User login
From debate to stalemate and hate: An epidemic of intellectual constipation
Groupthink is hazardous, especially when perfused with religious fervor. It can lead to adopting irrational thinking1 and aversion to new ideas or facts. Tenaciously clinging to 1 ideology as “the absolute truth” precludes an open-minded, constructive debate with any other point of view.
Three historical examples come to mind:
- The discovery of chlorpromazine in 1952 was a scientifically and clinically seismic and transformational event for the treatment of psychosis, which for centuries had been dogmatically deemed irreversible. Jean Delay, MD, the French psychiatrist and co-discoverer of chlorpromazine, was the first physician to witness the magical and dazzling dissolution of delusions and hallucinations in chronically institutionalized patients with psychosis.2 He published his landmark clinical observations and then traveled to the United States to share the great news and present his findings at a large psychiatric conference, hoping to enthrall American psychiatrists with the historic breakthrough in treating psychosis. This was an era in which psychoanalysis dominated American psychiatry (despite its dearth of empirical evidence). Dr. Delay was shocked when the audience of psychoanalysts booed him for saying that psychosis can be treated with a medication instead of with psychoanalysis (which, in the most intense groupthink in the history of psychiatry, they all believed was the only therapy for psychosis). Deeply disheartened, Dr. Delay returned to France and never returned to the United States. This groupthink was a prime example of intellectual constipation. Since then, not surprisingly, psychopharmacology grew meteorically while psychoanalysis declined precipitously.
- The monoamine hypothesis of depression, first propagated 60 years ago, became a groupthink dogma among psychiatric researchers for the next several decades, stultifying broader antidepressant medication development by focusing only on monoamines (eg, serotonin, norepinephrine, and dopamine). More recently, researchers have become more open-minded, and the monoamine hypothesis has taken a backseat to innovative new models of antidepressant therapy based on advances in the pathophysiology of depression, such as glutamatergic, opioid, and sigma pathways as well as neuroplasticity models.3 The consequence of groupthink in antidepressant research was a half-century delay in the development of effective alternative treatments that could have helped millions of patients recover from a life-threatening brain disorder such as major depressive disorder.
- Peptic ulcer and its serious gastritis were long believed to be due to stress and increased stomach acidity. So the groupthink gastroenterologists mocked 2 Australian researchers, Barry Marshall and Robin Warren, when they proposed that peptic ulcer may be due to an infection with a bacterium called Helicobacter pylori, and published their data demonstrating it.4 Marshall and Warren had the last laugh when they were awarded the 2005 Nobel Prize in Medicine and Physiology. It is ironic that even gastroenterologists are not immune to the affliction of intellectual constipation!
Intellectual constipation’s effects on youth
The principle of a civilized debate of contrarian ideas must be inculcated early, especially during college years. Youth should be mentored about not cowering into an ideological cocoon and shun listening to different or opposing points of view.5 Institutions of higher learning are incubators of future leaders. They must provide their young students with a wide diversity of ideas and philosophies and encourage them to critique those ideas, not “shelter” or isolate them from any ideas. Youth need to recognize that the complex societies in which we all live and work are not placid or unidimensional but a hotbed of clashing ideas and perspectives. An open-minded approach to education will inoculate young minds from developing intellectual constipation in adulthood.
Avoiding or insulating oneself from the ideas of others—no matter how disagreeable—leads to cognitive cowardice and behavioral intolerance. Healthy and vibrant debate is necessary as an inoculation against extremism, hate, paranoia, and, ultimately, violence. Psychiatrists help patients to self-reflect, gain insight, and consider changing their view of themselves and the world to help them grow into mature and resilient individuals. But for the millions of people with intellectual constipation, a potent cerebral enema comprised of a salubrious concoction of insight, common sense, and compromise may be the prescription to forestall lethal intellectual ileus.
1. Nasrallah HA. Irrational beliefs: a ubiquitous human trait. Current Psychiatry. 2007;6(2):15-16.
2. Ban TA. Fifty years chlorpromazine: a historical perspective. Neuropsychiatr Dis Treat. 2007;3(4):495-500.
3. Boku S, Nakagawa S, Toda H, et al. Neural basis of major depressive disorder: beyond monoamine hypothesis. Psychiatry Clin Neurosci. 2018;72(1):3-12.
4. Warren JR, Marshall B. Unidentified curved bacilli on gastric epithelium in active chronic gastritis. Lancet. 1983;1(8336):1273-1275.
5. Lukianoff G, Haidt J. The Coddling of the American Mind: How Good Intentions and Bad Ideas Are Setting Up a Generation for Failure. Penguin Books; 2018.
Groupthink is hazardous, especially when perfused with religious fervor. It can lead to adopting irrational thinking1 and aversion to new ideas or facts. Tenaciously clinging to 1 ideology as “the absolute truth” precludes an open-minded, constructive debate with any other point of view.
Three historical examples come to mind:
- The discovery of chlorpromazine in 1952 was a scientifically and clinically seismic and transformational event for the treatment of psychosis, which for centuries had been dogmatically deemed irreversible. Jean Delay, MD, the French psychiatrist and co-discoverer of chlorpromazine, was the first physician to witness the magical and dazzling dissolution of delusions and hallucinations in chronically institutionalized patients with psychosis.2 He published his landmark clinical observations and then traveled to the United States to share the great news and present his findings at a large psychiatric conference, hoping to enthrall American psychiatrists with the historic breakthrough in treating psychosis. This was an era in which psychoanalysis dominated American psychiatry (despite its dearth of empirical evidence). Dr. Delay was shocked when the audience of psychoanalysts booed him for saying that psychosis can be treated with a medication instead of with psychoanalysis (which, in the most intense groupthink in the history of psychiatry, they all believed was the only therapy for psychosis). Deeply disheartened, Dr. Delay returned to France and never returned to the United States. This groupthink was a prime example of intellectual constipation. Since then, not surprisingly, psychopharmacology grew meteorically while psychoanalysis declined precipitously.
- The monoamine hypothesis of depression, first propagated 60 years ago, became a groupthink dogma among psychiatric researchers for the next several decades, stultifying broader antidepressant medication development by focusing only on monoamines (eg, serotonin, norepinephrine, and dopamine). More recently, researchers have become more open-minded, and the monoamine hypothesis has taken a backseat to innovative new models of antidepressant therapy based on advances in the pathophysiology of depression, such as glutamatergic, opioid, and sigma pathways as well as neuroplasticity models.3 The consequence of groupthink in antidepressant research was a half-century delay in the development of effective alternative treatments that could have helped millions of patients recover from a life-threatening brain disorder such as major depressive disorder.
- Peptic ulcer and its serious gastritis were long believed to be due to stress and increased stomach acidity. So the groupthink gastroenterologists mocked 2 Australian researchers, Barry Marshall and Robin Warren, when they proposed that peptic ulcer may be due to an infection with a bacterium called Helicobacter pylori, and published their data demonstrating it.4 Marshall and Warren had the last laugh when they were awarded the 2005 Nobel Prize in Medicine and Physiology. It is ironic that even gastroenterologists are not immune to the affliction of intellectual constipation!
Intellectual constipation’s effects on youth
The principle of a civilized debate of contrarian ideas must be inculcated early, especially during college years. Youth should be mentored about not cowering into an ideological cocoon and shun listening to different or opposing points of view.5 Institutions of higher learning are incubators of future leaders. They must provide their young students with a wide diversity of ideas and philosophies and encourage them to critique those ideas, not “shelter” or isolate them from any ideas. Youth need to recognize that the complex societies in which we all live and work are not placid or unidimensional but a hotbed of clashing ideas and perspectives. An open-minded approach to education will inoculate young minds from developing intellectual constipation in adulthood.
Avoiding or insulating oneself from the ideas of others—no matter how disagreeable—leads to cognitive cowardice and behavioral intolerance. Healthy and vibrant debate is necessary as an inoculation against extremism, hate, paranoia, and, ultimately, violence. Psychiatrists help patients to self-reflect, gain insight, and consider changing their view of themselves and the world to help them grow into mature and resilient individuals. But for the millions of people with intellectual constipation, a potent cerebral enema comprised of a salubrious concoction of insight, common sense, and compromise may be the prescription to forestall lethal intellectual ileus.
Groupthink is hazardous, especially when perfused with religious fervor. It can lead to adopting irrational thinking1 and aversion to new ideas or facts. Tenaciously clinging to 1 ideology as “the absolute truth” precludes an open-minded, constructive debate with any other point of view.
Three historical examples come to mind:
- The discovery of chlorpromazine in 1952 was a scientifically and clinically seismic and transformational event for the treatment of psychosis, which for centuries had been dogmatically deemed irreversible. Jean Delay, MD, the French psychiatrist and co-discoverer of chlorpromazine, was the first physician to witness the magical and dazzling dissolution of delusions and hallucinations in chronically institutionalized patients with psychosis.2 He published his landmark clinical observations and then traveled to the United States to share the great news and present his findings at a large psychiatric conference, hoping to enthrall American psychiatrists with the historic breakthrough in treating psychosis. This was an era in which psychoanalysis dominated American psychiatry (despite its dearth of empirical evidence). Dr. Delay was shocked when the audience of psychoanalysts booed him for saying that psychosis can be treated with a medication instead of with psychoanalysis (which, in the most intense groupthink in the history of psychiatry, they all believed was the only therapy for psychosis). Deeply disheartened, Dr. Delay returned to France and never returned to the United States. This groupthink was a prime example of intellectual constipation. Since then, not surprisingly, psychopharmacology grew meteorically while psychoanalysis declined precipitously.
- The monoamine hypothesis of depression, first propagated 60 years ago, became a groupthink dogma among psychiatric researchers for the next several decades, stultifying broader antidepressant medication development by focusing only on monoamines (eg, serotonin, norepinephrine, and dopamine). More recently, researchers have become more open-minded, and the monoamine hypothesis has taken a backseat to innovative new models of antidepressant therapy based on advances in the pathophysiology of depression, such as glutamatergic, opioid, and sigma pathways as well as neuroplasticity models.3 The consequence of groupthink in antidepressant research was a half-century delay in the development of effective alternative treatments that could have helped millions of patients recover from a life-threatening brain disorder such as major depressive disorder.
- Peptic ulcer and its serious gastritis were long believed to be due to stress and increased stomach acidity. So the groupthink gastroenterologists mocked 2 Australian researchers, Barry Marshall and Robin Warren, when they proposed that peptic ulcer may be due to an infection with a bacterium called Helicobacter pylori, and published their data demonstrating it.4 Marshall and Warren had the last laugh when they were awarded the 2005 Nobel Prize in Medicine and Physiology. It is ironic that even gastroenterologists are not immune to the affliction of intellectual constipation!
Intellectual constipation’s effects on youth
The principle of a civilized debate of contrarian ideas must be inculcated early, especially during college years. Youth should be mentored about not cowering into an ideological cocoon and shun listening to different or opposing points of view.5 Institutions of higher learning are incubators of future leaders. They must provide their young students with a wide diversity of ideas and philosophies and encourage them to critique those ideas, not “shelter” or isolate them from any ideas. Youth need to recognize that the complex societies in which we all live and work are not placid or unidimensional but a hotbed of clashing ideas and perspectives. An open-minded approach to education will inoculate young minds from developing intellectual constipation in adulthood.
Avoiding or insulating oneself from the ideas of others—no matter how disagreeable—leads to cognitive cowardice and behavioral intolerance. Healthy and vibrant debate is necessary as an inoculation against extremism, hate, paranoia, and, ultimately, violence. Psychiatrists help patients to self-reflect, gain insight, and consider changing their view of themselves and the world to help them grow into mature and resilient individuals. But for the millions of people with intellectual constipation, a potent cerebral enema comprised of a salubrious concoction of insight, common sense, and compromise may be the prescription to forestall lethal intellectual ileus.
1. Nasrallah HA. Irrational beliefs: a ubiquitous human trait. Current Psychiatry. 2007;6(2):15-16.
2. Ban TA. Fifty years chlorpromazine: a historical perspective. Neuropsychiatr Dis Treat. 2007;3(4):495-500.
3. Boku S, Nakagawa S, Toda H, et al. Neural basis of major depressive disorder: beyond monoamine hypothesis. Psychiatry Clin Neurosci. 2018;72(1):3-12.
4. Warren JR, Marshall B. Unidentified curved bacilli on gastric epithelium in active chronic gastritis. Lancet. 1983;1(8336):1273-1275.
5. Lukianoff G, Haidt J. The Coddling of the American Mind: How Good Intentions and Bad Ideas Are Setting Up a Generation for Failure. Penguin Books; 2018.
1. Nasrallah HA. Irrational beliefs: a ubiquitous human trait. Current Psychiatry. 2007;6(2):15-16.
2. Ban TA. Fifty years chlorpromazine: a historical perspective. Neuropsychiatr Dis Treat. 2007;3(4):495-500.
3. Boku S, Nakagawa S, Toda H, et al. Neural basis of major depressive disorder: beyond monoamine hypothesis. Psychiatry Clin Neurosci. 2018;72(1):3-12.
4. Warren JR, Marshall B. Unidentified curved bacilli on gastric epithelium in active chronic gastritis. Lancet. 1983;1(8336):1273-1275.
5. Lukianoff G, Haidt J. The Coddling of the American Mind: How Good Intentions and Bad Ideas Are Setting Up a Generation for Failure. Penguin Books; 2018.
GLP-1 agonists for weight loss: What you need to know
Obesity and overweight, with or without metabolic dysregulation, pose vexing problems for many patients with mood, anxiety, or psychotic disorders. More than one-half of individuals with severe mental illnesses are obese or overweight,1 resulting from multiple factors that may include psychiatric symptoms (eg, anergia and hyperphagia), poor dietary choices, sedentary lifestyle, underlying inflammatory processes, medical comorbidities, and iatrogenic consequences of certain medications. Unfortunately, numerous psychotropic medications can increase weight and appetite due to a variety of mechanisms, including antihistaminergic effects, direct appetite-stimulating effects, and proclivities to cause insulin resistance. While individual agents can vary, a recent review identified an overall 2-fold increased risk for rapid, significant weight gain during treatment with antipsychotics as a class.2 In addition to lifestyle modifications (diet and exercise), many pharmacologic strategies have been proposed to counter iatrogenic weight gain, including appetite suppressants (eg, pro-dopaminergic agents such as phentermine, stimulants, and amantadine), pro-anorectant anticonvulsants (eg, topiramate or zonisamide), opioid receptor antagonists (eg, olanzapine/samidorphan or naltrexone) and oral hypoglycemics such as metformin. However, the magnitude of impact for most of these agents to reverse iatrogenic weight gain tends to be modest, particularly once significant weight gain (ie, ≥7% of initial body weight) has already occurred.
Pharmacologic strategies to modulate or enhance the effects of insulin hold particular importance for combatting psychotropic-associated weight gain. Insulin transports glucose from the intravascular space to end organs for fuel consumption; to varying degrees, second-generation antipsychotics (SGAs) and some other psychotropic medications can cause insulin resistance. This in turn leads to excessive storage of underutilized glucose in the liver (glycogenesis), the potential for developing fatty liver (ie, nonalcoholic steatohepatitis), and conversion of excess carbohydrates to fatty acids and triglycerides, with subsequent storage in adipose tissue. Medications that can enhance the activity of insulin (so-called incretin mimetics) can help to overcome insulin resistance caused by SGAs (and potentially by other psychotropic medications) and essentially lead to weight loss through enhanced “fuel efficiency.”
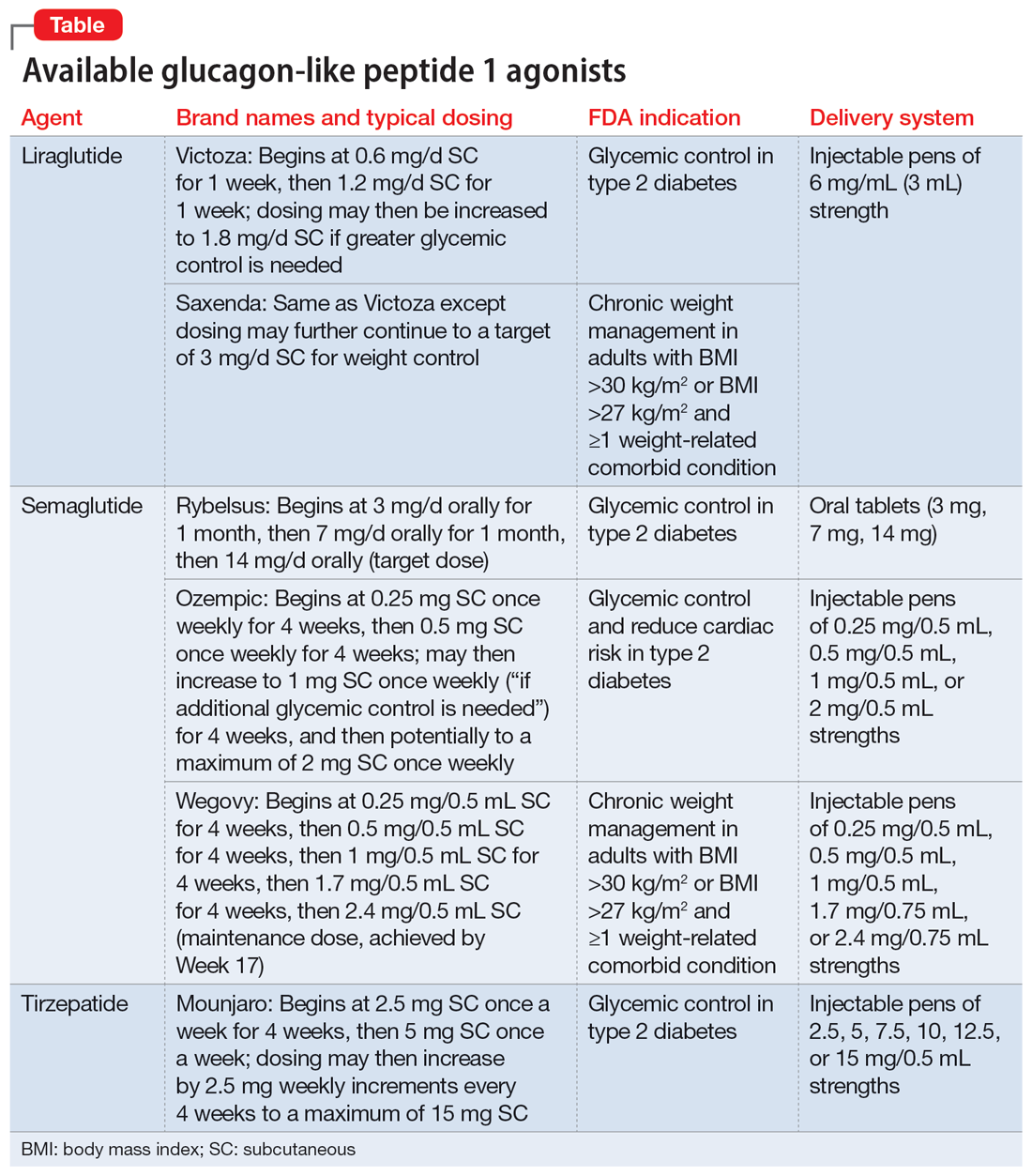
Metformin, typically dosed up to 1,000 mg twice daily with meals, has increasingly become recognized as a first-line strategy to attenuate weight gain and glycemic dysregulation from SGAs via its ability to reduce insulin resistance. Yet meta-analyses have shown that although results are significantly better than placebo, overall long-term weight loss from metformin alone tends to be rather modest (<4 kg) and associated with a reduction in body mass index (BMI) of only approximately 1 point.3 Psychiatrists (and other clinicians who prescribe psychotropic medications that can cause weight gain or metabolic dysregulation) therefore need to become familiar with alternative or adjunctive weight loss options. The use of a relatively new class of incretin mimetics called glucagon-like peptide 1 (GLP-1) agonists (Table) has been associated with profound and often dramatic weight loss and improvement of glycemic parameters in patients with obesity and glycemic dysregulation.
What are GLP-1 agonists?
GLP-1 is a hormone secreted by L cells in the intestinal mucosa in response to food. GLP-1 agonists reduce blood sugar by increasing insulin secretion, decreasing glucagon release (thus downregulating further increases in blood sugar), and reducing insulin resistance. GLP-1 agonists also reduce appetite by directly stimulating the satiety center and slowing gastric emptying and GI motility. In addition to GLP-1 agonism, some medications in this family (notably tirzepatide) also agonize a second hormone, glucose-dependent insulinotropic polypeptide, which can further induce insulin secretion as well as decrease stomach acid secretion, potentially delivering an even more substantial reduction in appetite and weight.
Routes of administration and FDA indications
Due to limited bioavailability, most GLP-1 agonists require subcutaneous (SC) injections (the sole exception is the Rybelsus brand of semaglutide, which comes in a daily pill form). Most are FDA-approved not specifically for weight loss but for patients with type 2 diabetes (defined as a hemoglobin A1C ≥6.5% or a fasting blood glucose level ≥126 mg/dL). Weight loss represents a secondary outcome for GLP-1 agonists FDA-approved for glycemic control in patients with type 2 diabetes. The 2 current exceptions to this classification are the Wegovy brand of semaglutide (ie, dosing of 2.4 mg) and the Saxenda brand of liraglutide, both of which carry FDA indications for chronic weight management alone (when paired with dietary and lifestyle modification) in individuals who are obese (BMI >30 kg/m2) regardless of the presence or absence of diabetes, or for persons who are overweight (BMI >27 kg/m2) and have ≥1 weight-related comorbid condition (eg, hypertension, type 2 diabetes, or dyslipidemia). Although patients at risk for diabetes (ie, prediabetes, defined as a hemoglobin A1C 5.7% to 6.4% or a fasting blood glucose level 100 to 125 mg/dL) were included in FDA registration trials of Saxenda or Wegovy, prediabetes is not an FDA indication for any GLP-1 agonist.
Data in weight loss
Most of the existing empirical data on weight loss with GLP-1 agonists come from studies of individuals who are overweight or obese, with or without type 2 diabetes, rather than from studies using these agents to counteract iatrogenic weight gain. In a retrospective cohort study of patients with type 2 diabetes, coadministration with serotonergic antidepressants (eg, citalopram/escitalopram) was associated with attenuation of the weight loss effects of GLP-1 agonists.4
Liraglutide currently is the sole GLP-1 agonist studied for treating SGA-associated weight gain. A 16-week randomized trial compared once-daily SC injected liraglutide vs placebo in patients with schizophrenia who incurred weight gain and prediabetes after taking olanzapine or clozapine.5 Significantly more patients taking liraglutide than placebo developed normal glucose tolerance (64% vs 16%), and body weight decreased by a mean of 5.3 kg.
Continue to: In studies of semaglutide...
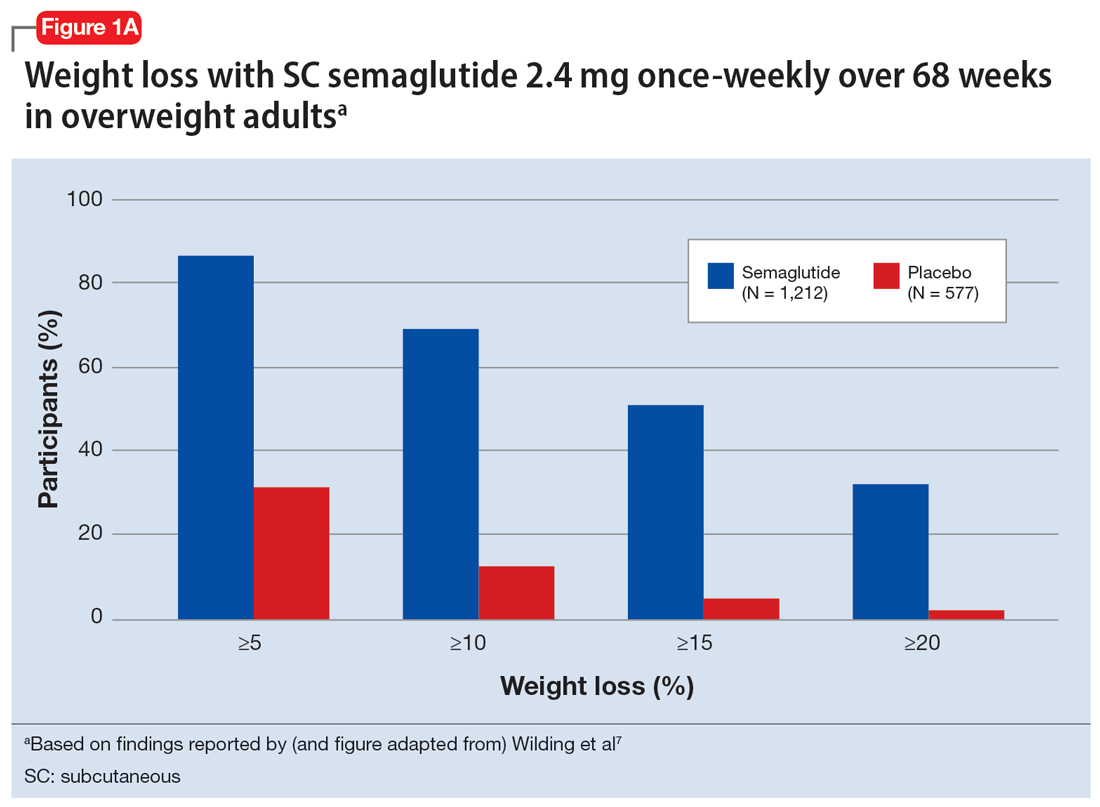
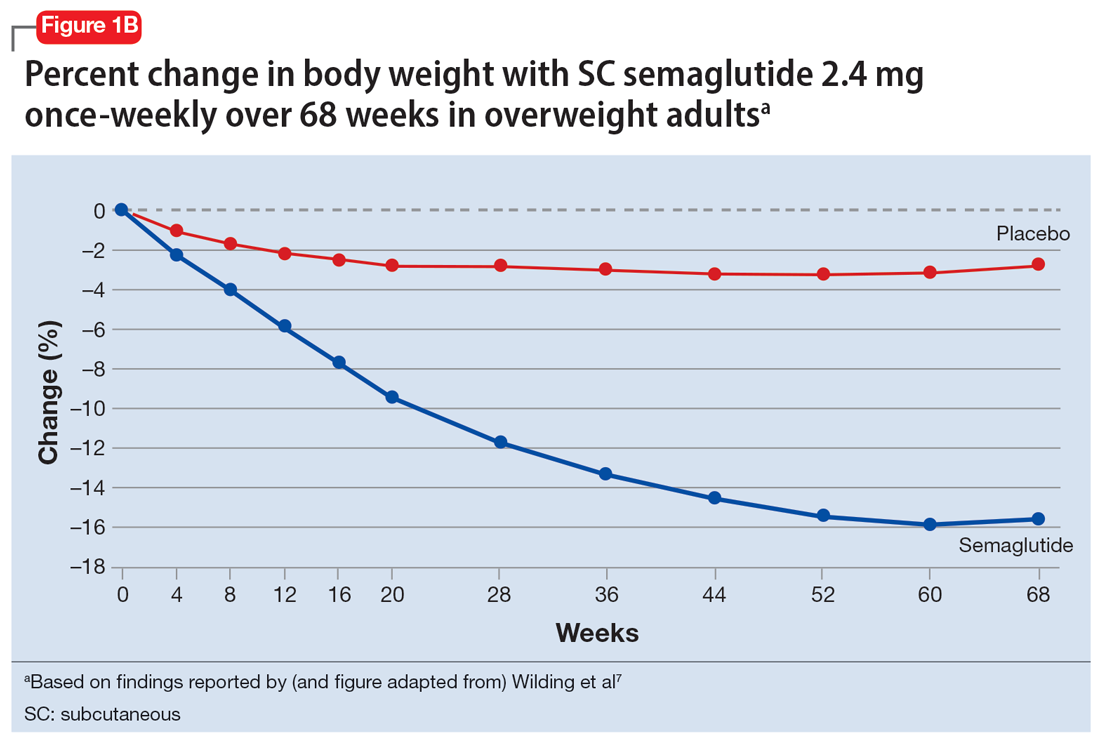
In studies of semaglutide for overweight/obese patients with type 2 diabetes or prediabetes, clinical trials of oral semaglutide (Rybelsus) found a mean weight loss over 26 weeks of -1.0 kg with dosing at 7 mg/d and -2.6 kg with dosing at 14 mg/d.6 A 68-week placebo-controlled trial of semaglutide (dosed at 2.4 mg SC weekly) for overweight/obese adults who did not have diabetes yielded a -15.3 kg weight loss (vs -2.6 kg with placebo); one-half of those who received semaglutide lost 15% of their initial body weight (Figure 1A and Figure 1B).7 Similar findings with semaglutide 2.4 mg SC weekly (Wegovy) were observed in overweight/obese adolescents, with 73% of participants losing ≥5% of their baseline weight.8 A comparative randomized trial in patients with type 2 diabetes also found modestly but significantly greater weight loss with oral semaglutide than with SC liraglutide.9
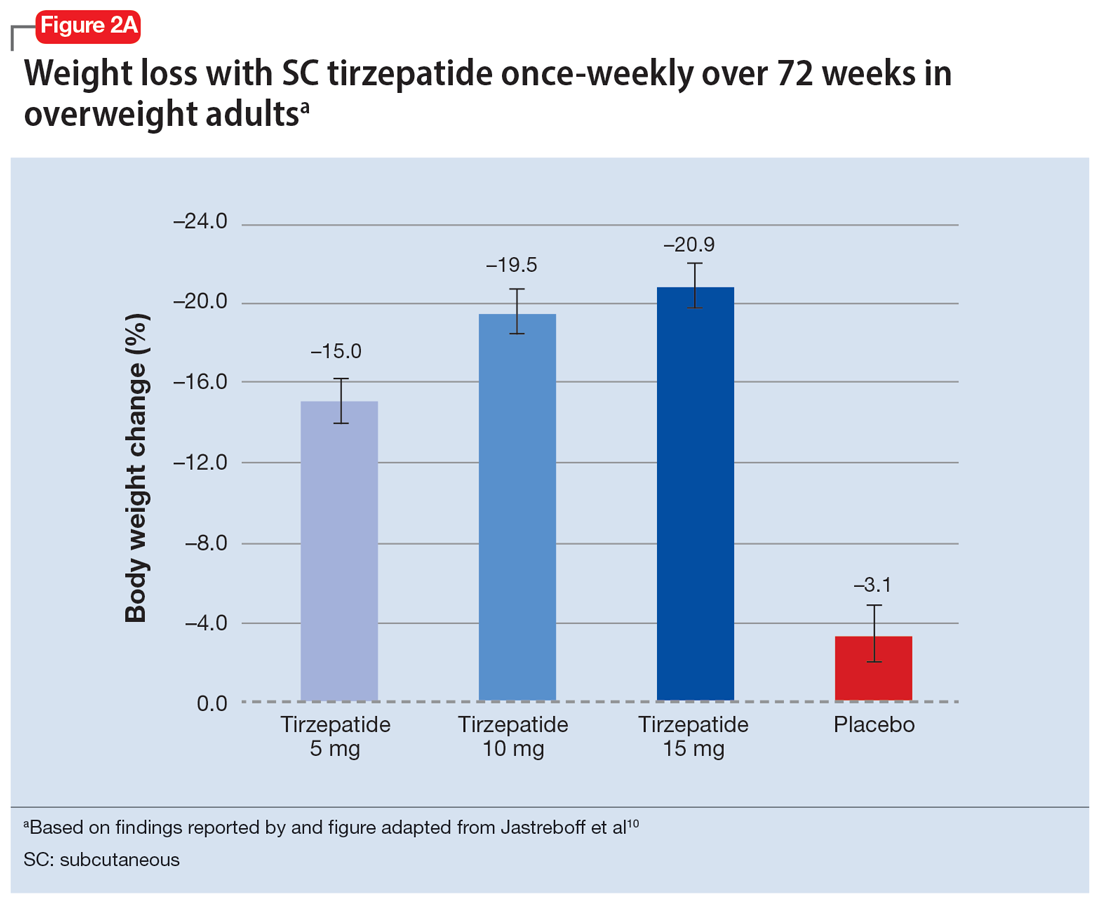
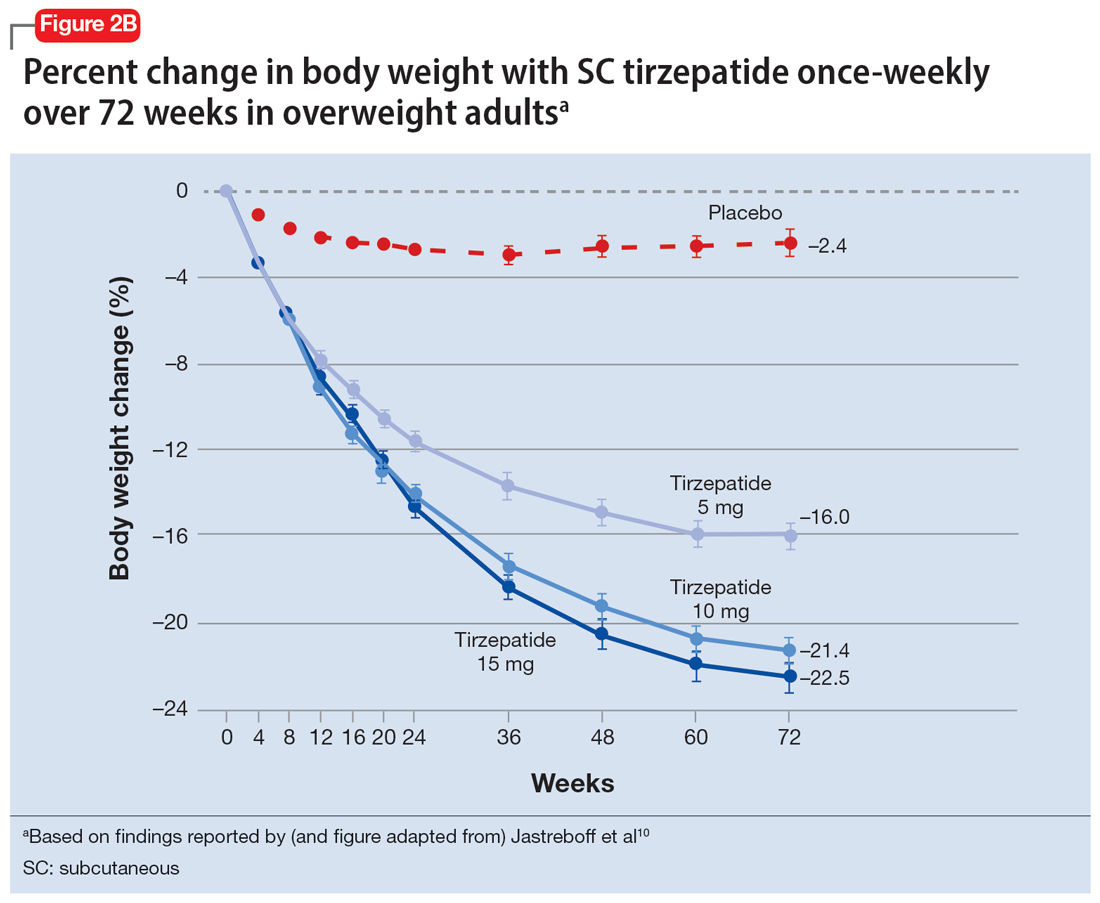
In a 72-week study of tirzepatide specifically for weight loss in nondiabetic patients who were overweight or obese, findings were especially dramatic (Figure 2A and Figure 2B).10 An overall 15% decrease in body weight was observed with 5 mg/week dosing alongside a 19.5% decrease in body weight with 10 mg/week dosing and a 20.9% weight reduction with 15 mg/week dosing.10 As noted in Figure 2B, the observed pattern of weight loss occurred along an exponential decay curve. Notably, a comparative study of tirzepatide vs once-weekly semaglutide (1 mg) in patients with type 2 diabetes11 found significantly greater dose-dependent weight loss with tirzepatide than semaglutide (-1.9 kg at 5 mg, -3.6 kg at 10 mg, and -5.5 kg at 15 mg)—although the somewhat low dosing of semaglutide may have limited its optimal possible weight loss benefit.
Tolerability
Adverse effects with GLP-1 agonists are mainly gastrointestinal (eg, nausea, vomiting, abdominal pain, diarrhea, or constipation)5-11 and generally transient. SC administration is performed in fatty tissue of the abdomen, thigh, or upper arm; site rotation is recommended to minimize injection site pain. All GLP-1 agonists carry manufacturers’ warning and precaution statements identifying the rare potential for acute pancreatitis, acute gall bladder disease, acute kidney injury, and hypoglycemia. Animal studies also have suggested an increased, dose-dependent risk for thyroid C-cell tumors with GLP-1 agonists; this has not been observed in human trials, although postmarketing pharmacovigilance reports have identified cases of medullary thyroid carcinoma in patients who took liraglutide. A manufacturer’s boxed warning indicates that a personal or family history of medullary carcinoma of the thyroid poses a contraindication for taking semaglutide, liraglutide, or tirzepatide.
Initial evidence prompts additional questions
GLP-1 agonists represent an emerging class of novel agents that can modulate glycemic dysregulation and overweight/obesity, often with dramatic results whose magnitude rivals the efficacy of bariatric surgery. Once-weekly formulations of semaglutide (Wegovy) and daily liraglutide (Saxenda) are FDA-approved for weight loss in patients who are overweight or obese while other existing formulations are approved solely for patients with type 2 diabetes, although it is likely that broader indications for weight loss (regardless of glycemic status) are forthcoming. Targeted use of GLP-1 agonists to counteract SGA-associated weight gain is supported by a handful of preliminary reports, with additional studies likely to come. Unanswered questions include:
- When should GLP-1 agonists be considered within a treatment algorithm for iatrogenic weight gain relative to other antidote strategies such as metformin or appetite-suppressing anticonvulsants?
- How effective might GLP-1 agonists be for iatrogenic weight gain from non-SGA psychotropic medications, such as serotonergic antidepressants?
- When and how can GLP-1 agonists be safely coprescribed with other nonincretin mimetic weight loss medications?
- When should psychiatrists prescribe GLP-1 agonists, or do so collaboratively with primary care physicians or endocrinologists, particularly in patients with metabolic syndrome?
Followers of the rapidly emerging literature in this area will likely find themselves best positioned to address these and other questions about optimal management of psychotropic-induced weight gain for the patients they treat.
Bottom Line
The use of glucagon-like peptide 1 (GLP-1) agonists, a relatively new class of incretin mimetics, has been associated with profound and often dramatic weight loss and improvement of glycemic parameters in patients with obesity and glycemic dysregulation. Preliminary reports support the potential targeted use of GLP-1 agonists to counteract weight gain associated with second-generation antipsychotics.
Related Resources
- Singh F, Allen A, Ianni A. Managing metabolic syndrome in patients with schizophrenia. Current Psychiatry. 2020;19(12):20-24,26. doi:10.12788/cp.0064
- Ard J, Fitch A, Fruh S, et al. Weight loss and maintenance related to the mechanism of action of glucagon-like peptide 1 receptor agonists. Adv Ther. 2021;38(6):2821- 2839. doi:10.1007/s12325-021-01710-0
Drug Brand Names
Amantadine • Gocovri
Citalopram • Celexa
Clozapine • Clozaril
Escitalopram • Lexapro
Liraglutide • Victoza, Saxenda
Metformin • Glucophage
Naltrexone • ReVia
Olanzapine • Zyprexa
Olanzapine/samidorphan • Lybalvi
Phentermine • Ionamin
Semaglutide • Rybelsus, Ozempic, Wegovy
Tirzepatide • Mounjaro
Topiramate • Topamax
Zonisamide • Zonegran
1. Afzal M, Siddiqi N, Ahmad B, et al. Prevalence of overweight and obesity in people with severe mental illness: systematic review and meta-analysis. Front Endocrinol (Lausanne). 2021;25;12:769309.
2. Barton BB, Segger F, Fischer K, et al. Update on weight-gain caused by antipsychotics: a systematic review and meta-analysis. Expert Opin Drug Safety. 2020;19(3):295-314.
3. de Silva AV, Suraweera C, Ratnatunga SS, et al. Metformin in prevention and treatment of antipsychotic induced weight gain: a systematic review and meta-analysis. BMC Psychiatry. 2016;16(1):341.
4. Durell N, Franks R, Coon S, et al. Effects of antidepressants on glucagon-like peptide-1 receptor agonist-related weight loss. J Pharm Technol. 2022;38(5):283-288.
5. Larsen JR, Vedtofte L, Jakobsen MSL, et al. Effect of liraglutide treatment on prediabetes and overweight or obesity in clozapine- or olanzapine-treated patients with schizophrenia spectrum disorder: a randomized clinical trial. JAMA Psychiatry. 2017;74(7):719-728.
6. Aroda VR, Rosenstock J, Terauchi Y, et al. PIONEER 1: randomized clinical trial of the efficacy and safety of oral semaglutide monotherapy in comparison with placebo in patients with type 2 diabetes. Diabetes Care. 2019;42(9):1724-1732.
7. Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384(11):989-1002.
8. Weghuber D, Barrett T, Barrientos-Pérez M, et al. Once-weekly semaglutide in adolescents with obesity. N Engl J Med. Published online November 2, 2022. doi:10.1056/NEJMoa2208601.
9. Pratley R, Amod A, Hoff ST, et al. Oral semaglutide versus subcutaneous liraglutide and placebo in type 2 diabetes (PIONEER 4): a randomized, double-blind, phase 3a trial. Lancet. 2019;394(10192):39-50.
10. Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387(3):205-216.
11. Frías JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med. 2021;385(6):503-515.
Obesity and overweight, with or without metabolic dysregulation, pose vexing problems for many patients with mood, anxiety, or psychotic disorders. More than one-half of individuals with severe mental illnesses are obese or overweight,1 resulting from multiple factors that may include psychiatric symptoms (eg, anergia and hyperphagia), poor dietary choices, sedentary lifestyle, underlying inflammatory processes, medical comorbidities, and iatrogenic consequences of certain medications. Unfortunately, numerous psychotropic medications can increase weight and appetite due to a variety of mechanisms, including antihistaminergic effects, direct appetite-stimulating effects, and proclivities to cause insulin resistance. While individual agents can vary, a recent review identified an overall 2-fold increased risk for rapid, significant weight gain during treatment with antipsychotics as a class.2 In addition to lifestyle modifications (diet and exercise), many pharmacologic strategies have been proposed to counter iatrogenic weight gain, including appetite suppressants (eg, pro-dopaminergic agents such as phentermine, stimulants, and amantadine), pro-anorectant anticonvulsants (eg, topiramate or zonisamide), opioid receptor antagonists (eg, olanzapine/samidorphan or naltrexone) and oral hypoglycemics such as metformin. However, the magnitude of impact for most of these agents to reverse iatrogenic weight gain tends to be modest, particularly once significant weight gain (ie, ≥7% of initial body weight) has already occurred.
Pharmacologic strategies to modulate or enhance the effects of insulin hold particular importance for combatting psychotropic-associated weight gain. Insulin transports glucose from the intravascular space to end organs for fuel consumption; to varying degrees, second-generation antipsychotics (SGAs) and some other psychotropic medications can cause insulin resistance. This in turn leads to excessive storage of underutilized glucose in the liver (glycogenesis), the potential for developing fatty liver (ie, nonalcoholic steatohepatitis), and conversion of excess carbohydrates to fatty acids and triglycerides, with subsequent storage in adipose tissue. Medications that can enhance the activity of insulin (so-called incretin mimetics) can help to overcome insulin resistance caused by SGAs (and potentially by other psychotropic medications) and essentially lead to weight loss through enhanced “fuel efficiency.”
Metformin, typically dosed up to 1,000 mg twice daily with meals, has increasingly become recognized as a first-line strategy to attenuate weight gain and glycemic dysregulation from SGAs via its ability to reduce insulin resistance. Yet meta-analyses have shown that although results are significantly better than placebo, overall long-term weight loss from metformin alone tends to be rather modest (<4 kg) and associated with a reduction in body mass index (BMI) of only approximately 1 point.3 Psychiatrists (and other clinicians who prescribe psychotropic medications that can cause weight gain or metabolic dysregulation) therefore need to become familiar with alternative or adjunctive weight loss options. The use of a relatively new class of incretin mimetics called glucagon-like peptide 1 (GLP-1) agonists (Table) has been associated with profound and often dramatic weight loss and improvement of glycemic parameters in patients with obesity and glycemic dysregulation.

What are GLP-1 agonists?
GLP-1 is a hormone secreted by L cells in the intestinal mucosa in response to food. GLP-1 agonists reduce blood sugar by increasing insulin secretion, decreasing glucagon release (thus downregulating further increases in blood sugar), and reducing insulin resistance. GLP-1 agonists also reduce appetite by directly stimulating the satiety center and slowing gastric emptying and GI motility. In addition to GLP-1 agonism, some medications in this family (notably tirzepatide) also agonize a second hormone, glucose-dependent insulinotropic polypeptide, which can further induce insulin secretion as well as decrease stomach acid secretion, potentially delivering an even more substantial reduction in appetite and weight.
Routes of administration and FDA indications
Due to limited bioavailability, most GLP-1 agonists require subcutaneous (SC) injections (the sole exception is the Rybelsus brand of semaglutide, which comes in a daily pill form). Most are FDA-approved not specifically for weight loss but for patients with type 2 diabetes (defined as a hemoglobin A1C ≥6.5% or a fasting blood glucose level ≥126 mg/dL). Weight loss represents a secondary outcome for GLP-1 agonists FDA-approved for glycemic control in patients with type 2 diabetes. The 2 current exceptions to this classification are the Wegovy brand of semaglutide (ie, dosing of 2.4 mg) and the Saxenda brand of liraglutide, both of which carry FDA indications for chronic weight management alone (when paired with dietary and lifestyle modification) in individuals who are obese (BMI >30 kg/m2) regardless of the presence or absence of diabetes, or for persons who are overweight (BMI >27 kg/m2) and have ≥1 weight-related comorbid condition (eg, hypertension, type 2 diabetes, or dyslipidemia). Although patients at risk for diabetes (ie, prediabetes, defined as a hemoglobin A1C 5.7% to 6.4% or a fasting blood glucose level 100 to 125 mg/dL) were included in FDA registration trials of Saxenda or Wegovy, prediabetes is not an FDA indication for any GLP-1 agonist.
Data in weight loss
Most of the existing empirical data on weight loss with GLP-1 agonists come from studies of individuals who are overweight or obese, with or without type 2 diabetes, rather than from studies using these agents to counteract iatrogenic weight gain. In a retrospective cohort study of patients with type 2 diabetes, coadministration with serotonergic antidepressants (eg, citalopram/escitalopram) was associated with attenuation of the weight loss effects of GLP-1 agonists.4
Liraglutide currently is the sole GLP-1 agonist studied for treating SGA-associated weight gain. A 16-week randomized trial compared once-daily SC injected liraglutide vs placebo in patients with schizophrenia who incurred weight gain and prediabetes after taking olanzapine or clozapine.5 Significantly more patients taking liraglutide than placebo developed normal glucose tolerance (64% vs 16%), and body weight decreased by a mean of 5.3 kg.
Continue to: In studies of semaglutide...
In studies of semaglutide for overweight/obese patients with type 2 diabetes or prediabetes, clinical trials of oral semaglutide (Rybelsus) found a mean weight loss over 26 weeks of -1.0 kg with dosing at 7 mg/d and -2.6 kg with dosing at 14 mg/d.6 A 68-week placebo-controlled trial of semaglutide (dosed at 2.4 mg SC weekly) for overweight/obese adults who did not have diabetes yielded a -15.3 kg weight loss (vs -2.6 kg with placebo); one-half of those who received semaglutide lost 15% of their initial body weight (Figure 1A and Figure 1B).7 Similar findings with semaglutide 2.4 mg SC weekly (Wegovy) were observed in overweight/obese adolescents, with 73% of participants losing ≥5% of their baseline weight.8 A comparative randomized trial in patients with type 2 diabetes also found modestly but significantly greater weight loss with oral semaglutide than with SC liraglutide.9


In a 72-week study of tirzepatide specifically for weight loss in nondiabetic patients who were overweight or obese, findings were especially dramatic (Figure 2A and Figure 2B).10 An overall 15% decrease in body weight was observed with 5 mg/week dosing alongside a 19.5% decrease in body weight with 10 mg/week dosing and a 20.9% weight reduction with 15 mg/week dosing.10 As noted in Figure 2B, the observed pattern of weight loss occurred along an exponential decay curve. Notably, a comparative study of tirzepatide vs once-weekly semaglutide (1 mg) in patients with type 2 diabetes11 found significantly greater dose-dependent weight loss with tirzepatide than semaglutide (-1.9 kg at 5 mg, -3.6 kg at 10 mg, and -5.5 kg at 15 mg)—although the somewhat low dosing of semaglutide may have limited its optimal possible weight loss benefit.


Tolerability
Adverse effects with GLP-1 agonists are mainly gastrointestinal (eg, nausea, vomiting, abdominal pain, diarrhea, or constipation)5-11 and generally transient. SC administration is performed in fatty tissue of the abdomen, thigh, or upper arm; site rotation is recommended to minimize injection site pain. All GLP-1 agonists carry manufacturers’ warning and precaution statements identifying the rare potential for acute pancreatitis, acute gall bladder disease, acute kidney injury, and hypoglycemia. Animal studies also have suggested an increased, dose-dependent risk for thyroid C-cell tumors with GLP-1 agonists; this has not been observed in human trials, although postmarketing pharmacovigilance reports have identified cases of medullary thyroid carcinoma in patients who took liraglutide. A manufacturer’s boxed warning indicates that a personal or family history of medullary carcinoma of the thyroid poses a contraindication for taking semaglutide, liraglutide, or tirzepatide.
Initial evidence prompts additional questions
GLP-1 agonists represent an emerging class of novel agents that can modulate glycemic dysregulation and overweight/obesity, often with dramatic results whose magnitude rivals the efficacy of bariatric surgery. Once-weekly formulations of semaglutide (Wegovy) and daily liraglutide (Saxenda) are FDA-approved for weight loss in patients who are overweight or obese while other existing formulations are approved solely for patients with type 2 diabetes, although it is likely that broader indications for weight loss (regardless of glycemic status) are forthcoming. Targeted use of GLP-1 agonists to counteract SGA-associated weight gain is supported by a handful of preliminary reports, with additional studies likely to come. Unanswered questions include:
- When should GLP-1 agonists be considered within a treatment algorithm for iatrogenic weight gain relative to other antidote strategies such as metformin or appetite-suppressing anticonvulsants?
- How effective might GLP-1 agonists be for iatrogenic weight gain from non-SGA psychotropic medications, such as serotonergic antidepressants?
- When and how can GLP-1 agonists be safely coprescribed with other nonincretin mimetic weight loss medications?
- When should psychiatrists prescribe GLP-1 agonists, or do so collaboratively with primary care physicians or endocrinologists, particularly in patients with metabolic syndrome?
Followers of the rapidly emerging literature in this area will likely find themselves best positioned to address these and other questions about optimal management of psychotropic-induced weight gain for the patients they treat.
Bottom Line
The use of glucagon-like peptide 1 (GLP-1) agonists, a relatively new class of incretin mimetics, has been associated with profound and often dramatic weight loss and improvement of glycemic parameters in patients with obesity and glycemic dysregulation. Preliminary reports support the potential targeted use of GLP-1 agonists to counteract weight gain associated with second-generation antipsychotics.
Related Resources
- Singh F, Allen A, Ianni A. Managing metabolic syndrome in patients with schizophrenia. Current Psychiatry. 2020;19(12):20-24,26. doi:10.12788/cp.0064
- Ard J, Fitch A, Fruh S, et al. Weight loss and maintenance related to the mechanism of action of glucagon-like peptide 1 receptor agonists. Adv Ther. 2021;38(6):2821- 2839. doi:10.1007/s12325-021-01710-0
Drug Brand Names
Amantadine • Gocovri
Citalopram • Celexa
Clozapine • Clozaril
Escitalopram • Lexapro
Liraglutide • Victoza, Saxenda
Metformin • Glucophage
Naltrexone • ReVia
Olanzapine • Zyprexa
Olanzapine/samidorphan • Lybalvi
Phentermine • Ionamin
Semaglutide • Rybelsus, Ozempic, Wegovy
Tirzepatide • Mounjaro
Topiramate • Topamax
Zonisamide • Zonegran
Obesity and overweight, with or without metabolic dysregulation, pose vexing problems for many patients with mood, anxiety, or psychotic disorders. More than one-half of individuals with severe mental illnesses are obese or overweight,1 resulting from multiple factors that may include psychiatric symptoms (eg, anergia and hyperphagia), poor dietary choices, sedentary lifestyle, underlying inflammatory processes, medical comorbidities, and iatrogenic consequences of certain medications. Unfortunately, numerous psychotropic medications can increase weight and appetite due to a variety of mechanisms, including antihistaminergic effects, direct appetite-stimulating effects, and proclivities to cause insulin resistance. While individual agents can vary, a recent review identified an overall 2-fold increased risk for rapid, significant weight gain during treatment with antipsychotics as a class.2 In addition to lifestyle modifications (diet and exercise), many pharmacologic strategies have been proposed to counter iatrogenic weight gain, including appetite suppressants (eg, pro-dopaminergic agents such as phentermine, stimulants, and amantadine), pro-anorectant anticonvulsants (eg, topiramate or zonisamide), opioid receptor antagonists (eg, olanzapine/samidorphan or naltrexone) and oral hypoglycemics such as metformin. However, the magnitude of impact for most of these agents to reverse iatrogenic weight gain tends to be modest, particularly once significant weight gain (ie, ≥7% of initial body weight) has already occurred.
Pharmacologic strategies to modulate or enhance the effects of insulin hold particular importance for combatting psychotropic-associated weight gain. Insulin transports glucose from the intravascular space to end organs for fuel consumption; to varying degrees, second-generation antipsychotics (SGAs) and some other psychotropic medications can cause insulin resistance. This in turn leads to excessive storage of underutilized glucose in the liver (glycogenesis), the potential for developing fatty liver (ie, nonalcoholic steatohepatitis), and conversion of excess carbohydrates to fatty acids and triglycerides, with subsequent storage in adipose tissue. Medications that can enhance the activity of insulin (so-called incretin mimetics) can help to overcome insulin resistance caused by SGAs (and potentially by other psychotropic medications) and essentially lead to weight loss through enhanced “fuel efficiency.”
Metformin, typically dosed up to 1,000 mg twice daily with meals, has increasingly become recognized as a first-line strategy to attenuate weight gain and glycemic dysregulation from SGAs via its ability to reduce insulin resistance. Yet meta-analyses have shown that although results are significantly better than placebo, overall long-term weight loss from metformin alone tends to be rather modest (<4 kg) and associated with a reduction in body mass index (BMI) of only approximately 1 point.3 Psychiatrists (and other clinicians who prescribe psychotropic medications that can cause weight gain or metabolic dysregulation) therefore need to become familiar with alternative or adjunctive weight loss options. The use of a relatively new class of incretin mimetics called glucagon-like peptide 1 (GLP-1) agonists (Table) has been associated with profound and often dramatic weight loss and improvement of glycemic parameters in patients with obesity and glycemic dysregulation.

What are GLP-1 agonists?
GLP-1 is a hormone secreted by L cells in the intestinal mucosa in response to food. GLP-1 agonists reduce blood sugar by increasing insulin secretion, decreasing glucagon release (thus downregulating further increases in blood sugar), and reducing insulin resistance. GLP-1 agonists also reduce appetite by directly stimulating the satiety center and slowing gastric emptying and GI motility. In addition to GLP-1 agonism, some medications in this family (notably tirzepatide) also agonize a second hormone, glucose-dependent insulinotropic polypeptide, which can further induce insulin secretion as well as decrease stomach acid secretion, potentially delivering an even more substantial reduction in appetite and weight.
Routes of administration and FDA indications
Due to limited bioavailability, most GLP-1 agonists require subcutaneous (SC) injections (the sole exception is the Rybelsus brand of semaglutide, which comes in a daily pill form). Most are FDA-approved not specifically for weight loss but for patients with type 2 diabetes (defined as a hemoglobin A1C ≥6.5% or a fasting blood glucose level ≥126 mg/dL). Weight loss represents a secondary outcome for GLP-1 agonists FDA-approved for glycemic control in patients with type 2 diabetes. The 2 current exceptions to this classification are the Wegovy brand of semaglutide (ie, dosing of 2.4 mg) and the Saxenda brand of liraglutide, both of which carry FDA indications for chronic weight management alone (when paired with dietary and lifestyle modification) in individuals who are obese (BMI >30 kg/m2) regardless of the presence or absence of diabetes, or for persons who are overweight (BMI >27 kg/m2) and have ≥1 weight-related comorbid condition (eg, hypertension, type 2 diabetes, or dyslipidemia). Although patients at risk for diabetes (ie, prediabetes, defined as a hemoglobin A1C 5.7% to 6.4% or a fasting blood glucose level 100 to 125 mg/dL) were included in FDA registration trials of Saxenda or Wegovy, prediabetes is not an FDA indication for any GLP-1 agonist.
Data in weight loss
Most of the existing empirical data on weight loss with GLP-1 agonists come from studies of individuals who are overweight or obese, with or without type 2 diabetes, rather than from studies using these agents to counteract iatrogenic weight gain. In a retrospective cohort study of patients with type 2 diabetes, coadministration with serotonergic antidepressants (eg, citalopram/escitalopram) was associated with attenuation of the weight loss effects of GLP-1 agonists.4
Liraglutide currently is the sole GLP-1 agonist studied for treating SGA-associated weight gain. A 16-week randomized trial compared once-daily SC injected liraglutide vs placebo in patients with schizophrenia who incurred weight gain and prediabetes after taking olanzapine or clozapine.5 Significantly more patients taking liraglutide than placebo developed normal glucose tolerance (64% vs 16%), and body weight decreased by a mean of 5.3 kg.
Continue to: In studies of semaglutide...
In studies of semaglutide for overweight/obese patients with type 2 diabetes or prediabetes, clinical trials of oral semaglutide (Rybelsus) found a mean weight loss over 26 weeks of -1.0 kg with dosing at 7 mg/d and -2.6 kg with dosing at 14 mg/d.6 A 68-week placebo-controlled trial of semaglutide (dosed at 2.4 mg SC weekly) for overweight/obese adults who did not have diabetes yielded a -15.3 kg weight loss (vs -2.6 kg with placebo); one-half of those who received semaglutide lost 15% of their initial body weight (Figure 1A and Figure 1B).7 Similar findings with semaglutide 2.4 mg SC weekly (Wegovy) were observed in overweight/obese adolescents, with 73% of participants losing ≥5% of their baseline weight.8 A comparative randomized trial in patients with type 2 diabetes also found modestly but significantly greater weight loss with oral semaglutide than with SC liraglutide.9


In a 72-week study of tirzepatide specifically for weight loss in nondiabetic patients who were overweight or obese, findings were especially dramatic (Figure 2A and Figure 2B).10 An overall 15% decrease in body weight was observed with 5 mg/week dosing alongside a 19.5% decrease in body weight with 10 mg/week dosing and a 20.9% weight reduction with 15 mg/week dosing.10 As noted in Figure 2B, the observed pattern of weight loss occurred along an exponential decay curve. Notably, a comparative study of tirzepatide vs once-weekly semaglutide (1 mg) in patients with type 2 diabetes11 found significantly greater dose-dependent weight loss with tirzepatide than semaglutide (-1.9 kg at 5 mg, -3.6 kg at 10 mg, and -5.5 kg at 15 mg)—although the somewhat low dosing of semaglutide may have limited its optimal possible weight loss benefit.


Tolerability
Adverse effects with GLP-1 agonists are mainly gastrointestinal (eg, nausea, vomiting, abdominal pain, diarrhea, or constipation)5-11 and generally transient. SC administration is performed in fatty tissue of the abdomen, thigh, or upper arm; site rotation is recommended to minimize injection site pain. All GLP-1 agonists carry manufacturers’ warning and precaution statements identifying the rare potential for acute pancreatitis, acute gall bladder disease, acute kidney injury, and hypoglycemia. Animal studies also have suggested an increased, dose-dependent risk for thyroid C-cell tumors with GLP-1 agonists; this has not been observed in human trials, although postmarketing pharmacovigilance reports have identified cases of medullary thyroid carcinoma in patients who took liraglutide. A manufacturer’s boxed warning indicates that a personal or family history of medullary carcinoma of the thyroid poses a contraindication for taking semaglutide, liraglutide, or tirzepatide.
Initial evidence prompts additional questions
GLP-1 agonists represent an emerging class of novel agents that can modulate glycemic dysregulation and overweight/obesity, often with dramatic results whose magnitude rivals the efficacy of bariatric surgery. Once-weekly formulations of semaglutide (Wegovy) and daily liraglutide (Saxenda) are FDA-approved for weight loss in patients who are overweight or obese while other existing formulations are approved solely for patients with type 2 diabetes, although it is likely that broader indications for weight loss (regardless of glycemic status) are forthcoming. Targeted use of GLP-1 agonists to counteract SGA-associated weight gain is supported by a handful of preliminary reports, with additional studies likely to come. Unanswered questions include:
- When should GLP-1 agonists be considered within a treatment algorithm for iatrogenic weight gain relative to other antidote strategies such as metformin or appetite-suppressing anticonvulsants?
- How effective might GLP-1 agonists be for iatrogenic weight gain from non-SGA psychotropic medications, such as serotonergic antidepressants?
- When and how can GLP-1 agonists be safely coprescribed with other nonincretin mimetic weight loss medications?
- When should psychiatrists prescribe GLP-1 agonists, or do so collaboratively with primary care physicians or endocrinologists, particularly in patients with metabolic syndrome?
Followers of the rapidly emerging literature in this area will likely find themselves best positioned to address these and other questions about optimal management of psychotropic-induced weight gain for the patients they treat.
Bottom Line
The use of glucagon-like peptide 1 (GLP-1) agonists, a relatively new class of incretin mimetics, has been associated with profound and often dramatic weight loss and improvement of glycemic parameters in patients with obesity and glycemic dysregulation. Preliminary reports support the potential targeted use of GLP-1 agonists to counteract weight gain associated with second-generation antipsychotics.
Related Resources
- Singh F, Allen A, Ianni A. Managing metabolic syndrome in patients with schizophrenia. Current Psychiatry. 2020;19(12):20-24,26. doi:10.12788/cp.0064
- Ard J, Fitch A, Fruh S, et al. Weight loss and maintenance related to the mechanism of action of glucagon-like peptide 1 receptor agonists. Adv Ther. 2021;38(6):2821- 2839. doi:10.1007/s12325-021-01710-0
Drug Brand Names
Amantadine • Gocovri
Citalopram • Celexa
Clozapine • Clozaril
Escitalopram • Lexapro
Liraglutide • Victoza, Saxenda
Metformin • Glucophage
Naltrexone • ReVia
Olanzapine • Zyprexa
Olanzapine/samidorphan • Lybalvi
Phentermine • Ionamin
Semaglutide • Rybelsus, Ozempic, Wegovy
Tirzepatide • Mounjaro
Topiramate • Topamax
Zonisamide • Zonegran
1. Afzal M, Siddiqi N, Ahmad B, et al. Prevalence of overweight and obesity in people with severe mental illness: systematic review and meta-analysis. Front Endocrinol (Lausanne). 2021;25;12:769309.
2. Barton BB, Segger F, Fischer K, et al. Update on weight-gain caused by antipsychotics: a systematic review and meta-analysis. Expert Opin Drug Safety. 2020;19(3):295-314.
3. de Silva AV, Suraweera C, Ratnatunga SS, et al. Metformin in prevention and treatment of antipsychotic induced weight gain: a systematic review and meta-analysis. BMC Psychiatry. 2016;16(1):341.
4. Durell N, Franks R, Coon S, et al. Effects of antidepressants on glucagon-like peptide-1 receptor agonist-related weight loss. J Pharm Technol. 2022;38(5):283-288.
5. Larsen JR, Vedtofte L, Jakobsen MSL, et al. Effect of liraglutide treatment on prediabetes and overweight or obesity in clozapine- or olanzapine-treated patients with schizophrenia spectrum disorder: a randomized clinical trial. JAMA Psychiatry. 2017;74(7):719-728.
6. Aroda VR, Rosenstock J, Terauchi Y, et al. PIONEER 1: randomized clinical trial of the efficacy and safety of oral semaglutide monotherapy in comparison with placebo in patients with type 2 diabetes. Diabetes Care. 2019;42(9):1724-1732.
7. Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384(11):989-1002.
8. Weghuber D, Barrett T, Barrientos-Pérez M, et al. Once-weekly semaglutide in adolescents with obesity. N Engl J Med. Published online November 2, 2022. doi:10.1056/NEJMoa2208601.
9. Pratley R, Amod A, Hoff ST, et al. Oral semaglutide versus subcutaneous liraglutide and placebo in type 2 diabetes (PIONEER 4): a randomized, double-blind, phase 3a trial. Lancet. 2019;394(10192):39-50.
10. Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387(3):205-216.
11. Frías JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med. 2021;385(6):503-515.
1. Afzal M, Siddiqi N, Ahmad B, et al. Prevalence of overweight and obesity in people with severe mental illness: systematic review and meta-analysis. Front Endocrinol (Lausanne). 2021;25;12:769309.
2. Barton BB, Segger F, Fischer K, et al. Update on weight-gain caused by antipsychotics: a systematic review and meta-analysis. Expert Opin Drug Safety. 2020;19(3):295-314.
3. de Silva AV, Suraweera C, Ratnatunga SS, et al. Metformin in prevention and treatment of antipsychotic induced weight gain: a systematic review and meta-analysis. BMC Psychiatry. 2016;16(1):341.
4. Durell N, Franks R, Coon S, et al. Effects of antidepressants on glucagon-like peptide-1 receptor agonist-related weight loss. J Pharm Technol. 2022;38(5):283-288.
5. Larsen JR, Vedtofte L, Jakobsen MSL, et al. Effect of liraglutide treatment on prediabetes and overweight or obesity in clozapine- or olanzapine-treated patients with schizophrenia spectrum disorder: a randomized clinical trial. JAMA Psychiatry. 2017;74(7):719-728.
6. Aroda VR, Rosenstock J, Terauchi Y, et al. PIONEER 1: randomized clinical trial of the efficacy and safety of oral semaglutide monotherapy in comparison with placebo in patients with type 2 diabetes. Diabetes Care. 2019;42(9):1724-1732.
7. Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384(11):989-1002.
8. Weghuber D, Barrett T, Barrientos-Pérez M, et al. Once-weekly semaglutide in adolescents with obesity. N Engl J Med. Published online November 2, 2022. doi:10.1056/NEJMoa2208601.
9. Pratley R, Amod A, Hoff ST, et al. Oral semaglutide versus subcutaneous liraglutide and placebo in type 2 diabetes (PIONEER 4): a randomized, double-blind, phase 3a trial. Lancet. 2019;394(10192):39-50.
10. Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387(3):205-216.
11. Frías JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med. 2021;385(6):503-515.
Managing excited catatonia: A suggested approach
Catatonia is often difficult to identify and treat. The excited catatonia subtype can be particularly challenging to diagnose because it can present with symptoms similar to those seen in mania or psychosis. In this article, we present 3 cases of excited catatonia that illustrate how to identify it, how to treat the catatonia as well as the underlying pathology, and factors to consider during this process to mitigate the risk of adverse outcomes. We also outline a treatment algorithm we used for the 3 cases. Although we describe using this approach for patients with excited catatonia, it is generalizable to other types of catatonia.
Many causes, varying presentations
Catatonia is a psychomotor syndrome characterized by mutism, negativism, stereotypy, waxy flexibility, and other symptoms.1 It is defined by the presence of ≥3 of the 12 symptoms listed in the Table.2 Causes of catatonia include metabolic abnormalities, endocrine disorders, drug intoxication, neurodevelopmental disorders, medication adverse effects, psychosis, and mood disorders.1,3

A subtype of this syndrome, excited catatonia, can present with restlessness, agitation, emotional lability, poor sleep, and altered mental status in addition to the more typical symptoms.1,4 Because excited catatonia can resemble mania or psychosis, it is particularly challenging to identify the underlying disorder causing it and appropriate treatment. Fink et al4 discussed how clinicians have interpreted the different presentations of excited catatonia to gain insight into the underlying diagnosis. If the patient’s thought process appears disorganized, psychosis may be suspected.4 If the patient is delusional and grandiose, they may be manic, and when altered mental status dominates the presentation, delirium may be the culprit.4
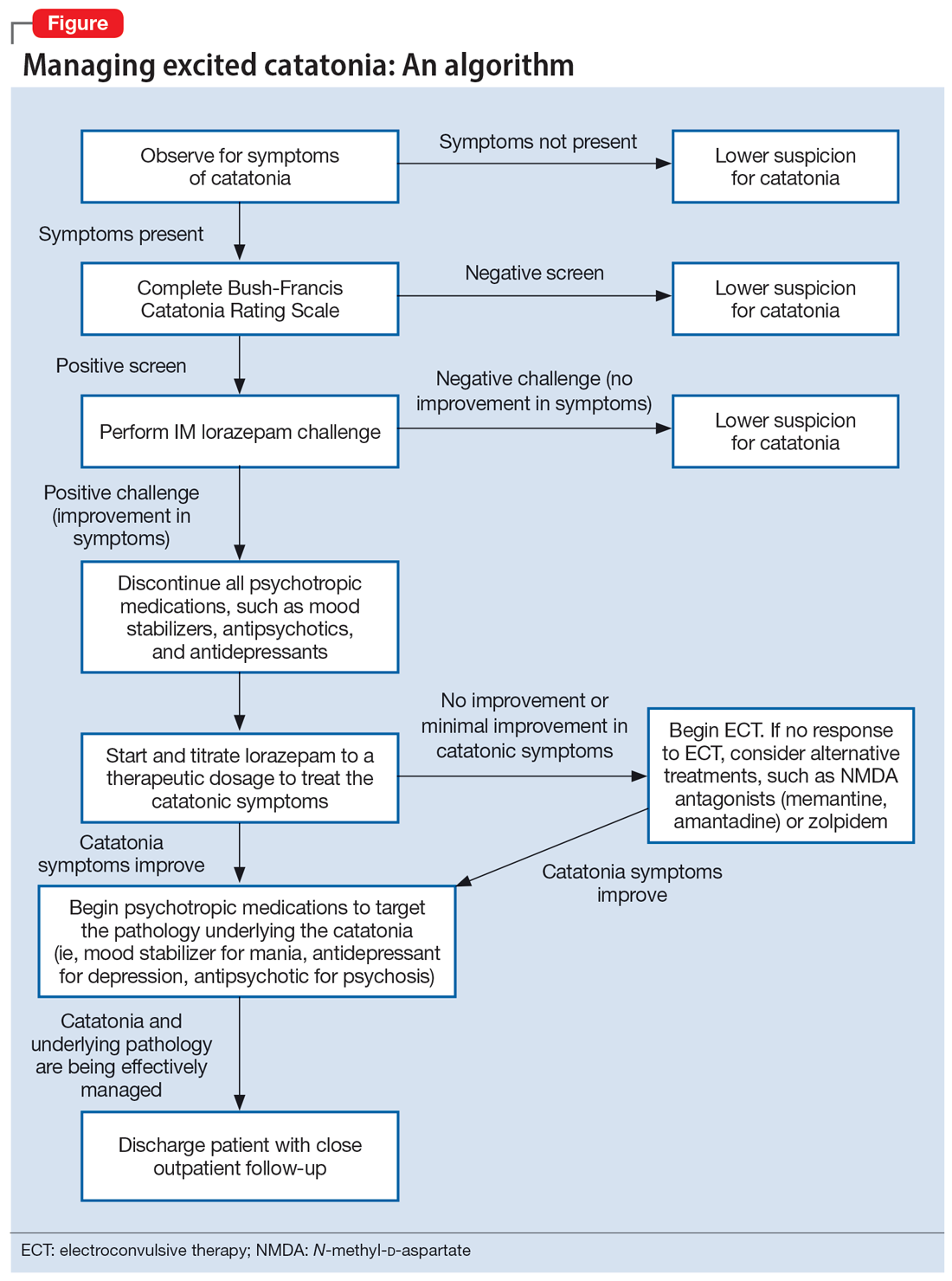
Regardless of the underlying cause, the first step is to treat the catatonia. Benzodiazepines and electroconvulsive therapy (ECT) are the most well validated treatments for catatonia and have been used to treat excited catatonia.1 Excited catatonia is often misdiagnosed and subsequently mistreated. In the following 3 cases, excited catatonia was successfully identified and treated using the same approach (Figure).
Case 1
Mr. A, age 27, has a history of bipolar I disorder. He was brought to the hospital by ambulance after being found to be yelling and acting belligerently, and he was admitted to the inpatient psychiatry unit for manic decompensation due to medication nonadherence. He was started on divalproex sodium 500 mg twice a day for mood stabilization, risperidone 1 mg twice a day for adjunct mood stabilization and psychosis, and lorazepam 1 mg 3 times a day for agitation. Mr. A exhibited odd behavior; he would take off his clothes in the hallway, run around the unit, and randomly yell at staff or to himself. At other times, he would stay silent, repeat the same statements, or oddly posture in the hallway for minutes at a time. These behaviors were seen primarily in the hour or 2 preceding lorazepam administration and improved after he received lorazepam.
Mr. A’s treating team completed the Bush-Francis Catatonia Rating Scale (BFCRS), which yielded a positive catatonia screen of 7/14. As a result, divalproex sodium and risperidone were held, and lorazepam was increased to 2 mg twice a day.
After several days, Mr. A was no longer acting oddly and was able to speak more spontaneously; however, he began to exhibit overt signs of mania. He would speak rapidly and make grandiose claims about managing millions of dollars as the CEO of a famous company. Divalproex sodium was restarted at 500 mg twice a day and increased to 500 mg 3 times a day for mood stabilization. Mr. A continued to receive lorazepam 2 mg 3 times a day for catatonia, and risperidone was restarted at 1 mg twice a day to more effectively target his manic symptoms. Risperidone was increased to 2 mg twice a day. After this change, Mr. A’s grandiosity dissipated, his speech normalized, and his thought process became organized. He was discharged on lorazepam 2 mg 3 times a day, divalproex sodium 500 mg 3 times a day, and risperidone 2 mg twice a day. Mr. A’s length of stay (LOS) for this admission was 11 days.
Continue to: Case 2
Case 2
Mr. B, age 49, presented with irritability and odd posturing. He has a history of schizoaffective disorder, bipolar type for which he was receiving a maintenance regimen of lithium 600 mg/d at bedtime and risperidone 2 mg/d at bedtime. He had multiple previous psychiatric admissions for catatonia. On this admission, Mr. B was irritable and difficult to redirect. He yelled at staff members and had a stiff gait. The BFCRS yielded a positive screening score of 3/14 and a severity score of 8/23. As a result, the treatment team conducted a lorazepam challenge.
After Mr. B received lorazepam 1 mg IM, his thought organization and irritability improved, which allowed him to have a coherent conversation with the interviewer. His gait stiffness also improved. His risperidone and lithium were held, and oral lorazepam 1 mg 3 times a day was started for catatonia. Lorazepam was gradually increased to 4 mg 3 times a day. Mr. B became euthymic and redirectable, and had an improved gait. However, he was also tangential and hyperverbal; these symptoms were indicative of the underlying mania that precipitated his catatonia.
Divalproex sodium extended release (ER) was started and increased to 1,500 mg/d at bedtime for mood stabilization. Lithium was restarted and increased to 300 mg twice a day for adjunct mood stabilization. Risperidone was not restarted. Toward the end of his admission, Mr. B was noted to be overly sedated, so the lorazepam dosage was decreased. He was discharged on lorazepam 2 mg 3 times a day, divalproex sodium ER 1,500 mg/d at bedtime, and lithium 300 mg twice a day. At discharge, Mr. B was calm and euthymic, with a linear thought process. His LOS was 25 days.
Case 3
Mr. C, age 62, presented to the emergency department (ED) because he had exhibited erratic behavior and had not slept for the past week. He has a history of bipolar I disorder, hypothyroidism, diabetes, and hypertension. For many years, he had been stable on divalproex sodium ER 2,500 mg/d at bedtime for mood stabilization and clozapine 100 mg/d at bedtime for adjunct mood stabilization and psychosis. In the ED, Mr. C was irritable, distractible, and tangential. On admission, he was speaking slowly with increased speech latency in response to questions, exhibiting stereotypy, repeating statements over and over, and walking very slowly.
The BFCRS yielded a positive screening score of 5/14 and a severity score of 10/23. Lorazepam 1 mg IM was administered. After 15 minutes, Mr. C’s speech, gait, and distractibility improved. As a result, clozapine and divalproex sodium were held, and he was started on oral lorazepam 1 mg 3 times a day. After several days, Mr. C was speaking fluently and no longer exhibiting stereotypy or having outbursts where he would make repetitive statements. However, he was tangential and irritable at times, which were signs of his underlying mania. Divalproex sodium ER was restarted at 250 mg/d at bedtime for mood stabilization and gradually increased to 2,500 mg/d at bedtime. Clozapine was also restarted at 25 mg/d at bedtime and gradually increased to 200 mg/d at bedtime. The lorazepam was gradually tapered and discontinued over the course of 3 weeks due to oversedation.
Continue to: At discharge...
At discharge, Mr. C was euthymic, calm, linear, and goal-directed. He was discharged on divalproex sodium ER 2,500 mg/d at bedtime and clozapine 200 mg/d at bedtime. His LOS for this admission was 22 days.
A stepwise approach can improve outcomes
The Figure outlines the method we used to manage excited catatonia in these 3 cases. Each of these patients exhibited signs of excited catatonia, but because those symptoms were nearly identical to those of mania, it was initially difficult to identify catatonia. Excited catatonia was suspected after more typical catatonic symptoms—such as a stiff gait, slowed speech, and stereotypy—were observed. The BFCRS was completed to get an objective measure of the likelihood that the patient was catatonic. In all 3 cases, the BFCRS resulted in a positive screen for catatonia. Following this, the patients described in Case 2 and Case 3 received a lorazepam challenge, which confirmed their catatonia. No lorazepam challenge was performed in Case 1 because the patient was already receiving lorazepam when the BFCRS was completed. Although most catatonic patients will respond to a lorazepam challenge, not all will. Therefore, clinicians should maintain some degree of suspicion for catatonia if a patient has a positive screen on the BFCRS but a negative lorazepam challenge.
In all 3 cases, after catatonia was confirmed, the patient’s psychotropic medications were discontinued. In all 3 cases, the antipsychotic was held to prevent progression to neuroleptic malignant syndrome (NMS) or malignant catatonia. Rasmussen et al3 found that 3.6% of the catatonic patients in their sample who were treated with antipsychotics developed NMS. A review of prospective studies looking at patients treated with antipsychotics found the incidence of NMS was .07% to 1.8%.5 Because NMS is often clinically indistinguishable from malignant catatonia,4,6 this incidence of NMS may have represented an increased incidence in malignant catatonia.
In all 3 cases, the mood stabilizer was held to prevent it from complicating the clinical picture. Discontinuing the mood stabilizer and focusing on treating the catatonia before targeting the underlying mania increased the likelihood of differentiating the patient’s catatonic symptoms from manic symptoms. This resulted in more precise medication selection and titration by allowing us to identify the specific symptoms that were being targeted by each medication.
Oral lorazepam was prescribed to target catatonia in all 3 cases, and the dosage was gradually increased until symptoms began to resolve. As the catatonia resolved, the manic symptoms became more easily identifiable, and at this point a mood stabilizer was started and titrated to a therapeutic dose to target the mania. In Case 1 and Case 3, the antipsychotic was restarted to treat the mania more effectively. It was not restarted in Case 2 because the patient’s mania was effectively being managed by 2 mood stabilizers. The risks and benefits of starting an antipsychotic in a catatonic or recently catatonic patient should be carefully considered. In the 2 cases where the antipsychotic was restarted, the patients were closely monitored, and there were no signs of NMS or malignant catatonia.
Continue to: As discharge approached...
As discharge approached, the dosages of oral lorazepam were reevaluated. Catatonic patients can typically tolerate high doses of benzodiazepines without becoming overly sedated, but each patient has a different threshold at which the dosage causes oversedation. In all 3 patients, lorazepam was initially titrated to a dose that treated their catatonic symptoms without causing intolerable sedation. In Case 2 and Case 3, as the catatonia began to resolve, the patients became increasingly sedated on their existing lorazepam dosage, so it was decreased. Because the patient in Case 1 did not become overly sedated, his lorazepam dosage did not need to be reduced.
For 2 of these patients, our approach resulted in a shorter LOS compared to their previous hospitalizations. The LOS in Case 2 was 25 days; 5 years earlier, he had a 49-day LOS for mania and catatonia. During the past admission, the identification and treatment of the catatonia was delayed, which resulted in the patient requiring multiple transfers to the medical unit for unstable vital signs. The LOS in Case 3 was 22 days; 6 months prior to this admission, the patient had 2 psychiatric admissions that totaled 37 days. Although the patient’s presentation in the 2 previous admissions was similar to his presentation as described in Case 3, catatonia had not been identified or treated in either admission. Since his catatonia and mania were treated in Case 3, he has not required a readmission. The patient in Case 1 was previously hospitalized, but information about the LOS of these admissions was not available. These results suggest that early identification and treatment of catatonia via the approach we used can improve patient outcomes.
Bottom Line
Excited catatonia can be challenging to diagnose and treat because it can present with symptoms similar to those seen in mania or psychosis. We describe 3 cases in which we used a stepwise approach to optimize treatment and improve outcomes for patients with excited catatonia. This approach may work equally well for other catatonia subtypes.
Related Resources
- Dubovsky SL, Dubovsky AN. Catatonia: how to identify and treat it. Current Psychiatry. 2018;17(8):16-26.
- Crouse EL, Joel B. Moran JB. Catatonia: recognition, management, and prevention of complications. Current Psychiatry. 2018;17(12):45-49.
Drug Brand Names
Clozapine • Clozaril
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Risperidone • Risperdal
Divalproex sodium • Depakote
1. Fink M, Taylor MA. The many varieties of catatonia. Eur Arch Psychiatry Clin Neurosci. 2001;251(Suppl 1):8-13.
2. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Association; 2013:119-121.
3. Rasmussen SA, Mazurek MF, Rosebush PI. Catatonia: our current understanding of its diagnosis, treatment and pathophysiology. World J Psychiatry. 2016;6(4):391-398.
4. Fink M, Taylor MA. Catatonia: A Clinician’s Guide to Diagnosis and Treatment. Cambridge University Press; 2003.
5. Adityanjee, Aderibigbe YA, Matthews T. Epidemiology of neuroleptic malignant syndrome. Clin Neuropharmacol. 1999;22(3):151-158.
6. Strawn JR, Keck PE Jr, Caroff SN. Neuroleptic malignant syndrome. Am J Psychiatry. 2007;164(6):870-876.
Catatonia is often difficult to identify and treat. The excited catatonia subtype can be particularly challenging to diagnose because it can present with symptoms similar to those seen in mania or psychosis. In this article, we present 3 cases of excited catatonia that illustrate how to identify it, how to treat the catatonia as well as the underlying pathology, and factors to consider during this process to mitigate the risk of adverse outcomes. We also outline a treatment algorithm we used for the 3 cases. Although we describe using this approach for patients with excited catatonia, it is generalizable to other types of catatonia.
Many causes, varying presentations
Catatonia is a psychomotor syndrome characterized by mutism, negativism, stereotypy, waxy flexibility, and other symptoms.1 It is defined by the presence of ≥3 of the 12 symptoms listed in the Table.2 Causes of catatonia include metabolic abnormalities, endocrine disorders, drug intoxication, neurodevelopmental disorders, medication adverse effects, psychosis, and mood disorders.1,3

A subtype of this syndrome, excited catatonia, can present with restlessness, agitation, emotional lability, poor sleep, and altered mental status in addition to the more typical symptoms.1,4 Because excited catatonia can resemble mania or psychosis, it is particularly challenging to identify the underlying disorder causing it and appropriate treatment. Fink et al4 discussed how clinicians have interpreted the different presentations of excited catatonia to gain insight into the underlying diagnosis. If the patient’s thought process appears disorganized, psychosis may be suspected.4 If the patient is delusional and grandiose, they may be manic, and when altered mental status dominates the presentation, delirium may be the culprit.4
Regardless of the underlying cause, the first step is to treat the catatonia. Benzodiazepines and electroconvulsive therapy (ECT) are the most well validated treatments for catatonia and have been used to treat excited catatonia.1 Excited catatonia is often misdiagnosed and subsequently mistreated. In the following 3 cases, excited catatonia was successfully identified and treated using the same approach (Figure).

Case 1
Mr. A, age 27, has a history of bipolar I disorder. He was brought to the hospital by ambulance after being found to be yelling and acting belligerently, and he was admitted to the inpatient psychiatry unit for manic decompensation due to medication nonadherence. He was started on divalproex sodium 500 mg twice a day for mood stabilization, risperidone 1 mg twice a day for adjunct mood stabilization and psychosis, and lorazepam 1 mg 3 times a day for agitation. Mr. A exhibited odd behavior; he would take off his clothes in the hallway, run around the unit, and randomly yell at staff or to himself. At other times, he would stay silent, repeat the same statements, or oddly posture in the hallway for minutes at a time. These behaviors were seen primarily in the hour or 2 preceding lorazepam administration and improved after he received lorazepam.
Mr. A’s treating team completed the Bush-Francis Catatonia Rating Scale (BFCRS), which yielded a positive catatonia screen of 7/14. As a result, divalproex sodium and risperidone were held, and lorazepam was increased to 2 mg twice a day.
After several days, Mr. A was no longer acting oddly and was able to speak more spontaneously; however, he began to exhibit overt signs of mania. He would speak rapidly and make grandiose claims about managing millions of dollars as the CEO of a famous company. Divalproex sodium was restarted at 500 mg twice a day and increased to 500 mg 3 times a day for mood stabilization. Mr. A continued to receive lorazepam 2 mg 3 times a day for catatonia, and risperidone was restarted at 1 mg twice a day to more effectively target his manic symptoms. Risperidone was increased to 2 mg twice a day. After this change, Mr. A’s grandiosity dissipated, his speech normalized, and his thought process became organized. He was discharged on lorazepam 2 mg 3 times a day, divalproex sodium 500 mg 3 times a day, and risperidone 2 mg twice a day. Mr. A’s length of stay (LOS) for this admission was 11 days.
Continue to: Case 2
Case 2
Mr. B, age 49, presented with irritability and odd posturing. He has a history of schizoaffective disorder, bipolar type for which he was receiving a maintenance regimen of lithium 600 mg/d at bedtime and risperidone 2 mg/d at bedtime. He had multiple previous psychiatric admissions for catatonia. On this admission, Mr. B was irritable and difficult to redirect. He yelled at staff members and had a stiff gait. The BFCRS yielded a positive screening score of 3/14 and a severity score of 8/23. As a result, the treatment team conducted a lorazepam challenge.
After Mr. B received lorazepam 1 mg IM, his thought organization and irritability improved, which allowed him to have a coherent conversation with the interviewer. His gait stiffness also improved. His risperidone and lithium were held, and oral lorazepam 1 mg 3 times a day was started for catatonia. Lorazepam was gradually increased to 4 mg 3 times a day. Mr. B became euthymic and redirectable, and had an improved gait. However, he was also tangential and hyperverbal; these symptoms were indicative of the underlying mania that precipitated his catatonia.
Divalproex sodium extended release (ER) was started and increased to 1,500 mg/d at bedtime for mood stabilization. Lithium was restarted and increased to 300 mg twice a day for adjunct mood stabilization. Risperidone was not restarted. Toward the end of his admission, Mr. B was noted to be overly sedated, so the lorazepam dosage was decreased. He was discharged on lorazepam 2 mg 3 times a day, divalproex sodium ER 1,500 mg/d at bedtime, and lithium 300 mg twice a day. At discharge, Mr. B was calm and euthymic, with a linear thought process. His LOS was 25 days.
Case 3
Mr. C, age 62, presented to the emergency department (ED) because he had exhibited erratic behavior and had not slept for the past week. He has a history of bipolar I disorder, hypothyroidism, diabetes, and hypertension. For many years, he had been stable on divalproex sodium ER 2,500 mg/d at bedtime for mood stabilization and clozapine 100 mg/d at bedtime for adjunct mood stabilization and psychosis. In the ED, Mr. C was irritable, distractible, and tangential. On admission, he was speaking slowly with increased speech latency in response to questions, exhibiting stereotypy, repeating statements over and over, and walking very slowly.
The BFCRS yielded a positive screening score of 5/14 and a severity score of 10/23. Lorazepam 1 mg IM was administered. After 15 minutes, Mr. C’s speech, gait, and distractibility improved. As a result, clozapine and divalproex sodium were held, and he was started on oral lorazepam 1 mg 3 times a day. After several days, Mr. C was speaking fluently and no longer exhibiting stereotypy or having outbursts where he would make repetitive statements. However, he was tangential and irritable at times, which were signs of his underlying mania. Divalproex sodium ER was restarted at 250 mg/d at bedtime for mood stabilization and gradually increased to 2,500 mg/d at bedtime. Clozapine was also restarted at 25 mg/d at bedtime and gradually increased to 200 mg/d at bedtime. The lorazepam was gradually tapered and discontinued over the course of 3 weeks due to oversedation.
Continue to: At discharge...
At discharge, Mr. C was euthymic, calm, linear, and goal-directed. He was discharged on divalproex sodium ER 2,500 mg/d at bedtime and clozapine 200 mg/d at bedtime. His LOS for this admission was 22 days.
A stepwise approach can improve outcomes
The Figure outlines the method we used to manage excited catatonia in these 3 cases. Each of these patients exhibited signs of excited catatonia, but because those symptoms were nearly identical to those of mania, it was initially difficult to identify catatonia. Excited catatonia was suspected after more typical catatonic symptoms—such as a stiff gait, slowed speech, and stereotypy—were observed. The BFCRS was completed to get an objective measure of the likelihood that the patient was catatonic. In all 3 cases, the BFCRS resulted in a positive screen for catatonia. Following this, the patients described in Case 2 and Case 3 received a lorazepam challenge, which confirmed their catatonia. No lorazepam challenge was performed in Case 1 because the patient was already receiving lorazepam when the BFCRS was completed. Although most catatonic patients will respond to a lorazepam challenge, not all will. Therefore, clinicians should maintain some degree of suspicion for catatonia if a patient has a positive screen on the BFCRS but a negative lorazepam challenge.
In all 3 cases, after catatonia was confirmed, the patient’s psychotropic medications were discontinued. In all 3 cases, the antipsychotic was held to prevent progression to neuroleptic malignant syndrome (NMS) or malignant catatonia. Rasmussen et al3 found that 3.6% of the catatonic patients in their sample who were treated with antipsychotics developed NMS. A review of prospective studies looking at patients treated with antipsychotics found the incidence of NMS was .07% to 1.8%.5 Because NMS is often clinically indistinguishable from malignant catatonia,4,6 this incidence of NMS may have represented an increased incidence in malignant catatonia.
In all 3 cases, the mood stabilizer was held to prevent it from complicating the clinical picture. Discontinuing the mood stabilizer and focusing on treating the catatonia before targeting the underlying mania increased the likelihood of differentiating the patient’s catatonic symptoms from manic symptoms. This resulted in more precise medication selection and titration by allowing us to identify the specific symptoms that were being targeted by each medication.
Oral lorazepam was prescribed to target catatonia in all 3 cases, and the dosage was gradually increased until symptoms began to resolve. As the catatonia resolved, the manic symptoms became more easily identifiable, and at this point a mood stabilizer was started and titrated to a therapeutic dose to target the mania. In Case 1 and Case 3, the antipsychotic was restarted to treat the mania more effectively. It was not restarted in Case 2 because the patient’s mania was effectively being managed by 2 mood stabilizers. The risks and benefits of starting an antipsychotic in a catatonic or recently catatonic patient should be carefully considered. In the 2 cases where the antipsychotic was restarted, the patients were closely monitored, and there were no signs of NMS or malignant catatonia.
Continue to: As discharge approached...
As discharge approached, the dosages of oral lorazepam were reevaluated. Catatonic patients can typically tolerate high doses of benzodiazepines without becoming overly sedated, but each patient has a different threshold at which the dosage causes oversedation. In all 3 patients, lorazepam was initially titrated to a dose that treated their catatonic symptoms without causing intolerable sedation. In Case 2 and Case 3, as the catatonia began to resolve, the patients became increasingly sedated on their existing lorazepam dosage, so it was decreased. Because the patient in Case 1 did not become overly sedated, his lorazepam dosage did not need to be reduced.
For 2 of these patients, our approach resulted in a shorter LOS compared to their previous hospitalizations. The LOS in Case 2 was 25 days; 5 years earlier, he had a 49-day LOS for mania and catatonia. During the past admission, the identification and treatment of the catatonia was delayed, which resulted in the patient requiring multiple transfers to the medical unit for unstable vital signs. The LOS in Case 3 was 22 days; 6 months prior to this admission, the patient had 2 psychiatric admissions that totaled 37 days. Although the patient’s presentation in the 2 previous admissions was similar to his presentation as described in Case 3, catatonia had not been identified or treated in either admission. Since his catatonia and mania were treated in Case 3, he has not required a readmission. The patient in Case 1 was previously hospitalized, but information about the LOS of these admissions was not available. These results suggest that early identification and treatment of catatonia via the approach we used can improve patient outcomes.
Bottom Line
Excited catatonia can be challenging to diagnose and treat because it can present with symptoms similar to those seen in mania or psychosis. We describe 3 cases in which we used a stepwise approach to optimize treatment and improve outcomes for patients with excited catatonia. This approach may work equally well for other catatonia subtypes.
Related Resources
- Dubovsky SL, Dubovsky AN. Catatonia: how to identify and treat it. Current Psychiatry. 2018;17(8):16-26.
- Crouse EL, Joel B. Moran JB. Catatonia: recognition, management, and prevention of complications. Current Psychiatry. 2018;17(12):45-49.
Drug Brand Names
Clozapine • Clozaril
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Risperidone • Risperdal
Divalproex sodium • Depakote
Catatonia is often difficult to identify and treat. The excited catatonia subtype can be particularly challenging to diagnose because it can present with symptoms similar to those seen in mania or psychosis. In this article, we present 3 cases of excited catatonia that illustrate how to identify it, how to treat the catatonia as well as the underlying pathology, and factors to consider during this process to mitigate the risk of adverse outcomes. We also outline a treatment algorithm we used for the 3 cases. Although we describe using this approach for patients with excited catatonia, it is generalizable to other types of catatonia.
Many causes, varying presentations
Catatonia is a psychomotor syndrome characterized by mutism, negativism, stereotypy, waxy flexibility, and other symptoms.1 It is defined by the presence of ≥3 of the 12 symptoms listed in the Table.2 Causes of catatonia include metabolic abnormalities, endocrine disorders, drug intoxication, neurodevelopmental disorders, medication adverse effects, psychosis, and mood disorders.1,3

A subtype of this syndrome, excited catatonia, can present with restlessness, agitation, emotional lability, poor sleep, and altered mental status in addition to the more typical symptoms.1,4 Because excited catatonia can resemble mania or psychosis, it is particularly challenging to identify the underlying disorder causing it and appropriate treatment. Fink et al4 discussed how clinicians have interpreted the different presentations of excited catatonia to gain insight into the underlying diagnosis. If the patient’s thought process appears disorganized, psychosis may be suspected.4 If the patient is delusional and grandiose, they may be manic, and when altered mental status dominates the presentation, delirium may be the culprit.4
Regardless of the underlying cause, the first step is to treat the catatonia. Benzodiazepines and electroconvulsive therapy (ECT) are the most well validated treatments for catatonia and have been used to treat excited catatonia.1 Excited catatonia is often misdiagnosed and subsequently mistreated. In the following 3 cases, excited catatonia was successfully identified and treated using the same approach (Figure).

Case 1
Mr. A, age 27, has a history of bipolar I disorder. He was brought to the hospital by ambulance after being found to be yelling and acting belligerently, and he was admitted to the inpatient psychiatry unit for manic decompensation due to medication nonadherence. He was started on divalproex sodium 500 mg twice a day for mood stabilization, risperidone 1 mg twice a day for adjunct mood stabilization and psychosis, and lorazepam 1 mg 3 times a day for agitation. Mr. A exhibited odd behavior; he would take off his clothes in the hallway, run around the unit, and randomly yell at staff or to himself. At other times, he would stay silent, repeat the same statements, or oddly posture in the hallway for minutes at a time. These behaviors were seen primarily in the hour or 2 preceding lorazepam administration and improved after he received lorazepam.
Mr. A’s treating team completed the Bush-Francis Catatonia Rating Scale (BFCRS), which yielded a positive catatonia screen of 7/14. As a result, divalproex sodium and risperidone were held, and lorazepam was increased to 2 mg twice a day.
After several days, Mr. A was no longer acting oddly and was able to speak more spontaneously; however, he began to exhibit overt signs of mania. He would speak rapidly and make grandiose claims about managing millions of dollars as the CEO of a famous company. Divalproex sodium was restarted at 500 mg twice a day and increased to 500 mg 3 times a day for mood stabilization. Mr. A continued to receive lorazepam 2 mg 3 times a day for catatonia, and risperidone was restarted at 1 mg twice a day to more effectively target his manic symptoms. Risperidone was increased to 2 mg twice a day. After this change, Mr. A’s grandiosity dissipated, his speech normalized, and his thought process became organized. He was discharged on lorazepam 2 mg 3 times a day, divalproex sodium 500 mg 3 times a day, and risperidone 2 mg twice a day. Mr. A’s length of stay (LOS) for this admission was 11 days.
Continue to: Case 2
Case 2
Mr. B, age 49, presented with irritability and odd posturing. He has a history of schizoaffective disorder, bipolar type for which he was receiving a maintenance regimen of lithium 600 mg/d at bedtime and risperidone 2 mg/d at bedtime. He had multiple previous psychiatric admissions for catatonia. On this admission, Mr. B was irritable and difficult to redirect. He yelled at staff members and had a stiff gait. The BFCRS yielded a positive screening score of 3/14 and a severity score of 8/23. As a result, the treatment team conducted a lorazepam challenge.
After Mr. B received lorazepam 1 mg IM, his thought organization and irritability improved, which allowed him to have a coherent conversation with the interviewer. His gait stiffness also improved. His risperidone and lithium were held, and oral lorazepam 1 mg 3 times a day was started for catatonia. Lorazepam was gradually increased to 4 mg 3 times a day. Mr. B became euthymic and redirectable, and had an improved gait. However, he was also tangential and hyperverbal; these symptoms were indicative of the underlying mania that precipitated his catatonia.
Divalproex sodium extended release (ER) was started and increased to 1,500 mg/d at bedtime for mood stabilization. Lithium was restarted and increased to 300 mg twice a day for adjunct mood stabilization. Risperidone was not restarted. Toward the end of his admission, Mr. B was noted to be overly sedated, so the lorazepam dosage was decreased. He was discharged on lorazepam 2 mg 3 times a day, divalproex sodium ER 1,500 mg/d at bedtime, and lithium 300 mg twice a day. At discharge, Mr. B was calm and euthymic, with a linear thought process. His LOS was 25 days.
Case 3
Mr. C, age 62, presented to the emergency department (ED) because he had exhibited erratic behavior and had not slept for the past week. He has a history of bipolar I disorder, hypothyroidism, diabetes, and hypertension. For many years, he had been stable on divalproex sodium ER 2,500 mg/d at bedtime for mood stabilization and clozapine 100 mg/d at bedtime for adjunct mood stabilization and psychosis. In the ED, Mr. C was irritable, distractible, and tangential. On admission, he was speaking slowly with increased speech latency in response to questions, exhibiting stereotypy, repeating statements over and over, and walking very slowly.
The BFCRS yielded a positive screening score of 5/14 and a severity score of 10/23. Lorazepam 1 mg IM was administered. After 15 minutes, Mr. C’s speech, gait, and distractibility improved. As a result, clozapine and divalproex sodium were held, and he was started on oral lorazepam 1 mg 3 times a day. After several days, Mr. C was speaking fluently and no longer exhibiting stereotypy or having outbursts where he would make repetitive statements. However, he was tangential and irritable at times, which were signs of his underlying mania. Divalproex sodium ER was restarted at 250 mg/d at bedtime for mood stabilization and gradually increased to 2,500 mg/d at bedtime. Clozapine was also restarted at 25 mg/d at bedtime and gradually increased to 200 mg/d at bedtime. The lorazepam was gradually tapered and discontinued over the course of 3 weeks due to oversedation.
Continue to: At discharge...
At discharge, Mr. C was euthymic, calm, linear, and goal-directed. He was discharged on divalproex sodium ER 2,500 mg/d at bedtime and clozapine 200 mg/d at bedtime. His LOS for this admission was 22 days.
A stepwise approach can improve outcomes
The Figure outlines the method we used to manage excited catatonia in these 3 cases. Each of these patients exhibited signs of excited catatonia, but because those symptoms were nearly identical to those of mania, it was initially difficult to identify catatonia. Excited catatonia was suspected after more typical catatonic symptoms—such as a stiff gait, slowed speech, and stereotypy—were observed. The BFCRS was completed to get an objective measure of the likelihood that the patient was catatonic. In all 3 cases, the BFCRS resulted in a positive screen for catatonia. Following this, the patients described in Case 2 and Case 3 received a lorazepam challenge, which confirmed their catatonia. No lorazepam challenge was performed in Case 1 because the patient was already receiving lorazepam when the BFCRS was completed. Although most catatonic patients will respond to a lorazepam challenge, not all will. Therefore, clinicians should maintain some degree of suspicion for catatonia if a patient has a positive screen on the BFCRS but a negative lorazepam challenge.
In all 3 cases, after catatonia was confirmed, the patient’s psychotropic medications were discontinued. In all 3 cases, the antipsychotic was held to prevent progression to neuroleptic malignant syndrome (NMS) or malignant catatonia. Rasmussen et al3 found that 3.6% of the catatonic patients in their sample who were treated with antipsychotics developed NMS. A review of prospective studies looking at patients treated with antipsychotics found the incidence of NMS was .07% to 1.8%.5 Because NMS is often clinically indistinguishable from malignant catatonia,4,6 this incidence of NMS may have represented an increased incidence in malignant catatonia.
In all 3 cases, the mood stabilizer was held to prevent it from complicating the clinical picture. Discontinuing the mood stabilizer and focusing on treating the catatonia before targeting the underlying mania increased the likelihood of differentiating the patient’s catatonic symptoms from manic symptoms. This resulted in more precise medication selection and titration by allowing us to identify the specific symptoms that were being targeted by each medication.
Oral lorazepam was prescribed to target catatonia in all 3 cases, and the dosage was gradually increased until symptoms began to resolve. As the catatonia resolved, the manic symptoms became more easily identifiable, and at this point a mood stabilizer was started and titrated to a therapeutic dose to target the mania. In Case 1 and Case 3, the antipsychotic was restarted to treat the mania more effectively. It was not restarted in Case 2 because the patient’s mania was effectively being managed by 2 mood stabilizers. The risks and benefits of starting an antipsychotic in a catatonic or recently catatonic patient should be carefully considered. In the 2 cases where the antipsychotic was restarted, the patients were closely monitored, and there were no signs of NMS or malignant catatonia.
Continue to: As discharge approached...
As discharge approached, the dosages of oral lorazepam were reevaluated. Catatonic patients can typically tolerate high doses of benzodiazepines without becoming overly sedated, but each patient has a different threshold at which the dosage causes oversedation. In all 3 patients, lorazepam was initially titrated to a dose that treated their catatonic symptoms without causing intolerable sedation. In Case 2 and Case 3, as the catatonia began to resolve, the patients became increasingly sedated on their existing lorazepam dosage, so it was decreased. Because the patient in Case 1 did not become overly sedated, his lorazepam dosage did not need to be reduced.
For 2 of these patients, our approach resulted in a shorter LOS compared to their previous hospitalizations. The LOS in Case 2 was 25 days; 5 years earlier, he had a 49-day LOS for mania and catatonia. During the past admission, the identification and treatment of the catatonia was delayed, which resulted in the patient requiring multiple transfers to the medical unit for unstable vital signs. The LOS in Case 3 was 22 days; 6 months prior to this admission, the patient had 2 psychiatric admissions that totaled 37 days. Although the patient’s presentation in the 2 previous admissions was similar to his presentation as described in Case 3, catatonia had not been identified or treated in either admission. Since his catatonia and mania were treated in Case 3, he has not required a readmission. The patient in Case 1 was previously hospitalized, but information about the LOS of these admissions was not available. These results suggest that early identification and treatment of catatonia via the approach we used can improve patient outcomes.
Bottom Line
Excited catatonia can be challenging to diagnose and treat because it can present with symptoms similar to those seen in mania or psychosis. We describe 3 cases in which we used a stepwise approach to optimize treatment and improve outcomes for patients with excited catatonia. This approach may work equally well for other catatonia subtypes.
Related Resources
- Dubovsky SL, Dubovsky AN. Catatonia: how to identify and treat it. Current Psychiatry. 2018;17(8):16-26.
- Crouse EL, Joel B. Moran JB. Catatonia: recognition, management, and prevention of complications. Current Psychiatry. 2018;17(12):45-49.
Drug Brand Names
Clozapine • Clozaril
Lithium • Eskalith, Lithobid
Lorazepam • Ativan
Risperidone • Risperdal
Divalproex sodium • Depakote
1. Fink M, Taylor MA. The many varieties of catatonia. Eur Arch Psychiatry Clin Neurosci. 2001;251(Suppl 1):8-13.
2. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Association; 2013:119-121.
3. Rasmussen SA, Mazurek MF, Rosebush PI. Catatonia: our current understanding of its diagnosis, treatment and pathophysiology. World J Psychiatry. 2016;6(4):391-398.
4. Fink M, Taylor MA. Catatonia: A Clinician’s Guide to Diagnosis and Treatment. Cambridge University Press; 2003.
5. Adityanjee, Aderibigbe YA, Matthews T. Epidemiology of neuroleptic malignant syndrome. Clin Neuropharmacol. 1999;22(3):151-158.
6. Strawn JR, Keck PE Jr, Caroff SN. Neuroleptic malignant syndrome. Am J Psychiatry. 2007;164(6):870-876.
1. Fink M, Taylor MA. The many varieties of catatonia. Eur Arch Psychiatry Clin Neurosci. 2001;251(Suppl 1):8-13.
2. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Association; 2013:119-121.
3. Rasmussen SA, Mazurek MF, Rosebush PI. Catatonia: our current understanding of its diagnosis, treatment and pathophysiology. World J Psychiatry. 2016;6(4):391-398.
4. Fink M, Taylor MA. Catatonia: A Clinician’s Guide to Diagnosis and Treatment. Cambridge University Press; 2003.
5. Adityanjee, Aderibigbe YA, Matthews T. Epidemiology of neuroleptic malignant syndrome. Clin Neuropharmacol. 1999;22(3):151-158.
6. Strawn JR, Keck PE Jr, Caroff SN. Neuroleptic malignant syndrome. Am J Psychiatry. 2007;164(6):870-876.
Behavioral treatment tied to lower medical, pharmacy costs
Results of a large retrospective study showed that patients newly diagnosed with a BHC who receive OPBHT following diagnosis incur lower medical and pharmacy costs over roughly the next 1 to 2 years, compared with peers who don’t receive OPBHT.
“Our findings suggest that promoting OPBHT as part of a population health strategy is associated with improved overall medical spending, particularly among adults,” the investigators write.
The study was published online in JAMA Network Open.
Common, undertreated
Nearly a quarter of adults in the United States have a BHC, and they incur greater medical costs than those without a BHC. However, diagnosis of a BHC is often delayed, and most affected individuals receive little to no treatment.
In their cost analysis, Johanna Bellon, PhD, and colleagues with Evernorth Health, St. Louis, analyzed commercial insurance claims data for 203,401 U.S. individuals newly diagnosed with one or more BHCs between 2017 and 2018.
About half of participants had depression and/or anxiety, 11% had substance use or alcohol use disorder, and 6% had a higher-acuity diagnosis, such as bipolar disorder, severe depression, eating disorder, psychotic disorder, or autism spectrum disorder.
About 1 in 5 (22%) had at least one chronic medical condition along with their BHC.
The researchers found that having at least one OPBHT visit was associated with lower medical and pharmacy costs during 15- and 27-month follow-up periods.
Over 15 months, the adjusted mean per member per month (PMPM) medical/pharmacy cost was $686 with no OPBHT visit, compared with $571 with one or more OPBHT visits.
Over 27 months, the adjusted mean PMPM was $464 with no OPBHT, versus $391 with one or more OPBHT visits.
Dose-response effect
In addition, there was a “dose-response” relationship between OPBHT and medical/pharmacy costs, such that estimated cost savings were significantly lower in the treated versus the untreated groups at almost every level of treatment.
“Our findings were also largely age independent, especially over 15 months, suggesting that OPBHT has favorable effects among children, young adults, and adults,” the researchers report.
“This is promising given that disease etiology and progression, treatment paradigms, presence of comorbid medical conditions, and overall medical and pharmacy costs differ among the three groups,” they say.
Notably, the dataset largely encompassed in-person OPBHT, because the study period preceded the transition into virtual care that occurred in 2020.
However, overall use of OPBHT was low – older adults, adults with lower income, individuals with comorbid medical conditions, and persons of racial and ethnic minorities were less likely to receive OPBHT, they found.
“These findings support the cost-effectiveness of practitioner- and insurance-based interventions to increase OPBHT utilization, which is a critical resource as new BHC diagnoses continue to increase,” the researchers say.
“Future research should validate these findings in other populations, including government-insured individuals, and explore data by chronic disease category, over longer time horizons, by type and quality of OPBHT, by type of medical spending, within subpopulations with BHCs, and including virtual and digital behavioral health services,” they suggest.
The study had no specific funding. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results of a large retrospective study showed that patients newly diagnosed with a BHC who receive OPBHT following diagnosis incur lower medical and pharmacy costs over roughly the next 1 to 2 years, compared with peers who don’t receive OPBHT.
“Our findings suggest that promoting OPBHT as part of a population health strategy is associated with improved overall medical spending, particularly among adults,” the investigators write.
The study was published online in JAMA Network Open.
Common, undertreated
Nearly a quarter of adults in the United States have a BHC, and they incur greater medical costs than those without a BHC. However, diagnosis of a BHC is often delayed, and most affected individuals receive little to no treatment.
In their cost analysis, Johanna Bellon, PhD, and colleagues with Evernorth Health, St. Louis, analyzed commercial insurance claims data for 203,401 U.S. individuals newly diagnosed with one or more BHCs between 2017 and 2018.
About half of participants had depression and/or anxiety, 11% had substance use or alcohol use disorder, and 6% had a higher-acuity diagnosis, such as bipolar disorder, severe depression, eating disorder, psychotic disorder, or autism spectrum disorder.
About 1 in 5 (22%) had at least one chronic medical condition along with their BHC.
The researchers found that having at least one OPBHT visit was associated with lower medical and pharmacy costs during 15- and 27-month follow-up periods.
Over 15 months, the adjusted mean per member per month (PMPM) medical/pharmacy cost was $686 with no OPBHT visit, compared with $571 with one or more OPBHT visits.
Over 27 months, the adjusted mean PMPM was $464 with no OPBHT, versus $391 with one or more OPBHT visits.
Dose-response effect
In addition, there was a “dose-response” relationship between OPBHT and medical/pharmacy costs, such that estimated cost savings were significantly lower in the treated versus the untreated groups at almost every level of treatment.
“Our findings were also largely age independent, especially over 15 months, suggesting that OPBHT has favorable effects among children, young adults, and adults,” the researchers report.
“This is promising given that disease etiology and progression, treatment paradigms, presence of comorbid medical conditions, and overall medical and pharmacy costs differ among the three groups,” they say.
Notably, the dataset largely encompassed in-person OPBHT, because the study period preceded the transition into virtual care that occurred in 2020.
However, overall use of OPBHT was low – older adults, adults with lower income, individuals with comorbid medical conditions, and persons of racial and ethnic minorities were less likely to receive OPBHT, they found.
“These findings support the cost-effectiveness of practitioner- and insurance-based interventions to increase OPBHT utilization, which is a critical resource as new BHC diagnoses continue to increase,” the researchers say.
“Future research should validate these findings in other populations, including government-insured individuals, and explore data by chronic disease category, over longer time horizons, by type and quality of OPBHT, by type of medical spending, within subpopulations with BHCs, and including virtual and digital behavioral health services,” they suggest.
The study had no specific funding. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results of a large retrospective study showed that patients newly diagnosed with a BHC who receive OPBHT following diagnosis incur lower medical and pharmacy costs over roughly the next 1 to 2 years, compared with peers who don’t receive OPBHT.
“Our findings suggest that promoting OPBHT as part of a population health strategy is associated with improved overall medical spending, particularly among adults,” the investigators write.
The study was published online in JAMA Network Open.
Common, undertreated
Nearly a quarter of adults in the United States have a BHC, and they incur greater medical costs than those without a BHC. However, diagnosis of a BHC is often delayed, and most affected individuals receive little to no treatment.
In their cost analysis, Johanna Bellon, PhD, and colleagues with Evernorth Health, St. Louis, analyzed commercial insurance claims data for 203,401 U.S. individuals newly diagnosed with one or more BHCs between 2017 and 2018.
About half of participants had depression and/or anxiety, 11% had substance use or alcohol use disorder, and 6% had a higher-acuity diagnosis, such as bipolar disorder, severe depression, eating disorder, psychotic disorder, or autism spectrum disorder.
About 1 in 5 (22%) had at least one chronic medical condition along with their BHC.
The researchers found that having at least one OPBHT visit was associated with lower medical and pharmacy costs during 15- and 27-month follow-up periods.
Over 15 months, the adjusted mean per member per month (PMPM) medical/pharmacy cost was $686 with no OPBHT visit, compared with $571 with one or more OPBHT visits.
Over 27 months, the adjusted mean PMPM was $464 with no OPBHT, versus $391 with one or more OPBHT visits.
Dose-response effect
In addition, there was a “dose-response” relationship between OPBHT and medical/pharmacy costs, such that estimated cost savings were significantly lower in the treated versus the untreated groups at almost every level of treatment.
“Our findings were also largely age independent, especially over 15 months, suggesting that OPBHT has favorable effects among children, young adults, and adults,” the researchers report.
“This is promising given that disease etiology and progression, treatment paradigms, presence of comorbid medical conditions, and overall medical and pharmacy costs differ among the three groups,” they say.
Notably, the dataset largely encompassed in-person OPBHT, because the study period preceded the transition into virtual care that occurred in 2020.
However, overall use of OPBHT was low – older adults, adults with lower income, individuals with comorbid medical conditions, and persons of racial and ethnic minorities were less likely to receive OPBHT, they found.
“These findings support the cost-effectiveness of practitioner- and insurance-based interventions to increase OPBHT utilization, which is a critical resource as new BHC diagnoses continue to increase,” the researchers say.
“Future research should validate these findings in other populations, including government-insured individuals, and explore data by chronic disease category, over longer time horizons, by type and quality of OPBHT, by type of medical spending, within subpopulations with BHCs, and including virtual and digital behavioral health services,” they suggest.
The study had no specific funding. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Does dopamine dysregulation cause schizophrenia?
Investigators identified a mechanism on the dopamine receptor, known as the autoreceptor, which regulates how much dopamine is released from the presynaptic neuron. Impairment of this autoreceptor leads to poorly controlled dopamine release and excessive dopamine flow.
The researchers found decreased expression of this autoreceptor accounts for the genetic evidence of schizophrenia risk, and, using a suite of statistical routines, they showed that this relationship is probably causative.
“Our research confirms the scientific hypothesis that too much dopamine plays a likely causative role in psychosis and precisely how this is based on genetic factors,” study investigator Daniel Weinberger, MD, director and CEO of the Lieber Institute for Brain Development, Baltimore, told this news organization.
“Drugs that treat psychosis symptoms by simply blocking dopamine receptors have harsh side effects. ... Theoretically, scientists could now develop therapies that target these malfunctioning autoreceptors to treat this devastating condition with fewer side effects,” he said.
The study was published online in Nature Neuroscience.
‘Privileged spot’
“Large international genetic studies known as genomewide association studies have identified hundreds of regions of the human genome housing potential risk genes for schizophrenia,” Dr. Weinberger said.
“However, these regions are still poorly resolved in terms of specific genes, and treatments and diagnostic techniques are far from what they should be.” Moreover, “treatments for schizophrenia address the symptoms of psychosis but not the cause,” he said.
“For more than 70 years, neuroscientists have suspected that dopamine plays a key role in schizophrenia, but what kind of role, exactly, has remained a mystery,” Dr. Weinberger noted. “It occupied a privileged spot in the principal hypothesis about schizophrenia for over 60 years – the so-called ‘dopamine hypothesis.’ ”
Antipsychotic drugs that reduce dopamine “are the principal medical treatments but they cause serious side effects, including an inability to experience pleasure and joy – a sad reality for patients and their families,” he continued.
The current study “set out to understand how dopamine acts in schizophrenia” using “analysis of the genetic and transcriptional landscape” of the postmortem caudate nucleus from 443 donors (245 neurotypical, 154 with schizophrenia, and 44 with bipolar disorder).
Brain samples were from individuals of diverse ancestry (210 were of African ancestry and 2,233 were of European ancestry).
New treatment target?
The researchers performed an analysis of transancestry expression quantitative trait loci, genetic variants that explain variations in gene expression levels, which express in the caudate, annotating “hundreds of caudate-specific cis-eQTLs.”
Then they integrated this analysis with gene expression that emerged from the latest genomewide association study and transcriptome-wide association study, identifying hundreds of genes that “showed a potential causal association with schizophrenia risk in the caudate nucleus,” including a specific isoform of the dopamine D2 receptor, which is upregulated in the caudate nucleus of those with schizophrenia.
“If autoreceptors don’t function properly the flow of dopamine in the brain is poorly controlled and too much dopamine flows for too long,” said Dr. Weinberger.
In particular, they observed “extensive differential gene expression” for schizophrenia in 2,701 genes in those with schizophrenia, compared with those without: glial cell–derived neurotrophic factor antisense RNA was a top-up gene and tyrosine hydroxylase, which is a rate-limiting enzyme in dopamine synthesis, was a down-regulated gene. Dopamine receptors DRD2 and DRD3 were differentially expressed.
Having done this, they looked at the effects of antipsychotic medications that target D2 regions on gene expression in the caudate by testing for differences between individuals with schizophrenia who were taking antipsychotics at the time of death, those not taking antipsychotics at the time of death (n = 104 and 49, respectively), and neurotypical individuals (n = 239).
There were 2,692 differentially expressed genes between individuals taking antipsychotics versus neurotypical individuals (false discovery rate < 0.05). By contrast, there were only 665 differentially expressed genes (FDR < .05) between those not taking antipsychotics and neurotypical individuals.
“We found that antipsychotic medication has an extensive influence on caudate gene expression,” the investigators noted.
They then developed a new approach to “infer gene networks from expression data.” This method is based on deep neural networks, obtaining a “low-dimensional representation of each gene’s expression across individuals.” The representation is then used to build a “gene neighborhood graph and assign genes to modules.”
This method identified “several modules enriched for genes associated with schizophrenia risk.” The expression representations captured in this approach placed genes in “biologically meaningful neighborhoods, which can provide insight into potential interactions if these genes are targeted for therapeutic intervention,” the authors summarized.
“Now that our new research has identified the specific mechanism by which dopamine plays a causative role in schizophrenia, we hope we have opened the door for more targeted drugs or diagnostic tests that could make life better for patients and their families,” Dr. Weinberger said.
No causal link?
Commenting on the study, Rifaat El-Mallakh, MD, director of the mood disorders research program, department of psychiatry and behavioral sciences, University of Louisville (Ky.), called it an “excellent study performed by an excellent research group” that “fills an important lacuna in our research database.”
However, Dr. El-Mallakh, who was not involved in the research, disagreed that the findings show causality. “The data that can be gleaned from this study is limited and the design has significant limitations. As with all genetic studies, this is an association study. It tells us nothing about the cause-effect relationship between the genes and the illness.
“We do not know why genes are associated with the illness. Genetic overrepresentation can have multiple causes, and more so when the data is a convenience sample. As noted by the authors, much of what they observed was probably related to medication effect. I don’t think this study specifically tells us anything clinically,” he added.
The study was supported by the LIBD, the BrainSeq Consortium, an National Institutes of Health fellowship to two of the authors, and a NARSAD Young Investigator Grant from the Brain & Behavior Research Foundation to one of the authors. Dr. Weinberger has reported no relevant financial relationships. Dr. El-Mallakh declared no specific financial relationships relevant to the study but has reported being a speaker for several companies that manufacture antipsychotics.
A version of this article first appeared on Medscape.com.
Investigators identified a mechanism on the dopamine receptor, known as the autoreceptor, which regulates how much dopamine is released from the presynaptic neuron. Impairment of this autoreceptor leads to poorly controlled dopamine release and excessive dopamine flow.
The researchers found decreased expression of this autoreceptor accounts for the genetic evidence of schizophrenia risk, and, using a suite of statistical routines, they showed that this relationship is probably causative.
“Our research confirms the scientific hypothesis that too much dopamine plays a likely causative role in psychosis and precisely how this is based on genetic factors,” study investigator Daniel Weinberger, MD, director and CEO of the Lieber Institute for Brain Development, Baltimore, told this news organization.
“Drugs that treat psychosis symptoms by simply blocking dopamine receptors have harsh side effects. ... Theoretically, scientists could now develop therapies that target these malfunctioning autoreceptors to treat this devastating condition with fewer side effects,” he said.
The study was published online in Nature Neuroscience.
‘Privileged spot’
“Large international genetic studies known as genomewide association studies have identified hundreds of regions of the human genome housing potential risk genes for schizophrenia,” Dr. Weinberger said.
“However, these regions are still poorly resolved in terms of specific genes, and treatments and diagnostic techniques are far from what they should be.” Moreover, “treatments for schizophrenia address the symptoms of psychosis but not the cause,” he said.
“For more than 70 years, neuroscientists have suspected that dopamine plays a key role in schizophrenia, but what kind of role, exactly, has remained a mystery,” Dr. Weinberger noted. “It occupied a privileged spot in the principal hypothesis about schizophrenia for over 60 years – the so-called ‘dopamine hypothesis.’ ”
Antipsychotic drugs that reduce dopamine “are the principal medical treatments but they cause serious side effects, including an inability to experience pleasure and joy – a sad reality for patients and their families,” he continued.
The current study “set out to understand how dopamine acts in schizophrenia” using “analysis of the genetic and transcriptional landscape” of the postmortem caudate nucleus from 443 donors (245 neurotypical, 154 with schizophrenia, and 44 with bipolar disorder).
Brain samples were from individuals of diverse ancestry (210 were of African ancestry and 2,233 were of European ancestry).
New treatment target?
The researchers performed an analysis of transancestry expression quantitative trait loci, genetic variants that explain variations in gene expression levels, which express in the caudate, annotating “hundreds of caudate-specific cis-eQTLs.”
Then they integrated this analysis with gene expression that emerged from the latest genomewide association study and transcriptome-wide association study, identifying hundreds of genes that “showed a potential causal association with schizophrenia risk in the caudate nucleus,” including a specific isoform of the dopamine D2 receptor, which is upregulated in the caudate nucleus of those with schizophrenia.
“If autoreceptors don’t function properly the flow of dopamine in the brain is poorly controlled and too much dopamine flows for too long,” said Dr. Weinberger.
In particular, they observed “extensive differential gene expression” for schizophrenia in 2,701 genes in those with schizophrenia, compared with those without: glial cell–derived neurotrophic factor antisense RNA was a top-up gene and tyrosine hydroxylase, which is a rate-limiting enzyme in dopamine synthesis, was a down-regulated gene. Dopamine receptors DRD2 and DRD3 were differentially expressed.
Having done this, they looked at the effects of antipsychotic medications that target D2 regions on gene expression in the caudate by testing for differences between individuals with schizophrenia who were taking antipsychotics at the time of death, those not taking antipsychotics at the time of death (n = 104 and 49, respectively), and neurotypical individuals (n = 239).
There were 2,692 differentially expressed genes between individuals taking antipsychotics versus neurotypical individuals (false discovery rate < 0.05). By contrast, there were only 665 differentially expressed genes (FDR < .05) between those not taking antipsychotics and neurotypical individuals.
“We found that antipsychotic medication has an extensive influence on caudate gene expression,” the investigators noted.
They then developed a new approach to “infer gene networks from expression data.” This method is based on deep neural networks, obtaining a “low-dimensional representation of each gene’s expression across individuals.” The representation is then used to build a “gene neighborhood graph and assign genes to modules.”
This method identified “several modules enriched for genes associated with schizophrenia risk.” The expression representations captured in this approach placed genes in “biologically meaningful neighborhoods, which can provide insight into potential interactions if these genes are targeted for therapeutic intervention,” the authors summarized.
“Now that our new research has identified the specific mechanism by which dopamine plays a causative role in schizophrenia, we hope we have opened the door for more targeted drugs or diagnostic tests that could make life better for patients and their families,” Dr. Weinberger said.
No causal link?
Commenting on the study, Rifaat El-Mallakh, MD, director of the mood disorders research program, department of psychiatry and behavioral sciences, University of Louisville (Ky.), called it an “excellent study performed by an excellent research group” that “fills an important lacuna in our research database.”
However, Dr. El-Mallakh, who was not involved in the research, disagreed that the findings show causality. “The data that can be gleaned from this study is limited and the design has significant limitations. As with all genetic studies, this is an association study. It tells us nothing about the cause-effect relationship between the genes and the illness.
“We do not know why genes are associated with the illness. Genetic overrepresentation can have multiple causes, and more so when the data is a convenience sample. As noted by the authors, much of what they observed was probably related to medication effect. I don’t think this study specifically tells us anything clinically,” he added.
The study was supported by the LIBD, the BrainSeq Consortium, an National Institutes of Health fellowship to two of the authors, and a NARSAD Young Investigator Grant from the Brain & Behavior Research Foundation to one of the authors. Dr. Weinberger has reported no relevant financial relationships. Dr. El-Mallakh declared no specific financial relationships relevant to the study but has reported being a speaker for several companies that manufacture antipsychotics.
A version of this article first appeared on Medscape.com.
Investigators identified a mechanism on the dopamine receptor, known as the autoreceptor, which regulates how much dopamine is released from the presynaptic neuron. Impairment of this autoreceptor leads to poorly controlled dopamine release and excessive dopamine flow.
The researchers found decreased expression of this autoreceptor accounts for the genetic evidence of schizophrenia risk, and, using a suite of statistical routines, they showed that this relationship is probably causative.
“Our research confirms the scientific hypothesis that too much dopamine plays a likely causative role in psychosis and precisely how this is based on genetic factors,” study investigator Daniel Weinberger, MD, director and CEO of the Lieber Institute for Brain Development, Baltimore, told this news organization.
“Drugs that treat psychosis symptoms by simply blocking dopamine receptors have harsh side effects. ... Theoretically, scientists could now develop therapies that target these malfunctioning autoreceptors to treat this devastating condition with fewer side effects,” he said.
The study was published online in Nature Neuroscience.
‘Privileged spot’
“Large international genetic studies known as genomewide association studies have identified hundreds of regions of the human genome housing potential risk genes for schizophrenia,” Dr. Weinberger said.
“However, these regions are still poorly resolved in terms of specific genes, and treatments and diagnostic techniques are far from what they should be.” Moreover, “treatments for schizophrenia address the symptoms of psychosis but not the cause,” he said.
“For more than 70 years, neuroscientists have suspected that dopamine plays a key role in schizophrenia, but what kind of role, exactly, has remained a mystery,” Dr. Weinberger noted. “It occupied a privileged spot in the principal hypothesis about schizophrenia for over 60 years – the so-called ‘dopamine hypothesis.’ ”
Antipsychotic drugs that reduce dopamine “are the principal medical treatments but they cause serious side effects, including an inability to experience pleasure and joy – a sad reality for patients and their families,” he continued.
The current study “set out to understand how dopamine acts in schizophrenia” using “analysis of the genetic and transcriptional landscape” of the postmortem caudate nucleus from 443 donors (245 neurotypical, 154 with schizophrenia, and 44 with bipolar disorder).
Brain samples were from individuals of diverse ancestry (210 were of African ancestry and 2,233 were of European ancestry).
New treatment target?
The researchers performed an analysis of transancestry expression quantitative trait loci, genetic variants that explain variations in gene expression levels, which express in the caudate, annotating “hundreds of caudate-specific cis-eQTLs.”
Then they integrated this analysis with gene expression that emerged from the latest genomewide association study and transcriptome-wide association study, identifying hundreds of genes that “showed a potential causal association with schizophrenia risk in the caudate nucleus,” including a specific isoform of the dopamine D2 receptor, which is upregulated in the caudate nucleus of those with schizophrenia.
“If autoreceptors don’t function properly the flow of dopamine in the brain is poorly controlled and too much dopamine flows for too long,” said Dr. Weinberger.
In particular, they observed “extensive differential gene expression” for schizophrenia in 2,701 genes in those with schizophrenia, compared with those without: glial cell–derived neurotrophic factor antisense RNA was a top-up gene and tyrosine hydroxylase, which is a rate-limiting enzyme in dopamine synthesis, was a down-regulated gene. Dopamine receptors DRD2 and DRD3 were differentially expressed.
Having done this, they looked at the effects of antipsychotic medications that target D2 regions on gene expression in the caudate by testing for differences between individuals with schizophrenia who were taking antipsychotics at the time of death, those not taking antipsychotics at the time of death (n = 104 and 49, respectively), and neurotypical individuals (n = 239).
There were 2,692 differentially expressed genes between individuals taking antipsychotics versus neurotypical individuals (false discovery rate < 0.05). By contrast, there were only 665 differentially expressed genes (FDR < .05) between those not taking antipsychotics and neurotypical individuals.
“We found that antipsychotic medication has an extensive influence on caudate gene expression,” the investigators noted.
They then developed a new approach to “infer gene networks from expression data.” This method is based on deep neural networks, obtaining a “low-dimensional representation of each gene’s expression across individuals.” The representation is then used to build a “gene neighborhood graph and assign genes to modules.”
This method identified “several modules enriched for genes associated with schizophrenia risk.” The expression representations captured in this approach placed genes in “biologically meaningful neighborhoods, which can provide insight into potential interactions if these genes are targeted for therapeutic intervention,” the authors summarized.
“Now that our new research has identified the specific mechanism by which dopamine plays a causative role in schizophrenia, we hope we have opened the door for more targeted drugs or diagnostic tests that could make life better for patients and their families,” Dr. Weinberger said.
No causal link?
Commenting on the study, Rifaat El-Mallakh, MD, director of the mood disorders research program, department of psychiatry and behavioral sciences, University of Louisville (Ky.), called it an “excellent study performed by an excellent research group” that “fills an important lacuna in our research database.”
However, Dr. El-Mallakh, who was not involved in the research, disagreed that the findings show causality. “The data that can be gleaned from this study is limited and the design has significant limitations. As with all genetic studies, this is an association study. It tells us nothing about the cause-effect relationship between the genes and the illness.
“We do not know why genes are associated with the illness. Genetic overrepresentation can have multiple causes, and more so when the data is a convenience sample. As noted by the authors, much of what they observed was probably related to medication effect. I don’t think this study specifically tells us anything clinically,” he added.
The study was supported by the LIBD, the BrainSeq Consortium, an National Institutes of Health fellowship to two of the authors, and a NARSAD Young Investigator Grant from the Brain & Behavior Research Foundation to one of the authors. Dr. Weinberger has reported no relevant financial relationships. Dr. El-Mallakh declared no specific financial relationships relevant to the study but has reported being a speaker for several companies that manufacture antipsychotics.
A version of this article first appeared on Medscape.com.
FROM NATURE NEUROSCIENCE
Clinical factors drive hospitalization after self-harm
Clinicians who assess suicidal patients in the emergency department setting face the challenge of whether to admit the patient to inpatient or outpatient care, and data on predictors of compulsory admission are limited, wrote Laurent Michaud, MD, of the University of Lausanne, Switzerland, and colleagues.
To better identify predictors of hospitalization after self-harm, the researchers reviewed data from 1,832 patients aged 18 years and older admitted to four emergency departments in Switzerland between December 2016 and November 2019 .
Self-harm (SH) was defined in this study as “all nonfatal intentional acts of self-poisoning or self-injury, irrespective of degree of suicidal intent or other types of motivation,” the researchers noted. The study included 2,142 episodes of self-harm.
The researchers conducted two analyses. They compared episodes followed by any hospitalization and those with outpatient follow-up (1,083 episodes vs. 1,059 episodes) and episodes followed by compulsory hospitalization (357 episodes) with all other episodes followed by either outpatient care or voluntary hospitalization (1,785 episodes).
Overall, women were significantly more likely to be referred to outpatient follow-up compared with men (61.8% vs. 38.1%), and hospitalized patients were significantly older than outpatients (mean age of 41 years vs. 36 years, P < .001 for both).
“Not surprisingly, major psychopathological conditions such as depression, mania, dementia, and schizophrenia were predictive of hospitalization,” the researchers noted.
Other sociodemographic factors associated with hospitalization included living alone, no children, problematic socioeconomic status, and unemployment. Clinical factors associated with hospitalization included physical pain, more lethal suicide attempt method, and clear intent to die.
In a multivariate analysis, independent predictors of any hospitalization included male gender, older age, assessment in the Neuchatel location vs. Lausanne, depression vs. personality disorders, substance use, or anxiety disorder, difficult socioeconomic status, a clear vs. unclear intent to die, and a serious suicide attempt vs. less serious.
Differences in hospitalization based on hospital setting was a striking finding, the researchers wrote in their discussion. These differences may be largely explained by the organization of local mental health services and specific institutional cultures; the workload of staff and availability of beds also may have played a role in decisions to hospitalize, they said.
The findings were limited by several factors including the lack of data on the realization level of a self-harm episode and significant events such as a breakup, the researchers explained. Other limitations included missing data, multiple analyses that could increase the risk of false positives, the reliance on clinical diagnosis rather than formal instruments, and the cross-sectional study design, they said.
However, the results have clinical implications, as the clinical factors identified could be used to target subgroups of suicidal populations and refine treatment strategies, they concluded.
The study was supported by institutional funding and the Swiss Federal Office of Public Health. The researchers had no financial conflicts to disclose.
Clinicians who assess suicidal patients in the emergency department setting face the challenge of whether to admit the patient to inpatient or outpatient care, and data on predictors of compulsory admission are limited, wrote Laurent Michaud, MD, of the University of Lausanne, Switzerland, and colleagues.
To better identify predictors of hospitalization after self-harm, the researchers reviewed data from 1,832 patients aged 18 years and older admitted to four emergency departments in Switzerland between December 2016 and November 2019 .
Self-harm (SH) was defined in this study as “all nonfatal intentional acts of self-poisoning or self-injury, irrespective of degree of suicidal intent or other types of motivation,” the researchers noted. The study included 2,142 episodes of self-harm.
The researchers conducted two analyses. They compared episodes followed by any hospitalization and those with outpatient follow-up (1,083 episodes vs. 1,059 episodes) and episodes followed by compulsory hospitalization (357 episodes) with all other episodes followed by either outpatient care or voluntary hospitalization (1,785 episodes).
Overall, women were significantly more likely to be referred to outpatient follow-up compared with men (61.8% vs. 38.1%), and hospitalized patients were significantly older than outpatients (mean age of 41 years vs. 36 years, P < .001 for both).
“Not surprisingly, major psychopathological conditions such as depression, mania, dementia, and schizophrenia were predictive of hospitalization,” the researchers noted.
Other sociodemographic factors associated with hospitalization included living alone, no children, problematic socioeconomic status, and unemployment. Clinical factors associated with hospitalization included physical pain, more lethal suicide attempt method, and clear intent to die.
In a multivariate analysis, independent predictors of any hospitalization included male gender, older age, assessment in the Neuchatel location vs. Lausanne, depression vs. personality disorders, substance use, or anxiety disorder, difficult socioeconomic status, a clear vs. unclear intent to die, and a serious suicide attempt vs. less serious.
Differences in hospitalization based on hospital setting was a striking finding, the researchers wrote in their discussion. These differences may be largely explained by the organization of local mental health services and specific institutional cultures; the workload of staff and availability of beds also may have played a role in decisions to hospitalize, they said.
The findings were limited by several factors including the lack of data on the realization level of a self-harm episode and significant events such as a breakup, the researchers explained. Other limitations included missing data, multiple analyses that could increase the risk of false positives, the reliance on clinical diagnosis rather than formal instruments, and the cross-sectional study design, they said.
However, the results have clinical implications, as the clinical factors identified could be used to target subgroups of suicidal populations and refine treatment strategies, they concluded.
The study was supported by institutional funding and the Swiss Federal Office of Public Health. The researchers had no financial conflicts to disclose.
Clinicians who assess suicidal patients in the emergency department setting face the challenge of whether to admit the patient to inpatient or outpatient care, and data on predictors of compulsory admission are limited, wrote Laurent Michaud, MD, of the University of Lausanne, Switzerland, and colleagues.
To better identify predictors of hospitalization after self-harm, the researchers reviewed data from 1,832 patients aged 18 years and older admitted to four emergency departments in Switzerland between December 2016 and November 2019 .
Self-harm (SH) was defined in this study as “all nonfatal intentional acts of self-poisoning or self-injury, irrespective of degree of suicidal intent or other types of motivation,” the researchers noted. The study included 2,142 episodes of self-harm.
The researchers conducted two analyses. They compared episodes followed by any hospitalization and those with outpatient follow-up (1,083 episodes vs. 1,059 episodes) and episodes followed by compulsory hospitalization (357 episodes) with all other episodes followed by either outpatient care or voluntary hospitalization (1,785 episodes).
Overall, women were significantly more likely to be referred to outpatient follow-up compared with men (61.8% vs. 38.1%), and hospitalized patients were significantly older than outpatients (mean age of 41 years vs. 36 years, P < .001 for both).
“Not surprisingly, major psychopathological conditions such as depression, mania, dementia, and schizophrenia were predictive of hospitalization,” the researchers noted.
Other sociodemographic factors associated with hospitalization included living alone, no children, problematic socioeconomic status, and unemployment. Clinical factors associated with hospitalization included physical pain, more lethal suicide attempt method, and clear intent to die.
In a multivariate analysis, independent predictors of any hospitalization included male gender, older age, assessment in the Neuchatel location vs. Lausanne, depression vs. personality disorders, substance use, or anxiety disorder, difficult socioeconomic status, a clear vs. unclear intent to die, and a serious suicide attempt vs. less serious.
Differences in hospitalization based on hospital setting was a striking finding, the researchers wrote in their discussion. These differences may be largely explained by the organization of local mental health services and specific institutional cultures; the workload of staff and availability of beds also may have played a role in decisions to hospitalize, they said.
The findings were limited by several factors including the lack of data on the realization level of a self-harm episode and significant events such as a breakup, the researchers explained. Other limitations included missing data, multiple analyses that could increase the risk of false positives, the reliance on clinical diagnosis rather than formal instruments, and the cross-sectional study design, they said.
However, the results have clinical implications, as the clinical factors identified could be used to target subgroups of suicidal populations and refine treatment strategies, they concluded.
The study was supported by institutional funding and the Swiss Federal Office of Public Health. The researchers had no financial conflicts to disclose.
FROM PSYCHIATRIC RESEARCH
Optimal psychiatric treatment: Target the brain and avoid the body
Pharmacotherapy for psychiatric disorders is a mixed blessing. The advent of psychotropic medications since the 1950s (antipsychotics, antidepressants, anxiolytics, mood stabilizers) has revolutionized the treatment of serious psychiatric brain disorders, allowing certain patients to be discharged to the community after a lifetime of institutionalization.
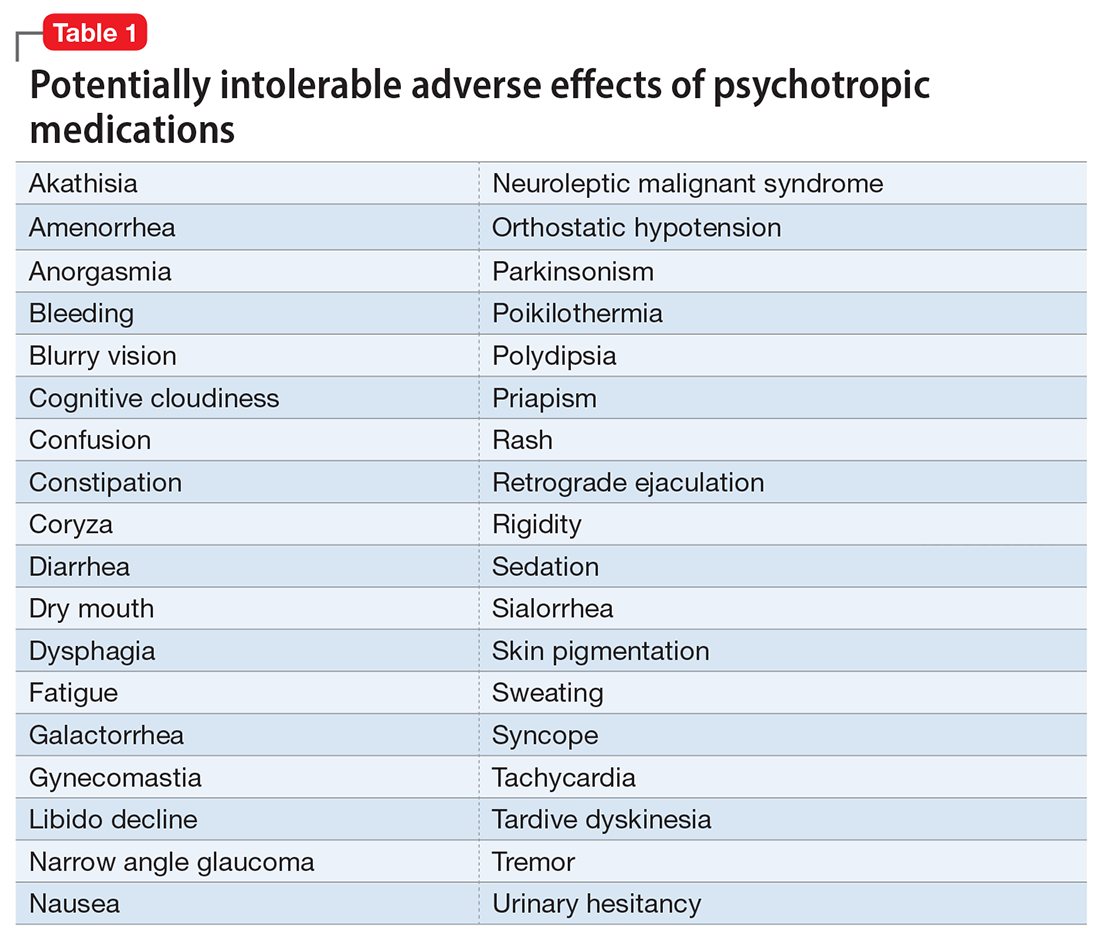
However, like all medications, psychotropic agents are often associated with various potentially intolerable symptoms (Table 1) or safety complications (Table 2) because they interact with every organ in the body besides their intended target, the brain, and its neurochemical circuitry.
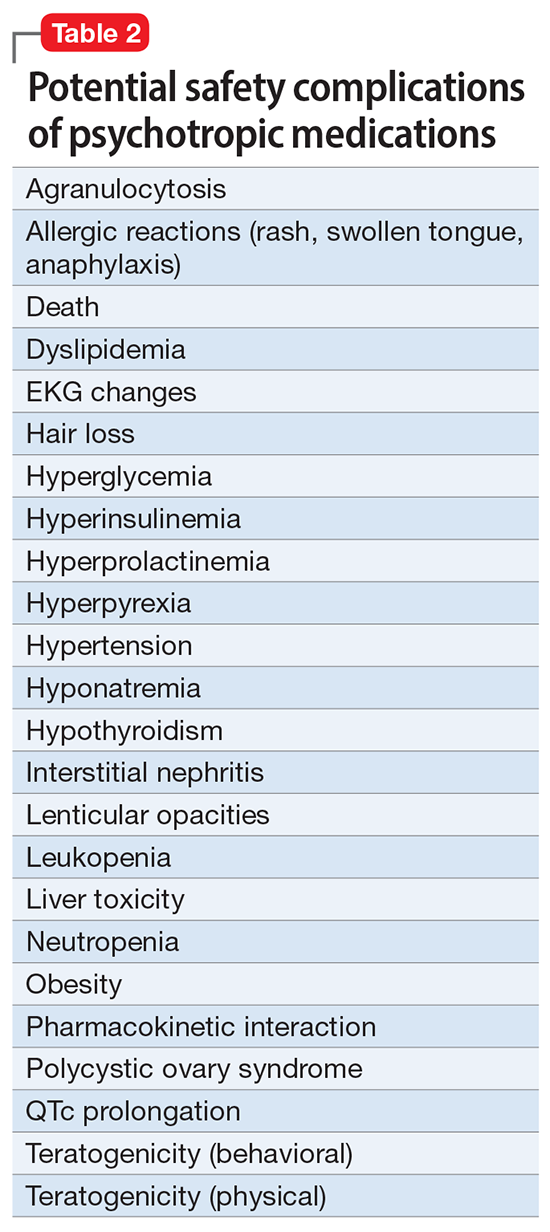
Imagine if we could treat our psychiatric patients while bypassing the body and achieve response, remission, and ultimately recovery without any systemic adverse effects. Adherence would dramatically improve, our patients’ quality of life would be enhanced, and the overall effectiveness (defined as the complex package of efficacy, safety, and tolerability) would be superior to current pharmacotherapies. This is important because most psychiatric medications must be taken daily for years, even a lifetime, to avoid a relapse of the illness. Psychiatrists frequently must manage adverse effects or switch the patient to a different medication if a tolerability or safety issue emerges, which is very common in psychiatric practice. A significant part of psychopharmacologic management includes ordering various laboratory tests to monitor adverse reactions in major organs, especially the liver, kidney, and heart. Additionally, psychiatric physicians must be constantly cognizant of medications prescribed by other clinicians for comorbid medical conditions to successfully navigate the turbulent seas of pharmacokinetic interactions.
I am sure you have noticed that whenever you watch a direct-to-consumer commercial for any medication, 90% of the advertisement is a background voice listing the various tolerability and safety complications of the medication as required by the FDA. Interestingly, these ads frequently contain colorful scenery and joyful clips, which I suspect are cleverly designed to distract the audience from focusing on the list of adverse effects.
Benefits of nonpharmacologic treatments
No wonder I am a fan of psychotherapy, a well-established psychiatric treatment modality that completely avoids body tissues. It directly targets the brain without needlessly interacting with any other organ. Psychotherapy’s many benefits (improving insight, enhancing adherence, improving self-esteem, reducing risky behaviors, guiding stress management and coping skills, modifying unhealthy beliefs, and ultimately relieving symptoms such as anxiety and depression) are achieved without any somatic adverse effects! Psychotherapy has also been shown to induce neuroplasticity and reduce inflammatory biomarkers.1 Unlike FDA-approved medications, psychotherapy does not include a “package insert,” 10 to 20 pages (in small print) that mostly focus on warnings, precautions, and sundry physical adverse effects. Even the dosing of psychotherapy is left entirely up to the treating clinician!
Although I have had many gratifying results with pharmacotherapy in my practice, especially in combination with psychotherapy,2 I also have observed excellent outcomes with nonpharmacologic approaches, especially neuromodulation therapies. The best antidepressant I have ever used since my residency training days is electroconvulsive therapy (ECT). My experience is consistent with a large meta-analysis3showing a huge effect size (Cohen d = .91) in contrast to the usual effect size of .3 to .5 for standard antidepressants (except IV ketamine). A recent study showed ECT is even better than the vaunted rapid-acting ketamine,4 which is further evidence of its remarkable efficacy in depression. Neuroimaging studies report that ECT rapidly increases the volume of the hippocampus,5,6 which shrinks in size in patients with unipolar or bipolar depression.
Neuromodulation may very well be the future of psychiatric therapeutics. It targets the brain and avoids the body, thus achieving efficacy with minimal systemic tolerability (ie, patient complaints) (Table 1) or safety (abnormal laboratory test results) issues (Table 2). This sounds ideal, and it is arguably an optimal approach to repairing the brain and healing the mind.
Continue to: ECT is the oldest...
ECT is the oldest neuromodulation technique (developed almost 100 years ago and significantly refined since then). Newer FDA-approved neuromodulation therapies include repetitive transcranial magnetic stimulation (rTMS), which was approved for depression in 2013, obsessive-compulsive disorder (OCD) in 2018, smoking cessation in 2020, and anxious depression in 2021.7 Vagus nerve stimulation (VNS) is used for drug-resistant epilepsy and was later approved for treatment-resistant depression,8,9 but some studies report it can be helpful for fear and anxiety in autism spectrum disorder10 and primary insomnia.11
There are many other neuromodulation therapies in development12 that have not yet been FDA approved (Table 3). The most prominent of these is deep brain stimulation (DBS), which is approved for Parkinson disease and has been reported in many studies to improve treatment-resistant depression13,14 and OCD.15 Another promising neuromodulation therapy is transcranial direct current stimulation (tDCS), which has promising results in schizophrenia16 similar to ECT’s effects in treatment-resistant schizophrenia.17
A particularly exciting neuromodulation approach published by Stanford University researchers is Stanford accelerated intelligent neuromodulation therapy (SAINT),18 which uses intermittent theta-burst stimulation (iTBS) daily for 5 days, targeted at the subgenual anterior cingulate gyrus (Brodman area 25). Remarkably, efficacy was rapid, with a very high remission rate (absence of symptoms) in approximately 90% of patients with severe depression.18
The future is bright for neuromodulation therapies, and for a good reason. Why send a chemical agent to every cell and organ in the body when the brain can be targeted directly? As psychiatric neuroscience advances to a point where we can localize the abnormal neurologic circuit in a specific brain region for each psychiatric disorder, it will be possible to treat almost all psychiatric disorders without burdening patients with the intolerable symptoms or safety adverse effects of medications. Psychiatrists should modulate their perspective about the future of psychiatric treatments. And finally, I propose that psychotherapy should be reclassified as a “verbal neuromodulation” technique.
1. Nasrallah HA. Repositioning psychotherapy as a neurobiological intervention. Current Psychiatry. 2013;12(12):18-19.
2. Nasrallah HA. Bipolar disorder: clinical questions beg for answers. Current Psychiatry. 2006;5(12):11-12.
3. UK ECT Review Group. Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and meta-analysis. Lancet. 2003;361(9360):799-808.
4. Rhee TG, Shim SR, Forester BP, et al. Efficacy and safety of ketamine vs electroconvulsive therapy among patients with major depressive episode: a systematic review and meta-analysis. JAMA Psychiatry. 2022:e223352. doi:10.1001/jamapsychiatry.2022.3352
5. Nuninga JO, Mandl RCW, Boks MP, et al. Volume increase in the dentate gyrus after electroconvulsive therapy in depressed patients as measured with 7T. Mol Psychiatry. 2020;25(7):1559-1568.
6. Joshi SH, Espinoza RT, Pirnia T, et al. Structural plasticity of the hippocampus and amygdala induced by electroconvulsive therapy in major depression. Biol Psychiatry. 2016;79(4):282-292.
7. Rhee TG, Olfson M, Nierenberg AA, et al. 20-year trends in the pharmacologic treatment of bipolar disorder by psychiatrists in outpatient care settings. Am J Psychiatry. 2020;177(8):706-715.
8. Hilz MJ. Transcutaneous vagus nerve stimulation - a brief introduction and overview. Auton Neurosci. 2022;243:103038. doi:10.1016/j.autneu.2022.103038
9. Pigato G, Rosson S, Bresolin N, et al. Vagus nerve stimulation in treatment-resistant depression: a case series of long-term follow-up. J ECT. 2022. doi:10.1097/YCT.0000000000000869
10. Shivaswamy T, Souza RR, Engineer CT, et al. Vagus nerve stimulation as a treatment for fear and anxiety in individuals with autism spectrum disorder. J Psychiatr Brain Sci. 2022;7(4):e220007. doi:10.20900/jpbs.20220007
11. Wu Y, Song L, Wang X, et al. Transcutaneous vagus nerve stimulation could improve the effective rate on the quality of sleep in the treatment of primary insomnia: a randomized control trial. Brain Sci. 2022;12(10):1296. doi:10.3390/brainsci12101296
12. Rosa MA, Lisanby SH. Somatic treatments for mood disorders. Neuropsychopharmacology. 2012;37(1):102-116.
13. Mayberg HS, Lozano AM, Voon V, et al. Deep brain stimulation for treatment-resistant depression. Neuron. 2005;45(5):651-660.
14. Choi KS, Mayberg H. Connectomic DBS in major depression. In: Horn A, ed. Connectomic Deep Brain Stimulation. Academic Press; 2022:433-447.
15. Cruz S, Gutiérrez-Rojas L, González-Domenech P, et al. Deep brain stimulation in obsessive-compulsive disorder: results from meta-analysis. Psychiatry Res. 2022;317:114869. doi:10.1016/j.psychres.2022.114869
16. Lisoni J, Baldacci G, Nibbio G, et al. Effects of bilateral, bipolar-nonbalanced, frontal transcranial direct current stimulation (tDCS) on negative symptoms and neurocognition in a sample of patients living with schizophrenia: results of a randomized double-blind sham-controlled trial. J Psychiatr Res. 2022;155:430-442.
17. Sinclair DJ, Zhao S, Qi F, et al. Electroconvulsive therapy for treatment-resistant schizophrenia. Cochrane Database Syst Rev. 2019;3(3):CD011847. doi:10.1002/14651858.CD011847.pub2
18. Cole EJ, Stimpson KH, Bentzley BS, et al. Stanford accelerated intelligent neuromodulation therapy for treatment-resistant depression. Am J Psychiatry. 2020;177(8):716-726.
Pharmacotherapy for psychiatric disorders is a mixed blessing. The advent of psychotropic medications since the 1950s (antipsychotics, antidepressants, anxiolytics, mood stabilizers) has revolutionized the treatment of serious psychiatric brain disorders, allowing certain patients to be discharged to the community after a lifetime of institutionalization.

However, like all medications, psychotropic agents are often associated with various potentially intolerable symptoms (Table 1) or safety complications (Table 2) because they interact with every organ in the body besides their intended target, the brain, and its neurochemical circuitry.

Imagine if we could treat our psychiatric patients while bypassing the body and achieve response, remission, and ultimately recovery without any systemic adverse effects. Adherence would dramatically improve, our patients’ quality of life would be enhanced, and the overall effectiveness (defined as the complex package of efficacy, safety, and tolerability) would be superior to current pharmacotherapies. This is important because most psychiatric medications must be taken daily for years, even a lifetime, to avoid a relapse of the illness. Psychiatrists frequently must manage adverse effects or switch the patient to a different medication if a tolerability or safety issue emerges, which is very common in psychiatric practice. A significant part of psychopharmacologic management includes ordering various laboratory tests to monitor adverse reactions in major organs, especially the liver, kidney, and heart. Additionally, psychiatric physicians must be constantly cognizant of medications prescribed by other clinicians for comorbid medical conditions to successfully navigate the turbulent seas of pharmacokinetic interactions.
I am sure you have noticed that whenever you watch a direct-to-consumer commercial for any medication, 90% of the advertisement is a background voice listing the various tolerability and safety complications of the medication as required by the FDA. Interestingly, these ads frequently contain colorful scenery and joyful clips, which I suspect are cleverly designed to distract the audience from focusing on the list of adverse effects.
Benefits of nonpharmacologic treatments
No wonder I am a fan of psychotherapy, a well-established psychiatric treatment modality that completely avoids body tissues. It directly targets the brain without needlessly interacting with any other organ. Psychotherapy’s many benefits (improving insight, enhancing adherence, improving self-esteem, reducing risky behaviors, guiding stress management and coping skills, modifying unhealthy beliefs, and ultimately relieving symptoms such as anxiety and depression) are achieved without any somatic adverse effects! Psychotherapy has also been shown to induce neuroplasticity and reduce inflammatory biomarkers.1 Unlike FDA-approved medications, psychotherapy does not include a “package insert,” 10 to 20 pages (in small print) that mostly focus on warnings, precautions, and sundry physical adverse effects. Even the dosing of psychotherapy is left entirely up to the treating clinician!
Although I have had many gratifying results with pharmacotherapy in my practice, especially in combination with psychotherapy,2 I also have observed excellent outcomes with nonpharmacologic approaches, especially neuromodulation therapies. The best antidepressant I have ever used since my residency training days is electroconvulsive therapy (ECT). My experience is consistent with a large meta-analysis3showing a huge effect size (Cohen d = .91) in contrast to the usual effect size of .3 to .5 for standard antidepressants (except IV ketamine). A recent study showed ECT is even better than the vaunted rapid-acting ketamine,4 which is further evidence of its remarkable efficacy in depression. Neuroimaging studies report that ECT rapidly increases the volume of the hippocampus,5,6 which shrinks in size in patients with unipolar or bipolar depression.
Neuromodulation may very well be the future of psychiatric therapeutics. It targets the brain and avoids the body, thus achieving efficacy with minimal systemic tolerability (ie, patient complaints) (Table 1) or safety (abnormal laboratory test results) issues (Table 2). This sounds ideal, and it is arguably an optimal approach to repairing the brain and healing the mind.
Continue to: ECT is the oldest...
ECT is the oldest neuromodulation technique (developed almost 100 years ago and significantly refined since then). Newer FDA-approved neuromodulation therapies include repetitive transcranial magnetic stimulation (rTMS), which was approved for depression in 2013, obsessive-compulsive disorder (OCD) in 2018, smoking cessation in 2020, and anxious depression in 2021.7 Vagus nerve stimulation (VNS) is used for drug-resistant epilepsy and was later approved for treatment-resistant depression,8,9 but some studies report it can be helpful for fear and anxiety in autism spectrum disorder10 and primary insomnia.11
There are many other neuromodulation therapies in development12 that have not yet been FDA approved (Table 3). The most prominent of these is deep brain stimulation (DBS), which is approved for Parkinson disease and has been reported in many studies to improve treatment-resistant depression13,14 and OCD.15 Another promising neuromodulation therapy is transcranial direct current stimulation (tDCS), which has promising results in schizophrenia16 similar to ECT’s effects in treatment-resistant schizophrenia.17

A particularly exciting neuromodulation approach published by Stanford University researchers is Stanford accelerated intelligent neuromodulation therapy (SAINT),18 which uses intermittent theta-burst stimulation (iTBS) daily for 5 days, targeted at the subgenual anterior cingulate gyrus (Brodman area 25). Remarkably, efficacy was rapid, with a very high remission rate (absence of symptoms) in approximately 90% of patients with severe depression.18
The future is bright for neuromodulation therapies, and for a good reason. Why send a chemical agent to every cell and organ in the body when the brain can be targeted directly? As psychiatric neuroscience advances to a point where we can localize the abnormal neurologic circuit in a specific brain region for each psychiatric disorder, it will be possible to treat almost all psychiatric disorders without burdening patients with the intolerable symptoms or safety adverse effects of medications. Psychiatrists should modulate their perspective about the future of psychiatric treatments. And finally, I propose that psychotherapy should be reclassified as a “verbal neuromodulation” technique.
Pharmacotherapy for psychiatric disorders is a mixed blessing. The advent of psychotropic medications since the 1950s (antipsychotics, antidepressants, anxiolytics, mood stabilizers) has revolutionized the treatment of serious psychiatric brain disorders, allowing certain patients to be discharged to the community after a lifetime of institutionalization.

However, like all medications, psychotropic agents are often associated with various potentially intolerable symptoms (Table 1) or safety complications (Table 2) because they interact with every organ in the body besides their intended target, the brain, and its neurochemical circuitry.

Imagine if we could treat our psychiatric patients while bypassing the body and achieve response, remission, and ultimately recovery without any systemic adverse effects. Adherence would dramatically improve, our patients’ quality of life would be enhanced, and the overall effectiveness (defined as the complex package of efficacy, safety, and tolerability) would be superior to current pharmacotherapies. This is important because most psychiatric medications must be taken daily for years, even a lifetime, to avoid a relapse of the illness. Psychiatrists frequently must manage adverse effects or switch the patient to a different medication if a tolerability or safety issue emerges, which is very common in psychiatric practice. A significant part of psychopharmacologic management includes ordering various laboratory tests to monitor adverse reactions in major organs, especially the liver, kidney, and heart. Additionally, psychiatric physicians must be constantly cognizant of medications prescribed by other clinicians for comorbid medical conditions to successfully navigate the turbulent seas of pharmacokinetic interactions.
I am sure you have noticed that whenever you watch a direct-to-consumer commercial for any medication, 90% of the advertisement is a background voice listing the various tolerability and safety complications of the medication as required by the FDA. Interestingly, these ads frequently contain colorful scenery and joyful clips, which I suspect are cleverly designed to distract the audience from focusing on the list of adverse effects.
Benefits of nonpharmacologic treatments
No wonder I am a fan of psychotherapy, a well-established psychiatric treatment modality that completely avoids body tissues. It directly targets the brain without needlessly interacting with any other organ. Psychotherapy’s many benefits (improving insight, enhancing adherence, improving self-esteem, reducing risky behaviors, guiding stress management and coping skills, modifying unhealthy beliefs, and ultimately relieving symptoms such as anxiety and depression) are achieved without any somatic adverse effects! Psychotherapy has also been shown to induce neuroplasticity and reduce inflammatory biomarkers.1 Unlike FDA-approved medications, psychotherapy does not include a “package insert,” 10 to 20 pages (in small print) that mostly focus on warnings, precautions, and sundry physical adverse effects. Even the dosing of psychotherapy is left entirely up to the treating clinician!
Although I have had many gratifying results with pharmacotherapy in my practice, especially in combination with psychotherapy,2 I also have observed excellent outcomes with nonpharmacologic approaches, especially neuromodulation therapies. The best antidepressant I have ever used since my residency training days is electroconvulsive therapy (ECT). My experience is consistent with a large meta-analysis3showing a huge effect size (Cohen d = .91) in contrast to the usual effect size of .3 to .5 for standard antidepressants (except IV ketamine). A recent study showed ECT is even better than the vaunted rapid-acting ketamine,4 which is further evidence of its remarkable efficacy in depression. Neuroimaging studies report that ECT rapidly increases the volume of the hippocampus,5,6 which shrinks in size in patients with unipolar or bipolar depression.
Neuromodulation may very well be the future of psychiatric therapeutics. It targets the brain and avoids the body, thus achieving efficacy with minimal systemic tolerability (ie, patient complaints) (Table 1) or safety (abnormal laboratory test results) issues (Table 2). This sounds ideal, and it is arguably an optimal approach to repairing the brain and healing the mind.
Continue to: ECT is the oldest...
ECT is the oldest neuromodulation technique (developed almost 100 years ago and significantly refined since then). Newer FDA-approved neuromodulation therapies include repetitive transcranial magnetic stimulation (rTMS), which was approved for depression in 2013, obsessive-compulsive disorder (OCD) in 2018, smoking cessation in 2020, and anxious depression in 2021.7 Vagus nerve stimulation (VNS) is used for drug-resistant epilepsy and was later approved for treatment-resistant depression,8,9 but some studies report it can be helpful for fear and anxiety in autism spectrum disorder10 and primary insomnia.11
There are many other neuromodulation therapies in development12 that have not yet been FDA approved (Table 3). The most prominent of these is deep brain stimulation (DBS), which is approved for Parkinson disease and has been reported in many studies to improve treatment-resistant depression13,14 and OCD.15 Another promising neuromodulation therapy is transcranial direct current stimulation (tDCS), which has promising results in schizophrenia16 similar to ECT’s effects in treatment-resistant schizophrenia.17

A particularly exciting neuromodulation approach published by Stanford University researchers is Stanford accelerated intelligent neuromodulation therapy (SAINT),18 which uses intermittent theta-burst stimulation (iTBS) daily for 5 days, targeted at the subgenual anterior cingulate gyrus (Brodman area 25). Remarkably, efficacy was rapid, with a very high remission rate (absence of symptoms) in approximately 90% of patients with severe depression.18
The future is bright for neuromodulation therapies, and for a good reason. Why send a chemical agent to every cell and organ in the body when the brain can be targeted directly? As psychiatric neuroscience advances to a point where we can localize the abnormal neurologic circuit in a specific brain region for each psychiatric disorder, it will be possible to treat almost all psychiatric disorders without burdening patients with the intolerable symptoms or safety adverse effects of medications. Psychiatrists should modulate their perspective about the future of psychiatric treatments. And finally, I propose that psychotherapy should be reclassified as a “verbal neuromodulation” technique.
1. Nasrallah HA. Repositioning psychotherapy as a neurobiological intervention. Current Psychiatry. 2013;12(12):18-19.
2. Nasrallah HA. Bipolar disorder: clinical questions beg for answers. Current Psychiatry. 2006;5(12):11-12.
3. UK ECT Review Group. Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and meta-analysis. Lancet. 2003;361(9360):799-808.
4. Rhee TG, Shim SR, Forester BP, et al. Efficacy and safety of ketamine vs electroconvulsive therapy among patients with major depressive episode: a systematic review and meta-analysis. JAMA Psychiatry. 2022:e223352. doi:10.1001/jamapsychiatry.2022.3352
5. Nuninga JO, Mandl RCW, Boks MP, et al. Volume increase in the dentate gyrus after electroconvulsive therapy in depressed patients as measured with 7T. Mol Psychiatry. 2020;25(7):1559-1568.
6. Joshi SH, Espinoza RT, Pirnia T, et al. Structural plasticity of the hippocampus and amygdala induced by electroconvulsive therapy in major depression. Biol Psychiatry. 2016;79(4):282-292.
7. Rhee TG, Olfson M, Nierenberg AA, et al. 20-year trends in the pharmacologic treatment of bipolar disorder by psychiatrists in outpatient care settings. Am J Psychiatry. 2020;177(8):706-715.
8. Hilz MJ. Transcutaneous vagus nerve stimulation - a brief introduction and overview. Auton Neurosci. 2022;243:103038. doi:10.1016/j.autneu.2022.103038
9. Pigato G, Rosson S, Bresolin N, et al. Vagus nerve stimulation in treatment-resistant depression: a case series of long-term follow-up. J ECT. 2022. doi:10.1097/YCT.0000000000000869
10. Shivaswamy T, Souza RR, Engineer CT, et al. Vagus nerve stimulation as a treatment for fear and anxiety in individuals with autism spectrum disorder. J Psychiatr Brain Sci. 2022;7(4):e220007. doi:10.20900/jpbs.20220007
11. Wu Y, Song L, Wang X, et al. Transcutaneous vagus nerve stimulation could improve the effective rate on the quality of sleep in the treatment of primary insomnia: a randomized control trial. Brain Sci. 2022;12(10):1296. doi:10.3390/brainsci12101296
12. Rosa MA, Lisanby SH. Somatic treatments for mood disorders. Neuropsychopharmacology. 2012;37(1):102-116.
13. Mayberg HS, Lozano AM, Voon V, et al. Deep brain stimulation for treatment-resistant depression. Neuron. 2005;45(5):651-660.
14. Choi KS, Mayberg H. Connectomic DBS in major depression. In: Horn A, ed. Connectomic Deep Brain Stimulation. Academic Press; 2022:433-447.
15. Cruz S, Gutiérrez-Rojas L, González-Domenech P, et al. Deep brain stimulation in obsessive-compulsive disorder: results from meta-analysis. Psychiatry Res. 2022;317:114869. doi:10.1016/j.psychres.2022.114869
16. Lisoni J, Baldacci G, Nibbio G, et al. Effects of bilateral, bipolar-nonbalanced, frontal transcranial direct current stimulation (tDCS) on negative symptoms and neurocognition in a sample of patients living with schizophrenia: results of a randomized double-blind sham-controlled trial. J Psychiatr Res. 2022;155:430-442.
17. Sinclair DJ, Zhao S, Qi F, et al. Electroconvulsive therapy for treatment-resistant schizophrenia. Cochrane Database Syst Rev. 2019;3(3):CD011847. doi:10.1002/14651858.CD011847.pub2
18. Cole EJ, Stimpson KH, Bentzley BS, et al. Stanford accelerated intelligent neuromodulation therapy for treatment-resistant depression. Am J Psychiatry. 2020;177(8):716-726.
1. Nasrallah HA. Repositioning psychotherapy as a neurobiological intervention. Current Psychiatry. 2013;12(12):18-19.
2. Nasrallah HA. Bipolar disorder: clinical questions beg for answers. Current Psychiatry. 2006;5(12):11-12.
3. UK ECT Review Group. Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and meta-analysis. Lancet. 2003;361(9360):799-808.
4. Rhee TG, Shim SR, Forester BP, et al. Efficacy and safety of ketamine vs electroconvulsive therapy among patients with major depressive episode: a systematic review and meta-analysis. JAMA Psychiatry. 2022:e223352. doi:10.1001/jamapsychiatry.2022.3352
5. Nuninga JO, Mandl RCW, Boks MP, et al. Volume increase in the dentate gyrus after electroconvulsive therapy in depressed patients as measured with 7T. Mol Psychiatry. 2020;25(7):1559-1568.
6. Joshi SH, Espinoza RT, Pirnia T, et al. Structural plasticity of the hippocampus and amygdala induced by electroconvulsive therapy in major depression. Biol Psychiatry. 2016;79(4):282-292.
7. Rhee TG, Olfson M, Nierenberg AA, et al. 20-year trends in the pharmacologic treatment of bipolar disorder by psychiatrists in outpatient care settings. Am J Psychiatry. 2020;177(8):706-715.
8. Hilz MJ. Transcutaneous vagus nerve stimulation - a brief introduction and overview. Auton Neurosci. 2022;243:103038. doi:10.1016/j.autneu.2022.103038
9. Pigato G, Rosson S, Bresolin N, et al. Vagus nerve stimulation in treatment-resistant depression: a case series of long-term follow-up. J ECT. 2022. doi:10.1097/YCT.0000000000000869
10. Shivaswamy T, Souza RR, Engineer CT, et al. Vagus nerve stimulation as a treatment for fear and anxiety in individuals with autism spectrum disorder. J Psychiatr Brain Sci. 2022;7(4):e220007. doi:10.20900/jpbs.20220007
11. Wu Y, Song L, Wang X, et al. Transcutaneous vagus nerve stimulation could improve the effective rate on the quality of sleep in the treatment of primary insomnia: a randomized control trial. Brain Sci. 2022;12(10):1296. doi:10.3390/brainsci12101296
12. Rosa MA, Lisanby SH. Somatic treatments for mood disorders. Neuropsychopharmacology. 2012;37(1):102-116.
13. Mayberg HS, Lozano AM, Voon V, et al. Deep brain stimulation for treatment-resistant depression. Neuron. 2005;45(5):651-660.
14. Choi KS, Mayberg H. Connectomic DBS in major depression. In: Horn A, ed. Connectomic Deep Brain Stimulation. Academic Press; 2022:433-447.
15. Cruz S, Gutiérrez-Rojas L, González-Domenech P, et al. Deep brain stimulation in obsessive-compulsive disorder: results from meta-analysis. Psychiatry Res. 2022;317:114869. doi:10.1016/j.psychres.2022.114869
16. Lisoni J, Baldacci G, Nibbio G, et al. Effects of bilateral, bipolar-nonbalanced, frontal transcranial direct current stimulation (tDCS) on negative symptoms and neurocognition in a sample of patients living with schizophrenia: results of a randomized double-blind sham-controlled trial. J Psychiatr Res. 2022;155:430-442.
17. Sinclair DJ, Zhao S, Qi F, et al. Electroconvulsive therapy for treatment-resistant schizophrenia. Cochrane Database Syst Rev. 2019;3(3):CD011847. doi:10.1002/14651858.CD011847.pub2
18. Cole EJ, Stimpson KH, Bentzley BS, et al. Stanford accelerated intelligent neuromodulation therapy for treatment-resistant depression. Am J Psychiatry. 2020;177(8):716-726.
Breast cancer screening in women receiving antipsychotics
Women with severe mental illness (SMI) are more likely to develop breast cancer and often have more advanced stages of breast cancer when it is detected.1 Antipsychotics have a wide variety of FDA-approved indications and many important life-saving properties. However, patients treated with antipsychotic medications that increase prolactin levels require special consideration with regards to referral for breast cancer screening. Although no clear causal link between antipsychotic use and breast cancer has been established, antipsychotics that raise serum prolactin levels (haloperidol, iloperidone, lurasidone, olanzapine, paliperidone, risperidone) are associated with a higher risk of breast cancer than antipsychotics that produce smaller increases in prolactin levels (aripiprazole, asenapine, brexpiprazole, cariprazine, clozapine, quetiapine, and ziprasidone).2,3 Risperidone and paliperidone have the highest propensities to increase prolactin (45 to >100 ng/mL), whereas other second-generation antipsychotics are associated with only modest elevations.4 Prolonged exposure to high serum prolactin levels should be avoided in women due to the increased risk for breast cancer.2,3 Although there are no clear rules regarding which number or cluster of personal risk factors necessitates a further risk assessment for breast cancer, women receiving antipsychotics (especially those age ≥40) can be referred for further assessment. An individualized, patient-centered approach should be used.
Recognize risk factors
Patients with SMI often need to take a regimen of medications, including antipsychotics, for weeks or months to stabilize their symptoms. Once a woman with SMI is stabilized, consider referral to a clinic that can comprehensively assess for breast cancer risk. Nonmodifiable risk factors include older age, certain genetic mutations (BRCA1 and BRCA2), early menarche, late menopause, high breast tissue density as detected by mammography, a family history of breast cancer, and exposure to radiation.5,6 Modifiable risk factors include physical inactivity, being overweight or obese, hormonal exposure, drinking alcohol, and the presence of certain factors in the patient’s reproductive history (first pregnancy after age 30, not breastfeeding, and never having a full-term pregnancy).2,3 When making such referrals, it is important to avoid making the patient feel alarmed or frightened of antipsychotics. Instead, explain that a referral for breast cancer screening is routine.
When to refer
All women age ≥40 should be offered a referral to a clinic that can provide screening mammography. If a woman has pain, detects a lump in her breast, has a bloody discharge from the nipple, or has changes in the shape or texture of the nipple or breast, a more urgent referral should be made.4 The most important thing to remember is that early breast lesion detection can be life-saving and can avert the need for more invasive surgeries as well as exposure to chemotherapy and radiation.
What to do when prolactin is elevated
Ongoing monitoring of serum prolactin levels can help ensure that the patient’s levels remain in a normal range (<25 ng/mL).2,3,5,6 If hyperprolactinemia is detected, consider switching to an antipsychotic less likely to increase prolactin. Alternatively, the addition of aripiprazole/brexpiprazole or a dopamine agonist as combination therapy can be considered to rapidly restore normal prolactin levels.2 Such changes should be carefully considered because patients may decompensate if antipsychotics are abruptly switched. An individualized risk vs benefit analysis is necessary for any patient in this situation. Risks include not only the recurrence of psychiatric symptoms but also a potential loss of their current level of functioning. Patients may need to continue to take an antipsychotic that is more likely to increase prolactin, in which case close monitoring is advised as well as collaboration with other physicians and members of the patient’s care team. Involving the patient’s support system is helpful.
1. Weinstein LC, Stefancic A, Cunningham AT, et al. Cancer screening, prevention, and treatment in people with mental illness. CA Cancer J Clin. 2016;66(2):134-151.
2. Rahman T, Sahrmann JM, Olsen MA, et al. Risk of breast cancer with prolactin elevating antipsychotic drugs: an observational study of US women (ages 18–64 years). J Clin Psychopharmacol. 2022;42(1):7-16.
3. Rahman T, Clevenger CV, Kaklamani V, et al. Antipsychotic treatment in breast cancer patients. Am J Psychiatry. 2014;171(6):616-621.
4. Peuskens J, Pani L, Detraux J, et al. The effects of novel and newly approved antipsychotics on serum prolactin levels: a comprehensive review. CNS Drugs. 2014;28(5):421-453.
5. Centers for Disease Control and Prevention, Division of Cancer Prevention and Control. Breast cancer. Accessed June 1, 2022. https://www.cdc.gov/cancer/breast/index.htm
6. Steiner E, Klubert D, Knutson D. Assessing breast cancer risk in women. Am Fam Physician. 2008;78(12):1361-1366.
Women with severe mental illness (SMI) are more likely to develop breast cancer and often have more advanced stages of breast cancer when it is detected.1 Antipsychotics have a wide variety of FDA-approved indications and many important life-saving properties. However, patients treated with antipsychotic medications that increase prolactin levels require special consideration with regards to referral for breast cancer screening. Although no clear causal link between antipsychotic use and breast cancer has been established, antipsychotics that raise serum prolactin levels (haloperidol, iloperidone, lurasidone, olanzapine, paliperidone, risperidone) are associated with a higher risk of breast cancer than antipsychotics that produce smaller increases in prolactin levels (aripiprazole, asenapine, brexpiprazole, cariprazine, clozapine, quetiapine, and ziprasidone).2,3 Risperidone and paliperidone have the highest propensities to increase prolactin (45 to >100 ng/mL), whereas other second-generation antipsychotics are associated with only modest elevations.4 Prolonged exposure to high serum prolactin levels should be avoided in women due to the increased risk for breast cancer.2,3 Although there are no clear rules regarding which number or cluster of personal risk factors necessitates a further risk assessment for breast cancer, women receiving antipsychotics (especially those age ≥40) can be referred for further assessment. An individualized, patient-centered approach should be used.
Recognize risk factors
Patients with SMI often need to take a regimen of medications, including antipsychotics, for weeks or months to stabilize their symptoms. Once a woman with SMI is stabilized, consider referral to a clinic that can comprehensively assess for breast cancer risk. Nonmodifiable risk factors include older age, certain genetic mutations (BRCA1 and BRCA2), early menarche, late menopause, high breast tissue density as detected by mammography, a family history of breast cancer, and exposure to radiation.5,6 Modifiable risk factors include physical inactivity, being overweight or obese, hormonal exposure, drinking alcohol, and the presence of certain factors in the patient’s reproductive history (first pregnancy after age 30, not breastfeeding, and never having a full-term pregnancy).2,3 When making such referrals, it is important to avoid making the patient feel alarmed or frightened of antipsychotics. Instead, explain that a referral for breast cancer screening is routine.
When to refer
All women age ≥40 should be offered a referral to a clinic that can provide screening mammography. If a woman has pain, detects a lump in her breast, has a bloody discharge from the nipple, or has changes in the shape or texture of the nipple or breast, a more urgent referral should be made.4 The most important thing to remember is that early breast lesion detection can be life-saving and can avert the need for more invasive surgeries as well as exposure to chemotherapy and radiation.
What to do when prolactin is elevated
Ongoing monitoring of serum prolactin levels can help ensure that the patient’s levels remain in a normal range (<25 ng/mL).2,3,5,6 If hyperprolactinemia is detected, consider switching to an antipsychotic less likely to increase prolactin. Alternatively, the addition of aripiprazole/brexpiprazole or a dopamine agonist as combination therapy can be considered to rapidly restore normal prolactin levels.2 Such changes should be carefully considered because patients may decompensate if antipsychotics are abruptly switched. An individualized risk vs benefit analysis is necessary for any patient in this situation. Risks include not only the recurrence of psychiatric symptoms but also a potential loss of their current level of functioning. Patients may need to continue to take an antipsychotic that is more likely to increase prolactin, in which case close monitoring is advised as well as collaboration with other physicians and members of the patient’s care team. Involving the patient’s support system is helpful.
Women with severe mental illness (SMI) are more likely to develop breast cancer and often have more advanced stages of breast cancer when it is detected.1 Antipsychotics have a wide variety of FDA-approved indications and many important life-saving properties. However, patients treated with antipsychotic medications that increase prolactin levels require special consideration with regards to referral for breast cancer screening. Although no clear causal link between antipsychotic use and breast cancer has been established, antipsychotics that raise serum prolactin levels (haloperidol, iloperidone, lurasidone, olanzapine, paliperidone, risperidone) are associated with a higher risk of breast cancer than antipsychotics that produce smaller increases in prolactin levels (aripiprazole, asenapine, brexpiprazole, cariprazine, clozapine, quetiapine, and ziprasidone).2,3 Risperidone and paliperidone have the highest propensities to increase prolactin (45 to >100 ng/mL), whereas other second-generation antipsychotics are associated with only modest elevations.4 Prolonged exposure to high serum prolactin levels should be avoided in women due to the increased risk for breast cancer.2,3 Although there are no clear rules regarding which number or cluster of personal risk factors necessitates a further risk assessment for breast cancer, women receiving antipsychotics (especially those age ≥40) can be referred for further assessment. An individualized, patient-centered approach should be used.
Recognize risk factors
Patients with SMI often need to take a regimen of medications, including antipsychotics, for weeks or months to stabilize their symptoms. Once a woman with SMI is stabilized, consider referral to a clinic that can comprehensively assess for breast cancer risk. Nonmodifiable risk factors include older age, certain genetic mutations (BRCA1 and BRCA2), early menarche, late menopause, high breast tissue density as detected by mammography, a family history of breast cancer, and exposure to radiation.5,6 Modifiable risk factors include physical inactivity, being overweight or obese, hormonal exposure, drinking alcohol, and the presence of certain factors in the patient’s reproductive history (first pregnancy after age 30, not breastfeeding, and never having a full-term pregnancy).2,3 When making such referrals, it is important to avoid making the patient feel alarmed or frightened of antipsychotics. Instead, explain that a referral for breast cancer screening is routine.
When to refer
All women age ≥40 should be offered a referral to a clinic that can provide screening mammography. If a woman has pain, detects a lump in her breast, has a bloody discharge from the nipple, or has changes in the shape or texture of the nipple or breast, a more urgent referral should be made.4 The most important thing to remember is that early breast lesion detection can be life-saving and can avert the need for more invasive surgeries as well as exposure to chemotherapy and radiation.
What to do when prolactin is elevated
Ongoing monitoring of serum prolactin levels can help ensure that the patient’s levels remain in a normal range (<25 ng/mL).2,3,5,6 If hyperprolactinemia is detected, consider switching to an antipsychotic less likely to increase prolactin. Alternatively, the addition of aripiprazole/brexpiprazole or a dopamine agonist as combination therapy can be considered to rapidly restore normal prolactin levels.2 Such changes should be carefully considered because patients may decompensate if antipsychotics are abruptly switched. An individualized risk vs benefit analysis is necessary for any patient in this situation. Risks include not only the recurrence of psychiatric symptoms but also a potential loss of their current level of functioning. Patients may need to continue to take an antipsychotic that is more likely to increase prolactin, in which case close monitoring is advised as well as collaboration with other physicians and members of the patient’s care team. Involving the patient’s support system is helpful.
1. Weinstein LC, Stefancic A, Cunningham AT, et al. Cancer screening, prevention, and treatment in people with mental illness. CA Cancer J Clin. 2016;66(2):134-151.
2. Rahman T, Sahrmann JM, Olsen MA, et al. Risk of breast cancer with prolactin elevating antipsychotic drugs: an observational study of US women (ages 18–64 years). J Clin Psychopharmacol. 2022;42(1):7-16.
3. Rahman T, Clevenger CV, Kaklamani V, et al. Antipsychotic treatment in breast cancer patients. Am J Psychiatry. 2014;171(6):616-621.
4. Peuskens J, Pani L, Detraux J, et al. The effects of novel and newly approved antipsychotics on serum prolactin levels: a comprehensive review. CNS Drugs. 2014;28(5):421-453.
5. Centers for Disease Control and Prevention, Division of Cancer Prevention and Control. Breast cancer. Accessed June 1, 2022. https://www.cdc.gov/cancer/breast/index.htm
6. Steiner E, Klubert D, Knutson D. Assessing breast cancer risk in women. Am Fam Physician. 2008;78(12):1361-1366.
1. Weinstein LC, Stefancic A, Cunningham AT, et al. Cancer screening, prevention, and treatment in people with mental illness. CA Cancer J Clin. 2016;66(2):134-151.
2. Rahman T, Sahrmann JM, Olsen MA, et al. Risk of breast cancer with prolactin elevating antipsychotic drugs: an observational study of US women (ages 18–64 years). J Clin Psychopharmacol. 2022;42(1):7-16.
3. Rahman T, Clevenger CV, Kaklamani V, et al. Antipsychotic treatment in breast cancer patients. Am J Psychiatry. 2014;171(6):616-621.
4. Peuskens J, Pani L, Detraux J, et al. The effects of novel and newly approved antipsychotics on serum prolactin levels: a comprehensive review. CNS Drugs. 2014;28(5):421-453.
5. Centers for Disease Control and Prevention, Division of Cancer Prevention and Control. Breast cancer. Accessed June 1, 2022. https://www.cdc.gov/cancer/breast/index.htm
6. Steiner E, Klubert D, Knutson D. Assessing breast cancer risk in women. Am Fam Physician. 2008;78(12):1361-1366.
What my Grandma’s schizophrenia taught me
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Grandma was sitting in her chair in the corner of the living room, and her eyes were wide, filled with fear and suspicion as she glanced between me, Mom, and Papa. “They are out to get me,” she said, slightly frantic. She glanced down at her right hand, fixated on a spot on the dorsum. Gingerly lifting her arm, she angled her hand toward my mom’s face. “You see that? They have been conducting experiments on me. I AM THE QUEEN,” she sobbed, “and you are planning together” she said, directing her attention to Papa and me. In that moment, Grandma was convinced Papa and I were conspiring to assassinate her. It hurt to see my grandmother look at me with genuine fear in her eyes. It was overwhelming to watch her deteriorate from the person I had been accustomed to for most of my life to the paranoid individual shaking in front of me.
This was the first time I had really observed my grandmother experiencing acute psychosis. My mom explained to me at a young age that my grandmother had an illness in her mind. I noticed that compared to other people in my life, my grandmother seemed to express less emotion and changed topics in conversations frequently, but by having an understanding provided by my mother, my brother and I didn’t think much of it; that was just Grandma. She would occasionally talk about her experiences with hearing voices or people on the television talking about her. For the most part, though, she was stable; she was able to carry out cleaning, cooking, and watching her favorite shows.
That was until she turned 65 and started on Medicare for insurance. The government required her to trial a less expensive medication and wanted her family practitioner to adjust the medications she had been on for years. This decision was made by people unfamiliar with my grandmother and her story. As a result, my family struggled alongside Grandma for over a month as she battled hallucinations and labile emotions. Living in rural Ohio, she had no access to a psychiatrist or other mental health professional during this period. The adjustments to her medications, changes in her insurance coverage, and lack of consistent psychiatric care led to a deterioration of her stability. This was the only time in my life that I saw Grandma at a place where she would have needed to be hospitalized if the symptoms lasted much longer. I spent evenings sitting with her in that dark and scary place, listening, sympathizing, and challenging her distortions of reality. This experience laid the foundation for my growing passion for providing care and advocating for people experiencing mental illness. I observed firsthand how the absence of consistent, compassionate, and informed care could lead to psychiatric hospitalization.
In the past, my grandfather hid my grandmother’s diagnosis from those around them. This approach prevented my uncle from disclosing the same information to my cousins. I observed how they would look at her with confusion and sometimes fear, which was rooted in a lack of understanding. This desire to hide Grandma’s schizophrenia stemmed from the marginalization society imposed upon her. There were sneers, comments regarding lack of religious faith, and expressions that she was not trying hard enough. My grandparents decided together to inform their church of my grandmother’s illness. The results were astounding. People looked at my grandmother not with confusion but with sympathy and would go out of their way to check on her. Knowledge is power, and awareness can break down stigma. Seeing the difference knowledge could have on a church community further solidified my desire to educate not only patients and their family members but also communities.
Access is another huge barrier my grandmother has faced. There is a lack of referring and awareness as well as large geographic disparities of psychiatrists around my hometown. My grandmother has also had struggles with being able to pay for services, medication, and therapy. This shows the desperate need for more mental health professionals who are competent and knowledgeable in how social determinants of health impact outcomes. These factors contributed to my decision to pursue a Master of Public Health degree. I aspire to use this background to prevent what happened to my Grandma from happening to other patients and to be an advocate for enhanced access to services, improving community mental health and awareness, and promoting continuity of care to increase treatment compliance. That is what my Grandma has fostered in me as a future psychiatrist.
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Grandma was sitting in her chair in the corner of the living room, and her eyes were wide, filled with fear and suspicion as she glanced between me, Mom, and Papa. “They are out to get me,” she said, slightly frantic. She glanced down at her right hand, fixated on a spot on the dorsum. Gingerly lifting her arm, she angled her hand toward my mom’s face. “You see that? They have been conducting experiments on me. I AM THE QUEEN,” she sobbed, “and you are planning together” she said, directing her attention to Papa and me. In that moment, Grandma was convinced Papa and I were conspiring to assassinate her. It hurt to see my grandmother look at me with genuine fear in her eyes. It was overwhelming to watch her deteriorate from the person I had been accustomed to for most of my life to the paranoid individual shaking in front of me.
This was the first time I had really observed my grandmother experiencing acute psychosis. My mom explained to me at a young age that my grandmother had an illness in her mind. I noticed that compared to other people in my life, my grandmother seemed to express less emotion and changed topics in conversations frequently, but by having an understanding provided by my mother, my brother and I didn’t think much of it; that was just Grandma. She would occasionally talk about her experiences with hearing voices or people on the television talking about her. For the most part, though, she was stable; she was able to carry out cleaning, cooking, and watching her favorite shows.
That was until she turned 65 and started on Medicare for insurance. The government required her to trial a less expensive medication and wanted her family practitioner to adjust the medications she had been on for years. This decision was made by people unfamiliar with my grandmother and her story. As a result, my family struggled alongside Grandma for over a month as she battled hallucinations and labile emotions. Living in rural Ohio, she had no access to a psychiatrist or other mental health professional during this period. The adjustments to her medications, changes in her insurance coverage, and lack of consistent psychiatric care led to a deterioration of her stability. This was the only time in my life that I saw Grandma at a place where she would have needed to be hospitalized if the symptoms lasted much longer. I spent evenings sitting with her in that dark and scary place, listening, sympathizing, and challenging her distortions of reality. This experience laid the foundation for my growing passion for providing care and advocating for people experiencing mental illness. I observed firsthand how the absence of consistent, compassionate, and informed care could lead to psychiatric hospitalization.
In the past, my grandfather hid my grandmother’s diagnosis from those around them. This approach prevented my uncle from disclosing the same information to my cousins. I observed how they would look at her with confusion and sometimes fear, which was rooted in a lack of understanding. This desire to hide Grandma’s schizophrenia stemmed from the marginalization society imposed upon her. There were sneers, comments regarding lack of religious faith, and expressions that she was not trying hard enough. My grandparents decided together to inform their church of my grandmother’s illness. The results were astounding. People looked at my grandmother not with confusion but with sympathy and would go out of their way to check on her. Knowledge is power, and awareness can break down stigma. Seeing the difference knowledge could have on a church community further solidified my desire to educate not only patients and their family members but also communities.
Access is another huge barrier my grandmother has faced. There is a lack of referring and awareness as well as large geographic disparities of psychiatrists around my hometown. My grandmother has also had struggles with being able to pay for services, medication, and therapy. This shows the desperate need for more mental health professionals who are competent and knowledgeable in how social determinants of health impact outcomes. These factors contributed to my decision to pursue a Master of Public Health degree. I aspire to use this background to prevent what happened to my Grandma from happening to other patients and to be an advocate for enhanced access to services, improving community mental health and awareness, and promoting continuity of care to increase treatment compliance. That is what my Grandma has fostered in me as a future psychiatrist.
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
Grandma was sitting in her chair in the corner of the living room, and her eyes were wide, filled with fear and suspicion as she glanced between me, Mom, and Papa. “They are out to get me,” she said, slightly frantic. She glanced down at her right hand, fixated on a spot on the dorsum. Gingerly lifting her arm, she angled her hand toward my mom’s face. “You see that? They have been conducting experiments on me. I AM THE QUEEN,” she sobbed, “and you are planning together” she said, directing her attention to Papa and me. In that moment, Grandma was convinced Papa and I were conspiring to assassinate her. It hurt to see my grandmother look at me with genuine fear in her eyes. It was overwhelming to watch her deteriorate from the person I had been accustomed to for most of my life to the paranoid individual shaking in front of me.
This was the first time I had really observed my grandmother experiencing acute psychosis. My mom explained to me at a young age that my grandmother had an illness in her mind. I noticed that compared to other people in my life, my grandmother seemed to express less emotion and changed topics in conversations frequently, but by having an understanding provided by my mother, my brother and I didn’t think much of it; that was just Grandma. She would occasionally talk about her experiences with hearing voices or people on the television talking about her. For the most part, though, she was stable; she was able to carry out cleaning, cooking, and watching her favorite shows.
That was until she turned 65 and started on Medicare for insurance. The government required her to trial a less expensive medication and wanted her family practitioner to adjust the medications she had been on for years. This decision was made by people unfamiliar with my grandmother and her story. As a result, my family struggled alongside Grandma for over a month as she battled hallucinations and labile emotions. Living in rural Ohio, she had no access to a psychiatrist or other mental health professional during this period. The adjustments to her medications, changes in her insurance coverage, and lack of consistent psychiatric care led to a deterioration of her stability. This was the only time in my life that I saw Grandma at a place where she would have needed to be hospitalized if the symptoms lasted much longer. I spent evenings sitting with her in that dark and scary place, listening, sympathizing, and challenging her distortions of reality. This experience laid the foundation for my growing passion for providing care and advocating for people experiencing mental illness. I observed firsthand how the absence of consistent, compassionate, and informed care could lead to psychiatric hospitalization.
In the past, my grandfather hid my grandmother’s diagnosis from those around them. This approach prevented my uncle from disclosing the same information to my cousins. I observed how they would look at her with confusion and sometimes fear, which was rooted in a lack of understanding. This desire to hide Grandma’s schizophrenia stemmed from the marginalization society imposed upon her. There were sneers, comments regarding lack of religious faith, and expressions that she was not trying hard enough. My grandparents decided together to inform their church of my grandmother’s illness. The results were astounding. People looked at my grandmother not with confusion but with sympathy and would go out of their way to check on her. Knowledge is power, and awareness can break down stigma. Seeing the difference knowledge could have on a church community further solidified my desire to educate not only patients and their family members but also communities.
Access is another huge barrier my grandmother has faced. There is a lack of referring and awareness as well as large geographic disparities of psychiatrists around my hometown. My grandmother has also had struggles with being able to pay for services, medication, and therapy. This shows the desperate need for more mental health professionals who are competent and knowledgeable in how social determinants of health impact outcomes. These factors contributed to my decision to pursue a Master of Public Health degree. I aspire to use this background to prevent what happened to my Grandma from happening to other patients and to be an advocate for enhanced access to services, improving community mental health and awareness, and promoting continuity of care to increase treatment compliance. That is what my Grandma has fostered in me as a future psychiatrist.
Clozapine underutilized in treatment-resistant schizophrenia
, and when it is used, the drug is often delayed by several crucial years, reducing chances of efficacy.
“Despite being the only pharmacological therapy approved for treatment-resistant schizophrenia, clozapine is underutilized globally, even in developed countries, where only about 30% of patients who would benefit from the drug receive it,” said John M. Kane, MD, of the department of psychiatry, Zucker Hillside Hospital, Northwell Health, Glen Oaks, N.Y., in a presentation on the subject at the 21st Annual Psychopharmacology Update presented by Current Psychiatry and the American Academy of Clinical Psychiatrists in Cincinnati, Ohio.
Clozapine, a tricyclic dibenzodiazepine available in branded and various generic versions, is approved by the U.S. Food and Drug Administration as a third-line therapy for severe, treatment-resistant schizophrenia, with studies showing benefits exceeding those of any other antipsychotics for the indication.
But while recommendations suggest use after a trial of two or more antipsychotics, with at least one being an atypical antipsychotic, one recent review finds delays in clozapine commencement ranging from 19.3 weeks to 5.5 years, and the duration of illness prior to clozapine use ranging from 1.1 to 9.7 years.
Blood monitoring, side effects
The key deterrents preventing many clinicians and patients from trying clozapine sooner are the drug’s safety and tolerability profiles, and notably the requirement of regular blood testing due to an increased risk of agranulocytosis.
Specifically, the blood testing is required every week for 6 months, then every other week for the next 6 months, and then once a month after that; however, “many of us think that that’s excessive at this point in time,” Dr. Kane noted.
Various other potential side effects are also of concern, including myocarditis, seizures, constipation, arrhythmia, hypersalivation, pneumonia, and metabolic symptoms including diabetes.
In terms of the common strategies that clinicians turn to when patients fail to respond to their current antipsychotic, including increasing doses, combining agents, or treatment switching, “none of the strategies likely rival clozapine in terms of efficacy,” Dr. Kane said.
Regarding higher dosing: “There is very little data suggesting that higher doses of antipsychotic drugs will work when the moderate or recommended dose has not worked,” he said.
Combination therapy strategies may provide benefits, but “they’re not a substitute for clozapine,” Dr. Kane added, noting that the combinations that do appear to be the most effective involve clozapine.
And regarding drug switching, studies suggest the likelihood of response in switching from one drug to another is “actually very low,” Dr. Kane added.
Clozapine also doesn’t work for all – the response rate runs between about 30% and 60%, Dr. Kane said, but when it is effective, the benefits can be profound.
“There are some patients who have a very pronounced response to clozapine – some patients describe it as life-changing,” he said.
Treatment delays reduce efficacy
Importantly, the delays before receiving clozapine are not inconsequential – data show that each outpatient antipsychotic trial prior to clozapine reduces the likelihood of response by 8%-11%, and each hospital admission further reduces the likelihood of response by 4%-8%, underscoring the need to identify treatment resistance as early as possible, Dr. Kane said.
“It’s critically important to try to identify treatment resistance earlier than we usually do because if we can get it under control sooner, we have a better chance of improving the patient’s outcome, and this has been shown in a number of studies,” he said.
“The longer you wait, the less likely you are to see a good response even to clozapine.”
Despite the concerns about clozapine, Dr. Kane notes that even the blood monitoring does not appear to be a big complaint for patients, especially they are improving.
“In our experience, the patients who benefit from clozapine don’t really have a problem with the monitoring,” he said.
“In fact, patients who benefit from clozapine are much more adherent to the medication than other patients that we see, which is understandable, because if you feel you’re really getting a benefit from medicine, you’re going to be much more motivated to take it even if it has side effects.”
A recent systematic review of 13 studies and 1,487 patients backs that up, concluding that “patients generally have a favorable experience when being treated with clozapine,” with the caveat that “conclusions are limited by the risk of bias, particularly survivorship bias.”
Preference for clozapine over other antipsychotic medications was reported by 54%-86% of patients in the review, with specific improvements in mood (11%-78%) and cognition (5%-68%).
Clinicians the biggest ‘obstacle’
Dr. Kane notes that an important factor in underutilization could indeed be the manner in which clinicians discuss clozapine with their patients – often opening the discussion by focusing on the negative aspects that, without the context of the potential benefits, can be deal-breakers for patient from the start.
“The clinicians in my opinion are really the obstacle,” Dr. Kane said. “What we always hear from clinicians is ‘I can’t do it because the patient refuses, or the patient doesn’t like the side effects’.”
Dr. Kane notes that most side effects can indeed be managed – regarding the risk for metabolic syndrome, for instance, he recommends that patients should be given metformin from the beginning when they’re started on clozapine.
He adds that in most cases, a 3-month trial is enough to answer the question of whether clozapine is working or not.
“Three months is a good trial, but it may not even tell you the total response to clozapine because that may actually accrue over time,” he said. “We’ve seen patients who actually get better and better beyond 3 months.”
Not offering the drug to patients, however, is doing them a serious disservice, Dr. Kane added.
“What I tell patients and families is that it would be a shame to miss this opportunity for a potential treatment that could be life-changing,” he said. “Does it have potential side effects? Yes. Do you have to get blood tests? Yes. And I can’t tell by evaluating a patient’s history or examining that patient whether or not they’re going to be a good responder. But would you really want to miss an opportunity to find that out?”
“To me the argument is – let’s try this drug for 3 months and see what effect it has, and at that point you’ll be in a much better position to make a decision about the benefits versus risk,” Dr. Kane said.
The only FDA-approved drug for treatment-resistant schizophrenia
Remarkably, clozapine isn’t just the only drug to currently have approval from the FDA for treatment-resistant schizophrenia – it has been for the last 3 decades.
“There have been attempts to develop medications with similar efficacy, but they have not succeeded,” Dr. Kane said in an interview. “We are still uncertain as to what accounts for clozapine’s unique qualities.”
Yet, with treatment-resistant schizophrenia patients representing some of the most dire mental illness cases clinicians may face, the need for better treatment decisions – and additional options – is pressing, Dr. Kane said.
“[The lack of any other drugs] is a big embarrassment to our field, in my opinion,” he said. “I’m a big proponent of clozapine, but we should have found another substance by now that could substitute for clozapine, which obviously has a lot of side effects and is not the easiest drug to use.”
Dr. Kane reported relationships either as a speaker or consultant/advisory board member and/or receives research grant support from Alkermes, Allergan, Click Therapeutics, Dainippon Sumitomo, H. Lundbeck, HLS Therapeutics, Indivior, Intra-Cellular Therapies, Janssen Pharmaceutical, Johnson & Johnson, LB Pharmaceuticals, Merck, Minerva, Neurocrine, Neumora Therapeutics, Novartis Pharmaceuticals, Otsuka, Reviva, Roche, Saladax, Sunovion, Takeda, and Teva. Dr. Kane receives non-mutual funds stock ownership/stock options from LB Pharmaceuticals, Vanguard Research Group, and North Shore Therapeutics, and receives patent holder/royalties paid by UpToDate.
The Psychopharmacology Update was sponsored by Medscape Live. Medscape Live and this news organization are owned by the same parent company.
, and when it is used, the drug is often delayed by several crucial years, reducing chances of efficacy.
“Despite being the only pharmacological therapy approved for treatment-resistant schizophrenia, clozapine is underutilized globally, even in developed countries, where only about 30% of patients who would benefit from the drug receive it,” said John M. Kane, MD, of the department of psychiatry, Zucker Hillside Hospital, Northwell Health, Glen Oaks, N.Y., in a presentation on the subject at the 21st Annual Psychopharmacology Update presented by Current Psychiatry and the American Academy of Clinical Psychiatrists in Cincinnati, Ohio.
Clozapine, a tricyclic dibenzodiazepine available in branded and various generic versions, is approved by the U.S. Food and Drug Administration as a third-line therapy for severe, treatment-resistant schizophrenia, with studies showing benefits exceeding those of any other antipsychotics for the indication.
But while recommendations suggest use after a trial of two or more antipsychotics, with at least one being an atypical antipsychotic, one recent review finds delays in clozapine commencement ranging from 19.3 weeks to 5.5 years, and the duration of illness prior to clozapine use ranging from 1.1 to 9.7 years.
Blood monitoring, side effects
The key deterrents preventing many clinicians and patients from trying clozapine sooner are the drug’s safety and tolerability profiles, and notably the requirement of regular blood testing due to an increased risk of agranulocytosis.
Specifically, the blood testing is required every week for 6 months, then every other week for the next 6 months, and then once a month after that; however, “many of us think that that’s excessive at this point in time,” Dr. Kane noted.
Various other potential side effects are also of concern, including myocarditis, seizures, constipation, arrhythmia, hypersalivation, pneumonia, and metabolic symptoms including diabetes.
In terms of the common strategies that clinicians turn to when patients fail to respond to their current antipsychotic, including increasing doses, combining agents, or treatment switching, “none of the strategies likely rival clozapine in terms of efficacy,” Dr. Kane said.
Regarding higher dosing: “There is very little data suggesting that higher doses of antipsychotic drugs will work when the moderate or recommended dose has not worked,” he said.
Combination therapy strategies may provide benefits, but “they’re not a substitute for clozapine,” Dr. Kane added, noting that the combinations that do appear to be the most effective involve clozapine.
And regarding drug switching, studies suggest the likelihood of response in switching from one drug to another is “actually very low,” Dr. Kane added.
Clozapine also doesn’t work for all – the response rate runs between about 30% and 60%, Dr. Kane said, but when it is effective, the benefits can be profound.
“There are some patients who have a very pronounced response to clozapine – some patients describe it as life-changing,” he said.
Treatment delays reduce efficacy
Importantly, the delays before receiving clozapine are not inconsequential – data show that each outpatient antipsychotic trial prior to clozapine reduces the likelihood of response by 8%-11%, and each hospital admission further reduces the likelihood of response by 4%-8%, underscoring the need to identify treatment resistance as early as possible, Dr. Kane said.
“It’s critically important to try to identify treatment resistance earlier than we usually do because if we can get it under control sooner, we have a better chance of improving the patient’s outcome, and this has been shown in a number of studies,” he said.
“The longer you wait, the less likely you are to see a good response even to clozapine.”
Despite the concerns about clozapine, Dr. Kane notes that even the blood monitoring does not appear to be a big complaint for patients, especially they are improving.
“In our experience, the patients who benefit from clozapine don’t really have a problem with the monitoring,” he said.
“In fact, patients who benefit from clozapine are much more adherent to the medication than other patients that we see, which is understandable, because if you feel you’re really getting a benefit from medicine, you’re going to be much more motivated to take it even if it has side effects.”
A recent systematic review of 13 studies and 1,487 patients backs that up, concluding that “patients generally have a favorable experience when being treated with clozapine,” with the caveat that “conclusions are limited by the risk of bias, particularly survivorship bias.”
Preference for clozapine over other antipsychotic medications was reported by 54%-86% of patients in the review, with specific improvements in mood (11%-78%) and cognition (5%-68%).
Clinicians the biggest ‘obstacle’
Dr. Kane notes that an important factor in underutilization could indeed be the manner in which clinicians discuss clozapine with their patients – often opening the discussion by focusing on the negative aspects that, without the context of the potential benefits, can be deal-breakers for patient from the start.
“The clinicians in my opinion are really the obstacle,” Dr. Kane said. “What we always hear from clinicians is ‘I can’t do it because the patient refuses, or the patient doesn’t like the side effects’.”
Dr. Kane notes that most side effects can indeed be managed – regarding the risk for metabolic syndrome, for instance, he recommends that patients should be given metformin from the beginning when they’re started on clozapine.
He adds that in most cases, a 3-month trial is enough to answer the question of whether clozapine is working or not.
“Three months is a good trial, but it may not even tell you the total response to clozapine because that may actually accrue over time,” he said. “We’ve seen patients who actually get better and better beyond 3 months.”
Not offering the drug to patients, however, is doing them a serious disservice, Dr. Kane added.
“What I tell patients and families is that it would be a shame to miss this opportunity for a potential treatment that could be life-changing,” he said. “Does it have potential side effects? Yes. Do you have to get blood tests? Yes. And I can’t tell by evaluating a patient’s history or examining that patient whether or not they’re going to be a good responder. But would you really want to miss an opportunity to find that out?”
“To me the argument is – let’s try this drug for 3 months and see what effect it has, and at that point you’ll be in a much better position to make a decision about the benefits versus risk,” Dr. Kane said.
The only FDA-approved drug for treatment-resistant schizophrenia
Remarkably, clozapine isn’t just the only drug to currently have approval from the FDA for treatment-resistant schizophrenia – it has been for the last 3 decades.
“There have been attempts to develop medications with similar efficacy, but they have not succeeded,” Dr. Kane said in an interview. “We are still uncertain as to what accounts for clozapine’s unique qualities.”
Yet, with treatment-resistant schizophrenia patients representing some of the most dire mental illness cases clinicians may face, the need for better treatment decisions – and additional options – is pressing, Dr. Kane said.
“[The lack of any other drugs] is a big embarrassment to our field, in my opinion,” he said. “I’m a big proponent of clozapine, but we should have found another substance by now that could substitute for clozapine, which obviously has a lot of side effects and is not the easiest drug to use.”
Dr. Kane reported relationships either as a speaker or consultant/advisory board member and/or receives research grant support from Alkermes, Allergan, Click Therapeutics, Dainippon Sumitomo, H. Lundbeck, HLS Therapeutics, Indivior, Intra-Cellular Therapies, Janssen Pharmaceutical, Johnson & Johnson, LB Pharmaceuticals, Merck, Minerva, Neurocrine, Neumora Therapeutics, Novartis Pharmaceuticals, Otsuka, Reviva, Roche, Saladax, Sunovion, Takeda, and Teva. Dr. Kane receives non-mutual funds stock ownership/stock options from LB Pharmaceuticals, Vanguard Research Group, and North Shore Therapeutics, and receives patent holder/royalties paid by UpToDate.
The Psychopharmacology Update was sponsored by Medscape Live. Medscape Live and this news organization are owned by the same parent company.
, and when it is used, the drug is often delayed by several crucial years, reducing chances of efficacy.
“Despite being the only pharmacological therapy approved for treatment-resistant schizophrenia, clozapine is underutilized globally, even in developed countries, where only about 30% of patients who would benefit from the drug receive it,” said John M. Kane, MD, of the department of psychiatry, Zucker Hillside Hospital, Northwell Health, Glen Oaks, N.Y., in a presentation on the subject at the 21st Annual Psychopharmacology Update presented by Current Psychiatry and the American Academy of Clinical Psychiatrists in Cincinnati, Ohio.
Clozapine, a tricyclic dibenzodiazepine available in branded and various generic versions, is approved by the U.S. Food and Drug Administration as a third-line therapy for severe, treatment-resistant schizophrenia, with studies showing benefits exceeding those of any other antipsychotics for the indication.
But while recommendations suggest use after a trial of two or more antipsychotics, with at least one being an atypical antipsychotic, one recent review finds delays in clozapine commencement ranging from 19.3 weeks to 5.5 years, and the duration of illness prior to clozapine use ranging from 1.1 to 9.7 years.
Blood monitoring, side effects
The key deterrents preventing many clinicians and patients from trying clozapine sooner are the drug’s safety and tolerability profiles, and notably the requirement of regular blood testing due to an increased risk of agranulocytosis.
Specifically, the blood testing is required every week for 6 months, then every other week for the next 6 months, and then once a month after that; however, “many of us think that that’s excessive at this point in time,” Dr. Kane noted.
Various other potential side effects are also of concern, including myocarditis, seizures, constipation, arrhythmia, hypersalivation, pneumonia, and metabolic symptoms including diabetes.
In terms of the common strategies that clinicians turn to when patients fail to respond to their current antipsychotic, including increasing doses, combining agents, or treatment switching, “none of the strategies likely rival clozapine in terms of efficacy,” Dr. Kane said.
Regarding higher dosing: “There is very little data suggesting that higher doses of antipsychotic drugs will work when the moderate or recommended dose has not worked,” he said.
Combination therapy strategies may provide benefits, but “they’re not a substitute for clozapine,” Dr. Kane added, noting that the combinations that do appear to be the most effective involve clozapine.
And regarding drug switching, studies suggest the likelihood of response in switching from one drug to another is “actually very low,” Dr. Kane added.
Clozapine also doesn’t work for all – the response rate runs between about 30% and 60%, Dr. Kane said, but when it is effective, the benefits can be profound.
“There are some patients who have a very pronounced response to clozapine – some patients describe it as life-changing,” he said.
Treatment delays reduce efficacy
Importantly, the delays before receiving clozapine are not inconsequential – data show that each outpatient antipsychotic trial prior to clozapine reduces the likelihood of response by 8%-11%, and each hospital admission further reduces the likelihood of response by 4%-8%, underscoring the need to identify treatment resistance as early as possible, Dr. Kane said.
“It’s critically important to try to identify treatment resistance earlier than we usually do because if we can get it under control sooner, we have a better chance of improving the patient’s outcome, and this has been shown in a number of studies,” he said.
“The longer you wait, the less likely you are to see a good response even to clozapine.”
Despite the concerns about clozapine, Dr. Kane notes that even the blood monitoring does not appear to be a big complaint for patients, especially they are improving.
“In our experience, the patients who benefit from clozapine don’t really have a problem with the monitoring,” he said.
“In fact, patients who benefit from clozapine are much more adherent to the medication than other patients that we see, which is understandable, because if you feel you’re really getting a benefit from medicine, you’re going to be much more motivated to take it even if it has side effects.”
A recent systematic review of 13 studies and 1,487 patients backs that up, concluding that “patients generally have a favorable experience when being treated with clozapine,” with the caveat that “conclusions are limited by the risk of bias, particularly survivorship bias.”
Preference for clozapine over other antipsychotic medications was reported by 54%-86% of patients in the review, with specific improvements in mood (11%-78%) and cognition (5%-68%).
Clinicians the biggest ‘obstacle’
Dr. Kane notes that an important factor in underutilization could indeed be the manner in which clinicians discuss clozapine with their patients – often opening the discussion by focusing on the negative aspects that, without the context of the potential benefits, can be deal-breakers for patient from the start.
“The clinicians in my opinion are really the obstacle,” Dr. Kane said. “What we always hear from clinicians is ‘I can’t do it because the patient refuses, or the patient doesn’t like the side effects’.”
Dr. Kane notes that most side effects can indeed be managed – regarding the risk for metabolic syndrome, for instance, he recommends that patients should be given metformin from the beginning when they’re started on clozapine.
He adds that in most cases, a 3-month trial is enough to answer the question of whether clozapine is working or not.
“Three months is a good trial, but it may not even tell you the total response to clozapine because that may actually accrue over time,” he said. “We’ve seen patients who actually get better and better beyond 3 months.”
Not offering the drug to patients, however, is doing them a serious disservice, Dr. Kane added.
“What I tell patients and families is that it would be a shame to miss this opportunity for a potential treatment that could be life-changing,” he said. “Does it have potential side effects? Yes. Do you have to get blood tests? Yes. And I can’t tell by evaluating a patient’s history or examining that patient whether or not they’re going to be a good responder. But would you really want to miss an opportunity to find that out?”
“To me the argument is – let’s try this drug for 3 months and see what effect it has, and at that point you’ll be in a much better position to make a decision about the benefits versus risk,” Dr. Kane said.
The only FDA-approved drug for treatment-resistant schizophrenia
Remarkably, clozapine isn’t just the only drug to currently have approval from the FDA for treatment-resistant schizophrenia – it has been for the last 3 decades.
“There have been attempts to develop medications with similar efficacy, but they have not succeeded,” Dr. Kane said in an interview. “We are still uncertain as to what accounts for clozapine’s unique qualities.”
Yet, with treatment-resistant schizophrenia patients representing some of the most dire mental illness cases clinicians may face, the need for better treatment decisions – and additional options – is pressing, Dr. Kane said.
“[The lack of any other drugs] is a big embarrassment to our field, in my opinion,” he said. “I’m a big proponent of clozapine, but we should have found another substance by now that could substitute for clozapine, which obviously has a lot of side effects and is not the easiest drug to use.”
Dr. Kane reported relationships either as a speaker or consultant/advisory board member and/or receives research grant support from Alkermes, Allergan, Click Therapeutics, Dainippon Sumitomo, H. Lundbeck, HLS Therapeutics, Indivior, Intra-Cellular Therapies, Janssen Pharmaceutical, Johnson & Johnson, LB Pharmaceuticals, Merck, Minerva, Neurocrine, Neumora Therapeutics, Novartis Pharmaceuticals, Otsuka, Reviva, Roche, Saladax, Sunovion, Takeda, and Teva. Dr. Kane receives non-mutual funds stock ownership/stock options from LB Pharmaceuticals, Vanguard Research Group, and North Shore Therapeutics, and receives patent holder/royalties paid by UpToDate.
The Psychopharmacology Update was sponsored by Medscape Live. Medscape Live and this news organization are owned by the same parent company.
FROM PSYCHOPHARMACOLOGY UPDATE