User login
Study eyes characteristics of pediatric patients with hidradenitis suppurativa
INDIANAPOLIS – in a study presented at the annual meeting of the Society for Pediatric Dermatology.
In addition, 44% presented with scarring, which suggests that HS may be underdiagnosed in this patient population. Those are the key findings from the study, a single-center retrospective chart review presented by Stephanie Sanchez during a poster session at the meeting.

“There is limited research on HS within the pediatric population,” said Ms. Sanchez, a fourth-year medical student at Boston University. “It’s not very well defined or characterized.” The “unusually high number of pediatric patients with HS” at Boston Medical Center provided “a unique opportunity to study this topic.”
Working with her mentor, Lisa Shen, MD, associate medical director of pediatric dermatology at Boston University, Ms. Sanchez and colleagues retrospectively reviewed the medical records of 303 patients aged 4-18 years who were diagnosed with HS at Boston Medical Center from 2012 to 2021. Boston Medical Center is the largest safety net hospital in New England. All data points and outcome measures were collected within 6 months of the patient’s HS diagnosis date.
Of the 303 patients with HS, 84% were female and 16% were male. Complete information about race was available in 286 patients. Of these, 65% were Black/African American, 11% were White, and the rest were from other racial groups. The mean age at symptom onset was 13 years, while the mean age at diagnosis was 15 years, and the mean delay to diagnosis was 2 years. A family history of HS was reported in 36% of patients.
The most common clinical features in these HS patients were pain/tenderness (90%), pustules/papules (65%), discharge/drainage (62%), and deep-seated nodules (51%). Scarring was present in 44% of patients at the time of diagnosis. The three most common sites of involvement were the axillary area (79%), the pubic area (36%), and the inguinal folds/inner thighs (34%).
Obesity was the most common comorbidity at the time of diagnosis, with 64% of patients affected. The next most common comorbidities were acne vulgaris (36%), acanthosis nigricans (25%), depression (18%), being overweight (17%), polycystic ovary syndrome (16%) and anxiety (13%). None had type 1 diabetes or metabolic syndrome.
Referring to the large population of underserved minority patients at Boston Medical Center, Dr. Shen noted, “we have to make sure not to underestimate the prevalence of obesity in this population as they get older. We need to start from a younger age to incorporate multidisciplinary care such as weight management, nutrition, and working with our pediatric surgery colleagues in trying to tackle [HS] because there is data to suggest that the earlier we intervene, the better outcomes they have. That makes sense.”
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the findings, said that the study “highlights the impressive and concerning gap and delays in diagnosis, not too dissimilar to what the literature shows in adult HS patients, which unfortunately has tremendous ramifications, both physically and emotionally/psychosocially.”
While this single-center study identified potential risk factors, such as obesity and self-identifying as Black, he said, “it is important to note that this condition does not discriminate and therefore it is important not to miss the cases that don’t follow the textbook nor stigmatize this condition as one that only impacts certain demographics.”
The researchers reported having no financial disclosures. Dr. Friedman, who was not involved with the study, reported that he serves as a consultant and/or advisor to numerous pharmaceutical companies. He is a speaker for companies including, Regeneron, Sanofi, AbbVie, Janssen, Incyte, and Brickell Biotech, and has received grants from Pfizer, the Dermatology Foundation, Almirall, Incyte, Galderma, and Janssen.
INDIANAPOLIS – in a study presented at the annual meeting of the Society for Pediatric Dermatology.
In addition, 44% presented with scarring, which suggests that HS may be underdiagnosed in this patient population. Those are the key findings from the study, a single-center retrospective chart review presented by Stephanie Sanchez during a poster session at the meeting.

“There is limited research on HS within the pediatric population,” said Ms. Sanchez, a fourth-year medical student at Boston University. “It’s not very well defined or characterized.” The “unusually high number of pediatric patients with HS” at Boston Medical Center provided “a unique opportunity to study this topic.”
Working with her mentor, Lisa Shen, MD, associate medical director of pediatric dermatology at Boston University, Ms. Sanchez and colleagues retrospectively reviewed the medical records of 303 patients aged 4-18 years who were diagnosed with HS at Boston Medical Center from 2012 to 2021. Boston Medical Center is the largest safety net hospital in New England. All data points and outcome measures were collected within 6 months of the patient’s HS diagnosis date.
Of the 303 patients with HS, 84% were female and 16% were male. Complete information about race was available in 286 patients. Of these, 65% were Black/African American, 11% were White, and the rest were from other racial groups. The mean age at symptom onset was 13 years, while the mean age at diagnosis was 15 years, and the mean delay to diagnosis was 2 years. A family history of HS was reported in 36% of patients.
The most common clinical features in these HS patients were pain/tenderness (90%), pustules/papules (65%), discharge/drainage (62%), and deep-seated nodules (51%). Scarring was present in 44% of patients at the time of diagnosis. The three most common sites of involvement were the axillary area (79%), the pubic area (36%), and the inguinal folds/inner thighs (34%).
Obesity was the most common comorbidity at the time of diagnosis, with 64% of patients affected. The next most common comorbidities were acne vulgaris (36%), acanthosis nigricans (25%), depression (18%), being overweight (17%), polycystic ovary syndrome (16%) and anxiety (13%). None had type 1 diabetes or metabolic syndrome.
Referring to the large population of underserved minority patients at Boston Medical Center, Dr. Shen noted, “we have to make sure not to underestimate the prevalence of obesity in this population as they get older. We need to start from a younger age to incorporate multidisciplinary care such as weight management, nutrition, and working with our pediatric surgery colleagues in trying to tackle [HS] because there is data to suggest that the earlier we intervene, the better outcomes they have. That makes sense.”
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the findings, said that the study “highlights the impressive and concerning gap and delays in diagnosis, not too dissimilar to what the literature shows in adult HS patients, which unfortunately has tremendous ramifications, both physically and emotionally/psychosocially.”
While this single-center study identified potential risk factors, such as obesity and self-identifying as Black, he said, “it is important to note that this condition does not discriminate and therefore it is important not to miss the cases that don’t follow the textbook nor stigmatize this condition as one that only impacts certain demographics.”
The researchers reported having no financial disclosures. Dr. Friedman, who was not involved with the study, reported that he serves as a consultant and/or advisor to numerous pharmaceutical companies. He is a speaker for companies including, Regeneron, Sanofi, AbbVie, Janssen, Incyte, and Brickell Biotech, and has received grants from Pfizer, the Dermatology Foundation, Almirall, Incyte, Galderma, and Janssen.
INDIANAPOLIS – in a study presented at the annual meeting of the Society for Pediatric Dermatology.
In addition, 44% presented with scarring, which suggests that HS may be underdiagnosed in this patient population. Those are the key findings from the study, a single-center retrospective chart review presented by Stephanie Sanchez during a poster session at the meeting.

“There is limited research on HS within the pediatric population,” said Ms. Sanchez, a fourth-year medical student at Boston University. “It’s not very well defined or characterized.” The “unusually high number of pediatric patients with HS” at Boston Medical Center provided “a unique opportunity to study this topic.”
Working with her mentor, Lisa Shen, MD, associate medical director of pediatric dermatology at Boston University, Ms. Sanchez and colleagues retrospectively reviewed the medical records of 303 patients aged 4-18 years who were diagnosed with HS at Boston Medical Center from 2012 to 2021. Boston Medical Center is the largest safety net hospital in New England. All data points and outcome measures were collected within 6 months of the patient’s HS diagnosis date.
Of the 303 patients with HS, 84% were female and 16% were male. Complete information about race was available in 286 patients. Of these, 65% were Black/African American, 11% were White, and the rest were from other racial groups. The mean age at symptom onset was 13 years, while the mean age at diagnosis was 15 years, and the mean delay to diagnosis was 2 years. A family history of HS was reported in 36% of patients.
The most common clinical features in these HS patients were pain/tenderness (90%), pustules/papules (65%), discharge/drainage (62%), and deep-seated nodules (51%). Scarring was present in 44% of patients at the time of diagnosis. The three most common sites of involvement were the axillary area (79%), the pubic area (36%), and the inguinal folds/inner thighs (34%).
Obesity was the most common comorbidity at the time of diagnosis, with 64% of patients affected. The next most common comorbidities were acne vulgaris (36%), acanthosis nigricans (25%), depression (18%), being overweight (17%), polycystic ovary syndrome (16%) and anxiety (13%). None had type 1 diabetes or metabolic syndrome.
Referring to the large population of underserved minority patients at Boston Medical Center, Dr. Shen noted, “we have to make sure not to underestimate the prevalence of obesity in this population as they get older. We need to start from a younger age to incorporate multidisciplinary care such as weight management, nutrition, and working with our pediatric surgery colleagues in trying to tackle [HS] because there is data to suggest that the earlier we intervene, the better outcomes they have. That makes sense.”
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the findings, said that the study “highlights the impressive and concerning gap and delays in diagnosis, not too dissimilar to what the literature shows in adult HS patients, which unfortunately has tremendous ramifications, both physically and emotionally/psychosocially.”
While this single-center study identified potential risk factors, such as obesity and self-identifying as Black, he said, “it is important to note that this condition does not discriminate and therefore it is important not to miss the cases that don’t follow the textbook nor stigmatize this condition as one that only impacts certain demographics.”
The researchers reported having no financial disclosures. Dr. Friedman, who was not involved with the study, reported that he serves as a consultant and/or advisor to numerous pharmaceutical companies. He is a speaker for companies including, Regeneron, Sanofi, AbbVie, Janssen, Incyte, and Brickell Biotech, and has received grants from Pfizer, the Dermatology Foundation, Almirall, Incyte, Galderma, and Janssen.
AT SPD 2022
Pembrolizumab for melanoma bittersweet, doctor says
CHICAGO – Pembrolizumab has shown promise as adjuvant therapy for stage IIB and IIC melanoma, shows the first interim analysis of the phase 3 KEYNOTE-716 study recently published in The Lancet.
The findings meet an unmet need as the recurrence risk in stage IIB and IIC melanoma is “underrecognized,” said author Georgina Long, MD, comedical director of the Melanoma Institute Australia, University of Sydney.
In fact, their risk of recurrence is similar to patients with stage IIIB disease, wrote David Killock, PhD, in a related commentary published in Nature Reviews.
The adjuvant treatment resulted in an 89% recurrence-free survival in patients who received pembrolizumab, compared with 83% of patients in the placebo group (hazard ratio, 0.65; P = .0066). These findings were used as the basis for Food and Drug Administration approval of pembrolizumab (Keytruda, Merck) for this patient population in December 2021.
Despite the positive findings, Dr. Killock called for more research on distant metastasis-free survival, overall survival, and quality of life data to “establish the true clinical benefit of adjuvant pembrolizumab.”
At the annual meeting of the American Society of Clinical Oncology, Dr. Long presented the third interim analysis which showed pembrolizumab reduced recurrence and distant metastases at 24 months, although the clinical benefit was relatively small at an approximately 8% improvement in recurrence-free survival and about a 6% improvement in distant metastasis-free survival. About 83% in the pembrolizumab group had treatment-related toxicities versus 64% in the placebo group. There were no deaths caused by treatment. About 90% of pembrolizumab-related endocrinopathies led to long-term hormone replacement.
In a discussion that followed the presentation at ASCO, Charlotte Eielson Ariyan, MD, PhD, said the results are bittersweet. Higher-risk stage IIC patients have a risk of recurrence of about 40%. “It’s high, but the absolute risk reduction is about 8%. This is a very personalized discussion with the patient and the physician in understanding their risk of toxicity is about 17% and higher than their absolute risk reduction with the treatment. For me, this is a bitter pill to swallow because you’re treating people longer and you’re not sure if you’re really helping them. Until we can further define who the highest-risk patients are, I think it’s hard to give it to everyone,” said Dr. Ariyan, who is a surgeon with Memorial Sloan Kettering Cancer Center, New York.
In addition to weighing short-term benefits and toxicity, there are longer-term concerns. Toxicity experienced from PD-1 inhibitors in the adjuvant setting could impact future treatment decisions. “We’re very lucky here in melanoma to know that systemic therapies are effective and we can cure people who recur. I would argue this is why we probably will never really see a difference in the survival benefit in this group because people who cross over will probably do well,” Dr. Ariyan said.
During the Q&A session, Vernon Sondek, MD, Moffitt Cancer Center, Tampa, encouraged physician colleagues to have an open mind about treatments. “Beware of dogma. We thought that adjuvant immunotherapy works much better in patients with ulcerated primary tumors. That’s a dogma in some parts of the world. Yet the T4a patients in KEYNOTE-716 dramatically outperformed the ulcerated T3b and T4b [patients]. We still don’t know what we don’t know.”
The study details
KEYNOTE-716 included 976 patients 12 years or older with newly diagnosed completely resected stage IIB or IIC melanoma with a negative sentinel lymph node. Patients were randomized to placebo or 200 mg pembrolizumab every 3 weeks, or 2 mg/kg in pediatric patients, over 17 cycles. Almost 40% of patients were age 65 or older. T3b and T4b were the most common melanoma subcategories at 41% and 35%, respectively.
The planned third interim analysis occurred after the occurrence of 146 distant metastases. After a median follow-up of 27.4 months, distant metastasis-free survival favored the pembrolizumab group (HR, 0.64; P = .0029). At 24 months, the pembrolizumab group had a higher distant metastasis-free survival at 88.1% versus 82.2% and a lower recurrence rate at 81.2% versus 72.8% (HR, 0.64; 95% confidence interval, 0.50-0.84).
At 24 months, only the T4a patients had a statistically significant reduction in distant metastases at 58% (HR, 0.42; 95% CI, 0.19-0.96), although there were numerical reductions in T3a (HR, 0.71; 95% CI, 0.41-1.22) and T4b (HR, 0.70; 95% CI, 0.44-1.33) patients. Of patients experiencing a distant metastasis, 73% of the placebo group had a first distant metastasis to the lung compared with 49% of the pembrolizumab group.
Dr. Long has held consulting or advisory roles for Merck Sharpe & Dohme, which funded this study.
CHICAGO – Pembrolizumab has shown promise as adjuvant therapy for stage IIB and IIC melanoma, shows the first interim analysis of the phase 3 KEYNOTE-716 study recently published in The Lancet.
The findings meet an unmet need as the recurrence risk in stage IIB and IIC melanoma is “underrecognized,” said author Georgina Long, MD, comedical director of the Melanoma Institute Australia, University of Sydney.
In fact, their risk of recurrence is similar to patients with stage IIIB disease, wrote David Killock, PhD, in a related commentary published in Nature Reviews.
The adjuvant treatment resulted in an 89% recurrence-free survival in patients who received pembrolizumab, compared with 83% of patients in the placebo group (hazard ratio, 0.65; P = .0066). These findings were used as the basis for Food and Drug Administration approval of pembrolizumab (Keytruda, Merck) for this patient population in December 2021.
Despite the positive findings, Dr. Killock called for more research on distant metastasis-free survival, overall survival, and quality of life data to “establish the true clinical benefit of adjuvant pembrolizumab.”
At the annual meeting of the American Society of Clinical Oncology, Dr. Long presented the third interim analysis which showed pembrolizumab reduced recurrence and distant metastases at 24 months, although the clinical benefit was relatively small at an approximately 8% improvement in recurrence-free survival and about a 6% improvement in distant metastasis-free survival. About 83% in the pembrolizumab group had treatment-related toxicities versus 64% in the placebo group. There were no deaths caused by treatment. About 90% of pembrolizumab-related endocrinopathies led to long-term hormone replacement.
In a discussion that followed the presentation at ASCO, Charlotte Eielson Ariyan, MD, PhD, said the results are bittersweet. Higher-risk stage IIC patients have a risk of recurrence of about 40%. “It’s high, but the absolute risk reduction is about 8%. This is a very personalized discussion with the patient and the physician in understanding their risk of toxicity is about 17% and higher than their absolute risk reduction with the treatment. For me, this is a bitter pill to swallow because you’re treating people longer and you’re not sure if you’re really helping them. Until we can further define who the highest-risk patients are, I think it’s hard to give it to everyone,” said Dr. Ariyan, who is a surgeon with Memorial Sloan Kettering Cancer Center, New York.
In addition to weighing short-term benefits and toxicity, there are longer-term concerns. Toxicity experienced from PD-1 inhibitors in the adjuvant setting could impact future treatment decisions. “We’re very lucky here in melanoma to know that systemic therapies are effective and we can cure people who recur. I would argue this is why we probably will never really see a difference in the survival benefit in this group because people who cross over will probably do well,” Dr. Ariyan said.
During the Q&A session, Vernon Sondek, MD, Moffitt Cancer Center, Tampa, encouraged physician colleagues to have an open mind about treatments. “Beware of dogma. We thought that adjuvant immunotherapy works much better in patients with ulcerated primary tumors. That’s a dogma in some parts of the world. Yet the T4a patients in KEYNOTE-716 dramatically outperformed the ulcerated T3b and T4b [patients]. We still don’t know what we don’t know.”
The study details
KEYNOTE-716 included 976 patients 12 years or older with newly diagnosed completely resected stage IIB or IIC melanoma with a negative sentinel lymph node. Patients were randomized to placebo or 200 mg pembrolizumab every 3 weeks, or 2 mg/kg in pediatric patients, over 17 cycles. Almost 40% of patients were age 65 or older. T3b and T4b were the most common melanoma subcategories at 41% and 35%, respectively.
The planned third interim analysis occurred after the occurrence of 146 distant metastases. After a median follow-up of 27.4 months, distant metastasis-free survival favored the pembrolizumab group (HR, 0.64; P = .0029). At 24 months, the pembrolizumab group had a higher distant metastasis-free survival at 88.1% versus 82.2% and a lower recurrence rate at 81.2% versus 72.8% (HR, 0.64; 95% confidence interval, 0.50-0.84).
At 24 months, only the T4a patients had a statistically significant reduction in distant metastases at 58% (HR, 0.42; 95% CI, 0.19-0.96), although there were numerical reductions in T3a (HR, 0.71; 95% CI, 0.41-1.22) and T4b (HR, 0.70; 95% CI, 0.44-1.33) patients. Of patients experiencing a distant metastasis, 73% of the placebo group had a first distant metastasis to the lung compared with 49% of the pembrolizumab group.
Dr. Long has held consulting or advisory roles for Merck Sharpe & Dohme, which funded this study.
CHICAGO – Pembrolizumab has shown promise as adjuvant therapy for stage IIB and IIC melanoma, shows the first interim analysis of the phase 3 KEYNOTE-716 study recently published in The Lancet.
The findings meet an unmet need as the recurrence risk in stage IIB and IIC melanoma is “underrecognized,” said author Georgina Long, MD, comedical director of the Melanoma Institute Australia, University of Sydney.
In fact, their risk of recurrence is similar to patients with stage IIIB disease, wrote David Killock, PhD, in a related commentary published in Nature Reviews.
The adjuvant treatment resulted in an 89% recurrence-free survival in patients who received pembrolizumab, compared with 83% of patients in the placebo group (hazard ratio, 0.65; P = .0066). These findings were used as the basis for Food and Drug Administration approval of pembrolizumab (Keytruda, Merck) for this patient population in December 2021.
Despite the positive findings, Dr. Killock called for more research on distant metastasis-free survival, overall survival, and quality of life data to “establish the true clinical benefit of adjuvant pembrolizumab.”
At the annual meeting of the American Society of Clinical Oncology, Dr. Long presented the third interim analysis which showed pembrolizumab reduced recurrence and distant metastases at 24 months, although the clinical benefit was relatively small at an approximately 8% improvement in recurrence-free survival and about a 6% improvement in distant metastasis-free survival. About 83% in the pembrolizumab group had treatment-related toxicities versus 64% in the placebo group. There were no deaths caused by treatment. About 90% of pembrolizumab-related endocrinopathies led to long-term hormone replacement.
In a discussion that followed the presentation at ASCO, Charlotte Eielson Ariyan, MD, PhD, said the results are bittersweet. Higher-risk stage IIC patients have a risk of recurrence of about 40%. “It’s high, but the absolute risk reduction is about 8%. This is a very personalized discussion with the patient and the physician in understanding their risk of toxicity is about 17% and higher than their absolute risk reduction with the treatment. For me, this is a bitter pill to swallow because you’re treating people longer and you’re not sure if you’re really helping them. Until we can further define who the highest-risk patients are, I think it’s hard to give it to everyone,” said Dr. Ariyan, who is a surgeon with Memorial Sloan Kettering Cancer Center, New York.
In addition to weighing short-term benefits and toxicity, there are longer-term concerns. Toxicity experienced from PD-1 inhibitors in the adjuvant setting could impact future treatment decisions. “We’re very lucky here in melanoma to know that systemic therapies are effective and we can cure people who recur. I would argue this is why we probably will never really see a difference in the survival benefit in this group because people who cross over will probably do well,” Dr. Ariyan said.
During the Q&A session, Vernon Sondek, MD, Moffitt Cancer Center, Tampa, encouraged physician colleagues to have an open mind about treatments. “Beware of dogma. We thought that adjuvant immunotherapy works much better in patients with ulcerated primary tumors. That’s a dogma in some parts of the world. Yet the T4a patients in KEYNOTE-716 dramatically outperformed the ulcerated T3b and T4b [patients]. We still don’t know what we don’t know.”
The study details
KEYNOTE-716 included 976 patients 12 years or older with newly diagnosed completely resected stage IIB or IIC melanoma with a negative sentinel lymph node. Patients were randomized to placebo or 200 mg pembrolizumab every 3 weeks, or 2 mg/kg in pediatric patients, over 17 cycles. Almost 40% of patients were age 65 or older. T3b and T4b were the most common melanoma subcategories at 41% and 35%, respectively.
The planned third interim analysis occurred after the occurrence of 146 distant metastases. After a median follow-up of 27.4 months, distant metastasis-free survival favored the pembrolizumab group (HR, 0.64; P = .0029). At 24 months, the pembrolizumab group had a higher distant metastasis-free survival at 88.1% versus 82.2% and a lower recurrence rate at 81.2% versus 72.8% (HR, 0.64; 95% confidence interval, 0.50-0.84).
At 24 months, only the T4a patients had a statistically significant reduction in distant metastases at 58% (HR, 0.42; 95% CI, 0.19-0.96), although there were numerical reductions in T3a (HR, 0.71; 95% CI, 0.41-1.22) and T4b (HR, 0.70; 95% CI, 0.44-1.33) patients. Of patients experiencing a distant metastasis, 73% of the placebo group had a first distant metastasis to the lung compared with 49% of the pembrolizumab group.
Dr. Long has held consulting or advisory roles for Merck Sharpe & Dohme, which funded this study.
AT ASCO 2022
What influences a trainee’s decision to choose pediatric dermatology as a career?
INDIANAPOLIS – Three during and after fellowship.
Those are key findings from a survey of current and prior pediatric dermatology fellows, which sought to investigate what factors influence their career decisions.
According to the study’s principal investigator, Lucia Z. Diaz, MD, pediatric dermatology suffers from workforce shortages and geographic maldistribution as a subspecialty in the United States. She also noted that, from 2016 to 2021, 100% of pediatric dermatology applicants matched, yet about 15 of every 31 positions remained unfilled during each of those years. This suggests that there may be a lack of trainee mentorship secondary to a lack of available pediatric dermatologists.
“Somewhere along the way, we lose trainees to general dermatology, or they may go through a pediatric dermatology fellowship but not actually see children upon completion of their training,” Dr. Diaz, chief of pediatric dermatology at the University of Texas at Austin, said in an interview at the annual meeting of the Society for Pediatric Dermatology, where the study was presented during a poster session. “We wanted to find out factors influencing this.”
For the study, Dr. Diaz, Courtney N. Haller, MD, a first-year dermatology resident at the University of Texas at Austin, and their colleagues emailed a 37-item survey to 59 current and prior pediatric dermatology fellows who trained in the United States in the past 4 years (classes of 2019-2022). Current fellows were asked to share their future plans, and past fellows were asked to share details about their current practice situation including practice type (such as academics, private practice, and a mix of adult and pediatrics), and the researchers used descriptive statistics and chi-square analyses to evaluate qualitative data.
In all, 41 survey participants gave complete responses, and 3 gave partial responses. Of these, 8 were current fellows, 36 were past fellows, and 38 were female. The researchers found that 67% of survey respondents first became interested in pediatric dermatology in medical school, while the decision to pursue a fellowship occurred then (33%) or during their third year of dermatology residency (33%). Early exposure to pediatric dermatology, from medical school through dermatology PGY-2, was significantly associated with an early decision to pursue a pediatric dermatology career (P = .004).
In addition, respondents at institutions with two or more pediatric dermatology faculty were significantly more likely to cite home institution mentorship as an influencing factor in their career decision (P = .035).
“I thought that the interest in pediatric dermatology would peak early on during dermatology residency, but it primarily happens during medical school,” said Dr. Diaz, who is also associate director of the dermatology residency program at the medical school. “Mentorship and early exposure to pediatric dermatology during medical school are really important.”
The top three factors that discouraged respondents from pursuing a pediatric dermatology fellowship included a lack of salary benefit with additional training (83%), additional time required to complete training (73%), and geographic relocation (20%). After fellowship, 51% of respondents said they plan to or currently work in academic settings, while 88% said they plan to work full time or currently were working full time.
Interestingly, fellows with additional pediatric training such as an internship or residency were not more likely to see a greater percentage of pediatric patients in practice than those without this training (P = .14). The top 3 reasons for not seeing pediatric patients 100% of the clinical time were interest in seeing adult patients (67%), financial factors (56%), and interest in performing more procedures (56%).
In other findings, the top three factors in deciding practice location were proximity to extended family (63%), practice type (59%), and income (51%).
Adelaide A. Hebert, MD, who was asked to comment on the study, said that the lack of salary benefit from additional training is a sticking point for many fellows. “The market trends of supply and demand do not work in pediatric dermatology,” said Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. “You would think that, because there are fewer of us, we should be paid more, but it does not work that way.”
She characterized the overall study findings as “a real testament to what the challenges are” in recruiting trainees to pediatric dermatology. “The influence of mentors resonates in this assessment, but influences that are somewhat beyond our control also play a role, such as lack of salary benefit from additional training, interest in seeing adult patients, and financial factors.”
Neither the researchers nor Dr. Hebert reported having relevant financial disclosures.
INDIANAPOLIS – Three during and after fellowship.
Those are key findings from a survey of current and prior pediatric dermatology fellows, which sought to investigate what factors influence their career decisions.
According to the study’s principal investigator, Lucia Z. Diaz, MD, pediatric dermatology suffers from workforce shortages and geographic maldistribution as a subspecialty in the United States. She also noted that, from 2016 to 2021, 100% of pediatric dermatology applicants matched, yet about 15 of every 31 positions remained unfilled during each of those years. This suggests that there may be a lack of trainee mentorship secondary to a lack of available pediatric dermatologists.
“Somewhere along the way, we lose trainees to general dermatology, or they may go through a pediatric dermatology fellowship but not actually see children upon completion of their training,” Dr. Diaz, chief of pediatric dermatology at the University of Texas at Austin, said in an interview at the annual meeting of the Society for Pediatric Dermatology, where the study was presented during a poster session. “We wanted to find out factors influencing this.”
For the study, Dr. Diaz, Courtney N. Haller, MD, a first-year dermatology resident at the University of Texas at Austin, and their colleagues emailed a 37-item survey to 59 current and prior pediatric dermatology fellows who trained in the United States in the past 4 years (classes of 2019-2022). Current fellows were asked to share their future plans, and past fellows were asked to share details about their current practice situation including practice type (such as academics, private practice, and a mix of adult and pediatrics), and the researchers used descriptive statistics and chi-square analyses to evaluate qualitative data.
In all, 41 survey participants gave complete responses, and 3 gave partial responses. Of these, 8 were current fellows, 36 were past fellows, and 38 were female. The researchers found that 67% of survey respondents first became interested in pediatric dermatology in medical school, while the decision to pursue a fellowship occurred then (33%) or during their third year of dermatology residency (33%). Early exposure to pediatric dermatology, from medical school through dermatology PGY-2, was significantly associated with an early decision to pursue a pediatric dermatology career (P = .004).
In addition, respondents at institutions with two or more pediatric dermatology faculty were significantly more likely to cite home institution mentorship as an influencing factor in their career decision (P = .035).
“I thought that the interest in pediatric dermatology would peak early on during dermatology residency, but it primarily happens during medical school,” said Dr. Diaz, who is also associate director of the dermatology residency program at the medical school. “Mentorship and early exposure to pediatric dermatology during medical school are really important.”
The top three factors that discouraged respondents from pursuing a pediatric dermatology fellowship included a lack of salary benefit with additional training (83%), additional time required to complete training (73%), and geographic relocation (20%). After fellowship, 51% of respondents said they plan to or currently work in academic settings, while 88% said they plan to work full time or currently were working full time.
Interestingly, fellows with additional pediatric training such as an internship or residency were not more likely to see a greater percentage of pediatric patients in practice than those without this training (P = .14). The top 3 reasons for not seeing pediatric patients 100% of the clinical time were interest in seeing adult patients (67%), financial factors (56%), and interest in performing more procedures (56%).
In other findings, the top three factors in deciding practice location were proximity to extended family (63%), practice type (59%), and income (51%).
Adelaide A. Hebert, MD, who was asked to comment on the study, said that the lack of salary benefit from additional training is a sticking point for many fellows. “The market trends of supply and demand do not work in pediatric dermatology,” said Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. “You would think that, because there are fewer of us, we should be paid more, but it does not work that way.”
She characterized the overall study findings as “a real testament to what the challenges are” in recruiting trainees to pediatric dermatology. “The influence of mentors resonates in this assessment, but influences that are somewhat beyond our control also play a role, such as lack of salary benefit from additional training, interest in seeing adult patients, and financial factors.”
Neither the researchers nor Dr. Hebert reported having relevant financial disclosures.
INDIANAPOLIS – Three during and after fellowship.
Those are key findings from a survey of current and prior pediatric dermatology fellows, which sought to investigate what factors influence their career decisions.
According to the study’s principal investigator, Lucia Z. Diaz, MD, pediatric dermatology suffers from workforce shortages and geographic maldistribution as a subspecialty in the United States. She also noted that, from 2016 to 2021, 100% of pediatric dermatology applicants matched, yet about 15 of every 31 positions remained unfilled during each of those years. This suggests that there may be a lack of trainee mentorship secondary to a lack of available pediatric dermatologists.
“Somewhere along the way, we lose trainees to general dermatology, or they may go through a pediatric dermatology fellowship but not actually see children upon completion of their training,” Dr. Diaz, chief of pediatric dermatology at the University of Texas at Austin, said in an interview at the annual meeting of the Society for Pediatric Dermatology, where the study was presented during a poster session. “We wanted to find out factors influencing this.”
For the study, Dr. Diaz, Courtney N. Haller, MD, a first-year dermatology resident at the University of Texas at Austin, and their colleagues emailed a 37-item survey to 59 current and prior pediatric dermatology fellows who trained in the United States in the past 4 years (classes of 2019-2022). Current fellows were asked to share their future plans, and past fellows were asked to share details about their current practice situation including practice type (such as academics, private practice, and a mix of adult and pediatrics), and the researchers used descriptive statistics and chi-square analyses to evaluate qualitative data.
In all, 41 survey participants gave complete responses, and 3 gave partial responses. Of these, 8 were current fellows, 36 were past fellows, and 38 were female. The researchers found that 67% of survey respondents first became interested in pediatric dermatology in medical school, while the decision to pursue a fellowship occurred then (33%) or during their third year of dermatology residency (33%). Early exposure to pediatric dermatology, from medical school through dermatology PGY-2, was significantly associated with an early decision to pursue a pediatric dermatology career (P = .004).
In addition, respondents at institutions with two or more pediatric dermatology faculty were significantly more likely to cite home institution mentorship as an influencing factor in their career decision (P = .035).
“I thought that the interest in pediatric dermatology would peak early on during dermatology residency, but it primarily happens during medical school,” said Dr. Diaz, who is also associate director of the dermatology residency program at the medical school. “Mentorship and early exposure to pediatric dermatology during medical school are really important.”
The top three factors that discouraged respondents from pursuing a pediatric dermatology fellowship included a lack of salary benefit with additional training (83%), additional time required to complete training (73%), and geographic relocation (20%). After fellowship, 51% of respondents said they plan to or currently work in academic settings, while 88% said they plan to work full time or currently were working full time.
Interestingly, fellows with additional pediatric training such as an internship or residency were not more likely to see a greater percentage of pediatric patients in practice than those without this training (P = .14). The top 3 reasons for not seeing pediatric patients 100% of the clinical time were interest in seeing adult patients (67%), financial factors (56%), and interest in performing more procedures (56%).
In other findings, the top three factors in deciding practice location were proximity to extended family (63%), practice type (59%), and income (51%).
Adelaide A. Hebert, MD, who was asked to comment on the study, said that the lack of salary benefit from additional training is a sticking point for many fellows. “The market trends of supply and demand do not work in pediatric dermatology,” said Dr. Hebert, professor of dermatology and pediatrics, and chief of pediatric dermatology at the University of Texas, Houston. “You would think that, because there are fewer of us, we should be paid more, but it does not work that way.”
She characterized the overall study findings as “a real testament to what the challenges are” in recruiting trainees to pediatric dermatology. “The influence of mentors resonates in this assessment, but influences that are somewhat beyond our control also play a role, such as lack of salary benefit from additional training, interest in seeing adult patients, and financial factors.”
Neither the researchers nor Dr. Hebert reported having relevant financial disclosures.
AT SPD 2022
Chest pain and difficulty swallowing
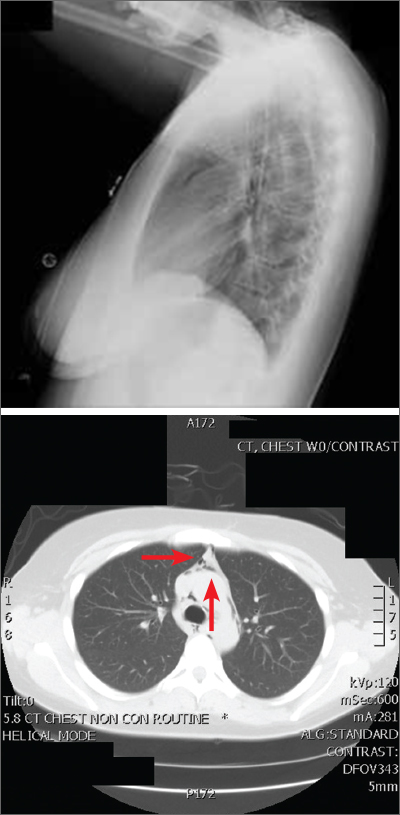
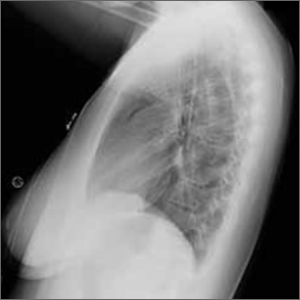
The posteroanterior (PA) and lateral view CXRs, as well as the CT scan, revealed the presence of retrosternal air and confirmed the patient’s diagnosis of pneumomediastinum.
Pneumomediastinum—the presence of free air in the mediastinum—can develop spontaneously (as was the case with our patient) or in response to trauma. Common causes include respiratory diseases such as asthma, and trauma to the esophagus secondary to mechanical ventilation, endoscopy, and excessive vomiting.1 Other possible causes include respiratory infections, foreign body aspiration, recent dental extraction, diabetic ketoacidosis, esophageal perforation, barotrauma (due to activities such as flying or scuba diving), and use of illicit drugs.1
Patients with pneumomediastinum often complain of retrosternal, pleuritic pain that radiates to their back, shoulders, and arms. They may also have difficulty swallowing (globus pharyngeus), a nasal voice, and/or dyspnea. Physical findings can include subcutaneous emphysema in the neck and supraclavicular fossa as manifested by the Hamman sign (a precordial “crunching” sound heard during systole), a fever, and distended neck veins.1
Diagnosis is made by CXR and/or chest CT. On a CXR, retrosternal air is best seen in the lateral projection. Small amounts of air can appear as linear lucencies outlining mediastinal contours. This air can be seen under the skin, surrounding the pericardium, around the pulmonary and/or aortic vasculature, and/or between the parietal pleura and diaphragm.2 A pleural effusion—particularly on the patient’s left side—should raise concern for esophageal perforation.
Pneumomediastinum is generally a self-limiting condition. Thus, patients who don’t have severe symptoms, such as respiratory distress or signs of inflammation, should be observed for 2 days, managed with rest and pain control, and discharged home. If severe symptoms or inflammatory signs are present, a Gastrografin swallow study is recommended to rule out esophageal perforation.3 If the result of this test is abnormal, a follow-up study with barium is recommended.3
The patient underwent both swallow studies, and they were negative. This patient received subsequent serial CXRs that showed improvement in the pneumomediastinum. Once the patient’s pain was well controlled with oral nonsteroidal anti-inflammatory drugs, she was discharged (after a 3-day hospitalization) with close follow-up. One week later, her pain had almost entirely resolved.
This case was adapted from: Gawrys B, Shaha D. Pleuritic chest pain and globus pharyngeus. J Fam Pract. 2015;64:305-307.
1. Park DE, Vallieres E. Pneumomediastinum and mediastinitis. In: Mason R, Broaddus V, Murray J, et al, eds. Murray and Nadel’s Textbook of Respiratory Medicine. 4th ed. Elsevier Health Sciences; 2005:2039-2068.
2. Zylak CM, Standen JR, Barnes GR, et al. Pneumomediastinum revisited. Radiographics. 2000;20:1043-1057. doi: 10.1148/radiographics.20.4.g00jl13104
3. Takada K, Matsumoto S, Hiramatsu T, et al. Management of spontaneous pneumomediastinum based on clinical experience of 25 cases. Respir Med. 2008;102:1329-1334. doi: 10.1016/j.rmed.2008.03.023

The posteroanterior (PA) and lateral view CXRs, as well as the CT scan, revealed the presence of retrosternal air and confirmed the patient’s diagnosis of pneumomediastinum.
Pneumomediastinum—the presence of free air in the mediastinum—can develop spontaneously (as was the case with our patient) or in response to trauma. Common causes include respiratory diseases such as asthma, and trauma to the esophagus secondary to mechanical ventilation, endoscopy, and excessive vomiting.1 Other possible causes include respiratory infections, foreign body aspiration, recent dental extraction, diabetic ketoacidosis, esophageal perforation, barotrauma (due to activities such as flying or scuba diving), and use of illicit drugs.1
Patients with pneumomediastinum often complain of retrosternal, pleuritic pain that radiates to their back, shoulders, and arms. They may also have difficulty swallowing (globus pharyngeus), a nasal voice, and/or dyspnea. Physical findings can include subcutaneous emphysema in the neck and supraclavicular fossa as manifested by the Hamman sign (a precordial “crunching” sound heard during systole), a fever, and distended neck veins.1
Diagnosis is made by CXR and/or chest CT. On a CXR, retrosternal air is best seen in the lateral projection. Small amounts of air can appear as linear lucencies outlining mediastinal contours. This air can be seen under the skin, surrounding the pericardium, around the pulmonary and/or aortic vasculature, and/or between the parietal pleura and diaphragm.2 A pleural effusion—particularly on the patient’s left side—should raise concern for esophageal perforation.
Pneumomediastinum is generally a self-limiting condition. Thus, patients who don’t have severe symptoms, such as respiratory distress or signs of inflammation, should be observed for 2 days, managed with rest and pain control, and discharged home. If severe symptoms or inflammatory signs are present, a Gastrografin swallow study is recommended to rule out esophageal perforation.3 If the result of this test is abnormal, a follow-up study with barium is recommended.3
The patient underwent both swallow studies, and they were negative. This patient received subsequent serial CXRs that showed improvement in the pneumomediastinum. Once the patient’s pain was well controlled with oral nonsteroidal anti-inflammatory drugs, she was discharged (after a 3-day hospitalization) with close follow-up. One week later, her pain had almost entirely resolved.
This case was adapted from: Gawrys B, Shaha D. Pleuritic chest pain and globus pharyngeus. J Fam Pract. 2015;64:305-307.

The posteroanterior (PA) and lateral view CXRs, as well as the CT scan, revealed the presence of retrosternal air and confirmed the patient’s diagnosis of pneumomediastinum.
Pneumomediastinum—the presence of free air in the mediastinum—can develop spontaneously (as was the case with our patient) or in response to trauma. Common causes include respiratory diseases such as asthma, and trauma to the esophagus secondary to mechanical ventilation, endoscopy, and excessive vomiting.1 Other possible causes include respiratory infections, foreign body aspiration, recent dental extraction, diabetic ketoacidosis, esophageal perforation, barotrauma (due to activities such as flying or scuba diving), and use of illicit drugs.1
Patients with pneumomediastinum often complain of retrosternal, pleuritic pain that radiates to their back, shoulders, and arms. They may also have difficulty swallowing (globus pharyngeus), a nasal voice, and/or dyspnea. Physical findings can include subcutaneous emphysema in the neck and supraclavicular fossa as manifested by the Hamman sign (a precordial “crunching” sound heard during systole), a fever, and distended neck veins.1
Diagnosis is made by CXR and/or chest CT. On a CXR, retrosternal air is best seen in the lateral projection. Small amounts of air can appear as linear lucencies outlining mediastinal contours. This air can be seen under the skin, surrounding the pericardium, around the pulmonary and/or aortic vasculature, and/or between the parietal pleura and diaphragm.2 A pleural effusion—particularly on the patient’s left side—should raise concern for esophageal perforation.
Pneumomediastinum is generally a self-limiting condition. Thus, patients who don’t have severe symptoms, such as respiratory distress or signs of inflammation, should be observed for 2 days, managed with rest and pain control, and discharged home. If severe symptoms or inflammatory signs are present, a Gastrografin swallow study is recommended to rule out esophageal perforation.3 If the result of this test is abnormal, a follow-up study with barium is recommended.3
The patient underwent both swallow studies, and they were negative. This patient received subsequent serial CXRs that showed improvement in the pneumomediastinum. Once the patient’s pain was well controlled with oral nonsteroidal anti-inflammatory drugs, she was discharged (after a 3-day hospitalization) with close follow-up. One week later, her pain had almost entirely resolved.
This case was adapted from: Gawrys B, Shaha D. Pleuritic chest pain and globus pharyngeus. J Fam Pract. 2015;64:305-307.
1. Park DE, Vallieres E. Pneumomediastinum and mediastinitis. In: Mason R, Broaddus V, Murray J, et al, eds. Murray and Nadel’s Textbook of Respiratory Medicine. 4th ed. Elsevier Health Sciences; 2005:2039-2068.
2. Zylak CM, Standen JR, Barnes GR, et al. Pneumomediastinum revisited. Radiographics. 2000;20:1043-1057. doi: 10.1148/radiographics.20.4.g00jl13104
3. Takada K, Matsumoto S, Hiramatsu T, et al. Management of spontaneous pneumomediastinum based on clinical experience of 25 cases. Respir Med. 2008;102:1329-1334. doi: 10.1016/j.rmed.2008.03.023
1. Park DE, Vallieres E. Pneumomediastinum and mediastinitis. In: Mason R, Broaddus V, Murray J, et al, eds. Murray and Nadel’s Textbook of Respiratory Medicine. 4th ed. Elsevier Health Sciences; 2005:2039-2068.
2. Zylak CM, Standen JR, Barnes GR, et al. Pneumomediastinum revisited. Radiographics. 2000;20:1043-1057. doi: 10.1148/radiographics.20.4.g00jl13104
3. Takada K, Matsumoto S, Hiramatsu T, et al. Management of spontaneous pneumomediastinum based on clinical experience of 25 cases. Respir Med. 2008;102:1329-1334. doi: 10.1016/j.rmed.2008.03.023
Topical gel for epidermolysis bullosa shows ongoing benefit
GLASGOW, Scotland – the phase 3 safety and efficacy study of the treatment.
Over 200 patients from the trial, including 105 who began treatment with a control gel, continued taking oleogel-S10 after 90 days. The current interim analysis at 12 months indicates there was a 55% reduction in the proportion of the body affected, compared with baseline.
Moreover, reductions in skin activity scores seen in the double-blind phase of the trial were maintained during the open-label extension. About 6% of patients experienced adverse events that led to withdrawal from the study.
The results show that oleogel-S10 was associated with “accelerated wound healing,” said study presenter Tracey Cunningham, MD, chief medical officer, Amryt Pharmaceuticals DAC, Dublin, which is developing the topical agent. “There were no new safety signals with this longer exposure to oleogel-S10, and patients had sustained improvement in wound burden,” she added.
The research was presented at the British Association of Dermatologists (BAD) 2022 Annual Meeting on July 6.
In April, European Medicines Agency recommended approval of oleogel-S10 for the treatment of partial-thickness skin wounds associated with dystrophic and junctional EB for patients aged 6 months and older.
However, just a month earlier, the U.S. Food and Drug Administration declined to approve the topical agent for use in EB, even after it extended its review by 3 months to include additional analyses of data previously submitted by the company.
In the post-presentation discussion, Dr. Cunningham said that the FDA had “not been satisfied at this point with the information that we have given them,” adding, “We don’t agree with the decision, and we will be appealing.”
Raman K. Madan, MD, a dermatologist at Northwell Health, Huntington, New York, who was not involved in the study, said that the reductions in wound healing seen in the study are “meaningful” and that the numbers represent a “big breakthrough.”
He told this news organization that there are “very few products on the market” for EB and that having an option for patients “would be amazing.”
“The big issue here would be cost and coverage for patients,” he said. If approved, “hopefully” it will be affordable, he added.
Dr. Madan noted that from his perspective, the majority of the reactions to the topical gel were “mild,” and there are “a lot of confounding factors” underlying the number of serious adverse events. “These patients with epidermolysis are prone to some of these issues regardless of treatment,” he said.
During her presentation, Dr. Cunningham noted that EB is a rare, debilitating condition that is characterized by varying degrees of skin fragility, blisters, and impaired wound healing that in turn lead to serious complications that affect quality of life.
While wound management is a “fundamental priority” for patients living with EB, she said, there is a “high, unmet” clinical need.
To those ends, EASE was the largest randomized controlled phase 3 efficacy and safety study in EB. In the study, 252 patients were allocated to receive oleogel-S10 or control gel plus standard-of-care nonadhesive wound dressing.
The double-blind phase of the trial met its primary endpoint: A higher proportion of patients who were given oleogel-S10 achieved first complete closure of the EB target wound by day 45, compared with patients who were given control gel, at 41.3% versus 28.9%. This equated to a relative risk of wound closure by day 45 of 1.44, or an odds ratio of 1.84 (P = .013).
However, as reported at the time by this news organization, the difference in time to wound healing by day 90 between the two patient groups was not statistically significant (P = .302), with 50.5% of oleogel-S10 patients achieving wound closure, versus 43.9% of those in the control group.
Dr. Cunningham discussed the open-label extension, which involved 205 patients from the double-blind phase (mean age, of 16.3 years) treated with oleogel-S10 or control gel plus standard-of-care nonadhesive wound dressing for 24 months.
In presenting the results of the first 12 months of the open-label extension, she said that oleogel-S10 led to “consistent” reductions in the body surface area percentage (BSAP) affected by EB. The overall reduction from baseline was 55% after receiving treatment for 15 months.
Between day 90 and month 12 of the open-label extension, the absolute BSAP was reduced from 7.4% to 5.4% for patients who had received oleogel-S10 from the start of the study. For those who started in the control group and then switched to the oleogel-S10 arm during the open-label extension, the reduction was from 8.3% to 6.4%.
Dr. Cunningham pointed out that a 1% reduction in BSAP equates approximately to the palmar surface of the hand.
Scores on the Epidermolysis Bullosa Disease Activity and Scarring Index (EBDASI) Skin activity subscale indicated that the reductions achieved in the double-blind phase of the trial were maintained.
Among patients who received oleogel-S10 from the start of the trial, EBDASI Skin scores were reduced from 19.6 at baseline to 13.5 at 12 months’ follow-up in the open-label extension. The reduction was from 19.6 to 13.5 for those who began the trial taking control gel.
Dr. Cunningham showed that adverse events of any grade were seen in 72.0% of patients who began taking oleogel-S10 at the start of the trial and in 69.5% of those who began the trial taking control gel.
Serious adverse events were recorded in 23.0% and 20.0% of patients, respectively, while 6.0% of those who initially received oleogel-S10 and 6.7% of those initially assigned to control gel experienced adverse events that led to study withdrawal during the open-label phase.
The most frequently reported adverse events in the open-label extension were wound complications, seen in 39.5% of patients; anemia, seen in 14.1%; wound infection, seen in 9.3%; pyrexia, seen in 8.3%; and pruritus, seen in 5.9%. No more details regarding adverse events were provided.
The study was funded by Amryt Pharmaceuticals DAC. Dr. Cunningham is an employee of Amryt Pharmaceuticals. No other relevant financial relationships have been disclosed.
A version of this article first appeared on Medscape.com.
GLASGOW, Scotland – the phase 3 safety and efficacy study of the treatment.
Over 200 patients from the trial, including 105 who began treatment with a control gel, continued taking oleogel-S10 after 90 days. The current interim analysis at 12 months indicates there was a 55% reduction in the proportion of the body affected, compared with baseline.
Moreover, reductions in skin activity scores seen in the double-blind phase of the trial were maintained during the open-label extension. About 6% of patients experienced adverse events that led to withdrawal from the study.
The results show that oleogel-S10 was associated with “accelerated wound healing,” said study presenter Tracey Cunningham, MD, chief medical officer, Amryt Pharmaceuticals DAC, Dublin, which is developing the topical agent. “There were no new safety signals with this longer exposure to oleogel-S10, and patients had sustained improvement in wound burden,” she added.
The research was presented at the British Association of Dermatologists (BAD) 2022 Annual Meeting on July 6.
In April, European Medicines Agency recommended approval of oleogel-S10 for the treatment of partial-thickness skin wounds associated with dystrophic and junctional EB for patients aged 6 months and older.
However, just a month earlier, the U.S. Food and Drug Administration declined to approve the topical agent for use in EB, even after it extended its review by 3 months to include additional analyses of data previously submitted by the company.
In the post-presentation discussion, Dr. Cunningham said that the FDA had “not been satisfied at this point with the information that we have given them,” adding, “We don’t agree with the decision, and we will be appealing.”
Raman K. Madan, MD, a dermatologist at Northwell Health, Huntington, New York, who was not involved in the study, said that the reductions in wound healing seen in the study are “meaningful” and that the numbers represent a “big breakthrough.”
He told this news organization that there are “very few products on the market” for EB and that having an option for patients “would be amazing.”
“The big issue here would be cost and coverage for patients,” he said. If approved, “hopefully” it will be affordable, he added.
Dr. Madan noted that from his perspective, the majority of the reactions to the topical gel were “mild,” and there are “a lot of confounding factors” underlying the number of serious adverse events. “These patients with epidermolysis are prone to some of these issues regardless of treatment,” he said.
During her presentation, Dr. Cunningham noted that EB is a rare, debilitating condition that is characterized by varying degrees of skin fragility, blisters, and impaired wound healing that in turn lead to serious complications that affect quality of life.
While wound management is a “fundamental priority” for patients living with EB, she said, there is a “high, unmet” clinical need.
To those ends, EASE was the largest randomized controlled phase 3 efficacy and safety study in EB. In the study, 252 patients were allocated to receive oleogel-S10 or control gel plus standard-of-care nonadhesive wound dressing.
The double-blind phase of the trial met its primary endpoint: A higher proportion of patients who were given oleogel-S10 achieved first complete closure of the EB target wound by day 45, compared with patients who were given control gel, at 41.3% versus 28.9%. This equated to a relative risk of wound closure by day 45 of 1.44, or an odds ratio of 1.84 (P = .013).
However, as reported at the time by this news organization, the difference in time to wound healing by day 90 between the two patient groups was not statistically significant (P = .302), with 50.5% of oleogel-S10 patients achieving wound closure, versus 43.9% of those in the control group.
Dr. Cunningham discussed the open-label extension, which involved 205 patients from the double-blind phase (mean age, of 16.3 years) treated with oleogel-S10 or control gel plus standard-of-care nonadhesive wound dressing for 24 months.
In presenting the results of the first 12 months of the open-label extension, she said that oleogel-S10 led to “consistent” reductions in the body surface area percentage (BSAP) affected by EB. The overall reduction from baseline was 55% after receiving treatment for 15 months.
Between day 90 and month 12 of the open-label extension, the absolute BSAP was reduced from 7.4% to 5.4% for patients who had received oleogel-S10 from the start of the study. For those who started in the control group and then switched to the oleogel-S10 arm during the open-label extension, the reduction was from 8.3% to 6.4%.
Dr. Cunningham pointed out that a 1% reduction in BSAP equates approximately to the palmar surface of the hand.
Scores on the Epidermolysis Bullosa Disease Activity and Scarring Index (EBDASI) Skin activity subscale indicated that the reductions achieved in the double-blind phase of the trial were maintained.
Among patients who received oleogel-S10 from the start of the trial, EBDASI Skin scores were reduced from 19.6 at baseline to 13.5 at 12 months’ follow-up in the open-label extension. The reduction was from 19.6 to 13.5 for those who began the trial taking control gel.
Dr. Cunningham showed that adverse events of any grade were seen in 72.0% of patients who began taking oleogel-S10 at the start of the trial and in 69.5% of those who began the trial taking control gel.
Serious adverse events were recorded in 23.0% and 20.0% of patients, respectively, while 6.0% of those who initially received oleogel-S10 and 6.7% of those initially assigned to control gel experienced adverse events that led to study withdrawal during the open-label phase.
The most frequently reported adverse events in the open-label extension were wound complications, seen in 39.5% of patients; anemia, seen in 14.1%; wound infection, seen in 9.3%; pyrexia, seen in 8.3%; and pruritus, seen in 5.9%. No more details regarding adverse events were provided.
The study was funded by Amryt Pharmaceuticals DAC. Dr. Cunningham is an employee of Amryt Pharmaceuticals. No other relevant financial relationships have been disclosed.
A version of this article first appeared on Medscape.com.
GLASGOW, Scotland – the phase 3 safety and efficacy study of the treatment.
Over 200 patients from the trial, including 105 who began treatment with a control gel, continued taking oleogel-S10 after 90 days. The current interim analysis at 12 months indicates there was a 55% reduction in the proportion of the body affected, compared with baseline.
Moreover, reductions in skin activity scores seen in the double-blind phase of the trial were maintained during the open-label extension. About 6% of patients experienced adverse events that led to withdrawal from the study.
The results show that oleogel-S10 was associated with “accelerated wound healing,” said study presenter Tracey Cunningham, MD, chief medical officer, Amryt Pharmaceuticals DAC, Dublin, which is developing the topical agent. “There were no new safety signals with this longer exposure to oleogel-S10, and patients had sustained improvement in wound burden,” she added.
The research was presented at the British Association of Dermatologists (BAD) 2022 Annual Meeting on July 6.
In April, European Medicines Agency recommended approval of oleogel-S10 for the treatment of partial-thickness skin wounds associated with dystrophic and junctional EB for patients aged 6 months and older.
However, just a month earlier, the U.S. Food and Drug Administration declined to approve the topical agent for use in EB, even after it extended its review by 3 months to include additional analyses of data previously submitted by the company.
In the post-presentation discussion, Dr. Cunningham said that the FDA had “not been satisfied at this point with the information that we have given them,” adding, “We don’t agree with the decision, and we will be appealing.”
Raman K. Madan, MD, a dermatologist at Northwell Health, Huntington, New York, who was not involved in the study, said that the reductions in wound healing seen in the study are “meaningful” and that the numbers represent a “big breakthrough.”
He told this news organization that there are “very few products on the market” for EB and that having an option for patients “would be amazing.”
“The big issue here would be cost and coverage for patients,” he said. If approved, “hopefully” it will be affordable, he added.
Dr. Madan noted that from his perspective, the majority of the reactions to the topical gel were “mild,” and there are “a lot of confounding factors” underlying the number of serious adverse events. “These patients with epidermolysis are prone to some of these issues regardless of treatment,” he said.
During her presentation, Dr. Cunningham noted that EB is a rare, debilitating condition that is characterized by varying degrees of skin fragility, blisters, and impaired wound healing that in turn lead to serious complications that affect quality of life.
While wound management is a “fundamental priority” for patients living with EB, she said, there is a “high, unmet” clinical need.
To those ends, EASE was the largest randomized controlled phase 3 efficacy and safety study in EB. In the study, 252 patients were allocated to receive oleogel-S10 or control gel plus standard-of-care nonadhesive wound dressing.
The double-blind phase of the trial met its primary endpoint: A higher proportion of patients who were given oleogel-S10 achieved first complete closure of the EB target wound by day 45, compared with patients who were given control gel, at 41.3% versus 28.9%. This equated to a relative risk of wound closure by day 45 of 1.44, or an odds ratio of 1.84 (P = .013).
However, as reported at the time by this news organization, the difference in time to wound healing by day 90 between the two patient groups was not statistically significant (P = .302), with 50.5% of oleogel-S10 patients achieving wound closure, versus 43.9% of those in the control group.
Dr. Cunningham discussed the open-label extension, which involved 205 patients from the double-blind phase (mean age, of 16.3 years) treated with oleogel-S10 or control gel plus standard-of-care nonadhesive wound dressing for 24 months.
In presenting the results of the first 12 months of the open-label extension, she said that oleogel-S10 led to “consistent” reductions in the body surface area percentage (BSAP) affected by EB. The overall reduction from baseline was 55% after receiving treatment for 15 months.
Between day 90 and month 12 of the open-label extension, the absolute BSAP was reduced from 7.4% to 5.4% for patients who had received oleogel-S10 from the start of the study. For those who started in the control group and then switched to the oleogel-S10 arm during the open-label extension, the reduction was from 8.3% to 6.4%.
Dr. Cunningham pointed out that a 1% reduction in BSAP equates approximately to the palmar surface of the hand.
Scores on the Epidermolysis Bullosa Disease Activity and Scarring Index (EBDASI) Skin activity subscale indicated that the reductions achieved in the double-blind phase of the trial were maintained.
Among patients who received oleogel-S10 from the start of the trial, EBDASI Skin scores were reduced from 19.6 at baseline to 13.5 at 12 months’ follow-up in the open-label extension. The reduction was from 19.6 to 13.5 for those who began the trial taking control gel.
Dr. Cunningham showed that adverse events of any grade were seen in 72.0% of patients who began taking oleogel-S10 at the start of the trial and in 69.5% of those who began the trial taking control gel.
Serious adverse events were recorded in 23.0% and 20.0% of patients, respectively, while 6.0% of those who initially received oleogel-S10 and 6.7% of those initially assigned to control gel experienced adverse events that led to study withdrawal during the open-label phase.
The most frequently reported adverse events in the open-label extension were wound complications, seen in 39.5% of patients; anemia, seen in 14.1%; wound infection, seen in 9.3%; pyrexia, seen in 8.3%; and pruritus, seen in 5.9%. No more details regarding adverse events were provided.
The study was funded by Amryt Pharmaceuticals DAC. Dr. Cunningham is an employee of Amryt Pharmaceuticals. No other relevant financial relationships have been disclosed.
A version of this article first appeared on Medscape.com.
Eczema causes substantial burden for many infants and preschoolers
INDIANAPOLIS – . Those are key findings from a large international web-based survey that was presented during a poster session at the annual meeting of the Society for Pediatric Dermatology.
“Improved knowledge of the AD-related burden may help reinforce the medical need in the pediatric population and contribute to better and earlier adequate management of the disease,” authors led by Stephan Weidinger , MD, PhD, vice head of the department of dermatology at University Hospital Schleswig-Holstein, Kiel, Germany, wrote in the abstract.
For the study, Dr. Weidinger and colleagues evaluated 1,486 infants and preschoolers with AD aged 6 months to under 6 years, who participated in the Epidemiology of Children with Atopic Dermatitis Reporting on their Experience (EPI-CARE), an international, cross-sectional, web-based survey of children and adolescents. The study population resided in 18 countries from five regions of the world, including North America, Latin America, Europe, Middle East/Eurasia, and East Asia. Parents or guardians answered all questions for infants/preschoolers younger than 4 years of age, while preschoolers aged 4 to younger than 6 years were asked to answer questions related to the impact of AD on their health-related quality of life.
AD severity was assessed using Patient Global Assessment (PtGA), where parents or guardians described their child’s eczema severity over the last week as mild, moderate, or severe. The researchers stratified outcomes by geographic region and AD severity, which included the following atopic comorbidities: worst itch, worst skin pain, and overall sleep disturbance in the past 24 hours as measured by the 0-10 numeric rating scale, where higher scores indicate worse severity; eczema-related hospitalization in the past 12 months; and frequency and average duration of flares over the past month.
The mean age of the study participants was 3 years and 61.6% had mild disease. The most common atopic comorbidities were hay fever, asthma, and seasonal allergies, and the incidence of atopic comorbidities increased with increasing AD severity. One or more atopic comorbidities was reported in 88.3% of patients with mild AD, compared with 92.1% of those with moderate disease and 95.8% of those with severe disease. In addition, infants and preschoolers with moderate or severe AD had worse itch, skin pain, and sleep disturbances over the past 24 hours, compared with those who had mild AD.
More than half of infants and preschoolers with severe AD (54.1%) were reported to have been hospitalized in the past 12 months (this ranged from 30.2% to 71.3% across regions), as did 35% of patients with moderate AD and 32.1% of those with mild AD. In addition, 50.6% of infants and preschoolers with severe AD had more than two flares in the past month, compared with 18.1% of those with moderate AD and 6.3% of those with mild disease.
In other findings, 50.7% of infants and preschoolers with severe AD had flares than lasted an average of 2 or more weeks, compared with 20.8% of those with moderate disease and 10% of those with mild disease. Also, 78.3% of preschoolers aged 4 to less than 6 years had missed one or more days of school in the previous 4 weeks: a mean of 5.1 days among those with mild AD, a mean of 7.3 days among those with moderate AD, and a mean of 12.1 days among those with severe disease.
Raj J. Chovatiya MD, PhD, of the department of dermatology at Northwestern University, Chicago, who was asked to comment on the study, said that infants and preschoolers remain an understudied group despite the high prevalence of AD in this age range. “The results of this study demonstrate a substantial burden of disease in this population, particularly among those with more severe disease,” said Dr. Chovatiya, who also directs the university’s Center for Eczema and Itch. “This includes longer and more frequent AD flares as well as high rates of inpatient hospitalization. These findings suggest that additional research is needed to better characterize disease burden and optimize outcomes for young children with AD.”
The study was funded by Regeneron Pharmaceuticals and Sanofi. Dr. Weidinger and other coauthors reported having received institutional research grants and consulting fees from many pharmaceutical companies that manufacture drugs used for the treatment of psoriasis and eczema.
Dr. Chovatiya disclosed that he has served as an advisory board member, consultant, speaker, and/or investigator for AbbVie, Arcutis, Arena, Beiersdorf, Bristol Myers Squibb, Dermavant, Eli Lilly, EPI Health, Incyte, L’Oréal, the National Eczema Association, Pfizer, Regeneron, Sanofi, and UCB.
INDIANAPOLIS – . Those are key findings from a large international web-based survey that was presented during a poster session at the annual meeting of the Society for Pediatric Dermatology.
“Improved knowledge of the AD-related burden may help reinforce the medical need in the pediatric population and contribute to better and earlier adequate management of the disease,” authors led by Stephan Weidinger , MD, PhD, vice head of the department of dermatology at University Hospital Schleswig-Holstein, Kiel, Germany, wrote in the abstract.
For the study, Dr. Weidinger and colleagues evaluated 1,486 infants and preschoolers with AD aged 6 months to under 6 years, who participated in the Epidemiology of Children with Atopic Dermatitis Reporting on their Experience (EPI-CARE), an international, cross-sectional, web-based survey of children and adolescents. The study population resided in 18 countries from five regions of the world, including North America, Latin America, Europe, Middle East/Eurasia, and East Asia. Parents or guardians answered all questions for infants/preschoolers younger than 4 years of age, while preschoolers aged 4 to younger than 6 years were asked to answer questions related to the impact of AD on their health-related quality of life.
AD severity was assessed using Patient Global Assessment (PtGA), where parents or guardians described their child’s eczema severity over the last week as mild, moderate, or severe. The researchers stratified outcomes by geographic region and AD severity, which included the following atopic comorbidities: worst itch, worst skin pain, and overall sleep disturbance in the past 24 hours as measured by the 0-10 numeric rating scale, where higher scores indicate worse severity; eczema-related hospitalization in the past 12 months; and frequency and average duration of flares over the past month.
The mean age of the study participants was 3 years and 61.6% had mild disease. The most common atopic comorbidities were hay fever, asthma, and seasonal allergies, and the incidence of atopic comorbidities increased with increasing AD severity. One or more atopic comorbidities was reported in 88.3% of patients with mild AD, compared with 92.1% of those with moderate disease and 95.8% of those with severe disease. In addition, infants and preschoolers with moderate or severe AD had worse itch, skin pain, and sleep disturbances over the past 24 hours, compared with those who had mild AD.
More than half of infants and preschoolers with severe AD (54.1%) were reported to have been hospitalized in the past 12 months (this ranged from 30.2% to 71.3% across regions), as did 35% of patients with moderate AD and 32.1% of those with mild AD. In addition, 50.6% of infants and preschoolers with severe AD had more than two flares in the past month, compared with 18.1% of those with moderate AD and 6.3% of those with mild disease.
In other findings, 50.7% of infants and preschoolers with severe AD had flares than lasted an average of 2 or more weeks, compared with 20.8% of those with moderate disease and 10% of those with mild disease. Also, 78.3% of preschoolers aged 4 to less than 6 years had missed one or more days of school in the previous 4 weeks: a mean of 5.1 days among those with mild AD, a mean of 7.3 days among those with moderate AD, and a mean of 12.1 days among those with severe disease.
Raj J. Chovatiya MD, PhD, of the department of dermatology at Northwestern University, Chicago, who was asked to comment on the study, said that infants and preschoolers remain an understudied group despite the high prevalence of AD in this age range. “The results of this study demonstrate a substantial burden of disease in this population, particularly among those with more severe disease,” said Dr. Chovatiya, who also directs the university’s Center for Eczema and Itch. “This includes longer and more frequent AD flares as well as high rates of inpatient hospitalization. These findings suggest that additional research is needed to better characterize disease burden and optimize outcomes for young children with AD.”
The study was funded by Regeneron Pharmaceuticals and Sanofi. Dr. Weidinger and other coauthors reported having received institutional research grants and consulting fees from many pharmaceutical companies that manufacture drugs used for the treatment of psoriasis and eczema.
Dr. Chovatiya disclosed that he has served as an advisory board member, consultant, speaker, and/or investigator for AbbVie, Arcutis, Arena, Beiersdorf, Bristol Myers Squibb, Dermavant, Eli Lilly, EPI Health, Incyte, L’Oréal, the National Eczema Association, Pfizer, Regeneron, Sanofi, and UCB.
INDIANAPOLIS – . Those are key findings from a large international web-based survey that was presented during a poster session at the annual meeting of the Society for Pediatric Dermatology.
“Improved knowledge of the AD-related burden may help reinforce the medical need in the pediatric population and contribute to better and earlier adequate management of the disease,” authors led by Stephan Weidinger , MD, PhD, vice head of the department of dermatology at University Hospital Schleswig-Holstein, Kiel, Germany, wrote in the abstract.
For the study, Dr. Weidinger and colleagues evaluated 1,486 infants and preschoolers with AD aged 6 months to under 6 years, who participated in the Epidemiology of Children with Atopic Dermatitis Reporting on their Experience (EPI-CARE), an international, cross-sectional, web-based survey of children and adolescents. The study population resided in 18 countries from five regions of the world, including North America, Latin America, Europe, Middle East/Eurasia, and East Asia. Parents or guardians answered all questions for infants/preschoolers younger than 4 years of age, while preschoolers aged 4 to younger than 6 years were asked to answer questions related to the impact of AD on their health-related quality of life.
AD severity was assessed using Patient Global Assessment (PtGA), where parents or guardians described their child’s eczema severity over the last week as mild, moderate, or severe. The researchers stratified outcomes by geographic region and AD severity, which included the following atopic comorbidities: worst itch, worst skin pain, and overall sleep disturbance in the past 24 hours as measured by the 0-10 numeric rating scale, where higher scores indicate worse severity; eczema-related hospitalization in the past 12 months; and frequency and average duration of flares over the past month.
The mean age of the study participants was 3 years and 61.6% had mild disease. The most common atopic comorbidities were hay fever, asthma, and seasonal allergies, and the incidence of atopic comorbidities increased with increasing AD severity. One or more atopic comorbidities was reported in 88.3% of patients with mild AD, compared with 92.1% of those with moderate disease and 95.8% of those with severe disease. In addition, infants and preschoolers with moderate or severe AD had worse itch, skin pain, and sleep disturbances over the past 24 hours, compared with those who had mild AD.
More than half of infants and preschoolers with severe AD (54.1%) were reported to have been hospitalized in the past 12 months (this ranged from 30.2% to 71.3% across regions), as did 35% of patients with moderate AD and 32.1% of those with mild AD. In addition, 50.6% of infants and preschoolers with severe AD had more than two flares in the past month, compared with 18.1% of those with moderate AD and 6.3% of those with mild disease.
In other findings, 50.7% of infants and preschoolers with severe AD had flares than lasted an average of 2 or more weeks, compared with 20.8% of those with moderate disease and 10% of those with mild disease. Also, 78.3% of preschoolers aged 4 to less than 6 years had missed one or more days of school in the previous 4 weeks: a mean of 5.1 days among those with mild AD, a mean of 7.3 days among those with moderate AD, and a mean of 12.1 days among those with severe disease.
Raj J. Chovatiya MD, PhD, of the department of dermatology at Northwestern University, Chicago, who was asked to comment on the study, said that infants and preschoolers remain an understudied group despite the high prevalence of AD in this age range. “The results of this study demonstrate a substantial burden of disease in this population, particularly among those with more severe disease,” said Dr. Chovatiya, who also directs the university’s Center for Eczema and Itch. “This includes longer and more frequent AD flares as well as high rates of inpatient hospitalization. These findings suggest that additional research is needed to better characterize disease burden and optimize outcomes for young children with AD.”
The study was funded by Regeneron Pharmaceuticals and Sanofi. Dr. Weidinger and other coauthors reported having received institutional research grants and consulting fees from many pharmaceutical companies that manufacture drugs used for the treatment of psoriasis and eczema.
Dr. Chovatiya disclosed that he has served as an advisory board member, consultant, speaker, and/or investigator for AbbVie, Arcutis, Arena, Beiersdorf, Bristol Myers Squibb, Dermavant, Eli Lilly, EPI Health, Incyte, L’Oréal, the National Eczema Association, Pfizer, Regeneron, Sanofi, and UCB.
AT SPD 2022
European survey finds wide variations in the use of phototherapy for atopic eczema
GLASGOW, Scotland – , which points to the need for management guidelines.
Over 140 phototherapy practitioners from 27 European countries responded to the survey. Of the practitioners surveyed, 96% used narrow-band ultraviolet B (NB-UVB), and about 50% prescribed psoralen and ultraviolet A (PUVA) for adults. Fewer than 10% did so for children.
There was considerable variation in prescribing practices, “especially when it comes to dosing and treatment duration,” said study presenter Mia Steyn, MD, dermatology registrar, St. John’s Institute of Dermatology, Guy’s and St. Thomas’s Hospital, London.
These results, she said, demonstrate that “an optimal treatment modality either is not known or agreed upon” and that studies are required to determine treatment efficacy, cost, and safety “in a range of skin types.”
Dr. Steyn said that what is needed first is a set of consensus treatment guidelines, “hopefully leading to a randomized controlled trial” that would compare the various treatment options.
The research was presented at the British Association of Dermatologists (BAD) 2022 Annual Meeting on July 7.
Session co-chair Adam Fityan, MD, a consultant dermatologist at University Hospital Southampton NHS Foundation Trust, U.K., commented that the study was “fascinating” and “really helpful.”
Dr. Fityan, who was not involved with the survey, told this news organization that, “clearly, what we’ve seen is that there is a huge variation in the way everyone uses the different modalities of phototherapy.”
“Having that sort of knowledge will hopefully help us to think a bit more clearly about the regimens and protocols that we use and to maybe find the evidence that everyone needs to have the most effective protocol.”
The data from the study are also useful on an individual level, Dr. Fityan continued, as “you have no idea what anyone is doing” and whether “you are an outlier.”
Dr. Steyn said that phototherapy is commonly used for the treatment of atopic eczema, but the evidence for its efficacy, its impact on quality on life, its cost-effectiveness, and short- and long-term safety is “weak,” particularly in relation to real-life use.
Electronic survey
In lieu of a well-designed randomized controlled trial to answer these questions, the researchers set up a task force to assess how phototherapy is currently being used to treat atopic eczema across the United Kingdom and Europe so as to guide further research.
An electronic survey was devised, and 144 members of phototherapy groups from 27 European countries submitted their responses during 2020. Most responses came from the Netherlands (20), Italy (16), the United Kingdom (14), France (11), and Germany (10).
The results showed that NB-UVB was the most widely used modality of phototherapy, chosen by 96% of respondents. In addition, 17% of respondents said they also prescribed home-based NB-UVB, which was available in eight of the 27 countries.
When asked how they used NB-UVB, the majority (68%) of respondents said they had an age cutoff for use in children, which was set at an average age of 9 years and older, although the range was age 2 years to 16 years.
NBUVB was used as a second-line therapy instead of systemic treatments in up to 93% of adults and in 69% of children. It was used concomitantly with systemic treatment in up to 58% of adults and 11% of children, according to the survey responses.
For about 70% of respondents, the use of NB-UVB was determined by assessing the Fitzpatrick skin type, although almost 40% relied on clinical experience.
Frequency of treatment
NB-UVB was prescribed three times a week by 59% of respondents; 31% of respondents prescribed it twice a week; 7%, five times per week; and 2%, four times a week. The typical number of treatments was 21-30 for 53% of respondents, 0-20 treatments for 24%, and 31-40 treatments for 20%.
The dose was typically increased in 10% increments, although there were wide variations in how the treatment was stepped up. Dose was increased after each treatment by almost 50% of respondents, after every two treatments by almost 25%, and after every three treatments by approximately 15%.
For the majority (53%) of respondents, response to NBUVB was assessed after 7-15 treatments, while 43% waited until after 16-30 treatments. Success was defined as a 75% reduction in eczema from baseline by 56% of respondents, while 54% looked to patient satisfaction, and 47% relied on quality of life to determine success of treatment.
Maintenance NB-UVB was never used by 54% of respondents, but 44% said they used it occasionally, and 83% said they did not follow a weaning schedule at the end of treatment.
The most commonly reported adverse effects of NB-UVB were significant erythema, hyperpigmentation, and eczema flare, while the most commonly cited absolute contraindications included a history of melanoma, a history of squamous cell carcinoma, the use of photosensitizing medications, and claustrophobia.
Use of PUVA, UVA1
The next most commonly used phototherapy for atopic eczema was PUVA. Although it was available to 83% of respondents, only 52% of respondents had personally prescribed the treatment for adults, and only 7% prescribed it for children.
Of the respondents, 71% said they would switch from NB-UVB to PUVA if desired treatment outcomes were not achieved with the former, and 44% said they would “sometimes consider” PUVA as second-line therapy instead of systemic treatments. Only 13% said they would use it concomitantly with systemic treatment.
Ultraviolet A1 (UVA1) phototherapy was not widely available, with 66% of respondents declaring that they did not have access to this option and just 29% saying they prescribed it.
But when it was used, UVA1 was cited as being used often in adults by 24% of respondents, while 33% used it was used sometimes, and 43% said it was used rarely. It was used for children by 26% of respondents. In addition, 29% said they favored using UVA1 for chronic atopic eczema, and 33% favored using it for acute eczema while 38% had no preference over whether to use it for chronic versus acute atopic eczema.
Similarly to NB-UVB, there were wide variations in the use of PUVA and UVA1 by respondents in terms of dosing schedules, duration of treatment, and how response to treatment was measured.
No funding for the study has been reported. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
GLASGOW, Scotland – , which points to the need for management guidelines.
Over 140 phototherapy practitioners from 27 European countries responded to the survey. Of the practitioners surveyed, 96% used narrow-band ultraviolet B (NB-UVB), and about 50% prescribed psoralen and ultraviolet A (PUVA) for adults. Fewer than 10% did so for children.
There was considerable variation in prescribing practices, “especially when it comes to dosing and treatment duration,” said study presenter Mia Steyn, MD, dermatology registrar, St. John’s Institute of Dermatology, Guy’s and St. Thomas’s Hospital, London.
These results, she said, demonstrate that “an optimal treatment modality either is not known or agreed upon” and that studies are required to determine treatment efficacy, cost, and safety “in a range of skin types.”
Dr. Steyn said that what is needed first is a set of consensus treatment guidelines, “hopefully leading to a randomized controlled trial” that would compare the various treatment options.
The research was presented at the British Association of Dermatologists (BAD) 2022 Annual Meeting on July 7.
Session co-chair Adam Fityan, MD, a consultant dermatologist at University Hospital Southampton NHS Foundation Trust, U.K., commented that the study was “fascinating” and “really helpful.”
Dr. Fityan, who was not involved with the survey, told this news organization that, “clearly, what we’ve seen is that there is a huge variation in the way everyone uses the different modalities of phototherapy.”
“Having that sort of knowledge will hopefully help us to think a bit more clearly about the regimens and protocols that we use and to maybe find the evidence that everyone needs to have the most effective protocol.”
The data from the study are also useful on an individual level, Dr. Fityan continued, as “you have no idea what anyone is doing” and whether “you are an outlier.”
Dr. Steyn said that phototherapy is commonly used for the treatment of atopic eczema, but the evidence for its efficacy, its impact on quality on life, its cost-effectiveness, and short- and long-term safety is “weak,” particularly in relation to real-life use.
Electronic survey
In lieu of a well-designed randomized controlled trial to answer these questions, the researchers set up a task force to assess how phototherapy is currently being used to treat atopic eczema across the United Kingdom and Europe so as to guide further research.
An electronic survey was devised, and 144 members of phototherapy groups from 27 European countries submitted their responses during 2020. Most responses came from the Netherlands (20), Italy (16), the United Kingdom (14), France (11), and Germany (10).
The results showed that NB-UVB was the most widely used modality of phototherapy, chosen by 96% of respondents. In addition, 17% of respondents said they also prescribed home-based NB-UVB, which was available in eight of the 27 countries.
When asked how they used NB-UVB, the majority (68%) of respondents said they had an age cutoff for use in children, which was set at an average age of 9 years and older, although the range was age 2 years to 16 years.
NBUVB was used as a second-line therapy instead of systemic treatments in up to 93% of adults and in 69% of children. It was used concomitantly with systemic treatment in up to 58% of adults and 11% of children, according to the survey responses.
For about 70% of respondents, the use of NB-UVB was determined by assessing the Fitzpatrick skin type, although almost 40% relied on clinical experience.
Frequency of treatment
NB-UVB was prescribed three times a week by 59% of respondents; 31% of respondents prescribed it twice a week; 7%, five times per week; and 2%, four times a week. The typical number of treatments was 21-30 for 53% of respondents, 0-20 treatments for 24%, and 31-40 treatments for 20%.
The dose was typically increased in 10% increments, although there were wide variations in how the treatment was stepped up. Dose was increased after each treatment by almost 50% of respondents, after every two treatments by almost 25%, and after every three treatments by approximately 15%.
For the majority (53%) of respondents, response to NBUVB was assessed after 7-15 treatments, while 43% waited until after 16-30 treatments. Success was defined as a 75% reduction in eczema from baseline by 56% of respondents, while 54% looked to patient satisfaction, and 47% relied on quality of life to determine success of treatment.
Maintenance NB-UVB was never used by 54% of respondents, but 44% said they used it occasionally, and 83% said they did not follow a weaning schedule at the end of treatment.
The most commonly reported adverse effects of NB-UVB were significant erythema, hyperpigmentation, and eczema flare, while the most commonly cited absolute contraindications included a history of melanoma, a history of squamous cell carcinoma, the use of photosensitizing medications, and claustrophobia.
Use of PUVA, UVA1
The next most commonly used phototherapy for atopic eczema was PUVA. Although it was available to 83% of respondents, only 52% of respondents had personally prescribed the treatment for adults, and only 7% prescribed it for children.
Of the respondents, 71% said they would switch from NB-UVB to PUVA if desired treatment outcomes were not achieved with the former, and 44% said they would “sometimes consider” PUVA as second-line therapy instead of systemic treatments. Only 13% said they would use it concomitantly with systemic treatment.
Ultraviolet A1 (UVA1) phototherapy was not widely available, with 66% of respondents declaring that they did not have access to this option and just 29% saying they prescribed it.
But when it was used, UVA1 was cited as being used often in adults by 24% of respondents, while 33% used it was used sometimes, and 43% said it was used rarely. It was used for children by 26% of respondents. In addition, 29% said they favored using UVA1 for chronic atopic eczema, and 33% favored using it for acute eczema while 38% had no preference over whether to use it for chronic versus acute atopic eczema.
Similarly to NB-UVB, there were wide variations in the use of PUVA and UVA1 by respondents in terms of dosing schedules, duration of treatment, and how response to treatment was measured.
No funding for the study has been reported. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
GLASGOW, Scotland – , which points to the need for management guidelines.
Over 140 phototherapy practitioners from 27 European countries responded to the survey. Of the practitioners surveyed, 96% used narrow-band ultraviolet B (NB-UVB), and about 50% prescribed psoralen and ultraviolet A (PUVA) for adults. Fewer than 10% did so for children.
There was considerable variation in prescribing practices, “especially when it comes to dosing and treatment duration,” said study presenter Mia Steyn, MD, dermatology registrar, St. John’s Institute of Dermatology, Guy’s and St. Thomas’s Hospital, London.
These results, she said, demonstrate that “an optimal treatment modality either is not known or agreed upon” and that studies are required to determine treatment efficacy, cost, and safety “in a range of skin types.”
Dr. Steyn said that what is needed first is a set of consensus treatment guidelines, “hopefully leading to a randomized controlled trial” that would compare the various treatment options.
The research was presented at the British Association of Dermatologists (BAD) 2022 Annual Meeting on July 7.
Session co-chair Adam Fityan, MD, a consultant dermatologist at University Hospital Southampton NHS Foundation Trust, U.K., commented that the study was “fascinating” and “really helpful.”
Dr. Fityan, who was not involved with the survey, told this news organization that, “clearly, what we’ve seen is that there is a huge variation in the way everyone uses the different modalities of phototherapy.”
“Having that sort of knowledge will hopefully help us to think a bit more clearly about the regimens and protocols that we use and to maybe find the evidence that everyone needs to have the most effective protocol.”
The data from the study are also useful on an individual level, Dr. Fityan continued, as “you have no idea what anyone is doing” and whether “you are an outlier.”
Dr. Steyn said that phototherapy is commonly used for the treatment of atopic eczema, but the evidence for its efficacy, its impact on quality on life, its cost-effectiveness, and short- and long-term safety is “weak,” particularly in relation to real-life use.
Electronic survey
In lieu of a well-designed randomized controlled trial to answer these questions, the researchers set up a task force to assess how phototherapy is currently being used to treat atopic eczema across the United Kingdom and Europe so as to guide further research.
An electronic survey was devised, and 144 members of phototherapy groups from 27 European countries submitted their responses during 2020. Most responses came from the Netherlands (20), Italy (16), the United Kingdom (14), France (11), and Germany (10).
The results showed that NB-UVB was the most widely used modality of phototherapy, chosen by 96% of respondents. In addition, 17% of respondents said they also prescribed home-based NB-UVB, which was available in eight of the 27 countries.
When asked how they used NB-UVB, the majority (68%) of respondents said they had an age cutoff for use in children, which was set at an average age of 9 years and older, although the range was age 2 years to 16 years.
NBUVB was used as a second-line therapy instead of systemic treatments in up to 93% of adults and in 69% of children. It was used concomitantly with systemic treatment in up to 58% of adults and 11% of children, according to the survey responses.
For about 70% of respondents, the use of NB-UVB was determined by assessing the Fitzpatrick skin type, although almost 40% relied on clinical experience.
Frequency of treatment
NB-UVB was prescribed three times a week by 59% of respondents; 31% of respondents prescribed it twice a week; 7%, five times per week; and 2%, four times a week. The typical number of treatments was 21-30 for 53% of respondents, 0-20 treatments for 24%, and 31-40 treatments for 20%.
The dose was typically increased in 10% increments, although there were wide variations in how the treatment was stepped up. Dose was increased after each treatment by almost 50% of respondents, after every two treatments by almost 25%, and after every three treatments by approximately 15%.
For the majority (53%) of respondents, response to NBUVB was assessed after 7-15 treatments, while 43% waited until after 16-30 treatments. Success was defined as a 75% reduction in eczema from baseline by 56% of respondents, while 54% looked to patient satisfaction, and 47% relied on quality of life to determine success of treatment.
Maintenance NB-UVB was never used by 54% of respondents, but 44% said they used it occasionally, and 83% said they did not follow a weaning schedule at the end of treatment.
The most commonly reported adverse effects of NB-UVB were significant erythema, hyperpigmentation, and eczema flare, while the most commonly cited absolute contraindications included a history of melanoma, a history of squamous cell carcinoma, the use of photosensitizing medications, and claustrophobia.
Use of PUVA, UVA1
The next most commonly used phototherapy for atopic eczema was PUVA. Although it was available to 83% of respondents, only 52% of respondents had personally prescribed the treatment for adults, and only 7% prescribed it for children.
Of the respondents, 71% said they would switch from NB-UVB to PUVA if desired treatment outcomes were not achieved with the former, and 44% said they would “sometimes consider” PUVA as second-line therapy instead of systemic treatments. Only 13% said they would use it concomitantly with systemic treatment.
Ultraviolet A1 (UVA1) phototherapy was not widely available, with 66% of respondents declaring that they did not have access to this option and just 29% saying they prescribed it.
But when it was used, UVA1 was cited as being used often in adults by 24% of respondents, while 33% used it was used sometimes, and 43% said it was used rarely. It was used for children by 26% of respondents. In addition, 29% said they favored using UVA1 for chronic atopic eczema, and 33% favored using it for acute eczema while 38% had no preference over whether to use it for chronic versus acute atopic eczema.
Similarly to NB-UVB, there were wide variations in the use of PUVA and UVA1 by respondents in terms of dosing schedules, duration of treatment, and how response to treatment was measured.
No funding for the study has been reported. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Zoster vaccination does not appear to increase flare risk in patients with immune-mediated inflammatory disease
, according to research published in Arthritis & Rheumatology.
The authors of the study noted that individuals with IMIDs are at increased risk for herpes zoster and related complications, including postherpetic neuralgia, and that vaccination has been recommended for certain groups of patients with rheumatoid arthritis, inflammatory bowel disease, and psoriasis, by the American College of Rheumatology and other professional organizations for individuals aged 50 and older.
The study investigators used medical claims from IBM MarketScan, which provided data on patients aged 50-64 years, and data from the Centers for Medicare and Medicaid Services’ Medicare on patients aged 65 and older.
They defined presumed flares in three ways: hospitalization/emergency department visits for IMIDs, steroid treatment with a short-acting oral glucocorticoid, or treatment with a parenteral glucocorticoid injection. The investigators conducted a self-controlled case series (SCCS) analysis to examine any temporal link between the RZV and disease flares.
Among enrollees with IMIDs, 14.8% of the 55,654 patients in the MarketScan database and 43.2% of the 160,545 patients in the Medicare database received at least one dose of RZV during 2018-2019. The two-dose series completion within 6 months was 76.6% in the MarketScan group (age range, 50-64 years) and 85.4% among Medicare enrollees (age range, 65 years and older). In the SCCS analysis, 10% and 13% of patients developed flares in the control group as compared to 9%, and 11%-12% in the risk window following one or two doses of RZV among MarketScan and Medicare enrollees, respectively.
Based on these findings, the investigators concluded there was no statistically significant increase in flares subsequent to RZV administration for any IMID in either patients aged 50-64 years or patients aged 65 years and older following the first dose or second dose.
Nilanjana Bose, MD, a rheumatologist with Lonestar Rheumatology, Houston, Texas, who was not involved with the study, said that the research addresses a topic where there is uneasiness, namely vaccination in patients with IMIDs.
“Anytime you are vaccinating a patient with an autoimmune disease, especially one on a biologic, you always worry about the risk of flares,” said Dr. Bose. “Any time you tamper with the immune system, there is a risk of flares.”
The study serves as a clarification for the primary care setting, said Dr. Bose. “A lot of the time, the shingles vaccine is administered not by rheumatology but by primary care or through the pharmacy,” she said. “This study puts them [primary care physicians] at ease.”
Findings from the study reflect that most RZV vaccinations were administered in pharmacies.
One of the weaknesses of the study is that the investigators did not include patients younger than 50 years old, said Dr. Bose. “It would have been nice if they could have looked at younger patients,” she said. “We try to vaccinate all our [immunocompromised] adult patients, even the younger ones, because they are also at risk for shingles.”
Given that there are increasing options of medical therapies in rheumatology that are immunomodulatory, the subject of vaccination for patients is often one of discussion, added Dr. Bose.
Arthur Kavanaugh, MD, professor of medicine, University of California San Diego (UCSD), La Jolla, Calif., and director of the Center for Innovative Therapy in the UCSD Division of Rheumatology, Allergy, and Immunology, told this news organization that a strength of the study is its large numbers of patients but noted the shortcoming of using claims data. “Claims data has inherent limitations, such as the lack of detailed granular data on the patients,” wrote Dr. Kavanaugh, who was not involved with the study. He described this investigation as “really about the first evidence that I am aware of addressing this issue.”
No funding source was listed. One author disclosed having received research grants and consulting fees received from Pfizer and GSK for unrelated work; the other authors had no disclosures. Dr. Bose and Dr. Kavanaugh had no relevant disclosures.
, according to research published in Arthritis & Rheumatology.
The authors of the study noted that individuals with IMIDs are at increased risk for herpes zoster and related complications, including postherpetic neuralgia, and that vaccination has been recommended for certain groups of patients with rheumatoid arthritis, inflammatory bowel disease, and psoriasis, by the American College of Rheumatology and other professional organizations for individuals aged 50 and older.
The study investigators used medical claims from IBM MarketScan, which provided data on patients aged 50-64 years, and data from the Centers for Medicare and Medicaid Services’ Medicare on patients aged 65 and older.
They defined presumed flares in three ways: hospitalization/emergency department visits for IMIDs, steroid treatment with a short-acting oral glucocorticoid, or treatment with a parenteral glucocorticoid injection. The investigators conducted a self-controlled case series (SCCS) analysis to examine any temporal link between the RZV and disease flares.
Among enrollees with IMIDs, 14.8% of the 55,654 patients in the MarketScan database and 43.2% of the 160,545 patients in the Medicare database received at least one dose of RZV during 2018-2019. The two-dose series completion within 6 months was 76.6% in the MarketScan group (age range, 50-64 years) and 85.4% among Medicare enrollees (age range, 65 years and older). In the SCCS analysis, 10% and 13% of patients developed flares in the control group as compared to 9%, and 11%-12% in the risk window following one or two doses of RZV among MarketScan and Medicare enrollees, respectively.
Based on these findings, the investigators concluded there was no statistically significant increase in flares subsequent to RZV administration for any IMID in either patients aged 50-64 years or patients aged 65 years and older following the first dose or second dose.
Nilanjana Bose, MD, a rheumatologist with Lonestar Rheumatology, Houston, Texas, who was not involved with the study, said that the research addresses a topic where there is uneasiness, namely vaccination in patients with IMIDs.
“Anytime you are vaccinating a patient with an autoimmune disease, especially one on a biologic, you always worry about the risk of flares,” said Dr. Bose. “Any time you tamper with the immune system, there is a risk of flares.”
The study serves as a clarification for the primary care setting, said Dr. Bose. “A lot of the time, the shingles vaccine is administered not by rheumatology but by primary care or through the pharmacy,” she said. “This study puts them [primary care physicians] at ease.”
Findings from the study reflect that most RZV vaccinations were administered in pharmacies.
One of the weaknesses of the study is that the investigators did not include patients younger than 50 years old, said Dr. Bose. “It would have been nice if they could have looked at younger patients,” she said. “We try to vaccinate all our [immunocompromised] adult patients, even the younger ones, because they are also at risk for shingles.”
Given that there are increasing options of medical therapies in rheumatology that are immunomodulatory, the subject of vaccination for patients is often one of discussion, added Dr. Bose.
Arthur Kavanaugh, MD, professor of medicine, University of California San Diego (UCSD), La Jolla, Calif., and director of the Center for Innovative Therapy in the UCSD Division of Rheumatology, Allergy, and Immunology, told this news organization that a strength of the study is its large numbers of patients but noted the shortcoming of using claims data. “Claims data has inherent limitations, such as the lack of detailed granular data on the patients,” wrote Dr. Kavanaugh, who was not involved with the study. He described this investigation as “really about the first evidence that I am aware of addressing this issue.”
No funding source was listed. One author disclosed having received research grants and consulting fees received from Pfizer and GSK for unrelated work; the other authors had no disclosures. Dr. Bose and Dr. Kavanaugh had no relevant disclosures.
, according to research published in Arthritis & Rheumatology.
The authors of the study noted that individuals with IMIDs are at increased risk for herpes zoster and related complications, including postherpetic neuralgia, and that vaccination has been recommended for certain groups of patients with rheumatoid arthritis, inflammatory bowel disease, and psoriasis, by the American College of Rheumatology and other professional organizations for individuals aged 50 and older.
The study investigators used medical claims from IBM MarketScan, which provided data on patients aged 50-64 years, and data from the Centers for Medicare and Medicaid Services’ Medicare on patients aged 65 and older.
They defined presumed flares in three ways: hospitalization/emergency department visits for IMIDs, steroid treatment with a short-acting oral glucocorticoid, or treatment with a parenteral glucocorticoid injection. The investigators conducted a self-controlled case series (SCCS) analysis to examine any temporal link between the RZV and disease flares.
Among enrollees with IMIDs, 14.8% of the 55,654 patients in the MarketScan database and 43.2% of the 160,545 patients in the Medicare database received at least one dose of RZV during 2018-2019. The two-dose series completion within 6 months was 76.6% in the MarketScan group (age range, 50-64 years) and 85.4% among Medicare enrollees (age range, 65 years and older). In the SCCS analysis, 10% and 13% of patients developed flares in the control group as compared to 9%, and 11%-12% in the risk window following one or two doses of RZV among MarketScan and Medicare enrollees, respectively.
Based on these findings, the investigators concluded there was no statistically significant increase in flares subsequent to RZV administration for any IMID in either patients aged 50-64 years or patients aged 65 years and older following the first dose or second dose.
Nilanjana Bose, MD, a rheumatologist with Lonestar Rheumatology, Houston, Texas, who was not involved with the study, said that the research addresses a topic where there is uneasiness, namely vaccination in patients with IMIDs.
“Anytime you are vaccinating a patient with an autoimmune disease, especially one on a biologic, you always worry about the risk of flares,” said Dr. Bose. “Any time you tamper with the immune system, there is a risk of flares.”
The study serves as a clarification for the primary care setting, said Dr. Bose. “A lot of the time, the shingles vaccine is administered not by rheumatology but by primary care or through the pharmacy,” she said. “This study puts them [primary care physicians] at ease.”
Findings from the study reflect that most RZV vaccinations were administered in pharmacies.
One of the weaknesses of the study is that the investigators did not include patients younger than 50 years old, said Dr. Bose. “It would have been nice if they could have looked at younger patients,” she said. “We try to vaccinate all our [immunocompromised] adult patients, even the younger ones, because they are also at risk for shingles.”
Given that there are increasing options of medical therapies in rheumatology that are immunomodulatory, the subject of vaccination for patients is often one of discussion, added Dr. Bose.
Arthur Kavanaugh, MD, professor of medicine, University of California San Diego (UCSD), La Jolla, Calif., and director of the Center for Innovative Therapy in the UCSD Division of Rheumatology, Allergy, and Immunology, told this news organization that a strength of the study is its large numbers of patients but noted the shortcoming of using claims data. “Claims data has inherent limitations, such as the lack of detailed granular data on the patients,” wrote Dr. Kavanaugh, who was not involved with the study. He described this investigation as “really about the first evidence that I am aware of addressing this issue.”
No funding source was listed. One author disclosed having received research grants and consulting fees received from Pfizer and GSK for unrelated work; the other authors had no disclosures. Dr. Bose and Dr. Kavanaugh had no relevant disclosures.
Large study reaffirms rare risk of TNF inhibitor–induced psoriasis in patients with RA, IBD
according to a new study published in JAMA Dermatology.
Despite this finding, the authors of the large Danish nationwide cohort study noted that TNFi-induced psoriasis is still a rare adverse event. “Practitioners and patients should be aware and observant of the potential for TNFi-associated psoriasis during TNFi treatment but keep in mind that the absolute risk appears to be low,” David Thein, MB, of the department of dermatology at Bispebjerg Hospital, University of Copenhagen, and colleagues wrote in the study.
They analyzed 109,085 patients with RA and IBD enrolled in Danish national registries between 1995 and 2018 without a previous diagnosis of psoriasis, who received either TNFi (20,910 patients) or conventional treatments (108,024 patients) and were followed for 5 years. They were a mean of 50 years old when they started treatment, 62% were women, with 87.8% of patients in the TNFi group receiving prior conventional therapy and 1% of patients in the conventional therapy group receiving prior TNFi treatment.
The investigators assessed the risk of developing any psoriasis, nonpustular psoriasis, and pustular psoriasis in the two groups using ICD-10 codes as well as a record of two consecutive prescriptions for topical vitamin D analogs.
Overall, 1,471 patients (1.4%) developed psoriasis of any type; 1,332 had non-pustular psoriasis, 127 had palmoplantar pustulosis, and 12 had generalized pustulosis.
The incidence rate of developing any psoriasis was 3.0 per 1,000 patient-years (95% confidence interval, 2.9-3.2) for patients receiving conventional therapy and 7.8 per 1,000 patient-years (95% CI, 7.5-8.9) for patients receiving TNFi treatment. Compared with conventional treatment, the risk of developing nonpustular psoriasis was twofold higher among patients receiving TNFi treatment (hazard ratio, 2.12; 95% CI, 1.87-2.40; P < .001). The risk of developing pustular psoriasis was more than sixfold higher among those on a TNFi (HR, 6.50; 95% CI, 4.60-9.23; P < .001).
Dr. Thein and colleagues estimated that the exposure needed to harm 1 additional patient was 241 patient-years for any psoriasis type, 342 patient-years for nonpustular psoriasis, and 909 patient-years for pustular psoriasis, with an estimated absolute risk difference of 5 per 1,000 patient-years.
Best evidence to date on risk
Asked to comment on the study findings, Anthony Fernandez, MD, PhD, director of medical dermatology at the Cleveland Clinic, said that he applauded the researchers for performing this well-designed study to determine the risk of TNF inhibitor–induced psoriasis in patients with RA and IBD.
The strengths of the study include excluding patients with a history of psoriasis to rule out disease recurrence and having a large comparator group of patients with IBD and RA who were taking medications other than TNF inhibitors, while one limitation was the potential accuracy of the ICD-10 codes used as the basis for diagnosing psoriasis. “It’s probably closer to the truth of what the true risk is compared to studies done in the past,” he said in an interview.
Dr. Fernandez noted that the results aren’t likely to change how dermatologists, rheumatologists, or gastroenterologists practice, but the message to stay the course in initially treating TNFi-induced psoriasis also holds value. “We don’t need to change anything in our clinical practice when it comes to TNF-alpha inhibitors.”
For patients with RA or IBD who develop TNFi-induced psoriasis with disease that is well controlled with TNFi treatment, keeping them on that treatment is a priority, Dr. Fernandez explained. “The first and foremost goal is, if the TNF inhibitor is working very well to control the disease that it was prescribed for, then you exhaust your efforts to try to control the psoriasis and allow those patients to stay on the TNF inhibitor.”
In his experience, most patients with RA and IBD who develop TNFi-induced psoriasis are controlled with topical medications. Switching to another TNFi is not recommended, he noted, as patients are “likely to have that reaction with any TNF inhibitor.”
However, Dr. Fernandez said that won’t be an option for all patients with RA and IBD. “In some patients you do simply have to stop the TNF inhibitor” and try an alternative treatment with a different mechanism of action.
The cause of TNFi-induced psoriasis is still not well understood. “There certainly is evidence to support that interferon alpha production by plasmacytoid dendritic cells is playing some role in this phenomenon,” but there is “more to the story” and unanswered questions remain, Dr. Fernandez said.
What’s most interesting about this phenomenon, he added, is that “patients can develop it at any time when exposed to a TNF inhibitor.” For instance, most patients develop drug reactions within 2-3 weeks of starting a treatment, but TNFi-induced psoriasis can appear after a single dose or several years after initiating treatment.
“Why so few patients, and why is there such variability in terms of how long they’re on the TNF inhibitor before the reaction occurs?” he asked. “That really points to ... some other trigger besides exposure to the TNF inhibitor needed for the initiation of this reaction.”
He noted that it would be valuable to identify triggers – or the most likely triggers – which would be challenging, but could “potentially impact clinical practice.”
The authors reported personal and institutional relationships in the form of personal and institutional research grants, honoraria, personal fees, investigator fees paid to university, consultancies, and speaker’s bureau positions for a variety of pharmaceutical companies, data companies, hospitals, and foundations. Dr. Fernandez reported he has nonbranded speaking, consulting, and research relationships with AbbVie and Novartis; and is a consultant for UCB, Bristol-Myers Squibb, and Boehringer Ingelheim on related products.
according to a new study published in JAMA Dermatology.
Despite this finding, the authors of the large Danish nationwide cohort study noted that TNFi-induced psoriasis is still a rare adverse event. “Practitioners and patients should be aware and observant of the potential for TNFi-associated psoriasis during TNFi treatment but keep in mind that the absolute risk appears to be low,” David Thein, MB, of the department of dermatology at Bispebjerg Hospital, University of Copenhagen, and colleagues wrote in the study.
They analyzed 109,085 patients with RA and IBD enrolled in Danish national registries between 1995 and 2018 without a previous diagnosis of psoriasis, who received either TNFi (20,910 patients) or conventional treatments (108,024 patients) and were followed for 5 years. They were a mean of 50 years old when they started treatment, 62% were women, with 87.8% of patients in the TNFi group receiving prior conventional therapy and 1% of patients in the conventional therapy group receiving prior TNFi treatment.
The investigators assessed the risk of developing any psoriasis, nonpustular psoriasis, and pustular psoriasis in the two groups using ICD-10 codes as well as a record of two consecutive prescriptions for topical vitamin D analogs.
Overall, 1,471 patients (1.4%) developed psoriasis of any type; 1,332 had non-pustular psoriasis, 127 had palmoplantar pustulosis, and 12 had generalized pustulosis.
The incidence rate of developing any psoriasis was 3.0 per 1,000 patient-years (95% confidence interval, 2.9-3.2) for patients receiving conventional therapy and 7.8 per 1,000 patient-years (95% CI, 7.5-8.9) for patients receiving TNFi treatment. Compared with conventional treatment, the risk of developing nonpustular psoriasis was twofold higher among patients receiving TNFi treatment (hazard ratio, 2.12; 95% CI, 1.87-2.40; P < .001). The risk of developing pustular psoriasis was more than sixfold higher among those on a TNFi (HR, 6.50; 95% CI, 4.60-9.23; P < .001).
Dr. Thein and colleagues estimated that the exposure needed to harm 1 additional patient was 241 patient-years for any psoriasis type, 342 patient-years for nonpustular psoriasis, and 909 patient-years for pustular psoriasis, with an estimated absolute risk difference of 5 per 1,000 patient-years.
Best evidence to date on risk
Asked to comment on the study findings, Anthony Fernandez, MD, PhD, director of medical dermatology at the Cleveland Clinic, said that he applauded the researchers for performing this well-designed study to determine the risk of TNF inhibitor–induced psoriasis in patients with RA and IBD.
The strengths of the study include excluding patients with a history of psoriasis to rule out disease recurrence and having a large comparator group of patients with IBD and RA who were taking medications other than TNF inhibitors, while one limitation was the potential accuracy of the ICD-10 codes used as the basis for diagnosing psoriasis. “It’s probably closer to the truth of what the true risk is compared to studies done in the past,” he said in an interview.
Dr. Fernandez noted that the results aren’t likely to change how dermatologists, rheumatologists, or gastroenterologists practice, but the message to stay the course in initially treating TNFi-induced psoriasis also holds value. “We don’t need to change anything in our clinical practice when it comes to TNF-alpha inhibitors.”
For patients with RA or IBD who develop TNFi-induced psoriasis with disease that is well controlled with TNFi treatment, keeping them on that treatment is a priority, Dr. Fernandez explained. “The first and foremost goal is, if the TNF inhibitor is working very well to control the disease that it was prescribed for, then you exhaust your efforts to try to control the psoriasis and allow those patients to stay on the TNF inhibitor.”
In his experience, most patients with RA and IBD who develop TNFi-induced psoriasis are controlled with topical medications. Switching to another TNFi is not recommended, he noted, as patients are “likely to have that reaction with any TNF inhibitor.”
However, Dr. Fernandez said that won’t be an option for all patients with RA and IBD. “In some patients you do simply have to stop the TNF inhibitor” and try an alternative treatment with a different mechanism of action.
The cause of TNFi-induced psoriasis is still not well understood. “There certainly is evidence to support that interferon alpha production by plasmacytoid dendritic cells is playing some role in this phenomenon,” but there is “more to the story” and unanswered questions remain, Dr. Fernandez said.
What’s most interesting about this phenomenon, he added, is that “patients can develop it at any time when exposed to a TNF inhibitor.” For instance, most patients develop drug reactions within 2-3 weeks of starting a treatment, but TNFi-induced psoriasis can appear after a single dose or several years after initiating treatment.
“Why so few patients, and why is there such variability in terms of how long they’re on the TNF inhibitor before the reaction occurs?” he asked. “That really points to ... some other trigger besides exposure to the TNF inhibitor needed for the initiation of this reaction.”
He noted that it would be valuable to identify triggers – or the most likely triggers – which would be challenging, but could “potentially impact clinical practice.”
The authors reported personal and institutional relationships in the form of personal and institutional research grants, honoraria, personal fees, investigator fees paid to university, consultancies, and speaker’s bureau positions for a variety of pharmaceutical companies, data companies, hospitals, and foundations. Dr. Fernandez reported he has nonbranded speaking, consulting, and research relationships with AbbVie and Novartis; and is a consultant for UCB, Bristol-Myers Squibb, and Boehringer Ingelheim on related products.
according to a new study published in JAMA Dermatology.
Despite this finding, the authors of the large Danish nationwide cohort study noted that TNFi-induced psoriasis is still a rare adverse event. “Practitioners and patients should be aware and observant of the potential for TNFi-associated psoriasis during TNFi treatment but keep in mind that the absolute risk appears to be low,” David Thein, MB, of the department of dermatology at Bispebjerg Hospital, University of Copenhagen, and colleagues wrote in the study.
They analyzed 109,085 patients with RA and IBD enrolled in Danish national registries between 1995 and 2018 without a previous diagnosis of psoriasis, who received either TNFi (20,910 patients) or conventional treatments (108,024 patients) and were followed for 5 years. They were a mean of 50 years old when they started treatment, 62% were women, with 87.8% of patients in the TNFi group receiving prior conventional therapy and 1% of patients in the conventional therapy group receiving prior TNFi treatment.
The investigators assessed the risk of developing any psoriasis, nonpustular psoriasis, and pustular psoriasis in the two groups using ICD-10 codes as well as a record of two consecutive prescriptions for topical vitamin D analogs.
Overall, 1,471 patients (1.4%) developed psoriasis of any type; 1,332 had non-pustular psoriasis, 127 had palmoplantar pustulosis, and 12 had generalized pustulosis.
The incidence rate of developing any psoriasis was 3.0 per 1,000 patient-years (95% confidence interval, 2.9-3.2) for patients receiving conventional therapy and 7.8 per 1,000 patient-years (95% CI, 7.5-8.9) for patients receiving TNFi treatment. Compared with conventional treatment, the risk of developing nonpustular psoriasis was twofold higher among patients receiving TNFi treatment (hazard ratio, 2.12; 95% CI, 1.87-2.40; P < .001). The risk of developing pustular psoriasis was more than sixfold higher among those on a TNFi (HR, 6.50; 95% CI, 4.60-9.23; P < .001).
Dr. Thein and colleagues estimated that the exposure needed to harm 1 additional patient was 241 patient-years for any psoriasis type, 342 patient-years for nonpustular psoriasis, and 909 patient-years for pustular psoriasis, with an estimated absolute risk difference of 5 per 1,000 patient-years.
Best evidence to date on risk
Asked to comment on the study findings, Anthony Fernandez, MD, PhD, director of medical dermatology at the Cleveland Clinic, said that he applauded the researchers for performing this well-designed study to determine the risk of TNF inhibitor–induced psoriasis in patients with RA and IBD.
The strengths of the study include excluding patients with a history of psoriasis to rule out disease recurrence and having a large comparator group of patients with IBD and RA who were taking medications other than TNF inhibitors, while one limitation was the potential accuracy of the ICD-10 codes used as the basis for diagnosing psoriasis. “It’s probably closer to the truth of what the true risk is compared to studies done in the past,” he said in an interview.
Dr. Fernandez noted that the results aren’t likely to change how dermatologists, rheumatologists, or gastroenterologists practice, but the message to stay the course in initially treating TNFi-induced psoriasis also holds value. “We don’t need to change anything in our clinical practice when it comes to TNF-alpha inhibitors.”
For patients with RA or IBD who develop TNFi-induced psoriasis with disease that is well controlled with TNFi treatment, keeping them on that treatment is a priority, Dr. Fernandez explained. “The first and foremost goal is, if the TNF inhibitor is working very well to control the disease that it was prescribed for, then you exhaust your efforts to try to control the psoriasis and allow those patients to stay on the TNF inhibitor.”
In his experience, most patients with RA and IBD who develop TNFi-induced psoriasis are controlled with topical medications. Switching to another TNFi is not recommended, he noted, as patients are “likely to have that reaction with any TNF inhibitor.”
However, Dr. Fernandez said that won’t be an option for all patients with RA and IBD. “In some patients you do simply have to stop the TNF inhibitor” and try an alternative treatment with a different mechanism of action.
The cause of TNFi-induced psoriasis is still not well understood. “There certainly is evidence to support that interferon alpha production by plasmacytoid dendritic cells is playing some role in this phenomenon,” but there is “more to the story” and unanswered questions remain, Dr. Fernandez said.
What’s most interesting about this phenomenon, he added, is that “patients can develop it at any time when exposed to a TNF inhibitor.” For instance, most patients develop drug reactions within 2-3 weeks of starting a treatment, but TNFi-induced psoriasis can appear after a single dose or several years after initiating treatment.
“Why so few patients, and why is there such variability in terms of how long they’re on the TNF inhibitor before the reaction occurs?” he asked. “That really points to ... some other trigger besides exposure to the TNF inhibitor needed for the initiation of this reaction.”
He noted that it would be valuable to identify triggers – or the most likely triggers – which would be challenging, but could “potentially impact clinical practice.”
The authors reported personal and institutional relationships in the form of personal and institutional research grants, honoraria, personal fees, investigator fees paid to university, consultancies, and speaker’s bureau positions for a variety of pharmaceutical companies, data companies, hospitals, and foundations. Dr. Fernandez reported he has nonbranded speaking, consulting, and research relationships with AbbVie and Novartis; and is a consultant for UCB, Bristol-Myers Squibb, and Boehringer Ingelheim on related products.
FROM JAMA DERMATOLOGY
Compulsivity contributes to poor outcomes in body-focused repetitive behaviors
Although body-focused repetitive behaviors (BFRBs), specifically trichotillomania and skin-picking disorder, are similar in clinical presentation to aspects of obsessive-compulsive disorder (OCD), the role of compulsivity in TTM and SPD has not been well studied, wrote Jon E. Grant, MD, of the University of Chicago and colleagues.
In a study published in the Journal of Psychiatric Research, the authors recruited 69 women and 22 men who met DSM-5 criteria for TTM and SPD. Participants completed diagnostic interviews, symptom inventories, and measures of disability/functioning. Compulsivity was measured using the 15-item Cambridge-Chicago Compulsivity Trait Scale (CHI-T). The average age of the participants was 30.9 years; 48 had TTM, 37 had SPD, and 2 had both conditions.
Overall, total CHI-T scores were significantly correlated with worse disability and quality of life, based on the Quality of Life Inventory (P = .0278) and the Sheehan Disability Scale (P = .0085) but not with severity of TTM or SPD symptoms. TTM and SPD symptoms were assessed using the Massachusetts General Hospital Hair Pulling Scale and the Skin Picking Symptom Symptom Assessment Scale.
“In the current study, we did not find a link between conventional symptom severity measures for BFRBs and disability or quality of life, whereas trans-diagnostic compulsivity did correlate with these clinically important parameters,” the researchers wrote in their discussion. “These findings might suggest the current symptom measures for BFRBs are not including an important aspect of the disease and that a fuller understanding of these symptoms requires measurement of compulsivity. Including validated measures of compulsivity in clinical trials of therapy or medication would also seem to be important for future work,” they said.
The study findings were limited by several factors including the use of a community sample that may not generalize to a clinical setting, the researchers noted. Other limitations include the cross-sectional design, which prevents conclusions about causality, the lack of a control group, and the relatively small sample size, they said.
However, the study is the first known to use a validated compulsivity measure to assess BFRBs, and the results suggest a clinically relevant impact of compulsivity on both psychosocial dysfunction and poor quality of life in this patient population, with possible implications for treatment, the researchers wrote.
The study received no outside funding. Lead author Dr. Grant disclosed research grants from Otsuka and Biohaven Pharmaceuticals, yearly compensation from Springer Publishing for acting as editor in chief of the Journal of Gambling Studies, and royalties from Oxford University Press, American Psychiatric Publishing, Norton Press, and McGraw Hill.
Although body-focused repetitive behaviors (BFRBs), specifically trichotillomania and skin-picking disorder, are similar in clinical presentation to aspects of obsessive-compulsive disorder (OCD), the role of compulsivity in TTM and SPD has not been well studied, wrote Jon E. Grant, MD, of the University of Chicago and colleagues.
In a study published in the Journal of Psychiatric Research, the authors recruited 69 women and 22 men who met DSM-5 criteria for TTM and SPD. Participants completed diagnostic interviews, symptom inventories, and measures of disability/functioning. Compulsivity was measured using the 15-item Cambridge-Chicago Compulsivity Trait Scale (CHI-T). The average age of the participants was 30.9 years; 48 had TTM, 37 had SPD, and 2 had both conditions.
Overall, total CHI-T scores were significantly correlated with worse disability and quality of life, based on the Quality of Life Inventory (P = .0278) and the Sheehan Disability Scale (P = .0085) but not with severity of TTM or SPD symptoms. TTM and SPD symptoms were assessed using the Massachusetts General Hospital Hair Pulling Scale and the Skin Picking Symptom Symptom Assessment Scale.
“In the current study, we did not find a link between conventional symptom severity measures for BFRBs and disability or quality of life, whereas trans-diagnostic compulsivity did correlate with these clinically important parameters,” the researchers wrote in their discussion. “These findings might suggest the current symptom measures for BFRBs are not including an important aspect of the disease and that a fuller understanding of these symptoms requires measurement of compulsivity. Including validated measures of compulsivity in clinical trials of therapy or medication would also seem to be important for future work,” they said.
The study findings were limited by several factors including the use of a community sample that may not generalize to a clinical setting, the researchers noted. Other limitations include the cross-sectional design, which prevents conclusions about causality, the lack of a control group, and the relatively small sample size, they said.
However, the study is the first known to use a validated compulsivity measure to assess BFRBs, and the results suggest a clinically relevant impact of compulsivity on both psychosocial dysfunction and poor quality of life in this patient population, with possible implications for treatment, the researchers wrote.
The study received no outside funding. Lead author Dr. Grant disclosed research grants from Otsuka and Biohaven Pharmaceuticals, yearly compensation from Springer Publishing for acting as editor in chief of the Journal of Gambling Studies, and royalties from Oxford University Press, American Psychiatric Publishing, Norton Press, and McGraw Hill.
Although body-focused repetitive behaviors (BFRBs), specifically trichotillomania and skin-picking disorder, are similar in clinical presentation to aspects of obsessive-compulsive disorder (OCD), the role of compulsivity in TTM and SPD has not been well studied, wrote Jon E. Grant, MD, of the University of Chicago and colleagues.
In a study published in the Journal of Psychiatric Research, the authors recruited 69 women and 22 men who met DSM-5 criteria for TTM and SPD. Participants completed diagnostic interviews, symptom inventories, and measures of disability/functioning. Compulsivity was measured using the 15-item Cambridge-Chicago Compulsivity Trait Scale (CHI-T). The average age of the participants was 30.9 years; 48 had TTM, 37 had SPD, and 2 had both conditions.
Overall, total CHI-T scores were significantly correlated with worse disability and quality of life, based on the Quality of Life Inventory (P = .0278) and the Sheehan Disability Scale (P = .0085) but not with severity of TTM or SPD symptoms. TTM and SPD symptoms were assessed using the Massachusetts General Hospital Hair Pulling Scale and the Skin Picking Symptom Symptom Assessment Scale.
“In the current study, we did not find a link between conventional symptom severity measures for BFRBs and disability or quality of life, whereas trans-diagnostic compulsivity did correlate with these clinically important parameters,” the researchers wrote in their discussion. “These findings might suggest the current symptom measures for BFRBs are not including an important aspect of the disease and that a fuller understanding of these symptoms requires measurement of compulsivity. Including validated measures of compulsivity in clinical trials of therapy or medication would also seem to be important for future work,” they said.
The study findings were limited by several factors including the use of a community sample that may not generalize to a clinical setting, the researchers noted. Other limitations include the cross-sectional design, which prevents conclusions about causality, the lack of a control group, and the relatively small sample size, they said.
However, the study is the first known to use a validated compulsivity measure to assess BFRBs, and the results suggest a clinically relevant impact of compulsivity on both psychosocial dysfunction and poor quality of life in this patient population, with possible implications for treatment, the researchers wrote.
The study received no outside funding. Lead author Dr. Grant disclosed research grants from Otsuka and Biohaven Pharmaceuticals, yearly compensation from Springer Publishing for acting as editor in chief of the Journal of Gambling Studies, and royalties from Oxford University Press, American Psychiatric Publishing, Norton Press, and McGraw Hill.
FROM THE JOURNAL OF PSYCHIATRIC RESEARCH