User login
Watchful waiting doesn't pay for asymptomatic inguinal hernias
INDIANAPOLIS – The luster has suddenly worn off the time-honored strategy of nonoperative watchful waiting in men with minimally symptomatic inguinal hernias.
New evidence indicates the vast majority of men with asymptomatic or minimally symptomatic inguinal hernias will eventually come to surgery. This may not occur until years down the road, when their advanced age may render surgery more arduous.
"Although watchful waiting remains a safe strategy, even on long-term follow-up, patients who present to their physician to have their hernia evaluated, especially if elderly, should be informed that almost certainly they will come to surgery eventually ... The logical assumption is that watchful waiting is not an effective strategy, as with time almost all men cross over," Dr. Robert J. Fitzgibbons Jr. explained at the annual meeting of the American Surgical Association.
He presented an extended follow-up of patients enrolled in a landmark randomized multicenter clinical trial, one of only two randomized studies ever done comparing watchful waiting versus routine surgical repair for men with minimally symptomatic inguinal hernia. In the earlier report by Dr. Fitzgibbons and coworkers, watchful waiting was deemed "an acceptable option" because only 23% of patients crossed over to surgery due to increased pain during the first 2 years of follow-up (JAMA 2006;295:285-92).
At the ASA meeting, however, he presented updated 10-year follow-up data on 167 patients from the cohort initially assigned to watchful waiting. The rate of crossover to surgery was 68% by 10 years, with a marked age-based divergence. Patients below age 65 had a 62% crossover rate, while those above that age had a 79% crossover rate, according to Dr. Fitzgibbons, professor of surgery and chief of the division of general surgery at Creighton University, Omaha, Neb.
The good news was that hernia incarceration was a rare event, occurring at a rate of just 0.2% per year over the course of 10 years.
"We as surgeons have been taught for many years that we must repair all our hernias to prevent hernia accidents. Well, only three patients in our whole study developed incarceration, for which they underwent surgery with no mortality," Dr. Fitzgibbons noted. "The risk of a hernia accident should not be considered an indication for surgery. Older studies in the literature which would suggest otherwise can no longer be considered relevant."
He offered a caveat regarding the study findings: Participants were enrolled after they sought medical attention because of their hernias, even though they were asymptomatic or only minimally symptomatic. So the study results are most applicable to men concerned enough about their hernias that they visit a physician for that reason.
"It’s probably not valid to extrapolate the conclusions in this study to the entire population of minimally symptomatic inguinal hernia patients," the surgeon said. "Physicians have been observing elderly patients for years and would be loath to believe a crossover rate this high."
Nevertheless, the results of this study are virtually identical to those of the only other randomized trial of watchful waiting, which was conducted by surgeons at the University of Glasgow. The most recent update from that study showed an estimated crossover rate in the watchful waiting group of 16% at 1 year, 54% at 5 years, and 72% at 7.5 years. As in the American study, the rate of acute incarceration was reassuringly small. The investigators concluded that watchful waiting appears pointless, and they recommended surgical repair for medically fit patients (Br. J. Surg. 2011;98:596-9).
Discussant Dr. Michael E. Zenilman commented that his own approach is to individualize patient management based in large part upon activity level.
"When I see patients who are 80 years old in the office with an asymptomatic hernia, my conversation with them is about what their lifestyle is like. If they’re an active golfer I know that they’re going to end up getting their hernia fixed. If they’re sedentary, sitting at home in retirement, they don’t. So I think the next step in your research project should be to find out what the activity level is of these patients who are getting older and have asymptomatic hernias," said Dr. Zenilman, vice chair and regional director of surgery for the Washington, D.C., region, Johns Hopkins Medicine.
Dr. Fitzgibbons’ trial was funded by the Agency for Healthcare Research and Quality with support from the American College of Surgeons. He reported having no financial conflicts.
This report is finally a victory for surgeons who plead for common sense in the pursuit of evidence-based practice. Dr. Robert J. Fitzgibbons, recognized as one of surgery's foremost experts in hernia repair, presents long-term follow-up of patients with inguinal hernia who are treated expectantly. There are three very significant results. First, complications are very rare (0.5% per year, and these patients did OK with urgent management). Second, most patients will decide to have their hernias repaired, eventually (68% at 10 years). Finally, older patients are the ones most likely to come to repair.
Dr. Savarise |
What this means for the thousands of surgeons who see these patients on a regular basis is that shared clinical decision making with our patients, based on the surgeon's judgment of operative risks and benefits, is the correct clinical pathway. Nonoperative management, when in the opinion of the surgeon and in the patient to be in the patient's best interest, is safe. Immediate operation, even in patients with asymptomatic hernias, is standard of care. And it would be perfectly reasonable for any good-risk patient to schedule an elective hernia repair at his convenience.
Dr. Mark Savarise is an ACS Fellow and clinical assistant professor of surgery, University of Utah, Salt Lake City.
This report is finally a victory for surgeons who plead for common sense in the pursuit of evidence-based practice. Dr. Robert J. Fitzgibbons, recognized as one of surgery's foremost experts in hernia repair, presents long-term follow-up of patients with inguinal hernia who are treated expectantly. There are three very significant results. First, complications are very rare (0.5% per year, and these patients did OK with urgent management). Second, most patients will decide to have their hernias repaired, eventually (68% at 10 years). Finally, older patients are the ones most likely to come to repair.
Dr. Savarise |
What this means for the thousands of surgeons who see these patients on a regular basis is that shared clinical decision making with our patients, based on the surgeon's judgment of operative risks and benefits, is the correct clinical pathway. Nonoperative management, when in the opinion of the surgeon and in the patient to be in the patient's best interest, is safe. Immediate operation, even in patients with asymptomatic hernias, is standard of care. And it would be perfectly reasonable for any good-risk patient to schedule an elective hernia repair at his convenience.
Dr. Mark Savarise is an ACS Fellow and clinical assistant professor of surgery, University of Utah, Salt Lake City.
This report is finally a victory for surgeons who plead for common sense in the pursuit of evidence-based practice. Dr. Robert J. Fitzgibbons, recognized as one of surgery's foremost experts in hernia repair, presents long-term follow-up of patients with inguinal hernia who are treated expectantly. There are three very significant results. First, complications are very rare (0.5% per year, and these patients did OK with urgent management). Second, most patients will decide to have their hernias repaired, eventually (68% at 10 years). Finally, older patients are the ones most likely to come to repair.
Dr. Savarise |
What this means for the thousands of surgeons who see these patients on a regular basis is that shared clinical decision making with our patients, based on the surgeon's judgment of operative risks and benefits, is the correct clinical pathway. Nonoperative management, when in the opinion of the surgeon and in the patient to be in the patient's best interest, is safe. Immediate operation, even in patients with asymptomatic hernias, is standard of care. And it would be perfectly reasonable for any good-risk patient to schedule an elective hernia repair at his convenience.
Dr. Mark Savarise is an ACS Fellow and clinical assistant professor of surgery, University of Utah, Salt Lake City.
INDIANAPOLIS – The luster has suddenly worn off the time-honored strategy of nonoperative watchful waiting in men with minimally symptomatic inguinal hernias.
New evidence indicates the vast majority of men with asymptomatic or minimally symptomatic inguinal hernias will eventually come to surgery. This may not occur until years down the road, when their advanced age may render surgery more arduous.
"Although watchful waiting remains a safe strategy, even on long-term follow-up, patients who present to their physician to have their hernia evaluated, especially if elderly, should be informed that almost certainly they will come to surgery eventually ... The logical assumption is that watchful waiting is not an effective strategy, as with time almost all men cross over," Dr. Robert J. Fitzgibbons Jr. explained at the annual meeting of the American Surgical Association.
He presented an extended follow-up of patients enrolled in a landmark randomized multicenter clinical trial, one of only two randomized studies ever done comparing watchful waiting versus routine surgical repair for men with minimally symptomatic inguinal hernia. In the earlier report by Dr. Fitzgibbons and coworkers, watchful waiting was deemed "an acceptable option" because only 23% of patients crossed over to surgery due to increased pain during the first 2 years of follow-up (JAMA 2006;295:285-92).
At the ASA meeting, however, he presented updated 10-year follow-up data on 167 patients from the cohort initially assigned to watchful waiting. The rate of crossover to surgery was 68% by 10 years, with a marked age-based divergence. Patients below age 65 had a 62% crossover rate, while those above that age had a 79% crossover rate, according to Dr. Fitzgibbons, professor of surgery and chief of the division of general surgery at Creighton University, Omaha, Neb.
The good news was that hernia incarceration was a rare event, occurring at a rate of just 0.2% per year over the course of 10 years.
"We as surgeons have been taught for many years that we must repair all our hernias to prevent hernia accidents. Well, only three patients in our whole study developed incarceration, for which they underwent surgery with no mortality," Dr. Fitzgibbons noted. "The risk of a hernia accident should not be considered an indication for surgery. Older studies in the literature which would suggest otherwise can no longer be considered relevant."
He offered a caveat regarding the study findings: Participants were enrolled after they sought medical attention because of their hernias, even though they were asymptomatic or only minimally symptomatic. So the study results are most applicable to men concerned enough about their hernias that they visit a physician for that reason.
"It’s probably not valid to extrapolate the conclusions in this study to the entire population of minimally symptomatic inguinal hernia patients," the surgeon said. "Physicians have been observing elderly patients for years and would be loath to believe a crossover rate this high."
Nevertheless, the results of this study are virtually identical to those of the only other randomized trial of watchful waiting, which was conducted by surgeons at the University of Glasgow. The most recent update from that study showed an estimated crossover rate in the watchful waiting group of 16% at 1 year, 54% at 5 years, and 72% at 7.5 years. As in the American study, the rate of acute incarceration was reassuringly small. The investigators concluded that watchful waiting appears pointless, and they recommended surgical repair for medically fit patients (Br. J. Surg. 2011;98:596-9).
Discussant Dr. Michael E. Zenilman commented that his own approach is to individualize patient management based in large part upon activity level.
"When I see patients who are 80 years old in the office with an asymptomatic hernia, my conversation with them is about what their lifestyle is like. If they’re an active golfer I know that they’re going to end up getting their hernia fixed. If they’re sedentary, sitting at home in retirement, they don’t. So I think the next step in your research project should be to find out what the activity level is of these patients who are getting older and have asymptomatic hernias," said Dr. Zenilman, vice chair and regional director of surgery for the Washington, D.C., region, Johns Hopkins Medicine.
Dr. Fitzgibbons’ trial was funded by the Agency for Healthcare Research and Quality with support from the American College of Surgeons. He reported having no financial conflicts.
INDIANAPOLIS – The luster has suddenly worn off the time-honored strategy of nonoperative watchful waiting in men with minimally symptomatic inguinal hernias.
New evidence indicates the vast majority of men with asymptomatic or minimally symptomatic inguinal hernias will eventually come to surgery. This may not occur until years down the road, when their advanced age may render surgery more arduous.
"Although watchful waiting remains a safe strategy, even on long-term follow-up, patients who present to their physician to have their hernia evaluated, especially if elderly, should be informed that almost certainly they will come to surgery eventually ... The logical assumption is that watchful waiting is not an effective strategy, as with time almost all men cross over," Dr. Robert J. Fitzgibbons Jr. explained at the annual meeting of the American Surgical Association.
He presented an extended follow-up of patients enrolled in a landmark randomized multicenter clinical trial, one of only two randomized studies ever done comparing watchful waiting versus routine surgical repair for men with minimally symptomatic inguinal hernia. In the earlier report by Dr. Fitzgibbons and coworkers, watchful waiting was deemed "an acceptable option" because only 23% of patients crossed over to surgery due to increased pain during the first 2 years of follow-up (JAMA 2006;295:285-92).
At the ASA meeting, however, he presented updated 10-year follow-up data on 167 patients from the cohort initially assigned to watchful waiting. The rate of crossover to surgery was 68% by 10 years, with a marked age-based divergence. Patients below age 65 had a 62% crossover rate, while those above that age had a 79% crossover rate, according to Dr. Fitzgibbons, professor of surgery and chief of the division of general surgery at Creighton University, Omaha, Neb.
The good news was that hernia incarceration was a rare event, occurring at a rate of just 0.2% per year over the course of 10 years.
"We as surgeons have been taught for many years that we must repair all our hernias to prevent hernia accidents. Well, only three patients in our whole study developed incarceration, for which they underwent surgery with no mortality," Dr. Fitzgibbons noted. "The risk of a hernia accident should not be considered an indication for surgery. Older studies in the literature which would suggest otherwise can no longer be considered relevant."
He offered a caveat regarding the study findings: Participants were enrolled after they sought medical attention because of their hernias, even though they were asymptomatic or only minimally symptomatic. So the study results are most applicable to men concerned enough about their hernias that they visit a physician for that reason.
"It’s probably not valid to extrapolate the conclusions in this study to the entire population of minimally symptomatic inguinal hernia patients," the surgeon said. "Physicians have been observing elderly patients for years and would be loath to believe a crossover rate this high."
Nevertheless, the results of this study are virtually identical to those of the only other randomized trial of watchful waiting, which was conducted by surgeons at the University of Glasgow. The most recent update from that study showed an estimated crossover rate in the watchful waiting group of 16% at 1 year, 54% at 5 years, and 72% at 7.5 years. As in the American study, the rate of acute incarceration was reassuringly small. The investigators concluded that watchful waiting appears pointless, and they recommended surgical repair for medically fit patients (Br. J. Surg. 2011;98:596-9).
Discussant Dr. Michael E. Zenilman commented that his own approach is to individualize patient management based in large part upon activity level.
"When I see patients who are 80 years old in the office with an asymptomatic hernia, my conversation with them is about what their lifestyle is like. If they’re an active golfer I know that they’re going to end up getting their hernia fixed. If they’re sedentary, sitting at home in retirement, they don’t. So I think the next step in your research project should be to find out what the activity level is of these patients who are getting older and have asymptomatic hernias," said Dr. Zenilman, vice chair and regional director of surgery for the Washington, D.C., region, Johns Hopkins Medicine.
Dr. Fitzgibbons’ trial was funded by the Agency for Healthcare Research and Quality with support from the American College of Surgeons. He reported having no financial conflicts.
AT THE ASA ANNUAL MEETING
Major finding: Sixty-eight percent of men randomized to nonoperative observation of their asymptomatic or minimally symptomatic inguinal hernia crossed over to surgical repair within 10 years.
Data source: This was an open registry long-term extension of a randomized trial in which 720 men with minimally symptomatic inguinal hernia were assigned to watchful waiting or routine surgical repair.
Disclosures: The sponsor was the Agency for Healthcare Research and Quality. The presenter reported having no conflicts of interest.
Obesity's price tag calculated in surgical patients
INDIANAPOLIS – Obesity adds an estimated $164 million annually to the total hospital cost of major nonbariatric/nonobstetric operative procedures, Dr. Rodney J. Mason reported at the annual meeting of the American Surgical Association.
And that figure, audience members were swift to point out, is a very, very conservative estimate.
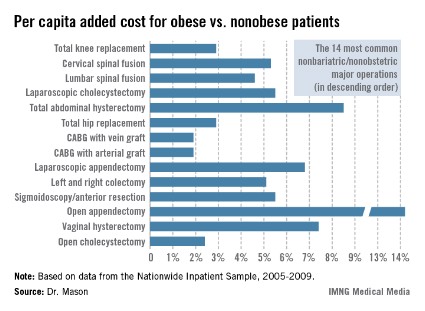
Dr. Mason presented an analysis of national cost estimates for obese vs. nonobese patients who underwent the 14 most common nonbariatric/nonobstetric major surgical procedures in the United States during 2005-2009 (see chart). He used data from the Nationwide Inpatient Sample (NIS) database sponsored by the Agency for Healthcare Research and Quality. The database provides detailed case information, including inpatient charges, from 20% of the nation’s hospitals.
One-quarter of the roughly 48 million hospitalized patients in the NIS database for this 5-year period underwent a major surgical procedure. Ten percent of patients who had 1 of the top 14 surgeries were obese. Dr. Mason and coworkers matched each of the 219,906 obese patients undergoing one of these operations to a nonobese patient who had the same operation; the tight one-to-one matching was based upon demographics and 28 defined comorbid conditions.
Because the NIS database records hospital charges, these charges had to be converted to estimated costs using a widely accepted formula. Once that was accomplished, the resultant annual total national estimated cost for the initial inpatient stay – not including physician costs – for patients undergoing one of the 14 surgical procedures was calculated at $4.38 billion in nonobese patients, compared with $4.55 billion in obese patients, for a $163,609,328 excess (3.7%) in the obese group, according to Dr. Mason of the University of Southern California, Los Angeles.
The mean per capita costs were $648 higher in obese than in nonobese surgical patients. To Dr. Mason’s surprise, this higher inpatient cost in the obese group was not driven by more postoperative complications; indeed, complication rates were equivalent in obese and nonobese patients. Instead, the explanation for the increased costs in the obese group was that they averaged a 0.025-day longer length of stay and they got significantly more secondary therapeutic, but not diagnostic, procedures.
Discussant Edward H. Livingston was sympathetic to the difficulties inherent in reliance upon the NIS database in conducting research, but he pointed out that the NIS is notoriously unreliable in recording obesity. This administrative database relies upon ICD-9 codes for obesity, and there’s good evidence that those codes often simply don’t get entered into the patient’s medical record. He noted that a mere 10% of the millions of surgical patients in the NIS database during the study years were categorized as obese, yet it’s well recognized that fully one-third of Americans are obese and another one-third are overweight. Clearly, then, a great many obese surgery patients were not captured in the study.
As for the study’s conclusion that the increased hospital costs in obese surgical patients were because of greater resource utilization and length of stay, Dr. Livingston said he thinks that’s probably all to the good.
"It may be that obese people do need more time in the hospital to recover and that they may need more resources to avoid more serious complications. We’ve learned how to better manage obese patients, and if those resources hadn’t been invested, then those patients might have done even worse," asserted Dr. Livingston, professor of surgery at the University of Texas Southwestern Medical Center, Dallas.
Taking care of obese patients is the reality of what physicians and surgeons deal with in hospitals now, he observed.
"Bariatric surgery has become the fifth most common operation in general surgery, and because of that we bariatric surgeons have taught everyone in the hospital how to take care of obese patients. We’ve learned over the years that there’s utility in using certain kinds of beds or having lifts available to move patients from one place to another. We’ve developed long instruments for facilitating laparoscopic surgery in obese patients. One of my own greatest learning experiences has been the utility of CPAP to manage morbidly obese patients in the postoperative period to help counter the effects of opiates and respiratory depression. The staff has become aware of how to take care of obese people and how to use medications appropriately in those people. All of that costs money," he said.
"Future research should aim to demonstrate that such costs are appropriate before Medicare does to us what it did with the so-called preventable complications, making a unilateral determination that we have too many complications or are spending too much money," the surgeon cautioned.
Dr. Mason responded that he’d like to see his study data shared with health policy makers in Washington.
"Hospitals get paid by [diagnosis-related group]. But if it costs more to manage obese patients, surely we should get more money to look after them. And right now we are not," he said.
Dr. Mason reported having no financial conflicts.
INDIANAPOLIS – Obesity adds an estimated $164 million annually to the total hospital cost of major nonbariatric/nonobstetric operative procedures, Dr. Rodney J. Mason reported at the annual meeting of the American Surgical Association.
And that figure, audience members were swift to point out, is a very, very conservative estimate.

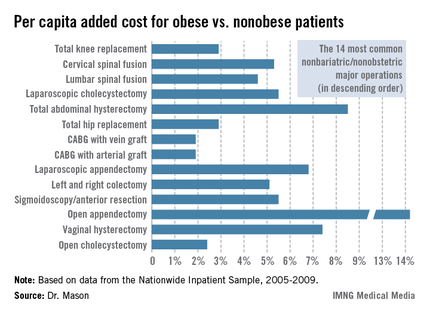
Dr. Mason presented an analysis of national cost estimates for obese vs. nonobese patients who underwent the 14 most common nonbariatric/nonobstetric major surgical procedures in the United States during 2005-2009 (see chart). He used data from the Nationwide Inpatient Sample (NIS) database sponsored by the Agency for Healthcare Research and Quality. The database provides detailed case information, including inpatient charges, from 20% of the nation’s hospitals.
One-quarter of the roughly 48 million hospitalized patients in the NIS database for this 5-year period underwent a major surgical procedure. Ten percent of patients who had 1 of the top 14 surgeries were obese. Dr. Mason and coworkers matched each of the 219,906 obese patients undergoing one of these operations to a nonobese patient who had the same operation; the tight one-to-one matching was based upon demographics and 28 defined comorbid conditions.
Because the NIS database records hospital charges, these charges had to be converted to estimated costs using a widely accepted formula. Once that was accomplished, the resultant annual total national estimated cost for the initial inpatient stay – not including physician costs – for patients undergoing one of the 14 surgical procedures was calculated at $4.38 billion in nonobese patients, compared with $4.55 billion in obese patients, for a $163,609,328 excess (3.7%) in the obese group, according to Dr. Mason of the University of Southern California, Los Angeles.
The mean per capita costs were $648 higher in obese than in nonobese surgical patients. To Dr. Mason’s surprise, this higher inpatient cost in the obese group was not driven by more postoperative complications; indeed, complication rates were equivalent in obese and nonobese patients. Instead, the explanation for the increased costs in the obese group was that they averaged a 0.025-day longer length of stay and they got significantly more secondary therapeutic, but not diagnostic, procedures.
Discussant Edward H. Livingston was sympathetic to the difficulties inherent in reliance upon the NIS database in conducting research, but he pointed out that the NIS is notoriously unreliable in recording obesity. This administrative database relies upon ICD-9 codes for obesity, and there’s good evidence that those codes often simply don’t get entered into the patient’s medical record. He noted that a mere 10% of the millions of surgical patients in the NIS database during the study years were categorized as obese, yet it’s well recognized that fully one-third of Americans are obese and another one-third are overweight. Clearly, then, a great many obese surgery patients were not captured in the study.
As for the study’s conclusion that the increased hospital costs in obese surgical patients were because of greater resource utilization and length of stay, Dr. Livingston said he thinks that’s probably all to the good.
"It may be that obese people do need more time in the hospital to recover and that they may need more resources to avoid more serious complications. We’ve learned how to better manage obese patients, and if those resources hadn’t been invested, then those patients might have done even worse," asserted Dr. Livingston, professor of surgery at the University of Texas Southwestern Medical Center, Dallas.
Taking care of obese patients is the reality of what physicians and surgeons deal with in hospitals now, he observed.
"Bariatric surgery has become the fifth most common operation in general surgery, and because of that we bariatric surgeons have taught everyone in the hospital how to take care of obese patients. We’ve learned over the years that there’s utility in using certain kinds of beds or having lifts available to move patients from one place to another. We’ve developed long instruments for facilitating laparoscopic surgery in obese patients. One of my own greatest learning experiences has been the utility of CPAP to manage morbidly obese patients in the postoperative period to help counter the effects of opiates and respiratory depression. The staff has become aware of how to take care of obese people and how to use medications appropriately in those people. All of that costs money," he said.
"Future research should aim to demonstrate that such costs are appropriate before Medicare does to us what it did with the so-called preventable complications, making a unilateral determination that we have too many complications or are spending too much money," the surgeon cautioned.
Dr. Mason responded that he’d like to see his study data shared with health policy makers in Washington.
"Hospitals get paid by [diagnosis-related group]. But if it costs more to manage obese patients, surely we should get more money to look after them. And right now we are not," he said.
Dr. Mason reported having no financial conflicts.
INDIANAPOLIS – Obesity adds an estimated $164 million annually to the total hospital cost of major nonbariatric/nonobstetric operative procedures, Dr. Rodney J. Mason reported at the annual meeting of the American Surgical Association.
And that figure, audience members were swift to point out, is a very, very conservative estimate.

Dr. Mason presented an analysis of national cost estimates for obese vs. nonobese patients who underwent the 14 most common nonbariatric/nonobstetric major surgical procedures in the United States during 2005-2009 (see chart). He used data from the Nationwide Inpatient Sample (NIS) database sponsored by the Agency for Healthcare Research and Quality. The database provides detailed case information, including inpatient charges, from 20% of the nation’s hospitals.
One-quarter of the roughly 48 million hospitalized patients in the NIS database for this 5-year period underwent a major surgical procedure. Ten percent of patients who had 1 of the top 14 surgeries were obese. Dr. Mason and coworkers matched each of the 219,906 obese patients undergoing one of these operations to a nonobese patient who had the same operation; the tight one-to-one matching was based upon demographics and 28 defined comorbid conditions.
Because the NIS database records hospital charges, these charges had to be converted to estimated costs using a widely accepted formula. Once that was accomplished, the resultant annual total national estimated cost for the initial inpatient stay – not including physician costs – for patients undergoing one of the 14 surgical procedures was calculated at $4.38 billion in nonobese patients, compared with $4.55 billion in obese patients, for a $163,609,328 excess (3.7%) in the obese group, according to Dr. Mason of the University of Southern California, Los Angeles.
The mean per capita costs were $648 higher in obese than in nonobese surgical patients. To Dr. Mason’s surprise, this higher inpatient cost in the obese group was not driven by more postoperative complications; indeed, complication rates were equivalent in obese and nonobese patients. Instead, the explanation for the increased costs in the obese group was that they averaged a 0.025-day longer length of stay and they got significantly more secondary therapeutic, but not diagnostic, procedures.
Discussant Edward H. Livingston was sympathetic to the difficulties inherent in reliance upon the NIS database in conducting research, but he pointed out that the NIS is notoriously unreliable in recording obesity. This administrative database relies upon ICD-9 codes for obesity, and there’s good evidence that those codes often simply don’t get entered into the patient’s medical record. He noted that a mere 10% of the millions of surgical patients in the NIS database during the study years were categorized as obese, yet it’s well recognized that fully one-third of Americans are obese and another one-third are overweight. Clearly, then, a great many obese surgery patients were not captured in the study.
As for the study’s conclusion that the increased hospital costs in obese surgical patients were because of greater resource utilization and length of stay, Dr. Livingston said he thinks that’s probably all to the good.
"It may be that obese people do need more time in the hospital to recover and that they may need more resources to avoid more serious complications. We’ve learned how to better manage obese patients, and if those resources hadn’t been invested, then those patients might have done even worse," asserted Dr. Livingston, professor of surgery at the University of Texas Southwestern Medical Center, Dallas.
Taking care of obese patients is the reality of what physicians and surgeons deal with in hospitals now, he observed.
"Bariatric surgery has become the fifth most common operation in general surgery, and because of that we bariatric surgeons have taught everyone in the hospital how to take care of obese patients. We’ve learned over the years that there’s utility in using certain kinds of beds or having lifts available to move patients from one place to another. We’ve developed long instruments for facilitating laparoscopic surgery in obese patients. One of my own greatest learning experiences has been the utility of CPAP to manage morbidly obese patients in the postoperative period to help counter the effects of opiates and respiratory depression. The staff has become aware of how to take care of obese people and how to use medications appropriately in those people. All of that costs money," he said.
"Future research should aim to demonstrate that such costs are appropriate before Medicare does to us what it did with the so-called preventable complications, making a unilateral determination that we have too many complications or are spending too much money," the surgeon cautioned.
Dr. Mason responded that he’d like to see his study data shared with health policy makers in Washington.
"Hospitals get paid by [diagnosis-related group]. But if it costs more to manage obese patients, surely we should get more money to look after them. And right now we are not," he said.
Dr. Mason reported having no financial conflicts.
AT THE ASA ANNUAL MEETING
Major Finding: Obese patients undergoing 1 of the 14 most common nonbariatric/nonobstetric major surgical procedures had a mean average total hospital cost excluding physician fees that was $648 higher than for tightly matched nonobese patients undergoing the same operation. That added up to a national cost of roughly $164 million/year annually
Data Source: A matched pair analysis of data on 219,906 and an equal number of nonobese patients obtained from the Nationwide Inpatient Sample.
Disclosures: The Nationwide Inpatient Sample is sponsored by the Agency for Healthcare Research and Quality. The study presenter reported having no conflicts of interest.
Early cholecystectomy beats delayed in acute cholecystitis
INDIANAPOLIS – Acute cholecystitis patients fared significantly better with early rather than delayed laparoscopic cholecystectomy in the largest-ever randomized trial addressing surgical timing for this common condition.
Patients assigned to early cholecystectomy – that is, surgery within 24 hours of presentation to the hospital – had one-third the morbidity, markedly shorter hospital lengths of stay, and correspondingly lower hospital costs compared with patients who underwent surgery on day 7-45, according to Dr. Markus W. Buchler of Heidelberg (Ger.) University.
"Early cholecystectomy in patients fit for surgery and in hospitals experienced in doing difficult laparoscopic cholecystectomies should become the standard of care in acute cholecystitis," he declared in presenting the results of the ACDC (Acute Cholecystitis: Early Versus Delayed Cholecystectomy) trial at the annual meeting of the American Surgical Association.
The optimal timing of surgical intervention in acute cholecystitis is a subject of long-standing controversy. The ACDC trial was conducted because in a Cochrane review of five smaller randomized trials totaling 451 acute cholecystitis patients, researchers concluded there was insufficient evidence to say which surgical strategy was best (Cochrane Database Syst. Rev. 2006 Oct 18;4:CD005440).
Dr. Buchler noted that surveys indicate many American surgeons prefer to delay laparoscopic cholecystectomy, while in Germany the surgical preference is for immediate surgery in patients with uncomplicated acute cholecystitis.
The ACDC trial involved 618 patients with uncomplicated acute cholecystitis who were placed on the same antibiotic – moxifloxacin – and randomized to early laparoscopic cholecystectomy or to delayed surgery on day 7-45. Pregnant patients were excluded from the trial, which was conducted at 35 European hospitals, including seven German university medical centers. All participating hospitals were staffed by surgical teams experienced in performing difficult laparoscopic cholecystectomies.
The primary endpoint was total morbidity within 75 days. This included cholangitis, pancreatitis, biliary leak, stroke, myocardial infarction, abscess, bleeding, peritonitis, infection, and renal failure. The rate was 11.6% in the early cholecystectomy group compared with 31.3% with delayed surgery. Among less challenging patients with an ASA score of 2 or less, the rates were 9.7% and 28.6%, respectively. Patients with an ASA score above 2 had an overall morbidity rate of 20% with early surgery compared with 47% with delayed laparoscopic cholecystectomy.
The rate of conversion to open surgery was 9.9% in the early laparoscopic cholecystectomy group and similar at 11.9% in the delayed surgery group. This came as a surprise to Dr. Buchler and his coinvestigators. They expected a significantly higher conversion rate in conjunction with delayed laparoscopic cholecystectomy.
"I think what this tells us is surgeons have gotten really good at laparoscopic cholecystectomy even in more difficult situations," he observed.
Total hospital stays averaged 5.4 days in the early surgery group compared with 10.0 days with delayed surgery. Mean total hospital costs calculated via the German DRG system were 2,919 euro in the early cholecystectomy group and 4,261 euro with delayed surgery.
Discussant Dr. Andrew L. Warshaw praised Dr. Buchler and his coworkers in the German surgical clinical trials study group for their "leadership in determining evidence-based standards of care."
"There’s no doubt in my mind that immediate cholecystectomy is superior in this patient population," said Dr. Warshaw, professor and chairman of the department of surgery at Harvard Medical School, Boston.
Noting that most acute cholecystitis patients are first seen by an internist or gastroenterologist who then makes the initial treatment decision, Dr. Warshaw asked Dr. Buchler if German internists and gastroenterologists have gotten on board this immediate surgery treatment pathway.
"Convincing internists and gastroenterologists will take a long time, at least in Germany," the surgeon replied. "It is much easier to convince the emergency department physicians to refer patients early to surgery; they’re much closer to the surgeons."
He noted that German surgical practice differs from that in the United States in several respects. For one, German patients routinely stay in the hospital longer, even if they don’t experience complications. That’s why the mean length of stay after cholecystectomy in ACDC was 4.68 days in the early surgery group and closely similar at 4.89 days in the delayed surgery group, even though the delayed surgery group had a threefold higher complication rate.
Another difference is that, unlike in this country, intraoperative cholangiography is rarely done in Germany.
"It’s the absolute exception that intraoperative cholangiography is used. It is used only when there’s a reason for it, such as jaundice. There was probably less than a 3% intraoperative cholangiography rate in this trial," said Dr. Buchler.
The ACDC trial was funded with government research grants. Dr. Buchler reported having no financial conflicts.
INDIANAPOLIS – Acute cholecystitis patients fared significantly better with early rather than delayed laparoscopic cholecystectomy in the largest-ever randomized trial addressing surgical timing for this common condition.
Patients assigned to early cholecystectomy – that is, surgery within 24 hours of presentation to the hospital – had one-third the morbidity, markedly shorter hospital lengths of stay, and correspondingly lower hospital costs compared with patients who underwent surgery on day 7-45, according to Dr. Markus W. Buchler of Heidelberg (Ger.) University.
"Early cholecystectomy in patients fit for surgery and in hospitals experienced in doing difficult laparoscopic cholecystectomies should become the standard of care in acute cholecystitis," he declared in presenting the results of the ACDC (Acute Cholecystitis: Early Versus Delayed Cholecystectomy) trial at the annual meeting of the American Surgical Association.
The optimal timing of surgical intervention in acute cholecystitis is a subject of long-standing controversy. The ACDC trial was conducted because in a Cochrane review of five smaller randomized trials totaling 451 acute cholecystitis patients, researchers concluded there was insufficient evidence to say which surgical strategy was best (Cochrane Database Syst. Rev. 2006 Oct 18;4:CD005440).
Dr. Buchler noted that surveys indicate many American surgeons prefer to delay laparoscopic cholecystectomy, while in Germany the surgical preference is for immediate surgery in patients with uncomplicated acute cholecystitis.
The ACDC trial involved 618 patients with uncomplicated acute cholecystitis who were placed on the same antibiotic – moxifloxacin – and randomized to early laparoscopic cholecystectomy or to delayed surgery on day 7-45. Pregnant patients were excluded from the trial, which was conducted at 35 European hospitals, including seven German university medical centers. All participating hospitals were staffed by surgical teams experienced in performing difficult laparoscopic cholecystectomies.
The primary endpoint was total morbidity within 75 days. This included cholangitis, pancreatitis, biliary leak, stroke, myocardial infarction, abscess, bleeding, peritonitis, infection, and renal failure. The rate was 11.6% in the early cholecystectomy group compared with 31.3% with delayed surgery. Among less challenging patients with an ASA score of 2 or less, the rates were 9.7% and 28.6%, respectively. Patients with an ASA score above 2 had an overall morbidity rate of 20% with early surgery compared with 47% with delayed laparoscopic cholecystectomy.
The rate of conversion to open surgery was 9.9% in the early laparoscopic cholecystectomy group and similar at 11.9% in the delayed surgery group. This came as a surprise to Dr. Buchler and his coinvestigators. They expected a significantly higher conversion rate in conjunction with delayed laparoscopic cholecystectomy.
"I think what this tells us is surgeons have gotten really good at laparoscopic cholecystectomy even in more difficult situations," he observed.
Total hospital stays averaged 5.4 days in the early surgery group compared with 10.0 days with delayed surgery. Mean total hospital costs calculated via the German DRG system were 2,919 euro in the early cholecystectomy group and 4,261 euro with delayed surgery.
Discussant Dr. Andrew L. Warshaw praised Dr. Buchler and his coworkers in the German surgical clinical trials study group for their "leadership in determining evidence-based standards of care."
"There’s no doubt in my mind that immediate cholecystectomy is superior in this patient population," said Dr. Warshaw, professor and chairman of the department of surgery at Harvard Medical School, Boston.
Noting that most acute cholecystitis patients are first seen by an internist or gastroenterologist who then makes the initial treatment decision, Dr. Warshaw asked Dr. Buchler if German internists and gastroenterologists have gotten on board this immediate surgery treatment pathway.
"Convincing internists and gastroenterologists will take a long time, at least in Germany," the surgeon replied. "It is much easier to convince the emergency department physicians to refer patients early to surgery; they’re much closer to the surgeons."
He noted that German surgical practice differs from that in the United States in several respects. For one, German patients routinely stay in the hospital longer, even if they don’t experience complications. That’s why the mean length of stay after cholecystectomy in ACDC was 4.68 days in the early surgery group and closely similar at 4.89 days in the delayed surgery group, even though the delayed surgery group had a threefold higher complication rate.
Another difference is that, unlike in this country, intraoperative cholangiography is rarely done in Germany.
"It’s the absolute exception that intraoperative cholangiography is used. It is used only when there’s a reason for it, such as jaundice. There was probably less than a 3% intraoperative cholangiography rate in this trial," said Dr. Buchler.
The ACDC trial was funded with government research grants. Dr. Buchler reported having no financial conflicts.
INDIANAPOLIS – Acute cholecystitis patients fared significantly better with early rather than delayed laparoscopic cholecystectomy in the largest-ever randomized trial addressing surgical timing for this common condition.
Patients assigned to early cholecystectomy – that is, surgery within 24 hours of presentation to the hospital – had one-third the morbidity, markedly shorter hospital lengths of stay, and correspondingly lower hospital costs compared with patients who underwent surgery on day 7-45, according to Dr. Markus W. Buchler of Heidelberg (Ger.) University.
"Early cholecystectomy in patients fit for surgery and in hospitals experienced in doing difficult laparoscopic cholecystectomies should become the standard of care in acute cholecystitis," he declared in presenting the results of the ACDC (Acute Cholecystitis: Early Versus Delayed Cholecystectomy) trial at the annual meeting of the American Surgical Association.
The optimal timing of surgical intervention in acute cholecystitis is a subject of long-standing controversy. The ACDC trial was conducted because in a Cochrane review of five smaller randomized trials totaling 451 acute cholecystitis patients, researchers concluded there was insufficient evidence to say which surgical strategy was best (Cochrane Database Syst. Rev. 2006 Oct 18;4:CD005440).
Dr. Buchler noted that surveys indicate many American surgeons prefer to delay laparoscopic cholecystectomy, while in Germany the surgical preference is for immediate surgery in patients with uncomplicated acute cholecystitis.
The ACDC trial involved 618 patients with uncomplicated acute cholecystitis who were placed on the same antibiotic – moxifloxacin – and randomized to early laparoscopic cholecystectomy or to delayed surgery on day 7-45. Pregnant patients were excluded from the trial, which was conducted at 35 European hospitals, including seven German university medical centers. All participating hospitals were staffed by surgical teams experienced in performing difficult laparoscopic cholecystectomies.
The primary endpoint was total morbidity within 75 days. This included cholangitis, pancreatitis, biliary leak, stroke, myocardial infarction, abscess, bleeding, peritonitis, infection, and renal failure. The rate was 11.6% in the early cholecystectomy group compared with 31.3% with delayed surgery. Among less challenging patients with an ASA score of 2 or less, the rates were 9.7% and 28.6%, respectively. Patients with an ASA score above 2 had an overall morbidity rate of 20% with early surgery compared with 47% with delayed laparoscopic cholecystectomy.
The rate of conversion to open surgery was 9.9% in the early laparoscopic cholecystectomy group and similar at 11.9% in the delayed surgery group. This came as a surprise to Dr. Buchler and his coinvestigators. They expected a significantly higher conversion rate in conjunction with delayed laparoscopic cholecystectomy.
"I think what this tells us is surgeons have gotten really good at laparoscopic cholecystectomy even in more difficult situations," he observed.
Total hospital stays averaged 5.4 days in the early surgery group compared with 10.0 days with delayed surgery. Mean total hospital costs calculated via the German DRG system were 2,919 euro in the early cholecystectomy group and 4,261 euro with delayed surgery.
Discussant Dr. Andrew L. Warshaw praised Dr. Buchler and his coworkers in the German surgical clinical trials study group for their "leadership in determining evidence-based standards of care."
"There’s no doubt in my mind that immediate cholecystectomy is superior in this patient population," said Dr. Warshaw, professor and chairman of the department of surgery at Harvard Medical School, Boston.
Noting that most acute cholecystitis patients are first seen by an internist or gastroenterologist who then makes the initial treatment decision, Dr. Warshaw asked Dr. Buchler if German internists and gastroenterologists have gotten on board this immediate surgery treatment pathway.
"Convincing internists and gastroenterologists will take a long time, at least in Germany," the surgeon replied. "It is much easier to convince the emergency department physicians to refer patients early to surgery; they’re much closer to the surgeons."
He noted that German surgical practice differs from that in the United States in several respects. For one, German patients routinely stay in the hospital longer, even if they don’t experience complications. That’s why the mean length of stay after cholecystectomy in ACDC was 4.68 days in the early surgery group and closely similar at 4.89 days in the delayed surgery group, even though the delayed surgery group had a threefold higher complication rate.
Another difference is that, unlike in this country, intraoperative cholangiography is rarely done in Germany.
"It’s the absolute exception that intraoperative cholangiography is used. It is used only when there’s a reason for it, such as jaundice. There was probably less than a 3% intraoperative cholangiography rate in this trial," said Dr. Buchler.
The ACDC trial was funded with government research grants. Dr. Buchler reported having no financial conflicts.
AT THE ASA ANNUAL MEETING
Major Finding: Acute cholecystitis patients randomized to laparoscopic cholecystectomy within 24 hours of hospital arrival had a 75-day total morbidity rate of 11.6% compared with 31.3% for those randomized to delayed laparoscopic cholecystectomy on days 7-45. Mean total hospital costs were one-third less in the early cholecystectomy group.
Data Source: ACDC trial of 618 randomized patients at 35 European medical centers.
Disclosures: The trial was funded by a German governmental research grant. The presenter reported having no conflict of interest.
Mortality after surgery for radiation-related cardiac problems unexpectedly high
Mortality is twice as high for cancer survivors undergoing cardiac surgery to correct damage done by thoracic radiation therapy as it is among matched patients who are undergoing the same procedures but who haven’t been exposed to radiotherapy, according to a report published online April 8 in Circulation.
In what they described as "the largest study to assess long-term survival in this population undergoing complex cardiac surgery and compare them to a well-matched comparison population," investigators found that mortality was 55% in the patients who had received radiation therapy, compared with 28% in the nonexposed controls. At least half of the deaths among the cancer survivors were attributed to cardiopulmonary disease, while only 5% were due to recurrent malignancy.
"Surgical intervention should be applied cautiously to patients who have had significant thoracic radiation previously," including survivors of breast, lung, hematologic, thyroid, and other cancers.
Trying to gauge mortality risk in such patients solely by using their preoperative scores on standard risk-prediction measures is inaccurate at best. In this study population, the survivors of breast, lung, hematologic, thyroid, and other cancers would have been deemed at intermediate risk for undergoing cardiac surgery, with an expected mortality of only 3%-5% based on their preoperative assessments – 10 times lower than their actual postoperative mortality, said Dr. Willis Wu and his associates at the Heart and Vascular Institute at the Cleveland Clinic.
"Our findings suggest that surgical intervention should be applied cautiously to patients who have had significant thoracic radiation previously," they wrote. Cardiac surgery that is considered conventional or even routine in most patients – including coronary artery bypass graft, valve repair, valve replacement, vascular surgery, pericardiectomy, left ventricular assist device implantation, and myectomy – may actually worsen the condition of radiation-exposed patients.
"It seems appropriate to examine other types of treatment options in patients with radiation heart disease who have significant valvular, coronary, or myocardial disease," the researchers noted.
Dr. Wu and his associates performed their retrospective observational study because of the dearth of information regarding long-term outcomes in cancer survivors who were exposed to chest irradiation. This knowledge gap makes treatment decisions "especially difficult" once radiation-related heart disease is discovered.
Such patients are known to have high rates of coronary artery, valvular, pericardial, myocardial, and conduction disease that progresses over time and may not become evident until decades after cancer treatment was completed.
Surgery is often thought necessary to effectively address these cardiac complications of prior radiation therapy. "These patients have multiple cardiac lesions and [often] have comorbidities such as pulmonary or vascular disease related to radiation," the investigators said.
They analyzed the medical records of 478 patients undergoing cardiac surgery at their tertiary care center in 2000-2003. A total of 173 had a prior history of malignancy treated with chest irradiation; the remaining 305 patients had no such history and were matched to the radiation group for age, sex, type of surgery, and date of surgery.
The patients exposed to radiation were relatively young but had advanced symptoms. Forty-five percent had obstructive proximal coronary artery disease; one-fourth had already undergone open heart surgery, including 16% who had prior coronary artery bypass graft surgery. There also was a high prevalence of valvular disease, with 51% of patients showing at least moderate mitral regurgitation, 6% showing severe mitral stenosis, 29% showing at least moderate aortic regurgitation, 23% showing severe aortic stenosis, and 34% showing at least moderate tricuspid regurgitation.
The mean interval between the current cardiac surgery and the last thoracic radiation treatment was 18 years. Approximately 53% of the study patients had survived breast cancer, 27% had survived Hodgkin’s lymphoma, 7% had survived lung cancer, 6% had survived non-Hodgkin’s lymphoma, and 8% had survived other malignancies including thyroid and testicular cancers.
Approximately 75% of both study groups were women.
During a mean follow-up of 7.6 years, overall mortality was 37%. Despite the success of the cardiac procedures, a significantly higher proportion of radiation-exposed patients died (55%), compared with the unexposed patients (28%), for a hazard ratio of 2.54, Dr. Wu and his colleagues said (Circulation 2013 April 8; doi:10.1161/CIRCULATIONAHA.13.001435).
Mortality was significantly higher across all subgroups of patients exposed to radiation. In particular, it was high among patients expected to have low mortality: 43% in patients aged younger than 65 years and 45% in those with lower preoperative risk scores. In fact, the youngest patients in the radiation group fared worse than the oldest patients in the comparison group.
Mortality also was significantly higher for the radiation-exposed patients than for controls across all types of cardiac surgery.
The cause of death was not known for a substantial proportion of subjects in this study, but cardiopulmonary causes were implicated in at least half of the deaths among radiation-exposed patients. "Why this should be the case in a group of patients who received potentially life-preserving revascularization, valve, and other procedures is unknown at present," Dr. Wu and his associates said.
In addition, patients with radiation-related heart disease had longer hospital stays, were more likely to develop atrial fibrillation after surgery, were more likely to require pacemaker implantation after surgery, had higher rates of ventricular dysfunction and of valvular regurgitation following surgery, and had higher 30-day mortality.
The finding that radiation-exposed patients "remained in hospital for a considerable period after their surgery" suggests that the procedures and postoperative courses were complicated.
"Radiation patients frequently develop pulmonary complications as a result of open heart surgery, not the least of which are recurrent pleural effusions and severe restrictive lung disease. It is our experience that respiratory complications may significantly compromise function and survival in patients with extensive prior radiation," the researchers said.
"Additionally, the presence of myocardial disease either as a result of the underlying cardiac condition (potentially exacerbated by prior chemotherapy) or as a consequence of a restrictive-type cardiomyopathy produced by the effects of radiation may play a role in impaired survival and is not necessarily improved by valvular or revascularization surgery," they said.
The radiation-exposed patients’ poor long-term survival after conventional cardiac surgery indicates that other, nonsurgical treatments may be necessary for this complex patient population, they added.
No relevant financial conflicts were reported.
There is little doubt that prior radiotherapy complicates surgery. Specifically, standard radiation used with curative intent for breast cancer, lymphoma, and a few other mediastinal/thorax malignancies, has important long-term effects.
Dr. Murthy |
These authors report on the increased complications and mortality of open heart operation following chest radiation given on average, some 20 years before. What is left out is that it is the radiotherapy itself that may be responsible for the current pathology for which many of the patients are requiring their heart operations. So you have the classic "insult-to-injury" of radiation being both the cause as well as the impediment to cure, in many of these circumstances. The caveat, of course, is that without their index radiotherapy, these patients would not have survived to now be burdened by the latent effects of ionizing radiation on the thorax.
Sudish Murthy, MD, FACS, is surgical director, Center of Major Airway Disease, The Cleveland Clinic.
There is little doubt that prior radiotherapy complicates surgery. Specifically, standard radiation used with curative intent for breast cancer, lymphoma, and a few other mediastinal/thorax malignancies, has important long-term effects.
Dr. Murthy |
These authors report on the increased complications and mortality of open heart operation following chest radiation given on average, some 20 years before. What is left out is that it is the radiotherapy itself that may be responsible for the current pathology for which many of the patients are requiring their heart operations. So you have the classic "insult-to-injury" of radiation being both the cause as well as the impediment to cure, in many of these circumstances. The caveat, of course, is that without their index radiotherapy, these patients would not have survived to now be burdened by the latent effects of ionizing radiation on the thorax.
Sudish Murthy, MD, FACS, is surgical director, Center of Major Airway Disease, The Cleveland Clinic.
There is little doubt that prior radiotherapy complicates surgery. Specifically, standard radiation used with curative intent for breast cancer, lymphoma, and a few other mediastinal/thorax malignancies, has important long-term effects.
Dr. Murthy |
These authors report on the increased complications and mortality of open heart operation following chest radiation given on average, some 20 years before. What is left out is that it is the radiotherapy itself that may be responsible for the current pathology for which many of the patients are requiring their heart operations. So you have the classic "insult-to-injury" of radiation being both the cause as well as the impediment to cure, in many of these circumstances. The caveat, of course, is that without their index radiotherapy, these patients would not have survived to now be burdened by the latent effects of ionizing radiation on the thorax.
Sudish Murthy, MD, FACS, is surgical director, Center of Major Airway Disease, The Cleveland Clinic.
Mortality is twice as high for cancer survivors undergoing cardiac surgery to correct damage done by thoracic radiation therapy as it is among matched patients who are undergoing the same procedures but who haven’t been exposed to radiotherapy, according to a report published online April 8 in Circulation.
In what they described as "the largest study to assess long-term survival in this population undergoing complex cardiac surgery and compare them to a well-matched comparison population," investigators found that mortality was 55% in the patients who had received radiation therapy, compared with 28% in the nonexposed controls. At least half of the deaths among the cancer survivors were attributed to cardiopulmonary disease, while only 5% were due to recurrent malignancy.
"Surgical intervention should be applied cautiously to patients who have had significant thoracic radiation previously," including survivors of breast, lung, hematologic, thyroid, and other cancers.
Trying to gauge mortality risk in such patients solely by using their preoperative scores on standard risk-prediction measures is inaccurate at best. In this study population, the survivors of breast, lung, hematologic, thyroid, and other cancers would have been deemed at intermediate risk for undergoing cardiac surgery, with an expected mortality of only 3%-5% based on their preoperative assessments – 10 times lower than their actual postoperative mortality, said Dr. Willis Wu and his associates at the Heart and Vascular Institute at the Cleveland Clinic.
"Our findings suggest that surgical intervention should be applied cautiously to patients who have had significant thoracic radiation previously," they wrote. Cardiac surgery that is considered conventional or even routine in most patients – including coronary artery bypass graft, valve repair, valve replacement, vascular surgery, pericardiectomy, left ventricular assist device implantation, and myectomy – may actually worsen the condition of radiation-exposed patients.
"It seems appropriate to examine other types of treatment options in patients with radiation heart disease who have significant valvular, coronary, or myocardial disease," the researchers noted.
Dr. Wu and his associates performed their retrospective observational study because of the dearth of information regarding long-term outcomes in cancer survivors who were exposed to chest irradiation. This knowledge gap makes treatment decisions "especially difficult" once radiation-related heart disease is discovered.
Such patients are known to have high rates of coronary artery, valvular, pericardial, myocardial, and conduction disease that progresses over time and may not become evident until decades after cancer treatment was completed.
Surgery is often thought necessary to effectively address these cardiac complications of prior radiation therapy. "These patients have multiple cardiac lesions and [often] have comorbidities such as pulmonary or vascular disease related to radiation," the investigators said.
They analyzed the medical records of 478 patients undergoing cardiac surgery at their tertiary care center in 2000-2003. A total of 173 had a prior history of malignancy treated with chest irradiation; the remaining 305 patients had no such history and were matched to the radiation group for age, sex, type of surgery, and date of surgery.
The patients exposed to radiation were relatively young but had advanced symptoms. Forty-five percent had obstructive proximal coronary artery disease; one-fourth had already undergone open heart surgery, including 16% who had prior coronary artery bypass graft surgery. There also was a high prevalence of valvular disease, with 51% of patients showing at least moderate mitral regurgitation, 6% showing severe mitral stenosis, 29% showing at least moderate aortic regurgitation, 23% showing severe aortic stenosis, and 34% showing at least moderate tricuspid regurgitation.
The mean interval between the current cardiac surgery and the last thoracic radiation treatment was 18 years. Approximately 53% of the study patients had survived breast cancer, 27% had survived Hodgkin’s lymphoma, 7% had survived lung cancer, 6% had survived non-Hodgkin’s lymphoma, and 8% had survived other malignancies including thyroid and testicular cancers.
Approximately 75% of both study groups were women.
During a mean follow-up of 7.6 years, overall mortality was 37%. Despite the success of the cardiac procedures, a significantly higher proportion of radiation-exposed patients died (55%), compared with the unexposed patients (28%), for a hazard ratio of 2.54, Dr. Wu and his colleagues said (Circulation 2013 April 8; doi:10.1161/CIRCULATIONAHA.13.001435).
Mortality was significantly higher across all subgroups of patients exposed to radiation. In particular, it was high among patients expected to have low mortality: 43% in patients aged younger than 65 years and 45% in those with lower preoperative risk scores. In fact, the youngest patients in the radiation group fared worse than the oldest patients in the comparison group.
Mortality also was significantly higher for the radiation-exposed patients than for controls across all types of cardiac surgery.
The cause of death was not known for a substantial proportion of subjects in this study, but cardiopulmonary causes were implicated in at least half of the deaths among radiation-exposed patients. "Why this should be the case in a group of patients who received potentially life-preserving revascularization, valve, and other procedures is unknown at present," Dr. Wu and his associates said.
In addition, patients with radiation-related heart disease had longer hospital stays, were more likely to develop atrial fibrillation after surgery, were more likely to require pacemaker implantation after surgery, had higher rates of ventricular dysfunction and of valvular regurgitation following surgery, and had higher 30-day mortality.
The finding that radiation-exposed patients "remained in hospital for a considerable period after their surgery" suggests that the procedures and postoperative courses were complicated.
"Radiation patients frequently develop pulmonary complications as a result of open heart surgery, not the least of which are recurrent pleural effusions and severe restrictive lung disease. It is our experience that respiratory complications may significantly compromise function and survival in patients with extensive prior radiation," the researchers said.
"Additionally, the presence of myocardial disease either as a result of the underlying cardiac condition (potentially exacerbated by prior chemotherapy) or as a consequence of a restrictive-type cardiomyopathy produced by the effects of radiation may play a role in impaired survival and is not necessarily improved by valvular or revascularization surgery," they said.
The radiation-exposed patients’ poor long-term survival after conventional cardiac surgery indicates that other, nonsurgical treatments may be necessary for this complex patient population, they added.
No relevant financial conflicts were reported.
Mortality is twice as high for cancer survivors undergoing cardiac surgery to correct damage done by thoracic radiation therapy as it is among matched patients who are undergoing the same procedures but who haven’t been exposed to radiotherapy, according to a report published online April 8 in Circulation.
In what they described as "the largest study to assess long-term survival in this population undergoing complex cardiac surgery and compare them to a well-matched comparison population," investigators found that mortality was 55% in the patients who had received radiation therapy, compared with 28% in the nonexposed controls. At least half of the deaths among the cancer survivors were attributed to cardiopulmonary disease, while only 5% were due to recurrent malignancy.
"Surgical intervention should be applied cautiously to patients who have had significant thoracic radiation previously," including survivors of breast, lung, hematologic, thyroid, and other cancers.
Trying to gauge mortality risk in such patients solely by using their preoperative scores on standard risk-prediction measures is inaccurate at best. In this study population, the survivors of breast, lung, hematologic, thyroid, and other cancers would have been deemed at intermediate risk for undergoing cardiac surgery, with an expected mortality of only 3%-5% based on their preoperative assessments – 10 times lower than their actual postoperative mortality, said Dr. Willis Wu and his associates at the Heart and Vascular Institute at the Cleveland Clinic.
"Our findings suggest that surgical intervention should be applied cautiously to patients who have had significant thoracic radiation previously," they wrote. Cardiac surgery that is considered conventional or even routine in most patients – including coronary artery bypass graft, valve repair, valve replacement, vascular surgery, pericardiectomy, left ventricular assist device implantation, and myectomy – may actually worsen the condition of radiation-exposed patients.
"It seems appropriate to examine other types of treatment options in patients with radiation heart disease who have significant valvular, coronary, or myocardial disease," the researchers noted.
Dr. Wu and his associates performed their retrospective observational study because of the dearth of information regarding long-term outcomes in cancer survivors who were exposed to chest irradiation. This knowledge gap makes treatment decisions "especially difficult" once radiation-related heart disease is discovered.
Such patients are known to have high rates of coronary artery, valvular, pericardial, myocardial, and conduction disease that progresses over time and may not become evident until decades after cancer treatment was completed.
Surgery is often thought necessary to effectively address these cardiac complications of prior radiation therapy. "These patients have multiple cardiac lesions and [often] have comorbidities such as pulmonary or vascular disease related to radiation," the investigators said.
They analyzed the medical records of 478 patients undergoing cardiac surgery at their tertiary care center in 2000-2003. A total of 173 had a prior history of malignancy treated with chest irradiation; the remaining 305 patients had no such history and were matched to the radiation group for age, sex, type of surgery, and date of surgery.
The patients exposed to radiation were relatively young but had advanced symptoms. Forty-five percent had obstructive proximal coronary artery disease; one-fourth had already undergone open heart surgery, including 16% who had prior coronary artery bypass graft surgery. There also was a high prevalence of valvular disease, with 51% of patients showing at least moderate mitral regurgitation, 6% showing severe mitral stenosis, 29% showing at least moderate aortic regurgitation, 23% showing severe aortic stenosis, and 34% showing at least moderate tricuspid regurgitation.
The mean interval between the current cardiac surgery and the last thoracic radiation treatment was 18 years. Approximately 53% of the study patients had survived breast cancer, 27% had survived Hodgkin’s lymphoma, 7% had survived lung cancer, 6% had survived non-Hodgkin’s lymphoma, and 8% had survived other malignancies including thyroid and testicular cancers.
Approximately 75% of both study groups were women.
During a mean follow-up of 7.6 years, overall mortality was 37%. Despite the success of the cardiac procedures, a significantly higher proportion of radiation-exposed patients died (55%), compared with the unexposed patients (28%), for a hazard ratio of 2.54, Dr. Wu and his colleagues said (Circulation 2013 April 8; doi:10.1161/CIRCULATIONAHA.13.001435).
Mortality was significantly higher across all subgroups of patients exposed to radiation. In particular, it was high among patients expected to have low mortality: 43% in patients aged younger than 65 years and 45% in those with lower preoperative risk scores. In fact, the youngest patients in the radiation group fared worse than the oldest patients in the comparison group.
Mortality also was significantly higher for the radiation-exposed patients than for controls across all types of cardiac surgery.
The cause of death was not known for a substantial proportion of subjects in this study, but cardiopulmonary causes were implicated in at least half of the deaths among radiation-exposed patients. "Why this should be the case in a group of patients who received potentially life-preserving revascularization, valve, and other procedures is unknown at present," Dr. Wu and his associates said.
In addition, patients with radiation-related heart disease had longer hospital stays, were more likely to develop atrial fibrillation after surgery, were more likely to require pacemaker implantation after surgery, had higher rates of ventricular dysfunction and of valvular regurgitation following surgery, and had higher 30-day mortality.
The finding that radiation-exposed patients "remained in hospital for a considerable period after their surgery" suggests that the procedures and postoperative courses were complicated.
"Radiation patients frequently develop pulmonary complications as a result of open heart surgery, not the least of which are recurrent pleural effusions and severe restrictive lung disease. It is our experience that respiratory complications may significantly compromise function and survival in patients with extensive prior radiation," the researchers said.
"Additionally, the presence of myocardial disease either as a result of the underlying cardiac condition (potentially exacerbated by prior chemotherapy) or as a consequence of a restrictive-type cardiomyopathy produced by the effects of radiation may play a role in impaired survival and is not necessarily improved by valvular or revascularization surgery," they said.
The radiation-exposed patients’ poor long-term survival after conventional cardiac surgery indicates that other, nonsurgical treatments may be necessary for this complex patient population, they added.
No relevant financial conflicts were reported.
FROM CIRCULATION
Major Finding: Despite the success of the cardiac procedures, a significantly higher proportion of radiation-exposed patients died during follow-up (55%), compared with the unexposed patients (28%).
Data Source: A retrospective, single-center, observational study involving 173 cancer survivors who were previously exposed to thoracic radiotherapy and 305 matched controls who underwent cardiac surgery in 2000-2003 and were followed for approximately 7 years.
Disclosures: No relevant financial conflicts were reported.
New blood test could identify early pancreatic cancer
An investigational serum biomarker panel accurately identified potentially resectable cases of pancreatic cancer, opening the possibility that a blood test could identify the cancer early and improve its dismal prognosis.
The panel measured four metabolites and had an overall sensitivity of 71% and a specificity of 78% for pancreatic cancer; the specificity for resectable tumors also approached 78%, reported Dr. Masaru Yoshida (Cancer Epidemiol. Biomarkers Prev. 2013 March 29 [doi: 10.1158/1055-9965.EPI-12-1033]).
The panel was more accurate than were any of the currently used biomarkers, including CA19-9 and CEA, said Dr. Yoshida, professor and chief of metabolomics research at Kobe University, Japan.
"This novel diagnostic approach, which is safe and easy to apply as a screening method, is expected to improve the prognosis of patients with pancreatic cancer by detecting their cancers early, when still in a resectable and curable state," Dr. Yoshida said in a press statement.
The researchers created the panel based on study of a cohort of 85 subjects: 43 with pancreatic cancer and 42 healthy controls. First, they pared down 113 potentially useful metabolites into a workable testing panel. Of the 45 candidate metabolites, four of them – xylitol, 1,5-anhydro-D-glucitol, histidine, and inositol – showed the highest variation between the healthy controls and the patients. In the training cohort, the panel of these four markers had a sensitivity of 86% and specificity of 88% for pancreatic cancer.
Next, the researchers evaluated a validation cohort of 42 cancer patients, 41 healthy controls, and 23 patients with chronic pancreatitis. In this cohort, with an area under the curve of 0.76, the panel’s sensitivity for pancreatic cancer was 71% and specificity, 78%. In contrast, the sensitivity of CA19-9 was 69% and specificity 86%; for CEA, those numbers were 36% and 80%, respectively.
In the subset of patients with resectable pancreatic cancer, the panel’s sensitivity was 78%, compared with 56% for CA19-9 and 44% for CEA.
The metabolic panel was also able to identify chronic pancreatitis, with a 17% rate of false positives, compared with a false positive rate of 30% for CA19-9 and 44% for CEA.
A blood panel could address three key problems in the field of pancreatic cancer, Dr. Yoshida and his colleagues said. "The first is the difficulty of detecting pancreatic cancer early in resectable stages. A sensitivity and specificity of about 80% for resectable disease ... should be acceptable for clinical use because most patients with resectable cancer have no symptoms, and blood examinations are useful tools for initial screening examinations."
The second problem is the difficulty in differentiating pancreatic cancer from chronic pancreatitis. "Many gastroenterologists follow up chronic pancreatitis with scheduled CT, magnetic resonance imaging (MRI), and endoscopic ultrasound scans (EUS); tumor marker tests; and endoscopic retrograde cholangiopancreatography (ERCP), but the initial malignant changes are frequently overlooked, and pancreatic tumors can rapidly become unresectable. In addition, unnecessary resections for benign inflammatory lesions are sometimes done because of false-positive results from CA19-9 and/or imaging examinations."
The third clinical problem is the risk of complications that result from pancreatic examinations. Serum samples are easy and safe to obtain, with a low risk of adverse events and, with this panel, a potentially good return of information. A blood test could also be a useful population screening tool, the researchers noted.
The metabolites probably reflect metabolic derangement that arises not only from focal tumorigenesis, but also from systemic reactions to pancreatic disease, Dr. Yoshida and his coinvestigators wrote. Pancreatic cancer causes significant decreases in some amino and fatty acids, probably because the tumor recruits these substances to aid its rapid cellular proliferation.
"Patients with pancreatic disease are also troubled by malnutrition because of pancreatic endocrine and exocrine insufficiency. Therefore, there is a possibility that the decreases in serum metabolite levels also reflect malnutrition."
Finally, they said, the decrease in 1,5-anhydro-D-glucitol indicates the presence of hyperglycemia and recent glycosuria. "These results suggest that glucose tolerance was impaired in these patients because of pancreatic insufficiency."
The authors reported no financial conflicts. The work was supported by grants administered by agencies of the Japanese government.
An investigational serum biomarker panel accurately identified potentially resectable cases of pancreatic cancer, opening the possibility that a blood test could identify the cancer early and improve its dismal prognosis.
The panel measured four metabolites and had an overall sensitivity of 71% and a specificity of 78% for pancreatic cancer; the specificity for resectable tumors also approached 78%, reported Dr. Masaru Yoshida (Cancer Epidemiol. Biomarkers Prev. 2013 March 29 [doi: 10.1158/1055-9965.EPI-12-1033]).
The panel was more accurate than were any of the currently used biomarkers, including CA19-9 and CEA, said Dr. Yoshida, professor and chief of metabolomics research at Kobe University, Japan.
"This novel diagnostic approach, which is safe and easy to apply as a screening method, is expected to improve the prognosis of patients with pancreatic cancer by detecting their cancers early, when still in a resectable and curable state," Dr. Yoshida said in a press statement.
The researchers created the panel based on study of a cohort of 85 subjects: 43 with pancreatic cancer and 42 healthy controls. First, they pared down 113 potentially useful metabolites into a workable testing panel. Of the 45 candidate metabolites, four of them – xylitol, 1,5-anhydro-D-glucitol, histidine, and inositol – showed the highest variation between the healthy controls and the patients. In the training cohort, the panel of these four markers had a sensitivity of 86% and specificity of 88% for pancreatic cancer.
Next, the researchers evaluated a validation cohort of 42 cancer patients, 41 healthy controls, and 23 patients with chronic pancreatitis. In this cohort, with an area under the curve of 0.76, the panel’s sensitivity for pancreatic cancer was 71% and specificity, 78%. In contrast, the sensitivity of CA19-9 was 69% and specificity 86%; for CEA, those numbers were 36% and 80%, respectively.
In the subset of patients with resectable pancreatic cancer, the panel’s sensitivity was 78%, compared with 56% for CA19-9 and 44% for CEA.
The metabolic panel was also able to identify chronic pancreatitis, with a 17% rate of false positives, compared with a false positive rate of 30% for CA19-9 and 44% for CEA.
A blood panel could address three key problems in the field of pancreatic cancer, Dr. Yoshida and his colleagues said. "The first is the difficulty of detecting pancreatic cancer early in resectable stages. A sensitivity and specificity of about 80% for resectable disease ... should be acceptable for clinical use because most patients with resectable cancer have no symptoms, and blood examinations are useful tools for initial screening examinations."
The second problem is the difficulty in differentiating pancreatic cancer from chronic pancreatitis. "Many gastroenterologists follow up chronic pancreatitis with scheduled CT, magnetic resonance imaging (MRI), and endoscopic ultrasound scans (EUS); tumor marker tests; and endoscopic retrograde cholangiopancreatography (ERCP), but the initial malignant changes are frequently overlooked, and pancreatic tumors can rapidly become unresectable. In addition, unnecessary resections for benign inflammatory lesions are sometimes done because of false-positive results from CA19-9 and/or imaging examinations."
The third clinical problem is the risk of complications that result from pancreatic examinations. Serum samples are easy and safe to obtain, with a low risk of adverse events and, with this panel, a potentially good return of information. A blood test could also be a useful population screening tool, the researchers noted.
The metabolites probably reflect metabolic derangement that arises not only from focal tumorigenesis, but also from systemic reactions to pancreatic disease, Dr. Yoshida and his coinvestigators wrote. Pancreatic cancer causes significant decreases in some amino and fatty acids, probably because the tumor recruits these substances to aid its rapid cellular proliferation.
"Patients with pancreatic disease are also troubled by malnutrition because of pancreatic endocrine and exocrine insufficiency. Therefore, there is a possibility that the decreases in serum metabolite levels also reflect malnutrition."
Finally, they said, the decrease in 1,5-anhydro-D-glucitol indicates the presence of hyperglycemia and recent glycosuria. "These results suggest that glucose tolerance was impaired in these patients because of pancreatic insufficiency."
The authors reported no financial conflicts. The work was supported by grants administered by agencies of the Japanese government.
An investigational serum biomarker panel accurately identified potentially resectable cases of pancreatic cancer, opening the possibility that a blood test could identify the cancer early and improve its dismal prognosis.
The panel measured four metabolites and had an overall sensitivity of 71% and a specificity of 78% for pancreatic cancer; the specificity for resectable tumors also approached 78%, reported Dr. Masaru Yoshida (Cancer Epidemiol. Biomarkers Prev. 2013 March 29 [doi: 10.1158/1055-9965.EPI-12-1033]).
The panel was more accurate than were any of the currently used biomarkers, including CA19-9 and CEA, said Dr. Yoshida, professor and chief of metabolomics research at Kobe University, Japan.
"This novel diagnostic approach, which is safe and easy to apply as a screening method, is expected to improve the prognosis of patients with pancreatic cancer by detecting their cancers early, when still in a resectable and curable state," Dr. Yoshida said in a press statement.
The researchers created the panel based on study of a cohort of 85 subjects: 43 with pancreatic cancer and 42 healthy controls. First, they pared down 113 potentially useful metabolites into a workable testing panel. Of the 45 candidate metabolites, four of them – xylitol, 1,5-anhydro-D-glucitol, histidine, and inositol – showed the highest variation between the healthy controls and the patients. In the training cohort, the panel of these four markers had a sensitivity of 86% and specificity of 88% for pancreatic cancer.
Next, the researchers evaluated a validation cohort of 42 cancer patients, 41 healthy controls, and 23 patients with chronic pancreatitis. In this cohort, with an area under the curve of 0.76, the panel’s sensitivity for pancreatic cancer was 71% and specificity, 78%. In contrast, the sensitivity of CA19-9 was 69% and specificity 86%; for CEA, those numbers were 36% and 80%, respectively.
In the subset of patients with resectable pancreatic cancer, the panel’s sensitivity was 78%, compared with 56% for CA19-9 and 44% for CEA.
The metabolic panel was also able to identify chronic pancreatitis, with a 17% rate of false positives, compared with a false positive rate of 30% for CA19-9 and 44% for CEA.
A blood panel could address three key problems in the field of pancreatic cancer, Dr. Yoshida and his colleagues said. "The first is the difficulty of detecting pancreatic cancer early in resectable stages. A sensitivity and specificity of about 80% for resectable disease ... should be acceptable for clinical use because most patients with resectable cancer have no symptoms, and blood examinations are useful tools for initial screening examinations."
The second problem is the difficulty in differentiating pancreatic cancer from chronic pancreatitis. "Many gastroenterologists follow up chronic pancreatitis with scheduled CT, magnetic resonance imaging (MRI), and endoscopic ultrasound scans (EUS); tumor marker tests; and endoscopic retrograde cholangiopancreatography (ERCP), but the initial malignant changes are frequently overlooked, and pancreatic tumors can rapidly become unresectable. In addition, unnecessary resections for benign inflammatory lesions are sometimes done because of false-positive results from CA19-9 and/or imaging examinations."
The third clinical problem is the risk of complications that result from pancreatic examinations. Serum samples are easy and safe to obtain, with a low risk of adverse events and, with this panel, a potentially good return of information. A blood test could also be a useful population screening tool, the researchers noted.
The metabolites probably reflect metabolic derangement that arises not only from focal tumorigenesis, but also from systemic reactions to pancreatic disease, Dr. Yoshida and his coinvestigators wrote. Pancreatic cancer causes significant decreases in some amino and fatty acids, probably because the tumor recruits these substances to aid its rapid cellular proliferation.
"Patients with pancreatic disease are also troubled by malnutrition because of pancreatic endocrine and exocrine insufficiency. Therefore, there is a possibility that the decreases in serum metabolite levels also reflect malnutrition."
Finally, they said, the decrease in 1,5-anhydro-D-glucitol indicates the presence of hyperglycemia and recent glycosuria. "These results suggest that glucose tolerance was impaired in these patients because of pancreatic insufficiency."
The authors reported no financial conflicts. The work was supported by grants administered by agencies of the Japanese government.
FROM CANCER EPIDEMIOLOGY, BIOMARKERS, AND PREVENTION
Major finding: A serum panel of four metabolites had a 78% sensitivity for detecting resectable pancreatic cancers.
Data source: The panel was derived from a test cohort of 85, and a validation cohort of 42 cancer patients, 41 healthy controls, and 23 patients with chronic pancreatitis.
Disclosures: The authors had no financial disclosures. Japanese government agencies funded the study.
Bariatric surgery advancement spurs guideline update
Weight loss surgery patients should get routine copper supplements along with other vitamins and minerals, according to newly updated bariatric surgery guidelines from the American Association of Clinical Endocrinologists, the Obesity Society, and the American Society for Metabolic and Bariatric Surgery.
The groups call for 2 mg/day to offset the potential for surgery to cause a deficiency. Although routine copper screening isn’t necessary after the procedure, copper levels should be assessed and treated as needed in patients with anemia, neutropenia, myeloneuropathy, and impaired wound healing.
The copper recommendations are new since the guidelines were last published in 2008. Other recommendations – there are 74 in all – have been revised to incorporate new advances in weight loss surgery and an improved evidence base. Changes are pointed out where they’ve been made, and the level of evidence cited for each assertion. Pre- and postoperative bariatric surgery checklists have been added as well, to help avoid errors.
"This is actually a very unique collaboration among the internists represented by the endocrinologists and the obesity people and the surgeons. We actually agreed on all these things. The main intent is to assist with clinical decision making," including selecting patients and procedures and perioperative management, said lead author Dr. Jeffrey Mechanick, president-elect of the American Association of Clinical Endocrinologists and director of metabolic support at the Mt. Sinai School of Medicine in New York.
"We scrutinized every recommendation one by one in the context of the new data. In many cases the recommendations changed," he said in an interview.
Another new recommendation is for patients to be followed by their primary care physicians and screened for cancer prior to surgery, as appropriate for age and risk. Dr. Mechanick and his colleagues have also given more attention to consent, behavioral, and psychiatric issues as well as weight loss surgery in patients with type 2 diabetes.
There’s more information on sleeve gastrectomy, as well. Considered experimental in 2008, it’s now "approved and being done more widely. There are some very nice data about its metabolic effects, independent from just the weight loss effect, effects on glycemic control, and cardiovascular risk. It was very important to devote a fair amount of time" to the procedure, he said.
The guidelines note that "sleeve gastrectomy has demonstrated benefits comparable to other bariatric procedures. ... A national risk-adjusted database positions [it] between the laparoscopic adjustable gastric band and laparoscopic Roux-en-Y gastric bypass in terms of weight loss, co-morbidity resolution, and complications."
"We [also] addressed two issues which were quite controversial, and are still rather unsettled. The first is the use of the lap band for mild obesity. The second is the use of these weight loss procedures specifically for patients with type 2 diabetes for glycemic control. Since 2008, there’ve been a lot more data" about the issues, he said, just as there’ve been more data about the need for copper supplementation.
As in 2008, the guidelines do not recommend bariatric surgery solely for glycemic control. "We still don’t have an absolute indication for ‘diabetes surgery,’ but we do recognize the existence of the salutary effects on glycemic control when these procedures are done for weight loss. It was important for the reader to be exposed to this information," Dr. Mechanick said.
Regarding surgery in the mildly obese, the guidelines note that patients with a body mass index of 30-34.9 kg/m2 with diabetes or metabolic syndrome "may also be offered a bariatric procedure, although current evidence is limited by the number of subjects studied and lack of long-term data demonstrating net benefit."
The guidelines will be published in the March/April 2013 issue of Endocrine Practice and March 2013 issue of Surgery for Obesity and Related Diseases.
Dr. Mechanick disclosed compensation from Abbott Nutrition for lectures and program development.
From preoperative evaluation through bariatric
surgery and onward through long-term postoperative health management, weight
loss surgery and the medical care associated with it is, obligatorily, a
thoroughly interdisciplinary effort. Endocrinologists and internists on the
bariatrics team spearhead lifestyle management, medical weight loss, and
long-term postoperative care and efforts to maintain durable weight loss.
Surgeons, endocrinologists, and internists work together to select patients
appropriate for bariatric surgery, to choose the weight-loss surgery best
suited to each individual patient, and to provide the proper preoperative
evaluation. Surgeons perform the appropriate bariatric operation and oversee
immediate postoperative and short-term perioperative care, and, frequently in
concert with gastroenterologists, internists, and endocrinologists, manage
complications that can result from bariatric surgery. Finally, long-term
continuity of medical care and durable maintenance of weight loss is again
directed by the endocrinologist and internist.
Thus, given that the entire bariatric care schema is
such an interdisciplinary effort, clinical practice guidelines for the
management of bariatric surgical patients must also be the product of an
analogous interdisciplinary effort. It is with this aim and in this spirit that
the American Association of Clinical Endocrinologists (AACE), The Obesity
Society (TOS), and American Society for Metabolic and Bariatric Surgery (AAMBS)
published their initial Medical Guidelines for Clinical Practice for the Perioperative
Nutritional, Metabolic, and Nonsurgical Support of the Bariatric Surgery
Patient in 2008. The same cooperating societies have just published their
sequel with numerous substantive additions, changes, and refinements. The
Clinical Practice Guidelines for the Perioperative Nutritional, Metabolic, and
Nonsurgical Support of the Bariatric Surgery Patient – 2013 Update: Cosponsored
by American Association of Clinical Endocrinologists, The Obesity Society, and
American Society for Metabolic & Bariatric Surgery was published jointly in
the March issue of Surgery for Obesity and Related Disease, and in the
March/April issue of Endocrine Practice.
Clearly, much has changed in the bariatric landscape
in the intervening half-decade. Laparoscopic gastric band surgery has declined,
while sleeve gastrectomy has gained traction as a restrictive bariatric
operation with more robust weight loss and glycemic effects. The
increasingly recognized impact of Roux-en-Y gastric bypass surgery not only on
weight loss, but also on glycemic control and other endocrinologic endpoints
has prompted studies to determine if such benefits might also result from
restrictive-only bariatric surgeries such as sleeve gastrectomy, and initial
results appear encouraging. The arrival of more and higher-quality data with
longer-term follow up of a greater variety of endpoints has led to the ability
of these updated guidelines to provide an increasing number of more specific,
data-driven recommendations related to the broader spectrum of bariatric
surgical procedures and anatomies managed by clinicians today. They cover every
aspect of the bariatric surgical patient, from preoperative evaluation through
surgery, to postoperative management, all with more solidly outcomes-based
recommendations from over 400 references, with user-friendly and more
error-proof preoperative and postoperative care checklists, while still
arriving at such expert guidelines through interdisciplinary study and
agreement in this timely update.
John A. Martin, M.D., is associate
professor of medicine and surgery and director of endoscopy, Northwestern
University Feinberg School of Medicine, Chicago.
From preoperative evaluation through bariatric
surgery and onward through long-term postoperative health management, weight
loss surgery and the medical care associated with it is, obligatorily, a
thoroughly interdisciplinary effort. Endocrinologists and internists on the
bariatrics team spearhead lifestyle management, medical weight loss, and
long-term postoperative care and efforts to maintain durable weight loss.
Surgeons, endocrinologists, and internists work together to select patients
appropriate for bariatric surgery, to choose the weight-loss surgery best
suited to each individual patient, and to provide the proper preoperative
evaluation. Surgeons perform the appropriate bariatric operation and oversee
immediate postoperative and short-term perioperative care, and, frequently in
concert with gastroenterologists, internists, and endocrinologists, manage
complications that can result from bariatric surgery. Finally, long-term
continuity of medical care and durable maintenance of weight loss is again
directed by the endocrinologist and internist.
Thus, given that the entire bariatric care schema is
such an interdisciplinary effort, clinical practice guidelines for the
management of bariatric surgical patients must also be the product of an
analogous interdisciplinary effort. It is with this aim and in this spirit that
the American Association of Clinical Endocrinologists (AACE), The Obesity
Society (TOS), and American Society for Metabolic and Bariatric Surgery (AAMBS)
published their initial Medical Guidelines for Clinical Practice for the Perioperative
Nutritional, Metabolic, and Nonsurgical Support of the Bariatric Surgery
Patient in 2008. The same cooperating societies have just published their
sequel with numerous substantive additions, changes, and refinements. The
Clinical Practice Guidelines for the Perioperative Nutritional, Metabolic, and
Nonsurgical Support of the Bariatric Surgery Patient – 2013 Update: Cosponsored
by American Association of Clinical Endocrinologists, The Obesity Society, and
American Society for Metabolic & Bariatric Surgery was published jointly in
the March issue of Surgery for Obesity and Related Disease, and in the
March/April issue of Endocrine Practice.
Clearly, much has changed in the bariatric landscape
in the intervening half-decade. Laparoscopic gastric band surgery has declined,
while sleeve gastrectomy has gained traction as a restrictive bariatric
operation with more robust weight loss and glycemic effects. The
increasingly recognized impact of Roux-en-Y gastric bypass surgery not only on
weight loss, but also on glycemic control and other endocrinologic endpoints
has prompted studies to determine if such benefits might also result from
restrictive-only bariatric surgeries such as sleeve gastrectomy, and initial
results appear encouraging. The arrival of more and higher-quality data with
longer-term follow up of a greater variety of endpoints has led to the ability
of these updated guidelines to provide an increasing number of more specific,
data-driven recommendations related to the broader spectrum of bariatric
surgical procedures and anatomies managed by clinicians today. They cover every
aspect of the bariatric surgical patient, from preoperative evaluation through
surgery, to postoperative management, all with more solidly outcomes-based
recommendations from over 400 references, with user-friendly and more
error-proof preoperative and postoperative care checklists, while still
arriving at such expert guidelines through interdisciplinary study and
agreement in this timely update.
John A. Martin, M.D., is associate
professor of medicine and surgery and director of endoscopy, Northwestern
University Feinberg School of Medicine, Chicago.
From preoperative evaluation through bariatric
surgery and onward through long-term postoperative health management, weight
loss surgery and the medical care associated with it is, obligatorily, a
thoroughly interdisciplinary effort. Endocrinologists and internists on the
bariatrics team spearhead lifestyle management, medical weight loss, and
long-term postoperative care and efforts to maintain durable weight loss.
Surgeons, endocrinologists, and internists work together to select patients
appropriate for bariatric surgery, to choose the weight-loss surgery best
suited to each individual patient, and to provide the proper preoperative
evaluation. Surgeons perform the appropriate bariatric operation and oversee
immediate postoperative and short-term perioperative care, and, frequently in
concert with gastroenterologists, internists, and endocrinologists, manage
complications that can result from bariatric surgery. Finally, long-term
continuity of medical care and durable maintenance of weight loss is again
directed by the endocrinologist and internist.
Thus, given that the entire bariatric care schema is
such an interdisciplinary effort, clinical practice guidelines for the
management of bariatric surgical patients must also be the product of an
analogous interdisciplinary effort. It is with this aim and in this spirit that
the American Association of Clinical Endocrinologists (AACE), The Obesity
Society (TOS), and American Society for Metabolic and Bariatric Surgery (AAMBS)
published their initial Medical Guidelines for Clinical Practice for the Perioperative
Nutritional, Metabolic, and Nonsurgical Support of the Bariatric Surgery
Patient in 2008. The same cooperating societies have just published their
sequel with numerous substantive additions, changes, and refinements. The
Clinical Practice Guidelines for the Perioperative Nutritional, Metabolic, and
Nonsurgical Support of the Bariatric Surgery Patient – 2013 Update: Cosponsored
by American Association of Clinical Endocrinologists, The Obesity Society, and
American Society for Metabolic & Bariatric Surgery was published jointly in
the March issue of Surgery for Obesity and Related Disease, and in the
March/April issue of Endocrine Practice.
Clearly, much has changed in the bariatric landscape
in the intervening half-decade. Laparoscopic gastric band surgery has declined,
while sleeve gastrectomy has gained traction as a restrictive bariatric
operation with more robust weight loss and glycemic effects. The
increasingly recognized impact of Roux-en-Y gastric bypass surgery not only on
weight loss, but also on glycemic control and other endocrinologic endpoints
has prompted studies to determine if such benefits might also result from
restrictive-only bariatric surgeries such as sleeve gastrectomy, and initial
results appear encouraging. The arrival of more and higher-quality data with
longer-term follow up of a greater variety of endpoints has led to the ability
of these updated guidelines to provide an increasing number of more specific,
data-driven recommendations related to the broader spectrum of bariatric
surgical procedures and anatomies managed by clinicians today. They cover every
aspect of the bariatric surgical patient, from preoperative evaluation through
surgery, to postoperative management, all with more solidly outcomes-based
recommendations from over 400 references, with user-friendly and more
error-proof preoperative and postoperative care checklists, while still
arriving at such expert guidelines through interdisciplinary study and
agreement in this timely update.
John A. Martin, M.D., is associate
professor of medicine and surgery and director of endoscopy, Northwestern
University Feinberg School of Medicine, Chicago.
Weight loss surgery patients should get routine copper supplements along with other vitamins and minerals, according to newly updated bariatric surgery guidelines from the American Association of Clinical Endocrinologists, the Obesity Society, and the American Society for Metabolic and Bariatric Surgery.
The groups call for 2 mg/day to offset the potential for surgery to cause a deficiency. Although routine copper screening isn’t necessary after the procedure, copper levels should be assessed and treated as needed in patients with anemia, neutropenia, myeloneuropathy, and impaired wound healing.
The copper recommendations are new since the guidelines were last published in 2008. Other recommendations – there are 74 in all – have been revised to incorporate new advances in weight loss surgery and an improved evidence base. Changes are pointed out where they’ve been made, and the level of evidence cited for each assertion. Pre- and postoperative bariatric surgery checklists have been added as well, to help avoid errors.
"This is actually a very unique collaboration among the internists represented by the endocrinologists and the obesity people and the surgeons. We actually agreed on all these things. The main intent is to assist with clinical decision making," including selecting patients and procedures and perioperative management, said lead author Dr. Jeffrey Mechanick, president-elect of the American Association of Clinical Endocrinologists and director of metabolic support at the Mt. Sinai School of Medicine in New York.
"We scrutinized every recommendation one by one in the context of the new data. In many cases the recommendations changed," he said in an interview.
Another new recommendation is for patients to be followed by their primary care physicians and screened for cancer prior to surgery, as appropriate for age and risk. Dr. Mechanick and his colleagues have also given more attention to consent, behavioral, and psychiatric issues as well as weight loss surgery in patients with type 2 diabetes.
There’s more information on sleeve gastrectomy, as well. Considered experimental in 2008, it’s now "approved and being done more widely. There are some very nice data about its metabolic effects, independent from just the weight loss effect, effects on glycemic control, and cardiovascular risk. It was very important to devote a fair amount of time" to the procedure, he said.
The guidelines note that "sleeve gastrectomy has demonstrated benefits comparable to other bariatric procedures. ... A national risk-adjusted database positions [it] between the laparoscopic adjustable gastric band and laparoscopic Roux-en-Y gastric bypass in terms of weight loss, co-morbidity resolution, and complications."
"We [also] addressed two issues which were quite controversial, and are still rather unsettled. The first is the use of the lap band for mild obesity. The second is the use of these weight loss procedures specifically for patients with type 2 diabetes for glycemic control. Since 2008, there’ve been a lot more data" about the issues, he said, just as there’ve been more data about the need for copper supplementation.
As in 2008, the guidelines do not recommend bariatric surgery solely for glycemic control. "We still don’t have an absolute indication for ‘diabetes surgery,’ but we do recognize the existence of the salutary effects on glycemic control when these procedures are done for weight loss. It was important for the reader to be exposed to this information," Dr. Mechanick said.
Regarding surgery in the mildly obese, the guidelines note that patients with a body mass index of 30-34.9 kg/m2 with diabetes or metabolic syndrome "may also be offered a bariatric procedure, although current evidence is limited by the number of subjects studied and lack of long-term data demonstrating net benefit."
The guidelines will be published in the March/April 2013 issue of Endocrine Practice and March 2013 issue of Surgery for Obesity and Related Diseases.
Dr. Mechanick disclosed compensation from Abbott Nutrition for lectures and program development.
Weight loss surgery patients should get routine copper supplements along with other vitamins and minerals, according to newly updated bariatric surgery guidelines from the American Association of Clinical Endocrinologists, the Obesity Society, and the American Society for Metabolic and Bariatric Surgery.
The groups call for 2 mg/day to offset the potential for surgery to cause a deficiency. Although routine copper screening isn’t necessary after the procedure, copper levels should be assessed and treated as needed in patients with anemia, neutropenia, myeloneuropathy, and impaired wound healing.
The copper recommendations are new since the guidelines were last published in 2008. Other recommendations – there are 74 in all – have been revised to incorporate new advances in weight loss surgery and an improved evidence base. Changes are pointed out where they’ve been made, and the level of evidence cited for each assertion. Pre- and postoperative bariatric surgery checklists have been added as well, to help avoid errors.
"This is actually a very unique collaboration among the internists represented by the endocrinologists and the obesity people and the surgeons. We actually agreed on all these things. The main intent is to assist with clinical decision making," including selecting patients and procedures and perioperative management, said lead author Dr. Jeffrey Mechanick, president-elect of the American Association of Clinical Endocrinologists and director of metabolic support at the Mt. Sinai School of Medicine in New York.
"We scrutinized every recommendation one by one in the context of the new data. In many cases the recommendations changed," he said in an interview.
Another new recommendation is for patients to be followed by their primary care physicians and screened for cancer prior to surgery, as appropriate for age and risk. Dr. Mechanick and his colleagues have also given more attention to consent, behavioral, and psychiatric issues as well as weight loss surgery in patients with type 2 diabetes.
There’s more information on sleeve gastrectomy, as well. Considered experimental in 2008, it’s now "approved and being done more widely. There are some very nice data about its metabolic effects, independent from just the weight loss effect, effects on glycemic control, and cardiovascular risk. It was very important to devote a fair amount of time" to the procedure, he said.
The guidelines note that "sleeve gastrectomy has demonstrated benefits comparable to other bariatric procedures. ... A national risk-adjusted database positions [it] between the laparoscopic adjustable gastric band and laparoscopic Roux-en-Y gastric bypass in terms of weight loss, co-morbidity resolution, and complications."
"We [also] addressed two issues which were quite controversial, and are still rather unsettled. The first is the use of the lap band for mild obesity. The second is the use of these weight loss procedures specifically for patients with type 2 diabetes for glycemic control. Since 2008, there’ve been a lot more data" about the issues, he said, just as there’ve been more data about the need for copper supplementation.
As in 2008, the guidelines do not recommend bariatric surgery solely for glycemic control. "We still don’t have an absolute indication for ‘diabetes surgery,’ but we do recognize the existence of the salutary effects on glycemic control when these procedures are done for weight loss. It was important for the reader to be exposed to this information," Dr. Mechanick said.
Regarding surgery in the mildly obese, the guidelines note that patients with a body mass index of 30-34.9 kg/m2 with diabetes or metabolic syndrome "may also be offered a bariatric procedure, although current evidence is limited by the number of subjects studied and lack of long-term data demonstrating net benefit."
The guidelines will be published in the March/April 2013 issue of Endocrine Practice and March 2013 issue of Surgery for Obesity and Related Diseases.
Dr. Mechanick disclosed compensation from Abbott Nutrition for lectures and program development.
Mold again found in compounded product
A compounding pharmacy has issued a nationwide voluntary recall of all of its products after mold was discovered in one of its intravenous solutions. To date, no injuries or illnesses have been reported in association with the contaminated product: magnesium sulfate 2 g in dextrose 5% in water, 50 mL for injection.
The compounding pharmacy, Med Prep Consulting Inc., announced in a statement that it has voluntarily recalled all lots of all products compounded at its facility after clinicians at a Connecticut hospital notified the company that visible particulate contaminants were observed in five 50-mL bags of the product. These were unique and distinct lots compounded and dispensed by the pharmacy to the Connecticut hospital, according to the company.
The contaminants were subsequently confirmed to be mold; the pharmacy then decided to voluntarily recall all of its compounded products because of lack of sterility assurance.
Injectable steroids contaminated with mold and produced by a different compounding pharmacy were discovered last October and resulted in numerous infections and deaths nationwide. The issue also was part of a recent Institute of Medicine report on drug quality concerns.
Products produced by Med Prep Consulting are used for a wide range of therapeutic purposes in hospitals, outpatient facilities, and physicians’ offices. None are dispensed directly to patients for either self-administration or nursing administration. All products are packaged in plastic infusion bags, plastic infusion devices, plastic syringes, or glass vials.
Recalled products packaged in plastic infusion bags, plastic infusion devices, plastic syringes, and glass vials were distributed directly to regional hospital pharmacies in New Jersey, Pennsylvania, Connecticut, and Delaware.
Recalled products packaged in plastic syringes were distributed nationwide to physician office practice facilities and clinics. All of these products were distributed through March 17, 2013, from Tinton Falls, N.J., to both regional and nationwide locations.
All recipients of products compounded by Med Prep Consulting have been notified by telephone, fax, e-mail, and regular mail of the recall and have been instructed to remove and return the product to the pharmacy. Facilities with questions may contact the company at 732-493-3390. The company has asked that product complaints related to this recall be reported to the same number.
Adverse events that may be related to the use of these products should be reported to FDA’s MedWatch Adverse Event Reporting Program.
A compounding pharmacy has issued a nationwide voluntary recall of all of its products after mold was discovered in one of its intravenous solutions. To date, no injuries or illnesses have been reported in association with the contaminated product: magnesium sulfate 2 g in dextrose 5% in water, 50 mL for injection.
The compounding pharmacy, Med Prep Consulting Inc., announced in a statement that it has voluntarily recalled all lots of all products compounded at its facility after clinicians at a Connecticut hospital notified the company that visible particulate contaminants were observed in five 50-mL bags of the product. These were unique and distinct lots compounded and dispensed by the pharmacy to the Connecticut hospital, according to the company.
The contaminants were subsequently confirmed to be mold; the pharmacy then decided to voluntarily recall all of its compounded products because of lack of sterility assurance.
Injectable steroids contaminated with mold and produced by a different compounding pharmacy were discovered last October and resulted in numerous infections and deaths nationwide. The issue also was part of a recent Institute of Medicine report on drug quality concerns.
Products produced by Med Prep Consulting are used for a wide range of therapeutic purposes in hospitals, outpatient facilities, and physicians’ offices. None are dispensed directly to patients for either self-administration or nursing administration. All products are packaged in plastic infusion bags, plastic infusion devices, plastic syringes, or glass vials.
Recalled products packaged in plastic infusion bags, plastic infusion devices, plastic syringes, and glass vials were distributed directly to regional hospital pharmacies in New Jersey, Pennsylvania, Connecticut, and Delaware.
Recalled products packaged in plastic syringes were distributed nationwide to physician office practice facilities and clinics. All of these products were distributed through March 17, 2013, from Tinton Falls, N.J., to both regional and nationwide locations.
All recipients of products compounded by Med Prep Consulting have been notified by telephone, fax, e-mail, and regular mail of the recall and have been instructed to remove and return the product to the pharmacy. Facilities with questions may contact the company at 732-493-3390. The company has asked that product complaints related to this recall be reported to the same number.
Adverse events that may be related to the use of these products should be reported to FDA’s MedWatch Adverse Event Reporting Program.
A compounding pharmacy has issued a nationwide voluntary recall of all of its products after mold was discovered in one of its intravenous solutions. To date, no injuries or illnesses have been reported in association with the contaminated product: magnesium sulfate 2 g in dextrose 5% in water, 50 mL for injection.
The compounding pharmacy, Med Prep Consulting Inc., announced in a statement that it has voluntarily recalled all lots of all products compounded at its facility after clinicians at a Connecticut hospital notified the company that visible particulate contaminants were observed in five 50-mL bags of the product. These were unique and distinct lots compounded and dispensed by the pharmacy to the Connecticut hospital, according to the company.
The contaminants were subsequently confirmed to be mold; the pharmacy then decided to voluntarily recall all of its compounded products because of lack of sterility assurance.
Injectable steroids contaminated with mold and produced by a different compounding pharmacy were discovered last October and resulted in numerous infections and deaths nationwide. The issue also was part of a recent Institute of Medicine report on drug quality concerns.
Products produced by Med Prep Consulting are used for a wide range of therapeutic purposes in hospitals, outpatient facilities, and physicians’ offices. None are dispensed directly to patients for either self-administration or nursing administration. All products are packaged in plastic infusion bags, plastic infusion devices, plastic syringes, or glass vials.
Recalled products packaged in plastic infusion bags, plastic infusion devices, plastic syringes, and glass vials were distributed directly to regional hospital pharmacies in New Jersey, Pennsylvania, Connecticut, and Delaware.
Recalled products packaged in plastic syringes were distributed nationwide to physician office practice facilities and clinics. All of these products were distributed through March 17, 2013, from Tinton Falls, N.J., to both regional and nationwide locations.
All recipients of products compounded by Med Prep Consulting have been notified by telephone, fax, e-mail, and regular mail of the recall and have been instructed to remove and return the product to the pharmacy. Facilities with questions may contact the company at 732-493-3390. The company has asked that product complaints related to this recall be reported to the same number.
Adverse events that may be related to the use of these products should be reported to FDA’s MedWatch Adverse Event Reporting Program.
Gastric bypass shows best results for glycemic control
Gastric bypass leads to more significant and durable improvements in glycemic control than does sleeve gastrectomy or intensive medical therapy in moderately obese patients with type 2 diabetes, a prospective, randomized controlled trial has found.
Two years after the procedure, patients randomized to Roux-en-Y gastric bypass plus intensive medical therapy (IMT) had a mean HbA1c of 6.7 ± 1.2%, compared with 7.1 ± 0.8% for those who underwent sleeve gastrectomy and IMT, and 8.4 ± 2.3% for IMT alone, reported Dr Sangeeta R. Kashyap of the Cleveland Clinic and colleagues.
While both surgical procedures resulted in similar reductions in body weight, body mass index, and total body fat percentage at 24 months, gastric bypass resulted in the greatest absolute truncal fat reductions (–16% vs. –10%; P = .04), according to the findings, which were published online (Diabetes Care 2013 Feb. 25 [doi:10.2337/dc12-1596]).
The 2-year study enrolled a subset of 60 patients from the 1-year STAMPEDE trial, which evaluated the efficacy and safety of IMT alone – pharmacotherapy in conjunction with lifestyle interventions – or IMT combined with gastric bypass or sleeve gastrectomy. Of the 54 patients who completed the trial, the average age was 48.4 years, with a mean BMI of 36 kg/m2; most patients were taking at least three different diabetes medications.
The study extension examined the effects of the three treatment approaches on glucose control, pancreatic beta-cell function including insulin secretion and sensitivity, and body composition.
"Other long-term observational studies have documented greater relapse rates for glycemic control after gastric restrictive procedures such as sleeve gastrectomy, suggesting that surgical weight loss from enforced caloric restriction itself is insufficient to halt the disease," reported Dr Kashyap and colleagues. "Our results extend the findings from our initial 12-month report and suggest factors beyond weight loss that are specific to intestinal bypass patients help regulate glucose levels and restore pancreatic beta-cell function."
Gastric bypass was the only treatment to have any significant effects on pancreatic beta-cell function, with a median 5.8-fold (quartile 1: –7.00; quartile Q3: 11.29) increase in beta-cell function from baseline compared with only negligible increases for sleeve gastrectomy and IMT.
Researchers also observed a 2.7-fold (N = 9, 3.8 vs. 1.4; P < .001) increase in insulin sensitivity with gastric bypass among subjects not using insulin, compared with a 1.2-fold (N=10; 5.8 vs. 5.3) increase after sleeve gastrectomy, and no change in those randomized to IMT (2.6 vs. 2.4; P = NS).
"Bariatric surgery, particularly gastric bypass surgery, uniquely restores normal glucose tolerance and pancreatic [beta]-cell function, presumably by targeting the truncal fat that represents the core metabolic defect involved in diabetes pathogenesis," the researchers wrote.
Primary funding for the study came from Ethicon Endo-Surgery, with ancillary funding from the American Diabetes Association and the National Institutes of Health. The authors reported receiving research grants, consultations and honoraria from various companies and organizations, and one author received grants and honoraria from Ethicon Endo-Surgery as scientific advisory board member, consultant, and speaker.
Gastric bypass leads to more significant and durable improvements in glycemic control than does sleeve gastrectomy or intensive medical therapy in moderately obese patients with type 2 diabetes, a prospective, randomized controlled trial has found.
Two years after the procedure, patients randomized to Roux-en-Y gastric bypass plus intensive medical therapy (IMT) had a mean HbA1c of 6.7 ± 1.2%, compared with 7.1 ± 0.8% for those who underwent sleeve gastrectomy and IMT, and 8.4 ± 2.3% for IMT alone, reported Dr Sangeeta R. Kashyap of the Cleveland Clinic and colleagues.
While both surgical procedures resulted in similar reductions in body weight, body mass index, and total body fat percentage at 24 months, gastric bypass resulted in the greatest absolute truncal fat reductions (–16% vs. –10%; P = .04), according to the findings, which were published online (Diabetes Care 2013 Feb. 25 [doi:10.2337/dc12-1596]).
The 2-year study enrolled a subset of 60 patients from the 1-year STAMPEDE trial, which evaluated the efficacy and safety of IMT alone – pharmacotherapy in conjunction with lifestyle interventions – or IMT combined with gastric bypass or sleeve gastrectomy. Of the 54 patients who completed the trial, the average age was 48.4 years, with a mean BMI of 36 kg/m2; most patients were taking at least three different diabetes medications.
The study extension examined the effects of the three treatment approaches on glucose control, pancreatic beta-cell function including insulin secretion and sensitivity, and body composition.
"Other long-term observational studies have documented greater relapse rates for glycemic control after gastric restrictive procedures such as sleeve gastrectomy, suggesting that surgical weight loss from enforced caloric restriction itself is insufficient to halt the disease," reported Dr Kashyap and colleagues. "Our results extend the findings from our initial 12-month report and suggest factors beyond weight loss that are specific to intestinal bypass patients help regulate glucose levels and restore pancreatic beta-cell function."
Gastric bypass was the only treatment to have any significant effects on pancreatic beta-cell function, with a median 5.8-fold (quartile 1: –7.00; quartile Q3: 11.29) increase in beta-cell function from baseline compared with only negligible increases for sleeve gastrectomy and IMT.
Researchers also observed a 2.7-fold (N = 9, 3.8 vs. 1.4; P < .001) increase in insulin sensitivity with gastric bypass among subjects not using insulin, compared with a 1.2-fold (N=10; 5.8 vs. 5.3) increase after sleeve gastrectomy, and no change in those randomized to IMT (2.6 vs. 2.4; P = NS).
"Bariatric surgery, particularly gastric bypass surgery, uniquely restores normal glucose tolerance and pancreatic [beta]-cell function, presumably by targeting the truncal fat that represents the core metabolic defect involved in diabetes pathogenesis," the researchers wrote.
Primary funding for the study came from Ethicon Endo-Surgery, with ancillary funding from the American Diabetes Association and the National Institutes of Health. The authors reported receiving research grants, consultations and honoraria from various companies and organizations, and one author received grants and honoraria from Ethicon Endo-Surgery as scientific advisory board member, consultant, and speaker.
Gastric bypass leads to more significant and durable improvements in glycemic control than does sleeve gastrectomy or intensive medical therapy in moderately obese patients with type 2 diabetes, a prospective, randomized controlled trial has found.
Two years after the procedure, patients randomized to Roux-en-Y gastric bypass plus intensive medical therapy (IMT) had a mean HbA1c of 6.7 ± 1.2%, compared with 7.1 ± 0.8% for those who underwent sleeve gastrectomy and IMT, and 8.4 ± 2.3% for IMT alone, reported Dr Sangeeta R. Kashyap of the Cleveland Clinic and colleagues.
While both surgical procedures resulted in similar reductions in body weight, body mass index, and total body fat percentage at 24 months, gastric bypass resulted in the greatest absolute truncal fat reductions (–16% vs. –10%; P = .04), according to the findings, which were published online (Diabetes Care 2013 Feb. 25 [doi:10.2337/dc12-1596]).
The 2-year study enrolled a subset of 60 patients from the 1-year STAMPEDE trial, which evaluated the efficacy and safety of IMT alone – pharmacotherapy in conjunction with lifestyle interventions – or IMT combined with gastric bypass or sleeve gastrectomy. Of the 54 patients who completed the trial, the average age was 48.4 years, with a mean BMI of 36 kg/m2; most patients were taking at least three different diabetes medications.
The study extension examined the effects of the three treatment approaches on glucose control, pancreatic beta-cell function including insulin secretion and sensitivity, and body composition.
"Other long-term observational studies have documented greater relapse rates for glycemic control after gastric restrictive procedures such as sleeve gastrectomy, suggesting that surgical weight loss from enforced caloric restriction itself is insufficient to halt the disease," reported Dr Kashyap and colleagues. "Our results extend the findings from our initial 12-month report and suggest factors beyond weight loss that are specific to intestinal bypass patients help regulate glucose levels and restore pancreatic beta-cell function."
Gastric bypass was the only treatment to have any significant effects on pancreatic beta-cell function, with a median 5.8-fold (quartile 1: –7.00; quartile Q3: 11.29) increase in beta-cell function from baseline compared with only negligible increases for sleeve gastrectomy and IMT.
Researchers also observed a 2.7-fold (N = 9, 3.8 vs. 1.4; P < .001) increase in insulin sensitivity with gastric bypass among subjects not using insulin, compared with a 1.2-fold (N=10; 5.8 vs. 5.3) increase after sleeve gastrectomy, and no change in those randomized to IMT (2.6 vs. 2.4; P = NS).
"Bariatric surgery, particularly gastric bypass surgery, uniquely restores normal glucose tolerance and pancreatic [beta]-cell function, presumably by targeting the truncal fat that represents the core metabolic defect involved in diabetes pathogenesis," the researchers wrote.
Primary funding for the study came from Ethicon Endo-Surgery, with ancillary funding from the American Diabetes Association and the National Institutes of Health. The authors reported receiving research grants, consultations and honoraria from various companies and organizations, and one author received grants and honoraria from Ethicon Endo-Surgery as scientific advisory board member, consultant, and speaker.
Major finding: Gastric bypass beats sleeve gastrectomy and intensive medical therapy in type 2 diabetes management.
Data source: Prospective, randomized controlled trial in 60 moderately obese patients with type 2 diabetes.
Disclosures: Primary funding for the study came from Ethicon Endo-Surgery, with ancillary funding from the American Diabetes Association and the National Institutes of Health. The authors reported receiving research grants, consultations, and honoraria from various organizations, and one author received grants and honoraria from Ethicon Endo-Surgery as scientific advisory board member, consultant, and speaker.
Bariatric surgery reduces mortality in obese diabetic patients
Bariatric surgery can significantly reduce the need for insulin and can even induce complete remission of type 2 diabetes mellitus, but some forms of the procedure pose too much risk and should be shunned, said a diabetes specialist at the annual advanced postgraduate course held by the American Diabetes Association.
Depending on the type of procedure, weight loss in the first year following bariatric surgery can range from about 30 kg to more than 50 kg, and the surgery can reduce the risk of mortality for formerly obese patients by about 25%, said Dr. John Bantle, professor of medicine and director of the division of endocrinology and diabetes at the University of Minnesota in Minneapolis.
Bariatric surgery should be considered in type 2 diabetic patients with a body mass index greater than 35 kg/m2, and may also benefit patients with a BMI of 30-35, although there is not enough evidence to support the latter assertion, Dr. Bantle said.
"I would choose laparoscopic gastric bypass as the procedure to recommend. I think laparoscopic gastric banding doesn’t have enough efficacy, and duodenal switch has too many complications," he said.
Looking back at Look AHEAD
The most recent follow-up data from the Action for Health in Diabetes (Look AHEAD) study compared cardiovascular event rates among patients randomized to either an intensive lifestyle intervention or a diabetes support and education program. Analysis showed that the lifestyle intervention was associated with significantly more weight loss (–4.7% vs. –1.1% at 4 years, P less than .001), and with a more favorable change in fitness levels, hemoglobin A1c, systolic blood pressure, high-density lipoprotein cholesterol levels, and triglyceride levels (P less than .001 for all comparisons) (Arch. Intern. Med. 2010;170:1566-75).
However, in September 2012, the National Institutes of Health, acting on the advice of the trial’s data and safety monitoring team, halted the lifestyle intervention because the primary endpoint of reduction in cardiovascular events had not been reached and would be unlikely to do so in the 2 years remaining in the study.
Although the trial failed to meet the cardiovascular endpoint, there was no evidence of harm, and patients who were randomized to the intervention had reduced need for diabetes medications, less sleep apnea, and increases in both physical mobility and quality of life.
"In data that are still under analysis and not yet published, I think we are going to see that there was some benefit on microvascular complications," Dr. Bantle said.
Choose procedures wisely
If lifestyle interventions are not sufficient, bariatric surgery may be helpful, Dr. Bantle said. Procedures commonly used in the United States include laparoscopic gastric banding, Roux-en-Y gastric bypass, and increasingly, sleeve gastrectomy and biliopancreatic diversion with duodenal switch.
The latter procedure is similar to a jejunoileal bypass, an early type of bariatric surgery that is no longer performed due to the high complication rate. Biliopancreatic diversion with duodenal switch involves division of the stomach and creation of a small gastric pouch that is drained with a loop of intestine attached downward to the ileum toward its head, so that biliary and pancreatic drainage occurs through the small intestine before the digestive juices meet up with food for digestion and absorption toward the end of the intestinal tract.
"I think this procedure is a really bad idea; it’s too much like a jejunoileal bypass with anastomosis, and I advise all my patients who are considering bariatric surgery to decline the option to pursue this procedure," he said.
Evidence favors surgery
Reviewing the evidence, Dr. Bantle pointed to a 2005 meta-analysis of 147 studies of bariatric surgery for obesity, which showed a mean 30.2-kg weight loss at 12 months and 34.8-kg loss after 36 months in patients who had a gastric banding procedure, compared with 43.5 kg and 41.5 kg, respectively, for gastric bypass, and 51.9 kg and 53.1 kg for those who had surgery with a duodenal switch procedure (Ann. Intern. Med. 2005;142:547-59).
The study also showed, however, that the rate of adverse events, including reflux, vomiting, dysphagia, and dumping syndrome, was markedly higher for patients who underwent a duodenal switch, at 37.7%, compared with 7.0% for gastric banding and 16.9% for gastric bypass.
A 2007 Swedish study also saw a reduction in mortality at a mean of 10.9 years of follow-up among 2,010 obese patients who underwent bariatric surgery compared with 2,037 patients who received standard medical management. The hazard ratio for death in the surgical patients was 0.76 (P = .04). The most common causes of death were myocardial infarction, which occurred in 13 surgical patients and 25 controls, and cancer, which occurred in 29 surgical patients and 47 controls (N. Engl. J. Med. 2007;357:741-52).
Similarly, a retrospective cohort study comparing 7,925 obese people who underwent gastric bypass surgery with the same number of obese controls matched by age, gender, and BMI showed that after a mean follow-up of 7.1 years, adjusted mortality for surgical patients was 37.6 per 10,000 person-years, compared with 57.1 per 10,000 for controls, a relative difference of 40%. Additionally, diabetes-specific mortality declined by 92% in the gastric bypass patients (N. Engl. J. Med. 2007;357:753-61).
Following surgery, it is important to ensure that patients have a protein intake of 60-120 g daily, and if they have undergone gastric bypass or duodenal switch procedures their diet should be supplemented with a multivitamin containing folate, thiamine, iron, vitamin B12, calcium, and vitamin D, Dr. Bantle said.
"I would suggest that the earlier the surgery is done, the better. Remission of diabetes is predicted by short duration of diabetes, need for few diabetes medications, and high postprandial C-peptide, so if a patient is considering [bariatric surgery], I typically get a postprandial C-peptide and use that to counsel them about whether or not they’re likely to have remission of diabetes after the procedure," he concluded.
Dr. Bantle reported having no financial disclosures.
Bariatric surgery can significantly reduce the need for insulin and can even induce complete remission of type 2 diabetes mellitus, but some forms of the procedure pose too much risk and should be shunned, said a diabetes specialist at the annual advanced postgraduate course held by the American Diabetes Association.
Depending on the type of procedure, weight loss in the first year following bariatric surgery can range from about 30 kg to more than 50 kg, and the surgery can reduce the risk of mortality for formerly obese patients by about 25%, said Dr. John Bantle, professor of medicine and director of the division of endocrinology and diabetes at the University of Minnesota in Minneapolis.
Bariatric surgery should be considered in type 2 diabetic patients with a body mass index greater than 35 kg/m2, and may also benefit patients with a BMI of 30-35, although there is not enough evidence to support the latter assertion, Dr. Bantle said.
"I would choose laparoscopic gastric bypass as the procedure to recommend. I think laparoscopic gastric banding doesn’t have enough efficacy, and duodenal switch has too many complications," he said.
Looking back at Look AHEAD
The most recent follow-up data from the Action for Health in Diabetes (Look AHEAD) study compared cardiovascular event rates among patients randomized to either an intensive lifestyle intervention or a diabetes support and education program. Analysis showed that the lifestyle intervention was associated with significantly more weight loss (–4.7% vs. –1.1% at 4 years, P less than .001), and with a more favorable change in fitness levels, hemoglobin A1c, systolic blood pressure, high-density lipoprotein cholesterol levels, and triglyceride levels (P less than .001 for all comparisons) (Arch. Intern. Med. 2010;170:1566-75).
However, in September 2012, the National Institutes of Health, acting on the advice of the trial’s data and safety monitoring team, halted the lifestyle intervention because the primary endpoint of reduction in cardiovascular events had not been reached and would be unlikely to do so in the 2 years remaining in the study.
Although the trial failed to meet the cardiovascular endpoint, there was no evidence of harm, and patients who were randomized to the intervention had reduced need for diabetes medications, less sleep apnea, and increases in both physical mobility and quality of life.
"In data that are still under analysis and not yet published, I think we are going to see that there was some benefit on microvascular complications," Dr. Bantle said.
Choose procedures wisely
If lifestyle interventions are not sufficient, bariatric surgery may be helpful, Dr. Bantle said. Procedures commonly used in the United States include laparoscopic gastric banding, Roux-en-Y gastric bypass, and increasingly, sleeve gastrectomy and biliopancreatic diversion with duodenal switch.
The latter procedure is similar to a jejunoileal bypass, an early type of bariatric surgery that is no longer performed due to the high complication rate. Biliopancreatic diversion with duodenal switch involves division of the stomach and creation of a small gastric pouch that is drained with a loop of intestine attached downward to the ileum toward its head, so that biliary and pancreatic drainage occurs through the small intestine before the digestive juices meet up with food for digestion and absorption toward the end of the intestinal tract.
"I think this procedure is a really bad idea; it’s too much like a jejunoileal bypass with anastomosis, and I advise all my patients who are considering bariatric surgery to decline the option to pursue this procedure," he said.
Evidence favors surgery
Reviewing the evidence, Dr. Bantle pointed to a 2005 meta-analysis of 147 studies of bariatric surgery for obesity, which showed a mean 30.2-kg weight loss at 12 months and 34.8-kg loss after 36 months in patients who had a gastric banding procedure, compared with 43.5 kg and 41.5 kg, respectively, for gastric bypass, and 51.9 kg and 53.1 kg for those who had surgery with a duodenal switch procedure (Ann. Intern. Med. 2005;142:547-59).
The study also showed, however, that the rate of adverse events, including reflux, vomiting, dysphagia, and dumping syndrome, was markedly higher for patients who underwent a duodenal switch, at 37.7%, compared with 7.0% for gastric banding and 16.9% for gastric bypass.
A 2007 Swedish study also saw a reduction in mortality at a mean of 10.9 years of follow-up among 2,010 obese patients who underwent bariatric surgery compared with 2,037 patients who received standard medical management. The hazard ratio for death in the surgical patients was 0.76 (P = .04). The most common causes of death were myocardial infarction, which occurred in 13 surgical patients and 25 controls, and cancer, which occurred in 29 surgical patients and 47 controls (N. Engl. J. Med. 2007;357:741-52).
Similarly, a retrospective cohort study comparing 7,925 obese people who underwent gastric bypass surgery with the same number of obese controls matched by age, gender, and BMI showed that after a mean follow-up of 7.1 years, adjusted mortality for surgical patients was 37.6 per 10,000 person-years, compared with 57.1 per 10,000 for controls, a relative difference of 40%. Additionally, diabetes-specific mortality declined by 92% in the gastric bypass patients (N. Engl. J. Med. 2007;357:753-61).
Following surgery, it is important to ensure that patients have a protein intake of 60-120 g daily, and if they have undergone gastric bypass or duodenal switch procedures their diet should be supplemented with a multivitamin containing folate, thiamine, iron, vitamin B12, calcium, and vitamin D, Dr. Bantle said.
"I would suggest that the earlier the surgery is done, the better. Remission of diabetes is predicted by short duration of diabetes, need for few diabetes medications, and high postprandial C-peptide, so if a patient is considering [bariatric surgery], I typically get a postprandial C-peptide and use that to counsel them about whether or not they’re likely to have remission of diabetes after the procedure," he concluded.
Dr. Bantle reported having no financial disclosures.
Bariatric surgery can significantly reduce the need for insulin and can even induce complete remission of type 2 diabetes mellitus, but some forms of the procedure pose too much risk and should be shunned, said a diabetes specialist at the annual advanced postgraduate course held by the American Diabetes Association.
Depending on the type of procedure, weight loss in the first year following bariatric surgery can range from about 30 kg to more than 50 kg, and the surgery can reduce the risk of mortality for formerly obese patients by about 25%, said Dr. John Bantle, professor of medicine and director of the division of endocrinology and diabetes at the University of Minnesota in Minneapolis.
Bariatric surgery should be considered in type 2 diabetic patients with a body mass index greater than 35 kg/m2, and may also benefit patients with a BMI of 30-35, although there is not enough evidence to support the latter assertion, Dr. Bantle said.
"I would choose laparoscopic gastric bypass as the procedure to recommend. I think laparoscopic gastric banding doesn’t have enough efficacy, and duodenal switch has too many complications," he said.
Looking back at Look AHEAD
The most recent follow-up data from the Action for Health in Diabetes (Look AHEAD) study compared cardiovascular event rates among patients randomized to either an intensive lifestyle intervention or a diabetes support and education program. Analysis showed that the lifestyle intervention was associated with significantly more weight loss (–4.7% vs. –1.1% at 4 years, P less than .001), and with a more favorable change in fitness levels, hemoglobin A1c, systolic blood pressure, high-density lipoprotein cholesterol levels, and triglyceride levels (P less than .001 for all comparisons) (Arch. Intern. Med. 2010;170:1566-75).
However, in September 2012, the National Institutes of Health, acting on the advice of the trial’s data and safety monitoring team, halted the lifestyle intervention because the primary endpoint of reduction in cardiovascular events had not been reached and would be unlikely to do so in the 2 years remaining in the study.
Although the trial failed to meet the cardiovascular endpoint, there was no evidence of harm, and patients who were randomized to the intervention had reduced need for diabetes medications, less sleep apnea, and increases in both physical mobility and quality of life.
"In data that are still under analysis and not yet published, I think we are going to see that there was some benefit on microvascular complications," Dr. Bantle said.
Choose procedures wisely
If lifestyle interventions are not sufficient, bariatric surgery may be helpful, Dr. Bantle said. Procedures commonly used in the United States include laparoscopic gastric banding, Roux-en-Y gastric bypass, and increasingly, sleeve gastrectomy and biliopancreatic diversion with duodenal switch.
The latter procedure is similar to a jejunoileal bypass, an early type of bariatric surgery that is no longer performed due to the high complication rate. Biliopancreatic diversion with duodenal switch involves division of the stomach and creation of a small gastric pouch that is drained with a loop of intestine attached downward to the ileum toward its head, so that biliary and pancreatic drainage occurs through the small intestine before the digestive juices meet up with food for digestion and absorption toward the end of the intestinal tract.
"I think this procedure is a really bad idea; it’s too much like a jejunoileal bypass with anastomosis, and I advise all my patients who are considering bariatric surgery to decline the option to pursue this procedure," he said.
Evidence favors surgery
Reviewing the evidence, Dr. Bantle pointed to a 2005 meta-analysis of 147 studies of bariatric surgery for obesity, which showed a mean 30.2-kg weight loss at 12 months and 34.8-kg loss after 36 months in patients who had a gastric banding procedure, compared with 43.5 kg and 41.5 kg, respectively, for gastric bypass, and 51.9 kg and 53.1 kg for those who had surgery with a duodenal switch procedure (Ann. Intern. Med. 2005;142:547-59).
The study also showed, however, that the rate of adverse events, including reflux, vomiting, dysphagia, and dumping syndrome, was markedly higher for patients who underwent a duodenal switch, at 37.7%, compared with 7.0% for gastric banding and 16.9% for gastric bypass.
A 2007 Swedish study also saw a reduction in mortality at a mean of 10.9 years of follow-up among 2,010 obese patients who underwent bariatric surgery compared with 2,037 patients who received standard medical management. The hazard ratio for death in the surgical patients was 0.76 (P = .04). The most common causes of death were myocardial infarction, which occurred in 13 surgical patients and 25 controls, and cancer, which occurred in 29 surgical patients and 47 controls (N. Engl. J. Med. 2007;357:741-52).
Similarly, a retrospective cohort study comparing 7,925 obese people who underwent gastric bypass surgery with the same number of obese controls matched by age, gender, and BMI showed that after a mean follow-up of 7.1 years, adjusted mortality for surgical patients was 37.6 per 10,000 person-years, compared with 57.1 per 10,000 for controls, a relative difference of 40%. Additionally, diabetes-specific mortality declined by 92% in the gastric bypass patients (N. Engl. J. Med. 2007;357:753-61).
Following surgery, it is important to ensure that patients have a protein intake of 60-120 g daily, and if they have undergone gastric bypass or duodenal switch procedures their diet should be supplemented with a multivitamin containing folate, thiamine, iron, vitamin B12, calcium, and vitamin D, Dr. Bantle said.
"I would suggest that the earlier the surgery is done, the better. Remission of diabetes is predicted by short duration of diabetes, need for few diabetes medications, and high postprandial C-peptide, so if a patient is considering [bariatric surgery], I typically get a postprandial C-peptide and use that to counsel them about whether or not they’re likely to have remission of diabetes after the procedure," he concluded.
Dr. Bantle reported having no financial disclosures.
AT THE ADA ADVANCED POSTGRADUATE COURSE
Major finding: Weight loss in the first year following bariatric surgery ranges from about 30 kg to more than 50 kg, and the surgery can reduce the risk of mortality for formerly obese patients by about 25%,
Data source: Review of medical literature on bariatric surgery for obese patients both with and without type 2 diabetes mellitus.
Disclosures: Dr. Bantle reported having no financial disclosures.
Outcomes no better at bariatric centers of excellence
Centers of Excellence for bariatric surgery – the only locations where the procedures are covered by Medicare – do not yield fewer complications or better outcomes for patients.
The Centers for Medicare and Medicaid Services established the Centers of Excellence in 2006 with an eye on increasing safety and decreasing negative outcomes.
Dr. Justin B. Dimick of the University of Michigan, Ann Arbor, and his colleagues, reviewed bariatric surgeries performed before and after that policy went into effect and found that there were no statistically significant improvement in complications, serious complications, or reoperations. Their findings were published Feb. 26 in JAMA.
The overall safety of bariatric surgery has increased over the years, Dr. Dimick noted, as surgeons have increasingly chosen less-invasive, lower-risk procedures such as laparoscopic gastric banding, rather than higher-risk, open procedures. Surgeons’ experience has increased and technology has improved as well; both trends have made the operations easier and less dangerous.
"Our study found large improvements in bariatric surgery outcomes over time even after adjusting for changes in procedure use," he wrote.
Taking into account patient factors such as age and comorbidities, procedure type, and year of operation, 5.5% of patients at a COE hospital had any complication, compared with 6% for those at a nondesignated facility. For serious complications, the rate was 2.2% at COEs vs. 2.5% at non-COEs. The reoperation rate was 0.83% for patients in COE hospitals, compared with 0.96% at nondesignated facilities (JAMA 2013;309:792-9).
The results were drawn from comparisons of discharge data from 2004-2009 in 12 states, chosen for geographic diversity. The discharges were for Medicare and non-Medicare patients, and the researchers examined outcomes for the 2 years before the CMS policy change and about 3 years after. Overall, there were 6,723 Medicare patients who had bariatric surgery before 2006, and 15,684 who had it afterward. For non-Medicare patients, the data covered 95,558 procedures before the change, and 155,117 afterward.
Facilities can gain the Centers of Excellence designation if they meet three primary criteria: provide accommodations for obese patients, and other structural elements; perform a minimum volume of 125 cases per year; and, submit data to either the American College of Surgeons or the American Society of Metabolic and Bariatric Surgery registry. Facilities are certified by the ACS or the ASMBS. Since 2006, the CMS has designated almost 600 facilities as a Bariatric Center of Excellence.
The structural resources required by the CMS do not differ much from what the Joint Commission requires, Dr. Dimick wrote, and volume standards don’t necessarily correlate with quality. He added that the registry data are not used in any kind of continuous quality improvement process, and thus probably do not have much of an impact.
The ACS and the ASMBS have been developing new outcomes measures and standards for that registry, the Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program (MBSAQIP). Public comment closed on Jan. 15. The organizations said they expected to incorporate any comments into a final draft.
The CMS policy restricting coverage to Centers of Excellence should be revisited, Dr. Dimick advised. The policy does not appear to improve outcomes and may even have the unintended consequence of "sacrificing long-term effectiveness for improved short-term safety."
The study was supported by grants from the National Institute on Aging and the Agency for Healthcare Research and Quality. The authors had no relevant disclosures.
On Twitter @aliciaault
Since the Medicare policy decision 7 years ago, the Centers of Excellence have contributed to notable improvements in bariatric surgery, primarily by increasing awareness of variation between centers and focusing the attention of physicians, hospital health care personnel and administrators, and payers on the need to improve quality and safety. However, the limitations of COEs are now recognized by the American Society for Metabolic and Bariatric Surgery and the American College of Surgeons, which have joined forces and have an initiative underway to develop new standards, with an increased focus on more robust outcome measures.
As the CMS and the surgical societies reexamine the COE policy in bariatric surgery, there is an opportunity for them to be creative; to catapult surgical outcomes science forward through scalable approaches to data sharing, measurement, collaborative networks and comparative effectiveness research; and to design a program that can not only identify high-quality hospitals, but also provide a sustained mechanism for quality improvement.
Dr. Caprice C. Greenberg is associate professor of surgery at the University of Wisconsin, Madison, and director of the Wisconsin Surgical Outcomes Research Program. Her remarks were made in an editorial accompanying Dr. Dimick’s report (JAMA 2013;309:827-8).
The Centers for Medicare and Medicaid Services,
Dr. Justin B. Dimick
Since the Medicare policy decision 7 years ago, the Centers of Excellence have contributed to notable improvements in bariatric surgery, primarily by increasing awareness of variation between centers and focusing the attention of physicians, hospital health care personnel and administrators, and payers on the need to improve quality and safety. However, the limitations of COEs are now recognized by the American Society for Metabolic and Bariatric Surgery and the American College of Surgeons, which have joined forces and have an initiative underway to develop new standards, with an increased focus on more robust outcome measures.
As the CMS and the surgical societies reexamine the COE policy in bariatric surgery, there is an opportunity for them to be creative; to catapult surgical outcomes science forward through scalable approaches to data sharing, measurement, collaborative networks and comparative effectiveness research; and to design a program that can not only identify high-quality hospitals, but also provide a sustained mechanism for quality improvement.
Dr. Caprice C. Greenberg is associate professor of surgery at the University of Wisconsin, Madison, and director of the Wisconsin Surgical Outcomes Research Program. Her remarks were made in an editorial accompanying Dr. Dimick’s report (JAMA 2013;309:827-8).
Since the Medicare policy decision 7 years ago, the Centers of Excellence have contributed to notable improvements in bariatric surgery, primarily by increasing awareness of variation between centers and focusing the attention of physicians, hospital health care personnel and administrators, and payers on the need to improve quality and safety. However, the limitations of COEs are now recognized by the American Society for Metabolic and Bariatric Surgery and the American College of Surgeons, which have joined forces and have an initiative underway to develop new standards, with an increased focus on more robust outcome measures.
As the CMS and the surgical societies reexamine the COE policy in bariatric surgery, there is an opportunity for them to be creative; to catapult surgical outcomes science forward through scalable approaches to data sharing, measurement, collaborative networks and comparative effectiveness research; and to design a program that can not only identify high-quality hospitals, but also provide a sustained mechanism for quality improvement.
Dr. Caprice C. Greenberg is associate professor of surgery at the University of Wisconsin, Madison, and director of the Wisconsin Surgical Outcomes Research Program. Her remarks were made in an editorial accompanying Dr. Dimick’s report (JAMA 2013;309:827-8).
Centers of Excellence for bariatric surgery – the only locations where the procedures are covered by Medicare – do not yield fewer complications or better outcomes for patients.
The Centers for Medicare and Medicaid Services established the Centers of Excellence in 2006 with an eye on increasing safety and decreasing negative outcomes.
Dr. Justin B. Dimick of the University of Michigan, Ann Arbor, and his colleagues, reviewed bariatric surgeries performed before and after that policy went into effect and found that there were no statistically significant improvement in complications, serious complications, or reoperations. Their findings were published Feb. 26 in JAMA.
The overall safety of bariatric surgery has increased over the years, Dr. Dimick noted, as surgeons have increasingly chosen less-invasive, lower-risk procedures such as laparoscopic gastric banding, rather than higher-risk, open procedures. Surgeons’ experience has increased and technology has improved as well; both trends have made the operations easier and less dangerous.
"Our study found large improvements in bariatric surgery outcomes over time even after adjusting for changes in procedure use," he wrote.
Taking into account patient factors such as age and comorbidities, procedure type, and year of operation, 5.5% of patients at a COE hospital had any complication, compared with 6% for those at a nondesignated facility. For serious complications, the rate was 2.2% at COEs vs. 2.5% at non-COEs. The reoperation rate was 0.83% for patients in COE hospitals, compared with 0.96% at nondesignated facilities (JAMA 2013;309:792-9).
The results were drawn from comparisons of discharge data from 2004-2009 in 12 states, chosen for geographic diversity. The discharges were for Medicare and non-Medicare patients, and the researchers examined outcomes for the 2 years before the CMS policy change and about 3 years after. Overall, there were 6,723 Medicare patients who had bariatric surgery before 2006, and 15,684 who had it afterward. For non-Medicare patients, the data covered 95,558 procedures before the change, and 155,117 afterward.
Facilities can gain the Centers of Excellence designation if they meet three primary criteria: provide accommodations for obese patients, and other structural elements; perform a minimum volume of 125 cases per year; and, submit data to either the American College of Surgeons or the American Society of Metabolic and Bariatric Surgery registry. Facilities are certified by the ACS or the ASMBS. Since 2006, the CMS has designated almost 600 facilities as a Bariatric Center of Excellence.
The structural resources required by the CMS do not differ much from what the Joint Commission requires, Dr. Dimick wrote, and volume standards don’t necessarily correlate with quality. He added that the registry data are not used in any kind of continuous quality improvement process, and thus probably do not have much of an impact.
The ACS and the ASMBS have been developing new outcomes measures and standards for that registry, the Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program (MBSAQIP). Public comment closed on Jan. 15. The organizations said they expected to incorporate any comments into a final draft.
The CMS policy restricting coverage to Centers of Excellence should be revisited, Dr. Dimick advised. The policy does not appear to improve outcomes and may even have the unintended consequence of "sacrificing long-term effectiveness for improved short-term safety."
The study was supported by grants from the National Institute on Aging and the Agency for Healthcare Research and Quality. The authors had no relevant disclosures.
On Twitter @aliciaault
Centers of Excellence for bariatric surgery – the only locations where the procedures are covered by Medicare – do not yield fewer complications or better outcomes for patients.
The Centers for Medicare and Medicaid Services established the Centers of Excellence in 2006 with an eye on increasing safety and decreasing negative outcomes.
Dr. Justin B. Dimick of the University of Michigan, Ann Arbor, and his colleagues, reviewed bariatric surgeries performed before and after that policy went into effect and found that there were no statistically significant improvement in complications, serious complications, or reoperations. Their findings were published Feb. 26 in JAMA.
The overall safety of bariatric surgery has increased over the years, Dr. Dimick noted, as surgeons have increasingly chosen less-invasive, lower-risk procedures such as laparoscopic gastric banding, rather than higher-risk, open procedures. Surgeons’ experience has increased and technology has improved as well; both trends have made the operations easier and less dangerous.
"Our study found large improvements in bariatric surgery outcomes over time even after adjusting for changes in procedure use," he wrote.
Taking into account patient factors such as age and comorbidities, procedure type, and year of operation, 5.5% of patients at a COE hospital had any complication, compared with 6% for those at a nondesignated facility. For serious complications, the rate was 2.2% at COEs vs. 2.5% at non-COEs. The reoperation rate was 0.83% for patients in COE hospitals, compared with 0.96% at nondesignated facilities (JAMA 2013;309:792-9).
The results were drawn from comparisons of discharge data from 2004-2009 in 12 states, chosen for geographic diversity. The discharges were for Medicare and non-Medicare patients, and the researchers examined outcomes for the 2 years before the CMS policy change and about 3 years after. Overall, there were 6,723 Medicare patients who had bariatric surgery before 2006, and 15,684 who had it afterward. For non-Medicare patients, the data covered 95,558 procedures before the change, and 155,117 afterward.
Facilities can gain the Centers of Excellence designation if they meet three primary criteria: provide accommodations for obese patients, and other structural elements; perform a minimum volume of 125 cases per year; and, submit data to either the American College of Surgeons or the American Society of Metabolic and Bariatric Surgery registry. Facilities are certified by the ACS or the ASMBS. Since 2006, the CMS has designated almost 600 facilities as a Bariatric Center of Excellence.
The structural resources required by the CMS do not differ much from what the Joint Commission requires, Dr. Dimick wrote, and volume standards don’t necessarily correlate with quality. He added that the registry data are not used in any kind of continuous quality improvement process, and thus probably do not have much of an impact.
The ACS and the ASMBS have been developing new outcomes measures and standards for that registry, the Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program (MBSAQIP). Public comment closed on Jan. 15. The organizations said they expected to incorporate any comments into a final draft.
The CMS policy restricting coverage to Centers of Excellence should be revisited, Dr. Dimick advised. The policy does not appear to improve outcomes and may even have the unintended consequence of "sacrificing long-term effectiveness for improved short-term safety."
The study was supported by grants from the National Institute on Aging and the Agency for Healthcare Research and Quality. The authors had no relevant disclosures.
On Twitter @aliciaault
The Centers for Medicare and Medicaid Services,
Dr. Justin B. Dimick
The Centers for Medicare and Medicaid Services,
Dr. Justin B. Dimick
FROM JAMA
Major finding: Complication rates were 5.5% at Centers of Excellence, compared with 6% at nondesignated facilities. Reoperation rates were 0.83% for COEs, compared with 0.96% at nondesignated facilities.
Data source: A retrospective, longitudinal study using Medicare and non-Medicare hospital discharge data for 321,464 patients from 12 states for 2004-2009.
Disclosures: The study was supported by grants from the National Institute on Aging and the Agency for Healthcare Research and Quality. The authors had no relevant disclosures.