User login
ASCO: Many women with triple-negative breast cancer aren’t screened for BRCA
SAN FRANCISCO – Many younger women diagnosed with triple-negative breast cancers do not get tested for BRCA, despite guideline recommendations, investigators report.
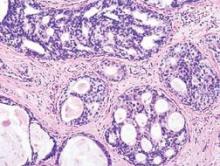
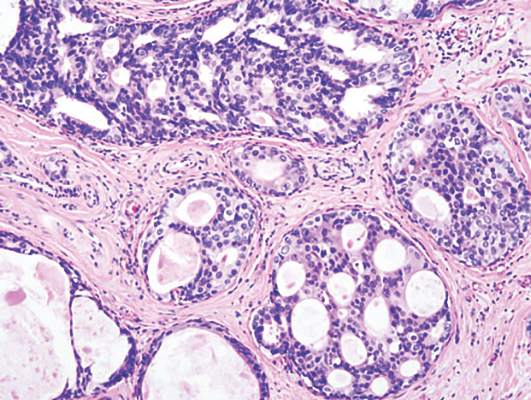
Among 173 women with triple-negative tumors -- lacking the HER2, estrogen and progesterone receptors –17% of those who should have been tested for BRCA according to National Comprehensive Cancer Network (NCCN) guidelines, were not tested.
Women less likely to be tested were those 55 years or older, African Americans, those who list Medicaid as their primary form of insurance, and those with stage 3 disease, reported Staci Aubry, a 4th-year medical student at Rush University Medical Center in Chicago, and her associates.
In an interview, Ms. Aubry said that some of the women who were eligible for genetic testing under the guidelines simply declined it.
“Often patients, if they didn’t have a daughter or if they were older and were in the 50 to 60 [year-old] range, even though they’re still included in the NCCN guidelines to be screened, still refused to be tested,” she said.
NCCN guidelines on genetic and familial high-risk assessment for breast and ovarian cancer susceptibility recommend BRCA genetic testing for all women below age 60 years who are diagnosed with triple-negative breast cancer.
“Some histopathologic features have reported to occur more frequently in breast cancers characterized by a BRCA1 or BRCA2 mutation. For example, several studies have shown that BRCA1 breast cancer is more likely to be characterized as ER-/PR-negative and HER2-negative (i.e., “triple negative”), the guidelines note.
To see whether clinicians were adhering to this recommendation, the investigators searched the Commission on Cancer registry tumor database for cases of triple-negative breast cancers diagnosed from 2006 through 2013.
They identified 173 patients, 105 of whom were younger than 60 and thus recommended for screening. Of this group, 87 (83%) were tested for BRCA, and 15 were found to be BRCA positive, but the remaining 18 patients (17%) were not tested.
When the authors looked at demographic and clinical factors that might have accounted for the differences between women who were tested for the gene and those who were not, four factors stood out. Women who did not get tested were more likely to be 55 or older (P = .002), African American (P = .001), Medicaid insured (P = .021) or to have American Joint Commission on Cancer stage 3 disease (P = .014).
“The main message for clinicians is to be aware that these disparities exist, and keep in mind that these four groups – people who are under Medicaid insurance, between 55 and 60 years of age, African American, or who have higher disease stage – should be targeted for screening,” Ms. Aubry said.
SAN FRANCISCO – Many younger women diagnosed with triple-negative breast cancers do not get tested for BRCA, despite guideline recommendations, investigators report.
Among 173 women with triple-negative tumors -- lacking the HER2, estrogen and progesterone receptors –17% of those who should have been tested for BRCA according to National Comprehensive Cancer Network (NCCN) guidelines, were not tested.
Women less likely to be tested were those 55 years or older, African Americans, those who list Medicaid as their primary form of insurance, and those with stage 3 disease, reported Staci Aubry, a 4th-year medical student at Rush University Medical Center in Chicago, and her associates.
In an interview, Ms. Aubry said that some of the women who were eligible for genetic testing under the guidelines simply declined it.
“Often patients, if they didn’t have a daughter or if they were older and were in the 50 to 60 [year-old] range, even though they’re still included in the NCCN guidelines to be screened, still refused to be tested,” she said.
NCCN guidelines on genetic and familial high-risk assessment for breast and ovarian cancer susceptibility recommend BRCA genetic testing for all women below age 60 years who are diagnosed with triple-negative breast cancer.
“Some histopathologic features have reported to occur more frequently in breast cancers characterized by a BRCA1 or BRCA2 mutation. For example, several studies have shown that BRCA1 breast cancer is more likely to be characterized as ER-/PR-negative and HER2-negative (i.e., “triple negative”), the guidelines note.
To see whether clinicians were adhering to this recommendation, the investigators searched the Commission on Cancer registry tumor database for cases of triple-negative breast cancers diagnosed from 2006 through 2013.
They identified 173 patients, 105 of whom were younger than 60 and thus recommended for screening. Of this group, 87 (83%) were tested for BRCA, and 15 were found to be BRCA positive, but the remaining 18 patients (17%) were not tested.
When the authors looked at demographic and clinical factors that might have accounted for the differences between women who were tested for the gene and those who were not, four factors stood out. Women who did not get tested were more likely to be 55 or older (P = .002), African American (P = .001), Medicaid insured (P = .021) or to have American Joint Commission on Cancer stage 3 disease (P = .014).
“The main message for clinicians is to be aware that these disparities exist, and keep in mind that these four groups – people who are under Medicaid insurance, between 55 and 60 years of age, African American, or who have higher disease stage – should be targeted for screening,” Ms. Aubry said.
SAN FRANCISCO – Many younger women diagnosed with triple-negative breast cancers do not get tested for BRCA, despite guideline recommendations, investigators report.
Among 173 women with triple-negative tumors -- lacking the HER2, estrogen and progesterone receptors –17% of those who should have been tested for BRCA according to National Comprehensive Cancer Network (NCCN) guidelines, were not tested.
Women less likely to be tested were those 55 years or older, African Americans, those who list Medicaid as their primary form of insurance, and those with stage 3 disease, reported Staci Aubry, a 4th-year medical student at Rush University Medical Center in Chicago, and her associates.
In an interview, Ms. Aubry said that some of the women who were eligible for genetic testing under the guidelines simply declined it.
“Often patients, if they didn’t have a daughter or if they were older and were in the 50 to 60 [year-old] range, even though they’re still included in the NCCN guidelines to be screened, still refused to be tested,” she said.
NCCN guidelines on genetic and familial high-risk assessment for breast and ovarian cancer susceptibility recommend BRCA genetic testing for all women below age 60 years who are diagnosed with triple-negative breast cancer.
“Some histopathologic features have reported to occur more frequently in breast cancers characterized by a BRCA1 or BRCA2 mutation. For example, several studies have shown that BRCA1 breast cancer is more likely to be characterized as ER-/PR-negative and HER2-negative (i.e., “triple negative”), the guidelines note.
To see whether clinicians were adhering to this recommendation, the investigators searched the Commission on Cancer registry tumor database for cases of triple-negative breast cancers diagnosed from 2006 through 2013.
They identified 173 patients, 105 of whom were younger than 60 and thus recommended for screening. Of this group, 87 (83%) were tested for BRCA, and 15 were found to be BRCA positive, but the remaining 18 patients (17%) were not tested.
When the authors looked at demographic and clinical factors that might have accounted for the differences between women who were tested for the gene and those who were not, four factors stood out. Women who did not get tested were more likely to be 55 or older (P = .002), African American (P = .001), Medicaid insured (P = .021) or to have American Joint Commission on Cancer stage 3 disease (P = .014).
“The main message for clinicians is to be aware that these disparities exist, and keep in mind that these four groups – people who are under Medicaid insurance, between 55 and 60 years of age, African American, or who have higher disease stage – should be targeted for screening,” Ms. Aubry said.
AT THE ASCO BREAST CANCER SYMPOSIUM
Key clinical point: Guidelines recommend screening for BRCA in women under 60 years of age with triple-negative breast cancer.
Major finding: Of 105 women eligible for BRCA screening, 18 (17%) were not tested.
Data source: Review of registry data on 173 women with triple-negative breast cancer, including 105 under age 60 years and therefore recommended for BRCA testing.
Disclosures: The investigators did not disclosed a funding source. Ms. Aubry reported having no conflicts of interest.
ASCO: Model predicts risk for breast cancer from atypical hyperplasia
SAN FRANCISCO – A woman’s age at biopsy and the number of atypical hyperplasia foci appear to be good predictors of risk for subsequent breast cancer, investigators say.
A review of pathology records and medical history on more than 13,000 women with benign breast disease showed that a predictive model including age and atypia effectively identified those women with atypical hyperplasia at highest risk for developing breast cancer, reported Dr. Amy C. Degnim from the Mayo Clinic in Rochester, Minnesota.
“In our country, approximately 10% of all benign breast biopsies are showing atypical hyperplasia, and with about a million women who undergo biopsy every year, this about 100,000 women every year who are diagnosed with atypical hyperplasia. These women are known to have an increased risk of breast cancer,” Dr. Degnim said at a breast symposium here jointly sponsored by the American Society of Clinical Oncology, American Society For Radiation Oncology, and Society of Surgical Oncology.
Current risk prediction models such as the Breast Cancer Risk Assessment Tool and IBIS Breast Cancer Risk Evaluation Tool tend to underestimate or overestimate risk of breast cancer in women with atypical hyperplasia, prompting the investigators to explore developing a reliable prediction tool, she said
The investigators identified a cohort of 13,538 women diagnosed with benign breast disease at the Mayo Clinic from 1967 through 2001, and they found data on 699 with atypical hyperplasia confirmed by pathology review blinded to outcomes. They collected data on clinical and histologic features and identified breast cancer event through review of medical records and questionnaires.
In addition to the Mayo patients used to develop the model, the authors tested it in a validation sample of 461 women with atypical hyperplasia treated at Vanderibilt University in Nashville, Tennessee.
They rounded up data on potential contributors to a risk-prediction model using Lasso-identified variables that they then plugged into a Cox regression model.
They found that of all possible co-variates, including body-mass index at biopsy, age at menarche, indication for biopsy, number of live births, breastfeeding, family features, and histologic features such as involution or calcifications, only the age at biopsy and number of foci of atypical hyperplasia remained as robust predictors for breast cancer risk.
They then tested the model on data from the 699 women in the development set, who had had a total of 142 breast cancer events over a median follow-up of 8.1 years, and in the external validation set of 461 women who had a total of 114 breast cancer events over a median follow-up of 11.4 years.
They found that the concordance between the prediction model and the actual outcomes at 5, 10, and 30 years was 0.607. 0.633, and 0.607, respectively, for the development set.
The model performed a little less well for the validation set, with 5-, 10-, and 30-year concordance of 0.557, 0.584, and 0.557.
Based on their findings, they developed a risk-prediction table showing relative risk for women by age and number of foci (1, 2, or 3 or more). For example, the table shows a 5-year absolute risk of 9.69% for a woman 70-74 years in age at the time of biopsy with 3 or more foci of atypia, compared with just 4.5% for a woman of the same age with only 1 focus. There are risk prediction tables for 5-, 10-, and 30-year absolute risk.
In her commentary on the study, Dr. A. Marilyn Leitch from the University of Texas Southwestern Medical Center in Dallas, noted that the risk-prediction model relies on specific detail in the pathology report for the description of the number of foci of atypical hyperplasia.
“This model can provide estimates that are more informative to the patient than ‘4.5 times risk of breast cancer.’ However, while a high score might motivate a patient for intervention, most patients in the less than 20% risk category, and so we may not have as much persuasion from this model,” she said.
SAN FRANCISCO – A woman’s age at biopsy and the number of atypical hyperplasia foci appear to be good predictors of risk for subsequent breast cancer, investigators say.
A review of pathology records and medical history on more than 13,000 women with benign breast disease showed that a predictive model including age and atypia effectively identified those women with atypical hyperplasia at highest risk for developing breast cancer, reported Dr. Amy C. Degnim from the Mayo Clinic in Rochester, Minnesota.
“In our country, approximately 10% of all benign breast biopsies are showing atypical hyperplasia, and with about a million women who undergo biopsy every year, this about 100,000 women every year who are diagnosed with atypical hyperplasia. These women are known to have an increased risk of breast cancer,” Dr. Degnim said at a breast symposium here jointly sponsored by the American Society of Clinical Oncology, American Society For Radiation Oncology, and Society of Surgical Oncology.
Current risk prediction models such as the Breast Cancer Risk Assessment Tool and IBIS Breast Cancer Risk Evaluation Tool tend to underestimate or overestimate risk of breast cancer in women with atypical hyperplasia, prompting the investigators to explore developing a reliable prediction tool, she said
The investigators identified a cohort of 13,538 women diagnosed with benign breast disease at the Mayo Clinic from 1967 through 2001, and they found data on 699 with atypical hyperplasia confirmed by pathology review blinded to outcomes. They collected data on clinical and histologic features and identified breast cancer event through review of medical records and questionnaires.
In addition to the Mayo patients used to develop the model, the authors tested it in a validation sample of 461 women with atypical hyperplasia treated at Vanderibilt University in Nashville, Tennessee.
They rounded up data on potential contributors to a risk-prediction model using Lasso-identified variables that they then plugged into a Cox regression model.
They found that of all possible co-variates, including body-mass index at biopsy, age at menarche, indication for biopsy, number of live births, breastfeeding, family features, and histologic features such as involution or calcifications, only the age at biopsy and number of foci of atypical hyperplasia remained as robust predictors for breast cancer risk.
They then tested the model on data from the 699 women in the development set, who had had a total of 142 breast cancer events over a median follow-up of 8.1 years, and in the external validation set of 461 women who had a total of 114 breast cancer events over a median follow-up of 11.4 years.
They found that the concordance between the prediction model and the actual outcomes at 5, 10, and 30 years was 0.607. 0.633, and 0.607, respectively, for the development set.
The model performed a little less well for the validation set, with 5-, 10-, and 30-year concordance of 0.557, 0.584, and 0.557.
Based on their findings, they developed a risk-prediction table showing relative risk for women by age and number of foci (1, 2, or 3 or more). For example, the table shows a 5-year absolute risk of 9.69% for a woman 70-74 years in age at the time of biopsy with 3 or more foci of atypia, compared with just 4.5% for a woman of the same age with only 1 focus. There are risk prediction tables for 5-, 10-, and 30-year absolute risk.
In her commentary on the study, Dr. A. Marilyn Leitch from the University of Texas Southwestern Medical Center in Dallas, noted that the risk-prediction model relies on specific detail in the pathology report for the description of the number of foci of atypical hyperplasia.
“This model can provide estimates that are more informative to the patient than ‘4.5 times risk of breast cancer.’ However, while a high score might motivate a patient for intervention, most patients in the less than 20% risk category, and so we may not have as much persuasion from this model,” she said.
SAN FRANCISCO – A woman’s age at biopsy and the number of atypical hyperplasia foci appear to be good predictors of risk for subsequent breast cancer, investigators say.
A review of pathology records and medical history on more than 13,000 women with benign breast disease showed that a predictive model including age and atypia effectively identified those women with atypical hyperplasia at highest risk for developing breast cancer, reported Dr. Amy C. Degnim from the Mayo Clinic in Rochester, Minnesota.
“In our country, approximately 10% of all benign breast biopsies are showing atypical hyperplasia, and with about a million women who undergo biopsy every year, this about 100,000 women every year who are diagnosed with atypical hyperplasia. These women are known to have an increased risk of breast cancer,” Dr. Degnim said at a breast symposium here jointly sponsored by the American Society of Clinical Oncology, American Society For Radiation Oncology, and Society of Surgical Oncology.
Current risk prediction models such as the Breast Cancer Risk Assessment Tool and IBIS Breast Cancer Risk Evaluation Tool tend to underestimate or overestimate risk of breast cancer in women with atypical hyperplasia, prompting the investigators to explore developing a reliable prediction tool, she said
The investigators identified a cohort of 13,538 women diagnosed with benign breast disease at the Mayo Clinic from 1967 through 2001, and they found data on 699 with atypical hyperplasia confirmed by pathology review blinded to outcomes. They collected data on clinical and histologic features and identified breast cancer event through review of medical records and questionnaires.
In addition to the Mayo patients used to develop the model, the authors tested it in a validation sample of 461 women with atypical hyperplasia treated at Vanderibilt University in Nashville, Tennessee.
They rounded up data on potential contributors to a risk-prediction model using Lasso-identified variables that they then plugged into a Cox regression model.
They found that of all possible co-variates, including body-mass index at biopsy, age at menarche, indication for biopsy, number of live births, breastfeeding, family features, and histologic features such as involution or calcifications, only the age at biopsy and number of foci of atypical hyperplasia remained as robust predictors for breast cancer risk.
They then tested the model on data from the 699 women in the development set, who had had a total of 142 breast cancer events over a median follow-up of 8.1 years, and in the external validation set of 461 women who had a total of 114 breast cancer events over a median follow-up of 11.4 years.
They found that the concordance between the prediction model and the actual outcomes at 5, 10, and 30 years was 0.607. 0.633, and 0.607, respectively, for the development set.
The model performed a little less well for the validation set, with 5-, 10-, and 30-year concordance of 0.557, 0.584, and 0.557.
Based on their findings, they developed a risk-prediction table showing relative risk for women by age and number of foci (1, 2, or 3 or more). For example, the table shows a 5-year absolute risk of 9.69% for a woman 70-74 years in age at the time of biopsy with 3 or more foci of atypia, compared with just 4.5% for a woman of the same age with only 1 focus. There are risk prediction tables for 5-, 10-, and 30-year absolute risk.
In her commentary on the study, Dr. A. Marilyn Leitch from the University of Texas Southwestern Medical Center in Dallas, noted that the risk-prediction model relies on specific detail in the pathology report for the description of the number of foci of atypical hyperplasia.
“This model can provide estimates that are more informative to the patient than ‘4.5 times risk of breast cancer.’ However, while a high score might motivate a patient for intervention, most patients in the less than 20% risk category, and so we may not have as much persuasion from this model,” she said.
AT THE ASCO BREAST CANCER SYMPOSIUM
Key clinical point: Age at biopsy and number of atypical hyperplasia foci may predict risk for subsequent breast cancer.
Major finding: A 70-74 year-old woman with three foci of atypia has double the risk for breast cancer as a same-age peer with just one focus.
Data source: Retrospective review of data on 699 women in a model-development set and 461 in a validation set.
Disclosures: The study was supported by grants from the Mayo Clinic and Komen Foundation. Dr. Degnim and Dr. Leitch reported no relevant conflicts of interest.
ASCO: 80-gene profile pegs breast tumors resistant to trastuzumab
SAN FRANCISCO – Molecular subtyping of breast tumors with an 80-gene panel appears to be a better predictor of response or resistance to neoadjuvant therapy than do standard techniques and may identify patients who could benefit from dual HER-2 blockade, according to data presented by investigators.
In a prospective trial, molecular subtyping with the BluePrint 80-gene profile (Agendia) classified 23% of tumors into a different subgroup than did immunohistochemical staining and fluorescent in-situ hybridization (IHC/FISH) and identified a sub-group of so-called triple-positive tumors (positive for the HER-2, estrogen and progresterone receptors) with luminal features.
“This triple-positive, 80-gene luminal subtype, is relatively resistant to neoadjuvant chemotherapy with trastuzumab [Herceptin] alone. Pertuzumab [Perjeta] overcame resistance to chemotherapy with trastuzumab in a substantial proportion of the triple-positive 80-gene luminal subtype,” said Dr. Peter Beitsch from the Dallas (Texas) Surgical group, in an oral session at a breast cancer meeting sponsored jointly by the American Society of Clinical Oncology, American Society of Radiation Oncology, and Society of Surgical Oncology. The data were also presented in a poster session.
“This trial was about 4 years long, and over the course of the trial, we saw the triple-positive luminal group get more and more pathologically complete responses, and we finally figured it out. What had happened was that about midway through the trial, pertuzumab became the standard,” Dr. Beitsch said in an interview.
The investigators used the gene profile to subtype tumors from 889 women from the ages of 18 years through 90 years with histologically proven breast cancer who had not yet undergone excision biopsy, axillary dissection, or breast cancer therapy.
Patients were treated with neoadjuvant therapy at the discretion of their oncologists, provided that the regimen was peer-reviewed or accepted by the National Comprehensive Cancer Network.
As noted before, 23% of tumors were classified into a different subgroup than those assigned by ICH/FISH.
The gene profile re-classified 82 of 167 patients (49%) who were classified by IHC/FISH as triple-positive as BluePrint luminal instead. In this subgroup of patients, the pathologic complete response (pCR) rate was 17%. In contrast, of the 72 patients (43%) classified as triple-positive by IHC/FISH and as BluePrint HER2 by the gene profile, the pCR rate was 59%, a statistically significantly better response rate than patients with the BluePrint luminal designation (P = .0001).
The remaining 13 patients (8%) were reclassified as BluePrint basal.
“We looked at the patients who were triple-positive, luminal subtype, and they had an absymal [chance of achieving pathologic complete response] with just trastuzumab and chemotherapy, but, you add in pertuzumab, and it goes up 10-fold,” Dr. Beitsch said.
“Using this particular molecular platform may indeed allow us to better categorize patients over what ICH alone is able to do,” said Dr. William J. Gradishar from the Lurie Comprehensive Cancer Center at Northwestern University in Chicago, Illinois, the invited discussant.
He noted that therapy guided by intrinsic subtype could be even more potent and might result in superior outcomes.
“Of course, in the end, the only way we’re going to be able to definitively address this is by rigorously looking at this in a prospective manner,” he said.
SAN FRANCISCO – Molecular subtyping of breast tumors with an 80-gene panel appears to be a better predictor of response or resistance to neoadjuvant therapy than do standard techniques and may identify patients who could benefit from dual HER-2 blockade, according to data presented by investigators.
In a prospective trial, molecular subtyping with the BluePrint 80-gene profile (Agendia) classified 23% of tumors into a different subgroup than did immunohistochemical staining and fluorescent in-situ hybridization (IHC/FISH) and identified a sub-group of so-called triple-positive tumors (positive for the HER-2, estrogen and progresterone receptors) with luminal features.
“This triple-positive, 80-gene luminal subtype, is relatively resistant to neoadjuvant chemotherapy with trastuzumab [Herceptin] alone. Pertuzumab [Perjeta] overcame resistance to chemotherapy with trastuzumab in a substantial proportion of the triple-positive 80-gene luminal subtype,” said Dr. Peter Beitsch from the Dallas (Texas) Surgical group, in an oral session at a breast cancer meeting sponsored jointly by the American Society of Clinical Oncology, American Society of Radiation Oncology, and Society of Surgical Oncology. The data were also presented in a poster session.
“This trial was about 4 years long, and over the course of the trial, we saw the triple-positive luminal group get more and more pathologically complete responses, and we finally figured it out. What had happened was that about midway through the trial, pertuzumab became the standard,” Dr. Beitsch said in an interview.
The investigators used the gene profile to subtype tumors from 889 women from the ages of 18 years through 90 years with histologically proven breast cancer who had not yet undergone excision biopsy, axillary dissection, or breast cancer therapy.
Patients were treated with neoadjuvant therapy at the discretion of their oncologists, provided that the regimen was peer-reviewed or accepted by the National Comprehensive Cancer Network.
As noted before, 23% of tumors were classified into a different subgroup than those assigned by ICH/FISH.
The gene profile re-classified 82 of 167 patients (49%) who were classified by IHC/FISH as triple-positive as BluePrint luminal instead. In this subgroup of patients, the pathologic complete response (pCR) rate was 17%. In contrast, of the 72 patients (43%) classified as triple-positive by IHC/FISH and as BluePrint HER2 by the gene profile, the pCR rate was 59%, a statistically significantly better response rate than patients with the BluePrint luminal designation (P = .0001).
The remaining 13 patients (8%) were reclassified as BluePrint basal.
“We looked at the patients who were triple-positive, luminal subtype, and they had an absymal [chance of achieving pathologic complete response] with just trastuzumab and chemotherapy, but, you add in pertuzumab, and it goes up 10-fold,” Dr. Beitsch said.
“Using this particular molecular platform may indeed allow us to better categorize patients over what ICH alone is able to do,” said Dr. William J. Gradishar from the Lurie Comprehensive Cancer Center at Northwestern University in Chicago, Illinois, the invited discussant.
He noted that therapy guided by intrinsic subtype could be even more potent and might result in superior outcomes.
“Of course, in the end, the only way we’re going to be able to definitively address this is by rigorously looking at this in a prospective manner,” he said.
SAN FRANCISCO – Molecular subtyping of breast tumors with an 80-gene panel appears to be a better predictor of response or resistance to neoadjuvant therapy than do standard techniques and may identify patients who could benefit from dual HER-2 blockade, according to data presented by investigators.
In a prospective trial, molecular subtyping with the BluePrint 80-gene profile (Agendia) classified 23% of tumors into a different subgroup than did immunohistochemical staining and fluorescent in-situ hybridization (IHC/FISH) and identified a sub-group of so-called triple-positive tumors (positive for the HER-2, estrogen and progresterone receptors) with luminal features.
“This triple-positive, 80-gene luminal subtype, is relatively resistant to neoadjuvant chemotherapy with trastuzumab [Herceptin] alone. Pertuzumab [Perjeta] overcame resistance to chemotherapy with trastuzumab in a substantial proportion of the triple-positive 80-gene luminal subtype,” said Dr. Peter Beitsch from the Dallas (Texas) Surgical group, in an oral session at a breast cancer meeting sponsored jointly by the American Society of Clinical Oncology, American Society of Radiation Oncology, and Society of Surgical Oncology. The data were also presented in a poster session.
“This trial was about 4 years long, and over the course of the trial, we saw the triple-positive luminal group get more and more pathologically complete responses, and we finally figured it out. What had happened was that about midway through the trial, pertuzumab became the standard,” Dr. Beitsch said in an interview.
The investigators used the gene profile to subtype tumors from 889 women from the ages of 18 years through 90 years with histologically proven breast cancer who had not yet undergone excision biopsy, axillary dissection, or breast cancer therapy.
Patients were treated with neoadjuvant therapy at the discretion of their oncologists, provided that the regimen was peer-reviewed or accepted by the National Comprehensive Cancer Network.
As noted before, 23% of tumors were classified into a different subgroup than those assigned by ICH/FISH.
The gene profile re-classified 82 of 167 patients (49%) who were classified by IHC/FISH as triple-positive as BluePrint luminal instead. In this subgroup of patients, the pathologic complete response (pCR) rate was 17%. In contrast, of the 72 patients (43%) classified as triple-positive by IHC/FISH and as BluePrint HER2 by the gene profile, the pCR rate was 59%, a statistically significantly better response rate than patients with the BluePrint luminal designation (P = .0001).
The remaining 13 patients (8%) were reclassified as BluePrint basal.
“We looked at the patients who were triple-positive, luminal subtype, and they had an absymal [chance of achieving pathologic complete response] with just trastuzumab and chemotherapy, but, you add in pertuzumab, and it goes up 10-fold,” Dr. Beitsch said.
“Using this particular molecular platform may indeed allow us to better categorize patients over what ICH alone is able to do,” said Dr. William J. Gradishar from the Lurie Comprehensive Cancer Center at Northwestern University in Chicago, Illinois, the invited discussant.
He noted that therapy guided by intrinsic subtype could be even more potent and might result in superior outcomes.
“Of course, in the end, the only way we’re going to be able to definitively address this is by rigorously looking at this in a prospective manner,” he said.
AT THE ASCO BREAST CANCER SYMPOSIUM
Key clinical point: An 80-gene profile identified a subtype of breast cancer that may require dual HER2 blockade for effective treatment.
Major finding: A subgroup of patients with triple-positive breast cancer and luminal subtype had a pathologic complete response rate of 17%, compared with 59% of patients with tumors classified as HER2-driven.
Data source: Prospective study of 889 women enrolled in a gene profiling trial.
Disclosures: The study was sponsored by Agendia. Dr. Beitsch disclosed receiving research support and serving as a speaker for the company. Dr. Gradishar reported no relevant conflicts of interest.
Meta-analysis confirms no OS benefit from antiangiogenics for NSCLC patients
DENVER – Once again, angiogenesis inhibitors have been examined for benefit against advanced non–small cell lung cancer (NSCLC) and have been found wanting.
Results of a meta-analysis limited to only phase III randomized controlled trials showed that while angiogenesis inhibitors added to standard of care were associated with modest but significant improvements in progression-free survival (PFS) and response rates (RR), they did not improve overall survival (OS), reported Dr. Jacques Raphael from the Sunnybrook Odette Cancer Centre in Toronto.
“The role of antiangiogenic inhibitors in advanced lung cancer is still questionable,” he said at a world conference on lung cancer sponsored by the International Association for the Study of Lung Cancer.
The findings highlight the need for validated biomarkers that can help clinicians identify which patients are mostly likely to benefit from antiangiogenic therapy, he said.
The investigators conducted a meta-analysis to evaluate the effectiveness of adding any angiogenesis inhibitor or vascular disruptor to standard of care – chemotherapy, tyrosine kinase inhibitors (TKIs), and/or best supportive care – in patients with advanced NSCLC.
Two independent reviewers combed through electronic databases and the websites of international cancer organizations to identify all eligible phase III randomized, controlled trials.
The investigators then performed two separate meta-analyses, each combing data from all other drug trials with data on bevacizumab (Avastin) at either a 7.5-or 15-mg/kg dose.
They identified a total of 25 eligible trials containing data on 19,098 patients, including 9867 treated with an angiogenesis inhibitor and 9231 controls.
For the combined analysis of trials with the 7.5 mg/kg bevacizumab dose, they found no difference in overall survival (hazard ratio, 0.98; 95% confidence interval, 0.96-1.00). Similarly, there was no OS benefit from angiogenesis inhibitors in the analysis including the 15-mg/kg dose of bevacizumab (HR, 0.97; 95% CI, 0.94-1.00).
When they looked at PFS, however, there was a clear benefit in delaying disease progression with angiogenesis inhibitors in both the 7.5 mg/kg bevacizumab analysis (HR, 0.85; 95% CI, 0.79-0.91) and 15 mg/kg analysis (HR 0.85, 95% CI, 0.75-0.88).
Similarly, there was a significant benefit in terms of response rates with the addition of antiangiogenesis in both analyses (odds ratio, 1.61; 95% CI, 1.30-2.01 for the 7.5-mg/kg bevacizumab analysis, and 1.72, 95% CI, 1.39-2.14 for the 15-mg/kg dose).
The investigators also performed subgroup analyses of first-line therapy trials only, trials with monoclonal antibody-based agents, and trials with TKI-based antiangiogenics, and again found PFS and RR benefits, but no OS benefits, Dr. Raphael said.
The failure to see an OS benefit from angiogenesis inhibitors may be attributable to the heterogeneity of drugs in this class (that is, TKIs vs. monoclonal antibodies), clinical trial designs that may have confounded results by allowing crossover to a different drug in later lines of therapy, uncertainties about relative doses of investigational drugs and optimal combinations, and mechanisms of action and resistance.
“Some data show that using antiangiogenic inhibitors can change the microenvironment and even render those agents resistant,” he said.
He noted that there are currently no validated, reliable biomarkers that can aid clinicians in patient selection for antiangiogenic agents.
“This study really confirms a clinical reality that’s known to all of us sitting in the audience,” commented invited discussant Dr. Primo N. Lara Jr. from the University of California Davis Comprehensive Cancer Center in Sacramento.
“The unanswered question is, who actually benefits from bevacizumab? No meta-analysis will answer this question. We would need integrated databases – Big Data – which would include annotated biospecimens to be able to somehow get to this question,” he said.
DENVER – Once again, angiogenesis inhibitors have been examined for benefit against advanced non–small cell lung cancer (NSCLC) and have been found wanting.
Results of a meta-analysis limited to only phase III randomized controlled trials showed that while angiogenesis inhibitors added to standard of care were associated with modest but significant improvements in progression-free survival (PFS) and response rates (RR), they did not improve overall survival (OS), reported Dr. Jacques Raphael from the Sunnybrook Odette Cancer Centre in Toronto.
“The role of antiangiogenic inhibitors in advanced lung cancer is still questionable,” he said at a world conference on lung cancer sponsored by the International Association for the Study of Lung Cancer.
The findings highlight the need for validated biomarkers that can help clinicians identify which patients are mostly likely to benefit from antiangiogenic therapy, he said.
The investigators conducted a meta-analysis to evaluate the effectiveness of adding any angiogenesis inhibitor or vascular disruptor to standard of care – chemotherapy, tyrosine kinase inhibitors (TKIs), and/or best supportive care – in patients with advanced NSCLC.
Two independent reviewers combed through electronic databases and the websites of international cancer organizations to identify all eligible phase III randomized, controlled trials.
The investigators then performed two separate meta-analyses, each combing data from all other drug trials with data on bevacizumab (Avastin) at either a 7.5-or 15-mg/kg dose.
They identified a total of 25 eligible trials containing data on 19,098 patients, including 9867 treated with an angiogenesis inhibitor and 9231 controls.
For the combined analysis of trials with the 7.5 mg/kg bevacizumab dose, they found no difference in overall survival (hazard ratio, 0.98; 95% confidence interval, 0.96-1.00). Similarly, there was no OS benefit from angiogenesis inhibitors in the analysis including the 15-mg/kg dose of bevacizumab (HR, 0.97; 95% CI, 0.94-1.00).
When they looked at PFS, however, there was a clear benefit in delaying disease progression with angiogenesis inhibitors in both the 7.5 mg/kg bevacizumab analysis (HR, 0.85; 95% CI, 0.79-0.91) and 15 mg/kg analysis (HR 0.85, 95% CI, 0.75-0.88).
Similarly, there was a significant benefit in terms of response rates with the addition of antiangiogenesis in both analyses (odds ratio, 1.61; 95% CI, 1.30-2.01 for the 7.5-mg/kg bevacizumab analysis, and 1.72, 95% CI, 1.39-2.14 for the 15-mg/kg dose).
The investigators also performed subgroup analyses of first-line therapy trials only, trials with monoclonal antibody-based agents, and trials with TKI-based antiangiogenics, and again found PFS and RR benefits, but no OS benefits, Dr. Raphael said.
The failure to see an OS benefit from angiogenesis inhibitors may be attributable to the heterogeneity of drugs in this class (that is, TKIs vs. monoclonal antibodies), clinical trial designs that may have confounded results by allowing crossover to a different drug in later lines of therapy, uncertainties about relative doses of investigational drugs and optimal combinations, and mechanisms of action and resistance.
“Some data show that using antiangiogenic inhibitors can change the microenvironment and even render those agents resistant,” he said.
He noted that there are currently no validated, reliable biomarkers that can aid clinicians in patient selection for antiangiogenic agents.
“This study really confirms a clinical reality that’s known to all of us sitting in the audience,” commented invited discussant Dr. Primo N. Lara Jr. from the University of California Davis Comprehensive Cancer Center in Sacramento.
“The unanswered question is, who actually benefits from bevacizumab? No meta-analysis will answer this question. We would need integrated databases – Big Data – which would include annotated biospecimens to be able to somehow get to this question,” he said.
DENVER – Once again, angiogenesis inhibitors have been examined for benefit against advanced non–small cell lung cancer (NSCLC) and have been found wanting.
Results of a meta-analysis limited to only phase III randomized controlled trials showed that while angiogenesis inhibitors added to standard of care were associated with modest but significant improvements in progression-free survival (PFS) and response rates (RR), they did not improve overall survival (OS), reported Dr. Jacques Raphael from the Sunnybrook Odette Cancer Centre in Toronto.
“The role of antiangiogenic inhibitors in advanced lung cancer is still questionable,” he said at a world conference on lung cancer sponsored by the International Association for the Study of Lung Cancer.
The findings highlight the need for validated biomarkers that can help clinicians identify which patients are mostly likely to benefit from antiangiogenic therapy, he said.
The investigators conducted a meta-analysis to evaluate the effectiveness of adding any angiogenesis inhibitor or vascular disruptor to standard of care – chemotherapy, tyrosine kinase inhibitors (TKIs), and/or best supportive care – in patients with advanced NSCLC.
Two independent reviewers combed through electronic databases and the websites of international cancer organizations to identify all eligible phase III randomized, controlled trials.
The investigators then performed two separate meta-analyses, each combing data from all other drug trials with data on bevacizumab (Avastin) at either a 7.5-or 15-mg/kg dose.
They identified a total of 25 eligible trials containing data on 19,098 patients, including 9867 treated with an angiogenesis inhibitor and 9231 controls.
For the combined analysis of trials with the 7.5 mg/kg bevacizumab dose, they found no difference in overall survival (hazard ratio, 0.98; 95% confidence interval, 0.96-1.00). Similarly, there was no OS benefit from angiogenesis inhibitors in the analysis including the 15-mg/kg dose of bevacizumab (HR, 0.97; 95% CI, 0.94-1.00).
When they looked at PFS, however, there was a clear benefit in delaying disease progression with angiogenesis inhibitors in both the 7.5 mg/kg bevacizumab analysis (HR, 0.85; 95% CI, 0.79-0.91) and 15 mg/kg analysis (HR 0.85, 95% CI, 0.75-0.88).
Similarly, there was a significant benefit in terms of response rates with the addition of antiangiogenesis in both analyses (odds ratio, 1.61; 95% CI, 1.30-2.01 for the 7.5-mg/kg bevacizumab analysis, and 1.72, 95% CI, 1.39-2.14 for the 15-mg/kg dose).
The investigators also performed subgroup analyses of first-line therapy trials only, trials with monoclonal antibody-based agents, and trials with TKI-based antiangiogenics, and again found PFS and RR benefits, but no OS benefits, Dr. Raphael said.
The failure to see an OS benefit from angiogenesis inhibitors may be attributable to the heterogeneity of drugs in this class (that is, TKIs vs. monoclonal antibodies), clinical trial designs that may have confounded results by allowing crossover to a different drug in later lines of therapy, uncertainties about relative doses of investigational drugs and optimal combinations, and mechanisms of action and resistance.
“Some data show that using antiangiogenic inhibitors can change the microenvironment and even render those agents resistant,” he said.
He noted that there are currently no validated, reliable biomarkers that can aid clinicians in patient selection for antiangiogenic agents.
“This study really confirms a clinical reality that’s known to all of us sitting in the audience,” commented invited discussant Dr. Primo N. Lara Jr. from the University of California Davis Comprehensive Cancer Center in Sacramento.
“The unanswered question is, who actually benefits from bevacizumab? No meta-analysis will answer this question. We would need integrated databases – Big Data – which would include annotated biospecimens to be able to somehow get to this question,” he said.
AT THE IASLC WORLD CONFERENCE
Key clinical point: Angiogenesis inhibitors added to standard of care do not improve overall survival of advanced lung cancer.
Major finding: The hazard ratios for OS in meta-analyses with data on bevacizumab at either of two doses were not statistically significant compared with standard of care alone.
Data source: Meta-analysis of 25 randomized controlled phase III trials involving 19,098 including 9,867 treated with an angiogenesis inhibitor and 9,231 controls.
Disclosures: Dr. Raphael reported no conflicts of interest. Dr. Lara serving as a paid consultant to 15 different pharmaceutical companies, including makers of angiogenesis inhibitors.
Recent quitters win big in lung screening trials
DENVER – It’s never too late to quit smoking, results of lung cancer screening trials confirm.
Among more than 3,300 heavy smokers over age 50 who took part in two low-dose CT (LDCT) screening programs, former smokers had a 37% reduction in all-cause mortality, compared with current smokers, and those who were active smokers at the time of randomization but quit during the follow-up period had a 43% lower risk for death, compared with those who continued to smoke, reported Dr. Ugo Pastorino of the Instiuto Nazionale dei Tumori in Milan.
He noted that the U.S. National Lung Screening Trial (NSLT) showed that screening with low-dose helical CT was associated with a nearly 7% reduction in all-cause mortality over 7 years of follow-up, compared with patients screened with chest x-ray.
“But we have to keep clear in our minds that the benefit achieved by this trial of early detection in terms of mortality reduction is only 1% per year, so it’s a not a major improvement. It’s a start, but we have to aim to improve this mortality reduction,” he said at the conference sponsored by the International Association for the Study of Lung Cancer.
Neither the NSLT nor other randomized screening trials currently underway have examined in detail the effects of smoking status on screening outcomes, prompting Dr. Pastorino and colleagues to investigate the matter in two cohorts of smokers assigned to LDCT in screening trials.
The study included 3,381 heavy smokers with a median follow-up of 9.7 years and a total follow-up of 32,858 person-years. Men comprised 69% of the combined cohorts, who had a median age of 58 and a median smoking of 40 pack-years.
The investigators divided the participants into current smokers – those who continued to smoke throughout the screening period, or if they quit did so within 1 year of the end of follow-up or death – and former smokers, subdivided into early quitters, who had stopped smoking by the time of accrual, and late quitters, who were active smokers at the time of accrual or randomization but stopped smoking at least 1 year before the end of the follow-up period or at least 1 year before death.
In an analysis of the effects of smoking on mortality, controlled for gender, age, body mass index, lung function, and pack-years smoked, they found that the relative risk for death from any cause among both early and late quitters, compared with current smokers, was 0.74. When they excluded 239 quitters who had kicked the habit less than 2 years before the end of follow-up or death, the benefits of not smoking were even greater, with an RR of 0.61.
Interestingly, when they looked at the early quitters, compared with current smokers, the RR for quitting was 0.63, and the effect appeared even stronger among more recent (late) quitters, who had a RR for all-cause mortality of 0.57, compared with current smokers. (All comparisons significant as shown by 95% confidence intervals).
Also of note was the fact that lung cancer accounted for fewer than 30% of deaths, Dr. Pastorino noted.
Dr. Nise H. Yamaguchi of the Hospital Israelita Albert Enstein in Sao Paolo, Brazil, applauded Dr. Pastorino and colleagues for the study, and succinctly summarized the take home message.
“If you came here from all around the world to see all these fancy treatments and everything that you can’t do, go back home and help people stop smoking, because you cure lots of people and save many lives for sure,” she said.
DENVER – It’s never too late to quit smoking, results of lung cancer screening trials confirm.
Among more than 3,300 heavy smokers over age 50 who took part in two low-dose CT (LDCT) screening programs, former smokers had a 37% reduction in all-cause mortality, compared with current smokers, and those who were active smokers at the time of randomization but quit during the follow-up period had a 43% lower risk for death, compared with those who continued to smoke, reported Dr. Ugo Pastorino of the Instiuto Nazionale dei Tumori in Milan.
He noted that the U.S. National Lung Screening Trial (NSLT) showed that screening with low-dose helical CT was associated with a nearly 7% reduction in all-cause mortality over 7 years of follow-up, compared with patients screened with chest x-ray.
“But we have to keep clear in our minds that the benefit achieved by this trial of early detection in terms of mortality reduction is only 1% per year, so it’s a not a major improvement. It’s a start, but we have to aim to improve this mortality reduction,” he said at the conference sponsored by the International Association for the Study of Lung Cancer.
Neither the NSLT nor other randomized screening trials currently underway have examined in detail the effects of smoking status on screening outcomes, prompting Dr. Pastorino and colleagues to investigate the matter in two cohorts of smokers assigned to LDCT in screening trials.
The study included 3,381 heavy smokers with a median follow-up of 9.7 years and a total follow-up of 32,858 person-years. Men comprised 69% of the combined cohorts, who had a median age of 58 and a median smoking of 40 pack-years.
The investigators divided the participants into current smokers – those who continued to smoke throughout the screening period, or if they quit did so within 1 year of the end of follow-up or death – and former smokers, subdivided into early quitters, who had stopped smoking by the time of accrual, and late quitters, who were active smokers at the time of accrual or randomization but stopped smoking at least 1 year before the end of the follow-up period or at least 1 year before death.
In an analysis of the effects of smoking on mortality, controlled for gender, age, body mass index, lung function, and pack-years smoked, they found that the relative risk for death from any cause among both early and late quitters, compared with current smokers, was 0.74. When they excluded 239 quitters who had kicked the habit less than 2 years before the end of follow-up or death, the benefits of not smoking were even greater, with an RR of 0.61.
Interestingly, when they looked at the early quitters, compared with current smokers, the RR for quitting was 0.63, and the effect appeared even stronger among more recent (late) quitters, who had a RR for all-cause mortality of 0.57, compared with current smokers. (All comparisons significant as shown by 95% confidence intervals).
Also of note was the fact that lung cancer accounted for fewer than 30% of deaths, Dr. Pastorino noted.
Dr. Nise H. Yamaguchi of the Hospital Israelita Albert Enstein in Sao Paolo, Brazil, applauded Dr. Pastorino and colleagues for the study, and succinctly summarized the take home message.
“If you came here from all around the world to see all these fancy treatments and everything that you can’t do, go back home and help people stop smoking, because you cure lots of people and save many lives for sure,” she said.
DENVER – It’s never too late to quit smoking, results of lung cancer screening trials confirm.
Among more than 3,300 heavy smokers over age 50 who took part in two low-dose CT (LDCT) screening programs, former smokers had a 37% reduction in all-cause mortality, compared with current smokers, and those who were active smokers at the time of randomization but quit during the follow-up period had a 43% lower risk for death, compared with those who continued to smoke, reported Dr. Ugo Pastorino of the Instiuto Nazionale dei Tumori in Milan.
He noted that the U.S. National Lung Screening Trial (NSLT) showed that screening with low-dose helical CT was associated with a nearly 7% reduction in all-cause mortality over 7 years of follow-up, compared with patients screened with chest x-ray.
“But we have to keep clear in our minds that the benefit achieved by this trial of early detection in terms of mortality reduction is only 1% per year, so it’s a not a major improvement. It’s a start, but we have to aim to improve this mortality reduction,” he said at the conference sponsored by the International Association for the Study of Lung Cancer.
Neither the NSLT nor other randomized screening trials currently underway have examined in detail the effects of smoking status on screening outcomes, prompting Dr. Pastorino and colleagues to investigate the matter in two cohorts of smokers assigned to LDCT in screening trials.
The study included 3,381 heavy smokers with a median follow-up of 9.7 years and a total follow-up of 32,858 person-years. Men comprised 69% of the combined cohorts, who had a median age of 58 and a median smoking of 40 pack-years.
The investigators divided the participants into current smokers – those who continued to smoke throughout the screening period, or if they quit did so within 1 year of the end of follow-up or death – and former smokers, subdivided into early quitters, who had stopped smoking by the time of accrual, and late quitters, who were active smokers at the time of accrual or randomization but stopped smoking at least 1 year before the end of the follow-up period or at least 1 year before death.
In an analysis of the effects of smoking on mortality, controlled for gender, age, body mass index, lung function, and pack-years smoked, they found that the relative risk for death from any cause among both early and late quitters, compared with current smokers, was 0.74. When they excluded 239 quitters who had kicked the habit less than 2 years before the end of follow-up or death, the benefits of not smoking were even greater, with an RR of 0.61.
Interestingly, when they looked at the early quitters, compared with current smokers, the RR for quitting was 0.63, and the effect appeared even stronger among more recent (late) quitters, who had a RR for all-cause mortality of 0.57, compared with current smokers. (All comparisons significant as shown by 95% confidence intervals).
Also of note was the fact that lung cancer accounted for fewer than 30% of deaths, Dr. Pastorino noted.
Dr. Nise H. Yamaguchi of the Hospital Israelita Albert Enstein in Sao Paolo, Brazil, applauded Dr. Pastorino and colleagues for the study, and succinctly summarized the take home message.
“If you came here from all around the world to see all these fancy treatments and everything that you can’t do, go back home and help people stop smoking, because you cure lots of people and save many lives for sure,” she said.
AT THE IASLC WORLD CONFERENCE
Key clinical point: Quitting smoking results in a significant reduction in all-cause mortality among heavy smokers taking part in screening programs.
Major finding: Compared with current smokers, the relative risk for all-cause mortality among ex-smokers or recent quitters was 0.74
Data source: Data on two cohorts totaling 3,381 current or ex-smokers assigned to low-dose CT lung screening.
Disclosures: The study was supported by the Italian Ministry of Health.
Vaccines targeting MUC1 show activity against NSCLC
DENVER – Checkpoint inhibitors get all the love these days, but other forms of immunotherapy, delivered alone or in combination with conventional chemotherapy, also show promising activity against non–small cell lung cancer, investigators report.
Two recombinant vaccines targeted against the tumor-associated antigen MUC1 (mucin 1, cell surface–associated) were associated with improved progression-free survival (PFS) and/or overall survival (OS).
A modified version of a vaccinia virus strain, labeled TG4010, when delivered in combination with chemotherapy to patients with stage IV NSCLC, improved both PFS and OS, particularly for those with low baseline levels of triple-positive activated lymphocytes (TrPAL), reported Dr. Elisabeth Quoix from Hospital Civil in Strasbourg, France.
“The future development of TG4010 is already planned in combination with chemotherapy and with immune checkpoint inhibitors,” Dr. Quoix said here at a world lung cancer conference sponsored by the International Association for the Study of Lung Cancer.
In a separate study, a dendritic cell-based immunotherapeutic vaccine was associated with good overall survival among patients who received six or more biweekly vaccinations, said Dr. Koji Teramoto from the Shiga University of Medical Science in Otsu, Japan.
Recombinant vaccine
TG4010 is a recombinant version of the Vaccinia virus Ankara strain that has been modified to carry coding sequences targeting MUC1 and interleukin-2. MUC1 receptors have been detected in various cancers, including NSCLC.
Dr. Quoix reported results of the phase IIb stage of a placebo-controlled study comparing a platinum-based chemotherapy doublet plus TG4010 or placebo in 222 patients with previously untreated stage IV NSCLC. Patients were stratified prior to randomization according to low or high TrPAL groups, with low TrPAL defined as a value at or below the upper limit of normal, based on a cutoff value obtained from 369 healthy volunteers.
Among patients with low TrPAL after 24 months of follow-up, the median PFS was 5.8 months for those on the TG4010-chemotherapy combination, compared with 5.0 months for those on chemotherapy with placebo vaccination. The hazard ratio (HR) favoring the addition of the vaccine in this group was 0.66 (P = .0104). Overall PFS rates at 24 months were 33% and 20%, respectively.
Overall survival was also significantly better among patients with low TrPAL who received the vaccine, with a median OS of 13.0 months, compared with 10.4 months for placebo-treated controls (HR, 0.67; P = .0183). In this group, 18-month OS rates were 39% and 30%, respectively.
Among patients with high TrPAL, however, the addition of TG4010 to chemotherapy did not improve either PFS or OS, Dr. Quoix reported.
Dendritic cell vaccine
Dr. Teramoto and his colleagues delivered the dendritic cell vaccine to 41 patients with advanced NSCLC refractory to standard therapy.
The technique involves collection of patients’ peripheral blood mononuclear cells, inducing differentiation of the cells into helper T cells with interleukin 4, expanding the population with granulocyte-macrophage colony-stimulating factor, pulsing them with MUC1 peptides, and then administering the treated cells via subcutaneous injections delivered once every 2 weeks.
Median survival time after the first vaccination was 7.4 months, and the 1-year survival rate was 29.3%.
Of the 41 patients, 29 (70%) received more than six vaccinations, and for these patients, the median survival time from the start of vaccination was 10.1 months, and the 1-year survival rate was 41.4%.
Clinical responses after six vaccinations included 12 cases of stable disease, and 17 with disease progression, for a disease control rate of 41.4% (as assessed according to Response Evaluation Criteria in Solid Tumors).
Dr. Teramoto noted that it generally takes several months to induce antitumor effects with vaccine immunotherapy. The vaccine appears to be most effective in patients with immune-related adverse events and in those with higher peripheral lymphocyte levels (20% and higher), he said, suggesting that peripheral lymphocyte levels could serve in patient selection.
Dr. Johan Vansteenkiste from the University Hospitals Leuven, Belgium, the invited discussant, commented that the TG4010 phase IIb results are promising and indicate that further planned development of the concept is warranted. On the other hand, the dendritic cell vaccine is interesting, but “it’s too early to comment,” he said.
DENVER – Checkpoint inhibitors get all the love these days, but other forms of immunotherapy, delivered alone or in combination with conventional chemotherapy, also show promising activity against non–small cell lung cancer, investigators report.
Two recombinant vaccines targeted against the tumor-associated antigen MUC1 (mucin 1, cell surface–associated) were associated with improved progression-free survival (PFS) and/or overall survival (OS).
A modified version of a vaccinia virus strain, labeled TG4010, when delivered in combination with chemotherapy to patients with stage IV NSCLC, improved both PFS and OS, particularly for those with low baseline levels of triple-positive activated lymphocytes (TrPAL), reported Dr. Elisabeth Quoix from Hospital Civil in Strasbourg, France.
“The future development of TG4010 is already planned in combination with chemotherapy and with immune checkpoint inhibitors,” Dr. Quoix said here at a world lung cancer conference sponsored by the International Association for the Study of Lung Cancer.
In a separate study, a dendritic cell-based immunotherapeutic vaccine was associated with good overall survival among patients who received six or more biweekly vaccinations, said Dr. Koji Teramoto from the Shiga University of Medical Science in Otsu, Japan.
Recombinant vaccine
TG4010 is a recombinant version of the Vaccinia virus Ankara strain that has been modified to carry coding sequences targeting MUC1 and interleukin-2. MUC1 receptors have been detected in various cancers, including NSCLC.
Dr. Quoix reported results of the phase IIb stage of a placebo-controlled study comparing a platinum-based chemotherapy doublet plus TG4010 or placebo in 222 patients with previously untreated stage IV NSCLC. Patients were stratified prior to randomization according to low or high TrPAL groups, with low TrPAL defined as a value at or below the upper limit of normal, based on a cutoff value obtained from 369 healthy volunteers.
Among patients with low TrPAL after 24 months of follow-up, the median PFS was 5.8 months for those on the TG4010-chemotherapy combination, compared with 5.0 months for those on chemotherapy with placebo vaccination. The hazard ratio (HR) favoring the addition of the vaccine in this group was 0.66 (P = .0104). Overall PFS rates at 24 months were 33% and 20%, respectively.
Overall survival was also significantly better among patients with low TrPAL who received the vaccine, with a median OS of 13.0 months, compared with 10.4 months for placebo-treated controls (HR, 0.67; P = .0183). In this group, 18-month OS rates were 39% and 30%, respectively.
Among patients with high TrPAL, however, the addition of TG4010 to chemotherapy did not improve either PFS or OS, Dr. Quoix reported.
Dendritic cell vaccine
Dr. Teramoto and his colleagues delivered the dendritic cell vaccine to 41 patients with advanced NSCLC refractory to standard therapy.
The technique involves collection of patients’ peripheral blood mononuclear cells, inducing differentiation of the cells into helper T cells with interleukin 4, expanding the population with granulocyte-macrophage colony-stimulating factor, pulsing them with MUC1 peptides, and then administering the treated cells via subcutaneous injections delivered once every 2 weeks.
Median survival time after the first vaccination was 7.4 months, and the 1-year survival rate was 29.3%.
Of the 41 patients, 29 (70%) received more than six vaccinations, and for these patients, the median survival time from the start of vaccination was 10.1 months, and the 1-year survival rate was 41.4%.
Clinical responses after six vaccinations included 12 cases of stable disease, and 17 with disease progression, for a disease control rate of 41.4% (as assessed according to Response Evaluation Criteria in Solid Tumors).
Dr. Teramoto noted that it generally takes several months to induce antitumor effects with vaccine immunotherapy. The vaccine appears to be most effective in patients with immune-related adverse events and in those with higher peripheral lymphocyte levels (20% and higher), he said, suggesting that peripheral lymphocyte levels could serve in patient selection.
Dr. Johan Vansteenkiste from the University Hospitals Leuven, Belgium, the invited discussant, commented that the TG4010 phase IIb results are promising and indicate that further planned development of the concept is warranted. On the other hand, the dendritic cell vaccine is interesting, but “it’s too early to comment,” he said.
DENVER – Checkpoint inhibitors get all the love these days, but other forms of immunotherapy, delivered alone or in combination with conventional chemotherapy, also show promising activity against non–small cell lung cancer, investigators report.
Two recombinant vaccines targeted against the tumor-associated antigen MUC1 (mucin 1, cell surface–associated) were associated with improved progression-free survival (PFS) and/or overall survival (OS).
A modified version of a vaccinia virus strain, labeled TG4010, when delivered in combination with chemotherapy to patients with stage IV NSCLC, improved both PFS and OS, particularly for those with low baseline levels of triple-positive activated lymphocytes (TrPAL), reported Dr. Elisabeth Quoix from Hospital Civil in Strasbourg, France.
“The future development of TG4010 is already planned in combination with chemotherapy and with immune checkpoint inhibitors,” Dr. Quoix said here at a world lung cancer conference sponsored by the International Association for the Study of Lung Cancer.
In a separate study, a dendritic cell-based immunotherapeutic vaccine was associated with good overall survival among patients who received six or more biweekly vaccinations, said Dr. Koji Teramoto from the Shiga University of Medical Science in Otsu, Japan.
Recombinant vaccine
TG4010 is a recombinant version of the Vaccinia virus Ankara strain that has been modified to carry coding sequences targeting MUC1 and interleukin-2. MUC1 receptors have been detected in various cancers, including NSCLC.
Dr. Quoix reported results of the phase IIb stage of a placebo-controlled study comparing a platinum-based chemotherapy doublet plus TG4010 or placebo in 222 patients with previously untreated stage IV NSCLC. Patients were stratified prior to randomization according to low or high TrPAL groups, with low TrPAL defined as a value at or below the upper limit of normal, based on a cutoff value obtained from 369 healthy volunteers.
Among patients with low TrPAL after 24 months of follow-up, the median PFS was 5.8 months for those on the TG4010-chemotherapy combination, compared with 5.0 months for those on chemotherapy with placebo vaccination. The hazard ratio (HR) favoring the addition of the vaccine in this group was 0.66 (P = .0104). Overall PFS rates at 24 months were 33% and 20%, respectively.
Overall survival was also significantly better among patients with low TrPAL who received the vaccine, with a median OS of 13.0 months, compared with 10.4 months for placebo-treated controls (HR, 0.67; P = .0183). In this group, 18-month OS rates were 39% and 30%, respectively.
Among patients with high TrPAL, however, the addition of TG4010 to chemotherapy did not improve either PFS or OS, Dr. Quoix reported.
Dendritic cell vaccine
Dr. Teramoto and his colleagues delivered the dendritic cell vaccine to 41 patients with advanced NSCLC refractory to standard therapy.
The technique involves collection of patients’ peripheral blood mononuclear cells, inducing differentiation of the cells into helper T cells with interleukin 4, expanding the population with granulocyte-macrophage colony-stimulating factor, pulsing them with MUC1 peptides, and then administering the treated cells via subcutaneous injections delivered once every 2 weeks.
Median survival time after the first vaccination was 7.4 months, and the 1-year survival rate was 29.3%.
Of the 41 patients, 29 (70%) received more than six vaccinations, and for these patients, the median survival time from the start of vaccination was 10.1 months, and the 1-year survival rate was 41.4%.
Clinical responses after six vaccinations included 12 cases of stable disease, and 17 with disease progression, for a disease control rate of 41.4% (as assessed according to Response Evaluation Criteria in Solid Tumors).
Dr. Teramoto noted that it generally takes several months to induce antitumor effects with vaccine immunotherapy. The vaccine appears to be most effective in patients with immune-related adverse events and in those with higher peripheral lymphocyte levels (20% and higher), he said, suggesting that peripheral lymphocyte levels could serve in patient selection.
Dr. Johan Vansteenkiste from the University Hospitals Leuven, Belgium, the invited discussant, commented that the TG4010 phase IIb results are promising and indicate that further planned development of the concept is warranted. On the other hand, the dendritic cell vaccine is interesting, but “it’s too early to comment,” he said.
AT THE IASLC WORLD CONFERENCE
Key clinical point: Therapeutic vaccines targeting MUC1 show clinical activity against non–small cell lung cancer.
Major finding: Overall survival among patients with low levels of triple-positive activated lymphocytes (TrPAL) treated with chemotherapy and the TG4010 vaccine was 13.0 months, compared with 10.4 months for chemo/placebo-treated controls.
Data source: Retrospective analysis of a randomized clinical trial in 222 patients with advanced NSCLC; open-label prospective study in 41 patients with NSCLC.
Disclosures: The TG4010 study was supported by Transgene. Dr. Quoix disclosed financial ties with various companies other than Transgene, Dr. Teramato did not disclose the funding source for the dendritic vaccine study, but reported having no conflicts of interest. Dr. Vansteenkiste disclosed serving as a consultant and speaker for several companies, but not Transgene.
Radiation + lumpectomy tied to increased survival in older women with TNBC
Older women with triple-negative breast cancer appear to get an overall survival and disease-specific survival benefit with the addition of radiation to breast-conserving surgery, authors of a retrospective study said.
Among 974 women aged 70 and above with T1-2, N0, M0 triple-negative breast cancer (TNBC; lacking the Her2-neu, estrogen, and progesterone receptors), the addition of radiation to lumpectomy was associated at 23 months’ follow-up with an overall survival (OS) rate of 98.2%, compared with 85.6% for women who received lumpectomy alone (P less than .001). Respective rates of disease-specific survival (DSS) were 99% and 94% (P = .003).
“The use of adjuvant radiation therapy after lumpectomy for elderly women with early-stage TNBC was associated with improved OS and DSS. Noting the potential for selection bias in this study, future prospective study is required to define the management of early-stage triple-negative breast cancer,” wrote Dr. Sean Szjea and colleagues at the University of Texas, Galveston, in a meeting abstract. The study will be presented at a breast cancer symposium sponsored by the American Society of Clinical Oncology.
It’s known that older women with estrogen-receptor positive disease can have good clinical outcomes with lumpectomy and adjuvant therapy alone, but whether adding radiation to breast-conserving surgery in older women with TNBC offers clinical benefit is less certain, the investigators said, prompting them to dive for data into the Surveillance, Epidemiology, and End Results database.
They collected information on 974 women aged 70 or older who underwent lumpectomy for early-stage TNBC with no nodal invasion or metastatic disease from 2010 through 2011. Of this group, 662 (68%) also received radiation therapy.
In addition to determining the OS and DSS rates in the overall population, the investigators conducted multivariate regression modeling controlling for confounding variables, including the use of neoadjuvant chemotherapy, the number of lymph nodes sampled, age, laterality, grade, T stage, the extent of surgery, and the existence of other cancers. They found that the survival benefit for radiation held for both OS (hazard ratio [HR], 0.14; P less than .001) and DSS (HR, 0.14; P = .01).
The authors reported having no relevant financial disclosures.
Older women with triple-negative breast cancer appear to get an overall survival and disease-specific survival benefit with the addition of radiation to breast-conserving surgery, authors of a retrospective study said.
Among 974 women aged 70 and above with T1-2, N0, M0 triple-negative breast cancer (TNBC; lacking the Her2-neu, estrogen, and progesterone receptors), the addition of radiation to lumpectomy was associated at 23 months’ follow-up with an overall survival (OS) rate of 98.2%, compared with 85.6% for women who received lumpectomy alone (P less than .001). Respective rates of disease-specific survival (DSS) were 99% and 94% (P = .003).
“The use of adjuvant radiation therapy after lumpectomy for elderly women with early-stage TNBC was associated with improved OS and DSS. Noting the potential for selection bias in this study, future prospective study is required to define the management of early-stage triple-negative breast cancer,” wrote Dr. Sean Szjea and colleagues at the University of Texas, Galveston, in a meeting abstract. The study will be presented at a breast cancer symposium sponsored by the American Society of Clinical Oncology.
It’s known that older women with estrogen-receptor positive disease can have good clinical outcomes with lumpectomy and adjuvant therapy alone, but whether adding radiation to breast-conserving surgery in older women with TNBC offers clinical benefit is less certain, the investigators said, prompting them to dive for data into the Surveillance, Epidemiology, and End Results database.
They collected information on 974 women aged 70 or older who underwent lumpectomy for early-stage TNBC with no nodal invasion or metastatic disease from 2010 through 2011. Of this group, 662 (68%) also received radiation therapy.
In addition to determining the OS and DSS rates in the overall population, the investigators conducted multivariate regression modeling controlling for confounding variables, including the use of neoadjuvant chemotherapy, the number of lymph nodes sampled, age, laterality, grade, T stage, the extent of surgery, and the existence of other cancers. They found that the survival benefit for radiation held for both OS (hazard ratio [HR], 0.14; P less than .001) and DSS (HR, 0.14; P = .01).
The authors reported having no relevant financial disclosures.
Older women with triple-negative breast cancer appear to get an overall survival and disease-specific survival benefit with the addition of radiation to breast-conserving surgery, authors of a retrospective study said.
Among 974 women aged 70 and above with T1-2, N0, M0 triple-negative breast cancer (TNBC; lacking the Her2-neu, estrogen, and progesterone receptors), the addition of radiation to lumpectomy was associated at 23 months’ follow-up with an overall survival (OS) rate of 98.2%, compared with 85.6% for women who received lumpectomy alone (P less than .001). Respective rates of disease-specific survival (DSS) were 99% and 94% (P = .003).
“The use of adjuvant radiation therapy after lumpectomy for elderly women with early-stage TNBC was associated with improved OS and DSS. Noting the potential for selection bias in this study, future prospective study is required to define the management of early-stage triple-negative breast cancer,” wrote Dr. Sean Szjea and colleagues at the University of Texas, Galveston, in a meeting abstract. The study will be presented at a breast cancer symposium sponsored by the American Society of Clinical Oncology.
It’s known that older women with estrogen-receptor positive disease can have good clinical outcomes with lumpectomy and adjuvant therapy alone, but whether adding radiation to breast-conserving surgery in older women with TNBC offers clinical benefit is less certain, the investigators said, prompting them to dive for data into the Surveillance, Epidemiology, and End Results database.
They collected information on 974 women aged 70 or older who underwent lumpectomy for early-stage TNBC with no nodal invasion or metastatic disease from 2010 through 2011. Of this group, 662 (68%) also received radiation therapy.
In addition to determining the OS and DSS rates in the overall population, the investigators conducted multivariate regression modeling controlling for confounding variables, including the use of neoadjuvant chemotherapy, the number of lymph nodes sampled, age, laterality, grade, T stage, the extent of surgery, and the existence of other cancers. They found that the survival benefit for radiation held for both OS (hazard ratio [HR], 0.14; P less than .001) and DSS (HR, 0.14; P = .01).
The authors reported having no relevant financial disclosures.
FROM THE 2015 ASCO BREAST CANCER SYMPOSIUM
Key clinical point: Radiation added to breast-conserving surgery in women 70 and older with early-stage triple-negative breast cancer was associated with improved survival.
Major finding: Overall survival with radiation and lumpectomy was 98.2%, compared with 85.6% for women who received lumpectomy alone (P less than .001)
Data source: A prospective database review of 974 women aged 70 and older treated from 2010 through 2011.
Disclosures: The authors reported having no relevant financial disclosures.
DCIS recurrences have declined significantly
Recurrence rates of ductal carcinoma in situ (DCIS ) have declined significantly over 3 decades, with the improvements likely because of better screening and pathologic assessment, investigators say.
A retrospective review of 2,996 women who underwent breast-conserving surgery (BCS) for DCIS at Memorial Sloan Kettering Cancer Center in New York from 1978 through 2010 showed that there were 363 recurrences within 5 years of treatment (12%). The 5-year recurrence rate for women treated from 1978 through 1998 was 13.6%, compared with 6.6% for women treated from 1999 through 2010.
The hazard ratio (HR) for more recent treatment was 0.62 (P less than .0001), report Dr. Kimberly J. Van Zee and colleagues at MSKCC in an oral abstract to be presented at the symposium sponsored by the American Society of Clinical Oncology.
The period of treatment remained a significant predictor of lower recurrence after multivariate analysis adjusting for age, family history, radiologic vs. clinical presentation, nuclear grade (nonhigh vs. high), necrosis, number of excisions (two or few vs. three or more), margin status (positive or close margins vs. negative), radiation, and use of endocrine therapy (HR 0.74, P = 0.02).
When the investigators stratified patients by whether they received radiation in addition to BCS, they found that recurrence rates dropped only for those patients who did not receive radiation (HR 0.62, P = 0.003). Among patients who received radiation, recurrence rates did not decline over time (HR 1.13, P = 0.6).
The authors conclude that the lower risk of recurrence over time is only partially explained by changes in clinical practice, such as screening, emphasis on negative surgical margins, and use of adjuvant therapies. The fact that the decline was seen only for women who did not receive radiation suggests that the improvements were not attributable to improvement in radiation therapy techniques. Rather, they may be due to improvements in mammography and in pathologic assessment, the investigators maintain.
“The lower recurrence risk observed for DCIS patients treated in more recent years is important for patient education, especially in view of the widely reported recent increase in use of mastectomy,” they write in the study abstract.
Recurrence rates of ductal carcinoma in situ (DCIS ) have declined significantly over 3 decades, with the improvements likely because of better screening and pathologic assessment, investigators say.
A retrospective review of 2,996 women who underwent breast-conserving surgery (BCS) for DCIS at Memorial Sloan Kettering Cancer Center in New York from 1978 through 2010 showed that there were 363 recurrences within 5 years of treatment (12%). The 5-year recurrence rate for women treated from 1978 through 1998 was 13.6%, compared with 6.6% for women treated from 1999 through 2010.
The hazard ratio (HR) for more recent treatment was 0.62 (P less than .0001), report Dr. Kimberly J. Van Zee and colleagues at MSKCC in an oral abstract to be presented at the symposium sponsored by the American Society of Clinical Oncology.
The period of treatment remained a significant predictor of lower recurrence after multivariate analysis adjusting for age, family history, radiologic vs. clinical presentation, nuclear grade (nonhigh vs. high), necrosis, number of excisions (two or few vs. three or more), margin status (positive or close margins vs. negative), radiation, and use of endocrine therapy (HR 0.74, P = 0.02).
When the investigators stratified patients by whether they received radiation in addition to BCS, they found that recurrence rates dropped only for those patients who did not receive radiation (HR 0.62, P = 0.003). Among patients who received radiation, recurrence rates did not decline over time (HR 1.13, P = 0.6).
The authors conclude that the lower risk of recurrence over time is only partially explained by changes in clinical practice, such as screening, emphasis on negative surgical margins, and use of adjuvant therapies. The fact that the decline was seen only for women who did not receive radiation suggests that the improvements were not attributable to improvement in radiation therapy techniques. Rather, they may be due to improvements in mammography and in pathologic assessment, the investigators maintain.
“The lower recurrence risk observed for DCIS patients treated in more recent years is important for patient education, especially in view of the widely reported recent increase in use of mastectomy,” they write in the study abstract.
Recurrence rates of ductal carcinoma in situ (DCIS ) have declined significantly over 3 decades, with the improvements likely because of better screening and pathologic assessment, investigators say.
A retrospective review of 2,996 women who underwent breast-conserving surgery (BCS) for DCIS at Memorial Sloan Kettering Cancer Center in New York from 1978 through 2010 showed that there were 363 recurrences within 5 years of treatment (12%). The 5-year recurrence rate for women treated from 1978 through 1998 was 13.6%, compared with 6.6% for women treated from 1999 through 2010.
The hazard ratio (HR) for more recent treatment was 0.62 (P less than .0001), report Dr. Kimberly J. Van Zee and colleagues at MSKCC in an oral abstract to be presented at the symposium sponsored by the American Society of Clinical Oncology.
The period of treatment remained a significant predictor of lower recurrence after multivariate analysis adjusting for age, family history, radiologic vs. clinical presentation, nuclear grade (nonhigh vs. high), necrosis, number of excisions (two or few vs. three or more), margin status (positive or close margins vs. negative), radiation, and use of endocrine therapy (HR 0.74, P = 0.02).
When the investigators stratified patients by whether they received radiation in addition to BCS, they found that recurrence rates dropped only for those patients who did not receive radiation (HR 0.62, P = 0.003). Among patients who received radiation, recurrence rates did not decline over time (HR 1.13, P = 0.6).
The authors conclude that the lower risk of recurrence over time is only partially explained by changes in clinical practice, such as screening, emphasis on negative surgical margins, and use of adjuvant therapies. The fact that the decline was seen only for women who did not receive radiation suggests that the improvements were not attributable to improvement in radiation therapy techniques. Rather, they may be due to improvements in mammography and in pathologic assessment, the investigators maintain.
“The lower recurrence risk observed for DCIS patients treated in more recent years is important for patient education, especially in view of the widely reported recent increase in use of mastectomy,” they write in the study abstract.
Key clinical point: Recurrence rates of DCIS in patients treated with breast-conserving surgery declined significantly from 1978 through 2010.
Major finding: The 5-year recurrence rate for women treated from 1978 through 1998 was 13.6%, compared with 6.6% for women treated from 1999 through 2010.
Data source: Retrospective case review of 2,996 women with DCIS treated with breast-conserving surgery with or without radiation.
Disclosures: The funding source was not disclosed. Coauthor Dr. Monica Morrow disclosed receiving honoraria from Genomic Health.
Clinical advances drive lung cancer staging, classification changes
DENVER – The term “precision medicine” can be applied to both clinical care and to pathology, as newly updated staging and classification systems for lung cancer show.
The proposed revised (8th) edition of the TNM staging system for lung cancer gives more weight to tumor size as a prognostic factor, reclassifies some primary tumor (T) descriptors, validates current nodal status (N) descriptors, modifies the definition of some types of metastases (M), and includes additional stages for better prognostic stratification, reported Dr. Ramón Rami-Porta from the Universitari Mútua Terrassa in Barcelona, at a world conference on lung cancer sponsored by the International Association for the Study of Lung Cancer.
Similarly, the updated World Health Organization (WHO) Classification of Lung Tumors, described by Dr. William D. Travis from the Memorial Sloan Kettering Cancer Center in New York, incorporates knowledge gained from immunohistochemistry and molecular testing for common genetic mutations into recommendations for treating the specific clinical circumstances of patients with lung cancer.
WHO’s Next
“The 2015 WHO Classification captures a remarkable decade of advances in every lung cancer specialty, from pathology – including histology, cytology, immunohistochemistry, genetics – to oncology, surgery, radiology, and epidemiology. The rapid expansion of immunohistochemical and molecular tools has had a profound impact on how we were able to reclassify a number of tumors, in addition to how we were able to contribute to improvement of subtyping of lung cancers, particularly non–small cell lung cancer,” Dr. Travis said at a media briefing following his discussion of the new classification at a plenary session.
The changes are expected to improve clinical management of patients with advanced lung cancer by clarifying criteria and terminology for small biopsies and cytology, establishing more accurate histologic subtyping, suggesting strategic management of small tissues, and streamlining the work flow for molecular testing. The classification also emphasizes the need for multidisciplinary cooperation among myriad clinicians, he said.
For surgically resected patients, the classification officially recognizes for the first time subsets of non–small cell lung cancer of adenocarcinoma histology with survival rates of 100% (adenocarcinoma in situ), or nearly 100% (minimally invasive adenocarcinoma).
Among the major changes that will affect the diagnosis of surgically resected patients are the adoption of the 2011 IASLC/ATS/ERS Lung Adenocarcinoma Classification, restriction of a diagnosis of large cell carcinoma to tumors lacking clear differentiation by both immunohistochemistry and morphology, reclassifying of squamous cancers into keratinizing, nonkeratinizing, and basaloid subtypes with elimination of clear cell, small cell, and papillary subtypes. Neuroendocrine subtypes are grouped together, but their classification otherwise remains largely unchanged.
The revised classification is expected to improve prediction of survival and recurrence, predict whether a patient is likely to have a survival benefit with platinum-based chemotherapy, allow radiologic pathologic correlations, and affects TNM staging by emphasizing solid tumor size (vs. whole tumor size), Dr. Travis said.
TNM Changes
The proposed changes to the TNM tumor staging have been submitted for approval to the American Joint Committee on Cancer and the Union for International Cancer Control.
If adopted, they would represent the first significant changes since the 7th edition’s publication in 2009. The changes are based on data on more than 77,000 patients diagnosed with lung cancer from 1999 through 2010.
The proposed changes are not intended, however, to alter clinical practice, and instead “imply a taxonomic refinement rather than new indications of already established treatment protocols,” Dr. Rami-Porta said.
In some cases, the proposed changes would result in an upgrading of the T stage, while others would result in downgrading. For example, tumors that range in size between 1 and 2 cm, designated T1a in the 7th edition, would be T1b in the 8th edition. Similarly, tumors larger than 2 cm and up to 3 cm would be upgraded from T1b to T1c, those larger than 4 up to 5 would go from T2a to T2b, those larger 5 and up to 7 cm would rise from T2b to T3, and those larger than 7 cm would be reclassified from T3 to T4. Tumors invading the diaphragm would also be upgraded from T3 to T4 under the proposed revisions.
In contrast, tumors with limited invasion of the trachea (bronchus less than 2 cm from the carina) would be downgraded from T3 to T2, as would tumors associated with total atelectasis and/or pneumonitis.
The current N descriptors are adequate for predicting prognosis, the investigators determined, prompting the recommendation to retain them in the new edition.
The investigators propose slight changes to the M descriptors of metastases. Although they found no significant differences in survival found among patients with M1a (metastases within the chest cavity) descriptors, when distant metastases outside the chest cavity (M1b) were assessed by to the number of metastases, they found that patients with tumors with one metastasis in one organ had significantly better outcomes than those who had multiple metastases in one or more organs.
The proposed revision would continue to group in the M1a category cases with pleural/pericardial effusions, contralateral/bilateral lung nodules, contralateral/bilateral pleural nodules, or a combination of multiple parameters. However, single metastatic lesions in a single distant organ would be reclassified as M1b, and multiple lesions in a single organ or multiple lesions in multiple organs would be reclassified as M1c.
DENVER – The term “precision medicine” can be applied to both clinical care and to pathology, as newly updated staging and classification systems for lung cancer show.
The proposed revised (8th) edition of the TNM staging system for lung cancer gives more weight to tumor size as a prognostic factor, reclassifies some primary tumor (T) descriptors, validates current nodal status (N) descriptors, modifies the definition of some types of metastases (M), and includes additional stages for better prognostic stratification, reported Dr. Ramón Rami-Porta from the Universitari Mútua Terrassa in Barcelona, at a world conference on lung cancer sponsored by the International Association for the Study of Lung Cancer.
Similarly, the updated World Health Organization (WHO) Classification of Lung Tumors, described by Dr. William D. Travis from the Memorial Sloan Kettering Cancer Center in New York, incorporates knowledge gained from immunohistochemistry and molecular testing for common genetic mutations into recommendations for treating the specific clinical circumstances of patients with lung cancer.
WHO’s Next
“The 2015 WHO Classification captures a remarkable decade of advances in every lung cancer specialty, from pathology – including histology, cytology, immunohistochemistry, genetics – to oncology, surgery, radiology, and epidemiology. The rapid expansion of immunohistochemical and molecular tools has had a profound impact on how we were able to reclassify a number of tumors, in addition to how we were able to contribute to improvement of subtyping of lung cancers, particularly non–small cell lung cancer,” Dr. Travis said at a media briefing following his discussion of the new classification at a plenary session.
The changes are expected to improve clinical management of patients with advanced lung cancer by clarifying criteria and terminology for small biopsies and cytology, establishing more accurate histologic subtyping, suggesting strategic management of small tissues, and streamlining the work flow for molecular testing. The classification also emphasizes the need for multidisciplinary cooperation among myriad clinicians, he said.
For surgically resected patients, the classification officially recognizes for the first time subsets of non–small cell lung cancer of adenocarcinoma histology with survival rates of 100% (adenocarcinoma in situ), or nearly 100% (minimally invasive adenocarcinoma).
Among the major changes that will affect the diagnosis of surgically resected patients are the adoption of the 2011 IASLC/ATS/ERS Lung Adenocarcinoma Classification, restriction of a diagnosis of large cell carcinoma to tumors lacking clear differentiation by both immunohistochemistry and morphology, reclassifying of squamous cancers into keratinizing, nonkeratinizing, and basaloid subtypes with elimination of clear cell, small cell, and papillary subtypes. Neuroendocrine subtypes are grouped together, but their classification otherwise remains largely unchanged.
The revised classification is expected to improve prediction of survival and recurrence, predict whether a patient is likely to have a survival benefit with platinum-based chemotherapy, allow radiologic pathologic correlations, and affects TNM staging by emphasizing solid tumor size (vs. whole tumor size), Dr. Travis said.
TNM Changes
The proposed changes to the TNM tumor staging have been submitted for approval to the American Joint Committee on Cancer and the Union for International Cancer Control.
If adopted, they would represent the first significant changes since the 7th edition’s publication in 2009. The changes are based on data on more than 77,000 patients diagnosed with lung cancer from 1999 through 2010.
The proposed changes are not intended, however, to alter clinical practice, and instead “imply a taxonomic refinement rather than new indications of already established treatment protocols,” Dr. Rami-Porta said.
In some cases, the proposed changes would result in an upgrading of the T stage, while others would result in downgrading. For example, tumors that range in size between 1 and 2 cm, designated T1a in the 7th edition, would be T1b in the 8th edition. Similarly, tumors larger than 2 cm and up to 3 cm would be upgraded from T1b to T1c, those larger than 4 up to 5 would go from T2a to T2b, those larger 5 and up to 7 cm would rise from T2b to T3, and those larger than 7 cm would be reclassified from T3 to T4. Tumors invading the diaphragm would also be upgraded from T3 to T4 under the proposed revisions.
In contrast, tumors with limited invasion of the trachea (bronchus less than 2 cm from the carina) would be downgraded from T3 to T2, as would tumors associated with total atelectasis and/or pneumonitis.
The current N descriptors are adequate for predicting prognosis, the investigators determined, prompting the recommendation to retain them in the new edition.
The investigators propose slight changes to the M descriptors of metastases. Although they found no significant differences in survival found among patients with M1a (metastases within the chest cavity) descriptors, when distant metastases outside the chest cavity (M1b) were assessed by to the number of metastases, they found that patients with tumors with one metastasis in one organ had significantly better outcomes than those who had multiple metastases in one or more organs.
The proposed revision would continue to group in the M1a category cases with pleural/pericardial effusions, contralateral/bilateral lung nodules, contralateral/bilateral pleural nodules, or a combination of multiple parameters. However, single metastatic lesions in a single distant organ would be reclassified as M1b, and multiple lesions in a single organ or multiple lesions in multiple organs would be reclassified as M1c.
DENVER – The term “precision medicine” can be applied to both clinical care and to pathology, as newly updated staging and classification systems for lung cancer show.
The proposed revised (8th) edition of the TNM staging system for lung cancer gives more weight to tumor size as a prognostic factor, reclassifies some primary tumor (T) descriptors, validates current nodal status (N) descriptors, modifies the definition of some types of metastases (M), and includes additional stages for better prognostic stratification, reported Dr. Ramón Rami-Porta from the Universitari Mútua Terrassa in Barcelona, at a world conference on lung cancer sponsored by the International Association for the Study of Lung Cancer.
Similarly, the updated World Health Organization (WHO) Classification of Lung Tumors, described by Dr. William D. Travis from the Memorial Sloan Kettering Cancer Center in New York, incorporates knowledge gained from immunohistochemistry and molecular testing for common genetic mutations into recommendations for treating the specific clinical circumstances of patients with lung cancer.
WHO’s Next
“The 2015 WHO Classification captures a remarkable decade of advances in every lung cancer specialty, from pathology – including histology, cytology, immunohistochemistry, genetics – to oncology, surgery, radiology, and epidemiology. The rapid expansion of immunohistochemical and molecular tools has had a profound impact on how we were able to reclassify a number of tumors, in addition to how we were able to contribute to improvement of subtyping of lung cancers, particularly non–small cell lung cancer,” Dr. Travis said at a media briefing following his discussion of the new classification at a plenary session.
The changes are expected to improve clinical management of patients with advanced lung cancer by clarifying criteria and terminology for small biopsies and cytology, establishing more accurate histologic subtyping, suggesting strategic management of small tissues, and streamlining the work flow for molecular testing. The classification also emphasizes the need for multidisciplinary cooperation among myriad clinicians, he said.
For surgically resected patients, the classification officially recognizes for the first time subsets of non–small cell lung cancer of adenocarcinoma histology with survival rates of 100% (adenocarcinoma in situ), or nearly 100% (minimally invasive adenocarcinoma).
Among the major changes that will affect the diagnosis of surgically resected patients are the adoption of the 2011 IASLC/ATS/ERS Lung Adenocarcinoma Classification, restriction of a diagnosis of large cell carcinoma to tumors lacking clear differentiation by both immunohistochemistry and morphology, reclassifying of squamous cancers into keratinizing, nonkeratinizing, and basaloid subtypes with elimination of clear cell, small cell, and papillary subtypes. Neuroendocrine subtypes are grouped together, but their classification otherwise remains largely unchanged.
The revised classification is expected to improve prediction of survival and recurrence, predict whether a patient is likely to have a survival benefit with platinum-based chemotherapy, allow radiologic pathologic correlations, and affects TNM staging by emphasizing solid tumor size (vs. whole tumor size), Dr. Travis said.
TNM Changes
The proposed changes to the TNM tumor staging have been submitted for approval to the American Joint Committee on Cancer and the Union for International Cancer Control.
If adopted, they would represent the first significant changes since the 7th edition’s publication in 2009. The changes are based on data on more than 77,000 patients diagnosed with lung cancer from 1999 through 2010.
The proposed changes are not intended, however, to alter clinical practice, and instead “imply a taxonomic refinement rather than new indications of already established treatment protocols,” Dr. Rami-Porta said.
In some cases, the proposed changes would result in an upgrading of the T stage, while others would result in downgrading. For example, tumors that range in size between 1 and 2 cm, designated T1a in the 7th edition, would be T1b in the 8th edition. Similarly, tumors larger than 2 cm and up to 3 cm would be upgraded from T1b to T1c, those larger than 4 up to 5 would go from T2a to T2b, those larger 5 and up to 7 cm would rise from T2b to T3, and those larger than 7 cm would be reclassified from T3 to T4. Tumors invading the diaphragm would also be upgraded from T3 to T4 under the proposed revisions.
In contrast, tumors with limited invasion of the trachea (bronchus less than 2 cm from the carina) would be downgraded from T3 to T2, as would tumors associated with total atelectasis and/or pneumonitis.
The current N descriptors are adequate for predicting prognosis, the investigators determined, prompting the recommendation to retain them in the new edition.
The investigators propose slight changes to the M descriptors of metastases. Although they found no significant differences in survival found among patients with M1a (metastases within the chest cavity) descriptors, when distant metastases outside the chest cavity (M1b) were assessed by to the number of metastases, they found that patients with tumors with one metastasis in one organ had significantly better outcomes than those who had multiple metastases in one or more organs.
The proposed revision would continue to group in the M1a category cases with pleural/pericardial effusions, contralateral/bilateral lung nodules, contralateral/bilateral pleural nodules, or a combination of multiple parameters. However, single metastatic lesions in a single distant organ would be reclassified as M1b, and multiple lesions in a single organ or multiple lesions in multiple organs would be reclassified as M1c.
AT THE IASLC WORLD CONFERENCE
Key clinical point: Improved understanding of lung cancer over the last decade has prompted updates to international tumor staging and classification systems.
Major finding: The WHO 2015 Classification of Lung Tumors is expected to aid clinical practice.
Data source: Conference presentation of key changes to the WHO Classification and TNM staging system.
Disclosures: Dr. Travis and Dr. Rami-Porta reported having no disclosures relevant to their presentations.
Nivolumab benefits heavily pretreated squamous NSCLC patients
DENVER – Patients with heavily pretreated advanced squamous cell non–small cell lung cancer (NSCLC) continued to receive clinical benefit from treatment with the checkpoint inhibitor nivolumab, longer-term results from a phase II clinical trial show.
Among 117 patients with squamous cell NSCLC that had progressed either during or after platinum-based doublet chemotherapy and at least one additional systemic regimen, the 12-month and 18-month overall survival (OS) rates were 39% and 27% respectively.
Historically, 1-year OS rates for patients with advanced squamous NSCLC treated with systemic chemotherapy ranged from 5% to 22%, and 2-year OS was less than 5%, said Dr. Leora Horn of the Vanderbilt-Ingram Cancer Center in Nashville, Tennessee.
“Consistent with randomized phase III trials, most treatment-related adverse events were of low grade and manageable with established safety guidelines, and most patients who experienced a treatment-related selected adverse event reported their first event within 3-6 months of treatment initiation. Clinical benefit with nivolumab was observed independent of [programmed death ligand 1] expression,” Dr. Horn said at a world conference on lung cancer.
Nivolumab (Opdivo) is an immune checkpoint inhibitor that has shown a survival benefit compared with docetaxel in patients with NSCLC of both adenocarcinoma and squamous cell histologies. It is approved in the U.S. for the treatment of metastatic squamous cell NSCLC which has progressed on therapy with a platinum-based doublet chemotherapy.
Dr. Horn presented updated efficacy and safety results from the previously reported phase II Checkmate 063 trial.
The investigators screened 140 and treated 117 patients with stage IIIB to stage IV squamous NSCLC who had disease progression on two or more prior lines of systemic chemotherapy and an Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1.
The patients received nivolumab 3 mg/kg by intravenous infusion every 2 weeks until disease progression or unacceptable toxicity.
As of July 2014, the date of the first analysis, after a median follow-up of 8 months the median overall survival was 8.2 months, and the 1-year overall survival rate was 41%.
Updated survival data as of June 2015, showed that the median OS was 8.1 months, and the overall survival at 1 year was 39%. As noted before, the 18-month overall survival was 27%.
An analysis stratified by PD-L1 expression showed a median OS of 8.3 months for patients with less than 1% of tumor cells expressing PD-L1, compared with 10.1 months for patients with 1% or more of tumor cells expressing the ligand. Among 10 patients not evaluable for PD-L1 expression, the median OS was 13 months.
Adverse event (AE) rates remained stable from 1 year to the next, with 74% of patients in July 2014 having had any grade of AE, compared with 75% of patients in June 2015. The percentage of patients who experienced a grade 3 or 4 adverse event was 17% for each analysis, indicating that the grade 3/4 events occurred early in the course of treatment. The two treatment-related deaths, one from hypoxic pneumonia and the other from ischemic stroke, were reported in the previous analysis.
The most common treatment-related select adverse events of any grade were gastrointestinal (10% in the current and previous analyses), pulmonary (6% vs. 5% in the previous analysis), and skin related (16% vs. 15%, respectively).
The survival data are consistent with those seen in the long-term follow-up data from the Checkmate 017 trial, in which nivolumab was compared with docetaxel in patients with advanced squamous NSCLC, commented Dr. Solange Peters of the Lausanne (Switzerland) University Hospital, the invited discussant.
The slightly better survival in the 017 trial is likely due to the fact the majority of patients in the 063 trial were heavily pretreated, with 65% having received three or more prior lines of therapy, Dr. Peters said at the conference sponsored by the International Association for the Study of Lung Cancer.
DENVER – Patients with heavily pretreated advanced squamous cell non–small cell lung cancer (NSCLC) continued to receive clinical benefit from treatment with the checkpoint inhibitor nivolumab, longer-term results from a phase II clinical trial show.
Among 117 patients with squamous cell NSCLC that had progressed either during or after platinum-based doublet chemotherapy and at least one additional systemic regimen, the 12-month and 18-month overall survival (OS) rates were 39% and 27% respectively.
Historically, 1-year OS rates for patients with advanced squamous NSCLC treated with systemic chemotherapy ranged from 5% to 22%, and 2-year OS was less than 5%, said Dr. Leora Horn of the Vanderbilt-Ingram Cancer Center in Nashville, Tennessee.
“Consistent with randomized phase III trials, most treatment-related adverse events were of low grade and manageable with established safety guidelines, and most patients who experienced a treatment-related selected adverse event reported their first event within 3-6 months of treatment initiation. Clinical benefit with nivolumab was observed independent of [programmed death ligand 1] expression,” Dr. Horn said at a world conference on lung cancer.
Nivolumab (Opdivo) is an immune checkpoint inhibitor that has shown a survival benefit compared with docetaxel in patients with NSCLC of both adenocarcinoma and squamous cell histologies. It is approved in the U.S. for the treatment of metastatic squamous cell NSCLC which has progressed on therapy with a platinum-based doublet chemotherapy.
Dr. Horn presented updated efficacy and safety results from the previously reported phase II Checkmate 063 trial.
The investigators screened 140 and treated 117 patients with stage IIIB to stage IV squamous NSCLC who had disease progression on two or more prior lines of systemic chemotherapy and an Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1.
The patients received nivolumab 3 mg/kg by intravenous infusion every 2 weeks until disease progression or unacceptable toxicity.
As of July 2014, the date of the first analysis, after a median follow-up of 8 months the median overall survival was 8.2 months, and the 1-year overall survival rate was 41%.
Updated survival data as of June 2015, showed that the median OS was 8.1 months, and the overall survival at 1 year was 39%. As noted before, the 18-month overall survival was 27%.
An analysis stratified by PD-L1 expression showed a median OS of 8.3 months for patients with less than 1% of tumor cells expressing PD-L1, compared with 10.1 months for patients with 1% or more of tumor cells expressing the ligand. Among 10 patients not evaluable for PD-L1 expression, the median OS was 13 months.
Adverse event (AE) rates remained stable from 1 year to the next, with 74% of patients in July 2014 having had any grade of AE, compared with 75% of patients in June 2015. The percentage of patients who experienced a grade 3 or 4 adverse event was 17% for each analysis, indicating that the grade 3/4 events occurred early in the course of treatment. The two treatment-related deaths, one from hypoxic pneumonia and the other from ischemic stroke, were reported in the previous analysis.
The most common treatment-related select adverse events of any grade were gastrointestinal (10% in the current and previous analyses), pulmonary (6% vs. 5% in the previous analysis), and skin related (16% vs. 15%, respectively).
The survival data are consistent with those seen in the long-term follow-up data from the Checkmate 017 trial, in which nivolumab was compared with docetaxel in patients with advanced squamous NSCLC, commented Dr. Solange Peters of the Lausanne (Switzerland) University Hospital, the invited discussant.
The slightly better survival in the 017 trial is likely due to the fact the majority of patients in the 063 trial were heavily pretreated, with 65% having received three or more prior lines of therapy, Dr. Peters said at the conference sponsored by the International Association for the Study of Lung Cancer.
DENVER – Patients with heavily pretreated advanced squamous cell non–small cell lung cancer (NSCLC) continued to receive clinical benefit from treatment with the checkpoint inhibitor nivolumab, longer-term results from a phase II clinical trial show.
Among 117 patients with squamous cell NSCLC that had progressed either during or after platinum-based doublet chemotherapy and at least one additional systemic regimen, the 12-month and 18-month overall survival (OS) rates were 39% and 27% respectively.
Historically, 1-year OS rates for patients with advanced squamous NSCLC treated with systemic chemotherapy ranged from 5% to 22%, and 2-year OS was less than 5%, said Dr. Leora Horn of the Vanderbilt-Ingram Cancer Center in Nashville, Tennessee.
“Consistent with randomized phase III trials, most treatment-related adverse events were of low grade and manageable with established safety guidelines, and most patients who experienced a treatment-related selected adverse event reported their first event within 3-6 months of treatment initiation. Clinical benefit with nivolumab was observed independent of [programmed death ligand 1] expression,” Dr. Horn said at a world conference on lung cancer.
Nivolumab (Opdivo) is an immune checkpoint inhibitor that has shown a survival benefit compared with docetaxel in patients with NSCLC of both adenocarcinoma and squamous cell histologies. It is approved in the U.S. for the treatment of metastatic squamous cell NSCLC which has progressed on therapy with a platinum-based doublet chemotherapy.
Dr. Horn presented updated efficacy and safety results from the previously reported phase II Checkmate 063 trial.
The investigators screened 140 and treated 117 patients with stage IIIB to stage IV squamous NSCLC who had disease progression on two or more prior lines of systemic chemotherapy and an Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1.
The patients received nivolumab 3 mg/kg by intravenous infusion every 2 weeks until disease progression or unacceptable toxicity.
As of July 2014, the date of the first analysis, after a median follow-up of 8 months the median overall survival was 8.2 months, and the 1-year overall survival rate was 41%.
Updated survival data as of June 2015, showed that the median OS was 8.1 months, and the overall survival at 1 year was 39%. As noted before, the 18-month overall survival was 27%.
An analysis stratified by PD-L1 expression showed a median OS of 8.3 months for patients with less than 1% of tumor cells expressing PD-L1, compared with 10.1 months for patients with 1% or more of tumor cells expressing the ligand. Among 10 patients not evaluable for PD-L1 expression, the median OS was 13 months.
Adverse event (AE) rates remained stable from 1 year to the next, with 74% of patients in July 2014 having had any grade of AE, compared with 75% of patients in June 2015. The percentage of patients who experienced a grade 3 or 4 adverse event was 17% for each analysis, indicating that the grade 3/4 events occurred early in the course of treatment. The two treatment-related deaths, one from hypoxic pneumonia and the other from ischemic stroke, were reported in the previous analysis.
The most common treatment-related select adverse events of any grade were gastrointestinal (10% in the current and previous analyses), pulmonary (6% vs. 5% in the previous analysis), and skin related (16% vs. 15%, respectively).
The survival data are consistent with those seen in the long-term follow-up data from the Checkmate 017 trial, in which nivolumab was compared with docetaxel in patients with advanced squamous NSCLC, commented Dr. Solange Peters of the Lausanne (Switzerland) University Hospital, the invited discussant.
The slightly better survival in the 017 trial is likely due to the fact the majority of patients in the 063 trial were heavily pretreated, with 65% having received three or more prior lines of therapy, Dr. Peters said at the conference sponsored by the International Association for the Study of Lung Cancer.
Key clinical point: Nivolumab was associated with improved survival among patients with heavily pretreated squamous cell NSCLC.
Major finding: 1-year overall survival with nivolumab was 39%; 18-month OS was 27%.
Data source: Long-term results of an open-label, phase II trial in 117 patients with advanced squamous cell NSCLC.
Disclosures: The study was funded by Bristol-Myers Squibb. Dr. Horn and Dr. Peters disclosed serving as consultants to the company.