User login
Canagliflozin reduces total CV burden in T2D patients with CKD
Key clinical point: Canagliflozin reduced not only the risk for the first cardiovascular (CV) events but also subsequent CV events in patients with type 2 diabetes and chronic kidney disease (CKD), with absolute benefits being greater for total CV events.
Major finding: Canagliflozin reduced the risk for the first (hazard ratio 0.74; P < .001) and total (incidence rate ratio 0.71; P < .001) CV events by 26% and 29%, respectively, with the absolute risk difference per 1000 patients treated over 2.5 years being −44 (95% CI −67 to −21) and −73 (95% CI −114 to −33) for the first and total CV events, respectively.
Study details: This was a post hoc analysis of the CREDENCE trial including patients with type 2 diabetes (T2D; glycated hemoglobin 6.5%-12.0%) and CKD who were randomly assigned to receive canagliflozin or placebo.
Disclosures: The CREDENCE trial was sponsored by Janssen Research & Development, LLC. Some authors reported serving as advisory board members, speakers, or consultants, or receiving honoraria, research support, consulting, travel, or scientific presentation fees from various sources, including Janssen.
Source: Li JW et al. Effect of canagliflozin on total cardiovascular burden in patients with diabetes and chronic kidney disease: A post hoc analysis from the CREDENCE trial. J Am Heart Assoc. 2022;11(16):e025045 (Aug 5). Doi: 10.1161/JAHA.121.025045
Key clinical point: Canagliflozin reduced not only the risk for the first cardiovascular (CV) events but also subsequent CV events in patients with type 2 diabetes and chronic kidney disease (CKD), with absolute benefits being greater for total CV events.
Major finding: Canagliflozin reduced the risk for the first (hazard ratio 0.74; P < .001) and total (incidence rate ratio 0.71; P < .001) CV events by 26% and 29%, respectively, with the absolute risk difference per 1000 patients treated over 2.5 years being −44 (95% CI −67 to −21) and −73 (95% CI −114 to −33) for the first and total CV events, respectively.
Study details: This was a post hoc analysis of the CREDENCE trial including patients with type 2 diabetes (T2D; glycated hemoglobin 6.5%-12.0%) and CKD who were randomly assigned to receive canagliflozin or placebo.
Disclosures: The CREDENCE trial was sponsored by Janssen Research & Development, LLC. Some authors reported serving as advisory board members, speakers, or consultants, or receiving honoraria, research support, consulting, travel, or scientific presentation fees from various sources, including Janssen.
Source: Li JW et al. Effect of canagliflozin on total cardiovascular burden in patients with diabetes and chronic kidney disease: A post hoc analysis from the CREDENCE trial. J Am Heart Assoc. 2022;11(16):e025045 (Aug 5). Doi: 10.1161/JAHA.121.025045
Key clinical point: Canagliflozin reduced not only the risk for the first cardiovascular (CV) events but also subsequent CV events in patients with type 2 diabetes and chronic kidney disease (CKD), with absolute benefits being greater for total CV events.
Major finding: Canagliflozin reduced the risk for the first (hazard ratio 0.74; P < .001) and total (incidence rate ratio 0.71; P < .001) CV events by 26% and 29%, respectively, with the absolute risk difference per 1000 patients treated over 2.5 years being −44 (95% CI −67 to −21) and −73 (95% CI −114 to −33) for the first and total CV events, respectively.
Study details: This was a post hoc analysis of the CREDENCE trial including patients with type 2 diabetes (T2D; glycated hemoglobin 6.5%-12.0%) and CKD who were randomly assigned to receive canagliflozin or placebo.
Disclosures: The CREDENCE trial was sponsored by Janssen Research & Development, LLC. Some authors reported serving as advisory board members, speakers, or consultants, or receiving honoraria, research support, consulting, travel, or scientific presentation fees from various sources, including Janssen.
Source: Li JW et al. Effect of canagliflozin on total cardiovascular burden in patients with diabetes and chronic kidney disease: A post hoc analysis from the CREDENCE trial. J Am Heart Assoc. 2022;11(16):e025045 (Aug 5). Doi: 10.1161/JAHA.121.025045
At EASD, docs to eye new tactics for type 2 diabetes
Highlights of the European Association for the Study of Diabetes 2022 annual meeting include new data on weight loss with the blockbuster twincretin tirzepatide and on the effects of dapagliflozin on heart failure in people with diabetes, as well as updated guidelines for type 2 diabetes management.
The EASD meeting will take place Sept. 19-23 in Stockholm. It will be the first in-person meeting since 2019 but will also feature live-streamed content for participants around the world.
“The EASD congress will cover all the different areas and aspects of diabetes research – clinical, basic, epidemiologic, and psychological,” EASD President Stefano Del Prato, MD, told this news organization.
What attendees should expect, said Del Prato of the University of Pisa (Italy), “is the pleasure to be able to participate in person at a meeting and get useful information, not only in terms of the knowledge and intellectual aspects of diabetes, but also something that can be implemented the following day in their daily clinical activities.”
EASD Honorary Secretary Mikael Rydén, MD, added: “I think meeting attendees will really be able to get the absolutely latest developments in all the areas that are relevant to diabetes treatments. It’s the best way to keep yourself up to date.”
This year, in particular, there’s a focus on past, present, and future trends in type 2 diabetes management, along with the co-occurring conditions of obesity, heart failure, and metabolic fatty liver disease.
DELIVER: The diabetes side
On Sept. 22, new data will be presented from the DELIVER trial on the sodium-glucose cotransporter 2 (SGLT2) inhibitor dapagliflozin (Farxiga) in patients with heart failure with preserved ejection fraction, comparing data for participants with diabetes, prediabetes, and normoglycemia.
Primary results from DELIVER were presented Aug. 26 at the European Society of Cardiology Congress 2022 in Barcelona and simultaneously published in the New England Journal of Medicine. The results showed that dapagliflozin benefits patients with heart failure with preserved ejection fraction, as previously demonstrated in those with reduced ejection fraction in the DAPA-HF trial.
“This information is quite important and is becoming of major interest in the field of diabetes,” Dr. Del Prato said, adding that a related joint EASD/ESC symposium will take place the next morning, on Sept. 23, entitled, “New perspectives on heart function and failure in diabetes.”
“So, within the congress, you get the background, pathophysiology, the diagnostic aspects, and the results of the effect of dapagliflozin on those individuals.”
Dr. Rydén commented, “I think this underlines how important it is for diabetologists to screen our patients better for heart failure because we can actually treat them now.”
However, Dr. Rydén of the Karolinska Institute, Stockholm, also cautioned about use of SGLT2 inhibitors in people with diabetes who use insulin, given the risk of euglycemic diabetic ketoacidosis. “These drugs have side effects and you have to be wary who you prescribe them to. For those on multiple daily [insulin] injections, the side effects probably outweigh the benefits.”
Tirzepatide, weight loss, and type 2 diabetes remission
On Sept. 21, a symposium will provide new data for the dual glucagonlike peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) agonist tirzepatide, approved for the treatment of type 2 diabetes in the United States in May with the brand name Mounjaro. The agent is now being studied as an obesity treatment.
Data from the SURMOUNT-1 trial presented at the ADA meeting in June showed the drug produced “unprecedented” weight loss of up to 22.5%.
At EASD, those findings will be reviewed and new data presented on morbidity and mortality, along with a new commentary. The degree of weight loss seen with this new twincretin has furthered discussion about the concept of remission in type 2 diabetes, Dr. Rydén noted. That will also be the subject of the Diabetologia symposium on Sept. 21, entitled, “Remission of type 2 diabetes – fact or fiction?”
Regarding tirzepatide, Dr. Rydén said: “It’s amazing, the most powerful antiobesity drug we have at our disposal. These drugs slow gastric emptying and have other beneficial effects. … We’re now closing in on drugs that produce more than 15% weight loss. That appears to be the ‘magic bullet’ where you can achieve type 2 diabetes remission.” He pointed to a symposium sponsored by The Lancet on this topic at last year’s EASD meeting.
“I think what we want with our drugs is not to treat but actually to combat type 2 diabetes and really to achieve remission. Of course, if you’ve had it for many decades that might be impossible, but we know that particularly in the first 5-10 years it’s very important to have good glucose control and we know we can also achieve remission.”
Dr. Del Prato noted the importance of weight reduction at the time of type 2 diabetes diagnosis will be emphasized in the ADA/EASD consensus document on the management of hyperglycemia in type 2 diabetes, to be presented in its final form on Sept. 23.
“I think we’ll be learning more about potential remission in the future, both because of metabolic surgery and agents like tirzepatide. The reduction in body weight that can be achieved [with these newer drugs], or that has been reported so far, is the closest to what can be obtained with metabolic surgery. I think there will be more and more information and a lot of discussion about this, and of course about the definition of remission and what to do after remission has occurred,” Dr. Del Prato said.
The revised ADA/EASD consensus document is expected to endorse weight loss as a “co-primary goal” of care for those without cardiorenal disease, along with early initiation of combination therapies – for example, taking two drugs immediately upon diagnosis, rather than just metformin – as opposed to the prior stepwise approach. The document will also cover use of newer glucose-lowering therapies, surgery, and behavioral interventions.
The key is a holistic approach, Dr. Del Prato said. “Of course, glucose control is important, but it’s not the only thing. The heterogeneity of the population with diabetes is also important. Some may already have microvascular complications, kidney dysfunction, are more or less obese, and older or younger. We need to keep these differences in mind to provide more and more individualized treatment.”
Related to that, he noted, will be a joint EASD/ADA symposium on Sept. 19, entitled, “Precision medicine in type 2 diabetes: How far can we get?”
COVID-19 and diabetes, UKPDS, type 1 diabetes, and much more
As always, there’s a whole lot more. On Sept. 21, there will be a symposium on COVID-19 and diabetes.
Another, on diabetes technology, has a somewhat cautionary theme: “A new hope (Star Wars) or strange new worlds (Star Trek): Submerging diabetes into emerging technologies.” One of the speakers will address the question: “Are we becoming robots? Automated insulin delivery (AID) systems for everyone with type 1 diabetes: Strengths and limitations.” And this year’s EASD/JDRF symposium topic will be prevention of type 1 diabetes.
Yet another symposium on Sept. 21 will present 44-year follow-up data from the landmark United Kingdom Prospective Diabetes Study (UKPDS), including an economic analysis and a look at dementia outcomes. “It’s a historical thing. This big trial represents a gold mine of information,” Dr. Del Prato commented.
On Sept. 22, new data will be presented for the investigational once-weekly insulins during a symposium entitled, “Re-inventing the insulin experience: Exploring the prospects of once-weekly insulins.”
And lest anyone was thinking of leaving the conference early, there’s a full agenda on Sept. 23, including symposia on diabetic nephropathy, type 1 diabetes, diabetes in old age, dietary management, and the role of primary care, among others. There will also be 12 separate oral presentation sessions that day.
Overall, the meeting will reflect the multidisciplinary direction the field is headed, Dr. Rydén said.
“We’re still in an era of medicine where a lot of things happen every year. Now we have the next generation of drugs that are coming that combine many areas of treatment – obesity, cardiology, and nephrology. So, we’re integrating. The future is integrating the diabetes world with our friends in other areas of clinical medicine.”
Dr. Del Prato has reported being a consultant, advisory board member, and/or lecturer for AstraZeneca, Boehringer Ingelheim, Novo Nordisk, Sanofi, Takeda, Eli Lilly, Abbott, and Applied Therapeutics. Dr. Rydén has reported receiving lecture fees from the Novo Nordisk Foundation and serving on advisory boards for MSD, Lilly, Boehringer Ingelheim, and AstraZeneca.
A version of this article first appeared on Medscape.com.
Highlights of the European Association for the Study of Diabetes 2022 annual meeting include new data on weight loss with the blockbuster twincretin tirzepatide and on the effects of dapagliflozin on heart failure in people with diabetes, as well as updated guidelines for type 2 diabetes management.
The EASD meeting will take place Sept. 19-23 in Stockholm. It will be the first in-person meeting since 2019 but will also feature live-streamed content for participants around the world.
“The EASD congress will cover all the different areas and aspects of diabetes research – clinical, basic, epidemiologic, and psychological,” EASD President Stefano Del Prato, MD, told this news organization.
What attendees should expect, said Del Prato of the University of Pisa (Italy), “is the pleasure to be able to participate in person at a meeting and get useful information, not only in terms of the knowledge and intellectual aspects of diabetes, but also something that can be implemented the following day in their daily clinical activities.”
EASD Honorary Secretary Mikael Rydén, MD, added: “I think meeting attendees will really be able to get the absolutely latest developments in all the areas that are relevant to diabetes treatments. It’s the best way to keep yourself up to date.”
This year, in particular, there’s a focus on past, present, and future trends in type 2 diabetes management, along with the co-occurring conditions of obesity, heart failure, and metabolic fatty liver disease.
DELIVER: The diabetes side
On Sept. 22, new data will be presented from the DELIVER trial on the sodium-glucose cotransporter 2 (SGLT2) inhibitor dapagliflozin (Farxiga) in patients with heart failure with preserved ejection fraction, comparing data for participants with diabetes, prediabetes, and normoglycemia.
Primary results from DELIVER were presented Aug. 26 at the European Society of Cardiology Congress 2022 in Barcelona and simultaneously published in the New England Journal of Medicine. The results showed that dapagliflozin benefits patients with heart failure with preserved ejection fraction, as previously demonstrated in those with reduced ejection fraction in the DAPA-HF trial.
“This information is quite important and is becoming of major interest in the field of diabetes,” Dr. Del Prato said, adding that a related joint EASD/ESC symposium will take place the next morning, on Sept. 23, entitled, “New perspectives on heart function and failure in diabetes.”
“So, within the congress, you get the background, pathophysiology, the diagnostic aspects, and the results of the effect of dapagliflozin on those individuals.”
Dr. Rydén commented, “I think this underlines how important it is for diabetologists to screen our patients better for heart failure because we can actually treat them now.”
However, Dr. Rydén of the Karolinska Institute, Stockholm, also cautioned about use of SGLT2 inhibitors in people with diabetes who use insulin, given the risk of euglycemic diabetic ketoacidosis. “These drugs have side effects and you have to be wary who you prescribe them to. For those on multiple daily [insulin] injections, the side effects probably outweigh the benefits.”
Tirzepatide, weight loss, and type 2 diabetes remission
On Sept. 21, a symposium will provide new data for the dual glucagonlike peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) agonist tirzepatide, approved for the treatment of type 2 diabetes in the United States in May with the brand name Mounjaro. The agent is now being studied as an obesity treatment.
Data from the SURMOUNT-1 trial presented at the ADA meeting in June showed the drug produced “unprecedented” weight loss of up to 22.5%.
At EASD, those findings will be reviewed and new data presented on morbidity and mortality, along with a new commentary. The degree of weight loss seen with this new twincretin has furthered discussion about the concept of remission in type 2 diabetes, Dr. Rydén noted. That will also be the subject of the Diabetologia symposium on Sept. 21, entitled, “Remission of type 2 diabetes – fact or fiction?”
Regarding tirzepatide, Dr. Rydén said: “It’s amazing, the most powerful antiobesity drug we have at our disposal. These drugs slow gastric emptying and have other beneficial effects. … We’re now closing in on drugs that produce more than 15% weight loss. That appears to be the ‘magic bullet’ where you can achieve type 2 diabetes remission.” He pointed to a symposium sponsored by The Lancet on this topic at last year’s EASD meeting.
“I think what we want with our drugs is not to treat but actually to combat type 2 diabetes and really to achieve remission. Of course, if you’ve had it for many decades that might be impossible, but we know that particularly in the first 5-10 years it’s very important to have good glucose control and we know we can also achieve remission.”
Dr. Del Prato noted the importance of weight reduction at the time of type 2 diabetes diagnosis will be emphasized in the ADA/EASD consensus document on the management of hyperglycemia in type 2 diabetes, to be presented in its final form on Sept. 23.
“I think we’ll be learning more about potential remission in the future, both because of metabolic surgery and agents like tirzepatide. The reduction in body weight that can be achieved [with these newer drugs], or that has been reported so far, is the closest to what can be obtained with metabolic surgery. I think there will be more and more information and a lot of discussion about this, and of course about the definition of remission and what to do after remission has occurred,” Dr. Del Prato said.
The revised ADA/EASD consensus document is expected to endorse weight loss as a “co-primary goal” of care for those without cardiorenal disease, along with early initiation of combination therapies – for example, taking two drugs immediately upon diagnosis, rather than just metformin – as opposed to the prior stepwise approach. The document will also cover use of newer glucose-lowering therapies, surgery, and behavioral interventions.
The key is a holistic approach, Dr. Del Prato said. “Of course, glucose control is important, but it’s not the only thing. The heterogeneity of the population with diabetes is also important. Some may already have microvascular complications, kidney dysfunction, are more or less obese, and older or younger. We need to keep these differences in mind to provide more and more individualized treatment.”
Related to that, he noted, will be a joint EASD/ADA symposium on Sept. 19, entitled, “Precision medicine in type 2 diabetes: How far can we get?”
COVID-19 and diabetes, UKPDS, type 1 diabetes, and much more
As always, there’s a whole lot more. On Sept. 21, there will be a symposium on COVID-19 and diabetes.
Another, on diabetes technology, has a somewhat cautionary theme: “A new hope (Star Wars) or strange new worlds (Star Trek): Submerging diabetes into emerging technologies.” One of the speakers will address the question: “Are we becoming robots? Automated insulin delivery (AID) systems for everyone with type 1 diabetes: Strengths and limitations.” And this year’s EASD/JDRF symposium topic will be prevention of type 1 diabetes.
Yet another symposium on Sept. 21 will present 44-year follow-up data from the landmark United Kingdom Prospective Diabetes Study (UKPDS), including an economic analysis and a look at dementia outcomes. “It’s a historical thing. This big trial represents a gold mine of information,” Dr. Del Prato commented.
On Sept. 22, new data will be presented for the investigational once-weekly insulins during a symposium entitled, “Re-inventing the insulin experience: Exploring the prospects of once-weekly insulins.”
And lest anyone was thinking of leaving the conference early, there’s a full agenda on Sept. 23, including symposia on diabetic nephropathy, type 1 diabetes, diabetes in old age, dietary management, and the role of primary care, among others. There will also be 12 separate oral presentation sessions that day.
Overall, the meeting will reflect the multidisciplinary direction the field is headed, Dr. Rydén said.
“We’re still in an era of medicine where a lot of things happen every year. Now we have the next generation of drugs that are coming that combine many areas of treatment – obesity, cardiology, and nephrology. So, we’re integrating. The future is integrating the diabetes world with our friends in other areas of clinical medicine.”
Dr. Del Prato has reported being a consultant, advisory board member, and/or lecturer for AstraZeneca, Boehringer Ingelheim, Novo Nordisk, Sanofi, Takeda, Eli Lilly, Abbott, and Applied Therapeutics. Dr. Rydén has reported receiving lecture fees from the Novo Nordisk Foundation and serving on advisory boards for MSD, Lilly, Boehringer Ingelheim, and AstraZeneca.
A version of this article first appeared on Medscape.com.
Highlights of the European Association for the Study of Diabetes 2022 annual meeting include new data on weight loss with the blockbuster twincretin tirzepatide and on the effects of dapagliflozin on heart failure in people with diabetes, as well as updated guidelines for type 2 diabetes management.
The EASD meeting will take place Sept. 19-23 in Stockholm. It will be the first in-person meeting since 2019 but will also feature live-streamed content for participants around the world.
“The EASD congress will cover all the different areas and aspects of diabetes research – clinical, basic, epidemiologic, and psychological,” EASD President Stefano Del Prato, MD, told this news organization.
What attendees should expect, said Del Prato of the University of Pisa (Italy), “is the pleasure to be able to participate in person at a meeting and get useful information, not only in terms of the knowledge and intellectual aspects of diabetes, but also something that can be implemented the following day in their daily clinical activities.”
EASD Honorary Secretary Mikael Rydén, MD, added: “I think meeting attendees will really be able to get the absolutely latest developments in all the areas that are relevant to diabetes treatments. It’s the best way to keep yourself up to date.”
This year, in particular, there’s a focus on past, present, and future trends in type 2 diabetes management, along with the co-occurring conditions of obesity, heart failure, and metabolic fatty liver disease.
DELIVER: The diabetes side
On Sept. 22, new data will be presented from the DELIVER trial on the sodium-glucose cotransporter 2 (SGLT2) inhibitor dapagliflozin (Farxiga) in patients with heart failure with preserved ejection fraction, comparing data for participants with diabetes, prediabetes, and normoglycemia.
Primary results from DELIVER were presented Aug. 26 at the European Society of Cardiology Congress 2022 in Barcelona and simultaneously published in the New England Journal of Medicine. The results showed that dapagliflozin benefits patients with heart failure with preserved ejection fraction, as previously demonstrated in those with reduced ejection fraction in the DAPA-HF trial.
“This information is quite important and is becoming of major interest in the field of diabetes,” Dr. Del Prato said, adding that a related joint EASD/ESC symposium will take place the next morning, on Sept. 23, entitled, “New perspectives on heart function and failure in diabetes.”
“So, within the congress, you get the background, pathophysiology, the diagnostic aspects, and the results of the effect of dapagliflozin on those individuals.”
Dr. Rydén commented, “I think this underlines how important it is for diabetologists to screen our patients better for heart failure because we can actually treat them now.”
However, Dr. Rydén of the Karolinska Institute, Stockholm, also cautioned about use of SGLT2 inhibitors in people with diabetes who use insulin, given the risk of euglycemic diabetic ketoacidosis. “These drugs have side effects and you have to be wary who you prescribe them to. For those on multiple daily [insulin] injections, the side effects probably outweigh the benefits.”
Tirzepatide, weight loss, and type 2 diabetes remission
On Sept. 21, a symposium will provide new data for the dual glucagonlike peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) agonist tirzepatide, approved for the treatment of type 2 diabetes in the United States in May with the brand name Mounjaro. The agent is now being studied as an obesity treatment.
Data from the SURMOUNT-1 trial presented at the ADA meeting in June showed the drug produced “unprecedented” weight loss of up to 22.5%.
At EASD, those findings will be reviewed and new data presented on morbidity and mortality, along with a new commentary. The degree of weight loss seen with this new twincretin has furthered discussion about the concept of remission in type 2 diabetes, Dr. Rydén noted. That will also be the subject of the Diabetologia symposium on Sept. 21, entitled, “Remission of type 2 diabetes – fact or fiction?”
Regarding tirzepatide, Dr. Rydén said: “It’s amazing, the most powerful antiobesity drug we have at our disposal. These drugs slow gastric emptying and have other beneficial effects. … We’re now closing in on drugs that produce more than 15% weight loss. That appears to be the ‘magic bullet’ where you can achieve type 2 diabetes remission.” He pointed to a symposium sponsored by The Lancet on this topic at last year’s EASD meeting.
“I think what we want with our drugs is not to treat but actually to combat type 2 diabetes and really to achieve remission. Of course, if you’ve had it for many decades that might be impossible, but we know that particularly in the first 5-10 years it’s very important to have good glucose control and we know we can also achieve remission.”
Dr. Del Prato noted the importance of weight reduction at the time of type 2 diabetes diagnosis will be emphasized in the ADA/EASD consensus document on the management of hyperglycemia in type 2 diabetes, to be presented in its final form on Sept. 23.
“I think we’ll be learning more about potential remission in the future, both because of metabolic surgery and agents like tirzepatide. The reduction in body weight that can be achieved [with these newer drugs], or that has been reported so far, is the closest to what can be obtained with metabolic surgery. I think there will be more and more information and a lot of discussion about this, and of course about the definition of remission and what to do after remission has occurred,” Dr. Del Prato said.
The revised ADA/EASD consensus document is expected to endorse weight loss as a “co-primary goal” of care for those without cardiorenal disease, along with early initiation of combination therapies – for example, taking two drugs immediately upon diagnosis, rather than just metformin – as opposed to the prior stepwise approach. The document will also cover use of newer glucose-lowering therapies, surgery, and behavioral interventions.
The key is a holistic approach, Dr. Del Prato said. “Of course, glucose control is important, but it’s not the only thing. The heterogeneity of the population with diabetes is also important. Some may already have microvascular complications, kidney dysfunction, are more or less obese, and older or younger. We need to keep these differences in mind to provide more and more individualized treatment.”
Related to that, he noted, will be a joint EASD/ADA symposium on Sept. 19, entitled, “Precision medicine in type 2 diabetes: How far can we get?”
COVID-19 and diabetes, UKPDS, type 1 diabetes, and much more
As always, there’s a whole lot more. On Sept. 21, there will be a symposium on COVID-19 and diabetes.
Another, on diabetes technology, has a somewhat cautionary theme: “A new hope (Star Wars) or strange new worlds (Star Trek): Submerging diabetes into emerging technologies.” One of the speakers will address the question: “Are we becoming robots? Automated insulin delivery (AID) systems for everyone with type 1 diabetes: Strengths and limitations.” And this year’s EASD/JDRF symposium topic will be prevention of type 1 diabetes.
Yet another symposium on Sept. 21 will present 44-year follow-up data from the landmark United Kingdom Prospective Diabetes Study (UKPDS), including an economic analysis and a look at dementia outcomes. “It’s a historical thing. This big trial represents a gold mine of information,” Dr. Del Prato commented.
On Sept. 22, new data will be presented for the investigational once-weekly insulins during a symposium entitled, “Re-inventing the insulin experience: Exploring the prospects of once-weekly insulins.”
And lest anyone was thinking of leaving the conference early, there’s a full agenda on Sept. 23, including symposia on diabetic nephropathy, type 1 diabetes, diabetes in old age, dietary management, and the role of primary care, among others. There will also be 12 separate oral presentation sessions that day.
Overall, the meeting will reflect the multidisciplinary direction the field is headed, Dr. Rydén said.
“We’re still in an era of medicine where a lot of things happen every year. Now we have the next generation of drugs that are coming that combine many areas of treatment – obesity, cardiology, and nephrology. So, we’re integrating. The future is integrating the diabetes world with our friends in other areas of clinical medicine.”
Dr. Del Prato has reported being a consultant, advisory board member, and/or lecturer for AstraZeneca, Boehringer Ingelheim, Novo Nordisk, Sanofi, Takeda, Eli Lilly, Abbott, and Applied Therapeutics. Dr. Rydén has reported receiving lecture fees from the Novo Nordisk Foundation and serving on advisory boards for MSD, Lilly, Boehringer Ingelheim, and AstraZeneca.
A version of this article first appeared on Medscape.com.
Diabetes Population Health Innovations in the Age of COVID-19: Insights From the T1D Exchange Quality Improvement Collaborative
From the T1D Exchange, Boston, MA (Ann Mungmode, Nicole Rioles, Jesse Cases, Dr. Ebekozien); The Leona M. and Harry B. Hemsley Charitable Trust, New York, NY (Laurel Koester); and the University of Mississippi School of Population Health, Jackson, MS (Dr. Ebekozien).
Abstract
There have been remarkable innovations in diabetes management since the start of the COVID-19 pandemic, but these groundbreaking innovations are drawing limited focus as the field focuses on the adverse impact of the pandemic on patients with diabetes. This article reviews select population health innovations in diabetes management that have become available over the past 2 years of the COVID-19 pandemic from the perspective of the T1D Exchange Quality Improvement Collaborative, a learning health network that focuses on improving care and outcomes for individuals with type 1 diabetes (T1D). Such innovations include expanded telemedicine access, collection of real-world data, machine learning and artificial intelligence, and new diabetes medications and devices. In addition, multiple innovative studies have been undertaken to explore contributors to health inequities in diabetes, and advocacy efforts for specific populations have been successful. Looking to the future, work is required to explore additional health equity successes that do not further exacerbate inequities and to look for additional innovative ways to engage people with T1D in their health care through conversations on social determinants of health and societal structures.
Keywords: type 1 diabetes, learning health network, continuous glucose monitoring, health equity
One in 10 people in the United States has diabetes.1 Diabetes is the nation’s second leading cause of death, costing the US health system more than $300 billion annually.2 The COVID-19 pandemic presented additional health burdens for people living with diabetes. For example, preexisting diabetes was identified as a risk factor for COVID-19–associated morbidity and mortality.3,4 Over the past 2 years, there have been remarkable innovations in diabetes management, including stem cell therapy and new medication options. Additionally, improved technology solutions have aided in diabetes management through continuous glucose monitors (CGM), smart insulin pens, advanced hybrid closed-loop systems, and continuous subcutaneous insulin injections.5,6 Unfortunately, these groundbreaking innovations are drawing limited focus, as the field is rightfully focused on the adverse impact of the pandemic on patients with diabetes.
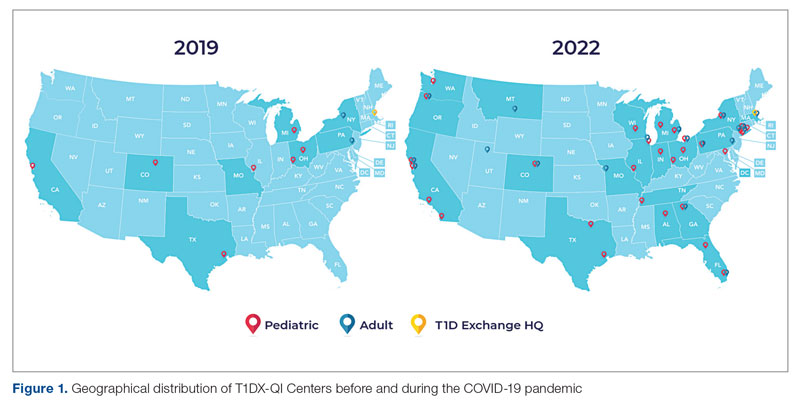
Learning health networks like the T1D Exchange Quality Improvement Collaborative (T1DX-QI) have implemented some of these innovative solutions to improve care for people with diabetes.7 T1DX-QI has more than 50 data-sharing endocrinology centers that care for over 75,000 people with diabetes across the United States (Figure 1). Centers participating in the T1DX-QI use quality improvement (QI) and implementation science methods to quickly translate research into evidence-based clinical practice. T1DX-QI leads diabetes population health and health system research and supports widespread transferability across health care organizations through regular collaborative calls, conferences, and case study documentation.8
In this review, we summarize impactful population health innovations in diabetes management that have become available over the past 2 years of the COVID-19 pandemic from the perspective of T1DX-QI (see Figure 2 for relevant definitions). This review is limited in scope and is not meant to be an exhaustive list of innovations. The review also reflects significant changes from the perspective of academic diabetes centers, which may not apply to rural or primary care diabetes practices.
Methods
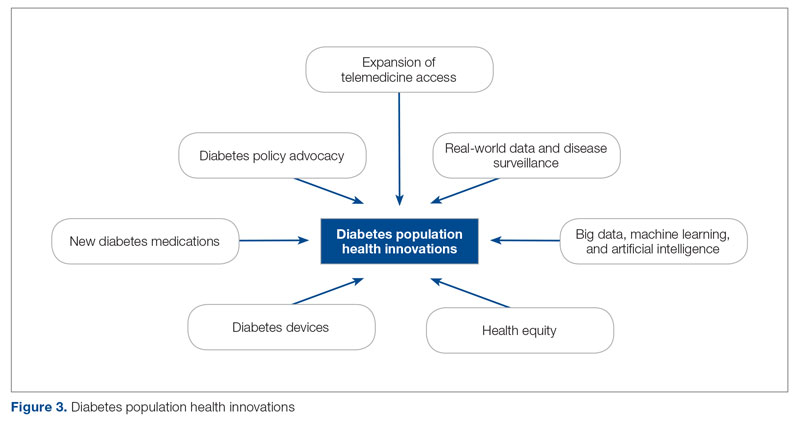
The first (A.M.), second (H.H.), and senior (O.E.) authors conducted a scoping review of published literature using terms related to diabetes, population health, and innovation on PubMed Central and Google Scholar for the period March 2020 to June 2022. To complement the review, A.M. and O.E. also reviewed abstracts from presentations at major international diabetes conferences, including the American Diabetes Association (ADA), the International Society for Pediatric and Adolescent Diabetes (ISPAD), the T1DX-QI Learning Session Conference, and the Advanced Technologies & Treatments for Diabetes (ATTD) 2020 to 2022 conferences.9-14 The authors also searched FDA.gov and ClinicalTrials.gov for relevant insights. A.M. and O.E. sorted the reviewed literature into major themes (Figure 3) from the population health improvement perspective of the T1DX-QI.
Population Health Innovations in Diabetes Management
Expansion of Telemedicine Access
Telemedicine is cost-effective for patients with diabetes,15 including those with complex cases.16 Before the COVID-19 pandemic, telemedicine and virtual care were rare in diabetes management. However, the pandemic offered a new opportunity to expand the practice of telemedicine in diabetes management. A study from the T1DX-QI showed that telemedicine visits grew from comprising <1% of visits pre-pandemic (December 2019) to 95.2% during the pandemic (August 2020).17 Additional studies, like those conducted by Phillip et al,18 confirmed the noninferiority of telemedicine practice for patients with diabetes.Telemedicine was also found to be an effective strategy to educate patients on the use of diabetes technologies.19
Real-World Data and Disease Surveillance
As the COVID-19 pandemic exacerbated outcomes for people with type 1 diabetes (T1D), a need arose to understand the immediate effects of the pandemic on people with T1D through real-world data and disease surveillance. In April 2020, the T1DX-QI initiated a multicenter surveillance study to collect data and analyze the impact of COVID-19 on people with T1D. The existing health collaborative served as a springboard for robust surveillance study, documenting numerous works on the effects of COVID-19.3,4,20-28 Other investigators also embraced the power of real-world surveillance and real-world data.29,30
Big Data, Machine Learning, and Artificial Intelligence
The past 2 years have seen a shift toward embracing the incredible opportunity to tap the large volume of data generated from routine care for practical insights.31 In particular, researchers have demonstrated the widespread application of machine learning and artificial intelligence to improve diabetes management.32 The T1DX-QI also harnessed the growing power of big data by expanding the functionality of innovative benchmarking software. The T1DX QI Portal uses electronic medical record data of diabetes patients for clinic-to-clinic benchmarking and data analysis, using business intelligence solutions.33
Health Equity
While inequities across various health outcomes have been well documented for years,34 the COVID-19 pandemic further exaggerated racial/ethnic health inequities in T1D.23,35 In response, several organizations have outlined specific strategies to address these health inequities. Emboldened by the pandemic, the T1DX-QI announced a multipronged approach to address health inequities among patients with T1D through the Health Equity Advancement Lab (HEAL).36 One of HEAL’s main components is using real-world data to champion population-level insights and demonstrate progress in QI efforts.
Multiple innovative studies have been undertaken to explore contributors to health inequities in diabetes, and these studies are expanding our understanding of the chasm.37 There have also been innovative solutions to addressing these inequities, with multiple studies published over the past 2 years.38 A source of inequity among patients with T1D is the lack of representation of racial/ethnic minorities with T1D in clinical trials.39 The T1DX-QI suggests that the equity-adapted framework for QI can be applied by research leaders to support trial diversity and representation, ensuring future device innovations are meaningful for all people with T1D.40
Diabetes Devices
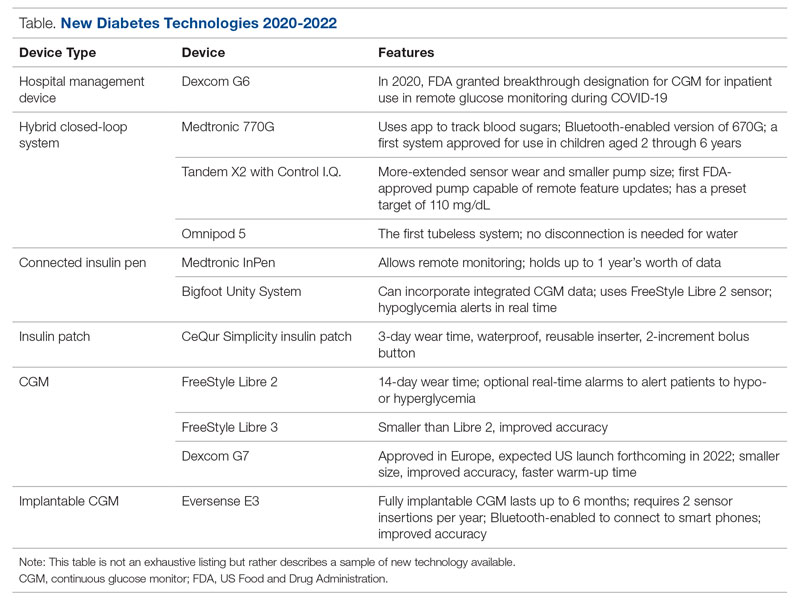
Glucose monitoring and insulin therapy are vital tools to support individuals living with T1D, and devices such as CGM and insulin pumps have become the standard of care for diabetes management (Table).41 Innovations in diabetes technology and device access are imperative for a chronic disease with no cure.
The COVID-19 pandemic created an opportunity to increase access to diabetes devices in inpatient settings. In 2020, the US Food and Drug Administration expanded the use of CGM to support remote monitoring of patients in inpatient hospital settings, simultaneously supporting the glucose monitoring needs of patients with T1D and reducing COVID-19 transmission through reduced patient-clinician contact.42 This effort has been expanded and will continue in 2022 and beyond,43 and aligns with the growing consensus that supports patients wearing both CGMs and insulin pumps in ambulatory settings to improve patient health outcomes.44
Since 2020, innovations in diabetes technology have improved and increased the variety of options available to people with T1D and made them easier to use (Table). New, advanced hybrid closed-loop systems have progressed to offer Bluetooth features, including automatic software upgrades, tubeless systems, and the ability to allow parents to use their smartphones to bolus for children.45-47 The next big step in insulin delivery innovation is the release of functioning, fully closed loop systems, of which several are currently in clinical trials.48 These systems support reduced hypoglycemia and improved time in range.49
Additional innovations in insulin delivery have improved the user experience and expanded therapeutic options, including a variety of smart insulin pens complete with dosing logs50,51 and even a patch to deliver insulin without the burden of injections.52 As barriers to diabetes technology persist,53 innovations in alternate insulin delivery provide people with T1D more options to align with their personal access and technology preferences.
Innovations in CGM address cited barriers to their use, including size or overall wear.53-55 CGMs released in the past few years are smaller in physical size, have longer durations of time between changings, are more accurate, and do not require calibrations for accuracy.
New Diabetes Medications
Many new medications and therapeutic advances have become available in the past 2 years.56 Additionally, more medications are being tested as adjunct therapies to support glycemic management in patients with T1D, including metformin, sodium-glucose cotransporter 1 and 2 inhibitors, pramlintide, glucagon-like polypeptide-1 analogs, and glucagon receptor agonists.57 Other recent advances include stem cell replacement therapy for patients with T1D.58 The ultra-long-acting biosimilar insulins are one medical innovation that has been stalled, rather than propelled, during the COVID-19 pandemic.59
Diabetes Policy Advocacy
People with T1D require insulin to survive. The cost of insulin has increased in recent years, with some studies citing a 64% to 100% increase in the past decade.60,61 In fact, 1 in 4 insulin users report that cost has impacted their insulin use, including rationing their insulin.62 Lockdowns during the COVID-19 pandemic stressed US families financially, increasing the urgency for insulin cost caps.
Although the COVID-19 pandemic halted national conversations on drug financing,63 advocacy efforts have succeeded for specific populations. The new Medicare Part D Senior Savings Model will cap the cost of insulin at $35 for a 30-day supply,64 and 20 states passed legislation capping insulin pricing.62 Efforts to codify national cost caps are under debate, including the passage of the Affordable Insulin Now Act, which passed the House in March 2022 and is currently under review in the Senate.65
Perspective: The Role of Private Philanthropy in Supporting Population Health Innovations
Funders and industry partners play a crucial role in leading and supporting innovations that improve the lives of people with T1D and reduce society’s costs of living with the disease. Data infrastructure is critical to supporting population health. While building the data infrastructure to support population health is both time- and resource-intensive, private foundations such as Helmsley are uniquely positioned—and have a responsibility—to take large, informed risks to help reach all communities with T1D.
The T1DX-QI is the largest source of population health data on T1D in the United States and is becoming the premiere data authority on its incidence, prevalence, and outcomes. The T1DX-QI enables a robust understanding of T1D-related health trends at the population level, as well as trends among clinics and providers. Pilot centers in the T1DX-QI have reported reductions in patients’ A1c and acute diabetes-related events, as well as improvements in device usage and depression screening. The ability to capture changes speaks to the promise and power of these data to demonstrate the clinical impact of QI interventions and to support the spread of best practices and learnings across health systems.
Additional philanthropic efforts have supported innovation in the last 2 years. For example, the JDRF, a nonprofit philanthropic equity firm, has supported efforts in developing artificial pancreas systems and cell therapies currently in clinical trials like teplizumab, a drug that has demonstrated delayed onset of T1D through JDRF’s T1D Fund.66 Industry partners also have an opportunity for significant influence in this area, as they continue to fund meaningful projects to advance care for people with T1D.67
Conclusion
We are optimistic that the innovations summarized here describe a shift in the tide of equitable T1D outcomes; however, future work is required to explore additional health equity successes that do not further exacerbate inequities. We also see further opportunities for innovative ways to engage people with T1D in their health care through conversations on social determinants of health and societal structures.
Corresponding author: Ann Mungmode, MPH, T1D Exchange, 11 Avenue de Lafayette, Boston, MA 02111; Email: amungmode@t1dexchange.org
Disclosures: Dr. Ebekozien serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for the Medtronic Advisory Board and received research grants from Medtronic Diabetes, Eli Lilly, and Dexcom.
Funding: The T1DX-QI is funded by The Leona M. and Harry B. Hemsley Charitable Trust.
1. Centers for Disease Control and Prevention. National diabetes statistics report. Accessed August 30, 2022. www.cdc.gov/diabetes/data/statistics-report/index.html
2. Centers for Disease Control and Prevention. Diabetes fast facts. Accessed August 30, 2022. www.cdc.gov/diabetes/basics/quick-facts.html
3. O’Malley G, Ebekozien O, Desimone M, et al. COVID-19 hospitalization in adults with type 1 diabetes: results from the T1D Exchange Multicenter Surveillance Study. J Clin Endocrinol Metab. 2020;106(2):e936-e942. doi:10.1210/clinem/dgaa825
4. Ebekozien OA, Noor N, Gallagher MP, Alonso GT. Type 1 diabetes and COVID-19: preliminary findings from a multicenter surveillance study in the U.S. Diabetes Care. 2020;43(8):e83-e85. doi:10.2337/dc20-1088
5. Zimmerman C, Albanese-O’Neill A, Haller MJ. Advances in type 1 diabetes technology over the last decade. Eur Endocrinol. 2019;15(2):70-76. doi:10.17925/ee.2019.15.2.70
6. Wake DJ, Gibb FW, Kar P, et al. Endocrinology in the time of COVID-19: remodelling diabetes services and emerging innovation. Eur J Endocrinol. 2020;183(2):G67-G77. doi:10.1530/eje-20-0377
7. Alonso GT, Corathers S, Shah A, et al. Establishment of the T1D Exchange Quality Improvement Collaborative (T1DX-QI). Clin Diabetes. 2020;38(2):141-151. doi:10.2337/cd19-0032
8. Ginnard OZB, Alonso GT, Corathers SD, et al. Quality improvement in diabetes care: a review of initiatives and outcomes in the T1D Exchange Quality Improvement Collaborative. Clin Diabetes. 2021;39(3):256-263. doi:10.2337/cd21-0029
9. ATTD 2021 invited speaker abstracts. Diabetes Technol Ther. 2021;23(S2):A1-A206. doi:10.1089/dia.2021.2525.abstracts
10. Rompicherla SN, Edelen N, Gallagher R, et al. Children and adolescent patients with pre-existing type 1 diabetes and additional comorbidities have an increased risk of hospitalization from COVID-19; data from the T1D Exchange COVID Registry. Pediatr Diabetes. 2021;22(S30):3-32. doi:10.1111/pedi.13268
11. Abstracts for the T1D Exchange QI Collaborative (T1DX-QI) Learning Session 2021. November 8-9, 2021. J Diabetes. 2021;13(S1):3-17. doi:10.1111/1753-0407.13227
12. The Official Journal of ATTD Advanced Technologies & Treatments for Diabetes conference 27-30 April 2022. Barcelona and online. Diabetes Technol Ther. 2022;24(S1):A1-A237. doi:10.1089/dia.2022.2525.abstracts
13. Ebekozien ON, Kamboj N, Odugbesan MK, et al. Inequities in glycemic outcomes for patients with type 1 diabetes: six-year (2016-2021) longitudinal follow-up by race and ethnicity of 36,390 patients in the T1DX-QI Collaborative. Diabetes. 2022;71(suppl 1). doi:10.2337/db22-167-OR
14. Narayan KA, Noor M, Rompicherla N, et al. No BMI increase during the COVID-pandemic in children and adults with T1D in three continents: joint analysis of ADDN, T1DX, and DPV registries. Diabetes. 2022;71(suppl 1). doi:10.2337/db22-269-OR
15. Lee JY, Lee SWH. Telemedicine cost-effectiveness for diabetes management: a systematic review. Diabetes Technol Ther. 2018;20(7):492-500. doi:10.1089/dia.2018.0098
16. McDonnell ME. Telemedicine in complex diabetes management. Curr Diab Rep. 2018;18(7):42. doi:10.1007/s11892-018-1015-3
17. Lee JM, Carlson E, Albanese-O’Neill A, et al. Adoption of telemedicine for type 1 diabetes care during the COVID-19 pandemic. Diabetes Technol Ther. 2021;23(9):642-651. doi:10.1089/dia.2021.0080
18. Phillip M, Bergenstal RM, Close KL, et al. The digital/virtual diabetes clinic: the future is now–recommendations from an international panel on diabetes digital technologies introduction. Diabetes Technol Ther. 2021;23(2):146-154. doi:10.1089/dia.2020.0375
19. Garg SK, Rodriguez E. COVID‐19 pandemic and diabetes care. Diabetes Technol Ther. 2022;24(S1):S2-S20. doi:10.1089/dia.2022.2501
20. Beliard K, Ebekozien O, Demeterco-Berggren C, et al. Increased DKA at presentation among newly diagnosed type 1 diabetes patients with or without COVID-19: data from a multi-site surveillance registry. J Diabetes. 2021;13(3):270-272. doi:10.1111/1753-0407.13141
21. Ebekozien O, Agarwal S, Noor N, et al. Inequities in diabetic ketoacidosis among patients with type 1 diabetes and COVID-19: data from 52 US clinical centers. J Clin Endocrinol Metab. 2020;106(4):1755-1762. doi:10.1210/clinem/dgaa920
22. Alonso GT, Ebekozien O, Gallagher MP, et al. Diabetic ketoacidosis drives COVID-19 related hospitalizations in children with type 1 diabetes. J Diabetes. 2021;13(8):681-687. doi:10.1111/1753-0407.13184
23. Noor N, Ebekozien O, Levin L, et al. Diabetes technology use for management of type 1 diabetes is associated with fewer adverse COVID-19 outcomes: findings from the T1D Exchange COVID-19 Surveillance Registry. Diabetes Care. 2021;44(8):e160-e162. doi:10.2337/dc21-0074
24. Demeterco-Berggren C, Ebekozien O, Rompicherla S, et al. Age and hospitalization risk in people with type 1 diabetes and COVID-19: data from the T1D Exchange Surveillance Study. J Clin Endocrinol Metab. 2021;107(2):410-418. doi:10.1210/clinem/dgab668
25. DeSalvo DJ, Noor N, Xie C, et al. Patient demographics and clinical outcomes among type 1 diabetes patients using continuous glucose monitors: data from T1D Exchange real-world observational study. J Diabetes Sci Technol. 2021 Oct 9. [Epub ahead of print] doi:10.1177/19322968211049783
26. Gallagher MP, Rompicherla S, Ebekozien O, et al. Differences in COVID-19 outcomes among patients with type 1 diabetes: first vs later surges. J Clin Outcomes Manage. 2022;29(1):27-31. doi:10.12788/jcom.0084
27. Wolf RM, Noor N, Izquierdo R, et al. Increase in newly diagnosed type 1 diabetes in youth during the COVID-19 pandemic in the United States: a multi-center analysis. Pediatr Diabetes. 2022;23(4):433-438. doi:10.1111/pedi.13328
28. Lavik AR, Ebekozien O, Noor N, et al. Trends in type 1 diabetic ketoacidosis during COVID-19 surges at 7 US centers: highest burden on non-Hispanic Black patients. J Clin Endocrinol Metab. 2022;107(7):1948-1955. doi:10.1210/clinem/dgac158
29. van der Linden J, Welsh JB, Hirsch IB, Garg SK. Real-time continuous glucose monitoring during the coronavirus disease 2019 pandemic and its impact on time in range. Diabetes Technol Ther. 2021;23(S1):S1-S7. doi:10.1089/dia.2020.0649
30. Nwosu BU, Al-Halbouni L, Parajuli S, et al. COVID-19 pandemic and pediatric type 1 diabetes: no significant change in glycemic control during the pandemic lockdown of 2020. Front Endocrinol (Lausanne). 2021;12:703905. doi:10.3389/fendo.2021.703905
31. Ellahham S. Artificial intelligence: the future for diabetes care. Am J Med. 2020;133(8):895-900. doi:10.1016/j.amjmed.2020.03.033
32. Nomura A, Noguchi M, Kometani M, et al. Artificial intelligence in current diabetes management and prediction. Curr Diab Rep. 2021;21(12):61. doi:10.1007/s11892-021-01423-2
33. Mungmode A, Noor N, Weinstock RS, et al. Making diabetes electronic medical record data actionable: promoting benchmarking and population health using the T1D Exchange Quality Improvement Portal. Clin Diabetes. Forthcoming 2022.
34. Lavizzo-Mourey RJ, Besser RE, Williams DR. Understanding and mitigating health inequities—past, current, and future directions. N Engl J Med. 2021;384(18):1681-1684. doi:10.1056/NEJMp2008628
35. Majidi S, Ebekozien O, Noor N, et al. Inequities in health outcomes in children and adults with type 1 diabetes: data from the T1D Exchange Quality Improvement Collaborative. Clin Diabetes. 2021;39(3):278-283. doi:10.2337/cd21-0028
36. Ebekozien O, Mungmode A, Odugbesan O, et al. Addressing type 1 diabetes health inequities in the United States: approaches from the T1D Exchange QI Collaborative. J Diabetes. 2022;14(1):79-82. doi:10.1111/1753-0407.13235
37. Odugbesan O, Addala A, Nelson G, et al. Implicit racial-ethnic and insurance-mediated bias to recommending diabetes technology: insights from T1D Exchange multicenter pediatric and adult diabetes provider cohort. Diabetes Technol Ther. 2022 Jun 13. [Epub ahead of print] doi:10.1089/dia.2022.0042
38. Schmitt J, Fogle K, Scott ML, Iyer P. Improving equitable access to continuous glucose monitors for Alabama’s children with type 1 diabetes: a quality improvement project. Diabetes Technol Ther. 2022;24(7):481-491. doi:10.1089/dia.2021.0511
39. Akturk HK, Agarwal S, Hoffecker L, Shah VN. Inequity in racial-ethnic representation in randomized controlled trials of diabetes technologies in type 1 diabetes: critical need for new standards. Diabetes Care. 2021;44(6):e121-e123. doi:10.2337/dc20-3063
40. Ebekozien O, Mungmode A, Buckingham D, et al. Achieving equity in diabetes research: borrowing from the field of quality improvement using a practical framework and improvement tools. Diabetes Spectr. 2022;35(3):304-312. doi:10.2237/dsi22-0002
41. Zhang J, Xu J, Lim J, et al. Wearable glucose monitoring and implantable drug delivery systems for diabetes management. Adv Healthc Mater. 2021;10(17):e2100194. doi:10.1002/adhm.202100194
42. FDA expands remote patient monitoring in hospitals for people with diabetes during COVID-19; manufacturers donate CGM supplies. News release. April 21, 2020. Accessed August 30, 2022. https://www.diabetes.org/newsroom/press-releases/2020/fda-remote-patient-monitoring-cgm
43. Campbell P. FDA grants Dexcom CGM breakthrough designation for in-hospital use. March 2, 2022. Accessed August 30, 2022. https://www.endocrinologynetwork.com/view/fda-grants-dexcom-cgm-breakthrough-designation-for-in-hospital-use
44. Yeh T, Yeung M, Mendelsohn Curanaj FA. Managing patients with insulin pumps and continuous glucose monitors in the hospital: to wear or not to wear. Curr Diab Rep. 2021;21(2):7. doi:10.1007/s11892-021-01375-7
45. Medtronic announces FDA approval for MiniMed 770G insulin pump system. News release. September 21, 2020. Accessed August 30, 2022. https://bit.ly/3TyEna4
46. Tandem Diabetes Care announces commercial launch of the t:slim X2 insulin pump with Control-IQ technology in the United States. News release. January 15, 2020. Accessed August 30, 2022. https://investor.tandemdiabetes.com/news-releases/news-release-details/tandem-diabetes-care-announces-commercial-launch-tslim-x2-0
47. Garza M, Gutow H, Mahoney K. Omnipod 5 cleared by the FDA. Updated August 22, 2022. Accessed August 30, 2022.https://diatribe.org/omnipod-5-approved-fda
48. Boughton CK. Fully closed-loop insulin delivery—are we nearly there yet? Lancet Digit Health. 2021;3(11):e689-e690. doi:10.1016/s2589-7500(21)00218-1
49. Noor N, Kamboj MK, Triolo T, et al. Hybrid closed-loop systems and glycemic outcomes in children and adults with type 1 diabetes: real-world evidence from a U.S.-based multicenter collaborative. Diabetes Care. 2022;45(8):e118-e119. doi:10.2337/dc22-0329
50. Medtronic launches InPen with real-time Guardian Connect CGM data--the first integrated smart insulin pen for people with diabetes on MDI. News release. November 12, 2020. Accessed August 30, 2022. https://bit.ly/3CTSWPL
51. Bigfoot Biomedical receives FDA clearance for Bigfoot Unity Diabetes Management System, featuring first-of-its-kind smart pen caps for insulin pens used to treat type 1 and type 2 diabetes. News release. May 10, 2021. Accessed August 30, 2022. https://bit.ly/3BeyoAh
52. Vieira G. All about the CeQur Simplicity insulin patch. Updated May 24, 2022. Accessed August 30, 2022. https://beyondtype1.org/cequr-simplicity-insulin-patch/.
53. Messer LH, Tanenbaum ML, Cook PF, et al. Cost, hassle, and on-body experience: barriers to diabetes device use in adolescents and potential intervention targets. Diabetes Technol Ther. 2020;22(10):760-767. doi:10.1089/dia.2019.0509
54. Hilliard ME, Levy W, Anderson BJ, et al. Benefits and barriers of continuous glucose monitoring in young children with type 1 diabetes. Diabetes Technol Ther. 2019;21(9):493-498. doi:10.1089/dia.2019.0142
55. Dexcom G7 Release Delayed Until Late 2022. News release. August 8, 2022. Accessed September 7, 2022. https://diatribe.org/dexcom-g7-release-delayed-until-late-2022
56. Drucker DJ. Transforming type 1 diabetes: the next wave of innovation. Diabetologia. 2021;64(5):1059-1065. doi:10.1007/s00125-021-05396-5
57. Garg SK, Rodriguez E, Shah VN, Hirsch IB. New medications for the treatment of diabetes. Diabetes Technol Ther. 2022;24(S1):S190-S208. doi:10.1089/dia.2022.2513
58. Melton D. The promise of stem cell-derived islet replacement therapy. Diabetologia. 2021;64(5):1030-1036. doi:10.1007/s00125-020-05367-2
59. Danne T, Heinemann L, Bolinder J. New insulins, biosimilars, and insulin therapy. Diabetes Technol Ther. 2022;24(S1):S35-S57. doi:10.1089/dia.2022.2503
60. Kenney J. Insulin copay caps–a path to affordability. July 6, 2021. Accessed August 30, 2022.https://diatribechange.org/news/insulin-copay-caps-path-affordability
61. Glied SA, Zhu B. Not so sweet: insulin affordability over time. September 25, 2020. Accessed August 30, 2022. https://www.commonwealthfund.org/publications/issue-briefs/2020/sep/not-so-sweet-insulin-affordability-over-time
62. American Diabetes Association. Insulin and drug affordability. Accessed August 30, 2022. https://www.diabetes.org/advocacy/insulin-and-drug-affordability
63. Sullivan P. Chances for drug pricing, surprise billing action fade until November. March 24, 2020. Accessed August 30, 2022. https://thehill.com/policy/healthcare/489334-chances-for-drug-pricing-surprise-billing-action-fade-until-november/
64. Brown TD. How Medicare’s new Senior Savings Model makes insulin more affordable. June 4, 2020. Accessed August 30, 2022. https://www.diabetes.org/blog/how-medicares-new-senior-savings-model-makes-insulin-more-affordable
65. American Diabetes Association. ADA applauds the U.S. House of Representatives passage of the Affordable Insulin Now Act. News release. April 1, 2022. https://www.diabetes.org/newsroom/official-statement/2022/ada-applauds-us-house-of-representatives-passage-of-the-affordable-insulin-now-act
66. JDRF. Driving T1D cures during challenging times. 2022.
67. Medtronic announces ongoing initiatives to address health equity for people of color living with diabetes. News release. April 7, 2021. Access August 30, 2022. https://bit.ly/3KGTOZU
From the T1D Exchange, Boston, MA (Ann Mungmode, Nicole Rioles, Jesse Cases, Dr. Ebekozien); The Leona M. and Harry B. Hemsley Charitable Trust, New York, NY (Laurel Koester); and the University of Mississippi School of Population Health, Jackson, MS (Dr. Ebekozien).
Abstract
There have been remarkable innovations in diabetes management since the start of the COVID-19 pandemic, but these groundbreaking innovations are drawing limited focus as the field focuses on the adverse impact of the pandemic on patients with diabetes. This article reviews select population health innovations in diabetes management that have become available over the past 2 years of the COVID-19 pandemic from the perspective of the T1D Exchange Quality Improvement Collaborative, a learning health network that focuses on improving care and outcomes for individuals with type 1 diabetes (T1D). Such innovations include expanded telemedicine access, collection of real-world data, machine learning and artificial intelligence, and new diabetes medications and devices. In addition, multiple innovative studies have been undertaken to explore contributors to health inequities in diabetes, and advocacy efforts for specific populations have been successful. Looking to the future, work is required to explore additional health equity successes that do not further exacerbate inequities and to look for additional innovative ways to engage people with T1D in their health care through conversations on social determinants of health and societal structures.
Keywords: type 1 diabetes, learning health network, continuous glucose monitoring, health equity
One in 10 people in the United States has diabetes.1 Diabetes is the nation’s second leading cause of death, costing the US health system more than $300 billion annually.2 The COVID-19 pandemic presented additional health burdens for people living with diabetes. For example, preexisting diabetes was identified as a risk factor for COVID-19–associated morbidity and mortality.3,4 Over the past 2 years, there have been remarkable innovations in diabetes management, including stem cell therapy and new medication options. Additionally, improved technology solutions have aided in diabetes management through continuous glucose monitors (CGM), smart insulin pens, advanced hybrid closed-loop systems, and continuous subcutaneous insulin injections.5,6 Unfortunately, these groundbreaking innovations are drawing limited focus, as the field is rightfully focused on the adverse impact of the pandemic on patients with diabetes.

Learning health networks like the T1D Exchange Quality Improvement Collaborative (T1DX-QI) have implemented some of these innovative solutions to improve care for people with diabetes.7 T1DX-QI has more than 50 data-sharing endocrinology centers that care for over 75,000 people with diabetes across the United States (Figure 1). Centers participating in the T1DX-QI use quality improvement (QI) and implementation science methods to quickly translate research into evidence-based clinical practice. T1DX-QI leads diabetes population health and health system research and supports widespread transferability across health care organizations through regular collaborative calls, conferences, and case study documentation.8
In this review, we summarize impactful population health innovations in diabetes management that have become available over the past 2 years of the COVID-19 pandemic from the perspective of T1DX-QI (see Figure 2 for relevant definitions). This review is limited in scope and is not meant to be an exhaustive list of innovations. The review also reflects significant changes from the perspective of academic diabetes centers, which may not apply to rural or primary care diabetes practices.
Methods
The first (A.M.), second (H.H.), and senior (O.E.) authors conducted a scoping review of published literature using terms related to diabetes, population health, and innovation on PubMed Central and Google Scholar for the period March 2020 to June 2022. To complement the review, A.M. and O.E. also reviewed abstracts from presentations at major international diabetes conferences, including the American Diabetes Association (ADA), the International Society for Pediatric and Adolescent Diabetes (ISPAD), the T1DX-QI Learning Session Conference, and the Advanced Technologies & Treatments for Diabetes (ATTD) 2020 to 2022 conferences.9-14 The authors also searched FDA.gov and ClinicalTrials.gov for relevant insights. A.M. and O.E. sorted the reviewed literature into major themes (Figure 3) from the population health improvement perspective of the T1DX-QI.

Population Health Innovations in Diabetes Management
Expansion of Telemedicine Access
Telemedicine is cost-effective for patients with diabetes,15 including those with complex cases.16 Before the COVID-19 pandemic, telemedicine and virtual care were rare in diabetes management. However, the pandemic offered a new opportunity to expand the practice of telemedicine in diabetes management. A study from the T1DX-QI showed that telemedicine visits grew from comprising <1% of visits pre-pandemic (December 2019) to 95.2% during the pandemic (August 2020).17 Additional studies, like those conducted by Phillip et al,18 confirmed the noninferiority of telemedicine practice for patients with diabetes.Telemedicine was also found to be an effective strategy to educate patients on the use of diabetes technologies.19
Real-World Data and Disease Surveillance
As the COVID-19 pandemic exacerbated outcomes for people with type 1 diabetes (T1D), a need arose to understand the immediate effects of the pandemic on people with T1D through real-world data and disease surveillance. In April 2020, the T1DX-QI initiated a multicenter surveillance study to collect data and analyze the impact of COVID-19 on people with T1D. The existing health collaborative served as a springboard for robust surveillance study, documenting numerous works on the effects of COVID-19.3,4,20-28 Other investigators also embraced the power of real-world surveillance and real-world data.29,30
Big Data, Machine Learning, and Artificial Intelligence
The past 2 years have seen a shift toward embracing the incredible opportunity to tap the large volume of data generated from routine care for practical insights.31 In particular, researchers have demonstrated the widespread application of machine learning and artificial intelligence to improve diabetes management.32 The T1DX-QI also harnessed the growing power of big data by expanding the functionality of innovative benchmarking software. The T1DX QI Portal uses electronic medical record data of diabetes patients for clinic-to-clinic benchmarking and data analysis, using business intelligence solutions.33
Health Equity
While inequities across various health outcomes have been well documented for years,34 the COVID-19 pandemic further exaggerated racial/ethnic health inequities in T1D.23,35 In response, several organizations have outlined specific strategies to address these health inequities. Emboldened by the pandemic, the T1DX-QI announced a multipronged approach to address health inequities among patients with T1D through the Health Equity Advancement Lab (HEAL).36 One of HEAL’s main components is using real-world data to champion population-level insights and demonstrate progress in QI efforts.
Multiple innovative studies have been undertaken to explore contributors to health inequities in diabetes, and these studies are expanding our understanding of the chasm.37 There have also been innovative solutions to addressing these inequities, with multiple studies published over the past 2 years.38 A source of inequity among patients with T1D is the lack of representation of racial/ethnic minorities with T1D in clinical trials.39 The T1DX-QI suggests that the equity-adapted framework for QI can be applied by research leaders to support trial diversity and representation, ensuring future device innovations are meaningful for all people with T1D.40
Diabetes Devices
Glucose monitoring and insulin therapy are vital tools to support individuals living with T1D, and devices such as CGM and insulin pumps have become the standard of care for diabetes management (Table).41 Innovations in diabetes technology and device access are imperative for a chronic disease with no cure.

The COVID-19 pandemic created an opportunity to increase access to diabetes devices in inpatient settings. In 2020, the US Food and Drug Administration expanded the use of CGM to support remote monitoring of patients in inpatient hospital settings, simultaneously supporting the glucose monitoring needs of patients with T1D and reducing COVID-19 transmission through reduced patient-clinician contact.42 This effort has been expanded and will continue in 2022 and beyond,43 and aligns with the growing consensus that supports patients wearing both CGMs and insulin pumps in ambulatory settings to improve patient health outcomes.44
Since 2020, innovations in diabetes technology have improved and increased the variety of options available to people with T1D and made them easier to use (Table). New, advanced hybrid closed-loop systems have progressed to offer Bluetooth features, including automatic software upgrades, tubeless systems, and the ability to allow parents to use their smartphones to bolus for children.45-47 The next big step in insulin delivery innovation is the release of functioning, fully closed loop systems, of which several are currently in clinical trials.48 These systems support reduced hypoglycemia and improved time in range.49
Additional innovations in insulin delivery have improved the user experience and expanded therapeutic options, including a variety of smart insulin pens complete with dosing logs50,51 and even a patch to deliver insulin without the burden of injections.52 As barriers to diabetes technology persist,53 innovations in alternate insulin delivery provide people with T1D more options to align with their personal access and technology preferences.
Innovations in CGM address cited barriers to their use, including size or overall wear.53-55 CGMs released in the past few years are smaller in physical size, have longer durations of time between changings, are more accurate, and do not require calibrations for accuracy.
New Diabetes Medications
Many new medications and therapeutic advances have become available in the past 2 years.56 Additionally, more medications are being tested as adjunct therapies to support glycemic management in patients with T1D, including metformin, sodium-glucose cotransporter 1 and 2 inhibitors, pramlintide, glucagon-like polypeptide-1 analogs, and glucagon receptor agonists.57 Other recent advances include stem cell replacement therapy for patients with T1D.58 The ultra-long-acting biosimilar insulins are one medical innovation that has been stalled, rather than propelled, during the COVID-19 pandemic.59
Diabetes Policy Advocacy
People with T1D require insulin to survive. The cost of insulin has increased in recent years, with some studies citing a 64% to 100% increase in the past decade.60,61 In fact, 1 in 4 insulin users report that cost has impacted their insulin use, including rationing their insulin.62 Lockdowns during the COVID-19 pandemic stressed US families financially, increasing the urgency for insulin cost caps.
Although the COVID-19 pandemic halted national conversations on drug financing,63 advocacy efforts have succeeded for specific populations. The new Medicare Part D Senior Savings Model will cap the cost of insulin at $35 for a 30-day supply,64 and 20 states passed legislation capping insulin pricing.62 Efforts to codify national cost caps are under debate, including the passage of the Affordable Insulin Now Act, which passed the House in March 2022 and is currently under review in the Senate.65
Perspective: The Role of Private Philanthropy in Supporting Population Health Innovations
Funders and industry partners play a crucial role in leading and supporting innovations that improve the lives of people with T1D and reduce society’s costs of living with the disease. Data infrastructure is critical to supporting population health. While building the data infrastructure to support population health is both time- and resource-intensive, private foundations such as Helmsley are uniquely positioned—and have a responsibility—to take large, informed risks to help reach all communities with T1D.
The T1DX-QI is the largest source of population health data on T1D in the United States and is becoming the premiere data authority on its incidence, prevalence, and outcomes. The T1DX-QI enables a robust understanding of T1D-related health trends at the population level, as well as trends among clinics and providers. Pilot centers in the T1DX-QI have reported reductions in patients’ A1c and acute diabetes-related events, as well as improvements in device usage and depression screening. The ability to capture changes speaks to the promise and power of these data to demonstrate the clinical impact of QI interventions and to support the spread of best practices and learnings across health systems.
Additional philanthropic efforts have supported innovation in the last 2 years. For example, the JDRF, a nonprofit philanthropic equity firm, has supported efforts in developing artificial pancreas systems and cell therapies currently in clinical trials like teplizumab, a drug that has demonstrated delayed onset of T1D through JDRF’s T1D Fund.66 Industry partners also have an opportunity for significant influence in this area, as they continue to fund meaningful projects to advance care for people with T1D.67
Conclusion
We are optimistic that the innovations summarized here describe a shift in the tide of equitable T1D outcomes; however, future work is required to explore additional health equity successes that do not further exacerbate inequities. We also see further opportunities for innovative ways to engage people with T1D in their health care through conversations on social determinants of health and societal structures.
Corresponding author: Ann Mungmode, MPH, T1D Exchange, 11 Avenue de Lafayette, Boston, MA 02111; Email: amungmode@t1dexchange.org
Disclosures: Dr. Ebekozien serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for the Medtronic Advisory Board and received research grants from Medtronic Diabetes, Eli Lilly, and Dexcom.
Funding: The T1DX-QI is funded by The Leona M. and Harry B. Hemsley Charitable Trust.
From the T1D Exchange, Boston, MA (Ann Mungmode, Nicole Rioles, Jesse Cases, Dr. Ebekozien); The Leona M. and Harry B. Hemsley Charitable Trust, New York, NY (Laurel Koester); and the University of Mississippi School of Population Health, Jackson, MS (Dr. Ebekozien).
Abstract
There have been remarkable innovations in diabetes management since the start of the COVID-19 pandemic, but these groundbreaking innovations are drawing limited focus as the field focuses on the adverse impact of the pandemic on patients with diabetes. This article reviews select population health innovations in diabetes management that have become available over the past 2 years of the COVID-19 pandemic from the perspective of the T1D Exchange Quality Improvement Collaborative, a learning health network that focuses on improving care and outcomes for individuals with type 1 diabetes (T1D). Such innovations include expanded telemedicine access, collection of real-world data, machine learning and artificial intelligence, and new diabetes medications and devices. In addition, multiple innovative studies have been undertaken to explore contributors to health inequities in diabetes, and advocacy efforts for specific populations have been successful. Looking to the future, work is required to explore additional health equity successes that do not further exacerbate inequities and to look for additional innovative ways to engage people with T1D in their health care through conversations on social determinants of health and societal structures.
Keywords: type 1 diabetes, learning health network, continuous glucose monitoring, health equity
One in 10 people in the United States has diabetes.1 Diabetes is the nation’s second leading cause of death, costing the US health system more than $300 billion annually.2 The COVID-19 pandemic presented additional health burdens for people living with diabetes. For example, preexisting diabetes was identified as a risk factor for COVID-19–associated morbidity and mortality.3,4 Over the past 2 years, there have been remarkable innovations in diabetes management, including stem cell therapy and new medication options. Additionally, improved technology solutions have aided in diabetes management through continuous glucose monitors (CGM), smart insulin pens, advanced hybrid closed-loop systems, and continuous subcutaneous insulin injections.5,6 Unfortunately, these groundbreaking innovations are drawing limited focus, as the field is rightfully focused on the adverse impact of the pandemic on patients with diabetes.

Learning health networks like the T1D Exchange Quality Improvement Collaborative (T1DX-QI) have implemented some of these innovative solutions to improve care for people with diabetes.7 T1DX-QI has more than 50 data-sharing endocrinology centers that care for over 75,000 people with diabetes across the United States (Figure 1). Centers participating in the T1DX-QI use quality improvement (QI) and implementation science methods to quickly translate research into evidence-based clinical practice. T1DX-QI leads diabetes population health and health system research and supports widespread transferability across health care organizations through regular collaborative calls, conferences, and case study documentation.8
In this review, we summarize impactful population health innovations in diabetes management that have become available over the past 2 years of the COVID-19 pandemic from the perspective of T1DX-QI (see Figure 2 for relevant definitions). This review is limited in scope and is not meant to be an exhaustive list of innovations. The review also reflects significant changes from the perspective of academic diabetes centers, which may not apply to rural or primary care diabetes practices.
Methods
The first (A.M.), second (H.H.), and senior (O.E.) authors conducted a scoping review of published literature using terms related to diabetes, population health, and innovation on PubMed Central and Google Scholar for the period March 2020 to June 2022. To complement the review, A.M. and O.E. also reviewed abstracts from presentations at major international diabetes conferences, including the American Diabetes Association (ADA), the International Society for Pediatric and Adolescent Diabetes (ISPAD), the T1DX-QI Learning Session Conference, and the Advanced Technologies & Treatments for Diabetes (ATTD) 2020 to 2022 conferences.9-14 The authors also searched FDA.gov and ClinicalTrials.gov for relevant insights. A.M. and O.E. sorted the reviewed literature into major themes (Figure 3) from the population health improvement perspective of the T1DX-QI.

Population Health Innovations in Diabetes Management
Expansion of Telemedicine Access
Telemedicine is cost-effective for patients with diabetes,15 including those with complex cases.16 Before the COVID-19 pandemic, telemedicine and virtual care were rare in diabetes management. However, the pandemic offered a new opportunity to expand the practice of telemedicine in diabetes management. A study from the T1DX-QI showed that telemedicine visits grew from comprising <1% of visits pre-pandemic (December 2019) to 95.2% during the pandemic (August 2020).17 Additional studies, like those conducted by Phillip et al,18 confirmed the noninferiority of telemedicine practice for patients with diabetes.Telemedicine was also found to be an effective strategy to educate patients on the use of diabetes technologies.19
Real-World Data and Disease Surveillance
As the COVID-19 pandemic exacerbated outcomes for people with type 1 diabetes (T1D), a need arose to understand the immediate effects of the pandemic on people with T1D through real-world data and disease surveillance. In April 2020, the T1DX-QI initiated a multicenter surveillance study to collect data and analyze the impact of COVID-19 on people with T1D. The existing health collaborative served as a springboard for robust surveillance study, documenting numerous works on the effects of COVID-19.3,4,20-28 Other investigators also embraced the power of real-world surveillance and real-world data.29,30
Big Data, Machine Learning, and Artificial Intelligence
The past 2 years have seen a shift toward embracing the incredible opportunity to tap the large volume of data generated from routine care for practical insights.31 In particular, researchers have demonstrated the widespread application of machine learning and artificial intelligence to improve diabetes management.32 The T1DX-QI also harnessed the growing power of big data by expanding the functionality of innovative benchmarking software. The T1DX QI Portal uses electronic medical record data of diabetes patients for clinic-to-clinic benchmarking and data analysis, using business intelligence solutions.33
Health Equity
While inequities across various health outcomes have been well documented for years,34 the COVID-19 pandemic further exaggerated racial/ethnic health inequities in T1D.23,35 In response, several organizations have outlined specific strategies to address these health inequities. Emboldened by the pandemic, the T1DX-QI announced a multipronged approach to address health inequities among patients with T1D through the Health Equity Advancement Lab (HEAL).36 One of HEAL’s main components is using real-world data to champion population-level insights and demonstrate progress in QI efforts.
Multiple innovative studies have been undertaken to explore contributors to health inequities in diabetes, and these studies are expanding our understanding of the chasm.37 There have also been innovative solutions to addressing these inequities, with multiple studies published over the past 2 years.38 A source of inequity among patients with T1D is the lack of representation of racial/ethnic minorities with T1D in clinical trials.39 The T1DX-QI suggests that the equity-adapted framework for QI can be applied by research leaders to support trial diversity and representation, ensuring future device innovations are meaningful for all people with T1D.40
Diabetes Devices
Glucose monitoring and insulin therapy are vital tools to support individuals living with T1D, and devices such as CGM and insulin pumps have become the standard of care for diabetes management (Table).41 Innovations in diabetes technology and device access are imperative for a chronic disease with no cure.

The COVID-19 pandemic created an opportunity to increase access to diabetes devices in inpatient settings. In 2020, the US Food and Drug Administration expanded the use of CGM to support remote monitoring of patients in inpatient hospital settings, simultaneously supporting the glucose monitoring needs of patients with T1D and reducing COVID-19 transmission through reduced patient-clinician contact.42 This effort has been expanded and will continue in 2022 and beyond,43 and aligns with the growing consensus that supports patients wearing both CGMs and insulin pumps in ambulatory settings to improve patient health outcomes.44
Since 2020, innovations in diabetes technology have improved and increased the variety of options available to people with T1D and made them easier to use (Table). New, advanced hybrid closed-loop systems have progressed to offer Bluetooth features, including automatic software upgrades, tubeless systems, and the ability to allow parents to use their smartphones to bolus for children.45-47 The next big step in insulin delivery innovation is the release of functioning, fully closed loop systems, of which several are currently in clinical trials.48 These systems support reduced hypoglycemia and improved time in range.49
Additional innovations in insulin delivery have improved the user experience and expanded therapeutic options, including a variety of smart insulin pens complete with dosing logs50,51 and even a patch to deliver insulin without the burden of injections.52 As barriers to diabetes technology persist,53 innovations in alternate insulin delivery provide people with T1D more options to align with their personal access and technology preferences.
Innovations in CGM address cited barriers to their use, including size or overall wear.53-55 CGMs released in the past few years are smaller in physical size, have longer durations of time between changings, are more accurate, and do not require calibrations for accuracy.
New Diabetes Medications
Many new medications and therapeutic advances have become available in the past 2 years.56 Additionally, more medications are being tested as adjunct therapies to support glycemic management in patients with T1D, including metformin, sodium-glucose cotransporter 1 and 2 inhibitors, pramlintide, glucagon-like polypeptide-1 analogs, and glucagon receptor agonists.57 Other recent advances include stem cell replacement therapy for patients with T1D.58 The ultra-long-acting biosimilar insulins are one medical innovation that has been stalled, rather than propelled, during the COVID-19 pandemic.59
Diabetes Policy Advocacy
People with T1D require insulin to survive. The cost of insulin has increased in recent years, with some studies citing a 64% to 100% increase in the past decade.60,61 In fact, 1 in 4 insulin users report that cost has impacted their insulin use, including rationing their insulin.62 Lockdowns during the COVID-19 pandemic stressed US families financially, increasing the urgency for insulin cost caps.
Although the COVID-19 pandemic halted national conversations on drug financing,63 advocacy efforts have succeeded for specific populations. The new Medicare Part D Senior Savings Model will cap the cost of insulin at $35 for a 30-day supply,64 and 20 states passed legislation capping insulin pricing.62 Efforts to codify national cost caps are under debate, including the passage of the Affordable Insulin Now Act, which passed the House in March 2022 and is currently under review in the Senate.65
Perspective: The Role of Private Philanthropy in Supporting Population Health Innovations
Funders and industry partners play a crucial role in leading and supporting innovations that improve the lives of people with T1D and reduce society’s costs of living with the disease. Data infrastructure is critical to supporting population health. While building the data infrastructure to support population health is both time- and resource-intensive, private foundations such as Helmsley are uniquely positioned—and have a responsibility—to take large, informed risks to help reach all communities with T1D.
The T1DX-QI is the largest source of population health data on T1D in the United States and is becoming the premiere data authority on its incidence, prevalence, and outcomes. The T1DX-QI enables a robust understanding of T1D-related health trends at the population level, as well as trends among clinics and providers. Pilot centers in the T1DX-QI have reported reductions in patients’ A1c and acute diabetes-related events, as well as improvements in device usage and depression screening. The ability to capture changes speaks to the promise and power of these data to demonstrate the clinical impact of QI interventions and to support the spread of best practices and learnings across health systems.
Additional philanthropic efforts have supported innovation in the last 2 years. For example, the JDRF, a nonprofit philanthropic equity firm, has supported efforts in developing artificial pancreas systems and cell therapies currently in clinical trials like teplizumab, a drug that has demonstrated delayed onset of T1D through JDRF’s T1D Fund.66 Industry partners also have an opportunity for significant influence in this area, as they continue to fund meaningful projects to advance care for people with T1D.67
Conclusion
We are optimistic that the innovations summarized here describe a shift in the tide of equitable T1D outcomes; however, future work is required to explore additional health equity successes that do not further exacerbate inequities. We also see further opportunities for innovative ways to engage people with T1D in their health care through conversations on social determinants of health and societal structures.
Corresponding author: Ann Mungmode, MPH, T1D Exchange, 11 Avenue de Lafayette, Boston, MA 02111; Email: amungmode@t1dexchange.org
Disclosures: Dr. Ebekozien serve(d) as a director, officer, partner, employee, advisor, consultant, or trustee for the Medtronic Advisory Board and received research grants from Medtronic Diabetes, Eli Lilly, and Dexcom.
Funding: The T1DX-QI is funded by The Leona M. and Harry B. Hemsley Charitable Trust.
1. Centers for Disease Control and Prevention. National diabetes statistics report. Accessed August 30, 2022. www.cdc.gov/diabetes/data/statistics-report/index.html
2. Centers for Disease Control and Prevention. Diabetes fast facts. Accessed August 30, 2022. www.cdc.gov/diabetes/basics/quick-facts.html
3. O’Malley G, Ebekozien O, Desimone M, et al. COVID-19 hospitalization in adults with type 1 diabetes: results from the T1D Exchange Multicenter Surveillance Study. J Clin Endocrinol Metab. 2020;106(2):e936-e942. doi:10.1210/clinem/dgaa825
4. Ebekozien OA, Noor N, Gallagher MP, Alonso GT. Type 1 diabetes and COVID-19: preliminary findings from a multicenter surveillance study in the U.S. Diabetes Care. 2020;43(8):e83-e85. doi:10.2337/dc20-1088
5. Zimmerman C, Albanese-O’Neill A, Haller MJ. Advances in type 1 diabetes technology over the last decade. Eur Endocrinol. 2019;15(2):70-76. doi:10.17925/ee.2019.15.2.70
6. Wake DJ, Gibb FW, Kar P, et al. Endocrinology in the time of COVID-19: remodelling diabetes services and emerging innovation. Eur J Endocrinol. 2020;183(2):G67-G77. doi:10.1530/eje-20-0377
7. Alonso GT, Corathers S, Shah A, et al. Establishment of the T1D Exchange Quality Improvement Collaborative (T1DX-QI). Clin Diabetes. 2020;38(2):141-151. doi:10.2337/cd19-0032
8. Ginnard OZB, Alonso GT, Corathers SD, et al. Quality improvement in diabetes care: a review of initiatives and outcomes in the T1D Exchange Quality Improvement Collaborative. Clin Diabetes. 2021;39(3):256-263. doi:10.2337/cd21-0029
9. ATTD 2021 invited speaker abstracts. Diabetes Technol Ther. 2021;23(S2):A1-A206. doi:10.1089/dia.2021.2525.abstracts
10. Rompicherla SN, Edelen N, Gallagher R, et al. Children and adolescent patients with pre-existing type 1 diabetes and additional comorbidities have an increased risk of hospitalization from COVID-19; data from the T1D Exchange COVID Registry. Pediatr Diabetes. 2021;22(S30):3-32. doi:10.1111/pedi.13268
11. Abstracts for the T1D Exchange QI Collaborative (T1DX-QI) Learning Session 2021. November 8-9, 2021. J Diabetes. 2021;13(S1):3-17. doi:10.1111/1753-0407.13227
12. The Official Journal of ATTD Advanced Technologies & Treatments for Diabetes conference 27-30 April 2022. Barcelona and online. Diabetes Technol Ther. 2022;24(S1):A1-A237. doi:10.1089/dia.2022.2525.abstracts
13. Ebekozien ON, Kamboj N, Odugbesan MK, et al. Inequities in glycemic outcomes for patients with type 1 diabetes: six-year (2016-2021) longitudinal follow-up by race and ethnicity of 36,390 patients in the T1DX-QI Collaborative. Diabetes. 2022;71(suppl 1). doi:10.2337/db22-167-OR
14. Narayan KA, Noor M, Rompicherla N, et al. No BMI increase during the COVID-pandemic in children and adults with T1D in three continents: joint analysis of ADDN, T1DX, and DPV registries. Diabetes. 2022;71(suppl 1). doi:10.2337/db22-269-OR
15. Lee JY, Lee SWH. Telemedicine cost-effectiveness for diabetes management: a systematic review. Diabetes Technol Ther. 2018;20(7):492-500. doi:10.1089/dia.2018.0098
16. McDonnell ME. Telemedicine in complex diabetes management. Curr Diab Rep. 2018;18(7):42. doi:10.1007/s11892-018-1015-3
17. Lee JM, Carlson E, Albanese-O’Neill A, et al. Adoption of telemedicine for type 1 diabetes care during the COVID-19 pandemic. Diabetes Technol Ther. 2021;23(9):642-651. doi:10.1089/dia.2021.0080
18. Phillip M, Bergenstal RM, Close KL, et al. The digital/virtual diabetes clinic: the future is now–recommendations from an international panel on diabetes digital technologies introduction. Diabetes Technol Ther. 2021;23(2):146-154. doi:10.1089/dia.2020.0375
19. Garg SK, Rodriguez E. COVID‐19 pandemic and diabetes care. Diabetes Technol Ther. 2022;24(S1):S2-S20. doi:10.1089/dia.2022.2501
20. Beliard K, Ebekozien O, Demeterco-Berggren C, et al. Increased DKA at presentation among newly diagnosed type 1 diabetes patients with or without COVID-19: data from a multi-site surveillance registry. J Diabetes. 2021;13(3):270-272. doi:10.1111/1753-0407.13141
21. Ebekozien O, Agarwal S, Noor N, et al. Inequities in diabetic ketoacidosis among patients with type 1 diabetes and COVID-19: data from 52 US clinical centers. J Clin Endocrinol Metab. 2020;106(4):1755-1762. doi:10.1210/clinem/dgaa920
22. Alonso GT, Ebekozien O, Gallagher MP, et al. Diabetic ketoacidosis drives COVID-19 related hospitalizations in children with type 1 diabetes. J Diabetes. 2021;13(8):681-687. doi:10.1111/1753-0407.13184
23. Noor N, Ebekozien O, Levin L, et al. Diabetes technology use for management of type 1 diabetes is associated with fewer adverse COVID-19 outcomes: findings from the T1D Exchange COVID-19 Surveillance Registry. Diabetes Care. 2021;44(8):e160-e162. doi:10.2337/dc21-0074
24. Demeterco-Berggren C, Ebekozien O, Rompicherla S, et al. Age and hospitalization risk in people with type 1 diabetes and COVID-19: data from the T1D Exchange Surveillance Study. J Clin Endocrinol Metab. 2021;107(2):410-418. doi:10.1210/clinem/dgab668
25. DeSalvo DJ, Noor N, Xie C, et al. Patient demographics and clinical outcomes among type 1 diabetes patients using continuous glucose monitors: data from T1D Exchange real-world observational study. J Diabetes Sci Technol. 2021 Oct 9. [Epub ahead of print] doi:10.1177/19322968211049783
26. Gallagher MP, Rompicherla S, Ebekozien O, et al. Differences in COVID-19 outcomes among patients with type 1 diabetes: first vs later surges. J Clin Outcomes Manage. 2022;29(1):27-31. doi:10.12788/jcom.0084
27. Wolf RM, Noor N, Izquierdo R, et al. Increase in newly diagnosed type 1 diabetes in youth during the COVID-19 pandemic in the United States: a multi-center analysis. Pediatr Diabetes. 2022;23(4):433-438. doi:10.1111/pedi.13328
28. Lavik AR, Ebekozien O, Noor N, et al. Trends in type 1 diabetic ketoacidosis during COVID-19 surges at 7 US centers: highest burden on non-Hispanic Black patients. J Clin Endocrinol Metab. 2022;107(7):1948-1955. doi:10.1210/clinem/dgac158
29. van der Linden J, Welsh JB, Hirsch IB, Garg SK. Real-time continuous glucose monitoring during the coronavirus disease 2019 pandemic and its impact on time in range. Diabetes Technol Ther. 2021;23(S1):S1-S7. doi:10.1089/dia.2020.0649
30. Nwosu BU, Al-Halbouni L, Parajuli S, et al. COVID-19 pandemic and pediatric type 1 diabetes: no significant change in glycemic control during the pandemic lockdown of 2020. Front Endocrinol (Lausanne). 2021;12:703905. doi:10.3389/fendo.2021.703905
31. Ellahham S. Artificial intelligence: the future for diabetes care. Am J Med. 2020;133(8):895-900. doi:10.1016/j.amjmed.2020.03.033
32. Nomura A, Noguchi M, Kometani M, et al. Artificial intelligence in current diabetes management and prediction. Curr Diab Rep. 2021;21(12):61. doi:10.1007/s11892-021-01423-2
33. Mungmode A, Noor N, Weinstock RS, et al. Making diabetes electronic medical record data actionable: promoting benchmarking and population health using the T1D Exchange Quality Improvement Portal. Clin Diabetes. Forthcoming 2022.
34. Lavizzo-Mourey RJ, Besser RE, Williams DR. Understanding and mitigating health inequities—past, current, and future directions. N Engl J Med. 2021;384(18):1681-1684. doi:10.1056/NEJMp2008628
35. Majidi S, Ebekozien O, Noor N, et al. Inequities in health outcomes in children and adults with type 1 diabetes: data from the T1D Exchange Quality Improvement Collaborative. Clin Diabetes. 2021;39(3):278-283. doi:10.2337/cd21-0028
36. Ebekozien O, Mungmode A, Odugbesan O, et al. Addressing type 1 diabetes health inequities in the United States: approaches from the T1D Exchange QI Collaborative. J Diabetes. 2022;14(1):79-82. doi:10.1111/1753-0407.13235
37. Odugbesan O, Addala A, Nelson G, et al. Implicit racial-ethnic and insurance-mediated bias to recommending diabetes technology: insights from T1D Exchange multicenter pediatric and adult diabetes provider cohort. Diabetes Technol Ther. 2022 Jun 13. [Epub ahead of print] doi:10.1089/dia.2022.0042
38. Schmitt J, Fogle K, Scott ML, Iyer P. Improving equitable access to continuous glucose monitors for Alabama’s children with type 1 diabetes: a quality improvement project. Diabetes Technol Ther. 2022;24(7):481-491. doi:10.1089/dia.2021.0511
39. Akturk HK, Agarwal S, Hoffecker L, Shah VN. Inequity in racial-ethnic representation in randomized controlled trials of diabetes technologies in type 1 diabetes: critical need for new standards. Diabetes Care. 2021;44(6):e121-e123. doi:10.2337/dc20-3063
40. Ebekozien O, Mungmode A, Buckingham D, et al. Achieving equity in diabetes research: borrowing from the field of quality improvement using a practical framework and improvement tools. Diabetes Spectr. 2022;35(3):304-312. doi:10.2237/dsi22-0002
41. Zhang J, Xu J, Lim J, et al. Wearable glucose monitoring and implantable drug delivery systems for diabetes management. Adv Healthc Mater. 2021;10(17):e2100194. doi:10.1002/adhm.202100194
42. FDA expands remote patient monitoring in hospitals for people with diabetes during COVID-19; manufacturers donate CGM supplies. News release. April 21, 2020. Accessed August 30, 2022. https://www.diabetes.org/newsroom/press-releases/2020/fda-remote-patient-monitoring-cgm
43. Campbell P. FDA grants Dexcom CGM breakthrough designation for in-hospital use. March 2, 2022. Accessed August 30, 2022. https://www.endocrinologynetwork.com/view/fda-grants-dexcom-cgm-breakthrough-designation-for-in-hospital-use
44. Yeh T, Yeung M, Mendelsohn Curanaj FA. Managing patients with insulin pumps and continuous glucose monitors in the hospital: to wear or not to wear. Curr Diab Rep. 2021;21(2):7. doi:10.1007/s11892-021-01375-7
45. Medtronic announces FDA approval for MiniMed 770G insulin pump system. News release. September 21, 2020. Accessed August 30, 2022. https://bit.ly/3TyEna4
46. Tandem Diabetes Care announces commercial launch of the t:slim X2 insulin pump with Control-IQ technology in the United States. News release. January 15, 2020. Accessed August 30, 2022. https://investor.tandemdiabetes.com/news-releases/news-release-details/tandem-diabetes-care-announces-commercial-launch-tslim-x2-0
47. Garza M, Gutow H, Mahoney K. Omnipod 5 cleared by the FDA. Updated August 22, 2022. Accessed August 30, 2022.https://diatribe.org/omnipod-5-approved-fda
48. Boughton CK. Fully closed-loop insulin delivery—are we nearly there yet? Lancet Digit Health. 2021;3(11):e689-e690. doi:10.1016/s2589-7500(21)00218-1
49. Noor N, Kamboj MK, Triolo T, et al. Hybrid closed-loop systems and glycemic outcomes in children and adults with type 1 diabetes: real-world evidence from a U.S.-based multicenter collaborative. Diabetes Care. 2022;45(8):e118-e119. doi:10.2337/dc22-0329
50. Medtronic launches InPen with real-time Guardian Connect CGM data--the first integrated smart insulin pen for people with diabetes on MDI. News release. November 12, 2020. Accessed August 30, 2022. https://bit.ly/3CTSWPL
51. Bigfoot Biomedical receives FDA clearance for Bigfoot Unity Diabetes Management System, featuring first-of-its-kind smart pen caps for insulin pens used to treat type 1 and type 2 diabetes. News release. May 10, 2021. Accessed August 30, 2022. https://bit.ly/3BeyoAh
52. Vieira G. All about the CeQur Simplicity insulin patch. Updated May 24, 2022. Accessed August 30, 2022. https://beyondtype1.org/cequr-simplicity-insulin-patch/.
53. Messer LH, Tanenbaum ML, Cook PF, et al. Cost, hassle, and on-body experience: barriers to diabetes device use in adolescents and potential intervention targets. Diabetes Technol Ther. 2020;22(10):760-767. doi:10.1089/dia.2019.0509
54. Hilliard ME, Levy W, Anderson BJ, et al. Benefits and barriers of continuous glucose monitoring in young children with type 1 diabetes. Diabetes Technol Ther. 2019;21(9):493-498. doi:10.1089/dia.2019.0142
55. Dexcom G7 Release Delayed Until Late 2022. News release. August 8, 2022. Accessed September 7, 2022. https://diatribe.org/dexcom-g7-release-delayed-until-late-2022
56. Drucker DJ. Transforming type 1 diabetes: the next wave of innovation. Diabetologia. 2021;64(5):1059-1065. doi:10.1007/s00125-021-05396-5
57. Garg SK, Rodriguez E, Shah VN, Hirsch IB. New medications for the treatment of diabetes. Diabetes Technol Ther. 2022;24(S1):S190-S208. doi:10.1089/dia.2022.2513
58. Melton D. The promise of stem cell-derived islet replacement therapy. Diabetologia. 2021;64(5):1030-1036. doi:10.1007/s00125-020-05367-2
59. Danne T, Heinemann L, Bolinder J. New insulins, biosimilars, and insulin therapy. Diabetes Technol Ther. 2022;24(S1):S35-S57. doi:10.1089/dia.2022.2503
60. Kenney J. Insulin copay caps–a path to affordability. July 6, 2021. Accessed August 30, 2022.https://diatribechange.org/news/insulin-copay-caps-path-affordability
61. Glied SA, Zhu B. Not so sweet: insulin affordability over time. September 25, 2020. Accessed August 30, 2022. https://www.commonwealthfund.org/publications/issue-briefs/2020/sep/not-so-sweet-insulin-affordability-over-time
62. American Diabetes Association. Insulin and drug affordability. Accessed August 30, 2022. https://www.diabetes.org/advocacy/insulin-and-drug-affordability
63. Sullivan P. Chances for drug pricing, surprise billing action fade until November. March 24, 2020. Accessed August 30, 2022. https://thehill.com/policy/healthcare/489334-chances-for-drug-pricing-surprise-billing-action-fade-until-november/
64. Brown TD. How Medicare’s new Senior Savings Model makes insulin more affordable. June 4, 2020. Accessed August 30, 2022. https://www.diabetes.org/blog/how-medicares-new-senior-savings-model-makes-insulin-more-affordable
65. American Diabetes Association. ADA applauds the U.S. House of Representatives passage of the Affordable Insulin Now Act. News release. April 1, 2022. https://www.diabetes.org/newsroom/official-statement/2022/ada-applauds-us-house-of-representatives-passage-of-the-affordable-insulin-now-act
66. JDRF. Driving T1D cures during challenging times. 2022.
67. Medtronic announces ongoing initiatives to address health equity for people of color living with diabetes. News release. April 7, 2021. Access August 30, 2022. https://bit.ly/3KGTOZU
1. Centers for Disease Control and Prevention. National diabetes statistics report. Accessed August 30, 2022. www.cdc.gov/diabetes/data/statistics-report/index.html
2. Centers for Disease Control and Prevention. Diabetes fast facts. Accessed August 30, 2022. www.cdc.gov/diabetes/basics/quick-facts.html
3. O’Malley G, Ebekozien O, Desimone M, et al. COVID-19 hospitalization in adults with type 1 diabetes: results from the T1D Exchange Multicenter Surveillance Study. J Clin Endocrinol Metab. 2020;106(2):e936-e942. doi:10.1210/clinem/dgaa825
4. Ebekozien OA, Noor N, Gallagher MP, Alonso GT. Type 1 diabetes and COVID-19: preliminary findings from a multicenter surveillance study in the U.S. Diabetes Care. 2020;43(8):e83-e85. doi:10.2337/dc20-1088
5. Zimmerman C, Albanese-O’Neill A, Haller MJ. Advances in type 1 diabetes technology over the last decade. Eur Endocrinol. 2019;15(2):70-76. doi:10.17925/ee.2019.15.2.70
6. Wake DJ, Gibb FW, Kar P, et al. Endocrinology in the time of COVID-19: remodelling diabetes services and emerging innovation. Eur J Endocrinol. 2020;183(2):G67-G77. doi:10.1530/eje-20-0377
7. Alonso GT, Corathers S, Shah A, et al. Establishment of the T1D Exchange Quality Improvement Collaborative (T1DX-QI). Clin Diabetes. 2020;38(2):141-151. doi:10.2337/cd19-0032
8. Ginnard OZB, Alonso GT, Corathers SD, et al. Quality improvement in diabetes care: a review of initiatives and outcomes in the T1D Exchange Quality Improvement Collaborative. Clin Diabetes. 2021;39(3):256-263. doi:10.2337/cd21-0029
9. ATTD 2021 invited speaker abstracts. Diabetes Technol Ther. 2021;23(S2):A1-A206. doi:10.1089/dia.2021.2525.abstracts
10. Rompicherla SN, Edelen N, Gallagher R, et al. Children and adolescent patients with pre-existing type 1 diabetes and additional comorbidities have an increased risk of hospitalization from COVID-19; data from the T1D Exchange COVID Registry. Pediatr Diabetes. 2021;22(S30):3-32. doi:10.1111/pedi.13268
11. Abstracts for the T1D Exchange QI Collaborative (T1DX-QI) Learning Session 2021. November 8-9, 2021. J Diabetes. 2021;13(S1):3-17. doi:10.1111/1753-0407.13227
12. The Official Journal of ATTD Advanced Technologies & Treatments for Diabetes conference 27-30 April 2022. Barcelona and online. Diabetes Technol Ther. 2022;24(S1):A1-A237. doi:10.1089/dia.2022.2525.abstracts
13. Ebekozien ON, Kamboj N, Odugbesan MK, et al. Inequities in glycemic outcomes for patients with type 1 diabetes: six-year (2016-2021) longitudinal follow-up by race and ethnicity of 36,390 patients in the T1DX-QI Collaborative. Diabetes. 2022;71(suppl 1). doi:10.2337/db22-167-OR
14. Narayan KA, Noor M, Rompicherla N, et al. No BMI increase during the COVID-pandemic in children and adults with T1D in three continents: joint analysis of ADDN, T1DX, and DPV registries. Diabetes. 2022;71(suppl 1). doi:10.2337/db22-269-OR
15. Lee JY, Lee SWH. Telemedicine cost-effectiveness for diabetes management: a systematic review. Diabetes Technol Ther. 2018;20(7):492-500. doi:10.1089/dia.2018.0098
16. McDonnell ME. Telemedicine in complex diabetes management. Curr Diab Rep. 2018;18(7):42. doi:10.1007/s11892-018-1015-3
17. Lee JM, Carlson E, Albanese-O’Neill A, et al. Adoption of telemedicine for type 1 diabetes care during the COVID-19 pandemic. Diabetes Technol Ther. 2021;23(9):642-651. doi:10.1089/dia.2021.0080
18. Phillip M, Bergenstal RM, Close KL, et al. The digital/virtual diabetes clinic: the future is now–recommendations from an international panel on diabetes digital technologies introduction. Diabetes Technol Ther. 2021;23(2):146-154. doi:10.1089/dia.2020.0375
19. Garg SK, Rodriguez E. COVID‐19 pandemic and diabetes care. Diabetes Technol Ther. 2022;24(S1):S2-S20. doi:10.1089/dia.2022.2501
20. Beliard K, Ebekozien O, Demeterco-Berggren C, et al. Increased DKA at presentation among newly diagnosed type 1 diabetes patients with or without COVID-19: data from a multi-site surveillance registry. J Diabetes. 2021;13(3):270-272. doi:10.1111/1753-0407.13141
21. Ebekozien O, Agarwal S, Noor N, et al. Inequities in diabetic ketoacidosis among patients with type 1 diabetes and COVID-19: data from 52 US clinical centers. J Clin Endocrinol Metab. 2020;106(4):1755-1762. doi:10.1210/clinem/dgaa920
22. Alonso GT, Ebekozien O, Gallagher MP, et al. Diabetic ketoacidosis drives COVID-19 related hospitalizations in children with type 1 diabetes. J Diabetes. 2021;13(8):681-687. doi:10.1111/1753-0407.13184
23. Noor N, Ebekozien O, Levin L, et al. Diabetes technology use for management of type 1 diabetes is associated with fewer adverse COVID-19 outcomes: findings from the T1D Exchange COVID-19 Surveillance Registry. Diabetes Care. 2021;44(8):e160-e162. doi:10.2337/dc21-0074
24. Demeterco-Berggren C, Ebekozien O, Rompicherla S, et al. Age and hospitalization risk in people with type 1 diabetes and COVID-19: data from the T1D Exchange Surveillance Study. J Clin Endocrinol Metab. 2021;107(2):410-418. doi:10.1210/clinem/dgab668
25. DeSalvo DJ, Noor N, Xie C, et al. Patient demographics and clinical outcomes among type 1 diabetes patients using continuous glucose monitors: data from T1D Exchange real-world observational study. J Diabetes Sci Technol. 2021 Oct 9. [Epub ahead of print] doi:10.1177/19322968211049783
26. Gallagher MP, Rompicherla S, Ebekozien O, et al. Differences in COVID-19 outcomes among patients with type 1 diabetes: first vs later surges. J Clin Outcomes Manage. 2022;29(1):27-31. doi:10.12788/jcom.0084
27. Wolf RM, Noor N, Izquierdo R, et al. Increase in newly diagnosed type 1 diabetes in youth during the COVID-19 pandemic in the United States: a multi-center analysis. Pediatr Diabetes. 2022;23(4):433-438. doi:10.1111/pedi.13328
28. Lavik AR, Ebekozien O, Noor N, et al. Trends in type 1 diabetic ketoacidosis during COVID-19 surges at 7 US centers: highest burden on non-Hispanic Black patients. J Clin Endocrinol Metab. 2022;107(7):1948-1955. doi:10.1210/clinem/dgac158
29. van der Linden J, Welsh JB, Hirsch IB, Garg SK. Real-time continuous glucose monitoring during the coronavirus disease 2019 pandemic and its impact on time in range. Diabetes Technol Ther. 2021;23(S1):S1-S7. doi:10.1089/dia.2020.0649
30. Nwosu BU, Al-Halbouni L, Parajuli S, et al. COVID-19 pandemic and pediatric type 1 diabetes: no significant change in glycemic control during the pandemic lockdown of 2020. Front Endocrinol (Lausanne). 2021;12:703905. doi:10.3389/fendo.2021.703905
31. Ellahham S. Artificial intelligence: the future for diabetes care. Am J Med. 2020;133(8):895-900. doi:10.1016/j.amjmed.2020.03.033
32. Nomura A, Noguchi M, Kometani M, et al. Artificial intelligence in current diabetes management and prediction. Curr Diab Rep. 2021;21(12):61. doi:10.1007/s11892-021-01423-2
33. Mungmode A, Noor N, Weinstock RS, et al. Making diabetes electronic medical record data actionable: promoting benchmarking and population health using the T1D Exchange Quality Improvement Portal. Clin Diabetes. Forthcoming 2022.
34. Lavizzo-Mourey RJ, Besser RE, Williams DR. Understanding and mitigating health inequities—past, current, and future directions. N Engl J Med. 2021;384(18):1681-1684. doi:10.1056/NEJMp2008628
35. Majidi S, Ebekozien O, Noor N, et al. Inequities in health outcomes in children and adults with type 1 diabetes: data from the T1D Exchange Quality Improvement Collaborative. Clin Diabetes. 2021;39(3):278-283. doi:10.2337/cd21-0028
36. Ebekozien O, Mungmode A, Odugbesan O, et al. Addressing type 1 diabetes health inequities in the United States: approaches from the T1D Exchange QI Collaborative. J Diabetes. 2022;14(1):79-82. doi:10.1111/1753-0407.13235
37. Odugbesan O, Addala A, Nelson G, et al. Implicit racial-ethnic and insurance-mediated bias to recommending diabetes technology: insights from T1D Exchange multicenter pediatric and adult diabetes provider cohort. Diabetes Technol Ther. 2022 Jun 13. [Epub ahead of print] doi:10.1089/dia.2022.0042
38. Schmitt J, Fogle K, Scott ML, Iyer P. Improving equitable access to continuous glucose monitors for Alabama’s children with type 1 diabetes: a quality improvement project. Diabetes Technol Ther. 2022;24(7):481-491. doi:10.1089/dia.2021.0511
39. Akturk HK, Agarwal S, Hoffecker L, Shah VN. Inequity in racial-ethnic representation in randomized controlled trials of diabetes technologies in type 1 diabetes: critical need for new standards. Diabetes Care. 2021;44(6):e121-e123. doi:10.2337/dc20-3063
40. Ebekozien O, Mungmode A, Buckingham D, et al. Achieving equity in diabetes research: borrowing from the field of quality improvement using a practical framework and improvement tools. Diabetes Spectr. 2022;35(3):304-312. doi:10.2237/dsi22-0002
41. Zhang J, Xu J, Lim J, et al. Wearable glucose monitoring and implantable drug delivery systems for diabetes management. Adv Healthc Mater. 2021;10(17):e2100194. doi:10.1002/adhm.202100194
42. FDA expands remote patient monitoring in hospitals for people with diabetes during COVID-19; manufacturers donate CGM supplies. News release. April 21, 2020. Accessed August 30, 2022. https://www.diabetes.org/newsroom/press-releases/2020/fda-remote-patient-monitoring-cgm
43. Campbell P. FDA grants Dexcom CGM breakthrough designation for in-hospital use. March 2, 2022. Accessed August 30, 2022. https://www.endocrinologynetwork.com/view/fda-grants-dexcom-cgm-breakthrough-designation-for-in-hospital-use
44. Yeh T, Yeung M, Mendelsohn Curanaj FA. Managing patients with insulin pumps and continuous glucose monitors in the hospital: to wear or not to wear. Curr Diab Rep. 2021;21(2):7. doi:10.1007/s11892-021-01375-7
45. Medtronic announces FDA approval for MiniMed 770G insulin pump system. News release. September 21, 2020. Accessed August 30, 2022. https://bit.ly/3TyEna4
46. Tandem Diabetes Care announces commercial launch of the t:slim X2 insulin pump with Control-IQ technology in the United States. News release. January 15, 2020. Accessed August 30, 2022. https://investor.tandemdiabetes.com/news-releases/news-release-details/tandem-diabetes-care-announces-commercial-launch-tslim-x2-0
47. Garza M, Gutow H, Mahoney K. Omnipod 5 cleared by the FDA. Updated August 22, 2022. Accessed August 30, 2022.https://diatribe.org/omnipod-5-approved-fda
48. Boughton CK. Fully closed-loop insulin delivery—are we nearly there yet? Lancet Digit Health. 2021;3(11):e689-e690. doi:10.1016/s2589-7500(21)00218-1
49. Noor N, Kamboj MK, Triolo T, et al. Hybrid closed-loop systems and glycemic outcomes in children and adults with type 1 diabetes: real-world evidence from a U.S.-based multicenter collaborative. Diabetes Care. 2022;45(8):e118-e119. doi:10.2337/dc22-0329
50. Medtronic launches InPen with real-time Guardian Connect CGM data--the first integrated smart insulin pen for people with diabetes on MDI. News release. November 12, 2020. Accessed August 30, 2022. https://bit.ly/3CTSWPL
51. Bigfoot Biomedical receives FDA clearance for Bigfoot Unity Diabetes Management System, featuring first-of-its-kind smart pen caps for insulin pens used to treat type 1 and type 2 diabetes. News release. May 10, 2021. Accessed August 30, 2022. https://bit.ly/3BeyoAh
52. Vieira G. All about the CeQur Simplicity insulin patch. Updated May 24, 2022. Accessed August 30, 2022. https://beyondtype1.org/cequr-simplicity-insulin-patch/.
53. Messer LH, Tanenbaum ML, Cook PF, et al. Cost, hassle, and on-body experience: barriers to diabetes device use in adolescents and potential intervention targets. Diabetes Technol Ther. 2020;22(10):760-767. doi:10.1089/dia.2019.0509
54. Hilliard ME, Levy W, Anderson BJ, et al. Benefits and barriers of continuous glucose monitoring in young children with type 1 diabetes. Diabetes Technol Ther. 2019;21(9):493-498. doi:10.1089/dia.2019.0142
55. Dexcom G7 Release Delayed Until Late 2022. News release. August 8, 2022. Accessed September 7, 2022. https://diatribe.org/dexcom-g7-release-delayed-until-late-2022
56. Drucker DJ. Transforming type 1 diabetes: the next wave of innovation. Diabetologia. 2021;64(5):1059-1065. doi:10.1007/s00125-021-05396-5
57. Garg SK, Rodriguez E, Shah VN, Hirsch IB. New medications for the treatment of diabetes. Diabetes Technol Ther. 2022;24(S1):S190-S208. doi:10.1089/dia.2022.2513
58. Melton D. The promise of stem cell-derived islet replacement therapy. Diabetologia. 2021;64(5):1030-1036. doi:10.1007/s00125-020-05367-2
59. Danne T, Heinemann L, Bolinder J. New insulins, biosimilars, and insulin therapy. Diabetes Technol Ther. 2022;24(S1):S35-S57. doi:10.1089/dia.2022.2503
60. Kenney J. Insulin copay caps–a path to affordability. July 6, 2021. Accessed August 30, 2022.https://diatribechange.org/news/insulin-copay-caps-path-affordability
61. Glied SA, Zhu B. Not so sweet: insulin affordability over time. September 25, 2020. Accessed August 30, 2022. https://www.commonwealthfund.org/publications/issue-briefs/2020/sep/not-so-sweet-insulin-affordability-over-time
62. American Diabetes Association. Insulin and drug affordability. Accessed August 30, 2022. https://www.diabetes.org/advocacy/insulin-and-drug-affordability
63. Sullivan P. Chances for drug pricing, surprise billing action fade until November. March 24, 2020. Accessed August 30, 2022. https://thehill.com/policy/healthcare/489334-chances-for-drug-pricing-surprise-billing-action-fade-until-november/
64. Brown TD. How Medicare’s new Senior Savings Model makes insulin more affordable. June 4, 2020. Accessed August 30, 2022. https://www.diabetes.org/blog/how-medicares-new-senior-savings-model-makes-insulin-more-affordable
65. American Diabetes Association. ADA applauds the U.S. House of Representatives passage of the Affordable Insulin Now Act. News release. April 1, 2022. https://www.diabetes.org/newsroom/official-statement/2022/ada-applauds-us-house-of-representatives-passage-of-the-affordable-insulin-now-act
66. JDRF. Driving T1D cures during challenging times. 2022.
67. Medtronic announces ongoing initiatives to address health equity for people of color living with diabetes. News release. April 7, 2021. Access August 30, 2022. https://bit.ly/3KGTOZU
Your poop may hold the secret to long life
Lots of things can disrupt your gut health over the years. A high-sugar diet, stress, antibiotics – all are linked to bad changes in the gut microbiome, the microbes that live in your intestinal tract. And this can raise the risk of diseases.
It could be possible, scientists say, by having people take a sample of their own stool when they are young to be put back into their colons when they are older.
While the science to back this up isn’t quite there yet, some researchers are saying we shouldn’t wait. They are calling on existing stool banks to let people start banking their stool now, so it’s there for them to use if the science becomes available.
But how would that work?
First, you’d go to a stool bank and provide a fresh sample of your poop, which would be screened for diseases, washed, processed, and deposited into a long-term storage facility.
Then, down the road, if you get a condition such as inflammatory bowel disease, heart disease, or type 2 diabetes – or if you have a procedure that wipes out your microbiome, like a course of antibiotics or chemotherapy – doctors could use your preserved stool to “re-colonize” your gut, restoring it to its earlier, healthier state, said Scott Weiss, MD, professor of medicine at Harvard Medical School, Boston, and a coauthor of a recent paper on the topic. They would do that using fecal microbiota transplantation, or FMT.
Timing is everything. You’d want a sample from when you’re healthy – say, between the ages of 18 and 35, or before a chronic condition is likely, said Dr. Weiss. But if you’re still healthy into your late 30s, 40s, or even 50s, providing a sample then could still benefit you later in life.
If we could pull off a banking system like this, it could have the potential to treat autoimmune disease, inflammatory bowel disease, diabetes, obesity, and heart disease – or even reverse the effects of aging. How can we make this happen?
Stool banks of today
While stool banks do exist today, the samples inside are destined not for the original donors but rather for sick patients hoping to treat an illness. Using FMT, doctors transfer the fecal material to the patient’s colon, restoring helpful gut microbiota.
Some research shows FMT may help treat inflammatory bowel diseases, such as Crohn’s or ulcerative colitis. Animal studies suggest it could help treat obesity, lengthen lifespan, and reverse some effects of aging, such as age-related decline in brain function. Other clinical trials are looking into its potential as a cancer treatment, said Dr. Weiss.
But outside the lab, FMT is mainly used for one purpose: to treat Clostridioides difficile infection. It works even better than antibiotics, research shows.
But first you need to find a healthy donor, and that’s harder than you might think.
Finding healthy stool samples
Banking our bodily substances is nothing new. Blood banks, for example, are common throughout the United States, and cord blood banking – preserving blood from a baby’s umbilical cord to aid possible future medical needs of the child – is becoming more popular. Sperm donors are highly sought after, and doctors regularly transplant kidneys and bone marrow to patients in need.
So why are we so particular about poop?
Part of the reason may be because feces (like blood, for that matter) can harbor disease – which is why it’s so important to find healthy stool donors. Problem is, this can be surprisingly hard to do.
To donate fecal matter, people must go through a rigorous screening process, said Majdi Osman, MD, chief medical officer for OpenBiome, a nonprofit microbiome research organization.
Until recently, OpenBiome operated a stool donation program, though it has since shifted its focus to research. Potential donors were screened for diseases and mental health conditions, pathogens, and antibiotic resistance. The pass rate was less than 3%.
“We take a very cautious approach because the association between diseases and the microbiome is still being understood,” Dr. Osman said.
FMT also carries risks – though so far, they seem mild. Side effects include mild diarrhea, nausea, belly pain, and fatigue. (The reason? Even the healthiest donor stool may not mix perfectly with your own.)
That’s where the idea of using your own stool comes in, said Yang-Yu Liu, PhD, a Harvard researcher who studies the microbiome and the lead author of the paper mentioned above. It’s not just more appealing but may also be a better “match” for your body.
Should you bank your stool?
While the researchers say we have reason to be optimistic about the future, it’s important to remember that many challenges remain. FMT is early in development, and there’s a lot about the microbiome we still don’t know.
There’s no guarantee, for example, that restoring a person’s microbiome to its formerly disease-free state will keep diseases at bay forever, said Dr. Weiss. If your genes raise your odds of having Crohn’s, for instance, it’s possible the disease could come back.
We also don’t know how long stool samples can be preserved, said Dr. Liu. Stool banks currently store fecal matter for 1 or 2 years, not decades. To protect the proteins and DNA structures for that long, samples would likely need to be stashed at the liquid nitrogen storage temperature of –196° C. (Currently, samples are stored at about –80° C.) Even then, testing would be needed to confirm if the fragile microorganisms in the stool can survive.
This raises another question: Who’s going to regulate all this?
The FDA regulates the use of FMT as a drug for the treatment of C. diff, but as Dr. Liu pointed out, many gastroenterologists consider the gut microbiota an organ. In that case, human fecal matter could be regulated the same way blood, bone, or even egg cells are.
Cord blood banking may be a helpful model, Dr. Liu said.
“We don’t have to start from scratch.”
Then there’s the question of cost. Cord blood banks could be a point of reference for that too, the researchers say. They charge about $1,500 to $2,820 for the first collection and processing, plus a yearly storage fee of $185 to $370.
Despite the unknowns, one thing is for sure: The interest in fecal banking is real – and growing. At least one microbiome firm, Cordlife Group Limited, based in Singapore, announced that it has started to allow people to bank their stool for future use.
“More people should talk about it and think about it,” said Dr. Liu.
A version of this article first appeared on WebMD.com.
Lots of things can disrupt your gut health over the years. A high-sugar diet, stress, antibiotics – all are linked to bad changes in the gut microbiome, the microbes that live in your intestinal tract. And this can raise the risk of diseases.
It could be possible, scientists say, by having people take a sample of their own stool when they are young to be put back into their colons when they are older.
While the science to back this up isn’t quite there yet, some researchers are saying we shouldn’t wait. They are calling on existing stool banks to let people start banking their stool now, so it’s there for them to use if the science becomes available.
But how would that work?
First, you’d go to a stool bank and provide a fresh sample of your poop, which would be screened for diseases, washed, processed, and deposited into a long-term storage facility.
Then, down the road, if you get a condition such as inflammatory bowel disease, heart disease, or type 2 diabetes – or if you have a procedure that wipes out your microbiome, like a course of antibiotics or chemotherapy – doctors could use your preserved stool to “re-colonize” your gut, restoring it to its earlier, healthier state, said Scott Weiss, MD, professor of medicine at Harvard Medical School, Boston, and a coauthor of a recent paper on the topic. They would do that using fecal microbiota transplantation, or FMT.
Timing is everything. You’d want a sample from when you’re healthy – say, between the ages of 18 and 35, or before a chronic condition is likely, said Dr. Weiss. But if you’re still healthy into your late 30s, 40s, or even 50s, providing a sample then could still benefit you later in life.
If we could pull off a banking system like this, it could have the potential to treat autoimmune disease, inflammatory bowel disease, diabetes, obesity, and heart disease – or even reverse the effects of aging. How can we make this happen?
Stool banks of today
While stool banks do exist today, the samples inside are destined not for the original donors but rather for sick patients hoping to treat an illness. Using FMT, doctors transfer the fecal material to the patient’s colon, restoring helpful gut microbiota.
Some research shows FMT may help treat inflammatory bowel diseases, such as Crohn’s or ulcerative colitis. Animal studies suggest it could help treat obesity, lengthen lifespan, and reverse some effects of aging, such as age-related decline in brain function. Other clinical trials are looking into its potential as a cancer treatment, said Dr. Weiss.
But outside the lab, FMT is mainly used for one purpose: to treat Clostridioides difficile infection. It works even better than antibiotics, research shows.
But first you need to find a healthy donor, and that’s harder than you might think.
Finding healthy stool samples
Banking our bodily substances is nothing new. Blood banks, for example, are common throughout the United States, and cord blood banking – preserving blood from a baby’s umbilical cord to aid possible future medical needs of the child – is becoming more popular. Sperm donors are highly sought after, and doctors regularly transplant kidneys and bone marrow to patients in need.
So why are we so particular about poop?
Part of the reason may be because feces (like blood, for that matter) can harbor disease – which is why it’s so important to find healthy stool donors. Problem is, this can be surprisingly hard to do.
To donate fecal matter, people must go through a rigorous screening process, said Majdi Osman, MD, chief medical officer for OpenBiome, a nonprofit microbiome research organization.
Until recently, OpenBiome operated a stool donation program, though it has since shifted its focus to research. Potential donors were screened for diseases and mental health conditions, pathogens, and antibiotic resistance. The pass rate was less than 3%.
“We take a very cautious approach because the association between diseases and the microbiome is still being understood,” Dr. Osman said.
FMT also carries risks – though so far, they seem mild. Side effects include mild diarrhea, nausea, belly pain, and fatigue. (The reason? Even the healthiest donor stool may not mix perfectly with your own.)
That’s where the idea of using your own stool comes in, said Yang-Yu Liu, PhD, a Harvard researcher who studies the microbiome and the lead author of the paper mentioned above. It’s not just more appealing but may also be a better “match” for your body.
Should you bank your stool?
While the researchers say we have reason to be optimistic about the future, it’s important to remember that many challenges remain. FMT is early in development, and there’s a lot about the microbiome we still don’t know.
There’s no guarantee, for example, that restoring a person’s microbiome to its formerly disease-free state will keep diseases at bay forever, said Dr. Weiss. If your genes raise your odds of having Crohn’s, for instance, it’s possible the disease could come back.
We also don’t know how long stool samples can be preserved, said Dr. Liu. Stool banks currently store fecal matter for 1 or 2 years, not decades. To protect the proteins and DNA structures for that long, samples would likely need to be stashed at the liquid nitrogen storage temperature of –196° C. (Currently, samples are stored at about –80° C.) Even then, testing would be needed to confirm if the fragile microorganisms in the stool can survive.
This raises another question: Who’s going to regulate all this?
The FDA regulates the use of FMT as a drug for the treatment of C. diff, but as Dr. Liu pointed out, many gastroenterologists consider the gut microbiota an organ. In that case, human fecal matter could be regulated the same way blood, bone, or even egg cells are.
Cord blood banking may be a helpful model, Dr. Liu said.
“We don’t have to start from scratch.”
Then there’s the question of cost. Cord blood banks could be a point of reference for that too, the researchers say. They charge about $1,500 to $2,820 for the first collection and processing, plus a yearly storage fee of $185 to $370.
Despite the unknowns, one thing is for sure: The interest in fecal banking is real – and growing. At least one microbiome firm, Cordlife Group Limited, based in Singapore, announced that it has started to allow people to bank their stool for future use.
“More people should talk about it and think about it,” said Dr. Liu.
A version of this article first appeared on WebMD.com.
Lots of things can disrupt your gut health over the years. A high-sugar diet, stress, antibiotics – all are linked to bad changes in the gut microbiome, the microbes that live in your intestinal tract. And this can raise the risk of diseases.
It could be possible, scientists say, by having people take a sample of their own stool when they are young to be put back into their colons when they are older.
While the science to back this up isn’t quite there yet, some researchers are saying we shouldn’t wait. They are calling on existing stool banks to let people start banking their stool now, so it’s there for them to use if the science becomes available.
But how would that work?
First, you’d go to a stool bank and provide a fresh sample of your poop, which would be screened for diseases, washed, processed, and deposited into a long-term storage facility.
Then, down the road, if you get a condition such as inflammatory bowel disease, heart disease, or type 2 diabetes – or if you have a procedure that wipes out your microbiome, like a course of antibiotics or chemotherapy – doctors could use your preserved stool to “re-colonize” your gut, restoring it to its earlier, healthier state, said Scott Weiss, MD, professor of medicine at Harvard Medical School, Boston, and a coauthor of a recent paper on the topic. They would do that using fecal microbiota transplantation, or FMT.
Timing is everything. You’d want a sample from when you’re healthy – say, between the ages of 18 and 35, or before a chronic condition is likely, said Dr. Weiss. But if you’re still healthy into your late 30s, 40s, or even 50s, providing a sample then could still benefit you later in life.
If we could pull off a banking system like this, it could have the potential to treat autoimmune disease, inflammatory bowel disease, diabetes, obesity, and heart disease – or even reverse the effects of aging. How can we make this happen?
Stool banks of today
While stool banks do exist today, the samples inside are destined not for the original donors but rather for sick patients hoping to treat an illness. Using FMT, doctors transfer the fecal material to the patient’s colon, restoring helpful gut microbiota.
Some research shows FMT may help treat inflammatory bowel diseases, such as Crohn’s or ulcerative colitis. Animal studies suggest it could help treat obesity, lengthen lifespan, and reverse some effects of aging, such as age-related decline in brain function. Other clinical trials are looking into its potential as a cancer treatment, said Dr. Weiss.
But outside the lab, FMT is mainly used for one purpose: to treat Clostridioides difficile infection. It works even better than antibiotics, research shows.
But first you need to find a healthy donor, and that’s harder than you might think.
Finding healthy stool samples
Banking our bodily substances is nothing new. Blood banks, for example, are common throughout the United States, and cord blood banking – preserving blood from a baby’s umbilical cord to aid possible future medical needs of the child – is becoming more popular. Sperm donors are highly sought after, and doctors regularly transplant kidneys and bone marrow to patients in need.
So why are we so particular about poop?
Part of the reason may be because feces (like blood, for that matter) can harbor disease – which is why it’s so important to find healthy stool donors. Problem is, this can be surprisingly hard to do.
To donate fecal matter, people must go through a rigorous screening process, said Majdi Osman, MD, chief medical officer for OpenBiome, a nonprofit microbiome research organization.
Until recently, OpenBiome operated a stool donation program, though it has since shifted its focus to research. Potential donors were screened for diseases and mental health conditions, pathogens, and antibiotic resistance. The pass rate was less than 3%.
“We take a very cautious approach because the association between diseases and the microbiome is still being understood,” Dr. Osman said.
FMT also carries risks – though so far, they seem mild. Side effects include mild diarrhea, nausea, belly pain, and fatigue. (The reason? Even the healthiest donor stool may not mix perfectly with your own.)
That’s where the idea of using your own stool comes in, said Yang-Yu Liu, PhD, a Harvard researcher who studies the microbiome and the lead author of the paper mentioned above. It’s not just more appealing but may also be a better “match” for your body.
Should you bank your stool?
While the researchers say we have reason to be optimistic about the future, it’s important to remember that many challenges remain. FMT is early in development, and there’s a lot about the microbiome we still don’t know.
There’s no guarantee, for example, that restoring a person’s microbiome to its formerly disease-free state will keep diseases at bay forever, said Dr. Weiss. If your genes raise your odds of having Crohn’s, for instance, it’s possible the disease could come back.
We also don’t know how long stool samples can be preserved, said Dr. Liu. Stool banks currently store fecal matter for 1 or 2 years, not decades. To protect the proteins and DNA structures for that long, samples would likely need to be stashed at the liquid nitrogen storage temperature of –196° C. (Currently, samples are stored at about –80° C.) Even then, testing would be needed to confirm if the fragile microorganisms in the stool can survive.
This raises another question: Who’s going to regulate all this?
The FDA regulates the use of FMT as a drug for the treatment of C. diff, but as Dr. Liu pointed out, many gastroenterologists consider the gut microbiota an organ. In that case, human fecal matter could be regulated the same way blood, bone, or even egg cells are.
Cord blood banking may be a helpful model, Dr. Liu said.
“We don’t have to start from scratch.”
Then there’s the question of cost. Cord blood banks could be a point of reference for that too, the researchers say. They charge about $1,500 to $2,820 for the first collection and processing, plus a yearly storage fee of $185 to $370.
Despite the unknowns, one thing is for sure: The interest in fecal banking is real – and growing. At least one microbiome firm, Cordlife Group Limited, based in Singapore, announced that it has started to allow people to bank their stool for future use.
“More people should talk about it and think about it,” said Dr. Liu.
A version of this article first appeared on WebMD.com.
Weight gain during pregnancy may play role in child ADHD risk
Obesity in women of reproductive age has emerged as one of the main risk factors associated with neonatal complications and long-term neuropsychiatric consequences in offspring, including attention-deficit/hyperactivity disorder.
Research has also linked pregestational diabetes and gestational diabetes mellitus (GDM) to an increased risk for ADHD in offspring. Now, an observational study of 1,036 singleton births at one hospital between 1998 and 2008 suggests that in the presence of GDM, maternal obesity combined with excessive weight gain during pregnancy may be jointly associated with increased risk of offspring ADHD. The median follow-up was 17.7 years.
Maternal obesity was independently associated with ADHD (adjusted hazard ratio, 1.66; 95% confidence interval: 1.07-2.60), but excessive weight gain during pregnancy and maternal overweight were not, reported Verónica Perea, MD, PhD, of the Hospital Universitari Mútua de Terrassa, Barcelona, and colleagues in the Journal of Clinical Endocrinology & Metabolism.
However, in women with pregestation obesity who gained more weight than recommended by the National Academy of Medicine (NAM), the risk of offspring ADHD was higher, compared with women of normal weight whose pregnancy weight stayed within NAM guidelines (adjusted hazard ratio, 2.13; 95% confidence interval: 1.14-4.01).
“The results of this study suggest that the negative repercussions of excessive weight gain on children within the setting of a high-risk population with GDM and obesity were not only observed during the prenatal period but also years later with a development of ADHD,” the researchers wrote.
The study also showed that when maternal weight gain did not exceed NAM guidelines, maternal obesity was no longer independently associated with ADHD in offspring (aHR, 1.36; 95% CI: 0.78-2.36). This finding conflicts with earlier studies focusing primarily on the role of pregestational maternal weight, the researchers said. A 2018 nationwide Finnish cohort study in newborns showed an increased long-term risk of ADHD in those born to women with GDM, compared with the nondiabetic population. This long-term risk of ADHD increased in the presence of pregestational obesity (HR, 1.64).
Similarly, evidence from systematic reviews and meta-analyses has demonstrated that antenatal lifestyle interventions to prevent excessive weight gain during pregnancy were associated with a reduction in adverse pregnancy outcomes. However, evidence on offspring mental health was lacking, especially in high-risk pregnancies with gestational diabetes, the study authors said.
Although causal inferences can’t be drawn from the current observational study, “it seems that the higher risk [of ADHD] observed would be explained by the role of gestational weight gain during the antenatal period,” Dr. Perea said in an interview. Importantly, the study highlights a window of opportunity for promoting healthy weight gain during pregnancy, Dr. Perea said. ”This should be a priority in the current management of gestation.”
Fatima Cody Stanford, MD, MPH, an associate professor of medicine and pediatrics at Harvard Medical School, Boston, agreed. “I think one of the key issues is that there’s very little attention paid to how weight gain is regulated during pregnancy,” she said in an interview. On many other points, however, Dr. Stanford, who is a specialist in obesity medicine at Massachusetts General Hospital Weight Center, did not agree.
The association between ADHD and obesity has already been well established by a 2019 meta-analysis and systematic review of studies over the last 10 years, she emphasized. “These studies were able to show a much stronger association between maternal obesity and ADHD in offspring because they were powered to detect differences.”
The current study does not say “anything new or novel,” Dr. Stanford added. “Maternal obesity and the association with an increased risk of ADHD in offspring is the main issue. I don’t think there was any appreciable increase when weight gain during pregnancy was factored in. It’s mild at best.”
Eran Bornstein, MD, vice-chair of obstetrics and gynecology at Lenox Hill Hospital, New York, expressed a similar point of view. Although the study findings “add to the current literature,” they should be interpreted “cautiously,” Dr. Bornstein said in an interview.
The size of the effect on ADHD risk attributable to maternal weight gain during pregnancy “was not clear,” he said. “Cohort studies of this sort are excellent for finding associations which help us generate the hypothesis, but this doesn’t demonstrate a cause and effect or a magnitude for this effect.”
Physicians should follow cumulative data suggesting that maternal obesity is associated with a number of pregnancy complications and neonatal outcomes in women with and without diabetes, Dr. Bornstein suggested. “Optimizing maternal weight prior to pregnancy and adhering to recommendations regarding weight gain has the potential to improve some of these outcomes.”
Treating obesity prior to conception mitigates GDM risk, agreed Dr. Stanford. “The issue,” she explained, “is that all of the drugs approved for the treatment of obesity are contraindicated in pregnancy and lifestyle modification fails in 96% of cases, even when there is no pregnancy.” Drugs such as metformin are being used off-label to treat obesity and to safely manage gestational weight gain, she said. “Those of us who practice obesity medicine know that metformin can be safely used throughout pregnancy with no harm to the fetus.”
This study was partially funded by Fundació Docència i Recerca MútuaTerrassa. Dr. Perea and study coauthors reporting have no conflicts of interest. Dr. Stanford disclosed relationships with Novo Nordisk, Eli Lilly, Boehringer Ingelheim, Gelesis, Pfizer, Currax, and Rhythm. Dr. Bornstein reported having no conflicts of interest.
This story was updated on 11/7/2022.
Obesity in women of reproductive age has emerged as one of the main risk factors associated with neonatal complications and long-term neuropsychiatric consequences in offspring, including attention-deficit/hyperactivity disorder.
Research has also linked pregestational diabetes and gestational diabetes mellitus (GDM) to an increased risk for ADHD in offspring. Now, an observational study of 1,036 singleton births at one hospital between 1998 and 2008 suggests that in the presence of GDM, maternal obesity combined with excessive weight gain during pregnancy may be jointly associated with increased risk of offspring ADHD. The median follow-up was 17.7 years.
Maternal obesity was independently associated with ADHD (adjusted hazard ratio, 1.66; 95% confidence interval: 1.07-2.60), but excessive weight gain during pregnancy and maternal overweight were not, reported Verónica Perea, MD, PhD, of the Hospital Universitari Mútua de Terrassa, Barcelona, and colleagues in the Journal of Clinical Endocrinology & Metabolism.
However, in women with pregestation obesity who gained more weight than recommended by the National Academy of Medicine (NAM), the risk of offspring ADHD was higher, compared with women of normal weight whose pregnancy weight stayed within NAM guidelines (adjusted hazard ratio, 2.13; 95% confidence interval: 1.14-4.01).
“The results of this study suggest that the negative repercussions of excessive weight gain on children within the setting of a high-risk population with GDM and obesity were not only observed during the prenatal period but also years later with a development of ADHD,” the researchers wrote.
The study also showed that when maternal weight gain did not exceed NAM guidelines, maternal obesity was no longer independently associated with ADHD in offspring (aHR, 1.36; 95% CI: 0.78-2.36). This finding conflicts with earlier studies focusing primarily on the role of pregestational maternal weight, the researchers said. A 2018 nationwide Finnish cohort study in newborns showed an increased long-term risk of ADHD in those born to women with GDM, compared with the nondiabetic population. This long-term risk of ADHD increased in the presence of pregestational obesity (HR, 1.64).
Similarly, evidence from systematic reviews and meta-analyses has demonstrated that antenatal lifestyle interventions to prevent excessive weight gain during pregnancy were associated with a reduction in adverse pregnancy outcomes. However, evidence on offspring mental health was lacking, especially in high-risk pregnancies with gestational diabetes, the study authors said.
Although causal inferences can’t be drawn from the current observational study, “it seems that the higher risk [of ADHD] observed would be explained by the role of gestational weight gain during the antenatal period,” Dr. Perea said in an interview. Importantly, the study highlights a window of opportunity for promoting healthy weight gain during pregnancy, Dr. Perea said. ”This should be a priority in the current management of gestation.”
Fatima Cody Stanford, MD, MPH, an associate professor of medicine and pediatrics at Harvard Medical School, Boston, agreed. “I think one of the key issues is that there’s very little attention paid to how weight gain is regulated during pregnancy,” she said in an interview. On many other points, however, Dr. Stanford, who is a specialist in obesity medicine at Massachusetts General Hospital Weight Center, did not agree.
The association between ADHD and obesity has already been well established by a 2019 meta-analysis and systematic review of studies over the last 10 years, she emphasized. “These studies were able to show a much stronger association between maternal obesity and ADHD in offspring because they were powered to detect differences.”
The current study does not say “anything new or novel,” Dr. Stanford added. “Maternal obesity and the association with an increased risk of ADHD in offspring is the main issue. I don’t think there was any appreciable increase when weight gain during pregnancy was factored in. It’s mild at best.”
Eran Bornstein, MD, vice-chair of obstetrics and gynecology at Lenox Hill Hospital, New York, expressed a similar point of view. Although the study findings “add to the current literature,” they should be interpreted “cautiously,” Dr. Bornstein said in an interview.
The size of the effect on ADHD risk attributable to maternal weight gain during pregnancy “was not clear,” he said. “Cohort studies of this sort are excellent for finding associations which help us generate the hypothesis, but this doesn’t demonstrate a cause and effect or a magnitude for this effect.”
Physicians should follow cumulative data suggesting that maternal obesity is associated with a number of pregnancy complications and neonatal outcomes in women with and without diabetes, Dr. Bornstein suggested. “Optimizing maternal weight prior to pregnancy and adhering to recommendations regarding weight gain has the potential to improve some of these outcomes.”
Treating obesity prior to conception mitigates GDM risk, agreed Dr. Stanford. “The issue,” she explained, “is that all of the drugs approved for the treatment of obesity are contraindicated in pregnancy and lifestyle modification fails in 96% of cases, even when there is no pregnancy.” Drugs such as metformin are being used off-label to treat obesity and to safely manage gestational weight gain, she said. “Those of us who practice obesity medicine know that metformin can be safely used throughout pregnancy with no harm to the fetus.”
This study was partially funded by Fundació Docència i Recerca MútuaTerrassa. Dr. Perea and study coauthors reporting have no conflicts of interest. Dr. Stanford disclosed relationships with Novo Nordisk, Eli Lilly, Boehringer Ingelheim, Gelesis, Pfizer, Currax, and Rhythm. Dr. Bornstein reported having no conflicts of interest.
This story was updated on 11/7/2022.
Obesity in women of reproductive age has emerged as one of the main risk factors associated with neonatal complications and long-term neuropsychiatric consequences in offspring, including attention-deficit/hyperactivity disorder.
Research has also linked pregestational diabetes and gestational diabetes mellitus (GDM) to an increased risk for ADHD in offspring. Now, an observational study of 1,036 singleton births at one hospital between 1998 and 2008 suggests that in the presence of GDM, maternal obesity combined with excessive weight gain during pregnancy may be jointly associated with increased risk of offspring ADHD. The median follow-up was 17.7 years.
Maternal obesity was independently associated with ADHD (adjusted hazard ratio, 1.66; 95% confidence interval: 1.07-2.60), but excessive weight gain during pregnancy and maternal overweight were not, reported Verónica Perea, MD, PhD, of the Hospital Universitari Mútua de Terrassa, Barcelona, and colleagues in the Journal of Clinical Endocrinology & Metabolism.
However, in women with pregestation obesity who gained more weight than recommended by the National Academy of Medicine (NAM), the risk of offspring ADHD was higher, compared with women of normal weight whose pregnancy weight stayed within NAM guidelines (adjusted hazard ratio, 2.13; 95% confidence interval: 1.14-4.01).
“The results of this study suggest that the negative repercussions of excessive weight gain on children within the setting of a high-risk population with GDM and obesity were not only observed during the prenatal period but also years later with a development of ADHD,” the researchers wrote.
The study also showed that when maternal weight gain did not exceed NAM guidelines, maternal obesity was no longer independently associated with ADHD in offspring (aHR, 1.36; 95% CI: 0.78-2.36). This finding conflicts with earlier studies focusing primarily on the role of pregestational maternal weight, the researchers said. A 2018 nationwide Finnish cohort study in newborns showed an increased long-term risk of ADHD in those born to women with GDM, compared with the nondiabetic population. This long-term risk of ADHD increased in the presence of pregestational obesity (HR, 1.64).
Similarly, evidence from systematic reviews and meta-analyses has demonstrated that antenatal lifestyle interventions to prevent excessive weight gain during pregnancy were associated with a reduction in adverse pregnancy outcomes. However, evidence on offspring mental health was lacking, especially in high-risk pregnancies with gestational diabetes, the study authors said.
Although causal inferences can’t be drawn from the current observational study, “it seems that the higher risk [of ADHD] observed would be explained by the role of gestational weight gain during the antenatal period,” Dr. Perea said in an interview. Importantly, the study highlights a window of opportunity for promoting healthy weight gain during pregnancy, Dr. Perea said. ”This should be a priority in the current management of gestation.”
Fatima Cody Stanford, MD, MPH, an associate professor of medicine and pediatrics at Harvard Medical School, Boston, agreed. “I think one of the key issues is that there’s very little attention paid to how weight gain is regulated during pregnancy,” she said in an interview. On many other points, however, Dr. Stanford, who is a specialist in obesity medicine at Massachusetts General Hospital Weight Center, did not agree.
The association between ADHD and obesity has already been well established by a 2019 meta-analysis and systematic review of studies over the last 10 years, she emphasized. “These studies were able to show a much stronger association between maternal obesity and ADHD in offspring because they were powered to detect differences.”
The current study does not say “anything new or novel,” Dr. Stanford added. “Maternal obesity and the association with an increased risk of ADHD in offspring is the main issue. I don’t think there was any appreciable increase when weight gain during pregnancy was factored in. It’s mild at best.”
Eran Bornstein, MD, vice-chair of obstetrics and gynecology at Lenox Hill Hospital, New York, expressed a similar point of view. Although the study findings “add to the current literature,” they should be interpreted “cautiously,” Dr. Bornstein said in an interview.
The size of the effect on ADHD risk attributable to maternal weight gain during pregnancy “was not clear,” he said. “Cohort studies of this sort are excellent for finding associations which help us generate the hypothesis, but this doesn’t demonstrate a cause and effect or a magnitude for this effect.”
Physicians should follow cumulative data suggesting that maternal obesity is associated with a number of pregnancy complications and neonatal outcomes in women with and without diabetes, Dr. Bornstein suggested. “Optimizing maternal weight prior to pregnancy and adhering to recommendations regarding weight gain has the potential to improve some of these outcomes.”
Treating obesity prior to conception mitigates GDM risk, agreed Dr. Stanford. “The issue,” she explained, “is that all of the drugs approved for the treatment of obesity are contraindicated in pregnancy and lifestyle modification fails in 96% of cases, even when there is no pregnancy.” Drugs such as metformin are being used off-label to treat obesity and to safely manage gestational weight gain, she said. “Those of us who practice obesity medicine know that metformin can be safely used throughout pregnancy with no harm to the fetus.”
This study was partially funded by Fundació Docència i Recerca MútuaTerrassa. Dr. Perea and study coauthors reporting have no conflicts of interest. Dr. Stanford disclosed relationships with Novo Nordisk, Eli Lilly, Boehringer Ingelheim, Gelesis, Pfizer, Currax, and Rhythm. Dr. Bornstein reported having no conflicts of interest.
This story was updated on 11/7/2022.
The Journal of Clinical Endocrinology & Metabolism
57-year-old man • type 2 diabetes • neuropathy • bilateral foot blisters • Dx?
THE CASE
A 57-year-old man with type 2 diabetes, hyperlipidemia, and obesity presented to the emergency department (ED) for bilateral foot blisters, both of which appeared 1 day prior to evaluation. The patient’s history also included right-side Charcot foot diagnosed 4 years earlier and right foot osteomyelitis diagnosed 2 years prior. He had ongoing neuropathy in both feet but denied any significant pain.
The patient wore orthotics daily and he’d had new orthotics made 6 months prior; however, a recent COVID-19 diagnosis and prolonged hospital stay resulted in a 30-pound weight loss and decreased swelling in his ankles. He acquired new shoes 2 weeks prior to ED presentation.
Physical examination revealed large blisters along the medial aspect of the patient’s feet, with both hemorrhagic and serous fluid-filled bullae. The lesions were flaccid but intact, without drainage or surrounding erythema, warmth, or tenderness. The blister on the left foot measured 8 x 5 cm and extended from the great toe to mid-arch (FIGURE), while the one on the right foot measured 8 x 3 cm and extended from the great toe to the base of the proximal arch. Sensation was decreased in the bilateral first and second digits but unchanged from prior documented exams. Bilateral dorsalis pedis pulses were normal.
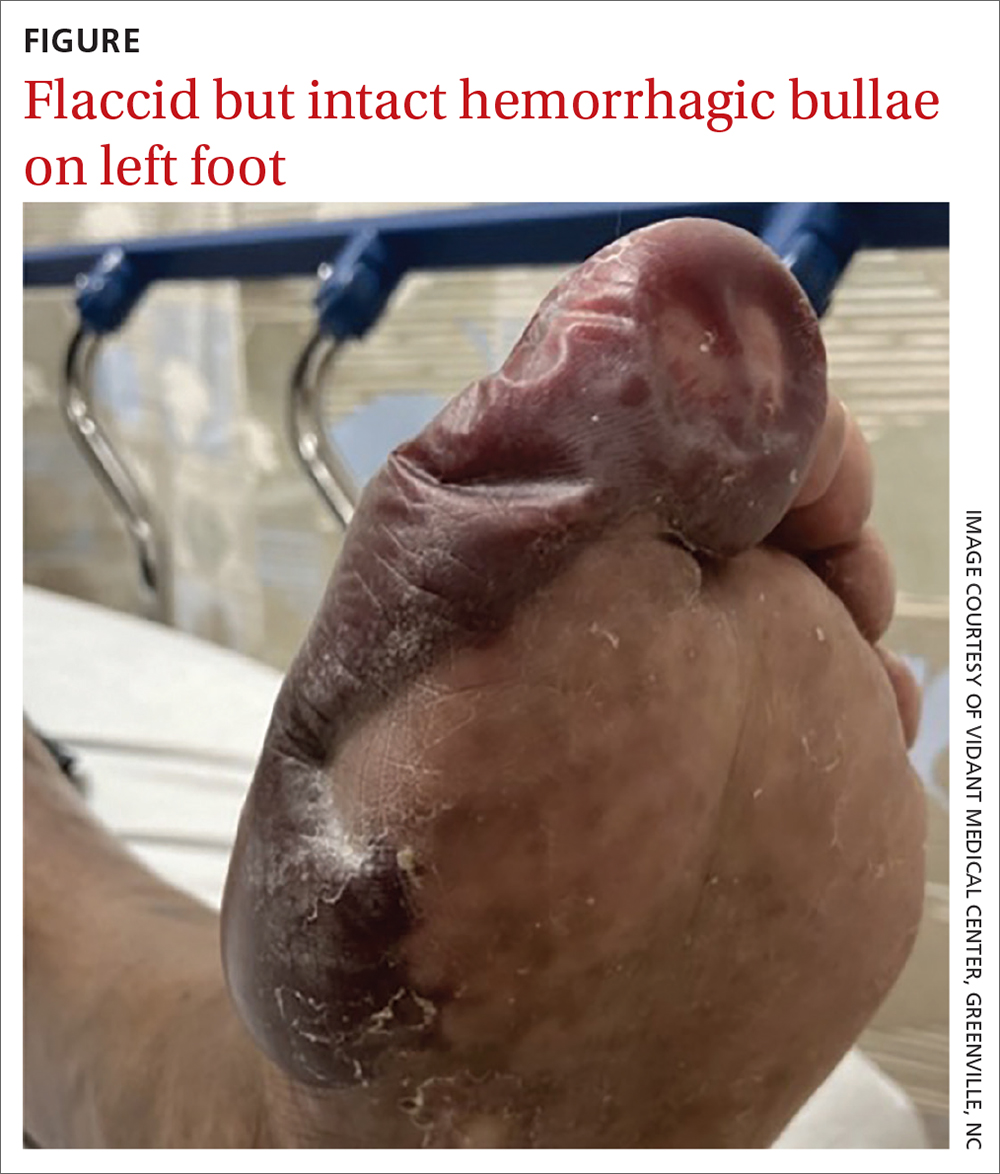
Work-up included imaging and lab work. The patient’s complete blood count was normal, as were his erythrocyte sedimentation rate and C-reactive protein level. Radiographs of the right foot were normal, but those of the left foot were concerning, although inconclusive, for osteomyelitis. Further evaluation with magnetic resonance imaging of his left foot revealed a deformity of the first digit with some subchondral signal change that was thought to be posttraumatic or degenerative, but unlikely osteomyelitis.
THE DIAGNOSIS
Podiatry was consulted for blister management. Based on atraumatic history, rapid appearance, location of blisters, unremarkable lab work and imaging, and concurrent diabetes, the patient received a diagnosis of bilateral bullous diabeticorum (BD).
DISCUSSION
Roughly one-third of patients with diabetes will experience some cutaneous adverse effect because of the disease.1 Common iterations include acanthosis nigricans, rash, or even infection.2 BD is a rare bullous skin lesion that occurs in patients with diabetes; it has a reported annual incidence of 0.16% and may be underdiagnosed.1
Cases of BD have been described both in patients with longstanding diabetes and in those newly diagnosed, although the former group is more often affected.1 BD is reported more frequently in males than females, at a ratio of 2:1.1,3 Patients ages 17 to 80 years (average age, 55 years) have received a diagnosis of BD.1 Most affected patients will have a concomitant peripheral neuropathy and sometimes nephropathy or retinopathy.1
Continue to: The etiology of BD...
The etiology of BD is unclear but appears to be multifactorial. Hypotheses suggest that there’s a link to neuropathy/nephropathy, excessive exposure to ultraviolet light, or a vascular cause secondary to hyaline deposition in the capillary walls.4,5
What you’ll see at presentation
The typical manifestation of BD is the rapid appearance of tense blisters, which may occur overnight or even within hours.1 They are usually painless; common locations include the feet, distal legs, hands, and forearms.1,5 The bullae can be serous or hemorrhagic.1
Most notable in the patient’s history will be a lack of trauma or injury to the area.1 Although A1C values do not correlate with blister formation, patients with hypoglycemic episodes and highly varying blood glucose values seem to have higher rates of occurrence.1
Other sources of blistering must be ruled out
The diagnosis of BD is clinical and based on history, exam, and exclusion of other bullous diagnoses.6 A key clue in the history is the spontaneous and rapid onset without associated trauma in a patient with diabetes.6 Direct immunofluorescence, although nonspecific, can be helpful to rule out other disorders (such as porphyria cutanea tarda and bullous pemphigoid) if the history and exam are inconclusive. Direct and indirect immunofluorescence is typically negative in BD.4,6
The differential diagnosis includes other conditions that involve bullae—such as frictional bullae, bullous pemphigoid, and bullous systemic lupus erythematosus—as well as porphyria, erythema multiforme, insect bites, or even fixed drug eruption.2,7
Continue to: Porphyria
Porphyria tends to develop on the hands, whereas BD most commonly occurs on the feet.5
Erythema multiforme typically includes inflammatory skin changes.5
Trauma or fixed drug eruption as a cause of blistering lesions would be revealed during history taking.
Considerations for treatment and follow-up
Without treatment, blisters often self-resolve in 2 to 6 weeks, but there is high likelihood of recurrence.6,8 There is no consensus on treatment, although a typical course of action is to deroof the blister and examine the area to rule out infection.6 The wound is then covered with wet-to-dry gauze that is changed regularly. If there is suspicion for or signs of underlying infection, such as an ulcer or skin necrosis, antibiotics should be included in the treatment plan.7
Additional considerations. Patients will often need therapeutic footwear if the blisters are located on the feet. Given the higher prevalence of microvascular complications in patients with diabetes who develop BD, routine ophthalmologic examination and renal function testing to monitor for microalbuminuria are recommended.5
Our patient underwent bedside incision and drainage and was discharged home with appropriate wound care and follow-up.
THE TAKEAWAY
BD cases may be underdiagnosed in clinical practice, perhaps due to patients not seeking help for a seemingly nonthreatening condition or lack of clinician recognition that bullae are related to a patient’s diabetes status. Prompt recognition and proper wound care are important to prevent poor outcomes, such as ulceration or necrosis.
CORRESPONDENCE
Kathleen S. Kinderwater, MD, 101 Heart Drive, Greenville, NC 27834; salmondk19@ecu.edu
1. Larsen K, Jensen T, Karlsmark T, et al. Incidence of bullosis diabeticorum—a controversial cause of chronic foot ulceration. Int Wound J. 2008;5:591-596. doi: 10.1111/j.1742-481X.2008.00476.x
2. Lipsky BA, Baker PD, Ahroni JH. Diabetic bullae: 12 cases of a purportedly rare cutaneous disorder. Int J Dermatol. 2000;39:196-200. doi: 10.1046/j.1365-4362.2000.00947.x
3. Gupta V, Gulati N, Bahl J, et al. Bullosis diabeticorum: rare presentation in a common disease. Case Rep Endocrinol. 2014;2014:862912.
4. Sonani H, Abdul Salim S, Garla VV, et al. Bullosis diabeticorum: a rare presentation with immunoglobulin G (IgG) deposition related vasculopathy. Case report and focused review. Am J Case Rep. 2018;19:52-56. doi: 10.12659/ajcr.905452
5. Chouk C, Litaiem N. Bullosis diabeticorum. StatPearls [Internet]. Updated June 5, 2021. Accessed July 14, 2022. www.ncbi.nlm.nih.gov/books/NBK539872/
6. Chatterjee D, Radotra A, Radotra BD, et al. Bullous diabeticorum: a rare blistering manifestation of diabetes. Indian Dermatol Online J. 2017;8:274-275. doi: 10.4103/idoj.IDOJ_340_16
7. Kansal NK, Anuragi RP. Bullous lesions in diabetes mellitus: bullous diabeticorum (diabetic bulla). BMJ Case Rep. 2020;13:e238617. doi: 10.1136/bcr-2020-238617
8. Bello F, Samaila OM, Lawal Y, et al. 2 cases of bullosis diabeticorum following long-distance journeys by road: a report of 2 cases. Case Rep Endocrinol. 2012;2012:367218. doi: 10.1155/2012/367218
THE CASE
A 57-year-old man with type 2 diabetes, hyperlipidemia, and obesity presented to the emergency department (ED) for bilateral foot blisters, both of which appeared 1 day prior to evaluation. The patient’s history also included right-side Charcot foot diagnosed 4 years earlier and right foot osteomyelitis diagnosed 2 years prior. He had ongoing neuropathy in both feet but denied any significant pain.
The patient wore orthotics daily and he’d had new orthotics made 6 months prior; however, a recent COVID-19 diagnosis and prolonged hospital stay resulted in a 30-pound weight loss and decreased swelling in his ankles. He acquired new shoes 2 weeks prior to ED presentation.
Physical examination revealed large blisters along the medial aspect of the patient’s feet, with both hemorrhagic and serous fluid-filled bullae. The lesions were flaccid but intact, without drainage or surrounding erythema, warmth, or tenderness. The blister on the left foot measured 8 x 5 cm and extended from the great toe to mid-arch (FIGURE), while the one on the right foot measured 8 x 3 cm and extended from the great toe to the base of the proximal arch. Sensation was decreased in the bilateral first and second digits but unchanged from prior documented exams. Bilateral dorsalis pedis pulses were normal.

Work-up included imaging and lab work. The patient’s complete blood count was normal, as were his erythrocyte sedimentation rate and C-reactive protein level. Radiographs of the right foot were normal, but those of the left foot were concerning, although inconclusive, for osteomyelitis. Further evaluation with magnetic resonance imaging of his left foot revealed a deformity of the first digit with some subchondral signal change that was thought to be posttraumatic or degenerative, but unlikely osteomyelitis.
THE DIAGNOSIS
Podiatry was consulted for blister management. Based on atraumatic history, rapid appearance, location of blisters, unremarkable lab work and imaging, and concurrent diabetes, the patient received a diagnosis of bilateral bullous diabeticorum (BD).
DISCUSSION
Roughly one-third of patients with diabetes will experience some cutaneous adverse effect because of the disease.1 Common iterations include acanthosis nigricans, rash, or even infection.2 BD is a rare bullous skin lesion that occurs in patients with diabetes; it has a reported annual incidence of 0.16% and may be underdiagnosed.1
Cases of BD have been described both in patients with longstanding diabetes and in those newly diagnosed, although the former group is more often affected.1 BD is reported more frequently in males than females, at a ratio of 2:1.1,3 Patients ages 17 to 80 years (average age, 55 years) have received a diagnosis of BD.1 Most affected patients will have a concomitant peripheral neuropathy and sometimes nephropathy or retinopathy.1
Continue to: The etiology of BD...
The etiology of BD is unclear but appears to be multifactorial. Hypotheses suggest that there’s a link to neuropathy/nephropathy, excessive exposure to ultraviolet light, or a vascular cause secondary to hyaline deposition in the capillary walls.4,5
What you’ll see at presentation
The typical manifestation of BD is the rapid appearance of tense blisters, which may occur overnight or even within hours.1 They are usually painless; common locations include the feet, distal legs, hands, and forearms.1,5 The bullae can be serous or hemorrhagic.1
Most notable in the patient’s history will be a lack of trauma or injury to the area.1 Although A1C values do not correlate with blister formation, patients with hypoglycemic episodes and highly varying blood glucose values seem to have higher rates of occurrence.1
Other sources of blistering must be ruled out
The diagnosis of BD is clinical and based on history, exam, and exclusion of other bullous diagnoses.6 A key clue in the history is the spontaneous and rapid onset without associated trauma in a patient with diabetes.6 Direct immunofluorescence, although nonspecific, can be helpful to rule out other disorders (such as porphyria cutanea tarda and bullous pemphigoid) if the history and exam are inconclusive. Direct and indirect immunofluorescence is typically negative in BD.4,6
The differential diagnosis includes other conditions that involve bullae—such as frictional bullae, bullous pemphigoid, and bullous systemic lupus erythematosus—as well as porphyria, erythema multiforme, insect bites, or even fixed drug eruption.2,7
Continue to: Porphyria
Porphyria tends to develop on the hands, whereas BD most commonly occurs on the feet.5
Erythema multiforme typically includes inflammatory skin changes.5
Trauma or fixed drug eruption as a cause of blistering lesions would be revealed during history taking.
Considerations for treatment and follow-up
Without treatment, blisters often self-resolve in 2 to 6 weeks, but there is high likelihood of recurrence.6,8 There is no consensus on treatment, although a typical course of action is to deroof the blister and examine the area to rule out infection.6 The wound is then covered with wet-to-dry gauze that is changed regularly. If there is suspicion for or signs of underlying infection, such as an ulcer or skin necrosis, antibiotics should be included in the treatment plan.7
Additional considerations. Patients will often need therapeutic footwear if the blisters are located on the feet. Given the higher prevalence of microvascular complications in patients with diabetes who develop BD, routine ophthalmologic examination and renal function testing to monitor for microalbuminuria are recommended.5
Our patient underwent bedside incision and drainage and was discharged home with appropriate wound care and follow-up.
THE TAKEAWAY
BD cases may be underdiagnosed in clinical practice, perhaps due to patients not seeking help for a seemingly nonthreatening condition or lack of clinician recognition that bullae are related to a patient’s diabetes status. Prompt recognition and proper wound care are important to prevent poor outcomes, such as ulceration or necrosis.
CORRESPONDENCE
Kathleen S. Kinderwater, MD, 101 Heart Drive, Greenville, NC 27834; salmondk19@ecu.edu
THE CASE
A 57-year-old man with type 2 diabetes, hyperlipidemia, and obesity presented to the emergency department (ED) for bilateral foot blisters, both of which appeared 1 day prior to evaluation. The patient’s history also included right-side Charcot foot diagnosed 4 years earlier and right foot osteomyelitis diagnosed 2 years prior. He had ongoing neuropathy in both feet but denied any significant pain.
The patient wore orthotics daily and he’d had new orthotics made 6 months prior; however, a recent COVID-19 diagnosis and prolonged hospital stay resulted in a 30-pound weight loss and decreased swelling in his ankles. He acquired new shoes 2 weeks prior to ED presentation.
Physical examination revealed large blisters along the medial aspect of the patient’s feet, with both hemorrhagic and serous fluid-filled bullae. The lesions were flaccid but intact, without drainage or surrounding erythema, warmth, or tenderness. The blister on the left foot measured 8 x 5 cm and extended from the great toe to mid-arch (FIGURE), while the one on the right foot measured 8 x 3 cm and extended from the great toe to the base of the proximal arch. Sensation was decreased in the bilateral first and second digits but unchanged from prior documented exams. Bilateral dorsalis pedis pulses were normal.

Work-up included imaging and lab work. The patient’s complete blood count was normal, as were his erythrocyte sedimentation rate and C-reactive protein level. Radiographs of the right foot were normal, but those of the left foot were concerning, although inconclusive, for osteomyelitis. Further evaluation with magnetic resonance imaging of his left foot revealed a deformity of the first digit with some subchondral signal change that was thought to be posttraumatic or degenerative, but unlikely osteomyelitis.
THE DIAGNOSIS
Podiatry was consulted for blister management. Based on atraumatic history, rapid appearance, location of blisters, unremarkable lab work and imaging, and concurrent diabetes, the patient received a diagnosis of bilateral bullous diabeticorum (BD).
DISCUSSION
Roughly one-third of patients with diabetes will experience some cutaneous adverse effect because of the disease.1 Common iterations include acanthosis nigricans, rash, or even infection.2 BD is a rare bullous skin lesion that occurs in patients with diabetes; it has a reported annual incidence of 0.16% and may be underdiagnosed.1
Cases of BD have been described both in patients with longstanding diabetes and in those newly diagnosed, although the former group is more often affected.1 BD is reported more frequently in males than females, at a ratio of 2:1.1,3 Patients ages 17 to 80 years (average age, 55 years) have received a diagnosis of BD.1 Most affected patients will have a concomitant peripheral neuropathy and sometimes nephropathy or retinopathy.1
Continue to: The etiology of BD...
The etiology of BD is unclear but appears to be multifactorial. Hypotheses suggest that there’s a link to neuropathy/nephropathy, excessive exposure to ultraviolet light, or a vascular cause secondary to hyaline deposition in the capillary walls.4,5
What you’ll see at presentation
The typical manifestation of BD is the rapid appearance of tense blisters, which may occur overnight or even within hours.1 They are usually painless; common locations include the feet, distal legs, hands, and forearms.1,5 The bullae can be serous or hemorrhagic.1
Most notable in the patient’s history will be a lack of trauma or injury to the area.1 Although A1C values do not correlate with blister formation, patients with hypoglycemic episodes and highly varying blood glucose values seem to have higher rates of occurrence.1
Other sources of blistering must be ruled out
The diagnosis of BD is clinical and based on history, exam, and exclusion of other bullous diagnoses.6 A key clue in the history is the spontaneous and rapid onset without associated trauma in a patient with diabetes.6 Direct immunofluorescence, although nonspecific, can be helpful to rule out other disorders (such as porphyria cutanea tarda and bullous pemphigoid) if the history and exam are inconclusive. Direct and indirect immunofluorescence is typically negative in BD.4,6
The differential diagnosis includes other conditions that involve bullae—such as frictional bullae, bullous pemphigoid, and bullous systemic lupus erythematosus—as well as porphyria, erythema multiforme, insect bites, or even fixed drug eruption.2,7
Continue to: Porphyria
Porphyria tends to develop on the hands, whereas BD most commonly occurs on the feet.5
Erythema multiforme typically includes inflammatory skin changes.5
Trauma or fixed drug eruption as a cause of blistering lesions would be revealed during history taking.
Considerations for treatment and follow-up
Without treatment, blisters often self-resolve in 2 to 6 weeks, but there is high likelihood of recurrence.6,8 There is no consensus on treatment, although a typical course of action is to deroof the blister and examine the area to rule out infection.6 The wound is then covered with wet-to-dry gauze that is changed regularly. If there is suspicion for or signs of underlying infection, such as an ulcer or skin necrosis, antibiotics should be included in the treatment plan.7
Additional considerations. Patients will often need therapeutic footwear if the blisters are located on the feet. Given the higher prevalence of microvascular complications in patients with diabetes who develop BD, routine ophthalmologic examination and renal function testing to monitor for microalbuminuria are recommended.5
Our patient underwent bedside incision and drainage and was discharged home with appropriate wound care and follow-up.
THE TAKEAWAY
BD cases may be underdiagnosed in clinical practice, perhaps due to patients not seeking help for a seemingly nonthreatening condition or lack of clinician recognition that bullae are related to a patient’s diabetes status. Prompt recognition and proper wound care are important to prevent poor outcomes, such as ulceration or necrosis.
CORRESPONDENCE
Kathleen S. Kinderwater, MD, 101 Heart Drive, Greenville, NC 27834; salmondk19@ecu.edu
1. Larsen K, Jensen T, Karlsmark T, et al. Incidence of bullosis diabeticorum—a controversial cause of chronic foot ulceration. Int Wound J. 2008;5:591-596. doi: 10.1111/j.1742-481X.2008.00476.x
2. Lipsky BA, Baker PD, Ahroni JH. Diabetic bullae: 12 cases of a purportedly rare cutaneous disorder. Int J Dermatol. 2000;39:196-200. doi: 10.1046/j.1365-4362.2000.00947.x
3. Gupta V, Gulati N, Bahl J, et al. Bullosis diabeticorum: rare presentation in a common disease. Case Rep Endocrinol. 2014;2014:862912.
4. Sonani H, Abdul Salim S, Garla VV, et al. Bullosis diabeticorum: a rare presentation with immunoglobulin G (IgG) deposition related vasculopathy. Case report and focused review. Am J Case Rep. 2018;19:52-56. doi: 10.12659/ajcr.905452
5. Chouk C, Litaiem N. Bullosis diabeticorum. StatPearls [Internet]. Updated June 5, 2021. Accessed July 14, 2022. www.ncbi.nlm.nih.gov/books/NBK539872/
6. Chatterjee D, Radotra A, Radotra BD, et al. Bullous diabeticorum: a rare blistering manifestation of diabetes. Indian Dermatol Online J. 2017;8:274-275. doi: 10.4103/idoj.IDOJ_340_16
7. Kansal NK, Anuragi RP. Bullous lesions in diabetes mellitus: bullous diabeticorum (diabetic bulla). BMJ Case Rep. 2020;13:e238617. doi: 10.1136/bcr-2020-238617
8. Bello F, Samaila OM, Lawal Y, et al. 2 cases of bullosis diabeticorum following long-distance journeys by road: a report of 2 cases. Case Rep Endocrinol. 2012;2012:367218. doi: 10.1155/2012/367218
1. Larsen K, Jensen T, Karlsmark T, et al. Incidence of bullosis diabeticorum—a controversial cause of chronic foot ulceration. Int Wound J. 2008;5:591-596. doi: 10.1111/j.1742-481X.2008.00476.x
2. Lipsky BA, Baker PD, Ahroni JH. Diabetic bullae: 12 cases of a purportedly rare cutaneous disorder. Int J Dermatol. 2000;39:196-200. doi: 10.1046/j.1365-4362.2000.00947.x
3. Gupta V, Gulati N, Bahl J, et al. Bullosis diabeticorum: rare presentation in a common disease. Case Rep Endocrinol. 2014;2014:862912.
4. Sonani H, Abdul Salim S, Garla VV, et al. Bullosis diabeticorum: a rare presentation with immunoglobulin G (IgG) deposition related vasculopathy. Case report and focused review. Am J Case Rep. 2018;19:52-56. doi: 10.12659/ajcr.905452
5. Chouk C, Litaiem N. Bullosis diabeticorum. StatPearls [Internet]. Updated June 5, 2021. Accessed July 14, 2022. www.ncbi.nlm.nih.gov/books/NBK539872/
6. Chatterjee D, Radotra A, Radotra BD, et al. Bullous diabeticorum: a rare blistering manifestation of diabetes. Indian Dermatol Online J. 2017;8:274-275. doi: 10.4103/idoj.IDOJ_340_16
7. Kansal NK, Anuragi RP. Bullous lesions in diabetes mellitus: bullous diabeticorum (diabetic bulla). BMJ Case Rep. 2020;13:e238617. doi: 10.1136/bcr-2020-238617
8. Bello F, Samaila OM, Lawal Y, et al. 2 cases of bullosis diabeticorum following long-distance journeys by road: a report of 2 cases. Case Rep Endocrinol. 2012;2012:367218. doi: 10.1155/2012/367218
Lack of exercise linked to small heart, HFpEF
Chronic lack of exercise – dubbed “exercise deficiency” – is associated with cardiac atrophy, reduced cardiac output and chamber size, and diminished cardiorespiratory fitness (CRF) in a subgroup of patients with heart failure with preserved ejection fraction (HFpEF), researchers say.
Increasing the physical activity levels of these sedentary individuals could be an effective preventive strategy, particularly for those who are younger and middle-aged, they suggest.
Thinking of HFpEF as an exercise deficiency syndrome leading to a small heart “flies in the face of decades of cardiovascular teaching, because traditionally, we’ve thought of heart failure as the big floppy heart,” Andre La Gerche, MBBS, PhD, of the Baker Heart and Diabetes Institute, Melbourne, told this news organization.
“While it is true that some people with HFpEF have thick, stiff hearts, we propose that another subset has a normal heart, except it’s small because it’s been underexercised,” he said.
The article, published online as part of a Focus Seminar series in the Journal of the American College of Cardiology, has “gone viral on social media,” Jason C. Kovacic, MBBS, PhD, of the Victor Chang Cardiac Research Institute, Darlinghurst, Australia, told this news organization.
Dr. Kovacic is a JACC section editor and the coordinating and senior author of the series, which covers other issues surrounding physical activity, both in athletes and the general public.
‘Coin-dropping moment’
To support their hypothesis that HFpEF is an exercise deficiency in certain patients, Dr. La Gerche and colleagues conducted a literature review that highlights the following points:
- There is a strong association between physical activity and both CRF and heart function.
- Exercise deficiency is a major risk factor for HFpEF in a subset of patients.
- Increasing physical activity is associated with greater cardiac mass, stroke volumes, cardiac output, and peak oxygen consumption.
- Physical inactivity leads to loss of heart muscle, reduced output and chamber size, and less ability to improve cardiac performance with exercise.
- Aging results in a smaller, stiffer heart; however, this effect is mitigated by regular exercise.
- Individuals who are sedentary throughout life cannot attenuate age-related reductions in heart size and have increasing chamber stiffness.
“When we explain it, it’s like a coin-dropping moment, because it’s actually a really simple concept,” Dr. La Gerche said. “A small heart has a small stroke volume. A patient with a small heart with a maximal stroke volume of 60 mL can generate a cardiac output of 9 L/min at a heart rate of 150 beats/min during exercise – an output that just isn’t enough. It’s like trying to drive a truck with a 50cc motorbike engine.”
“Plus,” Dr. La Gerche added, “exercise deficiency also sets the stage for comorbidities such as obesity, diabetes, and high blood pressure, all of which can ultimately lead to HFpEF.”
Considering HFpEF as an exercise deficiency syndrome has two clinical implications, Dr. La Gerche said. “First, it helps us understand the condition and diagnose more cases. For example, I think practitioners will start to recognize that breathlessness in some of their patients is associated with a small heart.”
“Second,” he said, “if it’s an exercise deficiency syndrome, the treatment is exercise. For most people, that means exercising regularly before the age of 60 to prevent HFpEF, because studies have found that after the age of 60, the heart is a bit fixed and harder to remodel. That doesn’t mean you shouldn’t try after 60 or that you won’t get benefit. But the real sweet spot is in middle age and younger.”
The bigger picture
The JACC Focus Seminar series starts with an article that underscores the benefits of regular physical activity. “The key is getting our patients to meet the guidelines: 150 to 300 minutes of moderate intensity exercise per week, or 75 to 250 minutes of vigorous activity per week,” Dr. Kovacic emphasized.
“Yes, we can give a statin to lower cholesterol. Yes, we can give a blood pressure medication to lower blood pressure. But when you prescribe exercise, you impact patients’ blood pressure, their cholesterol, their weight, their sense of well-being,” he said. “It cuts across so many different aspects of people’s lives that it’s important to underscore the value of exercise to everybody.”
That includes physicians, he affirmed. “It behooves all physicians to be leading by example. I would encourage those who are overweight or aren’t exercising as much as they should be to make the time to be healthy and to exercise. If you don’t, then bad health will force you to make the time to deal with bad health issues.”
Other articles in the series deal with the athlete’s heart. Christopher Semsarian, MBBS, PhD, MPH, University of Sydney, and colleagues discuss emerging data on hypertrophic cardiomyopathy and other genetic cardiovascular diseases, with the conclusion that it is probably okay for more athletes with these conditions to participate in recreational and competitive sports than was previously thought – another paradigm shift, according to Dr. Kovacic.
The final article addresses some of the challenges and controversies related to the athlete’s heart, including whether extreme exercise is associated with vulnerability to atrial fibrillation and other arrhythmias, and the impact of gender on the cardiac response to exercise, which can’t be determined now because of a paucity of data on women in sports.
Overall, Dr. Kovacic said, the series makes for “compelling” reading that should encourage readers to embark on their own studies to add to the data and support exercise prescription across the board.
No commercial funding or relevant conflicts of interest were reported.
A version of this article first appeared on Medscape.com.
Chronic lack of exercise – dubbed “exercise deficiency” – is associated with cardiac atrophy, reduced cardiac output and chamber size, and diminished cardiorespiratory fitness (CRF) in a subgroup of patients with heart failure with preserved ejection fraction (HFpEF), researchers say.
Increasing the physical activity levels of these sedentary individuals could be an effective preventive strategy, particularly for those who are younger and middle-aged, they suggest.
Thinking of HFpEF as an exercise deficiency syndrome leading to a small heart “flies in the face of decades of cardiovascular teaching, because traditionally, we’ve thought of heart failure as the big floppy heart,” Andre La Gerche, MBBS, PhD, of the Baker Heart and Diabetes Institute, Melbourne, told this news organization.
“While it is true that some people with HFpEF have thick, stiff hearts, we propose that another subset has a normal heart, except it’s small because it’s been underexercised,” he said.
The article, published online as part of a Focus Seminar series in the Journal of the American College of Cardiology, has “gone viral on social media,” Jason C. Kovacic, MBBS, PhD, of the Victor Chang Cardiac Research Institute, Darlinghurst, Australia, told this news organization.
Dr. Kovacic is a JACC section editor and the coordinating and senior author of the series, which covers other issues surrounding physical activity, both in athletes and the general public.
‘Coin-dropping moment’
To support their hypothesis that HFpEF is an exercise deficiency in certain patients, Dr. La Gerche and colleagues conducted a literature review that highlights the following points:
- There is a strong association between physical activity and both CRF and heart function.
- Exercise deficiency is a major risk factor for HFpEF in a subset of patients.
- Increasing physical activity is associated with greater cardiac mass, stroke volumes, cardiac output, and peak oxygen consumption.
- Physical inactivity leads to loss of heart muscle, reduced output and chamber size, and less ability to improve cardiac performance with exercise.
- Aging results in a smaller, stiffer heart; however, this effect is mitigated by regular exercise.
- Individuals who are sedentary throughout life cannot attenuate age-related reductions in heart size and have increasing chamber stiffness.
“When we explain it, it’s like a coin-dropping moment, because it’s actually a really simple concept,” Dr. La Gerche said. “A small heart has a small stroke volume. A patient with a small heart with a maximal stroke volume of 60 mL can generate a cardiac output of 9 L/min at a heart rate of 150 beats/min during exercise – an output that just isn’t enough. It’s like trying to drive a truck with a 50cc motorbike engine.”
“Plus,” Dr. La Gerche added, “exercise deficiency also sets the stage for comorbidities such as obesity, diabetes, and high blood pressure, all of which can ultimately lead to HFpEF.”
Considering HFpEF as an exercise deficiency syndrome has two clinical implications, Dr. La Gerche said. “First, it helps us understand the condition and diagnose more cases. For example, I think practitioners will start to recognize that breathlessness in some of their patients is associated with a small heart.”
“Second,” he said, “if it’s an exercise deficiency syndrome, the treatment is exercise. For most people, that means exercising regularly before the age of 60 to prevent HFpEF, because studies have found that after the age of 60, the heart is a bit fixed and harder to remodel. That doesn’t mean you shouldn’t try after 60 or that you won’t get benefit. But the real sweet spot is in middle age and younger.”
The bigger picture
The JACC Focus Seminar series starts with an article that underscores the benefits of regular physical activity. “The key is getting our patients to meet the guidelines: 150 to 300 minutes of moderate intensity exercise per week, or 75 to 250 minutes of vigorous activity per week,” Dr. Kovacic emphasized.
“Yes, we can give a statin to lower cholesterol. Yes, we can give a blood pressure medication to lower blood pressure. But when you prescribe exercise, you impact patients’ blood pressure, their cholesterol, their weight, their sense of well-being,” he said. “It cuts across so many different aspects of people’s lives that it’s important to underscore the value of exercise to everybody.”
That includes physicians, he affirmed. “It behooves all physicians to be leading by example. I would encourage those who are overweight or aren’t exercising as much as they should be to make the time to be healthy and to exercise. If you don’t, then bad health will force you to make the time to deal with bad health issues.”
Other articles in the series deal with the athlete’s heart. Christopher Semsarian, MBBS, PhD, MPH, University of Sydney, and colleagues discuss emerging data on hypertrophic cardiomyopathy and other genetic cardiovascular diseases, with the conclusion that it is probably okay for more athletes with these conditions to participate in recreational and competitive sports than was previously thought – another paradigm shift, according to Dr. Kovacic.
The final article addresses some of the challenges and controversies related to the athlete’s heart, including whether extreme exercise is associated with vulnerability to atrial fibrillation and other arrhythmias, and the impact of gender on the cardiac response to exercise, which can’t be determined now because of a paucity of data on women in sports.
Overall, Dr. Kovacic said, the series makes for “compelling” reading that should encourage readers to embark on their own studies to add to the data and support exercise prescription across the board.
No commercial funding or relevant conflicts of interest were reported.
A version of this article first appeared on Medscape.com.
Chronic lack of exercise – dubbed “exercise deficiency” – is associated with cardiac atrophy, reduced cardiac output and chamber size, and diminished cardiorespiratory fitness (CRF) in a subgroup of patients with heart failure with preserved ejection fraction (HFpEF), researchers say.
Increasing the physical activity levels of these sedentary individuals could be an effective preventive strategy, particularly for those who are younger and middle-aged, they suggest.
Thinking of HFpEF as an exercise deficiency syndrome leading to a small heart “flies in the face of decades of cardiovascular teaching, because traditionally, we’ve thought of heart failure as the big floppy heart,” Andre La Gerche, MBBS, PhD, of the Baker Heart and Diabetes Institute, Melbourne, told this news organization.
“While it is true that some people with HFpEF have thick, stiff hearts, we propose that another subset has a normal heart, except it’s small because it’s been underexercised,” he said.
The article, published online as part of a Focus Seminar series in the Journal of the American College of Cardiology, has “gone viral on social media,” Jason C. Kovacic, MBBS, PhD, of the Victor Chang Cardiac Research Institute, Darlinghurst, Australia, told this news organization.
Dr. Kovacic is a JACC section editor and the coordinating and senior author of the series, which covers other issues surrounding physical activity, both in athletes and the general public.
‘Coin-dropping moment’
To support their hypothesis that HFpEF is an exercise deficiency in certain patients, Dr. La Gerche and colleagues conducted a literature review that highlights the following points:
- There is a strong association between physical activity and both CRF and heart function.
- Exercise deficiency is a major risk factor for HFpEF in a subset of patients.
- Increasing physical activity is associated with greater cardiac mass, stroke volumes, cardiac output, and peak oxygen consumption.
- Physical inactivity leads to loss of heart muscle, reduced output and chamber size, and less ability to improve cardiac performance with exercise.
- Aging results in a smaller, stiffer heart; however, this effect is mitigated by regular exercise.
- Individuals who are sedentary throughout life cannot attenuate age-related reductions in heart size and have increasing chamber stiffness.
“When we explain it, it’s like a coin-dropping moment, because it’s actually a really simple concept,” Dr. La Gerche said. “A small heart has a small stroke volume. A patient with a small heart with a maximal stroke volume of 60 mL can generate a cardiac output of 9 L/min at a heart rate of 150 beats/min during exercise – an output that just isn’t enough. It’s like trying to drive a truck with a 50cc motorbike engine.”
“Plus,” Dr. La Gerche added, “exercise deficiency also sets the stage for comorbidities such as obesity, diabetes, and high blood pressure, all of which can ultimately lead to HFpEF.”
Considering HFpEF as an exercise deficiency syndrome has two clinical implications, Dr. La Gerche said. “First, it helps us understand the condition and diagnose more cases. For example, I think practitioners will start to recognize that breathlessness in some of their patients is associated with a small heart.”
“Second,” he said, “if it’s an exercise deficiency syndrome, the treatment is exercise. For most people, that means exercising regularly before the age of 60 to prevent HFpEF, because studies have found that after the age of 60, the heart is a bit fixed and harder to remodel. That doesn’t mean you shouldn’t try after 60 or that you won’t get benefit. But the real sweet spot is in middle age and younger.”
The bigger picture
The JACC Focus Seminar series starts with an article that underscores the benefits of regular physical activity. “The key is getting our patients to meet the guidelines: 150 to 300 minutes of moderate intensity exercise per week, or 75 to 250 minutes of vigorous activity per week,” Dr. Kovacic emphasized.
“Yes, we can give a statin to lower cholesterol. Yes, we can give a blood pressure medication to lower blood pressure. But when you prescribe exercise, you impact patients’ blood pressure, their cholesterol, their weight, their sense of well-being,” he said. “It cuts across so many different aspects of people’s lives that it’s important to underscore the value of exercise to everybody.”
That includes physicians, he affirmed. “It behooves all physicians to be leading by example. I would encourage those who are overweight or aren’t exercising as much as they should be to make the time to be healthy and to exercise. If you don’t, then bad health will force you to make the time to deal with bad health issues.”
Other articles in the series deal with the athlete’s heart. Christopher Semsarian, MBBS, PhD, MPH, University of Sydney, and colleagues discuss emerging data on hypertrophic cardiomyopathy and other genetic cardiovascular diseases, with the conclusion that it is probably okay for more athletes with these conditions to participate in recreational and competitive sports than was previously thought – another paradigm shift, according to Dr. Kovacic.
The final article addresses some of the challenges and controversies related to the athlete’s heart, including whether extreme exercise is associated with vulnerability to atrial fibrillation and other arrhythmias, and the impact of gender on the cardiac response to exercise, which can’t be determined now because of a paucity of data on women in sports.
Overall, Dr. Kovacic said, the series makes for “compelling” reading that should encourage readers to embark on their own studies to add to the data and support exercise prescription across the board.
No commercial funding or relevant conflicts of interest were reported.
A version of this article first appeared on Medscape.com.
New AI tech could detect type 2 diabetes without a blood test
Imagine that instead of a patient visiting their doctor for blood tests, they could rely on a noninvasive at-home test to predict their risk of diabetes, a disease that affects nearly 15% of U.S. adults (23% of whom are undiagnosed), according to the U.S. Centers for Disease Control and Prevention.
This technology could become a reality thanks to a research team that developed a machine learning algorithm to predict whether people had type 2 diabetes, prediabetes, or no diabetes. In an article published in BMJ Innovations, the researchers describe how their algorithm sorted people into these three categories with 97% accuracy on the basis of measurements of the heart’s electrical activity, determined from an electrocardiogram.
To develop and train their machine learning model – a type of artificial intelligence (AI) that keeps getting smarter over time – researchers used ECG measurements from 1,262 people in Central India. The study participants were part of the Sindhi population, an ethnic group that has been shown in past studies to be at elevated risk for type 2 diabetes.
Why ECG data? Because “cardiovascular abnormalities and diabetes, they go hand in hand,” says study author Manju Mamtani, MD, general manager of M&H Research, San Antonio, and treasurer of the Lata Medical Research Foundation. Subtle cardiovascular changes can occur even early in the development of diabetes.
“ECG has the power to detect these fluctuations, at least in theory, but those fluctuations are so tiny that many times we as humans looking at that might miss it,” says study author Hemant Kulkarni, MD, chief executive officer of M&H Research and president of the Lata Medical Research Foundation. “But the AI, which is powered to detect such specific fluctuations or subtle features, we hypothesized for the study that the AI algorithm might be able to pick those things up. And it did.”
Although this isn’t the first AI algorithm developed to predict diabetes risk, it outperforms previous models, the researchers say.
The team hopes to test and validate the algorithm in a variety of populations so that it can eventually be developed into an accessible, user-friendly technology. They envision that someday their algorithm could be used in smartwatches or other smart devices and could be integrated into telehealth so that people could be screened for diabetes even if they weren’t able to travel to a health care facility for blood testing.
The team is also studying other noninvasive methods of early disease detection and predictive models for adverse outcomes using AI.
“The fact that these algorithms are able to pick up the things of interest and learn on their own and keep learning in the future also adds excitement to their use in these settings,” says Dr. Kulkarni.
A version of this article first appeared on Medscape.com.
Imagine that instead of a patient visiting their doctor for blood tests, they could rely on a noninvasive at-home test to predict their risk of diabetes, a disease that affects nearly 15% of U.S. adults (23% of whom are undiagnosed), according to the U.S. Centers for Disease Control and Prevention.
This technology could become a reality thanks to a research team that developed a machine learning algorithm to predict whether people had type 2 diabetes, prediabetes, or no diabetes. In an article published in BMJ Innovations, the researchers describe how their algorithm sorted people into these three categories with 97% accuracy on the basis of measurements of the heart’s electrical activity, determined from an electrocardiogram.
To develop and train their machine learning model – a type of artificial intelligence (AI) that keeps getting smarter over time – researchers used ECG measurements from 1,262 people in Central India. The study participants were part of the Sindhi population, an ethnic group that has been shown in past studies to be at elevated risk for type 2 diabetes.
Why ECG data? Because “cardiovascular abnormalities and diabetes, they go hand in hand,” says study author Manju Mamtani, MD, general manager of M&H Research, San Antonio, and treasurer of the Lata Medical Research Foundation. Subtle cardiovascular changes can occur even early in the development of diabetes.
“ECG has the power to detect these fluctuations, at least in theory, but those fluctuations are so tiny that many times we as humans looking at that might miss it,” says study author Hemant Kulkarni, MD, chief executive officer of M&H Research and president of the Lata Medical Research Foundation. “But the AI, which is powered to detect such specific fluctuations or subtle features, we hypothesized for the study that the AI algorithm might be able to pick those things up. And it did.”
Although this isn’t the first AI algorithm developed to predict diabetes risk, it outperforms previous models, the researchers say.
The team hopes to test and validate the algorithm in a variety of populations so that it can eventually be developed into an accessible, user-friendly technology. They envision that someday their algorithm could be used in smartwatches or other smart devices and could be integrated into telehealth so that people could be screened for diabetes even if they weren’t able to travel to a health care facility for blood testing.
The team is also studying other noninvasive methods of early disease detection and predictive models for adverse outcomes using AI.
“The fact that these algorithms are able to pick up the things of interest and learn on their own and keep learning in the future also adds excitement to their use in these settings,” says Dr. Kulkarni.
A version of this article first appeared on Medscape.com.
Imagine that instead of a patient visiting their doctor for blood tests, they could rely on a noninvasive at-home test to predict their risk of diabetes, a disease that affects nearly 15% of U.S. adults (23% of whom are undiagnosed), according to the U.S. Centers for Disease Control and Prevention.
This technology could become a reality thanks to a research team that developed a machine learning algorithm to predict whether people had type 2 diabetes, prediabetes, or no diabetes. In an article published in BMJ Innovations, the researchers describe how their algorithm sorted people into these three categories with 97% accuracy on the basis of measurements of the heart’s electrical activity, determined from an electrocardiogram.
To develop and train their machine learning model – a type of artificial intelligence (AI) that keeps getting smarter over time – researchers used ECG measurements from 1,262 people in Central India. The study participants were part of the Sindhi population, an ethnic group that has been shown in past studies to be at elevated risk for type 2 diabetes.
Why ECG data? Because “cardiovascular abnormalities and diabetes, they go hand in hand,” says study author Manju Mamtani, MD, general manager of M&H Research, San Antonio, and treasurer of the Lata Medical Research Foundation. Subtle cardiovascular changes can occur even early in the development of diabetes.
“ECG has the power to detect these fluctuations, at least in theory, but those fluctuations are so tiny that many times we as humans looking at that might miss it,” says study author Hemant Kulkarni, MD, chief executive officer of M&H Research and president of the Lata Medical Research Foundation. “But the AI, which is powered to detect such specific fluctuations or subtle features, we hypothesized for the study that the AI algorithm might be able to pick those things up. And it did.”
Although this isn’t the first AI algorithm developed to predict diabetes risk, it outperforms previous models, the researchers say.
The team hopes to test and validate the algorithm in a variety of populations so that it can eventually be developed into an accessible, user-friendly technology. They envision that someday their algorithm could be used in smartwatches or other smart devices and could be integrated into telehealth so that people could be screened for diabetes even if they weren’t able to travel to a health care facility for blood testing.
The team is also studying other noninvasive methods of early disease detection and predictive models for adverse outcomes using AI.
“The fact that these algorithms are able to pick up the things of interest and learn on their own and keep learning in the future also adds excitement to their use in these settings,” says Dr. Kulkarni.
A version of this article first appeared on Medscape.com.
Commentary: Clinical Use of SGLT2 Inhibitors, GLP-1RA, and Insulin, September 2022
Many sodium-glucose cotransporter-2 (SGLT2) inhibitors are approved for use at two doses, but there are few clinical data regarding the metabolic impact of uptitrating an SGLT2 inhibitor from the lower to the higher dose in clinical practice. Matsumura and colleagues published the results of a retrospective, longitudinal study at a single institution in Japan. A total of 52 participants who were treated with 10 mg empagliflozin once daily were analyzed at 26 weeks after the dose had been increased to 25 mg once daily. The researchers reported a 0.6 kg weight reduction, a 0.15% reduction in A1c, and a 22.1 mg/dL reduction in triglycerides in the participants on the higher dose of empagliflozin. Although the benefits of the higher dose were rather small, this study does aid the clinician regarding the clinical impact of increasing the dose of empagliflozin.
Outcome studies with SGLT2 inhibitors have shown reductions in major adverse cardiovascular events (MACE), heart failure hospitalization, and mortality. However, clinicians may be reluctant to initiate SGLT2 inhibitors in frail individuals as they are often excluded from randomized trials and may be more likely to have side effects from this class of medications. Wood and colleagues conducted a cohort study in Australia, comparing the effectiveness of SGLT2 inhibitors to that of dipeptidyl peptidase-4 (DPP-4) inhibitors. The study was done with individuals with type 2 diabetes who were initiated on these agents within 60 days of a hospital discharge. It was noted that SGLT2 inhibitors significantly reduced MACE, heart failure hospitalization, and mortality compared with DPP-4 inhibitors, and this benefit was present in both frail and nonfrail individuals. The study did not report on tolerability issues and is limited by the cohort design, but it does suggest a cardiovascular benefit among frail patients with type 2 diabetes who are treated with SGLT2 inhibitors, and it may be reassuring when considering an SGLT2 inhibitor in a frail person.
In my July 2022 commentary, I discussed the results of AWARD-PEDS, which demonstrated a significant A1c reduction but no weight loss with the glucagon-like peptide-1 receptor agonist (GLP-1RA) dulaglutide in youth with type 2 diabetes. Tamborlane and colleagues have now reported the results of a randomized trial that studied the efficacy and safety of 2 mg exenatide once weekly in youth with type 2 diabetes. Similarly to the AWARD-PEDS study, A1c was significantly reduced compared with placebo, with a difference of -0.85% at 24 weeks. Also similarly to AWARD-PEDS, there was no significant difference in body weight between the GLP-1RA and placebo groups. There are now three studies showing glycemic benefits but little weight loss with GLP-1RA treatment in youth with type 2 diabetes, and while the glycemic benefits are encouraging, it remains perplexing why these studies have not demonstrated the weight loss that has consistently been demonstrated in adult studies of GLP-1RA.
Clinicians often choose a second-generation basal insulin analog (glargine U300, degludec) over a first-generation basal analog (glargine U100, detemir) because of lower rates of hypoglycemia. Randomized clinical trials and real-world evidence (RWE) studies comparing glargine U100 vs degludec have shown somewhat inconsistent results. In the newest RWE study comparing these two second-generation analogs, RESTORE-2 NAIVE, Fadini and colleagues reported that 6 months after initiating either glargine U300 or degludec in insulin-naive type 2 diabetes, there was a similar improvement in glycemia, no weight gain, and low hypoglycemia rates in each group. RESTORE-2 is another study demonstrating similar results between the two second-generation insulin analogs and helps build our understanding that these two insulins are more similar than different.
Many sodium-glucose cotransporter-2 (SGLT2) inhibitors are approved for use at two doses, but there are few clinical data regarding the metabolic impact of uptitrating an SGLT2 inhibitor from the lower to the higher dose in clinical practice. Matsumura and colleagues published the results of a retrospective, longitudinal study at a single institution in Japan. A total of 52 participants who were treated with 10 mg empagliflozin once daily were analyzed at 26 weeks after the dose had been increased to 25 mg once daily. The researchers reported a 0.6 kg weight reduction, a 0.15% reduction in A1c, and a 22.1 mg/dL reduction in triglycerides in the participants on the higher dose of empagliflozin. Although the benefits of the higher dose were rather small, this study does aid the clinician regarding the clinical impact of increasing the dose of empagliflozin.
Outcome studies with SGLT2 inhibitors have shown reductions in major adverse cardiovascular events (MACE), heart failure hospitalization, and mortality. However, clinicians may be reluctant to initiate SGLT2 inhibitors in frail individuals as they are often excluded from randomized trials and may be more likely to have side effects from this class of medications. Wood and colleagues conducted a cohort study in Australia, comparing the effectiveness of SGLT2 inhibitors to that of dipeptidyl peptidase-4 (DPP-4) inhibitors. The study was done with individuals with type 2 diabetes who were initiated on these agents within 60 days of a hospital discharge. It was noted that SGLT2 inhibitors significantly reduced MACE, heart failure hospitalization, and mortality compared with DPP-4 inhibitors, and this benefit was present in both frail and nonfrail individuals. The study did not report on tolerability issues and is limited by the cohort design, but it does suggest a cardiovascular benefit among frail patients with type 2 diabetes who are treated with SGLT2 inhibitors, and it may be reassuring when considering an SGLT2 inhibitor in a frail person.
In my July 2022 commentary, I discussed the results of AWARD-PEDS, which demonstrated a significant A1c reduction but no weight loss with the glucagon-like peptide-1 receptor agonist (GLP-1RA) dulaglutide in youth with type 2 diabetes. Tamborlane and colleagues have now reported the results of a randomized trial that studied the efficacy and safety of 2 mg exenatide once weekly in youth with type 2 diabetes. Similarly to the AWARD-PEDS study, A1c was significantly reduced compared with placebo, with a difference of -0.85% at 24 weeks. Also similarly to AWARD-PEDS, there was no significant difference in body weight between the GLP-1RA and placebo groups. There are now three studies showing glycemic benefits but little weight loss with GLP-1RA treatment in youth with type 2 diabetes, and while the glycemic benefits are encouraging, it remains perplexing why these studies have not demonstrated the weight loss that has consistently been demonstrated in adult studies of GLP-1RA.
Clinicians often choose a second-generation basal insulin analog (glargine U300, degludec) over a first-generation basal analog (glargine U100, detemir) because of lower rates of hypoglycemia. Randomized clinical trials and real-world evidence (RWE) studies comparing glargine U100 vs degludec have shown somewhat inconsistent results. In the newest RWE study comparing these two second-generation analogs, RESTORE-2 NAIVE, Fadini and colleagues reported that 6 months after initiating either glargine U300 or degludec in insulin-naive type 2 diabetes, there was a similar improvement in glycemia, no weight gain, and low hypoglycemia rates in each group. RESTORE-2 is another study demonstrating similar results between the two second-generation insulin analogs and helps build our understanding that these two insulins are more similar than different.
Many sodium-glucose cotransporter-2 (SGLT2) inhibitors are approved for use at two doses, but there are few clinical data regarding the metabolic impact of uptitrating an SGLT2 inhibitor from the lower to the higher dose in clinical practice. Matsumura and colleagues published the results of a retrospective, longitudinal study at a single institution in Japan. A total of 52 participants who were treated with 10 mg empagliflozin once daily were analyzed at 26 weeks after the dose had been increased to 25 mg once daily. The researchers reported a 0.6 kg weight reduction, a 0.15% reduction in A1c, and a 22.1 mg/dL reduction in triglycerides in the participants on the higher dose of empagliflozin. Although the benefits of the higher dose were rather small, this study does aid the clinician regarding the clinical impact of increasing the dose of empagliflozin.
Outcome studies with SGLT2 inhibitors have shown reductions in major adverse cardiovascular events (MACE), heart failure hospitalization, and mortality. However, clinicians may be reluctant to initiate SGLT2 inhibitors in frail individuals as they are often excluded from randomized trials and may be more likely to have side effects from this class of medications. Wood and colleagues conducted a cohort study in Australia, comparing the effectiveness of SGLT2 inhibitors to that of dipeptidyl peptidase-4 (DPP-4) inhibitors. The study was done with individuals with type 2 diabetes who were initiated on these agents within 60 days of a hospital discharge. It was noted that SGLT2 inhibitors significantly reduced MACE, heart failure hospitalization, and mortality compared with DPP-4 inhibitors, and this benefit was present in both frail and nonfrail individuals. The study did not report on tolerability issues and is limited by the cohort design, but it does suggest a cardiovascular benefit among frail patients with type 2 diabetes who are treated with SGLT2 inhibitors, and it may be reassuring when considering an SGLT2 inhibitor in a frail person.
In my July 2022 commentary, I discussed the results of AWARD-PEDS, which demonstrated a significant A1c reduction but no weight loss with the glucagon-like peptide-1 receptor agonist (GLP-1RA) dulaglutide in youth with type 2 diabetes. Tamborlane and colleagues have now reported the results of a randomized trial that studied the efficacy and safety of 2 mg exenatide once weekly in youth with type 2 diabetes. Similarly to the AWARD-PEDS study, A1c was significantly reduced compared with placebo, with a difference of -0.85% at 24 weeks. Also similarly to AWARD-PEDS, there was no significant difference in body weight between the GLP-1RA and placebo groups. There are now three studies showing glycemic benefits but little weight loss with GLP-1RA treatment in youth with type 2 diabetes, and while the glycemic benefits are encouraging, it remains perplexing why these studies have not demonstrated the weight loss that has consistently been demonstrated in adult studies of GLP-1RA.
Clinicians often choose a second-generation basal insulin analog (glargine U300, degludec) over a first-generation basal analog (glargine U100, detemir) because of lower rates of hypoglycemia. Randomized clinical trials and real-world evidence (RWE) studies comparing glargine U100 vs degludec have shown somewhat inconsistent results. In the newest RWE study comparing these two second-generation analogs, RESTORE-2 NAIVE, Fadini and colleagues reported that 6 months after initiating either glargine U300 or degludec in insulin-naive type 2 diabetes, there was a similar improvement in glycemia, no weight gain, and low hypoglycemia rates in each group. RESTORE-2 is another study demonstrating similar results between the two second-generation insulin analogs and helps build our understanding that these two insulins are more similar than different.
Exercise may counteract genetics for gestational diabetes
Women giving birth for the first time have significantly higher odds of developing gestational diabetes if they have a high polygenic risk score (PRS) and low physical activity, new data suggest.
Researchers, led by Kymberleigh A. Pagel, PhD, with the department of computer science, Indiana University, Bloomington, concluded that physical activity early in pregnancy is associated with reduced risk of gestational diabetes and may help women who are at high risk because of genetic predisposition, age, family history of diabetes, and body mass index.
The researchers included 3,533 women in the analysis (average age, 28.6 years) which was a subcohort of a larger study. They found that physical activity’s association with lower gestational diabetes risk “was particularly significant in individuals who were genetically predisposed to diabetes through PRS or family history,” the authors wrote.
Women with high PRS and low level of physical activity had three times the odds of developing gestational diabetes (odds ratio, 3.4; 95% confidence interval, 2.3-5.3).
Those with high PRS and moderate to high activity levels in early pregnancy (metabolic equivalents of task [METs] of at least 450) had gestational diabetes risk similar to that of the general population, according to the researchers.
The findings were published in JAMA Network Open.
Maisa Feghali, MD, a maternal-fetal specialist at the University of Pittsburgh Medical Center, who was not part of the study, said in an interview she found the link of physical activity and compensation for high predisposition to gestational diabetes most interesting.
“That’s interesting because a lot of studies that have looked at prevention of gestational diabetes either through limited weight gain or through some form of counseling on physical activity have not really shown any benefit,” she noted. “It might just be it’s not just one size fits all and it may be that physical activity is mostly beneficial in those with a high predisposition.”
Research in this area is particularly important as 7% of pregnancies in the United States each year are affected by gestational diabetes and the risk for developing type 2 diabetes “has doubled in the past decade among patients with GD [gestational diabetes],” the authors wrote.
Researchers looked at risks for gestational diabetes in high-risk subgroups, including women who had a body mass index of more than 25 kg/m2 or were at least 35 years old. In that group, women who were either in the in the top 25th percentile for PRS or had low physical activity (METs less than 450) had from 25% to 75% greater risk of developing gestational diabetes.
The findings are consistent with previous research and suggest exercise interventions may be important in improving pregnancy outcomes, the authors wrote.
Christina Han, MD, division director for maternal-fetal medicine at University of California, Los Angeles, who was not part of the study, pointed out several limitations of the study, however.
One of the biggest limitations, she said, was that “they excluded two-thirds of the original study. Essentially, they took only Caucasian [White] patients, which is about one-third of the study.” Additionally, the cohort was made up of people who had never had babies.
“Lots of our gestational diabetes patients are not first-time moms, so this makes the generalizability of the study very limited,” Dr. Han said.
She added that none of the sites where the study was conducted were in the South or Northwest, which also adds questions about generalizability.
Dr. Feghali and Dr. Han reported no relevant financial relationships.
Women giving birth for the first time have significantly higher odds of developing gestational diabetes if they have a high polygenic risk score (PRS) and low physical activity, new data suggest.
Researchers, led by Kymberleigh A. Pagel, PhD, with the department of computer science, Indiana University, Bloomington, concluded that physical activity early in pregnancy is associated with reduced risk of gestational diabetes and may help women who are at high risk because of genetic predisposition, age, family history of diabetes, and body mass index.
The researchers included 3,533 women in the analysis (average age, 28.6 years) which was a subcohort of a larger study. They found that physical activity’s association with lower gestational diabetes risk “was particularly significant in individuals who were genetically predisposed to diabetes through PRS or family history,” the authors wrote.
Women with high PRS and low level of physical activity had three times the odds of developing gestational diabetes (odds ratio, 3.4; 95% confidence interval, 2.3-5.3).
Those with high PRS and moderate to high activity levels in early pregnancy (metabolic equivalents of task [METs] of at least 450) had gestational diabetes risk similar to that of the general population, according to the researchers.
The findings were published in JAMA Network Open.
Maisa Feghali, MD, a maternal-fetal specialist at the University of Pittsburgh Medical Center, who was not part of the study, said in an interview she found the link of physical activity and compensation for high predisposition to gestational diabetes most interesting.
“That’s interesting because a lot of studies that have looked at prevention of gestational diabetes either through limited weight gain or through some form of counseling on physical activity have not really shown any benefit,” she noted. “It might just be it’s not just one size fits all and it may be that physical activity is mostly beneficial in those with a high predisposition.”
Research in this area is particularly important as 7% of pregnancies in the United States each year are affected by gestational diabetes and the risk for developing type 2 diabetes “has doubled in the past decade among patients with GD [gestational diabetes],” the authors wrote.
Researchers looked at risks for gestational diabetes in high-risk subgroups, including women who had a body mass index of more than 25 kg/m2 or were at least 35 years old. In that group, women who were either in the in the top 25th percentile for PRS or had low physical activity (METs less than 450) had from 25% to 75% greater risk of developing gestational diabetes.
The findings are consistent with previous research and suggest exercise interventions may be important in improving pregnancy outcomes, the authors wrote.
Christina Han, MD, division director for maternal-fetal medicine at University of California, Los Angeles, who was not part of the study, pointed out several limitations of the study, however.
One of the biggest limitations, she said, was that “they excluded two-thirds of the original study. Essentially, they took only Caucasian [White] patients, which is about one-third of the study.” Additionally, the cohort was made up of people who had never had babies.
“Lots of our gestational diabetes patients are not first-time moms, so this makes the generalizability of the study very limited,” Dr. Han said.
She added that none of the sites where the study was conducted were in the South or Northwest, which also adds questions about generalizability.
Dr. Feghali and Dr. Han reported no relevant financial relationships.
Women giving birth for the first time have significantly higher odds of developing gestational diabetes if they have a high polygenic risk score (PRS) and low physical activity, new data suggest.
Researchers, led by Kymberleigh A. Pagel, PhD, with the department of computer science, Indiana University, Bloomington, concluded that physical activity early in pregnancy is associated with reduced risk of gestational diabetes and may help women who are at high risk because of genetic predisposition, age, family history of diabetes, and body mass index.
The researchers included 3,533 women in the analysis (average age, 28.6 years) which was a subcohort of a larger study. They found that physical activity’s association with lower gestational diabetes risk “was particularly significant in individuals who were genetically predisposed to diabetes through PRS or family history,” the authors wrote.
Women with high PRS and low level of physical activity had three times the odds of developing gestational diabetes (odds ratio, 3.4; 95% confidence interval, 2.3-5.3).
Those with high PRS and moderate to high activity levels in early pregnancy (metabolic equivalents of task [METs] of at least 450) had gestational diabetes risk similar to that of the general population, according to the researchers.
The findings were published in JAMA Network Open.
Maisa Feghali, MD, a maternal-fetal specialist at the University of Pittsburgh Medical Center, who was not part of the study, said in an interview she found the link of physical activity and compensation for high predisposition to gestational diabetes most interesting.
“That’s interesting because a lot of studies that have looked at prevention of gestational diabetes either through limited weight gain or through some form of counseling on physical activity have not really shown any benefit,” she noted. “It might just be it’s not just one size fits all and it may be that physical activity is mostly beneficial in those with a high predisposition.”
Research in this area is particularly important as 7% of pregnancies in the United States each year are affected by gestational diabetes and the risk for developing type 2 diabetes “has doubled in the past decade among patients with GD [gestational diabetes],” the authors wrote.
Researchers looked at risks for gestational diabetes in high-risk subgroups, including women who had a body mass index of more than 25 kg/m2 or were at least 35 years old. In that group, women who were either in the in the top 25th percentile for PRS or had low physical activity (METs less than 450) had from 25% to 75% greater risk of developing gestational diabetes.
The findings are consistent with previous research and suggest exercise interventions may be important in improving pregnancy outcomes, the authors wrote.
Christina Han, MD, division director for maternal-fetal medicine at University of California, Los Angeles, who was not part of the study, pointed out several limitations of the study, however.
One of the biggest limitations, she said, was that “they excluded two-thirds of the original study. Essentially, they took only Caucasian [White] patients, which is about one-third of the study.” Additionally, the cohort was made up of people who had never had babies.
“Lots of our gestational diabetes patients are not first-time moms, so this makes the generalizability of the study very limited,” Dr. Han said.
She added that none of the sites where the study was conducted were in the South or Northwest, which also adds questions about generalizability.
Dr. Feghali and Dr. Han reported no relevant financial relationships.
FROM JAMA NETWORK OPEN