User login
Implementation and Evaluation of a 90-Minute Rituximab Infusion Protocol at the Richard L. Roudebush VA Medical Center
Rituximab is a genetically engineered chimeric immunoglobulin G1 monoclonal antibody. It functions by binding to the CD20 antigen on the surface of B-cell lymphocytes, leading to complement-dependent cytotoxicity and antibody-dependent cellular cytotoxicity.1 The US Food and Drug Administration approved this therapy to treat patients with B-cell non-Hodgkin lymphoma and chronic lymphocytic leukemia, along with other nonmalignant indications, including pemphigus vulgaris and rheumatoid arthritis (RA). Historically, a significant amount of time and labor on behalf of medical personnel has been required to administer rituximab according to the original manufacturer’s labeling due to the boxed warning associated with infusion-related reactions (IRRs).2
Originally, the elongated infusion times that were recommended for rituximab were largely due to the perceived risk of serious infusion-related adverse drug reactions. Slower infusion times should reduce the risk of a reaction and are considered to be a good option for those patients who are at a high risk of having a severe IRRs to rituximab. Examples of high-risk patients from previous studies include those with significant cardiovascular disease, a circulating lymphocyte count ≤ 5,000/µL at the start of infusion, and those who have previously had a reaction to rituximab.3-5 In appropriate patients, research has shown a decreasing incidence of all-grade IRRs for patients who are prescribed rituximab as they receive more doses of the drug.2,6 The ability to identify suitable patients for 90-minute infusions of rituximab and the prospect of better health system resource utilization has led investigators to study the effects of shortened infusion times.
The RATE trial addressed this subject with a phase 3 safety study on the effects of a 90-minute rituximab infusion for patients with previously untreated diffuse large B-cell and follicular lymphoma.3 The patients in this study received their first dose of rituximab using the traditional infusion approach. If it was well-tolerated, they received subsequent rituximab infusions using a 90-minute protocol. Only 1.1% of patients who had previously received a rituximab infusion developed a grade 3 or 4 IRR when receiving a faster infusion of the drug for the first time.3 This result led to the addition of instructions for a 90-minute infusion to the package insert.2
In contrast to the RATE trial, the RATE-RA trial evaluated the incidence of IRRs in patients who received rituximab for nonmalignant indications. This study assessed patients with RA receiving rituximab for > 120 minutes. The authors reported 0.6% of the patients in the study developed a grade 3 or 4 IRR associated with the first 120-minute infusion of the medication.5 The researchers concluded that rituximab can be administered at a faster rate during second and subsequent infusions in patients who have been shown to tolerate traditional infusions without increasing the risk or severity of IRRs.5
The US Department of Veterans Affairs (VA) Richard L. Roudebush VA Medical Center (RLRVAMC) in Indianapolis, Indiana, uses traditional directions for the infusion of rituximab due to perceived tolerability and safety concerns specifically in a veteran population—even while other VA medical centers have implemented shortened infusion protocols. This also is despite the fact that available research shows rapid infusions of the drug are well tolerated in a variety of community settings.7,8 Anticipated benefits of implementing a protocol include savings in chair time at the institution’s infusion clinic along with increased nursing and patient satisfaction. This project was conducted to prepare, implement, and assess the safety of a 90-minute rituximab protocol at the RLRVAMC.
Methods
Proactive measures were required before and during the implementation of the 90-minute protocol to ensure patient safety and staff satisfaction. Updates to the RLRVAMC policy for the management of medical emergencies within the infusion center were reviewed and approved by the acute care committee and nursing leadership. A protocol was developed to identify eligible patients, outline the hypersensitivity protocol, instruct pharmacy personnel on admixture preparation, and provide a titration schedule based on dose. Order sets also were created to assist health care providers (HCPs) with the prescribing of rituximab for nonantineoplastic indications. Educational materials were crafted to assist with order verification, product preparation, labeling, and programming of infusion pumps. Live education was provided for physicians, pharmacists, and nurses to ensure smooth implementation of the protocol and appropriate management of medical emergencies based on the updated policy.
Study Design
Nursing staff in the infusion clinic were surveyed once before a live education session and again after the conclusion of the study. The purpose of the survey was to assess the prior experience and current comfort level of the nursing staff with administering rituximab over 90 minutes. Nurses were asked the following questions: (1) Do you have prior experience administering rituximab via 90-minute infusion; and (2) do you feel comfortable administering rituximab via 90-minute infusion?
A weekly report of patients who received rituximab between November 1, 2018 through April 1, 2019 at the RLRVAMC was generated. HCPs were alerted to eligible patients based on protocol requirements. The HCPs then made the final determination and entered orders accordingly.
This study was a retrospective chart review of all who patients received a rapid infusion of rituximab. Patients who were included if they were aged ≥ 18 years, received rituximab infusions in the RLRVAMC infusion clinic, had an absolute lymphocyte count ≤ 5,000/mm3 at the time of their rapid infusions, had no significant baseline cardiovascular disease or respiratory compromise, and had no prior grade 3 or 4 rituximab IRRs as defined by Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0.9 This study was a quality improvement initiative and considered exempt by the institutional review board. All data were deidentified and secured to ensure patient privacy.
The primary endpoint for this study was the incidence of grade 3 or 4 IRRs associated with the rapid infusion of rituximab. Secondary endpoints included the proportion of patients who experienced a grade 3 or 4 infusion reaction, who received proper treatment according to the institution’s hypersensitivity protocol, savings in infusion clinic chair time, and nursing satisfaction with education and implementation of the rapid infusion rituximab protocol.
The following data were collected for all included patients: demographics, lactic acid dehydrogenase level, white blood cell count, and absolute lymphocyte count prior to rituximab infusion, indication for treatment, dose of rituximab for 90-minute infusion, date of infusion, starting time, ending time, number of previous rituximab infusions within the past 3 months, symptoms of infusion reactions during rituximab infusion, and grade of any infusion reactions that occurred.
Estimated savings in infusion clinic chair time was calculated by taking the difference in time between each completed rapid infusion and the estimated amount of time it would have taken for each patient to receive a traditional infusion. The estimated amount of time for traditional infusion was determined by following the institution’s protocol for administering rituximab to patients who previously tolerated their first dose of the drug (eg, 100 mg/h starting rate and increasing by 100 mg/h every 30 minutes to a maximum infusion rate of 400 mg/h). All endpoints were analyzed using descriptive statistics.
Results
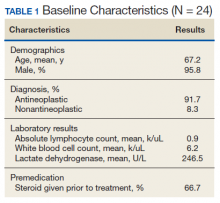
Between November 1, 2018 and April 1, 2019, 11 patients received a total of 24 rapid infusions of rituximab. The majority of patients included in the study were older males, and the most common indication for rapid infusion was follicular lymphoma (Table 1).
Primary Endpoint
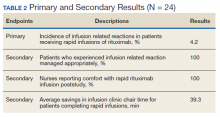
All patients who received a rapid infusion of rituximab were reviewed in the analysis of the primary and secondary endpoints. Among the 24 rapid infusions of rituximab, 1 infusion was stopped due to the patient experiencing a grade 3 IRR according to criteria from CTCAE Version 5.0. The patient was found to have dysphagia at baseline and experienced severe symptoms in the days following the first infusion that put the patient at high risk for subsequent infusion related concerns. Eligibility criteria for the 90-minute protocol were updated based on these findings. No patient experienced a grade 4 or 5 IRR. The remaining 23 infusions were well tolerated by the patients with no clinically significant events.
Secondary Endpoints
The patient who experienced a grade 3 IRR to rituximab received proper treatment by infusion clinic nurses according to the RLRVAMC hypersensitivity protocol. Patients who received rapid infusions of rituximab had a mean length of infusion of 95.0 minutes. This was in contrast to the mean time of each patient’s previous nonrapid infusion of 134.3 minutes. The difference between the 2 values equated to a savings in infusion clinic chair mean time of 39.3 minutes per patient.
Nurses were asked whether they had prior experience administering rituximab via 90-minute infusion and whether they felt comfortable administering a 90-minute rituximab infusion. Before the live education session, none of the nurses surveyed had prior experience or felt comfortable administering rituximab over 90 minutes. When the nurses were surveyed poststudy, all reported that they were experienced administering rituximab and felt comfortable with the process (Table 2).
Discussion
The infusion of rituximab has been associated with significant challenges related to the time and labor required. Although a vast number of institutions across the country now infuse the medication over an abbreviated time, HCP concerns for patient safety and appropriate use of hypersensitivity protocol in a veteran population delayed implementation at RLRVAMC. The results from this quality improvement initiative highlight the positive impact of the proactive measures that were used to implement the rapid infusion protocol for rituximab on improving HCP prescribing rates, nursing satisfaction, and appropriate management of IRRs.
Rapid infusion saved on average 39.3 minutes per patient in infusion clinic chair time. Each successful rapid infusion of rituximab potentially opened additional time in clinic for ≥ 1 patients to receive an infusion therapy. The RLRVAMC usually operated at maximum capacity, so the ability to accommodate more patients helped decrease hospital admittances for time-sensitive infusions.
The initial criteria used to screen patients to determine whether a rapid infusion of rituximab would be appropriate was based on inclusion and exclusion criteria for past studies on the same subject.3-5 The incidence of hypersensitivity reactions associated with study participants who received rapid rituximab infusions also resembles past research done on the subject, which is important to note due to prior misconceptions of staff at the institution of a higher risk of reaction in this specific veteran population. One patient with RA experienced a grade 3 IRR in this study. Although this patient met the original inclusion criteria, the patient had baseline dysphagia, and following the first infusion, reported to the emergency department (ED) with symptoms of delayed anaphylaxis. In this case, the order for rapid infusion was placed in advance and the prescriber was unaware of the ED visit. Based on this event, eligibility criteria for 90-minute rituximab infusions were updated to include additional information specifying that candidates for a rapid infusion also may have no baseline airway compromise. This hypersensitivity reaction also highlighted the need for decision support technology to assist HCPs in patient selection as well as empowering nursing and pharmacy staff to identify concerns once they place orders.
Over the course of the study, investigators assisted the HCPs with preparation of orders for the rapid infusion of rituximab for antineoplastic indications. Due to feasibility issues with this practice moving forward, order sets containing rituximab were updated to include a 90-minute option. This created a more standardized process that allowed HCPs to screen potential patients on their own. The expectation is that HCPs will be more likely to order 90-minute infusions for eligible patients in the future with this efficient and safer process.
Limitations
The small sample size in this study was a limitation. Retrospective data related to the management of infusion reactions and length of infusions were collected from nursing notes. The prospective use of a standardized evaluation tool for adverse drug reactions as well as bar code medication administration technology would improve the data available for this study. Additional studies also would be useful to validate the results.
Conclusions
The proactive measures that were used to implement the rapid infusion rituximab protocol improved HCP prescribing rates, nursing satisfaction, and the management of IRRs. Potential time savings with each infusion was significant. This study confirmed appropriateness of rapid administration of rituximab in this veteran population and has increased interest in implementing other rapid infusion protocols. Protocols, education, and order sets are being developed for daratumumab and infliximab.
1. Feugier P. A review of rituximab, the first anti-CD20 monoclonal antibody used in the treatment of B non-Hodgkin’s lymphomas. Future Oncol. 2015;11(9):1327-1342. doi:10.2217/fon.15.57
2. Rituxan [package insert]. South San Francisco, CA: Genentech; 2016.
3. Dakhil S, Hermann R, Schreeder MT, et al. Phase III safety study of rituximab administered as a 90-minute infusion in patients with previously untreated diffuse large B-cell and follicular lymphoma. Leuk Lymphoma. 2014;55(10):2335-2340. doi:10.3109/10428194.2013.877135
4. Dotson E, Crawford B, Phillips G, Jones J. Sixty-minute infusion rituximab protocol allows for safe and efficient workflow. Support Care Cancer. 2016;24(3):1125-1129. doi:10.1007/s00520-015-2869-4
5. Pritchard CH, Greenwald MW, Kremer JM, et al. Safety of infusing rituximab at a more rapid rate in patients with rheumatoid arthritis: results from the RATE-RA study. BMC Musculoskelet Disord. 2014;15:177. doi:10.1186/1471-2474-15-177
6. Hainsworth JD, Litchy S, Barton JH, et al. Single-agent rituximab as first-line and maintenance treatment for patients with chronic lymphocytic leukemia or small lymphocytic lymphoma: a phase II trial of the Minnie Pearl Cancer Research Network. J Clin Oncol. 2003;21(9):1746-1751. doi:10.1200/JCO.2003.09.027
7. Can M, Alibaz-Öner F, Yılmaz-Öner S, Atagündüz P, Înanç N, Direskeneli H. Accelerated infusion rates of rituximab are well tolerated and safe in rheumatology practice: a single-centre experience. Clin Rheumatol. 2013;32(1):87-90. doi:10.1007/s10067-012-2094-1
8. Sehn LH, Donaldson J, Filewich A, et al. Rapid infusion rituximab in combination with corticosteroid-containing chemotherapy or as maintenance therapy is well tolerated and can safely be delivered in the community setting. Blood. 2007;109(10):4171-4173. doi:10.1182/blood-2006-11-059469
9. National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE). https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm. Updated March 27 2020. Accessed June 15, 2020.
Rituximab is a genetically engineered chimeric immunoglobulin G1 monoclonal antibody. It functions by binding to the CD20 antigen on the surface of B-cell lymphocytes, leading to complement-dependent cytotoxicity and antibody-dependent cellular cytotoxicity.1 The US Food and Drug Administration approved this therapy to treat patients with B-cell non-Hodgkin lymphoma and chronic lymphocytic leukemia, along with other nonmalignant indications, including pemphigus vulgaris and rheumatoid arthritis (RA). Historically, a significant amount of time and labor on behalf of medical personnel has been required to administer rituximab according to the original manufacturer’s labeling due to the boxed warning associated with infusion-related reactions (IRRs).2
Originally, the elongated infusion times that were recommended for rituximab were largely due to the perceived risk of serious infusion-related adverse drug reactions. Slower infusion times should reduce the risk of a reaction and are considered to be a good option for those patients who are at a high risk of having a severe IRRs to rituximab. Examples of high-risk patients from previous studies include those with significant cardiovascular disease, a circulating lymphocyte count ≤ 5,000/µL at the start of infusion, and those who have previously had a reaction to rituximab.3-5 In appropriate patients, research has shown a decreasing incidence of all-grade IRRs for patients who are prescribed rituximab as they receive more doses of the drug.2,6 The ability to identify suitable patients for 90-minute infusions of rituximab and the prospect of better health system resource utilization has led investigators to study the effects of shortened infusion times.
The RATE trial addressed this subject with a phase 3 safety study on the effects of a 90-minute rituximab infusion for patients with previously untreated diffuse large B-cell and follicular lymphoma.3 The patients in this study received their first dose of rituximab using the traditional infusion approach. If it was well-tolerated, they received subsequent rituximab infusions using a 90-minute protocol. Only 1.1% of patients who had previously received a rituximab infusion developed a grade 3 or 4 IRR when receiving a faster infusion of the drug for the first time.3 This result led to the addition of instructions for a 90-minute infusion to the package insert.2
In contrast to the RATE trial, the RATE-RA trial evaluated the incidence of IRRs in patients who received rituximab for nonmalignant indications. This study assessed patients with RA receiving rituximab for > 120 minutes. The authors reported 0.6% of the patients in the study developed a grade 3 or 4 IRR associated with the first 120-minute infusion of the medication.5 The researchers concluded that rituximab can be administered at a faster rate during second and subsequent infusions in patients who have been shown to tolerate traditional infusions without increasing the risk or severity of IRRs.5
The US Department of Veterans Affairs (VA) Richard L. Roudebush VA Medical Center (RLRVAMC) in Indianapolis, Indiana, uses traditional directions for the infusion of rituximab due to perceived tolerability and safety concerns specifically in a veteran population—even while other VA medical centers have implemented shortened infusion protocols. This also is despite the fact that available research shows rapid infusions of the drug are well tolerated in a variety of community settings.7,8 Anticipated benefits of implementing a protocol include savings in chair time at the institution’s infusion clinic along with increased nursing and patient satisfaction. This project was conducted to prepare, implement, and assess the safety of a 90-minute rituximab protocol at the RLRVAMC.
Methods
Proactive measures were required before and during the implementation of the 90-minute protocol to ensure patient safety and staff satisfaction. Updates to the RLRVAMC policy for the management of medical emergencies within the infusion center were reviewed and approved by the acute care committee and nursing leadership. A protocol was developed to identify eligible patients, outline the hypersensitivity protocol, instruct pharmacy personnel on admixture preparation, and provide a titration schedule based on dose. Order sets also were created to assist health care providers (HCPs) with the prescribing of rituximab for nonantineoplastic indications. Educational materials were crafted to assist with order verification, product preparation, labeling, and programming of infusion pumps. Live education was provided for physicians, pharmacists, and nurses to ensure smooth implementation of the protocol and appropriate management of medical emergencies based on the updated policy.
Study Design
Nursing staff in the infusion clinic were surveyed once before a live education session and again after the conclusion of the study. The purpose of the survey was to assess the prior experience and current comfort level of the nursing staff with administering rituximab over 90 minutes. Nurses were asked the following questions: (1) Do you have prior experience administering rituximab via 90-minute infusion; and (2) do you feel comfortable administering rituximab via 90-minute infusion?
A weekly report of patients who received rituximab between November 1, 2018 through April 1, 2019 at the RLRVAMC was generated. HCPs were alerted to eligible patients based on protocol requirements. The HCPs then made the final determination and entered orders accordingly.
This study was a retrospective chart review of all who patients received a rapid infusion of rituximab. Patients who were included if they were aged ≥ 18 years, received rituximab infusions in the RLRVAMC infusion clinic, had an absolute lymphocyte count ≤ 5,000/mm3 at the time of their rapid infusions, had no significant baseline cardiovascular disease or respiratory compromise, and had no prior grade 3 or 4 rituximab IRRs as defined by Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0.9 This study was a quality improvement initiative and considered exempt by the institutional review board. All data were deidentified and secured to ensure patient privacy.
The primary endpoint for this study was the incidence of grade 3 or 4 IRRs associated with the rapid infusion of rituximab. Secondary endpoints included the proportion of patients who experienced a grade 3 or 4 infusion reaction, who received proper treatment according to the institution’s hypersensitivity protocol, savings in infusion clinic chair time, and nursing satisfaction with education and implementation of the rapid infusion rituximab protocol.
The following data were collected for all included patients: demographics, lactic acid dehydrogenase level, white blood cell count, and absolute lymphocyte count prior to rituximab infusion, indication for treatment, dose of rituximab for 90-minute infusion, date of infusion, starting time, ending time, number of previous rituximab infusions within the past 3 months, symptoms of infusion reactions during rituximab infusion, and grade of any infusion reactions that occurred.
Estimated savings in infusion clinic chair time was calculated by taking the difference in time between each completed rapid infusion and the estimated amount of time it would have taken for each patient to receive a traditional infusion. The estimated amount of time for traditional infusion was determined by following the institution’s protocol for administering rituximab to patients who previously tolerated their first dose of the drug (eg, 100 mg/h starting rate and increasing by 100 mg/h every 30 minutes to a maximum infusion rate of 400 mg/h). All endpoints were analyzed using descriptive statistics.
Results
Between November 1, 2018 and April 1, 2019, 11 patients received a total of 24 rapid infusions of rituximab. The majority of patients included in the study were older males, and the most common indication for rapid infusion was follicular lymphoma (Table 1).
Primary Endpoint
All patients who received a rapid infusion of rituximab were reviewed in the analysis of the primary and secondary endpoints. Among the 24 rapid infusions of rituximab, 1 infusion was stopped due to the patient experiencing a grade 3 IRR according to criteria from CTCAE Version 5.0. The patient was found to have dysphagia at baseline and experienced severe symptoms in the days following the first infusion that put the patient at high risk for subsequent infusion related concerns. Eligibility criteria for the 90-minute protocol were updated based on these findings. No patient experienced a grade 4 or 5 IRR. The remaining 23 infusions were well tolerated by the patients with no clinically significant events.
Secondary Endpoints
The patient who experienced a grade 3 IRR to rituximab received proper treatment by infusion clinic nurses according to the RLRVAMC hypersensitivity protocol. Patients who received rapid infusions of rituximab had a mean length of infusion of 95.0 minutes. This was in contrast to the mean time of each patient’s previous nonrapid infusion of 134.3 minutes. The difference between the 2 values equated to a savings in infusion clinic chair mean time of 39.3 minutes per patient.
Nurses were asked whether they had prior experience administering rituximab via 90-minute infusion and whether they felt comfortable administering a 90-minute rituximab infusion. Before the live education session, none of the nurses surveyed had prior experience or felt comfortable administering rituximab over 90 minutes. When the nurses were surveyed poststudy, all reported that they were experienced administering rituximab and felt comfortable with the process (Table 2).
Discussion
The infusion of rituximab has been associated with significant challenges related to the time and labor required. Although a vast number of institutions across the country now infuse the medication over an abbreviated time, HCP concerns for patient safety and appropriate use of hypersensitivity protocol in a veteran population delayed implementation at RLRVAMC. The results from this quality improvement initiative highlight the positive impact of the proactive measures that were used to implement the rapid infusion protocol for rituximab on improving HCP prescribing rates, nursing satisfaction, and appropriate management of IRRs.
Rapid infusion saved on average 39.3 minutes per patient in infusion clinic chair time. Each successful rapid infusion of rituximab potentially opened additional time in clinic for ≥ 1 patients to receive an infusion therapy. The RLRVAMC usually operated at maximum capacity, so the ability to accommodate more patients helped decrease hospital admittances for time-sensitive infusions.
The initial criteria used to screen patients to determine whether a rapid infusion of rituximab would be appropriate was based on inclusion and exclusion criteria for past studies on the same subject.3-5 The incidence of hypersensitivity reactions associated with study participants who received rapid rituximab infusions also resembles past research done on the subject, which is important to note due to prior misconceptions of staff at the institution of a higher risk of reaction in this specific veteran population. One patient with RA experienced a grade 3 IRR in this study. Although this patient met the original inclusion criteria, the patient had baseline dysphagia, and following the first infusion, reported to the emergency department (ED) with symptoms of delayed anaphylaxis. In this case, the order for rapid infusion was placed in advance and the prescriber was unaware of the ED visit. Based on this event, eligibility criteria for 90-minute rituximab infusions were updated to include additional information specifying that candidates for a rapid infusion also may have no baseline airway compromise. This hypersensitivity reaction also highlighted the need for decision support technology to assist HCPs in patient selection as well as empowering nursing and pharmacy staff to identify concerns once they place orders.
Over the course of the study, investigators assisted the HCPs with preparation of orders for the rapid infusion of rituximab for antineoplastic indications. Due to feasibility issues with this practice moving forward, order sets containing rituximab were updated to include a 90-minute option. This created a more standardized process that allowed HCPs to screen potential patients on their own. The expectation is that HCPs will be more likely to order 90-minute infusions for eligible patients in the future with this efficient and safer process.
Limitations
The small sample size in this study was a limitation. Retrospective data related to the management of infusion reactions and length of infusions were collected from nursing notes. The prospective use of a standardized evaluation tool for adverse drug reactions as well as bar code medication administration technology would improve the data available for this study. Additional studies also would be useful to validate the results.
Conclusions
The proactive measures that were used to implement the rapid infusion rituximab protocol improved HCP prescribing rates, nursing satisfaction, and the management of IRRs. Potential time savings with each infusion was significant. This study confirmed appropriateness of rapid administration of rituximab in this veteran population and has increased interest in implementing other rapid infusion protocols. Protocols, education, and order sets are being developed for daratumumab and infliximab.
Rituximab is a genetically engineered chimeric immunoglobulin G1 monoclonal antibody. It functions by binding to the CD20 antigen on the surface of B-cell lymphocytes, leading to complement-dependent cytotoxicity and antibody-dependent cellular cytotoxicity.1 The US Food and Drug Administration approved this therapy to treat patients with B-cell non-Hodgkin lymphoma and chronic lymphocytic leukemia, along with other nonmalignant indications, including pemphigus vulgaris and rheumatoid arthritis (RA). Historically, a significant amount of time and labor on behalf of medical personnel has been required to administer rituximab according to the original manufacturer’s labeling due to the boxed warning associated with infusion-related reactions (IRRs).2
Originally, the elongated infusion times that were recommended for rituximab were largely due to the perceived risk of serious infusion-related adverse drug reactions. Slower infusion times should reduce the risk of a reaction and are considered to be a good option for those patients who are at a high risk of having a severe IRRs to rituximab. Examples of high-risk patients from previous studies include those with significant cardiovascular disease, a circulating lymphocyte count ≤ 5,000/µL at the start of infusion, and those who have previously had a reaction to rituximab.3-5 In appropriate patients, research has shown a decreasing incidence of all-grade IRRs for patients who are prescribed rituximab as they receive more doses of the drug.2,6 The ability to identify suitable patients for 90-minute infusions of rituximab and the prospect of better health system resource utilization has led investigators to study the effects of shortened infusion times.
The RATE trial addressed this subject with a phase 3 safety study on the effects of a 90-minute rituximab infusion for patients with previously untreated diffuse large B-cell and follicular lymphoma.3 The patients in this study received their first dose of rituximab using the traditional infusion approach. If it was well-tolerated, they received subsequent rituximab infusions using a 90-minute protocol. Only 1.1% of patients who had previously received a rituximab infusion developed a grade 3 or 4 IRR when receiving a faster infusion of the drug for the first time.3 This result led to the addition of instructions for a 90-minute infusion to the package insert.2
In contrast to the RATE trial, the RATE-RA trial evaluated the incidence of IRRs in patients who received rituximab for nonmalignant indications. This study assessed patients with RA receiving rituximab for > 120 minutes. The authors reported 0.6% of the patients in the study developed a grade 3 or 4 IRR associated with the first 120-minute infusion of the medication.5 The researchers concluded that rituximab can be administered at a faster rate during second and subsequent infusions in patients who have been shown to tolerate traditional infusions without increasing the risk or severity of IRRs.5
The US Department of Veterans Affairs (VA) Richard L. Roudebush VA Medical Center (RLRVAMC) in Indianapolis, Indiana, uses traditional directions for the infusion of rituximab due to perceived tolerability and safety concerns specifically in a veteran population—even while other VA medical centers have implemented shortened infusion protocols. This also is despite the fact that available research shows rapid infusions of the drug are well tolerated in a variety of community settings.7,8 Anticipated benefits of implementing a protocol include savings in chair time at the institution’s infusion clinic along with increased nursing and patient satisfaction. This project was conducted to prepare, implement, and assess the safety of a 90-minute rituximab protocol at the RLRVAMC.
Methods
Proactive measures were required before and during the implementation of the 90-minute protocol to ensure patient safety and staff satisfaction. Updates to the RLRVAMC policy for the management of medical emergencies within the infusion center were reviewed and approved by the acute care committee and nursing leadership. A protocol was developed to identify eligible patients, outline the hypersensitivity protocol, instruct pharmacy personnel on admixture preparation, and provide a titration schedule based on dose. Order sets also were created to assist health care providers (HCPs) with the prescribing of rituximab for nonantineoplastic indications. Educational materials were crafted to assist with order verification, product preparation, labeling, and programming of infusion pumps. Live education was provided for physicians, pharmacists, and nurses to ensure smooth implementation of the protocol and appropriate management of medical emergencies based on the updated policy.
Study Design
Nursing staff in the infusion clinic were surveyed once before a live education session and again after the conclusion of the study. The purpose of the survey was to assess the prior experience and current comfort level of the nursing staff with administering rituximab over 90 minutes. Nurses were asked the following questions: (1) Do you have prior experience administering rituximab via 90-minute infusion; and (2) do you feel comfortable administering rituximab via 90-minute infusion?
A weekly report of patients who received rituximab between November 1, 2018 through April 1, 2019 at the RLRVAMC was generated. HCPs were alerted to eligible patients based on protocol requirements. The HCPs then made the final determination and entered orders accordingly.
This study was a retrospective chart review of all who patients received a rapid infusion of rituximab. Patients who were included if they were aged ≥ 18 years, received rituximab infusions in the RLRVAMC infusion clinic, had an absolute lymphocyte count ≤ 5,000/mm3 at the time of their rapid infusions, had no significant baseline cardiovascular disease or respiratory compromise, and had no prior grade 3 or 4 rituximab IRRs as defined by Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0.9 This study was a quality improvement initiative and considered exempt by the institutional review board. All data were deidentified and secured to ensure patient privacy.
The primary endpoint for this study was the incidence of grade 3 or 4 IRRs associated with the rapid infusion of rituximab. Secondary endpoints included the proportion of patients who experienced a grade 3 or 4 infusion reaction, who received proper treatment according to the institution’s hypersensitivity protocol, savings in infusion clinic chair time, and nursing satisfaction with education and implementation of the rapid infusion rituximab protocol.
The following data were collected for all included patients: demographics, lactic acid dehydrogenase level, white blood cell count, and absolute lymphocyte count prior to rituximab infusion, indication for treatment, dose of rituximab for 90-minute infusion, date of infusion, starting time, ending time, number of previous rituximab infusions within the past 3 months, symptoms of infusion reactions during rituximab infusion, and grade of any infusion reactions that occurred.
Estimated savings in infusion clinic chair time was calculated by taking the difference in time between each completed rapid infusion and the estimated amount of time it would have taken for each patient to receive a traditional infusion. The estimated amount of time for traditional infusion was determined by following the institution’s protocol for administering rituximab to patients who previously tolerated their first dose of the drug (eg, 100 mg/h starting rate and increasing by 100 mg/h every 30 minutes to a maximum infusion rate of 400 mg/h). All endpoints were analyzed using descriptive statistics.
Results
Between November 1, 2018 and April 1, 2019, 11 patients received a total of 24 rapid infusions of rituximab. The majority of patients included in the study were older males, and the most common indication for rapid infusion was follicular lymphoma (Table 1).
Primary Endpoint
All patients who received a rapid infusion of rituximab were reviewed in the analysis of the primary and secondary endpoints. Among the 24 rapid infusions of rituximab, 1 infusion was stopped due to the patient experiencing a grade 3 IRR according to criteria from CTCAE Version 5.0. The patient was found to have dysphagia at baseline and experienced severe symptoms in the days following the first infusion that put the patient at high risk for subsequent infusion related concerns. Eligibility criteria for the 90-minute protocol were updated based on these findings. No patient experienced a grade 4 or 5 IRR. The remaining 23 infusions were well tolerated by the patients with no clinically significant events.
Secondary Endpoints
The patient who experienced a grade 3 IRR to rituximab received proper treatment by infusion clinic nurses according to the RLRVAMC hypersensitivity protocol. Patients who received rapid infusions of rituximab had a mean length of infusion of 95.0 minutes. This was in contrast to the mean time of each patient’s previous nonrapid infusion of 134.3 minutes. The difference between the 2 values equated to a savings in infusion clinic chair mean time of 39.3 minutes per patient.
Nurses were asked whether they had prior experience administering rituximab via 90-minute infusion and whether they felt comfortable administering a 90-minute rituximab infusion. Before the live education session, none of the nurses surveyed had prior experience or felt comfortable administering rituximab over 90 minutes. When the nurses were surveyed poststudy, all reported that they were experienced administering rituximab and felt comfortable with the process (Table 2).
Discussion
The infusion of rituximab has been associated with significant challenges related to the time and labor required. Although a vast number of institutions across the country now infuse the medication over an abbreviated time, HCP concerns for patient safety and appropriate use of hypersensitivity protocol in a veteran population delayed implementation at RLRVAMC. The results from this quality improvement initiative highlight the positive impact of the proactive measures that were used to implement the rapid infusion protocol for rituximab on improving HCP prescribing rates, nursing satisfaction, and appropriate management of IRRs.
Rapid infusion saved on average 39.3 minutes per patient in infusion clinic chair time. Each successful rapid infusion of rituximab potentially opened additional time in clinic for ≥ 1 patients to receive an infusion therapy. The RLRVAMC usually operated at maximum capacity, so the ability to accommodate more patients helped decrease hospital admittances for time-sensitive infusions.
The initial criteria used to screen patients to determine whether a rapid infusion of rituximab would be appropriate was based on inclusion and exclusion criteria for past studies on the same subject.3-5 The incidence of hypersensitivity reactions associated with study participants who received rapid rituximab infusions also resembles past research done on the subject, which is important to note due to prior misconceptions of staff at the institution of a higher risk of reaction in this specific veteran population. One patient with RA experienced a grade 3 IRR in this study. Although this patient met the original inclusion criteria, the patient had baseline dysphagia, and following the first infusion, reported to the emergency department (ED) with symptoms of delayed anaphylaxis. In this case, the order for rapid infusion was placed in advance and the prescriber was unaware of the ED visit. Based on this event, eligibility criteria for 90-minute rituximab infusions were updated to include additional information specifying that candidates for a rapid infusion also may have no baseline airway compromise. This hypersensitivity reaction also highlighted the need for decision support technology to assist HCPs in patient selection as well as empowering nursing and pharmacy staff to identify concerns once they place orders.
Over the course of the study, investigators assisted the HCPs with preparation of orders for the rapid infusion of rituximab for antineoplastic indications. Due to feasibility issues with this practice moving forward, order sets containing rituximab were updated to include a 90-minute option. This created a more standardized process that allowed HCPs to screen potential patients on their own. The expectation is that HCPs will be more likely to order 90-minute infusions for eligible patients in the future with this efficient and safer process.
Limitations
The small sample size in this study was a limitation. Retrospective data related to the management of infusion reactions and length of infusions were collected from nursing notes. The prospective use of a standardized evaluation tool for adverse drug reactions as well as bar code medication administration technology would improve the data available for this study. Additional studies also would be useful to validate the results.
Conclusions
The proactive measures that were used to implement the rapid infusion rituximab protocol improved HCP prescribing rates, nursing satisfaction, and the management of IRRs. Potential time savings with each infusion was significant. This study confirmed appropriateness of rapid administration of rituximab in this veteran population and has increased interest in implementing other rapid infusion protocols. Protocols, education, and order sets are being developed for daratumumab and infliximab.
1. Feugier P. A review of rituximab, the first anti-CD20 monoclonal antibody used in the treatment of B non-Hodgkin’s lymphomas. Future Oncol. 2015;11(9):1327-1342. doi:10.2217/fon.15.57
2. Rituxan [package insert]. South San Francisco, CA: Genentech; 2016.
3. Dakhil S, Hermann R, Schreeder MT, et al. Phase III safety study of rituximab administered as a 90-minute infusion in patients with previously untreated diffuse large B-cell and follicular lymphoma. Leuk Lymphoma. 2014;55(10):2335-2340. doi:10.3109/10428194.2013.877135
4. Dotson E, Crawford B, Phillips G, Jones J. Sixty-minute infusion rituximab protocol allows for safe and efficient workflow. Support Care Cancer. 2016;24(3):1125-1129. doi:10.1007/s00520-015-2869-4
5. Pritchard CH, Greenwald MW, Kremer JM, et al. Safety of infusing rituximab at a more rapid rate in patients with rheumatoid arthritis: results from the RATE-RA study. BMC Musculoskelet Disord. 2014;15:177. doi:10.1186/1471-2474-15-177
6. Hainsworth JD, Litchy S, Barton JH, et al. Single-agent rituximab as first-line and maintenance treatment for patients with chronic lymphocytic leukemia or small lymphocytic lymphoma: a phase II trial of the Minnie Pearl Cancer Research Network. J Clin Oncol. 2003;21(9):1746-1751. doi:10.1200/JCO.2003.09.027
7. Can M, Alibaz-Öner F, Yılmaz-Öner S, Atagündüz P, Înanç N, Direskeneli H. Accelerated infusion rates of rituximab are well tolerated and safe in rheumatology practice: a single-centre experience. Clin Rheumatol. 2013;32(1):87-90. doi:10.1007/s10067-012-2094-1
8. Sehn LH, Donaldson J, Filewich A, et al. Rapid infusion rituximab in combination with corticosteroid-containing chemotherapy or as maintenance therapy is well tolerated and can safely be delivered in the community setting. Blood. 2007;109(10):4171-4173. doi:10.1182/blood-2006-11-059469
9. National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE). https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm. Updated March 27 2020. Accessed June 15, 2020.
1. Feugier P. A review of rituximab, the first anti-CD20 monoclonal antibody used in the treatment of B non-Hodgkin’s lymphomas. Future Oncol. 2015;11(9):1327-1342. doi:10.2217/fon.15.57
2. Rituxan [package insert]. South San Francisco, CA: Genentech; 2016.
3. Dakhil S, Hermann R, Schreeder MT, et al. Phase III safety study of rituximab administered as a 90-minute infusion in patients with previously untreated diffuse large B-cell and follicular lymphoma. Leuk Lymphoma. 2014;55(10):2335-2340. doi:10.3109/10428194.2013.877135
4. Dotson E, Crawford B, Phillips G, Jones J. Sixty-minute infusion rituximab protocol allows for safe and efficient workflow. Support Care Cancer. 2016;24(3):1125-1129. doi:10.1007/s00520-015-2869-4
5. Pritchard CH, Greenwald MW, Kremer JM, et al. Safety of infusing rituximab at a more rapid rate in patients with rheumatoid arthritis: results from the RATE-RA study. BMC Musculoskelet Disord. 2014;15:177. doi:10.1186/1471-2474-15-177
6. Hainsworth JD, Litchy S, Barton JH, et al. Single-agent rituximab as first-line and maintenance treatment for patients with chronic lymphocytic leukemia or small lymphocytic lymphoma: a phase II trial of the Minnie Pearl Cancer Research Network. J Clin Oncol. 2003;21(9):1746-1751. doi:10.1200/JCO.2003.09.027
7. Can M, Alibaz-Öner F, Yılmaz-Öner S, Atagündüz P, Înanç N, Direskeneli H. Accelerated infusion rates of rituximab are well tolerated and safe in rheumatology practice: a single-centre experience. Clin Rheumatol. 2013;32(1):87-90. doi:10.1007/s10067-012-2094-1
8. Sehn LH, Donaldson J, Filewich A, et al. Rapid infusion rituximab in combination with corticosteroid-containing chemotherapy or as maintenance therapy is well tolerated and can safely be delivered in the community setting. Blood. 2007;109(10):4171-4173. doi:10.1182/blood-2006-11-059469
9. National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE). https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm. Updated March 27 2020. Accessed June 15, 2020.
Gardasil-9 approved for prevention of head and neck cancers
The US Food and Drug Administration (FDA) has expanded the indication for the Gardasil-9 (Merck) vaccine to include prevention of oropharyngeal and other head and neck cancers caused by HPV types 16, 18, 31, 33, 45, 52, and 58.
This new indication is approved under the FDA’s accelerated approval program and is based on the vaccine’s effectiveness in preventing HPV-related anogenital disease. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory clinical trial, which is currently underway.
“At Merck, working to help prevent certain HPV-related cancers has been a priority for more than two decades,” Alain Luxembourg, MD, director, clinical research, Merck Research Laboratories, said in a statement. “Today’s approval for the prevention of HPV-related oropharyngeal and other head and neck cancers represents an important step in Merck’s mission to help reduce the number of men and women affected by certain HPV-related cancers.”
This new indication doesn’t affect the current recommendations that are already in place. In 2018, a supplemental application for Gardasil 9 was approved to include women and men aged 27 through 45 years for preventing a variety of cancers including cervical, vulvar, vaginal, and anal cancer as well as genital warts. But cancers of the head and neck were not included.
The original Gardasil vaccine came on the market in 2006, with an indication to prevent certain cancers and diseases caused by HPV types 6, 11, 16, and 18. It is no longer distributed in the United States.
In 2014, the FDA approved Gardasil 9, which extends the vaccine coverage for the initial four HPV types as five additional types (31, 33, 45, 52, and 58), and its initial indication was for use in both men and women between the ages of 9 through 26 years.
Head and neck cancers surpass cervical cancer
More than 2 decades ago, researchers first found a connection between HPV and a subset of head and neck cancers (Curr Opin Oncol. 1999;11(3):191-199). The cancers associated with HPV also appeared to have a different biology and disease pattern, as well as a better prognosis, compared with those that were unrelated. HPV is now responsible for the majority of oropharyngeal squamous cell cancers diagnosed in the United States.
A study published last year found that oral HPV infections were occurring with significantly less frequency among sexually active female adolescents who had received the quadrivalent vaccine, as compared with those who were unvaccinated.
These findings provided evidence that HPV vaccination was associated with a reduced frequency of HPV infection in the oral cavity, suggesting that vaccination could decrease the future risk of HPV-associated head and neck cancers.
The omission of head and neck cancers from the initial list of indications for the vaccine is notable because, according to data from the Centers for Disease Control and Prevention (CDC), oropharyngeal cancers are now the most common malignancy caused by HPV, surpassing cervical cancer.
Who will benefit?
An estimated 14 million new HPV infections occur every year in the United States, according to the CDC, and about 80% of individuals who are sexually active have been exposed at some point during their lifetime. In most people, however, the virus will clear on its own without causing any illness or symptoms.
In a Medscape videoblog, Sandra Adamson Fryhofer, MD, MACP, FRCP, helped clarify the adult population most likely to benefit from the vaccine. She pointed out that the HPV vaccine doesn’t treat HPV-related disease or help clear infections, and there are currently no clinical antibody tests or titers that can predict immunity.
“Many adults aged 27-45 have already been exposed to HPV early in life,” she said. Those in a long-term mutually monogamous relationship are not likely to get a new HPV infection. Those with multiple prior sex partners are more likely to have already been exposed to vaccine serotypes. For them, the vaccine will be less effective.”
Fryhofer added that individuals who are now at risk for exposure to a new HPV infection from a new sex partner are the ones most likely to benefit from HPV vaccination.
Confirmation needed
The FDA’s accelerated approval is contingent on confirmatory data, and Merck opened a clinical trial this past February to evaluate the efficacy, immunogenicity, and safety of the 9-valent HPV vaccine in men 20 to 45 years of age. The phase 3 multicenter randomized trial will have an estimated enrollment of 6000 men.
This article first appeared on Medscape.com.
The US Food and Drug Administration (FDA) has expanded the indication for the Gardasil-9 (Merck) vaccine to include prevention of oropharyngeal and other head and neck cancers caused by HPV types 16, 18, 31, 33, 45, 52, and 58.
This new indication is approved under the FDA’s accelerated approval program and is based on the vaccine’s effectiveness in preventing HPV-related anogenital disease. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory clinical trial, which is currently underway.
“At Merck, working to help prevent certain HPV-related cancers has been a priority for more than two decades,” Alain Luxembourg, MD, director, clinical research, Merck Research Laboratories, said in a statement. “Today’s approval for the prevention of HPV-related oropharyngeal and other head and neck cancers represents an important step in Merck’s mission to help reduce the number of men and women affected by certain HPV-related cancers.”
This new indication doesn’t affect the current recommendations that are already in place. In 2018, a supplemental application for Gardasil 9 was approved to include women and men aged 27 through 45 years for preventing a variety of cancers including cervical, vulvar, vaginal, and anal cancer as well as genital warts. But cancers of the head and neck were not included.
The original Gardasil vaccine came on the market in 2006, with an indication to prevent certain cancers and diseases caused by HPV types 6, 11, 16, and 18. It is no longer distributed in the United States.
In 2014, the FDA approved Gardasil 9, which extends the vaccine coverage for the initial four HPV types as five additional types (31, 33, 45, 52, and 58), and its initial indication was for use in both men and women between the ages of 9 through 26 years.
Head and neck cancers surpass cervical cancer
More than 2 decades ago, researchers first found a connection between HPV and a subset of head and neck cancers (Curr Opin Oncol. 1999;11(3):191-199). The cancers associated with HPV also appeared to have a different biology and disease pattern, as well as a better prognosis, compared with those that were unrelated. HPV is now responsible for the majority of oropharyngeal squamous cell cancers diagnosed in the United States.
A study published last year found that oral HPV infections were occurring with significantly less frequency among sexually active female adolescents who had received the quadrivalent vaccine, as compared with those who were unvaccinated.
These findings provided evidence that HPV vaccination was associated with a reduced frequency of HPV infection in the oral cavity, suggesting that vaccination could decrease the future risk of HPV-associated head and neck cancers.
The omission of head and neck cancers from the initial list of indications for the vaccine is notable because, according to data from the Centers for Disease Control and Prevention (CDC), oropharyngeal cancers are now the most common malignancy caused by HPV, surpassing cervical cancer.
Who will benefit?
An estimated 14 million new HPV infections occur every year in the United States, according to the CDC, and about 80% of individuals who are sexually active have been exposed at some point during their lifetime. In most people, however, the virus will clear on its own without causing any illness or symptoms.
In a Medscape videoblog, Sandra Adamson Fryhofer, MD, MACP, FRCP, helped clarify the adult population most likely to benefit from the vaccine. She pointed out that the HPV vaccine doesn’t treat HPV-related disease or help clear infections, and there are currently no clinical antibody tests or titers that can predict immunity.
“Many adults aged 27-45 have already been exposed to HPV early in life,” she said. Those in a long-term mutually monogamous relationship are not likely to get a new HPV infection. Those with multiple prior sex partners are more likely to have already been exposed to vaccine serotypes. For them, the vaccine will be less effective.”
Fryhofer added that individuals who are now at risk for exposure to a new HPV infection from a new sex partner are the ones most likely to benefit from HPV vaccination.
Confirmation needed
The FDA’s accelerated approval is contingent on confirmatory data, and Merck opened a clinical trial this past February to evaluate the efficacy, immunogenicity, and safety of the 9-valent HPV vaccine in men 20 to 45 years of age. The phase 3 multicenter randomized trial will have an estimated enrollment of 6000 men.
This article first appeared on Medscape.com.
The US Food and Drug Administration (FDA) has expanded the indication for the Gardasil-9 (Merck) vaccine to include prevention of oropharyngeal and other head and neck cancers caused by HPV types 16, 18, 31, 33, 45, 52, and 58.
This new indication is approved under the FDA’s accelerated approval program and is based on the vaccine’s effectiveness in preventing HPV-related anogenital disease. Continued approval for this indication may be contingent upon verification and description of clinical benefit in a confirmatory clinical trial, which is currently underway.
“At Merck, working to help prevent certain HPV-related cancers has been a priority for more than two decades,” Alain Luxembourg, MD, director, clinical research, Merck Research Laboratories, said in a statement. “Today’s approval for the prevention of HPV-related oropharyngeal and other head and neck cancers represents an important step in Merck’s mission to help reduce the number of men and women affected by certain HPV-related cancers.”
This new indication doesn’t affect the current recommendations that are already in place. In 2018, a supplemental application for Gardasil 9 was approved to include women and men aged 27 through 45 years for preventing a variety of cancers including cervical, vulvar, vaginal, and anal cancer as well as genital warts. But cancers of the head and neck were not included.
The original Gardasil vaccine came on the market in 2006, with an indication to prevent certain cancers and diseases caused by HPV types 6, 11, 16, and 18. It is no longer distributed in the United States.
In 2014, the FDA approved Gardasil 9, which extends the vaccine coverage for the initial four HPV types as five additional types (31, 33, 45, 52, and 58), and its initial indication was for use in both men and women between the ages of 9 through 26 years.
Head and neck cancers surpass cervical cancer
More than 2 decades ago, researchers first found a connection between HPV and a subset of head and neck cancers (Curr Opin Oncol. 1999;11(3):191-199). The cancers associated with HPV also appeared to have a different biology and disease pattern, as well as a better prognosis, compared with those that were unrelated. HPV is now responsible for the majority of oropharyngeal squamous cell cancers diagnosed in the United States.
A study published last year found that oral HPV infections were occurring with significantly less frequency among sexually active female adolescents who had received the quadrivalent vaccine, as compared with those who were unvaccinated.
These findings provided evidence that HPV vaccination was associated with a reduced frequency of HPV infection in the oral cavity, suggesting that vaccination could decrease the future risk of HPV-associated head and neck cancers.
The omission of head and neck cancers from the initial list of indications for the vaccine is notable because, according to data from the Centers for Disease Control and Prevention (CDC), oropharyngeal cancers are now the most common malignancy caused by HPV, surpassing cervical cancer.
Who will benefit?
An estimated 14 million new HPV infections occur every year in the United States, according to the CDC, and about 80% of individuals who are sexually active have been exposed at some point during their lifetime. In most people, however, the virus will clear on its own without causing any illness or symptoms.
In a Medscape videoblog, Sandra Adamson Fryhofer, MD, MACP, FRCP, helped clarify the adult population most likely to benefit from the vaccine. She pointed out that the HPV vaccine doesn’t treat HPV-related disease or help clear infections, and there are currently no clinical antibody tests or titers that can predict immunity.
“Many adults aged 27-45 have already been exposed to HPV early in life,” she said. Those in a long-term mutually monogamous relationship are not likely to get a new HPV infection. Those with multiple prior sex partners are more likely to have already been exposed to vaccine serotypes. For them, the vaccine will be less effective.”
Fryhofer added that individuals who are now at risk for exposure to a new HPV infection from a new sex partner are the ones most likely to benefit from HPV vaccination.
Confirmation needed
The FDA’s accelerated approval is contingent on confirmatory data, and Merck opened a clinical trial this past February to evaluate the efficacy, immunogenicity, and safety of the 9-valent HPV vaccine in men 20 to 45 years of age. The phase 3 multicenter randomized trial will have an estimated enrollment of 6000 men.
This article first appeared on Medscape.com.
Surgeon General scolds docs for failing to help patients quit smoking
The U.S. Surgeon General is calling on all physicians to help patients stop smoking, noting that two-thirds of adult smokers say they want to quit, but only 40% report that their doctor has advised them to stop.
“I’ve got to own this as the nation’s doctor, and our health providers in this room and in this country need to own this stat,” said Surgeon General Jerome Adams, MD, at a press briefing releasing a new report on smoking cessation.
“Smoking is the No. 1 preventable cause of death, disease, and disability in the United States,” he said. “So why are 40% of our health providers out there not advising smokers to quit when they come in?”
In the first U.S. Surgeon General report on smoking cessation in 30 years, the 700-page report suggests smoking cessation-related quality measures that include physician reimbursement would increase treatment.
The evidence also suggests that using electronic health records to prompt clinicians to inquire about smoking would increase cessation treatment.
EHRs could be used to “empower and enable” physicians to advise people to quit, said Dr. Adams. Physicians also need “the education and the confidence to be able to have that conversation, because too many of them look at someone and say: ‘Nope, too hard, too much effort, no, that’s not what they’re here for today,’ ” he said.
However, “simply asking, advising, and referring can be enough to get someone on the pathway to quitting,” Dr. Adams said.
34 million still smoke
The new report is the first on the topic released since 1990, and the 34th on tobacco control since the first one was issued in 1964, said Dr. Adams. Since that first report, adult smoking has declined 70%, but some 34 million Americans (14%) still smoke, he said.
In addition, Dr. Adams said that many subpopulations have been left behind, noting: “Cigarette smoking remains highest among LGBTQ adults, people with disabilities or limitations, American Indians and Alaska Natives, and people with mental health conditions or substance use disorders.”
He also noted that 40% of cigarettes are consumed by those with a mental illness or a substance use disorder.
Quitting is beneficial at any age and can add as much as a decade to life expectancy, the report notes. Quitting also reduces the risk of 12 cancers, cuts the risk of chronic obstructive pulmonary disease, and reduces cardiovascular and stroke morbidity and mortality.
Pregnant women who quit also reduce their own morbidity and mortality risk and that of unborn children and infants, the report says.
“We know more about the science of quitting than ever before. We can, and must, do more to ensure that evidence-based cessation treatments are reaching the people that need them,” said Dr. Adams.
Less than one-third of those who have quit have used Food and Drug Administration–approved cessation medications or behavioral counseling, Dr. Adams said.
Barriers to care
Despite the existence of five nicotine replacement therapies and two nonnicotine oral medications, and more widespread availability of proven counseling methods – including web- or text-based programs – barriers to access remain.
These include a lack of insurance coverage for comprehensive, evidence-based smoking cessation treatment, which, when offered, increases availability and use.
“These are cost-effective interventions,” said Dr. Adams. “It’s penny wise and pound foolish to not give someone access to what we know works,” he said.
Because of the diversity of e-cigarette products and the variety of ways they are used, coupled with little research, it’s not currently possible to determine whether they are, or are not, useful smoking cessation tools, the report notes.
However, experts who compiled the report found some evidence to suggest that e-cigarettes containing nicotine may be “associated with increased smoking cessation compared with the use of e-cigarettes not containing nicotine.”
Asked whether the report’s conclusions might be interpreted as supportive of e-cigarettes, Dr. Adams said the report focused on smoking cessation, not initiation.
“I’m terribly concerned about the clear data that shows youth are initiating tobacco product use with e-cigarettes,” he said.
The Trump administration’s current proposal to partially restrict sales of some flavored e-cigarettes “reflects the science,” and “a balance between a desire to really make sure that people aren’t initiating with these products, but also a desire to again try to maintain a pathway for adults who want to use these products to quit to use them,” Dr. Adams said.
The focus, said Dr. Adams, should not be on e-cigarettes and whether they do, or do not, work.
“People want to quit,” he said. “We know what works. Not enough of them are getting it, and there are terrible disparities in who is and who is not getting access to effective and evidence-based treatment – that’s the story here.”
This article first appeared on Medscape.com.
The U.S. Surgeon General is calling on all physicians to help patients stop smoking, noting that two-thirds of adult smokers say they want to quit, but only 40% report that their doctor has advised them to stop.
“I’ve got to own this as the nation’s doctor, and our health providers in this room and in this country need to own this stat,” said Surgeon General Jerome Adams, MD, at a press briefing releasing a new report on smoking cessation.
“Smoking is the No. 1 preventable cause of death, disease, and disability in the United States,” he said. “So why are 40% of our health providers out there not advising smokers to quit when they come in?”
In the first U.S. Surgeon General report on smoking cessation in 30 years, the 700-page report suggests smoking cessation-related quality measures that include physician reimbursement would increase treatment.
The evidence also suggests that using electronic health records to prompt clinicians to inquire about smoking would increase cessation treatment.
EHRs could be used to “empower and enable” physicians to advise people to quit, said Dr. Adams. Physicians also need “the education and the confidence to be able to have that conversation, because too many of them look at someone and say: ‘Nope, too hard, too much effort, no, that’s not what they’re here for today,’ ” he said.
However, “simply asking, advising, and referring can be enough to get someone on the pathway to quitting,” Dr. Adams said.
34 million still smoke
The new report is the first on the topic released since 1990, and the 34th on tobacco control since the first one was issued in 1964, said Dr. Adams. Since that first report, adult smoking has declined 70%, but some 34 million Americans (14%) still smoke, he said.
In addition, Dr. Adams said that many subpopulations have been left behind, noting: “Cigarette smoking remains highest among LGBTQ adults, people with disabilities or limitations, American Indians and Alaska Natives, and people with mental health conditions or substance use disorders.”
He also noted that 40% of cigarettes are consumed by those with a mental illness or a substance use disorder.
Quitting is beneficial at any age and can add as much as a decade to life expectancy, the report notes. Quitting also reduces the risk of 12 cancers, cuts the risk of chronic obstructive pulmonary disease, and reduces cardiovascular and stroke morbidity and mortality.
Pregnant women who quit also reduce their own morbidity and mortality risk and that of unborn children and infants, the report says.
“We know more about the science of quitting than ever before. We can, and must, do more to ensure that evidence-based cessation treatments are reaching the people that need them,” said Dr. Adams.
Less than one-third of those who have quit have used Food and Drug Administration–approved cessation medications or behavioral counseling, Dr. Adams said.
Barriers to care
Despite the existence of five nicotine replacement therapies and two nonnicotine oral medications, and more widespread availability of proven counseling methods – including web- or text-based programs – barriers to access remain.
These include a lack of insurance coverage for comprehensive, evidence-based smoking cessation treatment, which, when offered, increases availability and use.
“These are cost-effective interventions,” said Dr. Adams. “It’s penny wise and pound foolish to not give someone access to what we know works,” he said.
Because of the diversity of e-cigarette products and the variety of ways they are used, coupled with little research, it’s not currently possible to determine whether they are, or are not, useful smoking cessation tools, the report notes.
However, experts who compiled the report found some evidence to suggest that e-cigarettes containing nicotine may be “associated with increased smoking cessation compared with the use of e-cigarettes not containing nicotine.”
Asked whether the report’s conclusions might be interpreted as supportive of e-cigarettes, Dr. Adams said the report focused on smoking cessation, not initiation.
“I’m terribly concerned about the clear data that shows youth are initiating tobacco product use with e-cigarettes,” he said.
The Trump administration’s current proposal to partially restrict sales of some flavored e-cigarettes “reflects the science,” and “a balance between a desire to really make sure that people aren’t initiating with these products, but also a desire to again try to maintain a pathway for adults who want to use these products to quit to use them,” Dr. Adams said.
The focus, said Dr. Adams, should not be on e-cigarettes and whether they do, or do not, work.
“People want to quit,” he said. “We know what works. Not enough of them are getting it, and there are terrible disparities in who is and who is not getting access to effective and evidence-based treatment – that’s the story here.”
This article first appeared on Medscape.com.
The U.S. Surgeon General is calling on all physicians to help patients stop smoking, noting that two-thirds of adult smokers say they want to quit, but only 40% report that their doctor has advised them to stop.
“I’ve got to own this as the nation’s doctor, and our health providers in this room and in this country need to own this stat,” said Surgeon General Jerome Adams, MD, at a press briefing releasing a new report on smoking cessation.
“Smoking is the No. 1 preventable cause of death, disease, and disability in the United States,” he said. “So why are 40% of our health providers out there not advising smokers to quit when they come in?”
In the first U.S. Surgeon General report on smoking cessation in 30 years, the 700-page report suggests smoking cessation-related quality measures that include physician reimbursement would increase treatment.
The evidence also suggests that using electronic health records to prompt clinicians to inquire about smoking would increase cessation treatment.
EHRs could be used to “empower and enable” physicians to advise people to quit, said Dr. Adams. Physicians also need “the education and the confidence to be able to have that conversation, because too many of them look at someone and say: ‘Nope, too hard, too much effort, no, that’s not what they’re here for today,’ ” he said.
However, “simply asking, advising, and referring can be enough to get someone on the pathway to quitting,” Dr. Adams said.
34 million still smoke
The new report is the first on the topic released since 1990, and the 34th on tobacco control since the first one was issued in 1964, said Dr. Adams. Since that first report, adult smoking has declined 70%, but some 34 million Americans (14%) still smoke, he said.
In addition, Dr. Adams said that many subpopulations have been left behind, noting: “Cigarette smoking remains highest among LGBTQ adults, people with disabilities or limitations, American Indians and Alaska Natives, and people with mental health conditions or substance use disorders.”
He also noted that 40% of cigarettes are consumed by those with a mental illness or a substance use disorder.
Quitting is beneficial at any age and can add as much as a decade to life expectancy, the report notes. Quitting also reduces the risk of 12 cancers, cuts the risk of chronic obstructive pulmonary disease, and reduces cardiovascular and stroke morbidity and mortality.
Pregnant women who quit also reduce their own morbidity and mortality risk and that of unborn children and infants, the report says.
“We know more about the science of quitting than ever before. We can, and must, do more to ensure that evidence-based cessation treatments are reaching the people that need them,” said Dr. Adams.
Less than one-third of those who have quit have used Food and Drug Administration–approved cessation medications or behavioral counseling, Dr. Adams said.
Barriers to care
Despite the existence of five nicotine replacement therapies and two nonnicotine oral medications, and more widespread availability of proven counseling methods – including web- or text-based programs – barriers to access remain.
These include a lack of insurance coverage for comprehensive, evidence-based smoking cessation treatment, which, when offered, increases availability and use.
“These are cost-effective interventions,” said Dr. Adams. “It’s penny wise and pound foolish to not give someone access to what we know works,” he said.
Because of the diversity of e-cigarette products and the variety of ways they are used, coupled with little research, it’s not currently possible to determine whether they are, or are not, useful smoking cessation tools, the report notes.
However, experts who compiled the report found some evidence to suggest that e-cigarettes containing nicotine may be “associated with increased smoking cessation compared with the use of e-cigarettes not containing nicotine.”
Asked whether the report’s conclusions might be interpreted as supportive of e-cigarettes, Dr. Adams said the report focused on smoking cessation, not initiation.
“I’m terribly concerned about the clear data that shows youth are initiating tobacco product use with e-cigarettes,” he said.
The Trump administration’s current proposal to partially restrict sales of some flavored e-cigarettes “reflects the science,” and “a balance between a desire to really make sure that people aren’t initiating with these products, but also a desire to again try to maintain a pathway for adults who want to use these products to quit to use them,” Dr. Adams said.
The focus, said Dr. Adams, should not be on e-cigarettes and whether they do, or do not, work.
“People want to quit,” he said. “We know what works. Not enough of them are getting it, and there are terrible disparities in who is and who is not getting access to effective and evidence-based treatment – that’s the story here.”
This article first appeared on Medscape.com.