User login
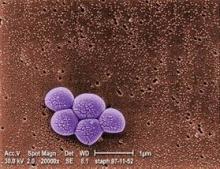
Health care-associated infections in hospitals continue to decline
The rates of three major types of health care–associated infections have continued to decrease in U.S. hospitals, according to a new report from the Centers for Disease Control and Prevention.
The rate of central line–associated bloodstream infections (CLABSI) is down nationally by 41%, catheter-associated urinary tract infections (CAUTI) are down by 7%, and surgical site infections (SSI) for a combined 10 surgical procedures are down by 17%.
These declines are measured against the 2008 baseline rates of CLABSIs, CAUTIs and SSIs reported when the U.S. Department of Health and Human Services established its 5-year goals for reducing health care–associated infections by the end of 2013. The HHS goals include reducing CLABSIs by 50%, CAUTIs by 25%, and SSIs by 25%. The American College of Surgeons and the CDC have partnered to develop the means to report, measure, and prevent health care–associated infections, and the ACS has been instrumental in collecting and submitting standard SSI measure data and other data to the CDC’s National Healthcare Safety Network (NHSN) and the ACS’s National Surgical Quality Improvement Program (NSQIP).
“One thing these numbers show us is the complexity of achieving im provement,” said Dr. Clifford Y. Ko, MD, MS, FACS, Director of the American College of Surgeons (ACS) Division of Research and Optimal Patient Care. “The College’s recent effort with the Joint Commission Center for Transforming Healthcare to reduce SSI has shown us that SSIs are very multifactorial, and not every provider or facility has the same is sues to address.
“Similarly, even the measurement and analytical techniques used in this study can be improved upon,” Dr. Ko added. “While better than they used to be, we know through ACS NSQIP®[the College’s National Surgical Quality Improvement Program] that we can measure and feedback risk-adjusted infection rates on all procedures, not just 10. This is important for gaining traction with all providers because it will likely require the effort of all providers to achieve system- wide, sustained improvement.”
Paul J. Malpiedi and associates at the CDC reported the findings in the 2011 National and State Healthcare-Associated Infections Standard Infection Ratio Report. Mr. Malpiedi’s team compared the standard infection ratios (SIRs) between 2010 and 2011 to determine progress in preventing health care–associated infections.
Standard infection ratios developed at the national, state, and facility levels compare the number of infections that actually occurred to the number that would be expected based on the referent years: 2008 for CLABSIs and SSIs, and 2009 for CAUTIs. The standard infection ratios were adjusted to account for hospital type, hospital size (based on bed number), and hospital affiliation with a medical school.
Mr. Malpiedi’s team analyzed the data reported for the 2011 calendar year to the NHSN from 3,472 facilities for CLABSIs, 1,807 facilities for CAUTIs and 2,130 facilities for SSIs, based on reports submitted through Sept. 4, 2012. Non–acute care hospitals, outpatient dialysis facilities, inpatient dialysis wards, long-term care facilities, and outpatient surgical settings were excluded from the analysis.
A total of 18,113 CLABSIs were reported during 2011, compared with 30,617 that were predicted to occur based on the 2008 referent population, for an SIR of 0.592. This 41% reduction is an improvement over the 2010 reduction of 32%. With a median SIR of 0.469, half the reporting facilities in 2011 had reduced their CLABSIs by 53%. The lowest rate of CLABSIs was reported in ICUs, where the infections had declined 44% since 2008.
The number of facilities reporting infection data increased from 2010 to 2011. The 3,472 facilities in 50 states and the District of Columbia that reported data for CLABSIs represented a 55% increase from those reporting in 2010. The 2011 data came from 12,122 patient care locations, which included 5,722 ICUs (47%), 5,436 wards (45%) and 946 NICUs (8%).
The overall reduction in CAUTIs was less substantial, with no significant overall change since 2010. The 7% reduction in CAUTIs, with an SIR of 0.93, came from 14,315 reported CAUTIs, compared to the 15,398 predicted infections based on the 2009 referent population. Specifically, CAUTIs in wards declined about 15% while the infection rates in ICUs remained unchanged.
When only the 550 facilities that reported in both 2010 and 2011 were included in the analysis, the reduction since 2010 was statistically significant. A total of 1,807 facilities in 50 states and the District of Columbia reported CAUTI data, an 84% increase from the 2010 number of 981 reporting facilities. The 6,402 patient care locations included in the CAUTI data came from 2,633 ICUs (41%) and 3,769 wards (59%).
A total of 2,130 facilities from 48 states and the District of Columbia reported SSI data. Among the 748,192 surgical procedures included were 6,357 deep incisional and organ/space infections occurred, compared to the 7,683 SSIs that were predicted using the 2008 baseline, for an SIR of 0.827 .
This lower SIR represents a 17% decline in SSIs since 2008. SSIs declined for hip arthroplasty (10.4% decline), knee arthroplasty (14.3%), coronary artery bypass graft (22.1%), cardiac surgery (30.2%), peripheral vascular bypass surgery (25.5%), abdominal aortic aneurysm repair (45.7%), colon surgery (20.4%), rectal surgery (25.6%), abdominal hysterectomy (16.6%),and vaginal hysterectomy (13.3%).
The increase in reporting facilities in 2011 is partly a result of new state requirements for reporting health care–associated infections to the NHSN (30 states plus the District of Columbia as of December 2012) and from the federal requirement that all hospitals participating in the CMS Hospital Inpatient Quality Reporting Program report these infections to the NHSN.
The authors estimated that each CLABSI occurring in ICU patients cost the CMS approximately $26,000. However, the report did not have information on the insurance status of the patients with CLABSIs, so this figure would not apply to the private insurance patients.
The report was funded by the CDC, and no disclosures were noted.
The CDC report on health care–associated infections is great news. It shows that we have been making significant and substantial progress in the often preventable infections that occur in our hospitals. Reductions of 41% (CLABSI) are very impressive. This is a significant number of patients who did not get infected, receive otherwise unnecessary antibiotics and remain in the hospital longer than necessary. This also represents a significant cost savings. As we strive for improved value for our patients – higher quality care at lower costs – improvements like this are amazing.
One interesting finding is that, while there are reductions in CAUTIs and SSIs, they are not as significant as those with CLABSI. I think part of this has to do with the research into CLABSI and the fact that it lent itself well to the use of protocols and checklists, which are easily adopted by institutions. Peter Pronovost’s 2006 New England Journal of Medicine study detailed the 66% reduction in CLABSI throughout Michigan ICUs via the use of a simple checklist. SSIs also lend themselves to "protocol-ization." CAUTIs are slightly more difficult because a different human factor is introduced – the convenience and wishes of the patient. We need to continue educating our patients about CAUTIs and developing protocols that make the early removal of catheters the norm rather than the exception.
Physicians should be proud of their efforts in reducing health care–associated infections. We need to continue working hard to sustain these gains and identify other areas where similar interventions will yield positive outcomes. Sustained education and intervention will get us close to the HHS goals by the end of 2013, if not achieve them outright. One simple method of preventing health care–associated infections is to (a) implement a standardized checklist of proven steps to reduce said infections, and (b) empower members of the health care team to stop the provider when those steps are not being followed. A team approach, both in the development and implementation of these protocols, is essential to initial and sustained success.
Dr. Michael Pistoria is an internal medicine specialist and hospitalist at Allentown Hospital and Bethlehem Hospital in New Jersey. He is a senior fellow of the Society of Hospital Medicine and served as lead editor of the publication "Core Competencies in Hospital Medicine," which defined hospitalists’ roles. He made these comments in an e-mail interview with this news organization.
The CDC report on health care–associated infections is great news. It shows that we have been making significant and substantial progress in the often preventable infections that occur in our hospitals. Reductions of 41% (CLABSI) are very impressive. This is a significant number of patients who did not get infected, receive otherwise unnecessary antibiotics and remain in the hospital longer than necessary. This also represents a significant cost savings. As we strive for improved value for our patients – higher quality care at lower costs – improvements like this are amazing.
One interesting finding is that, while there are reductions in CAUTIs and SSIs, they are not as significant as those with CLABSI. I think part of this has to do with the research into CLABSI and the fact that it lent itself well to the use of protocols and checklists, which are easily adopted by institutions. Peter Pronovost’s 2006 New England Journal of Medicine study detailed the 66% reduction in CLABSI throughout Michigan ICUs via the use of a simple checklist. SSIs also lend themselves to "protocol-ization." CAUTIs are slightly more difficult because a different human factor is introduced – the convenience and wishes of the patient. We need to continue educating our patients about CAUTIs and developing protocols that make the early removal of catheters the norm rather than the exception.
Physicians should be proud of their efforts in reducing health care–associated infections. We need to continue working hard to sustain these gains and identify other areas where similar interventions will yield positive outcomes. Sustained education and intervention will get us close to the HHS goals by the end of 2013, if not achieve them outright. One simple method of preventing health care–associated infections is to (a) implement a standardized checklist of proven steps to reduce said infections, and (b) empower members of the health care team to stop the provider when those steps are not being followed. A team approach, both in the development and implementation of these protocols, is essential to initial and sustained success.
Dr. Michael Pistoria is an internal medicine specialist and hospitalist at Allentown Hospital and Bethlehem Hospital in New Jersey. He is a senior fellow of the Society of Hospital Medicine and served as lead editor of the publication "Core Competencies in Hospital Medicine," which defined hospitalists’ roles. He made these comments in an e-mail interview with this news organization.
The CDC report on health care–associated infections is great news. It shows that we have been making significant and substantial progress in the often preventable infections that occur in our hospitals. Reductions of 41% (CLABSI) are very impressive. This is a significant number of patients who did not get infected, receive otherwise unnecessary antibiotics and remain in the hospital longer than necessary. This also represents a significant cost savings. As we strive for improved value for our patients – higher quality care at lower costs – improvements like this are amazing.
One interesting finding is that, while there are reductions in CAUTIs and SSIs, they are not as significant as those with CLABSI. I think part of this has to do with the research into CLABSI and the fact that it lent itself well to the use of protocols and checklists, which are easily adopted by institutions. Peter Pronovost’s 2006 New England Journal of Medicine study detailed the 66% reduction in CLABSI throughout Michigan ICUs via the use of a simple checklist. SSIs also lend themselves to "protocol-ization." CAUTIs are slightly more difficult because a different human factor is introduced – the convenience and wishes of the patient. We need to continue educating our patients about CAUTIs and developing protocols that make the early removal of catheters the norm rather than the exception.
Physicians should be proud of their efforts in reducing health care–associated infections. We need to continue working hard to sustain these gains and identify other areas where similar interventions will yield positive outcomes. Sustained education and intervention will get us close to the HHS goals by the end of 2013, if not achieve them outright. One simple method of preventing health care–associated infections is to (a) implement a standardized checklist of proven steps to reduce said infections, and (b) empower members of the health care team to stop the provider when those steps are not being followed. A team approach, both in the development and implementation of these protocols, is essential to initial and sustained success.
Dr. Michael Pistoria is an internal medicine specialist and hospitalist at Allentown Hospital and Bethlehem Hospital in New Jersey. He is a senior fellow of the Society of Hospital Medicine and served as lead editor of the publication "Core Competencies in Hospital Medicine," which defined hospitalists’ roles. He made these comments in an e-mail interview with this news organization.
The rates of three major types of health care–associated infections have continued to decrease in U.S. hospitals, according to a new report from the Centers for Disease Control and Prevention.
The rate of central line–associated bloodstream infections (CLABSI) is down nationally by 41%, catheter-associated urinary tract infections (CAUTI) are down by 7%, and surgical site infections (SSI) for a combined 10 surgical procedures are down by 17%.
These declines are measured against the 2008 baseline rates of CLABSIs, CAUTIs and SSIs reported when the U.S. Department of Health and Human Services established its 5-year goals for reducing health care–associated infections by the end of 2013. The HHS goals include reducing CLABSIs by 50%, CAUTIs by 25%, and SSIs by 25%. The American College of Surgeons and the CDC have partnered to develop the means to report, measure, and prevent health care–associated infections, and the ACS has been instrumental in collecting and submitting standard SSI measure data and other data to the CDC’s National Healthcare Safety Network (NHSN) and the ACS’s National Surgical Quality Improvement Program (NSQIP).
“One thing these numbers show us is the complexity of achieving im provement,” said Dr. Clifford Y. Ko, MD, MS, FACS, Director of the American College of Surgeons (ACS) Division of Research and Optimal Patient Care. “The College’s recent effort with the Joint Commission Center for Transforming Healthcare to reduce SSI has shown us that SSIs are very multifactorial, and not every provider or facility has the same is sues to address.
“Similarly, even the measurement and analytical techniques used in this study can be improved upon,” Dr. Ko added. “While better than they used to be, we know through ACS NSQIP®[the College’s National Surgical Quality Improvement Program] that we can measure and feedback risk-adjusted infection rates on all procedures, not just 10. This is important for gaining traction with all providers because it will likely require the effort of all providers to achieve system- wide, sustained improvement.”
Paul J. Malpiedi and associates at the CDC reported the findings in the 2011 National and State Healthcare-Associated Infections Standard Infection Ratio Report. Mr. Malpiedi’s team compared the standard infection ratios (SIRs) between 2010 and 2011 to determine progress in preventing health care–associated infections.
Standard infection ratios developed at the national, state, and facility levels compare the number of infections that actually occurred to the number that would be expected based on the referent years: 2008 for CLABSIs and SSIs, and 2009 for CAUTIs. The standard infection ratios were adjusted to account for hospital type, hospital size (based on bed number), and hospital affiliation with a medical school.
Mr. Malpiedi’s team analyzed the data reported for the 2011 calendar year to the NHSN from 3,472 facilities for CLABSIs, 1,807 facilities for CAUTIs and 2,130 facilities for SSIs, based on reports submitted through Sept. 4, 2012. Non–acute care hospitals, outpatient dialysis facilities, inpatient dialysis wards, long-term care facilities, and outpatient surgical settings were excluded from the analysis.
A total of 18,113 CLABSIs were reported during 2011, compared with 30,617 that were predicted to occur based on the 2008 referent population, for an SIR of 0.592. This 41% reduction is an improvement over the 2010 reduction of 32%. With a median SIR of 0.469, half the reporting facilities in 2011 had reduced their CLABSIs by 53%. The lowest rate of CLABSIs was reported in ICUs, where the infections had declined 44% since 2008.
The number of facilities reporting infection data increased from 2010 to 2011. The 3,472 facilities in 50 states and the District of Columbia that reported data for CLABSIs represented a 55% increase from those reporting in 2010. The 2011 data came from 12,122 patient care locations, which included 5,722 ICUs (47%), 5,436 wards (45%) and 946 NICUs (8%).
The overall reduction in CAUTIs was less substantial, with no significant overall change since 2010. The 7% reduction in CAUTIs, with an SIR of 0.93, came from 14,315 reported CAUTIs, compared to the 15,398 predicted infections based on the 2009 referent population. Specifically, CAUTIs in wards declined about 15% while the infection rates in ICUs remained unchanged.
When only the 550 facilities that reported in both 2010 and 2011 were included in the analysis, the reduction since 2010 was statistically significant. A total of 1,807 facilities in 50 states and the District of Columbia reported CAUTI data, an 84% increase from the 2010 number of 981 reporting facilities. The 6,402 patient care locations included in the CAUTI data came from 2,633 ICUs (41%) and 3,769 wards (59%).
A total of 2,130 facilities from 48 states and the District of Columbia reported SSI data. Among the 748,192 surgical procedures included were 6,357 deep incisional and organ/space infections occurred, compared to the 7,683 SSIs that were predicted using the 2008 baseline, for an SIR of 0.827 .
This lower SIR represents a 17% decline in SSIs since 2008. SSIs declined for hip arthroplasty (10.4% decline), knee arthroplasty (14.3%), coronary artery bypass graft (22.1%), cardiac surgery (30.2%), peripheral vascular bypass surgery (25.5%), abdominal aortic aneurysm repair (45.7%), colon surgery (20.4%), rectal surgery (25.6%), abdominal hysterectomy (16.6%),and vaginal hysterectomy (13.3%).
The increase in reporting facilities in 2011 is partly a result of new state requirements for reporting health care–associated infections to the NHSN (30 states plus the District of Columbia as of December 2012) and from the federal requirement that all hospitals participating in the CMS Hospital Inpatient Quality Reporting Program report these infections to the NHSN.
The authors estimated that each CLABSI occurring in ICU patients cost the CMS approximately $26,000. However, the report did not have information on the insurance status of the patients with CLABSIs, so this figure would not apply to the private insurance patients.
The report was funded by the CDC, and no disclosures were noted.
The rates of three major types of health care–associated infections have continued to decrease in U.S. hospitals, according to a new report from the Centers for Disease Control and Prevention.
The rate of central line–associated bloodstream infections (CLABSI) is down nationally by 41%, catheter-associated urinary tract infections (CAUTI) are down by 7%, and surgical site infections (SSI) for a combined 10 surgical procedures are down by 17%.
These declines are measured against the 2008 baseline rates of CLABSIs, CAUTIs and SSIs reported when the U.S. Department of Health and Human Services established its 5-year goals for reducing health care–associated infections by the end of 2013. The HHS goals include reducing CLABSIs by 50%, CAUTIs by 25%, and SSIs by 25%. The American College of Surgeons and the CDC have partnered to develop the means to report, measure, and prevent health care–associated infections, and the ACS has been instrumental in collecting and submitting standard SSI measure data and other data to the CDC’s National Healthcare Safety Network (NHSN) and the ACS’s National Surgical Quality Improvement Program (NSQIP).
“One thing these numbers show us is the complexity of achieving im provement,” said Dr. Clifford Y. Ko, MD, MS, FACS, Director of the American College of Surgeons (ACS) Division of Research and Optimal Patient Care. “The College’s recent effort with the Joint Commission Center for Transforming Healthcare to reduce SSI has shown us that SSIs are very multifactorial, and not every provider or facility has the same is sues to address.
“Similarly, even the measurement and analytical techniques used in this study can be improved upon,” Dr. Ko added. “While better than they used to be, we know through ACS NSQIP®[the College’s National Surgical Quality Improvement Program] that we can measure and feedback risk-adjusted infection rates on all procedures, not just 10. This is important for gaining traction with all providers because it will likely require the effort of all providers to achieve system- wide, sustained improvement.”
Paul J. Malpiedi and associates at the CDC reported the findings in the 2011 National and State Healthcare-Associated Infections Standard Infection Ratio Report. Mr. Malpiedi’s team compared the standard infection ratios (SIRs) between 2010 and 2011 to determine progress in preventing health care–associated infections.
Standard infection ratios developed at the national, state, and facility levels compare the number of infections that actually occurred to the number that would be expected based on the referent years: 2008 for CLABSIs and SSIs, and 2009 for CAUTIs. The standard infection ratios were adjusted to account for hospital type, hospital size (based on bed number), and hospital affiliation with a medical school.
Mr. Malpiedi’s team analyzed the data reported for the 2011 calendar year to the NHSN from 3,472 facilities for CLABSIs, 1,807 facilities for CAUTIs and 2,130 facilities for SSIs, based on reports submitted through Sept. 4, 2012. Non–acute care hospitals, outpatient dialysis facilities, inpatient dialysis wards, long-term care facilities, and outpatient surgical settings were excluded from the analysis.
A total of 18,113 CLABSIs were reported during 2011, compared with 30,617 that were predicted to occur based on the 2008 referent population, for an SIR of 0.592. This 41% reduction is an improvement over the 2010 reduction of 32%. With a median SIR of 0.469, half the reporting facilities in 2011 had reduced their CLABSIs by 53%. The lowest rate of CLABSIs was reported in ICUs, where the infections had declined 44% since 2008.
The number of facilities reporting infection data increased from 2010 to 2011. The 3,472 facilities in 50 states and the District of Columbia that reported data for CLABSIs represented a 55% increase from those reporting in 2010. The 2011 data came from 12,122 patient care locations, which included 5,722 ICUs (47%), 5,436 wards (45%) and 946 NICUs (8%).
The overall reduction in CAUTIs was less substantial, with no significant overall change since 2010. The 7% reduction in CAUTIs, with an SIR of 0.93, came from 14,315 reported CAUTIs, compared to the 15,398 predicted infections based on the 2009 referent population. Specifically, CAUTIs in wards declined about 15% while the infection rates in ICUs remained unchanged.
When only the 550 facilities that reported in both 2010 and 2011 were included in the analysis, the reduction since 2010 was statistically significant. A total of 1,807 facilities in 50 states and the District of Columbia reported CAUTI data, an 84% increase from the 2010 number of 981 reporting facilities. The 6,402 patient care locations included in the CAUTI data came from 2,633 ICUs (41%) and 3,769 wards (59%).
A total of 2,130 facilities from 48 states and the District of Columbia reported SSI data. Among the 748,192 surgical procedures included were 6,357 deep incisional and organ/space infections occurred, compared to the 7,683 SSIs that were predicted using the 2008 baseline, for an SIR of 0.827 .
This lower SIR represents a 17% decline in SSIs since 2008. SSIs declined for hip arthroplasty (10.4% decline), knee arthroplasty (14.3%), coronary artery bypass graft (22.1%), cardiac surgery (30.2%), peripheral vascular bypass surgery (25.5%), abdominal aortic aneurysm repair (45.7%), colon surgery (20.4%), rectal surgery (25.6%), abdominal hysterectomy (16.6%),and vaginal hysterectomy (13.3%).
The increase in reporting facilities in 2011 is partly a result of new state requirements for reporting health care–associated infections to the NHSN (30 states plus the District of Columbia as of December 2012) and from the federal requirement that all hospitals participating in the CMS Hospital Inpatient Quality Reporting Program report these infections to the NHSN.
The authors estimated that each CLABSI occurring in ICU patients cost the CMS approximately $26,000. However, the report did not have information on the insurance status of the patients with CLABSIs, so this figure would not apply to the private insurance patients.
The report was funded by the CDC, and no disclosures were noted.
Major Finding: Since 2008, central line-associated bloodstream infections in facilities from all 50 states and the District of Columbia have declined 41% and surgical site infections have declined 17%, with catheter-associated urinary tract infections showing a decline of 7% since 2009.
Data Source: The data come from 3,472 facilities’ reports for central line-associated bloodstream infections, 1,807 facilities’ reports for catheter-associated urinary tract infections and 2,130 facilities’ reports for surgical site infections, which occurred during the 2011 calendar year, included in reports submitted up through Sept. 4, 2012.
Disclosures: The study was funded by the U.S. Centers for Disease Control and Prevention. No disclosures were noted.
Direct peritoneal resuscitation beneficial after damage control surgery
SCOTTSDALE, ARIZ. – Direct peritoneal resuscitation with 2.5% Delflex solution significantly reduced abdominal closure time and operative complications in patients with hemorrhagic shock undergoing damage control surgery, a prospective case-control study showed.
The time to definitive wound closure fell from 5.7 days with standard resuscitation to 3.6 days with the addition of direct peritoneal resuscitation (DPR) with 2.5% Delflex solution (P = .006).
In addition, the control group was 3.2 times more likely to develop an intra-abdominal complication than was the DPR group (odds ratio, 3.2; P = .03), Dr. Jason W. Smith reported at the annual scientific assembly of the Eastern Association for the Surgery of Trauma..
"DPR is a safe, simple, and inexpensive method of managing an open abdomen following damage control surgery," he said. "DPR may attenuate persistent hepatic injury following hemorrhagic shock."
Dr. Smith and his colleagues at the University of Louisville (Ky.) have been strong proponents of the technique, and previously reported that DPR with Delflex shortened abdominal closure by 2.7 days in a retrospective proof-of-concept study in 19 trauma patients (J. Am. Coll. Surg. 2010;210:658-67).
Vasodilators and heparin can be used to augment microvascular circulation and prevent blood clots, but in patients with hemorrhagic shock, this generally leads to bad results. Activated drotrecogin alfa (Xigris), which was pulled from the market in October 2011, was initially used, but the sepsis drug treats only the symptoms, Dr. Smith explained.
"We selected Delflex because of its ability to have an effect on microvascular permeability as well as microvascular blood flow," he said. "So really this is a new take on what is a very old medicine."
Between 2008 and 2011, the investigators prospectively enrolled 42 patients who underwent damage control surgery and conventional resuscitation plus DPR consisting of peritoneal lavage with an 800-mL bolus of 2.5% Delflex for the first hour and a 400-mL/hr infusion thereafter. A 19 Fr drain was placed along the root of the mesentery into the pelvis, and suction dressing was applied to drain fluid. The infusion was stopped when the abdomen was closed definitively either by primary closure or by the use of an absorbable or a biologic mesh.
The patients (average age, 30.6 years) were propensity matched against 42 case controls (average age, 29.1 years) who had undergone conventional resuscitation at the discretion of the treating surgeon after damage control surgery. All patients underwent blood product resuscitation in the first 24-48 hours of treatment.
The average Injury Severity Score was 19 among patients and 21 among controls. The hepatic Abbreviated Injury Scale (AIS) score was 3.4 in both groups, and roughly 40% of patients underwent a liver operation.
Control patients had significantly elevated alanine transaminase levels at 48 (731 vs. 507 U/L) and 72 hours (791 vs. 501 U/L) post resuscitation, compared with DPR patients, and this difference continued up to 14 days after the initial injury (all P less than .01), Dr. Smith said.
International normalized ratio (INR) levels were persistently higher in the control group, reaching statistical significance at 72 hours (1.3 vs. 1.8; P = .03). Time to INR normalization was no different whether the red blood cell to fresh frozen plasma (FFP) ratio was 1:1 or 1:2, he said.
Controls achieved a slightly better transfusion ratio (1.57 vs. 1.76; P = .52), although DPR patients had significantly lower FFP requirements after 48 hours (P = .04), and received significantly fewer transfusions at 72 hours post admission (31 vs. 41; P = .007).
As observed in the pilot study, the primary fascial closure rate was significantly higher with DPR than conventional resuscitation alone (88% vs. 66%; P = .01), and mortality was similar (10% vs. 17%; P = .25), Dr. Smith said. There were no complications with DPR administration.
In a multivariate analysis, the two strongest predictors of persistent hepatic injury following hemorrhagic shock were hepatic AIS greater than 3 (OR, 3.18), followed by nonutilization of DPR (OR, 0.63).
Invited discussant Dr. Greta Piper of Yale University, New Haven, Conn., asked whether the results would have been even better with a higher infusion rate or concentration of Delflex since one effect of the infusion is continuous dialysis, with a potential reduction in total body salt and water content.
Dr. Smith replied that they have tried DPR with other agents such as mannitol and Delflex 1.5% and 4.25%, but said the 2.5% concentration "is kind of a Goldilocks dose," as 1.5% is too weak to do anything and 4.25% desiccates the intravascular space.
"The 2.5% doesn’t desiccate the intravascular space, but you still get all the benefits of microvascular augmentation," he added.
Dr. Smith noted that DPR with 2.5% Delflex has produced similar results in patients undergoing emergency surgery as well as in those with intestinal obstruction and significant bowel edema. The researchers are currently enrolling adult trauma patients into a prospective randomized trial that will evaluate changes in hepatic blood flow as well as the effect of DPR on the inflammatory process following hemorrhagic shock.
The National Institute of General Medicine Sciences supported this research. Dr. Smith and his coauthors reported no relevant financial disclosures.
SCOTTSDALE, ARIZ. – Direct peritoneal resuscitation with 2.5% Delflex solution significantly reduced abdominal closure time and operative complications in patients with hemorrhagic shock undergoing damage control surgery, a prospective case-control study showed.
The time to definitive wound closure fell from 5.7 days with standard resuscitation to 3.6 days with the addition of direct peritoneal resuscitation (DPR) with 2.5% Delflex solution (P = .006).
In addition, the control group was 3.2 times more likely to develop an intra-abdominal complication than was the DPR group (odds ratio, 3.2; P = .03), Dr. Jason W. Smith reported at the annual scientific assembly of the Eastern Association for the Surgery of Trauma..
"DPR is a safe, simple, and inexpensive method of managing an open abdomen following damage control surgery," he said. "DPR may attenuate persistent hepatic injury following hemorrhagic shock."
Dr. Smith and his colleagues at the University of Louisville (Ky.) have been strong proponents of the technique, and previously reported that DPR with Delflex shortened abdominal closure by 2.7 days in a retrospective proof-of-concept study in 19 trauma patients (J. Am. Coll. Surg. 2010;210:658-67).
Vasodilators and heparin can be used to augment microvascular circulation and prevent blood clots, but in patients with hemorrhagic shock, this generally leads to bad results. Activated drotrecogin alfa (Xigris), which was pulled from the market in October 2011, was initially used, but the sepsis drug treats only the symptoms, Dr. Smith explained.
"We selected Delflex because of its ability to have an effect on microvascular permeability as well as microvascular blood flow," he said. "So really this is a new take on what is a very old medicine."
Between 2008 and 2011, the investigators prospectively enrolled 42 patients who underwent damage control surgery and conventional resuscitation plus DPR consisting of peritoneal lavage with an 800-mL bolus of 2.5% Delflex for the first hour and a 400-mL/hr infusion thereafter. A 19 Fr drain was placed along the root of the mesentery into the pelvis, and suction dressing was applied to drain fluid. The infusion was stopped when the abdomen was closed definitively either by primary closure or by the use of an absorbable or a biologic mesh.
The patients (average age, 30.6 years) were propensity matched against 42 case controls (average age, 29.1 years) who had undergone conventional resuscitation at the discretion of the treating surgeon after damage control surgery. All patients underwent blood product resuscitation in the first 24-48 hours of treatment.
The average Injury Severity Score was 19 among patients and 21 among controls. The hepatic Abbreviated Injury Scale (AIS) score was 3.4 in both groups, and roughly 40% of patients underwent a liver operation.
Control patients had significantly elevated alanine transaminase levels at 48 (731 vs. 507 U/L) and 72 hours (791 vs. 501 U/L) post resuscitation, compared with DPR patients, and this difference continued up to 14 days after the initial injury (all P less than .01), Dr. Smith said.
International normalized ratio (INR) levels were persistently higher in the control group, reaching statistical significance at 72 hours (1.3 vs. 1.8; P = .03). Time to INR normalization was no different whether the red blood cell to fresh frozen plasma (FFP) ratio was 1:1 or 1:2, he said.
Controls achieved a slightly better transfusion ratio (1.57 vs. 1.76; P = .52), although DPR patients had significantly lower FFP requirements after 48 hours (P = .04), and received significantly fewer transfusions at 72 hours post admission (31 vs. 41; P = .007).
As observed in the pilot study, the primary fascial closure rate was significantly higher with DPR than conventional resuscitation alone (88% vs. 66%; P = .01), and mortality was similar (10% vs. 17%; P = .25), Dr. Smith said. There were no complications with DPR administration.
In a multivariate analysis, the two strongest predictors of persistent hepatic injury following hemorrhagic shock were hepatic AIS greater than 3 (OR, 3.18), followed by nonutilization of DPR (OR, 0.63).
Invited discussant Dr. Greta Piper of Yale University, New Haven, Conn., asked whether the results would have been even better with a higher infusion rate or concentration of Delflex since one effect of the infusion is continuous dialysis, with a potential reduction in total body salt and water content.
Dr. Smith replied that they have tried DPR with other agents such as mannitol and Delflex 1.5% and 4.25%, but said the 2.5% concentration "is kind of a Goldilocks dose," as 1.5% is too weak to do anything and 4.25% desiccates the intravascular space.
"The 2.5% doesn’t desiccate the intravascular space, but you still get all the benefits of microvascular augmentation," he added.
Dr. Smith noted that DPR with 2.5% Delflex has produced similar results in patients undergoing emergency surgery as well as in those with intestinal obstruction and significant bowel edema. The researchers are currently enrolling adult trauma patients into a prospective randomized trial that will evaluate changes in hepatic blood flow as well as the effect of DPR on the inflammatory process following hemorrhagic shock.
The National Institute of General Medicine Sciences supported this research. Dr. Smith and his coauthors reported no relevant financial disclosures.
SCOTTSDALE, ARIZ. – Direct peritoneal resuscitation with 2.5% Delflex solution significantly reduced abdominal closure time and operative complications in patients with hemorrhagic shock undergoing damage control surgery, a prospective case-control study showed.
The time to definitive wound closure fell from 5.7 days with standard resuscitation to 3.6 days with the addition of direct peritoneal resuscitation (DPR) with 2.5% Delflex solution (P = .006).
In addition, the control group was 3.2 times more likely to develop an intra-abdominal complication than was the DPR group (odds ratio, 3.2; P = .03), Dr. Jason W. Smith reported at the annual scientific assembly of the Eastern Association for the Surgery of Trauma..
"DPR is a safe, simple, and inexpensive method of managing an open abdomen following damage control surgery," he said. "DPR may attenuate persistent hepatic injury following hemorrhagic shock."
Dr. Smith and his colleagues at the University of Louisville (Ky.) have been strong proponents of the technique, and previously reported that DPR with Delflex shortened abdominal closure by 2.7 days in a retrospective proof-of-concept study in 19 trauma patients (J. Am. Coll. Surg. 2010;210:658-67).
Vasodilators and heparin can be used to augment microvascular circulation and prevent blood clots, but in patients with hemorrhagic shock, this generally leads to bad results. Activated drotrecogin alfa (Xigris), which was pulled from the market in October 2011, was initially used, but the sepsis drug treats only the symptoms, Dr. Smith explained.
"We selected Delflex because of its ability to have an effect on microvascular permeability as well as microvascular blood flow," he said. "So really this is a new take on what is a very old medicine."
Between 2008 and 2011, the investigators prospectively enrolled 42 patients who underwent damage control surgery and conventional resuscitation plus DPR consisting of peritoneal lavage with an 800-mL bolus of 2.5% Delflex for the first hour and a 400-mL/hr infusion thereafter. A 19 Fr drain was placed along the root of the mesentery into the pelvis, and suction dressing was applied to drain fluid. The infusion was stopped when the abdomen was closed definitively either by primary closure or by the use of an absorbable or a biologic mesh.
The patients (average age, 30.6 years) were propensity matched against 42 case controls (average age, 29.1 years) who had undergone conventional resuscitation at the discretion of the treating surgeon after damage control surgery. All patients underwent blood product resuscitation in the first 24-48 hours of treatment.
The average Injury Severity Score was 19 among patients and 21 among controls. The hepatic Abbreviated Injury Scale (AIS) score was 3.4 in both groups, and roughly 40% of patients underwent a liver operation.
Control patients had significantly elevated alanine transaminase levels at 48 (731 vs. 507 U/L) and 72 hours (791 vs. 501 U/L) post resuscitation, compared with DPR patients, and this difference continued up to 14 days after the initial injury (all P less than .01), Dr. Smith said.
International normalized ratio (INR) levels were persistently higher in the control group, reaching statistical significance at 72 hours (1.3 vs. 1.8; P = .03). Time to INR normalization was no different whether the red blood cell to fresh frozen plasma (FFP) ratio was 1:1 or 1:2, he said.
Controls achieved a slightly better transfusion ratio (1.57 vs. 1.76; P = .52), although DPR patients had significantly lower FFP requirements after 48 hours (P = .04), and received significantly fewer transfusions at 72 hours post admission (31 vs. 41; P = .007).
As observed in the pilot study, the primary fascial closure rate was significantly higher with DPR than conventional resuscitation alone (88% vs. 66%; P = .01), and mortality was similar (10% vs. 17%; P = .25), Dr. Smith said. There were no complications with DPR administration.
In a multivariate analysis, the two strongest predictors of persistent hepatic injury following hemorrhagic shock were hepatic AIS greater than 3 (OR, 3.18), followed by nonutilization of DPR (OR, 0.63).
Invited discussant Dr. Greta Piper of Yale University, New Haven, Conn., asked whether the results would have been even better with a higher infusion rate or concentration of Delflex since one effect of the infusion is continuous dialysis, with a potential reduction in total body salt and water content.
Dr. Smith replied that they have tried DPR with other agents such as mannitol and Delflex 1.5% and 4.25%, but said the 2.5% concentration "is kind of a Goldilocks dose," as 1.5% is too weak to do anything and 4.25% desiccates the intravascular space.
"The 2.5% doesn’t desiccate the intravascular space, but you still get all the benefits of microvascular augmentation," he added.
Dr. Smith noted that DPR with 2.5% Delflex has produced similar results in patients undergoing emergency surgery as well as in those with intestinal obstruction and significant bowel edema. The researchers are currently enrolling adult trauma patients into a prospective randomized trial that will evaluate changes in hepatic blood flow as well as the effect of DPR on the inflammatory process following hemorrhagic shock.
The National Institute of General Medicine Sciences supported this research. Dr. Smith and his coauthors reported no relevant financial disclosures.
AT THE EAST SCIENTIFIC ASSEMBLY
Major Finding: Time to definitive wound closure was 5.7 days with standard resuscitation vs. 3.6 days with the addition of direct peritoneal resuscitation with 2.5% Delflex (P = .006).
Data Source: Prospective, case-control study in 84 patients with hemorrhagic shock undergoing damage control surgery.
Disclosures: The National Institute of General Medicine Sciences supported this research. Dr. Smith and his coauthors reported no relevant financial disclosures.
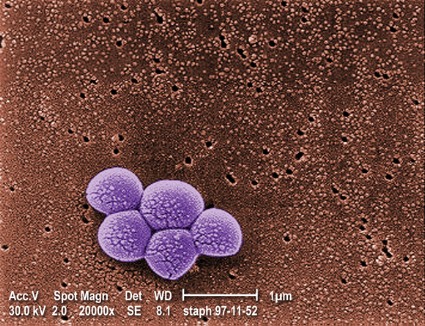
Daily chlorhexidine bath cuts bloodstream, MRSA infections in ICUs
For patients in intensive care units and bone marrow transplantation units, daily bathing with chlorhexidine-impregnated washcloths significantly reduced the incidence of bloodstream infections and decreased the acquisition of multidrug-resistant organisms in a randomized controlled trial reported online Feb. 7 in the New England Journal of Medicine.
The multicenter study, which included nine medical, coronary care, surgical, cardiac surgery, and bone marrow transplantation units in several geographic regions of the United States, confirms the results of previous single-center studies and demonstrates that the benefit of daily chlorhexidine bathing is widely generalizable, said Dr. Michael W. Climo of the Hunter Holmes McGuire Veterans Affairs Medical Center, Richmond, Va., and his associates.
Each hospital unit participating in the trial was randomized to perform daily bathing of patients using either washcloths impregnated with 2% chlorhexidine gluconate (intervention) or washcloths with no impregnated antimicrobial (control) for 6 months, followed by a crossover period in which patients were bathed daily with the alternate product for another 6 months. A total of 7,727 patients were included in the study.
Nurses were trained in the manufacturer’s instructions for bathing, which involved using the cloths in a sequential order to rinse all body surfaces except the face (to avoid getting chlorhexidine in the patient’s eyes and mouth). Implementing this cost-effective strategy was "relatively straightforward" because it did not require much of a change from current routine patient-bathing practices, the investigators said.
All the hospital units performed active surveillance for methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant enterococcus (VRE) throughout the study.
There were 119 hospital-acquired bloodstream infections during the intervention and 165 during the control period, representing a 28% reduction with chlorhexidine bathing. The rate of primary bloodstream infections was 31% lower and the rate of central catheter–associated bloodstream infections was 53% lower with the intervention, Dr. Climo and his associates reported (N. Engl. J. Med. 2013 Feb. 6 [doi: 10.1056/NEJMoa1113849]).
This benefit was greatest among patients who had longer lengths of stay in the hospital units.
One "unanticipated" finding was that rates of fungal infection in particular were dramatically reduced with chlorhexidine bathing. The incidence of primary bloodstream infection caused by fungi was 53% lower and the incidence of central catheter–associated fungal bloodstream infection was 90% lower during the intervention period than during the control period.
This antifungal effect has not been reported previously. "If our results are confirmed, topical use of chlorhexidine could be added to strategies to prevent fungal infection," the researchers noted.
The rate of MRSA or VRE acquisition was 23% lower during the intervention period (5.10 cases per 1,000 patient-days) than during the control period (6.60 cases per 1,000 patient-days). However, reductions in the incidence of MRSA and VRE bacteremia did not reach statistical significance, most likely because the overall number of cases was so low, Dr. Climo and his colleagues said.
No serious adverse effects were observed. The incidence of skin reactions was higher with the control washcloths (3.4%) than with the chlorhexidine washcloths (2.0%), and none of the skin reactions were considered to be related to the bathing intervention.
It was reassuring that none of the MRSA or VRE isolates that were detected in this study showed resistance to chlorhexidine. However, the development of resistance to biocides and disinfectants among nosocomial organisms remains "a substantial concern" and should continue to be monitored, they added.
This study was supported by the Centers for Disease Control and Prevention and Sage Products. Sage Products supplied the washcloths, provided technical and educational support, and participated in weekly teleconferences with the research group but was not involved in the study design, data analysis, or reporting of the study results. Dr. Climo and his associates reported ties to Sage Products, Centene, and other companies.
For patients in intensive care units and bone marrow transplantation units, daily bathing with chlorhexidine-impregnated washcloths significantly reduced the incidence of bloodstream infections and decreased the acquisition of multidrug-resistant organisms in a randomized controlled trial reported online Feb. 7 in the New England Journal of Medicine.
The multicenter study, which included nine medical, coronary care, surgical, cardiac surgery, and bone marrow transplantation units in several geographic regions of the United States, confirms the results of previous single-center studies and demonstrates that the benefit of daily chlorhexidine bathing is widely generalizable, said Dr. Michael W. Climo of the Hunter Holmes McGuire Veterans Affairs Medical Center, Richmond, Va., and his associates.
Each hospital unit participating in the trial was randomized to perform daily bathing of patients using either washcloths impregnated with 2% chlorhexidine gluconate (intervention) or washcloths with no impregnated antimicrobial (control) for 6 months, followed by a crossover period in which patients were bathed daily with the alternate product for another 6 months. A total of 7,727 patients were included in the study.
Nurses were trained in the manufacturer’s instructions for bathing, which involved using the cloths in a sequential order to rinse all body surfaces except the face (to avoid getting chlorhexidine in the patient’s eyes and mouth). Implementing this cost-effective strategy was "relatively straightforward" because it did not require much of a change from current routine patient-bathing practices, the investigators said.
All the hospital units performed active surveillance for methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant enterococcus (VRE) throughout the study.
There were 119 hospital-acquired bloodstream infections during the intervention and 165 during the control period, representing a 28% reduction with chlorhexidine bathing. The rate of primary bloodstream infections was 31% lower and the rate of central catheter–associated bloodstream infections was 53% lower with the intervention, Dr. Climo and his associates reported (N. Engl. J. Med. 2013 Feb. 6 [doi: 10.1056/NEJMoa1113849]).
This benefit was greatest among patients who had longer lengths of stay in the hospital units.
One "unanticipated" finding was that rates of fungal infection in particular were dramatically reduced with chlorhexidine bathing. The incidence of primary bloodstream infection caused by fungi was 53% lower and the incidence of central catheter–associated fungal bloodstream infection was 90% lower during the intervention period than during the control period.
This antifungal effect has not been reported previously. "If our results are confirmed, topical use of chlorhexidine could be added to strategies to prevent fungal infection," the researchers noted.
The rate of MRSA or VRE acquisition was 23% lower during the intervention period (5.10 cases per 1,000 patient-days) than during the control period (6.60 cases per 1,000 patient-days). However, reductions in the incidence of MRSA and VRE bacteremia did not reach statistical significance, most likely because the overall number of cases was so low, Dr. Climo and his colleagues said.
No serious adverse effects were observed. The incidence of skin reactions was higher with the control washcloths (3.4%) than with the chlorhexidine washcloths (2.0%), and none of the skin reactions were considered to be related to the bathing intervention.
It was reassuring that none of the MRSA or VRE isolates that were detected in this study showed resistance to chlorhexidine. However, the development of resistance to biocides and disinfectants among nosocomial organisms remains "a substantial concern" and should continue to be monitored, they added.
This study was supported by the Centers for Disease Control and Prevention and Sage Products. Sage Products supplied the washcloths, provided technical and educational support, and participated in weekly teleconferences with the research group but was not involved in the study design, data analysis, or reporting of the study results. Dr. Climo and his associates reported ties to Sage Products, Centene, and other companies.
For patients in intensive care units and bone marrow transplantation units, daily bathing with chlorhexidine-impregnated washcloths significantly reduced the incidence of bloodstream infections and decreased the acquisition of multidrug-resistant organisms in a randomized controlled trial reported online Feb. 7 in the New England Journal of Medicine.
The multicenter study, which included nine medical, coronary care, surgical, cardiac surgery, and bone marrow transplantation units in several geographic regions of the United States, confirms the results of previous single-center studies and demonstrates that the benefit of daily chlorhexidine bathing is widely generalizable, said Dr. Michael W. Climo of the Hunter Holmes McGuire Veterans Affairs Medical Center, Richmond, Va., and his associates.
Each hospital unit participating in the trial was randomized to perform daily bathing of patients using either washcloths impregnated with 2% chlorhexidine gluconate (intervention) or washcloths with no impregnated antimicrobial (control) for 6 months, followed by a crossover period in which patients were bathed daily with the alternate product for another 6 months. A total of 7,727 patients were included in the study.
Nurses were trained in the manufacturer’s instructions for bathing, which involved using the cloths in a sequential order to rinse all body surfaces except the face (to avoid getting chlorhexidine in the patient’s eyes and mouth). Implementing this cost-effective strategy was "relatively straightforward" because it did not require much of a change from current routine patient-bathing practices, the investigators said.
All the hospital units performed active surveillance for methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant enterococcus (VRE) throughout the study.
There were 119 hospital-acquired bloodstream infections during the intervention and 165 during the control period, representing a 28% reduction with chlorhexidine bathing. The rate of primary bloodstream infections was 31% lower and the rate of central catheter–associated bloodstream infections was 53% lower with the intervention, Dr. Climo and his associates reported (N. Engl. J. Med. 2013 Feb. 6 [doi: 10.1056/NEJMoa1113849]).
This benefit was greatest among patients who had longer lengths of stay in the hospital units.
One "unanticipated" finding was that rates of fungal infection in particular were dramatically reduced with chlorhexidine bathing. The incidence of primary bloodstream infection caused by fungi was 53% lower and the incidence of central catheter–associated fungal bloodstream infection was 90% lower during the intervention period than during the control period.
This antifungal effect has not been reported previously. "If our results are confirmed, topical use of chlorhexidine could be added to strategies to prevent fungal infection," the researchers noted.
The rate of MRSA or VRE acquisition was 23% lower during the intervention period (5.10 cases per 1,000 patient-days) than during the control period (6.60 cases per 1,000 patient-days). However, reductions in the incidence of MRSA and VRE bacteremia did not reach statistical significance, most likely because the overall number of cases was so low, Dr. Climo and his colleagues said.
No serious adverse effects were observed. The incidence of skin reactions was higher with the control washcloths (3.4%) than with the chlorhexidine washcloths (2.0%), and none of the skin reactions were considered to be related to the bathing intervention.
It was reassuring that none of the MRSA or VRE isolates that were detected in this study showed resistance to chlorhexidine. However, the development of resistance to biocides and disinfectants among nosocomial organisms remains "a substantial concern" and should continue to be monitored, they added.
This study was supported by the Centers for Disease Control and Prevention and Sage Products. Sage Products supplied the washcloths, provided technical and educational support, and participated in weekly teleconferences with the research group but was not involved in the study design, data analysis, or reporting of the study results. Dr. Climo and his associates reported ties to Sage Products, Centene, and other companies.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: Bathing patients daily with chlorhexidine-impregnated washcloths reduced the rate of bloodstream infections by 28%, the rate of central catheter–associated infections by 53%, the rate of fungal bloodstream infections by 53%, and the rate of MRSA or VRE acquisition by 23%.
Data Source: A multicenter, randomized controlled trial assessing the ability of chlorhexidine bathing of 7,727 intensive care patients to reduce bloodstream infections and the acquisition of resistant organisms.
Disclosures: This study was supported by the Centers for Disease Control and Prevention and Sage Products. Sage Products supplied the washcloths, provided technical and educational support, and participated in weekly teleconferences with the research group but was not involved in the study design, data analysis, or reporting of the study results. Dr. Climo and his associates reported ties to Sage Products, Centene, and other companies.
MAGELLAN trial results support rivaroxaban for DVT prophylaxis
Nearly 2 years after their presentation at a national meeting, the results of MAGELLAN, comparing rivaroxaban to enoxaparin for thromboprophylaxis in critically ill hospitalized patients, have been published online Feb. 6 in the New England Journal of Medicine.
In that large, multicenter trial, rivaroxaban was noninferior to enoxaparin when used for the standard 10-day duration, and superior when used for an extended 35-day duration, for thromboprophylaxis.
However, the rate of clinically relevant bleeding and the incidence of major bleeding events were significantly higher with rivaroxaban than with enoxaparin. This adverse effect apparently balanced out the reduction in venous thromboembolism, as a prespecified analysis of net clinical benefit and harm did not show either one at day 10 or day 35, reported Dr. Alexander T. Cohen and his associates in MAGELLAN (Multicenter, Randomized, Parallel Group Efficacy and Safety Study for the Prevention of Venous Thromboembolism in Hospitalized Acutely Ill Medical Patients Comparing Rivaroxaban with Enoxaparin). The results were first presented by Dr. Cohen, of King's College Hospital, London, at the annual meeting of the American College of Cardiology in spring 2011.
In the 2.5-year study, 8,101 patients at 556 sites in 52 countries were randomized to receive rivaroxaban (4,050 subjects) or enoxaparin (4,051 subjects). Hundreds of patients in both study groups were excluded from the final analyses for various reasons, usually because they could not be assessed for venous thromboembolism using ultrasonography and other imaging techniques in a timely fashion, as required by the study protocol.
At 10 days' follow-up, 2.7% of patients in both groups had a primary outcome event - asymptomatic proximal deep vein thrombosis (DVT), symptomatic nonfatal pulmonary embolism (PE), or thromboembolism-related death - so rivaroxaban met the criteria for noninferiority to enoxaparin.
At the 35-day follow-up, 4.4% of subjects who received extended-duration rivaroxaban and 5.7% of those who received enoxaparin plus placebo had a primary outcome event, so rivaroxaban met the criteria for superiority to enoxaparin, the investigators reported (N. Engl. J. Med. 2013 Feb. 6 [doi: 10.1056/NEJMoa1111096]).
Rivaroxaban (Xarelto)has several indications: for stroke prevention in nonvalvular atrial fibrillation, for the treatment of DVT and PE, for reducing the risk of recurrence of DVT and PE, and for the prophylaxis of DVT in patients undergoing knee or hip replacement surgery. It is not indicated for the condition studied in MAGELLAN, namely prevention of VTE in acutely medically ill patients.
This study was funded by Bayer HealthCare and Janssen, sponsors that also collected and analyzed the study data. Dr. Cohen and his associates reported numerous ties to industry sources.
Nearly 2 years after their presentation at a national meeting, the results of MAGELLAN, comparing rivaroxaban to enoxaparin for thromboprophylaxis in critically ill hospitalized patients, have been published online Feb. 6 in the New England Journal of Medicine.
In that large, multicenter trial, rivaroxaban was noninferior to enoxaparin when used for the standard 10-day duration, and superior when used for an extended 35-day duration, for thromboprophylaxis.
However, the rate of clinically relevant bleeding and the incidence of major bleeding events were significantly higher with rivaroxaban than with enoxaparin. This adverse effect apparently balanced out the reduction in venous thromboembolism, as a prespecified analysis of net clinical benefit and harm did not show either one at day 10 or day 35, reported Dr. Alexander T. Cohen and his associates in MAGELLAN (Multicenter, Randomized, Parallel Group Efficacy and Safety Study for the Prevention of Venous Thromboembolism in Hospitalized Acutely Ill Medical Patients Comparing Rivaroxaban with Enoxaparin). The results were first presented by Dr. Cohen, of King's College Hospital, London, at the annual meeting of the American College of Cardiology in spring 2011.
In the 2.5-year study, 8,101 patients at 556 sites in 52 countries were randomized to receive rivaroxaban (4,050 subjects) or enoxaparin (4,051 subjects). Hundreds of patients in both study groups were excluded from the final analyses for various reasons, usually because they could not be assessed for venous thromboembolism using ultrasonography and other imaging techniques in a timely fashion, as required by the study protocol.
At 10 days' follow-up, 2.7% of patients in both groups had a primary outcome event - asymptomatic proximal deep vein thrombosis (DVT), symptomatic nonfatal pulmonary embolism (PE), or thromboembolism-related death - so rivaroxaban met the criteria for noninferiority to enoxaparin.
At the 35-day follow-up, 4.4% of subjects who received extended-duration rivaroxaban and 5.7% of those who received enoxaparin plus placebo had a primary outcome event, so rivaroxaban met the criteria for superiority to enoxaparin, the investigators reported (N. Engl. J. Med. 2013 Feb. 6 [doi: 10.1056/NEJMoa1111096]).
Rivaroxaban (Xarelto)has several indications: for stroke prevention in nonvalvular atrial fibrillation, for the treatment of DVT and PE, for reducing the risk of recurrence of DVT and PE, and for the prophylaxis of DVT in patients undergoing knee or hip replacement surgery. It is not indicated for the condition studied in MAGELLAN, namely prevention of VTE in acutely medically ill patients.
This study was funded by Bayer HealthCare and Janssen, sponsors that also collected and analyzed the study data. Dr. Cohen and his associates reported numerous ties to industry sources.
Nearly 2 years after their presentation at a national meeting, the results of MAGELLAN, comparing rivaroxaban to enoxaparin for thromboprophylaxis in critically ill hospitalized patients, have been published online Feb. 6 in the New England Journal of Medicine.
In that large, multicenter trial, rivaroxaban was noninferior to enoxaparin when used for the standard 10-day duration, and superior when used for an extended 35-day duration, for thromboprophylaxis.
However, the rate of clinically relevant bleeding and the incidence of major bleeding events were significantly higher with rivaroxaban than with enoxaparin. This adverse effect apparently balanced out the reduction in venous thromboembolism, as a prespecified analysis of net clinical benefit and harm did not show either one at day 10 or day 35, reported Dr. Alexander T. Cohen and his associates in MAGELLAN (Multicenter, Randomized, Parallel Group Efficacy and Safety Study for the Prevention of Venous Thromboembolism in Hospitalized Acutely Ill Medical Patients Comparing Rivaroxaban with Enoxaparin). The results were first presented by Dr. Cohen, of King's College Hospital, London, at the annual meeting of the American College of Cardiology in spring 2011.
In the 2.5-year study, 8,101 patients at 556 sites in 52 countries were randomized to receive rivaroxaban (4,050 subjects) or enoxaparin (4,051 subjects). Hundreds of patients in both study groups were excluded from the final analyses for various reasons, usually because they could not be assessed for venous thromboembolism using ultrasonography and other imaging techniques in a timely fashion, as required by the study protocol.
At 10 days' follow-up, 2.7% of patients in both groups had a primary outcome event - asymptomatic proximal deep vein thrombosis (DVT), symptomatic nonfatal pulmonary embolism (PE), or thromboembolism-related death - so rivaroxaban met the criteria for noninferiority to enoxaparin.
At the 35-day follow-up, 4.4% of subjects who received extended-duration rivaroxaban and 5.7% of those who received enoxaparin plus placebo had a primary outcome event, so rivaroxaban met the criteria for superiority to enoxaparin, the investigators reported (N. Engl. J. Med. 2013 Feb. 6 [doi: 10.1056/NEJMoa1111096]).
Rivaroxaban (Xarelto)has several indications: for stroke prevention in nonvalvular atrial fibrillation, for the treatment of DVT and PE, for reducing the risk of recurrence of DVT and PE, and for the prophylaxis of DVT in patients undergoing knee or hip replacement surgery. It is not indicated for the condition studied in MAGELLAN, namely prevention of VTE in acutely medically ill patients.
This study was funded by Bayer HealthCare and Janssen, sponsors that also collected and analyzed the study data. Dr. Cohen and his associates reported numerous ties to industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: At 10-day follow-up, 2.7% of both study groups
had a thromboembolic event; at 35-day follow-up, 4.4% of subjects who received
rivaroxaban and 5.7% of those who received enoxaparin had a thromboembolic
event.
Data Source: A randomized, double-blind trial in 5,931 inpatients
with acute medical illness comparing rivaroxaban with enoxaparin for
thromboprophylaxis.
Disclosures: This study was funded by Bayer HealthCare and
Janssen, sponsors that also collected and analyzed the study data. Dr. Cohen
and his associates reported numerous ties to industry sources.
Robotic surgery called 'in the destiny of humanity'
The way Dr. Pier Cristoforo Giulianotti sees it, robots will play an increasingly prominent role in the future of surgery. He should know. Dr. Giulianotti, the Lloyd Nyhus Professor of Surgery and chief, division of minimally invasive, general, and robotic surgery at the University of Illinois at Chicago, pioneered robotic lung resection in 2002 and was the first surgeon to perform a robotic Whipple procedure in 2001.
"In my opinion there is no way back from robotic surgery," he said in an interview. "It’s in the destiny of humanity, for the simple reason: to perform more precisely and to overcome our limitations – not only manual limitations, the ability to control movements at a microscopic level, for example – but also diagnostic limitations. The computer and the artificial intelligence of the future will integrate our senses and our mechanical abilities, so we will be able to perform more precisely on selected targets."
Currently, most abdominal procedures performed at the University of Illinois Medical Center are done robotically, including the Whipple procedure, splenectomy, total gastrectomy, lung lobectomy, colorectal surgery, thyroidectomy, adrenalectomy, esophagectomy, major hepatectomies, and common bile duct procedures. The robot "has enormous benefits for the patients," Dr. Giulianotti said. "Open procedures involve a longer postoperative stay and more complications, and it takes more time to enter adjuvant oncological treatment after surgery, and there is less blood loss."
In Dr. Giulianotti’s published experience of 134 robotic-assisted laparoscopic pancreatic surgery procedures, morbidity was 26%, mortality was 2.2%, the conversion rate was 10.4%, and the fistula rate was 20.9%. The majority were grade A fistulas not requiring any treatment (Surg. Endosc. 2010; 24:1646-57). He said that robotic surgery "enables difficult technical maneuvers to be performed that facilitate the success of pancreatic minimally invasive surgery. The results in this series demonstrate feasibility and safety with clinical outcomes."
In a separate study, investigators who compared 32 open vs. 28 laparoscopic vs. 17 robotic distal pancreatectomies found that all three procedures resulted in a similar cost, while the robotic group had a shorter hospital stay, a higher rate of spleen-preserving cases, and an increased operative time, compared with patients in the other two groups (Surgery 2010;148:814-23).
Pelvic indications that are becoming the gold standard for robotic surgery include robotic prostatectomy and resection for rectal cancer. "When you have a very deep and complex surgical field like the pelvis, and you need to do a radical resection for cancer while at the same time sparing nerves that can be important for urinary function, the robot combines three-dimensional vision plus improved functionality of the instruments, allowing for a more precise dissection," Dr. Giulianotti said.
Dr. Leela M. Prasad, chief of the division of colon and rectal surgery at the University of Illinois at Chicago, described robotic-assisted surgery for rectal cancer as "far superior to anything we know right now," especially in overweight males. According to combined results from three published studies, circumferential margin positivity ranged from 0 to 7%, which, Dr. Prasad said, is significantly lower than the average number of positive margins reported following open and laparoscopic rectal surgery procedures (14% and 16%, respectively) (Ann. Surg. Oncol. 2007;14:3168-73; Surg. Oncol. 2009;16:1480-7; Dis. Colon Rectum 2009;52:1824-30).
At the present time, however, in his clinical experience there are no significant differences in outcomes between robotic-assisted and laparoscopically assisted open surgery for colon cancer. "We looked at our data, and it did not make any difference robotically or laparoscopically in terms of length of stay, lymph node harvest, or survival," he said. A recent study from Korean investigators supports those findings (Surg. Endosc. 2012 Dec. 13).
Dr. Giulianotti estimated that there are more than 1,600 robotic surgery systems in the United States. Some of them are devoted to urologic or gynecologic applications, "but in the majority of hospitals there is a growing tendency to use the robot as part of a multidisciplinary program," he said. "That means at the same institution different surgical teams on different days are using the robot for different indications. Of the 1,600 systems out there, I would estimate that about half are used for general surgery."
While hospital administrators have warmed up to the idea of initiating robotic surgery programs at their institutions in recent years, Dr. Giulianotti and Dr. Prasad both called for a culture change in terms of how surgeons and medical educators think about the robot’s place in clinical medicine. "There is a cultural resistance in accepting big changes in surgery," Dr. Giulianotti said. "Academic medical institutions can play a key role here. They need to start teaching robotics in a mandatory way by offering training opportunities in a lab. We also believe that simulation has a big future in this kind of training."
The economic investment required to launch a robotic surgery program is another challenge. "Some hospitals are only concerned about the cost," he said. "When you are doing the same procedure laparoscopically, the cost is probably about 30% less. In the future I hope we can better impact the overall economic aspect of surgical procedures. That means speeding up the postoperative and outpatient treatment of some pathologies. We are already doing Nissen fundoplication and gallbladder removal with robotic surgery, and patients are being discharged a few hours after the surgery."
Dr. Prasad said the field will be poised to further advance when makers of robotic surgery devices and instruments improve on existing technology. "I am using the same robot and instruments as I was using 3 years ago," he said. "I think we need new technologies – including a smaller robot to cut down the costs and speed up the operations. This will make it easier for patients."
To launch a successful robotic surgery program in this day and age, "you need a good team of nurses and surgeons who are dedicated to doing robotic surgery," Dr. Prasad concluded. "It is dangerous in the hands of a surgeon who is not qualified."
Dr. Giulianotti said that he had no relevant financial conflicts to disclose. Dr. Prasad disclosed that he has received honoraria from Intuitive Surgical, Ethicon Endo-Surgery, and Covidien.
I would like to congratulate Dr. Pier Cristoforo Giulianotti for his work to date demonstrating the effectiveness of robotic pancreatic surgery. Clearly the “robot,” as it is affectionately termed, is here to stay.
It has clear applications in all surgical subspecialties and is being embraced by surgeons, administrators, and patients as an important tool. It is a platform that will facilitate future developments that are certain to change the way we practice our craft.
Dr. John Sweeney |
Given the focus being placed on the value of health care deliveredin the United States, it is incumbent upon surgeons to continuously evaluate the quality and costs of each intervention that we offer to our patients. To many, this type of evaluative process might seem to come up short when it comes to robotic applications for many minimally invasive procedures. Quality in most studies has been equivalent, while the costs associated with this expensive technology can be very significant. Because this new piece of technology is the first step in a new direction, we must continue to place the time, effort and cost into refining, enhancing and improving it’s applications as the technology stands today. However unless we are able to accomplish this goal, I fear we fall into the trap of “using a Cadillac for a golf cart” which is something that the U.S. health care system cannot afford nor sustain.
Dr. John Sweeney, is an ACS Fellow and the W. Dean Warren Distinguished Chair in Surgery at Emory University, Atlanta.
I would like to congratulate Dr. Pier Cristoforo Giulianotti for his work to date demonstrating the effectiveness of robotic pancreatic surgery. Clearly the “robot,” as it is affectionately termed, is here to stay.
It has clear applications in all surgical subspecialties and is being embraced by surgeons, administrators, and patients as an important tool. It is a platform that will facilitate future developments that are certain to change the way we practice our craft.
Dr. John Sweeney |
Given the focus being placed on the value of health care deliveredin the United States, it is incumbent upon surgeons to continuously evaluate the quality and costs of each intervention that we offer to our patients. To many, this type of evaluative process might seem to come up short when it comes to robotic applications for many minimally invasive procedures. Quality in most studies has been equivalent, while the costs associated with this expensive technology can be very significant. Because this new piece of technology is the first step in a new direction, we must continue to place the time, effort and cost into refining, enhancing and improving it’s applications as the technology stands today. However unless we are able to accomplish this goal, I fear we fall into the trap of “using a Cadillac for a golf cart” which is something that the U.S. health care system cannot afford nor sustain.
Dr. John Sweeney, is an ACS Fellow and the W. Dean Warren Distinguished Chair in Surgery at Emory University, Atlanta.
I would like to congratulate Dr. Pier Cristoforo Giulianotti for his work to date demonstrating the effectiveness of robotic pancreatic surgery. Clearly the “robot,” as it is affectionately termed, is here to stay.
It has clear applications in all surgical subspecialties and is being embraced by surgeons, administrators, and patients as an important tool. It is a platform that will facilitate future developments that are certain to change the way we practice our craft.
Dr. John Sweeney |
Given the focus being placed on the value of health care deliveredin the United States, it is incumbent upon surgeons to continuously evaluate the quality and costs of each intervention that we offer to our patients. To many, this type of evaluative process might seem to come up short when it comes to robotic applications for many minimally invasive procedures. Quality in most studies has been equivalent, while the costs associated with this expensive technology can be very significant. Because this new piece of technology is the first step in a new direction, we must continue to place the time, effort and cost into refining, enhancing and improving it’s applications as the technology stands today. However unless we are able to accomplish this goal, I fear we fall into the trap of “using a Cadillac for a golf cart” which is something that the U.S. health care system cannot afford nor sustain.
Dr. John Sweeney, is an ACS Fellow and the W. Dean Warren Distinguished Chair in Surgery at Emory University, Atlanta.
The way Dr. Pier Cristoforo Giulianotti sees it, robots will play an increasingly prominent role in the future of surgery. He should know. Dr. Giulianotti, the Lloyd Nyhus Professor of Surgery and chief, division of minimally invasive, general, and robotic surgery at the University of Illinois at Chicago, pioneered robotic lung resection in 2002 and was the first surgeon to perform a robotic Whipple procedure in 2001.
"In my opinion there is no way back from robotic surgery," he said in an interview. "It’s in the destiny of humanity, for the simple reason: to perform more precisely and to overcome our limitations – not only manual limitations, the ability to control movements at a microscopic level, for example – but also diagnostic limitations. The computer and the artificial intelligence of the future will integrate our senses and our mechanical abilities, so we will be able to perform more precisely on selected targets."
Currently, most abdominal procedures performed at the University of Illinois Medical Center are done robotically, including the Whipple procedure, splenectomy, total gastrectomy, lung lobectomy, colorectal surgery, thyroidectomy, adrenalectomy, esophagectomy, major hepatectomies, and common bile duct procedures. The robot "has enormous benefits for the patients," Dr. Giulianotti said. "Open procedures involve a longer postoperative stay and more complications, and it takes more time to enter adjuvant oncological treatment after surgery, and there is less blood loss."
In Dr. Giulianotti’s published experience of 134 robotic-assisted laparoscopic pancreatic surgery procedures, morbidity was 26%, mortality was 2.2%, the conversion rate was 10.4%, and the fistula rate was 20.9%. The majority were grade A fistulas not requiring any treatment (Surg. Endosc. 2010; 24:1646-57). He said that robotic surgery "enables difficult technical maneuvers to be performed that facilitate the success of pancreatic minimally invasive surgery. The results in this series demonstrate feasibility and safety with clinical outcomes."
In a separate study, investigators who compared 32 open vs. 28 laparoscopic vs. 17 robotic distal pancreatectomies found that all three procedures resulted in a similar cost, while the robotic group had a shorter hospital stay, a higher rate of spleen-preserving cases, and an increased operative time, compared with patients in the other two groups (Surgery 2010;148:814-23).
Pelvic indications that are becoming the gold standard for robotic surgery include robotic prostatectomy and resection for rectal cancer. "When you have a very deep and complex surgical field like the pelvis, and you need to do a radical resection for cancer while at the same time sparing nerves that can be important for urinary function, the robot combines three-dimensional vision plus improved functionality of the instruments, allowing for a more precise dissection," Dr. Giulianotti said.
Dr. Leela M. Prasad, chief of the division of colon and rectal surgery at the University of Illinois at Chicago, described robotic-assisted surgery for rectal cancer as "far superior to anything we know right now," especially in overweight males. According to combined results from three published studies, circumferential margin positivity ranged from 0 to 7%, which, Dr. Prasad said, is significantly lower than the average number of positive margins reported following open and laparoscopic rectal surgery procedures (14% and 16%, respectively) (Ann. Surg. Oncol. 2007;14:3168-73; Surg. Oncol. 2009;16:1480-7; Dis. Colon Rectum 2009;52:1824-30).
At the present time, however, in his clinical experience there are no significant differences in outcomes between robotic-assisted and laparoscopically assisted open surgery for colon cancer. "We looked at our data, and it did not make any difference robotically or laparoscopically in terms of length of stay, lymph node harvest, or survival," he said. A recent study from Korean investigators supports those findings (Surg. Endosc. 2012 Dec. 13).
Dr. Giulianotti estimated that there are more than 1,600 robotic surgery systems in the United States. Some of them are devoted to urologic or gynecologic applications, "but in the majority of hospitals there is a growing tendency to use the robot as part of a multidisciplinary program," he said. "That means at the same institution different surgical teams on different days are using the robot for different indications. Of the 1,600 systems out there, I would estimate that about half are used for general surgery."
While hospital administrators have warmed up to the idea of initiating robotic surgery programs at their institutions in recent years, Dr. Giulianotti and Dr. Prasad both called for a culture change in terms of how surgeons and medical educators think about the robot’s place in clinical medicine. "There is a cultural resistance in accepting big changes in surgery," Dr. Giulianotti said. "Academic medical institutions can play a key role here. They need to start teaching robotics in a mandatory way by offering training opportunities in a lab. We also believe that simulation has a big future in this kind of training."
The economic investment required to launch a robotic surgery program is another challenge. "Some hospitals are only concerned about the cost," he said. "When you are doing the same procedure laparoscopically, the cost is probably about 30% less. In the future I hope we can better impact the overall economic aspect of surgical procedures. That means speeding up the postoperative and outpatient treatment of some pathologies. We are already doing Nissen fundoplication and gallbladder removal with robotic surgery, and patients are being discharged a few hours after the surgery."
Dr. Prasad said the field will be poised to further advance when makers of robotic surgery devices and instruments improve on existing technology. "I am using the same robot and instruments as I was using 3 years ago," he said. "I think we need new technologies – including a smaller robot to cut down the costs and speed up the operations. This will make it easier for patients."
To launch a successful robotic surgery program in this day and age, "you need a good team of nurses and surgeons who are dedicated to doing robotic surgery," Dr. Prasad concluded. "It is dangerous in the hands of a surgeon who is not qualified."
Dr. Giulianotti said that he had no relevant financial conflicts to disclose. Dr. Prasad disclosed that he has received honoraria from Intuitive Surgical, Ethicon Endo-Surgery, and Covidien.
The way Dr. Pier Cristoforo Giulianotti sees it, robots will play an increasingly prominent role in the future of surgery. He should know. Dr. Giulianotti, the Lloyd Nyhus Professor of Surgery and chief, division of minimally invasive, general, and robotic surgery at the University of Illinois at Chicago, pioneered robotic lung resection in 2002 and was the first surgeon to perform a robotic Whipple procedure in 2001.
"In my opinion there is no way back from robotic surgery," he said in an interview. "It’s in the destiny of humanity, for the simple reason: to perform more precisely and to overcome our limitations – not only manual limitations, the ability to control movements at a microscopic level, for example – but also diagnostic limitations. The computer and the artificial intelligence of the future will integrate our senses and our mechanical abilities, so we will be able to perform more precisely on selected targets."
Currently, most abdominal procedures performed at the University of Illinois Medical Center are done robotically, including the Whipple procedure, splenectomy, total gastrectomy, lung lobectomy, colorectal surgery, thyroidectomy, adrenalectomy, esophagectomy, major hepatectomies, and common bile duct procedures. The robot "has enormous benefits for the patients," Dr. Giulianotti said. "Open procedures involve a longer postoperative stay and more complications, and it takes more time to enter adjuvant oncological treatment after surgery, and there is less blood loss."
In Dr. Giulianotti’s published experience of 134 robotic-assisted laparoscopic pancreatic surgery procedures, morbidity was 26%, mortality was 2.2%, the conversion rate was 10.4%, and the fistula rate was 20.9%. The majority were grade A fistulas not requiring any treatment (Surg. Endosc. 2010; 24:1646-57). He said that robotic surgery "enables difficult technical maneuvers to be performed that facilitate the success of pancreatic minimally invasive surgery. The results in this series demonstrate feasibility and safety with clinical outcomes."
In a separate study, investigators who compared 32 open vs. 28 laparoscopic vs. 17 robotic distal pancreatectomies found that all three procedures resulted in a similar cost, while the robotic group had a shorter hospital stay, a higher rate of spleen-preserving cases, and an increased operative time, compared with patients in the other two groups (Surgery 2010;148:814-23).
Pelvic indications that are becoming the gold standard for robotic surgery include robotic prostatectomy and resection for rectal cancer. "When you have a very deep and complex surgical field like the pelvis, and you need to do a radical resection for cancer while at the same time sparing nerves that can be important for urinary function, the robot combines three-dimensional vision plus improved functionality of the instruments, allowing for a more precise dissection," Dr. Giulianotti said.
Dr. Leela M. Prasad, chief of the division of colon and rectal surgery at the University of Illinois at Chicago, described robotic-assisted surgery for rectal cancer as "far superior to anything we know right now," especially in overweight males. According to combined results from three published studies, circumferential margin positivity ranged from 0 to 7%, which, Dr. Prasad said, is significantly lower than the average number of positive margins reported following open and laparoscopic rectal surgery procedures (14% and 16%, respectively) (Ann. Surg. Oncol. 2007;14:3168-73; Surg. Oncol. 2009;16:1480-7; Dis. Colon Rectum 2009;52:1824-30).
At the present time, however, in his clinical experience there are no significant differences in outcomes between robotic-assisted and laparoscopically assisted open surgery for colon cancer. "We looked at our data, and it did not make any difference robotically or laparoscopically in terms of length of stay, lymph node harvest, or survival," he said. A recent study from Korean investigators supports those findings (Surg. Endosc. 2012 Dec. 13).
Dr. Giulianotti estimated that there are more than 1,600 robotic surgery systems in the United States. Some of them are devoted to urologic or gynecologic applications, "but in the majority of hospitals there is a growing tendency to use the robot as part of a multidisciplinary program," he said. "That means at the same institution different surgical teams on different days are using the robot for different indications. Of the 1,600 systems out there, I would estimate that about half are used for general surgery."
While hospital administrators have warmed up to the idea of initiating robotic surgery programs at their institutions in recent years, Dr. Giulianotti and Dr. Prasad both called for a culture change in terms of how surgeons and medical educators think about the robot’s place in clinical medicine. "There is a cultural resistance in accepting big changes in surgery," Dr. Giulianotti said. "Academic medical institutions can play a key role here. They need to start teaching robotics in a mandatory way by offering training opportunities in a lab. We also believe that simulation has a big future in this kind of training."
The economic investment required to launch a robotic surgery program is another challenge. "Some hospitals are only concerned about the cost," he said. "When you are doing the same procedure laparoscopically, the cost is probably about 30% less. In the future I hope we can better impact the overall economic aspect of surgical procedures. That means speeding up the postoperative and outpatient treatment of some pathologies. We are already doing Nissen fundoplication and gallbladder removal with robotic surgery, and patients are being discharged a few hours after the surgery."
Dr. Prasad said the field will be poised to further advance when makers of robotic surgery devices and instruments improve on existing technology. "I am using the same robot and instruments as I was using 3 years ago," he said. "I think we need new technologies – including a smaller robot to cut down the costs and speed up the operations. This will make it easier for patients."
To launch a successful robotic surgery program in this day and age, "you need a good team of nurses and surgeons who are dedicated to doing robotic surgery," Dr. Prasad concluded. "It is dangerous in the hands of a surgeon who is not qualified."
Dr. Giulianotti said that he had no relevant financial conflicts to disclose. Dr. Prasad disclosed that he has received honoraria from Intuitive Surgical, Ethicon Endo-Surgery, and Covidien.
High-frequency oscillatory ventilation may worsen ARDS
High-frequency oscillatory ventilation doesn’t improve and may actually worsen moderate to severe acute respiratory distress syndrome in adults, compared with standard ventilation, according to two reports published online Jan. 23 in the New England Journal of Medicine.
In two large, separate, randomized controlled trials comparing the two ventilation strategies, 1-month mortality in critically ill adults with ARDS who received high-frequency oscillatory ventilation (HFOV) was either higher or not significantly different from that in patients who received standard low tidal volume and high positive end–expiratory pressure ventilation.
In one study, which was terminated early because of the large discrepancy in short-term mortality, HFOV also was associated with higher mean airway pressures and significantly greater need for sedatives, neuromuscular blockers, and vasoactive drugs.
Both trials call into question the current widespread use of HFOV early in the course of ARDS when patients don’t show an adequate response to conventional mechanical ventilation, the two research groups noted.
HFOV, which delivers very small tidal volumes at very high rates, is thought to minimize the lung damage done by ventilation’s repeated forced opening and collapsing of lung structures. Many clinicians now use it earlier in the course of ARDS, even though there are other approaches for improving oxygenation, based solely on the results of animal studies and small trials that used outdated ventilation methods as a control. The commercial availability of HFOV equipment has accelerated this trend.
In the absence of good evidence of HFOV’s effectiveness, experts in Canada and the United Kingdom called for rigorous randomized controlled trials.
Dr. Niall D. Ferguson and his associates in the Oscillation for Acute Respiratory Distress Syndrome Treated Early (OSCILLATE) trial compared the two strategies in patients aged 16-85 years who had moderate to severe ARDS and were treated at 39 intensive care units in Canada, Saudi Arabia, the United States, Chile, and India.
The OSCILLATE steering committee terminated the trial early, after only 548 subjects had been randomized, because three consecutive interim analyses showed that 1-month mortality was consistently higher with HFOV. "The effect size was sufficiently large that we concluded that even if early HFOV did not increase mortality, it would be very unlikely to decrease mortality," wrote Dr. Ferguson of the division of critical care medicine and the departments of medicine and physiology, University of Toronto, and his colleagues.
At that time, 1-month mortality was 47% for HFOV (129 of 275 patients), compared with 35% (96 of 273 patients) in the control group (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMoa1215554]).
This result remained robust in three further analyses of the data that controlled for numerous variables. It also persisted in several subgroup analyses, regardless of the subjects’ baseline severity of hypoxemia, the subjects’ BMI, the use or nonuse of vasopressors, or the level of experience with ventilation at each medical center.
In addition, the use of vasopressors, which had been equivalent between the two study groups at baseline at approximately 63%, increased in the HFOV group to 73% within 4 hours of beginning the procedure and to 78% the next day. In contrast, vasopressors were used in only 62% and 58%, respectively, in the control group.
The use of neuromuscular blockers followed the same pattern, increasing in the HFOV group from 27% at baseline to 46% at 4 hours and 46% at 24 hours but remaining more steady in the control group at 29%, 31%, and 26%, respectively, they reported.
Cumulatively, vasoactive agents were used in 91% and neuromuscular blockers in 83% of the HFOV group, compared with 84% and 68%, respectively, in the control group. Vasoactive agents were required for an average of 2 days longer in the HFOV group, and neuromuscular blockers were required for an average of 1 day longer, compared with the control group.
Sedatives and opioids (primarily midazolam and fentanyl) were given for the same duration to the two study groups, but doses were higher in the HFOV group.
"Our results raise serious concerns about the early use of HFOV for the management of ARDS in adults. The results of this study increase the uncertainty about possible benefits of HFOV even when applied in patients with life-threatening refractory hypoxemia," Dr. Ferguson and his associates said.
In the Oscillation in ARDS (OSCAR) study, commissioned by the U.K.’s National Institute for Health Research, two ventilation techniques were compared in 795 patients in general ICUs at 12 university hospitals, 4 university-affiliated hospitals, and 13 district general hospitals in England, Wales, and Scotland.
As this was a pragmatic study meant to reflect real-world practice, HFOV was compared against whatever conventional ventilation was used in local practice, and the ICUs were "encouraged" to use the recommended combinations of positive end-expiratory pressure and fraction of inspired oxygen values. Consequently, management of the control group varied greatly from one ICU to the next, according to the researchers, led by Dr. Duncan Young of John Radcliffe Hospital and the University of Oxford, both in Oxford.
The primary outcome, 30-day all-cause mortality, occurred in 41.7% of the HFOV group (166 of 398 patients) and in 41.1% of the control group (163 of 397 patients), a nonsignificant difference. These rates remained largely unchanged in further analyses that adjusted for several variables, Dr. Young and his colleagues said (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMoa1215716]).
The duration of ICU stay was 17.6 days with HFOV and 16.1 days with conventional ventilation, and the duration of hospital stay was 33.1 days and 33.9 days, respectively.
"Since data collection is ongoing, we cannot yet report the longer-term outcomes (including survival and health-related quality of life)," they added.
In conclusion, "we were unable to find any benefit or harm from the use of HFOV in adult patients with ARDS. We recommend that this mode of ventilation not be used for routine care," Dr. Young and his associates said.
The OSCILLATE study was supported by the Canadian Institutes of Health Research, the King Abdullah International Medical Research Center, and Fonds de Recherche de Quebec. Dr. Ferguson’s associates reported ties to numerous industry sources. The OSCAR study was supported by the National Institute for Health Research Health Technology Assessment Programme. Dr. Young and his associates reported no relevant financial conflicts of interest.
CareFusion provided the SensorMedics HFO ventilator and technical support for the OSCILLATE trial but had no role in study design, data collection or analysis, or manuscript preparation. Inspiration Healthcare supplied the ventilators for the OSCAR study but had no role in the study design, data acquisition or analysis, or manuscript preparation.
Both the OSCILLATE and the OSCAR trial raise concerns about the widespread and routine use of HFOV, said Dr. Atul Malhotra and Dr. Jeffrey M. Drazen.
Dr. Jeffrey M. Drazen |
"However, one could argue that it is not HFOV itself but the HFOV protocols studied in these trials that were ineffective, and perhaps worse, than usual care," they wrote. Questions remain concerning this method’s usefulness after more aggressive volume resuscitation, less aggressive use of sedation and paralysis, and selection of patients who have homgeneous, recruitable lung.
"Perhaps patients with ARDS will require individualized therapy that takes into consideration their body habitus, the cause of their disease, and the mechanisms leading to lung injury ... but for now clinicians should be cautious about applying HFOV routinely in patients with ARDS," they said.
Dr. Malhotra and Dr. Drazen are with the divisions of sleep medicine and pulmonary and critical care medicine at Brigham and Women’s Hospital, Boston. Dr. Malhotra reported ties to Philips Respironics, Pfizer, Apnex, and Apnicure. These remarks were taken from their editorial accompanying the two reports (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMe1300103]).
Both the OSCILLATE and the OSCAR trial raise concerns about the widespread and routine use of HFOV, said Dr. Atul Malhotra and Dr. Jeffrey M. Drazen.
Dr. Jeffrey M. Drazen |
"However, one could argue that it is not HFOV itself but the HFOV protocols studied in these trials that were ineffective, and perhaps worse, than usual care," they wrote. Questions remain concerning this method’s usefulness after more aggressive volume resuscitation, less aggressive use of sedation and paralysis, and selection of patients who have homgeneous, recruitable lung.
"Perhaps patients with ARDS will require individualized therapy that takes into consideration their body habitus, the cause of their disease, and the mechanisms leading to lung injury ... but for now clinicians should be cautious about applying HFOV routinely in patients with ARDS," they said.
Dr. Malhotra and Dr. Drazen are with the divisions of sleep medicine and pulmonary and critical care medicine at Brigham and Women’s Hospital, Boston. Dr. Malhotra reported ties to Philips Respironics, Pfizer, Apnex, and Apnicure. These remarks were taken from their editorial accompanying the two reports (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMe1300103]).
Both the OSCILLATE and the OSCAR trial raise concerns about the widespread and routine use of HFOV, said Dr. Atul Malhotra and Dr. Jeffrey M. Drazen.
Dr. Jeffrey M. Drazen |
"However, one could argue that it is not HFOV itself but the HFOV protocols studied in these trials that were ineffective, and perhaps worse, than usual care," they wrote. Questions remain concerning this method’s usefulness after more aggressive volume resuscitation, less aggressive use of sedation and paralysis, and selection of patients who have homgeneous, recruitable lung.
"Perhaps patients with ARDS will require individualized therapy that takes into consideration their body habitus, the cause of their disease, and the mechanisms leading to lung injury ... but for now clinicians should be cautious about applying HFOV routinely in patients with ARDS," they said.
Dr. Malhotra and Dr. Drazen are with the divisions of sleep medicine and pulmonary and critical care medicine at Brigham and Women’s Hospital, Boston. Dr. Malhotra reported ties to Philips Respironics, Pfizer, Apnex, and Apnicure. These remarks were taken from their editorial accompanying the two reports (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMe1300103]).
High-frequency oscillatory ventilation doesn’t improve and may actually worsen moderate to severe acute respiratory distress syndrome in adults, compared with standard ventilation, according to two reports published online Jan. 23 in the New England Journal of Medicine.
In two large, separate, randomized controlled trials comparing the two ventilation strategies, 1-month mortality in critically ill adults with ARDS who received high-frequency oscillatory ventilation (HFOV) was either higher or not significantly different from that in patients who received standard low tidal volume and high positive end–expiratory pressure ventilation.
In one study, which was terminated early because of the large discrepancy in short-term mortality, HFOV also was associated with higher mean airway pressures and significantly greater need for sedatives, neuromuscular blockers, and vasoactive drugs.
Both trials call into question the current widespread use of HFOV early in the course of ARDS when patients don’t show an adequate response to conventional mechanical ventilation, the two research groups noted.
HFOV, which delivers very small tidal volumes at very high rates, is thought to minimize the lung damage done by ventilation’s repeated forced opening and collapsing of lung structures. Many clinicians now use it earlier in the course of ARDS, even though there are other approaches for improving oxygenation, based solely on the results of animal studies and small trials that used outdated ventilation methods as a control. The commercial availability of HFOV equipment has accelerated this trend.
In the absence of good evidence of HFOV’s effectiveness, experts in Canada and the United Kingdom called for rigorous randomized controlled trials.
Dr. Niall D. Ferguson and his associates in the Oscillation for Acute Respiratory Distress Syndrome Treated Early (OSCILLATE) trial compared the two strategies in patients aged 16-85 years who had moderate to severe ARDS and were treated at 39 intensive care units in Canada, Saudi Arabia, the United States, Chile, and India.
The OSCILLATE steering committee terminated the trial early, after only 548 subjects had been randomized, because three consecutive interim analyses showed that 1-month mortality was consistently higher with HFOV. "The effect size was sufficiently large that we concluded that even if early HFOV did not increase mortality, it would be very unlikely to decrease mortality," wrote Dr. Ferguson of the division of critical care medicine and the departments of medicine and physiology, University of Toronto, and his colleagues.
At that time, 1-month mortality was 47% for HFOV (129 of 275 patients), compared with 35% (96 of 273 patients) in the control group (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMoa1215554]).
This result remained robust in three further analyses of the data that controlled for numerous variables. It also persisted in several subgroup analyses, regardless of the subjects’ baseline severity of hypoxemia, the subjects’ BMI, the use or nonuse of vasopressors, or the level of experience with ventilation at each medical center.
In addition, the use of vasopressors, which had been equivalent between the two study groups at baseline at approximately 63%, increased in the HFOV group to 73% within 4 hours of beginning the procedure and to 78% the next day. In contrast, vasopressors were used in only 62% and 58%, respectively, in the control group.
The use of neuromuscular blockers followed the same pattern, increasing in the HFOV group from 27% at baseline to 46% at 4 hours and 46% at 24 hours but remaining more steady in the control group at 29%, 31%, and 26%, respectively, they reported.
Cumulatively, vasoactive agents were used in 91% and neuromuscular blockers in 83% of the HFOV group, compared with 84% and 68%, respectively, in the control group. Vasoactive agents were required for an average of 2 days longer in the HFOV group, and neuromuscular blockers were required for an average of 1 day longer, compared with the control group.
Sedatives and opioids (primarily midazolam and fentanyl) were given for the same duration to the two study groups, but doses were higher in the HFOV group.
"Our results raise serious concerns about the early use of HFOV for the management of ARDS in adults. The results of this study increase the uncertainty about possible benefits of HFOV even when applied in patients with life-threatening refractory hypoxemia," Dr. Ferguson and his associates said.
In the Oscillation in ARDS (OSCAR) study, commissioned by the U.K.’s National Institute for Health Research, two ventilation techniques were compared in 795 patients in general ICUs at 12 university hospitals, 4 university-affiliated hospitals, and 13 district general hospitals in England, Wales, and Scotland.
As this was a pragmatic study meant to reflect real-world practice, HFOV was compared against whatever conventional ventilation was used in local practice, and the ICUs were "encouraged" to use the recommended combinations of positive end-expiratory pressure and fraction of inspired oxygen values. Consequently, management of the control group varied greatly from one ICU to the next, according to the researchers, led by Dr. Duncan Young of John Radcliffe Hospital and the University of Oxford, both in Oxford.
The primary outcome, 30-day all-cause mortality, occurred in 41.7% of the HFOV group (166 of 398 patients) and in 41.1% of the control group (163 of 397 patients), a nonsignificant difference. These rates remained largely unchanged in further analyses that adjusted for several variables, Dr. Young and his colleagues said (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMoa1215716]).
The duration of ICU stay was 17.6 days with HFOV and 16.1 days with conventional ventilation, and the duration of hospital stay was 33.1 days and 33.9 days, respectively.
"Since data collection is ongoing, we cannot yet report the longer-term outcomes (including survival and health-related quality of life)," they added.
In conclusion, "we were unable to find any benefit or harm from the use of HFOV in adult patients with ARDS. We recommend that this mode of ventilation not be used for routine care," Dr. Young and his associates said.
The OSCILLATE study was supported by the Canadian Institutes of Health Research, the King Abdullah International Medical Research Center, and Fonds de Recherche de Quebec. Dr. Ferguson’s associates reported ties to numerous industry sources. The OSCAR study was supported by the National Institute for Health Research Health Technology Assessment Programme. Dr. Young and his associates reported no relevant financial conflicts of interest.
CareFusion provided the SensorMedics HFO ventilator and technical support for the OSCILLATE trial but had no role in study design, data collection or analysis, or manuscript preparation. Inspiration Healthcare supplied the ventilators for the OSCAR study but had no role in the study design, data acquisition or analysis, or manuscript preparation.
High-frequency oscillatory ventilation doesn’t improve and may actually worsen moderate to severe acute respiratory distress syndrome in adults, compared with standard ventilation, according to two reports published online Jan. 23 in the New England Journal of Medicine.
In two large, separate, randomized controlled trials comparing the two ventilation strategies, 1-month mortality in critically ill adults with ARDS who received high-frequency oscillatory ventilation (HFOV) was either higher or not significantly different from that in patients who received standard low tidal volume and high positive end–expiratory pressure ventilation.
In one study, which was terminated early because of the large discrepancy in short-term mortality, HFOV also was associated with higher mean airway pressures and significantly greater need for sedatives, neuromuscular blockers, and vasoactive drugs.
Both trials call into question the current widespread use of HFOV early in the course of ARDS when patients don’t show an adequate response to conventional mechanical ventilation, the two research groups noted.
HFOV, which delivers very small tidal volumes at very high rates, is thought to minimize the lung damage done by ventilation’s repeated forced opening and collapsing of lung structures. Many clinicians now use it earlier in the course of ARDS, even though there are other approaches for improving oxygenation, based solely on the results of animal studies and small trials that used outdated ventilation methods as a control. The commercial availability of HFOV equipment has accelerated this trend.
In the absence of good evidence of HFOV’s effectiveness, experts in Canada and the United Kingdom called for rigorous randomized controlled trials.
Dr. Niall D. Ferguson and his associates in the Oscillation for Acute Respiratory Distress Syndrome Treated Early (OSCILLATE) trial compared the two strategies in patients aged 16-85 years who had moderate to severe ARDS and were treated at 39 intensive care units in Canada, Saudi Arabia, the United States, Chile, and India.
The OSCILLATE steering committee terminated the trial early, after only 548 subjects had been randomized, because three consecutive interim analyses showed that 1-month mortality was consistently higher with HFOV. "The effect size was sufficiently large that we concluded that even if early HFOV did not increase mortality, it would be very unlikely to decrease mortality," wrote Dr. Ferguson of the division of critical care medicine and the departments of medicine and physiology, University of Toronto, and his colleagues.
At that time, 1-month mortality was 47% for HFOV (129 of 275 patients), compared with 35% (96 of 273 patients) in the control group (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMoa1215554]).
This result remained robust in three further analyses of the data that controlled for numerous variables. It also persisted in several subgroup analyses, regardless of the subjects’ baseline severity of hypoxemia, the subjects’ BMI, the use or nonuse of vasopressors, or the level of experience with ventilation at each medical center.
In addition, the use of vasopressors, which had been equivalent between the two study groups at baseline at approximately 63%, increased in the HFOV group to 73% within 4 hours of beginning the procedure and to 78% the next day. In contrast, vasopressors were used in only 62% and 58%, respectively, in the control group.
The use of neuromuscular blockers followed the same pattern, increasing in the HFOV group from 27% at baseline to 46% at 4 hours and 46% at 24 hours but remaining more steady in the control group at 29%, 31%, and 26%, respectively, they reported.
Cumulatively, vasoactive agents were used in 91% and neuromuscular blockers in 83% of the HFOV group, compared with 84% and 68%, respectively, in the control group. Vasoactive agents were required for an average of 2 days longer in the HFOV group, and neuromuscular blockers were required for an average of 1 day longer, compared with the control group.
Sedatives and opioids (primarily midazolam and fentanyl) were given for the same duration to the two study groups, but doses were higher in the HFOV group.
"Our results raise serious concerns about the early use of HFOV for the management of ARDS in adults. The results of this study increase the uncertainty about possible benefits of HFOV even when applied in patients with life-threatening refractory hypoxemia," Dr. Ferguson and his associates said.
In the Oscillation in ARDS (OSCAR) study, commissioned by the U.K.’s National Institute for Health Research, two ventilation techniques were compared in 795 patients in general ICUs at 12 university hospitals, 4 university-affiliated hospitals, and 13 district general hospitals in England, Wales, and Scotland.
As this was a pragmatic study meant to reflect real-world practice, HFOV was compared against whatever conventional ventilation was used in local practice, and the ICUs were "encouraged" to use the recommended combinations of positive end-expiratory pressure and fraction of inspired oxygen values. Consequently, management of the control group varied greatly from one ICU to the next, according to the researchers, led by Dr. Duncan Young of John Radcliffe Hospital and the University of Oxford, both in Oxford.
The primary outcome, 30-day all-cause mortality, occurred in 41.7% of the HFOV group (166 of 398 patients) and in 41.1% of the control group (163 of 397 patients), a nonsignificant difference. These rates remained largely unchanged in further analyses that adjusted for several variables, Dr. Young and his colleagues said (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMoa1215716]).
The duration of ICU stay was 17.6 days with HFOV and 16.1 days with conventional ventilation, and the duration of hospital stay was 33.1 days and 33.9 days, respectively.
"Since data collection is ongoing, we cannot yet report the longer-term outcomes (including survival and health-related quality of life)," they added.
In conclusion, "we were unable to find any benefit or harm from the use of HFOV in adult patients with ARDS. We recommend that this mode of ventilation not be used for routine care," Dr. Young and his associates said.
The OSCILLATE study was supported by the Canadian Institutes of Health Research, the King Abdullah International Medical Research Center, and Fonds de Recherche de Quebec. Dr. Ferguson’s associates reported ties to numerous industry sources. The OSCAR study was supported by the National Institute for Health Research Health Technology Assessment Programme. Dr. Young and his associates reported no relevant financial conflicts of interest.
CareFusion provided the SensorMedics HFO ventilator and technical support for the OSCILLATE trial but had no role in study design, data collection or analysis, or manuscript preparation. Inspiration Healthcare supplied the ventilators for the OSCAR study but had no role in the study design, data acquisition or analysis, or manuscript preparation.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: 1-month mortality was significantly higher with HFOV (47%) than with control ventilation (35%) in the OSCILLATE study; no significant difference was seen between groups in the OSCAR study (41.7% vs 41.1%).
Data Source: Two randomized controlled trials comparing HFOV against either low tidal volume, high positive end–expiratory pressure ventilation or conventional ventilation among 548 ARDS patients in Canada, Saudi Arabia, the United States, Chile, and India and among 795 ARDS patients in England, Wales, and Scotland.
Disclosures: The OSCILLATE study was supported by the Canadian Institutes of Health Research, the King Abdullah International Medical Research Center, and Fonds de Recherche de Quebec. Dr. Ferguson's associates reported ties to numerous industry sources. The OSCAR study was supported by the National Institute for Health Research Health Technology Assessment Programme. Dr. Young and his associates reported no relevant financial conflicts of interest.
TORS: Postop bleeding risk rises with antithrombotic use
The risk of postoperative hemorrhage after transoral robotic-assisted surgery was significantly higher in patients taking antithrombotic medication than in those not taking it, based on data from 147 consecutive patients.
"Even with this small sample size, we were able to identify that increased risk was associated with antithrombotic medication use," said Dr. Scott Asher of the University of Alabama at Birmingham.
Transoral robotic-assisted surgery (TORS) is gaining in popularity among head and neck surgeons, but the potential for postop bleeding in patients taking antithrombotic medication remains a problem, Dr. Asher said at the annual meeting of the American Academy of Otolaryngology – Head and Neck Surgery Foundation.
To assess the postop bleeding complications in TORS patients, Dr. Asher and his colleagues reviewed data from patients seen at a single tertiary academic medical center between March 2007 and September 2011.
Overall, 11 patients (8%) experienced some postop hemorrhage, but 8 of these hemorrhages (72%) occurred in patients taking antithrombotics, Dr. Asher said. Nine patients who hemorrhaged returned to the operating room for further examination and bleeding control, he noted. All postop hemorrhage events were controlled with standard techniques.
The incidence of postop hemorrhage was significantly higher among patients on antithrombotics (17%) compared with those not on antithrombotics (3%). However, no significant difference in the incidence of bleeding occurred between patients undergoing primary surgery and those undergoing salvage surgery (7% vs. 10%). Bleeding occurred an average of 11 days after surgery.
The bleeding events occurred past the time points when most patients would resume their antithrombotic medications, Dr. Asher noted.
"Our recommendation is that patients taking these meds should receive additional preop counseling when considering a TORS procedure," he said.
"A second recommendation is to collaborate preoperatively with the physicians who are prescribing," as well as the anesthesia team, to closely analyze the indications for use of antithrombotics, Dr. Asher added. "If you can safely discontinue them, you can potentially improve your TORS outcomes," he said.
Additional long-term safety and outcomes data are needed for TORS procedures, said Dr. Asher. "We are constantly reflecting on our own experience," he said. "We would encourage other institutions to collect and publish their complications-related data to continue to improve TORS outcomes."
Dr. Asher said he had no relevant financial conflicts.
The risk of postoperative hemorrhage after transoral robotic-assisted surgery was significantly higher in patients taking antithrombotic medication than in those not taking it, based on data from 147 consecutive patients.
"Even with this small sample size, we were able to identify that increased risk was associated with antithrombotic medication use," said Dr. Scott Asher of the University of Alabama at Birmingham.
Transoral robotic-assisted surgery (TORS) is gaining in popularity among head and neck surgeons, but the potential for postop bleeding in patients taking antithrombotic medication remains a problem, Dr. Asher said at the annual meeting of the American Academy of Otolaryngology – Head and Neck Surgery Foundation.
To assess the postop bleeding complications in TORS patients, Dr. Asher and his colleagues reviewed data from patients seen at a single tertiary academic medical center between March 2007 and September 2011.
Overall, 11 patients (8%) experienced some postop hemorrhage, but 8 of these hemorrhages (72%) occurred in patients taking antithrombotics, Dr. Asher said. Nine patients who hemorrhaged returned to the operating room for further examination and bleeding control, he noted. All postop hemorrhage events were controlled with standard techniques.
The incidence of postop hemorrhage was significantly higher among patients on antithrombotics (17%) compared with those not on antithrombotics (3%). However, no significant difference in the incidence of bleeding occurred between patients undergoing primary surgery and those undergoing salvage surgery (7% vs. 10%). Bleeding occurred an average of 11 days after surgery.
The bleeding events occurred past the time points when most patients would resume their antithrombotic medications, Dr. Asher noted.
"Our recommendation is that patients taking these meds should receive additional preop counseling when considering a TORS procedure," he said.
"A second recommendation is to collaborate preoperatively with the physicians who are prescribing," as well as the anesthesia team, to closely analyze the indications for use of antithrombotics, Dr. Asher added. "If you can safely discontinue them, you can potentially improve your TORS outcomes," he said.
Additional long-term safety and outcomes data are needed for TORS procedures, said Dr. Asher. "We are constantly reflecting on our own experience," he said. "We would encourage other institutions to collect and publish their complications-related data to continue to improve TORS outcomes."
Dr. Asher said he had no relevant financial conflicts.
The risk of postoperative hemorrhage after transoral robotic-assisted surgery was significantly higher in patients taking antithrombotic medication than in those not taking it, based on data from 147 consecutive patients.
"Even with this small sample size, we were able to identify that increased risk was associated with antithrombotic medication use," said Dr. Scott Asher of the University of Alabama at Birmingham.
Transoral robotic-assisted surgery (TORS) is gaining in popularity among head and neck surgeons, but the potential for postop bleeding in patients taking antithrombotic medication remains a problem, Dr. Asher said at the annual meeting of the American Academy of Otolaryngology – Head and Neck Surgery Foundation.
To assess the postop bleeding complications in TORS patients, Dr. Asher and his colleagues reviewed data from patients seen at a single tertiary academic medical center between March 2007 and September 2011.
Overall, 11 patients (8%) experienced some postop hemorrhage, but 8 of these hemorrhages (72%) occurred in patients taking antithrombotics, Dr. Asher said. Nine patients who hemorrhaged returned to the operating room for further examination and bleeding control, he noted. All postop hemorrhage events were controlled with standard techniques.
The incidence of postop hemorrhage was significantly higher among patients on antithrombotics (17%) compared with those not on antithrombotics (3%). However, no significant difference in the incidence of bleeding occurred between patients undergoing primary surgery and those undergoing salvage surgery (7% vs. 10%). Bleeding occurred an average of 11 days after surgery.
The bleeding events occurred past the time points when most patients would resume their antithrombotic medications, Dr. Asher noted.
"Our recommendation is that patients taking these meds should receive additional preop counseling when considering a TORS procedure," he said.
"A second recommendation is to collaborate preoperatively with the physicians who are prescribing," as well as the anesthesia team, to closely analyze the indications for use of antithrombotics, Dr. Asher added. "If you can safely discontinue them, you can potentially improve your TORS outcomes," he said.
Additional long-term safety and outcomes data are needed for TORS procedures, said Dr. Asher. "We are constantly reflecting on our own experience," he said. "We would encourage other institutions to collect and publish their complications-related data to continue to improve TORS outcomes."
Dr. Asher said he had no relevant financial conflicts.
AT THE ANNUAL MEETING OF THE AMERICAN ACADEMY OF OTOLARYNGOLOGY; HEAD AND NECK SURGERY FOUNDATION
Major Finding: Approximately three-quarters of patients (8 of 11) who underwent TORS and experienced some postop bleeding were on an antithrombotic medication.
Data Source: A review of 147 consecutive patients at a single surgery center.
Disclosures: Dr. Asher said he had no relevant financial conflicts.
Novel OR scheduling and staffing boosts efficiency
A more realistic approach to scheduling elective surgeries, and the staffing of operating rooms to support more efficient use, led to increased surgery volume but with less overtime, better staff satisfaction, and ultimately an increased margin for the hospital, according to the recent experience at one mid-size U.S. hospital.
The result was a surgical schedule that became "more predictable and reliable," and led to "increased capacity without increased operational costs," Dr. C. Daniel Smith said at the meeting.
"The issue is predictability and reliability, so that we can adapt [staffing and operating room needs] appropriately," said Dr. Smith, professor and chairman of the department of surgery at the Mayo Clinic in Jacksonville, Fla.
Dr. Smith and several other staffers at Mayo began by identifying a few shortcomings in the surgical scheduling methods at their hospital, a 214-bed facility that opened in 2008 with 21 operating rooms, 28 ICUs, and about 12,000 surgeries done each year. Operating-room volume was highly variable, fluctuating between 35 and 62 cases per day. "That’s a huge day-to-day variability. If you staff for the average you’ll be chronically understaffed or overstaffed each day," he noted.
Another big problem was that the schedule had no way to easily insert the inevitable emergency cases without severely disrupting elective cases. "As emergencies showed up, [elective] cases got bumped past 5 p.m. We had on average five cases a day where everything [scheduled] got changed. Surgeons no longer could operate with the team they expected."
When they began their self assessment in 2009, they found that their operating rooms had less than 65% utilization during prime-time hours, they routinely paid overtime for 15 full-time equivalent employees, and they had low levels of surgeon and staff satisfaction with OR management and efficiency.
They set out in 2010 to revamp their OR scheduling and staffing with the goal of increasing case volume, reducing overtime, and maintaining emergency surgeries without disrupting the elective schedule. Their strategy included designating some ORs that would handle only emergency and urgent cases. They also did not allow elective cases to be scheduled if they would finish later than 5 p.m., unless it was for a group of surgeons who consistently were used to working until the later time.
"We told surgeons [who wanted to schedule an elective case late in the day] that they would need to move the case to another day," Dr. Smith said. "The surgeons we struggled with the most were the high-volume, academic surgeons who try to handle 12 months of cases in 9 months. They run two rooms and a clustered bay. We told them that they could no longer bleed out into other rooms. They could no longer book a room that ended at 10 a.m. and then show up that morning and pound their fists and say they wanted more empty rooms to push their cases out to.
"That was a big cultural change, and it’s been hard," Dr. Smith said.
Another, ongoing facet has been to "reengineer flow out of the OR and into the hospital," he added. The goal is to "have a streamlined path from entry into the surgical practice to the postop bed and then out the door as quickly as possible."
During November 2010 to October 2011, the first year after full implementation of the revamped scheduling process, the number of surgical cases increased by nearly 500 patients, a 4% rise; prime time OR use rose from 61% to 64%; the number of overtime full-time equivalents fell by two staffers (27%) – a cost savings of more than $111,000; daily elective room changes fell by 69%; staff turnover, considered a measure of satisfaction, dropped from 20% to 12%; and the total net margin to the hospital rose by nearly $5 million, a 5% increase. That happened despite the addition of 15 full-time equivalents to the surgical staff, which boosted payroll by nearly $800,000, because the number of cases rose while the costs per case remained unchanged.
Despite these successes, Dr. Smith had a warning for any surgery department considering similar changes because of the substantial cultural change it involves: "We spent a year planning this before implementation, and that wasn’t enough time. There is never enough time. You need to take your time, find a group that likes this, get early wins, and build on that. It will take years to fully effect this; take it one step at a time."
Dr. Smith said that he had no disclosures.
At my institution, Johns Hopkins, we recently moved into new operating rooms, and we wanted to change the way we did things in the department just like at Mayo.
In general, the way surgeries are scheduled at U.S. hospitals is a mess. As a resident at UCLA, I would sit outside the OR at 6 a.m. to get a case scheduled for that day.

|
|
The key element we identified for the changes we made at Hopkins were preparation and buy-in from all the staff: nursing, anesthesia, and surgery. We needed champions in each of these areas, and we had to stay focused on what was best for patients. Before this, the way we ran our operating rooms – and the way most hospitals still run their ORs – is based on what is best for the surgeons. Changing the culture in OR scheduling to focus on patients is essential for making the operation efficient.
We now do a great deal of negotiation on whether a patient needs surgery right away. This requires transparency about the real urgency of a case.
Redesigning the culture and eliminating block time was painful. But we now do six to eight additional cases each day, and we have seen reductions in costs and gains in efficiency similar to what Dr. Smith reports happened at Mayo. Fewer elective cases get interrupted, while the urgent cases still get scheduled.
Once we began this effort, our elective ORs increased to greater than 80% use, and now we are up to 95%. We still offer block time, but once OR usage falls below the 80% level, we take block time away; some surgeons on our staff believe that I am evil because I took away their block time. We also have five emergency ORs that run with about a 50% usage rate.
We now find that we run out of beds for the postsurgical patients before we run out of available ORs. Our next challenge is how to discharge postoperative patients faster.
Dr. Julie A. Freischlag is a professor and chairman of the department of surgery at Johns Hopkins University, Baltimore. She made these comments as a designated discussant for Dr. Smith’s report. She had no disclosures.
At my institution, Johns Hopkins, we recently moved into new operating rooms, and we wanted to change the way we did things in the department just like at Mayo.
In general, the way surgeries are scheduled at U.S. hospitals is a mess. As a resident at UCLA, I would sit outside the OR at 6 a.m. to get a case scheduled for that day.

|
|
The key element we identified for the changes we made at Hopkins were preparation and buy-in from all the staff: nursing, anesthesia, and surgery. We needed champions in each of these areas, and we had to stay focused on what was best for patients. Before this, the way we ran our operating rooms – and the way most hospitals still run their ORs – is based on what is best for the surgeons. Changing the culture in OR scheduling to focus on patients is essential for making the operation efficient.
We now do a great deal of negotiation on whether a patient needs surgery right away. This requires transparency about the real urgency of a case.
Redesigning the culture and eliminating block time was painful. But we now do six to eight additional cases each day, and we have seen reductions in costs and gains in efficiency similar to what Dr. Smith reports happened at Mayo. Fewer elective cases get interrupted, while the urgent cases still get scheduled.
Once we began this effort, our elective ORs increased to greater than 80% use, and now we are up to 95%. We still offer block time, but once OR usage falls below the 80% level, we take block time away; some surgeons on our staff believe that I am evil because I took away their block time. We also have five emergency ORs that run with about a 50% usage rate.
We now find that we run out of beds for the postsurgical patients before we run out of available ORs. Our next challenge is how to discharge postoperative patients faster.
Dr. Julie A. Freischlag is a professor and chairman of the department of surgery at Johns Hopkins University, Baltimore. She made these comments as a designated discussant for Dr. Smith’s report. She had no disclosures.
At my institution, Johns Hopkins, we recently moved into new operating rooms, and we wanted to change the way we did things in the department just like at Mayo.
In general, the way surgeries are scheduled at U.S. hospitals is a mess. As a resident at UCLA, I would sit outside the OR at 6 a.m. to get a case scheduled for that day.

|
|
The key element we identified for the changes we made at Hopkins were preparation and buy-in from all the staff: nursing, anesthesia, and surgery. We needed champions in each of these areas, and we had to stay focused on what was best for patients. Before this, the way we ran our operating rooms – and the way most hospitals still run their ORs – is based on what is best for the surgeons. Changing the culture in OR scheduling to focus on patients is essential for making the operation efficient.
We now do a great deal of negotiation on whether a patient needs surgery right away. This requires transparency about the real urgency of a case.
Redesigning the culture and eliminating block time was painful. But we now do six to eight additional cases each day, and we have seen reductions in costs and gains in efficiency similar to what Dr. Smith reports happened at Mayo. Fewer elective cases get interrupted, while the urgent cases still get scheduled.
Once we began this effort, our elective ORs increased to greater than 80% use, and now we are up to 95%. We still offer block time, but once OR usage falls below the 80% level, we take block time away; some surgeons on our staff believe that I am evil because I took away their block time. We also have five emergency ORs that run with about a 50% usage rate.
We now find that we run out of beds for the postsurgical patients before we run out of available ORs. Our next challenge is how to discharge postoperative patients faster.
Dr. Julie A. Freischlag is a professor and chairman of the department of surgery at Johns Hopkins University, Baltimore. She made these comments as a designated discussant for Dr. Smith’s report. She had no disclosures.
A more realistic approach to scheduling elective surgeries, and the staffing of operating rooms to support more efficient use, led to increased surgery volume but with less overtime, better staff satisfaction, and ultimately an increased margin for the hospital, according to the recent experience at one mid-size U.S. hospital.
The result was a surgical schedule that became "more predictable and reliable," and led to "increased capacity without increased operational costs," Dr. C. Daniel Smith said at the meeting.
"The issue is predictability and reliability, so that we can adapt [staffing and operating room needs] appropriately," said Dr. Smith, professor and chairman of the department of surgery at the Mayo Clinic in Jacksonville, Fla.
Dr. Smith and several other staffers at Mayo began by identifying a few shortcomings in the surgical scheduling methods at their hospital, a 214-bed facility that opened in 2008 with 21 operating rooms, 28 ICUs, and about 12,000 surgeries done each year. Operating-room volume was highly variable, fluctuating between 35 and 62 cases per day. "That’s a huge day-to-day variability. If you staff for the average you’ll be chronically understaffed or overstaffed each day," he noted.
Another big problem was that the schedule had no way to easily insert the inevitable emergency cases without severely disrupting elective cases. "As emergencies showed up, [elective] cases got bumped past 5 p.m. We had on average five cases a day where everything [scheduled] got changed. Surgeons no longer could operate with the team they expected."
When they began their self assessment in 2009, they found that their operating rooms had less than 65% utilization during prime-time hours, they routinely paid overtime for 15 full-time equivalent employees, and they had low levels of surgeon and staff satisfaction with OR management and efficiency.
They set out in 2010 to revamp their OR scheduling and staffing with the goal of increasing case volume, reducing overtime, and maintaining emergency surgeries without disrupting the elective schedule. Their strategy included designating some ORs that would handle only emergency and urgent cases. They also did not allow elective cases to be scheduled if they would finish later than 5 p.m., unless it was for a group of surgeons who consistently were used to working until the later time.
"We told surgeons [who wanted to schedule an elective case late in the day] that they would need to move the case to another day," Dr. Smith said. "The surgeons we struggled with the most were the high-volume, academic surgeons who try to handle 12 months of cases in 9 months. They run two rooms and a clustered bay. We told them that they could no longer bleed out into other rooms. They could no longer book a room that ended at 10 a.m. and then show up that morning and pound their fists and say they wanted more empty rooms to push their cases out to.
"That was a big cultural change, and it’s been hard," Dr. Smith said.
Another, ongoing facet has been to "reengineer flow out of the OR and into the hospital," he added. The goal is to "have a streamlined path from entry into the surgical practice to the postop bed and then out the door as quickly as possible."
During November 2010 to October 2011, the first year after full implementation of the revamped scheduling process, the number of surgical cases increased by nearly 500 patients, a 4% rise; prime time OR use rose from 61% to 64%; the number of overtime full-time equivalents fell by two staffers (27%) – a cost savings of more than $111,000; daily elective room changes fell by 69%; staff turnover, considered a measure of satisfaction, dropped from 20% to 12%; and the total net margin to the hospital rose by nearly $5 million, a 5% increase. That happened despite the addition of 15 full-time equivalents to the surgical staff, which boosted payroll by nearly $800,000, because the number of cases rose while the costs per case remained unchanged.
Despite these successes, Dr. Smith had a warning for any surgery department considering similar changes because of the substantial cultural change it involves: "We spent a year planning this before implementation, and that wasn’t enough time. There is never enough time. You need to take your time, find a group that likes this, get early wins, and build on that. It will take years to fully effect this; take it one step at a time."
Dr. Smith said that he had no disclosures.
A more realistic approach to scheduling elective surgeries, and the staffing of operating rooms to support more efficient use, led to increased surgery volume but with less overtime, better staff satisfaction, and ultimately an increased margin for the hospital, according to the recent experience at one mid-size U.S. hospital.
The result was a surgical schedule that became "more predictable and reliable," and led to "increased capacity without increased operational costs," Dr. C. Daniel Smith said at the meeting.
"The issue is predictability and reliability, so that we can adapt [staffing and operating room needs] appropriately," said Dr. Smith, professor and chairman of the department of surgery at the Mayo Clinic in Jacksonville, Fla.
Dr. Smith and several other staffers at Mayo began by identifying a few shortcomings in the surgical scheduling methods at their hospital, a 214-bed facility that opened in 2008 with 21 operating rooms, 28 ICUs, and about 12,000 surgeries done each year. Operating-room volume was highly variable, fluctuating between 35 and 62 cases per day. "That’s a huge day-to-day variability. If you staff for the average you’ll be chronically understaffed or overstaffed each day," he noted.
Another big problem was that the schedule had no way to easily insert the inevitable emergency cases without severely disrupting elective cases. "As emergencies showed up, [elective] cases got bumped past 5 p.m. We had on average five cases a day where everything [scheduled] got changed. Surgeons no longer could operate with the team they expected."
When they began their self assessment in 2009, they found that their operating rooms had less than 65% utilization during prime-time hours, they routinely paid overtime for 15 full-time equivalent employees, and they had low levels of surgeon and staff satisfaction with OR management and efficiency.
They set out in 2010 to revamp their OR scheduling and staffing with the goal of increasing case volume, reducing overtime, and maintaining emergency surgeries without disrupting the elective schedule. Their strategy included designating some ORs that would handle only emergency and urgent cases. They also did not allow elective cases to be scheduled if they would finish later than 5 p.m., unless it was for a group of surgeons who consistently were used to working until the later time.
"We told surgeons [who wanted to schedule an elective case late in the day] that they would need to move the case to another day," Dr. Smith said. "The surgeons we struggled with the most were the high-volume, academic surgeons who try to handle 12 months of cases in 9 months. They run two rooms and a clustered bay. We told them that they could no longer bleed out into other rooms. They could no longer book a room that ended at 10 a.m. and then show up that morning and pound their fists and say they wanted more empty rooms to push their cases out to.
"That was a big cultural change, and it’s been hard," Dr. Smith said.
Another, ongoing facet has been to "reengineer flow out of the OR and into the hospital," he added. The goal is to "have a streamlined path from entry into the surgical practice to the postop bed and then out the door as quickly as possible."
During November 2010 to October 2011, the first year after full implementation of the revamped scheduling process, the number of surgical cases increased by nearly 500 patients, a 4% rise; prime time OR use rose from 61% to 64%; the number of overtime full-time equivalents fell by two staffers (27%) – a cost savings of more than $111,000; daily elective room changes fell by 69%; staff turnover, considered a measure of satisfaction, dropped from 20% to 12%; and the total net margin to the hospital rose by nearly $5 million, a 5% increase. That happened despite the addition of 15 full-time equivalents to the surgical staff, which boosted payroll by nearly $800,000, because the number of cases rose while the costs per case remained unchanged.
Despite these successes, Dr. Smith had a warning for any surgery department considering similar changes because of the substantial cultural change it involves: "We spent a year planning this before implementation, and that wasn’t enough time. There is never enough time. You need to take your time, find a group that likes this, get early wins, and build on that. It will take years to fully effect this; take it one step at a time."
Dr. Smith said that he had no disclosures.
AT THE ANNUAL MEETING OF THE SOUTHERN SURGICAL ASSOCIATION
Major Finding: During the first year, surgical cases rose 4%, overtime fell 27%, staff turnover fell 8%, and hospital margin rose 5%.
Data Source: The first-year experience of new operating room staffing and scheduling procedures at one U.S. hospital.
Disclosures: Dr. Smith said that he had no disclosures.
Night on call has no effect on next-day operations
PALM BEACH, FLA. – Surgeons who performed routine, elective operations the day after they spent all night working an in-hospital shift in the trauma unit had no increased rate of complications, no need for hospital readmissions, and no mortality among their elective cases in a single-center review of 869 cases.
The findings suggest that "there remains no compelling evidence to mandate work-hour restrictions for attending general surgeons," Dr. Martin A. Croce said at the annual meeting of the Southern Surgical Association.
"No study to date has evaluated the effect of sleep deprivation on outcomes specific to the practice of general surgery. This study looked at the impact of an overnight shift in a busy trauma center on the outcomes of typical general surgery procedures performed the next day" and found no significant difference in outcomes between post-call and non–post-call surgeons with respect to postoperative complications, readmissions, or deaths, said Dr. Croce, professor of surgery and chief of the division of trauma and critical care at the University of Tennessee Health Science Center in Memphis.
"These results confirm my bias, but the study was limited by its retrospective, single-institution nature, and by only including tertiary teaching patients. Each [surgeon] must continue to use judgment when deciding whether to perform elective operations after a busy night on call," commented Dr. William G. Cioffi Jr., professor and chairman of surgery at Brown University, Providence, R.I.
"It is unclear to me that the surgical community will need to present these results because there has been no call for work-hour restrictions for attendings, nor do I feel there will be," said Dr. L.D. Britt, professor and chairman of surgery at Eastern Virginia Medical School, Norfolk.
In his study, Dr. Croce reviewed patients who underwent any of three types of general surgeries at his institution during January 2006 to April 2009 by one of the nine attending surgeons in the department. The review comprised 869 procedures: 46% were hernia repairs, 35% were cholecystectomies, and 19% were bowel operations. The primary outcomes were 30-day postsurgical mortality, 30-day postsurgical readmissions, and procedure-related complications: wound infections, seroma, injured adjacent structures, intra-abdominal abscess, hemorrhage, dehiscence, leak, or perforation of the gallbladder during laparoscopic cholecystectomy. Overall, patients had a 13% complication rate, a 5% readmission rate, and mortality of less than 1%.
Among the 869 procedures, 132 (15%) were done by surgeons following a night on call and the remaining 737 (85%) were done by surgeons not on call the prior night. In a multivariable regression analysis that controlled for patient’s age, urgency of the elective surgery, and comorbidities, the rates of complications, readmissions, and deaths were similar between those done by surgeons following a night on call and those done without working the night before, Dr. Croce reported.
Further analyses showed no significant difference between the two surgeon subgroups when the types of procedures were broken down into hernia repair, cholecystectomy, or bowel operation.
A limitation of the study was that the findings apply only to these three types of surgeries; they should not be extrapolated to other, more complex surgeries, said Dr. John P. Sharpe, a surgeon at the University of Tennessee who collaborated with Dr. Croce on the study.
Dr. Croce, Dr. Cioffi, Dr. Britt, and Dr. Sharpe had no disclosures.
PALM BEACH, FLA. – Surgeons who performed routine, elective operations the day after they spent all night working an in-hospital shift in the trauma unit had no increased rate of complications, no need for hospital readmissions, and no mortality among their elective cases in a single-center review of 869 cases.
The findings suggest that "there remains no compelling evidence to mandate work-hour restrictions for attending general surgeons," Dr. Martin A. Croce said at the annual meeting of the Southern Surgical Association.
"No study to date has evaluated the effect of sleep deprivation on outcomes specific to the practice of general surgery. This study looked at the impact of an overnight shift in a busy trauma center on the outcomes of typical general surgery procedures performed the next day" and found no significant difference in outcomes between post-call and non–post-call surgeons with respect to postoperative complications, readmissions, or deaths, said Dr. Croce, professor of surgery and chief of the division of trauma and critical care at the University of Tennessee Health Science Center in Memphis.
"These results confirm my bias, but the study was limited by its retrospective, single-institution nature, and by only including tertiary teaching patients. Each [surgeon] must continue to use judgment when deciding whether to perform elective operations after a busy night on call," commented Dr. William G. Cioffi Jr., professor and chairman of surgery at Brown University, Providence, R.I.
"It is unclear to me that the surgical community will need to present these results because there has been no call for work-hour restrictions for attendings, nor do I feel there will be," said Dr. L.D. Britt, professor and chairman of surgery at Eastern Virginia Medical School, Norfolk.
In his study, Dr. Croce reviewed patients who underwent any of three types of general surgeries at his institution during January 2006 to April 2009 by one of the nine attending surgeons in the department. The review comprised 869 procedures: 46% were hernia repairs, 35% were cholecystectomies, and 19% were bowel operations. The primary outcomes were 30-day postsurgical mortality, 30-day postsurgical readmissions, and procedure-related complications: wound infections, seroma, injured adjacent structures, intra-abdominal abscess, hemorrhage, dehiscence, leak, or perforation of the gallbladder during laparoscopic cholecystectomy. Overall, patients had a 13% complication rate, a 5% readmission rate, and mortality of less than 1%.
Among the 869 procedures, 132 (15%) were done by surgeons following a night on call and the remaining 737 (85%) were done by surgeons not on call the prior night. In a multivariable regression analysis that controlled for patient’s age, urgency of the elective surgery, and comorbidities, the rates of complications, readmissions, and deaths were similar between those done by surgeons following a night on call and those done without working the night before, Dr. Croce reported.
Further analyses showed no significant difference between the two surgeon subgroups when the types of procedures were broken down into hernia repair, cholecystectomy, or bowel operation.
A limitation of the study was that the findings apply only to these three types of surgeries; they should not be extrapolated to other, more complex surgeries, said Dr. John P. Sharpe, a surgeon at the University of Tennessee who collaborated with Dr. Croce on the study.
Dr. Croce, Dr. Cioffi, Dr. Britt, and Dr. Sharpe had no disclosures.
PALM BEACH, FLA. – Surgeons who performed routine, elective operations the day after they spent all night working an in-hospital shift in the trauma unit had no increased rate of complications, no need for hospital readmissions, and no mortality among their elective cases in a single-center review of 869 cases.
The findings suggest that "there remains no compelling evidence to mandate work-hour restrictions for attending general surgeons," Dr. Martin A. Croce said at the annual meeting of the Southern Surgical Association.
"No study to date has evaluated the effect of sleep deprivation on outcomes specific to the practice of general surgery. This study looked at the impact of an overnight shift in a busy trauma center on the outcomes of typical general surgery procedures performed the next day" and found no significant difference in outcomes between post-call and non–post-call surgeons with respect to postoperative complications, readmissions, or deaths, said Dr. Croce, professor of surgery and chief of the division of trauma and critical care at the University of Tennessee Health Science Center in Memphis.
"These results confirm my bias, but the study was limited by its retrospective, single-institution nature, and by only including tertiary teaching patients. Each [surgeon] must continue to use judgment when deciding whether to perform elective operations after a busy night on call," commented Dr. William G. Cioffi Jr., professor and chairman of surgery at Brown University, Providence, R.I.
"It is unclear to me that the surgical community will need to present these results because there has been no call for work-hour restrictions for attendings, nor do I feel there will be," said Dr. L.D. Britt, professor and chairman of surgery at Eastern Virginia Medical School, Norfolk.
In his study, Dr. Croce reviewed patients who underwent any of three types of general surgeries at his institution during January 2006 to April 2009 by one of the nine attending surgeons in the department. The review comprised 869 procedures: 46% were hernia repairs, 35% were cholecystectomies, and 19% were bowel operations. The primary outcomes were 30-day postsurgical mortality, 30-day postsurgical readmissions, and procedure-related complications: wound infections, seroma, injured adjacent structures, intra-abdominal abscess, hemorrhage, dehiscence, leak, or perforation of the gallbladder during laparoscopic cholecystectomy. Overall, patients had a 13% complication rate, a 5% readmission rate, and mortality of less than 1%.
Among the 869 procedures, 132 (15%) were done by surgeons following a night on call and the remaining 737 (85%) were done by surgeons not on call the prior night. In a multivariable regression analysis that controlled for patient’s age, urgency of the elective surgery, and comorbidities, the rates of complications, readmissions, and deaths were similar between those done by surgeons following a night on call and those done without working the night before, Dr. Croce reported.
Further analyses showed no significant difference between the two surgeon subgroups when the types of procedures were broken down into hernia repair, cholecystectomy, or bowel operation.
A limitation of the study was that the findings apply only to these three types of surgeries; they should not be extrapolated to other, more complex surgeries, said Dr. John P. Sharpe, a surgeon at the University of Tennessee who collaborated with Dr. Croce on the study.
Dr. Croce, Dr. Cioffi, Dr. Britt, and Dr. Sharpe had no disclosures.
AT THE ANNUAL MEETING OF THE SOUTHERN SURGICAL ASSOCIATION
Major Finding: General surgeons who performed 132 elective operations after a night on call had no increased complications, readmissions, or deaths, compared with surgeons not on call the previous night (737 surgeries).
Data Source: A review of 869 surgeries at one U.S. center during 2006-2009.
Disclosures: Dr. Croce, Dr. Cioffi, Dr. Britt, and Dr. Sharpe had no disclosures.
FDA proposal encourages development of abuse deterrent opioids
Encouraging manufacturers to develop opioid products that are formulated to deter abuse is one of the main goals of a new draft guidance released Jan. 9 by the Food and Drug Administration.
"Guidance for Industry: Abuse-Deterrent Opioids – Evaluation and Labeling" provides information for companies interested in developing formulations of opioids with potential abuse deterrent properties, Dr. Douglas Throckmorton, deputy director for regulatory programs in the FDA’s Center for Drug Evaluation and Research, said at a briefing.
The document explains the agency’s views on the types of studies that should be conducted to show that a product can deter abuse, how those studies will be evaluated by the agency, and what labeling claims would be allowed based on the results of the FDA’s evaluations of those data. The ability to add abuse deterrent claims to a product’s label is expected to encourage companies to develop these products, he said.
The technology of formulations that deter abuse and the clinical epidemiology and statistical methods used to evaluate the potential of such products for abuse deterrence is a relatively new area of research, and the FDA plans "to take a flexible approach" in evaluating these products as the science evolves, Dr. Throckmorton said.
During the briefing, he pointed out that the guidance is one of the components of the FDA’s effort to prevent prescription drug abuse and misuse, "while ensuring patients in pain continue to have access to these medicines." In 2009, almost 425,000 visits to emergency departments involving nonmedical or inappropriate use of opioids and an estimated 15,600 deaths in the United States were attributed to opioid products, he said.
This draft guidance fulfills mandates under the Food and Drug Administration Safety and Innovation Act and the Office of National Drug Control Policy’s Prescription Drug Abuse Prevention Plan.
The FDA statement announcing the guidance explains that abuse deterrent formulations "target the known or expected routes of abuse, such as crushing in order to snort or dissolving in order to inject, for the specific opioid drug substance in that formulation" and that the agency believes these formulations have "promise to help reduce prescription drug abuse."
Two opioid products with abuse deterrent formulations are currently available: Since 2010, OxyContin (controlled-release oxycodone) has been available in a formulation that prevents it from being chewed, crushed, or dissolved. A crush-resistant formulation of hydromorphone (Opana ER) was approved in September 2012. But the prescribing information for these products do not include claims regarding abuse deterrence properties.
The lack of abuse deterrent features was the main reason an FDA advisory panel recommended against approval of an extended-release, single-ingredient capsule formulation of hydrocodone at a meeting in December. Without such a feature, the product would quickly become widely abused once it was approved and marketed, some of the panelists predicted.
The FDA plans to hold a public meeting to discuss public feedback on the draft guidance.
During the briefing, Dr. Throckmorton said that the FDA had not yet decided what to do about generic formulations of opioids that have no abuse deterrent properties.
The guidance "is an important part of a larger effort by FDA aimed at preventing prescription drug abuse and misuse," and the agency is "extremely concerned about the inappropriate use of prescription opioids, which is a major public health challenge for our nation," FDA commissioner Dr. Margaret Hamburg said in the statement.
Comments and suggestions can be submitted to the FDA within 60 days of publication in the Federal Register, to the Division of Dockets Management (HFA-305), Food and Drug Administration, 5630 Fishers Lane, Room 1061, Rockville, Md., 20852 , with the docket number listed.
Encouraging manufacturers to develop opioid products that are formulated to deter abuse is one of the main goals of a new draft guidance released Jan. 9 by the Food and Drug Administration.
"Guidance for Industry: Abuse-Deterrent Opioids – Evaluation and Labeling" provides information for companies interested in developing formulations of opioids with potential abuse deterrent properties, Dr. Douglas Throckmorton, deputy director for regulatory programs in the FDA’s Center for Drug Evaluation and Research, said at a briefing.
The document explains the agency’s views on the types of studies that should be conducted to show that a product can deter abuse, how those studies will be evaluated by the agency, and what labeling claims would be allowed based on the results of the FDA’s evaluations of those data. The ability to add abuse deterrent claims to a product’s label is expected to encourage companies to develop these products, he said.
The technology of formulations that deter abuse and the clinical epidemiology and statistical methods used to evaluate the potential of such products for abuse deterrence is a relatively new area of research, and the FDA plans "to take a flexible approach" in evaluating these products as the science evolves, Dr. Throckmorton said.
During the briefing, he pointed out that the guidance is one of the components of the FDA’s effort to prevent prescription drug abuse and misuse, "while ensuring patients in pain continue to have access to these medicines." In 2009, almost 425,000 visits to emergency departments involving nonmedical or inappropriate use of opioids and an estimated 15,600 deaths in the United States were attributed to opioid products, he said.
This draft guidance fulfills mandates under the Food and Drug Administration Safety and Innovation Act and the Office of National Drug Control Policy’s Prescription Drug Abuse Prevention Plan.
The FDA statement announcing the guidance explains that abuse deterrent formulations "target the known or expected routes of abuse, such as crushing in order to snort or dissolving in order to inject, for the specific opioid drug substance in that formulation" and that the agency believes these formulations have "promise to help reduce prescription drug abuse."
Two opioid products with abuse deterrent formulations are currently available: Since 2010, OxyContin (controlled-release oxycodone) has been available in a formulation that prevents it from being chewed, crushed, or dissolved. A crush-resistant formulation of hydromorphone (Opana ER) was approved in September 2012. But the prescribing information for these products do not include claims regarding abuse deterrence properties.
The lack of abuse deterrent features was the main reason an FDA advisory panel recommended against approval of an extended-release, single-ingredient capsule formulation of hydrocodone at a meeting in December. Without such a feature, the product would quickly become widely abused once it was approved and marketed, some of the panelists predicted.
The FDA plans to hold a public meeting to discuss public feedback on the draft guidance.
During the briefing, Dr. Throckmorton said that the FDA had not yet decided what to do about generic formulations of opioids that have no abuse deterrent properties.
The guidance "is an important part of a larger effort by FDA aimed at preventing prescription drug abuse and misuse," and the agency is "extremely concerned about the inappropriate use of prescription opioids, which is a major public health challenge for our nation," FDA commissioner Dr. Margaret Hamburg said in the statement.
Comments and suggestions can be submitted to the FDA within 60 days of publication in the Federal Register, to the Division of Dockets Management (HFA-305), Food and Drug Administration, 5630 Fishers Lane, Room 1061, Rockville, Md., 20852 , with the docket number listed.
Encouraging manufacturers to develop opioid products that are formulated to deter abuse is one of the main goals of a new draft guidance released Jan. 9 by the Food and Drug Administration.
"Guidance for Industry: Abuse-Deterrent Opioids – Evaluation and Labeling" provides information for companies interested in developing formulations of opioids with potential abuse deterrent properties, Dr. Douglas Throckmorton, deputy director for regulatory programs in the FDA’s Center for Drug Evaluation and Research, said at a briefing.
The document explains the agency’s views on the types of studies that should be conducted to show that a product can deter abuse, how those studies will be evaluated by the agency, and what labeling claims would be allowed based on the results of the FDA’s evaluations of those data. The ability to add abuse deterrent claims to a product’s label is expected to encourage companies to develop these products, he said.
The technology of formulations that deter abuse and the clinical epidemiology and statistical methods used to evaluate the potential of such products for abuse deterrence is a relatively new area of research, and the FDA plans "to take a flexible approach" in evaluating these products as the science evolves, Dr. Throckmorton said.
During the briefing, he pointed out that the guidance is one of the components of the FDA’s effort to prevent prescription drug abuse and misuse, "while ensuring patients in pain continue to have access to these medicines." In 2009, almost 425,000 visits to emergency departments involving nonmedical or inappropriate use of opioids and an estimated 15,600 deaths in the United States were attributed to opioid products, he said.
This draft guidance fulfills mandates under the Food and Drug Administration Safety and Innovation Act and the Office of National Drug Control Policy’s Prescription Drug Abuse Prevention Plan.
The FDA statement announcing the guidance explains that abuse deterrent formulations "target the known or expected routes of abuse, such as crushing in order to snort or dissolving in order to inject, for the specific opioid drug substance in that formulation" and that the agency believes these formulations have "promise to help reduce prescription drug abuse."
Two opioid products with abuse deterrent formulations are currently available: Since 2010, OxyContin (controlled-release oxycodone) has been available in a formulation that prevents it from being chewed, crushed, or dissolved. A crush-resistant formulation of hydromorphone (Opana ER) was approved in September 2012. But the prescribing information for these products do not include claims regarding abuse deterrence properties.
The lack of abuse deterrent features was the main reason an FDA advisory panel recommended against approval of an extended-release, single-ingredient capsule formulation of hydrocodone at a meeting in December. Without such a feature, the product would quickly become widely abused once it was approved and marketed, some of the panelists predicted.
The FDA plans to hold a public meeting to discuss public feedback on the draft guidance.
During the briefing, Dr. Throckmorton said that the FDA had not yet decided what to do about generic formulations of opioids that have no abuse deterrent properties.
The guidance "is an important part of a larger effort by FDA aimed at preventing prescription drug abuse and misuse," and the agency is "extremely concerned about the inappropriate use of prescription opioids, which is a major public health challenge for our nation," FDA commissioner Dr. Margaret Hamburg said in the statement.
Comments and suggestions can be submitted to the FDA within 60 days of publication in the Federal Register, to the Division of Dockets Management (HFA-305), Food and Drug Administration, 5630 Fishers Lane, Room 1061, Rockville, Md., 20852 , with the docket number listed.