User login
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
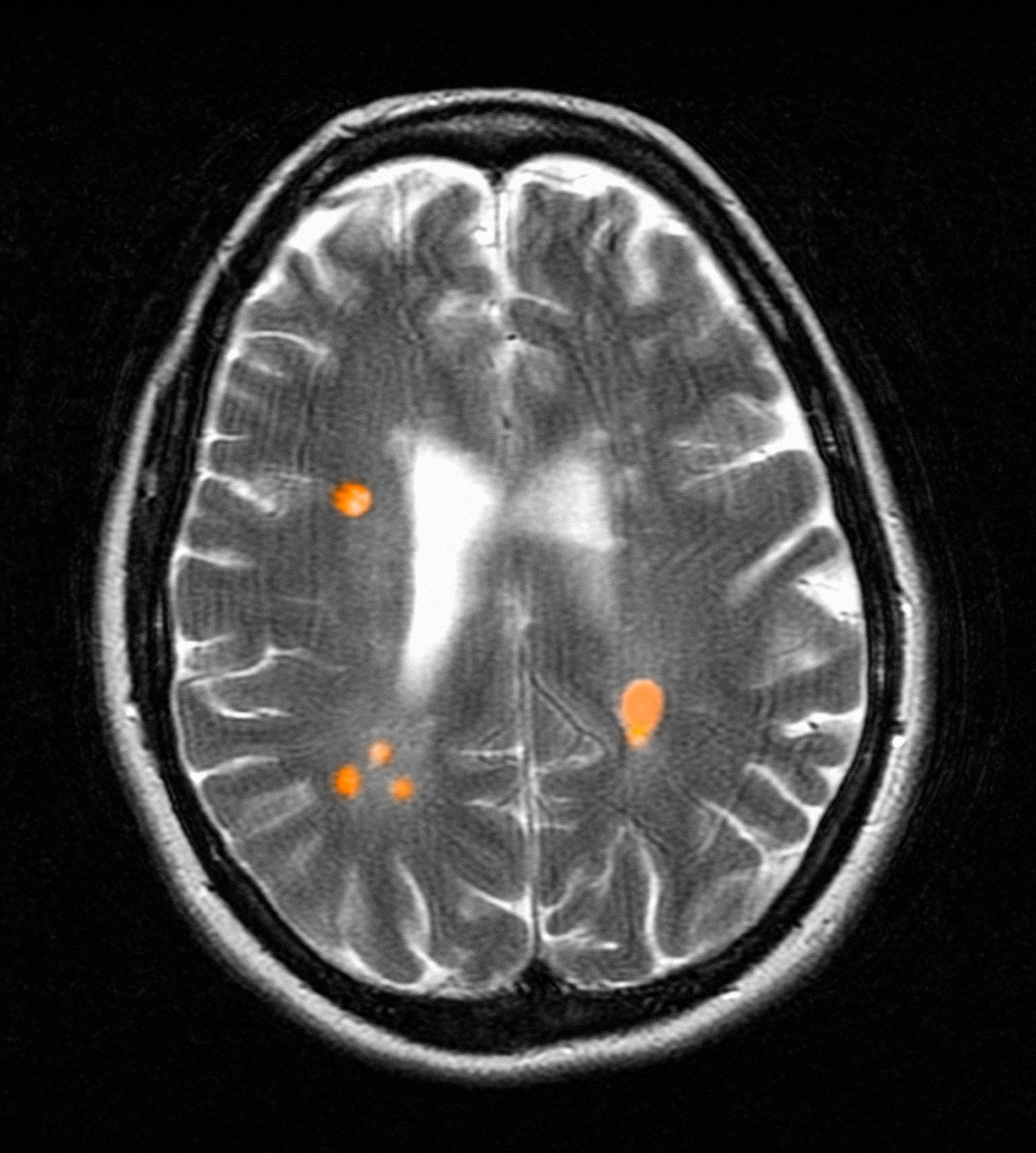
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
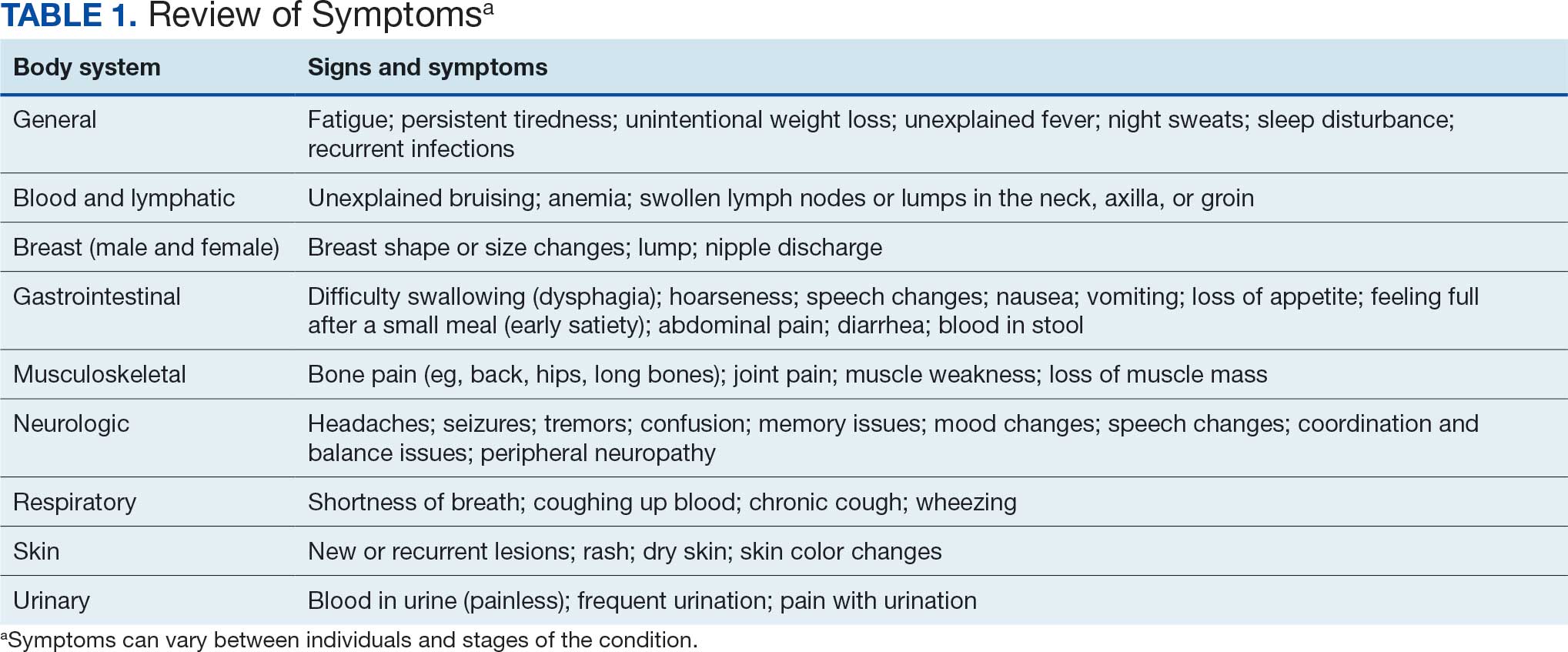
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).
Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
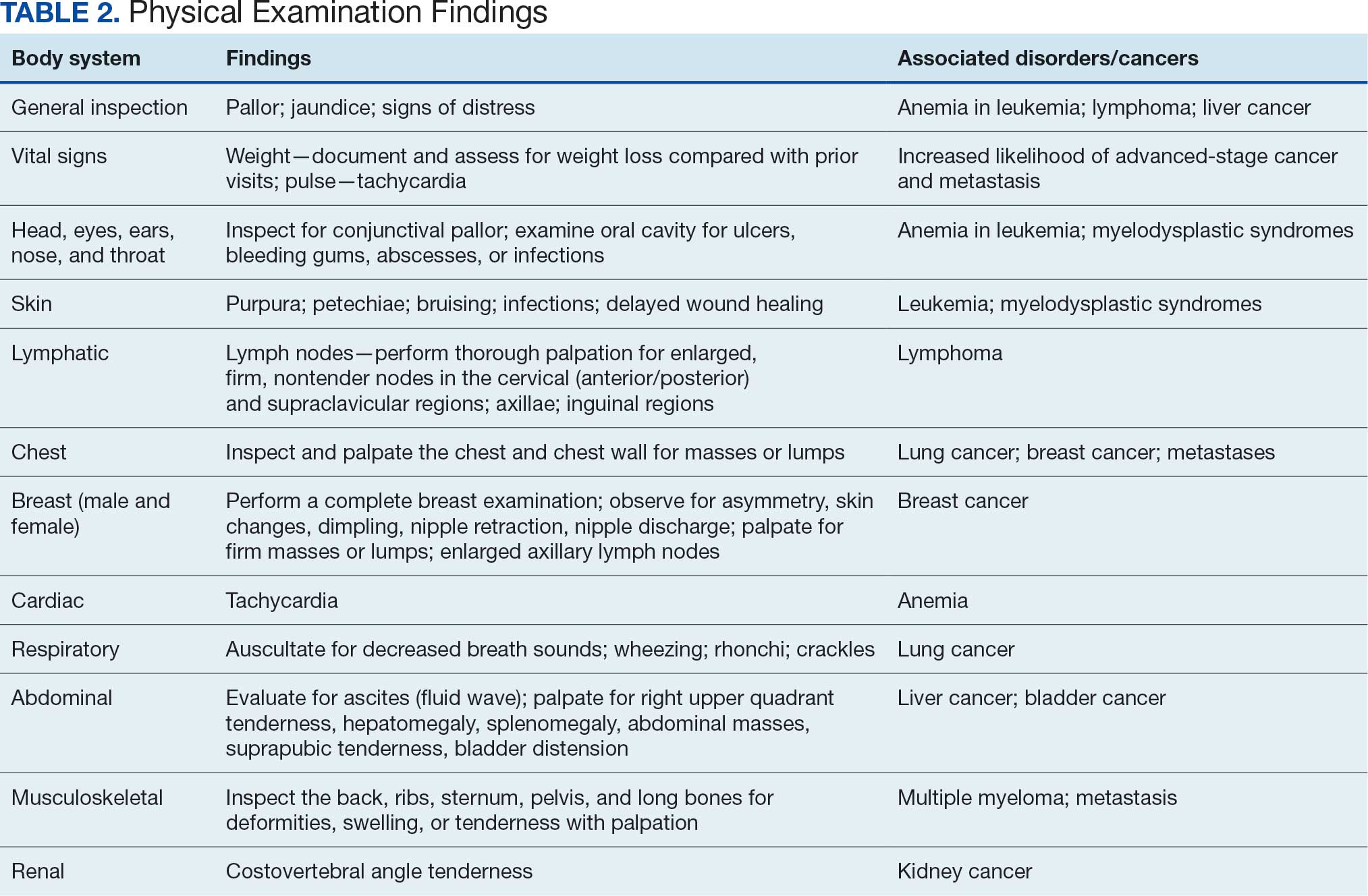
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.
Proposed Diagnostic Screening Panel
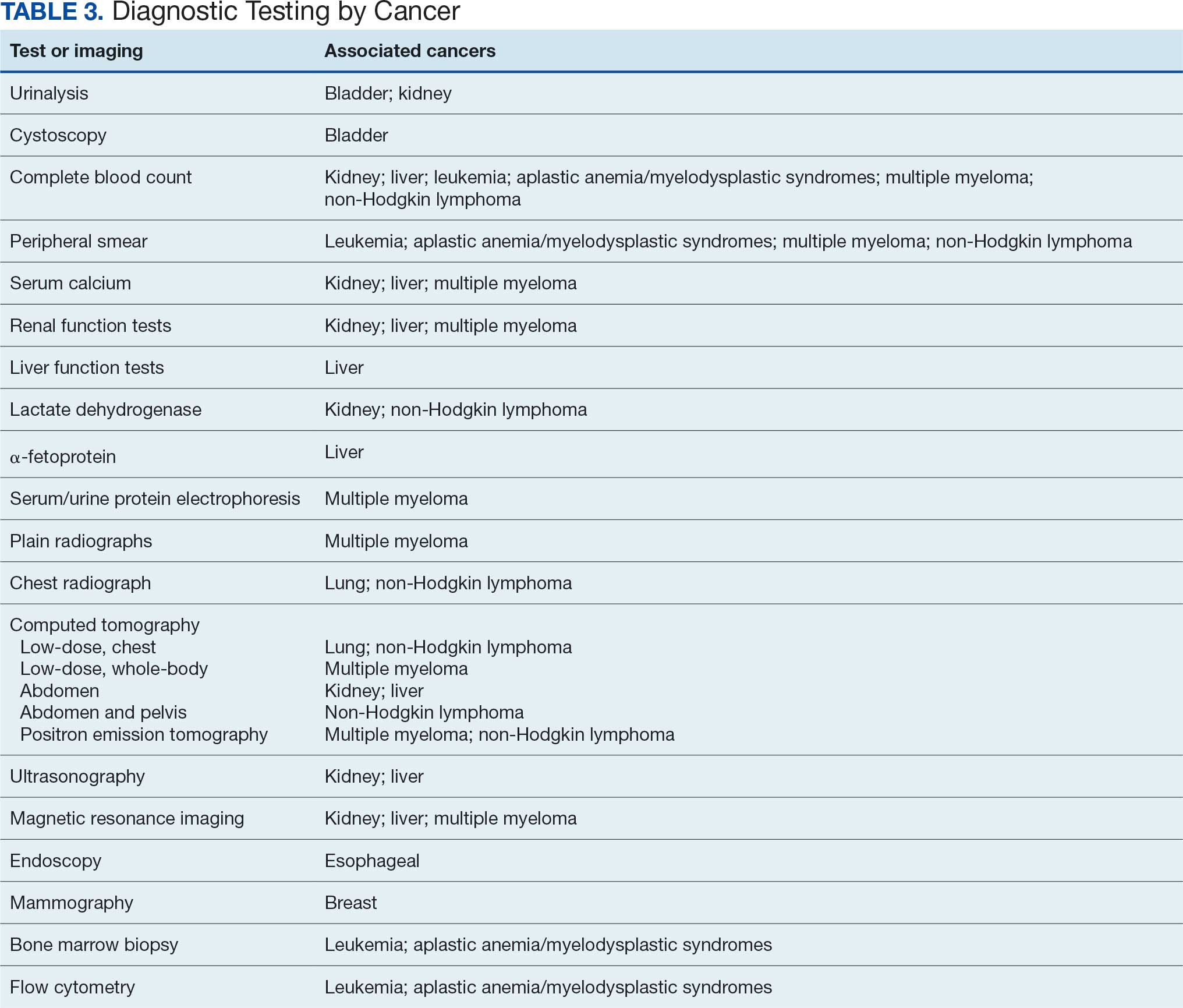
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).
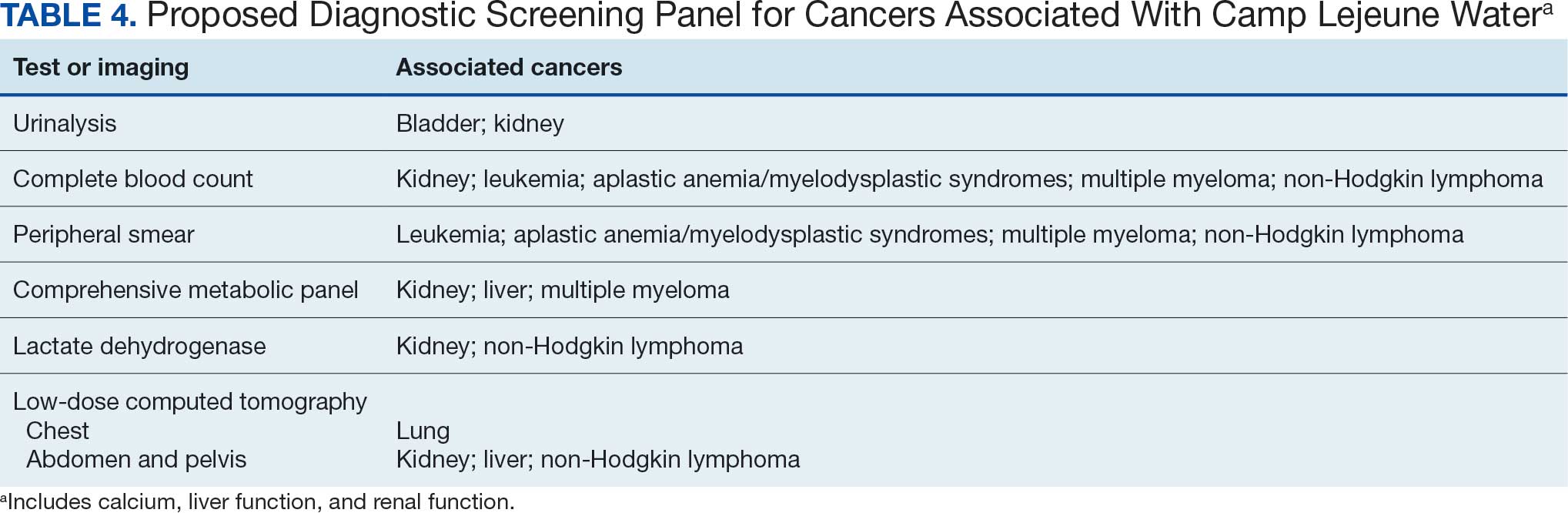
A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).

Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.

Proposed Diagnostic Screening Panel
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).


A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
Clinical awareness of cancers associated with Camp Lejeune water contamination exposure remains limited despite legal and policy advances. Gaps persist in early symptom recognition and timely diagnostic evaluation before a definitive cancer diagnosis among exposed personnel. This may represent missed opportunities for earlier identification of volatile organic compounds (VOCs)-related cancers and for less invasive treatment options for veterans in this high-risk population.
Federal health care practitioners (HCPs), especially those in primary care and internal medicine, are uniquely positioned to bridge this gap. By improving the recognition of symptoms, pertinent physical examination findings, and implementing a diagnostic screening panel, HCPs can support accurate diagnoses and facilitate earlier treatment to improve health and quality of life for this population.
From 1953 to 1985, as many as 1 million military personnel, civilian workers, and their families stationed at US Marine Corps Base Camp Lejeune were unknowingly exposed to toxic and carcinogenic chemicals in drinking and bathing water.1 Three of the 8 main water sources on base were contaminated with VOCs, which are associated with multiple cancers.1-3
The US Department of Veterans Affairs (VA) recognizes 15 conditions associated with Camp Lejeune contaminated water exposure for VA benefits, including 10 cancers: adult leukemia; aplastic anemia and other myelodysplastic syndromes (MDS); bladder, esophageal, kidney, liver, breast (male and female), and lung cancers; multiple myeloma; and non-Hodgkin lymphoma (NHL).4
BACKGROUND
Established in 1942, Camp Lejeune is an important Marine Corps training installation. Between 1953 and 1985, multiple on-base water systems were contaminated with VOCs, including trichloroethylene (TCE), perchloroethylene (PCE), benzene, and vinyl chloride, due to improper waste disposal and industrial runoff from on- and off-base sources.5 Tarawa Terrace water treatment plant (WTP) was contaminated primarily with PCE from November 1957 to February 1987. Hadnot Point WTP was contaminated with TCE from August 1953 to December 1984, along with PCE, and benzene, toluene, ethylbenzene, and xylene (BTEX). Holcomb Boulevard WTP, established in 1972, was contaminated with TCE from June 1972 to February 1985.2 These contaminants entered the drinking and bathing water supply over decades, and exposure often occurred concurrently across = 1 VOC, compounding health risks.2,3 This prolonged 32-year VOC exposure window underlies current concerns regarding long-term cancer risk among affected service members, civilian employees, and family members. Epidemiologic research has found statistically significant associations between VOC exposure and multiple cancers, neurologic conditions, and reproductive issues.6 Specifically, TCE is associated with higher risks of hematologic cancers, multiple myeloma, NHL, and kidney cancer.3 PCE is linked with kidney cancer, benzene with multiple myeloma and NHL, and vinyl chloride with hepatobiliary cancers.3 A cohort mortality study compared Camp Lejeune personnel with a control group at Camp Pendleton from 1972 to 1985 and found a 3-fold higher incidence or mortality rate for kidney, esophageal, and female breast cancers, leukemia, and lymphoma among exposed Camp Lejeune personnel.6 Notably, personnel assigned to Camp Lejeune for as little as 6 months faced up to a 6-fold increase in cancer risk; the average military assignment between 1975 and 1985 was 18 months.3,6
Honoring America's Veterans and Caring for Camp Lejeune Families Act of 2012, the Sergeant First Class Heath Robinson Honoring Our Promise to Address Comprehensive Toxics (PACT) Act of 2022, the Camp Lejeune Justice Act of 2022, and the pending Ensuring Justice for Camp Lejeune Victims Act of 2025 provide health care and legal resources for personnel and families affected by Camp Lejeune’s contaminated water.6-8 These laws acknowledge associations between exposure and specific health conditions and expanded health care, benefits, and legal recourse for affected veterans, survivors, and their families.8,9
CANCERS LINKED TO CAMP LEJEUNE
Camp Lejeune VOC-contaminated water exposure is associated with solid tumor and hematologic cancers. Symptoms, physical examination findings, and diagnostic considerations vary by cancer type (Table 1).

Bladder Cancer
The US incidence rate of bladder cancer for both males and females is 18 per 100,000 individuals per year, with a death rate of 4.1 per 100,000 individuals per year, and a 2.1% lifetime diagnosis risk.10 Personnel exposed to VOCs at Camp Lejeune had a 9% higher risk of developing bladder cancer and a 2% increased mortality compared with an unexposed control group at Camp Pendleton.1,7 Other bladder cancer subtypes at increased risk are papillary transitional cell carcinoma, nonpapillary transition cell carcinoma, and urothelial carcinoma.7 This is consistent with prior research that found PCE exposure is associated with an increased risk for bladder cancer.3,7,11 Smoking and tobacco use remain significant risk factors for bladder cancer.12
Symptomatology. The most common symptom associated with bladder cancer is painless hematuria (gross or microscopic). Other often delayed symptoms include urinary frequency, urgency, or nocturia.13,14
Diagnostics. Screening tests include urinalysis for hematuria, urine cytology, and cystoscopy with biopsy as the gold standard for diagnosis and staging.15,16
Kidney Cancer
The US incidence rate of kidney cancer and renal pelvis cancer for both males and females is 17.5 per 100,000 individuals per year, with a death rate of 3.4 per 100,000, and a 1.8% lifetime diagnosis risk.17 Camp Lejeune personnel exposed to VOCs had a 6% increased risk of developing kidney cancer and renal pelvis cancer and a 21% higher mortality risk compared with Camp Pendleton controls.1,7 Subtypes at risk include renal cell carcinoma and papillary carcinoma.7 This is consistent with prior research that found exposures to TCE and PCE are associated with a 3-fold increased risk of kidney cancer.3,7
Symptomatology. Hematuria, flank pain, and a palpable abdominal mass are common symptoms associated with kidney cancer. In advanced stages, other symptoms may include left-sided varicocele, anemia, weight loss, fatigue, fever, and night sweats.18
Diagnostics. Screening tests include urinalysis to assess the presence of blood, complete blood count (CBC) to assess anemia, calcium (elevated), and lactate dehydrogenase (LDH), which may be elevated. Imaging strategies include abdominal computed tomography (CT), magnetic resonance imaging (MRI), or ultrasound.19
Esophageal Cancer
The US incidence rate of esophageal cancer for both males and females is 4.2 per 100,000 individuals per year, the death rate is 3.7 per 100,000 individuals per year, and a 0.5% lifetime diagnosis risk.20 VOC-exposed Camp Lejeune personnel had a 27% increased incidence and 25% increased mortality compared with the control group.1,7 Esophageal cancer subtypes at elevated risk include squamous cell carcinoma and adenocarcinoma. This is consistent with prior research that found Camp Lejeune water exposure is associated with a 3-fold increased risk for esophageal cancer.7 Additional risk factors include history of smoking and alcohol use.21
Symptomatology. Esophageal cancer is often asymptomatic with potential symptoms that include dysphagia, hoarseness, and weight loss in advanced disease.22
Diagnostics. Endoscopy with biopsy is the definitive method for diagnosis.23
Liver Cancer
The US incidence rate of liver cancer and intrahepatic bile duct cancer for both males and females is 9.4 per 100,000 individuals per year, with a death rate of 6.6 per 100,000 individuals per year, and a 1.1% lifetime diagnosis risk.24 VOC-exposed personnel had a 1% higher mortality than controls.1
Symptomatology. Liver cancer is often asymptomatic and appears in late stages.25 Common symptoms include right upper quadrant pain, early satiety, nausea, vomiting, loss of appetite, weight loss, ascites, jaundice, and abnormal bleeding or bruising.25,26
Diagnostics. Diagnostic tests may include an ultrasound, CT, or MRI. Additional laboratory testing may include liver function, a-fetoprotein blood, CBC, renal function, calcium, and hepatitis panel screening for hepatitis B and C.27,28
Lung Cancer
The US incidence rate of lung cancer for both males and females is 47.8 per 100,000 individuals per year, with a death rate of 31.5 per 100,000 individuals per year, and a 5.4% lifetime diagnosis risk.29 VOC-exposed personnel had a 16% increased risk and 19% higher mortality.1,7 Subtypes include large cell, small cell, non-small cell, squamous cell, and adenocarcinoma.7 Smoking is an additional risk factor.30
Symptomatology. Symptoms of lung cancer include cough, shortness of breath, chest pain worse with deep breathing, unexplained weight loss, fatigue, night sweats, and recurrent fevers. Advanced stages may metastasize or spread to the liver, bones, and brain.31
Diagnostics. Low-dose CT and chest X-ray are used for screening.32
Breast Cancer
The US incidence rate of female breast cancer is 130.8 per 100,000 individuals per year, with a death rate of 19.2 per 100,000 individuals per year, and a 13.0% lifetime risk of diagnosis.33 For female VOC-exposed personnel, there was an equal risk of developing breast cancer as the control group.1 However, exposed females at Camp Lejeune had a 23% higher mortality risk compared to the control group.7 Breast cancer subtypes among females include ductal carcinoma, lobular carcinoma, and ductal-lobular carcinoma.1
The US incidence rate of male breast cancer is 1.3 per 100,000 individuals per year, with a death rate of 0.3 per 100,000 individuals per year.34,35 The lifetime risk for males developing breast cancer is 137.7 per 100,000 and about 70 to 100 times less common in men than women.36
Male personnel exposed at Camp Lejeune had a 4% increased risk for developing breast cancer compared to Camp Pendleton.7 However, mortality was lower in the Camp Lejeune group.1 Although male breast cancer is rare, males at Camp Lejeune had a higher incidence, indicating a link between TCE, PCE, vinyl chloride exposures and male breast cancer.37 Male breast cancer is more often diagnosed in advanced stages than female breast cancer due to the lack of awareness or absence of routine screenings.38 The most common breast cancer type in males is invasive ductal carcinoma, accounting for 85% to 90% of cases; lobular carcinoma is the second most common type.39
Symptomatology. In both females and males, breast cancer symptoms include painless, firm mass or lump in the breast (left breast slightly more common than right), skin changes or dimpling, nipple retraction or turning inward, and nipple discharge. Breast cancer can spread to the lymph nodes and can be appreciated in axilla or clavicular regions.40
Diagnostics. The diagnostic evaluation for breast cancer is similar for females and males. It includes a clinical breast examination, diagnostic mammogram, and ultrasound.41 Mammograms can distinguish between gynecomastia and cancer, especially in males.42 A core or fine needle biopsy is needed to confirm diagnosis.41
Adult Leukemia
The US incidence rate of leukemia for both male and female was 14.4 per 100,000 individuals per year, with a death rate of 5.8 per 100,000 individuals per year, and a 1.5% lifetime diagnosis risk.43
VOC-exposed personnel had a 7% higher risk of developing leukemia and a 13% increased mortality risk compared with the control group.1,7 Subtypes of leukemia at risk included a 38% increased incidence of acute myeloid/monocytic leukemia (AML) and a 2% increased incidence of chronic lymphocytic leukemia (CLL).1 Benzene and TCE exposures are known risk factors for AML and other leukemias.7 Personnel at Camp Lejeune had 3 times the incidence or mortality for leukemia, specifically AML mortality at 20%.7 Smoking is an additional risk factor for certain leukemias, especially AML.30
Symptomatology. Symptoms associated with leukemia are often nonspecific and may include fatigue, pallor, easy bruising or bleeding (skin or gums), recurrent infections secondary to neutropenia, fever, night sweats, pain or feeling full after a small meal due to enlarged spleen or liver, and weight loss.44,45
Diagnostics. An initial screening includes a CBC with differential, a peripheral smear to detect the presence of blast cells, as well as Auer rods in myeloid blast cells in AML or smudge cells in CLL. Confirmatory tests may include bone marrow biopsy or flow cytometry. A referral to a hematologist is recommended for any suspected leukemia.46,47
Myelodysplastic Syndromes
Aplastic anemia and MDS are considered rare disorders.48 Aplastic anemia is a nonmalignant bone marrow failure disorder with pancytopenia and hypocellular bone marrow due to the loss of hematopoietic stem cells.48 MDS is a type of hematopoietic cancer where the bone marrow produces abnormal blood cells or does not make enough healthy cells.49 This can lead to an increased risk for infection, cytopenias, neutropenia, refractory anemia, and thrombocytopenia, and progression to AML in some patients.49
The reported US incidence of MDS from 1975 to 2013 was 6.7 per 100,000 for males and 3.7 per 100,000 for females.50 Benzene exposure is linked to MDS and a known cause of AML.1 VOC-exposed personnel had a 68% increased risk of developing MDS and a 2.3-fold increased mortality risk compared to controls.1,7
Symptomatology. Some patients are asymptomatic at diagnosis.51 Symptoms related to cytopenia include fatigue, pallor, purpura, petechiae, bleeding of skin, gum, or nose, recurrent infections, fever, bone pain, loss of appetite, and weight loss.50,51
Diagnostics. Initial workup includes a CBC with differential to assess for anemia, white blood cell and absolute neutrophil counts (low), and thrombocytopenia.52 A peripheral blood smear may show myeloid blast cells. A bone marrow aspiration and biopsy, flow cytometry, and cytogenetic or molecular testing may be performed. If MDS is suspected, a referral to a hematologist should be considered.52
Multiple Myeloma
The US incidence rate of multiple myeloma for both males and females is 7.3 per 100,000 individuals per year, with a mortality rate of 2.9 per 100,000 individuals per year, and a 0.8% lifetime diagnosis risk.53 VOC-exposed personnel had a 13% increased risk of developing multiple myeloma and an 8% increased mortality risk compared to unexposed personnel.1,7
Symptomatology. Multiple myeloma may be asymptomatic in early stages. The most common presenting symptom is bone pain, especially in the back, hips, and long bones, due to hypercalcemia from increased reabsorption, plasma cell tumor overgrowth in the bone marrow, and lytic lesions.54 Additional symptoms include fatigue and pallor related to anemia, leukopenia, thrombocytopenia, recurrent infections, extreme thirst, frequent urination, dehydration, confusion associated with hypercalcemia, peripheral neuropathy, loss of appetite, weight loss, and renal impairment or failure.54
Diagnostics. Testing considerations include a CBC with a peripheral blood smear to evaluate anemia and rouleaux formation of red blood cells (seen in > 50% of patients with multiple myeloma), comprehensive metabolic panel (CMP) to assess kidney function, calcium levels (elevated), serum and urine protein electrophoresis with immunofixation to detect monoclonal protein (detected in > 80% of patients with multiple myeloma) and Bence-Jones proteins, serum free light chain assay, and a bone marrow biopsy for diagnosis.55,56
MRI of the spine and pelvis is the most sensitive to detecting bone marrow involvement and focal lesions before lytic lesion progression occurs and for assessing spinal cord compression.57 PET/CT is more sensitive at detecting extramedullary disease, outside of the spine, and for patients that cannot undergo MRI.57 A whole-body low-dose CT, either alone or with PET, is more sensitive than an X-ray at detecting lytic lesions, fractures, or osteoporosis associated with multiple myeloma.57
Non-Hodgkin Lymphoma
The US incidence rate of NHL for both males and females are 18.7 per 100,000 individuals per year, the death rate is 4.9 per 100,000 individuals per year, and a 2% lifetime diagnosis risk.58 VOC-exposed personnel had a 1% higher risk of developing NHL and a decreased mortality risk compared to the control group.1,7 Specific NHL subtypes with increased risk in the exposed cohort are mantle cell (26%), follicular (7%), Burkitt (53%), and marginal zone B-cell (45%).7
Symptomatology. NHL often presents with painless lymphadenopathy or enlarged lymph nodes involving the cervical, axillary, inguinal regions.59,60 Other symptoms include frequent infections, unexplained bruising, weight loss, and “B symptoms,” such as fever and night sweats.59,60 Some patients develop a mediastinal mass in the thorax, which if large may lead to cough or shortness of breath.59
Diagnostics. The initial diagnostic workup includes CBC with differential and LDH, which may be elevated.60,61 Imaging may begin with a chest X-ray to assess for a mediastinal mass; however, CTs of the chest, abdomen, and pelvis provide more detail to better assess for NHL. Whole body PET/CT is considered the gold standard for assessing and staging systemic involvement. If enlarged lymph nodes are present, a biopsy can confirm the subtype of NHL.60,61
PHYSICAL EXAMINATION
A focused physical examination may aid HCPs in early detection of the cancers associated with Camp Lejeune (Table 2). The physical examination can guide diagnostic testing and imaging for further assessment and workup for VOC-related cancers.

Proposed Diagnostic Screening Panel
Primary care and internal medicine HCPs have the opportunity to improve patient health outcomes by implementing a targeted diagnostic screening panel for identified veterans previously stationed at Camp Lejeune. Early identification of cancers associated with VOCs exposure can facilitate earlier treatment interventions and improve health and quality of life outcomes. The following diagnostic screening panel outlines a potential cost-effective strategy for evaluating and detecting the 10 cancers associated with VOC exposure in Camp Lejeune water.
Baseline Screening
Implementing a diagnostic screening panel in this high-risk cohort can lead to earlier diagnosis, reduce mortality, and improve patient outcomes through early intervention, which in turn may result in less invasive treatment. This approach may also reduce health care costs by avoiding costs associated with delayed diagnosis and advanced-stage cancer care (Tables 3 and 4).


A baseline panel of tests for exposed veterans could include:
- A CBC with differential and peripheral smear to assess for anemia, leukemia, thrombocytopenia, and blast cells associated with leukemias, MDS, multiple myeloma, and NHL.19,46,47,52,55,56,60,61
- CMP evaluates calcium, total protein, renal and liver renal function. Elevated test results may indicate kidney or liver cancer or multiple myeloma.19,27,28,55,56
- LDH testing may reveal levels that are elevated from tissue damage or high cell turnover in kidney cancer, multiple myeloma, and NHL.19,55,56,60,61
- Urinalysis with microscopy may detect hematuria, proteinuria and cellular casts in bladder and kidney cancers.13,24,19
- Low-dose CTs of the chest, abdomen, and pelvis are recommended for early identification of any masses or lymphadenopathy in lung, kidney, liver cancers, and NHL.19,27,28,32,60,61
COST EFFICIENCY
Screening Panel Cost
According to the Medicare Clinical Laboratory Fee Schedule payment cap for 2018, the mean cost for the proposed blood workup was $35 (CBC, $10; CMP, $13; LDH, $8; urinalysis, $4).62 Medicare procedure price schedule for 2025 includes $351 for a CT of the abdomen and pelvis with and without contrast (Current Procedural Terminology [CPT] code 74177) and $187 for a CT of the chest with and without contrast (CPT code 71270).63,64 The total proposed diagnostic screening panel payment cost about $572.
Cancer Care Cost
The average cost for initial cancer care across all cancer sites from 2007 to 2013 was $43,516 per patient; Camp Lejeune-associated cancers ranged from $26,443 for bladder cancer to $89,947 for esophageal cancer care.64 Further, the last year of life cost across all cancer sites averaged $109,727, and Camp Lejeune-associated cancer types ranged from $76,101 for breast cancer to $169,588 for leukemia.65
CONCLUSIONS
From 1953 to 1985, up to 1 million military personnel, civilian workers, and their families stationed at Camp Lejeune were unknowingly exposed to toxic and carcinogenic VOCs, which are associated with = 10 cancers, including bladder, kidney, esophageal, liver, lung, breast, and hematologic malignancies.1-4 Some veterans may be asymptomatic, whereas others present with subtle or specific symptoms that can vary by individual and the type and stage of cancer. HCPs have an opportunity to improve patient outcomes through awareness in identifying symptoms associated with Camp Lejeune water exposure and performing a thorough baseline physical examination, especially noting lymphadenopathy, unexplained weight loss, or masses, which can guide further diagnostic evaluation. Timely screening can identify cancers earlier, reducing delays in care, mitigating the cost burden associated with advanced-stage cancer treatment, improving survival outcomes, and enhancing quality of life. Primary care and internal medicine HCPs specifically play a crucial role in early recognition, physical assessment, and appropriate screening tools. A proposed panel includes CBC with differential and peripheral smear, CMP, LDH, urinalysis, and low-dose CTs of the chest, abdomen and pelvis. Implementation should be guided by clinical judgment and patient-specific risk factors. The proposed diagnostic screening panel is a small price to pay for those who served in any capacity at Camp Lejeune.
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
- Bove FJ, Greek A, Gatiba R, et al. Cancer incidence among Marines and Navy personnel and civilian workers exposed to industrial solvents in drinking water at US Marine Corps Base Camp Lejeune: a cohort study. Environ Health Perspect. 2024;132:107008. doi:10.1289/EHP14966
- Maslia ML, Aral MM, Ruckart PZ, Bove FJ. Reconstructing historical VOC concentrations in drinking water for epidemiological studies at a US military base: summary of results. Water (Basel). 2016;8:449. doi:10.3390/w8100449
- Rosenfeld PE, Spaeth KR, McCarthy SJ, et al. Camp Lejeune Marine cancer risk assessment for exposure to contaminated drinking water from 1955 to 1987. Water Air Soil Pollut. 2024;235(2). doi:10.1007/s11270-023-06863-y
- US Department of Veterans Affairs, Veterans Health Administration. Camp Lejeune: past water contamination. Updated April 15, 2025. Accessed March 3, 2026. https://www.publichealth.va.gov/exposures/camp-lejeune/
- Jung K, Khan A, Mocharnuk R, et al. Clinical encounter with three cancer patients affected by groundwater contamination at Camp Lejeune: a case series and review of the literature. J Med Case Rep. 2022;16(1):272. doi:10.1186/s13256-022-03501-9
- Honoring America's Veterans and Caring for Camp Lejeune Familes Act of 2012, Pub L No. 112-154. Janey Ensminger Act. Congress.gov. Accessed April 15, 2026. https://ww.congress.gov/bill/112th-congress/house-bill/1627
- Bove FJ, Greek A, Gatiba R, et al. Evaluation of mortality among Marines, Navy personnel, and civilian workers exposed to contaminated drinking water at USMC Base Camp Lejeune: a cohort study. Environ Health. 2024;23(1):61. doi:10.1186/s12940-024-01099-7
- Honoring our PACT Act of 2022 (Pub L No. 117-168): expansion of health care eligibility and toxic exposure screenings. Congress.gov. Accessed March 3, 2026. https://www.congress.gov/bill/117th-congress/house-bill/3967
- Ensuring Justice for Camp Lejeune Victims Act of 2025. Congress.gov. Accessed March 24, 2026. https://www.congress.gov/bill/119th-congress/house-bill/4145
- SEER. Cancer stat facts: bladder cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/urinb.html
- Agency for Toxic Substances and Disease Registry. ATSDR assessment of the evidence for the drinking water contaminants at Camp Lejeune and specific cancers and other diseases. Published January 13, 2017. Accessed March 3, 2026. https://www.atsdr.cdc.gov/camp-lejeune/media/pdfs/2024/10/ATSDR_summary_of_the_evidence_for_causality_TCE_PCE_508.pdf
- National Cancer Institute. What is bladder cancer? Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder
- National Cancer Institute. Bladder cancer symptoms. Updated February 16, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/symptoms
- American Cancer Society. Bladder cancer signs and symptoms. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- National Cancer Institute. Bladder cancer screening. Updated April 27, 2023. Accessed March 3, 2026. https://www.cancer.gov/types/bladder/screening
- American Cancer Society. Tests for bladder cancer. Updated March 12, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/bladder-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: kidney and renal pelvis cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/kidrp.html
- American Cancer Society. Kidney cancer signs and symptoms. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for kidney cancer. Updated May 1, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/kidney-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: esophageal cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/esoph.html
- Engel LS, Chow WH, Vaughan TL, et al. Population attributable risks of esophageal and gastric cancers.
J Natl Cancer Inst. 2003;95(18):1404-1413. doi:10.1093/jnci/djg047 - American Cancer Society. Signs and symptoms of esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/signs-and-symptoms.html
- American Cancer Society. Tests for esophageal cancer. Updated March 20, 2020. Accessed March 3, 2026. https://www.cancer.org/cancer/types/esophagus-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: liver and intrahepatic bile duct cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/livibd.html
- American Cancer Society. Signs and symptoms of liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. What is liver cancer? Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer
- American Cancer Society. Tests for liver cancer. Updated February 11, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/liver-cancer/detection-diagnosis-staging/how-diagnosed.html
- National Cancer Institute. Liver cancer screening. Updated May 15, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/liver/what-is-liver-cancer/screening
- SEER. Cancer stat facts: lung cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/lungb.html
- US Department of Health and Human Services. The Health Consequences of Smoking: A Report of the Surgeon General. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. Accessed March 3, 2026. https://www.ncbi.nlm.nih.gov/books/NBK44695/pdf/Bookshelf_NBK44695.pdf
- American Cancer Society. Lung cancer signs and symptoms. Updated February 27, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for lung cancer. Updated January 29, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/lung-cancer/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: female breast cancer. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/breast.html
- SEER. SEER*Explorer breast incidence and mortality comparison. Updated July 2, 2025. Accessed March 3, 2026. https://seer.cancer.gov/statistics-network/explorer/application.html
- Susan G. Komen. Male breast cancer. Updated June 3, 2025. Accessed March 3, 2026. https://www.komen.org/breast-cancer/facts-statistics/male-breast-cancer/
- American Cancer Society. Key statistics for breast cancer in men. Updated January 16, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/about/key-statistics.html
- Ruckart PZ, Bove FJ, Shanley E 3rd, et al. Evaluation of contaminated drinking water and male breast cancer at Marine Corps Base Camp Lejeune, North Carolina: a case-control study. Environ Health. 2015;14:74. doi:10.1186/s12940-015-0061-4
- Culell P, Solernou L, Tarazona J, et al. Male breast cancer: a multicentric study. Breast J. 2007;13:213-215. doi:10.1111/j.1524-4741.2007.00412.x
- Giordano SH, Cohen DS, Buzdar AU, et al. Breast carcinoma in men: a population-based study. Cancer. 2004;101:51-57. doi:10.1002/cncr.20312
- American Cancer Society. Signs and symptoms of breast cancer in men. Updated April 27, 2018. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests to help diagnose breast cancer in men. Updated December 20, 2021. Accessed March 3, 2026. https://www.cancer.org/cancer/types/breast-cancer-in-men/detection-diagnosis-staging/how-diagnosed.html
- Evans GF, Anthony T, Turnage RH, et al. The diagnostic accuracy of mammography in the evaluation of male breast disease. Am J Surg. 2001;181:96-100. doi:10.1016/S0002-9610(00)00571-7
- SEER. Cancer stat facts: leukemia. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/leuks.html
- American Cancer Society. Signs and symptoms of acute myeloid leukemia (AML). Updated March 20, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Signs and symptoms of chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for acute myeloid leukemia (AML). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/acute-myeloid-leukemia/detection-diagnosis-staging/how-diagnosed.html
- American Cancer Society. Tests for chronic lymphocytic leukemia (CLL). Updated March 4, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/chronic-lymphocytic-leukemia/detection-diagnosis-staging/how-diagnosed.html
- Giudice V, Selleri C. Aplastic anemia: pathophysiology. Semin Hematol. 2022;59:13-20. doi:10.1053/j.seminhematol.2021.12.002
- National Cancer Institute. Myelodysplastic syndromes treatment (PDQ®)–patient version. Updated October 4, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/myeloproliferative/patient/myelodysplastic-treatment-pdq
- SEER. Cancer statistics review (CSR) 1975-2013: myelodysplastic syndromes. Accessed March 3, 2026. https://seer.cancer.gov/archive/csr/1975_2013/browse_csr.php?sectionSEL=30&pageSEL=sect_30_table.01
- American Cancer Society. Signs and symptoms of myelodysplastic syndrome (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for myelodysplastic syndromes (MDS). Updated November 21, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/myelodysplastic-syndrome/detection-diagnosis-staging/how-diagnosed.html
- SEER. Cancer stat facts: myeloma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/mulmy.html
- American Cancer Society. Signs and symptoms of multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/signs-symptoms.html
- American Cancer Society. Tests for multiple myeloma. Updated February 28, 2025. Accessed March 3, 2026. https://www.cancer.org/cancer/types/multiple-myeloma/detection-diagnosis-staging/testing.html
- Laubach JP. Multiple myeloma: clinical features, laboratory manifestations, and diagnosis. UpToDate. Updated April 28, 2025. Accessed March 3, 2026. https://www.uptodate.com/contents/multiple-myeloma-clinical-features-laboratory-manifestations-and-diagnosis
- Hillengass J, Usmani S, Rajkumar SV, et al. International Myeloma Working Group consensus recommendations on imaging in monoclonal plasma cell disorders. Lancet Oncol. 2019;20:e302-e312. doi:10.1016/S1470-2045(19)30309-2
- SEER. Cancer stat facts: non-Hodgkin lymphoma. Accessed March 3, 2026. https://seer.cancer.gov/statfacts/html/nhl.html
- American Cancer Society. Signs and symptoms of non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/signs-symptoms.html
- National Cancer Institute. Non-Hodgkin lymphoma treatment (PDQ®)–patient version. Updated August 22, 2024. Accessed March 3, 2026. https://www.cancer.gov/types/lymphoma/patient/adult-nhl-treatment-pdq
- American Cancer Society. Tests for non-Hodgkin lymphoma. Updated February 15, 2024. Accessed March 3, 2026. https://www.cancer.org/cancer/types/non-hodgkin-lymphoma/detection-diagnosis-staging/how-diagnosed.html
- College of American Pathologists. Medicare clinical laboratory fee schedule. Published November 2017. Accessed March 3, 2026. https://documents.cap.org/documents/2018-final-medicare-clfs-rates.pdf
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/71270/
- Medicare.gov. Procedure price lookup for outpatient services. Accessed March 3, 2026. https://www.medicare.gov/procedure-price-lookup/cost/74177/
- National Cancer Institute. Cancer trends progress report: financial burden of cancer care. Updated April 2025. Accessed March 3, 2026. https://progressreport.cancer.gov/after/economic_burden
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
Optimizing Care for Veterans at Risk of Cancer From Camp Lejeune Water Exposure
The Integration of Extended Reality in Arthroplasty: Reviewing Technological Progress and Clinical Benefits
The Integration of Extended Reality in Arthroplasty: Reviewing Technological Progress and Clinical Benefits
The introduction of extended reality (XR) to the operating room (OR) has proved promising for enhancing surgical precision and improving patient outcomes. In the field of orthopedic surgery, precise alignment of implants is integral to maintaining functional range of motion and preventing impingement of adjacent neurovascular structures. XR systems have shown promise in arthroplasty including by improving precision and streamlining surgery by allowing surgeons to create 3D preoperative plans that are accessible intraoperatively. This article explores the current applications of XR in arthroplasty, highlights recent advancements and benefits, and describes limitations in comparison to traditional techniques.
Methods
A literature search identified studies involving the use of XR in arthroplasty and current US Food and Drug Administration (FDA)-approved XR systems. Multiple electronic databases were used, including PubMed, Google Scholar, and IEEE Xplore. Search terms included: extended reality, augmented reality, virtual reality, arthroplasty, joint replacement, total knee arthroplasty, total shoulder arthroplasty, and total hip arthroplasty. The study design, intervention details, outcomes, and comparisons with traditional surgical techniques were thematically analyzed, with identification of common ideas associated with XR use in arthroplasty. This narrative report highlights the integration of XR in arthroplasty.
Extended Reality Fundamentals
XR encompasses augmented reality (AR), virtual reality (VR), and mixed reality (MR). AR involves superimposing digitally rendered information and images onto the surgeon’s view of the real world, typically through the use of a headset and smart glasses.1 AR allows the surgeon to move and interact freely within the OR, removing the need for additional screens or devices to display patient information or imaging. VR is a fully immersive simulation using a headset that obstructs the view of the real world but allows the user to move freely within this virtual setting, often with audio or other sensory stimuli. MR combines AR and VR to create a digital model that allows for real-world interaction, with the advantage of adapting information and models in real time.2 Whereas in AR the surgeon can view the data projected from the headset, MR provides the ability to interact with and manipulate the digital content (Figure). Both AR and MR have been adapted for use in the OR, while VR has been adapted for use in surgical planning and training.
Extended Reality Use in Orthopedics
The HipNav system was introduced in 1995 to create preoperative plans that assist surgeons in accurately implanting the acetabular cup during total hip arthroplasty (THA).3 Although not commercially successful, this system spurred surgeons to experiment with XR to improve the accuracy and alignment of orthopedic implants. Systems capable of displaying the desired intraoperative implant placement have flourished, with applications in fracture reduction, arthroplasty, solid tumor resection, and hardware placement.4-7 Accurate alignment has been linked to improvements in patient outcomes.8-10 XR has great potential within the field of arthroplasty, with multiple new systems approved by the FDA and currently available in the US (Table).
Hip Arthroplasty
Orientation of the acetabular cup is a technically challenging part of THA. Accuracy in the anteversion and inclination angles of the acetabular cup is required to maintain implant stability, preserve functional range of motion (ROM), and prevent precocious wear.11,12 Despite preoperative planning, surgeons often overestimate the inclination angle and underestimate anteversion.13 Improper implantation of the acetabular cup can lead to joint instability caused by aseptic loosening, increasing the risk of dislocation and the need for revision surgery.14,15 Dislocations typically present to the emergency department, but primary care practitioners may encounter patients with pain or diminished sensation due to impingement or instability.16
The introduction of XR into the OR has provided the opportunity for real-time navigation and adjustment of the acetabular cup to maximize anteversion and inclination angles. Currently, 2 FDA-approved systems are available for THA: the Zimmer and Surgical Planning Associates HipInsight system, and the Insight Augmented Reality Visualization and Information System (ARVIS). The HipInsight system consists of a hologram projection using the Microsoft HoloLens2 device and optimizes preoperative planning, producing accuracy of anteversion and inclination angles within 3°.17 ARVIS employs existing surgical helmets and 2 mounted tracking cameras to provide navigation intraoperatively. ARVIS has also been approved for use in total knee arthroplasty (TKA) and unicompartmental knee arthroplasty.18
HipInsight has shown utility in increasing the accuracy of acetabular cup placement along with the use of biplanar radiographic scans.19 However, there are no studies validating the efficacy of ARVIS and HipInsight and assessing long-term disease-oriented or patient-oriented outcomes.
Knee Arthroplasty
In the setting of TKA, XR is most effective in ensuring accurate resection of the tibial and femoral components. Achieving the planned femoral coronal, axial, and sagittal angles allows the prosthesis to be on the femoral axis of rotation, improving functional outcomes. XR systems for TKA have been shown to increase the accuracy of distal femoral resection with a limited increase in surgery duration.20,21 For TKA in particular, patients are often less satisfied with the result than surgeons expect.22 Accurate alignment can improve patient satisfaction and reduce return-to-clinic rates for postoperative pain management, a factor that primary care practitioners should consider when recommending a patient for TKA.23
Along with ARVIS, 3 additional XR systems are FDA-approved for use in TKA. The Pixee Medical Knee+ system uses smart glasses and trackers to aid in the positioning of instruments for improved accuracy while allowing real-time navigation.24 The Medacta NextAR Knee’s single-use tracking system allows for intraoperative navigation with the use of AR glasses.25 The Polaris STELLAR Knee uses MR and avoids the need for preoperative imaging by capturing real-time anatomic data.26
The Pixee Medical Knee+ system was commercially available in Europe for several years prior to FDA approval, so more research exists on its efficacy. One study found that the Pixee Medical Knee+ system initially demonstrated an inferior clinical outcome, attributed to the learning curve associated with using the system.27 However, more recent studies have shown its utility in improving alignment, regardless of implant specifications.28,29 The Medacta NextAR Knee system has been shown to improve accuracy of tibial rotation and soft tissue balance and even increase OR efficiency.30,31 The Polaris STELLAR Knee system received FDA approval in 2023; no published research exists on its accuracy and outcomes.26
Shoulder Arthroplasty
Minimally invasive techniques are favored in total shoulder arthroplasty (TSA) due to the vitality of maintaining the surrounding soft tissue to maximize preservation of motility and strength.32 However, this complicates the procedure by decreasing the ability to effectively access and visualize key structures of the shoulder. Accordingly, issues with implant positioning and alignment are more common with TSA than other joint arthroplasties, making XR particularly promising.33 Some studies report that up to 67% of patients experience glenohumeral instability, which can clinically present as weakness, decreased range of motion, and persistent shoulder pain.34,35 The use of preoperative computed tomography to improve understanding of glenoid anatomy and glenohumeral subluxation is becoming increasingly common, and it can be combined with XR to improve accuracy.36,37
Two FDA-approved systems are available. The Stryker Blueprint MR system is used for intraoperative guidance and integration for patient imaging used for preoperative planning. The Medacta NextAR Shoulder system is a parallel of the company’s TKA system. The Stryker Blueprint MR system combines the Microsoft HoloLens 2 headset to display preoperative plans with a secondary display for coordination with the rest of the surgical team.38 Similar to the Medacta NextAR Knee, the Medacta NextAR Shoulder system uses the same single-use tracking system and AR glasses for intraoperative guidance.39
Data on the long-term outcomes of using these systems are still limited, but the Stryker Blueprint MR system has not been shown to accurately predict postoperative ROM.40 Cadaveric studies have demonstrated that the Medacta NextAR Shoulder system can provide accurate inclination, retroversion, entry point, depth, and rotation values based on the preoperative planned values.41,42 However, this accuracy has yet to be confirmed in vivo, and the impact of using XR in TSA on long-term outcomes is still unknown.
Challenges and Limitations
Though XR has proven to be promising in arthroplasty, several limitations regarding widespread implementation exist. In particular, there is a steep learning curve associated with the use of XR systems, which can cause increased operative time and even initial inferior outcomes, as demonstrated with the Pixee Medical Knee+ system. The need for extensive practice and training prior to use could delay widespread adoption and may cause discrepancies in surgical outcomes. Unfamiliarity with the system and technological difficulties that may require troubleshooting can also increase operative time, particularly for surgeons new to using the XR system. Though intraoperative navigation is expected to improve accuracy of implant alignment, its added complexity may also result in longer surgeries.
In addition to the steep learning curve and increased operative time, there is a high upfront cost associated with XR systems. Exact costs of XR systems are not typically disclosed, but available estimates suggest an average sales price of about $1000 per case. Given the proprietary nature of these technologies, publicly available cost data are limited, making it challenging to fully assess the financial burden on health care institutions. Though some systems, such as ARVIS, can be integrated with existing surgical helmets, many require the purchase of AR glasses and secondary displays. This can cause further variation in the total expense for each system. In low-resource settings, this represents a significant challenge to widespread implementation. To justify this cost, additional research on long-term patient outcomes is needed to ensure the benefits of XR systems outweigh the cost.
Although early studies on XR systems in arthroplasty have shown improvements in precision and short-term outcomes, long-term data regarding effectiveness remains. Even systems such as ARVIS and HipInsight have limited long-term follow-up, making it difficult to assess whether the improved accuracy with these XR systems translates into improved patient outcomes compared with traditional arthroplasty.
CONCLUSIONS
XR technologies have shown significant potential in enhancing precision and patient outcomes. Through the integration of XR in the OR, surgeons can visualize preoperative plans and even make intraoperative changes, with the benefit of improving implant alignment.
There are some disadvantages to its use, however, including high cost and increased operative time. Despite this, the integration of XR into surgical practice can deliver more precise implant alignment and address other challenges faced with conventional techniques. As these technologies evolve and studies on long-term outcomes validate their utility, XR has the potential to transform the field of arthroplasty.
Azuma RT. A survey of augmented reality. Presence-Teleop Virt. 1997;6:355-385. doi:10.1162/pres.1997.6.4.355
Speicher M, Hall BD, Nebeling M. What is Mixed Reality? In: Proceedings of the 2019 CHI Conference on Human Factors in Computing Systems. Association for Computing Machinery; 2019:1-15. doi:10.1145/3290605.3300767
Digioia AM, Jaramaz B, Nikou C, et al. Surgical navigation for total hip replacement with the use of hipnav. Oper Tech Orthop. 2000;10:3-8. doi:10.1016/S1048-6666(00)80036-1
Ogawa H, Hasegawa S, Tsukada S, et al. A pilot study of augmented reality technology applied to the acetabular cup placement during total hip arthroplasty. J Arthroplasty. 2018;33:1833-1837. doi:10.1016/j.arth.2018.01.067
Shen F, Chen B, Guo Q, et al. Augmented reality patient-specific reconstruction plate design for pelvic and acetabular fracture surgery. Int J CARS. 2013;8:169-179. doi:10.1007/s11548-012-0775-5
Cho HS, Park YK, Gupta S, et al. Augmented reality in bone tumour resection: an experimental study. Bone Joint Res. 2017;6:137-143. doi:10.1302/2046-3758.63.bjr-2016-0289.r1
Wu X, Liu R, Yu J, et al. Mixed reality technology launches in orthopedic surgery for comprehensive preoperative management of complicated cervical fractures. Surg Innov. 2018;25:421-422. doi:10.1177/1553350618761758
Dossett HG, Arthur JR, Makovicka JL, et al. A randomized controlled trial of kinematically and mechanically aligned total knee arthroplasties: long-term follow-up. J Arthroplasty. 2023;38:S209-S214. doi:10.1016/j.arth.2023.03.065
Kazarian GS, Haddad FS, Donaldson MJ, et al. Implant malalignment may be a risk factor for poor patient-reported outcomes measures (PROMs) following total knee arthroplasty (TKA). J Arthroplasty. 2022;37:S129-S133. doi:10.1016/j.arth.2022.02.087
Peng Y, Arauz P, An S, et al. Does component alignment affect patient reported outcomes following bicruciate retaining total knee arthroplasty? An in vivo three-dimensional analysis. J Knee Surg. 2020;33:798-803. doi:10.1055/s-0039-1688500
D’Lima DD, Urquhart AG, Buehler KO, et al. The effect of the orientation of the acetabular and femoral components on the range of motion of the hip at different head-neck ratios. J Bone Joint Surg Am. 2000;82:315-321. doi:10.2106/00004623-200003000-00003
Yamaguchi M, Akisue T, Bauer TW, et al. The spatial location of impingement in total hip arthroplasty. J Arthroplasty. 2000;15:305-313. doi:10.1016/s0883-5403(00)90601-6
Grammatopoulos G, Alvand A, Monk AP, et al. Surgeons’ accuracy in achieving their desired acetabular component orientation. J Bone Joint Surg. 2016;98:e72. doi:10.2106/JBJS.15.01080
Kennedy JG, Rogers WB, Soffe KE, et al. Effect of acetabular component orientation on recurrent dislocation, pelvic osteolysis, polyethylene wear, and component migration. J Arthroplasty. 1998;13:530-534. doi:10.1016/S0883-5403(98)90052-3
Del Schutte H, Lipman AJ, Bannar SM, et al. Effects of acetabular abduction on cup wear rates in total hip arthroplasty. J Arthroplasty. 1998;13:621-626. doi:10.1016/S0883-5403(98)80003-X
Aresti N, Kassam J, Bartlett D, et al. Primary care management of postoperative shoulder, hip, and knee arthroplasty. BMJ. 2017;359:j4431. doi:10.1136/bmj.j4431
HipInsightTM System. Zimmer Biomet. Accessed September 3, 2025. https://www.zimmerbiomet.com/en/products-and-solutions/zb-edge/mixed-reality-portfolio/hipinsight-system.html
ARVIS. Insight Medical Systems. Accessed September 3, 2025. https://www.insightmedsys.com/arvis
Sun DC, Murphy WS, Amundson AJ, et al. Validation of a novel method of measuring cup orientation using biplanar simultaneous radiographic images. J Arthroplasty. 2023;38:S252-S256. doi:10.1016/j.arth.2023.04.011
Tsukada S, Ogawa H, Nishino M, et al. Augmented reality-assisted femoral bone resection in total knee arthroplasty. JBJS Open Access. 2021;6:e21.00001. doi:10.2106/JBJS.OA.21.00001
Castellarin G, Bori E, Barbieux E, et al. Is total knee arthroplasty surgical performance enhanced using augmented reality? A single-center study on 76 consecutive patients. J Arthroplasty. 2024;39:332-335. doi:10.1016/j.arth.2023.08.013
Choi YJ, Ra HJ. Patient satisfaction after total knee arthroplasty. Knee Surg Relat Res. 2016;28:1. doi:10.5792/ksrr.2016.28.1.1
Hazratwala K, Gouk C, Wilkinson MPR, et al. Navigated functional alignment total knee arthroplasty achieves reliable, reproducible and accurate results with high patient satisfaction. Knee Surg Sports Traumatol Arthrosc. 2023;31:3861-3870. doi:10.1007/s00167-023-07327-w
Knee+. Pixee Medical. Accessed September 3, 2025. https://www.pixee-medical.com/en/products/knee-nexsight/
KNEE | NEXTAR. Nextar. Accessed September 3, 2025. https://nextar.medacta.com/knee
POLARIS AR receives clearance from the U.S. Food and Drug Administration for STELLAR Knee. News release. PRNewswire. November 3, 2023. Accessed September 3, 2025. https://www.prnewswire.com/news-releases/polarisar-receives-clearance-from-the-us-food-and-drug-administration-for-stellar-knee-301976747.html
van Overschelde P, Vansintjan P, Byn P, Lapierre C, van Lysebettens W. Does augmented reality improve clinical outcome in TKA? A prospective observational report. In: The 20th Annual Meeting of the International Society for Computer Assisted Orthopaedic Surgery. 2022:170-174.
Sakellariou E, Alevrogiannis P, Alevrogianni F, et al. Single-center experience with Knee+TM augmented reality navigation system in primary total knee arthroplasty. World J Orthop. 2024;15:247-256. doi:10.5312/wjo.v15.i3.247
León-Muñoz VJ, Moya-Angeler J, López-López M, et al. Integration of square fiducial markers in patient-specific instrumentation and their applicability in knee surgery. J Pers Med. 2023;13:727. doi:10.3390/jpm13050727
Fucentese SF, Koch PP. A novel augmented reality-based surgical guidance system for total knee arthroplasty. Arch Orthop Trauma Surg. 2021;141:2227-2233. doi:10.1007/s00402-021-04204-4
Sabatini L, Ascani D, Vezza D, et al. Novel surgical technique for total knee arthroplasty integrating kinematic alignment and real-time elongation of the ligaments using the NextAR system. J Pers Med. 2024;14:794. doi:10.3390/jpm14080794
Daher M, Ghanimeh J, Otayek J, et al. Augmented reality and shoulder replacement: a state-of-the-art review article. JSES Rev Rep Tech. 2023;3:274-278. doi:10.1016/j.xrrt.2023.01.008
Atmani H, Merienne F, Fofi D, et al. Computer aided surgery system for shoulder prosthesis placement. Comput Aided Surg. 2007;12:60-70. doi:10.3109/10929080701210832
Eichinger JK, Galvin JW. Management of complications after total shoulder arthroplasty. Curr Rev Musculoskelet Med. 2015;8:83-91. doi:10.1007/s12178-014-9251-x
Bonnevialle N, Melis B, Neyton L, et al. Aseptic glenoid loosening or failure in total shoulder arthroplasty: revision with glenoid reimplantation. J Shoulder Elbow Surg. 2013;22:745-751. doi:10.1016/j.jse.2012.08.009
Erickson BJ, Chalmers PN, Denard P, et al. Does commercially available shoulder arthroplasty preoperative planning software agree with surgeon measurements of version, inclination, and subluxation? J Shoulder Elbow Surg. 2021;30:413-420. doi:10.1016/j.jse.2020.05.027
Werner BS, Hudek R, Burkhart KJ, et al. The influence of three-dimensional planning on decision-making in total shoulder arthroplasty. J Shoulder Elbow Surg. 2017;26:1477-1483. doi:10.1016/j.jse.2017.01.006
Blueprint. Stryker. Updated August 2025. Accessed September 3, 2025. https://www.stryker.com/us/en/trauma-and-extremities/products/blueprint.html
NextAR Shoulder. Medacta. Accessed September 3, 2025. https://nextar.medacta.com/shoulder
Baumgarten KM. Accuracy of Blueprint software in predicting range of motion 1 year after reverse total shoulder arthroplasty. J Shoulder Elbow Surg. 2023;32:1088-1094. doi:10.1016/j.jse.2022.12.009
Rojas JT, Jost B, Zipeto C, et al. Glenoid component placement in reverse shoulder arthroplasty assisted with augmented reality through a head-mounted display leads to low deviation between planned and postoperative parameters. J Shoulder Elbow Surg. 2023;32:e587-e596. doi:10.1016/j.jse.2023.05.002
Dey Hazra RO, Paksoy A, Imiolczyk JP, et al. Augmented reality–assisted intraoperative navigation increases precision of glenoid inclination in reverse shoulder arthroplasty. J Shoulder Elbow Surg. 2025;34(2):577-583. doi:10.1016/j.jse.2024.05.039
The introduction of extended reality (XR) to the operating room (OR) has proved promising for enhancing surgical precision and improving patient outcomes. In the field of orthopedic surgery, precise alignment of implants is integral to maintaining functional range of motion and preventing impingement of adjacent neurovascular structures. XR systems have shown promise in arthroplasty including by improving precision and streamlining surgery by allowing surgeons to create 3D preoperative plans that are accessible intraoperatively. This article explores the current applications of XR in arthroplasty, highlights recent advancements and benefits, and describes limitations in comparison to traditional techniques.
Methods
A literature search identified studies involving the use of XR in arthroplasty and current US Food and Drug Administration (FDA)-approved XR systems. Multiple electronic databases were used, including PubMed, Google Scholar, and IEEE Xplore. Search terms included: extended reality, augmented reality, virtual reality, arthroplasty, joint replacement, total knee arthroplasty, total shoulder arthroplasty, and total hip arthroplasty. The study design, intervention details, outcomes, and comparisons with traditional surgical techniques were thematically analyzed, with identification of common ideas associated with XR use in arthroplasty. This narrative report highlights the integration of XR in arthroplasty.
Extended Reality Fundamentals
XR encompasses augmented reality (AR), virtual reality (VR), and mixed reality (MR). AR involves superimposing digitally rendered information and images onto the surgeon’s view of the real world, typically through the use of a headset and smart glasses.1 AR allows the surgeon to move and interact freely within the OR, removing the need for additional screens or devices to display patient information or imaging. VR is a fully immersive simulation using a headset that obstructs the view of the real world but allows the user to move freely within this virtual setting, often with audio or other sensory stimuli. MR combines AR and VR to create a digital model that allows for real-world interaction, with the advantage of adapting information and models in real time.2 Whereas in AR the surgeon can view the data projected from the headset, MR provides the ability to interact with and manipulate the digital content (Figure). Both AR and MR have been adapted for use in the OR, while VR has been adapted for use in surgical planning and training.
Extended Reality Use in Orthopedics
The HipNav system was introduced in 1995 to create preoperative plans that assist surgeons in accurately implanting the acetabular cup during total hip arthroplasty (THA).3 Although not commercially successful, this system spurred surgeons to experiment with XR to improve the accuracy and alignment of orthopedic implants. Systems capable of displaying the desired intraoperative implant placement have flourished, with applications in fracture reduction, arthroplasty, solid tumor resection, and hardware placement.4-7 Accurate alignment has been linked to improvements in patient outcomes.8-10 XR has great potential within the field of arthroplasty, with multiple new systems approved by the FDA and currently available in the US (Table).
Hip Arthroplasty
Orientation of the acetabular cup is a technically challenging part of THA. Accuracy in the anteversion and inclination angles of the acetabular cup is required to maintain implant stability, preserve functional range of motion (ROM), and prevent precocious wear.11,12 Despite preoperative planning, surgeons often overestimate the inclination angle and underestimate anteversion.13 Improper implantation of the acetabular cup can lead to joint instability caused by aseptic loosening, increasing the risk of dislocation and the need for revision surgery.14,15 Dislocations typically present to the emergency department, but primary care practitioners may encounter patients with pain or diminished sensation due to impingement or instability.16
The introduction of XR into the OR has provided the opportunity for real-time navigation and adjustment of the acetabular cup to maximize anteversion and inclination angles. Currently, 2 FDA-approved systems are available for THA: the Zimmer and Surgical Planning Associates HipInsight system, and the Insight Augmented Reality Visualization and Information System (ARVIS). The HipInsight system consists of a hologram projection using the Microsoft HoloLens2 device and optimizes preoperative planning, producing accuracy of anteversion and inclination angles within 3°.17 ARVIS employs existing surgical helmets and 2 mounted tracking cameras to provide navigation intraoperatively. ARVIS has also been approved for use in total knee arthroplasty (TKA) and unicompartmental knee arthroplasty.18
HipInsight has shown utility in increasing the accuracy of acetabular cup placement along with the use of biplanar radiographic scans.19 However, there are no studies validating the efficacy of ARVIS and HipInsight and assessing long-term disease-oriented or patient-oriented outcomes.
Knee Arthroplasty
In the setting of TKA, XR is most effective in ensuring accurate resection of the tibial and femoral components. Achieving the planned femoral coronal, axial, and sagittal angles allows the prosthesis to be on the femoral axis of rotation, improving functional outcomes. XR systems for TKA have been shown to increase the accuracy of distal femoral resection with a limited increase in surgery duration.20,21 For TKA in particular, patients are often less satisfied with the result than surgeons expect.22 Accurate alignment can improve patient satisfaction and reduce return-to-clinic rates for postoperative pain management, a factor that primary care practitioners should consider when recommending a patient for TKA.23
Along with ARVIS, 3 additional XR systems are FDA-approved for use in TKA. The Pixee Medical Knee+ system uses smart glasses and trackers to aid in the positioning of instruments for improved accuracy while allowing real-time navigation.24 The Medacta NextAR Knee’s single-use tracking system allows for intraoperative navigation with the use of AR glasses.25 The Polaris STELLAR Knee uses MR and avoids the need for preoperative imaging by capturing real-time anatomic data.26
The Pixee Medical Knee+ system was commercially available in Europe for several years prior to FDA approval, so more research exists on its efficacy. One study found that the Pixee Medical Knee+ system initially demonstrated an inferior clinical outcome, attributed to the learning curve associated with using the system.27 However, more recent studies have shown its utility in improving alignment, regardless of implant specifications.28,29 The Medacta NextAR Knee system has been shown to improve accuracy of tibial rotation and soft tissue balance and even increase OR efficiency.30,31 The Polaris STELLAR Knee system received FDA approval in 2023; no published research exists on its accuracy and outcomes.26
Shoulder Arthroplasty
Minimally invasive techniques are favored in total shoulder arthroplasty (TSA) due to the vitality of maintaining the surrounding soft tissue to maximize preservation of motility and strength.32 However, this complicates the procedure by decreasing the ability to effectively access and visualize key structures of the shoulder. Accordingly, issues with implant positioning and alignment are more common with TSA than other joint arthroplasties, making XR particularly promising.33 Some studies report that up to 67% of patients experience glenohumeral instability, which can clinically present as weakness, decreased range of motion, and persistent shoulder pain.34,35 The use of preoperative computed tomography to improve understanding of glenoid anatomy and glenohumeral subluxation is becoming increasingly common, and it can be combined with XR to improve accuracy.36,37
Two FDA-approved systems are available. The Stryker Blueprint MR system is used for intraoperative guidance and integration for patient imaging used for preoperative planning. The Medacta NextAR Shoulder system is a parallel of the company’s TKA system. The Stryker Blueprint MR system combines the Microsoft HoloLens 2 headset to display preoperative plans with a secondary display for coordination with the rest of the surgical team.38 Similar to the Medacta NextAR Knee, the Medacta NextAR Shoulder system uses the same single-use tracking system and AR glasses for intraoperative guidance.39
Data on the long-term outcomes of using these systems are still limited, but the Stryker Blueprint MR system has not been shown to accurately predict postoperative ROM.40 Cadaveric studies have demonstrated that the Medacta NextAR Shoulder system can provide accurate inclination, retroversion, entry point, depth, and rotation values based on the preoperative planned values.41,42 However, this accuracy has yet to be confirmed in vivo, and the impact of using XR in TSA on long-term outcomes is still unknown.
Challenges and Limitations
Though XR has proven to be promising in arthroplasty, several limitations regarding widespread implementation exist. In particular, there is a steep learning curve associated with the use of XR systems, which can cause increased operative time and even initial inferior outcomes, as demonstrated with the Pixee Medical Knee+ system. The need for extensive practice and training prior to use could delay widespread adoption and may cause discrepancies in surgical outcomes. Unfamiliarity with the system and technological difficulties that may require troubleshooting can also increase operative time, particularly for surgeons new to using the XR system. Though intraoperative navigation is expected to improve accuracy of implant alignment, its added complexity may also result in longer surgeries.
In addition to the steep learning curve and increased operative time, there is a high upfront cost associated with XR systems. Exact costs of XR systems are not typically disclosed, but available estimates suggest an average sales price of about $1000 per case. Given the proprietary nature of these technologies, publicly available cost data are limited, making it challenging to fully assess the financial burden on health care institutions. Though some systems, such as ARVIS, can be integrated with existing surgical helmets, many require the purchase of AR glasses and secondary displays. This can cause further variation in the total expense for each system. In low-resource settings, this represents a significant challenge to widespread implementation. To justify this cost, additional research on long-term patient outcomes is needed to ensure the benefits of XR systems outweigh the cost.
Although early studies on XR systems in arthroplasty have shown improvements in precision and short-term outcomes, long-term data regarding effectiveness remains. Even systems such as ARVIS and HipInsight have limited long-term follow-up, making it difficult to assess whether the improved accuracy with these XR systems translates into improved patient outcomes compared with traditional arthroplasty.
CONCLUSIONS
XR technologies have shown significant potential in enhancing precision and patient outcomes. Through the integration of XR in the OR, surgeons can visualize preoperative plans and even make intraoperative changes, with the benefit of improving implant alignment.
There are some disadvantages to its use, however, including high cost and increased operative time. Despite this, the integration of XR into surgical practice can deliver more precise implant alignment and address other challenges faced with conventional techniques. As these technologies evolve and studies on long-term outcomes validate their utility, XR has the potential to transform the field of arthroplasty.
The introduction of extended reality (XR) to the operating room (OR) has proved promising for enhancing surgical precision and improving patient outcomes. In the field of orthopedic surgery, precise alignment of implants is integral to maintaining functional range of motion and preventing impingement of adjacent neurovascular structures. XR systems have shown promise in arthroplasty including by improving precision and streamlining surgery by allowing surgeons to create 3D preoperative plans that are accessible intraoperatively. This article explores the current applications of XR in arthroplasty, highlights recent advancements and benefits, and describes limitations in comparison to traditional techniques.
Methods
A literature search identified studies involving the use of XR in arthroplasty and current US Food and Drug Administration (FDA)-approved XR systems. Multiple electronic databases were used, including PubMed, Google Scholar, and IEEE Xplore. Search terms included: extended reality, augmented reality, virtual reality, arthroplasty, joint replacement, total knee arthroplasty, total shoulder arthroplasty, and total hip arthroplasty. The study design, intervention details, outcomes, and comparisons with traditional surgical techniques were thematically analyzed, with identification of common ideas associated with XR use in arthroplasty. This narrative report highlights the integration of XR in arthroplasty.
Extended Reality Fundamentals
XR encompasses augmented reality (AR), virtual reality (VR), and mixed reality (MR). AR involves superimposing digitally rendered information and images onto the surgeon’s view of the real world, typically through the use of a headset and smart glasses.1 AR allows the surgeon to move and interact freely within the OR, removing the need for additional screens or devices to display patient information or imaging. VR is a fully immersive simulation using a headset that obstructs the view of the real world but allows the user to move freely within this virtual setting, often with audio or other sensory stimuli. MR combines AR and VR to create a digital model that allows for real-world interaction, with the advantage of adapting information and models in real time.2 Whereas in AR the surgeon can view the data projected from the headset, MR provides the ability to interact with and manipulate the digital content (Figure). Both AR and MR have been adapted for use in the OR, while VR has been adapted for use in surgical planning and training.
Extended Reality Use in Orthopedics
The HipNav system was introduced in 1995 to create preoperative plans that assist surgeons in accurately implanting the acetabular cup during total hip arthroplasty (THA).3 Although not commercially successful, this system spurred surgeons to experiment with XR to improve the accuracy and alignment of orthopedic implants. Systems capable of displaying the desired intraoperative implant placement have flourished, with applications in fracture reduction, arthroplasty, solid tumor resection, and hardware placement.4-7 Accurate alignment has been linked to improvements in patient outcomes.8-10 XR has great potential within the field of arthroplasty, with multiple new systems approved by the FDA and currently available in the US (Table).
Hip Arthroplasty
Orientation of the acetabular cup is a technically challenging part of THA. Accuracy in the anteversion and inclination angles of the acetabular cup is required to maintain implant stability, preserve functional range of motion (ROM), and prevent precocious wear.11,12 Despite preoperative planning, surgeons often overestimate the inclination angle and underestimate anteversion.13 Improper implantation of the acetabular cup can lead to joint instability caused by aseptic loosening, increasing the risk of dislocation and the need for revision surgery.14,15 Dislocations typically present to the emergency department, but primary care practitioners may encounter patients with pain or diminished sensation due to impingement or instability.16
The introduction of XR into the OR has provided the opportunity for real-time navigation and adjustment of the acetabular cup to maximize anteversion and inclination angles. Currently, 2 FDA-approved systems are available for THA: the Zimmer and Surgical Planning Associates HipInsight system, and the Insight Augmented Reality Visualization and Information System (ARVIS). The HipInsight system consists of a hologram projection using the Microsoft HoloLens2 device and optimizes preoperative planning, producing accuracy of anteversion and inclination angles within 3°.17 ARVIS employs existing surgical helmets and 2 mounted tracking cameras to provide navigation intraoperatively. ARVIS has also been approved for use in total knee arthroplasty (TKA) and unicompartmental knee arthroplasty.18
HipInsight has shown utility in increasing the accuracy of acetabular cup placement along with the use of biplanar radiographic scans.19 However, there are no studies validating the efficacy of ARVIS and HipInsight and assessing long-term disease-oriented or patient-oriented outcomes.
Knee Arthroplasty
In the setting of TKA, XR is most effective in ensuring accurate resection of the tibial and femoral components. Achieving the planned femoral coronal, axial, and sagittal angles allows the prosthesis to be on the femoral axis of rotation, improving functional outcomes. XR systems for TKA have been shown to increase the accuracy of distal femoral resection with a limited increase in surgery duration.20,21 For TKA in particular, patients are often less satisfied with the result than surgeons expect.22 Accurate alignment can improve patient satisfaction and reduce return-to-clinic rates for postoperative pain management, a factor that primary care practitioners should consider when recommending a patient for TKA.23
Along with ARVIS, 3 additional XR systems are FDA-approved for use in TKA. The Pixee Medical Knee+ system uses smart glasses and trackers to aid in the positioning of instruments for improved accuracy while allowing real-time navigation.24 The Medacta NextAR Knee’s single-use tracking system allows for intraoperative navigation with the use of AR glasses.25 The Polaris STELLAR Knee uses MR and avoids the need for preoperative imaging by capturing real-time anatomic data.26
The Pixee Medical Knee+ system was commercially available in Europe for several years prior to FDA approval, so more research exists on its efficacy. One study found that the Pixee Medical Knee+ system initially demonstrated an inferior clinical outcome, attributed to the learning curve associated with using the system.27 However, more recent studies have shown its utility in improving alignment, regardless of implant specifications.28,29 The Medacta NextAR Knee system has been shown to improve accuracy of tibial rotation and soft tissue balance and even increase OR efficiency.30,31 The Polaris STELLAR Knee system received FDA approval in 2023; no published research exists on its accuracy and outcomes.26
Shoulder Arthroplasty
Minimally invasive techniques are favored in total shoulder arthroplasty (TSA) due to the vitality of maintaining the surrounding soft tissue to maximize preservation of motility and strength.32 However, this complicates the procedure by decreasing the ability to effectively access and visualize key structures of the shoulder. Accordingly, issues with implant positioning and alignment are more common with TSA than other joint arthroplasties, making XR particularly promising.33 Some studies report that up to 67% of patients experience glenohumeral instability, which can clinically present as weakness, decreased range of motion, and persistent shoulder pain.34,35 The use of preoperative computed tomography to improve understanding of glenoid anatomy and glenohumeral subluxation is becoming increasingly common, and it can be combined with XR to improve accuracy.36,37
Two FDA-approved systems are available. The Stryker Blueprint MR system is used for intraoperative guidance and integration for patient imaging used for preoperative planning. The Medacta NextAR Shoulder system is a parallel of the company’s TKA system. The Stryker Blueprint MR system combines the Microsoft HoloLens 2 headset to display preoperative plans with a secondary display for coordination with the rest of the surgical team.38 Similar to the Medacta NextAR Knee, the Medacta NextAR Shoulder system uses the same single-use tracking system and AR glasses for intraoperative guidance.39
Data on the long-term outcomes of using these systems are still limited, but the Stryker Blueprint MR system has not been shown to accurately predict postoperative ROM.40 Cadaveric studies have demonstrated that the Medacta NextAR Shoulder system can provide accurate inclination, retroversion, entry point, depth, and rotation values based on the preoperative planned values.41,42 However, this accuracy has yet to be confirmed in vivo, and the impact of using XR in TSA on long-term outcomes is still unknown.
Challenges and Limitations
Though XR has proven to be promising in arthroplasty, several limitations regarding widespread implementation exist. In particular, there is a steep learning curve associated with the use of XR systems, which can cause increased operative time and even initial inferior outcomes, as demonstrated with the Pixee Medical Knee+ system. The need for extensive practice and training prior to use could delay widespread adoption and may cause discrepancies in surgical outcomes. Unfamiliarity with the system and technological difficulties that may require troubleshooting can also increase operative time, particularly for surgeons new to using the XR system. Though intraoperative navigation is expected to improve accuracy of implant alignment, its added complexity may also result in longer surgeries.
In addition to the steep learning curve and increased operative time, there is a high upfront cost associated with XR systems. Exact costs of XR systems are not typically disclosed, but available estimates suggest an average sales price of about $1000 per case. Given the proprietary nature of these technologies, publicly available cost data are limited, making it challenging to fully assess the financial burden on health care institutions. Though some systems, such as ARVIS, can be integrated with existing surgical helmets, many require the purchase of AR glasses and secondary displays. This can cause further variation in the total expense for each system. In low-resource settings, this represents a significant challenge to widespread implementation. To justify this cost, additional research on long-term patient outcomes is needed to ensure the benefits of XR systems outweigh the cost.
Although early studies on XR systems in arthroplasty have shown improvements in precision and short-term outcomes, long-term data regarding effectiveness remains. Even systems such as ARVIS and HipInsight have limited long-term follow-up, making it difficult to assess whether the improved accuracy with these XR systems translates into improved patient outcomes compared with traditional arthroplasty.
CONCLUSIONS
XR technologies have shown significant potential in enhancing precision and patient outcomes. Through the integration of XR in the OR, surgeons can visualize preoperative plans and even make intraoperative changes, with the benefit of improving implant alignment.
There are some disadvantages to its use, however, including high cost and increased operative time. Despite this, the integration of XR into surgical practice can deliver more precise implant alignment and address other challenges faced with conventional techniques. As these technologies evolve and studies on long-term outcomes validate their utility, XR has the potential to transform the field of arthroplasty.
Azuma RT. A survey of augmented reality. Presence-Teleop Virt. 1997;6:355-385. doi:10.1162/pres.1997.6.4.355
Speicher M, Hall BD, Nebeling M. What is Mixed Reality? In: Proceedings of the 2019 CHI Conference on Human Factors in Computing Systems. Association for Computing Machinery; 2019:1-15. doi:10.1145/3290605.3300767
Digioia AM, Jaramaz B, Nikou C, et al. Surgical navigation for total hip replacement with the use of hipnav. Oper Tech Orthop. 2000;10:3-8. doi:10.1016/S1048-6666(00)80036-1
Ogawa H, Hasegawa S, Tsukada S, et al. A pilot study of augmented reality technology applied to the acetabular cup placement during total hip arthroplasty. J Arthroplasty. 2018;33:1833-1837. doi:10.1016/j.arth.2018.01.067
Shen F, Chen B, Guo Q, et al. Augmented reality patient-specific reconstruction plate design for pelvic and acetabular fracture surgery. Int J CARS. 2013;8:169-179. doi:10.1007/s11548-012-0775-5
Cho HS, Park YK, Gupta S, et al. Augmented reality in bone tumour resection: an experimental study. Bone Joint Res. 2017;6:137-143. doi:10.1302/2046-3758.63.bjr-2016-0289.r1
Wu X, Liu R, Yu J, et al. Mixed reality technology launches in orthopedic surgery for comprehensive preoperative management of complicated cervical fractures. Surg Innov. 2018;25:421-422. doi:10.1177/1553350618761758
Dossett HG, Arthur JR, Makovicka JL, et al. A randomized controlled trial of kinematically and mechanically aligned total knee arthroplasties: long-term follow-up. J Arthroplasty. 2023;38:S209-S214. doi:10.1016/j.arth.2023.03.065
Kazarian GS, Haddad FS, Donaldson MJ, et al. Implant malalignment may be a risk factor for poor patient-reported outcomes measures (PROMs) following total knee arthroplasty (TKA). J Arthroplasty. 2022;37:S129-S133. doi:10.1016/j.arth.2022.02.087
Peng Y, Arauz P, An S, et al. Does component alignment affect patient reported outcomes following bicruciate retaining total knee arthroplasty? An in vivo three-dimensional analysis. J Knee Surg. 2020;33:798-803. doi:10.1055/s-0039-1688500
D’Lima DD, Urquhart AG, Buehler KO, et al. The effect of the orientation of the acetabular and femoral components on the range of motion of the hip at different head-neck ratios. J Bone Joint Surg Am. 2000;82:315-321. doi:10.2106/00004623-200003000-00003
Yamaguchi M, Akisue T, Bauer TW, et al. The spatial location of impingement in total hip arthroplasty. J Arthroplasty. 2000;15:305-313. doi:10.1016/s0883-5403(00)90601-6
Grammatopoulos G, Alvand A, Monk AP, et al. Surgeons’ accuracy in achieving their desired acetabular component orientation. J Bone Joint Surg. 2016;98:e72. doi:10.2106/JBJS.15.01080
Kennedy JG, Rogers WB, Soffe KE, et al. Effect of acetabular component orientation on recurrent dislocation, pelvic osteolysis, polyethylene wear, and component migration. J Arthroplasty. 1998;13:530-534. doi:10.1016/S0883-5403(98)90052-3
Del Schutte H, Lipman AJ, Bannar SM, et al. Effects of acetabular abduction on cup wear rates in total hip arthroplasty. J Arthroplasty. 1998;13:621-626. doi:10.1016/S0883-5403(98)80003-X
Aresti N, Kassam J, Bartlett D, et al. Primary care management of postoperative shoulder, hip, and knee arthroplasty. BMJ. 2017;359:j4431. doi:10.1136/bmj.j4431
HipInsightTM System. Zimmer Biomet. Accessed September 3, 2025. https://www.zimmerbiomet.com/en/products-and-solutions/zb-edge/mixed-reality-portfolio/hipinsight-system.html
ARVIS. Insight Medical Systems. Accessed September 3, 2025. https://www.insightmedsys.com/arvis
Sun DC, Murphy WS, Amundson AJ, et al. Validation of a novel method of measuring cup orientation using biplanar simultaneous radiographic images. J Arthroplasty. 2023;38:S252-S256. doi:10.1016/j.arth.2023.04.011
Tsukada S, Ogawa H, Nishino M, et al. Augmented reality-assisted femoral bone resection in total knee arthroplasty. JBJS Open Access. 2021;6:e21.00001. doi:10.2106/JBJS.OA.21.00001
Castellarin G, Bori E, Barbieux E, et al. Is total knee arthroplasty surgical performance enhanced using augmented reality? A single-center study on 76 consecutive patients. J Arthroplasty. 2024;39:332-335. doi:10.1016/j.arth.2023.08.013
Choi YJ, Ra HJ. Patient satisfaction after total knee arthroplasty. Knee Surg Relat Res. 2016;28:1. doi:10.5792/ksrr.2016.28.1.1
Hazratwala K, Gouk C, Wilkinson MPR, et al. Navigated functional alignment total knee arthroplasty achieves reliable, reproducible and accurate results with high patient satisfaction. Knee Surg Sports Traumatol Arthrosc. 2023;31:3861-3870. doi:10.1007/s00167-023-07327-w
Knee+. Pixee Medical. Accessed September 3, 2025. https://www.pixee-medical.com/en/products/knee-nexsight/
KNEE | NEXTAR. Nextar. Accessed September 3, 2025. https://nextar.medacta.com/knee
POLARIS AR receives clearance from the U.S. Food and Drug Administration for STELLAR Knee. News release. PRNewswire. November 3, 2023. Accessed September 3, 2025. https://www.prnewswire.com/news-releases/polarisar-receives-clearance-from-the-us-food-and-drug-administration-for-stellar-knee-301976747.html
van Overschelde P, Vansintjan P, Byn P, Lapierre C, van Lysebettens W. Does augmented reality improve clinical outcome in TKA? A prospective observational report. In: The 20th Annual Meeting of the International Society for Computer Assisted Orthopaedic Surgery. 2022:170-174.
Sakellariou E, Alevrogiannis P, Alevrogianni F, et al. Single-center experience with Knee+TM augmented reality navigation system in primary total knee arthroplasty. World J Orthop. 2024;15:247-256. doi:10.5312/wjo.v15.i3.247
León-Muñoz VJ, Moya-Angeler J, López-López M, et al. Integration of square fiducial markers in patient-specific instrumentation and their applicability in knee surgery. J Pers Med. 2023;13:727. doi:10.3390/jpm13050727
Fucentese SF, Koch PP. A novel augmented reality-based surgical guidance system for total knee arthroplasty. Arch Orthop Trauma Surg. 2021;141:2227-2233. doi:10.1007/s00402-021-04204-4
Sabatini L, Ascani D, Vezza D, et al. Novel surgical technique for total knee arthroplasty integrating kinematic alignment and real-time elongation of the ligaments using the NextAR system. J Pers Med. 2024;14:794. doi:10.3390/jpm14080794
Daher M, Ghanimeh J, Otayek J, et al. Augmented reality and shoulder replacement: a state-of-the-art review article. JSES Rev Rep Tech. 2023;3:274-278. doi:10.1016/j.xrrt.2023.01.008
Atmani H, Merienne F, Fofi D, et al. Computer aided surgery system for shoulder prosthesis placement. Comput Aided Surg. 2007;12:60-70. doi:10.3109/10929080701210832
Eichinger JK, Galvin JW. Management of complications after total shoulder arthroplasty. Curr Rev Musculoskelet Med. 2015;8:83-91. doi:10.1007/s12178-014-9251-x
Bonnevialle N, Melis B, Neyton L, et al. Aseptic glenoid loosening or failure in total shoulder arthroplasty: revision with glenoid reimplantation. J Shoulder Elbow Surg. 2013;22:745-751. doi:10.1016/j.jse.2012.08.009
Erickson BJ, Chalmers PN, Denard P, et al. Does commercially available shoulder arthroplasty preoperative planning software agree with surgeon measurements of version, inclination, and subluxation? J Shoulder Elbow Surg. 2021;30:413-420. doi:10.1016/j.jse.2020.05.027
Werner BS, Hudek R, Burkhart KJ, et al. The influence of three-dimensional planning on decision-making in total shoulder arthroplasty. J Shoulder Elbow Surg. 2017;26:1477-1483. doi:10.1016/j.jse.2017.01.006
Blueprint. Stryker. Updated August 2025. Accessed September 3, 2025. https://www.stryker.com/us/en/trauma-and-extremities/products/blueprint.html
NextAR Shoulder. Medacta. Accessed September 3, 2025. https://nextar.medacta.com/shoulder
Baumgarten KM. Accuracy of Blueprint software in predicting range of motion 1 year after reverse total shoulder arthroplasty. J Shoulder Elbow Surg. 2023;32:1088-1094. doi:10.1016/j.jse.2022.12.009
Rojas JT, Jost B, Zipeto C, et al. Glenoid component placement in reverse shoulder arthroplasty assisted with augmented reality through a head-mounted display leads to low deviation between planned and postoperative parameters. J Shoulder Elbow Surg. 2023;32:e587-e596. doi:10.1016/j.jse.2023.05.002
Dey Hazra RO, Paksoy A, Imiolczyk JP, et al. Augmented reality–assisted intraoperative navigation increases precision of glenoid inclination in reverse shoulder arthroplasty. J Shoulder Elbow Surg. 2025;34(2):577-583. doi:10.1016/j.jse.2024.05.039
Azuma RT. A survey of augmented reality. Presence-Teleop Virt. 1997;6:355-385. doi:10.1162/pres.1997.6.4.355
Speicher M, Hall BD, Nebeling M. What is Mixed Reality? In: Proceedings of the 2019 CHI Conference on Human Factors in Computing Systems. Association for Computing Machinery; 2019:1-15. doi:10.1145/3290605.3300767
Digioia AM, Jaramaz B, Nikou C, et al. Surgical navigation for total hip replacement with the use of hipnav. Oper Tech Orthop. 2000;10:3-8. doi:10.1016/S1048-6666(00)80036-1
Ogawa H, Hasegawa S, Tsukada S, et al. A pilot study of augmented reality technology applied to the acetabular cup placement during total hip arthroplasty. J Arthroplasty. 2018;33:1833-1837. doi:10.1016/j.arth.2018.01.067
Shen F, Chen B, Guo Q, et al. Augmented reality patient-specific reconstruction plate design for pelvic and acetabular fracture surgery. Int J CARS. 2013;8:169-179. doi:10.1007/s11548-012-0775-5
Cho HS, Park YK, Gupta S, et al. Augmented reality in bone tumour resection: an experimental study. Bone Joint Res. 2017;6:137-143. doi:10.1302/2046-3758.63.bjr-2016-0289.r1
Wu X, Liu R, Yu J, et al. Mixed reality technology launches in orthopedic surgery for comprehensive preoperative management of complicated cervical fractures. Surg Innov. 2018;25:421-422. doi:10.1177/1553350618761758
Dossett HG, Arthur JR, Makovicka JL, et al. A randomized controlled trial of kinematically and mechanically aligned total knee arthroplasties: long-term follow-up. J Arthroplasty. 2023;38:S209-S214. doi:10.1016/j.arth.2023.03.065
Kazarian GS, Haddad FS, Donaldson MJ, et al. Implant malalignment may be a risk factor for poor patient-reported outcomes measures (PROMs) following total knee arthroplasty (TKA). J Arthroplasty. 2022;37:S129-S133. doi:10.1016/j.arth.2022.02.087
Peng Y, Arauz P, An S, et al. Does component alignment affect patient reported outcomes following bicruciate retaining total knee arthroplasty? An in vivo three-dimensional analysis. J Knee Surg. 2020;33:798-803. doi:10.1055/s-0039-1688500
D’Lima DD, Urquhart AG, Buehler KO, et al. The effect of the orientation of the acetabular and femoral components on the range of motion of the hip at different head-neck ratios. J Bone Joint Surg Am. 2000;82:315-321. doi:10.2106/00004623-200003000-00003
Yamaguchi M, Akisue T, Bauer TW, et al. The spatial location of impingement in total hip arthroplasty. J Arthroplasty. 2000;15:305-313. doi:10.1016/s0883-5403(00)90601-6
Grammatopoulos G, Alvand A, Monk AP, et al. Surgeons’ accuracy in achieving their desired acetabular component orientation. J Bone Joint Surg. 2016;98:e72. doi:10.2106/JBJS.15.01080
Kennedy JG, Rogers WB, Soffe KE, et al. Effect of acetabular component orientation on recurrent dislocation, pelvic osteolysis, polyethylene wear, and component migration. J Arthroplasty. 1998;13:530-534. doi:10.1016/S0883-5403(98)90052-3
Del Schutte H, Lipman AJ, Bannar SM, et al. Effects of acetabular abduction on cup wear rates in total hip arthroplasty. J Arthroplasty. 1998;13:621-626. doi:10.1016/S0883-5403(98)80003-X
Aresti N, Kassam J, Bartlett D, et al. Primary care management of postoperative shoulder, hip, and knee arthroplasty. BMJ. 2017;359:j4431. doi:10.1136/bmj.j4431
HipInsightTM System. Zimmer Biomet. Accessed September 3, 2025. https://www.zimmerbiomet.com/en/products-and-solutions/zb-edge/mixed-reality-portfolio/hipinsight-system.html
ARVIS. Insight Medical Systems. Accessed September 3, 2025. https://www.insightmedsys.com/arvis
Sun DC, Murphy WS, Amundson AJ, et al. Validation of a novel method of measuring cup orientation using biplanar simultaneous radiographic images. J Arthroplasty. 2023;38:S252-S256. doi:10.1016/j.arth.2023.04.011
Tsukada S, Ogawa H, Nishino M, et al. Augmented reality-assisted femoral bone resection in total knee arthroplasty. JBJS Open Access. 2021;6:e21.00001. doi:10.2106/JBJS.OA.21.00001
Castellarin G, Bori E, Barbieux E, et al. Is total knee arthroplasty surgical performance enhanced using augmented reality? A single-center study on 76 consecutive patients. J Arthroplasty. 2024;39:332-335. doi:10.1016/j.arth.2023.08.013
Choi YJ, Ra HJ. Patient satisfaction after total knee arthroplasty. Knee Surg Relat Res. 2016;28:1. doi:10.5792/ksrr.2016.28.1.1
Hazratwala K, Gouk C, Wilkinson MPR, et al. Navigated functional alignment total knee arthroplasty achieves reliable, reproducible and accurate results with high patient satisfaction. Knee Surg Sports Traumatol Arthrosc. 2023;31:3861-3870. doi:10.1007/s00167-023-07327-w
Knee+. Pixee Medical. Accessed September 3, 2025. https://www.pixee-medical.com/en/products/knee-nexsight/
KNEE | NEXTAR. Nextar. Accessed September 3, 2025. https://nextar.medacta.com/knee
POLARIS AR receives clearance from the U.S. Food and Drug Administration for STELLAR Knee. News release. PRNewswire. November 3, 2023. Accessed September 3, 2025. https://www.prnewswire.com/news-releases/polarisar-receives-clearance-from-the-us-food-and-drug-administration-for-stellar-knee-301976747.html
van Overschelde P, Vansintjan P, Byn P, Lapierre C, van Lysebettens W. Does augmented reality improve clinical outcome in TKA? A prospective observational report. In: The 20th Annual Meeting of the International Society for Computer Assisted Orthopaedic Surgery. 2022:170-174.
Sakellariou E, Alevrogiannis P, Alevrogianni F, et al. Single-center experience with Knee+TM augmented reality navigation system in primary total knee arthroplasty. World J Orthop. 2024;15:247-256. doi:10.5312/wjo.v15.i3.247
León-Muñoz VJ, Moya-Angeler J, López-López M, et al. Integration of square fiducial markers in patient-specific instrumentation and their applicability in knee surgery. J Pers Med. 2023;13:727. doi:10.3390/jpm13050727
Fucentese SF, Koch PP. A novel augmented reality-based surgical guidance system for total knee arthroplasty. Arch Orthop Trauma Surg. 2021;141:2227-2233. doi:10.1007/s00402-021-04204-4
Sabatini L, Ascani D, Vezza D, et al. Novel surgical technique for total knee arthroplasty integrating kinematic alignment and real-time elongation of the ligaments using the NextAR system. J Pers Med. 2024;14:794. doi:10.3390/jpm14080794
Daher M, Ghanimeh J, Otayek J, et al. Augmented reality and shoulder replacement: a state-of-the-art review article. JSES Rev Rep Tech. 2023;3:274-278. doi:10.1016/j.xrrt.2023.01.008
Atmani H, Merienne F, Fofi D, et al. Computer aided surgery system for shoulder prosthesis placement. Comput Aided Surg. 2007;12:60-70. doi:10.3109/10929080701210832
Eichinger JK, Galvin JW. Management of complications after total shoulder arthroplasty. Curr Rev Musculoskelet Med. 2015;8:83-91. doi:10.1007/s12178-014-9251-x
Bonnevialle N, Melis B, Neyton L, et al. Aseptic glenoid loosening or failure in total shoulder arthroplasty: revision with glenoid reimplantation. J Shoulder Elbow Surg. 2013;22:745-751. doi:10.1016/j.jse.2012.08.009
Erickson BJ, Chalmers PN, Denard P, et al. Does commercially available shoulder arthroplasty preoperative planning software agree with surgeon measurements of version, inclination, and subluxation? J Shoulder Elbow Surg. 2021;30:413-420. doi:10.1016/j.jse.2020.05.027
Werner BS, Hudek R, Burkhart KJ, et al. The influence of three-dimensional planning on decision-making in total shoulder arthroplasty. J Shoulder Elbow Surg. 2017;26:1477-1483. doi:10.1016/j.jse.2017.01.006
Blueprint. Stryker. Updated August 2025. Accessed September 3, 2025. https://www.stryker.com/us/en/trauma-and-extremities/products/blueprint.html
NextAR Shoulder. Medacta. Accessed September 3, 2025. https://nextar.medacta.com/shoulder
Baumgarten KM. Accuracy of Blueprint software in predicting range of motion 1 year after reverse total shoulder arthroplasty. J Shoulder Elbow Surg. 2023;32:1088-1094. doi:10.1016/j.jse.2022.12.009
Rojas JT, Jost B, Zipeto C, et al. Glenoid component placement in reverse shoulder arthroplasty assisted with augmented reality through a head-mounted display leads to low deviation between planned and postoperative parameters. J Shoulder Elbow Surg. 2023;32:e587-e596. doi:10.1016/j.jse.2023.05.002
Dey Hazra RO, Paksoy A, Imiolczyk JP, et al. Augmented reality–assisted intraoperative navigation increases precision of glenoid inclination in reverse shoulder arthroplasty. J Shoulder Elbow Surg. 2025;34(2):577-583. doi:10.1016/j.jse.2024.05.039
The Integration of Extended Reality in Arthroplasty: Reviewing Technological Progress and Clinical Benefits
The Integration of Extended Reality in Arthroplasty: Reviewing Technological Progress and Clinical Benefits
Diagnostic Testing for Patients With Suspected Ocular Manifestations of Lyme Disease
Diagnostic Testing for Patients With Suspected Ocular Manifestations of Lyme Disease
Since Lyme disease (LD) was first identified in 1975, there has been uncertainty regarding the proper diagnostic testing for suspected cases.1 Challenges involved with ordering Lyme serology testing include navigating tests with an array of false negatives and false positives.2 Confounding these challenges is the wide variety of ocular manifestations of LD, ranging from nonspecific conjunctivitis, cranial palsies, and anterior and posterior segment inflammation.2,3 This article provides diagnostic testing guidelines for eye care clinicians who encounter patients with suspected LD.
BACKGROUND
LD is a bacterial infection caused by the spirochete Borrelia burgdorferi sensu lato complex transmitted by the Ixodes tick genus. There are 4 species of Ixodes ticks that can infect humans, and only 2 have been identified as principal vectors in North America: Ixodes scapularis and Ixodes pacificus. The incidence of LD is on the rise due to increasing global temperatures and expanding geographic borders for the organism. Cases in endemic areas range from 10 per 100,000 people to 50 per 100,000 people.4
LD occurs in 3 stages: early localized (stage 1), early disseminated (stage 2), and late disseminated (stage 3). In stage 1, patients typically present with erythema migrans (EM) rash (bull’s-eye cutaneous rash) and other nonspecific flu-like symptoms of fever, fatigue, and arthralgia. Stage 2 occurs several weeks to months after the initial infection and the infection has invaded other systemic organs, causing conditions like carditis, meningitis, and arthritis. A small subset of patients may progress to stage 3, which is characterized by chronic arthritis and chronic neurological LD.2,4,5 Ocular manifestations have been well-documented in all stages of LD but are more prevalent in early disseminated disease (Table).2,3,6,7
Indications
Recognizing common ocular manifestations associated with LD will allow eye care practitioners to make a timely diagnosis and initiate treatment. The most common ocular findings from LD include conjunctivitis, keratitis, cranial nerve VII palsy, optic neuritis, granulomatous iridocyclitis, and pars planitis.2,6 While retrospective studies suggest that up to 10% of patients with early localized LD have a nonspecific follicular conjunctivitis, those patients are unlikely to present for ocular evaluation. If a patient does present with an acute conjunctivitis, many clinicians do not consider LD in their differential diagnosis.8 In endemic areas, it is important to query patients for additional symptoms that may indicate LD.
Obtaining a complete patient history is vital in aiding a clinician’s decision to order Lyme serology for suspected LD. Epidemiology, history of geography/travel, pet exposure, sexual history (necessary to rule out other conditions [ie, syphilis] to direct appropriate diagnostic testing), and a complete review of systems should be obtained.2,4 LD may mimic other inflammatory autoimmune conditions or infectious diseases such as syphilis.2,5 This can lead to obtaining unnecessary Lyme serologies or failing to diagnose LD.5,7
Diagnostic testing is not indicated when a patient presents with an asymptomatic tick bite (ie, has no fever, malaise, or EM rash) or if a patient does not live in or has not recently traveled to an endemic area because it would be highly unlikely the patient has LD.9,10 If the patient reports known contact with a tick and has a rash suspicious for EM, the diagnosis may be made without confirmatory testing because EM is pathognomonic for LD.7,11 Serologic testing is not recommended in these cases, particularly if there is a single EM lesion, since the lesion often presents prior to development of an immune response leading to seronegative results.8
Lyme serology is necessary if a patient presents with ocular manifestations known to be associated with LD and resides in, or has recently traveled to, an area where LD is endemic (ie, New England, Minnesota, or Wisconsin).7,12 These criteria are of particular importance: about 50% of patients do not recall a tick bite and 20% to 40% do not present with an EM.2,9
Diagnostic Testing
In 2019 the Centers for Disease Control and Prevention (CDC) updated their testing guidelines to the modified 2-tier testing (MTTT) method. The MTTT first recommends a Lyme enzyme immunoassay (EIA), with a second EIA recommended only if the first is positive.12-14 The MTTT method has better sensitivity in early localized LD compared to standard 2-tier testing.9,11,12 The CDC advises against the use of any laboratory serology tests not approved by the US Food and Drug Administration.13 The CDC also advises that LD serology testing should not be performed as a “test for cure,” because even after successful treatment, an individual may still test positive.1,9 Follow-up testing in patients treated early in the disease course (ie, in the setting of EM) may never have an antibody response. In these cases, a negative test should not exclude an LD diagnosis. 9 For patients with suspected neuroborreliosis, a lumbar puncture may not be needed if a patient already has a positive peripheral serology via the MTTT method.12 The Figure depicts a flow chart for the process of ordering and interpreting testing.
Most LD testing, if correlated with clinical disease, is positive after 4 to 6 weeks.9 If an eye disease is noted and the patient has positive Lyme serology, the patient should still be screened for Lyme neuroborreliosis of the central nervous system (CNS). Examination of the fundus for papilledema, review of symptoms of aseptic meningitis, and a careful neurologic examination should be performed.15
If CNS disease is suspected, the patient may need additional CNS testing to support treatment decisions. The 2020 Infectious Diseases Society of America Lyme guidelines recommend to: (1) obtain simultaneous samples of cerebrospinal fluid (CSF) and serum for determination of the CSF:serum antibody index; (2) do not obtain CSF serology without measurement of the CSF:serum antibody index; and (3) do not obtain routine polymerase chain reaction or culture of CSF or serum.15 Once an LD diagnosis is confirmed, the CDC recommends a course of 100 mg of oral doxycycline twice daily for 14 to 21 days or an antimicrobial equivalent (eg, amoxicillin) if doxycycline is contraindicated. However, the antimicrobial dosage may vary depending on the stage of LD.11 Patients with confirmed neuroborreliosis should be admitted for 14 days of intravenous ceftriaxone or intravenous penicillin.2
CONCLUSIONS
To ensure timely diagnosis and treatment, eye care clinicians should be familiar with the appropriate diagnostic testing for patients suspected to have ocular manifestations of LD. For patients with suspected LD and a high pretest probability, clinicians should obtain a first-order Lyme EIA.12-14 If testing confirms LD, refer the patient to an infectious disease specialist for antimicrobial treatment and additional management.11
- Kullberg BJ, Vrijmoeth HD, van de Schoor F, Hovius JW. Lyme borreliosis: diagnosis and management. BMJ. 2020;369:m1041. doi:10.1136/bmj.m1041
- Zaidman GW. The ocular manifestations of Lyme disease. Int Ophthalmol Clin. 1993;33(1):9-22. doi:10.1097/00004397-199303310-00004
- Lesser RL. Ocular manifestations of Lyme disease. Am J Med. 1995; 98(4A):60S-62S. doi:10.1016/s0002-9343(99)80045-x
- Mead P. Epidemiology of Lyme disease. Infect Dis Clin North Am. 2022;36(3):495-521. doi:10.1016/j.idc.2022.03.004
- Klig JE. Ophthalmologic complications of systemic disease. Emerg Med Clin North Am. 2008;26(1):217-viii. doi:10.1016/j.emc.2007.10.003
- Raja H, Starr MR, Bakri SJ. Ocular manifestations of tickborne diseases. Surv Ophthalmol. 2016;61(6):726-744. doi:10.1016/j.survophthal.2016.03.011
- Mora P, Carta A. Ocular manifestations of Lyme borreliosis in Europe. Int J Med Sci. 2009;6(3):124-125. doi:10.7150/ijms.6.124
- Mikkilä HO, Seppälä IJ, Viljanen MK, Peltomaa MP, Karma A. The expanding clinical spectrum of ocular lyme borreliosis. Ophthalmology. 2000;107(3):581-587. doi:10.1016/s0161-6420(99)00128-1
- Schriefer ME. Lyme disease diagnosis: serology. Clin Lab Med. 2015;35(4):797-814. doi:10.1016/j.cll.2015.08.001
- Beck AR, Marx GE, Hinckley AF. Diagnosis, treatment, and prevention practices for Lyme disease by clinicians, United States, 2013-2015. Public Health Rep. 2021;136(5):609- 617. doi:10.1177/0033354920973235
- Wormser GP, McKenna D, Nowakowski J. Management approaches for suspected and established Lyme disease used at the Lyme disease diagnostic center. Wien Klin Wochenschr. 2018;130(15-16):463-467. doi:10.1007/s00508-015-0936-y
- Kobayashi T, Auwaerter PG. Diagnostic testing for Lyme disease. Infect Dis Clin North Am. 2022;36(3):605-620. doi:10.1016/j.idc.2022.04.001
- Mead P, Petersen J, Hinckley A. Updated CDC recommendation for serologic diagnosis of Lyme disease. MMWR Morb Mortal Wkly Rep. 2019;68(32):703. doi:10.15585/mmwr.mm6832a4
- Association of Public Health Laboratories. Suggested Reporting Language, Interpretation and Guidance Regarding Lyme Disease Serologic Test Results. April 2024. Accessed December 3, 2024. https://www.aphl.org/aboutAPHL/publications/Documents/ID-2024-Lyme-Disease-Serologic-Testing-Reporting.pdf
- Lantos PM, Rumbaugh P, Bockenstedt L, et al. Clinical practice guidelines by the Infectious Diseases Society of America (IDSA), American Academy of Neurology (AAN), and American College of Rheumatology (ACR): 2020 guidelines for the prevention, diagnosis and treatment of Lyme Disease. Clin Infect Dis. 2021;72(1):e1-e48. doi:10.1093/cid/ciaa1215
Since Lyme disease (LD) was first identified in 1975, there has been uncertainty regarding the proper diagnostic testing for suspected cases.1 Challenges involved with ordering Lyme serology testing include navigating tests with an array of false negatives and false positives.2 Confounding these challenges is the wide variety of ocular manifestations of LD, ranging from nonspecific conjunctivitis, cranial palsies, and anterior and posterior segment inflammation.2,3 This article provides diagnostic testing guidelines for eye care clinicians who encounter patients with suspected LD.
BACKGROUND
LD is a bacterial infection caused by the spirochete Borrelia burgdorferi sensu lato complex transmitted by the Ixodes tick genus. There are 4 species of Ixodes ticks that can infect humans, and only 2 have been identified as principal vectors in North America: Ixodes scapularis and Ixodes pacificus. The incidence of LD is on the rise due to increasing global temperatures and expanding geographic borders for the organism. Cases in endemic areas range from 10 per 100,000 people to 50 per 100,000 people.4
LD occurs in 3 stages: early localized (stage 1), early disseminated (stage 2), and late disseminated (stage 3). In stage 1, patients typically present with erythema migrans (EM) rash (bull’s-eye cutaneous rash) and other nonspecific flu-like symptoms of fever, fatigue, and arthralgia. Stage 2 occurs several weeks to months after the initial infection and the infection has invaded other systemic organs, causing conditions like carditis, meningitis, and arthritis. A small subset of patients may progress to stage 3, which is characterized by chronic arthritis and chronic neurological LD.2,4,5 Ocular manifestations have been well-documented in all stages of LD but are more prevalent in early disseminated disease (Table).2,3,6,7

Indications
Recognizing common ocular manifestations associated with LD will allow eye care practitioners to make a timely diagnosis and initiate treatment. The most common ocular findings from LD include conjunctivitis, keratitis, cranial nerve VII palsy, optic neuritis, granulomatous iridocyclitis, and pars planitis.2,6 While retrospective studies suggest that up to 10% of patients with early localized LD have a nonspecific follicular conjunctivitis, those patients are unlikely to present for ocular evaluation. If a patient does present with an acute conjunctivitis, many clinicians do not consider LD in their differential diagnosis.8 In endemic areas, it is important to query patients for additional symptoms that may indicate LD.
Obtaining a complete patient history is vital in aiding a clinician’s decision to order Lyme serology for suspected LD. Epidemiology, history of geography/travel, pet exposure, sexual history (necessary to rule out other conditions [ie, syphilis] to direct appropriate diagnostic testing), and a complete review of systems should be obtained.2,4 LD may mimic other inflammatory autoimmune conditions or infectious diseases such as syphilis.2,5 This can lead to obtaining unnecessary Lyme serologies or failing to diagnose LD.5,7
Diagnostic testing is not indicated when a patient presents with an asymptomatic tick bite (ie, has no fever, malaise, or EM rash) or if a patient does not live in or has not recently traveled to an endemic area because it would be highly unlikely the patient has LD.9,10 If the patient reports known contact with a tick and has a rash suspicious for EM, the diagnosis may be made without confirmatory testing because EM is pathognomonic for LD.7,11 Serologic testing is not recommended in these cases, particularly if there is a single EM lesion, since the lesion often presents prior to development of an immune response leading to seronegative results.8
Lyme serology is necessary if a patient presents with ocular manifestations known to be associated with LD and resides in, or has recently traveled to, an area where LD is endemic (ie, New England, Minnesota, or Wisconsin).7,12 These criteria are of particular importance: about 50% of patients do not recall a tick bite and 20% to 40% do not present with an EM.2,9
Diagnostic Testing
In 2019 the Centers for Disease Control and Prevention (CDC) updated their testing guidelines to the modified 2-tier testing (MTTT) method. The MTTT first recommends a Lyme enzyme immunoassay (EIA), with a second EIA recommended only if the first is positive.12-14 The MTTT method has better sensitivity in early localized LD compared to standard 2-tier testing.9,11,12 The CDC advises against the use of any laboratory serology tests not approved by the US Food and Drug Administration.13 The CDC also advises that LD serology testing should not be performed as a “test for cure,” because even after successful treatment, an individual may still test positive.1,9 Follow-up testing in patients treated early in the disease course (ie, in the setting of EM) may never have an antibody response. In these cases, a negative test should not exclude an LD diagnosis. 9 For patients with suspected neuroborreliosis, a lumbar puncture may not be needed if a patient already has a positive peripheral serology via the MTTT method.12 The Figure depicts a flow chart for the process of ordering and interpreting testing.

Most LD testing, if correlated with clinical disease, is positive after 4 to 6 weeks.9 If an eye disease is noted and the patient has positive Lyme serology, the patient should still be screened for Lyme neuroborreliosis of the central nervous system (CNS). Examination of the fundus for papilledema, review of symptoms of aseptic meningitis, and a careful neurologic examination should be performed.15
If CNS disease is suspected, the patient may need additional CNS testing to support treatment decisions. The 2020 Infectious Diseases Society of America Lyme guidelines recommend to: (1) obtain simultaneous samples of cerebrospinal fluid (CSF) and serum for determination of the CSF:serum antibody index; (2) do not obtain CSF serology without measurement of the CSF:serum antibody index; and (3) do not obtain routine polymerase chain reaction or culture of CSF or serum.15 Once an LD diagnosis is confirmed, the CDC recommends a course of 100 mg of oral doxycycline twice daily for 14 to 21 days or an antimicrobial equivalent (eg, amoxicillin) if doxycycline is contraindicated. However, the antimicrobial dosage may vary depending on the stage of LD.11 Patients with confirmed neuroborreliosis should be admitted for 14 days of intravenous ceftriaxone or intravenous penicillin.2
CONCLUSIONS
To ensure timely diagnosis and treatment, eye care clinicians should be familiar with the appropriate diagnostic testing for patients suspected to have ocular manifestations of LD. For patients with suspected LD and a high pretest probability, clinicians should obtain a first-order Lyme EIA.12-14 If testing confirms LD, refer the patient to an infectious disease specialist for antimicrobial treatment and additional management.11
Since Lyme disease (LD) was first identified in 1975, there has been uncertainty regarding the proper diagnostic testing for suspected cases.1 Challenges involved with ordering Lyme serology testing include navigating tests with an array of false negatives and false positives.2 Confounding these challenges is the wide variety of ocular manifestations of LD, ranging from nonspecific conjunctivitis, cranial palsies, and anterior and posterior segment inflammation.2,3 This article provides diagnostic testing guidelines for eye care clinicians who encounter patients with suspected LD.
BACKGROUND
LD is a bacterial infection caused by the spirochete Borrelia burgdorferi sensu lato complex transmitted by the Ixodes tick genus. There are 4 species of Ixodes ticks that can infect humans, and only 2 have been identified as principal vectors in North America: Ixodes scapularis and Ixodes pacificus. The incidence of LD is on the rise due to increasing global temperatures and expanding geographic borders for the organism. Cases in endemic areas range from 10 per 100,000 people to 50 per 100,000 people.4
LD occurs in 3 stages: early localized (stage 1), early disseminated (stage 2), and late disseminated (stage 3). In stage 1, patients typically present with erythema migrans (EM) rash (bull’s-eye cutaneous rash) and other nonspecific flu-like symptoms of fever, fatigue, and arthralgia. Stage 2 occurs several weeks to months after the initial infection and the infection has invaded other systemic organs, causing conditions like carditis, meningitis, and arthritis. A small subset of patients may progress to stage 3, which is characterized by chronic arthritis and chronic neurological LD.2,4,5 Ocular manifestations have been well-documented in all stages of LD but are more prevalent in early disseminated disease (Table).2,3,6,7

Indications
Recognizing common ocular manifestations associated with LD will allow eye care practitioners to make a timely diagnosis and initiate treatment. The most common ocular findings from LD include conjunctivitis, keratitis, cranial nerve VII palsy, optic neuritis, granulomatous iridocyclitis, and pars planitis.2,6 While retrospective studies suggest that up to 10% of patients with early localized LD have a nonspecific follicular conjunctivitis, those patients are unlikely to present for ocular evaluation. If a patient does present with an acute conjunctivitis, many clinicians do not consider LD in their differential diagnosis.8 In endemic areas, it is important to query patients for additional symptoms that may indicate LD.
Obtaining a complete patient history is vital in aiding a clinician’s decision to order Lyme serology for suspected LD. Epidemiology, history of geography/travel, pet exposure, sexual history (necessary to rule out other conditions [ie, syphilis] to direct appropriate diagnostic testing), and a complete review of systems should be obtained.2,4 LD may mimic other inflammatory autoimmune conditions or infectious diseases such as syphilis.2,5 This can lead to obtaining unnecessary Lyme serologies or failing to diagnose LD.5,7
Diagnostic testing is not indicated when a patient presents with an asymptomatic tick bite (ie, has no fever, malaise, or EM rash) or if a patient does not live in or has not recently traveled to an endemic area because it would be highly unlikely the patient has LD.9,10 If the patient reports known contact with a tick and has a rash suspicious for EM, the diagnosis may be made without confirmatory testing because EM is pathognomonic for LD.7,11 Serologic testing is not recommended in these cases, particularly if there is a single EM lesion, since the lesion often presents prior to development of an immune response leading to seronegative results.8
Lyme serology is necessary if a patient presents with ocular manifestations known to be associated with LD and resides in, or has recently traveled to, an area where LD is endemic (ie, New England, Minnesota, or Wisconsin).7,12 These criteria are of particular importance: about 50% of patients do not recall a tick bite and 20% to 40% do not present with an EM.2,9
Diagnostic Testing
In 2019 the Centers for Disease Control and Prevention (CDC) updated their testing guidelines to the modified 2-tier testing (MTTT) method. The MTTT first recommends a Lyme enzyme immunoassay (EIA), with a second EIA recommended only if the first is positive.12-14 The MTTT method has better sensitivity in early localized LD compared to standard 2-tier testing.9,11,12 The CDC advises against the use of any laboratory serology tests not approved by the US Food and Drug Administration.13 The CDC also advises that LD serology testing should not be performed as a “test for cure,” because even after successful treatment, an individual may still test positive.1,9 Follow-up testing in patients treated early in the disease course (ie, in the setting of EM) may never have an antibody response. In these cases, a negative test should not exclude an LD diagnosis. 9 For patients with suspected neuroborreliosis, a lumbar puncture may not be needed if a patient already has a positive peripheral serology via the MTTT method.12 The Figure depicts a flow chart for the process of ordering and interpreting testing.

Most LD testing, if correlated with clinical disease, is positive after 4 to 6 weeks.9 If an eye disease is noted and the patient has positive Lyme serology, the patient should still be screened for Lyme neuroborreliosis of the central nervous system (CNS). Examination of the fundus for papilledema, review of symptoms of aseptic meningitis, and a careful neurologic examination should be performed.15
If CNS disease is suspected, the patient may need additional CNS testing to support treatment decisions. The 2020 Infectious Diseases Society of America Lyme guidelines recommend to: (1) obtain simultaneous samples of cerebrospinal fluid (CSF) and serum for determination of the CSF:serum antibody index; (2) do not obtain CSF serology without measurement of the CSF:serum antibody index; and (3) do not obtain routine polymerase chain reaction or culture of CSF or serum.15 Once an LD diagnosis is confirmed, the CDC recommends a course of 100 mg of oral doxycycline twice daily for 14 to 21 days or an antimicrobial equivalent (eg, amoxicillin) if doxycycline is contraindicated. However, the antimicrobial dosage may vary depending on the stage of LD.11 Patients with confirmed neuroborreliosis should be admitted for 14 days of intravenous ceftriaxone or intravenous penicillin.2
CONCLUSIONS
To ensure timely diagnosis and treatment, eye care clinicians should be familiar with the appropriate diagnostic testing for patients suspected to have ocular manifestations of LD. For patients with suspected LD and a high pretest probability, clinicians should obtain a first-order Lyme EIA.12-14 If testing confirms LD, refer the patient to an infectious disease specialist for antimicrobial treatment and additional management.11
- Kullberg BJ, Vrijmoeth HD, van de Schoor F, Hovius JW. Lyme borreliosis: diagnosis and management. BMJ. 2020;369:m1041. doi:10.1136/bmj.m1041
- Zaidman GW. The ocular manifestations of Lyme disease. Int Ophthalmol Clin. 1993;33(1):9-22. doi:10.1097/00004397-199303310-00004
- Lesser RL. Ocular manifestations of Lyme disease. Am J Med. 1995; 98(4A):60S-62S. doi:10.1016/s0002-9343(99)80045-x
- Mead P. Epidemiology of Lyme disease. Infect Dis Clin North Am. 2022;36(3):495-521. doi:10.1016/j.idc.2022.03.004
- Klig JE. Ophthalmologic complications of systemic disease. Emerg Med Clin North Am. 2008;26(1):217-viii. doi:10.1016/j.emc.2007.10.003
- Raja H, Starr MR, Bakri SJ. Ocular manifestations of tickborne diseases. Surv Ophthalmol. 2016;61(6):726-744. doi:10.1016/j.survophthal.2016.03.011
- Mora P, Carta A. Ocular manifestations of Lyme borreliosis in Europe. Int J Med Sci. 2009;6(3):124-125. doi:10.7150/ijms.6.124
- Mikkilä HO, Seppälä IJ, Viljanen MK, Peltomaa MP, Karma A. The expanding clinical spectrum of ocular lyme borreliosis. Ophthalmology. 2000;107(3):581-587. doi:10.1016/s0161-6420(99)00128-1
- Schriefer ME. Lyme disease diagnosis: serology. Clin Lab Med. 2015;35(4):797-814. doi:10.1016/j.cll.2015.08.001
- Beck AR, Marx GE, Hinckley AF. Diagnosis, treatment, and prevention practices for Lyme disease by clinicians, United States, 2013-2015. Public Health Rep. 2021;136(5):609- 617. doi:10.1177/0033354920973235
- Wormser GP, McKenna D, Nowakowski J. Management approaches for suspected and established Lyme disease used at the Lyme disease diagnostic center. Wien Klin Wochenschr. 2018;130(15-16):463-467. doi:10.1007/s00508-015-0936-y
- Kobayashi T, Auwaerter PG. Diagnostic testing for Lyme disease. Infect Dis Clin North Am. 2022;36(3):605-620. doi:10.1016/j.idc.2022.04.001
- Mead P, Petersen J, Hinckley A. Updated CDC recommendation for serologic diagnosis of Lyme disease. MMWR Morb Mortal Wkly Rep. 2019;68(32):703. doi:10.15585/mmwr.mm6832a4
- Association of Public Health Laboratories. Suggested Reporting Language, Interpretation and Guidance Regarding Lyme Disease Serologic Test Results. April 2024. Accessed December 3, 2024. https://www.aphl.org/aboutAPHL/publications/Documents/ID-2024-Lyme-Disease-Serologic-Testing-Reporting.pdf
- Lantos PM, Rumbaugh P, Bockenstedt L, et al. Clinical practice guidelines by the Infectious Diseases Society of America (IDSA), American Academy of Neurology (AAN), and American College of Rheumatology (ACR): 2020 guidelines for the prevention, diagnosis and treatment of Lyme Disease. Clin Infect Dis. 2021;72(1):e1-e48. doi:10.1093/cid/ciaa1215
- Kullberg BJ, Vrijmoeth HD, van de Schoor F, Hovius JW. Lyme borreliosis: diagnosis and management. BMJ. 2020;369:m1041. doi:10.1136/bmj.m1041
- Zaidman GW. The ocular manifestations of Lyme disease. Int Ophthalmol Clin. 1993;33(1):9-22. doi:10.1097/00004397-199303310-00004
- Lesser RL. Ocular manifestations of Lyme disease. Am J Med. 1995; 98(4A):60S-62S. doi:10.1016/s0002-9343(99)80045-x
- Mead P. Epidemiology of Lyme disease. Infect Dis Clin North Am. 2022;36(3):495-521. doi:10.1016/j.idc.2022.03.004
- Klig JE. Ophthalmologic complications of systemic disease. Emerg Med Clin North Am. 2008;26(1):217-viii. doi:10.1016/j.emc.2007.10.003
- Raja H, Starr MR, Bakri SJ. Ocular manifestations of tickborne diseases. Surv Ophthalmol. 2016;61(6):726-744. doi:10.1016/j.survophthal.2016.03.011
- Mora P, Carta A. Ocular manifestations of Lyme borreliosis in Europe. Int J Med Sci. 2009;6(3):124-125. doi:10.7150/ijms.6.124
- Mikkilä HO, Seppälä IJ, Viljanen MK, Peltomaa MP, Karma A. The expanding clinical spectrum of ocular lyme borreliosis. Ophthalmology. 2000;107(3):581-587. doi:10.1016/s0161-6420(99)00128-1
- Schriefer ME. Lyme disease diagnosis: serology. Clin Lab Med. 2015;35(4):797-814. doi:10.1016/j.cll.2015.08.001
- Beck AR, Marx GE, Hinckley AF. Diagnosis, treatment, and prevention practices for Lyme disease by clinicians, United States, 2013-2015. Public Health Rep. 2021;136(5):609- 617. doi:10.1177/0033354920973235
- Wormser GP, McKenna D, Nowakowski J. Management approaches for suspected and established Lyme disease used at the Lyme disease diagnostic center. Wien Klin Wochenschr. 2018;130(15-16):463-467. doi:10.1007/s00508-015-0936-y
- Kobayashi T, Auwaerter PG. Diagnostic testing for Lyme disease. Infect Dis Clin North Am. 2022;36(3):605-620. doi:10.1016/j.idc.2022.04.001
- Mead P, Petersen J, Hinckley A. Updated CDC recommendation for serologic diagnosis of Lyme disease. MMWR Morb Mortal Wkly Rep. 2019;68(32):703. doi:10.15585/mmwr.mm6832a4
- Association of Public Health Laboratories. Suggested Reporting Language, Interpretation and Guidance Regarding Lyme Disease Serologic Test Results. April 2024. Accessed December 3, 2024. https://www.aphl.org/aboutAPHL/publications/Documents/ID-2024-Lyme-Disease-Serologic-Testing-Reporting.pdf
- Lantos PM, Rumbaugh P, Bockenstedt L, et al. Clinical practice guidelines by the Infectious Diseases Society of America (IDSA), American Academy of Neurology (AAN), and American College of Rheumatology (ACR): 2020 guidelines for the prevention, diagnosis and treatment of Lyme Disease. Clin Infect Dis. 2021;72(1):e1-e48. doi:10.1093/cid/ciaa1215
Diagnostic Testing for Patients With Suspected Ocular Manifestations of Lyme Disease
Diagnostic Testing for Patients With Suspected Ocular Manifestations of Lyme Disease
The Effect of GLP-1 Receptor Agonists on Hidradenitis Suppurativa: A Comprehensive Systematic Review
The Effect of GLP-1 Receptor Agonists on Hidradenitis Suppurativa: A Comprehensive Systematic Review
Hidradenitis suppurativa (HS) is a chronic relapsing inflammatory skin disorder affecting apocrine gland–bearing areas such as the axillae, inguinal regions, and anogenital area.1 It manifests with painful nodules, abscesses, sinus tract formation, and scarring.2 The disease strongly impacts patients’ quality of life due to pain, malodor, and psychosocial burden.3
The exact etiology of HS is multifactorial, involving genetic predisposition, mechanical stress, hormonal influences, dysbiosis, and immune dysregulation.4 Obesity and metabolic syndrome are highly prevalent among patients with HS and are considered exacerbating factors.5 Adipose tissue contributes to systemic inflammation through the secretion of proinflammatory cytokines such as tumor necrosis factor (TNF) α and interleukins (ILs).6
Management of HS includes lifestyle modifications, medical therapy, and surgical interventions. Medical treatments encompass antibiotics, retinoids, hormonal therapy, immunosuppressants, and immunomodulators such as anti-TNF and anti–IL-17 agents.7 Despite available therapies, many patients have suboptimal responses or experience adverse effects and dramatic reductions in their quality of life.3
Glucagonlike peptide 1 receptor agonists (GLP-1 RAs) are incretin-based therapies used in type 2 diabetes and obesity management.8 They enhance insulin secretion, suppress glucagon release, delay gastric emptying, and promote satiety.9 Beyond glycemic control, GLP-1 RAs exhibit anti-inflammatory properties and cardiovascular benefits.10
Given the high prevalence of obesity and metabolic syndrome in patients with HS as well as the anti-inflammatory effects of GLP-1 RAs, these agents may offer therapeutic benefits in HS.11 We conducted a systematic review to evaluate the existing evidence on the efficacy and safety of GLP-1 RAs in the treatment of HS.
Methods
A systematic review was conducted via a PubMed search of articles indexed for MEDLINE in October 2024, following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines12 using the terms hidradenitis suppurativa OR acne inversa AND GLP-1 receptor agonist OR glucagon-like peptide-1 receptor agonist OR liraglutide OR semaglutide OR exenatide OR dulaglutide. No filters were applied to limit the search by language or publication date.
Inclusion criteria were clinical trials, observational studies (cohort, case control, cross-sectional), and case reports/series involving patients diagnosed with HS treated with GLP-1 RAs. Outcomes of interest included clinical improvement in HS severity (eg, lesion count, pain assessment, HS-specific scores), safety, and adverse events. Exclusion criteria included animal studies or in vitro experiments, reviews, editorials, and opinion pieces without original patient data; studies not in English; and studies not reporting clinical outcomes related to HS.
Two independent reviewers (N.R.K. and S.K.C.) screened the titles and abstracts for relevance. Full-text articles of potentially eligible studies were retrieved for detailed evaluation. Data extracted included study design, patient demographics, intervention details, outcomes, and adverse events. Discrepancies were resolved through discussion.
Results
The initial search yielded 11 articles (Figure). After screening titles and abstracts, 9 articles were selected for full-text review. Of these, 3 articles met the inclusion criteria. These studies included 3 case reports. Interventions involved liraglutide (2 reports)13,14 and semaglutide15 (1 report)(Table). The patient population consisted of adult patients with HS with comorbid diabetes, obesity, and/or metabolic syndrome.
Jennings et al13 reported a 31-year-old obese woman with a history of smoking and Hurley stage 2 HS, a Hidradenitis Suppurativa Physician’s Global Assessment score of 4, a Dermatology Life Quality Index score of 24, and a body mass index of 45.3. She was treated with liraglutide monotherapy, starting with 0.6 mg subcutaneously once daily then titrating weekly to 1.8 mg subcutaneously. After 4 weeks, outcomes showed a reduction in Hidradenitis Suppurativa Physician’s Global Assessment (score=1) and Dermatology Life Quality Index (score=14) scores, and the patient lost 4.5 kg from baseline. The patient’s Hurley stage decreased from 2 to 1. After another 4 weeks, the patient’s weight decreased by a further 2 kg and HS remained controlled. No adverse events were recorded.
Khandalavala14 reported a single case of a 19-year-old woman with severe HS, obesity, and metabolic syndrome of 8 years’ duration treated with liraglutide. The patient had a weight of 215 lb with a body mass index of 37. With a combination of metformin 2000 mg/d, liraglutide 0.6 mg/d subcutaneously increased to 1.8 mg/d over 2 months, levonorgestrel-ethinyl estradiol (no dosage listed), dapsone 100 mg/d, and finasteride 5 mg/d, there was a marked reduction in nodules and abscesses after 6 months, with a weight loss of 40 lb (19% body weight). No adverse events were reported.
Mainville et al15 described a 59-year-old woman with refractory HS who showed improvement with a combination of intravenous ertapenem 1 g/d for 6 weeks, minocycline 100 mg/d for 3 months, metformin 500 mg three times daily for 2 months, doxycycline 100 mg/d to bridge to adalimumab (160 mg subcutaneously starting dose then 80 mg subcutaneously), and semaglutide (no dosage listed). After semaglutide was introduced, the patient lost 10 kg. The only adverse event was diarrhea.
Comment
The limited but growing body of evidence suggests that GLP-1 RAs may be beneficial in managing HS, particularly in patients with comorbid obesity. Treatment with liraglutide or semaglutide was associated with marked improvements in clinical severity scores, lesion counts, pain reduction, and quality of life.
As adjunct therapy, GLP-1 RAs could serve alongside standard HS treatments such as antibiotics and biologics. Addressing obesity, a known risk factor and disease modifier in HS, may lead to better disease control. The therapeutic benefits of GLP-1 RAs in HS are attributed to weight loss, which reduces adipose tissue and systemic inflammation.16 The anti-inflammatory effects of GLP-1 RAs involve the reduction of proinflammatory cytokines such as IL-6 and TNF-α.17 Metabolic improvements, including enhanced insulin sensitivity and lipid profile, also may contribute to disease modulation.17
Limitations—Because our analysis was limited to 3 case reports, the strength of the evidence is limited. These case reports also lack the standardized use of the Hidradenitis Suppurativa Clinical Response scoring system that generally is found in randomized controlled trials (RCTs). The lack of RCTs precludes definitive conclusions about efficacy. Future directions include the need for well-designed RCTs with large sample sizes to confirm findings, assessment of long-term safety and tolerability in patients with HS, and further research into the molecular mechanisms by which GLP-1 RAs affect HS pathophysiology. Of note, it is imperative to be aware of the medication shortage for all GLP-1 RAs when prescribing these medications for patients with HS.
Conclusion
Glucagonlike peptide 1 RAs show promise as a therapeutic option for HS, especially in patients with obesity and metabolic disturbances. The observed benefits likely result from weight loss and anti-inflammatory effects. Other drugs targeting glucose-dependent insulinotropic polypeptide and glucagon also are being studied thoroughly as options for managing HS. Although preliminary results are encouraging, robust clinical trials are needed to establish efficacy, optimal dosing, and safety in this patient population.
- Vinkel C, Thomsen SF. Hidradenitis suppurativa: causes, features, and current treatments. J Clin Aesthet Dermatol. 2018;11:17-23.
- Napolitano M, Megna M, Timoshchuk EA, et al. Hidradenitis suppurativa: from pathogenesis to diagnosis and treatment. Clin Cosmet Investig Dermatol. 2017;10:105-115. doi:10.2147/CCID.S111019
- Chernyshov PV, Finlay AY, Tomas-Aragones L, et al. Quality of life in hidradenitis suppurativa: an update. Int J Environ Res Public Health. 2021;18:6131. doi:10.3390/ijerph18116131
- Seyed Jafari SM, Hunger RE, Schlapbach C. Hidradenitis suppurativa: current understanding of pathogenic mechanisms and suggestion for treatment algorithm. Front Med (Lausanne). 2020;7:68. doi:10.3389/fmed.2020.00068
- Alotaibi HM. Incidence, risk factors, and prognosis of hidradenitis suppurativa across the globe: insights from the literature. Clin Cosmet Investig Dermatol. 2023;16:545-552. doi:10.2147/CCID.S402453
- Vossen ARJV, van der Zee HH, Prens EP. Hidradenitis suppurativa: a systematic review integrating inflammatory pathways into a cohesive pathogenic model. Front Immunol. 2018;9:2965. doi:10.3389/fimmu.2018.02965
- Orenstein LAV, Nguyen TV, Damiani G, et al. Medical and surgical management of hidradenitis suppurativa: a review of international treatment guidelines and implementation in general dermatology practice. Dermatology. 2020;236:393-412. doi:10.1159/000507323
- Brown E, Cuthbertson DJ, Wilding JP. Newer GLP-1 receptor agonists and obesity-diabetes. Peptides. 2018;100:61-67. doi:10.1016/j.peptides.2017.12.009
- Cornell S. A review of GLP‐1 receptor agonists in type 2 diabetes: a focus on the mechanism of action of once‐weekly agents. J Clin Pharm Ther. 2020;45(suppl 1):17-27. doi:10.1111/jcpt.13230
- Lee YS, Jun HS. Anti-inflammatory effects of GLP-1-based therapies beyond glucose control. Mediators Inflamm. 2016;2016:3094642. doi:10.1155/2016/3094642
- Mintoff D, Benhadou F, Pace NP, et al. Metabolic syndrome and hidradenitis suppurativa: epidemiological, molecular, and therapeutic aspects. Int J Dermatol. 2022;61:1175-1186. doi:10.1111/ijd.15910
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi:10.1136/bmj.n71
- Jennings L, Nestor L, Molloy O, et al. The treatment of hidradenitis suppurativa with the glucagon-like peptide-1 agonist liraglutide. Br J Dermatol. 2017;177:858-859. doi:10.1111/bjd.15233
- Khandalavala BN. A disease-modifying approach for advanced hidradenitis suppurativa (regimen with metformin, liraglutide, dapsone, and finasteride): a case report. Case Rep Dermatol. 2017;9:70-78. doi:10.1159/000473873
- Mainville L, MacHaalany J, Veillette H. Hidradenitis suppurativa patient requiring cardiac procedure with inguinal access: case management with ertapenem. SAGE Open Med Case Rep. 2024;12:2050313X241274819. doi:10.1177/2050313X241274819
- Hamed K, Alosaimi MN, Ali BA, et al. Glucagon-like peptide-1 (GLP-1) receptor agonists: exploring their impact on diabetes, obesity, and cardiovascular health through a comprehensive literature review. Cureus. 2024;16:E68390. doi:10.7759/cureus.68390
- Alharbi SH. Anti-inflammatory role of glucagon-like peptide 1 receptor agonists and its clinical implications. Ther Adv Endocrinol Metab. 2024;15:20420188231222367. doi:10.1177/20420188231222367
Hidradenitis suppurativa (HS) is a chronic relapsing inflammatory skin disorder affecting apocrine gland–bearing areas such as the axillae, inguinal regions, and anogenital area.1 It manifests with painful nodules, abscesses, sinus tract formation, and scarring.2 The disease strongly impacts patients’ quality of life due to pain, malodor, and psychosocial burden.3
The exact etiology of HS is multifactorial, involving genetic predisposition, mechanical stress, hormonal influences, dysbiosis, and immune dysregulation.4 Obesity and metabolic syndrome are highly prevalent among patients with HS and are considered exacerbating factors.5 Adipose tissue contributes to systemic inflammation through the secretion of proinflammatory cytokines such as tumor necrosis factor (TNF) α and interleukins (ILs).6
Management of HS includes lifestyle modifications, medical therapy, and surgical interventions. Medical treatments encompass antibiotics, retinoids, hormonal therapy, immunosuppressants, and immunomodulators such as anti-TNF and anti–IL-17 agents.7 Despite available therapies, many patients have suboptimal responses or experience adverse effects and dramatic reductions in their quality of life.3
Glucagonlike peptide 1 receptor agonists (GLP-1 RAs) are incretin-based therapies used in type 2 diabetes and obesity management.8 They enhance insulin secretion, suppress glucagon release, delay gastric emptying, and promote satiety.9 Beyond glycemic control, GLP-1 RAs exhibit anti-inflammatory properties and cardiovascular benefits.10
Given the high prevalence of obesity and metabolic syndrome in patients with HS as well as the anti-inflammatory effects of GLP-1 RAs, these agents may offer therapeutic benefits in HS.11 We conducted a systematic review to evaluate the existing evidence on the efficacy and safety of GLP-1 RAs in the treatment of HS.
Methods
A systematic review was conducted via a PubMed search of articles indexed for MEDLINE in October 2024, following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines12 using the terms hidradenitis suppurativa OR acne inversa AND GLP-1 receptor agonist OR glucagon-like peptide-1 receptor agonist OR liraglutide OR semaglutide OR exenatide OR dulaglutide. No filters were applied to limit the search by language or publication date.
Inclusion criteria were clinical trials, observational studies (cohort, case control, cross-sectional), and case reports/series involving patients diagnosed with HS treated with GLP-1 RAs. Outcomes of interest included clinical improvement in HS severity (eg, lesion count, pain assessment, HS-specific scores), safety, and adverse events. Exclusion criteria included animal studies or in vitro experiments, reviews, editorials, and opinion pieces without original patient data; studies not in English; and studies not reporting clinical outcomes related to HS.
Two independent reviewers (N.R.K. and S.K.C.) screened the titles and abstracts for relevance. Full-text articles of potentially eligible studies were retrieved for detailed evaluation. Data extracted included study design, patient demographics, intervention details, outcomes, and adverse events. Discrepancies were resolved through discussion.
Results
The initial search yielded 11 articles (Figure). After screening titles and abstracts, 9 articles were selected for full-text review. Of these, 3 articles met the inclusion criteria. These studies included 3 case reports. Interventions involved liraglutide (2 reports)13,14 and semaglutide15 (1 report)(Table). The patient population consisted of adult patients with HS with comorbid diabetes, obesity, and/or metabolic syndrome.


Jennings et al13 reported a 31-year-old obese woman with a history of smoking and Hurley stage 2 HS, a Hidradenitis Suppurativa Physician’s Global Assessment score of 4, a Dermatology Life Quality Index score of 24, and a body mass index of 45.3. She was treated with liraglutide monotherapy, starting with 0.6 mg subcutaneously once daily then titrating weekly to 1.8 mg subcutaneously. After 4 weeks, outcomes showed a reduction in Hidradenitis Suppurativa Physician’s Global Assessment (score=1) and Dermatology Life Quality Index (score=14) scores, and the patient lost 4.5 kg from baseline. The patient’s Hurley stage decreased from 2 to 1. After another 4 weeks, the patient’s weight decreased by a further 2 kg and HS remained controlled. No adverse events were recorded.
Khandalavala14 reported a single case of a 19-year-old woman with severe HS, obesity, and metabolic syndrome of 8 years’ duration treated with liraglutide. The patient had a weight of 215 lb with a body mass index of 37. With a combination of metformin 2000 mg/d, liraglutide 0.6 mg/d subcutaneously increased to 1.8 mg/d over 2 months, levonorgestrel-ethinyl estradiol (no dosage listed), dapsone 100 mg/d, and finasteride 5 mg/d, there was a marked reduction in nodules and abscesses after 6 months, with a weight loss of 40 lb (19% body weight). No adverse events were reported.
Mainville et al15 described a 59-year-old woman with refractory HS who showed improvement with a combination of intravenous ertapenem 1 g/d for 6 weeks, minocycline 100 mg/d for 3 months, metformin 500 mg three times daily for 2 months, doxycycline 100 mg/d to bridge to adalimumab (160 mg subcutaneously starting dose then 80 mg subcutaneously), and semaglutide (no dosage listed). After semaglutide was introduced, the patient lost 10 kg. The only adverse event was diarrhea.
Comment
The limited but growing body of evidence suggests that GLP-1 RAs may be beneficial in managing HS, particularly in patients with comorbid obesity. Treatment with liraglutide or semaglutide was associated with marked improvements in clinical severity scores, lesion counts, pain reduction, and quality of life.
As adjunct therapy, GLP-1 RAs could serve alongside standard HS treatments such as antibiotics and biologics. Addressing obesity, a known risk factor and disease modifier in HS, may lead to better disease control. The therapeutic benefits of GLP-1 RAs in HS are attributed to weight loss, which reduces adipose tissue and systemic inflammation.16 The anti-inflammatory effects of GLP-1 RAs involve the reduction of proinflammatory cytokines such as IL-6 and TNF-α.17 Metabolic improvements, including enhanced insulin sensitivity and lipid profile, also may contribute to disease modulation.17
Limitations—Because our analysis was limited to 3 case reports, the strength of the evidence is limited. These case reports also lack the standardized use of the Hidradenitis Suppurativa Clinical Response scoring system that generally is found in randomized controlled trials (RCTs). The lack of RCTs precludes definitive conclusions about efficacy. Future directions include the need for well-designed RCTs with large sample sizes to confirm findings, assessment of long-term safety and tolerability in patients with HS, and further research into the molecular mechanisms by which GLP-1 RAs affect HS pathophysiology. Of note, it is imperative to be aware of the medication shortage for all GLP-1 RAs when prescribing these medications for patients with HS.
Conclusion
Glucagonlike peptide 1 RAs show promise as a therapeutic option for HS, especially in patients with obesity and metabolic disturbances. The observed benefits likely result from weight loss and anti-inflammatory effects. Other drugs targeting glucose-dependent insulinotropic polypeptide and glucagon also are being studied thoroughly as options for managing HS. Although preliminary results are encouraging, robust clinical trials are needed to establish efficacy, optimal dosing, and safety in this patient population.
Hidradenitis suppurativa (HS) is a chronic relapsing inflammatory skin disorder affecting apocrine gland–bearing areas such as the axillae, inguinal regions, and anogenital area.1 It manifests with painful nodules, abscesses, sinus tract formation, and scarring.2 The disease strongly impacts patients’ quality of life due to pain, malodor, and psychosocial burden.3
The exact etiology of HS is multifactorial, involving genetic predisposition, mechanical stress, hormonal influences, dysbiosis, and immune dysregulation.4 Obesity and metabolic syndrome are highly prevalent among patients with HS and are considered exacerbating factors.5 Adipose tissue contributes to systemic inflammation through the secretion of proinflammatory cytokines such as tumor necrosis factor (TNF) α and interleukins (ILs).6
Management of HS includes lifestyle modifications, medical therapy, and surgical interventions. Medical treatments encompass antibiotics, retinoids, hormonal therapy, immunosuppressants, and immunomodulators such as anti-TNF and anti–IL-17 agents.7 Despite available therapies, many patients have suboptimal responses or experience adverse effects and dramatic reductions in their quality of life.3
Glucagonlike peptide 1 receptor agonists (GLP-1 RAs) are incretin-based therapies used in type 2 diabetes and obesity management.8 They enhance insulin secretion, suppress glucagon release, delay gastric emptying, and promote satiety.9 Beyond glycemic control, GLP-1 RAs exhibit anti-inflammatory properties and cardiovascular benefits.10
Given the high prevalence of obesity and metabolic syndrome in patients with HS as well as the anti-inflammatory effects of GLP-1 RAs, these agents may offer therapeutic benefits in HS.11 We conducted a systematic review to evaluate the existing evidence on the efficacy and safety of GLP-1 RAs in the treatment of HS.
Methods
A systematic review was conducted via a PubMed search of articles indexed for MEDLINE in October 2024, following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines12 using the terms hidradenitis suppurativa OR acne inversa AND GLP-1 receptor agonist OR glucagon-like peptide-1 receptor agonist OR liraglutide OR semaglutide OR exenatide OR dulaglutide. No filters were applied to limit the search by language or publication date.
Inclusion criteria were clinical trials, observational studies (cohort, case control, cross-sectional), and case reports/series involving patients diagnosed with HS treated with GLP-1 RAs. Outcomes of interest included clinical improvement in HS severity (eg, lesion count, pain assessment, HS-specific scores), safety, and adverse events. Exclusion criteria included animal studies or in vitro experiments, reviews, editorials, and opinion pieces without original patient data; studies not in English; and studies not reporting clinical outcomes related to HS.
Two independent reviewers (N.R.K. and S.K.C.) screened the titles and abstracts for relevance. Full-text articles of potentially eligible studies were retrieved for detailed evaluation. Data extracted included study design, patient demographics, intervention details, outcomes, and adverse events. Discrepancies were resolved through discussion.
Results
The initial search yielded 11 articles (Figure). After screening titles and abstracts, 9 articles were selected for full-text review. Of these, 3 articles met the inclusion criteria. These studies included 3 case reports. Interventions involved liraglutide (2 reports)13,14 and semaglutide15 (1 report)(Table). The patient population consisted of adult patients with HS with comorbid diabetes, obesity, and/or metabolic syndrome.


Jennings et al13 reported a 31-year-old obese woman with a history of smoking and Hurley stage 2 HS, a Hidradenitis Suppurativa Physician’s Global Assessment score of 4, a Dermatology Life Quality Index score of 24, and a body mass index of 45.3. She was treated with liraglutide monotherapy, starting with 0.6 mg subcutaneously once daily then titrating weekly to 1.8 mg subcutaneously. After 4 weeks, outcomes showed a reduction in Hidradenitis Suppurativa Physician’s Global Assessment (score=1) and Dermatology Life Quality Index (score=14) scores, and the patient lost 4.5 kg from baseline. The patient’s Hurley stage decreased from 2 to 1. After another 4 weeks, the patient’s weight decreased by a further 2 kg and HS remained controlled. No adverse events were recorded.
Khandalavala14 reported a single case of a 19-year-old woman with severe HS, obesity, and metabolic syndrome of 8 years’ duration treated with liraglutide. The patient had a weight of 215 lb with a body mass index of 37. With a combination of metformin 2000 mg/d, liraglutide 0.6 mg/d subcutaneously increased to 1.8 mg/d over 2 months, levonorgestrel-ethinyl estradiol (no dosage listed), dapsone 100 mg/d, and finasteride 5 mg/d, there was a marked reduction in nodules and abscesses after 6 months, with a weight loss of 40 lb (19% body weight). No adverse events were reported.
Mainville et al15 described a 59-year-old woman with refractory HS who showed improvement with a combination of intravenous ertapenem 1 g/d for 6 weeks, minocycline 100 mg/d for 3 months, metformin 500 mg three times daily for 2 months, doxycycline 100 mg/d to bridge to adalimumab (160 mg subcutaneously starting dose then 80 mg subcutaneously), and semaglutide (no dosage listed). After semaglutide was introduced, the patient lost 10 kg. The only adverse event was diarrhea.
Comment
The limited but growing body of evidence suggests that GLP-1 RAs may be beneficial in managing HS, particularly in patients with comorbid obesity. Treatment with liraglutide or semaglutide was associated with marked improvements in clinical severity scores, lesion counts, pain reduction, and quality of life.
As adjunct therapy, GLP-1 RAs could serve alongside standard HS treatments such as antibiotics and biologics. Addressing obesity, a known risk factor and disease modifier in HS, may lead to better disease control. The therapeutic benefits of GLP-1 RAs in HS are attributed to weight loss, which reduces adipose tissue and systemic inflammation.16 The anti-inflammatory effects of GLP-1 RAs involve the reduction of proinflammatory cytokines such as IL-6 and TNF-α.17 Metabolic improvements, including enhanced insulin sensitivity and lipid profile, also may contribute to disease modulation.17
Limitations—Because our analysis was limited to 3 case reports, the strength of the evidence is limited. These case reports also lack the standardized use of the Hidradenitis Suppurativa Clinical Response scoring system that generally is found in randomized controlled trials (RCTs). The lack of RCTs precludes definitive conclusions about efficacy. Future directions include the need for well-designed RCTs with large sample sizes to confirm findings, assessment of long-term safety and tolerability in patients with HS, and further research into the molecular mechanisms by which GLP-1 RAs affect HS pathophysiology. Of note, it is imperative to be aware of the medication shortage for all GLP-1 RAs when prescribing these medications for patients with HS.
Conclusion
Glucagonlike peptide 1 RAs show promise as a therapeutic option for HS, especially in patients with obesity and metabolic disturbances. The observed benefits likely result from weight loss and anti-inflammatory effects. Other drugs targeting glucose-dependent insulinotropic polypeptide and glucagon also are being studied thoroughly as options for managing HS. Although preliminary results are encouraging, robust clinical trials are needed to establish efficacy, optimal dosing, and safety in this patient population.
- Vinkel C, Thomsen SF. Hidradenitis suppurativa: causes, features, and current treatments. J Clin Aesthet Dermatol. 2018;11:17-23.
- Napolitano M, Megna M, Timoshchuk EA, et al. Hidradenitis suppurativa: from pathogenesis to diagnosis and treatment. Clin Cosmet Investig Dermatol. 2017;10:105-115. doi:10.2147/CCID.S111019
- Chernyshov PV, Finlay AY, Tomas-Aragones L, et al. Quality of life in hidradenitis suppurativa: an update. Int J Environ Res Public Health. 2021;18:6131. doi:10.3390/ijerph18116131
- Seyed Jafari SM, Hunger RE, Schlapbach C. Hidradenitis suppurativa: current understanding of pathogenic mechanisms and suggestion for treatment algorithm. Front Med (Lausanne). 2020;7:68. doi:10.3389/fmed.2020.00068
- Alotaibi HM. Incidence, risk factors, and prognosis of hidradenitis suppurativa across the globe: insights from the literature. Clin Cosmet Investig Dermatol. 2023;16:545-552. doi:10.2147/CCID.S402453
- Vossen ARJV, van der Zee HH, Prens EP. Hidradenitis suppurativa: a systematic review integrating inflammatory pathways into a cohesive pathogenic model. Front Immunol. 2018;9:2965. doi:10.3389/fimmu.2018.02965
- Orenstein LAV, Nguyen TV, Damiani G, et al. Medical and surgical management of hidradenitis suppurativa: a review of international treatment guidelines and implementation in general dermatology practice. Dermatology. 2020;236:393-412. doi:10.1159/000507323
- Brown E, Cuthbertson DJ, Wilding JP. Newer GLP-1 receptor agonists and obesity-diabetes. Peptides. 2018;100:61-67. doi:10.1016/j.peptides.2017.12.009
- Cornell S. A review of GLP‐1 receptor agonists in type 2 diabetes: a focus on the mechanism of action of once‐weekly agents. J Clin Pharm Ther. 2020;45(suppl 1):17-27. doi:10.1111/jcpt.13230
- Lee YS, Jun HS. Anti-inflammatory effects of GLP-1-based therapies beyond glucose control. Mediators Inflamm. 2016;2016:3094642. doi:10.1155/2016/3094642
- Mintoff D, Benhadou F, Pace NP, et al. Metabolic syndrome and hidradenitis suppurativa: epidemiological, molecular, and therapeutic aspects. Int J Dermatol. 2022;61:1175-1186. doi:10.1111/ijd.15910
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi:10.1136/bmj.n71
- Jennings L, Nestor L, Molloy O, et al. The treatment of hidradenitis suppurativa with the glucagon-like peptide-1 agonist liraglutide. Br J Dermatol. 2017;177:858-859. doi:10.1111/bjd.15233
- Khandalavala BN. A disease-modifying approach for advanced hidradenitis suppurativa (regimen with metformin, liraglutide, dapsone, and finasteride): a case report. Case Rep Dermatol. 2017;9:70-78. doi:10.1159/000473873
- Mainville L, MacHaalany J, Veillette H. Hidradenitis suppurativa patient requiring cardiac procedure with inguinal access: case management with ertapenem. SAGE Open Med Case Rep. 2024;12:2050313X241274819. doi:10.1177/2050313X241274819
- Hamed K, Alosaimi MN, Ali BA, et al. Glucagon-like peptide-1 (GLP-1) receptor agonists: exploring their impact on diabetes, obesity, and cardiovascular health through a comprehensive literature review. Cureus. 2024;16:E68390. doi:10.7759/cureus.68390
- Alharbi SH. Anti-inflammatory role of glucagon-like peptide 1 receptor agonists and its clinical implications. Ther Adv Endocrinol Metab. 2024;15:20420188231222367. doi:10.1177/20420188231222367
- Vinkel C, Thomsen SF. Hidradenitis suppurativa: causes, features, and current treatments. J Clin Aesthet Dermatol. 2018;11:17-23.
- Napolitano M, Megna M, Timoshchuk EA, et al. Hidradenitis suppurativa: from pathogenesis to diagnosis and treatment. Clin Cosmet Investig Dermatol. 2017;10:105-115. doi:10.2147/CCID.S111019
- Chernyshov PV, Finlay AY, Tomas-Aragones L, et al. Quality of life in hidradenitis suppurativa: an update. Int J Environ Res Public Health. 2021;18:6131. doi:10.3390/ijerph18116131
- Seyed Jafari SM, Hunger RE, Schlapbach C. Hidradenitis suppurativa: current understanding of pathogenic mechanisms and suggestion for treatment algorithm. Front Med (Lausanne). 2020;7:68. doi:10.3389/fmed.2020.00068
- Alotaibi HM. Incidence, risk factors, and prognosis of hidradenitis suppurativa across the globe: insights from the literature. Clin Cosmet Investig Dermatol. 2023;16:545-552. doi:10.2147/CCID.S402453
- Vossen ARJV, van der Zee HH, Prens EP. Hidradenitis suppurativa: a systematic review integrating inflammatory pathways into a cohesive pathogenic model. Front Immunol. 2018;9:2965. doi:10.3389/fimmu.2018.02965
- Orenstein LAV, Nguyen TV, Damiani G, et al. Medical and surgical management of hidradenitis suppurativa: a review of international treatment guidelines and implementation in general dermatology practice. Dermatology. 2020;236:393-412. doi:10.1159/000507323
- Brown E, Cuthbertson DJ, Wilding JP. Newer GLP-1 receptor agonists and obesity-diabetes. Peptides. 2018;100:61-67. doi:10.1016/j.peptides.2017.12.009
- Cornell S. A review of GLP‐1 receptor agonists in type 2 diabetes: a focus on the mechanism of action of once‐weekly agents. J Clin Pharm Ther. 2020;45(suppl 1):17-27. doi:10.1111/jcpt.13230
- Lee YS, Jun HS. Anti-inflammatory effects of GLP-1-based therapies beyond glucose control. Mediators Inflamm. 2016;2016:3094642. doi:10.1155/2016/3094642
- Mintoff D, Benhadou F, Pace NP, et al. Metabolic syndrome and hidradenitis suppurativa: epidemiological, molecular, and therapeutic aspects. Int J Dermatol. 2022;61:1175-1186. doi:10.1111/ijd.15910
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi:10.1136/bmj.n71
- Jennings L, Nestor L, Molloy O, et al. The treatment of hidradenitis suppurativa with the glucagon-like peptide-1 agonist liraglutide. Br J Dermatol. 2017;177:858-859. doi:10.1111/bjd.15233
- Khandalavala BN. A disease-modifying approach for advanced hidradenitis suppurativa (regimen with metformin, liraglutide, dapsone, and finasteride): a case report. Case Rep Dermatol. 2017;9:70-78. doi:10.1159/000473873
- Mainville L, MacHaalany J, Veillette H. Hidradenitis suppurativa patient requiring cardiac procedure with inguinal access: case management with ertapenem. SAGE Open Med Case Rep. 2024;12:2050313X241274819. doi:10.1177/2050313X241274819
- Hamed K, Alosaimi MN, Ali BA, et al. Glucagon-like peptide-1 (GLP-1) receptor agonists: exploring their impact on diabetes, obesity, and cardiovascular health through a comprehensive literature review. Cureus. 2024;16:E68390. doi:10.7759/cureus.68390
- Alharbi SH. Anti-inflammatory role of glucagon-like peptide 1 receptor agonists and its clinical implications. Ther Adv Endocrinol Metab. 2024;15:20420188231222367. doi:10.1177/20420188231222367
The Effect of GLP-1 Receptor Agonists on Hidradenitis Suppurativa: A Comprehensive Systematic Review
The Effect of GLP-1 Receptor Agonists on Hidradenitis Suppurativa: A Comprehensive Systematic Review
Practice Points
- Glucagonlike peptide 1 receptor agonists (GLP-1 RAs) can be used adjunctively to manage hidradenitis suppurativa (HS) symptoms.
- The anti-inflammatory properties of GLP-1 RAs as well as their tendency to cause weight loss and manage metabolic syndrome improve the outcome of HS.
- Although current evidence is limited to case reports, these agents can be successfully integrated with existing protocols (biologics, antibiotics, or metformin); however, clinicians should monitor for gastrointestinal adverse effects.
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
More than 40 million surgeries are performed annually in the United States, of which > 18 million are orthopedic, including > 1 million emergency orthopedic surgeries and > 2 million joint replacements.1-4 Notably, > 50% of patients undergoing orthopedic surgery are aged ≥ 65 years, a demographic shift driven by longer life expectancies and an increasing number of older adults remaining physically active for extended periods.5 Osteoarthritis, the most common joint disease, affects 10% of men and 18% of women aged > 60 years, often necessitating an orthopedic joint replacement.6 Perioperative morbidity and mortality are 2.9- to 6.7-times higher in older adults compared with younger adults.7 These risks include infection, venous thromboembolism (VTE), pressure ulcers, reduced mobility, and increased mortality. Due to the high incidence of these complications in older surgical patients, special perioperative protocols and considerations are needed when preparing an older patient for surgery. This review aims to establish concrete considerations and guidelines for perioperative management.
METHODOLOGY
A literature review of PubMed, Google Scholar, and IEEE Xplore identified research on perioperative challenges in geriatric orthopedic surgery. Keywords included geriatrics and orthopedic surgery, perioperative care in geriatric populations, and orthopedic perioperative care. Inclusion criteria were strictly defined to ensure relevance to the geriatric population, with studies focusing on patients aged ≥ 65 years. Exclusion criteria were applied to remove studies that did not involve geriatric populations or orthopedic surgeries or that lacked a clear perioperative focus. Studies were analyzed for design, interventions, and outcomes. Special attention was given to identifying common challenges and trends related to perioperative considerations. We developed a narrative report providing a comprehensive overview of the current understanding of perioperative care for geriatric orthopedic patients to offer practical recommendations for clinicians to use in their practice.
RESULTS
Consistent with the narrative review methodology described, the literature search yielded a broad range of publications addressing perioperative considerations in geriatric orthopedic patients. Articles were screened for relevance to patients aged ≥ 65 years undergoing orthopedic surgery and for applicability to perioperative optimization and postoperative outcomes. Given the heterogeneity in study design, population characteristics, and outcome reporting, findings are presented descriptively rather than being quantitatively pooled. Studies not focused on geriatric populations, orthopedic procedures, or perioperative management were excluded. Key themes included multimorbidity and comorbidity optimization, age-related physiologic changes, frailty assessment and fracture risk stratification, nutritional and bone health management, mechanism of injury considerations, prevention of postoperative complications, and the role of multidisciplinary perioperative care.
Unique Physiological Challenges
The aging process induces a range of physiological changes that can increase morbidity and mortality following surgery. One of the most essential elements to surgical recovery is wound healing, as impairments in this process can lead to adverse events, including infection, cosmetic deformity, and wound dehiscence. The general paradigm of aging involves cell senescence resulting in slower or disorganized functional capacity of these cells.8 While wound healing in older individuals was once thought to be defective, recent research has demonstrated that this process is not absent, but delayed.9
Wound healing is a tightly regulated and evolutionarily conserved process that proceeds through 3 main phases: inflammation, proliferation, and remodeling. Re-epithelialization begins with the migration of epithelial cells from hair follicles, sweat glands, or wound margins (depending on wound depth) and is influenced by oxygen levels, moisture, and growth factors.9 Several characteristics of aged skin contribute to the delayed healing process. Aged skin has fewer hair follicles and eccrine sweat glands, as well as decreased follicle thickness.10 This results in fewer proliferating cells for wound healing and lower amounts of sebum production for skin moisture.11 Furthermore, aged fibroblasts are fewer in number and less effective in synthesizing extracellular matrices, resulting in slower and less tensile wound healing.12 Additionally, microvascular changes associated with aging result in disorganized vasculature, which impairs oxygen delivery to the wound bed and diminishes the influx of proinflammatory cells necessary for effective healing.13 These senescent traits of aged skin contribute to the delayed wound healing process found in geriatric patients.
Compounding these age-related factors is the prevalence of multimorbidity, or coexisting chronic diagnoses, in 55% to 98% of older patients.14 Common comorbidities include peripheral arterial disease, chronic venous insufficiency, type 1 and type 2 diabetes, neoplasms, atherosclerotic disease, and hypertension. Older patients are more likely to be prescribed corticosteroids and chemotherapeutic agents that impair the function of inflammatory cells necessary for wound healing.15,16 Additionally, decreased mobility is more common in geriatric patients, which can increase the risk of wound formation, particularly pressure ulcers.17
Perioperative Considerations
All surgical patients undergo a formal or informal preoperative evaluation to assess their fitness for surgery, with the goal of minimizing both anesthesia-related risks and postoperative complications. A widely used tool in this assessment is the American Society of Anesthesiologists (ASA) physical status classification, which stratifies patients into 6 categories based on their medical history and overall health status.18 Classes range from healthy patients (Class I) to organ donors who are brain-dead (Class VI).
Cardiac optimization is an essential component of preoperative evaluation for older adults due to their higher risk of underlying cardiovascular disease.19 This process involves an in-depth review of the patient’s cardiac history, including the timing and nature of any prior interventions and the recurrence rate. Functional capacity is assessed through metabolic equivalents, where a threshold of > 4 metabolic equivalents (the ability to walk up a flight of stairs) is considered adequate for surgery. Risk is assessed based on the specific surgical procedure, and nonemergent orthopedic procedures are considered intermediate risk. If a patient is deemed high risk at any stage of this evaluation, further cardiac testing is indicated.
Pulmonary optimization is typically necessary for geriatric patients, who are more likely to have conditions such as chronic obstructive pulmonary disease or interstitial lung disease.14,20 In patients without severe systemic lung disease, pulmonary optimization involves assessing the functional expiratory volume and diffusing capacity for carbon monoxide. In addition, aggressive modification of risk factors, such as smoking cessation, is strongly recommended.
Additional perioperative conditions are disease-specific and involve evaluation of comorbid illnesses and recognition of absolute contraindications to noncardiac surgery. For instance, an ejection fraction of < 35%, a history of myocardial infarction within 6 months, or active diabetic ketoacidosis are all absolute contraindications to elective surgery. For orthopedic procedures, additional contraindications include symptomatic bacteremia, active joint or local tissue infection, severe malnutrition, uncontrolled metabolic syndrome or chronic disease, untreated immunodeficiency, and active deep venous thrombosis (DVT) or pulmonary embolism.21
Bone Health and Nutrition
In the context of orthopedic surgery, the hallmark of clinically defined optimal bone health is a musculoskeletal system that provides the ability for pain-free functional and occupational tasks with an adequate capacity to withstand the mechanical forces imparted by everyday life. Back pain and arthritis are the fourth- and sixth-most common complaints in primary care, underscoring suboptimal bone health management in developed countries.22
Optimizing bone health through proper nutrition is crucial in the perioperative management of geriatric orthopedic patients. The clinical diagnosis of malnutrition has well-studied associations with worse outcomes after orthopedic surgery, which include increased mortality, hospital length of stay, readmission rates, and health institution spending.23-25 Some studies show that up to 60% of geriatric patients may be malnourished.26
Regarding vitamin and mineral supplements, the general consensus before orthopedic surgery is that vitamins A, C, D, and E, and zinc are predictive in determining postoperative health.27 However, Curtis et al state that therapy should be targeted at correcting relative deficiencies; supraphysiologic concentrations of these vitamins do not appear to be helpful.27 This claim may merit serum studies to rule out deficiencies. Dietitians should be involved in the creation of a patient care plan in the spirit of multidisciplinary orthopedic surgery approaches, which have proven to result in superior patient outcomes.28 Additionally, directive counseling should be provided when necessary.
In patients with adequately managed nutrition, 7 to 10 days of diet optimization is typically sufficient, but patients with malnutrition may require sustained nutritional support for up to 6 weeks; a standardized time for adequate nutrition supplementation has not been identified.25-27 Postoperative management is similar in older patients who are malnourished and those receiving adequate nutrition after orthopedic surgery, which typically involves 3 weeks of a high-protein diet.26
Evaluating Mechanisms of Injury
Assessing the mechanism of injury (MOI) is essential to developing an appropriate and successful orthopedic treatment plan. MOI is typically categorized as low energy, which consists of ground-level falls and other minor trauma, or high energy, which can include motor vehicle crashes or falls from a height.29 Unlike younger patients who typically experience trauma from high-energy MOIs, geriatric patients often sustain fractures from low-energy MOIs. The importance of assessing MOI for the geriatric population is magnified as it provides vital clues that not only help determine the nature of the injury, but also highlight underlying frailty, comorbidities, and potential complications. Weakness or deconditioning related to older age is often not discovered before trauma, which is why assessing the MOI can provide valuable information regarding overall patient health.30
The MOI of trauma also is correlated with factors that influence postoperative recovery and overall prognosis (Figure). Falls comprise more than three-quarters of the MOI in geriatric patients with trauma, and > 90% of these falls are ground-level or other simple falls.30 Falls secondary to an intrinsic disorder, rather than an extrinsic environmental hazard, are more common in geriatric patients.31
These events may be associated with an underlying medical condition, such as osteopenia, osteoporosis, or neuromuscular disorders, such as Parkinson disease.32 They may also be attributed to normal age-related changes, such as decreased visual acuity, reduced reaction time, or mild cognitive impairment.30 An estimated 6% to 35% of geriatric patients who present to the emergency department have some degree of cognitive dysfunction.33 Accordingly, a thorough understanding of the events leading up to injury is vital for the management of older patients. Knowing the specific circumstances of a fall can provide insight into the patient’s gait, balance, and need for further investigations such as cognitive screening or evaluation of home safety. This information can guide decisions regarding preoperative optimization of medications and postoperative rehabilitation interventions.
Frailty and Risk of Fracture
Frailty is a clinical syndrome defined by overall decreased capacity for the body’s adaptive changes to various stressors.34 It is a common condition in geriatric populations due to cumulative degenerative changes and multisystem decline over a lifetime’s worth of disruptions to natural homeostasis.34 In orthopedics, frailty typically refers to musculoskeletal durability and resilience in response to mechanical forces (ie, falls, trauma, and high-acceleration movements). Globally, > 200 million people have osteoporotic frailty, leading to 9 million hip fractures annually.35 More than 30% of people aged ≥ 65 years fall ≥ 1 time per calendar year.36
Assessing frailty in geriatric patients undergoing orthopedic surgery is vital, as it predisposes patients to higher rates of morbidity, mortality, and institutionalization, particularly from falls and resultant fragility fractures.37-39 This is true for a wide range of orthopedic procedures, spanning elective to urgent surgeries and involving the axial and appendicular skeleton.40,41 Given the high rates of fractures, subsequent patient morbidity, and financial burden on the health care system, effective frailty screening is essential.
There are many strategies to assess frailty risk and subsequent fracture risk.42 Questionnaires or online medical calculators serve as easy-to-use tools for assessment of frailty or associated predictors of fragility fractures. Validated assessment tools are provided in Table 1.
Dual-emission X-ray absorptiometry is a well-established way to determine bone density and establish fracture risk. The Fried Frailty Phenotype score and Short Performance Physical Battery test are clinically applicable methods of assessing frailty in older outpatient populations. Although these examinations focus on different aspects of the patient, they have moderate agreeability, are sensitive, and can be readily performed in the clinical setting as demonstrated by a > 90% patient participation rate for both methods.42 Finally, several serum studies can be predictive of frailty, the most readily modifiable of which are vitamin D3, ferritin, albumin, and calcium.43 Although they are more invasive for the patient, serum studies can provide additional modifiable targets for perioperative optimization and contribute to risk stratification.
Risk stratification should take place around 6 weeks before surgery, which should provide adequate time for rectification of preoperative barriers to elective surgical intervention—namely nutritional status. In cases of urgent or emergent procedures (ie, femoral neck fracture with concern for avascular necrosis of the femoral head), this may not be possible but should be conducted nonetheless for patient-specific postoperative rehabilitation and risk reduction.
Postoperative Complication Risks
Postoperative complications affect nearly 15% of geriatric orthopedic patients, highlighting the need for comprehensive preoperative evaluations to assess risk factors.44 Age-related physiological changes, frailty, and comorbidities complicate recovery and management (Table 2).
Wound healing is impaired in older individuals due to suboptimal circulation and decreased oxygenation that is secondary to age-related changes, as well as the increased likelihood of comorbid conditions (eg, diabetes).7 Surgical site infections can be particularly malicious in geriatric patients, with a 4% incidence.45,46 Hospitalization can be prolonged by a mean 2 weeks, which increases the risk of hospital-associated delirium and iatrogenic complications.46 Both the mortality rate and costs associated with hospitalization are higher for older patients who develop surgical site infections compared with patients aged < 65 years, underscoring the importance of vigilant monitoring, early detection, and effective preoperative screening to identify and manage modifiable risk factors.47
Postoperative delirium is another common complication of orthopedic surgery in the geriatric population, increasing morbidity and mortality. The incidence is reported to be as high as 53.3% in the trauma setting and 28.3% in the elective setting, indicating a need to assess patient risk preoperatively.48,49 Several factors contribute to the high incidence of delirium, including advanced age, longer surgical durations, intraoperative hypotension and hypercapnia, pre-existing cognitive dysfunction, and postoperative sleep disorders.50
VTE is another common cause of complications following orthopedic surgery. The development of DVT can lead to subsequent pulmonary embolism, which can be fatal. Orthopedic surgery patients are already at higher risk of DVT and VTE than other surgical patients, with an incidence as high as 40% to 60%, though it is frequently asymptomatic.51,52 Geriatric patients may be more likely to have concurrent comorbidities that increase hypercoagulability.53 Congestive heart failure, chronic kidney disease, and cardiovascular disease are all more common in older individuals and can increase the risk of VTE by 2-fold.53 While anticoagulation is the standard of care to prevent VTE after orthopedic surgery, geriatric patients require more careful monitoring due to the higher incidence of bleeding complications. Additionally, early postoperative mobilization is critical to reduce the risk of DVT without significantly increasing pain or causing other adverse events.54
Respiratory complications are common after orthopedic surgery, particularly atelectasis and bronchospasm, which can result from intraoperative mechanical ventilation.55 While these conditions are typically self-limiting, more severe respiratory issues such as pneumonia are a significant concern because they may lead to the need for mechanical ventilation and admission to the intensive care unit (ICU). The more severe complications have an incidence of about 1% to 2% in orthopedic surgery patients.56 Preventive strategies, such as respiratory physiotherapy and guided breathing exercises, are crucial to minimize perioperative pulmonary complications and promote optimal recovery. Addressing these challenges through early intervention is essential to improve outcomes.
Multidisciplinary Perioperative Care
Multidisciplinary care in orthopedic surgery involves collaborative management of patient care by general practitioners, surgeons, anesthesiologists, dietitians, physical and occupational therapists, inpatient health care practitioners (HCPs), and social services. The goal of this form of care is to provide a longitudinal sequence of health-optimization tactics that prepare a patient for surgery and give them the best chance of postoperative recovery.
Given that many aspects of geriatric health play a role in orthopedic postoperative outcomes, there are many preoperative factors to consider. As previously discussed, preoperative evaluation of geriatric patients should include nutritional and fragility screening to determine surgical candidacy and target modifiable risk factors for risk reduction. This screening can be conducted by primary care practitioners and orthopedic surgeons in an outpatient setting. A multidisciplinary approach benefits patients by decreasing time to surgery.35
Several large studies have demonstrated the positive influence of a multidisciplinary approach on patient-oriented outcomes in orthopedic patients. Incorporation of this style of care in contrast to surgeon-led perioperative optimization leads to fewer floor and ICU admissions, shorter lengths of stay, and decreased mortality rates.35,57 These findings are broadly applicable to a wide range of orthopedic surgeries and even surgeries outside of the musculoskeletal system.58,59 In addition, this strategy has demonstrated reduced in-hospital health care costs due to shorter lengths of stay and fewer ICU admissions. Physical and occupational therapy also have irreplaceable roles in outcomes after orthopedic surgeries. They have independently been shown to decrease pain, increase range of motion, and increase functionality in daily life.60 These aspects of recovery are essential for geriatric well-being.
Screening Tools
The World Health Organization FRAX fracture risk assessment tool (www.fraxplus.org/calculation-tool) was developed to identify patients at high risk of fracture and subsequent complications and to guide clinical decision-making regarding pharmacologic interventions.61 FRAX calculates the 10-year probability of fracture based on demographic factors, such as age and body mass index, clinical measures (eg, femoral neck bone mineral density), and risk factors (eg, prior fragility fractures, substance use history, and prolonged glucocorticoid use).61 The online tool is easy to use, making it a valuable resource for assessing fracture risk and determining appropriate treatment strategies.
The fatigue, resistance, ambulation, illnesses, loss of weight (FRAIL) scale assesses frailty in older adults. The scale classifies patients into 3 categories: robust, prefrail, and frail. The frail category is associated with an increased frequency of hip fracture and an elevated ASA class.62 Additionally, the FRAIL scale has demonstrated value in predicting hospital length of stay and the risk of postoperative complications.62 It also has shown utility in quantifying frailty status, which is traditionally challenging to assess systematically.63
The Mini-Cog is commonly used in geriatric populations to screen for cognitive impairment. Preoperative Mini-Cog screening has been shown to predict the development of postoperative complications.64 Geriatric patients who screened positive for cognitive impairment prior to orthopedic surgery were more likely to develop postoperative delirium, require alternative discharge disposition, and have a longer hospital length of stay.64 Mini-Cog serves as an important preoperative tool for identifying patients who may benefit from closer postoperative monitoring and tailored care.
The Comprehensive Geriatric Assessment (CGA) is a multidimensional evaluation that has been validated for use in geriatric patients undergoing orthopedic surgery.65 The CGA assesses functional status and the ability to perform activities of daily living (ADLs), such as eating, dressing, and ambulating. Poor ADLs are associated with increased risk of falls and cardiopulmonary complications. The CGA allows HCPs to identify patients at higher risk of complications and tailor interventions that optimize functional recovery during the perioperative period.
Nutritional screening is another component of preoperative evaluation in older adults undergoing orthopedic surgery. The Perioperative Nutrition Screen is a preoperative phone assessment of unintentional weight loss in the past 6 months.66 Patients who screen positive are asked to come in for a preoperative visit with a registered dietitian who can further evaluate the nutritional status of the patient.
The Mini Nutritional Assessment Short Form (MNA-SF), Malnutrition Universal Screening Tool, and Nutrition Risk Screening 2002 have all been validated for use in older patients undergoing orthopedic surgery. Among these, the MNA-SF has demonstrated superior utility in predicting hospital readmission and mortality.67 Given the established links between malnutrition and poor surgical outcomes, routine nutritional screening is important for identifying geriatric patients who may require preoperative nutritional interventions.
CONCLUSIONS AND RECOMMENDATIONS
Perioperative management of geriatric patients undergoing orthopedic surgery requires an assessment and strategy focused on risk stratification, patient optimization, and mitigation of potential complications and mortality. Due to the complexity and comprehensive nature of an optimal perioperative plan, creating the plan early is essential to ensure adequate time for patient optimization and care coordination.
Nutrition plays a critical role in the success of surgical procedures, and orthopedics is no exception. Extra care should be taken to preoperatively optimize patient bone health before surgical intervention to enhance recovery and reduce the risk of complications. After an appropriate patient history and clinical picture are gathered, screening tools should be used on a case-by-case basis to further characterize comorbid conditions that may contribute to suboptimal outcomes. Additionally, given the proven association between frailty and fracture risk, frailty serves as a readily quantifiable predictor of patient-oriented outcomes. This should be assessed preoperatively with appropriate risk-stratification tools to determine appropriate postoperative measures to prevent morbidity and mortality.
Orthopedic surgery is increasingly common in geriatric patients, who face higher perioperative risks due to age-related physiological changes, multimorbidity, and frailty. Optimizing preoperative assessment and adopting a multidisciplinary approach—integrating surgeons, anesthesiologists, physical therapists, and dietitians—can improve outcomes, reduce complications, and enhance recovery. The successful use of the tools and strategies outlined in this article by primary care should facilitate access to and recovery from orthopedic surgery in the geriatric population.
Dobson GP. Trauma of major surgery: a global problem that is not going away. Int J Surg. 2020;81:47-54. doi:10.1016/j.ijsu.2020.07.017
United States (US) orthopedic procedures count by segments and forecast to 2030. GlobalData. February 17, 2023. Accessed April 29, 2026. https://www.globaldata.com/store/report/usa-orthopedic-procedures-analysis/
Jarman MP, Weaver MJ, Haider AH, Salim A, Harris MB. The national burden of orthopedic injury: cross-sectional estimates for trauma system planning and optimization. J Surg Res. 2020;249:197-204. doi:10.1016/j.jss.2019.12.023
Hegde V, Stambough JB, Levine BR, et al. Highlights of the 2022 American Joint Replacement Registry Annual Report. Arthroplast Today. 2023;21:101137. doi:10.1016/j.artd.2023.101137
Nakamura K, Ogata T. Locomotive syndrome: definition and management. Clin Rev Bone Miner Metab. 2016;14:56-67. doi:10.1007/s12018-016-9208-2
Glyn-Jones S, Palmer AJR, Agricola R, et al. Osteoarthritis. Lancet. 2015;386:376-387. doi:10.1016/S0140-6736(14)60802-3
Hughes S, Leary A, Zweizig S, Cain J. Surgery in elderly people: preoperative, operative and postoperative care to assist healing. Best Pract Res Clin Obstet Gynaecol. 2013;27:753-765. doi:10.1016/j.bpobgyn.2013.02.006
Regulski MJ. Cellular senescence: what, why, and how. Wounds. 2017;29:168-174.
Kremer M, Burkemper N. Aging skin and wound healing. Clin Geriatr Med. 2024;40:1-10. doi:10.1016/j.cger.2023.06.001
Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol. 1986;15:571-585. doi:10.1016/S0190-9622(86)70208-9
Van Neste D, Tobin DJ. Hair cycle and hair pigmentation: dynamic interactions and changes associated with aging. Micron. 2004;35:193-200. doi:10.1016/j.micron.2003.11.006
Salzer MC, Lafzi A, Berenguer-Llergo A, et al. Identity noise and adipogenic traits characterize dermal fibroblast aging. Cell. 2018;175:1575-1590.e22. doi:10.1016/j.cell.2018.10.012
Jin K. A microcirculatory theory of aging. Aging Dis. 2019;10:676-683. doi:10.14336/AD.2019.0315
Marengoni A, Angleman S, Melis R, et al. Aging with multimorbidity: a systematic review of the literature. Ageing Res Rev. 2011;10:430-439. doi:10.1016/j.arr.2011.03.003
Waljee AK, Rogers MAM, Lin P, et al. Short term use of oral corticosteroids and related harms among adults in the United States: population based cohort study. BMJ. 2017;357:j1415. doi:10.1136/bmj.j1415
Given B, Given CW. Older adults and cancer treatment. Cancer. 2008;113:3505-3511. doi:10.1002/cncr.23939
Ferrucci L, Cooper R, Shardell M, Simonsick EM, Schrack JA, Kuh D. Age-related change in mobility: perspectives from life course epidemiology and geroscience. J Gerontol A Biol Sci Med Sci. 2016;71:1184-1194. doi:10.1093/gerona/glw043
Mayhew D, Mendonca V, Murthy BVS. A review of ASA physical status - historical perspectives and modern developments. Anaesthesia. 2019;74:373-379. doi:10.1111/anae.14569
Eagle KA, Berger PB, Calkins H, et al. ACC/AHA guideline update for perioperative cardiovascular evaluation for noncardiac surgery—executive summary a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1996 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation. 2002;105:1257-1267. doi:10.1161/circ.105.10.1257
Bapoje SR, Whitaker JF, Schulz T, Chu ES, Albert RK. Preoperative evaluation of the patient with pulmonary disease. Chest. 2007;132:1637-1645. doi:10.1378/chest.07-0347
Choe H, Indelli PF, Ricciardi B, et al. What are the absolute contraindications for elective total knee or hip arthroplasty? J Arthroplasty. 2025;40(2 suppl 1):S45-S47. doi:10.1016/j.arth.2024.10.041
Finley CR, Chan DS, Garrison S, et al. What are the most common conditions in primary care? Systematic review. Can Fam Physician. 2018;64:832-840.
Vaid S, Bell T, Grim R, Ahuja V. Predicting risk of death in general surgery patients on the basis of preoperative variables using American College of Surgeons National Surgical Quality Improvement Program data. Perm J. 2012;16:10-17. doi:10.7812/TPP/12-019
Correia MTD, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22:235-239. doi:10.1016/S0261-5614(02)00215-7
Friedman J, Lussiez A, Sullivan J, Wang S, Englesbe M. Implications of sarcopenia in major surgery. Nutr Clin Pract. 2015;30:175-179. doi:10.1177/0884533615569888
Hirsch KR, Wolfe RR, Ferrando AA. Pre- and post-surgical nutrition for preservation of muscle mass, strength, and functionality following orthopedic surgery. Nutrients. 2021;13:1675. doi:10.3390/nu13051675
Curtis W, Choi T, Ahmad A, Shultz C. Perioperative nutritional considerations in orthopaedic surgery: a review of the literature. West J Orthop. 2023;12:1. https://digitalrepository.unm.edu/wjo/vol12/iss1/1
Wischmeyer PE, Carli F, Evans DC, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on nutrition screening and therapy within a surgical enhanced recovery pathway. Anesth Analg. 2018;126:1883-1895. doi:10.1213/ANE.0000000000002743
Mun F, Ringenbach K, Baer B, et al. Factors influencing geriatric orthopaedic trauma mortality. Injury. 2022;53:919-924. doi:10.1016/j.injury.2022.01.005
Bonne S, Schuerer DJE. Trauma in the older adult: epidemiology and evolving geriatric trauma principles. Clin Geriatr Med. 2013;29:137-150. doi:10.1016/j.cger.2012.10.008
Montero-Odasso MM. Falls as a geriatric syndrome: mechanisms and risk identification. In: Duque G, Kiel DP, eds. Osteoporosis in Older Persons: Advances in Pathophysiology and Therapeutic Approaches. 2nd ed. Springer International Publishing; 2016:171-186. doi:10.1007/978-3-319-25976-5_10
Lach HW, Reed AT, Arfken CL, et al. Falls in the elderly: reliability of a classification system. J Am Geriatr Soc. 1991;39:197-202. doi:10.1111/j.1532-5415.1991.tb01626.x
Carpenter CR, DesPain B, Keeling TN, Shah M, Rothenberger M. The six-item screener and AD8 for the detection of cognitive impairment in geriatric emergency department patients. Ann Emerg Med. 2011;57:653-661. doi:10.1016/j.annemergmed.2010.06.560
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381:752-762. doi:10.1016/S0140-6736(12)62167-9
Patel JN, Klein DS, Sreekumar S, Liporace FA, Yoon RS. Outcomes in multidisciplinary team-based approach in geriatric hip fracture care: a systematic review. J Am Acad Orthop Surg. 2020;28:128-133. doi:10.5435/JAAOS-D-18-00425
Amador LF, Loera JA. Preventing postoperative falls in the older adult. J Am Coll Surg. 2007;204:447-453. doi:10.1016/j.jamcollsurg.2006.12.010
Tembo MC, Holloway-Kew KL, Mohebbi M, et al. The association between a fracture risk tool and frailty: Geelong Osteoporosis Study. BMC Geriatr. 2020;20:196. doi:10.1186/s12877-020-01595-8
Demiris¸ B, Basat S, Kurt F, Aksakal B, Basat O. Evaluation of the relationship between frailty and fracture risk using Fracture Risk Assessment Tool in patients 65 years and over. South Clin Istanb Eurasia. 2023;34:42-48. doi:10.14744/scie.2022.66564
Partridge JSL, Harari D, Dhesi JK. Frailty in the older surgical patient: a review. Age Ageing. 2012;41:142-147. doi:10.1093/ageing/afr182
Mamtora PH, Fortier MA, Barnett SR, Schmid LN, Kain ZN. Peri-operative management of frailty in the orthopedic patient. J Orthop. 2020;22:304-307. doi:10.1016/j.jor.2020.05.024
Leven DM, Lee NJ, Kim JS, et al. Frailty is predictive of adverse postoperative events in patients undergoing lumbar fusion. Global Spine J. 2017;7:529-535. doi:10.1177/2192568217700099
Pritchard JM, Kennedy CC, Karampatos S, et al. Measuring frailty in clinical practice: a comparison of physical frailty assessment methods in a geriatric out-patient clinic. BMC Geriatr. 2017;17:264. doi:10.1186/s12877-017-0623-0
Kumar A, Dhar M, Agarwal M, Mukherjee A, Saxena V. Predictors of frailty in the elderly population: a cross-sectional study at a tertiary care center. Cureus. 2022;14:e30557. doi:10.7759/cureus.30557
Scarano KA, Philp FH, Westrick ER, Altman GT, Altman DT. Evaluating postoperative complications and outcomes of orthopedic fracture repair in nonagenarian patients. Geriatr Orthop Surg Rehabil. 2018;9:2151459318758106. doi:10.1177/2151459318758106
Liang Z, Rong K, Gu W, et al. Surgical site infection following elective orthopaedic surgeries in geriatric patients: incidence and associated risk factors. Int Wound J. 2019;16:773-780. doi:10.1111/iwj.13096
Ren M, Liang W, Wu Z, Zhao H, Wang J. Risk factors of surgical site infection in geriatric orthopedic surgery: a retrospective multicenter cohort study. Geriatr Gerontol Int. 2019;19:213-217. doi:10.1111/ggi.13590
Kaye KS, Schmader KE, Sawyer R. Surgical site infection in the elderly population. Clin Infect Dis. 2004;39:1835-1841. doi:10.1086/425744
Bruce AJ, Ritchie CW, Blizard R, Lai R, Raven P. The incidence of delirium associated with orthopedic surgery: a meta-analytic review. Int Psychogeriatr. 2007;19:197-214. doi:10.1017/S104161020600425X
Williams-Russo P, Urquhart BL, Sharrock NE, Charlson ME. Post-operative delirium: predictors and prognosis in elderly orthopedic patients. J Am Geriatr Soc. 1992;40:759-767. doi:10.1111/j.1532-5415.1992.tb01846.x
Wang J, Li Z, Yu Y, Li B, Shao G, Wang Q. Risk factors contributing to postoperative delirium in geriatric patients postorthopedic surgery. Asia Pac Psychiatry. 2015;7:375-382. doi:10.1111/appy.12193
Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126:338S-400S. doi:10.1378/chest.126.3_suppl.338S
Kahn SR, Shivakumar S. What’s new in VTE risk and prevention in orthopedic surgery. Res Pract Thromb Haemost. 2020;4:366-376. doi:10.1002/rth2.12323
Uzel K, Azboy I·, Parvizi J. Venous thromboembolism in orthopedic surgery: global guidelines. Acta Orthop Traumatol Turc. 2023;57:192-203. doi:10.5152/j.aott.2023.23074
Peck M, Holthaus A, Kingsbury K, Salsberry MG, Duggirala V. Mobility in acute care for geriatric patients with orthopedic conditions: a review of recent literature. Curr Geri Rep. 2020;9:300-310. doi:10.1007/s13670-020-00347-1
Leme LEG, Sitta MC, Toledo M, Henriques SS. Orthopedic surgery among the elderly: clinical characteristics. Rev Bras Ortop. 2015;46:238-246. doi:10.1016/S2255-4971(15)30189-0
Malcolm TL, Knezevic NN, Zouki CC, Tharian AR. Pulmonary complications after hip and knee arthroplasty in the United States, 2004-2014. Anesth Analg. 2020;130:917-924. doi:10.1213/ANE.0000000000004265
Kamal T, Conway RM, Littlejohn I, Ricketts D. The role of a multidisciplinary pre-assessment clinic in reducing mortality after complex orthopaedic surgery. Ann R Coll Surg Engl. 2011;93:149-151. doi:10.1308/003588411X561026
Davis MJ, Luu BC, Raj S, Abu-Ghname A, Buchanan EP. Multidisciplinary care in surgery: Are team-based interventions cost-effective? Surgeon. 2021;19:49-60. doi:10.1016/j.surge.2020.02.005
Frassanito L, Vergari A, Nestorini R, et al. Enhanced recovery after surgery (ERAS) in hip and knee replacement surgery: description of a multidisciplinary program to improve management of the patients undergoing major orthopedic surgery. Musculoskelet Surg. 2020;104:87-92. doi:10.1007/s12306-019-00603-4
Reddy RS, Alahmari KA, Alshahrani MS, et al. Exploring the impact of physiotherapy on health outcomes in older adults with chronic diseases: a cross-sectional analysis. Front Public Health. 2024;12:1415882. doi:10.3389/fpubh.2024.1415882
Watts NB. The Fracture Risk Assessment Tool (FRAX®): applications in clinical practice. J Womens Health (Larchmt). 2011;20:525-531. doi:10.1089/jwh.2010.2294
Gleason LJ, Benton EA, Alvarez-Nebreda ML, Weaver MJ, Harris MB, Javedan H. FRAIL questionnaire screening tool and short-term outcomes in geriatric fracture patients. J Am Med Dir Assoc. 2017;18:1082-1086. doi:10.1016/j.jamda.2017.07.005
Kojima G. Frailty defined by FRAIL scale as a predictor of mortality: a systematic review and meta-analysis. J Am Med Dir Assoc. 2018;19:480-483. doi:10.1016/j.jamda.2018.04.006
Culley DJ, Flaherty D, Fahey MC, et al. Poor performance on a preoperative cognitive screening test predicts postoperative complications in older orthopedic surgical patients. Anesthesiology. 2017;127:765-774. doi:10.1097/ALN.0000000000001859
Kong C, Zhang Y, Wang C, et al. Comprehensive geriatric assessment for older orthopedic patients and analysis of risk factors for postoperative complications. BMC Geriatr. 2022;22:644. doi:10.1186/s12877-022-03328-5
Williams DGA, Wischmeyer PE. Perioperative nutrition care of orthopedic surgery patient. Tech Orthop. 2020;35:15-18. doi:10.1097/BTO.0000000000000412
Koren-Hakim T, Weiss A, Hershkovitz A, et al. Comparing the adequacy of the MNA-SF, NRS-2002 and MUST nutritional tools in assessing malnutrition in hip fracture operated elderly patients. Clin Nutr. 2016;35:1053-1058. doi:10.1016/j.clnu.2015.07.014
More than 40 million surgeries are performed annually in the United States, of which > 18 million are orthopedic, including > 1 million emergency orthopedic surgeries and > 2 million joint replacements.1-4 Notably, > 50% of patients undergoing orthopedic surgery are aged ≥ 65 years, a demographic shift driven by longer life expectancies and an increasing number of older adults remaining physically active for extended periods.5 Osteoarthritis, the most common joint disease, affects 10% of men and 18% of women aged > 60 years, often necessitating an orthopedic joint replacement.6 Perioperative morbidity and mortality are 2.9- to 6.7-times higher in older adults compared with younger adults.7 These risks include infection, venous thromboembolism (VTE), pressure ulcers, reduced mobility, and increased mortality. Due to the high incidence of these complications in older surgical patients, special perioperative protocols and considerations are needed when preparing an older patient for surgery. This review aims to establish concrete considerations and guidelines for perioperative management.
METHODOLOGY
A literature review of PubMed, Google Scholar, and IEEE Xplore identified research on perioperative challenges in geriatric orthopedic surgery. Keywords included geriatrics and orthopedic surgery, perioperative care in geriatric populations, and orthopedic perioperative care. Inclusion criteria were strictly defined to ensure relevance to the geriatric population, with studies focusing on patients aged ≥ 65 years. Exclusion criteria were applied to remove studies that did not involve geriatric populations or orthopedic surgeries or that lacked a clear perioperative focus. Studies were analyzed for design, interventions, and outcomes. Special attention was given to identifying common challenges and trends related to perioperative considerations. We developed a narrative report providing a comprehensive overview of the current understanding of perioperative care for geriatric orthopedic patients to offer practical recommendations for clinicians to use in their practice.
RESULTS
Consistent with the narrative review methodology described, the literature search yielded a broad range of publications addressing perioperative considerations in geriatric orthopedic patients. Articles were screened for relevance to patients aged ≥ 65 years undergoing orthopedic surgery and for applicability to perioperative optimization and postoperative outcomes. Given the heterogeneity in study design, population characteristics, and outcome reporting, findings are presented descriptively rather than being quantitatively pooled. Studies not focused on geriatric populations, orthopedic procedures, or perioperative management were excluded. Key themes included multimorbidity and comorbidity optimization, age-related physiologic changes, frailty assessment and fracture risk stratification, nutritional and bone health management, mechanism of injury considerations, prevention of postoperative complications, and the role of multidisciplinary perioperative care.
Unique Physiological Challenges
The aging process induces a range of physiological changes that can increase morbidity and mortality following surgery. One of the most essential elements to surgical recovery is wound healing, as impairments in this process can lead to adverse events, including infection, cosmetic deformity, and wound dehiscence. The general paradigm of aging involves cell senescence resulting in slower or disorganized functional capacity of these cells.8 While wound healing in older individuals was once thought to be defective, recent research has demonstrated that this process is not absent, but delayed.9
Wound healing is a tightly regulated and evolutionarily conserved process that proceeds through 3 main phases: inflammation, proliferation, and remodeling. Re-epithelialization begins with the migration of epithelial cells from hair follicles, sweat glands, or wound margins (depending on wound depth) and is influenced by oxygen levels, moisture, and growth factors.9 Several characteristics of aged skin contribute to the delayed healing process. Aged skin has fewer hair follicles and eccrine sweat glands, as well as decreased follicle thickness.10 This results in fewer proliferating cells for wound healing and lower amounts of sebum production for skin moisture.11 Furthermore, aged fibroblasts are fewer in number and less effective in synthesizing extracellular matrices, resulting in slower and less tensile wound healing.12 Additionally, microvascular changes associated with aging result in disorganized vasculature, which impairs oxygen delivery to the wound bed and diminishes the influx of proinflammatory cells necessary for effective healing.13 These senescent traits of aged skin contribute to the delayed wound healing process found in geriatric patients.
Compounding these age-related factors is the prevalence of multimorbidity, or coexisting chronic diagnoses, in 55% to 98% of older patients.14 Common comorbidities include peripheral arterial disease, chronic venous insufficiency, type 1 and type 2 diabetes, neoplasms, atherosclerotic disease, and hypertension. Older patients are more likely to be prescribed corticosteroids and chemotherapeutic agents that impair the function of inflammatory cells necessary for wound healing.15,16 Additionally, decreased mobility is more common in geriatric patients, which can increase the risk of wound formation, particularly pressure ulcers.17
Perioperative Considerations
All surgical patients undergo a formal or informal preoperative evaluation to assess their fitness for surgery, with the goal of minimizing both anesthesia-related risks and postoperative complications. A widely used tool in this assessment is the American Society of Anesthesiologists (ASA) physical status classification, which stratifies patients into 6 categories based on their medical history and overall health status.18 Classes range from healthy patients (Class I) to organ donors who are brain-dead (Class VI).
Cardiac optimization is an essential component of preoperative evaluation for older adults due to their higher risk of underlying cardiovascular disease.19 This process involves an in-depth review of the patient’s cardiac history, including the timing and nature of any prior interventions and the recurrence rate. Functional capacity is assessed through metabolic equivalents, where a threshold of > 4 metabolic equivalents (the ability to walk up a flight of stairs) is considered adequate for surgery. Risk is assessed based on the specific surgical procedure, and nonemergent orthopedic procedures are considered intermediate risk. If a patient is deemed high risk at any stage of this evaluation, further cardiac testing is indicated.
Pulmonary optimization is typically necessary for geriatric patients, who are more likely to have conditions such as chronic obstructive pulmonary disease or interstitial lung disease.14,20 In patients without severe systemic lung disease, pulmonary optimization involves assessing the functional expiratory volume and diffusing capacity for carbon monoxide. In addition, aggressive modification of risk factors, such as smoking cessation, is strongly recommended.
Additional perioperative conditions are disease-specific and involve evaluation of comorbid illnesses and recognition of absolute contraindications to noncardiac surgery. For instance, an ejection fraction of < 35%, a history of myocardial infarction within 6 months, or active diabetic ketoacidosis are all absolute contraindications to elective surgery. For orthopedic procedures, additional contraindications include symptomatic bacteremia, active joint or local tissue infection, severe malnutrition, uncontrolled metabolic syndrome or chronic disease, untreated immunodeficiency, and active deep venous thrombosis (DVT) or pulmonary embolism.21
Bone Health and Nutrition
In the context of orthopedic surgery, the hallmark of clinically defined optimal bone health is a musculoskeletal system that provides the ability for pain-free functional and occupational tasks with an adequate capacity to withstand the mechanical forces imparted by everyday life. Back pain and arthritis are the fourth- and sixth-most common complaints in primary care, underscoring suboptimal bone health management in developed countries.22
Optimizing bone health through proper nutrition is crucial in the perioperative management of geriatric orthopedic patients. The clinical diagnosis of malnutrition has well-studied associations with worse outcomes after orthopedic surgery, which include increased mortality, hospital length of stay, readmission rates, and health institution spending.23-25 Some studies show that up to 60% of geriatric patients may be malnourished.26
Regarding vitamin and mineral supplements, the general consensus before orthopedic surgery is that vitamins A, C, D, and E, and zinc are predictive in determining postoperative health.27 However, Curtis et al state that therapy should be targeted at correcting relative deficiencies; supraphysiologic concentrations of these vitamins do not appear to be helpful.27 This claim may merit serum studies to rule out deficiencies. Dietitians should be involved in the creation of a patient care plan in the spirit of multidisciplinary orthopedic surgery approaches, which have proven to result in superior patient outcomes.28 Additionally, directive counseling should be provided when necessary.
In patients with adequately managed nutrition, 7 to 10 days of diet optimization is typically sufficient, but patients with malnutrition may require sustained nutritional support for up to 6 weeks; a standardized time for adequate nutrition supplementation has not been identified.25-27 Postoperative management is similar in older patients who are malnourished and those receiving adequate nutrition after orthopedic surgery, which typically involves 3 weeks of a high-protein diet.26
Evaluating Mechanisms of Injury
Assessing the mechanism of injury (MOI) is essential to developing an appropriate and successful orthopedic treatment plan. MOI is typically categorized as low energy, which consists of ground-level falls and other minor trauma, or high energy, which can include motor vehicle crashes or falls from a height.29 Unlike younger patients who typically experience trauma from high-energy MOIs, geriatric patients often sustain fractures from low-energy MOIs. The importance of assessing MOI for the geriatric population is magnified as it provides vital clues that not only help determine the nature of the injury, but also highlight underlying frailty, comorbidities, and potential complications. Weakness or deconditioning related to older age is often not discovered before trauma, which is why assessing the MOI can provide valuable information regarding overall patient health.30

The MOI of trauma also is correlated with factors that influence postoperative recovery and overall prognosis (Figure). Falls comprise more than three-quarters of the MOI in geriatric patients with trauma, and > 90% of these falls are ground-level or other simple falls.30 Falls secondary to an intrinsic disorder, rather than an extrinsic environmental hazard, are more common in geriatric patients.31
These events may be associated with an underlying medical condition, such as osteopenia, osteoporosis, or neuromuscular disorders, such as Parkinson disease.32 They may also be attributed to normal age-related changes, such as decreased visual acuity, reduced reaction time, or mild cognitive impairment.30 An estimated 6% to 35% of geriatric patients who present to the emergency department have some degree of cognitive dysfunction.33 Accordingly, a thorough understanding of the events leading up to injury is vital for the management of older patients. Knowing the specific circumstances of a fall can provide insight into the patient’s gait, balance, and need for further investigations such as cognitive screening or evaluation of home safety. This information can guide decisions regarding preoperative optimization of medications and postoperative rehabilitation interventions.
Frailty and Risk of Fracture
Frailty is a clinical syndrome defined by overall decreased capacity for the body’s adaptive changes to various stressors.34 It is a common condition in geriatric populations due to cumulative degenerative changes and multisystem decline over a lifetime’s worth of disruptions to natural homeostasis.34 In orthopedics, frailty typically refers to musculoskeletal durability and resilience in response to mechanical forces (ie, falls, trauma, and high-acceleration movements). Globally, > 200 million people have osteoporotic frailty, leading to 9 million hip fractures annually.35 More than 30% of people aged ≥ 65 years fall ≥ 1 time per calendar year.36
Assessing frailty in geriatric patients undergoing orthopedic surgery is vital, as it predisposes patients to higher rates of morbidity, mortality, and institutionalization, particularly from falls and resultant fragility fractures.37-39 This is true for a wide range of orthopedic procedures, spanning elective to urgent surgeries and involving the axial and appendicular skeleton.40,41 Given the high rates of fractures, subsequent patient morbidity, and financial burden on the health care system, effective frailty screening is essential.
There are many strategies to assess frailty risk and subsequent fracture risk.42 Questionnaires or online medical calculators serve as easy-to-use tools for assessment of frailty or associated predictors of fragility fractures. Validated assessment tools are provided in Table 1.
Dual-emission X-ray absorptiometry is a well-established way to determine bone density and establish fracture risk. The Fried Frailty Phenotype score and Short Performance Physical Battery test are clinically applicable methods of assessing frailty in older outpatient populations. Although these examinations focus on different aspects of the patient, they have moderate agreeability, are sensitive, and can be readily performed in the clinical setting as demonstrated by a > 90% patient participation rate for both methods.42 Finally, several serum studies can be predictive of frailty, the most readily modifiable of which are vitamin D3, ferritin, albumin, and calcium.43 Although they are more invasive for the patient, serum studies can provide additional modifiable targets for perioperative optimization and contribute to risk stratification.
Risk stratification should take place around 6 weeks before surgery, which should provide adequate time for rectification of preoperative barriers to elective surgical intervention—namely nutritional status. In cases of urgent or emergent procedures (ie, femoral neck fracture with concern for avascular necrosis of the femoral head), this may not be possible but should be conducted nonetheless for patient-specific postoperative rehabilitation and risk reduction.


Postoperative Complication Risks
Postoperative complications affect nearly 15% of geriatric orthopedic patients, highlighting the need for comprehensive preoperative evaluations to assess risk factors.44 Age-related physiological changes, frailty, and comorbidities complicate recovery and management (Table 2).
Wound healing is impaired in older individuals due to suboptimal circulation and decreased oxygenation that is secondary to age-related changes, as well as the increased likelihood of comorbid conditions (eg, diabetes).7 Surgical site infections can be particularly malicious in geriatric patients, with a 4% incidence.45,46 Hospitalization can be prolonged by a mean 2 weeks, which increases the risk of hospital-associated delirium and iatrogenic complications.46 Both the mortality rate and costs associated with hospitalization are higher for older patients who develop surgical site infections compared with patients aged < 65 years, underscoring the importance of vigilant monitoring, early detection, and effective preoperative screening to identify and manage modifiable risk factors.47
Postoperative delirium is another common complication of orthopedic surgery in the geriatric population, increasing morbidity and mortality. The incidence is reported to be as high as 53.3% in the trauma setting and 28.3% in the elective setting, indicating a need to assess patient risk preoperatively.48,49 Several factors contribute to the high incidence of delirium, including advanced age, longer surgical durations, intraoperative hypotension and hypercapnia, pre-existing cognitive dysfunction, and postoperative sleep disorders.50
VTE is another common cause of complications following orthopedic surgery. The development of DVT can lead to subsequent pulmonary embolism, which can be fatal. Orthopedic surgery patients are already at higher risk of DVT and VTE than other surgical patients, with an incidence as high as 40% to 60%, though it is frequently asymptomatic.51,52 Geriatric patients may be more likely to have concurrent comorbidities that increase hypercoagulability.53 Congestive heart failure, chronic kidney disease, and cardiovascular disease are all more common in older individuals and can increase the risk of VTE by 2-fold.53 While anticoagulation is the standard of care to prevent VTE after orthopedic surgery, geriatric patients require more careful monitoring due to the higher incidence of bleeding complications. Additionally, early postoperative mobilization is critical to reduce the risk of DVT without significantly increasing pain or causing other adverse events.54
Respiratory complications are common after orthopedic surgery, particularly atelectasis and bronchospasm, which can result from intraoperative mechanical ventilation.55 While these conditions are typically self-limiting, more severe respiratory issues such as pneumonia are a significant concern because they may lead to the need for mechanical ventilation and admission to the intensive care unit (ICU). The more severe complications have an incidence of about 1% to 2% in orthopedic surgery patients.56 Preventive strategies, such as respiratory physiotherapy and guided breathing exercises, are crucial to minimize perioperative pulmonary complications and promote optimal recovery. Addressing these challenges through early intervention is essential to improve outcomes.
Multidisciplinary Perioperative Care
Multidisciplinary care in orthopedic surgery involves collaborative management of patient care by general practitioners, surgeons, anesthesiologists, dietitians, physical and occupational therapists, inpatient health care practitioners (HCPs), and social services. The goal of this form of care is to provide a longitudinal sequence of health-optimization tactics that prepare a patient for surgery and give them the best chance of postoperative recovery.
Given that many aspects of geriatric health play a role in orthopedic postoperative outcomes, there are many preoperative factors to consider. As previously discussed, preoperative evaluation of geriatric patients should include nutritional and fragility screening to determine surgical candidacy and target modifiable risk factors for risk reduction. This screening can be conducted by primary care practitioners and orthopedic surgeons in an outpatient setting. A multidisciplinary approach benefits patients by decreasing time to surgery.35
Several large studies have demonstrated the positive influence of a multidisciplinary approach on patient-oriented outcomes in orthopedic patients. Incorporation of this style of care in contrast to surgeon-led perioperative optimization leads to fewer floor and ICU admissions, shorter lengths of stay, and decreased mortality rates.35,57 These findings are broadly applicable to a wide range of orthopedic surgeries and even surgeries outside of the musculoskeletal system.58,59 In addition, this strategy has demonstrated reduced in-hospital health care costs due to shorter lengths of stay and fewer ICU admissions. Physical and occupational therapy also have irreplaceable roles in outcomes after orthopedic surgeries. They have independently been shown to decrease pain, increase range of motion, and increase functionality in daily life.60 These aspects of recovery are essential for geriatric well-being.
Screening Tools
The World Health Organization FRAX fracture risk assessment tool (www.fraxplus.org/calculation-tool) was developed to identify patients at high risk of fracture and subsequent complications and to guide clinical decision-making regarding pharmacologic interventions.61 FRAX calculates the 10-year probability of fracture based on demographic factors, such as age and body mass index, clinical measures (eg, femoral neck bone mineral density), and risk factors (eg, prior fragility fractures, substance use history, and prolonged glucocorticoid use).61 The online tool is easy to use, making it a valuable resource for assessing fracture risk and determining appropriate treatment strategies.
The fatigue, resistance, ambulation, illnesses, loss of weight (FRAIL) scale assesses frailty in older adults. The scale classifies patients into 3 categories: robust, prefrail, and frail. The frail category is associated with an increased frequency of hip fracture and an elevated ASA class.62 Additionally, the FRAIL scale has demonstrated value in predicting hospital length of stay and the risk of postoperative complications.62 It also has shown utility in quantifying frailty status, which is traditionally challenging to assess systematically.63
The Mini-Cog is commonly used in geriatric populations to screen for cognitive impairment. Preoperative Mini-Cog screening has been shown to predict the development of postoperative complications.64 Geriatric patients who screened positive for cognitive impairment prior to orthopedic surgery were more likely to develop postoperative delirium, require alternative discharge disposition, and have a longer hospital length of stay.64 Mini-Cog serves as an important preoperative tool for identifying patients who may benefit from closer postoperative monitoring and tailored care.
The Comprehensive Geriatric Assessment (CGA) is a multidimensional evaluation that has been validated for use in geriatric patients undergoing orthopedic surgery.65 The CGA assesses functional status and the ability to perform activities of daily living (ADLs), such as eating, dressing, and ambulating. Poor ADLs are associated with increased risk of falls and cardiopulmonary complications. The CGA allows HCPs to identify patients at higher risk of complications and tailor interventions that optimize functional recovery during the perioperative period.
Nutritional screening is another component of preoperative evaluation in older adults undergoing orthopedic surgery. The Perioperative Nutrition Screen is a preoperative phone assessment of unintentional weight loss in the past 6 months.66 Patients who screen positive are asked to come in for a preoperative visit with a registered dietitian who can further evaluate the nutritional status of the patient.
The Mini Nutritional Assessment Short Form (MNA-SF), Malnutrition Universal Screening Tool, and Nutrition Risk Screening 2002 have all been validated for use in older patients undergoing orthopedic surgery. Among these, the MNA-SF has demonstrated superior utility in predicting hospital readmission and mortality.67 Given the established links between malnutrition and poor surgical outcomes, routine nutritional screening is important for identifying geriatric patients who may require preoperative nutritional interventions.
CONCLUSIONS AND RECOMMENDATIONS
Perioperative management of geriatric patients undergoing orthopedic surgery requires an assessment and strategy focused on risk stratification, patient optimization, and mitigation of potential complications and mortality. Due to the complexity and comprehensive nature of an optimal perioperative plan, creating the plan early is essential to ensure adequate time for patient optimization and care coordination.
Nutrition plays a critical role in the success of surgical procedures, and orthopedics is no exception. Extra care should be taken to preoperatively optimize patient bone health before surgical intervention to enhance recovery and reduce the risk of complications. After an appropriate patient history and clinical picture are gathered, screening tools should be used on a case-by-case basis to further characterize comorbid conditions that may contribute to suboptimal outcomes. Additionally, given the proven association between frailty and fracture risk, frailty serves as a readily quantifiable predictor of patient-oriented outcomes. This should be assessed preoperatively with appropriate risk-stratification tools to determine appropriate postoperative measures to prevent morbidity and mortality.
Orthopedic surgery is increasingly common in geriatric patients, who face higher perioperative risks due to age-related physiological changes, multimorbidity, and frailty. Optimizing preoperative assessment and adopting a multidisciplinary approach—integrating surgeons, anesthesiologists, physical therapists, and dietitians—can improve outcomes, reduce complications, and enhance recovery. The successful use of the tools and strategies outlined in this article by primary care should facilitate access to and recovery from orthopedic surgery in the geriatric population.
More than 40 million surgeries are performed annually in the United States, of which > 18 million are orthopedic, including > 1 million emergency orthopedic surgeries and > 2 million joint replacements.1-4 Notably, > 50% of patients undergoing orthopedic surgery are aged ≥ 65 years, a demographic shift driven by longer life expectancies and an increasing number of older adults remaining physically active for extended periods.5 Osteoarthritis, the most common joint disease, affects 10% of men and 18% of women aged > 60 years, often necessitating an orthopedic joint replacement.6 Perioperative morbidity and mortality are 2.9- to 6.7-times higher in older adults compared with younger adults.7 These risks include infection, venous thromboembolism (VTE), pressure ulcers, reduced mobility, and increased mortality. Due to the high incidence of these complications in older surgical patients, special perioperative protocols and considerations are needed when preparing an older patient for surgery. This review aims to establish concrete considerations and guidelines for perioperative management.
METHODOLOGY
A literature review of PubMed, Google Scholar, and IEEE Xplore identified research on perioperative challenges in geriatric orthopedic surgery. Keywords included geriatrics and orthopedic surgery, perioperative care in geriatric populations, and orthopedic perioperative care. Inclusion criteria were strictly defined to ensure relevance to the geriatric population, with studies focusing on patients aged ≥ 65 years. Exclusion criteria were applied to remove studies that did not involve geriatric populations or orthopedic surgeries or that lacked a clear perioperative focus. Studies were analyzed for design, interventions, and outcomes. Special attention was given to identifying common challenges and trends related to perioperative considerations. We developed a narrative report providing a comprehensive overview of the current understanding of perioperative care for geriatric orthopedic patients to offer practical recommendations for clinicians to use in their practice.
RESULTS
Consistent with the narrative review methodology described, the literature search yielded a broad range of publications addressing perioperative considerations in geriatric orthopedic patients. Articles were screened for relevance to patients aged ≥ 65 years undergoing orthopedic surgery and for applicability to perioperative optimization and postoperative outcomes. Given the heterogeneity in study design, population characteristics, and outcome reporting, findings are presented descriptively rather than being quantitatively pooled. Studies not focused on geriatric populations, orthopedic procedures, or perioperative management were excluded. Key themes included multimorbidity and comorbidity optimization, age-related physiologic changes, frailty assessment and fracture risk stratification, nutritional and bone health management, mechanism of injury considerations, prevention of postoperative complications, and the role of multidisciplinary perioperative care.
Unique Physiological Challenges
The aging process induces a range of physiological changes that can increase morbidity and mortality following surgery. One of the most essential elements to surgical recovery is wound healing, as impairments in this process can lead to adverse events, including infection, cosmetic deformity, and wound dehiscence. The general paradigm of aging involves cell senescence resulting in slower or disorganized functional capacity of these cells.8 While wound healing in older individuals was once thought to be defective, recent research has demonstrated that this process is not absent, but delayed.9
Wound healing is a tightly regulated and evolutionarily conserved process that proceeds through 3 main phases: inflammation, proliferation, and remodeling. Re-epithelialization begins with the migration of epithelial cells from hair follicles, sweat glands, or wound margins (depending on wound depth) and is influenced by oxygen levels, moisture, and growth factors.9 Several characteristics of aged skin contribute to the delayed healing process. Aged skin has fewer hair follicles and eccrine sweat glands, as well as decreased follicle thickness.10 This results in fewer proliferating cells for wound healing and lower amounts of sebum production for skin moisture.11 Furthermore, aged fibroblasts are fewer in number and less effective in synthesizing extracellular matrices, resulting in slower and less tensile wound healing.12 Additionally, microvascular changes associated with aging result in disorganized vasculature, which impairs oxygen delivery to the wound bed and diminishes the influx of proinflammatory cells necessary for effective healing.13 These senescent traits of aged skin contribute to the delayed wound healing process found in geriatric patients.
Compounding these age-related factors is the prevalence of multimorbidity, or coexisting chronic diagnoses, in 55% to 98% of older patients.14 Common comorbidities include peripheral arterial disease, chronic venous insufficiency, type 1 and type 2 diabetes, neoplasms, atherosclerotic disease, and hypertension. Older patients are more likely to be prescribed corticosteroids and chemotherapeutic agents that impair the function of inflammatory cells necessary for wound healing.15,16 Additionally, decreased mobility is more common in geriatric patients, which can increase the risk of wound formation, particularly pressure ulcers.17
Perioperative Considerations
All surgical patients undergo a formal or informal preoperative evaluation to assess their fitness for surgery, with the goal of minimizing both anesthesia-related risks and postoperative complications. A widely used tool in this assessment is the American Society of Anesthesiologists (ASA) physical status classification, which stratifies patients into 6 categories based on their medical history and overall health status.18 Classes range from healthy patients (Class I) to organ donors who are brain-dead (Class VI).
Cardiac optimization is an essential component of preoperative evaluation for older adults due to their higher risk of underlying cardiovascular disease.19 This process involves an in-depth review of the patient’s cardiac history, including the timing and nature of any prior interventions and the recurrence rate. Functional capacity is assessed through metabolic equivalents, where a threshold of > 4 metabolic equivalents (the ability to walk up a flight of stairs) is considered adequate for surgery. Risk is assessed based on the specific surgical procedure, and nonemergent orthopedic procedures are considered intermediate risk. If a patient is deemed high risk at any stage of this evaluation, further cardiac testing is indicated.
Pulmonary optimization is typically necessary for geriatric patients, who are more likely to have conditions such as chronic obstructive pulmonary disease or interstitial lung disease.14,20 In patients without severe systemic lung disease, pulmonary optimization involves assessing the functional expiratory volume and diffusing capacity for carbon monoxide. In addition, aggressive modification of risk factors, such as smoking cessation, is strongly recommended.
Additional perioperative conditions are disease-specific and involve evaluation of comorbid illnesses and recognition of absolute contraindications to noncardiac surgery. For instance, an ejection fraction of < 35%, a history of myocardial infarction within 6 months, or active diabetic ketoacidosis are all absolute contraindications to elective surgery. For orthopedic procedures, additional contraindications include symptomatic bacteremia, active joint or local tissue infection, severe malnutrition, uncontrolled metabolic syndrome or chronic disease, untreated immunodeficiency, and active deep venous thrombosis (DVT) or pulmonary embolism.21
Bone Health and Nutrition
In the context of orthopedic surgery, the hallmark of clinically defined optimal bone health is a musculoskeletal system that provides the ability for pain-free functional and occupational tasks with an adequate capacity to withstand the mechanical forces imparted by everyday life. Back pain and arthritis are the fourth- and sixth-most common complaints in primary care, underscoring suboptimal bone health management in developed countries.22
Optimizing bone health through proper nutrition is crucial in the perioperative management of geriatric orthopedic patients. The clinical diagnosis of malnutrition has well-studied associations with worse outcomes after orthopedic surgery, which include increased mortality, hospital length of stay, readmission rates, and health institution spending.23-25 Some studies show that up to 60% of geriatric patients may be malnourished.26
Regarding vitamin and mineral supplements, the general consensus before orthopedic surgery is that vitamins A, C, D, and E, and zinc are predictive in determining postoperative health.27 However, Curtis et al state that therapy should be targeted at correcting relative deficiencies; supraphysiologic concentrations of these vitamins do not appear to be helpful.27 This claim may merit serum studies to rule out deficiencies. Dietitians should be involved in the creation of a patient care plan in the spirit of multidisciplinary orthopedic surgery approaches, which have proven to result in superior patient outcomes.28 Additionally, directive counseling should be provided when necessary.
In patients with adequately managed nutrition, 7 to 10 days of diet optimization is typically sufficient, but patients with malnutrition may require sustained nutritional support for up to 6 weeks; a standardized time for adequate nutrition supplementation has not been identified.25-27 Postoperative management is similar in older patients who are malnourished and those receiving adequate nutrition after orthopedic surgery, which typically involves 3 weeks of a high-protein diet.26
Evaluating Mechanisms of Injury
Assessing the mechanism of injury (MOI) is essential to developing an appropriate and successful orthopedic treatment plan. MOI is typically categorized as low energy, which consists of ground-level falls and other minor trauma, or high energy, which can include motor vehicle crashes or falls from a height.29 Unlike younger patients who typically experience trauma from high-energy MOIs, geriatric patients often sustain fractures from low-energy MOIs. The importance of assessing MOI for the geriatric population is magnified as it provides vital clues that not only help determine the nature of the injury, but also highlight underlying frailty, comorbidities, and potential complications. Weakness or deconditioning related to older age is often not discovered before trauma, which is why assessing the MOI can provide valuable information regarding overall patient health.30

The MOI of trauma also is correlated with factors that influence postoperative recovery and overall prognosis (Figure). Falls comprise more than three-quarters of the MOI in geriatric patients with trauma, and > 90% of these falls are ground-level or other simple falls.30 Falls secondary to an intrinsic disorder, rather than an extrinsic environmental hazard, are more common in geriatric patients.31
These events may be associated with an underlying medical condition, such as osteopenia, osteoporosis, or neuromuscular disorders, such as Parkinson disease.32 They may also be attributed to normal age-related changes, such as decreased visual acuity, reduced reaction time, or mild cognitive impairment.30 An estimated 6% to 35% of geriatric patients who present to the emergency department have some degree of cognitive dysfunction.33 Accordingly, a thorough understanding of the events leading up to injury is vital for the management of older patients. Knowing the specific circumstances of a fall can provide insight into the patient’s gait, balance, and need for further investigations such as cognitive screening or evaluation of home safety. This information can guide decisions regarding preoperative optimization of medications and postoperative rehabilitation interventions.
Frailty and Risk of Fracture
Frailty is a clinical syndrome defined by overall decreased capacity for the body’s adaptive changes to various stressors.34 It is a common condition in geriatric populations due to cumulative degenerative changes and multisystem decline over a lifetime’s worth of disruptions to natural homeostasis.34 In orthopedics, frailty typically refers to musculoskeletal durability and resilience in response to mechanical forces (ie, falls, trauma, and high-acceleration movements). Globally, > 200 million people have osteoporotic frailty, leading to 9 million hip fractures annually.35 More than 30% of people aged ≥ 65 years fall ≥ 1 time per calendar year.36
Assessing frailty in geriatric patients undergoing orthopedic surgery is vital, as it predisposes patients to higher rates of morbidity, mortality, and institutionalization, particularly from falls and resultant fragility fractures.37-39 This is true for a wide range of orthopedic procedures, spanning elective to urgent surgeries and involving the axial and appendicular skeleton.40,41 Given the high rates of fractures, subsequent patient morbidity, and financial burden on the health care system, effective frailty screening is essential.
There are many strategies to assess frailty risk and subsequent fracture risk.42 Questionnaires or online medical calculators serve as easy-to-use tools for assessment of frailty or associated predictors of fragility fractures. Validated assessment tools are provided in Table 1.
Dual-emission X-ray absorptiometry is a well-established way to determine bone density and establish fracture risk. The Fried Frailty Phenotype score and Short Performance Physical Battery test are clinically applicable methods of assessing frailty in older outpatient populations. Although these examinations focus on different aspects of the patient, they have moderate agreeability, are sensitive, and can be readily performed in the clinical setting as demonstrated by a > 90% patient participation rate for both methods.42 Finally, several serum studies can be predictive of frailty, the most readily modifiable of which are vitamin D3, ferritin, albumin, and calcium.43 Although they are more invasive for the patient, serum studies can provide additional modifiable targets for perioperative optimization and contribute to risk stratification.
Risk stratification should take place around 6 weeks before surgery, which should provide adequate time for rectification of preoperative barriers to elective surgical intervention—namely nutritional status. In cases of urgent or emergent procedures (ie, femoral neck fracture with concern for avascular necrosis of the femoral head), this may not be possible but should be conducted nonetheless for patient-specific postoperative rehabilitation and risk reduction.


Postoperative Complication Risks
Postoperative complications affect nearly 15% of geriatric orthopedic patients, highlighting the need for comprehensive preoperative evaluations to assess risk factors.44 Age-related physiological changes, frailty, and comorbidities complicate recovery and management (Table 2).
Wound healing is impaired in older individuals due to suboptimal circulation and decreased oxygenation that is secondary to age-related changes, as well as the increased likelihood of comorbid conditions (eg, diabetes).7 Surgical site infections can be particularly malicious in geriatric patients, with a 4% incidence.45,46 Hospitalization can be prolonged by a mean 2 weeks, which increases the risk of hospital-associated delirium and iatrogenic complications.46 Both the mortality rate and costs associated with hospitalization are higher for older patients who develop surgical site infections compared with patients aged < 65 years, underscoring the importance of vigilant monitoring, early detection, and effective preoperative screening to identify and manage modifiable risk factors.47
Postoperative delirium is another common complication of orthopedic surgery in the geriatric population, increasing morbidity and mortality. The incidence is reported to be as high as 53.3% in the trauma setting and 28.3% in the elective setting, indicating a need to assess patient risk preoperatively.48,49 Several factors contribute to the high incidence of delirium, including advanced age, longer surgical durations, intraoperative hypotension and hypercapnia, pre-existing cognitive dysfunction, and postoperative sleep disorders.50
VTE is another common cause of complications following orthopedic surgery. The development of DVT can lead to subsequent pulmonary embolism, which can be fatal. Orthopedic surgery patients are already at higher risk of DVT and VTE than other surgical patients, with an incidence as high as 40% to 60%, though it is frequently asymptomatic.51,52 Geriatric patients may be more likely to have concurrent comorbidities that increase hypercoagulability.53 Congestive heart failure, chronic kidney disease, and cardiovascular disease are all more common in older individuals and can increase the risk of VTE by 2-fold.53 While anticoagulation is the standard of care to prevent VTE after orthopedic surgery, geriatric patients require more careful monitoring due to the higher incidence of bleeding complications. Additionally, early postoperative mobilization is critical to reduce the risk of DVT without significantly increasing pain or causing other adverse events.54
Respiratory complications are common after orthopedic surgery, particularly atelectasis and bronchospasm, which can result from intraoperative mechanical ventilation.55 While these conditions are typically self-limiting, more severe respiratory issues such as pneumonia are a significant concern because they may lead to the need for mechanical ventilation and admission to the intensive care unit (ICU). The more severe complications have an incidence of about 1% to 2% in orthopedic surgery patients.56 Preventive strategies, such as respiratory physiotherapy and guided breathing exercises, are crucial to minimize perioperative pulmonary complications and promote optimal recovery. Addressing these challenges through early intervention is essential to improve outcomes.
Multidisciplinary Perioperative Care
Multidisciplinary care in orthopedic surgery involves collaborative management of patient care by general practitioners, surgeons, anesthesiologists, dietitians, physical and occupational therapists, inpatient health care practitioners (HCPs), and social services. The goal of this form of care is to provide a longitudinal sequence of health-optimization tactics that prepare a patient for surgery and give them the best chance of postoperative recovery.
Given that many aspects of geriatric health play a role in orthopedic postoperative outcomes, there are many preoperative factors to consider. As previously discussed, preoperative evaluation of geriatric patients should include nutritional and fragility screening to determine surgical candidacy and target modifiable risk factors for risk reduction. This screening can be conducted by primary care practitioners and orthopedic surgeons in an outpatient setting. A multidisciplinary approach benefits patients by decreasing time to surgery.35
Several large studies have demonstrated the positive influence of a multidisciplinary approach on patient-oriented outcomes in orthopedic patients. Incorporation of this style of care in contrast to surgeon-led perioperative optimization leads to fewer floor and ICU admissions, shorter lengths of stay, and decreased mortality rates.35,57 These findings are broadly applicable to a wide range of orthopedic surgeries and even surgeries outside of the musculoskeletal system.58,59 In addition, this strategy has demonstrated reduced in-hospital health care costs due to shorter lengths of stay and fewer ICU admissions. Physical and occupational therapy also have irreplaceable roles in outcomes after orthopedic surgeries. They have independently been shown to decrease pain, increase range of motion, and increase functionality in daily life.60 These aspects of recovery are essential for geriatric well-being.
Screening Tools
The World Health Organization FRAX fracture risk assessment tool (www.fraxplus.org/calculation-tool) was developed to identify patients at high risk of fracture and subsequent complications and to guide clinical decision-making regarding pharmacologic interventions.61 FRAX calculates the 10-year probability of fracture based on demographic factors, such as age and body mass index, clinical measures (eg, femoral neck bone mineral density), and risk factors (eg, prior fragility fractures, substance use history, and prolonged glucocorticoid use).61 The online tool is easy to use, making it a valuable resource for assessing fracture risk and determining appropriate treatment strategies.
The fatigue, resistance, ambulation, illnesses, loss of weight (FRAIL) scale assesses frailty in older adults. The scale classifies patients into 3 categories: robust, prefrail, and frail. The frail category is associated with an increased frequency of hip fracture and an elevated ASA class.62 Additionally, the FRAIL scale has demonstrated value in predicting hospital length of stay and the risk of postoperative complications.62 It also has shown utility in quantifying frailty status, which is traditionally challenging to assess systematically.63
The Mini-Cog is commonly used in geriatric populations to screen for cognitive impairment. Preoperative Mini-Cog screening has been shown to predict the development of postoperative complications.64 Geriatric patients who screened positive for cognitive impairment prior to orthopedic surgery were more likely to develop postoperative delirium, require alternative discharge disposition, and have a longer hospital length of stay.64 Mini-Cog serves as an important preoperative tool for identifying patients who may benefit from closer postoperative monitoring and tailored care.
The Comprehensive Geriatric Assessment (CGA) is a multidimensional evaluation that has been validated for use in geriatric patients undergoing orthopedic surgery.65 The CGA assesses functional status and the ability to perform activities of daily living (ADLs), such as eating, dressing, and ambulating. Poor ADLs are associated with increased risk of falls and cardiopulmonary complications. The CGA allows HCPs to identify patients at higher risk of complications and tailor interventions that optimize functional recovery during the perioperative period.
Nutritional screening is another component of preoperative evaluation in older adults undergoing orthopedic surgery. The Perioperative Nutrition Screen is a preoperative phone assessment of unintentional weight loss in the past 6 months.66 Patients who screen positive are asked to come in for a preoperative visit with a registered dietitian who can further evaluate the nutritional status of the patient.
The Mini Nutritional Assessment Short Form (MNA-SF), Malnutrition Universal Screening Tool, and Nutrition Risk Screening 2002 have all been validated for use in older patients undergoing orthopedic surgery. Among these, the MNA-SF has demonstrated superior utility in predicting hospital readmission and mortality.67 Given the established links between malnutrition and poor surgical outcomes, routine nutritional screening is important for identifying geriatric patients who may require preoperative nutritional interventions.
CONCLUSIONS AND RECOMMENDATIONS
Perioperative management of geriatric patients undergoing orthopedic surgery requires an assessment and strategy focused on risk stratification, patient optimization, and mitigation of potential complications and mortality. Due to the complexity and comprehensive nature of an optimal perioperative plan, creating the plan early is essential to ensure adequate time for patient optimization and care coordination.
Nutrition plays a critical role in the success of surgical procedures, and orthopedics is no exception. Extra care should be taken to preoperatively optimize patient bone health before surgical intervention to enhance recovery and reduce the risk of complications. After an appropriate patient history and clinical picture are gathered, screening tools should be used on a case-by-case basis to further characterize comorbid conditions that may contribute to suboptimal outcomes. Additionally, given the proven association between frailty and fracture risk, frailty serves as a readily quantifiable predictor of patient-oriented outcomes. This should be assessed preoperatively with appropriate risk-stratification tools to determine appropriate postoperative measures to prevent morbidity and mortality.
Orthopedic surgery is increasingly common in geriatric patients, who face higher perioperative risks due to age-related physiological changes, multimorbidity, and frailty. Optimizing preoperative assessment and adopting a multidisciplinary approach—integrating surgeons, anesthesiologists, physical therapists, and dietitians—can improve outcomes, reduce complications, and enhance recovery. The successful use of the tools and strategies outlined in this article by primary care should facilitate access to and recovery from orthopedic surgery in the geriatric population.
Dobson GP. Trauma of major surgery: a global problem that is not going away. Int J Surg. 2020;81:47-54. doi:10.1016/j.ijsu.2020.07.017
United States (US) orthopedic procedures count by segments and forecast to 2030. GlobalData. February 17, 2023. Accessed April 29, 2026. https://www.globaldata.com/store/report/usa-orthopedic-procedures-analysis/
Jarman MP, Weaver MJ, Haider AH, Salim A, Harris MB. The national burden of orthopedic injury: cross-sectional estimates for trauma system planning and optimization. J Surg Res. 2020;249:197-204. doi:10.1016/j.jss.2019.12.023
Hegde V, Stambough JB, Levine BR, et al. Highlights of the 2022 American Joint Replacement Registry Annual Report. Arthroplast Today. 2023;21:101137. doi:10.1016/j.artd.2023.101137
Nakamura K, Ogata T. Locomotive syndrome: definition and management. Clin Rev Bone Miner Metab. 2016;14:56-67. doi:10.1007/s12018-016-9208-2
Glyn-Jones S, Palmer AJR, Agricola R, et al. Osteoarthritis. Lancet. 2015;386:376-387. doi:10.1016/S0140-6736(14)60802-3
Hughes S, Leary A, Zweizig S, Cain J. Surgery in elderly people: preoperative, operative and postoperative care to assist healing. Best Pract Res Clin Obstet Gynaecol. 2013;27:753-765. doi:10.1016/j.bpobgyn.2013.02.006
Regulski MJ. Cellular senescence: what, why, and how. Wounds. 2017;29:168-174.
Kremer M, Burkemper N. Aging skin and wound healing. Clin Geriatr Med. 2024;40:1-10. doi:10.1016/j.cger.2023.06.001
Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol. 1986;15:571-585. doi:10.1016/S0190-9622(86)70208-9
Van Neste D, Tobin DJ. Hair cycle and hair pigmentation: dynamic interactions and changes associated with aging. Micron. 2004;35:193-200. doi:10.1016/j.micron.2003.11.006
Salzer MC, Lafzi A, Berenguer-Llergo A, et al. Identity noise and adipogenic traits characterize dermal fibroblast aging. Cell. 2018;175:1575-1590.e22. doi:10.1016/j.cell.2018.10.012
Jin K. A microcirculatory theory of aging. Aging Dis. 2019;10:676-683. doi:10.14336/AD.2019.0315
Marengoni A, Angleman S, Melis R, et al. Aging with multimorbidity: a systematic review of the literature. Ageing Res Rev. 2011;10:430-439. doi:10.1016/j.arr.2011.03.003
Waljee AK, Rogers MAM, Lin P, et al. Short term use of oral corticosteroids and related harms among adults in the United States: population based cohort study. BMJ. 2017;357:j1415. doi:10.1136/bmj.j1415
Given B, Given CW. Older adults and cancer treatment. Cancer. 2008;113:3505-3511. doi:10.1002/cncr.23939
Ferrucci L, Cooper R, Shardell M, Simonsick EM, Schrack JA, Kuh D. Age-related change in mobility: perspectives from life course epidemiology and geroscience. J Gerontol A Biol Sci Med Sci. 2016;71:1184-1194. doi:10.1093/gerona/glw043
Mayhew D, Mendonca V, Murthy BVS. A review of ASA physical status - historical perspectives and modern developments. Anaesthesia. 2019;74:373-379. doi:10.1111/anae.14569
Eagle KA, Berger PB, Calkins H, et al. ACC/AHA guideline update for perioperative cardiovascular evaluation for noncardiac surgery—executive summary a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1996 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation. 2002;105:1257-1267. doi:10.1161/circ.105.10.1257
Bapoje SR, Whitaker JF, Schulz T, Chu ES, Albert RK. Preoperative evaluation of the patient with pulmonary disease. Chest. 2007;132:1637-1645. doi:10.1378/chest.07-0347
Choe H, Indelli PF, Ricciardi B, et al. What are the absolute contraindications for elective total knee or hip arthroplasty? J Arthroplasty. 2025;40(2 suppl 1):S45-S47. doi:10.1016/j.arth.2024.10.041
Finley CR, Chan DS, Garrison S, et al. What are the most common conditions in primary care? Systematic review. Can Fam Physician. 2018;64:832-840.
Vaid S, Bell T, Grim R, Ahuja V. Predicting risk of death in general surgery patients on the basis of preoperative variables using American College of Surgeons National Surgical Quality Improvement Program data. Perm J. 2012;16:10-17. doi:10.7812/TPP/12-019
Correia MTD, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22:235-239. doi:10.1016/S0261-5614(02)00215-7
Friedman J, Lussiez A, Sullivan J, Wang S, Englesbe M. Implications of sarcopenia in major surgery. Nutr Clin Pract. 2015;30:175-179. doi:10.1177/0884533615569888
Hirsch KR, Wolfe RR, Ferrando AA. Pre- and post-surgical nutrition for preservation of muscle mass, strength, and functionality following orthopedic surgery. Nutrients. 2021;13:1675. doi:10.3390/nu13051675
Curtis W, Choi T, Ahmad A, Shultz C. Perioperative nutritional considerations in orthopaedic surgery: a review of the literature. West J Orthop. 2023;12:1. https://digitalrepository.unm.edu/wjo/vol12/iss1/1
Wischmeyer PE, Carli F, Evans DC, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on nutrition screening and therapy within a surgical enhanced recovery pathway. Anesth Analg. 2018;126:1883-1895. doi:10.1213/ANE.0000000000002743
Mun F, Ringenbach K, Baer B, et al. Factors influencing geriatric orthopaedic trauma mortality. Injury. 2022;53:919-924. doi:10.1016/j.injury.2022.01.005
Bonne S, Schuerer DJE. Trauma in the older adult: epidemiology and evolving geriatric trauma principles. Clin Geriatr Med. 2013;29:137-150. doi:10.1016/j.cger.2012.10.008
Montero-Odasso MM. Falls as a geriatric syndrome: mechanisms and risk identification. In: Duque G, Kiel DP, eds. Osteoporosis in Older Persons: Advances in Pathophysiology and Therapeutic Approaches. 2nd ed. Springer International Publishing; 2016:171-186. doi:10.1007/978-3-319-25976-5_10
Lach HW, Reed AT, Arfken CL, et al. Falls in the elderly: reliability of a classification system. J Am Geriatr Soc. 1991;39:197-202. doi:10.1111/j.1532-5415.1991.tb01626.x
Carpenter CR, DesPain B, Keeling TN, Shah M, Rothenberger M. The six-item screener and AD8 for the detection of cognitive impairment in geriatric emergency department patients. Ann Emerg Med. 2011;57:653-661. doi:10.1016/j.annemergmed.2010.06.560
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381:752-762. doi:10.1016/S0140-6736(12)62167-9
Patel JN, Klein DS, Sreekumar S, Liporace FA, Yoon RS. Outcomes in multidisciplinary team-based approach in geriatric hip fracture care: a systematic review. J Am Acad Orthop Surg. 2020;28:128-133. doi:10.5435/JAAOS-D-18-00425
Amador LF, Loera JA. Preventing postoperative falls in the older adult. J Am Coll Surg. 2007;204:447-453. doi:10.1016/j.jamcollsurg.2006.12.010
Tembo MC, Holloway-Kew KL, Mohebbi M, et al. The association between a fracture risk tool and frailty: Geelong Osteoporosis Study. BMC Geriatr. 2020;20:196. doi:10.1186/s12877-020-01595-8
Demiris¸ B, Basat S, Kurt F, Aksakal B, Basat O. Evaluation of the relationship between frailty and fracture risk using Fracture Risk Assessment Tool in patients 65 years and over. South Clin Istanb Eurasia. 2023;34:42-48. doi:10.14744/scie.2022.66564
Partridge JSL, Harari D, Dhesi JK. Frailty in the older surgical patient: a review. Age Ageing. 2012;41:142-147. doi:10.1093/ageing/afr182
Mamtora PH, Fortier MA, Barnett SR, Schmid LN, Kain ZN. Peri-operative management of frailty in the orthopedic patient. J Orthop. 2020;22:304-307. doi:10.1016/j.jor.2020.05.024
Leven DM, Lee NJ, Kim JS, et al. Frailty is predictive of adverse postoperative events in patients undergoing lumbar fusion. Global Spine J. 2017;7:529-535. doi:10.1177/2192568217700099
Pritchard JM, Kennedy CC, Karampatos S, et al. Measuring frailty in clinical practice: a comparison of physical frailty assessment methods in a geriatric out-patient clinic. BMC Geriatr. 2017;17:264. doi:10.1186/s12877-017-0623-0
Kumar A, Dhar M, Agarwal M, Mukherjee A, Saxena V. Predictors of frailty in the elderly population: a cross-sectional study at a tertiary care center. Cureus. 2022;14:e30557. doi:10.7759/cureus.30557
Scarano KA, Philp FH, Westrick ER, Altman GT, Altman DT. Evaluating postoperative complications and outcomes of orthopedic fracture repair in nonagenarian patients. Geriatr Orthop Surg Rehabil. 2018;9:2151459318758106. doi:10.1177/2151459318758106
Liang Z, Rong K, Gu W, et al. Surgical site infection following elective orthopaedic surgeries in geriatric patients: incidence and associated risk factors. Int Wound J. 2019;16:773-780. doi:10.1111/iwj.13096
Ren M, Liang W, Wu Z, Zhao H, Wang J. Risk factors of surgical site infection in geriatric orthopedic surgery: a retrospective multicenter cohort study. Geriatr Gerontol Int. 2019;19:213-217. doi:10.1111/ggi.13590
Kaye KS, Schmader KE, Sawyer R. Surgical site infection in the elderly population. Clin Infect Dis. 2004;39:1835-1841. doi:10.1086/425744
Bruce AJ, Ritchie CW, Blizard R, Lai R, Raven P. The incidence of delirium associated with orthopedic surgery: a meta-analytic review. Int Psychogeriatr. 2007;19:197-214. doi:10.1017/S104161020600425X
Williams-Russo P, Urquhart BL, Sharrock NE, Charlson ME. Post-operative delirium: predictors and prognosis in elderly orthopedic patients. J Am Geriatr Soc. 1992;40:759-767. doi:10.1111/j.1532-5415.1992.tb01846.x
Wang J, Li Z, Yu Y, Li B, Shao G, Wang Q. Risk factors contributing to postoperative delirium in geriatric patients postorthopedic surgery. Asia Pac Psychiatry. 2015;7:375-382. doi:10.1111/appy.12193
Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126:338S-400S. doi:10.1378/chest.126.3_suppl.338S
Kahn SR, Shivakumar S. What’s new in VTE risk and prevention in orthopedic surgery. Res Pract Thromb Haemost. 2020;4:366-376. doi:10.1002/rth2.12323
Uzel K, Azboy I·, Parvizi J. Venous thromboembolism in orthopedic surgery: global guidelines. Acta Orthop Traumatol Turc. 2023;57:192-203. doi:10.5152/j.aott.2023.23074
Peck M, Holthaus A, Kingsbury K, Salsberry MG, Duggirala V. Mobility in acute care for geriatric patients with orthopedic conditions: a review of recent literature. Curr Geri Rep. 2020;9:300-310. doi:10.1007/s13670-020-00347-1
Leme LEG, Sitta MC, Toledo M, Henriques SS. Orthopedic surgery among the elderly: clinical characteristics. Rev Bras Ortop. 2015;46:238-246. doi:10.1016/S2255-4971(15)30189-0
Malcolm TL, Knezevic NN, Zouki CC, Tharian AR. Pulmonary complications after hip and knee arthroplasty in the United States, 2004-2014. Anesth Analg. 2020;130:917-924. doi:10.1213/ANE.0000000000004265
Kamal T, Conway RM, Littlejohn I, Ricketts D. The role of a multidisciplinary pre-assessment clinic in reducing mortality after complex orthopaedic surgery. Ann R Coll Surg Engl. 2011;93:149-151. doi:10.1308/003588411X561026
Davis MJ, Luu BC, Raj S, Abu-Ghname A, Buchanan EP. Multidisciplinary care in surgery: Are team-based interventions cost-effective? Surgeon. 2021;19:49-60. doi:10.1016/j.surge.2020.02.005
Frassanito L, Vergari A, Nestorini R, et al. Enhanced recovery after surgery (ERAS) in hip and knee replacement surgery: description of a multidisciplinary program to improve management of the patients undergoing major orthopedic surgery. Musculoskelet Surg. 2020;104:87-92. doi:10.1007/s12306-019-00603-4
Reddy RS, Alahmari KA, Alshahrani MS, et al. Exploring the impact of physiotherapy on health outcomes in older adults with chronic diseases: a cross-sectional analysis. Front Public Health. 2024;12:1415882. doi:10.3389/fpubh.2024.1415882
Watts NB. The Fracture Risk Assessment Tool (FRAX®): applications in clinical practice. J Womens Health (Larchmt). 2011;20:525-531. doi:10.1089/jwh.2010.2294
Gleason LJ, Benton EA, Alvarez-Nebreda ML, Weaver MJ, Harris MB, Javedan H. FRAIL questionnaire screening tool and short-term outcomes in geriatric fracture patients. J Am Med Dir Assoc. 2017;18:1082-1086. doi:10.1016/j.jamda.2017.07.005
Kojima G. Frailty defined by FRAIL scale as a predictor of mortality: a systematic review and meta-analysis. J Am Med Dir Assoc. 2018;19:480-483. doi:10.1016/j.jamda.2018.04.006
Culley DJ, Flaherty D, Fahey MC, et al. Poor performance on a preoperative cognitive screening test predicts postoperative complications in older orthopedic surgical patients. Anesthesiology. 2017;127:765-774. doi:10.1097/ALN.0000000000001859
Kong C, Zhang Y, Wang C, et al. Comprehensive geriatric assessment for older orthopedic patients and analysis of risk factors for postoperative complications. BMC Geriatr. 2022;22:644. doi:10.1186/s12877-022-03328-5
Williams DGA, Wischmeyer PE. Perioperative nutrition care of orthopedic surgery patient. Tech Orthop. 2020;35:15-18. doi:10.1097/BTO.0000000000000412
Koren-Hakim T, Weiss A, Hershkovitz A, et al. Comparing the adequacy of the MNA-SF, NRS-2002 and MUST nutritional tools in assessing malnutrition in hip fracture operated elderly patients. Clin Nutr. 2016;35:1053-1058. doi:10.1016/j.clnu.2015.07.014
Dobson GP. Trauma of major surgery: a global problem that is not going away. Int J Surg. 2020;81:47-54. doi:10.1016/j.ijsu.2020.07.017
United States (US) orthopedic procedures count by segments and forecast to 2030. GlobalData. February 17, 2023. Accessed April 29, 2026. https://www.globaldata.com/store/report/usa-orthopedic-procedures-analysis/
Jarman MP, Weaver MJ, Haider AH, Salim A, Harris MB. The national burden of orthopedic injury: cross-sectional estimates for trauma system planning and optimization. J Surg Res. 2020;249:197-204. doi:10.1016/j.jss.2019.12.023
Hegde V, Stambough JB, Levine BR, et al. Highlights of the 2022 American Joint Replacement Registry Annual Report. Arthroplast Today. 2023;21:101137. doi:10.1016/j.artd.2023.101137
Nakamura K, Ogata T. Locomotive syndrome: definition and management. Clin Rev Bone Miner Metab. 2016;14:56-67. doi:10.1007/s12018-016-9208-2
Glyn-Jones S, Palmer AJR, Agricola R, et al. Osteoarthritis. Lancet. 2015;386:376-387. doi:10.1016/S0140-6736(14)60802-3
Hughes S, Leary A, Zweizig S, Cain J. Surgery in elderly people: preoperative, operative and postoperative care to assist healing. Best Pract Res Clin Obstet Gynaecol. 2013;27:753-765. doi:10.1016/j.bpobgyn.2013.02.006
Regulski MJ. Cellular senescence: what, why, and how. Wounds. 2017;29:168-174.
Kremer M, Burkemper N. Aging skin and wound healing. Clin Geriatr Med. 2024;40:1-10. doi:10.1016/j.cger.2023.06.001
Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol. 1986;15:571-585. doi:10.1016/S0190-9622(86)70208-9
Van Neste D, Tobin DJ. Hair cycle and hair pigmentation: dynamic interactions and changes associated with aging. Micron. 2004;35:193-200. doi:10.1016/j.micron.2003.11.006
Salzer MC, Lafzi A, Berenguer-Llergo A, et al. Identity noise and adipogenic traits characterize dermal fibroblast aging. Cell. 2018;175:1575-1590.e22. doi:10.1016/j.cell.2018.10.012
Jin K. A microcirculatory theory of aging. Aging Dis. 2019;10:676-683. doi:10.14336/AD.2019.0315
Marengoni A, Angleman S, Melis R, et al. Aging with multimorbidity: a systematic review of the literature. Ageing Res Rev. 2011;10:430-439. doi:10.1016/j.arr.2011.03.003
Waljee AK, Rogers MAM, Lin P, et al. Short term use of oral corticosteroids and related harms among adults in the United States: population based cohort study. BMJ. 2017;357:j1415. doi:10.1136/bmj.j1415
Given B, Given CW. Older adults and cancer treatment. Cancer. 2008;113:3505-3511. doi:10.1002/cncr.23939
Ferrucci L, Cooper R, Shardell M, Simonsick EM, Schrack JA, Kuh D. Age-related change in mobility: perspectives from life course epidemiology and geroscience. J Gerontol A Biol Sci Med Sci. 2016;71:1184-1194. doi:10.1093/gerona/glw043
Mayhew D, Mendonca V, Murthy BVS. A review of ASA physical status - historical perspectives and modern developments. Anaesthesia. 2019;74:373-379. doi:10.1111/anae.14569
Eagle KA, Berger PB, Calkins H, et al. ACC/AHA guideline update for perioperative cardiovascular evaluation for noncardiac surgery—executive summary a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1996 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation. 2002;105:1257-1267. doi:10.1161/circ.105.10.1257
Bapoje SR, Whitaker JF, Schulz T, Chu ES, Albert RK. Preoperative evaluation of the patient with pulmonary disease. Chest. 2007;132:1637-1645. doi:10.1378/chest.07-0347
Choe H, Indelli PF, Ricciardi B, et al. What are the absolute contraindications for elective total knee or hip arthroplasty? J Arthroplasty. 2025;40(2 suppl 1):S45-S47. doi:10.1016/j.arth.2024.10.041
Finley CR, Chan DS, Garrison S, et al. What are the most common conditions in primary care? Systematic review. Can Fam Physician. 2018;64:832-840.
Vaid S, Bell T, Grim R, Ahuja V. Predicting risk of death in general surgery patients on the basis of preoperative variables using American College of Surgeons National Surgical Quality Improvement Program data. Perm J. 2012;16:10-17. doi:10.7812/TPP/12-019
Correia MTD, Waitzberg DL. The impact of malnutrition on morbidity, mortality, length of hospital stay and costs evaluated through a multivariate model analysis. Clin Nutr. 2003;22:235-239. doi:10.1016/S0261-5614(02)00215-7
Friedman J, Lussiez A, Sullivan J, Wang S, Englesbe M. Implications of sarcopenia in major surgery. Nutr Clin Pract. 2015;30:175-179. doi:10.1177/0884533615569888
Hirsch KR, Wolfe RR, Ferrando AA. Pre- and post-surgical nutrition for preservation of muscle mass, strength, and functionality following orthopedic surgery. Nutrients. 2021;13:1675. doi:10.3390/nu13051675
Curtis W, Choi T, Ahmad A, Shultz C. Perioperative nutritional considerations in orthopaedic surgery: a review of the literature. West J Orthop. 2023;12:1. https://digitalrepository.unm.edu/wjo/vol12/iss1/1
Wischmeyer PE, Carli F, Evans DC, et al. American Society for Enhanced Recovery and Perioperative Quality Initiative joint consensus statement on nutrition screening and therapy within a surgical enhanced recovery pathway. Anesth Analg. 2018;126:1883-1895. doi:10.1213/ANE.0000000000002743
Mun F, Ringenbach K, Baer B, et al. Factors influencing geriatric orthopaedic trauma mortality. Injury. 2022;53:919-924. doi:10.1016/j.injury.2022.01.005
Bonne S, Schuerer DJE. Trauma in the older adult: epidemiology and evolving geriatric trauma principles. Clin Geriatr Med. 2013;29:137-150. doi:10.1016/j.cger.2012.10.008
Montero-Odasso MM. Falls as a geriatric syndrome: mechanisms and risk identification. In: Duque G, Kiel DP, eds. Osteoporosis in Older Persons: Advances in Pathophysiology and Therapeutic Approaches. 2nd ed. Springer International Publishing; 2016:171-186. doi:10.1007/978-3-319-25976-5_10
Lach HW, Reed AT, Arfken CL, et al. Falls in the elderly: reliability of a classification system. J Am Geriatr Soc. 1991;39:197-202. doi:10.1111/j.1532-5415.1991.tb01626.x
Carpenter CR, DesPain B, Keeling TN, Shah M, Rothenberger M. The six-item screener and AD8 for the detection of cognitive impairment in geriatric emergency department patients. Ann Emerg Med. 2011;57:653-661. doi:10.1016/j.annemergmed.2010.06.560
Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381:752-762. doi:10.1016/S0140-6736(12)62167-9
Patel JN, Klein DS, Sreekumar S, Liporace FA, Yoon RS. Outcomes in multidisciplinary team-based approach in geriatric hip fracture care: a systematic review. J Am Acad Orthop Surg. 2020;28:128-133. doi:10.5435/JAAOS-D-18-00425
Amador LF, Loera JA. Preventing postoperative falls in the older adult. J Am Coll Surg. 2007;204:447-453. doi:10.1016/j.jamcollsurg.2006.12.010
Tembo MC, Holloway-Kew KL, Mohebbi M, et al. The association between a fracture risk tool and frailty: Geelong Osteoporosis Study. BMC Geriatr. 2020;20:196. doi:10.1186/s12877-020-01595-8
Demiris¸ B, Basat S, Kurt F, Aksakal B, Basat O. Evaluation of the relationship between frailty and fracture risk using Fracture Risk Assessment Tool in patients 65 years and over. South Clin Istanb Eurasia. 2023;34:42-48. doi:10.14744/scie.2022.66564
Partridge JSL, Harari D, Dhesi JK. Frailty in the older surgical patient: a review. Age Ageing. 2012;41:142-147. doi:10.1093/ageing/afr182
Mamtora PH, Fortier MA, Barnett SR, Schmid LN, Kain ZN. Peri-operative management of frailty in the orthopedic patient. J Orthop. 2020;22:304-307. doi:10.1016/j.jor.2020.05.024
Leven DM, Lee NJ, Kim JS, et al. Frailty is predictive of adverse postoperative events in patients undergoing lumbar fusion. Global Spine J. 2017;7:529-535. doi:10.1177/2192568217700099
Pritchard JM, Kennedy CC, Karampatos S, et al. Measuring frailty in clinical practice: a comparison of physical frailty assessment methods in a geriatric out-patient clinic. BMC Geriatr. 2017;17:264. doi:10.1186/s12877-017-0623-0
Kumar A, Dhar M, Agarwal M, Mukherjee A, Saxena V. Predictors of frailty in the elderly population: a cross-sectional study at a tertiary care center. Cureus. 2022;14:e30557. doi:10.7759/cureus.30557
Scarano KA, Philp FH, Westrick ER, Altman GT, Altman DT. Evaluating postoperative complications and outcomes of orthopedic fracture repair in nonagenarian patients. Geriatr Orthop Surg Rehabil. 2018;9:2151459318758106. doi:10.1177/2151459318758106
Liang Z, Rong K, Gu W, et al. Surgical site infection following elective orthopaedic surgeries in geriatric patients: incidence and associated risk factors. Int Wound J. 2019;16:773-780. doi:10.1111/iwj.13096
Ren M, Liang W, Wu Z, Zhao H, Wang J. Risk factors of surgical site infection in geriatric orthopedic surgery: a retrospective multicenter cohort study. Geriatr Gerontol Int. 2019;19:213-217. doi:10.1111/ggi.13590
Kaye KS, Schmader KE, Sawyer R. Surgical site infection in the elderly population. Clin Infect Dis. 2004;39:1835-1841. doi:10.1086/425744
Bruce AJ, Ritchie CW, Blizard R, Lai R, Raven P. The incidence of delirium associated with orthopedic surgery: a meta-analytic review. Int Psychogeriatr. 2007;19:197-214. doi:10.1017/S104161020600425X
Williams-Russo P, Urquhart BL, Sharrock NE, Charlson ME. Post-operative delirium: predictors and prognosis in elderly orthopedic patients. J Am Geriatr Soc. 1992;40:759-767. doi:10.1111/j.1532-5415.1992.tb01846.x
Wang J, Li Z, Yu Y, Li B, Shao G, Wang Q. Risk factors contributing to postoperative delirium in geriatric patients postorthopedic surgery. Asia Pac Psychiatry. 2015;7:375-382. doi:10.1111/appy.12193
Geerts WH, Pineo GF, Heit JA, et al. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126:338S-400S. doi:10.1378/chest.126.3_suppl.338S
Kahn SR, Shivakumar S. What’s new in VTE risk and prevention in orthopedic surgery. Res Pract Thromb Haemost. 2020;4:366-376. doi:10.1002/rth2.12323
Uzel K, Azboy I·, Parvizi J. Venous thromboembolism in orthopedic surgery: global guidelines. Acta Orthop Traumatol Turc. 2023;57:192-203. doi:10.5152/j.aott.2023.23074
Peck M, Holthaus A, Kingsbury K, Salsberry MG, Duggirala V. Mobility in acute care for geriatric patients with orthopedic conditions: a review of recent literature. Curr Geri Rep. 2020;9:300-310. doi:10.1007/s13670-020-00347-1
Leme LEG, Sitta MC, Toledo M, Henriques SS. Orthopedic surgery among the elderly: clinical characteristics. Rev Bras Ortop. 2015;46:238-246. doi:10.1016/S2255-4971(15)30189-0
Malcolm TL, Knezevic NN, Zouki CC, Tharian AR. Pulmonary complications after hip and knee arthroplasty in the United States, 2004-2014. Anesth Analg. 2020;130:917-924. doi:10.1213/ANE.0000000000004265
Kamal T, Conway RM, Littlejohn I, Ricketts D. The role of a multidisciplinary pre-assessment clinic in reducing mortality after complex orthopaedic surgery. Ann R Coll Surg Engl. 2011;93:149-151. doi:10.1308/003588411X561026
Davis MJ, Luu BC, Raj S, Abu-Ghname A, Buchanan EP. Multidisciplinary care in surgery: Are team-based interventions cost-effective? Surgeon. 2021;19:49-60. doi:10.1016/j.surge.2020.02.005
Frassanito L, Vergari A, Nestorini R, et al. Enhanced recovery after surgery (ERAS) in hip and knee replacement surgery: description of a multidisciplinary program to improve management of the patients undergoing major orthopedic surgery. Musculoskelet Surg. 2020;104:87-92. doi:10.1007/s12306-019-00603-4
Reddy RS, Alahmari KA, Alshahrani MS, et al. Exploring the impact of physiotherapy on health outcomes in older adults with chronic diseases: a cross-sectional analysis. Front Public Health. 2024;12:1415882. doi:10.3389/fpubh.2024.1415882
Watts NB. The Fracture Risk Assessment Tool (FRAX®): applications in clinical practice. J Womens Health (Larchmt). 2011;20:525-531. doi:10.1089/jwh.2010.2294
Gleason LJ, Benton EA, Alvarez-Nebreda ML, Weaver MJ, Harris MB, Javedan H. FRAIL questionnaire screening tool and short-term outcomes in geriatric fracture patients. J Am Med Dir Assoc. 2017;18:1082-1086. doi:10.1016/j.jamda.2017.07.005
Kojima G. Frailty defined by FRAIL scale as a predictor of mortality: a systematic review and meta-analysis. J Am Med Dir Assoc. 2018;19:480-483. doi:10.1016/j.jamda.2018.04.006
Culley DJ, Flaherty D, Fahey MC, et al. Poor performance on a preoperative cognitive screening test predicts postoperative complications in older orthopedic surgical patients. Anesthesiology. 2017;127:765-774. doi:10.1097/ALN.0000000000001859
Kong C, Zhang Y, Wang C, et al. Comprehensive geriatric assessment for older orthopedic patients and analysis of risk factors for postoperative complications. BMC Geriatr. 2022;22:644. doi:10.1186/s12877-022-03328-5
Williams DGA, Wischmeyer PE. Perioperative nutrition care of orthopedic surgery patient. Tech Orthop. 2020;35:15-18. doi:10.1097/BTO.0000000000000412
Koren-Hakim T, Weiss A, Hershkovitz A, et al. Comparing the adequacy of the MNA-SF, NRS-2002 and MUST nutritional tools in assessing malnutrition in hip fracture operated elderly patients. Clin Nutr. 2016;35:1053-1058. doi:10.1016/j.clnu.2015.07.014
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
Perioperative Considerations for Orthopedic Surgery in a Geriatric Population
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)
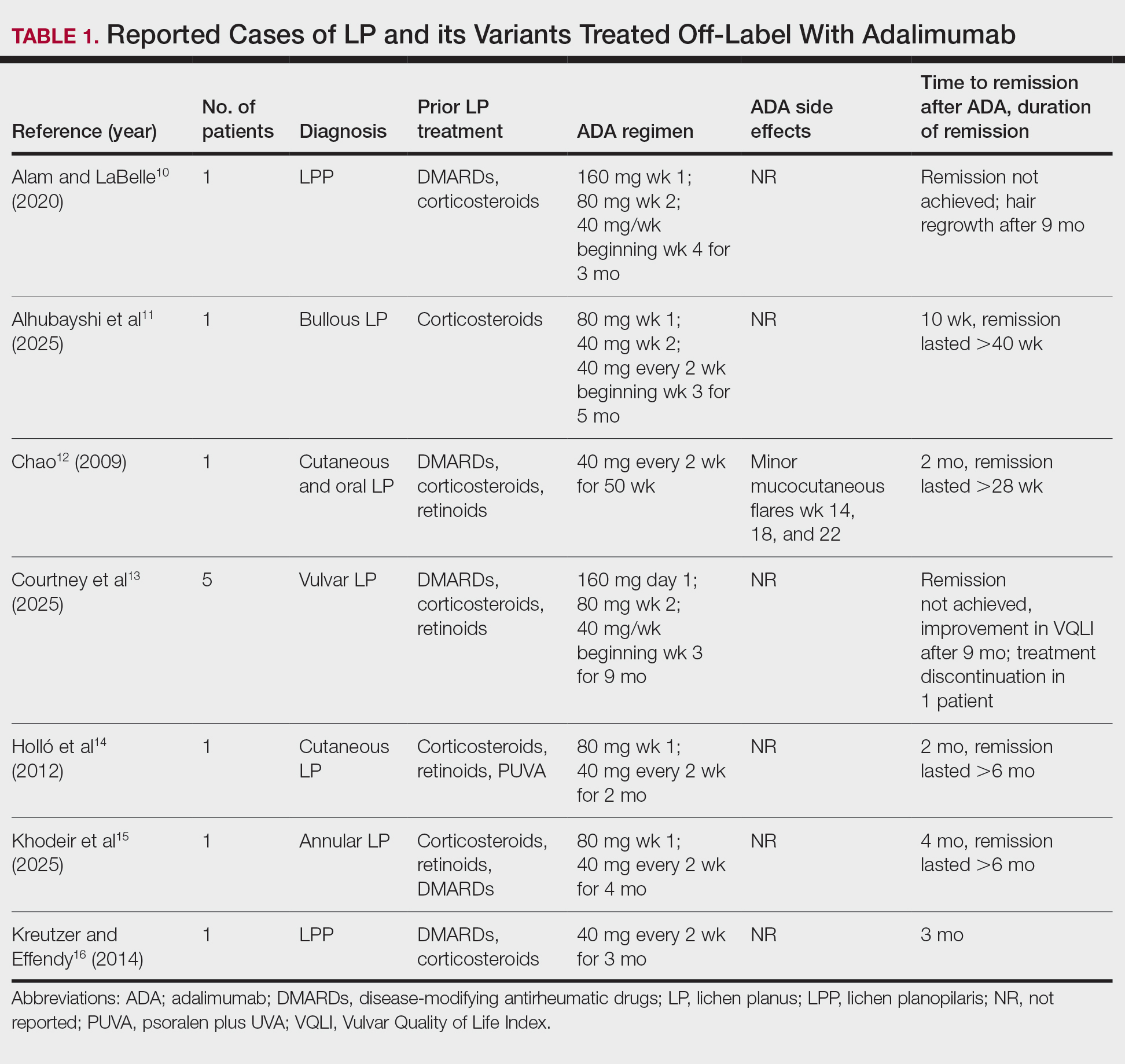
Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22
In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)

Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22

In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)

Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22

In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Practice Points
- Adalimumab can be beneficial when used off label for treatment of lichen planus in patients who do not respond to conventional therapies, including corticosteroids and immunosuppressants.
- Clinicians should be aware that adalimumab could potentially lead to paradoxical lichenoid eruptions and should monitor patients closely during treatment.
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Psoriasis, a chronic immune-mediated skin disease, is increasingly recognized for its systemic inflammation and associated cardiometabolic risk. The global rise of obesity has revealed a critical link between these 2 conditions. Current evidence confirms that obesity is an independent risk factor that can trigger psoriasis onset, drive more severe disease, and substantially impair treatment efficacy.1,2 This review synthesizes the latest evidence on the shared pathophysiology, clinical consequences, and integrated management strategies for patients with both psoriasis and obesity.
Pathophysiologic Links Between Psoriasis and Obesity
Emerging evidence highlights a bidirectional relationship between psoriasis and obesity rooted in overlapping inflammatory pathways. Both conditions are characterized by chronic inflammation mediated by cytokines that sustain systemic immune activation and metabolic dysfunction. This interplay creates a reciprocal process in which psoriatic inflammation promotes metabolic disturbances while obesity amplifies systemic inflammation and disease severity.3
Psoriasis may contribute to obesity through cytokine-driven metabolic alterations in insulin signaling and adipocyte function. The psoriatic immune response is dominated by T helper (Th) 1, Th17, and Th22 cell activation, leading to elevated levels of interferon-γ, tumor necrosis factor (TNF) α, interleukin (IL) 6, IL-17, and IL-22 from keratinocytes.4 These cytokines contribute not only to cutaneous inflammation but also to insulin resistance and adipocyte dysfunction.5 Tumor necrosis factor α and IL-6 interfere with insulin signaling via activation of stress kinases (eg, IκB kinase and c-Jun N-terminal kinase), implicating these molecules in insulin resistance and weight gain.6 Moreover, IL-17, a central cytokine in psoriasis, has been implicated in vascular inflammation, insulin resistance, and type 2 diabetes, suggesting a mechanistic link between psoriatic inflammation and metabolic disease.5 Additionally, chronic systemic inflammation in psoriasis suppresses adiponectin, a protective adipokine that enhances insulin sensitivity and exerts anti-inflammatory effects by inhibiting TNF-α and IL-6 production while promoting IL-10 synthesis. Reduced adiponectin levels have consistently been observed in patients with psoriasis and concomitant obesity or metabolic syndrome.3 The resultant imbalance between proinflammatory and anti-inflammatory mediators creates a metabolic environment conducive to obesity.3
Conversely, obesity itself may intensify both the incidence and severity of psoriasis through shared inflammatory pathways. Leptin, whose expression rises proportionally with adipocyte mass, acts as a proinflammatory mediator linking obesity to psoriasis exacerbation. By promoting Th1 and Th17 cell differentiation and suppressing regulatory T-cell activity, leptin increases IL-17A, IL-6, and TNF-α production.7 These cytokines stimulate keratinocyte proliferation and perpetuate cutaneous inflammation, thereby intensifying disease activity. Similarly, resistin, another adipokine that is elevated in obesity, stimulates monocytes and macrophages to secrete TNF-α and IL-6, creating a proinflammatory state in the body that drives the relationship between excessive fat storage (adiposity) and the development and severity of psoriasis.8 In contrast, reduced adiponectin levels in obesity remove a key anti-inflammatory regulator that normally inhibits TNF-α and IL-6 synthesis and promotes IL-10 production. This deficiency provokes unrestrained cytokine activation within both adipose and cutaneous tissue, exacerbating psoriatic immune dysregulation.9
Free fatty acids derived from abundant adipocytes in obesity further activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)–signaling and induce oxidative stress, augmenting systemic inflammation. Adipose tissue macrophages additionally secrete IL-1Β, IL-6, and TNF-α, which promote keratinocyte proliferation and amplify the psoriatic inflammatory cascade. Importantly, anti–TNF-α therapy has been shown to improve metabolic parameters, reducing low-density lipoprotein and total cholesterol levels and enhancing insulin sensitivity, highlighting the reciprocal benefits of dampening the inflammatory signaling from TNF-α.5
Together, these findings highlight that psoriasis and obesity are interconnected inflammatory disorders driven by overlapping cytokine networks, most notably TNF-α, IL-6, IL-17, and IL-23, and by imbalances in adipokines such as leptin, resistin, and adiponectin (Table).
Clinical Implications of Obesity-Related Comorbidities
Higher body mass index (BMI) has been associated with an increased and earlier incidence of psoriasis. When treated as continuous variables, both Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index scores were positively correlated with increases in BMI.10,11 This close association suggests BMI may function as a practical indicator of disease severity and prognosis.7,8,11
Psoriatic arthritis (PsA) is a major comorbidity of psoriasis that impacts disease severity and quality of life. Obesity is associated with an increased risk for PsA after accounting for traditional risk factors. Psoriatic arthritis can cause chronic mobility issues and complicate a patient’s ability to stay active. Early rheumatologist involvement may be warranted to prevent PsA development in patients with obesity and psoriasis.11,12
Patients with psoriasis have an increased baseline risk for metabolic syndrome, including obesity, type 2 diabetes, hypertension, and dyslipidemia11,13,14; therefore, the presence of obesity warrants regular metabolic evaluation. Unmanaged metabolic syndrome contributes to the increased prevalence of myocardial infarction, stroke, and heart failure seen with psoriasis.13,14 Obesity also may cause obstructive sleep apnea, which can exacerbate hypertension and heart disease due to chronic hypoxia.11 The interplay of these metabolic factors puts patients with psoriasis and obesity at heightened cardiovascular risk.13,14
Both psoriasis and obesity present psychosocial challenges such as elevated rates of depression, anxiety, and body-image concerns, all of which become more pronounced when these conditions coexist. These psychological stressors may hinder a patient’s motivation for lifestyle changes or treatment adherence.13
Dermatology visits serve as an important opportunity to monitor obesity-related comorbidities.13,15 Dermatologists are uniquely positioned to initiate metabolic screening while collaborating with primary care physicians for ongoing cardiometabolic management. Metabolic and cardiovascular baselines should be measured when care is established and should be followed longitudinally—these include BMI, waist circumference measurements, blood pressure, lipid panels, fasting glucose or A1C levels, and liver enzymes.10,14,15 Regular screening for depression, suicidality, and disordered eating also is encouraged.13 Clinicians should follow established guidelines to identify and manage metabolic, cardiovascular, and psychological comorbidities.13
Impact of Obesity on Psoriasis Treatment
Obesity is a critical factor in clinical decision-making, as it consistently is associated with diminished response to numerous systemic psoriasis therapies. This reduced efficacy has been observed with conventional oral agents such as methotrexate and cyclosporine and is particularly well documented in the context of biologic therapies.15,16 Several meta-analyses and large real-world studies have shown that higher BMI is associated with a suboptimal treatment response, with patients in higher BMI categories achieving lower rates of PASI 75 and PASI 90 than their nonobese counterparts receiving the same regimen.15,17
This efficacy gap often is rooted in pharmacokinetic challenges. For many biologics administered via subcutaneous injection, increased BMI can lead to altered drug distribution, such as sequestration in adipose tissue. Altered distribution combined with potentially increased drug clearance can result in lower overall serum drug concentrations and subsequent underdosing for a patient’s inflammatory burden.15 This reality highlights the important distinction between fixed and weight-based dosing strategies. Therapies dosed by weight, such as infliximab, have demonstrated more consistent efficacy in populations with obesity, as the dose is escalated to match patient size.18 Despite the weight-dependent effect, recent real-world studies have suggested that fixed dosing of some IL-17A inhibitors (eg, ixekizumab) remains highly effective across BMI categories, while others (eg, secukinumab) show diminished efficacy in obese patients.16,19 Furthermore, some real-world studies have reported an inverse relationship between elevated BMI and efficacy of IL-23 inhibitors, particularly guselkumab, while other studies reported no association.20,21 These mixed observations support a nuanced interpretation of BMI’s role in treatment modification; consideration should be given to the specific medication and the dosing strategy over biologic class alone.
Whether obesity independently drives psoriasis severity or mainly diminishes treatment efficacy through pharmacokinetic effects remains debated. Observational studies show a dose-dependent relationship between BMI and disease severity even in untreated patients, supporting a proinflammatory role for obesity.10 Concurrently, higher BMI may predict lower responses to some fixed-dose biologics, likely due to altered distribution and sequestration in adipose tissue.15,20 The precise contributions are challenging to delineate; both likely converge to heighten disease severity and reduce treatment response.
Beyond efficacy, obesity compounds the safety considerations of systemic treatments. Psoriasis and obesity are both strong independent risk factors for metabolic dysfunction–associated steatotic liver disease. This prevalent comorbidity creates a substantial clinical dilemma, as a first-line, cost-effective agent such as methotrexate carries a known risk for hepatotoxicity, which is amplified in patients with pre-existing liver steatosis.13 Consequently, the presence of obesity and metabolic dysfunction–associated steatotic liver disease often limits the use of methotrexate, forcing a change in therapy. Thorough baseline comorbidity screening in all patients with psoriasis and obesity is necessary to select a therapy that balances efficacy with safety.13,14
Management Strategies
Weight management plays a pivotal role in improving psoriasis outcomes. Even modest weight loss of 5% to 10% has been shown in randomized and observational studies to substantially reduce disease severity, reflected by lower PASI and Dermatology Life Quality Index scores and enhance treatment responsiveness.22,23 Dietary approaches emphasizing hypocaloric, Mediterranean, or anti-inflammatory patterns have demonstrated additional improvements in disease activity, likely through attenuation of systemic inflammation and metabolic dysregulation.21 Bariatric surgery provides more sustained benefits, with multiple studies reporting long-term remission or reduced psoriasis severity following substantial postoperative weight loss.23,24
Pharmacologic weight-loss therapies, particularly glucagonlike peptide-1 receptor agonists, have emerged as potential adjuncts in psoriasis management. Although data remain limited, these agents may reduce systemic inflammation, improve insulin sensitivity, and indirectly enhance biologic response.25
Conclusion
Psoriasis and obesity are interconnected chronic inflammatory conditions that share overlapping cytokine pathways and mutually exacerbate the clinical course. Systemic inflammation driven by cytokines such as TNF-α, IL-6, and IL-17 not only promotes psoriatic skin disease but also contributes to metabolic dysfunction and cardiovascular risk. In turn, excess adiposity amplifies inflammatory signaling and diminishes therapeutic response, creating a self-perpetuating cycle of disease.
Dermatologists should identify obesity-related risks early; counsel patients on lifestyle changes; initiate metabolic screening; and coordinate care across primary care, nutrition, and rheumatology. Regular screening for metabolic syndrome, cardiovascular comorbidities, and psychosocial distress should be integrated into psoriasis management. Future research should focus on personalized treatment strategies that integrate management of inflammatory skin disease with underlying metabolic health, such as optimizing biologic dosing and identifying novel targets that disrupt the pathophysiologic loop. By recognizing and addressing the shared inflammatory mechanisms of psoriasis and obesity, clinicians can improve both dermatologic and systemic outcomes for affected patients.
- Barrea L, Muscogiuri G, Annunziata G, et al. Update on obesity in psoriasis patients: narrative review and practical insights. Clin Cosmet Investig Dermatol. 2023;16:3089-3104.
- Owczarczyk-Saczonek A, Gornowicz-Porowska J, Zegarska B. Psoriasis comorbidities: obesity, diet, and metabolic syndrome. Int J Mol Sci. 2024;25:1832.
- Vata D, Tarcau BM, Popescu IA, et al. Update on obesity in psoriasis patients. Life (Basel). 2023;13:1947.
- Piaserico S, Orlando G, Messina F. Psoriasis and cardiometabolic diseases: shared genetic and molecular pathways. Int J Mol Sci. 2022;23:9063.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Kern L, Mittenbühler MJ, Vesting AJ, et al. Obesity-induced TNF-α and IL-6 signaling: the missing link between obesity and inflammation-driven liver and colorectal cancers. Cancers (Basel). 2019;11:24.
- Hwang J, Yoo JA, Yoon H, et al. Role of leptin in the association between obesity and psoriasis. Biomol Ther (Seoul). 2021;29:11-21.
- Smith B, Devjani S, Collier MR, et al. Association between psoriasis and obesity among US adults in the 2009-2014 National Health and Nutrition Examination Survey. Cutis. 2023;112:49-51. doi:10.12788/cutis.0807
- Ellulu MS, Patimah I, Khaza’ai H. Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci. 2017;13:851-863.
- Wang H, Hou S, Kang X, et al. BMI matters: understanding the link between weight and severe psoriasis. Sci Rep. 2025;15:11158.
- Norden A, Rekhtman S, Strunk A, et al. Risk of psoriasis according to body mass index: a retrospective cohort analysis. J Am Acad Dermatol. 2022;86:1020-1026.
- Di Caprio R, Nigro E, Di Brizzi EV, et al. Exploring the link between psoriasis and adipose tissue: one amplifies the other. Int J Mol Sci. 2024;25:13435.
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113.
- Secchiero P, Rimondi E, Marcuzzi A, et al. Metabolic syndrome and psoriasis: pivotal roles of chronic inflammation and gut microbiota. Int J Mol Sci. 2024;25:8098.
- Burshtein J, Armstrong A, Chow M, et al. Association between obesity and efficacy of psoriasis therapies: an expert consensus panel. J Am Acad Dermatol. 2025;92:807-815. doi:10.1016/j.jaad.2024.12.016
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925.
- Hjort G, Schwarz CW, Skov L, et al. Clinical characteristics associated with response to biologics in the treatment of psoriasis: a meta-analysis. JAMA Dermatol. 2024;160:830-837.
- Naldi L, Chimenti S, Girolomoni G, et al. Efficacy and safety of infliximab in obese and non-obese patients with plaque-type psoriasis: subanalysis of the EXPRESS II trial. Br J Dermatol. 2008;159:761-766.
- Puig L, Thom H, Mollon P, et al. Effect of body weight on the efficacy of biologics in moderate-to-severe plaque psoriasis: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2020;34:237-245.
- Dai M, Jiang Y, Wang Y, et al. Differential clinical factors influencing the effectiveness of distinct biologic agents in psoriasis: insights from a prospective cohort study in China. Inflamm Res. 2026;75:25. doi:10.1007/s00011-025-02179-1
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti–IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Jensen P, Zachariae C, Christensen R, et al. Effect of weight loss on the severity of psoriasis: a randomized clinical study. Br J Dermatol. 2013;168:319-327.
- Hossler EW, Wood GC, Still CD, et al. Psoriasis improvement following bariatric surgery is durable: 5-year follow-up in the Geisinger bariatric surgery cohort. Obes Surg. 2020;30:3350-3356.
- Romero-Talamás H, Daigle CR, Aminian A, et al. Psoriasis improvement after bariatric surgery. Surg Obes Relat Dis. 2014;10:1155-1159.
- Buonanno S, Gaggiano C, Terribili R, et al. Potential role of GLP-1 receptor agonists in the management of psoriatic disease: a scoping review. Inflamm Res. 2025;74:167. doi:10.1007/s00011-025-02140-2
Psoriasis, a chronic immune-mediated skin disease, is increasingly recognized for its systemic inflammation and associated cardiometabolic risk. The global rise of obesity has revealed a critical link between these 2 conditions. Current evidence confirms that obesity is an independent risk factor that can trigger psoriasis onset, drive more severe disease, and substantially impair treatment efficacy.1,2 This review synthesizes the latest evidence on the shared pathophysiology, clinical consequences, and integrated management strategies for patients with both psoriasis and obesity.
Pathophysiologic Links Between Psoriasis and Obesity
Emerging evidence highlights a bidirectional relationship between psoriasis and obesity rooted in overlapping inflammatory pathways. Both conditions are characterized by chronic inflammation mediated by cytokines that sustain systemic immune activation and metabolic dysfunction. This interplay creates a reciprocal process in which psoriatic inflammation promotes metabolic disturbances while obesity amplifies systemic inflammation and disease severity.3
Psoriasis may contribute to obesity through cytokine-driven metabolic alterations in insulin signaling and adipocyte function. The psoriatic immune response is dominated by T helper (Th) 1, Th17, and Th22 cell activation, leading to elevated levels of interferon-γ, tumor necrosis factor (TNF) α, interleukin (IL) 6, IL-17, and IL-22 from keratinocytes.4 These cytokines contribute not only to cutaneous inflammation but also to insulin resistance and adipocyte dysfunction.5 Tumor necrosis factor α and IL-6 interfere with insulin signaling via activation of stress kinases (eg, IκB kinase and c-Jun N-terminal kinase), implicating these molecules in insulin resistance and weight gain.6 Moreover, IL-17, a central cytokine in psoriasis, has been implicated in vascular inflammation, insulin resistance, and type 2 diabetes, suggesting a mechanistic link between psoriatic inflammation and metabolic disease.5 Additionally, chronic systemic inflammation in psoriasis suppresses adiponectin, a protective adipokine that enhances insulin sensitivity and exerts anti-inflammatory effects by inhibiting TNF-α and IL-6 production while promoting IL-10 synthesis. Reduced adiponectin levels have consistently been observed in patients with psoriasis and concomitant obesity or metabolic syndrome.3 The resultant imbalance between proinflammatory and anti-inflammatory mediators creates a metabolic environment conducive to obesity.3
Conversely, obesity itself may intensify both the incidence and severity of psoriasis through shared inflammatory pathways. Leptin, whose expression rises proportionally with adipocyte mass, acts as a proinflammatory mediator linking obesity to psoriasis exacerbation. By promoting Th1 and Th17 cell differentiation and suppressing regulatory T-cell activity, leptin increases IL-17A, IL-6, and TNF-α production.7 These cytokines stimulate keratinocyte proliferation and perpetuate cutaneous inflammation, thereby intensifying disease activity. Similarly, resistin, another adipokine that is elevated in obesity, stimulates monocytes and macrophages to secrete TNF-α and IL-6, creating a proinflammatory state in the body that drives the relationship between excessive fat storage (adiposity) and the development and severity of psoriasis.8 In contrast, reduced adiponectin levels in obesity remove a key anti-inflammatory regulator that normally inhibits TNF-α and IL-6 synthesis and promotes IL-10 production. This deficiency provokes unrestrained cytokine activation within both adipose and cutaneous tissue, exacerbating psoriatic immune dysregulation.9
Free fatty acids derived from abundant adipocytes in obesity further activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)–signaling and induce oxidative stress, augmenting systemic inflammation. Adipose tissue macrophages additionally secrete IL-1Β, IL-6, and TNF-α, which promote keratinocyte proliferation and amplify the psoriatic inflammatory cascade. Importantly, anti–TNF-α therapy has been shown to improve metabolic parameters, reducing low-density lipoprotein and total cholesterol levels and enhancing insulin sensitivity, highlighting the reciprocal benefits of dampening the inflammatory signaling from TNF-α.5
Together, these findings highlight that psoriasis and obesity are interconnected inflammatory disorders driven by overlapping cytokine networks, most notably TNF-α, IL-6, IL-17, and IL-23, and by imbalances in adipokines such as leptin, resistin, and adiponectin (Table).

Clinical Implications of Obesity-Related Comorbidities
Higher body mass index (BMI) has been associated with an increased and earlier incidence of psoriasis. When treated as continuous variables, both Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index scores were positively correlated with increases in BMI.10,11 This close association suggests BMI may function as a practical indicator of disease severity and prognosis.7,8,11
Psoriatic arthritis (PsA) is a major comorbidity of psoriasis that impacts disease severity and quality of life. Obesity is associated with an increased risk for PsA after accounting for traditional risk factors. Psoriatic arthritis can cause chronic mobility issues and complicate a patient’s ability to stay active. Early rheumatologist involvement may be warranted to prevent PsA development in patients with obesity and psoriasis.11,12
Patients with psoriasis have an increased baseline risk for metabolic syndrome, including obesity, type 2 diabetes, hypertension, and dyslipidemia11,13,14; therefore, the presence of obesity warrants regular metabolic evaluation. Unmanaged metabolic syndrome contributes to the increased prevalence of myocardial infarction, stroke, and heart failure seen with psoriasis.13,14 Obesity also may cause obstructive sleep apnea, which can exacerbate hypertension and heart disease due to chronic hypoxia.11 The interplay of these metabolic factors puts patients with psoriasis and obesity at heightened cardiovascular risk.13,14
Both psoriasis and obesity present psychosocial challenges such as elevated rates of depression, anxiety, and body-image concerns, all of which become more pronounced when these conditions coexist. These psychological stressors may hinder a patient’s motivation for lifestyle changes or treatment adherence.13
Dermatology visits serve as an important opportunity to monitor obesity-related comorbidities.13,15 Dermatologists are uniquely positioned to initiate metabolic screening while collaborating with primary care physicians for ongoing cardiometabolic management. Metabolic and cardiovascular baselines should be measured when care is established and should be followed longitudinally—these include BMI, waist circumference measurements, blood pressure, lipid panels, fasting glucose or A1C levels, and liver enzymes.10,14,15 Regular screening for depression, suicidality, and disordered eating also is encouraged.13 Clinicians should follow established guidelines to identify and manage metabolic, cardiovascular, and psychological comorbidities.13
Impact of Obesity on Psoriasis Treatment
Obesity is a critical factor in clinical decision-making, as it consistently is associated with diminished response to numerous systemic psoriasis therapies. This reduced efficacy has been observed with conventional oral agents such as methotrexate and cyclosporine and is particularly well documented in the context of biologic therapies.15,16 Several meta-analyses and large real-world studies have shown that higher BMI is associated with a suboptimal treatment response, with patients in higher BMI categories achieving lower rates of PASI 75 and PASI 90 than their nonobese counterparts receiving the same regimen.15,17
This efficacy gap often is rooted in pharmacokinetic challenges. For many biologics administered via subcutaneous injection, increased BMI can lead to altered drug distribution, such as sequestration in adipose tissue. Altered distribution combined with potentially increased drug clearance can result in lower overall serum drug concentrations and subsequent underdosing for a patient’s inflammatory burden.15 This reality highlights the important distinction between fixed and weight-based dosing strategies. Therapies dosed by weight, such as infliximab, have demonstrated more consistent efficacy in populations with obesity, as the dose is escalated to match patient size.18 Despite the weight-dependent effect, recent real-world studies have suggested that fixed dosing of some IL-17A inhibitors (eg, ixekizumab) remains highly effective across BMI categories, while others (eg, secukinumab) show diminished efficacy in obese patients.16,19 Furthermore, some real-world studies have reported an inverse relationship between elevated BMI and efficacy of IL-23 inhibitors, particularly guselkumab, while other studies reported no association.20,21 These mixed observations support a nuanced interpretation of BMI’s role in treatment modification; consideration should be given to the specific medication and the dosing strategy over biologic class alone.
Whether obesity independently drives psoriasis severity or mainly diminishes treatment efficacy through pharmacokinetic effects remains debated. Observational studies show a dose-dependent relationship between BMI and disease severity even in untreated patients, supporting a proinflammatory role for obesity.10 Concurrently, higher BMI may predict lower responses to some fixed-dose biologics, likely due to altered distribution and sequestration in adipose tissue.15,20 The precise contributions are challenging to delineate; both likely converge to heighten disease severity and reduce treatment response.
Beyond efficacy, obesity compounds the safety considerations of systemic treatments. Psoriasis and obesity are both strong independent risk factors for metabolic dysfunction–associated steatotic liver disease. This prevalent comorbidity creates a substantial clinical dilemma, as a first-line, cost-effective agent such as methotrexate carries a known risk for hepatotoxicity, which is amplified in patients with pre-existing liver steatosis.13 Consequently, the presence of obesity and metabolic dysfunction–associated steatotic liver disease often limits the use of methotrexate, forcing a change in therapy. Thorough baseline comorbidity screening in all patients with psoriasis and obesity is necessary to select a therapy that balances efficacy with safety.13,14
Management Strategies
Weight management plays a pivotal role in improving psoriasis outcomes. Even modest weight loss of 5% to 10% has been shown in randomized and observational studies to substantially reduce disease severity, reflected by lower PASI and Dermatology Life Quality Index scores and enhance treatment responsiveness.22,23 Dietary approaches emphasizing hypocaloric, Mediterranean, or anti-inflammatory patterns have demonstrated additional improvements in disease activity, likely through attenuation of systemic inflammation and metabolic dysregulation.21 Bariatric surgery provides more sustained benefits, with multiple studies reporting long-term remission or reduced psoriasis severity following substantial postoperative weight loss.23,24
Pharmacologic weight-loss therapies, particularly glucagonlike peptide-1 receptor agonists, have emerged as potential adjuncts in psoriasis management. Although data remain limited, these agents may reduce systemic inflammation, improve insulin sensitivity, and indirectly enhance biologic response.25
Conclusion
Psoriasis and obesity are interconnected chronic inflammatory conditions that share overlapping cytokine pathways and mutually exacerbate the clinical course. Systemic inflammation driven by cytokines such as TNF-α, IL-6, and IL-17 not only promotes psoriatic skin disease but also contributes to metabolic dysfunction and cardiovascular risk. In turn, excess adiposity amplifies inflammatory signaling and diminishes therapeutic response, creating a self-perpetuating cycle of disease.
Dermatologists should identify obesity-related risks early; counsel patients on lifestyle changes; initiate metabolic screening; and coordinate care across primary care, nutrition, and rheumatology. Regular screening for metabolic syndrome, cardiovascular comorbidities, and psychosocial distress should be integrated into psoriasis management. Future research should focus on personalized treatment strategies that integrate management of inflammatory skin disease with underlying metabolic health, such as optimizing biologic dosing and identifying novel targets that disrupt the pathophysiologic loop. By recognizing and addressing the shared inflammatory mechanisms of psoriasis and obesity, clinicians can improve both dermatologic and systemic outcomes for affected patients.
Psoriasis, a chronic immune-mediated skin disease, is increasingly recognized for its systemic inflammation and associated cardiometabolic risk. The global rise of obesity has revealed a critical link between these 2 conditions. Current evidence confirms that obesity is an independent risk factor that can trigger psoriasis onset, drive more severe disease, and substantially impair treatment efficacy.1,2 This review synthesizes the latest evidence on the shared pathophysiology, clinical consequences, and integrated management strategies for patients with both psoriasis and obesity.
Pathophysiologic Links Between Psoriasis and Obesity
Emerging evidence highlights a bidirectional relationship between psoriasis and obesity rooted in overlapping inflammatory pathways. Both conditions are characterized by chronic inflammation mediated by cytokines that sustain systemic immune activation and metabolic dysfunction. This interplay creates a reciprocal process in which psoriatic inflammation promotes metabolic disturbances while obesity amplifies systemic inflammation and disease severity.3
Psoriasis may contribute to obesity through cytokine-driven metabolic alterations in insulin signaling and adipocyte function. The psoriatic immune response is dominated by T helper (Th) 1, Th17, and Th22 cell activation, leading to elevated levels of interferon-γ, tumor necrosis factor (TNF) α, interleukin (IL) 6, IL-17, and IL-22 from keratinocytes.4 These cytokines contribute not only to cutaneous inflammation but also to insulin resistance and adipocyte dysfunction.5 Tumor necrosis factor α and IL-6 interfere with insulin signaling via activation of stress kinases (eg, IκB kinase and c-Jun N-terminal kinase), implicating these molecules in insulin resistance and weight gain.6 Moreover, IL-17, a central cytokine in psoriasis, has been implicated in vascular inflammation, insulin resistance, and type 2 diabetes, suggesting a mechanistic link between psoriatic inflammation and metabolic disease.5 Additionally, chronic systemic inflammation in psoriasis suppresses adiponectin, a protective adipokine that enhances insulin sensitivity and exerts anti-inflammatory effects by inhibiting TNF-α and IL-6 production while promoting IL-10 synthesis. Reduced adiponectin levels have consistently been observed in patients with psoriasis and concomitant obesity or metabolic syndrome.3 The resultant imbalance between proinflammatory and anti-inflammatory mediators creates a metabolic environment conducive to obesity.3
Conversely, obesity itself may intensify both the incidence and severity of psoriasis through shared inflammatory pathways. Leptin, whose expression rises proportionally with adipocyte mass, acts as a proinflammatory mediator linking obesity to psoriasis exacerbation. By promoting Th1 and Th17 cell differentiation and suppressing regulatory T-cell activity, leptin increases IL-17A, IL-6, and TNF-α production.7 These cytokines stimulate keratinocyte proliferation and perpetuate cutaneous inflammation, thereby intensifying disease activity. Similarly, resistin, another adipokine that is elevated in obesity, stimulates monocytes and macrophages to secrete TNF-α and IL-6, creating a proinflammatory state in the body that drives the relationship between excessive fat storage (adiposity) and the development and severity of psoriasis.8 In contrast, reduced adiponectin levels in obesity remove a key anti-inflammatory regulator that normally inhibits TNF-α and IL-6 synthesis and promotes IL-10 production. This deficiency provokes unrestrained cytokine activation within both adipose and cutaneous tissue, exacerbating psoriatic immune dysregulation.9
Free fatty acids derived from abundant adipocytes in obesity further activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)–signaling and induce oxidative stress, augmenting systemic inflammation. Adipose tissue macrophages additionally secrete IL-1Β, IL-6, and TNF-α, which promote keratinocyte proliferation and amplify the psoriatic inflammatory cascade. Importantly, anti–TNF-α therapy has been shown to improve metabolic parameters, reducing low-density lipoprotein and total cholesterol levels and enhancing insulin sensitivity, highlighting the reciprocal benefits of dampening the inflammatory signaling from TNF-α.5
Together, these findings highlight that psoriasis and obesity are interconnected inflammatory disorders driven by overlapping cytokine networks, most notably TNF-α, IL-6, IL-17, and IL-23, and by imbalances in adipokines such as leptin, resistin, and adiponectin (Table).

Clinical Implications of Obesity-Related Comorbidities
Higher body mass index (BMI) has been associated with an increased and earlier incidence of psoriasis. When treated as continuous variables, both Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index scores were positively correlated with increases in BMI.10,11 This close association suggests BMI may function as a practical indicator of disease severity and prognosis.7,8,11
Psoriatic arthritis (PsA) is a major comorbidity of psoriasis that impacts disease severity and quality of life. Obesity is associated with an increased risk for PsA after accounting for traditional risk factors. Psoriatic arthritis can cause chronic mobility issues and complicate a patient’s ability to stay active. Early rheumatologist involvement may be warranted to prevent PsA development in patients with obesity and psoriasis.11,12
Patients with psoriasis have an increased baseline risk for metabolic syndrome, including obesity, type 2 diabetes, hypertension, and dyslipidemia11,13,14; therefore, the presence of obesity warrants regular metabolic evaluation. Unmanaged metabolic syndrome contributes to the increased prevalence of myocardial infarction, stroke, and heart failure seen with psoriasis.13,14 Obesity also may cause obstructive sleep apnea, which can exacerbate hypertension and heart disease due to chronic hypoxia.11 The interplay of these metabolic factors puts patients with psoriasis and obesity at heightened cardiovascular risk.13,14
Both psoriasis and obesity present psychosocial challenges such as elevated rates of depression, anxiety, and body-image concerns, all of which become more pronounced when these conditions coexist. These psychological stressors may hinder a patient’s motivation for lifestyle changes or treatment adherence.13
Dermatology visits serve as an important opportunity to monitor obesity-related comorbidities.13,15 Dermatologists are uniquely positioned to initiate metabolic screening while collaborating with primary care physicians for ongoing cardiometabolic management. Metabolic and cardiovascular baselines should be measured when care is established and should be followed longitudinally—these include BMI, waist circumference measurements, blood pressure, lipid panels, fasting glucose or A1C levels, and liver enzymes.10,14,15 Regular screening for depression, suicidality, and disordered eating also is encouraged.13 Clinicians should follow established guidelines to identify and manage metabolic, cardiovascular, and psychological comorbidities.13
Impact of Obesity on Psoriasis Treatment
Obesity is a critical factor in clinical decision-making, as it consistently is associated with diminished response to numerous systemic psoriasis therapies. This reduced efficacy has been observed with conventional oral agents such as methotrexate and cyclosporine and is particularly well documented in the context of biologic therapies.15,16 Several meta-analyses and large real-world studies have shown that higher BMI is associated with a suboptimal treatment response, with patients in higher BMI categories achieving lower rates of PASI 75 and PASI 90 than their nonobese counterparts receiving the same regimen.15,17
This efficacy gap often is rooted in pharmacokinetic challenges. For many biologics administered via subcutaneous injection, increased BMI can lead to altered drug distribution, such as sequestration in adipose tissue. Altered distribution combined with potentially increased drug clearance can result in lower overall serum drug concentrations and subsequent underdosing for a patient’s inflammatory burden.15 This reality highlights the important distinction between fixed and weight-based dosing strategies. Therapies dosed by weight, such as infliximab, have demonstrated more consistent efficacy in populations with obesity, as the dose is escalated to match patient size.18 Despite the weight-dependent effect, recent real-world studies have suggested that fixed dosing of some IL-17A inhibitors (eg, ixekizumab) remains highly effective across BMI categories, while others (eg, secukinumab) show diminished efficacy in obese patients.16,19 Furthermore, some real-world studies have reported an inverse relationship between elevated BMI and efficacy of IL-23 inhibitors, particularly guselkumab, while other studies reported no association.20,21 These mixed observations support a nuanced interpretation of BMI’s role in treatment modification; consideration should be given to the specific medication and the dosing strategy over biologic class alone.
Whether obesity independently drives psoriasis severity or mainly diminishes treatment efficacy through pharmacokinetic effects remains debated. Observational studies show a dose-dependent relationship between BMI and disease severity even in untreated patients, supporting a proinflammatory role for obesity.10 Concurrently, higher BMI may predict lower responses to some fixed-dose biologics, likely due to altered distribution and sequestration in adipose tissue.15,20 The precise contributions are challenging to delineate; both likely converge to heighten disease severity and reduce treatment response.
Beyond efficacy, obesity compounds the safety considerations of systemic treatments. Psoriasis and obesity are both strong independent risk factors for metabolic dysfunction–associated steatotic liver disease. This prevalent comorbidity creates a substantial clinical dilemma, as a first-line, cost-effective agent such as methotrexate carries a known risk for hepatotoxicity, which is amplified in patients with pre-existing liver steatosis.13 Consequently, the presence of obesity and metabolic dysfunction–associated steatotic liver disease often limits the use of methotrexate, forcing a change in therapy. Thorough baseline comorbidity screening in all patients with psoriasis and obesity is necessary to select a therapy that balances efficacy with safety.13,14
Management Strategies
Weight management plays a pivotal role in improving psoriasis outcomes. Even modest weight loss of 5% to 10% has been shown in randomized and observational studies to substantially reduce disease severity, reflected by lower PASI and Dermatology Life Quality Index scores and enhance treatment responsiveness.22,23 Dietary approaches emphasizing hypocaloric, Mediterranean, or anti-inflammatory patterns have demonstrated additional improvements in disease activity, likely through attenuation of systemic inflammation and metabolic dysregulation.21 Bariatric surgery provides more sustained benefits, with multiple studies reporting long-term remission or reduced psoriasis severity following substantial postoperative weight loss.23,24
Pharmacologic weight-loss therapies, particularly glucagonlike peptide-1 receptor agonists, have emerged as potential adjuncts in psoriasis management. Although data remain limited, these agents may reduce systemic inflammation, improve insulin sensitivity, and indirectly enhance biologic response.25
Conclusion
Psoriasis and obesity are interconnected chronic inflammatory conditions that share overlapping cytokine pathways and mutually exacerbate the clinical course. Systemic inflammation driven by cytokines such as TNF-α, IL-6, and IL-17 not only promotes psoriatic skin disease but also contributes to metabolic dysfunction and cardiovascular risk. In turn, excess adiposity amplifies inflammatory signaling and diminishes therapeutic response, creating a self-perpetuating cycle of disease.
Dermatologists should identify obesity-related risks early; counsel patients on lifestyle changes; initiate metabolic screening; and coordinate care across primary care, nutrition, and rheumatology. Regular screening for metabolic syndrome, cardiovascular comorbidities, and psychosocial distress should be integrated into psoriasis management. Future research should focus on personalized treatment strategies that integrate management of inflammatory skin disease with underlying metabolic health, such as optimizing biologic dosing and identifying novel targets that disrupt the pathophysiologic loop. By recognizing and addressing the shared inflammatory mechanisms of psoriasis and obesity, clinicians can improve both dermatologic and systemic outcomes for affected patients.
- Barrea L, Muscogiuri G, Annunziata G, et al. Update on obesity in psoriasis patients: narrative review and practical insights. Clin Cosmet Investig Dermatol. 2023;16:3089-3104.
- Owczarczyk-Saczonek A, Gornowicz-Porowska J, Zegarska B. Psoriasis comorbidities: obesity, diet, and metabolic syndrome. Int J Mol Sci. 2024;25:1832.
- Vata D, Tarcau BM, Popescu IA, et al. Update on obesity in psoriasis patients. Life (Basel). 2023;13:1947.
- Piaserico S, Orlando G, Messina F. Psoriasis and cardiometabolic diseases: shared genetic and molecular pathways. Int J Mol Sci. 2022;23:9063.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Kern L, Mittenbühler MJ, Vesting AJ, et al. Obesity-induced TNF-α and IL-6 signaling: the missing link between obesity and inflammation-driven liver and colorectal cancers. Cancers (Basel). 2019;11:24.
- Hwang J, Yoo JA, Yoon H, et al. Role of leptin in the association between obesity and psoriasis. Biomol Ther (Seoul). 2021;29:11-21.
- Smith B, Devjani S, Collier MR, et al. Association between psoriasis and obesity among US adults in the 2009-2014 National Health and Nutrition Examination Survey. Cutis. 2023;112:49-51. doi:10.12788/cutis.0807
- Ellulu MS, Patimah I, Khaza’ai H. Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci. 2017;13:851-863.
- Wang H, Hou S, Kang X, et al. BMI matters: understanding the link between weight and severe psoriasis. Sci Rep. 2025;15:11158.
- Norden A, Rekhtman S, Strunk A, et al. Risk of psoriasis according to body mass index: a retrospective cohort analysis. J Am Acad Dermatol. 2022;86:1020-1026.
- Di Caprio R, Nigro E, Di Brizzi EV, et al. Exploring the link between psoriasis and adipose tissue: one amplifies the other. Int J Mol Sci. 2024;25:13435.
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113.
- Secchiero P, Rimondi E, Marcuzzi A, et al. Metabolic syndrome and psoriasis: pivotal roles of chronic inflammation and gut microbiota. Int J Mol Sci. 2024;25:8098.
- Burshtein J, Armstrong A, Chow M, et al. Association between obesity and efficacy of psoriasis therapies: an expert consensus panel. J Am Acad Dermatol. 2025;92:807-815. doi:10.1016/j.jaad.2024.12.016
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925.
- Hjort G, Schwarz CW, Skov L, et al. Clinical characteristics associated with response to biologics in the treatment of psoriasis: a meta-analysis. JAMA Dermatol. 2024;160:830-837.
- Naldi L, Chimenti S, Girolomoni G, et al. Efficacy and safety of infliximab in obese and non-obese patients with plaque-type psoriasis: subanalysis of the EXPRESS II trial. Br J Dermatol. 2008;159:761-766.
- Puig L, Thom H, Mollon P, et al. Effect of body weight on the efficacy of biologics in moderate-to-severe plaque psoriasis: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2020;34:237-245.
- Dai M, Jiang Y, Wang Y, et al. Differential clinical factors influencing the effectiveness of distinct biologic agents in psoriasis: insights from a prospective cohort study in China. Inflamm Res. 2026;75:25. doi:10.1007/s00011-025-02179-1
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti–IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Jensen P, Zachariae C, Christensen R, et al. Effect of weight loss on the severity of psoriasis: a randomized clinical study. Br J Dermatol. 2013;168:319-327.
- Hossler EW, Wood GC, Still CD, et al. Psoriasis improvement following bariatric surgery is durable: 5-year follow-up in the Geisinger bariatric surgery cohort. Obes Surg. 2020;30:3350-3356.
- Romero-Talamás H, Daigle CR, Aminian A, et al. Psoriasis improvement after bariatric surgery. Surg Obes Relat Dis. 2014;10:1155-1159.
- Buonanno S, Gaggiano C, Terribili R, et al. Potential role of GLP-1 receptor agonists in the management of psoriatic disease: a scoping review. Inflamm Res. 2025;74:167. doi:10.1007/s00011-025-02140-2
- Barrea L, Muscogiuri G, Annunziata G, et al. Update on obesity in psoriasis patients: narrative review and practical insights. Clin Cosmet Investig Dermatol. 2023;16:3089-3104.
- Owczarczyk-Saczonek A, Gornowicz-Porowska J, Zegarska B. Psoriasis comorbidities: obesity, diet, and metabolic syndrome. Int J Mol Sci. 2024;25:1832.
- Vata D, Tarcau BM, Popescu IA, et al. Update on obesity in psoriasis patients. Life (Basel). 2023;13:1947.
- Piaserico S, Orlando G, Messina F. Psoriasis and cardiometabolic diseases: shared genetic and molecular pathways. Int J Mol Sci. 2022;23:9063.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Kern L, Mittenbühler MJ, Vesting AJ, et al. Obesity-induced TNF-α and IL-6 signaling: the missing link between obesity and inflammation-driven liver and colorectal cancers. Cancers (Basel). 2019;11:24.
- Hwang J, Yoo JA, Yoon H, et al. Role of leptin in the association between obesity and psoriasis. Biomol Ther (Seoul). 2021;29:11-21.
- Smith B, Devjani S, Collier MR, et al. Association between psoriasis and obesity among US adults in the 2009-2014 National Health and Nutrition Examination Survey. Cutis. 2023;112:49-51. doi:10.12788/cutis.0807
- Ellulu MS, Patimah I, Khaza’ai H. Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci. 2017;13:851-863.
- Wang H, Hou S, Kang X, et al. BMI matters: understanding the link between weight and severe psoriasis. Sci Rep. 2025;15:11158.
- Norden A, Rekhtman S, Strunk A, et al. Risk of psoriasis according to body mass index: a retrospective cohort analysis. J Am Acad Dermatol. 2022;86:1020-1026.
- Di Caprio R, Nigro E, Di Brizzi EV, et al. Exploring the link between psoriasis and adipose tissue: one amplifies the other. Int J Mol Sci. 2024;25:13435.
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113.
- Secchiero P, Rimondi E, Marcuzzi A, et al. Metabolic syndrome and psoriasis: pivotal roles of chronic inflammation and gut microbiota. Int J Mol Sci. 2024;25:8098.
- Burshtein J, Armstrong A, Chow M, et al. Association between obesity and efficacy of psoriasis therapies: an expert consensus panel. J Am Acad Dermatol. 2025;92:807-815. doi:10.1016/j.jaad.2024.12.016
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925.
- Hjort G, Schwarz CW, Skov L, et al. Clinical characteristics associated with response to biologics in the treatment of psoriasis: a meta-analysis. JAMA Dermatol. 2024;160:830-837.
- Naldi L, Chimenti S, Girolomoni G, et al. Efficacy and safety of infliximab in obese and non-obese patients with plaque-type psoriasis: subanalysis of the EXPRESS II trial. Br J Dermatol. 2008;159:761-766.
- Puig L, Thom H, Mollon P, et al. Effect of body weight on the efficacy of biologics in moderate-to-severe plaque psoriasis: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2020;34:237-245.
- Dai M, Jiang Y, Wang Y, et al. Differential clinical factors influencing the effectiveness of distinct biologic agents in psoriasis: insights from a prospective cohort study in China. Inflamm Res. 2026;75:25. doi:10.1007/s00011-025-02179-1
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti–IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Jensen P, Zachariae C, Christensen R, et al. Effect of weight loss on the severity of psoriasis: a randomized clinical study. Br J Dermatol. 2013;168:319-327.
- Hossler EW, Wood GC, Still CD, et al. Psoriasis improvement following bariatric surgery is durable: 5-year follow-up in the Geisinger bariatric surgery cohort. Obes Surg. 2020;30:3350-3356.
- Romero-Talamás H, Daigle CR, Aminian A, et al. Psoriasis improvement after bariatric surgery. Surg Obes Relat Dis. 2014;10:1155-1159.
- Buonanno S, Gaggiano C, Terribili R, et al. Potential role of GLP-1 receptor agonists in the management of psoriatic disease: a scoping review. Inflamm Res. 2025;74:167. doi:10.1007/s00011-025-02140-2
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Practice Points
- Obesity is an independent risk factor for psoriasis onset and severity. Both conditions share overlapping inflammatory pathways that create a self-perpetuating cycle of metabolic and cutaneous dysfunction.
- Dermatologists play a key role in early detection of comorbidities, and patients with psoriasis and obesity should undergo regular screening for metabolic syndrome, liver disease, and psoriatic arthritis.
- Weight loss is a critical therapeutic intervention that may improve Psoriasis Area and Severity Index scores and restore therapeutic responsiveness.
Environmental and Lifestyle Triggers of Rosacea
Environmental and Lifestyle Triggers of Rosacea
Rosacea is a chronic inflammatory skin disease characterized by erythema, flushing, telangiectasias, papules, pustules, and rarely, phymatous changes that primarily manifest in a centrofacial distribution.1,2 Although establishing the true prevalence of rosacea may be challenging due to a wide spectrum of clinical manifestations, current studies estimate that it is between 5% to 6% of the global adult population and that rosacea most commonly is diagnosed in patients aged 30 and 60 years, though it occasionally can affect adolescents and children.3,4 Although the origin and pathophysiology of rosacea remain incompletely understood, the condition arises from a complex interplay of genetic, environmental, immune, microbial, and neurovascular factors; this interplay ultimately leads to excessive production of inflammatory and vasoactive peptides, chronic inflammation, and neurovascular hyperreactivity.1,5-7
Identifying triggers can be valuable in managing rosacea, as avoidance of these exposures may lead to disease improvement. In this review, we highlight 4 major environmental triggers of rosacea—UV radiation exposure, temperature fluctuation, skin care practices, and diet—and their roles in its pathogenesis and management. A high-level summary of recommendations can be found in the Table.
UV Radiation Exposure
Exposure to UV radiation is a known trigger of rosacea and may worsen symptoms through several mechanisms.8,9 It increases the production of inflammatory cytokines, which enhance the release of vascular endothelial growth factor, promoting angiogenesis and vasodilation.10 Exposure to UV radiation also contributes to tissue inflammation through the production of reactive oxygen species, further mediating inflammatory cascades and leading to immune dysregulation.11,12 Interestingly, though the mechanisms by which UV radiation may contribute to the pathophysiology of rosacea are well described, it remains unclear whether chronic UV exposure plays a major role in the pathogenesis or disease progression of rosacea.1 Studies have observed that increased exposure to sunlight seems to be correlated with increased severity of redness but not of papules and pustules.13,14
Despite some uncertainty regarding the relationship between rosacea and chronic UV exposure, sun protection is a prudent recommendation in this patient population, particularly given other risks of exposure to UV radiation, such as photoaging and skin cancer.9,15,16 Sun protection can be accomplished using broad-spectrum sunscreen (sun protection factor 50 or higher, reapplied every 2 to 4 hours) or by wearing physical protection (eg, hats, sun-protective clothing) along with avoidance of sun exposure during peak UV hours (ie, 10 am-
Temperature Fluctuation
Both heat and cold exposure have been suggested as triggers for rosacea, thought to be mediated through dysregulations in neurovascular and thermal pathways, resulting in increased flushing and erythema.6 Skin affected by rosacea exhibits a lower threshold for temperature and pain stimuli, resulting in heightened hypersensitivity compared to normal skin.18 Exposure to heat activates thermosensitive receptors found in neuronal and nonneuronal tissues, triggering the release of vasoactive neuropeptides.1 Among these, transient receptor potential (TRP) channels seem to play a crucial role in neurovascular reactivity and have been studied in the pathophysiology of rosacea.1,8 Overexpression or excessive stimulation of TRPs by various environmental triggers, such as heat or cold, leads to increased neuropeptide production, ultimately contributing to persistent erythema and vascular dysfunction, as well as a burning or stinging sensation.1,2 Moreover, rapid temperature changes, such as moving from freezing outdoor conditions into a heated environment, may also trigger flushing due to sudden vasodilation.2
Adopting behavioral strategies such as preventing overheating, minimizing sudden temperature shifts, and protecting the skin from cold can help reduce rosacea flare-ups, particularly flushing. For patients who do not achieve sufficient relief through lifestyle modifications alone, targeted pharmacologic treatments are available to help manage these symptoms. Topical α-adrenergic agonists (eg, brimonidine, oxymetazoline) are effective in reducing erythema and flushing by causing vasoconstriction.15,19 For persistent erythema and telangiectasias, pulsed dye laser and intense pulsed light therapies can be effective treatments, as they target hemoglobin in blood vessels, leading to their destruction and a subsequent reduction in erythema.20 Other medications such as topical metronidazole, azelaic acid, calcitonin-gene related peptide inhibitors, and systemic ß-blockers also can be used to treat flushing and redness.15,21
Skin Care Practices
Due to the increased tissue inflammation and potential skin barrier dysfunction, rosacea-affected skin is highly sensitive, and skin care practices or products that disrupt the already compromised skin barrier can contribute to flare-ups. General recommendations should include use of gentle cleansers and moisturizers to prevent dry skin and improve skin barrier function22 as well as avoidance of ingredients that are common irritants and inducers of allergic contact dermatitis (eg, fragrances).9
Cleansing the face should be limited to 1 to 2 times daily, as excessive cleansing and use of harsh formulations with exfoliative ingredients can lead to skin irritation and worsening of symptoms.9 Overcleansing can lead to alterations in cutaneous pH and strip the stratum corneum of healthy components such as lipids and natural moisturizing factors. Common ingredients in cleansers that should be avoided due to their irritant nature include alcohol, acetone, benzyl alcohol, propylene glycol, and α- and ß-hydroxy acids. Instead, syndet (synthetic detergent) cleansers that contain ceramides, hyaluronic acid, or other hydrating agents with a near-physiological pH can be helpful for dry and sensitive skin.23 Toners with high alcohol content and astringent-based products also should be avoided.
Optimal moisturizers for rosacea-affected skin should contain physiologic lipids that help replace a healthy skin barrier as well as relieve dryness and seal in moisture. Beneficial barrier-restoring ingredients include ceramides, dimethicone, cholesterol, and free fatty acids as well as humectants such as glycerin and hyaluronic acid.9,23,24 Applying moisturizer immediately after cleansing and prior to the application of any topical treatments also can help decrease irritation.
As mentioned previously, sun protection is a cornerstone in the management of rosacea and can help reduce redness and skin irritation. Using combination formulas, such as moisturizers with a sun protection factor of at least 50, can be effective.25 Additionally, products with antioxidant or anti-inflammatory ingredients such as niacinamide and allantoin can further support skin health. Lastly, formulations containing green pigments may also be beneficial, as they provide cosmetic camouflage to neutralize redness.26
Dietary Factors
Several dietary factors have been proposed as triggers for rosacea, but conclusive evidence remains limited.27 Foods and beverages that generate heat (eg, hot drinks, spicy foods) may exacerbate rosacea by causing vasodilation and stimulating TRP channels, resulting in flushing.18 While capsaicin, found in spicy foods, may lead to flushing through similar activation of TRP channels, current evidence has not proved a specific and consistent role in the pathogenesis of rosacea.18,27 Similarly, cinnamaldehyde, found in cinnamon and many commercial cinnamon-containing foods as well as various fruits and vegetables, activates thermosensitive receptors that may worsen rosacea symptoms.28 Other potential triggers include histamine-rich foods (eg, avocados, bananas, dried fruits, nuts, smoked fish, aged cheeses), which can lead to skin hypersensitivity and flushing, and formaldehyde-containing foods (eg, apples, carrots, cauliflower, shiitake mushrooms, fish), though the role these types of foods play in rosacea remains unclear.1,29-31
The relationship between caffeine and rosacea is complex. While caffeine commonly is found in coffee, tea, and soda, some studies have suggested that coffee consumption may reduce rosacea risk due to its vasoconstrictive and anti-inflammatory effects.28,32 In contrast, alcohol—particularly white wine and liquor—has been associated with increased rosacea risk due to its effect on vasodilation, inflammation, and oxidative stress.33 Despite anecdotal reports, the role of dairy products in rosacea remains unclear, with conflicting studies suggesting dairy consumption may exacerbate or protect against rosacea.27,28 Given the variability in dietary triggers, patients with rosacea may benefit from using a dietary journal to identify and avoid foods that exacerbate their symptoms, though more research is needed to establish clear recommendations.
Conclusion
Rosacea is a complex condition influenced by genetic, immune, microbial, and environmental factors. Triggers such as UV exposure, temperature fluctuations, alterations in the skin microbiome, and diet contribute to disease exacerbation through mechanisms like vasodilation, neurogenic inflammation, and immune dysregulation. These triggers often interact, compounding their effects and making symptom management more challenging and multifaceted.
Successful rosacea treatment relies on identifying and minimizing patient-specific triggers, as lifestyle modifications can reduce flare-ups and improve outcomes. When combined with interventional, oral, and topical therapies, these adjustments enhance treatment effectiveness and contribute to better long-term disease control. Clinicians should adopt a personalized holistic approach by educating patients on common triggers, recommending lifestyle changes, and integrating medical treatments as necessary. Future research should continue exploring the relationships between rosacea and environmental factors to develop more targeted and evidence-based recommendations.
- Steinhoff M, Schauber J, Leyden JJ. New insights into rosacea pathophysiology: a review of recent findings. J Am Acad Dermatol. 2013;69(6 suppl 1):S15-S26.
- Buddenkotte J, Steinhoff M. Recent advances in understanding and managing rosacea. F1000Res. 2018;7:F1000 Faculty Rev-1885.
- Gether L, Overgaard LK, Egeberg A, et al. Incidence and prevalence of rosacea: a systematic review and meta-analysis. Br J Dermatol. 2018;179:282-289. doi:10.1111/bjd.16481
- Chamaillard M, Mortemousque B, Boralevi F, et al. Cutaneous and ocular signs of childhood rosacea. Arch Dermatol. 2008;144:167-171.
- Abram K, Silm H, Maaroos H, et al. Risk factors associated with rosacea. J Eur Acad Dermatol Venereol. 2010;24:565-571.
- Gerber PA, Buhren BA, Steinhoff M, et al. Rosacea: the cytokine and chemokine network. J Investig Dermatol Symp Proc. 2011;15:40-47.
- Steinhoff M, Buddenkotte J, Aubert J, et al. Clinical, cellular, and molecular aspects in the pathophysiology of rosacea. J Investig Dermatol Symp Proc. 2011;15:2-11.
- Two AM, Wu W, Gallo RL, et al. Rosacea. J Am Acad Dermatol. 2015;72:749-758.
- Morgado‐Carrasco D, Granger C, Trullas C, et al. Impact of ultraviolet radiation and exposome on rosacea: key role of photoprotection in optimizing treatment. J Cosmet Dermatol. 2021;20:3415-3421.
- Suhng E, Kim BH, Choi YW, et al. Increased expression of IL‐33 in rosacea skin and UVB‐irradiated and LL‐37‐treated HaCaT cells. Exp Dermatol. 2018;27:1023-1029.
- Tisma VS, Basta-Juzbasic A, Jaganjac M, et al. Oxidative stress and ferritin expression in the skin of patients with rosacea. J Am Acad Dermatol. 2009;60:270-276.
- Kulkarni NN, Takahashi T, Sanford JA, et al. Innate immune dysfunction in rosacea promotes photosensitivity and vascular adhesion molecule expression. J Invest Dermatol. 2020;140:645-655.E6.
- Bae YI, Yun SJ, Lee JB, et al. Clinical evaluation of 168 Korean patients with rosacea: the sun exposure correlates with the erythematotelangiectatic subtype. Ann Dermatol. 2009;21:243-249.
- McAleer, MA, Fitzpatrick P, Powell FC. Papulopustular rosacea: prevalence and relationship to photodamage. J Am Acad Dermatol. 2010;63:33-39.
- Van Zuuren EJ. Rosacea. N Engl J Med. 2017;377:1754-1764.
- Two AM, Wu W, Gallo RL, et al. Rosacea. J Am Acad Dermatol. 2015;72:761-770.
- Nichols K, Desai N, Lebwohl MG. Effective sunscreen ingredients and cutaneous irritation in patients with rosacea. Cutis. 1998;61:344-346.
- Guzman-Sanchez DA, Ishiuji Y, Patel T, et al. Enhanced skin blood flow and sensitivity to noxious heat stimuli in papulopustular rosacea. J Am Acad Dermatol. 2007;57:800-805.
- Fowler J Jr, Jackson M, Moore A, et al. Efficacy and safety of once-daily topical brimonidine tartrate gel 0.5% for the treatment of moderate to severe facial erythema of rosacea: results of two randomized, double-blind, and vehicle-controlled pivotal studies. J Drugs Dermatol. 2013;12:650-656.
- van Zuuren EJ, Fedorowicz Z, Tan J, et al. Interventions for rosacea based on the phenotype approach: an updated systematic review including GRADE assessments. Br J Dermatol.2019;181:65-79.
- Wienholtz NKF, Christensen CE, Do TP, et al. Erenumab for treatment of persistent erythema and flushing in rosacea: a nonrandomized controlled trial. JAMA Dermatol.2024;160:612-619.
- Del Rosso JQ, Thiboutot D, Gallo R, et al. Consensus recommendations from the American Acne & Rosacea Society on the management of rosacea, part 1: a status report on the disease state, general measures, and adjunctive skin care. Cutis. 2013;92:234-240.
- Baldwin H, Alexis AF, Andriessen A, et al. Evidence of barrier deficiency in rosacea and the importance of integrating OTC skincare products into treatment regimens. J Drugs Dermatol. 2021;20:384-392.
- Schlesinger TE, Powell CR. Efficacy and tolerability of low molecular weight hyaluronic acid sodium salt 0.2% cream in rosacea. J Drugs Dermatol. 2013;12:664-667.
- Williams JD, Maitra P, Atillasoy E, et al. SPF 100+ sunscreen is more protective against sunburn than SPF 50+ in actual use: results of a randomized, double-blind, split-face, natural sunlight exposure clinical trial. J Am Acad Dermatol. 2018;78:902-910.E2.
- Draelos ZD. Cosmeceuticals for rosacea. Clin Dermatol. 2017;35:213-217.
- Yuan X, Huang X, Wang B, et al. Relationship between rosacea and dietary factors: a multicenter retrospective case–control survey. J Dermatol. 2019;46:219-225.
- Alia E, Feng H. Rosacea pathogenesis, common triggers, and dietary role: the cause, the trigger, and the positive effects of different foods. Clin Dermatol. 2022;40:122-127.
- Branco ACCC, Yoshikawa FSY, Pietrobon AJ, et al. Role of histamine in modulating the immune response and inflammation. Mediators Inflamm. 2018;2018:1-10.
- Darrigade A, Dendooven E, Aerts O. Contact allergy to fragrances and formaldehyde contributing to papulopustular rosacea. Contact Dermatitis. 2019;81:395-397.
- Linauskiene K, Isaksson M. Allergic contact dermatitis from formaldehyde mimicking impetigo and initiating rosacea. Contact Dermatitis. 2018;78:359-361.
- Al Reef T, Ghanem E. Caffeine: well-known as psychotropic substance, but little as immunomodulator. Immunobiology. 2018;223:818-825.
- Drago F, Ciccarese G, Herzum A, et al. Rosacea and alcohol intake. J Am Acad Dermatol. 2018;78:E25.
Rosacea is a chronic inflammatory skin disease characterized by erythema, flushing, telangiectasias, papules, pustules, and rarely, phymatous changes that primarily manifest in a centrofacial distribution.1,2 Although establishing the true prevalence of rosacea may be challenging due to a wide spectrum of clinical manifestations, current studies estimate that it is between 5% to 6% of the global adult population and that rosacea most commonly is diagnosed in patients aged 30 and 60 years, though it occasionally can affect adolescents and children.3,4 Although the origin and pathophysiology of rosacea remain incompletely understood, the condition arises from a complex interplay of genetic, environmental, immune, microbial, and neurovascular factors; this interplay ultimately leads to excessive production of inflammatory and vasoactive peptides, chronic inflammation, and neurovascular hyperreactivity.1,5-7
Identifying triggers can be valuable in managing rosacea, as avoidance of these exposures may lead to disease improvement. In this review, we highlight 4 major environmental triggers of rosacea—UV radiation exposure, temperature fluctuation, skin care practices, and diet—and their roles in its pathogenesis and management. A high-level summary of recommendations can be found in the Table.

UV Radiation Exposure
Exposure to UV radiation is a known trigger of rosacea and may worsen symptoms through several mechanisms.8,9 It increases the production of inflammatory cytokines, which enhance the release of vascular endothelial growth factor, promoting angiogenesis and vasodilation.10 Exposure to UV radiation also contributes to tissue inflammation through the production of reactive oxygen species, further mediating inflammatory cascades and leading to immune dysregulation.11,12 Interestingly, though the mechanisms by which UV radiation may contribute to the pathophysiology of rosacea are well described, it remains unclear whether chronic UV exposure plays a major role in the pathogenesis or disease progression of rosacea.1 Studies have observed that increased exposure to sunlight seems to be correlated with increased severity of redness but not of papules and pustules.13,14
Despite some uncertainty regarding the relationship between rosacea and chronic UV exposure, sun protection is a prudent recommendation in this patient population, particularly given other risks of exposure to UV radiation, such as photoaging and skin cancer.9,15,16 Sun protection can be accomplished using broad-spectrum sunscreen (sun protection factor 50 or higher, reapplied every 2 to 4 hours) or by wearing physical protection (eg, hats, sun-protective clothing) along with avoidance of sun exposure during peak UV hours (ie, 10 am-
Temperature Fluctuation
Both heat and cold exposure have been suggested as triggers for rosacea, thought to be mediated through dysregulations in neurovascular and thermal pathways, resulting in increased flushing and erythema.6 Skin affected by rosacea exhibits a lower threshold for temperature and pain stimuli, resulting in heightened hypersensitivity compared to normal skin.18 Exposure to heat activates thermosensitive receptors found in neuronal and nonneuronal tissues, triggering the release of vasoactive neuropeptides.1 Among these, transient receptor potential (TRP) channels seem to play a crucial role in neurovascular reactivity and have been studied in the pathophysiology of rosacea.1,8 Overexpression or excessive stimulation of TRPs by various environmental triggers, such as heat or cold, leads to increased neuropeptide production, ultimately contributing to persistent erythema and vascular dysfunction, as well as a burning or stinging sensation.1,2 Moreover, rapid temperature changes, such as moving from freezing outdoor conditions into a heated environment, may also trigger flushing due to sudden vasodilation.2
Adopting behavioral strategies such as preventing overheating, minimizing sudden temperature shifts, and protecting the skin from cold can help reduce rosacea flare-ups, particularly flushing. For patients who do not achieve sufficient relief through lifestyle modifications alone, targeted pharmacologic treatments are available to help manage these symptoms. Topical α-adrenergic agonists (eg, brimonidine, oxymetazoline) are effective in reducing erythema and flushing by causing vasoconstriction.15,19 For persistent erythema and telangiectasias, pulsed dye laser and intense pulsed light therapies can be effective treatments, as they target hemoglobin in blood vessels, leading to their destruction and a subsequent reduction in erythema.20 Other medications such as topical metronidazole, azelaic acid, calcitonin-gene related peptide inhibitors, and systemic ß-blockers also can be used to treat flushing and redness.15,21
Skin Care Practices
Due to the increased tissue inflammation and potential skin barrier dysfunction, rosacea-affected skin is highly sensitive, and skin care practices or products that disrupt the already compromised skin barrier can contribute to flare-ups. General recommendations should include use of gentle cleansers and moisturizers to prevent dry skin and improve skin barrier function22 as well as avoidance of ingredients that are common irritants and inducers of allergic contact dermatitis (eg, fragrances).9
Cleansing the face should be limited to 1 to 2 times daily, as excessive cleansing and use of harsh formulations with exfoliative ingredients can lead to skin irritation and worsening of symptoms.9 Overcleansing can lead to alterations in cutaneous pH and strip the stratum corneum of healthy components such as lipids and natural moisturizing factors. Common ingredients in cleansers that should be avoided due to their irritant nature include alcohol, acetone, benzyl alcohol, propylene glycol, and α- and ß-hydroxy acids. Instead, syndet (synthetic detergent) cleansers that contain ceramides, hyaluronic acid, or other hydrating agents with a near-physiological pH can be helpful for dry and sensitive skin.23 Toners with high alcohol content and astringent-based products also should be avoided.
Optimal moisturizers for rosacea-affected skin should contain physiologic lipids that help replace a healthy skin barrier as well as relieve dryness and seal in moisture. Beneficial barrier-restoring ingredients include ceramides, dimethicone, cholesterol, and free fatty acids as well as humectants such as glycerin and hyaluronic acid.9,23,24 Applying moisturizer immediately after cleansing and prior to the application of any topical treatments also can help decrease irritation.
As mentioned previously, sun protection is a cornerstone in the management of rosacea and can help reduce redness and skin irritation. Using combination formulas, such as moisturizers with a sun protection factor of at least 50, can be effective.25 Additionally, products with antioxidant or anti-inflammatory ingredients such as niacinamide and allantoin can further support skin health. Lastly, formulations containing green pigments may also be beneficial, as they provide cosmetic camouflage to neutralize redness.26
Dietary Factors
Several dietary factors have been proposed as triggers for rosacea, but conclusive evidence remains limited.27 Foods and beverages that generate heat (eg, hot drinks, spicy foods) may exacerbate rosacea by causing vasodilation and stimulating TRP channels, resulting in flushing.18 While capsaicin, found in spicy foods, may lead to flushing through similar activation of TRP channels, current evidence has not proved a specific and consistent role in the pathogenesis of rosacea.18,27 Similarly, cinnamaldehyde, found in cinnamon and many commercial cinnamon-containing foods as well as various fruits and vegetables, activates thermosensitive receptors that may worsen rosacea symptoms.28 Other potential triggers include histamine-rich foods (eg, avocados, bananas, dried fruits, nuts, smoked fish, aged cheeses), which can lead to skin hypersensitivity and flushing, and formaldehyde-containing foods (eg, apples, carrots, cauliflower, shiitake mushrooms, fish), though the role these types of foods play in rosacea remains unclear.1,29-31
The relationship between caffeine and rosacea is complex. While caffeine commonly is found in coffee, tea, and soda, some studies have suggested that coffee consumption may reduce rosacea risk due to its vasoconstrictive and anti-inflammatory effects.28,32 In contrast, alcohol—particularly white wine and liquor—has been associated with increased rosacea risk due to its effect on vasodilation, inflammation, and oxidative stress.33 Despite anecdotal reports, the role of dairy products in rosacea remains unclear, with conflicting studies suggesting dairy consumption may exacerbate or protect against rosacea.27,28 Given the variability in dietary triggers, patients with rosacea may benefit from using a dietary journal to identify and avoid foods that exacerbate their symptoms, though more research is needed to establish clear recommendations.
Conclusion
Rosacea is a complex condition influenced by genetic, immune, microbial, and environmental factors. Triggers such as UV exposure, temperature fluctuations, alterations in the skin microbiome, and diet contribute to disease exacerbation through mechanisms like vasodilation, neurogenic inflammation, and immune dysregulation. These triggers often interact, compounding their effects and making symptom management more challenging and multifaceted.
Successful rosacea treatment relies on identifying and minimizing patient-specific triggers, as lifestyle modifications can reduce flare-ups and improve outcomes. When combined with interventional, oral, and topical therapies, these adjustments enhance treatment effectiveness and contribute to better long-term disease control. Clinicians should adopt a personalized holistic approach by educating patients on common triggers, recommending lifestyle changes, and integrating medical treatments as necessary. Future research should continue exploring the relationships between rosacea and environmental factors to develop more targeted and evidence-based recommendations.
Rosacea is a chronic inflammatory skin disease characterized by erythema, flushing, telangiectasias, papules, pustules, and rarely, phymatous changes that primarily manifest in a centrofacial distribution.1,2 Although establishing the true prevalence of rosacea may be challenging due to a wide spectrum of clinical manifestations, current studies estimate that it is between 5% to 6% of the global adult population and that rosacea most commonly is diagnosed in patients aged 30 and 60 years, though it occasionally can affect adolescents and children.3,4 Although the origin and pathophysiology of rosacea remain incompletely understood, the condition arises from a complex interplay of genetic, environmental, immune, microbial, and neurovascular factors; this interplay ultimately leads to excessive production of inflammatory and vasoactive peptides, chronic inflammation, and neurovascular hyperreactivity.1,5-7
Identifying triggers can be valuable in managing rosacea, as avoidance of these exposures may lead to disease improvement. In this review, we highlight 4 major environmental triggers of rosacea—UV radiation exposure, temperature fluctuation, skin care practices, and diet—and their roles in its pathogenesis and management. A high-level summary of recommendations can be found in the Table.

UV Radiation Exposure
Exposure to UV radiation is a known trigger of rosacea and may worsen symptoms through several mechanisms.8,9 It increases the production of inflammatory cytokines, which enhance the release of vascular endothelial growth factor, promoting angiogenesis and vasodilation.10 Exposure to UV radiation also contributes to tissue inflammation through the production of reactive oxygen species, further mediating inflammatory cascades and leading to immune dysregulation.11,12 Interestingly, though the mechanisms by which UV radiation may contribute to the pathophysiology of rosacea are well described, it remains unclear whether chronic UV exposure plays a major role in the pathogenesis or disease progression of rosacea.1 Studies have observed that increased exposure to sunlight seems to be correlated with increased severity of redness but not of papules and pustules.13,14
Despite some uncertainty regarding the relationship between rosacea and chronic UV exposure, sun protection is a prudent recommendation in this patient population, particularly given other risks of exposure to UV radiation, such as photoaging and skin cancer.9,15,16 Sun protection can be accomplished using broad-spectrum sunscreen (sun protection factor 50 or higher, reapplied every 2 to 4 hours) or by wearing physical protection (eg, hats, sun-protective clothing) along with avoidance of sun exposure during peak UV hours (ie, 10 am-
Temperature Fluctuation
Both heat and cold exposure have been suggested as triggers for rosacea, thought to be mediated through dysregulations in neurovascular and thermal pathways, resulting in increased flushing and erythema.6 Skin affected by rosacea exhibits a lower threshold for temperature and pain stimuli, resulting in heightened hypersensitivity compared to normal skin.18 Exposure to heat activates thermosensitive receptors found in neuronal and nonneuronal tissues, triggering the release of vasoactive neuropeptides.1 Among these, transient receptor potential (TRP) channels seem to play a crucial role in neurovascular reactivity and have been studied in the pathophysiology of rosacea.1,8 Overexpression or excessive stimulation of TRPs by various environmental triggers, such as heat or cold, leads to increased neuropeptide production, ultimately contributing to persistent erythema and vascular dysfunction, as well as a burning or stinging sensation.1,2 Moreover, rapid temperature changes, such as moving from freezing outdoor conditions into a heated environment, may also trigger flushing due to sudden vasodilation.2
Adopting behavioral strategies such as preventing overheating, minimizing sudden temperature shifts, and protecting the skin from cold can help reduce rosacea flare-ups, particularly flushing. For patients who do not achieve sufficient relief through lifestyle modifications alone, targeted pharmacologic treatments are available to help manage these symptoms. Topical α-adrenergic agonists (eg, brimonidine, oxymetazoline) are effective in reducing erythema and flushing by causing vasoconstriction.15,19 For persistent erythema and telangiectasias, pulsed dye laser and intense pulsed light therapies can be effective treatments, as they target hemoglobin in blood vessels, leading to their destruction and a subsequent reduction in erythema.20 Other medications such as topical metronidazole, azelaic acid, calcitonin-gene related peptide inhibitors, and systemic ß-blockers also can be used to treat flushing and redness.15,21
Skin Care Practices
Due to the increased tissue inflammation and potential skin barrier dysfunction, rosacea-affected skin is highly sensitive, and skin care practices or products that disrupt the already compromised skin barrier can contribute to flare-ups. General recommendations should include use of gentle cleansers and moisturizers to prevent dry skin and improve skin barrier function22 as well as avoidance of ingredients that are common irritants and inducers of allergic contact dermatitis (eg, fragrances).9
Cleansing the face should be limited to 1 to 2 times daily, as excessive cleansing and use of harsh formulations with exfoliative ingredients can lead to skin irritation and worsening of symptoms.9 Overcleansing can lead to alterations in cutaneous pH and strip the stratum corneum of healthy components such as lipids and natural moisturizing factors. Common ingredients in cleansers that should be avoided due to their irritant nature include alcohol, acetone, benzyl alcohol, propylene glycol, and α- and ß-hydroxy acids. Instead, syndet (synthetic detergent) cleansers that contain ceramides, hyaluronic acid, or other hydrating agents with a near-physiological pH can be helpful for dry and sensitive skin.23 Toners with high alcohol content and astringent-based products also should be avoided.
Optimal moisturizers for rosacea-affected skin should contain physiologic lipids that help replace a healthy skin barrier as well as relieve dryness and seal in moisture. Beneficial barrier-restoring ingredients include ceramides, dimethicone, cholesterol, and free fatty acids as well as humectants such as glycerin and hyaluronic acid.9,23,24 Applying moisturizer immediately after cleansing and prior to the application of any topical treatments also can help decrease irritation.
As mentioned previously, sun protection is a cornerstone in the management of rosacea and can help reduce redness and skin irritation. Using combination formulas, such as moisturizers with a sun protection factor of at least 50, can be effective.25 Additionally, products with antioxidant or anti-inflammatory ingredients such as niacinamide and allantoin can further support skin health. Lastly, formulations containing green pigments may also be beneficial, as they provide cosmetic camouflage to neutralize redness.26
Dietary Factors
Several dietary factors have been proposed as triggers for rosacea, but conclusive evidence remains limited.27 Foods and beverages that generate heat (eg, hot drinks, spicy foods) may exacerbate rosacea by causing vasodilation and stimulating TRP channels, resulting in flushing.18 While capsaicin, found in spicy foods, may lead to flushing through similar activation of TRP channels, current evidence has not proved a specific and consistent role in the pathogenesis of rosacea.18,27 Similarly, cinnamaldehyde, found in cinnamon and many commercial cinnamon-containing foods as well as various fruits and vegetables, activates thermosensitive receptors that may worsen rosacea symptoms.28 Other potential triggers include histamine-rich foods (eg, avocados, bananas, dried fruits, nuts, smoked fish, aged cheeses), which can lead to skin hypersensitivity and flushing, and formaldehyde-containing foods (eg, apples, carrots, cauliflower, shiitake mushrooms, fish), though the role these types of foods play in rosacea remains unclear.1,29-31
The relationship between caffeine and rosacea is complex. While caffeine commonly is found in coffee, tea, and soda, some studies have suggested that coffee consumption may reduce rosacea risk due to its vasoconstrictive and anti-inflammatory effects.28,32 In contrast, alcohol—particularly white wine and liquor—has been associated with increased rosacea risk due to its effect on vasodilation, inflammation, and oxidative stress.33 Despite anecdotal reports, the role of dairy products in rosacea remains unclear, with conflicting studies suggesting dairy consumption may exacerbate or protect against rosacea.27,28 Given the variability in dietary triggers, patients with rosacea may benefit from using a dietary journal to identify and avoid foods that exacerbate their symptoms, though more research is needed to establish clear recommendations.
Conclusion
Rosacea is a complex condition influenced by genetic, immune, microbial, and environmental factors. Triggers such as UV exposure, temperature fluctuations, alterations in the skin microbiome, and diet contribute to disease exacerbation through mechanisms like vasodilation, neurogenic inflammation, and immune dysregulation. These triggers often interact, compounding their effects and making symptom management more challenging and multifaceted.
Successful rosacea treatment relies on identifying and minimizing patient-specific triggers, as lifestyle modifications can reduce flare-ups and improve outcomes. When combined with interventional, oral, and topical therapies, these adjustments enhance treatment effectiveness and contribute to better long-term disease control. Clinicians should adopt a personalized holistic approach by educating patients on common triggers, recommending lifestyle changes, and integrating medical treatments as necessary. Future research should continue exploring the relationships between rosacea and environmental factors to develop more targeted and evidence-based recommendations.
- Steinhoff M, Schauber J, Leyden JJ. New insights into rosacea pathophysiology: a review of recent findings. J Am Acad Dermatol. 2013;69(6 suppl 1):S15-S26.
- Buddenkotte J, Steinhoff M. Recent advances in understanding and managing rosacea. F1000Res. 2018;7:F1000 Faculty Rev-1885.
- Gether L, Overgaard LK, Egeberg A, et al. Incidence and prevalence of rosacea: a systematic review and meta-analysis. Br J Dermatol. 2018;179:282-289. doi:10.1111/bjd.16481
- Chamaillard M, Mortemousque B, Boralevi F, et al. Cutaneous and ocular signs of childhood rosacea. Arch Dermatol. 2008;144:167-171.
- Abram K, Silm H, Maaroos H, et al. Risk factors associated with rosacea. J Eur Acad Dermatol Venereol. 2010;24:565-571.
- Gerber PA, Buhren BA, Steinhoff M, et al. Rosacea: the cytokine and chemokine network. J Investig Dermatol Symp Proc. 2011;15:40-47.
- Steinhoff M, Buddenkotte J, Aubert J, et al. Clinical, cellular, and molecular aspects in the pathophysiology of rosacea. J Investig Dermatol Symp Proc. 2011;15:2-11.
- Two AM, Wu W, Gallo RL, et al. Rosacea. J Am Acad Dermatol. 2015;72:749-758.
- Morgado‐Carrasco D, Granger C, Trullas C, et al. Impact of ultraviolet radiation and exposome on rosacea: key role of photoprotection in optimizing treatment. J Cosmet Dermatol. 2021;20:3415-3421.
- Suhng E, Kim BH, Choi YW, et al. Increased expression of IL‐33 in rosacea skin and UVB‐irradiated and LL‐37‐treated HaCaT cells. Exp Dermatol. 2018;27:1023-1029.
- Tisma VS, Basta-Juzbasic A, Jaganjac M, et al. Oxidative stress and ferritin expression in the skin of patients with rosacea. J Am Acad Dermatol. 2009;60:270-276.
- Kulkarni NN, Takahashi T, Sanford JA, et al. Innate immune dysfunction in rosacea promotes photosensitivity and vascular adhesion molecule expression. J Invest Dermatol. 2020;140:645-655.E6.
- Bae YI, Yun SJ, Lee JB, et al. Clinical evaluation of 168 Korean patients with rosacea: the sun exposure correlates with the erythematotelangiectatic subtype. Ann Dermatol. 2009;21:243-249.
- McAleer, MA, Fitzpatrick P, Powell FC. Papulopustular rosacea: prevalence and relationship to photodamage. J Am Acad Dermatol. 2010;63:33-39.
- Van Zuuren EJ. Rosacea. N Engl J Med. 2017;377:1754-1764.
- Two AM, Wu W, Gallo RL, et al. Rosacea. J Am Acad Dermatol. 2015;72:761-770.
- Nichols K, Desai N, Lebwohl MG. Effective sunscreen ingredients and cutaneous irritation in patients with rosacea. Cutis. 1998;61:344-346.
- Guzman-Sanchez DA, Ishiuji Y, Patel T, et al. Enhanced skin blood flow and sensitivity to noxious heat stimuli in papulopustular rosacea. J Am Acad Dermatol. 2007;57:800-805.
- Fowler J Jr, Jackson M, Moore A, et al. Efficacy and safety of once-daily topical brimonidine tartrate gel 0.5% for the treatment of moderate to severe facial erythema of rosacea: results of two randomized, double-blind, and vehicle-controlled pivotal studies. J Drugs Dermatol. 2013;12:650-656.
- van Zuuren EJ, Fedorowicz Z, Tan J, et al. Interventions for rosacea based on the phenotype approach: an updated systematic review including GRADE assessments. Br J Dermatol.2019;181:65-79.
- Wienholtz NKF, Christensen CE, Do TP, et al. Erenumab for treatment of persistent erythema and flushing in rosacea: a nonrandomized controlled trial. JAMA Dermatol.2024;160:612-619.
- Del Rosso JQ, Thiboutot D, Gallo R, et al. Consensus recommendations from the American Acne & Rosacea Society on the management of rosacea, part 1: a status report on the disease state, general measures, and adjunctive skin care. Cutis. 2013;92:234-240.
- Baldwin H, Alexis AF, Andriessen A, et al. Evidence of barrier deficiency in rosacea and the importance of integrating OTC skincare products into treatment regimens. J Drugs Dermatol. 2021;20:384-392.
- Schlesinger TE, Powell CR. Efficacy and tolerability of low molecular weight hyaluronic acid sodium salt 0.2% cream in rosacea. J Drugs Dermatol. 2013;12:664-667.
- Williams JD, Maitra P, Atillasoy E, et al. SPF 100+ sunscreen is more protective against sunburn than SPF 50+ in actual use: results of a randomized, double-blind, split-face, natural sunlight exposure clinical trial. J Am Acad Dermatol. 2018;78:902-910.E2.
- Draelos ZD. Cosmeceuticals for rosacea. Clin Dermatol. 2017;35:213-217.
- Yuan X, Huang X, Wang B, et al. Relationship between rosacea and dietary factors: a multicenter retrospective case–control survey. J Dermatol. 2019;46:219-225.
- Alia E, Feng H. Rosacea pathogenesis, common triggers, and dietary role: the cause, the trigger, and the positive effects of different foods. Clin Dermatol. 2022;40:122-127.
- Branco ACCC, Yoshikawa FSY, Pietrobon AJ, et al. Role of histamine in modulating the immune response and inflammation. Mediators Inflamm. 2018;2018:1-10.
- Darrigade A, Dendooven E, Aerts O. Contact allergy to fragrances and formaldehyde contributing to papulopustular rosacea. Contact Dermatitis. 2019;81:395-397.
- Linauskiene K, Isaksson M. Allergic contact dermatitis from formaldehyde mimicking impetigo and initiating rosacea. Contact Dermatitis. 2018;78:359-361.
- Al Reef T, Ghanem E. Caffeine: well-known as psychotropic substance, but little as immunomodulator. Immunobiology. 2018;223:818-825.
- Drago F, Ciccarese G, Herzum A, et al. Rosacea and alcohol intake. J Am Acad Dermatol. 2018;78:E25.
- Steinhoff M, Schauber J, Leyden JJ. New insights into rosacea pathophysiology: a review of recent findings. J Am Acad Dermatol. 2013;69(6 suppl 1):S15-S26.
- Buddenkotte J, Steinhoff M. Recent advances in understanding and managing rosacea. F1000Res. 2018;7:F1000 Faculty Rev-1885.
- Gether L, Overgaard LK, Egeberg A, et al. Incidence and prevalence of rosacea: a systematic review and meta-analysis. Br J Dermatol. 2018;179:282-289. doi:10.1111/bjd.16481
- Chamaillard M, Mortemousque B, Boralevi F, et al. Cutaneous and ocular signs of childhood rosacea. Arch Dermatol. 2008;144:167-171.
- Abram K, Silm H, Maaroos H, et al. Risk factors associated with rosacea. J Eur Acad Dermatol Venereol. 2010;24:565-571.
- Gerber PA, Buhren BA, Steinhoff M, et al. Rosacea: the cytokine and chemokine network. J Investig Dermatol Symp Proc. 2011;15:40-47.
- Steinhoff M, Buddenkotte J, Aubert J, et al. Clinical, cellular, and molecular aspects in the pathophysiology of rosacea. J Investig Dermatol Symp Proc. 2011;15:2-11.
- Two AM, Wu W, Gallo RL, et al. Rosacea. J Am Acad Dermatol. 2015;72:749-758.
- Morgado‐Carrasco D, Granger C, Trullas C, et al. Impact of ultraviolet radiation and exposome on rosacea: key role of photoprotection in optimizing treatment. J Cosmet Dermatol. 2021;20:3415-3421.
- Suhng E, Kim BH, Choi YW, et al. Increased expression of IL‐33 in rosacea skin and UVB‐irradiated and LL‐37‐treated HaCaT cells. Exp Dermatol. 2018;27:1023-1029.
- Tisma VS, Basta-Juzbasic A, Jaganjac M, et al. Oxidative stress and ferritin expression in the skin of patients with rosacea. J Am Acad Dermatol. 2009;60:270-276.
- Kulkarni NN, Takahashi T, Sanford JA, et al. Innate immune dysfunction in rosacea promotes photosensitivity and vascular adhesion molecule expression. J Invest Dermatol. 2020;140:645-655.E6.
- Bae YI, Yun SJ, Lee JB, et al. Clinical evaluation of 168 Korean patients with rosacea: the sun exposure correlates with the erythematotelangiectatic subtype. Ann Dermatol. 2009;21:243-249.
- McAleer, MA, Fitzpatrick P, Powell FC. Papulopustular rosacea: prevalence and relationship to photodamage. J Am Acad Dermatol. 2010;63:33-39.
- Van Zuuren EJ. Rosacea. N Engl J Med. 2017;377:1754-1764.
- Two AM, Wu W, Gallo RL, et al. Rosacea. J Am Acad Dermatol. 2015;72:761-770.
- Nichols K, Desai N, Lebwohl MG. Effective sunscreen ingredients and cutaneous irritation in patients with rosacea. Cutis. 1998;61:344-346.
- Guzman-Sanchez DA, Ishiuji Y, Patel T, et al. Enhanced skin blood flow and sensitivity to noxious heat stimuli in papulopustular rosacea. J Am Acad Dermatol. 2007;57:800-805.
- Fowler J Jr, Jackson M, Moore A, et al. Efficacy and safety of once-daily topical brimonidine tartrate gel 0.5% for the treatment of moderate to severe facial erythema of rosacea: results of two randomized, double-blind, and vehicle-controlled pivotal studies. J Drugs Dermatol. 2013;12:650-656.
- van Zuuren EJ, Fedorowicz Z, Tan J, et al. Interventions for rosacea based on the phenotype approach: an updated systematic review including GRADE assessments. Br J Dermatol.2019;181:65-79.
- Wienholtz NKF, Christensen CE, Do TP, et al. Erenumab for treatment of persistent erythema and flushing in rosacea: a nonrandomized controlled trial. JAMA Dermatol.2024;160:612-619.
- Del Rosso JQ, Thiboutot D, Gallo R, et al. Consensus recommendations from the American Acne & Rosacea Society on the management of rosacea, part 1: a status report on the disease state, general measures, and adjunctive skin care. Cutis. 2013;92:234-240.
- Baldwin H, Alexis AF, Andriessen A, et al. Evidence of barrier deficiency in rosacea and the importance of integrating OTC skincare products into treatment regimens. J Drugs Dermatol. 2021;20:384-392.
- Schlesinger TE, Powell CR. Efficacy and tolerability of low molecular weight hyaluronic acid sodium salt 0.2% cream in rosacea. J Drugs Dermatol. 2013;12:664-667.
- Williams JD, Maitra P, Atillasoy E, et al. SPF 100+ sunscreen is more protective against sunburn than SPF 50+ in actual use: results of a randomized, double-blind, split-face, natural sunlight exposure clinical trial. J Am Acad Dermatol. 2018;78:902-910.E2.
- Draelos ZD. Cosmeceuticals for rosacea. Clin Dermatol. 2017;35:213-217.
- Yuan X, Huang X, Wang B, et al. Relationship between rosacea and dietary factors: a multicenter retrospective case–control survey. J Dermatol. 2019;46:219-225.
- Alia E, Feng H. Rosacea pathogenesis, common triggers, and dietary role: the cause, the trigger, and the positive effects of different foods. Clin Dermatol. 2022;40:122-127.
- Branco ACCC, Yoshikawa FSY, Pietrobon AJ, et al. Role of histamine in modulating the immune response and inflammation. Mediators Inflamm. 2018;2018:1-10.
- Darrigade A, Dendooven E, Aerts O. Contact allergy to fragrances and formaldehyde contributing to papulopustular rosacea. Contact Dermatitis. 2019;81:395-397.
- Linauskiene K, Isaksson M. Allergic contact dermatitis from formaldehyde mimicking impetigo and initiating rosacea. Contact Dermatitis. 2018;78:359-361.
- Al Reef T, Ghanem E. Caffeine: well-known as psychotropic substance, but little as immunomodulator. Immunobiology. 2018;223:818-825.
- Drago F, Ciccarese G, Herzum A, et al. Rosacea and alcohol intake. J Am Acad Dermatol. 2018;78:E25.
Environmental and Lifestyle Triggers of Rosacea
Environmental and Lifestyle Triggers of Rosacea
PRACTICE POINTS
- It is important to routinely assess and individualize rosacea management by encouraging use of symptom and trigger diaries to guide lifestyle modifications.
- Patients with rosacea should be encouraged to use mild, fragrance-free cleansers, barrier-supporting moisturizers, and daily broad-spectrum sunscreen and to avoid common irritants.
- Address flushing and erythema with behavioral and medical strategies; counsel patients on minimizing abrupt temperature shifts and consider topical Symbolα-adrenergic agonists, anti-inflammatory agents, or laser therapies when lifestyle measures alone are insufficient.
- Lifestyle recommendations (eg, optimal skin care practices, avoidance of dietary triggers) should be incorporated in treatment plans.
Sim and Learn: Simulation and its Value in Neurology Education
Sim and Learn: Simulation and its Value in Neurology Education
Clinical simulation is a technique, not a technology, used to replace or amplify real experiences with guided experiences that evoke or replicate substantial aspects of the real world in a fully interactive fashion.1 Simulation is widely used in medical education and spans a spectrum of sophistication, from simple reproduction of isolated body parts to high-fidelity human patient simulators that replicate whole body appearance and variable physiological parameters.2,3
Simulation-based medical education can be a valuable tool for safe health care delivery.4Simulation-based education is typically provided via 5 modalities: mannequins, computer-based mannequins, standardized patients, computer-based simulators, and software-based simulations. Simulation technology increases procedural skill by allowing for deliberate practice in a safe environment.5 Mastery learning is a stringent form of competency-based education that requires trainees to acquire clinical skill measured against a fixed achievement standard.6 In mastery learning, educational practice time varies but results are uniform. This approach improves patient outcomes and is more effective than clinical training alone.7-9
Advanced simulation models are helpful tools for neurologic education and training, especially for emergency department encounters.10 In recent years, advanced simulation models have been applied in various fields of medicine, especially emergency medicine and anesthesia.11-14
Acute neurology
In acute neurologic conditions (eg, stroke, intracerebral hemorrhage, status epilepticus, and neuromuscular respiratory failure) clinical outcomes are highly time dependent; consequently, a reduction in treatment delays can improve patient care. The application of simulation methodology allows trainees to address acute and potentially life-threatening emergencies in a safe, controlled, and reproducible environment. In addition to improving trainees’ knowledge base, simulation also helps to enhance team skills, communication, multidisciplinary collaboration, and leadership. Research has shown that deliberate practice leads to a decrease in clinical errors and improved procedural performance in the operating room.8,15 These results can be extrapolated to acute neurology settings to improve adherence to set protocols, thus streamlining management in acute settings.
Scenarios can be built to teach skills such as eliciting an appropriate history, establishing inclusion or exclusion criteria for the use of certain medications, evaluating neuroimaging and laboratory studies (while avoiding related common pitfalls), and managing treatment complications. Simulation also provides an opportunity for interprofessional education by training nurses and collaborative staff. It can be used to enhance nontechnical skills (eg, communication, situation awareness, decision making, and leadership) that further contribute to patient safety.
Simulation can be performed with the help of mannequins such as the SimMan 3G(Laerdal), which can display neurologic symptoms and physiological findings, or live actors who portray a patient by mimicking focal neurologic deficits.16,17 A briefing familiarizes the trainees with the equipment and explains the simulation process. The documentation and equipment are the same as that which is used in emergency departments or intensive care units.
Once the simulation is completed, a trainee’s performance is checked against a critical action checklist before a debriefing process during which the scenario is reviewed and learning goals are assessed. Immediate feedback is given to trainees to identify weaknesses and the simulation is repeated if multiple critical action items are missed. (Figure).17
RESIDENCY TRAINING
Simulation training in stroke is mandatory in some residency programs for neurology postgraduate year (PGY) 2 residents.18 These simulations are a part of a boot camp for incoming neurology residents after completing an internal medicine internship. The simulation program is not standardized across various training programs. The European Stroke Organization Simulation Committee has published an opinion paper with a consensus of experts about the implementation of simulation techniques in the stroke field.19,20 Residents participating in these mandatory programs are required to complete certification in the National Institutes of Health Stroke Scale (NIHSS) and the modified Rankin Scale, including a pretest that assesses their knowledge of acute stroke protocols prior to live simulation.17 A stepwise algorithm that incorporates faculty specialized in the field is used to evaluate and debrief the simulation.
Stroke vignettes are typically selected by the vascular neurology attending physician to cover thrombolytic therapy (indications and contraindications), mechanical thrombectomy, early arterial blood pressure management, anticoagulant reversal protocols, and management of thrombolytic complications (eg, neurologic worsening). Nursing staff is educated on the acute stroke protocol. Computed tomography (CT) and CT angiography scans are retrieved from teaching files. These are provided as live responses along with pertinent laboratory work, vital signs, and electrocardiogram tracings. Trainee performance is based on adherence to a critical action checklist, which includes (but is not limited to) identification of relative and absolute contraindications of thrombolytic treatments, estimation of NIHSS within 5 minutes of arrival, and consideration of candidacy for endovascular intervention.17
EVIDENCE FOR SIMULATION TRAINING
Simulations for acute ischemic stroke also improve cohesive teamwork to improve the door-to-needle and door-to-puncture time. A retrospective analysis involving first-year neurology residents at a comprehensive stroke center that compared patient cohort data before and after implementation of simulation training found that there was an improvement in door-to-needle time after implementation of stroke simulation training program by nearly 10 minutes.17 This was likely due to improvement in the comfort of the flow of management across multidisciplinary teams.
Discussing goals of care, communicating poor prognosis or complex decisions with distraught family members or patients requires practice. Simulation programs with video playback help focus on trainee’s body language, avoiding medical jargon and handling ethical dilemmas while adjusting the communication style to the patient’s personality.20 Enhanced communication skills improve patient satisfaction, trust, and adherence to treatments, all of which lead to better outcomes.21
Simulation has been effectively used as a training tool for recognizing and managing acute neuromuscular respiratory failure. These scenarios emphasize the importance of obtaining a focused clinical history, performing key neurological assessments (such as neck flexion strength and breath counting), evaluating pulmonary function tests, and identifying when to initiate ventilatory support.22 In a study designed as a simulation-based learning curriculum for status epilepticus, there was an improvement in the performance of PGY-2 residents after completing the curriculum from a median of 44.2% at pretest to 94.2% at posttest.23 In this curriculum, an emphasis was placed on the following: recognizing the delay in identification and treatment of status epilepticus; evaluating contraindications of certain antiseizure medication (ASM) based on history or laboratory work; giving first-line ASM within 5 minutes of seizure onset; airway and blood pressure assessment; suctioning the patient; use of second-line ASMs after first-line has failed; ordering a head CT and re-evaluating the case with postload ASM level; ordering a stat electroencephalography (EEG); and communicating the decision regarding patient disposition/level of care.24
There is a growing need for well designed simulation education programs targeted at the management of disorders requiring acute neurologic care, including not only stroke and status epilepticus, but also traumatic brain injury, subarachnoid hemorrhage, neuromuscular respiratory failure, flare of multiple sclerosis, acutely elevated intracranial pressure, malignant cerebral infarction, deterioration of Parkinson disease, and brain death evaluation with family counseling.25 This novel approach to teaching provides an opportunity to learn in addition to remediation with repetition of scenario and might be used for maintenance of recertification programs.
PROCEDURAL SKILLs
Perhaps one of the most studied uses for simulation in neurology is in procedural skills. This extends beyond neurology trainees and can include pulmonary critical care fellows, pediatric residents, and internal medicine residents receiving training in neurology-based procedures such as lumbar punctures (LPs). Other examples of neurology procedures and protocols in which simulation has been studied include fundoscopy, brain death evaluation, EEG interpretation in context of status epilepticus, and simulated stroke code responses. Additional procedures that lack research but may benefit from simulation-based training include the use of Doppler ultrasound and botulinum toxin injections practiced on mannequins.
Proficiency in LP procedural skills has been extensively studied by multiple institutions, with trainee levels ranging from medical students to fellows. One study in France enrolled 115 medical students without prior LP experience and randomized them to either a simulation or a control group.26 Those in the simulation group received instruction using a mannequin, and those in the control group received clinical training through hospital rotations. Both groups received an email containing literature-based information on the procedure as well as a self-assessment questionnaire before participating in either educational program.
The study showed that those students who received simulation training had a success rate of 67% on their first LP on a live patient compared with a success rate of 14% in those with traditional training. Students receiving simulation training required less assistance during the procedure from a supervisor and had higher satisfaction rates and confidence in their procedural skills.26
Another study of 128 medical students at the University of Pittsburgh found that a hybrid LP simulation significantly improved students’ confidence and perceived skill in performing LPs, obtaining informed consent, and electronic order entry. For example, confidence with LP increased from 5.95% presimulation to 90% postsimulation, with 58.24% of students reporting an improvement from minimal or no confidence to average or better (P < .001). Similarly, the proportion of students who felt able to perform LP with minimal or no assistance rose from 0% to 38.57% (P < .001). Confidence and perceived skill in obtaining informed consent and electronic order entry also saw significant gains. Although real-world skill assessments were limited by low survey response rates, preceptor evaluations and follow-up surveys suggested that students who participated in the simulation were more likely to perform these tasks independently or with minimal supervision during clinical rotations.27
Research on simulation training involving nonneurology residents is also encouraging. One study compared the LP skills of traditionally trained neurology residents (PGY-2 to PGY-4) to internal medicine residents (PGY-1) who underwent simulation on a mannequin.28 The internal medicine residents first underwent a pretest on LP performance, watched an educational video, underwent an LP demonstration, and practiced on a mannequin with feedback. The neurology residents completed the checklist-style pretest and performed an LP on a mannequin. Internal medicine residents were found to increase their pretest scores from a mean of 46.3% to 95.7% following training, whereas neurology residents scored a mean of 65.4%. More than half of neurology residents were unable to identify the correct anatomic location or standard cerebrospinal fluid (CSF) tests to be ordered on a routine LP.28
A pediatric resident study in Canada found that following simulation-based training, LP procedural skill improved in 15 of 16 residents tested, and PGY-1 residents showed a reduction in anxiety related to performing the procedure.29
Virtual Reality
An additional tool for simulation is the use of virtual reality (VR) in combination with mannequins. A French study used videos of LPs on actual patients, from equipment set up to final CSF collection and termination of the procedure.30 These videos included a 360-degree view of the procedure. The short video was administered through a VR device (the Oculus Go headset by Microsoft) or by a YouTube video (if VR was not desired).
Participants in the study watched the video then performed an LP on a mannequin. Those who used the VR option had minimal adverse effects (eg, low rates of cybersickness, blurred vision, nausea) and high satisfaction regarding their training environment.30Another VR-based program is the vascular intervention system trainer, which allows clinicians to use endovascular devices and simulate procedures such as thrombectomies. VR simulation is used for trainees and to retrain experienced physicians in performance of high-risk procedures.31
Fundoscopic and Ultrasound Simulations
The AR403 eye stimulator device for fundoscopic examinations is a mannequin-based simulation.32 In a single-center, prospective, single-blind study of neurology and pediatric neurology residents, trainees were split into control and intervention groups, with the intervention group receiving simulator training. Both groups received video lectures on fundoscopy techniques. Pre- and postintervention measurements included knowledge, skill, and total scores on the skills assessment. Of the 48 trainees who participated, the intervention group demonstrated significantly higher increases in skills (P = .01) and total (P = .02) scores, although knowledge scores did not improve. The intervention group also reported higher comfort levels, higher confidence, and higher success rates.
Areas that would benefit from simulation training and development include ultrasound training, such as transcranial Doppler evaluation. In a national survey of residents in anesthesia and critical care, trainees reported that simulation was not frequently used in ultrasound training and that bedside teaching was more common. Interestingly, there was a discrepancy between the opinions of residents and program directors. The program directors felt simulation was in fact used (18.2% of program directors reported this vs 5.3% of trainees).33
A new program, the NewroSim (Gaumard), is a computer-based model of cerebral perfusion that may be a useful tool in this setting. It can simulate blood flow velocities, including pathologic ones, both with a mannequin or without.34
Another potential area for development is the use of mannequins to teach botulinum toxin injections for migraine, dystonia and spasticity in a training environment This is typically led by pharmaceutical representatives who are not necessarily clinicians. Residents and fellows may benefit instead from clinician-led education during their training programs.
Simulation in Patient Communication
Simulation provides a realistic environment for teaching rapid decision-making, leadership, and appropriate management of acutely ill neurologic patients; this includes the communication skills needed in response to neurologic injury.35 Simulation can be particularly useful in situations involving brain death determination, where the communication techniques differ significantly from those used in shared decision-making. Simulation provides a low-stakes setting for clinicians to practice the process of brain death determination and communication, leading to improved confidence and knowledge.36
In the context of acute neurologic emergencies, simulation exercises have been used to investigate the consistency of prognostication across a spectrum of neurology physicians. These exercises revealed that acute neuroprognostication is highly variable and often inaccurate among neurology clinicians, suggesting a potential area for improvement through further simulation training.37
FUTURE DIRECTIONS
Simulation education in neurology can be directed towards learners at all levels, including medical students, residents, fellows, nurses, and medical technologists. In addition, simulation has great value to different disciplines, including emergency medicine, intensive care, and psychiatry. In our view simulation is not being used to full potential in neurology.
Simulation can be used to expose clinicians to rare pathology, play an integral role in competency-based evaluations, and serve as the foundation for simulation-based neurology curriculums, teleneurology simulation training programs, and team training for neurologic emergencies.38Another under-recognized aspect of neurology education is teaching interpersonal communication and professionalism. A survey conducted at a neurology department (20 residents and 73 faculty respondents) asked about residents’ comfort level in performing a number of interpersonal communication and professionalism tasks.38 While none of the residents said they were “very uncomfortable” with these tasks, only 1 resident reported being “very comfortable.” In addition, fewer than 50% noted that they had been directly observed by a faculty member while performing these tasks. The results prompted the facility to develop a simulation curriculum that including observation and feedback from 8 objective structured clinical examinations at a simulation center. A standardized professional simulated the role of a patient, caregiver, medical student, or a faculty member. Residents indicated in postsimulation surveys that it was very useful, and a majority voted for the activity to be repeated for future classes.38
Simulation models may also provide a more objective method to evaluate neurology residents. Accreditation Council for Graduate Medical Education has provided Milestones that are used for assessment of neurology residents. Most of the programs rely on end-of-rotation faculty evaluations. These are subjective evaluations, rely on chance evaluations and may not reflect the exact caliber of a trainee in different clinical situations. Simulation models can serve as alternatives to provide an objective and accurate assessment of resident’s competency in different neurologic scenarios.
In a study of PGY-4 neurology residents from 3 tertiary care academic medical centers were evaluated using simulation-based assessment. Their skills in identifying and managing status epilepticus were assessed via a simulation-based model and compared with clinical experience. No graduating neurology residents were able to meet or exceed the minimum passing score during the testing. It was suggested that end-of-rotation evaluations are inadequate for assigning level of Milestones.24 To move forward with use of simulation-based assessments, these models need to be trialed more extensively and validated.
Morris et al developed simulations for assessment in neurocritical care.39 Ten evaluative simulation cases were developed. Researchers reported on 64 trainee participants in 274 evaluative simulation scenarios. The participants were very satisfied with the cases, found them to be very realistic and appropriately difficult. Interrater reliability was acceptable for both checklist action items and global rating scales. The researchers concluded that they were able to demonstrate validity evidence via the 10 simulation cases for assessment in neurologic emergencies.39 It is the authors’ belief that the future of residents’ competency assessment should include more widespread use of similar simulation models.
Finally, VR and augmented reality (AR) have shown promise in various fields, including neurology. In neurology, these technologies are being explored for applications in rehabilitation, therapy, and medical training. Ongoing research aims to leverage these technologies for improved patient outcomes and medical education. Virtual simulations can recreate neurologic scenarios, allowing learners to interact with 3-dimensional (3D) models of the brain or experience virtual patient cases. AR can enhance traditional learning materials by overlaying digital information onto real-world objects, aiding in the understanding of complex neuroanatomy and medical concepts. These technologies contribute to more engaging and effective neurology education.39In a study of 84 graduate medical students divided into 3 groups, the first group attended a traditional lecture on neuroanatomy, the second group was shown VR-based 3D images, and the third group had a VR-based, interactive and stereoscopic session.40 Groups 2 and 3 showed the highest mean scores in evaluations and differed significantly from Group 1 (P < .05). Groups 2 and 3 did not differ significantly from each other. The researchers concluded that VR-based resources for teaching neuroanatomy fostered significantly higher learning when compared to the traditional methods.40
- Corvetto M, Bravo MP, Montaña R, et al. Simulación en educación médica: una sinopsis. Rev Med Chil. 2013;141:70-79. doi:10.4067/S0034-98872013000100010
- Lane JL, Slavin S, Ziv A. Simulation in medical education: a review. Simul Gaming. 2001;32:297-314. doi:10.1177/104687810103200302
- Bradley P. The history of simulation in medical education and possible future directions. Med Educ. 2006;40:254-262. doi:10.1111/j.1365-2929.2006.02394.x
- Jones F, Passos-Neto C, Melro Braghiroli O. Simulation in medical education: brief history and methodology. Princ Pract Clin Res J. 2015;1:46-54. doi:10.21801/ppcrj.2015.12.8
- Issenberg SB. Simulation technology for health care professional skills training and assessment. JAMA. 1999;28:861-866. doi:10.1001/jama.282.9.861
- McGaghie WC, Miller GE, Sajid AW, et al. Competency-based curriculum development on medical education: an introduction. Public Health Pap. 1978;68:11-91.
- Barsuk JH, Cohen ER, Feinglass J, et al. Use of simulation-based education to reduce catheter-related bloodstream infections. Arch Intern Med. 2009;169:1420-1423. doi:10.1001/archinternmed.2009.215
- Wayne DB, Didwania A, Feinglass J, et al. Simulation-based education improves quality of care during cardiac arrest team responses at an academic teaching hospital: a case-control study. Chest. 2008;133:56-61. doi:10.1378/chest.07-0131
- McGaghie WC, Issenberg SB, Cohen ER, et al. Does simulation-based medical education with deliberate practice yield better results than traditional clinical education? A meta-analytic comparative review of the evidence. Acad Med. 2011;86:706-711. doi:10.1097/ACM.0b013e318217e119
- Micieli G, Cavallini A, Santalucia P, et al. Simulation in neurology. Neurol Sci. 2015;36:1967-1971. doi:10.1007/s10072-015-2228-8
- Bond WF, Lammers RL, Spillane LL, et al. The use of simulation in emergency medicine: a research agenda. Acad Emerg Med. 2007;14:353-363. doi:10.1197/j.aem.2006.11.02112.
- McLaughlin SA, Doezema D, Sklar DP. Human simulation in emergency medicine training: a model curriculum. Acad Emerg Med. 2002;9:1310-1318. doi:10.1111/j.1553-2712.2002.tb01593.x
- Howard SK, Gaba DM, Fish KJ, et al. Anesthesia crisis resource management training: teaching anesthesiologists to handle critical incidents. Aviat Space Environ Med. 1992;63:763-770.
- Gaba DM. Anaesthesiology as a model for patient safety in health care. BMJ. 2000;320:785-788. doi:10.1136/bmj.320.7237.785
- Sedlack RE, Kolars JC. Computer simulator training enhances the competency of gastroenterology fellows at colonoscopy: results of a pilot study. Am J Gastroenterol. 2004;99:33-37. doi:10.1111/j.1572-0241.2004.04007.x
- Tchopev ZN, Nelson AE, Hunninghake JC, et al. Curriculum innovations: high-fidelity simulation of acute neurology enhances rising resident confidence: results from a multicohort study. Neurol Educ. 2022;1:e200022. doi:10.1212/ne9.0000000000200022
- Mehta T, Strauss S, Beland D, et al. Stroke simulation improves acute stroke management: a systems-based practice experience. J Grad Med Educ. 2018;10:57-62. doi:10.4300/JGME-D-17-00167.1
- Pergakis MB, Chang WTW, Tabatabai A, et al. Simulation-based assessment of graduate neurology trainees’ performance managing acute ischemic stroke. Neurology. 2021;97:e2414-e2422. doi:10.1212/WNL.0000000000012972
- Casolla B. Simulation for neurology training: acute setting and beyond. Rev Neurol (Paris). 2021;177:1207-1213. doi:10.1016/j.neurol.2021.03.008
- Casolla B, de Leciñana MA, Neves R, et al. Simulation training programs for acute stroke care: Objectives and standards of methodology. Eur Stroke J. 2020;5:328-335. doi:10.1177/2396987320971105
- Zolnierek KB, Dimatteo MR. Physician communication and patient adherence to treatment: a meta-analysis. Med Care. 2009;47:826-834.doi:10.1097/MLR.0b013e31819a5acc
- Patel RA, Mohl L, Paetow G, Maiser S. Acute neuromuscular respiratory weakness due to acute inflammatory demyelinating polyneuropathy (AIDP): a simulation scenario for neurology providers. MedEdPORTAL. 2019;15:10811. doi:10.15766/mep_2374-8265.10811
- Mikhaeil-Demo Y, Barsuk JH, Culler GW, et al. Use of a simulation-based mastery learning curriculum for neurology residents to improve the identification and management of status epilepticus. Epilepsy Behav. 2020;111:107247. doi:10.1016/j.yebeh.2020.107247
- Mikhaeil-Demo Y, Holmboe E, Gerard EE, et al. Simulation-based assessments and graduating neurology residents’ milestones: status epilepticus milestones. J Grad Med Educ. 2021;13:223-230. doi:10.4300/JGME-D-20-00832.1
- Hocker S, Wijdicks EFM, Feske SK, et al. Use of simulation in acute neurology training: point and counterpoint. Ann Neurol. 2015;78:337-342. doi:10.1002/ana.24473
- Gaubert S, Blet A, Dib F, et al. Positive effects of lumbar puncture simulation training for medical students in clinical practice. BMC Med Educ. 2021;21:1-6. doi:10.1186/S12909-020-02452-327.
- Yanta C, Knepper L, Van Deusen R, et al. The use of hybrid lumbar puncture simulation to teach entrustable professional activities during a medical student neurology clerkship. MedEdPublish (2016). 2021;9:266. doi:10.15694/mep.2020.000266.2
- Barsuk JH, Cohen ER, Caprio T, et al. Simulation-based education with mastery learning improves residents’ lumbar puncture skills. Neurology. 2012;79:132-137. doi:10.1212/WNL.0B013E31825DD39D
- McMillan HJ, Writer H, Moreau KA, et al. Lumbar puncture simulation in pediatric residency training: improving procedural competence and decreasing anxiety. BMC Med Educ. 2016;16:198. doi:10.1186/S12909-016-0722-1
- Vrillon A, Gonzales-Marabal L, Ceccaldi PF, et al. Using virtual reality in lumbar puncture training improves students learning experience. BMC Med Educ. 2022;22:244. doi:10.1186/S12909-022-03317-7
- Liebig T, Holtmannspötter M, Crossley R, et al. Metric-based virtual reality simulation: a paradigm shift in training for mechanical thrombectomy in acute stroke. Stroke. 2018;49:e239-e242.doi:10.1161/STROKEAHA.118.021089
- Gupta DK, Khandker N, Stacy K, et al. Utility of combining a simulation-based method with a lecture-based method for fundoscopy training in neurology residency. JAMA Neurol. 2017;74:1223-1227. doi:10.1001/JAMANEUROL.2017.2073
- Mongodi S, Bonomi F, Vaschetto R, et al. Point-of-care ultrasound training for residents in anaesthesia and critical care: results of a national survey comparing residents and training program directors’ perspectives. BMC Med Educ. 2022;22:647. doi:10.1186/S12909-022-03708-W
- Morris NA, Czeisler BM, Sarwal A. Simulation in neurocritical care: past, present, and future. Neurocrit Care. 2019;30:522-533. doi:10.1007/S12028-018-0629-2
- Wijdicks EFM, Hocker SE. A future for simulation in acute neurology. Semin Neurol. 2018;38:465-470. doi:10.1055/s-0038-1666986
- Kramer NM, O’Mahony S, Deamant C. Brain death determination and communication: an innovative approach using simulation and standardized patients. J Pain Symptom Manage. 2022;63:e765-e772. doi:10.1016/j.jpainsymman.2022.01.020
- Sloane KL, Miller JJ, Piquet A, et al. Prognostication in acute neurological emergencies. J Stroke Cerebrovasc Dis. 2022;31:106277. doi:10.1016/J.JSTROKECEREBROVASDIS.2021.106277
- Kurzweil AM, Lewis A, Pleninger P, et al. Education research: teaching and assessing communication and professionalism in neurology residency with simulation. Neurology. 2020;94:229-232. doi:10.1212/WNL.0000000000008895
- Morris NA, Chang WT, Tabatabai A, et al. Development of neurological emergency simulations for assessment: content evidence and response process. Neurocrit Care. 2021;35:389-396. doi:10.1007/S12028-020-01176-Y
- De Faria JWV, Teixeira MJ, De Moura Sousa Júnior L, et al. Virtual and stereoscopic anatomy: when virtual reality meets medical education. J Neurosurg. 2016;125:1105-1111. doi:10.3171/2015.8.JNS141563
Clinical simulation is a technique, not a technology, used to replace or amplify real experiences with guided experiences that evoke or replicate substantial aspects of the real world in a fully interactive fashion.1 Simulation is widely used in medical education and spans a spectrum of sophistication, from simple reproduction of isolated body parts to high-fidelity human patient simulators that replicate whole body appearance and variable physiological parameters.2,3
Simulation-based medical education can be a valuable tool for safe health care delivery.4Simulation-based education is typically provided via 5 modalities: mannequins, computer-based mannequins, standardized patients, computer-based simulators, and software-based simulations. Simulation technology increases procedural skill by allowing for deliberate practice in a safe environment.5 Mastery learning is a stringent form of competency-based education that requires trainees to acquire clinical skill measured against a fixed achievement standard.6 In mastery learning, educational practice time varies but results are uniform. This approach improves patient outcomes and is more effective than clinical training alone.7-9
Advanced simulation models are helpful tools for neurologic education and training, especially for emergency department encounters.10 In recent years, advanced simulation models have been applied in various fields of medicine, especially emergency medicine and anesthesia.11-14
Acute neurology
In acute neurologic conditions (eg, stroke, intracerebral hemorrhage, status epilepticus, and neuromuscular respiratory failure) clinical outcomes are highly time dependent; consequently, a reduction in treatment delays can improve patient care. The application of simulation methodology allows trainees to address acute and potentially life-threatening emergencies in a safe, controlled, and reproducible environment. In addition to improving trainees’ knowledge base, simulation also helps to enhance team skills, communication, multidisciplinary collaboration, and leadership. Research has shown that deliberate practice leads to a decrease in clinical errors and improved procedural performance in the operating room.8,15 These results can be extrapolated to acute neurology settings to improve adherence to set protocols, thus streamlining management in acute settings.
Scenarios can be built to teach skills such as eliciting an appropriate history, establishing inclusion or exclusion criteria for the use of certain medications, evaluating neuroimaging and laboratory studies (while avoiding related common pitfalls), and managing treatment complications. Simulation also provides an opportunity for interprofessional education by training nurses and collaborative staff. It can be used to enhance nontechnical skills (eg, communication, situation awareness, decision making, and leadership) that further contribute to patient safety.
Simulation can be performed with the help of mannequins such as the SimMan 3G(Laerdal), which can display neurologic symptoms and physiological findings, or live actors who portray a patient by mimicking focal neurologic deficits.16,17 A briefing familiarizes the trainees with the equipment and explains the simulation process. The documentation and equipment are the same as that which is used in emergency departments or intensive care units.
Once the simulation is completed, a trainee’s performance is checked against a critical action checklist before a debriefing process during which the scenario is reviewed and learning goals are assessed. Immediate feedback is given to trainees to identify weaknesses and the simulation is repeated if multiple critical action items are missed. (Figure).17

RESIDENCY TRAINING
Simulation training in stroke is mandatory in some residency programs for neurology postgraduate year (PGY) 2 residents.18 These simulations are a part of a boot camp for incoming neurology residents after completing an internal medicine internship. The simulation program is not standardized across various training programs. The European Stroke Organization Simulation Committee has published an opinion paper with a consensus of experts about the implementation of simulation techniques in the stroke field.19,20 Residents participating in these mandatory programs are required to complete certification in the National Institutes of Health Stroke Scale (NIHSS) and the modified Rankin Scale, including a pretest that assesses their knowledge of acute stroke protocols prior to live simulation.17 A stepwise algorithm that incorporates faculty specialized in the field is used to evaluate and debrief the simulation.
Stroke vignettes are typically selected by the vascular neurology attending physician to cover thrombolytic therapy (indications and contraindications), mechanical thrombectomy, early arterial blood pressure management, anticoagulant reversal protocols, and management of thrombolytic complications (eg, neurologic worsening). Nursing staff is educated on the acute stroke protocol. Computed tomography (CT) and CT angiography scans are retrieved from teaching files. These are provided as live responses along with pertinent laboratory work, vital signs, and electrocardiogram tracings. Trainee performance is based on adherence to a critical action checklist, which includes (but is not limited to) identification of relative and absolute contraindications of thrombolytic treatments, estimation of NIHSS within 5 minutes of arrival, and consideration of candidacy for endovascular intervention.17
EVIDENCE FOR SIMULATION TRAINING
Simulations for acute ischemic stroke also improve cohesive teamwork to improve the door-to-needle and door-to-puncture time. A retrospective analysis involving first-year neurology residents at a comprehensive stroke center that compared patient cohort data before and after implementation of simulation training found that there was an improvement in door-to-needle time after implementation of stroke simulation training program by nearly 10 minutes.17 This was likely due to improvement in the comfort of the flow of management across multidisciplinary teams.
Discussing goals of care, communicating poor prognosis or complex decisions with distraught family members or patients requires practice. Simulation programs with video playback help focus on trainee’s body language, avoiding medical jargon and handling ethical dilemmas while adjusting the communication style to the patient’s personality.20 Enhanced communication skills improve patient satisfaction, trust, and adherence to treatments, all of which lead to better outcomes.21
Simulation has been effectively used as a training tool for recognizing and managing acute neuromuscular respiratory failure. These scenarios emphasize the importance of obtaining a focused clinical history, performing key neurological assessments (such as neck flexion strength and breath counting), evaluating pulmonary function tests, and identifying when to initiate ventilatory support.22 In a study designed as a simulation-based learning curriculum for status epilepticus, there was an improvement in the performance of PGY-2 residents after completing the curriculum from a median of 44.2% at pretest to 94.2% at posttest.23 In this curriculum, an emphasis was placed on the following: recognizing the delay in identification and treatment of status epilepticus; evaluating contraindications of certain antiseizure medication (ASM) based on history or laboratory work; giving first-line ASM within 5 minutes of seizure onset; airway and blood pressure assessment; suctioning the patient; use of second-line ASMs after first-line has failed; ordering a head CT and re-evaluating the case with postload ASM level; ordering a stat electroencephalography (EEG); and communicating the decision regarding patient disposition/level of care.24
There is a growing need for well designed simulation education programs targeted at the management of disorders requiring acute neurologic care, including not only stroke and status epilepticus, but also traumatic brain injury, subarachnoid hemorrhage, neuromuscular respiratory failure, flare of multiple sclerosis, acutely elevated intracranial pressure, malignant cerebral infarction, deterioration of Parkinson disease, and brain death evaluation with family counseling.25 This novel approach to teaching provides an opportunity to learn in addition to remediation with repetition of scenario and might be used for maintenance of recertification programs.
PROCEDURAL SKILLs
Perhaps one of the most studied uses for simulation in neurology is in procedural skills. This extends beyond neurology trainees and can include pulmonary critical care fellows, pediatric residents, and internal medicine residents receiving training in neurology-based procedures such as lumbar punctures (LPs). Other examples of neurology procedures and protocols in which simulation has been studied include fundoscopy, brain death evaluation, EEG interpretation in context of status epilepticus, and simulated stroke code responses. Additional procedures that lack research but may benefit from simulation-based training include the use of Doppler ultrasound and botulinum toxin injections practiced on mannequins.
Proficiency in LP procedural skills has been extensively studied by multiple institutions, with trainee levels ranging from medical students to fellows. One study in France enrolled 115 medical students without prior LP experience and randomized them to either a simulation or a control group.26 Those in the simulation group received instruction using a mannequin, and those in the control group received clinical training through hospital rotations. Both groups received an email containing literature-based information on the procedure as well as a self-assessment questionnaire before participating in either educational program.
The study showed that those students who received simulation training had a success rate of 67% on their first LP on a live patient compared with a success rate of 14% in those with traditional training. Students receiving simulation training required less assistance during the procedure from a supervisor and had higher satisfaction rates and confidence in their procedural skills.26
Another study of 128 medical students at the University of Pittsburgh found that a hybrid LP simulation significantly improved students’ confidence and perceived skill in performing LPs, obtaining informed consent, and electronic order entry. For example, confidence with LP increased from 5.95% presimulation to 90% postsimulation, with 58.24% of students reporting an improvement from minimal or no confidence to average or better (P < .001). Similarly, the proportion of students who felt able to perform LP with minimal or no assistance rose from 0% to 38.57% (P < .001). Confidence and perceived skill in obtaining informed consent and electronic order entry also saw significant gains. Although real-world skill assessments were limited by low survey response rates, preceptor evaluations and follow-up surveys suggested that students who participated in the simulation were more likely to perform these tasks independently or with minimal supervision during clinical rotations.27
Research on simulation training involving nonneurology residents is also encouraging. One study compared the LP skills of traditionally trained neurology residents (PGY-2 to PGY-4) to internal medicine residents (PGY-1) who underwent simulation on a mannequin.28 The internal medicine residents first underwent a pretest on LP performance, watched an educational video, underwent an LP demonstration, and practiced on a mannequin with feedback. The neurology residents completed the checklist-style pretest and performed an LP on a mannequin. Internal medicine residents were found to increase their pretest scores from a mean of 46.3% to 95.7% following training, whereas neurology residents scored a mean of 65.4%. More than half of neurology residents were unable to identify the correct anatomic location or standard cerebrospinal fluid (CSF) tests to be ordered on a routine LP.28
A pediatric resident study in Canada found that following simulation-based training, LP procedural skill improved in 15 of 16 residents tested, and PGY-1 residents showed a reduction in anxiety related to performing the procedure.29
Virtual Reality
An additional tool for simulation is the use of virtual reality (VR) in combination with mannequins. A French study used videos of LPs on actual patients, from equipment set up to final CSF collection and termination of the procedure.30 These videos included a 360-degree view of the procedure. The short video was administered through a VR device (the Oculus Go headset by Microsoft) or by a YouTube video (if VR was not desired).
Participants in the study watched the video then performed an LP on a mannequin. Those who used the VR option had minimal adverse effects (eg, low rates of cybersickness, blurred vision, nausea) and high satisfaction regarding their training environment.30Another VR-based program is the vascular intervention system trainer, which allows clinicians to use endovascular devices and simulate procedures such as thrombectomies. VR simulation is used for trainees and to retrain experienced physicians in performance of high-risk procedures.31
Fundoscopic and Ultrasound Simulations
The AR403 eye stimulator device for fundoscopic examinations is a mannequin-based simulation.32 In a single-center, prospective, single-blind study of neurology and pediatric neurology residents, trainees were split into control and intervention groups, with the intervention group receiving simulator training. Both groups received video lectures on fundoscopy techniques. Pre- and postintervention measurements included knowledge, skill, and total scores on the skills assessment. Of the 48 trainees who participated, the intervention group demonstrated significantly higher increases in skills (P = .01) and total (P = .02) scores, although knowledge scores did not improve. The intervention group also reported higher comfort levels, higher confidence, and higher success rates.
Areas that would benefit from simulation training and development include ultrasound training, such as transcranial Doppler evaluation. In a national survey of residents in anesthesia and critical care, trainees reported that simulation was not frequently used in ultrasound training and that bedside teaching was more common. Interestingly, there was a discrepancy between the opinions of residents and program directors. The program directors felt simulation was in fact used (18.2% of program directors reported this vs 5.3% of trainees).33
A new program, the NewroSim (Gaumard), is a computer-based model of cerebral perfusion that may be a useful tool in this setting. It can simulate blood flow velocities, including pathologic ones, both with a mannequin or without.34
Another potential area for development is the use of mannequins to teach botulinum toxin injections for migraine, dystonia and spasticity in a training environment This is typically led by pharmaceutical representatives who are not necessarily clinicians. Residents and fellows may benefit instead from clinician-led education during their training programs.
Simulation in Patient Communication
Simulation provides a realistic environment for teaching rapid decision-making, leadership, and appropriate management of acutely ill neurologic patients; this includes the communication skills needed in response to neurologic injury.35 Simulation can be particularly useful in situations involving brain death determination, where the communication techniques differ significantly from those used in shared decision-making. Simulation provides a low-stakes setting for clinicians to practice the process of brain death determination and communication, leading to improved confidence and knowledge.36
In the context of acute neurologic emergencies, simulation exercises have been used to investigate the consistency of prognostication across a spectrum of neurology physicians. These exercises revealed that acute neuroprognostication is highly variable and often inaccurate among neurology clinicians, suggesting a potential area for improvement through further simulation training.37
FUTURE DIRECTIONS
Simulation education in neurology can be directed towards learners at all levels, including medical students, residents, fellows, nurses, and medical technologists. In addition, simulation has great value to different disciplines, including emergency medicine, intensive care, and psychiatry. In our view simulation is not being used to full potential in neurology.
Simulation can be used to expose clinicians to rare pathology, play an integral role in competency-based evaluations, and serve as the foundation for simulation-based neurology curriculums, teleneurology simulation training programs, and team training for neurologic emergencies.38Another under-recognized aspect of neurology education is teaching interpersonal communication and professionalism. A survey conducted at a neurology department (20 residents and 73 faculty respondents) asked about residents’ comfort level in performing a number of interpersonal communication and professionalism tasks.38 While none of the residents said they were “very uncomfortable” with these tasks, only 1 resident reported being “very comfortable.” In addition, fewer than 50% noted that they had been directly observed by a faculty member while performing these tasks. The results prompted the facility to develop a simulation curriculum that including observation and feedback from 8 objective structured clinical examinations at a simulation center. A standardized professional simulated the role of a patient, caregiver, medical student, or a faculty member. Residents indicated in postsimulation surveys that it was very useful, and a majority voted for the activity to be repeated for future classes.38
Simulation models may also provide a more objective method to evaluate neurology residents. Accreditation Council for Graduate Medical Education has provided Milestones that are used for assessment of neurology residents. Most of the programs rely on end-of-rotation faculty evaluations. These are subjective evaluations, rely on chance evaluations and may not reflect the exact caliber of a trainee in different clinical situations. Simulation models can serve as alternatives to provide an objective and accurate assessment of resident’s competency in different neurologic scenarios.
In a study of PGY-4 neurology residents from 3 tertiary care academic medical centers were evaluated using simulation-based assessment. Their skills in identifying and managing status epilepticus were assessed via a simulation-based model and compared with clinical experience. No graduating neurology residents were able to meet or exceed the minimum passing score during the testing. It was suggested that end-of-rotation evaluations are inadequate for assigning level of Milestones.24 To move forward with use of simulation-based assessments, these models need to be trialed more extensively and validated.
Morris et al developed simulations for assessment in neurocritical care.39 Ten evaluative simulation cases were developed. Researchers reported on 64 trainee participants in 274 evaluative simulation scenarios. The participants were very satisfied with the cases, found them to be very realistic and appropriately difficult. Interrater reliability was acceptable for both checklist action items and global rating scales. The researchers concluded that they were able to demonstrate validity evidence via the 10 simulation cases for assessment in neurologic emergencies.39 It is the authors’ belief that the future of residents’ competency assessment should include more widespread use of similar simulation models.
Finally, VR and augmented reality (AR) have shown promise in various fields, including neurology. In neurology, these technologies are being explored for applications in rehabilitation, therapy, and medical training. Ongoing research aims to leverage these technologies for improved patient outcomes and medical education. Virtual simulations can recreate neurologic scenarios, allowing learners to interact with 3-dimensional (3D) models of the brain or experience virtual patient cases. AR can enhance traditional learning materials by overlaying digital information onto real-world objects, aiding in the understanding of complex neuroanatomy and medical concepts. These technologies contribute to more engaging and effective neurology education.39In a study of 84 graduate medical students divided into 3 groups, the first group attended a traditional lecture on neuroanatomy, the second group was shown VR-based 3D images, and the third group had a VR-based, interactive and stereoscopic session.40 Groups 2 and 3 showed the highest mean scores in evaluations and differed significantly from Group 1 (P < .05). Groups 2 and 3 did not differ significantly from each other. The researchers concluded that VR-based resources for teaching neuroanatomy fostered significantly higher learning when compared to the traditional methods.40
Clinical simulation is a technique, not a technology, used to replace or amplify real experiences with guided experiences that evoke or replicate substantial aspects of the real world in a fully interactive fashion.1 Simulation is widely used in medical education and spans a spectrum of sophistication, from simple reproduction of isolated body parts to high-fidelity human patient simulators that replicate whole body appearance and variable physiological parameters.2,3
Simulation-based medical education can be a valuable tool for safe health care delivery.4Simulation-based education is typically provided via 5 modalities: mannequins, computer-based mannequins, standardized patients, computer-based simulators, and software-based simulations. Simulation technology increases procedural skill by allowing for deliberate practice in a safe environment.5 Mastery learning is a stringent form of competency-based education that requires trainees to acquire clinical skill measured against a fixed achievement standard.6 In mastery learning, educational practice time varies but results are uniform. This approach improves patient outcomes and is more effective than clinical training alone.7-9
Advanced simulation models are helpful tools for neurologic education and training, especially for emergency department encounters.10 In recent years, advanced simulation models have been applied in various fields of medicine, especially emergency medicine and anesthesia.11-14
Acute neurology
In acute neurologic conditions (eg, stroke, intracerebral hemorrhage, status epilepticus, and neuromuscular respiratory failure) clinical outcomes are highly time dependent; consequently, a reduction in treatment delays can improve patient care. The application of simulation methodology allows trainees to address acute and potentially life-threatening emergencies in a safe, controlled, and reproducible environment. In addition to improving trainees’ knowledge base, simulation also helps to enhance team skills, communication, multidisciplinary collaboration, and leadership. Research has shown that deliberate practice leads to a decrease in clinical errors and improved procedural performance in the operating room.8,15 These results can be extrapolated to acute neurology settings to improve adherence to set protocols, thus streamlining management in acute settings.
Scenarios can be built to teach skills such as eliciting an appropriate history, establishing inclusion or exclusion criteria for the use of certain medications, evaluating neuroimaging and laboratory studies (while avoiding related common pitfalls), and managing treatment complications. Simulation also provides an opportunity for interprofessional education by training nurses and collaborative staff. It can be used to enhance nontechnical skills (eg, communication, situation awareness, decision making, and leadership) that further contribute to patient safety.
Simulation can be performed with the help of mannequins such as the SimMan 3G(Laerdal), which can display neurologic symptoms and physiological findings, or live actors who portray a patient by mimicking focal neurologic deficits.16,17 A briefing familiarizes the trainees with the equipment and explains the simulation process. The documentation and equipment are the same as that which is used in emergency departments or intensive care units.
Once the simulation is completed, a trainee’s performance is checked against a critical action checklist before a debriefing process during which the scenario is reviewed and learning goals are assessed. Immediate feedback is given to trainees to identify weaknesses and the simulation is repeated if multiple critical action items are missed. (Figure).17

RESIDENCY TRAINING
Simulation training in stroke is mandatory in some residency programs for neurology postgraduate year (PGY) 2 residents.18 These simulations are a part of a boot camp for incoming neurology residents after completing an internal medicine internship. The simulation program is not standardized across various training programs. The European Stroke Organization Simulation Committee has published an opinion paper with a consensus of experts about the implementation of simulation techniques in the stroke field.19,20 Residents participating in these mandatory programs are required to complete certification in the National Institutes of Health Stroke Scale (NIHSS) and the modified Rankin Scale, including a pretest that assesses their knowledge of acute stroke protocols prior to live simulation.17 A stepwise algorithm that incorporates faculty specialized in the field is used to evaluate and debrief the simulation.
Stroke vignettes are typically selected by the vascular neurology attending physician to cover thrombolytic therapy (indications and contraindications), mechanical thrombectomy, early arterial blood pressure management, anticoagulant reversal protocols, and management of thrombolytic complications (eg, neurologic worsening). Nursing staff is educated on the acute stroke protocol. Computed tomography (CT) and CT angiography scans are retrieved from teaching files. These are provided as live responses along with pertinent laboratory work, vital signs, and electrocardiogram tracings. Trainee performance is based on adherence to a critical action checklist, which includes (but is not limited to) identification of relative and absolute contraindications of thrombolytic treatments, estimation of NIHSS within 5 minutes of arrival, and consideration of candidacy for endovascular intervention.17
EVIDENCE FOR SIMULATION TRAINING
Simulations for acute ischemic stroke also improve cohesive teamwork to improve the door-to-needle and door-to-puncture time. A retrospective analysis involving first-year neurology residents at a comprehensive stroke center that compared patient cohort data before and after implementation of simulation training found that there was an improvement in door-to-needle time after implementation of stroke simulation training program by nearly 10 minutes.17 This was likely due to improvement in the comfort of the flow of management across multidisciplinary teams.
Discussing goals of care, communicating poor prognosis or complex decisions with distraught family members or patients requires practice. Simulation programs with video playback help focus on trainee’s body language, avoiding medical jargon and handling ethical dilemmas while adjusting the communication style to the patient’s personality.20 Enhanced communication skills improve patient satisfaction, trust, and adherence to treatments, all of which lead to better outcomes.21
Simulation has been effectively used as a training tool for recognizing and managing acute neuromuscular respiratory failure. These scenarios emphasize the importance of obtaining a focused clinical history, performing key neurological assessments (such as neck flexion strength and breath counting), evaluating pulmonary function tests, and identifying when to initiate ventilatory support.22 In a study designed as a simulation-based learning curriculum for status epilepticus, there was an improvement in the performance of PGY-2 residents after completing the curriculum from a median of 44.2% at pretest to 94.2% at posttest.23 In this curriculum, an emphasis was placed on the following: recognizing the delay in identification and treatment of status epilepticus; evaluating contraindications of certain antiseizure medication (ASM) based on history or laboratory work; giving first-line ASM within 5 minutes of seizure onset; airway and blood pressure assessment; suctioning the patient; use of second-line ASMs after first-line has failed; ordering a head CT and re-evaluating the case with postload ASM level; ordering a stat electroencephalography (EEG); and communicating the decision regarding patient disposition/level of care.24
There is a growing need for well designed simulation education programs targeted at the management of disorders requiring acute neurologic care, including not only stroke and status epilepticus, but also traumatic brain injury, subarachnoid hemorrhage, neuromuscular respiratory failure, flare of multiple sclerosis, acutely elevated intracranial pressure, malignant cerebral infarction, deterioration of Parkinson disease, and brain death evaluation with family counseling.25 This novel approach to teaching provides an opportunity to learn in addition to remediation with repetition of scenario and might be used for maintenance of recertification programs.
PROCEDURAL SKILLs
Perhaps one of the most studied uses for simulation in neurology is in procedural skills. This extends beyond neurology trainees and can include pulmonary critical care fellows, pediatric residents, and internal medicine residents receiving training in neurology-based procedures such as lumbar punctures (LPs). Other examples of neurology procedures and protocols in which simulation has been studied include fundoscopy, brain death evaluation, EEG interpretation in context of status epilepticus, and simulated stroke code responses. Additional procedures that lack research but may benefit from simulation-based training include the use of Doppler ultrasound and botulinum toxin injections practiced on mannequins.
Proficiency in LP procedural skills has been extensively studied by multiple institutions, with trainee levels ranging from medical students to fellows. One study in France enrolled 115 medical students without prior LP experience and randomized them to either a simulation or a control group.26 Those in the simulation group received instruction using a mannequin, and those in the control group received clinical training through hospital rotations. Both groups received an email containing literature-based information on the procedure as well as a self-assessment questionnaire before participating in either educational program.
The study showed that those students who received simulation training had a success rate of 67% on their first LP on a live patient compared with a success rate of 14% in those with traditional training. Students receiving simulation training required less assistance during the procedure from a supervisor and had higher satisfaction rates and confidence in their procedural skills.26
Another study of 128 medical students at the University of Pittsburgh found that a hybrid LP simulation significantly improved students’ confidence and perceived skill in performing LPs, obtaining informed consent, and electronic order entry. For example, confidence with LP increased from 5.95% presimulation to 90% postsimulation, with 58.24% of students reporting an improvement from minimal or no confidence to average or better (P < .001). Similarly, the proportion of students who felt able to perform LP with minimal or no assistance rose from 0% to 38.57% (P < .001). Confidence and perceived skill in obtaining informed consent and electronic order entry also saw significant gains. Although real-world skill assessments were limited by low survey response rates, preceptor evaluations and follow-up surveys suggested that students who participated in the simulation were more likely to perform these tasks independently or with minimal supervision during clinical rotations.27
Research on simulation training involving nonneurology residents is also encouraging. One study compared the LP skills of traditionally trained neurology residents (PGY-2 to PGY-4) to internal medicine residents (PGY-1) who underwent simulation on a mannequin.28 The internal medicine residents first underwent a pretest on LP performance, watched an educational video, underwent an LP demonstration, and practiced on a mannequin with feedback. The neurology residents completed the checklist-style pretest and performed an LP on a mannequin. Internal medicine residents were found to increase their pretest scores from a mean of 46.3% to 95.7% following training, whereas neurology residents scored a mean of 65.4%. More than half of neurology residents were unable to identify the correct anatomic location or standard cerebrospinal fluid (CSF) tests to be ordered on a routine LP.28
A pediatric resident study in Canada found that following simulation-based training, LP procedural skill improved in 15 of 16 residents tested, and PGY-1 residents showed a reduction in anxiety related to performing the procedure.29
Virtual Reality
An additional tool for simulation is the use of virtual reality (VR) in combination with mannequins. A French study used videos of LPs on actual patients, from equipment set up to final CSF collection and termination of the procedure.30 These videos included a 360-degree view of the procedure. The short video was administered through a VR device (the Oculus Go headset by Microsoft) or by a YouTube video (if VR was not desired).
Participants in the study watched the video then performed an LP on a mannequin. Those who used the VR option had minimal adverse effects (eg, low rates of cybersickness, blurred vision, nausea) and high satisfaction regarding their training environment.30Another VR-based program is the vascular intervention system trainer, which allows clinicians to use endovascular devices and simulate procedures such as thrombectomies. VR simulation is used for trainees and to retrain experienced physicians in performance of high-risk procedures.31
Fundoscopic and Ultrasound Simulations
The AR403 eye stimulator device for fundoscopic examinations is a mannequin-based simulation.32 In a single-center, prospective, single-blind study of neurology and pediatric neurology residents, trainees were split into control and intervention groups, with the intervention group receiving simulator training. Both groups received video lectures on fundoscopy techniques. Pre- and postintervention measurements included knowledge, skill, and total scores on the skills assessment. Of the 48 trainees who participated, the intervention group demonstrated significantly higher increases in skills (P = .01) and total (P = .02) scores, although knowledge scores did not improve. The intervention group also reported higher comfort levels, higher confidence, and higher success rates.
Areas that would benefit from simulation training and development include ultrasound training, such as transcranial Doppler evaluation. In a national survey of residents in anesthesia and critical care, trainees reported that simulation was not frequently used in ultrasound training and that bedside teaching was more common. Interestingly, there was a discrepancy between the opinions of residents and program directors. The program directors felt simulation was in fact used (18.2% of program directors reported this vs 5.3% of trainees).33
A new program, the NewroSim (Gaumard), is a computer-based model of cerebral perfusion that may be a useful tool in this setting. It can simulate blood flow velocities, including pathologic ones, both with a mannequin or without.34
Another potential area for development is the use of mannequins to teach botulinum toxin injections for migraine, dystonia and spasticity in a training environment This is typically led by pharmaceutical representatives who are not necessarily clinicians. Residents and fellows may benefit instead from clinician-led education during their training programs.
Simulation in Patient Communication
Simulation provides a realistic environment for teaching rapid decision-making, leadership, and appropriate management of acutely ill neurologic patients; this includes the communication skills needed in response to neurologic injury.35 Simulation can be particularly useful in situations involving brain death determination, where the communication techniques differ significantly from those used in shared decision-making. Simulation provides a low-stakes setting for clinicians to practice the process of brain death determination and communication, leading to improved confidence and knowledge.36
In the context of acute neurologic emergencies, simulation exercises have been used to investigate the consistency of prognostication across a spectrum of neurology physicians. These exercises revealed that acute neuroprognostication is highly variable and often inaccurate among neurology clinicians, suggesting a potential area for improvement through further simulation training.37
FUTURE DIRECTIONS
Simulation education in neurology can be directed towards learners at all levels, including medical students, residents, fellows, nurses, and medical technologists. In addition, simulation has great value to different disciplines, including emergency medicine, intensive care, and psychiatry. In our view simulation is not being used to full potential in neurology.
Simulation can be used to expose clinicians to rare pathology, play an integral role in competency-based evaluations, and serve as the foundation for simulation-based neurology curriculums, teleneurology simulation training programs, and team training for neurologic emergencies.38Another under-recognized aspect of neurology education is teaching interpersonal communication and professionalism. A survey conducted at a neurology department (20 residents and 73 faculty respondents) asked about residents’ comfort level in performing a number of interpersonal communication and professionalism tasks.38 While none of the residents said they were “very uncomfortable” with these tasks, only 1 resident reported being “very comfortable.” In addition, fewer than 50% noted that they had been directly observed by a faculty member while performing these tasks. The results prompted the facility to develop a simulation curriculum that including observation and feedback from 8 objective structured clinical examinations at a simulation center. A standardized professional simulated the role of a patient, caregiver, medical student, or a faculty member. Residents indicated in postsimulation surveys that it was very useful, and a majority voted for the activity to be repeated for future classes.38
Simulation models may also provide a more objective method to evaluate neurology residents. Accreditation Council for Graduate Medical Education has provided Milestones that are used for assessment of neurology residents. Most of the programs rely on end-of-rotation faculty evaluations. These are subjective evaluations, rely on chance evaluations and may not reflect the exact caliber of a trainee in different clinical situations. Simulation models can serve as alternatives to provide an objective and accurate assessment of resident’s competency in different neurologic scenarios.
In a study of PGY-4 neurology residents from 3 tertiary care academic medical centers were evaluated using simulation-based assessment. Their skills in identifying and managing status epilepticus were assessed via a simulation-based model and compared with clinical experience. No graduating neurology residents were able to meet or exceed the minimum passing score during the testing. It was suggested that end-of-rotation evaluations are inadequate for assigning level of Milestones.24 To move forward with use of simulation-based assessments, these models need to be trialed more extensively and validated.
Morris et al developed simulations for assessment in neurocritical care.39 Ten evaluative simulation cases were developed. Researchers reported on 64 trainee participants in 274 evaluative simulation scenarios. The participants were very satisfied with the cases, found them to be very realistic and appropriately difficult. Interrater reliability was acceptable for both checklist action items and global rating scales. The researchers concluded that they were able to demonstrate validity evidence via the 10 simulation cases for assessment in neurologic emergencies.39 It is the authors’ belief that the future of residents’ competency assessment should include more widespread use of similar simulation models.
Finally, VR and augmented reality (AR) have shown promise in various fields, including neurology. In neurology, these technologies are being explored for applications in rehabilitation, therapy, and medical training. Ongoing research aims to leverage these technologies for improved patient outcomes and medical education. Virtual simulations can recreate neurologic scenarios, allowing learners to interact with 3-dimensional (3D) models of the brain or experience virtual patient cases. AR can enhance traditional learning materials by overlaying digital information onto real-world objects, aiding in the understanding of complex neuroanatomy and medical concepts. These technologies contribute to more engaging and effective neurology education.39In a study of 84 graduate medical students divided into 3 groups, the first group attended a traditional lecture on neuroanatomy, the second group was shown VR-based 3D images, and the third group had a VR-based, interactive and stereoscopic session.40 Groups 2 and 3 showed the highest mean scores in evaluations and differed significantly from Group 1 (P < .05). Groups 2 and 3 did not differ significantly from each other. The researchers concluded that VR-based resources for teaching neuroanatomy fostered significantly higher learning when compared to the traditional methods.40
- Corvetto M, Bravo MP, Montaña R, et al. Simulación en educación médica: una sinopsis. Rev Med Chil. 2013;141:70-79. doi:10.4067/S0034-98872013000100010
- Lane JL, Slavin S, Ziv A. Simulation in medical education: a review. Simul Gaming. 2001;32:297-314. doi:10.1177/104687810103200302
- Bradley P. The history of simulation in medical education and possible future directions. Med Educ. 2006;40:254-262. doi:10.1111/j.1365-2929.2006.02394.x
- Jones F, Passos-Neto C, Melro Braghiroli O. Simulation in medical education: brief history and methodology. Princ Pract Clin Res J. 2015;1:46-54. doi:10.21801/ppcrj.2015.12.8
- Issenberg SB. Simulation technology for health care professional skills training and assessment. JAMA. 1999;28:861-866. doi:10.1001/jama.282.9.861
- McGaghie WC, Miller GE, Sajid AW, et al. Competency-based curriculum development on medical education: an introduction. Public Health Pap. 1978;68:11-91.
- Barsuk JH, Cohen ER, Feinglass J, et al. Use of simulation-based education to reduce catheter-related bloodstream infections. Arch Intern Med. 2009;169:1420-1423. doi:10.1001/archinternmed.2009.215
- Wayne DB, Didwania A, Feinglass J, et al. Simulation-based education improves quality of care during cardiac arrest team responses at an academic teaching hospital: a case-control study. Chest. 2008;133:56-61. doi:10.1378/chest.07-0131
- McGaghie WC, Issenberg SB, Cohen ER, et al. Does simulation-based medical education with deliberate practice yield better results than traditional clinical education? A meta-analytic comparative review of the evidence. Acad Med. 2011;86:706-711. doi:10.1097/ACM.0b013e318217e119
- Micieli G, Cavallini A, Santalucia P, et al. Simulation in neurology. Neurol Sci. 2015;36:1967-1971. doi:10.1007/s10072-015-2228-8
- Bond WF, Lammers RL, Spillane LL, et al. The use of simulation in emergency medicine: a research agenda. Acad Emerg Med. 2007;14:353-363. doi:10.1197/j.aem.2006.11.02112.
- McLaughlin SA, Doezema D, Sklar DP. Human simulation in emergency medicine training: a model curriculum. Acad Emerg Med. 2002;9:1310-1318. doi:10.1111/j.1553-2712.2002.tb01593.x
- Howard SK, Gaba DM, Fish KJ, et al. Anesthesia crisis resource management training: teaching anesthesiologists to handle critical incidents. Aviat Space Environ Med. 1992;63:763-770.
- Gaba DM. Anaesthesiology as a model for patient safety in health care. BMJ. 2000;320:785-788. doi:10.1136/bmj.320.7237.785
- Sedlack RE, Kolars JC. Computer simulator training enhances the competency of gastroenterology fellows at colonoscopy: results of a pilot study. Am J Gastroenterol. 2004;99:33-37. doi:10.1111/j.1572-0241.2004.04007.x
- Tchopev ZN, Nelson AE, Hunninghake JC, et al. Curriculum innovations: high-fidelity simulation of acute neurology enhances rising resident confidence: results from a multicohort study. Neurol Educ. 2022;1:e200022. doi:10.1212/ne9.0000000000200022
- Mehta T, Strauss S, Beland D, et al. Stroke simulation improves acute stroke management: a systems-based practice experience. J Grad Med Educ. 2018;10:57-62. doi:10.4300/JGME-D-17-00167.1
- Pergakis MB, Chang WTW, Tabatabai A, et al. Simulation-based assessment of graduate neurology trainees’ performance managing acute ischemic stroke. Neurology. 2021;97:e2414-e2422. doi:10.1212/WNL.0000000000012972
- Casolla B. Simulation for neurology training: acute setting and beyond. Rev Neurol (Paris). 2021;177:1207-1213. doi:10.1016/j.neurol.2021.03.008
- Casolla B, de Leciñana MA, Neves R, et al. Simulation training programs for acute stroke care: Objectives and standards of methodology. Eur Stroke J. 2020;5:328-335. doi:10.1177/2396987320971105
- Zolnierek KB, Dimatteo MR. Physician communication and patient adherence to treatment: a meta-analysis. Med Care. 2009;47:826-834.doi:10.1097/MLR.0b013e31819a5acc
- Patel RA, Mohl L, Paetow G, Maiser S. Acute neuromuscular respiratory weakness due to acute inflammatory demyelinating polyneuropathy (AIDP): a simulation scenario for neurology providers. MedEdPORTAL. 2019;15:10811. doi:10.15766/mep_2374-8265.10811
- Mikhaeil-Demo Y, Barsuk JH, Culler GW, et al. Use of a simulation-based mastery learning curriculum for neurology residents to improve the identification and management of status epilepticus. Epilepsy Behav. 2020;111:107247. doi:10.1016/j.yebeh.2020.107247
- Mikhaeil-Demo Y, Holmboe E, Gerard EE, et al. Simulation-based assessments and graduating neurology residents’ milestones: status epilepticus milestones. J Grad Med Educ. 2021;13:223-230. doi:10.4300/JGME-D-20-00832.1
- Hocker S, Wijdicks EFM, Feske SK, et al. Use of simulation in acute neurology training: point and counterpoint. Ann Neurol. 2015;78:337-342. doi:10.1002/ana.24473
- Gaubert S, Blet A, Dib F, et al. Positive effects of lumbar puncture simulation training for medical students in clinical practice. BMC Med Educ. 2021;21:1-6. doi:10.1186/S12909-020-02452-327.
- Yanta C, Knepper L, Van Deusen R, et al. The use of hybrid lumbar puncture simulation to teach entrustable professional activities during a medical student neurology clerkship. MedEdPublish (2016). 2021;9:266. doi:10.15694/mep.2020.000266.2
- Barsuk JH, Cohen ER, Caprio T, et al. Simulation-based education with mastery learning improves residents’ lumbar puncture skills. Neurology. 2012;79:132-137. doi:10.1212/WNL.0B013E31825DD39D
- McMillan HJ, Writer H, Moreau KA, et al. Lumbar puncture simulation in pediatric residency training: improving procedural competence and decreasing anxiety. BMC Med Educ. 2016;16:198. doi:10.1186/S12909-016-0722-1
- Vrillon A, Gonzales-Marabal L, Ceccaldi PF, et al. Using virtual reality in lumbar puncture training improves students learning experience. BMC Med Educ. 2022;22:244. doi:10.1186/S12909-022-03317-7
- Liebig T, Holtmannspötter M, Crossley R, et al. Metric-based virtual reality simulation: a paradigm shift in training for mechanical thrombectomy in acute stroke. Stroke. 2018;49:e239-e242.doi:10.1161/STROKEAHA.118.021089
- Gupta DK, Khandker N, Stacy K, et al. Utility of combining a simulation-based method with a lecture-based method for fundoscopy training in neurology residency. JAMA Neurol. 2017;74:1223-1227. doi:10.1001/JAMANEUROL.2017.2073
- Mongodi S, Bonomi F, Vaschetto R, et al. Point-of-care ultrasound training for residents in anaesthesia and critical care: results of a national survey comparing residents and training program directors’ perspectives. BMC Med Educ. 2022;22:647. doi:10.1186/S12909-022-03708-W
- Morris NA, Czeisler BM, Sarwal A. Simulation in neurocritical care: past, present, and future. Neurocrit Care. 2019;30:522-533. doi:10.1007/S12028-018-0629-2
- Wijdicks EFM, Hocker SE. A future for simulation in acute neurology. Semin Neurol. 2018;38:465-470. doi:10.1055/s-0038-1666986
- Kramer NM, O’Mahony S, Deamant C. Brain death determination and communication: an innovative approach using simulation and standardized patients. J Pain Symptom Manage. 2022;63:e765-e772. doi:10.1016/j.jpainsymman.2022.01.020
- Sloane KL, Miller JJ, Piquet A, et al. Prognostication in acute neurological emergencies. J Stroke Cerebrovasc Dis. 2022;31:106277. doi:10.1016/J.JSTROKECEREBROVASDIS.2021.106277
- Kurzweil AM, Lewis A, Pleninger P, et al. Education research: teaching and assessing communication and professionalism in neurology residency with simulation. Neurology. 2020;94:229-232. doi:10.1212/WNL.0000000000008895
- Morris NA, Chang WT, Tabatabai A, et al. Development of neurological emergency simulations for assessment: content evidence and response process. Neurocrit Care. 2021;35:389-396. doi:10.1007/S12028-020-01176-Y
- De Faria JWV, Teixeira MJ, De Moura Sousa Júnior L, et al. Virtual and stereoscopic anatomy: when virtual reality meets medical education. J Neurosurg. 2016;125:1105-1111. doi:10.3171/2015.8.JNS141563
- Corvetto M, Bravo MP, Montaña R, et al. Simulación en educación médica: una sinopsis. Rev Med Chil. 2013;141:70-79. doi:10.4067/S0034-98872013000100010
- Lane JL, Slavin S, Ziv A. Simulation in medical education: a review. Simul Gaming. 2001;32:297-314. doi:10.1177/104687810103200302
- Bradley P. The history of simulation in medical education and possible future directions. Med Educ. 2006;40:254-262. doi:10.1111/j.1365-2929.2006.02394.x
- Jones F, Passos-Neto C, Melro Braghiroli O. Simulation in medical education: brief history and methodology. Princ Pract Clin Res J. 2015;1:46-54. doi:10.21801/ppcrj.2015.12.8
- Issenberg SB. Simulation technology for health care professional skills training and assessment. JAMA. 1999;28:861-866. doi:10.1001/jama.282.9.861
- McGaghie WC, Miller GE, Sajid AW, et al. Competency-based curriculum development on medical education: an introduction. Public Health Pap. 1978;68:11-91.
- Barsuk JH, Cohen ER, Feinglass J, et al. Use of simulation-based education to reduce catheter-related bloodstream infections. Arch Intern Med. 2009;169:1420-1423. doi:10.1001/archinternmed.2009.215
- Wayne DB, Didwania A, Feinglass J, et al. Simulation-based education improves quality of care during cardiac arrest team responses at an academic teaching hospital: a case-control study. Chest. 2008;133:56-61. doi:10.1378/chest.07-0131
- McGaghie WC, Issenberg SB, Cohen ER, et al. Does simulation-based medical education with deliberate practice yield better results than traditional clinical education? A meta-analytic comparative review of the evidence. Acad Med. 2011;86:706-711. doi:10.1097/ACM.0b013e318217e119
- Micieli G, Cavallini A, Santalucia P, et al. Simulation in neurology. Neurol Sci. 2015;36:1967-1971. doi:10.1007/s10072-015-2228-8
- Bond WF, Lammers RL, Spillane LL, et al. The use of simulation in emergency medicine: a research agenda. Acad Emerg Med. 2007;14:353-363. doi:10.1197/j.aem.2006.11.02112.
- McLaughlin SA, Doezema D, Sklar DP. Human simulation in emergency medicine training: a model curriculum. Acad Emerg Med. 2002;9:1310-1318. doi:10.1111/j.1553-2712.2002.tb01593.x
- Howard SK, Gaba DM, Fish KJ, et al. Anesthesia crisis resource management training: teaching anesthesiologists to handle critical incidents. Aviat Space Environ Med. 1992;63:763-770.
- Gaba DM. Anaesthesiology as a model for patient safety in health care. BMJ. 2000;320:785-788. doi:10.1136/bmj.320.7237.785
- Sedlack RE, Kolars JC. Computer simulator training enhances the competency of gastroenterology fellows at colonoscopy: results of a pilot study. Am J Gastroenterol. 2004;99:33-37. doi:10.1111/j.1572-0241.2004.04007.x
- Tchopev ZN, Nelson AE, Hunninghake JC, et al. Curriculum innovations: high-fidelity simulation of acute neurology enhances rising resident confidence: results from a multicohort study. Neurol Educ. 2022;1:e200022. doi:10.1212/ne9.0000000000200022
- Mehta T, Strauss S, Beland D, et al. Stroke simulation improves acute stroke management: a systems-based practice experience. J Grad Med Educ. 2018;10:57-62. doi:10.4300/JGME-D-17-00167.1
- Pergakis MB, Chang WTW, Tabatabai A, et al. Simulation-based assessment of graduate neurology trainees’ performance managing acute ischemic stroke. Neurology. 2021;97:e2414-e2422. doi:10.1212/WNL.0000000000012972
- Casolla B. Simulation for neurology training: acute setting and beyond. Rev Neurol (Paris). 2021;177:1207-1213. doi:10.1016/j.neurol.2021.03.008
- Casolla B, de Leciñana MA, Neves R, et al. Simulation training programs for acute stroke care: Objectives and standards of methodology. Eur Stroke J. 2020;5:328-335. doi:10.1177/2396987320971105
- Zolnierek KB, Dimatteo MR. Physician communication and patient adherence to treatment: a meta-analysis. Med Care. 2009;47:826-834.doi:10.1097/MLR.0b013e31819a5acc
- Patel RA, Mohl L, Paetow G, Maiser S. Acute neuromuscular respiratory weakness due to acute inflammatory demyelinating polyneuropathy (AIDP): a simulation scenario for neurology providers. MedEdPORTAL. 2019;15:10811. doi:10.15766/mep_2374-8265.10811
- Mikhaeil-Demo Y, Barsuk JH, Culler GW, et al. Use of a simulation-based mastery learning curriculum for neurology residents to improve the identification and management of status epilepticus. Epilepsy Behav. 2020;111:107247. doi:10.1016/j.yebeh.2020.107247
- Mikhaeil-Demo Y, Holmboe E, Gerard EE, et al. Simulation-based assessments and graduating neurology residents’ milestones: status epilepticus milestones. J Grad Med Educ. 2021;13:223-230. doi:10.4300/JGME-D-20-00832.1
- Hocker S, Wijdicks EFM, Feske SK, et al. Use of simulation in acute neurology training: point and counterpoint. Ann Neurol. 2015;78:337-342. doi:10.1002/ana.24473
- Gaubert S, Blet A, Dib F, et al. Positive effects of lumbar puncture simulation training for medical students in clinical practice. BMC Med Educ. 2021;21:1-6. doi:10.1186/S12909-020-02452-327.
- Yanta C, Knepper L, Van Deusen R, et al. The use of hybrid lumbar puncture simulation to teach entrustable professional activities during a medical student neurology clerkship. MedEdPublish (2016). 2021;9:266. doi:10.15694/mep.2020.000266.2
- Barsuk JH, Cohen ER, Caprio T, et al. Simulation-based education with mastery learning improves residents’ lumbar puncture skills. Neurology. 2012;79:132-137. doi:10.1212/WNL.0B013E31825DD39D
- McMillan HJ, Writer H, Moreau KA, et al. Lumbar puncture simulation in pediatric residency training: improving procedural competence and decreasing anxiety. BMC Med Educ. 2016;16:198. doi:10.1186/S12909-016-0722-1
- Vrillon A, Gonzales-Marabal L, Ceccaldi PF, et al. Using virtual reality in lumbar puncture training improves students learning experience. BMC Med Educ. 2022;22:244. doi:10.1186/S12909-022-03317-7
- Liebig T, Holtmannspötter M, Crossley R, et al. Metric-based virtual reality simulation: a paradigm shift in training for mechanical thrombectomy in acute stroke. Stroke. 2018;49:e239-e242.doi:10.1161/STROKEAHA.118.021089
- Gupta DK, Khandker N, Stacy K, et al. Utility of combining a simulation-based method with a lecture-based method for fundoscopy training in neurology residency. JAMA Neurol. 2017;74:1223-1227. doi:10.1001/JAMANEUROL.2017.2073
- Mongodi S, Bonomi F, Vaschetto R, et al. Point-of-care ultrasound training for residents in anaesthesia and critical care: results of a national survey comparing residents and training program directors’ perspectives. BMC Med Educ. 2022;22:647. doi:10.1186/S12909-022-03708-W
- Morris NA, Czeisler BM, Sarwal A. Simulation in neurocritical care: past, present, and future. Neurocrit Care. 2019;30:522-533. doi:10.1007/S12028-018-0629-2
- Wijdicks EFM, Hocker SE. A future for simulation in acute neurology. Semin Neurol. 2018;38:465-470. doi:10.1055/s-0038-1666986
- Kramer NM, O’Mahony S, Deamant C. Brain death determination and communication: an innovative approach using simulation and standardized patients. J Pain Symptom Manage. 2022;63:e765-e772. doi:10.1016/j.jpainsymman.2022.01.020
- Sloane KL, Miller JJ, Piquet A, et al. Prognostication in acute neurological emergencies. J Stroke Cerebrovasc Dis. 2022;31:106277. doi:10.1016/J.JSTROKECEREBROVASDIS.2021.106277
- Kurzweil AM, Lewis A, Pleninger P, et al. Education research: teaching and assessing communication and professionalism in neurology residency with simulation. Neurology. 2020;94:229-232. doi:10.1212/WNL.0000000000008895
- Morris NA, Chang WT, Tabatabai A, et al. Development of neurological emergency simulations for assessment: content evidence and response process. Neurocrit Care. 2021;35:389-396. doi:10.1007/S12028-020-01176-Y
- De Faria JWV, Teixeira MJ, De Moura Sousa Júnior L, et al. Virtual and stereoscopic anatomy: when virtual reality meets medical education. J Neurosurg. 2016;125:1105-1111. doi:10.3171/2015.8.JNS141563
Sim and Learn: Simulation and its Value in Neurology Education
Sim and Learn: Simulation and its Value in Neurology Education
Updates in Multiple Sclerosis Imaging
Updates in Multiple Sclerosis Imaging
Multiple sclerosis (MS) is a complex, chronic immune-mediated disease of the central nervous system characterized by focal inflammation, demyelination, and neurodegeneration. Magnetic resonance imaging (MRI), first incorporated into the McDonald Criteria for the diagnosis of MS in 2001, is an integral tool in the diagnosis, prognosis, and therapeutic monitoring of people with MS (PwMS).1
MRI research in MS is rapidly expanding and offers insights into the pathophysiology of MS with important implications for the routine clinical care of PwMS. At the Consortium of Multiple Sclerosis Centers 2024 Annual Meeting, the US Department of Veterans Affairs (VA) MS Centers of Excellence hosted an educational symposium highlighting MRI biomarkers in MS, including T2-lesions, chronic black holes (cBHs), brain atrophy, paramagnetic rim lesions (PRLs), and the central vein sign (CVS). The symposium also provided a brief overview of quantitative MRI techniques used to characterize MS lesion severity and research applications of these techniques. This clinical review summarizes the main points of that symposium with the goal of introducing key concepts to federal health care practitioners caring for PwMS.
MRI Biomarkers in MS
T2-lesions, Chronic Black Holes, and Brain Atrophy
Focal immune-mediated inflammation and demyelination in MS may be detected by MRI as hyperintense foci on T2-weighted (T2-w) imaging (eg, T2-w turbo spin echo or T2-w fluid attenuated inversion recovery sequences). These T2-lesions, critical for diagnosing MS, are typically ovoid and occur in the periventricular, juxtacortical, infratentorial spinal cord white matter (Figure 1A). T2-lesion number and volume show some association with disability and optic nerve.
Wattjes et al highlight 2 cases to demonstrate this point: a man aged 52 years with MS for 23 years and a woman aged 50 years with MS for 11 years. Despite having MS for a much shorter duration, the woman had worse disability due to a higher lesion number and volume.2 T2-lesion volume also impacts disability progression in PwMS. Gauthier et al compared the probability of progression in 3 women, all of whom were aged 39 years and had MS for 6 years. The profile with highest probability of disability progression had the highest quartile of T2-lesion volume.3 T2-lesion volume over 2 years correlates with worse scores on disability metrics such as the MS functional composite, paced auditory serial addition task, and brain volume.4 A 2024 systematic review and meta-analysis demonstrated that T2-lesion volume is significantly correlated with clinical disability in PwMS.5
Select T2-lesions are also hypointense on T1-w spin echo images and are known as cBHs (Figure 1B). Histologically, T2-lesions with cBHs have more severe architectural disruption than those without cBHs.6 cBH number and volume are significantly correlated with disability, regardless of the degree of hypointensity on T1-w imaging.5,7 A 10-year longitudinal study demonstrated that cBHs were associated with disease progression after 5 years while T2-lesion volume was not, indicating that cBHs may be a more accurate predictor of disability.8
Brain atrophy, another imaging biomarker of MS, affects both the cerebral white and gray matter. White matter fraction (the volume of white matter relative to the intracranial compartment volume) and gray matter fraction (the volume of gray matter relative to the intracranial compartment) are significantly lower among PwMS compared with healthy controls. In addition, gray matter fraction is lower among patients with primary and secondary progressive MS compared with those with relapsing-remitting MS, clinically isolated syndrome (CIS), and radiologically isolated syndrome (RIS). Gray matter fraction is also correlated with several motor and cognitive disability indices.9
Paramagnetic Rim Lesions
Neurologic worsening in PwMS occurs by 2 distinct mechanisms: relapse-associated worsening, a stepwise worsening of symptoms due to incomplete recovery following a relapse; and progression independent of relapse activity (PIRA), which is an irreversible neurologic deterioration in the absence of clinical or radiological relapses.10 PIRA is associated with neurodegeneration and predominates in both primary and secondary progressive MS. However, recent data demonstrated that PIRA may contribute to as much as 50% of disability worsening in relapsing MS and occurs early in the RMS disease course.10,11 Current high-efficacy disease modifying therapy, such as ocrelizumab, are extraordinarily successful at preventing focal inflammation and relapses but are less effective for preventing the slow march of disability progression characterizing PIRA.12,13 The prevention of PIRA is therefore an unmet treatment need.
Chronic active lesions (CALs) are an important driver of PIRA. When an acute gadolinium-enhancing lesion develops in PwMS, there are 3 possible fates of this lesion. The lesion may become chronically inactive, remyelinate, or transition to CALs.14 The histopathologic signature of CALs is compartmentalized, low-grade inflammation behind an intact blood-brain barrier with evidence of both active and chronic components.15 CALs may be found not only in cerebral white matter but also in the cerebral cortex and spinal cord.16,17 Combined MRI and histopathological studies have shown that iron-laden microglia/macrophages can be detected by susceptibility-based MRI as a rim of paramagnetic signal surrounding select T2-lesions.19 These PRLs represent an in vivo imaging biomarker of CAL (Figure 1C). According to the North American Imaging in MS Cooperative (NAIMS) consensus criteria, a PRL must surround at least two-thirds of the outer edge of a T2-lesion, be visible in ≥ 2 consecutive MRI slices, and cannot be contrast enhancing.20
PRLs can be visualized on multiple susceptibility-based imaging methods, including multiecho derived R2*/T2*, phase maps, susceptibility-weighted imaging, and quantitative susceptibility mapping.21-23 Retrospective analyses have shown no significant differences in sensitivity across these imaging modalities.24 Although first visualized with 7T MRI, PRLs may also be detected by 1.5T and 3T MRI with comparable sensitivities.25-27 However, there remains a significant knowledge gap regarding the accuracy of each imaging modality. Systematic, prospectively designed studies are needed to ascertain the comparative value of each method.
The presence of PRL is a poor prognostic indicator. PwMS without PRLs have higher levels of disability, are more likely to progress, and demonstrate greater gray matter atrophy and cognitive dysfunction when compared with PwMS with PRLs.27-29 Lesions with PRL tend to slowly expand, exhibit greater demyelination, and have diminished white matter integrity.21,22,30
PRLs may also be used as a diagnostic tool. PRLs are highly specific for MS/CIS with a 99.7% specificity and 98.4% positive predictive value, although the sensitivity is limited to 24%.31 Taken together, these data indicate that the presence of a PRL substantially increases the likelihood of an MS/CIS diagnosis, whereas the absence of a PRL does not exclude these diagnoses.
Several unanswered questions remain: Why do select acute MS lesions transition to CALs? How may investigators utilize PRLs as outcome measures in future clinical trials? How should PRLs be incorporated into the routine care of PwMS? As the role of this imaging biomarker is clarified both in the research and clinical settings, clinicians caring for PwMS can expect to increasingly encounter the topic of PRLs in the near future.
Central Vein Sign
A CVS is defined by the presence of a central vessel within a demyelinating plaque (Figure 1D). As early as the 1820s, MS plaques on gross pathology were noted to follow the course of a vessel. Early histological studies reported that up to 91% of MS plaques had a central vessel present.32 Lesion formation is dependent on the movement of lymphocytes and other inflammatory cells from the systemic circulation across the blood brain barrier into the perivascular space, a privileged site where immune cells interact with antigen presenting cells to launch an inflammatory cascade and eventual demyelinating lesion.33
CVS can be visualized on 1.5T, 3T and 7T MRI. However, 7T MRI is superior to 3T in the detection of CVS, with 85% of MS lesions having CVS visible compared with 45% on 3T.34 With advances in 7T MRI, fluid attenuated inversion recovery and T2* susceptibility, weighted sequences can be overlaid, allowing simultaneous visualization of the vessel and the demyelinating lesion. With higher density of parenchymal veins in the periventricular regions, the CVS is most seen in lesions of this territory but can also be present in juxtacortical, thalamic and infratentorial lesions with decreasing prevalence as these approach the cortex.35
MS lesions are more likely to have CVS than T2 hyperintense white matter lesions of other causes, with a large study reporting 78% of MS lesions were CVS positive. Further, CVS positive lesions can be found across all MS phenotypes including relapsing remitting, primary progressive, and secondary progressive.35 The CVS is also specific to MS lesions and is an effective tool for differentiating MS lesions from other common causes of T2 hyperintense lesions including chronic ischemic white matter disease,36 migraines,37 neuromyelitis optica spectrum disorders,38,39 Susac syndrome,40 and systemic autoimmune diseases (Behcet disease, systemic lupus erythematosus, and antiphospholipid syndrome).41
With CVS emerging as a promising radiographic biomarker for MS, NAIMS issued a consensus statement on necessary properties of a CVS. These criteria included appearance of a thin hypointense line or small dot, visualized in ≥ 2 perpendicular planes, with diameter < 2 mm, and running partially or entirely through the center of the lesion. They also clarified that lesions < 3 mm, confluent lesions, lesions with multiple vessels present or poorly visualized lesions were excluded.42
A shared CVS definition was a necessary step toward routine use of CVS as a radiographic biomarker and its incorporation in the 2024 revised McDonald criteria.43 Remaining limitations including 7T MRI is primarily available in research settings and the lack of consensus on a diagnostic threshold. There have been many proposed methods, including a 40% cut off,44 60% cut off,45 and Select 3* or Select 6* methods.46 The goal of each method is to optimize sensitivity and specificity while not compromising efficiency of MRI review for both neurologists and radiologists.
The CVS has significant potential as a radiographic biomarker for MS and may allow the early stages of MS to be differentiated from other common causes of white matter lesions on MRI. However, it remains unclear whether CVS holds prognostic value for patients, if CVS is suggestive of differing underlying pathology, or if the presence of a CVS is dynamic over time. Progress in these areas is anticipated as CVS is incorporated into routine clinical practice.
Quantitative MRI Techniques
In the research setting, several imaging modalities can be used to quantify the degree of microstructural injury in PwMS. The goal of these methods is to identify and quantify myelin and axonal damage, the major drivers of neurodegeneration. Among these methods, diffusion-based imaging is a measure of the amount of diffusion or fluid mobility across the tissues of the brain.47 Diffusion-weighted imaging (DWI) yields several parametric maps including axial diffusivity (AD), radial diffusivity (RD), and mean diffusivity (Figure 2 A, B, and C). These parametric maps provide information on different directions of water molecules’ movements. Myelin surrounds the axons preventing water molecules diffusion perpendicular to axons (RD) while axonal content prevents water diffusion horizontal to the axons (AD).Thus, AD is considered more specific to axonal injury, whereas RD is specific to myelin content.48 A higher value of any of these metrics is associated with a higher degree of tissue injury.
Although sensitive to axonal and myelin injury, AD and RD computed from single b-shell DWI experience several limitations including being affected by nonpathologic factors such as fiber orientation, distribution, and crossing, and by various nonmyelin specific pathologies including fluid accumulation during inflammation, myelin sheath thickness, and axonal intactness.48 Several multi b-shell methods have been developed to overcome diffusion imaging limitations. For example, work at the Nashville VA MS Center of Excellence has focused on the use of the multicompartment diffusion MRI with spherical mean technique (SMT). This method removes the orientation dependency of the diffusion MRI signal, increasing the signal-to-noise ratio and reducing biases from fiber undulation, crossing, and dispersion.49 SMT generates the apparent axonal volume fraction (Vax), which is a direct measure of axonal integrity with lower values indicating lower axonal content and higher tissue destruction (Figure 2D). Vax was previously validated in MS as a measure of axonal integrity.49
In terms of myelin, several other specific measures have been developed. Magnetization transfer ratio (MTR) is another measure of tissue integrity that has been validated as a measure of tissue injury in MS (Figure 2E).50,51 Zheng et al found that the percentage of lesions with low MTR was significantly higher among patients whose disease disability progressed compared with patients who did not.52Selective inversion recovery with quantitative magnetization transfer (SIR-qMT) was developed to account for the limitations of MTR, including its sensitivity to edema and axonal density.52 Germane to myelin measurements, SIR-qMT generates the macromolecular to free size ratio (PSR). PSR represents the ratio of protons bound to macromolecules (myelin) to free protons (Figure 2F). PSR is considered a marker of myelin integrity, with lower values correlating with disability severity and indicating higher tissue damage and lower myelin content. Previous studies from the Nashville VA MS Center of Excellence validated the use of SIR-qMT among patients with MS, CIS, RIS, and healthy controls.53
Quantitative MRI has several research applications in the field of MS. We demonstrated that PRL harbor a higher degree of myelin injury indicated by PSR compared with rimless lesions.54 These MRI techniques are also helpful to investigate tissues surrounding the lesions, called normal appearing white matter (NAWM). Using quantitative MRI techniques such as MTR,52 PSR,53 and Vax,49 investigators have demonstrated that NAWM is injured in PwMS, and proximal NAWM may have higher degree of tissue damage compared with distant NAWM.55
Anticipated Innovations and Challenges
In the field of quantitative MRI, several new techniques are being adopted. Researchers are developing techniques such as myelin water fraction which evaluates the interaction between water and protons to measure myelin content. This is considered an advancement as it takes into account edema resulting from MS injury.56 Another example is multicompartment diffusion imaging, such as standard model imaging,57 and neurite orientation dispersion and density imaging,58 which considers water as an additional compartment compared with the SMT derived Vax. For PRL identification, more advanced methodologic techniques are developing such quantitative susceptibility mapping (QSM), which can detect iron deposits that surround the lesions with relatively high sensitivity and specificity of identifying PRL.59
Despite these innovations, several challenges remain before possible incorporation into the clinical setting. These limitations include longer scan time, familiarity of clinicians in using these maps, higher financial cost, and the necessity of advanced imaging processing skills. Artificial intelligence is a promising tool that may overcome these challenges through creating automated processing pipelines and developing synthetic maps without the need for additional acquisition.60
Conclusions
MRI is the most important tool for diagnosing and treating PwMS. Imaging biomarkers such as T2-lesions, cBHs, brain atrophy, PRLs, and CVS provide insight into the disease’s pathogenesis and are invaluable for the accurate diagnosis and prognostication of MS. Quantitative MRI techniques, while not available in the clinical setting, are important tools for translational research that may help direct the development of future therapeutics. In the near future, clinicians caring for PwMS should expect to encounter these imaging biomarkers more frequently in the clinical setting, especially with the inclusion of PRLs and CVS in the next iteration of the McDonald diagnostic criteria.
McDonald WI, Compston A, Edan G, et al. Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol. 2001;50:121-127. doi:10.1002/ana.1032
Wattjes MP, Steenwijk MD, Stangel M. MRI in the diagnosis and monitoring of multiple sclerosis: an update. Clin Neuroradiol. 2015;25:157-165. doi:10.1007/s00062-015-0430-y
Gauthier SA, Mandel M, Guttmann CR, et al. Predicting short-term disability in multiple sclerosis. Neurology. 2007;68:2059-2065.doi:10.1212/01.wnl.0000264890.97479.b1
Rudick RA, Lee JC, Simon J, Fisher E. Significance of T2 lesions in multiple sclerosis: a 13-year longitudinal study. Ann Neurol. 2006;60:236-242. doi:10.1002/ana.20883
Nabizadeh F, Zafari R, Mohamadi M, et al. MRI features and disability in multiple sclerosis: a systematic review and meta-analysis. J Neuroradiol. 2024;51:24-37. doi:10.1016/j.neurad.2023.11.007
Bagnato F, Jeffries N, Richert ND, et al. Evolution of T1 black holes in patients with multiple sclerosis imaged monthly for 4 years. Brain. 2003;126:1782-1789. doi:10.1093/brain/awg182
Jacobsen C, Hagemeier J, Myhr KM, et al. Brain atrophy and disability progression in multiple sclerosis patients: a 10-year follow-up study. J Neurol Neurosurg Psychiatry. 2014;85:1109-1115. doi:10.1136/jnnp-2013-306906
Rovaris M, Gass A, Bammer R, et al. Diffusion MRI in multiple sclerosis. Neurology. 2005;65:1526-1532. doi:10.1212/01.wnl.0000184471.83948.e0
Fisniku LK, Chard DT, Jackson JS, et al. Gray matter atrophy is related to long-term disability in multiple sclerosis. Ann Neurol. 2008;64:247-254. doi:10.1002/ana.21423
Lublin FD, Häring DA, Ganjgahi H, et al. How patients with multiple sclerosis acquire disability. Brain. 2022;145:3147-3161. doi:10.1093/brain/awac016
Kappos L, Wolinsky JS, Giovannoni G, et al. Contribution of relapse-independent progression vs relapse-associated worsening to overall confirmed disability accumulation in typical relapsing multiple sclerosis in a pooled analysis of 2 randomized clinical trials. JAMA Neurol. 2020;77:1132-1140. doi:10.1001/jamaneurol.2020.1568
Hauser SL, Bar-Or A, Comi G, et al. Ocrelizumab versus interferon beta-1a in relapsing multiple sclerosis. N Engl J Med. 2017;376:221-234. doi:10.1056/NEJMoa1601277
Montalban X, Hauser SL, Kappos L, et al. Ocrelizumab versus placebo in primary progressive multiple sclerosis. N Engl J Med. 2017;376:209-220. doi:10.1056/NEJMoa1606468
Prineas JW, Kwon EE, Cho ES, et al. Immunopathology of secondary-progressive multiple sclerosis. Ann Neurol. 2001;50:646-657. doi:10.1002/ana.1255
Kuhlmann T, Ludwin S, Prat A, Antel J, Brück W, Lassmann H. An updated histological classification system for multiple sclerosis lesions. Acta Neuropathol. 2017;133:13-24. doi:10.1007/s00401-016-1653-y
Pitt D, Boster A, Pei W, et al. Imaging cortical lesions in multiple sclerosis with ultra-high-field magnetic resonance imaging. Arch Neurol. 2010;67:812-818. doi:10.1001/archneurol.2010.148
Gilmore CP, Geurts JJ, Evangelou N, et al. Spinal cord grey matter lesions in multiple sclerosis detected by post-mortem high field MR imaging. Mult Scler. 2009;15:180-188. doi:10.1177/1352458508096876
Lassmann H, Brück W, Lucchinetti CF. The immunopathology of multiple sclerosis: an overview. Brain Pathol. 2007;17:210-218. doi:10.1111/j.1750-3639.2007.00064.x
Bagnato F, Hametner S, Yao B, et al. Tracking iron in multiple sclerosis: a combined imaging and histopathological study at 7 Tesla. Brain. 2011;134:3602-3615. doi:10.1093/brain/awr278
Bagnato F, Sati P, Hemond CC, et al. Imaging chronic active lesions in multiple sclerosis: a consensus statement. Brain. 2024;147:2913-2933. doi:10.1093/brain/awae013
Dal-Bianco A, Grabner G, Kronnerwetter C, et al. Slow expansion of multiple sclerosis iron rim lesions: pathology and 7 T magnetic resonance imaging. Acta Neuropathol. 2017;133:25-42. doi:10.1007/s00401-016-1636-z
Absinta M, Sati P, Schindler M, et al. Persistent 7-tesla phase rim predicts poor outcome in new multiple sclerosis patient lesions. J Clin Invest. 2016;126:2597-2609. doi:10.1172/JCI86198
Gillen KM, Mubarak M, Park C, et al. QSM is an imaging biomarker for chronic glial activation in multiple sclerosis lesions. Ann Clin Transl Neurol. 2021;8:877-886. doi:10.1002/acn3.51338
Ng Kee Kwong KC, Mollison D, Meijboom R, et al. The prevalence of paramagnetic rim lesions in multiple sclerosis: a systematic review and meta-analysis. PLoS One. 2021;16:e0256845. doi:10.1371/journal.pone.0256845
Absinta M, Sati P, Fechner A, et al. Identification of chronic active multiple sclerosis lesions on 3T MRI. AJNR Am J Neuroradiol. 2018;39:1233-1238. doi:10.3174/ajnr.A5660
Hemond CC, Reich DS, Dundamadappa SK. Paramagnetic rim lesions in multiple sclerosis: comparison of visualization at 1.5-T and 3-T MRI. AJR Am J Roentgenol. 2022;219:120-131. doi:10.2214/AJR.21.26777
Altokhis AI, Hibbert AM, Allen CM, et al. Longitudinal clinical study of patients with iron rim lesions in multiple sclerosis. Mult Scler. 2022;28:2202-2211. doi:10.1177/13524585221114750
Choi S, Lake S, Harrison DM. Evaluation of the blood-brain barrier, demyelination, and neurodegeneration in paramagnetic rim lesions in multiple sclerosis on 7 tesla MRI. J Magn Reson Imaging. 2024;59:941-951. doi:10.1002/jmri.28847
Kazimuddin HF, Wang J, Hernandez B, et al. Paramagnetic rim lesions and their relationship with neurodegeneration and clinical disability at the time of multiple sclerosis diagnosis. Poster presented at: 2024 Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS) Forum; February 26-March 2; West Palm Beach, FL.
Rohm Z, Koch C, Kazimuddin H, et al. Longitudinal characterization of paramagnetic rim lesions in early multiple sclerosis. Poster presented at: 2024 Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS) Forum; February 26-March 2; West Palm Beach, FL.
Meaton I, Altokhis A, Allen CM, et al. Paramagnetic rims are a promising diagnostic imaging biomarker in multiple sclerosis. Mult Scler. 2022;28:2212-2220. doi:10.1177/13524585221118677
Fog T. On the vessel-plaque relationships in the brain in multiple sclerosis. Acta Neurol Scand Suppl. 1964;40:9-15.
Ineichen BV, Okar SV, Proulx ST, et al. Perivascular spaces and their role in neuroinflammation. Neuron. 2022;110:3566-3581. doi:10.1016/j.neuron.2022.10.024
Tallantyre EC, Morgan PS, Dixon JE, et al. A comparison of 3T and 7T in the detection of small parenchymal veins within MS lesions. Invest Radiol. 2009;44:491-494. doi:10.1097/RLI.0b013e3181b4c144
Kilsdonk ID, Lopez-Soriano A, Kuijer JP, et al. Morphological features of MS lesions on FLAIR* at 7 T and their relation to patient characteristics. J Neurol. 2014;261:1356-1364. doi:10.1007/s00415-014-7351-6
Tallantyre EC, Dixon JE, Donaldson I, et al. Ultra-high-field imaging distinguishes MS lesions from asymptomatic white matter lesions. Neurology. 2011;76:534-539. doi:10.1212/WNL.0b013e31820b7630
Solomon AJ, Schindler MK, Howard DB, et al. “Central vessel sign” on 3T FLAIR* MRI for the differentiation of multiple sclerosis from migraine. Ann Clin Transl Neurol. 2015;3:82-87. doi:10.1002/acn3.273
Sinnecker T, Dörr J, Pfueller CF, et al. Distinct lesion morphology at 7-T MRI differentiates neuromyelitis optica from multiple sclerosis. Neurology. 2012;79:708-714. doi:10.1212/WNL.0b013e3182648bc8
Kister I, Herbert J, Zhou Y, Ge Y. Ultrahigh-field MR (7 T) imaging of brain lesions in neuromyelitis optica. Mult Scler Int. 2013;2013:398259. doi:10.1155/2013/398259
Wuerfel J, Sinnecker T, Ringelstein EB, et al. Lesion morphology at 7 Tesla MRI differentiates Susac syndrome from multiple sclerosis. Mult Scler. 2012;18:1592-1599. doi:10.1177/1352458512441270
Massacesi L. Perivenular distribution of white matter lesions evaluated by MRI can differentiate MS lesions from inflammatory small vessel diseases. Eur J Neurol. 2016;23:86. doi:10.1212/WNL.86.16_supplement.P6.121
Sati P, Oh J, Constable RT, et al. The central vein sign and its clinical evaluation for the diagnosis of multiple sclerosis: a consensus statement from the North American Imaging in Multiple Sclerosis Cooperative. Nat Rev Neurol. 2016;12:714-722. doi:10.1038/nrneurol.2016.166
Montalban X, Lebrun-Frénay C, Oh J, et al. Diagnosis of multiple sclerosis: 2024 revisions of the McDonald criteria. Lancet Neurol. 2025;24:850-865. doi:10.1016/S1474-4422(25)00270-4
Mistry N, Dixon J, Tallantyre E, et al. Central veins in brain lesions visualized with high-field magnetic resonance imaging: a pathologically specific diagnostic biomarker for inflammatory demyelination in the brain. JAMA Neurol. 2013;70:623-628. doi:10.1001/jamaneurol.2013.1405
Campion T, Smith RJP, Altmann DR, et al. FLAIR* to visualize veins in white matter lesions: a new tool for the diagnosis of multiple sclerosis? Eur Radiol. 2017;27:4257-4263. doi:10.1007/s00330-017-4822-z
Solomon AJ, Watts R, Ontaneda D, et al. Diagnostic performance of central vein sign for multiple sclerosis with a simplified three-lesion algorithm. Mult Scler. 2018;24:750-757. doi:10.1177/1352458517726383
Cercignani M, Bozzali M, Iannucci G, Comi G, Filippi M. Intra-voxel and inter-voxel coherence in patients with multiple sclerosis assessed using diffusion tensor MRI. J Neurol. 2002;249:875-883. doi:10.1007/s00415-002-0752-y
Song SK, Yoshino J, Le TQ, et al. Demyelination increases radial diffusivity in corpus callosum of mouse brain. Neuroimage. 2005;26:132-140. doi:10.1016/j.neuroimage.2005.01.028
Bagnato F, Franco G, Li H, et al. Probing axons using multi-compartmental diffusion in multiple sclerosis. Ann Clin Transl Neurol. 2019;6:1595-1605. doi:10.1002/acn3.50836
Filippi M, Cercignani M, Inglese M, et al. Diffusion tensor magnetic resonance imaging in multiple sclerosis. Neurology. 2001;56:304-311. doi:10.1212/wnl.56.3.304
Bagnato F. Clinical application of magnetization transfer imaging. In: Advanced Neuro MR Techniques and Applications. Elsevier; 2022:403-417. doi:10.1016/B978-0-12-822479-3.00041-5
Zheng Y, Lee JC, Rudick R, Fisher E. Long-term magnetization transfer ratio evolution in multiple sclerosis white matter lesions. J Neuroimaging. 2018;28:191-198. doi:10.1111/jon.12480
Bagnato F, Hametner S, Franco G, et al. Selective inversion recovery quantitative magnetization transfer brain MRI at 7T: clinical and postmortem validation in multiple sclerosis. J Neuroimaging. 2018;28:380-388. doi:10.1111/jon.12511
Clarke MA, Cheek R, Hernandez B, et al. Paramagnetic rim lesions and the central vein sign: characterizing multiple sclerosis imaging markers. J Neuroimaging. 2024;34:86-94. doi:10.1111/jon.13173
Clarke MA, Lakhani DA, Wen S, et al. Perilesional neurodegenerative injury in multiple sclerosis: relation to focal lesions and impact on disability. Mult Scler Relat Disord. 2021;49:102738. doi:10.1016/j.msard.2021.102738
Laule C, Moore GRW. Myelin water imaging to detect demyelination and remyelination and its validation in pathology. Brain Pathol. 2018;28:750-764. doi:10.1111/bpa.12645
Coelho S, Baete SH, Lemberskiy G, et al. Reproducibility of the standard model of diffusion in white matter on clinical MRI systems. Neuroimage. 2022;257:119290. doi:10.1016/j.neuroimage.2022.119290
Novikov DS, Veraart J, Jelescu IO, et al. Rotationally-invariant mapping of scalar and orientational metrics of neuronal microstructure with diffusion MRI. Neuroimage. 2018;174:518-538. doi:10.1016/j.neuroimage.2018.03.006
Langkammer C, Liu T, Khalil M, et al. Quantitative susceptibility mapping in multiple sclerosis. Radiology. 2013;267:551-559. doi:10.1148/radiol.12120707
Collorone S, Coll L, Lorenzi M, et al. Artificial intelligence applied to MRI data to tackle key challenges in multiple sclerosis. Mult Scler. 2024;30:767-784. doi:10.1177/13524585241249422
Multiple sclerosis (MS) is a complex, chronic immune-mediated disease of the central nervous system characterized by focal inflammation, demyelination, and neurodegeneration. Magnetic resonance imaging (MRI), first incorporated into the McDonald Criteria for the diagnosis of MS in 2001, is an integral tool in the diagnosis, prognosis, and therapeutic monitoring of people with MS (PwMS).1
MRI research in MS is rapidly expanding and offers insights into the pathophysiology of MS with important implications for the routine clinical care of PwMS. At the Consortium of Multiple Sclerosis Centers 2024 Annual Meeting, the US Department of Veterans Affairs (VA) MS Centers of Excellence hosted an educational symposium highlighting MRI biomarkers in MS, including T2-lesions, chronic black holes (cBHs), brain atrophy, paramagnetic rim lesions (PRLs), and the central vein sign (CVS). The symposium also provided a brief overview of quantitative MRI techniques used to characterize MS lesion severity and research applications of these techniques. This clinical review summarizes the main points of that symposium with the goal of introducing key concepts to federal health care practitioners caring for PwMS.
MRI Biomarkers in MS
T2-lesions, Chronic Black Holes, and Brain Atrophy
Focal immune-mediated inflammation and demyelination in MS may be detected by MRI as hyperintense foci on T2-weighted (T2-w) imaging (eg, T2-w turbo spin echo or T2-w fluid attenuated inversion recovery sequences). These T2-lesions, critical for diagnosing MS, are typically ovoid and occur in the periventricular, juxtacortical, infratentorial spinal cord white matter (Figure 1A). T2-lesion number and volume show some association with disability and optic nerve.
Wattjes et al highlight 2 cases to demonstrate this point: a man aged 52 years with MS for 23 years and a woman aged 50 years with MS for 11 years. Despite having MS for a much shorter duration, the woman had worse disability due to a higher lesion number and volume.2 T2-lesion volume also impacts disability progression in PwMS. Gauthier et al compared the probability of progression in 3 women, all of whom were aged 39 years and had MS for 6 years. The profile with highest probability of disability progression had the highest quartile of T2-lesion volume.3 T2-lesion volume over 2 years correlates with worse scores on disability metrics such as the MS functional composite, paced auditory serial addition task, and brain volume.4 A 2024 systematic review and meta-analysis demonstrated that T2-lesion volume is significantly correlated with clinical disability in PwMS.5
Select T2-lesions are also hypointense on T1-w spin echo images and are known as cBHs (Figure 1B). Histologically, T2-lesions with cBHs have more severe architectural disruption than those without cBHs.6 cBH number and volume are significantly correlated with disability, regardless of the degree of hypointensity on T1-w imaging.5,7 A 10-year longitudinal study demonstrated that cBHs were associated with disease progression after 5 years while T2-lesion volume was not, indicating that cBHs may be a more accurate predictor of disability.8
Brain atrophy, another imaging biomarker of MS, affects both the cerebral white and gray matter. White matter fraction (the volume of white matter relative to the intracranial compartment volume) and gray matter fraction (the volume of gray matter relative to the intracranial compartment) are significantly lower among PwMS compared with healthy controls. In addition, gray matter fraction is lower among patients with primary and secondary progressive MS compared with those with relapsing-remitting MS, clinically isolated syndrome (CIS), and radiologically isolated syndrome (RIS). Gray matter fraction is also correlated with several motor and cognitive disability indices.9
Paramagnetic Rim Lesions
Neurologic worsening in PwMS occurs by 2 distinct mechanisms: relapse-associated worsening, a stepwise worsening of symptoms due to incomplete recovery following a relapse; and progression independent of relapse activity (PIRA), which is an irreversible neurologic deterioration in the absence of clinical or radiological relapses.10 PIRA is associated with neurodegeneration and predominates in both primary and secondary progressive MS. However, recent data demonstrated that PIRA may contribute to as much as 50% of disability worsening in relapsing MS and occurs early in the RMS disease course.10,11 Current high-efficacy disease modifying therapy, such as ocrelizumab, are extraordinarily successful at preventing focal inflammation and relapses but are less effective for preventing the slow march of disability progression characterizing PIRA.12,13 The prevention of PIRA is therefore an unmet treatment need.
Chronic active lesions (CALs) are an important driver of PIRA. When an acute gadolinium-enhancing lesion develops in PwMS, there are 3 possible fates of this lesion. The lesion may become chronically inactive, remyelinate, or transition to CALs.14 The histopathologic signature of CALs is compartmentalized, low-grade inflammation behind an intact blood-brain barrier with evidence of both active and chronic components.15 CALs may be found not only in cerebral white matter but also in the cerebral cortex and spinal cord.16,17 Combined MRI and histopathological studies have shown that iron-laden microglia/macrophages can be detected by susceptibility-based MRI as a rim of paramagnetic signal surrounding select T2-lesions.19 These PRLs represent an in vivo imaging biomarker of CAL (Figure 1C). According to the North American Imaging in MS Cooperative (NAIMS) consensus criteria, a PRL must surround at least two-thirds of the outer edge of a T2-lesion, be visible in ≥ 2 consecutive MRI slices, and cannot be contrast enhancing.20
PRLs can be visualized on multiple susceptibility-based imaging methods, including multiecho derived R2*/T2*, phase maps, susceptibility-weighted imaging, and quantitative susceptibility mapping.21-23 Retrospective analyses have shown no significant differences in sensitivity across these imaging modalities.24 Although first visualized with 7T MRI, PRLs may also be detected by 1.5T and 3T MRI with comparable sensitivities.25-27 However, there remains a significant knowledge gap regarding the accuracy of each imaging modality. Systematic, prospectively designed studies are needed to ascertain the comparative value of each method.
The presence of PRL is a poor prognostic indicator. PwMS without PRLs have higher levels of disability, are more likely to progress, and demonstrate greater gray matter atrophy and cognitive dysfunction when compared with PwMS with PRLs.27-29 Lesions with PRL tend to slowly expand, exhibit greater demyelination, and have diminished white matter integrity.21,22,30
PRLs may also be used as a diagnostic tool. PRLs are highly specific for MS/CIS with a 99.7% specificity and 98.4% positive predictive value, although the sensitivity is limited to 24%.31 Taken together, these data indicate that the presence of a PRL substantially increases the likelihood of an MS/CIS diagnosis, whereas the absence of a PRL does not exclude these diagnoses.
Several unanswered questions remain: Why do select acute MS lesions transition to CALs? How may investigators utilize PRLs as outcome measures in future clinical trials? How should PRLs be incorporated into the routine care of PwMS? As the role of this imaging biomarker is clarified both in the research and clinical settings, clinicians caring for PwMS can expect to increasingly encounter the topic of PRLs in the near future.
Central Vein Sign
A CVS is defined by the presence of a central vessel within a demyelinating plaque (Figure 1D). As early as the 1820s, MS plaques on gross pathology were noted to follow the course of a vessel. Early histological studies reported that up to 91% of MS plaques had a central vessel present.32 Lesion formation is dependent on the movement of lymphocytes and other inflammatory cells from the systemic circulation across the blood brain barrier into the perivascular space, a privileged site where immune cells interact with antigen presenting cells to launch an inflammatory cascade and eventual demyelinating lesion.33
CVS can be visualized on 1.5T, 3T and 7T MRI. However, 7T MRI is superior to 3T in the detection of CVS, with 85% of MS lesions having CVS visible compared with 45% on 3T.34 With advances in 7T MRI, fluid attenuated inversion recovery and T2* susceptibility, weighted sequences can be overlaid, allowing simultaneous visualization of the vessel and the demyelinating lesion. With higher density of parenchymal veins in the periventricular regions, the CVS is most seen in lesions of this territory but can also be present in juxtacortical, thalamic and infratentorial lesions with decreasing prevalence as these approach the cortex.35
MS lesions are more likely to have CVS than T2 hyperintense white matter lesions of other causes, with a large study reporting 78% of MS lesions were CVS positive. Further, CVS positive lesions can be found across all MS phenotypes including relapsing remitting, primary progressive, and secondary progressive.35 The CVS is also specific to MS lesions and is an effective tool for differentiating MS lesions from other common causes of T2 hyperintense lesions including chronic ischemic white matter disease,36 migraines,37 neuromyelitis optica spectrum disorders,38,39 Susac syndrome,40 and systemic autoimmune diseases (Behcet disease, systemic lupus erythematosus, and antiphospholipid syndrome).41
With CVS emerging as a promising radiographic biomarker for MS, NAIMS issued a consensus statement on necessary properties of a CVS. These criteria included appearance of a thin hypointense line or small dot, visualized in ≥ 2 perpendicular planes, with diameter < 2 mm, and running partially or entirely through the center of the lesion. They also clarified that lesions < 3 mm, confluent lesions, lesions with multiple vessels present or poorly visualized lesions were excluded.42
A shared CVS definition was a necessary step toward routine use of CVS as a radiographic biomarker and its incorporation in the 2024 revised McDonald criteria.43 Remaining limitations including 7T MRI is primarily available in research settings and the lack of consensus on a diagnostic threshold. There have been many proposed methods, including a 40% cut off,44 60% cut off,45 and Select 3* or Select 6* methods.46 The goal of each method is to optimize sensitivity and specificity while not compromising efficiency of MRI review for both neurologists and radiologists.
The CVS has significant potential as a radiographic biomarker for MS and may allow the early stages of MS to be differentiated from other common causes of white matter lesions on MRI. However, it remains unclear whether CVS holds prognostic value for patients, if CVS is suggestive of differing underlying pathology, or if the presence of a CVS is dynamic over time. Progress in these areas is anticipated as CVS is incorporated into routine clinical practice.
Quantitative MRI Techniques
In the research setting, several imaging modalities can be used to quantify the degree of microstructural injury in PwMS. The goal of these methods is to identify and quantify myelin and axonal damage, the major drivers of neurodegeneration. Among these methods, diffusion-based imaging is a measure of the amount of diffusion or fluid mobility across the tissues of the brain.47 Diffusion-weighted imaging (DWI) yields several parametric maps including axial diffusivity (AD), radial diffusivity (RD), and mean diffusivity (Figure 2 A, B, and C). These parametric maps provide information on different directions of water molecules’ movements. Myelin surrounds the axons preventing water molecules diffusion perpendicular to axons (RD) while axonal content prevents water diffusion horizontal to the axons (AD).Thus, AD is considered more specific to axonal injury, whereas RD is specific to myelin content.48 A higher value of any of these metrics is associated with a higher degree of tissue injury.
Although sensitive to axonal and myelin injury, AD and RD computed from single b-shell DWI experience several limitations including being affected by nonpathologic factors such as fiber orientation, distribution, and crossing, and by various nonmyelin specific pathologies including fluid accumulation during inflammation, myelin sheath thickness, and axonal intactness.48 Several multi b-shell methods have been developed to overcome diffusion imaging limitations. For example, work at the Nashville VA MS Center of Excellence has focused on the use of the multicompartment diffusion MRI with spherical mean technique (SMT). This method removes the orientation dependency of the diffusion MRI signal, increasing the signal-to-noise ratio and reducing biases from fiber undulation, crossing, and dispersion.49 SMT generates the apparent axonal volume fraction (Vax), which is a direct measure of axonal integrity with lower values indicating lower axonal content and higher tissue destruction (Figure 2D). Vax was previously validated in MS as a measure of axonal integrity.49
In terms of myelin, several other specific measures have been developed. Magnetization transfer ratio (MTR) is another measure of tissue integrity that has been validated as a measure of tissue injury in MS (Figure 2E).50,51 Zheng et al found that the percentage of lesions with low MTR was significantly higher among patients whose disease disability progressed compared with patients who did not.52Selective inversion recovery with quantitative magnetization transfer (SIR-qMT) was developed to account for the limitations of MTR, including its sensitivity to edema and axonal density.52 Germane to myelin measurements, SIR-qMT generates the macromolecular to free size ratio (PSR). PSR represents the ratio of protons bound to macromolecules (myelin) to free protons (Figure 2F). PSR is considered a marker of myelin integrity, with lower values correlating with disability severity and indicating higher tissue damage and lower myelin content. Previous studies from the Nashville VA MS Center of Excellence validated the use of SIR-qMT among patients with MS, CIS, RIS, and healthy controls.53
Quantitative MRI has several research applications in the field of MS. We demonstrated that PRL harbor a higher degree of myelin injury indicated by PSR compared with rimless lesions.54 These MRI techniques are also helpful to investigate tissues surrounding the lesions, called normal appearing white matter (NAWM). Using quantitative MRI techniques such as MTR,52 PSR,53 and Vax,49 investigators have demonstrated that NAWM is injured in PwMS, and proximal NAWM may have higher degree of tissue damage compared with distant NAWM.55
Anticipated Innovations and Challenges
In the field of quantitative MRI, several new techniques are being adopted. Researchers are developing techniques such as myelin water fraction which evaluates the interaction between water and protons to measure myelin content. This is considered an advancement as it takes into account edema resulting from MS injury.56 Another example is multicompartment diffusion imaging, such as standard model imaging,57 and neurite orientation dispersion and density imaging,58 which considers water as an additional compartment compared with the SMT derived Vax. For PRL identification, more advanced methodologic techniques are developing such quantitative susceptibility mapping (QSM), which can detect iron deposits that surround the lesions with relatively high sensitivity and specificity of identifying PRL.59
Despite these innovations, several challenges remain before possible incorporation into the clinical setting. These limitations include longer scan time, familiarity of clinicians in using these maps, higher financial cost, and the necessity of advanced imaging processing skills. Artificial intelligence is a promising tool that may overcome these challenges through creating automated processing pipelines and developing synthetic maps without the need for additional acquisition.60
Conclusions
MRI is the most important tool for diagnosing and treating PwMS. Imaging biomarkers such as T2-lesions, cBHs, brain atrophy, PRLs, and CVS provide insight into the disease’s pathogenesis and are invaluable for the accurate diagnosis and prognostication of MS. Quantitative MRI techniques, while not available in the clinical setting, are important tools for translational research that may help direct the development of future therapeutics. In the near future, clinicians caring for PwMS should expect to encounter these imaging biomarkers more frequently in the clinical setting, especially with the inclusion of PRLs and CVS in the next iteration of the McDonald diagnostic criteria.
Multiple sclerosis (MS) is a complex, chronic immune-mediated disease of the central nervous system characterized by focal inflammation, demyelination, and neurodegeneration. Magnetic resonance imaging (MRI), first incorporated into the McDonald Criteria for the diagnosis of MS in 2001, is an integral tool in the diagnosis, prognosis, and therapeutic monitoring of people with MS (PwMS).1
MRI research in MS is rapidly expanding and offers insights into the pathophysiology of MS with important implications for the routine clinical care of PwMS. At the Consortium of Multiple Sclerosis Centers 2024 Annual Meeting, the US Department of Veterans Affairs (VA) MS Centers of Excellence hosted an educational symposium highlighting MRI biomarkers in MS, including T2-lesions, chronic black holes (cBHs), brain atrophy, paramagnetic rim lesions (PRLs), and the central vein sign (CVS). The symposium also provided a brief overview of quantitative MRI techniques used to characterize MS lesion severity and research applications of these techniques. This clinical review summarizes the main points of that symposium with the goal of introducing key concepts to federal health care practitioners caring for PwMS.
MRI Biomarkers in MS
T2-lesions, Chronic Black Holes, and Brain Atrophy
Focal immune-mediated inflammation and demyelination in MS may be detected by MRI as hyperintense foci on T2-weighted (T2-w) imaging (eg, T2-w turbo spin echo or T2-w fluid attenuated inversion recovery sequences). These T2-lesions, critical for diagnosing MS, are typically ovoid and occur in the periventricular, juxtacortical, infratentorial spinal cord white matter (Figure 1A). T2-lesion number and volume show some association with disability and optic nerve.
Wattjes et al highlight 2 cases to demonstrate this point: a man aged 52 years with MS for 23 years and a woman aged 50 years with MS for 11 years. Despite having MS for a much shorter duration, the woman had worse disability due to a higher lesion number and volume.2 T2-lesion volume also impacts disability progression in PwMS. Gauthier et al compared the probability of progression in 3 women, all of whom were aged 39 years and had MS for 6 years. The profile with highest probability of disability progression had the highest quartile of T2-lesion volume.3 T2-lesion volume over 2 years correlates with worse scores on disability metrics such as the MS functional composite, paced auditory serial addition task, and brain volume.4 A 2024 systematic review and meta-analysis demonstrated that T2-lesion volume is significantly correlated with clinical disability in PwMS.5
Select T2-lesions are also hypointense on T1-w spin echo images and are known as cBHs (Figure 1B). Histologically, T2-lesions with cBHs have more severe architectural disruption than those without cBHs.6 cBH number and volume are significantly correlated with disability, regardless of the degree of hypointensity on T1-w imaging.5,7 A 10-year longitudinal study demonstrated that cBHs were associated with disease progression after 5 years while T2-lesion volume was not, indicating that cBHs may be a more accurate predictor of disability.8
Brain atrophy, another imaging biomarker of MS, affects both the cerebral white and gray matter. White matter fraction (the volume of white matter relative to the intracranial compartment volume) and gray matter fraction (the volume of gray matter relative to the intracranial compartment) are significantly lower among PwMS compared with healthy controls. In addition, gray matter fraction is lower among patients with primary and secondary progressive MS compared with those with relapsing-remitting MS, clinically isolated syndrome (CIS), and radiologically isolated syndrome (RIS). Gray matter fraction is also correlated with several motor and cognitive disability indices.9
Paramagnetic Rim Lesions
Neurologic worsening in PwMS occurs by 2 distinct mechanisms: relapse-associated worsening, a stepwise worsening of symptoms due to incomplete recovery following a relapse; and progression independent of relapse activity (PIRA), which is an irreversible neurologic deterioration in the absence of clinical or radiological relapses.10 PIRA is associated with neurodegeneration and predominates in both primary and secondary progressive MS. However, recent data demonstrated that PIRA may contribute to as much as 50% of disability worsening in relapsing MS and occurs early in the RMS disease course.10,11 Current high-efficacy disease modifying therapy, such as ocrelizumab, are extraordinarily successful at preventing focal inflammation and relapses but are less effective for preventing the slow march of disability progression characterizing PIRA.12,13 The prevention of PIRA is therefore an unmet treatment need.
Chronic active lesions (CALs) are an important driver of PIRA. When an acute gadolinium-enhancing lesion develops in PwMS, there are 3 possible fates of this lesion. The lesion may become chronically inactive, remyelinate, or transition to CALs.14 The histopathologic signature of CALs is compartmentalized, low-grade inflammation behind an intact blood-brain barrier with evidence of both active and chronic components.15 CALs may be found not only in cerebral white matter but also in the cerebral cortex and spinal cord.16,17 Combined MRI and histopathological studies have shown that iron-laden microglia/macrophages can be detected by susceptibility-based MRI as a rim of paramagnetic signal surrounding select T2-lesions.19 These PRLs represent an in vivo imaging biomarker of CAL (Figure 1C). According to the North American Imaging in MS Cooperative (NAIMS) consensus criteria, a PRL must surround at least two-thirds of the outer edge of a T2-lesion, be visible in ≥ 2 consecutive MRI slices, and cannot be contrast enhancing.20
PRLs can be visualized on multiple susceptibility-based imaging methods, including multiecho derived R2*/T2*, phase maps, susceptibility-weighted imaging, and quantitative susceptibility mapping.21-23 Retrospective analyses have shown no significant differences in sensitivity across these imaging modalities.24 Although first visualized with 7T MRI, PRLs may also be detected by 1.5T and 3T MRI with comparable sensitivities.25-27 However, there remains a significant knowledge gap regarding the accuracy of each imaging modality. Systematic, prospectively designed studies are needed to ascertain the comparative value of each method.
The presence of PRL is a poor prognostic indicator. PwMS without PRLs have higher levels of disability, are more likely to progress, and demonstrate greater gray matter atrophy and cognitive dysfunction when compared with PwMS with PRLs.27-29 Lesions with PRL tend to slowly expand, exhibit greater demyelination, and have diminished white matter integrity.21,22,30
PRLs may also be used as a diagnostic tool. PRLs are highly specific for MS/CIS with a 99.7% specificity and 98.4% positive predictive value, although the sensitivity is limited to 24%.31 Taken together, these data indicate that the presence of a PRL substantially increases the likelihood of an MS/CIS diagnosis, whereas the absence of a PRL does not exclude these diagnoses.
Several unanswered questions remain: Why do select acute MS lesions transition to CALs? How may investigators utilize PRLs as outcome measures in future clinical trials? How should PRLs be incorporated into the routine care of PwMS? As the role of this imaging biomarker is clarified both in the research and clinical settings, clinicians caring for PwMS can expect to increasingly encounter the topic of PRLs in the near future.
Central Vein Sign
A CVS is defined by the presence of a central vessel within a demyelinating plaque (Figure 1D). As early as the 1820s, MS plaques on gross pathology were noted to follow the course of a vessel. Early histological studies reported that up to 91% of MS plaques had a central vessel present.32 Lesion formation is dependent on the movement of lymphocytes and other inflammatory cells from the systemic circulation across the blood brain barrier into the perivascular space, a privileged site where immune cells interact with antigen presenting cells to launch an inflammatory cascade and eventual demyelinating lesion.33
CVS can be visualized on 1.5T, 3T and 7T MRI. However, 7T MRI is superior to 3T in the detection of CVS, with 85% of MS lesions having CVS visible compared with 45% on 3T.34 With advances in 7T MRI, fluid attenuated inversion recovery and T2* susceptibility, weighted sequences can be overlaid, allowing simultaneous visualization of the vessel and the demyelinating lesion. With higher density of parenchymal veins in the periventricular regions, the CVS is most seen in lesions of this territory but can also be present in juxtacortical, thalamic and infratentorial lesions with decreasing prevalence as these approach the cortex.35
MS lesions are more likely to have CVS than T2 hyperintense white matter lesions of other causes, with a large study reporting 78% of MS lesions were CVS positive. Further, CVS positive lesions can be found across all MS phenotypes including relapsing remitting, primary progressive, and secondary progressive.35 The CVS is also specific to MS lesions and is an effective tool for differentiating MS lesions from other common causes of T2 hyperintense lesions including chronic ischemic white matter disease,36 migraines,37 neuromyelitis optica spectrum disorders,38,39 Susac syndrome,40 and systemic autoimmune diseases (Behcet disease, systemic lupus erythematosus, and antiphospholipid syndrome).41
With CVS emerging as a promising radiographic biomarker for MS, NAIMS issued a consensus statement on necessary properties of a CVS. These criteria included appearance of a thin hypointense line or small dot, visualized in ≥ 2 perpendicular planes, with diameter < 2 mm, and running partially or entirely through the center of the lesion. They also clarified that lesions < 3 mm, confluent lesions, lesions with multiple vessels present or poorly visualized lesions were excluded.42
A shared CVS definition was a necessary step toward routine use of CVS as a radiographic biomarker and its incorporation in the 2024 revised McDonald criteria.43 Remaining limitations including 7T MRI is primarily available in research settings and the lack of consensus on a diagnostic threshold. There have been many proposed methods, including a 40% cut off,44 60% cut off,45 and Select 3* or Select 6* methods.46 The goal of each method is to optimize sensitivity and specificity while not compromising efficiency of MRI review for both neurologists and radiologists.
The CVS has significant potential as a radiographic biomarker for MS and may allow the early stages of MS to be differentiated from other common causes of white matter lesions on MRI. However, it remains unclear whether CVS holds prognostic value for patients, if CVS is suggestive of differing underlying pathology, or if the presence of a CVS is dynamic over time. Progress in these areas is anticipated as CVS is incorporated into routine clinical practice.
Quantitative MRI Techniques
In the research setting, several imaging modalities can be used to quantify the degree of microstructural injury in PwMS. The goal of these methods is to identify and quantify myelin and axonal damage, the major drivers of neurodegeneration. Among these methods, diffusion-based imaging is a measure of the amount of diffusion or fluid mobility across the tissues of the brain.47 Diffusion-weighted imaging (DWI) yields several parametric maps including axial diffusivity (AD), radial diffusivity (RD), and mean diffusivity (Figure 2 A, B, and C). These parametric maps provide information on different directions of water molecules’ movements. Myelin surrounds the axons preventing water molecules diffusion perpendicular to axons (RD) while axonal content prevents water diffusion horizontal to the axons (AD).Thus, AD is considered more specific to axonal injury, whereas RD is specific to myelin content.48 A higher value of any of these metrics is associated with a higher degree of tissue injury.
Although sensitive to axonal and myelin injury, AD and RD computed from single b-shell DWI experience several limitations including being affected by nonpathologic factors such as fiber orientation, distribution, and crossing, and by various nonmyelin specific pathologies including fluid accumulation during inflammation, myelin sheath thickness, and axonal intactness.48 Several multi b-shell methods have been developed to overcome diffusion imaging limitations. For example, work at the Nashville VA MS Center of Excellence has focused on the use of the multicompartment diffusion MRI with spherical mean technique (SMT). This method removes the orientation dependency of the diffusion MRI signal, increasing the signal-to-noise ratio and reducing biases from fiber undulation, crossing, and dispersion.49 SMT generates the apparent axonal volume fraction (Vax), which is a direct measure of axonal integrity with lower values indicating lower axonal content and higher tissue destruction (Figure 2D). Vax was previously validated in MS as a measure of axonal integrity.49
In terms of myelin, several other specific measures have been developed. Magnetization transfer ratio (MTR) is another measure of tissue integrity that has been validated as a measure of tissue injury in MS (Figure 2E).50,51 Zheng et al found that the percentage of lesions with low MTR was significantly higher among patients whose disease disability progressed compared with patients who did not.52Selective inversion recovery with quantitative magnetization transfer (SIR-qMT) was developed to account for the limitations of MTR, including its sensitivity to edema and axonal density.52 Germane to myelin measurements, SIR-qMT generates the macromolecular to free size ratio (PSR). PSR represents the ratio of protons bound to macromolecules (myelin) to free protons (Figure 2F). PSR is considered a marker of myelin integrity, with lower values correlating with disability severity and indicating higher tissue damage and lower myelin content. Previous studies from the Nashville VA MS Center of Excellence validated the use of SIR-qMT among patients with MS, CIS, RIS, and healthy controls.53
Quantitative MRI has several research applications in the field of MS. We demonstrated that PRL harbor a higher degree of myelin injury indicated by PSR compared with rimless lesions.54 These MRI techniques are also helpful to investigate tissues surrounding the lesions, called normal appearing white matter (NAWM). Using quantitative MRI techniques such as MTR,52 PSR,53 and Vax,49 investigators have demonstrated that NAWM is injured in PwMS, and proximal NAWM may have higher degree of tissue damage compared with distant NAWM.55
Anticipated Innovations and Challenges
In the field of quantitative MRI, several new techniques are being adopted. Researchers are developing techniques such as myelin water fraction which evaluates the interaction between water and protons to measure myelin content. This is considered an advancement as it takes into account edema resulting from MS injury.56 Another example is multicompartment diffusion imaging, such as standard model imaging,57 and neurite orientation dispersion and density imaging,58 which considers water as an additional compartment compared with the SMT derived Vax. For PRL identification, more advanced methodologic techniques are developing such quantitative susceptibility mapping (QSM), which can detect iron deposits that surround the lesions with relatively high sensitivity and specificity of identifying PRL.59
Despite these innovations, several challenges remain before possible incorporation into the clinical setting. These limitations include longer scan time, familiarity of clinicians in using these maps, higher financial cost, and the necessity of advanced imaging processing skills. Artificial intelligence is a promising tool that may overcome these challenges through creating automated processing pipelines and developing synthetic maps without the need for additional acquisition.60
Conclusions
MRI is the most important tool for diagnosing and treating PwMS. Imaging biomarkers such as T2-lesions, cBHs, brain atrophy, PRLs, and CVS provide insight into the disease’s pathogenesis and are invaluable for the accurate diagnosis and prognostication of MS. Quantitative MRI techniques, while not available in the clinical setting, are important tools for translational research that may help direct the development of future therapeutics. In the near future, clinicians caring for PwMS should expect to encounter these imaging biomarkers more frequently in the clinical setting, especially with the inclusion of PRLs and CVS in the next iteration of the McDonald diagnostic criteria.
McDonald WI, Compston A, Edan G, et al. Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol. 2001;50:121-127. doi:10.1002/ana.1032
Wattjes MP, Steenwijk MD, Stangel M. MRI in the diagnosis and monitoring of multiple sclerosis: an update. Clin Neuroradiol. 2015;25:157-165. doi:10.1007/s00062-015-0430-y
Gauthier SA, Mandel M, Guttmann CR, et al. Predicting short-term disability in multiple sclerosis. Neurology. 2007;68:2059-2065.doi:10.1212/01.wnl.0000264890.97479.b1
Rudick RA, Lee JC, Simon J, Fisher E. Significance of T2 lesions in multiple sclerosis: a 13-year longitudinal study. Ann Neurol. 2006;60:236-242. doi:10.1002/ana.20883
Nabizadeh F, Zafari R, Mohamadi M, et al. MRI features and disability in multiple sclerosis: a systematic review and meta-analysis. J Neuroradiol. 2024;51:24-37. doi:10.1016/j.neurad.2023.11.007
Bagnato F, Jeffries N, Richert ND, et al. Evolution of T1 black holes in patients with multiple sclerosis imaged monthly for 4 years. Brain. 2003;126:1782-1789. doi:10.1093/brain/awg182
Jacobsen C, Hagemeier J, Myhr KM, et al. Brain atrophy and disability progression in multiple sclerosis patients: a 10-year follow-up study. J Neurol Neurosurg Psychiatry. 2014;85:1109-1115. doi:10.1136/jnnp-2013-306906
Rovaris M, Gass A, Bammer R, et al. Diffusion MRI in multiple sclerosis. Neurology. 2005;65:1526-1532. doi:10.1212/01.wnl.0000184471.83948.e0
Fisniku LK, Chard DT, Jackson JS, et al. Gray matter atrophy is related to long-term disability in multiple sclerosis. Ann Neurol. 2008;64:247-254. doi:10.1002/ana.21423
Lublin FD, Häring DA, Ganjgahi H, et al. How patients with multiple sclerosis acquire disability. Brain. 2022;145:3147-3161. doi:10.1093/brain/awac016
Kappos L, Wolinsky JS, Giovannoni G, et al. Contribution of relapse-independent progression vs relapse-associated worsening to overall confirmed disability accumulation in typical relapsing multiple sclerosis in a pooled analysis of 2 randomized clinical trials. JAMA Neurol. 2020;77:1132-1140. doi:10.1001/jamaneurol.2020.1568
Hauser SL, Bar-Or A, Comi G, et al. Ocrelizumab versus interferon beta-1a in relapsing multiple sclerosis. N Engl J Med. 2017;376:221-234. doi:10.1056/NEJMoa1601277
Montalban X, Hauser SL, Kappos L, et al. Ocrelizumab versus placebo in primary progressive multiple sclerosis. N Engl J Med. 2017;376:209-220. doi:10.1056/NEJMoa1606468
Prineas JW, Kwon EE, Cho ES, et al. Immunopathology of secondary-progressive multiple sclerosis. Ann Neurol. 2001;50:646-657. doi:10.1002/ana.1255
Kuhlmann T, Ludwin S, Prat A, Antel J, Brück W, Lassmann H. An updated histological classification system for multiple sclerosis lesions. Acta Neuropathol. 2017;133:13-24. doi:10.1007/s00401-016-1653-y
Pitt D, Boster A, Pei W, et al. Imaging cortical lesions in multiple sclerosis with ultra-high-field magnetic resonance imaging. Arch Neurol. 2010;67:812-818. doi:10.1001/archneurol.2010.148
Gilmore CP, Geurts JJ, Evangelou N, et al. Spinal cord grey matter lesions in multiple sclerosis detected by post-mortem high field MR imaging. Mult Scler. 2009;15:180-188. doi:10.1177/1352458508096876
Lassmann H, Brück W, Lucchinetti CF. The immunopathology of multiple sclerosis: an overview. Brain Pathol. 2007;17:210-218. doi:10.1111/j.1750-3639.2007.00064.x
Bagnato F, Hametner S, Yao B, et al. Tracking iron in multiple sclerosis: a combined imaging and histopathological study at 7 Tesla. Brain. 2011;134:3602-3615. doi:10.1093/brain/awr278
Bagnato F, Sati P, Hemond CC, et al. Imaging chronic active lesions in multiple sclerosis: a consensus statement. Brain. 2024;147:2913-2933. doi:10.1093/brain/awae013
Dal-Bianco A, Grabner G, Kronnerwetter C, et al. Slow expansion of multiple sclerosis iron rim lesions: pathology and 7 T magnetic resonance imaging. Acta Neuropathol. 2017;133:25-42. doi:10.1007/s00401-016-1636-z
Absinta M, Sati P, Schindler M, et al. Persistent 7-tesla phase rim predicts poor outcome in new multiple sclerosis patient lesions. J Clin Invest. 2016;126:2597-2609. doi:10.1172/JCI86198
Gillen KM, Mubarak M, Park C, et al. QSM is an imaging biomarker for chronic glial activation in multiple sclerosis lesions. Ann Clin Transl Neurol. 2021;8:877-886. doi:10.1002/acn3.51338
Ng Kee Kwong KC, Mollison D, Meijboom R, et al. The prevalence of paramagnetic rim lesions in multiple sclerosis: a systematic review and meta-analysis. PLoS One. 2021;16:e0256845. doi:10.1371/journal.pone.0256845
Absinta M, Sati P, Fechner A, et al. Identification of chronic active multiple sclerosis lesions on 3T MRI. AJNR Am J Neuroradiol. 2018;39:1233-1238. doi:10.3174/ajnr.A5660
Hemond CC, Reich DS, Dundamadappa SK. Paramagnetic rim lesions in multiple sclerosis: comparison of visualization at 1.5-T and 3-T MRI. AJR Am J Roentgenol. 2022;219:120-131. doi:10.2214/AJR.21.26777
Altokhis AI, Hibbert AM, Allen CM, et al. Longitudinal clinical study of patients with iron rim lesions in multiple sclerosis. Mult Scler. 2022;28:2202-2211. doi:10.1177/13524585221114750
Choi S, Lake S, Harrison DM. Evaluation of the blood-brain barrier, demyelination, and neurodegeneration in paramagnetic rim lesions in multiple sclerosis on 7 tesla MRI. J Magn Reson Imaging. 2024;59:941-951. doi:10.1002/jmri.28847
Kazimuddin HF, Wang J, Hernandez B, et al. Paramagnetic rim lesions and their relationship with neurodegeneration and clinical disability at the time of multiple sclerosis diagnosis. Poster presented at: 2024 Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS) Forum; February 26-March 2; West Palm Beach, FL.
Rohm Z, Koch C, Kazimuddin H, et al. Longitudinal characterization of paramagnetic rim lesions in early multiple sclerosis. Poster presented at: 2024 Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS) Forum; February 26-March 2; West Palm Beach, FL.
Meaton I, Altokhis A, Allen CM, et al. Paramagnetic rims are a promising diagnostic imaging biomarker in multiple sclerosis. Mult Scler. 2022;28:2212-2220. doi:10.1177/13524585221118677
Fog T. On the vessel-plaque relationships in the brain in multiple sclerosis. Acta Neurol Scand Suppl. 1964;40:9-15.
Ineichen BV, Okar SV, Proulx ST, et al. Perivascular spaces and their role in neuroinflammation. Neuron. 2022;110:3566-3581. doi:10.1016/j.neuron.2022.10.024
Tallantyre EC, Morgan PS, Dixon JE, et al. A comparison of 3T and 7T in the detection of small parenchymal veins within MS lesions. Invest Radiol. 2009;44:491-494. doi:10.1097/RLI.0b013e3181b4c144
Kilsdonk ID, Lopez-Soriano A, Kuijer JP, et al. Morphological features of MS lesions on FLAIR* at 7 T and their relation to patient characteristics. J Neurol. 2014;261:1356-1364. doi:10.1007/s00415-014-7351-6
Tallantyre EC, Dixon JE, Donaldson I, et al. Ultra-high-field imaging distinguishes MS lesions from asymptomatic white matter lesions. Neurology. 2011;76:534-539. doi:10.1212/WNL.0b013e31820b7630
Solomon AJ, Schindler MK, Howard DB, et al. “Central vessel sign” on 3T FLAIR* MRI for the differentiation of multiple sclerosis from migraine. Ann Clin Transl Neurol. 2015;3:82-87. doi:10.1002/acn3.273
Sinnecker T, Dörr J, Pfueller CF, et al. Distinct lesion morphology at 7-T MRI differentiates neuromyelitis optica from multiple sclerosis. Neurology. 2012;79:708-714. doi:10.1212/WNL.0b013e3182648bc8
Kister I, Herbert J, Zhou Y, Ge Y. Ultrahigh-field MR (7 T) imaging of brain lesions in neuromyelitis optica. Mult Scler Int. 2013;2013:398259. doi:10.1155/2013/398259
Wuerfel J, Sinnecker T, Ringelstein EB, et al. Lesion morphology at 7 Tesla MRI differentiates Susac syndrome from multiple sclerosis. Mult Scler. 2012;18:1592-1599. doi:10.1177/1352458512441270
Massacesi L. Perivenular distribution of white matter lesions evaluated by MRI can differentiate MS lesions from inflammatory small vessel diseases. Eur J Neurol. 2016;23:86. doi:10.1212/WNL.86.16_supplement.P6.121
Sati P, Oh J, Constable RT, et al. The central vein sign and its clinical evaluation for the diagnosis of multiple sclerosis: a consensus statement from the North American Imaging in Multiple Sclerosis Cooperative. Nat Rev Neurol. 2016;12:714-722. doi:10.1038/nrneurol.2016.166
Montalban X, Lebrun-Frénay C, Oh J, et al. Diagnosis of multiple sclerosis: 2024 revisions of the McDonald criteria. Lancet Neurol. 2025;24:850-865. doi:10.1016/S1474-4422(25)00270-4
Mistry N, Dixon J, Tallantyre E, et al. Central veins in brain lesions visualized with high-field magnetic resonance imaging: a pathologically specific diagnostic biomarker for inflammatory demyelination in the brain. JAMA Neurol. 2013;70:623-628. doi:10.1001/jamaneurol.2013.1405
Campion T, Smith RJP, Altmann DR, et al. FLAIR* to visualize veins in white matter lesions: a new tool for the diagnosis of multiple sclerosis? Eur Radiol. 2017;27:4257-4263. doi:10.1007/s00330-017-4822-z
Solomon AJ, Watts R, Ontaneda D, et al. Diagnostic performance of central vein sign for multiple sclerosis with a simplified three-lesion algorithm. Mult Scler. 2018;24:750-757. doi:10.1177/1352458517726383
Cercignani M, Bozzali M, Iannucci G, Comi G, Filippi M. Intra-voxel and inter-voxel coherence in patients with multiple sclerosis assessed using diffusion tensor MRI. J Neurol. 2002;249:875-883. doi:10.1007/s00415-002-0752-y
Song SK, Yoshino J, Le TQ, et al. Demyelination increases radial diffusivity in corpus callosum of mouse brain. Neuroimage. 2005;26:132-140. doi:10.1016/j.neuroimage.2005.01.028
Bagnato F, Franco G, Li H, et al. Probing axons using multi-compartmental diffusion in multiple sclerosis. Ann Clin Transl Neurol. 2019;6:1595-1605. doi:10.1002/acn3.50836
Filippi M, Cercignani M, Inglese M, et al. Diffusion tensor magnetic resonance imaging in multiple sclerosis. Neurology. 2001;56:304-311. doi:10.1212/wnl.56.3.304
Bagnato F. Clinical application of magnetization transfer imaging. In: Advanced Neuro MR Techniques and Applications. Elsevier; 2022:403-417. doi:10.1016/B978-0-12-822479-3.00041-5
Zheng Y, Lee JC, Rudick R, Fisher E. Long-term magnetization transfer ratio evolution in multiple sclerosis white matter lesions. J Neuroimaging. 2018;28:191-198. doi:10.1111/jon.12480
Bagnato F, Hametner S, Franco G, et al. Selective inversion recovery quantitative magnetization transfer brain MRI at 7T: clinical and postmortem validation in multiple sclerosis. J Neuroimaging. 2018;28:380-388. doi:10.1111/jon.12511
Clarke MA, Cheek R, Hernandez B, et al. Paramagnetic rim lesions and the central vein sign: characterizing multiple sclerosis imaging markers. J Neuroimaging. 2024;34:86-94. doi:10.1111/jon.13173
Clarke MA, Lakhani DA, Wen S, et al. Perilesional neurodegenerative injury in multiple sclerosis: relation to focal lesions and impact on disability. Mult Scler Relat Disord. 2021;49:102738. doi:10.1016/j.msard.2021.102738
Laule C, Moore GRW. Myelin water imaging to detect demyelination and remyelination and its validation in pathology. Brain Pathol. 2018;28:750-764. doi:10.1111/bpa.12645
Coelho S, Baete SH, Lemberskiy G, et al. Reproducibility of the standard model of diffusion in white matter on clinical MRI systems. Neuroimage. 2022;257:119290. doi:10.1016/j.neuroimage.2022.119290
Novikov DS, Veraart J, Jelescu IO, et al. Rotationally-invariant mapping of scalar and orientational metrics of neuronal microstructure with diffusion MRI. Neuroimage. 2018;174:518-538. doi:10.1016/j.neuroimage.2018.03.006
Langkammer C, Liu T, Khalil M, et al. Quantitative susceptibility mapping in multiple sclerosis. Radiology. 2013;267:551-559. doi:10.1148/radiol.12120707
Collorone S, Coll L, Lorenzi M, et al. Artificial intelligence applied to MRI data to tackle key challenges in multiple sclerosis. Mult Scler. 2024;30:767-784. doi:10.1177/13524585241249422
McDonald WI, Compston A, Edan G, et al. Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol. 2001;50:121-127. doi:10.1002/ana.1032
Wattjes MP, Steenwijk MD, Stangel M. MRI in the diagnosis and monitoring of multiple sclerosis: an update. Clin Neuroradiol. 2015;25:157-165. doi:10.1007/s00062-015-0430-y
Gauthier SA, Mandel M, Guttmann CR, et al. Predicting short-term disability in multiple sclerosis. Neurology. 2007;68:2059-2065.doi:10.1212/01.wnl.0000264890.97479.b1
Rudick RA, Lee JC, Simon J, Fisher E. Significance of T2 lesions in multiple sclerosis: a 13-year longitudinal study. Ann Neurol. 2006;60:236-242. doi:10.1002/ana.20883
Nabizadeh F, Zafari R, Mohamadi M, et al. MRI features and disability in multiple sclerosis: a systematic review and meta-analysis. J Neuroradiol. 2024;51:24-37. doi:10.1016/j.neurad.2023.11.007
Bagnato F, Jeffries N, Richert ND, et al. Evolution of T1 black holes in patients with multiple sclerosis imaged monthly for 4 years. Brain. 2003;126:1782-1789. doi:10.1093/brain/awg182
Jacobsen C, Hagemeier J, Myhr KM, et al. Brain atrophy and disability progression in multiple sclerosis patients: a 10-year follow-up study. J Neurol Neurosurg Psychiatry. 2014;85:1109-1115. doi:10.1136/jnnp-2013-306906
Rovaris M, Gass A, Bammer R, et al. Diffusion MRI in multiple sclerosis. Neurology. 2005;65:1526-1532. doi:10.1212/01.wnl.0000184471.83948.e0
Fisniku LK, Chard DT, Jackson JS, et al. Gray matter atrophy is related to long-term disability in multiple sclerosis. Ann Neurol. 2008;64:247-254. doi:10.1002/ana.21423
Lublin FD, Häring DA, Ganjgahi H, et al. How patients with multiple sclerosis acquire disability. Brain. 2022;145:3147-3161. doi:10.1093/brain/awac016
Kappos L, Wolinsky JS, Giovannoni G, et al. Contribution of relapse-independent progression vs relapse-associated worsening to overall confirmed disability accumulation in typical relapsing multiple sclerosis in a pooled analysis of 2 randomized clinical trials. JAMA Neurol. 2020;77:1132-1140. doi:10.1001/jamaneurol.2020.1568
Hauser SL, Bar-Or A, Comi G, et al. Ocrelizumab versus interferon beta-1a in relapsing multiple sclerosis. N Engl J Med. 2017;376:221-234. doi:10.1056/NEJMoa1601277
Montalban X, Hauser SL, Kappos L, et al. Ocrelizumab versus placebo in primary progressive multiple sclerosis. N Engl J Med. 2017;376:209-220. doi:10.1056/NEJMoa1606468
Prineas JW, Kwon EE, Cho ES, et al. Immunopathology of secondary-progressive multiple sclerosis. Ann Neurol. 2001;50:646-657. doi:10.1002/ana.1255
Kuhlmann T, Ludwin S, Prat A, Antel J, Brück W, Lassmann H. An updated histological classification system for multiple sclerosis lesions. Acta Neuropathol. 2017;133:13-24. doi:10.1007/s00401-016-1653-y
Pitt D, Boster A, Pei W, et al. Imaging cortical lesions in multiple sclerosis with ultra-high-field magnetic resonance imaging. Arch Neurol. 2010;67:812-818. doi:10.1001/archneurol.2010.148
Gilmore CP, Geurts JJ, Evangelou N, et al. Spinal cord grey matter lesions in multiple sclerosis detected by post-mortem high field MR imaging. Mult Scler. 2009;15:180-188. doi:10.1177/1352458508096876
Lassmann H, Brück W, Lucchinetti CF. The immunopathology of multiple sclerosis: an overview. Brain Pathol. 2007;17:210-218. doi:10.1111/j.1750-3639.2007.00064.x
Bagnato F, Hametner S, Yao B, et al. Tracking iron in multiple sclerosis: a combined imaging and histopathological study at 7 Tesla. Brain. 2011;134:3602-3615. doi:10.1093/brain/awr278
Bagnato F, Sati P, Hemond CC, et al. Imaging chronic active lesions in multiple sclerosis: a consensus statement. Brain. 2024;147:2913-2933. doi:10.1093/brain/awae013
Dal-Bianco A, Grabner G, Kronnerwetter C, et al. Slow expansion of multiple sclerosis iron rim lesions: pathology and 7 T magnetic resonance imaging. Acta Neuropathol. 2017;133:25-42. doi:10.1007/s00401-016-1636-z
Absinta M, Sati P, Schindler M, et al. Persistent 7-tesla phase rim predicts poor outcome in new multiple sclerosis patient lesions. J Clin Invest. 2016;126:2597-2609. doi:10.1172/JCI86198
Gillen KM, Mubarak M, Park C, et al. QSM is an imaging biomarker for chronic glial activation in multiple sclerosis lesions. Ann Clin Transl Neurol. 2021;8:877-886. doi:10.1002/acn3.51338
Ng Kee Kwong KC, Mollison D, Meijboom R, et al. The prevalence of paramagnetic rim lesions in multiple sclerosis: a systematic review and meta-analysis. PLoS One. 2021;16:e0256845. doi:10.1371/journal.pone.0256845
Absinta M, Sati P, Fechner A, et al. Identification of chronic active multiple sclerosis lesions on 3T MRI. AJNR Am J Neuroradiol. 2018;39:1233-1238. doi:10.3174/ajnr.A5660
Hemond CC, Reich DS, Dundamadappa SK. Paramagnetic rim lesions in multiple sclerosis: comparison of visualization at 1.5-T and 3-T MRI. AJR Am J Roentgenol. 2022;219:120-131. doi:10.2214/AJR.21.26777
Altokhis AI, Hibbert AM, Allen CM, et al. Longitudinal clinical study of patients with iron rim lesions in multiple sclerosis. Mult Scler. 2022;28:2202-2211. doi:10.1177/13524585221114750
Choi S, Lake S, Harrison DM. Evaluation of the blood-brain barrier, demyelination, and neurodegeneration in paramagnetic rim lesions in multiple sclerosis on 7 tesla MRI. J Magn Reson Imaging. 2024;59:941-951. doi:10.1002/jmri.28847
Kazimuddin HF, Wang J, Hernandez B, et al. Paramagnetic rim lesions and their relationship with neurodegeneration and clinical disability at the time of multiple sclerosis diagnosis. Poster presented at: 2024 Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS) Forum; February 26-March 2; West Palm Beach, FL.
Rohm Z, Koch C, Kazimuddin H, et al. Longitudinal characterization of paramagnetic rim lesions in early multiple sclerosis. Poster presented at: 2024 Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS) Forum; February 26-March 2; West Palm Beach, FL.
Meaton I, Altokhis A, Allen CM, et al. Paramagnetic rims are a promising diagnostic imaging biomarker in multiple sclerosis. Mult Scler. 2022;28:2212-2220. doi:10.1177/13524585221118677
Fog T. On the vessel-plaque relationships in the brain in multiple sclerosis. Acta Neurol Scand Suppl. 1964;40:9-15.
Ineichen BV, Okar SV, Proulx ST, et al. Perivascular spaces and their role in neuroinflammation. Neuron. 2022;110:3566-3581. doi:10.1016/j.neuron.2022.10.024
Tallantyre EC, Morgan PS, Dixon JE, et al. A comparison of 3T and 7T in the detection of small parenchymal veins within MS lesions. Invest Radiol. 2009;44:491-494. doi:10.1097/RLI.0b013e3181b4c144
Kilsdonk ID, Lopez-Soriano A, Kuijer JP, et al. Morphological features of MS lesions on FLAIR* at 7 T and their relation to patient characteristics. J Neurol. 2014;261:1356-1364. doi:10.1007/s00415-014-7351-6
Tallantyre EC, Dixon JE, Donaldson I, et al. Ultra-high-field imaging distinguishes MS lesions from asymptomatic white matter lesions. Neurology. 2011;76:534-539. doi:10.1212/WNL.0b013e31820b7630
Solomon AJ, Schindler MK, Howard DB, et al. “Central vessel sign” on 3T FLAIR* MRI for the differentiation of multiple sclerosis from migraine. Ann Clin Transl Neurol. 2015;3:82-87. doi:10.1002/acn3.273
Sinnecker T, Dörr J, Pfueller CF, et al. Distinct lesion morphology at 7-T MRI differentiates neuromyelitis optica from multiple sclerosis. Neurology. 2012;79:708-714. doi:10.1212/WNL.0b013e3182648bc8
Kister I, Herbert J, Zhou Y, Ge Y. Ultrahigh-field MR (7 T) imaging of brain lesions in neuromyelitis optica. Mult Scler Int. 2013;2013:398259. doi:10.1155/2013/398259
Wuerfel J, Sinnecker T, Ringelstein EB, et al. Lesion morphology at 7 Tesla MRI differentiates Susac syndrome from multiple sclerosis. Mult Scler. 2012;18:1592-1599. doi:10.1177/1352458512441270
Massacesi L. Perivenular distribution of white matter lesions evaluated by MRI can differentiate MS lesions from inflammatory small vessel diseases. Eur J Neurol. 2016;23:86. doi:10.1212/WNL.86.16_supplement.P6.121
Sati P, Oh J, Constable RT, et al. The central vein sign and its clinical evaluation for the diagnosis of multiple sclerosis: a consensus statement from the North American Imaging in Multiple Sclerosis Cooperative. Nat Rev Neurol. 2016;12:714-722. doi:10.1038/nrneurol.2016.166
Montalban X, Lebrun-Frénay C, Oh J, et al. Diagnosis of multiple sclerosis: 2024 revisions of the McDonald criteria. Lancet Neurol. 2025;24:850-865. doi:10.1016/S1474-4422(25)00270-4
Mistry N, Dixon J, Tallantyre E, et al. Central veins in brain lesions visualized with high-field magnetic resonance imaging: a pathologically specific diagnostic biomarker for inflammatory demyelination in the brain. JAMA Neurol. 2013;70:623-628. doi:10.1001/jamaneurol.2013.1405
Campion T, Smith RJP, Altmann DR, et al. FLAIR* to visualize veins in white matter lesions: a new tool for the diagnosis of multiple sclerosis? Eur Radiol. 2017;27:4257-4263. doi:10.1007/s00330-017-4822-z
Solomon AJ, Watts R, Ontaneda D, et al. Diagnostic performance of central vein sign for multiple sclerosis with a simplified three-lesion algorithm. Mult Scler. 2018;24:750-757. doi:10.1177/1352458517726383
Cercignani M, Bozzali M, Iannucci G, Comi G, Filippi M. Intra-voxel and inter-voxel coherence in patients with multiple sclerosis assessed using diffusion tensor MRI. J Neurol. 2002;249:875-883. doi:10.1007/s00415-002-0752-y
Song SK, Yoshino J, Le TQ, et al. Demyelination increases radial diffusivity in corpus callosum of mouse brain. Neuroimage. 2005;26:132-140. doi:10.1016/j.neuroimage.2005.01.028
Bagnato F, Franco G, Li H, et al. Probing axons using multi-compartmental diffusion in multiple sclerosis. Ann Clin Transl Neurol. 2019;6:1595-1605. doi:10.1002/acn3.50836
Filippi M, Cercignani M, Inglese M, et al. Diffusion tensor magnetic resonance imaging in multiple sclerosis. Neurology. 2001;56:304-311. doi:10.1212/wnl.56.3.304
Bagnato F. Clinical application of magnetization transfer imaging. In: Advanced Neuro MR Techniques and Applications. Elsevier; 2022:403-417. doi:10.1016/B978-0-12-822479-3.00041-5
Zheng Y, Lee JC, Rudick R, Fisher E. Long-term magnetization transfer ratio evolution in multiple sclerosis white matter lesions. J Neuroimaging. 2018;28:191-198. doi:10.1111/jon.12480
Bagnato F, Hametner S, Franco G, et al. Selective inversion recovery quantitative magnetization transfer brain MRI at 7T: clinical and postmortem validation in multiple sclerosis. J Neuroimaging. 2018;28:380-388. doi:10.1111/jon.12511
Clarke MA, Cheek R, Hernandez B, et al. Paramagnetic rim lesions and the central vein sign: characterizing multiple sclerosis imaging markers. J Neuroimaging. 2024;34:86-94. doi:10.1111/jon.13173
Clarke MA, Lakhani DA, Wen S, et al. Perilesional neurodegenerative injury in multiple sclerosis: relation to focal lesions and impact on disability. Mult Scler Relat Disord. 2021;49:102738. doi:10.1016/j.msard.2021.102738
Laule C, Moore GRW. Myelin water imaging to detect demyelination and remyelination and its validation in pathology. Brain Pathol. 2018;28:750-764. doi:10.1111/bpa.12645
Coelho S, Baete SH, Lemberskiy G, et al. Reproducibility of the standard model of diffusion in white matter on clinical MRI systems. Neuroimage. 2022;257:119290. doi:10.1016/j.neuroimage.2022.119290
Novikov DS, Veraart J, Jelescu IO, et al. Rotationally-invariant mapping of scalar and orientational metrics of neuronal microstructure with diffusion MRI. Neuroimage. 2018;174:518-538. doi:10.1016/j.neuroimage.2018.03.006
Langkammer C, Liu T, Khalil M, et al. Quantitative susceptibility mapping in multiple sclerosis. Radiology. 2013;267:551-559. doi:10.1148/radiol.12120707
Collorone S, Coll L, Lorenzi M, et al. Artificial intelligence applied to MRI data to tackle key challenges in multiple sclerosis. Mult Scler. 2024;30:767-784. doi:10.1177/13524585241249422
Updates in Multiple Sclerosis Imaging
Updates in Multiple Sclerosis Imaging