User login
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
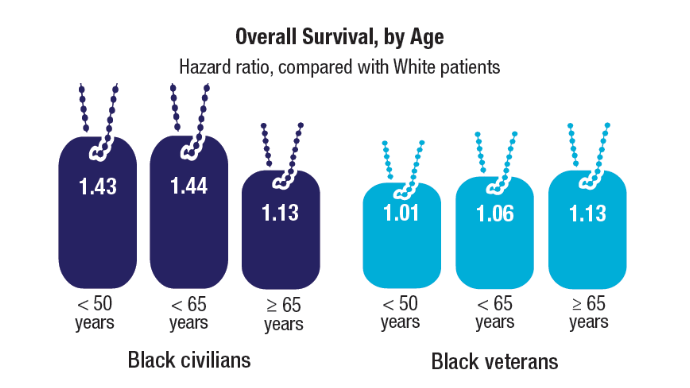
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
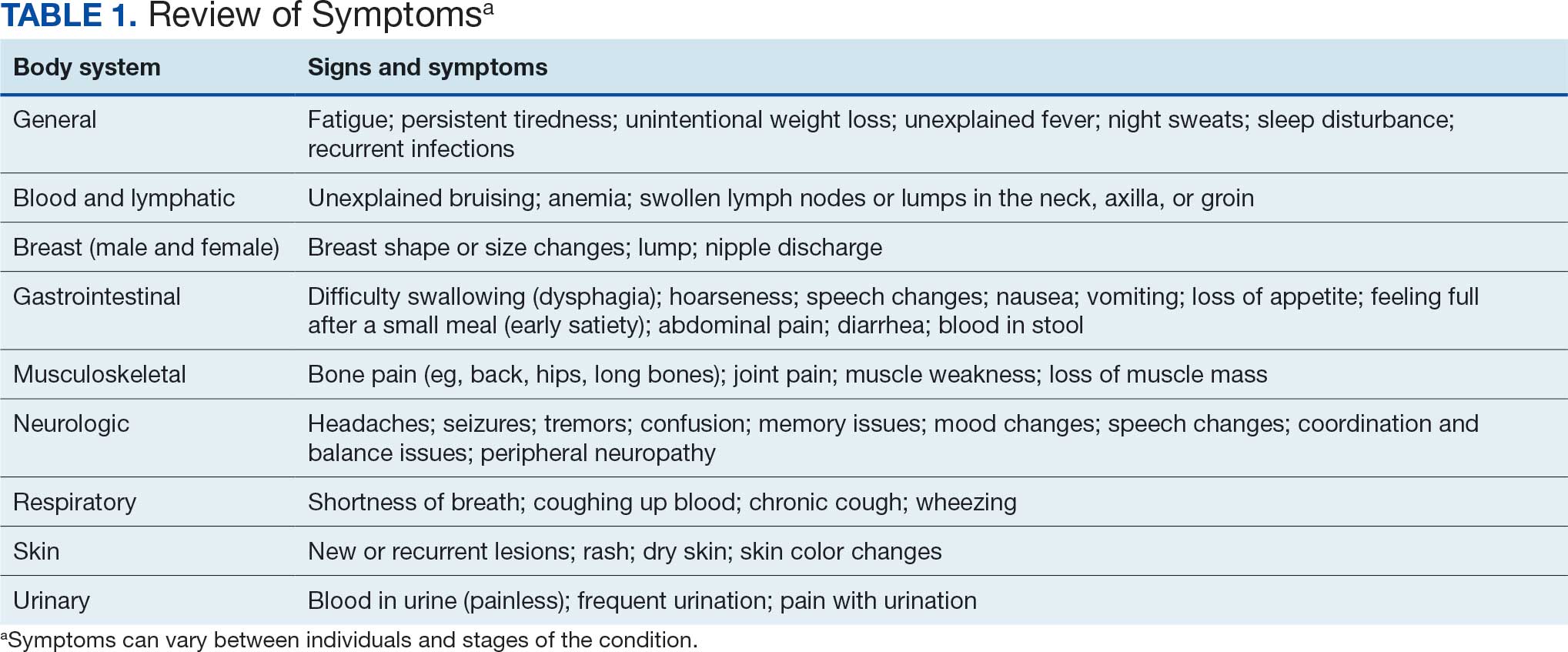
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).
Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
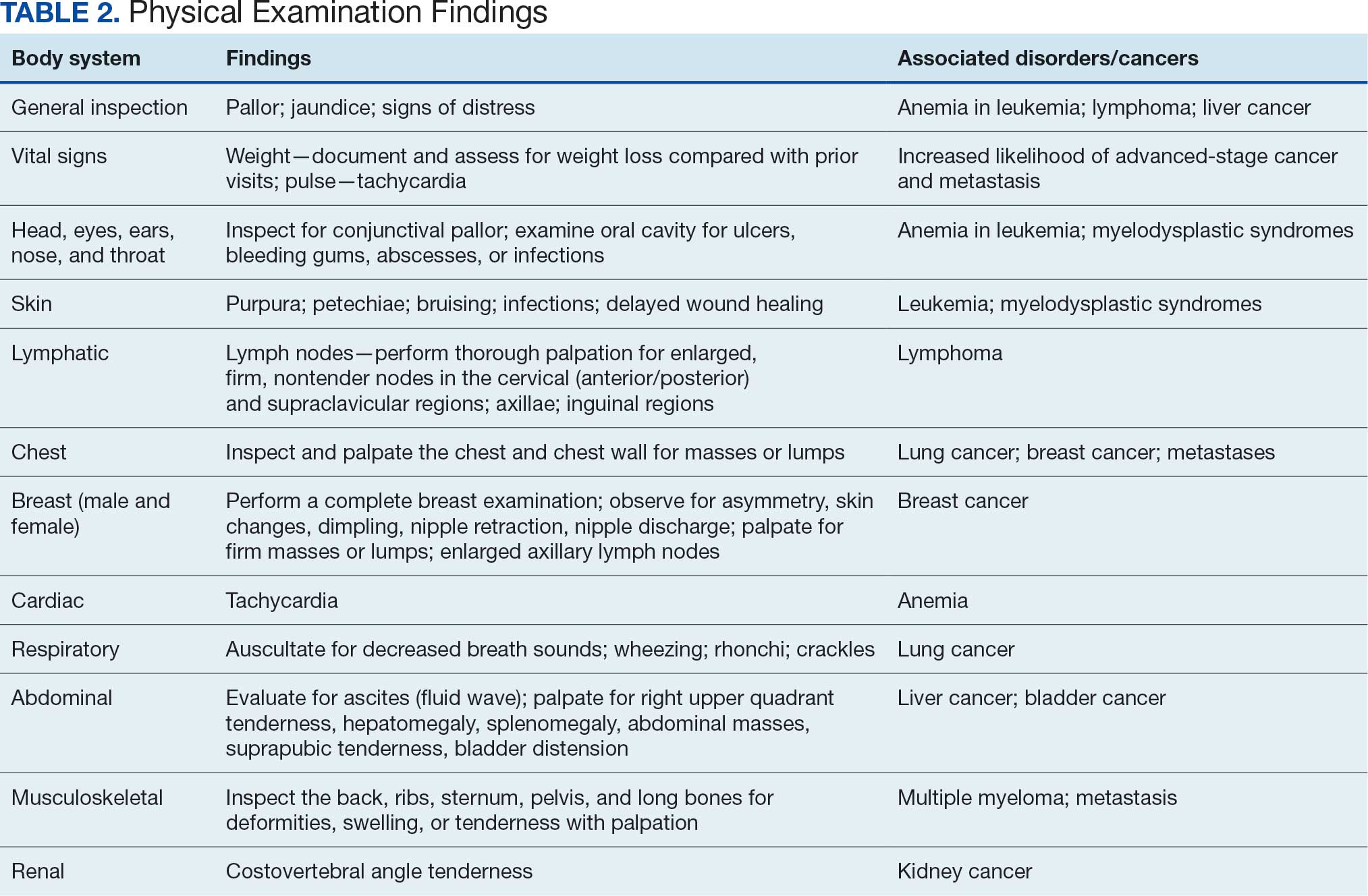
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.
Proposed Diagnostic Screening Panel
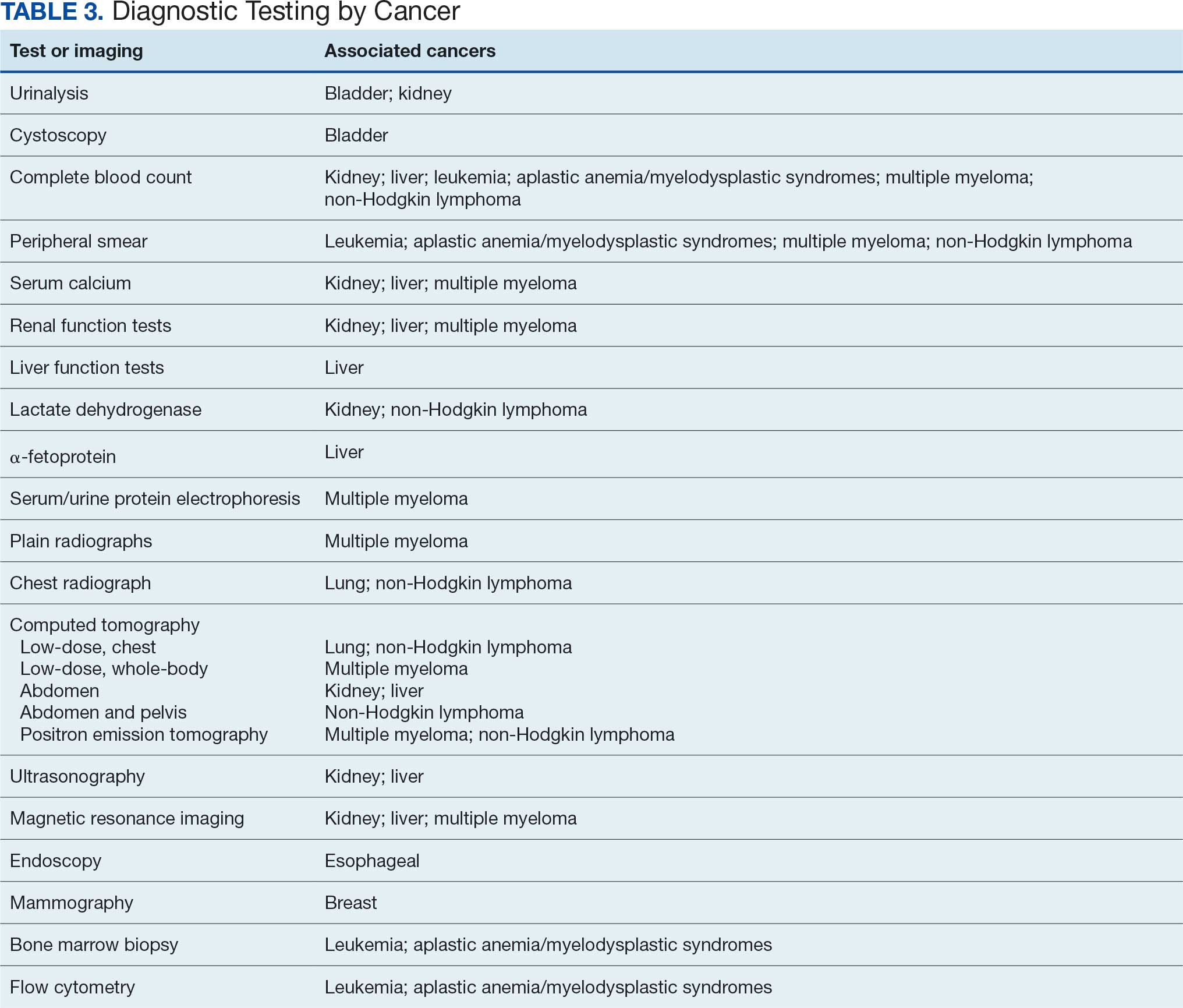
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).
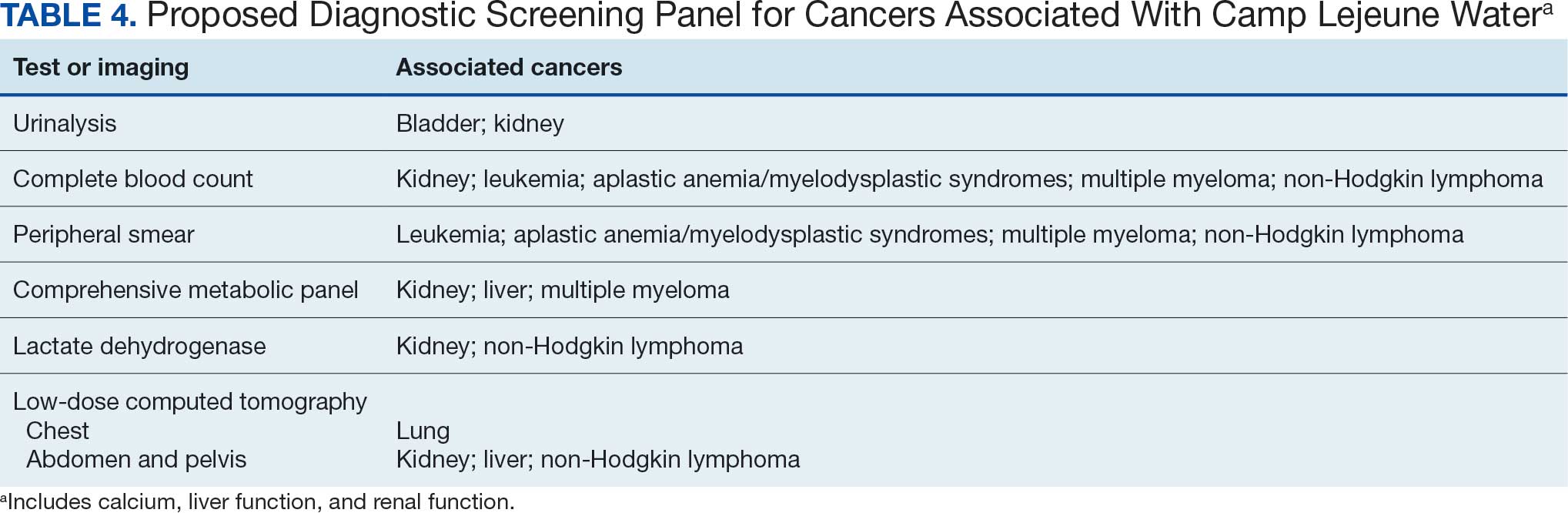
A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).

Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.

Proposed Diagnostic Screening Panel
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).


A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).

Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.

Proposed Diagnostic Screening Panel
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).


A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Cancer Data Trends 2025
The annual issue of Cancer Data Trends, produced in collaboration with the Association of VA Hematology/Oncology (AVAHO), highlights the latest research in some of the top cancers impacting US veterans.
In this issue:
- Access, Race, and "Colon Age": Improving CRC Screening
- Lung Cancer: Mortality Trends in Veterans and New Treatments
- Racial Disparities, Germline Testing, and Improved Overall Survival in Prostate Cancer
- Breast and Uterine Cancer: Screening Guidelines, Genetic Testing, and Mortality Trends
- HCC Updates: Quality Care Framework and Risk Stratification Data
- Rising Kidney Cancer Cases and Emerging Treatments for Veterans
- Advances in Blood Cancer Care for Veterans
- AI-Based Risk Stratification for Oropharyngeal Carcinomas: AIROC
- Brain Cancer: Epidemiology, TBI, and New Treatments
The annual issue of Cancer Data Trends, produced in collaboration with the Association of VA Hematology/Oncology (AVAHO), highlights the latest research in some of the top cancers impacting US veterans.
In this issue:
- Access, Race, and "Colon Age": Improving CRC Screening
- Lung Cancer: Mortality Trends in Veterans and New Treatments
- Racial Disparities, Germline Testing, and Improved Overall Survival in Prostate Cancer
- Breast and Uterine Cancer: Screening Guidelines, Genetic Testing, and Mortality Trends
- HCC Updates: Quality Care Framework and Risk Stratification Data
- Rising Kidney Cancer Cases and Emerging Treatments for Veterans
- Advances in Blood Cancer Care for Veterans
- AI-Based Risk Stratification for Oropharyngeal Carcinomas: AIROC
- Brain Cancer: Epidemiology, TBI, and New Treatments
The annual issue of Cancer Data Trends, produced in collaboration with the Association of VA Hematology/Oncology (AVAHO), highlights the latest research in some of the top cancers impacting US veterans.
In this issue:
- Access, Race, and "Colon Age": Improving CRC Screening
- Lung Cancer: Mortality Trends in Veterans and New Treatments
- Racial Disparities, Germline Testing, and Improved Overall Survival in Prostate Cancer
- Breast and Uterine Cancer: Screening Guidelines, Genetic Testing, and Mortality Trends
- HCC Updates: Quality Care Framework and Risk Stratification Data
- Rising Kidney Cancer Cases and Emerging Treatments for Veterans
- Advances in Blood Cancer Care for Veterans
- AI-Based Risk Stratification for Oropharyngeal Carcinomas: AIROC
- Brain Cancer: Epidemiology, TBI, and New Treatments
Wildfire Smoke Linked to Potential Risks for Some Cancers
Wildfire smoke exposure may be associated with increased risks for multiple types of cancer, suggests an analysis of prospective cohort data from over 90,000 individuals.
To determine how this widespread pollution might be affecting cancer risk, senior author Shuguang Leng, MBBS, PhD, and colleagues analyzed data from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial. That prospective national study enrolled approximately 154,000 participants between 1993 and 2001 and tracked cancer incidence through 2018. Of these, 91,460 participants had wildfire smoke exposure data and were included in the analysis.
During the 2006-2018 exposure period, the investigators identified incident cases of 242 ovarian, 800 colorectal, 896 bladder, 1696 hematopoietic, 1739 breast, and 1758 lung cancers, as well as 1127 melanoma cases. The median 36-month moving average for wildfire smoke PM2.5 (fine particulate matter) across the cohort was 0.37 µg/m3.
Wildfire smoke exposure was significantly associated with increased risks for lung, colorectal, breast, bladder, and hematopoietic cancer, according to the results of the study presented by Leng at American Association for Cancer Research (AACR) Annual Meeting 2026.
Each 1 µg/m3 increase in the 36-month moving average of wildfire smoke PM2.5 was associated with a 63% higher risk for hematopoietic cancer (HR, 1.63; 95% CI, 1.02-2.60), a nearly twofold higher risk for lung cancer (hazard ratio [HR], 1.92; 95% CI, 1.18-3.15), more than twofold higher risks for breast cancer (HR, 2.09; 95% CI, 1.34-3.26) and colorectal cancer (HR, 2.31; 95% CI, 1.11-4.81), and a more than threefold higher risk for bladder cancer (HR, 3.49; 95% CI, 1.66-7.34). No significant associations were observed for ovarian cancer or melanoma.
The investigators quantified wildfire smoke exposure at each participant’s residence on a monthly basis using three measures: near-ground wildfire smoke PM2.5, wildfire smoke black carbon, and satellite-derived wildfire smoke plume-day counts, with measurements available from 2006 until first cancer diagnosis or last contact.
Given evidence that 3 years of air pollution exposure can influence the development of epidermal growth factor receptor-positive lung adenocarcinoma, the team modeled exposure as a time-varying variable using 36-month moving averages preceding each month. HRs were estimated using Cox proportional hazards models stratified by study center, with restricted cubic splines applied to evaluate dose-response relationships. Models were adjusted for age, sex, race and ethnicity, education, smoking history, BMI, and trial arm.
All five cancer types linked with wildfire smoke exposure showed linear dose-response relationships, Leng noted, “which means the higher the exposure, the higher the cancer risk.”
Results based on wildfire smoke plume-day counts were generally consistent with those for PM2.5, while associations for black carbon exposure were observed only for breast and bladder cancers.
With wildfires on the rise, these findings suggest that the resulting smoke may become a “major driver for cancer burden in the US in the coming decades,” said Leng, of the University of New Mexico Comprehensive Cancer Center, Albuquerque, New Mexico.
“Wildfire smoke has become a major source of air pollution in the United States,” he continued. Large fires in the US are three times more common than they were 50 years ago, and the “tons of toxicants and particles” released by these fires “can travel hundreds of miles to affect communities far away.”
The investigators also conducted histology-specific analyses, finding that adenocarcinoma showed the strongest association with wildfire smoke among lung cancer subtypes. Among colorectal cancers, proximal tumors appeared more sensitive to wildfire smoke exposure, while among bladder cancers, the association was strongest for muscle-invasive disease.
Wildfire Smoke Exposure Expected to Rise
Under even the most conservative climate projections, wildfire smoke exposure in the US is expected to rise over the next 20-30 years, Leng said.
Annual average wildfire smoke PM2.5 levels, currently estimated at around 0.5 µg/m3, could rise to 1 µg/m3. Based on the study’s dose-response data, this would correspond to substantially greater cancer risk.
There will be “a much larger area” of the US exposed “at a much higher dose,” Leng predicted.
Mitigating the Risks of Wildfire Smoke
This is a “strong hypothesis-generating study,” Jun Wu, PhD, professor of environmental and occupational health at the UC Irvine Program in Public Health, Irvine, California, told Medscape Medical News.
“This is one of the first large, prospective US cohort studies to examine wildfire smoke specifically in relation to cancer risk, especially cancer sites beyond the lung,” Wu said. “A major strength is that the PLCO platform has around 91,000 participants with longitudinal follow-up and detailed covariate data, including smoking history, which is often a weak point in previous air pollution-cancer studies.”
According to Wu, who was not involved in the analysis but recently published data linking wildfire smoke exposure to preterm birth, the reported risks for colorectal, breast, bladder, and hematopoietic cancers represent novel contributions to the literature. However, she cautioned against viewing the specific HRs as a precise estimates of risk due to wide confidence intervals.
The findings should encourage individuals, public health officials, and clinicians to mitigate the risks of wildfire smoke, Wu said.
Specifically, she suggested that public health assessments expand beyond acute outcomes like emergency department visits to include long-term endpoints such as cancer, while community clean-air shelters need to be made more widely available.
She advised clinicians to incorporate wildfire exposure into routine patient histories and to provide vulnerable patients — such as those with asthma, chronic obstructive pulmonary disease, heart failure, or pregnancy — with smoke-season action plans.
Risk mitigation begins with awareness, according to Wu, who advised individuals check their local air quality index on AirNow.gov or PurpleAir.
On smoky days, she suggested prioritizing indoor air quality by keeping windows closed and running air purifiers. If going outside on such days is necessary, she suggested an N95 or KN95 mask, as these offer “meaningful protection,” while cloth and surgical masks do not.
These preventive steps may have once been out of the ordinary, Wu said, but the risk for wildfire smoke exposure is becoming a part of everyday life.
“The common thread is a shift in framing,” Wu said. “Wildfire smoke has traditionally been treated as an acute event, but the emerging evidence points to a chronic environmental exposure. Both our clinical and public health systems have room to grow into that reality.”
The analysis was funded by the National Institutes of Health. The investigators and Wu reported having no conflicts of interest.
This article was previously published on Medscape.
Wildfire smoke exposure may be associated with increased risks for multiple types of cancer, suggests an analysis of prospective cohort data from over 90,000 individuals.
To determine how this widespread pollution might be affecting cancer risk, senior author Shuguang Leng, MBBS, PhD, and colleagues analyzed data from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial. That prospective national study enrolled approximately 154,000 participants between 1993 and 2001 and tracked cancer incidence through 2018. Of these, 91,460 participants had wildfire smoke exposure data and were included in the analysis.
During the 2006-2018 exposure period, the investigators identified incident cases of 242 ovarian, 800 colorectal, 896 bladder, 1696 hematopoietic, 1739 breast, and 1758 lung cancers, as well as 1127 melanoma cases. The median 36-month moving average for wildfire smoke PM2.5 (fine particulate matter) across the cohort was 0.37 µg/m3.
Wildfire smoke exposure was significantly associated with increased risks for lung, colorectal, breast, bladder, and hematopoietic cancer, according to the results of the study presented by Leng at American Association for Cancer Research (AACR) Annual Meeting 2026.
Each 1 µg/m3 increase in the 36-month moving average of wildfire smoke PM2.5 was associated with a 63% higher risk for hematopoietic cancer (HR, 1.63; 95% CI, 1.02-2.60), a nearly twofold higher risk for lung cancer (hazard ratio [HR], 1.92; 95% CI, 1.18-3.15), more than twofold higher risks for breast cancer (HR, 2.09; 95% CI, 1.34-3.26) and colorectal cancer (HR, 2.31; 95% CI, 1.11-4.81), and a more than threefold higher risk for bladder cancer (HR, 3.49; 95% CI, 1.66-7.34). No significant associations were observed for ovarian cancer or melanoma.
The investigators quantified wildfire smoke exposure at each participant’s residence on a monthly basis using three measures: near-ground wildfire smoke PM2.5, wildfire smoke black carbon, and satellite-derived wildfire smoke plume-day counts, with measurements available from 2006 until first cancer diagnosis or last contact.
Given evidence that 3 years of air pollution exposure can influence the development of epidermal growth factor receptor-positive lung adenocarcinoma, the team modeled exposure as a time-varying variable using 36-month moving averages preceding each month. HRs were estimated using Cox proportional hazards models stratified by study center, with restricted cubic splines applied to evaluate dose-response relationships. Models were adjusted for age, sex, race and ethnicity, education, smoking history, BMI, and trial arm.
All five cancer types linked with wildfire smoke exposure showed linear dose-response relationships, Leng noted, “which means the higher the exposure, the higher the cancer risk.”
Results based on wildfire smoke plume-day counts were generally consistent with those for PM2.5, while associations for black carbon exposure were observed only for breast and bladder cancers.
With wildfires on the rise, these findings suggest that the resulting smoke may become a “major driver for cancer burden in the US in the coming decades,” said Leng, of the University of New Mexico Comprehensive Cancer Center, Albuquerque, New Mexico.
“Wildfire smoke has become a major source of air pollution in the United States,” he continued. Large fires in the US are three times more common than they were 50 years ago, and the “tons of toxicants and particles” released by these fires “can travel hundreds of miles to affect communities far away.”
The investigators also conducted histology-specific analyses, finding that adenocarcinoma showed the strongest association with wildfire smoke among lung cancer subtypes. Among colorectal cancers, proximal tumors appeared more sensitive to wildfire smoke exposure, while among bladder cancers, the association was strongest for muscle-invasive disease.
Wildfire Smoke Exposure Expected to Rise
Under even the most conservative climate projections, wildfire smoke exposure in the US is expected to rise over the next 20-30 years, Leng said.
Annual average wildfire smoke PM2.5 levels, currently estimated at around 0.5 µg/m3, could rise to 1 µg/m3. Based on the study’s dose-response data, this would correspond to substantially greater cancer risk.
There will be “a much larger area” of the US exposed “at a much higher dose,” Leng predicted.
Mitigating the Risks of Wildfire Smoke
This is a “strong hypothesis-generating study,” Jun Wu, PhD, professor of environmental and occupational health at the UC Irvine Program in Public Health, Irvine, California, told Medscape Medical News.
“This is one of the first large, prospective US cohort studies to examine wildfire smoke specifically in relation to cancer risk, especially cancer sites beyond the lung,” Wu said. “A major strength is that the PLCO platform has around 91,000 participants with longitudinal follow-up and detailed covariate data, including smoking history, which is often a weak point in previous air pollution-cancer studies.”
According to Wu, who was not involved in the analysis but recently published data linking wildfire smoke exposure to preterm birth, the reported risks for colorectal, breast, bladder, and hematopoietic cancers represent novel contributions to the literature. However, she cautioned against viewing the specific HRs as a precise estimates of risk due to wide confidence intervals.
The findings should encourage individuals, public health officials, and clinicians to mitigate the risks of wildfire smoke, Wu said.
Specifically, she suggested that public health assessments expand beyond acute outcomes like emergency department visits to include long-term endpoints such as cancer, while community clean-air shelters need to be made more widely available.
She advised clinicians to incorporate wildfire exposure into routine patient histories and to provide vulnerable patients — such as those with asthma, chronic obstructive pulmonary disease, heart failure, or pregnancy — with smoke-season action plans.
Risk mitigation begins with awareness, according to Wu, who advised individuals check their local air quality index on AirNow.gov or PurpleAir.
On smoky days, she suggested prioritizing indoor air quality by keeping windows closed and running air purifiers. If going outside on such days is necessary, she suggested an N95 or KN95 mask, as these offer “meaningful protection,” while cloth and surgical masks do not.
These preventive steps may have once been out of the ordinary, Wu said, but the risk for wildfire smoke exposure is becoming a part of everyday life.
“The common thread is a shift in framing,” Wu said. “Wildfire smoke has traditionally been treated as an acute event, but the emerging evidence points to a chronic environmental exposure. Both our clinical and public health systems have room to grow into that reality.”
The analysis was funded by the National Institutes of Health. The investigators and Wu reported having no conflicts of interest.
This article was previously published on Medscape.
Wildfire smoke exposure may be associated with increased risks for multiple types of cancer, suggests an analysis of prospective cohort data from over 90,000 individuals.
To determine how this widespread pollution might be affecting cancer risk, senior author Shuguang Leng, MBBS, PhD, and colleagues analyzed data from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial. That prospective national study enrolled approximately 154,000 participants between 1993 and 2001 and tracked cancer incidence through 2018. Of these, 91,460 participants had wildfire smoke exposure data and were included in the analysis.
During the 2006-2018 exposure period, the investigators identified incident cases of 242 ovarian, 800 colorectal, 896 bladder, 1696 hematopoietic, 1739 breast, and 1758 lung cancers, as well as 1127 melanoma cases. The median 36-month moving average for wildfire smoke PM2.5 (fine particulate matter) across the cohort was 0.37 µg/m3.
Wildfire smoke exposure was significantly associated with increased risks for lung, colorectal, breast, bladder, and hematopoietic cancer, according to the results of the study presented by Leng at American Association for Cancer Research (AACR) Annual Meeting 2026.
Each 1 µg/m3 increase in the 36-month moving average of wildfire smoke PM2.5 was associated with a 63% higher risk for hematopoietic cancer (HR, 1.63; 95% CI, 1.02-2.60), a nearly twofold higher risk for lung cancer (hazard ratio [HR], 1.92; 95% CI, 1.18-3.15), more than twofold higher risks for breast cancer (HR, 2.09; 95% CI, 1.34-3.26) and colorectal cancer (HR, 2.31; 95% CI, 1.11-4.81), and a more than threefold higher risk for bladder cancer (HR, 3.49; 95% CI, 1.66-7.34). No significant associations were observed for ovarian cancer or melanoma.
The investigators quantified wildfire smoke exposure at each participant’s residence on a monthly basis using three measures: near-ground wildfire smoke PM2.5, wildfire smoke black carbon, and satellite-derived wildfire smoke plume-day counts, with measurements available from 2006 until first cancer diagnosis or last contact.
Given evidence that 3 years of air pollution exposure can influence the development of epidermal growth factor receptor-positive lung adenocarcinoma, the team modeled exposure as a time-varying variable using 36-month moving averages preceding each month. HRs were estimated using Cox proportional hazards models stratified by study center, with restricted cubic splines applied to evaluate dose-response relationships. Models were adjusted for age, sex, race and ethnicity, education, smoking history, BMI, and trial arm.
All five cancer types linked with wildfire smoke exposure showed linear dose-response relationships, Leng noted, “which means the higher the exposure, the higher the cancer risk.”
Results based on wildfire smoke plume-day counts were generally consistent with those for PM2.5, while associations for black carbon exposure were observed only for breast and bladder cancers.
With wildfires on the rise, these findings suggest that the resulting smoke may become a “major driver for cancer burden in the US in the coming decades,” said Leng, of the University of New Mexico Comprehensive Cancer Center, Albuquerque, New Mexico.
“Wildfire smoke has become a major source of air pollution in the United States,” he continued. Large fires in the US are three times more common than they were 50 years ago, and the “tons of toxicants and particles” released by these fires “can travel hundreds of miles to affect communities far away.”
The investigators also conducted histology-specific analyses, finding that adenocarcinoma showed the strongest association with wildfire smoke among lung cancer subtypes. Among colorectal cancers, proximal tumors appeared more sensitive to wildfire smoke exposure, while among bladder cancers, the association was strongest for muscle-invasive disease.
Wildfire Smoke Exposure Expected to Rise
Under even the most conservative climate projections, wildfire smoke exposure in the US is expected to rise over the next 20-30 years, Leng said.
Annual average wildfire smoke PM2.5 levels, currently estimated at around 0.5 µg/m3, could rise to 1 µg/m3. Based on the study’s dose-response data, this would correspond to substantially greater cancer risk.
There will be “a much larger area” of the US exposed “at a much higher dose,” Leng predicted.
Mitigating the Risks of Wildfire Smoke
This is a “strong hypothesis-generating study,” Jun Wu, PhD, professor of environmental and occupational health at the UC Irvine Program in Public Health, Irvine, California, told Medscape Medical News.
“This is one of the first large, prospective US cohort studies to examine wildfire smoke specifically in relation to cancer risk, especially cancer sites beyond the lung,” Wu said. “A major strength is that the PLCO platform has around 91,000 participants with longitudinal follow-up and detailed covariate data, including smoking history, which is often a weak point in previous air pollution-cancer studies.”
According to Wu, who was not involved in the analysis but recently published data linking wildfire smoke exposure to preterm birth, the reported risks for colorectal, breast, bladder, and hematopoietic cancers represent novel contributions to the literature. However, she cautioned against viewing the specific HRs as a precise estimates of risk due to wide confidence intervals.
The findings should encourage individuals, public health officials, and clinicians to mitigate the risks of wildfire smoke, Wu said.
Specifically, she suggested that public health assessments expand beyond acute outcomes like emergency department visits to include long-term endpoints such as cancer, while community clean-air shelters need to be made more widely available.
She advised clinicians to incorporate wildfire exposure into routine patient histories and to provide vulnerable patients — such as those with asthma, chronic obstructive pulmonary disease, heart failure, or pregnancy — with smoke-season action plans.
Risk mitigation begins with awareness, according to Wu, who advised individuals check their local air quality index on AirNow.gov or PurpleAir.
On smoky days, she suggested prioritizing indoor air quality by keeping windows closed and running air purifiers. If going outside on such days is necessary, she suggested an N95 or KN95 mask, as these offer “meaningful protection,” while cloth and surgical masks do not.
These preventive steps may have once been out of the ordinary, Wu said, but the risk for wildfire smoke exposure is becoming a part of everyday life.
“The common thread is a shift in framing,” Wu said. “Wildfire smoke has traditionally been treated as an acute event, but the emerging evidence points to a chronic environmental exposure. Both our clinical and public health systems have room to grow into that reality.”
The analysis was funded by the National Institutes of Health. The investigators and Wu reported having no conflicts of interest.
This article was previously published on Medscape.
Rising Lung Cancer Burden Among Women
Rising Lung Cancer Burden Among Women
While the incidence of lung cancer is decreasing in men, it continues to rise in women. With more than 19,000 new cases in France each year, lung cancer is now the third most commonly diagnosed cancer among women. This trend is also seen in other European countries but appears to be region-specific because other continents report a decline in incidence among women. Moreover, although overall prognosis remains better in the female population, the trend is worrying: Mortality associated with the disease is increasing in women, unlike in men with lung cancer. A session at the French-Language Pneumology Congress held from January 30 to February 1, 2026, in Lille, France, provided an opportunity to review the situation.
Efficacy and Toxicity
Lung tumors in women have a distinct tumor profile: Women have a higher proportion of adenocarcinomas than men and a higher frequency of somatic mutations (EGFR, BRAF, or HER2), including in nonsmokers. In addition, 65% of lung cancers in women are associated with smoking compared with 87% of those in men.
The role of estrogens is central because they interact directly with tumor growth signaling pathways. Moreover, “sex is the second leading factor of variability in drug pharmacokinetics after weight and accounts for 28% of anticancer drug kinetics,” emphasized Julien Mazières, pulmonologist, Toulouse University Hospital, Toulouse, France. Also involved in this equation are a higher body fat percentage, lower gastric acidity, and, above all, reduced renal and hepatic clearance.
As a result, exposure to drugs — represented by the area under the curve — is often greater in women and translates into not only improved progression-free survival with targeted therapies and chemotherapy but also increased toxicity. Carboplatin and paclitaxel are among the drugs whose kinetics are most affected by clearance. There are differences in clearance of more than 20% for these drugs in women vs men, though dosages are not systematically adjusted except for weight-based dosing. This vulnerability to adverse effects is particularly pronounced with targeted therapies, with more neuropsychiatric and gastrointestinal disorders. Data on the efficacy of immunotherapy in lung cancer by sex are contradictory. However, endocrine-related adverse effects and pneumonitis are more frequent in women, especially before menopause.
Women remain underrepresented in clinical trials, and sex-specific analyses of results are too rarely performed, which limits understanding of mechanisms and prevents tailoring management recommendations according to sex.
Impaired Quality of Life
Lung cancer most severely impairs physical functioning in women. “In the absence of sex-stratified studies, psycho-oncologists’ experience suggests that women have more cognitive disorders, anxiety, and depression associated with this disease. Its impact on quality of life is major, with deterioration of social relationships and reduced treatment adherence,” summarized Céline Mascaux, MD, PhD, pulmonologist, Strasbourg University Hospital, Strasbourg, France. Women also face social and family pressure — a mental burden that pushes them to “hold on” for their loved ones. Regarding sexual health, women with lung cancer who are sexually active often report dissatisfaction with the quality of their sexual relations because of fatigue, lack of energy, sadness, and shortness of breath, not to mention treatment-related sexual dysfunction. These problems are often not given sufficient attention by physicians.
Finally, fertility requires greater attention from the medical community: According to the VICAN study conducted by France’s National Health Insurance Fund, a discussion about fertility preservation did not take place at the time of cancer diagnosis for 60% of men and 67% of women of childbearing age. “In lung cancer specifically, the desire for children nevertheless exists in nearly 40% of patients of childbearing age,” regretted Jacques Cadranel, pulmonologist, Tenon Hospital, Paris, France. This desire does not appear to have influenced therapeutic strategy, and fertility preservation was ultimately proposed in only a third of cases and was carried out in only 3% of women compared with21% of men.
This story has been translated from Univadis France, part of the Medscape Professional Network.
A version of this story first appeared on Medscape.com
While the incidence of lung cancer is decreasing in men, it continues to rise in women. With more than 19,000 new cases in France each year, lung cancer is now the third most commonly diagnosed cancer among women. This trend is also seen in other European countries but appears to be region-specific because other continents report a decline in incidence among women. Moreover, although overall prognosis remains better in the female population, the trend is worrying: Mortality associated with the disease is increasing in women, unlike in men with lung cancer. A session at the French-Language Pneumology Congress held from January 30 to February 1, 2026, in Lille, France, provided an opportunity to review the situation.
Efficacy and Toxicity
Lung tumors in women have a distinct tumor profile: Women have a higher proportion of adenocarcinomas than men and a higher frequency of somatic mutations (EGFR, BRAF, or HER2), including in nonsmokers. In addition, 65% of lung cancers in women are associated with smoking compared with 87% of those in men.
The role of estrogens is central because they interact directly with tumor growth signaling pathways. Moreover, “sex is the second leading factor of variability in drug pharmacokinetics after weight and accounts for 28% of anticancer drug kinetics,” emphasized Julien Mazières, pulmonologist, Toulouse University Hospital, Toulouse, France. Also involved in this equation are a higher body fat percentage, lower gastric acidity, and, above all, reduced renal and hepatic clearance.
As a result, exposure to drugs — represented by the area under the curve — is often greater in women and translates into not only improved progression-free survival with targeted therapies and chemotherapy but also increased toxicity. Carboplatin and paclitaxel are among the drugs whose kinetics are most affected by clearance. There are differences in clearance of more than 20% for these drugs in women vs men, though dosages are not systematically adjusted except for weight-based dosing. This vulnerability to adverse effects is particularly pronounced with targeted therapies, with more neuropsychiatric and gastrointestinal disorders. Data on the efficacy of immunotherapy in lung cancer by sex are contradictory. However, endocrine-related adverse effects and pneumonitis are more frequent in women, especially before menopause.
Women remain underrepresented in clinical trials, and sex-specific analyses of results are too rarely performed, which limits understanding of mechanisms and prevents tailoring management recommendations according to sex.
Impaired Quality of Life
Lung cancer most severely impairs physical functioning in women. “In the absence of sex-stratified studies, psycho-oncologists’ experience suggests that women have more cognitive disorders, anxiety, and depression associated with this disease. Its impact on quality of life is major, with deterioration of social relationships and reduced treatment adherence,” summarized Céline Mascaux, MD, PhD, pulmonologist, Strasbourg University Hospital, Strasbourg, France. Women also face social and family pressure — a mental burden that pushes them to “hold on” for their loved ones. Regarding sexual health, women with lung cancer who are sexually active often report dissatisfaction with the quality of their sexual relations because of fatigue, lack of energy, sadness, and shortness of breath, not to mention treatment-related sexual dysfunction. These problems are often not given sufficient attention by physicians.
Finally, fertility requires greater attention from the medical community: According to the VICAN study conducted by France’s National Health Insurance Fund, a discussion about fertility preservation did not take place at the time of cancer diagnosis for 60% of men and 67% of women of childbearing age. “In lung cancer specifically, the desire for children nevertheless exists in nearly 40% of patients of childbearing age,” regretted Jacques Cadranel, pulmonologist, Tenon Hospital, Paris, France. This desire does not appear to have influenced therapeutic strategy, and fertility preservation was ultimately proposed in only a third of cases and was carried out in only 3% of women compared with21% of men.
This story has been translated from Univadis France, part of the Medscape Professional Network.
A version of this story first appeared on Medscape.com
While the incidence of lung cancer is decreasing in men, it continues to rise in women. With more than 19,000 new cases in France each year, lung cancer is now the third most commonly diagnosed cancer among women. This trend is also seen in other European countries but appears to be region-specific because other continents report a decline in incidence among women. Moreover, although overall prognosis remains better in the female population, the trend is worrying: Mortality associated with the disease is increasing in women, unlike in men with lung cancer. A session at the French-Language Pneumology Congress held from January 30 to February 1, 2026, in Lille, France, provided an opportunity to review the situation.
Efficacy and Toxicity
Lung tumors in women have a distinct tumor profile: Women have a higher proportion of adenocarcinomas than men and a higher frequency of somatic mutations (EGFR, BRAF, or HER2), including in nonsmokers. In addition, 65% of lung cancers in women are associated with smoking compared with 87% of those in men.
The role of estrogens is central because they interact directly with tumor growth signaling pathways. Moreover, “sex is the second leading factor of variability in drug pharmacokinetics after weight and accounts for 28% of anticancer drug kinetics,” emphasized Julien Mazières, pulmonologist, Toulouse University Hospital, Toulouse, France. Also involved in this equation are a higher body fat percentage, lower gastric acidity, and, above all, reduced renal and hepatic clearance.
As a result, exposure to drugs — represented by the area under the curve — is often greater in women and translates into not only improved progression-free survival with targeted therapies and chemotherapy but also increased toxicity. Carboplatin and paclitaxel are among the drugs whose kinetics are most affected by clearance. There are differences in clearance of more than 20% for these drugs in women vs men, though dosages are not systematically adjusted except for weight-based dosing. This vulnerability to adverse effects is particularly pronounced with targeted therapies, with more neuropsychiatric and gastrointestinal disorders. Data on the efficacy of immunotherapy in lung cancer by sex are contradictory. However, endocrine-related adverse effects and pneumonitis are more frequent in women, especially before menopause.
Women remain underrepresented in clinical trials, and sex-specific analyses of results are too rarely performed, which limits understanding of mechanisms and prevents tailoring management recommendations according to sex.
Impaired Quality of Life
Lung cancer most severely impairs physical functioning in women. “In the absence of sex-stratified studies, psycho-oncologists’ experience suggests that women have more cognitive disorders, anxiety, and depression associated with this disease. Its impact on quality of life is major, with deterioration of social relationships and reduced treatment adherence,” summarized Céline Mascaux, MD, PhD, pulmonologist, Strasbourg University Hospital, Strasbourg, France. Women also face social and family pressure — a mental burden that pushes them to “hold on” for their loved ones. Regarding sexual health, women with lung cancer who are sexually active often report dissatisfaction with the quality of their sexual relations because of fatigue, lack of energy, sadness, and shortness of breath, not to mention treatment-related sexual dysfunction. These problems are often not given sufficient attention by physicians.
Finally, fertility requires greater attention from the medical community: According to the VICAN study conducted by France’s National Health Insurance Fund, a discussion about fertility preservation did not take place at the time of cancer diagnosis for 60% of men and 67% of women of childbearing age. “In lung cancer specifically, the desire for children nevertheless exists in nearly 40% of patients of childbearing age,” regretted Jacques Cadranel, pulmonologist, Tenon Hospital, Paris, France. This desire does not appear to have influenced therapeutic strategy, and fertility preservation was ultimately proposed in only a third of cases and was carried out in only 3% of women compared with21% of men.
This story has been translated from Univadis France, part of the Medscape Professional Network.
A version of this story first appeared on Medscape.com
Rising Lung Cancer Burden Among Women
Rising Lung Cancer Burden Among Women
TB, Chronic Bronchitis Tied to Lung Cancer in Never Smokers
TB, Chronic Bronchitis Tied to Lung Cancer in Never Smokers
TOPLINE:
A history of tuberculosis (TB) and a history of chronic bronchitis were associated with an increased risk for lung cancer in individuals who had never smoked, whereas asthma had a positive, nonsignificant association overall and a significant association in women.
METHODOLOGY:
- Researchers conducted a systematic review and meta-analysis of clinical databases from inception to July 2025, to assess the association between asthma, TB, and/or chronic bronchitis and the risk for lung cancer among participants aged 18 years or older who had never smoked.
- They included data from 20 case-control studies involving 54,135 participants and five cohort studies involving 377,983 participants.
- The primary outcome was the risk for lung cancer among participants with a history of TB, asthma, or chronic bronchitis.
- Participants were labeled as “never smokers” if they were explicitly described in the manuscripts as having “never smoked” or reported smoking < 100 cigarettes in their lifetime.
TAKEAWAY:
- In case-control studies, TB (16 studies) and chronic bronchitis (9 studies) were significantly associated with an increased risk for lung cancer (odds ratio [OR], 1.76; P < .001 and OR, 1.36; P = .012, respectively).
- In four case-cohort studies, TB was associated with an increased but nonsignificant risk for lung cancer (hazard ratio, 1.64).
- Eleven case-control studies demonstrated a positive but nonsignificant association between asthma and the risk for lung cancer (OR, 1.34). However, a significant association emerged when analyses were limited to women (five studies; OR, 1.61; P < .01).
IN PRACTICE:
History of TB was especially associated with increased LC [lung cancer] risk, meriting particular attention for prospective CT screening studies,” the authors of the study wrote.
SOURCE:
This study was led by Nishwant Swami, MD, Hospital of the University of Pennsylvania, Philadelphia. It was published online on January 11, 2026, in Chest.
LIMITATIONS:
Most studies lacked uniform adjustment for key confounders, increasing the risk for residual confounding. The inclusion of few cohort studies in the analysis may have limited the assessment of temporality and precision. Additionally, differences in covariate adjustment, variable definitions, and language restrictions may have limited comparability and generalizability.
DISCLOSURES:
No specific funding was reported for this study. One author reported serving as a consultant or advisor for various companies, including AstraZeneca, Merck, and Pfizer. Another author reported receiving funding, in part, through the Prostate Cancer Foundation Young Investigator Award and through the Cancer Center Support Grant from the National Cancer Institute.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
A history of tuberculosis (TB) and a history of chronic bronchitis were associated with an increased risk for lung cancer in individuals who had never smoked, whereas asthma had a positive, nonsignificant association overall and a significant association in women.
METHODOLOGY:
- Researchers conducted a systematic review and meta-analysis of clinical databases from inception to July 2025, to assess the association between asthma, TB, and/or chronic bronchitis and the risk for lung cancer among participants aged 18 years or older who had never smoked.
- They included data from 20 case-control studies involving 54,135 participants and five cohort studies involving 377,983 participants.
- The primary outcome was the risk for lung cancer among participants with a history of TB, asthma, or chronic bronchitis.
- Participants were labeled as “never smokers” if they were explicitly described in the manuscripts as having “never smoked” or reported smoking < 100 cigarettes in their lifetime.
TAKEAWAY:
- In case-control studies, TB (16 studies) and chronic bronchitis (9 studies) were significantly associated with an increased risk for lung cancer (odds ratio [OR], 1.76; P < .001 and OR, 1.36; P = .012, respectively).
- In four case-cohort studies, TB was associated with an increased but nonsignificant risk for lung cancer (hazard ratio, 1.64).
- Eleven case-control studies demonstrated a positive but nonsignificant association between asthma and the risk for lung cancer (OR, 1.34). However, a significant association emerged when analyses were limited to women (five studies; OR, 1.61; P < .01).
IN PRACTICE:
History of TB was especially associated with increased LC [lung cancer] risk, meriting particular attention for prospective CT screening studies,” the authors of the study wrote.
SOURCE:
This study was led by Nishwant Swami, MD, Hospital of the University of Pennsylvania, Philadelphia. It was published online on January 11, 2026, in Chest.
LIMITATIONS:
Most studies lacked uniform adjustment for key confounders, increasing the risk for residual confounding. The inclusion of few cohort studies in the analysis may have limited the assessment of temporality and precision. Additionally, differences in covariate adjustment, variable definitions, and language restrictions may have limited comparability and generalizability.
DISCLOSURES:
No specific funding was reported for this study. One author reported serving as a consultant or advisor for various companies, including AstraZeneca, Merck, and Pfizer. Another author reported receiving funding, in part, through the Prostate Cancer Foundation Young Investigator Award and through the Cancer Center Support Grant from the National Cancer Institute.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
A history of tuberculosis (TB) and a history of chronic bronchitis were associated with an increased risk for lung cancer in individuals who had never smoked, whereas asthma had a positive, nonsignificant association overall and a significant association in women.
METHODOLOGY:
- Researchers conducted a systematic review and meta-analysis of clinical databases from inception to July 2025, to assess the association between asthma, TB, and/or chronic bronchitis and the risk for lung cancer among participants aged 18 years or older who had never smoked.
- They included data from 20 case-control studies involving 54,135 participants and five cohort studies involving 377,983 participants.
- The primary outcome was the risk for lung cancer among participants with a history of TB, asthma, or chronic bronchitis.
- Participants were labeled as “never smokers” if they were explicitly described in the manuscripts as having “never smoked” or reported smoking < 100 cigarettes in their lifetime.
TAKEAWAY:
- In case-control studies, TB (16 studies) and chronic bronchitis (9 studies) were significantly associated with an increased risk for lung cancer (odds ratio [OR], 1.76; P < .001 and OR, 1.36; P = .012, respectively).
- In four case-cohort studies, TB was associated with an increased but nonsignificant risk for lung cancer (hazard ratio, 1.64).
- Eleven case-control studies demonstrated a positive but nonsignificant association between asthma and the risk for lung cancer (OR, 1.34). However, a significant association emerged when analyses were limited to women (five studies; OR, 1.61; P < .01).
IN PRACTICE:
History of TB was especially associated with increased LC [lung cancer] risk, meriting particular attention for prospective CT screening studies,” the authors of the study wrote.
SOURCE:
This study was led by Nishwant Swami, MD, Hospital of the University of Pennsylvania, Philadelphia. It was published online on January 11, 2026, in Chest.
LIMITATIONS:
Most studies lacked uniform adjustment for key confounders, increasing the risk for residual confounding. The inclusion of few cohort studies in the analysis may have limited the assessment of temporality and precision. Additionally, differences in covariate adjustment, variable definitions, and language restrictions may have limited comparability and generalizability.
DISCLOSURES:
No specific funding was reported for this study. One author reported serving as a consultant or advisor for various companies, including AstraZeneca, Merck, and Pfizer. Another author reported receiving funding, in part, through the Prostate Cancer Foundation Young Investigator Award and through the Cancer Center Support Grant from the National Cancer Institute.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TB, Chronic Bronchitis Tied to Lung Cancer in Never Smokers
TB, Chronic Bronchitis Tied to Lung Cancer in Never Smokers
What Drives Lung Cancer in Nonsmokers?
TOPLINE:
A comprehensive review of 92 studies found that 15% to 20% of lung cancers occurred among nonsmokers and were associated with environmental and germline risk factors. These cancers frequently harbored actionable genomic drivers, and targeted EGFR and ALK therapies produced significant diseasefree survival (DFS) and overall survival benefits.
METHODOLOGY:
- Lung cancer continues to be the leading cause of cancer death worldwide, causing about 1.8 million deaths in 2022, with smoking remaining the predominant risk factor. However, the incidence of lung cancer among nonsmokers (those who have smoked less than 100 cigarettes in their lifetime) is rising, varies by sex and geography, and is linked to environmental exposures and family history. The misperception that lung cancer is almost invariably caused by smoking may delay assessment and diagnosis.
- Researchers conducted a review of 92 studies on lung cancer in nonsmokers: 6 meta-analyses or systematic reviews, 16 randomized clinical trials, eight prospective cohort studies, seven retrospective cohort studies, three cross-sectional studies, four observational or case-control studies, 13 genomic studies, and 35 other studies.
- Overall, lung cancer among nonsmokers accounted for 15% to 20% of all lung cancer cases. Most lung cancers in nonsmokers were adenocarcinomas (60% to 80%), with a median age at diagnosis of 67 years in this group compared with 70 years in people with a history of smoking.
- Data analysis from three US hospital networks showed that the proportion of lung cancer among nonsmokers increased from 8.0% to 14.9% between 1990 and 2013. A pooled analysis of seven Finnish cohorts reported an absolute increase in lung cancer among nonsmokers from 6.9 per 100,000 person-years in 1972 to 12.9 per 100,000 person-years in 2015.
- The age-adjusted incidence rate of lung cancer in the US between 2000 and 2013 was 17.5 per 100,000 individuals among Asian female nonsmokers compared with 10.1 per 100,000 among non-Hispanic White female nonsmokers.
TAKEAWAY:
- Environmental and occupational risk factors were secondhand smoke, residential radon, outdoor and household air pollution (PM2.5), asbestos and silica exposure, and prior thoracic radiotherapy. Having a first-degree relative with lung cancer increased the risk of developing lung cancer, and genome-wide association studies identified susceptibility loci associated with lung cancer risk in nonsmokers.
- Family history and inherited susceptibility increased lung cancer risk in never smokers (odds ratio [OR] for lung cancer in those with a first–degree relative, 1.51), and clonal hematopoiesis was also associated with higher risk (OR, 1.43). Importantly, tumors in nonsmokers were frequently driven by actionable somatic alterations (EGFR mutations, 40% to 60% in nonsmokers compared with 10% in smokers) and enrichment of ALK/ROS1/RET/ERBB2/NTRK/NRG1 fusions; 78% to 92% of adenocarcinomas in nonsmokers harbored actionable drivers (compared with 49.5% in ever smokers), and nonsmokers had a substantially lower tumor mutational burden (10–fold lower).
- Similar to individuals with a history of smoking, nonsmokers with lung cancer presented with cough, pain, dyspnea, or weight loss or had disease detected incidentally. Surgical resection remained the preferred treatment for anatomically resectable lung cancer (stages I-III) in medically eligible patients, with follow-up CT screening recommended every 6 months for 2 to 3 years and then annually.
- Targeted adjuvant therapy substantially improved outcomes for resected EGFR–mutant or ALK–rearranged non-small cell lung cancer (NSCLC). Four-year DFS was increased to 70% with osimertinib compared with 29% with placebo (hazard ratio [HR], 0.23) and 5–year overall survival was increased to 85% compared with 73% (HR, 0.49). Two–year DFS was 93.8% with alectinib compared with 63% with placebo (HR, 0.24). In unresectable EGFR-mutated stage III NSCLC, median progression-free survival was 39.1 months with adjuvant osimertinib compared with 5.6 months with placebo. For resected ALKpositive disease, 2–year DFS was 93.8% with adjuvant alectinib compared with 63.0% with chemotherapy (HR, 0.24).
- However, singleagent single agent programmed cell death protein 1 inhibitors or programmed death-ligand 1 inhibitors demonstrated limited efficacy in EGFR or ALK–driven tumors, and benefit was attenuated in never smokers. Regarding screening and early detection, the US Preventive Services Task Force did not recommend lowdose CT screening for nonsmokers, whereas Taiwan implemented a biennial screening program for selected nonsmoking high–risk groups.
IN PRACTICE:
“Among patients with lung cancer, nonsmoking individuals are more likely to have genomic alterations, such as EGFR mutations or ALK gene rearrangements, and these patients have improved survival when treated with TKIs compared with chemotherapy,” the authors of the study wrote.
SOURCE:
The study, led by Cian Murphy, PhD, Cancer Evolution and Genome Instability Laboratory, Francis Crick Institute, London, England, was published online in JAMA.
LIMITATIONS:
Becausesmoking history was often not included in many databases, cancer registries, and trials, the incidence and prevalence of lung cancer in nonsmokers could not be accurately determined. Additionally, accurate quantification of environmental exposures, such as air pollution, presented significant challenges. The quality of the evidence was not formally evaluated, and some relevant articles may have been missed in the literature review.
DISCLOSURES:
The study received support from multiple organizations, including the Rosetrees Trust, Ruth Strauss Foundation, Cancer Research UK, and the National Health and Medical Research Council. Several authors reported receiving grants or personal fees from and having other ties with various sources. Full disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
A comprehensive review of 92 studies found that 15% to 20% of lung cancers occurred among nonsmokers and were associated with environmental and germline risk factors. These cancers frequently harbored actionable genomic drivers, and targeted EGFR and ALK therapies produced significant diseasefree survival (DFS) and overall survival benefits.
METHODOLOGY:
- Lung cancer continues to be the leading cause of cancer death worldwide, causing about 1.8 million deaths in 2022, with smoking remaining the predominant risk factor. However, the incidence of lung cancer among nonsmokers (those who have smoked less than 100 cigarettes in their lifetime) is rising, varies by sex and geography, and is linked to environmental exposures and family history. The misperception that lung cancer is almost invariably caused by smoking may delay assessment and diagnosis.
- Researchers conducted a review of 92 studies on lung cancer in nonsmokers: 6 meta-analyses or systematic reviews, 16 randomized clinical trials, eight prospective cohort studies, seven retrospective cohort studies, three cross-sectional studies, four observational or case-control studies, 13 genomic studies, and 35 other studies.
- Overall, lung cancer among nonsmokers accounted for 15% to 20% of all lung cancer cases. Most lung cancers in nonsmokers were adenocarcinomas (60% to 80%), with a median age at diagnosis of 67 years in this group compared with 70 years in people with a history of smoking.
- Data analysis from three US hospital networks showed that the proportion of lung cancer among nonsmokers increased from 8.0% to 14.9% between 1990 and 2013. A pooled analysis of seven Finnish cohorts reported an absolute increase in lung cancer among nonsmokers from 6.9 per 100,000 person-years in 1972 to 12.9 per 100,000 person-years in 2015.
- The age-adjusted incidence rate of lung cancer in the US between 2000 and 2013 was 17.5 per 100,000 individuals among Asian female nonsmokers compared with 10.1 per 100,000 among non-Hispanic White female nonsmokers.
TAKEAWAY:
- Environmental and occupational risk factors were secondhand smoke, residential radon, outdoor and household air pollution (PM2.5), asbestos and silica exposure, and prior thoracic radiotherapy. Having a first-degree relative with lung cancer increased the risk of developing lung cancer, and genome-wide association studies identified susceptibility loci associated with lung cancer risk in nonsmokers.
- Family history and inherited susceptibility increased lung cancer risk in never smokers (odds ratio [OR] for lung cancer in those with a first–degree relative, 1.51), and clonal hematopoiesis was also associated with higher risk (OR, 1.43). Importantly, tumors in nonsmokers were frequently driven by actionable somatic alterations (EGFR mutations, 40% to 60% in nonsmokers compared with 10% in smokers) and enrichment of ALK/ROS1/RET/ERBB2/NTRK/NRG1 fusions; 78% to 92% of adenocarcinomas in nonsmokers harbored actionable drivers (compared with 49.5% in ever smokers), and nonsmokers had a substantially lower tumor mutational burden (10–fold lower).
- Similar to individuals with a history of smoking, nonsmokers with lung cancer presented with cough, pain, dyspnea, or weight loss or had disease detected incidentally. Surgical resection remained the preferred treatment for anatomically resectable lung cancer (stages I-III) in medically eligible patients, with follow-up CT screening recommended every 6 months for 2 to 3 years and then annually.
- Targeted adjuvant therapy substantially improved outcomes for resected EGFR–mutant or ALK–rearranged non-small cell lung cancer (NSCLC). Four-year DFS was increased to 70% with osimertinib compared with 29% with placebo (hazard ratio [HR], 0.23) and 5–year overall survival was increased to 85% compared with 73% (HR, 0.49). Two–year DFS was 93.8% with alectinib compared with 63% with placebo (HR, 0.24). In unresectable EGFR-mutated stage III NSCLC, median progression-free survival was 39.1 months with adjuvant osimertinib compared with 5.6 months with placebo. For resected ALKpositive disease, 2–year DFS was 93.8% with adjuvant alectinib compared with 63.0% with chemotherapy (HR, 0.24).
- However, singleagent single agent programmed cell death protein 1 inhibitors or programmed death-ligand 1 inhibitors demonstrated limited efficacy in EGFR or ALK–driven tumors, and benefit was attenuated in never smokers. Regarding screening and early detection, the US Preventive Services Task Force did not recommend lowdose CT screening for nonsmokers, whereas Taiwan implemented a biennial screening program for selected nonsmoking high–risk groups.
IN PRACTICE:
“Among patients with lung cancer, nonsmoking individuals are more likely to have genomic alterations, such as EGFR mutations or ALK gene rearrangements, and these patients have improved survival when treated with TKIs compared with chemotherapy,” the authors of the study wrote.
SOURCE:
The study, led by Cian Murphy, PhD, Cancer Evolution and Genome Instability Laboratory, Francis Crick Institute, London, England, was published online in JAMA.
LIMITATIONS:
Becausesmoking history was often not included in many databases, cancer registries, and trials, the incidence and prevalence of lung cancer in nonsmokers could not be accurately determined. Additionally, accurate quantification of environmental exposures, such as air pollution, presented significant challenges. The quality of the evidence was not formally evaluated, and some relevant articles may have been missed in the literature review.
DISCLOSURES:
The study received support from multiple organizations, including the Rosetrees Trust, Ruth Strauss Foundation, Cancer Research UK, and the National Health and Medical Research Council. Several authors reported receiving grants or personal fees from and having other ties with various sources. Full disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
A comprehensive review of 92 studies found that 15% to 20% of lung cancers occurred among nonsmokers and were associated with environmental and germline risk factors. These cancers frequently harbored actionable genomic drivers, and targeted EGFR and ALK therapies produced significant diseasefree survival (DFS) and overall survival benefits.
METHODOLOGY:
- Lung cancer continues to be the leading cause of cancer death worldwide, causing about 1.8 million deaths in 2022, with smoking remaining the predominant risk factor. However, the incidence of lung cancer among nonsmokers (those who have smoked less than 100 cigarettes in their lifetime) is rising, varies by sex and geography, and is linked to environmental exposures and family history. The misperception that lung cancer is almost invariably caused by smoking may delay assessment and diagnosis.
- Researchers conducted a review of 92 studies on lung cancer in nonsmokers: 6 meta-analyses or systematic reviews, 16 randomized clinical trials, eight prospective cohort studies, seven retrospective cohort studies, three cross-sectional studies, four observational or case-control studies, 13 genomic studies, and 35 other studies.
- Overall, lung cancer among nonsmokers accounted for 15% to 20% of all lung cancer cases. Most lung cancers in nonsmokers were adenocarcinomas (60% to 80%), with a median age at diagnosis of 67 years in this group compared with 70 years in people with a history of smoking.
- Data analysis from three US hospital networks showed that the proportion of lung cancer among nonsmokers increased from 8.0% to 14.9% between 1990 and 2013. A pooled analysis of seven Finnish cohorts reported an absolute increase in lung cancer among nonsmokers from 6.9 per 100,000 person-years in 1972 to 12.9 per 100,000 person-years in 2015.
- The age-adjusted incidence rate of lung cancer in the US between 2000 and 2013 was 17.5 per 100,000 individuals among Asian female nonsmokers compared with 10.1 per 100,000 among non-Hispanic White female nonsmokers.
TAKEAWAY:
- Environmental and occupational risk factors were secondhand smoke, residential radon, outdoor and household air pollution (PM2.5), asbestos and silica exposure, and prior thoracic radiotherapy. Having a first-degree relative with lung cancer increased the risk of developing lung cancer, and genome-wide association studies identified susceptibility loci associated with lung cancer risk in nonsmokers.
- Family history and inherited susceptibility increased lung cancer risk in never smokers (odds ratio [OR] for lung cancer in those with a first–degree relative, 1.51), and clonal hematopoiesis was also associated with higher risk (OR, 1.43). Importantly, tumors in nonsmokers were frequently driven by actionable somatic alterations (EGFR mutations, 40% to 60% in nonsmokers compared with 10% in smokers) and enrichment of ALK/ROS1/RET/ERBB2/NTRK/NRG1 fusions; 78% to 92% of adenocarcinomas in nonsmokers harbored actionable drivers (compared with 49.5% in ever smokers), and nonsmokers had a substantially lower tumor mutational burden (10–fold lower).
- Similar to individuals with a history of smoking, nonsmokers with lung cancer presented with cough, pain, dyspnea, or weight loss or had disease detected incidentally. Surgical resection remained the preferred treatment for anatomically resectable lung cancer (stages I-III) in medically eligible patients, with follow-up CT screening recommended every 6 months for 2 to 3 years and then annually.
- Targeted adjuvant therapy substantially improved outcomes for resected EGFR–mutant or ALK–rearranged non-small cell lung cancer (NSCLC). Four-year DFS was increased to 70% with osimertinib compared with 29% with placebo (hazard ratio [HR], 0.23) and 5–year overall survival was increased to 85% compared with 73% (HR, 0.49). Two–year DFS was 93.8% with alectinib compared with 63% with placebo (HR, 0.24). In unresectable EGFR-mutated stage III NSCLC, median progression-free survival was 39.1 months with adjuvant osimertinib compared with 5.6 months with placebo. For resected ALKpositive disease, 2–year DFS was 93.8% with adjuvant alectinib compared with 63.0% with chemotherapy (HR, 0.24).
- However, singleagent single agent programmed cell death protein 1 inhibitors or programmed death-ligand 1 inhibitors demonstrated limited efficacy in EGFR or ALK–driven tumors, and benefit was attenuated in never smokers. Regarding screening and early detection, the US Preventive Services Task Force did not recommend lowdose CT screening for nonsmokers, whereas Taiwan implemented a biennial screening program for selected nonsmoking high–risk groups.
IN PRACTICE:
“Among patients with lung cancer, nonsmoking individuals are more likely to have genomic alterations, such as EGFR mutations or ALK gene rearrangements, and these patients have improved survival when treated with TKIs compared with chemotherapy,” the authors of the study wrote.
SOURCE:
The study, led by Cian Murphy, PhD, Cancer Evolution and Genome Instability Laboratory, Francis Crick Institute, London, England, was published online in JAMA.
LIMITATIONS:
Becausesmoking history was often not included in many databases, cancer registries, and trials, the incidence and prevalence of lung cancer in nonsmokers could not be accurately determined. Additionally, accurate quantification of environmental exposures, such as air pollution, presented significant challenges. The quality of the evidence was not formally evaluated, and some relevant articles may have been missed in the literature review.
DISCLOSURES:
The study received support from multiple organizations, including the Rosetrees Trust, Ruth Strauss Foundation, Cancer Research UK, and the National Health and Medical Research Council. Several authors reported receiving grants or personal fees from and having other ties with various sources. Full disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Weekend Warrior and Regular Physical Activity Patterns Are Associated With Reduced Lung Cancer Risk
TOPLINE:
Compared with inactive patterns, weekend warrior (moderate-to-vigorous physical activity [MVPA] condensed into 1-2 days per week) and regular physical activity patterns were found to be equally effective at reducing the risk for lung cancer. Neither pattern showed significant associations with the overall risk for cancer or specific risks for prostate, breast, and colorectal cancers.
METHODOLOGY:
- This analysis included 80,896 participants (mean age, 55.5 years; 56% women) with valid accelerometer data collected between June 2013 and December 2015.
- Participants were classified into three groups: 32,213 active weekend warriors (≥ 150 minutes of weekly MVPA with ≥ 50% achieved in 1-2 days), 22,162 active regular participants (≥ 150 minutes of MVPA but not meeting a weekend warrior pattern), and 26,521 inactive participants (< 150 minutes of MVPA).
- Researchers tracked associations between physical activity patterns and incident cases of all types of cancer plus specific cases of prostate, breast, colorectal, and lung cancer over a median follow-up duration of 6 years.
TAKEAWAY:
- Compared with inactive patterns, active weekend warrior patterns showed a significant inverse association with the risk for lung cancer (hazard ratio [HR], 0.77; 95% CI, 0.61-0.98).
- Active regular activity patterns demonstrated similar protective effects against lung cancer as inactive patterns (HR, 0.73; 95% CI, 0.56-0.96).
- Neither of the physical activity patterns showed any significant association with the overall risk for cancer or specific risks for prostate, breast, and colorectal cancers.
IN PRACTICE:
"Physical activity condensed into one to two days per week compared with a more balanced weekly distribution was associated with similar risk reductions of incident lung cancer, while neither pattern was associated with reduced overall, prostate, breast, and colorectal cancers," the authors of the study wrote.
SOURCE:
This study was led by Rubén López-Bueno, Department of Physical Medicine and Nursing, University of Zaragoza, Zaragoza, Spain. It was published online on September 06, 2025, in Annals of Medicine.
A version of this article first appeared on Medscape.com.
TOPLINE:
Compared with inactive patterns, weekend warrior (moderate-to-vigorous physical activity [MVPA] condensed into 1-2 days per week) and regular physical activity patterns were found to be equally effective at reducing the risk for lung cancer. Neither pattern showed significant associations with the overall risk for cancer or specific risks for prostate, breast, and colorectal cancers.
METHODOLOGY:
- This analysis included 80,896 participants (mean age, 55.5 years; 56% women) with valid accelerometer data collected between June 2013 and December 2015.
- Participants were classified into three groups: 32,213 active weekend warriors (≥ 150 minutes of weekly MVPA with ≥ 50% achieved in 1-2 days), 22,162 active regular participants (≥ 150 minutes of MVPA but not meeting a weekend warrior pattern), and 26,521 inactive participants (< 150 minutes of MVPA).
- Researchers tracked associations between physical activity patterns and incident cases of all types of cancer plus specific cases of prostate, breast, colorectal, and lung cancer over a median follow-up duration of 6 years.
TAKEAWAY:
- Compared with inactive patterns, active weekend warrior patterns showed a significant inverse association with the risk for lung cancer (hazard ratio [HR], 0.77; 95% CI, 0.61-0.98).
- Active regular activity patterns demonstrated similar protective effects against lung cancer as inactive patterns (HR, 0.73; 95% CI, 0.56-0.96).
- Neither of the physical activity patterns showed any significant association with the overall risk for cancer or specific risks for prostate, breast, and colorectal cancers.
IN PRACTICE:
"Physical activity condensed into one to two days per week compared with a more balanced weekly distribution was associated with similar risk reductions of incident lung cancer, while neither pattern was associated with reduced overall, prostate, breast, and colorectal cancers," the authors of the study wrote.
SOURCE:
This study was led by Rubén López-Bueno, Department of Physical Medicine and Nursing, University of Zaragoza, Zaragoza, Spain. It was published online on September 06, 2025, in Annals of Medicine.
A version of this article first appeared on Medscape.com.
TOPLINE:
Compared with inactive patterns, weekend warrior (moderate-to-vigorous physical activity [MVPA] condensed into 1-2 days per week) and regular physical activity patterns were found to be equally effective at reducing the risk for lung cancer. Neither pattern showed significant associations with the overall risk for cancer or specific risks for prostate, breast, and colorectal cancers.
METHODOLOGY:
- This analysis included 80,896 participants (mean age, 55.5 years; 56% women) with valid accelerometer data collected between June 2013 and December 2015.
- Participants were classified into three groups: 32,213 active weekend warriors (≥ 150 minutes of weekly MVPA with ≥ 50% achieved in 1-2 days), 22,162 active regular participants (≥ 150 minutes of MVPA but not meeting a weekend warrior pattern), and 26,521 inactive participants (< 150 minutes of MVPA).
- Researchers tracked associations between physical activity patterns and incident cases of all types of cancer plus specific cases of prostate, breast, colorectal, and lung cancer over a median follow-up duration of 6 years.
TAKEAWAY:
- Compared with inactive patterns, active weekend warrior patterns showed a significant inverse association with the risk for lung cancer (hazard ratio [HR], 0.77; 95% CI, 0.61-0.98).
- Active regular activity patterns demonstrated similar protective effects against lung cancer as inactive patterns (HR, 0.73; 95% CI, 0.56-0.96).
- Neither of the physical activity patterns showed any significant association with the overall risk for cancer or specific risks for prostate, breast, and colorectal cancers.
IN PRACTICE:
"Physical activity condensed into one to two days per week compared with a more balanced weekly distribution was associated with similar risk reductions of incident lung cancer, while neither pattern was associated with reduced overall, prostate, breast, and colorectal cancers," the authors of the study wrote.
SOURCE:
This study was led by Rubén López-Bueno, Department of Physical Medicine and Nursing, University of Zaragoza, Zaragoza, Spain. It was published online on September 06, 2025, in Annals of Medicine.
A version of this article first appeared on Medscape.com.
Organs of Metastasis Predominate with Age in Non-Small Cell Lung Cancer Subtypes: National Cancer Database Analysis
Background
Patients diagnosed with lung cancer are predominantly non-small cell lung cancer (NSCLC), a leading cause of cancer-related deaths. Thus, it is imperative to investigate and distinguish the differences present at diagnosis to possibly improve survival outcomes. NSCLC commonly metastasizes within older patients near the mean age of 71 years, but also in early onset patients which represents the patients younger than the earliest lung cancer screening age of 50.
Objective
To reveal differences in ratios of metastasis locations in squamous cell carcinoma (SCC), adenocarcinoma (ACC), and adenosquamous carcinoma (ASC).
Methods
The National Cancer Database (NCDB) was utilized to identify patients diagnosed with SCC, ACC, and ASC using the histology codes 8070, 8140, and 8560 from the ICD-O-3.2 from 2004 to 2022. Age groups were 70 years. Metastases located to the brain, liver, bone, and lung were included. Chi-Square tests were performed. The data was analyzed using R version 4.4.2 and statistical significance was set to α = 0.05.
Results
In this study, 1,445,119 patients were analyzed. Chi-Square tests identified significant differences in the ratios of organ metastasis locations between age groups in each subtype (p < 0.001). SCC in each age group similarly metastasized most to bone (36.3%, 34.7%, 34.5%), but notably more local lung metastasis was observed in the oldest group (33.6%). In ACC and ASC, the oldest group also had greater ratios of spread within the lungs (28.0%, 27.2%). Overall, the younger the age group, distant spread to the brain increased (ex. 29.0%, 24.4%, 17.5%). This suggests a widely heterogenous distribution of metastases at diagnosis of NSCLC subtypes and patient age.
Conclusions
This study demonstrated that patients with SCC, ACC, or ASC subtypes of NSCLC share similar predominant locations based in part on patient age, irrespective of cancer origin. NSCLC may more distantly metastasize in younger patients to the brain, while older patients may have locally metastatic cancer. Further analysis of key demographic variables as well as common undertaken treatment options may prove informative and reveal existing differences in survival outcomes.
Background
Patients diagnosed with lung cancer are predominantly non-small cell lung cancer (NSCLC), a leading cause of cancer-related deaths. Thus, it is imperative to investigate and distinguish the differences present at diagnosis to possibly improve survival outcomes. NSCLC commonly metastasizes within older patients near the mean age of 71 years, but also in early onset patients which represents the patients younger than the earliest lung cancer screening age of 50.
Objective
To reveal differences in ratios of metastasis locations in squamous cell carcinoma (SCC), adenocarcinoma (ACC), and adenosquamous carcinoma (ASC).
Methods
The National Cancer Database (NCDB) was utilized to identify patients diagnosed with SCC, ACC, and ASC using the histology codes 8070, 8140, and 8560 from the ICD-O-3.2 from 2004 to 2022. Age groups were 70 years. Metastases located to the brain, liver, bone, and lung were included. Chi-Square tests were performed. The data was analyzed using R version 4.4.2 and statistical significance was set to α = 0.05.
Results
In this study, 1,445,119 patients were analyzed. Chi-Square tests identified significant differences in the ratios of organ metastasis locations between age groups in each subtype (p < 0.001). SCC in each age group similarly metastasized most to bone (36.3%, 34.7%, 34.5%), but notably more local lung metastasis was observed in the oldest group (33.6%). In ACC and ASC, the oldest group also had greater ratios of spread within the lungs (28.0%, 27.2%). Overall, the younger the age group, distant spread to the brain increased (ex. 29.0%, 24.4%, 17.5%). This suggests a widely heterogenous distribution of metastases at diagnosis of NSCLC subtypes and patient age.
Conclusions
This study demonstrated that patients with SCC, ACC, or ASC subtypes of NSCLC share similar predominant locations based in part on patient age, irrespective of cancer origin. NSCLC may more distantly metastasize in younger patients to the brain, while older patients may have locally metastatic cancer. Further analysis of key demographic variables as well as common undertaken treatment options may prove informative and reveal existing differences in survival outcomes.
Background
Patients diagnosed with lung cancer are predominantly non-small cell lung cancer (NSCLC), a leading cause of cancer-related deaths. Thus, it is imperative to investigate and distinguish the differences present at diagnosis to possibly improve survival outcomes. NSCLC commonly metastasizes within older patients near the mean age of 71 years, but also in early onset patients which represents the patients younger than the earliest lung cancer screening age of 50.
Objective
To reveal differences in ratios of metastasis locations in squamous cell carcinoma (SCC), adenocarcinoma (ACC), and adenosquamous carcinoma (ASC).
Methods
The National Cancer Database (NCDB) was utilized to identify patients diagnosed with SCC, ACC, and ASC using the histology codes 8070, 8140, and 8560 from the ICD-O-3.2 from 2004 to 2022. Age groups were 70 years. Metastases located to the brain, liver, bone, and lung were included. Chi-Square tests were performed. The data was analyzed using R version 4.4.2 and statistical significance was set to α = 0.05.
Results
In this study, 1,445,119 patients were analyzed. Chi-Square tests identified significant differences in the ratios of organ metastasis locations between age groups in each subtype (p < 0.001). SCC in each age group similarly metastasized most to bone (36.3%, 34.7%, 34.5%), but notably more local lung metastasis was observed in the oldest group (33.6%). In ACC and ASC, the oldest group also had greater ratios of spread within the lungs (28.0%, 27.2%). Overall, the younger the age group, distant spread to the brain increased (ex. 29.0%, 24.4%, 17.5%). This suggests a widely heterogenous distribution of metastases at diagnosis of NSCLC subtypes and patient age.
Conclusions
This study demonstrated that patients with SCC, ACC, or ASC subtypes of NSCLC share similar predominant locations based in part on patient age, irrespective of cancer origin. NSCLC may more distantly metastasize in younger patients to the brain, while older patients may have locally metastatic cancer. Further analysis of key demographic variables as well as common undertaken treatment options may prove informative and reveal existing differences in survival outcomes.
A Rare Delayed Presentation of Immune-Related Hepatitis in a Patient Treated With Pembrolizumab
Background
Immune checkpoint inhibitors, including pembrolizumab, are associated with a spectrum of immune-related adverse events (irAEs), including immune- mediated hepatitis. Typically, this toxicity manifests within the first 14 weeks of therapy. Delayed presentations beyond one year are exceedingly rare and pose diagnostic challenges.
Case Presentation
We report an elderly patient (over 90 years old) with stage IVa squamous cell carcinoma of the lung and high microsatellite instability (MSI) who had been receiving pembrolizumab since 2023. In 2024—13 months into therapy—he presented with subjective fevers, weakness, and altered mental status. Laboratory evaluation revealed cholestatic jaundice with AST 310 U/L, ALT 291 U/L, alkaline phosphatase 860 U/L, and total bilirubin 5.7 mg/dL. Infectious workup was negative. Imaging via MRCP showed multiple scattered hepatic cysts and a small pancreatic cyst, without biliary obstruction.
Further evaluation, including serologies for hepatitis B and C, CMV, HSV, autoimmune hepatitis panel, iron studies, and ceruloplasmin, was unremarkable except for mildly elevated alpha-1 antitrypsin. Scattered liver cysts were seen on an MRI. The overall findings were most consistent with immune-related hepatitis, as pembrolizumab is known to cause both hepatocellular and cholestatic patterns of liver injury.
The patient was started on high-dose prednisone, resulting in rapid clinical and biochemical improvement. Two weeks post-discharge, liver function tests (LFTs) had markedly improved (bilirubin 1.3, AST 19, ALT 40, ALP 193). Given the severity of transaminitis and hyperbilirubinemia (AST >8x ULN, bilirubin >3x ULN), pembrolizumab was permanently discontinued. LFTs normalized after completion of the steroid taper.
Conclusions
This case highlights a rare instance of delayed immune-related hepatitis occurring over a year after initiation of pembrolizumab, far beyond the typical window of onset. Clinicians should maintain a high index of suspicion for irAEs even in late stages of immunotherapy, particularly when common etiologies are excluded. Prompt recognition and corticosteroid treatment can lead to favorable outcomes, even in older patients.
Background
Immune checkpoint inhibitors, including pembrolizumab, are associated with a spectrum of immune-related adverse events (irAEs), including immune- mediated hepatitis. Typically, this toxicity manifests within the first 14 weeks of therapy. Delayed presentations beyond one year are exceedingly rare and pose diagnostic challenges.
Case Presentation
We report an elderly patient (over 90 years old) with stage IVa squamous cell carcinoma of the lung and high microsatellite instability (MSI) who had been receiving pembrolizumab since 2023. In 2024—13 months into therapy—he presented with subjective fevers, weakness, and altered mental status. Laboratory evaluation revealed cholestatic jaundice with AST 310 U/L, ALT 291 U/L, alkaline phosphatase 860 U/L, and total bilirubin 5.7 mg/dL. Infectious workup was negative. Imaging via MRCP showed multiple scattered hepatic cysts and a small pancreatic cyst, without biliary obstruction.
Further evaluation, including serologies for hepatitis B and C, CMV, HSV, autoimmune hepatitis panel, iron studies, and ceruloplasmin, was unremarkable except for mildly elevated alpha-1 antitrypsin. Scattered liver cysts were seen on an MRI. The overall findings were most consistent with immune-related hepatitis, as pembrolizumab is known to cause both hepatocellular and cholestatic patterns of liver injury.
The patient was started on high-dose prednisone, resulting in rapid clinical and biochemical improvement. Two weeks post-discharge, liver function tests (LFTs) had markedly improved (bilirubin 1.3, AST 19, ALT 40, ALP 193). Given the severity of transaminitis and hyperbilirubinemia (AST >8x ULN, bilirubin >3x ULN), pembrolizumab was permanently discontinued. LFTs normalized after completion of the steroid taper.
Conclusions
This case highlights a rare instance of delayed immune-related hepatitis occurring over a year after initiation of pembrolizumab, far beyond the typical window of onset. Clinicians should maintain a high index of suspicion for irAEs even in late stages of immunotherapy, particularly when common etiologies are excluded. Prompt recognition and corticosteroid treatment can lead to favorable outcomes, even in older patients.
Background
Immune checkpoint inhibitors, including pembrolizumab, are associated with a spectrum of immune-related adverse events (irAEs), including immune- mediated hepatitis. Typically, this toxicity manifests within the first 14 weeks of therapy. Delayed presentations beyond one year are exceedingly rare and pose diagnostic challenges.
Case Presentation
We report an elderly patient (over 90 years old) with stage IVa squamous cell carcinoma of the lung and high microsatellite instability (MSI) who had been receiving pembrolizumab since 2023. In 2024—13 months into therapy—he presented with subjective fevers, weakness, and altered mental status. Laboratory evaluation revealed cholestatic jaundice with AST 310 U/L, ALT 291 U/L, alkaline phosphatase 860 U/L, and total bilirubin 5.7 mg/dL. Infectious workup was negative. Imaging via MRCP showed multiple scattered hepatic cysts and a small pancreatic cyst, without biliary obstruction.
Further evaluation, including serologies for hepatitis B and C, CMV, HSV, autoimmune hepatitis panel, iron studies, and ceruloplasmin, was unremarkable except for mildly elevated alpha-1 antitrypsin. Scattered liver cysts were seen on an MRI. The overall findings were most consistent with immune-related hepatitis, as pembrolizumab is known to cause both hepatocellular and cholestatic patterns of liver injury.
The patient was started on high-dose prednisone, resulting in rapid clinical and biochemical improvement. Two weeks post-discharge, liver function tests (LFTs) had markedly improved (bilirubin 1.3, AST 19, ALT 40, ALP 193). Given the severity of transaminitis and hyperbilirubinemia (AST >8x ULN, bilirubin >3x ULN), pembrolizumab was permanently discontinued. LFTs normalized after completion of the steroid taper.
Conclusions
This case highlights a rare instance of delayed immune-related hepatitis occurring over a year after initiation of pembrolizumab, far beyond the typical window of onset. Clinicians should maintain a high index of suspicion for irAEs even in late stages of immunotherapy, particularly when common etiologies are excluded. Prompt recognition and corticosteroid treatment can lead to favorable outcomes, even in older patients.
Prognosis Paradox: Does HLA-B27 Improve the Prognosis of Immune-Related Pneumonitis in Metastatic Lung Cancer?
Background
Immune related adverse events (irAE) are a well-known complication in the treatment of nonsmall cell lung cancer (NSCLCA) with checkpoint inhibitors and have been shown to improve overall survival (OS) and progression free survival (PFS) across multiple studies. However, studies have shown that the prognosis of NSCLCA differs depending on the type of immune related adverse event and the grade of the irAE. For instance, patients who experienced endocrine irAEs like thyroid, or adrenal insufficiency tended to have an improved OS and PFS, whereas patients who developed pneumonitis that required discontinuation of checkpoint inhibitors had worse OS and PFS. While the literature describes the prognostic impacts of irAEs on NSCLCA, there is still a dearth of information on the implications of HLA supertypes on the prognosis of NSCLCA following irAEs.
Case Presentation
To address this point and to ask a question, we would like to share the case of a patient with a 10-year history of inflammatory arthropathy related to HLA-B27 antigen prior to his diagnosis of T2bN2M1b adenosquamous lung cancer with liver metastases. The tumor was 100% PD-L1 expressive and the patient was treated with pembrolizumab. The patient developed central adrenal insufficiency 10 months after pembrolizumab was initiated which was treated with physiologic dosing of hydrocortisone. The patient later developed a grade 3 pneumonitis 62 months after initiation of pembrolizumab and was treated with systemic glucocorticoids. Due to recurrent hospitalizations for pneumonitis, pembrolizumab was discontinued at 70 months post initiation. At the time of discontinuation PET was positive. However, there was a decrease in hyperactivity of the primary tumor at 4 months post discontinuation of pembrolizumab and there have been serial negative PETS from 7 months to 13 months post discontinuation. This led us to ask the question of whether HLA-B27 is protective of the poor prognostic immune related pneumonitis in this patient?
Background
Immune related adverse events (irAE) are a well-known complication in the treatment of nonsmall cell lung cancer (NSCLCA) with checkpoint inhibitors and have been shown to improve overall survival (OS) and progression free survival (PFS) across multiple studies. However, studies have shown that the prognosis of NSCLCA differs depending on the type of immune related adverse event and the grade of the irAE. For instance, patients who experienced endocrine irAEs like thyroid, or adrenal insufficiency tended to have an improved OS and PFS, whereas patients who developed pneumonitis that required discontinuation of checkpoint inhibitors had worse OS and PFS. While the literature describes the prognostic impacts of irAEs on NSCLCA, there is still a dearth of information on the implications of HLA supertypes on the prognosis of NSCLCA following irAEs.
Case Presentation
To address this point and to ask a question, we would like to share the case of a patient with a 10-year history of inflammatory arthropathy related to HLA-B27 antigen prior to his diagnosis of T2bN2M1b adenosquamous lung cancer with liver metastases. The tumor was 100% PD-L1 expressive and the patient was treated with pembrolizumab. The patient developed central adrenal insufficiency 10 months after pembrolizumab was initiated which was treated with physiologic dosing of hydrocortisone. The patient later developed a grade 3 pneumonitis 62 months after initiation of pembrolizumab and was treated with systemic glucocorticoids. Due to recurrent hospitalizations for pneumonitis, pembrolizumab was discontinued at 70 months post initiation. At the time of discontinuation PET was positive. However, there was a decrease in hyperactivity of the primary tumor at 4 months post discontinuation of pembrolizumab and there have been serial negative PETS from 7 months to 13 months post discontinuation. This led us to ask the question of whether HLA-B27 is protective of the poor prognostic immune related pneumonitis in this patient?
Background
Immune related adverse events (irAE) are a well-known complication in the treatment of nonsmall cell lung cancer (NSCLCA) with checkpoint inhibitors and have been shown to improve overall survival (OS) and progression free survival (PFS) across multiple studies. However, studies have shown that the prognosis of NSCLCA differs depending on the type of immune related adverse event and the grade of the irAE. For instance, patients who experienced endocrine irAEs like thyroid, or adrenal insufficiency tended to have an improved OS and PFS, whereas patients who developed pneumonitis that required discontinuation of checkpoint inhibitors had worse OS and PFS. While the literature describes the prognostic impacts of irAEs on NSCLCA, there is still a dearth of information on the implications of HLA supertypes on the prognosis of NSCLCA following irAEs.
Case Presentation
To address this point and to ask a question, we would like to share the case of a patient with a 10-year history of inflammatory arthropathy related to HLA-B27 antigen prior to his diagnosis of T2bN2M1b adenosquamous lung cancer with liver metastases. The tumor was 100% PD-L1 expressive and the patient was treated with pembrolizumab. The patient developed central adrenal insufficiency 10 months after pembrolizumab was initiated which was treated with physiologic dosing of hydrocortisone. The patient later developed a grade 3 pneumonitis 62 months after initiation of pembrolizumab and was treated with systemic glucocorticoids. Due to recurrent hospitalizations for pneumonitis, pembrolizumab was discontinued at 70 months post initiation. At the time of discontinuation PET was positive. However, there was a decrease in hyperactivity of the primary tumor at 4 months post discontinuation of pembrolizumab and there have been serial negative PETS from 7 months to 13 months post discontinuation. This led us to ask the question of whether HLA-B27 is protective of the poor prognostic immune related pneumonitis in this patient?