User login
Potential Tyrosine Kinase Inhibitor Therapy Discontinuation for Patients With Chronic Myeloid Leukemia in a VA Regional Network
Potential Tyrosine Kinase Inhibitor Therapy Discontinuation for Patients With Chronic Myeloid Leukemia in a VA Regional Network
Chronic myeloid leukemia (CML) is a hematologic malignancy resulting from an acquired mutation. The mutation results in a reciprocal translocation between the long arms of chromosomes 9 and 22 and is known as the Philadelphia chromosome (Ph), or Ph-positive (Ph+) when present. The translocation results in the formation of a BCR-ABL fusion oncogene, which leads to continuous cell cycling and proliferation, altered differentiation, and a loss of apoptosis.1,2
Until the 1980s, CML was considered fatal.3 The mainstay of treatment consisted of 2 oral chemotherapeutic agents, busulfan and hydroxyurea. These medications did not prevent blast crisis, a fatal form of leukemia.4,5 The introduction of tyrosine kinase inhibitors (TKIs) transformed CML management and improved 10-year overall survival from about 20% to > 80% by delaying the transition to blast crisis. Now, the risk of death from general health conditions or comorbidities is higher than that of CML.6
TKIs target the root cause of CML through inhibition of the BCR-ABL oncoprotein.1,2 For CML, the goals of treatment include maintaining hematologic, cytogenetic, and molecular remission; preventing progression to accelerated phase or blast crisis; minimizing toxicity; and enabling potential cessation of therapy in carefully selected patients.7,8
Small cohort studies suggest that dose reduction of TKIs in patients who achieve optimal responses may reduce the risk of long-term adverse effects (AEs). However, optimal dose-reduction and minimum effective dose of each agent are unknown.7 The ability to maintain undetectable minimal residual disease or disease detectable at a stable low level after TKI discontinuation has been called treatment-free remission. Studies suggest that about 40% to 50% of patients who have achieved a stable deep molecular response remain in treatment-free remission after stopping first-line treatment.9,10 Of the patients who relapse following TKI discontinuation, 80% relapse within the first 6 months of treatment cessation. Molecular response is regained in almost all patients when treatment is resumed with the same TKI.11
The National Comprehensive Cancer Network (NCCN) recommends considering discontinuation of TKI therapy only outside the setting of a clinical trial and only in patients who consent to discontinuation after a thorough discussion of the potential risks and benefits. The NCCN criteria for patients who may be eligible for discontinuation are listed in Table 1. The Life After Stopping TKIs study reported that 80% of patients with well-controlled chronic phase CML who discontinued TKIs had a clinically meaningful improvement in fatigue. Patients also reported clinically meaningful improvements in depression, diarrhea, sleep disturbance, and pain interference. These symptoms worsened after restarting TKI therapy.12

TKI DISCONTINUATION
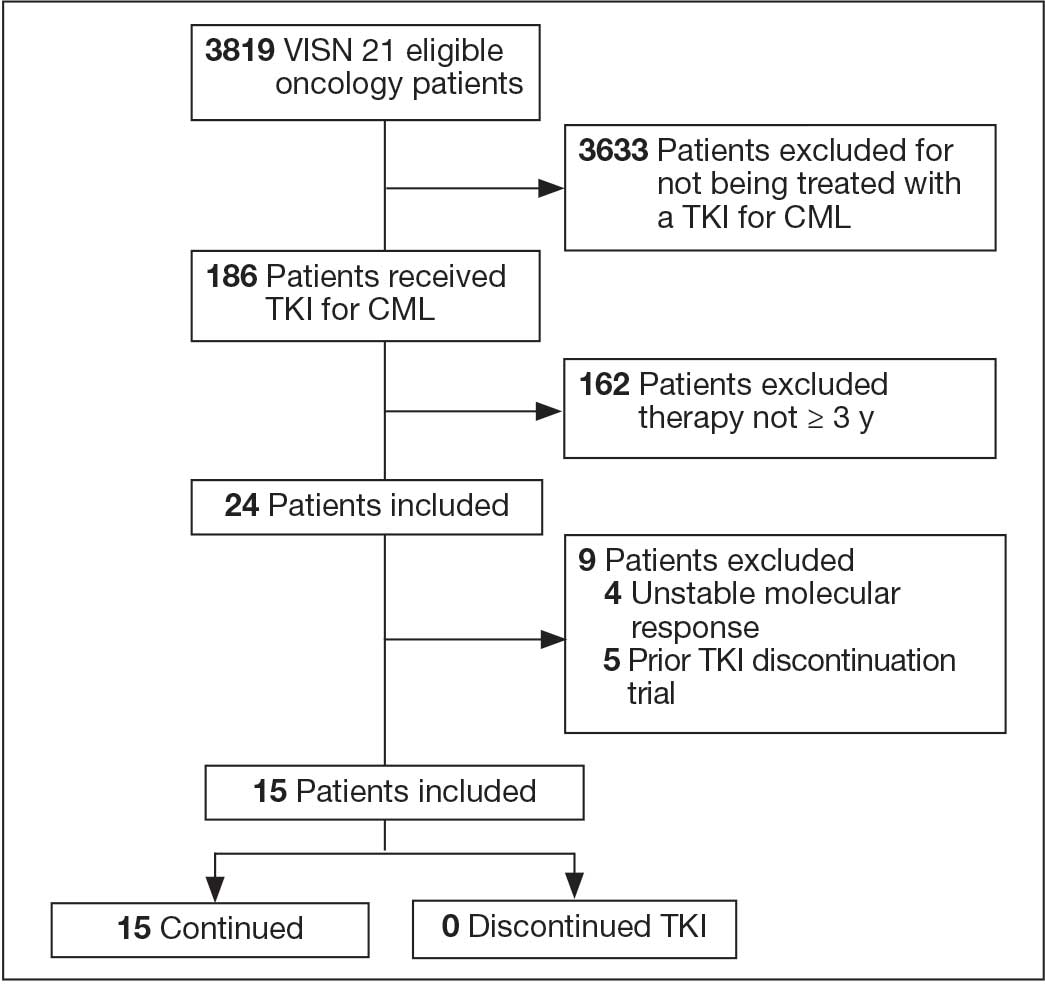
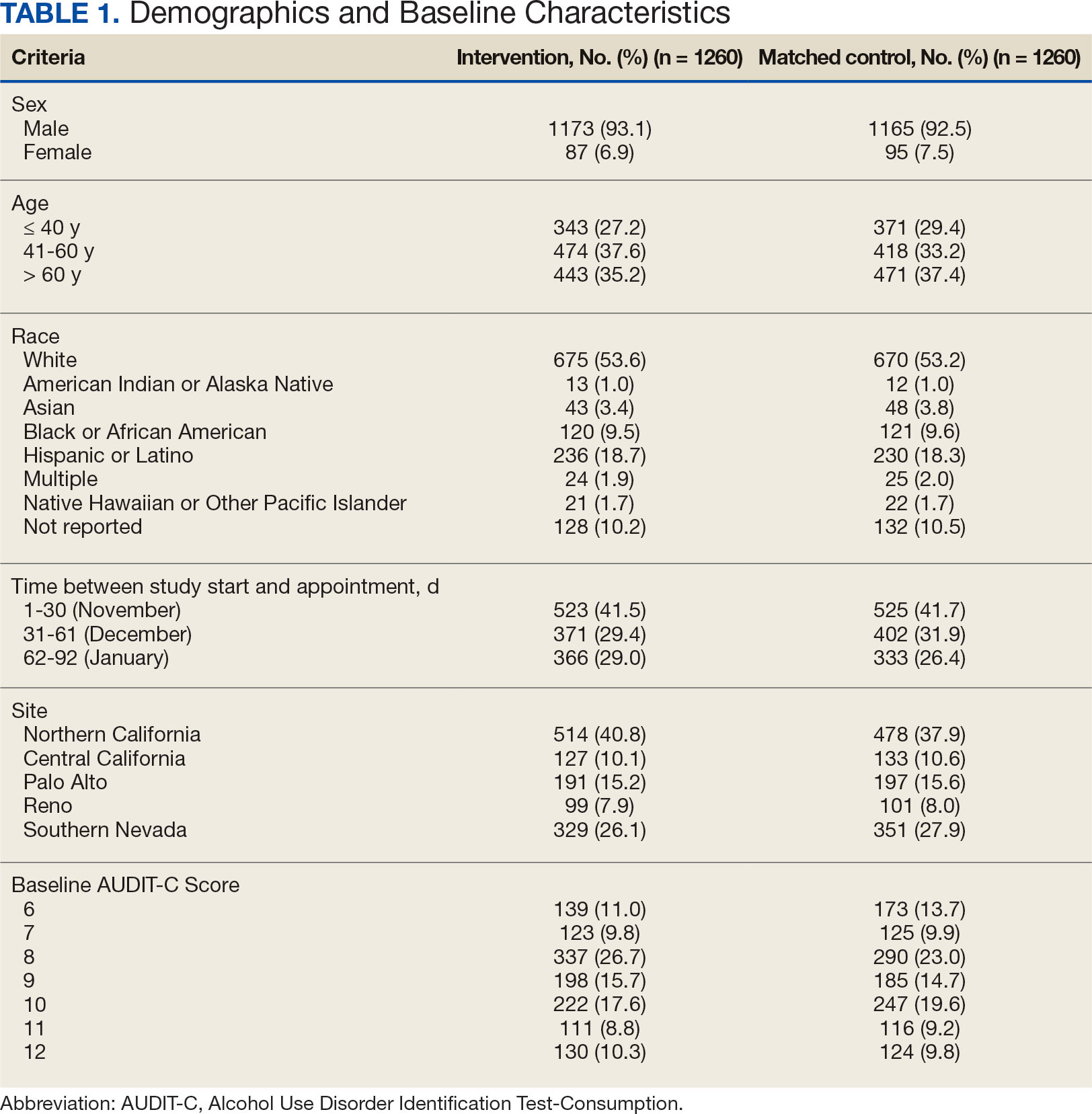
Electronic health record data were extracted using structured query language from the US Department of Veterans Affairs (VA) Corporate Data Warehouse (CDW). To be eligible for discontinuation, veterans had to be aged > 18 years, receive oncology care within a Veterans Integrated Services Network (VISN) 21 health care system (HCS) (VA Sierra Nevada HCS, VA Southern Nevada HCS, VA Central California HCS, VA Palo Alto HCS, VA Northern California HCS, and VA San Francisco HCS) or be a veteran referred to a community-based oncology practitioner. Patients had to have a documented diagnosis of chronic phase CML, have an active order for a TKI, be on TKI therapy for ≥ 3 years, and have a stable molecular response (BCR-ABL1 ≤ 0.01% on the International Scale for ≥ 2 years with ≥ 4 tests done ≥ 3 months apart) as of October 1, 2024. Veterans were excluded if they had a history of advanced accelerated phase CML, previous TKI discontinuation trials, nonadherence to the TKI, or if they did not want to consider TKI discontinuation.
This analysis evaluated the potential cost avoidance associated with TKI discontinuation. Cost avoidance was calculated using the average wholesale price of each TKI. Secondary objectives evaluated health outcomes of TKI discontinuation including CML relapse, reported AEs, long-term remission, and TKI withdrawal syndrome. Health outcomes were determined through chart review of AEs and clinic notes documented in the electronic health record during the study time frame.
Baseline information for eligible patients was collected, including age, sex, and race, and chart reviews were completed to evaluate reported AEs associated with therapy. Oncology clinical pharmacy practitioners (CPPs) at each VISN 21 facility were notified of eligible patients to facilitate discussion with oncologists and establish monitoring if therapy was discontinued. Following TKI discontinuation, health outcomes were evaluated, including CML relapse, changes in reported AEs, long-term remission, and TKI withdrawal syndrome. Descriptive statistics were used to analyze the baseline characteristics. Cost avoidance was calculated using the average wholesale price for each TKI. The number of tablets required to reach each patient’s individual dose was taken into consideration when determining the cost avoidance. A dashboard was created using the query from the CDW and was developed in Microsoft Power BI.
Preliminary Results
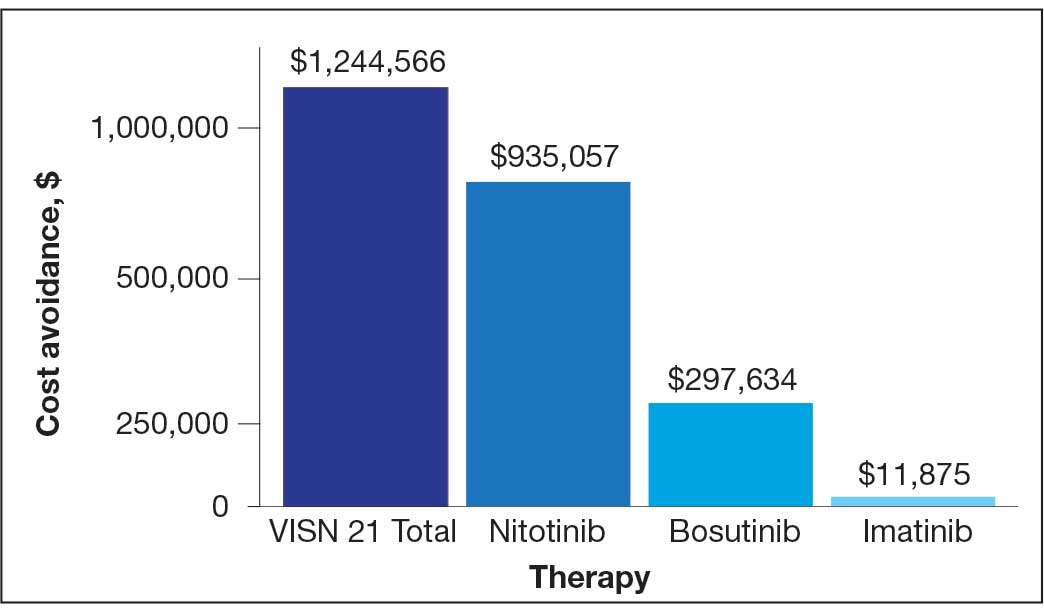
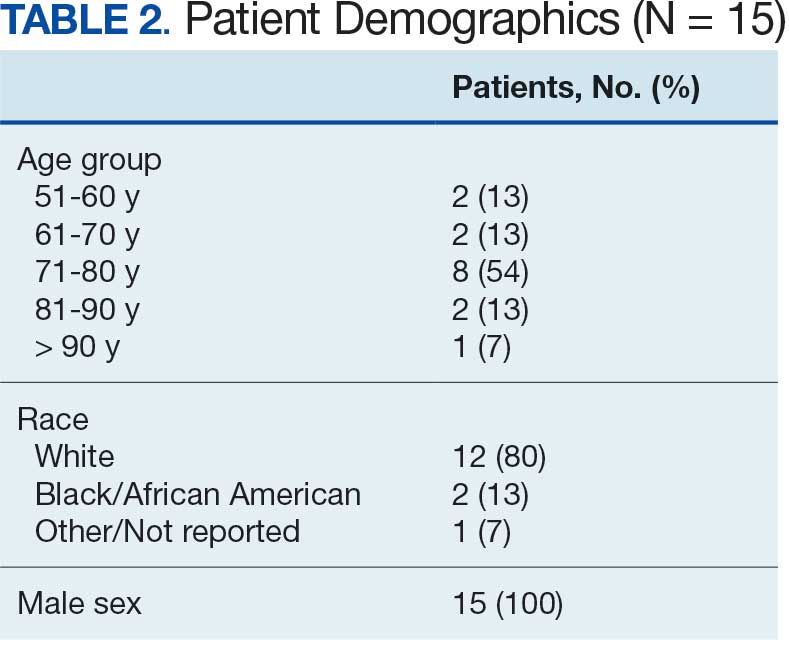
In FY 2024, VISN 21 had 3819 oncology patients. Twenty-four patients had taken a TKI for ≥ 3 years, 20 had a stable molecular response, and 15 had not previously attempted to discontinue their TKI (Figure 1). Fifteen veterans were eligible for therapy discontinuation for a total potential annual cost avoidance of $1.2 million (Figure 2). Most of the cost avoidance, $935,057 (78%), was attributed to 3 patients on nilotinib. The mean age of the population was 74 years. All patients were male, and 12 (80%) were White. (Table 2). At baseline, 11 patients (73%) were taking imatinib. One patient received oncology care from a community care clinician. All 15 patients decided to remain on therapy.
Abbreviations: CML, chronic myeloid leukemia; TKI, tyrosine kinase inhibitor;
VISN, Veterans Integrated Service Network.
for 15 patients at Veterans Integrated Services Network 21.
DISCUSSION
As a multisite quality improvement initiative, this project raised awareness of TKI therapy discontinuation in select patients with CML. It also sparked collaboration among oncology CPPs and clinicians and stimulated conversations about CML treatment. The development of the TKI discontinuation dashboard provides a population health management tool for CPPs and clinicians to identify eligible patients in the future.
Adherence to TKIs is crucial for disease control and survival in patients with CML. Patients are counseled that poor adherence to therapy may contribute to worsening disease or suboptimal response, the development of resistance, and greater health care costs.13 Therefore, it was a challenge for patients to understand and accept that they could stop TKI therapy after achieving a stable deep molecular response. Discussions with patients about the goal of therapy—suppressing the BCR-ABL oncogene, which they have achieved—could encourage patients to trial therapy discontinuation.
Only small cohort studies have been completed to evaluate the outcomes of therapy discontinuation. Much remains unknown regarding the optimal dose-reduction strategy and the minimum effective dose of each agent. Additionally, understanding the qualities of a good candidate for TKI discontinuation remains a barrier. A similar project was conducted in VISN 17. Five patients were counseled on TKI discontinuation; however, only 1 discontinued TKI therapy. Unfortunately, soon after discontinuing treatment, the patient had to restart therapy. Additional literature will enhance understanding of therapy discontinuation.
An unexpected finding of TKI discontinuation trials has been a reversible phenomenon known as TKI withdrawal syndrome.9 It can occur regardless of the TKI used and results in pruritus and new or worsening musculoskeletal pain within several weeks of TKI discontinuation in about 30% of patients. Symptoms may last several months and may require acetaminophen or nonsteroidal anti-inflammatory drugs for pain control.9,10,14
The potential cost avoidance of $1.2 million is an underestimation because VA contracts allow for greater cost savings. However, that information is confidential and therefore average wholesale price had to be used for this project. Most of the cost avoidance was due to 4 patients who could not tolerate imatinib and used nilotinib, which is more expensive.
Limitations
The small sample size presented some limitations. Of the 3819 oncology patients within VISN 21 in FY 2024, 186 received a TKI and only 15 were eligible for discontinuation. Additionally, challenges emerged when discussing discontinuation with community care clinicians and patients. Community care clinicians were difficult to contact, making it challenging to discuss the project with them. CPPs noted hesitancy among VA clinicians and patients to discontinue a medication for which adherence was continually emphasized.
Conclusions
Discussions about CML TKI discontinuation led to collaboration with the oncology care team and could lead to significant cost avoidance. Barriers to TKI discontinuation included patients’ concern for relapse, risk of discontinuation syndrome, the requirement for close monitoring, and clinician buy-in. Outcome studies are needed to gain a greater understanding of the benefits and risks of therapy discontinuation. In the future, evaluation of possible clinical and biological predictors of successful TKI discontinuation may be beneficial.
- Schiffer CA. BCR-ABL tyrosine kinase inhibitors for chronic myelogenous leukemia. N Engl J Med. 2007;357:258-265. doi:10.1056/NEJMct071828
- Hehlmann R, Hochhaus A, Baccarani M; European LeukemiaNet. Chronic myeloid leukaemia. Lancet. 2007;370:342-350. doi:10.1016/S0140-6736(07)61165-9
- Goldman JM, Melo JV. Chronic myeloid leukemia--advances in biology and new approaches to treatment. N Engl J Med. 2003;349:1451-1464. doi:10.1056/NEJMra020777
- Pasic I, Lipton JH. Current approach to the treatment of chronic myeloid leukaemia. Leuk Res. 2017;55:65-78. doi:10.1016/j.leukres.2017.01.005
- Rao KV, Iannucci A, Jabbour E. Current and future clinical strategies in the management of chronic myeloid leukemia. Pharmacotherapy. 2010;30:77S-101S. doi:10.1592/phco.30.pt2.77S
- Cortes J, Pavlovsky C, Saußele S. Chronic myeloid leukaemia. Lancet. 2021;398:1914-1926. doi:10.1016/S0140-6736(21)01204-6
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Chronic myeloid leukemia. Version 1.2026. July 16, 2025. Accessed February 8, 2026. https://www.nccn.org /guidelines/guidelines-detail?id=1427
- Hochhaus A, Baccarani M, Silver RT, et al. European LeukemiaNet 2020 recommendations for treating chronic myeloid leukemia. Leukemia. 2020;34:966-984. doi:10.1038/s41375-020-0776-2
- Saußele S, Richter J, Hochhaus A, Mahon F-X. The concept of treatment-free remission in chronic myeloid leukemia. Leukemia. 2016;30:1638-1647. doi:10.1038/leu.2016.115
- Atallah E, Sweet K. Treatment-free remission: the new goal in CML therapy. Curr Hematol Malig Rep. 2021;16:433-439. doi:10.1007/s11899-021-00653-1
- Hehlmann R. The new ELN recommendations for treating CML. J Clin Med. 2020;9:3671. doi:10.3390/jcm9113671
- Atallah E, Schiffer CA, Radich JP , et al. Assessment of outcomes after stopping tyrosine kinase inhibitors among patients with chronic myeloid leukemia: a non-randomized clinical trial. JAMA Oncol. 2021;7:42-50. doi:10.1001/jamaoncol.2020.5774
- Breccia M, Efficace F, Alimena G. Imatinib treatment in chronic myelogenous leukemia: what have we learned so far? Cancer Lett. 2011;300:115-121. doi:10.1016/j.canlet.2010.10.018
- Berman E. How I treat chronic-phase chronic myelogenous leukemia. Blood. 2022;139:3138-3147. doi:10.1182/blood.2021011722
Chronic myeloid leukemia (CML) is a hematologic malignancy resulting from an acquired mutation. The mutation results in a reciprocal translocation between the long arms of chromosomes 9 and 22 and is known as the Philadelphia chromosome (Ph), or Ph-positive (Ph+) when present. The translocation results in the formation of a BCR-ABL fusion oncogene, which leads to continuous cell cycling and proliferation, altered differentiation, and a loss of apoptosis.1,2
Until the 1980s, CML was considered fatal.3 The mainstay of treatment consisted of 2 oral chemotherapeutic agents, busulfan and hydroxyurea. These medications did not prevent blast crisis, a fatal form of leukemia.4,5 The introduction of tyrosine kinase inhibitors (TKIs) transformed CML management and improved 10-year overall survival from about 20% to > 80% by delaying the transition to blast crisis. Now, the risk of death from general health conditions or comorbidities is higher than that of CML.6
TKIs target the root cause of CML through inhibition of the BCR-ABL oncoprotein.1,2 For CML, the goals of treatment include maintaining hematologic, cytogenetic, and molecular remission; preventing progression to accelerated phase or blast crisis; minimizing toxicity; and enabling potential cessation of therapy in carefully selected patients.7,8
Small cohort studies suggest that dose reduction of TKIs in patients who achieve optimal responses may reduce the risk of long-term adverse effects (AEs). However, optimal dose-reduction and minimum effective dose of each agent are unknown.7 The ability to maintain undetectable minimal residual disease or disease detectable at a stable low level after TKI discontinuation has been called treatment-free remission. Studies suggest that about 40% to 50% of patients who have achieved a stable deep molecular response remain in treatment-free remission after stopping first-line treatment.9,10 Of the patients who relapse following TKI discontinuation, 80% relapse within the first 6 months of treatment cessation. Molecular response is regained in almost all patients when treatment is resumed with the same TKI.11
The National Comprehensive Cancer Network (NCCN) recommends considering discontinuation of TKI therapy only outside the setting of a clinical trial and only in patients who consent to discontinuation after a thorough discussion of the potential risks and benefits. The NCCN criteria for patients who may be eligible for discontinuation are listed in Table 1. The Life After Stopping TKIs study reported that 80% of patients with well-controlled chronic phase CML who discontinued TKIs had a clinically meaningful improvement in fatigue. Patients also reported clinically meaningful improvements in depression, diarrhea, sleep disturbance, and pain interference. These symptoms worsened after restarting TKI therapy.12

TKI DISCONTINUATION
Electronic health record data were extracted using structured query language from the US Department of Veterans Affairs (VA) Corporate Data Warehouse (CDW). To be eligible for discontinuation, veterans had to be aged > 18 years, receive oncology care within a Veterans Integrated Services Network (VISN) 21 health care system (HCS) (VA Sierra Nevada HCS, VA Southern Nevada HCS, VA Central California HCS, VA Palo Alto HCS, VA Northern California HCS, and VA San Francisco HCS) or be a veteran referred to a community-based oncology practitioner. Patients had to have a documented diagnosis of chronic phase CML, have an active order for a TKI, be on TKI therapy for ≥ 3 years, and have a stable molecular response (BCR-ABL1 ≤ 0.01% on the International Scale for ≥ 2 years with ≥ 4 tests done ≥ 3 months apart) as of October 1, 2024. Veterans were excluded if they had a history of advanced accelerated phase CML, previous TKI discontinuation trials, nonadherence to the TKI, or if they did not want to consider TKI discontinuation.
This analysis evaluated the potential cost avoidance associated with TKI discontinuation. Cost avoidance was calculated using the average wholesale price of each TKI. Secondary objectives evaluated health outcomes of TKI discontinuation including CML relapse, reported AEs, long-term remission, and TKI withdrawal syndrome. Health outcomes were determined through chart review of AEs and clinic notes documented in the electronic health record during the study time frame.
Baseline information for eligible patients was collected, including age, sex, and race, and chart reviews were completed to evaluate reported AEs associated with therapy. Oncology clinical pharmacy practitioners (CPPs) at each VISN 21 facility were notified of eligible patients to facilitate discussion with oncologists and establish monitoring if therapy was discontinued. Following TKI discontinuation, health outcomes were evaluated, including CML relapse, changes in reported AEs, long-term remission, and TKI withdrawal syndrome. Descriptive statistics were used to analyze the baseline characteristics. Cost avoidance was calculated using the average wholesale price for each TKI. The number of tablets required to reach each patient’s individual dose was taken into consideration when determining the cost avoidance. A dashboard was created using the query from the CDW and was developed in Microsoft Power BI.
Preliminary Results
In FY 2024, VISN 21 had 3819 oncology patients. Twenty-four patients had taken a TKI for ≥ 3 years, 20 had a stable molecular response, and 15 had not previously attempted to discontinue their TKI (Figure 1). Fifteen veterans were eligible for therapy discontinuation for a total potential annual cost avoidance of $1.2 million (Figure 2). Most of the cost avoidance, $935,057 (78%), was attributed to 3 patients on nilotinib. The mean age of the population was 74 years. All patients were male, and 12 (80%) were White. (Table 2). At baseline, 11 patients (73%) were taking imatinib. One patient received oncology care from a community care clinician. All 15 patients decided to remain on therapy.

Abbreviations: CML, chronic myeloid leukemia; TKI, tyrosine kinase inhibitor;
VISN, Veterans Integrated Service Network.

for 15 patients at Veterans Integrated Services Network 21.

DISCUSSION
As a multisite quality improvement initiative, this project raised awareness of TKI therapy discontinuation in select patients with CML. It also sparked collaboration among oncology CPPs and clinicians and stimulated conversations about CML treatment. The development of the TKI discontinuation dashboard provides a population health management tool for CPPs and clinicians to identify eligible patients in the future.
Adherence to TKIs is crucial for disease control and survival in patients with CML. Patients are counseled that poor adherence to therapy may contribute to worsening disease or suboptimal response, the development of resistance, and greater health care costs.13 Therefore, it was a challenge for patients to understand and accept that they could stop TKI therapy after achieving a stable deep molecular response. Discussions with patients about the goal of therapy—suppressing the BCR-ABL oncogene, which they have achieved—could encourage patients to trial therapy discontinuation.
Only small cohort studies have been completed to evaluate the outcomes of therapy discontinuation. Much remains unknown regarding the optimal dose-reduction strategy and the minimum effective dose of each agent. Additionally, understanding the qualities of a good candidate for TKI discontinuation remains a barrier. A similar project was conducted in VISN 17. Five patients were counseled on TKI discontinuation; however, only 1 discontinued TKI therapy. Unfortunately, soon after discontinuing treatment, the patient had to restart therapy. Additional literature will enhance understanding of therapy discontinuation.
An unexpected finding of TKI discontinuation trials has been a reversible phenomenon known as TKI withdrawal syndrome.9 It can occur regardless of the TKI used and results in pruritus and new or worsening musculoskeletal pain within several weeks of TKI discontinuation in about 30% of patients. Symptoms may last several months and may require acetaminophen or nonsteroidal anti-inflammatory drugs for pain control.9,10,14
The potential cost avoidance of $1.2 million is an underestimation because VA contracts allow for greater cost savings. However, that information is confidential and therefore average wholesale price had to be used for this project. Most of the cost avoidance was due to 4 patients who could not tolerate imatinib and used nilotinib, which is more expensive.
Limitations
The small sample size presented some limitations. Of the 3819 oncology patients within VISN 21 in FY 2024, 186 received a TKI and only 15 were eligible for discontinuation. Additionally, challenges emerged when discussing discontinuation with community care clinicians and patients. Community care clinicians were difficult to contact, making it challenging to discuss the project with them. CPPs noted hesitancy among VA clinicians and patients to discontinue a medication for which adherence was continually emphasized.
Conclusions
Discussions about CML TKI discontinuation led to collaboration with the oncology care team and could lead to significant cost avoidance. Barriers to TKI discontinuation included patients’ concern for relapse, risk of discontinuation syndrome, the requirement for close monitoring, and clinician buy-in. Outcome studies are needed to gain a greater understanding of the benefits and risks of therapy discontinuation. In the future, evaluation of possible clinical and biological predictors of successful TKI discontinuation may be beneficial.
Chronic myeloid leukemia (CML) is a hematologic malignancy resulting from an acquired mutation. The mutation results in a reciprocal translocation between the long arms of chromosomes 9 and 22 and is known as the Philadelphia chromosome (Ph), or Ph-positive (Ph+) when present. The translocation results in the formation of a BCR-ABL fusion oncogene, which leads to continuous cell cycling and proliferation, altered differentiation, and a loss of apoptosis.1,2
Until the 1980s, CML was considered fatal.3 The mainstay of treatment consisted of 2 oral chemotherapeutic agents, busulfan and hydroxyurea. These medications did not prevent blast crisis, a fatal form of leukemia.4,5 The introduction of tyrosine kinase inhibitors (TKIs) transformed CML management and improved 10-year overall survival from about 20% to > 80% by delaying the transition to blast crisis. Now, the risk of death from general health conditions or comorbidities is higher than that of CML.6
TKIs target the root cause of CML through inhibition of the BCR-ABL oncoprotein.1,2 For CML, the goals of treatment include maintaining hematologic, cytogenetic, and molecular remission; preventing progression to accelerated phase or blast crisis; minimizing toxicity; and enabling potential cessation of therapy in carefully selected patients.7,8
Small cohort studies suggest that dose reduction of TKIs in patients who achieve optimal responses may reduce the risk of long-term adverse effects (AEs). However, optimal dose-reduction and minimum effective dose of each agent are unknown.7 The ability to maintain undetectable minimal residual disease or disease detectable at a stable low level after TKI discontinuation has been called treatment-free remission. Studies suggest that about 40% to 50% of patients who have achieved a stable deep molecular response remain in treatment-free remission after stopping first-line treatment.9,10 Of the patients who relapse following TKI discontinuation, 80% relapse within the first 6 months of treatment cessation. Molecular response is regained in almost all patients when treatment is resumed with the same TKI.11
The National Comprehensive Cancer Network (NCCN) recommends considering discontinuation of TKI therapy only outside the setting of a clinical trial and only in patients who consent to discontinuation after a thorough discussion of the potential risks and benefits. The NCCN criteria for patients who may be eligible for discontinuation are listed in Table 1. The Life After Stopping TKIs study reported that 80% of patients with well-controlled chronic phase CML who discontinued TKIs had a clinically meaningful improvement in fatigue. Patients also reported clinically meaningful improvements in depression, diarrhea, sleep disturbance, and pain interference. These symptoms worsened after restarting TKI therapy.12

TKI DISCONTINUATION
Electronic health record data were extracted using structured query language from the US Department of Veterans Affairs (VA) Corporate Data Warehouse (CDW). To be eligible for discontinuation, veterans had to be aged > 18 years, receive oncology care within a Veterans Integrated Services Network (VISN) 21 health care system (HCS) (VA Sierra Nevada HCS, VA Southern Nevada HCS, VA Central California HCS, VA Palo Alto HCS, VA Northern California HCS, and VA San Francisco HCS) or be a veteran referred to a community-based oncology practitioner. Patients had to have a documented diagnosis of chronic phase CML, have an active order for a TKI, be on TKI therapy for ≥ 3 years, and have a stable molecular response (BCR-ABL1 ≤ 0.01% on the International Scale for ≥ 2 years with ≥ 4 tests done ≥ 3 months apart) as of October 1, 2024. Veterans were excluded if they had a history of advanced accelerated phase CML, previous TKI discontinuation trials, nonadherence to the TKI, or if they did not want to consider TKI discontinuation.
This analysis evaluated the potential cost avoidance associated with TKI discontinuation. Cost avoidance was calculated using the average wholesale price of each TKI. Secondary objectives evaluated health outcomes of TKI discontinuation including CML relapse, reported AEs, long-term remission, and TKI withdrawal syndrome. Health outcomes were determined through chart review of AEs and clinic notes documented in the electronic health record during the study time frame.
Baseline information for eligible patients was collected, including age, sex, and race, and chart reviews were completed to evaluate reported AEs associated with therapy. Oncology clinical pharmacy practitioners (CPPs) at each VISN 21 facility were notified of eligible patients to facilitate discussion with oncologists and establish monitoring if therapy was discontinued. Following TKI discontinuation, health outcomes were evaluated, including CML relapse, changes in reported AEs, long-term remission, and TKI withdrawal syndrome. Descriptive statistics were used to analyze the baseline characteristics. Cost avoidance was calculated using the average wholesale price for each TKI. The number of tablets required to reach each patient’s individual dose was taken into consideration when determining the cost avoidance. A dashboard was created using the query from the CDW and was developed in Microsoft Power BI.
Preliminary Results
In FY 2024, VISN 21 had 3819 oncology patients. Twenty-four patients had taken a TKI for ≥ 3 years, 20 had a stable molecular response, and 15 had not previously attempted to discontinue their TKI (Figure 1). Fifteen veterans were eligible for therapy discontinuation for a total potential annual cost avoidance of $1.2 million (Figure 2). Most of the cost avoidance, $935,057 (78%), was attributed to 3 patients on nilotinib. The mean age of the population was 74 years. All patients were male, and 12 (80%) were White. (Table 2). At baseline, 11 patients (73%) were taking imatinib. One patient received oncology care from a community care clinician. All 15 patients decided to remain on therapy.

Abbreviations: CML, chronic myeloid leukemia; TKI, tyrosine kinase inhibitor;
VISN, Veterans Integrated Service Network.

for 15 patients at Veterans Integrated Services Network 21.

DISCUSSION
As a multisite quality improvement initiative, this project raised awareness of TKI therapy discontinuation in select patients with CML. It also sparked collaboration among oncology CPPs and clinicians and stimulated conversations about CML treatment. The development of the TKI discontinuation dashboard provides a population health management tool for CPPs and clinicians to identify eligible patients in the future.
Adherence to TKIs is crucial for disease control and survival in patients with CML. Patients are counseled that poor adherence to therapy may contribute to worsening disease or suboptimal response, the development of resistance, and greater health care costs.13 Therefore, it was a challenge for patients to understand and accept that they could stop TKI therapy after achieving a stable deep molecular response. Discussions with patients about the goal of therapy—suppressing the BCR-ABL oncogene, which they have achieved—could encourage patients to trial therapy discontinuation.
Only small cohort studies have been completed to evaluate the outcomes of therapy discontinuation. Much remains unknown regarding the optimal dose-reduction strategy and the minimum effective dose of each agent. Additionally, understanding the qualities of a good candidate for TKI discontinuation remains a barrier. A similar project was conducted in VISN 17. Five patients were counseled on TKI discontinuation; however, only 1 discontinued TKI therapy. Unfortunately, soon after discontinuing treatment, the patient had to restart therapy. Additional literature will enhance understanding of therapy discontinuation.
An unexpected finding of TKI discontinuation trials has been a reversible phenomenon known as TKI withdrawal syndrome.9 It can occur regardless of the TKI used and results in pruritus and new or worsening musculoskeletal pain within several weeks of TKI discontinuation in about 30% of patients. Symptoms may last several months and may require acetaminophen or nonsteroidal anti-inflammatory drugs for pain control.9,10,14
The potential cost avoidance of $1.2 million is an underestimation because VA contracts allow for greater cost savings. However, that information is confidential and therefore average wholesale price had to be used for this project. Most of the cost avoidance was due to 4 patients who could not tolerate imatinib and used nilotinib, which is more expensive.
Limitations
The small sample size presented some limitations. Of the 3819 oncology patients within VISN 21 in FY 2024, 186 received a TKI and only 15 were eligible for discontinuation. Additionally, challenges emerged when discussing discontinuation with community care clinicians and patients. Community care clinicians were difficult to contact, making it challenging to discuss the project with them. CPPs noted hesitancy among VA clinicians and patients to discontinue a medication for which adherence was continually emphasized.
Conclusions
Discussions about CML TKI discontinuation led to collaboration with the oncology care team and could lead to significant cost avoidance. Barriers to TKI discontinuation included patients’ concern for relapse, risk of discontinuation syndrome, the requirement for close monitoring, and clinician buy-in. Outcome studies are needed to gain a greater understanding of the benefits and risks of therapy discontinuation. In the future, evaluation of possible clinical and biological predictors of successful TKI discontinuation may be beneficial.
- Schiffer CA. BCR-ABL tyrosine kinase inhibitors for chronic myelogenous leukemia. N Engl J Med. 2007;357:258-265. doi:10.1056/NEJMct071828
- Hehlmann R, Hochhaus A, Baccarani M; European LeukemiaNet. Chronic myeloid leukaemia. Lancet. 2007;370:342-350. doi:10.1016/S0140-6736(07)61165-9
- Goldman JM, Melo JV. Chronic myeloid leukemia--advances in biology and new approaches to treatment. N Engl J Med. 2003;349:1451-1464. doi:10.1056/NEJMra020777
- Pasic I, Lipton JH. Current approach to the treatment of chronic myeloid leukaemia. Leuk Res. 2017;55:65-78. doi:10.1016/j.leukres.2017.01.005
- Rao KV, Iannucci A, Jabbour E. Current and future clinical strategies in the management of chronic myeloid leukemia. Pharmacotherapy. 2010;30:77S-101S. doi:10.1592/phco.30.pt2.77S
- Cortes J, Pavlovsky C, Saußele S. Chronic myeloid leukaemia. Lancet. 2021;398:1914-1926. doi:10.1016/S0140-6736(21)01204-6
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Chronic myeloid leukemia. Version 1.2026. July 16, 2025. Accessed February 8, 2026. https://www.nccn.org /guidelines/guidelines-detail?id=1427
- Hochhaus A, Baccarani M, Silver RT, et al. European LeukemiaNet 2020 recommendations for treating chronic myeloid leukemia. Leukemia. 2020;34:966-984. doi:10.1038/s41375-020-0776-2
- Saußele S, Richter J, Hochhaus A, Mahon F-X. The concept of treatment-free remission in chronic myeloid leukemia. Leukemia. 2016;30:1638-1647. doi:10.1038/leu.2016.115
- Atallah E, Sweet K. Treatment-free remission: the new goal in CML therapy. Curr Hematol Malig Rep. 2021;16:433-439. doi:10.1007/s11899-021-00653-1
- Hehlmann R. The new ELN recommendations for treating CML. J Clin Med. 2020;9:3671. doi:10.3390/jcm9113671
- Atallah E, Schiffer CA, Radich JP , et al. Assessment of outcomes after stopping tyrosine kinase inhibitors among patients with chronic myeloid leukemia: a non-randomized clinical trial. JAMA Oncol. 2021;7:42-50. doi:10.1001/jamaoncol.2020.5774
- Breccia M, Efficace F, Alimena G. Imatinib treatment in chronic myelogenous leukemia: what have we learned so far? Cancer Lett. 2011;300:115-121. doi:10.1016/j.canlet.2010.10.018
- Berman E. How I treat chronic-phase chronic myelogenous leukemia. Blood. 2022;139:3138-3147. doi:10.1182/blood.2021011722
- Schiffer CA. BCR-ABL tyrosine kinase inhibitors for chronic myelogenous leukemia. N Engl J Med. 2007;357:258-265. doi:10.1056/NEJMct071828
- Hehlmann R, Hochhaus A, Baccarani M; European LeukemiaNet. Chronic myeloid leukaemia. Lancet. 2007;370:342-350. doi:10.1016/S0140-6736(07)61165-9
- Goldman JM, Melo JV. Chronic myeloid leukemia--advances in biology and new approaches to treatment. N Engl J Med. 2003;349:1451-1464. doi:10.1056/NEJMra020777
- Pasic I, Lipton JH. Current approach to the treatment of chronic myeloid leukaemia. Leuk Res. 2017;55:65-78. doi:10.1016/j.leukres.2017.01.005
- Rao KV, Iannucci A, Jabbour E. Current and future clinical strategies in the management of chronic myeloid leukemia. Pharmacotherapy. 2010;30:77S-101S. doi:10.1592/phco.30.pt2.77S
- Cortes J, Pavlovsky C, Saußele S. Chronic myeloid leukaemia. Lancet. 2021;398:1914-1926. doi:10.1016/S0140-6736(21)01204-6
- National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Chronic myeloid leukemia. Version 1.2026. July 16, 2025. Accessed February 8, 2026. https://www.nccn.org /guidelines/guidelines-detail?id=1427
- Hochhaus A, Baccarani M, Silver RT, et al. European LeukemiaNet 2020 recommendations for treating chronic myeloid leukemia. Leukemia. 2020;34:966-984. doi:10.1038/s41375-020-0776-2
- Saußele S, Richter J, Hochhaus A, Mahon F-X. The concept of treatment-free remission in chronic myeloid leukemia. Leukemia. 2016;30:1638-1647. doi:10.1038/leu.2016.115
- Atallah E, Sweet K. Treatment-free remission: the new goal in CML therapy. Curr Hematol Malig Rep. 2021;16:433-439. doi:10.1007/s11899-021-00653-1
- Hehlmann R. The new ELN recommendations for treating CML. J Clin Med. 2020;9:3671. doi:10.3390/jcm9113671
- Atallah E, Schiffer CA, Radich JP , et al. Assessment of outcomes after stopping tyrosine kinase inhibitors among patients with chronic myeloid leukemia: a non-randomized clinical trial. JAMA Oncol. 2021;7:42-50. doi:10.1001/jamaoncol.2020.5774
- Breccia M, Efficace F, Alimena G. Imatinib treatment in chronic myelogenous leukemia: what have we learned so far? Cancer Lett. 2011;300:115-121. doi:10.1016/j.canlet.2010.10.018
- Berman E. How I treat chronic-phase chronic myelogenous leukemia. Blood. 2022;139:3138-3147. doi:10.1182/blood.2021011722
Potential Tyrosine Kinase Inhibitor Therapy Discontinuation for Patients With Chronic Myeloid Leukemia in a VA Regional Network
Potential Tyrosine Kinase Inhibitor Therapy Discontinuation for Patients With Chronic Myeloid Leukemia in a VA Regional Network
Impact of Multisite Patient Education on Pharmacotherapy for Veterans With Alcohol Use Disorder
Impact of Multisite Patient Education on Pharmacotherapy for Veterans With Alcohol Use Disorder
Excessive alcohol use is one of the leading preventable causes of death in the United States, responsible for about 178,000 deaths annually and an average of 488 daily deaths in 2020 and 2021.1Alcohol-related deaths increased by 49% between 2006 and 2019.2 This trend continued during the COVID-19 pandemic, with death certificates that listed alcohol increasing by > 25% from 2019 to 2020, and another 10% in 2021.3 This increase of alcohol-related deaths includes those as a direct result of chronic alcohol use, such as alcoholic cardiomyopathy, alcoholic hepatitis and cirrhosis, and alcohol-induced pancreatitis, as well as a result of acute use such as alcohol poisoning, suicide by exposure to alcohol, and alcohol-impaired driving fatalities.4
Excessive alcohol consumption poses other serious risks, including cases when intake is abruptly reduced without proper management. Alcohol withdrawal syndrome (AWS) can vary in severity, with potentially life-threatening complications such as hallucinations, seizures, and delirium tremens.5
These risks highlight the importance of professional intervention and support, not only to mitigate risks associated with AWS, but provide a pathway towards recovery from alcohol use disorder (AUD).
According to the 2022 National Survey on Drug Use and Health, 28.8 million US adults had AUD in the prior year, yet only 7.6% of these individuals received treatment and an even smaller group (2.2%) received medication-assisted treatment for alcohol.6,7 This is despite American Psychiatric Association guidelines for the pharmacological treatment of patients with AUD, including the use of naltrexone, acamprosate, disulfiram, topiramate, or gabapentin, depending on therapy goals, past medication trials, medication contraindications, and patient preference.8 Several of these medications are approved by the US Food and Drug Administration (FDA) for the treatment of AUD and have support for effectiveness from randomized controlled trials and meta-analyses.9-11
Clinical practice guidelines for the management of substance use disorders (SUDs) from the US Department of Veterans Affairs (VA) and US Department of Defense have strong recommendations for naltrexone and topiramate as first-line pharmacotherapies for moderate to severe AUD. Acamprosate and disulfiram are weak recommendations as alternative options. Gabapentin is a weak recommendation for cases where first-line treatments are contraindicated or ineffective. The guidelines emphasize the importance of a comprehensive approach to AUD treatment, including psychosocial interventions in addition to pharmacotherapy.12
A 2023 national survey found veterans reported higher alcohol consumption than nonveterans.13 At the end of fiscal year 2023, > 4.4 million veterans—6% of Veterans Health Administration patients—had been diagnosed with AUD.14 However, > 87% of these patients nationally, and 88% of Veterans Integrated Service Network (VISN) 21 patients, were not receiving naltrexone, acamprosate, disulfiram, or topiramate as part of their treatment. The VA Academic Detailing Service (ADS) now includes AUD pharmacotherapy as a campaign focus, highlighting its importance. The ADS is a pharmacy educational outreach program that uses unbiased clinical guidelines to promote aligning prescribing behavior with best practices. Academic detailing methods include speaking with health care practitioners (HCPs), and direct-to-consumer (DTC) patient education.
ADS campaigns include DTC educational handouts. Past ADS projects and research using DTC have demonstrated a significant improvement in outcomes and positively influencing patients’ pharmacotherapy treatment. 15,16 A VA quality improvement project found a positive correlation between the initiation of AUD pharmacotherapy and engagement with mental health care following the distribution of AUD DTC patient education. 17 This project aimed to apply the same principles of prior research to explore the use of DTC across multiple facilities within VISN 21 to increase AUD pharmacotherapy. VISN 21 includes VA facilities and clinics across the Pacific Islands, Nevada, and California and serves about 350,000 veterans.
METHODS
A prospective cohort of VISN 21 veterans with or at high risk for AUD was identified using the VA ADS AUD Dashboard. The cohort included those not on acamprosate, disulfiram, naltrexone, topiramate, or gabapentin for treatment of AUD and had an elevated Alcohol Use Disorder Identification Test-Consumption (AUDIT-C) score of ≥ 6 (high risk) with an AUD diagnosis or ≥ 8 (severe risk) without a diagnosis. The AUDIT-C scores used in the dashboard are supported by the VA AUD clinician guide as the minimum scores when AUD pharmacotherapy should be offered to patients.18 Prescriptions filled outside the VA were not included in this dashboard.
Data and patient information were collected using the VA Corporate Data Warehouse. To be eligible, veterans needed a valid mailing address within the VISN 21 region and a primary care, mental health, or SUD clinician prescriber visit scheduled between October 1, 2023, and January 31, 2024. Veterans were excluded if they were in hospice, had a 1-year mortality risk score > 50% based on their Care Assessment Need (CAN) score, or facility leadership opted out of project involvement. Patients with both severe renal and hepatic impairments were excluded because they were ineligible for AUD pharmacotherapy. However, veterans with either renal or hepatic impairment (but not both) were included, as they could be potential candidates for ≥ 1 AUD pharmacotherapy option.
Initial correspondence with facilities was initiated through local academic detailers. A local champion was identified for the 1 facility without an academic detailer. Facilities could opt in or out of the project. Approval was provided by the local pharmacy and therapeutics committee, pharmacy, primary care, or psychiatry leadership. Approval process and clinician involvement varied by site.
Education
The selected AUD patient education was designed and approved by the national VA ADS (eappendix). The DTC patient education provided general knowledge about alcohol, including what constitutes a standard amount of alcohol, what is considered heavy drinking, risks of heavy drinking, creating a plan with a clinician to reduce and manage withdrawal symptoms, and additional resources. The DTC was accompanied by a cover letter that included a local facility contact number.
A centralized mailing facility was used for all materials. VA Northern California Health Care System provided the funding to cover the cost of postage. The list of veterans to be contacted was updated on a rolling basis and DTC education was mailed 2 weeks prior to their scheduled prescriber visit.
The eligible cohort of 1260 veterans received DTC education. A comparator group of 2048 veterans that did not receive DTC education was obtained retrospectively by using the same inclusion and exclusion criteria with a scheduled primary care, mental health, or SUD HCP visit from October 1, 2022, to January 31, 2023. The outcomes assessed were within 30 days of the scheduled visit, with the primary outcome as the initiation of AUD-related pharmacotherapy and the secondary outcome as the placement of a consultation for mental health or SUD services. Any consultations sent to Behavioral Health, Addiction, Mental Health, Psychiatric, and SUD services following the HCP visit, within the specified time frame, were used for the secondary outcome.
Matching and Analysis
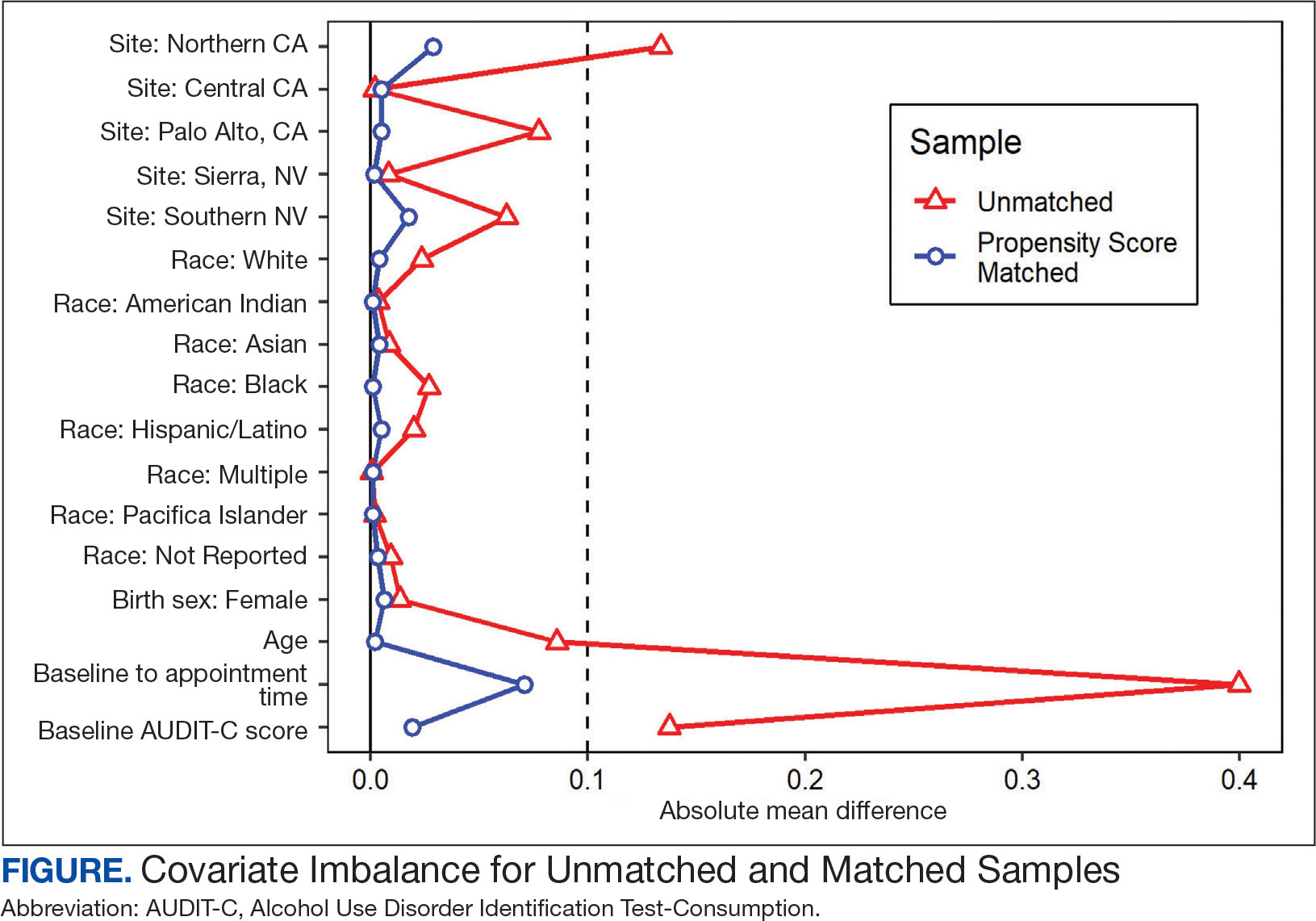
A 1-to-1 nearest neighbor propensity score (PS) matching without replacement was used to pair the 1260 veterans from the intervention group with similarly scored comparator group veterans for a PS-matched final dataset of 2520 veterans. The PS model was a multivariate logistic regression with the outcome being exposure and comparator group status. Baseline characteristics used in the PS model were age, birth sex, race, facility of care, baseline AUDIT-C score, and days between project start and scheduled appointment. Covariate imbalance for the PS-matched sample was assessed to ensure the standardized mean difference for all covariates fell under a 0.1 threshold (Figure).19
A frequency table was provided to compare the discrete distributions of the baseline characteristics in the intervention and comparator groups. Logistic regression analysis was performed to evaluate the association between DTC education exposure and pharmacotherapy initiation, while controlling for potential confounders. Univariate and multivariate P value results for each variable included in the model were reported along with the multivariate odds ratios (ORs) and their associated 95% CIs. Logistic regression analyses were run for both outcomes. Each model included the exposure and comparator group status as well as the baseline characteristics included in the PS model. Statistical significance was set at P < .05. All statistical analyses were performed with R version 4.2.1.
RESULTS
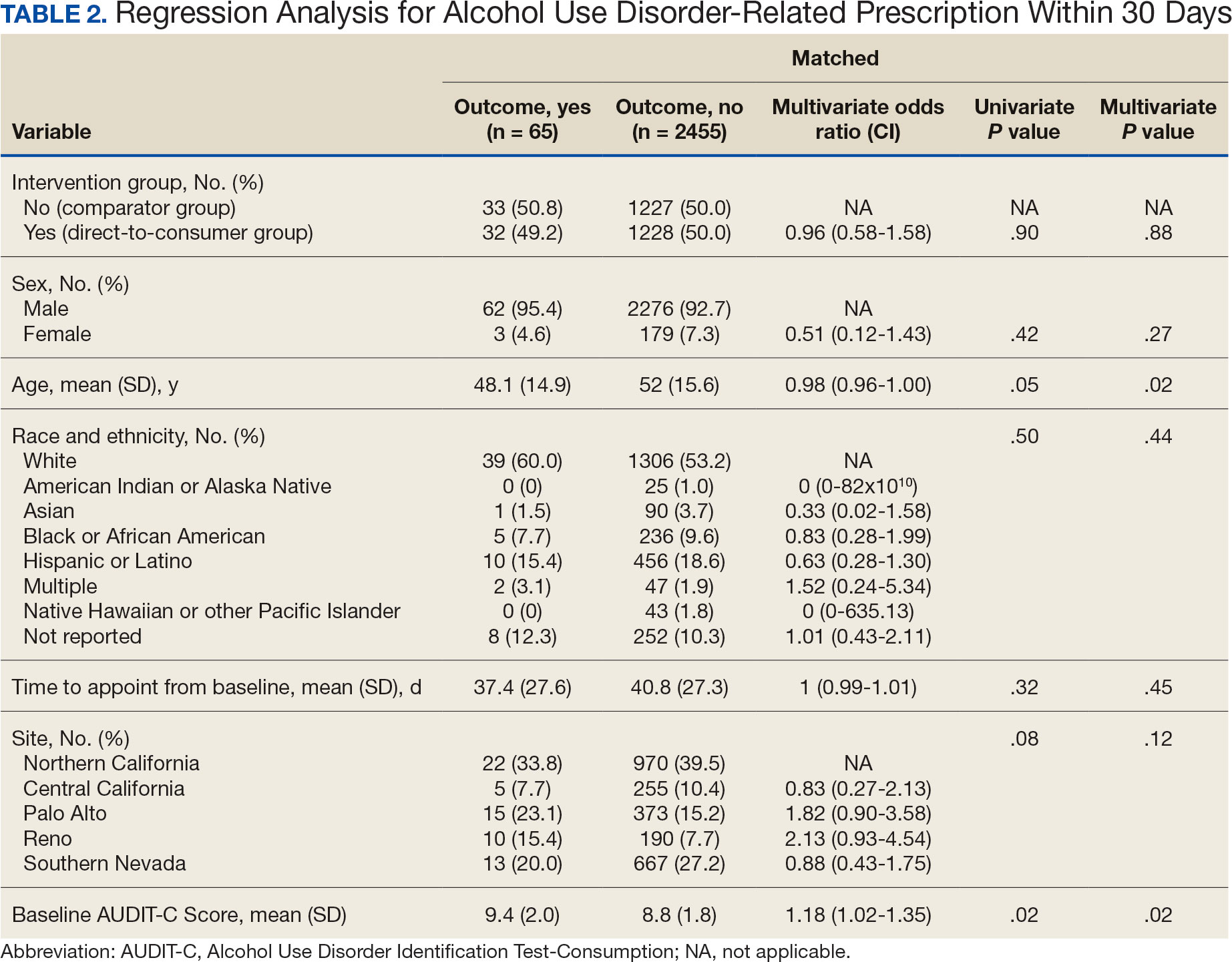
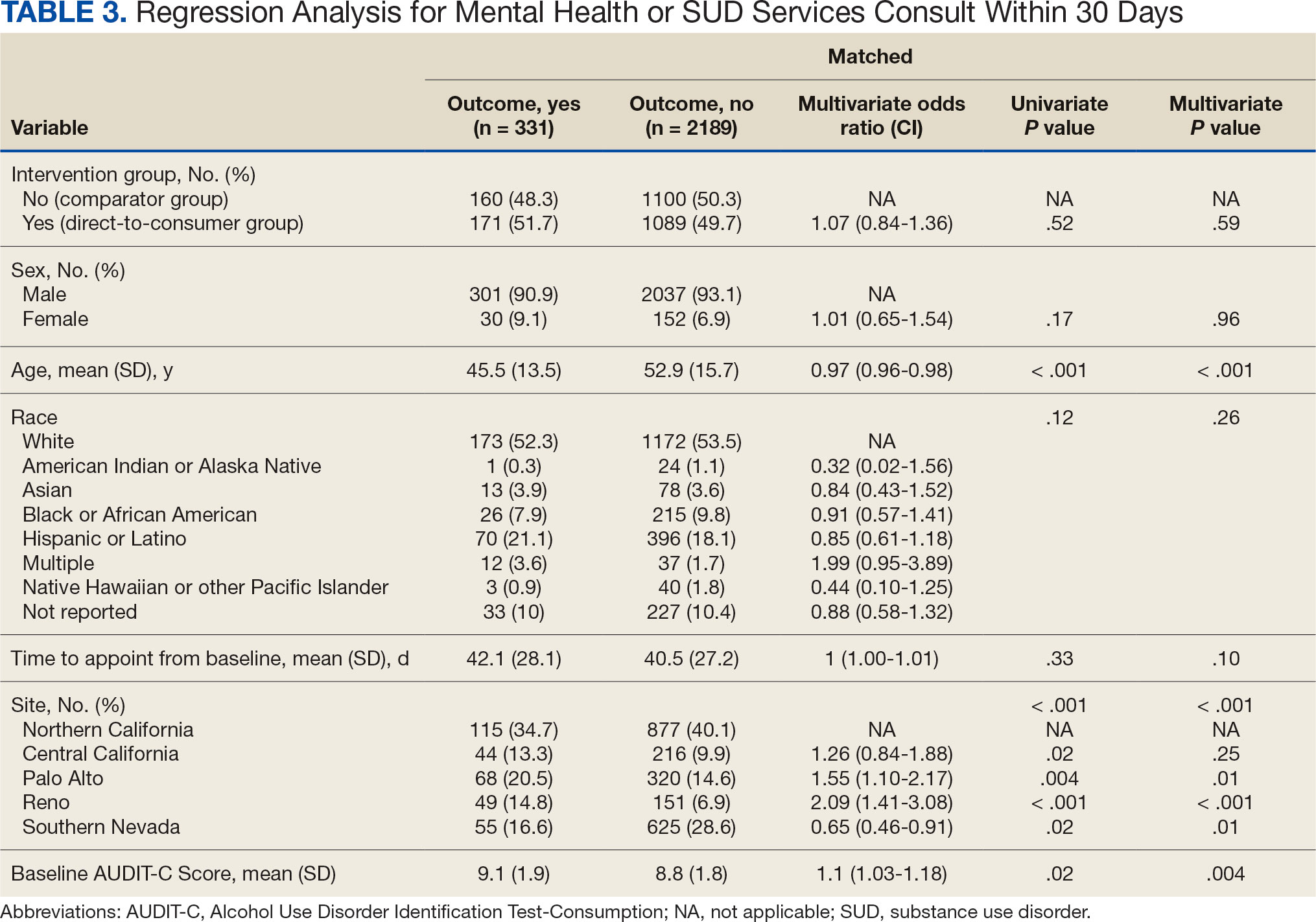
Two of 7 VISN 21 sites did not participate, and 3 had restrictions on participation. DTC education was mailed about 2 weeks prior to scheduled visit for 1260 veterans; 53.6% identified as White, 37.6% were aged 41 to 60 years, and 79.2% had an AUDIT-C ≥ 8 (Table 1). Of those mailed education, there were 173 no-show appointments (13.7%). Thirty-two veterans (2.5%) in the DTC group and 33 veterans (2.6%) in the comparator group received an AUD-related pharmacotherapy prescription (P = .88) (Table 2). One hundred seventy-one veterans (13.6%) in the DTC group and 160 veterans (12.7%) in the comparator group had a consult placed for mental health or SUD services within 30 days of their appointment (P = .59) (Table 3).
DISCUSSION
This project did not yield statistically significant differences in either the primary or secondary outcomes within the 30-day follow-up window and found limited impact from the DTC educational outreach to veterans. The percentage of veterans that received AUD-related pharmacotherapy or consultations for mental health or SUD services was similarly low in the DTC and comparator groups. These findings suggest that although DTC education may raise awareness, it may not be sufficient on its own to drive changes in prescribing behavior or referral patterns without system-level support.
Addiction is a complex disease faced with stigma and requiring readiness by both the HCP and patient to move forward in support and treatment. The consequences of stigma can be severe: the more stigma perceived by a person with AUD, the less likely they are to seek treatment.20 Stigma may exist even within HCPs and may lead to compromised care including shortened visits, less engagement, and less empathy.19 Cultural attitude towards alcohol use and intoxication can also be influenced through a wide range of sources including social media, movies, music, and television. Studies have shown targeted alcohol marketing may result in the development of positive beliefs about drinking and expand environments where alcohol use is socially acceptable and encouraged.21 These factors can impact drinking behavior, including the onset of drinking, binge drinking, and increased alcohol consumption.22
Three VISN 21 sites in this study had restrictions on or excluded primary care from participation. Leadership at some of these facilities were concerned that primary care teams did not have the bandwidth to take on additional items and/or there was variable primary care readiness for initiating AUD pharmacotherapy. Further attempts should be made to integrate primary care into the process of initiating AUD treatment as significant research suggests that integrated care models for AUD may be associated with improved process and outcome measures of care.23
There are several differences between this quality improvement project and prior research investigating the impact of DTC education for other conditions, such as the EMPOWER randomized controlled trial and VISN 22 project, which both demonstrated effectiveness of DTC education for reducing benzodiazepine use in geriatric veterans. 15,16 These studies focused on reducing or stopping pharmacotherapy use, whereas this project sought to promote the initiation of AUD pharmacotherapy. These studies evaluated outcomes at least 6 months postindex date, whereas this project evaluated outcomes within 30 days postappointment. Furthermore, the educational content varied significantly. Other projects provided patients with information focused on specific medications and interventions, such as benzodiazepine tapering, while this project mailed general information on heavy drinking, its risks, and strategies for cutting back, without mentioning pharmacotherapy. The DTC material used in this project was chosen because it was a preapproved national VA ADS resource, which expedited the project timeline by avoiding the need for additional approvals at each participating site. These differences may impact the observed effectiveness of DTC education in this project, especially regarding the primary outcome.
Strengths and Limitations
This quality improvement project sent a large sample of veterans DTC education in a clinical setting across multiple sites. Additionally, PS matching methods were used to balance covariates between the comparator and DTC education group, thereby simulating a randomized controlled trial and reducing selection bias. The project brought attention to the VISN 21 AUD treatment rates, stimulated conversation across sites about available treatments and resources for AUD, and sparked collaboration between academic detailing, mental health, and primary care services. The time frame for visits was selected during the winter; the National Institute on Alcohol Abuse and Alcoholism notes this is a time when people may be more likely to engage in excessive alcohol consumption than at other times of the year.24
The 30-day time frame for outcomes may have been too short to observe changes in prescribing or referral patterns. Additionally, the comparator group was comprised of veterans seen from October 1, 2022, to January 31, 2023, where seasonal timing may have influenced alcohol consumption behaviors and skewed the results. There were also no-show appointments in the DTC education group (13.7%), though it is likely some patients rescheduled and still received AUD pharmacotherapy within 30 days of the original appointment. Finally, it was not possible to confirm whether a patient opened and read the education that was mailed to them. This may be another reason to explore electronic distribution of DTC education. This all may have contributed to the lack of statistically significant differences in both the primary and secondary outcomes.
There was a high level of variability between facility participation in the project. Two of 7 sites did not participate, and 3 sites restricted primary care engagement. This represents a significant limitation, particularly for the secondary outcome of placing consultations for MH or SUD services. Facilities that only included mental health or SUD HCPs may have resulted in lower consultation rates due to their inherent specialization, reducing the likelihood of self-referrals.
The project may overestimate prescribed AUD pharmacotherapy in the primary outcome due to potential misclassification of medications. While the project adhered to the national VA ADS AUD dashboard’s definition of AUD pharmacotherapy, including acamprosate, disulfiram, naltrexone, topiramate, and gabapentin, some of these medications have multiple indications. For example, gabapentin is commonly prescribed for peripheral neuropathy, and topiramate is used to treat migraines and seizures. The multipurpose use adds uncertainty about whether they were prescribed specifically for AUD treatment, especially in cases where the HCP is responsible for treating a broad range of disease states, as in primary care.
CONCLUSIONS
Results of this quality improvement project did not show a statistically significant difference between patients sent DTC education and the comparator group for the initiation of AUD pharmacotherapy or placement of a consult to mental health or SUD services within 30 days of their scheduled visit. Future studies may seek to implement stricter criteria to confirm the intended use of topiramate and gabapentin, such as looking for keywords in the prescription instructions for use, performing chart reviews, and/or only including these medications if prescribed by a mental health or SUD HCP. Alternatively, future studies may consider limiting the analysis to only FDA-approved AUD medications: acamprosate, disulfiram, and naltrexone. It is vital to continue to enhance primary care HCP readiness to treat AUD, given the existing relationships and trust they often have with patients. Electronic methods for distributing DTC education could also be advantageous, as these methods may have the ability to track whether a message has been opened and read. Despite a lack of statistical significance, this project sparked crucial conversations and collaboration around AUD, available treatments, and addressing potential barriers to connecting patients to care within VISN 21.
- Centers for Disease Control and Prevention. Facts about U.S. deaths from excessive alcohol use. August 6, 2024. Accessed February 5, 2025. https://www.cdc.gov/alcohol/facts-stats/
- State Health Access Data Assistance Center. Escalating alcohol-involved death rates: trends and variation across the nation and in the states from 2006 to 2019. April 19, 2021. Accessed February 5, 2025. https://www.shadac.org/escalating-alcohol-involved-death-rates-trends-and-variation-across-nation-and-states-2006-2019
- National Institute on Alcohol Abuse and Alcoholism. Alcohol- related emergencies and deaths in the United States. Updated November 2024. Accessed February 5, 2025. https://www.niaaa.nih.gov/alcohols-effects-health/alcohol-topics/alcohol-facts-and-statistics/alcohol-related-emergencies-and-deaths-united-states
- Esser MB, Sherk A, Liu Y, Naimi TS. Deaths from excessive alcohol use - United States, 2016- 2021. MMWR Morb Mortal Wkly Rep. 2024;73(8):154-161. doi:10.15585/mmwr.mm7308a1
- Canver BR, Newman RK, Gomez AE. Alcohol Withdrawal Syndrome. In: StatPearls. StatPearls Publishing; 2024.
- National Institute on Alcohol Abuse and Alcoholism. Alcohol treatment in the United States. Updated January 2025. Accessed February 5, 2025. https://www.niaaa.nih.gov/alcohols-effects-health/alcohol-topics/alcohol-facts-and-statistics/alcohol-treatment-united-states
- National Institute on Alcohol Abuse and Alcoholism. Alcohol use disorder (AUD) in the United States: age groups and demographic characteristics. Updated September 2024. Accessed February 5, 2025. https://www.niaaa.nih.gov/alcohols-effects-health/alcohol-topics/alcohol-facts-and-statistics/alcohol-use-disorder-aud-united-states-age-groups-and-demographic-characteristics
- Reus VI, Fochtmann LJ, Bukstein O, et al. The American Psychiatric Association practice guideline for the pharmacological treatment of patients with alcohol use disorder. Am J Psychiatry. 2018;175(1):86-90. doi:10.1176/appi.ajp.2017.1750101
- Blodgett JC, Del Re AC, Maisel NC, Finney JW. A meta-analysis of topiramate’s effects for individuals with alcohol use disorders. Alcohol Clin Exp Res. 2014;38(6):1481-1488. doi:10.1111/acer.12411
- Maisel NC, Blodgett JC, Wilbourne PL, Humphreys K, Finney JW. Meta-analysis of naltrexone and acamprosate for treating alcohol use disorders: when are these medications most helpful? Addiction. 2013;108(2):275-293. doi:10.1111/j.1360-0443.2012.04054.x
- Jonas DE, Amick HR, Feltner C, et al. Pharmacotherapy for adults with alcohol use disorders in outpatient settings: a systematic review and meta-analysis. JAMA. 2014;311(18):1889-1900. doi:10.1001/jama.2014.3628
- US Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the management of substance use disorders. August 2021. Accessed February 5, 2025. https://www.healthquality.va.gov/guidelines/MH/sud/VADODSUDCPG.pdf
- Ranney RM, Bernhard PA, Vogt D, et al. Alcohol use and treatment utilization in a national sample of veterans and nonveterans. J Subst Use Addict Treat. 2023;146:208964. doi:10.1016/j.josat.2023.208964
- US Department of Veterans Affairs, Pharmacy Benefit Management Service, Academic Detailing Service. AUD Trend Report. https://vaww.pbi.cdw.va.gov/PBIRS/Pages/ReportViewer.aspx?/GPE/PBM_AD/SSRS/AUD/AUD_TrendReport
- Mendes MA, Smith JP, Marin JK, et al. Reducing benzodiazepine prescribing in older veterans: a direct-to-consumer educational brochure. Fed Pract. 2018;35(9):36-43.
- Tannenbaum C, Martin P, Tamblyn R, Benedetti A, Ahmed S. Reduction of inappropriate benzodiazepine prescriptions among older adults through direct patient education: the EMPOWER cluster randomized trial. JAMA Intern Med. 2014;174(6):890-898. doi:10.1001/jamainternmed.2014.949
- Maloney R, Funmilayo M. Acting on the AUDIT-C: implementation of direct-to-consumer education on unhealth alcohol use. Presented on March 31, 2023; Central Virginia Veterans Affairs Health Care System, Richmond, Virginia.
- US Department of Veterans Affairs, Pharmacy Benefit Management Service. Alcohol use disorder (AUD) – leading the charge in the treatment of AUD: a VA clinician’s guide. February 2022. Accessed February 5, 2025. https://www.pbm.va.gov/PBM/AcademicDetailingService/Documents/508/10-1530_AUD_ClinicianGuide_508Conformant.pdf
- Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011;46(3):399-424. doi:10.1080/00273171.2011.568786
- National Institute on Alcohol Abuse and Alcoholism. Stigma: overcoming a pervasive barrier to optimal care. Updated January 6, 2025. Accessed February 5, 2025. https://www.niaaa.nih.gov/health-professionals-communities/core-resource-on-alcohol/stigma-overcoming-pervasive-barrier-optimal-care
- Sudhinaraset M, Wigglesworth C, Takeuchi DT. Social and cultural contexts of alcohol use: influences in a socialecological framework. Alcohol Res. 2016;38(1):35-45.
- Tanski SE, McClure AC, Li Z, et al. Cued recall of alcohol advertising on television and underage drinking behavior. JAMA Pediatr. 2015;169(3):264-271. doi:10.1001/jamapediatrics.2014.3345
- Hyland CJ, McDowell MJ, Bain PA, Huskamp HA, Busch AB. Integration of pharmacotherapy for alcohol use disorder treatment in primary care settings: a scoping review. J Subst Abuse Treat. 2023;144:108919. doi:10.1016/j.jsat.2022.108919
- National Institute on Alcohol Abuse and Alcoholism. The truth about holiday spirits. Updated November 2023. Accessed February 5, 2025. ,a href="https://www.niaaa.nih.gov/publications/brochures-and-fact-sheets/truth-about-holiday-spirits">https://www.niaaa.nih.gov/publications/brochures-and-fact-sheets/truth-about-holiday-spirits
Excessive alcohol use is one of the leading preventable causes of death in the United States, responsible for about 178,000 deaths annually and an average of 488 daily deaths in 2020 and 2021.1Alcohol-related deaths increased by 49% between 2006 and 2019.2 This trend continued during the COVID-19 pandemic, with death certificates that listed alcohol increasing by > 25% from 2019 to 2020, and another 10% in 2021.3 This increase of alcohol-related deaths includes those as a direct result of chronic alcohol use, such as alcoholic cardiomyopathy, alcoholic hepatitis and cirrhosis, and alcohol-induced pancreatitis, as well as a result of acute use such as alcohol poisoning, suicide by exposure to alcohol, and alcohol-impaired driving fatalities.4
Excessive alcohol consumption poses other serious risks, including cases when intake is abruptly reduced without proper management. Alcohol withdrawal syndrome (AWS) can vary in severity, with potentially life-threatening complications such as hallucinations, seizures, and delirium tremens.5
These risks highlight the importance of professional intervention and support, not only to mitigate risks associated with AWS, but provide a pathway towards recovery from alcohol use disorder (AUD).
According to the 2022 National Survey on Drug Use and Health, 28.8 million US adults had AUD in the prior year, yet only 7.6% of these individuals received treatment and an even smaller group (2.2%) received medication-assisted treatment for alcohol.6,7 This is despite American Psychiatric Association guidelines for the pharmacological treatment of patients with AUD, including the use of naltrexone, acamprosate, disulfiram, topiramate, or gabapentin, depending on therapy goals, past medication trials, medication contraindications, and patient preference.8 Several of these medications are approved by the US Food and Drug Administration (FDA) for the treatment of AUD and have support for effectiveness from randomized controlled trials and meta-analyses.9-11
Clinical practice guidelines for the management of substance use disorders (SUDs) from the US Department of Veterans Affairs (VA) and US Department of Defense have strong recommendations for naltrexone and topiramate as first-line pharmacotherapies for moderate to severe AUD. Acamprosate and disulfiram are weak recommendations as alternative options. Gabapentin is a weak recommendation for cases where first-line treatments are contraindicated or ineffective. The guidelines emphasize the importance of a comprehensive approach to AUD treatment, including psychosocial interventions in addition to pharmacotherapy.12
A 2023 national survey found veterans reported higher alcohol consumption than nonveterans.13 At the end of fiscal year 2023, > 4.4 million veterans—6% of Veterans Health Administration patients—had been diagnosed with AUD.14 However, > 87% of these patients nationally, and 88% of Veterans Integrated Service Network (VISN) 21 patients, were not receiving naltrexone, acamprosate, disulfiram, or topiramate as part of their treatment. The VA Academic Detailing Service (ADS) now includes AUD pharmacotherapy as a campaign focus, highlighting its importance. The ADS is a pharmacy educational outreach program that uses unbiased clinical guidelines to promote aligning prescribing behavior with best practices. Academic detailing methods include speaking with health care practitioners (HCPs), and direct-to-consumer (DTC) patient education.
ADS campaigns include DTC educational handouts. Past ADS projects and research using DTC have demonstrated a significant improvement in outcomes and positively influencing patients’ pharmacotherapy treatment. 15,16 A VA quality improvement project found a positive correlation between the initiation of AUD pharmacotherapy and engagement with mental health care following the distribution of AUD DTC patient education. 17 This project aimed to apply the same principles of prior research to explore the use of DTC across multiple facilities within VISN 21 to increase AUD pharmacotherapy. VISN 21 includes VA facilities and clinics across the Pacific Islands, Nevada, and California and serves about 350,000 veterans.
METHODS
A prospective cohort of VISN 21 veterans with or at high risk for AUD was identified using the VA ADS AUD Dashboard. The cohort included those not on acamprosate, disulfiram, naltrexone, topiramate, or gabapentin for treatment of AUD and had an elevated Alcohol Use Disorder Identification Test-Consumption (AUDIT-C) score of ≥ 6 (high risk) with an AUD diagnosis or ≥ 8 (severe risk) without a diagnosis. The AUDIT-C scores used in the dashboard are supported by the VA AUD clinician guide as the minimum scores when AUD pharmacotherapy should be offered to patients.18 Prescriptions filled outside the VA were not included in this dashboard.
Data and patient information were collected using the VA Corporate Data Warehouse. To be eligible, veterans needed a valid mailing address within the VISN 21 region and a primary care, mental health, or SUD clinician prescriber visit scheduled between October 1, 2023, and January 31, 2024. Veterans were excluded if they were in hospice, had a 1-year mortality risk score > 50% based on their Care Assessment Need (CAN) score, or facility leadership opted out of project involvement. Patients with both severe renal and hepatic impairments were excluded because they were ineligible for AUD pharmacotherapy. However, veterans with either renal or hepatic impairment (but not both) were included, as they could be potential candidates for ≥ 1 AUD pharmacotherapy option.
Initial correspondence with facilities was initiated through local academic detailers. A local champion was identified for the 1 facility without an academic detailer. Facilities could opt in or out of the project. Approval was provided by the local pharmacy and therapeutics committee, pharmacy, primary care, or psychiatry leadership. Approval process and clinician involvement varied by site.
Education
The selected AUD patient education was designed and approved by the national VA ADS (eappendix). The DTC patient education provided general knowledge about alcohol, including what constitutes a standard amount of alcohol, what is considered heavy drinking, risks of heavy drinking, creating a plan with a clinician to reduce and manage withdrawal symptoms, and additional resources. The DTC was accompanied by a cover letter that included a local facility contact number.
A centralized mailing facility was used for all materials. VA Northern California Health Care System provided the funding to cover the cost of postage. The list of veterans to be contacted was updated on a rolling basis and DTC education was mailed 2 weeks prior to their scheduled prescriber visit.
The eligible cohort of 1260 veterans received DTC education. A comparator group of 2048 veterans that did not receive DTC education was obtained retrospectively by using the same inclusion and exclusion criteria with a scheduled primary care, mental health, or SUD HCP visit from October 1, 2022, to January 31, 2023. The outcomes assessed were within 30 days of the scheduled visit, with the primary outcome as the initiation of AUD-related pharmacotherapy and the secondary outcome as the placement of a consultation for mental health or SUD services. Any consultations sent to Behavioral Health, Addiction, Mental Health, Psychiatric, and SUD services following the HCP visit, within the specified time frame, were used for the secondary outcome.
Matching and Analysis
A 1-to-1 nearest neighbor propensity score (PS) matching without replacement was used to pair the 1260 veterans from the intervention group with similarly scored comparator group veterans for a PS-matched final dataset of 2520 veterans. The PS model was a multivariate logistic regression with the outcome being exposure and comparator group status. Baseline characteristics used in the PS model were age, birth sex, race, facility of care, baseline AUDIT-C score, and days between project start and scheduled appointment. Covariate imbalance for the PS-matched sample was assessed to ensure the standardized mean difference for all covariates fell under a 0.1 threshold (Figure).19

A frequency table was provided to compare the discrete distributions of the baseline characteristics in the intervention and comparator groups. Logistic regression analysis was performed to evaluate the association between DTC education exposure and pharmacotherapy initiation, while controlling for potential confounders. Univariate and multivariate P value results for each variable included in the model were reported along with the multivariate odds ratios (ORs) and their associated 95% CIs. Logistic regression analyses were run for both outcomes. Each model included the exposure and comparator group status as well as the baseline characteristics included in the PS model. Statistical significance was set at P < .05. All statistical analyses were performed with R version 4.2.1.
RESULTS
Two of 7 VISN 21 sites did not participate, and 3 had restrictions on participation. DTC education was mailed about 2 weeks prior to scheduled visit for 1260 veterans; 53.6% identified as White, 37.6% were aged 41 to 60 years, and 79.2% had an AUDIT-C ≥ 8 (Table 1). Of those mailed education, there were 173 no-show appointments (13.7%). Thirty-two veterans (2.5%) in the DTC group and 33 veterans (2.6%) in the comparator group received an AUD-related pharmacotherapy prescription (P = .88) (Table 2). One hundred seventy-one veterans (13.6%) in the DTC group and 160 veterans (12.7%) in the comparator group had a consult placed for mental health or SUD services within 30 days of their appointment (P = .59) (Table 3).



DISCUSSION
This project did not yield statistically significant differences in either the primary or secondary outcomes within the 30-day follow-up window and found limited impact from the DTC educational outreach to veterans. The percentage of veterans that received AUD-related pharmacotherapy or consultations for mental health or SUD services was similarly low in the DTC and comparator groups. These findings suggest that although DTC education may raise awareness, it may not be sufficient on its own to drive changes in prescribing behavior or referral patterns without system-level support.
Addiction is a complex disease faced with stigma and requiring readiness by both the HCP and patient to move forward in support and treatment. The consequences of stigma can be severe: the more stigma perceived by a person with AUD, the less likely they are to seek treatment.20 Stigma may exist even within HCPs and may lead to compromised care including shortened visits, less engagement, and less empathy.19 Cultural attitude towards alcohol use and intoxication can also be influenced through a wide range of sources including social media, movies, music, and television. Studies have shown targeted alcohol marketing may result in the development of positive beliefs about drinking and expand environments where alcohol use is socially acceptable and encouraged.21 These factors can impact drinking behavior, including the onset of drinking, binge drinking, and increased alcohol consumption.22
Three VISN 21 sites in this study had restrictions on or excluded primary care from participation. Leadership at some of these facilities were concerned that primary care teams did not have the bandwidth to take on additional items and/or there was variable primary care readiness for initiating AUD pharmacotherapy. Further attempts should be made to integrate primary care into the process of initiating AUD treatment as significant research suggests that integrated care models for AUD may be associated with improved process and outcome measures of care.23
There are several differences between this quality improvement project and prior research investigating the impact of DTC education for other conditions, such as the EMPOWER randomized controlled trial and VISN 22 project, which both demonstrated effectiveness of DTC education for reducing benzodiazepine use in geriatric veterans. 15,16 These studies focused on reducing or stopping pharmacotherapy use, whereas this project sought to promote the initiation of AUD pharmacotherapy. These studies evaluated outcomes at least 6 months postindex date, whereas this project evaluated outcomes within 30 days postappointment. Furthermore, the educational content varied significantly. Other projects provided patients with information focused on specific medications and interventions, such as benzodiazepine tapering, while this project mailed general information on heavy drinking, its risks, and strategies for cutting back, without mentioning pharmacotherapy. The DTC material used in this project was chosen because it was a preapproved national VA ADS resource, which expedited the project timeline by avoiding the need for additional approvals at each participating site. These differences may impact the observed effectiveness of DTC education in this project, especially regarding the primary outcome.
Strengths and Limitations
This quality improvement project sent a large sample of veterans DTC education in a clinical setting across multiple sites. Additionally, PS matching methods were used to balance covariates between the comparator and DTC education group, thereby simulating a randomized controlled trial and reducing selection bias. The project brought attention to the VISN 21 AUD treatment rates, stimulated conversation across sites about available treatments and resources for AUD, and sparked collaboration between academic detailing, mental health, and primary care services. The time frame for visits was selected during the winter; the National Institute on Alcohol Abuse and Alcoholism notes this is a time when people may be more likely to engage in excessive alcohol consumption than at other times of the year.24
The 30-day time frame for outcomes may have been too short to observe changes in prescribing or referral patterns. Additionally, the comparator group was comprised of veterans seen from October 1, 2022, to January 31, 2023, where seasonal timing may have influenced alcohol consumption behaviors and skewed the results. There were also no-show appointments in the DTC education group (13.7%), though it is likely some patients rescheduled and still received AUD pharmacotherapy within 30 days of the original appointment. Finally, it was not possible to confirm whether a patient opened and read the education that was mailed to them. This may be another reason to explore electronic distribution of DTC education. This all may have contributed to the lack of statistically significant differences in both the primary and secondary outcomes.
There was a high level of variability between facility participation in the project. Two of 7 sites did not participate, and 3 sites restricted primary care engagement. This represents a significant limitation, particularly for the secondary outcome of placing consultations for MH or SUD services. Facilities that only included mental health or SUD HCPs may have resulted in lower consultation rates due to their inherent specialization, reducing the likelihood of self-referrals.
The project may overestimate prescribed AUD pharmacotherapy in the primary outcome due to potential misclassification of medications. While the project adhered to the national VA ADS AUD dashboard’s definition of AUD pharmacotherapy, including acamprosate, disulfiram, naltrexone, topiramate, and gabapentin, some of these medications have multiple indications. For example, gabapentin is commonly prescribed for peripheral neuropathy, and topiramate is used to treat migraines and seizures. The multipurpose use adds uncertainty about whether they were prescribed specifically for AUD treatment, especially in cases where the HCP is responsible for treating a broad range of disease states, as in primary care.
CONCLUSIONS
Results of this quality improvement project did not show a statistically significant difference between patients sent DTC education and the comparator group for the initiation of AUD pharmacotherapy or placement of a consult to mental health or SUD services within 30 days of their scheduled visit. Future studies may seek to implement stricter criteria to confirm the intended use of topiramate and gabapentin, such as looking for keywords in the prescription instructions for use, performing chart reviews, and/or only including these medications if prescribed by a mental health or SUD HCP. Alternatively, future studies may consider limiting the analysis to only FDA-approved AUD medications: acamprosate, disulfiram, and naltrexone. It is vital to continue to enhance primary care HCP readiness to treat AUD, given the existing relationships and trust they often have with patients. Electronic methods for distributing DTC education could also be advantageous, as these methods may have the ability to track whether a message has been opened and read. Despite a lack of statistical significance, this project sparked crucial conversations and collaboration around AUD, available treatments, and addressing potential barriers to connecting patients to care within VISN 21.
Excessive alcohol use is one of the leading preventable causes of death in the United States, responsible for about 178,000 deaths annually and an average of 488 daily deaths in 2020 and 2021.1Alcohol-related deaths increased by 49% between 2006 and 2019.2 This trend continued during the COVID-19 pandemic, with death certificates that listed alcohol increasing by > 25% from 2019 to 2020, and another 10% in 2021.3 This increase of alcohol-related deaths includes those as a direct result of chronic alcohol use, such as alcoholic cardiomyopathy, alcoholic hepatitis and cirrhosis, and alcohol-induced pancreatitis, as well as a result of acute use such as alcohol poisoning, suicide by exposure to alcohol, and alcohol-impaired driving fatalities.4
Excessive alcohol consumption poses other serious risks, including cases when intake is abruptly reduced without proper management. Alcohol withdrawal syndrome (AWS) can vary in severity, with potentially life-threatening complications such as hallucinations, seizures, and delirium tremens.5
These risks highlight the importance of professional intervention and support, not only to mitigate risks associated with AWS, but provide a pathway towards recovery from alcohol use disorder (AUD).
According to the 2022 National Survey on Drug Use and Health, 28.8 million US adults had AUD in the prior year, yet only 7.6% of these individuals received treatment and an even smaller group (2.2%) received medication-assisted treatment for alcohol.6,7 This is despite American Psychiatric Association guidelines for the pharmacological treatment of patients with AUD, including the use of naltrexone, acamprosate, disulfiram, topiramate, or gabapentin, depending on therapy goals, past medication trials, medication contraindications, and patient preference.8 Several of these medications are approved by the US Food and Drug Administration (FDA) for the treatment of AUD and have support for effectiveness from randomized controlled trials and meta-analyses.9-11
Clinical practice guidelines for the management of substance use disorders (SUDs) from the US Department of Veterans Affairs (VA) and US Department of Defense have strong recommendations for naltrexone and topiramate as first-line pharmacotherapies for moderate to severe AUD. Acamprosate and disulfiram are weak recommendations as alternative options. Gabapentin is a weak recommendation for cases where first-line treatments are contraindicated or ineffective. The guidelines emphasize the importance of a comprehensive approach to AUD treatment, including psychosocial interventions in addition to pharmacotherapy.12
A 2023 national survey found veterans reported higher alcohol consumption than nonveterans.13 At the end of fiscal year 2023, > 4.4 million veterans—6% of Veterans Health Administration patients—had been diagnosed with AUD.14 However, > 87% of these patients nationally, and 88% of Veterans Integrated Service Network (VISN) 21 patients, were not receiving naltrexone, acamprosate, disulfiram, or topiramate as part of their treatment. The VA Academic Detailing Service (ADS) now includes AUD pharmacotherapy as a campaign focus, highlighting its importance. The ADS is a pharmacy educational outreach program that uses unbiased clinical guidelines to promote aligning prescribing behavior with best practices. Academic detailing methods include speaking with health care practitioners (HCPs), and direct-to-consumer (DTC) patient education.
ADS campaigns include DTC educational handouts. Past ADS projects and research using DTC have demonstrated a significant improvement in outcomes and positively influencing patients’ pharmacotherapy treatment. 15,16 A VA quality improvement project found a positive correlation between the initiation of AUD pharmacotherapy and engagement with mental health care following the distribution of AUD DTC patient education. 17 This project aimed to apply the same principles of prior research to explore the use of DTC across multiple facilities within VISN 21 to increase AUD pharmacotherapy. VISN 21 includes VA facilities and clinics across the Pacific Islands, Nevada, and California and serves about 350,000 veterans.
METHODS
A prospective cohort of VISN 21 veterans with or at high risk for AUD was identified using the VA ADS AUD Dashboard. The cohort included those not on acamprosate, disulfiram, naltrexone, topiramate, or gabapentin for treatment of AUD and had an elevated Alcohol Use Disorder Identification Test-Consumption (AUDIT-C) score of ≥ 6 (high risk) with an AUD diagnosis or ≥ 8 (severe risk) without a diagnosis. The AUDIT-C scores used in the dashboard are supported by the VA AUD clinician guide as the minimum scores when AUD pharmacotherapy should be offered to patients.18 Prescriptions filled outside the VA were not included in this dashboard.
Data and patient information were collected using the VA Corporate Data Warehouse. To be eligible, veterans needed a valid mailing address within the VISN 21 region and a primary care, mental health, or SUD clinician prescriber visit scheduled between October 1, 2023, and January 31, 2024. Veterans were excluded if they were in hospice, had a 1-year mortality risk score > 50% based on their Care Assessment Need (CAN) score, or facility leadership opted out of project involvement. Patients with both severe renal and hepatic impairments were excluded because they were ineligible for AUD pharmacotherapy. However, veterans with either renal or hepatic impairment (but not both) were included, as they could be potential candidates for ≥ 1 AUD pharmacotherapy option.
Initial correspondence with facilities was initiated through local academic detailers. A local champion was identified for the 1 facility without an academic detailer. Facilities could opt in or out of the project. Approval was provided by the local pharmacy and therapeutics committee, pharmacy, primary care, or psychiatry leadership. Approval process and clinician involvement varied by site.
Education
The selected AUD patient education was designed and approved by the national VA ADS (eappendix). The DTC patient education provided general knowledge about alcohol, including what constitutes a standard amount of alcohol, what is considered heavy drinking, risks of heavy drinking, creating a plan with a clinician to reduce and manage withdrawal symptoms, and additional resources. The DTC was accompanied by a cover letter that included a local facility contact number.
A centralized mailing facility was used for all materials. VA Northern California Health Care System provided the funding to cover the cost of postage. The list of veterans to be contacted was updated on a rolling basis and DTC education was mailed 2 weeks prior to their scheduled prescriber visit.
The eligible cohort of 1260 veterans received DTC education. A comparator group of 2048 veterans that did not receive DTC education was obtained retrospectively by using the same inclusion and exclusion criteria with a scheduled primary care, mental health, or SUD HCP visit from October 1, 2022, to January 31, 2023. The outcomes assessed were within 30 days of the scheduled visit, with the primary outcome as the initiation of AUD-related pharmacotherapy and the secondary outcome as the placement of a consultation for mental health or SUD services. Any consultations sent to Behavioral Health, Addiction, Mental Health, Psychiatric, and SUD services following the HCP visit, within the specified time frame, were used for the secondary outcome.
Matching and Analysis
A 1-to-1 nearest neighbor propensity score (PS) matching without replacement was used to pair the 1260 veterans from the intervention group with similarly scored comparator group veterans for a PS-matched final dataset of 2520 veterans. The PS model was a multivariate logistic regression with the outcome being exposure and comparator group status. Baseline characteristics used in the PS model were age, birth sex, race, facility of care, baseline AUDIT-C score, and days between project start and scheduled appointment. Covariate imbalance for the PS-matched sample was assessed to ensure the standardized mean difference for all covariates fell under a 0.1 threshold (Figure).19

A frequency table was provided to compare the discrete distributions of the baseline characteristics in the intervention and comparator groups. Logistic regression analysis was performed to evaluate the association between DTC education exposure and pharmacotherapy initiation, while controlling for potential confounders. Univariate and multivariate P value results for each variable included in the model were reported along with the multivariate odds ratios (ORs) and their associated 95% CIs. Logistic regression analyses were run for both outcomes. Each model included the exposure and comparator group status as well as the baseline characteristics included in the PS model. Statistical significance was set at P < .05. All statistical analyses were performed with R version 4.2.1.
RESULTS
Two of 7 VISN 21 sites did not participate, and 3 had restrictions on participation. DTC education was mailed about 2 weeks prior to scheduled visit for 1260 veterans; 53.6% identified as White, 37.6% were aged 41 to 60 years, and 79.2% had an AUDIT-C ≥ 8 (Table 1). Of those mailed education, there were 173 no-show appointments (13.7%). Thirty-two veterans (2.5%) in the DTC group and 33 veterans (2.6%) in the comparator group received an AUD-related pharmacotherapy prescription (P = .88) (Table 2). One hundred seventy-one veterans (13.6%) in the DTC group and 160 veterans (12.7%) in the comparator group had a consult placed for mental health or SUD services within 30 days of their appointment (P = .59) (Table 3).



DISCUSSION
This project did not yield statistically significant differences in either the primary or secondary outcomes within the 30-day follow-up window and found limited impact from the DTC educational outreach to veterans. The percentage of veterans that received AUD-related pharmacotherapy or consultations for mental health or SUD services was similarly low in the DTC and comparator groups. These findings suggest that although DTC education may raise awareness, it may not be sufficient on its own to drive changes in prescribing behavior or referral patterns without system-level support.
Addiction is a complex disease faced with stigma and requiring readiness by both the HCP and patient to move forward in support and treatment. The consequences of stigma can be severe: the more stigma perceived by a person with AUD, the less likely they are to seek treatment.20 Stigma may exist even within HCPs and may lead to compromised care including shortened visits, less engagement, and less empathy.19 Cultural attitude towards alcohol use and intoxication can also be influenced through a wide range of sources including social media, movies, music, and television. Studies have shown targeted alcohol marketing may result in the development of positive beliefs about drinking and expand environments where alcohol use is socially acceptable and encouraged.21 These factors can impact drinking behavior, including the onset of drinking, binge drinking, and increased alcohol consumption.22
Three VISN 21 sites in this study had restrictions on or excluded primary care from participation. Leadership at some of these facilities were concerned that primary care teams did not have the bandwidth to take on additional items and/or there was variable primary care readiness for initiating AUD pharmacotherapy. Further attempts should be made to integrate primary care into the process of initiating AUD treatment as significant research suggests that integrated care models for AUD may be associated with improved process and outcome measures of care.23
There are several differences between this quality improvement project and prior research investigating the impact of DTC education for other conditions, such as the EMPOWER randomized controlled trial and VISN 22 project, which both demonstrated effectiveness of DTC education for reducing benzodiazepine use in geriatric veterans. 15,16 These studies focused on reducing or stopping pharmacotherapy use, whereas this project sought to promote the initiation of AUD pharmacotherapy. These studies evaluated outcomes at least 6 months postindex date, whereas this project evaluated outcomes within 30 days postappointment. Furthermore, the educational content varied significantly. Other projects provided patients with information focused on specific medications and interventions, such as benzodiazepine tapering, while this project mailed general information on heavy drinking, its risks, and strategies for cutting back, without mentioning pharmacotherapy. The DTC material used in this project was chosen because it was a preapproved national VA ADS resource, which expedited the project timeline by avoiding the need for additional approvals at each participating site. These differences may impact the observed effectiveness of DTC education in this project, especially regarding the primary outcome.
Strengths and Limitations
This quality improvement project sent a large sample of veterans DTC education in a clinical setting across multiple sites. Additionally, PS matching methods were used to balance covariates between the comparator and DTC education group, thereby simulating a randomized controlled trial and reducing selection bias. The project brought attention to the VISN 21 AUD treatment rates, stimulated conversation across sites about available treatments and resources for AUD, and sparked collaboration between academic detailing, mental health, and primary care services. The time frame for visits was selected during the winter; the National Institute on Alcohol Abuse and Alcoholism notes this is a time when people may be more likely to engage in excessive alcohol consumption than at other times of the year.24
The 30-day time frame for outcomes may have been too short to observe changes in prescribing or referral patterns. Additionally, the comparator group was comprised of veterans seen from October 1, 2022, to January 31, 2023, where seasonal timing may have influenced alcohol consumption behaviors and skewed the results. There were also no-show appointments in the DTC education group (13.7%), though it is likely some patients rescheduled and still received AUD pharmacotherapy within 30 days of the original appointment. Finally, it was not possible to confirm whether a patient opened and read the education that was mailed to them. This may be another reason to explore electronic distribution of DTC education. This all may have contributed to the lack of statistically significant differences in both the primary and secondary outcomes.
There was a high level of variability between facility participation in the project. Two of 7 sites did not participate, and 3 sites restricted primary care engagement. This represents a significant limitation, particularly for the secondary outcome of placing consultations for MH or SUD services. Facilities that only included mental health or SUD HCPs may have resulted in lower consultation rates due to their inherent specialization, reducing the likelihood of self-referrals.
The project may overestimate prescribed AUD pharmacotherapy in the primary outcome due to potential misclassification of medications. While the project adhered to the national VA ADS AUD dashboard’s definition of AUD pharmacotherapy, including acamprosate, disulfiram, naltrexone, topiramate, and gabapentin, some of these medications have multiple indications. For example, gabapentin is commonly prescribed for peripheral neuropathy, and topiramate is used to treat migraines and seizures. The multipurpose use adds uncertainty about whether they were prescribed specifically for AUD treatment, especially in cases where the HCP is responsible for treating a broad range of disease states, as in primary care.
CONCLUSIONS
Results of this quality improvement project did not show a statistically significant difference between patients sent DTC education and the comparator group for the initiation of AUD pharmacotherapy or placement of a consult to mental health or SUD services within 30 days of their scheduled visit. Future studies may seek to implement stricter criteria to confirm the intended use of topiramate and gabapentin, such as looking for keywords in the prescription instructions for use, performing chart reviews, and/or only including these medications if prescribed by a mental health or SUD HCP. Alternatively, future studies may consider limiting the analysis to only FDA-approved AUD medications: acamprosate, disulfiram, and naltrexone. It is vital to continue to enhance primary care HCP readiness to treat AUD, given the existing relationships and trust they often have with patients. Electronic methods for distributing DTC education could also be advantageous, as these methods may have the ability to track whether a message has been opened and read. Despite a lack of statistical significance, this project sparked crucial conversations and collaboration around AUD, available treatments, and addressing potential barriers to connecting patients to care within VISN 21.
- Centers for Disease Control and Prevention. Facts about U.S. deaths from excessive alcohol use. August 6, 2024. Accessed February 5, 2025. https://www.cdc.gov/alcohol/facts-stats/
- State Health Access Data Assistance Center. Escalating alcohol-involved death rates: trends and variation across the nation and in the states from 2006 to 2019. April 19, 2021. Accessed February 5, 2025. https://www.shadac.org/escalating-alcohol-involved-death-rates-trends-and-variation-across-nation-and-states-2006-2019
- National Institute on Alcohol Abuse and Alcoholism. Alcohol- related emergencies and deaths in the United States. Updated November 2024. Accessed February 5, 2025. https://www.niaaa.nih.gov/alcohols-effects-health/alcohol-topics/alcohol-facts-and-statistics/alcohol-related-emergencies-and-deaths-united-states
- Esser MB, Sherk A, Liu Y, Naimi TS. Deaths from excessive alcohol use - United States, 2016- 2021. MMWR Morb Mortal Wkly Rep. 2024;73(8):154-161. doi:10.15585/mmwr.mm7308a1
- Canver BR, Newman RK, Gomez AE. Alcohol Withdrawal Syndrome. In: StatPearls. StatPearls Publishing; 2024.
- National Institute on Alcohol Abuse and Alcoholism. Alcohol treatment in the United States. Updated January 2025. Accessed February 5, 2025. https://www.niaaa.nih.gov/alcohols-effects-health/alcohol-topics/alcohol-facts-and-statistics/alcohol-treatment-united-states
- National Institute on Alcohol Abuse and Alcoholism. Alcohol use disorder (AUD) in the United States: age groups and demographic characteristics. Updated September 2024. Accessed February 5, 2025. https://www.niaaa.nih.gov/alcohols-effects-health/alcohol-topics/alcohol-facts-and-statistics/alcohol-use-disorder-aud-united-states-age-groups-and-demographic-characteristics
- Reus VI, Fochtmann LJ, Bukstein O, et al. The American Psychiatric Association practice guideline for the pharmacological treatment of patients with alcohol use disorder. Am J Psychiatry. 2018;175(1):86-90. doi:10.1176/appi.ajp.2017.1750101
- Blodgett JC, Del Re AC, Maisel NC, Finney JW. A meta-analysis of topiramate’s effects for individuals with alcohol use disorders. Alcohol Clin Exp Res. 2014;38(6):1481-1488. doi:10.1111/acer.12411
- Maisel NC, Blodgett JC, Wilbourne PL, Humphreys K, Finney JW. Meta-analysis of naltrexone and acamprosate for treating alcohol use disorders: when are these medications most helpful? Addiction. 2013;108(2):275-293. doi:10.1111/j.1360-0443.2012.04054.x
- Jonas DE, Amick HR, Feltner C, et al. Pharmacotherapy for adults with alcohol use disorders in outpatient settings: a systematic review and meta-analysis. JAMA. 2014;311(18):1889-1900. doi:10.1001/jama.2014.3628
- US Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the management of substance use disorders. August 2021. Accessed February 5, 2025. https://www.healthquality.va.gov/guidelines/MH/sud/VADODSUDCPG.pdf
- Ranney RM, Bernhard PA, Vogt D, et al. Alcohol use and treatment utilization in a national sample of veterans and nonveterans. J Subst Use Addict Treat. 2023;146:208964. doi:10.1016/j.josat.2023.208964
- US Department of Veterans Affairs, Pharmacy Benefit Management Service, Academic Detailing Service. AUD Trend Report. https://vaww.pbi.cdw.va.gov/PBIRS/Pages/ReportViewer.aspx?/GPE/PBM_AD/SSRS/AUD/AUD_TrendReport
- Mendes MA, Smith JP, Marin JK, et al. Reducing benzodiazepine prescribing in older veterans: a direct-to-consumer educational brochure. Fed Pract. 2018;35(9):36-43.
- Tannenbaum C, Martin P, Tamblyn R, Benedetti A, Ahmed S. Reduction of inappropriate benzodiazepine prescriptions among older adults through direct patient education: the EMPOWER cluster randomized trial. JAMA Intern Med. 2014;174(6):890-898. doi:10.1001/jamainternmed.2014.949
- Maloney R, Funmilayo M. Acting on the AUDIT-C: implementation of direct-to-consumer education on unhealth alcohol use. Presented on March 31, 2023; Central Virginia Veterans Affairs Health Care System, Richmond, Virginia.
- US Department of Veterans Affairs, Pharmacy Benefit Management Service. Alcohol use disorder (AUD) – leading the charge in the treatment of AUD: a VA clinician’s guide. February 2022. Accessed February 5, 2025. https://www.pbm.va.gov/PBM/AcademicDetailingService/Documents/508/10-1530_AUD_ClinicianGuide_508Conformant.pdf
- Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011;46(3):399-424. doi:10.1080/00273171.2011.568786
- National Institute on Alcohol Abuse and Alcoholism. Stigma: overcoming a pervasive barrier to optimal care. Updated January 6, 2025. Accessed February 5, 2025. https://www.niaaa.nih.gov/health-professionals-communities/core-resource-on-alcohol/stigma-overcoming-pervasive-barrier-optimal-care
- Sudhinaraset M, Wigglesworth C, Takeuchi DT. Social and cultural contexts of alcohol use: influences in a socialecological framework. Alcohol Res. 2016;38(1):35-45.
- Tanski SE, McClure AC, Li Z, et al. Cued recall of alcohol advertising on television and underage drinking behavior. JAMA Pediatr. 2015;169(3):264-271. doi:10.1001/jamapediatrics.2014.3345
- Hyland CJ, McDowell MJ, Bain PA, Huskamp HA, Busch AB. Integration of pharmacotherapy for alcohol use disorder treatment in primary care settings: a scoping review. J Subst Abuse Treat. 2023;144:108919. doi:10.1016/j.jsat.2022.108919
- National Institute on Alcohol Abuse and Alcoholism. The truth about holiday spirits. Updated November 2023. Accessed February 5, 2025. ,a href="https://www.niaaa.nih.gov/publications/brochures-and-fact-sheets/truth-about-holiday-spirits">https://www.niaaa.nih.gov/publications/brochures-and-fact-sheets/truth-about-holiday-spirits
- Centers for Disease Control and Prevention. Facts about U.S. deaths from excessive alcohol use. August 6, 2024. Accessed February 5, 2025. https://www.cdc.gov/alcohol/facts-stats/
- State Health Access Data Assistance Center. Escalating alcohol-involved death rates: trends and variation across the nation and in the states from 2006 to 2019. April 19, 2021. Accessed February 5, 2025. https://www.shadac.org/escalating-alcohol-involved-death-rates-trends-and-variation-across-nation-and-states-2006-2019
- National Institute on Alcohol Abuse and Alcoholism. Alcohol- related emergencies and deaths in the United States. Updated November 2024. Accessed February 5, 2025. https://www.niaaa.nih.gov/alcohols-effects-health/alcohol-topics/alcohol-facts-and-statistics/alcohol-related-emergencies-and-deaths-united-states
- Esser MB, Sherk A, Liu Y, Naimi TS. Deaths from excessive alcohol use - United States, 2016- 2021. MMWR Morb Mortal Wkly Rep. 2024;73(8):154-161. doi:10.15585/mmwr.mm7308a1
- Canver BR, Newman RK, Gomez AE. Alcohol Withdrawal Syndrome. In: StatPearls. StatPearls Publishing; 2024.
- National Institute on Alcohol Abuse and Alcoholism. Alcohol treatment in the United States. Updated January 2025. Accessed February 5, 2025. https://www.niaaa.nih.gov/alcohols-effects-health/alcohol-topics/alcohol-facts-and-statistics/alcohol-treatment-united-states
- National Institute on Alcohol Abuse and Alcoholism. Alcohol use disorder (AUD) in the United States: age groups and demographic characteristics. Updated September 2024. Accessed February 5, 2025. https://www.niaaa.nih.gov/alcohols-effects-health/alcohol-topics/alcohol-facts-and-statistics/alcohol-use-disorder-aud-united-states-age-groups-and-demographic-characteristics
- Reus VI, Fochtmann LJ, Bukstein O, et al. The American Psychiatric Association practice guideline for the pharmacological treatment of patients with alcohol use disorder. Am J Psychiatry. 2018;175(1):86-90. doi:10.1176/appi.ajp.2017.1750101
- Blodgett JC, Del Re AC, Maisel NC, Finney JW. A meta-analysis of topiramate’s effects for individuals with alcohol use disorders. Alcohol Clin Exp Res. 2014;38(6):1481-1488. doi:10.1111/acer.12411
- Maisel NC, Blodgett JC, Wilbourne PL, Humphreys K, Finney JW. Meta-analysis of naltrexone and acamprosate for treating alcohol use disorders: when are these medications most helpful? Addiction. 2013;108(2):275-293. doi:10.1111/j.1360-0443.2012.04054.x
- Jonas DE, Amick HR, Feltner C, et al. Pharmacotherapy for adults with alcohol use disorders in outpatient settings: a systematic review and meta-analysis. JAMA. 2014;311(18):1889-1900. doi:10.1001/jama.2014.3628
- US Department of Veterans Affairs, Department of Defense. VA/DoD clinical practice guideline for the management of substance use disorders. August 2021. Accessed February 5, 2025. https://www.healthquality.va.gov/guidelines/MH/sud/VADODSUDCPG.pdf
- Ranney RM, Bernhard PA, Vogt D, et al. Alcohol use and treatment utilization in a national sample of veterans and nonveterans. J Subst Use Addict Treat. 2023;146:208964. doi:10.1016/j.josat.2023.208964
- US Department of Veterans Affairs, Pharmacy Benefit Management Service, Academic Detailing Service. AUD Trend Report. https://vaww.pbi.cdw.va.gov/PBIRS/Pages/ReportViewer.aspx?/GPE/PBM_AD/SSRS/AUD/AUD_TrendReport
- Mendes MA, Smith JP, Marin JK, et al. Reducing benzodiazepine prescribing in older veterans: a direct-to-consumer educational brochure. Fed Pract. 2018;35(9):36-43.
- Tannenbaum C, Martin P, Tamblyn R, Benedetti A, Ahmed S. Reduction of inappropriate benzodiazepine prescriptions among older adults through direct patient education: the EMPOWER cluster randomized trial. JAMA Intern Med. 2014;174(6):890-898. doi:10.1001/jamainternmed.2014.949
- Maloney R, Funmilayo M. Acting on the AUDIT-C: implementation of direct-to-consumer education on unhealth alcohol use. Presented on March 31, 2023; Central Virginia Veterans Affairs Health Care System, Richmond, Virginia.
- US Department of Veterans Affairs, Pharmacy Benefit Management Service. Alcohol use disorder (AUD) – leading the charge in the treatment of AUD: a VA clinician’s guide. February 2022. Accessed February 5, 2025. https://www.pbm.va.gov/PBM/AcademicDetailingService/Documents/508/10-1530_AUD_ClinicianGuide_508Conformant.pdf
- Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011;46(3):399-424. doi:10.1080/00273171.2011.568786
- National Institute on Alcohol Abuse and Alcoholism. Stigma: overcoming a pervasive barrier to optimal care. Updated January 6, 2025. Accessed February 5, 2025. https://www.niaaa.nih.gov/health-professionals-communities/core-resource-on-alcohol/stigma-overcoming-pervasive-barrier-optimal-care
- Sudhinaraset M, Wigglesworth C, Takeuchi DT. Social and cultural contexts of alcohol use: influences in a socialecological framework. Alcohol Res. 2016;38(1):35-45.
- Tanski SE, McClure AC, Li Z, et al. Cued recall of alcohol advertising on television and underage drinking behavior. JAMA Pediatr. 2015;169(3):264-271. doi:10.1001/jamapediatrics.2014.3345
- Hyland CJ, McDowell MJ, Bain PA, Huskamp HA, Busch AB. Integration of pharmacotherapy for alcohol use disorder treatment in primary care settings: a scoping review. J Subst Abuse Treat. 2023;144:108919. doi:10.1016/j.jsat.2022.108919
- National Institute on Alcohol Abuse and Alcoholism. The truth about holiday spirits. Updated November 2023. Accessed February 5, 2025. ,a href="https://www.niaaa.nih.gov/publications/brochures-and-fact-sheets/truth-about-holiday-spirits">https://www.niaaa.nih.gov/publications/brochures-and-fact-sheets/truth-about-holiday-spirits
Impact of Multisite Patient Education on Pharmacotherapy for Veterans With Alcohol Use Disorder
Impact of Multisite Patient Education on Pharmacotherapy for Veterans With Alcohol Use Disorder