User login
Erythrasma
THE COMPARISON
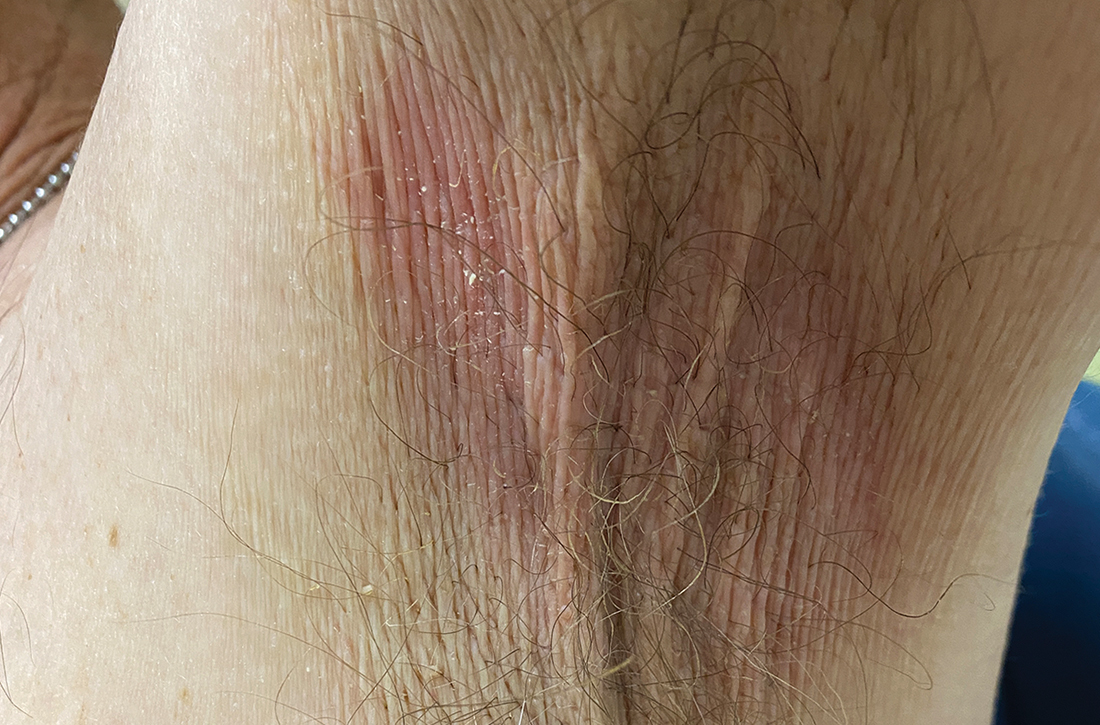
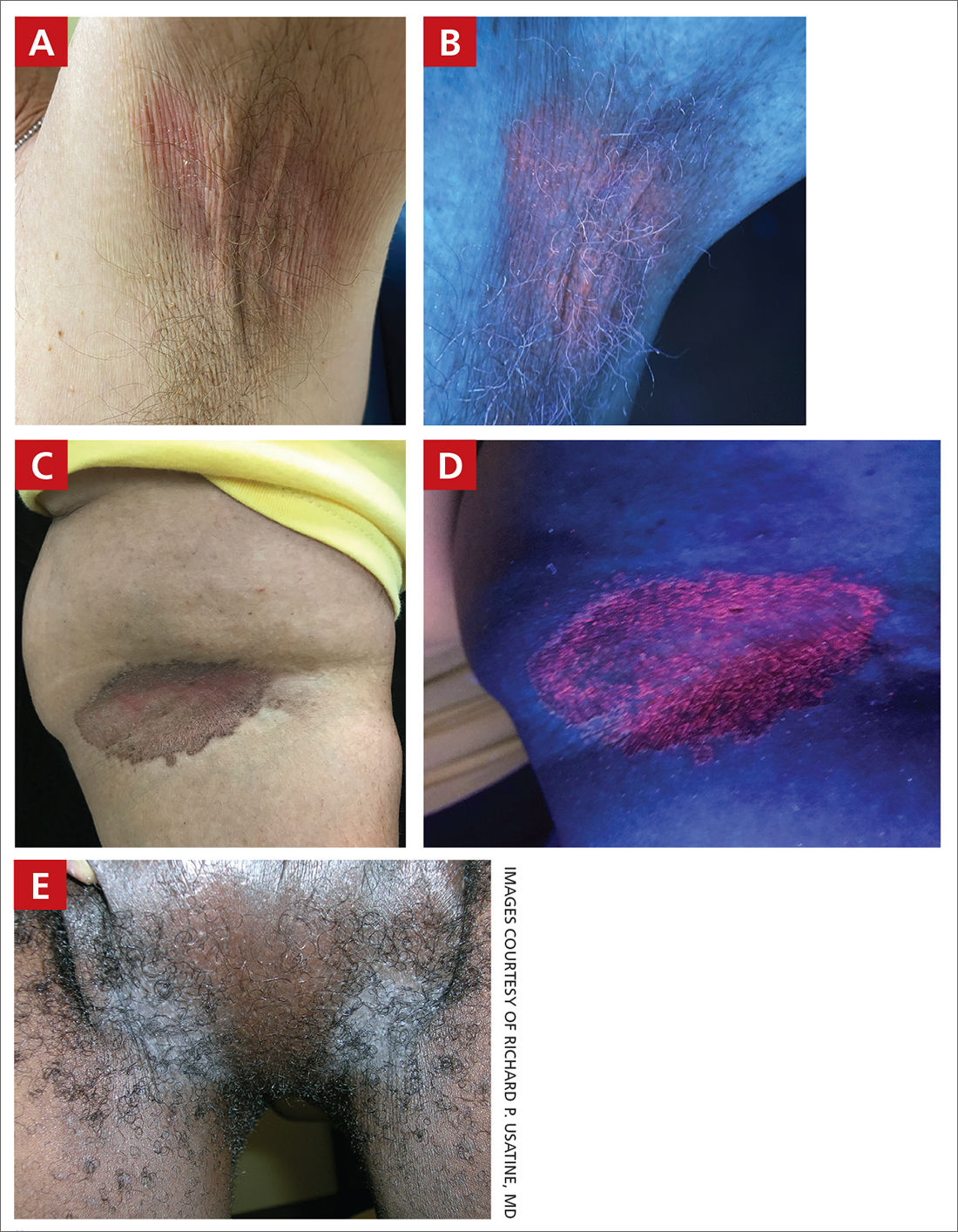
A and B Axilla of a 65-year-old White man with erythrasma showing a well-demarcated erythematous plaque with fine scale (A). Wood-lamp examination of the area showed characteristic bright coral red fluorescence (B).
C and D A well-demarcated, red-brown plaque with fine scale in the antecubital fossa of an obese Hispanic woman (C). Wood-lamp examination revealed bright coral red fluorescence (D).
E Hypopigmented patches (with pruritus) in the groin of a Black man. He also had erythrasma between the toes.
Erythrasma is a skin condition caused by acute or chronic infection of the outermost layer of the epidermis (stratum corneum) with Corynebacterium minutissimum. It has a predilection for intertriginous regions such as the axillae, groin, and interdigital spaces of the toes. It can be associated with pruritus or can be asymptomatic.
Epidemiology
Erythrasma typically affects adults, with greater prevalence among those residing in shared living facilities, such as dormitories or nursing homes, or in humid climates.1 It is a common disorder with an estimated prevalence of 17.6% of bacterial skin infections in elderly patients and 44% of diabetic interdigital toe space infections.2,3
Key clinical features
Erythrasma can manifest as red-brown hyperpigmented plaques with fine scale and little central clearing (FIGURES A and C) or as a hypopigmented patch (FIGURE E) with a sharply marginated, hyperpigmented border in patients with skin of color. In the interdigital toe spaces, the skin often is white and macerated. These findings may appear in patients of all skin tones.
Worth noting
- C minutissimum produces coproporphyrin III, which glows fluorescent red under Wood-lamp examination (FIGURES B and D). A recent shower or bath may remove the fluorescent coproporphyrins and cause a false-negative result. The interdigital space between the fourth and fifth toes is a common location for C minutissimum; thus clinicians should consider examining these areas with a Wood lamp.
- Associated risk factors include obesity, immunosuppression, diabetes mellitus, and excessive sweating.1
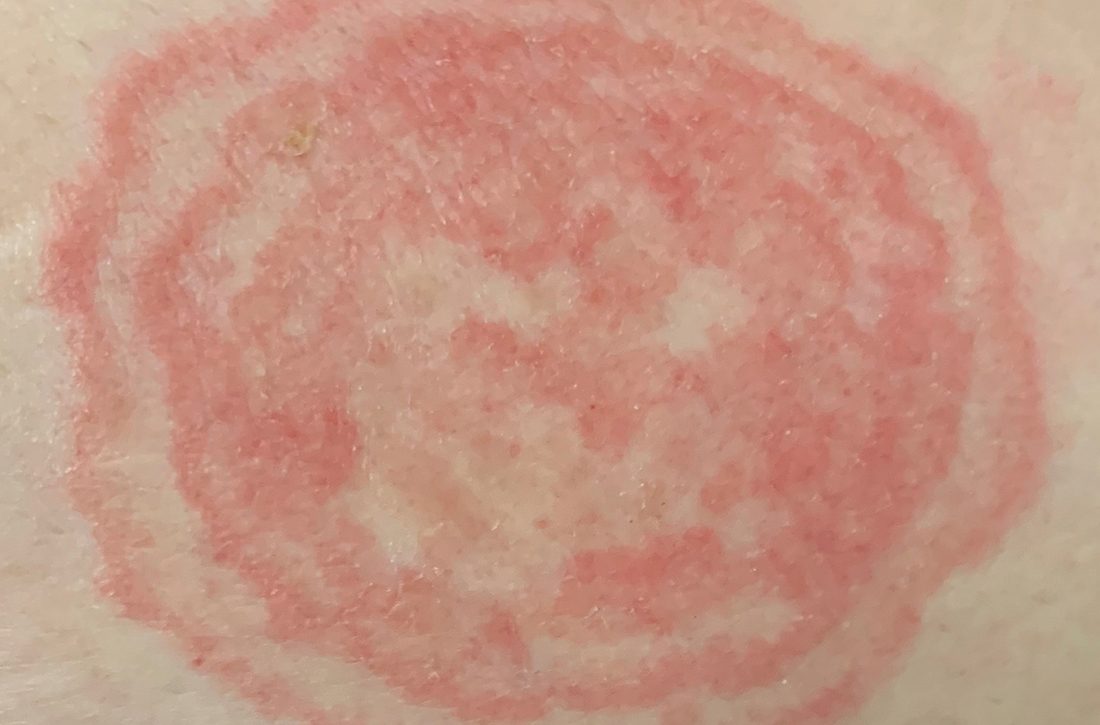
- The differential diagnosis includes intertrigo, inverse psoriasis, confluent and reticulated papillomatosis (Gougerot-Carteaud syndrome), acanthosis nigricans, seborrheic dermatitis, and tinea pedis when present in the interdigital toe spaces. Plaques occurring in circular patterns may be mistaken for tinea corporis or pityriasis rotunda.
- There is a high prevalence of erythrasma in patients with inverse psoriasis, and it may exacerbate psoriatic plaques.4
- Treatment options include application of topical clindamycin or erythromycin to the affected area.1 Some patients have responded to topical mupiricin.2 For larger areas, a 1-g dose of clarithromycin5 or a 14-day course of erythromycin may be appropriate.1 Avoid prescribing clarithromycin to patients with preexisting heart disease due to its increased risk for cardiac events or death; consider other agents.
Health disparity highlight
Obesity, most prevalent in non-Hispanic Black adults (49.9%) and Hispanic adults (45.6%) followed by non-Hispanic White adults (41.4%),6 may cause velvety dark plaques on the neck called acanthosis nigricans. However, acute or chronic erythrasma also may cause hyperpigmentation of the body folds. Although the pathology of erythrasma is due to bacterial infection of the superficial layer of the stratum corneum, acanthosis nigricans is due to fibroblast proliferation and stimulation of epidermal keratinocytes, likely from increased growth factors and insulinlike growth factor.7 If erythrasma is mistaken for acanthosis nigricans, the patient may be counseled inappropriately that the hyperpigmentation is something not easily resolved and subsequently left with an active treatable condition that adversely affects their quality of life.
1. Groves JB, Nassereddin A, Freeman AM. Erythrasma. In: StatPearls. StatPearls Publishing; August 11, 2021. Accessed November 17, 2022. https://www.ncbi.nlm.nih.gov/books/NBK513352/
2. Forouzan P, Cohen PR. Erythrasma revisited: diagnosis, differential diagnoses, and comprehensive review of treatment. Cureus. 2020;12:E10733. doi:10.7759/cureus.10733
3. Polat M, I˙lhan MN. Dermatological complaints of the elderly attending a dermatology outpatient clinic in Turkey: a prospective study over a one-year period. Acta Dermatovenerol Croat. 2015;23:277-281.
4. Janeczek M, Kozel Z, Bhasin R, et al. High prevalence of erythrasma in patients with inverse psoriasis: a cross-sectional study. J Clin Aesthet Dermatol. 2020;13:12-14.
5. Khan MJ. Interdigital pedal erythrasma treated with one-time dose of oral clarithromycin 1 g: two case reports. Clin Case Rep. 2020;8:672-674. doi:10.1002/ccr3.2712
6. Stierman B, Afful J, Carroll M, et al. National Health and Nutrition Examination Survey 2017–March 2020 Prepandemic Data Files Development of Files and Prevalence Estimates for Selected Health Outcomes. National Health Statistics Reports. Published June 14, 2021. Accessed November 17, 2022. https://stacks.cdc.gov/view/cdc/106273
7. Brady MF, Rawla P. Acanthosis nigricans. In: StatPearls. Stat- Pearls Publishing; 2022. Updated October 9, 2022. Accessed November 30, 2022. https://www.ncbi.nlm.nih.gov/books/NBK431057
THE COMPARISON
A and B Axilla of a 65-year-old White man with erythrasma showing a well-demarcated erythematous plaque with fine scale (A). Wood-lamp examination of the area showed characteristic bright coral red fluorescence (B).
C and D A well-demarcated, red-brown plaque with fine scale in the antecubital fossa of an obese Hispanic woman (C). Wood-lamp examination revealed bright coral red fluorescence (D).
E Hypopigmented patches (with pruritus) in the groin of a Black man. He also had erythrasma between the toes.
Erythrasma is a skin condition caused by acute or chronic infection of the outermost layer of the epidermis (stratum corneum) with Corynebacterium minutissimum. It has a predilection for intertriginous regions such as the axillae, groin, and interdigital spaces of the toes. It can be associated with pruritus or can be asymptomatic.

Epidemiology
Erythrasma typically affects adults, with greater prevalence among those residing in shared living facilities, such as dormitories or nursing homes, or in humid climates.1 It is a common disorder with an estimated prevalence of 17.6% of bacterial skin infections in elderly patients and 44% of diabetic interdigital toe space infections.2,3
Key clinical features
Erythrasma can manifest as red-brown hyperpigmented plaques with fine scale and little central clearing (FIGURES A and C) or as a hypopigmented patch (FIGURE E) with a sharply marginated, hyperpigmented border in patients with skin of color. In the interdigital toe spaces, the skin often is white and macerated. These findings may appear in patients of all skin tones.
Worth noting
- C minutissimum produces coproporphyrin III, which glows fluorescent red under Wood-lamp examination (FIGURES B and D). A recent shower or bath may remove the fluorescent coproporphyrins and cause a false-negative result. The interdigital space between the fourth and fifth toes is a common location for C minutissimum; thus clinicians should consider examining these areas with a Wood lamp.
- Associated risk factors include obesity, immunosuppression, diabetes mellitus, and excessive sweating.1
- The differential diagnosis includes intertrigo, inverse psoriasis, confluent and reticulated papillomatosis (Gougerot-Carteaud syndrome), acanthosis nigricans, seborrheic dermatitis, and tinea pedis when present in the interdigital toe spaces. Plaques occurring in circular patterns may be mistaken for tinea corporis or pityriasis rotunda.
- There is a high prevalence of erythrasma in patients with inverse psoriasis, and it may exacerbate psoriatic plaques.4
- Treatment options include application of topical clindamycin or erythromycin to the affected area.1 Some patients have responded to topical mupiricin.2 For larger areas, a 1-g dose of clarithromycin5 or a 14-day course of erythromycin may be appropriate.1 Avoid prescribing clarithromycin to patients with preexisting heart disease due to its increased risk for cardiac events or death; consider other agents.
Health disparity highlight
Obesity, most prevalent in non-Hispanic Black adults (49.9%) and Hispanic adults (45.6%) followed by non-Hispanic White adults (41.4%),6 may cause velvety dark plaques on the neck called acanthosis nigricans. However, acute or chronic erythrasma also may cause hyperpigmentation of the body folds. Although the pathology of erythrasma is due to bacterial infection of the superficial layer of the stratum corneum, acanthosis nigricans is due to fibroblast proliferation and stimulation of epidermal keratinocytes, likely from increased growth factors and insulinlike growth factor.7 If erythrasma is mistaken for acanthosis nigricans, the patient may be counseled inappropriately that the hyperpigmentation is something not easily resolved and subsequently left with an active treatable condition that adversely affects their quality of life.
THE COMPARISON
A and B Axilla of a 65-year-old White man with erythrasma showing a well-demarcated erythematous plaque with fine scale (A). Wood-lamp examination of the area showed characteristic bright coral red fluorescence (B).
C and D A well-demarcated, red-brown plaque with fine scale in the antecubital fossa of an obese Hispanic woman (C). Wood-lamp examination revealed bright coral red fluorescence (D).
E Hypopigmented patches (with pruritus) in the groin of a Black man. He also had erythrasma between the toes.
Erythrasma is a skin condition caused by acute or chronic infection of the outermost layer of the epidermis (stratum corneum) with Corynebacterium minutissimum. It has a predilection for intertriginous regions such as the axillae, groin, and interdigital spaces of the toes. It can be associated with pruritus or can be asymptomatic.

Epidemiology
Erythrasma typically affects adults, with greater prevalence among those residing in shared living facilities, such as dormitories or nursing homes, or in humid climates.1 It is a common disorder with an estimated prevalence of 17.6% of bacterial skin infections in elderly patients and 44% of diabetic interdigital toe space infections.2,3
Key clinical features
Erythrasma can manifest as red-brown hyperpigmented plaques with fine scale and little central clearing (FIGURES A and C) or as a hypopigmented patch (FIGURE E) with a sharply marginated, hyperpigmented border in patients with skin of color. In the interdigital toe spaces, the skin often is white and macerated. These findings may appear in patients of all skin tones.
Worth noting
- C minutissimum produces coproporphyrin III, which glows fluorescent red under Wood-lamp examination (FIGURES B and D). A recent shower or bath may remove the fluorescent coproporphyrins and cause a false-negative result. The interdigital space between the fourth and fifth toes is a common location for C minutissimum; thus clinicians should consider examining these areas with a Wood lamp.
- Associated risk factors include obesity, immunosuppression, diabetes mellitus, and excessive sweating.1
- The differential diagnosis includes intertrigo, inverse psoriasis, confluent and reticulated papillomatosis (Gougerot-Carteaud syndrome), acanthosis nigricans, seborrheic dermatitis, and tinea pedis when present in the interdigital toe spaces. Plaques occurring in circular patterns may be mistaken for tinea corporis or pityriasis rotunda.
- There is a high prevalence of erythrasma in patients with inverse psoriasis, and it may exacerbate psoriatic plaques.4
- Treatment options include application of topical clindamycin or erythromycin to the affected area.1 Some patients have responded to topical mupiricin.2 For larger areas, a 1-g dose of clarithromycin5 or a 14-day course of erythromycin may be appropriate.1 Avoid prescribing clarithromycin to patients with preexisting heart disease due to its increased risk for cardiac events or death; consider other agents.
Health disparity highlight
Obesity, most prevalent in non-Hispanic Black adults (49.9%) and Hispanic adults (45.6%) followed by non-Hispanic White adults (41.4%),6 may cause velvety dark plaques on the neck called acanthosis nigricans. However, acute or chronic erythrasma also may cause hyperpigmentation of the body folds. Although the pathology of erythrasma is due to bacterial infection of the superficial layer of the stratum corneum, acanthosis nigricans is due to fibroblast proliferation and stimulation of epidermal keratinocytes, likely from increased growth factors and insulinlike growth factor.7 If erythrasma is mistaken for acanthosis nigricans, the patient may be counseled inappropriately that the hyperpigmentation is something not easily resolved and subsequently left with an active treatable condition that adversely affects their quality of life.
1. Groves JB, Nassereddin A, Freeman AM. Erythrasma. In: StatPearls. StatPearls Publishing; August 11, 2021. Accessed November 17, 2022. https://www.ncbi.nlm.nih.gov/books/NBK513352/
2. Forouzan P, Cohen PR. Erythrasma revisited: diagnosis, differential diagnoses, and comprehensive review of treatment. Cureus. 2020;12:E10733. doi:10.7759/cureus.10733
3. Polat M, I˙lhan MN. Dermatological complaints of the elderly attending a dermatology outpatient clinic in Turkey: a prospective study over a one-year period. Acta Dermatovenerol Croat. 2015;23:277-281.
4. Janeczek M, Kozel Z, Bhasin R, et al. High prevalence of erythrasma in patients with inverse psoriasis: a cross-sectional study. J Clin Aesthet Dermatol. 2020;13:12-14.
5. Khan MJ. Interdigital pedal erythrasma treated with one-time dose of oral clarithromycin 1 g: two case reports. Clin Case Rep. 2020;8:672-674. doi:10.1002/ccr3.2712
6. Stierman B, Afful J, Carroll M, et al. National Health and Nutrition Examination Survey 2017–March 2020 Prepandemic Data Files Development of Files and Prevalence Estimates for Selected Health Outcomes. National Health Statistics Reports. Published June 14, 2021. Accessed November 17, 2022. https://stacks.cdc.gov/view/cdc/106273
7. Brady MF, Rawla P. Acanthosis nigricans. In: StatPearls. Stat- Pearls Publishing; 2022. Updated October 9, 2022. Accessed November 30, 2022. https://www.ncbi.nlm.nih.gov/books/NBK431057
1. Groves JB, Nassereddin A, Freeman AM. Erythrasma. In: StatPearls. StatPearls Publishing; August 11, 2021. Accessed November 17, 2022. https://www.ncbi.nlm.nih.gov/books/NBK513352/
2. Forouzan P, Cohen PR. Erythrasma revisited: diagnosis, differential diagnoses, and comprehensive review of treatment. Cureus. 2020;12:E10733. doi:10.7759/cureus.10733
3. Polat M, I˙lhan MN. Dermatological complaints of the elderly attending a dermatology outpatient clinic in Turkey: a prospective study over a one-year period. Acta Dermatovenerol Croat. 2015;23:277-281.
4. Janeczek M, Kozel Z, Bhasin R, et al. High prevalence of erythrasma in patients with inverse psoriasis: a cross-sectional study. J Clin Aesthet Dermatol. 2020;13:12-14.
5. Khan MJ. Interdigital pedal erythrasma treated with one-time dose of oral clarithromycin 1 g: two case reports. Clin Case Rep. 2020;8:672-674. doi:10.1002/ccr3.2712
6. Stierman B, Afful J, Carroll M, et al. National Health and Nutrition Examination Survey 2017–March 2020 Prepandemic Data Files Development of Files and Prevalence Estimates for Selected Health Outcomes. National Health Statistics Reports. Published June 14, 2021. Accessed November 17, 2022. https://stacks.cdc.gov/view/cdc/106273
7. Brady MF, Rawla P. Acanthosis nigricans. In: StatPearls. Stat- Pearls Publishing; 2022. Updated October 9, 2022. Accessed November 30, 2022. https://www.ncbi.nlm.nih.gov/books/NBK431057
Melanoma mortality rates fell in 2010s as new therapies took hold
, a new study finds, although the dip appeared to stabilize over the next 2 years.
“This data is very encouraging and represents the real-world effectiveness of these newer therapies, which include immunotherapies and targeted therapies,” hematologist/oncologist Navkirat Kahlon, MD, MPH, of Seacoast Cancer Center and Massachusetts General Brigham Wentworth-Douglass Hospital, Dover, N.H., one of the study authors, said in an interview. In clinical trials, these new treatments “have been very effective ... so the timing as well as magnitude of drop seen in melanoma-specific population mortality was not at all surprising. But it’s still very exciting.”
The report, published in JAMA Network Open, tracked mortality rates for the deadliest form of skin cancer from 1975 to 2019. The researchers launched the study to better understand outcomes in cutaneous melanoma following the rise of new therapies that now provide options in addition to chemotherapy. “With the use of novel therapies, the survival of these patients has increased from a few weeks or months to many years in clinical trials,” Dr. Kahlon said. “Given the magnitude of benefit compared to traditional chemotherapy in clinical trials, we decided to see if the real-world U.S. population is deriving the same benefit.”
New drugs introduced in recent years include immunotherapy agents such as ipilimumab and targeted therapies such as vemurafenib.
The researchers analyzed age-adjusted melanoma outcome data from the Surveillance, Epidemiology, and End Results (SEER) database. In 1975, the long-term melanoma mortality rate for melanoma was 2.07 per 100,000 people (95% confidence interval [CI], 2.00-2.13). It rose to 2.65 (95% CI, 2.58-2.65) in 1988 and 2.67 (95% CI, 2.61-2.72) in 2013, then fell to 2.09 (95% CI, 2.05-2.14) in 2017 and 2.01 (95% CI, 1.97-2.06) in 2019.
Per the analysis, the rate grew at an annual percentage change (APC) of 1.65% (95% CI, 1.30%-2.00%, P < .001) from 1975 to 1988 and remained stable from 1988 to 2013 (P = .85). Then it fell by an APC of 6.28% (95% CI, –8.52% to –3.97%, P < .001) from 2013 to 2017. There was no statistically significant difference between 2017 and 2019, although “the trend was downward,” the authors noted.
“Our study didn’t study the parameters that can answer the question about how many more years of life patients are getting or how many lives aren’t lost,” Dr. Kahlon said in the interview. “But looking at other studies and clinical trial data, the prognosis of these patients with a historical median overall survival of a few weeks to months has improved to many months to years.”
But why do melanoma mortality rates remain roughly about the same as they were in 1975? “The incidence of melanoma has continued to rise,” she said. “Also, over time, we have become better at collecting more accurate information, so the [rate] in 1975 could potentially be an underestimated rate.”
In an interview, dermatologist Adewole “Ade” Adamson, MD, MPP, of the University of Texas at Austin, noted that a 2020 study examined melanoma death rates in Whites – who are most affected by melanoma – and found similar trends from 2013 to 2016. “Nothing about these [new] findings surprises me as they have been shown before. However, these confirmatory findings are reassuring because they show the powerful effect of novel treatments at a population level.”
Which treatments are making the biggest difference? “It is difficult to say, but it’s likely immunotherapy because some patients on these medications have durable responses for many years,” Dr. Adamson said. “Studies are ongoing to figure out just how much more life some patients may expect after treatment.”
He added that “while this study did not evaluate mortality trends by race or ethnicity, it is important to note that the sharp decline in melanoma mortality rates is exclusively among non-Hispanic White Americans.”
Dermatologist David Polsky, MD, PhD, professor of dermatologic oncology at New York (N.Y.) University, said in an interview that the findings reflect extraordinary progress in melanoma treatment. “Historically, only 10% of metastatic melanoma patients would live 5 years. And now 30% to 50% of metastatic patients are living 5 years. That’s amazing to me,” said Dr. Polsky, who coauthored the 2020 report cited by Dr. Adamson.
A few years ago, Dr. Polsky added, he talked to an oncologist about how life at her clinic had changed as a result of new melanoma treatments. “She said, ‘My clinic has gotten really crowded. It used to be that patients died in a span of about a year and a half, and I would turn over my patient population. Now all those patients are still alive, and I’m getting new patients.’”
The study was funded by the University of Toledo College of Medicine and Life Sciences. One author reported receiving honoraria from Boston Healthcare Associates and research funding from Bayer, ImmunoVaccine, and the Ludwig Institute for Cancer Research. Dr. Polsky disclosed relationships with Merck (advisory board) and Novartis and Bristol Myers Squibb (consulting). Dr. Adamson disclosed he is web editor and associate editor at JAMA Dermatology.
, a new study finds, although the dip appeared to stabilize over the next 2 years.
“This data is very encouraging and represents the real-world effectiveness of these newer therapies, which include immunotherapies and targeted therapies,” hematologist/oncologist Navkirat Kahlon, MD, MPH, of Seacoast Cancer Center and Massachusetts General Brigham Wentworth-Douglass Hospital, Dover, N.H., one of the study authors, said in an interview. In clinical trials, these new treatments “have been very effective ... so the timing as well as magnitude of drop seen in melanoma-specific population mortality was not at all surprising. But it’s still very exciting.”
The report, published in JAMA Network Open, tracked mortality rates for the deadliest form of skin cancer from 1975 to 2019. The researchers launched the study to better understand outcomes in cutaneous melanoma following the rise of new therapies that now provide options in addition to chemotherapy. “With the use of novel therapies, the survival of these patients has increased from a few weeks or months to many years in clinical trials,” Dr. Kahlon said. “Given the magnitude of benefit compared to traditional chemotherapy in clinical trials, we decided to see if the real-world U.S. population is deriving the same benefit.”
New drugs introduced in recent years include immunotherapy agents such as ipilimumab and targeted therapies such as vemurafenib.
The researchers analyzed age-adjusted melanoma outcome data from the Surveillance, Epidemiology, and End Results (SEER) database. In 1975, the long-term melanoma mortality rate for melanoma was 2.07 per 100,000 people (95% confidence interval [CI], 2.00-2.13). It rose to 2.65 (95% CI, 2.58-2.65) in 1988 and 2.67 (95% CI, 2.61-2.72) in 2013, then fell to 2.09 (95% CI, 2.05-2.14) in 2017 and 2.01 (95% CI, 1.97-2.06) in 2019.
Per the analysis, the rate grew at an annual percentage change (APC) of 1.65% (95% CI, 1.30%-2.00%, P < .001) from 1975 to 1988 and remained stable from 1988 to 2013 (P = .85). Then it fell by an APC of 6.28% (95% CI, –8.52% to –3.97%, P < .001) from 2013 to 2017. There was no statistically significant difference between 2017 and 2019, although “the trend was downward,” the authors noted.
“Our study didn’t study the parameters that can answer the question about how many more years of life patients are getting or how many lives aren’t lost,” Dr. Kahlon said in the interview. “But looking at other studies and clinical trial data, the prognosis of these patients with a historical median overall survival of a few weeks to months has improved to many months to years.”
But why do melanoma mortality rates remain roughly about the same as they were in 1975? “The incidence of melanoma has continued to rise,” she said. “Also, over time, we have become better at collecting more accurate information, so the [rate] in 1975 could potentially be an underestimated rate.”
In an interview, dermatologist Adewole “Ade” Adamson, MD, MPP, of the University of Texas at Austin, noted that a 2020 study examined melanoma death rates in Whites – who are most affected by melanoma – and found similar trends from 2013 to 2016. “Nothing about these [new] findings surprises me as they have been shown before. However, these confirmatory findings are reassuring because they show the powerful effect of novel treatments at a population level.”
Which treatments are making the biggest difference? “It is difficult to say, but it’s likely immunotherapy because some patients on these medications have durable responses for many years,” Dr. Adamson said. “Studies are ongoing to figure out just how much more life some patients may expect after treatment.”
He added that “while this study did not evaluate mortality trends by race or ethnicity, it is important to note that the sharp decline in melanoma mortality rates is exclusively among non-Hispanic White Americans.”
Dermatologist David Polsky, MD, PhD, professor of dermatologic oncology at New York (N.Y.) University, said in an interview that the findings reflect extraordinary progress in melanoma treatment. “Historically, only 10% of metastatic melanoma patients would live 5 years. And now 30% to 50% of metastatic patients are living 5 years. That’s amazing to me,” said Dr. Polsky, who coauthored the 2020 report cited by Dr. Adamson.
A few years ago, Dr. Polsky added, he talked to an oncologist about how life at her clinic had changed as a result of new melanoma treatments. “She said, ‘My clinic has gotten really crowded. It used to be that patients died in a span of about a year and a half, and I would turn over my patient population. Now all those patients are still alive, and I’m getting new patients.’”
The study was funded by the University of Toledo College of Medicine and Life Sciences. One author reported receiving honoraria from Boston Healthcare Associates and research funding from Bayer, ImmunoVaccine, and the Ludwig Institute for Cancer Research. Dr. Polsky disclosed relationships with Merck (advisory board) and Novartis and Bristol Myers Squibb (consulting). Dr. Adamson disclosed he is web editor and associate editor at JAMA Dermatology.
, a new study finds, although the dip appeared to stabilize over the next 2 years.
“This data is very encouraging and represents the real-world effectiveness of these newer therapies, which include immunotherapies and targeted therapies,” hematologist/oncologist Navkirat Kahlon, MD, MPH, of Seacoast Cancer Center and Massachusetts General Brigham Wentworth-Douglass Hospital, Dover, N.H., one of the study authors, said in an interview. In clinical trials, these new treatments “have been very effective ... so the timing as well as magnitude of drop seen in melanoma-specific population mortality was not at all surprising. But it’s still very exciting.”
The report, published in JAMA Network Open, tracked mortality rates for the deadliest form of skin cancer from 1975 to 2019. The researchers launched the study to better understand outcomes in cutaneous melanoma following the rise of new therapies that now provide options in addition to chemotherapy. “With the use of novel therapies, the survival of these patients has increased from a few weeks or months to many years in clinical trials,” Dr. Kahlon said. “Given the magnitude of benefit compared to traditional chemotherapy in clinical trials, we decided to see if the real-world U.S. population is deriving the same benefit.”
New drugs introduced in recent years include immunotherapy agents such as ipilimumab and targeted therapies such as vemurafenib.
The researchers analyzed age-adjusted melanoma outcome data from the Surveillance, Epidemiology, and End Results (SEER) database. In 1975, the long-term melanoma mortality rate for melanoma was 2.07 per 100,000 people (95% confidence interval [CI], 2.00-2.13). It rose to 2.65 (95% CI, 2.58-2.65) in 1988 and 2.67 (95% CI, 2.61-2.72) in 2013, then fell to 2.09 (95% CI, 2.05-2.14) in 2017 and 2.01 (95% CI, 1.97-2.06) in 2019.
Per the analysis, the rate grew at an annual percentage change (APC) of 1.65% (95% CI, 1.30%-2.00%, P < .001) from 1975 to 1988 and remained stable from 1988 to 2013 (P = .85). Then it fell by an APC of 6.28% (95% CI, –8.52% to –3.97%, P < .001) from 2013 to 2017. There was no statistically significant difference between 2017 and 2019, although “the trend was downward,” the authors noted.
“Our study didn’t study the parameters that can answer the question about how many more years of life patients are getting or how many lives aren’t lost,” Dr. Kahlon said in the interview. “But looking at other studies and clinical trial data, the prognosis of these patients with a historical median overall survival of a few weeks to months has improved to many months to years.”
But why do melanoma mortality rates remain roughly about the same as they were in 1975? “The incidence of melanoma has continued to rise,” she said. “Also, over time, we have become better at collecting more accurate information, so the [rate] in 1975 could potentially be an underestimated rate.”
In an interview, dermatologist Adewole “Ade” Adamson, MD, MPP, of the University of Texas at Austin, noted that a 2020 study examined melanoma death rates in Whites – who are most affected by melanoma – and found similar trends from 2013 to 2016. “Nothing about these [new] findings surprises me as they have been shown before. However, these confirmatory findings are reassuring because they show the powerful effect of novel treatments at a population level.”
Which treatments are making the biggest difference? “It is difficult to say, but it’s likely immunotherapy because some patients on these medications have durable responses for many years,” Dr. Adamson said. “Studies are ongoing to figure out just how much more life some patients may expect after treatment.”
He added that “while this study did not evaluate mortality trends by race or ethnicity, it is important to note that the sharp decline in melanoma mortality rates is exclusively among non-Hispanic White Americans.”
Dermatologist David Polsky, MD, PhD, professor of dermatologic oncology at New York (N.Y.) University, said in an interview that the findings reflect extraordinary progress in melanoma treatment. “Historically, only 10% of metastatic melanoma patients would live 5 years. And now 30% to 50% of metastatic patients are living 5 years. That’s amazing to me,” said Dr. Polsky, who coauthored the 2020 report cited by Dr. Adamson.
A few years ago, Dr. Polsky added, he talked to an oncologist about how life at her clinic had changed as a result of new melanoma treatments. “She said, ‘My clinic has gotten really crowded. It used to be that patients died in a span of about a year and a half, and I would turn over my patient population. Now all those patients are still alive, and I’m getting new patients.’”
The study was funded by the University of Toledo College of Medicine and Life Sciences. One author reported receiving honoraria from Boston Healthcare Associates and research funding from Bayer, ImmunoVaccine, and the Ludwig Institute for Cancer Research. Dr. Polsky disclosed relationships with Merck (advisory board) and Novartis and Bristol Myers Squibb (consulting). Dr. Adamson disclosed he is web editor and associate editor at JAMA Dermatology.
FROM JAMA NETWORK OPEN
A practical guide to hidradenitis suppurativa
Hidradenitis suppurativa (HS), also known as acne inversa or Verneuil disease, is a chronic, recurrent, inflammatory occlusive disease affecting the terminal follicular epithelium in apocrine gland–bearing skin areas.1 HS manifests as painful nodules, abscesses, fistulas, and scarring and often has a severe psychological impact on the affected patient.2
When HS was first identified in the 1800s, it was believed to result from a dysfunction of the sweat glands.3 In 1939, scientists identified the true cause: follicular occlusion.3
Due to its chronic nature, heterogeneity in presentation, and apparent low prevalence,4 HS is considered an orphan disease.5 Over the past 10 years, there has been a surge in HS research—particularly in medical management—which has provided a better understanding of this condition.6,7
In this review, we discuss the most updated evidence regarding the diagnosis and treatment of HS to guide the family physician (FP)’s approach to managing this debilitating disease. But first, we offer a word about the etiology and pathophysiology of the condition.
3 events set the stage for hidradenitis suppurativa
Although the exact cause of HS is still unknown, some researchers have hypothesized that HS results from a combination of genetic predisposition and environmental and lifestyle factors.8-12 The primary mechanism of HS is the obstruction of the terminal follicular epithelium by a keratin plug.1,13,14 A systematic review of molecular inflammatory pathways involved in HS divides the pathogenesis of HS into 3 events: follicular occlusion followed by dilation, follicular rupture and inflammatory response, and chronic inflammatory state with sinus tracts.8
An underreported condition
HS is often underreported and misdiagnosed.4,15 Globally, the prevalence of HS varies from < 1% to 4%.15,16 A systematic review with meta-analysis showed a higher prevalence of HS in females compared to males in American and European populations.17 In the United States, the overall frequency of HS is 0.1%, or 98 per 100,000 persons.16 The prevalence of HS is highest among patients ages 30 to 39 years; there is decreased prevalence in patients ages 55 years and older.16,18
Who is at heightened risk?
Recent research has shown a relationship between ethnicity and HS.16,19,20 African American and biracial groups (defined as African American and White) have a 3-fold and 2-fold greater prevalence of HS, respectively, compared to White patients.16 However, the prevalence of HS in non-White ethnic groups may be underestimated in clinical trials due to a lack of representation and subgroup analyses based on ethnicity, which may affect generalizability in HS recommendations.21
Continue to: Genetic predisposition
Genetic predisposition. As many as 40% of patients with HS report having at least 1 affected family member. A positive family history of HS is associated with earlier onset, longer disease duration, and severe disease.22 HS is genetically heterogeneous, and several mutations (eg, gamma secretase, PSTPIP1, PSEN1 genes) have been identified in patients and in vitro as the cause of dysregulation of epidermal proliferation and differentiation, immune dysregulation, and promotion of amyloid formation.8,23-25
Obesity and metabolic risk factors. There is a strong relationship between HS and obesity. As many as 70% of patients with HS are obese, and 9% to 40% have metabolic syndrome.12,18,26-28 Obesity is associated with maceration and mechanical stress, increased fragility of the dermo-epidermal junction, changes in cutaneous blood flow, and subdermal fat inflammation—all of which favor the pathophysiology of HS.29,30
Smoking. Tobacco smoking is associated with severe HS and a lower chance of remission.12 Population-based studies have shown that as many as 90% of patients with HS have a history of smoking ≥ 20 packs of cigarettes per year.1,12,18,31,32 The nicotine and thousands of other chemicals present in cigarettes trigger keratinocytes and fibroblasts, resulting in epidermal hyperplasia, infundibular hyperkeratosis, excessive cornification, and dysbiosis.8,23,24
Hormones. The exact role sex hormones play in the pathogenesis of HS remains unclear.8,32 Most information is based primarily on small studies looking at antiandrogen treatments, HS activity during the menstrual cycle and pregnancy, HS exacerbation related to androgenic effects of hormonal contraception, and the association of HS with metabolic-endocrine disorders (eg, polycystic ovary syndrome [PCOS]).8,33
Androgens induce hyperkeratosis that may lead to follicular occlusion—the hallmark of HS pathology.34 A systematic review looking at the role of androgen and estrogen in HS found that while some patients with HS have elevated androgen levels, most have androgen and estrogen levels within normal range.35 Therefore, increased peripheral androgen receptor sensitivity has been hypothesized as the mechanism of action contributing to HS manifestation.34
Continue to: Host-defense defects
Host-defense defects. HS shares a similar cytokine profile with other well-established immune-mediated inflammatory diseases, including pyoderma gangrenosum (PG)36,37 and Crohn disease.38-40 HS is characterized by the expression of several immune mediators, including tumor necrosis factor-alpha (TNF-alpha), interleukin-1 alpha (IL-1 alpha), IL-1 beta, IL-8, IL-17, and the IL-23/T helper 17 pathway, all of which are upregulated in other inflammatory diseases and also result in an abnormal innate immune response.8,24 The recently described clinical triad of PG, acne, and HS (PASH) and the tetrad of pyogenic arthritis, PG, acne, and HS (PAPASH) further support the role of immune dysregulation in the pathogenesis of HS.40 Nonetheless, further studies are needed to determine the exact pathways of cytokine effect in HS.41
Use these criteria to make the diagnosis
The US and Canadian Hidradenitis Suppurativa Foundations (HSF) guidelines base the clinical diagnosis of HS on the following criteria2:
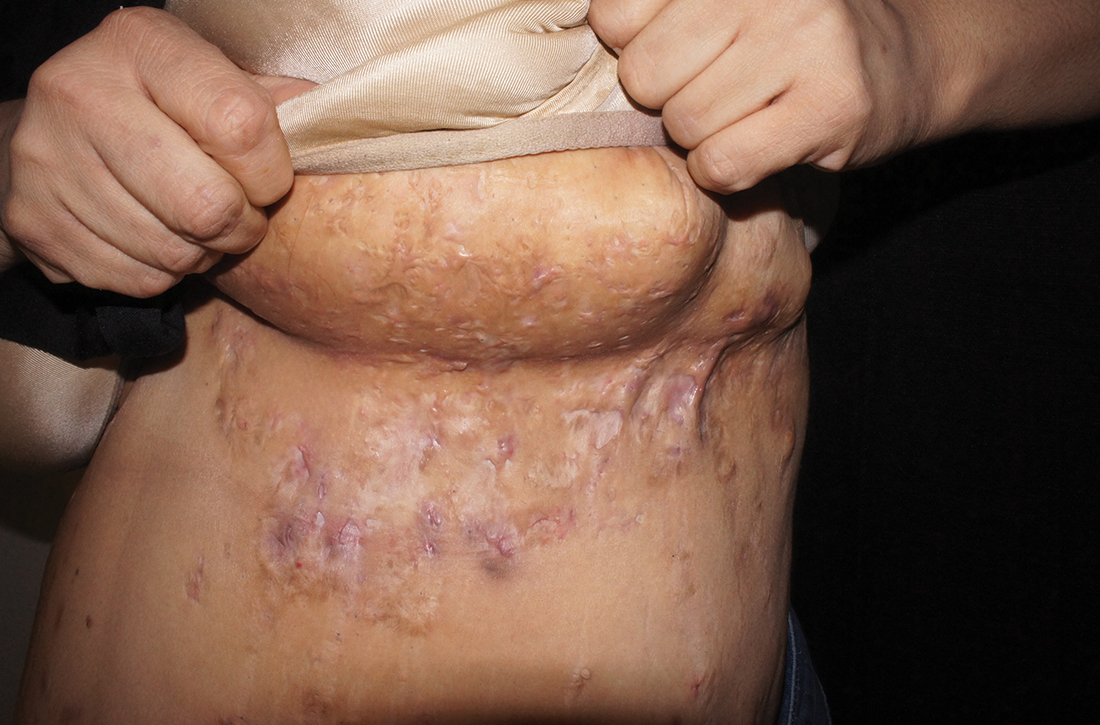
- Typical HS lesions: Erythematous skin lesions; inflamed, deep-seated painful nodules; “tombstone” double-ended comedones; sinus tracts; scarring; deformity. FIGURES 1A-1E show typical lesions seen in patients with HS.
- Typical locations: Intertriginous regions—apocrine gland–containing areas in axilla, groin, perineal region, buttocks, gluteal cleft, and mammary folds; beltline and waistband areas; areas of skin compression and friction.
- Recurrence and chronicity: Recurrent painful or suppurating lesions that appear more than twice in a 6-month period.2,41-43
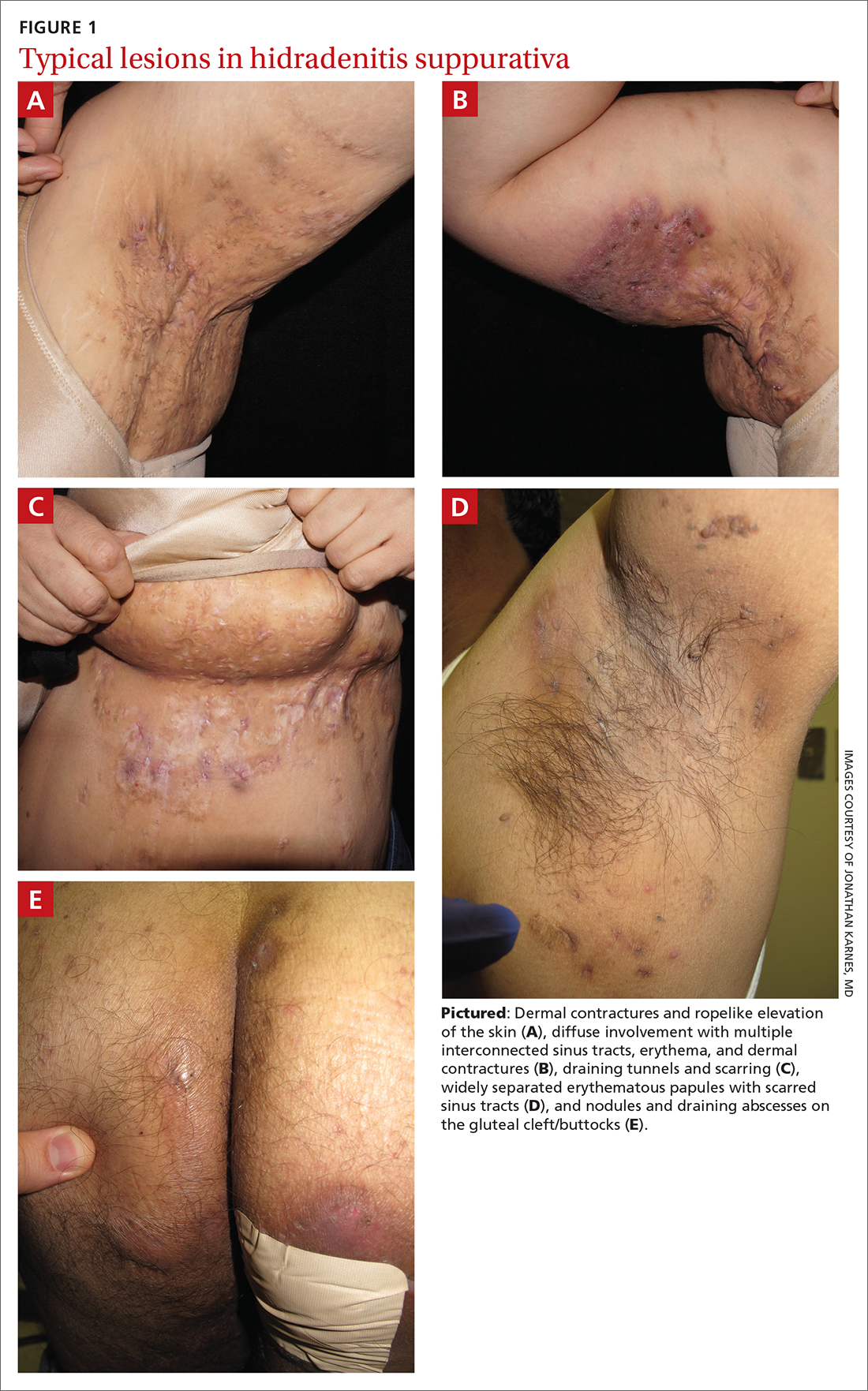
Patients with HS usually present with painful recurrent abscesses and scarring and often report multiple visits to the emergency department for drainage or failed antibiotic treatment for abscesses.15,44
Ask patients these 2 questions. Vinding et al45 developed a survey for the diagnosis of HS using 2 simple questions based on the 3 criteria established by the HSF:
- “Have you had an outbreak of boils during the last 6 months?” and
- “Where and how many boils have you had?” (This question includes a list of the typical HS locations—eg, axilla, groin, genitals, area under breast.)
In their questionnaire, Vinding et al45 found that an affirmative answer to Question 1 and reports of > 2 boils in response to Question 2 correlated to a sensitivity of 90%, specificity of 97%, positive predictive value of 96%, and negative predictive value of 92% for the diagnosis of HS. The differential diagnosis of HS is summarized in TABLE 1.42,45-52

Continue to: These tools can help you to stage hidradenitis suppurativa
These tools can help you to stage hidradenitis suppurativa
Multiple tools are available to assess the severity of HS.53 We will describe the Hurley staging system and the International Hidradenitis Suppurativa Severity Score System (IHS4). Other diagnostic tools, such as the Sartorius score and the Hidradenitis Suppurativa Physician’s Global Assessment Scale (HS-PGA), can be time-consuming and challenging to interpret, limiting their use in the clinical setting.2,54
Hurley staging system (available at www.hsdiseasesource.com/hs-disease-staging) considers the presence of nodules, abscesses, sinus tracts, and scarring affecting an entire anatomical area.13,55 This system is most useful as a rapid classification tool for patients with HS in the clinical setting but should not be used to assess clinical response.2,13,56
The IHS4 (available at https://online library.wiley.com/doi/10.1111/bjd.15748) is a validated and easy-to-use tool for assessing HS and guiding the therapeutic strategy in clinical practice.54 With IHS4, the clinician must calculate the following:
- total number of nodules > 10 mm in diameter
- total number of abscesses multiplied by 2, and
- total number of draining tunnels (fistulae/sinuses) multiplied by 4.
Mild HS is defined as a score ≤ 3 points; moderate HS, 4 to 10 points; and severe HS, ≥ 11 points.54
No diagnostic tests, but ultrasound may be helpful
There are currently no established biological markers or specific tests for diagnosing HS.15 Ultrasound is emerging as a tool to assess dermal thickness, hair follicle morphology, and number and extent of fluid collections. Two recent studies showed that pairing clinical assessment with ultrasound findings improves accuracy of scoring in 84% of cases.57,58 For patients with severe HS, skin biopsy can be considered to rule out squamous cell carcinoma. Cultures, however, have limited utility except for suspected superimposed bacterial infection.2
Continue to: Screening for comorbidities
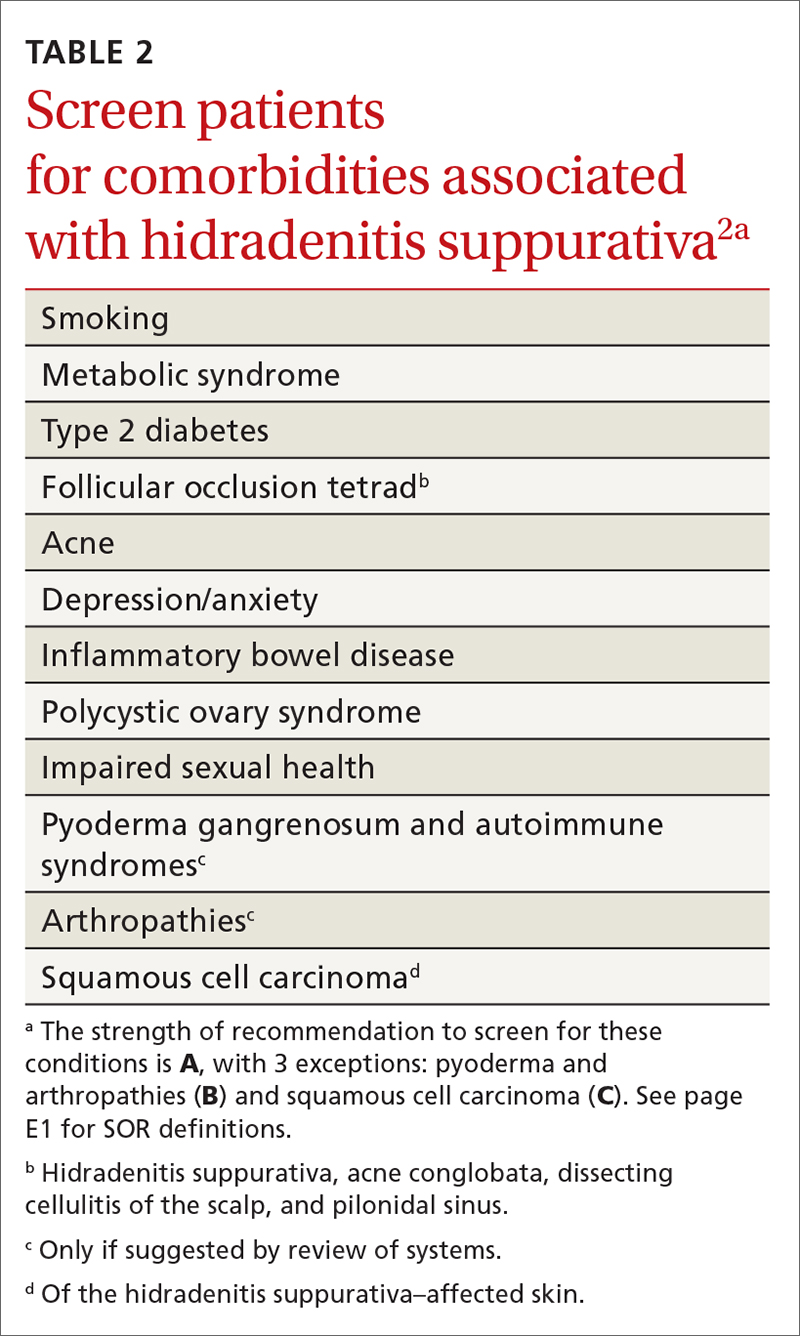
Screening for comorbidities
HSF recommends clinicians screen patients for comorbidities associated with HS (TABLE 2).2 Overall, screening patients for active and past history of smoking is strongly recommended, as is screening for metabolic syndrome, hyperlipidemia, type 2 diabetes (1.5- to 3-fold greater risk of type 2 diabetes in HS patients), and PCOS (3-fold greater risk).2,26,27,59 Screening patients for depression and anxiety is also routinely recommended.2 However, the authors of this article strongly recommend screening all patients with HS for psychiatric comorbidities, as research has shown a 2-fold greater risk of depression and anxiety, social isolation, and low self-esteem that severely limits quality of life (QOL) in this patient population.60,61
Management
Treat existing lesions, reduce formation of new ones
The main goals of treatment for patients with HS are to treat existing lesions and reduce associated symptoms, reduce the formation of new lesions, and minimize associated psychological morbidity.15 FPs play an important role in the early diagnosis, treatment, and comprehensive care of patients with HS. This includes monitoring patients, managing comorbidities, making appropriate referrals to dermatologists, and coordinating the multidisciplinary care that patients with HS require.
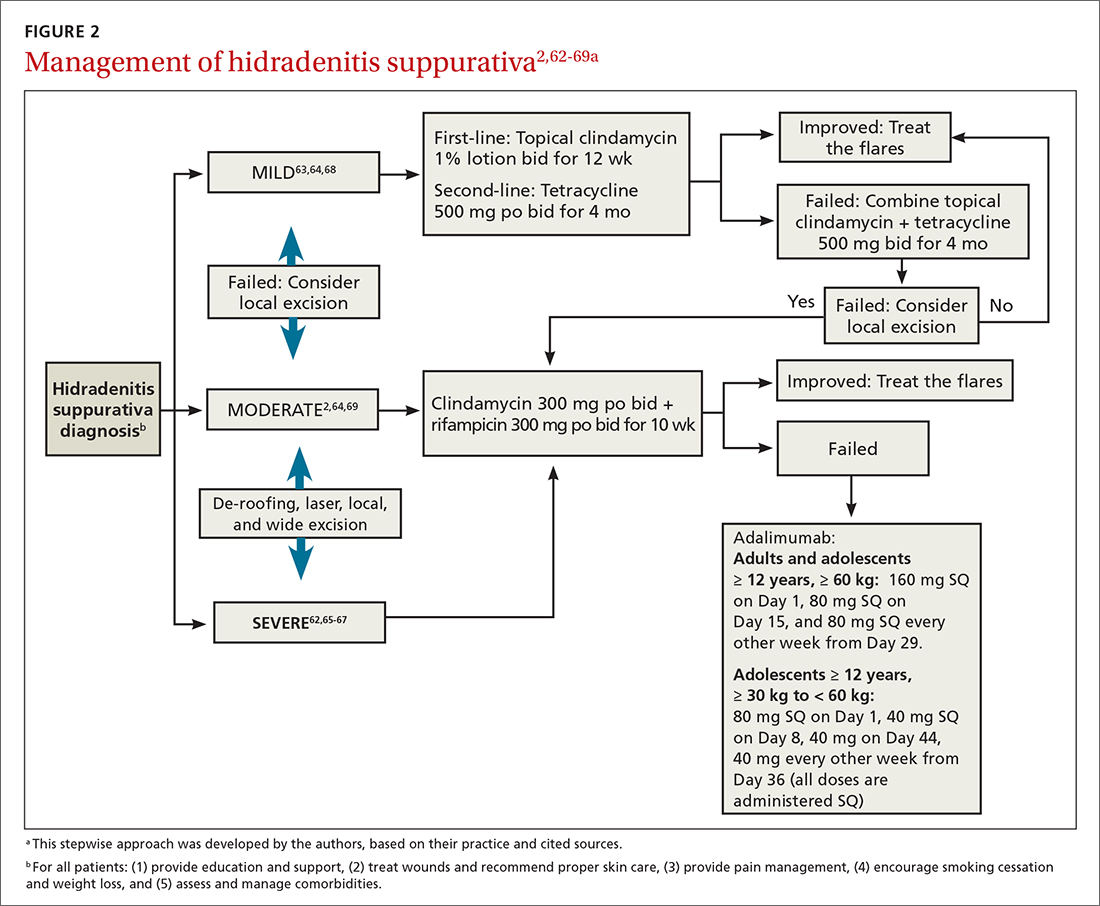
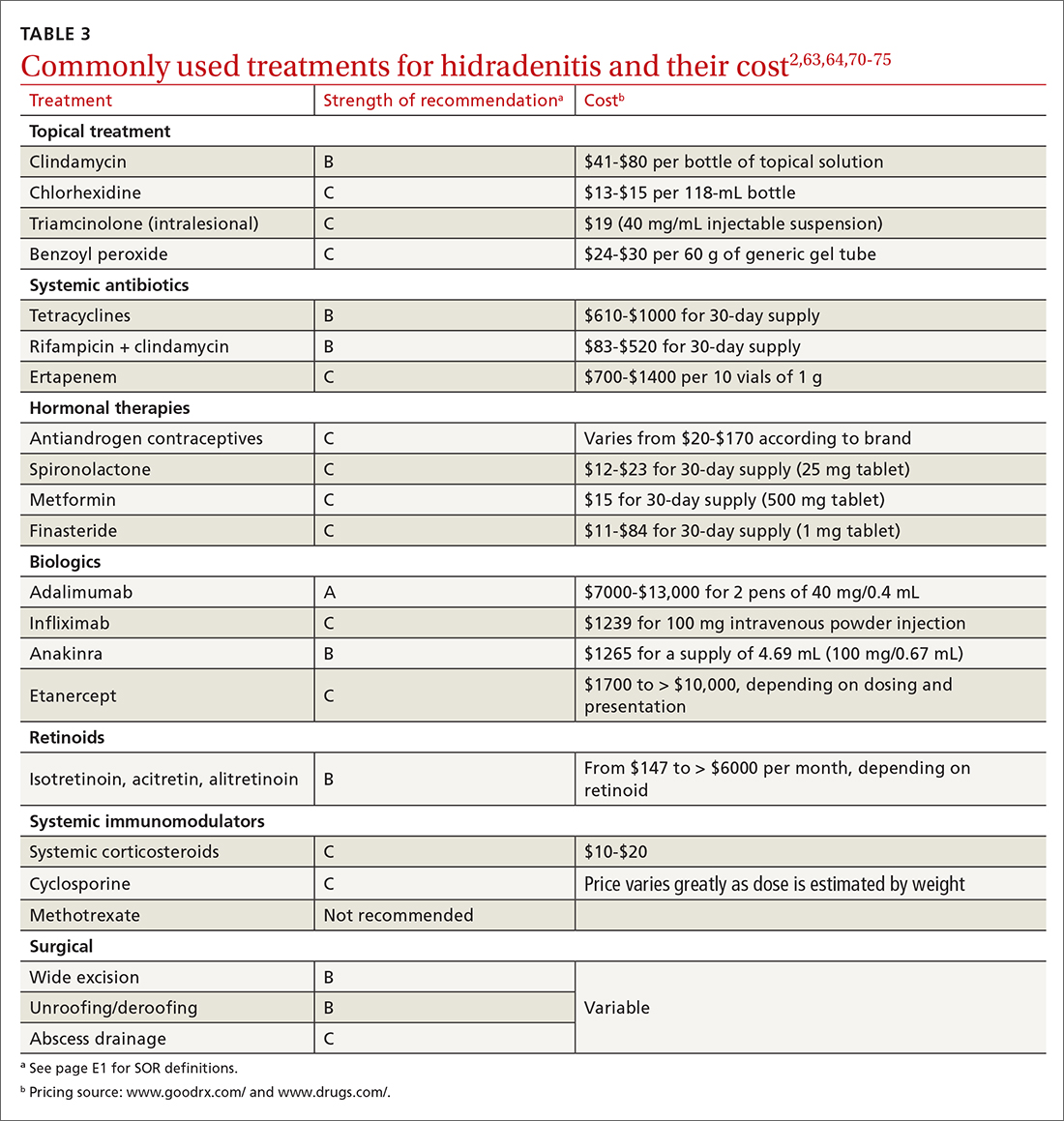
A systematic review identified more than 50 interventions used to treat HS, most based on small observational studies and randomized controlled trials (RCTs) with a high risk of bias.62 FIGURE 22,62-69 provides an evidence-based treatment algorithm for HS, and TABLE 32,63,64,70-75 summarizes the most commonly used treatments.
Biologic agents
Adalimumab (ADA) is a fully human immunoglobulin G1 monoclonal antibody that binds to TNF-alpha, neutralizes its bioactivity, and induces apoptosis of TNF-expressing mononuclear cells. It is the only medication approved by the US Food and Drug Administration for active refractory moderate and severe HS.62,65 Several double-blinded RCTs, including PIONEER I and PIONEER II, studied the effectiveness of ADA for HS and found significant clinical responses at Week 12, 50% reduction in abscess and nodule counts, no increase in abscesses or draining fistulas at Week 12, and sustained improvement in lesion counts, pain, and QOL.66,67,76
IL-1 and IL-23 inhibitors. The efficacy of etanercept and golimumab (anti-TNF), as well as anakinra (IL-1 inhibitor) and ustekinumab (IL-1/IL-23 inhibitor), continue to be investigated with variable results; they are considered second-line treatment for active refractory moderate and severe HS after ADA.65,77-80 Infliximab (IL-1 beta inhibitor) has shown no effect on reducing disease severity.70Compared to other treatments, biologic therapy is associated with higher costs (TABLE 3),2,63,64,70-75 an increased risk for reactivation of latent infections (eg, tuberculosis, herpes simplex, and hepatitis C virus [HCV], and B [HBV]), and an attenuated response to vaccines.81 Prior to starting biologic therapy, FPs should screen patients with HS for tuberculosis and HBV, consider HIV and HCV screening in at-risk patients, and optimize the immunization status of the patient.82,83 While inactivated vaccines can be administered without discontinuing biologic treatment, patients should avoid live-attenuated vaccines while taking biologics.83
Continue to: Antibiotic therapy
Antibiotic therapy
Topical antibiotics are considered first-line treatment for mild and moderate uncomplicated HS.63,64 Clindamycin 1%, the only topical antibiotic studied in a small double-blind RCT of patients with Hurley stage I and stage II HS, demonstrated significant clinical improvement after 12 weeks of treatment (twice- daily application), compared to placebo.84 Topical clindamycin is also recommended to treat flares in patients with mild disease.2,64
Oral antibiotics. Tetracycline (500 mg twice daily for 4 months) is considered a second-line treatment for patients with mild HS.64,68 Doxycycline (200 mg/d for 3 months) may also be considered as a second-line treatment in patients with mild disease.85
Combination oral clindamycin (300 mg) and rifampicin (300 mg) twice daily for 10 weeks is recommended as first-line treatment for patients with moderate HS.2,64,69 Combination rifampin (300 mg twice daily), moxifloxacin (400 mg/d), and metronidazole (500 mg three times a day) is not routinely recommended due to increased risk of toxicity.2
Ertapenem (1 g intravenously daily for 6 weeks) is supported by lower-level evidence as a third-line rescue therapy option and as a bridge to surgery; however, limitations for home infusions, costs, and concerns for antibiotic resistance limit its use.2,86
Corticosteroids and systemic immunomodulators
Intralesional triamcinolone (2-20 mg) may be beneficial in the early stages of HS, although its use is based on a small prospective open study of 33 patients.87 A recent double-blind placebo-controlled RCT comparing varying concentrations of intralesional triamcinolone (10 mg/mL and 40 mg/mL) vs normal saline showed no statistically significant difference in inflammatory clearance, pain reduction, or patient satisfaction.88
Continue to: Short-term systemic corticosteroid tapers...
Short-term systemic corticosteroid tapers (eg, prednisone, starting at 0.5-1 mg/kg) are recommended to treat flares. Long-term corticosteroids and cyclosporine are reserved for patients with severe refractory disease; however, due to safety concerns, their regular use is strongly discouraged.63,64,85 There is limited evidence to support the use of methotrexate for severe refractory disease, and its use is not recommended.63
Hormonal therapy
The use of hormonal therapy for HS is limited by the low-quality evidence (eg, anecdotal evidence, small retrospective analyses, uncontrolled trials).33,63 The only exception is a small double-blind controlled crossover trial from 1986 showing that the antiandrogen effects of combination oral contraceptives (ethinyloestradiol 50 mcg/cyproterone acetate in a reverse sequential regimen and ethinyloestradiol 50 mcg/norgestrel 500 mcg) improved HS lesions.89
Spironolactone, an antiandrogen diuretic, has been studied in small case report series with a high risk for bias. It is used mainly in female patients with mild or moderate disease, or in combination with other agents in patients with severe HS. Further research is needed to determine its utility in the treatment of HS.63,90,91
Metformin, alone or in combination with other therapies (dapsone, finasteride, liraglutide), has been analyzed in small prospective studies of primarily female patients with different severities of HS, obesity, and PCOS. These studies have shown improvement in lesions, QOL, and reduction of workdays lost.92,93
Finasteride. Studies have shown finasteride (1.25-5 mg/d) alone or in combination with other treatments (metformin, liraglutide, levonorgestrel-ethinyl estradiol, and dapsone) provided varying degrees of resolution or improvement in patients with severe and advanced HS. Finasteride has been used for 4 to 16 weeks with a good safety profile.92,94-96
Continue to: Retinoids
Retinoids
Acitretin, alitretinoin, and isotretinoin have been studied in small retrospective studies to manage HS, with variable results.97-99 Robust prospective studies are needed. Retinoids, in general, should be considered as a second- or third-line treatment for moderate to severe HS.63
Surgical intervention
Surgical interventions, which should be considered in patients with widespread mild, moderate, or severe disease, are associated with improved daily activity and work productivity.100 Incision and drainage should be avoided in patients with HS, as this technique does not remove the affected follicles and is associated with 100% recurrence.101
Wide excision is the preferred surgical technique for patients with Hurley stage II and stage III HS; it is associated with lower recurrence rates (13%) compared to local excision (22%) and deroofing (27%).102 Secondary intention healing is the most commonly chosen method, based on lower recurrence rates than primary closure.102
STEEP and laser techniques. The skin-tissue-sparing excision with electrosurgical peeling (STEEP) procedure involves successive tangential excision of affected tissue until the epithelized bottom of the sinus tracts has been reached. This allows for the removal of fibrotic tissue and the sparing of the deep subcutaneous fat. STEEP is associated with 30% of relapses after 43 months.71
Laser surgery has also been studied in patients with Hurley stage II and stage III HS. The most commonly used lasers for HS are the 1064-nm neodymium-doped yttrium aluminum garnet (Nd: YAG) and the carbon dioxide laser; they have been shown to reduce disease severity in inguinal, axillary, and inflammatory sites.72-74
Pain management: Start with lidocaine, NSAIDs
There are few studies about HS-associated pain management.103 For acute episodes, short-acting nonopioid local treatment with lidocaine, topical or oral nonsteroidal anti-inflammatory drugs, and acetaminophen are preferred. Opioids should be reserved for moderate-to-severe pain that has not responded to other analgesics. Adjuvant therapy with pregabalin, gabapentin, selective serotonin reuptake inhibitors, or serotonin-norepinephrine reuptake inhibitors can also be considered for the comanagement of pain and depression.62,104
Consider this tool to measure treatment response
The HS clinical response (HiSCR) tool is an outcome measure used to evaluate treatment outcomes. The tool uses an HS-specific binary score with the following criteria:
- ≥ 50% reduction in the number of inflammatory nodules;
- no increase in the number of abscesses; and
- no increase in the number of draining fistulas.105
The HiSCR was developed for the PIONEER studies105,106 to assess the response to ADA treatment. It is the only HS scoring system to undergo an extensive validation process with a meaningful clinical endpoint for HS treatment evaluation that is easy to use. Compared to the HS-PGA score (clear, minimal, mild), HiSCR was more responsive to change in patients with HS.105,106
CORRESPONDENCE
Cristina Marti-Amarista, MD, 101 Nicolls Road, Stony Brook, NY, 11794-8228; marti.amarista@gmail.com
1. Bergler-Czop B, Hadasik K, Brzezińska-Wcisło L. Acne inversa: difficulties in diagnostics and therapy. Postepy Dermatol Alergol. 2015;32:296-301. doi: 10.5114/pdia.2014.44012
2. Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part I: diagnosis, evaluation, and the use of complementary and procedural management. J Am Acad Dermatol. 2019;81:76-90. doi: 10.1016/j.jaad.2019.02.067
3. Fimmel S, Zouboulis CC. Comorbidities of hidradenitis suppurativa (acne inversa). Dermatoendocrinol. 2010;2:9-16. doi: 10.4161/derm.2.1.12490
4. Kokolakis G, Wolk K, Schneider-Burrus S, et al. Delayed diagnosis of hidradenitis suppurativa and its effect on patients and healthcare system. Dermatology. 2020;236:421-430. doi: 10.1159/000508787
5. Gulliver W, Landells IDR, Morgan D, et al. Hidradenitis suppurativa: a novel model of care and an integrative strategy to adopt an orphan disease. J Cutan Med Surg. 2018;22:71-77. doi: 10.1177/1203475417736290
6. Savage KT, Gonzalez Brant E, Flood KS, et al. Publication trends in hidradenitis suppurativa from 2008 to 2018. J Eur Acad Dermatol Venereol. 2020;34:1885-1889. doi: 10.1111/jdv.16213.
7. Narla S, Lyons AB, Hamzavi IH. The most recent advances in understanding and managing hidradenitis suppurativa. F1000Res. 2020;9:F1000. doi: 10.12688/f1000research.26083.1
8. Vossen ARJV, van der Zee HH, Prens EP. Hidradenitis suppurativa: a systematic review integrating inflammatory pathways into a cohesive pathogenic model. Front Immunol. 2018;9:2965. doi: 10.3389/fimmu.2018.02965
9. Frew JW, Hawkes JE, Krueger JG. A systematic review and critical evaluation of inflammatory cytokine associations in hidradenitis suppurativa. F1000Res. 2018;7:1930. doi: 10.12688/f1000 research.17267.1
10. Sabat R, Jemec GBE, Matusiak Ł. Hidradenitis suppurativa. Nat Rev Dis Primers. 2020;6:18. doi: 10.1038/s41572-020-0149-1
11. Shlyankevich J, Chen AJ, Kim GE, et al. Hidradenitis suppurativa is a systemic disease with substantial comorbidity burden: a chart-verified case-control analysis. J Am Acad Dermatol. 2014;71:1144-1150. doi: 10.1016/j.jaad.2014.09.012
12. Sartorius K, Emtestam L, Jemec GB, et al. Objective scoring of hidradenitis suppurativa reflecting the role of tobacco smoking and obesity. Br J Dermatol. 2009;161:831-839. doi: 10.1111/j.1365-2133.2009.09198.x
13. von Laffert M, Helmbold P, Wohlrab J, et al. Hidradenitis suppurativa (acne inversa): early inflammatory events at terminal follicles and at interfollicular epidermis. Exp Dermatol. 2010;19:533-537. doi: 10.1111/j.1600-0625.2009.00915.x
14. Jemec GB, Hansen U. Histology of hidradenitis suppurativa. J Am Acad Dermatol. 1996;34:994-999. doi: 10.1016/s0190-9622(96)90277-7
15. Ballard K, Shuman VL. Hidradenitis suppurativa. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021. Updated July 15, 2022. Accessed November 28, 2022. www.ncbi.nlm.nih.gov/books/NBK534867/
16. Garg A, Kirby JS, Lavian J, et al. Sex- and age-adjusted population analysis of prevalence estimates for hidradenitis suppurativa in the United States. JAMA Dermatol. 2017;153:760-764. doi: 10.1001/jamadermatol.2017.0201
17. Phan K, Charlton O, Smith SD. Global prevalence of hidradenitis suppurativa and geographical variation—systematic review and meta-analysis. BioMed Dermatol. 2020;4. doi: 10.1186/s41702-019-0052-0
18. Vazquez BG, Alikhan A, Weaver AL, et al. Incidence of hidradenitis suppurativa and associated factors: a population-based study of Olmsted County, Minnesota. J Invest Dermatol. 2013;133:97-103. doi: 10.1038/jid.2012.255
19. Sachdeva M, Shah M, Alavi A. Race-specific prevalence of hidradenitis suppurativa. J Cutan Med Surg. 2021;25:177-187. doi: 10.1177/1203475420972348
20. Vaidya T, Vangipuram R, Alikhan A. Examining the race-specific prevalence of hidradenitis suppurativa at a large academic center; results from a retrospective chart review. Dermatol Online J. 2017;23:13030/qt9xc0n0z1. doi: 10.5070/D3236035391
21. Price KN, Hsiao JL, Shi VY. Race and ethnicity gaps in global hidradenitis suppurativa clinical trials. Dermatology. 2021;237:97-102. doi: 10.1159/000504911
22. Schrader AM, Deckers IE, van der Zee HH, et al. Hidradenitis suppurativa: a retrospective study of 846 Dutch patients to identify factors associated with disease severity. J Am Acad Dermatol. 2014;71:460-467. doi: 10.1016/j.jaad.2014.04.001
23. Frew JW, Vekic DA, Wood J, et al. A systematic review and critical evaluation of reported pathogenic sequence variants in hidradenitis suppurativa. Br J Dermatol. 2017;177:987-998. doi: 10.1111/bjd.15441
24. Wolk K, Join-Lambert O, Sabat R. Aetiology and pathogenesis of hidradenitis suppurativa. Br J Dermatol. 2020;183:999-1010. doi: 10.1111/bjd.19556
25. Goldburg SR, Strober BE, Payette MJ. Hidradenitis suppurativa: epidemiology, clinical presentation, and pathogenesis. J Am Acad Dermatol. 2020;82:1045-1058. doi: 10.1016/j.jaad.2019.08.090
26. Sabat R, Chanwangpong A, Schneider-Burrus S, et al. Increased prevalence of metabolic syndrome in patients with acne inversa. PloS One. 2012;7:e31810. doi: 10.1371/journal.pone.0031810
27. Loh TY, Hendricks AJ, Hsiao JL, et al. Undergarment and fabric selection in the management of hidradenitis suppurativa. Dermatology. 2021;237:119-124. doi: 10.1159/000501611
28. Rodríguez-Zuñiga MJM, García-Perdomo HA, Ortega-Loayza AG. Association between hidradenitis suppurativa and metabolic syndrome: a systematic review and meta-analysis. Actas Dermosifiliogr (Engl Ed). 2019;110:279-288. doi: 10.1016/j.ad.2018.10.020
29. Walker JM, Garcet S, Aleman JO, et al. Obesity and ethnicity alter gene expression in skin. Sci Rep. 2020;10:14079. doi: 10.1038/s41598-020-70244-2.
30. Boer J, Nazary M, Riis PT. The role of mechanical stress in hidradenitis suppurativa. Dermatol Clin. 2016;34:37-43. doi: 10.1016/j.det.2015.08.011
31. Vossen ARJV, van Straalen KR, Swolfs EFH, et al. Nicotine dependency and readiness to quit smoking among patients with hidradenitis suppurativa. Dermatology. 2021;237:383-385. doi: 10.1159/000514028
32. Kromann CB, Deckers IE, Esmann S, et al. Risk factors, clinical course and long-term prognosis in hidradenitis suppurativa: a cross-sectional study. Br J Dermatol. 2014;171:819-824. doi: 10.1111/bjd.13090
33. Clark AK, Quinonez RL, Saric S, et al. Hormonal therapies for hidradenitis suppurativa: review. Dermatol Online J. 2017;23:13030/qt6383k0n4. doi: 10.5070/D32310036990
34. Saric-Bosanac S, Clark AK, Sivamani RK, et al. The role of hypothalamus-pituitary-adrenal (HPA)-like axis in inflammatory pilosebaceous disorders. Dermatol Online J. 2020;26:13030/qt8949296f. doi: 10.5070/D3262047430
35. Riis PT, Ring HC, Themstrup L, et al. The role of androgens and estrogens in hidradenitis suppurativa – a systematic review. Acta Dermatovenerol Croat. 2016;24:239-249.
36. Hsiao JL, Antaya RJ, Berger T, et al. Hidradenitis suppurativa and concomitant pyoderma gangrenosum: a case series and literature review. Arch Dermatol. 2010;146:1265-1270. doi: 10.1001/archdermatol.2010.328
37. Ah-Weng A, Langtry JAA, Velangi S, et al. Pyoderma gangrenosum associated with hidradenitis suppurativa. Clin Exp Dermatol. 2005;30:669-671. doi: 10.1111/j.1365-2230.2005.01897.x
38. Kirthi S, Hellen R, O’Connor R, et al. Hidradenitis suppurativa and Crohn’s disease: a case series. Ir Med J. 2017;110:618.
39. Dumont LM, Landman C, Sokol H, et al; CD-HS Study Group. Increased risk of permanent stoma in Crohn’s disease associated with hidradenitis suppurativa: a case-control study. Aliment Pharmacol Ther. 2020;52:303-310. doi: 10.1111/apt.15863
40. Marzano AV, Ceccherini I, Gattorno M, et al. Association of pyoderma gangrenosum, acne, and suppurative hidradenitis (PASH) shares genetic and cytokine profiles with other autoinflammatory diseases. Medicine (Baltimore). 2014;93:e187. doi: 10.1097/MD.0000000000000187.
41. Vinkel C, Thomsen SF. Hidradenitis suppurativa: causes, features, and current treatments. J Clin Aesthet Dermatol. 2018;11:17-23.
42. Wipperman J, Bragg DA, Litzner B. Hidradenitis suppurativa: rapid evidence review. Am Fam Physician. 2019;100:562-569.
43. Theut Riis P, Pedersen OB, Sigsgaard V, et al. Prevalence of patients with self-reported hidradenitis suppurativa in a cohort of Danish blood donors: a cross-sectional study. Br J Dermatol. 2019;180:774-781. doi: 10.1111/bjd.16998.
44. Dufour DN, Emtestam L, Jemec GB. Hidradenitis suppurativa: a common and burdensome, yet under-recognised, inflammatory skin disease. Postgrad Med J. 2014;90:216-221; doi: 10.1136/postgradmedj-2013-131994
45. Vinding GR, Miller IM, Zarchi K, et al. The prevalence of inverse recurrent suppuration: a population-based study of possible hidradenitis suppurativa. Br J Dermatol. 2014;170:884-889. doi: 10.1111/bjd.12787
46. Bassas-Vila J, González Lama Y. Hidradenitis suppurativa and perianal Crohn disease: differential diagnosis. Actas Dermosifiliogr. 2016;107(suppl 2):27-31. doi: 10.1016/S0001-7310(17) 30006-6
47. Valour F, Sénéchal A, Dupieux C, et al. Actinomycosis: etiology, clinical features, diagnosis, treatment, and management. Infect Drug Resist. 2014;7:183-197. doi: 10.2147/IDR.S39601
48. Fuchs W, Brockmeyer NH. Sexually transmitted infections. J Dtsch Dermatol Ges. 2014;12:451-463. doi: 10.1111/ddg.12310
49. Hap W, Frejlich E, Rudno-Rudzińska J, et al. Pilonidal sinus: finding the righttrack for treatment. Pol Przegl Chir. 2017;89:68-75. doi: 10.5604/01.3001.0009.6009
50. Al-Hamdi KI, Saadoon AQ. Acne onglobate of the scalp. Int J Trichology. 2020;12:35-37. doi: 10.4103/ijt.ijt_117_19
51. Balestra A, Bytyci H, Guillod C, et al. A case of ulceroglandular tularemia presenting with lymphadenopathy and an ulcer on a linear morphoea lesion surrounded by erysipelas. Int Med Case Rep J. 2018;11:313-318. doi: 10.2147/IMCRJ.S178561
52. Ibler KS, Kromann CB. Recurrent furunculosis – challenges and management: a review. Clin Cosmet Investig Dermatol. 2014;7:59-64. doi: 10.2147/CCID.S35302
53. Ingram JR, Hadjieconomou S, Piguet V. Development of core outcome sets in hidradenitis suppurativa: systematic review of outcome measure instruments to inform the process. Br J Dermatol. 2016;175:263-272. doi: 10.1111/bjd.14475
54. Zouboulis CC, Tzellos T, Kyrgidis A, et al; European Hidradenitis Suppurativa Foundation Investigator Group. Development and validation of the International Hidradenitis Suppurativa Severity Score System (I4), a novel dynamic scoring system to assess HS severity. Br J Dermatol. 2017;177:1401-1409. doi: 10.1111/bjd.15748
55. Hidradenitis Suppurativa Clinical Resource. Hidradenitis suppurativa stages: Hurley Staging System. www.hsdiseasesource.com/hs-disease-staging. Accessed October 11, 2022.
56. Ovadja ZN, Schuit MM, van der Horst CMAM, et al. Inter- and interrater reliability of Hurley staging for hidradenitis suppurativa. Br J Dermatol. 2019;181:344-349. doi: 10.1111/bjd.17588
57. Wortsman X, Jemec GBE. Real-time compound imaging ultrasound of hidradenitis suppurativa. Dermatol Surg. 2007;33:1340-1342. doi: 10.1111/j.1524-4725.2007.33286.x
58. Napolitano M, Calzavara-Pinton PG, Zanca A, et al. Comparison of clinical and ultrasound scores in patients with hidradenitis suppurativa: results from an Italian ultrasound working group. J Eur Acad Dermatol Venereol. 2019;33:e84-e87. doi: 10.1111/jdv.15235
59. Bukvić Mokos Z, Miše J, Balić A, et al. Understanding the relationship between smoking and hidradenitis suppurativa. Acta Dermatovenerol Croat. 2020;28:9-13.
60. Shavit E, Dreiher J, Freud T, et al. Psychiatric comorbidities in 3207 patients with hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2015;29:371-376. doi: 10.1111/jdv.12567
61. Kouris A, Platsidaki E, Christodoulou C, et al. Quality of life and psychosocial implications in patients with hidradenitis suppurativa. Dermatology. 2016;232:687-691. doi: 10.1159/000453355
62 Ingram JR, Woo PN, Chua SL, et al. Interventions for hidradenitis suppurativa: a Cochrane systematic review incorporating GRADE assessment of evidence quality. Br J Dermatol. 2016;174:970-978. doi: 10.1111/bjd.14418
63. Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part II: topical, intralesional, and systemic medical management. J Am Acad Dermatol. 2019;81:91-101. doi: 10.1016/j.jaad.2019.02.068
64. Gulliver W, Zouboulis CC, Prens E, et al. Evidence-based approach to the treatment of hidradenitis suppurativa/acne inversa, based on the European guidelines for hidradenitis suppurativa. Rev Endocr Metab Disord. 2016;17:343-351. doi: 10.1007/s11154-016-9328-5
65. Vena GA, Cassano N. Drug focus: adalimumab in the treatment of moderate to severe psoriasis. Biologics. 2007;1:93-103.
66. Kimball AB, Kerdel F, Adams D, et al. Adalimumab for the treatment of moderate to severe hidradenitis suppurativa: a parallel randomized trial. Ann Intern Med. 2012;157:846-55. doi: 10.7326/0003-4819-157-12-201212180-00004
67. Zouboulis CC, Okun MM, Prens EP, et al. Long-term adalimumab efficacy in patients with moderate-to-severe hidradenitis suppurativa/acne inversa: 3-year results of a phase 3 open-label extension study. J Am Acad Dermatol. 2019;80:60-69.e2. doi: 10.1016/j.jaad.2018.05.040
68. Jemec GB, Wendelboe P. Topical clindamycin versus systemic tetracycline in the treatment of hidradenitis suppurativa. J Am Acad Dermatol. 1998;39:971-974. doi: 10.1016/s0190-9622(98)70272-5
69. Gener G, Canoui-Poitrine F, Revuz JE, et al. Combination therapy with clindamycin and rifampicin for hidradenitis suppurativa: a series of 116 consecutive patients. Dermatology. 2009;219:148-154. doi: 10.1159/000228334
70. Grant A, Gonzalez T, Montgomery MO, et al. Infliximab therapy for patients with moderate to severe hidradenitis suppurativa: a randomized, double-blind, placebo-controlled crossover trial. J Am Acad Dermatol. 2010;62:205-217. doi: 10.1016/j.jaad.2009.06.050
71. Blok JL, Spoo JR, Leeman FWJ, et al. Skin-tissue-sparing excision with electrosurgical peeling (STEEP): a surgical treatment option for severe hidradenitis suppurativa Hurley stage II/III. J Eur Acad Dermatol Venereol. 2015;29:379-382. doi: 10.1111/jdv.12376
72. Mahmoud BH, Tierney E, Hexsel CL, et al. Prospective controlled clinical and histopathologic study of hidradenitis suppurativa treated with the long-pulsed neodymium:yttrium-aluminium-garnet laser. J Am Acad Dermatol. 2010;62:637-645. doi: 10.1016/j.jaad.2009.07.048
73. Tierney E, Mahmoud BH, Hexsel C, et al. Randomized control trial for the treatment of hidradenitis suppurativa with a neodymium-doped yttrium aluminium garnet laser. Dermatol Surg. 2009;35:1188-1198. doi: 10.1111/j.1524-4725.2009.01214.x
74. Hazen PG, Hazen BP. Hidradenitis suppurativa: successful treatment using carbon dioxide laser excision and marsupialization. Dermatol Surg. 2010;36:208-213. doi: 10.1111/j.1524-4725.2009.01427.x
75. van der Zee HH, Prens EP, Boer J. Deroofing: a tissue-saving surgical technique for the treatment of mild to moderate hidradenitis suppurativa lesions. J Am Acad Dermatol. 2010;63:475-480. doi: 10.1016/j.jaad.2009.12.018
76. Kimball AB, Okun MM, Williams DA, et al. Two phase 3 trials of adalimumab for hidradenitis suppurativa. N Engl J Med. 2016;375:422-434. doi: 10.1056/NEJMoa1504370. PMID: 27518661.
77. Adams DR, Yankura JA, Fogelberg AC, et al. Treatment of hidradenitis suppurativa with etanercept injection. Arch Dermatol. 2010;146:501-504. doi: 10.1001/archdermatol.2010.72
78. Tursi A. Concomitant hidradenitis suppurativa and pyostomatitis vegetans in silent ulcerative colitis successfully treated with golimumab. Dig Liver Dis. 2016;48:1511-1512. doi: 10.1016/j.dld.2016.09.010
79. Tzanetakou V, Kanni T, Giatrakou S, et al. Safety and efficacy of anakinra in severe hidradenitis suppurativa: a randomized clinical trial. JAMA Dermatol. 2016;152:52-59. doi: 10.1001/jamadermatol.2015.3903.
80. Romaní J, Vilarrasa E, Martorell A, et al. Ustekinumab with intravenous infusion: results in hidradenitis suppurativa. Dermatology. 2020;236:21-24. doi: 10.1159/000501075
81. Kane SV. Preparing for biologic or immunosuppressant therapy. Gastroenterol Hepatol (N Y). 2011;7:544-546.
82. Davis W, Vavilin I, Malhotra N. Biologic therapy in HIV: to screen or not to screen. Cureus. 2021;13:e15941. doi: 10.7759/cureus.15941
83. Papp KA, Haraoui B, Kumar D, et al. Vaccination guidelines for patients with immune-mediated disorders on immunosuppressive therapies. J Cutan Med Surg. 2019;23:50-74. doi: 10.1177/1203475418811335
84. Clemmensen OJ. Topical treatment of hidradenitis suppurativa with clindamycin. Int J Dermatol. 1983;22:325-328. doi: 10.1111/j.1365-4362.1983.tb02150.x
85. Hunger RE, Laffitte E, Läuchli S, et al. Swiss practice recommendations for the management of hidradenitis suppurativa/acne inversa. Dermatology. 2017;233:113-119. doi: 10.1159/000477459
86. Zouboulis CC, Bechara FG, Dickinson-Blok JL, et al. Hidradenitis suppurativa/acne inversa: a practical framework for treatment optimization - systematic review and recommendations from the HS ALLIANCE working group. J Eur Acad Dermatol Venereol. 2019;33:19-31. doi: 10.1111/jdv.15233
87. Riis PT, Boer J, Prens EP, et al. Intralesional triamcinolone for flares of hidradenitis suppurativa (HS): a case series. J Am Acad Dermatol. 2016;75:1151-1155. doi: 10.1016/j.jaad.2016.06.049
88. Fajgenbaum K, Crouse L, Dong L, et al. Intralesional triamcinolone may not be beneficial for treating acute hidradenitis suppurativa lesions: a double-blind, randomized, placebo-controlled trial. Dermatol Surg. 2020;46:685-689. doi: 10.1097/DSS.0000000000002112
89. Mortimer PS, Dawber RP, Gales MA, et al. A double-blind controlled cross-over trial of cyproterone acetate in females with hidradenitis suppurativa. Br J Dermatol. 1986;115:263-268. doi: 10.1111/j.1365-2133.1986.tb05740.x
90. Kraft JN, Searles GE. Hidradenitis suppurativa in 64 female patients: retrospective study comparing oral antibiotics and antiandrogen therapy. J Cutan Med Surg. 2007;11:125-131. doi: 10.2310/7750.2007.00019
91. Lee A, Fischer G. A case series of 20 women with hidradenitis suppurativa treated with spironolactone. Australas J Dermatol. 2015;56:192-196. doi: 10.1111/ajd.12362
92. Khandalavala BN. A disease-modifying approach for advanced hidradenitis suppurativa (regimen with metformin, liraglutide, dapsone, and finasteride): a case report. Case Rep Dermatol. 2017;9:70-78. doi: 10.1159/000473873
93. Verdolini R, Clayton N, Smith A, et al. Metformin for the treatment of hidradenitis suppurativa: a little help along the way. J Eur Acad Dermatol Venereol. 2013;27:1101-1108. doi: 10.1111/j.1468-3083.2012.04668.x
94. Khandalavala BN, Do MV. Finasteride in hidradenitis suppurativa: a “male” therapy for a predominantly “female” disease. J Clin Aesthet Dermatol. 2016;9:44-50.
95. Mota F, Machado S, Selores M. Hidradenitis suppurativa in children treated with finasteride-a case series. Pediatr Dermatol. 2017;34:578-583. doi: 10.1111/pde.13216
96. Doménech C, Matarredona J, Escribano-Stablé JC, et al. Facial hidradenitis suppurativa in a 28-year-old male responding to finasteride. Dermatology. 2012;224:307-308. doi: 10.1159/000339477
97. Patel N, McKenzie SA, Harview CL, et al. Isotretinoin in the treatment of hidradenitis suppurativa: a retrospective study. J Dermatolog Treat. 2021;32:473-475. doi: 10.1080/09546634.2019.1670779
98. Boer J, van Gemert MJ. Long-term results of isotretinoin in the treatment of 68 patients with hidradenitis suppurativa. J Am Acad Dermatol. 1999;40:73-76. doi: 10.1016/s0190-9622(99) 70530-x
99. Huang CM, Kirchhof MG. A new perspective on isotretinoin treatment of hidradenitis suppurativa: a retrospective chart review of patient outcomes. Dermatology. 2017;233:120-125. doi: 10.1159/000477207
100. Prens LM, Huizinga J, Janse IC. Surgical outcomes and the impact of major surgery on quality of life, activity impairment and sexual health in hidradenitis suppurativa patients: a prospective single centre study. J Eur Acad Dermatol Venereol. 2019;33:1941-1946. doi: 10.1111/jdv.15706
101. Ritz JP, Runkel N, Haier J, et al. Extent of surgery and recurrence rate of hidradenitis suppurativa. Int J Colorectal Dis. 1998;13:164-168. doi: 10.1007/s003840050159
102. Mehdizadeh A, Hazen PG, Bechara FG, et al. Recurrence of hidradenitis suppurativa after surgical management: a systematic review and meta-analysis. J Am Acad Dermatol. 2015;73(5 suppl 1):S70-S77. doi: 10.1016/j.jaad.2015.07.044.
103. Smith HS, Chao JD, Teitelbaum J. Painful hidradenitis suppurativa. Clin J Pain. 2010;26:435-444. doi: 10.1097/AJP.0b013e3181ceb80c
104. Horváth B, Janse IC, Sibbald GR. Pain management in patients with hidradenitis suppurativa. J Am Acad Dermatol. 2015;73(5 suppl 1):S47-S51. doi: 10.1016/j.jaad.2015.07.046
105. Kimball AB, Sobell JM, Zouboulis CC, et al. HiSCR (Hidradenitis Suppurativa Clinical Response): a novel clinical endpoint to evaluate therapeutic outcomes in patients with hidradenitis suppurativa from the placebo-controlled portion of a phase 2 adalimumab study. J Eur Acad Dermatol Venereol. 2016;30:989-994. doi: 10.1111/jdv.13216
106. Kimball AB, Jemec GB, Yang M, et al. Assessing the validity, responsiveness and meaningfulness of the Hidradenitis Suppurativa Clinical Response (HiSCR) as the clinical endpoint for hidradenitis suppurativa treatment. Br J Dermatol. 2014;171:1434-1442. doi: 10.1111/bjd.13270
Hidradenitis suppurativa (HS), also known as acne inversa or Verneuil disease, is a chronic, recurrent, inflammatory occlusive disease affecting the terminal follicular epithelium in apocrine gland–bearing skin areas.1 HS manifests as painful nodules, abscesses, fistulas, and scarring and often has a severe psychological impact on the affected patient.2
When HS was first identified in the 1800s, it was believed to result from a dysfunction of the sweat glands.3 In 1939, scientists identified the true cause: follicular occlusion.3
Due to its chronic nature, heterogeneity in presentation, and apparent low prevalence,4 HS is considered an orphan disease.5 Over the past 10 years, there has been a surge in HS research—particularly in medical management—which has provided a better understanding of this condition.6,7
In this review, we discuss the most updated evidence regarding the diagnosis and treatment of HS to guide the family physician (FP)’s approach to managing this debilitating disease. But first, we offer a word about the etiology and pathophysiology of the condition.
3 events set the stage for hidradenitis suppurativa
Although the exact cause of HS is still unknown, some researchers have hypothesized that HS results from a combination of genetic predisposition and environmental and lifestyle factors.8-12 The primary mechanism of HS is the obstruction of the terminal follicular epithelium by a keratin plug.1,13,14 A systematic review of molecular inflammatory pathways involved in HS divides the pathogenesis of HS into 3 events: follicular occlusion followed by dilation, follicular rupture and inflammatory response, and chronic inflammatory state with sinus tracts.8
An underreported condition
HS is often underreported and misdiagnosed.4,15 Globally, the prevalence of HS varies from < 1% to 4%.15,16 A systematic review with meta-analysis showed a higher prevalence of HS in females compared to males in American and European populations.17 In the United States, the overall frequency of HS is 0.1%, or 98 per 100,000 persons.16 The prevalence of HS is highest among patients ages 30 to 39 years; there is decreased prevalence in patients ages 55 years and older.16,18
Who is at heightened risk?
Recent research has shown a relationship between ethnicity and HS.16,19,20 African American and biracial groups (defined as African American and White) have a 3-fold and 2-fold greater prevalence of HS, respectively, compared to White patients.16 However, the prevalence of HS in non-White ethnic groups may be underestimated in clinical trials due to a lack of representation and subgroup analyses based on ethnicity, which may affect generalizability in HS recommendations.21
Continue to: Genetic predisposition
Genetic predisposition. As many as 40% of patients with HS report having at least 1 affected family member. A positive family history of HS is associated with earlier onset, longer disease duration, and severe disease.22 HS is genetically heterogeneous, and several mutations (eg, gamma secretase, PSTPIP1, PSEN1 genes) have been identified in patients and in vitro as the cause of dysregulation of epidermal proliferation and differentiation, immune dysregulation, and promotion of amyloid formation.8,23-25
Obesity and metabolic risk factors. There is a strong relationship between HS and obesity. As many as 70% of patients with HS are obese, and 9% to 40% have metabolic syndrome.12,18,26-28 Obesity is associated with maceration and mechanical stress, increased fragility of the dermo-epidermal junction, changes in cutaneous blood flow, and subdermal fat inflammation—all of which favor the pathophysiology of HS.29,30
Smoking. Tobacco smoking is associated with severe HS and a lower chance of remission.12 Population-based studies have shown that as many as 90% of patients with HS have a history of smoking ≥ 20 packs of cigarettes per year.1,12,18,31,32 The nicotine and thousands of other chemicals present in cigarettes trigger keratinocytes and fibroblasts, resulting in epidermal hyperplasia, infundibular hyperkeratosis, excessive cornification, and dysbiosis.8,23,24
Hormones. The exact role sex hormones play in the pathogenesis of HS remains unclear.8,32 Most information is based primarily on small studies looking at antiandrogen treatments, HS activity during the menstrual cycle and pregnancy, HS exacerbation related to androgenic effects of hormonal contraception, and the association of HS with metabolic-endocrine disorders (eg, polycystic ovary syndrome [PCOS]).8,33
Androgens induce hyperkeratosis that may lead to follicular occlusion—the hallmark of HS pathology.34 A systematic review looking at the role of androgen and estrogen in HS found that while some patients with HS have elevated androgen levels, most have androgen and estrogen levels within normal range.35 Therefore, increased peripheral androgen receptor sensitivity has been hypothesized as the mechanism of action contributing to HS manifestation.34
Continue to: Host-defense defects
Host-defense defects. HS shares a similar cytokine profile with other well-established immune-mediated inflammatory diseases, including pyoderma gangrenosum (PG)36,37 and Crohn disease.38-40 HS is characterized by the expression of several immune mediators, including tumor necrosis factor-alpha (TNF-alpha), interleukin-1 alpha (IL-1 alpha), IL-1 beta, IL-8, IL-17, and the IL-23/T helper 17 pathway, all of which are upregulated in other inflammatory diseases and also result in an abnormal innate immune response.8,24 The recently described clinical triad of PG, acne, and HS (PASH) and the tetrad of pyogenic arthritis, PG, acne, and HS (PAPASH) further support the role of immune dysregulation in the pathogenesis of HS.40 Nonetheless, further studies are needed to determine the exact pathways of cytokine effect in HS.41
Use these criteria to make the diagnosis
The US and Canadian Hidradenitis Suppurativa Foundations (HSF) guidelines base the clinical diagnosis of HS on the following criteria2:
- Typical HS lesions: Erythematous skin lesions; inflamed, deep-seated painful nodules; “tombstone” double-ended comedones; sinus tracts; scarring; deformity. FIGURES 1A-1E show typical lesions seen in patients with HS.
- Typical locations: Intertriginous regions—apocrine gland–containing areas in axilla, groin, perineal region, buttocks, gluteal cleft, and mammary folds; beltline and waistband areas; areas of skin compression and friction.
- Recurrence and chronicity: Recurrent painful or suppurating lesions that appear more than twice in a 6-month period.2,41-43

Patients with HS usually present with painful recurrent abscesses and scarring and often report multiple visits to the emergency department for drainage or failed antibiotic treatment for abscesses.15,44
Ask patients these 2 questions. Vinding et al45 developed a survey for the diagnosis of HS using 2 simple questions based on the 3 criteria established by the HSF:
- “Have you had an outbreak of boils during the last 6 months?” and
- “Where and how many boils have you had?” (This question includes a list of the typical HS locations—eg, axilla, groin, genitals, area under breast.)
In their questionnaire, Vinding et al45 found that an affirmative answer to Question 1 and reports of > 2 boils in response to Question 2 correlated to a sensitivity of 90%, specificity of 97%, positive predictive value of 96%, and negative predictive value of 92% for the diagnosis of HS. The differential diagnosis of HS is summarized in TABLE 1.42,45-52

Continue to: These tools can help you to stage hidradenitis suppurativa
These tools can help you to stage hidradenitis suppurativa
Multiple tools are available to assess the severity of HS.53 We will describe the Hurley staging system and the International Hidradenitis Suppurativa Severity Score System (IHS4). Other diagnostic tools, such as the Sartorius score and the Hidradenitis Suppurativa Physician’s Global Assessment Scale (HS-PGA), can be time-consuming and challenging to interpret, limiting their use in the clinical setting.2,54
Hurley staging system (available at www.hsdiseasesource.com/hs-disease-staging) considers the presence of nodules, abscesses, sinus tracts, and scarring affecting an entire anatomical area.13,55 This system is most useful as a rapid classification tool for patients with HS in the clinical setting but should not be used to assess clinical response.2,13,56
The IHS4 (available at https://online library.wiley.com/doi/10.1111/bjd.15748) is a validated and easy-to-use tool for assessing HS and guiding the therapeutic strategy in clinical practice.54 With IHS4, the clinician must calculate the following:
- total number of nodules > 10 mm in diameter
- total number of abscesses multiplied by 2, and
- total number of draining tunnels (fistulae/sinuses) multiplied by 4.
Mild HS is defined as a score ≤ 3 points; moderate HS, 4 to 10 points; and severe HS, ≥ 11 points.54
No diagnostic tests, but ultrasound may be helpful
There are currently no established biological markers or specific tests for diagnosing HS.15 Ultrasound is emerging as a tool to assess dermal thickness, hair follicle morphology, and number and extent of fluid collections. Two recent studies showed that pairing clinical assessment with ultrasound findings improves accuracy of scoring in 84% of cases.57,58 For patients with severe HS, skin biopsy can be considered to rule out squamous cell carcinoma. Cultures, however, have limited utility except for suspected superimposed bacterial infection.2
Continue to: Screening for comorbidities
Screening for comorbidities
HSF recommends clinicians screen patients for comorbidities associated with HS (TABLE 2).2 Overall, screening patients for active and past history of smoking is strongly recommended, as is screening for metabolic syndrome, hyperlipidemia, type 2 diabetes (1.5- to 3-fold greater risk of type 2 diabetes in HS patients), and PCOS (3-fold greater risk).2,26,27,59 Screening patients for depression and anxiety is also routinely recommended.2 However, the authors of this article strongly recommend screening all patients with HS for psychiatric comorbidities, as research has shown a 2-fold greater risk of depression and anxiety, social isolation, and low self-esteem that severely limits quality of life (QOL) in this patient population.60,61

Management
Treat existing lesions, reduce formation of new ones
The main goals of treatment for patients with HS are to treat existing lesions and reduce associated symptoms, reduce the formation of new lesions, and minimize associated psychological morbidity.15 FPs play an important role in the early diagnosis, treatment, and comprehensive care of patients with HS. This includes monitoring patients, managing comorbidities, making appropriate referrals to dermatologists, and coordinating the multidisciplinary care that patients with HS require.

A systematic review identified more than 50 interventions used to treat HS, most based on small observational studies and randomized controlled trials (RCTs) with a high risk of bias.62 FIGURE 22,62-69 provides an evidence-based treatment algorithm for HS, and TABLE 32,63,64,70-75 summarizes the most commonly used treatments.

Biologic agents
Adalimumab (ADA) is a fully human immunoglobulin G1 monoclonal antibody that binds to TNF-alpha, neutralizes its bioactivity, and induces apoptosis of TNF-expressing mononuclear cells. It is the only medication approved by the US Food and Drug Administration for active refractory moderate and severe HS.62,65 Several double-blinded RCTs, including PIONEER I and PIONEER II, studied the effectiveness of ADA for HS and found significant clinical responses at Week 12, 50% reduction in abscess and nodule counts, no increase in abscesses or draining fistulas at Week 12, and sustained improvement in lesion counts, pain, and QOL.66,67,76
IL-1 and IL-23 inhibitors. The efficacy of etanercept and golimumab (anti-TNF), as well as anakinra (IL-1 inhibitor) and ustekinumab (IL-1/IL-23 inhibitor), continue to be investigated with variable results; they are considered second-line treatment for active refractory moderate and severe HS after ADA.65,77-80 Infliximab (IL-1 beta inhibitor) has shown no effect on reducing disease severity.70Compared to other treatments, biologic therapy is associated with higher costs (TABLE 3),2,63,64,70-75 an increased risk for reactivation of latent infections (eg, tuberculosis, herpes simplex, and hepatitis C virus [HCV], and B [HBV]), and an attenuated response to vaccines.81 Prior to starting biologic therapy, FPs should screen patients with HS for tuberculosis and HBV, consider HIV and HCV screening in at-risk patients, and optimize the immunization status of the patient.82,83 While inactivated vaccines can be administered without discontinuing biologic treatment, patients should avoid live-attenuated vaccines while taking biologics.83
Continue to: Antibiotic therapy
Antibiotic therapy
Topical antibiotics are considered first-line treatment for mild and moderate uncomplicated HS.63,64 Clindamycin 1%, the only topical antibiotic studied in a small double-blind RCT of patients with Hurley stage I and stage II HS, demonstrated significant clinical improvement after 12 weeks of treatment (twice- daily application), compared to placebo.84 Topical clindamycin is also recommended to treat flares in patients with mild disease.2,64
Oral antibiotics. Tetracycline (500 mg twice daily for 4 months) is considered a second-line treatment for patients with mild HS.64,68 Doxycycline (200 mg/d for 3 months) may also be considered as a second-line treatment in patients with mild disease.85
Combination oral clindamycin (300 mg) and rifampicin (300 mg) twice daily for 10 weeks is recommended as first-line treatment for patients with moderate HS.2,64,69 Combination rifampin (300 mg twice daily), moxifloxacin (400 mg/d), and metronidazole (500 mg three times a day) is not routinely recommended due to increased risk of toxicity.2
Ertapenem (1 g intravenously daily for 6 weeks) is supported by lower-level evidence as a third-line rescue therapy option and as a bridge to surgery; however, limitations for home infusions, costs, and concerns for antibiotic resistance limit its use.2,86
Corticosteroids and systemic immunomodulators
Intralesional triamcinolone (2-20 mg) may be beneficial in the early stages of HS, although its use is based on a small prospective open study of 33 patients.87 A recent double-blind placebo-controlled RCT comparing varying concentrations of intralesional triamcinolone (10 mg/mL and 40 mg/mL) vs normal saline showed no statistically significant difference in inflammatory clearance, pain reduction, or patient satisfaction.88
Continue to: Short-term systemic corticosteroid tapers...
Short-term systemic corticosteroid tapers (eg, prednisone, starting at 0.5-1 mg/kg) are recommended to treat flares. Long-term corticosteroids and cyclosporine are reserved for patients with severe refractory disease; however, due to safety concerns, their regular use is strongly discouraged.63,64,85 There is limited evidence to support the use of methotrexate for severe refractory disease, and its use is not recommended.63
Hormonal therapy
The use of hormonal therapy for HS is limited by the low-quality evidence (eg, anecdotal evidence, small retrospective analyses, uncontrolled trials).33,63 The only exception is a small double-blind controlled crossover trial from 1986 showing that the antiandrogen effects of combination oral contraceptives (ethinyloestradiol 50 mcg/cyproterone acetate in a reverse sequential regimen and ethinyloestradiol 50 mcg/norgestrel 500 mcg) improved HS lesions.89
Spironolactone, an antiandrogen diuretic, has been studied in small case report series with a high risk for bias. It is used mainly in female patients with mild or moderate disease, or in combination with other agents in patients with severe HS. Further research is needed to determine its utility in the treatment of HS.63,90,91
Metformin, alone or in combination with other therapies (dapsone, finasteride, liraglutide), has been analyzed in small prospective studies of primarily female patients with different severities of HS, obesity, and PCOS. These studies have shown improvement in lesions, QOL, and reduction of workdays lost.92,93
Finasteride. Studies have shown finasteride (1.25-5 mg/d) alone or in combination with other treatments (metformin, liraglutide, levonorgestrel-ethinyl estradiol, and dapsone) provided varying degrees of resolution or improvement in patients with severe and advanced HS. Finasteride has been used for 4 to 16 weeks with a good safety profile.92,94-96
Continue to: Retinoids
Retinoids
Acitretin, alitretinoin, and isotretinoin have been studied in small retrospective studies to manage HS, with variable results.97-99 Robust prospective studies are needed. Retinoids, in general, should be considered as a second- or third-line treatment for moderate to severe HS.63
Surgical intervention
Surgical interventions, which should be considered in patients with widespread mild, moderate, or severe disease, are associated with improved daily activity and work productivity.100 Incision and drainage should be avoided in patients with HS, as this technique does not remove the affected follicles and is associated with 100% recurrence.101
Wide excision is the preferred surgical technique for patients with Hurley stage II and stage III HS; it is associated with lower recurrence rates (13%) compared to local excision (22%) and deroofing (27%).102 Secondary intention healing is the most commonly chosen method, based on lower recurrence rates than primary closure.102
STEEP and laser techniques. The skin-tissue-sparing excision with electrosurgical peeling (STEEP) procedure involves successive tangential excision of affected tissue until the epithelized bottom of the sinus tracts has been reached. This allows for the removal of fibrotic tissue and the sparing of the deep subcutaneous fat. STEEP is associated with 30% of relapses after 43 months.71
Laser surgery has also been studied in patients with Hurley stage II and stage III HS. The most commonly used lasers for HS are the 1064-nm neodymium-doped yttrium aluminum garnet (Nd: YAG) and the carbon dioxide laser; they have been shown to reduce disease severity in inguinal, axillary, and inflammatory sites.72-74
Pain management: Start with lidocaine, NSAIDs
There are few studies about HS-associated pain management.103 For acute episodes, short-acting nonopioid local treatment with lidocaine, topical or oral nonsteroidal anti-inflammatory drugs, and acetaminophen are preferred. Opioids should be reserved for moderate-to-severe pain that has not responded to other analgesics. Adjuvant therapy with pregabalin, gabapentin, selective serotonin reuptake inhibitors, or serotonin-norepinephrine reuptake inhibitors can also be considered for the comanagement of pain and depression.62,104
Consider this tool to measure treatment response
The HS clinical response (HiSCR) tool is an outcome measure used to evaluate treatment outcomes. The tool uses an HS-specific binary score with the following criteria:
- ≥ 50% reduction in the number of inflammatory nodules;
- no increase in the number of abscesses; and
- no increase in the number of draining fistulas.105
The HiSCR was developed for the PIONEER studies105,106 to assess the response to ADA treatment. It is the only HS scoring system to undergo an extensive validation process with a meaningful clinical endpoint for HS treatment evaluation that is easy to use. Compared to the HS-PGA score (clear, minimal, mild), HiSCR was more responsive to change in patients with HS.105,106
CORRESPONDENCE
Cristina Marti-Amarista, MD, 101 Nicolls Road, Stony Brook, NY, 11794-8228; marti.amarista@gmail.com
Hidradenitis suppurativa (HS), also known as acne inversa or Verneuil disease, is a chronic, recurrent, inflammatory occlusive disease affecting the terminal follicular epithelium in apocrine gland–bearing skin areas.1 HS manifests as painful nodules, abscesses, fistulas, and scarring and often has a severe psychological impact on the affected patient.2
When HS was first identified in the 1800s, it was believed to result from a dysfunction of the sweat glands.3 In 1939, scientists identified the true cause: follicular occlusion.3
Due to its chronic nature, heterogeneity in presentation, and apparent low prevalence,4 HS is considered an orphan disease.5 Over the past 10 years, there has been a surge in HS research—particularly in medical management—which has provided a better understanding of this condition.6,7
In this review, we discuss the most updated evidence regarding the diagnosis and treatment of HS to guide the family physician (FP)’s approach to managing this debilitating disease. But first, we offer a word about the etiology and pathophysiology of the condition.
3 events set the stage for hidradenitis suppurativa
Although the exact cause of HS is still unknown, some researchers have hypothesized that HS results from a combination of genetic predisposition and environmental and lifestyle factors.8-12 The primary mechanism of HS is the obstruction of the terminal follicular epithelium by a keratin plug.1,13,14 A systematic review of molecular inflammatory pathways involved in HS divides the pathogenesis of HS into 3 events: follicular occlusion followed by dilation, follicular rupture and inflammatory response, and chronic inflammatory state with sinus tracts.8
An underreported condition
HS is often underreported and misdiagnosed.4,15 Globally, the prevalence of HS varies from < 1% to 4%.15,16 A systematic review with meta-analysis showed a higher prevalence of HS in females compared to males in American and European populations.17 In the United States, the overall frequency of HS is 0.1%, or 98 per 100,000 persons.16 The prevalence of HS is highest among patients ages 30 to 39 years; there is decreased prevalence in patients ages 55 years and older.16,18
Who is at heightened risk?
Recent research has shown a relationship between ethnicity and HS.16,19,20 African American and biracial groups (defined as African American and White) have a 3-fold and 2-fold greater prevalence of HS, respectively, compared to White patients.16 However, the prevalence of HS in non-White ethnic groups may be underestimated in clinical trials due to a lack of representation and subgroup analyses based on ethnicity, which may affect generalizability in HS recommendations.21
Continue to: Genetic predisposition
Genetic predisposition. As many as 40% of patients with HS report having at least 1 affected family member. A positive family history of HS is associated with earlier onset, longer disease duration, and severe disease.22 HS is genetically heterogeneous, and several mutations (eg, gamma secretase, PSTPIP1, PSEN1 genes) have been identified in patients and in vitro as the cause of dysregulation of epidermal proliferation and differentiation, immune dysregulation, and promotion of amyloid formation.8,23-25
Obesity and metabolic risk factors. There is a strong relationship between HS and obesity. As many as 70% of patients with HS are obese, and 9% to 40% have metabolic syndrome.12,18,26-28 Obesity is associated with maceration and mechanical stress, increased fragility of the dermo-epidermal junction, changes in cutaneous blood flow, and subdermal fat inflammation—all of which favor the pathophysiology of HS.29,30
Smoking. Tobacco smoking is associated with severe HS and a lower chance of remission.12 Population-based studies have shown that as many as 90% of patients with HS have a history of smoking ≥ 20 packs of cigarettes per year.1,12,18,31,32 The nicotine and thousands of other chemicals present in cigarettes trigger keratinocytes and fibroblasts, resulting in epidermal hyperplasia, infundibular hyperkeratosis, excessive cornification, and dysbiosis.8,23,24
Hormones. The exact role sex hormones play in the pathogenesis of HS remains unclear.8,32 Most information is based primarily on small studies looking at antiandrogen treatments, HS activity during the menstrual cycle and pregnancy, HS exacerbation related to androgenic effects of hormonal contraception, and the association of HS with metabolic-endocrine disorders (eg, polycystic ovary syndrome [PCOS]).8,33
Androgens induce hyperkeratosis that may lead to follicular occlusion—the hallmark of HS pathology.34 A systematic review looking at the role of androgen and estrogen in HS found that while some patients with HS have elevated androgen levels, most have androgen and estrogen levels within normal range.35 Therefore, increased peripheral androgen receptor sensitivity has been hypothesized as the mechanism of action contributing to HS manifestation.34
Continue to: Host-defense defects
Host-defense defects. HS shares a similar cytokine profile with other well-established immune-mediated inflammatory diseases, including pyoderma gangrenosum (PG)36,37 and Crohn disease.38-40 HS is characterized by the expression of several immune mediators, including tumor necrosis factor-alpha (TNF-alpha), interleukin-1 alpha (IL-1 alpha), IL-1 beta, IL-8, IL-17, and the IL-23/T helper 17 pathway, all of which are upregulated in other inflammatory diseases and also result in an abnormal innate immune response.8,24 The recently described clinical triad of PG, acne, and HS (PASH) and the tetrad of pyogenic arthritis, PG, acne, and HS (PAPASH) further support the role of immune dysregulation in the pathogenesis of HS.40 Nonetheless, further studies are needed to determine the exact pathways of cytokine effect in HS.41
Use these criteria to make the diagnosis
The US and Canadian Hidradenitis Suppurativa Foundations (HSF) guidelines base the clinical diagnosis of HS on the following criteria2:
- Typical HS lesions: Erythematous skin lesions; inflamed, deep-seated painful nodules; “tombstone” double-ended comedones; sinus tracts; scarring; deformity. FIGURES 1A-1E show typical lesions seen in patients with HS.
- Typical locations: Intertriginous regions—apocrine gland–containing areas in axilla, groin, perineal region, buttocks, gluteal cleft, and mammary folds; beltline and waistband areas; areas of skin compression and friction.
- Recurrence and chronicity: Recurrent painful or suppurating lesions that appear more than twice in a 6-month period.2,41-43

Patients with HS usually present with painful recurrent abscesses and scarring and often report multiple visits to the emergency department for drainage or failed antibiotic treatment for abscesses.15,44
Ask patients these 2 questions. Vinding et al45 developed a survey for the diagnosis of HS using 2 simple questions based on the 3 criteria established by the HSF:
- “Have you had an outbreak of boils during the last 6 months?” and
- “Where and how many boils have you had?” (This question includes a list of the typical HS locations—eg, axilla, groin, genitals, area under breast.)
In their questionnaire, Vinding et al45 found that an affirmative answer to Question 1 and reports of > 2 boils in response to Question 2 correlated to a sensitivity of 90%, specificity of 97%, positive predictive value of 96%, and negative predictive value of 92% for the diagnosis of HS. The differential diagnosis of HS is summarized in TABLE 1.42,45-52

Continue to: These tools can help you to stage hidradenitis suppurativa
These tools can help you to stage hidradenitis suppurativa
Multiple tools are available to assess the severity of HS.53 We will describe the Hurley staging system and the International Hidradenitis Suppurativa Severity Score System (IHS4). Other diagnostic tools, such as the Sartorius score and the Hidradenitis Suppurativa Physician’s Global Assessment Scale (HS-PGA), can be time-consuming and challenging to interpret, limiting their use in the clinical setting.2,54
Hurley staging system (available at www.hsdiseasesource.com/hs-disease-staging) considers the presence of nodules, abscesses, sinus tracts, and scarring affecting an entire anatomical area.13,55 This system is most useful as a rapid classification tool for patients with HS in the clinical setting but should not be used to assess clinical response.2,13,56
The IHS4 (available at https://online library.wiley.com/doi/10.1111/bjd.15748) is a validated and easy-to-use tool for assessing HS and guiding the therapeutic strategy in clinical practice.54 With IHS4, the clinician must calculate the following:
- total number of nodules > 10 mm in diameter
- total number of abscesses multiplied by 2, and
- total number of draining tunnels (fistulae/sinuses) multiplied by 4.
Mild HS is defined as a score ≤ 3 points; moderate HS, 4 to 10 points; and severe HS, ≥ 11 points.54
No diagnostic tests, but ultrasound may be helpful
There are currently no established biological markers or specific tests for diagnosing HS.15 Ultrasound is emerging as a tool to assess dermal thickness, hair follicle morphology, and number and extent of fluid collections. Two recent studies showed that pairing clinical assessment with ultrasound findings improves accuracy of scoring in 84% of cases.57,58 For patients with severe HS, skin biopsy can be considered to rule out squamous cell carcinoma. Cultures, however, have limited utility except for suspected superimposed bacterial infection.2
Continue to: Screening for comorbidities
Screening for comorbidities
HSF recommends clinicians screen patients for comorbidities associated with HS (TABLE 2).2 Overall, screening patients for active and past history of smoking is strongly recommended, as is screening for metabolic syndrome, hyperlipidemia, type 2 diabetes (1.5- to 3-fold greater risk of type 2 diabetes in HS patients), and PCOS (3-fold greater risk).2,26,27,59 Screening patients for depression and anxiety is also routinely recommended.2 However, the authors of this article strongly recommend screening all patients with HS for psychiatric comorbidities, as research has shown a 2-fold greater risk of depression and anxiety, social isolation, and low self-esteem that severely limits quality of life (QOL) in this patient population.60,61

Management
Treat existing lesions, reduce formation of new ones
The main goals of treatment for patients with HS are to treat existing lesions and reduce associated symptoms, reduce the formation of new lesions, and minimize associated psychological morbidity.15 FPs play an important role in the early diagnosis, treatment, and comprehensive care of patients with HS. This includes monitoring patients, managing comorbidities, making appropriate referrals to dermatologists, and coordinating the multidisciplinary care that patients with HS require.

A systematic review identified more than 50 interventions used to treat HS, most based on small observational studies and randomized controlled trials (RCTs) with a high risk of bias.62 FIGURE 22,62-69 provides an evidence-based treatment algorithm for HS, and TABLE 32,63,64,70-75 summarizes the most commonly used treatments.

Biologic agents
Adalimumab (ADA) is a fully human immunoglobulin G1 monoclonal antibody that binds to TNF-alpha, neutralizes its bioactivity, and induces apoptosis of TNF-expressing mononuclear cells. It is the only medication approved by the US Food and Drug Administration for active refractory moderate and severe HS.62,65 Several double-blinded RCTs, including PIONEER I and PIONEER II, studied the effectiveness of ADA for HS and found significant clinical responses at Week 12, 50% reduction in abscess and nodule counts, no increase in abscesses or draining fistulas at Week 12, and sustained improvement in lesion counts, pain, and QOL.66,67,76
IL-1 and IL-23 inhibitors. The efficacy of etanercept and golimumab (anti-TNF), as well as anakinra (IL-1 inhibitor) and ustekinumab (IL-1/IL-23 inhibitor), continue to be investigated with variable results; they are considered second-line treatment for active refractory moderate and severe HS after ADA.65,77-80 Infliximab (IL-1 beta inhibitor) has shown no effect on reducing disease severity.70Compared to other treatments, biologic therapy is associated with higher costs (TABLE 3),2,63,64,70-75 an increased risk for reactivation of latent infections (eg, tuberculosis, herpes simplex, and hepatitis C virus [HCV], and B [HBV]), and an attenuated response to vaccines.81 Prior to starting biologic therapy, FPs should screen patients with HS for tuberculosis and HBV, consider HIV and HCV screening in at-risk patients, and optimize the immunization status of the patient.82,83 While inactivated vaccines can be administered without discontinuing biologic treatment, patients should avoid live-attenuated vaccines while taking biologics.83
Continue to: Antibiotic therapy
Antibiotic therapy
Topical antibiotics are considered first-line treatment for mild and moderate uncomplicated HS.63,64 Clindamycin 1%, the only topical antibiotic studied in a small double-blind RCT of patients with Hurley stage I and stage II HS, demonstrated significant clinical improvement after 12 weeks of treatment (twice- daily application), compared to placebo.84 Topical clindamycin is also recommended to treat flares in patients with mild disease.2,64
Oral antibiotics. Tetracycline (500 mg twice daily for 4 months) is considered a second-line treatment for patients with mild HS.64,68 Doxycycline (200 mg/d for 3 months) may also be considered as a second-line treatment in patients with mild disease.85
Combination oral clindamycin (300 mg) and rifampicin (300 mg) twice daily for 10 weeks is recommended as first-line treatment for patients with moderate HS.2,64,69 Combination rifampin (300 mg twice daily), moxifloxacin (400 mg/d), and metronidazole (500 mg three times a day) is not routinely recommended due to increased risk of toxicity.2
Ertapenem (1 g intravenously daily for 6 weeks) is supported by lower-level evidence as a third-line rescue therapy option and as a bridge to surgery; however, limitations for home infusions, costs, and concerns for antibiotic resistance limit its use.2,86
Corticosteroids and systemic immunomodulators
Intralesional triamcinolone (2-20 mg) may be beneficial in the early stages of HS, although its use is based on a small prospective open study of 33 patients.87 A recent double-blind placebo-controlled RCT comparing varying concentrations of intralesional triamcinolone (10 mg/mL and 40 mg/mL) vs normal saline showed no statistically significant difference in inflammatory clearance, pain reduction, or patient satisfaction.88
Continue to: Short-term systemic corticosteroid tapers...
Short-term systemic corticosteroid tapers (eg, prednisone, starting at 0.5-1 mg/kg) are recommended to treat flares. Long-term corticosteroids and cyclosporine are reserved for patients with severe refractory disease; however, due to safety concerns, their regular use is strongly discouraged.63,64,85 There is limited evidence to support the use of methotrexate for severe refractory disease, and its use is not recommended.63
Hormonal therapy
The use of hormonal therapy for HS is limited by the low-quality evidence (eg, anecdotal evidence, small retrospective analyses, uncontrolled trials).33,63 The only exception is a small double-blind controlled crossover trial from 1986 showing that the antiandrogen effects of combination oral contraceptives (ethinyloestradiol 50 mcg/cyproterone acetate in a reverse sequential regimen and ethinyloestradiol 50 mcg/norgestrel 500 mcg) improved HS lesions.89
Spironolactone, an antiandrogen diuretic, has been studied in small case report series with a high risk for bias. It is used mainly in female patients with mild or moderate disease, or in combination with other agents in patients with severe HS. Further research is needed to determine its utility in the treatment of HS.63,90,91
Metformin, alone or in combination with other therapies (dapsone, finasteride, liraglutide), has been analyzed in small prospective studies of primarily female patients with different severities of HS, obesity, and PCOS. These studies have shown improvement in lesions, QOL, and reduction of workdays lost.92,93
Finasteride. Studies have shown finasteride (1.25-5 mg/d) alone or in combination with other treatments (metformin, liraglutide, levonorgestrel-ethinyl estradiol, and dapsone) provided varying degrees of resolution or improvement in patients with severe and advanced HS. Finasteride has been used for 4 to 16 weeks with a good safety profile.92,94-96
Continue to: Retinoids
Retinoids
Acitretin, alitretinoin, and isotretinoin have been studied in small retrospective studies to manage HS, with variable results.97-99 Robust prospective studies are needed. Retinoids, in general, should be considered as a second- or third-line treatment for moderate to severe HS.63
Surgical intervention
Surgical interventions, which should be considered in patients with widespread mild, moderate, or severe disease, are associated with improved daily activity and work productivity.100 Incision and drainage should be avoided in patients with HS, as this technique does not remove the affected follicles and is associated with 100% recurrence.101
Wide excision is the preferred surgical technique for patients with Hurley stage II and stage III HS; it is associated with lower recurrence rates (13%) compared to local excision (22%) and deroofing (27%).102 Secondary intention healing is the most commonly chosen method, based on lower recurrence rates than primary closure.102
STEEP and laser techniques. The skin-tissue-sparing excision with electrosurgical peeling (STEEP) procedure involves successive tangential excision of affected tissue until the epithelized bottom of the sinus tracts has been reached. This allows for the removal of fibrotic tissue and the sparing of the deep subcutaneous fat. STEEP is associated with 30% of relapses after 43 months.71
Laser surgery has also been studied in patients with Hurley stage II and stage III HS. The most commonly used lasers for HS are the 1064-nm neodymium-doped yttrium aluminum garnet (Nd: YAG) and the carbon dioxide laser; they have been shown to reduce disease severity in inguinal, axillary, and inflammatory sites.72-74
Pain management: Start with lidocaine, NSAIDs
There are few studies about HS-associated pain management.103 For acute episodes, short-acting nonopioid local treatment with lidocaine, topical or oral nonsteroidal anti-inflammatory drugs, and acetaminophen are preferred. Opioids should be reserved for moderate-to-severe pain that has not responded to other analgesics. Adjuvant therapy with pregabalin, gabapentin, selective serotonin reuptake inhibitors, or serotonin-norepinephrine reuptake inhibitors can also be considered for the comanagement of pain and depression.62,104
Consider this tool to measure treatment response
The HS clinical response (HiSCR) tool is an outcome measure used to evaluate treatment outcomes. The tool uses an HS-specific binary score with the following criteria:
- ≥ 50% reduction in the number of inflammatory nodules;
- no increase in the number of abscesses; and
- no increase in the number of draining fistulas.105
The HiSCR was developed for the PIONEER studies105,106 to assess the response to ADA treatment. It is the only HS scoring system to undergo an extensive validation process with a meaningful clinical endpoint for HS treatment evaluation that is easy to use. Compared to the HS-PGA score (clear, minimal, mild), HiSCR was more responsive to change in patients with HS.105,106
CORRESPONDENCE
Cristina Marti-Amarista, MD, 101 Nicolls Road, Stony Brook, NY, 11794-8228; marti.amarista@gmail.com
1. Bergler-Czop B, Hadasik K, Brzezińska-Wcisło L. Acne inversa: difficulties in diagnostics and therapy. Postepy Dermatol Alergol. 2015;32:296-301. doi: 10.5114/pdia.2014.44012
2. Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part I: diagnosis, evaluation, and the use of complementary and procedural management. J Am Acad Dermatol. 2019;81:76-90. doi: 10.1016/j.jaad.2019.02.067
3. Fimmel S, Zouboulis CC. Comorbidities of hidradenitis suppurativa (acne inversa). Dermatoendocrinol. 2010;2:9-16. doi: 10.4161/derm.2.1.12490
4. Kokolakis G, Wolk K, Schneider-Burrus S, et al. Delayed diagnosis of hidradenitis suppurativa and its effect on patients and healthcare system. Dermatology. 2020;236:421-430. doi: 10.1159/000508787
5. Gulliver W, Landells IDR, Morgan D, et al. Hidradenitis suppurativa: a novel model of care and an integrative strategy to adopt an orphan disease. J Cutan Med Surg. 2018;22:71-77. doi: 10.1177/1203475417736290
6. Savage KT, Gonzalez Brant E, Flood KS, et al. Publication trends in hidradenitis suppurativa from 2008 to 2018. J Eur Acad Dermatol Venereol. 2020;34:1885-1889. doi: 10.1111/jdv.16213.
7. Narla S, Lyons AB, Hamzavi IH. The most recent advances in understanding and managing hidradenitis suppurativa. F1000Res. 2020;9:F1000. doi: 10.12688/f1000research.26083.1
8. Vossen ARJV, van der Zee HH, Prens EP. Hidradenitis suppurativa: a systematic review integrating inflammatory pathways into a cohesive pathogenic model. Front Immunol. 2018;9:2965. doi: 10.3389/fimmu.2018.02965
9. Frew JW, Hawkes JE, Krueger JG. A systematic review and critical evaluation of inflammatory cytokine associations in hidradenitis suppurativa. F1000Res. 2018;7:1930. doi: 10.12688/f1000 research.17267.1
10. Sabat R, Jemec GBE, Matusiak Ł. Hidradenitis suppurativa. Nat Rev Dis Primers. 2020;6:18. doi: 10.1038/s41572-020-0149-1
11. Shlyankevich J, Chen AJ, Kim GE, et al. Hidradenitis suppurativa is a systemic disease with substantial comorbidity burden: a chart-verified case-control analysis. J Am Acad Dermatol. 2014;71:1144-1150. doi: 10.1016/j.jaad.2014.09.012
12. Sartorius K, Emtestam L, Jemec GB, et al. Objective scoring of hidradenitis suppurativa reflecting the role of tobacco smoking and obesity. Br J Dermatol. 2009;161:831-839. doi: 10.1111/j.1365-2133.2009.09198.x
13. von Laffert M, Helmbold P, Wohlrab J, et al. Hidradenitis suppurativa (acne inversa): early inflammatory events at terminal follicles and at interfollicular epidermis. Exp Dermatol. 2010;19:533-537. doi: 10.1111/j.1600-0625.2009.00915.x
14. Jemec GB, Hansen U. Histology of hidradenitis suppurativa. J Am Acad Dermatol. 1996;34:994-999. doi: 10.1016/s0190-9622(96)90277-7
15. Ballard K, Shuman VL. Hidradenitis suppurativa. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021. Updated July 15, 2022. Accessed November 28, 2022. www.ncbi.nlm.nih.gov/books/NBK534867/
16. Garg A, Kirby JS, Lavian J, et al. Sex- and age-adjusted population analysis of prevalence estimates for hidradenitis suppurativa in the United States. JAMA Dermatol. 2017;153:760-764. doi: 10.1001/jamadermatol.2017.0201
17. Phan K, Charlton O, Smith SD. Global prevalence of hidradenitis suppurativa and geographical variation—systematic review and meta-analysis. BioMed Dermatol. 2020;4. doi: 10.1186/s41702-019-0052-0
18. Vazquez BG, Alikhan A, Weaver AL, et al. Incidence of hidradenitis suppurativa and associated factors: a population-based study of Olmsted County, Minnesota. J Invest Dermatol. 2013;133:97-103. doi: 10.1038/jid.2012.255
19. Sachdeva M, Shah M, Alavi A. Race-specific prevalence of hidradenitis suppurativa. J Cutan Med Surg. 2021;25:177-187. doi: 10.1177/1203475420972348
20. Vaidya T, Vangipuram R, Alikhan A. Examining the race-specific prevalence of hidradenitis suppurativa at a large academic center; results from a retrospective chart review. Dermatol Online J. 2017;23:13030/qt9xc0n0z1. doi: 10.5070/D3236035391
21. Price KN, Hsiao JL, Shi VY. Race and ethnicity gaps in global hidradenitis suppurativa clinical trials. Dermatology. 2021;237:97-102. doi: 10.1159/000504911
22. Schrader AM, Deckers IE, van der Zee HH, et al. Hidradenitis suppurativa: a retrospective study of 846 Dutch patients to identify factors associated with disease severity. J Am Acad Dermatol. 2014;71:460-467. doi: 10.1016/j.jaad.2014.04.001
23. Frew JW, Vekic DA, Wood J, et al. A systematic review and critical evaluation of reported pathogenic sequence variants in hidradenitis suppurativa. Br J Dermatol. 2017;177:987-998. doi: 10.1111/bjd.15441
24. Wolk K, Join-Lambert O, Sabat R. Aetiology and pathogenesis of hidradenitis suppurativa. Br J Dermatol. 2020;183:999-1010. doi: 10.1111/bjd.19556
25. Goldburg SR, Strober BE, Payette MJ. Hidradenitis suppurativa: epidemiology, clinical presentation, and pathogenesis. J Am Acad Dermatol. 2020;82:1045-1058. doi: 10.1016/j.jaad.2019.08.090
26. Sabat R, Chanwangpong A, Schneider-Burrus S, et al. Increased prevalence of metabolic syndrome in patients with acne inversa. PloS One. 2012;7:e31810. doi: 10.1371/journal.pone.0031810
27. Loh TY, Hendricks AJ, Hsiao JL, et al. Undergarment and fabric selection in the management of hidradenitis suppurativa. Dermatology. 2021;237:119-124. doi: 10.1159/000501611
28. Rodríguez-Zuñiga MJM, García-Perdomo HA, Ortega-Loayza AG. Association between hidradenitis suppurativa and metabolic syndrome: a systematic review and meta-analysis. Actas Dermosifiliogr (Engl Ed). 2019;110:279-288. doi: 10.1016/j.ad.2018.10.020
29. Walker JM, Garcet S, Aleman JO, et al. Obesity and ethnicity alter gene expression in skin. Sci Rep. 2020;10:14079. doi: 10.1038/s41598-020-70244-2.
30. Boer J, Nazary M, Riis PT. The role of mechanical stress in hidradenitis suppurativa. Dermatol Clin. 2016;34:37-43. doi: 10.1016/j.det.2015.08.011
31. Vossen ARJV, van Straalen KR, Swolfs EFH, et al. Nicotine dependency and readiness to quit smoking among patients with hidradenitis suppurativa. Dermatology. 2021;237:383-385. doi: 10.1159/000514028
32. Kromann CB, Deckers IE, Esmann S, et al. Risk factors, clinical course and long-term prognosis in hidradenitis suppurativa: a cross-sectional study. Br J Dermatol. 2014;171:819-824. doi: 10.1111/bjd.13090
33. Clark AK, Quinonez RL, Saric S, et al. Hormonal therapies for hidradenitis suppurativa: review. Dermatol Online J. 2017;23:13030/qt6383k0n4. doi: 10.5070/D32310036990
34. Saric-Bosanac S, Clark AK, Sivamani RK, et al. The role of hypothalamus-pituitary-adrenal (HPA)-like axis in inflammatory pilosebaceous disorders. Dermatol Online J. 2020;26:13030/qt8949296f. doi: 10.5070/D3262047430
35. Riis PT, Ring HC, Themstrup L, et al. The role of androgens and estrogens in hidradenitis suppurativa – a systematic review. Acta Dermatovenerol Croat. 2016;24:239-249.
36. Hsiao JL, Antaya RJ, Berger T, et al. Hidradenitis suppurativa and concomitant pyoderma gangrenosum: a case series and literature review. Arch Dermatol. 2010;146:1265-1270. doi: 10.1001/archdermatol.2010.328
37. Ah-Weng A, Langtry JAA, Velangi S, et al. Pyoderma gangrenosum associated with hidradenitis suppurativa. Clin Exp Dermatol. 2005;30:669-671. doi: 10.1111/j.1365-2230.2005.01897.x
38. Kirthi S, Hellen R, O’Connor R, et al. Hidradenitis suppurativa and Crohn’s disease: a case series. Ir Med J. 2017;110:618.
39. Dumont LM, Landman C, Sokol H, et al; CD-HS Study Group. Increased risk of permanent stoma in Crohn’s disease associated with hidradenitis suppurativa: a case-control study. Aliment Pharmacol Ther. 2020;52:303-310. doi: 10.1111/apt.15863
40. Marzano AV, Ceccherini I, Gattorno M, et al. Association of pyoderma gangrenosum, acne, and suppurative hidradenitis (PASH) shares genetic and cytokine profiles with other autoinflammatory diseases. Medicine (Baltimore). 2014;93:e187. doi: 10.1097/MD.0000000000000187.
41. Vinkel C, Thomsen SF. Hidradenitis suppurativa: causes, features, and current treatments. J Clin Aesthet Dermatol. 2018;11:17-23.
42. Wipperman J, Bragg DA, Litzner B. Hidradenitis suppurativa: rapid evidence review. Am Fam Physician. 2019;100:562-569.
43. Theut Riis P, Pedersen OB, Sigsgaard V, et al. Prevalence of patients with self-reported hidradenitis suppurativa in a cohort of Danish blood donors: a cross-sectional study. Br J Dermatol. 2019;180:774-781. doi: 10.1111/bjd.16998.
44. Dufour DN, Emtestam L, Jemec GB. Hidradenitis suppurativa: a common and burdensome, yet under-recognised, inflammatory skin disease. Postgrad Med J. 2014;90:216-221; doi: 10.1136/postgradmedj-2013-131994
45. Vinding GR, Miller IM, Zarchi K, et al. The prevalence of inverse recurrent suppuration: a population-based study of possible hidradenitis suppurativa. Br J Dermatol. 2014;170:884-889. doi: 10.1111/bjd.12787
46. Bassas-Vila J, González Lama Y. Hidradenitis suppurativa and perianal Crohn disease: differential diagnosis. Actas Dermosifiliogr. 2016;107(suppl 2):27-31. doi: 10.1016/S0001-7310(17) 30006-6
47. Valour F, Sénéchal A, Dupieux C, et al. Actinomycosis: etiology, clinical features, diagnosis, treatment, and management. Infect Drug Resist. 2014;7:183-197. doi: 10.2147/IDR.S39601
48. Fuchs W, Brockmeyer NH. Sexually transmitted infections. J Dtsch Dermatol Ges. 2014;12:451-463. doi: 10.1111/ddg.12310
49. Hap W, Frejlich E, Rudno-Rudzińska J, et al. Pilonidal sinus: finding the righttrack for treatment. Pol Przegl Chir. 2017;89:68-75. doi: 10.5604/01.3001.0009.6009
50. Al-Hamdi KI, Saadoon AQ. Acne onglobate of the scalp. Int J Trichology. 2020;12:35-37. doi: 10.4103/ijt.ijt_117_19
51. Balestra A, Bytyci H, Guillod C, et al. A case of ulceroglandular tularemia presenting with lymphadenopathy and an ulcer on a linear morphoea lesion surrounded by erysipelas. Int Med Case Rep J. 2018;11:313-318. doi: 10.2147/IMCRJ.S178561
52. Ibler KS, Kromann CB. Recurrent furunculosis – challenges and management: a review. Clin Cosmet Investig Dermatol. 2014;7:59-64. doi: 10.2147/CCID.S35302
53. Ingram JR, Hadjieconomou S, Piguet V. Development of core outcome sets in hidradenitis suppurativa: systematic review of outcome measure instruments to inform the process. Br J Dermatol. 2016;175:263-272. doi: 10.1111/bjd.14475
54. Zouboulis CC, Tzellos T, Kyrgidis A, et al; European Hidradenitis Suppurativa Foundation Investigator Group. Development and validation of the International Hidradenitis Suppurativa Severity Score System (I4), a novel dynamic scoring system to assess HS severity. Br J Dermatol. 2017;177:1401-1409. doi: 10.1111/bjd.15748
55. Hidradenitis Suppurativa Clinical Resource. Hidradenitis suppurativa stages: Hurley Staging System. www.hsdiseasesource.com/hs-disease-staging. Accessed October 11, 2022.
56. Ovadja ZN, Schuit MM, van der Horst CMAM, et al. Inter- and interrater reliability of Hurley staging for hidradenitis suppurativa. Br J Dermatol. 2019;181:344-349. doi: 10.1111/bjd.17588
57. Wortsman X, Jemec GBE. Real-time compound imaging ultrasound of hidradenitis suppurativa. Dermatol Surg. 2007;33:1340-1342. doi: 10.1111/j.1524-4725.2007.33286.x
58. Napolitano M, Calzavara-Pinton PG, Zanca A, et al. Comparison of clinical and ultrasound scores in patients with hidradenitis suppurativa: results from an Italian ultrasound working group. J Eur Acad Dermatol Venereol. 2019;33:e84-e87. doi: 10.1111/jdv.15235
59. Bukvić Mokos Z, Miše J, Balić A, et al. Understanding the relationship between smoking and hidradenitis suppurativa. Acta Dermatovenerol Croat. 2020;28:9-13.
60. Shavit E, Dreiher J, Freud T, et al. Psychiatric comorbidities in 3207 patients with hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2015;29:371-376. doi: 10.1111/jdv.12567
61. Kouris A, Platsidaki E, Christodoulou C, et al. Quality of life and psychosocial implications in patients with hidradenitis suppurativa. Dermatology. 2016;232:687-691. doi: 10.1159/000453355
62 Ingram JR, Woo PN, Chua SL, et al. Interventions for hidradenitis suppurativa: a Cochrane systematic review incorporating GRADE assessment of evidence quality. Br J Dermatol. 2016;174:970-978. doi: 10.1111/bjd.14418
63. Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part II: topical, intralesional, and systemic medical management. J Am Acad Dermatol. 2019;81:91-101. doi: 10.1016/j.jaad.2019.02.068
64. Gulliver W, Zouboulis CC, Prens E, et al. Evidence-based approach to the treatment of hidradenitis suppurativa/acne inversa, based on the European guidelines for hidradenitis suppurativa. Rev Endocr Metab Disord. 2016;17:343-351. doi: 10.1007/s11154-016-9328-5
65. Vena GA, Cassano N. Drug focus: adalimumab in the treatment of moderate to severe psoriasis. Biologics. 2007;1:93-103.
66. Kimball AB, Kerdel F, Adams D, et al. Adalimumab for the treatment of moderate to severe hidradenitis suppurativa: a parallel randomized trial. Ann Intern Med. 2012;157:846-55. doi: 10.7326/0003-4819-157-12-201212180-00004
67. Zouboulis CC, Okun MM, Prens EP, et al. Long-term adalimumab efficacy in patients with moderate-to-severe hidradenitis suppurativa/acne inversa: 3-year results of a phase 3 open-label extension study. J Am Acad Dermatol. 2019;80:60-69.e2. doi: 10.1016/j.jaad.2018.05.040
68. Jemec GB, Wendelboe P. Topical clindamycin versus systemic tetracycline in the treatment of hidradenitis suppurativa. J Am Acad Dermatol. 1998;39:971-974. doi: 10.1016/s0190-9622(98)70272-5
69. Gener G, Canoui-Poitrine F, Revuz JE, et al. Combination therapy with clindamycin and rifampicin for hidradenitis suppurativa: a series of 116 consecutive patients. Dermatology. 2009;219:148-154. doi: 10.1159/000228334
70. Grant A, Gonzalez T, Montgomery MO, et al. Infliximab therapy for patients with moderate to severe hidradenitis suppurativa: a randomized, double-blind, placebo-controlled crossover trial. J Am Acad Dermatol. 2010;62:205-217. doi: 10.1016/j.jaad.2009.06.050
71. Blok JL, Spoo JR, Leeman FWJ, et al. Skin-tissue-sparing excision with electrosurgical peeling (STEEP): a surgical treatment option for severe hidradenitis suppurativa Hurley stage II/III. J Eur Acad Dermatol Venereol. 2015;29:379-382. doi: 10.1111/jdv.12376
72. Mahmoud BH, Tierney E, Hexsel CL, et al. Prospective controlled clinical and histopathologic study of hidradenitis suppurativa treated with the long-pulsed neodymium:yttrium-aluminium-garnet laser. J Am Acad Dermatol. 2010;62:637-645. doi: 10.1016/j.jaad.2009.07.048
73. Tierney E, Mahmoud BH, Hexsel C, et al. Randomized control trial for the treatment of hidradenitis suppurativa with a neodymium-doped yttrium aluminium garnet laser. Dermatol Surg. 2009;35:1188-1198. doi: 10.1111/j.1524-4725.2009.01214.x
74. Hazen PG, Hazen BP. Hidradenitis suppurativa: successful treatment using carbon dioxide laser excision and marsupialization. Dermatol Surg. 2010;36:208-213. doi: 10.1111/j.1524-4725.2009.01427.x
75. van der Zee HH, Prens EP, Boer J. Deroofing: a tissue-saving surgical technique for the treatment of mild to moderate hidradenitis suppurativa lesions. J Am Acad Dermatol. 2010;63:475-480. doi: 10.1016/j.jaad.2009.12.018
76. Kimball AB, Okun MM, Williams DA, et al. Two phase 3 trials of adalimumab for hidradenitis suppurativa. N Engl J Med. 2016;375:422-434. doi: 10.1056/NEJMoa1504370. PMID: 27518661.
77. Adams DR, Yankura JA, Fogelberg AC, et al. Treatment of hidradenitis suppurativa with etanercept injection. Arch Dermatol. 2010;146:501-504. doi: 10.1001/archdermatol.2010.72
78. Tursi A. Concomitant hidradenitis suppurativa and pyostomatitis vegetans in silent ulcerative colitis successfully treated with golimumab. Dig Liver Dis. 2016;48:1511-1512. doi: 10.1016/j.dld.2016.09.010
79. Tzanetakou V, Kanni T, Giatrakou S, et al. Safety and efficacy of anakinra in severe hidradenitis suppurativa: a randomized clinical trial. JAMA Dermatol. 2016;152:52-59. doi: 10.1001/jamadermatol.2015.3903.
80. Romaní J, Vilarrasa E, Martorell A, et al. Ustekinumab with intravenous infusion: results in hidradenitis suppurativa. Dermatology. 2020;236:21-24. doi: 10.1159/000501075
81. Kane SV. Preparing for biologic or immunosuppressant therapy. Gastroenterol Hepatol (N Y). 2011;7:544-546.
82. Davis W, Vavilin I, Malhotra N. Biologic therapy in HIV: to screen or not to screen. Cureus. 2021;13:e15941. doi: 10.7759/cureus.15941
83. Papp KA, Haraoui B, Kumar D, et al. Vaccination guidelines for patients with immune-mediated disorders on immunosuppressive therapies. J Cutan Med Surg. 2019;23:50-74. doi: 10.1177/1203475418811335
84. Clemmensen OJ. Topical treatment of hidradenitis suppurativa with clindamycin. Int J Dermatol. 1983;22:325-328. doi: 10.1111/j.1365-4362.1983.tb02150.x
85. Hunger RE, Laffitte E, Läuchli S, et al. Swiss practice recommendations for the management of hidradenitis suppurativa/acne inversa. Dermatology. 2017;233:113-119. doi: 10.1159/000477459
86. Zouboulis CC, Bechara FG, Dickinson-Blok JL, et al. Hidradenitis suppurativa/acne inversa: a practical framework for treatment optimization - systematic review and recommendations from the HS ALLIANCE working group. J Eur Acad Dermatol Venereol. 2019;33:19-31. doi: 10.1111/jdv.15233
87. Riis PT, Boer J, Prens EP, et al. Intralesional triamcinolone for flares of hidradenitis suppurativa (HS): a case series. J Am Acad Dermatol. 2016;75:1151-1155. doi: 10.1016/j.jaad.2016.06.049
88. Fajgenbaum K, Crouse L, Dong L, et al. Intralesional triamcinolone may not be beneficial for treating acute hidradenitis suppurativa lesions: a double-blind, randomized, placebo-controlled trial. Dermatol Surg. 2020;46:685-689. doi: 10.1097/DSS.0000000000002112
89. Mortimer PS, Dawber RP, Gales MA, et al. A double-blind controlled cross-over trial of cyproterone acetate in females with hidradenitis suppurativa. Br J Dermatol. 1986;115:263-268. doi: 10.1111/j.1365-2133.1986.tb05740.x
90. Kraft JN, Searles GE. Hidradenitis suppurativa in 64 female patients: retrospective study comparing oral antibiotics and antiandrogen therapy. J Cutan Med Surg. 2007;11:125-131. doi: 10.2310/7750.2007.00019
91. Lee A, Fischer G. A case series of 20 women with hidradenitis suppurativa treated with spironolactone. Australas J Dermatol. 2015;56:192-196. doi: 10.1111/ajd.12362
92. Khandalavala BN. A disease-modifying approach for advanced hidradenitis suppurativa (regimen with metformin, liraglutide, dapsone, and finasteride): a case report. Case Rep Dermatol. 2017;9:70-78. doi: 10.1159/000473873
93. Verdolini R, Clayton N, Smith A, et al. Metformin for the treatment of hidradenitis suppurativa: a little help along the way. J Eur Acad Dermatol Venereol. 2013;27:1101-1108. doi: 10.1111/j.1468-3083.2012.04668.x
94. Khandalavala BN, Do MV. Finasteride in hidradenitis suppurativa: a “male” therapy for a predominantly “female” disease. J Clin Aesthet Dermatol. 2016;9:44-50.
95. Mota F, Machado S, Selores M. Hidradenitis suppurativa in children treated with finasteride-a case series. Pediatr Dermatol. 2017;34:578-583. doi: 10.1111/pde.13216
96. Doménech C, Matarredona J, Escribano-Stablé JC, et al. Facial hidradenitis suppurativa in a 28-year-old male responding to finasteride. Dermatology. 2012;224:307-308. doi: 10.1159/000339477
97. Patel N, McKenzie SA, Harview CL, et al. Isotretinoin in the treatment of hidradenitis suppurativa: a retrospective study. J Dermatolog Treat. 2021;32:473-475. doi: 10.1080/09546634.2019.1670779
98. Boer J, van Gemert MJ. Long-term results of isotretinoin in the treatment of 68 patients with hidradenitis suppurativa. J Am Acad Dermatol. 1999;40:73-76. doi: 10.1016/s0190-9622(99) 70530-x
99. Huang CM, Kirchhof MG. A new perspective on isotretinoin treatment of hidradenitis suppurativa: a retrospective chart review of patient outcomes. Dermatology. 2017;233:120-125. doi: 10.1159/000477207
100. Prens LM, Huizinga J, Janse IC. Surgical outcomes and the impact of major surgery on quality of life, activity impairment and sexual health in hidradenitis suppurativa patients: a prospective single centre study. J Eur Acad Dermatol Venereol. 2019;33:1941-1946. doi: 10.1111/jdv.15706
101. Ritz JP, Runkel N, Haier J, et al. Extent of surgery and recurrence rate of hidradenitis suppurativa. Int J Colorectal Dis. 1998;13:164-168. doi: 10.1007/s003840050159
102. Mehdizadeh A, Hazen PG, Bechara FG, et al. Recurrence of hidradenitis suppurativa after surgical management: a systematic review and meta-analysis. J Am Acad Dermatol. 2015;73(5 suppl 1):S70-S77. doi: 10.1016/j.jaad.2015.07.044.
103. Smith HS, Chao JD, Teitelbaum J. Painful hidradenitis suppurativa. Clin J Pain. 2010;26:435-444. doi: 10.1097/AJP.0b013e3181ceb80c
104. Horváth B, Janse IC, Sibbald GR. Pain management in patients with hidradenitis suppurativa. J Am Acad Dermatol. 2015;73(5 suppl 1):S47-S51. doi: 10.1016/j.jaad.2015.07.046
105. Kimball AB, Sobell JM, Zouboulis CC, et al. HiSCR (Hidradenitis Suppurativa Clinical Response): a novel clinical endpoint to evaluate therapeutic outcomes in patients with hidradenitis suppurativa from the placebo-controlled portion of a phase 2 adalimumab study. J Eur Acad Dermatol Venereol. 2016;30:989-994. doi: 10.1111/jdv.13216
106. Kimball AB, Jemec GB, Yang M, et al. Assessing the validity, responsiveness and meaningfulness of the Hidradenitis Suppurativa Clinical Response (HiSCR) as the clinical endpoint for hidradenitis suppurativa treatment. Br J Dermatol. 2014;171:1434-1442. doi: 10.1111/bjd.13270
1. Bergler-Czop B, Hadasik K, Brzezińska-Wcisło L. Acne inversa: difficulties in diagnostics and therapy. Postepy Dermatol Alergol. 2015;32:296-301. doi: 10.5114/pdia.2014.44012
2. Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part I: diagnosis, evaluation, and the use of complementary and procedural management. J Am Acad Dermatol. 2019;81:76-90. doi: 10.1016/j.jaad.2019.02.067
3. Fimmel S, Zouboulis CC. Comorbidities of hidradenitis suppurativa (acne inversa). Dermatoendocrinol. 2010;2:9-16. doi: 10.4161/derm.2.1.12490
4. Kokolakis G, Wolk K, Schneider-Burrus S, et al. Delayed diagnosis of hidradenitis suppurativa and its effect on patients and healthcare system. Dermatology. 2020;236:421-430. doi: 10.1159/000508787
5. Gulliver W, Landells IDR, Morgan D, et al. Hidradenitis suppurativa: a novel model of care and an integrative strategy to adopt an orphan disease. J Cutan Med Surg. 2018;22:71-77. doi: 10.1177/1203475417736290
6. Savage KT, Gonzalez Brant E, Flood KS, et al. Publication trends in hidradenitis suppurativa from 2008 to 2018. J Eur Acad Dermatol Venereol. 2020;34:1885-1889. doi: 10.1111/jdv.16213.
7. Narla S, Lyons AB, Hamzavi IH. The most recent advances in understanding and managing hidradenitis suppurativa. F1000Res. 2020;9:F1000. doi: 10.12688/f1000research.26083.1
8. Vossen ARJV, van der Zee HH, Prens EP. Hidradenitis suppurativa: a systematic review integrating inflammatory pathways into a cohesive pathogenic model. Front Immunol. 2018;9:2965. doi: 10.3389/fimmu.2018.02965
9. Frew JW, Hawkes JE, Krueger JG. A systematic review and critical evaluation of inflammatory cytokine associations in hidradenitis suppurativa. F1000Res. 2018;7:1930. doi: 10.12688/f1000 research.17267.1
10. Sabat R, Jemec GBE, Matusiak Ł. Hidradenitis suppurativa. Nat Rev Dis Primers. 2020;6:18. doi: 10.1038/s41572-020-0149-1
11. Shlyankevich J, Chen AJ, Kim GE, et al. Hidradenitis suppurativa is a systemic disease with substantial comorbidity burden: a chart-verified case-control analysis. J Am Acad Dermatol. 2014;71:1144-1150. doi: 10.1016/j.jaad.2014.09.012
12. Sartorius K, Emtestam L, Jemec GB, et al. Objective scoring of hidradenitis suppurativa reflecting the role of tobacco smoking and obesity. Br J Dermatol. 2009;161:831-839. doi: 10.1111/j.1365-2133.2009.09198.x
13. von Laffert M, Helmbold P, Wohlrab J, et al. Hidradenitis suppurativa (acne inversa): early inflammatory events at terminal follicles and at interfollicular epidermis. Exp Dermatol. 2010;19:533-537. doi: 10.1111/j.1600-0625.2009.00915.x
14. Jemec GB, Hansen U. Histology of hidradenitis suppurativa. J Am Acad Dermatol. 1996;34:994-999. doi: 10.1016/s0190-9622(96)90277-7
15. Ballard K, Shuman VL. Hidradenitis suppurativa. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021. Updated July 15, 2022. Accessed November 28, 2022. www.ncbi.nlm.nih.gov/books/NBK534867/
16. Garg A, Kirby JS, Lavian J, et al. Sex- and age-adjusted population analysis of prevalence estimates for hidradenitis suppurativa in the United States. JAMA Dermatol. 2017;153:760-764. doi: 10.1001/jamadermatol.2017.0201
17. Phan K, Charlton O, Smith SD. Global prevalence of hidradenitis suppurativa and geographical variation—systematic review and meta-analysis. BioMed Dermatol. 2020;4. doi: 10.1186/s41702-019-0052-0
18. Vazquez BG, Alikhan A, Weaver AL, et al. Incidence of hidradenitis suppurativa and associated factors: a population-based study of Olmsted County, Minnesota. J Invest Dermatol. 2013;133:97-103. doi: 10.1038/jid.2012.255
19. Sachdeva M, Shah M, Alavi A. Race-specific prevalence of hidradenitis suppurativa. J Cutan Med Surg. 2021;25:177-187. doi: 10.1177/1203475420972348
20. Vaidya T, Vangipuram R, Alikhan A. Examining the race-specific prevalence of hidradenitis suppurativa at a large academic center; results from a retrospective chart review. Dermatol Online J. 2017;23:13030/qt9xc0n0z1. doi: 10.5070/D3236035391
21. Price KN, Hsiao JL, Shi VY. Race and ethnicity gaps in global hidradenitis suppurativa clinical trials. Dermatology. 2021;237:97-102. doi: 10.1159/000504911
22. Schrader AM, Deckers IE, van der Zee HH, et al. Hidradenitis suppurativa: a retrospective study of 846 Dutch patients to identify factors associated with disease severity. J Am Acad Dermatol. 2014;71:460-467. doi: 10.1016/j.jaad.2014.04.001
23. Frew JW, Vekic DA, Wood J, et al. A systematic review and critical evaluation of reported pathogenic sequence variants in hidradenitis suppurativa. Br J Dermatol. 2017;177:987-998. doi: 10.1111/bjd.15441
24. Wolk K, Join-Lambert O, Sabat R. Aetiology and pathogenesis of hidradenitis suppurativa. Br J Dermatol. 2020;183:999-1010. doi: 10.1111/bjd.19556
25. Goldburg SR, Strober BE, Payette MJ. Hidradenitis suppurativa: epidemiology, clinical presentation, and pathogenesis. J Am Acad Dermatol. 2020;82:1045-1058. doi: 10.1016/j.jaad.2019.08.090
26. Sabat R, Chanwangpong A, Schneider-Burrus S, et al. Increased prevalence of metabolic syndrome in patients with acne inversa. PloS One. 2012;7:e31810. doi: 10.1371/journal.pone.0031810
27. Loh TY, Hendricks AJ, Hsiao JL, et al. Undergarment and fabric selection in the management of hidradenitis suppurativa. Dermatology. 2021;237:119-124. doi: 10.1159/000501611
28. Rodríguez-Zuñiga MJM, García-Perdomo HA, Ortega-Loayza AG. Association between hidradenitis suppurativa and metabolic syndrome: a systematic review and meta-analysis. Actas Dermosifiliogr (Engl Ed). 2019;110:279-288. doi: 10.1016/j.ad.2018.10.020
29. Walker JM, Garcet S, Aleman JO, et al. Obesity and ethnicity alter gene expression in skin. Sci Rep. 2020;10:14079. doi: 10.1038/s41598-020-70244-2.
30. Boer J, Nazary M, Riis PT. The role of mechanical stress in hidradenitis suppurativa. Dermatol Clin. 2016;34:37-43. doi: 10.1016/j.det.2015.08.011
31. Vossen ARJV, van Straalen KR, Swolfs EFH, et al. Nicotine dependency and readiness to quit smoking among patients with hidradenitis suppurativa. Dermatology. 2021;237:383-385. doi: 10.1159/000514028
32. Kromann CB, Deckers IE, Esmann S, et al. Risk factors, clinical course and long-term prognosis in hidradenitis suppurativa: a cross-sectional study. Br J Dermatol. 2014;171:819-824. doi: 10.1111/bjd.13090
33. Clark AK, Quinonez RL, Saric S, et al. Hormonal therapies for hidradenitis suppurativa: review. Dermatol Online J. 2017;23:13030/qt6383k0n4. doi: 10.5070/D32310036990
34. Saric-Bosanac S, Clark AK, Sivamani RK, et al. The role of hypothalamus-pituitary-adrenal (HPA)-like axis in inflammatory pilosebaceous disorders. Dermatol Online J. 2020;26:13030/qt8949296f. doi: 10.5070/D3262047430
35. Riis PT, Ring HC, Themstrup L, et al. The role of androgens and estrogens in hidradenitis suppurativa – a systematic review. Acta Dermatovenerol Croat. 2016;24:239-249.
36. Hsiao JL, Antaya RJ, Berger T, et al. Hidradenitis suppurativa and concomitant pyoderma gangrenosum: a case series and literature review. Arch Dermatol. 2010;146:1265-1270. doi: 10.1001/archdermatol.2010.328
37. Ah-Weng A, Langtry JAA, Velangi S, et al. Pyoderma gangrenosum associated with hidradenitis suppurativa. Clin Exp Dermatol. 2005;30:669-671. doi: 10.1111/j.1365-2230.2005.01897.x
38. Kirthi S, Hellen R, O’Connor R, et al. Hidradenitis suppurativa and Crohn’s disease: a case series. Ir Med J. 2017;110:618.
39. Dumont LM, Landman C, Sokol H, et al; CD-HS Study Group. Increased risk of permanent stoma in Crohn’s disease associated with hidradenitis suppurativa: a case-control study. Aliment Pharmacol Ther. 2020;52:303-310. doi: 10.1111/apt.15863
40. Marzano AV, Ceccherini I, Gattorno M, et al. Association of pyoderma gangrenosum, acne, and suppurative hidradenitis (PASH) shares genetic and cytokine profiles with other autoinflammatory diseases. Medicine (Baltimore). 2014;93:e187. doi: 10.1097/MD.0000000000000187.
41. Vinkel C, Thomsen SF. Hidradenitis suppurativa: causes, features, and current treatments. J Clin Aesthet Dermatol. 2018;11:17-23.
42. Wipperman J, Bragg DA, Litzner B. Hidradenitis suppurativa: rapid evidence review. Am Fam Physician. 2019;100:562-569.
43. Theut Riis P, Pedersen OB, Sigsgaard V, et al. Prevalence of patients with self-reported hidradenitis suppurativa in a cohort of Danish blood donors: a cross-sectional study. Br J Dermatol. 2019;180:774-781. doi: 10.1111/bjd.16998.
44. Dufour DN, Emtestam L, Jemec GB. Hidradenitis suppurativa: a common and burdensome, yet under-recognised, inflammatory skin disease. Postgrad Med J. 2014;90:216-221; doi: 10.1136/postgradmedj-2013-131994
45. Vinding GR, Miller IM, Zarchi K, et al. The prevalence of inverse recurrent suppuration: a population-based study of possible hidradenitis suppurativa. Br J Dermatol. 2014;170:884-889. doi: 10.1111/bjd.12787
46. Bassas-Vila J, González Lama Y. Hidradenitis suppurativa and perianal Crohn disease: differential diagnosis. Actas Dermosifiliogr. 2016;107(suppl 2):27-31. doi: 10.1016/S0001-7310(17) 30006-6
47. Valour F, Sénéchal A, Dupieux C, et al. Actinomycosis: etiology, clinical features, diagnosis, treatment, and management. Infect Drug Resist. 2014;7:183-197. doi: 10.2147/IDR.S39601
48. Fuchs W, Brockmeyer NH. Sexually transmitted infections. J Dtsch Dermatol Ges. 2014;12:451-463. doi: 10.1111/ddg.12310
49. Hap W, Frejlich E, Rudno-Rudzińska J, et al. Pilonidal sinus: finding the righttrack for treatment. Pol Przegl Chir. 2017;89:68-75. doi: 10.5604/01.3001.0009.6009
50. Al-Hamdi KI, Saadoon AQ. Acne onglobate of the scalp. Int J Trichology. 2020;12:35-37. doi: 10.4103/ijt.ijt_117_19
51. Balestra A, Bytyci H, Guillod C, et al. A case of ulceroglandular tularemia presenting with lymphadenopathy and an ulcer on a linear morphoea lesion surrounded by erysipelas. Int Med Case Rep J. 2018;11:313-318. doi: 10.2147/IMCRJ.S178561
52. Ibler KS, Kromann CB. Recurrent furunculosis – challenges and management: a review. Clin Cosmet Investig Dermatol. 2014;7:59-64. doi: 10.2147/CCID.S35302
53. Ingram JR, Hadjieconomou S, Piguet V. Development of core outcome sets in hidradenitis suppurativa: systematic review of outcome measure instruments to inform the process. Br J Dermatol. 2016;175:263-272. doi: 10.1111/bjd.14475
54. Zouboulis CC, Tzellos T, Kyrgidis A, et al; European Hidradenitis Suppurativa Foundation Investigator Group. Development and validation of the International Hidradenitis Suppurativa Severity Score System (I4), a novel dynamic scoring system to assess HS severity. Br J Dermatol. 2017;177:1401-1409. doi: 10.1111/bjd.15748
55. Hidradenitis Suppurativa Clinical Resource. Hidradenitis suppurativa stages: Hurley Staging System. www.hsdiseasesource.com/hs-disease-staging. Accessed October 11, 2022.
56. Ovadja ZN, Schuit MM, van der Horst CMAM, et al. Inter- and interrater reliability of Hurley staging for hidradenitis suppurativa. Br J Dermatol. 2019;181:344-349. doi: 10.1111/bjd.17588
57. Wortsman X, Jemec GBE. Real-time compound imaging ultrasound of hidradenitis suppurativa. Dermatol Surg. 2007;33:1340-1342. doi: 10.1111/j.1524-4725.2007.33286.x
58. Napolitano M, Calzavara-Pinton PG, Zanca A, et al. Comparison of clinical and ultrasound scores in patients with hidradenitis suppurativa: results from an Italian ultrasound working group. J Eur Acad Dermatol Venereol. 2019;33:e84-e87. doi: 10.1111/jdv.15235
59. Bukvić Mokos Z, Miše J, Balić A, et al. Understanding the relationship between smoking and hidradenitis suppurativa. Acta Dermatovenerol Croat. 2020;28:9-13.
60. Shavit E, Dreiher J, Freud T, et al. Psychiatric comorbidities in 3207 patients with hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2015;29:371-376. doi: 10.1111/jdv.12567
61. Kouris A, Platsidaki E, Christodoulou C, et al. Quality of life and psychosocial implications in patients with hidradenitis suppurativa. Dermatology. 2016;232:687-691. doi: 10.1159/000453355
62 Ingram JR, Woo PN, Chua SL, et al. Interventions for hidradenitis suppurativa: a Cochrane systematic review incorporating GRADE assessment of evidence quality. Br J Dermatol. 2016;174:970-978. doi: 10.1111/bjd.14418
63. Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part II: topical, intralesional, and systemic medical management. J Am Acad Dermatol. 2019;81:91-101. doi: 10.1016/j.jaad.2019.02.068
64. Gulliver W, Zouboulis CC, Prens E, et al. Evidence-based approach to the treatment of hidradenitis suppurativa/acne inversa, based on the European guidelines for hidradenitis suppurativa. Rev Endocr Metab Disord. 2016;17:343-351. doi: 10.1007/s11154-016-9328-5
65. Vena GA, Cassano N. Drug focus: adalimumab in the treatment of moderate to severe psoriasis. Biologics. 2007;1:93-103.
66. Kimball AB, Kerdel F, Adams D, et al. Adalimumab for the treatment of moderate to severe hidradenitis suppurativa: a parallel randomized trial. Ann Intern Med. 2012;157:846-55. doi: 10.7326/0003-4819-157-12-201212180-00004
67. Zouboulis CC, Okun MM, Prens EP, et al. Long-term adalimumab efficacy in patients with moderate-to-severe hidradenitis suppurativa/acne inversa: 3-year results of a phase 3 open-label extension study. J Am Acad Dermatol. 2019;80:60-69.e2. doi: 10.1016/j.jaad.2018.05.040
68. Jemec GB, Wendelboe P. Topical clindamycin versus systemic tetracycline in the treatment of hidradenitis suppurativa. J Am Acad Dermatol. 1998;39:971-974. doi: 10.1016/s0190-9622(98)70272-5
69. Gener G, Canoui-Poitrine F, Revuz JE, et al. Combination therapy with clindamycin and rifampicin for hidradenitis suppurativa: a series of 116 consecutive patients. Dermatology. 2009;219:148-154. doi: 10.1159/000228334
70. Grant A, Gonzalez T, Montgomery MO, et al. Infliximab therapy for patients with moderate to severe hidradenitis suppurativa: a randomized, double-blind, placebo-controlled crossover trial. J Am Acad Dermatol. 2010;62:205-217. doi: 10.1016/j.jaad.2009.06.050
71. Blok JL, Spoo JR, Leeman FWJ, et al. Skin-tissue-sparing excision with electrosurgical peeling (STEEP): a surgical treatment option for severe hidradenitis suppurativa Hurley stage II/III. J Eur Acad Dermatol Venereol. 2015;29:379-382. doi: 10.1111/jdv.12376
72. Mahmoud BH, Tierney E, Hexsel CL, et al. Prospective controlled clinical and histopathologic study of hidradenitis suppurativa treated with the long-pulsed neodymium:yttrium-aluminium-garnet laser. J Am Acad Dermatol. 2010;62:637-645. doi: 10.1016/j.jaad.2009.07.048
73. Tierney E, Mahmoud BH, Hexsel C, et al. Randomized control trial for the treatment of hidradenitis suppurativa with a neodymium-doped yttrium aluminium garnet laser. Dermatol Surg. 2009;35:1188-1198. doi: 10.1111/j.1524-4725.2009.01214.x
74. Hazen PG, Hazen BP. Hidradenitis suppurativa: successful treatment using carbon dioxide laser excision and marsupialization. Dermatol Surg. 2010;36:208-213. doi: 10.1111/j.1524-4725.2009.01427.x
75. van der Zee HH, Prens EP, Boer J. Deroofing: a tissue-saving surgical technique for the treatment of mild to moderate hidradenitis suppurativa lesions. J Am Acad Dermatol. 2010;63:475-480. doi: 10.1016/j.jaad.2009.12.018
76. Kimball AB, Okun MM, Williams DA, et al. Two phase 3 trials of adalimumab for hidradenitis suppurativa. N Engl J Med. 2016;375:422-434. doi: 10.1056/NEJMoa1504370. PMID: 27518661.
77. Adams DR, Yankura JA, Fogelberg AC, et al. Treatment of hidradenitis suppurativa with etanercept injection. Arch Dermatol. 2010;146:501-504. doi: 10.1001/archdermatol.2010.72
78. Tursi A. Concomitant hidradenitis suppurativa and pyostomatitis vegetans in silent ulcerative colitis successfully treated with golimumab. Dig Liver Dis. 2016;48:1511-1512. doi: 10.1016/j.dld.2016.09.010
79. Tzanetakou V, Kanni T, Giatrakou S, et al. Safety and efficacy of anakinra in severe hidradenitis suppurativa: a randomized clinical trial. JAMA Dermatol. 2016;152:52-59. doi: 10.1001/jamadermatol.2015.3903.
80. Romaní J, Vilarrasa E, Martorell A, et al. Ustekinumab with intravenous infusion: results in hidradenitis suppurativa. Dermatology. 2020;236:21-24. doi: 10.1159/000501075
81. Kane SV. Preparing for biologic or immunosuppressant therapy. Gastroenterol Hepatol (N Y). 2011;7:544-546.
82. Davis W, Vavilin I, Malhotra N. Biologic therapy in HIV: to screen or not to screen. Cureus. 2021;13:e15941. doi: 10.7759/cureus.15941
83. Papp KA, Haraoui B, Kumar D, et al. Vaccination guidelines for patients with immune-mediated disorders on immunosuppressive therapies. J Cutan Med Surg. 2019;23:50-74. doi: 10.1177/1203475418811335
84. Clemmensen OJ. Topical treatment of hidradenitis suppurativa with clindamycin. Int J Dermatol. 1983;22:325-328. doi: 10.1111/j.1365-4362.1983.tb02150.x
85. Hunger RE, Laffitte E, Läuchli S, et al. Swiss practice recommendations for the management of hidradenitis suppurativa/acne inversa. Dermatology. 2017;233:113-119. doi: 10.1159/000477459
86. Zouboulis CC, Bechara FG, Dickinson-Blok JL, et al. Hidradenitis suppurativa/acne inversa: a practical framework for treatment optimization - systematic review and recommendations from the HS ALLIANCE working group. J Eur Acad Dermatol Venereol. 2019;33:19-31. doi: 10.1111/jdv.15233
87. Riis PT, Boer J, Prens EP, et al. Intralesional triamcinolone for flares of hidradenitis suppurativa (HS): a case series. J Am Acad Dermatol. 2016;75:1151-1155. doi: 10.1016/j.jaad.2016.06.049
88. Fajgenbaum K, Crouse L, Dong L, et al. Intralesional triamcinolone may not be beneficial for treating acute hidradenitis suppurativa lesions: a double-blind, randomized, placebo-controlled trial. Dermatol Surg. 2020;46:685-689. doi: 10.1097/DSS.0000000000002112
89. Mortimer PS, Dawber RP, Gales MA, et al. A double-blind controlled cross-over trial of cyproterone acetate in females with hidradenitis suppurativa. Br J Dermatol. 1986;115:263-268. doi: 10.1111/j.1365-2133.1986.tb05740.x
90. Kraft JN, Searles GE. Hidradenitis suppurativa in 64 female patients: retrospective study comparing oral antibiotics and antiandrogen therapy. J Cutan Med Surg. 2007;11:125-131. doi: 10.2310/7750.2007.00019
91. Lee A, Fischer G. A case series of 20 women with hidradenitis suppurativa treated with spironolactone. Australas J Dermatol. 2015;56:192-196. doi: 10.1111/ajd.12362
92. Khandalavala BN. A disease-modifying approach for advanced hidradenitis suppurativa (regimen with metformin, liraglutide, dapsone, and finasteride): a case report. Case Rep Dermatol. 2017;9:70-78. doi: 10.1159/000473873
93. Verdolini R, Clayton N, Smith A, et al. Metformin for the treatment of hidradenitis suppurativa: a little help along the way. J Eur Acad Dermatol Venereol. 2013;27:1101-1108. doi: 10.1111/j.1468-3083.2012.04668.x
94. Khandalavala BN, Do MV. Finasteride in hidradenitis suppurativa: a “male” therapy for a predominantly “female” disease. J Clin Aesthet Dermatol. 2016;9:44-50.
95. Mota F, Machado S, Selores M. Hidradenitis suppurativa in children treated with finasteride-a case series. Pediatr Dermatol. 2017;34:578-583. doi: 10.1111/pde.13216
96. Doménech C, Matarredona J, Escribano-Stablé JC, et al. Facial hidradenitis suppurativa in a 28-year-old male responding to finasteride. Dermatology. 2012;224:307-308. doi: 10.1159/000339477
97. Patel N, McKenzie SA, Harview CL, et al. Isotretinoin in the treatment of hidradenitis suppurativa: a retrospective study. J Dermatolog Treat. 2021;32:473-475. doi: 10.1080/09546634.2019.1670779
98. Boer J, van Gemert MJ. Long-term results of isotretinoin in the treatment of 68 patients with hidradenitis suppurativa. J Am Acad Dermatol. 1999;40:73-76. doi: 10.1016/s0190-9622(99) 70530-x
99. Huang CM, Kirchhof MG. A new perspective on isotretinoin treatment of hidradenitis suppurativa: a retrospective chart review of patient outcomes. Dermatology. 2017;233:120-125. doi: 10.1159/000477207
100. Prens LM, Huizinga J, Janse IC. Surgical outcomes and the impact of major surgery on quality of life, activity impairment and sexual health in hidradenitis suppurativa patients: a prospective single centre study. J Eur Acad Dermatol Venereol. 2019;33:1941-1946. doi: 10.1111/jdv.15706
101. Ritz JP, Runkel N, Haier J, et al. Extent of surgery and recurrence rate of hidradenitis suppurativa. Int J Colorectal Dis. 1998;13:164-168. doi: 10.1007/s003840050159
102. Mehdizadeh A, Hazen PG, Bechara FG, et al. Recurrence of hidradenitis suppurativa after surgical management: a systematic review and meta-analysis. J Am Acad Dermatol. 2015;73(5 suppl 1):S70-S77. doi: 10.1016/j.jaad.2015.07.044.
103. Smith HS, Chao JD, Teitelbaum J. Painful hidradenitis suppurativa. Clin J Pain. 2010;26:435-444. doi: 10.1097/AJP.0b013e3181ceb80c
104. Horváth B, Janse IC, Sibbald GR. Pain management in patients with hidradenitis suppurativa. J Am Acad Dermatol. 2015;73(5 suppl 1):S47-S51. doi: 10.1016/j.jaad.2015.07.046
105. Kimball AB, Sobell JM, Zouboulis CC, et al. HiSCR (Hidradenitis Suppurativa Clinical Response): a novel clinical endpoint to evaluate therapeutic outcomes in patients with hidradenitis suppurativa from the placebo-controlled portion of a phase 2 adalimumab study. J Eur Acad Dermatol Venereol. 2016;30:989-994. doi: 10.1111/jdv.13216
106. Kimball AB, Jemec GB, Yang M, et al. Assessing the validity, responsiveness and meaningfulness of the Hidradenitis Suppurativa Clinical Response (HiSCR) as the clinical endpoint for hidradenitis suppurativa treatment. Br J Dermatol. 2014;171:1434-1442. doi: 10.1111/bjd.13270
PRACTICE RECOMMENDATIONS
› Screen patients with hidradenitis suppurativa (HS) for depression, anxiety, history of smoking, metabolic syndrome, and type 2 diabetes. A
› Look into early surgical and dermatology referrals for patients with mild diffused, moderate, and severe disease. B
› Consider biopsy to rule out skin cancer in patients with severe and longstanding HS refractory to treatment. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
A worsening abdominal rash
A 48-YEAR-OLD WOMAN presented to Dermatology for evaluation of a 6-cm abdominal lesion that had been present for 5 weeks (FIGURE 1). The lesion was originally about the size of a quarter, but it started to enlarge after treatment of an asthma exacerbation with a 4-day course of prednisone. It continued to grow after another physician, likely presuming the lesion was a corticosteroid-responsive dermatosis (eg, nummular eczema, granuloma annulare, or erythema annulare centrifugum), prescribed a 2-week trial of clobetasol ointment. Physical examination revealed a mildly pruritic, 6-cm erythematous plaque with scaly, annular, concentric rings on the left lower abdomen. The patient had no travel history.
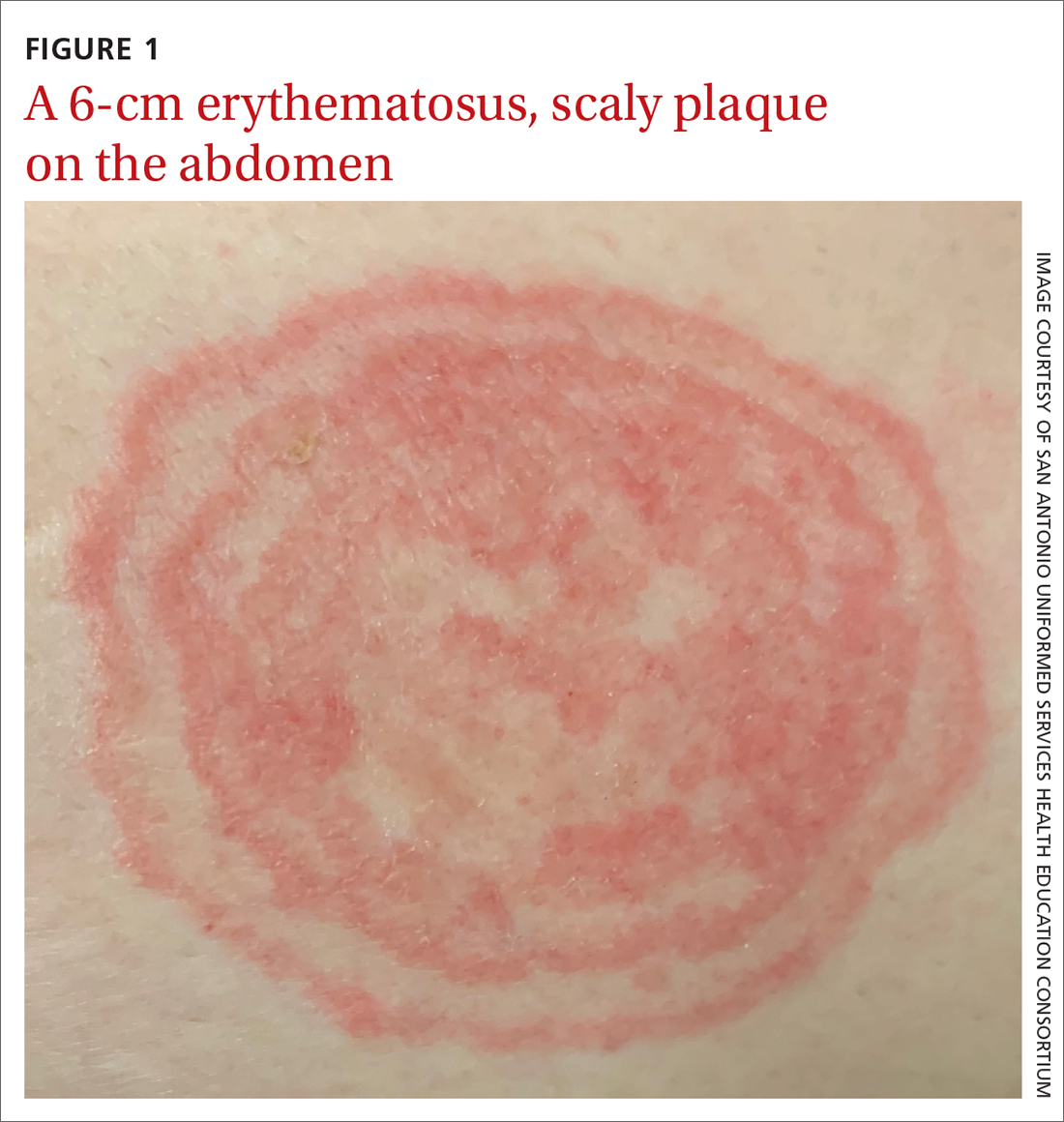
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Tinea incognito
The patient was given a diagnosis of tinea incognito, a form of tinea corporis that is exacerbated by the inappropriate use of corticosteroids in the management of a cutaneous fungal infection.1 Furthermore, this patient’s case was consistent with tinea pseudoimbricata, a variant of tinea incognito. Tinea pseudoimbricata is characterized by striking concentric scaly rings that mimic tinea imbricata, a fungal infection caused by the dermatophyte Trichophyton concentricum, which is commonly found in tropical areas.2
A common infection is alteredby steroid use
Tinea corporis has a relatively high prevalence. Approximately 10% to 20% of the world population is affected by fungal skin infections.3
T rubrum is the most common cause of tinea corporis. Other causes include T tonsurans, T interdigitale, T violaceum, Microsporum canis, M gypseum, and M audouinii.
Tinea corporis can be acquired through direct contact with an infected person, animal, or fomite. It may also be acquired through autoinoculation from another area of the body containing a dermatophyte fungal infection. Tinea corporis lesions are usually pruritic, erythematous, annular plaques with overlying scale and central clearing.
How steroid use can change the picture. Treatment with corticosteroids is ineffective for fungal skin infections and causes immunosuppression, allowing the fungus to thrive. This patient had been treated with a topical steroid (clobetasol) for the abdominal lesion caused by tinea corporis, as well as an oral steroid (prednisone) for an asthma exacerbation. These steroid treatments caused the abdominal lesion to morph from the typical appearance of tinea corporis—classically an annular erythematous plaque with overlying scale and central clearing—to an erythematous plaque with striking concentric scaly rings.
Continue to: Clinical exam can provide clues; KOH examination can reveal the Dx
Clinical exam can provide clues; KOH examination can reveal the Dx
The differential diagnosis for an annular skin lesion includes not only tinea corporis, but also superficial erythema annulare centrifugum, pityriasis rosea, granuloma annulare, subacute cutaneous lupus erythematosus (SCLE), and nummular eczema.
Superficial erythema annulare centrifugum, like tinea corporis, has scale. But the location of the scale sets the 2 apart. Superficial erythema annulare centrifugum lesions have a central trailing scale, whereas tinea corporis lesions have a peripheral leading scale.4
Pityriasis rosea forms multiple lesions in a “Christmas tree” pattern on the trunk, sometimes beginning with a single herald patch. Our patient’s single lesion with concentric scaly rings was inconsistent with the distribution and quality of the lesions in pityriasis rosea.4
Granuloma annulare lesions are smooth, nonscaly plaques that are most often seen on the dorsal hands and feet. The scaly manifestation of our patient’s lesion was not consistent with this diagnosis.4
SCLE lesions are typically photodistributed on sun-exposed skin (eg, the neck, upper trunk, or arms), whereas our patient’s lesion involved a sun-protected site.4
Continue to: Nummular eczema
Nummular eczema can be differentiated from tinea corporis by potassium hydroxide (KOH) examination. Nummular eczema is characterized by a negative KOH exam and response to topical corticosteroids.4
Performing a KOH examination, using the skin scrapings from the active border of a plaque, is useful on any lesion with potential fungal etiology. If the cause is indeed a dermatophyte infection, segmented fungal hyphae will be seen under light microscopy (FIGURE 2).1 If a KOH examination is not feasible, a skin scraping can be performed with a surgical scalpel blade and collected in a sterile urine cup for stain and culture at a qualified laboratory.
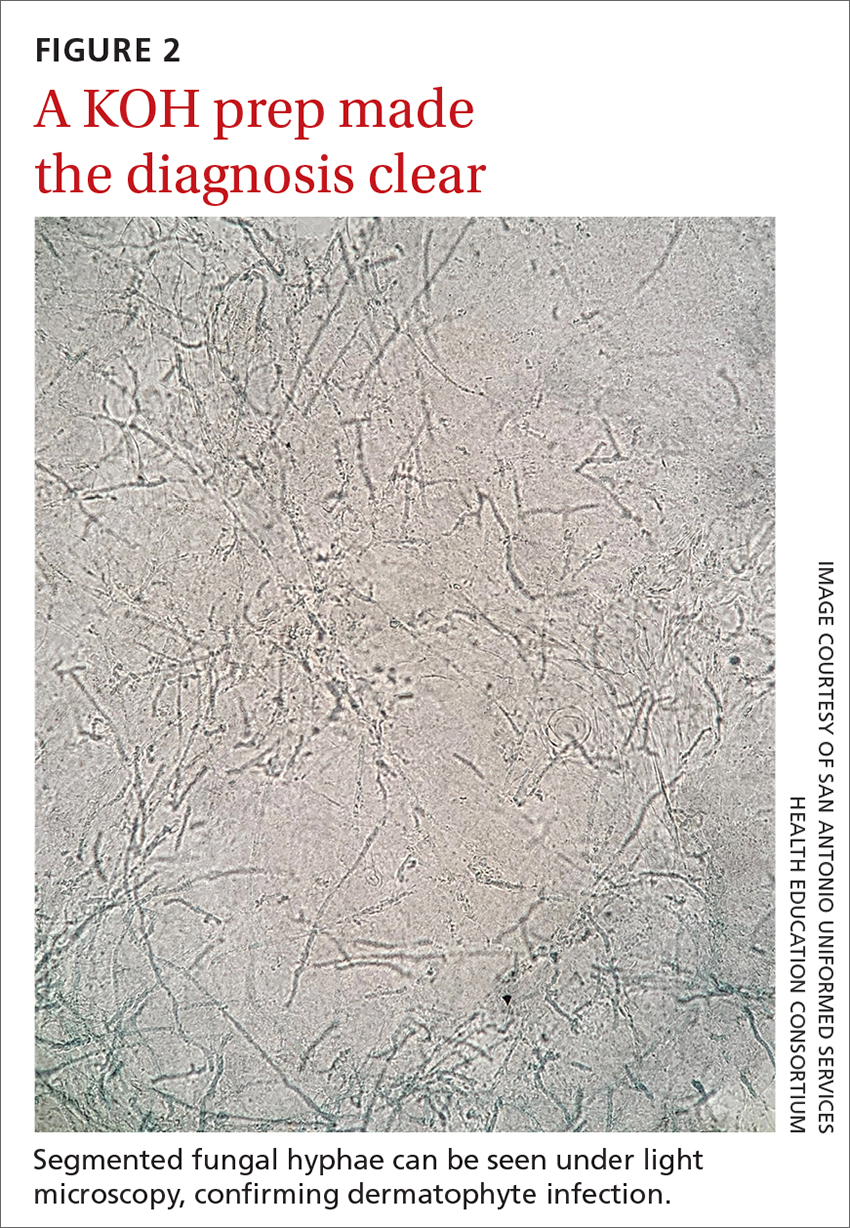
Topical and oral antifungal medications combat dermatophyte fungi
Treatments for cutaneous infections caused by dermatophyte fungi, such as tinea corporis, include topical and oral antifungals. The choice of agent depends on the extent of the disease.
Limited, localized disease can be treated topically with allylamines (terbinafine, naftifine) or imidazoles (clotrimazole). Other topical agents, such as butenafine, ciclopirox, and tolnaftate, also may be used.
Extensive disease, or tinea infection of vellus hairs, may require treatment with oral antifungal medications, such as the azoles (itraconazole, fluconazole), allylamines (terbinafine), or griseofulvin. Systemic therapy with oral antifungals has been associated with liver damage; therefore, oral therapy should not be used in patients with liver disease and liver enzymes should be monitored when appropriate.5 Nystatin is not effective in treating dermatophyte fungal infections.1
One complication of the inappropriate use of steroids on a dermatophyte infection is an increased risk of the fungus extending from the superficial skin into the hair follicles in the dermis, resulting in a condition known as Majocchi granuloma. Follicular infection is more severe and requires oral antifungal medication, such as terbinafine, itraconazole, fluconazole, or griseofulvin.1
Our patient was treated with terbinafine 250 mg/d for 4 weeks, due to the possibility of follicular infection. After the completion of 4 weeks of therapy, the patient’s cutaneous symptoms had resolved.
1. Goldstein AO, Goldstein BG. Dermatophyte (tinea) infections. In: Ofori AO, ed. UpToDate. 2022. Updated November 8, 2022. Accessed November 23, 2022. www.uptodate.com/contents/dermatophyte-tinea-infections
2. Lederman E, Craft N, Burgin S. Tinea imbricata in adult. VisualDx. Updated September 24, 2018. Accessed November 23, 2022. www.visualdx.com/visualdx/diagnosis/?moduleId=101&diagnosisId=52399
3. El-Gohary M, van Zuuren EJ, Fedorowicz Z, et al. Topical antifungal treatments for tinea cruris and tinea corporis. Cochrane Database Syst Rev. 2014:CD009992. doi: 10.1002/14651858.CD009992.pub2
4. Unwala R. Approach to the patient with annular skin lesions. In: Ofori AO, ed. UpToDate. 2022. Updated September 7, 2022. Accessed November 23, 2022. www.uptodate.com/contents/approach-to-the-patient-with-annular-skin-lesions
5. Wong V, High W, Burgin S. Tinea corporis in adult. VisualDx. Updated March 24, 2019. Accessed November 23, 2022. www.visualdx.com/visualdx/diagnosis/?moduleId=101&diagnosisId=52396#Therapy
A 48-YEAR-OLD WOMAN presented to Dermatology for evaluation of a 6-cm abdominal lesion that had been present for 5 weeks (FIGURE 1). The lesion was originally about the size of a quarter, but it started to enlarge after treatment of an asthma exacerbation with a 4-day course of prednisone. It continued to grow after another physician, likely presuming the lesion was a corticosteroid-responsive dermatosis (eg, nummular eczema, granuloma annulare, or erythema annulare centrifugum), prescribed a 2-week trial of clobetasol ointment. Physical examination revealed a mildly pruritic, 6-cm erythematous plaque with scaly, annular, concentric rings on the left lower abdomen. The patient had no travel history.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Tinea incognito
The patient was given a diagnosis of tinea incognito, a form of tinea corporis that is exacerbated by the inappropriate use of corticosteroids in the management of a cutaneous fungal infection.1 Furthermore, this patient’s case was consistent with tinea pseudoimbricata, a variant of tinea incognito. Tinea pseudoimbricata is characterized by striking concentric scaly rings that mimic tinea imbricata, a fungal infection caused by the dermatophyte Trichophyton concentricum, which is commonly found in tropical areas.2
A common infection is alteredby steroid use
Tinea corporis has a relatively high prevalence. Approximately 10% to 20% of the world population is affected by fungal skin infections.3
T rubrum is the most common cause of tinea corporis. Other causes include T tonsurans, T interdigitale, T violaceum, Microsporum canis, M gypseum, and M audouinii.
Tinea corporis can be acquired through direct contact with an infected person, animal, or fomite. It may also be acquired through autoinoculation from another area of the body containing a dermatophyte fungal infection. Tinea corporis lesions are usually pruritic, erythematous, annular plaques with overlying scale and central clearing.
How steroid use can change the picture. Treatment with corticosteroids is ineffective for fungal skin infections and causes immunosuppression, allowing the fungus to thrive. This patient had been treated with a topical steroid (clobetasol) for the abdominal lesion caused by tinea corporis, as well as an oral steroid (prednisone) for an asthma exacerbation. These steroid treatments caused the abdominal lesion to morph from the typical appearance of tinea corporis—classically an annular erythematous plaque with overlying scale and central clearing—to an erythematous plaque with striking concentric scaly rings.
Continue to: Clinical exam can provide clues; KOH examination can reveal the Dx
Clinical exam can provide clues; KOH examination can reveal the Dx
The differential diagnosis for an annular skin lesion includes not only tinea corporis, but also superficial erythema annulare centrifugum, pityriasis rosea, granuloma annulare, subacute cutaneous lupus erythematosus (SCLE), and nummular eczema.
Superficial erythema annulare centrifugum, like tinea corporis, has scale. But the location of the scale sets the 2 apart. Superficial erythema annulare centrifugum lesions have a central trailing scale, whereas tinea corporis lesions have a peripheral leading scale.4
Pityriasis rosea forms multiple lesions in a “Christmas tree” pattern on the trunk, sometimes beginning with a single herald patch. Our patient’s single lesion with concentric scaly rings was inconsistent with the distribution and quality of the lesions in pityriasis rosea.4
Granuloma annulare lesions are smooth, nonscaly plaques that are most often seen on the dorsal hands and feet. The scaly manifestation of our patient’s lesion was not consistent with this diagnosis.4
SCLE lesions are typically photodistributed on sun-exposed skin (eg, the neck, upper trunk, or arms), whereas our patient’s lesion involved a sun-protected site.4
Continue to: Nummular eczema
Nummular eczema can be differentiated from tinea corporis by potassium hydroxide (KOH) examination. Nummular eczema is characterized by a negative KOH exam and response to topical corticosteroids.4
Performing a KOH examination, using the skin scrapings from the active border of a plaque, is useful on any lesion with potential fungal etiology. If the cause is indeed a dermatophyte infection, segmented fungal hyphae will be seen under light microscopy (FIGURE 2).1 If a KOH examination is not feasible, a skin scraping can be performed with a surgical scalpel blade and collected in a sterile urine cup for stain and culture at a qualified laboratory.

Topical and oral antifungal medications combat dermatophyte fungi
Treatments for cutaneous infections caused by dermatophyte fungi, such as tinea corporis, include topical and oral antifungals. The choice of agent depends on the extent of the disease.
Limited, localized disease can be treated topically with allylamines (terbinafine, naftifine) or imidazoles (clotrimazole). Other topical agents, such as butenafine, ciclopirox, and tolnaftate, also may be used.
Extensive disease, or tinea infection of vellus hairs, may require treatment with oral antifungal medications, such as the azoles (itraconazole, fluconazole), allylamines (terbinafine), or griseofulvin. Systemic therapy with oral antifungals has been associated with liver damage; therefore, oral therapy should not be used in patients with liver disease and liver enzymes should be monitored when appropriate.5 Nystatin is not effective in treating dermatophyte fungal infections.1
One complication of the inappropriate use of steroids on a dermatophyte infection is an increased risk of the fungus extending from the superficial skin into the hair follicles in the dermis, resulting in a condition known as Majocchi granuloma. Follicular infection is more severe and requires oral antifungal medication, such as terbinafine, itraconazole, fluconazole, or griseofulvin.1
Our patient was treated with terbinafine 250 mg/d for 4 weeks, due to the possibility of follicular infection. After the completion of 4 weeks of therapy, the patient’s cutaneous symptoms had resolved.
A 48-YEAR-OLD WOMAN presented to Dermatology for evaluation of a 6-cm abdominal lesion that had been present for 5 weeks (FIGURE 1). The lesion was originally about the size of a quarter, but it started to enlarge after treatment of an asthma exacerbation with a 4-day course of prednisone. It continued to grow after another physician, likely presuming the lesion was a corticosteroid-responsive dermatosis (eg, nummular eczema, granuloma annulare, or erythema annulare centrifugum), prescribed a 2-week trial of clobetasol ointment. Physical examination revealed a mildly pruritic, 6-cm erythematous plaque with scaly, annular, concentric rings on the left lower abdomen. The patient had no travel history.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Tinea incognito
The patient was given a diagnosis of tinea incognito, a form of tinea corporis that is exacerbated by the inappropriate use of corticosteroids in the management of a cutaneous fungal infection.1 Furthermore, this patient’s case was consistent with tinea pseudoimbricata, a variant of tinea incognito. Tinea pseudoimbricata is characterized by striking concentric scaly rings that mimic tinea imbricata, a fungal infection caused by the dermatophyte Trichophyton concentricum, which is commonly found in tropical areas.2
A common infection is alteredby steroid use
Tinea corporis has a relatively high prevalence. Approximately 10% to 20% of the world population is affected by fungal skin infections.3
T rubrum is the most common cause of tinea corporis. Other causes include T tonsurans, T interdigitale, T violaceum, Microsporum canis, M gypseum, and M audouinii.
Tinea corporis can be acquired through direct contact with an infected person, animal, or fomite. It may also be acquired through autoinoculation from another area of the body containing a dermatophyte fungal infection. Tinea corporis lesions are usually pruritic, erythematous, annular plaques with overlying scale and central clearing.
How steroid use can change the picture. Treatment with corticosteroids is ineffective for fungal skin infections and causes immunosuppression, allowing the fungus to thrive. This patient had been treated with a topical steroid (clobetasol) for the abdominal lesion caused by tinea corporis, as well as an oral steroid (prednisone) for an asthma exacerbation. These steroid treatments caused the abdominal lesion to morph from the typical appearance of tinea corporis—classically an annular erythematous plaque with overlying scale and central clearing—to an erythematous plaque with striking concentric scaly rings.
Continue to: Clinical exam can provide clues; KOH examination can reveal the Dx
Clinical exam can provide clues; KOH examination can reveal the Dx
The differential diagnosis for an annular skin lesion includes not only tinea corporis, but also superficial erythema annulare centrifugum, pityriasis rosea, granuloma annulare, subacute cutaneous lupus erythematosus (SCLE), and nummular eczema.
Superficial erythema annulare centrifugum, like tinea corporis, has scale. But the location of the scale sets the 2 apart. Superficial erythema annulare centrifugum lesions have a central trailing scale, whereas tinea corporis lesions have a peripheral leading scale.4
Pityriasis rosea forms multiple lesions in a “Christmas tree” pattern on the trunk, sometimes beginning with a single herald patch. Our patient’s single lesion with concentric scaly rings was inconsistent with the distribution and quality of the lesions in pityriasis rosea.4
Granuloma annulare lesions are smooth, nonscaly plaques that are most often seen on the dorsal hands and feet. The scaly manifestation of our patient’s lesion was not consistent with this diagnosis.4
SCLE lesions are typically photodistributed on sun-exposed skin (eg, the neck, upper trunk, or arms), whereas our patient’s lesion involved a sun-protected site.4
Continue to: Nummular eczema
Nummular eczema can be differentiated from tinea corporis by potassium hydroxide (KOH) examination. Nummular eczema is characterized by a negative KOH exam and response to topical corticosteroids.4
Performing a KOH examination, using the skin scrapings from the active border of a plaque, is useful on any lesion with potential fungal etiology. If the cause is indeed a dermatophyte infection, segmented fungal hyphae will be seen under light microscopy (FIGURE 2).1 If a KOH examination is not feasible, a skin scraping can be performed with a surgical scalpel blade and collected in a sterile urine cup for stain and culture at a qualified laboratory.

Topical and oral antifungal medications combat dermatophyte fungi
Treatments for cutaneous infections caused by dermatophyte fungi, such as tinea corporis, include topical and oral antifungals. The choice of agent depends on the extent of the disease.
Limited, localized disease can be treated topically with allylamines (terbinafine, naftifine) or imidazoles (clotrimazole). Other topical agents, such as butenafine, ciclopirox, and tolnaftate, also may be used.
Extensive disease, or tinea infection of vellus hairs, may require treatment with oral antifungal medications, such as the azoles (itraconazole, fluconazole), allylamines (terbinafine), or griseofulvin. Systemic therapy with oral antifungals has been associated with liver damage; therefore, oral therapy should not be used in patients with liver disease and liver enzymes should be monitored when appropriate.5 Nystatin is not effective in treating dermatophyte fungal infections.1
One complication of the inappropriate use of steroids on a dermatophyte infection is an increased risk of the fungus extending from the superficial skin into the hair follicles in the dermis, resulting in a condition known as Majocchi granuloma. Follicular infection is more severe and requires oral antifungal medication, such as terbinafine, itraconazole, fluconazole, or griseofulvin.1
Our patient was treated with terbinafine 250 mg/d for 4 weeks, due to the possibility of follicular infection. After the completion of 4 weeks of therapy, the patient’s cutaneous symptoms had resolved.
1. Goldstein AO, Goldstein BG. Dermatophyte (tinea) infections. In: Ofori AO, ed. UpToDate. 2022. Updated November 8, 2022. Accessed November 23, 2022. www.uptodate.com/contents/dermatophyte-tinea-infections
2. Lederman E, Craft N, Burgin S. Tinea imbricata in adult. VisualDx. Updated September 24, 2018. Accessed November 23, 2022. www.visualdx.com/visualdx/diagnosis/?moduleId=101&diagnosisId=52399
3. El-Gohary M, van Zuuren EJ, Fedorowicz Z, et al. Topical antifungal treatments for tinea cruris and tinea corporis. Cochrane Database Syst Rev. 2014:CD009992. doi: 10.1002/14651858.CD009992.pub2
4. Unwala R. Approach to the patient with annular skin lesions. In: Ofori AO, ed. UpToDate. 2022. Updated September 7, 2022. Accessed November 23, 2022. www.uptodate.com/contents/approach-to-the-patient-with-annular-skin-lesions
5. Wong V, High W, Burgin S. Tinea corporis in adult. VisualDx. Updated March 24, 2019. Accessed November 23, 2022. www.visualdx.com/visualdx/diagnosis/?moduleId=101&diagnosisId=52396#Therapy
1. Goldstein AO, Goldstein BG. Dermatophyte (tinea) infections. In: Ofori AO, ed. UpToDate. 2022. Updated November 8, 2022. Accessed November 23, 2022. www.uptodate.com/contents/dermatophyte-tinea-infections
2. Lederman E, Craft N, Burgin S. Tinea imbricata in adult. VisualDx. Updated September 24, 2018. Accessed November 23, 2022. www.visualdx.com/visualdx/diagnosis/?moduleId=101&diagnosisId=52399
3. El-Gohary M, van Zuuren EJ, Fedorowicz Z, et al. Topical antifungal treatments for tinea cruris and tinea corporis. Cochrane Database Syst Rev. 2014:CD009992. doi: 10.1002/14651858.CD009992.pub2
4. Unwala R. Approach to the patient with annular skin lesions. In: Ofori AO, ed. UpToDate. 2022. Updated September 7, 2022. Accessed November 23, 2022. www.uptodate.com/contents/approach-to-the-patient-with-annular-skin-lesions
5. Wong V, High W, Burgin S. Tinea corporis in adult. VisualDx. Updated March 24, 2019. Accessed November 23, 2022. www.visualdx.com/visualdx/diagnosis/?moduleId=101&diagnosisId=52396#Therapy
Researchers use AI to diagnose infantile hemangioma
a proof-of-concept study reported.
Early diagnosis of infantile hemangiomas “is essential, as there is a narrow window of opportunity to treat high-risk lesions,” April J. Zhang, MD, and coauthors noted in the study. “AI algorithms optimized for image classification through use of convolutional neural networks have been widely utilized to classify lesions in which images are readily standardized, such as skin cancers and onychomycosis.”
The results were published in Pediatric Dermatology.
Dr. Zhang, of the department of dermatology at the Medical College of Wisconsin, Milwaukee, and colleagues trained a convoluted neural network to diagnose infantile hemangiomas based on clinical images from pediatric dermatology patients treated at Children’s Wisconsin between 2002 and 2019.
They used Microsoft’s ResNet-50, a publicly available network architecture, to train a binary infantile hemangioma classifier to group images as infantile hemangiomas or non–infantile hemangiomas. The team randomly split data from the model into training, validation, and test groups.
The preliminary data set contained 14,811 images, about half of which were facial lesions. The training group of images achieved an accuracy of 61.5%. Next, Dr. Zhang and colleagues limited the data set to facial-only lesions and removed poor-quality images, which left 5,834 images in the final data set: 4,110 infantile hemangiomas and 1,724 non–infantile hemangiomas. This model achieved an overall accuracy of 91.7%, with a sensitivity of 93% and a specificity of 90.5%.
“Our study is the first to demonstrate the applicability of AI in the pediatric dermatology population,” the authors wrote. “With current nationwide shortages in pediatric dermatologists, AI has the potential to improve patient access and outcomes through enhanced rapid diagnostic capabilities.”
They acknowledged certain limitations of the study, including a data set with greater numbers of infantile hemangiomas, compared with non–infantile hemangiomas.
“Random oversampling of the non–infantile hemangioma data set was used to combat this but may lead to model overfitting, where a model performs well on its training data but is unable to generalize to new data,” they wrote. “As infantile hemangiomas are rarely biopsied, expert clinical diagnoses were used as the gold standard without pathologic confirmation.”
The authors reported having no financial disclosures.
a proof-of-concept study reported.
Early diagnosis of infantile hemangiomas “is essential, as there is a narrow window of opportunity to treat high-risk lesions,” April J. Zhang, MD, and coauthors noted in the study. “AI algorithms optimized for image classification through use of convolutional neural networks have been widely utilized to classify lesions in which images are readily standardized, such as skin cancers and onychomycosis.”
The results were published in Pediatric Dermatology.
Dr. Zhang, of the department of dermatology at the Medical College of Wisconsin, Milwaukee, and colleagues trained a convoluted neural network to diagnose infantile hemangiomas based on clinical images from pediatric dermatology patients treated at Children’s Wisconsin between 2002 and 2019.
They used Microsoft’s ResNet-50, a publicly available network architecture, to train a binary infantile hemangioma classifier to group images as infantile hemangiomas or non–infantile hemangiomas. The team randomly split data from the model into training, validation, and test groups.
The preliminary data set contained 14,811 images, about half of which were facial lesions. The training group of images achieved an accuracy of 61.5%. Next, Dr. Zhang and colleagues limited the data set to facial-only lesions and removed poor-quality images, which left 5,834 images in the final data set: 4,110 infantile hemangiomas and 1,724 non–infantile hemangiomas. This model achieved an overall accuracy of 91.7%, with a sensitivity of 93% and a specificity of 90.5%.
“Our study is the first to demonstrate the applicability of AI in the pediatric dermatology population,” the authors wrote. “With current nationwide shortages in pediatric dermatologists, AI has the potential to improve patient access and outcomes through enhanced rapid diagnostic capabilities.”
They acknowledged certain limitations of the study, including a data set with greater numbers of infantile hemangiomas, compared with non–infantile hemangiomas.
“Random oversampling of the non–infantile hemangioma data set was used to combat this but may lead to model overfitting, where a model performs well on its training data but is unable to generalize to new data,” they wrote. “As infantile hemangiomas are rarely biopsied, expert clinical diagnoses were used as the gold standard without pathologic confirmation.”
The authors reported having no financial disclosures.
a proof-of-concept study reported.
Early diagnosis of infantile hemangiomas “is essential, as there is a narrow window of opportunity to treat high-risk lesions,” April J. Zhang, MD, and coauthors noted in the study. “AI algorithms optimized for image classification through use of convolutional neural networks have been widely utilized to classify lesions in which images are readily standardized, such as skin cancers and onychomycosis.”
The results were published in Pediatric Dermatology.
Dr. Zhang, of the department of dermatology at the Medical College of Wisconsin, Milwaukee, and colleagues trained a convoluted neural network to diagnose infantile hemangiomas based on clinical images from pediatric dermatology patients treated at Children’s Wisconsin between 2002 and 2019.
They used Microsoft’s ResNet-50, a publicly available network architecture, to train a binary infantile hemangioma classifier to group images as infantile hemangiomas or non–infantile hemangiomas. The team randomly split data from the model into training, validation, and test groups.
The preliminary data set contained 14,811 images, about half of which were facial lesions. The training group of images achieved an accuracy of 61.5%. Next, Dr. Zhang and colleagues limited the data set to facial-only lesions and removed poor-quality images, which left 5,834 images in the final data set: 4,110 infantile hemangiomas and 1,724 non–infantile hemangiomas. This model achieved an overall accuracy of 91.7%, with a sensitivity of 93% and a specificity of 90.5%.
“Our study is the first to demonstrate the applicability of AI in the pediatric dermatology population,” the authors wrote. “With current nationwide shortages in pediatric dermatologists, AI has the potential to improve patient access and outcomes through enhanced rapid diagnostic capabilities.”
They acknowledged certain limitations of the study, including a data set with greater numbers of infantile hemangiomas, compared with non–infantile hemangiomas.
“Random oversampling of the non–infantile hemangioma data set was used to combat this but may lead to model overfitting, where a model performs well on its training data but is unable to generalize to new data,” they wrote. “As infantile hemangiomas are rarely biopsied, expert clinical diagnoses were used as the gold standard without pathologic confirmation.”
The authors reported having no financial disclosures.
FROM PEDIATRIC DERMATOLOGY
Parental atopic dermatitis, asthma linked to risk of AD in offspring
of life, an analysis of a large birth cohort found.
“The prevalence of AD in children has increased dramatically in recent years, and most studies reporting the impact of parental atopic history on AD are based on older data,” wrote the study authors, led by Cathal O’Connor, MD. “Given the recent interest in early intervention to prevent AD and other allergic diseases, enhanced early identification of infants at risk of AD is increasingly important.”
The detailed analysis of AD risk associated with parental atopy in early life “may help to risk stratify infants to optimize early interventions for prevention or early treatment of AD,” they wrote.
The study was published in Pediatric Dermatology.
For the analysis, Dr. O’Connor of the department of pediatrics and child health at University College Cork (Ireland) and colleagues conducted a secondary analysis of the Cork Babies After Scope: Evaluating the Longitudinal Impact Using Neurological and Nutritional Endpoints (BASELINE) Birth Cohort Study.
The study recruited 2,183 healthy first-born babies between August 2009 and October 2011 to examine the effects of environmental factors during pregnancy and infancy on childhood health and development. Skin barrier assessments were performed at birth, 2 months, 6 months, 12 months, and 24 months using a validated open chamber system to measure transepidermal water loss.
Parental atopy was self-reported at 2 months. Parents were asked at 2 months if the infant had an “itchy rash on the face or in the folds of the arms or legs,” as a screening question for AD. Experienced health care personnel used UK Working Party criteria to diagnose AD at 6, 12, and 24 months.
Complete data on AD status was available for 1,505 children in the cohort. Dr. O’Connor and colleagues calculated an overall AD prevalence of 18.6% at 6 months, 15.2% at 12 months, and 16.5% at 24 months.
Overall prevalence of AD was highest at 6 months. The study showed a similar or slightly higher impact of paternal atopy on offspring AD development, compared to maternal atopy.
Multivariable logistic regression analysis revealed that the odds of AD were 1.57 at 6 months and 1.66 at 12 months for maternal AD; 1.90 at 6 months and 1.85 at 24 months for paternal AD; 1.76 at 6 months and 1.75 at 12 months for maternal asthma; and 1.70 at 6 months, 1.86 at 12 months, and 1.99 at 24 months for paternal asthma.
“Parental allergic rhinitis was not associated with AD in offspring in the first 2 years, except for maternal rhinitis at 24 months [an adjusted odds ratio of 1.79],” the authors wrote. “The genetic predisposition to allergic rhinitis, given the key role of aeroallergen sensitization in its pathogenesis, may not be associated with early onset AD, but may have a greater impact in later onset or persistent AD.”
The authors acknowledged certain limitations of the study, including the fact that it was a secondary data analysis, and that parental AD, asthma, and rhinitis were self-reported, “which may reduce reliability and may contribute to the differences seen between the impact of maternal and paternal reported atopy on offspring,” they wrote. “Data on siblings were not captured, as participants in the study were first-born children. Filaggrin mutational analysis was not performed, which would have provided richer detail.”
Kelly M. Cordoro, MD, professor of dermatology and pediatrics at the University of California, San Francisco, who was asked to comment on the work, said that the study confirms the well-known association between parental atopy and the risk of atopy in offspring, which has been shown in several studies dating back decades.
“The authors try to parse risk based on maternal or paternal or biparental history of AD and/or asthma and/or rhinitis, but this type of nuanced analysis when diagnosis is based solely on parental report may be an over-reach,” she said.
“Given that this data supports the association between parental atopy and risk of AD in infants at various time points, the clinically relevant immediate next question is how can we leverage this knowledge to prevent onset of AD in infants at risk?” she said. “To date, interventions such as early introduction of emollients have been evaluated with mixed results.”
A recent Cochrane analysis concluded that, based on available data, skin care interventions such as emollient use during the first year of life in otherwise healthy infants is probably not effective for preventing eczema and may increase risk of skin infection.
“Effects of skin care interventions on risk of asthma are also uncertain,” said Dr. Cordoro, who is also chief of the division of pediatric dermatology at UCSF.
“In sum, this study offers additional data in support of the link between atopy in parents and offspring,” she said. “Understanding how to mitigate risk and prevent atopy requires unraveling of the complex interplay between genetic, environmental, immunologic, microbial and other factors. For now, dermatologists are unable to make broad evidence-based recommendations for otherwise healthy (i.e., with normal skin) but at-risk infants in terms of approaches to skin care that might prevent eczema and asthma.”
of life, an analysis of a large birth cohort found.
“The prevalence of AD in children has increased dramatically in recent years, and most studies reporting the impact of parental atopic history on AD are based on older data,” wrote the study authors, led by Cathal O’Connor, MD. “Given the recent interest in early intervention to prevent AD and other allergic diseases, enhanced early identification of infants at risk of AD is increasingly important.”
The detailed analysis of AD risk associated with parental atopy in early life “may help to risk stratify infants to optimize early interventions for prevention or early treatment of AD,” they wrote.
The study was published in Pediatric Dermatology.
For the analysis, Dr. O’Connor of the department of pediatrics and child health at University College Cork (Ireland) and colleagues conducted a secondary analysis of the Cork Babies After Scope: Evaluating the Longitudinal Impact Using Neurological and Nutritional Endpoints (BASELINE) Birth Cohort Study.
The study recruited 2,183 healthy first-born babies between August 2009 and October 2011 to examine the effects of environmental factors during pregnancy and infancy on childhood health and development. Skin barrier assessments were performed at birth, 2 months, 6 months, 12 months, and 24 months using a validated open chamber system to measure transepidermal water loss.
Parental atopy was self-reported at 2 months. Parents were asked at 2 months if the infant had an “itchy rash on the face or in the folds of the arms or legs,” as a screening question for AD. Experienced health care personnel used UK Working Party criteria to diagnose AD at 6, 12, and 24 months.
Complete data on AD status was available for 1,505 children in the cohort. Dr. O’Connor and colleagues calculated an overall AD prevalence of 18.6% at 6 months, 15.2% at 12 months, and 16.5% at 24 months.
Overall prevalence of AD was highest at 6 months. The study showed a similar or slightly higher impact of paternal atopy on offspring AD development, compared to maternal atopy.
Multivariable logistic regression analysis revealed that the odds of AD were 1.57 at 6 months and 1.66 at 12 months for maternal AD; 1.90 at 6 months and 1.85 at 24 months for paternal AD; 1.76 at 6 months and 1.75 at 12 months for maternal asthma; and 1.70 at 6 months, 1.86 at 12 months, and 1.99 at 24 months for paternal asthma.
“Parental allergic rhinitis was not associated with AD in offspring in the first 2 years, except for maternal rhinitis at 24 months [an adjusted odds ratio of 1.79],” the authors wrote. “The genetic predisposition to allergic rhinitis, given the key role of aeroallergen sensitization in its pathogenesis, may not be associated with early onset AD, but may have a greater impact in later onset or persistent AD.”
The authors acknowledged certain limitations of the study, including the fact that it was a secondary data analysis, and that parental AD, asthma, and rhinitis were self-reported, “which may reduce reliability and may contribute to the differences seen between the impact of maternal and paternal reported atopy on offspring,” they wrote. “Data on siblings were not captured, as participants in the study were first-born children. Filaggrin mutational analysis was not performed, which would have provided richer detail.”
Kelly M. Cordoro, MD, professor of dermatology and pediatrics at the University of California, San Francisco, who was asked to comment on the work, said that the study confirms the well-known association between parental atopy and the risk of atopy in offspring, which has been shown in several studies dating back decades.
“The authors try to parse risk based on maternal or paternal or biparental history of AD and/or asthma and/or rhinitis, but this type of nuanced analysis when diagnosis is based solely on parental report may be an over-reach,” she said.
“Given that this data supports the association between parental atopy and risk of AD in infants at various time points, the clinically relevant immediate next question is how can we leverage this knowledge to prevent onset of AD in infants at risk?” she said. “To date, interventions such as early introduction of emollients have been evaluated with mixed results.”
A recent Cochrane analysis concluded that, based on available data, skin care interventions such as emollient use during the first year of life in otherwise healthy infants is probably not effective for preventing eczema and may increase risk of skin infection.
“Effects of skin care interventions on risk of asthma are also uncertain,” said Dr. Cordoro, who is also chief of the division of pediatric dermatology at UCSF.
“In sum, this study offers additional data in support of the link between atopy in parents and offspring,” she said. “Understanding how to mitigate risk and prevent atopy requires unraveling of the complex interplay between genetic, environmental, immunologic, microbial and other factors. For now, dermatologists are unable to make broad evidence-based recommendations for otherwise healthy (i.e., with normal skin) but at-risk infants in terms of approaches to skin care that might prevent eczema and asthma.”
of life, an analysis of a large birth cohort found.
“The prevalence of AD in children has increased dramatically in recent years, and most studies reporting the impact of parental atopic history on AD are based on older data,” wrote the study authors, led by Cathal O’Connor, MD. “Given the recent interest in early intervention to prevent AD and other allergic diseases, enhanced early identification of infants at risk of AD is increasingly important.”
The detailed analysis of AD risk associated with parental atopy in early life “may help to risk stratify infants to optimize early interventions for prevention or early treatment of AD,” they wrote.
The study was published in Pediatric Dermatology.
For the analysis, Dr. O’Connor of the department of pediatrics and child health at University College Cork (Ireland) and colleagues conducted a secondary analysis of the Cork Babies After Scope: Evaluating the Longitudinal Impact Using Neurological and Nutritional Endpoints (BASELINE) Birth Cohort Study.
The study recruited 2,183 healthy first-born babies between August 2009 and October 2011 to examine the effects of environmental factors during pregnancy and infancy on childhood health and development. Skin barrier assessments were performed at birth, 2 months, 6 months, 12 months, and 24 months using a validated open chamber system to measure transepidermal water loss.
Parental atopy was self-reported at 2 months. Parents were asked at 2 months if the infant had an “itchy rash on the face or in the folds of the arms or legs,” as a screening question for AD. Experienced health care personnel used UK Working Party criteria to diagnose AD at 6, 12, and 24 months.
Complete data on AD status was available for 1,505 children in the cohort. Dr. O’Connor and colleagues calculated an overall AD prevalence of 18.6% at 6 months, 15.2% at 12 months, and 16.5% at 24 months.
Overall prevalence of AD was highest at 6 months. The study showed a similar or slightly higher impact of paternal atopy on offspring AD development, compared to maternal atopy.
Multivariable logistic regression analysis revealed that the odds of AD were 1.57 at 6 months and 1.66 at 12 months for maternal AD; 1.90 at 6 months and 1.85 at 24 months for paternal AD; 1.76 at 6 months and 1.75 at 12 months for maternal asthma; and 1.70 at 6 months, 1.86 at 12 months, and 1.99 at 24 months for paternal asthma.
“Parental allergic rhinitis was not associated with AD in offspring in the first 2 years, except for maternal rhinitis at 24 months [an adjusted odds ratio of 1.79],” the authors wrote. “The genetic predisposition to allergic rhinitis, given the key role of aeroallergen sensitization in its pathogenesis, may not be associated with early onset AD, but may have a greater impact in later onset or persistent AD.”
The authors acknowledged certain limitations of the study, including the fact that it was a secondary data analysis, and that parental AD, asthma, and rhinitis were self-reported, “which may reduce reliability and may contribute to the differences seen between the impact of maternal and paternal reported atopy on offspring,” they wrote. “Data on siblings were not captured, as participants in the study were first-born children. Filaggrin mutational analysis was not performed, which would have provided richer detail.”
Kelly M. Cordoro, MD, professor of dermatology and pediatrics at the University of California, San Francisco, who was asked to comment on the work, said that the study confirms the well-known association between parental atopy and the risk of atopy in offspring, which has been shown in several studies dating back decades.
“The authors try to parse risk based on maternal or paternal or biparental history of AD and/or asthma and/or rhinitis, but this type of nuanced analysis when diagnosis is based solely on parental report may be an over-reach,” she said.
“Given that this data supports the association between parental atopy and risk of AD in infants at various time points, the clinically relevant immediate next question is how can we leverage this knowledge to prevent onset of AD in infants at risk?” she said. “To date, interventions such as early introduction of emollients have been evaluated with mixed results.”
A recent Cochrane analysis concluded that, based on available data, skin care interventions such as emollient use during the first year of life in otherwise healthy infants is probably not effective for preventing eczema and may increase risk of skin infection.
“Effects of skin care interventions on risk of asthma are also uncertain,” said Dr. Cordoro, who is also chief of the division of pediatric dermatology at UCSF.
“In sum, this study offers additional data in support of the link between atopy in parents and offspring,” she said. “Understanding how to mitigate risk and prevent atopy requires unraveling of the complex interplay between genetic, environmental, immunologic, microbial and other factors. For now, dermatologists are unable to make broad evidence-based recommendations for otherwise healthy (i.e., with normal skin) but at-risk infants in terms of approaches to skin care that might prevent eczema and asthma.”
FROM PEDIATRIC DERMATOLOGY
Subset of patients with melanoma have very low mortality risk
Although melanoma is the most serious skin cancer, most patients do have high chances of survival. New research has now identified a subset of patients with early disease who have a very low risk of dying from the disease.
In a cohort of almost 11,600 patients, the overall 7-year rate of death from melanoma was 2.5%, but the risk in a subset of 25% of patients was below 1%. Conversely, the study authors were also able to identify a small subset of high‐risk patients with a greater than 20% risk for death.
and may help to begin to address the problem of overdiagnosis, they note.
“While the topic of very low-risk melanomas has been presented at national and international meetings, there have been no formal discussions to define the classification of ‘melanocytic neoplasms of low malignant potential’ at this time,” first author Megan M. Eguchi, MPH, of the department of medicine, University of California, Los Angeles, said in an interview. “Criteria would need to be established using study designs beyond those available using SEER data.”
She emphasized that currently, they do not propose any change to treatment of these lesions, just a change to the terminology. “A diagnosis of ‘MNLMP’ rather than ‘melanoma’ may potentially alleviate people’s concerns related to prognosis and begin to address the problem of overdiagnosis,” said Ms. Eguchi. The study was recently published online in Cancer.
Even though melanoma is considered to be the most common potentially lethal tumor of the skin, prognosis is often very good for those with T1 tumors, the lowest risk category. Prognostic modeling has been used to predict survival in patients with melanoma and identify prognostic variables, the authors note, with the most prominent attributes being Breslow thickness and ulceration of the primary tumor, which form the basis of the current American Joint Committee on Cancer (AJCC) staging system.
There is evidence that the increasing incidence of melanoma is partly due to overdiagnosis, meaning the diagnosis of lesions that will not lead to symptoms or death. The authors write that they were interested in identifying lesions that are currently diagnosed as melanoma but might lack the capacity for metastasis, cases that could potentially be part of the phenomenon of overdiagnosis.
Subsets with low and high risk for death
In the study, Ms. Eguchi and colleagues analyzed information from the United States Surveillance, Epidemiology, and End Results (SEER) database and identified 11,594 patients who were diagnosed in 2010 and 2011 with stage 1 melanoma that was less than or equal to 1.0 mm in thickness and had not spread to the lymph nodes. Prognostic models for risk for death from melanoma in patients with low-risk melanomas were developed, then the ability of the models to identify very‐low risk subsets of patients with melanoma‐specific survival surpassing that of T1 overall was evaluated.
The median age of the patients was 58 years, the median Breslow thickness was 0.45 mm (interquartile range, 0.30-0.65 mm), and 71% were assigned stage IA. Ulceration was present in 4% of cases, 27% were mitogenic, and 45% were Clark level II, and within this cohort, 292 (2.5%) patients died of melanoma within 7 years. In the training data set, 177 of 7,652 (2.3%) patients died of melanoma within 7 years, and numbers were similar in the testing set (115 of 3,942; 2.9%).
Overall, the investigators identified three large subsets of patients who were in the AJCC seventh edition classification for stage I (“thin”) melanoma, who had a risk for death of approximately less than 1%. This was a marked improvement from the rate of the overall sample. In the simplest model (Model 1A), patients who were younger than 70 years at diagnosis with Clark level II invasion were deemed as very low risk.
In Model 1B, the same initial classification was used, but it was further refined and limited to patients who were either age 43 years or younger or 44-69 years with Breslow thickness less than 0.40 mm. At 10 years postdiagnosis, this subset also showed a less than 1% risk for death from melanoma. The logistic regression model (Model 2) was similar, as it identified about 25% of patients with a predicted risk for death of less than 0.5%, incorporating patient age, sex, mitogenicity, Clark level, and ulceration. Model 2 was also able to further identify a small subset of patients with no deaths.
The logistic regression model was also able to identify a very small subset (0.7% and 0.8%) of patients who had a risk for death that exceeded 20%, which was markedly higher, compared with most patients with T1b tumors.
This study was supported by the National Cancer Institute. Ms. Eguchi had no disclosures to report.
A version of this article first appeared on Medscape.com.
Although melanoma is the most serious skin cancer, most patients do have high chances of survival. New research has now identified a subset of patients with early disease who have a very low risk of dying from the disease.
In a cohort of almost 11,600 patients, the overall 7-year rate of death from melanoma was 2.5%, but the risk in a subset of 25% of patients was below 1%. Conversely, the study authors were also able to identify a small subset of high‐risk patients with a greater than 20% risk for death.
and may help to begin to address the problem of overdiagnosis, they note.
“While the topic of very low-risk melanomas has been presented at national and international meetings, there have been no formal discussions to define the classification of ‘melanocytic neoplasms of low malignant potential’ at this time,” first author Megan M. Eguchi, MPH, of the department of medicine, University of California, Los Angeles, said in an interview. “Criteria would need to be established using study designs beyond those available using SEER data.”
She emphasized that currently, they do not propose any change to treatment of these lesions, just a change to the terminology. “A diagnosis of ‘MNLMP’ rather than ‘melanoma’ may potentially alleviate people’s concerns related to prognosis and begin to address the problem of overdiagnosis,” said Ms. Eguchi. The study was recently published online in Cancer.
Even though melanoma is considered to be the most common potentially lethal tumor of the skin, prognosis is often very good for those with T1 tumors, the lowest risk category. Prognostic modeling has been used to predict survival in patients with melanoma and identify prognostic variables, the authors note, with the most prominent attributes being Breslow thickness and ulceration of the primary tumor, which form the basis of the current American Joint Committee on Cancer (AJCC) staging system.
There is evidence that the increasing incidence of melanoma is partly due to overdiagnosis, meaning the diagnosis of lesions that will not lead to symptoms or death. The authors write that they were interested in identifying lesions that are currently diagnosed as melanoma but might lack the capacity for metastasis, cases that could potentially be part of the phenomenon of overdiagnosis.
Subsets with low and high risk for death
In the study, Ms. Eguchi and colleagues analyzed information from the United States Surveillance, Epidemiology, and End Results (SEER) database and identified 11,594 patients who were diagnosed in 2010 and 2011 with stage 1 melanoma that was less than or equal to 1.0 mm in thickness and had not spread to the lymph nodes. Prognostic models for risk for death from melanoma in patients with low-risk melanomas were developed, then the ability of the models to identify very‐low risk subsets of patients with melanoma‐specific survival surpassing that of T1 overall was evaluated.
The median age of the patients was 58 years, the median Breslow thickness was 0.45 mm (interquartile range, 0.30-0.65 mm), and 71% were assigned stage IA. Ulceration was present in 4% of cases, 27% were mitogenic, and 45% were Clark level II, and within this cohort, 292 (2.5%) patients died of melanoma within 7 years. In the training data set, 177 of 7,652 (2.3%) patients died of melanoma within 7 years, and numbers were similar in the testing set (115 of 3,942; 2.9%).
Overall, the investigators identified three large subsets of patients who were in the AJCC seventh edition classification for stage I (“thin”) melanoma, who had a risk for death of approximately less than 1%. This was a marked improvement from the rate of the overall sample. In the simplest model (Model 1A), patients who were younger than 70 years at diagnosis with Clark level II invasion were deemed as very low risk.
In Model 1B, the same initial classification was used, but it was further refined and limited to patients who were either age 43 years or younger or 44-69 years with Breslow thickness less than 0.40 mm. At 10 years postdiagnosis, this subset also showed a less than 1% risk for death from melanoma. The logistic regression model (Model 2) was similar, as it identified about 25% of patients with a predicted risk for death of less than 0.5%, incorporating patient age, sex, mitogenicity, Clark level, and ulceration. Model 2 was also able to further identify a small subset of patients with no deaths.
The logistic regression model was also able to identify a very small subset (0.7% and 0.8%) of patients who had a risk for death that exceeded 20%, which was markedly higher, compared with most patients with T1b tumors.
This study was supported by the National Cancer Institute. Ms. Eguchi had no disclosures to report.
A version of this article first appeared on Medscape.com.
Although melanoma is the most serious skin cancer, most patients do have high chances of survival. New research has now identified a subset of patients with early disease who have a very low risk of dying from the disease.
In a cohort of almost 11,600 patients, the overall 7-year rate of death from melanoma was 2.5%, but the risk in a subset of 25% of patients was below 1%. Conversely, the study authors were also able to identify a small subset of high‐risk patients with a greater than 20% risk for death.
and may help to begin to address the problem of overdiagnosis, they note.
“While the topic of very low-risk melanomas has been presented at national and international meetings, there have been no formal discussions to define the classification of ‘melanocytic neoplasms of low malignant potential’ at this time,” first author Megan M. Eguchi, MPH, of the department of medicine, University of California, Los Angeles, said in an interview. “Criteria would need to be established using study designs beyond those available using SEER data.”
She emphasized that currently, they do not propose any change to treatment of these lesions, just a change to the terminology. “A diagnosis of ‘MNLMP’ rather than ‘melanoma’ may potentially alleviate people’s concerns related to prognosis and begin to address the problem of overdiagnosis,” said Ms. Eguchi. The study was recently published online in Cancer.
Even though melanoma is considered to be the most common potentially lethal tumor of the skin, prognosis is often very good for those with T1 tumors, the lowest risk category. Prognostic modeling has been used to predict survival in patients with melanoma and identify prognostic variables, the authors note, with the most prominent attributes being Breslow thickness and ulceration of the primary tumor, which form the basis of the current American Joint Committee on Cancer (AJCC) staging system.
There is evidence that the increasing incidence of melanoma is partly due to overdiagnosis, meaning the diagnosis of lesions that will not lead to symptoms or death. The authors write that they were interested in identifying lesions that are currently diagnosed as melanoma but might lack the capacity for metastasis, cases that could potentially be part of the phenomenon of overdiagnosis.
Subsets with low and high risk for death
In the study, Ms. Eguchi and colleagues analyzed information from the United States Surveillance, Epidemiology, and End Results (SEER) database and identified 11,594 patients who were diagnosed in 2010 and 2011 with stage 1 melanoma that was less than or equal to 1.0 mm in thickness and had not spread to the lymph nodes. Prognostic models for risk for death from melanoma in patients with low-risk melanomas were developed, then the ability of the models to identify very‐low risk subsets of patients with melanoma‐specific survival surpassing that of T1 overall was evaluated.
The median age of the patients was 58 years, the median Breslow thickness was 0.45 mm (interquartile range, 0.30-0.65 mm), and 71% were assigned stage IA. Ulceration was present in 4% of cases, 27% were mitogenic, and 45% were Clark level II, and within this cohort, 292 (2.5%) patients died of melanoma within 7 years. In the training data set, 177 of 7,652 (2.3%) patients died of melanoma within 7 years, and numbers were similar in the testing set (115 of 3,942; 2.9%).
Overall, the investigators identified three large subsets of patients who were in the AJCC seventh edition classification for stage I (“thin”) melanoma, who had a risk for death of approximately less than 1%. This was a marked improvement from the rate of the overall sample. In the simplest model (Model 1A), patients who were younger than 70 years at diagnosis with Clark level II invasion were deemed as very low risk.
In Model 1B, the same initial classification was used, but it was further refined and limited to patients who were either age 43 years or younger or 44-69 years with Breslow thickness less than 0.40 mm. At 10 years postdiagnosis, this subset also showed a less than 1% risk for death from melanoma. The logistic regression model (Model 2) was similar, as it identified about 25% of patients with a predicted risk for death of less than 0.5%, incorporating patient age, sex, mitogenicity, Clark level, and ulceration. Model 2 was also able to further identify a small subset of patients with no deaths.
The logistic regression model was also able to identify a very small subset (0.7% and 0.8%) of patients who had a risk for death that exceeded 20%, which was markedly higher, compared with most patients with T1b tumors.
This study was supported by the National Cancer Institute. Ms. Eguchi had no disclosures to report.
A version of this article first appeared on Medscape.com.
FROM CANCER
Study eyes sunscreens marketed to individuals with skin of color
, and more than 40% contain a UV blocker that may create a white cast.
Those are among the findings from a study by Michelle Xiong, a medical student at Brown University, Providence, R.I., and Erin M. Warshaw, MD, of the department of dermatology at Park Nicollet/Health Partners Health Services, Minneapolis, which was published online in the Journal of the American Academy of Dermatology.
“There is increasing awareness of the negative effects of ultraviolet (UV) light in individuals with skin of color (SOC), especially in regards to pigmentation disorders induced and/or exacerbated by UV exposure,” the authors wrote. “As a result, there has been a surge in sunscreens marketed to this population. We aimed to characterize cost, marketing claims, and potential allergenic ingredients in sunscreens marketed to individuals with SOC.”
Between December 2021 and October 2022, the researchers used the following search terms on Google: “sunscreen” plus “skin of 36 color,” “dark skin,” “brown skin,” “LatinX skin,” and/or “Black skin.” They extracted price, marketing claims, and ingredients from manufacturers’ websites and used 90 allergens contained in the American Contact Dermatitis Society 2020 Core series to identify potential allergens. Next, they combined cross-reactors/synonyms into allergen categories based on ACDS Contact Allergen Management Plan (CAMP) cross-reactor classification. If multiple ingredients in a sunscreen were represented by a single allergen category, it was counted only once. A similar approach was utilized for marketing categories.
A total of 12 sunscreens were included in the analysis: Absolute Joi, Black Girl Sunscreen, Black Girl Sunscreen Make It Matte, Bolden SPF Brightening Moisturizer, Eleven on the Defense Unrivaled Sun Serum, Kinlo Golden Rays Sunscreen, Live Tinted Hueguard 3-in-1 Mineral Sunscreen, Mele Dew The Most Sheer Moisturizer SPF30 Broad Spectrum Sunscreen, Mele No Shade Sunscreen Oil, Specific Beauty Active Radiance Day Moi, Unsun Mineral Sunscreen, and Urban Skin Rx Complexion Protection. Their average cost was $19.30 per ounce (range, $6.33-$50.00) and common marketing claims for these products were “no white cast” (91.7%), being free of an ingredient (83.3%), and “moisturizing” (75%).
Of the 12 sunscreens, 7 (58.3%) contained a chemical sunscreen agent, 5 (41.7%) contained a physical UV blocker, and all contained at least one allergen. The average number of allergens per product was 4.7, most commonly fragrance/botanicals (83.3%), tocopherol (83.3%), sodium benzoates/derivatives (58.3%), and sorbitan sesquiolate/derivatives (58.3%).
“Average cost of sunscreens marketed to individuals with SOC was $19.30/oz, much higher than the median price of $3.32/oz reported in a separate study of 65 popular sunscreens,” the study authors wrote. “As many of the sunscreens in our study were sold by smaller businesses, higher prices may be due to higher production costs or a perceived smaller market.”
The authors expressed surprise that five sunscreens marketed to individuals with SOC contained a physical UV blocker which may create a white cast. They contacted the manufacturers of these five sunscreens and confirmed that three used micronized formulations. “While ingested/inhaled nanoparticles of titanium dioxide may cause tissue effects, most studies of topical products show excellent safety,” they wrote.
They also noted that the average of 4.7 allergens per product observed in the analysis was similar to the average of 4.9 seen in a separate study of 52 popular sunscreens. “However, that study only included 34 allergens while this study evaluated 90 allergens,” the authors wrote. “Consumers and providers should be aware sunscreens marketed to individuals with SOC may cause allergic contact dermatitis,” they commented.
“It is interesting to see how costly these products are now compared to store bought and general commercially available sunscreens several years ago,” said Lawrence J. Green, clinical professor of dermatology at George Washington University, Washington, who was asked to comment on the study. “However, to me that is not surprising as products marketed and targeted to specific populations are often priced at a premium. It wasn’t clear to me how many of these specialized online SOC sunscreens are tinted. I wish the authors had compared the cost of tinted sunscreens in general to nontinted sunscreens because tinted ones are more useful for SOC, because when rubbed in, they can readily match SOC and can also offer protection in the visible light spectrum.”
The authors reported having no financial disclosures; the study had no funding source. Dr. Green disclosed that he is a speaker, consultant, or investigator for many pharmaceutical companies.
, and more than 40% contain a UV blocker that may create a white cast.
Those are among the findings from a study by Michelle Xiong, a medical student at Brown University, Providence, R.I., and Erin M. Warshaw, MD, of the department of dermatology at Park Nicollet/Health Partners Health Services, Minneapolis, which was published online in the Journal of the American Academy of Dermatology.
“There is increasing awareness of the negative effects of ultraviolet (UV) light in individuals with skin of color (SOC), especially in regards to pigmentation disorders induced and/or exacerbated by UV exposure,” the authors wrote. “As a result, there has been a surge in sunscreens marketed to this population. We aimed to characterize cost, marketing claims, and potential allergenic ingredients in sunscreens marketed to individuals with SOC.”
Between December 2021 and October 2022, the researchers used the following search terms on Google: “sunscreen” plus “skin of 36 color,” “dark skin,” “brown skin,” “LatinX skin,” and/or “Black skin.” They extracted price, marketing claims, and ingredients from manufacturers’ websites and used 90 allergens contained in the American Contact Dermatitis Society 2020 Core series to identify potential allergens. Next, they combined cross-reactors/synonyms into allergen categories based on ACDS Contact Allergen Management Plan (CAMP) cross-reactor classification. If multiple ingredients in a sunscreen were represented by a single allergen category, it was counted only once. A similar approach was utilized for marketing categories.
A total of 12 sunscreens were included in the analysis: Absolute Joi, Black Girl Sunscreen, Black Girl Sunscreen Make It Matte, Bolden SPF Brightening Moisturizer, Eleven on the Defense Unrivaled Sun Serum, Kinlo Golden Rays Sunscreen, Live Tinted Hueguard 3-in-1 Mineral Sunscreen, Mele Dew The Most Sheer Moisturizer SPF30 Broad Spectrum Sunscreen, Mele No Shade Sunscreen Oil, Specific Beauty Active Radiance Day Moi, Unsun Mineral Sunscreen, and Urban Skin Rx Complexion Protection. Their average cost was $19.30 per ounce (range, $6.33-$50.00) and common marketing claims for these products were “no white cast” (91.7%), being free of an ingredient (83.3%), and “moisturizing” (75%).
Of the 12 sunscreens, 7 (58.3%) contained a chemical sunscreen agent, 5 (41.7%) contained a physical UV blocker, and all contained at least one allergen. The average number of allergens per product was 4.7, most commonly fragrance/botanicals (83.3%), tocopherol (83.3%), sodium benzoates/derivatives (58.3%), and sorbitan sesquiolate/derivatives (58.3%).
“Average cost of sunscreens marketed to individuals with SOC was $19.30/oz, much higher than the median price of $3.32/oz reported in a separate study of 65 popular sunscreens,” the study authors wrote. “As many of the sunscreens in our study were sold by smaller businesses, higher prices may be due to higher production costs or a perceived smaller market.”
The authors expressed surprise that five sunscreens marketed to individuals with SOC contained a physical UV blocker which may create a white cast. They contacted the manufacturers of these five sunscreens and confirmed that three used micronized formulations. “While ingested/inhaled nanoparticles of titanium dioxide may cause tissue effects, most studies of topical products show excellent safety,” they wrote.
They also noted that the average of 4.7 allergens per product observed in the analysis was similar to the average of 4.9 seen in a separate study of 52 popular sunscreens. “However, that study only included 34 allergens while this study evaluated 90 allergens,” the authors wrote. “Consumers and providers should be aware sunscreens marketed to individuals with SOC may cause allergic contact dermatitis,” they commented.
“It is interesting to see how costly these products are now compared to store bought and general commercially available sunscreens several years ago,” said Lawrence J. Green, clinical professor of dermatology at George Washington University, Washington, who was asked to comment on the study. “However, to me that is not surprising as products marketed and targeted to specific populations are often priced at a premium. It wasn’t clear to me how many of these specialized online SOC sunscreens are tinted. I wish the authors had compared the cost of tinted sunscreens in general to nontinted sunscreens because tinted ones are more useful for SOC, because when rubbed in, they can readily match SOC and can also offer protection in the visible light spectrum.”
The authors reported having no financial disclosures; the study had no funding source. Dr. Green disclosed that he is a speaker, consultant, or investigator for many pharmaceutical companies.
, and more than 40% contain a UV blocker that may create a white cast.
Those are among the findings from a study by Michelle Xiong, a medical student at Brown University, Providence, R.I., and Erin M. Warshaw, MD, of the department of dermatology at Park Nicollet/Health Partners Health Services, Minneapolis, which was published online in the Journal of the American Academy of Dermatology.
“There is increasing awareness of the negative effects of ultraviolet (UV) light in individuals with skin of color (SOC), especially in regards to pigmentation disorders induced and/or exacerbated by UV exposure,” the authors wrote. “As a result, there has been a surge in sunscreens marketed to this population. We aimed to characterize cost, marketing claims, and potential allergenic ingredients in sunscreens marketed to individuals with SOC.”
Between December 2021 and October 2022, the researchers used the following search terms on Google: “sunscreen” plus “skin of 36 color,” “dark skin,” “brown skin,” “LatinX skin,” and/or “Black skin.” They extracted price, marketing claims, and ingredients from manufacturers’ websites and used 90 allergens contained in the American Contact Dermatitis Society 2020 Core series to identify potential allergens. Next, they combined cross-reactors/synonyms into allergen categories based on ACDS Contact Allergen Management Plan (CAMP) cross-reactor classification. If multiple ingredients in a sunscreen were represented by a single allergen category, it was counted only once. A similar approach was utilized for marketing categories.
A total of 12 sunscreens were included in the analysis: Absolute Joi, Black Girl Sunscreen, Black Girl Sunscreen Make It Matte, Bolden SPF Brightening Moisturizer, Eleven on the Defense Unrivaled Sun Serum, Kinlo Golden Rays Sunscreen, Live Tinted Hueguard 3-in-1 Mineral Sunscreen, Mele Dew The Most Sheer Moisturizer SPF30 Broad Spectrum Sunscreen, Mele No Shade Sunscreen Oil, Specific Beauty Active Radiance Day Moi, Unsun Mineral Sunscreen, and Urban Skin Rx Complexion Protection. Their average cost was $19.30 per ounce (range, $6.33-$50.00) and common marketing claims for these products were “no white cast” (91.7%), being free of an ingredient (83.3%), and “moisturizing” (75%).
Of the 12 sunscreens, 7 (58.3%) contained a chemical sunscreen agent, 5 (41.7%) contained a physical UV blocker, and all contained at least one allergen. The average number of allergens per product was 4.7, most commonly fragrance/botanicals (83.3%), tocopherol (83.3%), sodium benzoates/derivatives (58.3%), and sorbitan sesquiolate/derivatives (58.3%).
“Average cost of sunscreens marketed to individuals with SOC was $19.30/oz, much higher than the median price of $3.32/oz reported in a separate study of 65 popular sunscreens,” the study authors wrote. “As many of the sunscreens in our study were sold by smaller businesses, higher prices may be due to higher production costs or a perceived smaller market.”
The authors expressed surprise that five sunscreens marketed to individuals with SOC contained a physical UV blocker which may create a white cast. They contacted the manufacturers of these five sunscreens and confirmed that three used micronized formulations. “While ingested/inhaled nanoparticles of titanium dioxide may cause tissue effects, most studies of topical products show excellent safety,” they wrote.
They also noted that the average of 4.7 allergens per product observed in the analysis was similar to the average of 4.9 seen in a separate study of 52 popular sunscreens. “However, that study only included 34 allergens while this study evaluated 90 allergens,” the authors wrote. “Consumers and providers should be aware sunscreens marketed to individuals with SOC may cause allergic contact dermatitis,” they commented.
“It is interesting to see how costly these products are now compared to store bought and general commercially available sunscreens several years ago,” said Lawrence J. Green, clinical professor of dermatology at George Washington University, Washington, who was asked to comment on the study. “However, to me that is not surprising as products marketed and targeted to specific populations are often priced at a premium. It wasn’t clear to me how many of these specialized online SOC sunscreens are tinted. I wish the authors had compared the cost of tinted sunscreens in general to nontinted sunscreens because tinted ones are more useful for SOC, because when rubbed in, they can readily match SOC and can also offer protection in the visible light spectrum.”
The authors reported having no financial disclosures; the study had no funding source. Dr. Green disclosed that he is a speaker, consultant, or investigator for many pharmaceutical companies.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Pooled safety data analysis of tralokinumab reported
The most , according to a review published in the British Journal of Dermatology.
These findings underscore the mechanistic elegance of interleukin (IL)-13 inhibition and highlight potential advantages of flexible dosing, according to the study’s lead author, Eric Simpson, MD, MCR. Overall, the pooled analysis of safety data from five phase 2 and 3 trials shows that “blockade of a single cytokine provides excellent short- and long-term safety, which is useful for a severe chronic disease,” said Dr. Simpson, professor of dermatology at Oregon Health & Science University in Portland.
Most patients with AD require years of treatment. “So for clinicians to confidently report to patients the low rates of serious adverse events (AEs) and lack of immune suppression side-effect profile is very encouraging for both the provider and patient,” Dr. Simpson said, noting there were no new signals or concerning short-term AEs.
Tralokinumab (Adbry), an IL-13 antagonist administered subcutaneously, was approved by the Food and Drug Administration for treatment of moderate to severe AD in adults in December 2021.
Minor differences vs. placebo
In the pooled analysis involving 1,605 patients treated for 16 weeks with tralokinumab and 680 who received placebo, frequency of any AE was 65.7% and 67.2%, respectively. Severe AEs occurred in 4.6% and 6.3% of patients, respectively.
The most common AE overall was AD, which occurred less often in tralokinumab-treated patients (15.4%) than those on placebo (26.2%). Other common AEs that occurred more frequently with tralokinumab included viral upper respiratory tract infections (15.7% vs. 12.2%), upper respiratory tract infections (URTI, 5.6% vs. 4.8%), conjunctivitis (5.4% vs. 1.9%), and injection-site reactions (3.5% vs. 0.3%).
AEs that occurred less often with tralokinumab than placebo included skin infections (3.7% vs. 9.2%, respectively) and infected dermatitis (1.6% vs. 6.4%).
Regarding safety areas of special interest, eye disorders classified as conjunctivitis, keratoconjunctivitis, or keratitis occurred more commonly with tralokinumab (7.9%) than placebo (3.4%). Most eye disorders were mild or moderate and resolved during the study. During maintenance treatment up to 52 weeks, AE rates mirrored those in the initial treatment period and did not increase with treatment duration.
In fact, Dr. Simpson said, the low rate of AEs that are known to accompany type 2 blockade, such as conjunctivitis, do not increase but rather appear to drop with longer-term use. The fact that skin infections were reduced vs. placebo and decreased over time suggests that long-term IL-13 blockade with tralokinumab positively impacts skin infections, a well-known comorbidity in uncontrolled AD, he added.
Raj Chovatiya, MD, PhD, who was asked to comment on the study, said, “These findings provide additional data supporting the safety and tolerability of tralokinumab and support my personal real-world experience with tralokinumab as a safe and effective biologic therapy for patients with moderate to severe AD.”
Dr. Chovatiya is assistant professor, director of the Center for Eczema and Itch, and medical director of clinical trials at Northwestern University in Chicago.
Four-week dosing
Consistent with ECZTRA 3, the rates of URTIs and conjunctivitis were lower with maintenance dosing 300 mg every 4 weeks, consideration of which is approved for responders weighing less than 220 pounds, vs. 300 mg every 2 weeks. Specifically, 6.7% of patients on every 4-week dosing schedule experienced URTIs, vs. 9.4% on the every 2-week dosing schedule and 7% of those on the every 2-week dosing schedule plus optional topical corticosteroids. Corresponding figures for conjunctivitis were 3%, 5%, and 5.6%, respectively.
“Four-week dosing is a possibility in your patients with a good clinical response at 16 weeks,” Dr. Simpson said. Advantages include improved convenience for patients, he added, and this analysis shows that dosing every 4 weeks may improve tolerability, with a lower rate of conjunctivitis.
Although it is difficult to directly compare review data to other studies, said Dr. Chovatiya, findings also suggest that tralokinumab may be associated with reduced infections and conjunctivitis compared with other advanced AD therapies. Head-to-head trials and real-world studies are needed to better understand comparative safety, he added.
Some patients will lose a degree of response with the 4-week dosing schedule, Dr. Simpson said. In ECZTRA 1 and 2, 55.9% of patients who achieved investigator global assessment (IGA) scores of 0 or 1 after 16 weeks of dosing every 2 weeks maintained this response level through week 52, vs. 42.4% of responders who switched from dosing every 2 weeks to every 4 weeks after week 16. But according to data that Dr. Simpson recently presented, 95% of patients switched to monthly dosing who relapsed and returned to dosing every 2 weeks regained their original response level within approximately 4 weeks.
In his personal practice, Dr. Simpson has prescribed tralokinumab for patients with AD for up to a year. However, he and fellow investigators have been following much larger populations for more than 2 years and are planning additional publications. “Safety data will continue to accrue” said Dr. Simpson, “but I don’t expect any surprises.”
The clinical trials were sponsored by MedImmune (phase 2b) and LEO Pharma ( ECZTRA phase 3 trials), which also sponsored the review. Dr. Simpson reports grants and personal fees from numerous pharmaceutical companies. Dr. Chovatiya has been an advisory board member, consultant, investigator, and speaker for numerous pharmaceutical companies including LEO Pharma.
A version of this article first appeared on Medscape.com.
The most , according to a review published in the British Journal of Dermatology.
These findings underscore the mechanistic elegance of interleukin (IL)-13 inhibition and highlight potential advantages of flexible dosing, according to the study’s lead author, Eric Simpson, MD, MCR. Overall, the pooled analysis of safety data from five phase 2 and 3 trials shows that “blockade of a single cytokine provides excellent short- and long-term safety, which is useful for a severe chronic disease,” said Dr. Simpson, professor of dermatology at Oregon Health & Science University in Portland.
Most patients with AD require years of treatment. “So for clinicians to confidently report to patients the low rates of serious adverse events (AEs) and lack of immune suppression side-effect profile is very encouraging for both the provider and patient,” Dr. Simpson said, noting there were no new signals or concerning short-term AEs.
Tralokinumab (Adbry), an IL-13 antagonist administered subcutaneously, was approved by the Food and Drug Administration for treatment of moderate to severe AD in adults in December 2021.
Minor differences vs. placebo
In the pooled analysis involving 1,605 patients treated for 16 weeks with tralokinumab and 680 who received placebo, frequency of any AE was 65.7% and 67.2%, respectively. Severe AEs occurred in 4.6% and 6.3% of patients, respectively.
The most common AE overall was AD, which occurred less often in tralokinumab-treated patients (15.4%) than those on placebo (26.2%). Other common AEs that occurred more frequently with tralokinumab included viral upper respiratory tract infections (15.7% vs. 12.2%), upper respiratory tract infections (URTI, 5.6% vs. 4.8%), conjunctivitis (5.4% vs. 1.9%), and injection-site reactions (3.5% vs. 0.3%).
AEs that occurred less often with tralokinumab than placebo included skin infections (3.7% vs. 9.2%, respectively) and infected dermatitis (1.6% vs. 6.4%).
Regarding safety areas of special interest, eye disorders classified as conjunctivitis, keratoconjunctivitis, or keratitis occurred more commonly with tralokinumab (7.9%) than placebo (3.4%). Most eye disorders were mild or moderate and resolved during the study. During maintenance treatment up to 52 weeks, AE rates mirrored those in the initial treatment period and did not increase with treatment duration.
In fact, Dr. Simpson said, the low rate of AEs that are known to accompany type 2 blockade, such as conjunctivitis, do not increase but rather appear to drop with longer-term use. The fact that skin infections were reduced vs. placebo and decreased over time suggests that long-term IL-13 blockade with tralokinumab positively impacts skin infections, a well-known comorbidity in uncontrolled AD, he added.
Raj Chovatiya, MD, PhD, who was asked to comment on the study, said, “These findings provide additional data supporting the safety and tolerability of tralokinumab and support my personal real-world experience with tralokinumab as a safe and effective biologic therapy for patients with moderate to severe AD.”
Dr. Chovatiya is assistant professor, director of the Center for Eczema and Itch, and medical director of clinical trials at Northwestern University in Chicago.
Four-week dosing
Consistent with ECZTRA 3, the rates of URTIs and conjunctivitis were lower with maintenance dosing 300 mg every 4 weeks, consideration of which is approved for responders weighing less than 220 pounds, vs. 300 mg every 2 weeks. Specifically, 6.7% of patients on every 4-week dosing schedule experienced URTIs, vs. 9.4% on the every 2-week dosing schedule and 7% of those on the every 2-week dosing schedule plus optional topical corticosteroids. Corresponding figures for conjunctivitis were 3%, 5%, and 5.6%, respectively.
“Four-week dosing is a possibility in your patients with a good clinical response at 16 weeks,” Dr. Simpson said. Advantages include improved convenience for patients, he added, and this analysis shows that dosing every 4 weeks may improve tolerability, with a lower rate of conjunctivitis.
Although it is difficult to directly compare review data to other studies, said Dr. Chovatiya, findings also suggest that tralokinumab may be associated with reduced infections and conjunctivitis compared with other advanced AD therapies. Head-to-head trials and real-world studies are needed to better understand comparative safety, he added.
Some patients will lose a degree of response with the 4-week dosing schedule, Dr. Simpson said. In ECZTRA 1 and 2, 55.9% of patients who achieved investigator global assessment (IGA) scores of 0 or 1 after 16 weeks of dosing every 2 weeks maintained this response level through week 52, vs. 42.4% of responders who switched from dosing every 2 weeks to every 4 weeks after week 16. But according to data that Dr. Simpson recently presented, 95% of patients switched to monthly dosing who relapsed and returned to dosing every 2 weeks regained their original response level within approximately 4 weeks.
In his personal practice, Dr. Simpson has prescribed tralokinumab for patients with AD for up to a year. However, he and fellow investigators have been following much larger populations for more than 2 years and are planning additional publications. “Safety data will continue to accrue” said Dr. Simpson, “but I don’t expect any surprises.”
The clinical trials were sponsored by MedImmune (phase 2b) and LEO Pharma ( ECZTRA phase 3 trials), which also sponsored the review. Dr. Simpson reports grants and personal fees from numerous pharmaceutical companies. Dr. Chovatiya has been an advisory board member, consultant, investigator, and speaker for numerous pharmaceutical companies including LEO Pharma.
A version of this article first appeared on Medscape.com.
The most , according to a review published in the British Journal of Dermatology.
These findings underscore the mechanistic elegance of interleukin (IL)-13 inhibition and highlight potential advantages of flexible dosing, according to the study’s lead author, Eric Simpson, MD, MCR. Overall, the pooled analysis of safety data from five phase 2 and 3 trials shows that “blockade of a single cytokine provides excellent short- and long-term safety, which is useful for a severe chronic disease,” said Dr. Simpson, professor of dermatology at Oregon Health & Science University in Portland.
Most patients with AD require years of treatment. “So for clinicians to confidently report to patients the low rates of serious adverse events (AEs) and lack of immune suppression side-effect profile is very encouraging for both the provider and patient,” Dr. Simpson said, noting there were no new signals or concerning short-term AEs.
Tralokinumab (Adbry), an IL-13 antagonist administered subcutaneously, was approved by the Food and Drug Administration for treatment of moderate to severe AD in adults in December 2021.
Minor differences vs. placebo
In the pooled analysis involving 1,605 patients treated for 16 weeks with tralokinumab and 680 who received placebo, frequency of any AE was 65.7% and 67.2%, respectively. Severe AEs occurred in 4.6% and 6.3% of patients, respectively.
The most common AE overall was AD, which occurred less often in tralokinumab-treated patients (15.4%) than those on placebo (26.2%). Other common AEs that occurred more frequently with tralokinumab included viral upper respiratory tract infections (15.7% vs. 12.2%), upper respiratory tract infections (URTI, 5.6% vs. 4.8%), conjunctivitis (5.4% vs. 1.9%), and injection-site reactions (3.5% vs. 0.3%).
AEs that occurred less often with tralokinumab than placebo included skin infections (3.7% vs. 9.2%, respectively) and infected dermatitis (1.6% vs. 6.4%).
Regarding safety areas of special interest, eye disorders classified as conjunctivitis, keratoconjunctivitis, or keratitis occurred more commonly with tralokinumab (7.9%) than placebo (3.4%). Most eye disorders were mild or moderate and resolved during the study. During maintenance treatment up to 52 weeks, AE rates mirrored those in the initial treatment period and did not increase with treatment duration.
In fact, Dr. Simpson said, the low rate of AEs that are known to accompany type 2 blockade, such as conjunctivitis, do not increase but rather appear to drop with longer-term use. The fact that skin infections were reduced vs. placebo and decreased over time suggests that long-term IL-13 blockade with tralokinumab positively impacts skin infections, a well-known comorbidity in uncontrolled AD, he added.
Raj Chovatiya, MD, PhD, who was asked to comment on the study, said, “These findings provide additional data supporting the safety and tolerability of tralokinumab and support my personal real-world experience with tralokinumab as a safe and effective biologic therapy for patients with moderate to severe AD.”
Dr. Chovatiya is assistant professor, director of the Center for Eczema and Itch, and medical director of clinical trials at Northwestern University in Chicago.
Four-week dosing
Consistent with ECZTRA 3, the rates of URTIs and conjunctivitis were lower with maintenance dosing 300 mg every 4 weeks, consideration of which is approved for responders weighing less than 220 pounds, vs. 300 mg every 2 weeks. Specifically, 6.7% of patients on every 4-week dosing schedule experienced URTIs, vs. 9.4% on the every 2-week dosing schedule and 7% of those on the every 2-week dosing schedule plus optional topical corticosteroids. Corresponding figures for conjunctivitis were 3%, 5%, and 5.6%, respectively.
“Four-week dosing is a possibility in your patients with a good clinical response at 16 weeks,” Dr. Simpson said. Advantages include improved convenience for patients, he added, and this analysis shows that dosing every 4 weeks may improve tolerability, with a lower rate of conjunctivitis.
Although it is difficult to directly compare review data to other studies, said Dr. Chovatiya, findings also suggest that tralokinumab may be associated with reduced infections and conjunctivitis compared with other advanced AD therapies. Head-to-head trials and real-world studies are needed to better understand comparative safety, he added.
Some patients will lose a degree of response with the 4-week dosing schedule, Dr. Simpson said. In ECZTRA 1 and 2, 55.9% of patients who achieved investigator global assessment (IGA) scores of 0 or 1 after 16 weeks of dosing every 2 weeks maintained this response level through week 52, vs. 42.4% of responders who switched from dosing every 2 weeks to every 4 weeks after week 16. But according to data that Dr. Simpson recently presented, 95% of patients switched to monthly dosing who relapsed and returned to dosing every 2 weeks regained their original response level within approximately 4 weeks.
In his personal practice, Dr. Simpson has prescribed tralokinumab for patients with AD for up to a year. However, he and fellow investigators have been following much larger populations for more than 2 years and are planning additional publications. “Safety data will continue to accrue” said Dr. Simpson, “but I don’t expect any surprises.”
The clinical trials were sponsored by MedImmune (phase 2b) and LEO Pharma ( ECZTRA phase 3 trials), which also sponsored the review. Dr. Simpson reports grants and personal fees from numerous pharmaceutical companies. Dr. Chovatiya has been an advisory board member, consultant, investigator, and speaker for numerous pharmaceutical companies including LEO Pharma.
A version of this article first appeared on Medscape.com.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Current alopecia areata options include old and new therapies
LAS VEGAS – in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
“Some patients don’t have alopecia, but they have been managed for it,” he said. “Whenever there is an ounce of doubt, take a biopsy,” he advised.
Assessing disease severity in patients with alopecia areata (AA) is especially important as new therapies become available, said Dr. King, associate professor of dermatology at Yale University, New Haven, Conn. The Severity of Alopecia Tool (SALT) Score has been available since 2004, and remains a useful tool to estimate percent hair loss. The SALT Score divides the scalp into four sections: 18% each for the right and left sides, 40% for the top of the head, and 24% for the back of the head, said Dr. King. However, the SALT Score can be enhanced or modified based on a holistic approach to disease severity that categorizes alopecia as mild (scalp hair loss of 20% or less), moderate (scalp hair loss of 21 to 49%), or severe (scalp hair loss of 50% or more).
For example, if a patient’s hair loss based on SALT Score is mild or moderate, increase the severity by 1 level (from mild to moderate, or moderate to severe) if any of the following conditions apply: Noticeable eyebrow or eyelash involvement, inadequate treatment response after 6 months, diffuse positive hair pull test consistent with rapid progression of AA, or a negative impact on psychosocial functioning because of AA, he said.
Treatment advances
Understanding of the pathogenesis of AA has been slow to evolve, Dr. King noted. “We haven’t been able to shake this concept that people are causing the disease by being depressed,” as noted in the literature from the 1950s.
In 2014, breakthrough research changed the game by identifying the roles of interferon gamma and interleukin 15, Dr. King said. Since then, more research has been conducted on Janus kinase (JAK) inhibitors for AA. Dr. King was a coinvestigator on a 2014 case report in which a patient with psoriasis and alopecia universalis experienced regrowth of most of his body hair after 8 months of daily oral tofacitinib, a JAK inhibitor.
However, despite the dramatic results in some patients, “tofacitinib doesn’t always work,” said Dr. King. In his experience, patients for whom tofacitinib didn’t work were those with complete or nearly complete scalp hair loss for more than 10 years.
Approval of baricitinib
Dr. King’s recent work supported the approval in June 2022 of oral baricitinib, a JAK inhibitor, for AA. He reviewed data from his late-breaker abstract presented at the annual meeting of the American Academy of Dermatology in March 2022, where he reported that almost 40% of adults with AA treated with 4 mg of baricitinib daily had significant hair regrowth over 52 weeks.
Two other oral JAK inhibitors in the pipeline for AA are deuruxolitinib and ritlecitinib, which significantly increased the proportion of patients achieving SALT scores of 20 or less, compared with patients on placebo in early clinical trials. Data on both were presented at the annual meeting of the European Academy of Dermatology and Venereology.
So far, topical JAK inhibitors have not shown success in hair regrowth for AA patients, said Dr. King. Phase 2 studies of both ruxolitinib 1.5% cream and delgocitinib ointment were ineffective for AA.
Emerging role for oral minoxidil
Oral minoxidil has had a recent resurgence as an adjunct therapy to the new JAK inhibitors. A study published in 1987 found that, with oral minoxidil monotherapy, a cosmetic response was seen in 18% of patients with AA, Dr. King said.
In a study published in the Journal of the American Academy of Dermatology, Dr. King and colleagues noted that dose escalation is sometimes needed for effective treatment of AA with tofacitinib. They examined the effect of adding oral minoxidil to tofacitinib in patients with severe AA as a way to increase efficacy without increasing tofacitinib dosage. They reviewed data from 12 patients ages 18-51 years who were prescribed 5 mg of tofacitinib twice daily, plus 2.5 mg oral minoxidil daily for women and 2.5 mg of minoxidil twice daily for men; women received a lower dose to minimize the side effect of hypertrichosis.
After 6 months, 67% (eight patients) achieved at least 75% hair regrowth; of those eight patients, seven (58% of the total) had hair regrowth on a twice-daily dose of 5 mg tofacitinib with no need for dose escalation, Dr. King said.
More research is needed, but oral minoxidil may be a useful adjunct treatment for some patients with AA, he added.
During a question and answer session, Dr. King was asked to elaborate on the mechanism of minoxidil in combination with JAK inhibitors. “The truth is that I just don’t know” why the combination works for some patients. However, the majority of patients who succeed with this combination regrow hair by 4 months. “There is something special about that combination.”
Dr. King disclosed serving as a consultant or adviser for AbbVie, AltruBio, Almirall, AnaptysBio, Arena Pharmaceuticals, Bioniz, Bristol Myers Squibb, Concert Pharmaceuticals, Horizon, Incyte, Leo Pharma, Eli Lilly, Otsuka, Pfizer, Regeneron, Sanofi Genzyme, Twi Biotechnology, Viela Bio, and Visterra; serving as a speaker or as a member of the speakers bureau for Incyte, Pfizer, Regeneron, Sanofi Genzyme; and receiving research funding from Concert Pharmaceuticals, Eli Lilly, and Pfizer.
MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
“Some patients don’t have alopecia, but they have been managed for it,” he said. “Whenever there is an ounce of doubt, take a biopsy,” he advised.
Assessing disease severity in patients with alopecia areata (AA) is especially important as new therapies become available, said Dr. King, associate professor of dermatology at Yale University, New Haven, Conn. The Severity of Alopecia Tool (SALT) Score has been available since 2004, and remains a useful tool to estimate percent hair loss. The SALT Score divides the scalp into four sections: 18% each for the right and left sides, 40% for the top of the head, and 24% for the back of the head, said Dr. King. However, the SALT Score can be enhanced or modified based on a holistic approach to disease severity that categorizes alopecia as mild (scalp hair loss of 20% or less), moderate (scalp hair loss of 21 to 49%), or severe (scalp hair loss of 50% or more).
For example, if a patient’s hair loss based on SALT Score is mild or moderate, increase the severity by 1 level (from mild to moderate, or moderate to severe) if any of the following conditions apply: Noticeable eyebrow or eyelash involvement, inadequate treatment response after 6 months, diffuse positive hair pull test consistent with rapid progression of AA, or a negative impact on psychosocial functioning because of AA, he said.
Treatment advances
Understanding of the pathogenesis of AA has been slow to evolve, Dr. King noted. “We haven’t been able to shake this concept that people are causing the disease by being depressed,” as noted in the literature from the 1950s.
In 2014, breakthrough research changed the game by identifying the roles of interferon gamma and interleukin 15, Dr. King said. Since then, more research has been conducted on Janus kinase (JAK) inhibitors for AA. Dr. King was a coinvestigator on a 2014 case report in which a patient with psoriasis and alopecia universalis experienced regrowth of most of his body hair after 8 months of daily oral tofacitinib, a JAK inhibitor.
However, despite the dramatic results in some patients, “tofacitinib doesn’t always work,” said Dr. King. In his experience, patients for whom tofacitinib didn’t work were those with complete or nearly complete scalp hair loss for more than 10 years.
Approval of baricitinib
Dr. King’s recent work supported the approval in June 2022 of oral baricitinib, a JAK inhibitor, for AA. He reviewed data from his late-breaker abstract presented at the annual meeting of the American Academy of Dermatology in March 2022, where he reported that almost 40% of adults with AA treated with 4 mg of baricitinib daily had significant hair regrowth over 52 weeks.
Two other oral JAK inhibitors in the pipeline for AA are deuruxolitinib and ritlecitinib, which significantly increased the proportion of patients achieving SALT scores of 20 or less, compared with patients on placebo in early clinical trials. Data on both were presented at the annual meeting of the European Academy of Dermatology and Venereology.
So far, topical JAK inhibitors have not shown success in hair regrowth for AA patients, said Dr. King. Phase 2 studies of both ruxolitinib 1.5% cream and delgocitinib ointment were ineffective for AA.
Emerging role for oral minoxidil
Oral minoxidil has had a recent resurgence as an adjunct therapy to the new JAK inhibitors. A study published in 1987 found that, with oral minoxidil monotherapy, a cosmetic response was seen in 18% of patients with AA, Dr. King said.
In a study published in the Journal of the American Academy of Dermatology, Dr. King and colleagues noted that dose escalation is sometimes needed for effective treatment of AA with tofacitinib. They examined the effect of adding oral minoxidil to tofacitinib in patients with severe AA as a way to increase efficacy without increasing tofacitinib dosage. They reviewed data from 12 patients ages 18-51 years who were prescribed 5 mg of tofacitinib twice daily, plus 2.5 mg oral minoxidil daily for women and 2.5 mg of minoxidil twice daily for men; women received a lower dose to minimize the side effect of hypertrichosis.
After 6 months, 67% (eight patients) achieved at least 75% hair regrowth; of those eight patients, seven (58% of the total) had hair regrowth on a twice-daily dose of 5 mg tofacitinib with no need for dose escalation, Dr. King said.
More research is needed, but oral minoxidil may be a useful adjunct treatment for some patients with AA, he added.
During a question and answer session, Dr. King was asked to elaborate on the mechanism of minoxidil in combination with JAK inhibitors. “The truth is that I just don’t know” why the combination works for some patients. However, the majority of patients who succeed with this combination regrow hair by 4 months. “There is something special about that combination.”
Dr. King disclosed serving as a consultant or adviser for AbbVie, AltruBio, Almirall, AnaptysBio, Arena Pharmaceuticals, Bioniz, Bristol Myers Squibb, Concert Pharmaceuticals, Horizon, Incyte, Leo Pharma, Eli Lilly, Otsuka, Pfizer, Regeneron, Sanofi Genzyme, Twi Biotechnology, Viela Bio, and Visterra; serving as a speaker or as a member of the speakers bureau for Incyte, Pfizer, Regeneron, Sanofi Genzyme; and receiving research funding from Concert Pharmaceuticals, Eli Lilly, and Pfizer.
MedscapeLive and this news organization are owned by the same parent company.
LAS VEGAS – in a presentation at MedscapeLive’s annual Las Vegas Dermatology Seminar.
“Some patients don’t have alopecia, but they have been managed for it,” he said. “Whenever there is an ounce of doubt, take a biopsy,” he advised.
Assessing disease severity in patients with alopecia areata (AA) is especially important as new therapies become available, said Dr. King, associate professor of dermatology at Yale University, New Haven, Conn. The Severity of Alopecia Tool (SALT) Score has been available since 2004, and remains a useful tool to estimate percent hair loss. The SALT Score divides the scalp into four sections: 18% each for the right and left sides, 40% for the top of the head, and 24% for the back of the head, said Dr. King. However, the SALT Score can be enhanced or modified based on a holistic approach to disease severity that categorizes alopecia as mild (scalp hair loss of 20% or less), moderate (scalp hair loss of 21 to 49%), or severe (scalp hair loss of 50% or more).
For example, if a patient’s hair loss based on SALT Score is mild or moderate, increase the severity by 1 level (from mild to moderate, or moderate to severe) if any of the following conditions apply: Noticeable eyebrow or eyelash involvement, inadequate treatment response after 6 months, diffuse positive hair pull test consistent with rapid progression of AA, or a negative impact on psychosocial functioning because of AA, he said.
Treatment advances
Understanding of the pathogenesis of AA has been slow to evolve, Dr. King noted. “We haven’t been able to shake this concept that people are causing the disease by being depressed,” as noted in the literature from the 1950s.
In 2014, breakthrough research changed the game by identifying the roles of interferon gamma and interleukin 15, Dr. King said. Since then, more research has been conducted on Janus kinase (JAK) inhibitors for AA. Dr. King was a coinvestigator on a 2014 case report in which a patient with psoriasis and alopecia universalis experienced regrowth of most of his body hair after 8 months of daily oral tofacitinib, a JAK inhibitor.
However, despite the dramatic results in some patients, “tofacitinib doesn’t always work,” said Dr. King. In his experience, patients for whom tofacitinib didn’t work were those with complete or nearly complete scalp hair loss for more than 10 years.
Approval of baricitinib
Dr. King’s recent work supported the approval in June 2022 of oral baricitinib, a JAK inhibitor, for AA. He reviewed data from his late-breaker abstract presented at the annual meeting of the American Academy of Dermatology in March 2022, where he reported that almost 40% of adults with AA treated with 4 mg of baricitinib daily had significant hair regrowth over 52 weeks.
Two other oral JAK inhibitors in the pipeline for AA are deuruxolitinib and ritlecitinib, which significantly increased the proportion of patients achieving SALT scores of 20 or less, compared with patients on placebo in early clinical trials. Data on both were presented at the annual meeting of the European Academy of Dermatology and Venereology.
So far, topical JAK inhibitors have not shown success in hair regrowth for AA patients, said Dr. King. Phase 2 studies of both ruxolitinib 1.5% cream and delgocitinib ointment were ineffective for AA.
Emerging role for oral minoxidil
Oral minoxidil has had a recent resurgence as an adjunct therapy to the new JAK inhibitors. A study published in 1987 found that, with oral minoxidil monotherapy, a cosmetic response was seen in 18% of patients with AA, Dr. King said.
In a study published in the Journal of the American Academy of Dermatology, Dr. King and colleagues noted that dose escalation is sometimes needed for effective treatment of AA with tofacitinib. They examined the effect of adding oral minoxidil to tofacitinib in patients with severe AA as a way to increase efficacy without increasing tofacitinib dosage. They reviewed data from 12 patients ages 18-51 years who were prescribed 5 mg of tofacitinib twice daily, plus 2.5 mg oral minoxidil daily for women and 2.5 mg of minoxidil twice daily for men; women received a lower dose to minimize the side effect of hypertrichosis.
After 6 months, 67% (eight patients) achieved at least 75% hair regrowth; of those eight patients, seven (58% of the total) had hair regrowth on a twice-daily dose of 5 mg tofacitinib with no need for dose escalation, Dr. King said.
More research is needed, but oral minoxidil may be a useful adjunct treatment for some patients with AA, he added.
During a question and answer session, Dr. King was asked to elaborate on the mechanism of minoxidil in combination with JAK inhibitors. “The truth is that I just don’t know” why the combination works for some patients. However, the majority of patients who succeed with this combination regrow hair by 4 months. “There is something special about that combination.”
Dr. King disclosed serving as a consultant or adviser for AbbVie, AltruBio, Almirall, AnaptysBio, Arena Pharmaceuticals, Bioniz, Bristol Myers Squibb, Concert Pharmaceuticals, Horizon, Incyte, Leo Pharma, Eli Lilly, Otsuka, Pfizer, Regeneron, Sanofi Genzyme, Twi Biotechnology, Viela Bio, and Visterra; serving as a speaker or as a member of the speakers bureau for Incyte, Pfizer, Regeneron, Sanofi Genzyme; and receiving research funding from Concert Pharmaceuticals, Eli Lilly, and Pfizer.
MedscapeLive and this news organization are owned by the same parent company.
AT INNOVATIONS IN DERMATOLOGY