User login
How to manage hyperthyroid disease in pregnancy
The authors report no financial relationships relevant to this article.
CASE Life on the line
A 32-year-old woman in the 24th week of her fourth pregnancy arrives at the emergency department complaining of cough and congestion, shortness of breath, and swelling in her face, hands, and feet. The swelling has become worse over the past 2 weeks, and she had several episodes of bloody vomiting the day before her visit. The patient says she has not experienced any leakage of fluid, vaginal bleeding, or contractions. She reports good fetal movement.
The patient’s medical history is unremarkable, but a review of systems reveals a 15-lb weight loss over the past 2 weeks, racing heart, worsening edema and shortness of breath, and diarrhea.
Physical findings include exophthalmia and an enlarged thyroid with a nodule on the right side, as well as bilateral rales, tachycardia, tremor, and increased deep tendon reflexes. There is no evidence of fetal cardiac failure or goiter.
A computed tomography (CT) scan of the mother shows bilateral pleural effusions indicative of high-output cardiac failure. Thyroid ultrasonography (US) reveals a diffusely enlarged thyroid gland with a right-sided mass.
The thyroid-stimulating hormone (TSH) level is undetectable. Fetal heart rate is in the 160s, with normal variability and occasional variable deceleration. Fetal US is consistent with the estimated gestational age and shows adequate amniotic fluid and no gross fetal anomalies.
What is the likely diagnosis?
This is a classic example of undiagnosed hyperthyroidism in pregnancy manifesting as thyroid storm.
As the case illustrates, uncontrolled hyperthyroidism in pregnancy poses a significant challenge for the obstetrician. The condition can cause miscarriage, preterm delivery, intrauterine growth restriction, preeclampsia, and—at its most dangerous—thyroid storm.1 Thyroid storm is a life-threatening emergency, and treatment must be initiated even before hyperthyroidism is confirmed by thyroid function testing.2 The good news is that these complications can be successfully avoided with adequate control of thyroid function.
Overt hyperthyroidism, seen in 0.2% of pregnancies, requires active intervention to avert adverse pregnancy outcome and neurologic damage to the fetus. Subclinical disease, seen in 1.7% of pregnancies, can also create serious obstetrical problems.1
The effects of hyperthyroidism in pregnancy vary in severity, ranging from the fairly innocuous, transient, and self-limited state called gestational transient thyrotoxicosis to the life-threatening emergency of thyroid storm. This review will update you on how to manage this disorder for optimal pregnancy outcome.
To screen or not to screen
Routine screening for thyroid dysfunction has been recommended for women who have infertility, menstrual disorders, or type 1 diabetes mellitus, and for pregnant women who have signs and symptoms of the disorder. Some authors recommend screening all pregnant women, but routine screening is not endorsed by the American College of Obstetricians and Gynecologists.2,3
Thyroid testing in pregnancy is recommended in women who:
- have a family history of autoimmune thyroid disease
- are on thyroid therapy
- have a goiter or
- have insulin-dependent diabetes mellitus.
Pregnant women who have a history of high-dose neck radiation, thyroid therapy, postpartum thyroiditis, or an infant born with thyroid disease should also be tested at the first prenatal visit.4
Telltale signs and laboratory tests
The signs and symptoms of hyperthyroidism can include nervousness, heat intolerance, tachycardia, palpitations, goiter, weight loss, thyromegaly, exophthalmia, increased appetite, nausea and vomiting, sweating, and tremor.1 The difficulty here? Many of these symptoms are also seen in pregnant women who have normal thyroid function, so that symptoms alone are not a reliable guide.
Instead, the diagnosis of overt hyperthyroidism is made on the basis of laboratory tests indicating suppressed TSH and elevated levels of free thyroxine (FT4) and free triiodothyronine (FT3). Subclinical hyperthyroidism is defined as a suppressed TSH level with normal FT4 and FT3 levels.2
The effects of hyperthyroidism on laboratory values are shown in TABLE 1. A form of hyperthyroidism called the T3– toxicosis syndrome is diagnosed by suppressed TSH, normal FT4, and elevated FT3 levels.4
TABLE 1
Is your pregnant patient hyperthyroid? Five-test lab panel offers a guide
| TEST AND RESULT | |||||
|---|---|---|---|---|---|
| THYROID-STIMULATING HORMONE | FREE TRI-IODOTHYRONINE | FREE THYROXINE | TOTAL TRI-IODOTHYRONINE | TOTAL THYROXINE | THEN THE MOTHER’S CONDITION IS … |
| No change | No change | ↑ | ↑ | ↑ | Pregnancy |
| ↓ | ↑ | ↑ | ↑ | ↑ | Hyperthyroidism |
| ↓ | No change | No change | No change | No change | Subclinical hyperthyroidism |
What are the causes?
The most common cause of hyperthyroidism in pregnancy—accounting for some 95% of cases—is Graves’ disease.2 This autoimmune disorder is characterized by autoantibodies that activate the TSH receptor. These autoantibodies cross the placenta and can cause fetal and neonatal thyroid dysfunction even when the mother herself is in a euthyroid condition.4
Far less often, hyperthyroidism in pregnancy has a cause other than Graves’ disease; TABLE 2 summarizes the possibilities.1 Other causes of hyperthyroidism in early pregnancy include choriocarcinoma and gestational trophoblastic disease (partial and complete moles) (TABLE 3).
TABLE 2
Causes of hyperthyroidism in pregnancy
| Graves’ disease |
| Adenoma |
| Toxic nodular goiter |
| Thyroiditis |
| Excessive thyroid hormone intake |
| Choriocarcinoma |
| Molar pregnancy |
TABLE 3
What causes severe hyperthyroidism before 20 weeks’ gestation?
| Gestational transient thyrotoxicosis |
| Choriocarcinoma |
Gestational trophoblastic disease
|
Signs and symptoms of Graves’ disease
Women who have Graves’ disease usually have thyroid nodules and may have exophthalmia, pretibial myxedema, and tachycardia. They also display other classic signs and symptoms of hyperthyroidism, such as muscle weakness, tremor, and warm and moist skin.
During pregnancy, Graves’ disease usually becomes worse during the first trimester and postpartum period; symptoms resolve during the second and third trimesters.1
Thyrotoxin receptor and antithyroid antibodies
Antithyroid antibodies are common in patients with autoimmune thyroid disease, as a response to thyroid antigens. The two most common antithyroid antibodies are thyroglobulin and thyroid peroxidase (anti-TPO). Anti-TPO antibodies are associated with postpartum thyroiditis and fetal and neonatal hyperthyroidism. TSH-receptor antibodies include thyroid-stimulating immunoglobulin (TSI) and TSH-receptor antibody. TSI is associated with Graves’ disease. TSH-receptor antibody is associated with fetal goiter, congenital hypothyroidism, and chronic thyroiditis without goiter.4
Who do you test for antibodies? Test for maternal thyroid antibodies in patients who:
- had Graves’ disease with fetal or neonatal hyperthyroidism in a previous pregnancy
- have active Graves’ disease being treated with antithyroid drugs
- are euthyroid or have undergone ablative therapy and have fetal tachycardia or intrauterine growth restriction
- have chronic thyroiditis without goiter
- have fetal goiter on ultrasound.
Newborns who have congenital hypothyroidism should also be screened for thyroid antibodies.4
What are the consequences?
Hyperthyroidism can have multiple effects on the pregnant patient and her fetus, ranging in severity from the minimal to the catastrophic.
Gestational transient thyrotoxicosis
This condition is presumably related to high levels of human chorionic gonadotropin, a substance known to stimulate TSH receptors. Unhappily for your patient, the condition is usually heralded by severe bouts of nausea and vomiting starting at 4 to 8 weeks’ gestation. Laboratory tests show significantly elevated levels of FT4 and FT3 and suppressed TSH. Despite this significant derangement, patients generally have no evidence of a hypermetabolic state.
This condition resolves by 14 to 20 weeks of gestation, is not associated with poor pregnancy outcomes, and does not require treatment with antithyroid medication.1
Adverse pregnancy outcomes
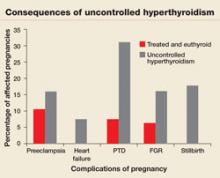
Pregnant women who have uncontrolled hyperthyroidism are at increased risk of spontaneous miscarriage, congestive heart failure, preterm delivery, intrauterine growth restriction, and preeclampsia.1 Studies that evaluated pregnancy outcomes in 239 women with overt hyperthyroidism showed increased risk of adverse pregnancy outcomes, compared with treated, euthyroid women (FIGURE 1).5-7
FIGURE 1 Consequences of uncontrolled hyperthyroidism
Several studies have found a much higher risk of pregnancy complications in women who have uncontrolled hyperthyroidism, compared with their treated and euthyroid peers.5-7
PTD=preterm delivery; FGR=fetal growth restrictions.
Fetal and neonatal hyperthyroidism
Hyperthyroidism in the fetus or newborn is caused by placental transfer of maternal immunoglobulin antibodies (TSI) to the fetus and is associated with maternal Graves’ disease. The incidence of neonatal hyperthyroidism is less than 1%. It can be predicted by rising levels of maternal TSI antibodies, to the point where levels in the third trimester are three to five times higher than they were at the beginning of pregnancy.4
Fetal hyperthyroidism develops at about 22 to 24 weeks’ gestation in mothers with a history of Graves’ disease who have been treated surgically or with ablative therapy prior to pregnancy. Even when these therapies achieve a euthyroid state in the mother, TSI levels may remain elevated and lead to fetal hyperthyroidism.
Characteristics of hyperthyroidism in the fetus include tachycardia, intrauterine growth restriction, congestive heart failure, oligohydramnios, and goiter. Treating the mother with antithyroid medications will ameliorate symptoms in the fetus.4
Thyroid storm
This is the worst-case scenario—a rare but potentially lethal complication of uncontrolled hyperthyroidism. Thyroid storm is a hypermetabolic state characterized by fever, nausea, vomiting, diarrhea, tachycardia, altered mental status, restlessness, nervousness, seizures, coma, and cardiac arrhythmias. It occurs in 1% to 2% of patients receiving thioamide therapy.8
In most instances, thyroid storm is a complication of uncontrolled hyperthyroidism, but it can also be precipitated by infection, surgery, thromboembolism, preeclampsia, labor, and delivery.
Thyroid storm is a medical emergency
This manifestation of uncontrolled hyperthyroidism is so urgent that treatment should be initiated before the results of TSH, FT4, and FT3 tests are available.2,8 Delivery should be avoided, if possible, until the mother’s condition can be stabilized but, if the status of the fetus is compromised, delivery is indicated.
Treatment of thyroid storm begins with stabilization of the patient, followed by initiation of a stepwise management approach (FIGURE 2).
FIGURE 2 Management of thyroid storm
Aggressive management of thyroid storm is indicated, following a stepwise approach. Each medication used to treat thyroid storm plays a specific role in suppressing thyroid function. Propylthiouracil (PTU) blocks additional synthesis of thyroid hormone and inhibits the conversion of thyroxine (T4) to triiodothyronine (T3). Methimazole blocks additional synthesis of thyroid hormones. Saturated solution of potassium iodide (SSKI), Lugol’s solution, and sodium iodide block the release of thyroid hormone from the gland. Dexamethasone is used to decrease thyroid hormone release and peripheral conversion of T4 to T3. Propranolol is used to treat maternal tachycardia by inhibiting the adrenergic effects of excessive thyroid hormones. Finally, phenobarbital is used to treat maternal agitation and restlessness caused by the increased catabolism of thyroid hormones.
SOURCE: Adapted from ACOG.2
Treatment of hyperthyroidism in pregnancy
Two medications are available to treat hyperthyroidism in pregnancy: propylthiouracil (PTU) and methimazole. These medications are known as thioamides.1,2
PTU blocks the oxidation of iodine in the thyroid gland, thereby preventing the synthesis of T4 and T3. The initial dosage for hyperthyroid women who are not pregnant is usually 300 to 450 mg/day in three divided doses every 8 hours, and this dosing strategy can also be applied to the pregnant patient. Maintenance therapy is usually achieved with 100 to 150 mg/day in divided doses every 8 to 12 hours.9
Methimazole works by blocking the organification of iodide, which decreases thyroid hormone production. The usual dosing, given in three divided doses every 8 hours, is 15 mg/day for mild hyperthyroidism, 30 to 40 mg/day for moderately severe hyperthyroidism, and 60 mg/day for severe hyperthyroidism. Maintenance therapy with methimazole is usually given at a dosage of 5 to 15 mg/day.9
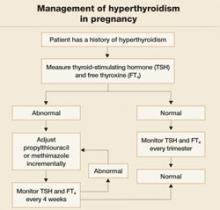
In the past, PTU was considered the drug of choice for treatment of hyperthyroidism in pregnancy because clinicians believed it crossed the placenta to a lesser degree than did methimazole, and because methimazole was associated with fetal esophageal and choanal atresia and fetal cutis aplasia (congenital skin defect of the scalp).1,2 Available evidence does not, however, support these conclusions.8,10 Whatever medication regimen you choose, thyroid function should be monitored 1) every 4 weeks until TSH and FT4 levels are within normal limits and 2) every trimester thereafter. FIGURE 3 presents an algorithm for managing hyperthyroidism in pregnancy.
FIGURE 3 Management of hyperthyroidism in pregnancy
CASE Resolved
The patient in thyroid storm described at the beginning of this article requires aggressive management, as outlined in the algorithm in FIGURE 2. As her symptoms diminish, fetal tachycardia resolves. The patient’s FT4 level begins to decline, consistent with appropriate treatment, and she is discharged home and instructed to continue PTU and labetalol and to follow up at the endocrinology and high-risk obstetrics clinics as soon as possible.
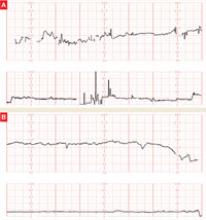
The patient does not follow this advice. Consequently, she presents at 33 5/7 weeks in a hypertensive crisis, with symptoms similar to those she first exhibited plus acute pulmonary edema. Fetal heart rate is initially in the 130s, with good variability and occasional decelerations (FIGURE 4A), but decelerations then become worse (FIGURE 4B) and emergency cesarean section is performed.
A male infant is delivered, weighing 2,390 g. Apgar scores are 0 at 1 minute and 9 at 5 minutes. A 25% placental abruption is noted at the time of delivery.
Mother and fetus are stabilized and discharged.
FIGURE 4 Weakening fetal status in a mother who is in thyroid storm
Fetal heart rate is initially in the 130s with good variability and occasional decelerations (A), but then deteriorates, with increasing decelerations (B), an indication for immediate delivery.
1. Casey BM, Leveno KJ. Thyroid disease in pregnancy. Obstet Gynecol. 2006;108:1283-1292.
2. American College of Obstetrics and Gynecology. ACOG Practice Bulletin. Clinical management guidelines for obstetrician-gynecologists. Number 37, August 2002. (Replaces Practice Bulletin Number 32, November 2001). Thyroid disease in pregnancy. Obstet Gynecol. 2002;100:387-396.
3. Mitchell ML, Klein RZ. The sequelae of untreated maternal hypothyroidism. Eur J Endocrinol. 2004;151 Suppl 3:U45-48.
4. Mestman JH. Endocrine diseases in pregnancy. In: Gabbe S, Niebyl JR, eds. Obstetrics: Normal and Problem Pregnancies. 4th ed. Philadelphia: Churchill Livingstone; 2002:1117-1168.
5. Davis LE, Leveno KJ, Cunningham FG. Hypothyroidism complicating pregnancy. Obstet Gynecol. 1988;72:108-112.
6. Davis LE, Lucas MJ, Hankins GD, Roark ML, Cunningham FG. Thyrotoxicosis complicating pregnancy. Am J Obstet Gynecol. 1989;160:63-70.
7. Kriplani A, Buckshee K, Bhargava VL, Takkar D, Ammini AC. Maternal and perinatal outcome in thyrotoxicosis complicating pregnancy. Eur J Obstet Gynecol Reprod Biol. 1994;54:159-163.
8. Belford MA. Navigating a thyroid storm. Contemporary OB/GYN. 2006; October:38–46.
9. Lazarus JH, Othman S. Thyroid disease in relation to pregnancy. Clin Endocrinol (Oxf). 1991;34:91-98.
10. Kent GN, Stuckey BG, Allen JR, Lambert T, Gee V. Postpartum thyroid dysfunction: clinical assessment and relationship to psychiatric affective morbidity. Clin Endocrinol (Oxf). 1999;51:429-438.
The authors report no financial relationships relevant to this article.
CASE Life on the line
A 32-year-old woman in the 24th week of her fourth pregnancy arrives at the emergency department complaining of cough and congestion, shortness of breath, and swelling in her face, hands, and feet. The swelling has become worse over the past 2 weeks, and she had several episodes of bloody vomiting the day before her visit. The patient says she has not experienced any leakage of fluid, vaginal bleeding, or contractions. She reports good fetal movement.
The patient’s medical history is unremarkable, but a review of systems reveals a 15-lb weight loss over the past 2 weeks, racing heart, worsening edema and shortness of breath, and diarrhea.
Physical findings include exophthalmia and an enlarged thyroid with a nodule on the right side, as well as bilateral rales, tachycardia, tremor, and increased deep tendon reflexes. There is no evidence of fetal cardiac failure or goiter.
A computed tomography (CT) scan of the mother shows bilateral pleural effusions indicative of high-output cardiac failure. Thyroid ultrasonography (US) reveals a diffusely enlarged thyroid gland with a right-sided mass.
The thyroid-stimulating hormone (TSH) level is undetectable. Fetal heart rate is in the 160s, with normal variability and occasional variable deceleration. Fetal US is consistent with the estimated gestational age and shows adequate amniotic fluid and no gross fetal anomalies.
What is the likely diagnosis?
This is a classic example of undiagnosed hyperthyroidism in pregnancy manifesting as thyroid storm.
As the case illustrates, uncontrolled hyperthyroidism in pregnancy poses a significant challenge for the obstetrician. The condition can cause miscarriage, preterm delivery, intrauterine growth restriction, preeclampsia, and—at its most dangerous—thyroid storm.1 Thyroid storm is a life-threatening emergency, and treatment must be initiated even before hyperthyroidism is confirmed by thyroid function testing.2 The good news is that these complications can be successfully avoided with adequate control of thyroid function.
Overt hyperthyroidism, seen in 0.2% of pregnancies, requires active intervention to avert adverse pregnancy outcome and neurologic damage to the fetus. Subclinical disease, seen in 1.7% of pregnancies, can also create serious obstetrical problems.1
The effects of hyperthyroidism in pregnancy vary in severity, ranging from the fairly innocuous, transient, and self-limited state called gestational transient thyrotoxicosis to the life-threatening emergency of thyroid storm. This review will update you on how to manage this disorder for optimal pregnancy outcome.
To screen or not to screen
Routine screening for thyroid dysfunction has been recommended for women who have infertility, menstrual disorders, or type 1 diabetes mellitus, and for pregnant women who have signs and symptoms of the disorder. Some authors recommend screening all pregnant women, but routine screening is not endorsed by the American College of Obstetricians and Gynecologists.2,3
Thyroid testing in pregnancy is recommended in women who:
- have a family history of autoimmune thyroid disease
- are on thyroid therapy
- have a goiter or
- have insulin-dependent diabetes mellitus.
Pregnant women who have a history of high-dose neck radiation, thyroid therapy, postpartum thyroiditis, or an infant born with thyroid disease should also be tested at the first prenatal visit.4
Telltale signs and laboratory tests
The signs and symptoms of hyperthyroidism can include nervousness, heat intolerance, tachycardia, palpitations, goiter, weight loss, thyromegaly, exophthalmia, increased appetite, nausea and vomiting, sweating, and tremor.1 The difficulty here? Many of these symptoms are also seen in pregnant women who have normal thyroid function, so that symptoms alone are not a reliable guide.
Instead, the diagnosis of overt hyperthyroidism is made on the basis of laboratory tests indicating suppressed TSH and elevated levels of free thyroxine (FT4) and free triiodothyronine (FT3). Subclinical hyperthyroidism is defined as a suppressed TSH level with normal FT4 and FT3 levels.2
The effects of hyperthyroidism on laboratory values are shown in TABLE 1. A form of hyperthyroidism called the T3– toxicosis syndrome is diagnosed by suppressed TSH, normal FT4, and elevated FT3 levels.4
TABLE 1
Is your pregnant patient hyperthyroid? Five-test lab panel offers a guide
| TEST AND RESULT | |||||
|---|---|---|---|---|---|
| THYROID-STIMULATING HORMONE | FREE TRI-IODOTHYRONINE | FREE THYROXINE | TOTAL TRI-IODOTHYRONINE | TOTAL THYROXINE | THEN THE MOTHER’S CONDITION IS … |
| No change | No change | ↑ | ↑ | ↑ | Pregnancy |
| ↓ | ↑ | ↑ | ↑ | ↑ | Hyperthyroidism |
| ↓ | No change | No change | No change | No change | Subclinical hyperthyroidism |
What are the causes?
The most common cause of hyperthyroidism in pregnancy—accounting for some 95% of cases—is Graves’ disease.2 This autoimmune disorder is characterized by autoantibodies that activate the TSH receptor. These autoantibodies cross the placenta and can cause fetal and neonatal thyroid dysfunction even when the mother herself is in a euthyroid condition.4
Far less often, hyperthyroidism in pregnancy has a cause other than Graves’ disease; TABLE 2 summarizes the possibilities.1 Other causes of hyperthyroidism in early pregnancy include choriocarcinoma and gestational trophoblastic disease (partial and complete moles) (TABLE 3).
TABLE 2
Causes of hyperthyroidism in pregnancy
| Graves’ disease |
| Adenoma |
| Toxic nodular goiter |
| Thyroiditis |
| Excessive thyroid hormone intake |
| Choriocarcinoma |
| Molar pregnancy |
TABLE 3
What causes severe hyperthyroidism before 20 weeks’ gestation?
| Gestational transient thyrotoxicosis |
| Choriocarcinoma |
Gestational trophoblastic disease
|
Signs and symptoms of Graves’ disease
Women who have Graves’ disease usually have thyroid nodules and may have exophthalmia, pretibial myxedema, and tachycardia. They also display other classic signs and symptoms of hyperthyroidism, such as muscle weakness, tremor, and warm and moist skin.
During pregnancy, Graves’ disease usually becomes worse during the first trimester and postpartum period; symptoms resolve during the second and third trimesters.1
Thyrotoxin receptor and antithyroid antibodies
Antithyroid antibodies are common in patients with autoimmune thyroid disease, as a response to thyroid antigens. The two most common antithyroid antibodies are thyroglobulin and thyroid peroxidase (anti-TPO). Anti-TPO antibodies are associated with postpartum thyroiditis and fetal and neonatal hyperthyroidism. TSH-receptor antibodies include thyroid-stimulating immunoglobulin (TSI) and TSH-receptor antibody. TSI is associated with Graves’ disease. TSH-receptor antibody is associated with fetal goiter, congenital hypothyroidism, and chronic thyroiditis without goiter.4
Who do you test for antibodies? Test for maternal thyroid antibodies in patients who:
- had Graves’ disease with fetal or neonatal hyperthyroidism in a previous pregnancy
- have active Graves’ disease being treated with antithyroid drugs
- are euthyroid or have undergone ablative therapy and have fetal tachycardia or intrauterine growth restriction
- have chronic thyroiditis without goiter
- have fetal goiter on ultrasound.
Newborns who have congenital hypothyroidism should also be screened for thyroid antibodies.4
What are the consequences?
Hyperthyroidism can have multiple effects on the pregnant patient and her fetus, ranging in severity from the minimal to the catastrophic.
Gestational transient thyrotoxicosis
This condition is presumably related to high levels of human chorionic gonadotropin, a substance known to stimulate TSH receptors. Unhappily for your patient, the condition is usually heralded by severe bouts of nausea and vomiting starting at 4 to 8 weeks’ gestation. Laboratory tests show significantly elevated levels of FT4 and FT3 and suppressed TSH. Despite this significant derangement, patients generally have no evidence of a hypermetabolic state.
This condition resolves by 14 to 20 weeks of gestation, is not associated with poor pregnancy outcomes, and does not require treatment with antithyroid medication.1
Adverse pregnancy outcomes
Pregnant women who have uncontrolled hyperthyroidism are at increased risk of spontaneous miscarriage, congestive heart failure, preterm delivery, intrauterine growth restriction, and preeclampsia.1 Studies that evaluated pregnancy outcomes in 239 women with overt hyperthyroidism showed increased risk of adverse pregnancy outcomes, compared with treated, euthyroid women (FIGURE 1).5-7
FIGURE 1 Consequences of uncontrolled hyperthyroidism
Several studies have found a much higher risk of pregnancy complications in women who have uncontrolled hyperthyroidism, compared with their treated and euthyroid peers.5-7
PTD=preterm delivery; FGR=fetal growth restrictions.
Fetal and neonatal hyperthyroidism
Hyperthyroidism in the fetus or newborn is caused by placental transfer of maternal immunoglobulin antibodies (TSI) to the fetus and is associated with maternal Graves’ disease. The incidence of neonatal hyperthyroidism is less than 1%. It can be predicted by rising levels of maternal TSI antibodies, to the point where levels in the third trimester are three to five times higher than they were at the beginning of pregnancy.4
Fetal hyperthyroidism develops at about 22 to 24 weeks’ gestation in mothers with a history of Graves’ disease who have been treated surgically or with ablative therapy prior to pregnancy. Even when these therapies achieve a euthyroid state in the mother, TSI levels may remain elevated and lead to fetal hyperthyroidism.
Characteristics of hyperthyroidism in the fetus include tachycardia, intrauterine growth restriction, congestive heart failure, oligohydramnios, and goiter. Treating the mother with antithyroid medications will ameliorate symptoms in the fetus.4
Thyroid storm
This is the worst-case scenario—a rare but potentially lethal complication of uncontrolled hyperthyroidism. Thyroid storm is a hypermetabolic state characterized by fever, nausea, vomiting, diarrhea, tachycardia, altered mental status, restlessness, nervousness, seizures, coma, and cardiac arrhythmias. It occurs in 1% to 2% of patients receiving thioamide therapy.8
In most instances, thyroid storm is a complication of uncontrolled hyperthyroidism, but it can also be precipitated by infection, surgery, thromboembolism, preeclampsia, labor, and delivery.
Thyroid storm is a medical emergency
This manifestation of uncontrolled hyperthyroidism is so urgent that treatment should be initiated before the results of TSH, FT4, and FT3 tests are available.2,8 Delivery should be avoided, if possible, until the mother’s condition can be stabilized but, if the status of the fetus is compromised, delivery is indicated.
Treatment of thyroid storm begins with stabilization of the patient, followed by initiation of a stepwise management approach (FIGURE 2).
FIGURE 2 Management of thyroid storm
Aggressive management of thyroid storm is indicated, following a stepwise approach. Each medication used to treat thyroid storm plays a specific role in suppressing thyroid function. Propylthiouracil (PTU) blocks additional synthesis of thyroid hormone and inhibits the conversion of thyroxine (T4) to triiodothyronine (T3). Methimazole blocks additional synthesis of thyroid hormones. Saturated solution of potassium iodide (SSKI), Lugol’s solution, and sodium iodide block the release of thyroid hormone from the gland. Dexamethasone is used to decrease thyroid hormone release and peripheral conversion of T4 to T3. Propranolol is used to treat maternal tachycardia by inhibiting the adrenergic effects of excessive thyroid hormones. Finally, phenobarbital is used to treat maternal agitation and restlessness caused by the increased catabolism of thyroid hormones.
SOURCE: Adapted from ACOG.2
Treatment of hyperthyroidism in pregnancy
Two medications are available to treat hyperthyroidism in pregnancy: propylthiouracil (PTU) and methimazole. These medications are known as thioamides.1,2
PTU blocks the oxidation of iodine in the thyroid gland, thereby preventing the synthesis of T4 and T3. The initial dosage for hyperthyroid women who are not pregnant is usually 300 to 450 mg/day in three divided doses every 8 hours, and this dosing strategy can also be applied to the pregnant patient. Maintenance therapy is usually achieved with 100 to 150 mg/day in divided doses every 8 to 12 hours.9
Methimazole works by blocking the organification of iodide, which decreases thyroid hormone production. The usual dosing, given in three divided doses every 8 hours, is 15 mg/day for mild hyperthyroidism, 30 to 40 mg/day for moderately severe hyperthyroidism, and 60 mg/day for severe hyperthyroidism. Maintenance therapy with methimazole is usually given at a dosage of 5 to 15 mg/day.9
In the past, PTU was considered the drug of choice for treatment of hyperthyroidism in pregnancy because clinicians believed it crossed the placenta to a lesser degree than did methimazole, and because methimazole was associated with fetal esophageal and choanal atresia and fetal cutis aplasia (congenital skin defect of the scalp).1,2 Available evidence does not, however, support these conclusions.8,10 Whatever medication regimen you choose, thyroid function should be monitored 1) every 4 weeks until TSH and FT4 levels are within normal limits and 2) every trimester thereafter. FIGURE 3 presents an algorithm for managing hyperthyroidism in pregnancy.
FIGURE 3 Management of hyperthyroidism in pregnancy
CASE Resolved
The patient in thyroid storm described at the beginning of this article requires aggressive management, as outlined in the algorithm in FIGURE 2. As her symptoms diminish, fetal tachycardia resolves. The patient’s FT4 level begins to decline, consistent with appropriate treatment, and she is discharged home and instructed to continue PTU and labetalol and to follow up at the endocrinology and high-risk obstetrics clinics as soon as possible.
The patient does not follow this advice. Consequently, she presents at 33 5/7 weeks in a hypertensive crisis, with symptoms similar to those she first exhibited plus acute pulmonary edema. Fetal heart rate is initially in the 130s, with good variability and occasional decelerations (FIGURE 4A), but decelerations then become worse (FIGURE 4B) and emergency cesarean section is performed.
A male infant is delivered, weighing 2,390 g. Apgar scores are 0 at 1 minute and 9 at 5 minutes. A 25% placental abruption is noted at the time of delivery.
Mother and fetus are stabilized and discharged.
FIGURE 4 Weakening fetal status in a mother who is in thyroid storm
Fetal heart rate is initially in the 130s with good variability and occasional decelerations (A), but then deteriorates, with increasing decelerations (B), an indication for immediate delivery.
The authors report no financial relationships relevant to this article.
CASE Life on the line
A 32-year-old woman in the 24th week of her fourth pregnancy arrives at the emergency department complaining of cough and congestion, shortness of breath, and swelling in her face, hands, and feet. The swelling has become worse over the past 2 weeks, and she had several episodes of bloody vomiting the day before her visit. The patient says she has not experienced any leakage of fluid, vaginal bleeding, or contractions. She reports good fetal movement.
The patient’s medical history is unremarkable, but a review of systems reveals a 15-lb weight loss over the past 2 weeks, racing heart, worsening edema and shortness of breath, and diarrhea.
Physical findings include exophthalmia and an enlarged thyroid with a nodule on the right side, as well as bilateral rales, tachycardia, tremor, and increased deep tendon reflexes. There is no evidence of fetal cardiac failure or goiter.
A computed tomography (CT) scan of the mother shows bilateral pleural effusions indicative of high-output cardiac failure. Thyroid ultrasonography (US) reveals a diffusely enlarged thyroid gland with a right-sided mass.
The thyroid-stimulating hormone (TSH) level is undetectable. Fetal heart rate is in the 160s, with normal variability and occasional variable deceleration. Fetal US is consistent with the estimated gestational age and shows adequate amniotic fluid and no gross fetal anomalies.
What is the likely diagnosis?
This is a classic example of undiagnosed hyperthyroidism in pregnancy manifesting as thyroid storm.
As the case illustrates, uncontrolled hyperthyroidism in pregnancy poses a significant challenge for the obstetrician. The condition can cause miscarriage, preterm delivery, intrauterine growth restriction, preeclampsia, and—at its most dangerous—thyroid storm.1 Thyroid storm is a life-threatening emergency, and treatment must be initiated even before hyperthyroidism is confirmed by thyroid function testing.2 The good news is that these complications can be successfully avoided with adequate control of thyroid function.
Overt hyperthyroidism, seen in 0.2% of pregnancies, requires active intervention to avert adverse pregnancy outcome and neurologic damage to the fetus. Subclinical disease, seen in 1.7% of pregnancies, can also create serious obstetrical problems.1
The effects of hyperthyroidism in pregnancy vary in severity, ranging from the fairly innocuous, transient, and self-limited state called gestational transient thyrotoxicosis to the life-threatening emergency of thyroid storm. This review will update you on how to manage this disorder for optimal pregnancy outcome.
To screen or not to screen
Routine screening for thyroid dysfunction has been recommended for women who have infertility, menstrual disorders, or type 1 diabetes mellitus, and for pregnant women who have signs and symptoms of the disorder. Some authors recommend screening all pregnant women, but routine screening is not endorsed by the American College of Obstetricians and Gynecologists.2,3
Thyroid testing in pregnancy is recommended in women who:
- have a family history of autoimmune thyroid disease
- are on thyroid therapy
- have a goiter or
- have insulin-dependent diabetes mellitus.
Pregnant women who have a history of high-dose neck radiation, thyroid therapy, postpartum thyroiditis, or an infant born with thyroid disease should also be tested at the first prenatal visit.4
Telltale signs and laboratory tests
The signs and symptoms of hyperthyroidism can include nervousness, heat intolerance, tachycardia, palpitations, goiter, weight loss, thyromegaly, exophthalmia, increased appetite, nausea and vomiting, sweating, and tremor.1 The difficulty here? Many of these symptoms are also seen in pregnant women who have normal thyroid function, so that symptoms alone are not a reliable guide.
Instead, the diagnosis of overt hyperthyroidism is made on the basis of laboratory tests indicating suppressed TSH and elevated levels of free thyroxine (FT4) and free triiodothyronine (FT3). Subclinical hyperthyroidism is defined as a suppressed TSH level with normal FT4 and FT3 levels.2
The effects of hyperthyroidism on laboratory values are shown in TABLE 1. A form of hyperthyroidism called the T3– toxicosis syndrome is diagnosed by suppressed TSH, normal FT4, and elevated FT3 levels.4
TABLE 1
Is your pregnant patient hyperthyroid? Five-test lab panel offers a guide
| TEST AND RESULT | |||||
|---|---|---|---|---|---|
| THYROID-STIMULATING HORMONE | FREE TRI-IODOTHYRONINE | FREE THYROXINE | TOTAL TRI-IODOTHYRONINE | TOTAL THYROXINE | THEN THE MOTHER’S CONDITION IS … |
| No change | No change | ↑ | ↑ | ↑ | Pregnancy |
| ↓ | ↑ | ↑ | ↑ | ↑ | Hyperthyroidism |
| ↓ | No change | No change | No change | No change | Subclinical hyperthyroidism |
What are the causes?
The most common cause of hyperthyroidism in pregnancy—accounting for some 95% of cases—is Graves’ disease.2 This autoimmune disorder is characterized by autoantibodies that activate the TSH receptor. These autoantibodies cross the placenta and can cause fetal and neonatal thyroid dysfunction even when the mother herself is in a euthyroid condition.4
Far less often, hyperthyroidism in pregnancy has a cause other than Graves’ disease; TABLE 2 summarizes the possibilities.1 Other causes of hyperthyroidism in early pregnancy include choriocarcinoma and gestational trophoblastic disease (partial and complete moles) (TABLE 3).
TABLE 2
Causes of hyperthyroidism in pregnancy
| Graves’ disease |
| Adenoma |
| Toxic nodular goiter |
| Thyroiditis |
| Excessive thyroid hormone intake |
| Choriocarcinoma |
| Molar pregnancy |
TABLE 3
What causes severe hyperthyroidism before 20 weeks’ gestation?
| Gestational transient thyrotoxicosis |
| Choriocarcinoma |
Gestational trophoblastic disease
|
Signs and symptoms of Graves’ disease
Women who have Graves’ disease usually have thyroid nodules and may have exophthalmia, pretibial myxedema, and tachycardia. They also display other classic signs and symptoms of hyperthyroidism, such as muscle weakness, tremor, and warm and moist skin.
During pregnancy, Graves’ disease usually becomes worse during the first trimester and postpartum period; symptoms resolve during the second and third trimesters.1
Thyrotoxin receptor and antithyroid antibodies
Antithyroid antibodies are common in patients with autoimmune thyroid disease, as a response to thyroid antigens. The two most common antithyroid antibodies are thyroglobulin and thyroid peroxidase (anti-TPO). Anti-TPO antibodies are associated with postpartum thyroiditis and fetal and neonatal hyperthyroidism. TSH-receptor antibodies include thyroid-stimulating immunoglobulin (TSI) and TSH-receptor antibody. TSI is associated with Graves’ disease. TSH-receptor antibody is associated with fetal goiter, congenital hypothyroidism, and chronic thyroiditis without goiter.4
Who do you test for antibodies? Test for maternal thyroid antibodies in patients who:
- had Graves’ disease with fetal or neonatal hyperthyroidism in a previous pregnancy
- have active Graves’ disease being treated with antithyroid drugs
- are euthyroid or have undergone ablative therapy and have fetal tachycardia or intrauterine growth restriction
- have chronic thyroiditis without goiter
- have fetal goiter on ultrasound.
Newborns who have congenital hypothyroidism should also be screened for thyroid antibodies.4
What are the consequences?
Hyperthyroidism can have multiple effects on the pregnant patient and her fetus, ranging in severity from the minimal to the catastrophic.
Gestational transient thyrotoxicosis
This condition is presumably related to high levels of human chorionic gonadotropin, a substance known to stimulate TSH receptors. Unhappily for your patient, the condition is usually heralded by severe bouts of nausea and vomiting starting at 4 to 8 weeks’ gestation. Laboratory tests show significantly elevated levels of FT4 and FT3 and suppressed TSH. Despite this significant derangement, patients generally have no evidence of a hypermetabolic state.
This condition resolves by 14 to 20 weeks of gestation, is not associated with poor pregnancy outcomes, and does not require treatment with antithyroid medication.1
Adverse pregnancy outcomes
Pregnant women who have uncontrolled hyperthyroidism are at increased risk of spontaneous miscarriage, congestive heart failure, preterm delivery, intrauterine growth restriction, and preeclampsia.1 Studies that evaluated pregnancy outcomes in 239 women with overt hyperthyroidism showed increased risk of adverse pregnancy outcomes, compared with treated, euthyroid women (FIGURE 1).5-7
FIGURE 1 Consequences of uncontrolled hyperthyroidism
Several studies have found a much higher risk of pregnancy complications in women who have uncontrolled hyperthyroidism, compared with their treated and euthyroid peers.5-7
PTD=preterm delivery; FGR=fetal growth restrictions.
Fetal and neonatal hyperthyroidism
Hyperthyroidism in the fetus or newborn is caused by placental transfer of maternal immunoglobulin antibodies (TSI) to the fetus and is associated with maternal Graves’ disease. The incidence of neonatal hyperthyroidism is less than 1%. It can be predicted by rising levels of maternal TSI antibodies, to the point where levels in the third trimester are three to five times higher than they were at the beginning of pregnancy.4
Fetal hyperthyroidism develops at about 22 to 24 weeks’ gestation in mothers with a history of Graves’ disease who have been treated surgically or with ablative therapy prior to pregnancy. Even when these therapies achieve a euthyroid state in the mother, TSI levels may remain elevated and lead to fetal hyperthyroidism.
Characteristics of hyperthyroidism in the fetus include tachycardia, intrauterine growth restriction, congestive heart failure, oligohydramnios, and goiter. Treating the mother with antithyroid medications will ameliorate symptoms in the fetus.4
Thyroid storm
This is the worst-case scenario—a rare but potentially lethal complication of uncontrolled hyperthyroidism. Thyroid storm is a hypermetabolic state characterized by fever, nausea, vomiting, diarrhea, tachycardia, altered mental status, restlessness, nervousness, seizures, coma, and cardiac arrhythmias. It occurs in 1% to 2% of patients receiving thioamide therapy.8
In most instances, thyroid storm is a complication of uncontrolled hyperthyroidism, but it can also be precipitated by infection, surgery, thromboembolism, preeclampsia, labor, and delivery.
Thyroid storm is a medical emergency
This manifestation of uncontrolled hyperthyroidism is so urgent that treatment should be initiated before the results of TSH, FT4, and FT3 tests are available.2,8 Delivery should be avoided, if possible, until the mother’s condition can be stabilized but, if the status of the fetus is compromised, delivery is indicated.
Treatment of thyroid storm begins with stabilization of the patient, followed by initiation of a stepwise management approach (FIGURE 2).
FIGURE 2 Management of thyroid storm
Aggressive management of thyroid storm is indicated, following a stepwise approach. Each medication used to treat thyroid storm plays a specific role in suppressing thyroid function. Propylthiouracil (PTU) blocks additional synthesis of thyroid hormone and inhibits the conversion of thyroxine (T4) to triiodothyronine (T3). Methimazole blocks additional synthesis of thyroid hormones. Saturated solution of potassium iodide (SSKI), Lugol’s solution, and sodium iodide block the release of thyroid hormone from the gland. Dexamethasone is used to decrease thyroid hormone release and peripheral conversion of T4 to T3. Propranolol is used to treat maternal tachycardia by inhibiting the adrenergic effects of excessive thyroid hormones. Finally, phenobarbital is used to treat maternal agitation and restlessness caused by the increased catabolism of thyroid hormones.
SOURCE: Adapted from ACOG.2
Treatment of hyperthyroidism in pregnancy
Two medications are available to treat hyperthyroidism in pregnancy: propylthiouracil (PTU) and methimazole. These medications are known as thioamides.1,2
PTU blocks the oxidation of iodine in the thyroid gland, thereby preventing the synthesis of T4 and T3. The initial dosage for hyperthyroid women who are not pregnant is usually 300 to 450 mg/day in three divided doses every 8 hours, and this dosing strategy can also be applied to the pregnant patient. Maintenance therapy is usually achieved with 100 to 150 mg/day in divided doses every 8 to 12 hours.9
Methimazole works by blocking the organification of iodide, which decreases thyroid hormone production. The usual dosing, given in three divided doses every 8 hours, is 15 mg/day for mild hyperthyroidism, 30 to 40 mg/day for moderately severe hyperthyroidism, and 60 mg/day for severe hyperthyroidism. Maintenance therapy with methimazole is usually given at a dosage of 5 to 15 mg/day.9
In the past, PTU was considered the drug of choice for treatment of hyperthyroidism in pregnancy because clinicians believed it crossed the placenta to a lesser degree than did methimazole, and because methimazole was associated with fetal esophageal and choanal atresia and fetal cutis aplasia (congenital skin defect of the scalp).1,2 Available evidence does not, however, support these conclusions.8,10 Whatever medication regimen you choose, thyroid function should be monitored 1) every 4 weeks until TSH and FT4 levels are within normal limits and 2) every trimester thereafter. FIGURE 3 presents an algorithm for managing hyperthyroidism in pregnancy.
FIGURE 3 Management of hyperthyroidism in pregnancy
CASE Resolved
The patient in thyroid storm described at the beginning of this article requires aggressive management, as outlined in the algorithm in FIGURE 2. As her symptoms diminish, fetal tachycardia resolves. The patient’s FT4 level begins to decline, consistent with appropriate treatment, and she is discharged home and instructed to continue PTU and labetalol and to follow up at the endocrinology and high-risk obstetrics clinics as soon as possible.
The patient does not follow this advice. Consequently, she presents at 33 5/7 weeks in a hypertensive crisis, with symptoms similar to those she first exhibited plus acute pulmonary edema. Fetal heart rate is initially in the 130s, with good variability and occasional decelerations (FIGURE 4A), but decelerations then become worse (FIGURE 4B) and emergency cesarean section is performed.
A male infant is delivered, weighing 2,390 g. Apgar scores are 0 at 1 minute and 9 at 5 minutes. A 25% placental abruption is noted at the time of delivery.
Mother and fetus are stabilized and discharged.
FIGURE 4 Weakening fetal status in a mother who is in thyroid storm
Fetal heart rate is initially in the 130s with good variability and occasional decelerations (A), but then deteriorates, with increasing decelerations (B), an indication for immediate delivery.
1. Casey BM, Leveno KJ. Thyroid disease in pregnancy. Obstet Gynecol. 2006;108:1283-1292.
2. American College of Obstetrics and Gynecology. ACOG Practice Bulletin. Clinical management guidelines for obstetrician-gynecologists. Number 37, August 2002. (Replaces Practice Bulletin Number 32, November 2001). Thyroid disease in pregnancy. Obstet Gynecol. 2002;100:387-396.
3. Mitchell ML, Klein RZ. The sequelae of untreated maternal hypothyroidism. Eur J Endocrinol. 2004;151 Suppl 3:U45-48.
4. Mestman JH. Endocrine diseases in pregnancy. In: Gabbe S, Niebyl JR, eds. Obstetrics: Normal and Problem Pregnancies. 4th ed. Philadelphia: Churchill Livingstone; 2002:1117-1168.
5. Davis LE, Leveno KJ, Cunningham FG. Hypothyroidism complicating pregnancy. Obstet Gynecol. 1988;72:108-112.
6. Davis LE, Lucas MJ, Hankins GD, Roark ML, Cunningham FG. Thyrotoxicosis complicating pregnancy. Am J Obstet Gynecol. 1989;160:63-70.
7. Kriplani A, Buckshee K, Bhargava VL, Takkar D, Ammini AC. Maternal and perinatal outcome in thyrotoxicosis complicating pregnancy. Eur J Obstet Gynecol Reprod Biol. 1994;54:159-163.
8. Belford MA. Navigating a thyroid storm. Contemporary OB/GYN. 2006; October:38–46.
9. Lazarus JH, Othman S. Thyroid disease in relation to pregnancy. Clin Endocrinol (Oxf). 1991;34:91-98.
10. Kent GN, Stuckey BG, Allen JR, Lambert T, Gee V. Postpartum thyroid dysfunction: clinical assessment and relationship to psychiatric affective morbidity. Clin Endocrinol (Oxf). 1999;51:429-438.
1. Casey BM, Leveno KJ. Thyroid disease in pregnancy. Obstet Gynecol. 2006;108:1283-1292.
2. American College of Obstetrics and Gynecology. ACOG Practice Bulletin. Clinical management guidelines for obstetrician-gynecologists. Number 37, August 2002. (Replaces Practice Bulletin Number 32, November 2001). Thyroid disease in pregnancy. Obstet Gynecol. 2002;100:387-396.
3. Mitchell ML, Klein RZ. The sequelae of untreated maternal hypothyroidism. Eur J Endocrinol. 2004;151 Suppl 3:U45-48.
4. Mestman JH. Endocrine diseases in pregnancy. In: Gabbe S, Niebyl JR, eds. Obstetrics: Normal and Problem Pregnancies. 4th ed. Philadelphia: Churchill Livingstone; 2002:1117-1168.
5. Davis LE, Leveno KJ, Cunningham FG. Hypothyroidism complicating pregnancy. Obstet Gynecol. 1988;72:108-112.
6. Davis LE, Lucas MJ, Hankins GD, Roark ML, Cunningham FG. Thyrotoxicosis complicating pregnancy. Am J Obstet Gynecol. 1989;160:63-70.
7. Kriplani A, Buckshee K, Bhargava VL, Takkar D, Ammini AC. Maternal and perinatal outcome in thyrotoxicosis complicating pregnancy. Eur J Obstet Gynecol Reprod Biol. 1994;54:159-163.
8. Belford MA. Navigating a thyroid storm. Contemporary OB/GYN. 2006; October:38–46.
9. Lazarus JH, Othman S. Thyroid disease in relation to pregnancy. Clin Endocrinol (Oxf). 1991;34:91-98.
10. Kent GN, Stuckey BG, Allen JR, Lambert T, Gee V. Postpartum thyroid dysfunction: clinical assessment and relationship to psychiatric affective morbidity. Clin Endocrinol (Oxf). 1999;51:429-438.
How to manage hypothyroid disease in pregnancy
The authors report no financial relationships relevant to this article.
A pregnant woman whose thyroid gland isn’t doing its job presents a serious management problem for her obstetrician. If she has overt hypothyroidism, seen in between 0.3% and 2.5% of pregnancies, active intervention is required to prevent serious damage to the fetus.1,2 Even if she has subclinical disease, seen in 2% to 3% of pregnancies, current research indicates that intervention may be indicated.
Fetal thyroxine requirements increase as early as 5 weeks of gestation, when the fetus is still dependent on maternal thyroxine. A deficiency of maternal thyroxine can have severe adverse outcomes, affecting the course of the pregnancy and the neurologic development of the fetus. To prevent such sequelae, patients who were on thyroid medication before pregnancy should increase the dosage by 30% once pregnancy is confirmed, and hypothyroidism that develops in pregnancy should be managed aggressively and meticulously.
Here, we’ll examine the published research to advise you on evidence-based approaches for diagnosis and management of this complex condition.
Maternal thyroid function
An elaborate negative-feedback loop prevails before pregnancy
In a nonpregnant woman, thyroid function is controlled by a negative-feedback loop that works like this:
- The hypothalamus releases thyroid-releasing hormone (TRH)
- TRH acts on the pituitary gland to release thyroid-stimulating hormone (TSH)
- TSH, in turn, acts on the thyroid gland to release the thyroid hormones iodothyronine (T3) and thyroxine (T4) that regulate metabolism
- TRH and TSH concentrations are inversely related to T3 and T4 concentrations. That is, the more TRH and TSH circulating in the blood stream, the less T3 and T4 will be produced by the thyroid gland3
- Almost all (approximately 99%) circulating T3 and T4 is bound to a protein called thyroxine-binding globulin (TBG). Only 1% of these hormones circulate in the free form, and only the free forms are biologically active.3
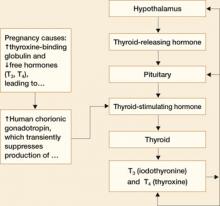
This relationship is illustrated in FIGURE 1.
FIGURE 1 Thyroid physiology and the impact of pregnancy
Pregnancy reduces free forms of T3 and T4, and increases TSH slightly
Pregnancy alters thyroid function in significant ways:
- Increases in circulating estrogen lead to the production of more TBG
- When TBG increases, more T3 and T4 are bound and fewer free forms of these hormones are available
- Because the total T3 (TT3) and total free T4 (TT4) are decreased in pregnancy, they are not good measures of thyroid function. Maternal thyroid function in pregnancy should be monitored using free T4 (FT4) and TSH levels
- Increased TBG also leads to a slight increase in TSH between the first trimester and term
- Human chorionic gonadotropin (hCG) concentrations also increase in pregnancy. Because hCG has thyrotropin-like activity, these higher levels cause a transient decrease in TSH by suppression of TSH production between approximately 8 and 14 weeks of gestation.
Fetal thyroid function
During early gestation, the fetus receives thyroid hormone from the mother.1 Maternal T4 crosses the placenta actively—the only hormone that does so.4 The fetus’s need for thyroxine starts to increase as early as 5 weeks of gestation.5
Fetal thyroid development does not begin until 10 to 12 weeks of gestation, and then continues until term. The fetus relies on maternal T4 exclusively before 12 weeks and partially thereafter for normal fetal neurologic development. It follows that maternal hypothyroidism could be detrimental to fetal development if not detected and corrected very early in gestation.
How (and whom) to screen for maternal hypothyroidism
Routine screening has been recommended for women who have infertility, menstrual disorders, or type 1 diabetes mellitus, and for pregnant women who have signs and symptoms of deficient thyroid function.6 In recent years, some authors have recommended screening all pregnant women for thyroid dysfunction, but such recommendations remain controversial.3,7,8 Routine screening is not endorsed by the American College of Obstetricians and Gynecologists.6
Symptoms overlap typical conditions of pregnancy
The difficulty here is that the characteristic signs and symptoms of hypothyroidism are very similar to physiologic conditions seen in most pregnancies. They include fatigue, constipation, cold intolerance, muscle cramps, hair loss, dry skin, brittle nails, weight gain, intellectual slowness, bradycardia, depression, insomnia, periorbital edema, myxedema, and myxedema coma.6 A side-by-side comparison of pregnancy conditions and hypothyroidism symptoms is provided in TABLE 1.
TABLE 1
Distinguishing hypothyroidism from a normal gestation can be challenging
| SYMPTOM | HYPOTHYROIDISM | PREGNANCY |
|---|---|---|
| Fatigue | • | • |
| Constipation | • | • |
| Hair loss | • | |
| Dry skin | • | |
| Brittle nails | • | |
| Weight gain | • | • |
| Fluid retention | • | • |
| Bradycardia | • | • |
| Goiter | • | |
| Carpal tunnel syndrome | • | • |
Which laboratory tests are informative?
Because screening is controversial and symptomatology does not reliably distinguish hypothyroidism from normal pregnancy, laboratory tests are the standard for diagnosis. Overt hypothyroidism is diagnosed in a symptomatic patient by elevated TSH level and low levels of FT4 and free T3 (FT3). Subclinical hypothyroidism is defined as elevated TSH with normal FT4 and FT3 in an asymptomatic patient. Level changes characteristic of normal pregnancy, overt hypothyroidism, and subclinical hypothyroidism are given in TABLE 2.6
TABLE 2
Laboratory diagnosis of hypothyroidism
| MATERNAL CONDITION | TSH | FREE T3 | FREE T4 | TOTAL T3 | TOTAL T4 |
|---|---|---|---|---|---|
| Normal pregnancy | No change | No change | ↑ | ↑ | ↑ |
| Hypothyroidism | ↑ | ↓ | ↓ | ↓ | ↓ |
| Subclinical hypothyroidism | ↑ | No change | No change | ↓ | ↓ |
| Adapted from American College of Obstetricians and Gynecologists6 | |||||
What causes hypothyroidism?
The most common cause of hypothyroidism in most of the world is iodine deficiency. In developed countries, however, where lack of iodine in the diet is not a problem, Hashimoto’s thyroiditis, also known as chronic autoimmune thyroiditis, is the most common cause. Hashimoto’s thyroiditis is characterized by the presence of antithyroid antibodies, including both thyroid antimicrosomial and antithyroglobulin antibodies. Both iodine deficiency and Hashimoto’s thyroiditis are associated with goiter.5 Other causes of hypothyroidism include radioactive iodine therapy for Graves’ disease, a condition we will discuss in Part 2 of this series in February; thyroidectomy; viral thyroiditis; pituitary tumors; Sheehan’s syndrome; and a number of medications.
Causes of hypothyroidism are summarized in TABLE 3.3
TABLE 3
Causes of hypothyroidism
| Iodine deficiency |
| Hashimoto’s thyroiditis |
| Radioactive iodine therapy |
| Thyroidectomy |
| Viral thyroiditis |
| Sheehan’s syndrome |
Medications
|
Effects vary by medication
Medications alter thyroid function in different ways. Iodine and lithium inhibit thyroid function and, along with dopamine antagonists, increase TSH levels. Conversely, thioamides, glucocorticoids, dopamine agonists, and somatostatins decrease TSH levels. Finally, ferrous sulfate, sucrafate, cholestyramine, and aluminum hydroxide antacids all inhibit gastrointestinal absorption of thyroid hormone and therefore should not be taken within 4 hours of thyroid medication.6
Maternal hypothyroidism: Effects on fetus, newborn
The impact of maternal hypothyroidism on the fetus depends on the severity of the condition.
- Uncontrolled hypothyroidism. The consequences of this condition can be dire. The possibilities include intrauterine fetal demise and stillbirth, preterm delivery, low birth weight, preeclampsia, and developmental anomalies including reduced intelligence quotient (IQ).1,2,4,6 Blazer and colleagues correlated intrauterine growth with maternal TSH and fetal FT4 and concluded that impaired intrauterine growth is related to abnormal thyroid function and might reflect an insufficient level of hormone production by hypothyroid mothers during pregnancy.9 Maternal and congenital hypothyroidism resulting from severe iodine deficiency are associated with profound neurologic impairment and mental retardation.1,3,10 If the condition is left untreated, cretinism can occur. Congenital cretinism is associated with growth failure, mental retardation, and other neuropsychologic deficits including deaf-mutism.3,4 However, if cretinism is identified and treated in the first 3 months of life, near-normal growth and intelligence can be expected.6 For this reason, all 50 states and the District of Columbia require newborn screening for congenital hypothyroidism.6
- Asymptomatic overt hypothyroidism. Several studies have evaluated neonatal outcomes in pregnancy complicated by asymptomatic overt hypothyroidism—that is, women who had previously been diagnosed with hypothyroidism, who have abnormal TSH and FT4 levels, but who do not have symptoms. Pop and colleagues have shown impaired psychomotor development at 10 months in infants born to mothers who had low T4 during the first 12 weeks of gestation.7 Haddow and colleagues correlated elevated maternal TSH levels at less than 17 weeks’ gestation with low IQ scores in the offspring at 7 to 9 years of age.8 Klein and colleagues demonstrated an inverse correlation between a woman’s TSH level during pregnancy and the IQ of her offspring.11 Kooistra and colleagues confirmed that maternal hypothyroxinemia is a risk for neurodevelopmental abnormalities that can be identified as early as 3 weeks of age.12 Studies of this relationship are summarized in TABLE 4.
- Subclinical hypothyroidism. During the past decade, researchers have focused attention on neonatal neurologic function in infants born to mothers who had subclinical disease. Mitchell and Klein evaluated the prevalence of subclinical hypothyroidism at less than 17 weeks’ gestation and subsequently compared the IQs in these children with those of controls.4 They found the mean and standard-deviation IQs of the children in the control and treated groups to be significantly higher than those of the children whose mothers were not treated. Casey and colleagues evaluated pregnancy outcomes in women who had undiagnosed subclinical hypothyroidism.10 They found that such pregnancies were more likely to be complicated by placental abruption and preterm birth, and speculated that the reduced IQ demonstrated in the Mitchell and Klein study might have been related to the effects of prematurity.
TABLE 4
Fetal and neonatal effects of asymptomatic overt hypothyroidism
| STUDY | LABORATORY FINDINGS | OUTCOMES AND RECOMMENDATIONS |
|---|---|---|
| Kooistra et al12 | ↓ FT4 | Maternal hypothyroxinemia is a risk for neurodevelopmental abnormalities as early as 3 weeks of age |
| Casey et al10 | ↑ TSH | Pregnancies with undiagnosed subclinical hypothyroidism were more likely to be complicated by placental abruption and preterm birth. The reduced IQ seen in a prior study (Mitchell and Klein4) may be related to effects of prematurity |
| Mitchell and Klein4 | ↑ TSH | The mean and standard deviation of IQs of the children of treated mothers with hypothyroidism and the control group were significantly higher than those for children of untreated hypothyroid women |
| Blazer et al9 | ↑ maternal TSH, ↑ fetal FT4 | Impaired intrauterine growth may reflect insufficient levels of hormone replacement therapy in hypothyroid mothers during pregnancy |
| Pop et al7 | ↓ FT4 | Impaired psychomotor development at 10 months of age in offspring of mothers with low T4 at ≤12 weeks |
| Haddow et al8 | ↑ TSH, ↓ FT4 | Elevated TSH levels at <17 weeks’ gestation are associated with low IQ scores at 7 to 9 years of age. Routine screening for thyroid deficiency may be warranted |
| Klein et al11 | ↑ TSH, ↓ FT4, ↓ TT4 | Inverse correlation between TSH during pregnancy and IQ of offspring |
| FT4=free thyroxine, TSH=thyroid-stimulating hormone, TT4=total thyroxine | ||
Managing hypothyroidism in pregnancy
The treatment of choice for correction of hypothyroidism is synthetic T4, or levothyroxine (Levothyroid, Levoxyl, Synthroid, and Unithroid). Initial treatment in the nonpregnant patient is 1.7 μg/kg/day or 12.5 to 25 μg/day adjusted by 25 μg/day every 2 to 4 weeks until a euthyroid state is achieved.13
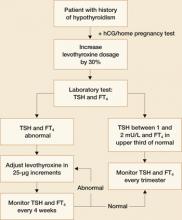
Patients who were on thyroxine therapy before pregnancy should increase the dose by 30% once pregnancy is confirmed.1,5 Serum thyrotropin levels should be monitored every 4 weeks to maintain a TSH level between 1 and 2 mU/L and FT4 in upper third of normal.1 Once a euthyroid state has been achieved, thyrotropin levels should be monitored every trimester until delivery. FIGURE 2 provides an algorithm for management of hypothyroidism in pregnancy.
FIGURE 2 During pregnancy, thyroid function merits regular monitoring, fine-tuning of treatment
Postpartum thyroiditis
About 5% of all obstetrical patients develop postpartum thyroiditis. Approximately 45% of these women present with hypothyroidism, with the rest evenly divided between thyrotoxicosis (hyperthyroidism) and thyrotoxicosis followed by hypothyroidism. Unfortunately, the signs and symptoms of hypo- and hyperthyroidism are similar to the postpartum state. Many of these patients are not diagnosed. A high index of suspicion warrants thyroid function testing. Women who have a history of type 1 diabetes mellitus have a 25% chance of developing postpartum thyroid dysfunction.
The diagnosis is made by documenting abnormal levels of TSH and FT4. Postpartum hyperthyroidism may be diagnosed by the presence of antimicrosomal or thyroperoxidase antithyroid peroxidase antibodies. Goiter may be present in up to 50% of patients.
Postpartum thyroiditis has two phases
The first phase, also known as the thyrotoxic phase, occurs 1 to 4 months after delivery when transient thyrotoxicosis develops from excessive release of thyroid hormones. The most common symptoms with early postpartum thyroiditis are fatigue and palpitations. Approximately 67% of these women will return to a euthyroid state, and thioamide therapy is generally considered ineffective. Hypothyroidism can develop within 1 month of the onset of thyroiditis.
The second phase occurs between 4 and 8 months postpartum, and these women present with hypothyroidism. Thyromegaly and associated symptoms are common. Unlike the first (thyrotoxic) phase, medical treatment is recommended. Thyroxine treatment should be initiated and maintained for 6 to 12 months. Postpartum thyroiditis carries a 30% risk of recurrence.14
Postpartum thyroiditis may be associated with depression or aggravate symptoms of depression, although the data on this association are conflicting. The largest study addressing this issue concluded that there was no difference in the clinical and psychiatric signs and symptoms between postpartum thyroiditis and controls.15 Nevertheless, it would seem prudent to evaluate thyroid function in postpartum depression if other signs of thyroid dysfunction are present.
1. Idris I, Srinivasan R, Simm A, Page RC. Effects of maternal hyperthyroidism during early gestation on neonatal and obstetric outcome. Clin Endocrinol. 2006;65:133-135.
2. Girling JC. Thyroid disorders in pregnancy. Curr Obstet Gynecol. 2006;16:47-53.
3. Creasy RK, Resnik R, Iams J. Maternal–Fetal Medicine. 5th ed. Philadelphia, Pa: Saunders Elsevier; 2004:1063-1082.
4. Mitchell ML, Klein RZ. The sequelae of untreated maternal hypothyroidism. Eur J Endocrinol. 2004;151 Suppl 3:U45-U48.
5. Alexander EK, Marqusee E, Lawrence J, Jarolim P, Fischer GA, Larsen PR. Timing and magnitude of increases in levothyroxine requirements during pregnancy in women with hypothyroidism. N Engl J Med. 2004;351:241-249.
6. American College of Obstetrics and Gynecology. ACOG Practice Bulletin. Clinical management guidelines for obstetrician-gynecologists. Number 37, August 2002. (Replaces Practice Bulletin Number 32, November 2001). Thyroid disease in pregnancy. Obstet Gynecol. 2002;100:387-396.
7. Pop VJ, Kuijpens JL, van Baar AL, et al. Low maternal free thyroxine concentrations during early pregnancy are associated with impaired psychomotor development in infancy. Clin Endocrinol. 1999;50:149-155.
8. Haddow JE, Palomaki GE, Allan WC, et al. Maternal thyroid deficiency during pregnancy and subsequent neuropsychological development of the child. N Engl J Med. 1999;341:549-555.
9. Blazer S, Moreh-Waterman Y, Miller-Lotan R, Tamir A, Hochberg Z. Maternal hypothyroidism may affect fetal growth and neonatal thyroid function. Obstet Gynecol. 2003;102:232-241.
10. Casey BM, Dashe JS, Wells CE, et al. Subclinical hypothyroidism and pregnancy outcomes. Obstet Gynecol. 2005;105:239-245.
11. Klein RZ, Haddow JE, Faix JD, et al. Prevalence of thyroid deficiency in pregnant women. Clin Endocrinol. 1991;35:41-46.
12. Kooistra L, Crawford S, van Baar AL, Brouwers EP, Pop VJ. Neonatal effects of maternal hypothyroxinemia during early pregnancy. Pediatrics. 2006;117:161-167.
13. Levothyroxine: Drug information. Lexicomp. http://www.utdol.com/utd/content/topic.do?topicKey=drug_l_z/143814&type=A&selectedTitle=2~39. Accessed December 14, 2007.
14. Casey BM, Leveno KJ. Thyroid disease in pregnancy. Obstet Gynecol. 2006;108:1283-1292.
15. Kent GN, Stuckey BG, Allen JR, Lambert T, Gee V. Postpartum thyroid dysfunction: clinical assessment and relationship to psychiatric affective morbidity. Clin Endocrinol. 1999;51:429-438.
The authors report no financial relationships relevant to this article.
A pregnant woman whose thyroid gland isn’t doing its job presents a serious management problem for her obstetrician. If she has overt hypothyroidism, seen in between 0.3% and 2.5% of pregnancies, active intervention is required to prevent serious damage to the fetus.1,2 Even if she has subclinical disease, seen in 2% to 3% of pregnancies, current research indicates that intervention may be indicated.
Fetal thyroxine requirements increase as early as 5 weeks of gestation, when the fetus is still dependent on maternal thyroxine. A deficiency of maternal thyroxine can have severe adverse outcomes, affecting the course of the pregnancy and the neurologic development of the fetus. To prevent such sequelae, patients who were on thyroid medication before pregnancy should increase the dosage by 30% once pregnancy is confirmed, and hypothyroidism that develops in pregnancy should be managed aggressively and meticulously.
Here, we’ll examine the published research to advise you on evidence-based approaches for diagnosis and management of this complex condition.
Maternal thyroid function
An elaborate negative-feedback loop prevails before pregnancy
In a nonpregnant woman, thyroid function is controlled by a negative-feedback loop that works like this:
- The hypothalamus releases thyroid-releasing hormone (TRH)
- TRH acts on the pituitary gland to release thyroid-stimulating hormone (TSH)
- TSH, in turn, acts on the thyroid gland to release the thyroid hormones iodothyronine (T3) and thyroxine (T4) that regulate metabolism
- TRH and TSH concentrations are inversely related to T3 and T4 concentrations. That is, the more TRH and TSH circulating in the blood stream, the less T3 and T4 will be produced by the thyroid gland3
- Almost all (approximately 99%) circulating T3 and T4 is bound to a protein called thyroxine-binding globulin (TBG). Only 1% of these hormones circulate in the free form, and only the free forms are biologically active.3
This relationship is illustrated in FIGURE 1.
FIGURE 1 Thyroid physiology and the impact of pregnancy
Pregnancy reduces free forms of T3 and T4, and increases TSH slightly
Pregnancy alters thyroid function in significant ways:
- Increases in circulating estrogen lead to the production of more TBG
- When TBG increases, more T3 and T4 are bound and fewer free forms of these hormones are available
- Because the total T3 (TT3) and total free T4 (TT4) are decreased in pregnancy, they are not good measures of thyroid function. Maternal thyroid function in pregnancy should be monitored using free T4 (FT4) and TSH levels
- Increased TBG also leads to a slight increase in TSH between the first trimester and term
- Human chorionic gonadotropin (hCG) concentrations also increase in pregnancy. Because hCG has thyrotropin-like activity, these higher levels cause a transient decrease in TSH by suppression of TSH production between approximately 8 and 14 weeks of gestation.
Fetal thyroid function
During early gestation, the fetus receives thyroid hormone from the mother.1 Maternal T4 crosses the placenta actively—the only hormone that does so.4 The fetus’s need for thyroxine starts to increase as early as 5 weeks of gestation.5
Fetal thyroid development does not begin until 10 to 12 weeks of gestation, and then continues until term. The fetus relies on maternal T4 exclusively before 12 weeks and partially thereafter for normal fetal neurologic development. It follows that maternal hypothyroidism could be detrimental to fetal development if not detected and corrected very early in gestation.
How (and whom) to screen for maternal hypothyroidism
Routine screening has been recommended for women who have infertility, menstrual disorders, or type 1 diabetes mellitus, and for pregnant women who have signs and symptoms of deficient thyroid function.6 In recent years, some authors have recommended screening all pregnant women for thyroid dysfunction, but such recommendations remain controversial.3,7,8 Routine screening is not endorsed by the American College of Obstetricians and Gynecologists.6
Symptoms overlap typical conditions of pregnancy
The difficulty here is that the characteristic signs and symptoms of hypothyroidism are very similar to physiologic conditions seen in most pregnancies. They include fatigue, constipation, cold intolerance, muscle cramps, hair loss, dry skin, brittle nails, weight gain, intellectual slowness, bradycardia, depression, insomnia, periorbital edema, myxedema, and myxedema coma.6 A side-by-side comparison of pregnancy conditions and hypothyroidism symptoms is provided in TABLE 1.
TABLE 1
Distinguishing hypothyroidism from a normal gestation can be challenging
| SYMPTOM | HYPOTHYROIDISM | PREGNANCY |
|---|---|---|
| Fatigue | • | • |
| Constipation | • | • |
| Hair loss | • | |
| Dry skin | • | |
| Brittle nails | • | |
| Weight gain | • | • |
| Fluid retention | • | • |
| Bradycardia | • | • |
| Goiter | • | |
| Carpal tunnel syndrome | • | • |
Which laboratory tests are informative?
Because screening is controversial and symptomatology does not reliably distinguish hypothyroidism from normal pregnancy, laboratory tests are the standard for diagnosis. Overt hypothyroidism is diagnosed in a symptomatic patient by elevated TSH level and low levels of FT4 and free T3 (FT3). Subclinical hypothyroidism is defined as elevated TSH with normal FT4 and FT3 in an asymptomatic patient. Level changes characteristic of normal pregnancy, overt hypothyroidism, and subclinical hypothyroidism are given in TABLE 2.6
TABLE 2
Laboratory diagnosis of hypothyroidism
| MATERNAL CONDITION | TSH | FREE T3 | FREE T4 | TOTAL T3 | TOTAL T4 |
|---|---|---|---|---|---|
| Normal pregnancy | No change | No change | ↑ | ↑ | ↑ |
| Hypothyroidism | ↑ | ↓ | ↓ | ↓ | ↓ |
| Subclinical hypothyroidism | ↑ | No change | No change | ↓ | ↓ |
| Adapted from American College of Obstetricians and Gynecologists6 | |||||
What causes hypothyroidism?
The most common cause of hypothyroidism in most of the world is iodine deficiency. In developed countries, however, where lack of iodine in the diet is not a problem, Hashimoto’s thyroiditis, also known as chronic autoimmune thyroiditis, is the most common cause. Hashimoto’s thyroiditis is characterized by the presence of antithyroid antibodies, including both thyroid antimicrosomial and antithyroglobulin antibodies. Both iodine deficiency and Hashimoto’s thyroiditis are associated with goiter.5 Other causes of hypothyroidism include radioactive iodine therapy for Graves’ disease, a condition we will discuss in Part 2 of this series in February; thyroidectomy; viral thyroiditis; pituitary tumors; Sheehan’s syndrome; and a number of medications.
Causes of hypothyroidism are summarized in TABLE 3.3
TABLE 3
Causes of hypothyroidism
| Iodine deficiency |
| Hashimoto’s thyroiditis |
| Radioactive iodine therapy |
| Thyroidectomy |
| Viral thyroiditis |
| Sheehan’s syndrome |
Medications
|
Effects vary by medication
Medications alter thyroid function in different ways. Iodine and lithium inhibit thyroid function and, along with dopamine antagonists, increase TSH levels. Conversely, thioamides, glucocorticoids, dopamine agonists, and somatostatins decrease TSH levels. Finally, ferrous sulfate, sucrafate, cholestyramine, and aluminum hydroxide antacids all inhibit gastrointestinal absorption of thyroid hormone and therefore should not be taken within 4 hours of thyroid medication.6
Maternal hypothyroidism: Effects on fetus, newborn
The impact of maternal hypothyroidism on the fetus depends on the severity of the condition.
- Uncontrolled hypothyroidism. The consequences of this condition can be dire. The possibilities include intrauterine fetal demise and stillbirth, preterm delivery, low birth weight, preeclampsia, and developmental anomalies including reduced intelligence quotient (IQ).1,2,4,6 Blazer and colleagues correlated intrauterine growth with maternal TSH and fetal FT4 and concluded that impaired intrauterine growth is related to abnormal thyroid function and might reflect an insufficient level of hormone production by hypothyroid mothers during pregnancy.9 Maternal and congenital hypothyroidism resulting from severe iodine deficiency are associated with profound neurologic impairment and mental retardation.1,3,10 If the condition is left untreated, cretinism can occur. Congenital cretinism is associated with growth failure, mental retardation, and other neuropsychologic deficits including deaf-mutism.3,4 However, if cretinism is identified and treated in the first 3 months of life, near-normal growth and intelligence can be expected.6 For this reason, all 50 states and the District of Columbia require newborn screening for congenital hypothyroidism.6
- Asymptomatic overt hypothyroidism. Several studies have evaluated neonatal outcomes in pregnancy complicated by asymptomatic overt hypothyroidism—that is, women who had previously been diagnosed with hypothyroidism, who have abnormal TSH and FT4 levels, but who do not have symptoms. Pop and colleagues have shown impaired psychomotor development at 10 months in infants born to mothers who had low T4 during the first 12 weeks of gestation.7 Haddow and colleagues correlated elevated maternal TSH levels at less than 17 weeks’ gestation with low IQ scores in the offspring at 7 to 9 years of age.8 Klein and colleagues demonstrated an inverse correlation between a woman’s TSH level during pregnancy and the IQ of her offspring.11 Kooistra and colleagues confirmed that maternal hypothyroxinemia is a risk for neurodevelopmental abnormalities that can be identified as early as 3 weeks of age.12 Studies of this relationship are summarized in TABLE 4.
- Subclinical hypothyroidism. During the past decade, researchers have focused attention on neonatal neurologic function in infants born to mothers who had subclinical disease. Mitchell and Klein evaluated the prevalence of subclinical hypothyroidism at less than 17 weeks’ gestation and subsequently compared the IQs in these children with those of controls.4 They found the mean and standard-deviation IQs of the children in the control and treated groups to be significantly higher than those of the children whose mothers were not treated. Casey and colleagues evaluated pregnancy outcomes in women who had undiagnosed subclinical hypothyroidism.10 They found that such pregnancies were more likely to be complicated by placental abruption and preterm birth, and speculated that the reduced IQ demonstrated in the Mitchell and Klein study might have been related to the effects of prematurity.
TABLE 4
Fetal and neonatal effects of asymptomatic overt hypothyroidism
| STUDY | LABORATORY FINDINGS | OUTCOMES AND RECOMMENDATIONS |
|---|---|---|
| Kooistra et al12 | ↓ FT4 | Maternal hypothyroxinemia is a risk for neurodevelopmental abnormalities as early as 3 weeks of age |
| Casey et al10 | ↑ TSH | Pregnancies with undiagnosed subclinical hypothyroidism were more likely to be complicated by placental abruption and preterm birth. The reduced IQ seen in a prior study (Mitchell and Klein4) may be related to effects of prematurity |
| Mitchell and Klein4 | ↑ TSH | The mean and standard deviation of IQs of the children of treated mothers with hypothyroidism and the control group were significantly higher than those for children of untreated hypothyroid women |
| Blazer et al9 | ↑ maternal TSH, ↑ fetal FT4 | Impaired intrauterine growth may reflect insufficient levels of hormone replacement therapy in hypothyroid mothers during pregnancy |
| Pop et al7 | ↓ FT4 | Impaired psychomotor development at 10 months of age in offspring of mothers with low T4 at ≤12 weeks |
| Haddow et al8 | ↑ TSH, ↓ FT4 | Elevated TSH levels at <17 weeks’ gestation are associated with low IQ scores at 7 to 9 years of age. Routine screening for thyroid deficiency may be warranted |
| Klein et al11 | ↑ TSH, ↓ FT4, ↓ TT4 | Inverse correlation between TSH during pregnancy and IQ of offspring |
| FT4=free thyroxine, TSH=thyroid-stimulating hormone, TT4=total thyroxine | ||
Managing hypothyroidism in pregnancy
The treatment of choice for correction of hypothyroidism is synthetic T4, or levothyroxine (Levothyroid, Levoxyl, Synthroid, and Unithroid). Initial treatment in the nonpregnant patient is 1.7 μg/kg/day or 12.5 to 25 μg/day adjusted by 25 μg/day every 2 to 4 weeks until a euthyroid state is achieved.13
Patients who were on thyroxine therapy before pregnancy should increase the dose by 30% once pregnancy is confirmed.1,5 Serum thyrotropin levels should be monitored every 4 weeks to maintain a TSH level between 1 and 2 mU/L and FT4 in upper third of normal.1 Once a euthyroid state has been achieved, thyrotropin levels should be monitored every trimester until delivery. FIGURE 2 provides an algorithm for management of hypothyroidism in pregnancy.
FIGURE 2 During pregnancy, thyroid function merits regular monitoring, fine-tuning of treatment
Postpartum thyroiditis
About 5% of all obstetrical patients develop postpartum thyroiditis. Approximately 45% of these women present with hypothyroidism, with the rest evenly divided between thyrotoxicosis (hyperthyroidism) and thyrotoxicosis followed by hypothyroidism. Unfortunately, the signs and symptoms of hypo- and hyperthyroidism are similar to the postpartum state. Many of these patients are not diagnosed. A high index of suspicion warrants thyroid function testing. Women who have a history of type 1 diabetes mellitus have a 25% chance of developing postpartum thyroid dysfunction.
The diagnosis is made by documenting abnormal levels of TSH and FT4. Postpartum hyperthyroidism may be diagnosed by the presence of antimicrosomal or thyroperoxidase antithyroid peroxidase antibodies. Goiter may be present in up to 50% of patients.
Postpartum thyroiditis has two phases
The first phase, also known as the thyrotoxic phase, occurs 1 to 4 months after delivery when transient thyrotoxicosis develops from excessive release of thyroid hormones. The most common symptoms with early postpartum thyroiditis are fatigue and palpitations. Approximately 67% of these women will return to a euthyroid state, and thioamide therapy is generally considered ineffective. Hypothyroidism can develop within 1 month of the onset of thyroiditis.
The second phase occurs between 4 and 8 months postpartum, and these women present with hypothyroidism. Thyromegaly and associated symptoms are common. Unlike the first (thyrotoxic) phase, medical treatment is recommended. Thyroxine treatment should be initiated and maintained for 6 to 12 months. Postpartum thyroiditis carries a 30% risk of recurrence.14
Postpartum thyroiditis may be associated with depression or aggravate symptoms of depression, although the data on this association are conflicting. The largest study addressing this issue concluded that there was no difference in the clinical and psychiatric signs and symptoms between postpartum thyroiditis and controls.15 Nevertheless, it would seem prudent to evaluate thyroid function in postpartum depression if other signs of thyroid dysfunction are present.
The authors report no financial relationships relevant to this article.
A pregnant woman whose thyroid gland isn’t doing its job presents a serious management problem for her obstetrician. If she has overt hypothyroidism, seen in between 0.3% and 2.5% of pregnancies, active intervention is required to prevent serious damage to the fetus.1,2 Even if she has subclinical disease, seen in 2% to 3% of pregnancies, current research indicates that intervention may be indicated.
Fetal thyroxine requirements increase as early as 5 weeks of gestation, when the fetus is still dependent on maternal thyroxine. A deficiency of maternal thyroxine can have severe adverse outcomes, affecting the course of the pregnancy and the neurologic development of the fetus. To prevent such sequelae, patients who were on thyroid medication before pregnancy should increase the dosage by 30% once pregnancy is confirmed, and hypothyroidism that develops in pregnancy should be managed aggressively and meticulously.
Here, we’ll examine the published research to advise you on evidence-based approaches for diagnosis and management of this complex condition.
Maternal thyroid function
An elaborate negative-feedback loop prevails before pregnancy
In a nonpregnant woman, thyroid function is controlled by a negative-feedback loop that works like this:
- The hypothalamus releases thyroid-releasing hormone (TRH)
- TRH acts on the pituitary gland to release thyroid-stimulating hormone (TSH)
- TSH, in turn, acts on the thyroid gland to release the thyroid hormones iodothyronine (T3) and thyroxine (T4) that regulate metabolism
- TRH and TSH concentrations are inversely related to T3 and T4 concentrations. That is, the more TRH and TSH circulating in the blood stream, the less T3 and T4 will be produced by the thyroid gland3
- Almost all (approximately 99%) circulating T3 and T4 is bound to a protein called thyroxine-binding globulin (TBG). Only 1% of these hormones circulate in the free form, and only the free forms are biologically active.3
This relationship is illustrated in FIGURE 1.
FIGURE 1 Thyroid physiology and the impact of pregnancy
Pregnancy reduces free forms of T3 and T4, and increases TSH slightly
Pregnancy alters thyroid function in significant ways:
- Increases in circulating estrogen lead to the production of more TBG
- When TBG increases, more T3 and T4 are bound and fewer free forms of these hormones are available
- Because the total T3 (TT3) and total free T4 (TT4) are decreased in pregnancy, they are not good measures of thyroid function. Maternal thyroid function in pregnancy should be monitored using free T4 (FT4) and TSH levels
- Increased TBG also leads to a slight increase in TSH between the first trimester and term
- Human chorionic gonadotropin (hCG) concentrations also increase in pregnancy. Because hCG has thyrotropin-like activity, these higher levels cause a transient decrease in TSH by suppression of TSH production between approximately 8 and 14 weeks of gestation.
Fetal thyroid function
During early gestation, the fetus receives thyroid hormone from the mother.1 Maternal T4 crosses the placenta actively—the only hormone that does so.4 The fetus’s need for thyroxine starts to increase as early as 5 weeks of gestation.5
Fetal thyroid development does not begin until 10 to 12 weeks of gestation, and then continues until term. The fetus relies on maternal T4 exclusively before 12 weeks and partially thereafter for normal fetal neurologic development. It follows that maternal hypothyroidism could be detrimental to fetal development if not detected and corrected very early in gestation.
How (and whom) to screen for maternal hypothyroidism
Routine screening has been recommended for women who have infertility, menstrual disorders, or type 1 diabetes mellitus, and for pregnant women who have signs and symptoms of deficient thyroid function.6 In recent years, some authors have recommended screening all pregnant women for thyroid dysfunction, but such recommendations remain controversial.3,7,8 Routine screening is not endorsed by the American College of Obstetricians and Gynecologists.6
Symptoms overlap typical conditions of pregnancy
The difficulty here is that the characteristic signs and symptoms of hypothyroidism are very similar to physiologic conditions seen in most pregnancies. They include fatigue, constipation, cold intolerance, muscle cramps, hair loss, dry skin, brittle nails, weight gain, intellectual slowness, bradycardia, depression, insomnia, periorbital edema, myxedema, and myxedema coma.6 A side-by-side comparison of pregnancy conditions and hypothyroidism symptoms is provided in TABLE 1.
TABLE 1
Distinguishing hypothyroidism from a normal gestation can be challenging
| SYMPTOM | HYPOTHYROIDISM | PREGNANCY |
|---|---|---|
| Fatigue | • | • |
| Constipation | • | • |
| Hair loss | • | |
| Dry skin | • | |
| Brittle nails | • | |
| Weight gain | • | • |
| Fluid retention | • | • |
| Bradycardia | • | • |
| Goiter | • | |
| Carpal tunnel syndrome | • | • |
Which laboratory tests are informative?
Because screening is controversial and symptomatology does not reliably distinguish hypothyroidism from normal pregnancy, laboratory tests are the standard for diagnosis. Overt hypothyroidism is diagnosed in a symptomatic patient by elevated TSH level and low levels of FT4 and free T3 (FT3). Subclinical hypothyroidism is defined as elevated TSH with normal FT4 and FT3 in an asymptomatic patient. Level changes characteristic of normal pregnancy, overt hypothyroidism, and subclinical hypothyroidism are given in TABLE 2.6
TABLE 2
Laboratory diagnosis of hypothyroidism
| MATERNAL CONDITION | TSH | FREE T3 | FREE T4 | TOTAL T3 | TOTAL T4 |
|---|---|---|---|---|---|
| Normal pregnancy | No change | No change | ↑ | ↑ | ↑ |
| Hypothyroidism | ↑ | ↓ | ↓ | ↓ | ↓ |
| Subclinical hypothyroidism | ↑ | No change | No change | ↓ | ↓ |
| Adapted from American College of Obstetricians and Gynecologists6 | |||||
What causes hypothyroidism?
The most common cause of hypothyroidism in most of the world is iodine deficiency. In developed countries, however, where lack of iodine in the diet is not a problem, Hashimoto’s thyroiditis, also known as chronic autoimmune thyroiditis, is the most common cause. Hashimoto’s thyroiditis is characterized by the presence of antithyroid antibodies, including both thyroid antimicrosomial and antithyroglobulin antibodies. Both iodine deficiency and Hashimoto’s thyroiditis are associated with goiter.5 Other causes of hypothyroidism include radioactive iodine therapy for Graves’ disease, a condition we will discuss in Part 2 of this series in February; thyroidectomy; viral thyroiditis; pituitary tumors; Sheehan’s syndrome; and a number of medications.
Causes of hypothyroidism are summarized in TABLE 3.3
TABLE 3
Causes of hypothyroidism
| Iodine deficiency |
| Hashimoto’s thyroiditis |
| Radioactive iodine therapy |
| Thyroidectomy |
| Viral thyroiditis |
| Sheehan’s syndrome |
Medications
|
Effects vary by medication
Medications alter thyroid function in different ways. Iodine and lithium inhibit thyroid function and, along with dopamine antagonists, increase TSH levels. Conversely, thioamides, glucocorticoids, dopamine agonists, and somatostatins decrease TSH levels. Finally, ferrous sulfate, sucrafate, cholestyramine, and aluminum hydroxide antacids all inhibit gastrointestinal absorption of thyroid hormone and therefore should not be taken within 4 hours of thyroid medication.6
Maternal hypothyroidism: Effects on fetus, newborn
The impact of maternal hypothyroidism on the fetus depends on the severity of the condition.
- Uncontrolled hypothyroidism. The consequences of this condition can be dire. The possibilities include intrauterine fetal demise and stillbirth, preterm delivery, low birth weight, preeclampsia, and developmental anomalies including reduced intelligence quotient (IQ).1,2,4,6 Blazer and colleagues correlated intrauterine growth with maternal TSH and fetal FT4 and concluded that impaired intrauterine growth is related to abnormal thyroid function and might reflect an insufficient level of hormone production by hypothyroid mothers during pregnancy.9 Maternal and congenital hypothyroidism resulting from severe iodine deficiency are associated with profound neurologic impairment and mental retardation.1,3,10 If the condition is left untreated, cretinism can occur. Congenital cretinism is associated with growth failure, mental retardation, and other neuropsychologic deficits including deaf-mutism.3,4 However, if cretinism is identified and treated in the first 3 months of life, near-normal growth and intelligence can be expected.6 For this reason, all 50 states and the District of Columbia require newborn screening for congenital hypothyroidism.6
- Asymptomatic overt hypothyroidism. Several studies have evaluated neonatal outcomes in pregnancy complicated by asymptomatic overt hypothyroidism—that is, women who had previously been diagnosed with hypothyroidism, who have abnormal TSH and FT4 levels, but who do not have symptoms. Pop and colleagues have shown impaired psychomotor development at 10 months in infants born to mothers who had low T4 during the first 12 weeks of gestation.7 Haddow and colleagues correlated elevated maternal TSH levels at less than 17 weeks’ gestation with low IQ scores in the offspring at 7 to 9 years of age.8 Klein and colleagues demonstrated an inverse correlation between a woman’s TSH level during pregnancy and the IQ of her offspring.11 Kooistra and colleagues confirmed that maternal hypothyroxinemia is a risk for neurodevelopmental abnormalities that can be identified as early as 3 weeks of age.12 Studies of this relationship are summarized in TABLE 4.
- Subclinical hypothyroidism. During the past decade, researchers have focused attention on neonatal neurologic function in infants born to mothers who had subclinical disease. Mitchell and Klein evaluated the prevalence of subclinical hypothyroidism at less than 17 weeks’ gestation and subsequently compared the IQs in these children with those of controls.4 They found the mean and standard-deviation IQs of the children in the control and treated groups to be significantly higher than those of the children whose mothers were not treated. Casey and colleagues evaluated pregnancy outcomes in women who had undiagnosed subclinical hypothyroidism.10 They found that such pregnancies were more likely to be complicated by placental abruption and preterm birth, and speculated that the reduced IQ demonstrated in the Mitchell and Klein study might have been related to the effects of prematurity.
TABLE 4
Fetal and neonatal effects of asymptomatic overt hypothyroidism
| STUDY | LABORATORY FINDINGS | OUTCOMES AND RECOMMENDATIONS |
|---|---|---|
| Kooistra et al12 | ↓ FT4 | Maternal hypothyroxinemia is a risk for neurodevelopmental abnormalities as early as 3 weeks of age |
| Casey et al10 | ↑ TSH | Pregnancies with undiagnosed subclinical hypothyroidism were more likely to be complicated by placental abruption and preterm birth. The reduced IQ seen in a prior study (Mitchell and Klein4) may be related to effects of prematurity |
| Mitchell and Klein4 | ↑ TSH | The mean and standard deviation of IQs of the children of treated mothers with hypothyroidism and the control group were significantly higher than those for children of untreated hypothyroid women |
| Blazer et al9 | ↑ maternal TSH, ↑ fetal FT4 | Impaired intrauterine growth may reflect insufficient levels of hormone replacement therapy in hypothyroid mothers during pregnancy |
| Pop et al7 | ↓ FT4 | Impaired psychomotor development at 10 months of age in offspring of mothers with low T4 at ≤12 weeks |
| Haddow et al8 | ↑ TSH, ↓ FT4 | Elevated TSH levels at <17 weeks’ gestation are associated with low IQ scores at 7 to 9 years of age. Routine screening for thyroid deficiency may be warranted |
| Klein et al11 | ↑ TSH, ↓ FT4, ↓ TT4 | Inverse correlation between TSH during pregnancy and IQ of offspring |
| FT4=free thyroxine, TSH=thyroid-stimulating hormone, TT4=total thyroxine | ||
Managing hypothyroidism in pregnancy
The treatment of choice for correction of hypothyroidism is synthetic T4, or levothyroxine (Levothyroid, Levoxyl, Synthroid, and Unithroid). Initial treatment in the nonpregnant patient is 1.7 μg/kg/day or 12.5 to 25 μg/day adjusted by 25 μg/day every 2 to 4 weeks until a euthyroid state is achieved.13
Patients who were on thyroxine therapy before pregnancy should increase the dose by 30% once pregnancy is confirmed.1,5 Serum thyrotropin levels should be monitored every 4 weeks to maintain a TSH level between 1 and 2 mU/L and FT4 in upper third of normal.1 Once a euthyroid state has been achieved, thyrotropin levels should be monitored every trimester until delivery. FIGURE 2 provides an algorithm for management of hypothyroidism in pregnancy.
FIGURE 2 During pregnancy, thyroid function merits regular monitoring, fine-tuning of treatment
Postpartum thyroiditis
About 5% of all obstetrical patients develop postpartum thyroiditis. Approximately 45% of these women present with hypothyroidism, with the rest evenly divided between thyrotoxicosis (hyperthyroidism) and thyrotoxicosis followed by hypothyroidism. Unfortunately, the signs and symptoms of hypo- and hyperthyroidism are similar to the postpartum state. Many of these patients are not diagnosed. A high index of suspicion warrants thyroid function testing. Women who have a history of type 1 diabetes mellitus have a 25% chance of developing postpartum thyroid dysfunction.
The diagnosis is made by documenting abnormal levels of TSH and FT4. Postpartum hyperthyroidism may be diagnosed by the presence of antimicrosomal or thyroperoxidase antithyroid peroxidase antibodies. Goiter may be present in up to 50% of patients.
Postpartum thyroiditis has two phases
The first phase, also known as the thyrotoxic phase, occurs 1 to 4 months after delivery when transient thyrotoxicosis develops from excessive release of thyroid hormones. The most common symptoms with early postpartum thyroiditis are fatigue and palpitations. Approximately 67% of these women will return to a euthyroid state, and thioamide therapy is generally considered ineffective. Hypothyroidism can develop within 1 month of the onset of thyroiditis.
The second phase occurs between 4 and 8 months postpartum, and these women present with hypothyroidism. Thyromegaly and associated symptoms are common. Unlike the first (thyrotoxic) phase, medical treatment is recommended. Thyroxine treatment should be initiated and maintained for 6 to 12 months. Postpartum thyroiditis carries a 30% risk of recurrence.14
Postpartum thyroiditis may be associated with depression or aggravate symptoms of depression, although the data on this association are conflicting. The largest study addressing this issue concluded that there was no difference in the clinical and psychiatric signs and symptoms between postpartum thyroiditis and controls.15 Nevertheless, it would seem prudent to evaluate thyroid function in postpartum depression if other signs of thyroid dysfunction are present.
1. Idris I, Srinivasan R, Simm A, Page RC. Effects of maternal hyperthyroidism during early gestation on neonatal and obstetric outcome. Clin Endocrinol. 2006;65:133-135.
2. Girling JC. Thyroid disorders in pregnancy. Curr Obstet Gynecol. 2006;16:47-53.
3. Creasy RK, Resnik R, Iams J. Maternal–Fetal Medicine. 5th ed. Philadelphia, Pa: Saunders Elsevier; 2004:1063-1082.
4. Mitchell ML, Klein RZ. The sequelae of untreated maternal hypothyroidism. Eur J Endocrinol. 2004;151 Suppl 3:U45-U48.
5. Alexander EK, Marqusee E, Lawrence J, Jarolim P, Fischer GA, Larsen PR. Timing and magnitude of increases in levothyroxine requirements during pregnancy in women with hypothyroidism. N Engl J Med. 2004;351:241-249.
6. American College of Obstetrics and Gynecology. ACOG Practice Bulletin. Clinical management guidelines for obstetrician-gynecologists. Number 37, August 2002. (Replaces Practice Bulletin Number 32, November 2001). Thyroid disease in pregnancy. Obstet Gynecol. 2002;100:387-396.
7. Pop VJ, Kuijpens JL, van Baar AL, et al. Low maternal free thyroxine concentrations during early pregnancy are associated with impaired psychomotor development in infancy. Clin Endocrinol. 1999;50:149-155.
8. Haddow JE, Palomaki GE, Allan WC, et al. Maternal thyroid deficiency during pregnancy and subsequent neuropsychological development of the child. N Engl J Med. 1999;341:549-555.
9. Blazer S, Moreh-Waterman Y, Miller-Lotan R, Tamir A, Hochberg Z. Maternal hypothyroidism may affect fetal growth and neonatal thyroid function. Obstet Gynecol. 2003;102:232-241.
10. Casey BM, Dashe JS, Wells CE, et al. Subclinical hypothyroidism and pregnancy outcomes. Obstet Gynecol. 2005;105:239-245.
11. Klein RZ, Haddow JE, Faix JD, et al. Prevalence of thyroid deficiency in pregnant women. Clin Endocrinol. 1991;35:41-46.
12. Kooistra L, Crawford S, van Baar AL, Brouwers EP, Pop VJ. Neonatal effects of maternal hypothyroxinemia during early pregnancy. Pediatrics. 2006;117:161-167.
13. Levothyroxine: Drug information. Lexicomp. http://www.utdol.com/utd/content/topic.do?topicKey=drug_l_z/143814&type=A&selectedTitle=2~39. Accessed December 14, 2007.
14. Casey BM, Leveno KJ. Thyroid disease in pregnancy. Obstet Gynecol. 2006;108:1283-1292.
15. Kent GN, Stuckey BG, Allen JR, Lambert T, Gee V. Postpartum thyroid dysfunction: clinical assessment and relationship to psychiatric affective morbidity. Clin Endocrinol. 1999;51:429-438.
1. Idris I, Srinivasan R, Simm A, Page RC. Effects of maternal hyperthyroidism during early gestation on neonatal and obstetric outcome. Clin Endocrinol. 2006;65:133-135.
2. Girling JC. Thyroid disorders in pregnancy. Curr Obstet Gynecol. 2006;16:47-53.
3. Creasy RK, Resnik R, Iams J. Maternal–Fetal Medicine. 5th ed. Philadelphia, Pa: Saunders Elsevier; 2004:1063-1082.
4. Mitchell ML, Klein RZ. The sequelae of untreated maternal hypothyroidism. Eur J Endocrinol. 2004;151 Suppl 3:U45-U48.
5. Alexander EK, Marqusee E, Lawrence J, Jarolim P, Fischer GA, Larsen PR. Timing and magnitude of increases in levothyroxine requirements during pregnancy in women with hypothyroidism. N Engl J Med. 2004;351:241-249.
6. American College of Obstetrics and Gynecology. ACOG Practice Bulletin. Clinical management guidelines for obstetrician-gynecologists. Number 37, August 2002. (Replaces Practice Bulletin Number 32, November 2001). Thyroid disease in pregnancy. Obstet Gynecol. 2002;100:387-396.
7. Pop VJ, Kuijpens JL, van Baar AL, et al. Low maternal free thyroxine concentrations during early pregnancy are associated with impaired psychomotor development in infancy. Clin Endocrinol. 1999;50:149-155.
8. Haddow JE, Palomaki GE, Allan WC, et al. Maternal thyroid deficiency during pregnancy and subsequent neuropsychological development of the child. N Engl J Med. 1999;341:549-555.
9. Blazer S, Moreh-Waterman Y, Miller-Lotan R, Tamir A, Hochberg Z. Maternal hypothyroidism may affect fetal growth and neonatal thyroid function. Obstet Gynecol. 2003;102:232-241.
10. Casey BM, Dashe JS, Wells CE, et al. Subclinical hypothyroidism and pregnancy outcomes. Obstet Gynecol. 2005;105:239-245.
11. Klein RZ, Haddow JE, Faix JD, et al. Prevalence of thyroid deficiency in pregnant women. Clin Endocrinol. 1991;35:41-46.
12. Kooistra L, Crawford S, van Baar AL, Brouwers EP, Pop VJ. Neonatal effects of maternal hypothyroxinemia during early pregnancy. Pediatrics. 2006;117:161-167.
13. Levothyroxine: Drug information. Lexicomp. http://www.utdol.com/utd/content/topic.do?topicKey=drug_l_z/143814&type=A&selectedTitle=2~39. Accessed December 14, 2007.
14. Casey BM, Leveno KJ. Thyroid disease in pregnancy. Obstet Gynecol. 2006;108:1283-1292.
15. Kent GN, Stuckey BG, Allen JR, Lambert T, Gee V. Postpartum thyroid dysfunction: clinical assessment and relationship to psychiatric affective morbidity. Clin Endocrinol. 1999;51:429-438.