malassezia furfur.2 Impaired immunity and excessive sweating are risk factors for pityriasis versicolor; other risk factors include having a family member with tinea versicolor and living in a hot, humid region.1 Adolescents and young adults are most often affected.1
An evoked scale sign is a helpful tool to confirm the diagnosis clinically.3 Scale will appear when you stretch the affected skin with your thumb and index finger (or you scrape the area with a scalpel blade).3 If there is doubt about the diagnosis, use a potassium hydroxide (KOH) preparation; a positive test will reveal the classic “spaghetti and meatballs” pattern.1
Selenium sulfide 2.5%, zinc pyrithione 1%, and ketoconazole 2% shampoo are effective topical treatments. The shampoo is applied full strength to the affected skin daily for 5 to 10 minutes before it’s washed off. This can be done for 1 to 4 weeks, with longer treatment courses resulting in higher cure rates. Systemic therapy is reserved for patients with widespread infection or those who do not respond to topical treatment.4 It’s important to advise patients that the restoration of normal skin pigmentation can takes months.
This patient was treated with ketoconazole 2% shampoo for 3 weeks. A complete return of skin color was achieved 6 months after completion of therapy.
Image courtesy of Judy Jasser, MD. Text courtesy of Judy Jasser, MD, Department of Pediatrics, and Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

These hyperpigmented lesions with fine scale on the outer edges are characteristic of pityriasis versicolor, also known as tinea versicolor. These macules (or patches) are commonly found on the trunk, proximal extremities, or neck. The lesions can be hypopigmented (pityriasis versicolor alba), hyperpigmented (pityriasis versicolor nigra), or erythematous (pityriasis versicolor rubra).1 It is common to see hyperpigmentation in people with darker skin tones, as was the case with this patient. Pityriasis versicolor is often asymptomatic, but patients may describe mild to moderate pruritis.
Pityriasis versicolor is a common fungal infection of the superficial layers of the dermis caused by Malassezia furfur.2 Impaired immunity and excessive sweating are risk factors for pityriasis versicolor; other risk factors include having a family member with tinea versicolor and living in a hot, humid region.1 Adolescents and young adults are most often affected.1
An evoked scale sign is a helpful tool to confirm the diagnosis clinically.3 Scale will appear when you stretch the affected skin with your thumb and index finger (or you scrape the area with a scalpel blade).3 If there is doubt about the diagnosis, use a potassium hydroxide (KOH) preparation; a positive test will reveal the classic “spaghetti and meatballs” pattern.1
Selenium sulfide 2.5%, zinc pyrithione 1%, and ketoconazole 2% shampoo are effective topical treatments. The shampoo is applied full strength to the affected skin daily for 5 to 10 minutes before it’s washed off. This can be done for 1 to 4 weeks, with longer treatment courses resulting in higher cure rates. Systemic therapy is reserved for patients with widespread infection or those who do not respond to topical treatment.4 It’s important to advise patients that the restoration of normal skin pigmentation can takes months.
This patient was treated with ketoconazole 2% shampoo for 3 weeks. A complete return of skin color was achieved 6 months after completion of therapy.
Image courtesy of Judy Jasser, MD. Text courtesy of Judy Jasser, MD, Department of Pediatrics, and Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Crespo-Erchiga, V, Florencio, VD. Malassezia yeasts and pityriasis versicolor. Curr Opin Infect Dis. 2006;19:139-147. doi: 10.1097/01.qco.0000216624.21069.61
2. Gupta, AK, Bluhm R, Summerbell R. Pityriasis versicolor. J Eur Acad Dermatol Venereol. 2002;16:19-33. doi: 10.1046/j.1468-3083.2002.00378.x
3. Han, A, Calcara DA, Stoecker WV, et al. Evoked scale sign of tinea versicolor. Arch Dermatol. 2009;145:1078. doi: 10.1001/archdermatol.2009.203
4. Renati S, Cukras A, Bigby M. Pityriasis versicolor. BMJ. 2015;350:h1394. doi: 10.1136/bmj.h1394
1. Crespo-Erchiga, V, Florencio, VD. Malassezia yeasts and pityriasis versicolor. Curr Opin Infect Dis. 2006;19:139-147. doi: 10.1097/01.qco.0000216624.21069.61
2. Gupta, AK, Bluhm R, Summerbell R. Pityriasis versicolor. J Eur Acad Dermatol Venereol. 2002;16:19-33. doi: 10.1046/j.1468-3083.2002.00378.x
3. Han, A, Calcara DA, Stoecker WV, et al. Evoked scale sign of tinea versicolor. Arch Dermatol. 2009;145:1078. doi: 10.1001/archdermatol.2009.203
4. Renati S, Cukras A, Bigby M. Pityriasis versicolor. BMJ. 2015;350:h1394. doi: 10.1136/bmj.h1394