User login
Potential Tyrosine Kinase Inhibitor Therapy Discontinuation for Patients With Chronic Myeloid Leukemia in a VA Regional Network
Potential Tyrosine Kinase Inhibitor Therapy Discontinuation for Patients With Chronic Myeloid Leukemia in a VA Regional Network
Chronic myeloid leukemia (CML) is a hematologic malignancy resulting from an acquired mutation. The mutation results in a reciprocal translocation between the long arms of chromosomes 9 and 22 and is known as the Philadelphia chromosome (Ph), or Ph-positive (Ph+) when present. The translocation results in the formation of a BCR-ABL fusion oncogene, which leads to continuous cell cycling and proliferation, altered differentiation, and a loss of apoptosis.1,2
Until the 1980s, CML was considered fatal.3 The mainstay of treatment consisted of 2 oral chemotherapeutic agents, busulfan and hydroxyurea. These medications did not prevent blast crisis, a fatal form of leukemia.4,5 The introduction of tyrosine kinase inhibitors (TKIs) transformed CML management and improved 10-year overall survival from about 20% to > 80% by delaying the transition to blast crisis. Now, the risk of death from general health conditions or comorbidities is higher than that of CML.6
TKIs target the root cause of CML through inhibition of the BCR-ABL oncoprotein.1,2 For CML, the goals of treatment include maintaining hematologic, cytogenetic, and molecular remission; preventing progression to accelerated phase or blast crisis; minimizing toxicity; and enabling potential cessation of therapy in carefully selected patients.7,8
Small cohort studies suggest that dose reduction of TKIs in patients who achieve optimal responses may reduce the risk of long-term adverse effects (AEs). However, optimal dose-reduction and minimum effective dose of each agent are unknown.7 The ability to maintain undetectable minimal residual disease or disease detectable at a stable low level after TKI discontinuation has been called treatment-free remission. Studies suggest that about 40% to 50% of patients who have achieved a stable deep molecular response remain in treatment-free remission after stopping first-line treatment.9,10 Of the patients who relapse following TKI discontinuation, 80% relapse within the first 6 months of treatment cessation. Molecular response is regained in almost all patients when treatment is resumed with the same TKI.11
The National Comprehensive Cancer Network (NCCN) recommends considering discontinuation of TKI therapy only outside the setting of a clinical trial and only in patients who consent to discontinuation after a thorough discussion of the potential risks and benefits. The NCCN criteria for patients who may be eligible for discontinuation are listed in Table 1. The Life After Stopping TKIs study reported that 80% of patients with well-controlled chronic phase CML who discontinued TKIs had a clinically meaningful improvement in fatigue. Patients also reported clinically meaningful improvements in depression, diarrhea, sleep disturbance, and pain interference. These symptoms worsened after restarting TKI therapy.12

TKI DISCONTINUATION
Electronic health record data were extracted using structured query language from the US Department of Veterans Affairs (VA) Corporate Data Warehouse (CDW). To be eligible for discontinuation, veterans had to be aged > 18 years, receive oncology care within a Veterans Integrated Services Network (VISN) 21 health care system (HCS) (VA Sierra Nevada HCS, VA Southern Nevada HCS, VA Central California HCS, VA Palo Alto HCS, VA Northern California HCS, and VA San Francisco HCS) or be a veteran referred to a community-based oncology practitioner. Patients had to have a documented diagnosis of chronic phase CML, have an active order for a TKI, be on TKI therapy for ≥ 3 years, and have a stable molecular response (BCR-ABL1 ≤ 0.01% on the International Scale for ≥ 2 years with ≥ 4 tests done ≥ 3 months apart) as of October 1, 2024. Veterans were excluded if they had a history of advanced accelerated phase CML, previous TKI discontinuation trials, nonadherence to the TKI, or if they did not want to consider TKI discontinuation.
This analysis evaluated the potential cost avoidance associated with TKI discontinuation. Cost avoidance was calculated using the average wholesale price of each TKI. Secondary objectives evaluated health outcomes of TKI discontinuation including CML relapse, reported AEs, long-term remission, and TKI withdrawal syndrome. Health outcomes were determined through chart review of AEs and clinic notes documented in the electronic health record during the study time frame.
Baseline information for eligible patients was collected, including age, sex, and race, and chart reviews were completed to evaluate reported AEs associated with therapy. Oncology clinical pharmacy practitioners (CPPs) at each VISN 21 facility were notified of eligible patients to facilitate discussion with oncologists and establish monitoring if therapy was discontinued. Following TKI discontinuation, health outcomes were evaluated, including CML relapse, changes in reported AEs, long-term remission, and TKI withdrawal syndrome. Descriptive statistics were used to analyze the baseline characteristics. Cost avoidance was calculated using the average wholesale price for each TKI. The number of tablets required to reach each patient’s individual dose was taken into consideration when determining the cost avoidance. A dashboard was created using the query from the CDW and was developed in Microsoft Power BI.
Preliminary Results
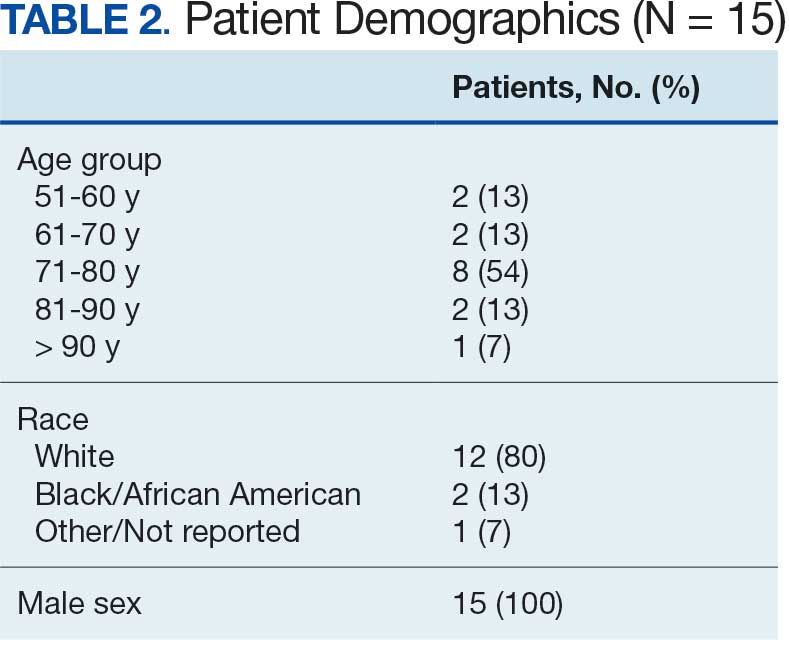
In FY 2024, VISN 21 had 3819 oncology patients. Twenty-four patients had taken a TKI for ≥ 3 years, 20 had a stable molecular response, and 15 had not previously attempted to discontinue their TKI (Figure 1). Fifteen veterans were eligible for therapy discontinuation for a total potential annual cost avoidance of $1.2 million (Figure 2). Most of the cost avoidance, $935,057 (78%), was attributed to 3 patients on nilotinib. The mean age of the population was 74 years. All patients were male, and 12 (80%) were White. (Table 2). At baseline, 11 patients (73%) were taking imatinib. One patient received oncology care from a community care clinician. All 15 patients decided to remain on therapy.
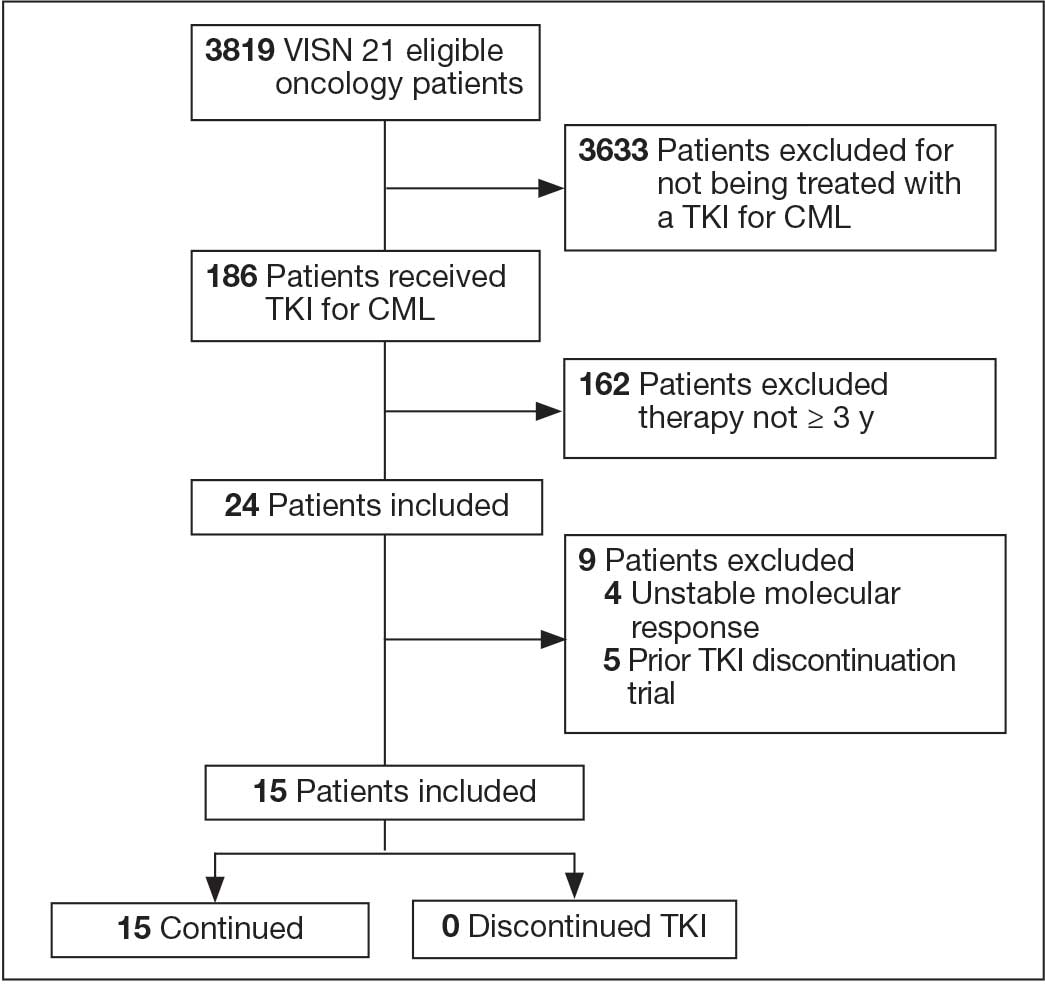
Abbreviations: CML, chronic myeloid leukemia; TKI, tyrosine kinase inhibitor;
VISN, Veterans Integrated Service Network.
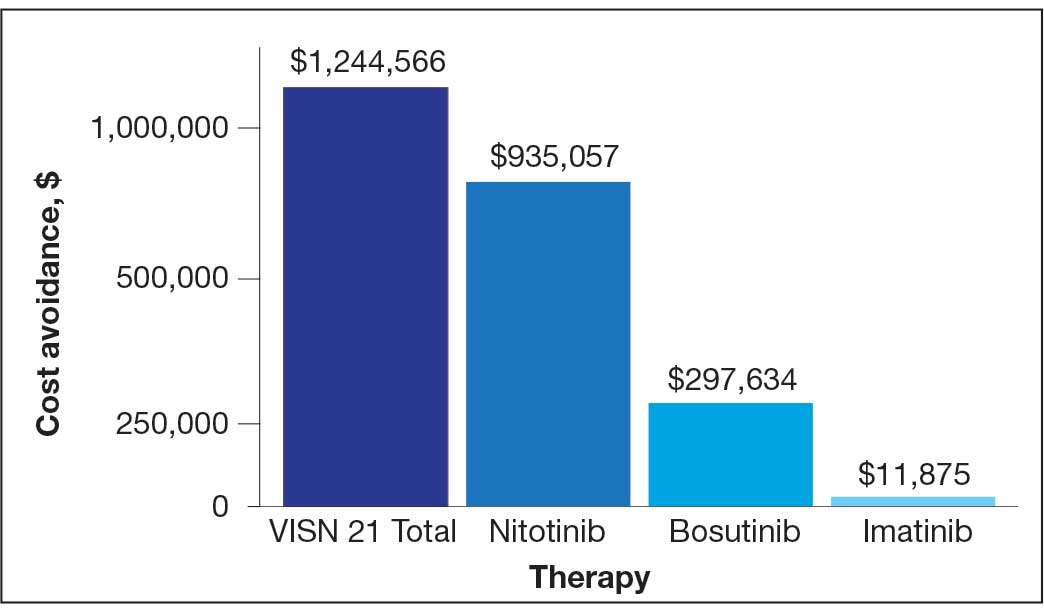
for 15 patients at Veterans Integrated Services Network 21.
DISCUSSION
As a multisite quality improvement initiative, this project raised awareness of TKI therapy discontinuation in select patients with CML. It also sparked collaboration among oncology CPPs and clinicians and stimulated conversations about CML treatment. The development of the TKI discontinuation dashboard provides a population health management tool for CPPs and clinicians to identify eligible patients in the future.
Adherence to TKIs is crucial for disease control and survival in patients with CML. Patients are counseled that poor adherence to therapy may contribute to worsening disease or suboptimal response, the development of resistance, and greater health care costs.13 Therefore, it was a challenge for patients to understand and accept that they could stop TKI therapy after achieving a stable deep molecular response. Discussions with patients about the goal of therapy—suppressing the BCR-ABL oncogene, which they have achieved—could encourage patients to trial therapy discontinuation.
Only small cohort studies have been completed to evaluate the outcomes of therapy discontinuation. Much remains unknown regarding the optimal dose-reduction strategy and the minimum effective dose of each agent. Additionally, understanding the qualities of a good candidate for TKI discontinuation remains a barrier. A similar project was conducted in VISN 17. Five patients were counseled on TKI discontinuation; however, only 1 discontinued TKI therapy. Unfortunately, soon after discontinuing treatment, the patient had to restart therapy. Additional literature will enhance understanding of therapy discontinuation.
An unexpected finding of TKI discontinuation trials has been a reversible phenomenon known as TKI withdrawal syndrome.9 It can occur regardless of the TKI used and results in pruritus and new or worsening musculoskeletal pain within several weeks of TKI discontinuation in about 30% of patients. Symptoms may last several months and may require acetaminophen or nonsteroidal anti-inflammatory drugs for pain control.9,10,14
The potential cost avoidance of $1.2 million is an underestimation because VA contracts allow for greater cost savings. However, that information is confidential and therefore average wholesale price had to be used for this project. Most of the cost avoidance was due to 4 patients who could not tolerate imatinib and used nilotinib, which is more expensive.
Limitations
The small sample size presented some limitations. Of the 3819 oncology patients within VISN 21 in FY 2024, 186 received a TKI and only 15 were eligible for discontinuation. Additionally, challenges emerged when discussing discontinuation with community care clinicians and patients. Community care clinicians were difficult to contact, making it challenging to discuss the project with them. CPPs noted hesitancy among VA clinicians and patients to discontinue a medication for which adherence was continually emphasized.
Conclusions
Discussions about CML TKI discontinuation led to collaboration with the oncology care team and could lead to significant cost avoidance. Barriers to TKI discontinuation included patients’ concern for relapse, risk of discontinuation syndrome, the requirement for close monitoring, and clinician buy-in. Outcome studies are needed to gain a greater understanding of the benefits and risks of therapy discontinuation. In the future, evaluation of possible clinical and biological predictors of successful TKI discontinuation may be beneficial.
- Schiffer CA. BCR-ABL tyrosine kinase inhibitors for chronic myelogenous leukemia. N Engl J Med. 2007;357:258-265. doi:10.1056/NEJMct071828
- Hehlmann R, Hochhaus A, Baccarani M; European LeukemiaNet. Chronic myeloid leukaemia. Lancet. 2007;370:342-350. doi:10.1016/S0140-6736(07)61165-9
- Goldman JM, Melo JV. Chronic myeloid leukemia--advances in biology and new approaches to treatment. N Engl J Med. 2003;349:1451-1464. doi:10.1056/NEJMra020777
- Pasic I, Lipton JH. Current approach to the treatment of chronic myeloid leukaemia. Leuk Res. 2017;55:65-78. doi:10.1016/j.leukres.2017.01.005
- Rao KV, Iannucci A, Jabbour E. Current and future clinical strategies in the management of chronic myeloid leukemia. Pharmacotherapy. 2010;30:77S-101S. doi:10.1592/phco.30.pt2.77S
- Cortes J, Pavlovsky C, Saußele S. Chronic myeloid leukaemia. Lancet. 2021;398:1914-1926. doi:10.1016/S0140-6736(21)01204-6
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Chronic myeloid leukemia. Version 1.2026. July 16, 2025. Accessed February 8, 2026. https://www.nccn.org /guidelines/guidelines-detail?id=1427
- Hochhaus A, Baccarani M, Silver RT, et al. European LeukemiaNet 2020 recommendations for treating chronic myeloid leukemia. Leukemia. 2020;34:966-984. doi:10.1038/s41375-020-0776-2
- Saußele S, Richter J, Hochhaus A, Mahon F-X. The concept of treatment-free remission in chronic myeloid leukemia. Leukemia. 2016;30:1638-1647. doi:10.1038/leu.2016.115
- Atallah E, Sweet K. Treatment-free remission: the new goal in CML therapy. Curr Hematol Malig Rep. 2021;16:433-439. doi:10.1007/s11899-021-00653-1
- Hehlmann R. The new ELN recommendations for treating CML. J Clin Med. 2020;9:3671. doi:10.3390/jcm9113671
- Atallah E, Schiffer CA, Radich JP , et al. Assessment of outcomes after stopping tyrosine kinase inhibitors among patients with chronic myeloid leukemia: a non-randomized clinical trial. JAMA Oncol. 2021;7:42-50. doi:10.1001/jamaoncol.2020.5774
- Breccia M, Efficace F, Alimena G. Imatinib treatment in chronic myelogenous leukemia: what have we learned so far? Cancer Lett. 2011;300:115-121. doi:10.1016/j.canlet.2010.10.018
- Berman E. How I treat chronic-phase chronic myelogenous leukemia. Blood. 2022;139:3138-3147. doi:10.1182/blood.2021011722
Chronic myeloid leukemia (CML) is a hematologic malignancy resulting from an acquired mutation. The mutation results in a reciprocal translocation between the long arms of chromosomes 9 and 22 and is known as the Philadelphia chromosome (Ph), or Ph-positive (Ph+) when present. The translocation results in the formation of a BCR-ABL fusion oncogene, which leads to continuous cell cycling and proliferation, altered differentiation, and a loss of apoptosis.1,2
Until the 1980s, CML was considered fatal.3 The mainstay of treatment consisted of 2 oral chemotherapeutic agents, busulfan and hydroxyurea. These medications did not prevent blast crisis, a fatal form of leukemia.4,5 The introduction of tyrosine kinase inhibitors (TKIs) transformed CML management and improved 10-year overall survival from about 20% to > 80% by delaying the transition to blast crisis. Now, the risk of death from general health conditions or comorbidities is higher than that of CML.6
TKIs target the root cause of CML through inhibition of the BCR-ABL oncoprotein.1,2 For CML, the goals of treatment include maintaining hematologic, cytogenetic, and molecular remission; preventing progression to accelerated phase or blast crisis; minimizing toxicity; and enabling potential cessation of therapy in carefully selected patients.7,8
Small cohort studies suggest that dose reduction of TKIs in patients who achieve optimal responses may reduce the risk of long-term adverse effects (AEs). However, optimal dose-reduction and minimum effective dose of each agent are unknown.7 The ability to maintain undetectable minimal residual disease or disease detectable at a stable low level after TKI discontinuation has been called treatment-free remission. Studies suggest that about 40% to 50% of patients who have achieved a stable deep molecular response remain in treatment-free remission after stopping first-line treatment.9,10 Of the patients who relapse following TKI discontinuation, 80% relapse within the first 6 months of treatment cessation. Molecular response is regained in almost all patients when treatment is resumed with the same TKI.11
The National Comprehensive Cancer Network (NCCN) recommends considering discontinuation of TKI therapy only outside the setting of a clinical trial and only in patients who consent to discontinuation after a thorough discussion of the potential risks and benefits. The NCCN criteria for patients who may be eligible for discontinuation are listed in Table 1. The Life After Stopping TKIs study reported that 80% of patients with well-controlled chronic phase CML who discontinued TKIs had a clinically meaningful improvement in fatigue. Patients also reported clinically meaningful improvements in depression, diarrhea, sleep disturbance, and pain interference. These symptoms worsened after restarting TKI therapy.12

TKI DISCONTINUATION
Electronic health record data were extracted using structured query language from the US Department of Veterans Affairs (VA) Corporate Data Warehouse (CDW). To be eligible for discontinuation, veterans had to be aged > 18 years, receive oncology care within a Veterans Integrated Services Network (VISN) 21 health care system (HCS) (VA Sierra Nevada HCS, VA Southern Nevada HCS, VA Central California HCS, VA Palo Alto HCS, VA Northern California HCS, and VA San Francisco HCS) or be a veteran referred to a community-based oncology practitioner. Patients had to have a documented diagnosis of chronic phase CML, have an active order for a TKI, be on TKI therapy for ≥ 3 years, and have a stable molecular response (BCR-ABL1 ≤ 0.01% on the International Scale for ≥ 2 years with ≥ 4 tests done ≥ 3 months apart) as of October 1, 2024. Veterans were excluded if they had a history of advanced accelerated phase CML, previous TKI discontinuation trials, nonadherence to the TKI, or if they did not want to consider TKI discontinuation.
This analysis evaluated the potential cost avoidance associated with TKI discontinuation. Cost avoidance was calculated using the average wholesale price of each TKI. Secondary objectives evaluated health outcomes of TKI discontinuation including CML relapse, reported AEs, long-term remission, and TKI withdrawal syndrome. Health outcomes were determined through chart review of AEs and clinic notes documented in the electronic health record during the study time frame.
Baseline information for eligible patients was collected, including age, sex, and race, and chart reviews were completed to evaluate reported AEs associated with therapy. Oncology clinical pharmacy practitioners (CPPs) at each VISN 21 facility were notified of eligible patients to facilitate discussion with oncologists and establish monitoring if therapy was discontinued. Following TKI discontinuation, health outcomes were evaluated, including CML relapse, changes in reported AEs, long-term remission, and TKI withdrawal syndrome. Descriptive statistics were used to analyze the baseline characteristics. Cost avoidance was calculated using the average wholesale price for each TKI. The number of tablets required to reach each patient’s individual dose was taken into consideration when determining the cost avoidance. A dashboard was created using the query from the CDW and was developed in Microsoft Power BI.
Preliminary Results
In FY 2024, VISN 21 had 3819 oncology patients. Twenty-four patients had taken a TKI for ≥ 3 years, 20 had a stable molecular response, and 15 had not previously attempted to discontinue their TKI (Figure 1). Fifteen veterans were eligible for therapy discontinuation for a total potential annual cost avoidance of $1.2 million (Figure 2). Most of the cost avoidance, $935,057 (78%), was attributed to 3 patients on nilotinib. The mean age of the population was 74 years. All patients were male, and 12 (80%) were White. (Table 2). At baseline, 11 patients (73%) were taking imatinib. One patient received oncology care from a community care clinician. All 15 patients decided to remain on therapy.

Abbreviations: CML, chronic myeloid leukemia; TKI, tyrosine kinase inhibitor;
VISN, Veterans Integrated Service Network.

for 15 patients at Veterans Integrated Services Network 21.

DISCUSSION
As a multisite quality improvement initiative, this project raised awareness of TKI therapy discontinuation in select patients with CML. It also sparked collaboration among oncology CPPs and clinicians and stimulated conversations about CML treatment. The development of the TKI discontinuation dashboard provides a population health management tool for CPPs and clinicians to identify eligible patients in the future.
Adherence to TKIs is crucial for disease control and survival in patients with CML. Patients are counseled that poor adherence to therapy may contribute to worsening disease or suboptimal response, the development of resistance, and greater health care costs.13 Therefore, it was a challenge for patients to understand and accept that they could stop TKI therapy after achieving a stable deep molecular response. Discussions with patients about the goal of therapy—suppressing the BCR-ABL oncogene, which they have achieved—could encourage patients to trial therapy discontinuation.
Only small cohort studies have been completed to evaluate the outcomes of therapy discontinuation. Much remains unknown regarding the optimal dose-reduction strategy and the minimum effective dose of each agent. Additionally, understanding the qualities of a good candidate for TKI discontinuation remains a barrier. A similar project was conducted in VISN 17. Five patients were counseled on TKI discontinuation; however, only 1 discontinued TKI therapy. Unfortunately, soon after discontinuing treatment, the patient had to restart therapy. Additional literature will enhance understanding of therapy discontinuation.
An unexpected finding of TKI discontinuation trials has been a reversible phenomenon known as TKI withdrawal syndrome.9 It can occur regardless of the TKI used and results in pruritus and new or worsening musculoskeletal pain within several weeks of TKI discontinuation in about 30% of patients. Symptoms may last several months and may require acetaminophen or nonsteroidal anti-inflammatory drugs for pain control.9,10,14
The potential cost avoidance of $1.2 million is an underestimation because VA contracts allow for greater cost savings. However, that information is confidential and therefore average wholesale price had to be used for this project. Most of the cost avoidance was due to 4 patients who could not tolerate imatinib and used nilotinib, which is more expensive.
Limitations
The small sample size presented some limitations. Of the 3819 oncology patients within VISN 21 in FY 2024, 186 received a TKI and only 15 were eligible for discontinuation. Additionally, challenges emerged when discussing discontinuation with community care clinicians and patients. Community care clinicians were difficult to contact, making it challenging to discuss the project with them. CPPs noted hesitancy among VA clinicians and patients to discontinue a medication for which adherence was continually emphasized.
Conclusions
Discussions about CML TKI discontinuation led to collaboration with the oncology care team and could lead to significant cost avoidance. Barriers to TKI discontinuation included patients’ concern for relapse, risk of discontinuation syndrome, the requirement for close monitoring, and clinician buy-in. Outcome studies are needed to gain a greater understanding of the benefits and risks of therapy discontinuation. In the future, evaluation of possible clinical and biological predictors of successful TKI discontinuation may be beneficial.
Chronic myeloid leukemia (CML) is a hematologic malignancy resulting from an acquired mutation. The mutation results in a reciprocal translocation between the long arms of chromosomes 9 and 22 and is known as the Philadelphia chromosome (Ph), or Ph-positive (Ph+) when present. The translocation results in the formation of a BCR-ABL fusion oncogene, which leads to continuous cell cycling and proliferation, altered differentiation, and a loss of apoptosis.1,2
Until the 1980s, CML was considered fatal.3 The mainstay of treatment consisted of 2 oral chemotherapeutic agents, busulfan and hydroxyurea. These medications did not prevent blast crisis, a fatal form of leukemia.4,5 The introduction of tyrosine kinase inhibitors (TKIs) transformed CML management and improved 10-year overall survival from about 20% to > 80% by delaying the transition to blast crisis. Now, the risk of death from general health conditions or comorbidities is higher than that of CML.6
TKIs target the root cause of CML through inhibition of the BCR-ABL oncoprotein.1,2 For CML, the goals of treatment include maintaining hematologic, cytogenetic, and molecular remission; preventing progression to accelerated phase or blast crisis; minimizing toxicity; and enabling potential cessation of therapy in carefully selected patients.7,8
Small cohort studies suggest that dose reduction of TKIs in patients who achieve optimal responses may reduce the risk of long-term adverse effects (AEs). However, optimal dose-reduction and minimum effective dose of each agent are unknown.7 The ability to maintain undetectable minimal residual disease or disease detectable at a stable low level after TKI discontinuation has been called treatment-free remission. Studies suggest that about 40% to 50% of patients who have achieved a stable deep molecular response remain in treatment-free remission after stopping first-line treatment.9,10 Of the patients who relapse following TKI discontinuation, 80% relapse within the first 6 months of treatment cessation. Molecular response is regained in almost all patients when treatment is resumed with the same TKI.11
The National Comprehensive Cancer Network (NCCN) recommends considering discontinuation of TKI therapy only outside the setting of a clinical trial and only in patients who consent to discontinuation after a thorough discussion of the potential risks and benefits. The NCCN criteria for patients who may be eligible for discontinuation are listed in Table 1. The Life After Stopping TKIs study reported that 80% of patients with well-controlled chronic phase CML who discontinued TKIs had a clinically meaningful improvement in fatigue. Patients also reported clinically meaningful improvements in depression, diarrhea, sleep disturbance, and pain interference. These symptoms worsened after restarting TKI therapy.12

TKI DISCONTINUATION
Electronic health record data were extracted using structured query language from the US Department of Veterans Affairs (VA) Corporate Data Warehouse (CDW). To be eligible for discontinuation, veterans had to be aged > 18 years, receive oncology care within a Veterans Integrated Services Network (VISN) 21 health care system (HCS) (VA Sierra Nevada HCS, VA Southern Nevada HCS, VA Central California HCS, VA Palo Alto HCS, VA Northern California HCS, and VA San Francisco HCS) or be a veteran referred to a community-based oncology practitioner. Patients had to have a documented diagnosis of chronic phase CML, have an active order for a TKI, be on TKI therapy for ≥ 3 years, and have a stable molecular response (BCR-ABL1 ≤ 0.01% on the International Scale for ≥ 2 years with ≥ 4 tests done ≥ 3 months apart) as of October 1, 2024. Veterans were excluded if they had a history of advanced accelerated phase CML, previous TKI discontinuation trials, nonadherence to the TKI, or if they did not want to consider TKI discontinuation.
This analysis evaluated the potential cost avoidance associated with TKI discontinuation. Cost avoidance was calculated using the average wholesale price of each TKI. Secondary objectives evaluated health outcomes of TKI discontinuation including CML relapse, reported AEs, long-term remission, and TKI withdrawal syndrome. Health outcomes were determined through chart review of AEs and clinic notes documented in the electronic health record during the study time frame.
Baseline information for eligible patients was collected, including age, sex, and race, and chart reviews were completed to evaluate reported AEs associated with therapy. Oncology clinical pharmacy practitioners (CPPs) at each VISN 21 facility were notified of eligible patients to facilitate discussion with oncologists and establish monitoring if therapy was discontinued. Following TKI discontinuation, health outcomes were evaluated, including CML relapse, changes in reported AEs, long-term remission, and TKI withdrawal syndrome. Descriptive statistics were used to analyze the baseline characteristics. Cost avoidance was calculated using the average wholesale price for each TKI. The number of tablets required to reach each patient’s individual dose was taken into consideration when determining the cost avoidance. A dashboard was created using the query from the CDW and was developed in Microsoft Power BI.
Preliminary Results
In FY 2024, VISN 21 had 3819 oncology patients. Twenty-four patients had taken a TKI for ≥ 3 years, 20 had a stable molecular response, and 15 had not previously attempted to discontinue their TKI (Figure 1). Fifteen veterans were eligible for therapy discontinuation for a total potential annual cost avoidance of $1.2 million (Figure 2). Most of the cost avoidance, $935,057 (78%), was attributed to 3 patients on nilotinib. The mean age of the population was 74 years. All patients were male, and 12 (80%) were White. (Table 2). At baseline, 11 patients (73%) were taking imatinib. One patient received oncology care from a community care clinician. All 15 patients decided to remain on therapy.

Abbreviations: CML, chronic myeloid leukemia; TKI, tyrosine kinase inhibitor;
VISN, Veterans Integrated Service Network.

for 15 patients at Veterans Integrated Services Network 21.

DISCUSSION
As a multisite quality improvement initiative, this project raised awareness of TKI therapy discontinuation in select patients with CML. It also sparked collaboration among oncology CPPs and clinicians and stimulated conversations about CML treatment. The development of the TKI discontinuation dashboard provides a population health management tool for CPPs and clinicians to identify eligible patients in the future.
Adherence to TKIs is crucial for disease control and survival in patients with CML. Patients are counseled that poor adherence to therapy may contribute to worsening disease or suboptimal response, the development of resistance, and greater health care costs.13 Therefore, it was a challenge for patients to understand and accept that they could stop TKI therapy after achieving a stable deep molecular response. Discussions with patients about the goal of therapy—suppressing the BCR-ABL oncogene, which they have achieved—could encourage patients to trial therapy discontinuation.
Only small cohort studies have been completed to evaluate the outcomes of therapy discontinuation. Much remains unknown regarding the optimal dose-reduction strategy and the minimum effective dose of each agent. Additionally, understanding the qualities of a good candidate for TKI discontinuation remains a barrier. A similar project was conducted in VISN 17. Five patients were counseled on TKI discontinuation; however, only 1 discontinued TKI therapy. Unfortunately, soon after discontinuing treatment, the patient had to restart therapy. Additional literature will enhance understanding of therapy discontinuation.
An unexpected finding of TKI discontinuation trials has been a reversible phenomenon known as TKI withdrawal syndrome.9 It can occur regardless of the TKI used and results in pruritus and new or worsening musculoskeletal pain within several weeks of TKI discontinuation in about 30% of patients. Symptoms may last several months and may require acetaminophen or nonsteroidal anti-inflammatory drugs for pain control.9,10,14
The potential cost avoidance of $1.2 million is an underestimation because VA contracts allow for greater cost savings. However, that information is confidential and therefore average wholesale price had to be used for this project. Most of the cost avoidance was due to 4 patients who could not tolerate imatinib and used nilotinib, which is more expensive.
Limitations
The small sample size presented some limitations. Of the 3819 oncology patients within VISN 21 in FY 2024, 186 received a TKI and only 15 were eligible for discontinuation. Additionally, challenges emerged when discussing discontinuation with community care clinicians and patients. Community care clinicians were difficult to contact, making it challenging to discuss the project with them. CPPs noted hesitancy among VA clinicians and patients to discontinue a medication for which adherence was continually emphasized.
Conclusions
Discussions about CML TKI discontinuation led to collaboration with the oncology care team and could lead to significant cost avoidance. Barriers to TKI discontinuation included patients’ concern for relapse, risk of discontinuation syndrome, the requirement for close monitoring, and clinician buy-in. Outcome studies are needed to gain a greater understanding of the benefits and risks of therapy discontinuation. In the future, evaluation of possible clinical and biological predictors of successful TKI discontinuation may be beneficial.
- Schiffer CA. BCR-ABL tyrosine kinase inhibitors for chronic myelogenous leukemia. N Engl J Med. 2007;357:258-265. doi:10.1056/NEJMct071828
- Hehlmann R, Hochhaus A, Baccarani M; European LeukemiaNet. Chronic myeloid leukaemia. Lancet. 2007;370:342-350. doi:10.1016/S0140-6736(07)61165-9
- Goldman JM, Melo JV. Chronic myeloid leukemia--advances in biology and new approaches to treatment. N Engl J Med. 2003;349:1451-1464. doi:10.1056/NEJMra020777
- Pasic I, Lipton JH. Current approach to the treatment of chronic myeloid leukaemia. Leuk Res. 2017;55:65-78. doi:10.1016/j.leukres.2017.01.005
- Rao KV, Iannucci A, Jabbour E. Current and future clinical strategies in the management of chronic myeloid leukemia. Pharmacotherapy. 2010;30:77S-101S. doi:10.1592/phco.30.pt2.77S
- Cortes J, Pavlovsky C, Saußele S. Chronic myeloid leukaemia. Lancet. 2021;398:1914-1926. doi:10.1016/S0140-6736(21)01204-6
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Chronic myeloid leukemia. Version 1.2026. July 16, 2025. Accessed February 8, 2026. https://www.nccn.org /guidelines/guidelines-detail?id=1427
- Hochhaus A, Baccarani M, Silver RT, et al. European LeukemiaNet 2020 recommendations for treating chronic myeloid leukemia. Leukemia. 2020;34:966-984. doi:10.1038/s41375-020-0776-2
- Saußele S, Richter J, Hochhaus A, Mahon F-X. The concept of treatment-free remission in chronic myeloid leukemia. Leukemia. 2016;30:1638-1647. doi:10.1038/leu.2016.115
- Atallah E, Sweet K. Treatment-free remission: the new goal in CML therapy. Curr Hematol Malig Rep. 2021;16:433-439. doi:10.1007/s11899-021-00653-1
- Hehlmann R. The new ELN recommendations for treating CML. J Clin Med. 2020;9:3671. doi:10.3390/jcm9113671
- Atallah E, Schiffer CA, Radich JP , et al. Assessment of outcomes after stopping tyrosine kinase inhibitors among patients with chronic myeloid leukemia: a non-randomized clinical trial. JAMA Oncol. 2021;7:42-50. doi:10.1001/jamaoncol.2020.5774
- Breccia M, Efficace F, Alimena G. Imatinib treatment in chronic myelogenous leukemia: what have we learned so far? Cancer Lett. 2011;300:115-121. doi:10.1016/j.canlet.2010.10.018
- Berman E. How I treat chronic-phase chronic myelogenous leukemia. Blood. 2022;139:3138-3147. doi:10.1182/blood.2021011722
- Schiffer CA. BCR-ABL tyrosine kinase inhibitors for chronic myelogenous leukemia. N Engl J Med. 2007;357:258-265. doi:10.1056/NEJMct071828
- Hehlmann R, Hochhaus A, Baccarani M; European LeukemiaNet. Chronic myeloid leukaemia. Lancet. 2007;370:342-350. doi:10.1016/S0140-6736(07)61165-9
- Goldman JM, Melo JV. Chronic myeloid leukemia--advances in biology and new approaches to treatment. N Engl J Med. 2003;349:1451-1464. doi:10.1056/NEJMra020777
- Pasic I, Lipton JH. Current approach to the treatment of chronic myeloid leukaemia. Leuk Res. 2017;55:65-78. doi:10.1016/j.leukres.2017.01.005
- Rao KV, Iannucci A, Jabbour E. Current and future clinical strategies in the management of chronic myeloid leukemia. Pharmacotherapy. 2010;30:77S-101S. doi:10.1592/phco.30.pt2.77S
- Cortes J, Pavlovsky C, Saußele S. Chronic myeloid leukaemia. Lancet. 2021;398:1914-1926. doi:10.1016/S0140-6736(21)01204-6
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Chronic myeloid leukemia. Version 1.2026. July 16, 2025. Accessed February 8, 2026. https://www.nccn.org /guidelines/guidelines-detail?id=1427
- Hochhaus A, Baccarani M, Silver RT, et al. European LeukemiaNet 2020 recommendations for treating chronic myeloid leukemia. Leukemia. 2020;34:966-984. doi:10.1038/s41375-020-0776-2
- Saußele S, Richter J, Hochhaus A, Mahon F-X. The concept of treatment-free remission in chronic myeloid leukemia. Leukemia. 2016;30:1638-1647. doi:10.1038/leu.2016.115
- Atallah E, Sweet K. Treatment-free remission: the new goal in CML therapy. Curr Hematol Malig Rep. 2021;16:433-439. doi:10.1007/s11899-021-00653-1
- Hehlmann R. The new ELN recommendations for treating CML. J Clin Med. 2020;9:3671. doi:10.3390/jcm9113671
- Atallah E, Schiffer CA, Radich JP , et al. Assessment of outcomes after stopping tyrosine kinase inhibitors among patients with chronic myeloid leukemia: a non-randomized clinical trial. JAMA Oncol. 2021;7:42-50. doi:10.1001/jamaoncol.2020.5774
- Breccia M, Efficace F, Alimena G. Imatinib treatment in chronic myelogenous leukemia: what have we learned so far? Cancer Lett. 2011;300:115-121. doi:10.1016/j.canlet.2010.10.018
- Berman E. How I treat chronic-phase chronic myelogenous leukemia. Blood. 2022;139:3138-3147. doi:10.1182/blood.2021011722
Potential Tyrosine Kinase Inhibitor Therapy Discontinuation for Patients With Chronic Myeloid Leukemia in a VA Regional Network
Potential Tyrosine Kinase Inhibitor Therapy Discontinuation for Patients With Chronic Myeloid Leukemia in a VA Regional Network