User login
Better pembrolizumab outcomes with increasing PD-L1 in NSCLC
CHICAGO – The PD-1 inhibitor, pembrolizumab, is superior to docetaxel in prolonging overall and progression-free survival in patients with non–small cell lung cancer (NSCLC), according to the results of the phase III KEYNOTE-010 trial.
Furthermore, increasing PD-L1 expression was associated with more favorable outcomes with pembrolizumab while level of PD-L1 expression had no effect on outcomes in docetaxel.
Results of the KEYNOTE-010 study were published earlier this year in the Lancet, but the results reported at the annual meeting of the American Society of Clinical Oncology further categorized study participants by tumor proportion score, a measure of viable tumor cells showing partial or complete membrane staining for PD-L1.
This finding verifies PD-L1 as a “predictive biomarker for ‘pembro’ in NSCLC,” lead study author Dr. Paul Baas of the Netherlands Cancer Institute, Amsterdam, said.
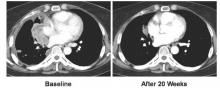
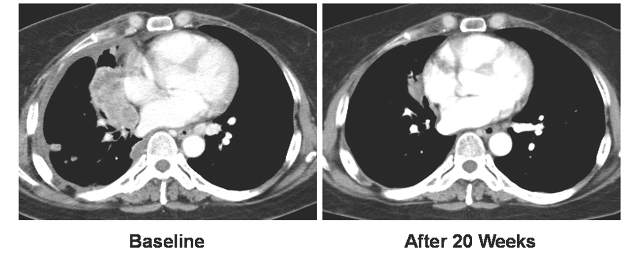
Out of 690 patients receiving pembrolizumab, 184 had a tumor proportion score (TPS) of 75% or more. For those 184 patients, median overall survival was 16.6 months and median progression-free survival was 6.2 months. The overall response rate was 33.7% for patients receiving pembrolizumab with TPS greater than 75%.
These survival and response rates were significantly higher, compared with the 8.2 month median overall survival, 4.0 month progression-free survival, and 7% overall response rate observed in 100 patients with the same TPS score who instead received docetaxel (overall survival: hazard ratio, 0.51; 95% confidence interval, 0.36-0.73; P less than .0001; progression-free survival: HR, 0.52; 95% CI, 0.38-0.69; P less than .0001; overall response rate: P less than .0001).
For the 106 patients receiving pembrolizumab who had a TPS between 50% and 74%, median OS was 15.8 months, PFS was 4.3 months, and the ORR was 22.6%. Median overall survival was significantly higher for pembrolizumab than docetaxel which was 8.2 months (HR, 0.58; 95% CI, 0.36-0.95; P = .01). ORR was also significantly higher for pembrolizumab than for docetaxel, which was 9.6% (P = .01).
The KEYNOTE-010 study was funded by Merck Sharp & Dohme. Dr. Baas reported having a consulting or advisory role and receiving honoraria and financial compensation from multiple companies including Merck Sharp & Dohme.
CHICAGO – The PD-1 inhibitor, pembrolizumab, is superior to docetaxel in prolonging overall and progression-free survival in patients with non–small cell lung cancer (NSCLC), according to the results of the phase III KEYNOTE-010 trial.
Furthermore, increasing PD-L1 expression was associated with more favorable outcomes with pembrolizumab while level of PD-L1 expression had no effect on outcomes in docetaxel.
Results of the KEYNOTE-010 study were published earlier this year in the Lancet, but the results reported at the annual meeting of the American Society of Clinical Oncology further categorized study participants by tumor proportion score, a measure of viable tumor cells showing partial or complete membrane staining for PD-L1.
This finding verifies PD-L1 as a “predictive biomarker for ‘pembro’ in NSCLC,” lead study author Dr. Paul Baas of the Netherlands Cancer Institute, Amsterdam, said.
Out of 690 patients receiving pembrolizumab, 184 had a tumor proportion score (TPS) of 75% or more. For those 184 patients, median overall survival was 16.6 months and median progression-free survival was 6.2 months. The overall response rate was 33.7% for patients receiving pembrolizumab with TPS greater than 75%.
These survival and response rates were significantly higher, compared with the 8.2 month median overall survival, 4.0 month progression-free survival, and 7% overall response rate observed in 100 patients with the same TPS score who instead received docetaxel (overall survival: hazard ratio, 0.51; 95% confidence interval, 0.36-0.73; P less than .0001; progression-free survival: HR, 0.52; 95% CI, 0.38-0.69; P less than .0001; overall response rate: P less than .0001).
For the 106 patients receiving pembrolizumab who had a TPS between 50% and 74%, median OS was 15.8 months, PFS was 4.3 months, and the ORR was 22.6%. Median overall survival was significantly higher for pembrolizumab than docetaxel which was 8.2 months (HR, 0.58; 95% CI, 0.36-0.95; P = .01). ORR was also significantly higher for pembrolizumab than for docetaxel, which was 9.6% (P = .01).
The KEYNOTE-010 study was funded by Merck Sharp & Dohme. Dr. Baas reported having a consulting or advisory role and receiving honoraria and financial compensation from multiple companies including Merck Sharp & Dohme.
CHICAGO – The PD-1 inhibitor, pembrolizumab, is superior to docetaxel in prolonging overall and progression-free survival in patients with non–small cell lung cancer (NSCLC), according to the results of the phase III KEYNOTE-010 trial.
Furthermore, increasing PD-L1 expression was associated with more favorable outcomes with pembrolizumab while level of PD-L1 expression had no effect on outcomes in docetaxel.
Results of the KEYNOTE-010 study were published earlier this year in the Lancet, but the results reported at the annual meeting of the American Society of Clinical Oncology further categorized study participants by tumor proportion score, a measure of viable tumor cells showing partial or complete membrane staining for PD-L1.
This finding verifies PD-L1 as a “predictive biomarker for ‘pembro’ in NSCLC,” lead study author Dr. Paul Baas of the Netherlands Cancer Institute, Amsterdam, said.
Out of 690 patients receiving pembrolizumab, 184 had a tumor proportion score (TPS) of 75% or more. For those 184 patients, median overall survival was 16.6 months and median progression-free survival was 6.2 months. The overall response rate was 33.7% for patients receiving pembrolizumab with TPS greater than 75%.
These survival and response rates were significantly higher, compared with the 8.2 month median overall survival, 4.0 month progression-free survival, and 7% overall response rate observed in 100 patients with the same TPS score who instead received docetaxel (overall survival: hazard ratio, 0.51; 95% confidence interval, 0.36-0.73; P less than .0001; progression-free survival: HR, 0.52; 95% CI, 0.38-0.69; P less than .0001; overall response rate: P less than .0001).
For the 106 patients receiving pembrolizumab who had a TPS between 50% and 74%, median OS was 15.8 months, PFS was 4.3 months, and the ORR was 22.6%. Median overall survival was significantly higher for pembrolizumab than docetaxel which was 8.2 months (HR, 0.58; 95% CI, 0.36-0.95; P = .01). ORR was also significantly higher for pembrolizumab than for docetaxel, which was 9.6% (P = .01).
The KEYNOTE-010 study was funded by Merck Sharp & Dohme. Dr. Baas reported having a consulting or advisory role and receiving honoraria and financial compensation from multiple companies including Merck Sharp & Dohme.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Pembrolizumab is superior to docetaxel in patients with non–small cell lung cancer especially at higher TPS. PD-L1 may be a predictive biomarker for pembrolizumab.
Major finding: For patients with tumor proportion scores of 75% or higher, median overall survival for pembrolizumab as single agent therapy was 16.6 months and only 8.2 months for single agent docetaxel (hazard ratio, .51; 95% confidence interval, .36-.73; P less than .0001).
Data source: The phase III KEYNOTE-010 trial of 1,034 patients with NSCLC.
Disclosures: The KEYNOTE-010 study was funded by Merck Sharp & Dohme. Dr. Baas reported having a consulting or advisory role and having received honoraria and financial compensation from multiple companies, including Merck Sharp & Dohme.
Study results differ on benefits of proton radiotherapy in NSCLC
CHICAGO – Whether proton radiotherapy is an option superior to photon radiotherapy for patients with non–small-cell lung cancer is still unclear, in light of results from a small randomized clinical trial and a review of the National Cancer Data Base.
The randomized trial of 149 patients showed there was no significant difference in radiation pneumonitis incidence rate nor overall survival by treatment received, while a retrospective analysis of 140,383 patients revealed that receipt of proton radiotherapy was associated with higher 5-year survival rates. The findings were presented at the annual meeting of the American Society of Clinical Oncology.
The Bayesian randomized trial compared rates of treatment failure and adverse events in patients receiving either intensity-modulated radiotherapy (IMRT) or 3D passively scattered proton therapy (3DPT). Of the 149 patients who met randomization requirements, 92 received IMRT and 57 received 3DPT. All patients received concurrent chemotherapy, and the patient characteristics were well balanced, although in the 3DPT group target volumes were larger.
Treatment failure rates at 12 months were 15.6% in the IMRT group and 24.6% in the 3DPT group. The median time to treatment failure was 10.5 months for both groups, reported Dr. Zhongxing Liao of the University of Texas MD Anderson Cancer Center, Houston.
The incident rates of radiation pneumonitis were 7.2% among patients receiving IMRT and 11% among patients receiving 3DPT; the median time to radiation pneumonitis was 4.5 and 4.0 months, respectively.
Dr. Liao pointed out that, historically, incidence rates of radiation pneumonitis were 15% in IMRT and 5% in 3DPT, and that rates observed in this study represented a deviation from those rates.
Local recurrence occurred in 22.8% of the IMRT patients and 24.6% of the 3DPT patients; time to local occurrence was 12.7 and 13.4 months, respectively. Finally, statistical analysis revealed no significant difference in overall survival by treatment received, Dr. Liao reported.
“Considerably fewer [adverse] events occurred in the current trial actually suggesting that both IMRT and the proton [therapy] are excellent treatments for non–small-cell lung cancer,” Dr. Liao said. “No differences were found between IMRT versus 3DPT in treatment failure in this randomized trial.”
Dr. Liao pointed out that as the trial went on, patients experienced fewer adverse events and went longer periods of time before experiencing treatment failure. This was an indication that the administration of both proton therapy and radiation therapy improved over time, he said.
In another study presented at the meeting, investigators used the National Cancer Data Base to collect demographic and clinical data on 140,383 patients with non–small-cell lung cancer who were treated with thoracic radiation from 2004 to 2012.
Across the entire cohort, median age was 68 years, 57% were male, 59% had stage II or III cancer, and 85% of patients were white. Only 348 patients received proton radiotherapy while the remaining 140,035 patients received photon radiotherapy, reported Madhusmita Behera, Ph.D., of Winship Cancer Institute at Emory University in Acworth, Georgia.
Multivariate analysis revealed that receipt of photon radiotherapy was associated with an increased risk of mortality, compared to proton radiotherapy (hazard ratio, 1.46; P less than .001).
For patients with stage II or III disease, 5-year overall survival rates were 15% for those who received photon radiotherapy and 22.3% for those who received proton radiotherapy (P = .01).
Patients were less likely to receive proton radiotherapy in community (odds ratio, .2; P less than .001) or comprehensive community (OR, .32; P less than .001) centers compared to academic centers, Dr. Behera reported.
Among the patients who received proton radiotherapy, 45.12% reported residing in a geographical location (defined by ZIP codes) with a median income quartile of $46,000 or more, the “highest median income quartile according to the U.S. census.” In addition, “only 14% of patients were from ZIP codes where more than 29% did not have a high school degree,” Dr. Behera said.
Both Dr. Behera and Dr. Liao noted that insurance denial continues to be a barrier to proton therapy. Following enrollment, 26 patients were denied insurance coverage for proton radiotherapy, Dr. Liao reported.
“This is only the beginning of the story of randomized trials [studying] proton and photon therapy in lung cancer in my opinion,” Dr. Liao said. “We only randomized patients with equivalent plans which may have excluded patients who could have benefited from protons the most.”
The study headed by Dr. Liao was funded by the MD Anderson Cancer Center and the National Cancer Institute. Dr. Liao had no relevant disclosures. One coinvestigator reported having stock or other ownership interest in Liquid Biotech, USA. The study headed by Dr. Behera was funded by the Biostatistics and Bioinformatics Shared Resource of Winship Cancer Institute of Emory University and the National Cancer Institute. Dr. Behera had no relevant disclosures; four of her coinvestigators disclosed having consulting or advisory roles or receiving financial compensation or honoraria from multiple companies.
On Twitter @jessnicolecraig
CHICAGO – Whether proton radiotherapy is an option superior to photon radiotherapy for patients with non–small-cell lung cancer is still unclear, in light of results from a small randomized clinical trial and a review of the National Cancer Data Base.
The randomized trial of 149 patients showed there was no significant difference in radiation pneumonitis incidence rate nor overall survival by treatment received, while a retrospective analysis of 140,383 patients revealed that receipt of proton radiotherapy was associated with higher 5-year survival rates. The findings were presented at the annual meeting of the American Society of Clinical Oncology.
The Bayesian randomized trial compared rates of treatment failure and adverse events in patients receiving either intensity-modulated radiotherapy (IMRT) or 3D passively scattered proton therapy (3DPT). Of the 149 patients who met randomization requirements, 92 received IMRT and 57 received 3DPT. All patients received concurrent chemotherapy, and the patient characteristics were well balanced, although in the 3DPT group target volumes were larger.
Treatment failure rates at 12 months were 15.6% in the IMRT group and 24.6% in the 3DPT group. The median time to treatment failure was 10.5 months for both groups, reported Dr. Zhongxing Liao of the University of Texas MD Anderson Cancer Center, Houston.
The incident rates of radiation pneumonitis were 7.2% among patients receiving IMRT and 11% among patients receiving 3DPT; the median time to radiation pneumonitis was 4.5 and 4.0 months, respectively.
Dr. Liao pointed out that, historically, incidence rates of radiation pneumonitis were 15% in IMRT and 5% in 3DPT, and that rates observed in this study represented a deviation from those rates.
Local recurrence occurred in 22.8% of the IMRT patients and 24.6% of the 3DPT patients; time to local occurrence was 12.7 and 13.4 months, respectively. Finally, statistical analysis revealed no significant difference in overall survival by treatment received, Dr. Liao reported.
“Considerably fewer [adverse] events occurred in the current trial actually suggesting that both IMRT and the proton [therapy] are excellent treatments for non–small-cell lung cancer,” Dr. Liao said. “No differences were found between IMRT versus 3DPT in treatment failure in this randomized trial.”
Dr. Liao pointed out that as the trial went on, patients experienced fewer adverse events and went longer periods of time before experiencing treatment failure. This was an indication that the administration of both proton therapy and radiation therapy improved over time, he said.
In another study presented at the meeting, investigators used the National Cancer Data Base to collect demographic and clinical data on 140,383 patients with non–small-cell lung cancer who were treated with thoracic radiation from 2004 to 2012.
Across the entire cohort, median age was 68 years, 57% were male, 59% had stage II or III cancer, and 85% of patients were white. Only 348 patients received proton radiotherapy while the remaining 140,035 patients received photon radiotherapy, reported Madhusmita Behera, Ph.D., of Winship Cancer Institute at Emory University in Acworth, Georgia.
Multivariate analysis revealed that receipt of photon radiotherapy was associated with an increased risk of mortality, compared to proton radiotherapy (hazard ratio, 1.46; P less than .001).
For patients with stage II or III disease, 5-year overall survival rates were 15% for those who received photon radiotherapy and 22.3% for those who received proton radiotherapy (P = .01).
Patients were less likely to receive proton radiotherapy in community (odds ratio, .2; P less than .001) or comprehensive community (OR, .32; P less than .001) centers compared to academic centers, Dr. Behera reported.
Among the patients who received proton radiotherapy, 45.12% reported residing in a geographical location (defined by ZIP codes) with a median income quartile of $46,000 or more, the “highest median income quartile according to the U.S. census.” In addition, “only 14% of patients were from ZIP codes where more than 29% did not have a high school degree,” Dr. Behera said.
Both Dr. Behera and Dr. Liao noted that insurance denial continues to be a barrier to proton therapy. Following enrollment, 26 patients were denied insurance coverage for proton radiotherapy, Dr. Liao reported.
“This is only the beginning of the story of randomized trials [studying] proton and photon therapy in lung cancer in my opinion,” Dr. Liao said. “We only randomized patients with equivalent plans which may have excluded patients who could have benefited from protons the most.”
The study headed by Dr. Liao was funded by the MD Anderson Cancer Center and the National Cancer Institute. Dr. Liao had no relevant disclosures. One coinvestigator reported having stock or other ownership interest in Liquid Biotech, USA. The study headed by Dr. Behera was funded by the Biostatistics and Bioinformatics Shared Resource of Winship Cancer Institute of Emory University and the National Cancer Institute. Dr. Behera had no relevant disclosures; four of her coinvestigators disclosed having consulting or advisory roles or receiving financial compensation or honoraria from multiple companies.
On Twitter @jessnicolecraig
CHICAGO – Whether proton radiotherapy is an option superior to photon radiotherapy for patients with non–small-cell lung cancer is still unclear, in light of results from a small randomized clinical trial and a review of the National Cancer Data Base.
The randomized trial of 149 patients showed there was no significant difference in radiation pneumonitis incidence rate nor overall survival by treatment received, while a retrospective analysis of 140,383 patients revealed that receipt of proton radiotherapy was associated with higher 5-year survival rates. The findings were presented at the annual meeting of the American Society of Clinical Oncology.
The Bayesian randomized trial compared rates of treatment failure and adverse events in patients receiving either intensity-modulated radiotherapy (IMRT) or 3D passively scattered proton therapy (3DPT). Of the 149 patients who met randomization requirements, 92 received IMRT and 57 received 3DPT. All patients received concurrent chemotherapy, and the patient characteristics were well balanced, although in the 3DPT group target volumes were larger.
Treatment failure rates at 12 months were 15.6% in the IMRT group and 24.6% in the 3DPT group. The median time to treatment failure was 10.5 months for both groups, reported Dr. Zhongxing Liao of the University of Texas MD Anderson Cancer Center, Houston.
The incident rates of radiation pneumonitis were 7.2% among patients receiving IMRT and 11% among patients receiving 3DPT; the median time to radiation pneumonitis was 4.5 and 4.0 months, respectively.
Dr. Liao pointed out that, historically, incidence rates of radiation pneumonitis were 15% in IMRT and 5% in 3DPT, and that rates observed in this study represented a deviation from those rates.
Local recurrence occurred in 22.8% of the IMRT patients and 24.6% of the 3DPT patients; time to local occurrence was 12.7 and 13.4 months, respectively. Finally, statistical analysis revealed no significant difference in overall survival by treatment received, Dr. Liao reported.
“Considerably fewer [adverse] events occurred in the current trial actually suggesting that both IMRT and the proton [therapy] are excellent treatments for non–small-cell lung cancer,” Dr. Liao said. “No differences were found between IMRT versus 3DPT in treatment failure in this randomized trial.”
Dr. Liao pointed out that as the trial went on, patients experienced fewer adverse events and went longer periods of time before experiencing treatment failure. This was an indication that the administration of both proton therapy and radiation therapy improved over time, he said.
In another study presented at the meeting, investigators used the National Cancer Data Base to collect demographic and clinical data on 140,383 patients with non–small-cell lung cancer who were treated with thoracic radiation from 2004 to 2012.
Across the entire cohort, median age was 68 years, 57% were male, 59% had stage II or III cancer, and 85% of patients were white. Only 348 patients received proton radiotherapy while the remaining 140,035 patients received photon radiotherapy, reported Madhusmita Behera, Ph.D., of Winship Cancer Institute at Emory University in Acworth, Georgia.
Multivariate analysis revealed that receipt of photon radiotherapy was associated with an increased risk of mortality, compared to proton radiotherapy (hazard ratio, 1.46; P less than .001).
For patients with stage II or III disease, 5-year overall survival rates were 15% for those who received photon radiotherapy and 22.3% for those who received proton radiotherapy (P = .01).
Patients were less likely to receive proton radiotherapy in community (odds ratio, .2; P less than .001) or comprehensive community (OR, .32; P less than .001) centers compared to academic centers, Dr. Behera reported.
Among the patients who received proton radiotherapy, 45.12% reported residing in a geographical location (defined by ZIP codes) with a median income quartile of $46,000 or more, the “highest median income quartile according to the U.S. census.” In addition, “only 14% of patients were from ZIP codes where more than 29% did not have a high school degree,” Dr. Behera said.
Both Dr. Behera and Dr. Liao noted that insurance denial continues to be a barrier to proton therapy. Following enrollment, 26 patients were denied insurance coverage for proton radiotherapy, Dr. Liao reported.
“This is only the beginning of the story of randomized trials [studying] proton and photon therapy in lung cancer in my opinion,” Dr. Liao said. “We only randomized patients with equivalent plans which may have excluded patients who could have benefited from protons the most.”
The study headed by Dr. Liao was funded by the MD Anderson Cancer Center and the National Cancer Institute. Dr. Liao had no relevant disclosures. One coinvestigator reported having stock or other ownership interest in Liquid Biotech, USA. The study headed by Dr. Behera was funded by the Biostatistics and Bioinformatics Shared Resource of Winship Cancer Institute of Emory University and the National Cancer Institute. Dr. Behera had no relevant disclosures; four of her coinvestigators disclosed having consulting or advisory roles or receiving financial compensation or honoraria from multiple companies.
On Twitter @jessnicolecraig
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Results of a small randomized trial and an analysis of the National Cancer Data Base show conflicting results about the benefits of proton therapy in NSCLC patients.
Major finding: In the small randomized trial, there was no significant difference in overall survival between photon and proton radiotherapy. The analysis of patient records from the NCDB revealed that receipt of photon radiotherapy was associated with an increased risk of mortality compared to proton radiotherapy (hazard ratio, 1.46; P less than .001).
Data source: A randomized trial of 149 patients and an analysis of 140,383 patients with NSCLC who received either proton radiotherapy or photon radiotherapy.
Disclosures: The study headed by Dr. Liao was funded by the MD Anderson Cancer Center and the National Cancer Institute. Dr. Liao had no relevant disclosures. One coinvestigator reported having stock or other ownership interest in Liquid Biotech, USA. The study headed by Dr. Behera was funded by the Biostatistics and Bioinformatics Shared Resource of Winship Cancer Institute of Emory University and the National Cancer Institute. Dr. Behera had no relevant disclosures, and four of her coinvestigators disclosed having consulting or advisory roles or receiving financial compensation or honoraria from multiple companies.
Pembrolizumab paired with immunostimulator is safe and tolerable
CHICAGO – Combining an immunostimulatory agent with the PD-1 checkpoint inhibitor pembrolizumab appeared quite safe and very tolerable, in a small phase Ib study.
There were some signs of efficacy against a variety of solid tumors, as well as biomarker trends showing immune activity.
In the phase Ib trial, researchers combined escalating doses (0.45-5.0 mg/kg) of PF-2566, an investigative immunostimulatory agent, with the anti–PD-1 checkpoint inhibitor pembrolizumab at 2 mg/kg, with both drugs given intravenously once every 3 weeks for a maximum of 32 cycles. A primary objective of the trial was to determine a maximum tolerated dose. Secondary objectives were to assess safety and tolerability and to determine any antitumor responses.
PF-2566 (Utomilumab/PF-05082566) is a monoclonal agonist targeting 4-1BB, a “costimulatory molecule that’s induced upon T-cell receptor activation and ultimately enhances cytotoxic T-cell response and effector status,” said Dr. Anthony Tolcher of the START Center for Cancer Care, San Antonio, at the annual meeting of the American Society of Clinical Oncology.
Eligible patients were 18 years or older, had a performance status of 0-1, and had advanced or metastatic solid tumors that had progressed on standard therapy or for which no standard therapy was available. They could not have had any form of immunosuppressive therapy in the 2 weeks prior to registration, a monoclonal antibody in the 2 months before the first dose, or any symptomatic or progressing central nervous system primary malignancies. Prior pembrolizumab was permitted.
Twenty-three patients (14 males) were heavily pretreated with a median of three prior therapies (range 0-9) for a variety of cancers, including six non–small-cell lung, five renal cell, three head and neck, and two each pancreatic and thyroid cancers.
Good safety and tolerability profiles
The most prevalent treatment-emergent adverse events (AEs) were fatigue, rash, cough, nausea, and decreased appetite, affecting 7-10 patients each. All were grade 1/2 except for one grade 3/4 case of fatigue and three cases of grade 3/4 anemia among the 23 patients. Most treatment-related AE’s were grade 1/2, largely fatigue (n = 8) and rash (n = 9). There was one case each of grade 3 adrenal insufficiency and hypokalemia. No patient discontinued the trial because of a treatment-related toxicity. Dr. Tolcher noted that adrenal insufficiency has been reported previously with the use of PD-1 inhibitors. “There does not appear to be any evidence of synergistic or additive toxicity in this patient population,” he said.
Neither drug affected the pharmacokinetics of the other drug or the development of antibodies to the other drug. The maximum tolerated dose of PF-2566 was at least 5 mg/kg every 3 weeks when combined with pembrolizumab 2 mg/kg. No dose-limiting toxicity was observed across the PF-2566 dosing range. And there were no treatment-emergent AEs of clinical relevance.
Pharmacodynamics and efficacy
By day 1 of cycle 5, “there [was] a trend toward increasing numbers of activated CD8 [cytotoxic] T cells in patients who ultimately responded or had a complete response, compared to those that had stable disease or progressive disease. The same actually applies to the effector memory T cells,” Dr. Tolcher said but was careful to point out that the sample sizes were small and it was only a trend. Similarly, circulating levels of gamma-interferon, often used as a biomarker of activated T cells, were higher at 6 and 24 hours post dose in cycle 5 for those patients who ultimately had partial or complete responses, compared with those with progressive or stable disease.
Among the 23 patients, there were two confirmed complete responses and four partial responses as well as one unconfirmed partial response. If responses occurred, they often were durable past 1 year and even out close to 2 years.
The strengths of this study were that it enrolled heavily pretreated patients and there were no drug-drug interactions, no dose-limiting toxicities, and no treatment-related AE’s leading to discontinuation, “so in general a very well-tolerated immunotherapy combination,” said discussant Dr. David Spigel of the Sarah Cannon Research Institute in Nashville, Tenn. There were also some durable responses, and he said it was interesting to see that there were some blood biomarkers that correlated with responses.
“It was hard for me to find any weaknesses to this,” Dr. Spigel said, beside the fact that it was a small study. “So what does this change?” He said the combination of pembrolizumab and PF-2566 looks promising in light of some sustained responses in refractory tumors and its safety profile. For the future, expansion trial cohorts are still needed to confirm activity and safety, especially hepatic safety based on trial results with similar drugs, and PF-2566 is already being tested with rituximab in lymphoma and with an anti-CCR4 compound (mogamulizumab).
The study was sponsored by Pfizer and Merck. Dr. Tolcher has ties to several companies, including Pfizer and Merck. Dr. Spigel has ties to several companies, including Pfizer.
CHICAGO – Combining an immunostimulatory agent with the PD-1 checkpoint inhibitor pembrolizumab appeared quite safe and very tolerable, in a small phase Ib study.
There were some signs of efficacy against a variety of solid tumors, as well as biomarker trends showing immune activity.
In the phase Ib trial, researchers combined escalating doses (0.45-5.0 mg/kg) of PF-2566, an investigative immunostimulatory agent, with the anti–PD-1 checkpoint inhibitor pembrolizumab at 2 mg/kg, with both drugs given intravenously once every 3 weeks for a maximum of 32 cycles. A primary objective of the trial was to determine a maximum tolerated dose. Secondary objectives were to assess safety and tolerability and to determine any antitumor responses.
PF-2566 (Utomilumab/PF-05082566) is a monoclonal agonist targeting 4-1BB, a “costimulatory molecule that’s induced upon T-cell receptor activation and ultimately enhances cytotoxic T-cell response and effector status,” said Dr. Anthony Tolcher of the START Center for Cancer Care, San Antonio, at the annual meeting of the American Society of Clinical Oncology.
Eligible patients were 18 years or older, had a performance status of 0-1, and had advanced or metastatic solid tumors that had progressed on standard therapy or for which no standard therapy was available. They could not have had any form of immunosuppressive therapy in the 2 weeks prior to registration, a monoclonal antibody in the 2 months before the first dose, or any symptomatic or progressing central nervous system primary malignancies. Prior pembrolizumab was permitted.
Twenty-three patients (14 males) were heavily pretreated with a median of three prior therapies (range 0-9) for a variety of cancers, including six non–small-cell lung, five renal cell, three head and neck, and two each pancreatic and thyroid cancers.
Good safety and tolerability profiles
The most prevalent treatment-emergent adverse events (AEs) were fatigue, rash, cough, nausea, and decreased appetite, affecting 7-10 patients each. All were grade 1/2 except for one grade 3/4 case of fatigue and three cases of grade 3/4 anemia among the 23 patients. Most treatment-related AE’s were grade 1/2, largely fatigue (n = 8) and rash (n = 9). There was one case each of grade 3 adrenal insufficiency and hypokalemia. No patient discontinued the trial because of a treatment-related toxicity. Dr. Tolcher noted that adrenal insufficiency has been reported previously with the use of PD-1 inhibitors. “There does not appear to be any evidence of synergistic or additive toxicity in this patient population,” he said.
Neither drug affected the pharmacokinetics of the other drug or the development of antibodies to the other drug. The maximum tolerated dose of PF-2566 was at least 5 mg/kg every 3 weeks when combined with pembrolizumab 2 mg/kg. No dose-limiting toxicity was observed across the PF-2566 dosing range. And there were no treatment-emergent AEs of clinical relevance.
Pharmacodynamics and efficacy
By day 1 of cycle 5, “there [was] a trend toward increasing numbers of activated CD8 [cytotoxic] T cells in patients who ultimately responded or had a complete response, compared to those that had stable disease or progressive disease. The same actually applies to the effector memory T cells,” Dr. Tolcher said but was careful to point out that the sample sizes were small and it was only a trend. Similarly, circulating levels of gamma-interferon, often used as a biomarker of activated T cells, were higher at 6 and 24 hours post dose in cycle 5 for those patients who ultimately had partial or complete responses, compared with those with progressive or stable disease.
Among the 23 patients, there were two confirmed complete responses and four partial responses as well as one unconfirmed partial response. If responses occurred, they often were durable past 1 year and even out close to 2 years.
The strengths of this study were that it enrolled heavily pretreated patients and there were no drug-drug interactions, no dose-limiting toxicities, and no treatment-related AE’s leading to discontinuation, “so in general a very well-tolerated immunotherapy combination,” said discussant Dr. David Spigel of the Sarah Cannon Research Institute in Nashville, Tenn. There were also some durable responses, and he said it was interesting to see that there were some blood biomarkers that correlated with responses.
“It was hard for me to find any weaknesses to this,” Dr. Spigel said, beside the fact that it was a small study. “So what does this change?” He said the combination of pembrolizumab and PF-2566 looks promising in light of some sustained responses in refractory tumors and its safety profile. For the future, expansion trial cohorts are still needed to confirm activity and safety, especially hepatic safety based on trial results with similar drugs, and PF-2566 is already being tested with rituximab in lymphoma and with an anti-CCR4 compound (mogamulizumab).
The study was sponsored by Pfizer and Merck. Dr. Tolcher has ties to several companies, including Pfizer and Merck. Dr. Spigel has ties to several companies, including Pfizer.
CHICAGO – Combining an immunostimulatory agent with the PD-1 checkpoint inhibitor pembrolizumab appeared quite safe and very tolerable, in a small phase Ib study.
There were some signs of efficacy against a variety of solid tumors, as well as biomarker trends showing immune activity.
In the phase Ib trial, researchers combined escalating doses (0.45-5.0 mg/kg) of PF-2566, an investigative immunostimulatory agent, with the anti–PD-1 checkpoint inhibitor pembrolizumab at 2 mg/kg, with both drugs given intravenously once every 3 weeks for a maximum of 32 cycles. A primary objective of the trial was to determine a maximum tolerated dose. Secondary objectives were to assess safety and tolerability and to determine any antitumor responses.
PF-2566 (Utomilumab/PF-05082566) is a monoclonal agonist targeting 4-1BB, a “costimulatory molecule that’s induced upon T-cell receptor activation and ultimately enhances cytotoxic T-cell response and effector status,” said Dr. Anthony Tolcher of the START Center for Cancer Care, San Antonio, at the annual meeting of the American Society of Clinical Oncology.
Eligible patients were 18 years or older, had a performance status of 0-1, and had advanced or metastatic solid tumors that had progressed on standard therapy or for which no standard therapy was available. They could not have had any form of immunosuppressive therapy in the 2 weeks prior to registration, a monoclonal antibody in the 2 months before the first dose, or any symptomatic or progressing central nervous system primary malignancies. Prior pembrolizumab was permitted.
Twenty-three patients (14 males) were heavily pretreated with a median of three prior therapies (range 0-9) for a variety of cancers, including six non–small-cell lung, five renal cell, three head and neck, and two each pancreatic and thyroid cancers.
Good safety and tolerability profiles
The most prevalent treatment-emergent adverse events (AEs) were fatigue, rash, cough, nausea, and decreased appetite, affecting 7-10 patients each. All were grade 1/2 except for one grade 3/4 case of fatigue and three cases of grade 3/4 anemia among the 23 patients. Most treatment-related AE’s were grade 1/2, largely fatigue (n = 8) and rash (n = 9). There was one case each of grade 3 adrenal insufficiency and hypokalemia. No patient discontinued the trial because of a treatment-related toxicity. Dr. Tolcher noted that adrenal insufficiency has been reported previously with the use of PD-1 inhibitors. “There does not appear to be any evidence of synergistic or additive toxicity in this patient population,” he said.
Neither drug affected the pharmacokinetics of the other drug or the development of antibodies to the other drug. The maximum tolerated dose of PF-2566 was at least 5 mg/kg every 3 weeks when combined with pembrolizumab 2 mg/kg. No dose-limiting toxicity was observed across the PF-2566 dosing range. And there were no treatment-emergent AEs of clinical relevance.
Pharmacodynamics and efficacy
By day 1 of cycle 5, “there [was] a trend toward increasing numbers of activated CD8 [cytotoxic] T cells in patients who ultimately responded or had a complete response, compared to those that had stable disease or progressive disease. The same actually applies to the effector memory T cells,” Dr. Tolcher said but was careful to point out that the sample sizes were small and it was only a trend. Similarly, circulating levels of gamma-interferon, often used as a biomarker of activated T cells, were higher at 6 and 24 hours post dose in cycle 5 for those patients who ultimately had partial or complete responses, compared with those with progressive or stable disease.
Among the 23 patients, there were two confirmed complete responses and four partial responses as well as one unconfirmed partial response. If responses occurred, they often were durable past 1 year and even out close to 2 years.
The strengths of this study were that it enrolled heavily pretreated patients and there were no drug-drug interactions, no dose-limiting toxicities, and no treatment-related AE’s leading to discontinuation, “so in general a very well-tolerated immunotherapy combination,” said discussant Dr. David Spigel of the Sarah Cannon Research Institute in Nashville, Tenn. There were also some durable responses, and he said it was interesting to see that there were some blood biomarkers that correlated with responses.
“It was hard for me to find any weaknesses to this,” Dr. Spigel said, beside the fact that it was a small study. “So what does this change?” He said the combination of pembrolizumab and PF-2566 looks promising in light of some sustained responses in refractory tumors and its safety profile. For the future, expansion trial cohorts are still needed to confirm activity and safety, especially hepatic safety based on trial results with similar drugs, and PF-2566 is already being tested with rituximab in lymphoma and with an anti-CCR4 compound (mogamulizumab).
The study was sponsored by Pfizer and Merck. Dr. Tolcher has ties to several companies, including Pfizer and Merck. Dr. Spigel has ties to several companies, including Pfizer.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Combining an immunostimulator with pembrolizumab had good tolerability and safety.
Major finding: Two complete and four partial responses occurred among 23 patients.
Data source: Phase Ib trial of 23 patients with a variety of solid tumors.
Disclosures: The study was sponsored by Pfizer and Merck. Dr. Tolcher has ties to several companies, including Pfizer and Merck. Dr. Spigel has ties to several companies, including Pfizer.
Immune agonist, checkpoint inhibitor combo shows good tolerability
CHICAGO – Combining two immunotherapies, one inhibiting immune suppression and the other stimulating immune activation, is well tolerated and shows activity for a variety of solid tumor types, according to a phase I trial presented at the annual meeting of the American Association of Clinical Oncology.
Investigators enrolled 51 patients with locally advanced or metastatic solid tumors of any type after progression on standard therapy to a phase Ib dose-escalation study using atezolizumab, a monoclonal antibody checkpoint inhibitor that targets PD-L1, in combination with MOXR0916 (MOXR), an agonist IgG1 monoclonal antibody targeting OX40, a costimulatory receptor. Atezolizumab received Food and Drug Administration approval in May 2016 for use in certain patients with urothelial carcinoma. There were 28 patients in a dose-escalation cohort of the study and 23 in a serial biopsy cohort. The dose of the drug combination was started at 12 mg and escalated to understand pharmacodynamic changes in the tumors.
“The pharmacokinetics of both MOXR0916 and atezolizumab were similar to their single-agent data, suggesting no interaction,” reported Dr. Jeffrey Infante of the Sarah Cannon Research Institute in Nashville, Tenn.
The drug combination was well tolerated through the entire escalation range of MOXR. There were no dose-limiting toxicities, and no maximal tolerated dose was reached. There were also no drug-related deaths or grade 4 toxicities or drug-related treatment discontinuations. One case of grade 3 pneumonitis, successfully managed with methylprednisolone and antibiotics, occurred at the MOXR 40-mg dose on cycle 4 of treatment in a patient with non–small-cell lung cancer, he said.
About half the patients (53%) experienced any form of adverse event on the drug combination, and only 8% were grade 2 or 3. There were very few adverse events of any one type, and they did not appear to cluster among patients on the higher MOXR doses. The most prevalent adverse events were nausea, fever, fatigue, and rash, and each was in the 8%-14% range and almost always grade 1.
Many patients showed efficacy of the regimens out to 6-7 cycles regardless of tumor type, and 8 of the 51 patients were still receiving the therapy past cycle 7 with partial responses.
The stimulatory molecule OX40 is not normally expressed on T cells, but it is expressed when antigen interacts with the T-cell receptor, and it can then interact with its ligand, OX40L. The result is production of inflammatory cytokines such as gamma-interferon, activation and survival of effector T cells, and production of memory T cells. At the same time, OX40 activity blocks the suppressive function of regulatory T cells.
“So a molecule that can be a cancer therapeutic such as an OX40 agonist has dual mechanisms of action,” Dr. Infante said. “It can costimulate effector T cells and at the same time inhibit regulatory T cells. Furthermore, there is a reduced risk of toxicity, potentially, as its activity is linked to antigen recognition.”
There is good rationale for using an OX40 agonist such as MOXR, either for its immune stimulatory function or to deactivate immune suppression by regulatory T cells, or both, said discussant Dr. Jedd Wolchok, chief, melanoma and immunotherapeutics service, Memorial Sloan-Kettering Cancer Center, New York. Dr. Infante’s dose-escalation study was “very nicely designed and showed quite good safety,” Dr. Wolchok said, though one thing he would have liked to have seen was a quantification of regulatory T cells in tumor biopsies.
“This [study] is very important considering that this is an agonist antibody, and the agonist agents need to be dosed very deliberatively, as was done here, to ensure safety of patients,” Dr. Wolchok said, adding that further research needs to target “optimal combinatorial partners” and explore other mechanistic biomarkers.
MOXR was given in this trial at escalating doses on a 3+3 design (0.8-1,200 mg) on the same day as atezolizumab 1,200 mg IV once every 3 weeks with a 21-day window for assessment of MOXR dose-limiting toxicities. MOXR doses of 300 mg maintained trough concentrations sufficient to saturate OX40 receptors. An expansion regimen using 300 mg MOXR with atezolizumab 1,200 mg every 3 weeks is underway and will assess efficacy in the treatment of melanoma, renal cell carcinoma, non–small-cell lung cancer, urothelial carcinoma, and triple-negative breast cancer.
The study was sponsored by Roche. Dr. Infante reported having no relevant financial disclosures. Dr. Wolchok owns stock in Potenza Therapeutics and Vesuvius Pharmaceuticals, has received travel expenses and/or has an advisory role with several other companies, and is a coinventor on an issued patent for DNA vaccines for the treatment of cancer in companion animals.
CHICAGO – Combining two immunotherapies, one inhibiting immune suppression and the other stimulating immune activation, is well tolerated and shows activity for a variety of solid tumor types, according to a phase I trial presented at the annual meeting of the American Association of Clinical Oncology.
Investigators enrolled 51 patients with locally advanced or metastatic solid tumors of any type after progression on standard therapy to a phase Ib dose-escalation study using atezolizumab, a monoclonal antibody checkpoint inhibitor that targets PD-L1, in combination with MOXR0916 (MOXR), an agonist IgG1 monoclonal antibody targeting OX40, a costimulatory receptor. Atezolizumab received Food and Drug Administration approval in May 2016 for use in certain patients with urothelial carcinoma. There were 28 patients in a dose-escalation cohort of the study and 23 in a serial biopsy cohort. The dose of the drug combination was started at 12 mg and escalated to understand pharmacodynamic changes in the tumors.
“The pharmacokinetics of both MOXR0916 and atezolizumab were similar to their single-agent data, suggesting no interaction,” reported Dr. Jeffrey Infante of the Sarah Cannon Research Institute in Nashville, Tenn.
The drug combination was well tolerated through the entire escalation range of MOXR. There were no dose-limiting toxicities, and no maximal tolerated dose was reached. There were also no drug-related deaths or grade 4 toxicities or drug-related treatment discontinuations. One case of grade 3 pneumonitis, successfully managed with methylprednisolone and antibiotics, occurred at the MOXR 40-mg dose on cycle 4 of treatment in a patient with non–small-cell lung cancer, he said.
About half the patients (53%) experienced any form of adverse event on the drug combination, and only 8% were grade 2 or 3. There were very few adverse events of any one type, and they did not appear to cluster among patients on the higher MOXR doses. The most prevalent adverse events were nausea, fever, fatigue, and rash, and each was in the 8%-14% range and almost always grade 1.
Many patients showed efficacy of the regimens out to 6-7 cycles regardless of tumor type, and 8 of the 51 patients were still receiving the therapy past cycle 7 with partial responses.
The stimulatory molecule OX40 is not normally expressed on T cells, but it is expressed when antigen interacts with the T-cell receptor, and it can then interact with its ligand, OX40L. The result is production of inflammatory cytokines such as gamma-interferon, activation and survival of effector T cells, and production of memory T cells. At the same time, OX40 activity blocks the suppressive function of regulatory T cells.
“So a molecule that can be a cancer therapeutic such as an OX40 agonist has dual mechanisms of action,” Dr. Infante said. “It can costimulate effector T cells and at the same time inhibit regulatory T cells. Furthermore, there is a reduced risk of toxicity, potentially, as its activity is linked to antigen recognition.”
There is good rationale for using an OX40 agonist such as MOXR, either for its immune stimulatory function or to deactivate immune suppression by regulatory T cells, or both, said discussant Dr. Jedd Wolchok, chief, melanoma and immunotherapeutics service, Memorial Sloan-Kettering Cancer Center, New York. Dr. Infante’s dose-escalation study was “very nicely designed and showed quite good safety,” Dr. Wolchok said, though one thing he would have liked to have seen was a quantification of regulatory T cells in tumor biopsies.
“This [study] is very important considering that this is an agonist antibody, and the agonist agents need to be dosed very deliberatively, as was done here, to ensure safety of patients,” Dr. Wolchok said, adding that further research needs to target “optimal combinatorial partners” and explore other mechanistic biomarkers.
MOXR was given in this trial at escalating doses on a 3+3 design (0.8-1,200 mg) on the same day as atezolizumab 1,200 mg IV once every 3 weeks with a 21-day window for assessment of MOXR dose-limiting toxicities. MOXR doses of 300 mg maintained trough concentrations sufficient to saturate OX40 receptors. An expansion regimen using 300 mg MOXR with atezolizumab 1,200 mg every 3 weeks is underway and will assess efficacy in the treatment of melanoma, renal cell carcinoma, non–small-cell lung cancer, urothelial carcinoma, and triple-negative breast cancer.
The study was sponsored by Roche. Dr. Infante reported having no relevant financial disclosures. Dr. Wolchok owns stock in Potenza Therapeutics and Vesuvius Pharmaceuticals, has received travel expenses and/or has an advisory role with several other companies, and is a coinventor on an issued patent for DNA vaccines for the treatment of cancer in companion animals.
CHICAGO – Combining two immunotherapies, one inhibiting immune suppression and the other stimulating immune activation, is well tolerated and shows activity for a variety of solid tumor types, according to a phase I trial presented at the annual meeting of the American Association of Clinical Oncology.
Investigators enrolled 51 patients with locally advanced or metastatic solid tumors of any type after progression on standard therapy to a phase Ib dose-escalation study using atezolizumab, a monoclonal antibody checkpoint inhibitor that targets PD-L1, in combination with MOXR0916 (MOXR), an agonist IgG1 monoclonal antibody targeting OX40, a costimulatory receptor. Atezolizumab received Food and Drug Administration approval in May 2016 for use in certain patients with urothelial carcinoma. There were 28 patients in a dose-escalation cohort of the study and 23 in a serial biopsy cohort. The dose of the drug combination was started at 12 mg and escalated to understand pharmacodynamic changes in the tumors.
“The pharmacokinetics of both MOXR0916 and atezolizumab were similar to their single-agent data, suggesting no interaction,” reported Dr. Jeffrey Infante of the Sarah Cannon Research Institute in Nashville, Tenn.
The drug combination was well tolerated through the entire escalation range of MOXR. There were no dose-limiting toxicities, and no maximal tolerated dose was reached. There were also no drug-related deaths or grade 4 toxicities or drug-related treatment discontinuations. One case of grade 3 pneumonitis, successfully managed with methylprednisolone and antibiotics, occurred at the MOXR 40-mg dose on cycle 4 of treatment in a patient with non–small-cell lung cancer, he said.
About half the patients (53%) experienced any form of adverse event on the drug combination, and only 8% were grade 2 or 3. There were very few adverse events of any one type, and they did not appear to cluster among patients on the higher MOXR doses. The most prevalent adverse events were nausea, fever, fatigue, and rash, and each was in the 8%-14% range and almost always grade 1.
Many patients showed efficacy of the regimens out to 6-7 cycles regardless of tumor type, and 8 of the 51 patients were still receiving the therapy past cycle 7 with partial responses.
The stimulatory molecule OX40 is not normally expressed on T cells, but it is expressed when antigen interacts with the T-cell receptor, and it can then interact with its ligand, OX40L. The result is production of inflammatory cytokines such as gamma-interferon, activation and survival of effector T cells, and production of memory T cells. At the same time, OX40 activity blocks the suppressive function of regulatory T cells.
“So a molecule that can be a cancer therapeutic such as an OX40 agonist has dual mechanisms of action,” Dr. Infante said. “It can costimulate effector T cells and at the same time inhibit regulatory T cells. Furthermore, there is a reduced risk of toxicity, potentially, as its activity is linked to antigen recognition.”
There is good rationale for using an OX40 agonist such as MOXR, either for its immune stimulatory function or to deactivate immune suppression by regulatory T cells, or both, said discussant Dr. Jedd Wolchok, chief, melanoma and immunotherapeutics service, Memorial Sloan-Kettering Cancer Center, New York. Dr. Infante’s dose-escalation study was “very nicely designed and showed quite good safety,” Dr. Wolchok said, though one thing he would have liked to have seen was a quantification of regulatory T cells in tumor biopsies.
“This [study] is very important considering that this is an agonist antibody, and the agonist agents need to be dosed very deliberatively, as was done here, to ensure safety of patients,” Dr. Wolchok said, adding that further research needs to target “optimal combinatorial partners” and explore other mechanistic biomarkers.
MOXR was given in this trial at escalating doses on a 3+3 design (0.8-1,200 mg) on the same day as atezolizumab 1,200 mg IV once every 3 weeks with a 21-day window for assessment of MOXR dose-limiting toxicities. MOXR doses of 300 mg maintained trough concentrations sufficient to saturate OX40 receptors. An expansion regimen using 300 mg MOXR with atezolizumab 1,200 mg every 3 weeks is underway and will assess efficacy in the treatment of melanoma, renal cell carcinoma, non–small-cell lung cancer, urothelial carcinoma, and triple-negative breast cancer.
The study was sponsored by Roche. Dr. Infante reported having no relevant financial disclosures. Dr. Wolchok owns stock in Potenza Therapeutics and Vesuvius Pharmaceuticals, has received travel expenses and/or has an advisory role with several other companies, and is a coinventor on an issued patent for DNA vaccines for the treatment of cancer in companion animals.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Combining an immune agonist and a checkpoint inhibitor shows good tolerability.
Major finding: Eighty-five percent of adverse effects were grade 1; the rest were grade 2/3.
Data source: A phase Ib, open-label multicenter study of 51 patients.
Disclosures: The study was sponsored by Roche. Dr. Infante reported having no relevant financial disclosures. Dr. Wolchok owns stock in Potenza Therapeutics and Vesuvius Pharmaceuticals, has received travel expenses and/or has an advisory role with several other companies, and is a coinventor on an issued patent for DNA vaccines for the treatment of cancer in companion animals.
Dual checkpoint blockade doubles response in SCLC
CHICAGO – A combination of checkpoint inhibitors nivolumab plus ipilimumab produced superior objective response rates, progression-free survival, and overall survival, compared with nivolumab alone for previously treated patients with small cell lung cancer but at a cost of added toxicity, according to a phase I/II study presented at the annual meeting of the American Society of Clinical Oncology.
The observed responses were independent of tumor platinum sensitivity and programmed death-ligand 1 (PD-L1) expression.
Nivolumab as a single agent had activity, showing a 10% objective response rate, which was nearly the same whether the tumors were platinum sensitive or platinum resistant. However, the response rates were doubled for patients treated with combination therapy, reported Dr. Scott Antonia of the H. Lee Moffitt Cancer Center & Research Institute in Tampa.
The phase I/II Checkmate 032 enrolled 216 patients with progressive small cell lung cancer (SCLC) after one or more prior lines of therapy including a first-line platinum-based regimen. Patients were not selected based on PD-L1 expression in their tumors. They were assigned to treatment with nivolumab 3 mg/kg intravenously (IV) every 2 weeks (Niv3; n = 98), to nivolumab 1 mg/kg + ipilimumab 3 mg/kg IV every 3 weeks for four cycles (Niv1/Ipi3; n = 61), or to nivolumab 3 mg/kg + ipilimumab 1 mg/kg IV every 3 weeks for four cycles (Niv3/Ipi1; n = 54). All patients then received nivolumab 3 mg/kg IV every 2 weeks.
The arms of the trial were well matched for age, sex, race, number of prior treatment regimens, smoking history, and tumor PD-L1 level of expression. “The interesting thing with small cell is that very few of the patients actually have tumors that express PD-L1,” Dr. Antonia said. “Only about a quarter of them express PD-L1, probably reflective of the fact that there are very few tumor-infiltrating lymphocytes generating gamma-interferon, and inducing this molecule.”
Combination therapy beats single agent
Nivolumab as a single agent had activity, showing a 10% objective response rate, which was nearly the same whether the tumors were platinum sensitive or platinum resistant. “The response rates were doubled when we treated the patients with combination therapy,” Dr. Antonia said, to 23% in the Niv1/Ipi3 arm and to 19% in the Niv3/Ipi1 arm. He said while it is still early to tell, the responses appear to have similar characteristics to what has been seen with non–small cell lung cancer, that is, even patients with bulky disease can have responses.
Only two complete responses occurred, and those were among the 61 patients in the Niv1/Ipi3 arm. All the rest of the responses were either partial or were stable disease. Most responses were rapid, occurring in the first 6-12 weeks. In many of the cases, responses were durable out past 1 year. A few rare cases of pseudoprogression were seen. “Responsiveness did not require PD-L1 expression. … The PD-L1–negative patients responded just as well as the PD-L1–positive patients,” Dr. Antonia said.
The greatest number of patients with tumor regression from baseline occurred in the Niv1/Ipi3 arm, contributing to the investigators’ decision to choose this combination to move forward into phase III trials. Again, while it is still early, the Niv1/Ipi3 combination produced the best overall survival, with a median of 7.7 months and a 1-year overall survival rate of 43%, with a median follow-up of 16.5 months.
Efficacy comes at a cost of toxicity
Toxicity of the treatments was consistent with what has been seen using these drugs with other cancers. Toxicity was greatest in the drug combination arms. For Niv3, 53% of patients had any grade of a treatment related adverse effect (AE). Grade 3/4 AEs affected 13%. For the arms combining nivolumab with ipilimumab, total AEs were in the 74%-79% range, with grade 3/4 AEs affecting 19%-30%. The most common AEs, largely grade 1/2, were fatigue, pruritus, and diarrhea, which were more prevalent when ipilimumab was used. Interestingly, with the higher dose of ipilimumab, 16% of patients experienced low-grade hypothyroidism, and 11% experienced low-grade hyperthyroidism. Treatment-related AEs leading to discontinuation occurred in 6% of the Niv3 arm, 18% of the Niv1/Ipi3 arm, and 13% of the Niv3/Ipi1 arm. Three treatment-related deaths occurred, all among the 115 patients in the combination arms. Dr. Antonia said even grade 3/4 immune-related AEs were well managed using established safety guidelines.
Trial results were simultaneously published in the Lancet Oncology (2016 Jun 3. pii: S1470-2045[16]30098-5. doi: 10.1016/S1470-2045[16]30098-5).
This study is being expanded to include more patients, and nivolumab alone and Niv1/Ipi3 are being tested earlier in the disease as front-line consolidation or maintenance therapy just after platinum-based therapy.
Discussant Dr. Jedd Wolchok of Memorial Sloan Kettering Cancer Center, New York, said people working in immunotherapy feel like they have been riding a wave over the past few years and have just washed up on a beach, wondering where to go from here. “I think combination therapies have really announced themselves as being the next step forward,” he said. Early studies with checkpoint inhibitors produced some strong and durable remissions, but for the most part, in only a minority of patients.
So investigators started to try checkpoint inhibitors with nonredundant targets, as Dr. Antonia’s study did. Nivolumab + ipilimumab has been tried in melanoma, but grade 3/4 toxicity was more prevalent than in this SCLC trial. Dr. Wolchok suggested maybe SCLC patients are more immunosuppressed and therefore do not react to the combination treatment as strongly. The toxicity was lower even than that seen in non-SCLC, “so even in diseases that occur at the same organ site, there is a different biology in the microenvironment that is leading to different tolerability, different degree of immune suppression,” he said.
He congratulated the Checkmate 032 investigators and sponsors for testing different doses and schedules “because this is not one-size-fits-all.” But he said more patients have to be studied. And remaining questions concern the nature of the response, that is, is the response deeper with the combination therapy, and balanced against the additional toxicity?
He noted that not all patients in Dr. Antonia’s study had fine needle aspirates, so their tumors could not be evaluated for PD-L1 expression. So it is still not entirely clear if a subset of patients could benefit the most, such as ones with higher expression.
CHICAGO – A combination of checkpoint inhibitors nivolumab plus ipilimumab produced superior objective response rates, progression-free survival, and overall survival, compared with nivolumab alone for previously treated patients with small cell lung cancer but at a cost of added toxicity, according to a phase I/II study presented at the annual meeting of the American Society of Clinical Oncology.
The observed responses were independent of tumor platinum sensitivity and programmed death-ligand 1 (PD-L1) expression.
Nivolumab as a single agent had activity, showing a 10% objective response rate, which was nearly the same whether the tumors were platinum sensitive or platinum resistant. However, the response rates were doubled for patients treated with combination therapy, reported Dr. Scott Antonia of the H. Lee Moffitt Cancer Center & Research Institute in Tampa.
The phase I/II Checkmate 032 enrolled 216 patients with progressive small cell lung cancer (SCLC) after one or more prior lines of therapy including a first-line platinum-based regimen. Patients were not selected based on PD-L1 expression in their tumors. They were assigned to treatment with nivolumab 3 mg/kg intravenously (IV) every 2 weeks (Niv3; n = 98), to nivolumab 1 mg/kg + ipilimumab 3 mg/kg IV every 3 weeks for four cycles (Niv1/Ipi3; n = 61), or to nivolumab 3 mg/kg + ipilimumab 1 mg/kg IV every 3 weeks for four cycles (Niv3/Ipi1; n = 54). All patients then received nivolumab 3 mg/kg IV every 2 weeks.
The arms of the trial were well matched for age, sex, race, number of prior treatment regimens, smoking history, and tumor PD-L1 level of expression. “The interesting thing with small cell is that very few of the patients actually have tumors that express PD-L1,” Dr. Antonia said. “Only about a quarter of them express PD-L1, probably reflective of the fact that there are very few tumor-infiltrating lymphocytes generating gamma-interferon, and inducing this molecule.”
Combination therapy beats single agent
Nivolumab as a single agent had activity, showing a 10% objective response rate, which was nearly the same whether the tumors were platinum sensitive or platinum resistant. “The response rates were doubled when we treated the patients with combination therapy,” Dr. Antonia said, to 23% in the Niv1/Ipi3 arm and to 19% in the Niv3/Ipi1 arm. He said while it is still early to tell, the responses appear to have similar characteristics to what has been seen with non–small cell lung cancer, that is, even patients with bulky disease can have responses.
Only two complete responses occurred, and those were among the 61 patients in the Niv1/Ipi3 arm. All the rest of the responses were either partial or were stable disease. Most responses were rapid, occurring in the first 6-12 weeks. In many of the cases, responses were durable out past 1 year. A few rare cases of pseudoprogression were seen. “Responsiveness did not require PD-L1 expression. … The PD-L1–negative patients responded just as well as the PD-L1–positive patients,” Dr. Antonia said.
The greatest number of patients with tumor regression from baseline occurred in the Niv1/Ipi3 arm, contributing to the investigators’ decision to choose this combination to move forward into phase III trials. Again, while it is still early, the Niv1/Ipi3 combination produced the best overall survival, with a median of 7.7 months and a 1-year overall survival rate of 43%, with a median follow-up of 16.5 months.
Efficacy comes at a cost of toxicity
Toxicity of the treatments was consistent with what has been seen using these drugs with other cancers. Toxicity was greatest in the drug combination arms. For Niv3, 53% of patients had any grade of a treatment related adverse effect (AE). Grade 3/4 AEs affected 13%. For the arms combining nivolumab with ipilimumab, total AEs were in the 74%-79% range, with grade 3/4 AEs affecting 19%-30%. The most common AEs, largely grade 1/2, were fatigue, pruritus, and diarrhea, which were more prevalent when ipilimumab was used. Interestingly, with the higher dose of ipilimumab, 16% of patients experienced low-grade hypothyroidism, and 11% experienced low-grade hyperthyroidism. Treatment-related AEs leading to discontinuation occurred in 6% of the Niv3 arm, 18% of the Niv1/Ipi3 arm, and 13% of the Niv3/Ipi1 arm. Three treatment-related deaths occurred, all among the 115 patients in the combination arms. Dr. Antonia said even grade 3/4 immune-related AEs were well managed using established safety guidelines.
Trial results were simultaneously published in the Lancet Oncology (2016 Jun 3. pii: S1470-2045[16]30098-5. doi: 10.1016/S1470-2045[16]30098-5).
This study is being expanded to include more patients, and nivolumab alone and Niv1/Ipi3 are being tested earlier in the disease as front-line consolidation or maintenance therapy just after platinum-based therapy.
Discussant Dr. Jedd Wolchok of Memorial Sloan Kettering Cancer Center, New York, said people working in immunotherapy feel like they have been riding a wave over the past few years and have just washed up on a beach, wondering where to go from here. “I think combination therapies have really announced themselves as being the next step forward,” he said. Early studies with checkpoint inhibitors produced some strong and durable remissions, but for the most part, in only a minority of patients.
So investigators started to try checkpoint inhibitors with nonredundant targets, as Dr. Antonia’s study did. Nivolumab + ipilimumab has been tried in melanoma, but grade 3/4 toxicity was more prevalent than in this SCLC trial. Dr. Wolchok suggested maybe SCLC patients are more immunosuppressed and therefore do not react to the combination treatment as strongly. The toxicity was lower even than that seen in non-SCLC, “so even in diseases that occur at the same organ site, there is a different biology in the microenvironment that is leading to different tolerability, different degree of immune suppression,” he said.
He congratulated the Checkmate 032 investigators and sponsors for testing different doses and schedules “because this is not one-size-fits-all.” But he said more patients have to be studied. And remaining questions concern the nature of the response, that is, is the response deeper with the combination therapy, and balanced against the additional toxicity?
He noted that not all patients in Dr. Antonia’s study had fine needle aspirates, so their tumors could not be evaluated for PD-L1 expression. So it is still not entirely clear if a subset of patients could benefit the most, such as ones with higher expression.
CHICAGO – A combination of checkpoint inhibitors nivolumab plus ipilimumab produced superior objective response rates, progression-free survival, and overall survival, compared with nivolumab alone for previously treated patients with small cell lung cancer but at a cost of added toxicity, according to a phase I/II study presented at the annual meeting of the American Society of Clinical Oncology.
The observed responses were independent of tumor platinum sensitivity and programmed death-ligand 1 (PD-L1) expression.
Nivolumab as a single agent had activity, showing a 10% objective response rate, which was nearly the same whether the tumors were platinum sensitive or platinum resistant. However, the response rates were doubled for patients treated with combination therapy, reported Dr. Scott Antonia of the H. Lee Moffitt Cancer Center & Research Institute in Tampa.
The phase I/II Checkmate 032 enrolled 216 patients with progressive small cell lung cancer (SCLC) after one or more prior lines of therapy including a first-line platinum-based regimen. Patients were not selected based on PD-L1 expression in their tumors. They were assigned to treatment with nivolumab 3 mg/kg intravenously (IV) every 2 weeks (Niv3; n = 98), to nivolumab 1 mg/kg + ipilimumab 3 mg/kg IV every 3 weeks for four cycles (Niv1/Ipi3; n = 61), or to nivolumab 3 mg/kg + ipilimumab 1 mg/kg IV every 3 weeks for four cycles (Niv3/Ipi1; n = 54). All patients then received nivolumab 3 mg/kg IV every 2 weeks.
The arms of the trial were well matched for age, sex, race, number of prior treatment regimens, smoking history, and tumor PD-L1 level of expression. “The interesting thing with small cell is that very few of the patients actually have tumors that express PD-L1,” Dr. Antonia said. “Only about a quarter of them express PD-L1, probably reflective of the fact that there are very few tumor-infiltrating lymphocytes generating gamma-interferon, and inducing this molecule.”
Combination therapy beats single agent
Nivolumab as a single agent had activity, showing a 10% objective response rate, which was nearly the same whether the tumors were platinum sensitive or platinum resistant. “The response rates were doubled when we treated the patients with combination therapy,” Dr. Antonia said, to 23% in the Niv1/Ipi3 arm and to 19% in the Niv3/Ipi1 arm. He said while it is still early to tell, the responses appear to have similar characteristics to what has been seen with non–small cell lung cancer, that is, even patients with bulky disease can have responses.
Only two complete responses occurred, and those were among the 61 patients in the Niv1/Ipi3 arm. All the rest of the responses were either partial or were stable disease. Most responses were rapid, occurring in the first 6-12 weeks. In many of the cases, responses were durable out past 1 year. A few rare cases of pseudoprogression were seen. “Responsiveness did not require PD-L1 expression. … The PD-L1–negative patients responded just as well as the PD-L1–positive patients,” Dr. Antonia said.
The greatest number of patients with tumor regression from baseline occurred in the Niv1/Ipi3 arm, contributing to the investigators’ decision to choose this combination to move forward into phase III trials. Again, while it is still early, the Niv1/Ipi3 combination produced the best overall survival, with a median of 7.7 months and a 1-year overall survival rate of 43%, with a median follow-up of 16.5 months.
Efficacy comes at a cost of toxicity
Toxicity of the treatments was consistent with what has been seen using these drugs with other cancers. Toxicity was greatest in the drug combination arms. For Niv3, 53% of patients had any grade of a treatment related adverse effect (AE). Grade 3/4 AEs affected 13%. For the arms combining nivolumab with ipilimumab, total AEs were in the 74%-79% range, with grade 3/4 AEs affecting 19%-30%. The most common AEs, largely grade 1/2, were fatigue, pruritus, and diarrhea, which were more prevalent when ipilimumab was used. Interestingly, with the higher dose of ipilimumab, 16% of patients experienced low-grade hypothyroidism, and 11% experienced low-grade hyperthyroidism. Treatment-related AEs leading to discontinuation occurred in 6% of the Niv3 arm, 18% of the Niv1/Ipi3 arm, and 13% of the Niv3/Ipi1 arm. Three treatment-related deaths occurred, all among the 115 patients in the combination arms. Dr. Antonia said even grade 3/4 immune-related AEs were well managed using established safety guidelines.
Trial results were simultaneously published in the Lancet Oncology (2016 Jun 3. pii: S1470-2045[16]30098-5. doi: 10.1016/S1470-2045[16]30098-5).
This study is being expanded to include more patients, and nivolumab alone and Niv1/Ipi3 are being tested earlier in the disease as front-line consolidation or maintenance therapy just after platinum-based therapy.
Discussant Dr. Jedd Wolchok of Memorial Sloan Kettering Cancer Center, New York, said people working in immunotherapy feel like they have been riding a wave over the past few years and have just washed up on a beach, wondering where to go from here. “I think combination therapies have really announced themselves as being the next step forward,” he said. Early studies with checkpoint inhibitors produced some strong and durable remissions, but for the most part, in only a minority of patients.
So investigators started to try checkpoint inhibitors with nonredundant targets, as Dr. Antonia’s study did. Nivolumab + ipilimumab has been tried in melanoma, but grade 3/4 toxicity was more prevalent than in this SCLC trial. Dr. Wolchok suggested maybe SCLC patients are more immunosuppressed and therefore do not react to the combination treatment as strongly. The toxicity was lower even than that seen in non-SCLC, “so even in diseases that occur at the same organ site, there is a different biology in the microenvironment that is leading to different tolerability, different degree of immune suppression,” he said.
He congratulated the Checkmate 032 investigators and sponsors for testing different doses and schedules “because this is not one-size-fits-all.” But he said more patients have to be studied. And remaining questions concern the nature of the response, that is, is the response deeper with the combination therapy, and balanced against the additional toxicity?
He noted that not all patients in Dr. Antonia’s study had fine needle aspirates, so their tumors could not be evaluated for PD-L1 expression. So it is still not entirely clear if a subset of patients could benefit the most, such as ones with higher expression.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Adding ipilimumab to nivolumab resulted in better responses in SCLC than nivolumab alone.
Major finding: Among 216 patients, response rates doubled from 10% to 19% and 23%, depending on dose.
Data source: Phase I/II open label trial of 216 patients with relapsed small cell lung cancer.
Disclosures: The study was sponsored by Bristol-Myers Squibb. Also contributing were Ono Pharmacuetical and Dako. Dr. Antonia and Dr. Wolchok disclosed ties to Bristol-Myers Squibb and several other pharmaceuticals companies.
Web app boosts lung cancer survival
CHICAGO – A simple Web-based mobile application (web-app) improved survival time and quality of life of patients with advanced lung cancer, according to a randomized study presented at the annual meeting of the American Society of Clinical Oncology.
The study was stopped at the planned interim survival analysis that occurred after 121 evaluable patients because of survival benefit favoring the web-app arm. The application, called Moovcare, allowed patients to report symptoms over time and stay in close touch with their care providers after their initial surgery, chemotherapy, or radiation therapy.
“The 1-year survival was 75% in the Moovcare vs. 49% in the control arm,” said lead author Dr. Fabrice Denis of the Institut Inter-régional de Cancérologie Jean Bernard in LeMans, France, in a press conference.
Dr. Denis identified several reasons why a web-app could be useful in treating patients with lung cancer. Even with more than 1 million lung cancer deaths a year worldwide, there is no standard follow-up, and relapses do not occur on a 3 or 6-month schedule of planned visits. So patients often wait several weeks until their next visit to report symptoms indicative of a relapse. They may also be reluctant to report symptoms because of shame over how they contracted the disease, for example, from smoking. And patients are often hesitant to “bother” the doctor with symptoms between visits. All these reasons can contribute to suboptimal therapy and worse outcomes.
Investigators designed Moovcare to allow patients to report symptoms weekly, facilitating early detection of relapse or dangerous medical conditions and triggering early supportive care. They compared the web-app to a control of usual, nonpersonalized follow-up in a French multicenter prospective, randomized trial.
Patients (n = 121) with stage II/node-positive to stage IV (90% stage III/IV) nonprogressive small cell or non–small cell lung cancer were randomly assigned 1:1 to the two arms of the trial. They had to have Internet access, prior experience with email, performance status of 0-2, and an initial symptom score less than 7. Patients could be taking tyrosine kinase inhibitors or on maintenance therapy. Monitoring visits were the same for both groups every 3 months or more frequently. Patients in the control arm received more frequent computed tomographic (CT) imaging than did ones in the web-app arm, and CT scans could be performed at any time in either group based on the investigator’s clinical judgment, or in the case of the web-app, as suggested by patient report in the algorithm.
The median follow up was 9 months. Relapse rates were close to 50% for both groups. The 1-year survival of 75% in the Moovcare to 49% in the control arm gave a 1-year absolute survival increase of 26%. Median survival was 19 months vs. 12 months, a 7-month improvement in median survival for the Moovcare arm. The hazard ratio for death in the web-app arm, compared with the control arm was 0.325 (95% confidence interval, 0.157-0.672; P = .0025).
When they relapsed, 77% of patients in the web-app arm had a good performance status, compared with 33% in the control arm. “This led to 74% of patients receiving optimal therapy in the Moovcare arm vs. 33% in the control arm,” Dr. Denis said. “And the number of imaging [procedures] was reduced by 50% per patient per year.”
Overall quality of life was better in the web-app arm, as assessed using standard quality of life questionnaires.
Moovcare works by having patients or their relatives report 12 symptoms weekly (for example, asthenia, cough, dyspnea, anorexia, etc.) using a smartphone, tablet, or computer. An algorithm analyzes an association of symptoms and triggers email alerts to health care providers if relapse or dangerous medical conditions may be occurring. Providers follow up alerts by phone and schedule visits and imaging. “The sensitivity of the algorithm was high and was validated in two prospective studies,” Dr. Denis said. Sensitivity was 86%-100%.
Moovcare allowed earlier detection of relapse and improved overall survival for three reasons. “It allowed higher performance status at relapse, leading to more optimal therapy for relapsing patients. Dangerous medical conditions were detected earlier and treated earlier. It favored earlier supportive care, which improved quality of life. Less imaging was needed and performed at the right time,” Dr. Denis said.
Patients were monitored on a weekly basis, allowing more personalized care. The Moovcare web-app has been evaluated prospectively in about 300 patients, providing a high level of evidence of its utility in improving outcomes for patients with advanced lung cancer.
Press conference moderator Dr. Patricia Ganz commented that Moovcare is an example of a new way to improve the delivery of high-quality care to patients. “If we had a drug or some new intervention that caused this level of survival benefit, wouldn’t we want to go out and use it?” she asked. “This is a tremendous advance. This is personalized medicine. This is really tailoring it to the patient, and you can see how simple it is to collect this kind of data from the patient and then bring them in in between what would have been a scheduled visit.” She said the app overcomes the barrier of patients putting off reporting symptoms until their next visit or their reluctance to “bother the doctor.”
She said the app alerts the health care team to potential problems and prompts them to “use tests when appropriate, not on a schedule, [which] leads to avoidance of waste in the follow-up of care of our patients.”
CHICAGO – A simple Web-based mobile application (web-app) improved survival time and quality of life of patients with advanced lung cancer, according to a randomized study presented at the annual meeting of the American Society of Clinical Oncology.
The study was stopped at the planned interim survival analysis that occurred after 121 evaluable patients because of survival benefit favoring the web-app arm. The application, called Moovcare, allowed patients to report symptoms over time and stay in close touch with their care providers after their initial surgery, chemotherapy, or radiation therapy.
“The 1-year survival was 75% in the Moovcare vs. 49% in the control arm,” said lead author Dr. Fabrice Denis of the Institut Inter-régional de Cancérologie Jean Bernard in LeMans, France, in a press conference.
Dr. Denis identified several reasons why a web-app could be useful in treating patients with lung cancer. Even with more than 1 million lung cancer deaths a year worldwide, there is no standard follow-up, and relapses do not occur on a 3 or 6-month schedule of planned visits. So patients often wait several weeks until their next visit to report symptoms indicative of a relapse. They may also be reluctant to report symptoms because of shame over how they contracted the disease, for example, from smoking. And patients are often hesitant to “bother” the doctor with symptoms between visits. All these reasons can contribute to suboptimal therapy and worse outcomes.
Investigators designed Moovcare to allow patients to report symptoms weekly, facilitating early detection of relapse or dangerous medical conditions and triggering early supportive care. They compared the web-app to a control of usual, nonpersonalized follow-up in a French multicenter prospective, randomized trial.
Patients (n = 121) with stage II/node-positive to stage IV (90% stage III/IV) nonprogressive small cell or non–small cell lung cancer were randomly assigned 1:1 to the two arms of the trial. They had to have Internet access, prior experience with email, performance status of 0-2, and an initial symptom score less than 7. Patients could be taking tyrosine kinase inhibitors or on maintenance therapy. Monitoring visits were the same for both groups every 3 months or more frequently. Patients in the control arm received more frequent computed tomographic (CT) imaging than did ones in the web-app arm, and CT scans could be performed at any time in either group based on the investigator’s clinical judgment, or in the case of the web-app, as suggested by patient report in the algorithm.
The median follow up was 9 months. Relapse rates were close to 50% for both groups. The 1-year survival of 75% in the Moovcare to 49% in the control arm gave a 1-year absolute survival increase of 26%. Median survival was 19 months vs. 12 months, a 7-month improvement in median survival for the Moovcare arm. The hazard ratio for death in the web-app arm, compared with the control arm was 0.325 (95% confidence interval, 0.157-0.672; P = .0025).
When they relapsed, 77% of patients in the web-app arm had a good performance status, compared with 33% in the control arm. “This led to 74% of patients receiving optimal therapy in the Moovcare arm vs. 33% in the control arm,” Dr. Denis said. “And the number of imaging [procedures] was reduced by 50% per patient per year.”
Overall quality of life was better in the web-app arm, as assessed using standard quality of life questionnaires.
Moovcare works by having patients or their relatives report 12 symptoms weekly (for example, asthenia, cough, dyspnea, anorexia, etc.) using a smartphone, tablet, or computer. An algorithm analyzes an association of symptoms and triggers email alerts to health care providers if relapse or dangerous medical conditions may be occurring. Providers follow up alerts by phone and schedule visits and imaging. “The sensitivity of the algorithm was high and was validated in two prospective studies,” Dr. Denis said. Sensitivity was 86%-100%.
Moovcare allowed earlier detection of relapse and improved overall survival for three reasons. “It allowed higher performance status at relapse, leading to more optimal therapy for relapsing patients. Dangerous medical conditions were detected earlier and treated earlier. It favored earlier supportive care, which improved quality of life. Less imaging was needed and performed at the right time,” Dr. Denis said.
Patients were monitored on a weekly basis, allowing more personalized care. The Moovcare web-app has been evaluated prospectively in about 300 patients, providing a high level of evidence of its utility in improving outcomes for patients with advanced lung cancer.
Press conference moderator Dr. Patricia Ganz commented that Moovcare is an example of a new way to improve the delivery of high-quality care to patients. “If we had a drug or some new intervention that caused this level of survival benefit, wouldn’t we want to go out and use it?” she asked. “This is a tremendous advance. This is personalized medicine. This is really tailoring it to the patient, and you can see how simple it is to collect this kind of data from the patient and then bring them in in between what would have been a scheduled visit.” She said the app overcomes the barrier of patients putting off reporting symptoms until their next visit or their reluctance to “bother the doctor.”
She said the app alerts the health care team to potential problems and prompts them to “use tests when appropriate, not on a schedule, [which] leads to avoidance of waste in the follow-up of care of our patients.”
CHICAGO – A simple Web-based mobile application (web-app) improved survival time and quality of life of patients with advanced lung cancer, according to a randomized study presented at the annual meeting of the American Society of Clinical Oncology.
The study was stopped at the planned interim survival analysis that occurred after 121 evaluable patients because of survival benefit favoring the web-app arm. The application, called Moovcare, allowed patients to report symptoms over time and stay in close touch with their care providers after their initial surgery, chemotherapy, or radiation therapy.
“The 1-year survival was 75% in the Moovcare vs. 49% in the control arm,” said lead author Dr. Fabrice Denis of the Institut Inter-régional de Cancérologie Jean Bernard in LeMans, France, in a press conference.
Dr. Denis identified several reasons why a web-app could be useful in treating patients with lung cancer. Even with more than 1 million lung cancer deaths a year worldwide, there is no standard follow-up, and relapses do not occur on a 3 or 6-month schedule of planned visits. So patients often wait several weeks until their next visit to report symptoms indicative of a relapse. They may also be reluctant to report symptoms because of shame over how they contracted the disease, for example, from smoking. And patients are often hesitant to “bother” the doctor with symptoms between visits. All these reasons can contribute to suboptimal therapy and worse outcomes.
Investigators designed Moovcare to allow patients to report symptoms weekly, facilitating early detection of relapse or dangerous medical conditions and triggering early supportive care. They compared the web-app to a control of usual, nonpersonalized follow-up in a French multicenter prospective, randomized trial.
Patients (n = 121) with stage II/node-positive to stage IV (90% stage III/IV) nonprogressive small cell or non–small cell lung cancer were randomly assigned 1:1 to the two arms of the trial. They had to have Internet access, prior experience with email, performance status of 0-2, and an initial symptom score less than 7. Patients could be taking tyrosine kinase inhibitors or on maintenance therapy. Monitoring visits were the same for both groups every 3 months or more frequently. Patients in the control arm received more frequent computed tomographic (CT) imaging than did ones in the web-app arm, and CT scans could be performed at any time in either group based on the investigator’s clinical judgment, or in the case of the web-app, as suggested by patient report in the algorithm.
The median follow up was 9 months. Relapse rates were close to 50% for both groups. The 1-year survival of 75% in the Moovcare to 49% in the control arm gave a 1-year absolute survival increase of 26%. Median survival was 19 months vs. 12 months, a 7-month improvement in median survival for the Moovcare arm. The hazard ratio for death in the web-app arm, compared with the control arm was 0.325 (95% confidence interval, 0.157-0.672; P = .0025).
When they relapsed, 77% of patients in the web-app arm had a good performance status, compared with 33% in the control arm. “This led to 74% of patients receiving optimal therapy in the Moovcare arm vs. 33% in the control arm,” Dr. Denis said. “And the number of imaging [procedures] was reduced by 50% per patient per year.”
Overall quality of life was better in the web-app arm, as assessed using standard quality of life questionnaires.
Moovcare works by having patients or their relatives report 12 symptoms weekly (for example, asthenia, cough, dyspnea, anorexia, etc.) using a smartphone, tablet, or computer. An algorithm analyzes an association of symptoms and triggers email alerts to health care providers if relapse or dangerous medical conditions may be occurring. Providers follow up alerts by phone and schedule visits and imaging. “The sensitivity of the algorithm was high and was validated in two prospective studies,” Dr. Denis said. Sensitivity was 86%-100%.
Moovcare allowed earlier detection of relapse and improved overall survival for three reasons. “It allowed higher performance status at relapse, leading to more optimal therapy for relapsing patients. Dangerous medical conditions were detected earlier and treated earlier. It favored earlier supportive care, which improved quality of life. Less imaging was needed and performed at the right time,” Dr. Denis said.
Patients were monitored on a weekly basis, allowing more personalized care. The Moovcare web-app has been evaluated prospectively in about 300 patients, providing a high level of evidence of its utility in improving outcomes for patients with advanced lung cancer.
Press conference moderator Dr. Patricia Ganz commented that Moovcare is an example of a new way to improve the delivery of high-quality care to patients. “If we had a drug or some new intervention that caused this level of survival benefit, wouldn’t we want to go out and use it?” she asked. “This is a tremendous advance. This is personalized medicine. This is really tailoring it to the patient, and you can see how simple it is to collect this kind of data from the patient and then bring them in in between what would have been a scheduled visit.” She said the app overcomes the barrier of patients putting off reporting symptoms until their next visit or their reluctance to “bother the doctor.”
She said the app alerts the health care team to potential problems and prompts them to “use tests when appropriate, not on a schedule, [which] leads to avoidance of waste in the follow-up of care of our patients.”
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: A Web-based app improves survival for advanced lung cancer patients.
Major finding: Survival improved by 26% for web-app patients vs. controls.
Data source: Multicenter, prospective, phase III, randomized trial of 121 patients.
Disclosures: Dr. Denis has received honoraria and expenses from several pharmaceutical companies and has received institutional research funding from Sivan. Dr. Patricia Ganz reported stock and other ownership interest in Abbott Laboratories, GlaxoSmithKline, Johnson & Johnson, Merck, Novartis, Pfizer, and Teva.
Vandetanib shows variable response, toxicity in RET-positive NSCLC
CHICAGO – The RET tyrosine kinase inhibitor vandetanib shows marked yet variable antitumor activity and toxicity in patients whose RET-positive non–small cell lung cancer (NSCLC) was unsuccessfully treated with chemotherapy, according to results of two small phase II trials presented at the annual meeting of the American Society of Clinical Oncology.
The overall response rates were 53% and 61% in two independent trials conducted by Dr. Takashi Seto of the National Kyushu Cancer Center, Japan, and by Dr. Se-Hoon Lee of Sungkyunkwan University, South Korea, respectively.
Compared to previous studies with other RET inhibitors, though all had small cohorts, there were many similarities in response rates. Progression-free survival was the most variable, but was much higher in the cabozantinib data reported at the 2015 ASCO annual meeting, according to moderator Dr. Karen Reckamp of the City of Hope Comprehensive Cancer Center, Duarte, Calif. “Vandetanib may have lower response rates than some of the others,” she said.
RET is a tyrosine kinase domain that fuses to and undergoes rearrangements with KIF5B and CCDC6 genes. This fusion and subsequent rearrangement results in ligand-dependent dimerization, which causes tumor growth. RET rearrangements were first identified in thyroid cancers.
“RET fusions were identified as new driver oncogenes of NSCLC in 2012 and observed in 1%-2% of all NSCLC,” Dr. Seto said. “Non–small cell lung cancer with RET rearrangement is regarded as a unique entity in terms of pathogenesis,” said Dr. Lee. There are currently multiple RET inhibitors in various stages of development.
In the trial headed by Dr. Seto, the Japanese genetic screening network was utilized to identify 34 NSCLC patients with RET rearrangements. Among the 34 patients, 17 met the eligibility requirements of having failed at least one prior chemotherapy treatment. Of those 17 patients, the median age was 59 years, 74% were female, all had adenocarcinomas, and 68% were nonsmokers.
All 17 patients received vandetanib at a dose of 300 mg once daily. The overall response rate was 53% (90% confidence interval, 31-74), and the disease control rate was 88%. The median progression-free survival was 4.7 months (90% CI, 2.8-8.3).
There was a marked difference in overall response rate and progression-free survival among RET fusion subtypes. For CCDC6-RET, the overall response was 83% and the progression-free survival was 8.3 months. For KIF5B-RET, the overall response was 20% with a median progression-free survival of 2.9 months.
Dr. Seto noted that there was no known biological explanation for the observed discrepancy in response rate or survival.
Dr. Seto reported that the safety profile was similar to previous reports. Four patients ended treatment due to adverse events while 16 experienced dose interruptions due to treatment-related toxicities. The most common grade three and four toxicities were hypertension (58%), rash (16%), and diarrhea (11%).
In the trial headed by Dr. Lee, 18 patients with RET rearrangements (confirmed by fluorescent in situ hybridization) met the eligibility requirement of having failed platinum-based chemotherapy. The median age of the cohort was 55 years, and 33% were female.
Similar to Dr. Seto’s study, all 18 patients received vandetanib at 300 mg once daily. Of the 18 patient cohort, 17% achieved partial remission and 44% achieved stable disease. Seven patients had no remission or stabilization. There were no treatment-related mortalities or grade 4 adverse events. Two grade 3 adverse events were reported.
“Looking at these two studies together, I think the important thing about the characteristics you see [is] that the age range is very similar [with a] median in the 50’s,” commented Dr. Reckamp. “The male to female ratio is actually opposite in both so [this] can occur in both men and women. The smoking status, interestingly is similar in both, where about a third of patients were former smokers. Most of the patients had adenocarcinoma. Many of these patients were highly previously treated. Only the Seto group looked at RET fusion partner, which may be important in looking at efficacy for these agents.” Vandetanib is a “challenging drug to tolerate,” Dr. Reckamp also noted.
“Is there a preferred RET inhibitor in small cell lung cancer?” Dr. Reckamp asked. “There are many RET inhibitors approved for other cancer types at this point, and they are multitargeted tyrosine kinases. In small studies they have similar efficacy. Toxicities vary because of the off-target effects, and most of the [treatment] decisions were made based on potential toxicities rather than differing efficacy. So none is really differentiated as the best choice, and it is unlikely that we are going to have the trials to evaluate them head to head.”
Dr. Reckamp suggested that “most patients with adenocarcinoma” should be tested for RET rearrangements. “Both men and women, both smokers and nonsmokers. And if you don’t test, you won’t find it. And if you don’t find it, you won’t be able to treat.” Dr. Reckamp also noted that next generation sequencing (NGS) should be the preferred method of identifying RET status because NGS uses less tissue, provides genetic sequencing, and allows for the identification of binding partners.
“Does targeting RET improve patient outcomes?” she continued. “Because that is really the question we need to answer if we need to move forward with RET inhibition for lung cancer. So there are variable response rates, less than other inhibitors of other oncogenic-driven tumors, that’s for sure... But if you look at the data, and these are [for] heavily pretreated patients for the most part, the response rates are better than second-line cytotoxic chemotherapy that we have had in the past and similar to unselected checkpoint inhibition. So there is potential for improving outcomes, and again if we don’t know someone is RET, we potentially are not going to offer a treatment that could help them live longer or better.”
When asked how she would treat a newly-diagnosed patient with RET-positive NSCLC, Dr. Reckamp said she would treat with first-line chemotherapy rather than a tyrosine kinase inhibitor (TKI) but would enroll the patient in whatever TKI trial was ongoing at that point in time.
“There are multiple trials that are ongoing,” Dr. Reckamp said. “It is unlikely that a comparison trial will be completed and so we are going to have to look at these trials next to each other and differentiate based on toxicity [and] perceived efficacy.”
Specifically, Dr. Reckamp believes the medical community needs to move toward “universal testing” for RET status in lung cancer patients. Resistance and combination therapies will also need to be assessed in future studies.
“RET is important in lung cancer, and should be targeted. We now need to find the best way to do that,” Dr. Reckamp concluded.
The trial headed by Dr. Seto was funded by the Japan Agency for Medical Research and Development, AMED, and AstraZeneca. Dr. Seto reported receiving honoraria and research funding from multiple companies including AstraZeneca. The trial headed by Dr. Lee was funded by AstraZeneca Korea. Dr. Lee reported having a consulting or advisory role and receiving honoraria and research funding from AstraZeneca, Pfizer, and Roche/Genentech.
CHICAGO – The RET tyrosine kinase inhibitor vandetanib shows marked yet variable antitumor activity and toxicity in patients whose RET-positive non–small cell lung cancer (NSCLC) was unsuccessfully treated with chemotherapy, according to results of two small phase II trials presented at the annual meeting of the American Society of Clinical Oncology.
The overall response rates were 53% and 61% in two independent trials conducted by Dr. Takashi Seto of the National Kyushu Cancer Center, Japan, and by Dr. Se-Hoon Lee of Sungkyunkwan University, South Korea, respectively.
Compared to previous studies with other RET inhibitors, though all had small cohorts, there were many similarities in response rates. Progression-free survival was the most variable, but was much higher in the cabozantinib data reported at the 2015 ASCO annual meeting, according to moderator Dr. Karen Reckamp of the City of Hope Comprehensive Cancer Center, Duarte, Calif. “Vandetanib may have lower response rates than some of the others,” she said.
RET is a tyrosine kinase domain that fuses to and undergoes rearrangements with KIF5B and CCDC6 genes. This fusion and subsequent rearrangement results in ligand-dependent dimerization, which causes tumor growth. RET rearrangements were first identified in thyroid cancers.
“RET fusions were identified as new driver oncogenes of NSCLC in 2012 and observed in 1%-2% of all NSCLC,” Dr. Seto said. “Non–small cell lung cancer with RET rearrangement is regarded as a unique entity in terms of pathogenesis,” said Dr. Lee. There are currently multiple RET inhibitors in various stages of development.
In the trial headed by Dr. Seto, the Japanese genetic screening network was utilized to identify 34 NSCLC patients with RET rearrangements. Among the 34 patients, 17 met the eligibility requirements of having failed at least one prior chemotherapy treatment. Of those 17 patients, the median age was 59 years, 74% were female, all had adenocarcinomas, and 68% were nonsmokers.
All 17 patients received vandetanib at a dose of 300 mg once daily. The overall response rate was 53% (90% confidence interval, 31-74), and the disease control rate was 88%. The median progression-free survival was 4.7 months (90% CI, 2.8-8.3).
There was a marked difference in overall response rate and progression-free survival among RET fusion subtypes. For CCDC6-RET, the overall response was 83% and the progression-free survival was 8.3 months. For KIF5B-RET, the overall response was 20% with a median progression-free survival of 2.9 months.
Dr. Seto noted that there was no known biological explanation for the observed discrepancy in response rate or survival.
Dr. Seto reported that the safety profile was similar to previous reports. Four patients ended treatment due to adverse events while 16 experienced dose interruptions due to treatment-related toxicities. The most common grade three and four toxicities were hypertension (58%), rash (16%), and diarrhea (11%).
In the trial headed by Dr. Lee, 18 patients with RET rearrangements (confirmed by fluorescent in situ hybridization) met the eligibility requirement of having failed platinum-based chemotherapy. The median age of the cohort was 55 years, and 33% were female.
Similar to Dr. Seto’s study, all 18 patients received vandetanib at 300 mg once daily. Of the 18 patient cohort, 17% achieved partial remission and 44% achieved stable disease. Seven patients had no remission or stabilization. There were no treatment-related mortalities or grade 4 adverse events. Two grade 3 adverse events were reported.
“Looking at these two studies together, I think the important thing about the characteristics you see [is] that the age range is very similar [with a] median in the 50’s,” commented Dr. Reckamp. “The male to female ratio is actually opposite in both so [this] can occur in both men and women. The smoking status, interestingly is similar in both, where about a third of patients were former smokers. Most of the patients had adenocarcinoma. Many of these patients were highly previously treated. Only the Seto group looked at RET fusion partner, which may be important in looking at efficacy for these agents.” Vandetanib is a “challenging drug to tolerate,” Dr. Reckamp also noted.
“Is there a preferred RET inhibitor in small cell lung cancer?” Dr. Reckamp asked. “There are many RET inhibitors approved for other cancer types at this point, and they are multitargeted tyrosine kinases. In small studies they have similar efficacy. Toxicities vary because of the off-target effects, and most of the [treatment] decisions were made based on potential toxicities rather than differing efficacy. So none is really differentiated as the best choice, and it is unlikely that we are going to have the trials to evaluate them head to head.”
Dr. Reckamp suggested that “most patients with adenocarcinoma” should be tested for RET rearrangements. “Both men and women, both smokers and nonsmokers. And if you don’t test, you won’t find it. And if you don’t find it, you won’t be able to treat.” Dr. Reckamp also noted that next generation sequencing (NGS) should be the preferred method of identifying RET status because NGS uses less tissue, provides genetic sequencing, and allows for the identification of binding partners.
“Does targeting RET improve patient outcomes?” she continued. “Because that is really the question we need to answer if we need to move forward with RET inhibition for lung cancer. So there are variable response rates, less than other inhibitors of other oncogenic-driven tumors, that’s for sure... But if you look at the data, and these are [for] heavily pretreated patients for the most part, the response rates are better than second-line cytotoxic chemotherapy that we have had in the past and similar to unselected checkpoint inhibition. So there is potential for improving outcomes, and again if we don’t know someone is RET, we potentially are not going to offer a treatment that could help them live longer or better.”
When asked how she would treat a newly-diagnosed patient with RET-positive NSCLC, Dr. Reckamp said she would treat with first-line chemotherapy rather than a tyrosine kinase inhibitor (TKI) but would enroll the patient in whatever TKI trial was ongoing at that point in time.
“There are multiple trials that are ongoing,” Dr. Reckamp said. “It is unlikely that a comparison trial will be completed and so we are going to have to look at these trials next to each other and differentiate based on toxicity [and] perceived efficacy.”
Specifically, Dr. Reckamp believes the medical community needs to move toward “universal testing” for RET status in lung cancer patients. Resistance and combination therapies will also need to be assessed in future studies.
“RET is important in lung cancer, and should be targeted. We now need to find the best way to do that,” Dr. Reckamp concluded.
The trial headed by Dr. Seto was funded by the Japan Agency for Medical Research and Development, AMED, and AstraZeneca. Dr. Seto reported receiving honoraria and research funding from multiple companies including AstraZeneca. The trial headed by Dr. Lee was funded by AstraZeneca Korea. Dr. Lee reported having a consulting or advisory role and receiving honoraria and research funding from AstraZeneca, Pfizer, and Roche/Genentech.
CHICAGO – The RET tyrosine kinase inhibitor vandetanib shows marked yet variable antitumor activity and toxicity in patients whose RET-positive non–small cell lung cancer (NSCLC) was unsuccessfully treated with chemotherapy, according to results of two small phase II trials presented at the annual meeting of the American Society of Clinical Oncology.
The overall response rates were 53% and 61% in two independent trials conducted by Dr. Takashi Seto of the National Kyushu Cancer Center, Japan, and by Dr. Se-Hoon Lee of Sungkyunkwan University, South Korea, respectively.
Compared to previous studies with other RET inhibitors, though all had small cohorts, there were many similarities in response rates. Progression-free survival was the most variable, but was much higher in the cabozantinib data reported at the 2015 ASCO annual meeting, according to moderator Dr. Karen Reckamp of the City of Hope Comprehensive Cancer Center, Duarte, Calif. “Vandetanib may have lower response rates than some of the others,” she said.
RET is a tyrosine kinase domain that fuses to and undergoes rearrangements with KIF5B and CCDC6 genes. This fusion and subsequent rearrangement results in ligand-dependent dimerization, which causes tumor growth. RET rearrangements were first identified in thyroid cancers.
“RET fusions were identified as new driver oncogenes of NSCLC in 2012 and observed in 1%-2% of all NSCLC,” Dr. Seto said. “Non–small cell lung cancer with RET rearrangement is regarded as a unique entity in terms of pathogenesis,” said Dr. Lee. There are currently multiple RET inhibitors in various stages of development.
In the trial headed by Dr. Seto, the Japanese genetic screening network was utilized to identify 34 NSCLC patients with RET rearrangements. Among the 34 patients, 17 met the eligibility requirements of having failed at least one prior chemotherapy treatment. Of those 17 patients, the median age was 59 years, 74% were female, all had adenocarcinomas, and 68% were nonsmokers.
All 17 patients received vandetanib at a dose of 300 mg once daily. The overall response rate was 53% (90% confidence interval, 31-74), and the disease control rate was 88%. The median progression-free survival was 4.7 months (90% CI, 2.8-8.3).
There was a marked difference in overall response rate and progression-free survival among RET fusion subtypes. For CCDC6-RET, the overall response was 83% and the progression-free survival was 8.3 months. For KIF5B-RET, the overall response was 20% with a median progression-free survival of 2.9 months.
Dr. Seto noted that there was no known biological explanation for the observed discrepancy in response rate or survival.
Dr. Seto reported that the safety profile was similar to previous reports. Four patients ended treatment due to adverse events while 16 experienced dose interruptions due to treatment-related toxicities. The most common grade three and four toxicities were hypertension (58%), rash (16%), and diarrhea (11%).
In the trial headed by Dr. Lee, 18 patients with RET rearrangements (confirmed by fluorescent in situ hybridization) met the eligibility requirement of having failed platinum-based chemotherapy. The median age of the cohort was 55 years, and 33% were female.
Similar to Dr. Seto’s study, all 18 patients received vandetanib at 300 mg once daily. Of the 18 patient cohort, 17% achieved partial remission and 44% achieved stable disease. Seven patients had no remission or stabilization. There were no treatment-related mortalities or grade 4 adverse events. Two grade 3 adverse events were reported.
“Looking at these two studies together, I think the important thing about the characteristics you see [is] that the age range is very similar [with a] median in the 50’s,” commented Dr. Reckamp. “The male to female ratio is actually opposite in both so [this] can occur in both men and women. The smoking status, interestingly is similar in both, where about a third of patients were former smokers. Most of the patients had adenocarcinoma. Many of these patients were highly previously treated. Only the Seto group looked at RET fusion partner, which may be important in looking at efficacy for these agents.” Vandetanib is a “challenging drug to tolerate,” Dr. Reckamp also noted.
“Is there a preferred RET inhibitor in small cell lung cancer?” Dr. Reckamp asked. “There are many RET inhibitors approved for other cancer types at this point, and they are multitargeted tyrosine kinases. In small studies they have similar efficacy. Toxicities vary because of the off-target effects, and most of the [treatment] decisions were made based on potential toxicities rather than differing efficacy. So none is really differentiated as the best choice, and it is unlikely that we are going to have the trials to evaluate them head to head.”
Dr. Reckamp suggested that “most patients with adenocarcinoma” should be tested for RET rearrangements. “Both men and women, both smokers and nonsmokers. And if you don’t test, you won’t find it. And if you don’t find it, you won’t be able to treat.” Dr. Reckamp also noted that next generation sequencing (NGS) should be the preferred method of identifying RET status because NGS uses less tissue, provides genetic sequencing, and allows for the identification of binding partners.
“Does targeting RET improve patient outcomes?” she continued. “Because that is really the question we need to answer if we need to move forward with RET inhibition for lung cancer. So there are variable response rates, less than other inhibitors of other oncogenic-driven tumors, that’s for sure... But if you look at the data, and these are [for] heavily pretreated patients for the most part, the response rates are better than second-line cytotoxic chemotherapy that we have had in the past and similar to unselected checkpoint inhibition. So there is potential for improving outcomes, and again if we don’t know someone is RET, we potentially are not going to offer a treatment that could help them live longer or better.”
When asked how she would treat a newly-diagnosed patient with RET-positive NSCLC, Dr. Reckamp said she would treat with first-line chemotherapy rather than a tyrosine kinase inhibitor (TKI) but would enroll the patient in whatever TKI trial was ongoing at that point in time.
“There are multiple trials that are ongoing,” Dr. Reckamp said. “It is unlikely that a comparison trial will be completed and so we are going to have to look at these trials next to each other and differentiate based on toxicity [and] perceived efficacy.”
Specifically, Dr. Reckamp believes the medical community needs to move toward “universal testing” for RET status in lung cancer patients. Resistance and combination therapies will also need to be assessed in future studies.
“RET is important in lung cancer, and should be targeted. We now need to find the best way to do that,” Dr. Reckamp concluded.
The trial headed by Dr. Seto was funded by the Japan Agency for Medical Research and Development, AMED, and AstraZeneca. Dr. Seto reported receiving honoraria and research funding from multiple companies including AstraZeneca. The trial headed by Dr. Lee was funded by AstraZeneca Korea. Dr. Lee reported having a consulting or advisory role and receiving honoraria and research funding from AstraZeneca, Pfizer, and Roche/Genentech.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Vandetanib shows marked yet variable anti-tumor activity and toxicity in patients with RET-rearranged NSCLC.
Major finding: The overall response rates were 53% and 61% in two independent trials.
Data source: Two independently-conducted multicenter phase II trials of patients with RET-rearranged non-small cell lung cancer.
Disclosures: The trial headed by Dr. Seto was funded by the Japan Agency for Medical Research and Development, AMED, and AstraZeneca. Dr. Seto reported receiving honoraria and research funding from multiple companies including AstraZeneca. The trial headed by Dr. Lee was funded by AstraZeneca Korea Ltd. Dr. Lee reported having a consulting or advisory role and receiving honoraria and research funding from AstraZeneca, Pfizer, and Roche/Genentech.
Novel antibody drug conjugate shows promise in SCLC
CHICAGO – The antibody drug conjugate rovalpituzumab tesirine (Rova-T) is active in DLL3-expressing recurrent and refractory small–cell lung cancer, according to results of a small, first-in-human trial.
Nearly a fifth of the 60 evaluable patients studied experienced tumor shrinkage, and 41% achieved clinical benefit, lead author Dr. Charles M. Rudin of Memorial Sloan Kettering Cancer Center, New York, reported at the annual meeting of the American Society of Clinical Oncology.
“We have demonstrated activity in recurrent and refractory small–cell lung cancer and comparable responses in the second- and third-line settings,” he summarized. “Responses and survival numerically improve [upon] historical controls.”
“This was an early but interesting study of a novel antibody drug conjugate demonstrating activity in progressive small–cell lung cancer which is a very aggressive disease with limited treatment options,” commented moderator Dr. Smitha Krishnamurthi of University Hospitals Case Medical Center, Cleveland.
“What we were focusing on was a new target in small–cell lung cancer, which we believe may be a predictive biomarker for therapeutics. DLL3 is expressed in many small–cell lung cancers and is not expressed in any normal adult tissue,” Dr. Rudin explained. Rova-T contains an anti-DLL3 antibody and pyrrolobenzodiazepine, which together selectively kill DLL3-expressing cells. The study focused on safety and efficacy.
Of the 74 patients enrolled in the study, 60 had tumor tissue samples and were selected for further study. DLL3 was detected in 88% of the patients, and 67% had high-level expression, which was defined as having measurable DLL3 in at least 50% of cells.
All patients received Rova-T as a first-, second-, or third-line therapy. Eleven out of the 60 patients experienced tumor shrinkage and 68% achieved clinical benefit. Among the high–DLL3-expressing patients, 89% achieved and maintained stable disease for at least two scans taken every 6 weeks.
Among the 26 patients with the highest levels of DLL3, 39% responded to the drug, while 32% achieved 1-year survival.
The 12 patients who received Rova-T as a third-line therapy responded “particularly well, with 50% having tumor shrinkage,” reported Dr. Rudin.
“Most importantly, this is the first biomarker-directed therapy to be defined for the treatment of small–cell lung cancer, and this is a biomarker that is expressed in a large majority of small–cell lung cancers so a biomarker that may [have] utility for small–cell lung cancer. Small–cell lung cancer is a terrible disease and is really unchanged in clinically meaningful endpoints for patients. ... Median survival [is] 9-10 months from time of diagnosis, and for recurrent disease, there is really one approved drug … so a lot of room for improvement here,” Dr. Rudin said. Although based on a small cohort, Dr. Rudin noted, “these data really look promising to us as a novel therapeutic for small–cell lung cancer.”
“These results clearly justify further clinical development. We are currently enrolling to those studies,” he said referring to TRINITY, a third-line, single-arm study with no comparative drug. TRINITY is currently enrolling patients in multiple centers. There are also several other trials in the works to look at other diseases with DLL3 expression and to assess Rova-T’s efficacy and safety in combination with other drugs.
Dr. Rudin disclosed that he has a consulting or advisory role with AbbVie, AVEO, Boehringer Ingelheim, GlaxoSmithKline, Merck, Celgene, Novartis, and has received funding from Biomarin. This study was funded by Stemcentrx.
CHICAGO – The antibody drug conjugate rovalpituzumab tesirine (Rova-T) is active in DLL3-expressing recurrent and refractory small–cell lung cancer, according to results of a small, first-in-human trial.
Nearly a fifth of the 60 evaluable patients studied experienced tumor shrinkage, and 41% achieved clinical benefit, lead author Dr. Charles M. Rudin of Memorial Sloan Kettering Cancer Center, New York, reported at the annual meeting of the American Society of Clinical Oncology.
“We have demonstrated activity in recurrent and refractory small–cell lung cancer and comparable responses in the second- and third-line settings,” he summarized. “Responses and survival numerically improve [upon] historical controls.”
“This was an early but interesting study of a novel antibody drug conjugate demonstrating activity in progressive small–cell lung cancer which is a very aggressive disease with limited treatment options,” commented moderator Dr. Smitha Krishnamurthi of University Hospitals Case Medical Center, Cleveland.
“What we were focusing on was a new target in small–cell lung cancer, which we believe may be a predictive biomarker for therapeutics. DLL3 is expressed in many small–cell lung cancers and is not expressed in any normal adult tissue,” Dr. Rudin explained. Rova-T contains an anti-DLL3 antibody and pyrrolobenzodiazepine, which together selectively kill DLL3-expressing cells. The study focused on safety and efficacy.
Of the 74 patients enrolled in the study, 60 had tumor tissue samples and were selected for further study. DLL3 was detected in 88% of the patients, and 67% had high-level expression, which was defined as having measurable DLL3 in at least 50% of cells.
All patients received Rova-T as a first-, second-, or third-line therapy. Eleven out of the 60 patients experienced tumor shrinkage and 68% achieved clinical benefit. Among the high–DLL3-expressing patients, 89% achieved and maintained stable disease for at least two scans taken every 6 weeks.
Among the 26 patients with the highest levels of DLL3, 39% responded to the drug, while 32% achieved 1-year survival.
The 12 patients who received Rova-T as a third-line therapy responded “particularly well, with 50% having tumor shrinkage,” reported Dr. Rudin.
“Most importantly, this is the first biomarker-directed therapy to be defined for the treatment of small–cell lung cancer, and this is a biomarker that is expressed in a large majority of small–cell lung cancers so a biomarker that may [have] utility for small–cell lung cancer. Small–cell lung cancer is a terrible disease and is really unchanged in clinically meaningful endpoints for patients. ... Median survival [is] 9-10 months from time of diagnosis, and for recurrent disease, there is really one approved drug … so a lot of room for improvement here,” Dr. Rudin said. Although based on a small cohort, Dr. Rudin noted, “these data really look promising to us as a novel therapeutic for small–cell lung cancer.”
“These results clearly justify further clinical development. We are currently enrolling to those studies,” he said referring to TRINITY, a third-line, single-arm study with no comparative drug. TRINITY is currently enrolling patients in multiple centers. There are also several other trials in the works to look at other diseases with DLL3 expression and to assess Rova-T’s efficacy and safety in combination with other drugs.
Dr. Rudin disclosed that he has a consulting or advisory role with AbbVie, AVEO, Boehringer Ingelheim, GlaxoSmithKline, Merck, Celgene, Novartis, and has received funding from Biomarin. This study was funded by Stemcentrx.
CHICAGO – The antibody drug conjugate rovalpituzumab tesirine (Rova-T) is active in DLL3-expressing recurrent and refractory small–cell lung cancer, according to results of a small, first-in-human trial.
Nearly a fifth of the 60 evaluable patients studied experienced tumor shrinkage, and 41% achieved clinical benefit, lead author Dr. Charles M. Rudin of Memorial Sloan Kettering Cancer Center, New York, reported at the annual meeting of the American Society of Clinical Oncology.
“We have demonstrated activity in recurrent and refractory small–cell lung cancer and comparable responses in the second- and third-line settings,” he summarized. “Responses and survival numerically improve [upon] historical controls.”
“This was an early but interesting study of a novel antibody drug conjugate demonstrating activity in progressive small–cell lung cancer which is a very aggressive disease with limited treatment options,” commented moderator Dr. Smitha Krishnamurthi of University Hospitals Case Medical Center, Cleveland.
“What we were focusing on was a new target in small–cell lung cancer, which we believe may be a predictive biomarker for therapeutics. DLL3 is expressed in many small–cell lung cancers and is not expressed in any normal adult tissue,” Dr. Rudin explained. Rova-T contains an anti-DLL3 antibody and pyrrolobenzodiazepine, which together selectively kill DLL3-expressing cells. The study focused on safety and efficacy.
Of the 74 patients enrolled in the study, 60 had tumor tissue samples and were selected for further study. DLL3 was detected in 88% of the patients, and 67% had high-level expression, which was defined as having measurable DLL3 in at least 50% of cells.
All patients received Rova-T as a first-, second-, or third-line therapy. Eleven out of the 60 patients experienced tumor shrinkage and 68% achieved clinical benefit. Among the high–DLL3-expressing patients, 89% achieved and maintained stable disease for at least two scans taken every 6 weeks.
Among the 26 patients with the highest levels of DLL3, 39% responded to the drug, while 32% achieved 1-year survival.
The 12 patients who received Rova-T as a third-line therapy responded “particularly well, with 50% having tumor shrinkage,” reported Dr. Rudin.
“Most importantly, this is the first biomarker-directed therapy to be defined for the treatment of small–cell lung cancer, and this is a biomarker that is expressed in a large majority of small–cell lung cancers so a biomarker that may [have] utility for small–cell lung cancer. Small–cell lung cancer is a terrible disease and is really unchanged in clinically meaningful endpoints for patients. ... Median survival [is] 9-10 months from time of diagnosis, and for recurrent disease, there is really one approved drug … so a lot of room for improvement here,” Dr. Rudin said. Although based on a small cohort, Dr. Rudin noted, “these data really look promising to us as a novel therapeutic for small–cell lung cancer.”
“These results clearly justify further clinical development. We are currently enrolling to those studies,” he said referring to TRINITY, a third-line, single-arm study with no comparative drug. TRINITY is currently enrolling patients in multiple centers. There are also several other trials in the works to look at other diseases with DLL3 expression and to assess Rova-T’s efficacy and safety in combination with other drugs.
Dr. Rudin disclosed that he has a consulting or advisory role with AbbVie, AVEO, Boehringer Ingelheim, GlaxoSmithKline, Merck, Celgene, Novartis, and has received funding from Biomarin. This study was funded by Stemcentrx.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Rova-T is active in DLL3-expressing recurrent and refractory small–cell lung cancer.
Major finding: The clinical benefit rate of Rova-T was 68% with 32% of patients achieving 1-year survival.
Data source: A first-in-human trial of 74 patients with SCLC.
Disclosures: This study was funded by Stemcentrx. Dr. Rudin disclosed that he has a consulting or advisory role with AbbVie, AVEO, Boehringer Ingelheim, GlaxoSmithKline, Merck, Celgene, Novartis, and has received funding from BioMarin.
MyPathway: Targeted therapies show promise in nonindicated tumors
CHICAGO – Agents that target the HER2, BRAF, Hedgehog, or EGFR pathways show promise in nonindicated tumor types that harbor these molecular alterations, according to early findings from the MyPathway study.
Of 129 patients enrolled in the multicenter, open-label, phase IIa study, 29 had a major response, defined as tumor shrinkage of at least 30%, to such treatment. One of those patients had a complete response, and 28 had a partial response. An additional 40 patients had stable disease on treatment. Fourteen of the 29 patients progressed after a median of 6 months’ follow-up, and 15 responses were ongoing at up to 11 months, Dr. John D. Hainsworth reported at the annual meeting of the American Society of Clinical Oncology.
No new safety signals were observed, said Dr. Hainsworth of Sarah Cannon Research Institute in Nashville, Tenn.
Treatments evaluated in MyPathway included:
• Trastuzumab + pertuzumab, which targets the HER2 pathway and is currently indicated for breast cancer.
• Vemurafenib, which targets the BRAF pathway and is currently indicated for melanoma.
• Vismodegib, which targets the Hedgehog pathway and is currently indicated for basal cell carcinoma of the skin.
• Erlotinib, which targets the EGFR pathway and is indicated for non–small-cell lung cancer.
Responses have been seen with all four of the treatments, but the best responses were seen among patients with HER2 and BRAF abnormalities.
Among 61 cancers with HER2 amplification/overexpression, trastuzumab + pertuzumab provided a benefit for colorectal, bladder, biliary, non–small-cell lung, pancreas, and head/neck cancers.
Of 20 colorectal tumors, 7 (35%) showed complete or partial response, and 3 (15%) remained stable for at least 120 days (clinical benefit rate, 50%). Complete/partial responses and stable disease, respectively, were also seen in three and two of eight bladder tumors (clinical benefit rate, 63%), in three and three of six biliary tumors (clinical benefit rate, 100%), in two and zero of seven non–small-cell lung tumors (clinical benefit rate, 29%), one and zero of six pancreas tumors (clinical benefit rate, 17%), and one and zero of three head and neck tumors (34%). One of 11 other types of tumors showed disease stability at 120 days (clinical benefit rate, 9%). The overall clinical benefit rate in the study was 43%, Dr. Hainsworth said.
Among 33 cancers with the BRAF mutation, vemurafenib showed activity for non–small-cell lung, ovary, unknown primary, colorectal, pancreas, and head/neck tumors. Of 15 non–small-cell lung tumors, 3 (20%) showed complete or partial responses and 2 (13%) remained stable for at least 120 days (clinical benefit rate, 33%). Complete/partial responses and stable disease, respectively, were also seen in one and two of four ovary tumors (clinical benefit rate, 75%), and complete or partial responses were seen in one each of three unknown primary tumors, two colorectal tumors, two pancreas tumors, and one head/neck tumor (clinical benefit rates of 33%, 50%, 50%, and 100%, respectively). No benefit was seen with tumors at other sites (total clinical benefit rate, 36%), Dr. Hainsworth said.
“Of interest in this group [of patients with BRAF mutations], seven of the eight responses were in V600E mutations, and as you know, that’s the mutation that’s been specifically correlated with high response to BRAF inhibition in melanoma where this treatment is now approved,” he said, adding that the response rate in those patients was 38%.
Based on these early results, enrollment of patients with HER2 abnormalities and colorectal, bladder, or biliary cancer, and of patients with BRAF mutations and lung cancer, will be expanded, he said.
Subjects enrolled in MyPathway have advanced cancer showing abnormalities in any of the pathways of interest. The first 129 received a mean of three prior therapies, and in the 29 who responded, 12 different types of cancer responded to the targeted treatment.
“An increasing number of targeted agents for advanced cancer are in use now based on the presence of molecular abnormalities in the cancer. … We’ve known that the same mutations that are in those cancers are found in a wide variety of other cancers, although at a lower incidence, and it’s been difficult to test how effective these same treatments are for the other cancers due to the difficulty in identifying the patient population,” he said, explaining that an increase in comprehensive genomic profiling in recent years has allowed for identification of more and more of these mutations in other cancers.
“I think we’ve shown now that this trial design is feasible, where patients are selected on the basis of molecular abnormalities in their cancers rather than on their primary tumor type or primary site, and certainly offers opportunities for patients with these molecular abnormalities,” Dr. Hainsworth concluded.
Thus far, MyPathway has enrolled more than 200 patients, and is designed to accrue up to 500, with adjustment of treatment groups based on response rates. Emerging new regimens that target these pathways, such as the MEK inhibitor cobemetinib, will also be added, as will new agents targeting additional molecular abnormalities.
The study design, using this “tumor-agnostic approach,” mirrors that of the ASCO-led TAPUR trial, according to ASCO spokesperson Dr. Sumanta Kumar Pal.
The findings of these and other precision medicine trials may ultimately shift the longstanding cancer treatment paradigm, Dr. Pal said.
MyPathway received funding from Genentech. Dr. Hainsworth reported that his institution has received research funding from Astellas Pharma, AstraZeneca, Celgene, Genentech, Johnson & Johnson, Lilly, and Novartis.
CHICAGO – Agents that target the HER2, BRAF, Hedgehog, or EGFR pathways show promise in nonindicated tumor types that harbor these molecular alterations, according to early findings from the MyPathway study.
Of 129 patients enrolled in the multicenter, open-label, phase IIa study, 29 had a major response, defined as tumor shrinkage of at least 30%, to such treatment. One of those patients had a complete response, and 28 had a partial response. An additional 40 patients had stable disease on treatment. Fourteen of the 29 patients progressed after a median of 6 months’ follow-up, and 15 responses were ongoing at up to 11 months, Dr. John D. Hainsworth reported at the annual meeting of the American Society of Clinical Oncology.
No new safety signals were observed, said Dr. Hainsworth of Sarah Cannon Research Institute in Nashville, Tenn.
Treatments evaluated in MyPathway included:
• Trastuzumab + pertuzumab, which targets the HER2 pathway and is currently indicated for breast cancer.
• Vemurafenib, which targets the BRAF pathway and is currently indicated for melanoma.
• Vismodegib, which targets the Hedgehog pathway and is currently indicated for basal cell carcinoma of the skin.
• Erlotinib, which targets the EGFR pathway and is indicated for non–small-cell lung cancer.
Responses have been seen with all four of the treatments, but the best responses were seen among patients with HER2 and BRAF abnormalities.
Among 61 cancers with HER2 amplification/overexpression, trastuzumab + pertuzumab provided a benefit for colorectal, bladder, biliary, non–small-cell lung, pancreas, and head/neck cancers.
Of 20 colorectal tumors, 7 (35%) showed complete or partial response, and 3 (15%) remained stable for at least 120 days (clinical benefit rate, 50%). Complete/partial responses and stable disease, respectively, were also seen in three and two of eight bladder tumors (clinical benefit rate, 63%), in three and three of six biliary tumors (clinical benefit rate, 100%), in two and zero of seven non–small-cell lung tumors (clinical benefit rate, 29%), one and zero of six pancreas tumors (clinical benefit rate, 17%), and one and zero of three head and neck tumors (34%). One of 11 other types of tumors showed disease stability at 120 days (clinical benefit rate, 9%). The overall clinical benefit rate in the study was 43%, Dr. Hainsworth said.
Among 33 cancers with the BRAF mutation, vemurafenib showed activity for non–small-cell lung, ovary, unknown primary, colorectal, pancreas, and head/neck tumors. Of 15 non–small-cell lung tumors, 3 (20%) showed complete or partial responses and 2 (13%) remained stable for at least 120 days (clinical benefit rate, 33%). Complete/partial responses and stable disease, respectively, were also seen in one and two of four ovary tumors (clinical benefit rate, 75%), and complete or partial responses were seen in one each of three unknown primary tumors, two colorectal tumors, two pancreas tumors, and one head/neck tumor (clinical benefit rates of 33%, 50%, 50%, and 100%, respectively). No benefit was seen with tumors at other sites (total clinical benefit rate, 36%), Dr. Hainsworth said.
“Of interest in this group [of patients with BRAF mutations], seven of the eight responses were in V600E mutations, and as you know, that’s the mutation that’s been specifically correlated with high response to BRAF inhibition in melanoma where this treatment is now approved,” he said, adding that the response rate in those patients was 38%.
Based on these early results, enrollment of patients with HER2 abnormalities and colorectal, bladder, or biliary cancer, and of patients with BRAF mutations and lung cancer, will be expanded, he said.
Subjects enrolled in MyPathway have advanced cancer showing abnormalities in any of the pathways of interest. The first 129 received a mean of three prior therapies, and in the 29 who responded, 12 different types of cancer responded to the targeted treatment.
“An increasing number of targeted agents for advanced cancer are in use now based on the presence of molecular abnormalities in the cancer. … We’ve known that the same mutations that are in those cancers are found in a wide variety of other cancers, although at a lower incidence, and it’s been difficult to test how effective these same treatments are for the other cancers due to the difficulty in identifying the patient population,” he said, explaining that an increase in comprehensive genomic profiling in recent years has allowed for identification of more and more of these mutations in other cancers.
“I think we’ve shown now that this trial design is feasible, where patients are selected on the basis of molecular abnormalities in their cancers rather than on their primary tumor type or primary site, and certainly offers opportunities for patients with these molecular abnormalities,” Dr. Hainsworth concluded.
Thus far, MyPathway has enrolled more than 200 patients, and is designed to accrue up to 500, with adjustment of treatment groups based on response rates. Emerging new regimens that target these pathways, such as the MEK inhibitor cobemetinib, will also be added, as will new agents targeting additional molecular abnormalities.
The study design, using this “tumor-agnostic approach,” mirrors that of the ASCO-led TAPUR trial, according to ASCO spokesperson Dr. Sumanta Kumar Pal.
The findings of these and other precision medicine trials may ultimately shift the longstanding cancer treatment paradigm, Dr. Pal said.
MyPathway received funding from Genentech. Dr. Hainsworth reported that his institution has received research funding from Astellas Pharma, AstraZeneca, Celgene, Genentech, Johnson & Johnson, Lilly, and Novartis.
CHICAGO – Agents that target the HER2, BRAF, Hedgehog, or EGFR pathways show promise in nonindicated tumor types that harbor these molecular alterations, according to early findings from the MyPathway study.
Of 129 patients enrolled in the multicenter, open-label, phase IIa study, 29 had a major response, defined as tumor shrinkage of at least 30%, to such treatment. One of those patients had a complete response, and 28 had a partial response. An additional 40 patients had stable disease on treatment. Fourteen of the 29 patients progressed after a median of 6 months’ follow-up, and 15 responses were ongoing at up to 11 months, Dr. John D. Hainsworth reported at the annual meeting of the American Society of Clinical Oncology.
No new safety signals were observed, said Dr. Hainsworth of Sarah Cannon Research Institute in Nashville, Tenn.
Treatments evaluated in MyPathway included:
• Trastuzumab + pertuzumab, which targets the HER2 pathway and is currently indicated for breast cancer.
• Vemurafenib, which targets the BRAF pathway and is currently indicated for melanoma.
• Vismodegib, which targets the Hedgehog pathway and is currently indicated for basal cell carcinoma of the skin.
• Erlotinib, which targets the EGFR pathway and is indicated for non–small-cell lung cancer.
Responses have been seen with all four of the treatments, but the best responses were seen among patients with HER2 and BRAF abnormalities.
Among 61 cancers with HER2 amplification/overexpression, trastuzumab + pertuzumab provided a benefit for colorectal, bladder, biliary, non–small-cell lung, pancreas, and head/neck cancers.
Of 20 colorectal tumors, 7 (35%) showed complete or partial response, and 3 (15%) remained stable for at least 120 days (clinical benefit rate, 50%). Complete/partial responses and stable disease, respectively, were also seen in three and two of eight bladder tumors (clinical benefit rate, 63%), in three and three of six biliary tumors (clinical benefit rate, 100%), in two and zero of seven non–small-cell lung tumors (clinical benefit rate, 29%), one and zero of six pancreas tumors (clinical benefit rate, 17%), and one and zero of three head and neck tumors (34%). One of 11 other types of tumors showed disease stability at 120 days (clinical benefit rate, 9%). The overall clinical benefit rate in the study was 43%, Dr. Hainsworth said.
Among 33 cancers with the BRAF mutation, vemurafenib showed activity for non–small-cell lung, ovary, unknown primary, colorectal, pancreas, and head/neck tumors. Of 15 non–small-cell lung tumors, 3 (20%) showed complete or partial responses and 2 (13%) remained stable for at least 120 days (clinical benefit rate, 33%). Complete/partial responses and stable disease, respectively, were also seen in one and two of four ovary tumors (clinical benefit rate, 75%), and complete or partial responses were seen in one each of three unknown primary tumors, two colorectal tumors, two pancreas tumors, and one head/neck tumor (clinical benefit rates of 33%, 50%, 50%, and 100%, respectively). No benefit was seen with tumors at other sites (total clinical benefit rate, 36%), Dr. Hainsworth said.
“Of interest in this group [of patients with BRAF mutations], seven of the eight responses were in V600E mutations, and as you know, that’s the mutation that’s been specifically correlated with high response to BRAF inhibition in melanoma where this treatment is now approved,” he said, adding that the response rate in those patients was 38%.
Based on these early results, enrollment of patients with HER2 abnormalities and colorectal, bladder, or biliary cancer, and of patients with BRAF mutations and lung cancer, will be expanded, he said.
Subjects enrolled in MyPathway have advanced cancer showing abnormalities in any of the pathways of interest. The first 129 received a mean of three prior therapies, and in the 29 who responded, 12 different types of cancer responded to the targeted treatment.
“An increasing number of targeted agents for advanced cancer are in use now based on the presence of molecular abnormalities in the cancer. … We’ve known that the same mutations that are in those cancers are found in a wide variety of other cancers, although at a lower incidence, and it’s been difficult to test how effective these same treatments are for the other cancers due to the difficulty in identifying the patient population,” he said, explaining that an increase in comprehensive genomic profiling in recent years has allowed for identification of more and more of these mutations in other cancers.
“I think we’ve shown now that this trial design is feasible, where patients are selected on the basis of molecular abnormalities in their cancers rather than on their primary tumor type or primary site, and certainly offers opportunities for patients with these molecular abnormalities,” Dr. Hainsworth concluded.
Thus far, MyPathway has enrolled more than 200 patients, and is designed to accrue up to 500, with adjustment of treatment groups based on response rates. Emerging new regimens that target these pathways, such as the MEK inhibitor cobemetinib, will also be added, as will new agents targeting additional molecular abnormalities.
The study design, using this “tumor-agnostic approach,” mirrors that of the ASCO-led TAPUR trial, according to ASCO spokesperson Dr. Sumanta Kumar Pal.
The findings of these and other precision medicine trials may ultimately shift the longstanding cancer treatment paradigm, Dr. Pal said.
MyPathway received funding from Genentech. Dr. Hainsworth reported that his institution has received research funding from Astellas Pharma, AstraZeneca, Celgene, Genentech, Johnson & Johnson, Lilly, and Novartis.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Agents that target the HER2, BRAF, Hedgehog, or EGFR pathways show promise in nonindicated tumor types that harbor these molecular alterations, according to early findings from the MyPathway study.
Major finding: Twenty-nine patients had a major response, and an additional 40 remained stable on treatment.
Data source: The ongoing open-label, phase IIa MyPathway study, including results from the first 129 patients.
Disclosures: MyPathway received funding from Genentech. Dr. Hainsworth reported that his institution has received research funding from Astellas Pharma, AstraZeneca, Celgene, Genentech, Johnson & Johnson, Lilly, and Novartis.
Liquid biopsies prove useful alternative to tissue biopsies
CHICAGO – Liquid biopsy, the testing of the blood for circulating tumor DNA (ctDNA), identified cancer mutations useful as biomarkers in 85% of all advanced cancer cases in the largest-ever genomic analysis performed using such technology.
In nearly half of those (49%), the biomarkers were associated with an approved targeted drug, Philip C. Mack, Ph.D., reported at the annual meeting of the American Society of Clinical Oncology.
The patterns of genetic changes detected in the 17,628 blood specimens analyzed for the study using a highly sensitive next-generation sequencing technique closely mirrored those identified using traditional tumor biopsies, suggesting that liquid biopsy provides a non-invasive alternative to tissue biopsy in certain cases, Dr. Mack, professor and director of molecular pharmacology at the University of California, Davis Comprehensive Cancer Center, said during a press briefing at the meeting.
Overall, taking into account FDA-approved drugs and eligibility for clinical trials, the ctDNA testing revealed a possible treatment option for 63.6% of the 15,191 patients who provided samples.
Further, the commercially available liquid biopsy assay used in the study (Guardant360) identified the presence of resistance alterations that could guide new therapy in patients with tumors that acquire resistance to an effective treatment, he said, noting that these resistance mutations are not typically present at the time of initial tissue-biopsy.
Of the patients included in the study, 37% had lung cancer, 14% had breast cancer, 10% had colorectal cancer, and 39% had other cancers. A comparison of genomic changes in ctDNA in 398 patients with available tumor tissue genetic test results showed that when ctDNA was positive for key abnormalities associated with tumor growth, the same mutations were reported in tissue 94% to 100% of the time.
Most of the ctDNA alterations were found at very low levels – with half occurring at a frequency below 0.4% of the total DNA in circulation; even at such low levels, the accuracy of the liquid biopsy assay remained high, Dr. Mack noted.
The ctDNA findings also compared well with those from publicly available population-scale sequencing projects, most notably the Cancer Genome Atlas, he said.
Alterations observed at ctDNA fractions as low as 0.06% responded to treatment, which highlights the importance of assay sensitivity.
Interest in comprehensive tumor genetic profiling to guide patients toward appropriate targeted therapies based on the molecular makeup of their tumors has been increasing, and the current findings underscore its potential value. Advantages of plasma testing include ease of use in any clinic setting, avoidance of biopsy-related complications, ability to monitor changes in disease over time, potential identification of mutations in metastatic lesions not observed in the original tumor biopsy (as genetic changes driving tumor growth often differ in different parts of the tumor), and the opportunity to identify treatment-induced resistance mechanisms, Dr. Mack said.
Additionally, liquid biopsy could be particularly helpful in cases involving tumors for which a traditional biopsy is difficult to obtain.
In fact, the clinical utility of liquid biopsy was evident among 362 lung cancer cases in the study. Tissue was insufficient for traditional biopsy in 63% of the cases, and ctDNA testing identified key genetic mutations at frequencies consistent with their prevalence in the published literature. Thus, ctDNA provided these patients with their only source of an actionable target, Dr. Mack noted.
As for whether liquid biopsy could or should replace tissue biopsy, he explained that it should be viewed more as an additional tool.
“I think there is always going to be a role for tissue-based biopsy,” he said, explaining that a tissue biopsy allows the pathologist to assess the morphological features of the cancer to diagnose it and determine what the tumor type is and where it originated. “That will always be required.”
Leftover tissue should be used for mutational testing, and this is the gold standard. It is in cases when the tissue is of insufficient quality or quantity to allow a broader array of testing that liquid biopsy can provide a complementary source of information, he said.
Further, while it would be ideal to have biopsies of any progressing lesion in a patient who initially had a great response to treatment, that is not feasible, he said.
“So probably, the biggest role for plasma analysis will be occurring down the road as patients are progressing on therapies, as their tumors are evolving, as a way to monitor progression of those cancers,” he said.
Dr. Sumanta Kumar Pal, an ASCO spokesperson and a panel member at the press briefing, noted that “the data that Dr. Mack reported provides key insights into the feasibility of the so-called liquid biopsy.”
“We’re increasingly using genomic data from day-to-day in our clinics to guide therapies,” he said, reiterating that tests such as this provide a useful alternative to tissue-based testing, particularly for the many patients in clinics who have tumors that are challenging to access, such as tumors on bone or near the brain.
“The authors have demonstrated not only the feasibility of the test in detecting alterations, but that the results are often potentially actionable as well ... As oncologists obtain this assay, it’s important to keep in mind trials such as the ASCO-led TAPUR study. TAPUR can potentially link patients with selected genomic alterations to relevant therapies that would otherwise be challenging to access,” he said.
Dr. Pal also noted that there are several other blood-based genomic tests emerging, and it will be important to “rigorously define which of these platforms deliver optimal results.”
Going forward, Dr. Mack and his colleagues will be working to increase the sensitivity of the Gaurdant360 assay to detect mutations at extremely low ctDNA level, as some tumors – glioblastomas, for example – diminish the ability to detect ctDNA. Improved sensitivity could also enable the use of the assay in earlier-stage cancers, he said.
This study was supported by funding from Guardant Health.
CHICAGO – Liquid biopsy, the testing of the blood for circulating tumor DNA (ctDNA), identified cancer mutations useful as biomarkers in 85% of all advanced cancer cases in the largest-ever genomic analysis performed using such technology.
In nearly half of those (49%), the biomarkers were associated with an approved targeted drug, Philip C. Mack, Ph.D., reported at the annual meeting of the American Society of Clinical Oncology.
The patterns of genetic changes detected in the 17,628 blood specimens analyzed for the study using a highly sensitive next-generation sequencing technique closely mirrored those identified using traditional tumor biopsies, suggesting that liquid biopsy provides a non-invasive alternative to tissue biopsy in certain cases, Dr. Mack, professor and director of molecular pharmacology at the University of California, Davis Comprehensive Cancer Center, said during a press briefing at the meeting.
Overall, taking into account FDA-approved drugs and eligibility for clinical trials, the ctDNA testing revealed a possible treatment option for 63.6% of the 15,191 patients who provided samples.
Further, the commercially available liquid biopsy assay used in the study (Guardant360) identified the presence of resistance alterations that could guide new therapy in patients with tumors that acquire resistance to an effective treatment, he said, noting that these resistance mutations are not typically present at the time of initial tissue-biopsy.
Of the patients included in the study, 37% had lung cancer, 14% had breast cancer, 10% had colorectal cancer, and 39% had other cancers. A comparison of genomic changes in ctDNA in 398 patients with available tumor tissue genetic test results showed that when ctDNA was positive for key abnormalities associated with tumor growth, the same mutations were reported in tissue 94% to 100% of the time.
Most of the ctDNA alterations were found at very low levels – with half occurring at a frequency below 0.4% of the total DNA in circulation; even at such low levels, the accuracy of the liquid biopsy assay remained high, Dr. Mack noted.
The ctDNA findings also compared well with those from publicly available population-scale sequencing projects, most notably the Cancer Genome Atlas, he said.
Alterations observed at ctDNA fractions as low as 0.06% responded to treatment, which highlights the importance of assay sensitivity.
Interest in comprehensive tumor genetic profiling to guide patients toward appropriate targeted therapies based on the molecular makeup of their tumors has been increasing, and the current findings underscore its potential value. Advantages of plasma testing include ease of use in any clinic setting, avoidance of biopsy-related complications, ability to monitor changes in disease over time, potential identification of mutations in metastatic lesions not observed in the original tumor biopsy (as genetic changes driving tumor growth often differ in different parts of the tumor), and the opportunity to identify treatment-induced resistance mechanisms, Dr. Mack said.
Additionally, liquid biopsy could be particularly helpful in cases involving tumors for which a traditional biopsy is difficult to obtain.
In fact, the clinical utility of liquid biopsy was evident among 362 lung cancer cases in the study. Tissue was insufficient for traditional biopsy in 63% of the cases, and ctDNA testing identified key genetic mutations at frequencies consistent with their prevalence in the published literature. Thus, ctDNA provided these patients with their only source of an actionable target, Dr. Mack noted.
As for whether liquid biopsy could or should replace tissue biopsy, he explained that it should be viewed more as an additional tool.
“I think there is always going to be a role for tissue-based biopsy,” he said, explaining that a tissue biopsy allows the pathologist to assess the morphological features of the cancer to diagnose it and determine what the tumor type is and where it originated. “That will always be required.”
Leftover tissue should be used for mutational testing, and this is the gold standard. It is in cases when the tissue is of insufficient quality or quantity to allow a broader array of testing that liquid biopsy can provide a complementary source of information, he said.
Further, while it would be ideal to have biopsies of any progressing lesion in a patient who initially had a great response to treatment, that is not feasible, he said.
“So probably, the biggest role for plasma analysis will be occurring down the road as patients are progressing on therapies, as their tumors are evolving, as a way to monitor progression of those cancers,” he said.
Dr. Sumanta Kumar Pal, an ASCO spokesperson and a panel member at the press briefing, noted that “the data that Dr. Mack reported provides key insights into the feasibility of the so-called liquid biopsy.”
“We’re increasingly using genomic data from day-to-day in our clinics to guide therapies,” he said, reiterating that tests such as this provide a useful alternative to tissue-based testing, particularly for the many patients in clinics who have tumors that are challenging to access, such as tumors on bone or near the brain.
“The authors have demonstrated not only the feasibility of the test in detecting alterations, but that the results are often potentially actionable as well ... As oncologists obtain this assay, it’s important to keep in mind trials such as the ASCO-led TAPUR study. TAPUR can potentially link patients with selected genomic alterations to relevant therapies that would otherwise be challenging to access,” he said.
Dr. Pal also noted that there are several other blood-based genomic tests emerging, and it will be important to “rigorously define which of these platforms deliver optimal results.”
Going forward, Dr. Mack and his colleagues will be working to increase the sensitivity of the Gaurdant360 assay to detect mutations at extremely low ctDNA level, as some tumors – glioblastomas, for example – diminish the ability to detect ctDNA. Improved sensitivity could also enable the use of the assay in earlier-stage cancers, he said.
This study was supported by funding from Guardant Health.
CHICAGO – Liquid biopsy, the testing of the blood for circulating tumor DNA (ctDNA), identified cancer mutations useful as biomarkers in 85% of all advanced cancer cases in the largest-ever genomic analysis performed using such technology.
In nearly half of those (49%), the biomarkers were associated with an approved targeted drug, Philip C. Mack, Ph.D., reported at the annual meeting of the American Society of Clinical Oncology.
The patterns of genetic changes detected in the 17,628 blood specimens analyzed for the study using a highly sensitive next-generation sequencing technique closely mirrored those identified using traditional tumor biopsies, suggesting that liquid biopsy provides a non-invasive alternative to tissue biopsy in certain cases, Dr. Mack, professor and director of molecular pharmacology at the University of California, Davis Comprehensive Cancer Center, said during a press briefing at the meeting.
Overall, taking into account FDA-approved drugs and eligibility for clinical trials, the ctDNA testing revealed a possible treatment option for 63.6% of the 15,191 patients who provided samples.
Further, the commercially available liquid biopsy assay used in the study (Guardant360) identified the presence of resistance alterations that could guide new therapy in patients with tumors that acquire resistance to an effective treatment, he said, noting that these resistance mutations are not typically present at the time of initial tissue-biopsy.
Of the patients included in the study, 37% had lung cancer, 14% had breast cancer, 10% had colorectal cancer, and 39% had other cancers. A comparison of genomic changes in ctDNA in 398 patients with available tumor tissue genetic test results showed that when ctDNA was positive for key abnormalities associated with tumor growth, the same mutations were reported in tissue 94% to 100% of the time.
Most of the ctDNA alterations were found at very low levels – with half occurring at a frequency below 0.4% of the total DNA in circulation; even at such low levels, the accuracy of the liquid biopsy assay remained high, Dr. Mack noted.
The ctDNA findings also compared well with those from publicly available population-scale sequencing projects, most notably the Cancer Genome Atlas, he said.
Alterations observed at ctDNA fractions as low as 0.06% responded to treatment, which highlights the importance of assay sensitivity.
Interest in comprehensive tumor genetic profiling to guide patients toward appropriate targeted therapies based on the molecular makeup of their tumors has been increasing, and the current findings underscore its potential value. Advantages of plasma testing include ease of use in any clinic setting, avoidance of biopsy-related complications, ability to monitor changes in disease over time, potential identification of mutations in metastatic lesions not observed in the original tumor biopsy (as genetic changes driving tumor growth often differ in different parts of the tumor), and the opportunity to identify treatment-induced resistance mechanisms, Dr. Mack said.
Additionally, liquid biopsy could be particularly helpful in cases involving tumors for which a traditional biopsy is difficult to obtain.
In fact, the clinical utility of liquid biopsy was evident among 362 lung cancer cases in the study. Tissue was insufficient for traditional biopsy in 63% of the cases, and ctDNA testing identified key genetic mutations at frequencies consistent with their prevalence in the published literature. Thus, ctDNA provided these patients with their only source of an actionable target, Dr. Mack noted.
As for whether liquid biopsy could or should replace tissue biopsy, he explained that it should be viewed more as an additional tool.
“I think there is always going to be a role for tissue-based biopsy,” he said, explaining that a tissue biopsy allows the pathologist to assess the morphological features of the cancer to diagnose it and determine what the tumor type is and where it originated. “That will always be required.”
Leftover tissue should be used for mutational testing, and this is the gold standard. It is in cases when the tissue is of insufficient quality or quantity to allow a broader array of testing that liquid biopsy can provide a complementary source of information, he said.
Further, while it would be ideal to have biopsies of any progressing lesion in a patient who initially had a great response to treatment, that is not feasible, he said.
“So probably, the biggest role for plasma analysis will be occurring down the road as patients are progressing on therapies, as their tumors are evolving, as a way to monitor progression of those cancers,” he said.
Dr. Sumanta Kumar Pal, an ASCO spokesperson and a panel member at the press briefing, noted that “the data that Dr. Mack reported provides key insights into the feasibility of the so-called liquid biopsy.”
“We’re increasingly using genomic data from day-to-day in our clinics to guide therapies,” he said, reiterating that tests such as this provide a useful alternative to tissue-based testing, particularly for the many patients in clinics who have tumors that are challenging to access, such as tumors on bone or near the brain.
“The authors have demonstrated not only the feasibility of the test in detecting alterations, but that the results are often potentially actionable as well ... As oncologists obtain this assay, it’s important to keep in mind trials such as the ASCO-led TAPUR study. TAPUR can potentially link patients with selected genomic alterations to relevant therapies that would otherwise be challenging to access,” he said.
Dr. Pal also noted that there are several other blood-based genomic tests emerging, and it will be important to “rigorously define which of these platforms deliver optimal results.”
Going forward, Dr. Mack and his colleagues will be working to increase the sensitivity of the Gaurdant360 assay to detect mutations at extremely low ctDNA level, as some tumors – glioblastomas, for example – diminish the ability to detect ctDNA. Improved sensitivity could also enable the use of the assay in earlier-stage cancers, he said.
This study was supported by funding from Guardant Health.
AT THE 2016 ASCO ANNUAL MEETING
Key clinical point: Liquid biopsy, the testing of the blood for circulating tumor DNA (ctDNA), identified cancer mutations useful as biomarkers – many associated with an approved targeted drug – in 85% of all advanced cancer cases in a large genomic analysis.
Major finding: A comparison of genomic changes in ctDNA in 398 patients with available tumor tissue genetic test results showed that when ctDNA was positive for key abnormalities associated with tumor growth, the same mutations were reported in tissue 94% to 100% of the time.
Data source: A genomic analysis of 17,628 blood specimens from 15,191 patients.
Disclosures: This study was supported by funding from Guardant Health.