User login
FDA approves rituximab biosimilar for lymphoma
(NHL).
Celltrion’s Truxima (rituximab-abbs) is a biosimilar of Genentech’s Rituxan (rituximab) and the first biosimilar approved in the United States to treat NHL.
Truxima (formerly CT-P10) is approved to treat adults with CD20-positive, B-cell NHL, either as a single agent or in combination with chemotherapy. Truxima is approved as a single agent to treat relapsed or refractory, low grade or follicular, CD20-positive, B-cell NHL. Truxima is approved in combination with first-line chemotherapy to treat previously untreated follicular, CD20-positive, B-cell NHL.
Truxima is approved as single-agent maintenance therapy in patients with follicular, CD20-positive, B-cell NHL who achieve a complete or partial response to a rituximab product in combination with chemotherapy. Truxima also is approved as a single agent to treat nonprogressing, low-grade, CD20-positive, B-cell NHL after first-line treatment with cyclophosphamide, vincristine, and prednisone.The label for Truxima contains a boxed warning detailing the risk of fatal infusion reactions, severe skin and mouth reactions (some with fatal outcomes), hepatitis B virus reactivation that may cause serious liver problems (including liver failure and death), and progressive multifocal leukoencephalopathy.
The FDA said its approval of Truxima is “based on a review of evidence that included extensive structural and functional characterization, animal study data, human pharmacokinetic data, clinical immunogenicity data, and other clinical data that demonstrates Truxima is biosimilar to Rituxan.”
Findings from a phase 3 trial suggested that Truxima is equivalent to the reference product in patients with low-tumor-burden follicular lymphoma (Lancet Haematol. 2018 Nov;5[11]:e543-53).
(NHL).
Celltrion’s Truxima (rituximab-abbs) is a biosimilar of Genentech’s Rituxan (rituximab) and the first biosimilar approved in the United States to treat NHL.
Truxima (formerly CT-P10) is approved to treat adults with CD20-positive, B-cell NHL, either as a single agent or in combination with chemotherapy. Truxima is approved as a single agent to treat relapsed or refractory, low grade or follicular, CD20-positive, B-cell NHL. Truxima is approved in combination with first-line chemotherapy to treat previously untreated follicular, CD20-positive, B-cell NHL.
Truxima is approved as single-agent maintenance therapy in patients with follicular, CD20-positive, B-cell NHL who achieve a complete or partial response to a rituximab product in combination with chemotherapy. Truxima also is approved as a single agent to treat nonprogressing, low-grade, CD20-positive, B-cell NHL after first-line treatment with cyclophosphamide, vincristine, and prednisone.The label for Truxima contains a boxed warning detailing the risk of fatal infusion reactions, severe skin and mouth reactions (some with fatal outcomes), hepatitis B virus reactivation that may cause serious liver problems (including liver failure and death), and progressive multifocal leukoencephalopathy.
The FDA said its approval of Truxima is “based on a review of evidence that included extensive structural and functional characterization, animal study data, human pharmacokinetic data, clinical immunogenicity data, and other clinical data that demonstrates Truxima is biosimilar to Rituxan.”
Findings from a phase 3 trial suggested that Truxima is equivalent to the reference product in patients with low-tumor-burden follicular lymphoma (Lancet Haematol. 2018 Nov;5[11]:e543-53).
(NHL).
Celltrion’s Truxima (rituximab-abbs) is a biosimilar of Genentech’s Rituxan (rituximab) and the first biosimilar approved in the United States to treat NHL.
Truxima (formerly CT-P10) is approved to treat adults with CD20-positive, B-cell NHL, either as a single agent or in combination with chemotherapy. Truxima is approved as a single agent to treat relapsed or refractory, low grade or follicular, CD20-positive, B-cell NHL. Truxima is approved in combination with first-line chemotherapy to treat previously untreated follicular, CD20-positive, B-cell NHL.
Truxima is approved as single-agent maintenance therapy in patients with follicular, CD20-positive, B-cell NHL who achieve a complete or partial response to a rituximab product in combination with chemotherapy. Truxima also is approved as a single agent to treat nonprogressing, low-grade, CD20-positive, B-cell NHL after first-line treatment with cyclophosphamide, vincristine, and prednisone.The label for Truxima contains a boxed warning detailing the risk of fatal infusion reactions, severe skin and mouth reactions (some with fatal outcomes), hepatitis B virus reactivation that may cause serious liver problems (including liver failure and death), and progressive multifocal leukoencephalopathy.
The FDA said its approval of Truxima is “based on a review of evidence that included extensive structural and functional characterization, animal study data, human pharmacokinetic data, clinical immunogenicity data, and other clinical data that demonstrates Truxima is biosimilar to Rituxan.”
Findings from a phase 3 trial suggested that Truxima is equivalent to the reference product in patients with low-tumor-burden follicular lymphoma (Lancet Haematol. 2018 Nov;5[11]:e543-53).
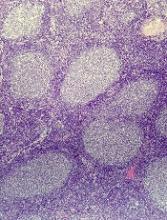
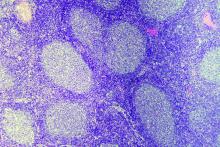
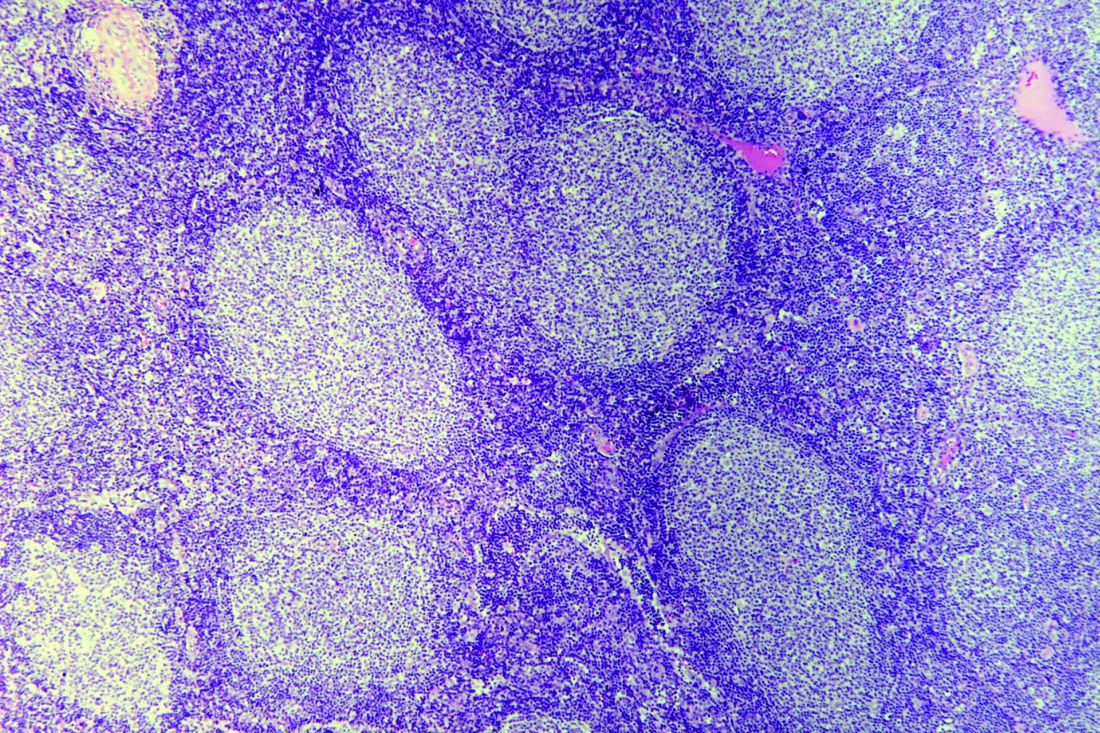
Cortactin expression aids in CLL-MCL differential
The presence or absence in tumor cells of cortactin, a cytoskeleton-remodeling adapter protein, may be a marker to help pathologists distinguish between chronic lymphocytic leukemia (CLL) and mantle cell lymphoma (MCL), investigators suggest.
A study of cortactin expression in tumor samples from patients with B-cell CLL, MCL, and other hematologic malignancies showed that while cortactin was present in 14 of 17 CLL samples, it was not expressed on any of 16 MCL samples, reported Marco Pizzi, MD, PhD, from the University of Padova (Italy) and his colleagues.
“In particular, cortactin may contribute to the differential diagnosis between CLL and MCL, two neoplasms with similar histological features but very different clinical outcome. Further studies are needed to clarify the molecular mechanisms of deranged cortactin expression in MCL and CLL and to investigate any possible relationship between cortactin status and the biological features of these lymphomas,” they wrote in Human Pathology.
Overexpression of cortactin has been reported in several solid tumors, and increased expression of CTTN, the gene encoding for cortactin, has been associated with aggressive, poor prognosis disease, the investigators noted.
To characterize cortactin expression in lymphoid and hematopoietic cells and detect potential associations between cortactin and virulence of hematologic malignancies, the investigators performed immunohistochemical analysis on samples from 131 patients treated at their center. The samples included 17 cases of CLL, 16 of MCL, 25 of follicular lymphoma (FL), 30 of marginal zone lymphoma (MZL), 10 of hairy cell leukemia, three of splenic diffuse red pulp small B-cell lymphomas (SDRPBL), and 30 of diffuse large B-cell lymphoma (DLBCL).
They found that cortactin was expressed in 14 of the 17 CLL samples, all 10 of the HCL samples, and 22 of the 30 DLBCL samples. In contrast, there was no cortactin expression detected in any of either 16 MCL or three SDRPBL samples. The researchers found that 13 of 30 MZL samples had low-level staining. In FL, cortactin was expressed in 2 of the samples but in the remaining 23 cases the researchers found only scattered cortactin-positive lymphoid elements of non–B-cell lineage.
The investigators also found that cortactin expression in CLL correlated with other CLL-specific markers, and found that expression of two or more of the markers had 89.1% sensitivity, 100% specificity, a 100% positive predictive value, and 90.5% negative predictive value for a diagnosis of CLL.
In addition, they saw that the immunohistochemical results were similar to those for CTTN gene expression assessed by in silico analysis.
The investigators noted that CLL and MCL are challenging to differentiate from one another because of morphologic similarities and partially overlapping immunophenotypes.
“In this context, cortactin expression would strongly sustain a diagnosis of CLL over MCL, particularly in association with other CLL markers (i.e., LEF1 and CD200),” they wrote.
The study was internally supported. The authors declared no conflicts of interest.
SOURCE: Pizzi M et al. Hum Pathol. 2018 Nov 17. doi: 10.1016/j.humpath.2018.10.038.
The presence or absence in tumor cells of cortactin, a cytoskeleton-remodeling adapter protein, may be a marker to help pathologists distinguish between chronic lymphocytic leukemia (CLL) and mantle cell lymphoma (MCL), investigators suggest.
A study of cortactin expression in tumor samples from patients with B-cell CLL, MCL, and other hematologic malignancies showed that while cortactin was present in 14 of 17 CLL samples, it was not expressed on any of 16 MCL samples, reported Marco Pizzi, MD, PhD, from the University of Padova (Italy) and his colleagues.
“In particular, cortactin may contribute to the differential diagnosis between CLL and MCL, two neoplasms with similar histological features but very different clinical outcome. Further studies are needed to clarify the molecular mechanisms of deranged cortactin expression in MCL and CLL and to investigate any possible relationship between cortactin status and the biological features of these lymphomas,” they wrote in Human Pathology.
Overexpression of cortactin has been reported in several solid tumors, and increased expression of CTTN, the gene encoding for cortactin, has been associated with aggressive, poor prognosis disease, the investigators noted.
To characterize cortactin expression in lymphoid and hematopoietic cells and detect potential associations between cortactin and virulence of hematologic malignancies, the investigators performed immunohistochemical analysis on samples from 131 patients treated at their center. The samples included 17 cases of CLL, 16 of MCL, 25 of follicular lymphoma (FL), 30 of marginal zone lymphoma (MZL), 10 of hairy cell leukemia, three of splenic diffuse red pulp small B-cell lymphomas (SDRPBL), and 30 of diffuse large B-cell lymphoma (DLBCL).
They found that cortactin was expressed in 14 of the 17 CLL samples, all 10 of the HCL samples, and 22 of the 30 DLBCL samples. In contrast, there was no cortactin expression detected in any of either 16 MCL or three SDRPBL samples. The researchers found that 13 of 30 MZL samples had low-level staining. In FL, cortactin was expressed in 2 of the samples but in the remaining 23 cases the researchers found only scattered cortactin-positive lymphoid elements of non–B-cell lineage.
The investigators also found that cortactin expression in CLL correlated with other CLL-specific markers, and found that expression of two or more of the markers had 89.1% sensitivity, 100% specificity, a 100% positive predictive value, and 90.5% negative predictive value for a diagnosis of CLL.
In addition, they saw that the immunohistochemical results were similar to those for CTTN gene expression assessed by in silico analysis.
The investigators noted that CLL and MCL are challenging to differentiate from one another because of morphologic similarities and partially overlapping immunophenotypes.
“In this context, cortactin expression would strongly sustain a diagnosis of CLL over MCL, particularly in association with other CLL markers (i.e., LEF1 and CD200),” they wrote.
The study was internally supported. The authors declared no conflicts of interest.
SOURCE: Pizzi M et al. Hum Pathol. 2018 Nov 17. doi: 10.1016/j.humpath.2018.10.038.
The presence or absence in tumor cells of cortactin, a cytoskeleton-remodeling adapter protein, may be a marker to help pathologists distinguish between chronic lymphocytic leukemia (CLL) and mantle cell lymphoma (MCL), investigators suggest.
A study of cortactin expression in tumor samples from patients with B-cell CLL, MCL, and other hematologic malignancies showed that while cortactin was present in 14 of 17 CLL samples, it was not expressed on any of 16 MCL samples, reported Marco Pizzi, MD, PhD, from the University of Padova (Italy) and his colleagues.
“In particular, cortactin may contribute to the differential diagnosis between CLL and MCL, two neoplasms with similar histological features but very different clinical outcome. Further studies are needed to clarify the molecular mechanisms of deranged cortactin expression in MCL and CLL and to investigate any possible relationship between cortactin status and the biological features of these lymphomas,” they wrote in Human Pathology.
Overexpression of cortactin has been reported in several solid tumors, and increased expression of CTTN, the gene encoding for cortactin, has been associated with aggressive, poor prognosis disease, the investigators noted.
To characterize cortactin expression in lymphoid and hematopoietic cells and detect potential associations between cortactin and virulence of hematologic malignancies, the investigators performed immunohistochemical analysis on samples from 131 patients treated at their center. The samples included 17 cases of CLL, 16 of MCL, 25 of follicular lymphoma (FL), 30 of marginal zone lymphoma (MZL), 10 of hairy cell leukemia, three of splenic diffuse red pulp small B-cell lymphomas (SDRPBL), and 30 of diffuse large B-cell lymphoma (DLBCL).
They found that cortactin was expressed in 14 of the 17 CLL samples, all 10 of the HCL samples, and 22 of the 30 DLBCL samples. In contrast, there was no cortactin expression detected in any of either 16 MCL or three SDRPBL samples. The researchers found that 13 of 30 MZL samples had low-level staining. In FL, cortactin was expressed in 2 of the samples but in the remaining 23 cases the researchers found only scattered cortactin-positive lymphoid elements of non–B-cell lineage.
The investigators also found that cortactin expression in CLL correlated with other CLL-specific markers, and found that expression of two or more of the markers had 89.1% sensitivity, 100% specificity, a 100% positive predictive value, and 90.5% negative predictive value for a diagnosis of CLL.
In addition, they saw that the immunohistochemical results were similar to those for CTTN gene expression assessed by in silico analysis.
The investigators noted that CLL and MCL are challenging to differentiate from one another because of morphologic similarities and partially overlapping immunophenotypes.
“In this context, cortactin expression would strongly sustain a diagnosis of CLL over MCL, particularly in association with other CLL markers (i.e., LEF1 and CD200),” they wrote.
The study was internally supported. The authors declared no conflicts of interest.
SOURCE: Pizzi M et al. Hum Pathol. 2018 Nov 17. doi: 10.1016/j.humpath.2018.10.038.
FROM HUMAN PATHOLOGY
Key clinical point:
Major finding: Cortactin was expressed on 14 of 17 CLL samples vs. none of 16 MCL samples.
Study details: Immunohistochemistry analysis of samples from 131 patients with B-cell lineage non-Hodgkin lymphomas.
Disclosures: The study was internally supported. The authors reported having no conflicts of interest.
Source: Pizzi M et al. Hum Pathol. 2018 Nov 17. doi: 10.1016/j.humpath.2018.10.038.
Biosimilar deemed equivalent to rituximab in FL
Phase 3 results suggest the biosimilar product CT-P10 is equivalent to rituximab in patients with low-tumor-burden follicular lymphoma (FL).
Overall response rates were similar—both exceeding 80%—in patients who received CT-P10 and those who received rituximab.
In addition, adverse event (AE) profiles were comparable between the treatment arms.
Larry W. Kwak, MD, PhD, of City of Hope in Duarte, California, and his colleagues reported these results in The Lancet Haematology.
CT-P10 was approved by the European Commission in 2017 and was recommended for approval by the U.S. Food and Drug Administration’s Oncologic Drugs Advisory Committee last month.
The phase 3 trial of CT-P10 included 258 patients with stage II-IV low-tumor-burden FL. They were randomized to receive CT-P10 (n=130) or rituximab (n=128).
Patients received intravenous CT-P10 or rituximab weekly for 4 weeks as induction therapy. Patients experiencing disease control went on to a maintenance phase with their assigned treatment, given every 8 weeks for six cycles, followed by another year of maintenance therapy with CT-P10 for those still on study.
Efficacy
The overall response rate at 7 months was 83% in patients randomized to CT-P10 and 81% in those randomized to rituximab.
The complete response rates were 28% and 34%, respectively. The unconfirmed complete response rates were 5% and 2%, respectively. And the partial response rates were 51% and 46%, respectively.
The two treatments were deemed therapeutically equivalent, as the two-sided 90% confidence intervals for the difference in proportion of responders between CT-P10 and rituximab were within the prespecified equivalence margin of 17%.
Safety
Treatment-emergent AEs occurred in 71% of patients in the CT-P10 arm and 67% of those in the rituximab arm.
The most common treatment-emergent AEs (in the CT-P10 and rituximab arms, respectively) were:
- Infusion-related reactions (31% and 29%)
- Infections (27% and 21%)
- Worsening neutropenia (22% for both)
- Upper respiratory tract infection (12% and 11%)
- Worsening anemia (10% and 14%)
- Worsening thrombocytopenia (8% and 7%)
- Fatigue (7% and 9%)
- Diarrhea (5% for both)
- Nausea (5% for both)
- Urinary tract infection (4% and 5%)
- Headache (3% and 5%).
Serious AEs were reported in six patients in the CT-P10 arm and three patients in the rituximab arm.
Two serious AEs—myocardial infarction and constipation—in the CT-P10 arm were considered related to treatment. None of the serious AEs in the rituximab arm were considered treatment-related.
Two patients in the CT-P10 arm discontinued treatment due to AEs—one due to myocardial infarction and one due to dermatitis. There were no AE-related discontinuations in the rituximab arm.
There were two deaths in the CT-P10 arm as of the cutoff date (January 4, 2018). One was due to myocardial infarction, and one was due to respiratory failure. The myocardial infarction was considered possibly related to treatment.
This trial was sponsored by Celltrion, the company developing CT-P10. Three study authors are employees of the company.
Dr. Kwak and several other authors not employed by Celltrion reported disclosures related to the company. Authors also reported relationships with Novartis, Roche, AbbVie, Celgene, and Takeda, among other entities.
Phase 3 results suggest the biosimilar product CT-P10 is equivalent to rituximab in patients with low-tumor-burden follicular lymphoma (FL).
Overall response rates were similar—both exceeding 80%—in patients who received CT-P10 and those who received rituximab.
In addition, adverse event (AE) profiles were comparable between the treatment arms.
Larry W. Kwak, MD, PhD, of City of Hope in Duarte, California, and his colleagues reported these results in The Lancet Haematology.
CT-P10 was approved by the European Commission in 2017 and was recommended for approval by the U.S. Food and Drug Administration’s Oncologic Drugs Advisory Committee last month.
The phase 3 trial of CT-P10 included 258 patients with stage II-IV low-tumor-burden FL. They were randomized to receive CT-P10 (n=130) or rituximab (n=128).
Patients received intravenous CT-P10 or rituximab weekly for 4 weeks as induction therapy. Patients experiencing disease control went on to a maintenance phase with their assigned treatment, given every 8 weeks for six cycles, followed by another year of maintenance therapy with CT-P10 for those still on study.
Efficacy
The overall response rate at 7 months was 83% in patients randomized to CT-P10 and 81% in those randomized to rituximab.
The complete response rates were 28% and 34%, respectively. The unconfirmed complete response rates were 5% and 2%, respectively. And the partial response rates were 51% and 46%, respectively.
The two treatments were deemed therapeutically equivalent, as the two-sided 90% confidence intervals for the difference in proportion of responders between CT-P10 and rituximab were within the prespecified equivalence margin of 17%.
Safety
Treatment-emergent AEs occurred in 71% of patients in the CT-P10 arm and 67% of those in the rituximab arm.
The most common treatment-emergent AEs (in the CT-P10 and rituximab arms, respectively) were:
- Infusion-related reactions (31% and 29%)
- Infections (27% and 21%)
- Worsening neutropenia (22% for both)
- Upper respiratory tract infection (12% and 11%)
- Worsening anemia (10% and 14%)
- Worsening thrombocytopenia (8% and 7%)
- Fatigue (7% and 9%)
- Diarrhea (5% for both)
- Nausea (5% for both)
- Urinary tract infection (4% and 5%)
- Headache (3% and 5%).
Serious AEs were reported in six patients in the CT-P10 arm and three patients in the rituximab arm.
Two serious AEs—myocardial infarction and constipation—in the CT-P10 arm were considered related to treatment. None of the serious AEs in the rituximab arm were considered treatment-related.
Two patients in the CT-P10 arm discontinued treatment due to AEs—one due to myocardial infarction and one due to dermatitis. There were no AE-related discontinuations in the rituximab arm.
There were two deaths in the CT-P10 arm as of the cutoff date (January 4, 2018). One was due to myocardial infarction, and one was due to respiratory failure. The myocardial infarction was considered possibly related to treatment.
This trial was sponsored by Celltrion, the company developing CT-P10. Three study authors are employees of the company.
Dr. Kwak and several other authors not employed by Celltrion reported disclosures related to the company. Authors also reported relationships with Novartis, Roche, AbbVie, Celgene, and Takeda, among other entities.
Phase 3 results suggest the biosimilar product CT-P10 is equivalent to rituximab in patients with low-tumor-burden follicular lymphoma (FL).
Overall response rates were similar—both exceeding 80%—in patients who received CT-P10 and those who received rituximab.
In addition, adverse event (AE) profiles were comparable between the treatment arms.
Larry W. Kwak, MD, PhD, of City of Hope in Duarte, California, and his colleagues reported these results in The Lancet Haematology.
CT-P10 was approved by the European Commission in 2017 and was recommended for approval by the U.S. Food and Drug Administration’s Oncologic Drugs Advisory Committee last month.
The phase 3 trial of CT-P10 included 258 patients with stage II-IV low-tumor-burden FL. They were randomized to receive CT-P10 (n=130) or rituximab (n=128).
Patients received intravenous CT-P10 or rituximab weekly for 4 weeks as induction therapy. Patients experiencing disease control went on to a maintenance phase with their assigned treatment, given every 8 weeks for six cycles, followed by another year of maintenance therapy with CT-P10 for those still on study.
Efficacy
The overall response rate at 7 months was 83% in patients randomized to CT-P10 and 81% in those randomized to rituximab.
The complete response rates were 28% and 34%, respectively. The unconfirmed complete response rates were 5% and 2%, respectively. And the partial response rates were 51% and 46%, respectively.
The two treatments were deemed therapeutically equivalent, as the two-sided 90% confidence intervals for the difference in proportion of responders between CT-P10 and rituximab were within the prespecified equivalence margin of 17%.
Safety
Treatment-emergent AEs occurred in 71% of patients in the CT-P10 arm and 67% of those in the rituximab arm.
The most common treatment-emergent AEs (in the CT-P10 and rituximab arms, respectively) were:
- Infusion-related reactions (31% and 29%)
- Infections (27% and 21%)
- Worsening neutropenia (22% for both)
- Upper respiratory tract infection (12% and 11%)
- Worsening anemia (10% and 14%)
- Worsening thrombocytopenia (8% and 7%)
- Fatigue (7% and 9%)
- Diarrhea (5% for both)
- Nausea (5% for both)
- Urinary tract infection (4% and 5%)
- Headache (3% and 5%).
Serious AEs were reported in six patients in the CT-P10 arm and three patients in the rituximab arm.
Two serious AEs—myocardial infarction and constipation—in the CT-P10 arm were considered related to treatment. None of the serious AEs in the rituximab arm were considered treatment-related.
Two patients in the CT-P10 arm discontinued treatment due to AEs—one due to myocardial infarction and one due to dermatitis. There were no AE-related discontinuations in the rituximab arm.
There were two deaths in the CT-P10 arm as of the cutoff date (January 4, 2018). One was due to myocardial infarction, and one was due to respiratory failure. The myocardial infarction was considered possibly related to treatment.
This trial was sponsored by Celltrion, the company developing CT-P10. Three study authors are employees of the company.
Dr. Kwak and several other authors not employed by Celltrion reported disclosures related to the company. Authors also reported relationships with Novartis, Roche, AbbVie, Celgene, and Takeda, among other entities.
Elderly NHL patients have higher NRM after HSCT
A retrospective study suggests elderly patients with non-Hodgkin lymphoma (NHL) are more likely to die, but not relapse, within a year of allogeneic hematopoietic stem cell transplant (allo-HSCT).
The rate of non-relapse mortality (NRM) at 1 year was significantly higher for elderly patients than for middle-aged or young patients.
However, the 3-year rate of relapse was similar across the age groups.
Charalampia Kyriakou, MD, PhD, of University College London in the U.K., and her colleagues reported these findings in Biology of Blood and Marrow Transplantation.
The investigators analyzed 3,919 patients with NHL who underwent allo-HSCT between 2003 and 2013.
The patients had follicular lymphoma (n=1,461), diffuse large B-cell lymphoma (n=1,192), mantle cell lymphoma (n=823), and peripheral T-cell lymphoma (n=443).
At the time of transplant, about 85% of patients were chemo-sensitive, with the remainder being chemo-refractory.
Results
The investigators compared outcomes in patients assigned to three age groups—young (18-50), middle-aged (51-65), and elderly (66-77).
NRM at 1 year was 13% for young patients, 20% for middle-aged patients, and 33% for elderly patients (P<0.001).
Overall survival at 3 years was 60% in young patients, 54% in middle-aged patients, and 38% in the elderly (P<0.001).
In contrast to these significant associations between age and survival, the rate of relapse at 3 years remained relatively consistent—30% in young patients, 31% in middle-aged patients, and 28% in elderly patients (P=0.355).
The increased risk of NRM in elderly patients could not be fully explained by comorbidities, although these were more common in the elderly.
After analyzing information from a subset of patients, the investigators concluded that “the presence of comorbidities is a significant risk factor for NRM and survival, but this does not fully explain the outcome disadvantages in our [elderly] group.”
Therefore, age remains an independent risk factor.
The investigators did not report conflicts of interest.
A retrospective study suggests elderly patients with non-Hodgkin lymphoma (NHL) are more likely to die, but not relapse, within a year of allogeneic hematopoietic stem cell transplant (allo-HSCT).
The rate of non-relapse mortality (NRM) at 1 year was significantly higher for elderly patients than for middle-aged or young patients.
However, the 3-year rate of relapse was similar across the age groups.
Charalampia Kyriakou, MD, PhD, of University College London in the U.K., and her colleagues reported these findings in Biology of Blood and Marrow Transplantation.
The investigators analyzed 3,919 patients with NHL who underwent allo-HSCT between 2003 and 2013.
The patients had follicular lymphoma (n=1,461), diffuse large B-cell lymphoma (n=1,192), mantle cell lymphoma (n=823), and peripheral T-cell lymphoma (n=443).
At the time of transplant, about 85% of patients were chemo-sensitive, with the remainder being chemo-refractory.
Results
The investigators compared outcomes in patients assigned to three age groups—young (18-50), middle-aged (51-65), and elderly (66-77).
NRM at 1 year was 13% for young patients, 20% for middle-aged patients, and 33% for elderly patients (P<0.001).
Overall survival at 3 years was 60% in young patients, 54% in middle-aged patients, and 38% in the elderly (P<0.001).
In contrast to these significant associations between age and survival, the rate of relapse at 3 years remained relatively consistent—30% in young patients, 31% in middle-aged patients, and 28% in elderly patients (P=0.355).
The increased risk of NRM in elderly patients could not be fully explained by comorbidities, although these were more common in the elderly.
After analyzing information from a subset of patients, the investigators concluded that “the presence of comorbidities is a significant risk factor for NRM and survival, but this does not fully explain the outcome disadvantages in our [elderly] group.”
Therefore, age remains an independent risk factor.
The investigators did not report conflicts of interest.
A retrospective study suggests elderly patients with non-Hodgkin lymphoma (NHL) are more likely to die, but not relapse, within a year of allogeneic hematopoietic stem cell transplant (allo-HSCT).
The rate of non-relapse mortality (NRM) at 1 year was significantly higher for elderly patients than for middle-aged or young patients.
However, the 3-year rate of relapse was similar across the age groups.
Charalampia Kyriakou, MD, PhD, of University College London in the U.K., and her colleagues reported these findings in Biology of Blood and Marrow Transplantation.
The investigators analyzed 3,919 patients with NHL who underwent allo-HSCT between 2003 and 2013.
The patients had follicular lymphoma (n=1,461), diffuse large B-cell lymphoma (n=1,192), mantle cell lymphoma (n=823), and peripheral T-cell lymphoma (n=443).
At the time of transplant, about 85% of patients were chemo-sensitive, with the remainder being chemo-refractory.
Results
The investigators compared outcomes in patients assigned to three age groups—young (18-50), middle-aged (51-65), and elderly (66-77).
NRM at 1 year was 13% for young patients, 20% for middle-aged patients, and 33% for elderly patients (P<0.001).
Overall survival at 3 years was 60% in young patients, 54% in middle-aged patients, and 38% in the elderly (P<0.001).
In contrast to these significant associations between age and survival, the rate of relapse at 3 years remained relatively consistent—30% in young patients, 31% in middle-aged patients, and 28% in elderly patients (P=0.355).
The increased risk of NRM in elderly patients could not be fully explained by comorbidities, although these were more common in the elderly.
After analyzing information from a subset of patients, the investigators concluded that “the presence of comorbidities is a significant risk factor for NRM and survival, but this does not fully explain the outcome disadvantages in our [elderly] group.”
Therefore, age remains an independent risk factor.
The investigators did not report conflicts of interest.
Rituximab biosimilar looks equivalent in follicular lymphoma
The rituximab biosimilar CT-P10 has equivalent efficacy, compared with rituximab, and is well tolerated in the treatment of low–tumor-burden follicular lymphoma, according to results from a multinational, randomized, phase 3 study.
Overall response after 7 months of treatment exceeded 80% for patients assigned to CT-P10 and for those assigned to rituximab, investigators reported in the Lancet Haematology.
Adverse event profiles were comparable for rituximab and the biosimilar over that time period, while pharmacokinetics, pharmacodynamics, and immunogenicity were likewise comparable between arms, according to investigators.
“Thus, CT-P10 monotherapy is suggested as a new therapeutic option for patients with low–tumor-burden follicular lymphoma,” wrote senior author Larry W Kwak, MD, PhD, of the Comprehensive Cancer Center, City of Hope, Duarte, Calif., and his colleagues.
CT-P10, the first rituximab biosimilar to be authorized by the European Medicines Agency, has been recommended for approval in the United States by the Food and Drug Administration’s Oncologic Drugs Advisory Committee.
If approved by the FDA, CT-P10 would be the first rituximab biosimilar available in the United States, according to the company, which noted three proposed indications in non-Hodgkin lymphoma.
In the current randomized, double-blind, parallel-group, phase 3 trial, 258 patients with stage II-IV low–tumor-burden follicular lymphoma were randomly assigned to CT-P10 (130 patients) or rituximab sourced in the United States (128 patients).
Treatment consisted of an induction period of intravenous CT-P10 or rituximab weekly for 4 weeks, while patients experiencing disease control went on to a maintenance phase with their assigned treatment given every 8 weeks for six cycles, followed by another year of maintenance therapy with CT-P10 for those still on study.
The primary endpoint of the study was overall response at 7 months, defined as a complete response, unconfirmed complete response, or partial response.
Overall response was seen in 83% of patients randomized to CT-P10 and 81% of patients randomized to rituximab at 7 months, Dr. Kwak and his colleagues reported.
The two treatments were deemed therapeutically equivalent, as illustrated by 90% confidence intervals within a prespecified equivalence margin of 17%, investigators said.
The most common treatment-emergent adverse events in either group were infusion-related reactions, which were of grade 1-2, except for one grade 3 reaction reported in the CT-P10 group, according to the report. Other common adverse events were upper respiratory tract infections and fatigue.
Serious adverse events were reported in six patients in the CT-P10 arm and three patients in the rituximab arm.
The availability of a rituximab biosimilar is anticipated to reduce the cost of treatment and improve patient access, according to investigators.
Introduction of CT-P10 in the European Union was projected to save between 90 and 150 million euros over a year, enabling more than 12,500 new patients to be treated with the biosimilar, according to results of a budget impact analysis investigators cited in their report.
“Widespread adoption of a rituximab biosimilar could have a substantial effect on health care budgets and might also have effects at a societal level,” Dr. Kwak and his coauthors said in the report.
The trial was sponsored by Celltrion and three coauthors of the study were employees of the company. Dr. Kwak and several other coinvestigators not employed by Celltrion reported disclosures related to the company. Other disclosures provided related to Novartis, Roche, AbbVie, Celgene, and Takeda, among other entities.
SOURCE: Ogura M et al. Lancet Haematol. 2018 Nov;5(11):e543-53.
The rituximab biosimilar CT-P10 has equivalent efficacy, compared with rituximab, and is well tolerated in the treatment of low–tumor-burden follicular lymphoma, according to results from a multinational, randomized, phase 3 study.
Overall response after 7 months of treatment exceeded 80% for patients assigned to CT-P10 and for those assigned to rituximab, investigators reported in the Lancet Haematology.
Adverse event profiles were comparable for rituximab and the biosimilar over that time period, while pharmacokinetics, pharmacodynamics, and immunogenicity were likewise comparable between arms, according to investigators.
“Thus, CT-P10 monotherapy is suggested as a new therapeutic option for patients with low–tumor-burden follicular lymphoma,” wrote senior author Larry W Kwak, MD, PhD, of the Comprehensive Cancer Center, City of Hope, Duarte, Calif., and his colleagues.
CT-P10, the first rituximab biosimilar to be authorized by the European Medicines Agency, has been recommended for approval in the United States by the Food and Drug Administration’s Oncologic Drugs Advisory Committee.
If approved by the FDA, CT-P10 would be the first rituximab biosimilar available in the United States, according to the company, which noted three proposed indications in non-Hodgkin lymphoma.
In the current randomized, double-blind, parallel-group, phase 3 trial, 258 patients with stage II-IV low–tumor-burden follicular lymphoma were randomly assigned to CT-P10 (130 patients) or rituximab sourced in the United States (128 patients).
Treatment consisted of an induction period of intravenous CT-P10 or rituximab weekly for 4 weeks, while patients experiencing disease control went on to a maintenance phase with their assigned treatment given every 8 weeks for six cycles, followed by another year of maintenance therapy with CT-P10 for those still on study.
The primary endpoint of the study was overall response at 7 months, defined as a complete response, unconfirmed complete response, or partial response.
Overall response was seen in 83% of patients randomized to CT-P10 and 81% of patients randomized to rituximab at 7 months, Dr. Kwak and his colleagues reported.
The two treatments were deemed therapeutically equivalent, as illustrated by 90% confidence intervals within a prespecified equivalence margin of 17%, investigators said.
The most common treatment-emergent adverse events in either group were infusion-related reactions, which were of grade 1-2, except for one grade 3 reaction reported in the CT-P10 group, according to the report. Other common adverse events were upper respiratory tract infections and fatigue.
Serious adverse events were reported in six patients in the CT-P10 arm and three patients in the rituximab arm.
The availability of a rituximab biosimilar is anticipated to reduce the cost of treatment and improve patient access, according to investigators.
Introduction of CT-P10 in the European Union was projected to save between 90 and 150 million euros over a year, enabling more than 12,500 new patients to be treated with the biosimilar, according to results of a budget impact analysis investigators cited in their report.
“Widespread adoption of a rituximab biosimilar could have a substantial effect on health care budgets and might also have effects at a societal level,” Dr. Kwak and his coauthors said in the report.
The trial was sponsored by Celltrion and three coauthors of the study were employees of the company. Dr. Kwak and several other coinvestigators not employed by Celltrion reported disclosures related to the company. Other disclosures provided related to Novartis, Roche, AbbVie, Celgene, and Takeda, among other entities.
SOURCE: Ogura M et al. Lancet Haematol. 2018 Nov;5(11):e543-53.
The rituximab biosimilar CT-P10 has equivalent efficacy, compared with rituximab, and is well tolerated in the treatment of low–tumor-burden follicular lymphoma, according to results from a multinational, randomized, phase 3 study.
Overall response after 7 months of treatment exceeded 80% for patients assigned to CT-P10 and for those assigned to rituximab, investigators reported in the Lancet Haematology.
Adverse event profiles were comparable for rituximab and the biosimilar over that time period, while pharmacokinetics, pharmacodynamics, and immunogenicity were likewise comparable between arms, according to investigators.
“Thus, CT-P10 monotherapy is suggested as a new therapeutic option for patients with low–tumor-burden follicular lymphoma,” wrote senior author Larry W Kwak, MD, PhD, of the Comprehensive Cancer Center, City of Hope, Duarte, Calif., and his colleagues.
CT-P10, the first rituximab biosimilar to be authorized by the European Medicines Agency, has been recommended for approval in the United States by the Food and Drug Administration’s Oncologic Drugs Advisory Committee.
If approved by the FDA, CT-P10 would be the first rituximab biosimilar available in the United States, according to the company, which noted three proposed indications in non-Hodgkin lymphoma.
In the current randomized, double-blind, parallel-group, phase 3 trial, 258 patients with stage II-IV low–tumor-burden follicular lymphoma were randomly assigned to CT-P10 (130 patients) or rituximab sourced in the United States (128 patients).
Treatment consisted of an induction period of intravenous CT-P10 or rituximab weekly for 4 weeks, while patients experiencing disease control went on to a maintenance phase with their assigned treatment given every 8 weeks for six cycles, followed by another year of maintenance therapy with CT-P10 for those still on study.
The primary endpoint of the study was overall response at 7 months, defined as a complete response, unconfirmed complete response, or partial response.
Overall response was seen in 83% of patients randomized to CT-P10 and 81% of patients randomized to rituximab at 7 months, Dr. Kwak and his colleagues reported.
The two treatments were deemed therapeutically equivalent, as illustrated by 90% confidence intervals within a prespecified equivalence margin of 17%, investigators said.
The most common treatment-emergent adverse events in either group were infusion-related reactions, which were of grade 1-2, except for one grade 3 reaction reported in the CT-P10 group, according to the report. Other common adverse events were upper respiratory tract infections and fatigue.
Serious adverse events were reported in six patients in the CT-P10 arm and three patients in the rituximab arm.
The availability of a rituximab biosimilar is anticipated to reduce the cost of treatment and improve patient access, according to investigators.
Introduction of CT-P10 in the European Union was projected to save between 90 and 150 million euros over a year, enabling more than 12,500 new patients to be treated with the biosimilar, according to results of a budget impact analysis investigators cited in their report.
“Widespread adoption of a rituximab biosimilar could have a substantial effect on health care budgets and might also have effects at a societal level,” Dr. Kwak and his coauthors said in the report.
The trial was sponsored by Celltrion and three coauthors of the study were employees of the company. Dr. Kwak and several other coinvestigators not employed by Celltrion reported disclosures related to the company. Other disclosures provided related to Novartis, Roche, AbbVie, Celgene, and Takeda, among other entities.
SOURCE: Ogura M et al. Lancet Haematol. 2018 Nov;5(11):e543-53.
FROM LANCET HAEMATOLOGY
Key clinical point:
Major finding: Overall response after 7 months of treatment was seen in 83% of patients randomized to CT-P10 and 81% of patients randomized to rituximab.
Study details: Analysis of 258 patients randomized to CT-P10 or rituximab in a phase 3, double-blind, parallel-group trial.
Disclosures: The trial was sponsored by Celltrion and three coauthors of the study were employees of the company. Other study coauthors reported disclosures related to Celltrion, Novartis, Roche, AbbVie, Celgene, and Takeda, among other companies.
Source: Ogura M et al. Lancet Haematol. 2018 Nov;5(11):e543-53.
R-CHOP effective as first-line treatment in FL
Long-term data suggest R-CHOP can be effective as first-line treatment for patients with follicular lymphoma (FL).
In a phase 2-3 trial, investigators compared R-CHOP-21 and R-CHOP-14 in a cohort of patients with indolent lymphomas, most of whom had FL.
Ten-year survival rates were similar between the R-CHOP-21 and R-CHOP-14 groups, with progression-free survival (PFS) rates of 33% and 39%, respectively, and overall survival (OS) rates of 81% and 85%, respectively.
The investigators did note that 9% of patients in each treatment group developed secondary malignancies, and grade 3 infections were a concern as well.
Takashi Watanabe, MD, PhD, of Mie University in Japan, and his colleagues reported these results in The Lancet Haematology.
The trial (JCOG0203) included 300 patients with stage III or IV indolent B-cell lymphomas from 44 Japanese hospitals.
Most patients (n=248) had grade 1-3a FL, 17 had grade 3b FL, 6 had marginal zone lymphoma, 6 had diffuse large B-cell lymphoma, 4 had mantle cell lymphoma, 2 had small lymphocytic lymphoma, 1 had plasmacytoma, 13 had other indolent B-cell lymphomas, and 3 had other lymphomas.
The patients were randomly assigned to receive six cycles of R-CHOP 21 (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone every 3 weeks) or R-CHOP 14 (R-CHOP every 2 weeks with granulocyte-colony stimulating factor support). Neither group received rituximab maintenance.
Overall results
The median follow-up was 11.2 years (interquartile range, 10.1 to 12.7 years).
The 10-year PFS was 33% in the R-CHOP-21 group and 39% in the R-CHOP-14 group (hazard ratio=0.89). The 10-year OS was 81% and 85%, respectively (hazard ratio=0.87).
At 10 years, the incidence of secondary malignancies was 9% in both the R-CHOP-21 group (14/148) and the R-CHOP-14 group (14/151).
The most frequent solid tumor malignancies were stomach (n=5), lung (n=4), colon (n=3), bladder (n=2), and prostate (n=2) cancers. Hematologic malignancies included myelodysplastic syndromes (n=6), acute myeloid leukemia (n=2), acute lymphoblastic leukemia (n=1), and chronic myeloid leukemia (n=1).
There were nine deaths from secondary malignancies, four in the R-CHOP-21 group and five in the R-CHOP-14 group.
The rate of grade 3 adverse events was 18% (n=53) for the entire cohort. Grade 3 infections occurred in 23% of the R-CHOP-21 group and 12% of the R-CHOP-14 group.
Focus on grade 1-3a FL
Among the 248 patients with grade 1-3a FL, the PFS (for both treatment groups) was 45% at 5 years, 39% at 8 years, and 36% at 10 years. The OS was 94% at 5 years, 87% at 8 years, and 85% at 10 years.
Histological transformation was observed in 11% of the patients who had grade 1-3a FL at enrollment. The cumulative incidence of histological transformation was 2.4% at 3 years, 3.2% at 5 years, 8.5% at 8 years, and 9.3% at 10 years.
Secondary malignancies occurred in 10% (12/125) of the R-CHOP-21 group and 11% (13/123) of the R-CHOP-14 group.
The cumulative incidence of hematologic secondary malignancies at 10 years was 2.9%.
The investigators noted that the actual incidence of secondary solid tumors or hematologic malignancies apart from the setting of autologous stem cell transplants is not known. They emphasized that patients should be followed beyond 10 years to ensure the risk of secondary malignancies is not underestimated.
“Clinicians choosing a first-line treatment for patients with follicular lymphoma should be cautious of secondary malignancies caused by immunochemotherapy and severe complications of infectious diseases in the long-term follow-up—both of which could lead to death,” the investigators wrote.
This study was supported by the Ministry of Health, Labour and Welfare of Japan and the National Cancer Center Research and Development Fund of Japan.
Dr. Wantanabe has received honoraria from Bristol-Myers Squibb, Takeda, Taisho Toyama, Celgene, Nippon Shinyaku, and Novartis and funding resources from TakaraBio and United Immunity to support the Department of Immuno-Gene Therapy at Mie University. Multiple co-authors reported similar relationships.
Long-term data suggest R-CHOP can be effective as first-line treatment for patients with follicular lymphoma (FL).
In a phase 2-3 trial, investigators compared R-CHOP-21 and R-CHOP-14 in a cohort of patients with indolent lymphomas, most of whom had FL.
Ten-year survival rates were similar between the R-CHOP-21 and R-CHOP-14 groups, with progression-free survival (PFS) rates of 33% and 39%, respectively, and overall survival (OS) rates of 81% and 85%, respectively.
The investigators did note that 9% of patients in each treatment group developed secondary malignancies, and grade 3 infections were a concern as well.
Takashi Watanabe, MD, PhD, of Mie University in Japan, and his colleagues reported these results in The Lancet Haematology.
The trial (JCOG0203) included 300 patients with stage III or IV indolent B-cell lymphomas from 44 Japanese hospitals.
Most patients (n=248) had grade 1-3a FL, 17 had grade 3b FL, 6 had marginal zone lymphoma, 6 had diffuse large B-cell lymphoma, 4 had mantle cell lymphoma, 2 had small lymphocytic lymphoma, 1 had plasmacytoma, 13 had other indolent B-cell lymphomas, and 3 had other lymphomas.
The patients were randomly assigned to receive six cycles of R-CHOP 21 (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone every 3 weeks) or R-CHOP 14 (R-CHOP every 2 weeks with granulocyte-colony stimulating factor support). Neither group received rituximab maintenance.
Overall results
The median follow-up was 11.2 years (interquartile range, 10.1 to 12.7 years).
The 10-year PFS was 33% in the R-CHOP-21 group and 39% in the R-CHOP-14 group (hazard ratio=0.89). The 10-year OS was 81% and 85%, respectively (hazard ratio=0.87).
At 10 years, the incidence of secondary malignancies was 9% in both the R-CHOP-21 group (14/148) and the R-CHOP-14 group (14/151).
The most frequent solid tumor malignancies were stomach (n=5), lung (n=4), colon (n=3), bladder (n=2), and prostate (n=2) cancers. Hematologic malignancies included myelodysplastic syndromes (n=6), acute myeloid leukemia (n=2), acute lymphoblastic leukemia (n=1), and chronic myeloid leukemia (n=1).
There were nine deaths from secondary malignancies, four in the R-CHOP-21 group and five in the R-CHOP-14 group.
The rate of grade 3 adverse events was 18% (n=53) for the entire cohort. Grade 3 infections occurred in 23% of the R-CHOP-21 group and 12% of the R-CHOP-14 group.
Focus on grade 1-3a FL
Among the 248 patients with grade 1-3a FL, the PFS (for both treatment groups) was 45% at 5 years, 39% at 8 years, and 36% at 10 years. The OS was 94% at 5 years, 87% at 8 years, and 85% at 10 years.
Histological transformation was observed in 11% of the patients who had grade 1-3a FL at enrollment. The cumulative incidence of histological transformation was 2.4% at 3 years, 3.2% at 5 years, 8.5% at 8 years, and 9.3% at 10 years.
Secondary malignancies occurred in 10% (12/125) of the R-CHOP-21 group and 11% (13/123) of the R-CHOP-14 group.
The cumulative incidence of hematologic secondary malignancies at 10 years was 2.9%.
The investigators noted that the actual incidence of secondary solid tumors or hematologic malignancies apart from the setting of autologous stem cell transplants is not known. They emphasized that patients should be followed beyond 10 years to ensure the risk of secondary malignancies is not underestimated.
“Clinicians choosing a first-line treatment for patients with follicular lymphoma should be cautious of secondary malignancies caused by immunochemotherapy and severe complications of infectious diseases in the long-term follow-up—both of which could lead to death,” the investigators wrote.
This study was supported by the Ministry of Health, Labour and Welfare of Japan and the National Cancer Center Research and Development Fund of Japan.
Dr. Wantanabe has received honoraria from Bristol-Myers Squibb, Takeda, Taisho Toyama, Celgene, Nippon Shinyaku, and Novartis and funding resources from TakaraBio and United Immunity to support the Department of Immuno-Gene Therapy at Mie University. Multiple co-authors reported similar relationships.
Long-term data suggest R-CHOP can be effective as first-line treatment for patients with follicular lymphoma (FL).
In a phase 2-3 trial, investigators compared R-CHOP-21 and R-CHOP-14 in a cohort of patients with indolent lymphomas, most of whom had FL.
Ten-year survival rates were similar between the R-CHOP-21 and R-CHOP-14 groups, with progression-free survival (PFS) rates of 33% and 39%, respectively, and overall survival (OS) rates of 81% and 85%, respectively.
The investigators did note that 9% of patients in each treatment group developed secondary malignancies, and grade 3 infections were a concern as well.
Takashi Watanabe, MD, PhD, of Mie University in Japan, and his colleagues reported these results in The Lancet Haematology.
The trial (JCOG0203) included 300 patients with stage III or IV indolent B-cell lymphomas from 44 Japanese hospitals.
Most patients (n=248) had grade 1-3a FL, 17 had grade 3b FL, 6 had marginal zone lymphoma, 6 had diffuse large B-cell lymphoma, 4 had mantle cell lymphoma, 2 had small lymphocytic lymphoma, 1 had plasmacytoma, 13 had other indolent B-cell lymphomas, and 3 had other lymphomas.
The patients were randomly assigned to receive six cycles of R-CHOP 21 (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone every 3 weeks) or R-CHOP 14 (R-CHOP every 2 weeks with granulocyte-colony stimulating factor support). Neither group received rituximab maintenance.
Overall results
The median follow-up was 11.2 years (interquartile range, 10.1 to 12.7 years).
The 10-year PFS was 33% in the R-CHOP-21 group and 39% in the R-CHOP-14 group (hazard ratio=0.89). The 10-year OS was 81% and 85%, respectively (hazard ratio=0.87).
At 10 years, the incidence of secondary malignancies was 9% in both the R-CHOP-21 group (14/148) and the R-CHOP-14 group (14/151).
The most frequent solid tumor malignancies were stomach (n=5), lung (n=4), colon (n=3), bladder (n=2), and prostate (n=2) cancers. Hematologic malignancies included myelodysplastic syndromes (n=6), acute myeloid leukemia (n=2), acute lymphoblastic leukemia (n=1), and chronic myeloid leukemia (n=1).
There were nine deaths from secondary malignancies, four in the R-CHOP-21 group and five in the R-CHOP-14 group.
The rate of grade 3 adverse events was 18% (n=53) for the entire cohort. Grade 3 infections occurred in 23% of the R-CHOP-21 group and 12% of the R-CHOP-14 group.
Focus on grade 1-3a FL
Among the 248 patients with grade 1-3a FL, the PFS (for both treatment groups) was 45% at 5 years, 39% at 8 years, and 36% at 10 years. The OS was 94% at 5 years, 87% at 8 years, and 85% at 10 years.
Histological transformation was observed in 11% of the patients who had grade 1-3a FL at enrollment. The cumulative incidence of histological transformation was 2.4% at 3 years, 3.2% at 5 years, 8.5% at 8 years, and 9.3% at 10 years.
Secondary malignancies occurred in 10% (12/125) of the R-CHOP-21 group and 11% (13/123) of the R-CHOP-14 group.
The cumulative incidence of hematologic secondary malignancies at 10 years was 2.9%.
The investigators noted that the actual incidence of secondary solid tumors or hematologic malignancies apart from the setting of autologous stem cell transplants is not known. They emphasized that patients should be followed beyond 10 years to ensure the risk of secondary malignancies is not underestimated.
“Clinicians choosing a first-line treatment for patients with follicular lymphoma should be cautious of secondary malignancies caused by immunochemotherapy and severe complications of infectious diseases in the long-term follow-up—both of which could lead to death,” the investigators wrote.
This study was supported by the Ministry of Health, Labour and Welfare of Japan and the National Cancer Center Research and Development Fund of Japan.
Dr. Wantanabe has received honoraria from Bristol-Myers Squibb, Takeda, Taisho Toyama, Celgene, Nippon Shinyaku, and Novartis and funding resources from TakaraBio and United Immunity to support the Department of Immuno-Gene Therapy at Mie University. Multiple co-authors reported similar relationships.
R-CHOP looks viable as first line in follicular lymphoma
A decade of follow-up data suggest that patients with newly diagnosed follicular lymphoma may derive long-term benefit from first-line therapy with the R-CHOP regimen, according to investigators in Japan.
Among patients with untreated follicular and other indolent B-cell lymphomas enrolled in a randomized phase 2/3 trial, the 10-year progression-free survival (PFS) rate for patients assigned to R-CHOP-21 (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone every 3 weeks) was 33%, and the 10-year PFS rate for patients assigned to R-CHOP-14 (R-CHOP every 2 weeks with granulocyte colony-stimulating factor [G-CSF] support] was 39%, and these PFS rates did not differ between the treatment arms, Takashi Watanabe, MD, PhD, from Mie University in Tsu, Japan, and his colleagues reported in the Lancet Haematology.
They also found that, compared with therapeutic regimens prior to the introduction of rituximab, R-CHOP was associated with a reduced likelihood of histological transformation of follicular lymphoma into a poor-prognosis diffuse large B-cell lymphoma (DLBCL), with no apparent increase in risk of secondary malignancies.
“The gold-standard, first-line treatment in patients with advanced-stage follicular lymphoma remains undetermined. However, because of the reduction in incidence of transformation without an increase in either secondary malignancies or fatal infectious events, the R-CHOP regimen should be a candidate for standard treatment, particularly from the viewpoint of long-term follow-up,” the investigators wrote
The JCOG0203 trial, which began enrollment in September 2002, included patients with stage III or IV indolent B-cell lymphomas, including grades 1-3 follicular lymphoma, from 44 Japanese hospitals. The patients were randomly assigned to receive six cycles of either R-CHOP-14 plus G-CSF or R-CHOP-21 administered once daily for 6 days beginning on day 8 of every cycle). Patients did not receive rituximab maintenance in either group.
In the primary analysis of the trial, published in 2011, the investigators reported that in 299 patients there were no significant differences in either PFS or 6-year overall survival (OS).
In the current analysis, the investigators reported that, for 248 patients with grade 1-3a follicular lymphoma, the 8-year PFS rate was 39% and the 10-year PFS rate was 36%.
The cumulative incidence of histological transformation was 3.2% at 5 years, 8.5% at 8 years, and 9.3% 10 years after the last patient was enrolled.
“In our study, survival after histological transformation was still poor; therefore, reducing histological transformation remains a crucial issue for patients with follicular lymphoma,” the investigators wrote.
The cumulative incidence of secondary malignancies at 10 years was 8.1%, and the cumulative incidence of hematological secondary malignancies was 2.9%.
The investigators noted that the actual incidence of secondary solid tumors or hematologic malignancies apart from the setting of autologous stem cell transplants is not known and emphasized that patients should be followed beyond 10 years to ensure that the risk of secondary malignancies is not underestimated.
“Clinicians choosing a first-line treatment for patients with follicular lymphoma should be cautious of secondary malignancies caused by immunochemotherapy and severe complications of infectious diseases in the long-term follow-up – both of which could lead to death,” they advised.
The study was supported by the Ministry of Health, Labour and Welfare of Japan and by the National Cancer Center Research and Development Fund of Japan. Dr. Watanabe has received honoraria from Bristol-Myers Squibb, Takeda, Taisho Toyama, Celgene, Nippon Shinyaku, and Novartis and funding resources from TakaraBio and United Immunity to support the Department of Immuno-Gene Therapy at Mie University. Multiple coauthors reported similar relationships.
SOURCE: Watanabe T et al. Lancet Haematol. 2018 Nov;5(11):e520-31.
A decade of follow-up data suggest that patients with newly diagnosed follicular lymphoma may derive long-term benefit from first-line therapy with the R-CHOP regimen, according to investigators in Japan.
Among patients with untreated follicular and other indolent B-cell lymphomas enrolled in a randomized phase 2/3 trial, the 10-year progression-free survival (PFS) rate for patients assigned to R-CHOP-21 (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone every 3 weeks) was 33%, and the 10-year PFS rate for patients assigned to R-CHOP-14 (R-CHOP every 2 weeks with granulocyte colony-stimulating factor [G-CSF] support] was 39%, and these PFS rates did not differ between the treatment arms, Takashi Watanabe, MD, PhD, from Mie University in Tsu, Japan, and his colleagues reported in the Lancet Haematology.
They also found that, compared with therapeutic regimens prior to the introduction of rituximab, R-CHOP was associated with a reduced likelihood of histological transformation of follicular lymphoma into a poor-prognosis diffuse large B-cell lymphoma (DLBCL), with no apparent increase in risk of secondary malignancies.
“The gold-standard, first-line treatment in patients with advanced-stage follicular lymphoma remains undetermined. However, because of the reduction in incidence of transformation without an increase in either secondary malignancies or fatal infectious events, the R-CHOP regimen should be a candidate for standard treatment, particularly from the viewpoint of long-term follow-up,” the investigators wrote
The JCOG0203 trial, which began enrollment in September 2002, included patients with stage III or IV indolent B-cell lymphomas, including grades 1-3 follicular lymphoma, from 44 Japanese hospitals. The patients were randomly assigned to receive six cycles of either R-CHOP-14 plus G-CSF or R-CHOP-21 administered once daily for 6 days beginning on day 8 of every cycle). Patients did not receive rituximab maintenance in either group.
In the primary analysis of the trial, published in 2011, the investigators reported that in 299 patients there were no significant differences in either PFS or 6-year overall survival (OS).
In the current analysis, the investigators reported that, for 248 patients with grade 1-3a follicular lymphoma, the 8-year PFS rate was 39% and the 10-year PFS rate was 36%.
The cumulative incidence of histological transformation was 3.2% at 5 years, 8.5% at 8 years, and 9.3% 10 years after the last patient was enrolled.
“In our study, survival after histological transformation was still poor; therefore, reducing histological transformation remains a crucial issue for patients with follicular lymphoma,” the investigators wrote.
The cumulative incidence of secondary malignancies at 10 years was 8.1%, and the cumulative incidence of hematological secondary malignancies was 2.9%.
The investigators noted that the actual incidence of secondary solid tumors or hematologic malignancies apart from the setting of autologous stem cell transplants is not known and emphasized that patients should be followed beyond 10 years to ensure that the risk of secondary malignancies is not underestimated.
“Clinicians choosing a first-line treatment for patients with follicular lymphoma should be cautious of secondary malignancies caused by immunochemotherapy and severe complications of infectious diseases in the long-term follow-up – both of which could lead to death,” they advised.
The study was supported by the Ministry of Health, Labour and Welfare of Japan and by the National Cancer Center Research and Development Fund of Japan. Dr. Watanabe has received honoraria from Bristol-Myers Squibb, Takeda, Taisho Toyama, Celgene, Nippon Shinyaku, and Novartis and funding resources from TakaraBio and United Immunity to support the Department of Immuno-Gene Therapy at Mie University. Multiple coauthors reported similar relationships.
SOURCE: Watanabe T et al. Lancet Haematol. 2018 Nov;5(11):e520-31.
A decade of follow-up data suggest that patients with newly diagnosed follicular lymphoma may derive long-term benefit from first-line therapy with the R-CHOP regimen, according to investigators in Japan.
Among patients with untreated follicular and other indolent B-cell lymphomas enrolled in a randomized phase 2/3 trial, the 10-year progression-free survival (PFS) rate for patients assigned to R-CHOP-21 (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone every 3 weeks) was 33%, and the 10-year PFS rate for patients assigned to R-CHOP-14 (R-CHOP every 2 weeks with granulocyte colony-stimulating factor [G-CSF] support] was 39%, and these PFS rates did not differ between the treatment arms, Takashi Watanabe, MD, PhD, from Mie University in Tsu, Japan, and his colleagues reported in the Lancet Haematology.
They also found that, compared with therapeutic regimens prior to the introduction of rituximab, R-CHOP was associated with a reduced likelihood of histological transformation of follicular lymphoma into a poor-prognosis diffuse large B-cell lymphoma (DLBCL), with no apparent increase in risk of secondary malignancies.
“The gold-standard, first-line treatment in patients with advanced-stage follicular lymphoma remains undetermined. However, because of the reduction in incidence of transformation without an increase in either secondary malignancies or fatal infectious events, the R-CHOP regimen should be a candidate for standard treatment, particularly from the viewpoint of long-term follow-up,” the investigators wrote
The JCOG0203 trial, which began enrollment in September 2002, included patients with stage III or IV indolent B-cell lymphomas, including grades 1-3 follicular lymphoma, from 44 Japanese hospitals. The patients were randomly assigned to receive six cycles of either R-CHOP-14 plus G-CSF or R-CHOP-21 administered once daily for 6 days beginning on day 8 of every cycle). Patients did not receive rituximab maintenance in either group.
In the primary analysis of the trial, published in 2011, the investigators reported that in 299 patients there were no significant differences in either PFS or 6-year overall survival (OS).
In the current analysis, the investigators reported that, for 248 patients with grade 1-3a follicular lymphoma, the 8-year PFS rate was 39% and the 10-year PFS rate was 36%.
The cumulative incidence of histological transformation was 3.2% at 5 years, 8.5% at 8 years, and 9.3% 10 years after the last patient was enrolled.
“In our study, survival after histological transformation was still poor; therefore, reducing histological transformation remains a crucial issue for patients with follicular lymphoma,” the investigators wrote.
The cumulative incidence of secondary malignancies at 10 years was 8.1%, and the cumulative incidence of hematological secondary malignancies was 2.9%.
The investigators noted that the actual incidence of secondary solid tumors or hematologic malignancies apart from the setting of autologous stem cell transplants is not known and emphasized that patients should be followed beyond 10 years to ensure that the risk of secondary malignancies is not underestimated.
“Clinicians choosing a first-line treatment for patients with follicular lymphoma should be cautious of secondary malignancies caused by immunochemotherapy and severe complications of infectious diseases in the long-term follow-up – both of which could lead to death,” they advised.
The study was supported by the Ministry of Health, Labour and Welfare of Japan and by the National Cancer Center Research and Development Fund of Japan. Dr. Watanabe has received honoraria from Bristol-Myers Squibb, Takeda, Taisho Toyama, Celgene, Nippon Shinyaku, and Novartis and funding resources from TakaraBio and United Immunity to support the Department of Immuno-Gene Therapy at Mie University. Multiple coauthors reported similar relationships.
SOURCE: Watanabe T et al. Lancet Haematol. 2018 Nov;5(11):e520-31.
FROM LANCET HAEMATOLOGY
Key clinical point:
Major finding: Approximately one-third of patients treated with R-CHOP-14 plus G-CSF or R-CHOP-21 had no disease progression after 10 years of follow-up.
Study details: Long-term analysis of a randomized phase 2/3 trial in 299 patients with indolent B-cell lymphomas, including follicular lymphoma.
Disclosures: The study was supported by the Ministry of Health, Labor and Welfare of Japan and by the National Cancer Center Research and Development Fund of Japan. Dr. Wantanabe has received honoraria from Bristol-Myers Squibb, Takeda, Taisho Toyama, Celgene, Nippon Shinyaku, and Novartis and funding resources from TakaraBio and United Immunity to support the Department of Immuno-Gene Therapy in Mie University. Multiple coauthors reported similar relationships.
Source: Wantanabe T et al. Lancet Haematol. 2018 Nov;5(11):e520-31.
Combo appears safe, active in rel/ref NHL
The combination of Hu5F9-G4 (5F9) and rituximab was considered safe and produced durable complete responses (CRs) in patients with relapsed or refractory non-Hodgkin lymphoma (NHL) in a phase 1b trial.
Mainly low-grade adverse events (AEs) were observed with rituximab and 5F9, a macrophage-activating immune checkpoint inhibitor blocking CD47.
In addition, the combination produced an objective response rate (ORR) of 50% and a CR rate of 36%.
Most of the responses were ongoing at the time of data cutoff.
“It was very gratifying to see how the treatment was well-tolerated and showed a clinically meaningful response,” said Ranjana Advani, MD, of Stanford University in California.
She and her colleagues reported these results in The New England Journal of Medicine.
The study included 22 patients with relapsed or refractory NHL. Fifteen had diffuse large B-cell lymphoma (DLBCL), and seven had follicular lymphoma (FL).
The patients had received a median of four prior therapies (range, 2-10). Twenty-one patients had disease that was refractory to rituximab (all FL and 14 DLBCL patients).
All patients received 5F9 starting with a priming dose of 1 mg/kg followed by weekly maintenance doses of 10 to 30 mg/kg in three dose-escalation cohorts. The treatment was given until disease progression or lack of clinical benefit.
Patients received rituximab at 375 mg/m2 weekly starting on the second week of the first cycle and then monthly for cycles two through six.
Results
The most common treatment-related AEs were chills (41%), headache (41%), anemia (41%), and infusion-related reactions (36%).
Serious AEs included infections (18%), anemia (4.5%), dyspnea (4.5%), pyrexia (4.5%), lactic acidosis (4.5%), retroperitoneal mass (4.5%), pulmonary embolism (4.5%), and infusion-related reaction (4.5%).
For the entire cohort, the ORR was 50% (n=11), and the CR rate was 36% (n=8).
Among DLBCL patients, the ORR was 40% (n=6), and the CR rate was 33% (n=5). In FL patients, the ORR was 71% (n=5), and the CR rate was 43% (n=3).
The median duration of response was not reached in either disease cohort. The median follow-up was 6.2 months for DLBCL and 8.1 months for FL.
Ten of 11 responders (91%) were still in response at the time of data cutoff.
The researchers said a phase 2 trial of 5F9 plus rituximab in relapsed or refractory B-cell NHL is ongoing.
The phase 1b study was supported by Forty Seven, Inc., and the Leukemia and Lymphoma Society. Dr. Advani reported disclosures related to Forty Seven, Inc., Bristol-Myers Squibb, Pharmacyclics, Seattle Genetics, and Roche/Genentech, among others.
The combination of Hu5F9-G4 (5F9) and rituximab was considered safe and produced durable complete responses (CRs) in patients with relapsed or refractory non-Hodgkin lymphoma (NHL) in a phase 1b trial.
Mainly low-grade adverse events (AEs) were observed with rituximab and 5F9, a macrophage-activating immune checkpoint inhibitor blocking CD47.
In addition, the combination produced an objective response rate (ORR) of 50% and a CR rate of 36%.
Most of the responses were ongoing at the time of data cutoff.
“It was very gratifying to see how the treatment was well-tolerated and showed a clinically meaningful response,” said Ranjana Advani, MD, of Stanford University in California.
She and her colleagues reported these results in The New England Journal of Medicine.
The study included 22 patients with relapsed or refractory NHL. Fifteen had diffuse large B-cell lymphoma (DLBCL), and seven had follicular lymphoma (FL).
The patients had received a median of four prior therapies (range, 2-10). Twenty-one patients had disease that was refractory to rituximab (all FL and 14 DLBCL patients).
All patients received 5F9 starting with a priming dose of 1 mg/kg followed by weekly maintenance doses of 10 to 30 mg/kg in three dose-escalation cohorts. The treatment was given until disease progression or lack of clinical benefit.
Patients received rituximab at 375 mg/m2 weekly starting on the second week of the first cycle and then monthly for cycles two through six.
Results
The most common treatment-related AEs were chills (41%), headache (41%), anemia (41%), and infusion-related reactions (36%).
Serious AEs included infections (18%), anemia (4.5%), dyspnea (4.5%), pyrexia (4.5%), lactic acidosis (4.5%), retroperitoneal mass (4.5%), pulmonary embolism (4.5%), and infusion-related reaction (4.5%).
For the entire cohort, the ORR was 50% (n=11), and the CR rate was 36% (n=8).
Among DLBCL patients, the ORR was 40% (n=6), and the CR rate was 33% (n=5). In FL patients, the ORR was 71% (n=5), and the CR rate was 43% (n=3).
The median duration of response was not reached in either disease cohort. The median follow-up was 6.2 months for DLBCL and 8.1 months for FL.
Ten of 11 responders (91%) were still in response at the time of data cutoff.
The researchers said a phase 2 trial of 5F9 plus rituximab in relapsed or refractory B-cell NHL is ongoing.
The phase 1b study was supported by Forty Seven, Inc., and the Leukemia and Lymphoma Society. Dr. Advani reported disclosures related to Forty Seven, Inc., Bristol-Myers Squibb, Pharmacyclics, Seattle Genetics, and Roche/Genentech, among others.
The combination of Hu5F9-G4 (5F9) and rituximab was considered safe and produced durable complete responses (CRs) in patients with relapsed or refractory non-Hodgkin lymphoma (NHL) in a phase 1b trial.
Mainly low-grade adverse events (AEs) were observed with rituximab and 5F9, a macrophage-activating immune checkpoint inhibitor blocking CD47.
In addition, the combination produced an objective response rate (ORR) of 50% and a CR rate of 36%.
Most of the responses were ongoing at the time of data cutoff.
“It was very gratifying to see how the treatment was well-tolerated and showed a clinically meaningful response,” said Ranjana Advani, MD, of Stanford University in California.
She and her colleagues reported these results in The New England Journal of Medicine.
The study included 22 patients with relapsed or refractory NHL. Fifteen had diffuse large B-cell lymphoma (DLBCL), and seven had follicular lymphoma (FL).
The patients had received a median of four prior therapies (range, 2-10). Twenty-one patients had disease that was refractory to rituximab (all FL and 14 DLBCL patients).
All patients received 5F9 starting with a priming dose of 1 mg/kg followed by weekly maintenance doses of 10 to 30 mg/kg in three dose-escalation cohorts. The treatment was given until disease progression or lack of clinical benefit.
Patients received rituximab at 375 mg/m2 weekly starting on the second week of the first cycle and then monthly for cycles two through six.
Results
The most common treatment-related AEs were chills (41%), headache (41%), anemia (41%), and infusion-related reactions (36%).
Serious AEs included infections (18%), anemia (4.5%), dyspnea (4.5%), pyrexia (4.5%), lactic acidosis (4.5%), retroperitoneal mass (4.5%), pulmonary embolism (4.5%), and infusion-related reaction (4.5%).
For the entire cohort, the ORR was 50% (n=11), and the CR rate was 36% (n=8).
Among DLBCL patients, the ORR was 40% (n=6), and the CR rate was 33% (n=5). In FL patients, the ORR was 71% (n=5), and the CR rate was 43% (n=3).
The median duration of response was not reached in either disease cohort. The median follow-up was 6.2 months for DLBCL and 8.1 months for FL.
Ten of 11 responders (91%) were still in response at the time of data cutoff.
The researchers said a phase 2 trial of 5F9 plus rituximab in relapsed or refractory B-cell NHL is ongoing.
The phase 1b study was supported by Forty Seven, Inc., and the Leukemia and Lymphoma Society. Dr. Advani reported disclosures related to Forty Seven, Inc., Bristol-Myers Squibb, Pharmacyclics, Seattle Genetics, and Roche/Genentech, among others.
Sandoz won’t seek U.S. approval for rituximab biosimilar
Sandoz has decided not to pursue U.S. approval for its rituximab product (GP2013), a proposed biosimiliar of Rituxan/Mabthera.
GP2013 (Rixathon, Riximyo) is already approved outside the U.S.
Sandoz was seeking U.S. approval of GP2013 for all the same indications as the reference product—B-cell non-Hodgkin lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, microscopic polyangiitis, and pemphigus vulgaris.
The U.S. Food and Drug Administration (FDA) had accepted the biologics license application (BLA) for GP2013 in September 2017.
In May of this year, the FDA issued a complete response letter saying it could not approve GP2013. The agency also requested additional information to complement the BLA submission for GP2013.
At the time of the complete response letter, Sandoz said it was still committed to bringing GP2013 to the U.S. market. Now, the company’s position has changed.
“We appreciate the important conversations with the FDA, which have provided specific requirements for our potential U.S. biosimilar rituximab, but believe the patient and marketplace needs in the U.S. will be satisfied before we can generate the data required,” said Stefan Hendriks, global head of biopharmaceuticals at Sandoz.
“We are disappointed to have to make this decision and stand behind the safety, efficacy, and quality of our medicine, which met the stringent criteria for approval in the European Union, Switzerland, Japan, New Zealand, and Australia.”
The BLA for GP2013 was supported, in part, by the ASSIST-FL trial (NCT01419665), in which researchers compared GP2013 to the reference product. Results from this trial were published in The Lancet Haematology in July 2017.
The phase 3 trial included adults with previously untreated, advanced stage follicular lymphoma. Patients received 8 cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% (271/311) in the GP2013 arm and 88% in the rituximab arm (274/313). Complete response rates were 15% (n=46) and 13% (n=42), respectively.
Rates of adverse events (AEs) were 93% in the GP2013 arm and 91% in the rituximab arm. Rates of serious AEs were 23% and 20%, respectively. The rate of discontinuation due to AEs was 7% in both arms.
The most common AE was neutropenia, which occurred in 26% of patients in the GP2013 arm and 30% of those in the rituximab arm in the combination phase. Rates of neutropenia in the maintenance phase were 10% and 6%, respectively.
Sandoz has decided not to pursue U.S. approval for its rituximab product (GP2013), a proposed biosimiliar of Rituxan/Mabthera.
GP2013 (Rixathon, Riximyo) is already approved outside the U.S.
Sandoz was seeking U.S. approval of GP2013 for all the same indications as the reference product—B-cell non-Hodgkin lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, microscopic polyangiitis, and pemphigus vulgaris.
The U.S. Food and Drug Administration (FDA) had accepted the biologics license application (BLA) for GP2013 in September 2017.
In May of this year, the FDA issued a complete response letter saying it could not approve GP2013. The agency also requested additional information to complement the BLA submission for GP2013.
At the time of the complete response letter, Sandoz said it was still committed to bringing GP2013 to the U.S. market. Now, the company’s position has changed.
“We appreciate the important conversations with the FDA, which have provided specific requirements for our potential U.S. biosimilar rituximab, but believe the patient and marketplace needs in the U.S. will be satisfied before we can generate the data required,” said Stefan Hendriks, global head of biopharmaceuticals at Sandoz.
“We are disappointed to have to make this decision and stand behind the safety, efficacy, and quality of our medicine, which met the stringent criteria for approval in the European Union, Switzerland, Japan, New Zealand, and Australia.”
The BLA for GP2013 was supported, in part, by the ASSIST-FL trial (NCT01419665), in which researchers compared GP2013 to the reference product. Results from this trial were published in The Lancet Haematology in July 2017.
The phase 3 trial included adults with previously untreated, advanced stage follicular lymphoma. Patients received 8 cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% (271/311) in the GP2013 arm and 88% in the rituximab arm (274/313). Complete response rates were 15% (n=46) and 13% (n=42), respectively.
Rates of adverse events (AEs) were 93% in the GP2013 arm and 91% in the rituximab arm. Rates of serious AEs were 23% and 20%, respectively. The rate of discontinuation due to AEs was 7% in both arms.
The most common AE was neutropenia, which occurred in 26% of patients in the GP2013 arm and 30% of those in the rituximab arm in the combination phase. Rates of neutropenia in the maintenance phase were 10% and 6%, respectively.
Sandoz has decided not to pursue U.S. approval for its rituximab product (GP2013), a proposed biosimiliar of Rituxan/Mabthera.
GP2013 (Rixathon, Riximyo) is already approved outside the U.S.
Sandoz was seeking U.S. approval of GP2013 for all the same indications as the reference product—B-cell non-Hodgkin lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, microscopic polyangiitis, and pemphigus vulgaris.
The U.S. Food and Drug Administration (FDA) had accepted the biologics license application (BLA) for GP2013 in September 2017.
In May of this year, the FDA issued a complete response letter saying it could not approve GP2013. The agency also requested additional information to complement the BLA submission for GP2013.
At the time of the complete response letter, Sandoz said it was still committed to bringing GP2013 to the U.S. market. Now, the company’s position has changed.
“We appreciate the important conversations with the FDA, which have provided specific requirements for our potential U.S. biosimilar rituximab, but believe the patient and marketplace needs in the U.S. will be satisfied before we can generate the data required,” said Stefan Hendriks, global head of biopharmaceuticals at Sandoz.
“We are disappointed to have to make this decision and stand behind the safety, efficacy, and quality of our medicine, which met the stringent criteria for approval in the European Union, Switzerland, Japan, New Zealand, and Australia.”
The BLA for GP2013 was supported, in part, by the ASSIST-FL trial (NCT01419665), in which researchers compared GP2013 to the reference product. Results from this trial were published in The Lancet Haematology in July 2017.
The phase 3 trial included adults with previously untreated, advanced stage follicular lymphoma. Patients received 8 cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% (271/311) in the GP2013 arm and 88% in the rituximab arm (274/313). Complete response rates were 15% (n=46) and 13% (n=42), respectively.
Rates of adverse events (AEs) were 93% in the GP2013 arm and 91% in the rituximab arm. Rates of serious AEs were 23% and 20%, respectively. The rate of discontinuation due to AEs was 7% in both arms.
The most common AE was neutropenia, which occurred in 26% of patients in the GP2013 arm and 30% of those in the rituximab arm in the combination phase. Rates of neutropenia in the maintenance phase were 10% and 6%, respectively.
Sandoz halts pursuit of U.S. approval for rituximab biosimilar
.
Sandoz, a division of Novartis, was seeking Food and Drug Administration approval of GP2013 for all the same indications as the reference product – B-cell non-Hodgkin lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, microscopic polyangiitis, and pemphigus vulgaris.
GP2013 already is approved in the European Union and elsewhere.
The FDA had accepted the biologics license application (BLA) for GP2013 in September 2017. In May 2018, the agency issued a complete response letter saying it could not approve GP2013. The agency also requested additional information to complement the BLA submission.
At the time of the complete response letter, Sandoz said it was still committed to bringing GP2013 to the U.S. market.
“We appreciate the important conversations with the FDA, which have provided specific requirements for our potential U.S. biosimilar rituximab but believe the patient and marketplace needs in the U.S. will be satisfied before we can generate the data required,” Stefan Hendriks, global head of biopharmaceuticals at Sandoz, said in a statement.
“We are disappointed to have to make this decision and stand behind the safety, efficacy, and quality of our medicine, which met the stringent criteria for approval in the European Union, Switzerland, Japan, New Zealand, and Australia.”
The BLA for GP2013 was supported, in part, by results from the ASSIST-FL trial, in which researchers compared GP2013 with the reference product (Lancet Haematol. 2017 Aug;4[8]:e350-61).
The phase 3 trial included adults with previously untreated, advanced-stage follicular lymphoma. Patients received eight cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% in the GP2013 arm and 88% in the rituximab arm.
.
Sandoz, a division of Novartis, was seeking Food and Drug Administration approval of GP2013 for all the same indications as the reference product – B-cell non-Hodgkin lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, microscopic polyangiitis, and pemphigus vulgaris.
GP2013 already is approved in the European Union and elsewhere.
The FDA had accepted the biologics license application (BLA) for GP2013 in September 2017. In May 2018, the agency issued a complete response letter saying it could not approve GP2013. The agency also requested additional information to complement the BLA submission.
At the time of the complete response letter, Sandoz said it was still committed to bringing GP2013 to the U.S. market.
“We appreciate the important conversations with the FDA, which have provided specific requirements for our potential U.S. biosimilar rituximab but believe the patient and marketplace needs in the U.S. will be satisfied before we can generate the data required,” Stefan Hendriks, global head of biopharmaceuticals at Sandoz, said in a statement.
“We are disappointed to have to make this decision and stand behind the safety, efficacy, and quality of our medicine, which met the stringent criteria for approval in the European Union, Switzerland, Japan, New Zealand, and Australia.”
The BLA for GP2013 was supported, in part, by results from the ASSIST-FL trial, in which researchers compared GP2013 with the reference product (Lancet Haematol. 2017 Aug;4[8]:e350-61).
The phase 3 trial included adults with previously untreated, advanced-stage follicular lymphoma. Patients received eight cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% in the GP2013 arm and 88% in the rituximab arm.
.
Sandoz, a division of Novartis, was seeking Food and Drug Administration approval of GP2013 for all the same indications as the reference product – B-cell non-Hodgkin lymphoma, chronic lymphocytic leukemia, rheumatoid arthritis, granulomatosis with polyangiitis, microscopic polyangiitis, and pemphigus vulgaris.
GP2013 already is approved in the European Union and elsewhere.
The FDA had accepted the biologics license application (BLA) for GP2013 in September 2017. In May 2018, the agency issued a complete response letter saying it could not approve GP2013. The agency also requested additional information to complement the BLA submission.
At the time of the complete response letter, Sandoz said it was still committed to bringing GP2013 to the U.S. market.
“We appreciate the important conversations with the FDA, which have provided specific requirements for our potential U.S. biosimilar rituximab but believe the patient and marketplace needs in the U.S. will be satisfied before we can generate the data required,” Stefan Hendriks, global head of biopharmaceuticals at Sandoz, said in a statement.
“We are disappointed to have to make this decision and stand behind the safety, efficacy, and quality of our medicine, which met the stringent criteria for approval in the European Union, Switzerland, Japan, New Zealand, and Australia.”
The BLA for GP2013 was supported, in part, by results from the ASSIST-FL trial, in which researchers compared GP2013 with the reference product (Lancet Haematol. 2017 Aug;4[8]:e350-61).
The phase 3 trial included adults with previously untreated, advanced-stage follicular lymphoma. Patients received eight cycles of cyclophosphamide, vincristine, and prednisone with either GP2013 or reference rituximab. Responders then received GP2013 or rituximab monotherapy as maintenance for up to 2 years.
At a median follow-up of 11.6 months, the overall response rate was 87% in the GP2013 arm and 88% in the rituximab arm.