User login
Effectiveness and Safety of Droperidol Use in the VA Greater Los Angeles Healthcare System Emergency Department
Effectiveness and Safety of Droperidol Use in the VA Greater Los Angeles Healthcare System Emergency Department
Droperidol is a butyrophenone antipsychotic approved by the US Food and Drug Administration (FDA) for use in postoperative nausea and vomiting (PONV). Off-label, it has also been utilized for its sedative, anxiolytic, and analgesic properties.1 While its exact mechanism of action remains elusive, it is believed that binding to postsynaptic γ-aminobutyric acid receptors induces anxiolysis and sedation, while dopaminergic activity in the chemoreceptor trigger zone contributes to its antiemetic effects.2 Since the introduction of droperidol in 1967, it has been widely used by emergency physicians, psychiatrists, and anesthesiologists globally.1
Despite its therapeutic efficacy, use of droperidol has been tempered by concerns regarding its cardiovascular safety profile, specifically its potential to prolong the QT interval and precipitate cardiac arrhythmias. In 2001, the FDA placed a boxed warning on droperidol that mandated electrocardiogram (EKG) monitoring before and after treatment. This requirement has led to a widespread decrease in use, and the FDA decision sparked significant controversy among clinicians, with many organizations arguing that the evidence did not support this mandate.1
Further review of the cases cited by the FDA revealed that there were 277 reported cases of droperidol-related adverse events (AEs), but many of these cases were duplicates and occurred outside the US.3 Additionally, the doses of droperidol used in these cases were significantly higher than the typical doses used in the emergency department (ED), ranging from 25 to 250 mg.4 Typical doses for PONV range from 0.625 to 2.5 mg intravenous (IV) or intramuscular (IM). Recommended doses for agitation typically range from 2.5 to 10 mg IV and 5 to 10 mg IM.5
There has been growing interest in reevaluating the risk-benefit profile of droperidol in the ED. Since the original decision by the FDA, multiple publications have challenged the idea that droperidol has significantly higher risks associated with its use. The 2014 review by the Clinical Guidelines Committee of the American Academy of Emergency Medicine did not find evidence that low-dose droperidol (< 2.5 is unsafe for use in the ED.6 A retrospective cohort study from 2020 found no fatalities in 5784 patients. Furthermore, a prospective observational study of 1009 patients at 6 EDs who received high-dose droperidol (≤ 20.0 mg) found no evidence of increased risk for QT prolongation.7 The evidence supports the safety of droperidol for use in prehospital and hospital settings as well as in pediatric, adult, and geriatric populations.8-14 Droperidol was eventually reintroduced in 2019, which led to increased use.
The US Department of Veterans Affairs (VA) formulary has limited options (eg, haloperidol and olanzapine) that have robust evidence supporting their use to treat aggression or psychosis-related agitation. Ziprasidone injections are not on the formulary and require authorization for use, which may delay patient care and pose a safety risk. In 2021, VA Greater Los Angeles Healthcare System (VAGLAHS) received Pharmacy and Therapeutics Committee approval to use droperidol in the ED for agitation or nausea and vomiting. The purpose of this study was to evaluate safety outcomes for patients prescribed droperidol and the need for rescue medications (ie, effectiveness) in the VAGLAHS ED.
Methods
This retrospective chart review analyzed patients administered droperidol in the VAGLAHS ED from February 1, 2021, through April 30, 2023. A list of patients who had droperidol ordered in the VAGLAHS ED was obtained from the Veterans Health Information Systems and Technology Architecture. Charts were reviewed using the Computerized Patient Record System to confirm droperidol administration. Nurse documentation was reviewed to confirm the time, dose, and route of administration. In addition, droperidol dosages were categorized as < 5 mg, 5 to 10 mg, and > 10 mg to review outcomes based on the total amount administered to each patient.
Patients included in the study received droperidol in the ED within the study period, were aged ≥ 18 years, and received droperidol for acute agitation or antiemesis. Patients were excluded if they received droperidol for an indication other than agitation or antiemesis.
The study team reviewed the list of patients and audited the collected data. Reviewers were trained on the study protocols and variables identified. The following data were collected: patient demographics (age, sex, race, height, weight, allergies), Charlson Comorbidity Index (CCI) conditions, cardiac comorbidities, laboratory values at admission, basic metabolic panels, liver function tests, droperidol use (doses, indications, and documentation of safety), concomitant medications ordered with the initial droperidol order, AEs (arrhythmias, extrapyramidal symptoms [EPS], respiratory depression, mortality), medications used within 60 minutes of droperidol administration (rescue medications), other medications used within 24 hours after droperidol administration, and EKG/QTc (corrected QT interval) intervals. The data reviewed and recorded were from the date of the initial patient ED visit.
Outcomes
The primary outcome was all-cause mortality within 24 hours after droperidol administration. This outcome was measured in all patients included in this study. Secondary outcomes included rescue medications needed after droperidol administration, incidence of QT prolongation, incidence of EPS (defined as akathisia, dystonia, parkinsonism, or tardive dyskinesia), and incidence of respiratory depression. Clinically significant QTc was defined as an interval of ≥ 500 ms with incidence of arrhythmias, code blues, or intubations. Baseline risk factors for QTc prolongation were taken into consideration including electrolyte abnormalities, concomitant QT-prolonging medications, CCI score, and cardiac comorbidities. Incidence of EPS was counted if patients received medications such as diphenhydramine or benztropine after droperidol administration in addition to documentation of EPS signs and symptoms. Incidences of EPS findings were reviewed by emergency department physicians to confirm the diagnosis.
Safety was assessed by quantifying mortality rates 24 hours after droperidol administration along with incidence of AEs associated with droperidol use including QT prolongation, EPS, and respiratory depression.
The necessity of rescue medication use was assessed by nursing documentation, additional medications ordered, and/or no additional medications required for agitation within 60 minutes of droperidol administration. Sixty minutes was the chosen timeframe given that the onset of droperidol action is between 3 and 10 minutes and peaks in about 30 minutes. Medications that were considered rescue medications included diphenhydramine < 25 mg, diphenhydramine 25 to 50 mg, lorazepam < 1 mg, lorazepam 1 to 2 mg, diphenhydramine < 25 mg and lorazepam < 1 mg, diphenhydramine < 25 mg and lorazepam 1 to 2 mg, diphenhydramine 25 to 50 mg and lorazepam 1 to 2 mg, and other medications, the names and doses of which were manually documented by investigators.
Statistical Analysis
For all variables in the study, descriptive analysis was used to categorize findings. Microsoft Excel was used to calculate means, frequency counts, percentages, and categorize data.
Results
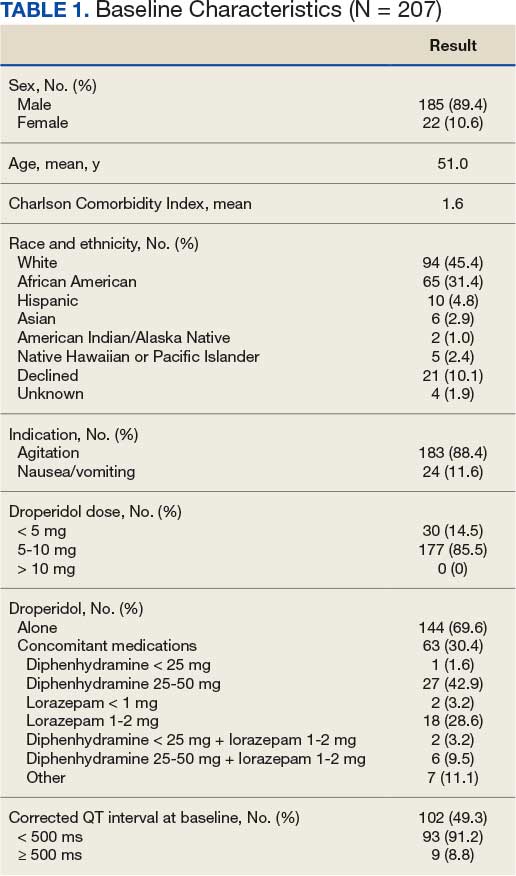
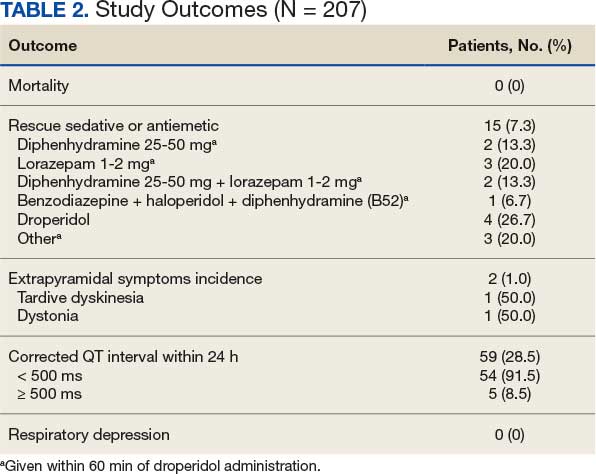
Between February 1, 2021, and April 16, 2023, 214 patients received droperidol in the VAGLAHS ED, and 207 patients were included in the study. Seven patients did not receive droperidol for the indications included (acute agitation or antiemesis). Most of the study population (89.4%) was male, and the mean age was 51.0 years. The mean CCI was 1.6. In the study, 183 (88.4%) patients received droperidol for agitation and 24 (11.6%) for nausea and vomiting (Table 1).
Primary Outcome
No deaths were observed in a 24-hour period after droperidol administration among the 207 patients included in the study. There were also no arrhythmias, code blues, or intubations observed with the administration of droperidol (Table 2).
Secondary Outcomes
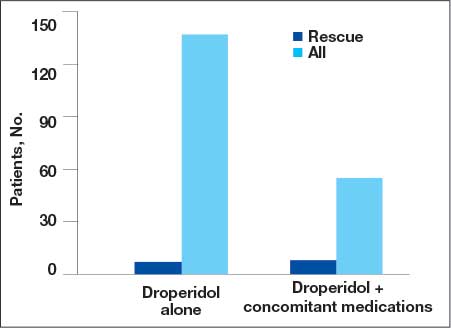
A total of 144 patients (69.6%) received droperidol alone to resolve agitation or nausea and vomiting. In the remaining population, 63 (30.4%) patients were given medications concomitantly with droperidol.
Fifteen patients (7.2%) required rescue medications that were administered within 60 minutes of droperidol administration. Rescue medications were required for 7 patients (4.9%) who initially received droperidol alone compared with 8 patients (12.7%) who were administered concomitant medications with droperidol (Figure).
Extrapyramidal Symptoms
EPS occurred in 2 patients (1.0%). There was 1 incidence of tardive dyskinesia (TD) in which the patient received droperidol 2.5 mg IM for emesis. TD was resolved with diphenhydramine 50 mg. A second patient who experienced dystonia received droperidol 10 mg IM for agitation. Dystonia was resolved with benztropine 2 mg. Both patients had a CCI of 0, no cardiac comorbidities, and laboratory test results were within reference ranges. The second patient received olanzapine within 24 hours of droperidol administration; however, it was after the EPS event.
QTc Prolongation
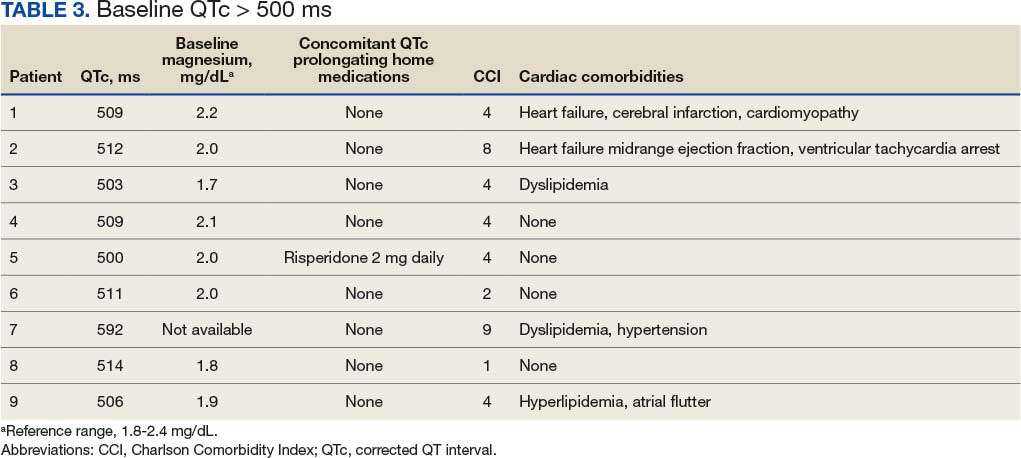
Baseline EKGs (within 6 months prior to ED visit) were available for 102 patients (49.3%). Nine patients (8.8%) had a reported baseline QTc of ≥ 500 ms (Table 3). Of these patients, 6 had a repeat EKG and 5 had a repeat QTc < 500 ms. One patient had a baseline and repeated QTc of 512 ms with essentially no change after droperidol administration. Only 1 patient was on a potentially QTc-prolonging medication at home. None of the patients with baseline QTc > 500 ms experienced arrhythmias after droperidol administration.
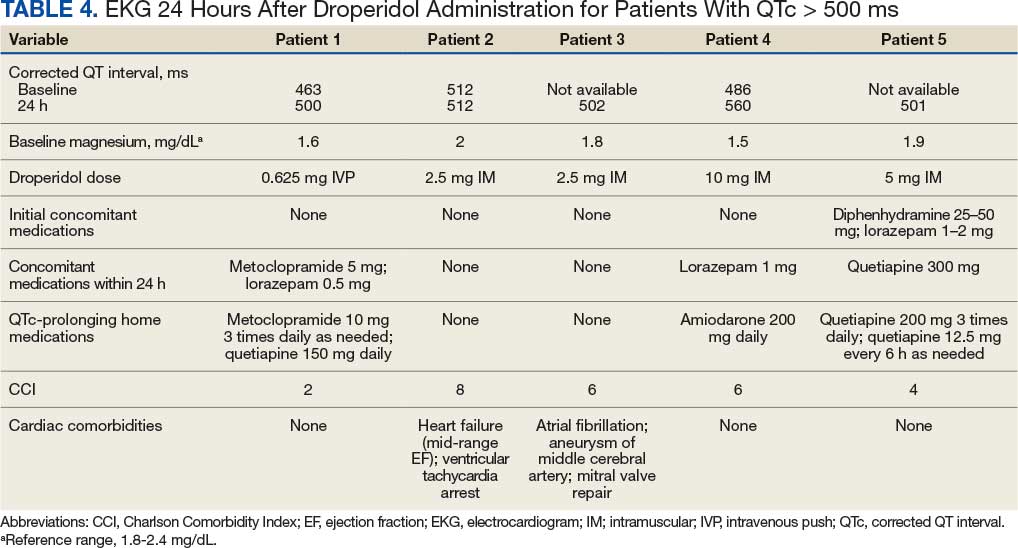
We found that 59 patients (28.5%) had EKGs performed within 24 hours after droperidol administration. Five patients had documented QTc ≥ 500 ms, but no arrhythmias were observed in a 24-hour period. Table 4 describes the additional medications administered after the 60-minute window but within 24 hours after droperidol administration. Quetiapine 300 mg and metoclopramide 5 mg were the only medications documented that can potentially increase QTc. Patient adherence to home medications and the timing of the last dose prior to ED visit were unknown. However, no arrhythmias were noted in these patients with QTc changes. No patients experienced respiratory depression within 24 hours of droperidol administration.
Older Adult Patients
Thirty-eight patients were aged ≥ 65 years with a mean age of 74.2 years. Thirty-four patients (89.5%) received droperidol for agitation and 4 (10.6%) for nausea and vomiting. Only 21 patients had a baseline EKG, and 4 had QTc ≤ 500 ms. At 24 hours, EKGs were performed for 18 patients and 3 had a QTc ≤ 500 ms. No mortality or arrhythmias were reported and there were no incidences of rescue medications, EPS, or respiratory depression.
Discussion
The study included 207 patients who received droperidol for either agitation or nausea/vomiting in the VAGLAHS ED. No mortality occurred within 24 hours of droperidol administration, which is consistent with recent studies.8-14
Furthermore, 59 patients (28.5%) had an EKG performed within 24 hours of droperidol administration; 5 patients had documented QTc ≥ 500 ms. Only 3 of the patients with prolonged QTc had baseline readings for comparison. Only 2 patients had an increase in QTc interval. No arrhythmias were observed; however, the effects of observing QTc prolongation were limited due to the lack of post-EKG readings following droperidol administration. Because of the retrospective nature of the study, neither standardization of EKG at baseline nor 24-hour postadministration were possible. The study found that droperidol was effective with only 15 patients (7.3%) requiring rescue medications. In the patients who were given medications concomitantly with droperidol, it was not possible to conclude whether the patients would have required rescue medications to resolve their agitation or nausea/vomiting. Administration of concomitant medications with droperidol may be attributed to practice patterns associated with haloperidol use, which is frequently administered with concomitant medications such as diphenhydramine and/or a benzodiazepine.
AEs were rare with no documentation of respiratory depression and 2 cases (1.0%) of EPS. Both incidences of EPS resolved with diphenhydramine or benztropine. However, given the reliance on nursing documentation to capture AEs, the number of events may have been underreported.
Limitations
Standardization of dosing was a limiting factor that could affect the need for rescue medications. Another limitation was reliance on nursing reports of resolution of symptoms and comfort with agitated patients. Given the retrospective design and small sample size, this study may not have captured all potential AEs. However, the doses administered within this study population were consistent with what was expected based on other studies.8-14
Conclusions
Droperidol, an antipsychotic, is currently approved for PONV, but is also used off-label for agitation. This study found no fatalities among patients who received droperidol in the ED. The findings suggest that droperidol used for agitation and as an antiemetic, despite its FDA boxed warning, appears to be safe and showed no evidence of mortality, arrhythmias, code blues, or intubations despite the lack of postdose EKG monitoring. Among the 38 patients aged ≥ 65 years, the use of droperidol revealed no increased risks. It should be noted that droperidol appeared safe and few patients required rescue medications within this study population.
- Perkins J, Ho JD, Vilke GM, DeMers G. American Academy of Emergency Medicine Position Statement: Safety of droperidol use in the emergency department. J Emerg Med. 2015;49:91-97. doi:10.1016/j.jemermed.2014.12.024
- Siegel RB, Motov SM, Marcolini EG. Droperidol use in the emergency department: a clinical review. J Emerg Med. 2023;64:289-294. doi:10.1016/j.jemermed.2022.12.012
- Jackson CW, Sheehan AH, Reddan JG. Evidencebased review of the black-box warning for droperidol. Am J Health Syst Pharm. 2007;64:1174-1186. doi:10.2146/ajhp060505
- Habib AS, Gan TJ. Food and Drug Administration black box warning on the perioperative use of droperidol: a review of the cases. Anesth Analg. 2003;96(5):1377-1379. doi:10.1213/01.ane.0000063923.87560.37
- Droperidol. In: Micromedex (electronic version). IBM Watson Health; 2019. Accessed March 2, 2026. https://www .micromedexsolutions.com
- Gaw CM, Cabrera D, Bellolio F, Mattson AE, Lohse CM, Jeffery MM. Effectiveness and safety of droperidol in a United States emergency department. Am J Emerg Med. 2020;38:1310-1314. doi:10.1016/j.ajem.2019.09.007
- Calver L, Page CB, Downes MA, et al. The safety and effectiveness of droperidol for sedation of acute behavioral disturbance in the emergency department. Ann Emerg Med. 2015;66(3):230-238.e1. doi:10.1016/j.annemergmed.2015.03.016
- Ernst R, Wagstaff H, Smith M, et al. Droperidol administration among emergency department patients with abdominal pain, nausea, and vomiting. Am J Emerg Med. 2024;85:44-47. doi:10.1016/j.ajem.2024.07.060
- Szwak K, Sacchetti A. Droperidol use in pediatric emergency department patients. Pediatr Emerg Care. 2010;26:248-250. doi:10.1097/pec.0b013e3181d6d9f2
- Chase PB, Biros MH. A retrospective review of the use and safety of droperidol in a large, high-risk, inner-city emergency department patient population. Acad Emerg Med. 2002;9:1402-1410. doi:10.1111/j.1553-2712.2002.tb01609.x
- Mattson A, Friend K, Brown CS, Cabrera D. Reintegrating droperidol into emergency medicine practice. Am J Health Syst Pharm. 2020;77(22):1838-1845. doi:10.1093/ajhp/zxaa271
- Cole JB, Stang JL, DeVries PA, Martel ML, Miner JR, Driver BE. A prospective study of intramuscular droperidol or olanzapine for acute agitation in the emergency department: a natural experiment owing to drug shortages. Ann Emerg Med. 2021;78(2):274-286. doi:10.1016/j.annemergmed.2021.01.005
- Page CB, Parker LE, Rashford SJ, et al. Prospective study of the safety and effectiveness of droperidol in elderly patients for pre-hospital acute behavioural disturbance. Emerg Med Australas. 2020;32(5):731-736. doi:10.1111/1742-6723.13496
- Page CB, Parker LE, Rashford SJ, et al. A prospective study of the safety and effectiveness of droperidol inchildren for prehospital acute behavioral disturbance. Prehosp Emerg Care. 2018;23:519-526. doi:10.1080/10903127.2018.1542473
Droperidol is a butyrophenone antipsychotic approved by the US Food and Drug Administration (FDA) for use in postoperative nausea and vomiting (PONV). Off-label, it has also been utilized for its sedative, anxiolytic, and analgesic properties.1 While its exact mechanism of action remains elusive, it is believed that binding to postsynaptic γ-aminobutyric acid receptors induces anxiolysis and sedation, while dopaminergic activity in the chemoreceptor trigger zone contributes to its antiemetic effects.2 Since the introduction of droperidol in 1967, it has been widely used by emergency physicians, psychiatrists, and anesthesiologists globally.1
Despite its therapeutic efficacy, use of droperidol has been tempered by concerns regarding its cardiovascular safety profile, specifically its potential to prolong the QT interval and precipitate cardiac arrhythmias. In 2001, the FDA placed a boxed warning on droperidol that mandated electrocardiogram (EKG) monitoring before and after treatment. This requirement has led to a widespread decrease in use, and the FDA decision sparked significant controversy among clinicians, with many organizations arguing that the evidence did not support this mandate.1
Further review of the cases cited by the FDA revealed that there were 277 reported cases of droperidol-related adverse events (AEs), but many of these cases were duplicates and occurred outside the US.3 Additionally, the doses of droperidol used in these cases were significantly higher than the typical doses used in the emergency department (ED), ranging from 25 to 250 mg.4 Typical doses for PONV range from 0.625 to 2.5 mg intravenous (IV) or intramuscular (IM). Recommended doses for agitation typically range from 2.5 to 10 mg IV and 5 to 10 mg IM.5
There has been growing interest in reevaluating the risk-benefit profile of droperidol in the ED. Since the original decision by the FDA, multiple publications have challenged the idea that droperidol has significantly higher risks associated with its use. The 2014 review by the Clinical Guidelines Committee of the American Academy of Emergency Medicine did not find evidence that low-dose droperidol (< 2.5 is unsafe for use in the ED.6 A retrospective cohort study from 2020 found no fatalities in 5784 patients. Furthermore, a prospective observational study of 1009 patients at 6 EDs who received high-dose droperidol (≤ 20.0 mg) found no evidence of increased risk for QT prolongation.7 The evidence supports the safety of droperidol for use in prehospital and hospital settings as well as in pediatric, adult, and geriatric populations.8-14 Droperidol was eventually reintroduced in 2019, which led to increased use.
The US Department of Veterans Affairs (VA) formulary has limited options (eg, haloperidol and olanzapine) that have robust evidence supporting their use to treat aggression or psychosis-related agitation. Ziprasidone injections are not on the formulary and require authorization for use, which may delay patient care and pose a safety risk. In 2021, VA Greater Los Angeles Healthcare System (VAGLAHS) received Pharmacy and Therapeutics Committee approval to use droperidol in the ED for agitation or nausea and vomiting. The purpose of this study was to evaluate safety outcomes for patients prescribed droperidol and the need for rescue medications (ie, effectiveness) in the VAGLAHS ED.
Methods
This retrospective chart review analyzed patients administered droperidol in the VAGLAHS ED from February 1, 2021, through April 30, 2023. A list of patients who had droperidol ordered in the VAGLAHS ED was obtained from the Veterans Health Information Systems and Technology Architecture. Charts were reviewed using the Computerized Patient Record System to confirm droperidol administration. Nurse documentation was reviewed to confirm the time, dose, and route of administration. In addition, droperidol dosages were categorized as < 5 mg, 5 to 10 mg, and > 10 mg to review outcomes based on the total amount administered to each patient.
Patients included in the study received droperidol in the ED within the study period, were aged ≥ 18 years, and received droperidol for acute agitation or antiemesis. Patients were excluded if they received droperidol for an indication other than agitation or antiemesis.
The study team reviewed the list of patients and audited the collected data. Reviewers were trained on the study protocols and variables identified. The following data were collected: patient demographics (age, sex, race, height, weight, allergies), Charlson Comorbidity Index (CCI) conditions, cardiac comorbidities, laboratory values at admission, basic metabolic panels, liver function tests, droperidol use (doses, indications, and documentation of safety), concomitant medications ordered with the initial droperidol order, AEs (arrhythmias, extrapyramidal symptoms [EPS], respiratory depression, mortality), medications used within 60 minutes of droperidol administration (rescue medications), other medications used within 24 hours after droperidol administration, and EKG/QTc (corrected QT interval) intervals. The data reviewed and recorded were from the date of the initial patient ED visit.
Outcomes
The primary outcome was all-cause mortality within 24 hours after droperidol administration. This outcome was measured in all patients included in this study. Secondary outcomes included rescue medications needed after droperidol administration, incidence of QT prolongation, incidence of EPS (defined as akathisia, dystonia, parkinsonism, or tardive dyskinesia), and incidence of respiratory depression. Clinically significant QTc was defined as an interval of ≥ 500 ms with incidence of arrhythmias, code blues, or intubations. Baseline risk factors for QTc prolongation were taken into consideration including electrolyte abnormalities, concomitant QT-prolonging medications, CCI score, and cardiac comorbidities. Incidence of EPS was counted if patients received medications such as diphenhydramine or benztropine after droperidol administration in addition to documentation of EPS signs and symptoms. Incidences of EPS findings were reviewed by emergency department physicians to confirm the diagnosis.
Safety was assessed by quantifying mortality rates 24 hours after droperidol administration along with incidence of AEs associated with droperidol use including QT prolongation, EPS, and respiratory depression.
The necessity of rescue medication use was assessed by nursing documentation, additional medications ordered, and/or no additional medications required for agitation within 60 minutes of droperidol administration. Sixty minutes was the chosen timeframe given that the onset of droperidol action is between 3 and 10 minutes and peaks in about 30 minutes. Medications that were considered rescue medications included diphenhydramine < 25 mg, diphenhydramine 25 to 50 mg, lorazepam < 1 mg, lorazepam 1 to 2 mg, diphenhydramine < 25 mg and lorazepam < 1 mg, diphenhydramine < 25 mg and lorazepam 1 to 2 mg, diphenhydramine 25 to 50 mg and lorazepam 1 to 2 mg, and other medications, the names and doses of which were manually documented by investigators.
Statistical Analysis
For all variables in the study, descriptive analysis was used to categorize findings. Microsoft Excel was used to calculate means, frequency counts, percentages, and categorize data.
Results
Between February 1, 2021, and April 16, 2023, 214 patients received droperidol in the VAGLAHS ED, and 207 patients were included in the study. Seven patients did not receive droperidol for the indications included (acute agitation or antiemesis). Most of the study population (89.4%) was male, and the mean age was 51.0 years. The mean CCI was 1.6. In the study, 183 (88.4%) patients received droperidol for agitation and 24 (11.6%) for nausea and vomiting (Table 1).

Primary Outcome
No deaths were observed in a 24-hour period after droperidol administration among the 207 patients included in the study. There were also no arrhythmias, code blues, or intubations observed with the administration of droperidol (Table 2).

Secondary Outcomes
A total of 144 patients (69.6%) received droperidol alone to resolve agitation or nausea and vomiting. In the remaining population, 63 (30.4%) patients were given medications concomitantly with droperidol.
Fifteen patients (7.2%) required rescue medications that were administered within 60 minutes of droperidol administration. Rescue medications were required for 7 patients (4.9%) who initially received droperidol alone compared with 8 patients (12.7%) who were administered concomitant medications with droperidol (Figure).

Extrapyramidal Symptoms
EPS occurred in 2 patients (1.0%). There was 1 incidence of tardive dyskinesia (TD) in which the patient received droperidol 2.5 mg IM for emesis. TD was resolved with diphenhydramine 50 mg. A second patient who experienced dystonia received droperidol 10 mg IM for agitation. Dystonia was resolved with benztropine 2 mg. Both patients had a CCI of 0, no cardiac comorbidities, and laboratory test results were within reference ranges. The second patient received olanzapine within 24 hours of droperidol administration; however, it was after the EPS event.
QTc Prolongation
Baseline EKGs (within 6 months prior to ED visit) were available for 102 patients (49.3%). Nine patients (8.8%) had a reported baseline QTc of ≥ 500 ms (Table 3). Of these patients, 6 had a repeat EKG and 5 had a repeat QTc < 500 ms. One patient had a baseline and repeated QTc of 512 ms with essentially no change after droperidol administration. Only 1 patient was on a potentially QTc-prolonging medication at home. None of the patients with baseline QTc > 500 ms experienced arrhythmias after droperidol administration.

We found that 59 patients (28.5%) had EKGs performed within 24 hours after droperidol administration. Five patients had documented QTc ≥ 500 ms, but no arrhythmias were observed in a 24-hour period. Table 4 describes the additional medications administered after the 60-minute window but within 24 hours after droperidol administration. Quetiapine 300 mg and metoclopramide 5 mg were the only medications documented that can potentially increase QTc. Patient adherence to home medications and the timing of the last dose prior to ED visit were unknown. However, no arrhythmias were noted in these patients with QTc changes. No patients experienced respiratory depression within 24 hours of droperidol administration.

Older Adult Patients
Thirty-eight patients were aged ≥ 65 years with a mean age of 74.2 years. Thirty-four patients (89.5%) received droperidol for agitation and 4 (10.6%) for nausea and vomiting. Only 21 patients had a baseline EKG, and 4 had QTc ≤ 500 ms. At 24 hours, EKGs were performed for 18 patients and 3 had a QTc ≤ 500 ms. No mortality or arrhythmias were reported and there were no incidences of rescue medications, EPS, or respiratory depression.
Discussion
The study included 207 patients who received droperidol for either agitation or nausea/vomiting in the VAGLAHS ED. No mortality occurred within 24 hours of droperidol administration, which is consistent with recent studies.8-14
Furthermore, 59 patients (28.5%) had an EKG performed within 24 hours of droperidol administration; 5 patients had documented QTc ≥ 500 ms. Only 3 of the patients with prolonged QTc had baseline readings for comparison. Only 2 patients had an increase in QTc interval. No arrhythmias were observed; however, the effects of observing QTc prolongation were limited due to the lack of post-EKG readings following droperidol administration. Because of the retrospective nature of the study, neither standardization of EKG at baseline nor 24-hour postadministration were possible. The study found that droperidol was effective with only 15 patients (7.3%) requiring rescue medications. In the patients who were given medications concomitantly with droperidol, it was not possible to conclude whether the patients would have required rescue medications to resolve their agitation or nausea/vomiting. Administration of concomitant medications with droperidol may be attributed to practice patterns associated with haloperidol use, which is frequently administered with concomitant medications such as diphenhydramine and/or a benzodiazepine.
AEs were rare with no documentation of respiratory depression and 2 cases (1.0%) of EPS. Both incidences of EPS resolved with diphenhydramine or benztropine. However, given the reliance on nursing documentation to capture AEs, the number of events may have been underreported.
Limitations
Standardization of dosing was a limiting factor that could affect the need for rescue medications. Another limitation was reliance on nursing reports of resolution of symptoms and comfort with agitated patients. Given the retrospective design and small sample size, this study may not have captured all potential AEs. However, the doses administered within this study population were consistent with what was expected based on other studies.8-14
Conclusions
Droperidol, an antipsychotic, is currently approved for PONV, but is also used off-label for agitation. This study found no fatalities among patients who received droperidol in the ED. The findings suggest that droperidol used for agitation and as an antiemetic, despite its FDA boxed warning, appears to be safe and showed no evidence of mortality, arrhythmias, code blues, or intubations despite the lack of postdose EKG monitoring. Among the 38 patients aged ≥ 65 years, the use of droperidol revealed no increased risks. It should be noted that droperidol appeared safe and few patients required rescue medications within this study population.
Droperidol is a butyrophenone antipsychotic approved by the US Food and Drug Administration (FDA) for use in postoperative nausea and vomiting (PONV). Off-label, it has also been utilized for its sedative, anxiolytic, and analgesic properties.1 While its exact mechanism of action remains elusive, it is believed that binding to postsynaptic γ-aminobutyric acid receptors induces anxiolysis and sedation, while dopaminergic activity in the chemoreceptor trigger zone contributes to its antiemetic effects.2 Since the introduction of droperidol in 1967, it has been widely used by emergency physicians, psychiatrists, and anesthesiologists globally.1
Despite its therapeutic efficacy, use of droperidol has been tempered by concerns regarding its cardiovascular safety profile, specifically its potential to prolong the QT interval and precipitate cardiac arrhythmias. In 2001, the FDA placed a boxed warning on droperidol that mandated electrocardiogram (EKG) monitoring before and after treatment. This requirement has led to a widespread decrease in use, and the FDA decision sparked significant controversy among clinicians, with many organizations arguing that the evidence did not support this mandate.1
Further review of the cases cited by the FDA revealed that there were 277 reported cases of droperidol-related adverse events (AEs), but many of these cases were duplicates and occurred outside the US.3 Additionally, the doses of droperidol used in these cases were significantly higher than the typical doses used in the emergency department (ED), ranging from 25 to 250 mg.4 Typical doses for PONV range from 0.625 to 2.5 mg intravenous (IV) or intramuscular (IM). Recommended doses for agitation typically range from 2.5 to 10 mg IV and 5 to 10 mg IM.5
There has been growing interest in reevaluating the risk-benefit profile of droperidol in the ED. Since the original decision by the FDA, multiple publications have challenged the idea that droperidol has significantly higher risks associated with its use. The 2014 review by the Clinical Guidelines Committee of the American Academy of Emergency Medicine did not find evidence that low-dose droperidol (< 2.5 is unsafe for use in the ED.6 A retrospective cohort study from 2020 found no fatalities in 5784 patients. Furthermore, a prospective observational study of 1009 patients at 6 EDs who received high-dose droperidol (≤ 20.0 mg) found no evidence of increased risk for QT prolongation.7 The evidence supports the safety of droperidol for use in prehospital and hospital settings as well as in pediatric, adult, and geriatric populations.8-14 Droperidol was eventually reintroduced in 2019, which led to increased use.
The US Department of Veterans Affairs (VA) formulary has limited options (eg, haloperidol and olanzapine) that have robust evidence supporting their use to treat aggression or psychosis-related agitation. Ziprasidone injections are not on the formulary and require authorization for use, which may delay patient care and pose a safety risk. In 2021, VA Greater Los Angeles Healthcare System (VAGLAHS) received Pharmacy and Therapeutics Committee approval to use droperidol in the ED for agitation or nausea and vomiting. The purpose of this study was to evaluate safety outcomes for patients prescribed droperidol and the need for rescue medications (ie, effectiveness) in the VAGLAHS ED.
Methods
This retrospective chart review analyzed patients administered droperidol in the VAGLAHS ED from February 1, 2021, through April 30, 2023. A list of patients who had droperidol ordered in the VAGLAHS ED was obtained from the Veterans Health Information Systems and Technology Architecture. Charts were reviewed using the Computerized Patient Record System to confirm droperidol administration. Nurse documentation was reviewed to confirm the time, dose, and route of administration. In addition, droperidol dosages were categorized as < 5 mg, 5 to 10 mg, and > 10 mg to review outcomes based on the total amount administered to each patient.
Patients included in the study received droperidol in the ED within the study period, were aged ≥ 18 years, and received droperidol for acute agitation or antiemesis. Patients were excluded if they received droperidol for an indication other than agitation or antiemesis.
The study team reviewed the list of patients and audited the collected data. Reviewers were trained on the study protocols and variables identified. The following data were collected: patient demographics (age, sex, race, height, weight, allergies), Charlson Comorbidity Index (CCI) conditions, cardiac comorbidities, laboratory values at admission, basic metabolic panels, liver function tests, droperidol use (doses, indications, and documentation of safety), concomitant medications ordered with the initial droperidol order, AEs (arrhythmias, extrapyramidal symptoms [EPS], respiratory depression, mortality), medications used within 60 minutes of droperidol administration (rescue medications), other medications used within 24 hours after droperidol administration, and EKG/QTc (corrected QT interval) intervals. The data reviewed and recorded were from the date of the initial patient ED visit.
Outcomes
The primary outcome was all-cause mortality within 24 hours after droperidol administration. This outcome was measured in all patients included in this study. Secondary outcomes included rescue medications needed after droperidol administration, incidence of QT prolongation, incidence of EPS (defined as akathisia, dystonia, parkinsonism, or tardive dyskinesia), and incidence of respiratory depression. Clinically significant QTc was defined as an interval of ≥ 500 ms with incidence of arrhythmias, code blues, or intubations. Baseline risk factors for QTc prolongation were taken into consideration including electrolyte abnormalities, concomitant QT-prolonging medications, CCI score, and cardiac comorbidities. Incidence of EPS was counted if patients received medications such as diphenhydramine or benztropine after droperidol administration in addition to documentation of EPS signs and symptoms. Incidences of EPS findings were reviewed by emergency department physicians to confirm the diagnosis.
Safety was assessed by quantifying mortality rates 24 hours after droperidol administration along with incidence of AEs associated with droperidol use including QT prolongation, EPS, and respiratory depression.
The necessity of rescue medication use was assessed by nursing documentation, additional medications ordered, and/or no additional medications required for agitation within 60 minutes of droperidol administration. Sixty minutes was the chosen timeframe given that the onset of droperidol action is between 3 and 10 minutes and peaks in about 30 minutes. Medications that were considered rescue medications included diphenhydramine < 25 mg, diphenhydramine 25 to 50 mg, lorazepam < 1 mg, lorazepam 1 to 2 mg, diphenhydramine < 25 mg and lorazepam < 1 mg, diphenhydramine < 25 mg and lorazepam 1 to 2 mg, diphenhydramine 25 to 50 mg and lorazepam 1 to 2 mg, and other medications, the names and doses of which were manually documented by investigators.
Statistical Analysis
For all variables in the study, descriptive analysis was used to categorize findings. Microsoft Excel was used to calculate means, frequency counts, percentages, and categorize data.
Results
Between February 1, 2021, and April 16, 2023, 214 patients received droperidol in the VAGLAHS ED, and 207 patients were included in the study. Seven patients did not receive droperidol for the indications included (acute agitation or antiemesis). Most of the study population (89.4%) was male, and the mean age was 51.0 years. The mean CCI was 1.6. In the study, 183 (88.4%) patients received droperidol for agitation and 24 (11.6%) for nausea and vomiting (Table 1).

Primary Outcome
No deaths were observed in a 24-hour period after droperidol administration among the 207 patients included in the study. There were also no arrhythmias, code blues, or intubations observed with the administration of droperidol (Table 2).

Secondary Outcomes
A total of 144 patients (69.6%) received droperidol alone to resolve agitation or nausea and vomiting. In the remaining population, 63 (30.4%) patients were given medications concomitantly with droperidol.
Fifteen patients (7.2%) required rescue medications that were administered within 60 minutes of droperidol administration. Rescue medications were required for 7 patients (4.9%) who initially received droperidol alone compared with 8 patients (12.7%) who were administered concomitant medications with droperidol (Figure).

Extrapyramidal Symptoms
EPS occurred in 2 patients (1.0%). There was 1 incidence of tardive dyskinesia (TD) in which the patient received droperidol 2.5 mg IM for emesis. TD was resolved with diphenhydramine 50 mg. A second patient who experienced dystonia received droperidol 10 mg IM for agitation. Dystonia was resolved with benztropine 2 mg. Both patients had a CCI of 0, no cardiac comorbidities, and laboratory test results were within reference ranges. The second patient received olanzapine within 24 hours of droperidol administration; however, it was after the EPS event.
QTc Prolongation
Baseline EKGs (within 6 months prior to ED visit) were available for 102 patients (49.3%). Nine patients (8.8%) had a reported baseline QTc of ≥ 500 ms (Table 3). Of these patients, 6 had a repeat EKG and 5 had a repeat QTc < 500 ms. One patient had a baseline and repeated QTc of 512 ms with essentially no change after droperidol administration. Only 1 patient was on a potentially QTc-prolonging medication at home. None of the patients with baseline QTc > 500 ms experienced arrhythmias after droperidol administration.

We found that 59 patients (28.5%) had EKGs performed within 24 hours after droperidol administration. Five patients had documented QTc ≥ 500 ms, but no arrhythmias were observed in a 24-hour period. Table 4 describes the additional medications administered after the 60-minute window but within 24 hours after droperidol administration. Quetiapine 300 mg and metoclopramide 5 mg were the only medications documented that can potentially increase QTc. Patient adherence to home medications and the timing of the last dose prior to ED visit were unknown. However, no arrhythmias were noted in these patients with QTc changes. No patients experienced respiratory depression within 24 hours of droperidol administration.

Older Adult Patients
Thirty-eight patients were aged ≥ 65 years with a mean age of 74.2 years. Thirty-four patients (89.5%) received droperidol for agitation and 4 (10.6%) for nausea and vomiting. Only 21 patients had a baseline EKG, and 4 had QTc ≤ 500 ms. At 24 hours, EKGs were performed for 18 patients and 3 had a QTc ≤ 500 ms. No mortality or arrhythmias were reported and there were no incidences of rescue medications, EPS, or respiratory depression.
Discussion
The study included 207 patients who received droperidol for either agitation or nausea/vomiting in the VAGLAHS ED. No mortality occurred within 24 hours of droperidol administration, which is consistent with recent studies.8-14
Furthermore, 59 patients (28.5%) had an EKG performed within 24 hours of droperidol administration; 5 patients had documented QTc ≥ 500 ms. Only 3 of the patients with prolonged QTc had baseline readings for comparison. Only 2 patients had an increase in QTc interval. No arrhythmias were observed; however, the effects of observing QTc prolongation were limited due to the lack of post-EKG readings following droperidol administration. Because of the retrospective nature of the study, neither standardization of EKG at baseline nor 24-hour postadministration were possible. The study found that droperidol was effective with only 15 patients (7.3%) requiring rescue medications. In the patients who were given medications concomitantly with droperidol, it was not possible to conclude whether the patients would have required rescue medications to resolve their agitation or nausea/vomiting. Administration of concomitant medications with droperidol may be attributed to practice patterns associated with haloperidol use, which is frequently administered with concomitant medications such as diphenhydramine and/or a benzodiazepine.
AEs were rare with no documentation of respiratory depression and 2 cases (1.0%) of EPS. Both incidences of EPS resolved with diphenhydramine or benztropine. However, given the reliance on nursing documentation to capture AEs, the number of events may have been underreported.
Limitations
Standardization of dosing was a limiting factor that could affect the need for rescue medications. Another limitation was reliance on nursing reports of resolution of symptoms and comfort with agitated patients. Given the retrospective design and small sample size, this study may not have captured all potential AEs. However, the doses administered within this study population were consistent with what was expected based on other studies.8-14
Conclusions
Droperidol, an antipsychotic, is currently approved for PONV, but is also used off-label for agitation. This study found no fatalities among patients who received droperidol in the ED. The findings suggest that droperidol used for agitation and as an antiemetic, despite its FDA boxed warning, appears to be safe and showed no evidence of mortality, arrhythmias, code blues, or intubations despite the lack of postdose EKG monitoring. Among the 38 patients aged ≥ 65 years, the use of droperidol revealed no increased risks. It should be noted that droperidol appeared safe and few patients required rescue medications within this study population.
- Perkins J, Ho JD, Vilke GM, DeMers G. American Academy of Emergency Medicine Position Statement: Safety of droperidol use in the emergency department. J Emerg Med. 2015;49:91-97. doi:10.1016/j.jemermed.2014.12.024
- Siegel RB, Motov SM, Marcolini EG. Droperidol use in the emergency department: a clinical review. J Emerg Med. 2023;64:289-294. doi:10.1016/j.jemermed.2022.12.012
- Jackson CW, Sheehan AH, Reddan JG. Evidencebased review of the black-box warning for droperidol. Am J Health Syst Pharm. 2007;64:1174-1186. doi:10.2146/ajhp060505
- Habib AS, Gan TJ. Food and Drug Administration black box warning on the perioperative use of droperidol: a review of the cases. Anesth Analg. 2003;96(5):1377-1379. doi:10.1213/01.ane.0000063923.87560.37
- Droperidol. In: Micromedex (electronic version). IBM Watson Health; 2019. Accessed March 2, 2026. https://www .micromedexsolutions.com
- Gaw CM, Cabrera D, Bellolio F, Mattson AE, Lohse CM, Jeffery MM. Effectiveness and safety of droperidol in a United States emergency department. Am J Emerg Med. 2020;38:1310-1314. doi:10.1016/j.ajem.2019.09.007
- Calver L, Page CB, Downes MA, et al. The safety and effectiveness of droperidol for sedation of acute behavioral disturbance in the emergency department. Ann Emerg Med. 2015;66(3):230-238.e1. doi:10.1016/j.annemergmed.2015.03.016
- Ernst R, Wagstaff H, Smith M, et al. Droperidol administration among emergency department patients with abdominal pain, nausea, and vomiting. Am J Emerg Med. 2024;85:44-47. doi:10.1016/j.ajem.2024.07.060
- Szwak K, Sacchetti A. Droperidol use in pediatric emergency department patients. Pediatr Emerg Care. 2010;26:248-250. doi:10.1097/pec.0b013e3181d6d9f2
- Chase PB, Biros MH. A retrospective review of the use and safety of droperidol in a large, high-risk, inner-city emergency department patient population. Acad Emerg Med. 2002;9:1402-1410. doi:10.1111/j.1553-2712.2002.tb01609.x
- Mattson A, Friend K, Brown CS, Cabrera D. Reintegrating droperidol into emergency medicine practice. Am J Health Syst Pharm. 2020;77(22):1838-1845. doi:10.1093/ajhp/zxaa271
- Cole JB, Stang JL, DeVries PA, Martel ML, Miner JR, Driver BE. A prospective study of intramuscular droperidol or olanzapine for acute agitation in the emergency department: a natural experiment owing to drug shortages. Ann Emerg Med. 2021;78(2):274-286. doi:10.1016/j.annemergmed.2021.01.005
- Page CB, Parker LE, Rashford SJ, et al. Prospective study of the safety and effectiveness of droperidol in elderly patients for pre-hospital acute behavioural disturbance. Emerg Med Australas. 2020;32(5):731-736. doi:10.1111/1742-6723.13496
- Page CB, Parker LE, Rashford SJ, et al. A prospective study of the safety and effectiveness of droperidol inchildren for prehospital acute behavioral disturbance. Prehosp Emerg Care. 2018;23:519-526. doi:10.1080/10903127.2018.1542473
- Perkins J, Ho JD, Vilke GM, DeMers G. American Academy of Emergency Medicine Position Statement: Safety of droperidol use in the emergency department. J Emerg Med. 2015;49:91-97. doi:10.1016/j.jemermed.2014.12.024
- Siegel RB, Motov SM, Marcolini EG. Droperidol use in the emergency department: a clinical review. J Emerg Med. 2023;64:289-294. doi:10.1016/j.jemermed.2022.12.012
- Jackson CW, Sheehan AH, Reddan JG. Evidencebased review of the black-box warning for droperidol. Am J Health Syst Pharm. 2007;64:1174-1186. doi:10.2146/ajhp060505
- Habib AS, Gan TJ. Food and Drug Administration black box warning on the perioperative use of droperidol: a review of the cases. Anesth Analg. 2003;96(5):1377-1379. doi:10.1213/01.ane.0000063923.87560.37
- Droperidol. In: Micromedex (electronic version). IBM Watson Health; 2019. Accessed March 2, 2026. https://www .micromedexsolutions.com
- Gaw CM, Cabrera D, Bellolio F, Mattson AE, Lohse CM, Jeffery MM. Effectiveness and safety of droperidol in a United States emergency department. Am J Emerg Med. 2020;38:1310-1314. doi:10.1016/j.ajem.2019.09.007
- Calver L, Page CB, Downes MA, et al. The safety and effectiveness of droperidol for sedation of acute behavioral disturbance in the emergency department. Ann Emerg Med. 2015;66(3):230-238.e1. doi:10.1016/j.annemergmed.2015.03.016
- Ernst R, Wagstaff H, Smith M, et al. Droperidol administration among emergency department patients with abdominal pain, nausea, and vomiting. Am J Emerg Med. 2024;85:44-47. doi:10.1016/j.ajem.2024.07.060
- Szwak K, Sacchetti A. Droperidol use in pediatric emergency department patients. Pediatr Emerg Care. 2010;26:248-250. doi:10.1097/pec.0b013e3181d6d9f2
- Chase PB, Biros MH. A retrospective review of the use and safety of droperidol in a large, high-risk, inner-city emergency department patient population. Acad Emerg Med. 2002;9:1402-1410. doi:10.1111/j.1553-2712.2002.tb01609.x
- Mattson A, Friend K, Brown CS, Cabrera D. Reintegrating droperidol into emergency medicine practice. Am J Health Syst Pharm. 2020;77(22):1838-1845. doi:10.1093/ajhp/zxaa271
- Cole JB, Stang JL, DeVries PA, Martel ML, Miner JR, Driver BE. A prospective study of intramuscular droperidol or olanzapine for acute agitation in the emergency department: a natural experiment owing to drug shortages. Ann Emerg Med. 2021;78(2):274-286. doi:10.1016/j.annemergmed.2021.01.005
- Page CB, Parker LE, Rashford SJ, et al. Prospective study of the safety and effectiveness of droperidol in elderly patients for pre-hospital acute behavioural disturbance. Emerg Med Australas. 2020;32(5):731-736. doi:10.1111/1742-6723.13496
- Page CB, Parker LE, Rashford SJ, et al. A prospective study of the safety and effectiveness of droperidol inchildren for prehospital acute behavioral disturbance. Prehosp Emerg Care. 2018;23:519-526. doi:10.1080/10903127.2018.1542473
Effectiveness and Safety of Droperidol Use in the VA Greater Los Angeles Healthcare System Emergency Department
Effectiveness and Safety of Droperidol Use in the VA Greater Los Angeles Healthcare System Emergency Department
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
Calcitonin gene-related peptide (CGRP) is a neuropeptide that plays a key role in migraine pathophysiology by promoting the dilation of cerebral blood vessels and transmitting pain signals.1 CGRP has generated interest for the prevention and acute treatment of migraine. Since 2018, 8 novel CGRP-targeting therapies have been approved by the US Food and Drug Administration (FDA) for the management of migraines.2,3 For migraine prevention, there are 4 injectable monoclonal antibodies (mAbs) directed against the CGRP receptor (erenumab) or the CGRP ligand (fremanezumab, galcanezumab, and eptinezumab). There are also 2 oral small-molecule CGRP receptor antagonists, termed gepants, that also are approved for migraine prevention (atogepant and rimegepant). Three gepants are approved for acute migraine treatment and are administered orally (rimegepant and ubrogepant) or intranasally (zavegepant) (Table 1).
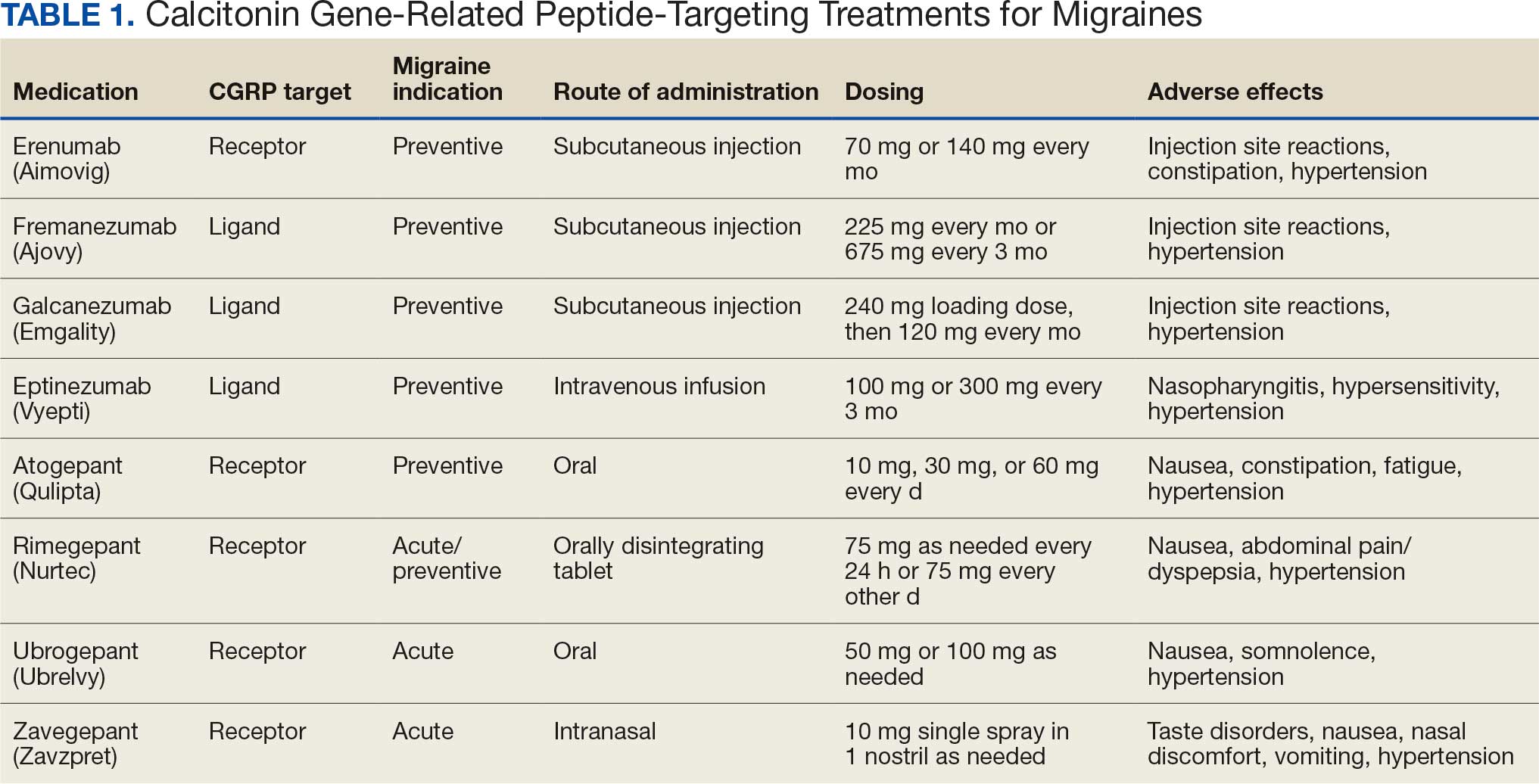
CGRP-targeting therapies have received attention for their role in vasodilation within the cerebral, coronary, and renal vasculature.4 CGRP-mediated vasodilatory effects cause systemic regulation of blood pressure (BP) and play a protective role in hypertension.2 Some studies, particularly with erenumab, have shown that the inhibitory role of the agent leads to an increase in BP, as well as gastrointestinal issues such as constipation.2,5 The FDA recently updated monitoring recommendations for all CGRP-targeting therapies to include the potential for BP elevations and hypertension. Outside of this, there is no definitive evidence linking dual CGRP-targeted therapy to higher cardiovascular or gastrointestinal risks and prescribing information does not carry contraindications.6
In a 2021 consensus statement, the American Headache Society (AHS) recommended CGRP-targeting therapies for migraine prevention after inability to tolerate or inadequate response to an 8-week trial of ≥ 2 drug classes including antihypertensives, antiseizure medications, antidepressants, and onabotulinumtoxinA.7 For acute treatment, AHS recommended gepant use after contraindication to or inadequate response to ≥ 2 triptans. Guidance on combination CGRP-targeting therapies for both prevention and acute treatment was not provided.7 More recently, the AHS published a position statement noting substantial efficacy and safety data for CGRP-targeting therapies and suggested its consideration as a first-line option for migraine prevention, though use for acute treatment or combination CGRP-targeting therapies for both prevention and acute treatment were not addressed.8
The International Headache Society guidelines for the acute treatment of migraines recommend nonopioid analgesics as first-line therapy for mild migraine attacks. For moderate to severe attacks, triptans with or without a nonopioid analgesic were recommended as first-line therapy, prior to consideration of CGRP-targeted therapy.9 The increased use of this new drug class has also led to combination use of CGRP-targeting therapies for migraine prevention and acute treatment as seen in clinical practice and reflected by some case reports, case series, and small studies describing such use.10-14 In light of the similar mechanism of action of these therapies and the physiologic role of CGRP, there have been calls for safety evaluation.15
To our knowledge, no studies have evaluated dual CGRP-targeting regimens for migraine in the veteran population. In 2023, the US Department of Veterans Affairs (VA) and US Department of Defense (DoD) updated their clinical practice guidelines for the management of headache.3 For migraine prevention, the VA/DoD guidelines include a strong recommendation for the use of erenumab, fremanezumab, and galcanezumab; a weak recommendation for the use of atogepant; and a recommendation neither for nor against the use of rimegepant. For acute treatment, the guidelines assign a weak recommendation for the use of rimegepant and ubrogepant. Combination use was not addressed.3
Prior to the VA/DoD guidelines, the Veterans Health Administration restricted the dual use of CGRP-targeting therapies for both preventive and acute migraine treatment. However, the VA Pharmacy Benefit Management Service removed the restriction in the Criteria for Use documents, allowing broader access to these medications for veterans.16-22 This change permits the use of CGRP-targeting drugs for both acute and preventive migraine treatment after initial data reflecting real-world case reports and open-label studies suggested possible efficacy without a clear safety concern.11,12 This study aims to fill the gap in the literature by evaluating the safety, efficacy, and overall outcomes of combination CGRP-targeting treatment for migraine prevention and acute treatment in a veteran population.
Methods
This single-center, retrospective, medication use evaluation at the Ralph H. Johnson VA Medical Center (RHJVAMC) was reviewed by the RHJVAMC Research and Development Committee and Quality Improvement Program Evaluation Self Certification Tool, which both determined that institutional review board approval was not required because it was considered part of routine care and quality improvement. Computerized Patient Record System (CPRS) data were reviewed between April 1, 2023 (after the Criteria for Use for CGRP-targeting therapies was updated), through January 31, 2025. Patients were included if they had a confirmed diagnosis of migraine using the International Classification of Headache Disorders, 3rd edition criteria and had concomitant active prescriptions for both a preventive and acute treatment CGRP-targeting agent during the project period.23 Only patients receiving care from the RHJVAMC neurology department were included.
The primary objective was to assess the safety of dual CGRP-targeting therapies for migraine treatment. Key safety endpoints included effects on liver function, kidney function, and BP. Safety outcomes were graded using Common Terminology Criteria for Adverse Events.24 Changes in liver function were categorized as grade 1, 2, or 3 elevations: grade 1 (aspartate aminotransferase [AST]/alanine aminotransferase [ALT] up to 3x the upper limit of normal [ULN] or bilirubin > 1.5 x ULN); grade 2 (AST/ALT 3-6 x ULN or bilirubin 1.5-3 x ULN); and grade 3 (AST/ALT 5-10 x ULN or bilirubin 3-10 x ULN). Kidney function changes were assessed by serum creatinine levels using a similar grading system: Grade 1 (≤ 1.5 x ULN); grade 2 (1.5-3 x baseline of normal); and grade 3 (3-6 x ULN or baseline). Changes in BP were monitored from baseline to the time of the first neurology follow-up. Elevations were grouped into 2 categories, defined as BP ≥ 140 mm Hg systolic and/or 90 mm Hg diastolic (category 1) and ≥ 160 mm Hg systolic and/or 100 mm Hg diastolic (category 2). Neurology documentation was also reviewed in CPRS for individual patient-reported adverse effects (AEs). Safety endpoints were tracked for any occurrence during the project period.
The secondary objective was to describe the patient-reported efficacy of adding a gepant for acute migraine treatment to existing CGRP-targeting therapies for migraine prevention, in those patients who were stable for ≥ 12 weeks on the preventive therapy. Neurology documentation of headache characteristics, including headache severity as rated on a numerical pain score from 0 (no pain) to 10 (worst pain), and duration of headaches (in hours) were recorded during the project period. Changes in headache characteristics were tracked from baseline (ie, the neurology visit when the gepant was first requested) to the first neurology follow-up within 6 months of initiating gepant for acute treatment. If ranges were provided within documentation, a mean was calculated and used for data collection. Neurology documentation was also reviewed for any patient report of overall effectiveness with the added gepant, and categorized as symptoms improved, worsened, or did not change based on subjective report. Descriptive statistics were used for data analysis. A 1-sample Wilcoxon signed rank test was performed as an exploratory analysis for change in headache characteristics from baseline to first neurology follow-up within 6 months. Each individual CGRP regimen was counted as a unique data point to adequately describe changes associated with each new medication and/or dose adjustment. Therefore, patients could be included more than once to account for each distinct treatment regimen.
Results
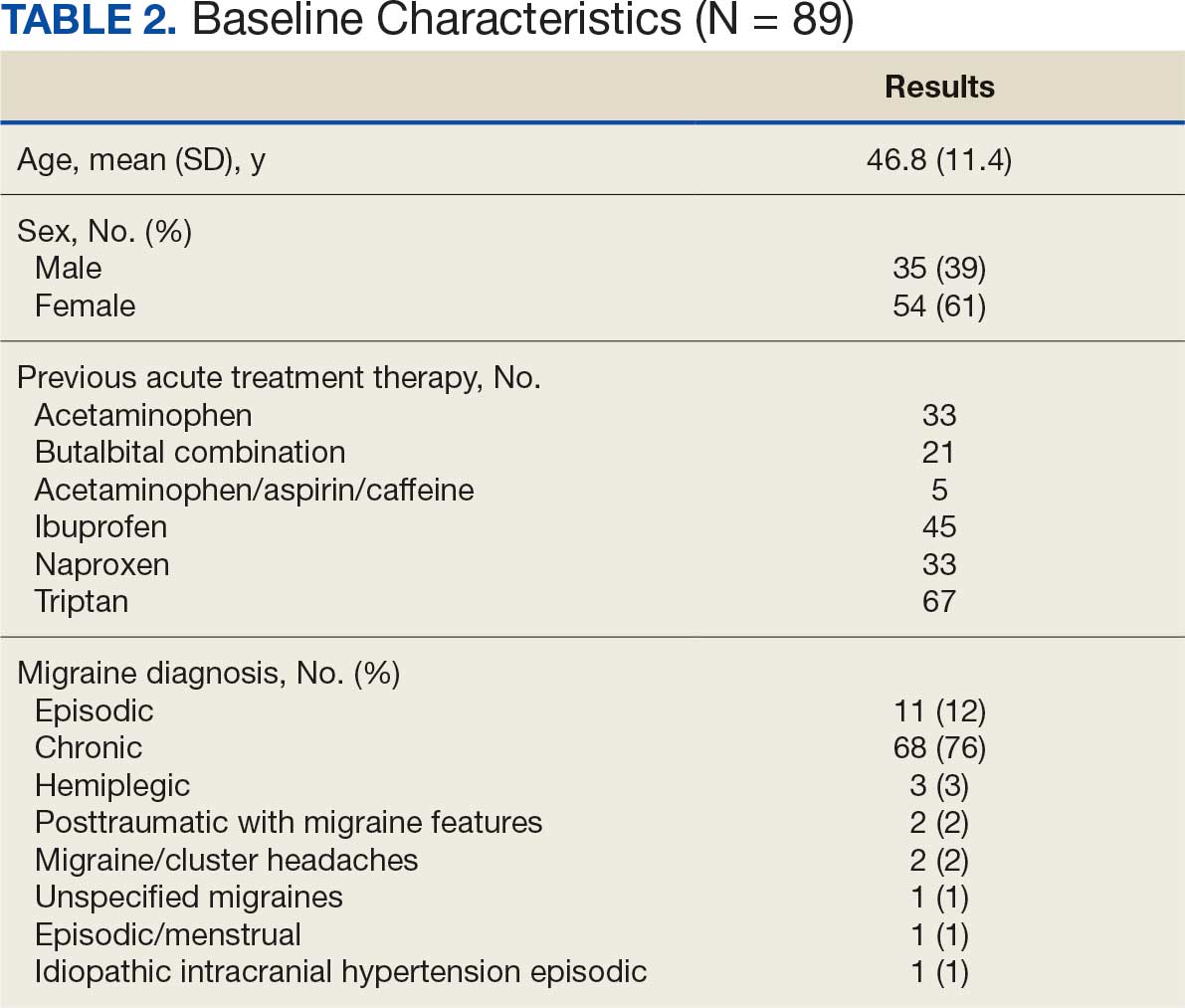
From April 1, 2023, to January 31, 2025, 96 patients were identified with active prescriptions for dual CGRP-targeting therapies. Of the 96 patients, 89 were included in the final analysis; 1 patient lacked a migraine diagnosis and 6 did not have a concomitant dual CGRP-targeted regimen and were excluded. The mean age of patients was 46.8 years and 54 (61%) were female. The most common migraine diagnosis was chronic migraine in 68 patients (76%). Triptans, ibuprofen, and acetaminophen were the most commonly used acute treatment medications (Table 2).
Safety Assessment
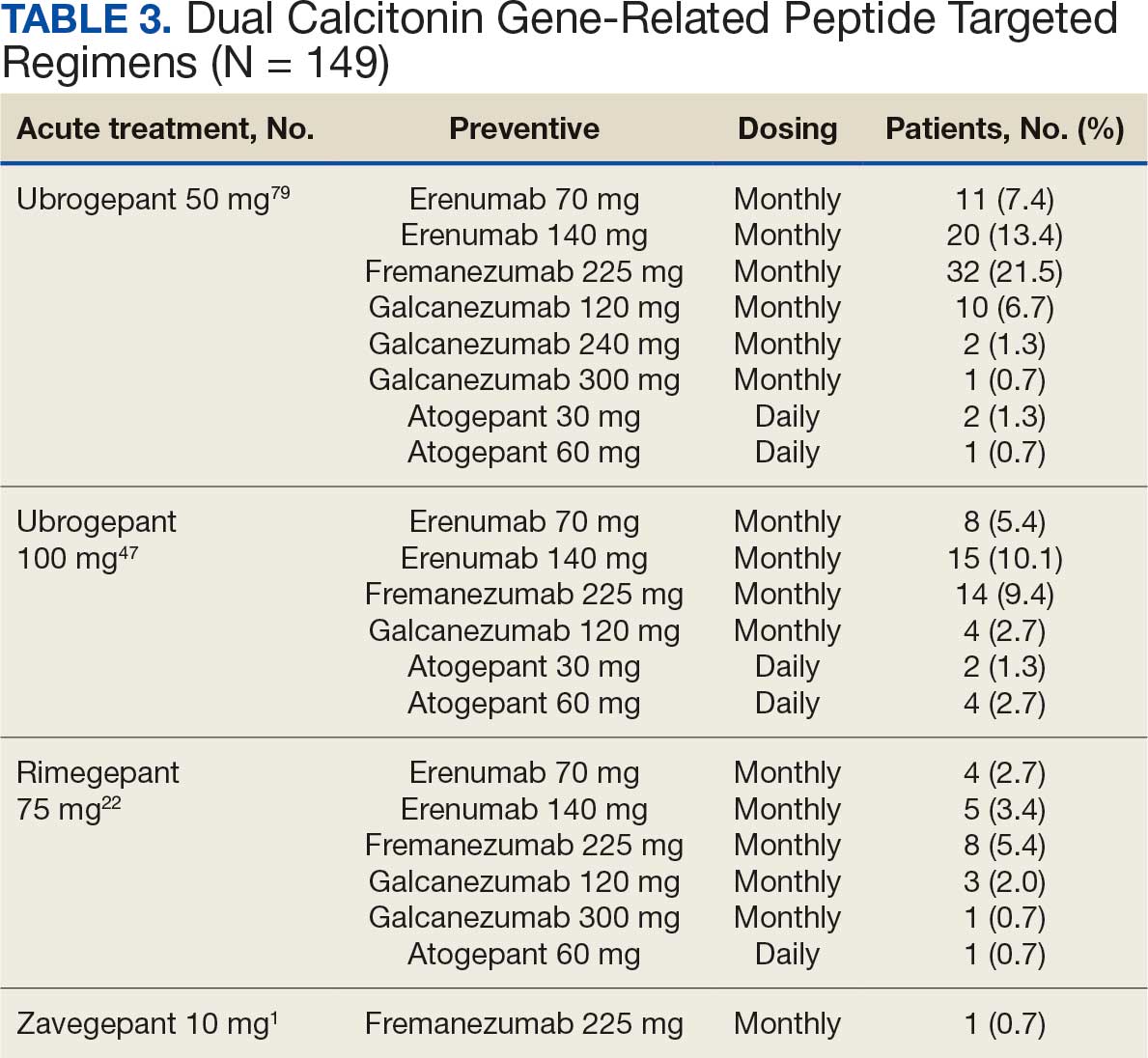
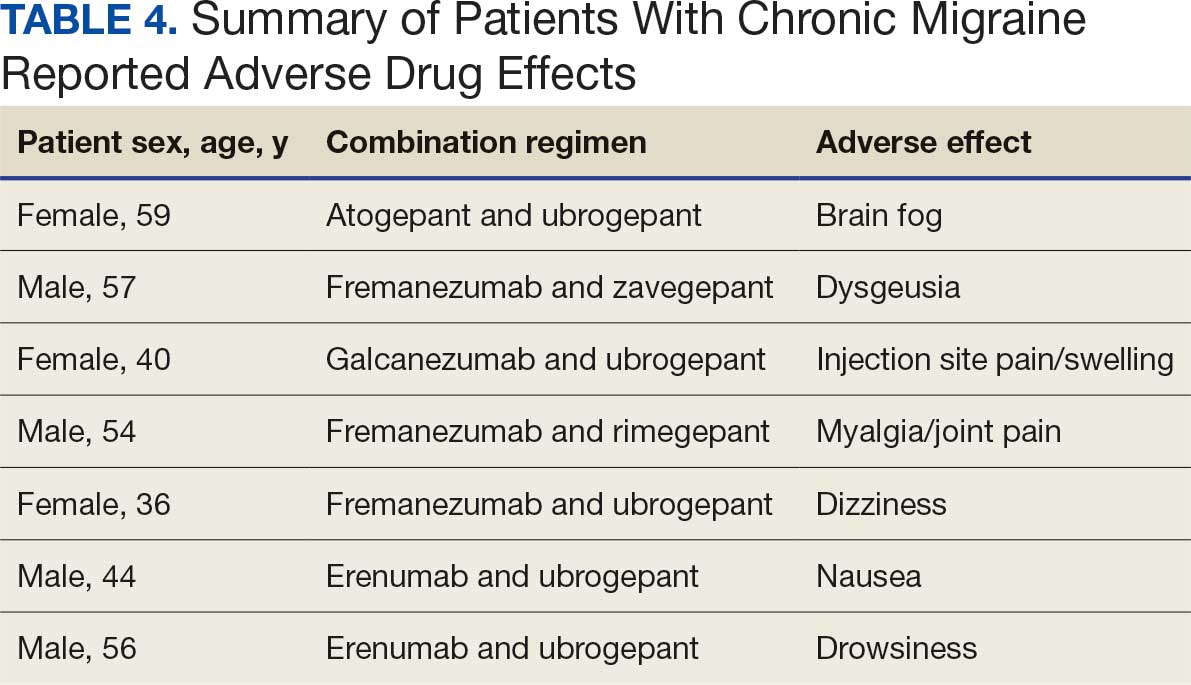
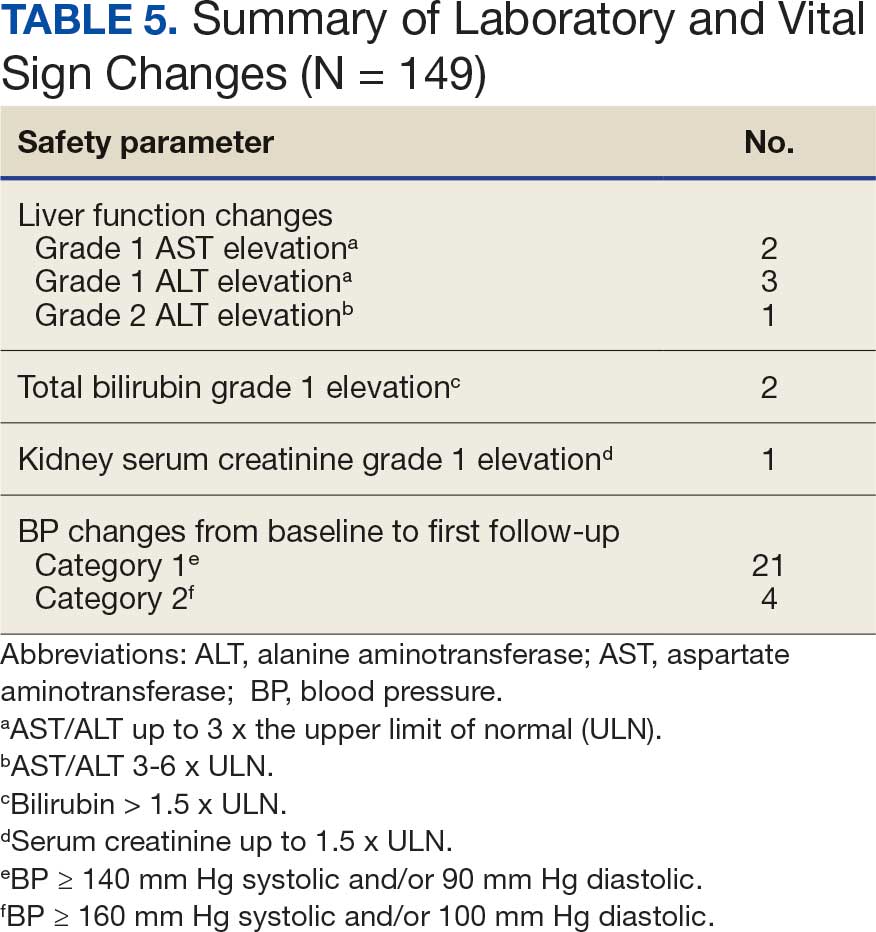
Many of the 89 unique patients trialed > 1 regimen. Thus, for the safety analysis, we analyzed 149 patients on unique dual CGRP-targeting regimens (Table 3). Ubrogepant was used by 126 patients (84.6%) for acute treatment. For preventive therapy, 63 patients (42.3%) used erenumab injections and 55 (36.9%) used fremanezumab injections. Seven patients (4.7%) reported AEs (Table 4). Five of the 7 AEs were noted in the package inserts.25-32 One patient taking both atogepant and ubrogepant reported brain fog that resolved after a dose reduction of atogepant to every other day dosing. A patient taking fremanezumab and rimegepant reported myalgia/joint pain after the first fremanezumab injection, which resolved after a few days and did not recur during the study period.
Nine of 149 patient regimens (6.0%) were associated with changes in liver function tests or serum creatinine, though all but 1 were grade 1 (1 patient had a grade 2 ALT elevation). Twenty-five patients (16.8%) experienced changes in BP, most of which were category 1 elevations. Four patients had systolic or diastolic BP ≥ 160 mm Hg or 100 mm Hg, respectively (Table 5).
Efficacy Assessment
Of the 149 unique dual CGRP regimens, 59 were eligible for the exploratory efficacy analysis. Data were excluded from the efficacy analysis if patients had not been on a stable CGRP preventive migraine regimen for ≥ 12 weeks prior to the addition of a gepant. Fourteen regimens were excluded due to a lack of clear documentation on efficacy, leaving 45 analyzed regimens. Of the 45 regimens, 34 were from unique patients. There was no median change in migraine intensity or duration found in the efficacy analysis (0.0, P = .18, and 0.0, P = .92, respectively). Ten patients on dual CGRP therapy reported that the addition of a gepant for acute treatment improved their symptoms, 20 reported that their symptoms were unchanged and/or worsened, and 29 lacked documentation.
Discussion
This study aimed to describe the safety and efficacy of concomitant CGRP regimens for migraine prevention and acute treatment. To our knowledge, this was the first descriptive study of these agents in a veteran population. The potential for increased AEs with concomitant use of CGRP antagonists is due to the similarities in the mechanism of action between the agents, which both target the same receptor/ligand pathway. Given CGRP activity in both the gastrointestinal and cardiovascular systems, the potential for related AEs is speculative. Patient-reported AEs occurred in 7 of 149 unique treatment regimens reviewed for an incidence rate < 5%. All AEs were nonserious and self-limiting.
Our findings are consistent with available research. A 2024 retrospective, exploratory real-world study evaluating the safety and tolerability of combining CGRP-targeting mAbs with gepants reported findings consistent with our results. This analysis included adult patients treated with ≥ 1 previous anti-CGRP mAb and found that 234 of 516 patients included received a combination of a gepant in addition to a CGRP-targeting mAb. Of these 234 patients, 1.3% reported nonserious AEs.33 Similarly, in a multicenter, open-label, long-term safety study in adults experiencing multiple monthly migraine attacks, a subgroup of 13 participants taking a stable dose of an anti-CGRP mAb also took rimegepant 75 mg as needed for acute treatment for 12 weeks. These patients experienced no serious AEs or any AEs leading to discontinuation.14 A study evaluating the drug-drug interaction, safety, and tolerability of dual therapy (atogepant 60 mg daily and ubrogepant 100 mg every 3 days) in 26 patients found no serious AEs, including no significant changes from baseline in laboratory results, vital signs, or safety-related 12-lead electrocardiogram parameters.15The TANDEM real-world, open-label, prospective study demonstrated similar results. It evaluated the safety and tolerability of concomitant use of ubrogepant and atogepant in patients with episodic migraines and found no increase in AEs when comparing atogepant alone with combination therapy. Twenty-six patients (9.9%) discontinued treatment due to AEs. The most common treatment-related AEs were constipation, nausea, decreased appetite, and fatigue. Efficacy data were also noted to be an exploratory endpoint in the TANDEM study; however, results have not been published.12
Within this safety analysis, new onset gastrointestinal AEs, specifically nausea, only occurred in 1 patient. Hypertension occurred in 25 treatment regimens (16.8%) for 21 unique patients (4 BP elevations occurred in 1 patient on 4 different regimens). However, the retrospective nature of reporting may limit accurate assessment. A closer analysis determined that elevated BP readings correlated with elevated pain scores at the time of the readings, which could have factored into the BP elevations. However, ongoing monitoring is needed due to an increased risk of hypertension, particularly given recent FDA labeling updates for CGRP-targeting therapies including gepants. In light of this, and the overall low incidence of hypertension reported, no new safety concerns were identified.
Limitations
Efficacy data in this project were exploratory. This evaluation did not show a significant difference in migraine intensity or duration after adding a gepant for acute treatment. The study was not powered to detect a significant difference. Limited data exist assessing efficacy outcomes with dual CGRP-targeting treatment regimens. The COURAGE study assessed the real-world effectiveness of ubrogepant and CGRP mAbs with or without the addition of onabotulinumtoxinA. The final analysis of the ubrogepant and CGRP mAb arm included 245 total patients and assessed meaningful migraine pain relief, restoration of normal function after a migraine, and treatment satisfaction. By hour 2, 61.6% of patients reported achieving migraine pain relief, rising to 80.4% by hour 4. Return to normal function occurred in 34.7% at hour 2 and 55.5% by hour 4.13 The long-term safety and efficacy of combining erenumab and rimegepant were described in a case series involving 2 patients. Both patients reported that the concomitant CGRP-targeted therapies were effective and reported no AEs.14
The retrospective design of this study meant that there was potential for limited documentation and introduction of bias into the results. Data were collected at a single VA health care system, and thus, results may not be generalizable to a broader population. However, the study population was consistent with the higher incidence of migraine expected in females in the general population. The sample size was limited, particularly in the exploratory efficacy endpoint assessment.
Limitations were observed due to inconsistent documentation regarding headache characteristics, making it challenging to draw meaningful conclusions from this data set. Additional confounding factors, including polypharmacy, nonadherence to medications, and comorbidities, may have skewed results. For example, while our study design required that the preventive CGRP-targeting medication be stable for 12 weeks for inclusion in further efficacy analysis, other medications commonly used for migraine prevention may have been adjusted (which was not accounted for in this analysis). Given this, more large-scale, placebo-controlled, randomized studies are needed to continue to assess the safety and efficacy of these combination treatment regimens.
Conclusions
Few AEs or safety events were reported with combination CGRP-targeting treatment for acute and preventive treatment of migraine. Those that were identified were considered mild. Efficacy data were limited, and further studies are needed to fully assess outcomes.
- Wattiez AS, Sowers LP, Russo AF. Calcitonin gene-related peptide (CGRP): role in migraine pathophysiology and therapeutic targeting. Expert Opin Ther Targets. 2020;24:91-100. doi:10.1080/14728222.2020.1724285
- Shah T, Bedrin K, Tinsley A. Calcitonin gene relating peptide inhibitors in combination for migraine treatment: a mini-review. Front Pain Res (Lausanne). 2023;4:1130239. doi:10.3389/fpain.2023.1130239
- Department of Veterans Affairs/Department of Defense. VA/DoD clinical practice guideline for management of headache. September 2023. Accessed February 4, 2026. https://www.healthquality.va.gov/guidelines/pain/headache/VA-DoD-CPG-Headache-Full-CPG.pdf
- Russell FA, King R, Smillie SJ, et al. Calcitonin gene-related peptide: physiology and pathophysiology. Physiol Rev. 2014;94:1099-1142. doi:10.1152/physrev.00034.2013
- de Vries Lentsch S, van der Arend BWH, VanDenBrink AM, et al. Blood pressure in patients with migraine treated with monoclonal anti-CGRP (receptor) antibodies: a prospective follow-up study. Neurology. 2022;99:e1897-e1904. doi:10.1212/WNL.0000000000201008
- Favoni V, Giani L, Al-Hassany L, et al. CGRP and migraine from a cardiovascular point of view: what do we expect from blocking CGRP?. J Headache Pain. 2019;20:27. doi:10.1186/s10194-019-0979-y
- Ailani J, Burch RC, Robbins MS, et al. The American Headache Society Consensus Statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61:1021-1039. doi:10.1111/head.14153
- Charles AC, Digre KB, Goadsby PJ, et al. Calcitonin gene-related peptide-targeting therapies are a first-line option for the prevention of migraine: an American Headache Society position statement update. Headache. 2024;64:333-341. doi:10.1111/head.14692
- Puledda F, Sacco S, Diener HC, et al. International Headache Society global practice recommendations for the acute pharmacological treatment of migraine. Cephalalgia. 2024;44:3331024241252666. doi:10.1177/03331024241252666
- Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi:10.1111/head.13930
- Blumenfeld AM, Boinpally R, De Abreu Ferreira R, et al. Phase Ib, open-label, fixed-sequence, drug-drug interaction, safety, and tolerability study between atogepant and ubrogepant in participants with a history of migraine. Headache. 2023;63:322-332. doi:10.1111/head.14433
- Ailani J, Lipton RB, Blumenfeld AM, et al. Safety and tolerability of ubrogepant for the acute treatment of migraine in participants taking atogepant for the preventive treatment of episodic migraine: results from the TANDEM study. Headache. 2025;65:1005-1014. doi:10.1111/head.14871
- Lipton RB, Contreras-De Lama J, Serrano D, et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: results from COURAGE. Neurol Ther. 2024;13:69-83. doi:10.1007/s40120-023-00556-8
- Mullin K, Kudrow D, Croop R, et al. Potential for treatment benefit of small molecule CGRP receptor antagonist plus monoclonal antibody in migraine therapy. Neurology. 2020;94:e2121-e2125. doi:10.1212/WNL.0000000000008944
- Ihara K, Takizawa T, Watanabe N, et al. Potential benefits and possible risks of CGRP-targeted multitherapy in migraine. Expert Opin Drug Metab Toxicol. 2024;20:1-4. doi:10.1080/17425255.2024.2316131
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Ubrogepant (Ubrelvy) criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Ubrogepant_UBRELVY_CFU_Rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for abortive migraine treatment criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_abortive_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_episodic_migraine_prevention_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_chronic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_episodic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Galcanezumab-gnlm (Emgality) for cluster headache criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Galcanezumab_EMGALITY_for_cluster_headache_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Atogepant (Qulipta) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Atogepant_QULIPTA_for_chronic_migraine_prevention_CFU_rev_Jul_2025.pdf
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi:10.1177/0333102417738202
- US Dept of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. November 27, 2017. Accessed March 4, 2026. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf
- Aimovig (erenumab-aooe) injection prescribing information. Amegen Inc. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761077s026lbl.pdf
- Ajovy (fremanezumab-vfrm) injection prescribing information. Teva Pharmaceuticals. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761089s031lbl.pdf
- Vyepti (eptinezumab-jjmr) injection prescribing information. Lundbeck Seattle Biopharmaceuticals. Updated October 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761119s011lbl.pdf
- Emgality (galcanezumab-gnlm) injection prescribing information. Eli Lilly and Company. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761063s010lbl.pdf
- Qulipta (atogepant) tablets prescribing information. AbbVie Inc. Updated September 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215206s013lbl.pdf
- Nurtec ODT (rimegepant) orally disintegrating tablets prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/212728s028lbl.pdf
- Ubrelvy (Ubrogepant) tablets prescribing information. AbbVie Inc. Updated June 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/211765s012lbl.pdf
- Zavzpret (zavegepant) intranasal spray prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/216386s007lbl.pdf
- Alsaadi T, Suliman R, Santos V, et al. Safety and tolerability of combining CGRP monoclonal antibodies with gepants in patients with migraine: a retrospective study. Neurol Ther. 2024;13:465-473. doi:10.1007/s40120-024-00586-w
Calcitonin gene-related peptide (CGRP) is a neuropeptide that plays a key role in migraine pathophysiology by promoting the dilation of cerebral blood vessels and transmitting pain signals.1 CGRP has generated interest for the prevention and acute treatment of migraine. Since 2018, 8 novel CGRP-targeting therapies have been approved by the US Food and Drug Administration (FDA) for the management of migraines.2,3 For migraine prevention, there are 4 injectable monoclonal antibodies (mAbs) directed against the CGRP receptor (erenumab) or the CGRP ligand (fremanezumab, galcanezumab, and eptinezumab). There are also 2 oral small-molecule CGRP receptor antagonists, termed gepants, that also are approved for migraine prevention (atogepant and rimegepant). Three gepants are approved for acute migraine treatment and are administered orally (rimegepant and ubrogepant) or intranasally (zavegepant) (Table 1).

CGRP-targeting therapies have received attention for their role in vasodilation within the cerebral, coronary, and renal vasculature.4 CGRP-mediated vasodilatory effects cause systemic regulation of blood pressure (BP) and play a protective role in hypertension.2 Some studies, particularly with erenumab, have shown that the inhibitory role of the agent leads to an increase in BP, as well as gastrointestinal issues such as constipation.2,5 The FDA recently updated monitoring recommendations for all CGRP-targeting therapies to include the potential for BP elevations and hypertension. Outside of this, there is no definitive evidence linking dual CGRP-targeted therapy to higher cardiovascular or gastrointestinal risks and prescribing information does not carry contraindications.6
In a 2021 consensus statement, the American Headache Society (AHS) recommended CGRP-targeting therapies for migraine prevention after inability to tolerate or inadequate response to an 8-week trial of ≥ 2 drug classes including antihypertensives, antiseizure medications, antidepressants, and onabotulinumtoxinA.7 For acute treatment, AHS recommended gepant use after contraindication to or inadequate response to ≥ 2 triptans. Guidance on combination CGRP-targeting therapies for both prevention and acute treatment was not provided.7 More recently, the AHS published a position statement noting substantial efficacy and safety data for CGRP-targeting therapies and suggested its consideration as a first-line option for migraine prevention, though use for acute treatment or combination CGRP-targeting therapies for both prevention and acute treatment were not addressed.8
The International Headache Society guidelines for the acute treatment of migraines recommend nonopioid analgesics as first-line therapy for mild migraine attacks. For moderate to severe attacks, triptans with or without a nonopioid analgesic were recommended as first-line therapy, prior to consideration of CGRP-targeted therapy.9 The increased use of this new drug class has also led to combination use of CGRP-targeting therapies for migraine prevention and acute treatment as seen in clinical practice and reflected by some case reports, case series, and small studies describing such use.10-14 In light of the similar mechanism of action of these therapies and the physiologic role of CGRP, there have been calls for safety evaluation.15
To our knowledge, no studies have evaluated dual CGRP-targeting regimens for migraine in the veteran population. In 2023, the US Department of Veterans Affairs (VA) and US Department of Defense (DoD) updated their clinical practice guidelines for the management of headache.3 For migraine prevention, the VA/DoD guidelines include a strong recommendation for the use of erenumab, fremanezumab, and galcanezumab; a weak recommendation for the use of atogepant; and a recommendation neither for nor against the use of rimegepant. For acute treatment, the guidelines assign a weak recommendation for the use of rimegepant and ubrogepant. Combination use was not addressed.3
Prior to the VA/DoD guidelines, the Veterans Health Administration restricted the dual use of CGRP-targeting therapies for both preventive and acute migraine treatment. However, the VA Pharmacy Benefit Management Service removed the restriction in the Criteria for Use documents, allowing broader access to these medications for veterans.16-22 This change permits the use of CGRP-targeting drugs for both acute and preventive migraine treatment after initial data reflecting real-world case reports and open-label studies suggested possible efficacy without a clear safety concern.11,12 This study aims to fill the gap in the literature by evaluating the safety, efficacy, and overall outcomes of combination CGRP-targeting treatment for migraine prevention and acute treatment in a veteran population.
Methods
This single-center, retrospective, medication use evaluation at the Ralph H. Johnson VA Medical Center (RHJVAMC) was reviewed by the RHJVAMC Research and Development Committee and Quality Improvement Program Evaluation Self Certification Tool, which both determined that institutional review board approval was not required because it was considered part of routine care and quality improvement. Computerized Patient Record System (CPRS) data were reviewed between April 1, 2023 (after the Criteria for Use for CGRP-targeting therapies was updated), through January 31, 2025. Patients were included if they had a confirmed diagnosis of migraine using the International Classification of Headache Disorders, 3rd edition criteria and had concomitant active prescriptions for both a preventive and acute treatment CGRP-targeting agent during the project period.23 Only patients receiving care from the RHJVAMC neurology department were included.
The primary objective was to assess the safety of dual CGRP-targeting therapies for migraine treatment. Key safety endpoints included effects on liver function, kidney function, and BP. Safety outcomes were graded using Common Terminology Criteria for Adverse Events.24 Changes in liver function were categorized as grade 1, 2, or 3 elevations: grade 1 (aspartate aminotransferase [AST]/alanine aminotransferase [ALT] up to 3x the upper limit of normal [ULN] or bilirubin > 1.5 x ULN); grade 2 (AST/ALT 3-6 x ULN or bilirubin 1.5-3 x ULN); and grade 3 (AST/ALT 5-10 x ULN or bilirubin 3-10 x ULN). Kidney function changes were assessed by serum creatinine levels using a similar grading system: Grade 1 (≤ 1.5 x ULN); grade 2 (1.5-3 x baseline of normal); and grade 3 (3-6 x ULN or baseline). Changes in BP were monitored from baseline to the time of the first neurology follow-up. Elevations were grouped into 2 categories, defined as BP ≥ 140 mm Hg systolic and/or 90 mm Hg diastolic (category 1) and ≥ 160 mm Hg systolic and/or 100 mm Hg diastolic (category 2). Neurology documentation was also reviewed in CPRS for individual patient-reported adverse effects (AEs). Safety endpoints were tracked for any occurrence during the project period.
The secondary objective was to describe the patient-reported efficacy of adding a gepant for acute migraine treatment to existing CGRP-targeting therapies for migraine prevention, in those patients who were stable for ≥ 12 weeks on the preventive therapy. Neurology documentation of headache characteristics, including headache severity as rated on a numerical pain score from 0 (no pain) to 10 (worst pain), and duration of headaches (in hours) were recorded during the project period. Changes in headache characteristics were tracked from baseline (ie, the neurology visit when the gepant was first requested) to the first neurology follow-up within 6 months of initiating gepant for acute treatment. If ranges were provided within documentation, a mean was calculated and used for data collection. Neurology documentation was also reviewed for any patient report of overall effectiveness with the added gepant, and categorized as symptoms improved, worsened, or did not change based on subjective report. Descriptive statistics were used for data analysis. A 1-sample Wilcoxon signed rank test was performed as an exploratory analysis for change in headache characteristics from baseline to first neurology follow-up within 6 months. Each individual CGRP regimen was counted as a unique data point to adequately describe changes associated with each new medication and/or dose adjustment. Therefore, patients could be included more than once to account for each distinct treatment regimen.
Results
From April 1, 2023, to January 31, 2025, 96 patients were identified with active prescriptions for dual CGRP-targeting therapies. Of the 96 patients, 89 were included in the final analysis; 1 patient lacked a migraine diagnosis and 6 did not have a concomitant dual CGRP-targeted regimen and were excluded. The mean age of patients was 46.8 years and 54 (61%) were female. The most common migraine diagnosis was chronic migraine in 68 patients (76%). Triptans, ibuprofen, and acetaminophen were the most commonly used acute treatment medications (Table 2).

Safety Assessment
Many of the 89 unique patients trialed > 1 regimen. Thus, for the safety analysis, we analyzed 149 patients on unique dual CGRP-targeting regimens (Table 3). Ubrogepant was used by 126 patients (84.6%) for acute treatment. For preventive therapy, 63 patients (42.3%) used erenumab injections and 55 (36.9%) used fremanezumab injections. Seven patients (4.7%) reported AEs (Table 4). Five of the 7 AEs were noted in the package inserts.25-32 One patient taking both atogepant and ubrogepant reported brain fog that resolved after a dose reduction of atogepant to every other day dosing. A patient taking fremanezumab and rimegepant reported myalgia/joint pain after the first fremanezumab injection, which resolved after a few days and did not recur during the study period.


Nine of 149 patient regimens (6.0%) were associated with changes in liver function tests or serum creatinine, though all but 1 were grade 1 (1 patient had a grade 2 ALT elevation). Twenty-five patients (16.8%) experienced changes in BP, most of which were category 1 elevations. Four patients had systolic or diastolic BP ≥ 160 mm Hg or 100 mm Hg, respectively (Table 5).

Efficacy Assessment
Of the 149 unique dual CGRP regimens, 59 were eligible for the exploratory efficacy analysis. Data were excluded from the efficacy analysis if patients had not been on a stable CGRP preventive migraine regimen for ≥ 12 weeks prior to the addition of a gepant. Fourteen regimens were excluded due to a lack of clear documentation on efficacy, leaving 45 analyzed regimens. Of the 45 regimens, 34 were from unique patients. There was no median change in migraine intensity or duration found in the efficacy analysis (0.0, P = .18, and 0.0, P = .92, respectively). Ten patients on dual CGRP therapy reported that the addition of a gepant for acute treatment improved their symptoms, 20 reported that their symptoms were unchanged and/or worsened, and 29 lacked documentation.
Discussion
This study aimed to describe the safety and efficacy of concomitant CGRP regimens for migraine prevention and acute treatment. To our knowledge, this was the first descriptive study of these agents in a veteran population. The potential for increased AEs with concomitant use of CGRP antagonists is due to the similarities in the mechanism of action between the agents, which both target the same receptor/ligand pathway. Given CGRP activity in both the gastrointestinal and cardiovascular systems, the potential for related AEs is speculative. Patient-reported AEs occurred in 7 of 149 unique treatment regimens reviewed for an incidence rate < 5%. All AEs were nonserious and self-limiting.
Our findings are consistent with available research. A 2024 retrospective, exploratory real-world study evaluating the safety and tolerability of combining CGRP-targeting mAbs with gepants reported findings consistent with our results. This analysis included adult patients treated with ≥ 1 previous anti-CGRP mAb and found that 234 of 516 patients included received a combination of a gepant in addition to a CGRP-targeting mAb. Of these 234 patients, 1.3% reported nonserious AEs.33 Similarly, in a multicenter, open-label, long-term safety study in adults experiencing multiple monthly migraine attacks, a subgroup of 13 participants taking a stable dose of an anti-CGRP mAb also took rimegepant 75 mg as needed for acute treatment for 12 weeks. These patients experienced no serious AEs or any AEs leading to discontinuation.14 A study evaluating the drug-drug interaction, safety, and tolerability of dual therapy (atogepant 60 mg daily and ubrogepant 100 mg every 3 days) in 26 patients found no serious AEs, including no significant changes from baseline in laboratory results, vital signs, or safety-related 12-lead electrocardiogram parameters.15The TANDEM real-world, open-label, prospective study demonstrated similar results. It evaluated the safety and tolerability of concomitant use of ubrogepant and atogepant in patients with episodic migraines and found no increase in AEs when comparing atogepant alone with combination therapy. Twenty-six patients (9.9%) discontinued treatment due to AEs. The most common treatment-related AEs were constipation, nausea, decreased appetite, and fatigue. Efficacy data were also noted to be an exploratory endpoint in the TANDEM study; however, results have not been published.12
Within this safety analysis, new onset gastrointestinal AEs, specifically nausea, only occurred in 1 patient. Hypertension occurred in 25 treatment regimens (16.8%) for 21 unique patients (4 BP elevations occurred in 1 patient on 4 different regimens). However, the retrospective nature of reporting may limit accurate assessment. A closer analysis determined that elevated BP readings correlated with elevated pain scores at the time of the readings, which could have factored into the BP elevations. However, ongoing monitoring is needed due to an increased risk of hypertension, particularly given recent FDA labeling updates for CGRP-targeting therapies including gepants. In light of this, and the overall low incidence of hypertension reported, no new safety concerns were identified.
Limitations
Efficacy data in this project were exploratory. This evaluation did not show a significant difference in migraine intensity or duration after adding a gepant for acute treatment. The study was not powered to detect a significant difference. Limited data exist assessing efficacy outcomes with dual CGRP-targeting treatment regimens. The COURAGE study assessed the real-world effectiveness of ubrogepant and CGRP mAbs with or without the addition of onabotulinumtoxinA. The final analysis of the ubrogepant and CGRP mAb arm included 245 total patients and assessed meaningful migraine pain relief, restoration of normal function after a migraine, and treatment satisfaction. By hour 2, 61.6% of patients reported achieving migraine pain relief, rising to 80.4% by hour 4. Return to normal function occurred in 34.7% at hour 2 and 55.5% by hour 4.13 The long-term safety and efficacy of combining erenumab and rimegepant were described in a case series involving 2 patients. Both patients reported that the concomitant CGRP-targeted therapies were effective and reported no AEs.14
The retrospective design of this study meant that there was potential for limited documentation and introduction of bias into the results. Data were collected at a single VA health care system, and thus, results may not be generalizable to a broader population. However, the study population was consistent with the higher incidence of migraine expected in females in the general population. The sample size was limited, particularly in the exploratory efficacy endpoint assessment.
Limitations were observed due to inconsistent documentation regarding headache characteristics, making it challenging to draw meaningful conclusions from this data set. Additional confounding factors, including polypharmacy, nonadherence to medications, and comorbidities, may have skewed results. For example, while our study design required that the preventive CGRP-targeting medication be stable for 12 weeks for inclusion in further efficacy analysis, other medications commonly used for migraine prevention may have been adjusted (which was not accounted for in this analysis). Given this, more large-scale, placebo-controlled, randomized studies are needed to continue to assess the safety and efficacy of these combination treatment regimens.
Conclusions
Few AEs or safety events were reported with combination CGRP-targeting treatment for acute and preventive treatment of migraine. Those that were identified were considered mild. Efficacy data were limited, and further studies are needed to fully assess outcomes.
Calcitonin gene-related peptide (CGRP) is a neuropeptide that plays a key role in migraine pathophysiology by promoting the dilation of cerebral blood vessels and transmitting pain signals.1 CGRP has generated interest for the prevention and acute treatment of migraine. Since 2018, 8 novel CGRP-targeting therapies have been approved by the US Food and Drug Administration (FDA) for the management of migraines.2,3 For migraine prevention, there are 4 injectable monoclonal antibodies (mAbs) directed against the CGRP receptor (erenumab) or the CGRP ligand (fremanezumab, galcanezumab, and eptinezumab). There are also 2 oral small-molecule CGRP receptor antagonists, termed gepants, that also are approved for migraine prevention (atogepant and rimegepant). Three gepants are approved for acute migraine treatment and are administered orally (rimegepant and ubrogepant) or intranasally (zavegepant) (Table 1).

CGRP-targeting therapies have received attention for their role in vasodilation within the cerebral, coronary, and renal vasculature.4 CGRP-mediated vasodilatory effects cause systemic regulation of blood pressure (BP) and play a protective role in hypertension.2 Some studies, particularly with erenumab, have shown that the inhibitory role of the agent leads to an increase in BP, as well as gastrointestinal issues such as constipation.2,5 The FDA recently updated monitoring recommendations for all CGRP-targeting therapies to include the potential for BP elevations and hypertension. Outside of this, there is no definitive evidence linking dual CGRP-targeted therapy to higher cardiovascular or gastrointestinal risks and prescribing information does not carry contraindications.6
In a 2021 consensus statement, the American Headache Society (AHS) recommended CGRP-targeting therapies for migraine prevention after inability to tolerate or inadequate response to an 8-week trial of ≥ 2 drug classes including antihypertensives, antiseizure medications, antidepressants, and onabotulinumtoxinA.7 For acute treatment, AHS recommended gepant use after contraindication to or inadequate response to ≥ 2 triptans. Guidance on combination CGRP-targeting therapies for both prevention and acute treatment was not provided.7 More recently, the AHS published a position statement noting substantial efficacy and safety data for CGRP-targeting therapies and suggested its consideration as a first-line option for migraine prevention, though use for acute treatment or combination CGRP-targeting therapies for both prevention and acute treatment were not addressed.8
The International Headache Society guidelines for the acute treatment of migraines recommend nonopioid analgesics as first-line therapy for mild migraine attacks. For moderate to severe attacks, triptans with or without a nonopioid analgesic were recommended as first-line therapy, prior to consideration of CGRP-targeted therapy.9 The increased use of this new drug class has also led to combination use of CGRP-targeting therapies for migraine prevention and acute treatment as seen in clinical practice and reflected by some case reports, case series, and small studies describing such use.10-14 In light of the similar mechanism of action of these therapies and the physiologic role of CGRP, there have been calls for safety evaluation.15
To our knowledge, no studies have evaluated dual CGRP-targeting regimens for migraine in the veteran population. In 2023, the US Department of Veterans Affairs (VA) and US Department of Defense (DoD) updated their clinical practice guidelines for the management of headache.3 For migraine prevention, the VA/DoD guidelines include a strong recommendation for the use of erenumab, fremanezumab, and galcanezumab; a weak recommendation for the use of atogepant; and a recommendation neither for nor against the use of rimegepant. For acute treatment, the guidelines assign a weak recommendation for the use of rimegepant and ubrogepant. Combination use was not addressed.3
Prior to the VA/DoD guidelines, the Veterans Health Administration restricted the dual use of CGRP-targeting therapies for both preventive and acute migraine treatment. However, the VA Pharmacy Benefit Management Service removed the restriction in the Criteria for Use documents, allowing broader access to these medications for veterans.16-22 This change permits the use of CGRP-targeting drugs for both acute and preventive migraine treatment after initial data reflecting real-world case reports and open-label studies suggested possible efficacy without a clear safety concern.11,12 This study aims to fill the gap in the literature by evaluating the safety, efficacy, and overall outcomes of combination CGRP-targeting treatment for migraine prevention and acute treatment in a veteran population.
Methods
This single-center, retrospective, medication use evaluation at the Ralph H. Johnson VA Medical Center (RHJVAMC) was reviewed by the RHJVAMC Research and Development Committee and Quality Improvement Program Evaluation Self Certification Tool, which both determined that institutional review board approval was not required because it was considered part of routine care and quality improvement. Computerized Patient Record System (CPRS) data were reviewed between April 1, 2023 (after the Criteria for Use for CGRP-targeting therapies was updated), through January 31, 2025. Patients were included if they had a confirmed diagnosis of migraine using the International Classification of Headache Disorders, 3rd edition criteria and had concomitant active prescriptions for both a preventive and acute treatment CGRP-targeting agent during the project period.23 Only patients receiving care from the RHJVAMC neurology department were included.
The primary objective was to assess the safety of dual CGRP-targeting therapies for migraine treatment. Key safety endpoints included effects on liver function, kidney function, and BP. Safety outcomes were graded using Common Terminology Criteria for Adverse Events.24 Changes in liver function were categorized as grade 1, 2, or 3 elevations: grade 1 (aspartate aminotransferase [AST]/alanine aminotransferase [ALT] up to 3x the upper limit of normal [ULN] or bilirubin > 1.5 x ULN); grade 2 (AST/ALT 3-6 x ULN or bilirubin 1.5-3 x ULN); and grade 3 (AST/ALT 5-10 x ULN or bilirubin 3-10 x ULN). Kidney function changes were assessed by serum creatinine levels using a similar grading system: Grade 1 (≤ 1.5 x ULN); grade 2 (1.5-3 x baseline of normal); and grade 3 (3-6 x ULN or baseline). Changes in BP were monitored from baseline to the time of the first neurology follow-up. Elevations were grouped into 2 categories, defined as BP ≥ 140 mm Hg systolic and/or 90 mm Hg diastolic (category 1) and ≥ 160 mm Hg systolic and/or 100 mm Hg diastolic (category 2). Neurology documentation was also reviewed in CPRS for individual patient-reported adverse effects (AEs). Safety endpoints were tracked for any occurrence during the project period.
The secondary objective was to describe the patient-reported efficacy of adding a gepant for acute migraine treatment to existing CGRP-targeting therapies for migraine prevention, in those patients who were stable for ≥ 12 weeks on the preventive therapy. Neurology documentation of headache characteristics, including headache severity as rated on a numerical pain score from 0 (no pain) to 10 (worst pain), and duration of headaches (in hours) were recorded during the project period. Changes in headache characteristics were tracked from baseline (ie, the neurology visit when the gepant was first requested) to the first neurology follow-up within 6 months of initiating gepant for acute treatment. If ranges were provided within documentation, a mean was calculated and used for data collection. Neurology documentation was also reviewed for any patient report of overall effectiveness with the added gepant, and categorized as symptoms improved, worsened, or did not change based on subjective report. Descriptive statistics were used for data analysis. A 1-sample Wilcoxon signed rank test was performed as an exploratory analysis for change in headache characteristics from baseline to first neurology follow-up within 6 months. Each individual CGRP regimen was counted as a unique data point to adequately describe changes associated with each new medication and/or dose adjustment. Therefore, patients could be included more than once to account for each distinct treatment regimen.
Results
From April 1, 2023, to January 31, 2025, 96 patients were identified with active prescriptions for dual CGRP-targeting therapies. Of the 96 patients, 89 were included in the final analysis; 1 patient lacked a migraine diagnosis and 6 did not have a concomitant dual CGRP-targeted regimen and were excluded. The mean age of patients was 46.8 years and 54 (61%) were female. The most common migraine diagnosis was chronic migraine in 68 patients (76%). Triptans, ibuprofen, and acetaminophen were the most commonly used acute treatment medications (Table 2).

Safety Assessment
Many of the 89 unique patients trialed > 1 regimen. Thus, for the safety analysis, we analyzed 149 patients on unique dual CGRP-targeting regimens (Table 3). Ubrogepant was used by 126 patients (84.6%) for acute treatment. For preventive therapy, 63 patients (42.3%) used erenumab injections and 55 (36.9%) used fremanezumab injections. Seven patients (4.7%) reported AEs (Table 4). Five of the 7 AEs were noted in the package inserts.25-32 One patient taking both atogepant and ubrogepant reported brain fog that resolved after a dose reduction of atogepant to every other day dosing. A patient taking fremanezumab and rimegepant reported myalgia/joint pain after the first fremanezumab injection, which resolved after a few days and did not recur during the study period.


Nine of 149 patient regimens (6.0%) were associated with changes in liver function tests or serum creatinine, though all but 1 were grade 1 (1 patient had a grade 2 ALT elevation). Twenty-five patients (16.8%) experienced changes in BP, most of which were category 1 elevations. Four patients had systolic or diastolic BP ≥ 160 mm Hg or 100 mm Hg, respectively (Table 5).

Efficacy Assessment
Of the 149 unique dual CGRP regimens, 59 were eligible for the exploratory efficacy analysis. Data were excluded from the efficacy analysis if patients had not been on a stable CGRP preventive migraine regimen for ≥ 12 weeks prior to the addition of a gepant. Fourteen regimens were excluded due to a lack of clear documentation on efficacy, leaving 45 analyzed regimens. Of the 45 regimens, 34 were from unique patients. There was no median change in migraine intensity or duration found in the efficacy analysis (0.0, P = .18, and 0.0, P = .92, respectively). Ten patients on dual CGRP therapy reported that the addition of a gepant for acute treatment improved their symptoms, 20 reported that their symptoms were unchanged and/or worsened, and 29 lacked documentation.
Discussion
This study aimed to describe the safety and efficacy of concomitant CGRP regimens for migraine prevention and acute treatment. To our knowledge, this was the first descriptive study of these agents in a veteran population. The potential for increased AEs with concomitant use of CGRP antagonists is due to the similarities in the mechanism of action between the agents, which both target the same receptor/ligand pathway. Given CGRP activity in both the gastrointestinal and cardiovascular systems, the potential for related AEs is speculative. Patient-reported AEs occurred in 7 of 149 unique treatment regimens reviewed for an incidence rate < 5%. All AEs were nonserious and self-limiting.
Our findings are consistent with available research. A 2024 retrospective, exploratory real-world study evaluating the safety and tolerability of combining CGRP-targeting mAbs with gepants reported findings consistent with our results. This analysis included adult patients treated with ≥ 1 previous anti-CGRP mAb and found that 234 of 516 patients included received a combination of a gepant in addition to a CGRP-targeting mAb. Of these 234 patients, 1.3% reported nonserious AEs.33 Similarly, in a multicenter, open-label, long-term safety study in adults experiencing multiple monthly migraine attacks, a subgroup of 13 participants taking a stable dose of an anti-CGRP mAb also took rimegepant 75 mg as needed for acute treatment for 12 weeks. These patients experienced no serious AEs or any AEs leading to discontinuation.14 A study evaluating the drug-drug interaction, safety, and tolerability of dual therapy (atogepant 60 mg daily and ubrogepant 100 mg every 3 days) in 26 patients found no serious AEs, including no significant changes from baseline in laboratory results, vital signs, or safety-related 12-lead electrocardiogram parameters.15The TANDEM real-world, open-label, prospective study demonstrated similar results. It evaluated the safety and tolerability of concomitant use of ubrogepant and atogepant in patients with episodic migraines and found no increase in AEs when comparing atogepant alone with combination therapy. Twenty-six patients (9.9%) discontinued treatment due to AEs. The most common treatment-related AEs were constipation, nausea, decreased appetite, and fatigue. Efficacy data were also noted to be an exploratory endpoint in the TANDEM study; however, results have not been published.12
Within this safety analysis, new onset gastrointestinal AEs, specifically nausea, only occurred in 1 patient. Hypertension occurred in 25 treatment regimens (16.8%) for 21 unique patients (4 BP elevations occurred in 1 patient on 4 different regimens). However, the retrospective nature of reporting may limit accurate assessment. A closer analysis determined that elevated BP readings correlated with elevated pain scores at the time of the readings, which could have factored into the BP elevations. However, ongoing monitoring is needed due to an increased risk of hypertension, particularly given recent FDA labeling updates for CGRP-targeting therapies including gepants. In light of this, and the overall low incidence of hypertension reported, no new safety concerns were identified.
Limitations
Efficacy data in this project were exploratory. This evaluation did not show a significant difference in migraine intensity or duration after adding a gepant for acute treatment. The study was not powered to detect a significant difference. Limited data exist assessing efficacy outcomes with dual CGRP-targeting treatment regimens. The COURAGE study assessed the real-world effectiveness of ubrogepant and CGRP mAbs with or without the addition of onabotulinumtoxinA. The final analysis of the ubrogepant and CGRP mAb arm included 245 total patients and assessed meaningful migraine pain relief, restoration of normal function after a migraine, and treatment satisfaction. By hour 2, 61.6% of patients reported achieving migraine pain relief, rising to 80.4% by hour 4. Return to normal function occurred in 34.7% at hour 2 and 55.5% by hour 4.13 The long-term safety and efficacy of combining erenumab and rimegepant were described in a case series involving 2 patients. Both patients reported that the concomitant CGRP-targeted therapies were effective and reported no AEs.14
The retrospective design of this study meant that there was potential for limited documentation and introduction of bias into the results. Data were collected at a single VA health care system, and thus, results may not be generalizable to a broader population. However, the study population was consistent with the higher incidence of migraine expected in females in the general population. The sample size was limited, particularly in the exploratory efficacy endpoint assessment.
Limitations were observed due to inconsistent documentation regarding headache characteristics, making it challenging to draw meaningful conclusions from this data set. Additional confounding factors, including polypharmacy, nonadherence to medications, and comorbidities, may have skewed results. For example, while our study design required that the preventive CGRP-targeting medication be stable for 12 weeks for inclusion in further efficacy analysis, other medications commonly used for migraine prevention may have been adjusted (which was not accounted for in this analysis). Given this, more large-scale, placebo-controlled, randomized studies are needed to continue to assess the safety and efficacy of these combination treatment regimens.
Conclusions
Few AEs or safety events were reported with combination CGRP-targeting treatment for acute and preventive treatment of migraine. Those that were identified were considered mild. Efficacy data were limited, and further studies are needed to fully assess outcomes.
- Wattiez AS, Sowers LP, Russo AF. Calcitonin gene-related peptide (CGRP): role in migraine pathophysiology and therapeutic targeting. Expert Opin Ther Targets. 2020;24:91-100. doi:10.1080/14728222.2020.1724285
- Shah T, Bedrin K, Tinsley A. Calcitonin gene relating peptide inhibitors in combination for migraine treatment: a mini-review. Front Pain Res (Lausanne). 2023;4:1130239. doi:10.3389/fpain.2023.1130239
- Department of Veterans Affairs/Department of Defense. VA/DoD clinical practice guideline for management of headache. September 2023. Accessed February 4, 2026. https://www.healthquality.va.gov/guidelines/pain/headache/VA-DoD-CPG-Headache-Full-CPG.pdf
- Russell FA, King R, Smillie SJ, et al. Calcitonin gene-related peptide: physiology and pathophysiology. Physiol Rev. 2014;94:1099-1142. doi:10.1152/physrev.00034.2013
- de Vries Lentsch S, van der Arend BWH, VanDenBrink AM, et al. Blood pressure in patients with migraine treated with monoclonal anti-CGRP (receptor) antibodies: a prospective follow-up study. Neurology. 2022;99:e1897-e1904. doi:10.1212/WNL.0000000000201008
- Favoni V, Giani L, Al-Hassany L, et al. CGRP and migraine from a cardiovascular point of view: what do we expect from blocking CGRP?. J Headache Pain. 2019;20:27. doi:10.1186/s10194-019-0979-y
- Ailani J, Burch RC, Robbins MS, et al. The American Headache Society Consensus Statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61:1021-1039. doi:10.1111/head.14153
- Charles AC, Digre KB, Goadsby PJ, et al. Calcitonin gene-related peptide-targeting therapies are a first-line option for the prevention of migraine: an American Headache Society position statement update. Headache. 2024;64:333-341. doi:10.1111/head.14692
- Puledda F, Sacco S, Diener HC, et al. International Headache Society global practice recommendations for the acute pharmacological treatment of migraine. Cephalalgia. 2024;44:3331024241252666. doi:10.1177/03331024241252666
- Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi:10.1111/head.13930
- Blumenfeld AM, Boinpally R, De Abreu Ferreira R, et al. Phase Ib, open-label, fixed-sequence, drug-drug interaction, safety, and tolerability study between atogepant and ubrogepant in participants with a history of migraine. Headache. 2023;63:322-332. doi:10.1111/head.14433
- Ailani J, Lipton RB, Blumenfeld AM, et al. Safety and tolerability of ubrogepant for the acute treatment of migraine in participants taking atogepant for the preventive treatment of episodic migraine: results from the TANDEM study. Headache. 2025;65:1005-1014. doi:10.1111/head.14871
- Lipton RB, Contreras-De Lama J, Serrano D, et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: results from COURAGE. Neurol Ther. 2024;13:69-83. doi:10.1007/s40120-023-00556-8
- Mullin K, Kudrow D, Croop R, et al. Potential for treatment benefit of small molecule CGRP receptor antagonist plus monoclonal antibody in migraine therapy. Neurology. 2020;94:e2121-e2125. doi:10.1212/WNL.0000000000008944
- Ihara K, Takizawa T, Watanabe N, et al. Potential benefits and possible risks of CGRP-targeted multitherapy in migraine. Expert Opin Drug Metab Toxicol. 2024;20:1-4. doi:10.1080/17425255.2024.2316131
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Ubrogepant (Ubrelvy) criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Ubrogepant_UBRELVY_CFU_Rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for abortive migraine treatment criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_abortive_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_episodic_migraine_prevention_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_chronic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_episodic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Galcanezumab-gnlm (Emgality) for cluster headache criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Galcanezumab_EMGALITY_for_cluster_headache_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Atogepant (Qulipta) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Atogepant_QULIPTA_for_chronic_migraine_prevention_CFU_rev_Jul_2025.pdf
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi:10.1177/0333102417738202
- US Dept of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. November 27, 2017. Accessed March 4, 2026. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf
- Aimovig (erenumab-aooe) injection prescribing information. Amegen Inc. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761077s026lbl.pdf
- Ajovy (fremanezumab-vfrm) injection prescribing information. Teva Pharmaceuticals. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761089s031lbl.pdf
- Vyepti (eptinezumab-jjmr) injection prescribing information. Lundbeck Seattle Biopharmaceuticals. Updated October 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761119s011lbl.pdf
- Emgality (galcanezumab-gnlm) injection prescribing information. Eli Lilly and Company. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761063s010lbl.pdf
- Qulipta (atogepant) tablets prescribing information. AbbVie Inc. Updated September 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215206s013lbl.pdf
- Nurtec ODT (rimegepant) orally disintegrating tablets prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/212728s028lbl.pdf
- Ubrelvy (Ubrogepant) tablets prescribing information. AbbVie Inc. Updated June 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/211765s012lbl.pdf
- Zavzpret (zavegepant) intranasal spray prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/216386s007lbl.pdf
- Alsaadi T, Suliman R, Santos V, et al. Safety and tolerability of combining CGRP monoclonal antibodies with gepants in patients with migraine: a retrospective study. Neurol Ther. 2024;13:465-473. doi:10.1007/s40120-024-00586-w
- Wattiez AS, Sowers LP, Russo AF. Calcitonin gene-related peptide (CGRP): role in migraine pathophysiology and therapeutic targeting. Expert Opin Ther Targets. 2020;24:91-100. doi:10.1080/14728222.2020.1724285
- Shah T, Bedrin K, Tinsley A. Calcitonin gene relating peptide inhibitors in combination for migraine treatment: a mini-review. Front Pain Res (Lausanne). 2023;4:1130239. doi:10.3389/fpain.2023.1130239
- Department of Veterans Affairs/Department of Defense. VA/DoD clinical practice guideline for management of headache. September 2023. Accessed February 4, 2026. https://www.healthquality.va.gov/guidelines/pain/headache/VA-DoD-CPG-Headache-Full-CPG.pdf
- Russell FA, King R, Smillie SJ, et al. Calcitonin gene-related peptide: physiology and pathophysiology. Physiol Rev. 2014;94:1099-1142. doi:10.1152/physrev.00034.2013
- de Vries Lentsch S, van der Arend BWH, VanDenBrink AM, et al. Blood pressure in patients with migraine treated with monoclonal anti-CGRP (receptor) antibodies: a prospective follow-up study. Neurology. 2022;99:e1897-e1904. doi:10.1212/WNL.0000000000201008
- Favoni V, Giani L, Al-Hassany L, et al. CGRP and migraine from a cardiovascular point of view: what do we expect from blocking CGRP?. J Headache Pain. 2019;20:27. doi:10.1186/s10194-019-0979-y
- Ailani J, Burch RC, Robbins MS, et al. The American Headache Society Consensus Statement: update on integrating new migraine treatments into clinical practice. Headache. 2021;61:1021-1039. doi:10.1111/head.14153
- Charles AC, Digre KB, Goadsby PJ, et al. Calcitonin gene-related peptide-targeting therapies are a first-line option for the prevention of migraine: an American Headache Society position statement update. Headache. 2024;64:333-341. doi:10.1111/head.14692
- Puledda F, Sacco S, Diener HC, et al. International Headache Society global practice recommendations for the acute pharmacological treatment of migraine. Cephalalgia. 2024;44:3331024241252666. doi:10.1177/03331024241252666
- Berman G, Croop R, Kudrow D, et al. Safety of rimegepant, an oral CGRP receptor antagonist, plus CGRP monoclonal antibodies for migraine. Headache. 2020;60:1734-1742. doi:10.1111/head.13930
- Blumenfeld AM, Boinpally R, De Abreu Ferreira R, et al. Phase Ib, open-label, fixed-sequence, drug-drug interaction, safety, and tolerability study between atogepant and ubrogepant in participants with a history of migraine. Headache. 2023;63:322-332. doi:10.1111/head.14433
- Ailani J, Lipton RB, Blumenfeld AM, et al. Safety and tolerability of ubrogepant for the acute treatment of migraine in participants taking atogepant for the preventive treatment of episodic migraine: results from the TANDEM study. Headache. 2025;65:1005-1014. doi:10.1111/head.14871
- Lipton RB, Contreras-De Lama J, Serrano D, et al. Real-world use of ubrogepant as acute treatment for migraine with an anti-calcitonin gene-related peptide monoclonal antibody: results from COURAGE. Neurol Ther. 2024;13:69-83. doi:10.1007/s40120-023-00556-8
- Mullin K, Kudrow D, Croop R, et al. Potential for treatment benefit of small molecule CGRP receptor antagonist plus monoclonal antibody in migraine therapy. Neurology. 2020;94:e2121-e2125. doi:10.1212/WNL.0000000000008944
- Ihara K, Takizawa T, Watanabe N, et al. Potential benefits and possible risks of CGRP-targeted multitherapy in migraine. Expert Opin Drug Metab Toxicol. 2024;20:1-4. doi:10.1080/17425255.2024.2316131
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Ubrogepant (Ubrelvy) criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Ubrogepant_UBRELVY_CFU_Rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for abortive migraine treatment criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_abortive_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Rimegepant (Nurtec) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Rimegepant_NURTEC_for_episodic_migraine_prevention_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_chronic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Erenumab-aooe (Aimovig) for episodic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Erenumab_AIMOVIG_for_episodic_migraine_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Galcanezumab-gnlm (Emgality) for cluster headache criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Galcanezumab_EMGALITY_for_cluster_headache_CFU_rev_Jul_2025.pdf
- US Department of Veterans Affairs, Pharmacy Benefits Management Services. Atogepant (Qulipta) for chronic migraine prevention criteria for use. July 2025. Accessed March 4, 2026. https://www.va.gov/formularyadvisor/DOC_PDF/CFU_Atogepant_QULIPTA_for_chronic_migraine_prevention_CFU_rev_Jul_2025.pdf
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38:1-211. doi:10.1177/0333102417738202
- US Dept of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. November 27, 2017. Accessed March 4, 2026. https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf
- Aimovig (erenumab-aooe) injection prescribing information. Amegen Inc. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761077s026lbl.pdf
- Ajovy (fremanezumab-vfrm) injection prescribing information. Teva Pharmaceuticals. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761089s031lbl.pdf
- Vyepti (eptinezumab-jjmr) injection prescribing information. Lundbeck Seattle Biopharmaceuticals. Updated October 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761119s011lbl.pdf
- Emgality (galcanezumab-gnlm) injection prescribing information. Eli Lilly and Company. Updated March 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/761063s010lbl.pdf
- Qulipta (atogepant) tablets prescribing information. AbbVie Inc. Updated September 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/215206s013lbl.pdf
- Nurtec ODT (rimegepant) orally disintegrating tablets prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/212728s028lbl.pdf
- Ubrelvy (Ubrogepant) tablets prescribing information. AbbVie Inc. Updated June 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/211765s012lbl.pdf
- Zavzpret (zavegepant) intranasal spray prescribing information. Pfzier Labs. Updated August 2025. Accessed March 4, 2026. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/216386s007lbl.pdf
- Alsaadi T, Suliman R, Santos V, et al. Safety and tolerability of combining CGRP monoclonal antibodies with gepants in patients with migraine: a retrospective study. Neurol Ther. 2024;13:465-473. doi:10.1007/s40120-024-00586-w
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
Retrospective Review of Dual CGRP-Targeted Regimens for Acute and Preventive Treatment of Migraines in a Veteran Population
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in a Patient With Advanced Melanoma
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in a Patient With Advanced Melanoma
Immune checkpoint inhibitors (ICIs) have become important in oncology and represent an evolving area of therapeutics. Since their approval by the US Food and Drug Administration (FDA) in 2011, ICIs have been increasingly used as modalities in neoadjuvant and adjuvant treatment for resectable solid malignancies and in unresectable disease, such as advanced melanoma, and are associated with improved survival.1
Immune checkpoints are present on the cell surface of activated T cells as well as other immune cells like B cells and natural killer cells. By regulating the length and amplitude of the body’s innate immune response, they maintain immune homeostasis and prevent its overactivation. Immune checkpoints are often thought of as the brakes on the immune system.2
Two glycoproteins that act as immune checkpoints and are targeted by ICIs are cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1). CTLA-4 is upregulated on activated T cells. PD-1 is also expressed on activated T cells, as well as macrophages, B cells, and dendritic cells. Cancer cells can evade immune surveillance by exploiting immune checkpoint pathways. Inhibition of these checkpoints with ICIs reactivates T cells and enables the immune system to recognize and attack cancer cells more effectively. Ipilimumab blocks the activity of CTLA-4 on T cells. Nivolumab and pembrolizumab block the interaction between PD-1 on T cells and its ligand PD-L1 on cancer cells.3,4
Inhibition of these checkpoints is often effective in cancer treatment but can result in the loss of immunologic tolerance with resultant immune-related adverse events (irAEs) and potentially permanent autoimmune disorders. Autoreactive T cells can damage host cell tissues including the colon, lungs, liver, pituitary gland, thyroid, and skin. Severe irAEs include type 1 diabetes mellitus, myositis, nephritis, colitis, pneumonitis, hepatitis, uveitis, hypophysitis, and adrenalitis.4
Hypophysitis is inflammation of the pituitary gland, often with thickening of the pituitary stalk, resulting in dysfunction and hormone deficiencies. While primary hypophysitis is idiopathic, secondary hypophysitis is the result of an underlying condition such as exposure to an ICI. Immune-mediated inflammation of the pituitary gland in hypophysitis may disrupt corticotroph function, leading to adrenocorticotropic hormone (ACTH) deficiency. Early warning features are often vague and nonspecific, such as headache, fatigue, and weakness, which makes diagnosis challenging.3,5
CASE PRESENTATION
A 73-year-old male veteran with a history of metastatic melanoma on ipilimumab 3 mg/kg and nivolumab 1 mg/kg every 3 weeks (a standard combination regimen for advanced melanoma) presented to the emergency department (ED) with 2 weeks of cough, nausea, and severe headache 3 weeks after cycle 2 of combination ICI therapy. The patient had undergone excision of multiple sites of melanoma in situ with recurrence and disease progression after 5 cycles of pembrolizumab. He was subsequently started on combination ICI therapy.
On ED arrival, the patient was afebrile and saturating well on room air. He was normotensive but found to have orthostatic blood pressure. Physical examination was remarkable for dry oral mucosa and decreased skin turgor. Initial laboratory results were significant for hyponatremia of 123 mmol/L (reference range, 136-145 mmol/L), low-normal free thyroxine (T4) level of 0.5 ng/dL (reference range, 0.6-1.2 ng/dL), a low total triiodothyronine level of 32.14 ng/dL (reference range, 85-178 ng/dL), and a low thyrotropin level of 0.19 mIU/L (reference range, 0.35-5.50 mIU/L). Serum osmolarity was low at 259 mOsm/kg (reference range, 285-315 mOsm/kg), urine sodium was high at 168 mEq/L (reference, 20 mEq/L), and urine osmolarity was inappropriately concentrated at 726 mOsm/kg (reference range, 250-1000 mOsm/kg). The patient was admitted for additional testing. His morning cortisol level was within normal limits at 15 mcg/dL (reference range, 6.7-22.5 mcg/dL).
Computed tomography (CT) of the patient’s head revealed no acute findings. Chest CT revealed posterior right lower lobe mild ground-glass opacities, with possible ICI-induced pneumonitis. The patient received fluid resuscitation. Given concern for syndrome of inappropriate antidiuretic hormone secretion, the patient was started on 3 g salt tablets 3 times a day and urea 30 g powder daily. The etiology of the abnormal thyroid levels was unclear to endocrinology at that time. The differential diagnosis included a nonthyroidal illness or central hypothyroidism.
The patient started levothyroxine 75 mcg due to abnormal thyroid levels and persistent fatigue and fludrocortisone 0.1 mg daily to manage orthostatic hypotension. His sodium levels improved to 132 mmol/L over 6 days and he was discharged with levothyroxine 75 mcg daily, fludrocortisone 0.1 mg daily, 3 g salt tabs 3 times a day, urea 30 g powder daily, as well as oral cefpodoxime 500 mg twice daily for 3 days and azithromycin 500 mg once daily for 2 days (for a total of 10 days of antibiotic therapy) to treat potential occult pneumonia.
The patient returned to the ED 3 days after discharge following an outpatient oncology appointment with ongoing severe headaches and persistent nausea. There was concern for recurrent hyponatremia. His sodium level was within normal limits at 133 mmol/L. Repeat morning cortisol was low-normal at 9 mcg/dL. Magnetic resonance imaging (MRI) of the brain was negative for metastatic disease, but showed a slight interval increase in size of the pituitary gland compared with an MRI from 6 months prior, with mild fullness and a slightly convex superior margin near homogeneous enhancement, raising concern for infection or hypophysitis (Figure 1).
The patient was readmitted to the general medicine service and was given intravenous hydrocortisone 100 mg every 8 hours because of concern for central adrenal insufficiency due to grade 3 hypophysitis in the setting of MRI imaging and severe headaches (Table 1). He was not hypotensive at the time of hydrocortisone initiation and other vital signs were stable. A cosyntropin stimulation test—a standard diagnostic test for central adrenal insufficiency—was not performed because the patient had already started high-dose hydrocortisone. The patient’s free T4 on this admission remained low at 0.6 ng/dL.

No adjustments were made to his levothyroxine dose given that he recently began the medication and levels may lag after initiation. After a 4-day hospitalization, the decision was made to continue with the steroid taper and follow up with outpatient endocrinology to obtain a cosyntropin stimulation test to complete a full assessment of his pituitary axis (Figure 2). Repeat thyroid function testing for levothyroxine titration was arranged. The levothyroxine dosage was later increased to 88 mcg daily, but the patient discontinued the medication and remained euthyroid. Endocrinology attributed a nonthyroidal illness as the etiology of his hypothyroidism, likely euthyroid sick syndrome in the setting of illness. His hydrocortisone was tapered during outpatient care and fludrocortisone was discontinued due to hypertension.
One month after his second discharge, the patient presented to the ED with 2 weeks of dizziness, associated lightheadedness, and blurred vision when standing from a sitting position. Upon assessment, symptoms were attributed to poor oral intake. The patient’s vital signs were again positive for orthostatic hypotension, though refractory to adequate fluid replacement. Laboratory testing was significant for a low ACTH level of 3.0 pg/mL (reference range, 7.2-63.3 pg/mL). Given that the patient had not received steroids for 1 week, he underwent a cosyntropin stimulation test, which revealed a blunted response supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis (Table 2).
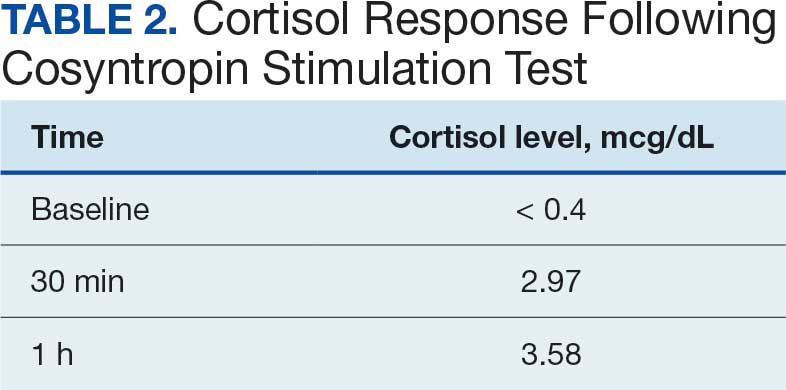
The patient was again readmitted to the general medicine service. A brain MRI showed interval shrinkage of the pituitary gland compared to imaging one month prior, which was attributed to hydrocortisone treatment during this month. CT of the patient’s abdomen demonstrated normal-sized adrenal glands. Positron emission tomography (PET)/CT showed no evidence of pituitary or adrenal metastases. Endocrinology recommended reinitiating oral hydrocortisone 50 mg in the morning and 50 mg around 3 pm daily with fludrocortisone 0.2 mg once daily, which resulted in near resolution of the patient’s symptoms. He was discharged after a 14-day hospitalization with home physical therapy services and endocrinology, nephrology, and oncology follow-up appointments.
The patient was readmitted twice to the general medicine service over the next 6 months for complications from hydrocortisone and fludrocortisone treatment including hypokalemia. He followed up with outpatient clinicians until his death 14 months later. He did not restart ICI therapy, and eventually joined a clinical trial for other advanced melanoma treatments at another institution. The patient’s family consented to the publication of this case report with the accompanying images.
DISCUSSION
The combination of ipilimumab (anti-CTLA-4 monoclonal antibody) and nivolumab (anti-PD-1 monoclonal antibody) is FDA-approved for treatment of advanced melanoma with the goal of harnessing complementary and synergistic mechanisms of dual therapy.6-8 Combination therapy, however, can increase the incidence of irAEs, which are often endocrine-related and more common in patients treated with dual immunotherapy than with monotherapy.9 Hypophysitis has the lowest reported fatality rate among ICI-related irAEs (< 1%), compared with higher mortality rates seen in myocarditis (25%-50%) and pneumonitis (10%-20%).4,10
The patient initially presented with ICI-related hypothyroidism, later identified as secondary (central) hypothyroidism. He was treated with levothyroxine until central hypothyroidism was confirmed. Subsequently, the patient developed headache, poor appetite, and lightheadedness, with MRI findings suggestive of hypophysitis, for which he was started on hydrocortisone. A component of primary adrenal insufficiency was initially considered, given the low ACTH level and blunted response to cosyntropin stimulation following prior high-dose steroid therapy. However, CT imaging demonstrated normal adrenal morphology without atrophy, supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis.
The estimated incidence of ICI-induced hypophysitis is 1.5% to 13.3% with anti-CTLA-4 agents, 0.3% to 3.0% with anti-PD-1 agents, and can be as high as 12.8% with combination therapy.1 ICI-induced hypophysitis is believed to arise from the direct binding of ICI antibodies to their targets on anterior pituitary cells, such as corticotrophs, thyrotrophs, and gonadotrophs, triggering an immune response. One theory for targeting these cells is high CTLA-4 expression in the anterior pituitary gland.11 PD-1 therapies tend to manifest as either hypothyroidism, hyperthyroidism, Graves’ disease, diabetes, or adrenal insufficiency.10
A concern in patients with advanced melanoma is metastasis. Melanoma has a high propensity for brain metastasis.12 There was moderate suspicion for pituitary gland metastasis in this case, though pituitary metastasis more often manifests with symptoms of posterior pituitary gland deficiency, such as polyuria and polydipsia.13 The adrenal gland is the fourth-most common site for melanoma metastases, after the lung, liver, and bone.14 This patient had no evidence of pituitary or adrenal metastases on PET/CT. Therefore, his symptoms were most likely due to ICI therapy. Cases of ≥ 1 endocrine dysfunction have been reported as an ICI therapy irAE.15 In these situations, diagnosing primary and central adrenal insufficiency in the same patient is complex because hormone profiles are intertwined.
Many patients who develop hypophysitis from ICI therapy will require permanent replacement therapy. It is unclear whether low-dose replacement steroids have a significant effect on the efficacy of ICIs. Given that ICI treatment works by enhancing the immune system, medications that suppress the body’s immune system, such as steroids, could interfere with treatment efficacy. However, there are speculations that the development of irAEs is an indicator of effective treatment. In a phase 1 trial of a CTLA-4 blocker in patients with metastatic melanoma, there was a correlation between reduced CTLA-4 expression as well as low rates of melanoma recurrence and a higher incidence of irAEs.16
When assessing patients on ICI treatment, clinicians must remain vigilant for all potential irAEs, especially in patients receiving combination therapy. ICI-induced irAEs can present with vague and nonspecific symptoms. Concurrent endocrine irAEs, such as hypophysitis with thyroiditis or adrenalitis, are not uncommon in combination therapy and can complicate interpretation of hormone profiles. It is prudent for clinicians to review known risk factors. Hypophysitis is typically associated with older adult male patients.17,18
The irAEs of ICI therapy deeply affected the quality of life of the patient in this case, as he was often experiencing many of the clinical symptoms of his hormone insufficiencies as well as the treatment modalities, thus requiring repeated hospital admissions. The risks and benefits of continuing ICI therapy should be an ongoing discussion between the physician and patient and should take into account the acuity and severity of irAEs and oncological disease burden, among other variables. Given the severity of his AEs, the patient stopped ICI therapy and instead opted to enroll in a clinical trial at another institution for continued alternative treatments.
CONCLUSIONS
This case offers a lesson in the diagnostic challenges of vague symptoms in patients with cancer who are receiving ICI therapy. ICI therapy is widely used in the treatment of solid malignancies, and as its use increases, it is expected that clinicians will likely see more cases of irAEs in hospitalized patients. The vague presentation of irAEs can often lead to treatment delays, especially when > 1 irAE presents concurrently. There are ongoing studies researching potential ways to predict the likelihood of developing these irAEs. It is imperative that clinicians are aware of these ICI-related complications and that more research be conducted to understand patient quality of life and treatment guidance based on irAE severity and disease burden.
- Villani A, Potestio L, Fabbrocini G, et al. The treatment of advanced melanoma: therapeutic update. Int J Mol Sci. 2022;23:6388. doi:10.3390/ijms23126388
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. doi:10.1038/nrc3239
- Chang LS, Barroso-Sousa R, Tolaney SM, et al. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40:17-65. doi:10.1210/er.2018-00006
- June CH, Warshauer JT, Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med. 2017;23:540-547. doi:10.1038/nm.4321
- Jessel S, Weiss SA, Austin M, et al. Immune checkpoint inhibitor-induced hypophysitis and patterns of loss of pituitary function. Front Oncol. 2022;12:836859. doi:10.3389/fonc.2022.836859
- Betof AS, Nipp RD, Giobbie-Hurder A, et al. Impact of age on outcomes with immunotherapy for patients with melanoma. Oncologist. 2017;22:963-971. doi:10.1634/theoncologist.2016-0450
- Wolchok JD, Kluger H, Callahan MK, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369:122-133. doi:10.1056/NEJMoa1302369
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723. doi:10.1056/NEJMoa1003466
- Benhima N, Belbaraka R, Langouo Fontsa MD. Single agent vs combination immunotherapy in advanced melanoma: a review of the evidence. Curr Opin Oncol. 2024;36:69-73. doi:10.1097/CCO.0000000000001014
- Tong J, Kartolo A, Yeung C, et al. Long-term toxicities of immune checkpoint inhibitor (ICI) in melanoma patients. Curr Oncol. 2022;29:7953-7963. doi:10.3390/curroncol29100629
- Grouthier V, Lebrun-Vignes B, Moey M, et al. Immune checkpoint inhibitor-associated primary adrenal insufficiency: WHO VigiBase report analysis. Oncologist. 2020;25:696-701. doi:10.1634/theoncologist.2019-0555
- Park BC, Jung S, Wright JJ, et al. Recurrence of hypophysitis after immune checkpoint inhibitor rechallenge. Oncologist. 2022;27:e967-e969. doi:10.1093/oncolo/oyac220
- Zhang D, Wang Z, Shang D, et al. Incidence and prognosis of brain metastases in cutaneous melanoma patients: a population-based study. Melanoma Res. 2019;29:77-84. doi:10.1097/CMR.0000000000000538
- Barnabei A, Carpano S, Chiefari A, et al. Case report: ipilimumab-induced panhypophysitis: an infrequent occurrence and literature review. Front Oncol. 2020;10:582394. doi:10.3389/fonc.2020.582394
- Shortreed H, Burute N, Aseyev O. Management of undifferentiated adrenal gland metastases from malignant melanoma: case report. Front Oncol. 2024;14:1419827. doi:10.3389/fonc.2024.1419827
- Rossi S, Silvetti F, Bordoni M, et al. Pembrolizumab-induced thyroiditis, hypophysitis and adrenalitis: a case of triple endocrine dysfunction. JCEM Case Rep. 2024;2:luae200. doi:10.1210/jcemcr/luae200
- Sanderson K, Scotland R, Lee P, et al. Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III and IV melanoma. J Clin Oncol. 2005;23:741-750. doi:10.1200/JCO.2005.01.128
- de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51:145-156. doi:10.1055/a-0843-3366
Immune checkpoint inhibitors (ICIs) have become important in oncology and represent an evolving area of therapeutics. Since their approval by the US Food and Drug Administration (FDA) in 2011, ICIs have been increasingly used as modalities in neoadjuvant and adjuvant treatment for resectable solid malignancies and in unresectable disease, such as advanced melanoma, and are associated with improved survival.1
Immune checkpoints are present on the cell surface of activated T cells as well as other immune cells like B cells and natural killer cells. By regulating the length and amplitude of the body’s innate immune response, they maintain immune homeostasis and prevent its overactivation. Immune checkpoints are often thought of as the brakes on the immune system.2
Two glycoproteins that act as immune checkpoints and are targeted by ICIs are cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1). CTLA-4 is upregulated on activated T cells. PD-1 is also expressed on activated T cells, as well as macrophages, B cells, and dendritic cells. Cancer cells can evade immune surveillance by exploiting immune checkpoint pathways. Inhibition of these checkpoints with ICIs reactivates T cells and enables the immune system to recognize and attack cancer cells more effectively. Ipilimumab blocks the activity of CTLA-4 on T cells. Nivolumab and pembrolizumab block the interaction between PD-1 on T cells and its ligand PD-L1 on cancer cells.3,4
Inhibition of these checkpoints is often effective in cancer treatment but can result in the loss of immunologic tolerance with resultant immune-related adverse events (irAEs) and potentially permanent autoimmune disorders. Autoreactive T cells can damage host cell tissues including the colon, lungs, liver, pituitary gland, thyroid, and skin. Severe irAEs include type 1 diabetes mellitus, myositis, nephritis, colitis, pneumonitis, hepatitis, uveitis, hypophysitis, and adrenalitis.4
Hypophysitis is inflammation of the pituitary gland, often with thickening of the pituitary stalk, resulting in dysfunction and hormone deficiencies. While primary hypophysitis is idiopathic, secondary hypophysitis is the result of an underlying condition such as exposure to an ICI. Immune-mediated inflammation of the pituitary gland in hypophysitis may disrupt corticotroph function, leading to adrenocorticotropic hormone (ACTH) deficiency. Early warning features are often vague and nonspecific, such as headache, fatigue, and weakness, which makes diagnosis challenging.3,5
CASE PRESENTATION
A 73-year-old male veteran with a history of metastatic melanoma on ipilimumab 3 mg/kg and nivolumab 1 mg/kg every 3 weeks (a standard combination regimen for advanced melanoma) presented to the emergency department (ED) with 2 weeks of cough, nausea, and severe headache 3 weeks after cycle 2 of combination ICI therapy. The patient had undergone excision of multiple sites of melanoma in situ with recurrence and disease progression after 5 cycles of pembrolizumab. He was subsequently started on combination ICI therapy.
On ED arrival, the patient was afebrile and saturating well on room air. He was normotensive but found to have orthostatic blood pressure. Physical examination was remarkable for dry oral mucosa and decreased skin turgor. Initial laboratory results were significant for hyponatremia of 123 mmol/L (reference range, 136-145 mmol/L), low-normal free thyroxine (T4) level of 0.5 ng/dL (reference range, 0.6-1.2 ng/dL), a low total triiodothyronine level of 32.14 ng/dL (reference range, 85-178 ng/dL), and a low thyrotropin level of 0.19 mIU/L (reference range, 0.35-5.50 mIU/L). Serum osmolarity was low at 259 mOsm/kg (reference range, 285-315 mOsm/kg), urine sodium was high at 168 mEq/L (reference, 20 mEq/L), and urine osmolarity was inappropriately concentrated at 726 mOsm/kg (reference range, 250-1000 mOsm/kg). The patient was admitted for additional testing. His morning cortisol level was within normal limits at 15 mcg/dL (reference range, 6.7-22.5 mcg/dL).
Computed tomography (CT) of the patient’s head revealed no acute findings. Chest CT revealed posterior right lower lobe mild ground-glass opacities, with possible ICI-induced pneumonitis. The patient received fluid resuscitation. Given concern for syndrome of inappropriate antidiuretic hormone secretion, the patient was started on 3 g salt tablets 3 times a day and urea 30 g powder daily. The etiology of the abnormal thyroid levels was unclear to endocrinology at that time. The differential diagnosis included a nonthyroidal illness or central hypothyroidism.
The patient started levothyroxine 75 mcg due to abnormal thyroid levels and persistent fatigue and fludrocortisone 0.1 mg daily to manage orthostatic hypotension. His sodium levels improved to 132 mmol/L over 6 days and he was discharged with levothyroxine 75 mcg daily, fludrocortisone 0.1 mg daily, 3 g salt tabs 3 times a day, urea 30 g powder daily, as well as oral cefpodoxime 500 mg twice daily for 3 days and azithromycin 500 mg once daily for 2 days (for a total of 10 days of antibiotic therapy) to treat potential occult pneumonia.
The patient returned to the ED 3 days after discharge following an outpatient oncology appointment with ongoing severe headaches and persistent nausea. There was concern for recurrent hyponatremia. His sodium level was within normal limits at 133 mmol/L. Repeat morning cortisol was low-normal at 9 mcg/dL. Magnetic resonance imaging (MRI) of the brain was negative for metastatic disease, but showed a slight interval increase in size of the pituitary gland compared with an MRI from 6 months prior, with mild fullness and a slightly convex superior margin near homogeneous enhancement, raising concern for infection or hypophysitis (Figure 1).

The patient was readmitted to the general medicine service and was given intravenous hydrocortisone 100 mg every 8 hours because of concern for central adrenal insufficiency due to grade 3 hypophysitis in the setting of MRI imaging and severe headaches (Table 1). He was not hypotensive at the time of hydrocortisone initiation and other vital signs were stable. A cosyntropin stimulation test—a standard diagnostic test for central adrenal insufficiency—was not performed because the patient had already started high-dose hydrocortisone. The patient’s free T4 on this admission remained low at 0.6 ng/dL.

No adjustments were made to his levothyroxine dose given that he recently began the medication and levels may lag after initiation. After a 4-day hospitalization, the decision was made to continue with the steroid taper and follow up with outpatient endocrinology to obtain a cosyntropin stimulation test to complete a full assessment of his pituitary axis (Figure 2). Repeat thyroid function testing for levothyroxine titration was arranged. The levothyroxine dosage was later increased to 88 mcg daily, but the patient discontinued the medication and remained euthyroid. Endocrinology attributed a nonthyroidal illness as the etiology of his hypothyroidism, likely euthyroid sick syndrome in the setting of illness. His hydrocortisone was tapered during outpatient care and fludrocortisone was discontinued due to hypertension.

One month after his second discharge, the patient presented to the ED with 2 weeks of dizziness, associated lightheadedness, and blurred vision when standing from a sitting position. Upon assessment, symptoms were attributed to poor oral intake. The patient’s vital signs were again positive for orthostatic hypotension, though refractory to adequate fluid replacement. Laboratory testing was significant for a low ACTH level of 3.0 pg/mL (reference range, 7.2-63.3 pg/mL). Given that the patient had not received steroids for 1 week, he underwent a cosyntropin stimulation test, which revealed a blunted response supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis (Table 2).

The patient was again readmitted to the general medicine service. A brain MRI showed interval shrinkage of the pituitary gland compared to imaging one month prior, which was attributed to hydrocortisone treatment during this month. CT of the patient’s abdomen demonstrated normal-sized adrenal glands. Positron emission tomography (PET)/CT showed no evidence of pituitary or adrenal metastases. Endocrinology recommended reinitiating oral hydrocortisone 50 mg in the morning and 50 mg around 3 pm daily with fludrocortisone 0.2 mg once daily, which resulted in near resolution of the patient’s symptoms. He was discharged after a 14-day hospitalization with home physical therapy services and endocrinology, nephrology, and oncology follow-up appointments.
The patient was readmitted twice to the general medicine service over the next 6 months for complications from hydrocortisone and fludrocortisone treatment including hypokalemia. He followed up with outpatient clinicians until his death 14 months later. He did not restart ICI therapy, and eventually joined a clinical trial for other advanced melanoma treatments at another institution. The patient’s family consented to the publication of this case report with the accompanying images.
DISCUSSION
The combination of ipilimumab (anti-CTLA-4 monoclonal antibody) and nivolumab (anti-PD-1 monoclonal antibody) is FDA-approved for treatment of advanced melanoma with the goal of harnessing complementary and synergistic mechanisms of dual therapy.6-8 Combination therapy, however, can increase the incidence of irAEs, which are often endocrine-related and more common in patients treated with dual immunotherapy than with monotherapy.9 Hypophysitis has the lowest reported fatality rate among ICI-related irAEs (< 1%), compared with higher mortality rates seen in myocarditis (25%-50%) and pneumonitis (10%-20%).4,10
The patient initially presented with ICI-related hypothyroidism, later identified as secondary (central) hypothyroidism. He was treated with levothyroxine until central hypothyroidism was confirmed. Subsequently, the patient developed headache, poor appetite, and lightheadedness, with MRI findings suggestive of hypophysitis, for which he was started on hydrocortisone. A component of primary adrenal insufficiency was initially considered, given the low ACTH level and blunted response to cosyntropin stimulation following prior high-dose steroid therapy. However, CT imaging demonstrated normal adrenal morphology without atrophy, supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis.
The estimated incidence of ICI-induced hypophysitis is 1.5% to 13.3% with anti-CTLA-4 agents, 0.3% to 3.0% with anti-PD-1 agents, and can be as high as 12.8% with combination therapy.1 ICI-induced hypophysitis is believed to arise from the direct binding of ICI antibodies to their targets on anterior pituitary cells, such as corticotrophs, thyrotrophs, and gonadotrophs, triggering an immune response. One theory for targeting these cells is high CTLA-4 expression in the anterior pituitary gland.11 PD-1 therapies tend to manifest as either hypothyroidism, hyperthyroidism, Graves’ disease, diabetes, or adrenal insufficiency.10
A concern in patients with advanced melanoma is metastasis. Melanoma has a high propensity for brain metastasis.12 There was moderate suspicion for pituitary gland metastasis in this case, though pituitary metastasis more often manifests with symptoms of posterior pituitary gland deficiency, such as polyuria and polydipsia.13 The adrenal gland is the fourth-most common site for melanoma metastases, after the lung, liver, and bone.14 This patient had no evidence of pituitary or adrenal metastases on PET/CT. Therefore, his symptoms were most likely due to ICI therapy. Cases of ≥ 1 endocrine dysfunction have been reported as an ICI therapy irAE.15 In these situations, diagnosing primary and central adrenal insufficiency in the same patient is complex because hormone profiles are intertwined.
Many patients who develop hypophysitis from ICI therapy will require permanent replacement therapy. It is unclear whether low-dose replacement steroids have a significant effect on the efficacy of ICIs. Given that ICI treatment works by enhancing the immune system, medications that suppress the body’s immune system, such as steroids, could interfere with treatment efficacy. However, there are speculations that the development of irAEs is an indicator of effective treatment. In a phase 1 trial of a CTLA-4 blocker in patients with metastatic melanoma, there was a correlation between reduced CTLA-4 expression as well as low rates of melanoma recurrence and a higher incidence of irAEs.16
When assessing patients on ICI treatment, clinicians must remain vigilant for all potential irAEs, especially in patients receiving combination therapy. ICI-induced irAEs can present with vague and nonspecific symptoms. Concurrent endocrine irAEs, such as hypophysitis with thyroiditis or adrenalitis, are not uncommon in combination therapy and can complicate interpretation of hormone profiles. It is prudent for clinicians to review known risk factors. Hypophysitis is typically associated with older adult male patients.17,18
The irAEs of ICI therapy deeply affected the quality of life of the patient in this case, as he was often experiencing many of the clinical symptoms of his hormone insufficiencies as well as the treatment modalities, thus requiring repeated hospital admissions. The risks and benefits of continuing ICI therapy should be an ongoing discussion between the physician and patient and should take into account the acuity and severity of irAEs and oncological disease burden, among other variables. Given the severity of his AEs, the patient stopped ICI therapy and instead opted to enroll in a clinical trial at another institution for continued alternative treatments.
CONCLUSIONS
This case offers a lesson in the diagnostic challenges of vague symptoms in patients with cancer who are receiving ICI therapy. ICI therapy is widely used in the treatment of solid malignancies, and as its use increases, it is expected that clinicians will likely see more cases of irAEs in hospitalized patients. The vague presentation of irAEs can often lead to treatment delays, especially when > 1 irAE presents concurrently. There are ongoing studies researching potential ways to predict the likelihood of developing these irAEs. It is imperative that clinicians are aware of these ICI-related complications and that more research be conducted to understand patient quality of life and treatment guidance based on irAE severity and disease burden.
Immune checkpoint inhibitors (ICIs) have become important in oncology and represent an evolving area of therapeutics. Since their approval by the US Food and Drug Administration (FDA) in 2011, ICIs have been increasingly used as modalities in neoadjuvant and adjuvant treatment for resectable solid malignancies and in unresectable disease, such as advanced melanoma, and are associated with improved survival.1
Immune checkpoints are present on the cell surface of activated T cells as well as other immune cells like B cells and natural killer cells. By regulating the length and amplitude of the body’s innate immune response, they maintain immune homeostasis and prevent its overactivation. Immune checkpoints are often thought of as the brakes on the immune system.2
Two glycoproteins that act as immune checkpoints and are targeted by ICIs are cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1). CTLA-4 is upregulated on activated T cells. PD-1 is also expressed on activated T cells, as well as macrophages, B cells, and dendritic cells. Cancer cells can evade immune surveillance by exploiting immune checkpoint pathways. Inhibition of these checkpoints with ICIs reactivates T cells and enables the immune system to recognize and attack cancer cells more effectively. Ipilimumab blocks the activity of CTLA-4 on T cells. Nivolumab and pembrolizumab block the interaction between PD-1 on T cells and its ligand PD-L1 on cancer cells.3,4
Inhibition of these checkpoints is often effective in cancer treatment but can result in the loss of immunologic tolerance with resultant immune-related adverse events (irAEs) and potentially permanent autoimmune disorders. Autoreactive T cells can damage host cell tissues including the colon, lungs, liver, pituitary gland, thyroid, and skin. Severe irAEs include type 1 diabetes mellitus, myositis, nephritis, colitis, pneumonitis, hepatitis, uveitis, hypophysitis, and adrenalitis.4
Hypophysitis is inflammation of the pituitary gland, often with thickening of the pituitary stalk, resulting in dysfunction and hormone deficiencies. While primary hypophysitis is idiopathic, secondary hypophysitis is the result of an underlying condition such as exposure to an ICI. Immune-mediated inflammation of the pituitary gland in hypophysitis may disrupt corticotroph function, leading to adrenocorticotropic hormone (ACTH) deficiency. Early warning features are often vague and nonspecific, such as headache, fatigue, and weakness, which makes diagnosis challenging.3,5
CASE PRESENTATION
A 73-year-old male veteran with a history of metastatic melanoma on ipilimumab 3 mg/kg and nivolumab 1 mg/kg every 3 weeks (a standard combination regimen for advanced melanoma) presented to the emergency department (ED) with 2 weeks of cough, nausea, and severe headache 3 weeks after cycle 2 of combination ICI therapy. The patient had undergone excision of multiple sites of melanoma in situ with recurrence and disease progression after 5 cycles of pembrolizumab. He was subsequently started on combination ICI therapy.
On ED arrival, the patient was afebrile and saturating well on room air. He was normotensive but found to have orthostatic blood pressure. Physical examination was remarkable for dry oral mucosa and decreased skin turgor. Initial laboratory results were significant for hyponatremia of 123 mmol/L (reference range, 136-145 mmol/L), low-normal free thyroxine (T4) level of 0.5 ng/dL (reference range, 0.6-1.2 ng/dL), a low total triiodothyronine level of 32.14 ng/dL (reference range, 85-178 ng/dL), and a low thyrotropin level of 0.19 mIU/L (reference range, 0.35-5.50 mIU/L). Serum osmolarity was low at 259 mOsm/kg (reference range, 285-315 mOsm/kg), urine sodium was high at 168 mEq/L (reference, 20 mEq/L), and urine osmolarity was inappropriately concentrated at 726 mOsm/kg (reference range, 250-1000 mOsm/kg). The patient was admitted for additional testing. His morning cortisol level was within normal limits at 15 mcg/dL (reference range, 6.7-22.5 mcg/dL).
Computed tomography (CT) of the patient’s head revealed no acute findings. Chest CT revealed posterior right lower lobe mild ground-glass opacities, with possible ICI-induced pneumonitis. The patient received fluid resuscitation. Given concern for syndrome of inappropriate antidiuretic hormone secretion, the patient was started on 3 g salt tablets 3 times a day and urea 30 g powder daily. The etiology of the abnormal thyroid levels was unclear to endocrinology at that time. The differential diagnosis included a nonthyroidal illness or central hypothyroidism.
The patient started levothyroxine 75 mcg due to abnormal thyroid levels and persistent fatigue and fludrocortisone 0.1 mg daily to manage orthostatic hypotension. His sodium levels improved to 132 mmol/L over 6 days and he was discharged with levothyroxine 75 mcg daily, fludrocortisone 0.1 mg daily, 3 g salt tabs 3 times a day, urea 30 g powder daily, as well as oral cefpodoxime 500 mg twice daily for 3 days and azithromycin 500 mg once daily for 2 days (for a total of 10 days of antibiotic therapy) to treat potential occult pneumonia.
The patient returned to the ED 3 days after discharge following an outpatient oncology appointment with ongoing severe headaches and persistent nausea. There was concern for recurrent hyponatremia. His sodium level was within normal limits at 133 mmol/L. Repeat morning cortisol was low-normal at 9 mcg/dL. Magnetic resonance imaging (MRI) of the brain was negative for metastatic disease, but showed a slight interval increase in size of the pituitary gland compared with an MRI from 6 months prior, with mild fullness and a slightly convex superior margin near homogeneous enhancement, raising concern for infection or hypophysitis (Figure 1).

The patient was readmitted to the general medicine service and was given intravenous hydrocortisone 100 mg every 8 hours because of concern for central adrenal insufficiency due to grade 3 hypophysitis in the setting of MRI imaging and severe headaches (Table 1). He was not hypotensive at the time of hydrocortisone initiation and other vital signs were stable. A cosyntropin stimulation test—a standard diagnostic test for central adrenal insufficiency—was not performed because the patient had already started high-dose hydrocortisone. The patient’s free T4 on this admission remained low at 0.6 ng/dL.

No adjustments were made to his levothyroxine dose given that he recently began the medication and levels may lag after initiation. After a 4-day hospitalization, the decision was made to continue with the steroid taper and follow up with outpatient endocrinology to obtain a cosyntropin stimulation test to complete a full assessment of his pituitary axis (Figure 2). Repeat thyroid function testing for levothyroxine titration was arranged. The levothyroxine dosage was later increased to 88 mcg daily, but the patient discontinued the medication and remained euthyroid. Endocrinology attributed a nonthyroidal illness as the etiology of his hypothyroidism, likely euthyroid sick syndrome in the setting of illness. His hydrocortisone was tapered during outpatient care and fludrocortisone was discontinued due to hypertension.

One month after his second discharge, the patient presented to the ED with 2 weeks of dizziness, associated lightheadedness, and blurred vision when standing from a sitting position. Upon assessment, symptoms were attributed to poor oral intake. The patient’s vital signs were again positive for orthostatic hypotension, though refractory to adequate fluid replacement. Laboratory testing was significant for a low ACTH level of 3.0 pg/mL (reference range, 7.2-63.3 pg/mL). Given that the patient had not received steroids for 1 week, he underwent a cosyntropin stimulation test, which revealed a blunted response supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis (Table 2).

The patient was again readmitted to the general medicine service. A brain MRI showed interval shrinkage of the pituitary gland compared to imaging one month prior, which was attributed to hydrocortisone treatment during this month. CT of the patient’s abdomen demonstrated normal-sized adrenal glands. Positron emission tomography (PET)/CT showed no evidence of pituitary or adrenal metastases. Endocrinology recommended reinitiating oral hydrocortisone 50 mg in the morning and 50 mg around 3 pm daily with fludrocortisone 0.2 mg once daily, which resulted in near resolution of the patient’s symptoms. He was discharged after a 14-day hospitalization with home physical therapy services and endocrinology, nephrology, and oncology follow-up appointments.
The patient was readmitted twice to the general medicine service over the next 6 months for complications from hydrocortisone and fludrocortisone treatment including hypokalemia. He followed up with outpatient clinicians until his death 14 months later. He did not restart ICI therapy, and eventually joined a clinical trial for other advanced melanoma treatments at another institution. The patient’s family consented to the publication of this case report with the accompanying images.
DISCUSSION
The combination of ipilimumab (anti-CTLA-4 monoclonal antibody) and nivolumab (anti-PD-1 monoclonal antibody) is FDA-approved for treatment of advanced melanoma with the goal of harnessing complementary and synergistic mechanisms of dual therapy.6-8 Combination therapy, however, can increase the incidence of irAEs, which are often endocrine-related and more common in patients treated with dual immunotherapy than with monotherapy.9 Hypophysitis has the lowest reported fatality rate among ICI-related irAEs (< 1%), compared with higher mortality rates seen in myocarditis (25%-50%) and pneumonitis (10%-20%).4,10
The patient initially presented with ICI-related hypothyroidism, later identified as secondary (central) hypothyroidism. He was treated with levothyroxine until central hypothyroidism was confirmed. Subsequently, the patient developed headache, poor appetite, and lightheadedness, with MRI findings suggestive of hypophysitis, for which he was started on hydrocortisone. A component of primary adrenal insufficiency was initially considered, given the low ACTH level and blunted response to cosyntropin stimulation following prior high-dose steroid therapy. However, CT imaging demonstrated normal adrenal morphology without atrophy, supporting a diagnosis of central adrenal insufficiency secondary to ICI-induced hypophysitis.
The estimated incidence of ICI-induced hypophysitis is 1.5% to 13.3% with anti-CTLA-4 agents, 0.3% to 3.0% with anti-PD-1 agents, and can be as high as 12.8% with combination therapy.1 ICI-induced hypophysitis is believed to arise from the direct binding of ICI antibodies to their targets on anterior pituitary cells, such as corticotrophs, thyrotrophs, and gonadotrophs, triggering an immune response. One theory for targeting these cells is high CTLA-4 expression in the anterior pituitary gland.11 PD-1 therapies tend to manifest as either hypothyroidism, hyperthyroidism, Graves’ disease, diabetes, or adrenal insufficiency.10
A concern in patients with advanced melanoma is metastasis. Melanoma has a high propensity for brain metastasis.12 There was moderate suspicion for pituitary gland metastasis in this case, though pituitary metastasis more often manifests with symptoms of posterior pituitary gland deficiency, such as polyuria and polydipsia.13 The adrenal gland is the fourth-most common site for melanoma metastases, after the lung, liver, and bone.14 This patient had no evidence of pituitary or adrenal metastases on PET/CT. Therefore, his symptoms were most likely due to ICI therapy. Cases of ≥ 1 endocrine dysfunction have been reported as an ICI therapy irAE.15 In these situations, diagnosing primary and central adrenal insufficiency in the same patient is complex because hormone profiles are intertwined.
Many patients who develop hypophysitis from ICI therapy will require permanent replacement therapy. It is unclear whether low-dose replacement steroids have a significant effect on the efficacy of ICIs. Given that ICI treatment works by enhancing the immune system, medications that suppress the body’s immune system, such as steroids, could interfere with treatment efficacy. However, there are speculations that the development of irAEs is an indicator of effective treatment. In a phase 1 trial of a CTLA-4 blocker in patients with metastatic melanoma, there was a correlation between reduced CTLA-4 expression as well as low rates of melanoma recurrence and a higher incidence of irAEs.16
When assessing patients on ICI treatment, clinicians must remain vigilant for all potential irAEs, especially in patients receiving combination therapy. ICI-induced irAEs can present with vague and nonspecific symptoms. Concurrent endocrine irAEs, such as hypophysitis with thyroiditis or adrenalitis, are not uncommon in combination therapy and can complicate interpretation of hormone profiles. It is prudent for clinicians to review known risk factors. Hypophysitis is typically associated with older adult male patients.17,18
The irAEs of ICI therapy deeply affected the quality of life of the patient in this case, as he was often experiencing many of the clinical symptoms of his hormone insufficiencies as well as the treatment modalities, thus requiring repeated hospital admissions. The risks and benefits of continuing ICI therapy should be an ongoing discussion between the physician and patient and should take into account the acuity and severity of irAEs and oncological disease burden, among other variables. Given the severity of his AEs, the patient stopped ICI therapy and instead opted to enroll in a clinical trial at another institution for continued alternative treatments.
CONCLUSIONS
This case offers a lesson in the diagnostic challenges of vague symptoms in patients with cancer who are receiving ICI therapy. ICI therapy is widely used in the treatment of solid malignancies, and as its use increases, it is expected that clinicians will likely see more cases of irAEs in hospitalized patients. The vague presentation of irAEs can often lead to treatment delays, especially when > 1 irAE presents concurrently. There are ongoing studies researching potential ways to predict the likelihood of developing these irAEs. It is imperative that clinicians are aware of these ICI-related complications and that more research be conducted to understand patient quality of life and treatment guidance based on irAE severity and disease burden.
- Villani A, Potestio L, Fabbrocini G, et al. The treatment of advanced melanoma: therapeutic update. Int J Mol Sci. 2022;23:6388. doi:10.3390/ijms23126388
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. doi:10.1038/nrc3239
- Chang LS, Barroso-Sousa R, Tolaney SM, et al. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40:17-65. doi:10.1210/er.2018-00006
- June CH, Warshauer JT, Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med. 2017;23:540-547. doi:10.1038/nm.4321
- Jessel S, Weiss SA, Austin M, et al. Immune checkpoint inhibitor-induced hypophysitis and patterns of loss of pituitary function. Front Oncol. 2022;12:836859. doi:10.3389/fonc.2022.836859
- Betof AS, Nipp RD, Giobbie-Hurder A, et al. Impact of age on outcomes with immunotherapy for patients with melanoma. Oncologist. 2017;22:963-971. doi:10.1634/theoncologist.2016-0450
- Wolchok JD, Kluger H, Callahan MK, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369:122-133. doi:10.1056/NEJMoa1302369
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723. doi:10.1056/NEJMoa1003466
- Benhima N, Belbaraka R, Langouo Fontsa MD. Single agent vs combination immunotherapy in advanced melanoma: a review of the evidence. Curr Opin Oncol. 2024;36:69-73. doi:10.1097/CCO.0000000000001014
- Tong J, Kartolo A, Yeung C, et al. Long-term toxicities of immune checkpoint inhibitor (ICI) in melanoma patients. Curr Oncol. 2022;29:7953-7963. doi:10.3390/curroncol29100629
- Grouthier V, Lebrun-Vignes B, Moey M, et al. Immune checkpoint inhibitor-associated primary adrenal insufficiency: WHO VigiBase report analysis. Oncologist. 2020;25:696-701. doi:10.1634/theoncologist.2019-0555
- Park BC, Jung S, Wright JJ, et al. Recurrence of hypophysitis after immune checkpoint inhibitor rechallenge. Oncologist. 2022;27:e967-e969. doi:10.1093/oncolo/oyac220
- Zhang D, Wang Z, Shang D, et al. Incidence and prognosis of brain metastases in cutaneous melanoma patients: a population-based study. Melanoma Res. 2019;29:77-84. doi:10.1097/CMR.0000000000000538
- Barnabei A, Carpano S, Chiefari A, et al. Case report: ipilimumab-induced panhypophysitis: an infrequent occurrence and literature review. Front Oncol. 2020;10:582394. doi:10.3389/fonc.2020.582394
- Shortreed H, Burute N, Aseyev O. Management of undifferentiated adrenal gland metastases from malignant melanoma: case report. Front Oncol. 2024;14:1419827. doi:10.3389/fonc.2024.1419827
- Rossi S, Silvetti F, Bordoni M, et al. Pembrolizumab-induced thyroiditis, hypophysitis and adrenalitis: a case of triple endocrine dysfunction. JCEM Case Rep. 2024;2:luae200. doi:10.1210/jcemcr/luae200
- Sanderson K, Scotland R, Lee P, et al. Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III and IV melanoma. J Clin Oncol. 2005;23:741-750. doi:10.1200/JCO.2005.01.128
- de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51:145-156. doi:10.1055/a-0843-3366
- Villani A, Potestio L, Fabbrocini G, et al. The treatment of advanced melanoma: therapeutic update. Int J Mol Sci. 2022;23:6388. doi:10.3390/ijms23126388
- Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252-264. doi:10.1038/nrc3239
- Chang LS, Barroso-Sousa R, Tolaney SM, et al. Endocrine toxicity of cancer immunotherapy targeting immune checkpoints. Endocr Rev. 2019;40:17-65. doi:10.1210/er.2018-00006
- June CH, Warshauer JT, Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med. 2017;23:540-547. doi:10.1038/nm.4321
- Jessel S, Weiss SA, Austin M, et al. Immune checkpoint inhibitor-induced hypophysitis and patterns of loss of pituitary function. Front Oncol. 2022;12:836859. doi:10.3389/fonc.2022.836859
- Betof AS, Nipp RD, Giobbie-Hurder A, et al. Impact of age on outcomes with immunotherapy for patients with melanoma. Oncologist. 2017;22:963-971. doi:10.1634/theoncologist.2016-0450
- Wolchok JD, Kluger H, Callahan MK, et al. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369:122-133. doi:10.1056/NEJMoa1302369
- Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723. doi:10.1056/NEJMoa1003466
- Benhima N, Belbaraka R, Langouo Fontsa MD. Single agent vs combination immunotherapy in advanced melanoma: a review of the evidence. Curr Opin Oncol. 2024;36:69-73. doi:10.1097/CCO.0000000000001014
- Tong J, Kartolo A, Yeung C, et al. Long-term toxicities of immune checkpoint inhibitor (ICI) in melanoma patients. Curr Oncol. 2022;29:7953-7963. doi:10.3390/curroncol29100629
- Grouthier V, Lebrun-Vignes B, Moey M, et al. Immune checkpoint inhibitor-associated primary adrenal insufficiency: WHO VigiBase report analysis. Oncologist. 2020;25:696-701. doi:10.1634/theoncologist.2019-0555
- Park BC, Jung S, Wright JJ, et al. Recurrence of hypophysitis after immune checkpoint inhibitor rechallenge. Oncologist. 2022;27:e967-e969. doi:10.1093/oncolo/oyac220
- Zhang D, Wang Z, Shang D, et al. Incidence and prognosis of brain metastases in cutaneous melanoma patients: a population-based study. Melanoma Res. 2019;29:77-84. doi:10.1097/CMR.0000000000000538
- Barnabei A, Carpano S, Chiefari A, et al. Case report: ipilimumab-induced panhypophysitis: an infrequent occurrence and literature review. Front Oncol. 2020;10:582394. doi:10.3389/fonc.2020.582394
- Shortreed H, Burute N, Aseyev O. Management of undifferentiated adrenal gland metastases from malignant melanoma: case report. Front Oncol. 2024;14:1419827. doi:10.3389/fonc.2024.1419827
- Rossi S, Silvetti F, Bordoni M, et al. Pembrolizumab-induced thyroiditis, hypophysitis and adrenalitis: a case of triple endocrine dysfunction. JCEM Case Rep. 2024;2:luae200. doi:10.1210/jcemcr/luae200
- Sanderson K, Scotland R, Lee P, et al. Autoimmunity in a phase I trial of a fully human anti-cytotoxic T-lymphocyte antigen-4 monoclonal antibody with multiple melanoma peptides and Montanide ISA 51 for patients with resected stages III and IV melanoma. J Clin Oncol. 2005;23:741-750. doi:10.1200/JCO.2005.01.128
- de Filette J, Andreescu CE, Cools F, Bravenboer B, Velkeniers B. A systematic review and meta-analysis of endocrine-related adverse events associated with immune checkpoint inhibitors. Horm Metab Res. 2019;51:145-156. doi:10.1055/a-0843-3366
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in a Patient With Advanced Melanoma
Diagnostic Challenge of Immune Checkpoint Inhibitor-Induced Hypophysitis in a Patient With Advanced Melanoma
Cannabis Use by Veterans and Potential Interactions With Antineoplastic Agents: Analysis and Literature Review
Cannabis Use by Veterans and Potential Interactions With Antineoplastic Agents: Analysis and Literature Review
Cannabis has a long history of use for medicinal and recreational purposes. Research illustrates the potential benefits and increased prevalence of cannabis use in patients with cancer.1 Cannabis products have been shown to possess antineoplastic and palliative activity, improving nociceptive and neuropathic pain in addition to chemotherapy-related nausea and vomiting.2-5 Despite these developments and changing social attitudes toward cannabis, there remains a lack of comprehensive data on patient perspectives regarding its use, especially in regions where cannabis remains illegal. This knowledge gap is notable among veterans undergoing cancer treatment in states where cannabis is prohibited. Up to 57% of veterans report lifetime marijuana use, making it crucial to understand this population’s cannabis use patterns and potential interactions with cancer treatments.6
This observational study sought to determine the prevalence of cannabis use among patients undergoing cancer treatment at the US Department of Veterans Affairs (VA) Memphis Healthcare System and evaluate the potential risks associated with combining cannabis products with anticancer therapies.
METHODS
This prospective observational study identified cannabis use among veterans receiving antineoplastic therapy at the Lt. Col. Luke Weathers Jr. VA Medical Center (WJVAMC) and analyzed potential interactions between cannabis products and their cancer treatments. Participants included adults aged > 18 years undergoing antineoplastic therapy at WJVAMC who consented to the study. Data collection involved a written survey approved by the WJVAMC Institutional Review Board and verbal consent from participants. The survey asked participants about their cannabis use in the previous 90 days, including details on quantity, frequency, and method of consumption (eg, inhalation, oral, topical). No incentives were offered for participation.
Surveys from 50 patients who used cannabis were analyzed and their electronic health records were reviewed for sex, age, diagnosis, and antineoplastic regimen. This information was securely stored. A literature review was conducted using PubMed and the Cochrane Library to explore potential interactions between cannabis and the antineoplastic agents that were prescribed to patients in the study, focusing on toxicity, efficacy, or synergistic effects.
Patients were categorized into 4 groups based on treatment: cytotoxic chemotherapy, immunotherapy, endocrine therapy, and targeted therapy. Patients undergoing multiple types of therapies were included in each applicable category.
RESULTS
A total of 132 patients agreed to participate. Fifty patients (38%) acknowledged using cannabis products within 90 days. The patients that used cannabis products within 90 days of the survey reported the following malignancies: 8 patients (16%) had prostate cancer, 3 patients (6%) had hepatocellular carcinoma, 7 patients (14%) had pancreatic carcinoma, 5 patients (10%) had multiple myeloma, 3 patients (6%) had chronic lymphocytic leukemia, 9 patients (18%) had non-small cell lung cancer, 3 patients (6%) had breast cancer, 3 (6%) patients had bladder cancer, 2 patients (4%) had renal cell carcinoma, 1 (2%) patient had chronic myeloid leukemia, 1 (2%) patient had renal amyloid, 1 patient (2%) had supraglottic squamous cell carcinoma, 1 patient (2%) had esophageal carcinoma, 1 (2%) patient had small cell lung cancer, 1 (2%) patient had gastric cancer, and 1 patient (2%) had follicular lymphoma.
Five (10%) of the cannabis users were female, and 45 (90%) were male. Twenty-nine patients (58%) were aged 66 to 75 years, 16 (32%) were aged 56 to 65 years, 3 (6%) were aged 46 to 55 years, and 2 (4%) were aged 76 to 85 years.
Thirty-five patients (70%) inhaled cannabis as opposed to using it via other formulations or a combination (eg, inhalation and topical). Thirty-eight percent of patients used cannabis once daily, 24% used < 1 daily, and 28% used it ≥ 2 times daily. Five patients (10%) did not report the frequency of their cannabis use. Among the patients who reported cannabis use, 21 (42%) were undergoing cytotoxic chemotherapy, 19 (38%) were undergoing immunotherapy, 12 (24%) were undergoing targeted therapy, and 10 (20%) were undergoing endocrine therapy. Some patients were treated with multiple types of antineoplastic agents and were counted in multiple categories (Table 1).
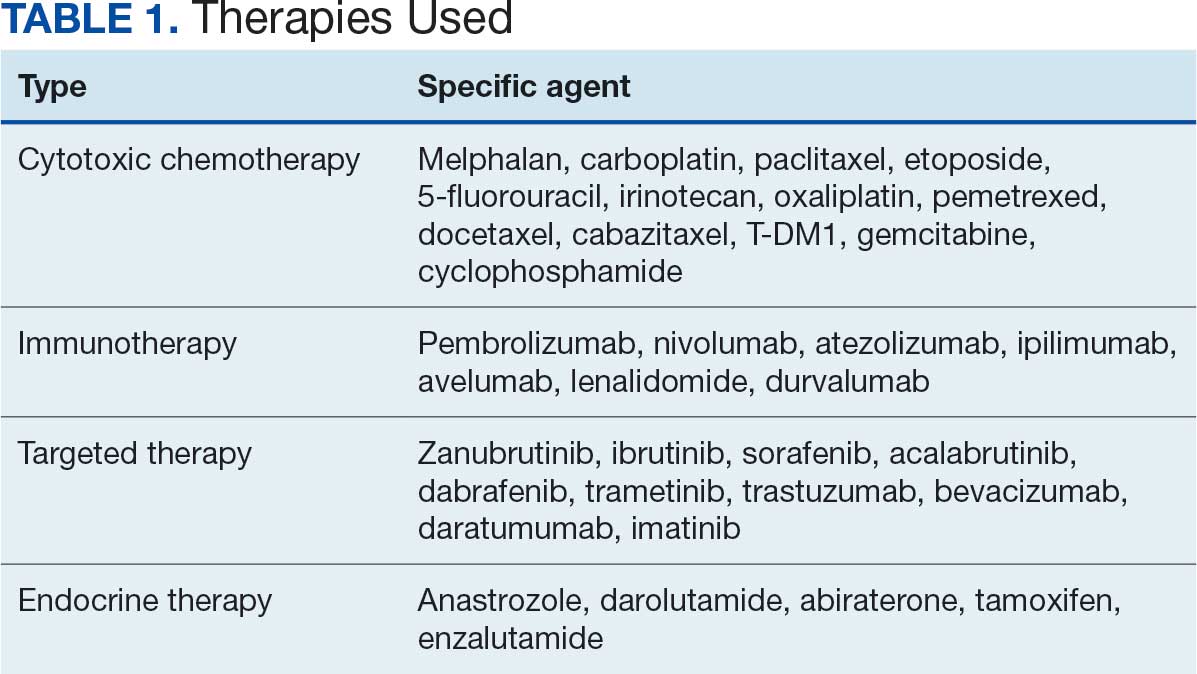
Following a literature review of cannabis and antineoplastic agents, patients were evaluated for the potential effects of cannabis on their treatment. The literature review revealed that 31% of cytotoxic chemotherapy agents received by patients in this study might have increased toxicity, and 19% could have reduced efficacy when combined with cannabis. Among immunotherapy agents received by patients in this study, 70% might have decreased efficacy when combined with cannabis use. For targeted therapies, 35% could have increased toxicity, and 70% of endocrine agents could potentially have decreased efficacy (Table 2).
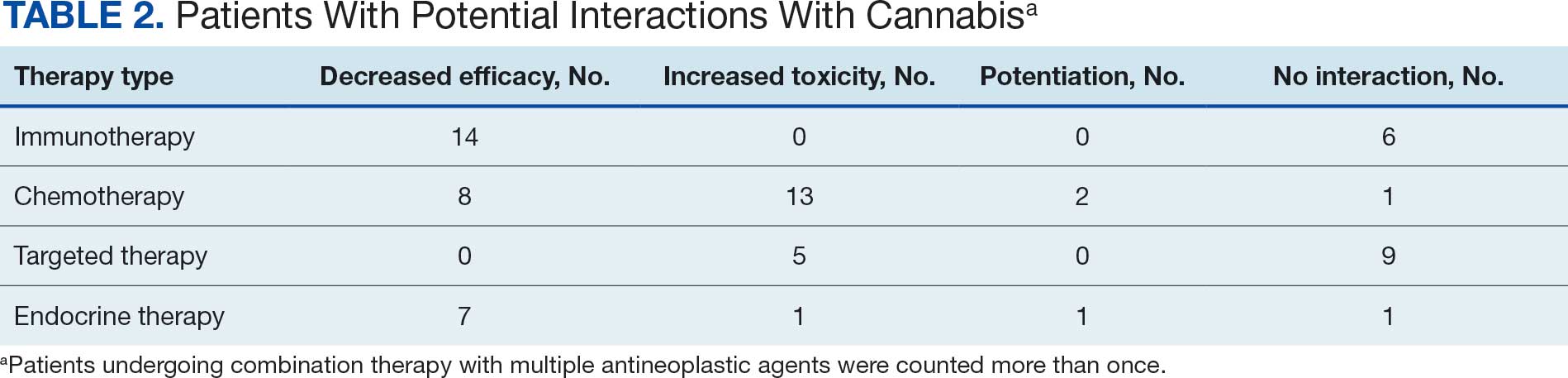
DISCUSSION
This prospective study corroborates previous research by demonstrating that more than one-third of patients receiving oncology care at WJVAMC use cannabis, most often inhaled. Cannabis use was observed among patients undergoing various cancer therapies, including cytotoxic chemotherapy, immunotherapy, targeted therapy, and endocrine therapy. The most common malignancies among cannabis users at WJVAMC include patients with lung cancer, prostate cancer, pancreatic cancer, and multiple myeloma. Cannabis use in patients with pancreatic cancer and multiple myeloma was significantly out of proportion to their prevalence at WJVAMC. This could potentially be due to their drastic effect on quality of life.
Cannabis use increased the risk of toxicity in patients treated with cytotoxic chemotherapy and targeted therapy. Cannabis use potentially decreased efficacy for patients treated with cytotoxic chemotherapy and/or immunotherapy. Cannabis use did not increase the risk of toxicity or efficacy in patients treated with endocrine therapy.
Antineoplastics/Cannabis Interactions
The potential interactions between cannabis and antineoplastic therapies administered at WJVAMC are worth exploring. While this review aims to shed light on possible interactions, it is important to acknowledge that much of the data is preliminary and derived from in vitro studies. The interactions should be interpreted as potential risks rather than established facts. Additional research is needed to confirm these interactions and effectively guide clinical practices. Understanding these dynamics is essential to optimize patient care and manage the complex interplay between cannabis use and cancer treatment.
Originating from Central Asia, the cannabis plant contains > 400 medicinally relevant compounds, of which about 100 are cannabinoids (CBs). Key CBs are cannabidiol (CBD), a nonpsychoactive compound, and ?-9-tetrahydrocannabinol (THC), a psychoactive compound. THC can make up 20% to 30% of the dry weight of female cannabis flowers.7
CBs act through the endocannabinoid system, involving CB1 and CB2 receptors, endogenous CBs like anandamide (AEA) and 2-arachidonoylglycerol, and various enzymes. These endogenous CBs, derived from arachidonic acid, play roles in cell growth and proliferation.8 In some studies, AEA has induced apoptosis in neuroblastoma cells and inhibited proliferation in breast cancer cells. However, other research suggests AEA may block apoptosis under certain conditions.9
CB receptors are transmembrane proteins that interact with CBs differently depending on tissue type and CB structure. Synthetic CBs are designed to target specific receptors, while natural CBs may act as both agonists and antagonists.10
Cytochrome P450 Metabolism
The human cytochrome P450 (CYP) 3A subfamily affects the metabolism of many therapeutic drugs, including cancer therapeutics.11 The various compositions of cannabis are primarily metabolized by the CYP450 pathway, the same as many cancer-directed pharmacologic treatments. CBs act as both CYP inducers and inhibitors. THC, for example, is a CYP inducer whereas CBD is a CYP inhibitor; both are found in the various compounds available for consumption.12,13 Pharmacology research has suggested potential interactions and effects on established adverse symptoms, but clinical data are lacking, and current research revealing interactions are only recognized in vitro.14
The Antineoplastic Activity of Cannabis
CBs can affect various cancer-related pathways such as PKB, AMPK, CAMKK-ß, mTOR, PDHK, HIF-1 a, and PPAR-γ. Δ-9-THC can selectively induce apoptosis in tumor cells without harming normal cells, though the exact mechanism remains unclear. Promising results from early mouse studies led to a 2006 human study where intracranial Δ-9-THC in patients with recurrent glioma yielded a median survival of 24 weeks, with 2 patients surviving > 1 year.15
In a 2022 review article, Cherkasova et al highlighted potential clinical benefits of cannabis across various cancers. They found that upregulated CB1 receptors in colon cancer might enhance the effect of 5-fluorouracil. However, many studies are preliminary and therefore not definitive.10
Additional research is needed to refine these findings. Challenges include variability in cannabis formulations, the complex tumor microenvironment, and the legal and psychoactive issues surrounding cannabis use. These factors complicate the design of multicenter randomized studies and may deter patients from disclosing cannabis use, thereby hindering efforts to fully understand its therapeutic potential.
Cannabis/Cytotoxic Chemotherapy Interactions
The chemotherapy agents used in this study included carboplatin, paclitaxel, 5-fluorouracil, etoposide, irinotecan, oxaliplatin, pemetrexed, docetaxel, cabazitaxel, T-DM1, gemcitabine, and cyclophosphamide. There is a paucity of research regarding the interactions between cytotoxic chemotherapy and cannabis. Most studies focused on CBD due to its inhibition of the CYP450 pathway, which is used for metabolizing cytotoxic chemotherapies. Through this mechanism, CBD could potentially increase the concentrations of chemotherapeutic agents, enhancing their toxicity.
When combined with irinotecan, cannabis can pose risks. Δ-9-THC undergoes first-pass metabolism in the liver, mediated by the CYP450 system and CYP3A4. The glucuronidation of irinotecan is mediated by uridine diphosphate glycosyltransferase, leading to its recirculation within the hepatic system and potentially increased toxicity due to prolonged drug presence. Cannabis may also compete with drug binding to albumin, altering the plasma concentrations of irinotecan and its conversion to the metabolite SN38.16
Cannabis products can affect chemotherapy levels by interacting with cellular transporters. The MRP1 transporter family, encoded by the ABCC gene family, is expressed mainly in the lung, kidney, skeletal muscle, and hematopoietic stem cells. A 2018 study investigating the effects of THC, CBD, and CBN on MRP1 transporters found that the presence of a cannabis component increased the concentration of vincristine 3-fold. Additional studies suggest the interaction with the CB1 receptor may lead to changes in the expression of MRP1 transporters.17
CBD inhibits the BCRP transporter, which functions as an efflux pump for methotrexate. Consequently, CBD can increase methotrexate levels, potentially enhancing efficacy but also worsening adverse effects.18
In pancreatic cancer, CBD specifically interacts with gemcitabine. CB1 and CB2 receptors are upregulated, and CBD inhibits the GPR55 receptor. These interactions may enhance the antineoplastic effect of gemcitabine, reducing cell cycle progression and growth.19
CBD also interacts with temozolomide (TMZ) by affecting extracellular vesicles used by cells for pro-oncogenic signaling and immune system evasion. Experiments on patient-derived glioblastoma cells, both chemotherapy-resistant and chemotherapy-sensitive, found that CBD increases the formation of extracellular vesicles with reduced levels of miR21 (pro-oncogenic) and elevated levels of miR126 (antioncogenic).20 CBD has also been found to decrease prohibitin levels, a protein associated with TMZ resistance.
In patients with glioblastoma, CBD combined with chemotherapeutic agents like TMZ, carmustine, doxorubicin, and cisplatin has shown increased sensitivity and improved tumor response. CBD is also known to inhibit NF-kB, a pathway that sustains tumor viability despite chemotherapy.21 Additionally, CBD inhibits the P-glycoprotein system, affecting chemotherapy efflux from neoplastic cells.14 In vitro studies have found that CBD is synergistic with bortezomib in inhibiting cancer cell viability. In another glioblastoma model, CBD enhanced the antiproliferative effects of both TMZ and carmustine.14
Different cannabis formulations may vary in how they interact with various cytotoxic chemotherapeutic agents. Some may potentiate the effects of chemotherapy and act synergistically to inhibit tumor growth, while others may lead to increased toxicity.10 More research is needed to determine which formulations, in combination with specific agents and doses, may have significant interactions that warrant adjustments in chemotherapy dosing.
Cannabis/Immunotherapy Interactions
Cannabis is an immunosuppressant. Data suggest the use of cannabis during immunotherapy worsens treatment outcomes in patients with cancer.22 Exogenous (THC) and endogenous (AEA) CBs negatively affect antitumor immunity by impairing the function of tumor-specific T cells via CB2 and by inhibiting the Jak1-STATs signaling in T cells through CNR2. Xiong et al found that THC reduces the therapeutic effect of anti-PD-1 therapy.22
In a prospective observational clinical study, Bar-Sela et al analyzed 102 patients with advanced cancer—of which 68 were cannabis users—that were started on immune checkpoint inhibitor therapy. The study found that cannabis users on anti-PD-1 (nivolumab, pembrolizumab), anti-CTLA-4 (ipilimumab), and anti-PD-L1 (durvalumab, atezolizumab) had a significant decrease in time to treatment progression and overall survival vs cannabis non-users.23 However, a 2023 study by Waissengrin et al found that concomitant use of medical cannabis with pembrolizumab had no harmful effect in advanced non-small cell lung cancer.24 Time to treatment progression of cannabis users did not differ from cannabis nonusers.25
Cannabis/Endocrine Therapy Interactions
In addition to having direct antineoplastic activity on tumor cells, data exist that show how cannabis affects the endocrine system. In animal models, cannabis has been found to suppress the whole hypothalamic-pituitary-adrenal axis as well as other hormones like thyroid, prolactin, and growth hormone. In breast cancer, cannabis competes with estrogen for the estrogen receptor and suppresses growth.26
The endocrine agents used by patients with cancer in this study were antiandrogens like abiraterone, enzalutamide, tamoxifen and anastrozole. Abiraterone is metabolized by CYP450 isoenzymes and uridine diphosphate glycosyltransferases. Cannabis inhibits both processes and therefore may lead to increased toxicities.27 Conversely, enzalutamide is a strong CYP3A inducer, and cannabis use during enzalutamide therapy may significantly increase the toxic effects of cannabis.
There is evidence that molecular pathways involving CB receptors and estrogens overlap, which may lead to interactions when antiestrogens are used in cannabis users with hormone receptor-positive breast cancer.26 In preclinical studies, tamoxifen has been shown to act as an inverse agonist on CB1 and CB2 receptors, though the significance of this finding is unclear. There is no research evaluating the effects of CBs on tamoxifen treatment. However, CBD has been found to potentiate the effectiveness of anastrozole or exemestane in breast cancer cell lines.28 Dobovišek et al demonstrated no inhibitory effect of CBD on the activity of tamoxifen, fulvestrant, or palbociclib in breast cancer cell lines.29 The interactions between hormone receptor-positive breast cancer and cannabinoids are complex, and the clinical significance of these interactions remains difficult to identify.
Cannabis/Targeted Therapy Interactions
The targeted therapies used by patients in this study included zanubrutinib, ibrutinib, sorafenib, acalabrutinib, dabrafenib, trametinib, trastuzumab, bevacizumab, daratumumab, and imatinib. Compared to other classes of cancer treatments, most studies have not demonstrated decreased efficacy or increased toxicity of targeted anticancer drugs when used concomitantly with CBD.29
Trastuzumab is a recombinant humanized monoclonal antibody that targets the proto-oncogene HER2/neu. It is used to treat select patients with metastatic breast cancer. Studies have shown that cannabis use does not attenuate the effectiveness of trastuzumab in HER2-positive and triple-negative breast cancer subtypes.29 One study found that CBD, in combination with chemotherapeutics and Bruton tyrosine kinase inhibitors, such as ibrutinib and zanubrutinib, has synergistic potential for treating diffuse large B-cell lymphoma and mantle cell lymphoma cell lines. This synergy is attributed to the CB1 antagonist activity of cannabis against diffuse large B-cell lymphoma and mantle cell lymphoma cell lines.30,31
Moreover, combining cannabinoids with bevacizumab (a monoclonal anti-VEGF antibody) has been shown to decrease tumor growth and intratumoral hypoxia in clinically relevant human glioblastoma models. This effect is mediated through the downregulation of HIF-1α.32 Long-term studies evaluating the potential harmful or synergistic potential of CBD on targeted anticancer therapy are needed.
CONCLUSIONS
This exploratory study of patients receiving cancer therapy at WJVAMC found a significant prevalence of concurrent cannabis use among patients undergoing antineoplastic treatments. Given that many antineoplastic agents are metabolized by the CYP450 enzyme system, the findings of this study suggest that concurrent cannabis use may pose risks of suboptimal therapeutic outcomes due to potential interactions affecting drug metabolism. These interactions could impact the efficacy and toxicity of the antineoplastic therapies, potentially leading to diminished therapeutic effects or exacerbated adverse reactions.
Patients should be informed regarding the potential decreased efficacy of immunotherapy with concurrent use of cannabis products. They should also be aware of the possibility of increased toxicity with other treatment modalities, though the exact impact on efficacy remains unclear. This highlights the necessity of caution when combining cannabis with prescribed cancer treatments.
While this study identified possible interactions, its data are preliminary and highlight the need for more rigorous research. Future studies should include larger, well-designed cohorts to compare outcomes between cannabis users and nonusers. Such research is essential to fully elucidate the clinical implications of cannabis use during cancer treatment, address the high prevalence of cannabis use among patients with cancer, and mitigate potential risks associated with combining cannabis products with antineoplastic therapies. This will ensure that treatment strategies are optimized for safety and efficacy in this complex patient population.
- Steele G, Arneson T, Zylla D. A comprehensive review of cannabis in patients with cancer: availability in the USA, general efficacy, and safety. Curr Oncol Rep. 2019;21:1-10. doi:10.1007/s11912-019-0757-7
- Brown D, Watson M, Schloss J. Pharmacological evidence of medicinal cannabis in oncology: a systematic review. Support Care Cancer. 2019;27:3195-320. doi:10.1007/s00520-019-04774-5
- Abrams DI. Integrating cannabis into clinical cancer care. Curr Oncol. 2016;23:S8-S14. doi:10.37.47/co.23.3099
- Serafimovska T, Darkovska-Serafimovska M, Stefkov G, Arsova-Sarafinovska Z, Balkanov T. Pharmacotherapeutic considerations for use of cannabinoids to relieve symptoms of nausea and vomiting induced by chemotherapy. Folia Medica (Plovdiv). 2020;62:668-678. doi:10.3897/folmed.62e51478
- Bar-Sela G, Zalman D, Semenysty V, Ballan E. The effects of dosage-controlled cannabis capsules on cancer-related cachexia and anorexia syndrome in advanced cancer patients: pilot study. Integr Cancer Ther. 2019;18:1534735419881498. doi:10.1177/1534735419881498
- Pederson ER, Villarosa-Hurlocker MC, Prince MA. Use of protective behavioral strategies among young adult veteran marijuana users. Cannabis. 2018;1:14-27.
- Schilling S, Melzer R, McCabe PF. Cannabis sativa. Curr Biol. 2020;30:R8-R9. doi:10.1016/j.cub.2019.10.039
- McDougle DR, Kambalyal A, Meling DD, Das A. Endocannabinoids anandamide and 2-arachidonoylglycerol are substrates for human CYP2J2 epoxygenase. J Pharmacol Exp Ther. 2014;351:616-627. doi:10.1124/jpet.114216598
- Movsesyan VA, Stoica BA, Yakovlev AG, et al. Anandamide-induced cell death in primary neuronal cultures: role of calpain and caspase pathways. Cell Death Differ. 2004;11:1121-1132. doi:10.1038/sj.cdd.4401442
- Cherkasova V, Wang B, Gerasymchuk M, Fiselier A, Kovalchuk O, Kovalchuk I. Use of cannabis and cannabinoids for treatment of cancer. Cancers (Basel). 2022;14:5142. doi:10.3390/cancers14205142
- Engels FK, Ten Tije AJ, Baker SD, et al. Effect of cytochrome P450 3A4 inhibition on the pharmacokinetics of docetaxel. Clin Pharmacol Ther. 2004;75:448-454. doi:10.1016/j.clpt.2004.01.001
- Alsherbiny MA, Li CG. Medicinal cannabis-potential drug interactions. Medicines (Basel). 2018;6:3. doi:10.3390/medicines6010003
- Stout SM, Cimino NM. Exogenous cannabinoids as substrates, inhibitors, and inducers of human drug metabolizing enzymes: a systematic review. Drug Metab Rev. 2014;46:86-95. doi:10.3109/03602532.2013.849268
- Opitz BJ, Ostroff ML, Whitman AC. The potential clinical implications and importance of drug interactions between anticancer agents and cannabidiol in patients with cancer. J Pharm Pract. 2020;33:506-512. doi:10.1177/0897190019828920
- Guzmán M, Duarte MJ, Blázquez C, et al. A pilot clinical study of D9-tetrahydrocannabinol in patients with recurrent glioblastoma multiforme. Br J Cancer. 2006;95:197-203. doi:10.1038/sj.bjc.6603236
- Kopjar N, Fuchs N, Brcic Karaconji I, et al. High doses of ?9-tetrahydrocannabinol might impair irinotecan chemotherapy: a review of potentially harmful interactions. Clin Drug Investig. 2020;40:775-787. doi:10.1007/s40261-020-00954-y
- Bouquié R, Deslandes G, Mazaré H, et al. Cannabis and anticancer drugs: societal usage and expected pharmacological interactions - a review. Fundam Clin Pharmacol. 2018;32:462-484. doi:10.1111/fcp.12373
- Buchtova T, Lukac D, Skrott Z, Chroma K, Bartek J, Mistrik M. Drug-drug interactions of cannabidiol with standard-of-care chemotherapeutics. Int J Mol Sci. 2023;24:2885. doi:10.3390/ijms24032885
- Sharafi G, He H, Nikfarjam M. Potential use of cannabinoids for the treatment of pancreatic cancer. J Pancreat Cancer. 2019;5:1-7. doi:10.1089/pancan.2018.0019
- Kosgodage US, Uysal-Onganer P, MacLatchy A, et al. Cannabidiol affects extracellular vesicle release, miR21 and miR126, and reduces prohibitin protein in glioblastoma multiforme cells. Transl Oncol. 2019;12:513-522. doi:10.1016/j.tranon.2018.12.004
- Elbaz M, Nasser MW, Ravi J, et al. Modulation of the tumor microenvironment and inhibition of EGF/EGFR pathway: novel anti-tumor mechanisms of cannabidiol in breast cancer. Mol Oncol. 2015;9:906-919. doi:10.1016/j.molonc.2014.12.010
- Xiong X, Chen S, Shen J, et al. Cannabis suppresses anti-tumor immunity by inhibiting JAK/STAT signaling in T cells through CNR2. Signal Transduct Target Ther. 2022;7:99. doi:10.1038/s41392-022-00918-y
- Bar-Sela G, Cohen I, Campisi-Pinto S, et al. Cannabis consumption used by cancer patients during immunotherapy correlates with poor clinical outcome. Cancers (Basel). 2020;12:2447. doi:10.3390/cancers12092447
- Waissengrin B, Leshem Y, Taya M, et al. The use of medical cannabis concomitantly with immune checkpoint inhibitors in non-small cell lung cancer: a sigh of relief? Eur J Cancer. 2023;180:52-61. doi:10.1016/j.ejca.2022.11.022
- Sarsembayeva A, Schicho R. Cannabinoids and the endocannabinoid system in immunotherapy: helpful or harmful? Front Oncol. 2023;13:1296906. doi:10.3389/fonc.2023.1296906
- Kisková T, Mungenast F, Suváková M, Jäger W, Thalhammer T. Future aspects for cannabinoids in breast cancer therapy. Int J Mol Sci. 2019;20:1673. doi:10.3390/ijms20071673
- Woerdenbag HJ, Olinga P, Kok EA, et al. Potential, limitations and risks of cannabis-derived products in cancer treatment. Cancers (Basel). 2023;15:2119. doi:10.3390/cancers15072119
- Almeida CF, Teixeira N, Valente MJ, Vinggaard AM, Correia-da-Silva G, Amaral C. Cannabidiol as a promising adjuvant therapy for estrogen receptor-positive breast tumors: unveiling its benefits with aromatase inhibitors. Cancers (Basel). 2023;15:2517. doi:10.3390/cancers15092517
- Dobovišek L, Novak M, Krstanovic F, Borštnar S, Turnšek TL, Debeljak N. Effect of combining CBD with standard breast cancer therapeutics. Adv Cancer Biol Metastasis. 2022;4:100038. doi:10.1016/j.adcanc.2022.100038
- Strong T, Rauvolfova J, Jackson E, Pham LV, Bryant J. Synergistic effect of cannabidiol with conventional chemotherapy treatment. Blood. 2018;132:5382. doi:10.1182/blood-2018-99-116749
- Maggi F, Morelli MB, Tomassoni D, et al. The effects of cannabidiol via TRPV2 channel in chronic myeloid leukemia cells and its combination with imatinib. Cancer Sci. 2022;113:1235-1249. doi:10.1111/cas.15257
- Obad N, Janji B, Prestegarden L, et al. ATPS-59 improving efficacy of bevacizumab treatment in glioblastoma by targeting hif1 alpha. Neuro Oncol. 2015;17:v31. doi:10.1093/neuonc/nov204.59
Cannabis has a long history of use for medicinal and recreational purposes. Research illustrates the potential benefits and increased prevalence of cannabis use in patients with cancer.1 Cannabis products have been shown to possess antineoplastic and palliative activity, improving nociceptive and neuropathic pain in addition to chemotherapy-related nausea and vomiting.2-5 Despite these developments and changing social attitudes toward cannabis, there remains a lack of comprehensive data on patient perspectives regarding its use, especially in regions where cannabis remains illegal. This knowledge gap is notable among veterans undergoing cancer treatment in states where cannabis is prohibited. Up to 57% of veterans report lifetime marijuana use, making it crucial to understand this population’s cannabis use patterns and potential interactions with cancer treatments.6
This observational study sought to determine the prevalence of cannabis use among patients undergoing cancer treatment at the US Department of Veterans Affairs (VA) Memphis Healthcare System and evaluate the potential risks associated with combining cannabis products with anticancer therapies.
METHODS
This prospective observational study identified cannabis use among veterans receiving antineoplastic therapy at the Lt. Col. Luke Weathers Jr. VA Medical Center (WJVAMC) and analyzed potential interactions between cannabis products and their cancer treatments. Participants included adults aged > 18 years undergoing antineoplastic therapy at WJVAMC who consented to the study. Data collection involved a written survey approved by the WJVAMC Institutional Review Board and verbal consent from participants. The survey asked participants about their cannabis use in the previous 90 days, including details on quantity, frequency, and method of consumption (eg, inhalation, oral, topical). No incentives were offered for participation.
Surveys from 50 patients who used cannabis were analyzed and their electronic health records were reviewed for sex, age, diagnosis, and antineoplastic regimen. This information was securely stored. A literature review was conducted using PubMed and the Cochrane Library to explore potential interactions between cannabis and the antineoplastic agents that were prescribed to patients in the study, focusing on toxicity, efficacy, or synergistic effects.
Patients were categorized into 4 groups based on treatment: cytotoxic chemotherapy, immunotherapy, endocrine therapy, and targeted therapy. Patients undergoing multiple types of therapies were included in each applicable category.
RESULTS
A total of 132 patients agreed to participate. Fifty patients (38%) acknowledged using cannabis products within 90 days. The patients that used cannabis products within 90 days of the survey reported the following malignancies: 8 patients (16%) had prostate cancer, 3 patients (6%) had hepatocellular carcinoma, 7 patients (14%) had pancreatic carcinoma, 5 patients (10%) had multiple myeloma, 3 patients (6%) had chronic lymphocytic leukemia, 9 patients (18%) had non-small cell lung cancer, 3 patients (6%) had breast cancer, 3 (6%) patients had bladder cancer, 2 patients (4%) had renal cell carcinoma, 1 (2%) patient had chronic myeloid leukemia, 1 (2%) patient had renal amyloid, 1 patient (2%) had supraglottic squamous cell carcinoma, 1 patient (2%) had esophageal carcinoma, 1 (2%) patient had small cell lung cancer, 1 (2%) patient had gastric cancer, and 1 patient (2%) had follicular lymphoma.
Five (10%) of the cannabis users were female, and 45 (90%) were male. Twenty-nine patients (58%) were aged 66 to 75 years, 16 (32%) were aged 56 to 65 years, 3 (6%) were aged 46 to 55 years, and 2 (4%) were aged 76 to 85 years.
Thirty-five patients (70%) inhaled cannabis as opposed to using it via other formulations or a combination (eg, inhalation and topical). Thirty-eight percent of patients used cannabis once daily, 24% used < 1 daily, and 28% used it ≥ 2 times daily. Five patients (10%) did not report the frequency of their cannabis use. Among the patients who reported cannabis use, 21 (42%) were undergoing cytotoxic chemotherapy, 19 (38%) were undergoing immunotherapy, 12 (24%) were undergoing targeted therapy, and 10 (20%) were undergoing endocrine therapy. Some patients were treated with multiple types of antineoplastic agents and were counted in multiple categories (Table 1).

Following a literature review of cannabis and antineoplastic agents, patients were evaluated for the potential effects of cannabis on their treatment. The literature review revealed that 31% of cytotoxic chemotherapy agents received by patients in this study might have increased toxicity, and 19% could have reduced efficacy when combined with cannabis. Among immunotherapy agents received by patients in this study, 70% might have decreased efficacy when combined with cannabis use. For targeted therapies, 35% could have increased toxicity, and 70% of endocrine agents could potentially have decreased efficacy (Table 2).

DISCUSSION
This prospective study corroborates previous research by demonstrating that more than one-third of patients receiving oncology care at WJVAMC use cannabis, most often inhaled. Cannabis use was observed among patients undergoing various cancer therapies, including cytotoxic chemotherapy, immunotherapy, targeted therapy, and endocrine therapy. The most common malignancies among cannabis users at WJVAMC include patients with lung cancer, prostate cancer, pancreatic cancer, and multiple myeloma. Cannabis use in patients with pancreatic cancer and multiple myeloma was significantly out of proportion to their prevalence at WJVAMC. This could potentially be due to their drastic effect on quality of life.
Cannabis use increased the risk of toxicity in patients treated with cytotoxic chemotherapy and targeted therapy. Cannabis use potentially decreased efficacy for patients treated with cytotoxic chemotherapy and/or immunotherapy. Cannabis use did not increase the risk of toxicity or efficacy in patients treated with endocrine therapy.
Antineoplastics/Cannabis Interactions
The potential interactions between cannabis and antineoplastic therapies administered at WJVAMC are worth exploring. While this review aims to shed light on possible interactions, it is important to acknowledge that much of the data is preliminary and derived from in vitro studies. The interactions should be interpreted as potential risks rather than established facts. Additional research is needed to confirm these interactions and effectively guide clinical practices. Understanding these dynamics is essential to optimize patient care and manage the complex interplay between cannabis use and cancer treatment.
Originating from Central Asia, the cannabis plant contains > 400 medicinally relevant compounds, of which about 100 are cannabinoids (CBs). Key CBs are cannabidiol (CBD), a nonpsychoactive compound, and ?-9-tetrahydrocannabinol (THC), a psychoactive compound. THC can make up 20% to 30% of the dry weight of female cannabis flowers.7
CBs act through the endocannabinoid system, involving CB1 and CB2 receptors, endogenous CBs like anandamide (AEA) and 2-arachidonoylglycerol, and various enzymes. These endogenous CBs, derived from arachidonic acid, play roles in cell growth and proliferation.8 In some studies, AEA has induced apoptosis in neuroblastoma cells and inhibited proliferation in breast cancer cells. However, other research suggests AEA may block apoptosis under certain conditions.9
CB receptors are transmembrane proteins that interact with CBs differently depending on tissue type and CB structure. Synthetic CBs are designed to target specific receptors, while natural CBs may act as both agonists and antagonists.10
Cytochrome P450 Metabolism
The human cytochrome P450 (CYP) 3A subfamily affects the metabolism of many therapeutic drugs, including cancer therapeutics.11 The various compositions of cannabis are primarily metabolized by the CYP450 pathway, the same as many cancer-directed pharmacologic treatments. CBs act as both CYP inducers and inhibitors. THC, for example, is a CYP inducer whereas CBD is a CYP inhibitor; both are found in the various compounds available for consumption.12,13 Pharmacology research has suggested potential interactions and effects on established adverse symptoms, but clinical data are lacking, and current research revealing interactions are only recognized in vitro.14
The Antineoplastic Activity of Cannabis
CBs can affect various cancer-related pathways such as PKB, AMPK, CAMKK-ß, mTOR, PDHK, HIF-1 a, and PPAR-γ. Δ-9-THC can selectively induce apoptosis in tumor cells without harming normal cells, though the exact mechanism remains unclear. Promising results from early mouse studies led to a 2006 human study where intracranial Δ-9-THC in patients with recurrent glioma yielded a median survival of 24 weeks, with 2 patients surviving > 1 year.15
In a 2022 review article, Cherkasova et al highlighted potential clinical benefits of cannabis across various cancers. They found that upregulated CB1 receptors in colon cancer might enhance the effect of 5-fluorouracil. However, many studies are preliminary and therefore not definitive.10
Additional research is needed to refine these findings. Challenges include variability in cannabis formulations, the complex tumor microenvironment, and the legal and psychoactive issues surrounding cannabis use. These factors complicate the design of multicenter randomized studies and may deter patients from disclosing cannabis use, thereby hindering efforts to fully understand its therapeutic potential.
Cannabis/Cytotoxic Chemotherapy Interactions
The chemotherapy agents used in this study included carboplatin, paclitaxel, 5-fluorouracil, etoposide, irinotecan, oxaliplatin, pemetrexed, docetaxel, cabazitaxel, T-DM1, gemcitabine, and cyclophosphamide. There is a paucity of research regarding the interactions between cytotoxic chemotherapy and cannabis. Most studies focused on CBD due to its inhibition of the CYP450 pathway, which is used for metabolizing cytotoxic chemotherapies. Through this mechanism, CBD could potentially increase the concentrations of chemotherapeutic agents, enhancing their toxicity.
When combined with irinotecan, cannabis can pose risks. Δ-9-THC undergoes first-pass metabolism in the liver, mediated by the CYP450 system and CYP3A4. The glucuronidation of irinotecan is mediated by uridine diphosphate glycosyltransferase, leading to its recirculation within the hepatic system and potentially increased toxicity due to prolonged drug presence. Cannabis may also compete with drug binding to albumin, altering the plasma concentrations of irinotecan and its conversion to the metabolite SN38.16
Cannabis products can affect chemotherapy levels by interacting with cellular transporters. The MRP1 transporter family, encoded by the ABCC gene family, is expressed mainly in the lung, kidney, skeletal muscle, and hematopoietic stem cells. A 2018 study investigating the effects of THC, CBD, and CBN on MRP1 transporters found that the presence of a cannabis component increased the concentration of vincristine 3-fold. Additional studies suggest the interaction with the CB1 receptor may lead to changes in the expression of MRP1 transporters.17
CBD inhibits the BCRP transporter, which functions as an efflux pump for methotrexate. Consequently, CBD can increase methotrexate levels, potentially enhancing efficacy but also worsening adverse effects.18
In pancreatic cancer, CBD specifically interacts with gemcitabine. CB1 and CB2 receptors are upregulated, and CBD inhibits the GPR55 receptor. These interactions may enhance the antineoplastic effect of gemcitabine, reducing cell cycle progression and growth.19
CBD also interacts with temozolomide (TMZ) by affecting extracellular vesicles used by cells for pro-oncogenic signaling and immune system evasion. Experiments on patient-derived glioblastoma cells, both chemotherapy-resistant and chemotherapy-sensitive, found that CBD increases the formation of extracellular vesicles with reduced levels of miR21 (pro-oncogenic) and elevated levels of miR126 (antioncogenic).20 CBD has also been found to decrease prohibitin levels, a protein associated with TMZ resistance.
In patients with glioblastoma, CBD combined with chemotherapeutic agents like TMZ, carmustine, doxorubicin, and cisplatin has shown increased sensitivity and improved tumor response. CBD is also known to inhibit NF-kB, a pathway that sustains tumor viability despite chemotherapy.21 Additionally, CBD inhibits the P-glycoprotein system, affecting chemotherapy efflux from neoplastic cells.14 In vitro studies have found that CBD is synergistic with bortezomib in inhibiting cancer cell viability. In another glioblastoma model, CBD enhanced the antiproliferative effects of both TMZ and carmustine.14
Different cannabis formulations may vary in how they interact with various cytotoxic chemotherapeutic agents. Some may potentiate the effects of chemotherapy and act synergistically to inhibit tumor growth, while others may lead to increased toxicity.10 More research is needed to determine which formulations, in combination with specific agents and doses, may have significant interactions that warrant adjustments in chemotherapy dosing.
Cannabis/Immunotherapy Interactions
Cannabis is an immunosuppressant. Data suggest the use of cannabis during immunotherapy worsens treatment outcomes in patients with cancer.22 Exogenous (THC) and endogenous (AEA) CBs negatively affect antitumor immunity by impairing the function of tumor-specific T cells via CB2 and by inhibiting the Jak1-STATs signaling in T cells through CNR2. Xiong et al found that THC reduces the therapeutic effect of anti-PD-1 therapy.22
In a prospective observational clinical study, Bar-Sela et al analyzed 102 patients with advanced cancer—of which 68 were cannabis users—that were started on immune checkpoint inhibitor therapy. The study found that cannabis users on anti-PD-1 (nivolumab, pembrolizumab), anti-CTLA-4 (ipilimumab), and anti-PD-L1 (durvalumab, atezolizumab) had a significant decrease in time to treatment progression and overall survival vs cannabis non-users.23 However, a 2023 study by Waissengrin et al found that concomitant use of medical cannabis with pembrolizumab had no harmful effect in advanced non-small cell lung cancer.24 Time to treatment progression of cannabis users did not differ from cannabis nonusers.25
Cannabis/Endocrine Therapy Interactions
In addition to having direct antineoplastic activity on tumor cells, data exist that show how cannabis affects the endocrine system. In animal models, cannabis has been found to suppress the whole hypothalamic-pituitary-adrenal axis as well as other hormones like thyroid, prolactin, and growth hormone. In breast cancer, cannabis competes with estrogen for the estrogen receptor and suppresses growth.26
The endocrine agents used by patients with cancer in this study were antiandrogens like abiraterone, enzalutamide, tamoxifen and anastrozole. Abiraterone is metabolized by CYP450 isoenzymes and uridine diphosphate glycosyltransferases. Cannabis inhibits both processes and therefore may lead to increased toxicities.27 Conversely, enzalutamide is a strong CYP3A inducer, and cannabis use during enzalutamide therapy may significantly increase the toxic effects of cannabis.
There is evidence that molecular pathways involving CB receptors and estrogens overlap, which may lead to interactions when antiestrogens are used in cannabis users with hormone receptor-positive breast cancer.26 In preclinical studies, tamoxifen has been shown to act as an inverse agonist on CB1 and CB2 receptors, though the significance of this finding is unclear. There is no research evaluating the effects of CBs on tamoxifen treatment. However, CBD has been found to potentiate the effectiveness of anastrozole or exemestane in breast cancer cell lines.28 Dobovišek et al demonstrated no inhibitory effect of CBD on the activity of tamoxifen, fulvestrant, or palbociclib in breast cancer cell lines.29 The interactions between hormone receptor-positive breast cancer and cannabinoids are complex, and the clinical significance of these interactions remains difficult to identify.
Cannabis/Targeted Therapy Interactions
The targeted therapies used by patients in this study included zanubrutinib, ibrutinib, sorafenib, acalabrutinib, dabrafenib, trametinib, trastuzumab, bevacizumab, daratumumab, and imatinib. Compared to other classes of cancer treatments, most studies have not demonstrated decreased efficacy or increased toxicity of targeted anticancer drugs when used concomitantly with CBD.29
Trastuzumab is a recombinant humanized monoclonal antibody that targets the proto-oncogene HER2/neu. It is used to treat select patients with metastatic breast cancer. Studies have shown that cannabis use does not attenuate the effectiveness of trastuzumab in HER2-positive and triple-negative breast cancer subtypes.29 One study found that CBD, in combination with chemotherapeutics and Bruton tyrosine kinase inhibitors, such as ibrutinib and zanubrutinib, has synergistic potential for treating diffuse large B-cell lymphoma and mantle cell lymphoma cell lines. This synergy is attributed to the CB1 antagonist activity of cannabis against diffuse large B-cell lymphoma and mantle cell lymphoma cell lines.30,31
Moreover, combining cannabinoids with bevacizumab (a monoclonal anti-VEGF antibody) has been shown to decrease tumor growth and intratumoral hypoxia in clinically relevant human glioblastoma models. This effect is mediated through the downregulation of HIF-1α.32 Long-term studies evaluating the potential harmful or synergistic potential of CBD on targeted anticancer therapy are needed.
CONCLUSIONS
This exploratory study of patients receiving cancer therapy at WJVAMC found a significant prevalence of concurrent cannabis use among patients undergoing antineoplastic treatments. Given that many antineoplastic agents are metabolized by the CYP450 enzyme system, the findings of this study suggest that concurrent cannabis use may pose risks of suboptimal therapeutic outcomes due to potential interactions affecting drug metabolism. These interactions could impact the efficacy and toxicity of the antineoplastic therapies, potentially leading to diminished therapeutic effects or exacerbated adverse reactions.
Patients should be informed regarding the potential decreased efficacy of immunotherapy with concurrent use of cannabis products. They should also be aware of the possibility of increased toxicity with other treatment modalities, though the exact impact on efficacy remains unclear. This highlights the necessity of caution when combining cannabis with prescribed cancer treatments.
While this study identified possible interactions, its data are preliminary and highlight the need for more rigorous research. Future studies should include larger, well-designed cohorts to compare outcomes between cannabis users and nonusers. Such research is essential to fully elucidate the clinical implications of cannabis use during cancer treatment, address the high prevalence of cannabis use among patients with cancer, and mitigate potential risks associated with combining cannabis products with antineoplastic therapies. This will ensure that treatment strategies are optimized for safety and efficacy in this complex patient population.
Cannabis has a long history of use for medicinal and recreational purposes. Research illustrates the potential benefits and increased prevalence of cannabis use in patients with cancer.1 Cannabis products have been shown to possess antineoplastic and palliative activity, improving nociceptive and neuropathic pain in addition to chemotherapy-related nausea and vomiting.2-5 Despite these developments and changing social attitudes toward cannabis, there remains a lack of comprehensive data on patient perspectives regarding its use, especially in regions where cannabis remains illegal. This knowledge gap is notable among veterans undergoing cancer treatment in states where cannabis is prohibited. Up to 57% of veterans report lifetime marijuana use, making it crucial to understand this population’s cannabis use patterns and potential interactions with cancer treatments.6
This observational study sought to determine the prevalence of cannabis use among patients undergoing cancer treatment at the US Department of Veterans Affairs (VA) Memphis Healthcare System and evaluate the potential risks associated with combining cannabis products with anticancer therapies.
METHODS
This prospective observational study identified cannabis use among veterans receiving antineoplastic therapy at the Lt. Col. Luke Weathers Jr. VA Medical Center (WJVAMC) and analyzed potential interactions between cannabis products and their cancer treatments. Participants included adults aged > 18 years undergoing antineoplastic therapy at WJVAMC who consented to the study. Data collection involved a written survey approved by the WJVAMC Institutional Review Board and verbal consent from participants. The survey asked participants about their cannabis use in the previous 90 days, including details on quantity, frequency, and method of consumption (eg, inhalation, oral, topical). No incentives were offered for participation.
Surveys from 50 patients who used cannabis were analyzed and their electronic health records were reviewed for sex, age, diagnosis, and antineoplastic regimen. This information was securely stored. A literature review was conducted using PubMed and the Cochrane Library to explore potential interactions between cannabis and the antineoplastic agents that were prescribed to patients in the study, focusing on toxicity, efficacy, or synergistic effects.
Patients were categorized into 4 groups based on treatment: cytotoxic chemotherapy, immunotherapy, endocrine therapy, and targeted therapy. Patients undergoing multiple types of therapies were included in each applicable category.
RESULTS
A total of 132 patients agreed to participate. Fifty patients (38%) acknowledged using cannabis products within 90 days. The patients that used cannabis products within 90 days of the survey reported the following malignancies: 8 patients (16%) had prostate cancer, 3 patients (6%) had hepatocellular carcinoma, 7 patients (14%) had pancreatic carcinoma, 5 patients (10%) had multiple myeloma, 3 patients (6%) had chronic lymphocytic leukemia, 9 patients (18%) had non-small cell lung cancer, 3 patients (6%) had breast cancer, 3 (6%) patients had bladder cancer, 2 patients (4%) had renal cell carcinoma, 1 (2%) patient had chronic myeloid leukemia, 1 (2%) patient had renal amyloid, 1 patient (2%) had supraglottic squamous cell carcinoma, 1 patient (2%) had esophageal carcinoma, 1 (2%) patient had small cell lung cancer, 1 (2%) patient had gastric cancer, and 1 patient (2%) had follicular lymphoma.
Five (10%) of the cannabis users were female, and 45 (90%) were male. Twenty-nine patients (58%) were aged 66 to 75 years, 16 (32%) were aged 56 to 65 years, 3 (6%) were aged 46 to 55 years, and 2 (4%) were aged 76 to 85 years.
Thirty-five patients (70%) inhaled cannabis as opposed to using it via other formulations or a combination (eg, inhalation and topical). Thirty-eight percent of patients used cannabis once daily, 24% used < 1 daily, and 28% used it ≥ 2 times daily. Five patients (10%) did not report the frequency of their cannabis use. Among the patients who reported cannabis use, 21 (42%) were undergoing cytotoxic chemotherapy, 19 (38%) were undergoing immunotherapy, 12 (24%) were undergoing targeted therapy, and 10 (20%) were undergoing endocrine therapy. Some patients were treated with multiple types of antineoplastic agents and were counted in multiple categories (Table 1).

Following a literature review of cannabis and antineoplastic agents, patients were evaluated for the potential effects of cannabis on their treatment. The literature review revealed that 31% of cytotoxic chemotherapy agents received by patients in this study might have increased toxicity, and 19% could have reduced efficacy when combined with cannabis. Among immunotherapy agents received by patients in this study, 70% might have decreased efficacy when combined with cannabis use. For targeted therapies, 35% could have increased toxicity, and 70% of endocrine agents could potentially have decreased efficacy (Table 2).

DISCUSSION
This prospective study corroborates previous research by demonstrating that more than one-third of patients receiving oncology care at WJVAMC use cannabis, most often inhaled. Cannabis use was observed among patients undergoing various cancer therapies, including cytotoxic chemotherapy, immunotherapy, targeted therapy, and endocrine therapy. The most common malignancies among cannabis users at WJVAMC include patients with lung cancer, prostate cancer, pancreatic cancer, and multiple myeloma. Cannabis use in patients with pancreatic cancer and multiple myeloma was significantly out of proportion to their prevalence at WJVAMC. This could potentially be due to their drastic effect on quality of life.
Cannabis use increased the risk of toxicity in patients treated with cytotoxic chemotherapy and targeted therapy. Cannabis use potentially decreased efficacy for patients treated with cytotoxic chemotherapy and/or immunotherapy. Cannabis use did not increase the risk of toxicity or efficacy in patients treated with endocrine therapy.
Antineoplastics/Cannabis Interactions
The potential interactions between cannabis and antineoplastic therapies administered at WJVAMC are worth exploring. While this review aims to shed light on possible interactions, it is important to acknowledge that much of the data is preliminary and derived from in vitro studies. The interactions should be interpreted as potential risks rather than established facts. Additional research is needed to confirm these interactions and effectively guide clinical practices. Understanding these dynamics is essential to optimize patient care and manage the complex interplay between cannabis use and cancer treatment.
Originating from Central Asia, the cannabis plant contains > 400 medicinally relevant compounds, of which about 100 are cannabinoids (CBs). Key CBs are cannabidiol (CBD), a nonpsychoactive compound, and ?-9-tetrahydrocannabinol (THC), a psychoactive compound. THC can make up 20% to 30% of the dry weight of female cannabis flowers.7
CBs act through the endocannabinoid system, involving CB1 and CB2 receptors, endogenous CBs like anandamide (AEA) and 2-arachidonoylglycerol, and various enzymes. These endogenous CBs, derived from arachidonic acid, play roles in cell growth and proliferation.8 In some studies, AEA has induced apoptosis in neuroblastoma cells and inhibited proliferation in breast cancer cells. However, other research suggests AEA may block apoptosis under certain conditions.9
CB receptors are transmembrane proteins that interact with CBs differently depending on tissue type and CB structure. Synthetic CBs are designed to target specific receptors, while natural CBs may act as both agonists and antagonists.10
Cytochrome P450 Metabolism
The human cytochrome P450 (CYP) 3A subfamily affects the metabolism of many therapeutic drugs, including cancer therapeutics.11 The various compositions of cannabis are primarily metabolized by the CYP450 pathway, the same as many cancer-directed pharmacologic treatments. CBs act as both CYP inducers and inhibitors. THC, for example, is a CYP inducer whereas CBD is a CYP inhibitor; both are found in the various compounds available for consumption.12,13 Pharmacology research has suggested potential interactions and effects on established adverse symptoms, but clinical data are lacking, and current research revealing interactions are only recognized in vitro.14
The Antineoplastic Activity of Cannabis
CBs can affect various cancer-related pathways such as PKB, AMPK, CAMKK-ß, mTOR, PDHK, HIF-1 a, and PPAR-γ. Δ-9-THC can selectively induce apoptosis in tumor cells without harming normal cells, though the exact mechanism remains unclear. Promising results from early mouse studies led to a 2006 human study where intracranial Δ-9-THC in patients with recurrent glioma yielded a median survival of 24 weeks, with 2 patients surviving > 1 year.15
In a 2022 review article, Cherkasova et al highlighted potential clinical benefits of cannabis across various cancers. They found that upregulated CB1 receptors in colon cancer might enhance the effect of 5-fluorouracil. However, many studies are preliminary and therefore not definitive.10
Additional research is needed to refine these findings. Challenges include variability in cannabis formulations, the complex tumor microenvironment, and the legal and psychoactive issues surrounding cannabis use. These factors complicate the design of multicenter randomized studies and may deter patients from disclosing cannabis use, thereby hindering efforts to fully understand its therapeutic potential.
Cannabis/Cytotoxic Chemotherapy Interactions
The chemotherapy agents used in this study included carboplatin, paclitaxel, 5-fluorouracil, etoposide, irinotecan, oxaliplatin, pemetrexed, docetaxel, cabazitaxel, T-DM1, gemcitabine, and cyclophosphamide. There is a paucity of research regarding the interactions between cytotoxic chemotherapy and cannabis. Most studies focused on CBD due to its inhibition of the CYP450 pathway, which is used for metabolizing cytotoxic chemotherapies. Through this mechanism, CBD could potentially increase the concentrations of chemotherapeutic agents, enhancing their toxicity.
When combined with irinotecan, cannabis can pose risks. Δ-9-THC undergoes first-pass metabolism in the liver, mediated by the CYP450 system and CYP3A4. The glucuronidation of irinotecan is mediated by uridine diphosphate glycosyltransferase, leading to its recirculation within the hepatic system and potentially increased toxicity due to prolonged drug presence. Cannabis may also compete with drug binding to albumin, altering the plasma concentrations of irinotecan and its conversion to the metabolite SN38.16
Cannabis products can affect chemotherapy levels by interacting with cellular transporters. The MRP1 transporter family, encoded by the ABCC gene family, is expressed mainly in the lung, kidney, skeletal muscle, and hematopoietic stem cells. A 2018 study investigating the effects of THC, CBD, and CBN on MRP1 transporters found that the presence of a cannabis component increased the concentration of vincristine 3-fold. Additional studies suggest the interaction with the CB1 receptor may lead to changes in the expression of MRP1 transporters.17
CBD inhibits the BCRP transporter, which functions as an efflux pump for methotrexate. Consequently, CBD can increase methotrexate levels, potentially enhancing efficacy but also worsening adverse effects.18
In pancreatic cancer, CBD specifically interacts with gemcitabine. CB1 and CB2 receptors are upregulated, and CBD inhibits the GPR55 receptor. These interactions may enhance the antineoplastic effect of gemcitabine, reducing cell cycle progression and growth.19
CBD also interacts with temozolomide (TMZ) by affecting extracellular vesicles used by cells for pro-oncogenic signaling and immune system evasion. Experiments on patient-derived glioblastoma cells, both chemotherapy-resistant and chemotherapy-sensitive, found that CBD increases the formation of extracellular vesicles with reduced levels of miR21 (pro-oncogenic) and elevated levels of miR126 (antioncogenic).20 CBD has also been found to decrease prohibitin levels, a protein associated with TMZ resistance.
In patients with glioblastoma, CBD combined with chemotherapeutic agents like TMZ, carmustine, doxorubicin, and cisplatin has shown increased sensitivity and improved tumor response. CBD is also known to inhibit NF-kB, a pathway that sustains tumor viability despite chemotherapy.21 Additionally, CBD inhibits the P-glycoprotein system, affecting chemotherapy efflux from neoplastic cells.14 In vitro studies have found that CBD is synergistic with bortezomib in inhibiting cancer cell viability. In another glioblastoma model, CBD enhanced the antiproliferative effects of both TMZ and carmustine.14
Different cannabis formulations may vary in how they interact with various cytotoxic chemotherapeutic agents. Some may potentiate the effects of chemotherapy and act synergistically to inhibit tumor growth, while others may lead to increased toxicity.10 More research is needed to determine which formulations, in combination with specific agents and doses, may have significant interactions that warrant adjustments in chemotherapy dosing.
Cannabis/Immunotherapy Interactions
Cannabis is an immunosuppressant. Data suggest the use of cannabis during immunotherapy worsens treatment outcomes in patients with cancer.22 Exogenous (THC) and endogenous (AEA) CBs negatively affect antitumor immunity by impairing the function of tumor-specific T cells via CB2 and by inhibiting the Jak1-STATs signaling in T cells through CNR2. Xiong et al found that THC reduces the therapeutic effect of anti-PD-1 therapy.22
In a prospective observational clinical study, Bar-Sela et al analyzed 102 patients with advanced cancer—of which 68 were cannabis users—that were started on immune checkpoint inhibitor therapy. The study found that cannabis users on anti-PD-1 (nivolumab, pembrolizumab), anti-CTLA-4 (ipilimumab), and anti-PD-L1 (durvalumab, atezolizumab) had a significant decrease in time to treatment progression and overall survival vs cannabis non-users.23 However, a 2023 study by Waissengrin et al found that concomitant use of medical cannabis with pembrolizumab had no harmful effect in advanced non-small cell lung cancer.24 Time to treatment progression of cannabis users did not differ from cannabis nonusers.25
Cannabis/Endocrine Therapy Interactions
In addition to having direct antineoplastic activity on tumor cells, data exist that show how cannabis affects the endocrine system. In animal models, cannabis has been found to suppress the whole hypothalamic-pituitary-adrenal axis as well as other hormones like thyroid, prolactin, and growth hormone. In breast cancer, cannabis competes with estrogen for the estrogen receptor and suppresses growth.26
The endocrine agents used by patients with cancer in this study were antiandrogens like abiraterone, enzalutamide, tamoxifen and anastrozole. Abiraterone is metabolized by CYP450 isoenzymes and uridine diphosphate glycosyltransferases. Cannabis inhibits both processes and therefore may lead to increased toxicities.27 Conversely, enzalutamide is a strong CYP3A inducer, and cannabis use during enzalutamide therapy may significantly increase the toxic effects of cannabis.
There is evidence that molecular pathways involving CB receptors and estrogens overlap, which may lead to interactions when antiestrogens are used in cannabis users with hormone receptor-positive breast cancer.26 In preclinical studies, tamoxifen has been shown to act as an inverse agonist on CB1 and CB2 receptors, though the significance of this finding is unclear. There is no research evaluating the effects of CBs on tamoxifen treatment. However, CBD has been found to potentiate the effectiveness of anastrozole or exemestane in breast cancer cell lines.28 Dobovišek et al demonstrated no inhibitory effect of CBD on the activity of tamoxifen, fulvestrant, or palbociclib in breast cancer cell lines.29 The interactions between hormone receptor-positive breast cancer and cannabinoids are complex, and the clinical significance of these interactions remains difficult to identify.
Cannabis/Targeted Therapy Interactions
The targeted therapies used by patients in this study included zanubrutinib, ibrutinib, sorafenib, acalabrutinib, dabrafenib, trametinib, trastuzumab, bevacizumab, daratumumab, and imatinib. Compared to other classes of cancer treatments, most studies have not demonstrated decreased efficacy or increased toxicity of targeted anticancer drugs when used concomitantly with CBD.29
Trastuzumab is a recombinant humanized monoclonal antibody that targets the proto-oncogene HER2/neu. It is used to treat select patients with metastatic breast cancer. Studies have shown that cannabis use does not attenuate the effectiveness of trastuzumab in HER2-positive and triple-negative breast cancer subtypes.29 One study found that CBD, in combination with chemotherapeutics and Bruton tyrosine kinase inhibitors, such as ibrutinib and zanubrutinib, has synergistic potential for treating diffuse large B-cell lymphoma and mantle cell lymphoma cell lines. This synergy is attributed to the CB1 antagonist activity of cannabis against diffuse large B-cell lymphoma and mantle cell lymphoma cell lines.30,31
Moreover, combining cannabinoids with bevacizumab (a monoclonal anti-VEGF antibody) has been shown to decrease tumor growth and intratumoral hypoxia in clinically relevant human glioblastoma models. This effect is mediated through the downregulation of HIF-1α.32 Long-term studies evaluating the potential harmful or synergistic potential of CBD on targeted anticancer therapy are needed.
CONCLUSIONS
This exploratory study of patients receiving cancer therapy at WJVAMC found a significant prevalence of concurrent cannabis use among patients undergoing antineoplastic treatments. Given that many antineoplastic agents are metabolized by the CYP450 enzyme system, the findings of this study suggest that concurrent cannabis use may pose risks of suboptimal therapeutic outcomes due to potential interactions affecting drug metabolism. These interactions could impact the efficacy and toxicity of the antineoplastic therapies, potentially leading to diminished therapeutic effects or exacerbated adverse reactions.
Patients should be informed regarding the potential decreased efficacy of immunotherapy with concurrent use of cannabis products. They should also be aware of the possibility of increased toxicity with other treatment modalities, though the exact impact on efficacy remains unclear. This highlights the necessity of caution when combining cannabis with prescribed cancer treatments.
While this study identified possible interactions, its data are preliminary and highlight the need for more rigorous research. Future studies should include larger, well-designed cohorts to compare outcomes between cannabis users and nonusers. Such research is essential to fully elucidate the clinical implications of cannabis use during cancer treatment, address the high prevalence of cannabis use among patients with cancer, and mitigate potential risks associated with combining cannabis products with antineoplastic therapies. This will ensure that treatment strategies are optimized for safety and efficacy in this complex patient population.
- Steele G, Arneson T, Zylla D. A comprehensive review of cannabis in patients with cancer: availability in the USA, general efficacy, and safety. Curr Oncol Rep. 2019;21:1-10. doi:10.1007/s11912-019-0757-7
- Brown D, Watson M, Schloss J. Pharmacological evidence of medicinal cannabis in oncology: a systematic review. Support Care Cancer. 2019;27:3195-320. doi:10.1007/s00520-019-04774-5
- Abrams DI. Integrating cannabis into clinical cancer care. Curr Oncol. 2016;23:S8-S14. doi:10.37.47/co.23.3099
- Serafimovska T, Darkovska-Serafimovska M, Stefkov G, Arsova-Sarafinovska Z, Balkanov T. Pharmacotherapeutic considerations for use of cannabinoids to relieve symptoms of nausea and vomiting induced by chemotherapy. Folia Medica (Plovdiv). 2020;62:668-678. doi:10.3897/folmed.62e51478
- Bar-Sela G, Zalman D, Semenysty V, Ballan E. The effects of dosage-controlled cannabis capsules on cancer-related cachexia and anorexia syndrome in advanced cancer patients: pilot study. Integr Cancer Ther. 2019;18:1534735419881498. doi:10.1177/1534735419881498
- Pederson ER, Villarosa-Hurlocker MC, Prince MA. Use of protective behavioral strategies among young adult veteran marijuana users. Cannabis. 2018;1:14-27.
- Schilling S, Melzer R, McCabe PF. Cannabis sativa. Curr Biol. 2020;30:R8-R9. doi:10.1016/j.cub.2019.10.039
- McDougle DR, Kambalyal A, Meling DD, Das A. Endocannabinoids anandamide and 2-arachidonoylglycerol are substrates for human CYP2J2 epoxygenase. J Pharmacol Exp Ther. 2014;351:616-627. doi:10.1124/jpet.114216598
- Movsesyan VA, Stoica BA, Yakovlev AG, et al. Anandamide-induced cell death in primary neuronal cultures: role of calpain and caspase pathways. Cell Death Differ. 2004;11:1121-1132. doi:10.1038/sj.cdd.4401442
- Cherkasova V, Wang B, Gerasymchuk M, Fiselier A, Kovalchuk O, Kovalchuk I. Use of cannabis and cannabinoids for treatment of cancer. Cancers (Basel). 2022;14:5142. doi:10.3390/cancers14205142
- Engels FK, Ten Tije AJ, Baker SD, et al. Effect of cytochrome P450 3A4 inhibition on the pharmacokinetics of docetaxel. Clin Pharmacol Ther. 2004;75:448-454. doi:10.1016/j.clpt.2004.01.001
- Alsherbiny MA, Li CG. Medicinal cannabis-potential drug interactions. Medicines (Basel). 2018;6:3. doi:10.3390/medicines6010003
- Stout SM, Cimino NM. Exogenous cannabinoids as substrates, inhibitors, and inducers of human drug metabolizing enzymes: a systematic review. Drug Metab Rev. 2014;46:86-95. doi:10.3109/03602532.2013.849268
- Opitz BJ, Ostroff ML, Whitman AC. The potential clinical implications and importance of drug interactions between anticancer agents and cannabidiol in patients with cancer. J Pharm Pract. 2020;33:506-512. doi:10.1177/0897190019828920
- Guzmán M, Duarte MJ, Blázquez C, et al. A pilot clinical study of D9-tetrahydrocannabinol in patients with recurrent glioblastoma multiforme. Br J Cancer. 2006;95:197-203. doi:10.1038/sj.bjc.6603236
- Kopjar N, Fuchs N, Brcic Karaconji I, et al. High doses of ?9-tetrahydrocannabinol might impair irinotecan chemotherapy: a review of potentially harmful interactions. Clin Drug Investig. 2020;40:775-787. doi:10.1007/s40261-020-00954-y
- Bouquié R, Deslandes G, Mazaré H, et al. Cannabis and anticancer drugs: societal usage and expected pharmacological interactions - a review. Fundam Clin Pharmacol. 2018;32:462-484. doi:10.1111/fcp.12373
- Buchtova T, Lukac D, Skrott Z, Chroma K, Bartek J, Mistrik M. Drug-drug interactions of cannabidiol with standard-of-care chemotherapeutics. Int J Mol Sci. 2023;24:2885. doi:10.3390/ijms24032885
- Sharafi G, He H, Nikfarjam M. Potential use of cannabinoids for the treatment of pancreatic cancer. J Pancreat Cancer. 2019;5:1-7. doi:10.1089/pancan.2018.0019
- Kosgodage US, Uysal-Onganer P, MacLatchy A, et al. Cannabidiol affects extracellular vesicle release, miR21 and miR126, and reduces prohibitin protein in glioblastoma multiforme cells. Transl Oncol. 2019;12:513-522. doi:10.1016/j.tranon.2018.12.004
- Elbaz M, Nasser MW, Ravi J, et al. Modulation of the tumor microenvironment and inhibition of EGF/EGFR pathway: novel anti-tumor mechanisms of cannabidiol in breast cancer. Mol Oncol. 2015;9:906-919. doi:10.1016/j.molonc.2014.12.010
- Xiong X, Chen S, Shen J, et al. Cannabis suppresses anti-tumor immunity by inhibiting JAK/STAT signaling in T cells through CNR2. Signal Transduct Target Ther. 2022;7:99. doi:10.1038/s41392-022-00918-y
- Bar-Sela G, Cohen I, Campisi-Pinto S, et al. Cannabis consumption used by cancer patients during immunotherapy correlates with poor clinical outcome. Cancers (Basel). 2020;12:2447. doi:10.3390/cancers12092447
- Waissengrin B, Leshem Y, Taya M, et al. The use of medical cannabis concomitantly with immune checkpoint inhibitors in non-small cell lung cancer: a sigh of relief? Eur J Cancer. 2023;180:52-61. doi:10.1016/j.ejca.2022.11.022
- Sarsembayeva A, Schicho R. Cannabinoids and the endocannabinoid system in immunotherapy: helpful or harmful? Front Oncol. 2023;13:1296906. doi:10.3389/fonc.2023.1296906
- Kisková T, Mungenast F, Suváková M, Jäger W, Thalhammer T. Future aspects for cannabinoids in breast cancer therapy. Int J Mol Sci. 2019;20:1673. doi:10.3390/ijms20071673
- Woerdenbag HJ, Olinga P, Kok EA, et al. Potential, limitations and risks of cannabis-derived products in cancer treatment. Cancers (Basel). 2023;15:2119. doi:10.3390/cancers15072119
- Almeida CF, Teixeira N, Valente MJ, Vinggaard AM, Correia-da-Silva G, Amaral C. Cannabidiol as a promising adjuvant therapy for estrogen receptor-positive breast tumors: unveiling its benefits with aromatase inhibitors. Cancers (Basel). 2023;15:2517. doi:10.3390/cancers15092517
- Dobovišek L, Novak M, Krstanovic F, Borštnar S, Turnšek TL, Debeljak N. Effect of combining CBD with standard breast cancer therapeutics. Adv Cancer Biol Metastasis. 2022;4:100038. doi:10.1016/j.adcanc.2022.100038
- Strong T, Rauvolfova J, Jackson E, Pham LV, Bryant J. Synergistic effect of cannabidiol with conventional chemotherapy treatment. Blood. 2018;132:5382. doi:10.1182/blood-2018-99-116749
- Maggi F, Morelli MB, Tomassoni D, et al. The effects of cannabidiol via TRPV2 channel in chronic myeloid leukemia cells and its combination with imatinib. Cancer Sci. 2022;113:1235-1249. doi:10.1111/cas.15257
- Obad N, Janji B, Prestegarden L, et al. ATPS-59 improving efficacy of bevacizumab treatment in glioblastoma by targeting hif1 alpha. Neuro Oncol. 2015;17:v31. doi:10.1093/neuonc/nov204.59
- Steele G, Arneson T, Zylla D. A comprehensive review of cannabis in patients with cancer: availability in the USA, general efficacy, and safety. Curr Oncol Rep. 2019;21:1-10. doi:10.1007/s11912-019-0757-7
- Brown D, Watson M, Schloss J. Pharmacological evidence of medicinal cannabis in oncology: a systematic review. Support Care Cancer. 2019;27:3195-320. doi:10.1007/s00520-019-04774-5
- Abrams DI. Integrating cannabis into clinical cancer care. Curr Oncol. 2016;23:S8-S14. doi:10.37.47/co.23.3099
- Serafimovska T, Darkovska-Serafimovska M, Stefkov G, Arsova-Sarafinovska Z, Balkanov T. Pharmacotherapeutic considerations for use of cannabinoids to relieve symptoms of nausea and vomiting induced by chemotherapy. Folia Medica (Plovdiv). 2020;62:668-678. doi:10.3897/folmed.62e51478
- Bar-Sela G, Zalman D, Semenysty V, Ballan E. The effects of dosage-controlled cannabis capsules on cancer-related cachexia and anorexia syndrome in advanced cancer patients: pilot study. Integr Cancer Ther. 2019;18:1534735419881498. doi:10.1177/1534735419881498
- Pederson ER, Villarosa-Hurlocker MC, Prince MA. Use of protective behavioral strategies among young adult veteran marijuana users. Cannabis. 2018;1:14-27.
- Schilling S, Melzer R, McCabe PF. Cannabis sativa. Curr Biol. 2020;30:R8-R9. doi:10.1016/j.cub.2019.10.039
- McDougle DR, Kambalyal A, Meling DD, Das A. Endocannabinoids anandamide and 2-arachidonoylglycerol are substrates for human CYP2J2 epoxygenase. J Pharmacol Exp Ther. 2014;351:616-627. doi:10.1124/jpet.114216598
- Movsesyan VA, Stoica BA, Yakovlev AG, et al. Anandamide-induced cell death in primary neuronal cultures: role of calpain and caspase pathways. Cell Death Differ. 2004;11:1121-1132. doi:10.1038/sj.cdd.4401442
- Cherkasova V, Wang B, Gerasymchuk M, Fiselier A, Kovalchuk O, Kovalchuk I. Use of cannabis and cannabinoids for treatment of cancer. Cancers (Basel). 2022;14:5142. doi:10.3390/cancers14205142
- Engels FK, Ten Tije AJ, Baker SD, et al. Effect of cytochrome P450 3A4 inhibition on the pharmacokinetics of docetaxel. Clin Pharmacol Ther. 2004;75:448-454. doi:10.1016/j.clpt.2004.01.001
- Alsherbiny MA, Li CG. Medicinal cannabis-potential drug interactions. Medicines (Basel). 2018;6:3. doi:10.3390/medicines6010003
- Stout SM, Cimino NM. Exogenous cannabinoids as substrates, inhibitors, and inducers of human drug metabolizing enzymes: a systematic review. Drug Metab Rev. 2014;46:86-95. doi:10.3109/03602532.2013.849268
- Opitz BJ, Ostroff ML, Whitman AC. The potential clinical implications and importance of drug interactions between anticancer agents and cannabidiol in patients with cancer. J Pharm Pract. 2020;33:506-512. doi:10.1177/0897190019828920
- Guzmán M, Duarte MJ, Blázquez C, et al. A pilot clinical study of D9-tetrahydrocannabinol in patients with recurrent glioblastoma multiforme. Br J Cancer. 2006;95:197-203. doi:10.1038/sj.bjc.6603236
- Kopjar N, Fuchs N, Brcic Karaconji I, et al. High doses of ?9-tetrahydrocannabinol might impair irinotecan chemotherapy: a review of potentially harmful interactions. Clin Drug Investig. 2020;40:775-787. doi:10.1007/s40261-020-00954-y
- Bouquié R, Deslandes G, Mazaré H, et al. Cannabis and anticancer drugs: societal usage and expected pharmacological interactions - a review. Fundam Clin Pharmacol. 2018;32:462-484. doi:10.1111/fcp.12373
- Buchtova T, Lukac D, Skrott Z, Chroma K, Bartek J, Mistrik M. Drug-drug interactions of cannabidiol with standard-of-care chemotherapeutics. Int J Mol Sci. 2023;24:2885. doi:10.3390/ijms24032885
- Sharafi G, He H, Nikfarjam M. Potential use of cannabinoids for the treatment of pancreatic cancer. J Pancreat Cancer. 2019;5:1-7. doi:10.1089/pancan.2018.0019
- Kosgodage US, Uysal-Onganer P, MacLatchy A, et al. Cannabidiol affects extracellular vesicle release, miR21 and miR126, and reduces prohibitin protein in glioblastoma multiforme cells. Transl Oncol. 2019;12:513-522. doi:10.1016/j.tranon.2018.12.004
- Elbaz M, Nasser MW, Ravi J, et al. Modulation of the tumor microenvironment and inhibition of EGF/EGFR pathway: novel anti-tumor mechanisms of cannabidiol in breast cancer. Mol Oncol. 2015;9:906-919. doi:10.1016/j.molonc.2014.12.010
- Xiong X, Chen S, Shen J, et al. Cannabis suppresses anti-tumor immunity by inhibiting JAK/STAT signaling in T cells through CNR2. Signal Transduct Target Ther. 2022;7:99. doi:10.1038/s41392-022-00918-y
- Bar-Sela G, Cohen I, Campisi-Pinto S, et al. Cannabis consumption used by cancer patients during immunotherapy correlates with poor clinical outcome. Cancers (Basel). 2020;12:2447. doi:10.3390/cancers12092447
- Waissengrin B, Leshem Y, Taya M, et al. The use of medical cannabis concomitantly with immune checkpoint inhibitors in non-small cell lung cancer: a sigh of relief? Eur J Cancer. 2023;180:52-61. doi:10.1016/j.ejca.2022.11.022
- Sarsembayeva A, Schicho R. Cannabinoids and the endocannabinoid system in immunotherapy: helpful or harmful? Front Oncol. 2023;13:1296906. doi:10.3389/fonc.2023.1296906
- Kisková T, Mungenast F, Suváková M, Jäger W, Thalhammer T. Future aspects for cannabinoids in breast cancer therapy. Int J Mol Sci. 2019;20:1673. doi:10.3390/ijms20071673
- Woerdenbag HJ, Olinga P, Kok EA, et al. Potential, limitations and risks of cannabis-derived products in cancer treatment. Cancers (Basel). 2023;15:2119. doi:10.3390/cancers15072119
- Almeida CF, Teixeira N, Valente MJ, Vinggaard AM, Correia-da-Silva G, Amaral C. Cannabidiol as a promising adjuvant therapy for estrogen receptor-positive breast tumors: unveiling its benefits with aromatase inhibitors. Cancers (Basel). 2023;15:2517. doi:10.3390/cancers15092517
- Dobovišek L, Novak M, Krstanovic F, Borštnar S, Turnšek TL, Debeljak N. Effect of combining CBD with standard breast cancer therapeutics. Adv Cancer Biol Metastasis. 2022;4:100038. doi:10.1016/j.adcanc.2022.100038
- Strong T, Rauvolfova J, Jackson E, Pham LV, Bryant J. Synergistic effect of cannabidiol with conventional chemotherapy treatment. Blood. 2018;132:5382. doi:10.1182/blood-2018-99-116749
- Maggi F, Morelli MB, Tomassoni D, et al. The effects of cannabidiol via TRPV2 channel in chronic myeloid leukemia cells and its combination with imatinib. Cancer Sci. 2022;113:1235-1249. doi:10.1111/cas.15257
- Obad N, Janji B, Prestegarden L, et al. ATPS-59 improving efficacy of bevacizumab treatment in glioblastoma by targeting hif1 alpha. Neuro Oncol. 2015;17:v31. doi:10.1093/neuonc/nov204.59
Cannabis Use by Veterans and Potential Interactions With Antineoplastic Agents: Analysis and Literature Review
Cannabis Use by Veterans and Potential Interactions With Antineoplastic Agents: Analysis and Literature Review
Investigating Real-World Tolerance and Dose Reductions of Oncology Multikinase Inhibitors in a VA Population
Investigating Real-World Tolerance and Dose Reductions of Oncology Multikinase Inhibitors in a VA Population
The US Department of Veterans Affairs (VA) annually treats around 450,000 veterans with cancer and diagnoses an additional 56,000.1,2 Oral multikinase inhibitors (MKIs) are widely used as targeted therapies for many different malignancies. Despite the ease of oral administration, these agents are often accompanied by significant adverse effects (AEs) and drug-drug interactions.3,4 Common AEs include hypertension, cutaneous reactions, gastrointestinal disturbances, proteinuria, and fatigue. Some serious outcomes that may occur are myocardial infarction, thrombosis, nephrotic syndrome, hemorrhage, hepatotoxicity, and gastrointestinal events.5,6 Due to poor tolerability of these AEs, dose reductions, frequent therapy holds, and discontinuation of therapy may occur.
The US Food and Drug Administration recognizes dosing challenges with novel therapies and has created the Oncology Center of Excellence (OCE) Project Optimus initiative to reform dose optimization in oncology drug development. The initiative aims to shift the focus from establishing dose regimens based on the maximum tolerated doses of cytotoxic chemotherapeutics to an emphasis on maximum efficacy, safety, and tolerability, which better reflect real-world dosing.7,8
MKIs can be challenging to manage because of the frequent toxicity-related dose reductions, interruptions, and discontinuations. In a multicenter retrospective study, Schnadig et al investigated dosing characteristics of first-line sunitinib for advanced renal cell carcinoma (RCC) and found that, among 114 patients who experienced AEs while taking sunitinib, 39.5% had dose reductions, 5.3% delayed therapy, 18.4% required additional supportive medications, and 22.8% discontinued sunitinib.9 Overall survival and median progression-free survival of these patients were lower than reported by Motzer et al in a phase 3 clinical trial.10 Schnadig et al concluded that patients treated with sunitinib for RCC in the community setting required more frequent dose reductions and had less time on therapy compared with patients in clinical trials, which ultimately impacted clinical outcomes.9
At the VA North Texas Health Care System (VANTHCS), patients with cancer have difficulty tolerating MKIs and often require dose alterations and/or discontinuation because of drug intolerance rather than discontinuation due to progression. Frequent dose adjustments for toxicity management can place more strain on patients and health care resources because of additional appointments, clinician time, and emergency department visits. Escalating drug costs can also cause concern when prescription doses are unused or changed frequently.
To capture and quantify prescribing practices and dose adjustments, this study evaluated the tolerability of MKIs at VANTHCS. This analysis may also guide clinicians in the selection of starting doses as well as dose titration expectations to optimize MKI therapy.
METHODS
This single-center, retrospective chart review analyzed patients receiving oral oncology MKIs for various malignancies at VANTHCS between January 1, 2014, and October 31, 2024. Participants included adults aged ≥ 18 years with a prescription for axitinib, cabozantinib, lenvatinib, pazopanib, regorafenib, sorafenib, or sunitinib initiated by the hematology/oncology service at VANTHCS. Patients were included if they had follow-up documentation with the hematology/oncology service and/or other VANTHCS clinicians outlining their course of therapy after MKI initiation. Patients were excluded if they did not have sufficient follow-up documentation (eg, transferred care to a non-VA health care practitioner [HCP], moved to another VA health care system), were enrolled in clinical trials, or were prescribed an MKI from a Care in the Community (CITC) prescriber. Electronic health record review and data collection were performed using the VA Computerized Patient Record System and Research Electronic Data Capture. Data were collected from the time of initiation to cessation of therapy and included information regarding therapy changes, progressive disease, and date of death, when available. Data collected included age, sex, race, comorbidities, date of death, type of malignancy and subtypes, cancer stage, MKI used (ie, drug, dose, frequency, schedule, and indication), dates of medication changes (ie, start, adjustment, hold, discontinuation), concurrent antineoplastic treatments, and AEs documented at times of dose change or interruption.
The primary outcome was MKI tolerance determined using relative dose intensity (RDI) and mean and median time on therapy. Two methods are used to calculate RDI that vary in how they approach time on therapy as outlined in Hawn et al.11 This study used method 2, which accounts for holds in therapy by comparing the actual duration of treatment with the duration expected according to treatment protocol. Method 1 compares the prescribed dose with the administered dose and does not adjust for holds.11 Using method 2, the RDI in this study was calculated by dividing the total actual dose given by the total indicated dose for the malignancy being treated, which accounts for duration of treatment.

The total actual dose was the strength, frequency, and days on therapy for each time frame of treatment multiplied together. This method accounted for all dose adjustments and time periods of treatment holds, including patient self-adjustments, prescriber-directed adjustments, and nonadherence determined by HCP documentation and/or prescription data. Similarly, the indicated total dose was calculated by multiplying the indicated strength, frequency, and all days that treatment should have occurred (time from start to finish). Indicated doses were derived from the prescribing information for each malignancy with the exception of sunitinib, for which the off-label dose of 37.5 mg daily was considered a full dose.12,13 The total indicated dose for axitinib was calculated by considering the dose escalation schedule from the prescribing information.
Patients who required dose reductions due to renal/hepatic impairments or drug-drug interactions had their total indicated dose calculated using dose adjustments listed in the prescribing information. The mean RDI for each MKI agent was calculated by averaging the RDI for each prescription. The overall combined mean RDI included the means of all the MKIs reviewed to avoid skewing the results toward an MKI with more prescriptions. RDIs were also calculated for each cancer type for each agent. Additional descriptive secondary outcomes included rates of AEs and adjustments in doses.
RESULTS
Electronic data extraction identified 278 patients with 366 MKI prescriptions, of which 108 veterans with 158 MKI prescriptions were excluded. The top reason for exclusion was patients managed through CITC. Ultimately, 170 veterans with 208 MKI prescriptions managed by the VANTHCS hematology/oncology clinic were included (Table 1). Among patients receiving MKIs, the mean age was 72.7 years, 98% were male, and 99% had metastatic disease.
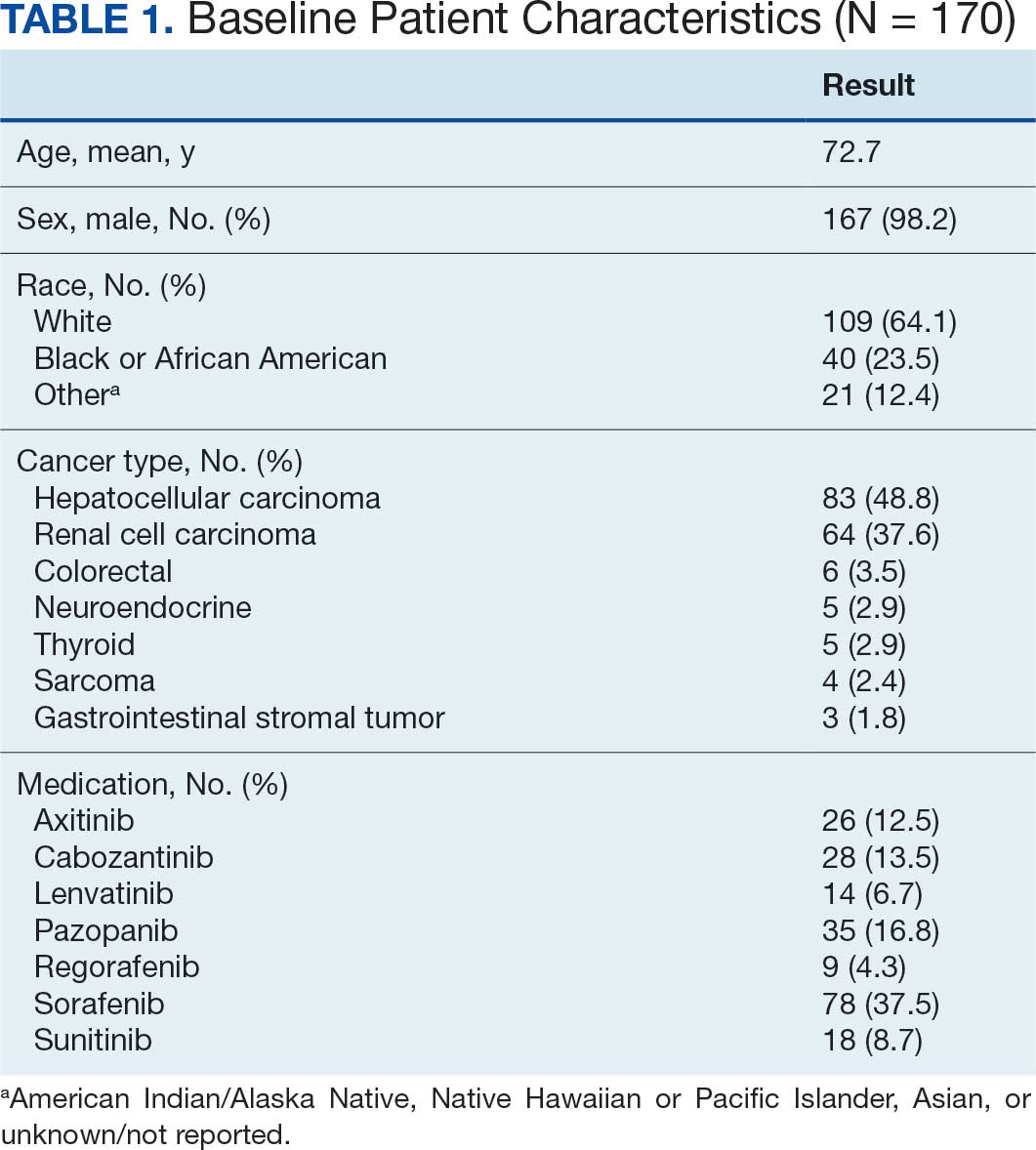
The overall combined mean MKI RDI was 67.5% using method 2 and ranged from 85.5% for sunitinib to 49.0% for sorafenib (Figure 1). Additional information regarding mean and median RDIs using method 2 is shown in Figure 1 and further subdivided by cancer type in Table 2. Median RDIs overall were similar to mean RDIs for most agents. Figure 2 indicates the mean and median time on therapy, reflecting time on therapy excluding days therapy was held. The overall combined mean and median days on therapy for all MKIs were 155 days and 95 days, respectively. Mean time on therapy depended on the agent used and ranged from 35 days (regorafenib) to 237 days (cabozantinib).
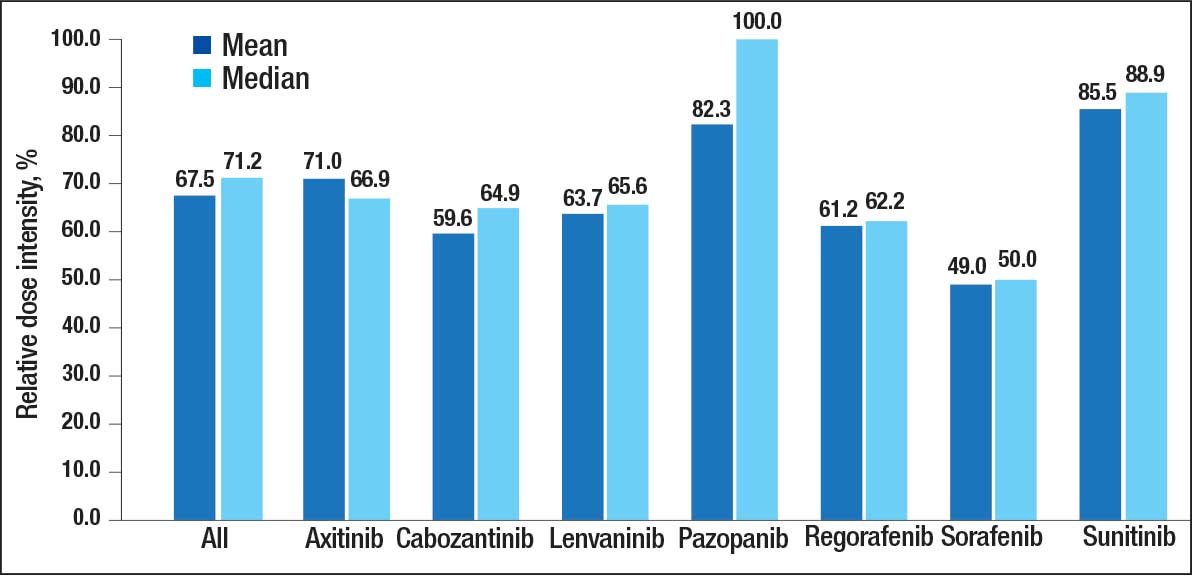
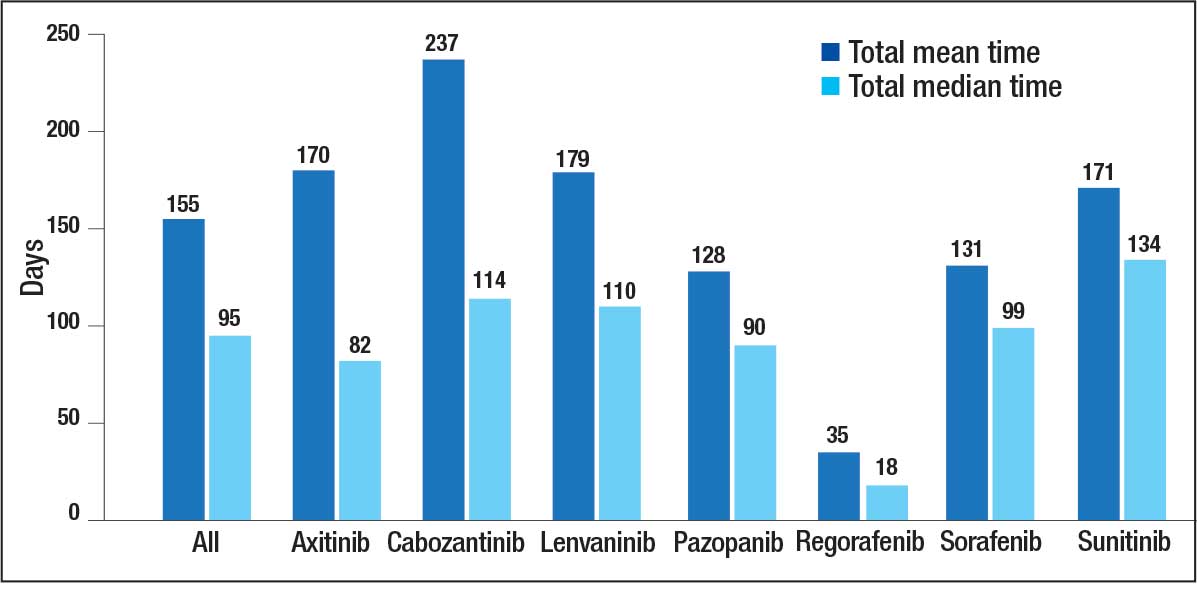
Of 208 MKI prescriptions, 127 (61.1%) were initiated at a reduced dose due to baseline concerns for tolerance such as performance status, frailty, and prior intolerance of other treatments. Eighty-one prescriptions (38.9%) were initiated at their indicated doses. Ninety prescriptions (43.3%) required dose reductions during treatment. Some MKI prescriptions had multiple dose increases and decreases, which is why RDI more accurately reflects dose adjustments. A total of 376 AEs that contributed to a dose adjustment, hold, or discontinuation occurred across all MKI prescriptions. The most common AEs were 82 failure-to-thrive events (21.8%) (fatigue, malaise, loss of appetite, reduced mobility, global decline), 79 gastrointestinal events (21.0%) (nausea, vomiting, diarrhea, abdominal pain), 62 dermatologic events (16.5%) (rash, hand-foot skin reactions, allergic response), 61 hepatic dysfunction events (16.2%) (liver enzyme elevations, hyperbilirubinemia), 40 cardiovascular events (10.6%) (hypertension, heart failure exacerbations, edema), and 33 renal dysfunction events (8.8%) (acute kidney injury, proteinuria) (Appendix 1).
DISCUSSION
The mean RDI of MKI prescriptions used in the veteran population at VANTHCS was about two-thirds of the indicated dose. These results indicate that most veterans required dose reductions and/or holds due to concerns over initial tolerance/performance status, worsening clinical condition, and/or intolerable AEs attributed to treatment. A retrospective study conducted by Denduluri et al suggested that an RDI of < 85% is a clinically meaningful reduction for traditional chemotherapy based on previous literature.14 However, it is less clear what RDI should be expected specifically for MKIs in real-world populations. The MKI phase 3 approval trials in RCC for axitinib, lenvatinib, and sunitinib found median RDIs of 89.4%, 69.6% to 70.4%, and 83.9%, respectively. Each trial cited dose reductions most commonly as the result of treatment-related AEs.15,16
Studies on the impact of RDIs on survival outcomes found that higher RDIs may improve overall and progression-free survival. Retrospective studies inspecting lenvatinib in hepatocellular carcinoma (HCC) indicated that an RDI > 70% in the initial 4 weeks resulted in favorable survival outcomes.17 Similarly, a retrospective study investigating sunitinib in RCC found that an RDI > 60% conferred favorable survival outcomes.18 Alghamdi et al noted that patients taking sorafenib for HCC who had RDI > 50% had a favorable trend in survival characteristics. Interestingly, the study found an RDI of 50% to 75% appeared to have better survival than an RDI > 75%.19 The authors of these studies hypothesized that additional dose reductions allowed for longer total time on therapy due to improved tolerability.17-19
This analysis found that the RDIs for most MKI agents at VANTHCS were < 85% and lower than the RDIs found in other review articles and phase 3 trials, with the exceptions of pazopanib in thyroid cancer and sunitinib in gastrointestinal stromal tumor (GIST), thyroid cancer, and neuroendocrine cancer. The reasons for the lower RDIs in this study are likely multifactorial, reflecting patient population characteristics, off-label dosing practices, and HCP experiences with these agents. Many veterans have chronic comorbidities that could contribute to reduced performance status and ability to tolerate these therapies. Despite attempts to preemptively reduce doses for patients and account for potential impaired tolerance, there were patients who required further dose reductions in our study.
Failure to thrive was the most common AE leading to dose adjustment or discontinuation, which illustrates the extensive effects these agents have on patient functioning in a real-world population. Notably though, the RDI for sunitinib was higher in this population because about half of patients were dosed using the off-label recommendation, whereas the prescribing information recommends a more intensive 6-week dosing cycle for certain cancer types.12,13,20 Sorafenib was also often dose-adjusted based on a pharmacokinetic study of sorafenib in renal/hepatic dysfunction, and the RDI likely reflects the off-label prescribing pattern.21
Patients with thyroid cancer were found to have higher RDIs compared with those receiving the same agents for other cancer types. Improved tolerability of MKIs in thyroid cancer may be due to a generally more tolerable disease course. Thyroid cancer is the most common cancer in individuals aged < 40 years, a population that is often more robust with fewer comorbidities. Moreover, the 5-year relative survival rate for thyroid cancer remains > 98%.22 This rate is in contrast to those for other cancer types such as HCC, with a 5-year relative survival rate of only 15%.23
It is challenging to compare the mean and median times on therapy found in this study with those in current literature, as this review included multiple different cancer types for each agent. However, the numbers are generally lower than durations of therapy found across the different disease states and further emphasize the difficulty in tolerating MKIs in the VANTHCS population. Regorafenib had a short duration of time on therapy, which highlights the importance of trials like ReDOS and initiatives such as OCE Project Optimus in helping improve tolerance.7,8,24
Comparing our results with other studies proved challenging because the RDI calculation methods were not specified. Calculating RDIs in this study using method 1, which does not account for holds, resulted in higher RDIs (Appendix 2). Using method 1, all MKIs had RDIs < 85%, except for pazopanib in thyroid cancer (100%) and RCC (87.9%), and sunitinib in GIST (93.6%), thyroid cancer (100%), and neuroendocrine cancer (93.7%). Notably, using method 1 increased the RDI for pazopanib in neuroendocrine cancer from 5.4% to 50.0%. The low RDI was attributed to a single veteran with a long hold duration, which demonstrates the discrepancy that can occur between the 2 methods.
Limitations
The retrospective design, lack of survival outcomes, and difficulty comparing results with other literature were limitations of this study. Because survival outcomes were not evaluated, future research should seek to investigate how RDIs and dose adjustments made among MKIs can affect survival outcomes in real-world populations. This veteran population with cancer often had multiple chronic comorbidities, which may have contributed to difficulty tolerating MKIs and could have impacted results. Disease-related factors may have influenced the poor tolerance of the MKIs and were not specifically accounted for. Adjustment for comorbidities was not possible because of discrepancies and/or incomplete diagnosis codes and Eastern Cooperative Oncology Group performance status scores documented in patient charts. Therefore, we decided not to report these findings due to potential inaccuracies.
CONCLUSIONS
Results of this study demonstrate that oncology MKI agents used at VANTHCS were difficult for patients to tolerate, leading to suboptimal dosing compared with indicated doses established in clinical trials and prescribing information. Clinicians may use these data to help guide clinical decision-making whenever initiating and managing MKI agents in this population. These findings reinforce that MKI agents are often difficult to tolerate in real-world practice, and indicated doses are often not achieved. Further studies should aim to investigate the effect that various RDIs have on overall survival. Further investigation into different dosing schemes for MKIs to improve tolerability and longer-term use may also prove beneficial.
This analysis may help guide clinicians to carefully approach dosing MKI agents in the veteran population. Given the RDI and AEs, more clinicians may consider starting at lower than indicated doses with the goal to titrate up as tolerated. Additionally, the results highlight the importance of considering palliative care consults and ensuring appropriate supportive care agents are preemptively engaged and adjusted as needed. Approaching dosing and titrations cautiously may help reduce the burden of management on the health care system.
- Frequently asked questions. VA National Oncology Program. 2025. Accessed December 15, 2025. https://www.cancer.va.gov/CANCER/faqs.html
- Torez L. Reigniting the cancer moonshot to beat cancer. VA News. April 20, 2023. Accessed April 6, 2026. https://news.va.gov/118378/reigniting-the-cancer-moonshot-to-beat-cancer
- Shah NN, Casella E, Capozzi D, et al. Improving the safety of oral chemotherapy at an academic medical center. J Oncol Pract. 2016;12:e71-e76. doi:10.1200/JOP.2015.007260
- Hussaarts KGAM, Veerman GDM, Jansman FGA, et al. Clinically relevant drug interactions with multikinase inhibitors: a review. Ther Adv Med Oncol. 2019;11:1758835918818347. doi:10.1177/1758835918818347
- Shyam Sunder S, Sharma UC, Pokharel S. Adverse effects of tyrosine kinase inhibitors in cancer therapy: pathophysiology, mechanisms and clinical management. Signal Transduct Target Ther. 2023;8:262. doi:10.1038/s41392-023-01469-6
- Thomson RJ, Moshirfar M, Ronquillo Y. Tyrosine kinase inhibitors. In: StatPearls [Internet]. StatPearls Publishing; updated July 18, 2023. Accessed December 15, 2025. https://www.ncbi.nlm.nih.gov/books/NBK563322/
- Project Optimus. US Food and Drug Administration. Updated December 6, 2024. Accessed December 15, 2025. https://www.fda.gov/about-fda/oncology-center-excellence/project-optimus
- Optimizing the dosage of human prescription drugs and biological products for the treatment of oncologic diseases: Guidance for Industry. Docket number FDA-2022-D-2827. US Food and Drug Administration. August 2024. Accessed December 15, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/optimizing-dosage-human-prescription-drugs-and-biological-products-treatment-oncologic-diseases
- Schnadig ID, Hutson TE, Chung H, et al. Dosing patterns, toxicity, and outcomes in patients treated with first-line sunitinib for advanced renal cell carcinoma in community-based practices. Clin Genitourin Cancer. 2014;12:413-421. doi:10.1016/j.clgc.2014.06.015
- Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115-124. doi:10.1056/nejmoa065044
- Hawn C, Bansal D. Relative dose intensity in oncology trials: a discussion of two approaches. PharmaSUG. 2024. Accessed April 6, 2026. https://pharmasug.org/proceedings/2024/ST/PharmaSUG-2024-ST-297.pdf
- George S, Merriam P, Maki RG, et al. Multicenter phase II trial of sunitinib in the treatment of nongastrointestinal stromal tumor sarcomas. J Clin Oncol. 2009;27:3154-3160. doi:10.1200/jco.2008.20.9890
- George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer. 2009;45:1959-1968. doi:10.1016/j.ejca.2009.02.011
- Denduluri N, Patt DA, Wang Y, et al. Dose delays, dose reductions, and relative dose intensity in patients with cancer who received adjuvant or neoadjuvant chemotherapy in community oncology practices. J Natl Compr Canc Netw. 2015;13:1383-1393. doi:10.6004/jnccn.2015.0166
- Motzer RJ, Penkov K, Haanen J, et al. Avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380:1103-1115. doi:10.1056/nejmoa1816047
- Motzer R, Alekseev B, Rha SY, et al. Lenvatinib plus pembrolizumab or everolimus for advanced renal cell carcinoma. N Engl J Med. 2021;384:1289-1300. doi:10.1056/nejmoa2035716
- Kirino S, Tsuchiya K, Kurosaki M, et al. Relative dose intensity over the first four weeks of lenvatinib therapy is a factor of favorable response and overall survival in patients with unresectable hepatocellular carcinoma. PloS One. 2020;15:e0231828. doi:10.1371/journal.pone.0231828
- Ishihara H, Takagi T, Kondo T, et al. Decreased relative dose intensity during the early phase of treatment impacts the therapeutic efficacy of sunitinib in metastatic renal cell carcinoma. Jpn J Clin Oncol. 2018;48:667-672. doi:10.1093/jjco/hyy078
- Alghamdi MA, Amaro CP, Lee-Ying R, et al. Effect of sorafenib starting dose and dose intensity on survival in patients with hepatocellular carcinoma: results from a Canadian Multicenter Database. Cancer Med. 2020;9:4918-4928. doi:10.1002/cam4.3228
- Motzer RJ, Rini BI, Bukowski RM, et al. Sunitinib in patients with metastatic renal cell carcinoma. JAMA. 2006;295:2516-2524. doi:10.1001/jama.295.21.2516
- Miller AA, Murry DJ, Owzar K, et al. Phase I and pharmacokinetic study of sorafenib in patients with hepatic or renal dysfunction: CALGB 60301. J Clin Oncol. 2009;27:1800-1805. doi:10.1200/jco.2008.20.0931
- Boucai L, Zafereo M, Cabanillas ME. Thyroid cancer: a review. JAMA. 2024;331:425-435. doi:10.1001/jama.2023.26348
- Amin N, Anwar J, Sulaiman A, et al. Hepatocellular carcinoma: a comprehensive review. Diseases. 2025;13:207. doi:10.3390/diseases13070207
- Bekaii-Saab TS, Ou FS, Ahn DH, et al. Regorafenib dose-optimisation in patients with refractory metastatic colorectal cancer (ReDOS): a randomised, multicentre, open-label, phase 2 study. Lancet Oncol. 2019;20:1070-1082. doi:10.1016/s1470-2045(19)30272-4
The US Department of Veterans Affairs (VA) annually treats around 450,000 veterans with cancer and diagnoses an additional 56,000.1,2 Oral multikinase inhibitors (MKIs) are widely used as targeted therapies for many different malignancies. Despite the ease of oral administration, these agents are often accompanied by significant adverse effects (AEs) and drug-drug interactions.3,4 Common AEs include hypertension, cutaneous reactions, gastrointestinal disturbances, proteinuria, and fatigue. Some serious outcomes that may occur are myocardial infarction, thrombosis, nephrotic syndrome, hemorrhage, hepatotoxicity, and gastrointestinal events.5,6 Due to poor tolerability of these AEs, dose reductions, frequent therapy holds, and discontinuation of therapy may occur.
The US Food and Drug Administration recognizes dosing challenges with novel therapies and has created the Oncology Center of Excellence (OCE) Project Optimus initiative to reform dose optimization in oncology drug development. The initiative aims to shift the focus from establishing dose regimens based on the maximum tolerated doses of cytotoxic chemotherapeutics to an emphasis on maximum efficacy, safety, and tolerability, which better reflect real-world dosing.7,8
MKIs can be challenging to manage because of the frequent toxicity-related dose reductions, interruptions, and discontinuations. In a multicenter retrospective study, Schnadig et al investigated dosing characteristics of first-line sunitinib for advanced renal cell carcinoma (RCC) and found that, among 114 patients who experienced AEs while taking sunitinib, 39.5% had dose reductions, 5.3% delayed therapy, 18.4% required additional supportive medications, and 22.8% discontinued sunitinib.9 Overall survival and median progression-free survival of these patients were lower than reported by Motzer et al in a phase 3 clinical trial.10 Schnadig et al concluded that patients treated with sunitinib for RCC in the community setting required more frequent dose reductions and had less time on therapy compared with patients in clinical trials, which ultimately impacted clinical outcomes.9
At the VA North Texas Health Care System (VANTHCS), patients with cancer have difficulty tolerating MKIs and often require dose alterations and/or discontinuation because of drug intolerance rather than discontinuation due to progression. Frequent dose adjustments for toxicity management can place more strain on patients and health care resources because of additional appointments, clinician time, and emergency department visits. Escalating drug costs can also cause concern when prescription doses are unused or changed frequently.
To capture and quantify prescribing practices and dose adjustments, this study evaluated the tolerability of MKIs at VANTHCS. This analysis may also guide clinicians in the selection of starting doses as well as dose titration expectations to optimize MKI therapy.
METHODS
This single-center, retrospective chart review analyzed patients receiving oral oncology MKIs for various malignancies at VANTHCS between January 1, 2014, and October 31, 2024. Participants included adults aged ≥ 18 years with a prescription for axitinib, cabozantinib, lenvatinib, pazopanib, regorafenib, sorafenib, or sunitinib initiated by the hematology/oncology service at VANTHCS. Patients were included if they had follow-up documentation with the hematology/oncology service and/or other VANTHCS clinicians outlining their course of therapy after MKI initiation. Patients were excluded if they did not have sufficient follow-up documentation (eg, transferred care to a non-VA health care practitioner [HCP], moved to another VA health care system), were enrolled in clinical trials, or were prescribed an MKI from a Care in the Community (CITC) prescriber. Electronic health record review and data collection were performed using the VA Computerized Patient Record System and Research Electronic Data Capture. Data were collected from the time of initiation to cessation of therapy and included information regarding therapy changes, progressive disease, and date of death, when available. Data collected included age, sex, race, comorbidities, date of death, type of malignancy and subtypes, cancer stage, MKI used (ie, drug, dose, frequency, schedule, and indication), dates of medication changes (ie, start, adjustment, hold, discontinuation), concurrent antineoplastic treatments, and AEs documented at times of dose change or interruption.
The primary outcome was MKI tolerance determined using relative dose intensity (RDI) and mean and median time on therapy. Two methods are used to calculate RDI that vary in how they approach time on therapy as outlined in Hawn et al.11 This study used method 2, which accounts for holds in therapy by comparing the actual duration of treatment with the duration expected according to treatment protocol. Method 1 compares the prescribed dose with the administered dose and does not adjust for holds.11 Using method 2, the RDI in this study was calculated by dividing the total actual dose given by the total indicated dose for the malignancy being treated, which accounts for duration of treatment.

The total actual dose was the strength, frequency, and days on therapy for each time frame of treatment multiplied together. This method accounted for all dose adjustments and time periods of treatment holds, including patient self-adjustments, prescriber-directed adjustments, and nonadherence determined by HCP documentation and/or prescription data. Similarly, the indicated total dose was calculated by multiplying the indicated strength, frequency, and all days that treatment should have occurred (time from start to finish). Indicated doses were derived from the prescribing information for each malignancy with the exception of sunitinib, for which the off-label dose of 37.5 mg daily was considered a full dose.12,13 The total indicated dose for axitinib was calculated by considering the dose escalation schedule from the prescribing information.
Patients who required dose reductions due to renal/hepatic impairments or drug-drug interactions had their total indicated dose calculated using dose adjustments listed in the prescribing information. The mean RDI for each MKI agent was calculated by averaging the RDI for each prescription. The overall combined mean RDI included the means of all the MKIs reviewed to avoid skewing the results toward an MKI with more prescriptions. RDIs were also calculated for each cancer type for each agent. Additional descriptive secondary outcomes included rates of AEs and adjustments in doses.
RESULTS
Electronic data extraction identified 278 patients with 366 MKI prescriptions, of which 108 veterans with 158 MKI prescriptions were excluded. The top reason for exclusion was patients managed through CITC. Ultimately, 170 veterans with 208 MKI prescriptions managed by the VANTHCS hematology/oncology clinic were included (Table 1). Among patients receiving MKIs, the mean age was 72.7 years, 98% were male, and 99% had metastatic disease.

The overall combined mean MKI RDI was 67.5% using method 2 and ranged from 85.5% for sunitinib to 49.0% for sorafenib (Figure 1). Additional information regarding mean and median RDIs using method 2 is shown in Figure 1 and further subdivided by cancer type in Table 2. Median RDIs overall were similar to mean RDIs for most agents. Figure 2 indicates the mean and median time on therapy, reflecting time on therapy excluding days therapy was held. The overall combined mean and median days on therapy for all MKIs were 155 days and 95 days, respectively. Mean time on therapy depended on the agent used and ranged from 35 days (regorafenib) to 237 days (cabozantinib).



Of 208 MKI prescriptions, 127 (61.1%) were initiated at a reduced dose due to baseline concerns for tolerance such as performance status, frailty, and prior intolerance of other treatments. Eighty-one prescriptions (38.9%) were initiated at their indicated doses. Ninety prescriptions (43.3%) required dose reductions during treatment. Some MKI prescriptions had multiple dose increases and decreases, which is why RDI more accurately reflects dose adjustments. A total of 376 AEs that contributed to a dose adjustment, hold, or discontinuation occurred across all MKI prescriptions. The most common AEs were 82 failure-to-thrive events (21.8%) (fatigue, malaise, loss of appetite, reduced mobility, global decline), 79 gastrointestinal events (21.0%) (nausea, vomiting, diarrhea, abdominal pain), 62 dermatologic events (16.5%) (rash, hand-foot skin reactions, allergic response), 61 hepatic dysfunction events (16.2%) (liver enzyme elevations, hyperbilirubinemia), 40 cardiovascular events (10.6%) (hypertension, heart failure exacerbations, edema), and 33 renal dysfunction events (8.8%) (acute kidney injury, proteinuria) (Appendix 1).

DISCUSSION
The mean RDI of MKI prescriptions used in the veteran population at VANTHCS was about two-thirds of the indicated dose. These results indicate that most veterans required dose reductions and/or holds due to concerns over initial tolerance/performance status, worsening clinical condition, and/or intolerable AEs attributed to treatment. A retrospective study conducted by Denduluri et al suggested that an RDI of < 85% is a clinically meaningful reduction for traditional chemotherapy based on previous literature.14 However, it is less clear what RDI should be expected specifically for MKIs in real-world populations. The MKI phase 3 approval trials in RCC for axitinib, lenvatinib, and sunitinib found median RDIs of 89.4%, 69.6% to 70.4%, and 83.9%, respectively. Each trial cited dose reductions most commonly as the result of treatment-related AEs.15,16
Studies on the impact of RDIs on survival outcomes found that higher RDIs may improve overall and progression-free survival. Retrospective studies inspecting lenvatinib in hepatocellular carcinoma (HCC) indicated that an RDI > 70% in the initial 4 weeks resulted in favorable survival outcomes.17 Similarly, a retrospective study investigating sunitinib in RCC found that an RDI > 60% conferred favorable survival outcomes.18 Alghamdi et al noted that patients taking sorafenib for HCC who had RDI > 50% had a favorable trend in survival characteristics. Interestingly, the study found an RDI of 50% to 75% appeared to have better survival than an RDI > 75%.19 The authors of these studies hypothesized that additional dose reductions allowed for longer total time on therapy due to improved tolerability.17-19
This analysis found that the RDIs for most MKI agents at VANTHCS were < 85% and lower than the RDIs found in other review articles and phase 3 trials, with the exceptions of pazopanib in thyroid cancer and sunitinib in gastrointestinal stromal tumor (GIST), thyroid cancer, and neuroendocrine cancer. The reasons for the lower RDIs in this study are likely multifactorial, reflecting patient population characteristics, off-label dosing practices, and HCP experiences with these agents. Many veterans have chronic comorbidities that could contribute to reduced performance status and ability to tolerate these therapies. Despite attempts to preemptively reduce doses for patients and account for potential impaired tolerance, there were patients who required further dose reductions in our study.
Failure to thrive was the most common AE leading to dose adjustment or discontinuation, which illustrates the extensive effects these agents have on patient functioning in a real-world population. Notably though, the RDI for sunitinib was higher in this population because about half of patients were dosed using the off-label recommendation, whereas the prescribing information recommends a more intensive 6-week dosing cycle for certain cancer types.12,13,20 Sorafenib was also often dose-adjusted based on a pharmacokinetic study of sorafenib in renal/hepatic dysfunction, and the RDI likely reflects the off-label prescribing pattern.21
Patients with thyroid cancer were found to have higher RDIs compared with those receiving the same agents for other cancer types. Improved tolerability of MKIs in thyroid cancer may be due to a generally more tolerable disease course. Thyroid cancer is the most common cancer in individuals aged < 40 years, a population that is often more robust with fewer comorbidities. Moreover, the 5-year relative survival rate for thyroid cancer remains > 98%.22 This rate is in contrast to those for other cancer types such as HCC, with a 5-year relative survival rate of only 15%.23
It is challenging to compare the mean and median times on therapy found in this study with those in current literature, as this review included multiple different cancer types for each agent. However, the numbers are generally lower than durations of therapy found across the different disease states and further emphasize the difficulty in tolerating MKIs in the VANTHCS population. Regorafenib had a short duration of time on therapy, which highlights the importance of trials like ReDOS and initiatives such as OCE Project Optimus in helping improve tolerance.7,8,24
Comparing our results with other studies proved challenging because the RDI calculation methods were not specified. Calculating RDIs in this study using method 1, which does not account for holds, resulted in higher RDIs (Appendix 2). Using method 1, all MKIs had RDIs < 85%, except for pazopanib in thyroid cancer (100%) and RCC (87.9%), and sunitinib in GIST (93.6%), thyroid cancer (100%), and neuroendocrine cancer (93.7%). Notably, using method 1 increased the RDI for pazopanib in neuroendocrine cancer from 5.4% to 50.0%. The low RDI was attributed to a single veteran with a long hold duration, which demonstrates the discrepancy that can occur between the 2 methods.

Limitations
The retrospective design, lack of survival outcomes, and difficulty comparing results with other literature were limitations of this study. Because survival outcomes were not evaluated, future research should seek to investigate how RDIs and dose adjustments made among MKIs can affect survival outcomes in real-world populations. This veteran population with cancer often had multiple chronic comorbidities, which may have contributed to difficulty tolerating MKIs and could have impacted results. Disease-related factors may have influenced the poor tolerance of the MKIs and were not specifically accounted for. Adjustment for comorbidities was not possible because of discrepancies and/or incomplete diagnosis codes and Eastern Cooperative Oncology Group performance status scores documented in patient charts. Therefore, we decided not to report these findings due to potential inaccuracies.
CONCLUSIONS
Results of this study demonstrate that oncology MKI agents used at VANTHCS were difficult for patients to tolerate, leading to suboptimal dosing compared with indicated doses established in clinical trials and prescribing information. Clinicians may use these data to help guide clinical decision-making whenever initiating and managing MKI agents in this population. These findings reinforce that MKI agents are often difficult to tolerate in real-world practice, and indicated doses are often not achieved. Further studies should aim to investigate the effect that various RDIs have on overall survival. Further investigation into different dosing schemes for MKIs to improve tolerability and longer-term use may also prove beneficial.
This analysis may help guide clinicians to carefully approach dosing MKI agents in the veteran population. Given the RDI and AEs, more clinicians may consider starting at lower than indicated doses with the goal to titrate up as tolerated. Additionally, the results highlight the importance of considering palliative care consults and ensuring appropriate supportive care agents are preemptively engaged and adjusted as needed. Approaching dosing and titrations cautiously may help reduce the burden of management on the health care system.
The US Department of Veterans Affairs (VA) annually treats around 450,000 veterans with cancer and diagnoses an additional 56,000.1,2 Oral multikinase inhibitors (MKIs) are widely used as targeted therapies for many different malignancies. Despite the ease of oral administration, these agents are often accompanied by significant adverse effects (AEs) and drug-drug interactions.3,4 Common AEs include hypertension, cutaneous reactions, gastrointestinal disturbances, proteinuria, and fatigue. Some serious outcomes that may occur are myocardial infarction, thrombosis, nephrotic syndrome, hemorrhage, hepatotoxicity, and gastrointestinal events.5,6 Due to poor tolerability of these AEs, dose reductions, frequent therapy holds, and discontinuation of therapy may occur.
The US Food and Drug Administration recognizes dosing challenges with novel therapies and has created the Oncology Center of Excellence (OCE) Project Optimus initiative to reform dose optimization in oncology drug development. The initiative aims to shift the focus from establishing dose regimens based on the maximum tolerated doses of cytotoxic chemotherapeutics to an emphasis on maximum efficacy, safety, and tolerability, which better reflect real-world dosing.7,8
MKIs can be challenging to manage because of the frequent toxicity-related dose reductions, interruptions, and discontinuations. In a multicenter retrospective study, Schnadig et al investigated dosing characteristics of first-line sunitinib for advanced renal cell carcinoma (RCC) and found that, among 114 patients who experienced AEs while taking sunitinib, 39.5% had dose reductions, 5.3% delayed therapy, 18.4% required additional supportive medications, and 22.8% discontinued sunitinib.9 Overall survival and median progression-free survival of these patients were lower than reported by Motzer et al in a phase 3 clinical trial.10 Schnadig et al concluded that patients treated with sunitinib for RCC in the community setting required more frequent dose reductions and had less time on therapy compared with patients in clinical trials, which ultimately impacted clinical outcomes.9
At the VA North Texas Health Care System (VANTHCS), patients with cancer have difficulty tolerating MKIs and often require dose alterations and/or discontinuation because of drug intolerance rather than discontinuation due to progression. Frequent dose adjustments for toxicity management can place more strain on patients and health care resources because of additional appointments, clinician time, and emergency department visits. Escalating drug costs can also cause concern when prescription doses are unused or changed frequently.
To capture and quantify prescribing practices and dose adjustments, this study evaluated the tolerability of MKIs at VANTHCS. This analysis may also guide clinicians in the selection of starting doses as well as dose titration expectations to optimize MKI therapy.
METHODS
This single-center, retrospective chart review analyzed patients receiving oral oncology MKIs for various malignancies at VANTHCS between January 1, 2014, and October 31, 2024. Participants included adults aged ≥ 18 years with a prescription for axitinib, cabozantinib, lenvatinib, pazopanib, regorafenib, sorafenib, or sunitinib initiated by the hematology/oncology service at VANTHCS. Patients were included if they had follow-up documentation with the hematology/oncology service and/or other VANTHCS clinicians outlining their course of therapy after MKI initiation. Patients were excluded if they did not have sufficient follow-up documentation (eg, transferred care to a non-VA health care practitioner [HCP], moved to another VA health care system), were enrolled in clinical trials, or were prescribed an MKI from a Care in the Community (CITC) prescriber. Electronic health record review and data collection were performed using the VA Computerized Patient Record System and Research Electronic Data Capture. Data were collected from the time of initiation to cessation of therapy and included information regarding therapy changes, progressive disease, and date of death, when available. Data collected included age, sex, race, comorbidities, date of death, type of malignancy and subtypes, cancer stage, MKI used (ie, drug, dose, frequency, schedule, and indication), dates of medication changes (ie, start, adjustment, hold, discontinuation), concurrent antineoplastic treatments, and AEs documented at times of dose change or interruption.
The primary outcome was MKI tolerance determined using relative dose intensity (RDI) and mean and median time on therapy. Two methods are used to calculate RDI that vary in how they approach time on therapy as outlined in Hawn et al.11 This study used method 2, which accounts for holds in therapy by comparing the actual duration of treatment with the duration expected according to treatment protocol. Method 1 compares the prescribed dose with the administered dose and does not adjust for holds.11 Using method 2, the RDI in this study was calculated by dividing the total actual dose given by the total indicated dose for the malignancy being treated, which accounts for duration of treatment.

The total actual dose was the strength, frequency, and days on therapy for each time frame of treatment multiplied together. This method accounted for all dose adjustments and time periods of treatment holds, including patient self-adjustments, prescriber-directed adjustments, and nonadherence determined by HCP documentation and/or prescription data. Similarly, the indicated total dose was calculated by multiplying the indicated strength, frequency, and all days that treatment should have occurred (time from start to finish). Indicated doses were derived from the prescribing information for each malignancy with the exception of sunitinib, for which the off-label dose of 37.5 mg daily was considered a full dose.12,13 The total indicated dose for axitinib was calculated by considering the dose escalation schedule from the prescribing information.
Patients who required dose reductions due to renal/hepatic impairments or drug-drug interactions had their total indicated dose calculated using dose adjustments listed in the prescribing information. The mean RDI for each MKI agent was calculated by averaging the RDI for each prescription. The overall combined mean RDI included the means of all the MKIs reviewed to avoid skewing the results toward an MKI with more prescriptions. RDIs were also calculated for each cancer type for each agent. Additional descriptive secondary outcomes included rates of AEs and adjustments in doses.
RESULTS
Electronic data extraction identified 278 patients with 366 MKI prescriptions, of which 108 veterans with 158 MKI prescriptions were excluded. The top reason for exclusion was patients managed through CITC. Ultimately, 170 veterans with 208 MKI prescriptions managed by the VANTHCS hematology/oncology clinic were included (Table 1). Among patients receiving MKIs, the mean age was 72.7 years, 98% were male, and 99% had metastatic disease.

The overall combined mean MKI RDI was 67.5% using method 2 and ranged from 85.5% for sunitinib to 49.0% for sorafenib (Figure 1). Additional information regarding mean and median RDIs using method 2 is shown in Figure 1 and further subdivided by cancer type in Table 2. Median RDIs overall were similar to mean RDIs for most agents. Figure 2 indicates the mean and median time on therapy, reflecting time on therapy excluding days therapy was held. The overall combined mean and median days on therapy for all MKIs were 155 days and 95 days, respectively. Mean time on therapy depended on the agent used and ranged from 35 days (regorafenib) to 237 days (cabozantinib).



Of 208 MKI prescriptions, 127 (61.1%) were initiated at a reduced dose due to baseline concerns for tolerance such as performance status, frailty, and prior intolerance of other treatments. Eighty-one prescriptions (38.9%) were initiated at their indicated doses. Ninety prescriptions (43.3%) required dose reductions during treatment. Some MKI prescriptions had multiple dose increases and decreases, which is why RDI more accurately reflects dose adjustments. A total of 376 AEs that contributed to a dose adjustment, hold, or discontinuation occurred across all MKI prescriptions. The most common AEs were 82 failure-to-thrive events (21.8%) (fatigue, malaise, loss of appetite, reduced mobility, global decline), 79 gastrointestinal events (21.0%) (nausea, vomiting, diarrhea, abdominal pain), 62 dermatologic events (16.5%) (rash, hand-foot skin reactions, allergic response), 61 hepatic dysfunction events (16.2%) (liver enzyme elevations, hyperbilirubinemia), 40 cardiovascular events (10.6%) (hypertension, heart failure exacerbations, edema), and 33 renal dysfunction events (8.8%) (acute kidney injury, proteinuria) (Appendix 1).

DISCUSSION
The mean RDI of MKI prescriptions used in the veteran population at VANTHCS was about two-thirds of the indicated dose. These results indicate that most veterans required dose reductions and/or holds due to concerns over initial tolerance/performance status, worsening clinical condition, and/or intolerable AEs attributed to treatment. A retrospective study conducted by Denduluri et al suggested that an RDI of < 85% is a clinically meaningful reduction for traditional chemotherapy based on previous literature.14 However, it is less clear what RDI should be expected specifically for MKIs in real-world populations. The MKI phase 3 approval trials in RCC for axitinib, lenvatinib, and sunitinib found median RDIs of 89.4%, 69.6% to 70.4%, and 83.9%, respectively. Each trial cited dose reductions most commonly as the result of treatment-related AEs.15,16
Studies on the impact of RDIs on survival outcomes found that higher RDIs may improve overall and progression-free survival. Retrospective studies inspecting lenvatinib in hepatocellular carcinoma (HCC) indicated that an RDI > 70% in the initial 4 weeks resulted in favorable survival outcomes.17 Similarly, a retrospective study investigating sunitinib in RCC found that an RDI > 60% conferred favorable survival outcomes.18 Alghamdi et al noted that patients taking sorafenib for HCC who had RDI > 50% had a favorable trend in survival characteristics. Interestingly, the study found an RDI of 50% to 75% appeared to have better survival than an RDI > 75%.19 The authors of these studies hypothesized that additional dose reductions allowed for longer total time on therapy due to improved tolerability.17-19
This analysis found that the RDIs for most MKI agents at VANTHCS were < 85% and lower than the RDIs found in other review articles and phase 3 trials, with the exceptions of pazopanib in thyroid cancer and sunitinib in gastrointestinal stromal tumor (GIST), thyroid cancer, and neuroendocrine cancer. The reasons for the lower RDIs in this study are likely multifactorial, reflecting patient population characteristics, off-label dosing practices, and HCP experiences with these agents. Many veterans have chronic comorbidities that could contribute to reduced performance status and ability to tolerate these therapies. Despite attempts to preemptively reduce doses for patients and account for potential impaired tolerance, there were patients who required further dose reductions in our study.
Failure to thrive was the most common AE leading to dose adjustment or discontinuation, which illustrates the extensive effects these agents have on patient functioning in a real-world population. Notably though, the RDI for sunitinib was higher in this population because about half of patients were dosed using the off-label recommendation, whereas the prescribing information recommends a more intensive 6-week dosing cycle for certain cancer types.12,13,20 Sorafenib was also often dose-adjusted based on a pharmacokinetic study of sorafenib in renal/hepatic dysfunction, and the RDI likely reflects the off-label prescribing pattern.21
Patients with thyroid cancer were found to have higher RDIs compared with those receiving the same agents for other cancer types. Improved tolerability of MKIs in thyroid cancer may be due to a generally more tolerable disease course. Thyroid cancer is the most common cancer in individuals aged < 40 years, a population that is often more robust with fewer comorbidities. Moreover, the 5-year relative survival rate for thyroid cancer remains > 98%.22 This rate is in contrast to those for other cancer types such as HCC, with a 5-year relative survival rate of only 15%.23
It is challenging to compare the mean and median times on therapy found in this study with those in current literature, as this review included multiple different cancer types for each agent. However, the numbers are generally lower than durations of therapy found across the different disease states and further emphasize the difficulty in tolerating MKIs in the VANTHCS population. Regorafenib had a short duration of time on therapy, which highlights the importance of trials like ReDOS and initiatives such as OCE Project Optimus in helping improve tolerance.7,8,24
Comparing our results with other studies proved challenging because the RDI calculation methods were not specified. Calculating RDIs in this study using method 1, which does not account for holds, resulted in higher RDIs (Appendix 2). Using method 1, all MKIs had RDIs < 85%, except for pazopanib in thyroid cancer (100%) and RCC (87.9%), and sunitinib in GIST (93.6%), thyroid cancer (100%), and neuroendocrine cancer (93.7%). Notably, using method 1 increased the RDI for pazopanib in neuroendocrine cancer from 5.4% to 50.0%. The low RDI was attributed to a single veteran with a long hold duration, which demonstrates the discrepancy that can occur between the 2 methods.

Limitations
The retrospective design, lack of survival outcomes, and difficulty comparing results with other literature were limitations of this study. Because survival outcomes were not evaluated, future research should seek to investigate how RDIs and dose adjustments made among MKIs can affect survival outcomes in real-world populations. This veteran population with cancer often had multiple chronic comorbidities, which may have contributed to difficulty tolerating MKIs and could have impacted results. Disease-related factors may have influenced the poor tolerance of the MKIs and were not specifically accounted for. Adjustment for comorbidities was not possible because of discrepancies and/or incomplete diagnosis codes and Eastern Cooperative Oncology Group performance status scores documented in patient charts. Therefore, we decided not to report these findings due to potential inaccuracies.
CONCLUSIONS
Results of this study demonstrate that oncology MKI agents used at VANTHCS were difficult for patients to tolerate, leading to suboptimal dosing compared with indicated doses established in clinical trials and prescribing information. Clinicians may use these data to help guide clinical decision-making whenever initiating and managing MKI agents in this population. These findings reinforce that MKI agents are often difficult to tolerate in real-world practice, and indicated doses are often not achieved. Further studies should aim to investigate the effect that various RDIs have on overall survival. Further investigation into different dosing schemes for MKIs to improve tolerability and longer-term use may also prove beneficial.
This analysis may help guide clinicians to carefully approach dosing MKI agents in the veteran population. Given the RDI and AEs, more clinicians may consider starting at lower than indicated doses with the goal to titrate up as tolerated. Additionally, the results highlight the importance of considering palliative care consults and ensuring appropriate supportive care agents are preemptively engaged and adjusted as needed. Approaching dosing and titrations cautiously may help reduce the burden of management on the health care system.
- Frequently asked questions. VA National Oncology Program. 2025. Accessed December 15, 2025. https://www.cancer.va.gov/CANCER/faqs.html
- Torez L. Reigniting the cancer moonshot to beat cancer. VA News. April 20, 2023. Accessed April 6, 2026. https://news.va.gov/118378/reigniting-the-cancer-moonshot-to-beat-cancer
- Shah NN, Casella E, Capozzi D, et al. Improving the safety of oral chemotherapy at an academic medical center. J Oncol Pract. 2016;12:e71-e76. doi:10.1200/JOP.2015.007260
- Hussaarts KGAM, Veerman GDM, Jansman FGA, et al. Clinically relevant drug interactions with multikinase inhibitors: a review. Ther Adv Med Oncol. 2019;11:1758835918818347. doi:10.1177/1758835918818347
- Shyam Sunder S, Sharma UC, Pokharel S. Adverse effects of tyrosine kinase inhibitors in cancer therapy: pathophysiology, mechanisms and clinical management. Signal Transduct Target Ther. 2023;8:262. doi:10.1038/s41392-023-01469-6
- Thomson RJ, Moshirfar M, Ronquillo Y. Tyrosine kinase inhibitors. In: StatPearls [Internet]. StatPearls Publishing; updated July 18, 2023. Accessed December 15, 2025. https://www.ncbi.nlm.nih.gov/books/NBK563322/
- Project Optimus. US Food and Drug Administration. Updated December 6, 2024. Accessed December 15, 2025. https://www.fda.gov/about-fda/oncology-center-excellence/project-optimus
- Optimizing the dosage of human prescription drugs and biological products for the treatment of oncologic diseases: Guidance for Industry. Docket number FDA-2022-D-2827. US Food and Drug Administration. August 2024. Accessed December 15, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/optimizing-dosage-human-prescription-drugs-and-biological-products-treatment-oncologic-diseases
- Schnadig ID, Hutson TE, Chung H, et al. Dosing patterns, toxicity, and outcomes in patients treated with first-line sunitinib for advanced renal cell carcinoma in community-based practices. Clin Genitourin Cancer. 2014;12:413-421. doi:10.1016/j.clgc.2014.06.015
- Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115-124. doi:10.1056/nejmoa065044
- Hawn C, Bansal D. Relative dose intensity in oncology trials: a discussion of two approaches. PharmaSUG. 2024. Accessed April 6, 2026. https://pharmasug.org/proceedings/2024/ST/PharmaSUG-2024-ST-297.pdf
- George S, Merriam P, Maki RG, et al. Multicenter phase II trial of sunitinib in the treatment of nongastrointestinal stromal tumor sarcomas. J Clin Oncol. 2009;27:3154-3160. doi:10.1200/jco.2008.20.9890
- George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer. 2009;45:1959-1968. doi:10.1016/j.ejca.2009.02.011
- Denduluri N, Patt DA, Wang Y, et al. Dose delays, dose reductions, and relative dose intensity in patients with cancer who received adjuvant or neoadjuvant chemotherapy in community oncology practices. J Natl Compr Canc Netw. 2015;13:1383-1393. doi:10.6004/jnccn.2015.0166
- Motzer RJ, Penkov K, Haanen J, et al. Avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380:1103-1115. doi:10.1056/nejmoa1816047
- Motzer R, Alekseev B, Rha SY, et al. Lenvatinib plus pembrolizumab or everolimus for advanced renal cell carcinoma. N Engl J Med. 2021;384:1289-1300. doi:10.1056/nejmoa2035716
- Kirino S, Tsuchiya K, Kurosaki M, et al. Relative dose intensity over the first four weeks of lenvatinib therapy is a factor of favorable response and overall survival in patients with unresectable hepatocellular carcinoma. PloS One. 2020;15:e0231828. doi:10.1371/journal.pone.0231828
- Ishihara H, Takagi T, Kondo T, et al. Decreased relative dose intensity during the early phase of treatment impacts the therapeutic efficacy of sunitinib in metastatic renal cell carcinoma. Jpn J Clin Oncol. 2018;48:667-672. doi:10.1093/jjco/hyy078
- Alghamdi MA, Amaro CP, Lee-Ying R, et al. Effect of sorafenib starting dose and dose intensity on survival in patients with hepatocellular carcinoma: results from a Canadian Multicenter Database. Cancer Med. 2020;9:4918-4928. doi:10.1002/cam4.3228
- Motzer RJ, Rini BI, Bukowski RM, et al. Sunitinib in patients with metastatic renal cell carcinoma. JAMA. 2006;295:2516-2524. doi:10.1001/jama.295.21.2516
- Miller AA, Murry DJ, Owzar K, et al. Phase I and pharmacokinetic study of sorafenib in patients with hepatic or renal dysfunction: CALGB 60301. J Clin Oncol. 2009;27:1800-1805. doi:10.1200/jco.2008.20.0931
- Boucai L, Zafereo M, Cabanillas ME. Thyroid cancer: a review. JAMA. 2024;331:425-435. doi:10.1001/jama.2023.26348
- Amin N, Anwar J, Sulaiman A, et al. Hepatocellular carcinoma: a comprehensive review. Diseases. 2025;13:207. doi:10.3390/diseases13070207
- Bekaii-Saab TS, Ou FS, Ahn DH, et al. Regorafenib dose-optimisation in patients with refractory metastatic colorectal cancer (ReDOS): a randomised, multicentre, open-label, phase 2 study. Lancet Oncol. 2019;20:1070-1082. doi:10.1016/s1470-2045(19)30272-4
- Frequently asked questions. VA National Oncology Program. 2025. Accessed December 15, 2025. https://www.cancer.va.gov/CANCER/faqs.html
- Torez L. Reigniting the cancer moonshot to beat cancer. VA News. April 20, 2023. Accessed April 6, 2026. https://news.va.gov/118378/reigniting-the-cancer-moonshot-to-beat-cancer
- Shah NN, Casella E, Capozzi D, et al. Improving the safety of oral chemotherapy at an academic medical center. J Oncol Pract. 2016;12:e71-e76. doi:10.1200/JOP.2015.007260
- Hussaarts KGAM, Veerman GDM, Jansman FGA, et al. Clinically relevant drug interactions with multikinase inhibitors: a review. Ther Adv Med Oncol. 2019;11:1758835918818347. doi:10.1177/1758835918818347
- Shyam Sunder S, Sharma UC, Pokharel S. Adverse effects of tyrosine kinase inhibitors in cancer therapy: pathophysiology, mechanisms and clinical management. Signal Transduct Target Ther. 2023;8:262. doi:10.1038/s41392-023-01469-6
- Thomson RJ, Moshirfar M, Ronquillo Y. Tyrosine kinase inhibitors. In: StatPearls [Internet]. StatPearls Publishing; updated July 18, 2023. Accessed December 15, 2025. https://www.ncbi.nlm.nih.gov/books/NBK563322/
- Project Optimus. US Food and Drug Administration. Updated December 6, 2024. Accessed December 15, 2025. https://www.fda.gov/about-fda/oncology-center-excellence/project-optimus
- Optimizing the dosage of human prescription drugs and biological products for the treatment of oncologic diseases: Guidance for Industry. Docket number FDA-2022-D-2827. US Food and Drug Administration. August 2024. Accessed December 15, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/optimizing-dosage-human-prescription-drugs-and-biological-products-treatment-oncologic-diseases
- Schnadig ID, Hutson TE, Chung H, et al. Dosing patterns, toxicity, and outcomes in patients treated with first-line sunitinib for advanced renal cell carcinoma in community-based practices. Clin Genitourin Cancer. 2014;12:413-421. doi:10.1016/j.clgc.2014.06.015
- Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115-124. doi:10.1056/nejmoa065044
- Hawn C, Bansal D. Relative dose intensity in oncology trials: a discussion of two approaches. PharmaSUG. 2024. Accessed April 6, 2026. https://pharmasug.org/proceedings/2024/ST/PharmaSUG-2024-ST-297.pdf
- George S, Merriam P, Maki RG, et al. Multicenter phase II trial of sunitinib in the treatment of nongastrointestinal stromal tumor sarcomas. J Clin Oncol. 2009;27:3154-3160. doi:10.1200/jco.2008.20.9890
- George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer. 2009;45:1959-1968. doi:10.1016/j.ejca.2009.02.011
- Denduluri N, Patt DA, Wang Y, et al. Dose delays, dose reductions, and relative dose intensity in patients with cancer who received adjuvant or neoadjuvant chemotherapy in community oncology practices. J Natl Compr Canc Netw. 2015;13:1383-1393. doi:10.6004/jnccn.2015.0166
- Motzer RJ, Penkov K, Haanen J, et al. Avelumab plus axitinib versus sunitinib for advanced renal-cell carcinoma. N Engl J Med. 2019;380:1103-1115. doi:10.1056/nejmoa1816047
- Motzer R, Alekseev B, Rha SY, et al. Lenvatinib plus pembrolizumab or everolimus for advanced renal cell carcinoma. N Engl J Med. 2021;384:1289-1300. doi:10.1056/nejmoa2035716
- Kirino S, Tsuchiya K, Kurosaki M, et al. Relative dose intensity over the first four weeks of lenvatinib therapy is a factor of favorable response and overall survival in patients with unresectable hepatocellular carcinoma. PloS One. 2020;15:e0231828. doi:10.1371/journal.pone.0231828
- Ishihara H, Takagi T, Kondo T, et al. Decreased relative dose intensity during the early phase of treatment impacts the therapeutic efficacy of sunitinib in metastatic renal cell carcinoma. Jpn J Clin Oncol. 2018;48:667-672. doi:10.1093/jjco/hyy078
- Alghamdi MA, Amaro CP, Lee-Ying R, et al. Effect of sorafenib starting dose and dose intensity on survival in patients with hepatocellular carcinoma: results from a Canadian Multicenter Database. Cancer Med. 2020;9:4918-4928. doi:10.1002/cam4.3228
- Motzer RJ, Rini BI, Bukowski RM, et al. Sunitinib in patients with metastatic renal cell carcinoma. JAMA. 2006;295:2516-2524. doi:10.1001/jama.295.21.2516
- Miller AA, Murry DJ, Owzar K, et al. Phase I and pharmacokinetic study of sorafenib in patients with hepatic or renal dysfunction: CALGB 60301. J Clin Oncol. 2009;27:1800-1805. doi:10.1200/jco.2008.20.0931
- Boucai L, Zafereo M, Cabanillas ME. Thyroid cancer: a review. JAMA. 2024;331:425-435. doi:10.1001/jama.2023.26348
- Amin N, Anwar J, Sulaiman A, et al. Hepatocellular carcinoma: a comprehensive review. Diseases. 2025;13:207. doi:10.3390/diseases13070207
- Bekaii-Saab TS, Ou FS, Ahn DH, et al. Regorafenib dose-optimisation in patients with refractory metastatic colorectal cancer (ReDOS): a randomised, multicentre, open-label, phase 2 study. Lancet Oncol. 2019;20:1070-1082. doi:10.1016/s1470-2045(19)30272-4
Investigating Real-World Tolerance and Dose Reductions of Oncology Multikinase Inhibitors in a VA Population
Investigating Real-World Tolerance and Dose Reductions of Oncology Multikinase Inhibitors in a VA Population
No Survival Gain With Adjuvant Therapy in Stage III Melanoma
Offering adjuvant therapy to patients with stage III melanoma offers no melanoma-specific or overall survival benefit, reveals extended follow-up from the first population-based national study to estimate the impact of the treatment.
Hildur Helgadottir, MD, PhD, presented the new findings at the 22nd European Association of Dermato-Oncology (EADO) Congress 2026 on April 24 and described the lead-up to the latest update on the study.
To investigate the impact of adjuvant treatment in patients with stage III melanoma, researchers initially conducted a study in which they used the Swedish Melanoma Registry (SweMR) to identify a precohort of those treated before the introduction of adjuvant therapy in 2018 and a postcohort of those treated subsequently, following both groups out to 2023, she explained.
The analysis revealed no significant difference in melanoma-specific survival between the two groups, at a hazard ratio of 0.92, nor in overall survival, at a hazard ratio of 0.93 (P = .60 for both). However, median follow-up differed between the groups, at 69 months vs 39 months for the precohort vs the postcohort.
Helgadottir, who is a senior research specialist at the Karolinska Comprehensive Cancer Center in Stockholm, Sweden, said that when the earlier results were presented at the European Society for Medical Oncology 2024, there was some criticism that the follow-up was not long enough and that there was no information on the actual adjuvant treatment received in the postcohort patients.
The researchers therefore extended their study out to 2024 to increase the median follow-up to 60 months vs 92 months in the postcohort group vs the precohort group.
They also focused patient selection on patients aged less than 75 years because exposure to adjuvant therapy in older patients was low and restricted the analysis to sentinel lymph node-positive stage IIIB-D cutaneous melanoma diagnosed between 2016 and 2020. This was because adjuvant exposure in stage IIIA disease was low, and patients with clinically detected stage III melanoma started to receive neoadjuvant therapy from 2022 onward.
The current analysis, which was recently published in the European Journal of Cancer, involved 287 patients in the precohort and 349 in the postcohort, who had a median age of 60.0 years and 61.0 years, respectively, and of whom 62.0% and 60.5%, respectively, were male. The groups were well balanced in terms of baseline disease characteristics.
Helgadottir explained that 73% of patients in the postcohort received some form of adjuvant treatment, with the majority treated with PD-1 inhibitors, and a smaller proportion given B-Raf serine-threonine kinase inhibitors. The main reasons for not giving adjuvant therapy were favorable tumor characteristics and the presence of comorbidities.
Five-year melanoma-specific survival rates in the precohorts and postcohorts were 71.4% vs 73.2%, at a hazard ratio adjusted for age, sex, and American Joint Committee on Cancer stage of 1.01 (P = .931). Five-year overall survival rates were 67.3% vs 70.1%, at an adjusted hazard ratio of 0.96 (P = .791).
Helgadottir showed that there were also no significant survival differences in any of the prespecified subgroups for neither melanoma-specific nor overall survival.
There were, again, no significant differences in survival outcomes between the two patient groups, she reported.
The latest results are similar to those from another study conducted in Netherlands and a Danish analysis, Helgadottir said.
Taken together, and “considering the side effects and the costs, it is possible that we will go back to closely following up our patients and treating only at relapse,” she said, “and optimally, of course, that will be already in the neoadjuvant setting.”
“And of course we will need biomarkers because there could be some patients that really need adjuvant treatment, but we need to identify these patients,” continued Helgadottir. Overall survival results from KEYNOTE-054, which compares pembrolizumab with placebo after resection of high-risk stage III melanoma, are awaited, she continued.
Helgadottir explained that adjuvant treatment for stage III melanoma was approved in Sweden in 2018, with treatments freely available to all Swedish residents.
The SweMR is a population-based national register that has near-complete and detailed data on primary cutaneous melanomas, including nodal status and satellite and in-transit disease, and is linked to the national Cause of Death Registry. Helgadottir noted, however, that the SweMR does not contain any information on relapses or the nature of the oncologic treatment received by patients with melanoma.
Following her presentation, she was challenged by an audience member as to whether, on the basis of her findings, she would go back to following up with patients and treating at relapses.
“Maybe we should do that and believe in our own data, and we do. But still, the gold standard must always be the randomized clinical trial,” Helgadottir responded. “So I think, although that we believe in this data, we also want to see the results of the randomized studies.”
The audience member commented that she can see in the data from her own institution that they treat fewer and fewer patients with melanoma with adjuvant therapy by discussing it more thoroughly and being stricter on who should receive it.
Helgadottir agreed, adding that “based on this experience, we did not introduce it to stage II patients because it’s always harder to go back” once a group of patients has started to receive a treatment.
The research was supported by Regional Cancer Centres in Sweden and with grants from the Swedish Cancer Society, Region Stockholm, and the Cancer Research Funds of Radiumhemmet. Helgadottir declared having relationships with Bristol Myers Squibb, Merck Sharp & Dohme, Pierre Fabre, and Novartis.
The trial was supported by SkinVision. The researchers declared having no relevant financial relationships.
This article was previously published by Medscape.
Offering adjuvant therapy to patients with stage III melanoma offers no melanoma-specific or overall survival benefit, reveals extended follow-up from the first population-based national study to estimate the impact of the treatment.
Hildur Helgadottir, MD, PhD, presented the new findings at the 22nd European Association of Dermato-Oncology (EADO) Congress 2026 on April 24 and described the lead-up to the latest update on the study.
To investigate the impact of adjuvant treatment in patients with stage III melanoma, researchers initially conducted a study in which they used the Swedish Melanoma Registry (SweMR) to identify a precohort of those treated before the introduction of adjuvant therapy in 2018 and a postcohort of those treated subsequently, following both groups out to 2023, she explained.
The analysis revealed no significant difference in melanoma-specific survival between the two groups, at a hazard ratio of 0.92, nor in overall survival, at a hazard ratio of 0.93 (P = .60 for both). However, median follow-up differed between the groups, at 69 months vs 39 months for the precohort vs the postcohort.
Helgadottir, who is a senior research specialist at the Karolinska Comprehensive Cancer Center in Stockholm, Sweden, said that when the earlier results were presented at the European Society for Medical Oncology 2024, there was some criticism that the follow-up was not long enough and that there was no information on the actual adjuvant treatment received in the postcohort patients.
The researchers therefore extended their study out to 2024 to increase the median follow-up to 60 months vs 92 months in the postcohort group vs the precohort group.
They also focused patient selection on patients aged less than 75 years because exposure to adjuvant therapy in older patients was low and restricted the analysis to sentinel lymph node-positive stage IIIB-D cutaneous melanoma diagnosed between 2016 and 2020. This was because adjuvant exposure in stage IIIA disease was low, and patients with clinically detected stage III melanoma started to receive neoadjuvant therapy from 2022 onward.
The current analysis, which was recently published in the European Journal of Cancer, involved 287 patients in the precohort and 349 in the postcohort, who had a median age of 60.0 years and 61.0 years, respectively, and of whom 62.0% and 60.5%, respectively, were male. The groups were well balanced in terms of baseline disease characteristics.
Helgadottir explained that 73% of patients in the postcohort received some form of adjuvant treatment, with the majority treated with PD-1 inhibitors, and a smaller proportion given B-Raf serine-threonine kinase inhibitors. The main reasons for not giving adjuvant therapy were favorable tumor characteristics and the presence of comorbidities.
Five-year melanoma-specific survival rates in the precohorts and postcohorts were 71.4% vs 73.2%, at a hazard ratio adjusted for age, sex, and American Joint Committee on Cancer stage of 1.01 (P = .931). Five-year overall survival rates were 67.3% vs 70.1%, at an adjusted hazard ratio of 0.96 (P = .791).
Helgadottir showed that there were also no significant survival differences in any of the prespecified subgroups for neither melanoma-specific nor overall survival.
There were, again, no significant differences in survival outcomes between the two patient groups, she reported.
The latest results are similar to those from another study conducted in Netherlands and a Danish analysis, Helgadottir said.
Taken together, and “considering the side effects and the costs, it is possible that we will go back to closely following up our patients and treating only at relapse,” she said, “and optimally, of course, that will be already in the neoadjuvant setting.”
“And of course we will need biomarkers because there could be some patients that really need adjuvant treatment, but we need to identify these patients,” continued Helgadottir. Overall survival results from KEYNOTE-054, which compares pembrolizumab with placebo after resection of high-risk stage III melanoma, are awaited, she continued.
Helgadottir explained that adjuvant treatment for stage III melanoma was approved in Sweden in 2018, with treatments freely available to all Swedish residents.
The SweMR is a population-based national register that has near-complete and detailed data on primary cutaneous melanomas, including nodal status and satellite and in-transit disease, and is linked to the national Cause of Death Registry. Helgadottir noted, however, that the SweMR does not contain any information on relapses or the nature of the oncologic treatment received by patients with melanoma.
Following her presentation, she was challenged by an audience member as to whether, on the basis of her findings, she would go back to following up with patients and treating at relapses.
“Maybe we should do that and believe in our own data, and we do. But still, the gold standard must always be the randomized clinical trial,” Helgadottir responded. “So I think, although that we believe in this data, we also want to see the results of the randomized studies.”
The audience member commented that she can see in the data from her own institution that they treat fewer and fewer patients with melanoma with adjuvant therapy by discussing it more thoroughly and being stricter on who should receive it.
Helgadottir agreed, adding that “based on this experience, we did not introduce it to stage II patients because it’s always harder to go back” once a group of patients has started to receive a treatment.
The research was supported by Regional Cancer Centres in Sweden and with grants from the Swedish Cancer Society, Region Stockholm, and the Cancer Research Funds of Radiumhemmet. Helgadottir declared having relationships with Bristol Myers Squibb, Merck Sharp & Dohme, Pierre Fabre, and Novartis.
The trial was supported by SkinVision. The researchers declared having no relevant financial relationships.
This article was previously published by Medscape.
Offering adjuvant therapy to patients with stage III melanoma offers no melanoma-specific or overall survival benefit, reveals extended follow-up from the first population-based national study to estimate the impact of the treatment.
Hildur Helgadottir, MD, PhD, presented the new findings at the 22nd European Association of Dermato-Oncology (EADO) Congress 2026 on April 24 and described the lead-up to the latest update on the study.
To investigate the impact of adjuvant treatment in patients with stage III melanoma, researchers initially conducted a study in which they used the Swedish Melanoma Registry (SweMR) to identify a precohort of those treated before the introduction of adjuvant therapy in 2018 and a postcohort of those treated subsequently, following both groups out to 2023, she explained.
The analysis revealed no significant difference in melanoma-specific survival between the two groups, at a hazard ratio of 0.92, nor in overall survival, at a hazard ratio of 0.93 (P = .60 for both). However, median follow-up differed between the groups, at 69 months vs 39 months for the precohort vs the postcohort.
Helgadottir, who is a senior research specialist at the Karolinska Comprehensive Cancer Center in Stockholm, Sweden, said that when the earlier results were presented at the European Society for Medical Oncology 2024, there was some criticism that the follow-up was not long enough and that there was no information on the actual adjuvant treatment received in the postcohort patients.
The researchers therefore extended their study out to 2024 to increase the median follow-up to 60 months vs 92 months in the postcohort group vs the precohort group.
They also focused patient selection on patients aged less than 75 years because exposure to adjuvant therapy in older patients was low and restricted the analysis to sentinel lymph node-positive stage IIIB-D cutaneous melanoma diagnosed between 2016 and 2020. This was because adjuvant exposure in stage IIIA disease was low, and patients with clinically detected stage III melanoma started to receive neoadjuvant therapy from 2022 onward.
The current analysis, which was recently published in the European Journal of Cancer, involved 287 patients in the precohort and 349 in the postcohort, who had a median age of 60.0 years and 61.0 years, respectively, and of whom 62.0% and 60.5%, respectively, were male. The groups were well balanced in terms of baseline disease characteristics.
Helgadottir explained that 73% of patients in the postcohort received some form of adjuvant treatment, with the majority treated with PD-1 inhibitors, and a smaller proportion given B-Raf serine-threonine kinase inhibitors. The main reasons for not giving adjuvant therapy were favorable tumor characteristics and the presence of comorbidities.
Five-year melanoma-specific survival rates in the precohorts and postcohorts were 71.4% vs 73.2%, at a hazard ratio adjusted for age, sex, and American Joint Committee on Cancer stage of 1.01 (P = .931). Five-year overall survival rates were 67.3% vs 70.1%, at an adjusted hazard ratio of 0.96 (P = .791).
Helgadottir showed that there were also no significant survival differences in any of the prespecified subgroups for neither melanoma-specific nor overall survival.
There were, again, no significant differences in survival outcomes between the two patient groups, she reported.
The latest results are similar to those from another study conducted in Netherlands and a Danish analysis, Helgadottir said.
Taken together, and “considering the side effects and the costs, it is possible that we will go back to closely following up our patients and treating only at relapse,” she said, “and optimally, of course, that will be already in the neoadjuvant setting.”
“And of course we will need biomarkers because there could be some patients that really need adjuvant treatment, but we need to identify these patients,” continued Helgadottir. Overall survival results from KEYNOTE-054, which compares pembrolizumab with placebo after resection of high-risk stage III melanoma, are awaited, she continued.
Helgadottir explained that adjuvant treatment for stage III melanoma was approved in Sweden in 2018, with treatments freely available to all Swedish residents.
The SweMR is a population-based national register that has near-complete and detailed data on primary cutaneous melanomas, including nodal status and satellite and in-transit disease, and is linked to the national Cause of Death Registry. Helgadottir noted, however, that the SweMR does not contain any information on relapses or the nature of the oncologic treatment received by patients with melanoma.
Following her presentation, she was challenged by an audience member as to whether, on the basis of her findings, she would go back to following up with patients and treating at relapses.
“Maybe we should do that and believe in our own data, and we do. But still, the gold standard must always be the randomized clinical trial,” Helgadottir responded. “So I think, although that we believe in this data, we also want to see the results of the randomized studies.”
The audience member commented that she can see in the data from her own institution that they treat fewer and fewer patients with melanoma with adjuvant therapy by discussing it more thoroughly and being stricter on who should receive it.
Helgadottir agreed, adding that “based on this experience, we did not introduce it to stage II patients because it’s always harder to go back” once a group of patients has started to receive a treatment.
The research was supported by Regional Cancer Centres in Sweden and with grants from the Swedish Cancer Society, Region Stockholm, and the Cancer Research Funds of Radiumhemmet. Helgadottir declared having relationships with Bristol Myers Squibb, Merck Sharp & Dohme, Pierre Fabre, and Novartis.
The trial was supported by SkinVision. The researchers declared having no relevant financial relationships.
This article was previously published by Medscape.
GLP-1s Lower Risk of SUDs in VA Studies
Two studies published in March by researchers at the Veterans Affairs Saint Louis Healthcare System highlight the clinical significance of glucagon-like peptide 1 receptor agonists (GLP-1s) and their impact on reducing substance use disorder (SUD) risks. The studies also explore the impact of GLP-1 discontinuation or interruption on their effectiveness in protection against the cardiovascular events.
In one study, Al-Aly et al assigned 606,434 veterans with type 2 diabetes to 1 of 2 protocols, comparing GLP-1s with sodium-glucose cotransporter-2 (SGLT-2) inhibitors, and followed the patients for up to 3 years. Al-Aly et al found that GLP-1s were “consistently associated” with a lower risk of developing SUDs, including those involving alcohol, cannabis, cocaine, nicotine, and opioids. The findings suggested “potential preventive effects across a broad range of addictive substances.”
In participants with pre-existing SUDs, GLP-1s were also associated with reduced risks of SUD-related emergency department visits, hospital admissions, and mortality, in addition to drug overdoses and suicidal behaviors. A study published in 2025 from the same research group reported that GLP-1s could have a variety of health benefits, including reducing the risk of incident alcohol and cannabis disorders, neurocognitive disorders (such as Alzheimer's disease and dementia), coagulation disorders, cardiometabolic disorders, infectious illnesses and several respiratory conditions, but less was known about the potential for preventing development of opioid use disorder and other SUDs.
GLP-1s target the brain’s reward pathways and have recently made attention-grabbing headlines regarding celebrity weight loss, with social media boosting public interest. One study, for example, found 100 videos on TikTok with the #Ozempic viewed nearly 70 million times.
Al-Aly et al used SGLT-2 inhibitors as active comparators because “they have no established direct actions on mesolimbic reward circuits in the brain, whereas GLP-1 receptors are present in areas of the brain involved in impulse control and reward signaling.”
The second study found that quitting or pausing GLP-1 treatment for 6 months could have a rebound effect and possibly reverse any progress. Discontinuing GLP-1 treatment is common, with rates ranging from 36% to 81% in the first year. Stopping or interrupting the treatment is often followed by weight regain and a rebound in inflammation, both major drivers in cardiovascular disease risk.
The study followed 132,551 VA patients using GLP-1s and 201,136 using sulfonylureas from 2017 through 2023. About two-thirds of participants took semaglutide, prescribed as Ozempic to treat diabetes and Wegovy to reduce obesity. A total of 26% of the participants stopped GLP-1 treatment during the follow-up period, with 64% occurring during the first year. Most (67%) treatment interruptions also came in the first year.
Compared with incident use of sulfonylureas, incident use of GLP-1s was associated with a reduced risk of heart attack, stroke, or death. Patients who took the GLP-1s without interruption > 3 years experienced an 18% lower risk for heart attack or stroke.
Cardiovascular benefits accumulated with continuous use over 3 years, but even brief periods of discontinuations or interruptions could progressively erode and ultimately reverse this protection, the researchers found. Discontinuing treatment for half a year was associated with an increased risk of major adverse cardiovascular events (incidence risk ratio [IRR], 1.04), while longer gaps were progressively associated with a higher risk of disease (IRR, 1.12 for 1 year; IRR, 1.16 for 2 years of interrupted use, respectively).
Dr. Ziyad Al-Aly, a study author and Chief of the Research and Education Service at the Veterans Affairs Saint Louis Healthcare System, called it “metabolic whiplash.” In an interview, he said it was important to caution patients that these medications “need to be taken for the long haul. This is not something (patients) can take for a month or 2 or 3 and get off of it. It's not going to work like that.”
Two studies published in March by researchers at the Veterans Affairs Saint Louis Healthcare System highlight the clinical significance of glucagon-like peptide 1 receptor agonists (GLP-1s) and their impact on reducing substance use disorder (SUD) risks. The studies also explore the impact of GLP-1 discontinuation or interruption on their effectiveness in protection against the cardiovascular events.
In one study, Al-Aly et al assigned 606,434 veterans with type 2 diabetes to 1 of 2 protocols, comparing GLP-1s with sodium-glucose cotransporter-2 (SGLT-2) inhibitors, and followed the patients for up to 3 years. Al-Aly et al found that GLP-1s were “consistently associated” with a lower risk of developing SUDs, including those involving alcohol, cannabis, cocaine, nicotine, and opioids. The findings suggested “potential preventive effects across a broad range of addictive substances.”
In participants with pre-existing SUDs, GLP-1s were also associated with reduced risks of SUD-related emergency department visits, hospital admissions, and mortality, in addition to drug overdoses and suicidal behaviors. A study published in 2025 from the same research group reported that GLP-1s could have a variety of health benefits, including reducing the risk of incident alcohol and cannabis disorders, neurocognitive disorders (such as Alzheimer's disease and dementia), coagulation disorders, cardiometabolic disorders, infectious illnesses and several respiratory conditions, but less was known about the potential for preventing development of opioid use disorder and other SUDs.
GLP-1s target the brain’s reward pathways and have recently made attention-grabbing headlines regarding celebrity weight loss, with social media boosting public interest. One study, for example, found 100 videos on TikTok with the #Ozempic viewed nearly 70 million times.
Al-Aly et al used SGLT-2 inhibitors as active comparators because “they have no established direct actions on mesolimbic reward circuits in the brain, whereas GLP-1 receptors are present in areas of the brain involved in impulse control and reward signaling.”
The second study found that quitting or pausing GLP-1 treatment for 6 months could have a rebound effect and possibly reverse any progress. Discontinuing GLP-1 treatment is common, with rates ranging from 36% to 81% in the first year. Stopping or interrupting the treatment is often followed by weight regain and a rebound in inflammation, both major drivers in cardiovascular disease risk.
The study followed 132,551 VA patients using GLP-1s and 201,136 using sulfonylureas from 2017 through 2023. About two-thirds of participants took semaglutide, prescribed as Ozempic to treat diabetes and Wegovy to reduce obesity. A total of 26% of the participants stopped GLP-1 treatment during the follow-up period, with 64% occurring during the first year. Most (67%) treatment interruptions also came in the first year.
Compared with incident use of sulfonylureas, incident use of GLP-1s was associated with a reduced risk of heart attack, stroke, or death. Patients who took the GLP-1s without interruption > 3 years experienced an 18% lower risk for heart attack or stroke.
Cardiovascular benefits accumulated with continuous use over 3 years, but even brief periods of discontinuations or interruptions could progressively erode and ultimately reverse this protection, the researchers found. Discontinuing treatment for half a year was associated with an increased risk of major adverse cardiovascular events (incidence risk ratio [IRR], 1.04), while longer gaps were progressively associated with a higher risk of disease (IRR, 1.12 for 1 year; IRR, 1.16 for 2 years of interrupted use, respectively).
Dr. Ziyad Al-Aly, a study author and Chief of the Research and Education Service at the Veterans Affairs Saint Louis Healthcare System, called it “metabolic whiplash.” In an interview, he said it was important to caution patients that these medications “need to be taken for the long haul. This is not something (patients) can take for a month or 2 or 3 and get off of it. It's not going to work like that.”
Two studies published in March by researchers at the Veterans Affairs Saint Louis Healthcare System highlight the clinical significance of glucagon-like peptide 1 receptor agonists (GLP-1s) and their impact on reducing substance use disorder (SUD) risks. The studies also explore the impact of GLP-1 discontinuation or interruption on their effectiveness in protection against the cardiovascular events.
In one study, Al-Aly et al assigned 606,434 veterans with type 2 diabetes to 1 of 2 protocols, comparing GLP-1s with sodium-glucose cotransporter-2 (SGLT-2) inhibitors, and followed the patients for up to 3 years. Al-Aly et al found that GLP-1s were “consistently associated” with a lower risk of developing SUDs, including those involving alcohol, cannabis, cocaine, nicotine, and opioids. The findings suggested “potential preventive effects across a broad range of addictive substances.”
In participants with pre-existing SUDs, GLP-1s were also associated with reduced risks of SUD-related emergency department visits, hospital admissions, and mortality, in addition to drug overdoses and suicidal behaviors. A study published in 2025 from the same research group reported that GLP-1s could have a variety of health benefits, including reducing the risk of incident alcohol and cannabis disorders, neurocognitive disorders (such as Alzheimer's disease and dementia), coagulation disorders, cardiometabolic disorders, infectious illnesses and several respiratory conditions, but less was known about the potential for preventing development of opioid use disorder and other SUDs.
GLP-1s target the brain’s reward pathways and have recently made attention-grabbing headlines regarding celebrity weight loss, with social media boosting public interest. One study, for example, found 100 videos on TikTok with the #Ozempic viewed nearly 70 million times.
Al-Aly et al used SGLT-2 inhibitors as active comparators because “they have no established direct actions on mesolimbic reward circuits in the brain, whereas GLP-1 receptors are present in areas of the brain involved in impulse control and reward signaling.”
The second study found that quitting or pausing GLP-1 treatment for 6 months could have a rebound effect and possibly reverse any progress. Discontinuing GLP-1 treatment is common, with rates ranging from 36% to 81% in the first year. Stopping or interrupting the treatment is often followed by weight regain and a rebound in inflammation, both major drivers in cardiovascular disease risk.
The study followed 132,551 VA patients using GLP-1s and 201,136 using sulfonylureas from 2017 through 2023. About two-thirds of participants took semaglutide, prescribed as Ozempic to treat diabetes and Wegovy to reduce obesity. A total of 26% of the participants stopped GLP-1 treatment during the follow-up period, with 64% occurring during the first year. Most (67%) treatment interruptions also came in the first year.
Compared with incident use of sulfonylureas, incident use of GLP-1s was associated with a reduced risk of heart attack, stroke, or death. Patients who took the GLP-1s without interruption > 3 years experienced an 18% lower risk for heart attack or stroke.
Cardiovascular benefits accumulated with continuous use over 3 years, but even brief periods of discontinuations or interruptions could progressively erode and ultimately reverse this protection, the researchers found. Discontinuing treatment for half a year was associated with an increased risk of major adverse cardiovascular events (incidence risk ratio [IRR], 1.04), while longer gaps were progressively associated with a higher risk of disease (IRR, 1.12 for 1 year; IRR, 1.16 for 2 years of interrupted use, respectively).
Dr. Ziyad Al-Aly, a study author and Chief of the Research and Education Service at the Veterans Affairs Saint Louis Healthcare System, called it “metabolic whiplash.” In an interview, he said it was important to caution patients that these medications “need to be taken for the long haul. This is not something (patients) can take for a month or 2 or 3 and get off of it. It's not going to work like that.”
Evaluation of Pharmacist-Driven Inhaled Corticosteroid De-escalation in Veterans
Evaluation of Pharmacist-Driven Inhaled Corticosteroid De-escalation in Veterans
Systemic glucocorticoids play an important role in the treatment of chronic obstructive pulmonary disease (COPD) exacerbations. They are recommended to shorten recovery time and increase forced expiratory volume in 1 second (FEV1) during exacerbations.1 However, the role of the chronic use of inhaled corticosteroids (ICSs) in the treatment of COPD is less clear.
When added to inhaled β-2 agonists and muscarinic antagonists, ICSs can decrease the risk of exacerbations.1 However, not all patients with COPD benefit from ICS therapy. The degree of benefit an ICS can provide has been shown to correlate with eosinophil count—a marker of inflammation. The expected benefit of using an ICS increases as the eosinophil count increases.1 Maximum benefit can be observed with eosinophil counts ≥ 300 cells/µL, and minimal benefit is observed with eosinophil counts < 100 cells/µL. Adverse effects (AEs) of ICSs include a hoarse voice, oral candidiasis, and an increased risk of pneumonia.1 Given the risk of AEs, it is important to limit ICS use in patients who are unlikely to reap any benefits.
The Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines suggest the use of ICSs in patients who experience exacerbations while using long-acting β agonist (LABA) plus long-acting muscarinic antagonist (LAMA) therapy and have an eosinophil count ≥ 100 cells/µL. Switching from LABA or LAMA monotherapy to triple therapy with LAMA/LABA/ICS may be considered if patients have continued exacerbations and an eosinophil count ≥ 300 cells/µL. De-escalation of ICS therapy should be considered if patients do not meet these criteria or if patients experience ICS AEs, such as pneumonia. The patients most likely to have increased exacerbations or decreased FEV1 with ICS withdrawal are those with eosinophil counts ≥ 300 cells/µL.1,2
Several studies have explored the effects of ICS de-escalation in real-world clinical settings. A systematic review of 11 studies indicated that de-escalation of ICS in COPD does not result in increased exacerbations.3 A prospective study by Rossi et al found that in a 6-month period, 141 of 482 patients on ICS therapy (29%) had an exacerbation. In the opposing arm of the study, 88 of 334 patients (26%) with deprescribed ICS experienced an exacerbation. The difference between these 2 groups was not statistically significant.4 The researchers concluded that in real-world practice, ICS withdrawal can be safe in patients at low risk of exacerbation.
About 25% of veterans (1.25 million) have been diagnosed with COPD.5 To address this, the US Department of Veterans Affairs (VA) and US Department of Defense published updated COPD guidelines in 2021 that specify criteria for de-escalation of ICS.6 Guidelines, however, may not be reflected in common clinical practice for several years following publication. The VA Academic Detailing Service (ADS) provides tools to help clinicians identify patients who may benefit from changes in treatment plans. A recent ADS focus was the implementation of a COPD dashboard, which identifies patients with COPD who are candidates for ICS de-escalation based on comorbid diagnoses, exacerbation history, and eosinophil count. VA pharmacists have an expanded role in the management of primary care disease states and are therefore well-positioned to increase adherence to guideline-directed therapy. The objective of this quality improvement project was to determine the impact of pharmacist-driven de-escalation on ICS usage in veterans with COPD.
Methods
This project was conducted in an outpatient clinic at the Robley Rex VA Medical Center beginning September 21, 2023, with a progress note in the Computerized Patient Record System (CPRS). Eligible patients were selected using the COPD Dashboard provided by ADS. The COPD Dashboard defined patients with COPD as those with ≥ 2 outpatient COPD diagnoses in the past 2 years, 1 inpatient discharge COPD diagnosis in the past year, or COPD listed as an active problem. COPD diagnoses were identified using International Statistical Classification of Disease, Tenth Revision (ICD-10) codes
Candidates identified for ICS de-escalation by the dashboard were excluded if they had a history of COPD exacerbation in the previous 2 years. The dashboard identified COPD exacerbations via ICD-10 codes for COPD or acute respiratory failure for inpatient discharges, emergency department (ED) visits, urgent care visits, and community care consults with 1 of the following terms: emergency, inpatient, hospital, urgent, ED (self). The COPD dashboard excluded patients with a diagnosis of asthma.
After patients were selected, they were screened for additional exclusion criteria. Patients were excluded if a pulmonary care practitioner managed their COPD; if identified via an active pulmonary consult in CPRS; if a non-VA clinician prescribed their ICS; or if they were being treated with roflumilast, theophylline, or chronic azithromycin. Individuals taking these 3 drugs were excluded due to potential severe and/or refractory COPD. Patients also were excluded if they: (1) had prior ICS de-escalation failure (defined as a COPD exacerbation following ICS de-escalation that resulted in ICS resumption); (2) had a COPD exacerbation requiring systemic corticosteroids or antibiotics in the previous year; (3) had active lung cancer; (4) did not have any eosinophil levels in CPRS within the previous 2 years; or (5) had any eosinophil levels ≥ 300 cells/µL in the previous year.
Each patient who met the inclusion criteria and was not excluded received a focused medication review by a pharmacist who created a templated progress note, with patient-specific recommendations, that was entered in the CPRS (eAppendix). The recommendations were also attached as an addendum to the patient’s last primary care visit note, and the primary care practitioner (PCP) was alerted via CPRS to consider ICS de-escalation and non-ICS alternatives. Tapering of ICS therapy was offered as an option to de-escalate if abrupt discontinuation was deemed inappropriate. PCPs were also prompted to consider referral to a primary care clinical pharmacy specialist for management and follow-up of ICS de-escalation.
The primary outcome was the number of patients with de-escalated ICS at 3 and 6 months following the recommendation. Secondary outcomes included the number of: patients who were no longer prescribed an ICS or who had a non-ICS alternative initiated at a pharmacist’s recommendation; patients who were referred to a primary care clinical pharmacy specialist for ICS de-escalation; COPD exacerbations requiring systemic steroids or antibiotics, or requiring an ED visit, inpatient admission, or urgent-care clinic visit; and cases of pneumonia or oral candidiasis. Primary and secondary outcomes were evaluated via chart review in CPRS. For secondary outcomes of pneumonia and COPD exacerbation, identification was made by documented diagnosis in CPRS. For continuous data such as age, the mean was calculated.
Results
Pharmacist ICS de-escalation recommendations were made between September 21, 2023, and November 19, 2023, for 106 patients. The mean age was 72 years and 99 (93%) patients were male (Table 1). Forty-one (39%) of the patients used tobacco at the time of the study. FEV1 was available for 69 patients with a mean of 63% (GOLD grade 2).1 Based on FEV1 values, 16 patients had mild COPD (GOLD grade 1), 37 patients had moderate COPD (GOLD grade 2), 14 patients had severe COPD (GOLD grade 3), and 2 patients had very severe COPD (GOLD grade 4).1 Thirty-four patients received LABA + LAMA + ICS, 65 received LABA + ICS, 2 received LAMA + ICS, and 5 received ICS monotherapy. The most common dose of ICS was a moderate dose (Table 2). Only 2 patients had an ICS AE in the previous year.
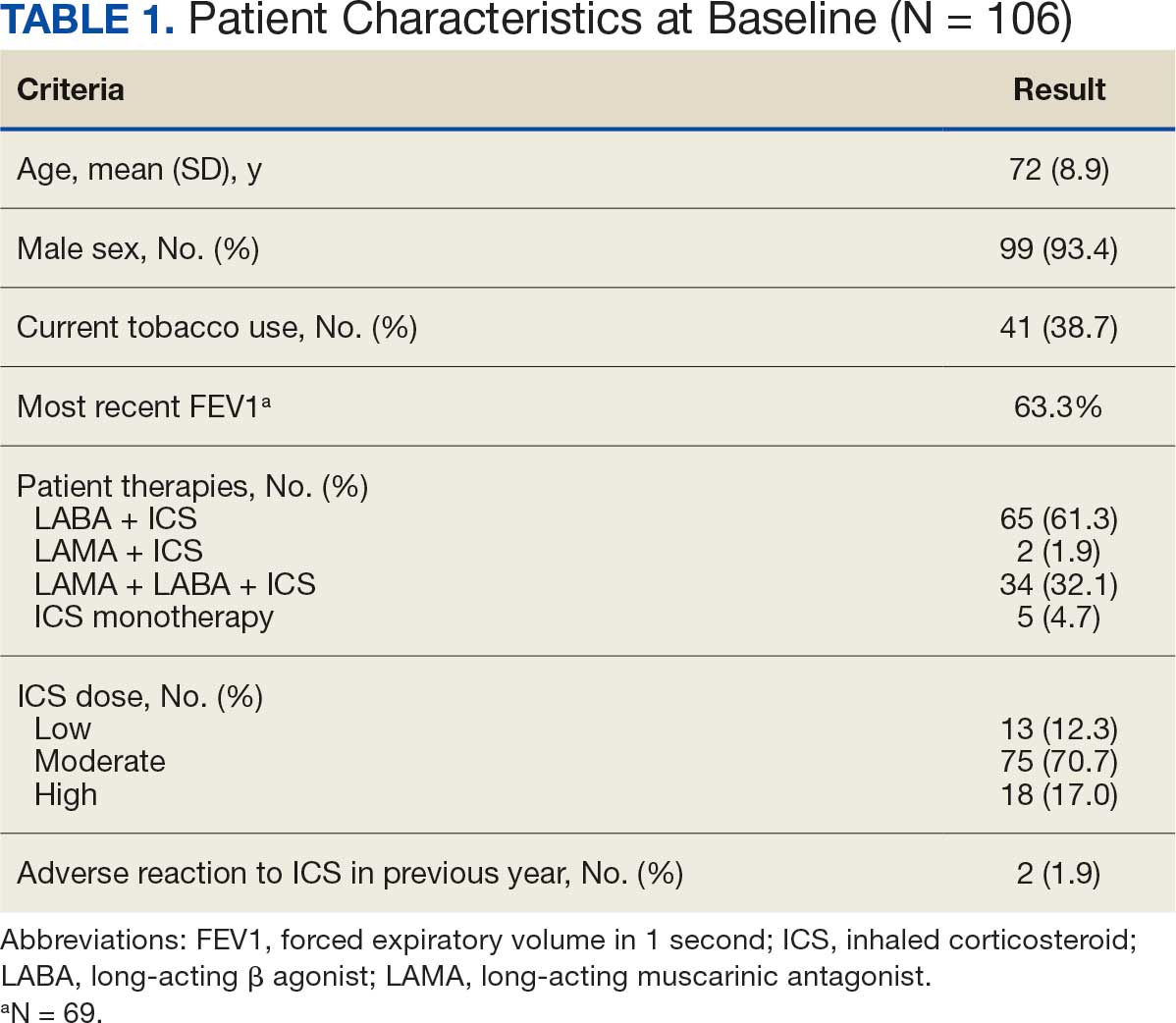
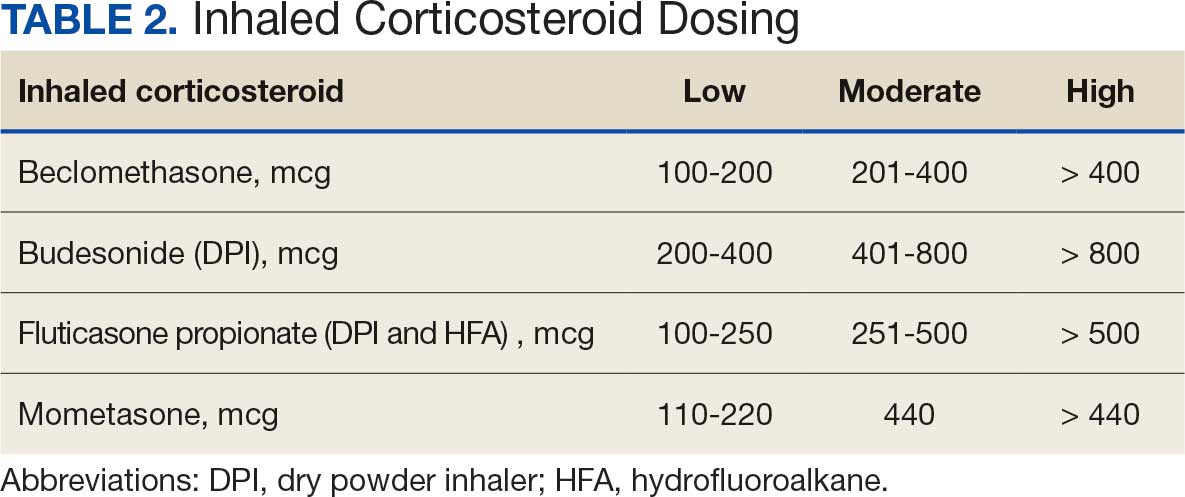
ICS de-escalation recommendations resulted in ICS de-escalation in 50 (47.2%) and 62 (58.5%) patients at 3 and 6 months, respectively. The 6-month ICS de-escalation rate by ICS dose at baseline was 72.2% (high dose), 60.0% (moderate), and 30.8% (low). De-escalation at 6 months by GOLD grade at baseline was 56.3% (9 of 16 patients, GOLD 1), 64.9% (24 of 37 patients, GOLD 2), 50% (7 of 14 patients, GOLD 3), and 50% (1 of 2 patients, GOLD 4). Six months after the ICS de-escalation recommendation appeared in the CPRS, the percentage of patients on LABA + ICS therapy dropped from 65 patients (61.3%) at baseline to 25 patients (23.6%).
Secondary outcomes were assessed at 3 and 6 months following the recommendation. Most patients with de-escalated ICS had their ICS discontinued and a non-ICS alternative initiated per pharmacist recommendations. At 6 months, 39 patients (36.8%) patients were referred to a patient aligned care team (PACT) pharmacist for de-escalation. Of the 39 patients referred to pharmacists, 69.2% (27 patients) were de-escalated; this compared to 52.2% (35 patients) who were not referred to pharmacists (Table 3).
ICS use increases the risk of pneumonia.1 At 6 months, 11 patients were diagnosed with pneumonia; 3 patients were diagnosed with pneumonia twice, resulting in a total of 14 cases. Ten cases occurred while patients were on ICS and 4 cases occurred following ICS de-escalation. One patient had a documented case of oral candidiasis that occurred while on ICS therapy; no patients with discontinued ICS were diagnosed with oral candidiasis. In addition, 10 patients had COPD exacerbations; however no patients had exacerbations both before and after de-escalation. Six patients were on ICS therapy when they experienced an exacerbation, and 4 patients had an exacerbation after ICS de-escalation.
Discussion
More than half of patients receiving the pharmacist intervention achieved the primary outcome of ICS de-escalation at 6 months. Furthermore, a larger percentage of patients referred to pharmacists for the management of ICS de-escalation successfully achieved de-escalation compared to those who were not referred. These outcomes reflect the important role pharmacists can play in identifying appropriate candidates for ICS de-escalation and assisting in the management of ICS de-escalation. Patients referred to pharmacists also received other services such as smoking cessation pharmacotherapy and counseling on inhaler technique and adherence. These interventions can support improved COPD clinical outcomes.
The purpose of de-escalating ICS therapy is to reduce the risk of AEs such as pneumonia and oral candidiasis.1 The secondary outcomes of this study support previous evidence that patients who have de-escalated ICS therapy may have reduced risk of AEs compared to those who remain on ICS therapy.3 Specifically, of the 14 cases of pneumonia that occurred during the study, 10 cases occurred while patients were on ICS and 4 cases occurred following ICS de-escalation.
ICS de-escalation may increase risk of increased COPD exacerbations.1 However, the secondary outcomes of this study do not indicate that those with de-escalated ICS had more COPD exacerbations compared to those who continued on ICS. Pharmacists’ recommendations were more effective for patients with less severe COPD based on baseline FEV1.
The previous GOLD Guidelines for COPD suggested LABA + ICS therapy as an option for patients with a high symptom and exacerbation burden (previously known as GOLD Group D). Guidelines no longer recommend LABA + ICS therapy due to the superiority of triple inhaled therapy for exacerbations and the superiority of LAMA + LABA therapy for dyspnea.7 A majority of identified patients in this project were on LABA + ICS therapy alone at baseline. The ICS de-escalation recommendation resulted in a 61.5% reduction in patients on LABA + ICS therapy at 6 months. By decreasing the number of patients on LABA + ICS without LAMA, recommendations increased the number of patients on guideline-directed therapy.
Limitations
This study lacked a control group, and the rate of ICS de-escalation in patients who did not receive a pharmacist recommendation was not assessed. Therefore, it could not be determined whether the pharmacist recommendation is more effective than no recommendation. Another limitation was our inability to access records from non-VA health care facilities. This may have resulted in missed COPD exacerbations, pneumonia, and oral candidiasis prior to or following the pharmacist recommendation.
In addition, the method used to notify PCPs of the pharmacist recommendation was a CPRS alert. Clinicians often receive multiple daily alerts and may not always pay close attention to them due to alert fatigue. Early in the study, some PCPs were unknowingly omitted from the alert of the pharmacist recommendation for 10 patients due to human error. For 8 of these 10 patients, the PCP was notified of the recommendations during the 3-month follow-up period. However, 2 patients had COPD exacerbations during the 3-month follow-up period. In these cases, the PCP was not alerted to de-escalate ICS. The data for these patients were collected at 3 and 6 months in the same manner as all other patients. Also, 7 of 35 patients who were referred to a pharmacist for ICS de-escalation did not have a scheduled appointment. These patients were considered to be lost to follow-up and this may have resulted in an underestimation of the ability of pharmacists to successfully de-escalate ICS in patients with COPD.
Other studies have evaluated the efficacy of a pharmacy-driven ICS de-escalation.8,9 Hegland et al reported ICS de-escalation for 22% of 141 eligible ambulatory patients with COPD on triple inhaled therapy following pharmacist appointments.8 A study by Hahn et al resulted in 63.8% of 58 patients with COPD being maintained off ICS following a pharmacist de-escalation initiative.9 However, these studies relied upon more time-consuming de-escalation interventions, including at least 1 phone, video, or in-person patient visit.8,9
This project used a single chart review and templated progress note to recommend ICS de-escalation and achieved similar or improved de-escalation rates compared to previous studies.8,9 Previous studies were conducted prior to the updated 2023 GOLD guidelines for COPD which no longer recommend LABA + ICS therapy. This project addressed ICS de-escalation in patients on LABA + ICS therapy in addition to those on triple inhaled therapy. Additionally, previous studies did not address rates of moderate to severe COPD exacerbation and adverse events to ICS following the pharmacist intervention.8,9
This study included COPD exacerbations and cases of pneumonia or oral candidiasis as secondary outcomes to assess the safety and efficacy of the ICS de-escalation. It appeared there were similar or lower rates of COPD exacerbations, pneumonia, and oral candidiasis in those with de-escalated ICS therapy in this study. However, these secondary outcomes are exploratory and would need to be confirmed by larger studies powered to address these outcomes.
CONCLUSIONS
Pharmacist-driven ICS de-escalation may be an effective method for reducing ICS usage in veterans as seen in this study. Additional controlled studies are required to evaluate the efficacy and safety of pharmacist-driven ICS de-escalation.
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2024 Report). Accessed October 14, 2025. https://goldcopd.org/2024-gold-report/
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2025 Report). Accessed November 14, 2025. https://goldcopd.org/2025-gold-report/
- Rogliani P, Ritondo BL, Gabriele M, et al. Optimizing de-escalation of inhaled corticosteroids in COPD: a systematic review of real-world findings. Expert Rev Clin Pharmacol. 2020;13(9):977-990. doi:10.1080/17512433.2020.1817739
- Rossi A, Guerriero M, Corrado A; OPTIMO/AIPO Study Group. Withdrawal of inhaled corticosteroids can be safe in COPD patients at low risk of exacerbation: a real-life study on the appropriateness of treatment in moderate COPD patients (OPTIMO). Respir Res. 2014;15(1):77. doi:10.1186/1465-9921-15-77
- Anderson E, Wiener RS, Resnick K, et al. Care coordination for veterans with COPD: a positive deviance study. Am J Manag Care. 2020;26(2):63-68. doi:10.37765/ajmc.2020.42394
- US Department of Veterans Affairs, US Department of Defense. VA/DoD Clinical Practice Guideline for the Management of Chronic Obstructive Pulmonary Disease. 2021. Accessed October 14, 2025. https://www.healthquality.va.gov/guidelines/CD/copd/
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2023 Report). Accessed October 14, 2025. https://goldcopd.org/wp-content/uploads/2023/03/GOLD-2023-ver-1.3-17Feb2023_WMV.pdf
- Hegland AJ, Bolduc J, Jones L, Kunisaki KM, Melzer AC. Pharmacist-driven deprescribing of inhaled corticosteroids in patients with stable chronic obstructive pulmonary disease. Ann Am Thorac Soc. 2021;18(4):730-733. doi:10.1513/AnnalsATS.202007-871RL
- Hahn NM, Nagy MW. Implementation of a targeted inhaled corticosteroid de-escalation process in patients with chronic obstructive pulmonary disease in the primary care setting. Innov Pharm. 2022;13(1):10.24926/iip.v13i1.4349. doi:10.24926/iip.v13i1.4349
Systemic glucocorticoids play an important role in the treatment of chronic obstructive pulmonary disease (COPD) exacerbations. They are recommended to shorten recovery time and increase forced expiratory volume in 1 second (FEV1) during exacerbations.1 However, the role of the chronic use of inhaled corticosteroids (ICSs) in the treatment of COPD is less clear.
When added to inhaled β-2 agonists and muscarinic antagonists, ICSs can decrease the risk of exacerbations.1 However, not all patients with COPD benefit from ICS therapy. The degree of benefit an ICS can provide has been shown to correlate with eosinophil count—a marker of inflammation. The expected benefit of using an ICS increases as the eosinophil count increases.1 Maximum benefit can be observed with eosinophil counts ≥ 300 cells/µL, and minimal benefit is observed with eosinophil counts < 100 cells/µL. Adverse effects (AEs) of ICSs include a hoarse voice, oral candidiasis, and an increased risk of pneumonia.1 Given the risk of AEs, it is important to limit ICS use in patients who are unlikely to reap any benefits.
The Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines suggest the use of ICSs in patients who experience exacerbations while using long-acting β agonist (LABA) plus long-acting muscarinic antagonist (LAMA) therapy and have an eosinophil count ≥ 100 cells/µL. Switching from LABA or LAMA monotherapy to triple therapy with LAMA/LABA/ICS may be considered if patients have continued exacerbations and an eosinophil count ≥ 300 cells/µL. De-escalation of ICS therapy should be considered if patients do not meet these criteria or if patients experience ICS AEs, such as pneumonia. The patients most likely to have increased exacerbations or decreased FEV1 with ICS withdrawal are those with eosinophil counts ≥ 300 cells/µL.1,2
Several studies have explored the effects of ICS de-escalation in real-world clinical settings. A systematic review of 11 studies indicated that de-escalation of ICS in COPD does not result in increased exacerbations.3 A prospective study by Rossi et al found that in a 6-month period, 141 of 482 patients on ICS therapy (29%) had an exacerbation. In the opposing arm of the study, 88 of 334 patients (26%) with deprescribed ICS experienced an exacerbation. The difference between these 2 groups was not statistically significant.4 The researchers concluded that in real-world practice, ICS withdrawal can be safe in patients at low risk of exacerbation.
About 25% of veterans (1.25 million) have been diagnosed with COPD.5 To address this, the US Department of Veterans Affairs (VA) and US Department of Defense published updated COPD guidelines in 2021 that specify criteria for de-escalation of ICS.6 Guidelines, however, may not be reflected in common clinical practice for several years following publication. The VA Academic Detailing Service (ADS) provides tools to help clinicians identify patients who may benefit from changes in treatment plans. A recent ADS focus was the implementation of a COPD dashboard, which identifies patients with COPD who are candidates for ICS de-escalation based on comorbid diagnoses, exacerbation history, and eosinophil count. VA pharmacists have an expanded role in the management of primary care disease states and are therefore well-positioned to increase adherence to guideline-directed therapy. The objective of this quality improvement project was to determine the impact of pharmacist-driven de-escalation on ICS usage in veterans with COPD.
Methods
This project was conducted in an outpatient clinic at the Robley Rex VA Medical Center beginning September 21, 2023, with a progress note in the Computerized Patient Record System (CPRS). Eligible patients were selected using the COPD Dashboard provided by ADS. The COPD Dashboard defined patients with COPD as those with ≥ 2 outpatient COPD diagnoses in the past 2 years, 1 inpatient discharge COPD diagnosis in the past year, or COPD listed as an active problem. COPD diagnoses were identified using International Statistical Classification of Disease, Tenth Revision (ICD-10) codes
Candidates identified for ICS de-escalation by the dashboard were excluded if they had a history of COPD exacerbation in the previous 2 years. The dashboard identified COPD exacerbations via ICD-10 codes for COPD or acute respiratory failure for inpatient discharges, emergency department (ED) visits, urgent care visits, and community care consults with 1 of the following terms: emergency, inpatient, hospital, urgent, ED (self). The COPD dashboard excluded patients with a diagnosis of asthma.
After patients were selected, they were screened for additional exclusion criteria. Patients were excluded if a pulmonary care practitioner managed their COPD; if identified via an active pulmonary consult in CPRS; if a non-VA clinician prescribed their ICS; or if they were being treated with roflumilast, theophylline, or chronic azithromycin. Individuals taking these 3 drugs were excluded due to potential severe and/or refractory COPD. Patients also were excluded if they: (1) had prior ICS de-escalation failure (defined as a COPD exacerbation following ICS de-escalation that resulted in ICS resumption); (2) had a COPD exacerbation requiring systemic corticosteroids or antibiotics in the previous year; (3) had active lung cancer; (4) did not have any eosinophil levels in CPRS within the previous 2 years; or (5) had any eosinophil levels ≥ 300 cells/µL in the previous year.
Each patient who met the inclusion criteria and was not excluded received a focused medication review by a pharmacist who created a templated progress note, with patient-specific recommendations, that was entered in the CPRS (eAppendix). The recommendations were also attached as an addendum to the patient’s last primary care visit note, and the primary care practitioner (PCP) was alerted via CPRS to consider ICS de-escalation and non-ICS alternatives. Tapering of ICS therapy was offered as an option to de-escalate if abrupt discontinuation was deemed inappropriate. PCPs were also prompted to consider referral to a primary care clinical pharmacy specialist for management and follow-up of ICS de-escalation.
The primary outcome was the number of patients with de-escalated ICS at 3 and 6 months following the recommendation. Secondary outcomes included the number of: patients who were no longer prescribed an ICS or who had a non-ICS alternative initiated at a pharmacist’s recommendation; patients who were referred to a primary care clinical pharmacy specialist for ICS de-escalation; COPD exacerbations requiring systemic steroids or antibiotics, or requiring an ED visit, inpatient admission, or urgent-care clinic visit; and cases of pneumonia or oral candidiasis. Primary and secondary outcomes were evaluated via chart review in CPRS. For secondary outcomes of pneumonia and COPD exacerbation, identification was made by documented diagnosis in CPRS. For continuous data such as age, the mean was calculated.
Results
Pharmacist ICS de-escalation recommendations were made between September 21, 2023, and November 19, 2023, for 106 patients. The mean age was 72 years and 99 (93%) patients were male (Table 1). Forty-one (39%) of the patients used tobacco at the time of the study. FEV1 was available for 69 patients with a mean of 63% (GOLD grade 2).1 Based on FEV1 values, 16 patients had mild COPD (GOLD grade 1), 37 patients had moderate COPD (GOLD grade 2), 14 patients had severe COPD (GOLD grade 3), and 2 patients had very severe COPD (GOLD grade 4).1 Thirty-four patients received LABA + LAMA + ICS, 65 received LABA + ICS, 2 received LAMA + ICS, and 5 received ICS monotherapy. The most common dose of ICS was a moderate dose (Table 2). Only 2 patients had an ICS AE in the previous year.


ICS de-escalation recommendations resulted in ICS de-escalation in 50 (47.2%) and 62 (58.5%) patients at 3 and 6 months, respectively. The 6-month ICS de-escalation rate by ICS dose at baseline was 72.2% (high dose), 60.0% (moderate), and 30.8% (low). De-escalation at 6 months by GOLD grade at baseline was 56.3% (9 of 16 patients, GOLD 1), 64.9% (24 of 37 patients, GOLD 2), 50% (7 of 14 patients, GOLD 3), and 50% (1 of 2 patients, GOLD 4). Six months after the ICS de-escalation recommendation appeared in the CPRS, the percentage of patients on LABA + ICS therapy dropped from 65 patients (61.3%) at baseline to 25 patients (23.6%).
Secondary outcomes were assessed at 3 and 6 months following the recommendation. Most patients with de-escalated ICS had their ICS discontinued and a non-ICS alternative initiated per pharmacist recommendations. At 6 months, 39 patients (36.8%) patients were referred to a patient aligned care team (PACT) pharmacist for de-escalation. Of the 39 patients referred to pharmacists, 69.2% (27 patients) were de-escalated; this compared to 52.2% (35 patients) who were not referred to pharmacists (Table 3).

ICS use increases the risk of pneumonia.1 At 6 months, 11 patients were diagnosed with pneumonia; 3 patients were diagnosed with pneumonia twice, resulting in a total of 14 cases. Ten cases occurred while patients were on ICS and 4 cases occurred following ICS de-escalation. One patient had a documented case of oral candidiasis that occurred while on ICS therapy; no patients with discontinued ICS were diagnosed with oral candidiasis. In addition, 10 patients had COPD exacerbations; however no patients had exacerbations both before and after de-escalation. Six patients were on ICS therapy when they experienced an exacerbation, and 4 patients had an exacerbation after ICS de-escalation.
Discussion
More than half of patients receiving the pharmacist intervention achieved the primary outcome of ICS de-escalation at 6 months. Furthermore, a larger percentage of patients referred to pharmacists for the management of ICS de-escalation successfully achieved de-escalation compared to those who were not referred. These outcomes reflect the important role pharmacists can play in identifying appropriate candidates for ICS de-escalation and assisting in the management of ICS de-escalation. Patients referred to pharmacists also received other services such as smoking cessation pharmacotherapy and counseling on inhaler technique and adherence. These interventions can support improved COPD clinical outcomes.
The purpose of de-escalating ICS therapy is to reduce the risk of AEs such as pneumonia and oral candidiasis.1 The secondary outcomes of this study support previous evidence that patients who have de-escalated ICS therapy may have reduced risk of AEs compared to those who remain on ICS therapy.3 Specifically, of the 14 cases of pneumonia that occurred during the study, 10 cases occurred while patients were on ICS and 4 cases occurred following ICS de-escalation.
ICS de-escalation may increase risk of increased COPD exacerbations.1 However, the secondary outcomes of this study do not indicate that those with de-escalated ICS had more COPD exacerbations compared to those who continued on ICS. Pharmacists’ recommendations were more effective for patients with less severe COPD based on baseline FEV1.
The previous GOLD Guidelines for COPD suggested LABA + ICS therapy as an option for patients with a high symptom and exacerbation burden (previously known as GOLD Group D). Guidelines no longer recommend LABA + ICS therapy due to the superiority of triple inhaled therapy for exacerbations and the superiority of LAMA + LABA therapy for dyspnea.7 A majority of identified patients in this project were on LABA + ICS therapy alone at baseline. The ICS de-escalation recommendation resulted in a 61.5% reduction in patients on LABA + ICS therapy at 6 months. By decreasing the number of patients on LABA + ICS without LAMA, recommendations increased the number of patients on guideline-directed therapy.
Limitations
This study lacked a control group, and the rate of ICS de-escalation in patients who did not receive a pharmacist recommendation was not assessed. Therefore, it could not be determined whether the pharmacist recommendation is more effective than no recommendation. Another limitation was our inability to access records from non-VA health care facilities. This may have resulted in missed COPD exacerbations, pneumonia, and oral candidiasis prior to or following the pharmacist recommendation.
In addition, the method used to notify PCPs of the pharmacist recommendation was a CPRS alert. Clinicians often receive multiple daily alerts and may not always pay close attention to them due to alert fatigue. Early in the study, some PCPs were unknowingly omitted from the alert of the pharmacist recommendation for 10 patients due to human error. For 8 of these 10 patients, the PCP was notified of the recommendations during the 3-month follow-up period. However, 2 patients had COPD exacerbations during the 3-month follow-up period. In these cases, the PCP was not alerted to de-escalate ICS. The data for these patients were collected at 3 and 6 months in the same manner as all other patients. Also, 7 of 35 patients who were referred to a pharmacist for ICS de-escalation did not have a scheduled appointment. These patients were considered to be lost to follow-up and this may have resulted in an underestimation of the ability of pharmacists to successfully de-escalate ICS in patients with COPD.
Other studies have evaluated the efficacy of a pharmacy-driven ICS de-escalation.8,9 Hegland et al reported ICS de-escalation for 22% of 141 eligible ambulatory patients with COPD on triple inhaled therapy following pharmacist appointments.8 A study by Hahn et al resulted in 63.8% of 58 patients with COPD being maintained off ICS following a pharmacist de-escalation initiative.9 However, these studies relied upon more time-consuming de-escalation interventions, including at least 1 phone, video, or in-person patient visit.8,9
This project used a single chart review and templated progress note to recommend ICS de-escalation and achieved similar or improved de-escalation rates compared to previous studies.8,9 Previous studies were conducted prior to the updated 2023 GOLD guidelines for COPD which no longer recommend LABA + ICS therapy. This project addressed ICS de-escalation in patients on LABA + ICS therapy in addition to those on triple inhaled therapy. Additionally, previous studies did not address rates of moderate to severe COPD exacerbation and adverse events to ICS following the pharmacist intervention.8,9
This study included COPD exacerbations and cases of pneumonia or oral candidiasis as secondary outcomes to assess the safety and efficacy of the ICS de-escalation. It appeared there were similar or lower rates of COPD exacerbations, pneumonia, and oral candidiasis in those with de-escalated ICS therapy in this study. However, these secondary outcomes are exploratory and would need to be confirmed by larger studies powered to address these outcomes.
CONCLUSIONS
Pharmacist-driven ICS de-escalation may be an effective method for reducing ICS usage in veterans as seen in this study. Additional controlled studies are required to evaluate the efficacy and safety of pharmacist-driven ICS de-escalation.

Systemic glucocorticoids play an important role in the treatment of chronic obstructive pulmonary disease (COPD) exacerbations. They are recommended to shorten recovery time and increase forced expiratory volume in 1 second (FEV1) during exacerbations.1 However, the role of the chronic use of inhaled corticosteroids (ICSs) in the treatment of COPD is less clear.
When added to inhaled β-2 agonists and muscarinic antagonists, ICSs can decrease the risk of exacerbations.1 However, not all patients with COPD benefit from ICS therapy. The degree of benefit an ICS can provide has been shown to correlate with eosinophil count—a marker of inflammation. The expected benefit of using an ICS increases as the eosinophil count increases.1 Maximum benefit can be observed with eosinophil counts ≥ 300 cells/µL, and minimal benefit is observed with eosinophil counts < 100 cells/µL. Adverse effects (AEs) of ICSs include a hoarse voice, oral candidiasis, and an increased risk of pneumonia.1 Given the risk of AEs, it is important to limit ICS use in patients who are unlikely to reap any benefits.
The Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines suggest the use of ICSs in patients who experience exacerbations while using long-acting β agonist (LABA) plus long-acting muscarinic antagonist (LAMA) therapy and have an eosinophil count ≥ 100 cells/µL. Switching from LABA or LAMA monotherapy to triple therapy with LAMA/LABA/ICS may be considered if patients have continued exacerbations and an eosinophil count ≥ 300 cells/µL. De-escalation of ICS therapy should be considered if patients do not meet these criteria or if patients experience ICS AEs, such as pneumonia. The patients most likely to have increased exacerbations or decreased FEV1 with ICS withdrawal are those with eosinophil counts ≥ 300 cells/µL.1,2
Several studies have explored the effects of ICS de-escalation in real-world clinical settings. A systematic review of 11 studies indicated that de-escalation of ICS in COPD does not result in increased exacerbations.3 A prospective study by Rossi et al found that in a 6-month period, 141 of 482 patients on ICS therapy (29%) had an exacerbation. In the opposing arm of the study, 88 of 334 patients (26%) with deprescribed ICS experienced an exacerbation. The difference between these 2 groups was not statistically significant.4 The researchers concluded that in real-world practice, ICS withdrawal can be safe in patients at low risk of exacerbation.
About 25% of veterans (1.25 million) have been diagnosed with COPD.5 To address this, the US Department of Veterans Affairs (VA) and US Department of Defense published updated COPD guidelines in 2021 that specify criteria for de-escalation of ICS.6 Guidelines, however, may not be reflected in common clinical practice for several years following publication. The VA Academic Detailing Service (ADS) provides tools to help clinicians identify patients who may benefit from changes in treatment plans. A recent ADS focus was the implementation of a COPD dashboard, which identifies patients with COPD who are candidates for ICS de-escalation based on comorbid diagnoses, exacerbation history, and eosinophil count. VA pharmacists have an expanded role in the management of primary care disease states and are therefore well-positioned to increase adherence to guideline-directed therapy. The objective of this quality improvement project was to determine the impact of pharmacist-driven de-escalation on ICS usage in veterans with COPD.
Methods
This project was conducted in an outpatient clinic at the Robley Rex VA Medical Center beginning September 21, 2023, with a progress note in the Computerized Patient Record System (CPRS). Eligible patients were selected using the COPD Dashboard provided by ADS. The COPD Dashboard defined patients with COPD as those with ≥ 2 outpatient COPD diagnoses in the past 2 years, 1 inpatient discharge COPD diagnosis in the past year, or COPD listed as an active problem. COPD diagnoses were identified using International Statistical Classification of Disease, Tenth Revision (ICD-10) codes
Candidates identified for ICS de-escalation by the dashboard were excluded if they had a history of COPD exacerbation in the previous 2 years. The dashboard identified COPD exacerbations via ICD-10 codes for COPD or acute respiratory failure for inpatient discharges, emergency department (ED) visits, urgent care visits, and community care consults with 1 of the following terms: emergency, inpatient, hospital, urgent, ED (self). The COPD dashboard excluded patients with a diagnosis of asthma.
After patients were selected, they were screened for additional exclusion criteria. Patients were excluded if a pulmonary care practitioner managed their COPD; if identified via an active pulmonary consult in CPRS; if a non-VA clinician prescribed their ICS; or if they were being treated with roflumilast, theophylline, or chronic azithromycin. Individuals taking these 3 drugs were excluded due to potential severe and/or refractory COPD. Patients also were excluded if they: (1) had prior ICS de-escalation failure (defined as a COPD exacerbation following ICS de-escalation that resulted in ICS resumption); (2) had a COPD exacerbation requiring systemic corticosteroids or antibiotics in the previous year; (3) had active lung cancer; (4) did not have any eosinophil levels in CPRS within the previous 2 years; or (5) had any eosinophil levels ≥ 300 cells/µL in the previous year.
Each patient who met the inclusion criteria and was not excluded received a focused medication review by a pharmacist who created a templated progress note, with patient-specific recommendations, that was entered in the CPRS (eAppendix). The recommendations were also attached as an addendum to the patient’s last primary care visit note, and the primary care practitioner (PCP) was alerted via CPRS to consider ICS de-escalation and non-ICS alternatives. Tapering of ICS therapy was offered as an option to de-escalate if abrupt discontinuation was deemed inappropriate. PCPs were also prompted to consider referral to a primary care clinical pharmacy specialist for management and follow-up of ICS de-escalation.
The primary outcome was the number of patients with de-escalated ICS at 3 and 6 months following the recommendation. Secondary outcomes included the number of: patients who were no longer prescribed an ICS or who had a non-ICS alternative initiated at a pharmacist’s recommendation; patients who were referred to a primary care clinical pharmacy specialist for ICS de-escalation; COPD exacerbations requiring systemic steroids or antibiotics, or requiring an ED visit, inpatient admission, or urgent-care clinic visit; and cases of pneumonia or oral candidiasis. Primary and secondary outcomes were evaluated via chart review in CPRS. For secondary outcomes of pneumonia and COPD exacerbation, identification was made by documented diagnosis in CPRS. For continuous data such as age, the mean was calculated.
Results
Pharmacist ICS de-escalation recommendations were made between September 21, 2023, and November 19, 2023, for 106 patients. The mean age was 72 years and 99 (93%) patients were male (Table 1). Forty-one (39%) of the patients used tobacco at the time of the study. FEV1 was available for 69 patients with a mean of 63% (GOLD grade 2).1 Based on FEV1 values, 16 patients had mild COPD (GOLD grade 1), 37 patients had moderate COPD (GOLD grade 2), 14 patients had severe COPD (GOLD grade 3), and 2 patients had very severe COPD (GOLD grade 4).1 Thirty-four patients received LABA + LAMA + ICS, 65 received LABA + ICS, 2 received LAMA + ICS, and 5 received ICS monotherapy. The most common dose of ICS was a moderate dose (Table 2). Only 2 patients had an ICS AE in the previous year.


ICS de-escalation recommendations resulted in ICS de-escalation in 50 (47.2%) and 62 (58.5%) patients at 3 and 6 months, respectively. The 6-month ICS de-escalation rate by ICS dose at baseline was 72.2% (high dose), 60.0% (moderate), and 30.8% (low). De-escalation at 6 months by GOLD grade at baseline was 56.3% (9 of 16 patients, GOLD 1), 64.9% (24 of 37 patients, GOLD 2), 50% (7 of 14 patients, GOLD 3), and 50% (1 of 2 patients, GOLD 4). Six months after the ICS de-escalation recommendation appeared in the CPRS, the percentage of patients on LABA + ICS therapy dropped from 65 patients (61.3%) at baseline to 25 patients (23.6%).
Secondary outcomes were assessed at 3 and 6 months following the recommendation. Most patients with de-escalated ICS had their ICS discontinued and a non-ICS alternative initiated per pharmacist recommendations. At 6 months, 39 patients (36.8%) patients were referred to a patient aligned care team (PACT) pharmacist for de-escalation. Of the 39 patients referred to pharmacists, 69.2% (27 patients) were de-escalated; this compared to 52.2% (35 patients) who were not referred to pharmacists (Table 3).

ICS use increases the risk of pneumonia.1 At 6 months, 11 patients were diagnosed with pneumonia; 3 patients were diagnosed with pneumonia twice, resulting in a total of 14 cases. Ten cases occurred while patients were on ICS and 4 cases occurred following ICS de-escalation. One patient had a documented case of oral candidiasis that occurred while on ICS therapy; no patients with discontinued ICS were diagnosed with oral candidiasis. In addition, 10 patients had COPD exacerbations; however no patients had exacerbations both before and after de-escalation. Six patients were on ICS therapy when they experienced an exacerbation, and 4 patients had an exacerbation after ICS de-escalation.
Discussion
More than half of patients receiving the pharmacist intervention achieved the primary outcome of ICS de-escalation at 6 months. Furthermore, a larger percentage of patients referred to pharmacists for the management of ICS de-escalation successfully achieved de-escalation compared to those who were not referred. These outcomes reflect the important role pharmacists can play in identifying appropriate candidates for ICS de-escalation and assisting in the management of ICS de-escalation. Patients referred to pharmacists also received other services such as smoking cessation pharmacotherapy and counseling on inhaler technique and adherence. These interventions can support improved COPD clinical outcomes.
The purpose of de-escalating ICS therapy is to reduce the risk of AEs such as pneumonia and oral candidiasis.1 The secondary outcomes of this study support previous evidence that patients who have de-escalated ICS therapy may have reduced risk of AEs compared to those who remain on ICS therapy.3 Specifically, of the 14 cases of pneumonia that occurred during the study, 10 cases occurred while patients were on ICS and 4 cases occurred following ICS de-escalation.
ICS de-escalation may increase risk of increased COPD exacerbations.1 However, the secondary outcomes of this study do not indicate that those with de-escalated ICS had more COPD exacerbations compared to those who continued on ICS. Pharmacists’ recommendations were more effective for patients with less severe COPD based on baseline FEV1.
The previous GOLD Guidelines for COPD suggested LABA + ICS therapy as an option for patients with a high symptom and exacerbation burden (previously known as GOLD Group D). Guidelines no longer recommend LABA + ICS therapy due to the superiority of triple inhaled therapy for exacerbations and the superiority of LAMA + LABA therapy for dyspnea.7 A majority of identified patients in this project were on LABA + ICS therapy alone at baseline. The ICS de-escalation recommendation resulted in a 61.5% reduction in patients on LABA + ICS therapy at 6 months. By decreasing the number of patients on LABA + ICS without LAMA, recommendations increased the number of patients on guideline-directed therapy.
Limitations
This study lacked a control group, and the rate of ICS de-escalation in patients who did not receive a pharmacist recommendation was not assessed. Therefore, it could not be determined whether the pharmacist recommendation is more effective than no recommendation. Another limitation was our inability to access records from non-VA health care facilities. This may have resulted in missed COPD exacerbations, pneumonia, and oral candidiasis prior to or following the pharmacist recommendation.
In addition, the method used to notify PCPs of the pharmacist recommendation was a CPRS alert. Clinicians often receive multiple daily alerts and may not always pay close attention to them due to alert fatigue. Early in the study, some PCPs were unknowingly omitted from the alert of the pharmacist recommendation for 10 patients due to human error. For 8 of these 10 patients, the PCP was notified of the recommendations during the 3-month follow-up period. However, 2 patients had COPD exacerbations during the 3-month follow-up period. In these cases, the PCP was not alerted to de-escalate ICS. The data for these patients were collected at 3 and 6 months in the same manner as all other patients. Also, 7 of 35 patients who were referred to a pharmacist for ICS de-escalation did not have a scheduled appointment. These patients were considered to be lost to follow-up and this may have resulted in an underestimation of the ability of pharmacists to successfully de-escalate ICS in patients with COPD.
Other studies have evaluated the efficacy of a pharmacy-driven ICS de-escalation.8,9 Hegland et al reported ICS de-escalation for 22% of 141 eligible ambulatory patients with COPD on triple inhaled therapy following pharmacist appointments.8 A study by Hahn et al resulted in 63.8% of 58 patients with COPD being maintained off ICS following a pharmacist de-escalation initiative.9 However, these studies relied upon more time-consuming de-escalation interventions, including at least 1 phone, video, or in-person patient visit.8,9
This project used a single chart review and templated progress note to recommend ICS de-escalation and achieved similar or improved de-escalation rates compared to previous studies.8,9 Previous studies were conducted prior to the updated 2023 GOLD guidelines for COPD which no longer recommend LABA + ICS therapy. This project addressed ICS de-escalation in patients on LABA + ICS therapy in addition to those on triple inhaled therapy. Additionally, previous studies did not address rates of moderate to severe COPD exacerbation and adverse events to ICS following the pharmacist intervention.8,9
This study included COPD exacerbations and cases of pneumonia or oral candidiasis as secondary outcomes to assess the safety and efficacy of the ICS de-escalation. It appeared there were similar or lower rates of COPD exacerbations, pneumonia, and oral candidiasis in those with de-escalated ICS therapy in this study. However, these secondary outcomes are exploratory and would need to be confirmed by larger studies powered to address these outcomes.
CONCLUSIONS
Pharmacist-driven ICS de-escalation may be an effective method for reducing ICS usage in veterans as seen in this study. Additional controlled studies are required to evaluate the efficacy and safety of pharmacist-driven ICS de-escalation.

- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2024 Report). Accessed October 14, 2025. https://goldcopd.org/2024-gold-report/
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2025 Report). Accessed November 14, 2025. https://goldcopd.org/2025-gold-report/
- Rogliani P, Ritondo BL, Gabriele M, et al. Optimizing de-escalation of inhaled corticosteroids in COPD: a systematic review of real-world findings. Expert Rev Clin Pharmacol. 2020;13(9):977-990. doi:10.1080/17512433.2020.1817739
- Rossi A, Guerriero M, Corrado A; OPTIMO/AIPO Study Group. Withdrawal of inhaled corticosteroids can be safe in COPD patients at low risk of exacerbation: a real-life study on the appropriateness of treatment in moderate COPD patients (OPTIMO). Respir Res. 2014;15(1):77. doi:10.1186/1465-9921-15-77
- Anderson E, Wiener RS, Resnick K, et al. Care coordination for veterans with COPD: a positive deviance study. Am J Manag Care. 2020;26(2):63-68. doi:10.37765/ajmc.2020.42394
- US Department of Veterans Affairs, US Department of Defense. VA/DoD Clinical Practice Guideline for the Management of Chronic Obstructive Pulmonary Disease. 2021. Accessed October 14, 2025. https://www.healthquality.va.gov/guidelines/CD/copd/
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2023 Report). Accessed October 14, 2025. https://goldcopd.org/wp-content/uploads/2023/03/GOLD-2023-ver-1.3-17Feb2023_WMV.pdf
- Hegland AJ, Bolduc J, Jones L, Kunisaki KM, Melzer AC. Pharmacist-driven deprescribing of inhaled corticosteroids in patients with stable chronic obstructive pulmonary disease. Ann Am Thorac Soc. 2021;18(4):730-733. doi:10.1513/AnnalsATS.202007-871RL
- Hahn NM, Nagy MW. Implementation of a targeted inhaled corticosteroid de-escalation process in patients with chronic obstructive pulmonary disease in the primary care setting. Innov Pharm. 2022;13(1):10.24926/iip.v13i1.4349. doi:10.24926/iip.v13i1.4349
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2024 Report). Accessed October 14, 2025. https://goldcopd.org/2024-gold-report/
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2025 Report). Accessed November 14, 2025. https://goldcopd.org/2025-gold-report/
- Rogliani P, Ritondo BL, Gabriele M, et al. Optimizing de-escalation of inhaled corticosteroids in COPD: a systematic review of real-world findings. Expert Rev Clin Pharmacol. 2020;13(9):977-990. doi:10.1080/17512433.2020.1817739
- Rossi A, Guerriero M, Corrado A; OPTIMO/AIPO Study Group. Withdrawal of inhaled corticosteroids can be safe in COPD patients at low risk of exacerbation: a real-life study on the appropriateness of treatment in moderate COPD patients (OPTIMO). Respir Res. 2014;15(1):77. doi:10.1186/1465-9921-15-77
- Anderson E, Wiener RS, Resnick K, et al. Care coordination for veterans with COPD: a positive deviance study. Am J Manag Care. 2020;26(2):63-68. doi:10.37765/ajmc.2020.42394
- US Department of Veterans Affairs, US Department of Defense. VA/DoD Clinical Practice Guideline for the Management of Chronic Obstructive Pulmonary Disease. 2021. Accessed October 14, 2025. https://www.healthquality.va.gov/guidelines/CD/copd/
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2023 Report). Accessed October 14, 2025. https://goldcopd.org/wp-content/uploads/2023/03/GOLD-2023-ver-1.3-17Feb2023_WMV.pdf
- Hegland AJ, Bolduc J, Jones L, Kunisaki KM, Melzer AC. Pharmacist-driven deprescribing of inhaled corticosteroids in patients with stable chronic obstructive pulmonary disease. Ann Am Thorac Soc. 2021;18(4):730-733. doi:10.1513/AnnalsATS.202007-871RL
- Hahn NM, Nagy MW. Implementation of a targeted inhaled corticosteroid de-escalation process in patients with chronic obstructive pulmonary disease in the primary care setting. Innov Pharm. 2022;13(1):10.24926/iip.v13i1.4349. doi:10.24926/iip.v13i1.4349
Evaluation of Pharmacist-Driven Inhaled Corticosteroid De-escalation in Veterans
Evaluation of Pharmacist-Driven Inhaled Corticosteroid De-escalation in Veterans
Nine VA Facilities to Open Research Trials for Psychedelics
Nine VA Facilities to Open Research Trials for Psychedelics
On Nov. 22, 2014, 8 years after he came back from Iraq with “crippling” posttraumatic stress disorder (PTSD), Jonathan Lubecky took his first dose of the psychedelic compound methylenedioxymethamphetamine (MDMA). Lubecky, a Marine, Army, and National Guard veteran, described his path to MDMA therapy in in the New Horizons in Health podcast.
After 5 suicide attempts and “the hundreds of times I thought about it or stood on a bridge or had a plan,” he felt he had run out of options. Then, in a counseling session, a psychiatric intern slid a piece of paper across the table to him. It read “Google MDMA PTSD.”
Luckily for Lubecky, a space in a clinical trial opened up, in which he had 8 hours of talk therapy with specially trained therapists, combined with MDMA. “MDMA is a tool that opens up the mind, body and spirit,” he said, “so you can heal and process all those memories and traumas that are causing yourissues. It puts you in a middle place where you can talk about trauma without having panic attacks, without your body betraying you, and look at it from a different perspective.” said he added, “It’s like doing therapy while being hugged by everyone who loves you in a bathtub full of puppies licking your face.” In 2023, 9 years after that first dose, Lubecky said, “I’ve been PTSD free longer than I had it.”
And now, in 2025, the research into psychedelic therapy for veterans like Lubecky is taking another step forward according to a report by Military.com. Nine VA facilities, in the Bronx, Los Angeles, Omaha, Palo Alto, Portland (Oregon), San Diego, San Francisco, West Haven, and White River Junction, are participating in long-term studies to test the safety and clinical impact of psychedelic compounds for PTSD, treatment-resistant depression, and anxiety disorders.
Early trials from Johns Hopkins University, the Multidisciplinary Association for Psychedelic Studies (MAPS), and others found significant symptom reductions for some participants with chronic PTSD. MAPP2, the multisite phase 3 study that extended the findings of MAPP1, found that MDMA-assisted therapy significantly improved PTSD symptoms and functional impairment, compared with placebo-assisted therapy. Notably, of the 52 participants (including 16 veterans) 45 (86%) achieved a clinically meaningful benefit, and 37 (71%) no longer met criteria for PTSD by study end. Despite the promising findings, a US Food and Drug Administration (FDA) advisory panel recommended against approving the treatment.
In 2024 the VA issued a request for applications for proposals from its network of VA researchers and academic institutions to gather “definitive scientific evidence” on the potential efficacy and safety of psychedelic compounds, such as MDMA and psilocybin, when used in conjunction with psychotherapy. It would be the first time since the 1960s that the VA had funded research on such compounds.
Funding proposals for such research have cycled in and out of Congress for years, but have gathered more steam in the last few years. The 2024 National Defense Authorization Act directed the US Department of Defense to establish a process for funding clinical research into the use of certain psychedelic substances to treat PTSD and traumatic brain injury. In April 2024, Representatives Lou Correa (D-CA) and Jack Bergman (R-MI), cochairs of the Psychedelics Advancing Therapies (PATH) caucus, introduced the Innovative Therapies Centers of Excellence Act of 2025, bipartisan legislation that would increase federally funded research on innovative therapies to treat veterans with PTSD, substance use disorder, and depression. It would also, if enacted, direct the VA to create ≥ 5 dedicated centers of excellence to study the therapeutic uses of psychedelic substances. The bill has also been endorsed by the American Legion, Veterans of Foreign Wars, Iraq and Afghanistan Veterans of America, Disabled American Veterans, and the Wounded Warrior Project.
The current administration has two strong high-level supporters of psychedelics research: VA Secretary Doug Collins and US Department of Health and Human Service Secretary Robert F. Kennedy Jr. Sec. Kennedy has castigated the FDA for what he calls “aggressive suppression” of alternative and complementary treatments, including psychedelics. This, although the FDA granted breakthrough therapy status for MDMA for treating PTSD and psilocybin for treating depression in 2018 and 2019, respectively, as well a pivotal draft guidance in 2023 for the development of psychedelic drugs for psychiatric disorders, substance use disorders, and various medical conditions.
Collins, citing an “eye-opening” discussion with Kennedy, enthusiastically backs the research into psychedelics. In a May 2025 hearing that was mainly a series of testy exchanges about his proposed budget slashing, he emphasized the importance of keeping and expanding VA programs and studies on psychedelic treatments, something he has been advocating for since the beginning of his appointment. “We want to make sure we’re not closing off any outlet for a veteran who could be helped by these programs,” he said.
Taking the intern’s advice to look into MDMA, Jonathan Lubecky said, was one of the best decisions he’d ever made. But “it’s not the MDMA that fixes you,” he said. “It’s the therapy. It’s the therapist working with you and you doing the hard work.”
On Nov. 22, 2014, 8 years after he came back from Iraq with “crippling” posttraumatic stress disorder (PTSD), Jonathan Lubecky took his first dose of the psychedelic compound methylenedioxymethamphetamine (MDMA). Lubecky, a Marine, Army, and National Guard veteran, described his path to MDMA therapy in in the New Horizons in Health podcast.
After 5 suicide attempts and “the hundreds of times I thought about it or stood on a bridge or had a plan,” he felt he had run out of options. Then, in a counseling session, a psychiatric intern slid a piece of paper across the table to him. It read “Google MDMA PTSD.”
Luckily for Lubecky, a space in a clinical trial opened up, in which he had 8 hours of talk therapy with specially trained therapists, combined with MDMA. “MDMA is a tool that opens up the mind, body and spirit,” he said, “so you can heal and process all those memories and traumas that are causing yourissues. It puts you in a middle place where you can talk about trauma without having panic attacks, without your body betraying you, and look at it from a different perspective.” said he added, “It’s like doing therapy while being hugged by everyone who loves you in a bathtub full of puppies licking your face.” In 2023, 9 years after that first dose, Lubecky said, “I’ve been PTSD free longer than I had it.”
And now, in 2025, the research into psychedelic therapy for veterans like Lubecky is taking another step forward according to a report by Military.com. Nine VA facilities, in the Bronx, Los Angeles, Omaha, Palo Alto, Portland (Oregon), San Diego, San Francisco, West Haven, and White River Junction, are participating in long-term studies to test the safety and clinical impact of psychedelic compounds for PTSD, treatment-resistant depression, and anxiety disorders.
Early trials from Johns Hopkins University, the Multidisciplinary Association for Psychedelic Studies (MAPS), and others found significant symptom reductions for some participants with chronic PTSD. MAPP2, the multisite phase 3 study that extended the findings of MAPP1, found that MDMA-assisted therapy significantly improved PTSD symptoms and functional impairment, compared with placebo-assisted therapy. Notably, of the 52 participants (including 16 veterans) 45 (86%) achieved a clinically meaningful benefit, and 37 (71%) no longer met criteria for PTSD by study end. Despite the promising findings, a US Food and Drug Administration (FDA) advisory panel recommended against approving the treatment.
In 2024 the VA issued a request for applications for proposals from its network of VA researchers and academic institutions to gather “definitive scientific evidence” on the potential efficacy and safety of psychedelic compounds, such as MDMA and psilocybin, when used in conjunction with psychotherapy. It would be the first time since the 1960s that the VA had funded research on such compounds.
Funding proposals for such research have cycled in and out of Congress for years, but have gathered more steam in the last few years. The 2024 National Defense Authorization Act directed the US Department of Defense to establish a process for funding clinical research into the use of certain psychedelic substances to treat PTSD and traumatic brain injury. In April 2024, Representatives Lou Correa (D-CA) and Jack Bergman (R-MI), cochairs of the Psychedelics Advancing Therapies (PATH) caucus, introduced the Innovative Therapies Centers of Excellence Act of 2025, bipartisan legislation that would increase federally funded research on innovative therapies to treat veterans with PTSD, substance use disorder, and depression. It would also, if enacted, direct the VA to create ≥ 5 dedicated centers of excellence to study the therapeutic uses of psychedelic substances. The bill has also been endorsed by the American Legion, Veterans of Foreign Wars, Iraq and Afghanistan Veterans of America, Disabled American Veterans, and the Wounded Warrior Project.
The current administration has two strong high-level supporters of psychedelics research: VA Secretary Doug Collins and US Department of Health and Human Service Secretary Robert F. Kennedy Jr. Sec. Kennedy has castigated the FDA for what he calls “aggressive suppression” of alternative and complementary treatments, including psychedelics. This, although the FDA granted breakthrough therapy status for MDMA for treating PTSD and psilocybin for treating depression in 2018 and 2019, respectively, as well a pivotal draft guidance in 2023 for the development of psychedelic drugs for psychiatric disorders, substance use disorders, and various medical conditions.
Collins, citing an “eye-opening” discussion with Kennedy, enthusiastically backs the research into psychedelics. In a May 2025 hearing that was mainly a series of testy exchanges about his proposed budget slashing, he emphasized the importance of keeping and expanding VA programs and studies on psychedelic treatments, something he has been advocating for since the beginning of his appointment. “We want to make sure we’re not closing off any outlet for a veteran who could be helped by these programs,” he said.
Taking the intern’s advice to look into MDMA, Jonathan Lubecky said, was one of the best decisions he’d ever made. But “it’s not the MDMA that fixes you,” he said. “It’s the therapy. It’s the therapist working with you and you doing the hard work.”
On Nov. 22, 2014, 8 years after he came back from Iraq with “crippling” posttraumatic stress disorder (PTSD), Jonathan Lubecky took his first dose of the psychedelic compound methylenedioxymethamphetamine (MDMA). Lubecky, a Marine, Army, and National Guard veteran, described his path to MDMA therapy in in the New Horizons in Health podcast.
After 5 suicide attempts and “the hundreds of times I thought about it or stood on a bridge or had a plan,” he felt he had run out of options. Then, in a counseling session, a psychiatric intern slid a piece of paper across the table to him. It read “Google MDMA PTSD.”
Luckily for Lubecky, a space in a clinical trial opened up, in which he had 8 hours of talk therapy with specially trained therapists, combined with MDMA. “MDMA is a tool that opens up the mind, body and spirit,” he said, “so you can heal and process all those memories and traumas that are causing yourissues. It puts you in a middle place where you can talk about trauma without having panic attacks, without your body betraying you, and look at it from a different perspective.” said he added, “It’s like doing therapy while being hugged by everyone who loves you in a bathtub full of puppies licking your face.” In 2023, 9 years after that first dose, Lubecky said, “I’ve been PTSD free longer than I had it.”
And now, in 2025, the research into psychedelic therapy for veterans like Lubecky is taking another step forward according to a report by Military.com. Nine VA facilities, in the Bronx, Los Angeles, Omaha, Palo Alto, Portland (Oregon), San Diego, San Francisco, West Haven, and White River Junction, are participating in long-term studies to test the safety and clinical impact of psychedelic compounds for PTSD, treatment-resistant depression, and anxiety disorders.
Early trials from Johns Hopkins University, the Multidisciplinary Association for Psychedelic Studies (MAPS), and others found significant symptom reductions for some participants with chronic PTSD. MAPP2, the multisite phase 3 study that extended the findings of MAPP1, found that MDMA-assisted therapy significantly improved PTSD symptoms and functional impairment, compared with placebo-assisted therapy. Notably, of the 52 participants (including 16 veterans) 45 (86%) achieved a clinically meaningful benefit, and 37 (71%) no longer met criteria for PTSD by study end. Despite the promising findings, a US Food and Drug Administration (FDA) advisory panel recommended against approving the treatment.
In 2024 the VA issued a request for applications for proposals from its network of VA researchers and academic institutions to gather “definitive scientific evidence” on the potential efficacy and safety of psychedelic compounds, such as MDMA and psilocybin, when used in conjunction with psychotherapy. It would be the first time since the 1960s that the VA had funded research on such compounds.
Funding proposals for such research have cycled in and out of Congress for years, but have gathered more steam in the last few years. The 2024 National Defense Authorization Act directed the US Department of Defense to establish a process for funding clinical research into the use of certain psychedelic substances to treat PTSD and traumatic brain injury. In April 2024, Representatives Lou Correa (D-CA) and Jack Bergman (R-MI), cochairs of the Psychedelics Advancing Therapies (PATH) caucus, introduced the Innovative Therapies Centers of Excellence Act of 2025, bipartisan legislation that would increase federally funded research on innovative therapies to treat veterans with PTSD, substance use disorder, and depression. It would also, if enacted, direct the VA to create ≥ 5 dedicated centers of excellence to study the therapeutic uses of psychedelic substances. The bill has also been endorsed by the American Legion, Veterans of Foreign Wars, Iraq and Afghanistan Veterans of America, Disabled American Veterans, and the Wounded Warrior Project.
The current administration has two strong high-level supporters of psychedelics research: VA Secretary Doug Collins and US Department of Health and Human Service Secretary Robert F. Kennedy Jr. Sec. Kennedy has castigated the FDA for what he calls “aggressive suppression” of alternative and complementary treatments, including psychedelics. This, although the FDA granted breakthrough therapy status for MDMA for treating PTSD and psilocybin for treating depression in 2018 and 2019, respectively, as well a pivotal draft guidance in 2023 for the development of psychedelic drugs for psychiatric disorders, substance use disorders, and various medical conditions.
Collins, citing an “eye-opening” discussion with Kennedy, enthusiastically backs the research into psychedelics. In a May 2025 hearing that was mainly a series of testy exchanges about his proposed budget slashing, he emphasized the importance of keeping and expanding VA programs and studies on psychedelic treatments, something he has been advocating for since the beginning of his appointment. “We want to make sure we’re not closing off any outlet for a veteran who could be helped by these programs,” he said.
Taking the intern’s advice to look into MDMA, Jonathan Lubecky said, was one of the best decisions he’d ever made. But “it’s not the MDMA that fixes you,” he said. “It’s the therapy. It’s the therapist working with you and you doing the hard work.”
Nine VA Facilities to Open Research Trials for Psychedelics
Nine VA Facilities to Open Research Trials for Psychedelics
New Drug Eases Side Effects of Weight-Loss Meds
A new drug currently known as NG101 reduced nausea and vomiting in patients with obesity using GLP-1s by 40% and 67%, respectively, based on data from a phase 2 trial presented at the Obesity Society’s Obesity Week 2025 in Atlanta.
Previous research published in JAMA Network Open showed a nearly 65% discontinuation rate for three GLP-1s (liraglutide, semaglutide, or tirzepatide) among adults with overweight or obesity and without type 2 diabetes. Gastrointestinal (GI) side effects topped the list of reasons for dropping the medications.
Given the impact of nausea and vomiting on discontinuation, there is an unmet need for therapies to manage GI symptoms, said Kimberley Cummings, PhD, of Neurogastrx, Inc., in her presentation.
In the new study, Cummings and colleagues randomly assigned 90 adults aged 18-55 years with overweight or obesity (defined as a BMI ranging from 22.0 to 35.0) to receive a single subcutaneous dose of semaglutide (0.5 mg) plus 5 days of NG101 at 20 mg twice daily, or a placebo.
NG101 is a peripherally acting D2 antagonist designed to reduce nausea and vomiting associated with GLP-1 use, Cummings said. NG101 targets the nausea center of the brain but is peripherally restricted to prevent central nervous system side effects, she explained.
Compared with placebo, NG101 significantly reduced the incidence of nausea and vomiting by 40% and 67%, respectively. Use of NG101 also was associated with a significant reduction in the duration of nausea and vomiting; GI events lasting longer than 1 day were reported in 22% and 51% of the NG101 patients and placebo patients, respectively.
In addition, participants who received NG101 reported a 70% decrease in nausea severity from baseline.
Overall, patients in the NG101 group also reported significantly fewer adverse events than those in the placebo group (74 vs 135), suggesting an improved safety profile when semaglutide is administered in conjunction with NG101, the researchers noted. No serious adverse events related to the study drug were reported in either group.
The findings were limited by several factors including the relatively small sample size. Additional research is needed with other GLP-1 agonists in larger populations with longer follow-up periods, Cummings said. However, the results suggest that NG101 was safe and effectively improved side effects associated with GLP-1 agonists.
“We know there are receptors for GLP-1 in the area postrema (nausea center of the brain), and that NG101 works on this area to reduce nausea and vomiting, so the study findings were not unexpected,” said Jim O’Mara, president and CEO of Neurogastrx, in an interview.
The study was a single-dose study designed to show proof of concept, and future studies would involve treating patients going through the recommended titration schedule for their GLP-1s, O’Mara said. However, NG101 offers an opportunity to keep more patients on GLP-1 therapy and help them reach their long-term therapeutic goals, he said.
Decrease Side Effects for Weight-Loss Success
“GI side effects are often the rate-limiting step in implementing an effective medication that patients want to take but may not be able to tolerate,” said Sean Wharton, MD, PharmD, medical director of the Wharton Medical Clinic for Weight and Diabetes Management, Burlington, Ontario, Canada, in an interview. “If we can decrease side effects, these medications could improve patients’ lives,” said Wharton, who was not involved in the study.
The improvement after a single dose of NG101 in patients receiving a single dose of semaglutide was impressive and in keeping with the mechanism of the drug action, said Wharton. “I was not surprised by the result but pleased that this single dose was shown to reduce the overall incidence of nausea and vomiting, the duration of nausea, the severity of nausea as rated by the study participants compared to placebo,” he said.
Ultimately, the clinical implications for NG101 are improved patient tolerance for GLP-1s and the ability to titrate and stay on them long term, incurring greater cardiometabolic benefit, Wharton told this news organization.
The current trial was limited to GLP1-1s on the market; newer medications may have fewer side effects, Wharton noted. “In clinical practice, patients often decrease the medication or titrate slower, and this could be the comparator,” he added.
The study was funded by Neurogastrx.
Wharton disclosed serving as a consultant for Neurogastrx but not as an investigator on the current study. He also reported having disclosed research on various GLP-1 medications.
A version of this article first appeared on Medscape.com.
A new drug currently known as NG101 reduced nausea and vomiting in patients with obesity using GLP-1s by 40% and 67%, respectively, based on data from a phase 2 trial presented at the Obesity Society’s Obesity Week 2025 in Atlanta.
Previous research published in JAMA Network Open showed a nearly 65% discontinuation rate for three GLP-1s (liraglutide, semaglutide, or tirzepatide) among adults with overweight or obesity and without type 2 diabetes. Gastrointestinal (GI) side effects topped the list of reasons for dropping the medications.
Given the impact of nausea and vomiting on discontinuation, there is an unmet need for therapies to manage GI symptoms, said Kimberley Cummings, PhD, of Neurogastrx, Inc., in her presentation.
In the new study, Cummings and colleagues randomly assigned 90 adults aged 18-55 years with overweight or obesity (defined as a BMI ranging from 22.0 to 35.0) to receive a single subcutaneous dose of semaglutide (0.5 mg) plus 5 days of NG101 at 20 mg twice daily, or a placebo.
NG101 is a peripherally acting D2 antagonist designed to reduce nausea and vomiting associated with GLP-1 use, Cummings said. NG101 targets the nausea center of the brain but is peripherally restricted to prevent central nervous system side effects, she explained.
Compared with placebo, NG101 significantly reduced the incidence of nausea and vomiting by 40% and 67%, respectively. Use of NG101 also was associated with a significant reduction in the duration of nausea and vomiting; GI events lasting longer than 1 day were reported in 22% and 51% of the NG101 patients and placebo patients, respectively.
In addition, participants who received NG101 reported a 70% decrease in nausea severity from baseline.
Overall, patients in the NG101 group also reported significantly fewer adverse events than those in the placebo group (74 vs 135), suggesting an improved safety profile when semaglutide is administered in conjunction with NG101, the researchers noted. No serious adverse events related to the study drug were reported in either group.
The findings were limited by several factors including the relatively small sample size. Additional research is needed with other GLP-1 agonists in larger populations with longer follow-up periods, Cummings said. However, the results suggest that NG101 was safe and effectively improved side effects associated with GLP-1 agonists.
“We know there are receptors for GLP-1 in the area postrema (nausea center of the brain), and that NG101 works on this area to reduce nausea and vomiting, so the study findings were not unexpected,” said Jim O’Mara, president and CEO of Neurogastrx, in an interview.
The study was a single-dose study designed to show proof of concept, and future studies would involve treating patients going through the recommended titration schedule for their GLP-1s, O’Mara said. However, NG101 offers an opportunity to keep more patients on GLP-1 therapy and help them reach their long-term therapeutic goals, he said.
Decrease Side Effects for Weight-Loss Success
“GI side effects are often the rate-limiting step in implementing an effective medication that patients want to take but may not be able to tolerate,” said Sean Wharton, MD, PharmD, medical director of the Wharton Medical Clinic for Weight and Diabetes Management, Burlington, Ontario, Canada, in an interview. “If we can decrease side effects, these medications could improve patients’ lives,” said Wharton, who was not involved in the study.
The improvement after a single dose of NG101 in patients receiving a single dose of semaglutide was impressive and in keeping with the mechanism of the drug action, said Wharton. “I was not surprised by the result but pleased that this single dose was shown to reduce the overall incidence of nausea and vomiting, the duration of nausea, the severity of nausea as rated by the study participants compared to placebo,” he said.
Ultimately, the clinical implications for NG101 are improved patient tolerance for GLP-1s and the ability to titrate and stay on them long term, incurring greater cardiometabolic benefit, Wharton told this news organization.
The current trial was limited to GLP1-1s on the market; newer medications may have fewer side effects, Wharton noted. “In clinical practice, patients often decrease the medication or titrate slower, and this could be the comparator,” he added.
The study was funded by Neurogastrx.
Wharton disclosed serving as a consultant for Neurogastrx but not as an investigator on the current study. He also reported having disclosed research on various GLP-1 medications.
A version of this article first appeared on Medscape.com.
A new drug currently known as NG101 reduced nausea and vomiting in patients with obesity using GLP-1s by 40% and 67%, respectively, based on data from a phase 2 trial presented at the Obesity Society’s Obesity Week 2025 in Atlanta.
Previous research published in JAMA Network Open showed a nearly 65% discontinuation rate for three GLP-1s (liraglutide, semaglutide, or tirzepatide) among adults with overweight or obesity and without type 2 diabetes. Gastrointestinal (GI) side effects topped the list of reasons for dropping the medications.
Given the impact of nausea and vomiting on discontinuation, there is an unmet need for therapies to manage GI symptoms, said Kimberley Cummings, PhD, of Neurogastrx, Inc., in her presentation.
In the new study, Cummings and colleagues randomly assigned 90 adults aged 18-55 years with overweight or obesity (defined as a BMI ranging from 22.0 to 35.0) to receive a single subcutaneous dose of semaglutide (0.5 mg) plus 5 days of NG101 at 20 mg twice daily, or a placebo.
NG101 is a peripherally acting D2 antagonist designed to reduce nausea and vomiting associated with GLP-1 use, Cummings said. NG101 targets the nausea center of the brain but is peripherally restricted to prevent central nervous system side effects, she explained.
Compared with placebo, NG101 significantly reduced the incidence of nausea and vomiting by 40% and 67%, respectively. Use of NG101 also was associated with a significant reduction in the duration of nausea and vomiting; GI events lasting longer than 1 day were reported in 22% and 51% of the NG101 patients and placebo patients, respectively.
In addition, participants who received NG101 reported a 70% decrease in nausea severity from baseline.
Overall, patients in the NG101 group also reported significantly fewer adverse events than those in the placebo group (74 vs 135), suggesting an improved safety profile when semaglutide is administered in conjunction with NG101, the researchers noted. No serious adverse events related to the study drug were reported in either group.
The findings were limited by several factors including the relatively small sample size. Additional research is needed with other GLP-1 agonists in larger populations with longer follow-up periods, Cummings said. However, the results suggest that NG101 was safe and effectively improved side effects associated with GLP-1 agonists.
“We know there are receptors for GLP-1 in the area postrema (nausea center of the brain), and that NG101 works on this area to reduce nausea and vomiting, so the study findings were not unexpected,” said Jim O’Mara, president and CEO of Neurogastrx, in an interview.
The study was a single-dose study designed to show proof of concept, and future studies would involve treating patients going through the recommended titration schedule for their GLP-1s, O’Mara said. However, NG101 offers an opportunity to keep more patients on GLP-1 therapy and help them reach their long-term therapeutic goals, he said.
Decrease Side Effects for Weight-Loss Success
“GI side effects are often the rate-limiting step in implementing an effective medication that patients want to take but may not be able to tolerate,” said Sean Wharton, MD, PharmD, medical director of the Wharton Medical Clinic for Weight and Diabetes Management, Burlington, Ontario, Canada, in an interview. “If we can decrease side effects, these medications could improve patients’ lives,” said Wharton, who was not involved in the study.
The improvement after a single dose of NG101 in patients receiving a single dose of semaglutide was impressive and in keeping with the mechanism of the drug action, said Wharton. “I was not surprised by the result but pleased that this single dose was shown to reduce the overall incidence of nausea and vomiting, the duration of nausea, the severity of nausea as rated by the study participants compared to placebo,” he said.
Ultimately, the clinical implications for NG101 are improved patient tolerance for GLP-1s and the ability to titrate and stay on them long term, incurring greater cardiometabolic benefit, Wharton told this news organization.
The current trial was limited to GLP1-1s on the market; newer medications may have fewer side effects, Wharton noted. “In clinical practice, patients often decrease the medication or titrate slower, and this could be the comparator,” he added.
The study was funded by Neurogastrx.
Wharton disclosed serving as a consultant for Neurogastrx but not as an investigator on the current study. He also reported having disclosed research on various GLP-1 medications.
A version of this article first appeared on Medscape.com.
FROM OBESITY WEEK 2025