User login
Transvaginal mesh, native tissue repair have similar outcomes in 3-year trial
Transvaginal mesh was found to be safe and effective for patients with pelvic organ prolapse (POP) when compared with native tissue repair (NTR) in a 3-year trial.
Researchers, led by Bruce S. Kahn, MD, with the department of obstetrics & gynecology at Scripps Clinic in San Diego evaluated the two surgical treatment methods and published their findings in Obstetrics & Gynecology.
At completion of the 3-year follow-up in 2016, there were 401 participants in the transvaginal mesh group and 171 in the NTR group.
The prospective, nonrandomized, parallel-cohort, 27-site trial used a primary composite endpoint of anatomical success; subjective success (vaginal bulging); retreatment measures; and serious device-related or serious procedure-related adverse events.
The secondary endpoint was a composite outcome similar to the primary composite outcome but with anatomical success more stringently defined as POP quantification (POP-Q) point Ba < 0 and/or C < 0.
The secondary outcome was added to this trial because investigators had criticized the primary endpoint, set by the Food and Drug Administration, because it included anatomic outcome measures that were the same for inclusion criteria (POP-Q point Ba < 0 and/or C < 0.)
The secondary-outcome composite also included quality-of-life measures, mesh exposure, and mesh- and procedure-related complications.
Outcomes similar for both groups
The primary outcome demonstrated transvaginal mesh was not superior to native tissue repair (P =.056).
In the secondary outcome, superiority of transvaginal mesh over native tissue repair was shown (P =.009), with a propensity score–adjusted difference of 10.6% (90% confidence interval, 3.3%-17.9%) in favor of transvaginal mesh.
The authors noted that subjective success regarding vaginal bulging, which is important in patient satisfaction, was high and not statistically different between the two groups.
Additionally, transvaginal mesh repair was as safe as NTR regarding serious device-related and/or serious procedure-related side effects.
For the primary safety endpoint, 3.1% in the mesh group and 2.7% in the native tissue repair group experienced serious adverse events, demonstrating that mesh was noninferior to NTR.
Research results have been mixed
Unanswered questions surround surgical options for POP, which, the authors wrote, “affects 3%-6% of women based on symptoms and up to 50% of women based on vaginal examination.”
The FDA in 2011 issued 522 postmarket surveillance study orders for companies that market transvaginal mesh for POP.
Research results have varied and contentious debate has continued in the field. Some studies have shown that mesh has better subjective and objective outcomes than NTR in the anterior compartment. Others have found more complications with transvaginal mesh, such as mesh exposure and painful intercourse.
Complicating comparisons, early versions of the mesh used were larger and denser than today’s versions.
In this postmarket study, patients received either the Uphold LITE brand of transvaginal mesh or native tissue repair for surgical treatment of POP.
Expert: This study unlikely to change minds
In an accompanying editorial, John O.L. DeLancey, MD, professor of gynecology at the University of Michigan, Ann Arbor, pointed out that so far there’s been a lack of randomized trials that could answer whether mesh surgeries result in fewer symptoms or result in sufficient improvements in anatomy to justify their additional risk.
This study may not help with the decision. Dr. DeLancey wrote: “Will this study change the minds of either side of this debate? Probably not. The two sides are deeply entrenched in their positions.”
Two considerations are important in thinking about the issue, he said. Surgical outcomes for POP are “not as good as we would hope.” Also, many women have had serious complications with mesh operations.
He wrote: “Mesh litigation has resulted in more $8 billion in settlements, which is many times the $1 billion annual national cost of providing care for prolapse. Those of us who practice in referral centers have seen women with devastating problems, even though they probably represent a small fraction of cases.”
Dr. DeLancey highlighted some limitations of the study by Dr. Kahn and colleagues, especially regarding differences in the groups studied and the design of the study.
“For example,” he explained, “65% of individuals in the mesh-repair group had a prior hysterectomy as opposed to 30% in the native tissue repair group. In addition, some of the operations in the native tissue group are not typical choices; for example, hysteropexy was used for some patients and had a 47% failure rate.”
He said the all-or-nothing approach to surgical solutions may be clouding the debate – in other words mesh or no mesh for women as a group.
“Rather than asking whether mesh is better than no mesh, knowing which women (if any) stand to benefit from mesh is the critical question. We need to understand, for each woman, what structural failures exist so that we can target our interventions to correct them,” he wrote.
This study was sponsored by Boston Scientific. Dr. Kahn disclosed research support from Solaire, payments from AbbVie and Douchenay as a speaker, payments from Caldera and Cytuity (Boston Scientific) as a medical consultant, and payment from Johnson & Johnson as an expert witness. One coauthor disclosed that money was paid to her institution from Medtronic and Boston Scientific (both unrestricted educational grants for cadaveric lab). Another is chief medical officer at Axonics. One study coauthor receives research funding from Axonics and is a consultant for Group Dynamics, Medpace, and FirstThought. One coauthor received research support, is a consultant for Boston Scientific, and is an expert witness for Johnson & Johnson. Dr. DeLancey declared no relevant financial relationships.
Transvaginal mesh was found to be safe and effective for patients with pelvic organ prolapse (POP) when compared with native tissue repair (NTR) in a 3-year trial.
Researchers, led by Bruce S. Kahn, MD, with the department of obstetrics & gynecology at Scripps Clinic in San Diego evaluated the two surgical treatment methods and published their findings in Obstetrics & Gynecology.
At completion of the 3-year follow-up in 2016, there were 401 participants in the transvaginal mesh group and 171 in the NTR group.
The prospective, nonrandomized, parallel-cohort, 27-site trial used a primary composite endpoint of anatomical success; subjective success (vaginal bulging); retreatment measures; and serious device-related or serious procedure-related adverse events.
The secondary endpoint was a composite outcome similar to the primary composite outcome but with anatomical success more stringently defined as POP quantification (POP-Q) point Ba < 0 and/or C < 0.
The secondary outcome was added to this trial because investigators had criticized the primary endpoint, set by the Food and Drug Administration, because it included anatomic outcome measures that were the same for inclusion criteria (POP-Q point Ba < 0 and/or C < 0.)
The secondary-outcome composite also included quality-of-life measures, mesh exposure, and mesh- and procedure-related complications.
Outcomes similar for both groups
The primary outcome demonstrated transvaginal mesh was not superior to native tissue repair (P =.056).
In the secondary outcome, superiority of transvaginal mesh over native tissue repair was shown (P =.009), with a propensity score–adjusted difference of 10.6% (90% confidence interval, 3.3%-17.9%) in favor of transvaginal mesh.
The authors noted that subjective success regarding vaginal bulging, which is important in patient satisfaction, was high and not statistically different between the two groups.
Additionally, transvaginal mesh repair was as safe as NTR regarding serious device-related and/or serious procedure-related side effects.
For the primary safety endpoint, 3.1% in the mesh group and 2.7% in the native tissue repair group experienced serious adverse events, demonstrating that mesh was noninferior to NTR.
Research results have been mixed
Unanswered questions surround surgical options for POP, which, the authors wrote, “affects 3%-6% of women based on symptoms and up to 50% of women based on vaginal examination.”
The FDA in 2011 issued 522 postmarket surveillance study orders for companies that market transvaginal mesh for POP.
Research results have varied and contentious debate has continued in the field. Some studies have shown that mesh has better subjective and objective outcomes than NTR in the anterior compartment. Others have found more complications with transvaginal mesh, such as mesh exposure and painful intercourse.
Complicating comparisons, early versions of the mesh used were larger and denser than today’s versions.
In this postmarket study, patients received either the Uphold LITE brand of transvaginal mesh or native tissue repair for surgical treatment of POP.
Expert: This study unlikely to change minds
In an accompanying editorial, John O.L. DeLancey, MD, professor of gynecology at the University of Michigan, Ann Arbor, pointed out that so far there’s been a lack of randomized trials that could answer whether mesh surgeries result in fewer symptoms or result in sufficient improvements in anatomy to justify their additional risk.
This study may not help with the decision. Dr. DeLancey wrote: “Will this study change the minds of either side of this debate? Probably not. The two sides are deeply entrenched in their positions.”
Two considerations are important in thinking about the issue, he said. Surgical outcomes for POP are “not as good as we would hope.” Also, many women have had serious complications with mesh operations.
He wrote: “Mesh litigation has resulted in more $8 billion in settlements, which is many times the $1 billion annual national cost of providing care for prolapse. Those of us who practice in referral centers have seen women with devastating problems, even though they probably represent a small fraction of cases.”
Dr. DeLancey highlighted some limitations of the study by Dr. Kahn and colleagues, especially regarding differences in the groups studied and the design of the study.
“For example,” he explained, “65% of individuals in the mesh-repair group had a prior hysterectomy as opposed to 30% in the native tissue repair group. In addition, some of the operations in the native tissue group are not typical choices; for example, hysteropexy was used for some patients and had a 47% failure rate.”
He said the all-or-nothing approach to surgical solutions may be clouding the debate – in other words mesh or no mesh for women as a group.
“Rather than asking whether mesh is better than no mesh, knowing which women (if any) stand to benefit from mesh is the critical question. We need to understand, for each woman, what structural failures exist so that we can target our interventions to correct them,” he wrote.
This study was sponsored by Boston Scientific. Dr. Kahn disclosed research support from Solaire, payments from AbbVie and Douchenay as a speaker, payments from Caldera and Cytuity (Boston Scientific) as a medical consultant, and payment from Johnson & Johnson as an expert witness. One coauthor disclosed that money was paid to her institution from Medtronic and Boston Scientific (both unrestricted educational grants for cadaveric lab). Another is chief medical officer at Axonics. One study coauthor receives research funding from Axonics and is a consultant for Group Dynamics, Medpace, and FirstThought. One coauthor received research support, is a consultant for Boston Scientific, and is an expert witness for Johnson & Johnson. Dr. DeLancey declared no relevant financial relationships.
Transvaginal mesh was found to be safe and effective for patients with pelvic organ prolapse (POP) when compared with native tissue repair (NTR) in a 3-year trial.
Researchers, led by Bruce S. Kahn, MD, with the department of obstetrics & gynecology at Scripps Clinic in San Diego evaluated the two surgical treatment methods and published their findings in Obstetrics & Gynecology.
At completion of the 3-year follow-up in 2016, there were 401 participants in the transvaginal mesh group and 171 in the NTR group.
The prospective, nonrandomized, parallel-cohort, 27-site trial used a primary composite endpoint of anatomical success; subjective success (vaginal bulging); retreatment measures; and serious device-related or serious procedure-related adverse events.
The secondary endpoint was a composite outcome similar to the primary composite outcome but with anatomical success more stringently defined as POP quantification (POP-Q) point Ba < 0 and/or C < 0.
The secondary outcome was added to this trial because investigators had criticized the primary endpoint, set by the Food and Drug Administration, because it included anatomic outcome measures that were the same for inclusion criteria (POP-Q point Ba < 0 and/or C < 0.)
The secondary-outcome composite also included quality-of-life measures, mesh exposure, and mesh- and procedure-related complications.
Outcomes similar for both groups
The primary outcome demonstrated transvaginal mesh was not superior to native tissue repair (P =.056).
In the secondary outcome, superiority of transvaginal mesh over native tissue repair was shown (P =.009), with a propensity score–adjusted difference of 10.6% (90% confidence interval, 3.3%-17.9%) in favor of transvaginal mesh.
The authors noted that subjective success regarding vaginal bulging, which is important in patient satisfaction, was high and not statistically different between the two groups.
Additionally, transvaginal mesh repair was as safe as NTR regarding serious device-related and/or serious procedure-related side effects.
For the primary safety endpoint, 3.1% in the mesh group and 2.7% in the native tissue repair group experienced serious adverse events, demonstrating that mesh was noninferior to NTR.
Research results have been mixed
Unanswered questions surround surgical options for POP, which, the authors wrote, “affects 3%-6% of women based on symptoms and up to 50% of women based on vaginal examination.”
The FDA in 2011 issued 522 postmarket surveillance study orders for companies that market transvaginal mesh for POP.
Research results have varied and contentious debate has continued in the field. Some studies have shown that mesh has better subjective and objective outcomes than NTR in the anterior compartment. Others have found more complications with transvaginal mesh, such as mesh exposure and painful intercourse.
Complicating comparisons, early versions of the mesh used were larger and denser than today’s versions.
In this postmarket study, patients received either the Uphold LITE brand of transvaginal mesh or native tissue repair for surgical treatment of POP.
Expert: This study unlikely to change minds
In an accompanying editorial, John O.L. DeLancey, MD, professor of gynecology at the University of Michigan, Ann Arbor, pointed out that so far there’s been a lack of randomized trials that could answer whether mesh surgeries result in fewer symptoms or result in sufficient improvements in anatomy to justify their additional risk.
This study may not help with the decision. Dr. DeLancey wrote: “Will this study change the minds of either side of this debate? Probably not. The two sides are deeply entrenched in their positions.”
Two considerations are important in thinking about the issue, he said. Surgical outcomes for POP are “not as good as we would hope.” Also, many women have had serious complications with mesh operations.
He wrote: “Mesh litigation has resulted in more $8 billion in settlements, which is many times the $1 billion annual national cost of providing care for prolapse. Those of us who practice in referral centers have seen women with devastating problems, even though they probably represent a small fraction of cases.”
Dr. DeLancey highlighted some limitations of the study by Dr. Kahn and colleagues, especially regarding differences in the groups studied and the design of the study.
“For example,” he explained, “65% of individuals in the mesh-repair group had a prior hysterectomy as opposed to 30% in the native tissue repair group. In addition, some of the operations in the native tissue group are not typical choices; for example, hysteropexy was used for some patients and had a 47% failure rate.”
He said the all-or-nothing approach to surgical solutions may be clouding the debate – in other words mesh or no mesh for women as a group.
“Rather than asking whether mesh is better than no mesh, knowing which women (if any) stand to benefit from mesh is the critical question. We need to understand, for each woman, what structural failures exist so that we can target our interventions to correct them,” he wrote.
This study was sponsored by Boston Scientific. Dr. Kahn disclosed research support from Solaire, payments from AbbVie and Douchenay as a speaker, payments from Caldera and Cytuity (Boston Scientific) as a medical consultant, and payment from Johnson & Johnson as an expert witness. One coauthor disclosed that money was paid to her institution from Medtronic and Boston Scientific (both unrestricted educational grants for cadaveric lab). Another is chief medical officer at Axonics. One study coauthor receives research funding from Axonics and is a consultant for Group Dynamics, Medpace, and FirstThought. One coauthor received research support, is a consultant for Boston Scientific, and is an expert witness for Johnson & Johnson. Dr. DeLancey declared no relevant financial relationships.
FROM OBSTETRICS & GYNECOLOGY
FDA clears diagnostic test for early Alzheimer’s
The Lumipulse G β-Amyloid Ratio 1-42/1-40 (Fujirebio Diagnostics) test detects amyloid plaques associated with AD in adults age 55 or older who are under investigation for AD and other causes of cognitive decline.
“The availability of an in vitro diagnostic test that can potentially eliminate the need for time-consuming and expensive [positron emission tomography (PET)] scans is great news for individuals and families concerned with the possibility of an Alzheimer’s disease diagnosis,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, said in a statement.
“With the Lumipulse test, there is a new option that can typically be completed the same day and can give doctors the same information regarding brain amyloid status, without the radiation risk, to help determine if a patient’s cognitive impairment is due to Alzheimer’s disease,” he added.
In its statement, the FDA notes that there is an “unmet need for a reliable and safe test that can accurately identify patients with amyloid plaques consistent with Alzheimer’s disease.”
The agency goes on to state that this new test may eliminate the need to use PET brain scans, a “potentially costly and cumbersome option” to visualize amyloid plaques for the diagnosis of AD.
The Lumipulse test measures the ratio of β-amyloid 1-42 and β-amyloid 1-40 concentrations in human cerebral spinal fluid (CSF). A positive Lumipulse G β-amyloid Ratio (1-42/1-40) test result is consistent with the presence of amyloid plaques, similar to that revealed in a PET scan. A negative result is consistent with a negative amyloid PET scan result.
However, the FDA notes that the test is not a stand-alone assay and should be used in conjunction with other clinical evaluations and additional tests to determine treatment options.
The FDA reports that it evaluated the safety and efficacy of the test in a clinical study of 292 CSF samples from the Alzheimer’s Disease Neuroimaging Initiative sample bank.
The samples were tested by the Lumipulse G β-amyloid Ratio (1-42/1-40) and compared with amyloid PET scan results. In this clinical study, 97% of individuals with Lumipulse G β-amyloid Ratio (1-42/1-40) positive results had the presence of amyloid plaques by PET scan and 84% of individuals with negative results had a negative amyloid PET scan.
The risks associated with the Lumipulse G β-amyloid Ratio (1-42/1-40) test are mainly the possibility of false-positive and false-negative test results.
False-positive results, in conjunction with other clinical information, could lead to an inappropriate diagnosis of, and unnecessary treatment for AD.
False-negative test results could result in additional unnecessary diagnostic tests and potential delay in effective treatment for AD.
The FDA reviewed the device through the De Novo premarket review pathway, a regulatory pathway for low- to moderate-risk devices of a new type.
The agency says this action “creates a new regulatory classification, which means that subsequent devices of the same type with the same intended use may go through FDA’s 510(k) premarket process, whereby devices can obtain marketing authorization by demonstrating substantial equivalence to a predicate device.”
The Lumipulse G β-amyloid Ratio (1-42/1-40) was granted Breakthrough Device designation, a process designed to expedite the development and review of devices that may provide for more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions.
A version of this article first appeared on Medscape.com.
The Lumipulse G β-Amyloid Ratio 1-42/1-40 (Fujirebio Diagnostics) test detects amyloid plaques associated with AD in adults age 55 or older who are under investigation for AD and other causes of cognitive decline.
“The availability of an in vitro diagnostic test that can potentially eliminate the need for time-consuming and expensive [positron emission tomography (PET)] scans is great news for individuals and families concerned with the possibility of an Alzheimer’s disease diagnosis,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, said in a statement.
“With the Lumipulse test, there is a new option that can typically be completed the same day and can give doctors the same information regarding brain amyloid status, without the radiation risk, to help determine if a patient’s cognitive impairment is due to Alzheimer’s disease,” he added.
In its statement, the FDA notes that there is an “unmet need for a reliable and safe test that can accurately identify patients with amyloid plaques consistent with Alzheimer’s disease.”
The agency goes on to state that this new test may eliminate the need to use PET brain scans, a “potentially costly and cumbersome option” to visualize amyloid plaques for the diagnosis of AD.
The Lumipulse test measures the ratio of β-amyloid 1-42 and β-amyloid 1-40 concentrations in human cerebral spinal fluid (CSF). A positive Lumipulse G β-amyloid Ratio (1-42/1-40) test result is consistent with the presence of amyloid plaques, similar to that revealed in a PET scan. A negative result is consistent with a negative amyloid PET scan result.
However, the FDA notes that the test is not a stand-alone assay and should be used in conjunction with other clinical evaluations and additional tests to determine treatment options.
The FDA reports that it evaluated the safety and efficacy of the test in a clinical study of 292 CSF samples from the Alzheimer’s Disease Neuroimaging Initiative sample bank.
The samples were tested by the Lumipulse G β-amyloid Ratio (1-42/1-40) and compared with amyloid PET scan results. In this clinical study, 97% of individuals with Lumipulse G β-amyloid Ratio (1-42/1-40) positive results had the presence of amyloid plaques by PET scan and 84% of individuals with negative results had a negative amyloid PET scan.
The risks associated with the Lumipulse G β-amyloid Ratio (1-42/1-40) test are mainly the possibility of false-positive and false-negative test results.
False-positive results, in conjunction with other clinical information, could lead to an inappropriate diagnosis of, and unnecessary treatment for AD.
False-negative test results could result in additional unnecessary diagnostic tests and potential delay in effective treatment for AD.
The FDA reviewed the device through the De Novo premarket review pathway, a regulatory pathway for low- to moderate-risk devices of a new type.
The agency says this action “creates a new regulatory classification, which means that subsequent devices of the same type with the same intended use may go through FDA’s 510(k) premarket process, whereby devices can obtain marketing authorization by demonstrating substantial equivalence to a predicate device.”
The Lumipulse G β-amyloid Ratio (1-42/1-40) was granted Breakthrough Device designation, a process designed to expedite the development and review of devices that may provide for more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions.
A version of this article first appeared on Medscape.com.
The Lumipulse G β-Amyloid Ratio 1-42/1-40 (Fujirebio Diagnostics) test detects amyloid plaques associated with AD in adults age 55 or older who are under investigation for AD and other causes of cognitive decline.
“The availability of an in vitro diagnostic test that can potentially eliminate the need for time-consuming and expensive [positron emission tomography (PET)] scans is great news for individuals and families concerned with the possibility of an Alzheimer’s disease diagnosis,” Jeff Shuren, MD, JD, director of the FDA’s Center for Devices and Radiological Health, said in a statement.
“With the Lumipulse test, there is a new option that can typically be completed the same day and can give doctors the same information regarding brain amyloid status, without the radiation risk, to help determine if a patient’s cognitive impairment is due to Alzheimer’s disease,” he added.
In its statement, the FDA notes that there is an “unmet need for a reliable and safe test that can accurately identify patients with amyloid plaques consistent with Alzheimer’s disease.”
The agency goes on to state that this new test may eliminate the need to use PET brain scans, a “potentially costly and cumbersome option” to visualize amyloid plaques for the diagnosis of AD.
The Lumipulse test measures the ratio of β-amyloid 1-42 and β-amyloid 1-40 concentrations in human cerebral spinal fluid (CSF). A positive Lumipulse G β-amyloid Ratio (1-42/1-40) test result is consistent with the presence of amyloid plaques, similar to that revealed in a PET scan. A negative result is consistent with a negative amyloid PET scan result.
However, the FDA notes that the test is not a stand-alone assay and should be used in conjunction with other clinical evaluations and additional tests to determine treatment options.
The FDA reports that it evaluated the safety and efficacy of the test in a clinical study of 292 CSF samples from the Alzheimer’s Disease Neuroimaging Initiative sample bank.
The samples were tested by the Lumipulse G β-amyloid Ratio (1-42/1-40) and compared with amyloid PET scan results. In this clinical study, 97% of individuals with Lumipulse G β-amyloid Ratio (1-42/1-40) positive results had the presence of amyloid plaques by PET scan and 84% of individuals with negative results had a negative amyloid PET scan.
The risks associated with the Lumipulse G β-amyloid Ratio (1-42/1-40) test are mainly the possibility of false-positive and false-negative test results.
False-positive results, in conjunction with other clinical information, could lead to an inappropriate diagnosis of, and unnecessary treatment for AD.
False-negative test results could result in additional unnecessary diagnostic tests and potential delay in effective treatment for AD.
The FDA reviewed the device through the De Novo premarket review pathway, a regulatory pathway for low- to moderate-risk devices of a new type.
The agency says this action “creates a new regulatory classification, which means that subsequent devices of the same type with the same intended use may go through FDA’s 510(k) premarket process, whereby devices can obtain marketing authorization by demonstrating substantial equivalence to a predicate device.”
The Lumipulse G β-amyloid Ratio (1-42/1-40) was granted Breakthrough Device designation, a process designed to expedite the development and review of devices that may provide for more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions.
A version of this article first appeared on Medscape.com.
Seven hours of sleep is ideal for middle aged and older
Sleep disturbances are common in older age, and previous studies have shown associations between too much or too little sleep and increased risk of cognitive decline, but the ideal amount of sleep for preserving mental health has not been well described, according to the authors of the new paper.
In the study published in Nature Aging, the team of researchers from China and the United Kingdom reviewed data from the UK Biobank, a national database of individuals in the United Kingdom that includes cognitive assessments, mental health questionnaires, and brain imaging data, as well as genetic information.
Sleep is important for physical and psychological health, and also serves a neuroprotective function by clearing waste products from the brain, lead author Yuzhu Li of Fudan University, Shanghai, China, and colleagues wrote.
The study population included 498,277 participants, aged 38-73 years, who completed touchscreen questionnaires about sleep duration between 2006 and 2010. The average age at baseline was 56.5 years, 54% were female, and the mean sleep duration was 7.15 hours.
The researchers also reviewed brain imaging data and genetic data from 39,692 participants in 2014 to examine the relationships between sleep duration and brain structure and between sleep duration and genetic risk. In addition, 156,884 participants completed an online follow-up mental health questionnaire in 2016-2017 to assess the longitudinal impact of sleep on mental health.
Both excessive and insufficient sleep was associated with impaired cognitive performance, evidenced by the U-shaped curve found by the researchers in their data analysis, which used quadratic associations.
Specific cognitive functions including pair matching, trail making, prospective memory, and reaction time were significantly impaired with too much or too little sleep, the researchers said. “This demonstrated the positive association of both insufficient and excessive sleep duration with inferior performance on cognitive tasks.”
When the researchers analyzed the association between sleep duration and mental health, sleep duration also showed a U-shaped association with symptoms of anxiety, depression, mental distress, mania, and self-harm, while well-being showed an inverted U-shape. All associations between sleep duration and mental health were statistically significant after controlling for confounding variables (P < .001).
On further analysis (using two-line tests), the researchers determined that consistent sleep duration of approximately 7 hours per night was optimal for cognitive performance and for good mental health.
The researchers also used neuroimaging data to examine the relationship between sleep duration and brain structure. Overall, greater changes were seen in the regions of the brain involved in cognitive processing and memory.
“The most significant cortical volumes nonlinearly associated with sleep duration included the precentral cortex, the superior frontal gyrus, the lateral orbitofrontal cortex, the pars orbitalis, the frontal pole, and the middle temporal cortex,” the researchers wrote (P < .05 for all).
The association between sleep duration and cognitive function diminished among individuals older than 65 years, compared with those aged approximately 40 years, which suggests that optimal sleep duration may be more beneficial in middle age, the researchers noted. However, no similar impact of age was seen for mental health. For brain structure, the nonlinear relationship between sleep duration and cortical volumes was greatest in those aged 44-59 years, and gradually flattened with older age.
Research supports sleep discussions with patients
“Primary care physicians can use this study in their discussions with middle-aged and older patients to recommend optimal sleep duration and measures to achieve this sleep target,” Noel Deep, MD, a general internist in group practice in Antigo, Wisc., who was not involved in the study, said in an interview.
“This study is important because it demonstrated that both inadequate and excessive sleep patterns were associated with cognitive and mental health changes,” said Dr. Deep. “It supported previous observations of cognitive decline and mental health disorders being linked to disturbed sleep. But this study was unique because it provides data supporting an optimal sleep duration of 7 hours and the ill effects of both insufficient and excessive sleep duration.
“The usual thought process has been to assume that older individuals may not require as much sleep as the younger individuals, but this study supports an optimal time duration of sleep of 7 hours that benefits the older individuals. It was also interesting to note the mental health effects caused by the inadequate and excessive sleep durations,” he added.
As for additional research, “I would like to look into the quality of the sleep, in addition to the duration of sleep,” said Dr. Deep. For example, whether the excessive sleep was caused by poor quality sleep or fragmented sleep leading to the structural and subsequent cognitive decline.
Study limitations
“The current study relied on self-reporting of the sleep duration and was not observed and recorded data,” Dr. Deep noted. “It would also be beneficial to not only rely on healthy volunteers reporting the sleep duration, but also obtain sleep data from individuals with known brain disorders.”
The study findings were limited by several other factors, including the use of total sleep duration only, without other measures of sleep hygiene, the researchers noted. More research is needed to investigate the mechanisms driving the association between too much and not enough sleep and poor mental health and cognitive function.
The study was supported by the National Key R&D Program of China, the Shanghai Municipal Science and Technology Major Project, the Shanghai Center for Brain Science and Brain-Inspired Technology, the 111 Project, the National Natural Sciences Foundation of China and the Shanghai Rising Star Program.
The researchers had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the editorial advisory board of Internal Medicine News.
Sleep disturbances are common in older age, and previous studies have shown associations between too much or too little sleep and increased risk of cognitive decline, but the ideal amount of sleep for preserving mental health has not been well described, according to the authors of the new paper.
In the study published in Nature Aging, the team of researchers from China and the United Kingdom reviewed data from the UK Biobank, a national database of individuals in the United Kingdom that includes cognitive assessments, mental health questionnaires, and brain imaging data, as well as genetic information.
Sleep is important for physical and psychological health, and also serves a neuroprotective function by clearing waste products from the brain, lead author Yuzhu Li of Fudan University, Shanghai, China, and colleagues wrote.
The study population included 498,277 participants, aged 38-73 years, who completed touchscreen questionnaires about sleep duration between 2006 and 2010. The average age at baseline was 56.5 years, 54% were female, and the mean sleep duration was 7.15 hours.
The researchers also reviewed brain imaging data and genetic data from 39,692 participants in 2014 to examine the relationships between sleep duration and brain structure and between sleep duration and genetic risk. In addition, 156,884 participants completed an online follow-up mental health questionnaire in 2016-2017 to assess the longitudinal impact of sleep on mental health.
Both excessive and insufficient sleep was associated with impaired cognitive performance, evidenced by the U-shaped curve found by the researchers in their data analysis, which used quadratic associations.
Specific cognitive functions including pair matching, trail making, prospective memory, and reaction time were significantly impaired with too much or too little sleep, the researchers said. “This demonstrated the positive association of both insufficient and excessive sleep duration with inferior performance on cognitive tasks.”
When the researchers analyzed the association between sleep duration and mental health, sleep duration also showed a U-shaped association with symptoms of anxiety, depression, mental distress, mania, and self-harm, while well-being showed an inverted U-shape. All associations between sleep duration and mental health were statistically significant after controlling for confounding variables (P < .001).
On further analysis (using two-line tests), the researchers determined that consistent sleep duration of approximately 7 hours per night was optimal for cognitive performance and for good mental health.
The researchers also used neuroimaging data to examine the relationship between sleep duration and brain structure. Overall, greater changes were seen in the regions of the brain involved in cognitive processing and memory.
“The most significant cortical volumes nonlinearly associated with sleep duration included the precentral cortex, the superior frontal gyrus, the lateral orbitofrontal cortex, the pars orbitalis, the frontal pole, and the middle temporal cortex,” the researchers wrote (P < .05 for all).
The association between sleep duration and cognitive function diminished among individuals older than 65 years, compared with those aged approximately 40 years, which suggests that optimal sleep duration may be more beneficial in middle age, the researchers noted. However, no similar impact of age was seen for mental health. For brain structure, the nonlinear relationship between sleep duration and cortical volumes was greatest in those aged 44-59 years, and gradually flattened with older age.
Research supports sleep discussions with patients
“Primary care physicians can use this study in their discussions with middle-aged and older patients to recommend optimal sleep duration and measures to achieve this sleep target,” Noel Deep, MD, a general internist in group practice in Antigo, Wisc., who was not involved in the study, said in an interview.
“This study is important because it demonstrated that both inadequate and excessive sleep patterns were associated with cognitive and mental health changes,” said Dr. Deep. “It supported previous observations of cognitive decline and mental health disorders being linked to disturbed sleep. But this study was unique because it provides data supporting an optimal sleep duration of 7 hours and the ill effects of both insufficient and excessive sleep duration.
“The usual thought process has been to assume that older individuals may not require as much sleep as the younger individuals, but this study supports an optimal time duration of sleep of 7 hours that benefits the older individuals. It was also interesting to note the mental health effects caused by the inadequate and excessive sleep durations,” he added.
As for additional research, “I would like to look into the quality of the sleep, in addition to the duration of sleep,” said Dr. Deep. For example, whether the excessive sleep was caused by poor quality sleep or fragmented sleep leading to the structural and subsequent cognitive decline.
Study limitations
“The current study relied on self-reporting of the sleep duration and was not observed and recorded data,” Dr. Deep noted. “It would also be beneficial to not only rely on healthy volunteers reporting the sleep duration, but also obtain sleep data from individuals with known brain disorders.”
The study findings were limited by several other factors, including the use of total sleep duration only, without other measures of sleep hygiene, the researchers noted. More research is needed to investigate the mechanisms driving the association between too much and not enough sleep and poor mental health and cognitive function.
The study was supported by the National Key R&D Program of China, the Shanghai Municipal Science and Technology Major Project, the Shanghai Center for Brain Science and Brain-Inspired Technology, the 111 Project, the National Natural Sciences Foundation of China and the Shanghai Rising Star Program.
The researchers had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the editorial advisory board of Internal Medicine News.
Sleep disturbances are common in older age, and previous studies have shown associations between too much or too little sleep and increased risk of cognitive decline, but the ideal amount of sleep for preserving mental health has not been well described, according to the authors of the new paper.
In the study published in Nature Aging, the team of researchers from China and the United Kingdom reviewed data from the UK Biobank, a national database of individuals in the United Kingdom that includes cognitive assessments, mental health questionnaires, and brain imaging data, as well as genetic information.
Sleep is important for physical and psychological health, and also serves a neuroprotective function by clearing waste products from the brain, lead author Yuzhu Li of Fudan University, Shanghai, China, and colleagues wrote.
The study population included 498,277 participants, aged 38-73 years, who completed touchscreen questionnaires about sleep duration between 2006 and 2010. The average age at baseline was 56.5 years, 54% were female, and the mean sleep duration was 7.15 hours.
The researchers also reviewed brain imaging data and genetic data from 39,692 participants in 2014 to examine the relationships between sleep duration and brain structure and between sleep duration and genetic risk. In addition, 156,884 participants completed an online follow-up mental health questionnaire in 2016-2017 to assess the longitudinal impact of sleep on mental health.
Both excessive and insufficient sleep was associated with impaired cognitive performance, evidenced by the U-shaped curve found by the researchers in their data analysis, which used quadratic associations.
Specific cognitive functions including pair matching, trail making, prospective memory, and reaction time were significantly impaired with too much or too little sleep, the researchers said. “This demonstrated the positive association of both insufficient and excessive sleep duration with inferior performance on cognitive tasks.”
When the researchers analyzed the association between sleep duration and mental health, sleep duration also showed a U-shaped association with symptoms of anxiety, depression, mental distress, mania, and self-harm, while well-being showed an inverted U-shape. All associations between sleep duration and mental health were statistically significant after controlling for confounding variables (P < .001).
On further analysis (using two-line tests), the researchers determined that consistent sleep duration of approximately 7 hours per night was optimal for cognitive performance and for good mental health.
The researchers also used neuroimaging data to examine the relationship between sleep duration and brain structure. Overall, greater changes were seen in the regions of the brain involved in cognitive processing and memory.
“The most significant cortical volumes nonlinearly associated with sleep duration included the precentral cortex, the superior frontal gyrus, the lateral orbitofrontal cortex, the pars orbitalis, the frontal pole, and the middle temporal cortex,” the researchers wrote (P < .05 for all).
The association between sleep duration and cognitive function diminished among individuals older than 65 years, compared with those aged approximately 40 years, which suggests that optimal sleep duration may be more beneficial in middle age, the researchers noted. However, no similar impact of age was seen for mental health. For brain structure, the nonlinear relationship between sleep duration and cortical volumes was greatest in those aged 44-59 years, and gradually flattened with older age.
Research supports sleep discussions with patients
“Primary care physicians can use this study in their discussions with middle-aged and older patients to recommend optimal sleep duration and measures to achieve this sleep target,” Noel Deep, MD, a general internist in group practice in Antigo, Wisc., who was not involved in the study, said in an interview.
“This study is important because it demonstrated that both inadequate and excessive sleep patterns were associated with cognitive and mental health changes,” said Dr. Deep. “It supported previous observations of cognitive decline and mental health disorders being linked to disturbed sleep. But this study was unique because it provides data supporting an optimal sleep duration of 7 hours and the ill effects of both insufficient and excessive sleep duration.
“The usual thought process has been to assume that older individuals may not require as much sleep as the younger individuals, but this study supports an optimal time duration of sleep of 7 hours that benefits the older individuals. It was also interesting to note the mental health effects caused by the inadequate and excessive sleep durations,” he added.
As for additional research, “I would like to look into the quality of the sleep, in addition to the duration of sleep,” said Dr. Deep. For example, whether the excessive sleep was caused by poor quality sleep or fragmented sleep leading to the structural and subsequent cognitive decline.
Study limitations
“The current study relied on self-reporting of the sleep duration and was not observed and recorded data,” Dr. Deep noted. “It would also be beneficial to not only rely on healthy volunteers reporting the sleep duration, but also obtain sleep data from individuals with known brain disorders.”
The study findings were limited by several other factors, including the use of total sleep duration only, without other measures of sleep hygiene, the researchers noted. More research is needed to investigate the mechanisms driving the association between too much and not enough sleep and poor mental health and cognitive function.
The study was supported by the National Key R&D Program of China, the Shanghai Municipal Science and Technology Major Project, the Shanghai Center for Brain Science and Brain-Inspired Technology, the 111 Project, the National Natural Sciences Foundation of China and the Shanghai Rising Star Program.
The researchers had no financial conflicts to disclose. Dr. Deep had no financial conflicts to disclose, but serves on the editorial advisory board of Internal Medicine News.
FROM NATURE AGING
Deprescribing in older adults: An overview
Mr. J, age 73, has a 25-year history of generalized anxiety disorder and major depressive disorder. His medical history includes hypertension, hyperlipidemia, type 2 diabetes mellitus, hypothyroidism, osteoarthritis, insomnia, and allergic rhinitis. His last laboratory test results indicate his hemoglobin A1c, thyroid-stimulating hormone, low-density lipoprotein, and blood pressure measurements are at goal. He believes his conditions are well controlled but cites concerns about taking multiple medications each day and being able to afford his medications.
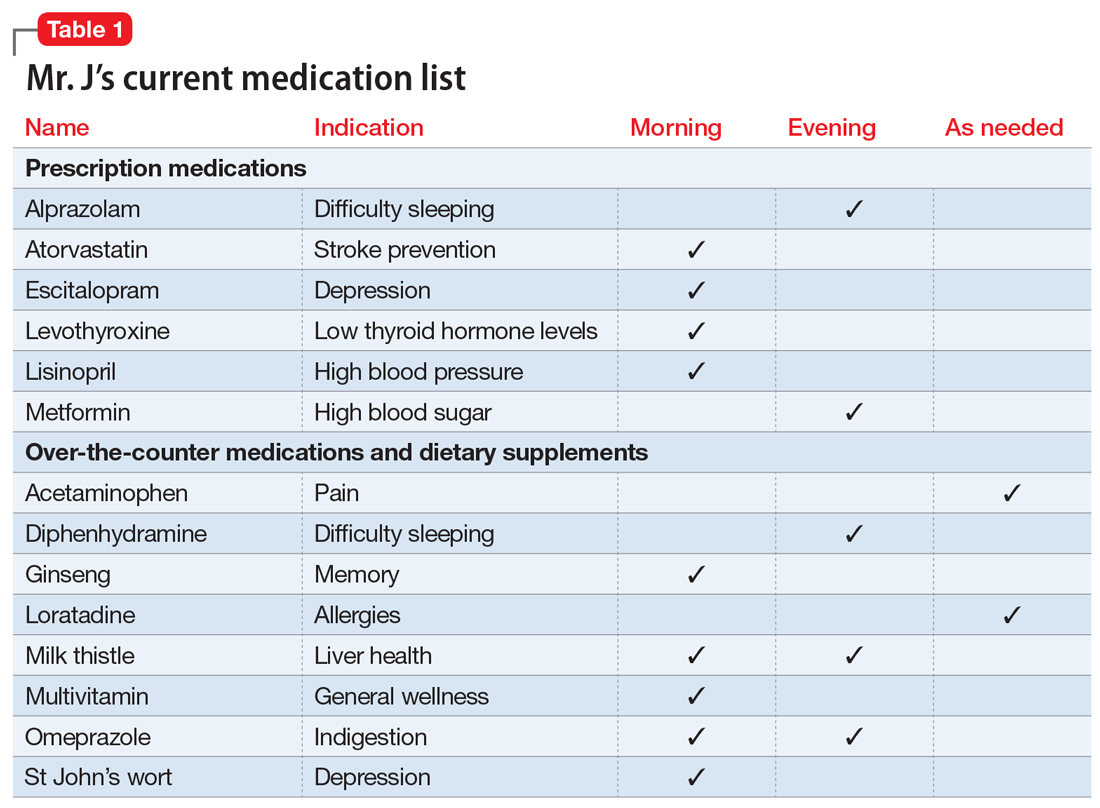
You review the list of Mr. J’s current prescription medications, which include alprazolam 0.5 mg/d, atorvastatin 40 mg/d, escitalopram 10 mg/d, levothyroxine 0.125 mg/d, lisinopril 20 mg/d, and metformin XR 1,000 mg/d. Mr. J reports taking over-the-counter (OTC) acetaminophen as needed for pain, diphenhydramine for insomnia, loratadine as needed for allergic rhinitis, and omeprazole for 2 years for indigestion. After further questioning, he also reports taking ginseng, milk thistle, a multivitamin, and, based on a friend’s recommendation, St John’s Wort (Table 1).
Similar to Mr. J, many older adults take multiple medications to manage chronic health conditions and promote their overall health. On average, 30% of older adults take ≥5 medications.1 Among commonly prescribed medications for these patients, an estimated 1 in 5 of may be inappropriate.1 Older adults have high rates of polypharmacy (often defined as taking ≥5 medications1), age-related physiological changes, increased number of comorbidities, and frailty, all of which can increase the risk of medication-related adverse events.2 As a result, older patients’ medications should be regularly evaluated to determine if each medication is appropriate to continue or should be tapered or stopped.
Deprescribing, in which medications are tapered or discontinued using a patient-centered approach, should be considered when a patient is no longer receiving benefit from a medication, or when the harm may exceed the benefit.1,3
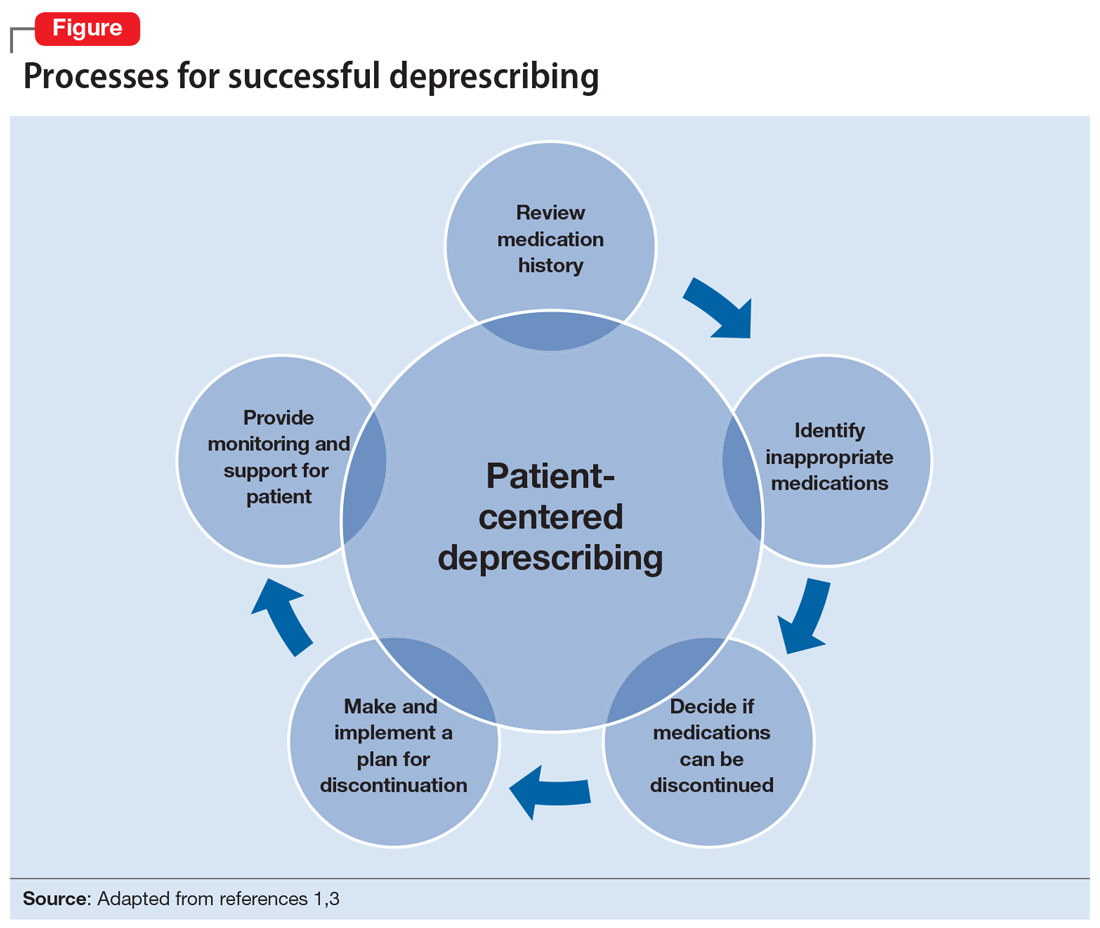
Several researchers1,3 and organizations have published detailed descriptions of and guidelines for the process of deprescribing (see Related Resources). Here we provide a brief overview of this process (Figure1,3). The first step is to assemble a list of all prescription and OTC medications, herbal products, vitamins, or nutritional supplements the patient is taking. It is important to specifically ask patients about their use of nonprescription products, because these products are infrequently documented in medical records.
The second step is to evaluate the indication, effectiveness, safety, and patient’s adherence to each medication while beginning to consider opportunities to limit treatment burden and the risk of harm from medications. Ideally, this assessment should involve a patient-centered conversation that considers the patient’s goals, preferences, and treatment values. Many resources can be used to evaluate which medications might be inappropriate for an older adult. Two examples are the American Geriatrics Society Beers Criteria5 and STOPP/START criteria.6 By looking at these resources, you could identify that (for example) anticholinergic medications should be avoided in older patients due to an increased risk of adverse effects, change in cognitive status, and falls.5,6 These resources can aid in identifying, prioritizing, and deprescribing potentially harmful and/or inappropriate medications.
The next step is to decide whether any medications should be discontinued. Whenever possible, include the patient in this conversation, as they may have strong feelings about their current medication regimen. When there are multiple medications that can be discontinued, consider which medication to stop first based on potential harm, patient resistance, and other factors.
Continue to: Subsequently, work with...
Subsequently, work with the patient to create a plan for stopping or lowering the dose or frequency of the medication. These changes should be individualized based on the patient’s preferences as well as the properties of the medication. For example, some medications can be immediately discontinued, while others (eg, benzodiazepines) may need to be slowly tapered. It is important to consider if the patient will need to switch to a safer medication, change their behaviors (eg, lifestyle changes), or engage in alternative treatments (such as cognitive-behavioral therapy for insomnia) when they stop their current medication. Take an active role in monitoring your patient during this process, and encourage them to reach out to you or to their primary clinician if they have concerns.
CASE CONTINUED
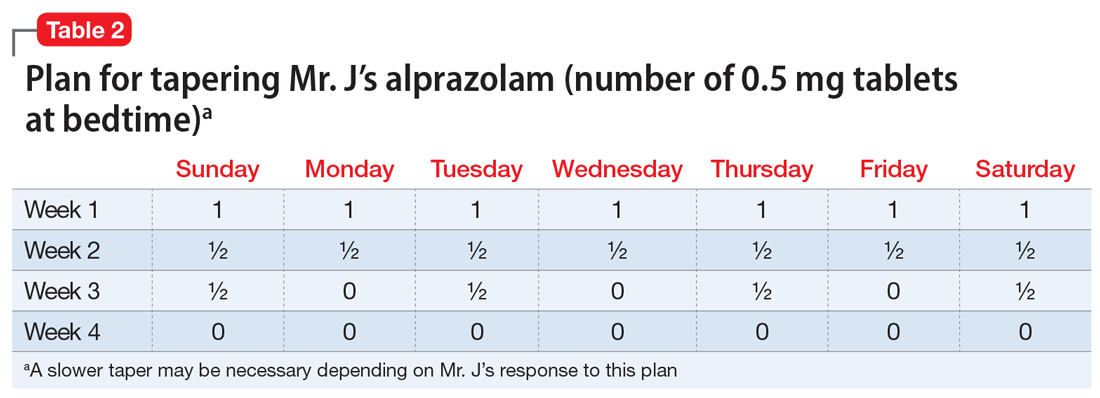
Mr. J is a candidate for deprescribing because he has expressed concerns about his current regimen, and because he is taking potentially unsafe medications. The 2 medications he’s taking that may cause the most harm are diphenhydramine and alprazolam, due to the risk of cognitive impairment and falls. Through a patient-centered conversation, Mr. J says he is willing to stop diphenhydramine immediately and taper off the alprazolam over the next month, with the support of a tapering chart (Table 2). You explain to him that a long tapering of alprazolam may be necessary. He is willing to try good sleep hygiene practices and will put off starting trazodone as an alternative to diphenhydramine until he sees if it will be necessary. You make a note to follow up with him in 1 week to assess his insomnia and adherence to the new treatment plan. You also teach Mr. J that some of his supplements may interact with his prescription medications, such as St John’s Wort with escitalopram (ie, risk of serotonin syndrome) and ginseng with metformin (ie, risk for hypoglycemia). He says he doesn’t take ginseng, milk thistle, or St John’s Wort regularly, and because he feels they do not offer any benefit, he will stop taking them. He says that at his next visit with his primary care physician, he will bring up the idea of stopping omeprazole.
Related Resources
- Deprescribing.org. Deprescribing guidelines and algorithms. https://deprescribing.org/resources/deprescribing-guidelines-algorithms/
- US Deprescribing Research Network. Resources for Clinicians. https://deprescribingresearch.org/resources-2/resources-for-clinicians/
Drug Brand Names
Alprazolam • Xanax
Atorvastatin • Lipitor
Escitalopram • Lexapro
Levothyroxine • Synthroid
Lisinopril • Zestril
Metformin XR • Glucophage XR
Trazodone • Desyrel
1. Scott IA, Hilmer SN, Reeve E, et al. Reducing inappropriate polypharmacy: the process of deprescribing. JAMA Intern Med. 2015;175(5):827-834.
2. Gibson G, Kennedy LH, Barlow G. Polypharmacy in older adults. Current Psychiatry. 2020;19(4):40-46.
3. Reeve E, Shakib S, Hendrix I, et al. Review of deprescribing processes and development of an evidence-based, patient-centred deprescribing process. Br J Clin Pharmcol. 2014;78(4):738-747.
4. Iyer S, Naganathan V, McLachlan AJ, et al. Medication withdrawal trials in people aged 65 years and older: a systematic review. Drugs Aging. 2008;25(12):1021-1031.
5. 2019 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2019 updated AGS Beers Criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2019;67(4):674-694.
6. O’Mahony D, O’Sullivan D, Byrne S, et al. STOPP/START criteria for potentially inappropriate prescribing in older people: version 2. Age Ageing. 2015;44(2):213-218.
Mr. J, age 73, has a 25-year history of generalized anxiety disorder and major depressive disorder. His medical history includes hypertension, hyperlipidemia, type 2 diabetes mellitus, hypothyroidism, osteoarthritis, insomnia, and allergic rhinitis. His last laboratory test results indicate his hemoglobin A1c, thyroid-stimulating hormone, low-density lipoprotein, and blood pressure measurements are at goal. He believes his conditions are well controlled but cites concerns about taking multiple medications each day and being able to afford his medications.
You review the list of Mr. J’s current prescription medications, which include alprazolam 0.5 mg/d, atorvastatin 40 mg/d, escitalopram 10 mg/d, levothyroxine 0.125 mg/d, lisinopril 20 mg/d, and metformin XR 1,000 mg/d. Mr. J reports taking over-the-counter (OTC) acetaminophen as needed for pain, diphenhydramine for insomnia, loratadine as needed for allergic rhinitis, and omeprazole for 2 years for indigestion. After further questioning, he also reports taking ginseng, milk thistle, a multivitamin, and, based on a friend’s recommendation, St John’s Wort (Table 1).
Similar to Mr. J, many older adults take multiple medications to manage chronic health conditions and promote their overall health. On average, 30% of older adults take ≥5 medications.1 Among commonly prescribed medications for these patients, an estimated 1 in 5 of may be inappropriate.1 Older adults have high rates of polypharmacy (often defined as taking ≥5 medications1), age-related physiological changes, increased number of comorbidities, and frailty, all of which can increase the risk of medication-related adverse events.2 As a result, older patients’ medications should be regularly evaluated to determine if each medication is appropriate to continue or should be tapered or stopped.
Deprescribing, in which medications are tapered or discontinued using a patient-centered approach, should be considered when a patient is no longer receiving benefit from a medication, or when the harm may exceed the benefit.1,3
Several researchers1,3 and organizations have published detailed descriptions of and guidelines for the process of deprescribing (see Related Resources). Here we provide a brief overview of this process (Figure1,3). The first step is to assemble a list of all prescription and OTC medications, herbal products, vitamins, or nutritional supplements the patient is taking. It is important to specifically ask patients about their use of nonprescription products, because these products are infrequently documented in medical records.
The second step is to evaluate the indication, effectiveness, safety, and patient’s adherence to each medication while beginning to consider opportunities to limit treatment burden and the risk of harm from medications. Ideally, this assessment should involve a patient-centered conversation that considers the patient’s goals, preferences, and treatment values. Many resources can be used to evaluate which medications might be inappropriate for an older adult. Two examples are the American Geriatrics Society Beers Criteria5 and STOPP/START criteria.6 By looking at these resources, you could identify that (for example) anticholinergic medications should be avoided in older patients due to an increased risk of adverse effects, change in cognitive status, and falls.5,6 These resources can aid in identifying, prioritizing, and deprescribing potentially harmful and/or inappropriate medications.
The next step is to decide whether any medications should be discontinued. Whenever possible, include the patient in this conversation, as they may have strong feelings about their current medication regimen. When there are multiple medications that can be discontinued, consider which medication to stop first based on potential harm, patient resistance, and other factors.
Continue to: Subsequently, work with...
Subsequently, work with the patient to create a plan for stopping or lowering the dose or frequency of the medication. These changes should be individualized based on the patient’s preferences as well as the properties of the medication. For example, some medications can be immediately discontinued, while others (eg, benzodiazepines) may need to be slowly tapered. It is important to consider if the patient will need to switch to a safer medication, change their behaviors (eg, lifestyle changes), or engage in alternative treatments (such as cognitive-behavioral therapy for insomnia) when they stop their current medication. Take an active role in monitoring your patient during this process, and encourage them to reach out to you or to their primary clinician if they have concerns.
CASE CONTINUED
Mr. J is a candidate for deprescribing because he has expressed concerns about his current regimen, and because he is taking potentially unsafe medications. The 2 medications he’s taking that may cause the most harm are diphenhydramine and alprazolam, due to the risk of cognitive impairment and falls. Through a patient-centered conversation, Mr. J says he is willing to stop diphenhydramine immediately and taper off the alprazolam over the next month, with the support of a tapering chart (Table 2). You explain to him that a long tapering of alprazolam may be necessary. He is willing to try good sleep hygiene practices and will put off starting trazodone as an alternative to diphenhydramine until he sees if it will be necessary. You make a note to follow up with him in 1 week to assess his insomnia and adherence to the new treatment plan. You also teach Mr. J that some of his supplements may interact with his prescription medications, such as St John’s Wort with escitalopram (ie, risk of serotonin syndrome) and ginseng with metformin (ie, risk for hypoglycemia). He says he doesn’t take ginseng, milk thistle, or St John’s Wort regularly, and because he feels they do not offer any benefit, he will stop taking them. He says that at his next visit with his primary care physician, he will bring up the idea of stopping omeprazole.
Related Resources
- Deprescribing.org. Deprescribing guidelines and algorithms. https://deprescribing.org/resources/deprescribing-guidelines-algorithms/
- US Deprescribing Research Network. Resources for Clinicians. https://deprescribingresearch.org/resources-2/resources-for-clinicians/
Drug Brand Names
Alprazolam • Xanax
Atorvastatin • Lipitor
Escitalopram • Lexapro
Levothyroxine • Synthroid
Lisinopril • Zestril
Metformin XR • Glucophage XR
Trazodone • Desyrel
Mr. J, age 73, has a 25-year history of generalized anxiety disorder and major depressive disorder. His medical history includes hypertension, hyperlipidemia, type 2 diabetes mellitus, hypothyroidism, osteoarthritis, insomnia, and allergic rhinitis. His last laboratory test results indicate his hemoglobin A1c, thyroid-stimulating hormone, low-density lipoprotein, and blood pressure measurements are at goal. He believes his conditions are well controlled but cites concerns about taking multiple medications each day and being able to afford his medications.
You review the list of Mr. J’s current prescription medications, which include alprazolam 0.5 mg/d, atorvastatin 40 mg/d, escitalopram 10 mg/d, levothyroxine 0.125 mg/d, lisinopril 20 mg/d, and metformin XR 1,000 mg/d. Mr. J reports taking over-the-counter (OTC) acetaminophen as needed for pain, diphenhydramine for insomnia, loratadine as needed for allergic rhinitis, and omeprazole for 2 years for indigestion. After further questioning, he also reports taking ginseng, milk thistle, a multivitamin, and, based on a friend’s recommendation, St John’s Wort (Table 1).
Similar to Mr. J, many older adults take multiple medications to manage chronic health conditions and promote their overall health. On average, 30% of older adults take ≥5 medications.1 Among commonly prescribed medications for these patients, an estimated 1 in 5 of may be inappropriate.1 Older adults have high rates of polypharmacy (often defined as taking ≥5 medications1), age-related physiological changes, increased number of comorbidities, and frailty, all of which can increase the risk of medication-related adverse events.2 As a result, older patients’ medications should be regularly evaluated to determine if each medication is appropriate to continue or should be tapered or stopped.
Deprescribing, in which medications are tapered or discontinued using a patient-centered approach, should be considered when a patient is no longer receiving benefit from a medication, or when the harm may exceed the benefit.1,3
Several researchers1,3 and organizations have published detailed descriptions of and guidelines for the process of deprescribing (see Related Resources). Here we provide a brief overview of this process (Figure1,3). The first step is to assemble a list of all prescription and OTC medications, herbal products, vitamins, or nutritional supplements the patient is taking. It is important to specifically ask patients about their use of nonprescription products, because these products are infrequently documented in medical records.
The second step is to evaluate the indication, effectiveness, safety, and patient’s adherence to each medication while beginning to consider opportunities to limit treatment burden and the risk of harm from medications. Ideally, this assessment should involve a patient-centered conversation that considers the patient’s goals, preferences, and treatment values. Many resources can be used to evaluate which medications might be inappropriate for an older adult. Two examples are the American Geriatrics Society Beers Criteria5 and STOPP/START criteria.6 By looking at these resources, you could identify that (for example) anticholinergic medications should be avoided in older patients due to an increased risk of adverse effects, change in cognitive status, and falls.5,6 These resources can aid in identifying, prioritizing, and deprescribing potentially harmful and/or inappropriate medications.
The next step is to decide whether any medications should be discontinued. Whenever possible, include the patient in this conversation, as they may have strong feelings about their current medication regimen. When there are multiple medications that can be discontinued, consider which medication to stop first based on potential harm, patient resistance, and other factors.
Continue to: Subsequently, work with...
Subsequently, work with the patient to create a plan for stopping or lowering the dose or frequency of the medication. These changes should be individualized based on the patient’s preferences as well as the properties of the medication. For example, some medications can be immediately discontinued, while others (eg, benzodiazepines) may need to be slowly tapered. It is important to consider if the patient will need to switch to a safer medication, change their behaviors (eg, lifestyle changes), or engage in alternative treatments (such as cognitive-behavioral therapy for insomnia) when they stop their current medication. Take an active role in monitoring your patient during this process, and encourage them to reach out to you or to their primary clinician if they have concerns.
CASE CONTINUED
Mr. J is a candidate for deprescribing because he has expressed concerns about his current regimen, and because he is taking potentially unsafe medications. The 2 medications he’s taking that may cause the most harm are diphenhydramine and alprazolam, due to the risk of cognitive impairment and falls. Through a patient-centered conversation, Mr. J says he is willing to stop diphenhydramine immediately and taper off the alprazolam over the next month, with the support of a tapering chart (Table 2). You explain to him that a long tapering of alprazolam may be necessary. He is willing to try good sleep hygiene practices and will put off starting trazodone as an alternative to diphenhydramine until he sees if it will be necessary. You make a note to follow up with him in 1 week to assess his insomnia and adherence to the new treatment plan. You also teach Mr. J that some of his supplements may interact with his prescription medications, such as St John’s Wort with escitalopram (ie, risk of serotonin syndrome) and ginseng with metformin (ie, risk for hypoglycemia). He says he doesn’t take ginseng, milk thistle, or St John’s Wort regularly, and because he feels they do not offer any benefit, he will stop taking them. He says that at his next visit with his primary care physician, he will bring up the idea of stopping omeprazole.
Related Resources
- Deprescribing.org. Deprescribing guidelines and algorithms. https://deprescribing.org/resources/deprescribing-guidelines-algorithms/
- US Deprescribing Research Network. Resources for Clinicians. https://deprescribingresearch.org/resources-2/resources-for-clinicians/
Drug Brand Names
Alprazolam • Xanax
Atorvastatin • Lipitor
Escitalopram • Lexapro
Levothyroxine • Synthroid
Lisinopril • Zestril
Metformin XR • Glucophage XR
Trazodone • Desyrel
1. Scott IA, Hilmer SN, Reeve E, et al. Reducing inappropriate polypharmacy: the process of deprescribing. JAMA Intern Med. 2015;175(5):827-834.
2. Gibson G, Kennedy LH, Barlow G. Polypharmacy in older adults. Current Psychiatry. 2020;19(4):40-46.
3. Reeve E, Shakib S, Hendrix I, et al. Review of deprescribing processes and development of an evidence-based, patient-centred deprescribing process. Br J Clin Pharmcol. 2014;78(4):738-747.
4. Iyer S, Naganathan V, McLachlan AJ, et al. Medication withdrawal trials in people aged 65 years and older: a systematic review. Drugs Aging. 2008;25(12):1021-1031.
5. 2019 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2019 updated AGS Beers Criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2019;67(4):674-694.
6. O’Mahony D, O’Sullivan D, Byrne S, et al. STOPP/START criteria for potentially inappropriate prescribing in older people: version 2. Age Ageing. 2015;44(2):213-218.
1. Scott IA, Hilmer SN, Reeve E, et al. Reducing inappropriate polypharmacy: the process of deprescribing. JAMA Intern Med. 2015;175(5):827-834.
2. Gibson G, Kennedy LH, Barlow G. Polypharmacy in older adults. Current Psychiatry. 2020;19(4):40-46.
3. Reeve E, Shakib S, Hendrix I, et al. Review of deprescribing processes and development of an evidence-based, patient-centred deprescribing process. Br J Clin Pharmcol. 2014;78(4):738-747.
4. Iyer S, Naganathan V, McLachlan AJ, et al. Medication withdrawal trials in people aged 65 years and older: a systematic review. Drugs Aging. 2008;25(12):1021-1031.
5. 2019 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2019 updated AGS Beers Criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2019;67(4):674-694.
6. O’Mahony D, O’Sullivan D, Byrne S, et al. STOPP/START criteria for potentially inappropriate prescribing in older people: version 2. Age Ageing. 2015;44(2):213-218.
‘Where does it hurt?’: Primary care tips for common ortho problems
Knee and shoulder pain are common complaints for patients in the primary care office.
But identifying the source of the pain can be complicated,
and an accurate diagnosis of the underlying cause of discomfort is key to appropriate management – whether that involves simple home care options of ice and rest or a recommendation for a follow-up with a specialist.
Speaking at the annual meeting of the American College of Physicians, Greg Nakamoto, MD, department of orthopedics, Virginia Mason Medical Center, Seattle, discussed common knee and shoulder problems that patients often present with in the primary care setting, and offered tips on diagnosis and appropriate management.
The most common conditions causing knee pain are osteoarthritis and meniscal tears. “The differential for knee pain is broad,” Dr. Nakamoto said. “You have to have a way to divide it down, such as if it’s acute or chronic.”
The initial workup has several key components. The first steps: Determine the location of the pain – anterior, medial, lateral, posterior – and then whether it stems from an injury or is atraumatic.
“If you have to ask one question – ask where it hurts,” he said. “And is it from an injury or just wear and tear? That helps me when deciding if surgery is needed.”
Pain in the knee generally localizes well to the site of pathology, and knee pain of acute traumatic onset requires more scrutiny for problems best treated with early surgery. “This also helps establish whether radiographic findings are due to injury or degeneration,” Dr. Nakamoto said. “The presence of swelling guides the need for anti-inflammatories or cortisone.”
Palpating for tenderness along the joint line is important, as is palpating above and below the joint line, Dr. Nakamoto said.
“Tenderness limited to the joint line, combined with a meniscal exam maneuver that reproduces joint-line pain, is suggestive of pain from meniscal pathology,” he said.
Imaging is an important component of evaluating knee symptoms, and the question often arises as to when to order an MRI.
Dr. Nakamoto offered the following scenario: If significant osteoarthritis is evident on weight-bearing x-ray, treat the patient for the condition. However, if little or no osteoarthritis appears on x-ray, and if the onset of symptoms was traumatic and both patient history and physical examination suggest a meniscal tear, order an MRI.
An early MRI also is needed if the patient has had either atraumatic or traumatic onset of symptoms and their history and physical exams are suspicious for a mechanically locked or locking meniscus. For suspicion of a ruptured quadriceps or patellar tendon or a stress fracture, an MRI is needed urgently.
An MRI would be ordered later if the patient’s symptoms have not improved significantly after 3 months of conservative management.
Dr. Nakamoto stressed how common undiagnosed meniscus tears are in the general population. A third of men aged 50-59 years and nearly 20% of women in that age group have a tear, he said. “That number goes up to 56% and 51% in men and women aged 70-90 years, and 61% of these tears were in patients who were asymptomatic in the last month.”
In the setting of osteoarthritis, 76% of asymptomatic patients had a meniscus tear, and 91% of patients with symptomatic osteoarthritis had a meniscus tear, he added.
Treating knee pain
Treatment will vary depending on the underlying etiology of pain. For a possible meniscus tear, the recommendation is for a conservative intervention with ice, ibuprofen, knee immobilizer, and crutches, with a follow-up appointment in a week.
Three types of injections also can help:
- Cortisone for osteoarthritis or meniscus tears, swelling, and inflammation, and prophylaxis against inflammation.
- Viscosupplementation (intra‐articular hyaluronic acid) for chronic, baseline osteoarthritis symptoms.
- Regenerative therapies (platelet-rich plasma, stem cells, etc.) are used primarily for osteoarthritis (these do not regrow cartilage, but some patients report decreased pain).
The data on injections are mixed, Dr. Nakamoto said. For example, the results of a 2015 Cochrane review on cortisone injections for osteoarthritis reported that the benefits were small to moderate at 4‐6 weeks, and small to none at 13 weeks.
“There is a lot of controversy for viscosupplementation despite all of the data on it,” he said. “But the recommendations from professional organizations are mixed.”
He noted that he has been using viscosupplementation since the 1990s, and some patients do benefit from it.
Shoulder pain
The most common causes of shoulder pain are adhesive capsulitis, rotator cuff tears and tendinopathy, and impingement.
As with knee pain, the same assessment routine largely applies.
First, pinpoint the location: Is the trouble spot the lateral shoulder and upper arm, the trapezial ridge, or the shoulder blade?
Next, assess pain on movement: Does the patient experience discomfort reaching overhead or behind the back, or moving at the glenohumeral joint/capsule and engaging the rotator cuff? Check for stiffness, weakness, and decreased range of motion in the rotator cuff.
Determine if the cause of the pain is traumatic or atraumatic and stems from an acute injury versus degeneration or overuse.
As with the knee, imaging is a major component of the assessment and typically involves the use of x-ray. An MRI may be required for evaluating full- and partial-thickness tears and when contemplating surgery.
MRI also is necessary for evaluating cases of acute, traumatic shoulder injury, and patients exhibiting disability suggestive of a rotator cuff tear in an otherwise healthy tendon.
Some pain can be treated with cortisone injections or regenerative therapies, which generally are given at the acromioclavicular or glenohumeral joints or in the subacromial space. A 2005 meta-analysis found that subacromial injections of corticosteroids are effective for improvement for rotator cuff tendinitis up to a 9‐month period.
Surgery may be warranted in some cases, Dr. Nakamoto said. These include adhesive capsulitis, rotator cuff tear, acute traumatic injury in an otherwise healthy tendon, and chronic (or acute-on-chronic) tears in a degenerative tendon following a trial of conservative therapy.
A version of this article first appeared on Medscape.com.
Knee and shoulder pain are common complaints for patients in the primary care office.
But identifying the source of the pain can be complicated,
and an accurate diagnosis of the underlying cause of discomfort is key to appropriate management – whether that involves simple home care options of ice and rest or a recommendation for a follow-up with a specialist.
Speaking at the annual meeting of the American College of Physicians, Greg Nakamoto, MD, department of orthopedics, Virginia Mason Medical Center, Seattle, discussed common knee and shoulder problems that patients often present with in the primary care setting, and offered tips on diagnosis and appropriate management.
The most common conditions causing knee pain are osteoarthritis and meniscal tears. “The differential for knee pain is broad,” Dr. Nakamoto said. “You have to have a way to divide it down, such as if it’s acute or chronic.”
The initial workup has several key components. The first steps: Determine the location of the pain – anterior, medial, lateral, posterior – and then whether it stems from an injury or is atraumatic.
“If you have to ask one question – ask where it hurts,” he said. “And is it from an injury or just wear and tear? That helps me when deciding if surgery is needed.”
Pain in the knee generally localizes well to the site of pathology, and knee pain of acute traumatic onset requires more scrutiny for problems best treated with early surgery. “This also helps establish whether radiographic findings are due to injury or degeneration,” Dr. Nakamoto said. “The presence of swelling guides the need for anti-inflammatories or cortisone.”
Palpating for tenderness along the joint line is important, as is palpating above and below the joint line, Dr. Nakamoto said.
“Tenderness limited to the joint line, combined with a meniscal exam maneuver that reproduces joint-line pain, is suggestive of pain from meniscal pathology,” he said.
Imaging is an important component of evaluating knee symptoms, and the question often arises as to when to order an MRI.
Dr. Nakamoto offered the following scenario: If significant osteoarthritis is evident on weight-bearing x-ray, treat the patient for the condition. However, if little or no osteoarthritis appears on x-ray, and if the onset of symptoms was traumatic and both patient history and physical examination suggest a meniscal tear, order an MRI.
An early MRI also is needed if the patient has had either atraumatic or traumatic onset of symptoms and their history and physical exams are suspicious for a mechanically locked or locking meniscus. For suspicion of a ruptured quadriceps or patellar tendon or a stress fracture, an MRI is needed urgently.
An MRI would be ordered later if the patient’s symptoms have not improved significantly after 3 months of conservative management.
Dr. Nakamoto stressed how common undiagnosed meniscus tears are in the general population. A third of men aged 50-59 years and nearly 20% of women in that age group have a tear, he said. “That number goes up to 56% and 51% in men and women aged 70-90 years, and 61% of these tears were in patients who were asymptomatic in the last month.”
In the setting of osteoarthritis, 76% of asymptomatic patients had a meniscus tear, and 91% of patients with symptomatic osteoarthritis had a meniscus tear, he added.
Treating knee pain
Treatment will vary depending on the underlying etiology of pain. For a possible meniscus tear, the recommendation is for a conservative intervention with ice, ibuprofen, knee immobilizer, and crutches, with a follow-up appointment in a week.
Three types of injections also can help:
- Cortisone for osteoarthritis or meniscus tears, swelling, and inflammation, and prophylaxis against inflammation.
- Viscosupplementation (intra‐articular hyaluronic acid) for chronic, baseline osteoarthritis symptoms.
- Regenerative therapies (platelet-rich plasma, stem cells, etc.) are used primarily for osteoarthritis (these do not regrow cartilage, but some patients report decreased pain).
The data on injections are mixed, Dr. Nakamoto said. For example, the results of a 2015 Cochrane review on cortisone injections for osteoarthritis reported that the benefits were small to moderate at 4‐6 weeks, and small to none at 13 weeks.
“There is a lot of controversy for viscosupplementation despite all of the data on it,” he said. “But the recommendations from professional organizations are mixed.”
He noted that he has been using viscosupplementation since the 1990s, and some patients do benefit from it.
Shoulder pain
The most common causes of shoulder pain are adhesive capsulitis, rotator cuff tears and tendinopathy, and impingement.
As with knee pain, the same assessment routine largely applies.
First, pinpoint the location: Is the trouble spot the lateral shoulder and upper arm, the trapezial ridge, or the shoulder blade?
Next, assess pain on movement: Does the patient experience discomfort reaching overhead or behind the back, or moving at the glenohumeral joint/capsule and engaging the rotator cuff? Check for stiffness, weakness, and decreased range of motion in the rotator cuff.
Determine if the cause of the pain is traumatic or atraumatic and stems from an acute injury versus degeneration or overuse.
As with the knee, imaging is a major component of the assessment and typically involves the use of x-ray. An MRI may be required for evaluating full- and partial-thickness tears and when contemplating surgery.
MRI also is necessary for evaluating cases of acute, traumatic shoulder injury, and patients exhibiting disability suggestive of a rotator cuff tear in an otherwise healthy tendon.
Some pain can be treated with cortisone injections or regenerative therapies, which generally are given at the acromioclavicular or glenohumeral joints or in the subacromial space. A 2005 meta-analysis found that subacromial injections of corticosteroids are effective for improvement for rotator cuff tendinitis up to a 9‐month period.
Surgery may be warranted in some cases, Dr. Nakamoto said. These include adhesive capsulitis, rotator cuff tear, acute traumatic injury in an otherwise healthy tendon, and chronic (or acute-on-chronic) tears in a degenerative tendon following a trial of conservative therapy.
A version of this article first appeared on Medscape.com.
Knee and shoulder pain are common complaints for patients in the primary care office.
But identifying the source of the pain can be complicated,
and an accurate diagnosis of the underlying cause of discomfort is key to appropriate management – whether that involves simple home care options of ice and rest or a recommendation for a follow-up with a specialist.
Speaking at the annual meeting of the American College of Physicians, Greg Nakamoto, MD, department of orthopedics, Virginia Mason Medical Center, Seattle, discussed common knee and shoulder problems that patients often present with in the primary care setting, and offered tips on diagnosis and appropriate management.
The most common conditions causing knee pain are osteoarthritis and meniscal tears. “The differential for knee pain is broad,” Dr. Nakamoto said. “You have to have a way to divide it down, such as if it’s acute or chronic.”
The initial workup has several key components. The first steps: Determine the location of the pain – anterior, medial, lateral, posterior – and then whether it stems from an injury or is atraumatic.
“If you have to ask one question – ask where it hurts,” he said. “And is it from an injury or just wear and tear? That helps me when deciding if surgery is needed.”
Pain in the knee generally localizes well to the site of pathology, and knee pain of acute traumatic onset requires more scrutiny for problems best treated with early surgery. “This also helps establish whether radiographic findings are due to injury or degeneration,” Dr. Nakamoto said. “The presence of swelling guides the need for anti-inflammatories or cortisone.”
Palpating for tenderness along the joint line is important, as is palpating above and below the joint line, Dr. Nakamoto said.
“Tenderness limited to the joint line, combined with a meniscal exam maneuver that reproduces joint-line pain, is suggestive of pain from meniscal pathology,” he said.
Imaging is an important component of evaluating knee symptoms, and the question often arises as to when to order an MRI.
Dr. Nakamoto offered the following scenario: If significant osteoarthritis is evident on weight-bearing x-ray, treat the patient for the condition. However, if little or no osteoarthritis appears on x-ray, and if the onset of symptoms was traumatic and both patient history and physical examination suggest a meniscal tear, order an MRI.
An early MRI also is needed if the patient has had either atraumatic or traumatic onset of symptoms and their history and physical exams are suspicious for a mechanically locked or locking meniscus. For suspicion of a ruptured quadriceps or patellar tendon or a stress fracture, an MRI is needed urgently.
An MRI would be ordered later if the patient’s symptoms have not improved significantly after 3 months of conservative management.
Dr. Nakamoto stressed how common undiagnosed meniscus tears are in the general population. A third of men aged 50-59 years and nearly 20% of women in that age group have a tear, he said. “That number goes up to 56% and 51% in men and women aged 70-90 years, and 61% of these tears were in patients who were asymptomatic in the last month.”
In the setting of osteoarthritis, 76% of asymptomatic patients had a meniscus tear, and 91% of patients with symptomatic osteoarthritis had a meniscus tear, he added.
Treating knee pain
Treatment will vary depending on the underlying etiology of pain. For a possible meniscus tear, the recommendation is for a conservative intervention with ice, ibuprofen, knee immobilizer, and crutches, with a follow-up appointment in a week.
Three types of injections also can help:
- Cortisone for osteoarthritis or meniscus tears, swelling, and inflammation, and prophylaxis against inflammation.
- Viscosupplementation (intra‐articular hyaluronic acid) for chronic, baseline osteoarthritis symptoms.
- Regenerative therapies (platelet-rich plasma, stem cells, etc.) are used primarily for osteoarthritis (these do not regrow cartilage, but some patients report decreased pain).
The data on injections are mixed, Dr. Nakamoto said. For example, the results of a 2015 Cochrane review on cortisone injections for osteoarthritis reported that the benefits were small to moderate at 4‐6 weeks, and small to none at 13 weeks.
“There is a lot of controversy for viscosupplementation despite all of the data on it,” he said. “But the recommendations from professional organizations are mixed.”
He noted that he has been using viscosupplementation since the 1990s, and some patients do benefit from it.
Shoulder pain
The most common causes of shoulder pain are adhesive capsulitis, rotator cuff tears and tendinopathy, and impingement.
As with knee pain, the same assessment routine largely applies.
First, pinpoint the location: Is the trouble spot the lateral shoulder and upper arm, the trapezial ridge, or the shoulder blade?
Next, assess pain on movement: Does the patient experience discomfort reaching overhead or behind the back, or moving at the glenohumeral joint/capsule and engaging the rotator cuff? Check for stiffness, weakness, and decreased range of motion in the rotator cuff.
Determine if the cause of the pain is traumatic or atraumatic and stems from an acute injury versus degeneration or overuse.
As with the knee, imaging is a major component of the assessment and typically involves the use of x-ray. An MRI may be required for evaluating full- and partial-thickness tears and when contemplating surgery.
MRI also is necessary for evaluating cases of acute, traumatic shoulder injury, and patients exhibiting disability suggestive of a rotator cuff tear in an otherwise healthy tendon.
Some pain can be treated with cortisone injections or regenerative therapies, which generally are given at the acromioclavicular or glenohumeral joints or in the subacromial space. A 2005 meta-analysis found that subacromial injections of corticosteroids are effective for improvement for rotator cuff tendinitis up to a 9‐month period.
Surgery may be warranted in some cases, Dr. Nakamoto said. These include adhesive capsulitis, rotator cuff tear, acute traumatic injury in an otherwise healthy tendon, and chronic (or acute-on-chronic) tears in a degenerative tendon following a trial of conservative therapy.
A version of this article first appeared on Medscape.com.
FROM INTERNAL MEDICINE 2022
New blood biomarker to detect early dementia?
Investigators found that plasma concentrations of 2-aminoethyl dihydrogen phosphate and taurine could distinguish adults with early-stage Alzheimer’s disease from cognitively normal adults.
“Our biomarker for early-stage Alzheimer’s disease represents new thinking and is unique from the amyloid-beta and p-tau molecules that are currently being investigated to diagnose AD,” Sandra Banack, PhD, senior scientist, Brain Chemistry Labs, Jackson, Wyoming, told this news organization.
If further studies pan out, Dr. Banack said this biomarker could “easily be transformed into a test to aid clinical evaluations for Alzheimer’s disease.”
The study was published online in PLOS ONE.
New drug target?
The researchers measured concentrations of 2-aminoethyl dihydrogen phosphate and taurine in blood plasma samples in 25 patients (21 men; mean age, 71) with a clinical diagnosis of early-stage Alzheimer’s based on a Clinical Dementia Rating (CDR) score of 0.5, suggesting very mild cognitive impairment, and 25 healthy controls (20 men; mean age, 39).
The concentration of 2-aminoethyl dihydrogen phosphate, normalized by the concentration of taurine, reliably distinguished blood samples of early-stage Alzheimer’s patients from controls in a blinded analysis.
This biomarker “could lead to new understanding of [AD] and lead to new drug candidates,” Dr. Banack told this news organization.
The researchers note that 2-aminoethyl dihydrogen phosphate plays an important role in the structure and function of cellular membranes.
Physiologic effects of increased 2-aminoethyl dihydrogen phosphate concentrations in the blood are not known. However, in one study, concentrations of this molecule were found to be significantly lower in the temporal cortex, frontal cortex, and hippocampus (40%) in patients with Alzheimer’s disease, compared with controls.
“New biomarkers take time before they can be implemented in the clinic. The next step will be to repeat the experiments using a large sample size of AD patient blood samples,” Dr. Banack told this news organization.
The study team is looking to source a larger sample size of AD blood samples to replicate these findings. They are also examining this biomarker relative to other neurodegenerative diseases.
“If verified with larger sample sizes, the quantification of 2-aminoethyl dihydrogen phosphate could potentially assist in the diagnosis of early-stage Alzheimer’s disease when used in conjunction with the patient’s CDR score and other potential AD biomarkers,” Dr. Banack and colleagues say.
Caveats, cautionary notes
Commenting on the findings, Rebecca M. Edelmayer, PhD, Alzheimer’s Association senior director of scientific engagement, said the study is “interesting, though very small-scale and very preliminary.”
Dr. Edelmayer said one “major limitation” is that participants did not have their Alzheimer’s diagnosis confirmed with “gold standard biomarkers. They have been diagnosed based only on their cognitive and behavioral symptoms.”
She also cautioned that the study population is not representative – either of the general public or people living with Alzheimer’s disease.
For example, 41 out of all 50 samples are from men, “though we know women are disproportionately impacted by Alzheimer’s.”
“There is a mismatch in the age of the study groups,” Dr. Edelmayer noted. The mean age of controls in the study was 39 and the mean age of people with dementia was 71. Race or ethnicity and other demographic information is also unclear from the article.
“There is an urgent need for simple, inexpensive, noninvasive and easily available diagnostic tools for Alzheimer’s, such as a blood test. A simple blood test for Alzheimer’s would be a great advance for individuals with – and at risk for – the disease, families, doctors, and researchers,” Dr. Edelmayer said.
“Bottom line,” Dr. Edelmayer continued, “these results need to be further tested and verified in long-term, large-scale studies with diverse populations that are representative of those living with Alzheimer’s disease.”
This research was supported by the William Stamps Farish Fund and the Josephine P. & John J. Louis Foundation. Brain Chemistry Labs has applied for a patent related to this research. Dr. Edelmayer has no relevant disclosures.
A version of this article first appeared on Medscape.com.
Investigators found that plasma concentrations of 2-aminoethyl dihydrogen phosphate and taurine could distinguish adults with early-stage Alzheimer’s disease from cognitively normal adults.
“Our biomarker for early-stage Alzheimer’s disease represents new thinking and is unique from the amyloid-beta and p-tau molecules that are currently being investigated to diagnose AD,” Sandra Banack, PhD, senior scientist, Brain Chemistry Labs, Jackson, Wyoming, told this news organization.
If further studies pan out, Dr. Banack said this biomarker could “easily be transformed into a test to aid clinical evaluations for Alzheimer’s disease.”
The study was published online in PLOS ONE.
New drug target?
The researchers measured concentrations of 2-aminoethyl dihydrogen phosphate and taurine in blood plasma samples in 25 patients (21 men; mean age, 71) with a clinical diagnosis of early-stage Alzheimer’s based on a Clinical Dementia Rating (CDR) score of 0.5, suggesting very mild cognitive impairment, and 25 healthy controls (20 men; mean age, 39).
The concentration of 2-aminoethyl dihydrogen phosphate, normalized by the concentration of taurine, reliably distinguished blood samples of early-stage Alzheimer’s patients from controls in a blinded analysis.
This biomarker “could lead to new understanding of [AD] and lead to new drug candidates,” Dr. Banack told this news organization.
The researchers note that 2-aminoethyl dihydrogen phosphate plays an important role in the structure and function of cellular membranes.
Physiologic effects of increased 2-aminoethyl dihydrogen phosphate concentrations in the blood are not known. However, in one study, concentrations of this molecule were found to be significantly lower in the temporal cortex, frontal cortex, and hippocampus (40%) in patients with Alzheimer’s disease, compared with controls.
“New biomarkers take time before they can be implemented in the clinic. The next step will be to repeat the experiments using a large sample size of AD patient blood samples,” Dr. Banack told this news organization.
The study team is looking to source a larger sample size of AD blood samples to replicate these findings. They are also examining this biomarker relative to other neurodegenerative diseases.
“If verified with larger sample sizes, the quantification of 2-aminoethyl dihydrogen phosphate could potentially assist in the diagnosis of early-stage Alzheimer’s disease when used in conjunction with the patient’s CDR score and other potential AD biomarkers,” Dr. Banack and colleagues say.
Caveats, cautionary notes
Commenting on the findings, Rebecca M. Edelmayer, PhD, Alzheimer’s Association senior director of scientific engagement, said the study is “interesting, though very small-scale and very preliminary.”
Dr. Edelmayer said one “major limitation” is that participants did not have their Alzheimer’s diagnosis confirmed with “gold standard biomarkers. They have been diagnosed based only on their cognitive and behavioral symptoms.”
She also cautioned that the study population is not representative – either of the general public or people living with Alzheimer’s disease.
For example, 41 out of all 50 samples are from men, “though we know women are disproportionately impacted by Alzheimer’s.”
“There is a mismatch in the age of the study groups,” Dr. Edelmayer noted. The mean age of controls in the study was 39 and the mean age of people with dementia was 71. Race or ethnicity and other demographic information is also unclear from the article.
“There is an urgent need for simple, inexpensive, noninvasive and easily available diagnostic tools for Alzheimer’s, such as a blood test. A simple blood test for Alzheimer’s would be a great advance for individuals with – and at risk for – the disease, families, doctors, and researchers,” Dr. Edelmayer said.
“Bottom line,” Dr. Edelmayer continued, “these results need to be further tested and verified in long-term, large-scale studies with diverse populations that are representative of those living with Alzheimer’s disease.”
This research was supported by the William Stamps Farish Fund and the Josephine P. & John J. Louis Foundation. Brain Chemistry Labs has applied for a patent related to this research. Dr. Edelmayer has no relevant disclosures.
A version of this article first appeared on Medscape.com.
Investigators found that plasma concentrations of 2-aminoethyl dihydrogen phosphate and taurine could distinguish adults with early-stage Alzheimer’s disease from cognitively normal adults.
“Our biomarker for early-stage Alzheimer’s disease represents new thinking and is unique from the amyloid-beta and p-tau molecules that are currently being investigated to diagnose AD,” Sandra Banack, PhD, senior scientist, Brain Chemistry Labs, Jackson, Wyoming, told this news organization.
If further studies pan out, Dr. Banack said this biomarker could “easily be transformed into a test to aid clinical evaluations for Alzheimer’s disease.”
The study was published online in PLOS ONE.
New drug target?
The researchers measured concentrations of 2-aminoethyl dihydrogen phosphate and taurine in blood plasma samples in 25 patients (21 men; mean age, 71) with a clinical diagnosis of early-stage Alzheimer’s based on a Clinical Dementia Rating (CDR) score of 0.5, suggesting very mild cognitive impairment, and 25 healthy controls (20 men; mean age, 39).
The concentration of 2-aminoethyl dihydrogen phosphate, normalized by the concentration of taurine, reliably distinguished blood samples of early-stage Alzheimer’s patients from controls in a blinded analysis.
This biomarker “could lead to new understanding of [AD] and lead to new drug candidates,” Dr. Banack told this news organization.
The researchers note that 2-aminoethyl dihydrogen phosphate plays an important role in the structure and function of cellular membranes.
Physiologic effects of increased 2-aminoethyl dihydrogen phosphate concentrations in the blood are not known. However, in one study, concentrations of this molecule were found to be significantly lower in the temporal cortex, frontal cortex, and hippocampus (40%) in patients with Alzheimer’s disease, compared with controls.
“New biomarkers take time before they can be implemented in the clinic. The next step will be to repeat the experiments using a large sample size of AD patient blood samples,” Dr. Banack told this news organization.
The study team is looking to source a larger sample size of AD blood samples to replicate these findings. They are also examining this biomarker relative to other neurodegenerative diseases.
“If verified with larger sample sizes, the quantification of 2-aminoethyl dihydrogen phosphate could potentially assist in the diagnosis of early-stage Alzheimer’s disease when used in conjunction with the patient’s CDR score and other potential AD biomarkers,” Dr. Banack and colleagues say.
Caveats, cautionary notes
Commenting on the findings, Rebecca M. Edelmayer, PhD, Alzheimer’s Association senior director of scientific engagement, said the study is “interesting, though very small-scale and very preliminary.”
Dr. Edelmayer said one “major limitation” is that participants did not have their Alzheimer’s diagnosis confirmed with “gold standard biomarkers. They have been diagnosed based only on their cognitive and behavioral symptoms.”
She also cautioned that the study population is not representative – either of the general public or people living with Alzheimer’s disease.
For example, 41 out of all 50 samples are from men, “though we know women are disproportionately impacted by Alzheimer’s.”
“There is a mismatch in the age of the study groups,” Dr. Edelmayer noted. The mean age of controls in the study was 39 and the mean age of people with dementia was 71. Race or ethnicity and other demographic information is also unclear from the article.
“There is an urgent need for simple, inexpensive, noninvasive and easily available diagnostic tools for Alzheimer’s, such as a blood test. A simple blood test for Alzheimer’s would be a great advance for individuals with – and at risk for – the disease, families, doctors, and researchers,” Dr. Edelmayer said.
“Bottom line,” Dr. Edelmayer continued, “these results need to be further tested and verified in long-term, large-scale studies with diverse populations that are representative of those living with Alzheimer’s disease.”
This research was supported by the William Stamps Farish Fund and the Josephine P. & John J. Louis Foundation. Brain Chemistry Labs has applied for a patent related to this research. Dr. Edelmayer has no relevant disclosures.
A version of this article first appeared on Medscape.com.
Treating frail patients requires ‘precision gerontology’
An estimated 15% of Americans over age 65 years who aren’t living in institutions are considered frail – a complex geriatric syndrome that raises the odds of disability, hospitalization, the need for nursing care, and death.
But while the word frailty may conjure images of wizened and weakened men and women, the clinical picture is far less clear.
“We’ve made a lot of progress in some ways, but still a lot of work to be done in others,” George A. Kuchel, MD, CM, the chair in geriatrics and gerontology and director of the UConn Center on Aging in Farmington, Conn., said at the annual meeting of the American College of Physicians.
“You have to be very careful about generalizations,” Dr. Kuchel said. “This is very important when you are thinking about managing it.”
One of the key take-home messages, Dr. Kuchel said, “and one of the first things I learned as a geriatrics fellow, is that when you have seen one older person, all you have seen is one older person.”
The second major take-home is that frailty is multifactorial – a critical consideration when it comes to managing elderly patients.
“Unlike other conditions, there is no single medication, there is no one single thing you can do – it is really multifactorial,” he said. “What it means is to match the components to target unique needs, and that is something that we are calling ‘precision gerontology,’ as opposed to precision medicine.”
The definitions of frailty vary but can involve increased vulnerability; enhanced risk of declining function, disability, and death; and a decline in functioning across multiple physiologic systems, accompanied by an increased vulnerability to stressors.
Key features that clinicians should emphasize include multifactorial etiology with each risk factor contributing only modestly:
- Multidimensional nature, with physical and psychosocial factors playing a part.
- Frailty represents an extreme consequence of the normal aging process.
- The process is dynamic, and individuals can fluctuate between frailty states.
Diagnosing frailty
Diagnosing frailty in the average clinical setting can be a challenge. Unlike other disorders, no single test or assessment tool exists for the condition. Most settings or patients, for example, do not even have the device to measure hand grip strength, Dr. Kuchel said. Other obstacles include a lack of time and reimbursement.
However, clinicians can quickly and easily assess patients for several warning signs, including the presence of multimorbidity (>5 diseases), slow walking speed (<1 m/sec), inability to climb a flight of stairs, and/or walk a block or rise from chair five times with arms folded.
“These are simple questions that can be asked by a medical assistant or even over the phone ahead of time,” he said.
Frailty and sarcopenia are closely linked but are not equivalent. As a result, dual-energy x-ray absorptiometry (DXA), which can measure both bone mineral density and muscle mass, is not a good assessment of frailty because muscle mass by itself is not necessarily tied to weakness. Instead, Dr. Kuchel said, measuring muscle function and quality is much more effective at identifying frail patients.
“Gait velocity is potentially the greatest single measure, and if there is one thing you should do with your patient, it is to check gait velocity,” Dr. Kuchel said. Researchers at his facility are working on radio technology identification-based device that allows for measuring gait when a patient walks down the hallway.
“Measuring gait should be the sixth vital sign, and you need to have that information in front of you when working with older patients,” he said. “We are working on integrating it into our system.”
Managing frailty
Although no single intervention for frailty exists, physical activity has been shown to delay its onset. Still, Dr. Kuchel said, clinicians can try a range of approaches, both biologic and social, to address the condition.
Assessing for and treating depression, for example, may help reduce frailty fatigue, as can stopping medications – including benzodiazepines, and corticosteroids – that might be worsening the condition. Another step is to check for low vitamin D levels and hypothyroidism, he said.
Some patients have unexplained anemia that could be corrected, as well as correcting basal and orthostatic hypotension, which can arise from overtreatment, Dr. Kuchel added.
People with HIV can experience accelerated aging, as can adults who were treated with chemotherapy and radiation as children. “We are also beginning to see some of this with long COVID, so there seems to be some overlap,” he said.
Finally, socioeconomic considerations include the possibility of elder neglect and/or abuse, and the effects of poverty on nutrition and the ability to pay for needed medications.
The bottom line, Dr. Kuchel said, is that managing frailty is possible, but doing so effectively may require stops and starts.
“Correct what is correctable, such as nutrition, vitamin D, depression, and stopping offending meds,” he said. “Match multicomponent interventions with deficits and interventions targeting health care systems will include better care coordination. A comprehensive geriatric assessment is important in the care of this geriatric syndrome.
Dr. Kuchel has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An estimated 15% of Americans over age 65 years who aren’t living in institutions are considered frail – a complex geriatric syndrome that raises the odds of disability, hospitalization, the need for nursing care, and death.
But while the word frailty may conjure images of wizened and weakened men and women, the clinical picture is far less clear.
“We’ve made a lot of progress in some ways, but still a lot of work to be done in others,” George A. Kuchel, MD, CM, the chair in geriatrics and gerontology and director of the UConn Center on Aging in Farmington, Conn., said at the annual meeting of the American College of Physicians.
“You have to be very careful about generalizations,” Dr. Kuchel said. “This is very important when you are thinking about managing it.”
One of the key take-home messages, Dr. Kuchel said, “and one of the first things I learned as a geriatrics fellow, is that when you have seen one older person, all you have seen is one older person.”
The second major take-home is that frailty is multifactorial – a critical consideration when it comes to managing elderly patients.
“Unlike other conditions, there is no single medication, there is no one single thing you can do – it is really multifactorial,” he said. “What it means is to match the components to target unique needs, and that is something that we are calling ‘precision gerontology,’ as opposed to precision medicine.”
The definitions of frailty vary but can involve increased vulnerability; enhanced risk of declining function, disability, and death; and a decline in functioning across multiple physiologic systems, accompanied by an increased vulnerability to stressors.
Key features that clinicians should emphasize include multifactorial etiology with each risk factor contributing only modestly:
- Multidimensional nature, with physical and psychosocial factors playing a part.
- Frailty represents an extreme consequence of the normal aging process.
- The process is dynamic, and individuals can fluctuate between frailty states.
Diagnosing frailty
Diagnosing frailty in the average clinical setting can be a challenge. Unlike other disorders, no single test or assessment tool exists for the condition. Most settings or patients, for example, do not even have the device to measure hand grip strength, Dr. Kuchel said. Other obstacles include a lack of time and reimbursement.
However, clinicians can quickly and easily assess patients for several warning signs, including the presence of multimorbidity (>5 diseases), slow walking speed (<1 m/sec), inability to climb a flight of stairs, and/or walk a block or rise from chair five times with arms folded.
“These are simple questions that can be asked by a medical assistant or even over the phone ahead of time,” he said.
Frailty and sarcopenia are closely linked but are not equivalent. As a result, dual-energy x-ray absorptiometry (DXA), which can measure both bone mineral density and muscle mass, is not a good assessment of frailty because muscle mass by itself is not necessarily tied to weakness. Instead, Dr. Kuchel said, measuring muscle function and quality is much more effective at identifying frail patients.
“Gait velocity is potentially the greatest single measure, and if there is one thing you should do with your patient, it is to check gait velocity,” Dr. Kuchel said. Researchers at his facility are working on radio technology identification-based device that allows for measuring gait when a patient walks down the hallway.
“Measuring gait should be the sixth vital sign, and you need to have that information in front of you when working with older patients,” he said. “We are working on integrating it into our system.”
Managing frailty
Although no single intervention for frailty exists, physical activity has been shown to delay its onset. Still, Dr. Kuchel said, clinicians can try a range of approaches, both biologic and social, to address the condition.
Assessing for and treating depression, for example, may help reduce frailty fatigue, as can stopping medications – including benzodiazepines, and corticosteroids – that might be worsening the condition. Another step is to check for low vitamin D levels and hypothyroidism, he said.
Some patients have unexplained anemia that could be corrected, as well as correcting basal and orthostatic hypotension, which can arise from overtreatment, Dr. Kuchel added.
People with HIV can experience accelerated aging, as can adults who were treated with chemotherapy and radiation as children. “We are also beginning to see some of this with long COVID, so there seems to be some overlap,” he said.
Finally, socioeconomic considerations include the possibility of elder neglect and/or abuse, and the effects of poverty on nutrition and the ability to pay for needed medications.
The bottom line, Dr. Kuchel said, is that managing frailty is possible, but doing so effectively may require stops and starts.
“Correct what is correctable, such as nutrition, vitamin D, depression, and stopping offending meds,” he said. “Match multicomponent interventions with deficits and interventions targeting health care systems will include better care coordination. A comprehensive geriatric assessment is important in the care of this geriatric syndrome.
Dr. Kuchel has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An estimated 15% of Americans over age 65 years who aren’t living in institutions are considered frail – a complex geriatric syndrome that raises the odds of disability, hospitalization, the need for nursing care, and death.
But while the word frailty may conjure images of wizened and weakened men and women, the clinical picture is far less clear.
“We’ve made a lot of progress in some ways, but still a lot of work to be done in others,” George A. Kuchel, MD, CM, the chair in geriatrics and gerontology and director of the UConn Center on Aging in Farmington, Conn., said at the annual meeting of the American College of Physicians.
“You have to be very careful about generalizations,” Dr. Kuchel said. “This is very important when you are thinking about managing it.”
One of the key take-home messages, Dr. Kuchel said, “and one of the first things I learned as a geriatrics fellow, is that when you have seen one older person, all you have seen is one older person.”
The second major take-home is that frailty is multifactorial – a critical consideration when it comes to managing elderly patients.
“Unlike other conditions, there is no single medication, there is no one single thing you can do – it is really multifactorial,” he said. “What it means is to match the components to target unique needs, and that is something that we are calling ‘precision gerontology,’ as opposed to precision medicine.”
The definitions of frailty vary but can involve increased vulnerability; enhanced risk of declining function, disability, and death; and a decline in functioning across multiple physiologic systems, accompanied by an increased vulnerability to stressors.
Key features that clinicians should emphasize include multifactorial etiology with each risk factor contributing only modestly:
- Multidimensional nature, with physical and psychosocial factors playing a part.
- Frailty represents an extreme consequence of the normal aging process.
- The process is dynamic, and individuals can fluctuate between frailty states.
Diagnosing frailty
Diagnosing frailty in the average clinical setting can be a challenge. Unlike other disorders, no single test or assessment tool exists for the condition. Most settings or patients, for example, do not even have the device to measure hand grip strength, Dr. Kuchel said. Other obstacles include a lack of time and reimbursement.
However, clinicians can quickly and easily assess patients for several warning signs, including the presence of multimorbidity (>5 diseases), slow walking speed (<1 m/sec), inability to climb a flight of stairs, and/or walk a block or rise from chair five times with arms folded.
“These are simple questions that can be asked by a medical assistant or even over the phone ahead of time,” he said.
Frailty and sarcopenia are closely linked but are not equivalent. As a result, dual-energy x-ray absorptiometry (DXA), which can measure both bone mineral density and muscle mass, is not a good assessment of frailty because muscle mass by itself is not necessarily tied to weakness. Instead, Dr. Kuchel said, measuring muscle function and quality is much more effective at identifying frail patients.
“Gait velocity is potentially the greatest single measure, and if there is one thing you should do with your patient, it is to check gait velocity,” Dr. Kuchel said. Researchers at his facility are working on radio technology identification-based device that allows for measuring gait when a patient walks down the hallway.
“Measuring gait should be the sixth vital sign, and you need to have that information in front of you when working with older patients,” he said. “We are working on integrating it into our system.”
Managing frailty
Although no single intervention for frailty exists, physical activity has been shown to delay its onset. Still, Dr. Kuchel said, clinicians can try a range of approaches, both biologic and social, to address the condition.
Assessing for and treating depression, for example, may help reduce frailty fatigue, as can stopping medications – including benzodiazepines, and corticosteroids – that might be worsening the condition. Another step is to check for low vitamin D levels and hypothyroidism, he said.
Some patients have unexplained anemia that could be corrected, as well as correcting basal and orthostatic hypotension, which can arise from overtreatment, Dr. Kuchel added.
People with HIV can experience accelerated aging, as can adults who were treated with chemotherapy and radiation as children. “We are also beginning to see some of this with long COVID, so there seems to be some overlap,” he said.
Finally, socioeconomic considerations include the possibility of elder neglect and/or abuse, and the effects of poverty on nutrition and the ability to pay for needed medications.
The bottom line, Dr. Kuchel said, is that managing frailty is possible, but doing so effectively may require stops and starts.
“Correct what is correctable, such as nutrition, vitamin D, depression, and stopping offending meds,” he said. “Match multicomponent interventions with deficits and interventions targeting health care systems will include better care coordination. A comprehensive geriatric assessment is important in the care of this geriatric syndrome.
Dr. Kuchel has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM INTERNAL MEDICINE 2022
Long-COVID symptoms a serious challenge for older patients, physicians
Even mundane tasks such as making a meal can be exhausting for Louise Salant.
“I’m totally wiped out,” said the 71-year-old former private music instructor with asthma who lives in New York City and has been coping with debilitating symptoms of fatigue, shortness of breath, and gastrointestinal symptoms since recovering from a severe bout of COVID-19 2 years ago. “I just don’t have the energy.”
Ms. Salant is not alone. Many older people who contract COVID-19 experience prolonged symptoms of the disease. An analysis of Medicare Advantage claims data published in the BMJ found that about one-third of roughly 87,000 adults aged 65 in the database with a COVID-19 diagnosis sought care for persistent or new symptoms 21 or more days later.
That figure is about twice the rate of persistent COVID-19 related symptoms seen in a cohort of adults younger than age 65 with commercial insurance analyzed by the same group of researchers in a separate BMJ study. Compared with a 2020 comparator group of patients in this age cohort, these patients had a greater likelihood of respiratory failure, fatigue, hypertension, memory problems, kidney injury, mental health conditions, hypercoagulability, and cardiac rhythm disorders. When they compared post–COVID-19 symptoms to lasting symptoms of another serious viral disease – influenza – the researchers found that only respiratory failure, dementia, and post-viral fatigue were more common in the COVID-19 group.
“It became clear early in the pandemic that there is going to be a second pandemic related to all of the complications that we’ve seen related to COVID-19 infections,” said Ken Cohen, MD, executive director of translational research and national senior medical director for Optum Labs in Minnetonka, Minn., who coauthored the BMJ studies.
The results are among a growing body of evidence suggesting that older adults are at high risk of persistent post-COVID-19 symptoms.
Researchers in Rome, for example, found that 83% of 165 patients aged 65 or older who had been hospitalized for COVID-19 reported at least one lasting symptom – problems like fatigue, shortness of breath, joint pain, and coughing – in the months after hospitalization. One-third of those had two symptoms, and 46% had three or more.
A similar study in Norway found that two-thirds of patients aged 60 or older reported reduced health-related quality of life during follow-up visits 6 months after hospitalization for COVID-19. The most-reported impairments among those patients were the inability to perform the tasks of daily life, reduced mobility, and increased pain and discomfort.
Cognitive concerns
Mounting evidence indicates that COVID-19 may contribute to chronic cognitive impairment in older adults. A multisite U.S. study found that 28% of 817 adults presenting to emergency departments with COVID-19 had delirium and poorer outcomes. A Chinese case-control study that enrolled 1,438 individuals hospitalized in Wuhan for COVID-19, along with 438 of their uninfected spouses, found that 12% of COVID-19 survivors experienced cognitive impairment a year after discharge. Matteo Tosato, MD, PhD, head of the outpatient clinic for patients with long COVID symptoms at Gemelli Hospital in Rome, called those findings “very concerning.”
Jin Ho Han, MD, associate professor of emergency medicine at Vanderbilt University, Nashville, Tenn., said cognitive impairment is common after an acute illness, particularly in frail or vulnerable patients.
“Hospitalization and the acute illness itself accelerate cognitive decline,” said Dr. Han, and previous evidence links delirium with worsening cognition. He and his colleagues are studying the potential role of delirium in longer-term cognitive decline in older patients after COVID-19.
Dr. Han emphasized the importance of preventing COVID-19-related delirium through vaccines and other strategies to reduce exposure of older patients to the virus. “Once you have cognitive decline, there are no interventions to reverse it,” he said.
Alarm bells for long-term care
Experts expressed concern that the situation might be even worse for people living in long-term care facilities. Many already need assistance with tasks of daily living and could be particularly vulnerable to lasting effects of COVID-19, said Karl Steinberg, MD, president of the Society for Post-Acute and Long-Term Care Medicine. He estimated that roughly half of his patients who have had COVID-19, regardless of the severity of their symptoms, have endured some degree of functional decline.
“It’s common for long-term care facility residents to experience functional and cognitive decline, even after seemingly minor things, like a cold or a trip to the hospital,” Dr. Steinberg, who has been a medical director of long-term care facilities in San Diego County for more than 2 decades, told this news organization. “It makes it a little harder to determine whether the declines we’ve been seeing post COVID in these residents are attributable to post COVID versus just an accelerated step in their overall expected decline.”
The pandemic may have contributed to worse outcomes for people in long-term care facilities in several ways: the disease itself, its effects on health care delivery, and necessary preventive measures to protect long-term care residents from exposure to the virus.
“During the many months where family visits were prohibited, we saw people – whether they had COVID-19 or not – suffer major clinical, functional, cognitive declines or severe psychological symptoms,” Dr. Steinberg said.
He emphasized the importance of preventive measures such as vaccines and boosters in patients in long-term care facilities. He said the benefit of preventing lasting symptoms is often a strong motivator for family caregivers of people with dementia to get them vaccinated or boosted.
“It’s clear that vaccination and booster reduce the incidence of post-COVID symptoms,” he said. Almost all studies have been in younger cohorts, but he expects the benefits would also apply to older patients.
Easing symptoms and offering support
As with long COVID generally, many questions remain about the causes of lasting symptoms of COVID-19 in older patients, and how best to treat them. Dr. Tosato, who led the study of long-COVID patients in Rome, is focusing on inflammation as a critical factor in the condition. He and colleagues across Europe hope to answer some of them by launching a multicenter study of lasting COVID-19 symptoms.
In the meantime, Dr. Steinberg and Dr. Tosato said they are doing their best to evaluate and treat patients empirically.
“We pull from our armamentarium to treat system-specific symptoms,” Dr. Steinberg said. “We want to improve the quality of life and help each day be the best it can.”
Physicians in long-term care facilities might use medications such as antidepressants or nonpharmacologic approaches for patients experiencing depression symptoms. Families are also crucial in helping patients by bringing in home-cooked meals and encouraging loved ones who may be experiencing loss of taste or smell to eat, Dr. Steinberg said.
“We’ve seen with the return of families and loved ones visiting to some extent has alleviated some people’s symptoms, especially psychological ones,” he said.
Dr. Tosato said he and his colleagues start with an individualized, multidisciplinary assessment to determine what types of care may help. He noted that physicians might recommend medications or rehabilitative therapies depending on the patient’s needs.
“A personalized approach is key,” Dr. Tosato said. His study also found that the proportion of older patients experiencing symptoms declined over time – a glimmer of hope that many will recover.
Dr. Cohen emphasized the need for a multimodal rehabilitation, an evidence-based approach used to care for patients who survived hospitalization with severe COVID-19 – a group that has substantially higher rates of persistent symptoms. This approach includes cognitive rehabilitation, physical therapy, occupational therapy, and a graded exercise program.
Dr. Han and colleagues are studying potential therapies such as cognitive rehabilitation in adults who’ve experienced delirium. But until evidence-based treatments are available, they stress the role of support for patients with cognitive decline and their families.
“A lot of the work we do is teach patients and their families to compensate for newly acquired cognitive deficits from any illness, including COVID-19,” Dr. Han said.
Ms. Salant said she has experienced some improvement in her energy since her pulmonologist recommended a new inhaler based on her symptoms. Her sense of smell and taste, lost to the infection, returned after she received her first dose of a vaccine against COVID-19. She takes comfort in participating in Survivor Corps, a group of more than 170,000 COVID-19 survivors and their families who advocate for more scientific research on the disease.
She also expressed gratitude for the support she receives from her primary care physician, who she said has taken the time to learn more about the symptoms of long COVID, listens to her, and respects what she has to say.
“I have hope that I will keep getting better by baby steps,” Ms. Salant said.
Dr. Tosato, Dr. Steinberg, and Dr. Han have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Even mundane tasks such as making a meal can be exhausting for Louise Salant.
“I’m totally wiped out,” said the 71-year-old former private music instructor with asthma who lives in New York City and has been coping with debilitating symptoms of fatigue, shortness of breath, and gastrointestinal symptoms since recovering from a severe bout of COVID-19 2 years ago. “I just don’t have the energy.”
Ms. Salant is not alone. Many older people who contract COVID-19 experience prolonged symptoms of the disease. An analysis of Medicare Advantage claims data published in the BMJ found that about one-third of roughly 87,000 adults aged 65 in the database with a COVID-19 diagnosis sought care for persistent or new symptoms 21 or more days later.
That figure is about twice the rate of persistent COVID-19 related symptoms seen in a cohort of adults younger than age 65 with commercial insurance analyzed by the same group of researchers in a separate BMJ study. Compared with a 2020 comparator group of patients in this age cohort, these patients had a greater likelihood of respiratory failure, fatigue, hypertension, memory problems, kidney injury, mental health conditions, hypercoagulability, and cardiac rhythm disorders. When they compared post–COVID-19 symptoms to lasting symptoms of another serious viral disease – influenza – the researchers found that only respiratory failure, dementia, and post-viral fatigue were more common in the COVID-19 group.
“It became clear early in the pandemic that there is going to be a second pandemic related to all of the complications that we’ve seen related to COVID-19 infections,” said Ken Cohen, MD, executive director of translational research and national senior medical director for Optum Labs in Minnetonka, Minn., who coauthored the BMJ studies.
The results are among a growing body of evidence suggesting that older adults are at high risk of persistent post-COVID-19 symptoms.
Researchers in Rome, for example, found that 83% of 165 patients aged 65 or older who had been hospitalized for COVID-19 reported at least one lasting symptom – problems like fatigue, shortness of breath, joint pain, and coughing – in the months after hospitalization. One-third of those had two symptoms, and 46% had three or more.
A similar study in Norway found that two-thirds of patients aged 60 or older reported reduced health-related quality of life during follow-up visits 6 months after hospitalization for COVID-19. The most-reported impairments among those patients were the inability to perform the tasks of daily life, reduced mobility, and increased pain and discomfort.
Cognitive concerns
Mounting evidence indicates that COVID-19 may contribute to chronic cognitive impairment in older adults. A multisite U.S. study found that 28% of 817 adults presenting to emergency departments with COVID-19 had delirium and poorer outcomes. A Chinese case-control study that enrolled 1,438 individuals hospitalized in Wuhan for COVID-19, along with 438 of their uninfected spouses, found that 12% of COVID-19 survivors experienced cognitive impairment a year after discharge. Matteo Tosato, MD, PhD, head of the outpatient clinic for patients with long COVID symptoms at Gemelli Hospital in Rome, called those findings “very concerning.”
Jin Ho Han, MD, associate professor of emergency medicine at Vanderbilt University, Nashville, Tenn., said cognitive impairment is common after an acute illness, particularly in frail or vulnerable patients.
“Hospitalization and the acute illness itself accelerate cognitive decline,” said Dr. Han, and previous evidence links delirium with worsening cognition. He and his colleagues are studying the potential role of delirium in longer-term cognitive decline in older patients after COVID-19.
Dr. Han emphasized the importance of preventing COVID-19-related delirium through vaccines and other strategies to reduce exposure of older patients to the virus. “Once you have cognitive decline, there are no interventions to reverse it,” he said.
Alarm bells for long-term care
Experts expressed concern that the situation might be even worse for people living in long-term care facilities. Many already need assistance with tasks of daily living and could be particularly vulnerable to lasting effects of COVID-19, said Karl Steinberg, MD, president of the Society for Post-Acute and Long-Term Care Medicine. He estimated that roughly half of his patients who have had COVID-19, regardless of the severity of their symptoms, have endured some degree of functional decline.
“It’s common for long-term care facility residents to experience functional and cognitive decline, even after seemingly minor things, like a cold or a trip to the hospital,” Dr. Steinberg, who has been a medical director of long-term care facilities in San Diego County for more than 2 decades, told this news organization. “It makes it a little harder to determine whether the declines we’ve been seeing post COVID in these residents are attributable to post COVID versus just an accelerated step in their overall expected decline.”
The pandemic may have contributed to worse outcomes for people in long-term care facilities in several ways: the disease itself, its effects on health care delivery, and necessary preventive measures to protect long-term care residents from exposure to the virus.
“During the many months where family visits were prohibited, we saw people – whether they had COVID-19 or not – suffer major clinical, functional, cognitive declines or severe psychological symptoms,” Dr. Steinberg said.
He emphasized the importance of preventive measures such as vaccines and boosters in patients in long-term care facilities. He said the benefit of preventing lasting symptoms is often a strong motivator for family caregivers of people with dementia to get them vaccinated or boosted.
“It’s clear that vaccination and booster reduce the incidence of post-COVID symptoms,” he said. Almost all studies have been in younger cohorts, but he expects the benefits would also apply to older patients.
Easing symptoms and offering support
As with long COVID generally, many questions remain about the causes of lasting symptoms of COVID-19 in older patients, and how best to treat them. Dr. Tosato, who led the study of long-COVID patients in Rome, is focusing on inflammation as a critical factor in the condition. He and colleagues across Europe hope to answer some of them by launching a multicenter study of lasting COVID-19 symptoms.
In the meantime, Dr. Steinberg and Dr. Tosato said they are doing their best to evaluate and treat patients empirically.
“We pull from our armamentarium to treat system-specific symptoms,” Dr. Steinberg said. “We want to improve the quality of life and help each day be the best it can.”
Physicians in long-term care facilities might use medications such as antidepressants or nonpharmacologic approaches for patients experiencing depression symptoms. Families are also crucial in helping patients by bringing in home-cooked meals and encouraging loved ones who may be experiencing loss of taste or smell to eat, Dr. Steinberg said.
“We’ve seen with the return of families and loved ones visiting to some extent has alleviated some people’s symptoms, especially psychological ones,” he said.
Dr. Tosato said he and his colleagues start with an individualized, multidisciplinary assessment to determine what types of care may help. He noted that physicians might recommend medications or rehabilitative therapies depending on the patient’s needs.
“A personalized approach is key,” Dr. Tosato said. His study also found that the proportion of older patients experiencing symptoms declined over time – a glimmer of hope that many will recover.
Dr. Cohen emphasized the need for a multimodal rehabilitation, an evidence-based approach used to care for patients who survived hospitalization with severe COVID-19 – a group that has substantially higher rates of persistent symptoms. This approach includes cognitive rehabilitation, physical therapy, occupational therapy, and a graded exercise program.
Dr. Han and colleagues are studying potential therapies such as cognitive rehabilitation in adults who’ve experienced delirium. But until evidence-based treatments are available, they stress the role of support for patients with cognitive decline and their families.
“A lot of the work we do is teach patients and their families to compensate for newly acquired cognitive deficits from any illness, including COVID-19,” Dr. Han said.
Ms. Salant said she has experienced some improvement in her energy since her pulmonologist recommended a new inhaler based on her symptoms. Her sense of smell and taste, lost to the infection, returned after she received her first dose of a vaccine against COVID-19. She takes comfort in participating in Survivor Corps, a group of more than 170,000 COVID-19 survivors and their families who advocate for more scientific research on the disease.
She also expressed gratitude for the support she receives from her primary care physician, who she said has taken the time to learn more about the symptoms of long COVID, listens to her, and respects what she has to say.
“I have hope that I will keep getting better by baby steps,” Ms. Salant said.
Dr. Tosato, Dr. Steinberg, and Dr. Han have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Even mundane tasks such as making a meal can be exhausting for Louise Salant.
“I’m totally wiped out,” said the 71-year-old former private music instructor with asthma who lives in New York City and has been coping with debilitating symptoms of fatigue, shortness of breath, and gastrointestinal symptoms since recovering from a severe bout of COVID-19 2 years ago. “I just don’t have the energy.”
Ms. Salant is not alone. Many older people who contract COVID-19 experience prolonged symptoms of the disease. An analysis of Medicare Advantage claims data published in the BMJ found that about one-third of roughly 87,000 adults aged 65 in the database with a COVID-19 diagnosis sought care for persistent or new symptoms 21 or more days later.
That figure is about twice the rate of persistent COVID-19 related symptoms seen in a cohort of adults younger than age 65 with commercial insurance analyzed by the same group of researchers in a separate BMJ study. Compared with a 2020 comparator group of patients in this age cohort, these patients had a greater likelihood of respiratory failure, fatigue, hypertension, memory problems, kidney injury, mental health conditions, hypercoagulability, and cardiac rhythm disorders. When they compared post–COVID-19 symptoms to lasting symptoms of another serious viral disease – influenza – the researchers found that only respiratory failure, dementia, and post-viral fatigue were more common in the COVID-19 group.
“It became clear early in the pandemic that there is going to be a second pandemic related to all of the complications that we’ve seen related to COVID-19 infections,” said Ken Cohen, MD, executive director of translational research and national senior medical director for Optum Labs in Minnetonka, Minn., who coauthored the BMJ studies.
The results are among a growing body of evidence suggesting that older adults are at high risk of persistent post-COVID-19 symptoms.
Researchers in Rome, for example, found that 83% of 165 patients aged 65 or older who had been hospitalized for COVID-19 reported at least one lasting symptom – problems like fatigue, shortness of breath, joint pain, and coughing – in the months after hospitalization. One-third of those had two symptoms, and 46% had three or more.
A similar study in Norway found that two-thirds of patients aged 60 or older reported reduced health-related quality of life during follow-up visits 6 months after hospitalization for COVID-19. The most-reported impairments among those patients were the inability to perform the tasks of daily life, reduced mobility, and increased pain and discomfort.
Cognitive concerns
Mounting evidence indicates that COVID-19 may contribute to chronic cognitive impairment in older adults. A multisite U.S. study found that 28% of 817 adults presenting to emergency departments with COVID-19 had delirium and poorer outcomes. A Chinese case-control study that enrolled 1,438 individuals hospitalized in Wuhan for COVID-19, along with 438 of their uninfected spouses, found that 12% of COVID-19 survivors experienced cognitive impairment a year after discharge. Matteo Tosato, MD, PhD, head of the outpatient clinic for patients with long COVID symptoms at Gemelli Hospital in Rome, called those findings “very concerning.”
Jin Ho Han, MD, associate professor of emergency medicine at Vanderbilt University, Nashville, Tenn., said cognitive impairment is common after an acute illness, particularly in frail or vulnerable patients.
“Hospitalization and the acute illness itself accelerate cognitive decline,” said Dr. Han, and previous evidence links delirium with worsening cognition. He and his colleagues are studying the potential role of delirium in longer-term cognitive decline in older patients after COVID-19.
Dr. Han emphasized the importance of preventing COVID-19-related delirium through vaccines and other strategies to reduce exposure of older patients to the virus. “Once you have cognitive decline, there are no interventions to reverse it,” he said.
Alarm bells for long-term care
Experts expressed concern that the situation might be even worse for people living in long-term care facilities. Many already need assistance with tasks of daily living and could be particularly vulnerable to lasting effects of COVID-19, said Karl Steinberg, MD, president of the Society for Post-Acute and Long-Term Care Medicine. He estimated that roughly half of his patients who have had COVID-19, regardless of the severity of their symptoms, have endured some degree of functional decline.
“It’s common for long-term care facility residents to experience functional and cognitive decline, even after seemingly minor things, like a cold or a trip to the hospital,” Dr. Steinberg, who has been a medical director of long-term care facilities in San Diego County for more than 2 decades, told this news organization. “It makes it a little harder to determine whether the declines we’ve been seeing post COVID in these residents are attributable to post COVID versus just an accelerated step in their overall expected decline.”
The pandemic may have contributed to worse outcomes for people in long-term care facilities in several ways: the disease itself, its effects on health care delivery, and necessary preventive measures to protect long-term care residents from exposure to the virus.
“During the many months where family visits were prohibited, we saw people – whether they had COVID-19 or not – suffer major clinical, functional, cognitive declines or severe psychological symptoms,” Dr. Steinberg said.
He emphasized the importance of preventive measures such as vaccines and boosters in patients in long-term care facilities. He said the benefit of preventing lasting symptoms is often a strong motivator for family caregivers of people with dementia to get them vaccinated or boosted.
“It’s clear that vaccination and booster reduce the incidence of post-COVID symptoms,” he said. Almost all studies have been in younger cohorts, but he expects the benefits would also apply to older patients.
Easing symptoms and offering support
As with long COVID generally, many questions remain about the causes of lasting symptoms of COVID-19 in older patients, and how best to treat them. Dr. Tosato, who led the study of long-COVID patients in Rome, is focusing on inflammation as a critical factor in the condition. He and colleagues across Europe hope to answer some of them by launching a multicenter study of lasting COVID-19 symptoms.
In the meantime, Dr. Steinberg and Dr. Tosato said they are doing their best to evaluate and treat patients empirically.
“We pull from our armamentarium to treat system-specific symptoms,” Dr. Steinberg said. “We want to improve the quality of life and help each day be the best it can.”
Physicians in long-term care facilities might use medications such as antidepressants or nonpharmacologic approaches for patients experiencing depression symptoms. Families are also crucial in helping patients by bringing in home-cooked meals and encouraging loved ones who may be experiencing loss of taste or smell to eat, Dr. Steinberg said.
“We’ve seen with the return of families and loved ones visiting to some extent has alleviated some people’s symptoms, especially psychological ones,” he said.
Dr. Tosato said he and his colleagues start with an individualized, multidisciplinary assessment to determine what types of care may help. He noted that physicians might recommend medications or rehabilitative therapies depending on the patient’s needs.
“A personalized approach is key,” Dr. Tosato said. His study also found that the proportion of older patients experiencing symptoms declined over time – a glimmer of hope that many will recover.
Dr. Cohen emphasized the need for a multimodal rehabilitation, an evidence-based approach used to care for patients who survived hospitalization with severe COVID-19 – a group that has substantially higher rates of persistent symptoms. This approach includes cognitive rehabilitation, physical therapy, occupational therapy, and a graded exercise program.
Dr. Han and colleagues are studying potential therapies such as cognitive rehabilitation in adults who’ve experienced delirium. But until evidence-based treatments are available, they stress the role of support for patients with cognitive decline and their families.
“A lot of the work we do is teach patients and their families to compensate for newly acquired cognitive deficits from any illness, including COVID-19,” Dr. Han said.
Ms. Salant said she has experienced some improvement in her energy since her pulmonologist recommended a new inhaler based on her symptoms. Her sense of smell and taste, lost to the infection, returned after she received her first dose of a vaccine against COVID-19. She takes comfort in participating in Survivor Corps, a group of more than 170,000 COVID-19 survivors and their families who advocate for more scientific research on the disease.
She also expressed gratitude for the support she receives from her primary care physician, who she said has taken the time to learn more about the symptoms of long COVID, listens to her, and respects what she has to say.
“I have hope that I will keep getting better by baby steps,” Ms. Salant said.
Dr. Tosato, Dr. Steinberg, and Dr. Han have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Virtual reality an ‘exciting opportunity’ for geriatric psychiatry
Researchers are increasingly turning their attention to virtual reality (VR) for the treatment of psychiatric disorders in older adults.
Recent studies have highlighted the usefulness of VR in treating depression and loneliness in older patients who may be socially isolated because of their age, comorbidities, or the COVID-19 pandemic.
“The unique capability of virtual reality to create an immersive and engaging setting is an exciting opportunity for geriatric psychiatry,” Harmehr Sekhon, PhD, postdoctoral research fellow, Lady Davis Institute/Jewish General Hospital, McGill University, Montreal, and McLean Hospital, Harvard Medical School, Boston, told this news organization.
, Dr. Sekhon said.
One novel approach involves using VR to administer a mindfulness intervention in older adults. Dr. Sekhon shared information on her own mindfulness study and on other developments in VR and telemedicine at the American Association for Geriatric Psychiatry annual meeting.
Potential bridging tool
As the population ages, the prevalence of mental health disorders increases. Telemedicine has proved to be a potential “bridge” to address the health care needs of older adults, Dr. Sekhon noted.
She cited her systematic review of telemedicine for older adults with dementia during COVID-19. Results showed that telemedicine was a “beneficial approach” to assisting these individuals and that it increased accessibility, said Dr. Sekhon.
In addition, a survey published last year showed that 87% of Americans in general want to continue using telehealth services after the pandemic. Most respondents agreed that telehealth had made it easier to get the care they needed. They also reported having received the same level of care via telehealth as with in-person care.
A growing body of research shows that VR has “positive influences on mood and well-being, cognition, pain management, [and] treatment of phobias in younger adults,” Dr. Sekhon said. She added that there is evidence that VR is feasible for older adults, with applications in cognitive disorders.
She cited a recent systematic review of 55 studies that assessed the impact of different types of VR on mental health in older adults. The results showed that VR could be helpful in screening for cognitive impairment – and it was comparable to some paper-based assessment. It was also useful as a training tool for those with cognitive impairment.
Examples of VR interventions that can be used to treat cognitive impairment include “virtual cities, kitchens, supermarkets,” Dr. Sekhon noted.
The technology is increasingly being used as a tool to deliver psychotherapy, in which patient engagement is “a key determinant” of outcomes, she added. “Virtual reality is a cutting-edge, engaging, and immersive technique to administer psychotherapy,” she said.
Such VR approaches are proving successful in older patients. Dr. Sekhon highlighted the case of an 85-year-old woman who engaged in ten sessions of psychodynamic psychotherapy that targeted persistent dysthymia and negativistic mood. The case was part of a proof-of-concept study published in the May issue of the American Journal of Geriatric Psychiatry.
Dr. Sekhon noted the intervention was well tolerated and was associated with minimal side effects.
VR-based meditation
Dr. Sekhon and her colleagues are now conducting a randomized controlled trial of VR meditation in older adults. VR-based meditation has been shown to increase relaxation and to decrease anxiety, sadness, and anger in younger adults. However, it has not been studied in the geriatric population.
The pilot study is assessing the feasibility and tolerability of VR meditation for older adults and its effects on stress, anxiety, depression, sleep, and quality of life. The study involves 30 adults aged 60 years and older.
Participants receive either 15-minute VR mindfulness meditation sessions twice a week for 4 weeks or are on a control wait list. The meditation sessions are user friendly and focus on breath meditation and body scans, Dr. Sekhon reported.
Because participants are older and balance is a concern, safety steps are incorporated into the sessions. “We ensure they’re doing this in a seated position, in a chair with arm rests, so that they’re very stable and there’s no risk of falls,” said Dr. Sekhon.
Another concern with VR is motion sickness, she noted. “It’s pretty minimal, but the best way we found so far is giving older adults time to adapt and feel comfortable with the VR,” she said. From the first session, participants learn how to put on the device and are checked to make sure they are comfortable with the process. To help them get used to everything, video and audio are not included during the first session.
Dr. Sekhon noted that results from the study are expected later this year.
In addition to mindfulness, researchers are using VR to deliver other established interventions, such as exposure therapy – and are implementing these approaches in varied environments, including long-term and palliative care settings.
VR-related technology is constantly improving and is becoming easier to use and more affordable, said Dr. Sekhon. She noted that the simplest devices that rely on smartphones cost as little as $15.
Although VR in older adults is promising, there are barriers to its adoption and use in research, she noted. For example, older adults may have cognitive, visual, or hearing impairments. They may have limited digital literacy, and/or they may not have access to the required technology.
These barriers can be overcome through workarounds, including providing instructional videos and digital literacy assistance via Zoom and working with community partners to facilitate study recruitment of older patients, Dr. Sekhon said.
Dr. Sekhon’s research is funded by the Canadian Institutes of Health Research and the Fonds de recherche du Quebec Sante.
A version of this article first appeared on Medscape.com.
Researchers are increasingly turning their attention to virtual reality (VR) for the treatment of psychiatric disorders in older adults.
Recent studies have highlighted the usefulness of VR in treating depression and loneliness in older patients who may be socially isolated because of their age, comorbidities, or the COVID-19 pandemic.
“The unique capability of virtual reality to create an immersive and engaging setting is an exciting opportunity for geriatric psychiatry,” Harmehr Sekhon, PhD, postdoctoral research fellow, Lady Davis Institute/Jewish General Hospital, McGill University, Montreal, and McLean Hospital, Harvard Medical School, Boston, told this news organization.
, Dr. Sekhon said.
One novel approach involves using VR to administer a mindfulness intervention in older adults. Dr. Sekhon shared information on her own mindfulness study and on other developments in VR and telemedicine at the American Association for Geriatric Psychiatry annual meeting.
Potential bridging tool
As the population ages, the prevalence of mental health disorders increases. Telemedicine has proved to be a potential “bridge” to address the health care needs of older adults, Dr. Sekhon noted.
She cited her systematic review of telemedicine for older adults with dementia during COVID-19. Results showed that telemedicine was a “beneficial approach” to assisting these individuals and that it increased accessibility, said Dr. Sekhon.
In addition, a survey published last year showed that 87% of Americans in general want to continue using telehealth services after the pandemic. Most respondents agreed that telehealth had made it easier to get the care they needed. They also reported having received the same level of care via telehealth as with in-person care.
A growing body of research shows that VR has “positive influences on mood and well-being, cognition, pain management, [and] treatment of phobias in younger adults,” Dr. Sekhon said. She added that there is evidence that VR is feasible for older adults, with applications in cognitive disorders.
She cited a recent systematic review of 55 studies that assessed the impact of different types of VR on mental health in older adults. The results showed that VR could be helpful in screening for cognitive impairment – and it was comparable to some paper-based assessment. It was also useful as a training tool for those with cognitive impairment.
Examples of VR interventions that can be used to treat cognitive impairment include “virtual cities, kitchens, supermarkets,” Dr. Sekhon noted.
The technology is increasingly being used as a tool to deliver psychotherapy, in which patient engagement is “a key determinant” of outcomes, she added. “Virtual reality is a cutting-edge, engaging, and immersive technique to administer psychotherapy,” she said.
Such VR approaches are proving successful in older patients. Dr. Sekhon highlighted the case of an 85-year-old woman who engaged in ten sessions of psychodynamic psychotherapy that targeted persistent dysthymia and negativistic mood. The case was part of a proof-of-concept study published in the May issue of the American Journal of Geriatric Psychiatry.
Dr. Sekhon noted the intervention was well tolerated and was associated with minimal side effects.
VR-based meditation
Dr. Sekhon and her colleagues are now conducting a randomized controlled trial of VR meditation in older adults. VR-based meditation has been shown to increase relaxation and to decrease anxiety, sadness, and anger in younger adults. However, it has not been studied in the geriatric population.
The pilot study is assessing the feasibility and tolerability of VR meditation for older adults and its effects on stress, anxiety, depression, sleep, and quality of life. The study involves 30 adults aged 60 years and older.
Participants receive either 15-minute VR mindfulness meditation sessions twice a week for 4 weeks or are on a control wait list. The meditation sessions are user friendly and focus on breath meditation and body scans, Dr. Sekhon reported.
Because participants are older and balance is a concern, safety steps are incorporated into the sessions. “We ensure they’re doing this in a seated position, in a chair with arm rests, so that they’re very stable and there’s no risk of falls,” said Dr. Sekhon.
Another concern with VR is motion sickness, she noted. “It’s pretty minimal, but the best way we found so far is giving older adults time to adapt and feel comfortable with the VR,” she said. From the first session, participants learn how to put on the device and are checked to make sure they are comfortable with the process. To help them get used to everything, video and audio are not included during the first session.
Dr. Sekhon noted that results from the study are expected later this year.
In addition to mindfulness, researchers are using VR to deliver other established interventions, such as exposure therapy – and are implementing these approaches in varied environments, including long-term and palliative care settings.
VR-related technology is constantly improving and is becoming easier to use and more affordable, said Dr. Sekhon. She noted that the simplest devices that rely on smartphones cost as little as $15.
Although VR in older adults is promising, there are barriers to its adoption and use in research, she noted. For example, older adults may have cognitive, visual, or hearing impairments. They may have limited digital literacy, and/or they may not have access to the required technology.
These barriers can be overcome through workarounds, including providing instructional videos and digital literacy assistance via Zoom and working with community partners to facilitate study recruitment of older patients, Dr. Sekhon said.
Dr. Sekhon’s research is funded by the Canadian Institutes of Health Research and the Fonds de recherche du Quebec Sante.
A version of this article first appeared on Medscape.com.
Researchers are increasingly turning their attention to virtual reality (VR) for the treatment of psychiatric disorders in older adults.
Recent studies have highlighted the usefulness of VR in treating depression and loneliness in older patients who may be socially isolated because of their age, comorbidities, or the COVID-19 pandemic.
“The unique capability of virtual reality to create an immersive and engaging setting is an exciting opportunity for geriatric psychiatry,” Harmehr Sekhon, PhD, postdoctoral research fellow, Lady Davis Institute/Jewish General Hospital, McGill University, Montreal, and McLean Hospital, Harvard Medical School, Boston, told this news organization.
, Dr. Sekhon said.
One novel approach involves using VR to administer a mindfulness intervention in older adults. Dr. Sekhon shared information on her own mindfulness study and on other developments in VR and telemedicine at the American Association for Geriatric Psychiatry annual meeting.
Potential bridging tool
As the population ages, the prevalence of mental health disorders increases. Telemedicine has proved to be a potential “bridge” to address the health care needs of older adults, Dr. Sekhon noted.
She cited her systematic review of telemedicine for older adults with dementia during COVID-19. Results showed that telemedicine was a “beneficial approach” to assisting these individuals and that it increased accessibility, said Dr. Sekhon.
In addition, a survey published last year showed that 87% of Americans in general want to continue using telehealth services after the pandemic. Most respondents agreed that telehealth had made it easier to get the care they needed. They also reported having received the same level of care via telehealth as with in-person care.
A growing body of research shows that VR has “positive influences on mood and well-being, cognition, pain management, [and] treatment of phobias in younger adults,” Dr. Sekhon said. She added that there is evidence that VR is feasible for older adults, with applications in cognitive disorders.
She cited a recent systematic review of 55 studies that assessed the impact of different types of VR on mental health in older adults. The results showed that VR could be helpful in screening for cognitive impairment – and it was comparable to some paper-based assessment. It was also useful as a training tool for those with cognitive impairment.
Examples of VR interventions that can be used to treat cognitive impairment include “virtual cities, kitchens, supermarkets,” Dr. Sekhon noted.
The technology is increasingly being used as a tool to deliver psychotherapy, in which patient engagement is “a key determinant” of outcomes, she added. “Virtual reality is a cutting-edge, engaging, and immersive technique to administer psychotherapy,” she said.
Such VR approaches are proving successful in older patients. Dr. Sekhon highlighted the case of an 85-year-old woman who engaged in ten sessions of psychodynamic psychotherapy that targeted persistent dysthymia and negativistic mood. The case was part of a proof-of-concept study published in the May issue of the American Journal of Geriatric Psychiatry.
Dr. Sekhon noted the intervention was well tolerated and was associated with minimal side effects.
VR-based meditation
Dr. Sekhon and her colleagues are now conducting a randomized controlled trial of VR meditation in older adults. VR-based meditation has been shown to increase relaxation and to decrease anxiety, sadness, and anger in younger adults. However, it has not been studied in the geriatric population.
The pilot study is assessing the feasibility and tolerability of VR meditation for older adults and its effects on stress, anxiety, depression, sleep, and quality of life. The study involves 30 adults aged 60 years and older.
Participants receive either 15-minute VR mindfulness meditation sessions twice a week for 4 weeks or are on a control wait list. The meditation sessions are user friendly and focus on breath meditation and body scans, Dr. Sekhon reported.
Because participants are older and balance is a concern, safety steps are incorporated into the sessions. “We ensure they’re doing this in a seated position, in a chair with arm rests, so that they’re very stable and there’s no risk of falls,” said Dr. Sekhon.
Another concern with VR is motion sickness, she noted. “It’s pretty minimal, but the best way we found so far is giving older adults time to adapt and feel comfortable with the VR,” she said. From the first session, participants learn how to put on the device and are checked to make sure they are comfortable with the process. To help them get used to everything, video and audio are not included during the first session.
Dr. Sekhon noted that results from the study are expected later this year.
In addition to mindfulness, researchers are using VR to deliver other established interventions, such as exposure therapy – and are implementing these approaches in varied environments, including long-term and palliative care settings.
VR-related technology is constantly improving and is becoming easier to use and more affordable, said Dr. Sekhon. She noted that the simplest devices that rely on smartphones cost as little as $15.
Although VR in older adults is promising, there are barriers to its adoption and use in research, she noted. For example, older adults may have cognitive, visual, or hearing impairments. They may have limited digital literacy, and/or they may not have access to the required technology.
These barriers can be overcome through workarounds, including providing instructional videos and digital literacy assistance via Zoom and working with community partners to facilitate study recruitment of older patients, Dr. Sekhon said.
Dr. Sekhon’s research is funded by the Canadian Institutes of Health Research and the Fonds de recherche du Quebec Sante.
A version of this article first appeared on Medscape.com.
FROM AAGP 2022
Atypical anxiety offers intervention target in Parkinson’s disease
Anxiety is common in Parkinson’s disease (PD) and has been shown to increase functional disability and decrease quality of life, but atypical presentations of anxiety are underrecognized and often undertreated in PD patients, wrote Nadeeka N. Dissanayaka, PhD, of the University of Queensland, Brisbane, Australia, and colleagues.
In a study published in the American Journal of Geriatric Psychiatry , the researchers conducted a systematic review of 60 studies to better characterize atypical PD-related anxiety. Fourteen studies involved Anxiety Not Otherwise Specified (NOS), 31 included fluctuating anxiety symptoms, and 22 included Fear of Falling (FOF).
Overall, the average prevalence rate for anxiety disorders in the PD population was 31%.
Anxiety NOS, fluctuating anxiety, and FOF accounted for a weighted mean prevalence of 14.9%, 34.19%, and 51.5%, respectively.
The symptomatology of anxiety NOS included psychological distress about the PD diagnosis, insecurity about the future, fear of losing control of motor and bodily functions, and social embarrassment. Clinically, anxiety NOS was associated with a range of factors including minor depression, on-off motor symptoms, muscle cramps, poor quality of life, and gait impairment.
The symptomatology of fluctuating anxiety was assessed in 9 studies of the “on” motor state and 16 studies of both “on” and “off.” Symptoms associated with the off state included panic attacks, feeling anxious or sad, and avoiding situations, as well as palpitations, dizziness, chills, and hot flashes.
Clinically, studies showed that anxiety was more severe in the off-medication state, and symptoms were reduced in the on state. Data from some studies showed that fluctuating anxiety was more common in PD patients who were female, and who had a younger age of PD onset and longer disease duration.
The symptomatology of FOF included associations between FOF and difficulty with walking and gait: Using a walker or other device, more frequent freezing in place, hesitation when turning, and slower speed while walking. Clinically, characteristics associated with FOF included older age, needing assistance for activities of daily living, a history of falls, and reduced quality of life.
The results of the review were limited by several factors including the varying assessment techniques, and the lack of data on treatment for atypical anxiety in PD, the researchers noted. “To our knowledge there are no treatment trials focused on Anxiety NOS,” and studies on the treatment of fluctuating anxiety and FOF are preliminary, they said.
However, the results support the need for early identification and classification of PD-related anxiety to improve treatment strategies and long-term outcomes, the researchers concluded. In the absence of evidence-based treatment strategies, “Given the heterogeneity of anxiety presentations in PD, the importance of tailoring interventions to meet the specific needs and unique symptom profiles of each individual cannot be overstated,” and routine screening of PD patients for anxiety every 6-12 months is recommended, they emphasized.
Dr. Dissanayaka disclosed support from the National Health and Medical Research Boosting Dementia Research Leadership Fellowship.
Anxiety is common in Parkinson’s disease (PD) and has been shown to increase functional disability and decrease quality of life, but atypical presentations of anxiety are underrecognized and often undertreated in PD patients, wrote Nadeeka N. Dissanayaka, PhD, of the University of Queensland, Brisbane, Australia, and colleagues.
In a study published in the American Journal of Geriatric Psychiatry , the researchers conducted a systematic review of 60 studies to better characterize atypical PD-related anxiety. Fourteen studies involved Anxiety Not Otherwise Specified (NOS), 31 included fluctuating anxiety symptoms, and 22 included Fear of Falling (FOF).
Overall, the average prevalence rate for anxiety disorders in the PD population was 31%.
Anxiety NOS, fluctuating anxiety, and FOF accounted for a weighted mean prevalence of 14.9%, 34.19%, and 51.5%, respectively.
The symptomatology of anxiety NOS included psychological distress about the PD diagnosis, insecurity about the future, fear of losing control of motor and bodily functions, and social embarrassment. Clinically, anxiety NOS was associated with a range of factors including minor depression, on-off motor symptoms, muscle cramps, poor quality of life, and gait impairment.
The symptomatology of fluctuating anxiety was assessed in 9 studies of the “on” motor state and 16 studies of both “on” and “off.” Symptoms associated with the off state included panic attacks, feeling anxious or sad, and avoiding situations, as well as palpitations, dizziness, chills, and hot flashes.
Clinically, studies showed that anxiety was more severe in the off-medication state, and symptoms were reduced in the on state. Data from some studies showed that fluctuating anxiety was more common in PD patients who were female, and who had a younger age of PD onset and longer disease duration.
The symptomatology of FOF included associations between FOF and difficulty with walking and gait: Using a walker or other device, more frequent freezing in place, hesitation when turning, and slower speed while walking. Clinically, characteristics associated with FOF included older age, needing assistance for activities of daily living, a history of falls, and reduced quality of life.
The results of the review were limited by several factors including the varying assessment techniques, and the lack of data on treatment for atypical anxiety in PD, the researchers noted. “To our knowledge there are no treatment trials focused on Anxiety NOS,” and studies on the treatment of fluctuating anxiety and FOF are preliminary, they said.
However, the results support the need for early identification and classification of PD-related anxiety to improve treatment strategies and long-term outcomes, the researchers concluded. In the absence of evidence-based treatment strategies, “Given the heterogeneity of anxiety presentations in PD, the importance of tailoring interventions to meet the specific needs and unique symptom profiles of each individual cannot be overstated,” and routine screening of PD patients for anxiety every 6-12 months is recommended, they emphasized.
Dr. Dissanayaka disclosed support from the National Health and Medical Research Boosting Dementia Research Leadership Fellowship.
Anxiety is common in Parkinson’s disease (PD) and has been shown to increase functional disability and decrease quality of life, but atypical presentations of anxiety are underrecognized and often undertreated in PD patients, wrote Nadeeka N. Dissanayaka, PhD, of the University of Queensland, Brisbane, Australia, and colleagues.
In a study published in the American Journal of Geriatric Psychiatry , the researchers conducted a systematic review of 60 studies to better characterize atypical PD-related anxiety. Fourteen studies involved Anxiety Not Otherwise Specified (NOS), 31 included fluctuating anxiety symptoms, and 22 included Fear of Falling (FOF).
Overall, the average prevalence rate for anxiety disorders in the PD population was 31%.
Anxiety NOS, fluctuating anxiety, and FOF accounted for a weighted mean prevalence of 14.9%, 34.19%, and 51.5%, respectively.
The symptomatology of anxiety NOS included psychological distress about the PD diagnosis, insecurity about the future, fear of losing control of motor and bodily functions, and social embarrassment. Clinically, anxiety NOS was associated with a range of factors including minor depression, on-off motor symptoms, muscle cramps, poor quality of life, and gait impairment.
The symptomatology of fluctuating anxiety was assessed in 9 studies of the “on” motor state and 16 studies of both “on” and “off.” Symptoms associated with the off state included panic attacks, feeling anxious or sad, and avoiding situations, as well as palpitations, dizziness, chills, and hot flashes.
Clinically, studies showed that anxiety was more severe in the off-medication state, and symptoms were reduced in the on state. Data from some studies showed that fluctuating anxiety was more common in PD patients who were female, and who had a younger age of PD onset and longer disease duration.
The symptomatology of FOF included associations between FOF and difficulty with walking and gait: Using a walker or other device, more frequent freezing in place, hesitation when turning, and slower speed while walking. Clinically, characteristics associated with FOF included older age, needing assistance for activities of daily living, a history of falls, and reduced quality of life.
The results of the review were limited by several factors including the varying assessment techniques, and the lack of data on treatment for atypical anxiety in PD, the researchers noted. “To our knowledge there are no treatment trials focused on Anxiety NOS,” and studies on the treatment of fluctuating anxiety and FOF are preliminary, they said.
However, the results support the need for early identification and classification of PD-related anxiety to improve treatment strategies and long-term outcomes, the researchers concluded. In the absence of evidence-based treatment strategies, “Given the heterogeneity of anxiety presentations in PD, the importance of tailoring interventions to meet the specific needs and unique symptom profiles of each individual cannot be overstated,” and routine screening of PD patients for anxiety every 6-12 months is recommended, they emphasized.
Dr. Dissanayaka disclosed support from the National Health and Medical Research Boosting Dementia Research Leadership Fellowship.
FROM THE AMERICAN JOURNAL OF GERIATRIC PSYCHIATRY