User login
Can Dual Immunotherapy Replace Surgery in Gastric Cancer?
Can Dual Immunotherapy Replace Surgery in Gastric Cancer?
Dual checkpoint blockade allowed 70.6% of patients with microsatellite instability-high (MSI-H) resectable gastric or gastroesophageal junction adenocarcinoma (G/GEJAC) to avoid surgery in a small cohort of the INFINITY study.
MSI-H tumors account for roughly 10% of early G/GEJACs. They respond well to immunotherapy, with high rates of pathologic complete responses. The Italian INFINITY trial set out to test whether some patients with these tumors might not need gastrectomy.
The trial treated MSI-H patients with durvalumab 1500 mg once a month for 3 months along with 1 300-mg dose of the cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) blocker tremelimumab on day 1. The 18 patients in cohort 1 proceeded to surgery, with a 60% pathologic complete response rate. An additional 18 patients in cohort 2 were the subject of a presentation at the American Association for Cancer Research (AACR) Annual Meeting 2026. These patients were assessed for clinical complete response; if present, they went on to surveillance; if not, they had surgery.
To qualify for a clinical complete response and surveillance, patients were required to have negative findings on CT and PET scans; tumor-informed circulating tumor DNA (ctDNA); and upper endoscopy with ultrasound, including bite-on-bite biopsies and nodal sampling. Surveillance afterward included CT, endoscopy with biopsies, and ctDNA every 12 weeks for up to 2 years.
Among 17 evaluable patients, 1 withdrew consent during immunotherapy, 13 (76%) had a clinical complete response and started surveillance, and the other 4 went to surgery. One patient in the surveillance group had a local regrowth after 4 months, underwent salvage surgery, and remained disease-free. At a median follow-up of 27.1 months, there were no additional progression events.
Overall, 12 of the 17 patients (70.6%) were gastrectomy-free at 2 years without additional treatment. Progression-free survival was 94.1%, and all patients were alive.
“The results are very encouraging,” lead investigator Alberto Leone, MD, said while presenting the results at the AACR annual meeting.
“Nonoperative management could be a safe and effective strategy for patients achieving a clinical complete response after only 3 months of dual immunotherapy,” said Leone, who is a gastrointestinal medical oncologist at the Istituto Nazionale dei Tumori, Milan, Italy. “However, the optimal strategy needs to be established in larger randomized trials.”
Study discussant Yelena Janjagian, MD, gastrointestinal medical oncologist at Memorial Sloan Kettering Cancer Center in New York City, said the findings were important, particularly given that 70.6% of patients avoided a potentially life-altering gastrectomy.
In addition to surgery, the study also calls into question the need for chemotherapy, long the backbone of management alongside surgery, she said. To replace it, however, “it appears that dual checkpoint blockade will be required for a chemotherapy-free approach to achieve organ preservation.”
“Anti-PD-1 alone is not sufficient; we need CTLA-4 to expand and reactivate tumor-specific immunity,” Janjagian continued.
Ultimately, she expects immunotherapy to shift management of MSI-H cancers away from surgery, although some patients will still likely need an operation.
In addition to being MSI-H, patients in the study were mismatch repair deficient and Epstein-Barr virus-negative with T2/T3 tumors; T4 tumors were excluded.
Tumor-agnostic plasma ctDNA was positive at baseline in 13 patients and cleared in 11 after treatment. Higher baseline plasma ctDNA trended toward a lower likelihood of reaching a clinical complete response. Specificity was 100%, so when positive, the test was “very highly informative,” Leone said.
Three patients had grade 3 adverse events (hyperthyroidism, increased gamma-glutamyl transferase, and colitis) that resolved with steroids. There were no grade 4 events, treatment discontinuation, or deaths.
The work was funded by the GONO Foundation and AstraZeneca, the maker of durvalumab and tremelimumab. Leone reported having no disclosures. Janjagian reported having extensive industry ties, including travel funding, consulting fees, and research support from AstraZeneca.
M. Alexander Otto is a physician assistant with a master’s degree in medical science and a journalism degree from Newhouse. He is an award-winning medical journalist who worked for several major news outlets before joining Medscape. Alex is also an MIT Knight Science Journalism fellow. Email: aotto@medscape.net
A version of this article first appeared on Medscape.com.
Dual checkpoint blockade allowed 70.6% of patients with microsatellite instability-high (MSI-H) resectable gastric or gastroesophageal junction adenocarcinoma (G/GEJAC) to avoid surgery in a small cohort of the INFINITY study.
MSI-H tumors account for roughly 10% of early G/GEJACs. They respond well to immunotherapy, with high rates of pathologic complete responses. The Italian INFINITY trial set out to test whether some patients with these tumors might not need gastrectomy.
The trial treated MSI-H patients with durvalumab 1500 mg once a month for 3 months along with 1 300-mg dose of the cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) blocker tremelimumab on day 1. The 18 patients in cohort 1 proceeded to surgery, with a 60% pathologic complete response rate. An additional 18 patients in cohort 2 were the subject of a presentation at the American Association for Cancer Research (AACR) Annual Meeting 2026. These patients were assessed for clinical complete response; if present, they went on to surveillance; if not, they had surgery.
To qualify for a clinical complete response and surveillance, patients were required to have negative findings on CT and PET scans; tumor-informed circulating tumor DNA (ctDNA); and upper endoscopy with ultrasound, including bite-on-bite biopsies and nodal sampling. Surveillance afterward included CT, endoscopy with biopsies, and ctDNA every 12 weeks for up to 2 years.
Among 17 evaluable patients, 1 withdrew consent during immunotherapy, 13 (76%) had a clinical complete response and started surveillance, and the other 4 went to surgery. One patient in the surveillance group had a local regrowth after 4 months, underwent salvage surgery, and remained disease-free. At a median follow-up of 27.1 months, there were no additional progression events.
Overall, 12 of the 17 patients (70.6%) were gastrectomy-free at 2 years without additional treatment. Progression-free survival was 94.1%, and all patients were alive.
“The results are very encouraging,” lead investigator Alberto Leone, MD, said while presenting the results at the AACR annual meeting.
“Nonoperative management could be a safe and effective strategy for patients achieving a clinical complete response after only 3 months of dual immunotherapy,” said Leone, who is a gastrointestinal medical oncologist at the Istituto Nazionale dei Tumori, Milan, Italy. “However, the optimal strategy needs to be established in larger randomized trials.”
Study discussant Yelena Janjagian, MD, gastrointestinal medical oncologist at Memorial Sloan Kettering Cancer Center in New York City, said the findings were important, particularly given that 70.6% of patients avoided a potentially life-altering gastrectomy.
In addition to surgery, the study also calls into question the need for chemotherapy, long the backbone of management alongside surgery, she said. To replace it, however, “it appears that dual checkpoint blockade will be required for a chemotherapy-free approach to achieve organ preservation.”
“Anti-PD-1 alone is not sufficient; we need CTLA-4 to expand and reactivate tumor-specific immunity,” Janjagian continued.
Ultimately, she expects immunotherapy to shift management of MSI-H cancers away from surgery, although some patients will still likely need an operation.
In addition to being MSI-H, patients in the study were mismatch repair deficient and Epstein-Barr virus-negative with T2/T3 tumors; T4 tumors were excluded.
Tumor-agnostic plasma ctDNA was positive at baseline in 13 patients and cleared in 11 after treatment. Higher baseline plasma ctDNA trended toward a lower likelihood of reaching a clinical complete response. Specificity was 100%, so when positive, the test was “very highly informative,” Leone said.
Three patients had grade 3 adverse events (hyperthyroidism, increased gamma-glutamyl transferase, and colitis) that resolved with steroids. There were no grade 4 events, treatment discontinuation, or deaths.
The work was funded by the GONO Foundation and AstraZeneca, the maker of durvalumab and tremelimumab. Leone reported having no disclosures. Janjagian reported having extensive industry ties, including travel funding, consulting fees, and research support from AstraZeneca.
M. Alexander Otto is a physician assistant with a master’s degree in medical science and a journalism degree from Newhouse. He is an award-winning medical journalist who worked for several major news outlets before joining Medscape. Alex is also an MIT Knight Science Journalism fellow. Email: aotto@medscape.net
A version of this article first appeared on Medscape.com.
Dual checkpoint blockade allowed 70.6% of patients with microsatellite instability-high (MSI-H) resectable gastric or gastroesophageal junction adenocarcinoma (G/GEJAC) to avoid surgery in a small cohort of the INFINITY study.
MSI-H tumors account for roughly 10% of early G/GEJACs. They respond well to immunotherapy, with high rates of pathologic complete responses. The Italian INFINITY trial set out to test whether some patients with these tumors might not need gastrectomy.
The trial treated MSI-H patients with durvalumab 1500 mg once a month for 3 months along with 1 300-mg dose of the cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) blocker tremelimumab on day 1. The 18 patients in cohort 1 proceeded to surgery, with a 60% pathologic complete response rate. An additional 18 patients in cohort 2 were the subject of a presentation at the American Association for Cancer Research (AACR) Annual Meeting 2026. These patients were assessed for clinical complete response; if present, they went on to surveillance; if not, they had surgery.
To qualify for a clinical complete response and surveillance, patients were required to have negative findings on CT and PET scans; tumor-informed circulating tumor DNA (ctDNA); and upper endoscopy with ultrasound, including bite-on-bite biopsies and nodal sampling. Surveillance afterward included CT, endoscopy with biopsies, and ctDNA every 12 weeks for up to 2 years.
Among 17 evaluable patients, 1 withdrew consent during immunotherapy, 13 (76%) had a clinical complete response and started surveillance, and the other 4 went to surgery. One patient in the surveillance group had a local regrowth after 4 months, underwent salvage surgery, and remained disease-free. At a median follow-up of 27.1 months, there were no additional progression events.
Overall, 12 of the 17 patients (70.6%) were gastrectomy-free at 2 years without additional treatment. Progression-free survival was 94.1%, and all patients were alive.
“The results are very encouraging,” lead investigator Alberto Leone, MD, said while presenting the results at the AACR annual meeting.
“Nonoperative management could be a safe and effective strategy for patients achieving a clinical complete response after only 3 months of dual immunotherapy,” said Leone, who is a gastrointestinal medical oncologist at the Istituto Nazionale dei Tumori, Milan, Italy. “However, the optimal strategy needs to be established in larger randomized trials.”
Study discussant Yelena Janjagian, MD, gastrointestinal medical oncologist at Memorial Sloan Kettering Cancer Center in New York City, said the findings were important, particularly given that 70.6% of patients avoided a potentially life-altering gastrectomy.
In addition to surgery, the study also calls into question the need for chemotherapy, long the backbone of management alongside surgery, she said. To replace it, however, “it appears that dual checkpoint blockade will be required for a chemotherapy-free approach to achieve organ preservation.”
“Anti-PD-1 alone is not sufficient; we need CTLA-4 to expand and reactivate tumor-specific immunity,” Janjagian continued.
Ultimately, she expects immunotherapy to shift management of MSI-H cancers away from surgery, although some patients will still likely need an operation.
In addition to being MSI-H, patients in the study were mismatch repair deficient and Epstein-Barr virus-negative with T2/T3 tumors; T4 tumors were excluded.
Tumor-agnostic plasma ctDNA was positive at baseline in 13 patients and cleared in 11 after treatment. Higher baseline plasma ctDNA trended toward a lower likelihood of reaching a clinical complete response. Specificity was 100%, so when positive, the test was “very highly informative,” Leone said.
Three patients had grade 3 adverse events (hyperthyroidism, increased gamma-glutamyl transferase, and colitis) that resolved with steroids. There were no grade 4 events, treatment discontinuation, or deaths.
The work was funded by the GONO Foundation and AstraZeneca, the maker of durvalumab and tremelimumab. Leone reported having no disclosures. Janjagian reported having extensive industry ties, including travel funding, consulting fees, and research support from AstraZeneca.
M. Alexander Otto is a physician assistant with a master’s degree in medical science and a journalism degree from Newhouse. He is an award-winning medical journalist who worked for several major news outlets before joining Medscape. Alex is also an MIT Knight Science Journalism fellow. Email: aotto@medscape.net
A version of this article first appeared on Medscape.com.
Can Dual Immunotherapy Replace Surgery in Gastric Cancer?
Can Dual Immunotherapy Replace Surgery in Gastric Cancer?
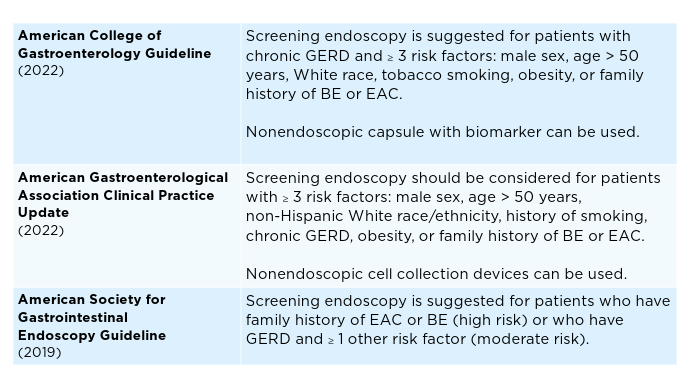
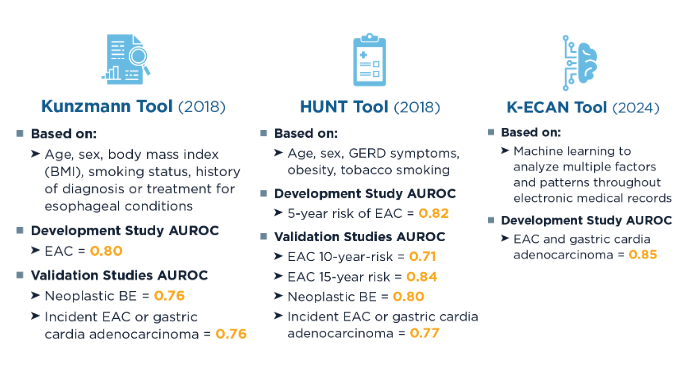
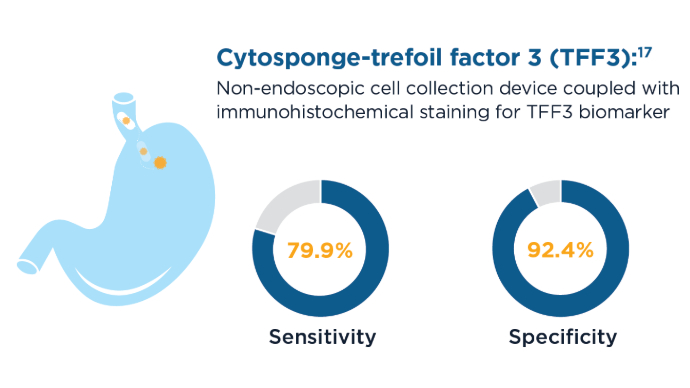
Advances in Screening for Barrett’s Esophagus and Esophageal Adenocarcinoma
Advances in Screening for Barrett’s Esophagus and Esophageal Adenocarcinoma
Click to view more from Gastroenterology Data Trends 2025.
- Vantanasiri K, Kamboj AK, Kisiel JB, Iyer PG. Advances in Screening for Barrett Esophagus and Esophageal Adenocarcinoma. Mayo Clin Proc. 2024;99(3):459-473. doi:10.1016/j.mayocp.2023.07.014
- Cancer Stat Facts: Esophageal Cancer. NIH National Cancer Institute: Survival, Epidemiology, and End Results Program web site. Accessed March 12, 2025. https://seer.cancer.gov/statfacts/html/esoph.html
- Seer*Explorer: Esophagus. NIH National Cancer Institute: Survival, Epidemiology, and End Results Program web site. Accessed March 4, 2025. https://seer.cancer.gov/statistics-network/explorer/application.html
- Kolb JM, Chen M, Tavakkoli A, et al. Understanding Compliance, Practice Patterns, and Barriers Among Gastroenterologists and Primary Care Providers Is Crucial for Developing Strategies to Improve Screening for Barrett’s Esophagus. Gastroenterology. 2022;162(6):1568-1573.e4. doi:10.1053/j.gastro.2022.02.003
- Kunzmann AT, Thrift AP, Cardwell CR, et al. Model for Identifying Individuals at Risk for Esophageal Adenocarcinoma. Clin Gastroenterol Hepatol. 2018;16(8):1229-1236.e4. doi:10.1016/j.cgh.2018.03.014
- Rubenstein JH, Evans RR, Burns JA, et al. Patients With Adenocarcinoma of the Esophagus or Esophagogastric Junction Frequently Have Potential Screening Opportunities. Gastroenterology. 2022;162(4):1349-1351.e5. doi:10.1053/j.gastro.2021.12.255
- Xie S-H, Ness-Jensen E, Medefelt N, Lagergren J. Assessing the feasibility of targeted screening for esophageal adenocarcinoma based on individual risk assessment in a population-based cohort study in Norway (The HUNT Study). Am J Gastroenterol. 2018;113(6):829-835. doi:10.1038/s41395-018-0069-9
- Rubenstein JH, Fontaine S, MacDonald PW, et al. Predicting Incident Adenocarcinoma of the Esophagus or Gastric Cardia Using Machine Learning of Electronic Health Records. Gastroenterology. 2023;165(6):1420-1429.e10. doi:10.1053/j.gastro.2023.08.011
- Fitzgerald RC, di Pietro M, O’Donovan M, et al. Cytosponge-trefoil factor 3 versus usual care to identify Barrett’s oesophagus in a primary care setting: a multicentre, pragmatic, randomised controlled trial. Lancet. 2020;396(10247):333-344. doi:10.1016/S0140-6736(20)31099-0
- Moinova HR, Verma S, Dumot J, et al. Multicenter, Prospective Trial of Nonendoscopic
Biomarker-Driven Detection of Barrett’s Esophagus and Esophageal Adenocarcinoma. Am J Gastroenterol. 2024;119(11):2206-2214. doi:10.14309/ajg.0000000000002850 - Shaheen NJ, Falk GW, Iyer PG, et al. Diagnosis and Management of Barrett’s Esophagus: An Updated ACG Guideline. Am J Gastroenterol. 2022;117(4):559-587. doi:10.14309/ajg.0000000000001680
- ASGE STANDARDS OF PRACTICE COMMITTEE; Qumseya B, Sultan S, Bain P, et al. ASGE guideline on screening and surveillance of Barrett’s esophagus. Gastrointest Endosc. 2019;90(3):335-359.e2. doi:10.1016/j.gie.2019.05.012
- Muthusamy VR, Wani S, Gyawali CP, Komanduri S. CGIT Barrett’s Esophagus Consensus Conference Participants. AGA Clinical Practice Update on New Technology and Innovation for Surveillance and Screening in Barrett’s Esophagus: Expert review. Clin Gastroenterol Hepatol. 2022;20(12):2696-2706. doi:10.1016/j.cgh.2022.06.003
- Xie SH, Lagergren J. A model for predicting individuals’ absolute risk of esophageal adenocarcinoma: Moving toward tailored screening and prevention. Int J Cancer. 2016;138(12):2813-2819. doi:10.1002/ijc.29988
- Rubenstein JH, McConnell D, Waljee AK, et al. Validation and Comparison of Tools for Selecting Individuals to Screen for Barrett’s Esophagus and Early Neoplasia. Gastroenterology. 2020;158(8):2082-2092. doi:10.1053/j.gastro.2020.02.037
- Iyer PG, Sachdeva K, Leggett CL, et al. Development of Electronic Health Record–Based Machine Learning Models to Predict Barrett’s Esophagus and Esophageal Adenocarcinoma Risk. Clin Transl Gastroenterol. 2023;14(10):e00637. doi:10.14309/ctg.0000000000000637
- Ross-Innes CS, Debiram-Beecham I, O’Donovan M, et al; BEST2 Study Group. Evaluation of a minimally invasive cell sampling device coupled with assessment of trefoil factor 3 expression for diagnosing Barrett’s esophagus: a multicenter case-control study. PLoS Med. 2015;12(1):e1001780. doi:10.1371/journal.pmed.1001780
Click to view more from Gastroenterology Data Trends 2025.
Click to view more from Gastroenterology Data Trends 2025.
- Vantanasiri K, Kamboj AK, Kisiel JB, Iyer PG. Advances in Screening for Barrett Esophagus and Esophageal Adenocarcinoma. Mayo Clin Proc. 2024;99(3):459-473. doi:10.1016/j.mayocp.2023.07.014
- Cancer Stat Facts: Esophageal Cancer. NIH National Cancer Institute: Survival, Epidemiology, and End Results Program web site. Accessed March 12, 2025. https://seer.cancer.gov/statfacts/html/esoph.html
- Seer*Explorer: Esophagus. NIH National Cancer Institute: Survival, Epidemiology, and End Results Program web site. Accessed March 4, 2025. https://seer.cancer.gov/statistics-network/explorer/application.html
- Kolb JM, Chen M, Tavakkoli A, et al. Understanding Compliance, Practice Patterns, and Barriers Among Gastroenterologists and Primary Care Providers Is Crucial for Developing Strategies to Improve Screening for Barrett’s Esophagus. Gastroenterology. 2022;162(6):1568-1573.e4. doi:10.1053/j.gastro.2022.02.003
- Kunzmann AT, Thrift AP, Cardwell CR, et al. Model for Identifying Individuals at Risk for Esophageal Adenocarcinoma. Clin Gastroenterol Hepatol. 2018;16(8):1229-1236.e4. doi:10.1016/j.cgh.2018.03.014
- Rubenstein JH, Evans RR, Burns JA, et al. Patients With Adenocarcinoma of the Esophagus or Esophagogastric Junction Frequently Have Potential Screening Opportunities. Gastroenterology. 2022;162(4):1349-1351.e5. doi:10.1053/j.gastro.2021.12.255
- Xie S-H, Ness-Jensen E, Medefelt N, Lagergren J. Assessing the feasibility of targeted screening for esophageal adenocarcinoma based on individual risk assessment in a population-based cohort study in Norway (The HUNT Study). Am J Gastroenterol. 2018;113(6):829-835. doi:10.1038/s41395-018-0069-9
- Rubenstein JH, Fontaine S, MacDonald PW, et al. Predicting Incident Adenocarcinoma of the Esophagus or Gastric Cardia Using Machine Learning of Electronic Health Records. Gastroenterology. 2023;165(6):1420-1429.e10. doi:10.1053/j.gastro.2023.08.011
- Fitzgerald RC, di Pietro M, O’Donovan M, et al. Cytosponge-trefoil factor 3 versus usual care to identify Barrett’s oesophagus in a primary care setting: a multicentre, pragmatic, randomised controlled trial. Lancet. 2020;396(10247):333-344. doi:10.1016/S0140-6736(20)31099-0
- Moinova HR, Verma S, Dumot J, et al. Multicenter, Prospective Trial of Nonendoscopic
Biomarker-Driven Detection of Barrett’s Esophagus and Esophageal Adenocarcinoma. Am J Gastroenterol. 2024;119(11):2206-2214. doi:10.14309/ajg.0000000000002850 - Shaheen NJ, Falk GW, Iyer PG, et al. Diagnosis and Management of Barrett’s Esophagus: An Updated ACG Guideline. Am J Gastroenterol. 2022;117(4):559-587. doi:10.14309/ajg.0000000000001680
- ASGE STANDARDS OF PRACTICE COMMITTEE; Qumseya B, Sultan S, Bain P, et al. ASGE guideline on screening and surveillance of Barrett’s esophagus. Gastrointest Endosc. 2019;90(3):335-359.e2. doi:10.1016/j.gie.2019.05.012
- Muthusamy VR, Wani S, Gyawali CP, Komanduri S. CGIT Barrett’s Esophagus Consensus Conference Participants. AGA Clinical Practice Update on New Technology and Innovation for Surveillance and Screening in Barrett’s Esophagus: Expert review. Clin Gastroenterol Hepatol. 2022;20(12):2696-2706. doi:10.1016/j.cgh.2022.06.003
- Xie SH, Lagergren J. A model for predicting individuals’ absolute risk of esophageal adenocarcinoma: Moving toward tailored screening and prevention. Int J Cancer. 2016;138(12):2813-2819. doi:10.1002/ijc.29988
- Rubenstein JH, McConnell D, Waljee AK, et al. Validation and Comparison of Tools for Selecting Individuals to Screen for Barrett’s Esophagus and Early Neoplasia. Gastroenterology. 2020;158(8):2082-2092. doi:10.1053/j.gastro.2020.02.037
- Iyer PG, Sachdeva K, Leggett CL, et al. Development of Electronic Health Record–Based Machine Learning Models to Predict Barrett’s Esophagus and Esophageal Adenocarcinoma Risk. Clin Transl Gastroenterol. 2023;14(10):e00637. doi:10.14309/ctg.0000000000000637
- Ross-Innes CS, Debiram-Beecham I, O’Donovan M, et al; BEST2 Study Group. Evaluation of a minimally invasive cell sampling device coupled with assessment of trefoil factor 3 expression for diagnosing Barrett’s esophagus: a multicenter case-control study. PLoS Med. 2015;12(1):e1001780. doi:10.1371/journal.pmed.1001780
- Vantanasiri K, Kamboj AK, Kisiel JB, Iyer PG. Advances in Screening for Barrett Esophagus and Esophageal Adenocarcinoma. Mayo Clin Proc. 2024;99(3):459-473. doi:10.1016/j.mayocp.2023.07.014
- Cancer Stat Facts: Esophageal Cancer. NIH National Cancer Institute: Survival, Epidemiology, and End Results Program web site. Accessed March 12, 2025. https://seer.cancer.gov/statfacts/html/esoph.html
- Seer*Explorer: Esophagus. NIH National Cancer Institute: Survival, Epidemiology, and End Results Program web site. Accessed March 4, 2025. https://seer.cancer.gov/statistics-network/explorer/application.html
- Kolb JM, Chen M, Tavakkoli A, et al. Understanding Compliance, Practice Patterns, and Barriers Among Gastroenterologists and Primary Care Providers Is Crucial for Developing Strategies to Improve Screening for Barrett’s Esophagus. Gastroenterology. 2022;162(6):1568-1573.e4. doi:10.1053/j.gastro.2022.02.003
- Kunzmann AT, Thrift AP, Cardwell CR, et al. Model for Identifying Individuals at Risk for Esophageal Adenocarcinoma. Clin Gastroenterol Hepatol. 2018;16(8):1229-1236.e4. doi:10.1016/j.cgh.2018.03.014
- Rubenstein JH, Evans RR, Burns JA, et al. Patients With Adenocarcinoma of the Esophagus or Esophagogastric Junction Frequently Have Potential Screening Opportunities. Gastroenterology. 2022;162(4):1349-1351.e5. doi:10.1053/j.gastro.2021.12.255
- Xie S-H, Ness-Jensen E, Medefelt N, Lagergren J. Assessing the feasibility of targeted screening for esophageal adenocarcinoma based on individual risk assessment in a population-based cohort study in Norway (The HUNT Study). Am J Gastroenterol. 2018;113(6):829-835. doi:10.1038/s41395-018-0069-9
- Rubenstein JH, Fontaine S, MacDonald PW, et al. Predicting Incident Adenocarcinoma of the Esophagus or Gastric Cardia Using Machine Learning of Electronic Health Records. Gastroenterology. 2023;165(6):1420-1429.e10. doi:10.1053/j.gastro.2023.08.011
- Fitzgerald RC, di Pietro M, O’Donovan M, et al. Cytosponge-trefoil factor 3 versus usual care to identify Barrett’s oesophagus in a primary care setting: a multicentre, pragmatic, randomised controlled trial. Lancet. 2020;396(10247):333-344. doi:10.1016/S0140-6736(20)31099-0
- Moinova HR, Verma S, Dumot J, et al. Multicenter, Prospective Trial of Nonendoscopic
Biomarker-Driven Detection of Barrett’s Esophagus and Esophageal Adenocarcinoma. Am J Gastroenterol. 2024;119(11):2206-2214. doi:10.14309/ajg.0000000000002850 - Shaheen NJ, Falk GW, Iyer PG, et al. Diagnosis and Management of Barrett’s Esophagus: An Updated ACG Guideline. Am J Gastroenterol. 2022;117(4):559-587. doi:10.14309/ajg.0000000000001680
- ASGE STANDARDS OF PRACTICE COMMITTEE; Qumseya B, Sultan S, Bain P, et al. ASGE guideline on screening and surveillance of Barrett’s esophagus. Gastrointest Endosc. 2019;90(3):335-359.e2. doi:10.1016/j.gie.2019.05.012
- Muthusamy VR, Wani S, Gyawali CP, Komanduri S. CGIT Barrett’s Esophagus Consensus Conference Participants. AGA Clinical Practice Update on New Technology and Innovation for Surveillance and Screening in Barrett’s Esophagus: Expert review. Clin Gastroenterol Hepatol. 2022;20(12):2696-2706. doi:10.1016/j.cgh.2022.06.003
- Xie SH, Lagergren J. A model for predicting individuals’ absolute risk of esophageal adenocarcinoma: Moving toward tailored screening and prevention. Int J Cancer. 2016;138(12):2813-2819. doi:10.1002/ijc.29988
- Rubenstein JH, McConnell D, Waljee AK, et al. Validation and Comparison of Tools for Selecting Individuals to Screen for Barrett’s Esophagus and Early Neoplasia. Gastroenterology. 2020;158(8):2082-2092. doi:10.1053/j.gastro.2020.02.037
- Iyer PG, Sachdeva K, Leggett CL, et al. Development of Electronic Health Record–Based Machine Learning Models to Predict Barrett’s Esophagus and Esophageal Adenocarcinoma Risk. Clin Transl Gastroenterol. 2023;14(10):e00637. doi:10.14309/ctg.0000000000000637
- Ross-Innes CS, Debiram-Beecham I, O’Donovan M, et al; BEST2 Study Group. Evaluation of a minimally invasive cell sampling device coupled with assessment of trefoil factor 3 expression for diagnosing Barrett’s esophagus: a multicenter case-control study. PLoS Med. 2015;12(1):e1001780. doi:10.1371/journal.pmed.1001780
Advances in Screening for Barrett’s Esophagus and Esophageal Adenocarcinoma
Advances in Screening for Barrett’s Esophagus and Esophageal Adenocarcinoma
New Model Estimates Hepatocellular Carcinoma Risk in Patients With Chronic Hepatitis B
The model, called Revised REACH-B or reREACH-B, stems from cohort studies in Hong Kong, South Korea, and Taiwan, and looks at the nonlinear parabolic association between serum hepatitis B virus (HBV) DNA levels and HCC risk.
“Current clinical practice guidelines don’t advocate antiviral treatment for patients with CHB who don’t show elevated alanine aminotransferase (ALT) levels, even in those with high HBV viral loads,” said coauthor Young-Suk Lim, MD, PhD, professor of gastroenterology at the University of Ulsan College of Medicine and Asan Medical Center in Seoul, South Korea.
“This stance is rooted in the notion that patients in the immune-tolerant phase are at very low risk for developing HCC,” Lim said. “However, the immune-tolerant phase includes patients with HBV DNA levels who face the highest risk for HCC, and many patients with moderate HBV viremia fall into an undefined gray zone.”
The study was published in Annals of Internal Medicine.
Validating reREACH-B
During a course of CHB, HBV viral loads and HCC risks evolve over time because of viral replication and host immune responses, Lim explained. Most patients typically move to seroclearance and an “inactive hepatitis” phase, but about 10%-20% can progress to a “reactivation” phase, where HBV DNA levels and ALT levels increase, which can increase HCC risk as well.
In a previous cohort study in Taiwan, a prognostic model called Risk Estimation for HCC in CHB — or REACH-B — found the risk for HCC increases tenfold with increasing levels of HBV DNA up to 5 log10IU/mL in noncirrhotic patients with CHB, regardless of ALT levels. Another cohort study in South Korea found a nonlinear parabolic association between HCC risk and HBV DNA levels up to 9 log10 IU/mL, with the highest risks found for moderate HBV DNA levels around 6 log10 IU/mL.
In this study, Lim and colleagues developed a prognostic model to integrate the nonlinear relationship and validated it externally, as well as compared it with the previous REACH-B model. The Revised REACH-B model incorporates six variables: age, sex, platelet count, HBV DNA level, ALT, and hepatitis B e-antigen (HBeAg).
The study included 14,378 treatment-naive, noncirrhotic adults with CHB and serum ALT levels < two times the upper limit of normal for at least 1 year and serum hepatitis B surface antigen for at least 6 months. The internal validation cohort included 6,949 patients from Asan Medical Center, and the external validation cohort included 7,429 patients from previous studies in Hong Kong, South Korea, and Taiwan.
Among the Asan cohort, the mean age was 45 years, 29.9% were HBeAg positive, median HBV DNA levels were 3.1 log10 IU/mL, and the median ALT level was 25 U/L. In the external cohort, the mean age was 46 years, 21% were HBeAg positive, median HBV DNA levels were 3.4 log10 IU/mL, and the median ALT level was 20 U/L.
In the Asan cohort, 435 patients (6.3%) developed HCC during a median follow-up of 10 years. The annual HCC incidence rate was 0.63 per 100 person-years, and the estimated cumulative probability of developing HCC at 10 years was 6.4%.
In the external cohort, 467 patients (6.3%) developed HCC during a median follow-up of 12 years. The annual HCC incidence rate was 0.42 per 100 person-years, and the estimated cumulative probability of developing HCC at 10 years was 3.1%.
Overall, the association between HBV viral load and HCC risk was linear in the HBeAg-negative groups and inverse in the HBeAg-positive groups, with the association between HBV viral load and HCC risk showing a nonlinear parabolic pattern.
Across both cohorts, patients with HBV DNA levels between 5 and 6 log10 IU/mL had the highest risk for HCC in both the HBeAg-negative and HBeAg-positive groups, which was more than eight times higher than those HBV DNA levels ≤ 3 log10 IU/mL.
For internal validation, the Revised REACH-B model had a c-statistic of 0.844 and 5-year area under the curve of 0.864. For external validation across the three external cohorts, the reREACH-B had c-statistics of 0.804, 0.808, and 0.813, and 5-year area under the curve of 0.839, 0.860, and 0.865.
In addition, the revised model yielded a greater positive net benefit than the REACH-B model in the threshold probability range between 0% and 18%.
“These analyses indicate the reREACH-B model can be a valuable tool in clinical practice, aiding in timely management decisions,” Lim said.
Considering Prognostic Models
This study highlights the importance of recognizing that the association between HBV DNA viral load and HCC risk isn’t linear, said Norah Terrault, MD, chief of Gastroenterology and Hepatology at the Keck School of Medicine at the University of Southern California, Los Angeles.
“In contrast to most chronic liver diseases where liver cancer develops only among those with advanced fibrosis/cirrhosis, people with chronic hepatitis B are at risk prior to the development of cirrhosis,” she said. “Risk prediction scores for HCC can be a useful means of identifying those without cirrhosis who should be enrolled in HCC surveillance programs.”
For instance, patients with HBV DNA levels < 3 log10 IU/mL or > 8 log10 IU/mL don’t have an increased risk, Terrault noted. However, the highest risk group appears to be around 5-6 log10 IU/mL.
“Future risk prediction models should acknowledge that relationship in modeling HCC risk,” she said. “The re-REACH-B provides modest improvement over the REACH-B, but further validation of this score in more diverse cohorts is essential.”
The study received financial support from the Korean government and grants from the Patient-Centered Clinical Research Coordinating Center of the National Evidence-based Healthcare Collaborating Agency and the National R&D Program for Cancer Control through the National Cancer Center, which is funded by Korea’s Ministry of Health and Welfare. Lim and Terrault reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
The model, called Revised REACH-B or reREACH-B, stems from cohort studies in Hong Kong, South Korea, and Taiwan, and looks at the nonlinear parabolic association between serum hepatitis B virus (HBV) DNA levels and HCC risk.
“Current clinical practice guidelines don’t advocate antiviral treatment for patients with CHB who don’t show elevated alanine aminotransferase (ALT) levels, even in those with high HBV viral loads,” said coauthor Young-Suk Lim, MD, PhD, professor of gastroenterology at the University of Ulsan College of Medicine and Asan Medical Center in Seoul, South Korea.
“This stance is rooted in the notion that patients in the immune-tolerant phase are at very low risk for developing HCC,” Lim said. “However, the immune-tolerant phase includes patients with HBV DNA levels who face the highest risk for HCC, and many patients with moderate HBV viremia fall into an undefined gray zone.”
The study was published in Annals of Internal Medicine.
Validating reREACH-B
During a course of CHB, HBV viral loads and HCC risks evolve over time because of viral replication and host immune responses, Lim explained. Most patients typically move to seroclearance and an “inactive hepatitis” phase, but about 10%-20% can progress to a “reactivation” phase, where HBV DNA levels and ALT levels increase, which can increase HCC risk as well.
In a previous cohort study in Taiwan, a prognostic model called Risk Estimation for HCC in CHB — or REACH-B — found the risk for HCC increases tenfold with increasing levels of HBV DNA up to 5 log10IU/mL in noncirrhotic patients with CHB, regardless of ALT levels. Another cohort study in South Korea found a nonlinear parabolic association between HCC risk and HBV DNA levels up to 9 log10 IU/mL, with the highest risks found for moderate HBV DNA levels around 6 log10 IU/mL.
In this study, Lim and colleagues developed a prognostic model to integrate the nonlinear relationship and validated it externally, as well as compared it with the previous REACH-B model. The Revised REACH-B model incorporates six variables: age, sex, platelet count, HBV DNA level, ALT, and hepatitis B e-antigen (HBeAg).
The study included 14,378 treatment-naive, noncirrhotic adults with CHB and serum ALT levels < two times the upper limit of normal for at least 1 year and serum hepatitis B surface antigen for at least 6 months. The internal validation cohort included 6,949 patients from Asan Medical Center, and the external validation cohort included 7,429 patients from previous studies in Hong Kong, South Korea, and Taiwan.
Among the Asan cohort, the mean age was 45 years, 29.9% were HBeAg positive, median HBV DNA levels were 3.1 log10 IU/mL, and the median ALT level was 25 U/L. In the external cohort, the mean age was 46 years, 21% were HBeAg positive, median HBV DNA levels were 3.4 log10 IU/mL, and the median ALT level was 20 U/L.
In the Asan cohort, 435 patients (6.3%) developed HCC during a median follow-up of 10 years. The annual HCC incidence rate was 0.63 per 100 person-years, and the estimated cumulative probability of developing HCC at 10 years was 6.4%.
In the external cohort, 467 patients (6.3%) developed HCC during a median follow-up of 12 years. The annual HCC incidence rate was 0.42 per 100 person-years, and the estimated cumulative probability of developing HCC at 10 years was 3.1%.
Overall, the association between HBV viral load and HCC risk was linear in the HBeAg-negative groups and inverse in the HBeAg-positive groups, with the association between HBV viral load and HCC risk showing a nonlinear parabolic pattern.
Across both cohorts, patients with HBV DNA levels between 5 and 6 log10 IU/mL had the highest risk for HCC in both the HBeAg-negative and HBeAg-positive groups, which was more than eight times higher than those HBV DNA levels ≤ 3 log10 IU/mL.
For internal validation, the Revised REACH-B model had a c-statistic of 0.844 and 5-year area under the curve of 0.864. For external validation across the three external cohorts, the reREACH-B had c-statistics of 0.804, 0.808, and 0.813, and 5-year area under the curve of 0.839, 0.860, and 0.865.
In addition, the revised model yielded a greater positive net benefit than the REACH-B model in the threshold probability range between 0% and 18%.
“These analyses indicate the reREACH-B model can be a valuable tool in clinical practice, aiding in timely management decisions,” Lim said.
Considering Prognostic Models
This study highlights the importance of recognizing that the association between HBV DNA viral load and HCC risk isn’t linear, said Norah Terrault, MD, chief of Gastroenterology and Hepatology at the Keck School of Medicine at the University of Southern California, Los Angeles.
“In contrast to most chronic liver diseases where liver cancer develops only among those with advanced fibrosis/cirrhosis, people with chronic hepatitis B are at risk prior to the development of cirrhosis,” she said. “Risk prediction scores for HCC can be a useful means of identifying those without cirrhosis who should be enrolled in HCC surveillance programs.”
For instance, patients with HBV DNA levels < 3 log10 IU/mL or > 8 log10 IU/mL don’t have an increased risk, Terrault noted. However, the highest risk group appears to be around 5-6 log10 IU/mL.
“Future risk prediction models should acknowledge that relationship in modeling HCC risk,” she said. “The re-REACH-B provides modest improvement over the REACH-B, but further validation of this score in more diverse cohorts is essential.”
The study received financial support from the Korean government and grants from the Patient-Centered Clinical Research Coordinating Center of the National Evidence-based Healthcare Collaborating Agency and the National R&D Program for Cancer Control through the National Cancer Center, which is funded by Korea’s Ministry of Health and Welfare. Lim and Terrault reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
The model, called Revised REACH-B or reREACH-B, stems from cohort studies in Hong Kong, South Korea, and Taiwan, and looks at the nonlinear parabolic association between serum hepatitis B virus (HBV) DNA levels and HCC risk.
“Current clinical practice guidelines don’t advocate antiviral treatment for patients with CHB who don’t show elevated alanine aminotransferase (ALT) levels, even in those with high HBV viral loads,” said coauthor Young-Suk Lim, MD, PhD, professor of gastroenterology at the University of Ulsan College of Medicine and Asan Medical Center in Seoul, South Korea.
“This stance is rooted in the notion that patients in the immune-tolerant phase are at very low risk for developing HCC,” Lim said. “However, the immune-tolerant phase includes patients with HBV DNA levels who face the highest risk for HCC, and many patients with moderate HBV viremia fall into an undefined gray zone.”
The study was published in Annals of Internal Medicine.
Validating reREACH-B
During a course of CHB, HBV viral loads and HCC risks evolve over time because of viral replication and host immune responses, Lim explained. Most patients typically move to seroclearance and an “inactive hepatitis” phase, but about 10%-20% can progress to a “reactivation” phase, where HBV DNA levels and ALT levels increase, which can increase HCC risk as well.
In a previous cohort study in Taiwan, a prognostic model called Risk Estimation for HCC in CHB — or REACH-B — found the risk for HCC increases tenfold with increasing levels of HBV DNA up to 5 log10IU/mL in noncirrhotic patients with CHB, regardless of ALT levels. Another cohort study in South Korea found a nonlinear parabolic association between HCC risk and HBV DNA levels up to 9 log10 IU/mL, with the highest risks found for moderate HBV DNA levels around 6 log10 IU/mL.
In this study, Lim and colleagues developed a prognostic model to integrate the nonlinear relationship and validated it externally, as well as compared it with the previous REACH-B model. The Revised REACH-B model incorporates six variables: age, sex, platelet count, HBV DNA level, ALT, and hepatitis B e-antigen (HBeAg).
The study included 14,378 treatment-naive, noncirrhotic adults with CHB and serum ALT levels < two times the upper limit of normal for at least 1 year and serum hepatitis B surface antigen for at least 6 months. The internal validation cohort included 6,949 patients from Asan Medical Center, and the external validation cohort included 7,429 patients from previous studies in Hong Kong, South Korea, and Taiwan.
Among the Asan cohort, the mean age was 45 years, 29.9% were HBeAg positive, median HBV DNA levels were 3.1 log10 IU/mL, and the median ALT level was 25 U/L. In the external cohort, the mean age was 46 years, 21% were HBeAg positive, median HBV DNA levels were 3.4 log10 IU/mL, and the median ALT level was 20 U/L.
In the Asan cohort, 435 patients (6.3%) developed HCC during a median follow-up of 10 years. The annual HCC incidence rate was 0.63 per 100 person-years, and the estimated cumulative probability of developing HCC at 10 years was 6.4%.
In the external cohort, 467 patients (6.3%) developed HCC during a median follow-up of 12 years. The annual HCC incidence rate was 0.42 per 100 person-years, and the estimated cumulative probability of developing HCC at 10 years was 3.1%.
Overall, the association between HBV viral load and HCC risk was linear in the HBeAg-negative groups and inverse in the HBeAg-positive groups, with the association between HBV viral load and HCC risk showing a nonlinear parabolic pattern.
Across both cohorts, patients with HBV DNA levels between 5 and 6 log10 IU/mL had the highest risk for HCC in both the HBeAg-negative and HBeAg-positive groups, which was more than eight times higher than those HBV DNA levels ≤ 3 log10 IU/mL.
For internal validation, the Revised REACH-B model had a c-statistic of 0.844 and 5-year area under the curve of 0.864. For external validation across the three external cohorts, the reREACH-B had c-statistics of 0.804, 0.808, and 0.813, and 5-year area under the curve of 0.839, 0.860, and 0.865.
In addition, the revised model yielded a greater positive net benefit than the REACH-B model in the threshold probability range between 0% and 18%.
“These analyses indicate the reREACH-B model can be a valuable tool in clinical practice, aiding in timely management decisions,” Lim said.
Considering Prognostic Models
This study highlights the importance of recognizing that the association between HBV DNA viral load and HCC risk isn’t linear, said Norah Terrault, MD, chief of Gastroenterology and Hepatology at the Keck School of Medicine at the University of Southern California, Los Angeles.
“In contrast to most chronic liver diseases where liver cancer develops only among those with advanced fibrosis/cirrhosis, people with chronic hepatitis B are at risk prior to the development of cirrhosis,” she said. “Risk prediction scores for HCC can be a useful means of identifying those without cirrhosis who should be enrolled in HCC surveillance programs.”
For instance, patients with HBV DNA levels < 3 log10 IU/mL or > 8 log10 IU/mL don’t have an increased risk, Terrault noted. However, the highest risk group appears to be around 5-6 log10 IU/mL.
“Future risk prediction models should acknowledge that relationship in modeling HCC risk,” she said. “The re-REACH-B provides modest improvement over the REACH-B, but further validation of this score in more diverse cohorts is essential.”
The study received financial support from the Korean government and grants from the Patient-Centered Clinical Research Coordinating Center of the National Evidence-based Healthcare Collaborating Agency and the National R&D Program for Cancer Control through the National Cancer Center, which is funded by Korea’s Ministry of Health and Welfare. Lim and Terrault reported no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE
Red Wine May Not Be a Health Tonic, But Is It a Cancer Risk?
Earlier this month, US surgeon general Vivek Murthy, MD, issued an advisory, calling for alcoholic beverages to carry a warning label about cancer risk. The advisory flagged alcohol as the third leading preventable cause of cancer in the United States, after tobacco and obesity, and highlighted people’s limited awareness about the relationship between alcohol and cancer risk.
But, when it comes to cancer risk, are all types of alcohol created equal?
For many years, red wine seemed to be an outlier, with studies indicating that, in moderation, it might even be good for you. Red wine has anti-inflammatory and antioxidant properties — most notably, it contains the antioxidant resveratrol. Starting in the 1990s, research began to hint that the compound might protect against heart disease, aging, and cancer, though much of this work was done in animals or test tubes.
The idea that red wine carries health benefits, however, has been called into question more recently. A recent meta-analysis, for instance, suggests that many previous studies touting the health benefits of more moderate drinking were likely biased, potentially leading to “misleading positive health associations.” And one recent study found that alcohol consumption, largely red wine and beer, at all levels was linked to an increased risk for cardiovascular disease.
Although wine’s health halo is dwindling, there might be an exception: Cancer risk.
Overall, research shows that even light to moderate drinking increases the risk for at least seven types of cancer, but when focusing on red wine, in particular, that risk calculus can look different.
“It’s very complicated and nuanced,” said Timothy Rebbeck, PhD, professor of cancer prevention, Harvard T.H. Chan School of Public Health, Boston. “And ‘complicated and nuanced’ doesn’t work very well in public health messages.”
The Knowns About Alcohol and Cancer Risk
Some things about the relationship between alcohol and cancer risk are crystal clear. “There’s no question that alcohol is a group 1 carcinogen,” Rebbeck said. “Alcohol can cause cancer.”
Groups including the International Agency for Research on Cancer (IARC) and American Cancer Society agree that alcohol use is an established cause of seven types of cancer: Those of the oral cavity, larynx, pharynx, esophagus (squamous cell carcinoma), liver (hepatocellular carcinoma), breast, and colon/rectum. Heavy drinking — at least 8 standard drinks a week for women and 15 for men — and binge drinking — 4 or more drinks in 2 hours for women and 5 or more for men — only amplify that risk. (A “standard” drink has 14 g of alcohol, which translates to a 5-oz glass of wine.)
“We’re most concerned about high-risk drinking — more than 2 drinks a day — and/or binge drinking,” said Noelle LoConte, MD, of the Division of Hematology, Medical Oncology and Palliative Care, University of Wisconsin School of Medicine and Public Health, Madison, who authored a 2018 statement on alcohol and cancer risk from the American Society of Clinical Oncology (ASCO).
Compared with not drinking, heavy drinking is linked with a roughly fivefold increase in the risk for oral cavity, pharyngeal, and esophageal cancers, and a 61% increase in the risk for breast cancer, according to LoConte and colleagues.
Things get murkier when it comes to moderate drinking — defined as up to 1 standard drink per day for women and 2 per day for men. There is evidence, LoConte said, that moderate drinking is associated with increased cancer risks, though the magnitude is generally much less than heavier drinking.
Cancer type also matters. One analysis found that the risk for breast cancer increased with even light to moderate alcohol consumption. Compared with no drinking, light to moderate drinking has also been linked to increased risks for oral cavity, pharynx, larynx, and esophageal cancers.
As for whether the type of alcoholic beverage matters, LoConte said, there’s no clear physiological reason that wine would be less risky than beer or liquor. Research indicates that ethanol is the problematic ingredient: Once ingested, it’s metabolized into acetaldehyde, a DNA-damaging substance that’s considered a probable human carcinogen. Ethanol can also alter circulating levels of estrogens and androgens, LoConte said, which is thought to drive its association with breast cancer risk.
“It likely doesn’t matter how you choose to get your ethanol,” she said. “It’s a question of volume.”
Hints That Wine Is an Outlier
Still, some studies suggest that how people ingest ethanol could make a difference.
A study published in August in JAMA Network Open is a case in point. The study found that, among older adults, light to heavy drinkers had an increased risk of dying from cancer, compared with occasional drinkers (though the increased risk among light to moderate drinkers occurred only among people who also had chronic health conditions, such as diabetes or high blood pressure, or were of lower socioeconomic status).
Wine drinkers fared differently. Most notably, drinkers who “preferred” wine — consuming over 80% of total ethanol from wine — or those who drank only with meals showed a small reduction in their risk for cancer mortality and all-cause mortality (hazard ratio [HR], 0.94 for both). The small protective association was somewhat stronger among people who reported both patterns (HR, 0.88), especially if they were of lower socioeconomic status (HR, 0.79).
The findings are in line with other research suggesting that wine drinkers may be outliers when it comes to cancer risk. A 2023 meta-analysis of 26 observational studies, for instance, found no association between wine consumption and any cancer type, with the caveat that there was «substantial» heterogeneity among the studies.
This heterogeneity caveat speaks to the inherent limitations of observational research, said Tim Stockwell, PhD, of the Canadian Institute for Substance Use Research, University of Victoria in British Columbia, Canada.
“Individual studies of alcohol and cancer risk do find differences by type of drink, or patterns of drinking,” Stockwell said. “But it’s so hard to unpack the confounding that goes along with the type of person who’s a wine drinker or a beer drinker or a spirit drinker. The beverage of choice seems to come with a lot of baggage.”
Compared with people who favor beer or liquor, he noted, wine aficionados are typically higher-income, exercise more often, smoke less, and have different diets, for example. The “best” studies, Rebbeck said, try to adjust for those differences, but it’s challenging.
The authors of the 2023 meta-analysis noted that “many components in wine could have anticarcinogenic effects” that theoretically could counter the ill effects of ethanol. Besides resveratrol, which is mainly found in red wine, the list includes anthocyanins, quercetin, and tannins. However, the authors also acknowledged that they couldn’t account for whether other lifestyle habits might explain why wine drinkers, overall, showed no increased cancer risks and sometimes lower risks.
Still, groups such as the IARC and ASCO hold that there is no known “safe” level, or type, of alcohol when it comes to cancer.
In the latest Canadian guidelines on alcohol use, the scientific panel calculated that people who have 6 drinks a week throughout adulthood (whatever the source of the alcohol) could shave 11 weeks from their life expectancy, on average, said Stockwell, who was on the guideline panel. Compare that with heavy drinking, where 4 drinks a day could rob the average person of 2 or 3 years. “If you’re drinking a lot, you could get huge benefits from cutting down,” Stockwell explained. “If you’re a moderate drinker, the benefits would obviously be less.”
Stockwell said that choices around drinking and breast cancer risk, specifically, can be “tough.” Unlike many of the other alcohol-associated cancers, he noted, breast cancer is common — so even small relative risk increases may be concerning. Based on a 2020 meta-analysis of 22 cohort studies, the risk for breast cancer rises by about 10%, on average, for every 10 g of alcohol a woman drinks per day. This study also found no evidence that wine is any different from other types of alcohol.
In real life, the calculus around wine consumption and cancer risk will probably vary widely from person to person, Rebbeck said. One woman with a family history of breast cancer might decide that having wine with dinner isn’t worth it. Another with the same family history might see that glass of wine as a stress reliever and opt to focus on other ways to reduce her breast cancer risk — by exercising and maintaining a healthy weight, for example.
“The bottom line is, in human studies, the data on light to moderate drinking and cancer are limited and messy, and you can’t draw firm conclusions from them,” Rebbeck said. “It probably raises risk in some people, but we don’t know who those people are. And the risk increases are relatively small.”
A Conversation Few Are Having
Even with many studies highlighting the connection between alcohol consumption and cancer risk, most people remain unaware about this risk.
A 2023 study by the National Cancer Institute found that only a minority of US adults knew that drinking alcohol is linked to increased cancer risk, and they were much less likely to say that was true of wine: Only 20% did, vs 31% who said that liquor can boost cancer risk. Meanwhile, 10% believed that wine helps prevent cancer. Other studies show that even among cancer survivors and patients undergoing active cancer treatment, many drink — often heavily.
“What we know right now is, physicians almost never talk about this,” LoConte said.
That could be due to time constraints, according to Rebbeck, or clinicians’ perceptions that the subject is too complicated and/or their own confusion about the data. There could also be some “cognitive dissonance” at play, LoConte noted, because many doctors drink alcohol.
It’s critical, she said, that conversations about drinking habits become “normalized,” and that should include informing patients that alcohol use is associated with certain cancers. Again, LoConte said, it’s high-risk drinking that’s most concerning and where reducing intake could have the biggest impact on cancer risk and other health outcomes.
“From a cancer prevention standpoint, it’s probably best not to drink,” she said. “But people don’t make choices based solely on cancer risk. We don’t want to come out with recommendations saying no one should drink. I don’t think the data support that, and people would buck against that advice.”
Rebbeck made a similar point. Even if there’s uncertainty about the risks for a daily glass of wine, he said, people can use that information to make decisions. “Everybody’s preferences and choices are going to be different,” Rebbeck said. “And that’s all we can really do.”
A version of this article appeared on Medscape.com.
Earlier this month, US surgeon general Vivek Murthy, MD, issued an advisory, calling for alcoholic beverages to carry a warning label about cancer risk. The advisory flagged alcohol as the third leading preventable cause of cancer in the United States, after tobacco and obesity, and highlighted people’s limited awareness about the relationship between alcohol and cancer risk.
But, when it comes to cancer risk, are all types of alcohol created equal?
For many years, red wine seemed to be an outlier, with studies indicating that, in moderation, it might even be good for you. Red wine has anti-inflammatory and antioxidant properties — most notably, it contains the antioxidant resveratrol. Starting in the 1990s, research began to hint that the compound might protect against heart disease, aging, and cancer, though much of this work was done in animals or test tubes.
The idea that red wine carries health benefits, however, has been called into question more recently. A recent meta-analysis, for instance, suggests that many previous studies touting the health benefits of more moderate drinking were likely biased, potentially leading to “misleading positive health associations.” And one recent study found that alcohol consumption, largely red wine and beer, at all levels was linked to an increased risk for cardiovascular disease.
Although wine’s health halo is dwindling, there might be an exception: Cancer risk.
Overall, research shows that even light to moderate drinking increases the risk for at least seven types of cancer, but when focusing on red wine, in particular, that risk calculus can look different.
“It’s very complicated and nuanced,” said Timothy Rebbeck, PhD, professor of cancer prevention, Harvard T.H. Chan School of Public Health, Boston. “And ‘complicated and nuanced’ doesn’t work very well in public health messages.”
The Knowns About Alcohol and Cancer Risk
Some things about the relationship between alcohol and cancer risk are crystal clear. “There’s no question that alcohol is a group 1 carcinogen,” Rebbeck said. “Alcohol can cause cancer.”
Groups including the International Agency for Research on Cancer (IARC) and American Cancer Society agree that alcohol use is an established cause of seven types of cancer: Those of the oral cavity, larynx, pharynx, esophagus (squamous cell carcinoma), liver (hepatocellular carcinoma), breast, and colon/rectum. Heavy drinking — at least 8 standard drinks a week for women and 15 for men — and binge drinking — 4 or more drinks in 2 hours for women and 5 or more for men — only amplify that risk. (A “standard” drink has 14 g of alcohol, which translates to a 5-oz glass of wine.)
“We’re most concerned about high-risk drinking — more than 2 drinks a day — and/or binge drinking,” said Noelle LoConte, MD, of the Division of Hematology, Medical Oncology and Palliative Care, University of Wisconsin School of Medicine and Public Health, Madison, who authored a 2018 statement on alcohol and cancer risk from the American Society of Clinical Oncology (ASCO).
Compared with not drinking, heavy drinking is linked with a roughly fivefold increase in the risk for oral cavity, pharyngeal, and esophageal cancers, and a 61% increase in the risk for breast cancer, according to LoConte and colleagues.
Things get murkier when it comes to moderate drinking — defined as up to 1 standard drink per day for women and 2 per day for men. There is evidence, LoConte said, that moderate drinking is associated with increased cancer risks, though the magnitude is generally much less than heavier drinking.
Cancer type also matters. One analysis found that the risk for breast cancer increased with even light to moderate alcohol consumption. Compared with no drinking, light to moderate drinking has also been linked to increased risks for oral cavity, pharynx, larynx, and esophageal cancers.
As for whether the type of alcoholic beverage matters, LoConte said, there’s no clear physiological reason that wine would be less risky than beer or liquor. Research indicates that ethanol is the problematic ingredient: Once ingested, it’s metabolized into acetaldehyde, a DNA-damaging substance that’s considered a probable human carcinogen. Ethanol can also alter circulating levels of estrogens and androgens, LoConte said, which is thought to drive its association with breast cancer risk.
“It likely doesn’t matter how you choose to get your ethanol,” she said. “It’s a question of volume.”
Hints That Wine Is an Outlier
Still, some studies suggest that how people ingest ethanol could make a difference.
A study published in August in JAMA Network Open is a case in point. The study found that, among older adults, light to heavy drinkers had an increased risk of dying from cancer, compared with occasional drinkers (though the increased risk among light to moderate drinkers occurred only among people who also had chronic health conditions, such as diabetes or high blood pressure, or were of lower socioeconomic status).
Wine drinkers fared differently. Most notably, drinkers who “preferred” wine — consuming over 80% of total ethanol from wine — or those who drank only with meals showed a small reduction in their risk for cancer mortality and all-cause mortality (hazard ratio [HR], 0.94 for both). The small protective association was somewhat stronger among people who reported both patterns (HR, 0.88), especially if they were of lower socioeconomic status (HR, 0.79).
The findings are in line with other research suggesting that wine drinkers may be outliers when it comes to cancer risk. A 2023 meta-analysis of 26 observational studies, for instance, found no association between wine consumption and any cancer type, with the caveat that there was «substantial» heterogeneity among the studies.
This heterogeneity caveat speaks to the inherent limitations of observational research, said Tim Stockwell, PhD, of the Canadian Institute for Substance Use Research, University of Victoria in British Columbia, Canada.
“Individual studies of alcohol and cancer risk do find differences by type of drink, or patterns of drinking,” Stockwell said. “But it’s so hard to unpack the confounding that goes along with the type of person who’s a wine drinker or a beer drinker or a spirit drinker. The beverage of choice seems to come with a lot of baggage.”
Compared with people who favor beer or liquor, he noted, wine aficionados are typically higher-income, exercise more often, smoke less, and have different diets, for example. The “best” studies, Rebbeck said, try to adjust for those differences, but it’s challenging.
The authors of the 2023 meta-analysis noted that “many components in wine could have anticarcinogenic effects” that theoretically could counter the ill effects of ethanol. Besides resveratrol, which is mainly found in red wine, the list includes anthocyanins, quercetin, and tannins. However, the authors also acknowledged that they couldn’t account for whether other lifestyle habits might explain why wine drinkers, overall, showed no increased cancer risks and sometimes lower risks.
Still, groups such as the IARC and ASCO hold that there is no known “safe” level, or type, of alcohol when it comes to cancer.
In the latest Canadian guidelines on alcohol use, the scientific panel calculated that people who have 6 drinks a week throughout adulthood (whatever the source of the alcohol) could shave 11 weeks from their life expectancy, on average, said Stockwell, who was on the guideline panel. Compare that with heavy drinking, where 4 drinks a day could rob the average person of 2 or 3 years. “If you’re drinking a lot, you could get huge benefits from cutting down,” Stockwell explained. “If you’re a moderate drinker, the benefits would obviously be less.”
Stockwell said that choices around drinking and breast cancer risk, specifically, can be “tough.” Unlike many of the other alcohol-associated cancers, he noted, breast cancer is common — so even small relative risk increases may be concerning. Based on a 2020 meta-analysis of 22 cohort studies, the risk for breast cancer rises by about 10%, on average, for every 10 g of alcohol a woman drinks per day. This study also found no evidence that wine is any different from other types of alcohol.
In real life, the calculus around wine consumption and cancer risk will probably vary widely from person to person, Rebbeck said. One woman with a family history of breast cancer might decide that having wine with dinner isn’t worth it. Another with the same family history might see that glass of wine as a stress reliever and opt to focus on other ways to reduce her breast cancer risk — by exercising and maintaining a healthy weight, for example.
“The bottom line is, in human studies, the data on light to moderate drinking and cancer are limited and messy, and you can’t draw firm conclusions from them,” Rebbeck said. “It probably raises risk in some people, but we don’t know who those people are. And the risk increases are relatively small.”
A Conversation Few Are Having
Even with many studies highlighting the connection between alcohol consumption and cancer risk, most people remain unaware about this risk.
A 2023 study by the National Cancer Institute found that only a minority of US adults knew that drinking alcohol is linked to increased cancer risk, and they were much less likely to say that was true of wine: Only 20% did, vs 31% who said that liquor can boost cancer risk. Meanwhile, 10% believed that wine helps prevent cancer. Other studies show that even among cancer survivors and patients undergoing active cancer treatment, many drink — often heavily.
“What we know right now is, physicians almost never talk about this,” LoConte said.
That could be due to time constraints, according to Rebbeck, or clinicians’ perceptions that the subject is too complicated and/or their own confusion about the data. There could also be some “cognitive dissonance” at play, LoConte noted, because many doctors drink alcohol.
It’s critical, she said, that conversations about drinking habits become “normalized,” and that should include informing patients that alcohol use is associated with certain cancers. Again, LoConte said, it’s high-risk drinking that’s most concerning and where reducing intake could have the biggest impact on cancer risk and other health outcomes.
“From a cancer prevention standpoint, it’s probably best not to drink,” she said. “But people don’t make choices based solely on cancer risk. We don’t want to come out with recommendations saying no one should drink. I don’t think the data support that, and people would buck against that advice.”
Rebbeck made a similar point. Even if there’s uncertainty about the risks for a daily glass of wine, he said, people can use that information to make decisions. “Everybody’s preferences and choices are going to be different,” Rebbeck said. “And that’s all we can really do.”
A version of this article appeared on Medscape.com.
Earlier this month, US surgeon general Vivek Murthy, MD, issued an advisory, calling for alcoholic beverages to carry a warning label about cancer risk. The advisory flagged alcohol as the third leading preventable cause of cancer in the United States, after tobacco and obesity, and highlighted people’s limited awareness about the relationship between alcohol and cancer risk.
But, when it comes to cancer risk, are all types of alcohol created equal?
For many years, red wine seemed to be an outlier, with studies indicating that, in moderation, it might even be good for you. Red wine has anti-inflammatory and antioxidant properties — most notably, it contains the antioxidant resveratrol. Starting in the 1990s, research began to hint that the compound might protect against heart disease, aging, and cancer, though much of this work was done in animals or test tubes.
The idea that red wine carries health benefits, however, has been called into question more recently. A recent meta-analysis, for instance, suggests that many previous studies touting the health benefits of more moderate drinking were likely biased, potentially leading to “misleading positive health associations.” And one recent study found that alcohol consumption, largely red wine and beer, at all levels was linked to an increased risk for cardiovascular disease.
Although wine’s health halo is dwindling, there might be an exception: Cancer risk.
Overall, research shows that even light to moderate drinking increases the risk for at least seven types of cancer, but when focusing on red wine, in particular, that risk calculus can look different.
“It’s very complicated and nuanced,” said Timothy Rebbeck, PhD, professor of cancer prevention, Harvard T.H. Chan School of Public Health, Boston. “And ‘complicated and nuanced’ doesn’t work very well in public health messages.”
The Knowns About Alcohol and Cancer Risk
Some things about the relationship between alcohol and cancer risk are crystal clear. “There’s no question that alcohol is a group 1 carcinogen,” Rebbeck said. “Alcohol can cause cancer.”
Groups including the International Agency for Research on Cancer (IARC) and American Cancer Society agree that alcohol use is an established cause of seven types of cancer: Those of the oral cavity, larynx, pharynx, esophagus (squamous cell carcinoma), liver (hepatocellular carcinoma), breast, and colon/rectum. Heavy drinking — at least 8 standard drinks a week for women and 15 for men — and binge drinking — 4 or more drinks in 2 hours for women and 5 or more for men — only amplify that risk. (A “standard” drink has 14 g of alcohol, which translates to a 5-oz glass of wine.)
“We’re most concerned about high-risk drinking — more than 2 drinks a day — and/or binge drinking,” said Noelle LoConte, MD, of the Division of Hematology, Medical Oncology and Palliative Care, University of Wisconsin School of Medicine and Public Health, Madison, who authored a 2018 statement on alcohol and cancer risk from the American Society of Clinical Oncology (ASCO).
Compared with not drinking, heavy drinking is linked with a roughly fivefold increase in the risk for oral cavity, pharyngeal, and esophageal cancers, and a 61% increase in the risk for breast cancer, according to LoConte and colleagues.
Things get murkier when it comes to moderate drinking — defined as up to 1 standard drink per day for women and 2 per day for men. There is evidence, LoConte said, that moderate drinking is associated with increased cancer risks, though the magnitude is generally much less than heavier drinking.
Cancer type also matters. One analysis found that the risk for breast cancer increased with even light to moderate alcohol consumption. Compared with no drinking, light to moderate drinking has also been linked to increased risks for oral cavity, pharynx, larynx, and esophageal cancers.
As for whether the type of alcoholic beverage matters, LoConte said, there’s no clear physiological reason that wine would be less risky than beer or liquor. Research indicates that ethanol is the problematic ingredient: Once ingested, it’s metabolized into acetaldehyde, a DNA-damaging substance that’s considered a probable human carcinogen. Ethanol can also alter circulating levels of estrogens and androgens, LoConte said, which is thought to drive its association with breast cancer risk.
“It likely doesn’t matter how you choose to get your ethanol,” she said. “It’s a question of volume.”
Hints That Wine Is an Outlier
Still, some studies suggest that how people ingest ethanol could make a difference.
A study published in August in JAMA Network Open is a case in point. The study found that, among older adults, light to heavy drinkers had an increased risk of dying from cancer, compared with occasional drinkers (though the increased risk among light to moderate drinkers occurred only among people who also had chronic health conditions, such as diabetes or high blood pressure, or were of lower socioeconomic status).
Wine drinkers fared differently. Most notably, drinkers who “preferred” wine — consuming over 80% of total ethanol from wine — or those who drank only with meals showed a small reduction in their risk for cancer mortality and all-cause mortality (hazard ratio [HR], 0.94 for both). The small protective association was somewhat stronger among people who reported both patterns (HR, 0.88), especially if they were of lower socioeconomic status (HR, 0.79).
The findings are in line with other research suggesting that wine drinkers may be outliers when it comes to cancer risk. A 2023 meta-analysis of 26 observational studies, for instance, found no association between wine consumption and any cancer type, with the caveat that there was «substantial» heterogeneity among the studies.
This heterogeneity caveat speaks to the inherent limitations of observational research, said Tim Stockwell, PhD, of the Canadian Institute for Substance Use Research, University of Victoria in British Columbia, Canada.
“Individual studies of alcohol and cancer risk do find differences by type of drink, or patterns of drinking,” Stockwell said. “But it’s so hard to unpack the confounding that goes along with the type of person who’s a wine drinker or a beer drinker or a spirit drinker. The beverage of choice seems to come with a lot of baggage.”
Compared with people who favor beer or liquor, he noted, wine aficionados are typically higher-income, exercise more often, smoke less, and have different diets, for example. The “best” studies, Rebbeck said, try to adjust for those differences, but it’s challenging.
The authors of the 2023 meta-analysis noted that “many components in wine could have anticarcinogenic effects” that theoretically could counter the ill effects of ethanol. Besides resveratrol, which is mainly found in red wine, the list includes anthocyanins, quercetin, and tannins. However, the authors also acknowledged that they couldn’t account for whether other lifestyle habits might explain why wine drinkers, overall, showed no increased cancer risks and sometimes lower risks.
Still, groups such as the IARC and ASCO hold that there is no known “safe” level, or type, of alcohol when it comes to cancer.
In the latest Canadian guidelines on alcohol use, the scientific panel calculated that people who have 6 drinks a week throughout adulthood (whatever the source of the alcohol) could shave 11 weeks from their life expectancy, on average, said Stockwell, who was on the guideline panel. Compare that with heavy drinking, where 4 drinks a day could rob the average person of 2 or 3 years. “If you’re drinking a lot, you could get huge benefits from cutting down,” Stockwell explained. “If you’re a moderate drinker, the benefits would obviously be less.”
Stockwell said that choices around drinking and breast cancer risk, specifically, can be “tough.” Unlike many of the other alcohol-associated cancers, he noted, breast cancer is common — so even small relative risk increases may be concerning. Based on a 2020 meta-analysis of 22 cohort studies, the risk for breast cancer rises by about 10%, on average, for every 10 g of alcohol a woman drinks per day. This study also found no evidence that wine is any different from other types of alcohol.
In real life, the calculus around wine consumption and cancer risk will probably vary widely from person to person, Rebbeck said. One woman with a family history of breast cancer might decide that having wine with dinner isn’t worth it. Another with the same family history might see that glass of wine as a stress reliever and opt to focus on other ways to reduce her breast cancer risk — by exercising and maintaining a healthy weight, for example.
“The bottom line is, in human studies, the data on light to moderate drinking and cancer are limited and messy, and you can’t draw firm conclusions from them,” Rebbeck said. “It probably raises risk in some people, but we don’t know who those people are. And the risk increases are relatively small.”
A Conversation Few Are Having
Even with many studies highlighting the connection between alcohol consumption and cancer risk, most people remain unaware about this risk.
A 2023 study by the National Cancer Institute found that only a minority of US adults knew that drinking alcohol is linked to increased cancer risk, and they were much less likely to say that was true of wine: Only 20% did, vs 31% who said that liquor can boost cancer risk. Meanwhile, 10% believed that wine helps prevent cancer. Other studies show that even among cancer survivors and patients undergoing active cancer treatment, many drink — often heavily.
“What we know right now is, physicians almost never talk about this,” LoConte said.
That could be due to time constraints, according to Rebbeck, or clinicians’ perceptions that the subject is too complicated and/or their own confusion about the data. There could also be some “cognitive dissonance” at play, LoConte noted, because many doctors drink alcohol.
It’s critical, she said, that conversations about drinking habits become “normalized,” and that should include informing patients that alcohol use is associated with certain cancers. Again, LoConte said, it’s high-risk drinking that’s most concerning and where reducing intake could have the biggest impact on cancer risk and other health outcomes.
“From a cancer prevention standpoint, it’s probably best not to drink,” she said. “But people don’t make choices based solely on cancer risk. We don’t want to come out with recommendations saying no one should drink. I don’t think the data support that, and people would buck against that advice.”
Rebbeck made a similar point. Even if there’s uncertainty about the risks for a daily glass of wine, he said, people can use that information to make decisions. “Everybody’s preferences and choices are going to be different,” Rebbeck said. “And that’s all we can really do.”
A version of this article appeared on Medscape.com.
Dietary Calcium Cuts Colorectal Cancer Risk by 17%
Cancer Research UK (CRUK), which funded the study, said that it demonstrated the benefits of a healthy, balanced diet for lowering cancer risk.
Colorectal cancer is the third most common cancer worldwide. Incidence rates vary markedly, with higher rates observed in high-income countries. The risk increases for individuals who migrate from low- to high-incidence areas, suggesting that lifestyle and environmental factors contribute to its development.
While alcohol and processed meats are established carcinogens, and red meat is classified as probably carcinogenic, there is a lack of consensus regarding the relationships between other dietary factors and colorectal cancer risk. This uncertainty may be due, at least in part, to relatively few studies giving comprehensive results on all food types, as well as dietary measurement errors, and/or small sample sizes.
Study Tracked 97 Dietary Factors
To address these gaps, the research team, led by the University of Oxford in England, tracked the intake of 97 dietary factors in 542,778 women from 2001 for an average of 16.6 years. During this period 12,251 participants developed colorectal cancer. The women completed detailed dietary questionnaires at baseline, with 7% participating in at least one subsequent 24-hour online dietary assessment.
Women diagnosed with colorectal cancer were generally older, taller, more likely to have a family history of bowel cancer, and have more adverse health behaviors, compared with participants overall.
Calcium Intake Showed the Strongest Protective Association
Relative risks (RR) for colorectal cancer were calculated for intakes of all 97 dietary factors, with significant associations found for 17 of them. Calcium intake showed the strongest protective effect, with each additional 300 mg per day – equivalent to a large glass of milk – associated with a 17% reduced RR.
Six dairy-related factors associated with calcium – dairy milk, yogurt, riboflavin, magnesium, phosphorus, and potassium intakes – also demonstrated inverse associations with colorectal cancer risk. Weaker protective effects were noted for breakfast cereal, fruit, wholegrains, carbohydrates, fibre, total sugars, folate, and vitamin C. However, the team commented that these inverse associations might reflect residual confounding from other lifestyle or other dietary factors.
Calcium’s protective role was independent of dairy milk intake. The study, published in Nature Communications, concluded that, while “dairy products help protect against colorectal cancer,” that protection is “driven largely or wholly by calcium.”
Alcohol and Processed Meat Confirmed as Risk Factors
As expected, alcohol showed the reverse association, with each additional 20 g daily – equivalent to one large glass of wine – associated with a 15% RR increase. Weaker associations were seen for the combined category of red and processed meat, with each additional 30 g per day associated with an 8% increased RR for colorectal cancer. This association was minimally affected by diet and lifestyle factors.
Commenting to the Science Media Centre (SMC), Tom Sanders, professor emeritus of nutrition and dietetics at King’s College London, England, said: “One theory is that the calcium may bind to free bile acids in the gut, preventing the harmful effects of free bile acids on gut mucosa.” However, the lactose content in milk also has effects on large bowel microflora, which may in turn affect risk.
Also commenting to the SMC, David Nunan, senior research fellow at the University of Oxford’s Centre for Evidence Based Medicine, who was not involved in the study, cautioned that the findings were subject to the bias inherent in observational studies. “These biases often inflate the estimated associations compared to controlled experiments,” he said. Nunan advised caution in interpreting the findings, as more robust research, such as randomized controlled trials, would be needed to establish causation.
A version of this article first appeared on Medscape.com.
Cancer Research UK (CRUK), which funded the study, said that it demonstrated the benefits of a healthy, balanced diet for lowering cancer risk.
Colorectal cancer is the third most common cancer worldwide. Incidence rates vary markedly, with higher rates observed in high-income countries. The risk increases for individuals who migrate from low- to high-incidence areas, suggesting that lifestyle and environmental factors contribute to its development.
While alcohol and processed meats are established carcinogens, and red meat is classified as probably carcinogenic, there is a lack of consensus regarding the relationships between other dietary factors and colorectal cancer risk. This uncertainty may be due, at least in part, to relatively few studies giving comprehensive results on all food types, as well as dietary measurement errors, and/or small sample sizes.
Study Tracked 97 Dietary Factors
To address these gaps, the research team, led by the University of Oxford in England, tracked the intake of 97 dietary factors in 542,778 women from 2001 for an average of 16.6 years. During this period 12,251 participants developed colorectal cancer. The women completed detailed dietary questionnaires at baseline, with 7% participating in at least one subsequent 24-hour online dietary assessment.
Women diagnosed with colorectal cancer were generally older, taller, more likely to have a family history of bowel cancer, and have more adverse health behaviors, compared with participants overall.
Calcium Intake Showed the Strongest Protective Association
Relative risks (RR) for colorectal cancer were calculated for intakes of all 97 dietary factors, with significant associations found for 17 of them. Calcium intake showed the strongest protective effect, with each additional 300 mg per day – equivalent to a large glass of milk – associated with a 17% reduced RR.
Six dairy-related factors associated with calcium – dairy milk, yogurt, riboflavin, magnesium, phosphorus, and potassium intakes – also demonstrated inverse associations with colorectal cancer risk. Weaker protective effects were noted for breakfast cereal, fruit, wholegrains, carbohydrates, fibre, total sugars, folate, and vitamin C. However, the team commented that these inverse associations might reflect residual confounding from other lifestyle or other dietary factors.
Calcium’s protective role was independent of dairy milk intake. The study, published in Nature Communications, concluded that, while “dairy products help protect against colorectal cancer,” that protection is “driven largely or wholly by calcium.”
Alcohol and Processed Meat Confirmed as Risk Factors
As expected, alcohol showed the reverse association, with each additional 20 g daily – equivalent to one large glass of wine – associated with a 15% RR increase. Weaker associations were seen for the combined category of red and processed meat, with each additional 30 g per day associated with an 8% increased RR for colorectal cancer. This association was minimally affected by diet and lifestyle factors.
Commenting to the Science Media Centre (SMC), Tom Sanders, professor emeritus of nutrition and dietetics at King’s College London, England, said: “One theory is that the calcium may bind to free bile acids in the gut, preventing the harmful effects of free bile acids on gut mucosa.” However, the lactose content in milk also has effects on large bowel microflora, which may in turn affect risk.
Also commenting to the SMC, David Nunan, senior research fellow at the University of Oxford’s Centre for Evidence Based Medicine, who was not involved in the study, cautioned that the findings were subject to the bias inherent in observational studies. “These biases often inflate the estimated associations compared to controlled experiments,” he said. Nunan advised caution in interpreting the findings, as more robust research, such as randomized controlled trials, would be needed to establish causation.
A version of this article first appeared on Medscape.com.
Cancer Research UK (CRUK), which funded the study, said that it demonstrated the benefits of a healthy, balanced diet for lowering cancer risk.
Colorectal cancer is the third most common cancer worldwide. Incidence rates vary markedly, with higher rates observed in high-income countries. The risk increases for individuals who migrate from low- to high-incidence areas, suggesting that lifestyle and environmental factors contribute to its development.
While alcohol and processed meats are established carcinogens, and red meat is classified as probably carcinogenic, there is a lack of consensus regarding the relationships between other dietary factors and colorectal cancer risk. This uncertainty may be due, at least in part, to relatively few studies giving comprehensive results on all food types, as well as dietary measurement errors, and/or small sample sizes.
Study Tracked 97 Dietary Factors
To address these gaps, the research team, led by the University of Oxford in England, tracked the intake of 97 dietary factors in 542,778 women from 2001 for an average of 16.6 years. During this period 12,251 participants developed colorectal cancer. The women completed detailed dietary questionnaires at baseline, with 7% participating in at least one subsequent 24-hour online dietary assessment.
Women diagnosed with colorectal cancer were generally older, taller, more likely to have a family history of bowel cancer, and have more adverse health behaviors, compared with participants overall.
Calcium Intake Showed the Strongest Protective Association
Relative risks (RR) for colorectal cancer were calculated for intakes of all 97 dietary factors, with significant associations found for 17 of them. Calcium intake showed the strongest protective effect, with each additional 300 mg per day – equivalent to a large glass of milk – associated with a 17% reduced RR.
Six dairy-related factors associated with calcium – dairy milk, yogurt, riboflavin, magnesium, phosphorus, and potassium intakes – also demonstrated inverse associations with colorectal cancer risk. Weaker protective effects were noted for breakfast cereal, fruit, wholegrains, carbohydrates, fibre, total sugars, folate, and vitamin C. However, the team commented that these inverse associations might reflect residual confounding from other lifestyle or other dietary factors.
Calcium’s protective role was independent of dairy milk intake. The study, published in Nature Communications, concluded that, while “dairy products help protect against colorectal cancer,” that protection is “driven largely or wholly by calcium.”
Alcohol and Processed Meat Confirmed as Risk Factors
As expected, alcohol showed the reverse association, with each additional 20 g daily – equivalent to one large glass of wine – associated with a 15% RR increase. Weaker associations were seen for the combined category of red and processed meat, with each additional 30 g per day associated with an 8% increased RR for colorectal cancer. This association was minimally affected by diet and lifestyle factors.
Commenting to the Science Media Centre (SMC), Tom Sanders, professor emeritus of nutrition and dietetics at King’s College London, England, said: “One theory is that the calcium may bind to free bile acids in the gut, preventing the harmful effects of free bile acids on gut mucosa.” However, the lactose content in milk also has effects on large bowel microflora, which may in turn affect risk.
Also commenting to the SMC, David Nunan, senior research fellow at the University of Oxford’s Centre for Evidence Based Medicine, who was not involved in the study, cautioned that the findings were subject to the bias inherent in observational studies. “These biases often inflate the estimated associations compared to controlled experiments,” he said. Nunan advised caution in interpreting the findings, as more robust research, such as randomized controlled trials, would be needed to establish causation.
A version of this article first appeared on Medscape.com.
FROM NATURE COMMUNICATIONS
MRI-Invisible Prostate Lesions: Are They Dangerous?
MRI-invisible prostate lesions. It sounds like the stuff of science fiction and fantasy, a creation from the minds of H.G. Wells, who wrote The Invisible Man, or J.K. Rowling, who authored the Harry Potter series.
But MRI-invisible prostate lesions are real. And what these lesions may, or may not, indicate is the subject of intense debate.
MRI plays an increasingly important role in detecting and diagnosing prostate cancer, staging prostate cancer as well as monitoring disease progression. However, on occasion, a puzzling phenomenon arises. Certain prostate lesions that appear when pathologists examine biopsied tissue samples under a microscope are not visible on MRI. The prostate tissue will, instead, appear normal to a radiologist’s eye.
Some experts believe these MRI-invisible lesions are nothing to worry about.
If the clinician can’t see the cancer on MRI, then it simply isn’t a threat, according to Mark Emberton, MD, a pioneer in prostate MRIs and director of interventional oncology at University College London, England.
Laurence Klotz, MD, of the University of Toronto, Ontario, Canada, agreed, noting that “invisible cancers are clinically insignificant and don’t require systematic biopsies.”
Emberton and Klotz compared MRI-invisible lesions to grade group 1 prostate cancer (Gleason score ≤ 6) — the least aggressive category that indicates the cancer that is not likely to spread or kill. For patients on active surveillance, those with MRI-invisible cancers do drastically better than those with visible cancers, Klotz explained.
But other experts in the field are skeptical that MRI-invisible lesions are truly innocuous.
Although statistically an MRI-visible prostate lesion indicates a more aggressive tumor, that is not always the case for every individual, said Brian Helfand, MD, PhD, chief of urology at NorthShore University Health System, Evanston, Illinois.
MRIs can lead to false negatives in about 10%-20% of patients who have clinically significant prostate cancer, though estimates vary.
In one analysis, 16% of men with no suspicious lesions on MRI had clinically significant prostate cancer identified after undergoing a systematic biopsy. Another analysis found that about 35% of MRI-invisible prostate cancers identified via biopsy were clinically significant.
Other studies, however, have indicated that negative MRI results accurately indicate patients at low risk of developing clinically significant cancers. A recent JAMA Oncology analysis, for instance, found that only seven of 233 men (3%) with negative MRI results at baseline who completed 3 years of monitoring were diagnosed with clinically significant prostate cancer.
When a patient has an MRI-invisible prostate tumor, there are a couple of reasons the MRI may not be picking it up, said urologic oncologist Alexander Putnam Cole, MD, assistant professor of surgery, Harvard Medical School, Boston, Massachusetts. “One is that the cancer is aggressive but just very small,” said Cole.
“Another possibility is that the cancer looks very similar to background prostate tissue, which is something that you might expect if you think about more of a low-grade cancer,” he explained.
The experience level of the radiologist interpreting the MRI can also play into the accuracy of the reading.
But Cole agreed that “in general, MRI visibility is associated with molecular and histologic features of progression and aggressiveness and non-visible cancers are less likely to have aggressive features.”
The genomic profiles of MRI-visible and -invisible cancers bear this out.
According to Todd Morgan, MD, chief of urologic oncology at Michigan Medicine, University of Michigan, Ann Arbor, the gene expression in visible disease tends to be linked to more aggressive prostate tumors whereas gene expression in invisible disease does not.
In one analysis, for instance, researchers found that four genes — PHYHD1, CENPF, ALDH2, and GDF15 — associated with worse progression-free survival and metastasis-free survival in prostate cancer also predicted MRI visibility.
“Genes that are associated with visibility are essentially the same genes that are associated with aggressive cancers,” Klotz said.
Next Steps After Negative MRI Result
What do MRI-invisible lesions mean for patient care? If, for instance, a patient has elevated PSA levels but a normal MRI, is a targeted or systematic biopsy warranted?
The overarching message, according to Klotz, is that “you don’t need to find them.” Klotz noted, however, that patients with a negative MRI result should still be followed with periodic repeat imaging.
Several trials support this approach of using MRI to decide who needs a biopsy and delaying a biopsy in men with normal MRIs.
The recent JAMA Oncology analysis found that, among men with negative MRI results, 86% avoided a biopsy over 3 years, with clinically significant prostate cancer detected in only 4% of men across the study period — four in the initial diagnostic phase and seven in the 3-year monitoring phase. However, during the initial diagnostic phase, more than half the men with positive MRI findings had clinically significant prostate cancer detected.
Another recent study found that patients with negative MRI results were much less likely to upgrade to higher Gleason scores over time. Among 522 patients who underwent a systematic and targeted biopsy within 18 months of their grade group 1 designation, 9.2% with negative MRI findings had tumors reclassified as grade group 2 or higher vs 27% with positive MRI findings, and 2.3% with negative MRI findings had tumors reclassified as grade group 3 or higher vs 7.8% with positive MRI findings.
These data suggest that men with grade group 1 cancer and negative MRI result “may be able to avoid confirmatory biopsies until a routine surveillance biopsy in 2-3 years,” according to study author Christian Pavlovich, MD, professor of urologic oncology at the Johns Hopkins University School of Medicine, Baltimore.
Cole used MRI findings to triage who gets a biopsy. When a biopsy is warranted, “I usually recommend adding in some systematic sampling of the other side to assess for nonvisible cancers,” he noted.
Sampling prostate tissue outside the target area “adds maybe 1-2 minutes to the procedure and doesn’t drastically increase the morbidity or risks,” Cole said. It also can help “confirm there is cancer in the MRI target and also confirm there is no cancer in the nonvisible areas.”
According to Klotz, if imaging demonstrates progression, patients should receive a biopsy — in most cases, a targeted biopsy only. And, Klotz noted, skipping routine prostate biopsies in men with negative MRI results can save thousands of men from these procedures, which carry risks for infections and sepsis.
Looking beyond Gleason scores for risk prediction, MRI “visibility is a very powerful risk stratifier,” he said.
A version of this article appeared on Medscape.com.
MRI-invisible prostate lesions. It sounds like the stuff of science fiction and fantasy, a creation from the minds of H.G. Wells, who wrote The Invisible Man, or J.K. Rowling, who authored the Harry Potter series.
But MRI-invisible prostate lesions are real. And what these lesions may, or may not, indicate is the subject of intense debate.
MRI plays an increasingly important role in detecting and diagnosing prostate cancer, staging prostate cancer as well as monitoring disease progression. However, on occasion, a puzzling phenomenon arises. Certain prostate lesions that appear when pathologists examine biopsied tissue samples under a microscope are not visible on MRI. The prostate tissue will, instead, appear normal to a radiologist’s eye.
Some experts believe these MRI-invisible lesions are nothing to worry about.
If the clinician can’t see the cancer on MRI, then it simply isn’t a threat, according to Mark Emberton, MD, a pioneer in prostate MRIs and director of interventional oncology at University College London, England.
Laurence Klotz, MD, of the University of Toronto, Ontario, Canada, agreed, noting that “invisible cancers are clinically insignificant and don’t require systematic biopsies.”
Emberton and Klotz compared MRI-invisible lesions to grade group 1 prostate cancer (Gleason score ≤ 6) — the least aggressive category that indicates the cancer that is not likely to spread or kill. For patients on active surveillance, those with MRI-invisible cancers do drastically better than those with visible cancers, Klotz explained.
But other experts in the field are skeptical that MRI-invisible lesions are truly innocuous.
Although statistically an MRI-visible prostate lesion indicates a more aggressive tumor, that is not always the case for every individual, said Brian Helfand, MD, PhD, chief of urology at NorthShore University Health System, Evanston, Illinois.
MRIs can lead to false negatives in about 10%-20% of patients who have clinically significant prostate cancer, though estimates vary.
In one analysis, 16% of men with no suspicious lesions on MRI had clinically significant prostate cancer identified after undergoing a systematic biopsy. Another analysis found that about 35% of MRI-invisible prostate cancers identified via biopsy were clinically significant.
Other studies, however, have indicated that negative MRI results accurately indicate patients at low risk of developing clinically significant cancers. A recent JAMA Oncology analysis, for instance, found that only seven of 233 men (3%) with negative MRI results at baseline who completed 3 years of monitoring were diagnosed with clinically significant prostate cancer.
When a patient has an MRI-invisible prostate tumor, there are a couple of reasons the MRI may not be picking it up, said urologic oncologist Alexander Putnam Cole, MD, assistant professor of surgery, Harvard Medical School, Boston, Massachusetts. “One is that the cancer is aggressive but just very small,” said Cole.
“Another possibility is that the cancer looks very similar to background prostate tissue, which is something that you might expect if you think about more of a low-grade cancer,” he explained.
The experience level of the radiologist interpreting the MRI can also play into the accuracy of the reading.
But Cole agreed that “in general, MRI visibility is associated with molecular and histologic features of progression and aggressiveness and non-visible cancers are less likely to have aggressive features.”
The genomic profiles of MRI-visible and -invisible cancers bear this out.
According to Todd Morgan, MD, chief of urologic oncology at Michigan Medicine, University of Michigan, Ann Arbor, the gene expression in visible disease tends to be linked to more aggressive prostate tumors whereas gene expression in invisible disease does not.
In one analysis, for instance, researchers found that four genes — PHYHD1, CENPF, ALDH2, and GDF15 — associated with worse progression-free survival and metastasis-free survival in prostate cancer also predicted MRI visibility.
“Genes that are associated with visibility are essentially the same genes that are associated with aggressive cancers,” Klotz said.
Next Steps After Negative MRI Result
What do MRI-invisible lesions mean for patient care? If, for instance, a patient has elevated PSA levels but a normal MRI, is a targeted or systematic biopsy warranted?
The overarching message, according to Klotz, is that “you don’t need to find them.” Klotz noted, however, that patients with a negative MRI result should still be followed with periodic repeat imaging.
Several trials support this approach of using MRI to decide who needs a biopsy and delaying a biopsy in men with normal MRIs.
The recent JAMA Oncology analysis found that, among men with negative MRI results, 86% avoided a biopsy over 3 years, with clinically significant prostate cancer detected in only 4% of men across the study period — four in the initial diagnostic phase and seven in the 3-year monitoring phase. However, during the initial diagnostic phase, more than half the men with positive MRI findings had clinically significant prostate cancer detected.
Another recent study found that patients with negative MRI results were much less likely to upgrade to higher Gleason scores over time. Among 522 patients who underwent a systematic and targeted biopsy within 18 months of their grade group 1 designation, 9.2% with negative MRI findings had tumors reclassified as grade group 2 or higher vs 27% with positive MRI findings, and 2.3% with negative MRI findings had tumors reclassified as grade group 3 or higher vs 7.8% with positive MRI findings.
These data suggest that men with grade group 1 cancer and negative MRI result “may be able to avoid confirmatory biopsies until a routine surveillance biopsy in 2-3 years,” according to study author Christian Pavlovich, MD, professor of urologic oncology at the Johns Hopkins University School of Medicine, Baltimore.
Cole used MRI findings to triage who gets a biopsy. When a biopsy is warranted, “I usually recommend adding in some systematic sampling of the other side to assess for nonvisible cancers,” he noted.
Sampling prostate tissue outside the target area “adds maybe 1-2 minutes to the procedure and doesn’t drastically increase the morbidity or risks,” Cole said. It also can help “confirm there is cancer in the MRI target and also confirm there is no cancer in the nonvisible areas.”
According to Klotz, if imaging demonstrates progression, patients should receive a biopsy — in most cases, a targeted biopsy only. And, Klotz noted, skipping routine prostate biopsies in men with negative MRI results can save thousands of men from these procedures, which carry risks for infections and sepsis.
Looking beyond Gleason scores for risk prediction, MRI “visibility is a very powerful risk stratifier,” he said.
A version of this article appeared on Medscape.com.
MRI-invisible prostate lesions. It sounds like the stuff of science fiction and fantasy, a creation from the minds of H.G. Wells, who wrote The Invisible Man, or J.K. Rowling, who authored the Harry Potter series.
But MRI-invisible prostate lesions are real. And what these lesions may, or may not, indicate is the subject of intense debate.
MRI plays an increasingly important role in detecting and diagnosing prostate cancer, staging prostate cancer as well as monitoring disease progression. However, on occasion, a puzzling phenomenon arises. Certain prostate lesions that appear when pathologists examine biopsied tissue samples under a microscope are not visible on MRI. The prostate tissue will, instead, appear normal to a radiologist’s eye.
Some experts believe these MRI-invisible lesions are nothing to worry about.
If the clinician can’t see the cancer on MRI, then it simply isn’t a threat, according to Mark Emberton, MD, a pioneer in prostate MRIs and director of interventional oncology at University College London, England.
Laurence Klotz, MD, of the University of Toronto, Ontario, Canada, agreed, noting that “invisible cancers are clinically insignificant and don’t require systematic biopsies.”
Emberton and Klotz compared MRI-invisible lesions to grade group 1 prostate cancer (Gleason score ≤ 6) — the least aggressive category that indicates the cancer that is not likely to spread or kill. For patients on active surveillance, those with MRI-invisible cancers do drastically better than those with visible cancers, Klotz explained.
But other experts in the field are skeptical that MRI-invisible lesions are truly innocuous.
Although statistically an MRI-visible prostate lesion indicates a more aggressive tumor, that is not always the case for every individual, said Brian Helfand, MD, PhD, chief of urology at NorthShore University Health System, Evanston, Illinois.
MRIs can lead to false negatives in about 10%-20% of patients who have clinically significant prostate cancer, though estimates vary.
In one analysis, 16% of men with no suspicious lesions on MRI had clinically significant prostate cancer identified after undergoing a systematic biopsy. Another analysis found that about 35% of MRI-invisible prostate cancers identified via biopsy were clinically significant.
Other studies, however, have indicated that negative MRI results accurately indicate patients at low risk of developing clinically significant cancers. A recent JAMA Oncology analysis, for instance, found that only seven of 233 men (3%) with negative MRI results at baseline who completed 3 years of monitoring were diagnosed with clinically significant prostate cancer.
When a patient has an MRI-invisible prostate tumor, there are a couple of reasons the MRI may not be picking it up, said urologic oncologist Alexander Putnam Cole, MD, assistant professor of surgery, Harvard Medical School, Boston, Massachusetts. “One is that the cancer is aggressive but just very small,” said Cole.
“Another possibility is that the cancer looks very similar to background prostate tissue, which is something that you might expect if you think about more of a low-grade cancer,” he explained.
The experience level of the radiologist interpreting the MRI can also play into the accuracy of the reading.
But Cole agreed that “in general, MRI visibility is associated with molecular and histologic features of progression and aggressiveness and non-visible cancers are less likely to have aggressive features.”
The genomic profiles of MRI-visible and -invisible cancers bear this out.
According to Todd Morgan, MD, chief of urologic oncology at Michigan Medicine, University of Michigan, Ann Arbor, the gene expression in visible disease tends to be linked to more aggressive prostate tumors whereas gene expression in invisible disease does not.
In one analysis, for instance, researchers found that four genes — PHYHD1, CENPF, ALDH2, and GDF15 — associated with worse progression-free survival and metastasis-free survival in prostate cancer also predicted MRI visibility.
“Genes that are associated with visibility are essentially the same genes that are associated with aggressive cancers,” Klotz said.
Next Steps After Negative MRI Result
What do MRI-invisible lesions mean for patient care? If, for instance, a patient has elevated PSA levels but a normal MRI, is a targeted or systematic biopsy warranted?
The overarching message, according to Klotz, is that “you don’t need to find them.” Klotz noted, however, that patients with a negative MRI result should still be followed with periodic repeat imaging.
Several trials support this approach of using MRI to decide who needs a biopsy and delaying a biopsy in men with normal MRIs.
The recent JAMA Oncology analysis found that, among men with negative MRI results, 86% avoided a biopsy over 3 years, with clinically significant prostate cancer detected in only 4% of men across the study period — four in the initial diagnostic phase and seven in the 3-year monitoring phase. However, during the initial diagnostic phase, more than half the men with positive MRI findings had clinically significant prostate cancer detected.
Another recent study found that patients with negative MRI results were much less likely to upgrade to higher Gleason scores over time. Among 522 patients who underwent a systematic and targeted biopsy within 18 months of their grade group 1 designation, 9.2% with negative MRI findings had tumors reclassified as grade group 2 or higher vs 27% with positive MRI findings, and 2.3% with negative MRI findings had tumors reclassified as grade group 3 or higher vs 7.8% with positive MRI findings.
These data suggest that men with grade group 1 cancer and negative MRI result “may be able to avoid confirmatory biopsies until a routine surveillance biopsy in 2-3 years,” according to study author Christian Pavlovich, MD, professor of urologic oncology at the Johns Hopkins University School of Medicine, Baltimore.
Cole used MRI findings to triage who gets a biopsy. When a biopsy is warranted, “I usually recommend adding in some systematic sampling of the other side to assess for nonvisible cancers,” he noted.
Sampling prostate tissue outside the target area “adds maybe 1-2 minutes to the procedure and doesn’t drastically increase the morbidity or risks,” Cole said. It also can help “confirm there is cancer in the MRI target and also confirm there is no cancer in the nonvisible areas.”
According to Klotz, if imaging demonstrates progression, patients should receive a biopsy — in most cases, a targeted biopsy only. And, Klotz noted, skipping routine prostate biopsies in men with negative MRI results can save thousands of men from these procedures, which carry risks for infections and sepsis.
Looking beyond Gleason scores for risk prediction, MRI “visibility is a very powerful risk stratifier,” he said.
A version of this article appeared on Medscape.com.
KRAS Mutations Linked to Varied Treatment Outcomes in Metastatic Pancreatic Cancer
TOPLINE:
METHODOLOGY:
- Researchers conducted a retrospective cohort study analyzing deidentified clinical data from 2,433 patients with metastatic pancreatic ductal adenocarcinoma (PDAC), diagnosed between February 2010 and September 2022.
- They assessed the association of KRAS mutations in metastatic PDAC with the clinical outcomes and responses to first-line chemotherapy regimens.
- Data originated from approximately 280 US cancer clinics, encompassing about 800 sites of care, with comprehensive genomic profiling performed on all patients.
- Analysis focused on median overall survival (OS) and time to next treatment (TTNT) across different KRAS mutation groups, using multivariate Cox proportional hazards models.
TAKEAWAY:
- Patients with KRAS G12D and G12V mutations showed significantly higher risk for disease progression (hazard ratio [HR], 1.15; 95% CI, 1.04-1.29; P = .009) and (HR, 1.16; 95% CI, 1.04-1.30; P = .01), respectively, compared with KRAS wild type.
- KRAS G12R mutations were associated with the longest median OS at 13.2 months (95% CI, 10.6-15.2) and longest median TTNT at 6.0 months (95% CI, 5.2-6.6).
- FOLFIRINOX treatment demonstrated better outcomes than gemcitabine-based therapies across all patients, with lower risk for treatment progression (HR, 1.19; 95% CI, 1.09-1.29; P < .001) and death (HR, 1.18; 95% CI, 1.07-1.29; P < .001).
- Specifically, when FOLFIRINOX was used as first-line treatment in patients with KRAS G12D and G12V mutations, the therapy was associated with improved TTNT and OS vs gemcitabine with or without nab-paclitaxel.
IN PRACTICE:
“In its totality, these data set a benchmark for future studies on KRAS inhibitors for specific KRAS variants and highlights the groups for which treatment combinations may ultimately be necessary,” the authors concluded.
SOURCE:
The study was led by Carter Norton, Huntsman Cancer Institute in Salt Lake City, Utah. It was published online on January 7 in JAMA Network Open.
LIMITATIONS:
According to the authors, the study’s limitations include the heterogeneity of clinical data collected retrospectively, which is subject to residual confounding. The sample size was limited for certain mutational groups, particularly KRAS G12C, leading to limited statistical power. Additionally, the detection rate of genomic alterations by commercially available assays may be affected by the high stromal content and low cellularity characteristic of PDAC.
DISCLOSURES:
The study was supported by Cancer Center Support grant P30CA042014 from the National Institutes of Health. Heloisa P. Soares, MD, PhD, one of the study authors, disclosed receiving consulting fees from Ipsen, Exelixis Inc, BMS, Novartis AG, AstraZeneca, and TerSera Therapeutics LLC and symposium speaker fees from ITM Radiopharma outside the submitted work. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Researchers conducted a retrospective cohort study analyzing deidentified clinical data from 2,433 patients with metastatic pancreatic ductal adenocarcinoma (PDAC), diagnosed between February 2010 and September 2022.
- They assessed the association of KRAS mutations in metastatic PDAC with the clinical outcomes and responses to first-line chemotherapy regimens.
- Data originated from approximately 280 US cancer clinics, encompassing about 800 sites of care, with comprehensive genomic profiling performed on all patients.
- Analysis focused on median overall survival (OS) and time to next treatment (TTNT) across different KRAS mutation groups, using multivariate Cox proportional hazards models.
TAKEAWAY:
- Patients with KRAS G12D and G12V mutations showed significantly higher risk for disease progression (hazard ratio [HR], 1.15; 95% CI, 1.04-1.29; P = .009) and (HR, 1.16; 95% CI, 1.04-1.30; P = .01), respectively, compared with KRAS wild type.
- KRAS G12R mutations were associated with the longest median OS at 13.2 months (95% CI, 10.6-15.2) and longest median TTNT at 6.0 months (95% CI, 5.2-6.6).
- FOLFIRINOX treatment demonstrated better outcomes than gemcitabine-based therapies across all patients, with lower risk for treatment progression (HR, 1.19; 95% CI, 1.09-1.29; P < .001) and death (HR, 1.18; 95% CI, 1.07-1.29; P < .001).
- Specifically, when FOLFIRINOX was used as first-line treatment in patients with KRAS G12D and G12V mutations, the therapy was associated with improved TTNT and OS vs gemcitabine with or without nab-paclitaxel.
IN PRACTICE:
“In its totality, these data set a benchmark for future studies on KRAS inhibitors for specific KRAS variants and highlights the groups for which treatment combinations may ultimately be necessary,” the authors concluded.
SOURCE:
The study was led by Carter Norton, Huntsman Cancer Institute in Salt Lake City, Utah. It was published online on January 7 in JAMA Network Open.
LIMITATIONS:
According to the authors, the study’s limitations include the heterogeneity of clinical data collected retrospectively, which is subject to residual confounding. The sample size was limited for certain mutational groups, particularly KRAS G12C, leading to limited statistical power. Additionally, the detection rate of genomic alterations by commercially available assays may be affected by the high stromal content and low cellularity characteristic of PDAC.
DISCLOSURES:
The study was supported by Cancer Center Support grant P30CA042014 from the National Institutes of Health. Heloisa P. Soares, MD, PhD, one of the study authors, disclosed receiving consulting fees from Ipsen, Exelixis Inc, BMS, Novartis AG, AstraZeneca, and TerSera Therapeutics LLC and symposium speaker fees from ITM Radiopharma outside the submitted work. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Researchers conducted a retrospective cohort study analyzing deidentified clinical data from 2,433 patients with metastatic pancreatic ductal adenocarcinoma (PDAC), diagnosed between February 2010 and September 2022.
- They assessed the association of KRAS mutations in metastatic PDAC with the clinical outcomes and responses to first-line chemotherapy regimens.
- Data originated from approximately 280 US cancer clinics, encompassing about 800 sites of care, with comprehensive genomic profiling performed on all patients.
- Analysis focused on median overall survival (OS) and time to next treatment (TTNT) across different KRAS mutation groups, using multivariate Cox proportional hazards models.
TAKEAWAY:
- Patients with KRAS G12D and G12V mutations showed significantly higher risk for disease progression (hazard ratio [HR], 1.15; 95% CI, 1.04-1.29; P = .009) and (HR, 1.16; 95% CI, 1.04-1.30; P = .01), respectively, compared with KRAS wild type.
- KRAS G12R mutations were associated with the longest median OS at 13.2 months (95% CI, 10.6-15.2) and longest median TTNT at 6.0 months (95% CI, 5.2-6.6).
- FOLFIRINOX treatment demonstrated better outcomes than gemcitabine-based therapies across all patients, with lower risk for treatment progression (HR, 1.19; 95% CI, 1.09-1.29; P < .001) and death (HR, 1.18; 95% CI, 1.07-1.29; P < .001).
- Specifically, when FOLFIRINOX was used as first-line treatment in patients with KRAS G12D and G12V mutations, the therapy was associated with improved TTNT and OS vs gemcitabine with or without nab-paclitaxel.
IN PRACTICE:
“In its totality, these data set a benchmark for future studies on KRAS inhibitors for specific KRAS variants and highlights the groups for which treatment combinations may ultimately be necessary,” the authors concluded.
SOURCE:
The study was led by Carter Norton, Huntsman Cancer Institute in Salt Lake City, Utah. It was published online on January 7 in JAMA Network Open.
LIMITATIONS:
According to the authors, the study’s limitations include the heterogeneity of clinical data collected retrospectively, which is subject to residual confounding. The sample size was limited for certain mutational groups, particularly KRAS G12C, leading to limited statistical power. Additionally, the detection rate of genomic alterations by commercially available assays may be affected by the high stromal content and low cellularity characteristic of PDAC.
DISCLOSURES:
The study was supported by Cancer Center Support grant P30CA042014 from the National Institutes of Health. Heloisa P. Soares, MD, PhD, one of the study authors, disclosed receiving consulting fees from Ipsen, Exelixis Inc, BMS, Novartis AG, AstraZeneca, and TerSera Therapeutics LLC and symposium speaker fees from ITM Radiopharma outside the submitted work. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Does Watch and Wait Increase Distant Metastasis Risk in Rectal Cancer?
TOPLINE:
The new study highlights the importance of timely surgical intervention to improve distant metastases–free survival rates.
METHODOLOGY:
- Organ preservation has become an attractive alternative to surgery for patients with rectal cancer who achieve a clinical complete response after neoadjuvant therapy, with the risk for local regrowth after initial clinical complete response being around 25%-30%.
- The new study aimed to compare the risk for distant metastases between patients with local regrowth after watch and wait and patients with near-complete pathologic response managed by total mesorectal excision.
- A total of 508 patients with local regrowth were included from the International Watch & Wait Database, and 893 patients with near-complete pathologic response were included from the Spanish Rectal Cancer Project.
- The primary endpoint was distant metastases–free survival at 3 years from the decision to watch and wait or total mesorectal excision, and the secondary endpoints included possible risk factors associated with distant metastases.
TAKEAWAY:
- Patients with local regrowth had a significantly higher rate of distant metastases (rate, 22.8% vs 10.2%; P ≤.001) than those with near-complete pathologic response managed by total mesorectal excision.
- Distant metastases–free survival at 3 years was significantly worse for patients with local regrowth (rate, 75% vs 87%; P < .001).
- Independent risk factors for distant metastases included local regrowth (vs total mesorectal excision at reassessment; P = .001), ypT3-4 status (P = .016), and ypN+ status (P = .001) at the time of surgery.
- Patients with local regrowth had worse distant metastases–free survival across all pathologic stages than those managed by total mesorectal excision.
IN PRACTICE:
“Patients with local regrowth appear to have a higher risk for subsequent distant metastases development than patients with near-complete pathologic response managed by total mesorectal excision at restaging irrespective of final pathology,” the authors wrote.
SOURCE:
This study was led by Laura M. Fernandez, MD, of the Champalimaud Foundation in Lisbon, Portugal. It was published online in Journal of Clinical Oncology.
LIMITATIONS:
This study’s limitations included the heterogeneity in defining clinical complete response and the decision to watch and wait across different institutions. The majority of patients did not receive total neoadjuvant therapy regimens, which may have affected the generalizability of the findings. The study had a considerable amount of follow-up losses, which could have introduced bias.
DISCLOSURES:
This study was supported by the European Society of Surgical Oncology, the Champalimaud Foundation, the Bas Mulder Award, the Alpe d’HuZes Foundation, the Dutch Cancer Society, the European Research Council Advanced Grant, and the National Institute of Health and Research Manchester Biomedical Research Centre. Fernandez disclosed receiving grants from Johnson & Johnson. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
The new study highlights the importance of timely surgical intervention to improve distant metastases–free survival rates.
METHODOLOGY:
- Organ preservation has become an attractive alternative to surgery for patients with rectal cancer who achieve a clinical complete response after neoadjuvant therapy, with the risk for local regrowth after initial clinical complete response being around 25%-30%.
- The new study aimed to compare the risk for distant metastases between patients with local regrowth after watch and wait and patients with near-complete pathologic response managed by total mesorectal excision.
- A total of 508 patients with local regrowth were included from the International Watch & Wait Database, and 893 patients with near-complete pathologic response were included from the Spanish Rectal Cancer Project.
- The primary endpoint was distant metastases–free survival at 3 years from the decision to watch and wait or total mesorectal excision, and the secondary endpoints included possible risk factors associated with distant metastases.
TAKEAWAY:
- Patients with local regrowth had a significantly higher rate of distant metastases (rate, 22.8% vs 10.2%; P ≤.001) than those with near-complete pathologic response managed by total mesorectal excision.
- Distant metastases–free survival at 3 years was significantly worse for patients with local regrowth (rate, 75% vs 87%; P < .001).
- Independent risk factors for distant metastases included local regrowth (vs total mesorectal excision at reassessment; P = .001), ypT3-4 status (P = .016), and ypN+ status (P = .001) at the time of surgery.
- Patients with local regrowth had worse distant metastases–free survival across all pathologic stages than those managed by total mesorectal excision.
IN PRACTICE:
“Patients with local regrowth appear to have a higher risk for subsequent distant metastases development than patients with near-complete pathologic response managed by total mesorectal excision at restaging irrespective of final pathology,” the authors wrote.
SOURCE:
This study was led by Laura M. Fernandez, MD, of the Champalimaud Foundation in Lisbon, Portugal. It was published online in Journal of Clinical Oncology.
LIMITATIONS:
This study’s limitations included the heterogeneity in defining clinical complete response and the decision to watch and wait across different institutions. The majority of patients did not receive total neoadjuvant therapy regimens, which may have affected the generalizability of the findings. The study had a considerable amount of follow-up losses, which could have introduced bias.
DISCLOSURES:
This study was supported by the European Society of Surgical Oncology, the Champalimaud Foundation, the Bas Mulder Award, the Alpe d’HuZes Foundation, the Dutch Cancer Society, the European Research Council Advanced Grant, and the National Institute of Health and Research Manchester Biomedical Research Centre. Fernandez disclosed receiving grants from Johnson & Johnson. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
The new study highlights the importance of timely surgical intervention to improve distant metastases–free survival rates.
METHODOLOGY:
- Organ preservation has become an attractive alternative to surgery for patients with rectal cancer who achieve a clinical complete response after neoadjuvant therapy, with the risk for local regrowth after initial clinical complete response being around 25%-30%.
- The new study aimed to compare the risk for distant metastases between patients with local regrowth after watch and wait and patients with near-complete pathologic response managed by total mesorectal excision.
- A total of 508 patients with local regrowth were included from the International Watch & Wait Database, and 893 patients with near-complete pathologic response were included from the Spanish Rectal Cancer Project.
- The primary endpoint was distant metastases–free survival at 3 years from the decision to watch and wait or total mesorectal excision, and the secondary endpoints included possible risk factors associated with distant metastases.
TAKEAWAY:
- Patients with local regrowth had a significantly higher rate of distant metastases (rate, 22.8% vs 10.2%; P ≤.001) than those with near-complete pathologic response managed by total mesorectal excision.
- Distant metastases–free survival at 3 years was significantly worse for patients with local regrowth (rate, 75% vs 87%; P < .001).
- Independent risk factors for distant metastases included local regrowth (vs total mesorectal excision at reassessment; P = .001), ypT3-4 status (P = .016), and ypN+ status (P = .001) at the time of surgery.
- Patients with local regrowth had worse distant metastases–free survival across all pathologic stages than those managed by total mesorectal excision.
IN PRACTICE:
“Patients with local regrowth appear to have a higher risk for subsequent distant metastases development than patients with near-complete pathologic response managed by total mesorectal excision at restaging irrespective of final pathology,” the authors wrote.
SOURCE:
This study was led by Laura M. Fernandez, MD, of the Champalimaud Foundation in Lisbon, Portugal. It was published online in Journal of Clinical Oncology.
LIMITATIONS:
This study’s limitations included the heterogeneity in defining clinical complete response and the decision to watch and wait across different institutions. The majority of patients did not receive total neoadjuvant therapy regimens, which may have affected the generalizability of the findings. The study had a considerable amount of follow-up losses, which could have introduced bias.
DISCLOSURES:
This study was supported by the European Society of Surgical Oncology, the Champalimaud Foundation, the Bas Mulder Award, the Alpe d’HuZes Foundation, the Dutch Cancer Society, the European Research Council Advanced Grant, and the National Institute of Health and Research Manchester Biomedical Research Centre. Fernandez disclosed receiving grants from Johnson & Johnson. Additional disclosures are noted in the original article.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Cellular Therapies for Solid Tumors: The Next Big Thing?
The cutting edge of treating solid tumors with cell therapies got notably sharper in 2024.
First came the US Food and Drug Administration (FDA) approval in February 2024 of the tumor-infiltrating lymphocyte (TIL) therapy lifileucel in unresectable or metastatic melanoma that had progressed on prior immunotherapy, the first cellular therapy for any solid tumor. Then came the August FDA approval of afamitresgene autoleucel in unresectable or metastatic synovial sarcoma with failed chemotherapy, the first engineered T-cell therapy for cancers in soft tissue.
“This was a pipe dream just a decade ago,” Alison Betof Warner, MD, PhD, lead author of a lifileucel study (NCT05640193), said in an interview with Medscape Medical News. “At the start of 2024, we had no approvals of these kinds of products in solid cancers. Now we have two.”
As the director of Solid Tumor Cell Therapy and leader of Stanford Medicine’s Melanoma and Cutaneous Oncology Clinical Research Group, Betof Warner has been at the forefront of developing commercial cell therapy using tumor-infiltrating lymphocytes (TILs).
“The approval of lifileucel increases confidence that we can get these therapies across the regulatory finish line and to patients,” Betof Warner said during the interview. She was not involved in the development of afamitresgene autoleucel.
‘Reverse Engineering’
In addition to her contributions to the work that led to lifileucel’s approval, Betof Warner was the lead author on the first consensus guidelines on management and best practices for tumor-infiltrating lymphocyte cell therapy.
Betof Warner began studying TILs after doing research with her mentors in immuno-oncology, Jedd D. Wolchok and Michael A. Postow. Their investigations — including one that Betof Warner coauthored — into how monoclonal antibodies and checkpoint inhibitors, such as ipilimumab or nivolumab, might extend the lives of people with advanced unresectable or metastatic melanoma inspired her to push further to find ways to minimize treatment while maximizing outcomes for patients. Betof Warner’s interest overall, she said in the interview, is in capitalizing on what can be learned about how the immune system controls cancer.
“What we know is that the immune system has the ability to kill cancer,” Betof Warner said. “Therefore we need to be thinking about how we can increase immune surveillance. How can we enhance that before a patient develops advanced cancer?
Betof Warner said that although TILs are now standard treatment in melanoma, there is about a 30% response rate compared with about a 50% response rate in immunotherapy, and the latter is easier for the patient to withstand.
“Antibodies on the frontline are better than going through a surgery and then waiting weeks to get your therapy,” Betof Warner said in the interview. “You can come into my clinic and get an antibody therapy in 30 minutes and go straight to work. TILs require patients to be in the hospital for weeks at a time and out of work for months at a time.”
In an effort to combine therapies to maximize best outcomes, a phase 3 trial (NCT05727904) is currently recruiting. The TILVANCE-301 trial will compare immunotherapy plus adoptive cell therapy vs immunotherapy alone in untreated unresectable or metastatic melanoma. Betof Warner is not a part of this study.
Cell Therapies Include CAR T Cells and TCRT
In general, adoptive T-cell therapies such as TILs involve the isolation of autologous immune cells that are removed from the body and either expanded or modified to optimize their efficacy in fighting antigens, before their transfer to the patient as a living drug by infusion.
In addition to TILs, adoptive cell therapies for antitumor therapeutics include chimeric antigen receptor (CAR) T cells and engineered T-cell receptor therapy (TCRT).
In CAR T-cell therapy and TCRT, naive T cells are harvested from the patient’s blood then engineered to target a tumor. In TIL therapy, tumor-specific T cells are taken from the patient’s tumor. Once extracted, the respective cells are expanded billions of times and then delivered back to the patient’s body, said Betof Warner.
“The main promise of this approach is to generate responses in what we know as ‘cold’ tumors, or tumors that do not have a lot of endogenous T-cell infiltration or where the T cells are not working well, to bring in tumor targeting T cells and then trigger an immune response,” Betof Warner told an audience at the American Society of Clinical Oncology (ASCO) 2024 annual meeting.
TIL patients also receive interleukin (IL)-2 infusions to further stimulate the cells. In patients being treated with TCRT, they either receive low or no IL-2, Betof Warner said in her ASCO presentation, “Adopting Cutting-Edge Cell Therapies in Melanoma,” part of the session Beyond the Tip of the Iceberg: Next-Generation Cell-Based Therapies.
Decades in the Making
The National Cancer Institute began investigating TILs in the late 1980s, with the current National Cancer Institute (NCI) surgery chief, Steven Rosenberg, MD, PhD, leading the first-ever trials that showed TILs could shrink tumors in people with advanced melanoma.
Since then, NCI staff and others have also investigated TILs beyond melanoma and additional cell therapies based on CAR T cells and TCRT for antitumor therapeutics.
“TCRs are different from CAR Ts because they go after intracellular antigens instead of extracellular antigens,” said Betof Warner. “That has appeal because many of the tumor antigens we’re looking for will be intracellular.”
Because CAR T cells only target extracellular antigens, their utility is somewhat limited. Although several CAR T-cell therapies exist for blood cancers, there currently are no approved CAR T-cell therapies for solid tumors. However, several trials of CAR T cells in gastrointestinal cancers and melanoma are ongoing, said Betof Warner, who is not a part of these studies.
“We are starting to see early-phase efficacy in pediatric gliomas,” Betof Warner said, mentioning a study conducted by colleagues at Stanford who demonstrated potential for anti-GD2 CAR T-cell therapy in deadly pediatric diffuse midline gliomas, tumors on the spine and brain.
In their study, nine out of 11 participants (median age, 15 years) showed benefit from the cell therapy, with one participant’s tumors resolving completely. The results paved the way for the FDA to grant a Regenerative Medicine Advanced Therapy designation for use of anti-GD2 CAR T cells in H3K27M-positive diffuse midline gliomas.
The investigators are now recruiting for a phase 1 trial (NCT04196413). Results of the initial study were published in Nature last month.
Another lesser-known cell therapy expected to advance at some point in the future for solid tumors is use of the body’s natural killer (NK) cells. “They’ve been known about for a long time, but they are more difficult to regulate, which is one reason why it has taken longer to make NK cell therapies,” said Betof Warner, who is not involved in the study of NK cells. “One of their advantages is that, potentially, there could be an ‘off the shelf’ NK product. They don’t necessarily have to be made with autologous cells.”
Risk-Benefit Profiles Depend on Mechanism of Action
If the corresponding TCR sequence of a tumor antigen is known, said Betof Warner, it is possible to use leukapheresis to generate naive circulating lymphocytes. Once infused, the manufactured TCRTs will activate in the body the same as native cells because the signaling is the same.
An advantage to TCRT compared with CAR T-cell therapy is that it targets intracellular proteins, which are significantly present in the tumor, Betof Warner said in her presentation at ASCO 2024. She clarified that tumors will usually be screened for the presence of this antigen before a patient is selected for treatment with that particular therapy, because not all antigens are highly expressed in every tumor.
“Furthermore, the tumor antigen has to be presented by a major histocompatibility complex, meaning there are human leukocyte antigen restrictions, which impacts patient selection,” she said.
A risk with both TCRT and CAR T-cell therapy, according to Betof Warner, is that because there are often shared antigens between tumor and normal tissues, on-target/off-tumor toxicity is a risk.
“TILs are different because they are nonengineered, at least not for antigen recognition. They are polyclonal and go after multiple targets,” Betof Warner said. “TCRs and CARs are engineered to go after one target. So, TILs have much lower rates of on-tumor/off-target effects, vs when you engineer a very high affinity receptor like a TCR or CAR.”
A good example of how this amplification of TCR affinity can lead to poor outcomes is in metastatic melanoma, said Betof Warner.
In investigations (NCI-07-C-0174 and NCI-07-C-0175) of TCRT in metastatic melanoma, for example, the researchers were targeting MART-1 or gp100, which are expressed in melanocytes.
“The problem was that these antigens are also expressed in the eyes and ears, so it caused eye inflammation and hearing loss in a number of patients because it wasn’t specific enough for the tumor,” said Betof Warner. “So, if that target is highly expressed on normal tissue, then you have a high risk.”
Promise of PRAME
Betof Warner said the most promising TCRT at present is the investigational autologous cell therapy IMA203 (NCT03688124), which targets the preferentially expressed antigen (PRAME). Although PRAME is found in many tumors, this testis antigen does not tend to express in normal, healthy adult tissues. Betof Warner is not affiliated with this study.
“It’s maybe the most exciting TCRT cell in melanoma,” Betof Warner told her audience at the ASCO 2024 meeting. Because the expression rate of PRAME in cutaneous and uveal melanoma is at or above 95% and 90%, respectively, she said “it is a really good target in melanoma.”
Phase 1a results reported in late 2023 from a first-in-human trial of IMA203 involving 13 persons with highly advanced melanoma and a median of 5.5 previous treatments showed a 50% objective response rate in the 12 evaluable results. The duration of response ranged between 2.2 and 14.7 months (median follow-up, 14 months).
The safety profile of the treatment was favorable, with no grade 3 adverse events occurring in more than 10% of the cohort, and no grade 5 adverse events at all.
Phase 1b results published in October by maker Immatics showed that in 28 heavily pretreated metastatic melanoma patients, IMA203 had a confirmed objective response rate of 54% with a median duration of response of 12.1 months, while maintaining a favorable tolerability profile.
Accelerated Approvals, Boxed Warnings
The FDA granted accelerated approvals for both lifileucel, the TIL therapy, and afamitresgene autoleucel, the TCRT.
Both were approved with boxed warnings. Lifileucel’s warning is for treatment-related mortality, prolonged severe cytopenia, severe infection, and cardiopulmonary and renal impairment. Afamitresgene autoleucel’s boxed warning is for serious or fatal cytokine release syndrome, which may be severe or life-threatening.
With these approvals, the bar is now raised on TILs and TCRTs, said Betof Warner.
The lifileucel trial studied 73 patients whose melanoma had continued to metastasize despite treatment with a programmed cell death protein (PD-1)/ programmed death-ligand (PD-L1)–targeted immune checkpoint inhibitor and a BRAF inhibitor (if appropriate based on tumor mutation status), and whose lifileucel dose was at least 7.5 billion cells (the approved dose). The cohort also received a median of six IL-2 (aldesleukin) doses.
The objective response rate was 31.5% (95% CI, 21.1-43.4), and median duration of response was not reached (lower bound of 95% CI, 4.1).
In the afamitresgene autoleucel study, 44 of 52 patients with synovial sarcoma received leukapheresis and a single infusion of afamitresgene autoleucel.
The overall response rate was 43.2% (95% CI, 28.4-59.0). The median time to response was 4.9 weeks (95% CI, 4.4-8), and the median duration of response was 6 months (lower bound of 95% CI, 4.6). Among patients who were responsive to the treatment, 45.6% and 39.0% had a duration of response of 6 months or longer and 12 months or longer, respectively.
New Hope for Patients
Betof Warner and her colleagues are now recruiting for an open-label, phase 1/2 investigation of the safety and efficacy of the TIL therapy OBX-115 in adult advanced solid tumors in melanoma or non–small cell lung cancer. The first-in-human results of a previous trial were presented at the ASCO 2024 meeting, and OBX-115 received FDA fast track designation in July.
“I think the results are really promising,” said Betof Warner. “This is an engineered TIL that does not require administering IL-2 to the patient. There were four out of the nine patients who responded to the treatment and there were no dose-limiting toxicities, no cytokine and no intracranial — all of which is excellent.”
For Betof Warner, the possibility that by using their own immune system, patients with advanced and refractory cancers could soon have a one-time treatment with a cell therapy rather than innumerable bouts of chemotherapy pushes her onward.
“The idea that we can treat cancer one time and have it not recur for years — that’s pushing the start of saying there’s a cure of cancer. That a person could move on from cancer like they move on from an infection. That is the potential of this work. We’re not there yet, but that’s where we need to think and dream big,” she said.
Betof Warner disclosed consulting/advisory roles with BluePath Solutions, Bristol-Myers Squibb/Medarex, Immatics, Instil Bio, Iovance Biotherapeutics, Lyell Immunopharma, Merck, Novartis, and Pfizer and research funding and travel expenses from Iovance Biotherapeutics.
A version of this article appeared on Medscape.com.
The cutting edge of treating solid tumors with cell therapies got notably sharper in 2024.
First came the US Food and Drug Administration (FDA) approval in February 2024 of the tumor-infiltrating lymphocyte (TIL) therapy lifileucel in unresectable or metastatic melanoma that had progressed on prior immunotherapy, the first cellular therapy for any solid tumor. Then came the August FDA approval of afamitresgene autoleucel in unresectable or metastatic synovial sarcoma with failed chemotherapy, the first engineered T-cell therapy for cancers in soft tissue.
“This was a pipe dream just a decade ago,” Alison Betof Warner, MD, PhD, lead author of a lifileucel study (NCT05640193), said in an interview with Medscape Medical News. “At the start of 2024, we had no approvals of these kinds of products in solid cancers. Now we have two.”
As the director of Solid Tumor Cell Therapy and leader of Stanford Medicine’s Melanoma and Cutaneous Oncology Clinical Research Group, Betof Warner has been at the forefront of developing commercial cell therapy using tumor-infiltrating lymphocytes (TILs).
“The approval of lifileucel increases confidence that we can get these therapies across the regulatory finish line and to patients,” Betof Warner said during the interview. She was not involved in the development of afamitresgene autoleucel.
‘Reverse Engineering’
In addition to her contributions to the work that led to lifileucel’s approval, Betof Warner was the lead author on the first consensus guidelines on management and best practices for tumor-infiltrating lymphocyte cell therapy.
Betof Warner began studying TILs after doing research with her mentors in immuno-oncology, Jedd D. Wolchok and Michael A. Postow. Their investigations — including one that Betof Warner coauthored — into how monoclonal antibodies and checkpoint inhibitors, such as ipilimumab or nivolumab, might extend the lives of people with advanced unresectable or metastatic melanoma inspired her to push further to find ways to minimize treatment while maximizing outcomes for patients. Betof Warner’s interest overall, she said in the interview, is in capitalizing on what can be learned about how the immune system controls cancer.
“What we know is that the immune system has the ability to kill cancer,” Betof Warner said. “Therefore we need to be thinking about how we can increase immune surveillance. How can we enhance that before a patient develops advanced cancer?
Betof Warner said that although TILs are now standard treatment in melanoma, there is about a 30% response rate compared with about a 50% response rate in immunotherapy, and the latter is easier for the patient to withstand.
“Antibodies on the frontline are better than going through a surgery and then waiting weeks to get your therapy,” Betof Warner said in the interview. “You can come into my clinic and get an antibody therapy in 30 minutes and go straight to work. TILs require patients to be in the hospital for weeks at a time and out of work for months at a time.”
In an effort to combine therapies to maximize best outcomes, a phase 3 trial (NCT05727904) is currently recruiting. The TILVANCE-301 trial will compare immunotherapy plus adoptive cell therapy vs immunotherapy alone in untreated unresectable or metastatic melanoma. Betof Warner is not a part of this study.
Cell Therapies Include CAR T Cells and TCRT
In general, adoptive T-cell therapies such as TILs involve the isolation of autologous immune cells that are removed from the body and either expanded or modified to optimize their efficacy in fighting antigens, before their transfer to the patient as a living drug by infusion.
In addition to TILs, adoptive cell therapies for antitumor therapeutics include chimeric antigen receptor (CAR) T cells and engineered T-cell receptor therapy (TCRT).
In CAR T-cell therapy and TCRT, naive T cells are harvested from the patient’s blood then engineered to target a tumor. In TIL therapy, tumor-specific T cells are taken from the patient’s tumor. Once extracted, the respective cells are expanded billions of times and then delivered back to the patient’s body, said Betof Warner.
“The main promise of this approach is to generate responses in what we know as ‘cold’ tumors, or tumors that do not have a lot of endogenous T-cell infiltration or where the T cells are not working well, to bring in tumor targeting T cells and then trigger an immune response,” Betof Warner told an audience at the American Society of Clinical Oncology (ASCO) 2024 annual meeting.
TIL patients also receive interleukin (IL)-2 infusions to further stimulate the cells. In patients being treated with TCRT, they either receive low or no IL-2, Betof Warner said in her ASCO presentation, “Adopting Cutting-Edge Cell Therapies in Melanoma,” part of the session Beyond the Tip of the Iceberg: Next-Generation Cell-Based Therapies.
Decades in the Making
The National Cancer Institute began investigating TILs in the late 1980s, with the current National Cancer Institute (NCI) surgery chief, Steven Rosenberg, MD, PhD, leading the first-ever trials that showed TILs could shrink tumors in people with advanced melanoma.
Since then, NCI staff and others have also investigated TILs beyond melanoma and additional cell therapies based on CAR T cells and TCRT for antitumor therapeutics.
“TCRs are different from CAR Ts because they go after intracellular antigens instead of extracellular antigens,” said Betof Warner. “That has appeal because many of the tumor antigens we’re looking for will be intracellular.”
Because CAR T cells only target extracellular antigens, their utility is somewhat limited. Although several CAR T-cell therapies exist for blood cancers, there currently are no approved CAR T-cell therapies for solid tumors. However, several trials of CAR T cells in gastrointestinal cancers and melanoma are ongoing, said Betof Warner, who is not a part of these studies.
“We are starting to see early-phase efficacy in pediatric gliomas,” Betof Warner said, mentioning a study conducted by colleagues at Stanford who demonstrated potential for anti-GD2 CAR T-cell therapy in deadly pediatric diffuse midline gliomas, tumors on the spine and brain.
In their study, nine out of 11 participants (median age, 15 years) showed benefit from the cell therapy, with one participant’s tumors resolving completely. The results paved the way for the FDA to grant a Regenerative Medicine Advanced Therapy designation for use of anti-GD2 CAR T cells in H3K27M-positive diffuse midline gliomas.
The investigators are now recruiting for a phase 1 trial (NCT04196413). Results of the initial study were published in Nature last month.
Another lesser-known cell therapy expected to advance at some point in the future for solid tumors is use of the body’s natural killer (NK) cells. “They’ve been known about for a long time, but they are more difficult to regulate, which is one reason why it has taken longer to make NK cell therapies,” said Betof Warner, who is not involved in the study of NK cells. “One of their advantages is that, potentially, there could be an ‘off the shelf’ NK product. They don’t necessarily have to be made with autologous cells.”
Risk-Benefit Profiles Depend on Mechanism of Action
If the corresponding TCR sequence of a tumor antigen is known, said Betof Warner, it is possible to use leukapheresis to generate naive circulating lymphocytes. Once infused, the manufactured TCRTs will activate in the body the same as native cells because the signaling is the same.
An advantage to TCRT compared with CAR T-cell therapy is that it targets intracellular proteins, which are significantly present in the tumor, Betof Warner said in her presentation at ASCO 2024. She clarified that tumors will usually be screened for the presence of this antigen before a patient is selected for treatment with that particular therapy, because not all antigens are highly expressed in every tumor.
“Furthermore, the tumor antigen has to be presented by a major histocompatibility complex, meaning there are human leukocyte antigen restrictions, which impacts patient selection,” she said.
A risk with both TCRT and CAR T-cell therapy, according to Betof Warner, is that because there are often shared antigens between tumor and normal tissues, on-target/off-tumor toxicity is a risk.
“TILs are different because they are nonengineered, at least not for antigen recognition. They are polyclonal and go after multiple targets,” Betof Warner said. “TCRs and CARs are engineered to go after one target. So, TILs have much lower rates of on-tumor/off-target effects, vs when you engineer a very high affinity receptor like a TCR or CAR.”
A good example of how this amplification of TCR affinity can lead to poor outcomes is in metastatic melanoma, said Betof Warner.
In investigations (NCI-07-C-0174 and NCI-07-C-0175) of TCRT in metastatic melanoma, for example, the researchers were targeting MART-1 or gp100, which are expressed in melanocytes.
“The problem was that these antigens are also expressed in the eyes and ears, so it caused eye inflammation and hearing loss in a number of patients because it wasn’t specific enough for the tumor,” said Betof Warner. “So, if that target is highly expressed on normal tissue, then you have a high risk.”
Promise of PRAME
Betof Warner said the most promising TCRT at present is the investigational autologous cell therapy IMA203 (NCT03688124), which targets the preferentially expressed antigen (PRAME). Although PRAME is found in many tumors, this testis antigen does not tend to express in normal, healthy adult tissues. Betof Warner is not affiliated with this study.
“It’s maybe the most exciting TCRT cell in melanoma,” Betof Warner told her audience at the ASCO 2024 meeting. Because the expression rate of PRAME in cutaneous and uveal melanoma is at or above 95% and 90%, respectively, she said “it is a really good target in melanoma.”
Phase 1a results reported in late 2023 from a first-in-human trial of IMA203 involving 13 persons with highly advanced melanoma and a median of 5.5 previous treatments showed a 50% objective response rate in the 12 evaluable results. The duration of response ranged between 2.2 and 14.7 months (median follow-up, 14 months).
The safety profile of the treatment was favorable, with no grade 3 adverse events occurring in more than 10% of the cohort, and no grade 5 adverse events at all.
Phase 1b results published in October by maker Immatics showed that in 28 heavily pretreated metastatic melanoma patients, IMA203 had a confirmed objective response rate of 54% with a median duration of response of 12.1 months, while maintaining a favorable tolerability profile.
Accelerated Approvals, Boxed Warnings
The FDA granted accelerated approvals for both lifileucel, the TIL therapy, and afamitresgene autoleucel, the TCRT.
Both were approved with boxed warnings. Lifileucel’s warning is for treatment-related mortality, prolonged severe cytopenia, severe infection, and cardiopulmonary and renal impairment. Afamitresgene autoleucel’s boxed warning is for serious or fatal cytokine release syndrome, which may be severe or life-threatening.
With these approvals, the bar is now raised on TILs and TCRTs, said Betof Warner.
The lifileucel trial studied 73 patients whose melanoma had continued to metastasize despite treatment with a programmed cell death protein (PD-1)/ programmed death-ligand (PD-L1)–targeted immune checkpoint inhibitor and a BRAF inhibitor (if appropriate based on tumor mutation status), and whose lifileucel dose was at least 7.5 billion cells (the approved dose). The cohort also received a median of six IL-2 (aldesleukin) doses.
The objective response rate was 31.5% (95% CI, 21.1-43.4), and median duration of response was not reached (lower bound of 95% CI, 4.1).
In the afamitresgene autoleucel study, 44 of 52 patients with synovial sarcoma received leukapheresis and a single infusion of afamitresgene autoleucel.
The overall response rate was 43.2% (95% CI, 28.4-59.0). The median time to response was 4.9 weeks (95% CI, 4.4-8), and the median duration of response was 6 months (lower bound of 95% CI, 4.6). Among patients who were responsive to the treatment, 45.6% and 39.0% had a duration of response of 6 months or longer and 12 months or longer, respectively.
New Hope for Patients
Betof Warner and her colleagues are now recruiting for an open-label, phase 1/2 investigation of the safety and efficacy of the TIL therapy OBX-115 in adult advanced solid tumors in melanoma or non–small cell lung cancer. The first-in-human results of a previous trial were presented at the ASCO 2024 meeting, and OBX-115 received FDA fast track designation in July.
“I think the results are really promising,” said Betof Warner. “This is an engineered TIL that does not require administering IL-2 to the patient. There were four out of the nine patients who responded to the treatment and there were no dose-limiting toxicities, no cytokine and no intracranial — all of which is excellent.”
For Betof Warner, the possibility that by using their own immune system, patients with advanced and refractory cancers could soon have a one-time treatment with a cell therapy rather than innumerable bouts of chemotherapy pushes her onward.
“The idea that we can treat cancer one time and have it not recur for years — that’s pushing the start of saying there’s a cure of cancer. That a person could move on from cancer like they move on from an infection. That is the potential of this work. We’re not there yet, but that’s where we need to think and dream big,” she said.
Betof Warner disclosed consulting/advisory roles with BluePath Solutions, Bristol-Myers Squibb/Medarex, Immatics, Instil Bio, Iovance Biotherapeutics, Lyell Immunopharma, Merck, Novartis, and Pfizer and research funding and travel expenses from Iovance Biotherapeutics.
A version of this article appeared on Medscape.com.
The cutting edge of treating solid tumors with cell therapies got notably sharper in 2024.
First came the US Food and Drug Administration (FDA) approval in February 2024 of the tumor-infiltrating lymphocyte (TIL) therapy lifileucel in unresectable or metastatic melanoma that had progressed on prior immunotherapy, the first cellular therapy for any solid tumor. Then came the August FDA approval of afamitresgene autoleucel in unresectable or metastatic synovial sarcoma with failed chemotherapy, the first engineered T-cell therapy for cancers in soft tissue.
“This was a pipe dream just a decade ago,” Alison Betof Warner, MD, PhD, lead author of a lifileucel study (NCT05640193), said in an interview with Medscape Medical News. “At the start of 2024, we had no approvals of these kinds of products in solid cancers. Now we have two.”
As the director of Solid Tumor Cell Therapy and leader of Stanford Medicine’s Melanoma and Cutaneous Oncology Clinical Research Group, Betof Warner has been at the forefront of developing commercial cell therapy using tumor-infiltrating lymphocytes (TILs).
“The approval of lifileucel increases confidence that we can get these therapies across the regulatory finish line and to patients,” Betof Warner said during the interview. She was not involved in the development of afamitresgene autoleucel.
‘Reverse Engineering’
In addition to her contributions to the work that led to lifileucel’s approval, Betof Warner was the lead author on the first consensus guidelines on management and best practices for tumor-infiltrating lymphocyte cell therapy.
Betof Warner began studying TILs after doing research with her mentors in immuno-oncology, Jedd D. Wolchok and Michael A. Postow. Their investigations — including one that Betof Warner coauthored — into how monoclonal antibodies and checkpoint inhibitors, such as ipilimumab or nivolumab, might extend the lives of people with advanced unresectable or metastatic melanoma inspired her to push further to find ways to minimize treatment while maximizing outcomes for patients. Betof Warner’s interest overall, she said in the interview, is in capitalizing on what can be learned about how the immune system controls cancer.
“What we know is that the immune system has the ability to kill cancer,” Betof Warner said. “Therefore we need to be thinking about how we can increase immune surveillance. How can we enhance that before a patient develops advanced cancer?
Betof Warner said that although TILs are now standard treatment in melanoma, there is about a 30% response rate compared with about a 50% response rate in immunotherapy, and the latter is easier for the patient to withstand.
“Antibodies on the frontline are better than going through a surgery and then waiting weeks to get your therapy,” Betof Warner said in the interview. “You can come into my clinic and get an antibody therapy in 30 minutes and go straight to work. TILs require patients to be in the hospital for weeks at a time and out of work for months at a time.”
In an effort to combine therapies to maximize best outcomes, a phase 3 trial (NCT05727904) is currently recruiting. The TILVANCE-301 trial will compare immunotherapy plus adoptive cell therapy vs immunotherapy alone in untreated unresectable or metastatic melanoma. Betof Warner is not a part of this study.
Cell Therapies Include CAR T Cells and TCRT
In general, adoptive T-cell therapies such as TILs involve the isolation of autologous immune cells that are removed from the body and either expanded or modified to optimize their efficacy in fighting antigens, before their transfer to the patient as a living drug by infusion.
In addition to TILs, adoptive cell therapies for antitumor therapeutics include chimeric antigen receptor (CAR) T cells and engineered T-cell receptor therapy (TCRT).
In CAR T-cell therapy and TCRT, naive T cells are harvested from the patient’s blood then engineered to target a tumor. In TIL therapy, tumor-specific T cells are taken from the patient’s tumor. Once extracted, the respective cells are expanded billions of times and then delivered back to the patient’s body, said Betof Warner.
“The main promise of this approach is to generate responses in what we know as ‘cold’ tumors, or tumors that do not have a lot of endogenous T-cell infiltration or where the T cells are not working well, to bring in tumor targeting T cells and then trigger an immune response,” Betof Warner told an audience at the American Society of Clinical Oncology (ASCO) 2024 annual meeting.
TIL patients also receive interleukin (IL)-2 infusions to further stimulate the cells. In patients being treated with TCRT, they either receive low or no IL-2, Betof Warner said in her ASCO presentation, “Adopting Cutting-Edge Cell Therapies in Melanoma,” part of the session Beyond the Tip of the Iceberg: Next-Generation Cell-Based Therapies.
Decades in the Making
The National Cancer Institute began investigating TILs in the late 1980s, with the current National Cancer Institute (NCI) surgery chief, Steven Rosenberg, MD, PhD, leading the first-ever trials that showed TILs could shrink tumors in people with advanced melanoma.
Since then, NCI staff and others have also investigated TILs beyond melanoma and additional cell therapies based on CAR T cells and TCRT for antitumor therapeutics.
“TCRs are different from CAR Ts because they go after intracellular antigens instead of extracellular antigens,” said Betof Warner. “That has appeal because many of the tumor antigens we’re looking for will be intracellular.”
Because CAR T cells only target extracellular antigens, their utility is somewhat limited. Although several CAR T-cell therapies exist for blood cancers, there currently are no approved CAR T-cell therapies for solid tumors. However, several trials of CAR T cells in gastrointestinal cancers and melanoma are ongoing, said Betof Warner, who is not a part of these studies.
“We are starting to see early-phase efficacy in pediatric gliomas,” Betof Warner said, mentioning a study conducted by colleagues at Stanford who demonstrated potential for anti-GD2 CAR T-cell therapy in deadly pediatric diffuse midline gliomas, tumors on the spine and brain.
In their study, nine out of 11 participants (median age, 15 years) showed benefit from the cell therapy, with one participant’s tumors resolving completely. The results paved the way for the FDA to grant a Regenerative Medicine Advanced Therapy designation for use of anti-GD2 CAR T cells in H3K27M-positive diffuse midline gliomas.
The investigators are now recruiting for a phase 1 trial (NCT04196413). Results of the initial study were published in Nature last month.
Another lesser-known cell therapy expected to advance at some point in the future for solid tumors is use of the body’s natural killer (NK) cells. “They’ve been known about for a long time, but they are more difficult to regulate, which is one reason why it has taken longer to make NK cell therapies,” said Betof Warner, who is not involved in the study of NK cells. “One of their advantages is that, potentially, there could be an ‘off the shelf’ NK product. They don’t necessarily have to be made with autologous cells.”
Risk-Benefit Profiles Depend on Mechanism of Action
If the corresponding TCR sequence of a tumor antigen is known, said Betof Warner, it is possible to use leukapheresis to generate naive circulating lymphocytes. Once infused, the manufactured TCRTs will activate in the body the same as native cells because the signaling is the same.
An advantage to TCRT compared with CAR T-cell therapy is that it targets intracellular proteins, which are significantly present in the tumor, Betof Warner said in her presentation at ASCO 2024. She clarified that tumors will usually be screened for the presence of this antigen before a patient is selected for treatment with that particular therapy, because not all antigens are highly expressed in every tumor.
“Furthermore, the tumor antigen has to be presented by a major histocompatibility complex, meaning there are human leukocyte antigen restrictions, which impacts patient selection,” she said.
A risk with both TCRT and CAR T-cell therapy, according to Betof Warner, is that because there are often shared antigens between tumor and normal tissues, on-target/off-tumor toxicity is a risk.
“TILs are different because they are nonengineered, at least not for antigen recognition. They are polyclonal and go after multiple targets,” Betof Warner said. “TCRs and CARs are engineered to go after one target. So, TILs have much lower rates of on-tumor/off-target effects, vs when you engineer a very high affinity receptor like a TCR or CAR.”
A good example of how this amplification of TCR affinity can lead to poor outcomes is in metastatic melanoma, said Betof Warner.
In investigations (NCI-07-C-0174 and NCI-07-C-0175) of TCRT in metastatic melanoma, for example, the researchers were targeting MART-1 or gp100, which are expressed in melanocytes.
“The problem was that these antigens are also expressed in the eyes and ears, so it caused eye inflammation and hearing loss in a number of patients because it wasn’t specific enough for the tumor,” said Betof Warner. “So, if that target is highly expressed on normal tissue, then you have a high risk.”
Promise of PRAME
Betof Warner said the most promising TCRT at present is the investigational autologous cell therapy IMA203 (NCT03688124), which targets the preferentially expressed antigen (PRAME). Although PRAME is found in many tumors, this testis antigen does not tend to express in normal, healthy adult tissues. Betof Warner is not affiliated with this study.
“It’s maybe the most exciting TCRT cell in melanoma,” Betof Warner told her audience at the ASCO 2024 meeting. Because the expression rate of PRAME in cutaneous and uveal melanoma is at or above 95% and 90%, respectively, she said “it is a really good target in melanoma.”
Phase 1a results reported in late 2023 from a first-in-human trial of IMA203 involving 13 persons with highly advanced melanoma and a median of 5.5 previous treatments showed a 50% objective response rate in the 12 evaluable results. The duration of response ranged between 2.2 and 14.7 months (median follow-up, 14 months).
The safety profile of the treatment was favorable, with no grade 3 adverse events occurring in more than 10% of the cohort, and no grade 5 adverse events at all.
Phase 1b results published in October by maker Immatics showed that in 28 heavily pretreated metastatic melanoma patients, IMA203 had a confirmed objective response rate of 54% with a median duration of response of 12.1 months, while maintaining a favorable tolerability profile.
Accelerated Approvals, Boxed Warnings
The FDA granted accelerated approvals for both lifileucel, the TIL therapy, and afamitresgene autoleucel, the TCRT.
Both were approved with boxed warnings. Lifileucel’s warning is for treatment-related mortality, prolonged severe cytopenia, severe infection, and cardiopulmonary and renal impairment. Afamitresgene autoleucel’s boxed warning is for serious or fatal cytokine release syndrome, which may be severe or life-threatening.
With these approvals, the bar is now raised on TILs and TCRTs, said Betof Warner.
The lifileucel trial studied 73 patients whose melanoma had continued to metastasize despite treatment with a programmed cell death protein (PD-1)/ programmed death-ligand (PD-L1)–targeted immune checkpoint inhibitor and a BRAF inhibitor (if appropriate based on tumor mutation status), and whose lifileucel dose was at least 7.5 billion cells (the approved dose). The cohort also received a median of six IL-2 (aldesleukin) doses.
The objective response rate was 31.5% (95% CI, 21.1-43.4), and median duration of response was not reached (lower bound of 95% CI, 4.1).
In the afamitresgene autoleucel study, 44 of 52 patients with synovial sarcoma received leukapheresis and a single infusion of afamitresgene autoleucel.
The overall response rate was 43.2% (95% CI, 28.4-59.0). The median time to response was 4.9 weeks (95% CI, 4.4-8), and the median duration of response was 6 months (lower bound of 95% CI, 4.6). Among patients who were responsive to the treatment, 45.6% and 39.0% had a duration of response of 6 months or longer and 12 months or longer, respectively.
New Hope for Patients
Betof Warner and her colleagues are now recruiting for an open-label, phase 1/2 investigation of the safety and efficacy of the TIL therapy OBX-115 in adult advanced solid tumors in melanoma or non–small cell lung cancer. The first-in-human results of a previous trial were presented at the ASCO 2024 meeting, and OBX-115 received FDA fast track designation in July.
“I think the results are really promising,” said Betof Warner. “This is an engineered TIL that does not require administering IL-2 to the patient. There were four out of the nine patients who responded to the treatment and there were no dose-limiting toxicities, no cytokine and no intracranial — all of which is excellent.”
For Betof Warner, the possibility that by using their own immune system, patients with advanced and refractory cancers could soon have a one-time treatment with a cell therapy rather than innumerable bouts of chemotherapy pushes her onward.
“The idea that we can treat cancer one time and have it not recur for years — that’s pushing the start of saying there’s a cure of cancer. That a person could move on from cancer like they move on from an infection. That is the potential of this work. We’re not there yet, but that’s where we need to think and dream big,” she said.
Betof Warner disclosed consulting/advisory roles with BluePath Solutions, Bristol-Myers Squibb/Medarex, Immatics, Instil Bio, Iovance Biotherapeutics, Lyell Immunopharma, Merck, Novartis, and Pfizer and research funding and travel expenses from Iovance Biotherapeutics.
A version of this article appeared on Medscape.com.
AI-Aided Colonoscopy’s ‘Intelligent’ Module Ups Polyp Detection
Colin J. Rees, a professor of gastroenterology in the Faculty of Medical Sciences at Newcastle University in Newcastle upon Tyne, England, and colleagues compared the real-world clinical effectiveness of computer-aided detection (CADe)–assisted colonoscopy using an “intelligent” module with that of standard colonoscopy in a study in The Lancet Gastroenterology & Hepatology.
They found the GI Genius Intelligent Endoscopy Module (Medtronic) increased the mean number of adenomas detected per procedure and the adenoma detection rate, especially for small, flat (type 0-IIa) polyps, and sessile serrated lesions, which are more likely to be missed.
“Missed sessile serrated lesions disproportionately increase the risk of post-colonoscopy colorectal cancer, thus the adoption of GI Genius into routine colonoscopy practice could not only increase polyp detection but also reduce the incidence of post-colonoscopy colorectal cancer,” the investigators wrote.
“AI is going to have a major impact upon most aspects of healthcare. Some areas of medical practice are now well established, and some are still in evolution,” Rees, who is also president of the British Society of Gastroenterology, said in an interview. “Within gastroenterology, the role of AI in endoscopic diagnostics is also evolving. The COLO-DETECT trial demonstrates that AI increases detection of lesions, and work is ongoing to see how AI might help with characterization and other elements of endoscopic practice.”
Study Details
The multicenter, open-label, parallel-arm, pragmatic randomized controlled trial was conducted at 12 National Health Service hospitals in England. The study cohort consisted of adults ≥ 18 years undergoing colorectal cancer (CRC) screening or colonoscopy for gastrointestinal symptom surveillance owing to personal or family history.
Recruiting staff, participants, and colonoscopists were unmasked to allocation, whereas histopathologists, cochief investigators, and trial statisticians were masked.
CADe-assisted colonoscopy consisted of standard colonoscopy plus the GI Genius module active for at least the entire inspection phase of colonoscope withdrawal.
The primary outcome was mean adenomas per procedure (total number of adenomas detected divided by total number of procedures). The key secondary outcome was adenoma detection rate (proportion of colonoscopies with at least one adenoma).
From March 2021 to April 2023, the investigators recruited 2032 participants, 55.7% men, with a mean cohort age of 62.4 years and randomly assigned them to CADe-assisted colonoscopy (n = 1015) or to standard colonoscopy (n = 1017). Of these, 60.6% were undergoing screening and 39.4% had symptomatic indications.
Mean adenomas per procedure were 1.56 (SD, 2.82; n = 1001 participants with data) in the CADe-assisted group vs 1.21 (n = 1009) in the standard group, for an adjusted mean difference of 0.36 (95% CI, 0.14-0.57; adjusted incidence rate ratio, 1.30; 95% CI, 1.15-1.47; P < .0001).
Adenomas were detected in 555 (56.6%) of 980 participants in the CADe-assisted group vs 477 (48.4%) of 986 in the standard group, representing a proportion difference of 8.3% (95% CI, 3.9-12.7; adjusted odds ratio, 1.47; 95% CI, 1.21-1.78; P < .0001).
As to safety, adverse events were numerically comparable in both the intervention and control groups, with overall events 25 vs 19 and serious events 4 vs 6. On independent review, no adverse events in the CADe-assisted colonoscopy group were related to GI Genius.
Offering a US perspective on the study, Nabil M. Mansour, MD, an associate professor and director of the McNair General GI Clinic at Baylor College of Medicine in Houston, Texas, said GI Genius and other CADe systems represent a significant advance over standard colonoscopy for identifying premalignant polyps. “While the data have been mixed, most studies, particularly randomized controlled trials have shown significant improvements with CADe in detection both terms of in adenomas per colonoscopy and reductions in adenoma miss rate,” he said in an interview.
He added that the main utility of CADe is for asymptomatic patients undergoing average-risk screening and surveillance colonoscopy for CRC screening and prevention, as well as for those with positive stool-based screening tests, “though there is no downside to using it in symptomatic patients as well.” Though AI colonoscopy likely still stands at < 50% of endoscopy centers overall, and is used mainly at academic centers, his clinic has been using it for the past year.
The main question, Mansour cautioned, is whether increased detection of small polyps will actually reduce CRC incidence or mortality, and it will likely be several years before clear, concrete data can answer that.
“Most studies have shown the improvement in adenoma detection is mainly for diminutive polyps < 5 mm in diameter, but whether that will actually translate to substantive improvements in hard outcomes is as yet unknown,” he said. “But if gastroenterologists are interested in doing everything they can today to help improve detection rates and lower miss rates of premalignant polyps, serious consideration should be given to adopting the use of CADe in practice.”
This study was supported by Medtronic. Rees reported receiving grant funding from ARC Medical, Norgine, Medtronic, 3-D Matrix, and Olympus Medical, and has been an expert witness for ARC Medical. Other authors disclosed receiving research funding, honoraria, or travel expenses from Medtronic or other private companies. Mansour had no competing interests to declare.
A version of this article appeared on Medscape.com.
Colin J. Rees, a professor of gastroenterology in the Faculty of Medical Sciences at Newcastle University in Newcastle upon Tyne, England, and colleagues compared the real-world clinical effectiveness of computer-aided detection (CADe)–assisted colonoscopy using an “intelligent” module with that of standard colonoscopy in a study in The Lancet Gastroenterology & Hepatology.
They found the GI Genius Intelligent Endoscopy Module (Medtronic) increased the mean number of adenomas detected per procedure and the adenoma detection rate, especially for small, flat (type 0-IIa) polyps, and sessile serrated lesions, which are more likely to be missed.
“Missed sessile serrated lesions disproportionately increase the risk of post-colonoscopy colorectal cancer, thus the adoption of GI Genius into routine colonoscopy practice could not only increase polyp detection but also reduce the incidence of post-colonoscopy colorectal cancer,” the investigators wrote.
“AI is going to have a major impact upon most aspects of healthcare. Some areas of medical practice are now well established, and some are still in evolution,” Rees, who is also president of the British Society of Gastroenterology, said in an interview. “Within gastroenterology, the role of AI in endoscopic diagnostics is also evolving. The COLO-DETECT trial demonstrates that AI increases detection of lesions, and work is ongoing to see how AI might help with characterization and other elements of endoscopic practice.”
Study Details
The multicenter, open-label, parallel-arm, pragmatic randomized controlled trial was conducted at 12 National Health Service hospitals in England. The study cohort consisted of adults ≥ 18 years undergoing colorectal cancer (CRC) screening or colonoscopy for gastrointestinal symptom surveillance owing to personal or family history.
Recruiting staff, participants, and colonoscopists were unmasked to allocation, whereas histopathologists, cochief investigators, and trial statisticians were masked.
CADe-assisted colonoscopy consisted of standard colonoscopy plus the GI Genius module active for at least the entire inspection phase of colonoscope withdrawal.
The primary outcome was mean adenomas per procedure (total number of adenomas detected divided by total number of procedures). The key secondary outcome was adenoma detection rate (proportion of colonoscopies with at least one adenoma).
From March 2021 to April 2023, the investigators recruited 2032 participants, 55.7% men, with a mean cohort age of 62.4 years and randomly assigned them to CADe-assisted colonoscopy (n = 1015) or to standard colonoscopy (n = 1017). Of these, 60.6% were undergoing screening and 39.4% had symptomatic indications.
Mean adenomas per procedure were 1.56 (SD, 2.82; n = 1001 participants with data) in the CADe-assisted group vs 1.21 (n = 1009) in the standard group, for an adjusted mean difference of 0.36 (95% CI, 0.14-0.57; adjusted incidence rate ratio, 1.30; 95% CI, 1.15-1.47; P < .0001).
Adenomas were detected in 555 (56.6%) of 980 participants in the CADe-assisted group vs 477 (48.4%) of 986 in the standard group, representing a proportion difference of 8.3% (95% CI, 3.9-12.7; adjusted odds ratio, 1.47; 95% CI, 1.21-1.78; P < .0001).
As to safety, adverse events were numerically comparable in both the intervention and control groups, with overall events 25 vs 19 and serious events 4 vs 6. On independent review, no adverse events in the CADe-assisted colonoscopy group were related to GI Genius.
Offering a US perspective on the study, Nabil M. Mansour, MD, an associate professor and director of the McNair General GI Clinic at Baylor College of Medicine in Houston, Texas, said GI Genius and other CADe systems represent a significant advance over standard colonoscopy for identifying premalignant polyps. “While the data have been mixed, most studies, particularly randomized controlled trials have shown significant improvements with CADe in detection both terms of in adenomas per colonoscopy and reductions in adenoma miss rate,” he said in an interview.
He added that the main utility of CADe is for asymptomatic patients undergoing average-risk screening and surveillance colonoscopy for CRC screening and prevention, as well as for those with positive stool-based screening tests, “though there is no downside to using it in symptomatic patients as well.” Though AI colonoscopy likely still stands at < 50% of endoscopy centers overall, and is used mainly at academic centers, his clinic has been using it for the past year.
The main question, Mansour cautioned, is whether increased detection of small polyps will actually reduce CRC incidence or mortality, and it will likely be several years before clear, concrete data can answer that.
“Most studies have shown the improvement in adenoma detection is mainly for diminutive polyps < 5 mm in diameter, but whether that will actually translate to substantive improvements in hard outcomes is as yet unknown,” he said. “But if gastroenterologists are interested in doing everything they can today to help improve detection rates and lower miss rates of premalignant polyps, serious consideration should be given to adopting the use of CADe in practice.”
This study was supported by Medtronic. Rees reported receiving grant funding from ARC Medical, Norgine, Medtronic, 3-D Matrix, and Olympus Medical, and has been an expert witness for ARC Medical. Other authors disclosed receiving research funding, honoraria, or travel expenses from Medtronic or other private companies. Mansour had no competing interests to declare.
A version of this article appeared on Medscape.com.
Colin J. Rees, a professor of gastroenterology in the Faculty of Medical Sciences at Newcastle University in Newcastle upon Tyne, England, and colleagues compared the real-world clinical effectiveness of computer-aided detection (CADe)–assisted colonoscopy using an “intelligent” module with that of standard colonoscopy in a study in The Lancet Gastroenterology & Hepatology.
They found the GI Genius Intelligent Endoscopy Module (Medtronic) increased the mean number of adenomas detected per procedure and the adenoma detection rate, especially for small, flat (type 0-IIa) polyps, and sessile serrated lesions, which are more likely to be missed.
“Missed sessile serrated lesions disproportionately increase the risk of post-colonoscopy colorectal cancer, thus the adoption of GI Genius into routine colonoscopy practice could not only increase polyp detection but also reduce the incidence of post-colonoscopy colorectal cancer,” the investigators wrote.
“AI is going to have a major impact upon most aspects of healthcare. Some areas of medical practice are now well established, and some are still in evolution,” Rees, who is also president of the British Society of Gastroenterology, said in an interview. “Within gastroenterology, the role of AI in endoscopic diagnostics is also evolving. The COLO-DETECT trial demonstrates that AI increases detection of lesions, and work is ongoing to see how AI might help with characterization and other elements of endoscopic practice.”
Study Details
The multicenter, open-label, parallel-arm, pragmatic randomized controlled trial was conducted at 12 National Health Service hospitals in England. The study cohort consisted of adults ≥ 18 years undergoing colorectal cancer (CRC) screening or colonoscopy for gastrointestinal symptom surveillance owing to personal or family history.
Recruiting staff, participants, and colonoscopists were unmasked to allocation, whereas histopathologists, cochief investigators, and trial statisticians were masked.
CADe-assisted colonoscopy consisted of standard colonoscopy plus the GI Genius module active for at least the entire inspection phase of colonoscope withdrawal.
The primary outcome was mean adenomas per procedure (total number of adenomas detected divided by total number of procedures). The key secondary outcome was adenoma detection rate (proportion of colonoscopies with at least one adenoma).
From March 2021 to April 2023, the investigators recruited 2032 participants, 55.7% men, with a mean cohort age of 62.4 years and randomly assigned them to CADe-assisted colonoscopy (n = 1015) or to standard colonoscopy (n = 1017). Of these, 60.6% were undergoing screening and 39.4% had symptomatic indications.
Mean adenomas per procedure were 1.56 (SD, 2.82; n = 1001 participants with data) in the CADe-assisted group vs 1.21 (n = 1009) in the standard group, for an adjusted mean difference of 0.36 (95% CI, 0.14-0.57; adjusted incidence rate ratio, 1.30; 95% CI, 1.15-1.47; P < .0001).
Adenomas were detected in 555 (56.6%) of 980 participants in the CADe-assisted group vs 477 (48.4%) of 986 in the standard group, representing a proportion difference of 8.3% (95% CI, 3.9-12.7; adjusted odds ratio, 1.47; 95% CI, 1.21-1.78; P < .0001).
As to safety, adverse events were numerically comparable in both the intervention and control groups, with overall events 25 vs 19 and serious events 4 vs 6. On independent review, no adverse events in the CADe-assisted colonoscopy group were related to GI Genius.
Offering a US perspective on the study, Nabil M. Mansour, MD, an associate professor and director of the McNair General GI Clinic at Baylor College of Medicine in Houston, Texas, said GI Genius and other CADe systems represent a significant advance over standard colonoscopy for identifying premalignant polyps. “While the data have been mixed, most studies, particularly randomized controlled trials have shown significant improvements with CADe in detection both terms of in adenomas per colonoscopy and reductions in adenoma miss rate,” he said in an interview.
He added that the main utility of CADe is for asymptomatic patients undergoing average-risk screening and surveillance colonoscopy for CRC screening and prevention, as well as for those with positive stool-based screening tests, “though there is no downside to using it in symptomatic patients as well.” Though AI colonoscopy likely still stands at < 50% of endoscopy centers overall, and is used mainly at academic centers, his clinic has been using it for the past year.
The main question, Mansour cautioned, is whether increased detection of small polyps will actually reduce CRC incidence or mortality, and it will likely be several years before clear, concrete data can answer that.
“Most studies have shown the improvement in adenoma detection is mainly for diminutive polyps < 5 mm in diameter, but whether that will actually translate to substantive improvements in hard outcomes is as yet unknown,” he said. “But if gastroenterologists are interested in doing everything they can today to help improve detection rates and lower miss rates of premalignant polyps, serious consideration should be given to adopting the use of CADe in practice.”
This study was supported by Medtronic. Rees reported receiving grant funding from ARC Medical, Norgine, Medtronic, 3-D Matrix, and Olympus Medical, and has been an expert witness for ARC Medical. Other authors disclosed receiving research funding, honoraria, or travel expenses from Medtronic or other private companies. Mansour had no competing interests to declare.
A version of this article appeared on Medscape.com.
FROM THE LANCET GASTROENTEROLOGY & HEPATOLOGY