User login
‘Staggering’ CVD rise projected in U.S., especially in minorities
A new analysis projects steep increases by 2060 in the prevalence of cardiovascular (CV) risk factors and disease that will disproportionately affect non-White populations who have limited access to health care.
The study by Reza Mohebi, MD, Massachusetts General Hospital and Harvard Medical School, both in Boston, and colleagues was published in the Journal of the American College of Cardiology.
“Even though several assumptions underlie these projections, the importance of this work cannot be overestimated,” Andreas P. Kalogeropoulos, MD, MPH, PhD, and Javed Butler, MD, MPH, MBA, wrote in an accompanying editorial. “The absolute numbers are staggering.”
From 2025 to 2060, the number of people with any one of four CV risk factors – type 2 diabetes, hypertension, dyslipidemia, and obesity – is projected to increase by 15.4 million, to 34.7 million.
And the number of people with of any one of four CV disease types – ischemic heart disease, heart failure, MI, and stroke – is projected to increase by 3.2 million, to 6.8 million.
Although the model predicts that the prevalence of CV risk factors will gradually decrease among White Americans, the highest prevalence of CV risk factors will be among the White population because of its overall size.
Conversely, the projected prevalence of CV risk factors is expected to increase in Black, Hispanic, Asian, and other race/ethnicity populations.
In parallel, the prevalence of CV disease is projected to decrease in the White population and increase among all other race/ethnicities, particularly in the Black and Hispanic populations.
“Our results project a worrisome increase with a particularly ominous increase in risk factors and disease in our most vulnerable patients, including Blacks and Hispanics,” senior author James L. Januzzi Jr., MD, summarized in a video issued by the society.
“The steep rise in CV risk factors and disease reflects the generally higher prevalence in populations projected to increase in the United States, owing to immigration and growth, including Black or Hispanic individuals,” Dr. Januzzi, also from Massachusetts General and Harvard, said in an interview.
“The disproportionate size of the risk is expected in a sense, as minority populations are disproportionately disadvantaged with respect to their health care,” he said. “But whether it is expected or not, the increase in projected prevalence is, nonetheless, concerning and a call to action.”
This study identifies “areas of opportunity for change in the U.S. health care system,” he continued. “Business as usual will result in us encountering a huge number of individuals with CV risk factors and diseases.”
The results from the current analysis assume there will be no modification in health care policies or changes in access to care for at-risk populations, Dr. Mohebi and colleagues noted.
To “stem the rising tide of CV disease in at-risk individuals,” would require strategies such as “emphasis on education regarding CV risk factors, improving access to quality healthcare, and facilitating lower-cost access to effective therapies for treatment of CV risk factors,” according to the researchers.
“Such advances need to be applied in a more equitable way throughout the United States, however,” they cautioned.
Census plus NHANES data
The researchers used 2020 U.S. census data and projected growth and 2013-2018 U.S. National Health and Nutrition Survey data to estimate the number of people with CV risk factors and CV disease from 2025 to 2060.
The estimates are based on a growing population and a fixed frequency.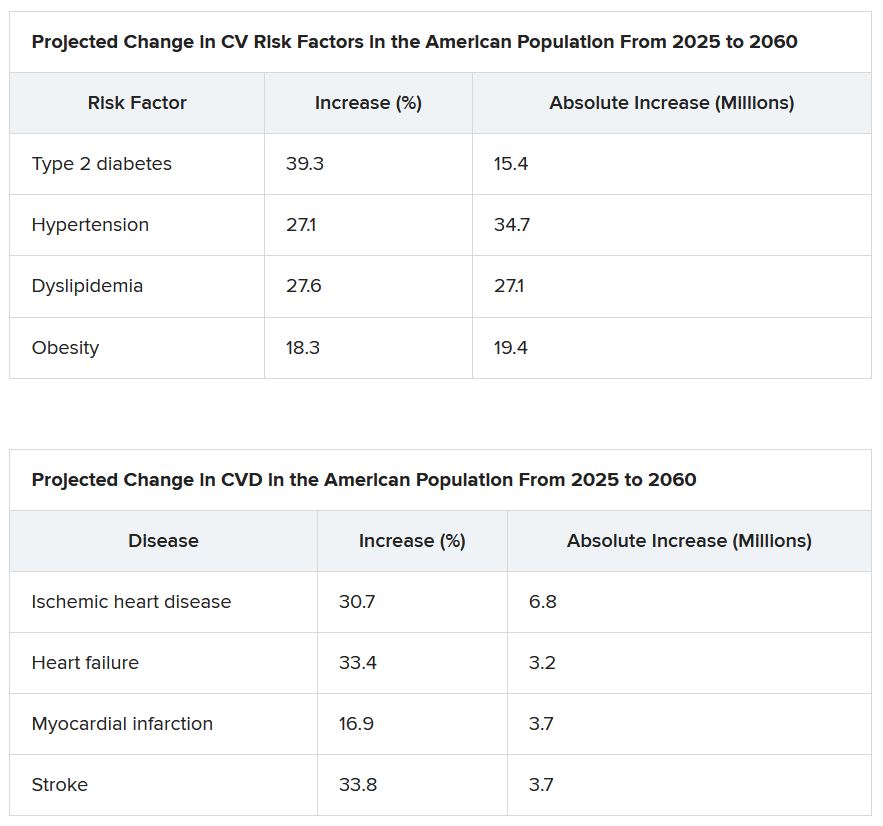
The projected changes in CV risk factors and disease over time were similar in men and women.
The researchers acknowledge that study limitations include the assumption that the prevalence patterns for CV risk factors and disease will be stable.
“To the extent the frequency of risk factors and disease are not likely to remain static, that assumption may reduce the accuracy of the projections,” Dr. Januzzi said. “However, we would point out that the goals of our analysis were to set general trends, and not to seek to project exact figures.”
Also, they did not take into account the effect of COVID-19. CV diseases were also based on self-report and CV risk factors could have been underestimated in minority populations that do not access health care.
Changing demographic landscape
It is “striking” that the numbers of non-White individuals with CV risk factors is projected to surpass the number of White individuals over time, and the number of non-White individuals with CV disease will be almost as many as White individuals by the year 2060, the editorialists noted.
“From a policy perspective, this means that unless appropriate, targeted action is taken, disparities in the burden of cardiovascular disease are only going to be exacerbated over time,” wrote Dr. Kalogeropoulos, from Stony Brook (N.Y.) University, and Dr. Butler, from Baylor College of Medicine, Dallas.
“On the positive side,” they continued, “the absolute increase in the percent prevalence of cardiovascular risk factors and conditions is projected to lie within a manageable range,” assuming that specific prevention policies are implemented.
“This is an opportunity for professional societies, including the cardiovascular care community, to re-evaluate priorities and strategies, for both training and practice, to best match the growing demands of a changing demographic landscape in the United States,” Dr. Kalogeropoulos and Dr. Butler concluded.
Dr. Mohebi is supported by the Barry Fellowship. Dr. Januzzi is supported by the Hutter Family Professorship; is a Trustee of the American College of Cardiology; is a board member of Imbria Pharmaceuticals; has received grant support from Abbott Diagnostics, Applied Therapeutics, Innolife, and Novartis; has received consulting income from Abbott Diagnostics, Boehringer Ingelheim, Janssen, Novartis, and Roche Diagnostics; and participates in clinical endpoint committees/data safety monitoring boards for AbbVie, Siemens, Takeda, and Vifor. Dr. Kalogeropoulos has received research funding from the National Heart, Lung, and Blood Institute; the American Heart Association; and the Centers for Disease Control and Prevention. Dr. Butler has been a consultant for numerous pharmaceutical companies.
A version of this article first appeared on Medscape.com.
A new analysis projects steep increases by 2060 in the prevalence of cardiovascular (CV) risk factors and disease that will disproportionately affect non-White populations who have limited access to health care.
The study by Reza Mohebi, MD, Massachusetts General Hospital and Harvard Medical School, both in Boston, and colleagues was published in the Journal of the American College of Cardiology.
“Even though several assumptions underlie these projections, the importance of this work cannot be overestimated,” Andreas P. Kalogeropoulos, MD, MPH, PhD, and Javed Butler, MD, MPH, MBA, wrote in an accompanying editorial. “The absolute numbers are staggering.”
From 2025 to 2060, the number of people with any one of four CV risk factors – type 2 diabetes, hypertension, dyslipidemia, and obesity – is projected to increase by 15.4 million, to 34.7 million.
And the number of people with of any one of four CV disease types – ischemic heart disease, heart failure, MI, and stroke – is projected to increase by 3.2 million, to 6.8 million.
Although the model predicts that the prevalence of CV risk factors will gradually decrease among White Americans, the highest prevalence of CV risk factors will be among the White population because of its overall size.
Conversely, the projected prevalence of CV risk factors is expected to increase in Black, Hispanic, Asian, and other race/ethnicity populations.
In parallel, the prevalence of CV disease is projected to decrease in the White population and increase among all other race/ethnicities, particularly in the Black and Hispanic populations.
“Our results project a worrisome increase with a particularly ominous increase in risk factors and disease in our most vulnerable patients, including Blacks and Hispanics,” senior author James L. Januzzi Jr., MD, summarized in a video issued by the society.
“The steep rise in CV risk factors and disease reflects the generally higher prevalence in populations projected to increase in the United States, owing to immigration and growth, including Black or Hispanic individuals,” Dr. Januzzi, also from Massachusetts General and Harvard, said in an interview.
“The disproportionate size of the risk is expected in a sense, as minority populations are disproportionately disadvantaged with respect to their health care,” he said. “But whether it is expected or not, the increase in projected prevalence is, nonetheless, concerning and a call to action.”
This study identifies “areas of opportunity for change in the U.S. health care system,” he continued. “Business as usual will result in us encountering a huge number of individuals with CV risk factors and diseases.”
The results from the current analysis assume there will be no modification in health care policies or changes in access to care for at-risk populations, Dr. Mohebi and colleagues noted.
To “stem the rising tide of CV disease in at-risk individuals,” would require strategies such as “emphasis on education regarding CV risk factors, improving access to quality healthcare, and facilitating lower-cost access to effective therapies for treatment of CV risk factors,” according to the researchers.
“Such advances need to be applied in a more equitable way throughout the United States, however,” they cautioned.
Census plus NHANES data
The researchers used 2020 U.S. census data and projected growth and 2013-2018 U.S. National Health and Nutrition Survey data to estimate the number of people with CV risk factors and CV disease from 2025 to 2060.
The estimates are based on a growing population and a fixed frequency.
The projected changes in CV risk factors and disease over time were similar in men and women.
The researchers acknowledge that study limitations include the assumption that the prevalence patterns for CV risk factors and disease will be stable.
“To the extent the frequency of risk factors and disease are not likely to remain static, that assumption may reduce the accuracy of the projections,” Dr. Januzzi said. “However, we would point out that the goals of our analysis were to set general trends, and not to seek to project exact figures.”
Also, they did not take into account the effect of COVID-19. CV diseases were also based on self-report and CV risk factors could have been underestimated in minority populations that do not access health care.
Changing demographic landscape
It is “striking” that the numbers of non-White individuals with CV risk factors is projected to surpass the number of White individuals over time, and the number of non-White individuals with CV disease will be almost as many as White individuals by the year 2060, the editorialists noted.
“From a policy perspective, this means that unless appropriate, targeted action is taken, disparities in the burden of cardiovascular disease are only going to be exacerbated over time,” wrote Dr. Kalogeropoulos, from Stony Brook (N.Y.) University, and Dr. Butler, from Baylor College of Medicine, Dallas.
“On the positive side,” they continued, “the absolute increase in the percent prevalence of cardiovascular risk factors and conditions is projected to lie within a manageable range,” assuming that specific prevention policies are implemented.
“This is an opportunity for professional societies, including the cardiovascular care community, to re-evaluate priorities and strategies, for both training and practice, to best match the growing demands of a changing demographic landscape in the United States,” Dr. Kalogeropoulos and Dr. Butler concluded.
Dr. Mohebi is supported by the Barry Fellowship. Dr. Januzzi is supported by the Hutter Family Professorship; is a Trustee of the American College of Cardiology; is a board member of Imbria Pharmaceuticals; has received grant support from Abbott Diagnostics, Applied Therapeutics, Innolife, and Novartis; has received consulting income from Abbott Diagnostics, Boehringer Ingelheim, Janssen, Novartis, and Roche Diagnostics; and participates in clinical endpoint committees/data safety monitoring boards for AbbVie, Siemens, Takeda, and Vifor. Dr. Kalogeropoulos has received research funding from the National Heart, Lung, and Blood Institute; the American Heart Association; and the Centers for Disease Control and Prevention. Dr. Butler has been a consultant for numerous pharmaceutical companies.
A version of this article first appeared on Medscape.com.
A new analysis projects steep increases by 2060 in the prevalence of cardiovascular (CV) risk factors and disease that will disproportionately affect non-White populations who have limited access to health care.
The study by Reza Mohebi, MD, Massachusetts General Hospital and Harvard Medical School, both in Boston, and colleagues was published in the Journal of the American College of Cardiology.
“Even though several assumptions underlie these projections, the importance of this work cannot be overestimated,” Andreas P. Kalogeropoulos, MD, MPH, PhD, and Javed Butler, MD, MPH, MBA, wrote in an accompanying editorial. “The absolute numbers are staggering.”
From 2025 to 2060, the number of people with any one of four CV risk factors – type 2 diabetes, hypertension, dyslipidemia, and obesity – is projected to increase by 15.4 million, to 34.7 million.
And the number of people with of any one of four CV disease types – ischemic heart disease, heart failure, MI, and stroke – is projected to increase by 3.2 million, to 6.8 million.
Although the model predicts that the prevalence of CV risk factors will gradually decrease among White Americans, the highest prevalence of CV risk factors will be among the White population because of its overall size.
Conversely, the projected prevalence of CV risk factors is expected to increase in Black, Hispanic, Asian, and other race/ethnicity populations.
In parallel, the prevalence of CV disease is projected to decrease in the White population and increase among all other race/ethnicities, particularly in the Black and Hispanic populations.
“Our results project a worrisome increase with a particularly ominous increase in risk factors and disease in our most vulnerable patients, including Blacks and Hispanics,” senior author James L. Januzzi Jr., MD, summarized in a video issued by the society.
“The steep rise in CV risk factors and disease reflects the generally higher prevalence in populations projected to increase in the United States, owing to immigration and growth, including Black or Hispanic individuals,” Dr. Januzzi, also from Massachusetts General and Harvard, said in an interview.
“The disproportionate size of the risk is expected in a sense, as minority populations are disproportionately disadvantaged with respect to their health care,” he said. “But whether it is expected or not, the increase in projected prevalence is, nonetheless, concerning and a call to action.”
This study identifies “areas of opportunity for change in the U.S. health care system,” he continued. “Business as usual will result in us encountering a huge number of individuals with CV risk factors and diseases.”
The results from the current analysis assume there will be no modification in health care policies or changes in access to care for at-risk populations, Dr. Mohebi and colleagues noted.
To “stem the rising tide of CV disease in at-risk individuals,” would require strategies such as “emphasis on education regarding CV risk factors, improving access to quality healthcare, and facilitating lower-cost access to effective therapies for treatment of CV risk factors,” according to the researchers.
“Such advances need to be applied in a more equitable way throughout the United States, however,” they cautioned.
Census plus NHANES data
The researchers used 2020 U.S. census data and projected growth and 2013-2018 U.S. National Health and Nutrition Survey data to estimate the number of people with CV risk factors and CV disease from 2025 to 2060.
The estimates are based on a growing population and a fixed frequency.
The projected changes in CV risk factors and disease over time were similar in men and women.
The researchers acknowledge that study limitations include the assumption that the prevalence patterns for CV risk factors and disease will be stable.
“To the extent the frequency of risk factors and disease are not likely to remain static, that assumption may reduce the accuracy of the projections,” Dr. Januzzi said. “However, we would point out that the goals of our analysis were to set general trends, and not to seek to project exact figures.”
Also, they did not take into account the effect of COVID-19. CV diseases were also based on self-report and CV risk factors could have been underestimated in minority populations that do not access health care.
Changing demographic landscape
It is “striking” that the numbers of non-White individuals with CV risk factors is projected to surpass the number of White individuals over time, and the number of non-White individuals with CV disease will be almost as many as White individuals by the year 2060, the editorialists noted.
“From a policy perspective, this means that unless appropriate, targeted action is taken, disparities in the burden of cardiovascular disease are only going to be exacerbated over time,” wrote Dr. Kalogeropoulos, from Stony Brook (N.Y.) University, and Dr. Butler, from Baylor College of Medicine, Dallas.
“On the positive side,” they continued, “the absolute increase in the percent prevalence of cardiovascular risk factors and conditions is projected to lie within a manageable range,” assuming that specific prevention policies are implemented.
“This is an opportunity for professional societies, including the cardiovascular care community, to re-evaluate priorities and strategies, for both training and practice, to best match the growing demands of a changing demographic landscape in the United States,” Dr. Kalogeropoulos and Dr. Butler concluded.
Dr. Mohebi is supported by the Barry Fellowship. Dr. Januzzi is supported by the Hutter Family Professorship; is a Trustee of the American College of Cardiology; is a board member of Imbria Pharmaceuticals; has received grant support from Abbott Diagnostics, Applied Therapeutics, Innolife, and Novartis; has received consulting income from Abbott Diagnostics, Boehringer Ingelheim, Janssen, Novartis, and Roche Diagnostics; and participates in clinical endpoint committees/data safety monitoring boards for AbbVie, Siemens, Takeda, and Vifor. Dr. Kalogeropoulos has received research funding from the National Heart, Lung, and Blood Institute; the American Heart Association; and the Centers for Disease Control and Prevention. Dr. Butler has been a consultant for numerous pharmaceutical companies.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF AMERICAN COLLEGE OF CARDIOLOGY
Commentary: Diabetes Drug Comparisons, August 2022
Meta-analyses of sodium-glucose cotransporter 2 inhibitor (SGLT2i) outcome trials have shown reductions in all-cause and cardiovascular mortality, but dipeptidyl peptidase 4 inhibitors (DPP4i) have been neutral for these outcomes. In a Taiwanese retrospective cohort study, Chung and colleagues compared 53,264 pairs of propensity-matched patients with type 2 diabetes who were treated with either an SGLT2i or DPP4i. They not only reported relative risk reductions of 34% and 32% for all-cause death and cardiovascular death, respectively, but also a reduction in cancer death of 27% and a reduction in noncancer, noncardiovascular death of 38%. Although limited by its retrospective, observational design, the finding of a benefit of SGLT2i treatment on both cancer death and noncancer, noncardiovascular death would benefit from further research.
In another large Taiwanese retrospective cohort study with propensity matching, Chan and colleagues compared patients treated with SGLT2i, DPP4i, and glucagon-like peptide 1 receptor agonists (GLP-1RA), with a main study outcome of new-onset atrial fibrillation (AF). They noted that SGLT2i treatment was associated with a 10% and 36% lower risk for AF compared with DPP4i and GLP-1RA treatment, respectively. These results are consistent with meta-analyses of SGLT2i outcome trials that have demonstrated reductions in AF with SGLT2i vs placebo. Perhaps it is time to recognize that another clinical benefit of the SGLT2i class is the reduction in risk for AF.
Although GLP-1RA have been linked to an increased risk for gallbladder-related events in many studies, there has been little data suggesting an increased risk with DPP4i. He and colleagues have published a pairwise meta-analysis of 82 randomized clinical trials and found that DPP4i compared with placebo or nonincretin drugs increased the risk for gallbladder or biliary diseases by 1.22-fold, with 11 more events per 10,000 person years. In a network meta-analysis of 184 randomized trials, they also found that DPP4i treatment increased the risk for gallbladder or biliary diseases compared with SGLT2i but not compared with GLP-1RA. This was the first meta-analysis to systematically study the association between DPP4i and gallbladder-related diseases. Although the absolute risk is small, clinicians need to be aware of this link and consider this adverse effect when deciding about the risks vs benefits of DPP4i treatment.
Individuals with obesity and prediabetes are at greater risk for type 2 diabetes. Trials of lifestyle modification and antiobesity agents have shown that restoration to normoglycemia can occur with weight loss. Phase 3A studies of the antiobesity agent semaglutide (2.4 mg/week) included 3375 individuals with prediabetes across three trials. In a post hoc analysis of these patients with prediabetes, Perreault and colleagues found that after 68 weeks of treatment, there was a much higher likelihood of normoglycemia with 2.4 mg/week of semaglutide compared with placebo. Though definite conclusions are limited owing to the post hoc nature of this analysis, these results make it very likely that the ongoing STEP 10 trial of 2.4 mg semaglutide vs placebo (201 participants with obesity and prediabetes) will probably show a significant benefit on the primary outcome of change to normoglycemia.
Meta-analyses of sodium-glucose cotransporter 2 inhibitor (SGLT2i) outcome trials have shown reductions in all-cause and cardiovascular mortality, but dipeptidyl peptidase 4 inhibitors (DPP4i) have been neutral for these outcomes. In a Taiwanese retrospective cohort study, Chung and colleagues compared 53,264 pairs of propensity-matched patients with type 2 diabetes who were treated with either an SGLT2i or DPP4i. They not only reported relative risk reductions of 34% and 32% for all-cause death and cardiovascular death, respectively, but also a reduction in cancer death of 27% and a reduction in noncancer, noncardiovascular death of 38%. Although limited by its retrospective, observational design, the finding of a benefit of SGLT2i treatment on both cancer death and noncancer, noncardiovascular death would benefit from further research.
In another large Taiwanese retrospective cohort study with propensity matching, Chan and colleagues compared patients treated with SGLT2i, DPP4i, and glucagon-like peptide 1 receptor agonists (GLP-1RA), with a main study outcome of new-onset atrial fibrillation (AF). They noted that SGLT2i treatment was associated with a 10% and 36% lower risk for AF compared with DPP4i and GLP-1RA treatment, respectively. These results are consistent with meta-analyses of SGLT2i outcome trials that have demonstrated reductions in AF with SGLT2i vs placebo. Perhaps it is time to recognize that another clinical benefit of the SGLT2i class is the reduction in risk for AF.
Although GLP-1RA have been linked to an increased risk for gallbladder-related events in many studies, there has been little data suggesting an increased risk with DPP4i. He and colleagues have published a pairwise meta-analysis of 82 randomized clinical trials and found that DPP4i compared with placebo or nonincretin drugs increased the risk for gallbladder or biliary diseases by 1.22-fold, with 11 more events per 10,000 person years. In a network meta-analysis of 184 randomized trials, they also found that DPP4i treatment increased the risk for gallbladder or biliary diseases compared with SGLT2i but not compared with GLP-1RA. This was the first meta-analysis to systematically study the association between DPP4i and gallbladder-related diseases. Although the absolute risk is small, clinicians need to be aware of this link and consider this adverse effect when deciding about the risks vs benefits of DPP4i treatment.
Individuals with obesity and prediabetes are at greater risk for type 2 diabetes. Trials of lifestyle modification and antiobesity agents have shown that restoration to normoglycemia can occur with weight loss. Phase 3A studies of the antiobesity agent semaglutide (2.4 mg/week) included 3375 individuals with prediabetes across three trials. In a post hoc analysis of these patients with prediabetes, Perreault and colleagues found that after 68 weeks of treatment, there was a much higher likelihood of normoglycemia with 2.4 mg/week of semaglutide compared with placebo. Though definite conclusions are limited owing to the post hoc nature of this analysis, these results make it very likely that the ongoing STEP 10 trial of 2.4 mg semaglutide vs placebo (201 participants with obesity and prediabetes) will probably show a significant benefit on the primary outcome of change to normoglycemia.
Meta-analyses of sodium-glucose cotransporter 2 inhibitor (SGLT2i) outcome trials have shown reductions in all-cause and cardiovascular mortality, but dipeptidyl peptidase 4 inhibitors (DPP4i) have been neutral for these outcomes. In a Taiwanese retrospective cohort study, Chung and colleagues compared 53,264 pairs of propensity-matched patients with type 2 diabetes who were treated with either an SGLT2i or DPP4i. They not only reported relative risk reductions of 34% and 32% for all-cause death and cardiovascular death, respectively, but also a reduction in cancer death of 27% and a reduction in noncancer, noncardiovascular death of 38%. Although limited by its retrospective, observational design, the finding of a benefit of SGLT2i treatment on both cancer death and noncancer, noncardiovascular death would benefit from further research.
In another large Taiwanese retrospective cohort study with propensity matching, Chan and colleagues compared patients treated with SGLT2i, DPP4i, and glucagon-like peptide 1 receptor agonists (GLP-1RA), with a main study outcome of new-onset atrial fibrillation (AF). They noted that SGLT2i treatment was associated with a 10% and 36% lower risk for AF compared with DPP4i and GLP-1RA treatment, respectively. These results are consistent with meta-analyses of SGLT2i outcome trials that have demonstrated reductions in AF with SGLT2i vs placebo. Perhaps it is time to recognize that another clinical benefit of the SGLT2i class is the reduction in risk for AF.
Although GLP-1RA have been linked to an increased risk for gallbladder-related events in many studies, there has been little data suggesting an increased risk with DPP4i. He and colleagues have published a pairwise meta-analysis of 82 randomized clinical trials and found that DPP4i compared with placebo or nonincretin drugs increased the risk for gallbladder or biliary diseases by 1.22-fold, with 11 more events per 10,000 person years. In a network meta-analysis of 184 randomized trials, they also found that DPP4i treatment increased the risk for gallbladder or biliary diseases compared with SGLT2i but not compared with GLP-1RA. This was the first meta-analysis to systematically study the association between DPP4i and gallbladder-related diseases. Although the absolute risk is small, clinicians need to be aware of this link and consider this adverse effect when deciding about the risks vs benefits of DPP4i treatment.
Individuals with obesity and prediabetes are at greater risk for type 2 diabetes. Trials of lifestyle modification and antiobesity agents have shown that restoration to normoglycemia can occur with weight loss. Phase 3A studies of the antiobesity agent semaglutide (2.4 mg/week) included 3375 individuals with prediabetes across three trials. In a post hoc analysis of these patients with prediabetes, Perreault and colleagues found that after 68 weeks of treatment, there was a much higher likelihood of normoglycemia with 2.4 mg/week of semaglutide compared with placebo. Though definite conclusions are limited owing to the post hoc nature of this analysis, these results make it very likely that the ongoing STEP 10 trial of 2.4 mg semaglutide vs placebo (201 participants with obesity and prediabetes) will probably show a significant benefit on the primary outcome of change to normoglycemia.
Ongoing debate whether COVID links to new diabetes in kids
compared with the pre-pandemic rate, in new research.
This contrasts with findings from a U.S. study and a German study, but this is “not the final word” about this possible association, lead author Rayzel Shulman, MD, admits, since the study may have been underpowered.
The population-based, cross-sectional study was published recently as a research letter in JAMA Open.
The researchers found a nonsignificant increase in the monthly rate of new diabetes during the first 18 months of the COVID-19 pandemic, compared with the 3 prior years (relative risk 1.09, 95% confidence interval).
New study contrasts with previous reports
This differs from a Morbidity and Mortality Weekly Report from the U.S. Centers for Disease Control and Prevention, in which COVID-19 infection was associated with a significant increase in new onset of diabetes in children during March 2020 through June 2021, “although some experts have criticized the study methods and conclusion validity,” Dr. Shulman and colleagues write.
Another study, from Germany, reported a significant 1.15-fold increase in type 1 diabetes in children during the pandemic, they note.
The current study may have been underpowered and too small to show a significant association between COVID-19 and new diabetes, the researchers acknowledge.
And the 1.30 upper limit of the confidence interval shows that it “cannot rule out a possible 1.3-fold increase” in relative risk of a diagnosis of diabetes related to COVID, Dr. Shulman explained to this news organization.
It will be important to see how the rates have changed since September 2021 (the end of the current study), added Dr. Shulman, an adjunct scientist at the Institute for Clinical Evaluative Sciences (ICES) and a physician and scientist at the Hospital for Sick Children, Toronto.
The current study did find a decreased (delayed) rate of diagnosis of new diabetes during the first months of the pandemic when there were lockdowns, followed by a “catch-up” increase in rates later on, as has been reported earlier.
“Our study is definitely not the final word on this,” Dr. Shulman summarized in a statement from ICES. “However, our findings call into question whether a direct association between COVID-19 and new-onset diabetes in children exists.”
COVID-diabetes link?
The researchers analyzed health administrative data from January 2017 to September 2021.
They identified 2,700,178 children and youth in Ontario who were under age 18 in 2021, who had a mean age of 9.2, and about half were girls.
Between November 2020 and April 2021, an estimated 3.3% of children in Ontario had a SARS-COV-2 infection.
New diagnoses of diabetes in this age group are mostly type 1 diabetes, based on previous studies.
The rate of incident diabetes was 15%-32% lower during the first 3 months of the pandemic, March-May 2020 (1.67-2.34 cases per 100,000), compared with the pre-pandemic monthly rate during 2017, 2018, and 2019 (2.54-2.59 cases per 100,000).
The rate of incident diabetes was 33%-50% higher during February to July 2021 (3.48-4.18 cases per 100,000), compared with the pre-pandemic rate.
The pre-pandemic and pandemic monthly rates of incident diabetes were similar during the other months.
The group concludes: “The lack of both an observable increase in overall diabetes incidence among children during the 18-month pandemic restrictions [in this Ontario study] and a plausible biological mechanism call into question an association between COVID-19 and new-onset diabetes.”
More research is needed. “Given the variability in monthly [relative risks], additional population-based, longer-term data are needed to examine the direct and indirect effects of COVID-19 and diabetes risk among children,” the authors write.
This study was supported by ICES (which is funded by the Ontario Ministry of Health) and by a grant from the Canadian Institutes of Health Research. Dr. Shulman reported receiving fees from Dexcom outside the submitted work, and she and three other authors reported receiving grants from the Canadian Institutes of Health Research outside the submitted work.
A version of this article first appeared on Medscape.com.
compared with the pre-pandemic rate, in new research.
This contrasts with findings from a U.S. study and a German study, but this is “not the final word” about this possible association, lead author Rayzel Shulman, MD, admits, since the study may have been underpowered.
The population-based, cross-sectional study was published recently as a research letter in JAMA Open.
The researchers found a nonsignificant increase in the monthly rate of new diabetes during the first 18 months of the COVID-19 pandemic, compared with the 3 prior years (relative risk 1.09, 95% confidence interval).
New study contrasts with previous reports
This differs from a Morbidity and Mortality Weekly Report from the U.S. Centers for Disease Control and Prevention, in which COVID-19 infection was associated with a significant increase in new onset of diabetes in children during March 2020 through June 2021, “although some experts have criticized the study methods and conclusion validity,” Dr. Shulman and colleagues write.
Another study, from Germany, reported a significant 1.15-fold increase in type 1 diabetes in children during the pandemic, they note.
The current study may have been underpowered and too small to show a significant association between COVID-19 and new diabetes, the researchers acknowledge.
And the 1.30 upper limit of the confidence interval shows that it “cannot rule out a possible 1.3-fold increase” in relative risk of a diagnosis of diabetes related to COVID, Dr. Shulman explained to this news organization.
It will be important to see how the rates have changed since September 2021 (the end of the current study), added Dr. Shulman, an adjunct scientist at the Institute for Clinical Evaluative Sciences (ICES) and a physician and scientist at the Hospital for Sick Children, Toronto.
The current study did find a decreased (delayed) rate of diagnosis of new diabetes during the first months of the pandemic when there were lockdowns, followed by a “catch-up” increase in rates later on, as has been reported earlier.
“Our study is definitely not the final word on this,” Dr. Shulman summarized in a statement from ICES. “However, our findings call into question whether a direct association between COVID-19 and new-onset diabetes in children exists.”
COVID-diabetes link?
The researchers analyzed health administrative data from January 2017 to September 2021.
They identified 2,700,178 children and youth in Ontario who were under age 18 in 2021, who had a mean age of 9.2, and about half were girls.
Between November 2020 and April 2021, an estimated 3.3% of children in Ontario had a SARS-COV-2 infection.
New diagnoses of diabetes in this age group are mostly type 1 diabetes, based on previous studies.
The rate of incident diabetes was 15%-32% lower during the first 3 months of the pandemic, March-May 2020 (1.67-2.34 cases per 100,000), compared with the pre-pandemic monthly rate during 2017, 2018, and 2019 (2.54-2.59 cases per 100,000).
The rate of incident diabetes was 33%-50% higher during February to July 2021 (3.48-4.18 cases per 100,000), compared with the pre-pandemic rate.
The pre-pandemic and pandemic monthly rates of incident diabetes were similar during the other months.
The group concludes: “The lack of both an observable increase in overall diabetes incidence among children during the 18-month pandemic restrictions [in this Ontario study] and a plausible biological mechanism call into question an association between COVID-19 and new-onset diabetes.”
More research is needed. “Given the variability in monthly [relative risks], additional population-based, longer-term data are needed to examine the direct and indirect effects of COVID-19 and diabetes risk among children,” the authors write.
This study was supported by ICES (which is funded by the Ontario Ministry of Health) and by a grant from the Canadian Institutes of Health Research. Dr. Shulman reported receiving fees from Dexcom outside the submitted work, and she and three other authors reported receiving grants from the Canadian Institutes of Health Research outside the submitted work.
A version of this article first appeared on Medscape.com.
compared with the pre-pandemic rate, in new research.
This contrasts with findings from a U.S. study and a German study, but this is “not the final word” about this possible association, lead author Rayzel Shulman, MD, admits, since the study may have been underpowered.
The population-based, cross-sectional study was published recently as a research letter in JAMA Open.
The researchers found a nonsignificant increase in the monthly rate of new diabetes during the first 18 months of the COVID-19 pandemic, compared with the 3 prior years (relative risk 1.09, 95% confidence interval).
New study contrasts with previous reports
This differs from a Morbidity and Mortality Weekly Report from the U.S. Centers for Disease Control and Prevention, in which COVID-19 infection was associated with a significant increase in new onset of diabetes in children during March 2020 through June 2021, “although some experts have criticized the study methods and conclusion validity,” Dr. Shulman and colleagues write.
Another study, from Germany, reported a significant 1.15-fold increase in type 1 diabetes in children during the pandemic, they note.
The current study may have been underpowered and too small to show a significant association between COVID-19 and new diabetes, the researchers acknowledge.
And the 1.30 upper limit of the confidence interval shows that it “cannot rule out a possible 1.3-fold increase” in relative risk of a diagnosis of diabetes related to COVID, Dr. Shulman explained to this news organization.
It will be important to see how the rates have changed since September 2021 (the end of the current study), added Dr. Shulman, an adjunct scientist at the Institute for Clinical Evaluative Sciences (ICES) and a physician and scientist at the Hospital for Sick Children, Toronto.
The current study did find a decreased (delayed) rate of diagnosis of new diabetes during the first months of the pandemic when there were lockdowns, followed by a “catch-up” increase in rates later on, as has been reported earlier.
“Our study is definitely not the final word on this,” Dr. Shulman summarized in a statement from ICES. “However, our findings call into question whether a direct association between COVID-19 and new-onset diabetes in children exists.”
COVID-diabetes link?
The researchers analyzed health administrative data from January 2017 to September 2021.
They identified 2,700,178 children and youth in Ontario who were under age 18 in 2021, who had a mean age of 9.2, and about half were girls.
Between November 2020 and April 2021, an estimated 3.3% of children in Ontario had a SARS-COV-2 infection.
New diagnoses of diabetes in this age group are mostly type 1 diabetes, based on previous studies.
The rate of incident diabetes was 15%-32% lower during the first 3 months of the pandemic, March-May 2020 (1.67-2.34 cases per 100,000), compared with the pre-pandemic monthly rate during 2017, 2018, and 2019 (2.54-2.59 cases per 100,000).
The rate of incident diabetes was 33%-50% higher during February to July 2021 (3.48-4.18 cases per 100,000), compared with the pre-pandemic rate.
The pre-pandemic and pandemic monthly rates of incident diabetes were similar during the other months.
The group concludes: “The lack of both an observable increase in overall diabetes incidence among children during the 18-month pandemic restrictions [in this Ontario study] and a plausible biological mechanism call into question an association between COVID-19 and new-onset diabetes.”
More research is needed. “Given the variability in monthly [relative risks], additional population-based, longer-term data are needed to examine the direct and indirect effects of COVID-19 and diabetes risk among children,” the authors write.
This study was supported by ICES (which is funded by the Ontario Ministry of Health) and by a grant from the Canadian Institutes of Health Research. Dr. Shulman reported receiving fees from Dexcom outside the submitted work, and she and three other authors reported receiving grants from the Canadian Institutes of Health Research outside the submitted work.
A version of this article first appeared on Medscape.com.
FROM JAMA OPEN
Supporting Patients on Complex Care Journeys: How Technology Can Bridge the Gaps
From Memora Health (Dr. Flyckt and Dr. Colbert), San Francisco, CA; and Harvard Medical School (Dr. Colbert), Boston, MA.
A close relative was recently diagnosed with follicular lymphoma. He was cared for at a high-ranked cancer center by physicians with demonstrated expertise, and even had the support of a care navigator. Still, he was often left feeling overwhelmed and confused, holding an inch-thick stack of papers, instructions, and pamphlets. As he left his treatment planning visit, reeling from the emotional burden of his diagnosis and all the unfamiliar terminology, he didn’t know what to do or what to expect. Later, when he experienced early signs of tumor lysis syndrome, he struggled to reach his care team for triage and guidance. When he went to the emergency room, his oncologist was never informed.
This scenario is unfortunately common, and versions of this scenario play out thousands of times each day across the US health system. Within the clinic and hospital setting, patients receive excellent care from their providers, but a disconnect emerges once the patient leaves these medical settings: patients at home struggle to find guidance and support, while care teams lack the tools to engage patients between visits or monitor their health across care settings, providers, or episodes of care.
Leveraging Technology to Move From Episodes of Care to Complex Care Journeys
The use of automated messaging, artificial intelligence and natural language processing–driven chat experiences, and text-based support is becoming more common. However, health care lags behind other industries in the adoption of these technologies.1,2 The slow pace can be warranted, given that health care is more complicated and higher risk than inquiring about a lost package, ordering groceries, or applying for a mortgage. At the same time, many of the consumer engagement tools used to guide an applicant through the multiple steps and complexities of their home loan process or to prompt viewers to select new shows to binge have applications in health care.
Over the past few years, technologies have emerged that guide patients through complex care journeys and allow care teams to monitor and engage patients between visits. These solutions come in different formats, but generally patients can receive messages on their phones that contain disease-specific educational content, prompts to fill prescriptions and take medications, and reminders and guidance on how to prepare for appointments and procedures. These programs also collect relevant data from patients through survey and electronic patient-reported outcomes instruments, as well as connected patient monitoring devices, that help track patient progress and identify issues as they arise. Many programs also incorporate symptom triage pathways and use natural language processing to respond automatically to patient questions and concerns.3,4
These technology solutions can automate many tasks that in the past required a care team member to spend hours on the phone. Newly freed from such repetitive tasks, care teams can now focus on more in-depth interactions with those patients who are most in need—the types of interactions that are more satisfying and rewarding. Such assistance is particularly needed today with the staffing shortages faced by most health systems.5
In addition, technology allows teams to see the panel of patients they are caring for and to quickly identify and take action on any specific needs or issues. Care teams can focus on any patient and see where they are in their journey. When appropriate, some solutions also allow care teams to engage directly with patients through text-messaging, creating a seamless experience and unified communication channel. Ideally, these solutions should be linked or embedded within the electronic health record or other primary system of record, so that teams can easily access these tools through their existing workflows and avoid creating yet another interface to navigate.
The Impact of Low-Tech Solutions to Deliver High-Touch Support
There is evidence showing that digital patient navigation tools impact patient care. In the oncology setting, patients with a digital navigator have achieved over 95% adherence rates with complex oral chemotherapy regimens (Memora Health Unpublished Data. 2022.). In the postpartum setting, a text message–based program improved screening rates for postpartum depression and did so with very high patient satisfaction ratings.6 Particularly notable is the fact that this depression screening program achieved these results in a population that was predominantly low income, with more than half belonging to underrepresented minority populations.6
We believe these digital patient navigation technologies, specifically low-tech solutions that don’t require app downloads, portal log-ins, or high-speed internet, will transform care delivery over the next 5 to 10 years. Successful management of complex conditions like diabetes or cancer requires more than 3 hours of care each day,7 yet most patients spend only 1 or 2 hours per month directly interacting with their health care providers. However, most patients carry their phones with them at all times, and artificial intelligence–enabled text support is “always on” to provide support, monitoring, and guidance, wherever a patient happens to be when assistance is needed.
Shifting the Model to Support a Lifetime of Care
While still in the early stages of development, these tools have the potential to radically alter the practice of medicine, shifting the focus from episodic interactions to continuous journey-based care delivery. Outside of an acute event bringing a patient into the clinic or emergency room, many patients go a year or more without seeing their primary care providers.8 During that time, an immense amount of information is underreported or completely lost. Capturing this information in real-time and more holistically over a person’s lifetime of care could provide physicians better insight to both better manage and more fully evaluate the success of treatment plans by tracking patient symptoms, pain, and functional status over time. With this more longitudinal view of the patient, we see a pathway towards achieving the Quadruple Aim: patients who are more supported will achieve better outcomes at lower cost, they will have a better experience, and care teams will be empowered to focus their time on more satisfying activities rather than repetitive administrative tasks.
Corresponding author: James A. Colbert, MD, MBA; jamie@memorahealth.com
Disclosures: Dr. Flyckt and Dr. Colbert are employed by Memora Health, an organization that helps health care systems digitize and automate care journeys.
1. Hermes S, Riasanow T, Clemons EK, et al. The digital transformation of the healthcare industry: exploring the rise of emerging platform ecosystems and their influence on the role of patients. Bus Res. 2020;13:1033-1069. doi:10.1007/s40685-020-00125-x
2. Van Velthoven MH, Cordon C. Sustainable adoption of digital health innovations: perspectives from a stakeholder workshop. J Med Internet Res. 2019;21(3):e11922. doi:10.2196/11922
3. Campbell K, Louie P, Levine B, Gililland J. Using patient engagement platforms in the postoperative management of patients. Curr Rev Musculoskelet Med. 2020;13(4):479-484. doi:10.1007/s12178-020-09638-8
4. Xu L, Sanders L, Li K, Chow JCL. Chatbot for health care and oncology applications using artificial intelligence and machine learning: systematic review. JMIR Cancer. 2021;7(4):e27850. doi:10.2196/27850
5. Data brief: health care workforce challenges threaten hospitals’ ability to care for patients. American Hospital Association. Accessed July 24, 2022. www.aha.org/fact-sheets/2021-11-01-data-brief-health-care-workforce-challenges-threaten-hospitals-ability-care
6. Gaulton JS, Leitner K, Hahn L, et al. Healing at home: applying innovation principles to redesign and optimise postpartum care. BMJ Innovations. 2022;8:37-41.
7. Østbye T, Yarnall KS, Krause KM, et al. Is there time for management of patients with chronic diseases in primary care? Ann Fam Med. 2005;3(3):209-214. doi:10.1370/afm.310
8. Ganguli I, Shi Z, E. Orav J, et al. Declining use of primary care among commercially insured adults in the united states, 2008–2016. Ann Intern Med. 2020;172:240-247. doi:10.7326/M19-1834
From Memora Health (Dr. Flyckt and Dr. Colbert), San Francisco, CA; and Harvard Medical School (Dr. Colbert), Boston, MA.
A close relative was recently diagnosed with follicular lymphoma. He was cared for at a high-ranked cancer center by physicians with demonstrated expertise, and even had the support of a care navigator. Still, he was often left feeling overwhelmed and confused, holding an inch-thick stack of papers, instructions, and pamphlets. As he left his treatment planning visit, reeling from the emotional burden of his diagnosis and all the unfamiliar terminology, he didn’t know what to do or what to expect. Later, when he experienced early signs of tumor lysis syndrome, he struggled to reach his care team for triage and guidance. When he went to the emergency room, his oncologist was never informed.
This scenario is unfortunately common, and versions of this scenario play out thousands of times each day across the US health system. Within the clinic and hospital setting, patients receive excellent care from their providers, but a disconnect emerges once the patient leaves these medical settings: patients at home struggle to find guidance and support, while care teams lack the tools to engage patients between visits or monitor their health across care settings, providers, or episodes of care.
Leveraging Technology to Move From Episodes of Care to Complex Care Journeys
The use of automated messaging, artificial intelligence and natural language processing–driven chat experiences, and text-based support is becoming more common. However, health care lags behind other industries in the adoption of these technologies.1,2 The slow pace can be warranted, given that health care is more complicated and higher risk than inquiring about a lost package, ordering groceries, or applying for a mortgage. At the same time, many of the consumer engagement tools used to guide an applicant through the multiple steps and complexities of their home loan process or to prompt viewers to select new shows to binge have applications in health care.
Over the past few years, technologies have emerged that guide patients through complex care journeys and allow care teams to monitor and engage patients between visits. These solutions come in different formats, but generally patients can receive messages on their phones that contain disease-specific educational content, prompts to fill prescriptions and take medications, and reminders and guidance on how to prepare for appointments and procedures. These programs also collect relevant data from patients through survey and electronic patient-reported outcomes instruments, as well as connected patient monitoring devices, that help track patient progress and identify issues as they arise. Many programs also incorporate symptom triage pathways and use natural language processing to respond automatically to patient questions and concerns.3,4
These technology solutions can automate many tasks that in the past required a care team member to spend hours on the phone. Newly freed from such repetitive tasks, care teams can now focus on more in-depth interactions with those patients who are most in need—the types of interactions that are more satisfying and rewarding. Such assistance is particularly needed today with the staffing shortages faced by most health systems.5
In addition, technology allows teams to see the panel of patients they are caring for and to quickly identify and take action on any specific needs or issues. Care teams can focus on any patient and see where they are in their journey. When appropriate, some solutions also allow care teams to engage directly with patients through text-messaging, creating a seamless experience and unified communication channel. Ideally, these solutions should be linked or embedded within the electronic health record or other primary system of record, so that teams can easily access these tools through their existing workflows and avoid creating yet another interface to navigate.
The Impact of Low-Tech Solutions to Deliver High-Touch Support
There is evidence showing that digital patient navigation tools impact patient care. In the oncology setting, patients with a digital navigator have achieved over 95% adherence rates with complex oral chemotherapy regimens (Memora Health Unpublished Data. 2022.). In the postpartum setting, a text message–based program improved screening rates for postpartum depression and did so with very high patient satisfaction ratings.6 Particularly notable is the fact that this depression screening program achieved these results in a population that was predominantly low income, with more than half belonging to underrepresented minority populations.6
We believe these digital patient navigation technologies, specifically low-tech solutions that don’t require app downloads, portal log-ins, or high-speed internet, will transform care delivery over the next 5 to 10 years. Successful management of complex conditions like diabetes or cancer requires more than 3 hours of care each day,7 yet most patients spend only 1 or 2 hours per month directly interacting with their health care providers. However, most patients carry their phones with them at all times, and artificial intelligence–enabled text support is “always on” to provide support, monitoring, and guidance, wherever a patient happens to be when assistance is needed.
Shifting the Model to Support a Lifetime of Care
While still in the early stages of development, these tools have the potential to radically alter the practice of medicine, shifting the focus from episodic interactions to continuous journey-based care delivery. Outside of an acute event bringing a patient into the clinic or emergency room, many patients go a year or more without seeing their primary care providers.8 During that time, an immense amount of information is underreported or completely lost. Capturing this information in real-time and more holistically over a person’s lifetime of care could provide physicians better insight to both better manage and more fully evaluate the success of treatment plans by tracking patient symptoms, pain, and functional status over time. With this more longitudinal view of the patient, we see a pathway towards achieving the Quadruple Aim: patients who are more supported will achieve better outcomes at lower cost, they will have a better experience, and care teams will be empowered to focus their time on more satisfying activities rather than repetitive administrative tasks.
Corresponding author: James A. Colbert, MD, MBA; jamie@memorahealth.com
Disclosures: Dr. Flyckt and Dr. Colbert are employed by Memora Health, an organization that helps health care systems digitize and automate care journeys.
From Memora Health (Dr. Flyckt and Dr. Colbert), San Francisco, CA; and Harvard Medical School (Dr. Colbert), Boston, MA.
A close relative was recently diagnosed with follicular lymphoma. He was cared for at a high-ranked cancer center by physicians with demonstrated expertise, and even had the support of a care navigator. Still, he was often left feeling overwhelmed and confused, holding an inch-thick stack of papers, instructions, and pamphlets. As he left his treatment planning visit, reeling from the emotional burden of his diagnosis and all the unfamiliar terminology, he didn’t know what to do or what to expect. Later, when he experienced early signs of tumor lysis syndrome, he struggled to reach his care team for triage and guidance. When he went to the emergency room, his oncologist was never informed.
This scenario is unfortunately common, and versions of this scenario play out thousands of times each day across the US health system. Within the clinic and hospital setting, patients receive excellent care from their providers, but a disconnect emerges once the patient leaves these medical settings: patients at home struggle to find guidance and support, while care teams lack the tools to engage patients between visits or monitor their health across care settings, providers, or episodes of care.
Leveraging Technology to Move From Episodes of Care to Complex Care Journeys
The use of automated messaging, artificial intelligence and natural language processing–driven chat experiences, and text-based support is becoming more common. However, health care lags behind other industries in the adoption of these technologies.1,2 The slow pace can be warranted, given that health care is more complicated and higher risk than inquiring about a lost package, ordering groceries, or applying for a mortgage. At the same time, many of the consumer engagement tools used to guide an applicant through the multiple steps and complexities of their home loan process or to prompt viewers to select new shows to binge have applications in health care.
Over the past few years, technologies have emerged that guide patients through complex care journeys and allow care teams to monitor and engage patients between visits. These solutions come in different formats, but generally patients can receive messages on their phones that contain disease-specific educational content, prompts to fill prescriptions and take medications, and reminders and guidance on how to prepare for appointments and procedures. These programs also collect relevant data from patients through survey and electronic patient-reported outcomes instruments, as well as connected patient monitoring devices, that help track patient progress and identify issues as they arise. Many programs also incorporate symptom triage pathways and use natural language processing to respond automatically to patient questions and concerns.3,4
These technology solutions can automate many tasks that in the past required a care team member to spend hours on the phone. Newly freed from such repetitive tasks, care teams can now focus on more in-depth interactions with those patients who are most in need—the types of interactions that are more satisfying and rewarding. Such assistance is particularly needed today with the staffing shortages faced by most health systems.5
In addition, technology allows teams to see the panel of patients they are caring for and to quickly identify and take action on any specific needs or issues. Care teams can focus on any patient and see where they are in their journey. When appropriate, some solutions also allow care teams to engage directly with patients through text-messaging, creating a seamless experience and unified communication channel. Ideally, these solutions should be linked or embedded within the electronic health record or other primary system of record, so that teams can easily access these tools through their existing workflows and avoid creating yet another interface to navigate.
The Impact of Low-Tech Solutions to Deliver High-Touch Support
There is evidence showing that digital patient navigation tools impact patient care. In the oncology setting, patients with a digital navigator have achieved over 95% adherence rates with complex oral chemotherapy regimens (Memora Health Unpublished Data. 2022.). In the postpartum setting, a text message–based program improved screening rates for postpartum depression and did so with very high patient satisfaction ratings.6 Particularly notable is the fact that this depression screening program achieved these results in a population that was predominantly low income, with more than half belonging to underrepresented minority populations.6
We believe these digital patient navigation technologies, specifically low-tech solutions that don’t require app downloads, portal log-ins, or high-speed internet, will transform care delivery over the next 5 to 10 years. Successful management of complex conditions like diabetes or cancer requires more than 3 hours of care each day,7 yet most patients spend only 1 or 2 hours per month directly interacting with their health care providers. However, most patients carry their phones with them at all times, and artificial intelligence–enabled text support is “always on” to provide support, monitoring, and guidance, wherever a patient happens to be when assistance is needed.
Shifting the Model to Support a Lifetime of Care
While still in the early stages of development, these tools have the potential to radically alter the practice of medicine, shifting the focus from episodic interactions to continuous journey-based care delivery. Outside of an acute event bringing a patient into the clinic or emergency room, many patients go a year or more without seeing their primary care providers.8 During that time, an immense amount of information is underreported or completely lost. Capturing this information in real-time and more holistically over a person’s lifetime of care could provide physicians better insight to both better manage and more fully evaluate the success of treatment plans by tracking patient symptoms, pain, and functional status over time. With this more longitudinal view of the patient, we see a pathway towards achieving the Quadruple Aim: patients who are more supported will achieve better outcomes at lower cost, they will have a better experience, and care teams will be empowered to focus their time on more satisfying activities rather than repetitive administrative tasks.
Corresponding author: James A. Colbert, MD, MBA; jamie@memorahealth.com
Disclosures: Dr. Flyckt and Dr. Colbert are employed by Memora Health, an organization that helps health care systems digitize and automate care journeys.
1. Hermes S, Riasanow T, Clemons EK, et al. The digital transformation of the healthcare industry: exploring the rise of emerging platform ecosystems and their influence on the role of patients. Bus Res. 2020;13:1033-1069. doi:10.1007/s40685-020-00125-x
2. Van Velthoven MH, Cordon C. Sustainable adoption of digital health innovations: perspectives from a stakeholder workshop. J Med Internet Res. 2019;21(3):e11922. doi:10.2196/11922
3. Campbell K, Louie P, Levine B, Gililland J. Using patient engagement platforms in the postoperative management of patients. Curr Rev Musculoskelet Med. 2020;13(4):479-484. doi:10.1007/s12178-020-09638-8
4. Xu L, Sanders L, Li K, Chow JCL. Chatbot for health care and oncology applications using artificial intelligence and machine learning: systematic review. JMIR Cancer. 2021;7(4):e27850. doi:10.2196/27850
5. Data brief: health care workforce challenges threaten hospitals’ ability to care for patients. American Hospital Association. Accessed July 24, 2022. www.aha.org/fact-sheets/2021-11-01-data-brief-health-care-workforce-challenges-threaten-hospitals-ability-care
6. Gaulton JS, Leitner K, Hahn L, et al. Healing at home: applying innovation principles to redesign and optimise postpartum care. BMJ Innovations. 2022;8:37-41.
7. Østbye T, Yarnall KS, Krause KM, et al. Is there time for management of patients with chronic diseases in primary care? Ann Fam Med. 2005;3(3):209-214. doi:10.1370/afm.310
8. Ganguli I, Shi Z, E. Orav J, et al. Declining use of primary care among commercially insured adults in the united states, 2008–2016. Ann Intern Med. 2020;172:240-247. doi:10.7326/M19-1834
1. Hermes S, Riasanow T, Clemons EK, et al. The digital transformation of the healthcare industry: exploring the rise of emerging platform ecosystems and their influence on the role of patients. Bus Res. 2020;13:1033-1069. doi:10.1007/s40685-020-00125-x
2. Van Velthoven MH, Cordon C. Sustainable adoption of digital health innovations: perspectives from a stakeholder workshop. J Med Internet Res. 2019;21(3):e11922. doi:10.2196/11922
3. Campbell K, Louie P, Levine B, Gililland J. Using patient engagement platforms in the postoperative management of patients. Curr Rev Musculoskelet Med. 2020;13(4):479-484. doi:10.1007/s12178-020-09638-8
4. Xu L, Sanders L, Li K, Chow JCL. Chatbot for health care and oncology applications using artificial intelligence and machine learning: systematic review. JMIR Cancer. 2021;7(4):e27850. doi:10.2196/27850
5. Data brief: health care workforce challenges threaten hospitals’ ability to care for patients. American Hospital Association. Accessed July 24, 2022. www.aha.org/fact-sheets/2021-11-01-data-brief-health-care-workforce-challenges-threaten-hospitals-ability-care
6. Gaulton JS, Leitner K, Hahn L, et al. Healing at home: applying innovation principles to redesign and optimise postpartum care. BMJ Innovations. 2022;8:37-41.
7. Østbye T, Yarnall KS, Krause KM, et al. Is there time for management of patients with chronic diseases in primary care? Ann Fam Med. 2005;3(3):209-214. doi:10.1370/afm.310
8. Ganguli I, Shi Z, E. Orav J, et al. Declining use of primary care among commercially insured adults in the united states, 2008–2016. Ann Intern Med. 2020;172:240-247. doi:10.7326/M19-1834
Serum 25-hydroxyvitamin D levels inversely correlated with albuminuria in patients with diabetes
Key clinical point: Serum level of 25-hydroxyvitamin D (25[OH]D) was inversely associated with albuminuria as an indicator of diabetic nephropathy in patients with type 2 diabetes mellitus (T2D), with the prevalence of macroalbuminuria increasing when the serum 25(OH)D level was <20 ng/mL.
Major finding: A strong negative association was observed between the serum 25(OH)D level and severity of albuminuria (r, −0.257; P < .001), with the prevalence of vitamin D deficiency being 61.8%, 33.3%, and 24.0% in patients with macroalbuminuria, microalbuminuria, and normoalbuminuria, respectively.
Study details: This was a cross-sectional study including 200 patients with T2D, of which 100, 66, and 34 had normoalbuminuria, microalbuminuria, and macroalbuminuria, respectively.
Disclosures: This study was supported by Shiraz University of Medical Sciences, Iran. The authors declared no competing interests.
Source: Zomorodian SA et al. Assessment of the relationship between 25-hydroxyvitamin D and albuminuria in type 2 diabetes mellitus. BMC Endocr Disord. 2022;22:171 (Jul 4). Doi: 10.1186/s12902-022-01088-2
Key clinical point: Serum level of 25-hydroxyvitamin D (25[OH]D) was inversely associated with albuminuria as an indicator of diabetic nephropathy in patients with type 2 diabetes mellitus (T2D), with the prevalence of macroalbuminuria increasing when the serum 25(OH)D level was <20 ng/mL.
Major finding: A strong negative association was observed between the serum 25(OH)D level and severity of albuminuria (r, −0.257; P < .001), with the prevalence of vitamin D deficiency being 61.8%, 33.3%, and 24.0% in patients with macroalbuminuria, microalbuminuria, and normoalbuminuria, respectively.
Study details: This was a cross-sectional study including 200 patients with T2D, of which 100, 66, and 34 had normoalbuminuria, microalbuminuria, and macroalbuminuria, respectively.
Disclosures: This study was supported by Shiraz University of Medical Sciences, Iran. The authors declared no competing interests.
Source: Zomorodian SA et al. Assessment of the relationship between 25-hydroxyvitamin D and albuminuria in type 2 diabetes mellitus. BMC Endocr Disord. 2022;22:171 (Jul 4). Doi: 10.1186/s12902-022-01088-2
Key clinical point: Serum level of 25-hydroxyvitamin D (25[OH]D) was inversely associated with albuminuria as an indicator of diabetic nephropathy in patients with type 2 diabetes mellitus (T2D), with the prevalence of macroalbuminuria increasing when the serum 25(OH)D level was <20 ng/mL.
Major finding: A strong negative association was observed between the serum 25(OH)D level and severity of albuminuria (r, −0.257; P < .001), with the prevalence of vitamin D deficiency being 61.8%, 33.3%, and 24.0% in patients with macroalbuminuria, microalbuminuria, and normoalbuminuria, respectively.
Study details: This was a cross-sectional study including 200 patients with T2D, of which 100, 66, and 34 had normoalbuminuria, microalbuminuria, and macroalbuminuria, respectively.
Disclosures: This study was supported by Shiraz University of Medical Sciences, Iran. The authors declared no competing interests.
Source: Zomorodian SA et al. Assessment of the relationship between 25-hydroxyvitamin D and albuminuria in type 2 diabetes mellitus. BMC Endocr Disord. 2022;22:171 (Jul 4). Doi: 10.1186/s12902-022-01088-2
Sarcopenia index significantly linked to subclinical atherosclerosis prevalence in T2D
Key clinical point: The sarcopenia index (SI; serum creatinine/serum cystatin C ratio) is significantly associated with the prevalence of subclinical atherosclerosis in patients with type 2 diabetes mellitus (T2D).
Major finding: After adjusting for all confounders, SI was significantly associated with the prevalence of subclinical atherosclerosis (adjusted odds ratio 0.95; P = .015).
Study details: The data come from a cross-sectional study of 174 patients with T2D, of which 43.7% were diagnosed with subclinical atherosclerosis.
Disclosures: The study received no specific funding. Some authors declared receiving honoraria, personal fees, r research grants from various sources.
Source: Hashimoto Y et al. Relationship between serum creatinine to cystatin C ratio and subclinical atherosclerosis in patients with type 2 diabetes. BMJ Open Diabetes Res Care. 2022;10:e002910 (Jun 23). Doi: 10.1136/bmjdrc-2022-002910
Key clinical point: The sarcopenia index (SI; serum creatinine/serum cystatin C ratio) is significantly associated with the prevalence of subclinical atherosclerosis in patients with type 2 diabetes mellitus (T2D).
Major finding: After adjusting for all confounders, SI was significantly associated with the prevalence of subclinical atherosclerosis (adjusted odds ratio 0.95; P = .015).
Study details: The data come from a cross-sectional study of 174 patients with T2D, of which 43.7% were diagnosed with subclinical atherosclerosis.
Disclosures: The study received no specific funding. Some authors declared receiving honoraria, personal fees, r research grants from various sources.
Source: Hashimoto Y et al. Relationship between serum creatinine to cystatin C ratio and subclinical atherosclerosis in patients with type 2 diabetes. BMJ Open Diabetes Res Care. 2022;10:e002910 (Jun 23). Doi: 10.1136/bmjdrc-2022-002910
Key clinical point: The sarcopenia index (SI; serum creatinine/serum cystatin C ratio) is significantly associated with the prevalence of subclinical atherosclerosis in patients with type 2 diabetes mellitus (T2D).
Major finding: After adjusting for all confounders, SI was significantly associated with the prevalence of subclinical atherosclerosis (adjusted odds ratio 0.95; P = .015).
Study details: The data come from a cross-sectional study of 174 patients with T2D, of which 43.7% were diagnosed with subclinical atherosclerosis.
Disclosures: The study received no specific funding. Some authors declared receiving honoraria, personal fees, r research grants from various sources.
Source: Hashimoto Y et al. Relationship between serum creatinine to cystatin C ratio and subclinical atherosclerosis in patients with type 2 diabetes. BMJ Open Diabetes Res Care. 2022;10:e002910 (Jun 23). Doi: 10.1136/bmjdrc-2022-002910
Real-world insights of initiating or switching to insulin degludec/aspart in T2D
Key clinical point: In patients with type 2 diabetes (T2D), initiating or switching to insulin degludec/ aspart (IDegAsp) from other antidiabetic treatments was associated with improved glycemic control, lower basal insulin dose requirement in insulin-experienced patients, and lower rates of hypoglycemia.
Major finding: Patients with T2D initiating or switching to IDegAsp had a significant reduction in glycated hemoglobin (estimated difference [Δ] −1.4%; P < .0001), basal insulin dose requirements in insulin-experienced participants (Δ −2.3 units; P = .0004), and rates of hypoglycemia (P < .001).
Study details: Findings are from a real-world, prospective study including 1102 patients with T2D who initiated or switched to IDegAsp from other antidiabetic treatments.
Disclosures: This study was funded by Novo Nordisk. Some authors declared being employees and shareholders of Novo Nordisk. Some authors declared receiving speaker or consulting honoraria, research contracts, and teaching or research sponsorships; being consultants; or serving as advisory board or speaker panel members for various sources, including Novo Nordisk.
Source: Fulcher GR et al. Initiating or switching to insulin degludec/insulin aspart in adults with type 2 diabetes: A real-world, prospective, non-interventional study across six countries. Adv Ther. 2022 (Jun 25). Doi: 10.1007/s12325-022-02212-3
Key clinical point: In patients with type 2 diabetes (T2D), initiating or switching to insulin degludec/ aspart (IDegAsp) from other antidiabetic treatments was associated with improved glycemic control, lower basal insulin dose requirement in insulin-experienced patients, and lower rates of hypoglycemia.
Major finding: Patients with T2D initiating or switching to IDegAsp had a significant reduction in glycated hemoglobin (estimated difference [Δ] −1.4%; P < .0001), basal insulin dose requirements in insulin-experienced participants (Δ −2.3 units; P = .0004), and rates of hypoglycemia (P < .001).
Study details: Findings are from a real-world, prospective study including 1102 patients with T2D who initiated or switched to IDegAsp from other antidiabetic treatments.
Disclosures: This study was funded by Novo Nordisk. Some authors declared being employees and shareholders of Novo Nordisk. Some authors declared receiving speaker or consulting honoraria, research contracts, and teaching or research sponsorships; being consultants; or serving as advisory board or speaker panel members for various sources, including Novo Nordisk.
Source: Fulcher GR et al. Initiating or switching to insulin degludec/insulin aspart in adults with type 2 diabetes: A real-world, prospective, non-interventional study across six countries. Adv Ther. 2022 (Jun 25). Doi: 10.1007/s12325-022-02212-3
Key clinical point: In patients with type 2 diabetes (T2D), initiating or switching to insulin degludec/ aspart (IDegAsp) from other antidiabetic treatments was associated with improved glycemic control, lower basal insulin dose requirement in insulin-experienced patients, and lower rates of hypoglycemia.
Major finding: Patients with T2D initiating or switching to IDegAsp had a significant reduction in glycated hemoglobin (estimated difference [Δ] −1.4%; P < .0001), basal insulin dose requirements in insulin-experienced participants (Δ −2.3 units; P = .0004), and rates of hypoglycemia (P < .001).
Study details: Findings are from a real-world, prospective study including 1102 patients with T2D who initiated or switched to IDegAsp from other antidiabetic treatments.
Disclosures: This study was funded by Novo Nordisk. Some authors declared being employees and shareholders of Novo Nordisk. Some authors declared receiving speaker or consulting honoraria, research contracts, and teaching or research sponsorships; being consultants; or serving as advisory board or speaker panel members for various sources, including Novo Nordisk.
Source: Fulcher GR et al. Initiating or switching to insulin degludec/insulin aspart in adults with type 2 diabetes: A real-world, prospective, non-interventional study across six countries. Adv Ther. 2022 (Jun 25). Doi: 10.1007/s12325-022-02212-3
SGLT2i vs DPP4i lowers death risk in T2D
Key clinical point: Sodium-glucose cotransporter 2 inhibitor (SGLT2i) use in patients with type 2 diabetes mellitus (T2D) is associated with a significantly lower risk for all-cause and cause-specific death compared with dipeptidyl peptidase 4 inhibitor (DPP4i) use.
Major finding: Patients receiving SGLT2i vs DPP4i had a lower risk for all-cause death (adjusted hazard ratio [aHR] 0.66; P < .001), cardiovascular death (aHR 0.68; P < .001), cancer death (aHR 0.73; P = .003), and noncancer and nonvascular death (aHR 0.62; P < .001).
Study details: This nationwide retrospective cohort study matched patients with T2D who initiated SGLT2i (n = 53,264) with those who initiated DPP4i (n = 53,264) using propensity score matching.
Disclosures: This study was partly supported by grants from China Medical University Hospital, Ditmanson Medical Foundation Chiayi Christian Hospital, and the Ministry of Science and Technology of Taiwan. No competing interests were declared.
Source: Chung M-C et al. Efficacy and safety of DBPR108 (prusogliptin) as an add-on to metformin therapy in patients with type 2 diabetes mellitus: A 24-week, multi-center, randomized, double-blind, placebo-controlled, superiority, phase III clinical trial. Sci Rep. 2022;12:10147 (Jun 16). Doi: 10.1038/s41598-022-13760-7
Key clinical point: Sodium-glucose cotransporter 2 inhibitor (SGLT2i) use in patients with type 2 diabetes mellitus (T2D) is associated with a significantly lower risk for all-cause and cause-specific death compared with dipeptidyl peptidase 4 inhibitor (DPP4i) use.
Major finding: Patients receiving SGLT2i vs DPP4i had a lower risk for all-cause death (adjusted hazard ratio [aHR] 0.66; P < .001), cardiovascular death (aHR 0.68; P < .001), cancer death (aHR 0.73; P = .003), and noncancer and nonvascular death (aHR 0.62; P < .001).
Study details: This nationwide retrospective cohort study matched patients with T2D who initiated SGLT2i (n = 53,264) with those who initiated DPP4i (n = 53,264) using propensity score matching.
Disclosures: This study was partly supported by grants from China Medical University Hospital, Ditmanson Medical Foundation Chiayi Christian Hospital, and the Ministry of Science and Technology of Taiwan. No competing interests were declared.
Source: Chung M-C et al. Efficacy and safety of DBPR108 (prusogliptin) as an add-on to metformin therapy in patients with type 2 diabetes mellitus: A 24-week, multi-center, randomized, double-blind, placebo-controlled, superiority, phase III clinical trial. Sci Rep. 2022;12:10147 (Jun 16). Doi: 10.1038/s41598-022-13760-7
Key clinical point: Sodium-glucose cotransporter 2 inhibitor (SGLT2i) use in patients with type 2 diabetes mellitus (T2D) is associated with a significantly lower risk for all-cause and cause-specific death compared with dipeptidyl peptidase 4 inhibitor (DPP4i) use.
Major finding: Patients receiving SGLT2i vs DPP4i had a lower risk for all-cause death (adjusted hazard ratio [aHR] 0.66; P < .001), cardiovascular death (aHR 0.68; P < .001), cancer death (aHR 0.73; P = .003), and noncancer and nonvascular death (aHR 0.62; P < .001).
Study details: This nationwide retrospective cohort study matched patients with T2D who initiated SGLT2i (n = 53,264) with those who initiated DPP4i (n = 53,264) using propensity score matching.
Disclosures: This study was partly supported by grants from China Medical University Hospital, Ditmanson Medical Foundation Chiayi Christian Hospital, and the Ministry of Science and Technology of Taiwan. No competing interests were declared.
Source: Chung M-C et al. Efficacy and safety of DBPR108 (prusogliptin) as an add-on to metformin therapy in patients with type 2 diabetes mellitus: A 24-week, multi-center, randomized, double-blind, placebo-controlled, superiority, phase III clinical trial. Sci Rep. 2022;12:10147 (Jun 16). Doi: 10.1038/s41598-022-13760-7
Prusogliptin add-on to metformin safe and effective in T2D
Key clinical point: Prusogliptin as an add-on therapy to metformin was superior to metformin monotherapy in improving glycemic control and was safe and well tolerated in patients with type 2 diabetes mellitus (T2D) inadequately controlled with metformin.
Major finding: At week 24, prusogliptin + metformin vs metformin + placebo led to significantly higher reductions in glycated hemoglobin (least squares mean change [LSM] −0.70% vs −0.07%; P < .001), fasting plasma glucose (LSM −0.63 vs 0.07 mmol/L; P = .025), and 2-hour postprandial plasma glucose (LSM −2.43 vs −0.70 mmol/L; P < .001) levels, with the incidence of adverse events being similar between the treatment groups.
Study details: Findings are from a 24-week, superiority, phase 3 trial including 206 patients with T2D with blood glucose levels inadequately controlled on metformin who were randomly assigned to receive prusogliptin + metformin (n = 138) or placebo + metformin (n = 68).
Disclosures: This study was funded by the CSPC Zhongqi Pharmaceutical Technology Co., Ltd. Some authors are employees of CSPC Zhongqi Pharmaceutical Technology.
Source: Xu J et al. Efficacy and safety of DBPR108 (prusogliptin) as an add-on to metformin therapy in patients with type 2 diabetes mellitus: A 24-week, multi-center, randomized, double-blind, placebo-controlled, superiority, phase III clinical trial. Diabetes Obes Metab. 2022 (Jul 6). Doi: 10.1111/dom.14810
Key clinical point: Prusogliptin as an add-on therapy to metformin was superior to metformin monotherapy in improving glycemic control and was safe and well tolerated in patients with type 2 diabetes mellitus (T2D) inadequately controlled with metformin.
Major finding: At week 24, prusogliptin + metformin vs metformin + placebo led to significantly higher reductions in glycated hemoglobin (least squares mean change [LSM] −0.70% vs −0.07%; P < .001), fasting plasma glucose (LSM −0.63 vs 0.07 mmol/L; P = .025), and 2-hour postprandial plasma glucose (LSM −2.43 vs −0.70 mmol/L; P < .001) levels, with the incidence of adverse events being similar between the treatment groups.
Study details: Findings are from a 24-week, superiority, phase 3 trial including 206 patients with T2D with blood glucose levels inadequately controlled on metformin who were randomly assigned to receive prusogliptin + metformin (n = 138) or placebo + metformin (n = 68).
Disclosures: This study was funded by the CSPC Zhongqi Pharmaceutical Technology Co., Ltd. Some authors are employees of CSPC Zhongqi Pharmaceutical Technology.
Source: Xu J et al. Efficacy and safety of DBPR108 (prusogliptin) as an add-on to metformin therapy in patients with type 2 diabetes mellitus: A 24-week, multi-center, randomized, double-blind, placebo-controlled, superiority, phase III clinical trial. Diabetes Obes Metab. 2022 (Jul 6). Doi: 10.1111/dom.14810
Key clinical point: Prusogliptin as an add-on therapy to metformin was superior to metformin monotherapy in improving glycemic control and was safe and well tolerated in patients with type 2 diabetes mellitus (T2D) inadequately controlled with metformin.
Major finding: At week 24, prusogliptin + metformin vs metformin + placebo led to significantly higher reductions in glycated hemoglobin (least squares mean change [LSM] −0.70% vs −0.07%; P < .001), fasting plasma glucose (LSM −0.63 vs 0.07 mmol/L; P = .025), and 2-hour postprandial plasma glucose (LSM −2.43 vs −0.70 mmol/L; P < .001) levels, with the incidence of adverse events being similar between the treatment groups.
Study details: Findings are from a 24-week, superiority, phase 3 trial including 206 patients with T2D with blood glucose levels inadequately controlled on metformin who were randomly assigned to receive prusogliptin + metformin (n = 138) or placebo + metformin (n = 68).
Disclosures: This study was funded by the CSPC Zhongqi Pharmaceutical Technology Co., Ltd. Some authors are employees of CSPC Zhongqi Pharmaceutical Technology.
Source: Xu J et al. Efficacy and safety of DBPR108 (prusogliptin) as an add-on to metformin therapy in patients with type 2 diabetes mellitus: A 24-week, multi-center, randomized, double-blind, placebo-controlled, superiority, phase III clinical trial. Diabetes Obes Metab. 2022 (Jul 6). Doi: 10.1111/dom.14810
Initiating semaglutide improves outcomes in patients on high daily doses of insulin
Key clinical point: Initiation of semaglutide in patients with type 2 diabetes mellitus (T2D) on high daily doses of insulin at baseline led to a significant improvement in glycemic control, body weight, and reduction in total daily dose (TDD) of insulin.
Major finding: From baseline to 6 months, the TDD of insulin (183 ± 98 to 143 ± 99 units, respectively), glycated hemoglobin level (8.9% ± 1.3% to 7.6% ± 1.5%, respectively), and body weight (123.9 ± 23.5 to 118.9 ± 22.9 kg, respectively; all P < .001) reduced significantly in patients on high daily doses of insulin who received semaglutide.
Study details: Findings are from a retrospective analysis including 72 patients with T2D on high TDD of insulin (≥100 units) who were prescribed semaglutide.
Disclosures: This study received no specific funding. The authors declared no conflicts of interest.
Source: Meyer J et al. The effects of adding semaglutide to high daily dose insulin regimens in patients with type 2 diabetes. Ann Pharmacother. 2022 (Jul 1). Doi: 10.1177/10600280221107381
Key clinical point: Initiation of semaglutide in patients with type 2 diabetes mellitus (T2D) on high daily doses of insulin at baseline led to a significant improvement in glycemic control, body weight, and reduction in total daily dose (TDD) of insulin.
Major finding: From baseline to 6 months, the TDD of insulin (183 ± 98 to 143 ± 99 units, respectively), glycated hemoglobin level (8.9% ± 1.3% to 7.6% ± 1.5%, respectively), and body weight (123.9 ± 23.5 to 118.9 ± 22.9 kg, respectively; all P < .001) reduced significantly in patients on high daily doses of insulin who received semaglutide.
Study details: Findings are from a retrospective analysis including 72 patients with T2D on high TDD of insulin (≥100 units) who were prescribed semaglutide.
Disclosures: This study received no specific funding. The authors declared no conflicts of interest.
Source: Meyer J et al. The effects of adding semaglutide to high daily dose insulin regimens in patients with type 2 diabetes. Ann Pharmacother. 2022 (Jul 1). Doi: 10.1177/10600280221107381
Key clinical point: Initiation of semaglutide in patients with type 2 diabetes mellitus (T2D) on high daily doses of insulin at baseline led to a significant improvement in glycemic control, body weight, and reduction in total daily dose (TDD) of insulin.
Major finding: From baseline to 6 months, the TDD of insulin (183 ± 98 to 143 ± 99 units, respectively), glycated hemoglobin level (8.9% ± 1.3% to 7.6% ± 1.5%, respectively), and body weight (123.9 ± 23.5 to 118.9 ± 22.9 kg, respectively; all P < .001) reduced significantly in patients on high daily doses of insulin who received semaglutide.
Study details: Findings are from a retrospective analysis including 72 patients with T2D on high TDD of insulin (≥100 units) who were prescribed semaglutide.
Disclosures: This study received no specific funding. The authors declared no conflicts of interest.
Source: Meyer J et al. The effects of adding semaglutide to high daily dose insulin regimens in patients with type 2 diabetes. Ann Pharmacother. 2022 (Jul 1). Doi: 10.1177/10600280221107381


