User login
Light therapy a beacon of hope for Alzheimer’s?
TOPLINE:
Light therapy leads to significant improvement in several sleep measures and helps alleviate depression and agitation in patients with Alzheimer’s disease (AD), a meta-analysis of 15 high-quality trials shows.
METHODOLOGY:
- This meta-analysis included 15 randomized controlled trials involving 598 patients with mild to moderate AD.
- The included trials were written in English, published between 2005 and 2022, and performed in seven countries. A fixed-effects model was used for data analysis.
TAKEAWAY:
- Light therapy significantly improved sleep efficiency (mean difference [MD], −2.42; P < .00001), increased interdaily stability (MD, −0.04; P < .00001), and reduced intradaily variability (MD, −0.04; P < .00001), indicating better sleep quality.
- Light therapy reduced agitation (MD, −3.97; P < .00001), depression (MD, −2.55; P < .00001), and caregiver burden (MD, −3.57; P < .00001).
- Light therapy also had a significant advantage over usual care in reducing the severity of psychobehavioral symptoms as assessed by the Neuropsychiatric Inventory (MD, −3.07; P < .00001).
- Light therapy had no statistically significant effect on improving cognitive function as measured by the Mini-Mental State Examination.
IN PRACTICE:
“These findings, combined with its low side-effects, suggest the role of light therapy as a promising treatment for AD. Although light therapy has fewer side effects than pharmacological treatment, adverse behavioral outcomes in patients due to bright light exposure should be considered,” the authors wrote.
SOURCE:
The study by Lili Zang and colleagues from Weifang Medical University School of Nursing, Shandong Province, China, was published online on December 6, 2023, in PLOS One.
LIMITATIONS:
The types and degrees of dementia in the included studies were inconsistent, potentially affecting the outcome indicators. Some articles did not clearly describe their randomization and allocation concealment methods, indicating possible bias in these studies.
DISCLOSURES:
The study was supported by the Natural Science Foundation of Shandong Province, China. The authors declared no competing interests.
Megan Brooks has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
Light therapy leads to significant improvement in several sleep measures and helps alleviate depression and agitation in patients with Alzheimer’s disease (AD), a meta-analysis of 15 high-quality trials shows.
METHODOLOGY:
- This meta-analysis included 15 randomized controlled trials involving 598 patients with mild to moderate AD.
- The included trials were written in English, published between 2005 and 2022, and performed in seven countries. A fixed-effects model was used for data analysis.
TAKEAWAY:
- Light therapy significantly improved sleep efficiency (mean difference [MD], −2.42; P < .00001), increased interdaily stability (MD, −0.04; P < .00001), and reduced intradaily variability (MD, −0.04; P < .00001), indicating better sleep quality.
- Light therapy reduced agitation (MD, −3.97; P < .00001), depression (MD, −2.55; P < .00001), and caregiver burden (MD, −3.57; P < .00001).
- Light therapy also had a significant advantage over usual care in reducing the severity of psychobehavioral symptoms as assessed by the Neuropsychiatric Inventory (MD, −3.07; P < .00001).
- Light therapy had no statistically significant effect on improving cognitive function as measured by the Mini-Mental State Examination.
IN PRACTICE:
“These findings, combined with its low side-effects, suggest the role of light therapy as a promising treatment for AD. Although light therapy has fewer side effects than pharmacological treatment, adverse behavioral outcomes in patients due to bright light exposure should be considered,” the authors wrote.
SOURCE:
The study by Lili Zang and colleagues from Weifang Medical University School of Nursing, Shandong Province, China, was published online on December 6, 2023, in PLOS One.
LIMITATIONS:
The types and degrees of dementia in the included studies were inconsistent, potentially affecting the outcome indicators. Some articles did not clearly describe their randomization and allocation concealment methods, indicating possible bias in these studies.
DISCLOSURES:
The study was supported by the Natural Science Foundation of Shandong Province, China. The authors declared no competing interests.
Megan Brooks has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
Light therapy leads to significant improvement in several sleep measures and helps alleviate depression and agitation in patients with Alzheimer’s disease (AD), a meta-analysis of 15 high-quality trials shows.
METHODOLOGY:
- This meta-analysis included 15 randomized controlled trials involving 598 patients with mild to moderate AD.
- The included trials were written in English, published between 2005 and 2022, and performed in seven countries. A fixed-effects model was used for data analysis.
TAKEAWAY:
- Light therapy significantly improved sleep efficiency (mean difference [MD], −2.42; P < .00001), increased interdaily stability (MD, −0.04; P < .00001), and reduced intradaily variability (MD, −0.04; P < .00001), indicating better sleep quality.
- Light therapy reduced agitation (MD, −3.97; P < .00001), depression (MD, −2.55; P < .00001), and caregiver burden (MD, −3.57; P < .00001).
- Light therapy also had a significant advantage over usual care in reducing the severity of psychobehavioral symptoms as assessed by the Neuropsychiatric Inventory (MD, −3.07; P < .00001).
- Light therapy had no statistically significant effect on improving cognitive function as measured by the Mini-Mental State Examination.
IN PRACTICE:
“These findings, combined with its low side-effects, suggest the role of light therapy as a promising treatment for AD. Although light therapy has fewer side effects than pharmacological treatment, adverse behavioral outcomes in patients due to bright light exposure should be considered,” the authors wrote.
SOURCE:
The study by Lili Zang and colleagues from Weifang Medical University School of Nursing, Shandong Province, China, was published online on December 6, 2023, in PLOS One.
LIMITATIONS:
The types and degrees of dementia in the included studies were inconsistent, potentially affecting the outcome indicators. Some articles did not clearly describe their randomization and allocation concealment methods, indicating possible bias in these studies.
DISCLOSURES:
The study was supported by the Natural Science Foundation of Shandong Province, China. The authors declared no competing interests.
Megan Brooks has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Depression, constipation, UTIs early signs of MS?
However, these prodromal symptoms are also more likely to occur in people with two other autoimmune diseases — lupus and Crohn’s disease — and therefore, will not help earlier diagnosis, study investigator, Céline Louapre, professor of neurology, Sorbonne University and Paris Brain Institute, Paris, France, said in an interview.
“On the other hand, in certain patients who may be at particular risk of developing MS, such as in certain familial forms or in patients with incidental inflammatory lesions discovered on MRI, the presence of these symptoms could suggest an already active process, prior to the first typical symptoms of the disease,” she noted.
Retracing MS Origins
The case-control study, published online in Neurology, included 20,174 people with newly diagnosed MS who were matched to 54,790 without MS, as well as 30,477 with Crohn’s disease and 7337 with lupus.
Using International Classification of Diseases, 10th revision (ICD-10) codes in electronic health records, the researchers assessed the associations between 113 diseases and symptoms in the 5 years before and after an MS diagnosis.
Twelve ICD-10 codes were significantly positively associated with the risk for MS compared with controls without MS.
After considering ICD-10 codes suggestive of neurologic symptoms as the first diagnosis of MS, the following five ICD-10 codes remained significantly associated with MS:
- Depression (odds ratio [OR], 1.22; 95% CI, 1.11-1.34)
- Sexual dysfunction (OR, 1.47; 95% CI, 1.11-1.95)
- Constipation (OR, 1.5; 95% CI, 1.27-1.78)
- Cystitis (OR, 1.21; 95% CI, 1.05-1.39)
- UTIs of unspecified site (OR, 1.38; 95% CI, 1.18-1.61)
However, none of these conditions was selectively associated with MS in comparison with both lupus and Crohn’s disease. All five ICD-10 codes identified were still associated with MS during the 5 years after diagnosis.
“The importance of investigating prodromal signs in MS is that it allows us to retrace the origins of the disease,” said Dr. Louapre.
“The main contribution of the data on prodromes in MS is to clarify that the disease and its mechanisms are frequently underway well before the first typical neurological symptoms, and that the causes of MS are probably present many years before diagnosis,” she added.
A limitation of the study was that data were not available for other factors that could influence people’s risk of developing MS, such as education level, ethnicity, body mass index, socioeconomic status, or genetic information.
It also remains unclear whether the conditions linked to MS are risk factors for the disease or nonspecific early MS symptoms.
Preventing Disease Evolution
In a linked editorial, Ruth Ann Marrie, MD, PhD, with the University of Manitoba, Manitoba, Canada, and Raffaele Palladino, MD, PhD, with the University of Naples Federico II, Naples, Italy, note these findings highlight the challenges of accurately identifying the prodromal stage of a specific disease.
“Commonalities of prodromal features are recognized across neurodegenerative diseases; this is also true for immune-mediated diseases, and it is not surprising, given shared etiologic factors and pathobiological mechanisms,” they point out.
“This suggests that we should be trying to link prodromal features to specific underlying pathobiological changes rather than specific diseases. This approach would require use of different study designs, including broad, deeply phenotyped cohorts, but would allow us to develop and test interventions targeted at those mechanisms, and could ultimately achieve the goal of preventing disease evolution,” they add.
The study was supported by the French National Research Agency. Dr. Louapre has received consulting or travel fees from Biogen, Novartis, Roche, Sanofi, Teva, and Merck Serono, unrelated to this study. Dr. Marrie is a coinvestigator on studies receiving funding from Biogen Idec and Roche Canada; receives research funding from CIHR, Research Manitoba, Multiple Sclerosis Society of Canada, Multiple Sclerosis Scientific Foundation, Crohn’s and Colitis Canada, National Multiple Sclerosis Society, CMSC, the Arthritis Society and the US Department of Defense; and serves on the editorial board of Neurology. Dr. Palladino has taken part in advisory boards/consultancy for MSD and Sanofi and has received support from the UK MS Society.
A version of this article appeared on Medscape.com.
However, these prodromal symptoms are also more likely to occur in people with two other autoimmune diseases — lupus and Crohn’s disease — and therefore, will not help earlier diagnosis, study investigator, Céline Louapre, professor of neurology, Sorbonne University and Paris Brain Institute, Paris, France, said in an interview.
“On the other hand, in certain patients who may be at particular risk of developing MS, such as in certain familial forms or in patients with incidental inflammatory lesions discovered on MRI, the presence of these symptoms could suggest an already active process, prior to the first typical symptoms of the disease,” she noted.
Retracing MS Origins
The case-control study, published online in Neurology, included 20,174 people with newly diagnosed MS who were matched to 54,790 without MS, as well as 30,477 with Crohn’s disease and 7337 with lupus.
Using International Classification of Diseases, 10th revision (ICD-10) codes in electronic health records, the researchers assessed the associations between 113 diseases and symptoms in the 5 years before and after an MS diagnosis.
Twelve ICD-10 codes were significantly positively associated with the risk for MS compared with controls without MS.
After considering ICD-10 codes suggestive of neurologic symptoms as the first diagnosis of MS, the following five ICD-10 codes remained significantly associated with MS:
- Depression (odds ratio [OR], 1.22; 95% CI, 1.11-1.34)
- Sexual dysfunction (OR, 1.47; 95% CI, 1.11-1.95)
- Constipation (OR, 1.5; 95% CI, 1.27-1.78)
- Cystitis (OR, 1.21; 95% CI, 1.05-1.39)
- UTIs of unspecified site (OR, 1.38; 95% CI, 1.18-1.61)
However, none of these conditions was selectively associated with MS in comparison with both lupus and Crohn’s disease. All five ICD-10 codes identified were still associated with MS during the 5 years after diagnosis.
“The importance of investigating prodromal signs in MS is that it allows us to retrace the origins of the disease,” said Dr. Louapre.
“The main contribution of the data on prodromes in MS is to clarify that the disease and its mechanisms are frequently underway well before the first typical neurological symptoms, and that the causes of MS are probably present many years before diagnosis,” she added.
A limitation of the study was that data were not available for other factors that could influence people’s risk of developing MS, such as education level, ethnicity, body mass index, socioeconomic status, or genetic information.
It also remains unclear whether the conditions linked to MS are risk factors for the disease or nonspecific early MS symptoms.
Preventing Disease Evolution
In a linked editorial, Ruth Ann Marrie, MD, PhD, with the University of Manitoba, Manitoba, Canada, and Raffaele Palladino, MD, PhD, with the University of Naples Federico II, Naples, Italy, note these findings highlight the challenges of accurately identifying the prodromal stage of a specific disease.
“Commonalities of prodromal features are recognized across neurodegenerative diseases; this is also true for immune-mediated diseases, and it is not surprising, given shared etiologic factors and pathobiological mechanisms,” they point out.
“This suggests that we should be trying to link prodromal features to specific underlying pathobiological changes rather than specific diseases. This approach would require use of different study designs, including broad, deeply phenotyped cohorts, but would allow us to develop and test interventions targeted at those mechanisms, and could ultimately achieve the goal of preventing disease evolution,” they add.
The study was supported by the French National Research Agency. Dr. Louapre has received consulting or travel fees from Biogen, Novartis, Roche, Sanofi, Teva, and Merck Serono, unrelated to this study. Dr. Marrie is a coinvestigator on studies receiving funding from Biogen Idec and Roche Canada; receives research funding from CIHR, Research Manitoba, Multiple Sclerosis Society of Canada, Multiple Sclerosis Scientific Foundation, Crohn’s and Colitis Canada, National Multiple Sclerosis Society, CMSC, the Arthritis Society and the US Department of Defense; and serves on the editorial board of Neurology. Dr. Palladino has taken part in advisory boards/consultancy for MSD and Sanofi and has received support from the UK MS Society.
A version of this article appeared on Medscape.com.
However, these prodromal symptoms are also more likely to occur in people with two other autoimmune diseases — lupus and Crohn’s disease — and therefore, will not help earlier diagnosis, study investigator, Céline Louapre, professor of neurology, Sorbonne University and Paris Brain Institute, Paris, France, said in an interview.
“On the other hand, in certain patients who may be at particular risk of developing MS, such as in certain familial forms or in patients with incidental inflammatory lesions discovered on MRI, the presence of these symptoms could suggest an already active process, prior to the first typical symptoms of the disease,” she noted.
Retracing MS Origins
The case-control study, published online in Neurology, included 20,174 people with newly diagnosed MS who were matched to 54,790 without MS, as well as 30,477 with Crohn’s disease and 7337 with lupus.
Using International Classification of Diseases, 10th revision (ICD-10) codes in electronic health records, the researchers assessed the associations between 113 diseases and symptoms in the 5 years before and after an MS diagnosis.
Twelve ICD-10 codes were significantly positively associated with the risk for MS compared with controls without MS.
After considering ICD-10 codes suggestive of neurologic symptoms as the first diagnosis of MS, the following five ICD-10 codes remained significantly associated with MS:
- Depression (odds ratio [OR], 1.22; 95% CI, 1.11-1.34)
- Sexual dysfunction (OR, 1.47; 95% CI, 1.11-1.95)
- Constipation (OR, 1.5; 95% CI, 1.27-1.78)
- Cystitis (OR, 1.21; 95% CI, 1.05-1.39)
- UTIs of unspecified site (OR, 1.38; 95% CI, 1.18-1.61)
However, none of these conditions was selectively associated with MS in comparison with both lupus and Crohn’s disease. All five ICD-10 codes identified were still associated with MS during the 5 years after diagnosis.
“The importance of investigating prodromal signs in MS is that it allows us to retrace the origins of the disease,” said Dr. Louapre.
“The main contribution of the data on prodromes in MS is to clarify that the disease and its mechanisms are frequently underway well before the first typical neurological symptoms, and that the causes of MS are probably present many years before diagnosis,” she added.
A limitation of the study was that data were not available for other factors that could influence people’s risk of developing MS, such as education level, ethnicity, body mass index, socioeconomic status, or genetic information.
It also remains unclear whether the conditions linked to MS are risk factors for the disease or nonspecific early MS symptoms.
Preventing Disease Evolution
In a linked editorial, Ruth Ann Marrie, MD, PhD, with the University of Manitoba, Manitoba, Canada, and Raffaele Palladino, MD, PhD, with the University of Naples Federico II, Naples, Italy, note these findings highlight the challenges of accurately identifying the prodromal stage of a specific disease.
“Commonalities of prodromal features are recognized across neurodegenerative diseases; this is also true for immune-mediated diseases, and it is not surprising, given shared etiologic factors and pathobiological mechanisms,” they point out.
“This suggests that we should be trying to link prodromal features to specific underlying pathobiological changes rather than specific diseases. This approach would require use of different study designs, including broad, deeply phenotyped cohorts, but would allow us to develop and test interventions targeted at those mechanisms, and could ultimately achieve the goal of preventing disease evolution,” they add.
The study was supported by the French National Research Agency. Dr. Louapre has received consulting or travel fees from Biogen, Novartis, Roche, Sanofi, Teva, and Merck Serono, unrelated to this study. Dr. Marrie is a coinvestigator on studies receiving funding from Biogen Idec and Roche Canada; receives research funding from CIHR, Research Manitoba, Multiple Sclerosis Society of Canada, Multiple Sclerosis Scientific Foundation, Crohn’s and Colitis Canada, National Multiple Sclerosis Society, CMSC, the Arthritis Society and the US Department of Defense; and serves on the editorial board of Neurology. Dr. Palladino has taken part in advisory boards/consultancy for MSD and Sanofi and has received support from the UK MS Society.
A version of this article appeared on Medscape.com.
FROM NEUROLOGY
Toward a better framework for postmarketing reproductive safety surveillance of medications
For the last 30 years, the Center for Women’s Mental Health at Massachusetts General Hospital (MGH) has had as part of its mission, the conveying of accurate information about the reproductive safety of psychiatric medications. There has been a spectrum of medicines developed across psychiatric indications over the last several decades, and many studies over those decades have attempted to delineate the reproductive safety of these agents.
With the development of new antidepressants and second-generation antipsychotics has come an appreciation of the utility of these agents across a wide range of psychiatric disease states and psychiatric symptoms. More and more data demonstrate the efficacy of these medicines for mood and anxiety disorders; these agents are also used for a broad array of symptoms from insomnia, irritability, and symptoms of posttraumatic stress disorder (PTSD) just as examples — even absent formal approval by the US Food and Drug Administration (FDA) for these specific indications. With the growing use of medicines, including new antidepressants like selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors, and second-generation atypical antipsychotics, there has been a greater interest and appreciation of the need to provide women with the best information about reproductive safety of these medicines as well.
When I began working in reproductive psychiatry, the FDA was using the pregnancy labeling categories introduced in 1979. The categories were simple, but also oversimplified in terms of incompletely conveying information about reproductive safety. For instance, category labels of B and C under the old labeling system could be nebulous, containing sparse information (in the case of category B) or animal data and some conflicting human data (in the case of category C) that may not have translated into relevant or easily interpretable safety information for patients and clinicians.
It was on that basis the current Pregnancy and Lactation Labeling (PLLR) Final Rule was published in 2014, which was a shift from categorical labeling to more descriptive labeling, including updated actual information on the package insert about available reproductive safety data, animal data, and data on lactation.
Even following the publication of the PLLR, there has still been an acknowledgment in the field that our assessment tools for postmarketing reproductive safety surveillance are incomplete. A recent 2-day FDA workshop hosted by the Duke-Margolis Center for Health Policy on optimizing the use of postapproval pregnancy safety studies sought to discuss the many questions that still surround this issue. Based on presentations at this workshop, a framework emerged for the future of assessing the reproductive safety of medications, which included an effort to develop the most effective model using tools such as pregnancy registries and harnessing “big data,” whether through electronic health records or large administrative databases from public and private insurers. Together, these various sources of information can provide signals of potential concern, prompting the need for a more rigorous look at the reproductive safety of a medication, or provide reassurance if data fail to indicate the absence of a signal of risk.
FDA’s new commitments under the latest reauthorization of the Prescription Drug User Fee Act (PDUFA VII) include pregnancy-specific postmarketing safety requirements as well as the creation of a framework for how data from pregnancy-specific postmarketing studies can be used. The agency is also conducting demonstration projects, including one for assessing the performance of pregnancy registries for the potential to detect safety signals for medications early in pregnancy. FDA is expanding its Sentinel Initiative to help accomplish these aims, and is implementing an Active Risk Identification and Analysis (ARIA) system to conduct active safety surveillance of medications used during pregnancy.
Pregnancy registries have now been available for decades, and some have been more successful than others across different classes of medicines, with the most rigorous registries including prospective follow-up of women across pregnancies and careful documentation of malformations (at best with original source data and with a blinded dysmorphologist). Still, with all of its rigor, even the best-intentioned efforts with respect to pregnancy registries have limitations. As I mentioned in my testimony during the public comment portion of the workshop, the sheer volume of pregnancy data from administrative databases we now have access to is attractive, but the quality of these data needs to be good enough to ascertain a signal of risk if they are to be used as a basis for reproductive safety determination.
The flip side of using data from large administrative databases is using carefully collected data from pregnancy registries. With a pregnancy registry, accrual of a substantial number of participants can also take a considerable period of time, and initial risk estimates of outcomes can have typically large confidence intervals, which can make it difficult to discern whether a drug is safe for women of reproductive age.
Another key issue is a lack of participation from manufacturers with respect to commitment to collection of high-quality reproductive safety data. History has shown that many medication manufacturers, unless required to have a dedicated registry as part of a postmarketing requirement or commitment, will invest sparse resources to track data on safety of fetal drug exposure. Participation is typically voluntary and varies from company to company unless, as noted previously, there is a postmarketing requirement or commitment tied to the approval of a medication. Just as a recent concrete example, the manufacturer of a new medication recently approved by the FDA for the treatment of postpartum depression (which will include presumably sexually active women well into the first postpartum year) has no plan to support the collection of reproductive safety data on this new medication because it is not required to, based on current FDA guidelines and the absence of a postmarketing requirement to do so.
Looking ahead
While the PLLR was a huge step forward in the field from the old pregnancy category system that could misinform women contemplating pregnancy, it also sets the stage for the next iteration of a system that allows us to generate information more quickly about the reproductive safety of medications. In psychiatry, as many as 10% of women use SSRIs during pregnancy. With drugs like atypical antipsychotics being used across disease states — in schizophrenia, bipolar disorder, depression, anxiety, insomnia, and PTSD — and where new classes of medicine are becoming available, like with ketamine or steroids, we need to have a system by which we can more quickly ascertain reproductive safety information. This information informs treatment decisions during a critical life event of deciding to try to become pregnant or during an actual pregnancy.
In my mind, it is reassuring when a registry has even as few as 50-60 cases of fetal exposure without an increase in the risk for malformation, because it can mean we are not seeing a repeat of the past with medications like thalidomide and sodium valproate. However, patients and clinicians are starved for better data. Risk assessment is also different from clinician to clinician and patient to patient. We want to empower patients to make decisions that work for them based on more rapidly accumulating information and help inform their decisions.
To come out on the “other side” of the PLLR, , which can be confusing when study results frequently conflict. I believe we have an obligation today to do this better, because the areas of reproductive toxicology and pharmacovigilance are growing incredibly quickly, and clinicians and patients are seeing these volumes of data being published without the ability to integrate that information in a systematic way.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital (MGH) in Boston, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Full disclosure information for Dr. Cohen is available at womensmentalhealth.org. Email Dr. Cohen at obnews@mdedge.com.
For the last 30 years, the Center for Women’s Mental Health at Massachusetts General Hospital (MGH) has had as part of its mission, the conveying of accurate information about the reproductive safety of psychiatric medications. There has been a spectrum of medicines developed across psychiatric indications over the last several decades, and many studies over those decades have attempted to delineate the reproductive safety of these agents.
With the development of new antidepressants and second-generation antipsychotics has come an appreciation of the utility of these agents across a wide range of psychiatric disease states and psychiatric symptoms. More and more data demonstrate the efficacy of these medicines for mood and anxiety disorders; these agents are also used for a broad array of symptoms from insomnia, irritability, and symptoms of posttraumatic stress disorder (PTSD) just as examples — even absent formal approval by the US Food and Drug Administration (FDA) for these specific indications. With the growing use of medicines, including new antidepressants like selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors, and second-generation atypical antipsychotics, there has been a greater interest and appreciation of the need to provide women with the best information about reproductive safety of these medicines as well.
When I began working in reproductive psychiatry, the FDA was using the pregnancy labeling categories introduced in 1979. The categories were simple, but also oversimplified in terms of incompletely conveying information about reproductive safety. For instance, category labels of B and C under the old labeling system could be nebulous, containing sparse information (in the case of category B) or animal data and some conflicting human data (in the case of category C) that may not have translated into relevant or easily interpretable safety information for patients and clinicians.
It was on that basis the current Pregnancy and Lactation Labeling (PLLR) Final Rule was published in 2014, which was a shift from categorical labeling to more descriptive labeling, including updated actual information on the package insert about available reproductive safety data, animal data, and data on lactation.
Even following the publication of the PLLR, there has still been an acknowledgment in the field that our assessment tools for postmarketing reproductive safety surveillance are incomplete. A recent 2-day FDA workshop hosted by the Duke-Margolis Center for Health Policy on optimizing the use of postapproval pregnancy safety studies sought to discuss the many questions that still surround this issue. Based on presentations at this workshop, a framework emerged for the future of assessing the reproductive safety of medications, which included an effort to develop the most effective model using tools such as pregnancy registries and harnessing “big data,” whether through electronic health records or large administrative databases from public and private insurers. Together, these various sources of information can provide signals of potential concern, prompting the need for a more rigorous look at the reproductive safety of a medication, or provide reassurance if data fail to indicate the absence of a signal of risk.
FDA’s new commitments under the latest reauthorization of the Prescription Drug User Fee Act (PDUFA VII) include pregnancy-specific postmarketing safety requirements as well as the creation of a framework for how data from pregnancy-specific postmarketing studies can be used. The agency is also conducting demonstration projects, including one for assessing the performance of pregnancy registries for the potential to detect safety signals for medications early in pregnancy. FDA is expanding its Sentinel Initiative to help accomplish these aims, and is implementing an Active Risk Identification and Analysis (ARIA) system to conduct active safety surveillance of medications used during pregnancy.
Pregnancy registries have now been available for decades, and some have been more successful than others across different classes of medicines, with the most rigorous registries including prospective follow-up of women across pregnancies and careful documentation of malformations (at best with original source data and with a blinded dysmorphologist). Still, with all of its rigor, even the best-intentioned efforts with respect to pregnancy registries have limitations. As I mentioned in my testimony during the public comment portion of the workshop, the sheer volume of pregnancy data from administrative databases we now have access to is attractive, but the quality of these data needs to be good enough to ascertain a signal of risk if they are to be used as a basis for reproductive safety determination.
The flip side of using data from large administrative databases is using carefully collected data from pregnancy registries. With a pregnancy registry, accrual of a substantial number of participants can also take a considerable period of time, and initial risk estimates of outcomes can have typically large confidence intervals, which can make it difficult to discern whether a drug is safe for women of reproductive age.
Another key issue is a lack of participation from manufacturers with respect to commitment to collection of high-quality reproductive safety data. History has shown that many medication manufacturers, unless required to have a dedicated registry as part of a postmarketing requirement or commitment, will invest sparse resources to track data on safety of fetal drug exposure. Participation is typically voluntary and varies from company to company unless, as noted previously, there is a postmarketing requirement or commitment tied to the approval of a medication. Just as a recent concrete example, the manufacturer of a new medication recently approved by the FDA for the treatment of postpartum depression (which will include presumably sexually active women well into the first postpartum year) has no plan to support the collection of reproductive safety data on this new medication because it is not required to, based on current FDA guidelines and the absence of a postmarketing requirement to do so.
Looking ahead
While the PLLR was a huge step forward in the field from the old pregnancy category system that could misinform women contemplating pregnancy, it also sets the stage for the next iteration of a system that allows us to generate information more quickly about the reproductive safety of medications. In psychiatry, as many as 10% of women use SSRIs during pregnancy. With drugs like atypical antipsychotics being used across disease states — in schizophrenia, bipolar disorder, depression, anxiety, insomnia, and PTSD — and where new classes of medicine are becoming available, like with ketamine or steroids, we need to have a system by which we can more quickly ascertain reproductive safety information. This information informs treatment decisions during a critical life event of deciding to try to become pregnant or during an actual pregnancy.
In my mind, it is reassuring when a registry has even as few as 50-60 cases of fetal exposure without an increase in the risk for malformation, because it can mean we are not seeing a repeat of the past with medications like thalidomide and sodium valproate. However, patients and clinicians are starved for better data. Risk assessment is also different from clinician to clinician and patient to patient. We want to empower patients to make decisions that work for them based on more rapidly accumulating information and help inform their decisions.
To come out on the “other side” of the PLLR, , which can be confusing when study results frequently conflict. I believe we have an obligation today to do this better, because the areas of reproductive toxicology and pharmacovigilance are growing incredibly quickly, and clinicians and patients are seeing these volumes of data being published without the ability to integrate that information in a systematic way.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital (MGH) in Boston, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Full disclosure information for Dr. Cohen is available at womensmentalhealth.org. Email Dr. Cohen at obnews@mdedge.com.
For the last 30 years, the Center for Women’s Mental Health at Massachusetts General Hospital (MGH) has had as part of its mission, the conveying of accurate information about the reproductive safety of psychiatric medications. There has been a spectrum of medicines developed across psychiatric indications over the last several decades, and many studies over those decades have attempted to delineate the reproductive safety of these agents.
With the development of new antidepressants and second-generation antipsychotics has come an appreciation of the utility of these agents across a wide range of psychiatric disease states and psychiatric symptoms. More and more data demonstrate the efficacy of these medicines for mood and anxiety disorders; these agents are also used for a broad array of symptoms from insomnia, irritability, and symptoms of posttraumatic stress disorder (PTSD) just as examples — even absent formal approval by the US Food and Drug Administration (FDA) for these specific indications. With the growing use of medicines, including new antidepressants like selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors, and second-generation atypical antipsychotics, there has been a greater interest and appreciation of the need to provide women with the best information about reproductive safety of these medicines as well.
When I began working in reproductive psychiatry, the FDA was using the pregnancy labeling categories introduced in 1979. The categories were simple, but also oversimplified in terms of incompletely conveying information about reproductive safety. For instance, category labels of B and C under the old labeling system could be nebulous, containing sparse information (in the case of category B) or animal data and some conflicting human data (in the case of category C) that may not have translated into relevant or easily interpretable safety information for patients and clinicians.
It was on that basis the current Pregnancy and Lactation Labeling (PLLR) Final Rule was published in 2014, which was a shift from categorical labeling to more descriptive labeling, including updated actual information on the package insert about available reproductive safety data, animal data, and data on lactation.
Even following the publication of the PLLR, there has still been an acknowledgment in the field that our assessment tools for postmarketing reproductive safety surveillance are incomplete. A recent 2-day FDA workshop hosted by the Duke-Margolis Center for Health Policy on optimizing the use of postapproval pregnancy safety studies sought to discuss the many questions that still surround this issue. Based on presentations at this workshop, a framework emerged for the future of assessing the reproductive safety of medications, which included an effort to develop the most effective model using tools such as pregnancy registries and harnessing “big data,” whether through electronic health records or large administrative databases from public and private insurers. Together, these various sources of information can provide signals of potential concern, prompting the need for a more rigorous look at the reproductive safety of a medication, or provide reassurance if data fail to indicate the absence of a signal of risk.
FDA’s new commitments under the latest reauthorization of the Prescription Drug User Fee Act (PDUFA VII) include pregnancy-specific postmarketing safety requirements as well as the creation of a framework for how data from pregnancy-specific postmarketing studies can be used. The agency is also conducting demonstration projects, including one for assessing the performance of pregnancy registries for the potential to detect safety signals for medications early in pregnancy. FDA is expanding its Sentinel Initiative to help accomplish these aims, and is implementing an Active Risk Identification and Analysis (ARIA) system to conduct active safety surveillance of medications used during pregnancy.
Pregnancy registries have now been available for decades, and some have been more successful than others across different classes of medicines, with the most rigorous registries including prospective follow-up of women across pregnancies and careful documentation of malformations (at best with original source data and with a blinded dysmorphologist). Still, with all of its rigor, even the best-intentioned efforts with respect to pregnancy registries have limitations. As I mentioned in my testimony during the public comment portion of the workshop, the sheer volume of pregnancy data from administrative databases we now have access to is attractive, but the quality of these data needs to be good enough to ascertain a signal of risk if they are to be used as a basis for reproductive safety determination.
The flip side of using data from large administrative databases is using carefully collected data from pregnancy registries. With a pregnancy registry, accrual of a substantial number of participants can also take a considerable period of time, and initial risk estimates of outcomes can have typically large confidence intervals, which can make it difficult to discern whether a drug is safe for women of reproductive age.
Another key issue is a lack of participation from manufacturers with respect to commitment to collection of high-quality reproductive safety data. History has shown that many medication manufacturers, unless required to have a dedicated registry as part of a postmarketing requirement or commitment, will invest sparse resources to track data on safety of fetal drug exposure. Participation is typically voluntary and varies from company to company unless, as noted previously, there is a postmarketing requirement or commitment tied to the approval of a medication. Just as a recent concrete example, the manufacturer of a new medication recently approved by the FDA for the treatment of postpartum depression (which will include presumably sexually active women well into the first postpartum year) has no plan to support the collection of reproductive safety data on this new medication because it is not required to, based on current FDA guidelines and the absence of a postmarketing requirement to do so.
Looking ahead
While the PLLR was a huge step forward in the field from the old pregnancy category system that could misinform women contemplating pregnancy, it also sets the stage for the next iteration of a system that allows us to generate information more quickly about the reproductive safety of medications. In psychiatry, as many as 10% of women use SSRIs during pregnancy. With drugs like atypical antipsychotics being used across disease states — in schizophrenia, bipolar disorder, depression, anxiety, insomnia, and PTSD — and where new classes of medicine are becoming available, like with ketamine or steroids, we need to have a system by which we can more quickly ascertain reproductive safety information. This information informs treatment decisions during a critical life event of deciding to try to become pregnant or during an actual pregnancy.
In my mind, it is reassuring when a registry has even as few as 50-60 cases of fetal exposure without an increase in the risk for malformation, because it can mean we are not seeing a repeat of the past with medications like thalidomide and sodium valproate. However, patients and clinicians are starved for better data. Risk assessment is also different from clinician to clinician and patient to patient. We want to empower patients to make decisions that work for them based on more rapidly accumulating information and help inform their decisions.
To come out on the “other side” of the PLLR, , which can be confusing when study results frequently conflict. I believe we have an obligation today to do this better, because the areas of reproductive toxicology and pharmacovigilance are growing incredibly quickly, and clinicians and patients are seeing these volumes of data being published without the ability to integrate that information in a systematic way.
Dr. Cohen is the director of the Ammon-Pinizzotto Center for Women’s Mental Health at Massachusetts General Hospital (MGH) in Boston, which provides information resources and conducts clinical care and research in reproductive mental health. He has been a consultant to manufacturers of psychiatric medications. Full disclosure information for Dr. Cohen is available at womensmentalhealth.org. Email Dr. Cohen at obnews@mdedge.com.
1 in 3 women have lasting health problems after giving birth: Study
Those problems include pain during sexual intercourse (35%), low back pain (32%), urinary incontinence (8% to 31%), anxiety (9% to 24%), anal incontinence (19%), depression (11% to 17%), fear of childbirth (6% to 15%), perineal pain (11%), and secondary infertility (11%).
Other problems included pelvic organ prolapse, posttraumatic stress disorder, thyroid dysfunction, mastitis, HIV seroconversion (when the body begins to produce detectable levels of HIV antibodies), nerve injury, and psychosis.
The study says most women see a doctor 6 to 12 weeks after birth and then rarely talk to doctors about these nagging health problems. Many of the problems don’t show up until 6 or more weeks after birth.
“To comprehensively address these conditions, broader and more comprehensive health service opportunities are needed, which should extend beyond 6 weeks postpartum and embrace multidisciplinary models of care,” the study says. “This approach can ensure that these conditions are promptly identified and given the attention that they deserve.”
The study is part of a series organized by the United Nation’s Special Program on Human Reproduction, the World Health Organization, and the U.S. Agency for International Development. The authors said most of the data came from high-income nations. There was little data from low-income and middle-income countries except for postpartum depression, anxiety, and psychosis.
“Many postpartum conditions cause considerable suffering in women’s daily life long after birth, both emotionally and physically, and yet they are largely underappreciated, underrecognized, and underreported,” Pascale Allotey, MD, director of Sexual and Reproductive Health and Research at WHO, said in a statement.
“Throughout their lives, and beyond motherhood, women need access to a range of services from health-care providers who listen to their concerns and meet their needs — so they not only survive childbirth but can enjoy good health and quality of life.”
A version of this article appeared on WebMD.com.
Those problems include pain during sexual intercourse (35%), low back pain (32%), urinary incontinence (8% to 31%), anxiety (9% to 24%), anal incontinence (19%), depression (11% to 17%), fear of childbirth (6% to 15%), perineal pain (11%), and secondary infertility (11%).
Other problems included pelvic organ prolapse, posttraumatic stress disorder, thyroid dysfunction, mastitis, HIV seroconversion (when the body begins to produce detectable levels of HIV antibodies), nerve injury, and psychosis.
The study says most women see a doctor 6 to 12 weeks after birth and then rarely talk to doctors about these nagging health problems. Many of the problems don’t show up until 6 or more weeks after birth.
“To comprehensively address these conditions, broader and more comprehensive health service opportunities are needed, which should extend beyond 6 weeks postpartum and embrace multidisciplinary models of care,” the study says. “This approach can ensure that these conditions are promptly identified and given the attention that they deserve.”
The study is part of a series organized by the United Nation’s Special Program on Human Reproduction, the World Health Organization, and the U.S. Agency for International Development. The authors said most of the data came from high-income nations. There was little data from low-income and middle-income countries except for postpartum depression, anxiety, and psychosis.
“Many postpartum conditions cause considerable suffering in women’s daily life long after birth, both emotionally and physically, and yet they are largely underappreciated, underrecognized, and underreported,” Pascale Allotey, MD, director of Sexual and Reproductive Health and Research at WHO, said in a statement.
“Throughout their lives, and beyond motherhood, women need access to a range of services from health-care providers who listen to their concerns and meet their needs — so they not only survive childbirth but can enjoy good health and quality of life.”
A version of this article appeared on WebMD.com.
Those problems include pain during sexual intercourse (35%), low back pain (32%), urinary incontinence (8% to 31%), anxiety (9% to 24%), anal incontinence (19%), depression (11% to 17%), fear of childbirth (6% to 15%), perineal pain (11%), and secondary infertility (11%).
Other problems included pelvic organ prolapse, posttraumatic stress disorder, thyroid dysfunction, mastitis, HIV seroconversion (when the body begins to produce detectable levels of HIV antibodies), nerve injury, and psychosis.
The study says most women see a doctor 6 to 12 weeks after birth and then rarely talk to doctors about these nagging health problems. Many of the problems don’t show up until 6 or more weeks after birth.
“To comprehensively address these conditions, broader and more comprehensive health service opportunities are needed, which should extend beyond 6 weeks postpartum and embrace multidisciplinary models of care,” the study says. “This approach can ensure that these conditions are promptly identified and given the attention that they deserve.”
The study is part of a series organized by the United Nation’s Special Program on Human Reproduction, the World Health Organization, and the U.S. Agency for International Development. The authors said most of the data came from high-income nations. There was little data from low-income and middle-income countries except for postpartum depression, anxiety, and psychosis.
“Many postpartum conditions cause considerable suffering in women’s daily life long after birth, both emotionally and physically, and yet they are largely underappreciated, underrecognized, and underreported,” Pascale Allotey, MD, director of Sexual and Reproductive Health and Research at WHO, said in a statement.
“Throughout their lives, and beyond motherhood, women need access to a range of services from health-care providers who listen to their concerns and meet their needs — so they not only survive childbirth but can enjoy good health and quality of life.”
A version of this article appeared on WebMD.com.
FROM THE LANCET GLOBAL HEALTH
Breastfeeding by patients with serious mental illness: An ethical approach
Difficult ethical situations can arise when treating perinatal women who have serious mental illness (SMI). Clinicians must consider ethical issues related to administering antipsychotic medications, the safety of breastfeeding, and concerns for child welfare. They need to carefully weigh the risks and benefits of each decision when treating perinatal women who have SMI. Ethical guidelines can help clinicians best support families in these situations.
In this article, we describe 2 cases of women with psychotic disorders who requested to breastfeed after delivering their child during an inpatient psychiatric hospitalization. The course of their hospitalizations illustrated common ethical questions and facilitated the creation of a framework to assist with complex decision-making regarding breastfeeding on inpatient psychiatric units.
CASE 1
Ms. C, age 41, is multigravida with a psychiatric history of chronic, severe schizoaffective disorder and lives in supportive housing. When Ms. C presents to the hospital in search of a rape kit, clinicians discover she is 22 weeks pregnant but has not received any prenatal care. Psychiatry is consulted because she is found to be intermittently agitated and endorses grandiose delusions. Ms. C requires involuntary hospitalization for decompensated psychosis because she refuses prenatal and psychiatric care. Because it has reassuring reproductive safety data,1 olanzapine 5 mg/d is started. However, Ms. C experiences minimal improvement from a maximum dose of 20 mg/d. After 13 weeks on the psychiatry unit, she is transferred to obstetrics service for preeclampsia with severe features. Ms. C requires an urgent cesarean delivery at 37 weeks. Her baby boy is transferred to the neonatal intensive care unit (NICU) for transient tachypnea. After delivery and in consultation with psychiatry, the pediatrics team calls Child Protective Services (CPS) due to concern for neglect driven by Ms. C’s psychiatric condition. Ms. C visits the child with medical unit staff supervision in the NICU without consulting with the psychiatry service or CPS. On postpartum Day 2, Ms. C is transferred back to psychiatry for persistent psychosis.
On postpartum Day 3, Ms. C starts to produce breastmilk and requests to breastfeed. At this time, the multidisciplinary team determines she is not able to visit her child in the NICU due to psychiatric instability. No plan is developed to facilitate hand expression or pumping of breastmilk while Ms. C is on the psychiatric unit. The clinical teams discuss whether the benefits of breastfeeding and/or pumping breastmilk would outweigh the risks. CPS determines that Ms. C is unable to retain custody and places the child in kinship foster care while awaiting clinical improvement from her.
CASE 2
Ms. S, age 32, has a history of schizophrenia. She lives with her husband and parents. She is pregnant for the first time and has been receiving consistent prenatal care. Ms. S is brought to the hospital by her husband for bizarre behavior and paranoia after self-discontinuing risperidone 2 mg twice daily due to concern about the medication’s influence on her pregnancy. An ultrasound confirms she is 37 weeks pregnant. Psychiatry is consulted because Ms. S is internally preoccupied, delusional, and endorses auditory hallucinations. She requires involuntary hospitalization for decompensated psychosis. During admission, Ms. S experiences improvement of her psychiatric symptoms while receiving risperidone 2 mg twice daily, which she takes consistently after receiving extensive psychoeducation regarding its safety profile during pregnancy and lactation.
After 2 weeks on the psychiatry unit, Ms. S’s care team transfers her to the obstetrics service with one-to-one supervision. At 39 weeks gestation, she has a vaginal delivery without complications. Because there are no concerns about infant harm, obstetrics, pediatrics, and psychiatry coordinate care so the baby can room in with Ms. S, her husband, and a staff supervisor to facilitate bonding. Ms. S starts to lactate, wishes to breastfeed, and meets with lactation, pediatric, obstetric, and psychiatric specialists to discuss the risks and benefits of breastfeeding and pumping breastmilk. She pursues direct breastfeeding until the baby is discharged home with the husband at postpartum Day 2. CPS is not called because there are no concerns for parental abuse or neglect at the infant’s discharge.
On postpartum Day 2, the obstetrics service transfers Ms. S back to the psychiatric unit for further treatment of her paranoia. She wishes to pump breastmilk while hospitalized, so the treatment team supplies a breast pump, facilitates the storage of breastmilk, and coordinates supervision during pumping to reduce the ligature risk. Ms. S’s husband visits daily to transport the milk and feed the infant breastmilk and formula to meet its nutritional needs. Ms. S maintains psychiatric stability while breast pumping, and the team helps transition her to breastfeeding during visitation with her husband and infant until she is discharged home at 2 weeks postpartum.
Continue to: Approaching care with a relational ethics framework
Approaching care with a relational ethics framework
A relational ethics framework was constructed to evaluate whether to support breastfeeding for both patients during their psychiatric hospitalizations. A relational ethics perspective is defined as “a moral responsibility within a context of human relations” [that] “recognizes the human interdependency and reciprocity within which personal autonomy is embedded.”2 This framework values connectedness and commonality between various and even conflicting parties. In the setting of a clinician-patient relationship, health care decisions are made with consideration of the patient’s traditional beliefs, values, and principles rather than the application of impartial moral principles. For these complex cases, this framework was chosen to determine the safest possible outcome for both mother and child.
Risks/benefits of breastfeeding by patients who have SMI
There are several methods of breastfeeding, including direct breastfeeding and other ways of expressing breastmilk such as pumping or hand expression.3 Unlike other forms of feeding using breastmilk, direct breastfeeding has been extensively studied, has well-established medical and psychological benefits for newborns and mothers, and enhances long-term bonding.4 Compared with their counterparts who do not breastfeed, mothers who breastfeed have lower rates of unintended pregnancy, cardiovascular disease, postpartum bleeding, osteoporosis, and breast and ovarian cancer.5 Among its key psychological benefits, breastfeeding is associated with an increase in maternal self-efficacy and, in some research, has been shown to be associated with a decreased risk of postpartum depression and stress.Additionally, breastfed infants experience lower rates of childhood infection and obesity, and improved nutrition, cognitive development, and immune function.6 The American Academy of Pediatrics recognizes these benefits and recommends that women exclusively breastfeed for 6 months postpartum and continue to breastfeed for 2 years or beyond if mutually desired by the mother and child.7 Absolute contraindications to breastfeeding must be ruled out (eg, infant classic galactosemia; maternal use of illicit substances such as cocaine, opioids, or phencyclidine; maternal HIV infection, etc).
The risks of breastfeeding by patients who have SMI must also be considered. In severe situations, the infant can be exposed to a mother’s agitation secondary to psychosis.8,9 The transmission of antipsychotic medication through breastmilk and associated adverse effects (eg, sedation, poor feeding, and extrapyramidal symptoms) are also potential risks and varies among different antipsychotic medications.1,10 Therefore, when prescribing an antipsychotic for a patient with SMI who breastfeeds, it is crucial to consider the medication’s safety profile as well as other factors, such as the relative infant dose (the weight-adjusted [ie, mg/kg] percentage of the maternal dosage ingested by a fully breastfed infant) and the molecular characteristics of the medication.10-12 Neonates should be routinely monitored for adverse effects, medication toxicity, and withdrawal symptoms, and care should be coordinated with the infant’s pediatrician. Certain antipsychotic medications, such as aripiprazole, may impact breastmilk production through the dopamine agonist’s interference of the prolactin reflex and anticholinergic properties.11,13 For a patient with SMI, perhaps the most significant risk involves the time and resources needed for breastfeeding, which can interfere with sleep and psychiatric treatment and possibly further exacerbate psychiatric symptoms.14-16 Additionally, breastfeeding difficulties or disruption can increase the risk of psychiatric symptoms and psychological distress.17 In Ms. C’s case, there was a delay in the baby latching as well as multiple medical and psychiatric factors that hindered the milk-ejection reflex to properly initiate; both of these factors rendered breastfeeding particularly difficult while Ms. C was on the inpatient psychiatry unit.17 In comparison, Ms. S was able to bond with her infant shortly after delivery, which facilitated the milk-ejection reflex and lactation.
Patients who wish to directly breastfeed but struggle to do so while tending to their acute psychiatric condition can benefit from expression of breastmilk that can be provided to the infant or discarded to facilitate breastfeeding in the future.18 While expression of breastmilk may not be as advantageous for infant health as direct breastfeeding due to the potential changes in breastmilk composition from collecting, storing, and heating, this option can be more protective than formula feeding and facilitate future breastfeeding.19 In these clinical scenarios, it is standard care to provide a hospital-grade breast pump to the patient, much like a continuous positive airway pressure machine is provided to patients with obstructive sleep apnea.20 However, there is often considerable difficulty obtaining proper breastfeeding equipment and a lack of services devoted to perinatal care in general inpatient settings. Barriers to direct breastfeeding and pumping of breastmilk are highlighted in the Table.21

Limitations on breastfeeding on an inpatient unit
The limitations in care and restrictions placed on breastfeeding are more optimally addressed in a mother and baby unit (MBU). MBUs are specialized inpatient psychiatric units designed for mothers experiencing severe perinatal psychiatric difficulties. Unlike general psychiatric units, MBUs allow for joint, full-time admission of mothers and their infants. These units also include multidisciplinary staff who specialize in treating perinatal mental health issues as well as infant care and child development.22 Admission into an MBU is considered best practice for new mothers requiring treatment, particularly in the United Kingdom, Australia, and France, as it is well-recognized that the separation of mother and baby can be psychologically harmful.23 In the UK, most patients admitted to an MBU showed significant improvement of their psychiatric symptoms and reported overall high satisfaction with care.24,25 Patients who experience postpartum psychosis prefer MBUs over general psychiatric units because the latter often lack specialized perinatal support, appropriate visitor arrangements, and adequate time with their infant.26-28
Continue to: The resistance to adopting MBUs in the United States...
The resistance to adopting MBUs in the United States has posed significant barriers in care for perinatal patients and has been attributed to financial barriers, medicolegal risk, staffing, and safety concerns.29 Though currently there are no MBUs in the US, other specialized units have been created. A partial day hospitalization program created in 2000 in Rhode Island for mothers and infants revolutionized the psychiatric care experience for new mothers.30 Since then, other institutions have significantly expanded their services to include perinatal psychiatry inpatient units, yet unlike MBUs, these units typically do not provide overnight rooming-in with infants.31 They have the necessary resources and facilities to accommodate the mother’s needs and maximize positive mother-infant interaction, while actively integrating the infant into the mother’s treatment. Breast pumping is treated as a necessary medical procedure and patients can easily access hospital-grade breast pumps with staff supervision. At one such perinatal psychiatric inpatient unit, high rates of treatment satisfaction and significant improvements in symptoms of depression, anxiety, active suicidal ideation, and overall functioning were observed at discharge.32 Therefore, it is crucial to incorporate strategies in general psychiatry units to improve perinatal care, acknowledging that most patients will not have access to these specialized units.21
A framework to approaching the relational ethics decisions
An interdisciplinary team used a relational ethics perspective to carefully analyze the risks and benefits of these complex cases. In Figure 1, we propose a framework for the relational ethics decisions of breastfeeding on general inpatient psychiatric units. In creating this framework, we considered principles of autonomy, beneficence, and nonmaleficence, along with the medical and logistical barriers to breastfeeding.
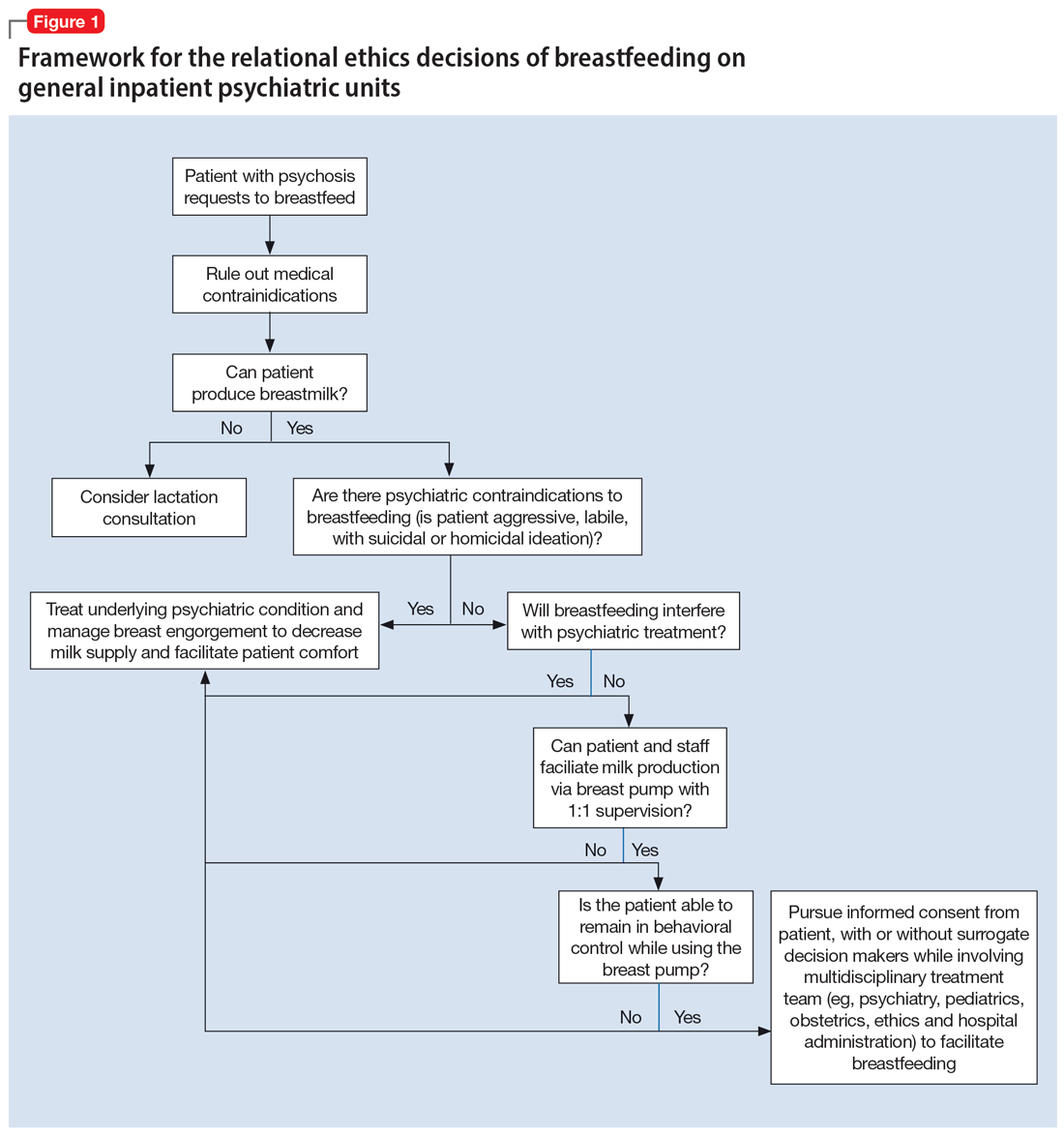
In Ms. C’s case, the team determined that the risks—which included disrupting the mother’s psychiatric treatment, exposing her to psychological harm due to increasing attachment before remanding the child to CPS custody, and risks to the child due to potential unpredictable agitation driven by the treatment-refractory psychosis of the mother as well as that of other psychiatric patients—outweighed the benefits of breastfeeding. We instead recommended breast pumping as an alternative once Ms. C’s psychiatric stability improved. We presented Ms. C with the option of breast pumping on postpartum Day 5. During a 1-day period in which she showed improved behavioral control, she was counseled on the risks and benefits of breastfeeding and exclusive pumping and was notified that the team would help her with the necessary resources, including consultation with a lactation specialist and breast pump. Despite lactation consultant support, Ms. C had low milk production and difficulty with hand expression, which was very discouraging to her. She produced 1 ounce of milk that was shared with the newborn while in the NICU. Because Ms. C’s psychiatric symptoms continued to be severe, with lability and aggression, and because pumping was triggering distress, the multidisciplinary team determined the best course of care would be to focus on her psychiatric recovery rather than on pumping breastmilk. To reduce milk production and minimize discomfort secondary to breast engorgement, the lactation consultant recommended cold compresses, pain management, and compression of breasts. Ultimately, the mother-infant dyad was unable to reap the benefits of breastfeeding (via pumping or direct breastfeeding) due to the mother’s underlying psychiatric illness, although the staffing, psychosocial support, and logistical limitations contributed to this outcome.33
In Ms. S’s case, the treatment team determined that there were no medical or psychiatric contraindications to breastfeeding, and she was counseled on the risks and benefits of direct breastfeeding and pumping. The treatment team determined it was safe for Ms. S to directly breastfeed as there were no concerns for infant harm postdelivery with constant supervision while on the obstetrics floor. The patient opted to directly breastfeed, which was successful with the guidance of a lactation specialist. When she was transferred to the psychiatric unit on postpartum Day 2, her child was discharged home with the husband. The patient was then encouraged to pump while the psychiatrists monitored her symptoms closely and facilitated increased staff and resources. Transportation of breastmilk was made possible by the family, and on postpartum Day 5, as the patient maintained psychiatric stability, the team discussed with Ms. S and her husband the prospect of direct breastfeeding. The treatment team arranged for separate visitation hours to minimize the possibility of exposing the infant to aggression from other patients on the unit and advocated with hospital leadership to approve of infant visitation on the unit.
Impact of involvement of Child Protective Services
The involvement of CPS also added complexity to Ms. C’s case. Without proper legal guidance, mothers with psychosis who lose custody can find it difficult to navigate the legal system and maintain contact with their children.34 As the prevalence of custody loss in mothers with psychosis is high (approximately 50% according to research published in the last 10 years), effective interventions to reunite the mother and child must be promoted (Figure 2).35-39 Ultimately, the goal of psychiatric hospitalization for perinatal women who have SMI is psychiatric stabilization. The preemptive involvement of psychiatry is crucial because it can allow for early postpartum planning and can provide an opportunity to address feeding options and custody concerns with the patient, social supports and services, and various medical teams. In Ms. C’s case, she visited her baby in the NICU on postpartum Day 2 without consultation with psychiatry or CPS, which posed risks to the patient, infant, and staff. It is vital that various clinicians collaborate with each other and the patient, working towards the goal of optimizing the patient’s mental health to allow for parenting rights in the future and maximizing a sustainable attachment between the parent and child. In Ms. S’s case, the husband was able to facilitate caring for the baby while the mother was hospitalized and played an integral role in the feeding process via pumped breastmilk and transport of the infant for direct breastfeeding.
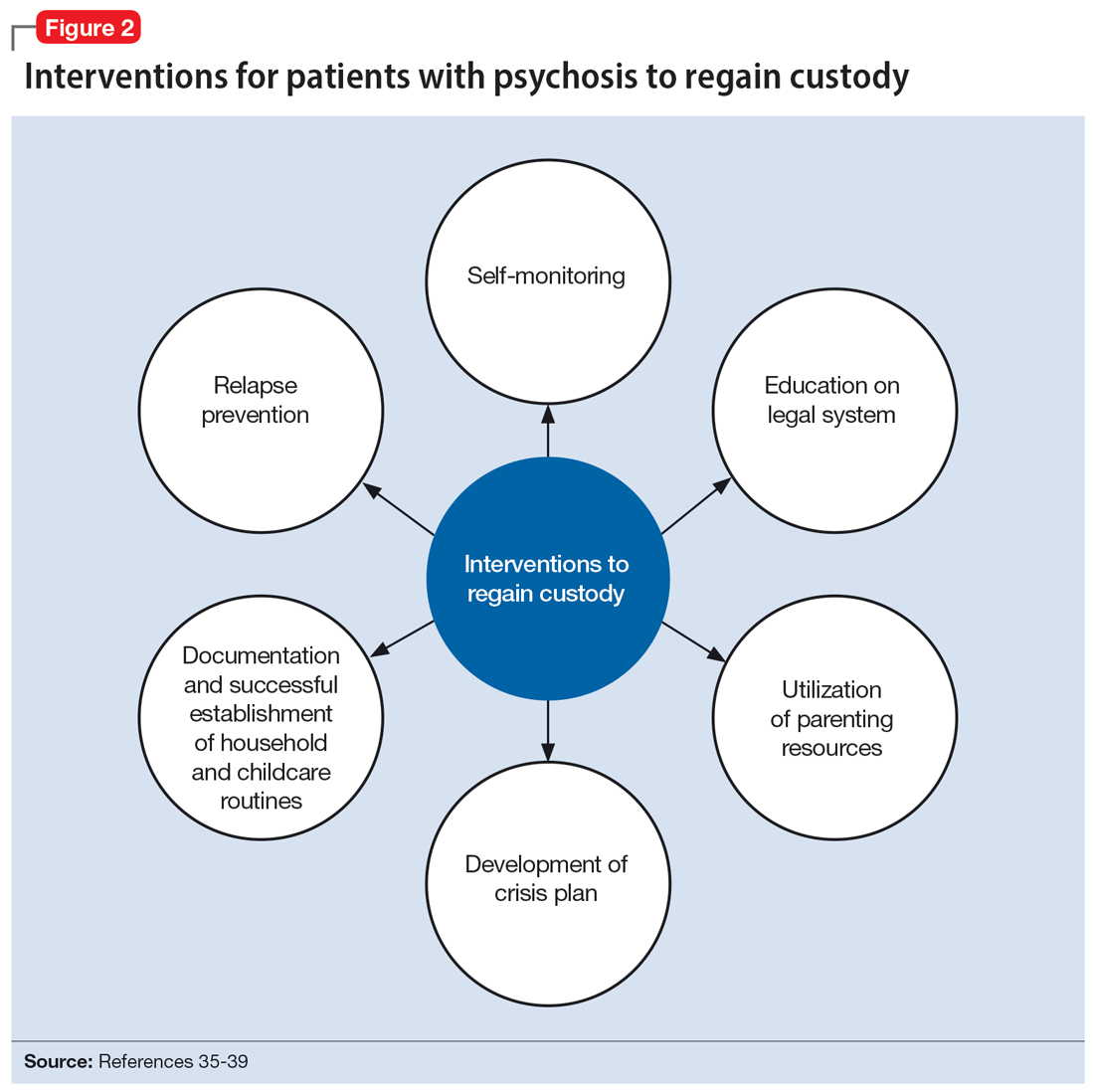
Continue to: The differences in these 2 cases...
The differences in these 2 cases show the extreme importance of social support to benefit both the mother and child, and the need for more comprehensive social services for women who do not have a social safety net.
Bottom Line
These complex cases highlight an ethical decision-making approach to breastfeeding in perinatal women who have serious mental illness. Collaborative care and shared decision-making, which highlight the interests of the mother and baby, are crucial when assessing the risks and benefits of breastfeeding and pumping breastmilk. Our relational ethics framework can be used to better evaluate and implement breastfeeding options on general psychiatric units.
Related Resources
- Tillman B, Sloan N, Westmoreland P. How COVID-19 affects peripartum women’s mental health. Current Psychiatry. 2021;20(6):18-22. doi:10.12788/cp.0129
- Koch J, Preinitz J. Antidepressants for patients who are breastfeeding: what to consider. Current Psychiatry. 2023;22(5):20-23,48. doi:10.12788/cp.0355
Drug Brand Names
Aripiprazole • Abilify
Olanzapine • Zyprexa
Risperidone • Risperdal
1. Brunner E, Falk DM, Jones M, et al. Olanzapine in pregnancy and breastfeeding: a review of data from global safety surveillance. BMC Pharmacol Toxicol. 2013;14:38. doi:10.1186/2050-6511-14-38
2. Seeman MV. Relational ethics: when mothers suffer from psychosis. Arch Womens Ment Health. 2004;7(3):201-210. doi:10.1007/s00737-004-0054-8
3. Motee A, Jeewon R. Importance of exclusive breastfeeding and complementary feeding among infants. Curr Res Nutr Food Sci. 2014;2(2). doi:10.12944/CRNFSJ.2.2.02
4. Committee Opinion No. 570: breastfeeding in underserved women: increasing initiation and continuation of breastfeeding. Obstet Gynecol. 2013;122(2 Pt 1):423-427. doi:10.1097/01.AOG.0000433008.93971.6a
5. Sibolboro Mezzacappa E, Endicott J. Parity mediates the association between infant feeding method and maternal depressive symptoms in the postpartum. Arch Womens Ment Health. 2007;10(6):259-266. doi:10.1007/s00737-007-0207-7
6. Kramer MS, Chalmers B, Hodnett ED, et al. Promotion of Breastfeeding Intervention Trial (PROBIT): a randomized trial in the Republic of Belarus. JAMA. 2001;285(4):413-420. doi:10.1001/jama.285.4.413
7. American Academy of Pediatrics. American Academy of Pediatrics calls for more support for breastfeeding mothers within updated policy recommendations. June 27, 2022. Accessed October 4, 2022. https://www.aap.org/en/news-room/news-releases/aap/2022/american-academy-of-pediatrics-calls-for-more-support-for-breastfeeding-mothers-within-updated-policy-recommendations
8. Hipwell AE, Kumar R. Maternal psychopathology and prediction of outcome based on mother-infant interaction ratings (BMIS). Br J Psychiatry. 1996;169(5):655-661. doi:10.1192/bjp.169.5.655
9. Chandra PS, Bhargavaraman RP, Raghunandan VN, et al. Delusions related to infant and their association with mother-infant interactions in postpartum psychotic disorders. Arch Womens Ment Health. 2006;9(5):285-288. doi:10.1007/s00737-006-0147-7
10. Klinger G, Stahl B, Fusar-Poli P, et al. Antipsychotic drugs and breastfeeding. Pediatr Endocrinol Rev. 2013;10(3):308-317.
11. Uguz F. A new safety scoring system for the use of psychotropic drugs during lactation. Am J Ther. 2021;28(1):e118-e126. doi:10.1097/MJT.0000000000000909
12. Hale TW, Krutsch K. Hale’s Medications & Mothers’ Milk, 2023: A Manual of Lactational Pharmacology. 20th ed. Springer Publishing Company; 2023.
13. Komaroff A. Aripiprazole and lactation failure: the importance of shared decision making. A case report. Case Rep Womens Health. 2021;30:e00308. doi:10.1016/j.crwh.2021.e00308
14. Dennis CL, McQueen K. Does maternal postpartum depressive symptomatology influence infant feeding outcomes? Acta Pediatr. 2007;96(4):590-594. doi:10.1111/j.1651-2227.2007.00184.x
15. Chaput KH, Nettel-Aguirre A, Musto R, et al. Breastfeeding difficulties and supports and risk of postpartum depression in a cohort of women who have given birth in Calgary: a prospective cohort study. CMAJ Open. 2016;4(1):E103-E109. doi:10.9778/cmajo.20150009
16. Dias CC, Figueiredo B. Breastfeeding and depression: a systematic review of the literature. J Affect Disord. 2015;171:142-154. doi:10.1016/j.jad.2014.09.022
17. Brown A, Rance J, Bennett P. Understanding the relationship between breastfeeding and postnatal depression: the role of pain and physical difficulties. J Adv Nurs. 2016;72(2):273-282. doi:10.1111/jan.12832
18. Rosenbaum KA. Exclusive breastmilk pumping: a concept analysis. Nurs Forum. 2022;57(5):946-953. doi:10.1111/nuf.12766
19. Boone KM, Geraghty SR, Keim SA. Feeding at the breast and expressed milk feeding: associations with otitis media and diarrhea in infants. J Pediatr. 2016;174:118-125. doi:10.1016/j.jpeds.2016.04.006
20. Epstein LJ, Kristo D, Strollo PJ Jr, et al; Adult Obstructive Sleep Apnea Task Force of the American Academy of Sleep Medicine. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med. 2009;5(3):263-276.
21. Caan MP, Sreshta NE, Okwerekwu JA, et al. Clinical and legal considerations regarding breastfeeding on psychiatric units. J Am Acad Psychiatry Law. 2022;50(2):200-207. doi:10.29158/JAAPL.210086-21
22. Glangeaud-Freudenthal NMC, Rainelli C, Cazas O, et al. Inpatient mother and baby psychiatric units (MBUs) and day cares. In: Sutter-Dallay AL, Glangeaud-Freudenthal NC, Guedeney A, et al, eds. Joint Care of Parents and Infants in Perinatal Psychiatry. Springer, Cham; 2016:147-164. doi:10.1007/978-3-319-21557-0_10
23. Dembosky A. A humane approach to caring for new mothers in psychiatric crisis. Health Aff (Millwood). 2021;40(10):1528-1533. doi:10.1377/hlthaff.2021.01288
24. Connellan K, Bartholomaeus C, Due C, et al. A systematic review of research on psychiatric mother-baby units. Arch Womens Ment Health. 2017;20(3):373-388. doi:10.1007/s00737-017-0718-9
25. Griffiths J, Lever Taylor B, Morant N, et al. A qualitative comparison of experiences of specialist mother and baby units versus general psychiatric wards. BMC Psychiatry. 2019;19(1):401. doi:10.1186/s12888-019-2389-8
26. Heron J, Gilbert N, Dolman C, et al. Information and support needs during recovery from postpartum psychosis. Arch Womens Ment Health. 2012;15(3):155-165. doi:10.1007/s00737-012-0267-1
27. Robertson E, Lyons A. Living with puerperal psychosis: a qualitative analysis. Psychol Psychother. 2003;76(Pt 4):411-431. doi:10.1348/147608303770584755
28. Mental Welfare Commission for Scotland. Perinatal Themed Visit Report: Keeping Mothers and Babies in Mind. Mental Welfare Commission for Scotland; 2016.
29. Wisner KL, Jennings KD, Conley B. Clinical dilemmas due to the lack of inpatient mother-baby units. Int J Psychiatry Med. 1996;26(4):479-493. doi:10.2190/NFJK-A4V7-CXUU-AM89
30. Battle CL, Howard MM. A mother-baby psychiatric day hospital: history, rationale, and why perinatal mental health is important for obstetric medicine. Obstet Med. 2014;7(2):66-70. doi:10.1177/1753495X13514402
31. Bullard ES, Meltzer-Brody S, Rubinow DR. The need for comprehensive psychiatric perinatal care-the University of North Carolina at Chapel Hill, Department of Psychiatry, Center for Women’s Mood Disorders launches the first dedicated inpatient program in the United States. Am J Obstet Gynecol. 2009;201(5):e10-e11. doi:10.1016/j.ajog.2009.05.004
32. Meltzer-Brody S, Brandon AR, Pearson B, et al. Evaluating the clinical effectiveness of a specialized perinatal psychiatry inpatient unit. Arch Womens Ment Health. 2014;17(2):107-113. doi:10.1007/s00737-013-0390-7
33. Alvarez-Toro V. Gender-specific care for women in psychiatric units. J Am Acad Psychiatry Law. 2022;JAAPL.220015-21. doi:10.29158/JAAPL.220015-21
34. Diaz-Caneja A, Johnson S. The views and experiences of severely mentally ill mothers--a qualitative study. Soc Psychiatry Psychiatr Epidemiol. 2004;39(6):472-482. doi:10.1007/s00127-004-0772-2
35. Gewurtz R, Krupa T, Eastabrook S, et al. Prevalence and characteristics of parenting among people served by assertive community treatment. Psychiatr Rehabil J. 2004;28(1):63-65. doi:10.2975/28.2004.63.65
36. Howard LM, Kumar R, Thornicroft G. Psychosocial characteristics and needs of mothers with psychotic disorders. Br J Psychiatry. 2001;178:427-432. doi:10.1192/bjp.178.5.427
37. Hollingsworth LD. Child custody loss among women with persistent severe mental illness. Social Work Research. 2004;28(4):199-209. doi:10.1093/swr/28.4.199
38. Dipple H, Smith S, Andrews H, et al. The experience of motherhood in women with severe and enduring mental illness. Soc Psychiatry Psychiatr Epidemiolf. 2002;37(7):336-340. doi:10.1007/s00127-002-0559-2
39. Seeman MV. Intervention to prevent child custody loss in mothers with schizophrenia. Schizophr Res Treatment. 2012;2012:796763. doi:10.1155/2012/796763
Difficult ethical situations can arise when treating perinatal women who have serious mental illness (SMI). Clinicians must consider ethical issues related to administering antipsychotic medications, the safety of breastfeeding, and concerns for child welfare. They need to carefully weigh the risks and benefits of each decision when treating perinatal women who have SMI. Ethical guidelines can help clinicians best support families in these situations.
In this article, we describe 2 cases of women with psychotic disorders who requested to breastfeed after delivering their child during an inpatient psychiatric hospitalization. The course of their hospitalizations illustrated common ethical questions and facilitated the creation of a framework to assist with complex decision-making regarding breastfeeding on inpatient psychiatric units.
CASE 1
Ms. C, age 41, is multigravida with a psychiatric history of chronic, severe schizoaffective disorder and lives in supportive housing. When Ms. C presents to the hospital in search of a rape kit, clinicians discover she is 22 weeks pregnant but has not received any prenatal care. Psychiatry is consulted because she is found to be intermittently agitated and endorses grandiose delusions. Ms. C requires involuntary hospitalization for decompensated psychosis because she refuses prenatal and psychiatric care. Because it has reassuring reproductive safety data,1 olanzapine 5 mg/d is started. However, Ms. C experiences minimal improvement from a maximum dose of 20 mg/d. After 13 weeks on the psychiatry unit, she is transferred to obstetrics service for preeclampsia with severe features. Ms. C requires an urgent cesarean delivery at 37 weeks. Her baby boy is transferred to the neonatal intensive care unit (NICU) for transient tachypnea. After delivery and in consultation with psychiatry, the pediatrics team calls Child Protective Services (CPS) due to concern for neglect driven by Ms. C’s psychiatric condition. Ms. C visits the child with medical unit staff supervision in the NICU without consulting with the psychiatry service or CPS. On postpartum Day 2, Ms. C is transferred back to psychiatry for persistent psychosis.
On postpartum Day 3, Ms. C starts to produce breastmilk and requests to breastfeed. At this time, the multidisciplinary team determines she is not able to visit her child in the NICU due to psychiatric instability. No plan is developed to facilitate hand expression or pumping of breastmilk while Ms. C is on the psychiatric unit. The clinical teams discuss whether the benefits of breastfeeding and/or pumping breastmilk would outweigh the risks. CPS determines that Ms. C is unable to retain custody and places the child in kinship foster care while awaiting clinical improvement from her.
CASE 2
Ms. S, age 32, has a history of schizophrenia. She lives with her husband and parents. She is pregnant for the first time and has been receiving consistent prenatal care. Ms. S is brought to the hospital by her husband for bizarre behavior and paranoia after self-discontinuing risperidone 2 mg twice daily due to concern about the medication’s influence on her pregnancy. An ultrasound confirms she is 37 weeks pregnant. Psychiatry is consulted because Ms. S is internally preoccupied, delusional, and endorses auditory hallucinations. She requires involuntary hospitalization for decompensated psychosis. During admission, Ms. S experiences improvement of her psychiatric symptoms while receiving risperidone 2 mg twice daily, which she takes consistently after receiving extensive psychoeducation regarding its safety profile during pregnancy and lactation.
After 2 weeks on the psychiatry unit, Ms. S’s care team transfers her to the obstetrics service with one-to-one supervision. At 39 weeks gestation, she has a vaginal delivery without complications. Because there are no concerns about infant harm, obstetrics, pediatrics, and psychiatry coordinate care so the baby can room in with Ms. S, her husband, and a staff supervisor to facilitate bonding. Ms. S starts to lactate, wishes to breastfeed, and meets with lactation, pediatric, obstetric, and psychiatric specialists to discuss the risks and benefits of breastfeeding and pumping breastmilk. She pursues direct breastfeeding until the baby is discharged home with the husband at postpartum Day 2. CPS is not called because there are no concerns for parental abuse or neglect at the infant’s discharge.
On postpartum Day 2, the obstetrics service transfers Ms. S back to the psychiatric unit for further treatment of her paranoia. She wishes to pump breastmilk while hospitalized, so the treatment team supplies a breast pump, facilitates the storage of breastmilk, and coordinates supervision during pumping to reduce the ligature risk. Ms. S’s husband visits daily to transport the milk and feed the infant breastmilk and formula to meet its nutritional needs. Ms. S maintains psychiatric stability while breast pumping, and the team helps transition her to breastfeeding during visitation with her husband and infant until she is discharged home at 2 weeks postpartum.
Continue to: Approaching care with a relational ethics framework
Approaching care with a relational ethics framework
A relational ethics framework was constructed to evaluate whether to support breastfeeding for both patients during their psychiatric hospitalizations. A relational ethics perspective is defined as “a moral responsibility within a context of human relations” [that] “recognizes the human interdependency and reciprocity within which personal autonomy is embedded.”2 This framework values connectedness and commonality between various and even conflicting parties. In the setting of a clinician-patient relationship, health care decisions are made with consideration of the patient’s traditional beliefs, values, and principles rather than the application of impartial moral principles. For these complex cases, this framework was chosen to determine the safest possible outcome for both mother and child.
Risks/benefits of breastfeeding by patients who have SMI
There are several methods of breastfeeding, including direct breastfeeding and other ways of expressing breastmilk such as pumping or hand expression.3 Unlike other forms of feeding using breastmilk, direct breastfeeding has been extensively studied, has well-established medical and psychological benefits for newborns and mothers, and enhances long-term bonding.4 Compared with their counterparts who do not breastfeed, mothers who breastfeed have lower rates of unintended pregnancy, cardiovascular disease, postpartum bleeding, osteoporosis, and breast and ovarian cancer.5 Among its key psychological benefits, breastfeeding is associated with an increase in maternal self-efficacy and, in some research, has been shown to be associated with a decreased risk of postpartum depression and stress.Additionally, breastfed infants experience lower rates of childhood infection and obesity, and improved nutrition, cognitive development, and immune function.6 The American Academy of Pediatrics recognizes these benefits and recommends that women exclusively breastfeed for 6 months postpartum and continue to breastfeed for 2 years or beyond if mutually desired by the mother and child.7 Absolute contraindications to breastfeeding must be ruled out (eg, infant classic galactosemia; maternal use of illicit substances such as cocaine, opioids, or phencyclidine; maternal HIV infection, etc).
The risks of breastfeeding by patients who have SMI must also be considered. In severe situations, the infant can be exposed to a mother’s agitation secondary to psychosis.8,9 The transmission of antipsychotic medication through breastmilk and associated adverse effects (eg, sedation, poor feeding, and extrapyramidal symptoms) are also potential risks and varies among different antipsychotic medications.1,10 Therefore, when prescribing an antipsychotic for a patient with SMI who breastfeeds, it is crucial to consider the medication’s safety profile as well as other factors, such as the relative infant dose (the weight-adjusted [ie, mg/kg] percentage of the maternal dosage ingested by a fully breastfed infant) and the molecular characteristics of the medication.10-12 Neonates should be routinely monitored for adverse effects, medication toxicity, and withdrawal symptoms, and care should be coordinated with the infant’s pediatrician. Certain antipsychotic medications, such as aripiprazole, may impact breastmilk production through the dopamine agonist’s interference of the prolactin reflex and anticholinergic properties.11,13 For a patient with SMI, perhaps the most significant risk involves the time and resources needed for breastfeeding, which can interfere with sleep and psychiatric treatment and possibly further exacerbate psychiatric symptoms.14-16 Additionally, breastfeeding difficulties or disruption can increase the risk of psychiatric symptoms and psychological distress.17 In Ms. C’s case, there was a delay in the baby latching as well as multiple medical and psychiatric factors that hindered the milk-ejection reflex to properly initiate; both of these factors rendered breastfeeding particularly difficult while Ms. C was on the inpatient psychiatry unit.17 In comparison, Ms. S was able to bond with her infant shortly after delivery, which facilitated the milk-ejection reflex and lactation.
Patients who wish to directly breastfeed but struggle to do so while tending to their acute psychiatric condition can benefit from expression of breastmilk that can be provided to the infant or discarded to facilitate breastfeeding in the future.18 While expression of breastmilk may not be as advantageous for infant health as direct breastfeeding due to the potential changes in breastmilk composition from collecting, storing, and heating, this option can be more protective than formula feeding and facilitate future breastfeeding.19 In these clinical scenarios, it is standard care to provide a hospital-grade breast pump to the patient, much like a continuous positive airway pressure machine is provided to patients with obstructive sleep apnea.20 However, there is often considerable difficulty obtaining proper breastfeeding equipment and a lack of services devoted to perinatal care in general inpatient settings. Barriers to direct breastfeeding and pumping of breastmilk are highlighted in the Table.21

Limitations on breastfeeding on an inpatient unit
The limitations in care and restrictions placed on breastfeeding are more optimally addressed in a mother and baby unit (MBU). MBUs are specialized inpatient psychiatric units designed for mothers experiencing severe perinatal psychiatric difficulties. Unlike general psychiatric units, MBUs allow for joint, full-time admission of mothers and their infants. These units also include multidisciplinary staff who specialize in treating perinatal mental health issues as well as infant care and child development.22 Admission into an MBU is considered best practice for new mothers requiring treatment, particularly in the United Kingdom, Australia, and France, as it is well-recognized that the separation of mother and baby can be psychologically harmful.23 In the UK, most patients admitted to an MBU showed significant improvement of their psychiatric symptoms and reported overall high satisfaction with care.24,25 Patients who experience postpartum psychosis prefer MBUs over general psychiatric units because the latter often lack specialized perinatal support, appropriate visitor arrangements, and adequate time with their infant.26-28
Continue to: The resistance to adopting MBUs in the United States...
The resistance to adopting MBUs in the United States has posed significant barriers in care for perinatal patients and has been attributed to financial barriers, medicolegal risk, staffing, and safety concerns.29 Though currently there are no MBUs in the US, other specialized units have been created. A partial day hospitalization program created in 2000 in Rhode Island for mothers and infants revolutionized the psychiatric care experience for new mothers.30 Since then, other institutions have significantly expanded their services to include perinatal psychiatry inpatient units, yet unlike MBUs, these units typically do not provide overnight rooming-in with infants.31 They have the necessary resources and facilities to accommodate the mother’s needs and maximize positive mother-infant interaction, while actively integrating the infant into the mother’s treatment. Breast pumping is treated as a necessary medical procedure and patients can easily access hospital-grade breast pumps with staff supervision. At one such perinatal psychiatric inpatient unit, high rates of treatment satisfaction and significant improvements in symptoms of depression, anxiety, active suicidal ideation, and overall functioning were observed at discharge.32 Therefore, it is crucial to incorporate strategies in general psychiatry units to improve perinatal care, acknowledging that most patients will not have access to these specialized units.21
A framework to approaching the relational ethics decisions
An interdisciplinary team used a relational ethics perspective to carefully analyze the risks and benefits of these complex cases. In Figure 1, we propose a framework for the relational ethics decisions of breastfeeding on general inpatient psychiatric units. In creating this framework, we considered principles of autonomy, beneficence, and nonmaleficence, along with the medical and logistical barriers to breastfeeding.

In Ms. C’s case, the team determined that the risks—which included disrupting the mother’s psychiatric treatment, exposing her to psychological harm due to increasing attachment before remanding the child to CPS custody, and risks to the child due to potential unpredictable agitation driven by the treatment-refractory psychosis of the mother as well as that of other psychiatric patients—outweighed the benefits of breastfeeding. We instead recommended breast pumping as an alternative once Ms. C’s psychiatric stability improved. We presented Ms. C with the option of breast pumping on postpartum Day 5. During a 1-day period in which she showed improved behavioral control, she was counseled on the risks and benefits of breastfeeding and exclusive pumping and was notified that the team would help her with the necessary resources, including consultation with a lactation specialist and breast pump. Despite lactation consultant support, Ms. C had low milk production and difficulty with hand expression, which was very discouraging to her. She produced 1 ounce of milk that was shared with the newborn while in the NICU. Because Ms. C’s psychiatric symptoms continued to be severe, with lability and aggression, and because pumping was triggering distress, the multidisciplinary team determined the best course of care would be to focus on her psychiatric recovery rather than on pumping breastmilk. To reduce milk production and minimize discomfort secondary to breast engorgement, the lactation consultant recommended cold compresses, pain management, and compression of breasts. Ultimately, the mother-infant dyad was unable to reap the benefits of breastfeeding (via pumping or direct breastfeeding) due to the mother’s underlying psychiatric illness, although the staffing, psychosocial support, and logistical limitations contributed to this outcome.33
In Ms. S’s case, the treatment team determined that there were no medical or psychiatric contraindications to breastfeeding, and she was counseled on the risks and benefits of direct breastfeeding and pumping. The treatment team determined it was safe for Ms. S to directly breastfeed as there were no concerns for infant harm postdelivery with constant supervision while on the obstetrics floor. The patient opted to directly breastfeed, which was successful with the guidance of a lactation specialist. When she was transferred to the psychiatric unit on postpartum Day 2, her child was discharged home with the husband. The patient was then encouraged to pump while the psychiatrists monitored her symptoms closely and facilitated increased staff and resources. Transportation of breastmilk was made possible by the family, and on postpartum Day 5, as the patient maintained psychiatric stability, the team discussed with Ms. S and her husband the prospect of direct breastfeeding. The treatment team arranged for separate visitation hours to minimize the possibility of exposing the infant to aggression from other patients on the unit and advocated with hospital leadership to approve of infant visitation on the unit.
Impact of involvement of Child Protective Services
The involvement of CPS also added complexity to Ms. C’s case. Without proper legal guidance, mothers with psychosis who lose custody can find it difficult to navigate the legal system and maintain contact with their children.34 As the prevalence of custody loss in mothers with psychosis is high (approximately 50% according to research published in the last 10 years), effective interventions to reunite the mother and child must be promoted (Figure 2).35-39 Ultimately, the goal of psychiatric hospitalization for perinatal women who have SMI is psychiatric stabilization. The preemptive involvement of psychiatry is crucial because it can allow for early postpartum planning and can provide an opportunity to address feeding options and custody concerns with the patient, social supports and services, and various medical teams. In Ms. C’s case, she visited her baby in the NICU on postpartum Day 2 without consultation with psychiatry or CPS, which posed risks to the patient, infant, and staff. It is vital that various clinicians collaborate with each other and the patient, working towards the goal of optimizing the patient’s mental health to allow for parenting rights in the future and maximizing a sustainable attachment between the parent and child. In Ms. S’s case, the husband was able to facilitate caring for the baby while the mother was hospitalized and played an integral role in the feeding process via pumped breastmilk and transport of the infant for direct breastfeeding.

Continue to: The differences in these 2 cases...
The differences in these 2 cases show the extreme importance of social support to benefit both the mother and child, and the need for more comprehensive social services for women who do not have a social safety net.
Bottom Line
These complex cases highlight an ethical decision-making approach to breastfeeding in perinatal women who have serious mental illness. Collaborative care and shared decision-making, which highlight the interests of the mother and baby, are crucial when assessing the risks and benefits of breastfeeding and pumping breastmilk. Our relational ethics framework can be used to better evaluate and implement breastfeeding options on general psychiatric units.
Related Resources
- Tillman B, Sloan N, Westmoreland P. How COVID-19 affects peripartum women’s mental health. Current Psychiatry. 2021;20(6):18-22. doi:10.12788/cp.0129
- Koch J, Preinitz J. Antidepressants for patients who are breastfeeding: what to consider. Current Psychiatry. 2023;22(5):20-23,48. doi:10.12788/cp.0355
Drug Brand Names
Aripiprazole • Abilify
Olanzapine • Zyprexa
Risperidone • Risperdal
Difficult ethical situations can arise when treating perinatal women who have serious mental illness (SMI). Clinicians must consider ethical issues related to administering antipsychotic medications, the safety of breastfeeding, and concerns for child welfare. They need to carefully weigh the risks and benefits of each decision when treating perinatal women who have SMI. Ethical guidelines can help clinicians best support families in these situations.
In this article, we describe 2 cases of women with psychotic disorders who requested to breastfeed after delivering their child during an inpatient psychiatric hospitalization. The course of their hospitalizations illustrated common ethical questions and facilitated the creation of a framework to assist with complex decision-making regarding breastfeeding on inpatient psychiatric units.
CASE 1
Ms. C, age 41, is multigravida with a psychiatric history of chronic, severe schizoaffective disorder and lives in supportive housing. When Ms. C presents to the hospital in search of a rape kit, clinicians discover she is 22 weeks pregnant but has not received any prenatal care. Psychiatry is consulted because she is found to be intermittently agitated and endorses grandiose delusions. Ms. C requires involuntary hospitalization for decompensated psychosis because she refuses prenatal and psychiatric care. Because it has reassuring reproductive safety data,1 olanzapine 5 mg/d is started. However, Ms. C experiences minimal improvement from a maximum dose of 20 mg/d. After 13 weeks on the psychiatry unit, she is transferred to obstetrics service for preeclampsia with severe features. Ms. C requires an urgent cesarean delivery at 37 weeks. Her baby boy is transferred to the neonatal intensive care unit (NICU) for transient tachypnea. After delivery and in consultation with psychiatry, the pediatrics team calls Child Protective Services (CPS) due to concern for neglect driven by Ms. C’s psychiatric condition. Ms. C visits the child with medical unit staff supervision in the NICU without consulting with the psychiatry service or CPS. On postpartum Day 2, Ms. C is transferred back to psychiatry for persistent psychosis.
On postpartum Day 3, Ms. C starts to produce breastmilk and requests to breastfeed. At this time, the multidisciplinary team determines she is not able to visit her child in the NICU due to psychiatric instability. No plan is developed to facilitate hand expression or pumping of breastmilk while Ms. C is on the psychiatric unit. The clinical teams discuss whether the benefits of breastfeeding and/or pumping breastmilk would outweigh the risks. CPS determines that Ms. C is unable to retain custody and places the child in kinship foster care while awaiting clinical improvement from her.
CASE 2
Ms. S, age 32, has a history of schizophrenia. She lives with her husband and parents. She is pregnant for the first time and has been receiving consistent prenatal care. Ms. S is brought to the hospital by her husband for bizarre behavior and paranoia after self-discontinuing risperidone 2 mg twice daily due to concern about the medication’s influence on her pregnancy. An ultrasound confirms she is 37 weeks pregnant. Psychiatry is consulted because Ms. S is internally preoccupied, delusional, and endorses auditory hallucinations. She requires involuntary hospitalization for decompensated psychosis. During admission, Ms. S experiences improvement of her psychiatric symptoms while receiving risperidone 2 mg twice daily, which she takes consistently after receiving extensive psychoeducation regarding its safety profile during pregnancy and lactation.
After 2 weeks on the psychiatry unit, Ms. S’s care team transfers her to the obstetrics service with one-to-one supervision. At 39 weeks gestation, she has a vaginal delivery without complications. Because there are no concerns about infant harm, obstetrics, pediatrics, and psychiatry coordinate care so the baby can room in with Ms. S, her husband, and a staff supervisor to facilitate bonding. Ms. S starts to lactate, wishes to breastfeed, and meets with lactation, pediatric, obstetric, and psychiatric specialists to discuss the risks and benefits of breastfeeding and pumping breastmilk. She pursues direct breastfeeding until the baby is discharged home with the husband at postpartum Day 2. CPS is not called because there are no concerns for parental abuse or neglect at the infant’s discharge.
On postpartum Day 2, the obstetrics service transfers Ms. S back to the psychiatric unit for further treatment of her paranoia. She wishes to pump breastmilk while hospitalized, so the treatment team supplies a breast pump, facilitates the storage of breastmilk, and coordinates supervision during pumping to reduce the ligature risk. Ms. S’s husband visits daily to transport the milk and feed the infant breastmilk and formula to meet its nutritional needs. Ms. S maintains psychiatric stability while breast pumping, and the team helps transition her to breastfeeding during visitation with her husband and infant until she is discharged home at 2 weeks postpartum.
Continue to: Approaching care with a relational ethics framework
Approaching care with a relational ethics framework
A relational ethics framework was constructed to evaluate whether to support breastfeeding for both patients during their psychiatric hospitalizations. A relational ethics perspective is defined as “a moral responsibility within a context of human relations” [that] “recognizes the human interdependency and reciprocity within which personal autonomy is embedded.”2 This framework values connectedness and commonality between various and even conflicting parties. In the setting of a clinician-patient relationship, health care decisions are made with consideration of the patient’s traditional beliefs, values, and principles rather than the application of impartial moral principles. For these complex cases, this framework was chosen to determine the safest possible outcome for both mother and child.
Risks/benefits of breastfeeding by patients who have SMI
There are several methods of breastfeeding, including direct breastfeeding and other ways of expressing breastmilk such as pumping or hand expression.3 Unlike other forms of feeding using breastmilk, direct breastfeeding has been extensively studied, has well-established medical and psychological benefits for newborns and mothers, and enhances long-term bonding.4 Compared with their counterparts who do not breastfeed, mothers who breastfeed have lower rates of unintended pregnancy, cardiovascular disease, postpartum bleeding, osteoporosis, and breast and ovarian cancer.5 Among its key psychological benefits, breastfeeding is associated with an increase in maternal self-efficacy and, in some research, has been shown to be associated with a decreased risk of postpartum depression and stress.Additionally, breastfed infants experience lower rates of childhood infection and obesity, and improved nutrition, cognitive development, and immune function.6 The American Academy of Pediatrics recognizes these benefits and recommends that women exclusively breastfeed for 6 months postpartum and continue to breastfeed for 2 years or beyond if mutually desired by the mother and child.7 Absolute contraindications to breastfeeding must be ruled out (eg, infant classic galactosemia; maternal use of illicit substances such as cocaine, opioids, or phencyclidine; maternal HIV infection, etc).
The risks of breastfeeding by patients who have SMI must also be considered. In severe situations, the infant can be exposed to a mother’s agitation secondary to psychosis.8,9 The transmission of antipsychotic medication through breastmilk and associated adverse effects (eg, sedation, poor feeding, and extrapyramidal symptoms) are also potential risks and varies among different antipsychotic medications.1,10 Therefore, when prescribing an antipsychotic for a patient with SMI who breastfeeds, it is crucial to consider the medication’s safety profile as well as other factors, such as the relative infant dose (the weight-adjusted [ie, mg/kg] percentage of the maternal dosage ingested by a fully breastfed infant) and the molecular characteristics of the medication.10-12 Neonates should be routinely monitored for adverse effects, medication toxicity, and withdrawal symptoms, and care should be coordinated with the infant’s pediatrician. Certain antipsychotic medications, such as aripiprazole, may impact breastmilk production through the dopamine agonist’s interference of the prolactin reflex and anticholinergic properties.11,13 For a patient with SMI, perhaps the most significant risk involves the time and resources needed for breastfeeding, which can interfere with sleep and psychiatric treatment and possibly further exacerbate psychiatric symptoms.14-16 Additionally, breastfeeding difficulties or disruption can increase the risk of psychiatric symptoms and psychological distress.17 In Ms. C’s case, there was a delay in the baby latching as well as multiple medical and psychiatric factors that hindered the milk-ejection reflex to properly initiate; both of these factors rendered breastfeeding particularly difficult while Ms. C was on the inpatient psychiatry unit.17 In comparison, Ms. S was able to bond with her infant shortly after delivery, which facilitated the milk-ejection reflex and lactation.
Patients who wish to directly breastfeed but struggle to do so while tending to their acute psychiatric condition can benefit from expression of breastmilk that can be provided to the infant or discarded to facilitate breastfeeding in the future.18 While expression of breastmilk may not be as advantageous for infant health as direct breastfeeding due to the potential changes in breastmilk composition from collecting, storing, and heating, this option can be more protective than formula feeding and facilitate future breastfeeding.19 In these clinical scenarios, it is standard care to provide a hospital-grade breast pump to the patient, much like a continuous positive airway pressure machine is provided to patients with obstructive sleep apnea.20 However, there is often considerable difficulty obtaining proper breastfeeding equipment and a lack of services devoted to perinatal care in general inpatient settings. Barriers to direct breastfeeding and pumping of breastmilk are highlighted in the Table.21

Limitations on breastfeeding on an inpatient unit
The limitations in care and restrictions placed on breastfeeding are more optimally addressed in a mother and baby unit (MBU). MBUs are specialized inpatient psychiatric units designed for mothers experiencing severe perinatal psychiatric difficulties. Unlike general psychiatric units, MBUs allow for joint, full-time admission of mothers and their infants. These units also include multidisciplinary staff who specialize in treating perinatal mental health issues as well as infant care and child development.22 Admission into an MBU is considered best practice for new mothers requiring treatment, particularly in the United Kingdom, Australia, and France, as it is well-recognized that the separation of mother and baby can be psychologically harmful.23 In the UK, most patients admitted to an MBU showed significant improvement of their psychiatric symptoms and reported overall high satisfaction with care.24,25 Patients who experience postpartum psychosis prefer MBUs over general psychiatric units because the latter often lack specialized perinatal support, appropriate visitor arrangements, and adequate time with their infant.26-28
Continue to: The resistance to adopting MBUs in the United States...
The resistance to adopting MBUs in the United States has posed significant barriers in care for perinatal patients and has been attributed to financial barriers, medicolegal risk, staffing, and safety concerns.29 Though currently there are no MBUs in the US, other specialized units have been created. A partial day hospitalization program created in 2000 in Rhode Island for mothers and infants revolutionized the psychiatric care experience for new mothers.30 Since then, other institutions have significantly expanded their services to include perinatal psychiatry inpatient units, yet unlike MBUs, these units typically do not provide overnight rooming-in with infants.31 They have the necessary resources and facilities to accommodate the mother’s needs and maximize positive mother-infant interaction, while actively integrating the infant into the mother’s treatment. Breast pumping is treated as a necessary medical procedure and patients can easily access hospital-grade breast pumps with staff supervision. At one such perinatal psychiatric inpatient unit, high rates of treatment satisfaction and significant improvements in symptoms of depression, anxiety, active suicidal ideation, and overall functioning were observed at discharge.32 Therefore, it is crucial to incorporate strategies in general psychiatry units to improve perinatal care, acknowledging that most patients will not have access to these specialized units.21
A framework to approaching the relational ethics decisions
An interdisciplinary team used a relational ethics perspective to carefully analyze the risks and benefits of these complex cases. In Figure 1, we propose a framework for the relational ethics decisions of breastfeeding on general inpatient psychiatric units. In creating this framework, we considered principles of autonomy, beneficence, and nonmaleficence, along with the medical and logistical barriers to breastfeeding.

In Ms. C’s case, the team determined that the risks—which included disrupting the mother’s psychiatric treatment, exposing her to psychological harm due to increasing attachment before remanding the child to CPS custody, and risks to the child due to potential unpredictable agitation driven by the treatment-refractory psychosis of the mother as well as that of other psychiatric patients—outweighed the benefits of breastfeeding. We instead recommended breast pumping as an alternative once Ms. C’s psychiatric stability improved. We presented Ms. C with the option of breast pumping on postpartum Day 5. During a 1-day period in which she showed improved behavioral control, she was counseled on the risks and benefits of breastfeeding and exclusive pumping and was notified that the team would help her with the necessary resources, including consultation with a lactation specialist and breast pump. Despite lactation consultant support, Ms. C had low milk production and difficulty with hand expression, which was very discouraging to her. She produced 1 ounce of milk that was shared with the newborn while in the NICU. Because Ms. C’s psychiatric symptoms continued to be severe, with lability and aggression, and because pumping was triggering distress, the multidisciplinary team determined the best course of care would be to focus on her psychiatric recovery rather than on pumping breastmilk. To reduce milk production and minimize discomfort secondary to breast engorgement, the lactation consultant recommended cold compresses, pain management, and compression of breasts. Ultimately, the mother-infant dyad was unable to reap the benefits of breastfeeding (via pumping or direct breastfeeding) due to the mother’s underlying psychiatric illness, although the staffing, psychosocial support, and logistical limitations contributed to this outcome.33
In Ms. S’s case, the treatment team determined that there were no medical or psychiatric contraindications to breastfeeding, and she was counseled on the risks and benefits of direct breastfeeding and pumping. The treatment team determined it was safe for Ms. S to directly breastfeed as there were no concerns for infant harm postdelivery with constant supervision while on the obstetrics floor. The patient opted to directly breastfeed, which was successful with the guidance of a lactation specialist. When she was transferred to the psychiatric unit on postpartum Day 2, her child was discharged home with the husband. The patient was then encouraged to pump while the psychiatrists monitored her symptoms closely and facilitated increased staff and resources. Transportation of breastmilk was made possible by the family, and on postpartum Day 5, as the patient maintained psychiatric stability, the team discussed with Ms. S and her husband the prospect of direct breastfeeding. The treatment team arranged for separate visitation hours to minimize the possibility of exposing the infant to aggression from other patients on the unit and advocated with hospital leadership to approve of infant visitation on the unit.
Impact of involvement of Child Protective Services
The involvement of CPS also added complexity to Ms. C’s case. Without proper legal guidance, mothers with psychosis who lose custody can find it difficult to navigate the legal system and maintain contact with their children.34 As the prevalence of custody loss in mothers with psychosis is high (approximately 50% according to research published in the last 10 years), effective interventions to reunite the mother and child must be promoted (Figure 2).35-39 Ultimately, the goal of psychiatric hospitalization for perinatal women who have SMI is psychiatric stabilization. The preemptive involvement of psychiatry is crucial because it can allow for early postpartum planning and can provide an opportunity to address feeding options and custody concerns with the patient, social supports and services, and various medical teams. In Ms. C’s case, she visited her baby in the NICU on postpartum Day 2 without consultation with psychiatry or CPS, which posed risks to the patient, infant, and staff. It is vital that various clinicians collaborate with each other and the patient, working towards the goal of optimizing the patient’s mental health to allow for parenting rights in the future and maximizing a sustainable attachment between the parent and child. In Ms. S’s case, the husband was able to facilitate caring for the baby while the mother was hospitalized and played an integral role in the feeding process via pumped breastmilk and transport of the infant for direct breastfeeding.

Continue to: The differences in these 2 cases...
The differences in these 2 cases show the extreme importance of social support to benefit both the mother and child, and the need for more comprehensive social services for women who do not have a social safety net.
Bottom Line
These complex cases highlight an ethical decision-making approach to breastfeeding in perinatal women who have serious mental illness. Collaborative care and shared decision-making, which highlight the interests of the mother and baby, are crucial when assessing the risks and benefits of breastfeeding and pumping breastmilk. Our relational ethics framework can be used to better evaluate and implement breastfeeding options on general psychiatric units.
Related Resources
- Tillman B, Sloan N, Westmoreland P. How COVID-19 affects peripartum women’s mental health. Current Psychiatry. 2021;20(6):18-22. doi:10.12788/cp.0129
- Koch J, Preinitz J. Antidepressants for patients who are breastfeeding: what to consider. Current Psychiatry. 2023;22(5):20-23,48. doi:10.12788/cp.0355
Drug Brand Names
Aripiprazole • Abilify
Olanzapine • Zyprexa
Risperidone • Risperdal
1. Brunner E, Falk DM, Jones M, et al. Olanzapine in pregnancy and breastfeeding: a review of data from global safety surveillance. BMC Pharmacol Toxicol. 2013;14:38. doi:10.1186/2050-6511-14-38
2. Seeman MV. Relational ethics: when mothers suffer from psychosis. Arch Womens Ment Health. 2004;7(3):201-210. doi:10.1007/s00737-004-0054-8
3. Motee A, Jeewon R. Importance of exclusive breastfeeding and complementary feeding among infants. Curr Res Nutr Food Sci. 2014;2(2). doi:10.12944/CRNFSJ.2.2.02
4. Committee Opinion No. 570: breastfeeding in underserved women: increasing initiation and continuation of breastfeeding. Obstet Gynecol. 2013;122(2 Pt 1):423-427. doi:10.1097/01.AOG.0000433008.93971.6a
5. Sibolboro Mezzacappa E, Endicott J. Parity mediates the association between infant feeding method and maternal depressive symptoms in the postpartum. Arch Womens Ment Health. 2007;10(6):259-266. doi:10.1007/s00737-007-0207-7
6. Kramer MS, Chalmers B, Hodnett ED, et al. Promotion of Breastfeeding Intervention Trial (PROBIT): a randomized trial in the Republic of Belarus. JAMA. 2001;285(4):413-420. doi:10.1001/jama.285.4.413
7. American Academy of Pediatrics. American Academy of Pediatrics calls for more support for breastfeeding mothers within updated policy recommendations. June 27, 2022. Accessed October 4, 2022. https://www.aap.org/en/news-room/news-releases/aap/2022/american-academy-of-pediatrics-calls-for-more-support-for-breastfeeding-mothers-within-updated-policy-recommendations
8. Hipwell AE, Kumar R. Maternal psychopathology and prediction of outcome based on mother-infant interaction ratings (BMIS). Br J Psychiatry. 1996;169(5):655-661. doi:10.1192/bjp.169.5.655
9. Chandra PS, Bhargavaraman RP, Raghunandan VN, et al. Delusions related to infant and their association with mother-infant interactions in postpartum psychotic disorders. Arch Womens Ment Health. 2006;9(5):285-288. doi:10.1007/s00737-006-0147-7
10. Klinger G, Stahl B, Fusar-Poli P, et al. Antipsychotic drugs and breastfeeding. Pediatr Endocrinol Rev. 2013;10(3):308-317.
11. Uguz F. A new safety scoring system for the use of psychotropic drugs during lactation. Am J Ther. 2021;28(1):e118-e126. doi:10.1097/MJT.0000000000000909
12. Hale TW, Krutsch K. Hale’s Medications & Mothers’ Milk, 2023: A Manual of Lactational Pharmacology. 20th ed. Springer Publishing Company; 2023.
13. Komaroff A. Aripiprazole and lactation failure: the importance of shared decision making. A case report. Case Rep Womens Health. 2021;30:e00308. doi:10.1016/j.crwh.2021.e00308
14. Dennis CL, McQueen K. Does maternal postpartum depressive symptomatology influence infant feeding outcomes? Acta Pediatr. 2007;96(4):590-594. doi:10.1111/j.1651-2227.2007.00184.x
15. Chaput KH, Nettel-Aguirre A, Musto R, et al. Breastfeeding difficulties and supports and risk of postpartum depression in a cohort of women who have given birth in Calgary: a prospective cohort study. CMAJ Open. 2016;4(1):E103-E109. doi:10.9778/cmajo.20150009
16. Dias CC, Figueiredo B. Breastfeeding and depression: a systematic review of the literature. J Affect Disord. 2015;171:142-154. doi:10.1016/j.jad.2014.09.022
17. Brown A, Rance J, Bennett P. Understanding the relationship between breastfeeding and postnatal depression: the role of pain and physical difficulties. J Adv Nurs. 2016;72(2):273-282. doi:10.1111/jan.12832
18. Rosenbaum KA. Exclusive breastmilk pumping: a concept analysis. Nurs Forum. 2022;57(5):946-953. doi:10.1111/nuf.12766
19. Boone KM, Geraghty SR, Keim SA. Feeding at the breast and expressed milk feeding: associations with otitis media and diarrhea in infants. J Pediatr. 2016;174:118-125. doi:10.1016/j.jpeds.2016.04.006
20. Epstein LJ, Kristo D, Strollo PJ Jr, et al; Adult Obstructive Sleep Apnea Task Force of the American Academy of Sleep Medicine. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med. 2009;5(3):263-276.
21. Caan MP, Sreshta NE, Okwerekwu JA, et al. Clinical and legal considerations regarding breastfeeding on psychiatric units. J Am Acad Psychiatry Law. 2022;50(2):200-207. doi:10.29158/JAAPL.210086-21
22. Glangeaud-Freudenthal NMC, Rainelli C, Cazas O, et al. Inpatient mother and baby psychiatric units (MBUs) and day cares. In: Sutter-Dallay AL, Glangeaud-Freudenthal NC, Guedeney A, et al, eds. Joint Care of Parents and Infants in Perinatal Psychiatry. Springer, Cham; 2016:147-164. doi:10.1007/978-3-319-21557-0_10
23. Dembosky A. A humane approach to caring for new mothers in psychiatric crisis. Health Aff (Millwood). 2021;40(10):1528-1533. doi:10.1377/hlthaff.2021.01288
24. Connellan K, Bartholomaeus C, Due C, et al. A systematic review of research on psychiatric mother-baby units. Arch Womens Ment Health. 2017;20(3):373-388. doi:10.1007/s00737-017-0718-9
25. Griffiths J, Lever Taylor B, Morant N, et al. A qualitative comparison of experiences of specialist mother and baby units versus general psychiatric wards. BMC Psychiatry. 2019;19(1):401. doi:10.1186/s12888-019-2389-8
26. Heron J, Gilbert N, Dolman C, et al. Information and support needs during recovery from postpartum psychosis. Arch Womens Ment Health. 2012;15(3):155-165. doi:10.1007/s00737-012-0267-1
27. Robertson E, Lyons A. Living with puerperal psychosis: a qualitative analysis. Psychol Psychother. 2003;76(Pt 4):411-431. doi:10.1348/147608303770584755
28. Mental Welfare Commission for Scotland. Perinatal Themed Visit Report: Keeping Mothers and Babies in Mind. Mental Welfare Commission for Scotland; 2016.
29. Wisner KL, Jennings KD, Conley B. Clinical dilemmas due to the lack of inpatient mother-baby units. Int J Psychiatry Med. 1996;26(4):479-493. doi:10.2190/NFJK-A4V7-CXUU-AM89
30. Battle CL, Howard MM. A mother-baby psychiatric day hospital: history, rationale, and why perinatal mental health is important for obstetric medicine. Obstet Med. 2014;7(2):66-70. doi:10.1177/1753495X13514402
31. Bullard ES, Meltzer-Brody S, Rubinow DR. The need for comprehensive psychiatric perinatal care-the University of North Carolina at Chapel Hill, Department of Psychiatry, Center for Women’s Mood Disorders launches the first dedicated inpatient program in the United States. Am J Obstet Gynecol. 2009;201(5):e10-e11. doi:10.1016/j.ajog.2009.05.004
32. Meltzer-Brody S, Brandon AR, Pearson B, et al. Evaluating the clinical effectiveness of a specialized perinatal psychiatry inpatient unit. Arch Womens Ment Health. 2014;17(2):107-113. doi:10.1007/s00737-013-0390-7
33. Alvarez-Toro V. Gender-specific care for women in psychiatric units. J Am Acad Psychiatry Law. 2022;JAAPL.220015-21. doi:10.29158/JAAPL.220015-21
34. Diaz-Caneja A, Johnson S. The views and experiences of severely mentally ill mothers--a qualitative study. Soc Psychiatry Psychiatr Epidemiol. 2004;39(6):472-482. doi:10.1007/s00127-004-0772-2
35. Gewurtz R, Krupa T, Eastabrook S, et al. Prevalence and characteristics of parenting among people served by assertive community treatment. Psychiatr Rehabil J. 2004;28(1):63-65. doi:10.2975/28.2004.63.65
36. Howard LM, Kumar R, Thornicroft G. Psychosocial characteristics and needs of mothers with psychotic disorders. Br J Psychiatry. 2001;178:427-432. doi:10.1192/bjp.178.5.427
37. Hollingsworth LD. Child custody loss among women with persistent severe mental illness. Social Work Research. 2004;28(4):199-209. doi:10.1093/swr/28.4.199
38. Dipple H, Smith S, Andrews H, et al. The experience of motherhood in women with severe and enduring mental illness. Soc Psychiatry Psychiatr Epidemiolf. 2002;37(7):336-340. doi:10.1007/s00127-002-0559-2
39. Seeman MV. Intervention to prevent child custody loss in mothers with schizophrenia. Schizophr Res Treatment. 2012;2012:796763. doi:10.1155/2012/796763
1. Brunner E, Falk DM, Jones M, et al. Olanzapine in pregnancy and breastfeeding: a review of data from global safety surveillance. BMC Pharmacol Toxicol. 2013;14:38. doi:10.1186/2050-6511-14-38
2. Seeman MV. Relational ethics: when mothers suffer from psychosis. Arch Womens Ment Health. 2004;7(3):201-210. doi:10.1007/s00737-004-0054-8
3. Motee A, Jeewon R. Importance of exclusive breastfeeding and complementary feeding among infants. Curr Res Nutr Food Sci. 2014;2(2). doi:10.12944/CRNFSJ.2.2.02
4. Committee Opinion No. 570: breastfeeding in underserved women: increasing initiation and continuation of breastfeeding. Obstet Gynecol. 2013;122(2 Pt 1):423-427. doi:10.1097/01.AOG.0000433008.93971.6a
5. Sibolboro Mezzacappa E, Endicott J. Parity mediates the association between infant feeding method and maternal depressive symptoms in the postpartum. Arch Womens Ment Health. 2007;10(6):259-266. doi:10.1007/s00737-007-0207-7
6. Kramer MS, Chalmers B, Hodnett ED, et al. Promotion of Breastfeeding Intervention Trial (PROBIT): a randomized trial in the Republic of Belarus. JAMA. 2001;285(4):413-420. doi:10.1001/jama.285.4.413
7. American Academy of Pediatrics. American Academy of Pediatrics calls for more support for breastfeeding mothers within updated policy recommendations. June 27, 2022. Accessed October 4, 2022. https://www.aap.org/en/news-room/news-releases/aap/2022/american-academy-of-pediatrics-calls-for-more-support-for-breastfeeding-mothers-within-updated-policy-recommendations
8. Hipwell AE, Kumar R. Maternal psychopathology and prediction of outcome based on mother-infant interaction ratings (BMIS). Br J Psychiatry. 1996;169(5):655-661. doi:10.1192/bjp.169.5.655
9. Chandra PS, Bhargavaraman RP, Raghunandan VN, et al. Delusions related to infant and their association with mother-infant interactions in postpartum psychotic disorders. Arch Womens Ment Health. 2006;9(5):285-288. doi:10.1007/s00737-006-0147-7
10. Klinger G, Stahl B, Fusar-Poli P, et al. Antipsychotic drugs and breastfeeding. Pediatr Endocrinol Rev. 2013;10(3):308-317.
11. Uguz F. A new safety scoring system for the use of psychotropic drugs during lactation. Am J Ther. 2021;28(1):e118-e126. doi:10.1097/MJT.0000000000000909
12. Hale TW, Krutsch K. Hale’s Medications & Mothers’ Milk, 2023: A Manual of Lactational Pharmacology. 20th ed. Springer Publishing Company; 2023.
13. Komaroff A. Aripiprazole and lactation failure: the importance of shared decision making. A case report. Case Rep Womens Health. 2021;30:e00308. doi:10.1016/j.crwh.2021.e00308
14. Dennis CL, McQueen K. Does maternal postpartum depressive symptomatology influence infant feeding outcomes? Acta Pediatr. 2007;96(4):590-594. doi:10.1111/j.1651-2227.2007.00184.x
15. Chaput KH, Nettel-Aguirre A, Musto R, et al. Breastfeeding difficulties and supports and risk of postpartum depression in a cohort of women who have given birth in Calgary: a prospective cohort study. CMAJ Open. 2016;4(1):E103-E109. doi:10.9778/cmajo.20150009
16. Dias CC, Figueiredo B. Breastfeeding and depression: a systematic review of the literature. J Affect Disord. 2015;171:142-154. doi:10.1016/j.jad.2014.09.022
17. Brown A, Rance J, Bennett P. Understanding the relationship between breastfeeding and postnatal depression: the role of pain and physical difficulties. J Adv Nurs. 2016;72(2):273-282. doi:10.1111/jan.12832
18. Rosenbaum KA. Exclusive breastmilk pumping: a concept analysis. Nurs Forum. 2022;57(5):946-953. doi:10.1111/nuf.12766
19. Boone KM, Geraghty SR, Keim SA. Feeding at the breast and expressed milk feeding: associations with otitis media and diarrhea in infants. J Pediatr. 2016;174:118-125. doi:10.1016/j.jpeds.2016.04.006
20. Epstein LJ, Kristo D, Strollo PJ Jr, et al; Adult Obstructive Sleep Apnea Task Force of the American Academy of Sleep Medicine. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med. 2009;5(3):263-276.
21. Caan MP, Sreshta NE, Okwerekwu JA, et al. Clinical and legal considerations regarding breastfeeding on psychiatric units. J Am Acad Psychiatry Law. 2022;50(2):200-207. doi:10.29158/JAAPL.210086-21
22. Glangeaud-Freudenthal NMC, Rainelli C, Cazas O, et al. Inpatient mother and baby psychiatric units (MBUs) and day cares. In: Sutter-Dallay AL, Glangeaud-Freudenthal NC, Guedeney A, et al, eds. Joint Care of Parents and Infants in Perinatal Psychiatry. Springer, Cham; 2016:147-164. doi:10.1007/978-3-319-21557-0_10
23. Dembosky A. A humane approach to caring for new mothers in psychiatric crisis. Health Aff (Millwood). 2021;40(10):1528-1533. doi:10.1377/hlthaff.2021.01288
24. Connellan K, Bartholomaeus C, Due C, et al. A systematic review of research on psychiatric mother-baby units. Arch Womens Ment Health. 2017;20(3):373-388. doi:10.1007/s00737-017-0718-9
25. Griffiths J, Lever Taylor B, Morant N, et al. A qualitative comparison of experiences of specialist mother and baby units versus general psychiatric wards. BMC Psychiatry. 2019;19(1):401. doi:10.1186/s12888-019-2389-8
26. Heron J, Gilbert N, Dolman C, et al. Information and support needs during recovery from postpartum psychosis. Arch Womens Ment Health. 2012;15(3):155-165. doi:10.1007/s00737-012-0267-1
27. Robertson E, Lyons A. Living with puerperal psychosis: a qualitative analysis. Psychol Psychother. 2003;76(Pt 4):411-431. doi:10.1348/147608303770584755
28. Mental Welfare Commission for Scotland. Perinatal Themed Visit Report: Keeping Mothers and Babies in Mind. Mental Welfare Commission for Scotland; 2016.
29. Wisner KL, Jennings KD, Conley B. Clinical dilemmas due to the lack of inpatient mother-baby units. Int J Psychiatry Med. 1996;26(4):479-493. doi:10.2190/NFJK-A4V7-CXUU-AM89
30. Battle CL, Howard MM. A mother-baby psychiatric day hospital: history, rationale, and why perinatal mental health is important for obstetric medicine. Obstet Med. 2014;7(2):66-70. doi:10.1177/1753495X13514402
31. Bullard ES, Meltzer-Brody S, Rubinow DR. The need for comprehensive psychiatric perinatal care-the University of North Carolina at Chapel Hill, Department of Psychiatry, Center for Women’s Mood Disorders launches the first dedicated inpatient program in the United States. Am J Obstet Gynecol. 2009;201(5):e10-e11. doi:10.1016/j.ajog.2009.05.004
32. Meltzer-Brody S, Brandon AR, Pearson B, et al. Evaluating the clinical effectiveness of a specialized perinatal psychiatry inpatient unit. Arch Womens Ment Health. 2014;17(2):107-113. doi:10.1007/s00737-013-0390-7
33. Alvarez-Toro V. Gender-specific care for women in psychiatric units. J Am Acad Psychiatry Law. 2022;JAAPL.220015-21. doi:10.29158/JAAPL.220015-21
34. Diaz-Caneja A, Johnson S. The views and experiences of severely mentally ill mothers--a qualitative study. Soc Psychiatry Psychiatr Epidemiol. 2004;39(6):472-482. doi:10.1007/s00127-004-0772-2
35. Gewurtz R, Krupa T, Eastabrook S, et al. Prevalence and characteristics of parenting among people served by assertive community treatment. Psychiatr Rehabil J. 2004;28(1):63-65. doi:10.2975/28.2004.63.65
36. Howard LM, Kumar R, Thornicroft G. Psychosocial characteristics and needs of mothers with psychotic disorders. Br J Psychiatry. 2001;178:427-432. doi:10.1192/bjp.178.5.427
37. Hollingsworth LD. Child custody loss among women with persistent severe mental illness. Social Work Research. 2004;28(4):199-209. doi:10.1093/swr/28.4.199
38. Dipple H, Smith S, Andrews H, et al. The experience of motherhood in women with severe and enduring mental illness. Soc Psychiatry Psychiatr Epidemiolf. 2002;37(7):336-340. doi:10.1007/s00127-002-0559-2
39. Seeman MV. Intervention to prevent child custody loss in mothers with schizophrenia. Schizophr Res Treatment. 2012;2012:796763. doi:10.1155/2012/796763
Excessive TV-watching tied to elevated risk for dementia, Parkinson’s disease, and depression
TOPLINE:
whereas a limited amount of daily computer use that is not work-related is linked to a lower risk for dementia.
METHODOLOGY:
- Investigators analyzed data on 473,184 people aged 39-72 years from the UK Biobank who were enrolled from 2006 to 2010 and followed until a diagnosis of dementia, PD, depression, death, or study end (2018 for Wales residents; 2021 for residents of England and Scotland).
- Participants reported on the number of hours they spent outside of work exercising, watching television, and using the computer.
- MRI was conducted to determine participants’ brain volume.
TAKEAWAY:
- During the study, 6096 people developed dementia, 3000 developed PD, 23,600 developed depression, 1200 developed dementia and depression, and 486 developed PD and depression.
- Compared with those who watched TV for under 1 hour per day, those who reported watching 4 or more hours per day had a 28% higher risk for dementia (adjusted hazard ratio [aHR], 1.28; 95% CI, 1.17-1.39), a 35% higher risk for depression, (aHR, 1.35; 95% CI, 1.29-1.40) and a 16% greater risk for PD (aHR, 1.16; 95% CI, 1.03-1.29).
- However, moderate computer use outside of work seemed somewhat protective. Participants who used the computer for 30-60 minutes per day had lower risks for dementia (aHR, 0.68; 95% CI, 0.64-0.72), PD, (aHR, 0.86; 95% CI, 0.79-0.93), and depression (aHR, 0.85; 95% CI, 0.83-0.88) compared with those who reported the lowest levels of computer usage.
- Replacing 30 minutes per day of computer time with an equal amount of structured exercise was associated with decreased risk for dementia (aHR, 0.74; 95% CI, 0.85-0.95) and PD (aHR, 0.84; 95% CI, 0.78-0.90).
IN PRACTICE:
The association between extended periods of TV use and higher risk for PD and dementia could be explained by a lack of activity, the authors note. They add that sedentary behavior is, “associated with biomarkers of low-grade inflammation and changes in inflammation markers that could initiate and or worsen neuroinflammation and contribute to neurodegeneration.”
SOURCE:
Hanzhang Wu, PhD, of Tianjin University of Traditional Medicine in Tianjin, China, led the study, which was published online in the International Journal of Behavioral Nutrition and Physical Activity.
LIMITATIONS:
Screen behaviors were assessed using self-report measures, which is subject to recall bias. Also, there may have been variables confounding the findings for which investigators did not account.
DISCLOSURES:
The study was funded by the National Natural Science Foundation of China, the Tianjin Major Public Health Science and Technology Project, the National Health Commission of China, the Food Science and Technology Foundation of Chinese Institute of Food Science and Technology, the China Cohort Consortium, and the Chinese Nutrition Society Nutrition Research Foundation–DSM Research Fund, China. There were no disclosures reported.
Eve Bender has no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
whereas a limited amount of daily computer use that is not work-related is linked to a lower risk for dementia.
METHODOLOGY:
- Investigators analyzed data on 473,184 people aged 39-72 years from the UK Biobank who were enrolled from 2006 to 2010 and followed until a diagnosis of dementia, PD, depression, death, or study end (2018 for Wales residents; 2021 for residents of England and Scotland).
- Participants reported on the number of hours they spent outside of work exercising, watching television, and using the computer.
- MRI was conducted to determine participants’ brain volume.
TAKEAWAY:
- During the study, 6096 people developed dementia, 3000 developed PD, 23,600 developed depression, 1200 developed dementia and depression, and 486 developed PD and depression.
- Compared with those who watched TV for under 1 hour per day, those who reported watching 4 or more hours per day had a 28% higher risk for dementia (adjusted hazard ratio [aHR], 1.28; 95% CI, 1.17-1.39), a 35% higher risk for depression, (aHR, 1.35; 95% CI, 1.29-1.40) and a 16% greater risk for PD (aHR, 1.16; 95% CI, 1.03-1.29).
- However, moderate computer use outside of work seemed somewhat protective. Participants who used the computer for 30-60 minutes per day had lower risks for dementia (aHR, 0.68; 95% CI, 0.64-0.72), PD, (aHR, 0.86; 95% CI, 0.79-0.93), and depression (aHR, 0.85; 95% CI, 0.83-0.88) compared with those who reported the lowest levels of computer usage.
- Replacing 30 minutes per day of computer time with an equal amount of structured exercise was associated with decreased risk for dementia (aHR, 0.74; 95% CI, 0.85-0.95) and PD (aHR, 0.84; 95% CI, 0.78-0.90).
IN PRACTICE:
The association between extended periods of TV use and higher risk for PD and dementia could be explained by a lack of activity, the authors note. They add that sedentary behavior is, “associated with biomarkers of low-grade inflammation and changes in inflammation markers that could initiate and or worsen neuroinflammation and contribute to neurodegeneration.”
SOURCE:
Hanzhang Wu, PhD, of Tianjin University of Traditional Medicine in Tianjin, China, led the study, which was published online in the International Journal of Behavioral Nutrition and Physical Activity.
LIMITATIONS:
Screen behaviors were assessed using self-report measures, which is subject to recall bias. Also, there may have been variables confounding the findings for which investigators did not account.
DISCLOSURES:
The study was funded by the National Natural Science Foundation of China, the Tianjin Major Public Health Science and Technology Project, the National Health Commission of China, the Food Science and Technology Foundation of Chinese Institute of Food Science and Technology, the China Cohort Consortium, and the Chinese Nutrition Society Nutrition Research Foundation–DSM Research Fund, China. There were no disclosures reported.
Eve Bender has no relevant financial relationships.
A version of this article appeared on Medscape.com.
TOPLINE:
whereas a limited amount of daily computer use that is not work-related is linked to a lower risk for dementia.
METHODOLOGY:
- Investigators analyzed data on 473,184 people aged 39-72 years from the UK Biobank who were enrolled from 2006 to 2010 and followed until a diagnosis of dementia, PD, depression, death, or study end (2018 for Wales residents; 2021 for residents of England and Scotland).
- Participants reported on the number of hours they spent outside of work exercising, watching television, and using the computer.
- MRI was conducted to determine participants’ brain volume.
TAKEAWAY:
- During the study, 6096 people developed dementia, 3000 developed PD, 23,600 developed depression, 1200 developed dementia and depression, and 486 developed PD and depression.
- Compared with those who watched TV for under 1 hour per day, those who reported watching 4 or more hours per day had a 28% higher risk for dementia (adjusted hazard ratio [aHR], 1.28; 95% CI, 1.17-1.39), a 35% higher risk for depression, (aHR, 1.35; 95% CI, 1.29-1.40) and a 16% greater risk for PD (aHR, 1.16; 95% CI, 1.03-1.29).
- However, moderate computer use outside of work seemed somewhat protective. Participants who used the computer for 30-60 minutes per day had lower risks for dementia (aHR, 0.68; 95% CI, 0.64-0.72), PD, (aHR, 0.86; 95% CI, 0.79-0.93), and depression (aHR, 0.85; 95% CI, 0.83-0.88) compared with those who reported the lowest levels of computer usage.
- Replacing 30 minutes per day of computer time with an equal amount of structured exercise was associated with decreased risk for dementia (aHR, 0.74; 95% CI, 0.85-0.95) and PD (aHR, 0.84; 95% CI, 0.78-0.90).
IN PRACTICE:
The association between extended periods of TV use and higher risk for PD and dementia could be explained by a lack of activity, the authors note. They add that sedentary behavior is, “associated with biomarkers of low-grade inflammation and changes in inflammation markers that could initiate and or worsen neuroinflammation and contribute to neurodegeneration.”
SOURCE:
Hanzhang Wu, PhD, of Tianjin University of Traditional Medicine in Tianjin, China, led the study, which was published online in the International Journal of Behavioral Nutrition and Physical Activity.
LIMITATIONS:
Screen behaviors were assessed using self-report measures, which is subject to recall bias. Also, there may have been variables confounding the findings for which investigators did not account.
DISCLOSURES:
The study was funded by the National Natural Science Foundation of China, the Tianjin Major Public Health Science and Technology Project, the National Health Commission of China, the Food Science and Technology Foundation of Chinese Institute of Food Science and Technology, the China Cohort Consortium, and the Chinese Nutrition Society Nutrition Research Foundation–DSM Research Fund, China. There were no disclosures reported.
Eve Bender has no relevant financial relationships.
A version of this article appeared on Medscape.com.
MDMA therapy for loneliness? Researchers say it could work
Some call the drug “ecstasy” or “molly.” Researchers are calling it a potential tool to help treat loneliness.
As public health experts sound the alarm on a rising loneliness epidemic in the United States and across the globe,
In the latest study, MDMA “led to a robust increase in feelings of connection” among people socializing in a controlled setting. Participants were dosed with either MDMA or a placebo and asked to chat with a stranger. Afterward, those who took MDMA said their companion was more responsive and attentive, and that they had plenty in common. The drug also “increased participants’ ratings of liking their partners, feeling connected and finding the conversation enjoyable and meaningful.”
The study was small — just 18 participants — but its results “have implications for MDMA-assisted therapy,” the authors wrote. “This feeling of connectedness could help patients feel safe and trusting, thereby facilitating deeper emotional exploration.”
MDMA “really does seem to make people want to interact more with other people,” says Harriet de Wit, PhD, a neuropharmacologist at the University of Chicago and one of the study’s authors. The results echo those of earlier research using psychedelics like LSD or psilocybin.
It’s important to note that any intervention involving MDMA or psychedelics would be a drug-assisted therapy — that is, used in conjunction with the appropriate therapy and in a therapeutic setting. MDMA-assisted therapy has already drawn popular and scientific attention, as it recently cleared clinical trials for treating posttraumatic stress disorder (PTSD) and may be nearing approval by the US Food and Drug Administration (FDA).
According to Friederike Holze, PhD, psychopharmacologist at the University of Basel, in Switzerland, “there could be a place” for MDMA and psychedelics in treating chronic loneliness, but only under professional supervision.
There would have to be clear guidelines too, says Joshua Woolley, MD, PhD, a psychiatrist at the University of California, San Francisco.
MDMA and psychedelics “induce this plastic state, a state where people can change. They feel open, they feel like things are possible,” Dr. Woolley says. Then, with therapy, “you can help them change.”
Loneliness Can Impact Our Health
On top of the mental health ramifications, the physiologic effects of loneliness could have grave consequences over time. In observational studies, loneliness has been linked to higher risks for cancer and heart disease, and shorter lifespan. One third of Americans over 45 say they are chronically lonely.
Chronic loneliness changes how we think and behave, research shows. It makes us fear contact with others and see them in a more negative light, as more threatening and less trustworthy. Lonely people prefer to stand farther apart from strangers and avoid touch.
This is where MDMA-assisted therapies could potentially help, by easing these defensive tendencies, according to Dr. Woolley.
MDMA, Psychedelics, and Social Behavior
MDMA, or 3,4-methylenedioxymethamphetamine, is a hybrid between a stimulant and a psychedelic. In Dr. de Wit’s earlier experiments, volunteers given MDMA engaged more in communal activities, chatting, and playing games. They used more positive words during social encounters than those who had received a placebo. And after MDMA, people felt less rejected if they were slighted in Cyberball — a virtual ball-tossing game commonly used to measure the effects of social exclusion.
MDMA has been shown to reduce people’s response to other’s negative emotions, diminishing activation of the amygdala (the brain’s fear center) while looking at pictures of angry faces.
This could be helpful. “If you perceive a person’s natural expression as being a little bit angry, if that disappears, then you might be more inclined to interact,” de Wit says.
However, there may be downsides, too. If a drug makes people more trusting and willing to connect, they could be taken advantage of. This is why, Dr. Woolley says, “psychedelics have been used in cults.”
MDMA may also make the experience of touch more pleasant. In a series of experiments in 2019, researchers gently stroked volunteers ’ arms with a goat-hair brush, mimicking the comforting gestures one may receive from a loved one. At the same time, the scientists monitored the volunteers’ facial muscles. People on MDMA perceived gentle touch as more pleasant than those on placebo, and their smile muscles activated more.
MDMA and psychedelics boost social behaviors in animals, too — suggesting that their effects on relationships have a biological basis. Rats on MDMA are more likely to lie next to each other, and mice become more resilient to social stress. Even octopuses become more outgoing after a dose of MDMA, choosing to spend more time with other octopuses instead of a new toy. Classic psychedelics show similar effects — LSD, for example, makes mice more social.
Psychedelics can induce a sense of a “dissolution of the self-other boundary,” Dr. Woolley says. People who take them often say it’s “helped them feel more connected to themselves and other people.” LSD, first synthesized in 1938, may help increase empathy in some people.
Psilocybin, a compound found in over 200 species of mushrooms and used for centuries in Mesoamerican rituals, also seems to boost empathy, with effects persisting for at least seven days. In Cyberball, the online ball-throwing game, people who took psilocybin felt less socially rejected, an outcome reflected in their brain activation patterns in one study — the areas responsible for social-pain processing appeared to dim after a dose.
Making It Legal and Putting It to Use
In 2020, Oregon became the first state to establish a regulatory framework for psilocybin for therapeutic use, and Colorado followed suit in 2022. Such therapeutic applications of psilocybin could help fight loneliness as well, Dr. Woolley believes, because a “ common symptom of depression is that people feel socially withdrawn and lack motivation, ” he says. As mentioned above, MDMA-assisted therapy is also nearing FDA approval for PTSD.
What remain unclear are the exact mechanisms at play.
“MDMA releases oxytocin, and it does that through serotonin receptors,” Dr. de Wit says. Serotonin activates 5-HT1A receptors in the hypothalamus, releasing oxytocin into the bloodstream. In Dr. de Wit’s recent experiments, the more people felt connected after taking MDMA, the more oxytocin was found circulating in their bodies. (Another drug, methamphetamine, also upped the levels of oxytocin but did not increase feelings of connectedness.)
“It’s likely that both something in the serotonin system independent of oxytocin, and oxytocin itself, contribute,” Dr. de Wit says. Dopamine, a neurotransmitter responsible for motivation, appears to increase as well.
The empathy-boosting effects of LSD also seem to be at least partly driven by oxytocin, experiments published in 2021 revealed. Studies in mice, meanwhile, suggest that glutamate, a chemical messenger in the brain, may be behind some of LSD’s prosocial effects.
Scientists are fairly certain which receptors these drugs bind to and which neurotransmitters they affect. “How that gets translated into these higher-order things like empathy and feeling connected to the world, we don’t totally understand,” Dr. Woolley says.
Challenges and the Future
Although MDMA and psychedelics are largely considered safe when taken in a legal, medically controlled setting, there is reason to be cautious.
“They have relatively low impact on the body, like heart rate increase or blood pressure increase. But they might leave some disturbing psychological effects,” says Dr. Holze. Scientists routinely screen experiment volunteers for their risk for psychiatric disorders.
Although risk for addiction is low with both MDMA and psychedelics, there is always some risk for misuse. MDMA “ can produce feelings of well-being, and then people might use it repeatedly, ” Dr. de Wit says. “ That doesn ’ t seem to be a problem for really a lot of people, but it could easily happen. ”
Still, possibilities remain for MDMA in the fight against loneliness.
“[People] feel open, they feel like things are possible, they feel like they’re unstuck,” Dr. Woolley says. “You can harness that in psychotherapy.”
A version of this article appeared on Medscape.com.
Some call the drug “ecstasy” or “molly.” Researchers are calling it a potential tool to help treat loneliness.
As public health experts sound the alarm on a rising loneliness epidemic in the United States and across the globe,
In the latest study, MDMA “led to a robust increase in feelings of connection” among people socializing in a controlled setting. Participants were dosed with either MDMA or a placebo and asked to chat with a stranger. Afterward, those who took MDMA said their companion was more responsive and attentive, and that they had plenty in common. The drug also “increased participants’ ratings of liking their partners, feeling connected and finding the conversation enjoyable and meaningful.”
The study was small — just 18 participants — but its results “have implications for MDMA-assisted therapy,” the authors wrote. “This feeling of connectedness could help patients feel safe and trusting, thereby facilitating deeper emotional exploration.”
MDMA “really does seem to make people want to interact more with other people,” says Harriet de Wit, PhD, a neuropharmacologist at the University of Chicago and one of the study’s authors. The results echo those of earlier research using psychedelics like LSD or psilocybin.
It’s important to note that any intervention involving MDMA or psychedelics would be a drug-assisted therapy — that is, used in conjunction with the appropriate therapy and in a therapeutic setting. MDMA-assisted therapy has already drawn popular and scientific attention, as it recently cleared clinical trials for treating posttraumatic stress disorder (PTSD) and may be nearing approval by the US Food and Drug Administration (FDA).
According to Friederike Holze, PhD, psychopharmacologist at the University of Basel, in Switzerland, “there could be a place” for MDMA and psychedelics in treating chronic loneliness, but only under professional supervision.
There would have to be clear guidelines too, says Joshua Woolley, MD, PhD, a psychiatrist at the University of California, San Francisco.
MDMA and psychedelics “induce this plastic state, a state where people can change. They feel open, they feel like things are possible,” Dr. Woolley says. Then, with therapy, “you can help them change.”
Loneliness Can Impact Our Health
On top of the mental health ramifications, the physiologic effects of loneliness could have grave consequences over time. In observational studies, loneliness has been linked to higher risks for cancer and heart disease, and shorter lifespan. One third of Americans over 45 say they are chronically lonely.
Chronic loneliness changes how we think and behave, research shows. It makes us fear contact with others and see them in a more negative light, as more threatening and less trustworthy. Lonely people prefer to stand farther apart from strangers and avoid touch.
This is where MDMA-assisted therapies could potentially help, by easing these defensive tendencies, according to Dr. Woolley.
MDMA, Psychedelics, and Social Behavior
MDMA, or 3,4-methylenedioxymethamphetamine, is a hybrid between a stimulant and a psychedelic. In Dr. de Wit’s earlier experiments, volunteers given MDMA engaged more in communal activities, chatting, and playing games. They used more positive words during social encounters than those who had received a placebo. And after MDMA, people felt less rejected if they were slighted in Cyberball — a virtual ball-tossing game commonly used to measure the effects of social exclusion.
MDMA has been shown to reduce people’s response to other’s negative emotions, diminishing activation of the amygdala (the brain’s fear center) while looking at pictures of angry faces.
This could be helpful. “If you perceive a person’s natural expression as being a little bit angry, if that disappears, then you might be more inclined to interact,” de Wit says.
However, there may be downsides, too. If a drug makes people more trusting and willing to connect, they could be taken advantage of. This is why, Dr. Woolley says, “psychedelics have been used in cults.”
MDMA may also make the experience of touch more pleasant. In a series of experiments in 2019, researchers gently stroked volunteers ’ arms with a goat-hair brush, mimicking the comforting gestures one may receive from a loved one. At the same time, the scientists monitored the volunteers’ facial muscles. People on MDMA perceived gentle touch as more pleasant than those on placebo, and their smile muscles activated more.
MDMA and psychedelics boost social behaviors in animals, too — suggesting that their effects on relationships have a biological basis. Rats on MDMA are more likely to lie next to each other, and mice become more resilient to social stress. Even octopuses become more outgoing after a dose of MDMA, choosing to spend more time with other octopuses instead of a new toy. Classic psychedelics show similar effects — LSD, for example, makes mice more social.
Psychedelics can induce a sense of a “dissolution of the self-other boundary,” Dr. Woolley says. People who take them often say it’s “helped them feel more connected to themselves and other people.” LSD, first synthesized in 1938, may help increase empathy in some people.
Psilocybin, a compound found in over 200 species of mushrooms and used for centuries in Mesoamerican rituals, also seems to boost empathy, with effects persisting for at least seven days. In Cyberball, the online ball-throwing game, people who took psilocybin felt less socially rejected, an outcome reflected in their brain activation patterns in one study — the areas responsible for social-pain processing appeared to dim after a dose.
Making It Legal and Putting It to Use
In 2020, Oregon became the first state to establish a regulatory framework for psilocybin for therapeutic use, and Colorado followed suit in 2022. Such therapeutic applications of psilocybin could help fight loneliness as well, Dr. Woolley believes, because a “ common symptom of depression is that people feel socially withdrawn and lack motivation, ” he says. As mentioned above, MDMA-assisted therapy is also nearing FDA approval for PTSD.
What remain unclear are the exact mechanisms at play.
“MDMA releases oxytocin, and it does that through serotonin receptors,” Dr. de Wit says. Serotonin activates 5-HT1A receptors in the hypothalamus, releasing oxytocin into the bloodstream. In Dr. de Wit’s recent experiments, the more people felt connected after taking MDMA, the more oxytocin was found circulating in their bodies. (Another drug, methamphetamine, also upped the levels of oxytocin but did not increase feelings of connectedness.)
“It’s likely that both something in the serotonin system independent of oxytocin, and oxytocin itself, contribute,” Dr. de Wit says. Dopamine, a neurotransmitter responsible for motivation, appears to increase as well.
The empathy-boosting effects of LSD also seem to be at least partly driven by oxytocin, experiments published in 2021 revealed. Studies in mice, meanwhile, suggest that glutamate, a chemical messenger in the brain, may be behind some of LSD’s prosocial effects.
Scientists are fairly certain which receptors these drugs bind to and which neurotransmitters they affect. “How that gets translated into these higher-order things like empathy and feeling connected to the world, we don’t totally understand,” Dr. Woolley says.
Challenges and the Future
Although MDMA and psychedelics are largely considered safe when taken in a legal, medically controlled setting, there is reason to be cautious.
“They have relatively low impact on the body, like heart rate increase or blood pressure increase. But they might leave some disturbing psychological effects,” says Dr. Holze. Scientists routinely screen experiment volunteers for their risk for psychiatric disorders.
Although risk for addiction is low with both MDMA and psychedelics, there is always some risk for misuse. MDMA “ can produce feelings of well-being, and then people might use it repeatedly, ” Dr. de Wit says. “ That doesn ’ t seem to be a problem for really a lot of people, but it could easily happen. ”
Still, possibilities remain for MDMA in the fight against loneliness.
“[People] feel open, they feel like things are possible, they feel like they’re unstuck,” Dr. Woolley says. “You can harness that in psychotherapy.”
A version of this article appeared on Medscape.com.
Some call the drug “ecstasy” or “molly.” Researchers are calling it a potential tool to help treat loneliness.
As public health experts sound the alarm on a rising loneliness epidemic in the United States and across the globe,
In the latest study, MDMA “led to a robust increase in feelings of connection” among people socializing in a controlled setting. Participants were dosed with either MDMA or a placebo and asked to chat with a stranger. Afterward, those who took MDMA said their companion was more responsive and attentive, and that they had plenty in common. The drug also “increased participants’ ratings of liking their partners, feeling connected and finding the conversation enjoyable and meaningful.”
The study was small — just 18 participants — but its results “have implications for MDMA-assisted therapy,” the authors wrote. “This feeling of connectedness could help patients feel safe and trusting, thereby facilitating deeper emotional exploration.”
MDMA “really does seem to make people want to interact more with other people,” says Harriet de Wit, PhD, a neuropharmacologist at the University of Chicago and one of the study’s authors. The results echo those of earlier research using psychedelics like LSD or psilocybin.
It’s important to note that any intervention involving MDMA or psychedelics would be a drug-assisted therapy — that is, used in conjunction with the appropriate therapy and in a therapeutic setting. MDMA-assisted therapy has already drawn popular and scientific attention, as it recently cleared clinical trials for treating posttraumatic stress disorder (PTSD) and may be nearing approval by the US Food and Drug Administration (FDA).
According to Friederike Holze, PhD, psychopharmacologist at the University of Basel, in Switzerland, “there could be a place” for MDMA and psychedelics in treating chronic loneliness, but only under professional supervision.
There would have to be clear guidelines too, says Joshua Woolley, MD, PhD, a psychiatrist at the University of California, San Francisco.
MDMA and psychedelics “induce this plastic state, a state where people can change. They feel open, they feel like things are possible,” Dr. Woolley says. Then, with therapy, “you can help them change.”
Loneliness Can Impact Our Health
On top of the mental health ramifications, the physiologic effects of loneliness could have grave consequences over time. In observational studies, loneliness has been linked to higher risks for cancer and heart disease, and shorter lifespan. One third of Americans over 45 say they are chronically lonely.
Chronic loneliness changes how we think and behave, research shows. It makes us fear contact with others and see them in a more negative light, as more threatening and less trustworthy. Lonely people prefer to stand farther apart from strangers and avoid touch.
This is where MDMA-assisted therapies could potentially help, by easing these defensive tendencies, according to Dr. Woolley.
MDMA, Psychedelics, and Social Behavior
MDMA, or 3,4-methylenedioxymethamphetamine, is a hybrid between a stimulant and a psychedelic. In Dr. de Wit’s earlier experiments, volunteers given MDMA engaged more in communal activities, chatting, and playing games. They used more positive words during social encounters than those who had received a placebo. And after MDMA, people felt less rejected if they were slighted in Cyberball — a virtual ball-tossing game commonly used to measure the effects of social exclusion.
MDMA has been shown to reduce people’s response to other’s negative emotions, diminishing activation of the amygdala (the brain’s fear center) while looking at pictures of angry faces.
This could be helpful. “If you perceive a person’s natural expression as being a little bit angry, if that disappears, then you might be more inclined to interact,” de Wit says.
However, there may be downsides, too. If a drug makes people more trusting and willing to connect, they could be taken advantage of. This is why, Dr. Woolley says, “psychedelics have been used in cults.”
MDMA may also make the experience of touch more pleasant. In a series of experiments in 2019, researchers gently stroked volunteers ’ arms with a goat-hair brush, mimicking the comforting gestures one may receive from a loved one. At the same time, the scientists monitored the volunteers’ facial muscles. People on MDMA perceived gentle touch as more pleasant than those on placebo, and their smile muscles activated more.
MDMA and psychedelics boost social behaviors in animals, too — suggesting that their effects on relationships have a biological basis. Rats on MDMA are more likely to lie next to each other, and mice become more resilient to social stress. Even octopuses become more outgoing after a dose of MDMA, choosing to spend more time with other octopuses instead of a new toy. Classic psychedelics show similar effects — LSD, for example, makes mice more social.
Psychedelics can induce a sense of a “dissolution of the self-other boundary,” Dr. Woolley says. People who take them often say it’s “helped them feel more connected to themselves and other people.” LSD, first synthesized in 1938, may help increase empathy in some people.
Psilocybin, a compound found in over 200 species of mushrooms and used for centuries in Mesoamerican rituals, also seems to boost empathy, with effects persisting for at least seven days. In Cyberball, the online ball-throwing game, people who took psilocybin felt less socially rejected, an outcome reflected in their brain activation patterns in one study — the areas responsible for social-pain processing appeared to dim after a dose.
Making It Legal and Putting It to Use
In 2020, Oregon became the first state to establish a regulatory framework for psilocybin for therapeutic use, and Colorado followed suit in 2022. Such therapeutic applications of psilocybin could help fight loneliness as well, Dr. Woolley believes, because a “ common symptom of depression is that people feel socially withdrawn and lack motivation, ” he says. As mentioned above, MDMA-assisted therapy is also nearing FDA approval for PTSD.
What remain unclear are the exact mechanisms at play.
“MDMA releases oxytocin, and it does that through serotonin receptors,” Dr. de Wit says. Serotonin activates 5-HT1A receptors in the hypothalamus, releasing oxytocin into the bloodstream. In Dr. de Wit’s recent experiments, the more people felt connected after taking MDMA, the more oxytocin was found circulating in their bodies. (Another drug, methamphetamine, also upped the levels of oxytocin but did not increase feelings of connectedness.)
“It’s likely that both something in the serotonin system independent of oxytocin, and oxytocin itself, contribute,” Dr. de Wit says. Dopamine, a neurotransmitter responsible for motivation, appears to increase as well.
The empathy-boosting effects of LSD also seem to be at least partly driven by oxytocin, experiments published in 2021 revealed. Studies in mice, meanwhile, suggest that glutamate, a chemical messenger in the brain, may be behind some of LSD’s prosocial effects.
Scientists are fairly certain which receptors these drugs bind to and which neurotransmitters they affect. “How that gets translated into these higher-order things like empathy and feeling connected to the world, we don’t totally understand,” Dr. Woolley says.
Challenges and the Future
Although MDMA and psychedelics are largely considered safe when taken in a legal, medically controlled setting, there is reason to be cautious.
“They have relatively low impact on the body, like heart rate increase or blood pressure increase. But they might leave some disturbing psychological effects,” says Dr. Holze. Scientists routinely screen experiment volunteers for their risk for psychiatric disorders.
Although risk for addiction is low with both MDMA and psychedelics, there is always some risk for misuse. MDMA “ can produce feelings of well-being, and then people might use it repeatedly, ” Dr. de Wit says. “ That doesn ’ t seem to be a problem for really a lot of people, but it could easily happen. ”
Still, possibilities remain for MDMA in the fight against loneliness.
“[People] feel open, they feel like things are possible, they feel like they’re unstuck,” Dr. Woolley says. “You can harness that in psychotherapy.”
A version of this article appeared on Medscape.com.
Higher blood levels of oleic acid tied to depression
TOPLINE:
National survey data reveal an association between higher serum oleic acid levels and depression in adults, new research shows.
METHODOLOGY:
Oleic acid is the most abundant fatty acid in plasma and has been associated with multiple neurologic diseases. However, the relationship between oleic acid and depression is unclear.
This cross-sectional analyzed data on 4459 adults from the 2011-2014 National Health and Nutrition Examination Survey (NHANES).
Multivariable logistic regression models adjusting for demographics, health and lifestyle factors quantified the association between oleic acid levels and depression. Depression was assessed using the Patient Health Questionnaire-9 (PHQ-9) with depression defined as a score ≥ 10.
TAKEAWAY:
Serum oleic acid levels were positively associated with depression before and after multivariable adjustment.
After adjusting for all covariates, for every 1 mmol/L increase in serum oleic acid levels, the prevalence of depression increased by 40% (adjusted odds ratio [aOR], 1.40; 95% CI, 1.03-1.90).
Adults in the top quartile of oleic acid (≥ 2.51 mmol/L) had a greater than twofold higher likelihood of depression (aOR, 2.22; 95% CI, 1.04-4.73) compared with peers in the lowest quartile (≤ 1.54 mmol/L).
IN PRACTICE:
“A better understanding of the role of oleic acid in depression may lead to new preventive and therapeutic methods. Thus, carefully designed prospective studies are necessary to explore the positive effects of changing serum oleic acid levels through diet, medicine, or other measures on depression,” the authors write.
SOURCE:
The study, with first author Jiahui Yin of Shandong University of Traditional Chinese Medicine in Jinan, China, was published online on November 16, 2023 in BMC Psychiatry .
LIMITATIONS:
The cross-sectional study can’t prove causality. The findings may not apply to clinically diagnosed major depressive disorder.
DISCLOSURES:
The study had no specific funding. The authors report no conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
National survey data reveal an association between higher serum oleic acid levels and depression in adults, new research shows.
METHODOLOGY:
Oleic acid is the most abundant fatty acid in plasma and has been associated with multiple neurologic diseases. However, the relationship between oleic acid and depression is unclear.
This cross-sectional analyzed data on 4459 adults from the 2011-2014 National Health and Nutrition Examination Survey (NHANES).
Multivariable logistic regression models adjusting for demographics, health and lifestyle factors quantified the association between oleic acid levels and depression. Depression was assessed using the Patient Health Questionnaire-9 (PHQ-9) with depression defined as a score ≥ 10.
TAKEAWAY:
Serum oleic acid levels were positively associated with depression before and after multivariable adjustment.
After adjusting for all covariates, for every 1 mmol/L increase in serum oleic acid levels, the prevalence of depression increased by 40% (adjusted odds ratio [aOR], 1.40; 95% CI, 1.03-1.90).
Adults in the top quartile of oleic acid (≥ 2.51 mmol/L) had a greater than twofold higher likelihood of depression (aOR, 2.22; 95% CI, 1.04-4.73) compared with peers in the lowest quartile (≤ 1.54 mmol/L).
IN PRACTICE:
“A better understanding of the role of oleic acid in depression may lead to new preventive and therapeutic methods. Thus, carefully designed prospective studies are necessary to explore the positive effects of changing serum oleic acid levels through diet, medicine, or other measures on depression,” the authors write.
SOURCE:
The study, with first author Jiahui Yin of Shandong University of Traditional Chinese Medicine in Jinan, China, was published online on November 16, 2023 in BMC Psychiatry .
LIMITATIONS:
The cross-sectional study can’t prove causality. The findings may not apply to clinically diagnosed major depressive disorder.
DISCLOSURES:
The study had no specific funding. The authors report no conflicts of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
National survey data reveal an association between higher serum oleic acid levels and depression in adults, new research shows.
METHODOLOGY:
Oleic acid is the most abundant fatty acid in plasma and has been associated with multiple neurologic diseases. However, the relationship between oleic acid and depression is unclear.
This cross-sectional analyzed data on 4459 adults from the 2011-2014 National Health and Nutrition Examination Survey (NHANES).
Multivariable logistic regression models adjusting for demographics, health and lifestyle factors quantified the association between oleic acid levels and depression. Depression was assessed using the Patient Health Questionnaire-9 (PHQ-9) with depression defined as a score ≥ 10.
TAKEAWAY:
Serum oleic acid levels were positively associated with depression before and after multivariable adjustment.
After adjusting for all covariates, for every 1 mmol/L increase in serum oleic acid levels, the prevalence of depression increased by 40% (adjusted odds ratio [aOR], 1.40; 95% CI, 1.03-1.90).
Adults in the top quartile of oleic acid (≥ 2.51 mmol/L) had a greater than twofold higher likelihood of depression (aOR, 2.22; 95% CI, 1.04-4.73) compared with peers in the lowest quartile (≤ 1.54 mmol/L).
IN PRACTICE:
“A better understanding of the role of oleic acid in depression may lead to new preventive and therapeutic methods. Thus, carefully designed prospective studies are necessary to explore the positive effects of changing serum oleic acid levels through diet, medicine, or other measures on depression,” the authors write.
SOURCE:
The study, with first author Jiahui Yin of Shandong University of Traditional Chinese Medicine in Jinan, China, was published online on November 16, 2023 in BMC Psychiatry .
LIMITATIONS:
The cross-sectional study can’t prove causality. The findings may not apply to clinically diagnosed major depressive disorder.
DISCLOSURES:
The study had no specific funding. The authors report no conflicts of interest.
A version of this article first appeared on Medscape.com.
Mobile mental health apps linked with ‘significantly reduced’ depressive symptoms
TOPLINE:
A meta-analysis supports the use of mobile mental health apps, both as a standalone and added to conventional treatment, for adults with moderate to severe depression.
METHODOLOGY:
Mobile mental health apps have proliferated but data on their effectiveness in different patient populations is lacking.
To investigate, researchers conducted a systematic review and meta-analysis of 13 randomized clinical trials assessing treatment efficacy of mobile mental health apps in 1470 adults with moderate to severe depression.
The primary outcome was change in depression symptoms from pre- to post-treatment; secondary outcomes included patient-level factors associated with app efficacy.
TAKEAWAY:
Mobile app interventions were associated with significantly reduced depressive symptoms vs both active and inactive control groups, with a medium effect size (standardized mean difference [SMD] 0.50).
App interventions delivered for < 8 weeks had a significantly greater effect size than those delivered for 8+ weeks (SMD 0.77 vs 0.43). Apps were more effective in patients not on medication or in therapy. Apps offering rewards or incentives also appeared to be more effective.
Interventions with in-app notifications were associated with significantly lower treatment outcomes (SMD 0.45) than interventions without (SMD 0.45 vs 0.71).
IN PRACTICE:
“The significant treatment efficacy of app-based interventions compared with active and inactive controls suggests the potential of mobile app interventions as an alternative to conventional psychotherapy, with further merits in accessibility, financial affordability, and safety from stigma,” the authors write.
SOURCE:
The study, with first author Hayoung Bae, BA, with Korea University School of Psychology, Seoul, South Korea, was published online November 20 in JAMA Network Open .
LIMITATIONS:
The findings are based on a small number of trials, with significant heterogeneity among the included trials. The analysis included only English-language publications. Using summary data for the subgroup analyses might have prevented a detailed understanding of the moderating associations of individual participant characteristics.
DISCLOSURES:
The study was supported by a grant from the National Research Foundation funded by the Korean government. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com .
TOPLINE:
A meta-analysis supports the use of mobile mental health apps, both as a standalone and added to conventional treatment, for adults with moderate to severe depression.
METHODOLOGY:
Mobile mental health apps have proliferated but data on their effectiveness in different patient populations is lacking.
To investigate, researchers conducted a systematic review and meta-analysis of 13 randomized clinical trials assessing treatment efficacy of mobile mental health apps in 1470 adults with moderate to severe depression.
The primary outcome was change in depression symptoms from pre- to post-treatment; secondary outcomes included patient-level factors associated with app efficacy.
TAKEAWAY:
Mobile app interventions were associated with significantly reduced depressive symptoms vs both active and inactive control groups, with a medium effect size (standardized mean difference [SMD] 0.50).
App interventions delivered for < 8 weeks had a significantly greater effect size than those delivered for 8+ weeks (SMD 0.77 vs 0.43). Apps were more effective in patients not on medication or in therapy. Apps offering rewards or incentives also appeared to be more effective.
Interventions with in-app notifications were associated with significantly lower treatment outcomes (SMD 0.45) than interventions without (SMD 0.45 vs 0.71).
IN PRACTICE:
“The significant treatment efficacy of app-based interventions compared with active and inactive controls suggests the potential of mobile app interventions as an alternative to conventional psychotherapy, with further merits in accessibility, financial affordability, and safety from stigma,” the authors write.
SOURCE:
The study, with first author Hayoung Bae, BA, with Korea University School of Psychology, Seoul, South Korea, was published online November 20 in JAMA Network Open .
LIMITATIONS:
The findings are based on a small number of trials, with significant heterogeneity among the included trials. The analysis included only English-language publications. Using summary data for the subgroup analyses might have prevented a detailed understanding of the moderating associations of individual participant characteristics.
DISCLOSURES:
The study was supported by a grant from the National Research Foundation funded by the Korean government. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com .
TOPLINE:
A meta-analysis supports the use of mobile mental health apps, both as a standalone and added to conventional treatment, for adults with moderate to severe depression.
METHODOLOGY:
Mobile mental health apps have proliferated but data on their effectiveness in different patient populations is lacking.
To investigate, researchers conducted a systematic review and meta-analysis of 13 randomized clinical trials assessing treatment efficacy of mobile mental health apps in 1470 adults with moderate to severe depression.
The primary outcome was change in depression symptoms from pre- to post-treatment; secondary outcomes included patient-level factors associated with app efficacy.
TAKEAWAY:
Mobile app interventions were associated with significantly reduced depressive symptoms vs both active and inactive control groups, with a medium effect size (standardized mean difference [SMD] 0.50).
App interventions delivered for < 8 weeks had a significantly greater effect size than those delivered for 8+ weeks (SMD 0.77 vs 0.43). Apps were more effective in patients not on medication or in therapy. Apps offering rewards or incentives also appeared to be more effective.
Interventions with in-app notifications were associated with significantly lower treatment outcomes (SMD 0.45) than interventions without (SMD 0.45 vs 0.71).
IN PRACTICE:
“The significant treatment efficacy of app-based interventions compared with active and inactive controls suggests the potential of mobile app interventions as an alternative to conventional psychotherapy, with further merits in accessibility, financial affordability, and safety from stigma,” the authors write.
SOURCE:
The study, with first author Hayoung Bae, BA, with Korea University School of Psychology, Seoul, South Korea, was published online November 20 in JAMA Network Open .
LIMITATIONS:
The findings are based on a small number of trials, with significant heterogeneity among the included trials. The analysis included only English-language publications. Using summary data for the subgroup analyses might have prevented a detailed understanding of the moderating associations of individual participant characteristics.
DISCLOSURES:
The study was supported by a grant from the National Research Foundation funded by the Korean government. The authors report no relevant financial relationships.
A version of this article first appeared on Medscape.com .
Monoamine oxidase inhibitors and tricyclic antidepressants for MDD
Ms. B, age 45, has a history of major depressive disorder (MDD) and migraines. She is admitted after presenting with anhedonia, hopelessness, and hypersomnia. These symptoms have become more severe over the last few weeks. Ms. B describes a past suicide attempt via overdose on doxylamine for which she required treatment in the intensive care unit. The only activity she enjoys is her weekly girls’ night, during which she drinks a few glasses of wine. Ms. B’s current medications are dextromethorphan/bupropion 45/105 mg twice daily and aripiprazole 5 mg/d, which she has taken for 3 months. She states she has “been on every antidepressant there is.”
When clinicians review Ms. B’s medication history, it is clear she has had adequate trials of selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), intranasal esketamine, multiple augmentation strategies, and electroconvulsive therapy (ECT). Ms. B seeks an alternative medication to improve her depressive symptoms.
Treatment-resistant depression (TRD) is commonly defined as depression that has not responded to ≥2 adequate trials of an antidepressant.1 Some guidelines recommend monoamine oxidase inhibitors (MAOIs) and tricyclic antidepressants (TCAs) as second- or even third-line options for MDD,2 while others recommend reserving them for patients with insufficient responses to alternative treatment modalities.3,4 Although MAOIs and TCAs have been available since the 1950s, prescribing these medications has become less prevalent due to safety concerns, the availability of other pharmacologic options, and a lack of clinical training and comfort.5,6 Most research notes that MAOIs are superior for treating atypical depression while TCAs are more effective for melancholic depression.2-4 In a review of 20 studies, Thase et al7 found that 50% of TCA nonresponders benefited from an MAOI. In the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial, monotherapy with the MAOI tranylcypromine was associated with a lower remission rate than the TCA nortriptyline; many argue the dose of tranylcypromine was suboptimal, and few participants completed an adequate trial in the last level.8,9 A more recent study by Kim et al10 found MAOIs to be “generally more effective” than TCAs for TRD, particularly in patients with fewer antidepressant trials; however, this was a small retrospective exploratory trial. A network meta-analysis found both classes to be “competitive” with SSRIs based on efficacy and tolerability, which leads to the question of whether these medications should be considered earlier in therapy.11 Considering patient-specific factors and particular medication properties is an effective strategy when prescribing an MAOI or TCA.
Monoamine oxidase inhibitors
Four MAOIs are FDA-approved for treating MDD (Table 15,12-17): phenelzine, isocarboxazid, tranylcypromine, and selegiline. These medications irreversibly inhibit MAO, which exists as isomers A and B. MAO-A primarily metabolizes serotonin and norepinephrine, which is largely responsible for these medications’ antidepressant effects. Both isomers equally metabolize dopamine.5,12,18 It is best to avoid using MAOIs in patients with cerebrovascular disease, hepatic disease, or pheochromocytoma. Patients with active substance use disorders (particularly sympathomimetics and hallucinogens) are at an increased risk for hypertensive crises and serotonin syndrome, respectively. The most common adverse effects are orthostatic hypotension (despite more well-known concerns regarding hypertension), alterations in sleep patterns (insomnia or hypersomnia, depending on the agent), gastrointestinal issues, and anticholinergic adverse effects such as dry mouth and constipation.13,19-21
In one review and meta-analysis, phenelzine displayed the highest efficacy across all MAOIs.11 It likely requires high doses to achieve adequate MAO inhibition.11 A metabolite of phenelzine inhibits gamma-aminobutyric acid transaminase and may be helpful for patients with comorbid anxiety disorders or MDD with anxious distress.18,21 Additional considerations include phenelzine’s propensity for orthostasis (with rapid titrations and higher doses), sedation, weight gain, sexual dysfunction, and a rare adverse effect of vitamin B6 deficiency.5,13,14,20-22
Use of isocarboxazid in clinical practice is rare. Its adverse effects are similar to those of phenelzine but isocarboxazid is less studied. Tranylcypromine has a similar chemical structure to amphetamine. It can be stimulating at higher doses, potentially benefitting patients with comorbid attention-deficit/hyperactivity disorder (ADHD) or significant apathy.13,23 Selegiline’s distinct quality is its availability as a transdermal patch, which may be useful for patients who struggle to take oral medications. At low doses (6 mg/24 h), the selegiline transdermal patch allows patients to disregard a dietary tyramine restriction because it avoids first-pass metabolism. It inhibits both MAO isomers in the brain but is only selective for MAO-B once concentrations are distributed to the liver. Higher doses require a tyramine-restricted diet because there is still some MAO-A inhibition in the gut. Selegiline is also stimulating because it is converted to amphetamine and methamphetamine.5,12,13,17,19,24
Despite promising results from the use of MAOIs, physicians and patients may be reluctant to use these medications due to perceived limitations. One prominent barrier is the infamous “cheese reaction.” Tyramine, an amino acid found in certain food and beverages (Table 25,13-18,25-28), is broken down by MAO-A in the gut. When this enzyme is inhibited, higher concentrations of tyramine reach systemic circulation. Tyramine’s release of norepinephrine (which now cannot be broken down) can lead to a hypertensive crisis. Consequently, a tyramine-restricted diet is recommended for patients taking an MAOI. However, the common notion that cheese, wine, and beer must be avoided is false, because most of the dietary restrictions developed following the discovery of MAOIs are antiquated.5,12,25-28 Patients who take an MAOI only need to slightly adjust their diet, as outlined in Table 2.5,13-18,25-28 A reasonable serving size of most foods and beverages containing tyramine is unlikely to elicit this “pressor” response. Of the 4 MAOIs FDA-approved for MDD, tranylcypromine appears to be the most sensitive to tyramine.21 Transient postdose hypertension (regardless of tyramine) may occur after taking an MAOI.29 Encourage patients to monitor their blood pressure.
Continue to: Additional hurdles include...
Additional hurdles include the required washout period from serotonergic medications and interactions with sympathomimetics. MAOIs pose the highest risk of serotonin syndrome; however, this usually occurs if given concomitantly with other serotonergic agents. The standard recommendation is a 14-day washout period from SSRIs (5 weeks for fluoxetine and 3 weeks for vortioxetine), SNRIs, mirtazapine, and other antidepressants. It can be distressing for patients to be without medication during that period. Because some antidepressants have much shorter half-lives, waiting 5 half-lives (typically 5 to 7 days) for the discontinued medication to be excreted is feasible if patients are closely monitored.5,12,13,25,27,30 There are rare instances where a TCA may be combined with an MAOI (typically initiated within 1 to 2 days of each other), but never clomipramine or imipramine due to their potent serotonin reuptake inhibition.31 If switching to an alternative MAOI, waiting 7 to 14 days is recommended to allow adequate time for the inhibited enzyme to regenerate.14-17,32 Taking medications that increase dopamine and norepinephrine (eg, stimulants or oral over-the-counter decongestants) with an MAOI is typically not recommended due to the risk of hypertensive crisis.25,27 In severe TRD or comorbid ADHD, successful simultaneous use of methylphenidate or amphetamine—typically at low doses—with close blood pressure monitoring has been reported.33 There have also been positive cases of the use of modafinil in combination with an MAOI; however, this should be done with caution.34,35 Clinicians must use clinical judgment when considering a combination of medications that pose a higher risk.
Tricyclic antidepressants
TCAs work differently than MAOIs to increase monoamines. They inhibit presynaptic serotonin and norepinephrine transporters in the CNS to increase levels of these chemicals in the synaptic cleft. While all TCAs inhibit these transporters, they do so at varying levels (Table 336-51). Based on their chemical structure, TCAs can be categorized into secondary and tertiary amines. Tertiary amines are metabolized via demethylation into their derivatives (Table 336-51). Patients who have recently suffered a myocardial infarction (MI) should avoid tertiary amines. TCAs can reduce heart rate variability, which is already decreased after an MI, thus presenting the potential for cardiac arrhythmias. TCAs should also be avoided in patients with cardiac conduction abnormalities.38-46,52 Patients with a prior baseline cardiac conduction defect, such as a bundle branch block, are at higher risk for further cardiac abnormalities. In those with a preexisting first-degree heart block, TCAs can still be used, but electrocardiogram monitoring is recommended.52,53 TCAs have also been reported to decrease the seizure threshold.38-46 They can be used with caution in patients who have a history of epilepsy or head trauma, or with concomitant medications that lower the seizure threshold.38-46
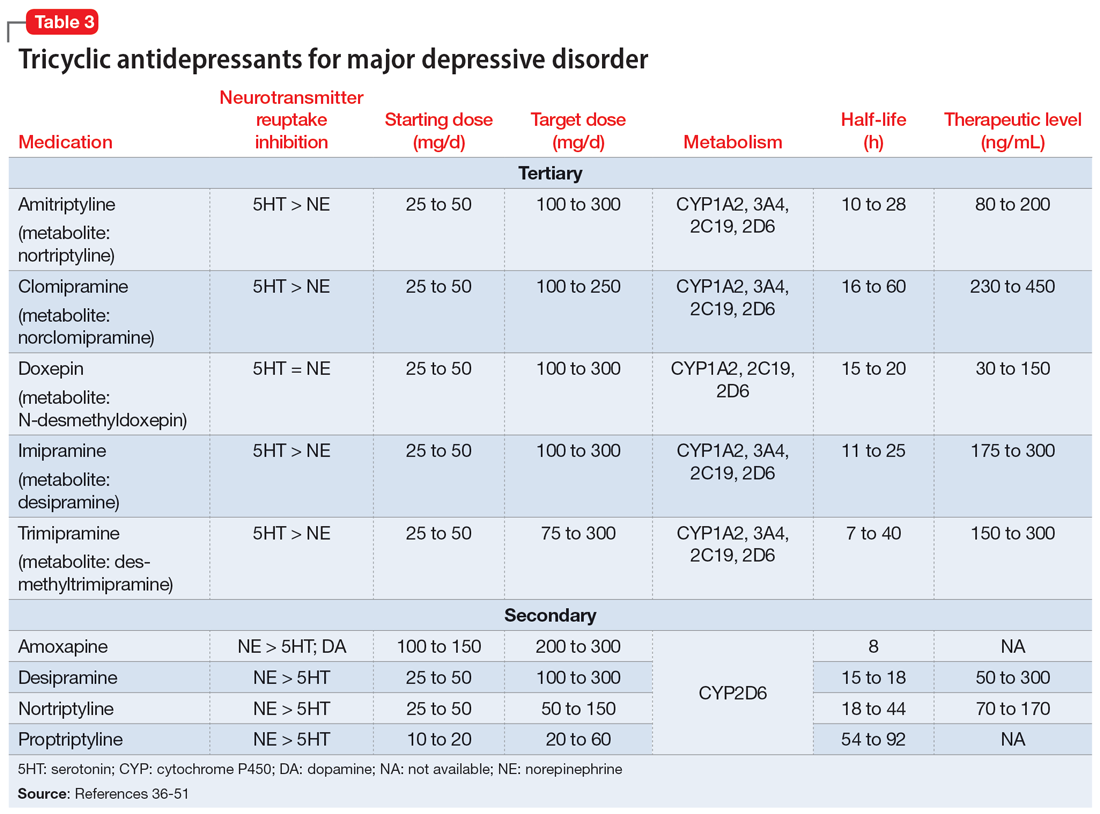
Overdose risk is a concern with TCAs because ingestion of 10 to 20 mg/kg can lead to significant toxicity.54 This is due to their blockage of voltage-gated sodium channels found in the CNS and heart, which contributes to overdose symptoms such as a widened QRS complex and seizures. Symptoms usually develop within 2 hours but may be delayed up to 6 hours.55 Patients with a history of overdose must be carefully assessed before initiating a TCA. Prescribing a limited supply of these medications may be valuable. The use of TCAs has often been limited due to their adverse effects, most of which are associated with their respective affinities for alpha 1, muscarinic 1, and histamine 1 receptors. Inhibition of the alpha 1 receptor is associated with hypotension, muscarinic 1 with anticholinergic adverse effects, and histamine 1 with sedation and weight gain. Tertiary amines have a higher affinity for these receptors compared to secondary amines, leading to a more significant adverse effect profile.36,50 Among TCAs, amitriptyline is the most likely to cause hypotension, whereas desipramine and nortriptyline are least likely. Amitriptyline and clomipramine are most likely to cause anticholinergic adverse effects, whereas desipramine and nortriptyline are the least likely. Amitriptyline, doxepin, and imipramine have the highest propensity for QTc prolongation.36
Beyond treating MDD, TCAs have shown benefits for treating other disease states (Table 438-46,49,56-61).These differing indications may help psychiatrists determine the best TCA to prescribe for a given patient. Amitriptyline is the most studied TCA for MDD; however, nortriptyline is typically preferred due to its favorable tolerability profile.4,62 Nortriptyline also has data supporting its use in ECT to prevent relapse.63 Amitriptyline and nortriptyline have shown benefits in patients with neuropathic pain and for migraine prophylaxis.56-60 Although frequently used for MDD, clomipramine is not FDA-approved for this indication, but is for obsessive-compulsive disorder.39 Doxepin is FDA-approved for insomnia at lower doses and for MDD at higher doses.40 Therefore, it may benefit patients with sleep difficulties secondary to depression. Desipramine has been used off-label to treat ADHD in children and has shown some benefits in adults.64-66 Protriptyline, trimipramine, and amoxapine are infrequently used in clinical practice.
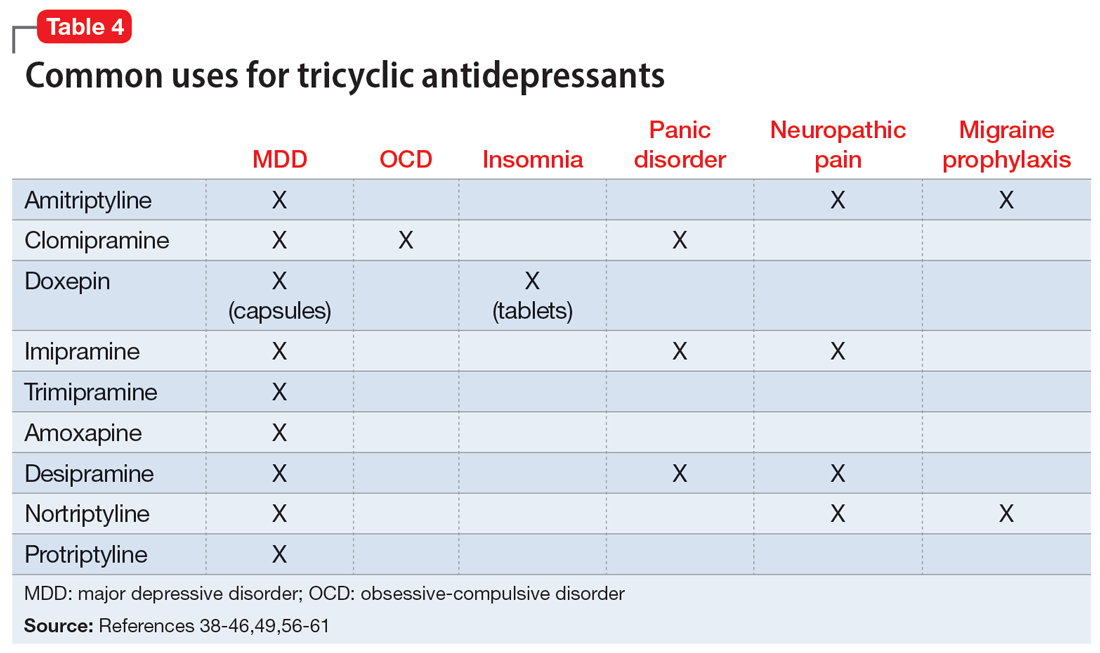
A unique feature of TCAs is the ability to monitor serum concentrations (Table 336-51).Guidelines recommend therapeutic drug monitoring (TDM) with amitriptyline, clomipramine, imipramine, and nortriptyline for routine use. TDM is still recommended for doxepin, desipramine, and trimipramine, but its utility is largely for treatment failure or resistance.37 These plasma levels can be altered based on coadministered medications (Table 538-46) and should be closely monitored. Physicians should obtain a trough level after at least 5 half-lives and before the next dose is due, and use TDM as indicated to optimize dosing.

Continue to: CASE CONTINUED
CASE CONTINUED
Ms. B’s outpatient psychiatrist provides collateral information about her medical history and confirms her long-standing MDD with multiple medication trials, though she has never received an MAOI or TCA. Ms. B is adamant she does not want a medication-free period between treatments and refuses to adjust her diet, despite being educated on the few changes necessary. She has no contraindications for TCAs and may benefit from a TCA for her comorbid migraines. The care team expresses concern for TCA overdose to Ms. B and her family. Ms. B’s sister reassures the team they will have someone monitor and dispense her medications at home. They decide to discontinue her current psychiatric regimen, and Ms. B is started on nortriptyline 50 mg/d at night, with plans to titrate based on tolerability.
Related Resources
- Meyer JM. A concise guide to monoamine oxidase inhibitors. Current Psychiatry. 2017;16(12):14-16,18-23,47,A.
- Espejo GD. Treating major depressive disorder after limited response to an initial agent. Current Psychiatry. 2021;20(10):51-53. doi:10.12788/cp.0178
- American Association of Psychiatric Pharmacists (AAPP) MAOI Pharmacist Toolkit. https://aapp.org/guideline/maoi
Drug Brand Names
Amitriptyline • Elavil
Amphetamine • Adzenys, Dyanavel
Aripiprazole • Abilify
Clomipramine • Anafranil
Desipramine • Norpramin
Dextromethorphan/bupropion • Auvelity
Doxepin • Sinequan, Adapin
Esketamine • Spravato
Fluoxetine • Prozac
Imipramine • Tofranil
Isocarboxazid • Marplan
Methamphetamine • Desoxyn
Mirtazapine • Remeron
Modafinil • Provigil
Nortriptyline • Pamelor
Phenelzine • Nardil
Protriptyline • Vivactil
Selegiline • Emsam
Tranylcypromine • Parnate
Trimipramine • Surmontil
Vortioxetine • Trintellix
1. Gaynes BN, Lux L, Gartlehner G, et al. Defining treatment-resistant depression. Depress Anxiety. 2020;37(2):134-145. doi:10.1002/da.22968
2. Kennedy SH, Lam RW, McIntyre RS, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 clinical guidelines for the management of adults with major depressive disorder: section 3. Pharmacological treatments. Can J Psychiatry. 2016;61(9):540-560. doi:10.1177/0706743716659417
3. VA/DoD clinical practice guideline for the management of major depressive disorder. Veterans Health Administration and Department of Defense; 2016. https://www.healthquality.va.gov/guidelines/MH/mdd/VADoDMDDCPGFINAL82916.pdf
4. Gelenberg AJ, Freeman MP, Markowitz JC, et al. Practice guideline for the treatment of patients with major depressive disorder. Am J Psychiatry. 2010;167(Suppl 10):9-118.
5. Meyer JM. A concise guide to monoamine oxidase inhibitors. Current Psychiatry. 2017;16(12):14-16,18-23,47,A.
6. Taylor D. Selective serotonin reuptake inhibitors and tricyclic antidepressants in combination. Interactions and therapeutic uses. Br J Psychiatry. 1995;167(5):575-580. doi:10.1192/bjp.167.5.575
7. Thase ME, Trivedi MH, Rush AJ. MAOIs in the contemporary treatment of depression. Neuropsychopharmacology. 1995;12(3):185-219. doi:10.1016/0893-133X(94)00058-8
8. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905-1917. doi:10.1176/ajp.2006.163.11.1905
9. McGrath PJ, Stewart JW, Fava M, et al. Tranylcypromine versus venlafaxine plus mirtazapine following three failed antidepressant medication trials for depression: a STAR*D report. Am J Psychiatry. 2006;163(9):1531-1666. doi:10.1176/ajp.2006.163.9.1531
10. Kim T, Xu C, Amsterdam JD. Relative effectiveness of tricyclic antidepressant versus monoamine oxidase inhibitor monotherapy for treatment-resistant depression. J Affect Disord. 2019;250:199-203. doi:10.1016/j.jad.2019.03.028
11. Suchting R, Tirumalajaru V, Gareeb R, et al. Revisiting monoamine oxidase inhibitors for the treatment of depressive disorders: a systematic review and network meta-analysis. J Affect Disord. 2021;282:1153-1160. doi:10.1016/j.jad.2021.01.021
12. Stahl SM, Felker A. Monoamine oxidase inhibitors: a modern guide to an unrequited class of antidepressants. CNS Spectr. 2008;13(10):855-870. doi:10.1017/s1092852900016965
13. Chamberlain SR, Baldwin DS. Monoamine oxidase inhibitors (MAOIs) in psychiatric practice: how to use them safely and effectively. CNS Drugs. 2021;35(7):703-716. doi:10.1007/s40263-021-00832-x
14. Nardil [package insert]. New York, NY: Parke-Davis; 2009.
15. Marplan [package insert]. Parsippany, NJ: Validus Pharmaceuticals LLC; 2020.
16. Parnate [package insert]. Saint Michael, Barbados: Concordia Pharmaceuticals; 2015.
17. Emsam [package insert]. Morgantown, WV: Mylan Specialty LP; 2014.
18. Shulman KI, Herrmann N, Walker SE. Current place of monoamine oxidase inhibitors in the treatment of depression. CNS Drugs. 2013;27(10):789-797. doi:10.1007/s40263-013-0097-3
19. Sub Laban T, Saadabadi A. Monoamine oxidase inhibitors (MAOI). StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK539848/
20. Rabkin JG, Quitkin FM, McGrath P, et al. Adverse reactions to monoamine oxidase inhibitors. Part II. Treatment correlates and clinical management. J Clin Psychopharmacol. 1985;5(1):2-9.
21. Gillman PK. Advances pertaining to the pharmacology and interactions of irreversible nonselective monoamine oxidase inhibitors. J Clin Psychopharmacol. 2011;31(1):66-74. doi:10.1097/JCP.0b013e31820469ea
22. Sidhu G, Marwaha R. Phenelzine. StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK554508/
23. Frieling H, Bleich S. Tranylcypromine: new perspectives on an “old” drug. Eur Arch Psychiatry Clin Neurosci. 2006;256(5):268-273. doi:10.1007/s00406-006-0660-8
24. Goodnick PJ. Seligiline transdermal system in depression. Expert Opin Pharmacother. 2007;8(1):59-64. doi:10.1517/14656566.8.1.59
25. Edinoff AN, Swinford CR, Odisho AS, et al. Clinically relevant drug interactions with monoamine oxidase inhibitors. Health Psychol Res. 2022;10(4):39576. doi:10.52965/001c.39576
26. Gillman PK. A reassessment of the safety profile of monoamine oxidase inhibitors: elucidating tired old tyramine myths. J Neural Transm (Vienna). 2018;125(11):1707-1717. doi:10.1007/s00702-018-1932-y
27. Flockhart DA. Dietary restrictions and drug interactions with monoamine oxidase inhibitors: an update. J Clin Psychiatry. 2012;73 Suppl 1:17-24. doi:10.4088/JCP.11096su1c.03
28. McCabe-Sellers BJ, Staggs CG, Bogle ML. Tyramine in foods and monoamine oxidase inhibitor drugs: a crossroad where medicine, nutrition, pharmacy, and food industry converge. J Food Composit Anal. 2006;19:S58-S65. doi:10.1016/j.jfca.2005.12.008
29. Keck PE Jr, Vuckovic A, Pope HG Jr, et al. Acute cardiovascular response to monoamine oxidase inhibitors: a prospective assessment. J Clin Psychopharmacol. 1989;9(3):203-206.
30. Bodkin JA, Dunlop BW. Moving on with monoamine oxidase inhibitors. Focus (Am Psychiatr Publ). 2021;19(1):50-52. doi:10.1176/appi.focus.20200046
31. Amsterdam JD, Kim TT. Relative effectiveness of monoamine oxidase inhibitor and tricyclic antidepressant combination therapy for treatment-resistant depression. J Clin Psychopharmacol. 2019;39(6):649-652. doi:10.1097/JCP.0000000000001130
32. Keks N, Hope J, Keogh S. Switching and stopping antidepressants. Aust Prescr. 2016;39(3):76-83. doi:10.18773/austprescr.2016.039
33. Israel JA. Combining stimulants and monoamine oxidase inhibitors: a reexamination of the literature and a report of a new treatment combination. Prim Care Companion CNS Disord. 2015;17(6):10.4088/PCC.15br01836. doi:10.4088/PCC.15br01836
34. Clemons WE, Makela E, Young J. Concomitant use of modafinil and tranylcypromine in a patient with narcolepsy: a case report. Sleep Med. 2004;5(5):509-511. doi:10.1016/j.sleep.2004.06.006
35. Ashton AK. Modafinil augmentation of phenelzine for residual fatigue in dysthymia. Am J Psychiatry. 2004;161(9):1716-1717. doi:10.1176/appi.ajp.161.9.1716-a
36. O’Donnell JM, Bies RR, Shelton RC. Drug therapy of depression and anxiety disorders. In: Brunton LL, Hilal-Dandan R, Knollmann BC, eds. Goodman & Gilman’s: The Pharmacological Basis of Therapeutics. 13th ed. McGraw Hill; 2017. Accessed June 4, 2023. https://accessanesthesiology.mhmedical.com/content.aspx?bookid=2189§ionid=169518711
37. Hiemke C, Bergemann N, Clement HW, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51(1-02):9-62. doi:10.1055/s-0043-116492
38. Amitriptyline hydrochloride [package insert]. East Brunswick, NJ: Unichem Pharmaceuticals (USA); 2021.
39. Clomipramine hydrochloride [package insert]. East Windsor, NJ: Aurobindo Pharma Limited; 2023.
40. Doxepin hydrochloride capsules, USP [package insert]. Bedminster, NJ: Alembic Pharmaceuticals Inc; 2021.
41. Imipramine hydrochloride tablet [package insert]. Fairfield, NJ: Leading Pharma LLC USA; 2022.
42. Trimipramine maleate [package insert]. Northvale, NJ: Elite Laboratories Inc; 2021.
43. Amoxapine [package insert]. Parsippany, NJ: Actavis Pharma Inc; 2015.
44. Desipramine hydrochloride tablets [package insert]. Bedminster, NJ: Alembic Pharmaceuticals Inc; 2023.
45. Nortriptyline hydrochloride capsules, USP [package insert]. Parsippany, NJ: Teva Pharmaceuticals Inc; 2021.
46. Protriptyline hydrochloride [package insert]. Bensalem, PA: Sigmapharm Laboratories, LLC; 2023.
47. Calvo B, García MJ, Pedraz JL, et al. Pharmacokinetics of amoxapine and its active metabolites. Int J Clin Pharmacol Ther Toxicol. 1985;23(4):180-185.
48. Ziegler VE, Biggs JT, Wylie LT, et al. Protriptyline kinetics. Clin Pharmacol Ther. 1978;23(5):580-584. doi:10.1002/cpt1978235580
49. Cleare A, Pariante CM, Young AH, et al. Evidence-based guidelines for treating depressive disorders with antidepressants: a revision of the 2008 British Association for Psychopharmacology guidelines. J Psychopharmacol. 2015;29(5):459-525. doi:10.1177/0269881115581093
50. Richelson E. Synaptic effects of antidepressants. J Clin Psychopharmacol. 1996;16(3 Suppl 2):1S-9S. doi:10.1097/00004714-199606002-00001
51. Vos CF, Aarnoutse RE, Op de Coul MJM, et al. Tricyclic antidepressants for major depressive disorder: a comprehensive evaluation of current practice in the Netherlands. BMC Psychiatry. 2021;21(1):481. doi:10.1186/s12888-021-03490-x
52. Alvarez W Jr, Pickworth KK. Safety of antidepressant drugs in the patient with cardiac disease: a review of the literature. Pharmacotherapy. 2003;23(6):754-771. doi:10.1592/phco.23.6.754.32185
53. Dietch JT, Fine M. The effect of nortriptyline in elderly patients with cardiac conduction disease. J Clin Psychiatry. 1990;51(2):65-67.
54. Valento M, Liebelt EL. Cyclic antidepressants. In: Nelson LS, Howland M, Lewin NA, et al, eds. Goldfrank’s Toxicologic Emergencies. 9th ed. McGraw Hill; 2011. Accessed June 10, 2023. https://accesspharmacy.mhmedical.com/content.aspx?bookid=2569§ionid=210274664
55. Woolf AD, Erdman AR, Nelson LS, et al. Tricyclic antidepressant poisoning: an evidence-based consensus guideline for out-of-hospital management. Clin Toxicol (Phila). 2007;45(3):203-233. doi:10.1080/15563650701226192
56. Baldwin DS, Anderson IM, Nutt DJ, et al. Evidence-based pharmacological treatment of anxiety disorders, post-traumatic stress disorder and obsessive-compulsive disorder: a revision of the 2005 guidelines from the British Association for Psychopharmacology. J Psychopharmacol. 2014;28(5):403-439. doi:10.1177/0269881114525674
57. Moulin DE, Clark AJ, Gilron I, et al. Pharmacological management of chronic neuropathic pain - consensus statement and guidelines from the Canadian Pain Society. Pain Res Manag. 2007;12(1):13-21. doi:10.1155/2007/730785
58. Finnerup NB, Attal N, Haroutounian S, et al. Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol. 2015;14(2):162-173. doi:10.1016/S1474-4422(14)70251-0
59. Saarto T, Wiffen PJ. Antidepressants for neuropathic pain. Cochrane Database Syst Rev. 2007;(4):CD005454. doi:10.1002/14651858.CD005454.pub2
60. Burch R. Antidepressants for preventive treatment of migraine. Curr Treat Options Neurol. 2019;21(4):18. doi:10.1007/s11940-019-0557-2
61. Ailani J, Burch RC, Robbins MS; Board of Directors of the American Headache Society. The American Headache Society consensus statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61(7):1021-1039. doi:10.1111/head.14153
62. Bauer M, Pfennig A, Severus E, et al. World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for biological treatment of unipolar depressive disorders, part 1: update 2013 on the acute and continuation treatment of unipolar depressive disorders. World J Biol Psychiatry. 2013;14(5):334-385. doi:10.3109/15622975.2013.804195
63. Jelovac A, Kolshus E, McLoughlin DM. Relapse following successful electroconvulsive therapy for major depression: a meta-analysis. Neuropsychopharmacology. 2013;38(12):2467-2474. doi:10.1038/npp.2013.149
64. Spencer T, Biederman J, Coffey B, et al. A double-blind comparison of desipramine and placebo in children and adolescents with chronic tic disorder and comorbid attention-deficit/hyperactivity disorder. Arch Gen Psychiatry. 2002;59(7):649-656. doi:10.1001/archpsyc.59.7.649
65. Spencer T, Biederman J, Wilens T, et al. Pharmacotherapy of attention-deficit hyperactivity disorder across the life cycle. J Am Acad Child Adolesc Psychiatry. 1996;35(4):409-432. doi:10.1097/00004583-199604000-00008
66. Wilens TE, Biederman J, Prince J, et al. Six-week, double-blind, placebo-controlled study of desipramine for adult attention deficit hyperactivity disorder. Am J Psychiatry. 1996;153(9):1147-1153. doi:10.1176/ajp.153.9.1147
Ms. B, age 45, has a history of major depressive disorder (MDD) and migraines. She is admitted after presenting with anhedonia, hopelessness, and hypersomnia. These symptoms have become more severe over the last few weeks. Ms. B describes a past suicide attempt via overdose on doxylamine for which she required treatment in the intensive care unit. The only activity she enjoys is her weekly girls’ night, during which she drinks a few glasses of wine. Ms. B’s current medications are dextromethorphan/bupropion 45/105 mg twice daily and aripiprazole 5 mg/d, which she has taken for 3 months. She states she has “been on every antidepressant there is.”
When clinicians review Ms. B’s medication history, it is clear she has had adequate trials of selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), intranasal esketamine, multiple augmentation strategies, and electroconvulsive therapy (ECT). Ms. B seeks an alternative medication to improve her depressive symptoms.
Treatment-resistant depression (TRD) is commonly defined as depression that has not responded to ≥2 adequate trials of an antidepressant.1 Some guidelines recommend monoamine oxidase inhibitors (MAOIs) and tricyclic antidepressants (TCAs) as second- or even third-line options for MDD,2 while others recommend reserving them for patients with insufficient responses to alternative treatment modalities.3,4 Although MAOIs and TCAs have been available since the 1950s, prescribing these medications has become less prevalent due to safety concerns, the availability of other pharmacologic options, and a lack of clinical training and comfort.5,6 Most research notes that MAOIs are superior for treating atypical depression while TCAs are more effective for melancholic depression.2-4 In a review of 20 studies, Thase et al7 found that 50% of TCA nonresponders benefited from an MAOI. In the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial, monotherapy with the MAOI tranylcypromine was associated with a lower remission rate than the TCA nortriptyline; many argue the dose of tranylcypromine was suboptimal, and few participants completed an adequate trial in the last level.8,9 A more recent study by Kim et al10 found MAOIs to be “generally more effective” than TCAs for TRD, particularly in patients with fewer antidepressant trials; however, this was a small retrospective exploratory trial. A network meta-analysis found both classes to be “competitive” with SSRIs based on efficacy and tolerability, which leads to the question of whether these medications should be considered earlier in therapy.11 Considering patient-specific factors and particular medication properties is an effective strategy when prescribing an MAOI or TCA.
Monoamine oxidase inhibitors
Four MAOIs are FDA-approved for treating MDD (Table 15,12-17): phenelzine, isocarboxazid, tranylcypromine, and selegiline. These medications irreversibly inhibit MAO, which exists as isomers A and B. MAO-A primarily metabolizes serotonin and norepinephrine, which is largely responsible for these medications’ antidepressant effects. Both isomers equally metabolize dopamine.5,12,18 It is best to avoid using MAOIs in patients with cerebrovascular disease, hepatic disease, or pheochromocytoma. Patients with active substance use disorders (particularly sympathomimetics and hallucinogens) are at an increased risk for hypertensive crises and serotonin syndrome, respectively. The most common adverse effects are orthostatic hypotension (despite more well-known concerns regarding hypertension), alterations in sleep patterns (insomnia or hypersomnia, depending on the agent), gastrointestinal issues, and anticholinergic adverse effects such as dry mouth and constipation.13,19-21

In one review and meta-analysis, phenelzine displayed the highest efficacy across all MAOIs.11 It likely requires high doses to achieve adequate MAO inhibition.11 A metabolite of phenelzine inhibits gamma-aminobutyric acid transaminase and may be helpful for patients with comorbid anxiety disorders or MDD with anxious distress.18,21 Additional considerations include phenelzine’s propensity for orthostasis (with rapid titrations and higher doses), sedation, weight gain, sexual dysfunction, and a rare adverse effect of vitamin B6 deficiency.5,13,14,20-22
Use of isocarboxazid in clinical practice is rare. Its adverse effects are similar to those of phenelzine but isocarboxazid is less studied. Tranylcypromine has a similar chemical structure to amphetamine. It can be stimulating at higher doses, potentially benefitting patients with comorbid attention-deficit/hyperactivity disorder (ADHD) or significant apathy.13,23 Selegiline’s distinct quality is its availability as a transdermal patch, which may be useful for patients who struggle to take oral medications. At low doses (6 mg/24 h), the selegiline transdermal patch allows patients to disregard a dietary tyramine restriction because it avoids first-pass metabolism. It inhibits both MAO isomers in the brain but is only selective for MAO-B once concentrations are distributed to the liver. Higher doses require a tyramine-restricted diet because there is still some MAO-A inhibition in the gut. Selegiline is also stimulating because it is converted to amphetamine and methamphetamine.5,12,13,17,19,24
Despite promising results from the use of MAOIs, physicians and patients may be reluctant to use these medications due to perceived limitations. One prominent barrier is the infamous “cheese reaction.” Tyramine, an amino acid found in certain food and beverages (Table 25,13-18,25-28), is broken down by MAO-A in the gut. When this enzyme is inhibited, higher concentrations of tyramine reach systemic circulation. Tyramine’s release of norepinephrine (which now cannot be broken down) can lead to a hypertensive crisis. Consequently, a tyramine-restricted diet is recommended for patients taking an MAOI. However, the common notion that cheese, wine, and beer must be avoided is false, because most of the dietary restrictions developed following the discovery of MAOIs are antiquated.5,12,25-28 Patients who take an MAOI only need to slightly adjust their diet, as outlined in Table 2.5,13-18,25-28 A reasonable serving size of most foods and beverages containing tyramine is unlikely to elicit this “pressor” response. Of the 4 MAOIs FDA-approved for MDD, tranylcypromine appears to be the most sensitive to tyramine.21 Transient postdose hypertension (regardless of tyramine) may occur after taking an MAOI.29 Encourage patients to monitor their blood pressure.

Continue to: Additional hurdles include...
Additional hurdles include the required washout period from serotonergic medications and interactions with sympathomimetics. MAOIs pose the highest risk of serotonin syndrome; however, this usually occurs if given concomitantly with other serotonergic agents. The standard recommendation is a 14-day washout period from SSRIs (5 weeks for fluoxetine and 3 weeks for vortioxetine), SNRIs, mirtazapine, and other antidepressants. It can be distressing for patients to be without medication during that period. Because some antidepressants have much shorter half-lives, waiting 5 half-lives (typically 5 to 7 days) for the discontinued medication to be excreted is feasible if patients are closely monitored.5,12,13,25,27,30 There are rare instances where a TCA may be combined with an MAOI (typically initiated within 1 to 2 days of each other), but never clomipramine or imipramine due to their potent serotonin reuptake inhibition.31 If switching to an alternative MAOI, waiting 7 to 14 days is recommended to allow adequate time for the inhibited enzyme to regenerate.14-17,32 Taking medications that increase dopamine and norepinephrine (eg, stimulants or oral over-the-counter decongestants) with an MAOI is typically not recommended due to the risk of hypertensive crisis.25,27 In severe TRD or comorbid ADHD, successful simultaneous use of methylphenidate or amphetamine—typically at low doses—with close blood pressure monitoring has been reported.33 There have also been positive cases of the use of modafinil in combination with an MAOI; however, this should be done with caution.34,35 Clinicians must use clinical judgment when considering a combination of medications that pose a higher risk.
Tricyclic antidepressants
TCAs work differently than MAOIs to increase monoamines. They inhibit presynaptic serotonin and norepinephrine transporters in the CNS to increase levels of these chemicals in the synaptic cleft. While all TCAs inhibit these transporters, they do so at varying levels (Table 336-51). Based on their chemical structure, TCAs can be categorized into secondary and tertiary amines. Tertiary amines are metabolized via demethylation into their derivatives (Table 336-51). Patients who have recently suffered a myocardial infarction (MI) should avoid tertiary amines. TCAs can reduce heart rate variability, which is already decreased after an MI, thus presenting the potential for cardiac arrhythmias. TCAs should also be avoided in patients with cardiac conduction abnormalities.38-46,52 Patients with a prior baseline cardiac conduction defect, such as a bundle branch block, are at higher risk for further cardiac abnormalities. In those with a preexisting first-degree heart block, TCAs can still be used, but electrocardiogram monitoring is recommended.52,53 TCAs have also been reported to decrease the seizure threshold.38-46 They can be used with caution in patients who have a history of epilepsy or head trauma, or with concomitant medications that lower the seizure threshold.38-46

Overdose risk is a concern with TCAs because ingestion of 10 to 20 mg/kg can lead to significant toxicity.54 This is due to their blockage of voltage-gated sodium channels found in the CNS and heart, which contributes to overdose symptoms such as a widened QRS complex and seizures. Symptoms usually develop within 2 hours but may be delayed up to 6 hours.55 Patients with a history of overdose must be carefully assessed before initiating a TCA. Prescribing a limited supply of these medications may be valuable. The use of TCAs has often been limited due to their adverse effects, most of which are associated with their respective affinities for alpha 1, muscarinic 1, and histamine 1 receptors. Inhibition of the alpha 1 receptor is associated with hypotension, muscarinic 1 with anticholinergic adverse effects, and histamine 1 with sedation and weight gain. Tertiary amines have a higher affinity for these receptors compared to secondary amines, leading to a more significant adverse effect profile.36,50 Among TCAs, amitriptyline is the most likely to cause hypotension, whereas desipramine and nortriptyline are least likely. Amitriptyline and clomipramine are most likely to cause anticholinergic adverse effects, whereas desipramine and nortriptyline are the least likely. Amitriptyline, doxepin, and imipramine have the highest propensity for QTc prolongation.36
Beyond treating MDD, TCAs have shown benefits for treating other disease states (Table 438-46,49,56-61).These differing indications may help psychiatrists determine the best TCA to prescribe for a given patient. Amitriptyline is the most studied TCA for MDD; however, nortriptyline is typically preferred due to its favorable tolerability profile.4,62 Nortriptyline also has data supporting its use in ECT to prevent relapse.63 Amitriptyline and nortriptyline have shown benefits in patients with neuropathic pain and for migraine prophylaxis.56-60 Although frequently used for MDD, clomipramine is not FDA-approved for this indication, but is for obsessive-compulsive disorder.39 Doxepin is FDA-approved for insomnia at lower doses and for MDD at higher doses.40 Therefore, it may benefit patients with sleep difficulties secondary to depression. Desipramine has been used off-label to treat ADHD in children and has shown some benefits in adults.64-66 Protriptyline, trimipramine, and amoxapine are infrequently used in clinical practice.

A unique feature of TCAs is the ability to monitor serum concentrations (Table 336-51).Guidelines recommend therapeutic drug monitoring (TDM) with amitriptyline, clomipramine, imipramine, and nortriptyline for routine use. TDM is still recommended for doxepin, desipramine, and trimipramine, but its utility is largely for treatment failure or resistance.37 These plasma levels can be altered based on coadministered medications (Table 538-46) and should be closely monitored. Physicians should obtain a trough level after at least 5 half-lives and before the next dose is due, and use TDM as indicated to optimize dosing.

Continue to: CASE CONTINUED
CASE CONTINUED
Ms. B’s outpatient psychiatrist provides collateral information about her medical history and confirms her long-standing MDD with multiple medication trials, though she has never received an MAOI or TCA. Ms. B is adamant she does not want a medication-free period between treatments and refuses to adjust her diet, despite being educated on the few changes necessary. She has no contraindications for TCAs and may benefit from a TCA for her comorbid migraines. The care team expresses concern for TCA overdose to Ms. B and her family. Ms. B’s sister reassures the team they will have someone monitor and dispense her medications at home. They decide to discontinue her current psychiatric regimen, and Ms. B is started on nortriptyline 50 mg/d at night, with plans to titrate based on tolerability.
Related Resources
- Meyer JM. A concise guide to monoamine oxidase inhibitors. Current Psychiatry. 2017;16(12):14-16,18-23,47,A.
- Espejo GD. Treating major depressive disorder after limited response to an initial agent. Current Psychiatry. 2021;20(10):51-53. doi:10.12788/cp.0178
- American Association of Psychiatric Pharmacists (AAPP) MAOI Pharmacist Toolkit. https://aapp.org/guideline/maoi
Drug Brand Names
Amitriptyline • Elavil
Amphetamine • Adzenys, Dyanavel
Aripiprazole • Abilify
Clomipramine • Anafranil
Desipramine • Norpramin
Dextromethorphan/bupropion • Auvelity
Doxepin • Sinequan, Adapin
Esketamine • Spravato
Fluoxetine • Prozac
Imipramine • Tofranil
Isocarboxazid • Marplan
Methamphetamine • Desoxyn
Mirtazapine • Remeron
Modafinil • Provigil
Nortriptyline • Pamelor
Phenelzine • Nardil
Protriptyline • Vivactil
Selegiline • Emsam
Tranylcypromine • Parnate
Trimipramine • Surmontil
Vortioxetine • Trintellix
Ms. B, age 45, has a history of major depressive disorder (MDD) and migraines. She is admitted after presenting with anhedonia, hopelessness, and hypersomnia. These symptoms have become more severe over the last few weeks. Ms. B describes a past suicide attempt via overdose on doxylamine for which she required treatment in the intensive care unit. The only activity she enjoys is her weekly girls’ night, during which she drinks a few glasses of wine. Ms. B’s current medications are dextromethorphan/bupropion 45/105 mg twice daily and aripiprazole 5 mg/d, which she has taken for 3 months. She states she has “been on every antidepressant there is.”
When clinicians review Ms. B’s medication history, it is clear she has had adequate trials of selective serotonin reuptake inhibitors (SSRIs), serotonin-norepinephrine reuptake inhibitors (SNRIs), intranasal esketamine, multiple augmentation strategies, and electroconvulsive therapy (ECT). Ms. B seeks an alternative medication to improve her depressive symptoms.
Treatment-resistant depression (TRD) is commonly defined as depression that has not responded to ≥2 adequate trials of an antidepressant.1 Some guidelines recommend monoamine oxidase inhibitors (MAOIs) and tricyclic antidepressants (TCAs) as second- or even third-line options for MDD,2 while others recommend reserving them for patients with insufficient responses to alternative treatment modalities.3,4 Although MAOIs and TCAs have been available since the 1950s, prescribing these medications has become less prevalent due to safety concerns, the availability of other pharmacologic options, and a lack of clinical training and comfort.5,6 Most research notes that MAOIs are superior for treating atypical depression while TCAs are more effective for melancholic depression.2-4 In a review of 20 studies, Thase et al7 found that 50% of TCA nonresponders benefited from an MAOI. In the Sequenced Treatment Alternatives to Relieve Depression (STAR*D) trial, monotherapy with the MAOI tranylcypromine was associated with a lower remission rate than the TCA nortriptyline; many argue the dose of tranylcypromine was suboptimal, and few participants completed an adequate trial in the last level.8,9 A more recent study by Kim et al10 found MAOIs to be “generally more effective” than TCAs for TRD, particularly in patients with fewer antidepressant trials; however, this was a small retrospective exploratory trial. A network meta-analysis found both classes to be “competitive” with SSRIs based on efficacy and tolerability, which leads to the question of whether these medications should be considered earlier in therapy.11 Considering patient-specific factors and particular medication properties is an effective strategy when prescribing an MAOI or TCA.
Monoamine oxidase inhibitors
Four MAOIs are FDA-approved for treating MDD (Table 15,12-17): phenelzine, isocarboxazid, tranylcypromine, and selegiline. These medications irreversibly inhibit MAO, which exists as isomers A and B. MAO-A primarily metabolizes serotonin and norepinephrine, which is largely responsible for these medications’ antidepressant effects. Both isomers equally metabolize dopamine.5,12,18 It is best to avoid using MAOIs in patients with cerebrovascular disease, hepatic disease, or pheochromocytoma. Patients with active substance use disorders (particularly sympathomimetics and hallucinogens) are at an increased risk for hypertensive crises and serotonin syndrome, respectively. The most common adverse effects are orthostatic hypotension (despite more well-known concerns regarding hypertension), alterations in sleep patterns (insomnia or hypersomnia, depending on the agent), gastrointestinal issues, and anticholinergic adverse effects such as dry mouth and constipation.13,19-21

In one review and meta-analysis, phenelzine displayed the highest efficacy across all MAOIs.11 It likely requires high doses to achieve adequate MAO inhibition.11 A metabolite of phenelzine inhibits gamma-aminobutyric acid transaminase and may be helpful for patients with comorbid anxiety disorders or MDD with anxious distress.18,21 Additional considerations include phenelzine’s propensity for orthostasis (with rapid titrations and higher doses), sedation, weight gain, sexual dysfunction, and a rare adverse effect of vitamin B6 deficiency.5,13,14,20-22
Use of isocarboxazid in clinical practice is rare. Its adverse effects are similar to those of phenelzine but isocarboxazid is less studied. Tranylcypromine has a similar chemical structure to amphetamine. It can be stimulating at higher doses, potentially benefitting patients with comorbid attention-deficit/hyperactivity disorder (ADHD) or significant apathy.13,23 Selegiline’s distinct quality is its availability as a transdermal patch, which may be useful for patients who struggle to take oral medications. At low doses (6 mg/24 h), the selegiline transdermal patch allows patients to disregard a dietary tyramine restriction because it avoids first-pass metabolism. It inhibits both MAO isomers in the brain but is only selective for MAO-B once concentrations are distributed to the liver. Higher doses require a tyramine-restricted diet because there is still some MAO-A inhibition in the gut. Selegiline is also stimulating because it is converted to amphetamine and methamphetamine.5,12,13,17,19,24
Despite promising results from the use of MAOIs, physicians and patients may be reluctant to use these medications due to perceived limitations. One prominent barrier is the infamous “cheese reaction.” Tyramine, an amino acid found in certain food and beverages (Table 25,13-18,25-28), is broken down by MAO-A in the gut. When this enzyme is inhibited, higher concentrations of tyramine reach systemic circulation. Tyramine’s release of norepinephrine (which now cannot be broken down) can lead to a hypertensive crisis. Consequently, a tyramine-restricted diet is recommended for patients taking an MAOI. However, the common notion that cheese, wine, and beer must be avoided is false, because most of the dietary restrictions developed following the discovery of MAOIs are antiquated.5,12,25-28 Patients who take an MAOI only need to slightly adjust their diet, as outlined in Table 2.5,13-18,25-28 A reasonable serving size of most foods and beverages containing tyramine is unlikely to elicit this “pressor” response. Of the 4 MAOIs FDA-approved for MDD, tranylcypromine appears to be the most sensitive to tyramine.21 Transient postdose hypertension (regardless of tyramine) may occur after taking an MAOI.29 Encourage patients to monitor their blood pressure.

Continue to: Additional hurdles include...
Additional hurdles include the required washout period from serotonergic medications and interactions with sympathomimetics. MAOIs pose the highest risk of serotonin syndrome; however, this usually occurs if given concomitantly with other serotonergic agents. The standard recommendation is a 14-day washout period from SSRIs (5 weeks for fluoxetine and 3 weeks for vortioxetine), SNRIs, mirtazapine, and other antidepressants. It can be distressing for patients to be without medication during that period. Because some antidepressants have much shorter half-lives, waiting 5 half-lives (typically 5 to 7 days) for the discontinued medication to be excreted is feasible if patients are closely monitored.5,12,13,25,27,30 There are rare instances where a TCA may be combined with an MAOI (typically initiated within 1 to 2 days of each other), but never clomipramine or imipramine due to their potent serotonin reuptake inhibition.31 If switching to an alternative MAOI, waiting 7 to 14 days is recommended to allow adequate time for the inhibited enzyme to regenerate.14-17,32 Taking medications that increase dopamine and norepinephrine (eg, stimulants or oral over-the-counter decongestants) with an MAOI is typically not recommended due to the risk of hypertensive crisis.25,27 In severe TRD or comorbid ADHD, successful simultaneous use of methylphenidate or amphetamine—typically at low doses—with close blood pressure monitoring has been reported.33 There have also been positive cases of the use of modafinil in combination with an MAOI; however, this should be done with caution.34,35 Clinicians must use clinical judgment when considering a combination of medications that pose a higher risk.
Tricyclic antidepressants
TCAs work differently than MAOIs to increase monoamines. They inhibit presynaptic serotonin and norepinephrine transporters in the CNS to increase levels of these chemicals in the synaptic cleft. While all TCAs inhibit these transporters, they do so at varying levels (Table 336-51). Based on their chemical structure, TCAs can be categorized into secondary and tertiary amines. Tertiary amines are metabolized via demethylation into their derivatives (Table 336-51). Patients who have recently suffered a myocardial infarction (MI) should avoid tertiary amines. TCAs can reduce heart rate variability, which is already decreased after an MI, thus presenting the potential for cardiac arrhythmias. TCAs should also be avoided in patients with cardiac conduction abnormalities.38-46,52 Patients with a prior baseline cardiac conduction defect, such as a bundle branch block, are at higher risk for further cardiac abnormalities. In those with a preexisting first-degree heart block, TCAs can still be used, but electrocardiogram monitoring is recommended.52,53 TCAs have also been reported to decrease the seizure threshold.38-46 They can be used with caution in patients who have a history of epilepsy or head trauma, or with concomitant medications that lower the seizure threshold.38-46

Overdose risk is a concern with TCAs because ingestion of 10 to 20 mg/kg can lead to significant toxicity.54 This is due to their blockage of voltage-gated sodium channels found in the CNS and heart, which contributes to overdose symptoms such as a widened QRS complex and seizures. Symptoms usually develop within 2 hours but may be delayed up to 6 hours.55 Patients with a history of overdose must be carefully assessed before initiating a TCA. Prescribing a limited supply of these medications may be valuable. The use of TCAs has often been limited due to their adverse effects, most of which are associated with their respective affinities for alpha 1, muscarinic 1, and histamine 1 receptors. Inhibition of the alpha 1 receptor is associated with hypotension, muscarinic 1 with anticholinergic adverse effects, and histamine 1 with sedation and weight gain. Tertiary amines have a higher affinity for these receptors compared to secondary amines, leading to a more significant adverse effect profile.36,50 Among TCAs, amitriptyline is the most likely to cause hypotension, whereas desipramine and nortriptyline are least likely. Amitriptyline and clomipramine are most likely to cause anticholinergic adverse effects, whereas desipramine and nortriptyline are the least likely. Amitriptyline, doxepin, and imipramine have the highest propensity for QTc prolongation.36
Beyond treating MDD, TCAs have shown benefits for treating other disease states (Table 438-46,49,56-61).These differing indications may help psychiatrists determine the best TCA to prescribe for a given patient. Amitriptyline is the most studied TCA for MDD; however, nortriptyline is typically preferred due to its favorable tolerability profile.4,62 Nortriptyline also has data supporting its use in ECT to prevent relapse.63 Amitriptyline and nortriptyline have shown benefits in patients with neuropathic pain and for migraine prophylaxis.56-60 Although frequently used for MDD, clomipramine is not FDA-approved for this indication, but is for obsessive-compulsive disorder.39 Doxepin is FDA-approved for insomnia at lower doses and for MDD at higher doses.40 Therefore, it may benefit patients with sleep difficulties secondary to depression. Desipramine has been used off-label to treat ADHD in children and has shown some benefits in adults.64-66 Protriptyline, trimipramine, and amoxapine are infrequently used in clinical practice.

A unique feature of TCAs is the ability to monitor serum concentrations (Table 336-51).Guidelines recommend therapeutic drug monitoring (TDM) with amitriptyline, clomipramine, imipramine, and nortriptyline for routine use. TDM is still recommended for doxepin, desipramine, and trimipramine, but its utility is largely for treatment failure or resistance.37 These plasma levels can be altered based on coadministered medications (Table 538-46) and should be closely monitored. Physicians should obtain a trough level after at least 5 half-lives and before the next dose is due, and use TDM as indicated to optimize dosing.

Continue to: CASE CONTINUED
CASE CONTINUED
Ms. B’s outpatient psychiatrist provides collateral information about her medical history and confirms her long-standing MDD with multiple medication trials, though she has never received an MAOI or TCA. Ms. B is adamant she does not want a medication-free period between treatments and refuses to adjust her diet, despite being educated on the few changes necessary. She has no contraindications for TCAs and may benefit from a TCA for her comorbid migraines. The care team expresses concern for TCA overdose to Ms. B and her family. Ms. B’s sister reassures the team they will have someone monitor and dispense her medications at home. They decide to discontinue her current psychiatric regimen, and Ms. B is started on nortriptyline 50 mg/d at night, with plans to titrate based on tolerability.
Related Resources
- Meyer JM. A concise guide to monoamine oxidase inhibitors. Current Psychiatry. 2017;16(12):14-16,18-23,47,A.
- Espejo GD. Treating major depressive disorder after limited response to an initial agent. Current Psychiatry. 2021;20(10):51-53. doi:10.12788/cp.0178
- American Association of Psychiatric Pharmacists (AAPP) MAOI Pharmacist Toolkit. https://aapp.org/guideline/maoi
Drug Brand Names
Amitriptyline • Elavil
Amphetamine • Adzenys, Dyanavel
Aripiprazole • Abilify
Clomipramine • Anafranil
Desipramine • Norpramin
Dextromethorphan/bupropion • Auvelity
Doxepin • Sinequan, Adapin
Esketamine • Spravato
Fluoxetine • Prozac
Imipramine • Tofranil
Isocarboxazid • Marplan
Methamphetamine • Desoxyn
Mirtazapine • Remeron
Modafinil • Provigil
Nortriptyline • Pamelor
Phenelzine • Nardil
Protriptyline • Vivactil
Selegiline • Emsam
Tranylcypromine • Parnate
Trimipramine • Surmontil
Vortioxetine • Trintellix
1. Gaynes BN, Lux L, Gartlehner G, et al. Defining treatment-resistant depression. Depress Anxiety. 2020;37(2):134-145. doi:10.1002/da.22968
2. Kennedy SH, Lam RW, McIntyre RS, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 clinical guidelines for the management of adults with major depressive disorder: section 3. Pharmacological treatments. Can J Psychiatry. 2016;61(9):540-560. doi:10.1177/0706743716659417
3. VA/DoD clinical practice guideline for the management of major depressive disorder. Veterans Health Administration and Department of Defense; 2016. https://www.healthquality.va.gov/guidelines/MH/mdd/VADoDMDDCPGFINAL82916.pdf
4. Gelenberg AJ, Freeman MP, Markowitz JC, et al. Practice guideline for the treatment of patients with major depressive disorder. Am J Psychiatry. 2010;167(Suppl 10):9-118.
5. Meyer JM. A concise guide to monoamine oxidase inhibitors. Current Psychiatry. 2017;16(12):14-16,18-23,47,A.
6. Taylor D. Selective serotonin reuptake inhibitors and tricyclic antidepressants in combination. Interactions and therapeutic uses. Br J Psychiatry. 1995;167(5):575-580. doi:10.1192/bjp.167.5.575
7. Thase ME, Trivedi MH, Rush AJ. MAOIs in the contemporary treatment of depression. Neuropsychopharmacology. 1995;12(3):185-219. doi:10.1016/0893-133X(94)00058-8
8. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905-1917. doi:10.1176/ajp.2006.163.11.1905
9. McGrath PJ, Stewart JW, Fava M, et al. Tranylcypromine versus venlafaxine plus mirtazapine following three failed antidepressant medication trials for depression: a STAR*D report. Am J Psychiatry. 2006;163(9):1531-1666. doi:10.1176/ajp.2006.163.9.1531
10. Kim T, Xu C, Amsterdam JD. Relative effectiveness of tricyclic antidepressant versus monoamine oxidase inhibitor monotherapy for treatment-resistant depression. J Affect Disord. 2019;250:199-203. doi:10.1016/j.jad.2019.03.028
11. Suchting R, Tirumalajaru V, Gareeb R, et al. Revisiting monoamine oxidase inhibitors for the treatment of depressive disorders: a systematic review and network meta-analysis. J Affect Disord. 2021;282:1153-1160. doi:10.1016/j.jad.2021.01.021
12. Stahl SM, Felker A. Monoamine oxidase inhibitors: a modern guide to an unrequited class of antidepressants. CNS Spectr. 2008;13(10):855-870. doi:10.1017/s1092852900016965
13. Chamberlain SR, Baldwin DS. Monoamine oxidase inhibitors (MAOIs) in psychiatric practice: how to use them safely and effectively. CNS Drugs. 2021;35(7):703-716. doi:10.1007/s40263-021-00832-x
14. Nardil [package insert]. New York, NY: Parke-Davis; 2009.
15. Marplan [package insert]. Parsippany, NJ: Validus Pharmaceuticals LLC; 2020.
16. Parnate [package insert]. Saint Michael, Barbados: Concordia Pharmaceuticals; 2015.
17. Emsam [package insert]. Morgantown, WV: Mylan Specialty LP; 2014.
18. Shulman KI, Herrmann N, Walker SE. Current place of monoamine oxidase inhibitors in the treatment of depression. CNS Drugs. 2013;27(10):789-797. doi:10.1007/s40263-013-0097-3
19. Sub Laban T, Saadabadi A. Monoamine oxidase inhibitors (MAOI). StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK539848/
20. Rabkin JG, Quitkin FM, McGrath P, et al. Adverse reactions to monoamine oxidase inhibitors. Part II. Treatment correlates and clinical management. J Clin Psychopharmacol. 1985;5(1):2-9.
21. Gillman PK. Advances pertaining to the pharmacology and interactions of irreversible nonselective monoamine oxidase inhibitors. J Clin Psychopharmacol. 2011;31(1):66-74. doi:10.1097/JCP.0b013e31820469ea
22. Sidhu G, Marwaha R. Phenelzine. StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK554508/
23. Frieling H, Bleich S. Tranylcypromine: new perspectives on an “old” drug. Eur Arch Psychiatry Clin Neurosci. 2006;256(5):268-273. doi:10.1007/s00406-006-0660-8
24. Goodnick PJ. Seligiline transdermal system in depression. Expert Opin Pharmacother. 2007;8(1):59-64. doi:10.1517/14656566.8.1.59
25. Edinoff AN, Swinford CR, Odisho AS, et al. Clinically relevant drug interactions with monoamine oxidase inhibitors. Health Psychol Res. 2022;10(4):39576. doi:10.52965/001c.39576
26. Gillman PK. A reassessment of the safety profile of monoamine oxidase inhibitors: elucidating tired old tyramine myths. J Neural Transm (Vienna). 2018;125(11):1707-1717. doi:10.1007/s00702-018-1932-y
27. Flockhart DA. Dietary restrictions and drug interactions with monoamine oxidase inhibitors: an update. J Clin Psychiatry. 2012;73 Suppl 1:17-24. doi:10.4088/JCP.11096su1c.03
28. McCabe-Sellers BJ, Staggs CG, Bogle ML. Tyramine in foods and monoamine oxidase inhibitor drugs: a crossroad where medicine, nutrition, pharmacy, and food industry converge. J Food Composit Anal. 2006;19:S58-S65. doi:10.1016/j.jfca.2005.12.008
29. Keck PE Jr, Vuckovic A, Pope HG Jr, et al. Acute cardiovascular response to monoamine oxidase inhibitors: a prospective assessment. J Clin Psychopharmacol. 1989;9(3):203-206.
30. Bodkin JA, Dunlop BW. Moving on with monoamine oxidase inhibitors. Focus (Am Psychiatr Publ). 2021;19(1):50-52. doi:10.1176/appi.focus.20200046
31. Amsterdam JD, Kim TT. Relative effectiveness of monoamine oxidase inhibitor and tricyclic antidepressant combination therapy for treatment-resistant depression. J Clin Psychopharmacol. 2019;39(6):649-652. doi:10.1097/JCP.0000000000001130
32. Keks N, Hope J, Keogh S. Switching and stopping antidepressants. Aust Prescr. 2016;39(3):76-83. doi:10.18773/austprescr.2016.039
33. Israel JA. Combining stimulants and monoamine oxidase inhibitors: a reexamination of the literature and a report of a new treatment combination. Prim Care Companion CNS Disord. 2015;17(6):10.4088/PCC.15br01836. doi:10.4088/PCC.15br01836
34. Clemons WE, Makela E, Young J. Concomitant use of modafinil and tranylcypromine in a patient with narcolepsy: a case report. Sleep Med. 2004;5(5):509-511. doi:10.1016/j.sleep.2004.06.006
35. Ashton AK. Modafinil augmentation of phenelzine for residual fatigue in dysthymia. Am J Psychiatry. 2004;161(9):1716-1717. doi:10.1176/appi.ajp.161.9.1716-a
36. O’Donnell JM, Bies RR, Shelton RC. Drug therapy of depression and anxiety disorders. In: Brunton LL, Hilal-Dandan R, Knollmann BC, eds. Goodman & Gilman’s: The Pharmacological Basis of Therapeutics. 13th ed. McGraw Hill; 2017. Accessed June 4, 2023. https://accessanesthesiology.mhmedical.com/content.aspx?bookid=2189§ionid=169518711
37. Hiemke C, Bergemann N, Clement HW, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51(1-02):9-62. doi:10.1055/s-0043-116492
38. Amitriptyline hydrochloride [package insert]. East Brunswick, NJ: Unichem Pharmaceuticals (USA); 2021.
39. Clomipramine hydrochloride [package insert]. East Windsor, NJ: Aurobindo Pharma Limited; 2023.
40. Doxepin hydrochloride capsules, USP [package insert]. Bedminster, NJ: Alembic Pharmaceuticals Inc; 2021.
41. Imipramine hydrochloride tablet [package insert]. Fairfield, NJ: Leading Pharma LLC USA; 2022.
42. Trimipramine maleate [package insert]. Northvale, NJ: Elite Laboratories Inc; 2021.
43. Amoxapine [package insert]. Parsippany, NJ: Actavis Pharma Inc; 2015.
44. Desipramine hydrochloride tablets [package insert]. Bedminster, NJ: Alembic Pharmaceuticals Inc; 2023.
45. Nortriptyline hydrochloride capsules, USP [package insert]. Parsippany, NJ: Teva Pharmaceuticals Inc; 2021.
46. Protriptyline hydrochloride [package insert]. Bensalem, PA: Sigmapharm Laboratories, LLC; 2023.
47. Calvo B, García MJ, Pedraz JL, et al. Pharmacokinetics of amoxapine and its active metabolites. Int J Clin Pharmacol Ther Toxicol. 1985;23(4):180-185.
48. Ziegler VE, Biggs JT, Wylie LT, et al. Protriptyline kinetics. Clin Pharmacol Ther. 1978;23(5):580-584. doi:10.1002/cpt1978235580
49. Cleare A, Pariante CM, Young AH, et al. Evidence-based guidelines for treating depressive disorders with antidepressants: a revision of the 2008 British Association for Psychopharmacology guidelines. J Psychopharmacol. 2015;29(5):459-525. doi:10.1177/0269881115581093
50. Richelson E. Synaptic effects of antidepressants. J Clin Psychopharmacol. 1996;16(3 Suppl 2):1S-9S. doi:10.1097/00004714-199606002-00001
51. Vos CF, Aarnoutse RE, Op de Coul MJM, et al. Tricyclic antidepressants for major depressive disorder: a comprehensive evaluation of current practice in the Netherlands. BMC Psychiatry. 2021;21(1):481. doi:10.1186/s12888-021-03490-x
52. Alvarez W Jr, Pickworth KK. Safety of antidepressant drugs in the patient with cardiac disease: a review of the literature. Pharmacotherapy. 2003;23(6):754-771. doi:10.1592/phco.23.6.754.32185
53. Dietch JT, Fine M. The effect of nortriptyline in elderly patients with cardiac conduction disease. J Clin Psychiatry. 1990;51(2):65-67.
54. Valento M, Liebelt EL. Cyclic antidepressants. In: Nelson LS, Howland M, Lewin NA, et al, eds. Goldfrank’s Toxicologic Emergencies. 9th ed. McGraw Hill; 2011. Accessed June 10, 2023. https://accesspharmacy.mhmedical.com/content.aspx?bookid=2569§ionid=210274664
55. Woolf AD, Erdman AR, Nelson LS, et al. Tricyclic antidepressant poisoning: an evidence-based consensus guideline for out-of-hospital management. Clin Toxicol (Phila). 2007;45(3):203-233. doi:10.1080/15563650701226192
56. Baldwin DS, Anderson IM, Nutt DJ, et al. Evidence-based pharmacological treatment of anxiety disorders, post-traumatic stress disorder and obsessive-compulsive disorder: a revision of the 2005 guidelines from the British Association for Psychopharmacology. J Psychopharmacol. 2014;28(5):403-439. doi:10.1177/0269881114525674
57. Moulin DE, Clark AJ, Gilron I, et al. Pharmacological management of chronic neuropathic pain - consensus statement and guidelines from the Canadian Pain Society. Pain Res Manag. 2007;12(1):13-21. doi:10.1155/2007/730785
58. Finnerup NB, Attal N, Haroutounian S, et al. Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol. 2015;14(2):162-173. doi:10.1016/S1474-4422(14)70251-0
59. Saarto T, Wiffen PJ. Antidepressants for neuropathic pain. Cochrane Database Syst Rev. 2007;(4):CD005454. doi:10.1002/14651858.CD005454.pub2
60. Burch R. Antidepressants for preventive treatment of migraine. Curr Treat Options Neurol. 2019;21(4):18. doi:10.1007/s11940-019-0557-2
61. Ailani J, Burch RC, Robbins MS; Board of Directors of the American Headache Society. The American Headache Society consensus statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61(7):1021-1039. doi:10.1111/head.14153
62. Bauer M, Pfennig A, Severus E, et al. World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for biological treatment of unipolar depressive disorders, part 1: update 2013 on the acute and continuation treatment of unipolar depressive disorders. World J Biol Psychiatry. 2013;14(5):334-385. doi:10.3109/15622975.2013.804195
63. Jelovac A, Kolshus E, McLoughlin DM. Relapse following successful electroconvulsive therapy for major depression: a meta-analysis. Neuropsychopharmacology. 2013;38(12):2467-2474. doi:10.1038/npp.2013.149
64. Spencer T, Biederman J, Coffey B, et al. A double-blind comparison of desipramine and placebo in children and adolescents with chronic tic disorder and comorbid attention-deficit/hyperactivity disorder. Arch Gen Psychiatry. 2002;59(7):649-656. doi:10.1001/archpsyc.59.7.649
65. Spencer T, Biederman J, Wilens T, et al. Pharmacotherapy of attention-deficit hyperactivity disorder across the life cycle. J Am Acad Child Adolesc Psychiatry. 1996;35(4):409-432. doi:10.1097/00004583-199604000-00008
66. Wilens TE, Biederman J, Prince J, et al. Six-week, double-blind, placebo-controlled study of desipramine for adult attention deficit hyperactivity disorder. Am J Psychiatry. 1996;153(9):1147-1153. doi:10.1176/ajp.153.9.1147
1. Gaynes BN, Lux L, Gartlehner G, et al. Defining treatment-resistant depression. Depress Anxiety. 2020;37(2):134-145. doi:10.1002/da.22968
2. Kennedy SH, Lam RW, McIntyre RS, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) 2016 clinical guidelines for the management of adults with major depressive disorder: section 3. Pharmacological treatments. Can J Psychiatry. 2016;61(9):540-560. doi:10.1177/0706743716659417
3. VA/DoD clinical practice guideline for the management of major depressive disorder. Veterans Health Administration and Department of Defense; 2016. https://www.healthquality.va.gov/guidelines/MH/mdd/VADoDMDDCPGFINAL82916.pdf
4. Gelenberg AJ, Freeman MP, Markowitz JC, et al. Practice guideline for the treatment of patients with major depressive disorder. Am J Psychiatry. 2010;167(Suppl 10):9-118.
5. Meyer JM. A concise guide to monoamine oxidase inhibitors. Current Psychiatry. 2017;16(12):14-16,18-23,47,A.
6. Taylor D. Selective serotonin reuptake inhibitors and tricyclic antidepressants in combination. Interactions and therapeutic uses. Br J Psychiatry. 1995;167(5):575-580. doi:10.1192/bjp.167.5.575
7. Thase ME, Trivedi MH, Rush AJ. MAOIs in the contemporary treatment of depression. Neuropsychopharmacology. 1995;12(3):185-219. doi:10.1016/0893-133X(94)00058-8
8. Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905-1917. doi:10.1176/ajp.2006.163.11.1905
9. McGrath PJ, Stewart JW, Fava M, et al. Tranylcypromine versus venlafaxine plus mirtazapine following three failed antidepressant medication trials for depression: a STAR*D report. Am J Psychiatry. 2006;163(9):1531-1666. doi:10.1176/ajp.2006.163.9.1531
10. Kim T, Xu C, Amsterdam JD. Relative effectiveness of tricyclic antidepressant versus monoamine oxidase inhibitor monotherapy for treatment-resistant depression. J Affect Disord. 2019;250:199-203. doi:10.1016/j.jad.2019.03.028
11. Suchting R, Tirumalajaru V, Gareeb R, et al. Revisiting monoamine oxidase inhibitors for the treatment of depressive disorders: a systematic review and network meta-analysis. J Affect Disord. 2021;282:1153-1160. doi:10.1016/j.jad.2021.01.021
12. Stahl SM, Felker A. Monoamine oxidase inhibitors: a modern guide to an unrequited class of antidepressants. CNS Spectr. 2008;13(10):855-870. doi:10.1017/s1092852900016965
13. Chamberlain SR, Baldwin DS. Monoamine oxidase inhibitors (MAOIs) in psychiatric practice: how to use them safely and effectively. CNS Drugs. 2021;35(7):703-716. doi:10.1007/s40263-021-00832-x
14. Nardil [package insert]. New York, NY: Parke-Davis; 2009.
15. Marplan [package insert]. Parsippany, NJ: Validus Pharmaceuticals LLC; 2020.
16. Parnate [package insert]. Saint Michael, Barbados: Concordia Pharmaceuticals; 2015.
17. Emsam [package insert]. Morgantown, WV: Mylan Specialty LP; 2014.
18. Shulman KI, Herrmann N, Walker SE. Current place of monoamine oxidase inhibitors in the treatment of depression. CNS Drugs. 2013;27(10):789-797. doi:10.1007/s40263-013-0097-3
19. Sub Laban T, Saadabadi A. Monoamine oxidase inhibitors (MAOI). StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK539848/
20. Rabkin JG, Quitkin FM, McGrath P, et al. Adverse reactions to monoamine oxidase inhibitors. Part II. Treatment correlates and clinical management. J Clin Psychopharmacol. 1985;5(1):2-9.
21. Gillman PK. Advances pertaining to the pharmacology and interactions of irreversible nonselective monoamine oxidase inhibitors. J Clin Psychopharmacol. 2011;31(1):66-74. doi:10.1097/JCP.0b013e31820469ea
22. Sidhu G, Marwaha R. Phenelzine. StatPearls Publishing; 2023. https://www.ncbi.nlm.nih.gov/books/NBK554508/
23. Frieling H, Bleich S. Tranylcypromine: new perspectives on an “old” drug. Eur Arch Psychiatry Clin Neurosci. 2006;256(5):268-273. doi:10.1007/s00406-006-0660-8
24. Goodnick PJ. Seligiline transdermal system in depression. Expert Opin Pharmacother. 2007;8(1):59-64. doi:10.1517/14656566.8.1.59
25. Edinoff AN, Swinford CR, Odisho AS, et al. Clinically relevant drug interactions with monoamine oxidase inhibitors. Health Psychol Res. 2022;10(4):39576. doi:10.52965/001c.39576
26. Gillman PK. A reassessment of the safety profile of monoamine oxidase inhibitors: elucidating tired old tyramine myths. J Neural Transm (Vienna). 2018;125(11):1707-1717. doi:10.1007/s00702-018-1932-y
27. Flockhart DA. Dietary restrictions and drug interactions with monoamine oxidase inhibitors: an update. J Clin Psychiatry. 2012;73 Suppl 1:17-24. doi:10.4088/JCP.11096su1c.03
28. McCabe-Sellers BJ, Staggs CG, Bogle ML. Tyramine in foods and monoamine oxidase inhibitor drugs: a crossroad where medicine, nutrition, pharmacy, and food industry converge. J Food Composit Anal. 2006;19:S58-S65. doi:10.1016/j.jfca.2005.12.008
29. Keck PE Jr, Vuckovic A, Pope HG Jr, et al. Acute cardiovascular response to monoamine oxidase inhibitors: a prospective assessment. J Clin Psychopharmacol. 1989;9(3):203-206.
30. Bodkin JA, Dunlop BW. Moving on with monoamine oxidase inhibitors. Focus (Am Psychiatr Publ). 2021;19(1):50-52. doi:10.1176/appi.focus.20200046
31. Amsterdam JD, Kim TT. Relative effectiveness of monoamine oxidase inhibitor and tricyclic antidepressant combination therapy for treatment-resistant depression. J Clin Psychopharmacol. 2019;39(6):649-652. doi:10.1097/JCP.0000000000001130
32. Keks N, Hope J, Keogh S. Switching and stopping antidepressants. Aust Prescr. 2016;39(3):76-83. doi:10.18773/austprescr.2016.039
33. Israel JA. Combining stimulants and monoamine oxidase inhibitors: a reexamination of the literature and a report of a new treatment combination. Prim Care Companion CNS Disord. 2015;17(6):10.4088/PCC.15br01836. doi:10.4088/PCC.15br01836
34. Clemons WE, Makela E, Young J. Concomitant use of modafinil and tranylcypromine in a patient with narcolepsy: a case report. Sleep Med. 2004;5(5):509-511. doi:10.1016/j.sleep.2004.06.006
35. Ashton AK. Modafinil augmentation of phenelzine for residual fatigue in dysthymia. Am J Psychiatry. 2004;161(9):1716-1717. doi:10.1176/appi.ajp.161.9.1716-a
36. O’Donnell JM, Bies RR, Shelton RC. Drug therapy of depression and anxiety disorders. In: Brunton LL, Hilal-Dandan R, Knollmann BC, eds. Goodman & Gilman’s: The Pharmacological Basis of Therapeutics. 13th ed. McGraw Hill; 2017. Accessed June 4, 2023. https://accessanesthesiology.mhmedical.com/content.aspx?bookid=2189§ionid=169518711
37. Hiemke C, Bergemann N, Clement HW, et al. Consensus guidelines for therapeutic drug monitoring in neuropsychopharmacology: update 2017. Pharmacopsychiatry. 2018;51(1-02):9-62. doi:10.1055/s-0043-116492
38. Amitriptyline hydrochloride [package insert]. East Brunswick, NJ: Unichem Pharmaceuticals (USA); 2021.
39. Clomipramine hydrochloride [package insert]. East Windsor, NJ: Aurobindo Pharma Limited; 2023.
40. Doxepin hydrochloride capsules, USP [package insert]. Bedminster, NJ: Alembic Pharmaceuticals Inc; 2021.
41. Imipramine hydrochloride tablet [package insert]. Fairfield, NJ: Leading Pharma LLC USA; 2022.
42. Trimipramine maleate [package insert]. Northvale, NJ: Elite Laboratories Inc; 2021.
43. Amoxapine [package insert]. Parsippany, NJ: Actavis Pharma Inc; 2015.
44. Desipramine hydrochloride tablets [package insert]. Bedminster, NJ: Alembic Pharmaceuticals Inc; 2023.
45. Nortriptyline hydrochloride capsules, USP [package insert]. Parsippany, NJ: Teva Pharmaceuticals Inc; 2021.
46. Protriptyline hydrochloride [package insert]. Bensalem, PA: Sigmapharm Laboratories, LLC; 2023.
47. Calvo B, García MJ, Pedraz JL, et al. Pharmacokinetics of amoxapine and its active metabolites. Int J Clin Pharmacol Ther Toxicol. 1985;23(4):180-185.
48. Ziegler VE, Biggs JT, Wylie LT, et al. Protriptyline kinetics. Clin Pharmacol Ther. 1978;23(5):580-584. doi:10.1002/cpt1978235580
49. Cleare A, Pariante CM, Young AH, et al. Evidence-based guidelines for treating depressive disorders with antidepressants: a revision of the 2008 British Association for Psychopharmacology guidelines. J Psychopharmacol. 2015;29(5):459-525. doi:10.1177/0269881115581093
50. Richelson E. Synaptic effects of antidepressants. J Clin Psychopharmacol. 1996;16(3 Suppl 2):1S-9S. doi:10.1097/00004714-199606002-00001
51. Vos CF, Aarnoutse RE, Op de Coul MJM, et al. Tricyclic antidepressants for major depressive disorder: a comprehensive evaluation of current practice in the Netherlands. BMC Psychiatry. 2021;21(1):481. doi:10.1186/s12888-021-03490-x
52. Alvarez W Jr, Pickworth KK. Safety of antidepressant drugs in the patient with cardiac disease: a review of the literature. Pharmacotherapy. 2003;23(6):754-771. doi:10.1592/phco.23.6.754.32185
53. Dietch JT, Fine M. The effect of nortriptyline in elderly patients with cardiac conduction disease. J Clin Psychiatry. 1990;51(2):65-67.
54. Valento M, Liebelt EL. Cyclic antidepressants. In: Nelson LS, Howland M, Lewin NA, et al, eds. Goldfrank’s Toxicologic Emergencies. 9th ed. McGraw Hill; 2011. Accessed June 10, 2023. https://accesspharmacy.mhmedical.com/content.aspx?bookid=2569§ionid=210274664
55. Woolf AD, Erdman AR, Nelson LS, et al. Tricyclic antidepressant poisoning: an evidence-based consensus guideline for out-of-hospital management. Clin Toxicol (Phila). 2007;45(3):203-233. doi:10.1080/15563650701226192
56. Baldwin DS, Anderson IM, Nutt DJ, et al. Evidence-based pharmacological treatment of anxiety disorders, post-traumatic stress disorder and obsessive-compulsive disorder: a revision of the 2005 guidelines from the British Association for Psychopharmacology. J Psychopharmacol. 2014;28(5):403-439. doi:10.1177/0269881114525674
57. Moulin DE, Clark AJ, Gilron I, et al. Pharmacological management of chronic neuropathic pain - consensus statement and guidelines from the Canadian Pain Society. Pain Res Manag. 2007;12(1):13-21. doi:10.1155/2007/730785
58. Finnerup NB, Attal N, Haroutounian S, et al. Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol. 2015;14(2):162-173. doi:10.1016/S1474-4422(14)70251-0
59. Saarto T, Wiffen PJ. Antidepressants for neuropathic pain. Cochrane Database Syst Rev. 2007;(4):CD005454. doi:10.1002/14651858.CD005454.pub2
60. Burch R. Antidepressants for preventive treatment of migraine. Curr Treat Options Neurol. 2019;21(4):18. doi:10.1007/s11940-019-0557-2
61. Ailani J, Burch RC, Robbins MS; Board of Directors of the American Headache Society. The American Headache Society consensus statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61(7):1021-1039. doi:10.1111/head.14153
62. Bauer M, Pfennig A, Severus E, et al. World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for biological treatment of unipolar depressive disorders, part 1: update 2013 on the acute and continuation treatment of unipolar depressive disorders. World J Biol Psychiatry. 2013;14(5):334-385. doi:10.3109/15622975.2013.804195
63. Jelovac A, Kolshus E, McLoughlin DM. Relapse following successful electroconvulsive therapy for major depression: a meta-analysis. Neuropsychopharmacology. 2013;38(12):2467-2474. doi:10.1038/npp.2013.149
64. Spencer T, Biederman J, Coffey B, et al. A double-blind comparison of desipramine and placebo in children and adolescents with chronic tic disorder and comorbid attention-deficit/hyperactivity disorder. Arch Gen Psychiatry. 2002;59(7):649-656. doi:10.1001/archpsyc.59.7.649
65. Spencer T, Biederman J, Wilens T, et al. Pharmacotherapy of attention-deficit hyperactivity disorder across the life cycle. J Am Acad Child Adolesc Psychiatry. 1996;35(4):409-432. doi:10.1097/00004583-199604000-00008
66. Wilens TE, Biederman J, Prince J, et al. Six-week, double-blind, placebo-controlled study of desipramine for adult attention deficit hyperactivity disorder. Am J Psychiatry. 1996;153(9):1147-1153. doi:10.1176/ajp.153.9.1147




